User login
Exploring the Utility of Artificial Intelligence During COVID-19 in Dermatology Practice
With the need to adapt to the given challenges associated with COVID-19, artificial intelligence (AI) serves as a potential tool in providing access to medical-based diagnosis in a novel way. Artificial intelligence is defined as intelligence harnessed by machines that have the ability to perform what is called cognitive thinking and to mimic the problem-solving abilities of the human mind. Virtual AI in dermatology entails neural network–based guidance that includes developing algorithms to detect skin pathology through photographs.1 To use AI in dermatology, recognition of visual patterns must be established to give diagnoses. These neural networks have been used to classify skin diseases, including cancer, actinic keratosis, and warts.2
AI for Skin Cancer
The use of AI to classify melanoma and nonmelanoma skin cancer has been studied extensively, including the following 2 research projects.
Convolutional Neural Network
In 2017, Stanford University published a study in which a deep-learning algorithm known as a convolutional neural network was used to classify skin lesions.3 The network was trained using a dataset of 129,450 clinical images of 2032 diseases. Its performance was compared to that of 21 board-certified dermatologists on biopsy-proven clinical images with 2 classifications of cases: (1) keratinocyte carcinoma as opposed to benign seborrheic keratosis and (2) malignant melanoma as opposed to benign nevi—the first representing the most common skin cancers, and the second, the deadliest skin cancers. The study showed that the machine could accurately identify and classify skin cancers compared to the work of board-certified dermatologists. The study did not include demographic information, which limits its external validity.3
Dermoscopic Image Classification
A 2019 study by Brinker and colleagues4 showed the superiority of automated dermoscopic melanoma image classifications compared to the work of board-certified dermatologists. For the study, 804 biopsy-proven images of melanoma and nevi (1:1 ratio) were randomly presented to dermatologists for their evaluation and recommended treatment (yielding 19,296 recommendations). The dermatologists classified the lesions with a sensitivity of 67.2% and specificity of 62.2%; the trained convolutional neural network attained both higher sensitivity (82.3%) and higher specificity (77.9%).4
Smartphone Diagnosis of Melanoma
An application of AI has been to use smartphone apps for the diagnosis of melanoma. The most utilized and novel algorithm-based smartphone app that assesses skin lesions for malignancy characteristics is SkinVision. With a simple download from Apple’s App Store, this technology allows a person to check their skin spots by taking a photograph and receiving algorithmic risk-assessment feedback. This inexpensive software ($51.78 a year) also allows a patient’s physician to assess the photograph and then validate their assessment by comparing it with the algorithmic analysis that the program provides.5
A review of SkinVision conducted by Thissen and colleagues6 found that, in a hypothetical population of 1000 adults of whom 3% actually had melanoma, 4 of those 30 people would not have been flagged as at “high risk” by SkinVision. There also was a high false-positive rate with the app, with more than 200 people flagged as at high risk. The analysis pegged SkinVision as having a sensitivity of 88% and specificity of 79%.6
In summary, systematic review of diagnostic accuracy has shown that, although there is accuracy in AI analyses, it should be used only as a guide for health care advice due to variability in algorithm performance.7
Utility of AI in Telehealth
Artificial intelligence algorithms could be created to ensure telehealth image accuracy, stratify risk, and track patient progress. With teledermatology visits on the rise during the COVID-19 pandemic, AI algorithms could ensure that photographs of appropriate quality are taken. Also, patients could be organized by risk factors with such algorithms, allowing physicians to save time on triage and stratification. Algorithms also could be used to track a telehealth patient’s treatment and progress.8
Furthermore, there is a need for an algorithm that has the ability to detect, quantify, and monitor changes in dermatologic conditions using images that patients have uploaded. This capability will lead to creation of a standardized quantification scale that will allow physicians to virtually track the progression of visible skin pathologies.
Hazards of Racial Bias in AI
Artificial intelligence is limited by racial disparity bias seen in computerized medicine. For years, the majority of dermatology research, especially in skin cancer, has been conducted on fairer-skinned populations. This bias has existed at the expense of darker-skinned patients, whose skin conditions and symptoms present differently,9 and reflects directly in available data sets that can be used to develop AI algorithms. Because these data are inadequate to the task, AI might misdiagnose skin cancer in people of color or miss an existing condition entirely.10 Consequently, the higher rate of skin cancer mortality that is reported in people of color is likely to persist with the rise of AI in dermatology.11 A more representative database of imaged skin lesions needs to be utilized to create a diversely representative and applicable data set for AI algorithms.12
Benefits of Conversational Agents
Another method by which AI could be incorporated into dermatology is through what is known as a conversational agent (CA)—AI software that engages in a dialogue with users by interpreting their voice and replying to them through text, image, or voice.13 Conversational agents facilitate remote patient management, allow clinicians to focus on other functions, and aid in data collection.14 A 2014 study showed that patients were significantly more likely to disclose history and emotions when informed they were interacting with a CA than with a human clinician (P=.007).15 Such benefits could be invaluable in dermatology, where emotions and patient perceptions of skin conditions play into the treatment process.
However, some evidence showed that CAs cannot respond to patients’ statements in all circumstances.16 It also is unclear how well CAs recognize nuanced statements that might signal potential harm. This fits into the greater theme of a major problem with AI: the lack of a reliable response in all circumstances.13
Final Thoughts
The practical implementations of AI in dermatology are still being explored. Given the uncertainty surrounding the COVID-19 pandemic and the future of patient care, AI might serve as an important asset in assisting with the diagnosis and treatment of dermatologic conditions, physician productivity, and patient monitoring.
