User login
Obinutuzumab-based regimens yield durable remissions in CLL
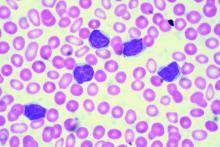
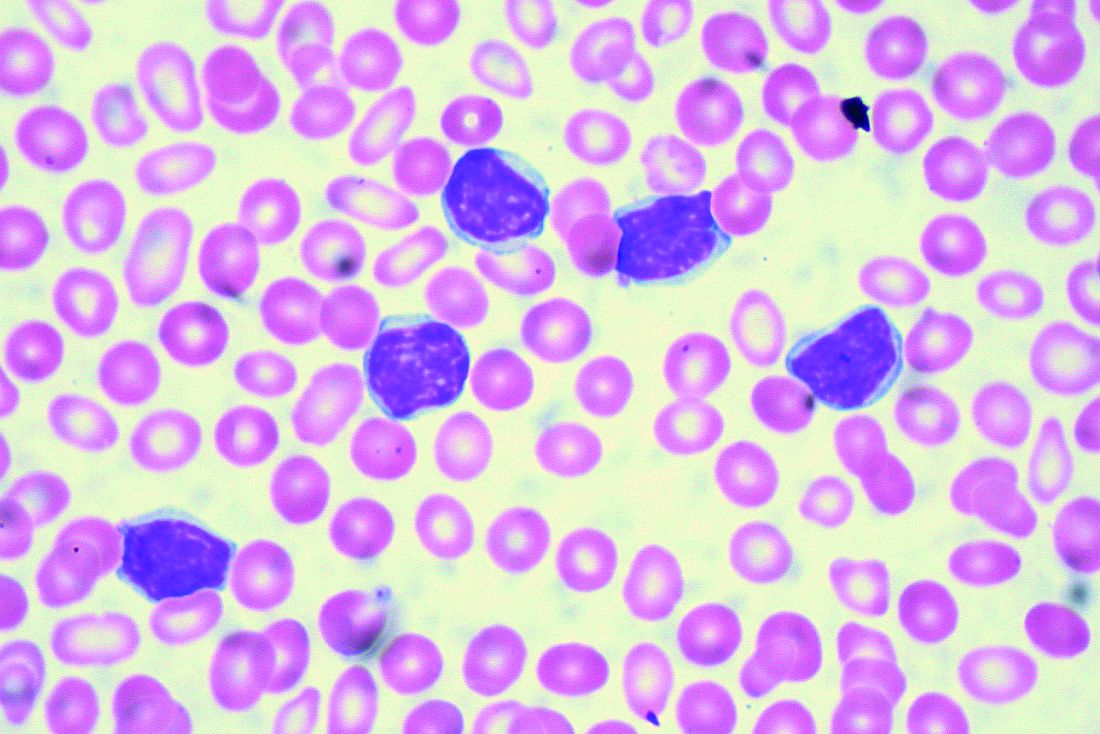
Two different obinutuzumab-based chemoimmunotherapy regimens resulted in excellent long-term disease control as front-line therapy for chronic lymphocytic leukemia (CLL), investigators said in a follow-up report on a phase 1b study.
Both obinutuzumab plus fludarabine/cyclophosphamide (G-FC) and obinutuzumab plus bendamustine (G-B) were well tolerated, with adverse events similar to what has been reported in rituximab-containing immunotherapy regimens, they said in the report of final results from the GALTON trial.
Most evaluable patients had B-cell recovery by 36 months in the study, which included a population of CLL patients largely without 17p deletions, said Jennifer R. Brown, MD, PhD, of Dana-Farber Cancer Institute, Boston, and her coinvestigators.
“These data support moving forward with these regimens in subsequent trials, which are currently ongoing,” they said in their report on the study, which appears in Blood.
The open-label, parallel-arm, multicenter phase 1b GALTON study included 41 patients with CLL, of whom 21 received G-FC and 20 received G-B for up to six cycles of 28 days each. The median age was 60 years, and about one-third of patients had Rai stage III or IV disease. Only one patient had del(17p), and nearly half of patients tested (17 of 38 patients) had unmutated immunoglobulin heavy-chain variable region gene (IGHV). Six patients had del(11q), including four in the G-FC arm and two in the G-B arm.
Both G-FC and G-B had manageable toxicities, with infusion-related reactions being the most common adverse event, occurring in 88% (20% grade 3 or 4), Dr. Brown and her colleagues reported, adding that grade 3 or 4 neutropenia was seen in 48% of the G-FC arm and 55% of the G-B arm.
The objective response rate (ORR) was 62% for G-FC and 90% for GB.
“The ORR in the G-FC arm likely does not reflect the true activity of the regimen, as it is based on an intent-to-treat analysis,” the investigators said.
With a median observation time of 40.4 months, 95% of patients were alive, and 90% had not experienced a progression-free survival event.
Nine patients in the G-FC arm underwent minimal residual disease (MRD) testing in peripheral blood; 100% had undetectable MRD, according to the report.
“With the caveat of small patient numbers and inevitable differences in patient populations across studies, these results suggest that G-FC may clear residual disease more effectively than rituximab plus FC,” the investigators wrote.
Previous studies of R-FC showed an undetectable MRD rate of 45% or less, they said.
The study was sponsored by Genentech. The investigators reported disclosures related to Genentech/Roche and other companies.
SOURCE: Brown JR et al. Blood. 2018 Dec 28. doi: 10.1182/blood-2018-06-857714.
Two different obinutuzumab-based chemoimmunotherapy regimens resulted in excellent long-term disease control as front-line therapy for chronic lymphocytic leukemia (CLL), investigators said in a follow-up report on a phase 1b study.
Both obinutuzumab plus fludarabine/cyclophosphamide (G-FC) and obinutuzumab plus bendamustine (G-B) were well tolerated, with adverse events similar to what has been reported in rituximab-containing immunotherapy regimens, they said in the report of final results from the GALTON trial.
Most evaluable patients had B-cell recovery by 36 months in the study, which included a population of CLL patients largely without 17p deletions, said Jennifer R. Brown, MD, PhD, of Dana-Farber Cancer Institute, Boston, and her coinvestigators.
“These data support moving forward with these regimens in subsequent trials, which are currently ongoing,” they said in their report on the study, which appears in Blood.
The open-label, parallel-arm, multicenter phase 1b GALTON study included 41 patients with CLL, of whom 21 received G-FC and 20 received G-B for up to six cycles of 28 days each. The median age was 60 years, and about one-third of patients had Rai stage III or IV disease. Only one patient had del(17p), and nearly half of patients tested (17 of 38 patients) had unmutated immunoglobulin heavy-chain variable region gene (IGHV). Six patients had del(11q), including four in the G-FC arm and two in the G-B arm.
Both G-FC and G-B had manageable toxicities, with infusion-related reactions being the most common adverse event, occurring in 88% (20% grade 3 or 4), Dr. Brown and her colleagues reported, adding that grade 3 or 4 neutropenia was seen in 48% of the G-FC arm and 55% of the G-B arm.
The objective response rate (ORR) was 62% for G-FC and 90% for GB.
“The ORR in the G-FC arm likely does not reflect the true activity of the regimen, as it is based on an intent-to-treat analysis,” the investigators said.
With a median observation time of 40.4 months, 95% of patients were alive, and 90% had not experienced a progression-free survival event.
Nine patients in the G-FC arm underwent minimal residual disease (MRD) testing in peripheral blood; 100% had undetectable MRD, according to the report.
“With the caveat of small patient numbers and inevitable differences in patient populations across studies, these results suggest that G-FC may clear residual disease more effectively than rituximab plus FC,” the investigators wrote.
Previous studies of R-FC showed an undetectable MRD rate of 45% or less, they said.
The study was sponsored by Genentech. The investigators reported disclosures related to Genentech/Roche and other companies.
SOURCE: Brown JR et al. Blood. 2018 Dec 28. doi: 10.1182/blood-2018-06-857714.
Two different obinutuzumab-based chemoimmunotherapy regimens resulted in excellent long-term disease control as front-line therapy for chronic lymphocytic leukemia (CLL), investigators said in a follow-up report on a phase 1b study.
Both obinutuzumab plus fludarabine/cyclophosphamide (G-FC) and obinutuzumab plus bendamustine (G-B) were well tolerated, with adverse events similar to what has been reported in rituximab-containing immunotherapy regimens, they said in the report of final results from the GALTON trial.
Most evaluable patients had B-cell recovery by 36 months in the study, which included a population of CLL patients largely without 17p deletions, said Jennifer R. Brown, MD, PhD, of Dana-Farber Cancer Institute, Boston, and her coinvestigators.
“These data support moving forward with these regimens in subsequent trials, which are currently ongoing,” they said in their report on the study, which appears in Blood.
The open-label, parallel-arm, multicenter phase 1b GALTON study included 41 patients with CLL, of whom 21 received G-FC and 20 received G-B for up to six cycles of 28 days each. The median age was 60 years, and about one-third of patients had Rai stage III or IV disease. Only one patient had del(17p), and nearly half of patients tested (17 of 38 patients) had unmutated immunoglobulin heavy-chain variable region gene (IGHV). Six patients had del(11q), including four in the G-FC arm and two in the G-B arm.
Both G-FC and G-B had manageable toxicities, with infusion-related reactions being the most common adverse event, occurring in 88% (20% grade 3 or 4), Dr. Brown and her colleagues reported, adding that grade 3 or 4 neutropenia was seen in 48% of the G-FC arm and 55% of the G-B arm.
The objective response rate (ORR) was 62% for G-FC and 90% for GB.
“The ORR in the G-FC arm likely does not reflect the true activity of the regimen, as it is based on an intent-to-treat analysis,” the investigators said.
With a median observation time of 40.4 months, 95% of patients were alive, and 90% had not experienced a progression-free survival event.
Nine patients in the G-FC arm underwent minimal residual disease (MRD) testing in peripheral blood; 100% had undetectable MRD, according to the report.
“With the caveat of small patient numbers and inevitable differences in patient populations across studies, these results suggest that G-FC may clear residual disease more effectively than rituximab plus FC,” the investigators wrote.
Previous studies of R-FC showed an undetectable MRD rate of 45% or less, they said.
The study was sponsored by Genentech. The investigators reported disclosures related to Genentech/Roche and other companies.
SOURCE: Brown JR et al. Blood. 2018 Dec 28. doi: 10.1182/blood-2018-06-857714.
FROM BLOOD
Key clinical point:
Major finding: With a median observation time of 40.4 months, 95% of patients were alive, and 90% had not experienced a progression-free survival event.
Study details: Long-term follow-up of the phase 1b GALTON trial, including 41 patients with CLL.
Disclosures: The study was sponsored by Genentech. The study authors reported disclosures related to Genentech/Roche and other companies.
Source: Brown JR et al. Blood. 2018 Dec 28. doi: 10.1182/blood-2018-06-857714.
Aspirin and Omega-3 fatty acids fail
Also today, New data reveal that college students are at greater risk of meningococcal B infection, children who survive Hodgkin lymphoma face a massive increased risk for second cancers down the road, and the 2018/19 flu season shows high activity in nine states.
Amazon Alexa
Apple Podcasts
Google Podcasts
Spotify
Also today, New data reveal that college students are at greater risk of meningococcal B infection, children who survive Hodgkin lymphoma face a massive increased risk for second cancers down the road, and the 2018/19 flu season shows high activity in nine states.
Amazon Alexa
Apple Podcasts
Google Podcasts
Spotify
Also today, New data reveal that college students are at greater risk of meningococcal B infection, children who survive Hodgkin lymphoma face a massive increased risk for second cancers down the road, and the 2018/19 flu season shows high activity in nine states.
