User login
Cutis is a peer-reviewed clinical journal for the dermatologist, allergist, and general practitioner published monthly since 1965. Concise clinical articles present the practical side of dermatology, helping physicians to improve patient care. Cutis is referenced in Index Medicus/MEDLINE and is written and edited by industry leaders.
ass lick
assault rifle
balls
ballsac
black jack
bleach
Boko Haram
bondage
causas
cheap
child abuse
cocaine
compulsive behaviors
cost of miracles
cunt
Daech
display network stats
drug paraphernalia
explosion
fart
fda and death
fda AND warn
fda AND warning
fda AND warns
feom
fuck
gambling
gfc
gun
human trafficking
humira AND expensive
illegal
ISIL
ISIS
Islamic caliphate
Islamic state
madvocate
masturbation
mixed martial arts
MMA
molestation
national rifle association
NRA
nsfw
nuccitelli
pedophile
pedophilia
poker
porn
porn
pornography
psychedelic drug
recreational drug
sex slave rings
shit
slot machine
snort
substance abuse
terrorism
terrorist
texarkana
Texas hold 'em
UFC
section[contains(@class, 'nav-hidden')]
section[contains(@class, 'nav-hidden active')
A peer-reviewed, indexed journal for dermatologists with original research, image quizzes, cases and reviews, and columns.
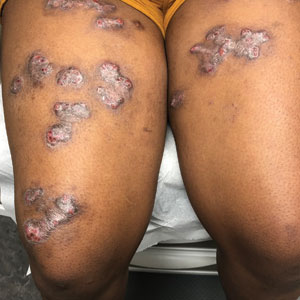
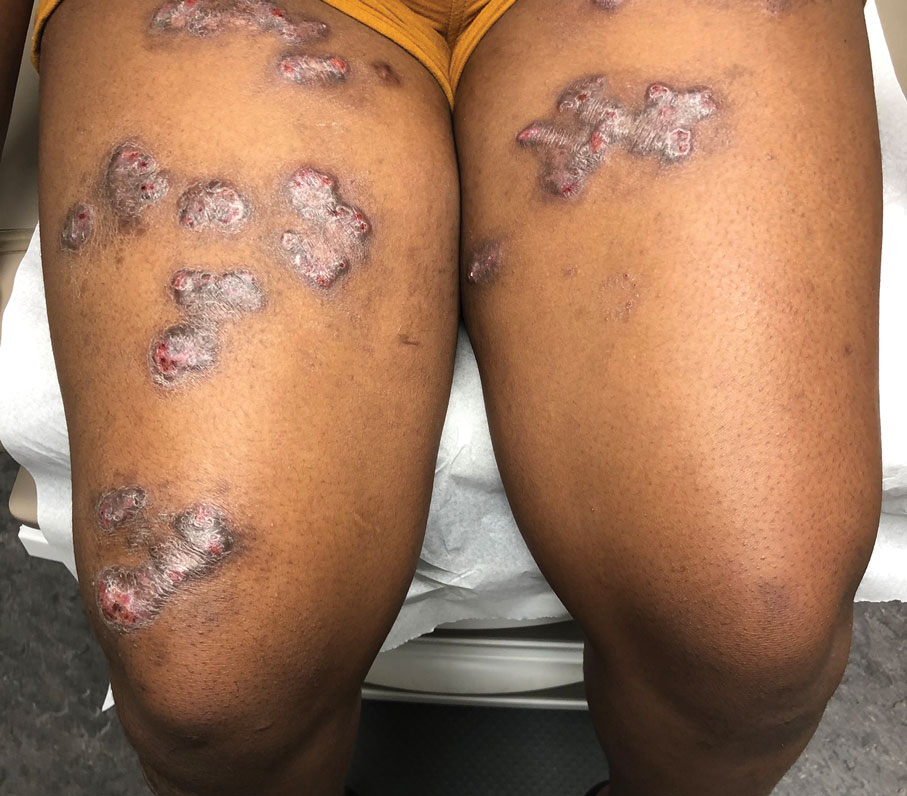
Spreading Painful Lesions on the Legs
The Diagnosis: Cutaneous Leishmaniasis
A punch biopsy of the skin showed pseudoepitheliomatous hyperplasia of the epidermis with dermal granulomatous and suppurative inflammation; tissue cultures remained sterile. Polymerase chain reaction testing of the skin revealed the presence of Leishmania guyanensis complex. Leishmaniasis is a widespread parasitic disease transmitted via sandflies that often is seen in children and young adults.1 Although leishmaniasis is endemic to several countries within Southeast Asia, East Africa, and Latin America, an increase in international travel has brought the disease to nonendemic regions. Therefore, it is crucial to obtain a detailed history of travel and exposure to sandflies in patients who have recently returned from endemic regions.
Leishmaniasis may present in 3 forms: cutaneous, mucocutaneous, or visceral. Cutaneous clinical findings vary depending on disease stage, causative species, and host immune activation. Presentation following a sandfly bite typically includes a papule that progresses to an erythematous nodule. Cutaneous leishmaniasis commonly occurs in areas of the body that are easily accessible to sandflies, such as the face, neck, and limbs. Mucocutaneous leishmaniasis presents with nasal or oral involvement several years after the onset of cutaneous leishmaniasis; however, it can coexist with cutaneous involvement. Without treatment, mucocutaneous leishmaniasis may lead to perforation of the nasal septum, destruction of the mouth, and life-threatening airway obstruction.1 Determining the specific species is important due to the variation in treatment options and prognosis. Because Leishmania organisms are fastidious, obtaining a positive culture often is challenging. Polymerase chain reaction can be utilized for identification, with detection rates of 97%.1 Systemic treatment is indicated for patients with multiple or large lesions; lesions on the hands, feet, face, or joints; or immunocompromised patients. Antimonial drugs are the first-line treatment for most forms of leishmaniasis, though increasing resistance has led to a decrease in efficacy.1 Our patient ultimately was treated with 4 weeks of miltefosine 50 mg 3 times daily. She obtained full resolution of the lesions with no further treatment indicated.
Pemphigus vegetans may present with various clinical manifestations that often can lead to a delay in diagnosis. The Hallopeau subtype typically presents as pustular lesions, while the Neumann subtype may present as large vesiculobullous erosive lesions that rupture and form verrucous, crusted, vegetative plaques. The groin, inguinal folds, axillae, thighs, and flexural areas commonly are affected, but reports of nasal, vaginal, and conjunctival involvement also exist.2
Granuloma inguinale is a sexually transmitted ulcerative disease that is caused by infection with Klebsiella granulomatis. It typically is found in tropical and subtropical climates, including Australia, Brazil, India, and South Africa. The initial presentation includes a single papule or multiple papules or nodules in the genital area that progress to a painless ulcer. It can be diagnosed via biopsies or tissue smears, which will demonstrate the presence of inclusion bodies known as Donovan bodies.3
Cutaneous tuberculosis (TB) can have variable clinical presentations and may be acquired exogenously or endogenously. Cutaneous TB can be divided into 2 categories: exogenous TB caused by inoculation and endogenous TB due to direct spread or autoinoculation. Exogenous TB subtypes include tuberculous chancre and TB verrucosa cutis, while endogenous TB includes scrofuloderma, orificial TB, and lupus vulgaris.4 Patches and plaques are found in patients with lupus vulgaris and TB verrucosa cutis. Scrofuloderma, tuberculous chancre, and orificial TB can present as ulcerative or erosive lesions. Cutaneous TB infection can be diagnosed through a smear, culture, or polymerase chain reaction.4
Deep cutaneous fungal infections most commonly present in immunocompromised individuals, particularly those who are severely neutropenic and are receiving broad-spectrum systemic antimicrobial agents. Deep cutaneous fungal infections initially present as a papule and evolve into a pustule followed by a necrotic ulcer. The lesions typically are accompanied by a fever and/or vital sign abnormalities.5
- Pace D. Leishmaniasis [published online September 17, 2014]. J Infect. 2014;69(suppl 1):S10-S18. doi:10.1016/j.jinf.2014.07.016
- Messersmith L, Krauland K. Pemphigus vegetans. StatPearls. StatPearls Publishing; 2022.
- Ornelas J, Kiuru M, Konia T, et al. Granuloma inguinale in a 51-year-old man. Dermatol Online J. 2016;22:13030/qt52k0c4hj.
- Chen Q, Chen W, Hao F. Cutaneous tuberculosis: a great imitator. Clin Dermatol. 2019;37:192-199.
- Marcoux D, Jafarian F, Joncas V, et al. Deep cutaneous fungal infections in immunocompromised children. J Am Acad Dermatol. 2009;61:857-864.
The Diagnosis: Cutaneous Leishmaniasis
A punch biopsy of the skin showed pseudoepitheliomatous hyperplasia of the epidermis with dermal granulomatous and suppurative inflammation; tissue cultures remained sterile. Polymerase chain reaction testing of the skin revealed the presence of Leishmania guyanensis complex. Leishmaniasis is a widespread parasitic disease transmitted via sandflies that often is seen in children and young adults.1 Although leishmaniasis is endemic to several countries within Southeast Asia, East Africa, and Latin America, an increase in international travel has brought the disease to nonendemic regions. Therefore, it is crucial to obtain a detailed history of travel and exposure to sandflies in patients who have recently returned from endemic regions.
Leishmaniasis may present in 3 forms: cutaneous, mucocutaneous, or visceral. Cutaneous clinical findings vary depending on disease stage, causative species, and host immune activation. Presentation following a sandfly bite typically includes a papule that progresses to an erythematous nodule. Cutaneous leishmaniasis commonly occurs in areas of the body that are easily accessible to sandflies, such as the face, neck, and limbs. Mucocutaneous leishmaniasis presents with nasal or oral involvement several years after the onset of cutaneous leishmaniasis; however, it can coexist with cutaneous involvement. Without treatment, mucocutaneous leishmaniasis may lead to perforation of the nasal septum, destruction of the mouth, and life-threatening airway obstruction.1 Determining the specific species is important due to the variation in treatment options and prognosis. Because Leishmania organisms are fastidious, obtaining a positive culture often is challenging. Polymerase chain reaction can be utilized for identification, with detection rates of 97%.1 Systemic treatment is indicated for patients with multiple or large lesions; lesions on the hands, feet, face, or joints; or immunocompromised patients. Antimonial drugs are the first-line treatment for most forms of leishmaniasis, though increasing resistance has led to a decrease in efficacy.1 Our patient ultimately was treated with 4 weeks of miltefosine 50 mg 3 times daily. She obtained full resolution of the lesions with no further treatment indicated.
Pemphigus vegetans may present with various clinical manifestations that often can lead to a delay in diagnosis. The Hallopeau subtype typically presents as pustular lesions, while the Neumann subtype may present as large vesiculobullous erosive lesions that rupture and form verrucous, crusted, vegetative plaques. The groin, inguinal folds, axillae, thighs, and flexural areas commonly are affected, but reports of nasal, vaginal, and conjunctival involvement also exist.2
Granuloma inguinale is a sexually transmitted ulcerative disease that is caused by infection with Klebsiella granulomatis. It typically is found in tropical and subtropical climates, including Australia, Brazil, India, and South Africa. The initial presentation includes a single papule or multiple papules or nodules in the genital area that progress to a painless ulcer. It can be diagnosed via biopsies or tissue smears, which will demonstrate the presence of inclusion bodies known as Donovan bodies.3
Cutaneous tuberculosis (TB) can have variable clinical presentations and may be acquired exogenously or endogenously. Cutaneous TB can be divided into 2 categories: exogenous TB caused by inoculation and endogenous TB due to direct spread or autoinoculation. Exogenous TB subtypes include tuberculous chancre and TB verrucosa cutis, while endogenous TB includes scrofuloderma, orificial TB, and lupus vulgaris.4 Patches and plaques are found in patients with lupus vulgaris and TB verrucosa cutis. Scrofuloderma, tuberculous chancre, and orificial TB can present as ulcerative or erosive lesions. Cutaneous TB infection can be diagnosed through a smear, culture, or polymerase chain reaction.4
Deep cutaneous fungal infections most commonly present in immunocompromised individuals, particularly those who are severely neutropenic and are receiving broad-spectrum systemic antimicrobial agents. Deep cutaneous fungal infections initially present as a papule and evolve into a pustule followed by a necrotic ulcer. The lesions typically are accompanied by a fever and/or vital sign abnormalities.5
The Diagnosis: Cutaneous Leishmaniasis
A punch biopsy of the skin showed pseudoepitheliomatous hyperplasia of the epidermis with dermal granulomatous and suppurative inflammation; tissue cultures remained sterile. Polymerase chain reaction testing of the skin revealed the presence of Leishmania guyanensis complex. Leishmaniasis is a widespread parasitic disease transmitted via sandflies that often is seen in children and young adults.1 Although leishmaniasis is endemic to several countries within Southeast Asia, East Africa, and Latin America, an increase in international travel has brought the disease to nonendemic regions. Therefore, it is crucial to obtain a detailed history of travel and exposure to sandflies in patients who have recently returned from endemic regions.
Leishmaniasis may present in 3 forms: cutaneous, mucocutaneous, or visceral. Cutaneous clinical findings vary depending on disease stage, causative species, and host immune activation. Presentation following a sandfly bite typically includes a papule that progresses to an erythematous nodule. Cutaneous leishmaniasis commonly occurs in areas of the body that are easily accessible to sandflies, such as the face, neck, and limbs. Mucocutaneous leishmaniasis presents with nasal or oral involvement several years after the onset of cutaneous leishmaniasis; however, it can coexist with cutaneous involvement. Without treatment, mucocutaneous leishmaniasis may lead to perforation of the nasal septum, destruction of the mouth, and life-threatening airway obstruction.1 Determining the specific species is important due to the variation in treatment options and prognosis. Because Leishmania organisms are fastidious, obtaining a positive culture often is challenging. Polymerase chain reaction can be utilized for identification, with detection rates of 97%.1 Systemic treatment is indicated for patients with multiple or large lesions; lesions on the hands, feet, face, or joints; or immunocompromised patients. Antimonial drugs are the first-line treatment for most forms of leishmaniasis, though increasing resistance has led to a decrease in efficacy.1 Our patient ultimately was treated with 4 weeks of miltefosine 50 mg 3 times daily. She obtained full resolution of the lesions with no further treatment indicated.
Pemphigus vegetans may present with various clinical manifestations that often can lead to a delay in diagnosis. The Hallopeau subtype typically presents as pustular lesions, while the Neumann subtype may present as large vesiculobullous erosive lesions that rupture and form verrucous, crusted, vegetative plaques. The groin, inguinal folds, axillae, thighs, and flexural areas commonly are affected, but reports of nasal, vaginal, and conjunctival involvement also exist.2
Granuloma inguinale is a sexually transmitted ulcerative disease that is caused by infection with Klebsiella granulomatis. It typically is found in tropical and subtropical climates, including Australia, Brazil, India, and South Africa. The initial presentation includes a single papule or multiple papules or nodules in the genital area that progress to a painless ulcer. It can be diagnosed via biopsies or tissue smears, which will demonstrate the presence of inclusion bodies known as Donovan bodies.3
Cutaneous tuberculosis (TB) can have variable clinical presentations and may be acquired exogenously or endogenously. Cutaneous TB can be divided into 2 categories: exogenous TB caused by inoculation and endogenous TB due to direct spread or autoinoculation. Exogenous TB subtypes include tuberculous chancre and TB verrucosa cutis, while endogenous TB includes scrofuloderma, orificial TB, and lupus vulgaris.4 Patches and plaques are found in patients with lupus vulgaris and TB verrucosa cutis. Scrofuloderma, tuberculous chancre, and orificial TB can present as ulcerative or erosive lesions. Cutaneous TB infection can be diagnosed through a smear, culture, or polymerase chain reaction.4
Deep cutaneous fungal infections most commonly present in immunocompromised individuals, particularly those who are severely neutropenic and are receiving broad-spectrum systemic antimicrobial agents. Deep cutaneous fungal infections initially present as a papule and evolve into a pustule followed by a necrotic ulcer. The lesions typically are accompanied by a fever and/or vital sign abnormalities.5
- Pace D. Leishmaniasis [published online September 17, 2014]. J Infect. 2014;69(suppl 1):S10-S18. doi:10.1016/j.jinf.2014.07.016
- Messersmith L, Krauland K. Pemphigus vegetans. StatPearls. StatPearls Publishing; 2022.
- Ornelas J, Kiuru M, Konia T, et al. Granuloma inguinale in a 51-year-old man. Dermatol Online J. 2016;22:13030/qt52k0c4hj.
- Chen Q, Chen W, Hao F. Cutaneous tuberculosis: a great imitator. Clin Dermatol. 2019;37:192-199.
- Marcoux D, Jafarian F, Joncas V, et al. Deep cutaneous fungal infections in immunocompromised children. J Am Acad Dermatol. 2009;61:857-864.
- Pace D. Leishmaniasis [published online September 17, 2014]. J Infect. 2014;69(suppl 1):S10-S18. doi:10.1016/j.jinf.2014.07.016
- Messersmith L, Krauland K. Pemphigus vegetans. StatPearls. StatPearls Publishing; 2022.
- Ornelas J, Kiuru M, Konia T, et al. Granuloma inguinale in a 51-year-old man. Dermatol Online J. 2016;22:13030/qt52k0c4hj.
- Chen Q, Chen W, Hao F. Cutaneous tuberculosis: a great imitator. Clin Dermatol. 2019;37:192-199.
- Marcoux D, Jafarian F, Joncas V, et al. Deep cutaneous fungal infections in immunocompromised children. J Am Acad Dermatol. 2009;61:857-864.
A 14-year-old adolescent girl presented with spreading painful lesions on the legs and left forearm of 2 years’ duration. Her travel history included several countries in South and Central America, traversing the Colombian jungle on foot. Near the end of the jungle trip, she noted a skin lesion on the left forearm around the site of an insect bite. Within 1 month, the lesions spread to the legs. She was treated with topical corticosteroids without improvement. Physical examination revealed verrucous, reddish-brown plaques on the legs and left forearm. Intranasal examination revealed a red rounded lesion inside the left nostril.
Epithelioma Cuniculatum (Plantar Verrucous Carcinoma): A Systematic Review of Treatment Options
Verrucous carcinoma (VC) is an uncommon type of well-differentiated squamous cell carcinoma (SCC) that most commonly affects men in the fifth to sixth decades of life. 1 The tumor grows slowly over a decade or more and does not frequently metastasize but has a high propensity for recurrence and local invasion. 2 There are 3 main subtypes of VC classified by anatomic site: oral florid papillomatosis (oral cavity), Buschke-Lowenstein tumor (anogenital region), and epithelioma cuniculatum (EC)(feet). 3 Epithelioma cuniculatum, also known as carcinoma cuniculatum or papillomatosis cutis carcinoides, most commonly presents as a solitary, warty or cauliflowerlike, exophytic mass with keratin-filled sinus tracts and malodorous discharge. 4 Diabetic foot ulcers and chronic inflammatory conditions are predisposing risk factors for EC, and it can result in difficulty walking/immobility, pain, and bleeding depending on anatomic involvement. 5-9
The differential diagnosis for VC includes refractory verruca vulgaris, clavus, SCC, keratoacanthoma, deep fungal or mycobacterial infection, eccrine poroma or porocarcinoma, amelanotic melanoma, and sarcoma.10-13 The slow-growing nature of VC, sampling error of superficial biopsies, and minimal cytological atypia on histologic examination can contribute to delayed diagnosis and appropriate treatment.14 Characteristic histologic features include hyperkeratosis, papillomatosis, marked acanthosis, broad blunt-ended rete ridges with a “bulldozing” architecture, and minimal cytologic atypia and mitoses.5,6 In some cases, pleomorphism and glassy eosinophilic cytoplasmic changes may be more pronounced than that of a common wart though less dramatic than that of conventional SCCs.15 Antigen Ki-67 and tumor protein p53 have been proposed to help differentiate between common plantar verruca, VC, and SCC, but the histologic diagnosis remains challenging, and repeat histopathologic examination often is required.16-19 Following diagnosis, computed tomography or magnetic resonance imaging may be necessary to determine tumor extension and assess for deep tissue and bony involvement.20-22
Treatment of EC is particularly challenging because of the anatomic location and need for margin control while maintaining adequate function, preserving healthy tissue, and providing coverage of defects. Surgical excision of EC is the first-line treatment, most commonly by wide local excision (WLE) or amputation. Mohs micrographic surgery (MMS) also has been utilized. One review found no recurrences in 5 cases of EC treated with MMS.23 As MMS is a tissue-sparing technique, this is a valuable modality for sites of functional importance such as the feet. Herein, we review various reported EC treatment modalities and outcomes, with an emphasis on recurrence rates for WLE and MMS.
METHODS
A systematic literature review of PubMed articles indexed for MEDLINE, as well as databases including the Cochrane Library, Web of Science, and Cumulative Index to Nursing and Allied Health Literature (CINAHL), was performed on January 14, 2020. Two authors (S.S.D. and S.V.C.) independently screened results using the search terms (plantar OR foot) AND (verrucous carcinoma OR epithelioma cuniculatum OR carcinoma cuniculatum). The search terms were chosen according to MeSH subject headings. All articles from the start date of the databases through the search date were screened, and articles pertaining to VC, EC, or carcinoma cuniculatum located on the foot were included. Of these, non–English-language articles were translated and included. Articles reporting VC on a site other than the foot (eg, the oral cavity) or benign verrucous skin lesions were excluded. The reference lists for all articles also were reviewed for additional reports that were absent from the initial search using both included and excluded articles. A full-text review was performed on 221 articles published between 1954 and 2019 per the PRISMA guidelines (Figure).
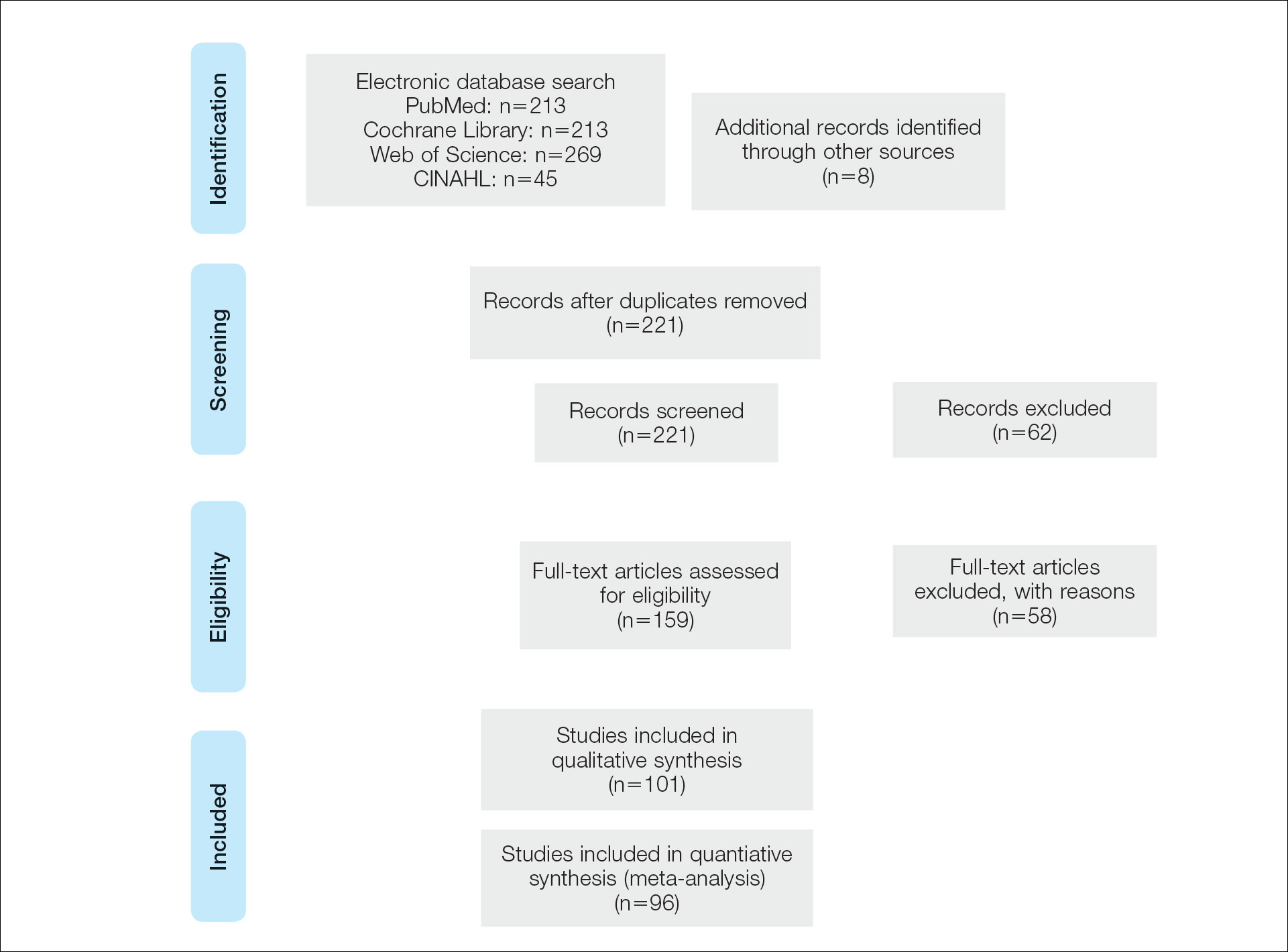
A total of 101 articles were included in the study for qualitative analysis. Nearly all articles identified were case reports, giving an evidence level of 5 by the Centre for Evidence-Based Medicine rating scale. Five articles reported data on multiple patients without individual demographic or clinical details and were excluded from analysis. Of the remaining 96 articles, information about patient characteristics, tumor size, treatment modality, and recurrence were extracted for 115 cases.
RESULTS
Of the 115 cases that were reviewed, 81 (70%) were male and 33 (29%) were female with a male-to-female ratio of 2.4:1. Ages of the patients ranged from 18 to 88 years; the mean and median age was 56 years. Nearly all reported cases of EC affected the plantar surface of one foot, with 4 reports of tumors affecting both feet.24-27 One case affecting both feet reported known exposure to lead arsenate pesticides27; all others were associated with a clinical history of chronic ulcers or warts persisting for several years to decades. Other less common sites of EC included the dorsal foot, interdigital web space, and subungual digit.28-30 The most common location reported was the anterior ball of the foot. Tumors were reported to arise within pre-existing lesions, such as hypertrophic lichen planus or chronic foot wounds associated with diabetes mellitus or leprosy.31-35 Tumor size ranged from 1 to 22 cm with a median of 4.5 cm.
Eight cases were reported to be associated with human papillomavirus; low-risk types 6 and 11 and high-risk types 16 and 18 were found in 6 cases.36-41 Two cases reported association with human papillomavirus type 2.7,42
Metastases to dermal and subdermal lymphatics, regional lymph nodes, and the lungs were reported in 3 cases, repectively.43-45 Of these, one primary tumor had received low-dose irradiation in the form of X-ray therapy.45
Treatment Modalities
The cases of EC that we reviewed included treatment with surgical and systemic therapies as well as other modalities such as acitretin, interferon alfa, topical imiquimod, curettage, debridement, electrodesiccation, and radiation. The Table includes a complete summary of the treatments we analyzed.
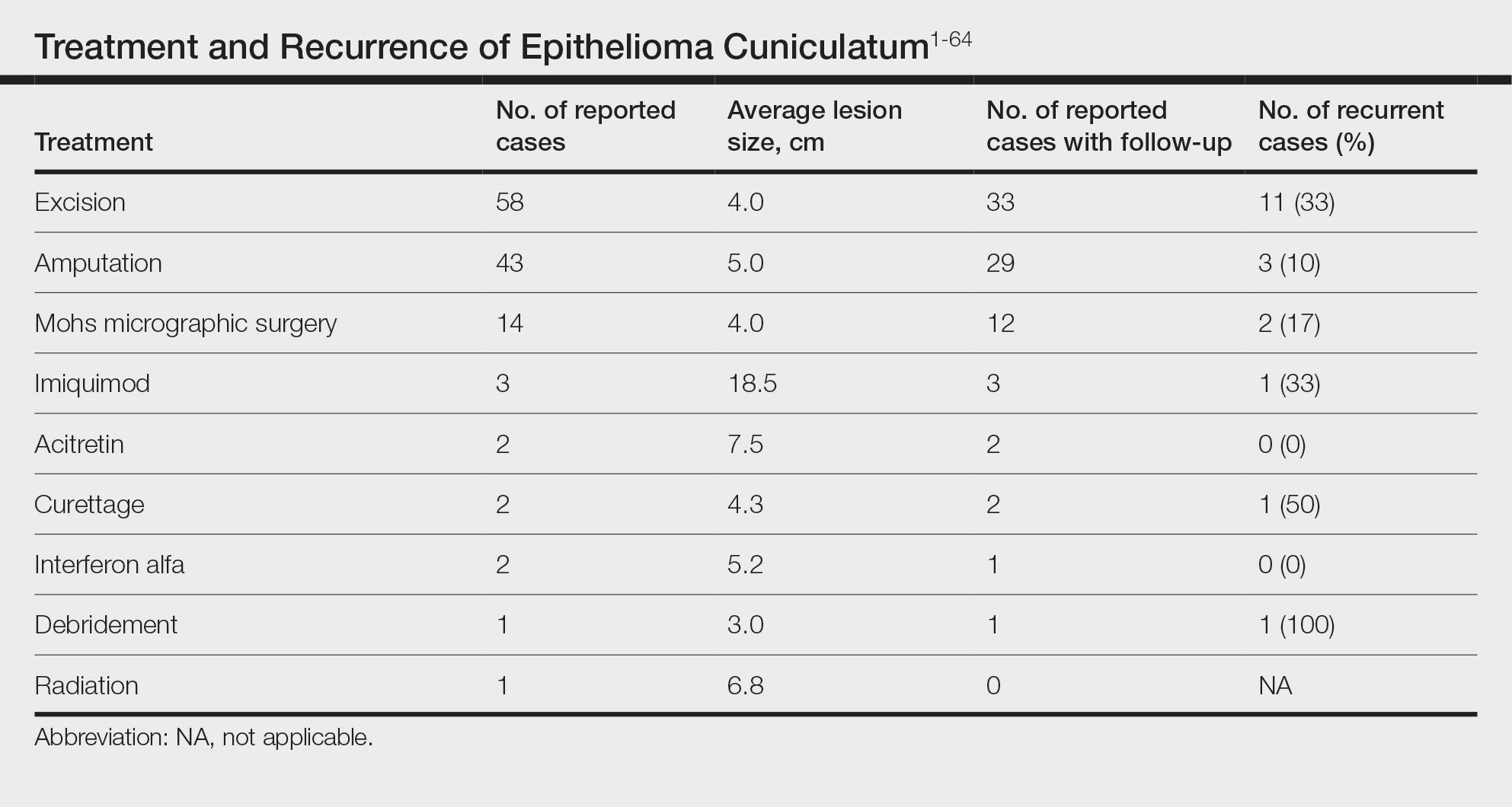
Surgical Therapy—The majority (91% [105/115]) of cases were treated surgically. The most common treatment modality was WLE (50% [58/115]), followed by amputation (37% [43/115]) and MMS (12% [14/115]).
Wide local excision was the most frequently reported treatment, with excision margins of at least 5 mm to 1 cm.48 Incidence of recurrence was reported for 57% (33/58) of cases treated with WLE; of these, the recurrence rate was 33% (11/33). For patients with EC recurrence, the most common secondary treatment was repeat excision with wider margins (1–2 cm) or amputation (5/11).49-52 Few postoperative complications were reported but included pain, infection, and difficulty walking, which were mostly associated with repair modality (eg, split-thickness skin grafts, rotational flaps).53 Amputation was the second most common treatment modality, with a 67% (29/43) incidence of recurrence. Types of amputation included transmetatarsal ray amputation (7/43 [16%]), foot or forefoot amputation (2/43 [5%]), above-the-knee amputation (1/43 [2%]), and below-the-knee amputation (1/43 [2%]). Complications associated with amputation included infection and requirement of prosthetics for ambulation. Split-thickness skin grafts and rotational flaps were the most common surgical repairs performed.52,53
Mohs micrographic surgery was the least frequently reported surgical treatment modality. Both traditional MMS on fresh tissue and “slow Mohs,” with formalin-fixed paraffin embedded tissue examination over several days, were performed for EC with horizontal en face sectioning.54-56 Incidence of recurrence was reported for 86% (12/14) of MMS cases. Of these, recurrence was seen in 17% (2/12) that utilized a flat horizontal processing of tissue sections coupled with saucerlike excisions to enable examination of the entire undersurface and margins. In one case, the patient was treated with MMS with recurrence noted 1 month later; thus, repeat MMS was performed, and the tumor was found to be entwined around the flexor tendon.57 The tendon was removed, and clear margins were obtained. Follow-up 3 years after the second MMS revealed no signs of recurrence.57 In the other case, the patient had a particularly aggressive course with bilateral VC in the setting of diabetic ulcers that was treated with WLE prior to MMS and recurrence still noted after MMS.26 No complications were reported with MMS.
Overall, recurrence was most frequently reported with WLE (11/33 [33%]), followed by MMS (2/12 [17%]) and amputation (3/29 [10%]). When comparing WLE and amputation, the relationship between treatment modality and recurrence was statistically significant using a χ2 test of independence (χ2=4.7; P=.03). However, results were not significant with Yates correction for continuity (χ2=3.4; P=.06). The χ2 test of independence showed no significant association between treatment method and recurrence when comparing WLE with MMS (χ2=1.2; P=.28). Reported follow-up times varied greatly from a few months to 10 years.
Systemic Therapy—Of the total cases, only 2 cases reported treatment with acitretin and 2 utilized interferon alfa.58,59 In one case, treatment of EC with interferon alfa alone required more aggressive therapy (ie, amputation).58 Neither of the 2 cases using acitretin reported recurrence.59,60 Complications of acitretin therapy included cheilitis and transaminitis.60
Other Treatment Modalities—Three cases utilized imiquimod, with 2 cases of imiquimod monotherapy and 1 case of imiquimod in combination with electrodesiccation and WLE.37 One of the cases of EC treated with imiquimod monotherapy recurred and required WLE.61
There were reports of other treatments including curettage alone (2% [2/115]),40,62 debridement alone (1% [1/115]),40 electrodesiccation (1% [1/115]),37 and radiation (1% [1/115]).43 Recurrence was found with curettage alone and debridement alone. Electrodesiccation was reported in conjunction with WLE without recurrence. Radiation was used to treat a case of VC that had metastasized to the lymph nodes; no follow-up was described.43
COMMENT
Epithelioma cuniculatum is an indolent malignancy of the plantar foot that likely is frequently underdiagnosed or misdiagnosed because of location, sampling error, and challenges in histopathologic diagnosis. Once diagnosed, surgical removal with margin control is the first-line therapy for EC. Our review found a number of surgical, systemic, and other treatment modalities that have been used to treat EC, but there remains a lack of evidence to provide clear guidelines as to which therapies are most effective. Current data on the treatment of EC largely are limited to case reports and case series. To date, there are no reports of higher-quality studies or randomized controlled trials to assess the efficacy of various treatment modalities.
Our review found that WLE is the most common treatment modality for EC, followed by amputation and MMS. Three cases43-45 that reported metastasis to lymph nodes also were treated with fine-needle aspiration or biopsy, and it is recommended that sentinel lymph node biopsy be performed when there is a history of radiation exposure or clinically and sonographically unsuspicious lymph nodes, while dissection of regional nodes should be performed if lymph node metastasis is suspected.53 Additional treatments reported included acitretin, interferon alfa, topical imiquimod, curettage, debridement, and electrodesiccation, but because of the limited number of cases and variable efficacy, no conclusions can be made on the utility of these alternative modalities.
The lowest rate of reported recurrence was found with amputation, followed by MMS and WLE. Amputation is the most aggressive treatment option, but its superiority in lower recurrence rates was not statistically significant when compared with either WLE or MMS after Yates correction. Despite treatment with radical surgery, recurrence is still possible and may be associated with factors including greater size (>2 cm) and depth (>4 mm), poor histologic differentiation, perineural involvement, failure of previous treatments, and immunosuppression.63 No statistically significant difference in recurrence rates was found among surgical methods, though data trended toward lower rates of recurrence with MMS compared with WLE, as recurrence with MMS was only reported in 2 cases.25,56
The efficacy of MMS is well documented for tumors with contiguous growth and enables maximum preservation of normal tissue structure and function with complete margin visualization. Thus, our results are in agreement with those of prior studies,54-56,64 suggesting that MMS is associated with lower recurrence rates for EC than WLE. Future studies and reporting of MMS for EC are particularly important because of the functional importance of the plantar foot.
It is important to note that there are local and systemic risk factors that increase the likelihood of developing EC and facilitate tumor growth, including antecedent trauma to the lesion site, chronic irritation or infection, and immunosuppression (HIV related or iatrogenic medication induced). These risk factors may play a role in the treatment modality utilized (eg, more aggressive EC may be treated with amputation instead of WLE). Underlying patient comorbidities could potentially affect recurrence rates, which is a variable we could not control for in our analysis.
Our findings are limited by study design, with supporting evidence consisting of case reports and series. The review is limited by interstudy variability and heterogeneity of results. Additionally, recurrence is not reported in all cases and may be a source of sampling bias. Further complicating the generalizability of these results is the lack of follow-up to evaluate morbidity and quality of life after treatment.
CONCLUSION
This review suggests that MMS is associated with lower recurrence rates than WLE for the treatment of EC. Further investigation of MMS for EC with appropriate follow-up is necessary to identify whether MMS is associated with lower recurrence and less functional impairment. Nonsurgical treatments, including topical imiquimod, interferon alfa, and acitretin, may be useful in cases where surgical therapies are contraindicated, but there is little evidence to support these treatment modalities. Treatment guidelines for EC are not established, and appropriate treatment guidelines should be developed in the future.
- McKee PH, Wilkinson JD, Black MM, et al. Carcinoma (epithelioma) cuniculatum: a clinicopathological study of nineteen cases and review of the literature. Histopathology. 1981;5:425-436.
- Aird I, Johnson HD, Lennox B, et al. Epithelioma cuniculatum: a variety of squamous carcinoma peculiar to the foot. Br J Surg. 1954;42:245-250.
- Seremet S, Erdemir AT, Kiremitci U, et al. Unusually early-onset plantar verrucous carcinoma. Cutis. 2019;104:34-36.
- Spyriounis PK, Tentis D, Sparveri IF, et al. Plantar epithelioma cuniculatum. a case report with review of the literature. Eur J Plast Surg. 2004;27:253-256.
- Ho J, Diven G, Bu J, et al. An ulcerating verrucous plaque on the foot. verrucous carcinoma (epithelioma cuniculatum). Arch Dermatol. 2000;136:547-548, 550-551.
- Kao GF, Graham JH, Helwig EB. Carcinoma cuniculatum (verrucous carcinoma of the skin): a clinicopathologic study of 46 cases with ultrastructural observations. Cancer. 1982;49:2395-2403.
- Zielonka E, Goldschmidt D, de Fontaine S. Verrucous carcinoma or epithelioma cuniculatum plantare. Eur J Surg Oncol. 1997;23:86-87.
- Dogan G, Oram Y, Hazneci E, et al. Three cases of verrucous carcinoma. Australas J Dermatol. 1998;39:251-254.
- Schwartz RA, Burgess GH. Verrucous carcinoma of the foot. J Surg Oncol. 1980;14:333-339.
- McKay C, McBride P, Muir J. Plantar verrucous carcinoma masquerading as toe web intertrigo. Australas J Dermatol. 2012;53:2010-2012.
- Shenoy AS, Waghmare RS, Kavishwar VS, et al. Carcinoma cuniculatum of foot. Foot. 2011;21:207-208.
- Lozzi G, Perris K. Carcinoma cuniculatum. CMAJ. 2007;177:249-251.
- Schein O, Orenstein A, Bar-Meir E. Plantar verrucous carcicoma (epithelioma cuniculatum): rare form of the common wart. Isr Med Assoc J. 2006;8:885.
- Rheingold LM, Roth LM. Carcinoma of the skin of the foot exhibiting some verrucous features. Plast Reconstr Surg. 1978;61:605-609.
- Klima M, Kurtis B, Jordan PH. Verrucous carcinoma of skin. J Cutan Pathol. 1980;7:88-98.
- Nakamura Y, Kashiwagi K, Nakamura A, et al. Verrucous carcinoma of the foot diagnosed using p53 and Ki-67 immunostaining in a patient with diabetic neuropathy. Am J Dermatopathol. 2015;37:257-259.
- Costache M, Desa LT, Mitrache LE, et al. Cutaneous verrucous carcinoma—report of three cases with review of literature. Rom J Morphol Embryol. 2014;55:383-388.
- Terada T. Verrucous carcinoma of the skin: a report on 5 Japanese cases. Ann Diagn Pathol. 2011;15:175-180.
- Noel JC, Heenen M, Peny MO, et al. Proliferating cell nuclear antigen distribution in verrucous carcinoma of the skin. Br J Dermatol. 1995;133:868-873.
- García-Gavín J, González-Vilas D, Rodríguez-Pazos L, et al. Verrucous carcinoma of the foot affecting the bone: utility of the computed tomography scanner. Dermatol Online J. 2010;16:3-5.
- Wasserman PL, Taylor RC, Pinillia J, et al. Verrucous carcinoma of the foot and enhancement assessment by MRI. Skeletal Radiol. 2009;38:393-395.
- Bhushan MH, Ferguson JE, Hutchinson CE. Carcinoma cuniculatum of the foot assessed by magnetic resonance scanning. Clin Exp Dermatol. 2001;26:419-422.
- Penera KE, Manji KA, Craig AB, et al. Atypical presentation of verrucous carcinoma: a case study and review of the literature. Foot Ankle Spec. 2013;6:318-322.
- Suen K, Wijeratne S, Patrikios J. An unusual case of bilateral verrucous carcinoma of the foot (epithelioma cuniculatum). J Surg Case Rep. 2012;2012:rjs020.
- Riccio C, King K, Elston JB, et al. Bilateral plantar verrucous carcinoma. Eplasty. 2016;16:ic46.
- Di Palma V, Stone JP, Schell A, et al. Mistaken diabetic ulcers: a case of bilateral foot verrucous carcinoma. Case Rep Dermatol Med. 2018;2018:4192657.
- Seehafer JR, Muller SA, Dicken CH. Bilateral verrucous carcinoma of the feet. Orthop Surv. 1979;3:205.
- Tosti A, Morelli R, Fanti PA, et al. Carcinoma cuniculatum of the nail apparatus: report of three cases. Dermatology. 1993;186:217-221.
- Melo CR, Melo IS, Souza LP. Epithelioma cuniculatum, a verrucous carcinoma of the foot. report of 2 cases. Dermatologica. 1981;163:338-342.
- Van Geertruyden JP, Olemans C, Laporte M, et al. Verrucous carcinoma of the nail bed. Foot Ankle Int. 1998;19:327-328.
- Thakur BK, Verma S, Raphael V. Verrucous carcinoma developing in a long standing case of ulcerative lichen planus of sole: a rare case report. J Eur Acad Dermatol Venereol. 2015;29:399-401.
- Mayron R, Grimwood RE, Siegle RJ, et al. Verrucous carcinoma arising in ulcerative lichen planus of the soles. J Dermatol Surg Oncol. 1988;14:547-551.
- Boussofara L, Belajouza-Noueiri C, Ghariani N, et al. Verrucous epidermoid carcinoma as a complication in cutaneous lichen planus [article in French]. Ann Dermatol Venereol. 2006;133:404-405.
- Khullar G, Mittal S, Sharma S. Verrucous carcinoma on the foot arising in a chronic neuropathic ulcer of leprosy. Australas J Dermatol. 2019;60:245-246.
- Ochsner PE, Hausman R, Olsthoorn PGM. Epithelioma cunicalutum developing in a neuropathic ulcer of leprous etiology. Arch Orthop Trauma Surg. 1979;94:227-231.
- Ray R, Bhagat A, Vasudevan B, et al. A rare case of plantar epithelioma cuniculatum arising from a wart. Indian J Dermatol. 2015;60:485-487.
- Imko-Walczuk B, Cegielska A, Placek W, et al. Human papillomavirus-related verrucous carcinoma in a renal transplant patient after long-term immunosuppression: a case report. Transplant Proc. 2014;46:2916-2919.
- Floristán MU, Feltes RA, Sáenz JC, et al. Verrucous carcinoma of the foot associated with human papillomavirus type 18. Actas Dermosifiliogr. 2009;100:433-435.
- Sasaoka R, Morimura T, Mihara M, et al. Detection of human pupillomavirus type 16 DNA in two cases of verriicous carcinoma of the foot. Br J Dermatol. 1996;134:983984.
- Schell BJ, Rosen T, Rády P, et al. Verrucous carcinoma of the foot associated with human papillomavirus type 16. J Am Acad Dermatol. 2001;45:49-55.
- Knobler RM, Schneider S, Neumann RA, et al. DNA dot‐blot hybridization implicates human papillomavirus type 11‐DNA in epithelioma cuniculatum. J Med Virol. 1989;29:33-37.
- Noel JC, Peny MO, Detremmerie O, et al. Demonstration of human papillomavirus type 2 in a verrucous carcinoma of the foot. Dermatology. 1993;187:58-61.
- Jungmann J, Vogt T, Müller CSL. Giant verrucous carcinoma of the lower extremity in women with dementia. BMJ Case Rep. 2012;2012:bcr2012006357.
- McKee PH, Wilkinson JD, Corbett MF, et al. Carcinoma cuniculatum: a case metastasizing to skin and lymph nodes. Clin Exp Dermatol. 1981;6:613-618.
- Owen WR, Wolfe ID, Burnett JW, et al. Epithelioma cuniculatum. South Med J. 1978;71:477-479.
- Patel AN, Bedforth N, Varma S. Pain-free treatment of carcinoma cuniculatum on the heel using Mohs micrographic surgery and ultrasonography-guided sciatic nerve block. Clin Exp Dermatol. 2013;38:569-571.
- Padilla RS, Bailin PL, Howard WR, et al. Verrucous carcinoma of the skin and its management by Mohs’ surgery. Plast Reconstr Surg. 1984;73:442-447.
- Kotwal M, Poflee S, Bobhate S. Carcinoma cuniculatum at various anatomical sites. Indian J Dermatol. 2005;50:216-220.
- Arefi M, Philipone E, Caprioli R, et al. A case of verrucous carcinoma (epithelioma cuniculatum) of the heel mimicking infected epidermal cyst and gout. Foot Ankle Spec. 2008;1:297-299.
- Trebing D, Brunner M, Kröning Y, et al. Young man with verrucous heel tumor [article in German]. J Dtsch Dermatol Ges. 2003;9:739-741.
- Thompson SG. Epithelioma cuniculatum: an unusual tumour of the foot. Br J Plast Surg. 1965;18:214-217.
- Thomas EJ, Graves NC, Meritt SM. Carcinoma cuniculatum: an atypical presentation in the foot. J Foot Ankle Surg. 2014;53:356-359.
- Koch H, Kowatsch E, Hödl S, et al. Verrucous carcinoma of the skin: long-term follow-up results following surgical therapy. Dermatol Surg. 2004;30:1124-1130.
- Mallatt BD, Ceilley RI, Dryer RF. Management of verrucous carcinoma on a foot by a combination of chemosurgery and plastic repair: report of a case. J Dermatol Surg Oncol. 1980;6:532-534.
- Mohs FE, Sahl WJ. Chemosurgery for verrucous carcinoma. J Dermatol Surg Oncol. 1979;5:302-306.
- Alkalay R, Alcalay J, Shiri J. Plantar verrucous carcinoma treated with Mohs micrographic surgery: a case report and literature review. J Drugs Dermatol. 2006;5:68-73.
- Mora RG. Microscopically controlled surgery (Mohs’ chemosurgery) for treatment of verrucous squamous cell carcinoma of the foot (epithelioma cuniculatum). J Am Acad Dermatol. 1983;8:354-362.
- Risse L, Negrier P, Dang PM, et al. Treatment of verrucous carcinoma with recombinant alfa-interferon. Dermatology. 1995;190:142-144.
- Rogozin´ski TT, Schwartz RA, Towpik E. Verrucous carcinoma in Unna-Thost hyperkeratosis of the palms and soles. J Am Acad Dermatol. 1994;31:1061-1062.
- Kuan YZ, Hsu HC, Kuo TT, et al. Multiple verrucous carcinomas treated with acitretin. J Am Acad Dermatol. 2007;56(2 suppl):S29-S32.
- Schalock PC, Kornik RI, Baughman RD, et al. Treatment of verrucous carcinoma with topical imiquimod. J Am Acad Dermatol. 2006;54:233-234.
- Brown SM, Freeman RG. Epithelioma cuniculatum. Arch Dermatol. 1976;112:1295-1296.
- Rowe DE, Carroll RJ, Day CL, et al. Prognostic factors for local recurrence, metastasis, and survival rates in squamous cell carcinoma of the skin, ear, and lip. J Am Acad Dermatol. 1992;26:976-990.
- Swanson NA, Taylor WB. Plantar verrucous carcinoma: literature review and treatment by the Mohs’ chemosurgery technique. Arch Dermatol. 1980;116:794-797.
Verrucous carcinoma (VC) is an uncommon type of well-differentiated squamous cell carcinoma (SCC) that most commonly affects men in the fifth to sixth decades of life. 1 The tumor grows slowly over a decade or more and does not frequently metastasize but has a high propensity for recurrence and local invasion. 2 There are 3 main subtypes of VC classified by anatomic site: oral florid papillomatosis (oral cavity), Buschke-Lowenstein tumor (anogenital region), and epithelioma cuniculatum (EC)(feet). 3 Epithelioma cuniculatum, also known as carcinoma cuniculatum or papillomatosis cutis carcinoides, most commonly presents as a solitary, warty or cauliflowerlike, exophytic mass with keratin-filled sinus tracts and malodorous discharge. 4 Diabetic foot ulcers and chronic inflammatory conditions are predisposing risk factors for EC, and it can result in difficulty walking/immobility, pain, and bleeding depending on anatomic involvement. 5-9
The differential diagnosis for VC includes refractory verruca vulgaris, clavus, SCC, keratoacanthoma, deep fungal or mycobacterial infection, eccrine poroma or porocarcinoma, amelanotic melanoma, and sarcoma.10-13 The slow-growing nature of VC, sampling error of superficial biopsies, and minimal cytological atypia on histologic examination can contribute to delayed diagnosis and appropriate treatment.14 Characteristic histologic features include hyperkeratosis, papillomatosis, marked acanthosis, broad blunt-ended rete ridges with a “bulldozing” architecture, and minimal cytologic atypia and mitoses.5,6 In some cases, pleomorphism and glassy eosinophilic cytoplasmic changes may be more pronounced than that of a common wart though less dramatic than that of conventional SCCs.15 Antigen Ki-67 and tumor protein p53 have been proposed to help differentiate between common plantar verruca, VC, and SCC, but the histologic diagnosis remains challenging, and repeat histopathologic examination often is required.16-19 Following diagnosis, computed tomography or magnetic resonance imaging may be necessary to determine tumor extension and assess for deep tissue and bony involvement.20-22
Treatment of EC is particularly challenging because of the anatomic location and need for margin control while maintaining adequate function, preserving healthy tissue, and providing coverage of defects. Surgical excision of EC is the first-line treatment, most commonly by wide local excision (WLE) or amputation. Mohs micrographic surgery (MMS) also has been utilized. One review found no recurrences in 5 cases of EC treated with MMS.23 As MMS is a tissue-sparing technique, this is a valuable modality for sites of functional importance such as the feet. Herein, we review various reported EC treatment modalities and outcomes, with an emphasis on recurrence rates for WLE and MMS.
METHODS
A systematic literature review of PubMed articles indexed for MEDLINE, as well as databases including the Cochrane Library, Web of Science, and Cumulative Index to Nursing and Allied Health Literature (CINAHL), was performed on January 14, 2020. Two authors (S.S.D. and S.V.C.) independently screened results using the search terms (plantar OR foot) AND (verrucous carcinoma OR epithelioma cuniculatum OR carcinoma cuniculatum). The search terms were chosen according to MeSH subject headings. All articles from the start date of the databases through the search date were screened, and articles pertaining to VC, EC, or carcinoma cuniculatum located on the foot were included. Of these, non–English-language articles were translated and included. Articles reporting VC on a site other than the foot (eg, the oral cavity) or benign verrucous skin lesions were excluded. The reference lists for all articles also were reviewed for additional reports that were absent from the initial search using both included and excluded articles. A full-text review was performed on 221 articles published between 1954 and 2019 per the PRISMA guidelines (Figure).