- Amisha, Malik P, Pathania M, et al. Overview of artificial intelligence in medicine. J Family Med Prim Care. 2019;8:2328-2331. doi:10.4103/jfmpc.jfmpc_440_19
- Han SS, Kim MS, Lim W, et al. Classification of the clinical images for benign and malignant cutaneous tumors using a deep learning algorithm. J Invest Dermatol. 2018;138:1529-1538. doi:10.1016/j.jid.2018.01.028
- Esteva A, Kuprel B, Novoa RA, et al. Dermatologist-level classification of skin cancer with deep neural networks. Nature. 2017;542:115-118. doi:10.1038/nature21056
- Brinker TJ, Hekler A, Enk AH, et al. Deep neural networks are superior to dermatologists in melanoma image classification. Eur J Cancer. 2019;119:11-17. doi:10.1016/j.ejca.2019.05.023
- Regulated medical device for detecting skin cancer. SkinVision website. Accessed July 23, 2021. https://www.skinvision.com/hcp/
- Thissen M, Udrea A, Hacking M, et al. mHealth app for risk assessment of pigmented and nonpigmented skin lesions—a study on sensitivity and specificity in detecting malignancy. Telemed J E Health. 2017;23:948-954. doi:10.1089/tmj.2016.0259
- Freeman K, Dinnes J, Chuchu N, et al. Algorithm based smartphone apps to assess risk of skin cancer in adults: systematic review of diagnostic accuracy studies. BMJ. 2020;368:m127. doi:10.1136/bmj.m127
- Puri P, Comfere N, Pittelkow MR, et al. COVID-19: an opportunity to build dermatology’s digital future. Dermatol Ther. 2020;33:e14149. doi:10.1111/dth.14149
- Buster KJ, Stevens EI, Elmets CA. Dermatologic health disparities. Dermatol Clin. 2012;30:53-59,viii. doi:10.1016/j.det.2011.08.002
- Adamson AS, Smith A. Machine learning and health care disparities in dermatology. JAMA Dermatol. 2018;154:1247-1248. doi:10.1001/jamadermatol.2018.2348
- Agbai ON, Buster K, Sanchez M, et al. Skin cancer and photoprotection in people of color: a review and recommendations for physicians and the public. J Am Acad Dermatol. 2014;70:748-762. doi:S0190-9622(13)01296-6
- Alabdulkareem A. Artificial intelligence and dermatologists: friends or foes? J Dermatol Dermatolog Surg. 2019;23:57-60. doi:10.4103/jdds.jdds_19_19
- McGreevey JD 3rd, Hanson CW 3rd, Koppel R. Clinical, legal, and ethical aspects of artificial intelligence-assisted conversational agents in health care. JAMA. 2020;324:552-553. doi:10.1001/jama.2020.2724
- Piau A, Crissey R, Brechemier D, et al. A smartphone chatbot application to optimize monitoring of older patients with cancer. Int J Med Inform. 2019;128:18-23. doi:10.1016/j.ijmedinf.2019.05.013
- Lucas GM, Gratch J, King A, et al. It’s only a computer: virtual humans increase willingness to disclose. Comput Human Behav. 2014;37:94-100. https://doi.org/10.1016/j.chb.2014.04.043
- Miner AS, Milstein A, Schueller S, et al. Smartphone-based conversational agents and responses to questions about mental health, interpersonal violence, and physical health. JAMA Intern Med. 2016;176:619-625. doi:10.1001/jamainternmed.2016.0400
With the need to adapt to the given challenges associated with COVID-19, artificial intelligence (AI) serves as a potential tool in providing access to medical-based diagnosis in a novel way. Artificial intelligence is defined as intelligence harnessed by machines that have the ability to perform what is called cognitive thinking and to mimic the problem-solving abilities of the human mind. Virtual AI in dermatology entails neural network–based guidance that includes developing algorithms to detect skin pathology through photographs.1 To use AI in dermatology, recognition of visual patterns must be established to give diagnoses. These neural networks have been used to classify skin diseases, including cancer, actinic keratosis, and warts.2
AI for Skin Cancer
The use of AI to classify melanoma and nonmelanoma skin cancer has been studied extensively, including the following 2 research projects.
Convolutional Neural Network
In 2017, Stanford University published a study in which a deep-learning algorithm known as a convolutional neural network was used to classify skin lesions.3 The network was trained using a dataset of 129,450 clinical images of 2032 diseases. Its performance was compared to that of 21 board-certified dermatologists on biopsy-proven clinical images with 2 classifications of cases: (1) keratinocyte carcinoma as opposed to benign seborrheic keratosis and (2) malignant melanoma as opposed to benign nevi—the first representing the most common skin cancers, and the second, the deadliest skin cancers. The study showed that the machine could accurately identify and classify skin cancers compared to the work of board-certified dermatologists. The study did not include demographic information, which limits its external validity.3
Dermoscopic Image Classification
A 2019 study by Brinker and colleagues4 showed the superiority of automated dermoscopic melanoma image classifications compared to the work of board-certified dermatologists. For the study, 804 biopsy-proven images of melanoma and nevi (1:1 ratio) were randomly presented to dermatologists for their evaluation and recommended treatment (yielding 19,296 recommendations). The dermatologists classified the lesions with a sensitivity of 67.2% and specificity of 62.2%; the trained convolutional neural network attained both higher sensitivity (82.3%) and higher specificity (77.9%).4
Smartphone Diagnosis of Melanoma
An application of AI has been to use smartphone apps for the diagnosis of melanoma. The most utilized and novel algorithm-based smartphone app that assesses skin lesions for malignancy characteristics is SkinVision. With a simple download from Apple’s App Store, this technology allows a person to check their skin spots by taking a photograph and receiving algorithmic risk-assessment feedback. This inexpensive software ($51.78 a year) also allows a patient’s physician to assess the photograph and then validate their assessment by comparing it with the algorithmic analysis that the program provides.5
A review of SkinVision conducted by Thissen and colleagues6 found that, in a hypothetical population of 1000 adults of whom 3% actually had melanoma, 4 of those 30 people would not have been flagged as at “high risk” by SkinVision. There also was a high false-positive rate with the app, with more than 200 people flagged as at high risk. The analysis pegged SkinVision as having a sensitivity of 88% and specificity of 79%.6
In summary, systematic review of diagnostic accuracy has shown that, although there is accuracy in AI analyses, it should be used only as a guide for health care advice due to variability in algorithm performance.7
Utility of AI in Telehealth
Artificial intelligence algorithms could be created to ensure telehealth image accuracy, stratify risk, and track patient progress. With teledermatology visits on the rise during the COVID-19 pandemic, AI algorithms could ensure that photographs of appropriate quality are taken. Also, patients could be organized by risk factors with such algorithms, allowing physicians to save time on triage and stratification. Algorithms also could be used to track a telehealth patient’s treatment and progress.8
Furthermore, there is a need for an algorithm that has the ability to detect, quantify, and monitor changes in dermatologic conditions using images that patients have uploaded. This capability will lead to creation of a standardized quantification scale that will allow physicians to virtually track the progression of visible skin pathologies.
Hazards of Racial Bias in AI
Artificial intelligence is limited by racial disparity bias seen in computerized medicine. For years, the majority of dermatology research, especially in skin cancer, has been conducted on fairer-skinned populations. This bias has existed at the expense of darker-skinned patients, whose skin conditions and symptoms present differently,9 and reflects directly in available data sets that can be used to develop AI algorithms. Because these data are inadequate to the task, AI might misdiagnose skin cancer in people of color or miss an existing condition entirely.10 Consequently, the higher rate of skin cancer mortality that is reported in people of color is likely to persist with the rise of AI in dermatology.11 A more representative database of imaged skin lesions needs to be utilized to create a diversely representative and applicable data set for AI algorithms.12
Benefits of Conversational Agents
Another method by which AI could be incorporated into dermatology is through what is known as a conversational agent (CA)—AI software that engages in a dialogue with users by interpreting their voice and replying to them through text, image, or voice.13 Conversational agents facilitate remote patient management, allow clinicians to focus on other functions, and aid in data collection.14 A 2014 study showed that patients were significantly more likely to disclose history and emotions when informed they were interacting with a CA than with a human clinician (P=.007).15 Such benefits could be invaluable in dermatology, where emotions and patient perceptions of skin conditions play into the treatment process.
However, some evidence showed that CAs cannot respond to patients’ statements in all circumstances.16 It also is unclear how well CAs recognize nuanced statements that might signal potential harm. This fits into the greater theme of a major problem with AI: the lack of a reliable response in all circumstances.13
Final Thoughts
The practical implementations of AI in dermatology are still being explored. Given the uncertainty surrounding the COVID-19 pandemic and the future of patient care, AI might serve as an important asset in assisting with the diagnosis and treatment of dermatologic conditions, physician productivity, and patient monitoring.
With the need to adapt to the given challenges associated with COVID-19, artificial intelligence (AI) serves as a potential tool in providing access to medical-based diagnosis in a novel way. Artificial intelligence is defined as intelligence harnessed by machines that have the ability to perform what is called cognitive thinking and to mimic the problem-solving abilities of the human mind. Virtual AI in dermatology entails neural network–based guidance that includes developing algorithms to detect skin pathology through photographs.1 To use AI in dermatology, recognition of visual patterns must be established to give diagnoses. These neural networks have been used to classify skin diseases, including cancer, actinic keratosis, and warts.2
AI for Skin Cancer
The use of AI to classify melanoma and nonmelanoma skin cancer has been studied extensively, including the following 2 research projects.