Amazon Alexa
Apple Podcasts
Google Podcasts
Spotify
ALL chemotherapy looks effective in mixed phenotype leukemia
SAN DIEGO – The majority of pediatric patients with mixed phenotype acute leukemia (MPAL) who were treated with acute lymphoblastic leukemia (ALL)–directed chemotherapy achieved a minimum residual disease (MRD)–negative complete response by the end of consolidation, according to findings from a multicenter retrospective cohort study.
The cohort included 94 patients aged 1-21 years who met strict World Health Organization MPAL criteria and were treated between 2008 and 2016 at one of six U.S. institutions. Most had B/myeloid phenotype (89%), and 87 patients were treated with an ALL regimen, Etan Orgel, MD, reported at the annual meeting of the American Society of Hematology.
Of those 87 patients, 81 (93%) experienced an end-of-induction (EOI) complete response. One patient died during induction and six had induction failures, defined as either disease progression before EOI (two patients) or EOI MRD of 5% or greater (three patients), said Dr. Orgel of the University of Southern California, Los Angeles, and Children’s Hospital Los Angeles.
The MRD-negative rates, defined as MRD less than 0.01%, were 70% at EOI and 86% at EOI or end of consolidation (EOC); 12 of 14 patients who were MRD positive at EOI and continued on ALL therapy achieved an EOC MRD-negative complete response, including 8 of 8 with EOI MRD of 0.01%-0.09% and 4 of 6 with EOI MRD of 1% or greater.
Event-free survival at 5 years in the 78 patients without hematopoietic stem cell transplant at first remission was 75%, and 5-year overall survival was 89%, “thus demonstrating that, for a majority of patients, transplant in first remission may not be necessary,” Dr. Orgel said. “This is very different from the approach used at many adult centers and many of the adult recommendations.”
Overall 5-year EOI event-free survival was 80% in the 59 patients who were MRD negative at EOI, and 13% in 25 patients who were MRD-positive at EOI. The corresponding overall survival rates were 91% and 84%.
Overall 5-year EOC event-free survival was 77% in 74 patients who were MRD negative at EOC and was unavailable in 3 patients who were MRD positive at EOC, although all three were salvaged. The corresponding EOC overall survival rates were 89% and “not available,” Dr. Orgel reported.
Multivariable analysis confirmed the predictive value of MRD at EOI (hazard ratio for event-free survival and overall survival, 3.77 and 3.54, respectively).
Of note, there was a possible trend toward earlier failure and a trend toward worse overall survival (HR, 4.49, P = .074) for T-lineage–containing MPAL.
“That indicates that this might be a group that needs careful scrutiny of which form of ALL therapy they receive,” he said.
MRD in pediatric MPAL is rare. Recent studies of MPAL biology show areas of similarity with ALL and AML, and while this could eventually help further subcategorize or classify the disease and lead to biology-driven therapies, it is important to know how to treat the disease today, Dr. Orgel said.
The evolving consensus is that ALL therapy is adequate for most MPAL, but there is no established threshold for MRD to enable a risk-stratified MPAL approach, he added.
The current findings suggest that ALL therapy – without hematopoietic stem cell transplant – may be sufficient to treat most patients with pediatric MPAL, Dr. Orgen reported, noting that clinical trials are necessary to prospectively validate MRD thresholds at EOI and EOC and to establish the threshold for favorable survival.
“Future research should explore either intensification of therapy or different therapies for patients with persistent MRD,” he said.
Dr. Orgel reported having no financial disclosures.
SOURCE: Oberley M et al. ASH 2018, Abstract 558.
SAN DIEGO – The majority of pediatric patients with mixed phenotype acute leukemia (MPAL) who were treated with acute lymphoblastic leukemia (ALL)–directed chemotherapy achieved a minimum residual disease (MRD)–negative complete response by the end of consolidation, according to findings from a multicenter retrospective cohort study.
The cohort included 94 patients aged 1-21 years who met strict World Health Organization MPAL criteria and were treated between 2008 and 2016 at one of six U.S. institutions. Most had B/myeloid phenotype (89%), and 87 patients were treated with an ALL regimen, Etan Orgel, MD, reported at the annual meeting of the American Society of Hematology.
Of those 87 patients, 81 (93%) experienced an end-of-induction (EOI) complete response. One patient died during induction and six had induction failures, defined as either disease progression before EOI (two patients) or EOI MRD of 5% or greater (three patients), said Dr. Orgel of the University of Southern California, Los Angeles, and Children’s Hospital Los Angeles.
The MRD-negative rates, defined as MRD less than 0.01%, were 70% at EOI and 86% at EOI or end of consolidation (EOC); 12 of 14 patients who were MRD positive at EOI and continued on ALL therapy achieved an EOC MRD-negative complete response, including 8 of 8 with EOI MRD of 0.01%-0.09% and 4 of 6 with EOI MRD of 1% or greater.
Event-free survival at 5 years in the 78 patients without hematopoietic stem cell transplant at first remission was 75%, and 5-year overall survival was 89%, “thus demonstrating that, for a majority of patients, transplant in first remission may not be necessary,” Dr. Orgel said. “This is very different from the approach used at many adult centers and many of the adult recommendations.”
Overall 5-year EOI event-free survival was 80% in the 59 patients who were MRD negative at EOI, and 13% in 25 patients who were MRD-positive at EOI. The corresponding overall survival rates were 91% and 84%.
Overall 5-year EOC event-free survival was 77% in 74 patients who were MRD negative at EOC and was unavailable in 3 patients who were MRD positive at EOC, although all three were salvaged. The corresponding EOC overall survival rates were 89% and “not available,” Dr. Orgel reported.
Multivariable analysis confirmed the predictive value of MRD at EOI (hazard ratio for event-free survival and overall survival, 3.77 and 3.54, respectively).
Of note, there was a possible trend toward earlier failure and a trend toward worse overall survival (HR, 4.49, P = .074) for T-lineage–containing MPAL.
“That indicates that this might be a group that needs careful scrutiny of which form of ALL therapy they receive,” he said.
MRD in pediatric MPAL is rare. Recent studies of MPAL biology show areas of similarity with ALL and AML, and while this could eventually help further subcategorize or classify the disease and lead to biology-driven therapies, it is important to know how to treat the disease today, Dr. Orgel said.
The evolving consensus is that ALL therapy is adequate for most MPAL, but there is no established threshold for MRD to enable a risk-stratified MPAL approach, he added.
The current findings suggest that ALL therapy – without hematopoietic stem cell transplant – may be sufficient to treat most patients with pediatric MPAL, Dr. Orgen reported, noting that clinical trials are necessary to prospectively validate MRD thresholds at EOI and EOC and to establish the threshold for favorable survival.
“Future research should explore either intensification of therapy or different therapies for patients with persistent MRD,” he said.
Dr. Orgel reported having no financial disclosures.
SOURCE: Oberley M et al. ASH 2018, Abstract 558.
SAN DIEGO – The majority of pediatric patients with mixed phenotype acute leukemia (MPAL) who were treated with acute lymphoblastic leukemia (ALL)–directed chemotherapy achieved a minimum residual disease (MRD)–negative complete response by the end of consolidation, according to findings from a multicenter retrospective cohort study.
The cohort included 94 patients aged 1-21 years who met strict World Health Organization MPAL criteria and were treated between 2008 and 2016 at one of six U.S. institutions. Most had B/myeloid phenotype (89%), and 87 patients were treated with an ALL regimen, Etan Orgel, MD, reported at the annual meeting of the American Society of Hematology.
Of those 87 patients, 81 (93%) experienced an end-of-induction (EOI) complete response. One patient died during induction and six had induction failures, defined as either disease progression before EOI (two patients) or EOI MRD of 5% or greater (three patients), said Dr. Orgel of the University of Southern California, Los Angeles, and Children’s Hospital Los Angeles.
The MRD-negative rates, defined as MRD less than 0.01%, were 70% at EOI and 86% at EOI or end of consolidation (EOC); 12 of 14 patients who were MRD positive at EOI and continued on ALL therapy achieved an EOC MRD-negative complete response, including 8 of 8 with EOI MRD of 0.01%-0.09% and 4 of 6 with EOI MRD of 1% or greater.
Event-free survival at 5 years in the 78 patients without hematopoietic stem cell transplant at first remission was 75%, and 5-year overall survival was 89%, “thus demonstrating that, for a majority of patients, transplant in first remission may not be necessary,” Dr. Orgel said. “This is very different from the approach used at many adult centers and many of the adult recommendations.”
Overall 5-year EOI event-free survival was 80% in the 59 patients who were MRD negative at EOI, and 13% in 25 patients who were MRD-positive at EOI. The corresponding overall survival rates were 91% and 84%.
Overall 5-year EOC event-free survival was 77% in 74 patients who were MRD negative at EOC and was unavailable in 3 patients who were MRD positive at EOC, although all three were salvaged. The corresponding EOC overall survival rates were 89% and “not available,” Dr. Orgel reported.
Multivariable analysis confirmed the predictive value of MRD at EOI (hazard ratio for event-free survival and overall survival, 3.77 and 3.54, respectively).
Of note, there was a possible trend toward earlier failure and a trend toward worse overall survival (HR, 4.49, P = .074) for T-lineage–containing MPAL.
“That indicates that this might be a group that needs careful scrutiny of which form of ALL therapy they receive,” he said.
MRD in pediatric MPAL is rare. Recent studies of MPAL biology show areas of similarity with ALL and AML, and while this could eventually help further subcategorize or classify the disease and lead to biology-driven therapies, it is important to know how to treat the disease today, Dr. Orgel said.
The evolving consensus is that ALL therapy is adequate for most MPAL, but there is no established threshold for MRD to enable a risk-stratified MPAL approach, he added.
The current findings suggest that ALL therapy – without hematopoietic stem cell transplant – may be sufficient to treat most patients with pediatric MPAL, Dr. Orgen reported, noting that clinical trials are necessary to prospectively validate MRD thresholds at EOI and EOC and to establish the threshold for favorable survival.
“Future research should explore either intensification of therapy or different therapies for patients with persistent MRD,” he said.
Dr. Orgel reported having no financial disclosures.
SOURCE: Oberley M et al. ASH 2018, Abstract 558.
REPORTING FROM ASH 2018
Key clinical point:
Major finding: MRD-negative rates were 70% at end of induction and 86% at end of induction or consolidation.
Study details: A retrospective cohort study of 87 pediatric MPAL patients.
Disclosures: Dr. Orgel reported having no financial disclosures.
Source: Oberley M et al. ASH 2018, Abstract 558.
FDA approves ravulizumab for treatment of paroxysmal nocturnal hemoglobinuria
The Food and Drug Administration has approved ravulizumab (Ultomiris) injection for the treatment of adult patients with paroxysmal nocturnal hemoglobinuria (PNH).
“The approval of Ultomiris will change the way that patients with PNH are treated. Prior to this approval, the only approved therapy for PNH required treatment every 2 weeks, which can be burdensome for patients and their families. Ultomiris uses a novel formulation so patients only need treatment every 8 weeks, without compromising efficacy,” Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence, said in a press release from the agency.
Patients with PNH, a rare disorder, lack a protein which protects red blood cells from being destroyed in the immune system. Episodes can be triggered by stresses on the body such as infection or physical exertion, and symptoms include severe anemia, profound fatigue, shortness of breath, intermittent episodes of dark-colored urine, kidney disease, or recurrent pain.