A total of 101 articles were included in the study for qualitative analysis. Nearly all articles identified were case reports, giving an evidence level of 5 by the Centre for Evidence-Based Medicine rating scale. Five articles reported data on multiple patients without individual demographic or clinical details and were excluded from analysis. Of the remaining 96 articles, information about patient characteristics, tumor size, treatment modality, and recurrence were extracted for 115 cases.
RESULTS
Of the 115 cases that were reviewed, 81 (70%) were male and 33 (29%) were female with a male-to-female ratio of 2.4:1. Ages of the patients ranged from 18 to 88 years; the mean and median age was 56 years. Nearly all reported cases of EC affected the plantar surface of one foot, with 4 reports of tumors affecting both feet.24-27 One case affecting both feet reported known exposure to lead arsenate pesticides27; all others were associated with a clinical history of chronic ulcers or warts persisting for several years to decades. Other less common sites of EC included the dorsal foot, interdigital web space, and subungual digit.28-30 The most common location reported was the anterior ball of the foot. Tumors were reported to arise within pre-existing lesions, such as hypertrophic lichen planus or chronic foot wounds associated with diabetes mellitus or leprosy.31-35 Tumor size ranged from 1 to 22 cm with a median of 4.5 cm.
Eight cases were reported to be associated with human papillomavirus; low-risk types 6 and 11 and high-risk types 16 and 18 were found in 6 cases.36-41 Two cases reported association with human papillomavirus type 2.7,42
Metastases to dermal and subdermal lymphatics, regional lymph nodes, and the lungs were reported in 3 cases, repectively.43-45 Of these, one primary tumor had received low-dose irradiation in the form of X-ray therapy.45
Treatment Modalities
The cases of EC that we reviewed included treatment with surgical and systemic therapies as well as other modalities such as acitretin, interferon alfa, topical imiquimod, curettage, debridement, electrodesiccation, and radiation. The Table includes a complete summary of the treatments we analyzed.

Surgical Therapy—The majority (91% [105/115]) of cases were treated surgically. The most common treatment modality was WLE (50% [58/115]), followed by amputation (37% [43/115]) and MMS (12% [14/115]).
Wide local excision was the most frequently reported treatment, with excision margins of at least 5 mm to 1 cm.48 Incidence of recurrence was reported for 57% (33/58) of cases treated with WLE; of these, the recurrence rate was 33% (11/33). For patients with EC recurrence, the most common secondary treatment was repeat excision with wider margins (1–2 cm) or amputation (5/11).49-52 Few postoperative complications were reported but included pain, infection, and difficulty walking, which were mostly associated with repair modality (eg, split-thickness skin grafts, rotational flaps).53 Amputation was the second most common treatment modality, with a 67% (29/43) incidence of recurrence. Types of amputation included transmetatarsal ray amputation (7/43 [16%]), foot or forefoot amputation (2/43 [5%]), above-the-knee amputation (1/43 [2%]), and below-the-knee amputation (1/43 [2%]). Complications associated with amputation included infection and requirement of prosthetics for ambulation. Split-thickness skin grafts and rotational flaps were the most common surgical repairs performed.52,53
Mohs micrographic surgery was the least frequently reported surgical treatment modality. Both traditional MMS on fresh tissue and “slow Mohs,” with formalin-fixed paraffin embedded tissue examination over several days, were performed for EC with horizontal en face sectioning.54-56 Incidence of recurrence was reported for 86% (12/14) of MMS cases. Of these, recurrence was seen in 17% (2/12) that utilized a flat horizontal processing of tissue sections coupled with saucerlike excisions to enable examination of the entire undersurface and margins. In one case, the patient was treated with MMS with recurrence noted 1 month later; thus, repeat MMS was performed, and the tumor was found to be entwined around the flexor tendon.57 The tendon was removed, and clear margins were obtained. Follow-up 3 years after the second MMS revealed no signs of recurrence.57 In the other case, the patient had a particularly aggressive course with bilateral VC in the setting of diabetic ulcers that was treated with WLE prior to MMS and recurrence still noted after MMS.26 No complications were reported with MMS.
Overall, recurrence was most frequently reported with WLE (11/33 [33%]), followed by MMS (2/12 [17%]) and amputation (3/29 [10%]). When comparing WLE and amputation, the relationship between treatment modality and recurrence was statistically significant using a χ2 test of independence (χ2=4.7; P=.03). However, results were not significant with Yates correction for continuity (χ2=3.4; P=.06). The χ2 test of independence showed no significant association between treatment method and recurrence when comparing WLE with MMS (χ2=1.2; P=.28). Reported follow-up times varied greatly from a few months to 10 years.
Systemic Therapy—Of the total cases, only 2 cases reported treatment with acitretin and 2 utilized interferon alfa.58,59 In one case, treatment of EC with interferon alfa alone required more aggressive therapy (ie, amputation).58 Neither of the 2 cases using acitretin reported recurrence.59,60 Complications of acitretin therapy included cheilitis and transaminitis.60
Other Treatment Modalities—Three cases utilized imiquimod, with 2 cases of imiquimod monotherapy and 1 case of imiquimod in combination with electrodesiccation and WLE.37 One of the cases of EC treated with imiquimod monotherapy recurred and required WLE.61
There were reports of other treatments including curettage alone (2% [2/115]),40,62 debridement alone (1% [1/115]),40 electrodesiccation (1% [1/115]),37 and radiation (1% [1/115]).43 Recurrence was found with curettage alone and debridement alone. Electrodesiccation was reported in conjunction with WLE without recurrence. Radiation was used to treat a case of VC that had metastasized to the lymph nodes; no follow-up was described.43
COMMENT
Epithelioma cuniculatum is an indolent malignancy of the plantar foot that likely is frequently underdiagnosed or misdiagnosed because of location, sampling error, and challenges in histopathologic diagnosis. Once diagnosed, surgical removal with margin control is the first-line therapy for EC. Our review found a number of surgical, systemic, and other treatment modalities that have been used to treat EC, but there remains a lack of evidence to provide clear guidelines as to which therapies are most effective. Current data on the treatment of EC largely are limited to case reports and case series. To date, there are no reports of higher-quality studies or randomized controlled trials to assess the efficacy of various treatment modalities.
Our review found that WLE is the most common treatment modality for EC, followed by amputation and MMS. Three cases43-45 that reported metastasis to lymph nodes also were treated with fine-needle aspiration or biopsy, and it is recommended that sentinel lymph node biopsy be performed when there is a history of radiation exposure or clinically and sonographically unsuspicious lymph nodes, while dissection of regional nodes should be performed if lymph node metastasis is suspected.53 Additional treatments reported included acitretin, interferon alfa, topical imiquimod, curettage, debridement, and electrodesiccation, but because of the limited number of cases and variable efficacy, no conclusions can be made on the utility of these alternative modalities.
The lowest rate of reported recurrence was found with amputation, followed by MMS and WLE. Amputation is the most aggressive treatment option, but its superiority in lower recurrence rates was not statistically significant when compared with either WLE or MMS after Yates correction. Despite treatment with radical surgery, recurrence is still possible and may be associated with factors including greater size (>2 cm) and depth (>4 mm), poor histologic differentiation, perineural involvement, failure of previous treatments, and immunosuppression.63 No statistically significant difference in recurrence rates was found among surgical methods, though data trended toward lower rates of recurrence with MMS compared with WLE, as recurrence with MMS was only reported in 2 cases.25,56
The efficacy of MMS is well documented for tumors with contiguous growth and enables maximum preservation of normal tissue structure and function with complete margin visualization. Thus, our results are in agreement with those of prior studies,54-56,64 suggesting that MMS is associated with lower recurrence rates for EC than WLE. Future studies and reporting of MMS for EC are particularly important because of the functional importance of the plantar foot.
It is important to note that there are local and systemic risk factors that increase the likelihood of developing EC and facilitate tumor growth, including antecedent trauma to the lesion site, chronic irritation or infection, and immunosuppression (HIV related or iatrogenic medication induced). These risk factors may play a role in the treatment modality utilized (eg, more aggressive EC may be treated with amputation instead of WLE). Underlying patient comorbidities could potentially affect recurrence rates, which is a variable we could not control for in our analysis.
Our findings are limited by study design, with supporting evidence consisting of case reports and series. The review is limited by interstudy variability and heterogeneity of results. Additionally, recurrence is not reported in all cases and may be a source of sampling bias. Further complicating the generalizability of these results is the lack of follow-up to evaluate morbidity and quality of life after treatment.
CONCLUSION
This review suggests that MMS is associated with lower recurrence rates than WLE for the treatment of EC. Further investigation of MMS for EC with appropriate follow-up is necessary to identify whether MMS is associated with lower recurrence and less functional impairment. Nonsurgical treatments, including topical imiquimod, interferon alfa, and acitretin, may be useful in cases where surgical therapies are contraindicated, but there is little evidence to support these treatment modalities. Treatment guidelines for EC are not established, and appropriate treatment guidelines should be developed in the future.
Verrucous carcinoma (VC) is an uncommon type of well-differentiated squamous cell carcinoma (SCC) that most commonly affects men in the fifth to sixth decades of life. 1 The tumor grows slowly over a decade or more and does not frequently metastasize but has a high propensity for recurrence and local invasion. 2 There are 3 main subtypes of VC classified by anatomic site: oral florid papillomatosis (oral cavity), Buschke-Lowenstein tumor (anogenital region), and epithelioma cuniculatum (EC)(feet). 3 Epithelioma cuniculatum, also known as carcinoma cuniculatum or papillomatosis cutis carcinoides, most commonly presents as a solitary, warty or cauliflowerlike, exophytic mass with keratin-filled sinus tracts and malodorous discharge. 4 Diabetic foot ulcers and chronic inflammatory conditions are predisposing risk factors for EC, and it can result in difficulty walking/immobility, pain, and bleeding depending on anatomic involvement. 5-9
The differential diagnosis for VC includes refractory verruca vulgaris, clavus, SCC, keratoacanthoma, deep fungal or mycobacterial infection, eccrine poroma or porocarcinoma, amelanotic melanoma, and sarcoma.10-13 The slow-growing nature of VC, sampling error of superficial biopsies, and minimal cytological atypia on histologic examination can contribute to delayed diagnosis and appropriate treatment.14 Characteristic histologic features include hyperkeratosis, papillomatosis, marked acanthosis, broad blunt-ended rete ridges with a “bulldozing” architecture, and minimal cytologic atypia and mitoses.5,6 In some cases, pleomorphism and glassy eosinophilic cytoplasmic changes may be more pronounced than that of a common wart though less dramatic than that of conventional SCCs.15 Antigen Ki-67 and tumor protein p53 have been proposed to help differentiate between common plantar verruca, VC, and SCC, but the histologic diagnosis remains challenging, and repeat histopathologic examination often is required.16-19 Following diagnosis, computed tomography or magnetic resonance imaging may be necessary to determine tumor extension and assess for deep tissue and bony involvement.20-22
Treatment of EC is particularly challenging because of the anatomic location and need for margin control while maintaining adequate function, preserving healthy tissue, and providing coverage of defects. Surgical excision of EC is the first-line treatment, most commonly by wide local excision (WLE) or amputation. Mohs micrographic surgery (MMS) also has been utilized. One review found no recurrences in 5 cases of EC treated with MMS.23 As MMS is a tissue-sparing technique, this is a valuable modality for sites of functional importance such as the feet. Herein, we review various reported EC treatment modalities and outcomes, with an emphasis on recurrence rates for WLE and MMS.
METHODS
A systematic literature review of PubMed articles indexed for MEDLINE, as well as databases including the Cochrane Library, Web of Science, and Cumulative Index to Nursing and Allied Health Literature (CINAHL), was performed on January 14, 2020. Two authors (S.S.D. and S.V.C.) independently screened results using the search terms (plantar OR foot) AND (verrucous carcinoma OR epithelioma cuniculatum OR carcinoma cuniculatum). The search terms were chosen according to MeSH subject headings. All articles from the start date of the databases through the search date were screened, and articles pertaining to VC, EC, or carcinoma cuniculatum located on the foot were included. Of these, non–English-language articles were translated and included. Articles reporting VC on a site other than the foot (eg, the oral cavity) or benign verrucous skin lesions were excluded. The reference lists for all articles also were reviewed for additional reports that were absent from the initial search using both included and excluded articles. A full-text review was performed on 221 articles published between 1954 and 2019 per the PRISMA guidelines (Figure).

A total of 101 articles were included in the study for qualitative analysis. Nearly all articles identified were case reports, giving an evidence level of 5 by the Centre for Evidence-Based Medicine rating scale. Five articles reported data on multiple patients without individual demographic or clinical details and were excluded from analysis. Of the remaining 96 articles, information about patient characteristics, tumor size, treatment modality, and recurrence were extracted for 115 cases.
RESULTS
Of the 115 cases that were reviewed, 81 (70%) were male and 33 (29%) were female with a male-to-female ratio of 2.4:1. Ages of the patients ranged from 18 to 88 years; the mean and median age was 56 years. Nearly all reported cases of EC affected the plantar surface of one foot, with 4 reports of tumors affecting both feet.24-27 One case affecting both feet reported known exposure to lead arsenate pesticides27; all others were associated with a clinical history of chronic ulcers or warts persisting for several years to decades. Other less common sites of EC included the dorsal foot, interdigital web space, and subungual digit.28-30 The most common location reported was the anterior ball of the foot. Tumors were reported to arise within pre-existing lesions, such as hypertrophic lichen planus or chronic foot wounds associated with diabetes mellitus or leprosy.31-35 Tumor size ranged from 1 to 22 cm with a median of 4.5 cm.
Eight cases were reported to be associated with human papillomavirus; low-risk types 6 and 11 and high-risk types 16 and 18 were found in 6 cases.36-41 Two cases reported association with human papillomavirus type 2.7,42
Metastases to dermal and subdermal lymphatics, regional lymph nodes, and the lungs were reported in 3 cases, repectively.43-45 Of these, one primary tumor had received low-dose irradiation in the form of X-ray therapy.45
Treatment Modalities
The cases of EC that we reviewed included treatment with surgical and systemic therapies as well as other modalities such as acitretin, interferon alfa, topical imiquimod, curettage, debridement, electrodesiccation, and radiation. The Table includes a complete summary of the treatments we analyzed.