Convolutional Neural Network
In 2017, Stanford University published a study in which a deep-learning algorithm known as a convolutional neural network was used to classify skin lesions.3 The network was trained using a dataset of 129,450 clinical images of 2032 diseases. Its performance was compared to that of 21 board-certified dermatologists on biopsy-proven clinical images with 2 classifications of cases: (1) keratinocyte carcinoma as opposed to benign seborrheic keratosis and (2) malignant melanoma as opposed to benign nevi—the first representing the most common skin cancers, and the second, the deadliest skin cancers. The study showed that the machine could accurately identify and classify skin cancers compared to the work of board-certified dermatologists. The study did not include demographic information, which limits its external validity.3
Dermoscopic Image Classification
A 2019 study by Brinker and colleagues4 showed the superiority of automated dermoscopic melanoma image classifications compared to the work of board-certified dermatologists. For the study, 804 biopsy-proven images of melanoma and nevi (1:1 ratio) were randomly presented to dermatologists for their evaluation and recommended treatment (yielding 19,296 recommendations). The dermatologists classified the lesions with a sensitivity of 67.2% and specificity of 62.2%; the trained convolutional neural network attained both higher sensitivity (82.3%) and higher specificity (77.9%).4
Smartphone Diagnosis of Melanoma
An application of AI has been to use smartphone apps for the diagnosis of melanoma. The most utilized and novel algorithm-based smartphone app that assesses skin lesions for malignancy characteristics is SkinVision. With a simple download from Apple’s App Store, this technology allows a person to check their skin spots by taking a photograph and receiving algorithmic risk-assessment feedback. This inexpensive software ($51.78 a year) also allows a patient’s physician to assess the photograph and then validate their assessment by comparing it with the algorithmic analysis that the program provides.5
A review of SkinVision conducted by Thissen and colleagues6 found that, in a hypothetical population of 1000 adults of whom 3% actually had melanoma, 4 of those 30 people would not have been flagged as at “high risk” by SkinVision. There also was a high false-positive rate with the app, with more than 200 people flagged as at high risk. The analysis pegged SkinVision as having a sensitivity of 88% and specificity of 79%.6
In summary, systematic review of diagnostic accuracy has shown that, although there is accuracy in AI analyses, it should be used only as a guide for health care advice due to variability in algorithm performance.7
Utility of AI in Telehealth
Artificial intelligence algorithms could be created to ensure telehealth image accuracy, stratify risk, and track patient progress. With teledermatology visits on the rise during the COVID-19 pandemic, AI algorithms could ensure that photographs of appropriate quality are taken. Also, patients could be organized by risk factors with such algorithms, allowing physicians to save time on triage and stratification. Algorithms also could be used to track a telehealth patient’s treatment and progress.8
Furthermore, there is a need for an algorithm that has the ability to detect, quantify, and monitor changes in dermatologic conditions using images that patients have uploaded. This capability will lead to creation of a standardized quantification scale that will allow physicians to virtually track the progression of visible skin pathologies.
Hazards of Racial Bias in AI
Artificial intelligence is limited by racial disparity bias seen in computerized medicine. For years, the majority of dermatology research, especially in skin cancer, has been conducted on fairer-skinned populations. This bias has existed at the expense of darker-skinned patients, whose skin conditions and symptoms present differently,9 and reflects directly in available data sets that can be used to develop AI algorithms. Because these data are inadequate to the task, AI might misdiagnose skin cancer in people of color or miss an existing condition entirely.10 Consequently, the higher rate of skin cancer mortality that is reported in people of color is likely to persist with the rise of AI in dermatology.11 A more representative database of imaged skin lesions needs to be utilized to create a diversely representative and applicable data set for AI algorithms.12
Benefits of Conversational Agents
Another method by which AI could be incorporated into dermatology is through what is known as a conversational agent (CA)—AI software that engages in a dialogue with users by interpreting their voice and replying to them through text, image, or voice.13 Conversational agents facilitate remote patient management, allow clinicians to focus on other functions, and aid in data collection.14 A 2014 study showed that patients were significantly more likely to disclose history and emotions when informed they were interacting with a CA than with a human clinician (P=.007).15 Such benefits could be invaluable in dermatology, where emotions and patient perceptions of skin conditions play into the treatment process.
However, some evidence showed that CAs cannot respond to patients’ statements in all circumstances.16 It also is unclear how well CAs recognize nuanced statements that might signal potential harm. This fits into the greater theme of a major problem with AI: the lack of a reliable response in all circumstances.13
Final Thoughts
The practical implementations of AI in dermatology are still being explored. Given the uncertainty surrounding the COVID-19 pandemic and the future of patient care, AI might serve as an important asset in assisting with the diagnosis and treatment of dermatologic conditions, physician productivity, and patient monitoring.
- Amisha, Malik P, Pathania M, et al. Overview of artificial intelligence in medicine. J Family Med Prim Care. 2019;8:2328-2331. doi:10.4103/jfmpc.jfmpc_440_19
- Han SS, Kim MS, Lim W, et al. Classification of the clinical images for benign and malignant cutaneous tumors using a deep learning algorithm. J Invest Dermatol. 2018;138:1529-1538. doi:10.1016/j.jid.2018.01.028
- Esteva A, Kuprel B, Novoa RA, et al. Dermatologist-level classification of skin cancer with deep neural networks. Nature. 2017;542:115-118. doi:10.1038/nature21056
- Brinker TJ, Hekler A, Enk AH, et al. Deep neural networks are superior to dermatologists in melanoma image classification. Eur J Cancer. 2019;119:11-17. doi:10.1016/j.ejca.2019.05.023
- Regulated medical device for detecting skin cancer. SkinVision website. Accessed July 23, 2021. https://www.skinvision.com/hcp/
- Thissen M, Udrea A, Hacking M, et al. mHealth app for risk assessment of pigmented and nonpigmented skin lesions—a study on sensitivity and specificity in detecting malignancy. Telemed J E Health. 2017;23:948-954. doi:10.1089/tmj.2016.0259
- Freeman K, Dinnes J, Chuchu N, et al. Algorithm based smartphone apps to assess risk of skin cancer in adults: systematic review of diagnostic accuracy studies. BMJ. 2020;368:m127. doi:10.1136/bmj.m127
- Puri P, Comfere N, Pittelkow MR, et al. COVID-19: an opportunity to build dermatology’s digital future. Dermatol Ther. 2020;33:e14149. doi:10.1111/dth.14149
- Buster KJ, Stevens EI, Elmets CA. Dermatologic health disparities. Dermatol Clin. 2012;30:53-59,viii. doi:10.1016/j.det.2011.08.002
- Adamson AS, Smith A. Machine learning and health care disparities in dermatology. JAMA Dermatol. 2018;154:1247-1248. doi:10.1001/jamadermatol.2018.2348
- Agbai ON, Buster K, Sanchez M, et al. Skin cancer and photoprotection in people of color: a review and recommendations for physicians and the public. J Am Acad Dermatol. 2014;70:748-762. doi:S0190-9622(13)01296-6