FDA approval for ravulizumab is based on results from a pair of clinical trials. In the first, 246 treatment-naive PNH patients received either ravulizumab or eculizumab, the current standard of care; ravulizumab was noninferior, with no patients undergoing a transfusion and all patients having similar incidence of hemolysis. In the second trial, 195 patients who had clinically stable PNH after receiving eculizumab for 6 months were randomized to receive ravulizumab or continue eculizumab; again, ravulizumab was noninferior.
The most common adverse events associated with ravulizumab were headache and respiratory tract infection. Caution is recommended when prescribing ravulizumab to patients with any type of infection.
Find the full press release on the FDA website.
The Food and Drug Administration has approved ravulizumab (Ultomiris) injection for the treatment of adult patients with paroxysmal nocturnal hemoglobinuria (PNH).
“The approval of Ultomiris will change the way that patients with PNH are treated. Prior to this approval, the only approved therapy for PNH required treatment every 2 weeks, which can be burdensome for patients and their families. Ultomiris uses a novel formulation so patients only need treatment every 8 weeks, without compromising efficacy,” Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence, said in a press release from the agency.
Patients with PNH, a rare disorder, lack a protein which protects red blood cells from being destroyed in the immune system. Episodes can be triggered by stresses on the body such as infection or physical exertion, and symptoms include severe anemia, profound fatigue, shortness of breath, intermittent episodes of dark-colored urine, kidney disease, or recurrent pain.
FDA approval for ravulizumab is based on results from a pair of clinical trials. In the first, 246 treatment-naive PNH patients received either ravulizumab or eculizumab, the current standard of care; ravulizumab was noninferior, with no patients undergoing a transfusion and all patients having similar incidence of hemolysis. In the second trial, 195 patients who had clinically stable PNH after receiving eculizumab for 6 months were randomized to receive ravulizumab or continue eculizumab; again, ravulizumab was noninferior.
The most common adverse events associated with ravulizumab were headache and respiratory tract infection. Caution is recommended when prescribing ravulizumab to patients with any type of infection.
Find the full press release on the FDA website.
The Food and Drug Administration has approved ravulizumab (Ultomiris) injection for the treatment of adult patients with paroxysmal nocturnal hemoglobinuria (PNH).
“The approval of Ultomiris will change the way that patients with PNH are treated. Prior to this approval, the only approved therapy for PNH required treatment every 2 weeks, which can be burdensome for patients and their families. Ultomiris uses a novel formulation so patients only need treatment every 8 weeks, without compromising efficacy,” Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence, said in a press release from the agency.
Patients with PNH, a rare disorder, lack a protein which protects red blood cells from being destroyed in the immune system. Episodes can be triggered by stresses on the body such as infection or physical exertion, and symptoms include severe anemia, profound fatigue, shortness of breath, intermittent episodes of dark-colored urine, kidney disease, or recurrent pain.
FDA approval for ravulizumab is based on results from a pair of clinical trials. In the first, 246 treatment-naive PNH patients received either ravulizumab or eculizumab, the current standard of care; ravulizumab was noninferior, with no patients undergoing a transfusion and all patients having similar incidence of hemolysis. In the second trial, 195 patients who had clinically stable PNH after receiving eculizumab for 6 months were randomized to receive ravulizumab or continue eculizumab; again, ravulizumab was noninferior.
The most common adverse events associated with ravulizumab were headache and respiratory tract infection. Caution is recommended when prescribing ravulizumab to patients with any type of infection.
Find the full press release on the FDA website.
FDA approves Elzonris for blastic plasmacytoid dendritic cell neoplasm
The Food and Drug Administration has approved tagraxofusp-erzs (Elzonris) infusion for the treatment of blastic plasmacytoid dendritic cell neoplasm (BPDCN) in adults and pediatric patients, 2 years of age and older.
Approval was based on efficacy in two cohorts of patients in a single-arm clinical trial. Seven patients (54%) out of 13 with untreated BPDCN achieved complete remission (CR) or CR with a skin abnormality not indicative of active disease (CRc) in the first cohort. In the second cohort of 15 patients, one patient achieved CR and one patient achieved CRc.
Common side effects for patients receiving tagraxofusp-erzs infusion were capillary leak syndrome, nausea, fatigue, peripheral edema, pyrexia, chills, and weight increase. Most common laboratory abnormalities were decreases in lymphocytes, albumin, platelets, hemoglobin, and calcium, and increases in glucose and liver enzymes (ALT and AST), the FDA said in a press statement.
The FDA placed a Boxed Warning on the drug to alert health care professionals and patients about the increased risk of capillary leak syndrome and recommends that health care providers monitor liver enzyme levels for signs of intolerance to the infusion.
BPDCN is an aggressive and rare disease of the bone marrow and blood that can affect multiple organs, including the lymph nodes and the skin. It often presents as leukemia or evolves into acute leukemia, the FDA said.
“Prior to today’s approval, there had been no FDA approved therapies for BPDCN. The standard of care has been intensive chemotherapy followed by bone marrow transplantation. Many patients with BPDCN are unable to tolerate this intensive therapy, so there is an urgent need for alternative treatment options,” Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence and acting director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research, said in the statement.
The Food and Drug Administration has approved tagraxofusp-erzs (Elzonris) infusion for the treatment of blastic plasmacytoid dendritic cell neoplasm (BPDCN) in adults and pediatric patients, 2 years of age and older.
Approval was based on efficacy in two cohorts of patients in a single-arm clinical trial. Seven patients (54%) out of 13 with untreated BPDCN achieved complete remission (CR) or CR with a skin abnormality not indicative of active disease (CRc) in the first cohort. In the second cohort of 15 patients, one patient achieved CR and one patient achieved CRc.
Common side effects for patients receiving tagraxofusp-erzs infusion were capillary leak syndrome, nausea, fatigue, peripheral edema, pyrexia, chills, and weight increase. Most common laboratory abnormalities were decreases in lymphocytes, albumin, platelets, hemoglobin, and calcium, and increases in glucose and liver enzymes (ALT and AST), the FDA said in a press statement.
The FDA placed a Boxed Warning on the drug to alert health care professionals and patients about the increased risk of capillary leak syndrome and recommends that health care providers monitor liver enzyme levels for signs of intolerance to the infusion.
BPDCN is an aggressive and rare disease of the bone marrow and blood that can affect multiple organs, including the lymph nodes and the skin. It often presents as leukemia or evolves into acute leukemia, the FDA said.
“Prior to today’s approval, there had been no FDA approved therapies for BPDCN. The standard of care has been intensive chemotherapy followed by bone marrow transplantation. Many patients with BPDCN are unable to tolerate this intensive therapy, so there is an urgent need for alternative treatment options,” Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence and acting director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research, said in the statement.
The Food and Drug Administration has approved tagraxofusp-erzs (Elzonris) infusion for the treatment of blastic plasmacytoid dendritic cell neoplasm (BPDCN) in adults and pediatric patients, 2 years of age and older.
Approval was based on efficacy in two cohorts of patients in a single-arm clinical trial. Seven patients (54%) out of 13 with untreated BPDCN achieved complete remission (CR) or CR with a skin abnormality not indicative of active disease (CRc) in the first cohort. In the second cohort of 15 patients, one patient achieved CR and one patient achieved CRc.
Common side effects for patients receiving tagraxofusp-erzs infusion were capillary leak syndrome, nausea, fatigue, peripheral edema, pyrexia, chills, and weight increase. Most common laboratory abnormalities were decreases in lymphocytes, albumin, platelets, hemoglobin, and calcium, and increases in glucose and liver enzymes (ALT and AST), the FDA said in a press statement.
The FDA placed a Boxed Warning on the drug to alert health care professionals and patients about the increased risk of capillary leak syndrome and recommends that health care providers monitor liver enzyme levels for signs of intolerance to the infusion.
BPDCN is an aggressive and rare disease of the bone marrow and blood that can affect multiple organs, including the lymph nodes and the skin. It often presents as leukemia or evolves into acute leukemia, the FDA said.
“Prior to today’s approval, there had been no FDA approved therapies for BPDCN. The standard of care has been intensive chemotherapy followed by bone marrow transplantation. Many patients with BPDCN are unable to tolerate this intensive therapy, so there is an urgent need for alternative treatment options,” Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence and acting director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research, said in the statement.
Check for neuromyelitis optica spectrum disorder in suspect HIV patients
HIV-associated neuromyelitis optica spectrum disorder (NMOSD) is a recently recognized entity and high index of suspicion is needed to diagnose these patients, according to Thomas Mathew, MD, and his colleagues at St. John’s Medical College Hospital, Bengaluru, India.
“NMOSD can be associated with a wide range of autoimmune diseases but clinicians rarely diagnose NMOSD in cases of HIV infection and HIV-associated NMOSD is rarely mentioned in the conventional classification of NMOSD,” they stated.
Dr. Mathew and his colleagues reported the results of a case study they made of six cases of HIV-NMOSD identified from the literature and 1 HIV-infected patient from a registry for NMOSD that they had established, which had a total of 25 patients with the condition.
There were four men and three women in the study, ranging from 8 years to 49 years of age. The duration of HIV infection in these patients ranged from newly detected to 15 years, according to the report, published in Multiple Sclerosis and Related Disorders (2019 Jan;27:289-93).
Optic neuritis followed by myelitis was the commonest presentation, occurring in five of the seven patients. Of these, six patients were assayed for anti–aquaporin 4 antibodies, which are considered a serological marker of neuromyelitis optica; three patients were positive and three were negative.
All patients received immunomodulatory treatment. Five of the seven patients had a poor recovery from acute attacks, but no patient had further relapses while on immunomodulatory treatment and antiretroviral therapy.
Dr. Mathew and his colleagues suggested that all patients with HIV infection presenting with optic neuritis or/and myelitis, should have their anti–aquaporin 4 antibody status checked and in all patients of NMOSD, HIV infection should be ruled out.
“Prognosis of these patients is variable; residual neurological deficits were common but treatment prevented further attacks. Increased awareness of this association will lead to earlier diagnosis, early treatment and prevention of disability,” the researchers concluded.
The authors reported that they had no conflicts.
SOURCE: Mathew T et al. Mult Scler Relat Disord. 2019 Jan;27:289-93.
HIV-associated neuromyelitis optica spectrum disorder (NMOSD) is a recently recognized entity and high index of suspicion is needed to diagnose these patients, according to Thomas Mathew, MD, and his colleagues at St. John’s Medical College Hospital, Bengaluru, India.
“NMOSD can be associated with a wide range of autoimmune diseases but clinicians rarely diagnose NMOSD in cases of HIV infection and HIV-associated NMOSD is rarely mentioned in the conventional classification of NMOSD,” they stated.
Dr. Mathew and his colleagues reported the results of a case study they made of six cases of HIV-NMOSD identified from the literature and 1 HIV-infected patient from a registry for NMOSD that they had established, which had a total of 25 patients with the condition.
There were four men and three women in the study, ranging from 8 years to 49 years of age. The duration of HIV infection in these patients ranged from newly detected to 15 years, according to the report, published in Multiple Sclerosis and Related Disorders (2019 Jan;27:289-93).
Optic neuritis followed by myelitis was the commonest presentation, occurring in five of the seven patients. Of these, six patients were assayed for anti–aquaporin 4 antibodies, which are considered a serological marker of neuromyelitis optica; three patients were positive and three were negative.
All patients received immunomodulatory treatment. Five of the seven patients had a poor recovery from acute attacks, but no patient had further relapses while on immunomodulatory treatment and antiretroviral therapy.
Dr. Mathew and his colleagues suggested that all patients with HIV infection presenting with optic neuritis or/and myelitis, should have their anti–aquaporin 4 antibody status checked and in all patients of NMOSD, HIV infection should be ruled out.