Surgical Therapy—The majority (91% [105/115]) of cases were treated surgically. The most common treatment modality was WLE (50% [58/115]), followed by amputation (37% [43/115]) and MMS (12% [14/115]).
Wide local excision was the most frequently reported treatment, with excision margins of at least 5 mm to 1 cm.48 Incidence of recurrence was reported for 57% (33/58) of cases treated with WLE; of these, the recurrence rate was 33% (11/33). For patients with EC recurrence, the most common secondary treatment was repeat excision with wider margins (1–2 cm) or amputation (5/11).49-52 Few postoperative complications were reported but included pain, infection, and difficulty walking, which were mostly associated with repair modality (eg, split-thickness skin grafts, rotational flaps).53 Amputation was the second most common treatment modality, with a 67% (29/43) incidence of recurrence. Types of amputation included transmetatarsal ray amputation (7/43 [16%]), foot or forefoot amputation (2/43 [5%]), above-the-knee amputation (1/43 [2%]), and below-the-knee amputation (1/43 [2%]). Complications associated with amputation included infection and requirement of prosthetics for ambulation. Split-thickness skin grafts and rotational flaps were the most common surgical repairs performed.52,53
Mohs micrographic surgery was the least frequently reported surgical treatment modality. Both traditional MMS on fresh tissue and “slow Mohs,” with formalin-fixed paraffin embedded tissue examination over several days, were performed for EC with horizontal en face sectioning.54-56 Incidence of recurrence was reported for 86% (12/14) of MMS cases. Of these, recurrence was seen in 17% (2/12) that utilized a flat horizontal processing of tissue sections coupled with saucerlike excisions to enable examination of the entire undersurface and margins. In one case, the patient was treated with MMS with recurrence noted 1 month later; thus, repeat MMS was performed, and the tumor was found to be entwined around the flexor tendon.57 The tendon was removed, and clear margins were obtained. Follow-up 3 years after the second MMS revealed no signs of recurrence.57 In the other case, the patient had a particularly aggressive course with bilateral VC in the setting of diabetic ulcers that was treated with WLE prior to MMS and recurrence still noted after MMS.26 No complications were reported with MMS.
Overall, recurrence was most frequently reported with WLE (11/33 [33%]), followed by MMS (2/12 [17%]) and amputation (3/29 [10%]). When comparing WLE and amputation, the relationship between treatment modality and recurrence was statistically significant using a χ2 test of independence (χ2=4.7; P=.03). However, results were not significant with Yates correction for continuity (χ2=3.4; P=.06). The χ2 test of independence showed no significant association between treatment method and recurrence when comparing WLE with MMS (χ2=1.2; P=.28). Reported follow-up times varied greatly from a few months to 10 years.
Systemic Therapy—Of the total cases, only 2 cases reported treatment with acitretin and 2 utilized interferon alfa.58,59 In one case, treatment of EC with interferon alfa alone required more aggressive therapy (ie, amputation).58 Neither of the 2 cases using acitretin reported recurrence.59,60 Complications of acitretin therapy included cheilitis and transaminitis.60
Other Treatment Modalities—Three cases utilized imiquimod, with 2 cases of imiquimod monotherapy and 1 case of imiquimod in combination with electrodesiccation and WLE.37 One of the cases of EC treated with imiquimod monotherapy recurred and required WLE.61
There were reports of other treatments including curettage alone (2% [2/115]),40,62 debridement alone (1% [1/115]),40 electrodesiccation (1% [1/115]),37 and radiation (1% [1/115]).43 Recurrence was found with curettage alone and debridement alone. Electrodesiccation was reported in conjunction with WLE without recurrence. Radiation was used to treat a case of VC that had metastasized to the lymph nodes; no follow-up was described.43
COMMENT
Epithelioma cuniculatum is an indolent malignancy of the plantar foot that likely is frequently underdiagnosed or misdiagnosed because of location, sampling error, and challenges in histopathologic diagnosis. Once diagnosed, surgical removal with margin control is the first-line therapy for EC. Our review found a number of surgical, systemic, and other treatment modalities that have been used to treat EC, but there remains a lack of evidence to provide clear guidelines as to which therapies are most effective. Current data on the treatment of EC largely are limited to case reports and case series. To date, there are no reports of higher-quality studies or randomized controlled trials to assess the efficacy of various treatment modalities.
Our review found that WLE is the most common treatment modality for EC, followed by amputation and MMS. Three cases43-45 that reported metastasis to lymph nodes also were treated with fine-needle aspiration or biopsy, and it is recommended that sentinel lymph node biopsy be performed when there is a history of radiation exposure or clinically and sonographically unsuspicious lymph nodes, while dissection of regional nodes should be performed if lymph node metastasis is suspected.53 Additional treatments reported included acitretin, interferon alfa, topical imiquimod, curettage, debridement, and electrodesiccation, but because of the limited number of cases and variable efficacy, no conclusions can be made on the utility of these alternative modalities.
The lowest rate of reported recurrence was found with amputation, followed by MMS and WLE. Amputation is the most aggressive treatment option, but its superiority in lower recurrence rates was not statistically significant when compared with either WLE or MMS after Yates correction. Despite treatment with radical surgery, recurrence is still possible and may be associated with factors including greater size (>2 cm) and depth (>4 mm), poor histologic differentiation, perineural involvement, failure of previous treatments, and immunosuppression.63 No statistically significant difference in recurrence rates was found among surgical methods, though data trended toward lower rates of recurrence with MMS compared with WLE, as recurrence with MMS was only reported in 2 cases.25,56
The efficacy of MMS is well documented for tumors with contiguous growth and enables maximum preservation of normal tissue structure and function with complete margin visualization. Thus, our results are in agreement with those of prior studies,54-56,64 suggesting that MMS is associated with lower recurrence rates for EC than WLE. Future studies and reporting of MMS for EC are particularly important because of the functional importance of the plantar foot.
It is important to note that there are local and systemic risk factors that increase the likelihood of developing EC and facilitate tumor growth, including antecedent trauma to the lesion site, chronic irritation or infection, and immunosuppression (HIV related or iatrogenic medication induced). These risk factors may play a role in the treatment modality utilized (eg, more aggressive EC may be treated with amputation instead of WLE). Underlying patient comorbidities could potentially affect recurrence rates, which is a variable we could not control for in our analysis.
Our findings are limited by study design, with supporting evidence consisting of case reports and series. The review is limited by interstudy variability and heterogeneity of results. Additionally, recurrence is not reported in all cases and may be a source of sampling bias. Further complicating the generalizability of these results is the lack of follow-up to evaluate morbidity and quality of life after treatment.
CONCLUSION
This review suggests that MMS is associated with lower recurrence rates than WLE for the treatment of EC. Further investigation of MMS for EC with appropriate follow-up is necessary to identify whether MMS is associated with lower recurrence and less functional impairment. Nonsurgical treatments, including topical imiquimod, interferon alfa, and acitretin, may be useful in cases where surgical therapies are contraindicated, but there is little evidence to support these treatment modalities. Treatment guidelines for EC are not established, and appropriate treatment guidelines should be developed in the future.
- McKee PH, Wilkinson JD, Black MM, et al. Carcinoma (epithelioma) cuniculatum: a clinicopathological study of nineteen cases and review of the literature. Histopathology. 1981;5:425-436.
- Aird I, Johnson HD, Lennox B, et al. Epithelioma cuniculatum: a variety of squamous carcinoma peculiar to the foot. Br J Surg. 1954;42:245-250.
- Seremet S, Erdemir AT, Kiremitci U, et al. Unusually early-onset plantar verrucous carcinoma. Cutis. 2019;104:34-36.
- Spyriounis PK, Tentis D, Sparveri IF, et al. Plantar epithelioma cuniculatum. a case report with review of the literature. Eur J Plast Surg. 2004;27:253-256.
- Ho J, Diven G, Bu J, et al. An ulcerating verrucous plaque on the foot. verrucous carcinoma (epithelioma cuniculatum). Arch Dermatol. 2000;136:547-548, 550-551.
- Kao GF, Graham JH, Helwig EB. Carcinoma cuniculatum (verrucous carcinoma of the skin): a clinicopathologic study of 46 cases with ultrastructural observations. Cancer. 1982;49:2395-2403.
- Zielonka E, Goldschmidt D, de Fontaine S. Verrucous carcinoma or epithelioma cuniculatum plantare. Eur J Surg Oncol. 1997;23:86-87.
- Dogan G, Oram Y, Hazneci E, et al. Three cases of verrucous carcinoma. Australas J Dermatol. 1998;39:251-254.
- Schwartz RA, Burgess GH. Verrucous carcinoma of the foot. J Surg Oncol. 1980;14:333-339.
- McKay C, McBride P, Muir J. Plantar verrucous carcinoma masquerading as toe web intertrigo. Australas J Dermatol. 2012;53:2010-2012.
- Shenoy AS, Waghmare RS, Kavishwar VS, et al. Carcinoma cuniculatum of foot. Foot. 2011;21:207-208.
- Lozzi G, Perris K. Carcinoma cuniculatum. CMAJ. 2007;177:249-251.
- Schein O, Orenstein A, Bar-Meir E. Plantar verrucous carcicoma (epithelioma cuniculatum): rare form of the common wart. Isr Med Assoc J. 2006;8:885.
- Rheingold LM, Roth LM. Carcinoma of the skin of the foot exhibiting some verrucous features. Plast Reconstr Surg. 1978;61:605-609.
- Klima M, Kurtis B, Jordan PH. Verrucous carcinoma of skin. J Cutan Pathol. 1980;7:88-98.
- Nakamura Y, Kashiwagi K, Nakamura A, et al. Verrucous carcinoma of the foot diagnosed using p53 and Ki-67 immunostaining in a patient with diabetic neuropathy. Am J Dermatopathol. 2015;37:257-259.
- Costache M, Desa LT, Mitrache LE, et al. Cutaneous verrucous carcinoma—report of three cases with review of literature. Rom J Morphol Embryol. 2014;55:383-388.
- Terada T. Verrucous carcinoma of the skin: a report on 5 Japanese cases. Ann Diagn Pathol. 2011;15:175-180.
- Noel JC, Heenen M, Peny MO, et al. Proliferating cell nuclear antigen distribution in verrucous carcinoma of the skin. Br J Dermatol. 1995;133:868-873.
- García-Gavín J, González-Vilas D, Rodríguez-Pazos L, et al. Verrucous carcinoma of the foot affecting the bone: utility of the computed tomography scanner. Dermatol Online J. 2010;16:3-5.
- Wasserman PL, Taylor RC, Pinillia J, et al. Verrucous carcinoma of the foot and enhancement assessment by MRI. Skeletal Radiol. 2009;38:393-395.
- Bhushan MH, Ferguson JE, Hutchinson CE. Carcinoma cuniculatum of the foot assessed by magnetic resonance scanning. Clin Exp Dermatol. 2001;26:419-422.
- Penera KE, Manji KA, Craig AB, et al. Atypical presentation of verrucous carcinoma: a case study and review of the literature. Foot Ankle Spec. 2013;6:318-322.
- Suen K, Wijeratne S, Patrikios J. An unusual case of bilateral verrucous carcinoma of the foot (epithelioma cuniculatum). J Surg Case Rep. 2012;2012:rjs020.
- Riccio C, King K, Elston JB, et al. Bilateral plantar verrucous carcinoma. Eplasty. 2016;16:ic46.
- Di Palma V, Stone JP, Schell A, et al. Mistaken diabetic ulcers: a case of bilateral foot verrucous carcinoma. Case Rep Dermatol Med. 2018;2018:4192657.
- Seehafer JR, Muller SA, Dicken CH. Bilateral verrucous carcinoma of the feet. Orthop Surv. 1979;3:205.
- Tosti A, Morelli R, Fanti PA, et al. Carcinoma cuniculatum of the nail apparatus: report of three cases. Dermatology. 1993;186:217-221.
- Melo CR, Melo IS, Souza LP. Epithelioma cuniculatum, a verrucous carcinoma of the foot. report of 2 cases. Dermatologica. 1981;163:338-342.
- Van Geertruyden JP, Olemans C, Laporte M, et al. Verrucous carcinoma of the nail bed. Foot Ankle Int. 1998;19:327-328.
- Thakur BK, Verma S, Raphael V. Verrucous carcinoma developing in a long standing case of ulcerative lichen planus of sole: a rare case report. J Eur Acad Dermatol Venereol. 2015;29:399-401.
- Mayron R, Grimwood RE, Siegle RJ, et al. Verrucous carcinoma arising in ulcerative lichen planus of the soles. J Dermatol Surg Oncol. 1988;14:547-551.
- Boussofara L, Belajouza-Noueiri C, Ghariani N, et al. Verrucous epidermoid carcinoma as a complication in cutaneous lichen planus [article in French]. Ann Dermatol Venereol. 2006;133:404-405.
- Khullar G, Mittal S, Sharma S. Verrucous carcinoma on the foot arising in a chronic neuropathic ulcer of leprosy. Australas J Dermatol. 2019;60:245-246.
- Ochsner PE, Hausman R, Olsthoorn PGM. Epithelioma cunicalutum developing in a neuropathic ulcer of leprous etiology. Arch Orthop Trauma Surg. 1979;94:227-231.
- Ray R, Bhagat A, Vasudevan B, et al. A rare case of plantar epithelioma cuniculatum arising from a wart. Indian J Dermatol. 2015;60:485-487.
- Imko-Walczuk B, Cegielska A, Placek W, et al. Human papillomavirus-related verrucous carcinoma in a renal transplant patient after long-term immunosuppression: a case report. Transplant Proc. 2014;46:2916-2919.
- Floristán MU, Feltes RA, Sáenz JC, et al. Verrucous carcinoma of the foot associated with human papillomavirus type 18. Actas Dermosifiliogr. 2009;100:433-435.
- Sasaoka R, Morimura T, Mihara M, et al. Detection of human pupillomavirus type 16 DNA in two cases of verriicous carcinoma of the foot. Br J Dermatol. 1996;134:983984.
- Schell BJ, Rosen T, Rády P, et al. Verrucous carcinoma of the foot associated with human papillomavirus type 16. J Am Acad Dermatol. 2001;45:49-55.
- Knobler RM, Schneider S, Neumann RA, et al. DNA dot‐blot hybridization implicates human papillomavirus type 11‐DNA in epithelioma cuniculatum. J Med Virol. 1989;29:33-37.
- Noel JC, Peny MO, Detremmerie O, et al. Demonstration of human papillomavirus type 2 in a verrucous carcinoma of the foot. Dermatology. 1993;187:58-61.
- Jungmann J, Vogt T, Müller CSL. Giant verrucous carcinoma of the lower extremity in women with dementia. BMJ Case Rep. 2012;2012:bcr2012006357.
- McKee PH, Wilkinson JD, Corbett MF, et al. Carcinoma cuniculatum: a case metastasizing to skin and lymph nodes. Clin Exp Dermatol. 1981;6:613-618.
- Owen WR, Wolfe ID, Burnett JW, et al. Epithelioma cuniculatum. South Med J. 1978;71:477-479.
- Patel AN, Bedforth N, Varma S. Pain-free treatment of carcinoma cuniculatum on the heel using Mohs micrographic surgery and ultrasonography-guided sciatic nerve block. Clin Exp Dermatol. 2013;38:569-571.
- Padilla RS, Bailin PL, Howard WR, et al. Verrucous carcinoma of the skin and its management by Mohs’ surgery. Plast Reconstr Surg. 1984;73:442-447.
- Kotwal M, Poflee S, Bobhate S. Carcinoma cuniculatum at various anatomical sites. Indian J Dermatol. 2005;50:216-220.
- Arefi M, Philipone E, Caprioli R, et al. A case of verrucous carcinoma (epithelioma cuniculatum) of the heel mimicking infected epidermal cyst and gout. Foot Ankle Spec. 2008;1:297-299.
- Trebing D, Brunner M, Kröning Y, et al. Young man with verrucous heel tumor [article in German]. J Dtsch Dermatol Ges. 2003;9:739-741.
- Thompson SG. Epithelioma cuniculatum: an unusual tumour of the foot. Br J Plast Surg. 1965;18:214-217.
- Thomas EJ, Graves NC, Meritt SM. Carcinoma cuniculatum: an atypical presentation in the foot. J Foot Ankle Surg. 2014;53:356-359.
- Koch H, Kowatsch E, Hödl S, et al. Verrucous carcinoma of the skin: long-term follow-up results following surgical therapy. Dermatol Surg. 2004;30:1124-1130.
- Mallatt BD, Ceilley RI, Dryer RF. Management of verrucous carcinoma on a foot by a combination of chemosurgery and plastic repair: report of a case. J Dermatol Surg Oncol. 1980;6:532-534.
- Mohs FE, Sahl WJ. Chemosurgery for verrucous carcinoma. J Dermatol Surg Oncol. 1979;5:302-306.
- Alkalay R, Alcalay J, Shiri J. Plantar verrucous carcinoma treated with Mohs micrographic surgery: a case report and literature review. J Drugs Dermatol. 2006;5:68-73.
- Mora RG. Microscopically controlled surgery (Mohs’ chemosurgery) for treatment of verrucous squamous cell carcinoma of the foot (epithelioma cuniculatum). J Am Acad Dermatol. 1983;8:354-362.
- Risse L, Negrier P, Dang PM, et al. Treatment of verrucous carcinoma with recombinant alfa-interferon. Dermatology. 1995;190:142-144.
- Rogozin´ski TT, Schwartz RA, Towpik E. Verrucous carcinoma in Unna-Thost hyperkeratosis of the palms and soles. J Am Acad Dermatol. 1994;31:1061-1062.
- Kuan YZ, Hsu HC, Kuo TT, et al. Multiple verrucous carcinomas treated with acitretin. J Am Acad Dermatol. 2007;56(2 suppl):S29-S32.
- Schalock PC, Kornik RI, Baughman RD, et al. Treatment of verrucous carcinoma with topical imiquimod. J Am Acad Dermatol. 2006;54:233-234.
- Brown SM, Freeman RG. Epithelioma cuniculatum. Arch Dermatol. 1976;112:1295-1296.
- Rowe DE, Carroll RJ, Day CL, et al. Prognostic factors for local recurrence, metastasis, and survival rates in squamous cell carcinoma of the skin, ear, and lip. J Am Acad Dermatol. 1992;26:976-990.
- Swanson NA, Taylor WB. Plantar verrucous carcinoma: literature review and treatment by the Mohs’ chemosurgery technique. Arch Dermatol. 1980;116:794-797.
- McKee PH, Wilkinson JD, Black MM, et al. Carcinoma (epithelioma) cuniculatum: a clinicopathological study of nineteen cases and review of the literature. Histopathology. 1981;5:425-436.
- Aird I, Johnson HD, Lennox B, et al. Epithelioma cuniculatum: a variety of squamous carcinoma peculiar to the foot. Br J Surg. 1954;42:245-250.
- Seremet S, Erdemir AT, Kiremitci U, et al. Unusually early-onset plantar verrucous carcinoma. Cutis. 2019;104:34-36.
- Spyriounis PK, Tentis D, Sparveri IF, et al. Plantar epithelioma cuniculatum. a case report with review of the literature. Eur J Plast Surg. 2004;27:253-256.
- Ho J, Diven G, Bu J, et al. An ulcerating verrucous plaque on the foot. verrucous carcinoma (epithelioma cuniculatum). Arch Dermatol. 2000;136:547-548, 550-551.
- Kao GF, Graham JH, Helwig EB. Carcinoma cuniculatum (verrucous carcinoma of the skin): a clinicopathologic study of 46 cases with ultrastructural observations. Cancer. 1982;49:2395-2403.
- Zielonka E, Goldschmidt D, de Fontaine S. Verrucous carcinoma or epithelioma cuniculatum plantare. Eur J Surg Oncol. 1997;23:86-87.
- Dogan G, Oram Y, Hazneci E, et al. Three cases of verrucous carcinoma. Australas J Dermatol. 1998;39:251-254.
- Schwartz RA, Burgess GH. Verrucous carcinoma of the foot. J Surg Oncol. 1980;14:333-339.
- McKay C, McBride P, Muir J. Plantar verrucous carcinoma masquerading as toe web intertrigo. Australas J Dermatol. 2012;53:2010-2012.
- Shenoy AS, Waghmare RS, Kavishwar VS, et al. Carcinoma cuniculatum of foot. Foot. 2011;21:207-208.
- Lozzi G, Perris K. Carcinoma cuniculatum. CMAJ. 2007;177:249-251.
- Schein O, Orenstein A, Bar-Meir E. Plantar verrucous carcicoma (epithelioma cuniculatum): rare form of the common wart. Isr Med Assoc J. 2006;8:885.
- Rheingold LM, Roth LM. Carcinoma of the skin of the foot exhibiting some verrucous features. Plast Reconstr Surg. 1978;61:605-609.
- Klima M, Kurtis B, Jordan PH. Verrucous carcinoma of skin. J Cutan Pathol. 1980;7:88-98.
- Nakamura Y, Kashiwagi K, Nakamura A, et al. Verrucous carcinoma of the foot diagnosed using p53 and Ki-67 immunostaining in a patient with diabetic neuropathy. Am J Dermatopathol. 2015;37:257-259.
- Costache M, Desa LT, Mitrache LE, et al. Cutaneous verrucous carcinoma—report of three cases with review of literature. Rom J Morphol Embryol. 2014;55:383-388.
- Terada T. Verrucous carcinoma of the skin: a report on 5 Japanese cases. Ann Diagn Pathol. 2011;15:175-180.
- Noel JC, Heenen M, Peny MO, et al. Proliferating cell nuclear antigen distribution in verrucous carcinoma of the skin. Br J Dermatol. 1995;133:868-873.
- García-Gavín J, González-Vilas D, Rodríguez-Pazos L, et al. Verrucous carcinoma of the foot affecting the bone: utility of the computed tomography scanner. Dermatol Online J. 2010;16:3-5.
- Wasserman PL, Taylor RC, Pinillia J, et al. Verrucous carcinoma of the foot and enhancement assessment by MRI. Skeletal Radiol. 2009;38:393-395.
- Bhushan MH, Ferguson JE, Hutchinson CE. Carcinoma cuniculatum of the foot assessed by magnetic resonance scanning. Clin Exp Dermatol. 2001;26:419-422.
- Penera KE, Manji KA, Craig AB, et al. Atypical presentation of verrucous carcinoma: a case study and review of the literature. Foot Ankle Spec. 2013;6:318-322.
- Suen K, Wijeratne S, Patrikios J. An unusual case of bilateral verrucous carcinoma of the foot (epithelioma cuniculatum). J Surg Case Rep. 2012;2012:rjs020.
- Riccio C, King K, Elston JB, et al. Bilateral plantar verrucous carcinoma. Eplasty. 2016;16:ic46.
- Di Palma V, Stone JP, Schell A, et al. Mistaken diabetic ulcers: a case of bilateral foot verrucous carcinoma. Case Rep Dermatol Med. 2018;2018:4192657.
- Seehafer JR, Muller SA, Dicken CH. Bilateral verrucous carcinoma of the feet. Orthop Surv. 1979;3:205.
- Tosti A, Morelli R, Fanti PA, et al. Carcinoma cuniculatum of the nail apparatus: report of three cases. Dermatology. 1993;186:217-221.
- Melo CR, Melo IS, Souza LP. Epithelioma cuniculatum, a verrucous carcinoma of the foot. report of 2 cases. Dermatologica. 1981;163:338-342.
- Van Geertruyden JP, Olemans C, Laporte M, et al. Verrucous carcinoma of the nail bed. Foot Ankle Int. 1998;19:327-328.
- Thakur BK, Verma S, Raphael V. Verrucous carcinoma developing in a long standing case of ulcerative lichen planus of sole: a rare case report. J Eur Acad Dermatol Venereol. 2015;29:399-401.
- Mayron R, Grimwood RE, Siegle RJ, et al. Verrucous carcinoma arising in ulcerative lichen planus of the soles. J Dermatol Surg Oncol. 1988;14:547-551.
- Boussofara L, Belajouza-Noueiri C, Ghariani N, et al. Verrucous epidermoid carcinoma as a complication in cutaneous lichen planus [article in French]. Ann Dermatol Venereol. 2006;133:404-405.
- Khullar G, Mittal S, Sharma S. Verrucous carcinoma on the foot arising in a chronic neuropathic ulcer of leprosy. Australas J Dermatol. 2019;60:245-246.
- Ochsner PE, Hausman R, Olsthoorn PGM. Epithelioma cunicalutum developing in a neuropathic ulcer of leprous etiology. Arch Orthop Trauma Surg. 1979;94:227-231.
- Ray R, Bhagat A, Vasudevan B, et al. A rare case of plantar epithelioma cuniculatum arising from a wart. Indian J Dermatol. 2015;60:485-487.
- Imko-Walczuk B, Cegielska A, Placek W, et al. Human papillomavirus-related verrucous carcinoma in a renal transplant patient after long-term immunosuppression: a case report. Transplant Proc. 2014;46:2916-2919.
- Floristán MU, Feltes RA, Sáenz JC, et al. Verrucous carcinoma of the foot associated with human papillomavirus type 18. Actas Dermosifiliogr. 2009;100:433-435.
- Sasaoka R, Morimura T, Mihara M, et al. Detection of human pupillomavirus type 16 DNA in two cases of verriicous carcinoma of the foot. Br J Dermatol. 1996;134:983984.
- Schell BJ, Rosen T, Rády P, et al. Verrucous carcinoma of the foot associated with human papillomavirus type 16. J Am Acad Dermatol. 2001;45:49-55.
- Knobler RM, Schneider S, Neumann RA, et al. DNA dot‐blot hybridization implicates human papillomavirus type 11‐DNA in epithelioma cuniculatum. J Med Virol. 1989;29:33-37.
- Noel JC, Peny MO, Detremmerie O, et al. Demonstration of human papillomavirus type 2 in a verrucous carcinoma of the foot. Dermatology. 1993;187:58-61.
- Jungmann J, Vogt T, Müller CSL. Giant verrucous carcinoma of the lower extremity in women with dementia. BMJ Case Rep. 2012;2012:bcr2012006357.
- McKee PH, Wilkinson JD, Corbett MF, et al. Carcinoma cuniculatum: a case metastasizing to skin and lymph nodes. Clin Exp Dermatol. 1981;6:613-618.
- Owen WR, Wolfe ID, Burnett JW, et al. Epithelioma cuniculatum. South Med J. 1978;71:477-479.
- Patel AN, Bedforth N, Varma S. Pain-free treatment of carcinoma cuniculatum on the heel using Mohs micrographic surgery and ultrasonography-guided sciatic nerve block. Clin Exp Dermatol. 2013;38:569-571.
- Padilla RS, Bailin PL, Howard WR, et al. Verrucous carcinoma of the skin and its management by Mohs’ surgery. Plast Reconstr Surg. 1984;73:442-447.
- Kotwal M, Poflee S, Bobhate S. Carcinoma cuniculatum at various anatomical sites. Indian J Dermatol. 2005;50:216-220.
- Arefi M, Philipone E, Caprioli R, et al. A case of verrucous carcinoma (epithelioma cuniculatum) of the heel mimicking infected epidermal cyst and gout. Foot Ankle Spec. 2008;1:297-299.
- Trebing D, Brunner M, Kröning Y, et al. Young man with verrucous heel tumor [article in German]. J Dtsch Dermatol Ges. 2003;9:739-741.
- Thompson SG. Epithelioma cuniculatum: an unusual tumour of the foot. Br J Plast Surg. 1965;18:214-217.
- Thomas EJ, Graves NC, Meritt SM. Carcinoma cuniculatum: an atypical presentation in the foot. J Foot Ankle Surg. 2014;53:356-359.
- Koch H, Kowatsch E, Hödl S, et al. Verrucous carcinoma of the skin: long-term follow-up results following surgical therapy. Dermatol Surg. 2004;30:1124-1130.
- Mallatt BD, Ceilley RI, Dryer RF. Management of verrucous carcinoma on a foot by a combination of chemosurgery and plastic repair: report of a case. J Dermatol Surg Oncol. 1980;6:532-534.
- Mohs FE, Sahl WJ. Chemosurgery for verrucous carcinoma. J Dermatol Surg Oncol. 1979;5:302-306.
- Alkalay R, Alcalay J, Shiri J. Plantar verrucous carcinoma treated with Mohs micrographic surgery: a case report and literature review. J Drugs Dermatol. 2006;5:68-73.
- Mora RG. Microscopically controlled surgery (Mohs’ chemosurgery) for treatment of verrucous squamous cell carcinoma of the foot (epithelioma cuniculatum). J Am Acad Dermatol. 1983;8:354-362.
- Risse L, Negrier P, Dang PM, et al. Treatment of verrucous carcinoma with recombinant alfa-interferon. Dermatology. 1995;190:142-144.
- Rogozin´ski TT, Schwartz RA, Towpik E. Verrucous carcinoma in Unna-Thost hyperkeratosis of the palms and soles. J Am Acad Dermatol. 1994;31:1061-1062.
- Kuan YZ, Hsu HC, Kuo TT, et al. Multiple verrucous carcinomas treated with acitretin. J Am Acad Dermatol. 2007;56(2 suppl):S29-S32.
- Schalock PC, Kornik RI, Baughman RD, et al. Treatment of verrucous carcinoma with topical imiquimod. J Am Acad Dermatol. 2006;54:233-234.
- Brown SM, Freeman RG. Epithelioma cuniculatum. Arch Dermatol. 1976;112:1295-1296.
- Rowe DE, Carroll RJ, Day CL, et al. Prognostic factors for local recurrence, metastasis, and survival rates in squamous cell carcinoma of the skin, ear, and lip. J Am Acad Dermatol. 1992;26:976-990.
- Swanson NA, Taylor WB. Plantar verrucous carcinoma: literature review and treatment by the Mohs’ chemosurgery technique. Arch Dermatol. 1980;116:794-797.
Practice Points
- Because of its slow-growing nature and propensity for local invasion and recurrence, diagnosis of epithelioma cuniculatum (EC) often is delayed and therefore can be associated with notable morbidity.
- Wide local excision with 5-mm to 1-cm margins is considered standard of care and is the most commonly reported treatment of EC. Amputation may be required in cases with extensive local destruction.
- Mohs micrographic surgery is a viable option for treatment of EC, with more recent cases suggesting favorable outcomes regarding recurrence rates.
The Evidence Behind Topical Hair Loss Remedies on TikTok
Hair loss is an exceedingly common chief concern in outpatient dermatology clinics. An estimated 50% of males and females will experience androgenetic alopecia.1 Approximately 2% of new dermatology outpatient visits in the United States and the United Kingdom are for alopecia areata, the second most common type of hair loss.2 As access to dermatology appointments remains an issue with some studies citing wait times ranging from 2 to 25 days for a dermatologic consultation, the ease of accessibility of medical information on social media continues to grow,3 which leaves many of our patients turning to social media as a first-line source of information. As dermatology resident physicians, it is essential to be aware of popular dermatologic therapies on social media so that we may provide evidence-based opinions to our patients.
Remedies for Hair Loss on Social Media
Many trends on hair loss therapies found on TikTok focus on natural remedies that are produced by ingredients accessible to patients at home and over the counter, which may increase the appeal due to ease of treatment.
Rosemary Oil—The top trends in hair loss remedies I have come across are rosemary oil and rosemary water. Rosemary (Rosmarinus officinalis) has been known to possess antimicrobial and antioxidant properties but also has shown enhancement of microcapillary perfusion, which could explain its role in the prevention of hair loss and aiding hair growth in a similar mechanism to minoxidil.4,5 Unlike many other natural hair loss remedies, there are randomized controlled trials that assess the efficacy of rosemary oil for the treatment of hair loss. In a 2015 study of 100 patients with androgenetic alopecia, there was no statistically significant difference in mean hair count measured by microphotographic assessment after 6 months of treatment in 2 groups treated with either minoxidil solution 2% or rosemary oil, and both groups experienced a significant increase in hair count at 6 months (P<.05) compared with baseline and 3 months.6 Additionally, essential oils, including a mixture of thyme, rosemary, lavender, and cedarwood oils for alopecia were superior to placebo carrier oils in a posttreatment photographic assessment of their efficacy.7
Rice Water—The use of rice water and rice bran extract is a common hair care practice in Asia. Rice bran extract preparations have been shown in vivo to increase the number of anagen hair follicles as well as the number of anagen-related molecules in the dermal papillae.8,9 However, there are limited clinical data to support the use of rice water for hair growth.10
Onion Juice—Sharquie and Al-Obaidi11 conducted a study comparing crude onion juice to tap water in 38 patients with alopecia areata. They found that onion juice produced hair regrowth in significantly more patients than tap water (P<.0001).11 The mechanism of crude onion juice in hair growth is unknown; however, the induction of irritant or allergic contact dermatitis to components in crude onion juice may stimulate antigenic competition.12
Garlic Gel—Garlic gel, which is in the genus Allium, produces organosulfur compounds that provide antimicrobial and anti-inflammatory benefits.12 Additionally, in a double-blind randomized controlled trial, garlic powder was shown to increase cutaneous capillary perfusion.5 One study in 40 patients with alopecia areata demonstrated garlic gel 5% added to betamethasone valerate cream 0.1% was statistically superior to betamethasone alone in stimulating terminal hair growth (P=.001).13
Limitations and Downsides to Hair Loss Remedies on Social Media
Social media continues to be a prominent source of medical information for our patients, but most sources of hair content on social media are not board-certified dermatologists. A recent review of alopecia-related content found only 4% and 10% of posts were created by medical professionals on Instagram and TikTok, respectively, making misinformation extremely likely.14 Natural hair loss remedies contrived by TikTok have little clinical evidence to support their claims. Few data are available that compare these treatments to gold-standard hair loss therapies. Additionally, while some of these agents may be beneficial, the lack of standardized dosing may counteract these benefits. For example, videos on rosemary water advise the viewer to boil fresh rosemary sprigs in water and apply the solution to the hair daily with a spray bottle or apply cloves of garlic directly to the scalp, as opposed to a measured and standardized percentage. Some preparations may even induce harm to patients. Over-the-counter oils with added fragrances and natural compounds in onion and garlic may cause contact dermatitis. Finally, by using these products, patients may delay consultation with a board-certified dermatologist, leading to delays in applying evidence-based therapies targeted to specific hair loss subtypes while also incurring unnecessary expenses for these preparations.