- Alabdulkareem A. Artificial intelligence and dermatologists: friends or foes? J Dermatol Dermatolog Surg. 2019;23:57-60. doi:10.4103/jdds.jdds_19_19
- McGreevey JD 3rd, Hanson CW 3rd, Koppel R. Clinical, legal, and ethical aspects of artificial intelligence-assisted conversational agents in health care. JAMA. 2020;324:552-553. doi:10.1001/jama.2020.2724
- Piau A, Crissey R, Brechemier D, et al. A smartphone chatbot application to optimize monitoring of older patients with cancer. Int J Med Inform. 2019;128:18-23. doi:10.1016/j.ijmedinf.2019.05.013
- Lucas GM, Gratch J, King A, et al. It’s only a computer: virtual humans increase willingness to disclose. Comput Human Behav. 2014;37:94-100. https://doi.org/10.1016/j.chb.2014.04.043
- Miner AS, Milstein A, Schueller S, et al. Smartphone-based conversational agents and responses to questions about mental health, interpersonal violence, and physical health. JAMA Intern Med. 2016;176:619-625. doi:10.1001/jamainternmed.2016.0400
- Amisha, Malik P, Pathania M, et al. Overview of artificial intelligence in medicine. J Family Med Prim Care. 2019;8:2328-2331. doi:10.4103/jfmpc.jfmpc_440_19
- Han SS, Kim MS, Lim W, et al. Classification of the clinical images for benign and malignant cutaneous tumors using a deep learning algorithm. J Invest Dermatol. 2018;138:1529-1538. doi:10.1016/j.jid.2018.01.028
- Esteva A, Kuprel B, Novoa RA, et al. Dermatologist-level classification of skin cancer with deep neural networks. Nature. 2017;542:115-118. doi:10.1038/nature21056
- Brinker TJ, Hekler A, Enk AH, et al. Deep neural networks are superior to dermatologists in melanoma image classification. Eur J Cancer. 2019;119:11-17. doi:10.1016/j.ejca.2019.05.023
- Regulated medical device for detecting skin cancer. SkinVision website. Accessed July 23, 2021. https://www.skinvision.com/hcp/
- Thissen M, Udrea A, Hacking M, et al. mHealth app for risk assessment of pigmented and nonpigmented skin lesions—a study on sensitivity and specificity in detecting malignancy. Telemed J E Health. 2017;23:948-954. doi:10.1089/tmj.2016.0259
- Freeman K, Dinnes J, Chuchu N, et al. Algorithm based smartphone apps to assess risk of skin cancer in adults: systematic review of diagnostic accuracy studies. BMJ. 2020;368:m127. doi:10.1136/bmj.m127
- Puri P, Comfere N, Pittelkow MR, et al. COVID-19: an opportunity to build dermatology’s digital future. Dermatol Ther. 2020;33:e14149. doi:10.1111/dth.14149
- Buster KJ, Stevens EI, Elmets CA. Dermatologic health disparities. Dermatol Clin. 2012;30:53-59,viii. doi:10.1016/j.det.2011.08.002
- Adamson AS, Smith A. Machine learning and health care disparities in dermatology. JAMA Dermatol. 2018;154:1247-1248. doi:10.1001/jamadermatol.2018.2348
- Agbai ON, Buster K, Sanchez M, et al. Skin cancer and photoprotection in people of color: a review and recommendations for physicians and the public. J Am Acad Dermatol. 2014;70:748-762. doi:S0190-9622(13)01296-6
- Alabdulkareem A. Artificial intelligence and dermatologists: friends or foes? J Dermatol Dermatolog Surg. 2019;23:57-60. doi:10.4103/jdds.jdds_19_19
- McGreevey JD 3rd, Hanson CW 3rd, Koppel R. Clinical, legal, and ethical aspects of artificial intelligence-assisted conversational agents in health care. JAMA. 2020;324:552-553. doi:10.1001/jama.2020.2724
- Piau A, Crissey R, Brechemier D, et al. A smartphone chatbot application to optimize monitoring of older patients with cancer. Int J Med Inform. 2019;128:18-23. doi:10.1016/j.ijmedinf.2019.05.013
- Lucas GM, Gratch J, King A, et al. It’s only a computer: virtual humans increase willingness to disclose. Comput Human Behav. 2014;37:94-100. https://doi.org/10.1016/j.chb.2014.04.043
- Miner AS, Milstein A, Schueller S, et al. Smartphone-based conversational agents and responses to questions about mental health, interpersonal violence, and physical health. JAMA Intern Med. 2016;176:619-625. doi:10.1001/jamainternmed.2016.0400
Practice Points
- Dermatologists should amass pictures of dermatologic conditions in skin of color to contribute to growing awareness and knowledge of presentation of disease in this population.
- Dermatologists should use artificial intelligence as a tool for delivering more efficient and beneficial patient care.
A sizzling hybrid meeting of the Society of Gynecologic Surgeons
The 47th Annual Scientific Meeting of the Society of Gynecologic Surgeons (SGS), like so many things in our modern world, endured many changes and had to stay nimble and evolve to changing times. In the end, however, SGS was able to adapt and succeed, just like a skilled gynecologic surgeon in the operating room, to deliver a fresh new type of meeting.
When we chose the meeting theme, “Working together: How collaboration enables us to better help our patients,” we anticipated a meeting discussing medical colleagues and consultants. In our forever-changed world, we knew we needed to reinterpret this to a broader social context. Our special lectures and panel discussions sought to open attendees’ eyes to disparities in health care for people of color and women.
While we highlighted the realities faced by colleagues in medicine, the topics addressed also were designed to grow awareness about struggles our patients encounter as well. Social disparities are sobering, long-standing, and sometimes require creative collaborations to achieve successful outcomes for all patients. The faculty of one of our postgraduate courses reviews in this special 2-part section to
The meeting also kicked off with a postgraduate course on fibroid management, with workshops on harnessing the power of social media and lessons on leadership from a female Fortune 500 CEO, Lori Ryerkerk, offered as well. As the scientific program launched, we were once again treated to strong science on gynecologic surgery, with only a small dip in abstract submissions, despite the challenges of research during a pandemic. Mark Walters, MD, gave the inaugural lecture in his name on the crucial topic of surgical education and teaching. We also heard a special report from the SGS SOCOVID research group, led by Dr. Rosanne Kho, on gynecologic surgery during the pandemic. We also convened a virtual panel for our hybrid attendees on the benefits to patients of a multidisciplinary approach to gynecologic surgery, presented here by Cecile Ferrando, MD.
As our practices continue to grow and evolve, the introduction of innovative technologies can pose a new challenge, as Miles Murphy, MD, and members of the panel on novel gynecologic office procedures will present in this series next month.
The TeLinde keynote speaker was Janet Dombrowski, who works as a coach for many surgeons in various disciplines across the country. She spoke to the resilience gained through community and collaboration.
While our meeting theme dated to the “before” pandemic era, those who were able to be in attendance in person can attest to the value we can all place now on community and personal interactions. With experience strengthened by science, I hope this meeting summary serves to highlight the many ways in which we can collaborate to improve outcomes for ourselves in medicine and for patients.
The 47th Annual Scientific Meeting of the Society of Gynecologic Surgeons (SGS), like so many things in our modern world, endured many changes and had to stay nimble and evolve to changing times. In the end, however, SGS was able to adapt and succeed, just like a skilled gynecologic surgeon in the operating room, to deliver a fresh new type of meeting.
When we chose the meeting theme, “Working together: How collaboration enables us to better help our patients,” we anticipated a meeting discussing medical colleagues and consultants. In our forever-changed world, we knew we needed to reinterpret this to a broader social context. Our special lectures and panel discussions sought to open attendees’ eyes to disparities in health care for people of color and women.