“Prognosis of these patients is variable; residual neurological deficits were common but treatment prevented further attacks. Increased awareness of this association will lead to earlier diagnosis, early treatment and prevention of disability,” the researchers concluded.
The authors reported that they had no conflicts.
SOURCE: Mathew T et al. Mult Scler Relat Disord. 2019 Jan;27:289-93.
HIV-associated neuromyelitis optica spectrum disorder (NMOSD) is a recently recognized entity and high index of suspicion is needed to diagnose these patients, according to Thomas Mathew, MD, and his colleagues at St. John’s Medical College Hospital, Bengaluru, India.
“NMOSD can be associated with a wide range of autoimmune diseases but clinicians rarely diagnose NMOSD in cases of HIV infection and HIV-associated NMOSD is rarely mentioned in the conventional classification of NMOSD,” they stated.
Dr. Mathew and his colleagues reported the results of a case study they made of six cases of HIV-NMOSD identified from the literature and 1 HIV-infected patient from a registry for NMOSD that they had established, which had a total of 25 patients with the condition.
There were four men and three women in the study, ranging from 8 years to 49 years of age. The duration of HIV infection in these patients ranged from newly detected to 15 years, according to the report, published in Multiple Sclerosis and Related Disorders (2019 Jan;27:289-93).
Optic neuritis followed by myelitis was the commonest presentation, occurring in five of the seven patients. Of these, six patients were assayed for anti–aquaporin 4 antibodies, which are considered a serological marker of neuromyelitis optica; three patients were positive and three were negative.
All patients received immunomodulatory treatment. Five of the seven patients had a poor recovery from acute attacks, but no patient had further relapses while on immunomodulatory treatment and antiretroviral therapy.
Dr. Mathew and his colleagues suggested that all patients with HIV infection presenting with optic neuritis or/and myelitis, should have their anti–aquaporin 4 antibody status checked and in all patients of NMOSD, HIV infection should be ruled out.
“Prognosis of these patients is variable; residual neurological deficits were common but treatment prevented further attacks. Increased awareness of this association will lead to earlier diagnosis, early treatment and prevention of disability,” the researchers concluded.
The authors reported that they had no conflicts.
SOURCE: Mathew T et al. Mult Scler Relat Disord. 2019 Jan;27:289-93.
FROM MULTIPLE SCLEROSIS AND RELATED DISORDERS
Parental leave for residents
Also today, exercise is important for patients with sickle cell, COPD patients are experiencing a risk in non-TB mycobacteria infections, and how to be an influencer on social media.
Amazon Alexa
Apple Podcasts
Google Podcasts
Spotify
Also today, exercise is important for patients with sickle cell, COPD patients are experiencing a risk in non-TB mycobacteria infections, and how to be an influencer on social media.
Amazon Alexa
Apple Podcasts
Google Podcasts
Spotify
Also today, exercise is important for patients with sickle cell, COPD patients are experiencing a risk in non-TB mycobacteria infections, and how to be an influencer on social media.
Amazon Alexa
Apple Podcasts
Google Podcasts
Spotify
IgA vasculitis increases risks for hypertension, chronic kidney disease
IgA vasculitis, also called Henoch-Schönlein purpura, increases risks for hypertension and chronic kidney disease (CKD), according to a retrospective study of more than 13,000 patients with IgAV.
In patients with adult-onset IgA vasculitis (IgAV), mortality risk is also increased, reported first author Alexander Tracy and his colleagues at the University of Birmingham (England).
“Long-term health outcomes of adult-onset IgAV are not well characterized,” the investigators wrote in Annals of Rheumatic Disease. “Most evidence regarding complications of IgAV in adults derives from case reports and case series; there is need for controlled epidemiological studies to address this question.”
The retrospective study compared 2,828 patients with adult-onset IgAV and 10,405 patients with childhood-onset IgAV against sex- and age-matched controls. Patients diagnosed at age 16 years or older were classified as having adult-onset disease. The investigators drew their data from The Health Improvement Network database, which includes 3.6 million active patients from more than 675 general practices in the United Kingdom. Patients in the present study were diagnosed with IgAV between 2005 and 2016. After diagnosis, participant follow-up continued until any of the following occurred: outcome event, patient left practice, death, the practice stopped contributing data, or the study ended. Primary outcomes for adult-onset patients were venous thromboembolism (VTE), ischemic heart disease, hypertension, stage 3-5 CKD, stroke/transient ischemic attack, and all-cause mortality. Primary outcomes for patients with childhood-onset disease were limited to CKD, hypertension, and VTE.
The incidence of childhood-onset IgAV was 27.22 per 100,000 person-years, whereas adult-onset disease was much less common at 2.20 per 100,000 person-years. Mean age at onset of childhood IgAV was 6.68 years. The adult-onset group had a mean age at diagnosis of 38.1 years.
Compared with controls, all patients with IgAV, regardless of onset age, had increased risks of hypertension (adult-onset adjusted hazard ratio, 1.42; P less than .001; childhood-onset aHR, 1.52; P less than .001) and CKD (adult-onset aHR, 1.54; P less than .001; childhood-onset aHR, 1.89; P = .01). Patients with adult-onset IgAV showed increased risk of death, compared with controls (aHR, 1.27; P = .006). No associations were found between IgAV and stroke/transient ischemic attack, VTE, or ischemic heart disease.
“These findings emphasize the importance of blood pressure and renal function monitoring in patients with IgAV,” the investigators concluded. “Our data also suggest that IgAV should not be considered a ‘single-hit’ disease, but that clinicians should monitor for long-term sequelae. Further research is required to clarify the cause of hypertension in patients with IgAV, and to investigate whether such patients suffer from additional long-term sequelae than that are currently unrecognized.”
The investigators reported no funding sources or conflicts or interest.
SOURCE: Tracy A et al. Ann Rheum Dis. 2018 Nov 28. doi: 10.1136/annrheumdis-2018-214142.
IgA vasculitis, also called Henoch-Schönlein purpura, increases risks for hypertension and chronic kidney disease (CKD), according to a retrospective study of more than 13,000 patients with IgAV.
In patients with adult-onset IgA vasculitis (IgAV), mortality risk is also increased, reported first author Alexander Tracy and his colleagues at the University of Birmingham (England).
“Long-term health outcomes of adult-onset IgAV are not well characterized,” the investigators wrote in Annals of Rheumatic Disease. “Most evidence regarding complications of IgAV in adults derives from case reports and case series; there is need for controlled epidemiological studies to address this question.”
The retrospective study compared 2,828 patients with adult-onset IgAV and 10,405 patients with childhood-onset IgAV against sex- and age-matched controls. Patients diagnosed at age 16 years or older were classified as having adult-onset disease. The investigators drew their data from The Health Improvement Network database, which includes 3.6 million active patients from more than 675 general practices in the United Kingdom. Patients in the present study were diagnosed with IgAV between 2005 and 2016. After diagnosis, participant follow-up continued until any of the following occurred: outcome event, patient left practice, death, the practice stopped contributing data, or the study ended. Primary outcomes for adult-onset patients were venous thromboembolism (VTE), ischemic heart disease, hypertension, stage 3-5 CKD, stroke/transient ischemic attack, and all-cause mortality. Primary outcomes for patients with childhood-onset disease were limited to CKD, hypertension, and VTE.
The incidence of childhood-onset IgAV was 27.22 per 100,000 person-years, whereas adult-onset disease was much less common at 2.20 per 100,000 person-years. Mean age at onset of childhood IgAV was 6.68 years. The adult-onset group had a mean age at diagnosis of 38.1 years.
Compared with controls, all patients with IgAV, regardless of onset age, had increased risks of hypertension (adult-onset adjusted hazard ratio, 1.42; P less than .001; childhood-onset aHR, 1.52; P less than .001) and CKD (adult-onset aHR, 1.54; P less than .001; childhood-onset aHR, 1.89; P = .01). Patients with adult-onset IgAV showed increased risk of death, compared with controls (aHR, 1.27; P = .006). No associations were found between IgAV and stroke/transient ischemic attack, VTE, or ischemic heart disease.
“These findings emphasize the importance of blood pressure and renal function monitoring in patients with IgAV,” the investigators concluded. “Our data also suggest that IgAV should not be considered a ‘single-hit’ disease, but that clinicians should monitor for long-term sequelae. Further research is required to clarify the cause of hypertension in patients with IgAV, and to investigate whether such patients suffer from additional long-term sequelae than that are currently unrecognized.”
The investigators reported no funding sources or conflicts or interest.
SOURCE: Tracy A et al. Ann Rheum Dis. 2018 Nov 28. doi: 10.1136/annrheumdis-2018-214142.
IgA vasculitis, also called Henoch-Schönlein purpura, increases risks for hypertension and chronic kidney disease (CKD), according to a retrospective study of more than 13,000 patients with IgAV.
In patients with adult-onset IgA vasculitis (IgAV), mortality risk is also increased, reported first author Alexander Tracy and his colleagues at the University of Birmingham (England).
“Long-term health outcomes of adult-onset IgAV are not well characterized,” the investigators wrote in Annals of Rheumatic Disease. “Most evidence regarding complications of IgAV in adults derives from case reports and case series; there is need for controlled epidemiological studies to address this question.”
The retrospective study compared 2,828 patients with adult-onset IgAV and 10,405 patients with childhood-onset IgAV against sex- and age-matched controls. Patients diagnosed at age 16 years or older were classified as having adult-onset disease. The investigators drew their data from The Health Improvement Network database, which includes 3.6 million active patients from more than 675 general practices in the United Kingdom. Patients in the present study were diagnosed with IgAV between 2005 and 2016. After diagnosis, participant follow-up continued until any of the following occurred: outcome event, patient left practice, death, the practice stopped contributing data, or the study ended. Primary outcomes for adult-onset patients were venous thromboembolism (VTE), ischemic heart disease, hypertension, stage 3-5 CKD, stroke/transient ischemic attack, and all-cause mortality. Primary outcomes for patients with childhood-onset disease were limited to CKD, hypertension, and VTE.
The incidence of childhood-onset IgAV was 27.22 per 100,000 person-years, whereas adult-onset disease was much less common at 2.20 per 100,000 person-years. Mean age at onset of childhood IgAV was 6.68 years. The adult-onset group had a mean age at diagnosis of 38.1 years.
Compared with controls, all patients with IgAV, regardless of onset age, had increased risks of hypertension (adult-onset adjusted hazard ratio, 1.42; P less than .001; childhood-onset aHR, 1.52; P less than .001) and CKD (adult-onset aHR, 1.54; P less than .001; childhood-onset aHR, 1.89; P = .01). Patients with adult-onset IgAV showed increased risk of death, compared with controls (aHR, 1.27; P = .006). No associations were found between IgAV and stroke/transient ischemic attack, VTE, or ischemic heart disease.
“These findings emphasize the importance of blood pressure and renal function monitoring in patients with IgAV,” the investigators concluded. “Our data also suggest that IgAV should not be considered a ‘single-hit’ disease, but that clinicians should monitor for long-term sequelae. Further research is required to clarify the cause of hypertension in patients with IgAV, and to investigate whether such patients suffer from additional long-term sequelae than that are currently unrecognized.”
The investigators reported no funding sources or conflicts or interest.
SOURCE: Tracy A et al. Ann Rheum Dis. 2018 Nov 28. doi: 10.1136/annrheumdis-2018-214142.
FROM ANNALS OF THE RHEUMATIC DISEASES
Key clinical point:
Major finding: There was significantly increased risk of stage 3-5 chronic kidney disease in patients with childhood-onset IgA vasculitis (adjusted hazard ratio, 1.89; P = .01).