Final Thoughts
Hair loss affects a notable portion of the population and is a common chief concern in dermatology clinics. Misinformation on social media continues to grow in prevalence. It is important to be aware of the hair loss remedies that are commonly touted to patients online and the evidence behind them.
- Ho CH, Sood T, Zito PM. Androgenetic alopecia. StatPearls. StatPearls Publishing; 2022.
- McMichael AJ, Pearce DJ, Wasserman D, et al. Alopecia in the United States: outpatient utilization and common prescribing patterns. J Am Acad Dermatol. 2007;57(2 suppl):S49-S51.
- Creadore A, Desai S, Li SJ, et al. Insurance acceptance, appointment wait time, and dermatologist access across practice types in the US. JAMA Dermatol. 2021;157:181-188. doi:10.1001/jamadermatol.2020.5173
- Bassino E, Gasparri F, Munaron L. Protective role of nutritional plants containing flavonoids in hair follicle disruption: a review. Int J Mol Sci. 2020;21:523. doi:10.3390/ijms21020523
- Ezekwe N, King M, Hollinger JC. The use of natural ingredients in the treatment of alopecias with an emphasis on central centrifugal cicatricial alopecia: a systematic review [published online August 1, 2020]. J Clin Aesthet Dermatol. 2020;13:23-27.
- Panahi Y, Taghizadeh M, Marzony ET, et al. Rosemary oil vs minoxidil 2% for the treatment of androgenetic alopecia: a randomized comparative trial. Skinmed. 2015;13:15-21.
- Hay IC, Jamieson M, Ormerod AD. Randomized trial of aromatherapy. successful treatment for alopecia areata. Arch Dermatol. 1998;134:1349-1352. doi:10.1001/archderm.134.11.1349
- Choi JS, Jeon MH, Moon WS, et al. In vivo hair growth-promoting effect of rice bran extract prepared by supercritical carbon dioxide fluid. Biol Pharm Bull. 2014;37:44-53. doi:10.1248/bpb.b13-00528
- Kim YM, Kwon SJ, Jang HJ, et al. Rice bran mineral extract increases the expression of anagen-related molecules in human dermal papilla through wnt/catenin pathway. Food Nutr Res. 2017;61:1412792. doi:10.1080/16546628.2017.1412792
- Hashemi K, Pham C, Sung C, et al. A systematic review: application of rice products for hair growth. J Drugs Dermatol. 2022;21:177-185. doi:10.36849/jdd.6345
- Sharquie KE, Al-Obaidi HK. Onion juice (Allium cepa L.), a new topical treatment for alopecia areata. J Dermatol. 2002;29:343-346. doi:10.1111/j.1346-8138.2002.tb00277.x
- Hosking AM, Juhasz M, Atanaskova Mesinkovska N. Complementary and alternative treatments for alopecia: a comprehensive review. Skin Appendage Disord. 2019;5:72-89. doi:10.1159/000492035
- Hajheydari Z, Jamshidi M, Akbari J, et al. Combination of topical garlic gel and betamethasone valerate cream in the treatment of localized alopecia areata: a double-blind randomized controlled study. Indian J Dermatol Venereol Leprol. 2007;73:29-32. doi:10.4103/0378-6323.30648
- Laughter M, Anderson J, Kolla A, et al. An analysis of alopecia related content on Instagram and TikTok. J Drugs Dermatol. 2022;21:1316-1321. doi:10.36849/JDD.6707
Hair loss is an exceedingly common chief concern in outpatient dermatology clinics. An estimated 50% of males and females will experience androgenetic alopecia.1 Approximately 2% of new dermatology outpatient visits in the United States and the United Kingdom are for alopecia areata, the second most common type of hair loss.2 As access to dermatology appointments remains an issue with some studies citing wait times ranging from 2 to 25 days for a dermatologic consultation, the ease of accessibility of medical information on social media continues to grow,3 which leaves many of our patients turning to social media as a first-line source of information. As dermatology resident physicians, it is essential to be aware of popular dermatologic therapies on social media so that we may provide evidence-based opinions to our patients.
Remedies for Hair Loss on Social Media
Many trends on hair loss therapies found on TikTok focus on natural remedies that are produced by ingredients accessible to patients at home and over the counter, which may increase the appeal due to ease of treatment.
Rosemary Oil—The top trends in hair loss remedies I have come across are rosemary oil and rosemary water. Rosemary (Rosmarinus officinalis) has been known to possess antimicrobial and antioxidant properties but also has shown enhancement of microcapillary perfusion, which could explain its role in the prevention of hair loss and aiding hair growth in a similar mechanism to minoxidil.4,5 Unlike many other natural hair loss remedies, there are randomized controlled trials that assess the efficacy of rosemary oil for the treatment of hair loss. In a 2015 study of 100 patients with androgenetic alopecia, there was no statistically significant difference in mean hair count measured by microphotographic assessment after 6 months of treatment in 2 groups treated with either minoxidil solution 2% or rosemary oil, and both groups experienced a significant increase in hair count at 6 months (P<.05) compared with baseline and 3 months.6 Additionally, essential oils, including a mixture of thyme, rosemary, lavender, and cedarwood oils for alopecia were superior to placebo carrier oils in a posttreatment photographic assessment of their efficacy.7
Rice Water—The use of rice water and rice bran extract is a common hair care practice in Asia. Rice bran extract preparations have been shown in vivo to increase the number of anagen hair follicles as well as the number of anagen-related molecules in the dermal papillae.8,9 However, there are limited clinical data to support the use of rice water for hair growth.10
Onion Juice—Sharquie and Al-Obaidi11 conducted a study comparing crude onion juice to tap water in 38 patients with alopecia areata. They found that onion juice produced hair regrowth in significantly more patients than tap water (P<.0001).11 The mechanism of crude onion juice in hair growth is unknown; however, the induction of irritant or allergic contact dermatitis to components in crude onion juice may stimulate antigenic competition.12
Garlic Gel—Garlic gel, which is in the genus Allium, produces organosulfur compounds that provide antimicrobial and anti-inflammatory benefits.12 Additionally, in a double-blind randomized controlled trial, garlic powder was shown to increase cutaneous capillary perfusion.5 One study in 40 patients with alopecia areata demonstrated garlic gel 5% added to betamethasone valerate cream 0.1% was statistically superior to betamethasone alone in stimulating terminal hair growth (P=.001).13
Limitations and Downsides to Hair Loss Remedies on Social Media
Social media continues to be a prominent source of medical information for our patients, but most sources of hair content on social media are not board-certified dermatologists. A recent review of alopecia-related content found only 4% and 10% of posts were created by medical professionals on Instagram and TikTok, respectively, making misinformation extremely likely.14 Natural hair loss remedies contrived by TikTok have little clinical evidence to support their claims. Few data are available that compare these treatments to gold-standard hair loss therapies. Additionally, while some of these agents may be beneficial, the lack of standardized dosing may counteract these benefits. For example, videos on rosemary water advise the viewer to boil fresh rosemary sprigs in water and apply the solution to the hair daily with a spray bottle or apply cloves of garlic directly to the scalp, as opposed to a measured and standardized percentage. Some preparations may even induce harm to patients. Over-the-counter oils with added fragrances and natural compounds in onion and garlic may cause contact dermatitis. Finally, by using these products, patients may delay consultation with a board-certified dermatologist, leading to delays in applying evidence-based therapies targeted to specific hair loss subtypes while also incurring unnecessary expenses for these preparations.
Final Thoughts
Hair loss affects a notable portion of the population and is a common chief concern in dermatology clinics. Misinformation on social media continues to grow in prevalence. It is important to be aware of the hair loss remedies that are commonly touted to patients online and the evidence behind them.
Hair loss is an exceedingly common chief concern in outpatient dermatology clinics. An estimated 50% of males and females will experience androgenetic alopecia.1 Approximately 2% of new dermatology outpatient visits in the United States and the United Kingdom are for alopecia areata, the second most common type of hair loss.2 As access to dermatology appointments remains an issue with some studies citing wait times ranging from 2 to 25 days for a dermatologic consultation, the ease of accessibility of medical information on social media continues to grow,3 which leaves many of our patients turning to social media as a first-line source of information. As dermatology resident physicians, it is essential to be aware of popular dermatologic therapies on social media so that we may provide evidence-based opinions to our patients.
Remedies for Hair Loss on Social Media
Many trends on hair loss therapies found on TikTok focus on natural remedies that are produced by ingredients accessible to patients at home and over the counter, which may increase the appeal due to ease of treatment.
Rosemary Oil—The top trends in hair loss remedies I have come across are rosemary oil and rosemary water. Rosemary (Rosmarinus officinalis) has been known to possess antimicrobial and antioxidant properties but also has shown enhancement of microcapillary perfusion, which could explain its role in the prevention of hair loss and aiding hair growth in a similar mechanism to minoxidil.4,5 Unlike many other natural hair loss remedies, there are randomized controlled trials that assess the efficacy of rosemary oil for the treatment of hair loss. In a 2015 study of 100 patients with androgenetic alopecia, there was no statistically significant difference in mean hair count measured by microphotographic assessment after 6 months of treatment in 2 groups treated with either minoxidil solution 2% or rosemary oil, and both groups experienced a significant increase in hair count at 6 months (P<.05) compared with baseline and 3 months.6 Additionally, essential oils, including a mixture of thyme, rosemary, lavender, and cedarwood oils for alopecia were superior to placebo carrier oils in a posttreatment photographic assessment of their efficacy.7
Rice Water—The use of rice water and rice bran extract is a common hair care practice in Asia. Rice bran extract preparations have been shown in vivo to increase the number of anagen hair follicles as well as the number of anagen-related molecules in the dermal papillae.8,9 However, there are limited clinical data to support the use of rice water for hair growth.10
Onion Juice—Sharquie and Al-Obaidi11 conducted a study comparing crude onion juice to tap water in 38 patients with alopecia areata. They found that onion juice produced hair regrowth in significantly more patients than tap water (P<.0001).11 The mechanism of crude onion juice in hair growth is unknown; however, the induction of irritant or allergic contact dermatitis to components in crude onion juice may stimulate antigenic competition.12
Garlic Gel—Garlic gel, which is in the genus Allium, produces organosulfur compounds that provide antimicrobial and anti-inflammatory benefits.12 Additionally, in a double-blind randomized controlled trial, garlic powder was shown to increase cutaneous capillary perfusion.5 One study in 40 patients with alopecia areata demonstrated garlic gel 5% added to betamethasone valerate cream 0.1% was statistically superior to betamethasone alone in stimulating terminal hair growth (P=.001).13
Limitations and Downsides to Hair Loss Remedies on Social Media
Social media continues to be a prominent source of medical information for our patients, but most sources of hair content on social media are not board-certified dermatologists. A recent review of alopecia-related content found only 4% and 10% of posts were created by medical professionals on Instagram and TikTok, respectively, making misinformation extremely likely.14 Natural hair loss remedies contrived by TikTok have little clinical evidence to support their claims. Few data are available that compare these treatments to gold-standard hair loss therapies. Additionally, while some of these agents may be beneficial, the lack of standardized dosing may counteract these benefits. For example, videos on rosemary water advise the viewer to boil fresh rosemary sprigs in water and apply the solution to the hair daily with a spray bottle or apply cloves of garlic directly to the scalp, as opposed to a measured and standardized percentage. Some preparations may even induce harm to patients. Over-the-counter oils with added fragrances and natural compounds in onion and garlic may cause contact dermatitis. Finally, by using these products, patients may delay consultation with a board-certified dermatologist, leading to delays in applying evidence-based therapies targeted to specific hair loss subtypes while also incurring unnecessary expenses for these preparations.
Final Thoughts
Hair loss affects a notable portion of the population and is a common chief concern in dermatology clinics. Misinformation on social media continues to grow in prevalence. It is important to be aware of the hair loss remedies that are commonly touted to patients online and the evidence behind them.
- Ho CH, Sood T, Zito PM. Androgenetic alopecia. StatPearls. StatPearls Publishing; 2022.
- McMichael AJ, Pearce DJ, Wasserman D, et al. Alopecia in the United States: outpatient utilization and common prescribing patterns. J Am Acad Dermatol. 2007;57(2 suppl):S49-S51.
- Creadore A, Desai S, Li SJ, et al. Insurance acceptance, appointment wait time, and dermatologist access across practice types in the US. JAMA Dermatol. 2021;157:181-188. doi:10.1001/jamadermatol.2020.5173
- Bassino E, Gasparri F, Munaron L. Protective role of nutritional plants containing flavonoids in hair follicle disruption: a review. Int J Mol Sci. 2020;21:523. doi:10.3390/ijms21020523
- Ezekwe N, King M, Hollinger JC. The use of natural ingredients in the treatment of alopecias with an emphasis on central centrifugal cicatricial alopecia: a systematic review [published online August 1, 2020]. J Clin Aesthet Dermatol. 2020;13:23-27.
- Panahi Y, Taghizadeh M, Marzony ET, et al. Rosemary oil vs minoxidil 2% for the treatment of androgenetic alopecia: a randomized comparative trial. Skinmed. 2015;13:15-21.
- Hay IC, Jamieson M, Ormerod AD. Randomized trial of aromatherapy. successful treatment for alopecia areata. Arch Dermatol. 1998;134:1349-1352. doi:10.1001/archderm.134.11.1349
- Choi JS, Jeon MH, Moon WS, et al. In vivo hair growth-promoting effect of rice bran extract prepared by supercritical carbon dioxide fluid. Biol Pharm Bull. 2014;37:44-53. doi:10.1248/bpb.b13-00528
- Kim YM, Kwon SJ, Jang HJ, et al. Rice bran mineral extract increases the expression of anagen-related molecules in human dermal papilla through wnt/catenin pathway. Food Nutr Res. 2017;61:1412792. doi:10.1080/16546628.2017.1412792
- Hashemi K, Pham C, Sung C, et al. A systematic review: application of rice products for hair growth. J Drugs Dermatol. 2022;21:177-185. doi:10.36849/jdd.6345
- Sharquie KE, Al-Obaidi HK. Onion juice (Allium cepa L.), a new topical treatment for alopecia areata. J Dermatol. 2002;29:343-346. doi:10.1111/j.1346-8138.2002.tb00277.x
- Hosking AM, Juhasz M, Atanaskova Mesinkovska N. Complementary and alternative treatments for alopecia: a comprehensive review. Skin Appendage Disord. 2019;5:72-89. doi:10.1159/000492035
- Hajheydari Z, Jamshidi M, Akbari J, et al. Combination of topical garlic gel and betamethasone valerate cream in the treatment of localized alopecia areata: a double-blind randomized controlled study. Indian J Dermatol Venereol Leprol. 2007;73:29-32. doi:10.4103/0378-6323.30648
- Laughter M, Anderson J, Kolla A, et al. An analysis of alopecia related content on Instagram and TikTok. J Drugs Dermatol. 2022;21:1316-1321. doi:10.36849/JDD.6707
- Ho CH, Sood T, Zito PM. Androgenetic alopecia. StatPearls. StatPearls Publishing; 2022.
- McMichael AJ, Pearce DJ, Wasserman D, et al. Alopecia in the United States: outpatient utilization and common prescribing patterns. J Am Acad Dermatol. 2007;57(2 suppl):S49-S51.
- Creadore A, Desai S, Li SJ, et al. Insurance acceptance, appointment wait time, and dermatologist access across practice types in the US. JAMA Dermatol. 2021;157:181-188. doi:10.1001/jamadermatol.2020.5173
- Bassino E, Gasparri F, Munaron L. Protective role of nutritional plants containing flavonoids in hair follicle disruption: a review. Int J Mol Sci. 2020;21:523. doi:10.3390/ijms21020523
- Ezekwe N, King M, Hollinger JC. The use of natural ingredients in the treatment of alopecias with an emphasis on central centrifugal cicatricial alopecia: a systematic review [published online August 1, 2020]. J Clin Aesthet Dermatol. 2020;13:23-27.
- Panahi Y, Taghizadeh M, Marzony ET, et al. Rosemary oil vs minoxidil 2% for the treatment of androgenetic alopecia: a randomized comparative trial. Skinmed. 2015;13:15-21.
- Hay IC, Jamieson M, Ormerod AD. Randomized trial of aromatherapy. successful treatment for alopecia areata. Arch Dermatol. 1998;134:1349-1352. doi:10.1001/archderm.134.11.1349
- Choi JS, Jeon MH, Moon WS, et al. In vivo hair growth-promoting effect of rice bran extract prepared by supercritical carbon dioxide fluid. Biol Pharm Bull. 2014;37:44-53. doi:10.1248/bpb.b13-00528
- Kim YM, Kwon SJ, Jang HJ, et al. Rice bran mineral extract increases the expression of anagen-related molecules in human dermal papilla through wnt/catenin pathway. Food Nutr Res. 2017;61:1412792. doi:10.1080/16546628.2017.1412792
- Hashemi K, Pham C, Sung C, et al. A systematic review: application of rice products for hair growth. J Drugs Dermatol. 2022;21:177-185. doi:10.36849/jdd.6345
- Sharquie KE, Al-Obaidi HK. Onion juice (Allium cepa L.), a new topical treatment for alopecia areata. J Dermatol. 2002;29:343-346. doi:10.1111/j.1346-8138.2002.tb00277.x
- Hosking AM, Juhasz M, Atanaskova Mesinkovska N. Complementary and alternative treatments for alopecia: a comprehensive review. Skin Appendage Disord. 2019;5:72-89. doi:10.1159/000492035
- Hajheydari Z, Jamshidi M, Akbari J, et al. Combination of topical garlic gel and betamethasone valerate cream in the treatment of localized alopecia areata: a double-blind randomized controlled study. Indian J Dermatol Venereol Leprol. 2007;73:29-32. doi:10.4103/0378-6323.30648
- Laughter M, Anderson J, Kolla A, et al. An analysis of alopecia related content on Instagram and TikTok. J Drugs Dermatol. 2022;21:1316-1321. doi:10.36849/JDD.6707
Resident Pearl
- With terabytes of information at their fingertips, patients often turn to social media for hair loss advice. Many recommended therapies lack evidence-based research, and some may even be harmful to patients or delay time to efficacious treatments.
Multimodal Treatment of Epidermodysplasia Verruciformis in an HIV-Positive Man
To the Editor:
Epidermodysplasia verruciformis (EDV) is a rare generalized form of epidermal dysplasia that is linked to certain subtypes of human papillomavirus (HPV) infection and inherited or acquired states of immunodeficiency.1-3 The inherited form most commonly manifests via autosomal-recessive inactivation of the EVER1 and EVER2 genes that encode integral membrane proteins in the endoplasmic reticulum, though cases of autosomal-dominant and X-linked inheritance have been reported.1-3 Acquired cases have been reported in patients lacking immunocompetency, including transplant recipients and patients living with HIV.4-11 We present the case of a patient with HIV-associated EDV who was treated successfully with intralesional Candida albicans antigen, oral acitretin, and cryotherapy.
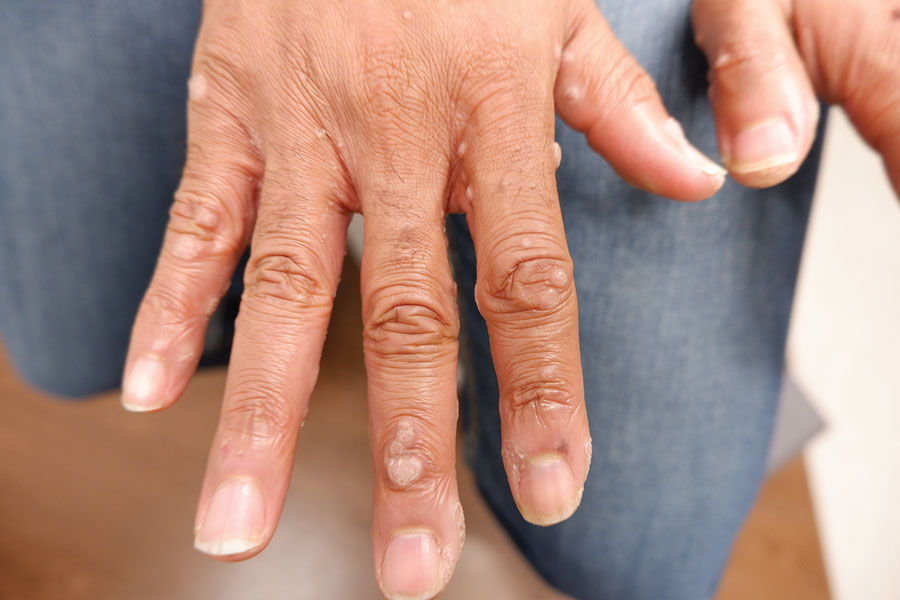
A 56-year-old man presented for evaluation of several cutaneous lesions that had developed over several months on the neck and over many years on the hands and feet. He had a 16-year history of HIV, Castleman disease, and primary effusion lymphoma in remission that was treated with rituximab, etoposide phosphate, prednisone, vincristine sulfate, cyclophosphamide, and doxorubicin hydrochloride 10 or more years ago. The patient denied pruritus or pain associated with the skin lesions. He was intermittently taking immunosuppressants and antiretrovirals including dolutegravir and emtricitabine-tenofovir for 3 years. Prior treatments of the lesions included cryotherapy and over-the-counter 17% salicylic acid. Physical examination revealed the presence of innumerable, clustered, verrucous, scaly papules on the dorsal and palmoplantar regions of the hands (Figure 1), as well as hypopigmented macules clustered on the neck that morphologically resembled tinea versicolor (Figure 2). The physical examination was otherwise unremarkable.
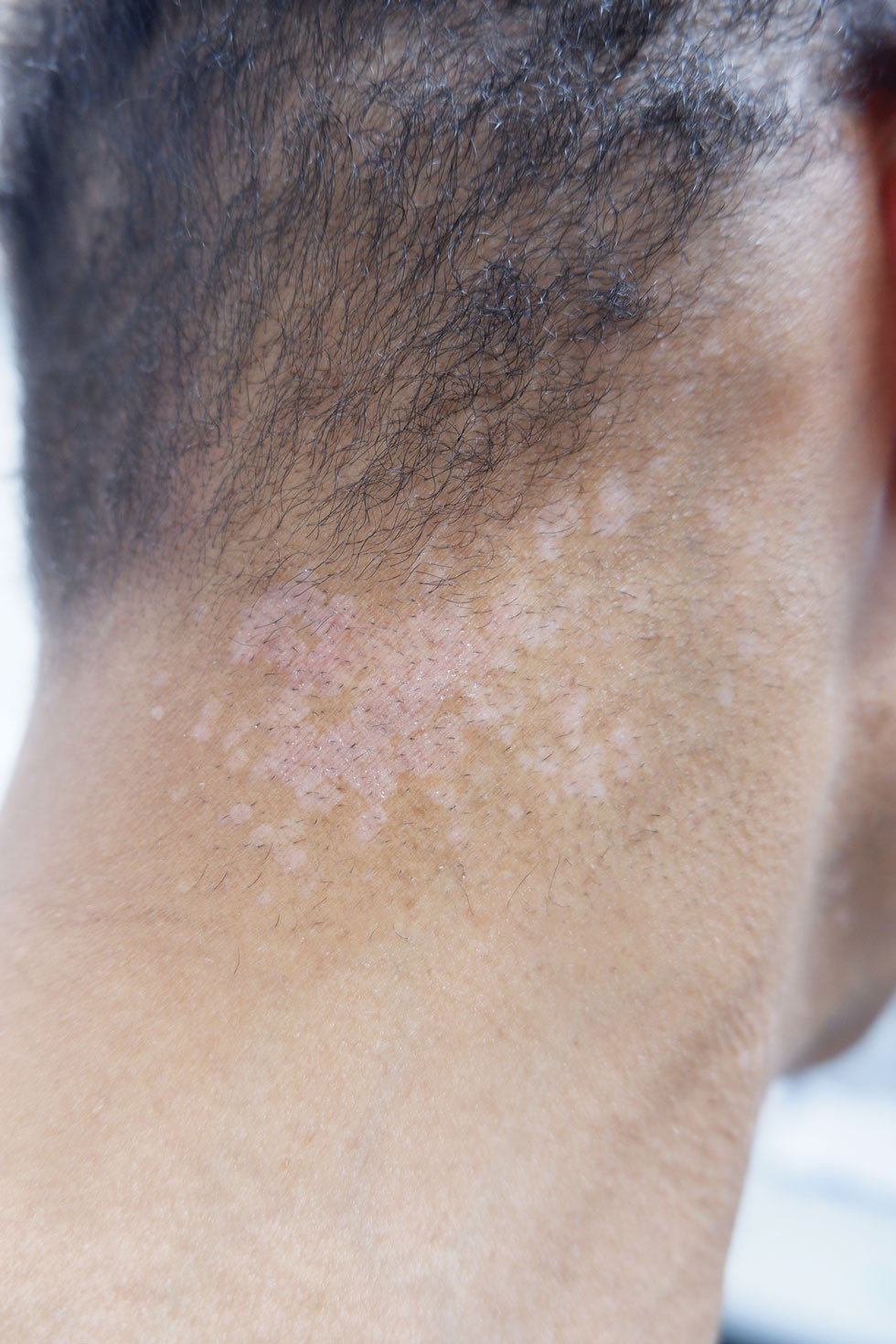
Complete blood cell counts as well as lipid, liver, and renal function panel results were unremarkable. Laboratory examination also revealed a CD4 cell count of 373/µL (reference range, 320–1900/µL) and an undetectable HIV copy number (<40 copies/mL). A punch biopsy of a hypopigmented macule on the left side of the neck revealed epidermal acanthosis, hypergranulosis, and hyperkeratosis, with blue-gray cytoplasm observed in the keratinocytes (Figure 3). Koilocytes with perinuclear clearing associated with keratinocytes in the upper epidermis were noted. Based on the clinical and histopathologic correlation, acquired EDV was diagnosed.
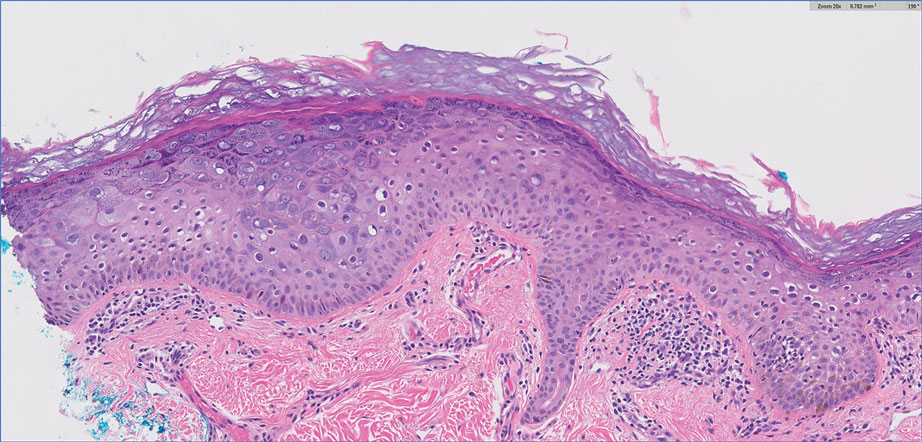
Given that HIV-associated EDV often is recalcitrant and there is a lack of consistent and effective treatment, the patient initially was prescribed oral acitretin 25 mg/d with intralesional C albicans antigen injected once per month into the lesions along with concurrent cryotherapy. At subsequent monthly follow-ups, the involved areas were notably thinner and flat. The patient reported no remarkable side effects from the systemic retinoid treatment such as abdominal pain, photosensitivity, or headaches, though he did experience mild xerosis. Complete resolution of EDV occurred with multimodal therapy—acitretin, cryotherapy, and intralesional Candida antigen. Palmar verrucae were much improved, and he is currently continuing therapy.
Epidermodysplasia verruciformis is a rare genodermatosis associated with an abnormal susceptibility to cutaneous HPV and can be acquired in immunocompromised patients. Patients with EDV present with a clinically heterogeneous disease that can manifest as hypopigmented, red-brown macules with scaling on the trunk, neck, and extremities, which are morphologically similar to tinea versicolor, or patients can present with flat wartlike papules that are most commonly found on the face, hands, and feet.2,3 Epidermodysplasia verruciformis can be distinguished from EDV-like eruptions and other generalized verrucoses by its characteristic histologic appearance and by the demonstration of HPV within the lesions, typically subtypes HPV-5 and HPV-8.1-3 Classic EDV histopathologic findings include mild to moderate acanthosis and hyperkeratosis with enlarged keratinocytes featuring blue-gray cytoplasm and perinuclear halos.1
The histologic differential diagnosis of EDV is quite broad and includes common verrucae, which may be distinguished by the absence of blue-gray discoloration of the cytoplasm among the individual keratinocytes.1 Verruca plana and condylomata also may mimic EDV, and patients may present with minimal papillomatosis of the surface epidermis.2 Squamous cell carcinoma in situ (SCC-IS) and particularly bowenoid papulosis also may share similar histologic features.2 However, in SCC-IS, there typically is full-thickness dysplasia of the epidermis, which is not present in EDV. Nonetheless, EDV is equivalent to SCC-IS in its clinical behavior. Bowenoid papulosis shares similar findings, but lesions generally are located in the genital areas and linked to HPV-16 and HPV-18.2 Additional histologic features of EDV have been described in the entity of EDV acanthoma, specifically incidental findings present in association with other cutaneous neoplasms including acantholytic acanthomas, condylomas, intradermal nevi, and seborrheic keratoses.12
The pathophysiology of EDV is thought to be specifically associated with patients with immunocompromised conditions. Particular attention has been paid to the association between EDV and HIV. Anselmo et al13 reported a case of HIV-associated acquired EDV with preexisting lesions that were spread along the distribution of the patient’s tattoo, suggesting potential autoinoculation. In individuals living with HIV, the cutaneous features of EDV are not associated with immune status.14
Acquired EDV also may be associated with other conditions including renal transplantation, IgM deficiency, severe combined immunodeficiency, common variable immunodeficiency, systemic lupus erythematosus, and myasthenia gravis.2 Hematologic malignancies such as Hodgkin disease,4 natural killer/T-cell lymphoma,5 cutaneous T-cell lymphoma,6 adult T-cell leukemia,7 intestinal diffuse large B-cell lymphoma,8,9 transformed acute myelogenous lymphoma,10 and chronic myelogenous leukemia11 also may be associated with EDV. In the inherited form, integral membrane proteins of the endoplasmic reticulum encoded by the genes EVER1 and EVER2 on chromosome 17 are thought to act as restriction factors for certain types of HPV.2,3 Inactivating mutations in EVER1 and EVER2 result in defects in cell-mediated immunity, rendering patients susceptible to both benign and oncogenic verrucous infections.2,3 Currently, it is believed that immunosuppressed states may result in defects in cell-mediated immunity that make patients similarly susceptible to these virulent strains of HPV, resulting in an acquired form of EDV.3 Interestingly, the clinical and histologic presentation is identical for acquired EDV and genetic EDV.
Due to the general resistance of EDV to treatment, a variety of options for acquired EDV have been explored including topical and systemic retinoids, cryotherapy, interferon alfa‐2a, zidovudine, ketoconazole, corticosteroids, podophyllotoxin, imiquimod, cidofovir, electrosurgery, 5‐fluorouracil, glycolic acid, temporized diathermy, and methyl aminolevulinate photodynamic therapy.3 Highly active antiretroviral therapy has been proposed as a potential treatment modality for HIV-associated cases; however, acquired EDV has been reported to develop as an immune reconstitution inflammatory syndrome after the initiation of highly active antiretroviral therapy.15
Combination therapy consisting of a systemic retinoid, immunotherapy, and cryotherapy was initiated for our patient. Human papillomavirus infection is marked by epithelial hyperplasia, and retinoids induce antiproliferation through the control of epithelial cell differentiation.16 The specific mechanism of action of retinoids in EDV treatment is unknown; however, the beneficial effects may result from the modification of terminal differentiation, a direct antiviral action, or the enhancement of killer T cells.17 Immunotherapy with C albicans antigen initiates an inflammatory reaction that leads to an immune response directed against the virus, thus reducing the number of warts.2 Cryotherapy aims to destroy the lesion but not the virus.2 The combination of systemic retinoids, immunotherapy, and destruction may target EDV via multiple potentially synergistic mechanisms. Thus, a multimodal approach can be beneficial in patients with recalcitrant acquired EDV.
The occurrence of EDV is rare, and data on treatment are limited in number resulting in general uncertainty about the efficacy of therapies. Elucidation of the specific mechanism of immunosuppression and its effects on T lymphocytes in acquired EDV may shed light on the most effective treatments. We present this novel case of a patient with HIV-associated acquired EDV who responded favorably to a combination treatment of acitretin, intralesional C albicans antigen, and cryotherapy.
- Nuovo GJ, Ishag M. The histologic spectrum of epidermodysplasia verruciformis. Am J Surg Pathol. 2000;24:1400-1406.
- Sri JC, Dubina MI, Kao GF, et al. Generalized verrucosis: a review of the associated diseases, evaluation, and treatments. J Am Acad Dermatol. 2012;66:292-311.
- Zampetti A, Giurdanella F, Manco S, et al. Acquired epidermodysplasia verruciformis: a comprehensive review and a proposal for treatment. Dermatol Surg. 2013;39:974-980.
- Gross G, Ellinger K, Roussaki A, et al. Epidermodysplasia verruciformis in a patient with Hodgkin’s disease: characterization of a new papillomavirus type and interferon treatment. J Invest Dermatol. 1988;91:43-48.
- Boran P, Tokuc G, Ozberk M, et al. Epidermodysplasia verruciformis associated with natural killer/T cell lymphoma. J Pediatr. 2010;156:340-340.e1.
- Cutlan JE, Rashid RM, Torres-Cabala C, et al. Epidermodysplasia verruciformis after cutaneous T-cell lymphoma: periungual presentation. Dermatol Online J. 2010;16:12.
- Kawai K, Egawa N, Kiyono T, et al. Epidermodysplasia-verruciformis-like eruption associated with gamma-papillomavirus infection in a patient with adult T-cell leukemia. Dermatology. 2009;219:274-278.
- Slawsky LD, Gilson RT, Hockley AJ, et al. Epidermodysplasia verruciformis associated with severe immunodeficiency, lymphoma, and disseminated molluscum contagiosum. J Am Acad Dermatol. 1992;27:448-450.
- Youssef M, Denguezli M, Ghariani N, et al. Epidermodysplasia verruciformis associated with intestinal lymphoma: a model of viral oncogenicity. Pediatr Dermatol. 2007;24:511-513.
- Kunishige JH, Hymes SR, Madkan V, et al. Epidermodysplasia verruciformis in the setting of graft-versus-host disease. J Am Acad Dermatol. 2007;57(5 suppl):S78-S80.
- Binkley GW. A case for diagnosis (epidermodysplasia verruciformis?) chronic myeloid leukemia. Arch Derm Syphilol. 1947;55:280-282.
- Ko CJ, Iftner T, Barr RJ, et al. Changes of epidermodysplasia verruciformis in benign skin lesions: the EV acanthoma. J Cutan Pathol. 2007;34:44-48.
- Anselmo F, Ansari U, Gagnier JM, et al. Verrucous lesions in an HIV-positive man. JAAD Case Reports. 2019;5:825-827.
- Huang S, Wu JH, Lewis DJ, et al. A novel approach to the classification of epidermodysplasia verruciformis. Int J Dermatol. 2018;57:1344-1350.
- Jacobelli S, Laude H, Carlotti A, et al. Epidermodysplasia verruciformis in human immunodeficiency virus-infected patients: a marker of human papillomavirus-related disorders not affected by antiretroviral therapy. Arch Dermatol. 2011;147:590-596.
- Limmer AL, Wu JH, Doan HQ, et al. Acquired epidermodysplasia verruciformis: a 10-year anniversary update. Br J Dermatol. 2020;182:790-792.
- Anadolu R, Oskay T, Erdem C, et al. Treatment of epidermodysplasia verruciformis with a combination of acitretin and interferon alfa-2a.J Am Acad Dermatol. 2001;45:296-299.
To the Editor:
Epidermodysplasia verruciformis (EDV) is a rare generalized form of epidermal dysplasia that is linked to certain subtypes of human papillomavirus (HPV) infection and inherited or acquired states of immunodeficiency.1-3 The inherited form most commonly manifests via autosomal-recessive inactivation of the EVER1 and EVER2 genes that encode integral membrane proteins in the endoplasmic reticulum, though cases of autosomal-dominant and X-linked inheritance have been reported.1-3 Acquired cases have been reported in patients lacking immunocompetency, including transplant recipients and patients living with HIV.4-11 We present the case of a patient with HIV-associated EDV who was treated successfully with intralesional Candida albicans antigen, oral acitretin, and cryotherapy.