While we highlighted the realities faced by colleagues in medicine, the topics addressed also were designed to grow awareness about struggles our patients encounter as well. Social disparities are sobering, long-standing, and sometimes require creative collaborations to achieve successful outcomes for all patients. The faculty of one of our postgraduate courses reviews in this special 2-part section to
The meeting also kicked off with a postgraduate course on fibroid management, with workshops on harnessing the power of social media and lessons on leadership from a female Fortune 500 CEO, Lori Ryerkerk, offered as well. As the scientific program launched, we were once again treated to strong science on gynecologic surgery, with only a small dip in abstract submissions, despite the challenges of research during a pandemic. Mark Walters, MD, gave the inaugural lecture in his name on the crucial topic of surgical education and teaching. We also heard a special report from the SGS SOCOVID research group, led by Dr. Rosanne Kho, on gynecologic surgery during the pandemic. We also convened a virtual panel for our hybrid attendees on the benefits to patients of a multidisciplinary approach to gynecologic surgery, presented here by Cecile Ferrando, MD.
As our practices continue to grow and evolve, the introduction of innovative technologies can pose a new challenge, as Miles Murphy, MD, and members of the panel on novel gynecologic office procedures will present in this series next month.
The TeLinde keynote speaker was Janet Dombrowski, who works as a coach for many surgeons in various disciplines across the country. She spoke to the resilience gained through community and collaboration.
While our meeting theme dated to the “before” pandemic era, those who were able to be in attendance in person can attest to the value we can all place now on community and personal interactions. With experience strengthened by science, I hope this meeting summary serves to highlight the many ways in which we can collaborate to improve outcomes for ourselves in medicine and for patients.
The 47th Annual Scientific Meeting of the Society of Gynecologic Surgeons (SGS), like so many things in our modern world, endured many changes and had to stay nimble and evolve to changing times. In the end, however, SGS was able to adapt and succeed, just like a skilled gynecologic surgeon in the operating room, to deliver a fresh new type of meeting.
When we chose the meeting theme, “Working together: How collaboration enables us to better help our patients,” we anticipated a meeting discussing medical colleagues and consultants. In our forever-changed world, we knew we needed to reinterpret this to a broader social context. Our special lectures and panel discussions sought to open attendees’ eyes to disparities in health care for people of color and women.
While we highlighted the realities faced by colleagues in medicine, the topics addressed also were designed to grow awareness about struggles our patients encounter as well. Social disparities are sobering, long-standing, and sometimes require creative collaborations to achieve successful outcomes for all patients. The faculty of one of our postgraduate courses reviews in this special 2-part section to
The meeting also kicked off with a postgraduate course on fibroid management, with workshops on harnessing the power of social media and lessons on leadership from a female Fortune 500 CEO, Lori Ryerkerk, offered as well. As the scientific program launched, we were once again treated to strong science on gynecologic surgery, with only a small dip in abstract submissions, despite the challenges of research during a pandemic. Mark Walters, MD, gave the inaugural lecture in his name on the crucial topic of surgical education and teaching. We also heard a special report from the SGS SOCOVID research group, led by Dr. Rosanne Kho, on gynecologic surgery during the pandemic. We also convened a virtual panel for our hybrid attendees on the benefits to patients of a multidisciplinary approach to gynecologic surgery, presented here by Cecile Ferrando, MD.
As our practices continue to grow and evolve, the introduction of innovative technologies can pose a new challenge, as Miles Murphy, MD, and members of the panel on novel gynecologic office procedures will present in this series next month.
The TeLinde keynote speaker was Janet Dombrowski, who works as a coach for many surgeons in various disciplines across the country. She spoke to the resilience gained through community and collaboration.
While our meeting theme dated to the “before” pandemic era, those who were able to be in attendance in person can attest to the value we can all place now on community and personal interactions. With experience strengthened by science, I hope this meeting summary serves to highlight the many ways in which we can collaborate to improve outcomes for ourselves in medicine and for patients.
U.S. health system ranks last among 11 high-income countries
The U.S. health care system ranked last overall among 11 high-income countries in an analysis by the nonprofit Commonwealth Fund, according to a report released on Aug. 4.
The report is the seventh international comparison of countries’ health systems by the Commonwealth Fund since 2004, and the United States has ranked last in every edition, David Blumenthal, MD, president of the Commonwealth Fund, told reporters during a press briefing.
Researchers analyzed survey answers from tens of thousands of patients and physicians in 11 countries. They analyzed performance on 71 measures across five categories – access to care, care process, administrative efficiency, equity, and health care outcomes. Administrative data were gathered from the Organisation for Economic Cooperation and Development and the World Health Organization.
Among contributors to the poor showing by the United States is that half (50%) of lower-income U.S. adults and 27% of higher-income U.S. adults say costs keep them from getting needed health care.
“In no other country does income inequality so profoundly limit access to care,” Dr. Blumenthal said.
In the United Kingdom, only 12% with lower incomes and 7% with higher incomes said costs kept them from care.
In a stark comparison, the researchers found that “a high-income person in the U.S. was more likely to report financial barriers than a low-income person in nearly all the other countries surveyed: Australia, Canada, France, Germany, the Netherlands, New Zealand, Norway, Sweden, Switzerland, and the U.K.”
Norway, the Netherlands, and Australia were ranked at the top overall in that order. Rounding out the 11 in overall ranking were the U.K., Germany, New Zealand, Sweden, France, Switzerland, Canada, and the United States.
“What this report tells us is that our health care system is not working for Americans, particularly those with lower incomes, who are at a severe disadvantage compared to citizens of other countries. And they are paying the price with their health and their lives,” Dr. Blumenthal said in a press release.
“To catch up with other high-income countries, the administration and Congress would have to expand access to health care, equitably, to all Americans, act aggressively to control costs, and invest in the social services we know can lead to a healthier population.”
High infant mortality, low life expectancy in U.S.
Several factors contributed to the U.S. ranking at the bottom of the outcomes category. Among them are that the United States has the highest infant mortality rate (5.7 deaths per 1,000 live births) and lowest life expectancy at age 60 (living on average 23.1 years after age 60), compared with the other countries surveyed. The U.S. rate of preventable mortality (177 deaths per 100,000 population) is more than double that of the best-performing country, Switzerland.
Lead author Eric Schneider, MD, senior vice president for policy and research at the Commonwealth Fund, pointed out that, in terms of the change in avoidable mortality over a decade, not only did the United States have the highest rate, compared with the other countries surveyed, “it also experienced the smallest decline in avoidable mortality over that 10-year period.”
The U.S. maternal mortality rate of 17.4 deaths per 100,000 live births is twice that of France, the country with the next-highest rate (7.6 deaths per 100,000 live births).
U.S. excelled in only one category
The only category in which the United States did not rank last was in “care process,” where it ranked second behind only New Zealand.
The care process category combines preventive care, safe care, coordinated care, and patient engagement and preferences. The category includes indicators such as mammography screening and influenza vaccination for older adults as well as the percentage of adults counseled by a health care provider about nutrition, smoking, or alcohol use.
The United States and Germany performed best on engagement and patient preferences, although U.S. adults have the lowest rates of continuity with the same doctor.
New Zealand and the United States ranked highest in the safe care category, with higher reported use of computerized alerts and routine review of medications.
‘Too little, too late’: Key recommendations for U.S. to improve
Reginald Williams, vice president of International Health Policy and Practice Innovations at the Commonwealth Fund, pointed out that the U.S. shortcomings in health care come despite spending more than twice as much of its GDP (17% in 2019) as the average OECD country.
“It appears that the US delivers too little of the care that is most needed and often delivers that care too late, especially for people with chronic illnesses,” he said.