Study details: A retrospective study of 2,828 patients with adult-onset IgA vasculitis and 10,405 patients with childhood-onset IgAV, compared with sex-matched and age-matched controls.
Disclosures: No funding sources or conflicts of interest were reported.
Source: Tracy A et al. Ann Rheum Dis. 2018 Nov 28. doi: 10.1136/annrheumdis-2018-214142.
What constitutes a clinically meaningful reduction in seizure frequency?
NEW ORLEANS – according to a study described at the annual meeting of the American Epilepsy Society. A reduction in seizure frequency of between 60% and 68% is associated with Clinical Global Impression of Improvement (CGI-I) ratings of “very much improved,” as assessed by caregivers and investigators.
“Further analyses from other phase III studies in Dravet syndrome and other patient populations should be performed to confirm these findings and explore other potential factors that contribute to caregiver and investigator CGI-I ratings, such as nonseizure outcomes and tolerability,” said Arnold Gammaitoni, PharmD, vice president of medical and scientific affairs at Zogenix in San Diego, and his colleagues.
A 50% reduction in seizure frequency is conventionally considered to be the cutoff for a clinically meaningful change. To develop an evidence-based definition of clinically meaningful seizure reduction, Dr. Gammaitoni and colleagues examined data from a phase III, randomized, double-blind, placebo-controlled trial of fenfluramine HCl oral solution for the adjunctive treatment of seizures associated with Dravet syndrome. The investigators took an anchor-based approach and examined the percentage change in seizure frequency, along with caregiver and investigator CGI-I ratings.
A total of 119 patients with Dravet syndrome were enrolled and randomized in equal groups to placebo, 0.2 mg/kg per day of fenfluramine HCl, or 0.8 mg/kg per day of fenfluramine HCl. After a 2-week titration period, patients entered a 12-week maintenance period. Patients in the 0.8-mg/kg per day group had a 63.9% greater reduction in seizure frequency than controls did.
After the 14-week titration and maintenance period, caregivers and investigators rated the change in participants’ clinical status from baseline, using the CGI-I scale, on which responses range from 1 (very much improved) to 7 (very much worse). The investigators considered patients with CGI-I scores of 1 or 2 (much improved) to have achieved a clinically meaningful response. A score of 3 (minimally improved) was not considered meaningful. The researchers pooled the results of the three treatment groups for this analysis. They estimated the clinically meaningful percentage change in seizure frequency using receiver operating characteristic analysis of binary CGI-I score, compared with percentage change in seizure frequency, and defined it as the cut-point for which specificity and sensitivity were equal or most similar.
Caregivers and investigators provided CGI-I assessments for 112 patients and 114 patients, respectively. The receiver operating characteristic analysis identified a 44% reduction in seizure frequency as a clinically meaningful cutoff point for caregiver and investigator assessments. Using this threshold, 75%, 46%, and 12.5% of patients in the 0.8-mg/kg per day, 0.2-mg/kg per day, and placebo groups, respectively, achieved a clinically meaningful reduction from baseline in seizure frequency in the phase III study.
“The use of external anchors is one method to define a clinically meaningful change in seizure frequency,” said Dr. Gammaitoni. “Having a defined minimum clinically important difference like this allows clinicians to assess impacts of treatments on an individual patient basis.... This is a chance for others to do similar types of analyses to confirm the findings that we have had in this first study with bigger data sets, in terms of using external anchors and data to define what a clinically meaningful change is.”
Zogenix, which is developing the fenfluramine formulation examined in this study, provided funding for this research.
SOURCE: Nabbout R et al. AES 2018, Abstract 3.202.
NEW ORLEANS – according to a study described at the annual meeting of the American Epilepsy Society. A reduction in seizure frequency of between 60% and 68% is associated with Clinical Global Impression of Improvement (CGI-I) ratings of “very much improved,” as assessed by caregivers and investigators.
“Further analyses from other phase III studies in Dravet syndrome and other patient populations should be performed to confirm these findings and explore other potential factors that contribute to caregiver and investigator CGI-I ratings, such as nonseizure outcomes and tolerability,” said Arnold Gammaitoni, PharmD, vice president of medical and scientific affairs at Zogenix in San Diego, and his colleagues.
A 50% reduction in seizure frequency is conventionally considered to be the cutoff for a clinically meaningful change. To develop an evidence-based definition of clinically meaningful seizure reduction, Dr. Gammaitoni and colleagues examined data from a phase III, randomized, double-blind, placebo-controlled trial of fenfluramine HCl oral solution for the adjunctive treatment of seizures associated with Dravet syndrome. The investigators took an anchor-based approach and examined the percentage change in seizure frequency, along with caregiver and investigator CGI-I ratings.
A total of 119 patients with Dravet syndrome were enrolled and randomized in equal groups to placebo, 0.2 mg/kg per day of fenfluramine HCl, or 0.8 mg/kg per day of fenfluramine HCl. After a 2-week titration period, patients entered a 12-week maintenance period. Patients in the 0.8-mg/kg per day group had a 63.9% greater reduction in seizure frequency than controls did.
After the 14-week titration and maintenance period, caregivers and investigators rated the change in participants’ clinical status from baseline, using the CGI-I scale, on which responses range from 1 (very much improved) to 7 (very much worse). The investigators considered patients with CGI-I scores of 1 or 2 (much improved) to have achieved a clinically meaningful response. A score of 3 (minimally improved) was not considered meaningful. The researchers pooled the results of the three treatment groups for this analysis. They estimated the clinically meaningful percentage change in seizure frequency using receiver operating characteristic analysis of binary CGI-I score, compared with percentage change in seizure frequency, and defined it as the cut-point for which specificity and sensitivity were equal or most similar.
Caregivers and investigators provided CGI-I assessments for 112 patients and 114 patients, respectively. The receiver operating characteristic analysis identified a 44% reduction in seizure frequency as a clinically meaningful cutoff point for caregiver and investigator assessments. Using this threshold, 75%, 46%, and 12.5% of patients in the 0.8-mg/kg per day, 0.2-mg/kg per day, and placebo groups, respectively, achieved a clinically meaningful reduction from baseline in seizure frequency in the phase III study.
“The use of external anchors is one method to define a clinically meaningful change in seizure frequency,” said Dr. Gammaitoni. “Having a defined minimum clinically important difference like this allows clinicians to assess impacts of treatments on an individual patient basis.... This is a chance for others to do similar types of analyses to confirm the findings that we have had in this first study with bigger data sets, in terms of using external anchors and data to define what a clinically meaningful change is.”
Zogenix, which is developing the fenfluramine formulation examined in this study, provided funding for this research.
SOURCE: Nabbout R et al. AES 2018, Abstract 3.202.
NEW ORLEANS – according to a study described at the annual meeting of the American Epilepsy Society. A reduction in seizure frequency of between 60% and 68% is associated with Clinical Global Impression of Improvement (CGI-I) ratings of “very much improved,” as assessed by caregivers and investigators.
“Further analyses from other phase III studies in Dravet syndrome and other patient populations should be performed to confirm these findings and explore other potential factors that contribute to caregiver and investigator CGI-I ratings, such as nonseizure outcomes and tolerability,” said Arnold Gammaitoni, PharmD, vice president of medical and scientific affairs at Zogenix in San Diego, and his colleagues.
A 50% reduction in seizure frequency is conventionally considered to be the cutoff for a clinically meaningful change. To develop an evidence-based definition of clinically meaningful seizure reduction, Dr. Gammaitoni and colleagues examined data from a phase III, randomized, double-blind, placebo-controlled trial of fenfluramine HCl oral solution for the adjunctive treatment of seizures associated with Dravet syndrome. The investigators took an anchor-based approach and examined the percentage change in seizure frequency, along with caregiver and investigator CGI-I ratings.
A total of 119 patients with Dravet syndrome were enrolled and randomized in equal groups to placebo, 0.2 mg/kg per day of fenfluramine HCl, or 0.8 mg/kg per day of fenfluramine HCl. After a 2-week titration period, patients entered a 12-week maintenance period. Patients in the 0.8-mg/kg per day group had a 63.9% greater reduction in seizure frequency than controls did.
After the 14-week titration and maintenance period, caregivers and investigators rated the change in participants’ clinical status from baseline, using the CGI-I scale, on which responses range from 1 (very much improved) to 7 (very much worse). The investigators considered patients with CGI-I scores of 1 or 2 (much improved) to have achieved a clinically meaningful response. A score of 3 (minimally improved) was not considered meaningful. The researchers pooled the results of the three treatment groups for this analysis. They estimated the clinically meaningful percentage change in seizure frequency using receiver operating characteristic analysis of binary CGI-I score, compared with percentage change in seizure frequency, and defined it as the cut-point for which specificity and sensitivity were equal or most similar.
Caregivers and investigators provided CGI-I assessments for 112 patients and 114 patients, respectively. The receiver operating characteristic analysis identified a 44% reduction in seizure frequency as a clinically meaningful cutoff point for caregiver and investigator assessments. Using this threshold, 75%, 46%, and 12.5% of patients in the 0.8-mg/kg per day, 0.2-mg/kg per day, and placebo groups, respectively, achieved a clinically meaningful reduction from baseline in seizure frequency in the phase III study.
“The use of external anchors is one method to define a clinically meaningful change in seizure frequency,” said Dr. Gammaitoni. “Having a defined minimum clinically important difference like this allows clinicians to assess impacts of treatments on an individual patient basis.... This is a chance for others to do similar types of analyses to confirm the findings that we have had in this first study with bigger data sets, in terms of using external anchors and data to define what a clinically meaningful change is.”
Zogenix, which is developing the fenfluramine formulation examined in this study, provided funding for this research.
SOURCE: Nabbout R et al. AES 2018, Abstract 3.202.
REPORTING FROM AES 2018
Key clinical point: Data support the convention of considering a 50% reduction in seizure frequency as the cutoff for a clinically meaningful change.
Major finding: Statistical analysis indicates that a 44% reduction in seizure frequency is clinically meaningful.
Study details: A phase III, randomized, double-blind, placebo-controlled clinical trial of fenfluramine HCl that included 119 patients.
Disclosures: Zogenix provided funding for the study.
Source: Nabbout R et al. Abstract 3.202.
Ice Pack–Induced Perniosis: A Rare and Underrecognized Association
Perniosis, or chilblain, is characterized by localized, tender, erythematous skin lesions that occur as an abnormal reaction to exposure to cold and damp conditions. Although the lesions favor the distal extremities, perniosis may present anywhere on the body. Lesions can develop within hours to days following exposure to temperature less than 10°C or damp environments with greater than 60% humidity.1 Acute cases may lead to pruritus and tenderness, whereas chronic cases may involve lesions that blister or ulcerate and can take weeks to heal. We report an unusual case of erythematous plaques arising on the buttocks of a 73-year-old woman using ice pack treatments for chronic low back pain.
Case Report
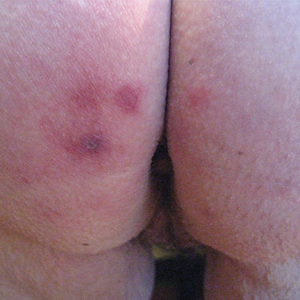
A 73-year-old woman presented with recurrent tender lesions on the buttocks of 5 years’ duration. Her medical history was remarkable for hypertension, hypothyroidism, and lumbar spinal fusion surgery 5 years prior. Physical examination revealed indurated erythematous plaques with areas of erosions on the left buttock with some involvement of the right buttock (Figure 1).