A 56-year-old man presented for evaluation of several cutaneous lesions that had developed over several months on the neck and over many years on the hands and feet. He had a 16-year history of HIV, Castleman disease, and primary effusion lymphoma in remission that was treated with rituximab, etoposide phosphate, prednisone, vincristine sulfate, cyclophosphamide, and doxorubicin hydrochloride 10 or more years ago. The patient denied pruritus or pain associated with the skin lesions. He was intermittently taking immunosuppressants and antiretrovirals including dolutegravir and emtricitabine-tenofovir for 3 years. Prior treatments of the lesions included cryotherapy and over-the-counter 17% salicylic acid. Physical examination revealed the presence of innumerable, clustered, verrucous, scaly papules on the dorsal and palmoplantar regions of the hands (Figure 1), as well as hypopigmented macules clustered on the neck that morphologically resembled tinea versicolor (Figure 2). The physical examination was otherwise unremarkable.

Complete blood cell counts as well as lipid, liver, and renal function panel results were unremarkable. Laboratory examination also revealed a CD4 cell count of 373/µL (reference range, 320–1900/µL) and an undetectable HIV copy number (<40 copies/mL). A punch biopsy of a hypopigmented macule on the left side of the neck revealed epidermal acanthosis, hypergranulosis, and hyperkeratosis, with blue-gray cytoplasm observed in the keratinocytes (Figure 3). Koilocytes with perinuclear clearing associated with keratinocytes in the upper epidermis were noted. Based on the clinical and histopathologic correlation, acquired EDV was diagnosed.