He then summarized the team’s recommendations on how the United States can change course.
First is expanding insurance coverage, he said, noting that the United States is the only one of the 11 countries that lacks universal coverage and nearly 30 million people remain uninsured.
Top-performing countries in the survey have universal coverage, annual out-of-pocket caps on covered benefits, and full coverage for primary care and treatment for chronic conditions, he said.
The United States must also improve access to care, he said.
“Top-ranking countries like the Netherlands and Norway ensure timely availability to care by telephone on nights and weekends, and in-person follow-up at home, if needed,” he said.
Mr. Williams said reducing administrative burdens is also critical to free up resources for improving health. He gave an example: “Norway determines patient copayments or physician fees on a regional basis, applying standardized copayments to all physicians within a specialty in a geographic area.”
Reducing income-related barriers is important as well, he said.
The fear of unpredictably high bills and other issues prevent people in the United States from getting the care they ultimately need, he said, adding that top-performing countries invest more in social services to reduce health risks.
That could have implications for the COVID-19 response.
Responding effectively to COVID-19 requires that patients can access affordable health care services, Mr. Williams noted.
“We know from our research that more than two-thirds of U.S. adults say their potential out-of-pocket costs would figure prominently in their decisions to get care if they had coronavirus symptoms,” he said.
Dr. Schneider summed up in the press release: “This study makes clear that higher U.S. spending on health care is not producing better health especially as the U.S. continues on a path of deepening inequality. A country that spends as much as we do should have the best health system in the world. We should adapt what works in other high-income countries to build a better health care system that provides affordable, high-quality health care for everyone.”
Dr. Blumenthal, Dr. Schneider, and Mr. Williams reported no relevant financial relationships outside their employment with the Commonwealth Fund.
A version of this article first appeared on Medscape.com.
The U.S. health care system ranked last overall among 11 high-income countries in an analysis by the nonprofit Commonwealth Fund, according to a report released on Aug. 4.
The report is the seventh international comparison of countries’ health systems by the Commonwealth Fund since 2004, and the United States has ranked last in every edition, David Blumenthal, MD, president of the Commonwealth Fund, told reporters during a press briefing.
Researchers analyzed survey answers from tens of thousands of patients and physicians in 11 countries. They analyzed performance on 71 measures across five categories – access to care, care process, administrative efficiency, equity, and health care outcomes. Administrative data were gathered from the Organisation for Economic Cooperation and Development and the World Health Organization.
Among contributors to the poor showing by the United States is that half (50%) of lower-income U.S. adults and 27% of higher-income U.S. adults say costs keep them from getting needed health care.
“In no other country does income inequality so profoundly limit access to care,” Dr. Blumenthal said.
In the United Kingdom, only 12% with lower incomes and 7% with higher incomes said costs kept them from care.
In a stark comparison, the researchers found that “a high-income person in the U.S. was more likely to report financial barriers than a low-income person in nearly all the other countries surveyed: Australia, Canada, France, Germany, the Netherlands, New Zealand, Norway, Sweden, Switzerland, and the U.K.”
Norway, the Netherlands, and Australia were ranked at the top overall in that order. Rounding out the 11 in overall ranking were the U.K., Germany, New Zealand, Sweden, France, Switzerland, Canada, and the United States.
“What this report tells us is that our health care system is not working for Americans, particularly those with lower incomes, who are at a severe disadvantage compared to citizens of other countries. And they are paying the price with their health and their lives,” Dr. Blumenthal said in a press release.
“To catch up with other high-income countries, the administration and Congress would have to expand access to health care, equitably, to all Americans, act aggressively to control costs, and invest in the social services we know can lead to a healthier population.”
High infant mortality, low life expectancy in U.S.
Several factors contributed to the U.S. ranking at the bottom of the outcomes category. Among them are that the United States has the highest infant mortality rate (5.7 deaths per 1,000 live births) and lowest life expectancy at age 60 (living on average 23.1 years after age 60), compared with the other countries surveyed. The U.S. rate of preventable mortality (177 deaths per 100,000 population) is more than double that of the best-performing country, Switzerland.
Lead author Eric Schneider, MD, senior vice president for policy and research at the Commonwealth Fund, pointed out that, in terms of the change in avoidable mortality over a decade, not only did the United States have the highest rate, compared with the other countries surveyed, “it also experienced the smallest decline in avoidable mortality over that 10-year period.”
The U.S. maternal mortality rate of 17.4 deaths per 100,000 live births is twice that of France, the country with the next-highest rate (7.6 deaths per 100,000 live births).
U.S. excelled in only one category
The only category in which the United States did not rank last was in “care process,” where it ranked second behind only New Zealand.
The care process category combines preventive care, safe care, coordinated care, and patient engagement and preferences. The category includes indicators such as mammography screening and influenza vaccination for older adults as well as the percentage of adults counseled by a health care provider about nutrition, smoking, or alcohol use.
The United States and Germany performed best on engagement and patient preferences, although U.S. adults have the lowest rates of continuity with the same doctor.
New Zealand and the United States ranked highest in the safe care category, with higher reported use of computerized alerts and routine review of medications.
‘Too little, too late’: Key recommendations for U.S. to improve
Reginald Williams, vice president of International Health Policy and Practice Innovations at the Commonwealth Fund, pointed out that the U.S. shortcomings in health care come despite spending more than twice as much of its GDP (17% in 2019) as the average OECD country.
“It appears that the US delivers too little of the care that is most needed and often delivers that care too late, especially for people with chronic illnesses,” he said.
He then summarized the team’s recommendations on how the United States can change course.
First is expanding insurance coverage, he said, noting that the United States is the only one of the 11 countries that lacks universal coverage and nearly 30 million people remain uninsured.
Top-performing countries in the survey have universal coverage, annual out-of-pocket caps on covered benefits, and full coverage for primary care and treatment for chronic conditions, he said.
The United States must also improve access to care, he said.
“Top-ranking countries like the Netherlands and Norway ensure timely availability to care by telephone on nights and weekends, and in-person follow-up at home, if needed,” he said.
Mr. Williams said reducing administrative burdens is also critical to free up resources for improving health. He gave an example: “Norway determines patient copayments or physician fees on a regional basis, applying standardized copayments to all physicians within a specialty in a geographic area.”
Reducing income-related barriers is important as well, he said.
The fear of unpredictably high bills and other issues prevent people in the United States from getting the care they ultimately need, he said, adding that top-performing countries invest more in social services to reduce health risks.
That could have implications for the COVID-19 response.
Responding effectively to COVID-19 requires that patients can access affordable health care services, Mr. Williams noted.
“We know from our research that more than two-thirds of U.S. adults say their potential out-of-pocket costs would figure prominently in their decisions to get care if they had coronavirus symptoms,” he said.
Dr. Schneider summed up in the press release: “This study makes clear that higher U.S. spending on health care is not producing better health especially as the U.S. continues on a path of deepening inequality. A country that spends as much as we do should have the best health system in the world. We should adapt what works in other high-income countries to build a better health care system that provides affordable, high-quality health care for everyone.”
Dr. Blumenthal, Dr. Schneider, and Mr. Williams reported no relevant financial relationships outside their employment with the Commonwealth Fund.
A version of this article first appeared on Medscape.com.
The U.S. health care system ranked last overall among 11 high-income countries in an analysis by the nonprofit Commonwealth Fund, according to a report released on Aug. 4.
The report is the seventh international comparison of countries’ health systems by the Commonwealth Fund since 2004, and the United States has ranked last in every edition, David Blumenthal, MD, president of the Commonwealth Fund, told reporters during a press briefing.