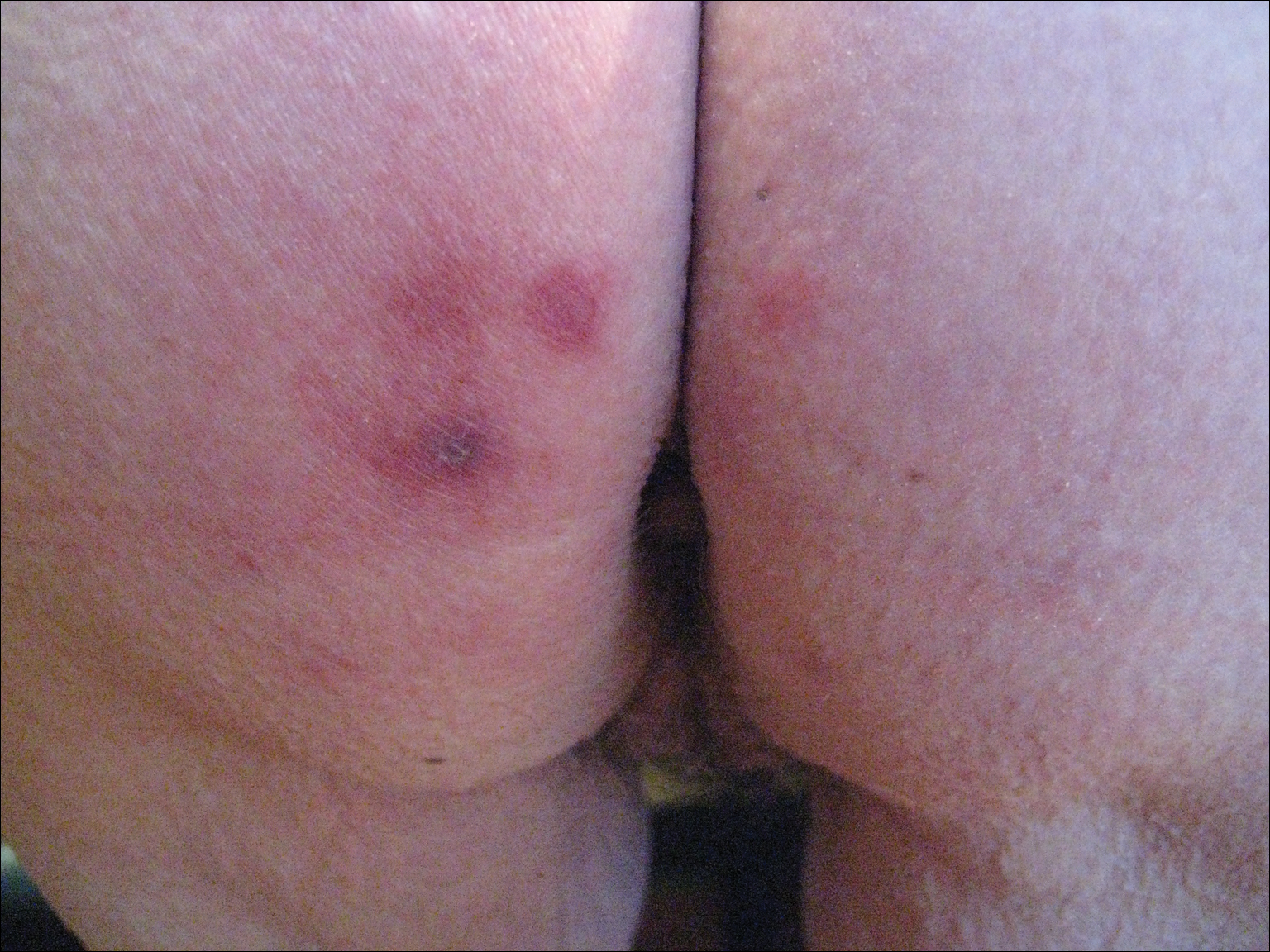
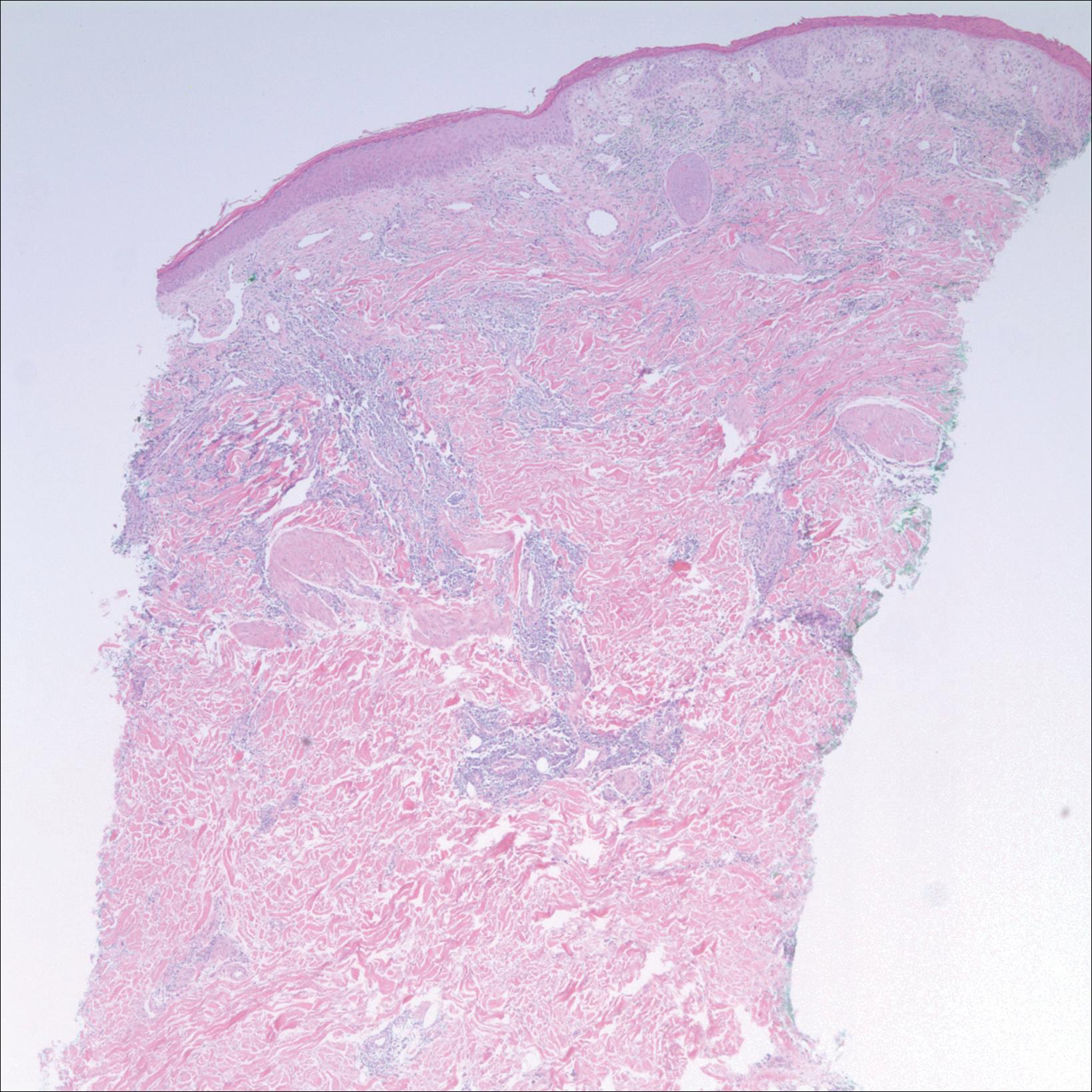
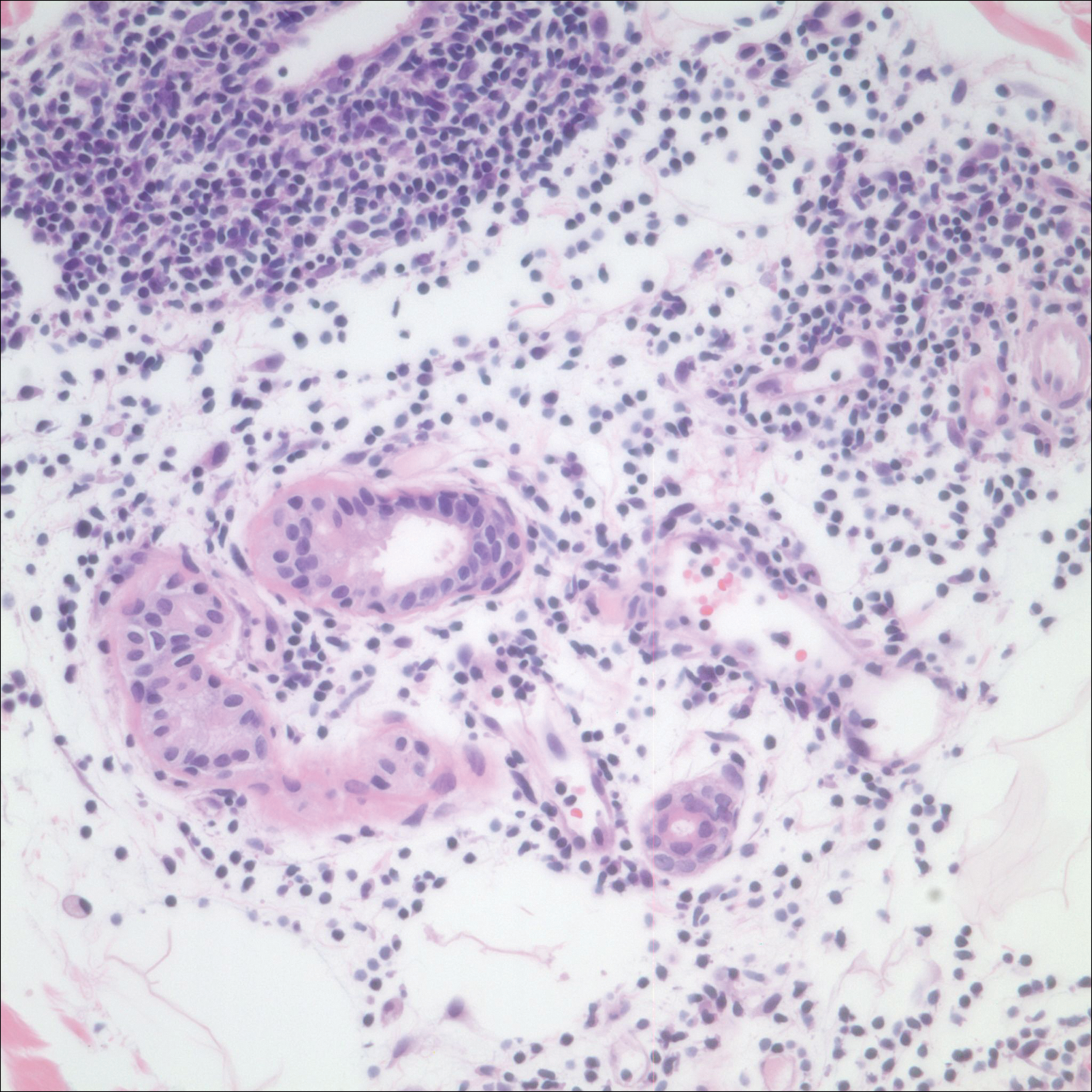
After a trial of oral valacyclovir for presumed herpes simplex infection provided no relief, a punch biopsy of the left buttock was performed, which revealed a cell-poor interface dermatitis with superficial and deep perivascular and periadnexal lymphocytic infiltrates (Figure 2). Perieccrine lymphocytes were present in a small portion of the reticular dermis (Figure 3). The patient revealed she had been sitting on ice packs for several hours daily since the lumbar spinal fusion surgery 5 years prior to alleviate chronic low back pain.