Given that HIV-associated EDV often is recalcitrant and there is a lack of consistent and effective treatment, the patient initially was prescribed oral acitretin 25 mg/d with intralesional C albicans antigen injected once per month into the lesions along with concurrent cryotherapy. At subsequent monthly follow-ups, the involved areas were notably thinner and flat. The patient reported no remarkable side effects from the systemic retinoid treatment such as abdominal pain, photosensitivity, or headaches, though he did experience mild xerosis. Complete resolution of EDV occurred with multimodal therapy—acitretin, cryotherapy, and intralesional Candida antigen. Palmar verrucae were much improved, and he is currently continuing therapy.
Epidermodysplasia verruciformis is a rare genodermatosis associated with an abnormal susceptibility to cutaneous HPV and can be acquired in immunocompromised patients. Patients with EDV present with a clinically heterogeneous disease that can manifest as hypopigmented, red-brown macules with scaling on the trunk, neck, and extremities, which are morphologically similar to tinea versicolor, or patients can present with flat wartlike papules that are most commonly found on the face, hands, and feet.2,3 Epidermodysplasia verruciformis can be distinguished from EDV-like eruptions and other generalized verrucoses by its characteristic histologic appearance and by the demonstration of HPV within the lesions, typically subtypes HPV-5 and HPV-8.1-3 Classic EDV histopathologic findings include mild to moderate acanthosis and hyperkeratosis with enlarged keratinocytes featuring blue-gray cytoplasm and perinuclear halos.1
The histologic differential diagnosis of EDV is quite broad and includes common verrucae, which may be distinguished by the absence of blue-gray discoloration of the cytoplasm among the individual keratinocytes.1 Verruca plana and condylomata also may mimic EDV, and patients may present with minimal papillomatosis of the surface epidermis.2 Squamous cell carcinoma in situ (SCC-IS) and particularly bowenoid papulosis also may share similar histologic features.2 However, in SCC-IS, there typically is full-thickness dysplasia of the epidermis, which is not present in EDV. Nonetheless, EDV is equivalent to SCC-IS in its clinical behavior. Bowenoid papulosis shares similar findings, but lesions generally are located in the genital areas and linked to HPV-16 and HPV-18.2 Additional histologic features of EDV have been described in the entity of EDV acanthoma, specifically incidental findings present in association with other cutaneous neoplasms including acantholytic acanthomas, condylomas, intradermal nevi, and seborrheic keratoses.12
The pathophysiology of EDV is thought to be specifically associated with patients with immunocompromised conditions. Particular attention has been paid to the association between EDV and HIV. Anselmo et al13 reported a case of HIV-associated acquired EDV with preexisting lesions that were spread along the distribution of the patient’s tattoo, suggesting potential autoinoculation. In individuals living with HIV, the cutaneous features of EDV are not associated with immune status.14
Acquired EDV also may be associated with other conditions including renal transplantation, IgM deficiency, severe combined immunodeficiency, common variable immunodeficiency, systemic lupus erythematosus, and myasthenia gravis.2 Hematologic malignancies such as Hodgkin disease,4 natural killer/T-cell lymphoma,5 cutaneous T-cell lymphoma,6 adult T-cell leukemia,7 intestinal diffuse large B-cell lymphoma,8,9 transformed acute myelogenous lymphoma,10 and chronic myelogenous leukemia11 also may be associated with EDV. In the inherited form, integral membrane proteins of the endoplasmic reticulum encoded by the genes EVER1 and EVER2 on chromosome 17 are thought to act as restriction factors for certain types of HPV.2,3 Inactivating mutations in EVER1 and EVER2 result in defects in cell-mediated immunity, rendering patients susceptible to both benign and oncogenic verrucous infections.2,3 Currently, it is believed that immunosuppressed states may result in defects in cell-mediated immunity that make patients similarly susceptible to these virulent strains of HPV, resulting in an acquired form of EDV.3 Interestingly, the clinical and histologic presentation is identical for acquired EDV and genetic EDV.
Due to the general resistance of EDV to treatment, a variety of options for acquired EDV have been explored including topical and systemic retinoids, cryotherapy, interferon alfa‐2a, zidovudine, ketoconazole, corticosteroids, podophyllotoxin, imiquimod, cidofovir, electrosurgery, 5‐fluorouracil, glycolic acid, temporized diathermy, and methyl aminolevulinate photodynamic therapy.3 Highly active antiretroviral therapy has been proposed as a potential treatment modality for HIV-associated cases; however, acquired EDV has been reported to develop as an immune reconstitution inflammatory syndrome after the initiation of highly active antiretroviral therapy.15
Combination therapy consisting of a systemic retinoid, immunotherapy, and cryotherapy was initiated for our patient. Human papillomavirus infection is marked by epithelial hyperplasia, and retinoids induce antiproliferation through the control of epithelial cell differentiation.16 The specific mechanism of action of retinoids in EDV treatment is unknown; however, the beneficial effects may result from the modification of terminal differentiation, a direct antiviral action, or the enhancement of killer T cells.17 Immunotherapy with C albicans antigen initiates an inflammatory reaction that leads to an immune response directed against the virus, thus reducing the number of warts.2 Cryotherapy aims to destroy the lesion but not the virus.2 The combination of systemic retinoids, immunotherapy, and destruction may target EDV via multiple potentially synergistic mechanisms. Thus, a multimodal approach can be beneficial in patients with recalcitrant acquired EDV.
The occurrence of EDV is rare, and data on treatment are limited in number resulting in general uncertainty about the efficacy of therapies. Elucidation of the specific mechanism of immunosuppression and its effects on T lymphocytes in acquired EDV may shed light on the most effective treatments. We present this novel case of a patient with HIV-associated acquired EDV who responded favorably to a combination treatment of acitretin, intralesional C albicans antigen, and cryotherapy.
To the Editor:
Epidermodysplasia verruciformis (EDV) is a rare generalized form of epidermal dysplasia that is linked to certain subtypes of human papillomavirus (HPV) infection and inherited or acquired states of immunodeficiency.1-3 The inherited form most commonly manifests via autosomal-recessive inactivation of the EVER1 and EVER2 genes that encode integral membrane proteins in the endoplasmic reticulum, though cases of autosomal-dominant and X-linked inheritance have been reported.1-3 Acquired cases have been reported in patients lacking immunocompetency, including transplant recipients and patients living with HIV.4-11 We present the case of a patient with HIV-associated EDV who was treated successfully with intralesional Candida albicans antigen, oral acitretin, and cryotherapy.

A 56-year-old man presented for evaluation of several cutaneous lesions that had developed over several months on the neck and over many years on the hands and feet. He had a 16-year history of HIV, Castleman disease, and primary effusion lymphoma in remission that was treated with rituximab, etoposide phosphate, prednisone, vincristine sulfate, cyclophosphamide, and doxorubicin hydrochloride 10 or more years ago. The patient denied pruritus or pain associated with the skin lesions. He was intermittently taking immunosuppressants and antiretrovirals including dolutegravir and emtricitabine-tenofovir for 3 years. Prior treatments of the lesions included cryotherapy and over-the-counter 17% salicylic acid. Physical examination revealed the presence of innumerable, clustered, verrucous, scaly papules on the dorsal and palmoplantar regions of the hands (Figure 1), as well as hypopigmented macules clustered on the neck that morphologically resembled tinea versicolor (Figure 2). The physical examination was otherwise unremarkable.

Complete blood cell counts as well as lipid, liver, and renal function panel results were unremarkable. Laboratory examination also revealed a CD4 cell count of 373/µL (reference range, 320–1900/µL) and an undetectable HIV copy number (<40 copies/mL). A punch biopsy of a hypopigmented macule on the left side of the neck revealed epidermal acanthosis, hypergranulosis, and hyperkeratosis, with blue-gray cytoplasm observed in the keratinocytes (Figure 3). Koilocytes with perinuclear clearing associated with keratinocytes in the upper epidermis were noted. Based on the clinical and histopathologic correlation, acquired EDV was diagnosed.

Given that HIV-associated EDV often is recalcitrant and there is a lack of consistent and effective treatment, the patient initially was prescribed oral acitretin 25 mg/d with intralesional C albicans antigen injected once per month into the lesions along with concurrent cryotherapy. At subsequent monthly follow-ups, the involved areas were notably thinner and flat. The patient reported no remarkable side effects from the systemic retinoid treatment such as abdominal pain, photosensitivity, or headaches, though he did experience mild xerosis. Complete resolution of EDV occurred with multimodal therapy—acitretin, cryotherapy, and intralesional Candida antigen. Palmar verrucae were much improved, and he is currently continuing therapy.
Epidermodysplasia verruciformis is a rare genodermatosis associated with an abnormal susceptibility to cutaneous HPV and can be acquired in immunocompromised patients. Patients with EDV present with a clinically heterogeneous disease that can manifest as hypopigmented, red-brown macules with scaling on the trunk, neck, and extremities, which are morphologically similar to tinea versicolor, or patients can present with flat wartlike papules that are most commonly found on the face, hands, and feet.2,3 Epidermodysplasia verruciformis can be distinguished from EDV-like eruptions and other generalized verrucoses by its characteristic histologic appearance and by the demonstration of HPV within the lesions, typically subtypes HPV-5 and HPV-8.1-3 Classic EDV histopathologic findings include mild to moderate acanthosis and hyperkeratosis with enlarged keratinocytes featuring blue-gray cytoplasm and perinuclear halos.1
The histologic differential diagnosis of EDV is quite broad and includes common verrucae, which may be distinguished by the absence of blue-gray discoloration of the cytoplasm among the individual keratinocytes.1 Verruca plana and condylomata also may mimic EDV, and patients may present with minimal papillomatosis of the surface epidermis.2 Squamous cell carcinoma in situ (SCC-IS) and particularly bowenoid papulosis also may share similar histologic features.2 However, in SCC-IS, there typically is full-thickness dysplasia of the epidermis, which is not present in EDV. Nonetheless, EDV is equivalent to SCC-IS in its clinical behavior. Bowenoid papulosis shares similar findings, but lesions generally are located in the genital areas and linked to HPV-16 and HPV-18.2 Additional histologic features of EDV have been described in the entity of EDV acanthoma, specifically incidental findings present in association with other cutaneous neoplasms including acantholytic acanthomas, condylomas, intradermal nevi, and seborrheic keratoses.12
The pathophysiology of EDV is thought to be specifically associated with patients with immunocompromised conditions. Particular attention has been paid to the association between EDV and HIV. Anselmo et al13 reported a case of HIV-associated acquired EDV with preexisting lesions that were spread along the distribution of the patient’s tattoo, suggesting potential autoinoculation. In individuals living with HIV, the cutaneous features of EDV are not associated with immune status.14
Acquired EDV also may be associated with other conditions including renal transplantation, IgM deficiency, severe combined immunodeficiency, common variable immunodeficiency, systemic lupus erythematosus, and myasthenia gravis.2 Hematologic malignancies such as Hodgkin disease,4 natural killer/T-cell lymphoma,5 cutaneous T-cell lymphoma,6 adult T-cell leukemia,7 intestinal diffuse large B-cell lymphoma,8,9 transformed acute myelogenous lymphoma,10 and chronic myelogenous leukemia11 also may be associated with EDV. In the inherited form, integral membrane proteins of the endoplasmic reticulum encoded by the genes EVER1 and EVER2 on chromosome 17 are thought to act as restriction factors for certain types of HPV.2,3 Inactivating mutations in EVER1 and EVER2 result in defects in cell-mediated immunity, rendering patients susceptible to both benign and oncogenic verrucous infections.2,3 Currently, it is believed that immunosuppressed states may result in defects in cell-mediated immunity that make patients similarly susceptible to these virulent strains of HPV, resulting in an acquired form of EDV.3 Interestingly, the clinical and histologic presentation is identical for acquired EDV and genetic EDV.
Due to the general resistance of EDV to treatment, a variety of options for acquired EDV have been explored including topical and systemic retinoids, cryotherapy, interferon alfa‐2a, zidovudine, ketoconazole, corticosteroids, podophyllotoxin, imiquimod, cidofovir, electrosurgery, 5‐fluorouracil, glycolic acid, temporized diathermy, and methyl aminolevulinate photodynamic therapy.3 Highly active antiretroviral therapy has been proposed as a potential treatment modality for HIV-associated cases; however, acquired EDV has been reported to develop as an immune reconstitution inflammatory syndrome after the initiation of highly active antiretroviral therapy.15
Combination therapy consisting of a systemic retinoid, immunotherapy, and cryotherapy was initiated for our patient. Human papillomavirus infection is marked by epithelial hyperplasia, and retinoids induce antiproliferation through the control of epithelial cell differentiation.16 The specific mechanism of action of retinoids in EDV treatment is unknown; however, the beneficial effects may result from the modification of terminal differentiation, a direct antiviral action, or the enhancement of killer T cells.17 Immunotherapy with C albicans antigen initiates an inflammatory reaction that leads to an immune response directed against the virus, thus reducing the number of warts.2 Cryotherapy aims to destroy the lesion but not the virus.2 The combination of systemic retinoids, immunotherapy, and destruction may target EDV via multiple potentially synergistic mechanisms. Thus, a multimodal approach can be beneficial in patients with recalcitrant acquired EDV.
The occurrence of EDV is rare, and data on treatment are limited in number resulting in general uncertainty about the efficacy of therapies. Elucidation of the specific mechanism of immunosuppression and its effects on T lymphocytes in acquired EDV may shed light on the most effective treatments. We present this novel case of a patient with HIV-associated acquired EDV who responded favorably to a combination treatment of acitretin, intralesional C albicans antigen, and cryotherapy.
- Nuovo GJ, Ishag M. The histologic spectrum of epidermodysplasia verruciformis. Am J Surg Pathol. 2000;24:1400-1406.
- Sri JC, Dubina MI, Kao GF, et al. Generalized verrucosis: a review of the associated diseases, evaluation, and treatments. J Am Acad Dermatol. 2012;66:292-311.
- Zampetti A, Giurdanella F, Manco S, et al. Acquired epidermodysplasia verruciformis: a comprehensive review and a proposal for treatment. Dermatol Surg. 2013;39:974-980.
- Gross G, Ellinger K, Roussaki A, et al. Epidermodysplasia verruciformis in a patient with Hodgkin’s disease: characterization of a new papillomavirus type and interferon treatment. J Invest Dermatol. 1988;91:43-48.
- Boran P, Tokuc G, Ozberk M, et al. Epidermodysplasia verruciformis associated with natural killer/T cell lymphoma. J Pediatr. 2010;156:340-340.e1.
- Cutlan JE, Rashid RM, Torres-Cabala C, et al. Epidermodysplasia verruciformis after cutaneous T-cell lymphoma: periungual presentation. Dermatol Online J. 2010;16:12.
- Kawai K, Egawa N, Kiyono T, et al. Epidermodysplasia-verruciformis-like eruption associated with gamma-papillomavirus infection in a patient with adult T-cell leukemia. Dermatology. 2009;219:274-278.
- Slawsky LD, Gilson RT, Hockley AJ, et al. Epidermodysplasia verruciformis associated with severe immunodeficiency, lymphoma, and disseminated molluscum contagiosum. J Am Acad Dermatol. 1992;27:448-450.
- Youssef M, Denguezli M, Ghariani N, et al. Epidermodysplasia verruciformis associated with intestinal lymphoma: a model of viral oncogenicity. Pediatr Dermatol. 2007;24:511-513.
- Kunishige JH, Hymes SR, Madkan V, et al. Epidermodysplasia verruciformis in the setting of graft-versus-host disease. J Am Acad Dermatol. 2007;57(5 suppl):S78-S80.
- Binkley GW. A case for diagnosis (epidermodysplasia verruciformis?) chronic myeloid leukemia. Arch Derm Syphilol. 1947;55:280-282.
- Ko CJ, Iftner T, Barr RJ, et al. Changes of epidermodysplasia verruciformis in benign skin lesions: the EV acanthoma. J Cutan Pathol. 2007;34:44-48.
- Anselmo F, Ansari U, Gagnier JM, et al. Verrucous lesions in an HIV-positive man. JAAD Case Reports. 2019;5:825-827.
- Huang S, Wu JH, Lewis DJ, et al. A novel approach to the classification of epidermodysplasia verruciformis. Int J Dermatol. 2018;57:1344-1350.
- Jacobelli S, Laude H, Carlotti A, et al. Epidermodysplasia verruciformis in human immunodeficiency virus-infected patients: a marker of human papillomavirus-related disorders not affected by antiretroviral therapy. Arch Dermatol. 2011;147:590-596.
- Limmer AL, Wu JH, Doan HQ, et al. Acquired epidermodysplasia verruciformis: a 10-year anniversary update. Br J Dermatol. 2020;182:790-792.
- Anadolu R, Oskay T, Erdem C, et al. Treatment of epidermodysplasia verruciformis with a combination of acitretin and interferon alfa-2a.J Am Acad Dermatol. 2001;45:296-299.
- Nuovo GJ, Ishag M. The histologic spectrum of epidermodysplasia verruciformis. Am J Surg Pathol. 2000;24:1400-1406.
- Sri JC, Dubina MI, Kao GF, et al. Generalized verrucosis: a review of the associated diseases, evaluation, and treatments. J Am Acad Dermatol. 2012;66:292-311.
- Zampetti A, Giurdanella F, Manco S, et al. Acquired epidermodysplasia verruciformis: a comprehensive review and a proposal for treatment. Dermatol Surg. 2013;39:974-980.
- Gross G, Ellinger K, Roussaki A, et al. Epidermodysplasia verruciformis in a patient with Hodgkin’s disease: characterization of a new papillomavirus type and interferon treatment. J Invest Dermatol. 1988;91:43-48.
- Boran P, Tokuc G, Ozberk M, et al. Epidermodysplasia verruciformis associated with natural killer/T cell lymphoma. J Pediatr. 2010;156:340-340.e1.
- Cutlan JE, Rashid RM, Torres-Cabala C, et al. Epidermodysplasia verruciformis after cutaneous T-cell lymphoma: periungual presentation. Dermatol Online J. 2010;16:12.
- Kawai K, Egawa N, Kiyono T, et al. Epidermodysplasia-verruciformis-like eruption associated with gamma-papillomavirus infection in a patient with adult T-cell leukemia. Dermatology. 2009;219:274-278.
- Slawsky LD, Gilson RT, Hockley AJ, et al. Epidermodysplasia verruciformis associated with severe immunodeficiency, lymphoma, and disseminated molluscum contagiosum. J Am Acad Dermatol. 1992;27:448-450.
- Youssef M, Denguezli M, Ghariani N, et al. Epidermodysplasia verruciformis associated with intestinal lymphoma: a model of viral oncogenicity. Pediatr Dermatol. 2007;24:511-513.
- Kunishige JH, Hymes SR, Madkan V, et al. Epidermodysplasia verruciformis in the setting of graft-versus-host disease. J Am Acad Dermatol. 2007;57(5 suppl):S78-S80.
- Binkley GW. A case for diagnosis (epidermodysplasia verruciformis?) chronic myeloid leukemia. Arch Derm Syphilol. 1947;55:280-282.
- Ko CJ, Iftner T, Barr RJ, et al. Changes of epidermodysplasia verruciformis in benign skin lesions: the EV acanthoma. J Cutan Pathol. 2007;34:44-48.
- Anselmo F, Ansari U, Gagnier JM, et al. Verrucous lesions in an HIV-positive man. JAAD Case Reports. 2019;5:825-827.
- Huang S, Wu JH, Lewis DJ, et al. A novel approach to the classification of epidermodysplasia verruciformis. Int J Dermatol. 2018;57:1344-1350.
- Jacobelli S, Laude H, Carlotti A, et al. Epidermodysplasia verruciformis in human immunodeficiency virus-infected patients: a marker of human papillomavirus-related disorders not affected by antiretroviral therapy. Arch Dermatol. 2011;147:590-596.
- Limmer AL, Wu JH, Doan HQ, et al. Acquired epidermodysplasia verruciformis: a 10-year anniversary update. Br J Dermatol. 2020;182:790-792.
- Anadolu R, Oskay T, Erdem C, et al. Treatment of epidermodysplasia verruciformis with a combination of acitretin and interferon alfa-2a.J Am Acad Dermatol. 2001;45:296-299.
Practice Points
- Acquired epidermodysplasia verruciformis (EDV) is associated with immunocompromised patients with conditions such as HIV.
- Multimodal treatment of HIV-associated acquired EDV with acitretin, intralesional Candida albicans antigen, and cryotherapy may be efficacious for patients with recalcitrant disease.
Violaceous Nodules on the Leg in a Patient with HIV
The Diagnosis: Plasmablastic Lymphoma
A punch biopsy of one of the leg nodules with hematoxylin and eosin staining revealed sheets of medium to large cells with plasmacytic differentiation (Figure, A and B). Immunohistochemistry showed CD79, epithelial membrane antigen, multiple myeloma 1, and CD138 positivity, as well as CD-19 negativity and positive staining on Epstein-Barr virus (EBV) in situ hybridization (Figure, C). Ki-67 stained greater than 90% of the neoplastic cells. Neoplastic cells were found to be λ restricted on κ and λ immunohistochemistry. Human herpesvirus 8 (HHV-8), CD3, and CD20 stains were negative. Subsequent fluorescent in situ hybridization was positive for MYC/immunoglobulin heavy chain (MYC/IGH) rearrangement t(8;14), confirming a diagnosis of plasmablastic lymphoma (PBL).
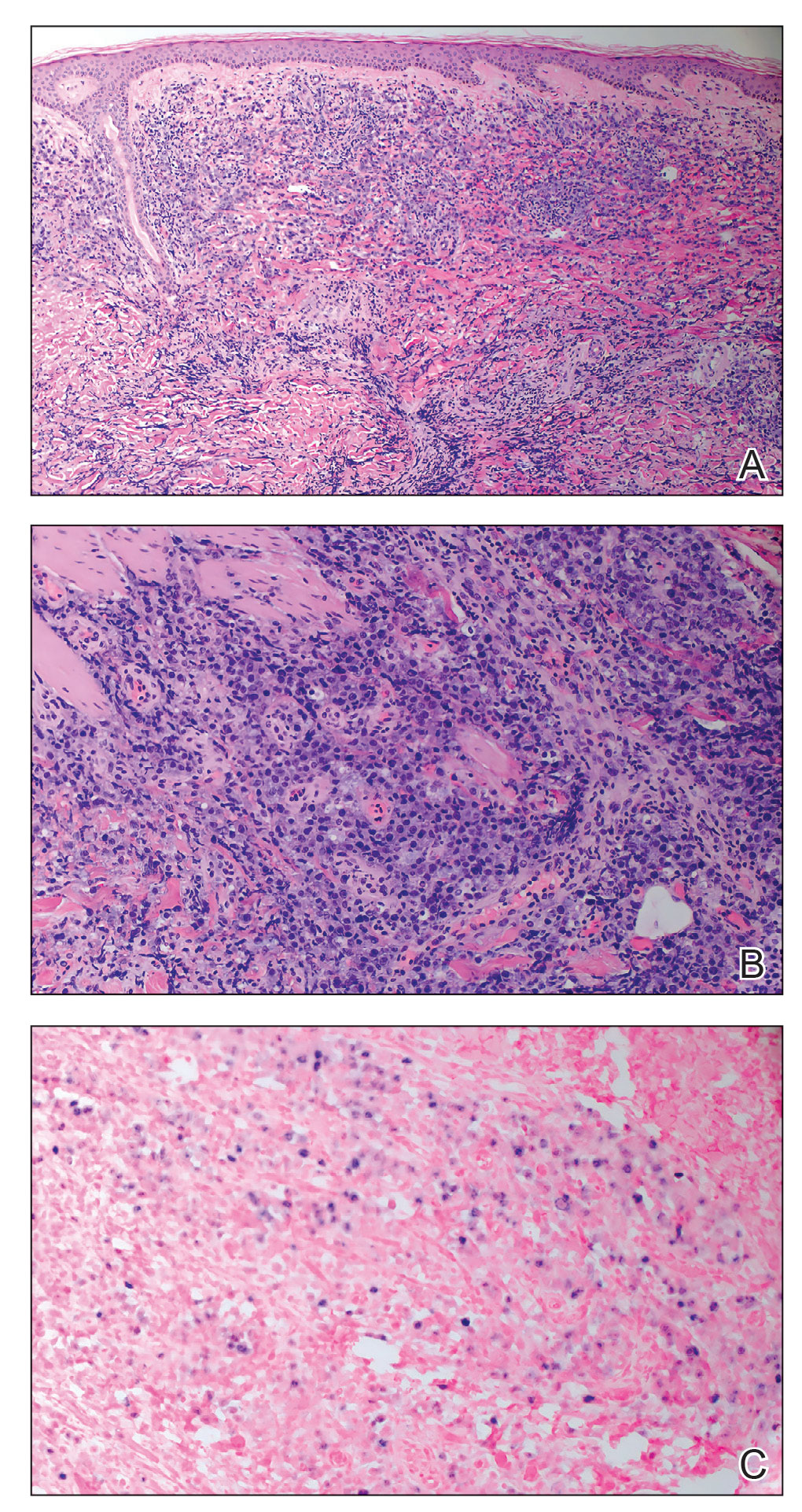
A bone marrow biopsy revealed normocellular bone marrow with trilineage hematopoiesis and no morphologic, immunophenotypic, or fluorescent in situ hybridization evidence of plasmablastic lymphoma or other pathology in the bone marrow. Our patient was started on hyper-CVAD (cyclophosphamide, vincristine, doxorubicin hydrochloride, dexamethasone) chemotherapy and was doing well with plans for a fourth course of chemotherapy. There is no standardized treatment course for cutaneous PBL, though excision with adjunctive chemotherapy treatment commonly has been reported in the literature.1
Plasmablastic lymphoma is a rare and aggressive diffuse large B-cell lymphoma associated with EBV infection that compromises approximately 2% to 3% of all HIV-related lymphomas.1,2 It frequently is associated with immunosuppression in patients with HIV or in transplant recipients on immunosuppression; however, it has been reported in immunocompetent individuals such as elderly patients.2 Plasmablastic lymphoma most commonly presents on the buccal mucosa but also can affect the gastrointestinal tract and occasionally has cutaneous manifestations.1,2 Cutaneous manifestations of PBL range from erythematous infiltrated plaques to ulcerated nodules presenting in an array of colors from flesh colored to violaceous.2 Primary cutaneous lesions can be seen on the legs, as in our patient.
Histopathologic examination reveals sheets of plasmablasts or large cells with eccentric nuclei and abundant basophilic cytoplasm.1 Plasmablastic lymphoma frequently is positive for mature B-cell markers such as CD38, CD138, multiple myeloma 1, and B lymphocyte–induced maturation protein 1.2,3 Uncommonly, PBL expresses paired box protein Pax-5 and CD20 markers.3 Although pathogenesis is poorly understood, it has been speculated that EBV infection is a common pathogenic factor. Epstein-Barr virus positivity has been noted in 60% of cases.2
Plasmablastic lymphoma and other malignant plasma cell processes such as plasmablastic myeloma (PBM) are morphologically similar. Proliferation of plasmablasts with rare plasmacytic cells is common in PBL, while plasmacytic cells are predominant in PBM. MYC rearrangement/ immunoglobulin heavy chain rearrangement t(8;14) was used to differentiate PBL from PBM in our patient; however, more cases of PBM with MYC/IGH rearrangement t(8;14) have been reported, making it an unreliable differentiating factor.4 A detailed clinical, pathologic, and genetic survey remains necessary for confirmatory diagnosis of PBL. Compared to other malignant plasma cell processes, PBL more commonly is seen in immunocompromised patients or those with HIV, such as our patient. Additionally, EBV testing is more likely to be positive in patients with PBL, further supporting this diagnosis in our patient.4
Presentations of bacillary angiomatosis, Kaposi sarcoma, and cutaneous lymphoma may be clinically similar; therefore, careful immunohistopathologic differentiation is necessary. Kaposi sarcoma is an angioproliferative disorder that develops from HHV-8 infection and commonly is associated with HIV. It presents as painless vascular lesions in a range of colors with typical progression from patch to plaque to nodules, frequently on the lower extremities. Histologically, admixtures of bland spindle cells, slitlike small vessel proliferation, and lymphocytic infiltration are typical. Neoplastic vessels lack basement membrane zones, resulting in microhemorrhages and hemosiderin deposition. Neoplastic vessels label with CD31 and CD34 endothelial markers in addition to HHV-8 antibodies, which is highly specific for Kaposi sarcoma and differentiates it from PBL.5
Bacillary angiomatosis is an infectious neovascular proliferation characterized by papular lesions that may resemble the lesions of PBL. Mixed cell infiltration in inflammatory cells with clumping of granular material is characteristic. Under Warthin-Starry staining, the granular material is abundant in gram-negative rods representing Bartonella species, which is the implicated infectious agent in bacillary angiomatosis.
Lymphomatoid papulosis (LyP) is the most common CD30+ lymphoproliferative disorder and also may present with exophytic nodules. The etiology of LyP remains unknown, but it is suspected that overexpression of CD30 plays a role. Lymphomatoid papulosis presents as red-violaceous papules and nodules in various stages of healing. Although variable histology among types of LyP exists, CD30+ T-cell lymphocytes remain the hallmark of LyP. Type A LyP, which accounts for 80% of LyP cases, reveals CD4+ and CD30+ cells scattered among neutrophils, eosinophils, and small lymphocytes.5 Lymphomatoid papulosis typically is self-healing, recurrent, and carries an excellent prognosis.
Plasmablastic lymphoma remains a rare and aggressive type of diffuse large B-cell lymphoma that can have primary cutaneous manifestations. It is prudent to consider PBL in the differential diagnosis of nodular lower extremity lesions, especially in immunosuppressed patients.
- Jambusaria A, Shafer D, Wu H, et al. Cutaneous plasmablastic lymphoma. J Am Acad Dermatol. 2008;58:676-678.
- Marques SA, Abbade LP, Guiotoku MM, et al. Primary cutaneous plasmablastic lymphoma revealing clinically unsuspected HIV infection. An Bras Dermatol. 2016;91:507-509.
- Bhatt R, Desai DS. Plasmablastic lymphoma. StatPearls. StatPearls Publishing; 2021. https://www.ncbi.nlm.nih.gov/books/NBK532975/
- Morris A, Monohan G. Plasmablastic myeloma versus plasmablastic lymphoma: different yet related diseases. Hematol Transfus Int J. 2018;6:25-28. doi:10.15406/htij.2018.06.00146
- Prieto-Torres L, Rodriguez-Pinilla SM, Onaindia A, et al. CD30-positive primary cutaneous lymphoproliferative disorders: molecular alterations and targeted therapies. Haematologica. 2019;104:226-235.
The Diagnosis: Plasmablastic Lymphoma
A punch biopsy of one of the leg nodules with hematoxylin and eosin staining revealed sheets of medium to large cells with plasmacytic differentiation (Figure, A and B). Immunohistochemistry showed CD79, epithelial membrane antigen, multiple myeloma 1, and CD138 positivity, as well as CD-19 negativity and positive staining on Epstein-Barr virus (EBV) in situ hybridization (Figure, C). Ki-67 stained greater than 90% of the neoplastic cells. Neoplastic cells were found to be λ restricted on κ and λ immunohistochemistry. Human herpesvirus 8 (HHV-8), CD3, and CD20 stains were negative. Subsequent fluorescent in situ hybridization was positive for MYC/immunoglobulin heavy chain (MYC/IGH) rearrangement t(8;14), confirming a diagnosis of plasmablastic lymphoma (PBL).

A bone marrow biopsy revealed normocellular bone marrow with trilineage hematopoiesis and no morphologic, immunophenotypic, or fluorescent in situ hybridization evidence of plasmablastic lymphoma or other pathology in the bone marrow. Our patient was started on hyper-CVAD (cyclophosphamide, vincristine, doxorubicin hydrochloride, dexamethasone) chemotherapy and was doing well with plans for a fourth course of chemotherapy. There is no standardized treatment course for cutaneous PBL, though excision with adjunctive chemotherapy treatment commonly has been reported in the literature.1
Plasmablastic lymphoma is a rare and aggressive diffuse large B-cell lymphoma associated with EBV infection that compromises approximately 2% to 3% of all HIV-related lymphomas.1,2 It frequently is associated with immunosuppression in patients with HIV or in transplant recipients on immunosuppression; however, it has been reported in immunocompetent individuals such as elderly patients.2 Plasmablastic lymphoma most commonly presents on the buccal mucosa but also can affect the gastrointestinal tract and occasionally has cutaneous manifestations.1,2 Cutaneous manifestations of PBL range from erythematous infiltrated plaques to ulcerated nodules presenting in an array of colors from flesh colored to violaceous.2 Primary cutaneous lesions can be seen on the legs, as in our patient.
Histopathologic examination reveals sheets of plasmablasts or large cells with eccentric nuclei and abundant basophilic cytoplasm.1 Plasmablastic lymphoma frequently is positive for mature B-cell markers such as CD38, CD138, multiple myeloma 1, and B lymphocyte–induced maturation protein 1.2,3 Uncommonly, PBL expresses paired box protein Pax-5 and CD20 markers.3 Although pathogenesis is poorly understood, it has been speculated that EBV infection is a common pathogenic factor. Epstein-Barr virus positivity has been noted in 60% of cases.2
Plasmablastic lymphoma and other malignant plasma cell processes such as plasmablastic myeloma (PBM) are morphologically similar. Proliferation of plasmablasts with rare plasmacytic cells is common in PBL, while plasmacytic cells are predominant in PBM. MYC rearrangement/ immunoglobulin heavy chain rearrangement t(8;14) was used to differentiate PBL from PBM in our patient; however, more cases of PBM with MYC/IGH rearrangement t(8;14) have been reported, making it an unreliable differentiating factor.4 A detailed clinical, pathologic, and genetic survey remains necessary for confirmatory diagnosis of PBL. Compared to other malignant plasma cell processes, PBL more commonly is seen in immunocompromised patients or those with HIV, such as our patient. Additionally, EBV testing is more likely to be positive in patients with PBL, further supporting this diagnosis in our patient.4
Presentations of bacillary angiomatosis, Kaposi sarcoma, and cutaneous lymphoma may be clinically similar; therefore, careful immunohistopathologic differentiation is necessary. Kaposi sarcoma is an angioproliferative disorder that develops from HHV-8 infection and commonly is associated with HIV. It presents as painless vascular lesions in a range of colors with typical progression from patch to plaque to nodules, frequently on the lower extremities. Histologically, admixtures of bland spindle cells, slitlike small vessel proliferation, and lymphocytic infiltration are typical. Neoplastic vessels lack basement membrane zones, resulting in microhemorrhages and hemosiderin deposition. Neoplastic vessels label with CD31 and CD34 endothelial markers in addition to HHV-8 antibodies, which is highly specific for Kaposi sarcoma and differentiates it from PBL.5
Bacillary angiomatosis is an infectious neovascular proliferation characterized by papular lesions that may resemble the lesions of PBL. Mixed cell infiltration in inflammatory cells with clumping of granular material is characteristic. Under Warthin-Starry staining, the granular material is abundant in gram-negative rods representing Bartonella species, which is the implicated infectious agent in bacillary angiomatosis.
Lymphomatoid papulosis (LyP) is the most common CD30+ lymphoproliferative disorder and also may present with exophytic nodules. The etiology of LyP remains unknown, but it is suspected that overexpression of CD30 plays a role. Lymphomatoid papulosis presents as red-violaceous papules and nodules in various stages of healing. Although variable histology among types of LyP exists, CD30+ T-cell lymphocytes remain the hallmark of LyP. Type A LyP, which accounts for 80% of LyP cases, reveals CD4+ and CD30+ cells scattered among neutrophils, eosinophils, and small lymphocytes.5 Lymphomatoid papulosis typically is self-healing, recurrent, and carries an excellent prognosis.
Plasmablastic lymphoma remains a rare and aggressive type of diffuse large B-cell lymphoma that can have primary cutaneous manifestations. It is prudent to consider PBL in the differential diagnosis of nodular lower extremity lesions, especially in immunosuppressed patients.
The Diagnosis: Plasmablastic Lymphoma
A punch biopsy of one of the leg nodules with hematoxylin and eosin staining revealed sheets of medium to large cells with plasmacytic differentiation (Figure, A and B). Immunohistochemistry showed CD79, epithelial membrane antigen, multiple myeloma 1, and CD138 positivity, as well as CD-19 negativity and positive staining on Epstein-Barr virus (EBV) in situ hybridization (Figure, C). Ki-67 stained greater than 90% of the neoplastic cells. Neoplastic cells were found to be λ restricted on κ and λ immunohistochemistry. Human herpesvirus 8 (HHV-8), CD3, and CD20 stains were negative. Subsequent fluorescent in situ hybridization was positive for MYC/immunoglobulin heavy chain (MYC/IGH) rearrangement t(8;14), confirming a diagnosis of plasmablastic lymphoma (PBL).