Researchers analyzed survey answers from tens of thousands of patients and physicians in 11 countries. They analyzed performance on 71 measures across five categories – access to care, care process, administrative efficiency, equity, and health care outcomes. Administrative data were gathered from the Organisation for Economic Cooperation and Development and the World Health Organization.
Among contributors to the poor showing by the United States is that half (50%) of lower-income U.S. adults and 27% of higher-income U.S. adults say costs keep them from getting needed health care.
“In no other country does income inequality so profoundly limit access to care,” Dr. Blumenthal said.
In the United Kingdom, only 12% with lower incomes and 7% with higher incomes said costs kept them from care.
In a stark comparison, the researchers found that “a high-income person in the U.S. was more likely to report financial barriers than a low-income person in nearly all the other countries surveyed: Australia, Canada, France, Germany, the Netherlands, New Zealand, Norway, Sweden, Switzerland, and the U.K.”
Norway, the Netherlands, and Australia were ranked at the top overall in that order. Rounding out the 11 in overall ranking were the U.K., Germany, New Zealand, Sweden, France, Switzerland, Canada, and the United States.
“What this report tells us is that our health care system is not working for Americans, particularly those with lower incomes, who are at a severe disadvantage compared to citizens of other countries. And they are paying the price with their health and their lives,” Dr. Blumenthal said in a press release.
“To catch up with other high-income countries, the administration and Congress would have to expand access to health care, equitably, to all Americans, act aggressively to control costs, and invest in the social services we know can lead to a healthier population.”
High infant mortality, low life expectancy in U.S.
Several factors contributed to the U.S. ranking at the bottom of the outcomes category. Among them are that the United States has the highest infant mortality rate (5.7 deaths per 1,000 live births) and lowest life expectancy at age 60 (living on average 23.1 years after age 60), compared with the other countries surveyed. The U.S. rate of preventable mortality (177 deaths per 100,000 population) is more than double that of the best-performing country, Switzerland.
Lead author Eric Schneider, MD, senior vice president for policy and research at the Commonwealth Fund, pointed out that, in terms of the change in avoidable mortality over a decade, not only did the United States have the highest rate, compared with the other countries surveyed, “it also experienced the smallest decline in avoidable mortality over that 10-year period.”
The U.S. maternal mortality rate of 17.4 deaths per 100,000 live births is twice that of France, the country with the next-highest rate (7.6 deaths per 100,000 live births).
U.S. excelled in only one category
The only category in which the United States did not rank last was in “care process,” where it ranked second behind only New Zealand.
The care process category combines preventive care, safe care, coordinated care, and patient engagement and preferences. The category includes indicators such as mammography screening and influenza vaccination for older adults as well as the percentage of adults counseled by a health care provider about nutrition, smoking, or alcohol use.
The United States and Germany performed best on engagement and patient preferences, although U.S. adults have the lowest rates of continuity with the same doctor.
New Zealand and the United States ranked highest in the safe care category, with higher reported use of computerized alerts and routine review of medications.
‘Too little, too late’: Key recommendations for U.S. to improve
Reginald Williams, vice president of International Health Policy and Practice Innovations at the Commonwealth Fund, pointed out that the U.S. shortcomings in health care come despite spending more than twice as much of its GDP (17% in 2019) as the average OECD country.
“It appears that the US delivers too little of the care that is most needed and often delivers that care too late, especially for people with chronic illnesses,” he said.
He then summarized the team’s recommendations on how the United States can change course.
First is expanding insurance coverage, he said, noting that the United States is the only one of the 11 countries that lacks universal coverage and nearly 30 million people remain uninsured.
Top-performing countries in the survey have universal coverage, annual out-of-pocket caps on covered benefits, and full coverage for primary care and treatment for chronic conditions, he said.
The United States must also improve access to care, he said.
“Top-ranking countries like the Netherlands and Norway ensure timely availability to care by telephone on nights and weekends, and in-person follow-up at home, if needed,” he said.
Mr. Williams said reducing administrative burdens is also critical to free up resources for improving health. He gave an example: “Norway determines patient copayments or physician fees on a regional basis, applying standardized copayments to all physicians within a specialty in a geographic area.”
Reducing income-related barriers is important as well, he said.
The fear of unpredictably high bills and other issues prevent people in the United States from getting the care they ultimately need, he said, adding that top-performing countries invest more in social services to reduce health risks.
That could have implications for the COVID-19 response.
Responding effectively to COVID-19 requires that patients can access affordable health care services, Mr. Williams noted.
“We know from our research that more than two-thirds of U.S. adults say their potential out-of-pocket costs would figure prominently in their decisions to get care if they had coronavirus symptoms,” he said.
Dr. Schneider summed up in the press release: “This study makes clear that higher U.S. spending on health care is not producing better health especially as the U.S. continues on a path of deepening inequality. A country that spends as much as we do should have the best health system in the world. We should adapt what works in other high-income countries to build a better health care system that provides affordable, high-quality health care for everyone.”
Dr. Blumenthal, Dr. Schneider, and Mr. Williams reported no relevant financial relationships outside their employment with the Commonwealth Fund.
A version of this article first appeared on Medscape.com.
Oncologists face nightmares every day with prior authorization
Editor’s note: Prior authorization has been flagged as the biggest payer-related cause of stress for U.S. oncologists. In one survey, 75% said prior authorization was their biggest burden, followed by coverage denials and appeals (62%). Another survey found that practices spent on average 16.4 hours a week dealing with prior authorizations.
Around 5% of my emails every day are from insurance companies denying my patients the treatments I have recommended. A part of every day is spent worrying about how I’m going to cover my patients’ therapy and what I need to order to make sure it doesn’t get delayed.
Many doctors are retiring because they don’t want to deal with this anymore. There are many times that I have thought about quitting for this reason. A partner of mine had a heart attack last year. He’s a few years older than I am – in his mid-50s – and that scared me. I actually had a CT angiogram just to make sure. They told me my heart is fine, but I worry because of all these frustrations every day. And I’m not alone. For every doctor I work with, it’s the same story, and it’s just ridiculous.
For example, I had a patient with a huge breast mass. My nurse got me the prior authorization for an emergency biopsy. I got back the results for estrogen and progesterone receptor status, but not the HER2-neu results because that test required another authorization.
Authorization shouldn’t be required for every single step. I understand if maybe you need to get an authorization to do something outside the standard of care or something that is unique or unheard of, but HER2-neu biopsy is standard of care and should not require additional authorization.
And the sad part is, that patient turned out to be HER2-neu positive. She lost 4 weeks just waiting for an authorization of a test that should be a no-brainer.
We cannot even do a blood count in our office before getting authorization from some insurances. This is a very important test when we give chemotherapy, and it’s very cheap.
And then another nightmare is if you want to give a patient growth factors when you see their blood count is going down. Sometimes the insurance company will say, “When they get neutropenic fever, we’ll allow it with the next cycle.” Why do I have to wait until the patient develops such a problem to start with a treatment that could avoid it? They may end up in the hospital.
I think I’m one of the more conservative doctors; I try to do everything scientifically and only order a test or a treatment if it’s indicated. But sometimes this guidance costs more money. For example, an insurance company may say to order a CT scan first and if you don’t find your answer, then get a PET scan.
So I order a CT scan, knowing it’s not going to help, and then I tell them, “Now I need a PET scan.” That’s another week delay and an extra cost that I don’t want the system to incur.