Based on the clinicopathologic correlation, a diagnosis of perniosis secondary to ice pack therapy was made. An evaluation for concomitant or underlying connective tissue disease (CTD) including a complete blood cell count with sedimentation rate, antinuclear antibodies (ANAs), serum protein electrophoresis, and serum levels of cryoglobulins and complement components was unremarkable. Our patient was treated with simple analgesia and was encouraged to avoid direct contact with ice packs for extended periods of time. Because of her low back pain, she continued to use ice packs but readjusted them sporadically and decreased frequency of use. She had complete resolution of the lesions at 6-month follow-up.
Comment
Perniosis is a self-limited condition, manifesting as erythematous plaques or nodules following exposure to cold and damp conditions. It was first reported in 1902 by Hochsinger2 as tender submental plaques occurring in children after exposure to cold weather. Since then, reports of perniosis have been described in equestrians and long-distance cyclists as well as in the context of other outdoor activities.3-5 In all cases, patients developed perniosis at sites of exposure to cold or damp conditions.
Perniosis arising in patients using ice pack therapy is a rare and recent phenomenon, with only 3 other known reported cases.6,7 In all cases, including ours, patients reported treating chronic low back pain with ice packs for more than 2 hours per day. Clinical presentations included erythematous to purpuric plaques with ulceration on the lower back or buttocks that reoccurred with subsequent use of ice packs. No concomitant CTD was reported.6
Much controversy exists as to whether idiopathic perniosis (IP) increases susceptibility to acquiring an autoimmune disease or if IP is a form of CTD that follows a more indolent course.8 In a prospective study of 33 patients with underlying IP, no patients developed lupus erythematosus (LE), with a median follow-up of 38 months.9 A study by Crowson and Magro8 revealed that 18 of 39 patients with perniotic lesions had an associated systemic disease including LE, human immunodeficiency virus, viral hepatitis, rheumatoid arthritis, cryofibrinogenemia, hypergammaglobulinemia, iritis, or Crohn disease. Of the 21 other patients who had no underlying CTD or systemic disease, 10 had a positive ANA test but no systemic symptoms; therefore, all 21 of these patients were classified as cases of IP.8
Cutaneous biopsy to distinguish between IP and autoimmune perniosis remains controversial; perniotic lesions and discoid LE share histopathologic features,9 as was evident with our case, which demonstrated overlapping findings of vacuolar change with superficial and deep perivascular and periadnexal lymphoid infiltrates. Typical features of IP include thrombosed capillaries in the papillary dermis and lymphocytic exocytosis localized to the acrosyringia, whereas secondary perniosis has superficial and deep perivascular and perieccrine lymphocytic infiltrates with vascular thrombosis in the reticular dermis. Vascular ectasia, dermal mucinosis, basement membrane zone thickening, and erythrocyte extravasation are not reliable and may be seen in both cases.8 One study revealed the only significant difference between both entities was the perieccrine distribution of lymphocytic infiltrate in cases of IP (P=.007), whereas an absence of perieccrine involvement was noted in autoimmune cases.9
Direct immunofluorescence (DIF) may help differentiate IP from autoimmune perniosis. In a prospective study by Viguier et al,9 6 of 9 patients with IP had negative DIF and 3 had slight nonspecific C3 immunoreactivity of dermal vessels. Conversely, in patients with autoimmune perniosis, positive DIF with the lupus band test was seen in 3 of 7 patients, all who had a positive ANA test9; however, positive ANA levels also were reported in patients with autoimmune perniosis but negative DIF, suggesting that DIF lacks specificity in diagnosing autoimmune perniosis.
Although histopathologic findings bear similarities to LE, there are no guidelines to suggest for or against laboratory testing for CTD in patients presenting with perniosis. Some investigators have suggested that any patient with clinical features suggestive of perniosis should undergo laboratory evaluation including a complete blood cell count and assessment for antibodies to Ro, ANA, rheumatoid factor, cryofibrinogens, and antiphospholipid antibodies.9 Serum protein electrophoresis and immunofixation electrophoresis may be done to exclude monoclonal gammopathy.
For idiopathic cases, treatment is aimed at limiting or removing cold exposure. Patients should be advised regarding the use of long-term ice pack use and the potential development of perniosis. For chronic perniosis lasting beyond several weeks, a combination of a slow taper of oral prednisone, hydroxychloroquine, and quinacrine has been successful in patients with persistent lesions despite making environmental modifications.3 Intralesional triamcinolone acetonide and nifedipine also have been effective in perniotic hand lesions.10
Conclusion
We report a rare case of perniosis on the buttocks that arose in a patient who utilized ice packs for treatment of chronic low back pain. Ice pack–induced perniosis may be an underreported entity. Histopathologic examination is nondescript, as overlapping features of perniosis and LE have been observed with no underlying CTD present. Correlation with patient history and clinical examination is paramount in diagnosis and management.
- Praminik T, Jha AK, Ghimire A. A retrospective study of cases with chilblains (perniosis) in Out Patient Department of Dermatology, Nepal Medical College and Teaching Hospital (NMCTH). Nepal Med Coll J. 2011;13:190-192.
- Hochsinger C. Acute perniosis in submental region of child [in German]. Monatsschr Kinderheilkd. 1902;1:323-327.
- Stewart CL, Adler DJ, Jacobson A, et al. Equestrian perniosis: a report of 2 cases and a review of the literature. Am J Dermatopathol. 2013;35:237-240.
- Neal AJ, Jarman AM, Bennett TG. Perniosis in a long-distance cyclist crossing Mongolia. J Travel Med. 2012;19:66-68.
- Price RD, Murdoch DR. Perniosis (chilblains) of the thigh: report of five cases including four following river crossings. High Alt Met Biol. 2001;2:535-538.
- West SA, McCalmont TH, North JP. Ice-pack dermatosis: a cold-induced dermatitis with similarities to cold panniculitis and perniosis that histopathologically resembles lupus. JAMA Dermatol. 2013;149:1314-1318.
- Haber JS, Ker KJ, Werth VP, et al. Ice‐pack dermatosis: a diagnositic pitfall for dermatopathologists that mimics lupus erythematosus. J Cutan Pathol. 2016;43:1-4.
- Crowson AN, Magro CM. Idiopathic perniosis and its mimics: a clinical and histological study of 38 cases. Hum Pathol. 1997;28:478-484.
- Viguier M, Pinguier L, Cavelier-Balloy B, et al. Clinical and histopathologic features and immunologic variables in patients with severe chilblains. a study of the relationship to lupus erythematosus. Medicine. 2001;80:180-188.
- Patra AK, Das AL, Ramadasan P. Diltiazem vs. nifedipine in chilblains: a clinical trial. Indian J Dermatol Venereol Leprol. 2003;69:209-211.
Perniosis, or chilblain, is characterized by localized, tender, erythematous skin lesions that occur as an abnormal reaction to exposure to cold and damp conditions. Although the lesions favor the distal extremities, perniosis may present anywhere on the body. Lesions can develop within hours to days following exposure to temperature less than 10°C or damp environments with greater than 60% humidity.1 Acute cases may lead to pruritus and tenderness, whereas chronic cases may involve lesions that blister or ulcerate and can take weeks to heal. We report an unusual case of erythematous plaques arising on the buttocks of a 73-year-old woman using ice pack treatments for chronic low back pain.
Case Report
A 73-year-old woman presented with recurrent tender lesions on the buttocks of 5 years’ duration. Her medical history was remarkable for hypertension, hypothyroidism, and lumbar spinal fusion surgery 5 years prior. Physical examination revealed indurated erythematous plaques with areas of erosions on the left buttock with some involvement of the right buttock (Figure 1).

After a trial of oral valacyclovir for presumed herpes simplex infection provided no relief, a punch biopsy of the left buttock was performed, which revealed a cell-poor interface dermatitis with superficial and deep perivascular and periadnexal lymphocytic infiltrates (Figure 2). Perieccrine lymphocytes were present in a small portion of the reticular dermis (Figure 3). The patient revealed she had been sitting on ice packs for several hours daily since the lumbar spinal fusion surgery 5 years prior to alleviate chronic low back pain.


Based on the clinicopathologic correlation, a diagnosis of perniosis secondary to ice pack therapy was made. An evaluation for concomitant or underlying connective tissue disease (CTD) including a complete blood cell count with sedimentation rate, antinuclear antibodies (ANAs), serum protein electrophoresis, and serum levels of cryoglobulins and complement components was unremarkable. Our patient was treated with simple analgesia and was encouraged to avoid direct contact with ice packs for extended periods of time. Because of her low back pain, she continued to use ice packs but readjusted them sporadically and decreased frequency of use. She had complete resolution of the lesions at 6-month follow-up.
Comment
Perniosis is a self-limited condition, manifesting as erythematous plaques or nodules following exposure to cold and damp conditions. It was first reported in 1902 by Hochsinger2 as tender submental plaques occurring in children after exposure to cold weather. Since then, reports of perniosis have been described in equestrians and long-distance cyclists as well as in the context of other outdoor activities.3-5 In all cases, patients developed perniosis at sites of exposure to cold or damp conditions.
Perniosis arising in patients using ice pack therapy is a rare and recent phenomenon, with only 3 other known reported cases.6,7 In all cases, including ours, patients reported treating chronic low back pain with ice packs for more than 2 hours per day. Clinical presentations included erythematous to purpuric plaques with ulceration on the lower back or buttocks that reoccurred with subsequent use of ice packs. No concomitant CTD was reported.6
Much controversy exists as to whether idiopathic perniosis (IP) increases susceptibility to acquiring an autoimmune disease or if IP is a form of CTD that follows a more indolent course.8 In a prospective study of 33 patients with underlying IP, no patients developed lupus erythematosus (LE), with a median follow-up of 38 months.9 A study by Crowson and Magro8 revealed that 18 of 39 patients with perniotic lesions had an associated systemic disease including LE, human immunodeficiency virus, viral hepatitis, rheumatoid arthritis, cryofibrinogenemia, hypergammaglobulinemia, iritis, or Crohn disease. Of the 21 other patients who had no underlying CTD or systemic disease, 10 had a positive ANA test but no systemic symptoms; therefore, all 21 of these patients were classified as cases of IP.8
Cutaneous biopsy to distinguish between IP and autoimmune perniosis remains controversial; perniotic lesions and discoid LE share histopathologic features,9 as was evident with our case, which demonstrated overlapping findings of vacuolar change with superficial and deep perivascular and periadnexal lymphoid infiltrates. Typical features of IP include thrombosed capillaries in the papillary dermis and lymphocytic exocytosis localized to the acrosyringia, whereas secondary perniosis has superficial and deep perivascular and perieccrine lymphocytic infiltrates with vascular thrombosis in the reticular dermis. Vascular ectasia, dermal mucinosis, basement membrane zone thickening, and erythrocyte extravasation are not reliable and may be seen in both cases.8 One study revealed the only significant difference between both entities was the perieccrine distribution of lymphocytic infiltrate in cases of IP (P=.007), whereas an absence of perieccrine involvement was noted in autoimmune cases.9
Direct immunofluorescence (DIF) may help differentiate IP from autoimmune perniosis. In a prospective study by Viguier et al,9 6 of 9 patients with IP had negative DIF and 3 had slight nonspecific C3 immunoreactivity of dermal vessels. Conversely, in patients with autoimmune perniosis, positive DIF with the lupus band test was seen in 3 of 7 patients, all who had a positive ANA test9; however, positive ANA levels also were reported in patients with autoimmune perniosis but negative DIF, suggesting that DIF lacks specificity in diagnosing autoimmune perniosis.