A bone marrow biopsy revealed normocellular bone marrow with trilineage hematopoiesis and no morphologic, immunophenotypic, or fluorescent in situ hybridization evidence of plasmablastic lymphoma or other pathology in the bone marrow. Our patient was started on hyper-CVAD (cyclophosphamide, vincristine, doxorubicin hydrochloride, dexamethasone) chemotherapy and was doing well with plans for a fourth course of chemotherapy. There is no standardized treatment course for cutaneous PBL, though excision with adjunctive chemotherapy treatment commonly has been reported in the literature.1
Plasmablastic lymphoma is a rare and aggressive diffuse large B-cell lymphoma associated with EBV infection that compromises approximately 2% to 3% of all HIV-related lymphomas.1,2 It frequently is associated with immunosuppression in patients with HIV or in transplant recipients on immunosuppression; however, it has been reported in immunocompetent individuals such as elderly patients.2 Plasmablastic lymphoma most commonly presents on the buccal mucosa but also can affect the gastrointestinal tract and occasionally has cutaneous manifestations.1,2 Cutaneous manifestations of PBL range from erythematous infiltrated plaques to ulcerated nodules presenting in an array of colors from flesh colored to violaceous.2 Primary cutaneous lesions can be seen on the legs, as in our patient.
Histopathologic examination reveals sheets of plasmablasts or large cells with eccentric nuclei and abundant basophilic cytoplasm.1 Plasmablastic lymphoma frequently is positive for mature B-cell markers such as CD38, CD138, multiple myeloma 1, and B lymphocyte–induced maturation protein 1.2,3 Uncommonly, PBL expresses paired box protein Pax-5 and CD20 markers.3 Although pathogenesis is poorly understood, it has been speculated that EBV infection is a common pathogenic factor. Epstein-Barr virus positivity has been noted in 60% of cases.2
Plasmablastic lymphoma and other malignant plasma cell processes such as plasmablastic myeloma (PBM) are morphologically similar. Proliferation of plasmablasts with rare plasmacytic cells is common in PBL, while plasmacytic cells are predominant in PBM. MYC rearrangement/ immunoglobulin heavy chain rearrangement t(8;14) was used to differentiate PBL from PBM in our patient; however, more cases of PBM with MYC/IGH rearrangement t(8;14) have been reported, making it an unreliable differentiating factor.4 A detailed clinical, pathologic, and genetic survey remains necessary for confirmatory diagnosis of PBL. Compared to other malignant plasma cell processes, PBL more commonly is seen in immunocompromised patients or those with HIV, such as our patient. Additionally, EBV testing is more likely to be positive in patients with PBL, further supporting this diagnosis in our patient.4
Presentations of bacillary angiomatosis, Kaposi sarcoma, and cutaneous lymphoma may be clinically similar; therefore, careful immunohistopathologic differentiation is necessary. Kaposi sarcoma is an angioproliferative disorder that develops from HHV-8 infection and commonly is associated with HIV. It presents as painless vascular lesions in a range of colors with typical progression from patch to plaque to nodules, frequently on the lower extremities. Histologically, admixtures of bland spindle cells, slitlike small vessel proliferation, and lymphocytic infiltration are typical. Neoplastic vessels lack basement membrane zones, resulting in microhemorrhages and hemosiderin deposition. Neoplastic vessels label with CD31 and CD34 endothelial markers in addition to HHV-8 antibodies, which is highly specific for Kaposi sarcoma and differentiates it from PBL.5
Bacillary angiomatosis is an infectious neovascular proliferation characterized by papular lesions that may resemble the lesions of PBL. Mixed cell infiltration in inflammatory cells with clumping of granular material is characteristic. Under Warthin-Starry staining, the granular material is abundant in gram-negative rods representing Bartonella species, which is the implicated infectious agent in bacillary angiomatosis.
Lymphomatoid papulosis (LyP) is the most common CD30+ lymphoproliferative disorder and also may present with exophytic nodules. The etiology of LyP remains unknown, but it is suspected that overexpression of CD30 plays a role. Lymphomatoid papulosis presents as red-violaceous papules and nodules in various stages of healing. Although variable histology among types of LyP exists, CD30+ T-cell lymphocytes remain the hallmark of LyP. Type A LyP, which accounts for 80% of LyP cases, reveals CD4+ and CD30+ cells scattered among neutrophils, eosinophils, and small lymphocytes.5 Lymphomatoid papulosis typically is self-healing, recurrent, and carries an excellent prognosis.
Plasmablastic lymphoma remains a rare and aggressive type of diffuse large B-cell lymphoma that can have primary cutaneous manifestations. It is prudent to consider PBL in the differential diagnosis of nodular lower extremity lesions, especially in immunosuppressed patients.
- Jambusaria A, Shafer D, Wu H, et al. Cutaneous plasmablastic lymphoma. J Am Acad Dermatol. 2008;58:676-678.
- Marques SA, Abbade LP, Guiotoku MM, et al. Primary cutaneous plasmablastic lymphoma revealing clinically unsuspected HIV infection. An Bras Dermatol. 2016;91:507-509.
- Bhatt R, Desai DS. Plasmablastic lymphoma. StatPearls. StatPearls Publishing; 2021. https://www.ncbi.nlm.nih.gov/books/NBK532975/
- Morris A, Monohan G. Plasmablastic myeloma versus plasmablastic lymphoma: different yet related diseases. Hematol Transfus Int J. 2018;6:25-28. doi:10.15406/htij.2018.06.00146
- Prieto-Torres L, Rodriguez-Pinilla SM, Onaindia A, et al. CD30-positive primary cutaneous lymphoproliferative disorders: molecular alterations and targeted therapies. Haematologica. 2019;104:226-235.
- Jambusaria A, Shafer D, Wu H, et al. Cutaneous plasmablastic lymphoma. J Am Acad Dermatol. 2008;58:676-678.
- Marques SA, Abbade LP, Guiotoku MM, et al. Primary cutaneous plasmablastic lymphoma revealing clinically unsuspected HIV infection. An Bras Dermatol. 2016;91:507-509.
- Bhatt R, Desai DS. Plasmablastic lymphoma. StatPearls. StatPearls Publishing; 2021. https://www.ncbi.nlm.nih.gov/books/NBK532975/
- Morris A, Monohan G. Plasmablastic myeloma versus plasmablastic lymphoma: different yet related diseases. Hematol Transfus Int J. 2018;6:25-28. doi:10.15406/htij.2018.06.00146
- Prieto-Torres L, Rodriguez-Pinilla SM, Onaindia A, et al. CD30-positive primary cutaneous lymphoproliferative disorders: molecular alterations and targeted therapies. Haematologica. 2019;104:226-235.
A 67-year-old man with long-standing hepatitis B virus and HIV managed with chronic antiretroviral therapy presented to an urgent care facility with worsening erythema and edema of the legs of 2 weeks’ duration. He was prescribed a 7-day course of cephalexin for presumed cellulitis. Two months later, he developed nodules on the lower extremities. He was seen by podiatry and prescribed a course of amoxicillin–clavulanic acid for presumed infection. Despite 2 courses of antibiotics, his symptoms progressed. The nodules expanded in number and some developed ulceration. Three months into his clinical course, he presented to our dermatology clinic. Physical examination revealed two 2- to 3-cm, violaceous, exophytic, tender nodules. He reported tactile allodynia of the lower extremities and denied fever, chills, night sweats, or weight loss. He also denied exposure to infectious or chemical agents and reported no recent travel. The patient was chronically taking lisinopril/hydrochlorothiazide, escitalopram, elvitegravir/cobicistat/emtricitabine/tenofovir alafenamide, bupropion, and aspirin with no recent changes. A complete hematologic and biochemical survey largely was unremarkable. His HIV viral load was undetectable with a CD4 count greater than 400/mm3 (reference range, 490–1436/mm3). Lactate dehydrogenase was elevated at 568 IU/L (reference range, 135–225 IU/L). The lower leg lesions were biopsied for confirmatory diagnosis.
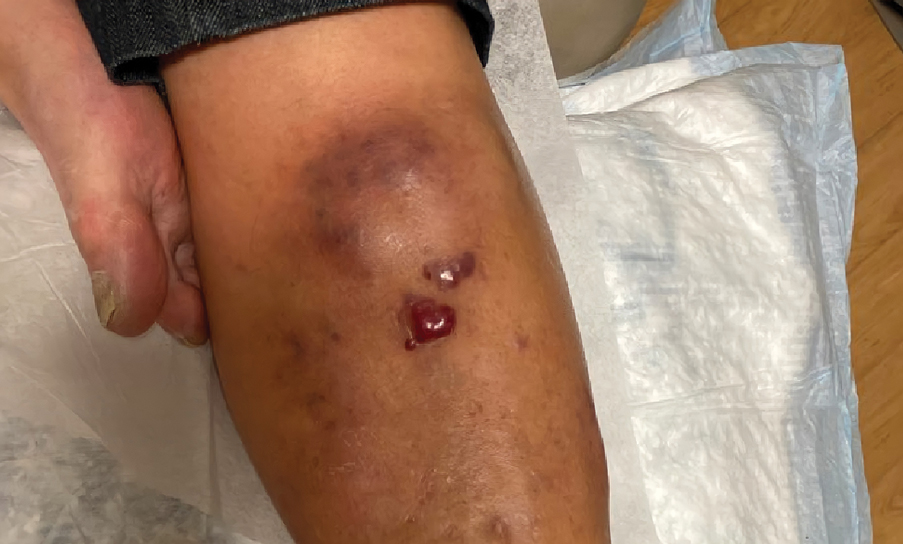
Ticking Time: Spreading Awareness About African Tick-Bite Fever
To the Editor:
One of the more common tick-borne infections seen in travelers returning from sub-Saharan Africa is caused by Rickettsia africae, which is the etiologic agent of African tick-bite fever (ATBF), the most common tick-borne bacterial zoonosis.1 There are 2 known tick vectors of disease: Amblyomma variegatum, found in sub-Saharan Africa and the West Indies, and Amblyomma hebraeum, found specifically in southern Africa.2,3
Unlike other disease-carrying ticks that passively wait on vegetation to be picked up by a host, A hebraeum uniquely attract other nearby ticks to the host. Studies have shown that male ticks feeding on a nonhuman host (usually cattle) can emit an aggression-attachment pheromone that attracts other ticks to the host. The presence of the pheromone allows unfed ticks to actively discriminate between hosts on which these parasites have fed successfully (ie, suitable hosts) and those on which they have not.4
The aggressive hunting nature of A hebraeum explains the clinical presentation of multiple eschars and why large groups of exposed travelers—such as soldiers, leisure safari tourists, game hunters, and foreign-aid workers—are affected.2
Another southern African spotted fever group, Rickettsia conorii is the causative agent of Mediterranean spotted fever (MSF). Ticks carrying R conorii exhibit a much less aggressive hunting stylethan A hebraeum; consequently, infected patients present with a single eschar site.5
Rickettsia africae is estimated to have very high prevalence (95.2%) in Amblyomma ticks and a fairly high prevalence (approximately 4.0% to 8.6%) in travelers coming from rural southern Africa,6,7 with an incubation period of 5 to 10 days after inoculation by an infected tick.8 Signs include fever, a generalized maculopapular or papulovesicular rash, and regional lymphadenopathy; symptoms include fatigue, headache, and myalgia.
The inoculation eschar—single or multiple—commonly presents on the legs and is accompanied by tender lymphadenopathy of draining nodes1,8 More severe findings, such as myocarditis and subacute neuropathy, have been reported in elderly patients.9
A 77-year-old man presented with a pruritic maculopapular and papulovesicular rash distributed over the upper and lower extremities of 3 weeks’ duration. The patient reported having been on a 12-day mission trip to Limpopo, South Africa, where he was constructing and installing safe toilets to replace dangerous toilet pits. He believed he had been bitten by 2 ticks, after which he noted a dark purple and black patch on the left lower leg by the third day of the trip. He developed a sudden persistent pruritic rash, first on the lower extremities and then spreading to the upper extremities. The patient was seen by an American physician in South Africa who gave him a 7-day course of oral doxycycline monohydrate 100 mg twice daily. He then returned to the United States.
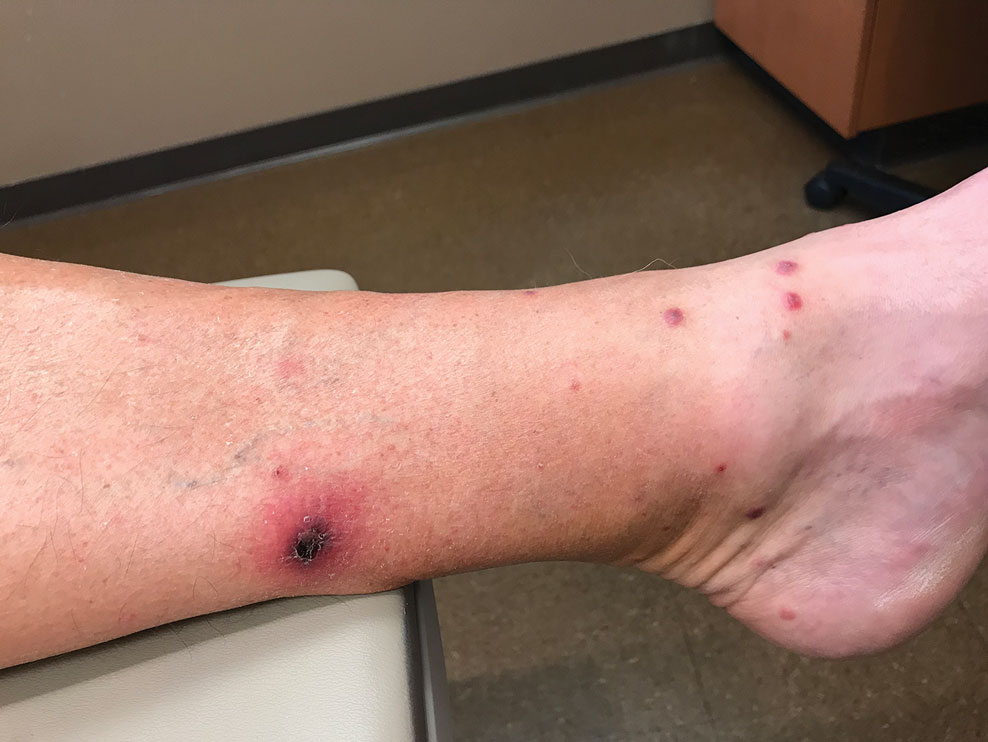
Sixteen days after being bitten by the ticks, the patient was examined in our dermatology office. Physical examination revealed an erythematous plaque with a central eschar over the medial aspect of the left leg (Figure 1) and multiple 3- to 6-mm, erythematous, dome-shaped papules scattered over the dorsal aspects of the feet and ankles (Figure 2). The examination was otherwise normal. Blood was drawn the same day for laboratory analysis; no abnormalities of platelets, red blood cells, or white blood cells were found. Results of a chemistry panel and liver enzyme tests were within reference range.
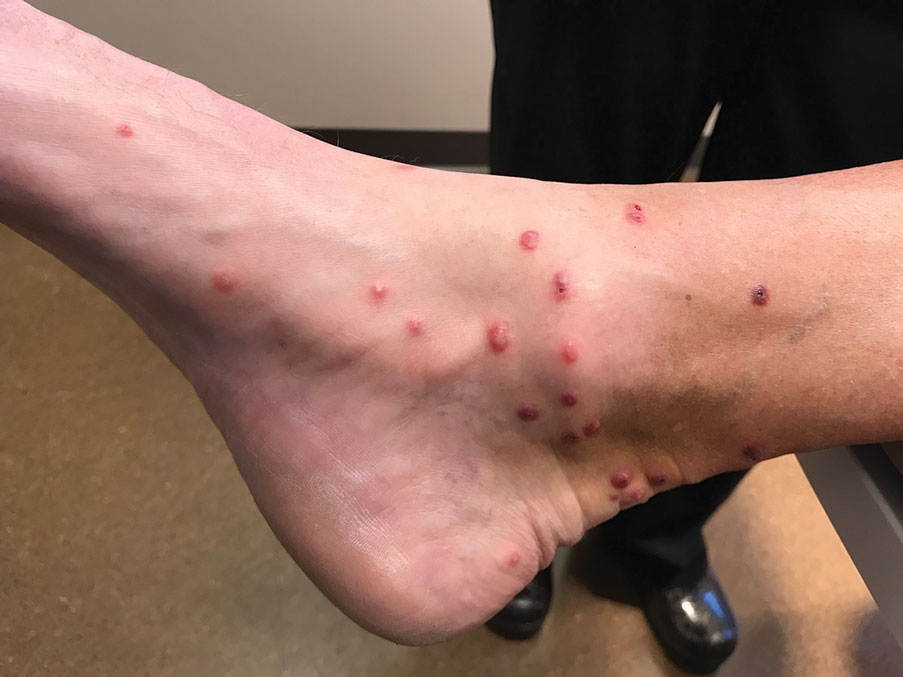
Skin biopsies were taken to elucidate the underlying pathology. Although an arthropod assault was suspected, there also was concern for deep vessel vasculitis because of the presence of considerable petechiae and purpura (Figure 3). Histologically, leukocytoclasia was seen in deep dermal blood vessels. A mild eosinophilic spongiosis with a mixed dermal infiltrate was identified—strengthening our suspicion of an arthropod assault. Bacterial cultures for aerobes and anaerobes using material taken from the right shin showed no growth.
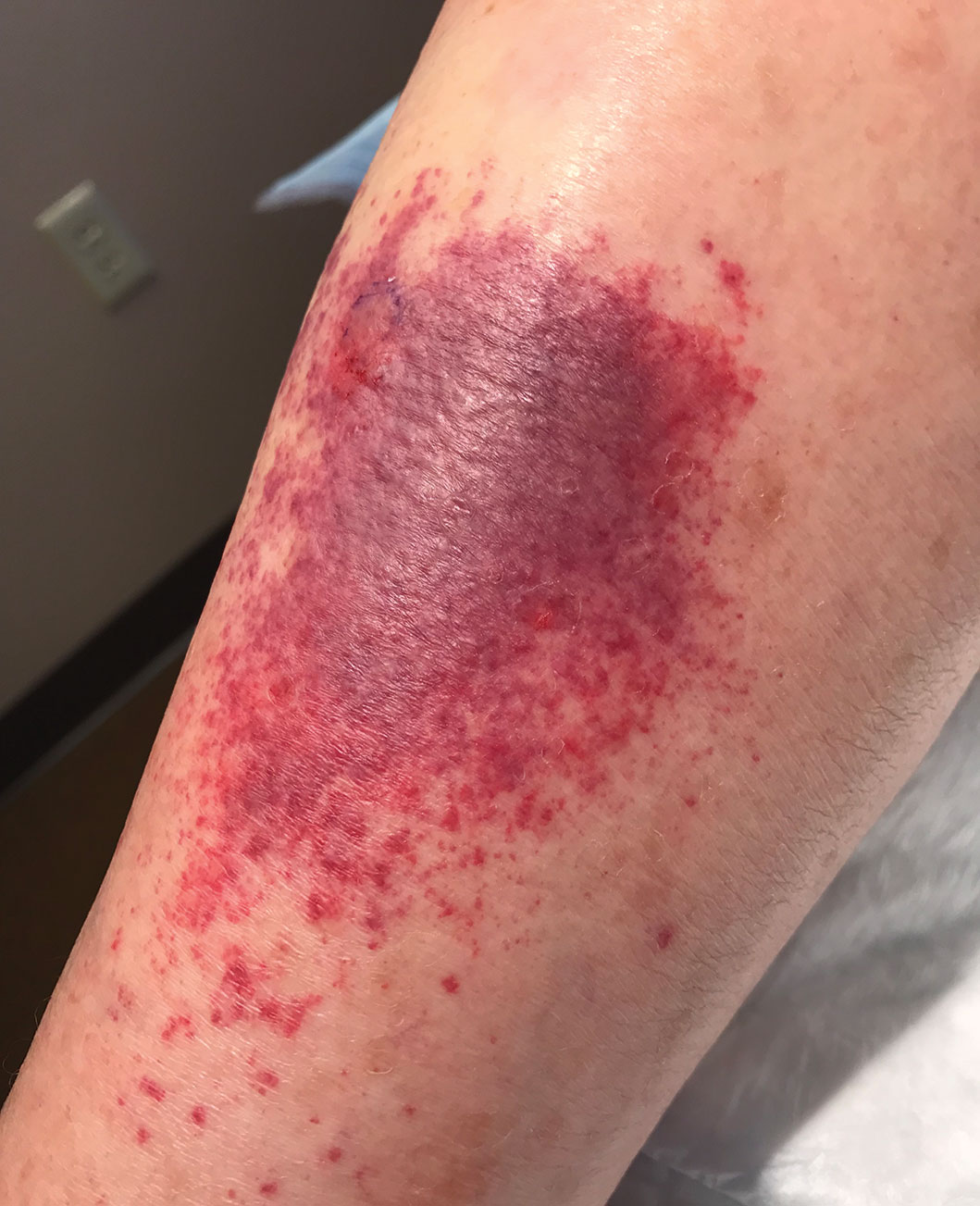
Ten days after the initial biopsies, serum specimens were drawn and swabs of eschar were collected and sent to the Centers for Disease Control and Prevention for further testing. Serum was tested by immunofluorescence assay (IFA) for spotted fever group IgG to detect Rocky Mountain spotted fever and ATBF antibodies; both tests were negative. Swab material from eschar was tested again by IFA for spotted fever group IgG (Rocky Mountain spotted fever) and antibodies to ATBF and with bacterial culture isolation and nucleic acid amplification; the culture and amplification came back positive for R africae.
Because the specialized tests confirmed infection with R africae, the patient was given triamcinolone cream 0.1% to apply twice daily to the pruritic lesions for as long as 4 weeks; an additional 14-day course of oral doxycycline monohydrate (100 mg twice daily) was given. At follow-up, the lesions had fully resolved without evident scarring.
Various diagnostic techniques can detect R africae. Bacterial culture and the polymerase chain reaction are specific and therefore diagnostic. In addition, the diagnosis of rickettsiosis can be made with serology testing, in which disease-specific antibodies are detected by indirect IFA using disease-specific antigens.
Antigens from R rickettsii (the agent of Rocky Mountain spotted fever), R conorii (Mediterranean spotted fever), and R africae (ATBF) are commercially available for making the diagnosis of rickettsiosis. However, antigens from R conorii exhibit cross-reactivity with R africae, which can confound the diagnosis.1,10 Serologic IFA tests have been shown to be less sensitive, especially when performed after antibacterial treatment has started.
In a study, 17 of 65 (26%) ATBF-confirmed patients were seronegative (acute and convalescent-phase sera) against R africae; 14 had received doxycycline during the first week of clinical signs. The current hypothesis is that R africae is highly sensitive to doxycycline and that early exposure to the drug prevented development of detectable titers of reactive antibodies, thus producing a negative serology test.11
Furthermore, it has been shown that seroconversion of IgG and IgM antibodies in R africae–infected sera is delayed compared to what is observed with R conorii–infected sera. Typically, seroconversion of R conorii–infected sera can be detected within 2 weeks; seroconversion in R africae–infected sera can take 4 weeks or longer.11
Our patient had a confirmed case of ATBF secondary to R africae infection, which was evident from tissue culture isolation and polymerase chain reaction analysis of swab material obtained from eschar sites, both of which yielded a positive result for R africae. The traveler’s negative serologic status might be due to his early exposure to doxycycline or to the 4-week delay in R africae seroconversion; his serum was collected only 3 weeks after the tick bites.
Clinical signs also aid in making the diagnosis of ATBF and distinguishing R conorii from R africae infection. Because of the aggressive hunting nature of the tick carrying R africae, they are associated with multiple eschars and tend to affect groups of multiple people, especially in rural areas.4,5 In contrast, ticks carrying R conorii yield a single eschar due to their passive style of infecting a host and because they favor a single host within urban areas.5 Both infections exhibit a maculopapular or papulovesicular rash and are accompanied by fatigue, headache, and myalgia, though ATBF tends to present with a milder rash than MSF.
Infection with either R conorii or R africae responds to tetracyclines, quinolones, and macrolides.10,12
African tick-bite fever is becoming more common, which should encourage clinicians to become familiar with the disease. Less than 2 decades ago, ATBF virtually was unknown outside of Zimbabwe, Botswana, Tanzania, Zambia, and Kenya, where it is endemic. However, after the abolition of apartheid in the 1990s, international tourism in southern Africa increased 6-fold.13 African tick-bite fever is now one of the most common rickettsial infections in Africa.7 In addition to diagnosing ATBF and managing infected patients, clinicians can help prevent ATBF in individuals who travel to endemic areas by recommending commercial topical insect repellents containing at least 19.5% N,N-diethyl-meta-toluamide (DEET).14
- Parola P, Paddock CD, Socolovschi C, et al. Update on tick-borne rickettsioses around the world: a geographic approach. Clin Microbiol Rev. 2013;26:657-702. doi:10.1128/CMR.00032-13
- Jensenius M, Fournier P-E, Vene S, et al; Norwegian African Tick Bite Fever Study Group. African tick bite fever in travelers to rural sub-equatorial Africa. Clin Infect Dis. 2003;36:1411-1417. doi:10.1086/375083
- Parola P, Jourdan J, Raoult D. Tick-borne infection caused by Rickettsia africae in the West Indies. N Engl J Med. 1998;338:1391-1392. doi:10.1056/NEJM199805073381918
- Norval RA, Andrew HR, Yunker CE. Pheromone-mediation of host-selection in bont ticks (Amblyomma hebraeum Koch). Science. 1989;243:364-365. doi:10.1126/science.2911745
- Parola P, Raoult D. Ticks and tickborne bacterial diseases in humans: an emerging infectious threat. Clin Infect Dis. 2001;32:897-928. doi:10.1086/319347
- Kelly PJ, Mason PR. Transmission of a spotted fever group rickettsia by Amblyomma hebraeum (Acari: Ixodidae). J Med Entomol. 1991;28:598-600. doi:10.1093/jmedent/28.5.598
- Maina AN, Jiang J, Omulo SA, et al. High prevalence of Rickettsia africae variants in Amblyomma variegatum ticks from domestic mammals in rural Western Kenya: implications for human health. Vector Borne Zoonotic Dis. 2014;14:693-702. doi:10.1089/vbz.2014.1578
- Jensenius M, Fournier P-E, Kelly P, et al. African tick bite fever. Lancet Infect Dis. 2003;3:557-564. doi:10.1016/s1473-3099(03)00739-4
- Roch N, Epaulard O, Pelloux I, et al. African tick bite fever in elderly patients: 8 cases in French tourists returning from South Africa. Clin Infect Dis. 2008;47:E28-E35. doi:10.1086/589868
- Palau L, Pankey GA. Mediterranean spotted fever in travelers from the United States. J Travel Med. 1997;4:179-182. doi:10.1111/j.1708-8305.1997.tb00816.x
- Fournier P-E, Jensenius M, Laferl H, et al. Kinetics of antibody responses in Rickettsia africae and Rickettsia conorii infections. Clin Diagn Lab Immunol. 2002;9:324-328. doi:10.1128/cdli.9.2.324-328.2002
- Brouqui P, Bacellar F, Baranton G, et al; ; . Guidelines for the diagnosis of tick-borne bacterial diseases in Europe. Clin Microbiol Infect. 2004;10:1108-1132. doi:10.1111/j.1469-0691.2004.01019.x
- Rolain JM, Jensenius M, Raoult D. Rickettsial infections—a threat to travelers? Curr Opin Infect Dis. 2004;17:433-437. doi:10.1097/00001432-200410000-00008
- Jensenius M, Pretorius AM, Clarke F, et al. Repellent efficacy of four commercial DEET lotions against Amblyomma hebraeum (Acari: Ixodidae), the principal vector of Rickettsia africae in southern Africa. Trans R Soc Trop Med Hyg. 2005;99:708-711. doi:10.1016/j.trstmh.2005.01.006
To the Editor:
One of the more common tick-borne infections seen in travelers returning from sub-Saharan Africa is caused by Rickettsia africae, which is the etiologic agent of African tick-bite fever (ATBF), the most common tick-borne bacterial zoonosis.1 There are 2 known tick vectors of disease: Amblyomma variegatum, found in sub-Saharan Africa and the West Indies, and Amblyomma hebraeum, found specifically in southern Africa.2,3
Unlike other disease-carrying ticks that passively wait on vegetation to be picked up by a host, A hebraeum uniquely attract other nearby ticks to the host. Studies have shown that male ticks feeding on a nonhuman host (usually cattle) can emit an aggression-attachment pheromone that attracts other ticks to the host. The presence of the pheromone allows unfed ticks to actively discriminate between hosts on which these parasites have fed successfully (ie, suitable hosts) and those on which they have not.4
The aggressive hunting nature of A hebraeum explains the clinical presentation of multiple eschars and why large groups of exposed travelers—such as soldiers, leisure safari tourists, game hunters, and foreign-aid workers—are affected.2
Another southern African spotted fever group, Rickettsia conorii is the causative agent of Mediterranean spotted fever (MSF). Ticks carrying R conorii exhibit a much less aggressive hunting stylethan A hebraeum; consequently, infected patients present with a single eschar site.5
Rickettsia africae is estimated to have very high prevalence (95.2%) in Amblyomma ticks and a fairly high prevalence (approximately 4.0% to 8.6%) in travelers coming from rural southern Africa,6,7 with an incubation period of 5 to 10 days after inoculation by an infected tick.8 Signs include fever, a generalized maculopapular or papulovesicular rash, and regional lymphadenopathy; symptoms include fatigue, headache, and myalgia.
The inoculation eschar—single or multiple—commonly presents on the legs and is accompanied by tender lymphadenopathy of draining nodes1,8 More severe findings, such as myocarditis and subacute neuropathy, have been reported in elderly patients.9
A 77-year-old man presented with a pruritic maculopapular and papulovesicular rash distributed over the upper and lower extremities of 3 weeks’ duration. The patient reported having been on a 12-day mission trip to Limpopo, South Africa, where he was constructing and installing safe toilets to replace dangerous toilet pits. He believed he had been bitten by 2 ticks, after which he noted a dark purple and black patch on the left lower leg by the third day of the trip. He developed a sudden persistent pruritic rash, first on the lower extremities and then spreading to the upper extremities. The patient was seen by an American physician in South Africa who gave him a 7-day course of oral doxycycline monohydrate 100 mg twice daily. He then returned to the United States.

Sixteen days after being bitten by the ticks, the patient was examined in our dermatology office. Physical examination revealed an erythematous plaque with a central eschar over the medial aspect of the left leg (Figure 1) and multiple 3- to 6-mm, erythematous, dome-shaped papules scattered over the dorsal aspects of the feet and ankles (Figure 2). The examination was otherwise normal. Blood was drawn the same day for laboratory analysis; no abnormalities of platelets, red blood cells, or white blood cells were found. Results of a chemistry panel and liver enzyme tests were within reference range.

Skin biopsies were taken to elucidate the underlying pathology. Although an arthropod assault was suspected, there also was concern for deep vessel vasculitis because of the presence of considerable petechiae and purpura (Figure 3). Histologically, leukocytoclasia was seen in deep dermal blood vessels. A mild eosinophilic spongiosis with a mixed dermal infiltrate was identified—strengthening our suspicion of an arthropod assault. Bacterial cultures for aerobes and anaerobes using material taken from the right shin showed no growth.

Ten days after the initial biopsies, serum specimens were drawn and swabs of eschar were collected and sent to the Centers for Disease Control and Prevention for further testing. Serum was tested by immunofluorescence assay (IFA) for spotted fever group IgG to detect Rocky Mountain spotted fever and ATBF antibodies; both tests were negative. Swab material from eschar was tested again by IFA for spotted fever group IgG (Rocky Mountain spotted fever) and antibodies to ATBF and with bacterial culture isolation and nucleic acid amplification; the culture and amplification came back positive for R africae.
Because the specialized tests confirmed infection with R africae, the patient was given triamcinolone cream 0.1% to apply twice daily to the pruritic lesions for as long as 4 weeks; an additional 14-day course of oral doxycycline monohydrate (100 mg twice daily) was given. At follow-up, the lesions had fully resolved without evident scarring.
Various diagnostic techniques can detect R africae. Bacterial culture and the polymerase chain reaction are specific and therefore diagnostic. In addition, the diagnosis of rickettsiosis can be made with serology testing, in which disease-specific antibodies are detected by indirect IFA using disease-specific antigens.
Antigens from R rickettsii (the agent of Rocky Mountain spotted fever), R conorii (Mediterranean spotted fever), and R africae (ATBF) are commercially available for making the diagnosis of rickettsiosis. However, antigens from R conorii exhibit cross-reactivity with R africae, which can confound the diagnosis.1,10 Serologic IFA tests have been shown to be less sensitive, especially when performed after antibacterial treatment has started.
In a study, 17 of 65 (26%) ATBF-confirmed patients were seronegative (acute and convalescent-phase sera) against R africae; 14 had received doxycycline during the first week of clinical signs. The current hypothesis is that R africae is highly sensitive to doxycycline and that early exposure to the drug prevented development of detectable titers of reactive antibodies, thus producing a negative serology test.11
Furthermore, it has been shown that seroconversion of IgG and IgM antibodies in R africae–infected sera is delayed compared to what is observed with R conorii–infected sera. Typically, seroconversion of R conorii–infected sera can be detected within 2 weeks; seroconversion in R africae–infected sera can take 4 weeks or longer.11
Our patient had a confirmed case of ATBF secondary to R africae infection, which was evident from tissue culture isolation and polymerase chain reaction analysis of swab material obtained from eschar sites, both of which yielded a positive result for R africae. The traveler’s negative serologic status might be due to his early exposure to doxycycline or to the 4-week delay in R africae seroconversion; his serum was collected only 3 weeks after the tick bites.
Clinical signs also aid in making the diagnosis of ATBF and distinguishing R conorii from R africae infection. Because of the aggressive hunting nature of the tick carrying R africae, they are associated with multiple eschars and tend to affect groups of multiple people, especially in rural areas.4,5 In contrast, ticks carrying R conorii yield a single eschar due to their passive style of infecting a host and because they favor a single host within urban areas.5 Both infections exhibit a maculopapular or papulovesicular rash and are accompanied by fatigue, headache, and myalgia, though ATBF tends to present with a milder rash than MSF.
Infection with either R conorii or R africae responds to tetracyclines, quinolones, and macrolides.10,12
African tick-bite fever is becoming more common, which should encourage clinicians to become familiar with the disease. Less than 2 decades ago, ATBF virtually was unknown outside of Zimbabwe, Botswana, Tanzania, Zambia, and Kenya, where it is endemic. However, after the abolition of apartheid in the 1990s, international tourism in southern Africa increased 6-fold.13 African tick-bite fever is now one of the most common rickettsial infections in Africa.7 In addition to diagnosing ATBF and managing infected patients, clinicians can help prevent ATBF in individuals who travel to endemic areas by recommending commercial topical insect repellents containing at least 19.5% N,N-diethyl-meta-toluamide (DEET).14
To the Editor:
One of the more common tick-borne infections seen in travelers returning from sub-Saharan Africa is caused by Rickettsia africae, which is the etiologic agent of African tick-bite fever (ATBF), the most common tick-borne bacterial zoonosis.1 There are 2 known tick vectors of disease: Amblyomma variegatum, found in sub-Saharan Africa and the West Indies, and Amblyomma hebraeum, found specifically in southern Africa.2,3
Unlike other disease-carrying ticks that passively wait on vegetation to be picked up by a host, A hebraeum uniquely attract other nearby ticks to the host. Studies have shown that male ticks feeding on a nonhuman host (usually cattle) can emit an aggression-attachment pheromone that attracts other ticks to the host. The presence of the pheromone allows unfed ticks to actively discriminate between hosts on which these parasites have fed successfully (ie, suitable hosts) and those on which they have not.4
The aggressive hunting nature of A hebraeum explains the clinical presentation of multiple eschars and why large groups of exposed travelers—such as soldiers, leisure safari tourists, game hunters, and foreign-aid workers—are affected.2
Another southern African spotted fever group, Rickettsia conorii is the causative agent of Mediterranean spotted fever (MSF). Ticks carrying R conorii exhibit a much less aggressive hunting stylethan A hebraeum; consequently, infected patients present with a single eschar site.5
Rickettsia africae is estimated to have very high prevalence (95.2%) in Amblyomma ticks and a fairly high prevalence (approximately 4.0% to 8.6%) in travelers coming from rural southern Africa,6,7 with an incubation period of 5 to 10 days after inoculation by an infected tick.8 Signs include fever, a generalized maculopapular or papulovesicular rash, and regional lymphadenopathy; symptoms include fatigue, headache, and myalgia.
The inoculation eschar—single or multiple—commonly presents on the legs and is accompanied by tender lymphadenopathy of draining nodes1,8 More severe findings, such as myocarditis and subacute neuropathy, have been reported in elderly patients.9
A 77-year-old man presented with a pruritic maculopapular and papulovesicular rash distributed over the upper and lower extremities of 3 weeks’ duration. The patient reported having been on a 12-day mission trip to Limpopo, South Africa, where he was constructing and installing safe toilets to replace dangerous toilet pits. He believed he had been bitten by 2 ticks, after which he noted a dark purple and black patch on the left lower leg by the third day of the trip. He developed a sudden persistent pruritic rash, first on the lower extremities and then spreading to the upper extremities. The patient was seen by an American physician in South Africa who gave him a 7-day course of oral doxycycline monohydrate 100 mg twice daily. He then returned to the United States.

Sixteen days after being bitten by the ticks, the patient was examined in our dermatology office. Physical examination revealed an erythematous plaque with a central eschar over the medial aspect of the left leg (Figure 1) and multiple 3- to 6-mm, erythematous, dome-shaped papules scattered over the dorsal aspects of the feet and ankles (Figure 2). The examination was otherwise normal. Blood was drawn the same day for laboratory analysis; no abnormalities of platelets, red blood cells, or white blood cells were found. Results of a chemistry panel and liver enzyme tests were within reference range.

Skin biopsies were taken to elucidate the underlying pathology. Although an arthropod assault was suspected, there also was concern for deep vessel vasculitis because of the presence of considerable petechiae and purpura (Figure 3). Histologically, leukocytoclasia was seen in deep dermal blood vessels. A mild eosinophilic spongiosis with a mixed dermal infiltrate was identified—strengthening our suspicion of an arthropod assault. Bacterial cultures for aerobes and anaerobes using material taken from the right shin showed no growth.

Ten days after the initial biopsies, serum specimens were drawn and swabs of eschar were collected and sent to the Centers for Disease Control and Prevention for further testing. Serum was tested by immunofluorescence assay (IFA) for spotted fever group IgG to detect Rocky Mountain spotted fever and ATBF antibodies; both tests were negative. Swab material from eschar was tested again by IFA for spotted fever group IgG (Rocky Mountain spotted fever) and antibodies to ATBF and with bacterial culture isolation and nucleic acid amplification; the culture and amplification came back positive for R africae.
Because the specialized tests confirmed infection with R africae, the patient was given triamcinolone cream 0.1% to apply twice daily to the pruritic lesions for as long as 4 weeks; an additional 14-day course of oral doxycycline monohydrate (100 mg twice daily) was given. At follow-up, the lesions had fully resolved without evident scarring.
Various diagnostic techniques can detect R africae. Bacterial culture and the polymerase chain reaction are specific and therefore diagnostic. In addition, the diagnosis of rickettsiosis can be made with serology testing, in which disease-specific antibodies are detected by indirect IFA using disease-specific antigens.
Antigens from R rickettsii (the agent of Rocky Mountain spotted fever), R conorii (Mediterranean spotted fever), and R africae (ATBF) are commercially available for making the diagnosis of rickettsiosis. However, antigens from R conorii exhibit cross-reactivity with R africae, which can confound the diagnosis.1,10 Serologic IFA tests have been shown to be less sensitive, especially when performed after antibacterial treatment has started.
In a study, 17 of 65 (26%) ATBF-confirmed patients were seronegative (acute and convalescent-phase sera) against R africae; 14 had received doxycycline during the first week of clinical signs. The current hypothesis is that R africae is highly sensitive to doxycycline and that early exposure to the drug prevented development of detectable titers of reactive antibodies, thus producing a negative serology test.11
Furthermore, it has been shown that seroconversion of IgG and IgM antibodies in R africae–infected sera is delayed compared to what is observed with R conorii–infected sera. Typically, seroconversion of R conorii–infected sera can be detected within 2 weeks; seroconversion in R africae–infected sera can take 4 weeks or longer.11
Our patient had a confirmed case of ATBF secondary to R africae infection, which was evident from tissue culture isolation and polymerase chain reaction analysis of swab material obtained from eschar sites, both of which yielded a positive result for R africae. The traveler’s negative serologic status might be due to his early exposure to doxycycline or to the 4-week delay in R africae seroconversion; his serum was collected only 3 weeks after the tick bites.
Clinical signs also aid in making the diagnosis of ATBF and distinguishing R conorii from R africae infection. Because of the aggressive hunting nature of the tick carrying R africae, they are associated with multiple eschars and tend to affect groups of multiple people, especially in rural areas.4,5 In contrast, ticks carrying R conorii yield a single eschar due to their passive style of infecting a host and because they favor a single host within urban areas.5 Both infections exhibit a maculopapular or papulovesicular rash and are accompanied by fatigue, headache, and myalgia, though ATBF tends to present with a milder rash than MSF.
Infection with either R conorii or R africae responds to tetracyclines, quinolones, and macrolides.10,12
African tick-bite fever is becoming more common, which should encourage clinicians to become familiar with the disease. Less than 2 decades ago, ATBF virtually was unknown outside of Zimbabwe, Botswana, Tanzania, Zambia, and Kenya, where it is endemic. However, after the abolition of apartheid in the 1990s, international tourism in southern Africa increased 6-fold.13 African tick-bite fever is now one of the most common rickettsial infections in Africa.7 In addition to diagnosing ATBF and managing infected patients, clinicians can help prevent ATBF in individuals who travel to endemic areas by recommending commercial topical insect repellents containing at least 19.5% N,N-diethyl-meta-toluamide (DEET).14
- Parola P, Paddock CD, Socolovschi C, et al. Update on tick-borne rickettsioses around the world: a geographic approach. Clin Microbiol Rev. 2013;26:657-702. doi:10.1128/CMR.00032-13
- Jensenius M, Fournier P-E, Vene S, et al; Norwegian African Tick Bite Fever Study Group. African tick bite fever in travelers to rural sub-equatorial Africa. Clin Infect Dis. 2003;36:1411-1417. doi:10.1086/375083
- Parola P, Jourdan J, Raoult D. Tick-borne infection caused by Rickettsia africae in the West Indies. N Engl J Med. 1998;338:1391-1392. doi:10.1056/NEJM199805073381918
- Norval RA, Andrew HR, Yunker CE. Pheromone-mediation of host-selection in bont ticks (Amblyomma hebraeum Koch). Science. 1989;243:364-365. doi:10.1126/science.2911745
- Parola P, Raoult D. Ticks and tickborne bacterial diseases in humans: an emerging infectious threat. Clin Infect Dis. 2001;32:897-928. doi:10.1086/319347
- Kelly PJ, Mason PR. Transmission of a spotted fever group rickettsia by Amblyomma hebraeum (Acari: Ixodidae). J Med Entomol. 1991;28:598-600. doi:10.1093/jmedent/28.5.598
- Maina AN, Jiang J, Omulo SA, et al. High prevalence of Rickettsia africae variants in Amblyomma variegatum ticks from domestic mammals in rural Western Kenya: implications for human health. Vector Borne Zoonotic Dis. 2014;14:693-702. doi:10.1089/vbz.2014.1578
- Jensenius M, Fournier P-E, Kelly P, et al. African tick bite fever. Lancet Infect Dis. 2003;3:557-564. doi:10.1016/s1473-3099(03)00739-4
- Roch N, Epaulard O, Pelloux I, et al. African tick bite fever in elderly patients: 8 cases in French tourists returning from South Africa. Clin Infect Dis. 2008;47:E28-E35. doi:10.1086/589868
- Palau L, Pankey GA. Mediterranean spotted fever in travelers from the United States. J Travel Med. 1997;4:179-182. doi:10.1111/j.1708-8305.1997.tb00816.x
- Fournier P-E, Jensenius M, Laferl H, et al. Kinetics of antibody responses in Rickettsia africae and Rickettsia conorii infections. Clin Diagn Lab Immunol. 2002;9:324-328. doi:10.1128/cdli.9.2.324-328.2002
- Brouqui P, Bacellar F, Baranton G, et al; ; . Guidelines for the diagnosis of tick-borne bacterial diseases in Europe. Clin Microbiol Infect. 2004;10:1108-1132. doi:10.1111/j.1469-0691.2004.01019.x
- Rolain JM, Jensenius M, Raoult D. Rickettsial infections—a threat to travelers? Curr Opin Infect Dis. 2004;17:433-437. doi:10.1097/00001432-200410000-00008
- Jensenius M, Pretorius AM, Clarke F, et al. Repellent efficacy of four commercial DEET lotions against Amblyomma hebraeum (Acari: Ixodidae), the principal vector of Rickettsia africae in southern Africa. Trans R Soc Trop Med Hyg. 2005;99:708-711. doi:10.1016/j.trstmh.2005.01.006
- Parola P, Paddock CD, Socolovschi C, et al. Update on tick-borne rickettsioses around the world: a geographic approach. Clin Microbiol Rev. 2013;26:657-702. doi:10.1128/CMR.00032-13
- Jensenius M, Fournier P-E, Vene S, et al; Norwegian African Tick Bite Fever Study Group. African tick bite fever in travelers to rural sub-equatorial Africa. Clin Infect Dis. 2003;36:1411-1417. doi:10.1086/375083
- Parola P, Jourdan J, Raoult D. Tick-borne infection caused by Rickettsia africae in the West Indies. N Engl J Med. 1998;338:1391-1392. doi:10.1056/NEJM199805073381918
- Norval RA, Andrew HR, Yunker CE. Pheromone-mediation of host-selection in bont ticks (Amblyomma hebraeum Koch). Science. 1989;243:364-365. doi:10.1126/science.2911745
- Parola P, Raoult D. Ticks and tickborne bacterial diseases in humans: an emerging infectious threat. Clin Infect Dis. 2001;32:897-928. doi:10.1086/319347
- Kelly PJ, Mason PR. Transmission of a spotted fever group rickettsia by Amblyomma hebraeum (Acari: Ixodidae). J Med Entomol. 1991;28:598-600. doi:10.1093/jmedent/28.5.598
- Maina AN, Jiang J, Omulo SA, et al. High prevalence of Rickettsia africae variants in Amblyomma variegatum ticks from domestic mammals in rural Western Kenya: implications for human health. Vector Borne Zoonotic Dis. 2014;14:693-702. doi:10.1089/vbz.2014.1578
- Jensenius M, Fournier P-E, Kelly P, et al. African tick bite fever. Lancet Infect Dis. 2003;3:557-564. doi:10.1016/s1473-3099(03)00739-4
- Roch N, Epaulard O, Pelloux I, et al. African tick bite fever in elderly patients: 8 cases in French tourists returning from South Africa. Clin Infect Dis. 2008;47:E28-E35. doi:10.1086/589868
- Palau L, Pankey GA. Mediterranean spotted fever in travelers from the United States. J Travel Med. 1997;4:179-182. doi:10.1111/j.1708-8305.1997.tb00816.x
- Fournier P-E, Jensenius M, Laferl H, et al. Kinetics of antibody responses in Rickettsia africae and Rickettsia conorii infections. Clin Diagn Lab Immunol. 2002;9:324-328. doi:10.1128/cdli.9.2.324-328.2002
- Brouqui P, Bacellar F, Baranton G, et al; ; . Guidelines for the diagnosis of tick-borne bacterial diseases in Europe. Clin Microbiol Infect. 2004;10:1108-1132. doi:10.1111/j.1469-0691.2004.01019.x
- Rolain JM, Jensenius M, Raoult D. Rickettsial infections—a threat to travelers? Curr Opin Infect Dis. 2004;17:433-437. doi:10.1097/00001432-200410000-00008
- Jensenius M, Pretorius AM, Clarke F, et al. Repellent efficacy of four commercial DEET lotions against Amblyomma hebraeum (Acari: Ixodidae), the principal vector of Rickettsia africae in southern Africa. Trans R Soc Trop Med Hyg. 2005;99:708-711. doi:10.1016/j.trstmh.2005.01.006
PRACTICE POINTS
- African tick-bite fever (ATBF) is one of the more common tick-borne bacterial zoonoses and should be considered in patients presenting with multiple eschar sites who have had exposure to rural areas of southern Africa in the preceding 2 weeks.
- Ticks carrying Rickettsia africae are unique, given their ability to actively be attracted to and hunt their nonhuman hosts via an aggression-attachment pheromone.
- Laboratory diagnosis of ATBF can be challenging due to the high cross-reactivity of Helvetica Neue LT StdR africae with Helvetica Neue LT StdRickettsia conorii in serologic testing and the delay in seroconversion in Helvetica Neue LT StdR africae infection.
Chronic Erythematous Plaques Around the Ears
The Diagnosis: Discoid Lupus Erythematosus
The biopsies demonstrated vacuolar interface changes with superficial and deep perivascular and periadnexal inflammation as well as increased background mucin deposition. The clinical morphology and distributions of the plaques limited to the photoexposed areas of the head suggested a diagnosis of discoid lupus erythematosus (DLE). The interface changes on histopathology supported this clinical impression. Our patient was treated with limited application of triamcinolone ointment 0.1% twice daily around the ears and neck, tacrolimus ointment 0.1% twice daily on the face, and hydroxychloroquine, as well as sun protection instructions. Smoking cessation was strongly advised.
Discoid lupus erythematosus is a disorder with chronic, erythematous, scaly, coin-shaped (discoid) plaques and is the most common form of chronic cutaneous lupus erythematosus.1 Lesions usually present on sun-exposed areas of the face, scalp, neck, ears, lips, or upper torso. They expand slowly with an active peripheral margin and a central scar that can result in induration, pigmentation changes, telangiectases, pruritus, or tenderness. Hair-bearing areas may be involved, causing hair loss due to follicular plugging; irreversible scarring alopecia can result. Facial DLE often spares the nasolabial folds. Ear involvement characteristically includes the conchal bowl and the outer external auditory canal. Discoid lupus erythematosus is considered localized if most of the head and neck region is involved or generalized if lesions also are present below the neck. Risk factors for DLE include genetic and environmental factors such as UV exposure, hormones, or exposure to toxins such as cigarette smoke.1 The disorder most commonly affects females and has a higher prevalence in patients of African descent than in Asian and White patients. Disease can occur at any age but usually occurs between 20 and 40 years of age.2 Discoid lupus erythematosus and other forms of chronic cutaneous lupus can occur independently or in conjunction with systemic lupus erythematosus (SLE), and approximately 15% to 30% of SLE patients develop DLE.1
Discoid lupus erythematosus is clinically diagnosed by the presentation of plaques in the characteristic distribution with confirmation via skin biopsy.1 Elman et al3 created a system for DLE classification that was only clinical and did not involve histopathology. Histologically, DLE often includes basement membrane thickening, follicular keratin plugs, mucin deposition, and vacuolar change with an interface, and a perivascular and periadnexal lymphocytic infiltrate.3,4 Antibodies such as antinuclear antibodies, rheumatoid factor, anti–double-stranded DNA, anti-Smith, and Sjögren syndrome A and B antibodies may be present (albeit with low positive frequency) in cutaneous lupus erythematosus.4 Characteristics of SLE also may be present, helping to confirm the diagnosis. Because there is an association of DLE with SLE, various laboratory tests should be ordered, including complete blood cell count, renal function panel, inflammatory markers, antibodies, and urinalysis for proteinuria.2,4
Treatment of DLE consists of preventative measures, such as sun protection with vitamin D supplementation, avoidance of drug triggers, and smoking cessation, as well as pharmacotherapy. The importance of wearing sun protective hats and garments with sunscreen use cannot be understated.1 Smoking cessation should be advised because smoking reduces the efficacy of antimalarial treatment and potentially increases the likelihood of patients requiring a second antimalarial drug. Quinacrine often is noted in both the dermatology and rheumatology literature to be used for escalating cutaneous lupus erythematosus care when hydroxychloroquine is ineffective or not tolerated, but no US manufacturer produces this medication; thus, compounding is required, which may be financially prohibitive, making this recommendation difficult to translate into clinical practice.5 Firstline therapy for acute flares is high-potency topical corticosteroids. If lesions are primarily on areas other than the face, a medium-potency topical steroid may be used. Topical calcineurin inhibitors or intralesional corticosteroids may be used if minimal improvement is seen after initial topical corticosteroid therapy. Treatment for widespread disease or disease that is resistant to local treatment is systemic therapy with antimalarial agents, followed by antimetabolites, systemic retinoids, thalidomide, or dapsone.1,2 The Cutaneous Lupus Erythematosus Disease Area and Severity Index is a valid tool to gauge the degree of disease and to help with disease progression and treatment response by noting the features of the plaques.1
Patients also should be educated that this disease can last for years, and long-standing DLE plaques infrequently can give rise to squamous cell carcinoma. In addition, isolated DLE can progress to SLE in 5% to 28% of patients.2
The differential diagnosis in our patient included other diseases with violaceous annular lesions and central clearing. Majocchi granuloma usually presents in areas of prior trauma, possibly due to shaving the face in our patient, or in the setting of topical corticosteroid use or immunosuppression. Scaling often is present within lesions, and histology shows fungal elements.6 Cutaneous sarcoidosis usually presents on the face, with scarring alopecia when appearing on the scalp; histology shows noncaseating granulomas, and 70% of patients with cutaneous symptoms will have systemic sarcoidosis.7 Granuloma annulare most commonly presents on the extremities, and histology shows lymphohistiocytic granulomas in a palisaded or interstitial pattern with connective-tissue degeneration and mucinous deposits.8 Annular psoriasis often is scaly and symmetric with parakeratosis, epidermal hyperplasia, dilated dermal capillaries, loss of granular layer, perivascular mononuclear cell infiltrate, and elongation of rete ridges on histology.9 Drug-induced lupus erythematosus always should be considered in patients taking triggering drugs such as antihypertensives, lipid-lowering drugs, antifungals, anti–tumor necrosis factor drugs, and proton pump inhibitors—the latter being a drug our patient was taking.10
- Sontheimer CJ, Costner MI, Sontheimer RD. Lupus erythematosus. In: Kang S, Amagai M, Bruckner A, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019:1037-1060.
- Lee KC. Discoid lupus. In: Ferri FF, ed. Ferri’s Clinical Advisor 2021. Elsevier; 2021:477.e15-477.e18.
- Elman SA, Joyce C, Braudis K, et al. Creation and validation of classification criteria for discoid lupus erythematosus. JAMA Dermatol. 2020;156:901-906. doi:10.1001/jamadermatol.2020.1698
- Patel P, Werth V. Cutaneous lupus erythematosus: a review. Dermatol Clin. 2002;20:373-385, v. doi:10.1016/s0733-8635(02)00016-5
- Mittal L, Werth VP. The quinacrine experience in a population of patients with cutaneous lupus erythematosus and dermatomyositis. J Am Acad Dermatol. 2017;77:374-377. doi:10.1016/j .jaad.2017.03.027
- Craddock LN, Schieke SM. Superficial fungal infection. In: Kang S, Amagai M, Bruckner A, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019:2925-2951.
- Tan J, Vleugels R. Dermatologic findings in systemic disease. In: McKean S, Dressler D, Ross J, et al, eds. Principles and Practice of Hospital Medicine. 2nd ed. McGraw Hill; 2017:1145-1170.
- Prendiville JS. Granuloma annulare. In: Kang S, Amagai M, Bruckner A, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019:564-571.
- Gudjonsson JE, Elder JT. Psoriasis. In: Kang S, Amagai M, Bruckner A, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019:457-497.
- He Y, Sawalha AH. Drug-induced lupus erythematosus: an update on drugs and mechanisms. Curr Opin Rheumatol. 2018;30:490-497. doi:10.1097/BOR.0000000000000522
The Diagnosis: Discoid Lupus Erythematosus
The biopsies demonstrated vacuolar interface changes with superficial and deep perivascular and periadnexal inflammation as well as increased background mucin deposition. The clinical morphology and distributions of the plaques limited to the photoexposed areas of the head suggested a diagnosis of discoid lupus erythematosus (DLE). The interface changes on histopathology supported this clinical impression. Our patient was treated with limited application of triamcinolone ointment 0.1% twice daily around the ears and neck, tacrolimus ointment 0.1% twice daily on the face, and hydroxychloroquine, as well as sun protection instructions. Smoking cessation was strongly advised.
Discoid lupus erythematosus is a disorder with chronic, erythematous, scaly, coin-shaped (discoid) plaques and is the most common form of chronic cutaneous lupus erythematosus.1 Lesions usually present on sun-exposed areas of the face, scalp, neck, ears, lips, or upper torso. They expand slowly with an active peripheral margin and a central scar that can result in induration, pigmentation changes, telangiectases, pruritus, or tenderness. Hair-bearing areas may be involved, causing hair loss due to follicular plugging; irreversible scarring alopecia can result. Facial DLE often spares the nasolabial folds. Ear involvement characteristically includes the conchal bowl and the outer external auditory canal. Discoid lupus erythematosus is considered localized if most of the head and neck region is involved or generalized if lesions also are present below the neck. Risk factors for DLE include genetic and environmental factors such as UV exposure, hormones, or exposure to toxins such as cigarette smoke.1 The disorder most commonly affects females and has a higher prevalence in patients of African descent than in Asian and White patients. Disease can occur at any age but usually occurs between 20 and 40 years of age.2 Discoid lupus erythematosus and other forms of chronic cutaneous lupus can occur independently or in conjunction with systemic lupus erythematosus (SLE), and approximately 15% to 30% of SLE patients develop DLE.1
Discoid lupus erythematosus is clinically diagnosed by the presentation of plaques in the characteristic distribution with confirmation via skin biopsy.1 Elman et al3 created a system for DLE classification that was only clinical and did not involve histopathology. Histologically, DLE often includes basement membrane thickening, follicular keratin plugs, mucin deposition, and vacuolar change with an interface, and a perivascular and periadnexal lymphocytic infiltrate.3,4 Antibodies such as antinuclear antibodies, rheumatoid factor, anti–double-stranded DNA, anti-Smith, and Sjögren syndrome A and B antibodies may be present (albeit with low positive frequency) in cutaneous lupus erythematosus.4 Characteristics of SLE also may be present, helping to confirm the diagnosis. Because there is an association of DLE with SLE, various laboratory tests should be ordered, including complete blood cell count, renal function panel, inflammatory markers, antibodies, and urinalysis for proteinuria.2,4
Treatment of DLE consists of preventative measures, such as sun protection with vitamin D supplementation, avoidance of drug triggers, and smoking cessation, as well as pharmacotherapy. The importance of wearing sun protective hats and garments with sunscreen use cannot be understated.1 Smoking cessation should be advised because smoking reduces the efficacy of antimalarial treatment and potentially increases the likelihood of patients requiring a second antimalarial drug. Quinacrine often is noted in both the dermatology and rheumatology literature to be used for escalating cutaneous lupus erythematosus care when hydroxychloroquine is ineffective or not tolerated, but no US manufacturer produces this medication; thus, compounding is required, which may be financially prohibitive, making this recommendation difficult to translate into clinical practice.5 Firstline therapy for acute flares is high-potency topical corticosteroids. If lesions are primarily on areas other than the face, a medium-potency topical steroid may be used. Topical calcineurin inhibitors or intralesional corticosteroids may be used if minimal improvement is seen after initial topical corticosteroid therapy. Treatment for widespread disease or disease that is resistant to local treatment is systemic therapy with antimalarial agents, followed by antimetabolites, systemic retinoids, thalidomide, or dapsone.1,2 The Cutaneous Lupus Erythematosus Disease Area and Severity Index is a valid tool to gauge the degree of disease and to help with disease progression and treatment response by noting the features of the plaques.1
Patients also should be educated that this disease can last for years, and long-standing DLE plaques infrequently can give rise to squamous cell carcinoma. In addition, isolated DLE can progress to SLE in 5% to 28% of patients.2
The differential diagnosis in our patient included other diseases with violaceous annular lesions and central clearing. Majocchi granuloma usually presents in areas of prior trauma, possibly due to shaving the face in our patient, or in the setting of topical corticosteroid use or immunosuppression. Scaling often is present within lesions, and histology shows fungal elements.6 Cutaneous sarcoidosis usually presents on the face, with scarring alopecia when appearing on the scalp; histology shows noncaseating granulomas, and 70% of patients with cutaneous symptoms will have systemic sarcoidosis.7 Granuloma annulare most commonly presents on the extremities, and histology shows lymphohistiocytic granulomas in a palisaded or interstitial pattern with connective-tissue degeneration and mucinous deposits.8 Annular psoriasis often is scaly and symmetric with parakeratosis, epidermal hyperplasia, dilated dermal capillaries, loss of granular layer, perivascular mononuclear cell infiltrate, and elongation of rete ridges on histology.9 Drug-induced lupus erythematosus always should be considered in patients taking triggering drugs such as antihypertensives, lipid-lowering drugs, antifungals, anti–tumor necrosis factor drugs, and proton pump inhibitors—the latter being a drug our patient was taking.10
The Diagnosis: Discoid Lupus Erythematosus
The biopsies demonstrated vacuolar interface changes with superficial and deep perivascular and periadnexal inflammation as well as increased background mucin deposition. The clinical morphology and distributions of the plaques limited to the photoexposed areas of the head suggested a diagnosis of discoid lupus erythematosus (DLE). The interface changes on histopathology supported this clinical impression. Our patient was treated with limited application of triamcinolone ointment 0.1% twice daily around the ears and neck, tacrolimus ointment 0.1% twice daily on the face, and hydroxychloroquine, as well as sun protection instructions. Smoking cessation was strongly advised.
Discoid lupus erythematosus is a disorder with chronic, erythematous, scaly, coin-shaped (discoid) plaques and is the most common form of chronic cutaneous lupus erythematosus.1 Lesions usually present on sun-exposed areas of the face, scalp, neck, ears, lips, or upper torso. They expand slowly with an active peripheral margin and a central scar that can result in induration, pigmentation changes, telangiectases, pruritus, or tenderness. Hair-bearing areas may be involved, causing hair loss due to follicular plugging; irreversible scarring alopecia can result. Facial DLE often spares the nasolabial folds. Ear involvement characteristically includes the conchal bowl and the outer external auditory canal. Discoid lupus erythematosus is considered localized if most of the head and neck region is involved or generalized if lesions also are present below the neck. Risk factors for DLE include genetic and environmental factors such as UV exposure, hormones, or exposure to toxins such as cigarette smoke.1 The disorder most commonly affects females and has a higher prevalence in patients of African descent than in Asian and White patients. Disease can occur at any age but usually occurs between 20 and 40 years of age.2 Discoid lupus erythematosus and other forms of chronic cutaneous lupus can occur independently or in conjunction with systemic lupus erythematosus (SLE), and approximately 15% to 30% of SLE patients develop DLE.1
Discoid lupus erythematosus is clinically diagnosed by the presentation of plaques in the characteristic distribution with confirmation via skin biopsy.1 Elman et al3 created a system for DLE classification that was only clinical and did not involve histopathology. Histologically, DLE often includes basement membrane thickening, follicular keratin plugs, mucin deposition, and vacuolar change with an interface, and a perivascular and periadnexal lymphocytic infiltrate.3,4 Antibodies such as antinuclear antibodies, rheumatoid factor, anti–double-stranded DNA, anti-Smith, and Sjögren syndrome A and B antibodies may be present (albeit with low positive frequency) in cutaneous lupus erythematosus.4 Characteristics of SLE also may be present, helping to confirm the diagnosis. Because there is an association of DLE with SLE, various laboratory tests should be ordered, including complete blood cell count, renal function panel, inflammatory markers, antibodies, and urinalysis for proteinuria.2,4
Treatment of DLE consists of preventative measures, such as sun protection with vitamin D supplementation, avoidance of drug triggers, and smoking cessation, as well as pharmacotherapy. The importance of wearing sun protective hats and garments with sunscreen use cannot be understated.1 Smoking cessation should be advised because smoking reduces the efficacy of antimalarial treatment and potentially increases the likelihood of patients requiring a second antimalarial drug. Quinacrine often is noted in both the dermatology and rheumatology literature to be used for escalating cutaneous lupus erythematosus care when hydroxychloroquine is ineffective or not tolerated, but no US manufacturer produces this medication; thus, compounding is required, which may be financially prohibitive, making this recommendation difficult to translate into clinical practice.5 Firstline therapy for acute flares is high-potency topical corticosteroids. If lesions are primarily on areas other than the face, a medium-potency topical steroid may be used. Topical calcineurin inhibitors or intralesional corticosteroids may be used if minimal improvement is seen after initial topical corticosteroid therapy. Treatment for widespread disease or disease that is resistant to local treatment is systemic therapy with antimalarial agents, followed by antimetabolites, systemic retinoids, thalidomide, or dapsone.1,2 The Cutaneous Lupus Erythematosus Disease Area and Severity Index is a valid tool to gauge the degree of disease and to help with disease progression and treatment response by noting the features of the plaques.1
Patients also should be educated that this disease can last for years, and long-standing DLE plaques infrequently can give rise to squamous cell carcinoma. In addition, isolated DLE can progress to SLE in 5% to 28% of patients.2
The differential diagnosis in our patient included other diseases with violaceous annular lesions and central clearing. Majocchi granuloma usually presents in areas of prior trauma, possibly due to shaving the face in our patient, or in the setting of topical corticosteroid use or immunosuppression. Scaling often is present within lesions, and histology shows fungal elements.6 Cutaneous sarcoidosis usually presents on the face, with scarring alopecia when appearing on the scalp; histology shows noncaseating granulomas, and 70% of patients with cutaneous symptoms will have systemic sarcoidosis.7 Granuloma annulare most commonly presents on the extremities, and histology shows lymphohistiocytic granulomas in a palisaded or interstitial pattern with connective-tissue degeneration and mucinous deposits.8 Annular psoriasis often is scaly and symmetric with parakeratosis, epidermal hyperplasia, dilated dermal capillaries, loss of granular layer, perivascular mononuclear cell infiltrate, and elongation of rete ridges on histology.9 Drug-induced lupus erythematosus always should be considered in patients taking triggering drugs such as antihypertensives, lipid-lowering drugs, antifungals, anti–tumor necrosis factor drugs, and proton pump inhibitors—the latter being a drug our patient was taking.10
- Sontheimer CJ, Costner MI, Sontheimer RD. Lupus erythematosus. In: Kang S, Amagai M, Bruckner A, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019:1037-1060.
- Lee KC. Discoid lupus. In: Ferri FF, ed. Ferri’s Clinical Advisor 2021. Elsevier; 2021:477.e15-477.e18.
- Elman SA, Joyce C, Braudis K, et al. Creation and validation of classification criteria for discoid lupus erythematosus. JAMA Dermatol. 2020;156:901-906. doi:10.1001/jamadermatol.2020.1698
- Patel P, Werth V. Cutaneous lupus erythematosus: a review. Dermatol Clin. 2002;20:373-385, v. doi:10.1016/s0733-8635(02)00016-5
- Mittal L, Werth VP. The quinacrine experience in a population of patients with cutaneous lupus erythematosus and dermatomyositis. J Am Acad Dermatol. 2017;77:374-377. doi:10.1016/j .jaad.2017.03.027
- Craddock LN, Schieke SM. Superficial fungal infection. In: Kang S, Amagai M, Bruckner A, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019:2925-2951.
- Tan J, Vleugels R. Dermatologic findings in systemic disease. In: McKean S, Dressler D, Ross J, et al, eds. Principles and Practice of Hospital Medicine. 2nd ed. McGraw Hill; 2017:1145-1170.
- Prendiville JS. Granuloma annulare. In: Kang S, Amagai M, Bruckner A, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019:564-571.
- Gudjonsson JE, Elder JT. Psoriasis. In: Kang S, Amagai M, Bruckner A, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019:457-497.
- He Y, Sawalha AH. Drug-induced lupus erythematosus: an update on drugs and mechanisms. Curr Opin Rheumatol. 2018;30:490-497. doi:10.1097/BOR.0000000000000522
- Sontheimer CJ, Costner MI, Sontheimer RD. Lupus erythematosus. In: Kang S, Amagai M, Bruckner A, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019:1037-1060.
- Lee KC. Discoid lupus. In: Ferri FF, ed. Ferri’s Clinical Advisor 2021. Elsevier; 2021:477.e15-477.e18.
- Elman SA, Joyce C, Braudis K, et al. Creation and validation of classification criteria for discoid lupus erythematosus. JAMA Dermatol. 2020;156:901-906. doi:10.1001/jamadermatol.2020.1698
- Patel P, Werth V. Cutaneous lupus erythematosus: a review. Dermatol Clin. 2002;20:373-385, v. doi:10.1016/s0733-8635(02)00016-5
- Mittal L, Werth VP. The quinacrine experience in a population of patients with cutaneous lupus erythematosus and dermatomyositis. J Am Acad Dermatol. 2017;77:374-377. doi:10.1016/j .jaad.2017.03.027
- Craddock LN, Schieke SM. Superficial fungal infection. In: Kang S, Amagai M, Bruckner A, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019:2925-2951.
- Tan J, Vleugels R. Dermatologic findings in systemic disease. In: McKean S, Dressler D, Ross J, et al, eds. Principles and Practice of Hospital Medicine. 2nd ed. McGraw Hill; 2017:1145-1170.
- Prendiville JS. Granuloma annulare. In: Kang S, Amagai M, Bruckner A, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019:564-571.
- Gudjonsson JE, Elder JT. Psoriasis. In: Kang S, Amagai M, Bruckner A, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019:457-497.
- He Y, Sawalha AH. Drug-induced lupus erythematosus: an update on drugs and mechanisms. Curr Opin Rheumatol. 2018;30:490-497. doi:10.1097/BOR.0000000000000522
A 41-year-old man presented to the dermatology clinic with erythematous, pruritic, and painful plaques around the ears of 6 years’ duration. He reported that application of topical steroids, antifungals, and most recently a topical calcineurin inhibitor did not change the appearance or symptoms. His medical history was notable for tobacco smoking and gastroesophageal reflux disease, for which he was taking omeprazole for the last 3 years. He was unsure if the lesions changed with UV exposure. He was an active-duty US military service member, and his job required frequently working outdoors. A review of systems was otherwise unremarkable. Physical examination revealed annular, erythematous, indurated plaques on both the preauricular and postauricular skin on the left ear with associated central atrophy and hypopigmentation. No alopecia was appreciated. The remainder of the skin examination was unremarkable. Ancillary laboratory test results were notable for a negative antinuclear antibody screen but positive (low titer) for Sjögren syndrome A and B antibodies. Two punch biopsies were performed.
Periorbital Orange Spots
The Diagnosis: Orange Palpebral Spots
The clinical presentation of our patient was consistent with a diagnosis of orange palpebral spots (OPSs), an uncommon discoloration that most often appears in White patients in the fifth or sixth decades of life. Orange palpebral spots were first described in 2008 by Assouly et al1 in 27 patients (23 females and 4 males). In 2015, Belliveau et al2 expanded the designation to yellow-orange palpebral spots because they felt the term more fully expressed the color variations depicted in their patients; however, this term more frequently is used in ophthalmology.
Orange palpebral spots commonly appear as asymptomatic, yellow-orange, symmetric lesions with a predilection for the recessed areas of the superior eyelids but also can present on the canthi and inferior eyelids. The discolorations are more easily visible on fair skin and have been reported to measure from 10 to 15 mm in the long axis.3 Assouly et al1 described the orange spots as having indistinct margins, with borders similar to “sand on a sea shore.” Orange palpebral spots can be a persistent discoloration, and there are no reports of spontaneous regression. No known association with malignancy or systemic illness has been reported.
Case reports of OPSs describe histologic similarities between specimens, including increased adipose tissue and pigment-laden macrophages in the superficial dermis.2 The pigmented deposits sometimes may be found in the basal keratinocytes of the epidermis and turn black with Fontana-Masson stain.1 No inflammatory infiltrates, necrosis, or xanthomization are characteristically found. Stains for iron, mucin, and amyloid also have been negative.2
The cause of pigmentation in OPSs is unknown; however, lipofuscin deposits and high-situated adipocytes in the reticular dermis colored by carotenoids have been proposed as possible mechanisms.1 No unifying cause for pigmentation in the serum (eg, cholesterol, triglycerides, thyroid-stimulating hormone, free retinol, vitamin E, carotenoids) was found in 11 of 27 patients with OPSs assessed by Assouly et al.1 In one case, lipofuscin, a degradation product of lysosomes, was detected by microscopic autofluorescence in the superficial dermis. However, lipofuscin typically is a breakdown product associated with aging, and OPSs have been present in patients as young as 28 years.1 Local trauma related to eye rubbing is another theory that has been proposed due to the finding of melanin in the superficial dermis. However, the absence of hemosiderin deposits as well as the extensive duration of the discolorations makes local trauma a less likely explanation for the etiology of OPSs.2
The clinical differential diagnosis for OPSs includes xanthelasma, jaundice, and carotenoderma. Xanthelasma presents as elevated yellow plaques usually found over the medial aspect of the eyes. In contrast, OPSs are nonelevated with both orange and yellow hues typically present. Histologic samples of xanthelasma are characterized by lipid-laden macrophages (foam cells) in the dermis in contrast to the adipose tissue seen in OPSs that has not been phagocytized.1,2 The lack of scleral icterus made jaundice an unlikely diagnosis in our patient. Bilirubin elevations substantial enough to cause skin discoloration also would be expected to discolor the conjunctiva. In carotenoderma, carotenoids are deposited in the sweat and sebum of the stratum corneum with the orange pigmentation most prominent in regions of increased sweating such as the palms, soles, and nasolabial folds.4 Our patient’s lack of discoloration in places other than the periorbital region made carotenoderma less likely.
In the study by Assouly et al,1 10 of 11 patients who underwent laboratory analysis self-reported eating a diet rich in fruit and vegetables, though no standardized questionnaire was given. One patient was found to have an elevated vitamin E level, and in 5 cases there was an elevated level of β-cryptoxanthin. The significance of these elevations in such a small minority is unknown, and increased β-cryptoxanthin has been attributed to increased consumption of citrus fruits during the winter season. Our patient reported ingesting a daily oral supplement rich in carotenoids that constituted 60% of the daily value of vitamin E including mixed tocopherols as well as 90% of the daily value of vitamin A with many sources of carotenoids including beta-carotenes, lutein/zeaxanthin, lycopene, and astaxanthin. An invasive biopsy was not taken in this case, as OPSs largely are diagnosed clinically. Greater awareness and recognition of OPSs may help to identify common underlying causes for this unique diagnosis.
- Assouly P, Cavelier-Balloy B, Dupré T. Orange palpebral spots. Dermatology. 2008;216:166-170.
- Belliveau MJ, Odashiro AN, Harvey JT. Yellow-orange palpebral spots. Ophthalmology. 2015;122:2139-2140.
- Kluger N, Guillot B. Bilateral orange discoloration of the upper eyelids: a quiz. Acta Derm Venereol. 2011;91:211-212.
- Maharshak N, Shapiro J, Trau H. Carotenoderma—a review of the current literature. Int J Dermatol. 2003;42:178-181.
The Diagnosis: Orange Palpebral Spots
The clinical presentation of our patient was consistent with a diagnosis of orange palpebral spots (OPSs), an uncommon discoloration that most often appears in White patients in the fifth or sixth decades of life. Orange palpebral spots were first described in 2008 by Assouly et al1 in 27 patients (23 females and 4 males). In 2015, Belliveau et al2 expanded the designation to yellow-orange palpebral spots because they felt the term more fully expressed the color variations depicted in their patients; however, this term more frequently is used in ophthalmology.
Orange palpebral spots commonly appear as asymptomatic, yellow-orange, symmetric lesions with a predilection for the recessed areas of the superior eyelids but also can present on the canthi and inferior eyelids. The discolorations are more easily visible on fair skin and have been reported to measure from 10 to 15 mm in the long axis.3 Assouly et al1 described the orange spots as having indistinct margins, with borders similar to “sand on a sea shore.” Orange palpebral spots can be a persistent discoloration, and there are no reports of spontaneous regression. No known association with malignancy or systemic illness has been reported.
Case reports of OPSs describe histologic similarities between specimens, including increased adipose tissue and pigment-laden macrophages in the superficial dermis.2 The pigmented deposits sometimes may be found in the basal keratinocytes of the epidermis and turn black with Fontana-Masson stain.1 No inflammatory infiltrates, necrosis, or xanthomization are characteristically found. Stains for iron, mucin, and amyloid also have been negative.2
The cause of pigmentation in OPSs is unknown; however, lipofuscin deposits and high-situated adipocytes in the reticular dermis colored by carotenoids have been proposed as possible mechanisms.1 No unifying cause for pigmentation in the serum (eg, cholesterol, triglycerides, thyroid-stimulating hormone, free retinol, vitamin E, carotenoids) was found in 11 of 27 patients with OPSs assessed by Assouly et al.1 In one case, lipofuscin, a degradation product of lysosomes, was detected by microscopic autofluorescence in the superficial dermis. However, lipofuscin typically is a breakdown product associated with aging, and OPSs have been present in patients as young as 28 years.1 Local trauma related to eye rubbing is another theory that has been proposed due to the finding of melanin in the superficial dermis. However, the absence of hemosiderin deposits as well as the extensive duration of the discolorations makes local trauma a less likely explanation for the etiology of OPSs.2
The clinical differential diagnosis for OPSs includes xanthelasma, jaundice, and carotenoderma. Xanthelasma presents as elevated yellow plaques usually found over the medial aspect of the eyes. In contrast, OPSs are nonelevated with both orange and yellow hues typically present. Histologic samples of xanthelasma are characterized by lipid-laden macrophages (foam cells) in the dermis in contrast to the adipose tissue seen in OPSs that has not been phagocytized.1,2 The lack of scleral icterus made jaundice an unlikely diagnosis in our patient. Bilirubin elevations substantial enough to cause skin discoloration also would be expected to discolor the conjunctiva. In carotenoderma, carotenoids are deposited in the sweat and sebum of the stratum corneum with the orange pigmentation most prominent in regions of increased sweating such as the palms, soles, and nasolabial folds.4 Our patient’s lack of discoloration in places other than the periorbital region made carotenoderma less likely.
In the study by Assouly et al,1 10 of 11 patients who underwent laboratory analysis self-reported eating a diet rich in fruit and vegetables, though no standardized questionnaire was given. One patient was found to have an elevated vitamin E level, and in 5 cases there was an elevated level of β-cryptoxanthin. The significance of these elevations in such a small minority is unknown, and increased β-cryptoxanthin has been attributed to increased consumption of citrus fruits during the winter season. Our patient reported ingesting a daily oral supplement rich in carotenoids that constituted 60% of the daily value of vitamin E including mixed tocopherols as well as 90% of the daily value of vitamin A with many sources of carotenoids including beta-carotenes, lutein/zeaxanthin, lycopene, and astaxanthin. An invasive biopsy was not taken in this case, as OPSs largely are diagnosed clinically. Greater awareness and recognition of OPSs may help to identify common underlying causes for this unique diagnosis.
The Diagnosis: Orange Palpebral Spots
The clinical presentation of our patient was consistent with a diagnosis of orange palpebral spots (OPSs), an uncommon discoloration that most often appears in White patients in the fifth or sixth decades of life. Orange palpebral spots were first described in 2008 by Assouly et al1 in 27 patients (23 females and 4 males). In 2015, Belliveau et al2 expanded the designation to yellow-orange palpebral spots because they felt the term more fully expressed the color variations depicted in their patients; however, this term more frequently is used in ophthalmology.
Orange palpebral spots commonly appear as asymptomatic, yellow-orange, symmetric lesions with a predilection for the recessed areas of the superior eyelids but also can present on the canthi and inferior eyelids. The discolorations are more easily visible on fair skin and have been reported to measure from 10 to 15 mm in the long axis.3 Assouly et al1 described the orange spots as having indistinct margins, with borders similar to “sand on a sea shore.” Orange palpebral spots can be a persistent discoloration, and there are no reports of spontaneous regression. No known association with malignancy or systemic illness has been reported.
Case reports of OPSs describe histologic similarities between specimens, including increased adipose tissue and pigment-laden macrophages in the superficial dermis.2 The pigmented deposits sometimes may be found in the basal keratinocytes of the epidermis and turn black with Fontana-Masson stain.1 No inflammatory infiltrates, necrosis, or xanthomization are characteristically found. Stains for iron, mucin, and amyloid also have been negative.2
The cause of pigmentation in OPSs is unknown; however, lipofuscin deposits and high-situated adipocytes in the reticular dermis colored by carotenoids have been proposed as possible mechanisms.1 No unifying cause for pigmentation in the serum (eg, cholesterol, triglycerides, thyroid-stimulating hormone, free retinol, vitamin E, carotenoids) was found in 11 of 27 patients with OPSs assessed by Assouly et al.1 In one case, lipofuscin, a degradation product of lysosomes, was detected by microscopic autofluorescence in the superficial dermis. However, lipofuscin typically is a breakdown product associated with aging, and OPSs have been present in patients as young as 28 years.1 Local trauma related to eye rubbing is another theory that has been proposed due to the finding of melanin in the superficial dermis. However, the absence of hemosiderin deposits as well as the extensive duration of the discolorations makes local trauma a less likely explanation for the etiology of OPSs.2
The clinical differential diagnosis for OPSs includes xanthelasma, jaundice, and carotenoderma. Xanthelasma presents as elevated yellow plaques usually found over the medial aspect of the eyes. In contrast, OPSs are nonelevated with both orange and yellow hues typically present. Histologic samples of xanthelasma are characterized by lipid-laden macrophages (foam cells) in the dermis in contrast to the adipose tissue seen in OPSs that has not been phagocytized.1,2 The lack of scleral icterus made jaundice an unlikely diagnosis in our patient. Bilirubin elevations substantial enough to cause skin discoloration also would be expected to discolor the conjunctiva. In carotenoderma, carotenoids are deposited in the sweat and sebum of the stratum corneum with the orange pigmentation most prominent in regions of increased sweating such as the palms, soles, and nasolabial folds.4 Our patient’s lack of discoloration in places other than the periorbital region made carotenoderma less likely.
In the study by Assouly et al,1 10 of 11 patients who underwent laboratory analysis self-reported eating a diet rich in fruit and vegetables, though no standardized questionnaire was given. One patient was found to have an elevated vitamin E level, and in 5 cases there was an elevated level of β-cryptoxanthin. The significance of these elevations in such a small minority is unknown, and increased β-cryptoxanthin has been attributed to increased consumption of citrus fruits during the winter season. Our patient reported ingesting a daily oral supplement rich in carotenoids that constituted 60% of the daily value of vitamin E including mixed tocopherols as well as 90% of the daily value of vitamin A with many sources of carotenoids including beta-carotenes, lutein/zeaxanthin, lycopene, and astaxanthin. An invasive biopsy was not taken in this case, as OPSs largely are diagnosed clinically. Greater awareness and recognition of OPSs may help to identify common underlying causes for this unique diagnosis.
- Assouly P, Cavelier-Balloy B, Dupré T. Orange palpebral spots. Dermatology. 2008;216:166-170.
- Belliveau MJ, Odashiro AN, Harvey JT. Yellow-orange palpebral spots. Ophthalmology. 2015;122:2139-2140.
- Kluger N, Guillot B. Bilateral orange discoloration of the upper eyelids: a quiz. Acta Derm Venereol. 2011;91:211-212.
- Maharshak N, Shapiro J, Trau H. Carotenoderma—a review of the current literature. Int J Dermatol. 2003;42:178-181.
- Assouly P, Cavelier-Balloy B, Dupré T. Orange palpebral spots. Dermatology. 2008;216:166-170.
- Belliveau MJ, Odashiro AN, Harvey JT. Yellow-orange palpebral spots. Ophthalmology. 2015;122:2139-2140.
- Kluger N, Guillot B. Bilateral orange discoloration of the upper eyelids: a quiz. Acta Derm Venereol. 2011;91:211-212.
- Maharshak N, Shapiro J, Trau H. Carotenoderma—a review of the current literature. Int J Dermatol. 2003;42:178-181.
A 63-year-old White man with a history of melanoma presented to our dermatology clinic for evaluation of gradually worsening yellow discoloration around the eyes of 2 years’ duration. Physical examination revealed periorbital yellow-orange patches (top). The discolorations were nonelevated and nonpalpable. Dermoscopy revealed yellow blotches with sparing of the hair follicles (bottom). The remainder of the skin examination was unremarkable.
A Dermatology Hospitalist Team’s Response to the Inpatient Consult Flowchart
To the Editor:
We read with interest the Cutis article by Dobkin et al1 (Cutis. 2022;109:218-220) regarding guidelines for inpatient and emergency department dermatology consultations. We agree with the authors that dermatology training is lacking in other medical specialties, which makes it challenging for teams to assess the appropriateness of a dermatology consultation in the inpatient setting. Inpatient dermatology consultation can be utilized in a hospital system to aid in rapid and accurate diagnosis, avoid inappropriate therapies, and decrease length of stay2 and readmission rates3 while providing education to the primary teams. This is precisely why in many instances the availability of inpatient dermatology consultation is so important because nondermatologists often are unable to determine whether a rash is life-threatening, benign, or something in between. From the perspective of dermatology hospitalists, there is room for improvement in the flowchart Dobkin et al1 presented to guide inpatient dermatology consultation.
To have a productive relationship with our internal medicine, surgery, pediatrics, psychiatry, and other hospital-based colleagues, we must keep an open mind when a consultation is received. We feel that the flowchart proposed by Dobkin et al1 presents too narrow a viewpoint on the utility of inpatient dermatology. It rests on assertions that other teams will be able to determine the appropriate dermatologic diagnosis without involving a dermatologist, which often is not the case.
We disagree with several recommendations in the flowchart, the first being the assertion that patients who are “hemodynamically unstable due to [a] nondermatologic problem (eg, intubated on pressors, febrile, and hypotensive)” are not appropriate for inpatient dermatology consultation.1 Although dermatologists do not commonly encounter patients with critical illness in the outpatient clinic, dermatology consultation can be extremely helpful and even lifesaving in the inpatient setting. It is unrealistic to expect the primary teams to know whether cutaneous manifestations potentially could be related to the patient’s overall clinical picture. On the contrary, we would encourage the primary team in charge of a hemodynamically unstable patient to consult dermatology at the first sign of an unexplained rash. Take for example an acutely ill patient who develops retiform purpura. There are well-established dermatology guidelines for the workup of retiform purpura,4 including prompt biopsy and assessment of broad, potentially life-threatening differential diagnoses from calciphylaxis to angioinvasive fungal infection. In this scenario, the dermatology consultant may render the correct diagnosis and recommend immediate treatment that could be lifesaving.
Secondly, we do not agree with the recommendation that a patient in hospice care is not appropriate for inpatient dermatology consultation. Patients receiving hospice or palliative care have high rates of potentially symptomatic cutaneous diseases,5 including intertrigo and dermatitis—comprising stasis, seborrheic, and contact dermatitis.6 Although aggressive intervention for asymptomatic benign or malignant skin conditions may not be in line with their goals of care, an inpatient dermatology consultation can reduce symptoms and improve quality of life. This population also is one that is unlikely to be able to attend an outpatient dermatology clinic appointment and therefore are good candidates for inpatient consultation.
Lastly, we want to highlight the difference between a stable chronic dermatologic disease and an acute flare that may occur while a patient is hospitalized, regardless of whether it is the reason for admission. For example, a patient with psoriasis affecting limited body surface area who is hospitalized for a myocardial infarction is not appropriate for a dermatology consultation. However, if that same patient develops erythroderma while they are hospitalized for cardiac monitoring, it would certainly be appropriate for dermatology to be consulted. Additionally, there are times when a chronic skin disease is the reason for hospitalization; dermatology, although technically a consulting service, would be the primary decision-maker for the patient’s care in this situation. In these scenarios, it is important for the patient to be able to establish care for long-term outpatient management of their condition; however, it is prudent to involve dermatology while the patient is acutely hospitalized to guide their treatment plan until they are able to see a dermatologist after discharge.
In conclusion, we believe that hospital dermatology is a valuable tool that can be utilized in many different scenarios. Although there are certainly situations more appropriate for outpatient dermatology referral, we would caution against overly simplified algorithms that could discourage valuable inpatient dermatology consultations. It often is worth a conversation with your dermatology consultant (when available at an institution) to determine the best course of action for each patient. Additionally, we recognize the need for more formalized guidelines on when to pursue inpatient dermatology consultation. We are members of the Society of Dermatology Hospitalists and encourage readers to reference their website, which provides additional resources on inpatient dermatology (https://societydermatologyhospitalists.com/inpatient-dermatology-literature/).
Authors’ Response
We appreciate the letter in response to our commentary on the appropriateness of inpatient dermatology consultations. It is the continued refining and re-evaluation of concepts such as these that allow our field to grow and improve knowledge and patient care.
We sought to provide a nonpatronizing yet simple consultation flowchart that would help guide triage of patients in need or not in need of dermatologic evaluation by the inpatient teams. Understandably, the impressions of our flowchart have been variable based on different readers’ medical backgrounds and experiences. It is certainly possible that our flowchart lacked certain exceptions and oversimplified certain concepts, and we welcome further refining of this flowchart to better guide inpatient dermatology consultations.
We do, however, disagree that the primary team would not know whether a patient is intubated in the intensive care unit for a dermatology reason. If the patient is in such a status, it would be pertinent for the primary team to conduct a timely workup that could include consultations until a diagnosis is made. We were not implying that every dermatology consultation in the intensive care unit is unwarranted, especially if it can lead to a primary dermatologic diagnosis. We do believe that a thorough history could elicit an allergy or other chronic skin condition that could save resources and spending within a hospital. Likewise, psoriasis comes in many different presentations, and although we do not believe a consultation for chronic psoriatic plaques is appropriate in the hospital, it is absolutely appropriate for a patient who is erythrodermic from any cause.
Our flowchart was intended to be the first step to providing education on when consultations are appropriate, and further refinement will be necessary.
Hershel Dobkin, MD; Timothy Blackwell, BS; Robin Ashinoff, MD
Drs. Dobkin and Ashinoff are from Hackensack University Medical Center, New Jersey. Mr. Blackwell is from the Rowan University School of Osteopathic Medicine, Stratford, New Jersey.
The authors report no conflict of interest.
Correspondence: Hershel Dobkin, MD, Hackensack University Medical Center, 30 Prospect Ave, Hackensack, NJ 07601 ([email protected]).
- Dobkin H, Blackwell T, Ashinoff R. When are inpatient and emergency dermatologic consultations appropriate? Cutis. 2022;109:218-220. doi:10.12788/cutis.0492
- Ko LN, Garza-Mayers AC, St John J, et al. Effect of dermatology consultation on outcomes for patients with presumed cellulitis: a randomized clinical trial. JAMA Dermatol. 2018;154:529-536. doi:10.1001/jamadermatol.2017.6196
- Hu L, Haynes H, Ferrazza D, et al. Impact of specialist consultations on inpatient admissions for dermatology-specific and related DRGs. J Gen Intern Med. 2013;28:1477-1482. doi:10.1007/s11606-013-2440-2
- Georgesen C, Fox LP, Harp J. Retiform purpura: a diagnostic approach. J Am Acad Dermatol. 2020;82:783-796. doi:10.1016/j.jaad.2019.07.112
- Pisano C, Paladichuk H, Keeling B. Dermatology in palliative medicine [published online October 14, 2021]. BMJ Support Palliat Care. doi:10.1136/bmjspcare-2021-003342
- Barnabé C, Daeninck P. “Beauty is only skin deep”: prevalence of dermatologic disease on a palliative care unit. J Pain Symptom Manage. 2005;29:419-422. doi:10.1016/j.jpainsymman.2004.08.009
To the Editor:
We read with interest the Cutis article by Dobkin et al1 (Cutis. 2022;109:218-220) regarding guidelines for inpatient and emergency department dermatology consultations. We agree with the authors that dermatology training is lacking in other medical specialties, which makes it challenging for teams to assess the appropriateness of a dermatology consultation in the inpatient setting. Inpatient dermatology consultation can be utilized in a hospital system to aid in rapid and accurate diagnosis, avoid inappropriate therapies, and decrease length of stay2 and readmission rates3 while providing education to the primary teams. This is precisely why in many instances the availability of inpatient dermatology consultation is so important because nondermatologists often are unable to determine whether a rash is life-threatening, benign, or something in between. From the perspective of dermatology hospitalists, there is room for improvement in the flowchart Dobkin et al1 presented to guide inpatient dermatology consultation.
To have a productive relationship with our internal medicine, surgery, pediatrics, psychiatry, and other hospital-based colleagues, we must keep an open mind when a consultation is received. We feel that the flowchart proposed by Dobkin et al1 presents too narrow a viewpoint on the utility of inpatient dermatology. It rests on assertions that other teams will be able to determine the appropriate dermatologic diagnosis without involving a dermatologist, which often is not the case.
We disagree with several recommendations in the flowchart, the first being the assertion that patients who are “hemodynamically unstable due to [a] nondermatologic problem (eg, intubated on pressors, febrile, and hypotensive)” are not appropriate for inpatient dermatology consultation.1 Although dermatologists do not commonly encounter patients with critical illness in the outpatient clinic, dermatology consultation can be extremely helpful and even lifesaving in the inpatient setting. It is unrealistic to expect the primary teams to know whether cutaneous manifestations potentially could be related to the patient’s overall clinical picture. On the contrary, we would encourage the primary team in charge of a hemodynamically unstable patient to consult dermatology at the first sign of an unexplained rash. Take for example an acutely ill patient who develops retiform purpura. There are well-established dermatology guidelines for the workup of retiform purpura,4 including prompt biopsy and assessment of broad, potentially life-threatening differential diagnoses from calciphylaxis to angioinvasive fungal infection. In this scenario, the dermatology consultant may render the correct diagnosis and recommend immediate treatment that could be lifesaving.
Secondly, we do not agree with the recommendation that a patient in hospice care is not appropriate for inpatient dermatology consultation. Patients receiving hospice or palliative care have high rates of potentially symptomatic cutaneous diseases,5 including intertrigo and dermatitis—comprising stasis, seborrheic, and contact dermatitis.6 Although aggressive intervention for asymptomatic benign or malignant skin conditions may not be in line with their goals of care, an inpatient dermatology consultation can reduce symptoms and improve quality of life. This population also is one that is unlikely to be able to attend an outpatient dermatology clinic appointment and therefore are good candidates for inpatient consultation.
Lastly, we want to highlight the difference between a stable chronic dermatologic disease and an acute flare that may occur while a patient is hospitalized, regardless of whether it is the reason for admission. For example, a patient with psoriasis affecting limited body surface area who is hospitalized for a myocardial infarction is not appropriate for a dermatology consultation. However, if that same patient develops erythroderma while they are hospitalized for cardiac monitoring, it would certainly be appropriate for dermatology to be consulted. Additionally, there are times when a chronic skin disease is the reason for hospitalization; dermatology, although technically a consulting service, would be the primary decision-maker for the patient’s care in this situation. In these scenarios, it is important for the patient to be able to establish care for long-term outpatient management of their condition; however, it is prudent to involve dermatology while the patient is acutely hospitalized to guide their treatment plan until they are able to see a dermatologist after discharge.
In conclusion, we believe that hospital dermatology is a valuable tool that can be utilized in many different scenarios. Although there are certainly situations more appropriate for outpatient dermatology referral, we would caution against overly simplified algorithms that could discourage valuable inpatient dermatology consultations. It often is worth a conversation with your dermatology consultant (when available at an institution) to determine the best course of action for each patient. Additionally, we recognize the need for more formalized guidelines on when to pursue inpatient dermatology consultation. We are members of the Society of Dermatology Hospitalists and encourage readers to reference their website, which provides additional resources on inpatient dermatology (https://societydermatologyhospitalists.com/inpatient-dermatology-literature/).
Authors’ Response
We appreciate the letter in response to our commentary on the appropriateness of inpatient dermatology consultations. It is the continued refining and re-evaluation of concepts such as these that allow our field to grow and improve knowledge and patient care.
We sought to provide a nonpatronizing yet simple consultation flowchart that would help guide triage of patients in need or not in need of dermatologic evaluation by the inpatient teams. Understandably, the impressions of our flowchart have been variable based on different readers’ medical backgrounds and experiences. It is certainly possible that our flowchart lacked certain exceptions and oversimplified certain concepts, and we welcome further refining of this flowchart to better guide inpatient dermatology consultations.
We do, however, disagree that the primary team would not know whether a patient is intubated in the intensive care unit for a dermatology reason. If the patient is in such a status, it would be pertinent for the primary team to conduct a timely workup that could include consultations until a diagnosis is made. We were not implying that every dermatology consultation in the intensive care unit is unwarranted, especially if it can lead to a primary dermatologic diagnosis. We do believe that a thorough history could elicit an allergy or other chronic skin condition that could save resources and spending within a hospital. Likewise, psoriasis comes in many different presentations, and although we do not believe a consultation for chronic psoriatic plaques is appropriate in the hospital, it is absolutely appropriate for a patient who is erythrodermic from any cause.
Our flowchart was intended to be the first step to providing education on when consultations are appropriate, and further refinement will be necessary.
Hershel Dobkin, MD; Timothy Blackwell, BS; Robin Ashinoff, MD
Drs. Dobkin and Ashinoff are from Hackensack University Medical Center, New Jersey. Mr. Blackwell is from the Rowan University School of Osteopathic Medicine, Stratford, New Jersey.
The authors report no conflict of interest.
Correspondence: Hershel Dobkin, MD, Hackensack University Medical Center, 30 Prospect Ave, Hackensack, NJ 07601 ([email protected]).
To the Editor:
We read with interest the Cutis article by Dobkin et al1 (Cutis. 2022;109:218-220) regarding guidelines for inpatient and emergency department dermatology consultations. We agree with the authors that dermatology training is lacking in other medical specialties, which makes it challenging for teams to assess the appropriateness of a dermatology consultation in the inpatient setting. Inpatient dermatology consultation can be utilized in a hospital system to aid in rapid and accurate diagnosis, avoid inappropriate therapies, and decrease length of stay2 and readmission rates3 while providing education to the primary teams. This is precisely why in many instances the availability of inpatient dermatology consultation is so important because nondermatologists often are unable to determine whether a rash is life-threatening, benign, or something in between. From the perspective of dermatology hospitalists, there is room for improvement in the flowchart Dobkin et al1 presented to guide inpatient dermatology consultation.
To have a productive relationship with our internal medicine, surgery, pediatrics, psychiatry, and other hospital-based colleagues, we must keep an open mind when a consultation is received. We feel that the flowchart proposed by Dobkin et al1 presents too narrow a viewpoint on the utility of inpatient dermatology. It rests on assertions that other teams will be able to determine the appropriate dermatologic diagnosis without involving a dermatologist, which often is not the case.
We disagree with several recommendations in the flowchart, the first being the assertion that patients who are “hemodynamically unstable due to [a] nondermatologic problem (eg, intubated on pressors, febrile, and hypotensive)” are not appropriate for inpatient dermatology consultation.1 Although dermatologists do not commonly encounter patients with critical illness in the outpatient clinic, dermatology consultation can be extremely helpful and even lifesaving in the inpatient setting. It is unrealistic to expect the primary teams to know whether cutaneous manifestations potentially could be related to the patient’s overall clinical picture. On the contrary, we would encourage the primary team in charge of a hemodynamically unstable patient to consult dermatology at the first sign of an unexplained rash. Take for example an acutely ill patient who develops retiform purpura. There are well-established dermatology guidelines for the workup of retiform purpura,4 including prompt biopsy and assessment of broad, potentially life-threatening differential diagnoses from calciphylaxis to angioinvasive fungal infection. In this scenario, the dermatology consultant may render the correct diagnosis and recommend immediate treatment that could be lifesaving.
Secondly, we do not agree with the recommendation that a patient in hospice care is not appropriate for inpatient dermatology consultation. Patients receiving hospice or palliative care have high rates of potentially symptomatic cutaneous diseases,5 including intertrigo and dermatitis—comprising stasis, seborrheic, and contact dermatitis.6 Although aggressive intervention for asymptomatic benign or malignant skin conditions may not be in line with their goals of care, an inpatient dermatology consultation can reduce symptoms and improve quality of life. This population also is one that is unlikely to be able to attend an outpatient dermatology clinic appointment and therefore are good candidates for inpatient consultation.
Lastly, we want to highlight the difference between a stable chronic dermatologic disease and an acute flare that may occur while a patient is hospitalized, regardless of whether it is the reason for admission. For example, a patient with psoriasis affecting limited body surface area who is hospitalized for a myocardial infarction is not appropriate for a dermatology consultation. However, if that same patient develops erythroderma while they are hospitalized for cardiac monitoring, it would certainly be appropriate for dermatology to be consulted. Additionally, there are times when a chronic skin disease is the reason for hospitalization; dermatology, although technically a consulting service, would be the primary decision-maker for the patient’s care in this situation. In these scenarios, it is important for the patient to be able to establish care for long-term outpatient management of their condition; however, it is prudent to involve dermatology while the patient is acutely hospitalized to guide their treatment plan until they are able to see a dermatologist after discharge.
In conclusion, we believe that hospital dermatology is a valuable tool that can be utilized in many different scenarios. Although there are certainly situations more appropriate for outpatient dermatology referral, we would caution against overly simplified algorithms that could discourage valuable inpatient dermatology consultations. It often is worth a conversation with your dermatology consultant (when available at an institution) to determine the best course of action for each patient. Additionally, we recognize the need for more formalized guidelines on when to pursue inpatient dermatology consultation. We are members of the Society of Dermatology Hospitalists and encourage readers to reference their website, which provides additional resources on inpatient dermatology (https://societydermatologyhospitalists.com/inpatient-dermatology-literature/).
Authors’ Response
We appreciate the letter in response to our commentary on the appropriateness of inpatient dermatology consultations. It is the continued refining and re-evaluation of concepts such as these that allow our field to grow and improve knowledge and patient care.
We sought to provide a nonpatronizing yet simple consultation flowchart that would help guide triage of patients in need or not in need of dermatologic evaluation by the inpatient teams. Understandably, the impressions of our flowchart have been variable based on different readers’ medical backgrounds and experiences. It is certainly possible that our flowchart lacked certain exceptions and oversimplified certain concepts, and we welcome further refining of this flowchart to better guide inpatient dermatology consultations.
We do, however, disagree that the primary team would not know whether a patient is intubated in the intensive care unit for a dermatology reason. If the patient is in such a status, it would be pertinent for the primary team to conduct a timely workup that could include consultations until a diagnosis is made. We were not implying that every dermatology consultation in the intensive care unit is unwarranted, especially if it can lead to a primary dermatologic diagnosis. We do believe that a thorough history could elicit an allergy or other chronic skin condition that could save resources and spending within a hospital. Likewise, psoriasis comes in many different presentations, and although we do not believe a consultation for chronic psoriatic plaques is appropriate in the hospital, it is absolutely appropriate for a patient who is erythrodermic from any cause.
Our flowchart was intended to be the first step to providing education on when consultations are appropriate, and further refinement will be necessary.
Hershel Dobkin, MD; Timothy Blackwell, BS; Robin Ashinoff, MD
Drs. Dobkin and Ashinoff are from Hackensack University Medical Center, New Jersey. Mr. Blackwell is from the Rowan University School of Osteopathic Medicine, Stratford, New Jersey.
The authors report no conflict of interest.
Correspondence: Hershel Dobkin, MD, Hackensack University Medical Center, 30 Prospect Ave, Hackensack, NJ 07601 ([email protected]).
- Dobkin H, Blackwell T, Ashinoff R. When are inpatient and emergency dermatologic consultations appropriate? Cutis. 2022;109:218-220. doi:10.12788/cutis.0492
- Ko LN, Garza-Mayers AC, St John J, et al. Effect of dermatology consultation on outcomes for patients with presumed cellulitis: a randomized clinical trial. JAMA Dermatol. 2018;154:529-536. doi:10.1001/jamadermatol.2017.6196
- Hu L, Haynes H, Ferrazza D, et al. Impact of specialist consultations on inpatient admissions for dermatology-specific and related DRGs. J Gen Intern Med. 2013;28:1477-1482. doi:10.1007/s11606-013-2440-2
- Georgesen C, Fox LP, Harp J. Retiform purpura: a diagnostic approach. J Am Acad Dermatol. 2020;82:783-796. doi:10.1016/j.jaad.2019.07.112
- Pisano C, Paladichuk H, Keeling B. Dermatology in palliative medicine [published online October 14, 2021]. BMJ Support Palliat Care. doi:10.1136/bmjspcare-2021-003342
- Barnabé C, Daeninck P. “Beauty is only skin deep”: prevalence of dermatologic disease on a palliative care unit. J Pain Symptom Manage. 2005;29:419-422. doi:10.1016/j.jpainsymman.2004.08.009
- Dobkin H, Blackwell T, Ashinoff R. When are inpatient and emergency dermatologic consultations appropriate? Cutis. 2022;109:218-220. doi:10.12788/cutis.0492
- Ko LN, Garza-Mayers AC, St John J, et al. Effect of dermatology consultation on outcomes for patients with presumed cellulitis: a randomized clinical trial. JAMA Dermatol. 2018;154:529-536. doi:10.1001/jamadermatol.2017.6196
- Hu L, Haynes H, Ferrazza D, et al. Impact of specialist consultations on inpatient admissions for dermatology-specific and related DRGs. J Gen Intern Med. 2013;28:1477-1482. doi:10.1007/s11606-013-2440-2
- Georgesen C, Fox LP, Harp J. Retiform purpura: a diagnostic approach. J Am Acad Dermatol. 2020;82:783-796. doi:10.1016/j.jaad.2019.07.112
- Pisano C, Paladichuk H, Keeling B. Dermatology in palliative medicine [published online October 14, 2021]. BMJ Support Palliat Care. doi:10.1136/bmjspcare-2021-003342
- Barnabé C, Daeninck P. “Beauty is only skin deep”: prevalence of dermatologic disease on a palliative care unit. J Pain Symptom Manage. 2005;29:419-422. doi:10.1016/j.jpainsymman.2004.08.009