Although histopathologic findings bear similarities to LE, there are no guidelines to suggest for or against laboratory testing for CTD in patients presenting with perniosis. Some investigators have suggested that any patient with clinical features suggestive of perniosis should undergo laboratory evaluation including a complete blood cell count and assessment for antibodies to Ro, ANA, rheumatoid factor, cryofibrinogens, and antiphospholipid antibodies.9 Serum protein electrophoresis and immunofixation electrophoresis may be done to exclude monoclonal gammopathy.
For idiopathic cases, treatment is aimed at limiting or removing cold exposure. Patients should be advised regarding the use of long-term ice pack use and the potential development of perniosis. For chronic perniosis lasting beyond several weeks, a combination of a slow taper of oral prednisone, hydroxychloroquine, and quinacrine has been successful in patients with persistent lesions despite making environmental modifications.3 Intralesional triamcinolone acetonide and nifedipine also have been effective in perniotic hand lesions.10
Conclusion
We report a rare case of perniosis on the buttocks that arose in a patient who utilized ice packs for treatment of chronic low back pain. Ice pack–induced perniosis may be an underreported entity. Histopathologic examination is nondescript, as overlapping features of perniosis and LE have been observed with no underlying CTD present. Correlation with patient history and clinical examination is paramount in diagnosis and management.
Perniosis, or chilblain, is characterized by localized, tender, erythematous skin lesions that occur as an abnormal reaction to exposure to cold and damp conditions. Although the lesions favor the distal extremities, perniosis may present anywhere on the body. Lesions can develop within hours to days following exposure to temperature less than 10°C or damp environments with greater than 60% humidity.1 Acute cases may lead to pruritus and tenderness, whereas chronic cases may involve lesions that blister or ulcerate and can take weeks to heal. We report an unusual case of erythematous plaques arising on the buttocks of a 73-year-old woman using ice pack treatments for chronic low back pain.
Case Report
A 73-year-old woman presented with recurrent tender lesions on the buttocks of 5 years’ duration. Her medical history was remarkable for hypertension, hypothyroidism, and lumbar spinal fusion surgery 5 years prior. Physical examination revealed indurated erythematous plaques with areas of erosions on the left buttock with some involvement of the right buttock (Figure 1).

After a trial of oral valacyclovir for presumed herpes simplex infection provided no relief, a punch biopsy of the left buttock was performed, which revealed a cell-poor interface dermatitis with superficial and deep perivascular and periadnexal lymphocytic infiltrates (Figure 2). Perieccrine lymphocytes were present in a small portion of the reticular dermis (Figure 3). The patient revealed she had been sitting on ice packs for several hours daily since the lumbar spinal fusion surgery 5 years prior to alleviate chronic low back pain.


Based on the clinicopathologic correlation, a diagnosis of perniosis secondary to ice pack therapy was made. An evaluation for concomitant or underlying connective tissue disease (CTD) including a complete blood cell count with sedimentation rate, antinuclear antibodies (ANAs), serum protein electrophoresis, and serum levels of cryoglobulins and complement components was unremarkable. Our patient was treated with simple analgesia and was encouraged to avoid direct contact with ice packs for extended periods of time. Because of her low back pain, she continued to use ice packs but readjusted them sporadically and decreased frequency of use. She had complete resolution of the lesions at 6-month follow-up.
Comment
Perniosis is a self-limited condition, manifesting as erythematous plaques or nodules following exposure to cold and damp conditions. It was first reported in 1902 by Hochsinger2 as tender submental plaques occurring in children after exposure to cold weather. Since then, reports of perniosis have been described in equestrians and long-distance cyclists as well as in the context of other outdoor activities.3-5 In all cases, patients developed perniosis at sites of exposure to cold or damp conditions.
Perniosis arising in patients using ice pack therapy is a rare and recent phenomenon, with only 3 other known reported cases.6,7 In all cases, including ours, patients reported treating chronic low back pain with ice packs for more than 2 hours per day. Clinical presentations included erythematous to purpuric plaques with ulceration on the lower back or buttocks that reoccurred with subsequent use of ice packs. No concomitant CTD was reported.6
Much controversy exists as to whether idiopathic perniosis (IP) increases susceptibility to acquiring an autoimmune disease or if IP is a form of CTD that follows a more indolent course.8 In a prospective study of 33 patients with underlying IP, no patients developed lupus erythematosus (LE), with a median follow-up of 38 months.9 A study by Crowson and Magro8 revealed that 18 of 39 patients with perniotic lesions had an associated systemic disease including LE, human immunodeficiency virus, viral hepatitis, rheumatoid arthritis, cryofibrinogenemia, hypergammaglobulinemia, iritis, or Crohn disease. Of the 21 other patients who had no underlying CTD or systemic disease, 10 had a positive ANA test but no systemic symptoms; therefore, all 21 of these patients were classified as cases of IP.8
Cutaneous biopsy to distinguish between IP and autoimmune perniosis remains controversial; perniotic lesions and discoid LE share histopathologic features,9 as was evident with our case, which demonstrated overlapping findings of vacuolar change with superficial and deep perivascular and periadnexal lymphoid infiltrates. Typical features of IP include thrombosed capillaries in the papillary dermis and lymphocytic exocytosis localized to the acrosyringia, whereas secondary perniosis has superficial and deep perivascular and perieccrine lymphocytic infiltrates with vascular thrombosis in the reticular dermis. Vascular ectasia, dermal mucinosis, basement membrane zone thickening, and erythrocyte extravasation are not reliable and may be seen in both cases.8 One study revealed the only significant difference between both entities was the perieccrine distribution of lymphocytic infiltrate in cases of IP (P=.007), whereas an absence of perieccrine involvement was noted in autoimmune cases.9
Direct immunofluorescence (DIF) may help differentiate IP from autoimmune perniosis. In a prospective study by Viguier et al,9 6 of 9 patients with IP had negative DIF and 3 had slight nonspecific C3 immunoreactivity of dermal vessels. Conversely, in patients with autoimmune perniosis, positive DIF with the lupus band test was seen in 3 of 7 patients, all who had a positive ANA test9; however, positive ANA levels also were reported in patients with autoimmune perniosis but negative DIF, suggesting that DIF lacks specificity in diagnosing autoimmune perniosis.
Although histopathologic findings bear similarities to LE, there are no guidelines to suggest for or against laboratory testing for CTD in patients presenting with perniosis. Some investigators have suggested that any patient with clinical features suggestive of perniosis should undergo laboratory evaluation including a complete blood cell count and assessment for antibodies to Ro, ANA, rheumatoid factor, cryofibrinogens, and antiphospholipid antibodies.9 Serum protein electrophoresis and immunofixation electrophoresis may be done to exclude monoclonal gammopathy.
For idiopathic cases, treatment is aimed at limiting or removing cold exposure. Patients should be advised regarding the use of long-term ice pack use and the potential development of perniosis. For chronic perniosis lasting beyond several weeks, a combination of a slow taper of oral prednisone, hydroxychloroquine, and quinacrine has been successful in patients with persistent lesions despite making environmental modifications.3 Intralesional triamcinolone acetonide and nifedipine also have been effective in perniotic hand lesions.10
Conclusion
We report a rare case of perniosis on the buttocks that arose in a patient who utilized ice packs for treatment of chronic low back pain. Ice pack–induced perniosis may be an underreported entity. Histopathologic examination is nondescript, as overlapping features of perniosis and LE have been observed with no underlying CTD present. Correlation with patient history and clinical examination is paramount in diagnosis and management.
- Praminik T, Jha AK, Ghimire A. A retrospective study of cases with chilblains (perniosis) in Out Patient Department of Dermatology, Nepal Medical College and Teaching Hospital (NMCTH). Nepal Med Coll J. 2011;13:190-192.
- Hochsinger C. Acute perniosis in submental region of child [in German]. Monatsschr Kinderheilkd. 1902;1:323-327.
- Stewart CL, Adler DJ, Jacobson A, et al. Equestrian perniosis: a report of 2 cases and a review of the literature. Am J Dermatopathol. 2013;35:237-240.
- Neal AJ, Jarman AM, Bennett TG. Perniosis in a long-distance cyclist crossing Mongolia. J Travel Med. 2012;19:66-68.
- Price RD, Murdoch DR. Perniosis (chilblains) of the thigh: report of five cases including four following river crossings. High Alt Met Biol. 2001;2:535-538.
- West SA, McCalmont TH, North JP. Ice-pack dermatosis: a cold-induced dermatitis with similarities to cold panniculitis and perniosis that histopathologically resembles lupus. JAMA Dermatol. 2013;149:1314-1318.
- Haber JS, Ker KJ, Werth VP, et al. Ice‐pack dermatosis: a diagnositic pitfall for dermatopathologists that mimics lupus erythematosus. J Cutan Pathol. 2016;43:1-4.
- Crowson AN, Magro CM. Idiopathic perniosis and its mimics: a clinical and histological study of 38 cases. Hum Pathol. 1997;28:478-484.
- Viguier M, Pinguier L, Cavelier-Balloy B, et al. Clinical and histopathologic features and immunologic variables in patients with severe chilblains. a study of the relationship to lupus erythematosus. Medicine. 2001;80:180-188.
- Patra AK, Das AL, Ramadasan P. Diltiazem vs. nifedipine in chilblains: a clinical trial. Indian J Dermatol Venereol Leprol. 2003;69:209-211.
- Praminik T, Jha AK, Ghimire A. A retrospective study of cases with chilblains (perniosis) in Out Patient Department of Dermatology, Nepal Medical College and Teaching Hospital (NMCTH). Nepal Med Coll J. 2011;13:190-192.
- Hochsinger C. Acute perniosis in submental region of child [in German]. Monatsschr Kinderheilkd. 1902;1:323-327.
- Stewart CL, Adler DJ, Jacobson A, et al. Equestrian perniosis: a report of 2 cases and a review of the literature. Am J Dermatopathol. 2013;35:237-240.
- Neal AJ, Jarman AM, Bennett TG. Perniosis in a long-distance cyclist crossing Mongolia. J Travel Med. 2012;19:66-68.
- Price RD, Murdoch DR. Perniosis (chilblains) of the thigh: report of five cases including four following river crossings. High Alt Met Biol. 2001;2:535-538.
- West SA, McCalmont TH, North JP. Ice-pack dermatosis: a cold-induced dermatitis with similarities to cold panniculitis and perniosis that histopathologically resembles lupus. JAMA Dermatol. 2013;149:1314-1318.
- Haber JS, Ker KJ, Werth VP, et al. Ice‐pack dermatosis: a diagnositic pitfall for dermatopathologists that mimics lupus erythematosus. J Cutan Pathol. 2016;43:1-4.
- Crowson AN, Magro CM. Idiopathic perniosis and its mimics: a clinical and histological study of 38 cases. Hum Pathol. 1997;28:478-484.
- Viguier M, Pinguier L, Cavelier-Balloy B, et al. Clinical and histopathologic features and immunologic variables in patients with severe chilblains. a study of the relationship to lupus erythematosus. Medicine. 2001;80:180-188.
- Patra AK, Das AL, Ramadasan P. Diltiazem vs. nifedipine in chilblains: a clinical trial. Indian J Dermatol Venereol Leprol. 2003;69:209-211.
Practice Points
- Ice pack-induced perniosis is a rare condition that can occur in patients using long-term ice pack therapy.
- This entity histopathologically mimics cutaneous lupus erythematosus and can present a diagnostic challenge.
- A thorough clinical history and awareness of this diagnosis is essential for diagnostic accuracy.