I’ve even had some issues with lung screening scans for smokers. This screening has reduced mortality by 20%; it should be a no-brainer to encourage smokers to do it because many of them may not even need chemotherapy if you find early-stage lung cancer. And the screening is not expensive, you can do it for $90 to $100. So why do we have to get authorization for that?
Sometimes I push back and request a “peer-to-peer,” where you challenge the decision of the insurance company and speak to one of their doctors. Out of 10 doctors, maybe three or four will do the peer-to-peer. The rest will give up because it’s so frustrating.
In one case, I wanted to modify a standard regimen and give only two out of three drugs because I thought the third would be too toxic. But the insurance company wouldn’t approve the regimen because the guidelines say you have to give three drugs.
Guidelines are guidance, they should not dictate how you treat an individual patient – there should be some allowance in there for a doctor’s discretion. If not, why do we even need doctors? We could just follow treatment regimens dictated by computers. They have to allow me to personalize the care that my patient deserves and make changes so that the treatment can be tolerated.
But then, I get that one patient whom I feel I really helped and I realize, “Okay, I can help more people.”
I had this one patient, a young, 40-year-old nurse with breast cancer – also HER2-neu positive. She’d had her surgery and finished her adjuvant chemotherapy. One of the things that you do as standard of care, after a year of trastuzumab, is you start them on neratinib. There are studies that show it improves progression-free survival if you give them an extra year of this drug as an adjuvant.
I prescribed the neratinib, but the insurance company denied it because the patient “did not have positive lymph nodes and was not considered high risk.” I told them, “That’s BS, that’s not what the indication is for.” I asked for a peer-to-peer and they said the policy did not allow for peer-to-peer. So, I made a big fuss about it. We appealed, and I finally spoke to a pharmacist who worked for the insurance company. I told him, “Why did you guys deny this? It’s standard of care.” He said, “Oh, I agree with you, this will be approved. And actually, we’re going to change the policy now.”
When that pharmacist told me they were going to change the policy, it was like someone gave me 1 million dollars. Because, you know what? I didn’t just help my patient; now other patients will also get it. The hope is that if you keep fighting for something, they will change it.
I think every doctor wants to do the best for their patients. It’s not like they don’t want to, but really, I am fortunate that I have the means to do it. We’re a big practice and we have dedicated staff who can help.
If you’re a small practice, it’s almost impossible to deal with this. I have two nurse practitioners, and a lot of their work is filling out paperwork for insurance companies.
We had a colleague, a solo practitioner, who would send us his patients with complicated therapies, because he couldn’t afford the time or the effort or the risk of not getting reimbursed. His practice could have paid out $100,000 for drugs and not get a reimbursement for a few months.
Even when an insurance company does give the preauthorization, there’s always this disclaimer that it doesn’t guarantee payment. If they find in the future that your patient didn’t meet the criteria, they can still deny payment.
If the insurers refuse coverage, we really work hard at getting patients free drugs, and most of the time, we manage to do that. We either look to charitable organizations, like the Leukemia and Lymphoma Society, or we look for rare disease societies or we go to the pharmaceutical company.
For really expensive drugs, pharmaceutical companies have a program where you can enroll the patient and they can help copay or even cover the drug. For less expensive drugs, it might not be a big problem, but for a drug that can cost $18,000 to $20,000 a month, that’s a big risk to take.
It’s confusing for patients, too. They get angry and frustrated, and that’s not good for their treatment, because attitude and psychology are very important. Sometimes they yell at us because they think it’s our fault. I encourage them to call their insurance companies themselves, and some of them do.
I don’t do it with every patient, but there are some more educated patients who are advocates, and if their condition is stable, I do encourage them to call their senators or congressmen or congresswomen to complain.
I don’t mind treating complicated patients. I don’t want to say I enjoy it, but I like challenges. That’s my field, that’s medicine, that’s what I’m supposed to do. But it’s really sad and frustrating that, when you want to treat a patient, you first have to look at their insurance to see how much care you can actually give them.
Maen Hussein, MD, is physician director of finance at Florida Cancer Specialists and Research Institute, Fort Myers. He is a board member of the Florida Cancer Specialists Foundation and sits on the board of directors for the Florida Society of Clinical Oncology.
A version of this article first appeared on Medscape.com.
Editor’s note: Prior authorization has been flagged as the biggest payer-related cause of stress for U.S. oncologists. In one survey, 75% said prior authorization was their biggest burden, followed by coverage denials and appeals (62%). Another survey found that practices spent on average 16.4 hours a week dealing with prior authorizations.
Around 5% of my emails every day are from insurance companies denying my patients the treatments I have recommended. A part of every day is spent worrying about how I’m going to cover my patients’ therapy and what I need to order to make sure it doesn’t get delayed.
Many doctors are retiring because they don’t want to deal with this anymore. There are many times that I have thought about quitting for this reason. A partner of mine had a heart attack last year. He’s a few years older than I am – in his mid-50s – and that scared me. I actually had a CT angiogram just to make sure. They told me my heart is fine, but I worry because of all these frustrations every day. And I’m not alone. For every doctor I work with, it’s the same story, and it’s just ridiculous.
For example, I had a patient with a huge breast mass. My nurse got me the prior authorization for an emergency biopsy. I got back the results for estrogen and progesterone receptor status, but not the HER2-neu results because that test required another authorization.
Authorization shouldn’t be required for every single step. I understand if maybe you need to get an authorization to do something outside the standard of care or something that is unique or unheard of, but HER2-neu biopsy is standard of care and should not require additional authorization.
And the sad part is, that patient turned out to be HER2-neu positive. She lost 4 weeks just waiting for an authorization of a test that should be a no-brainer.
We cannot even do a blood count in our office before getting authorization from some insurances. This is a very important test when we give chemotherapy, and it’s very cheap.
And then another nightmare is if you want to give a patient growth factors when you see their blood count is going down. Sometimes the insurance company will say, “When they get neutropenic fever, we’ll allow it with the next cycle.” Why do I have to wait until the patient develops such a problem to start with a treatment that could avoid it? They may end up in the hospital.
I think I’m one of the more conservative doctors; I try to do everything scientifically and only order a test or a treatment if it’s indicated. But sometimes this guidance costs more money. For example, an insurance company may say to order a CT scan first and if you don’t find your answer, then get a PET scan.
So I order a CT scan, knowing it’s not going to help, and then I tell them, “Now I need a PET scan.” That’s another week delay and an extra cost that I don’t want the system to incur.
I’ve even had some issues with lung screening scans for smokers. This screening has reduced mortality by 20%; it should be a no-brainer to encourage smokers to do it because many of them may not even need chemotherapy if you find early-stage lung cancer. And the screening is not expensive, you can do it for $90 to $100. So why do we have to get authorization for that?
Sometimes I push back and request a “peer-to-peer,” where you challenge the decision of the insurance company and speak to one of their doctors. Out of 10 doctors, maybe three or four will do the peer-to-peer. The rest will give up because it’s so frustrating.
In one case, I wanted to modify a standard regimen and give only two out of three drugs because I thought the third would be too toxic. But the insurance company wouldn’t approve the regimen because the guidelines say you have to give three drugs.
Guidelines are guidance, they should not dictate how you treat an individual patient – there should be some allowance in there for a doctor’s discretion. If not, why do we even need doctors? We could just follow treatment regimens dictated by computers. They have to allow me to personalize the care that my patient deserves and make changes so that the treatment can be tolerated.
But then, I get that one patient whom I feel I really helped and I realize, “Okay, I can help more people.”
I had this one patient, a young, 40-year-old nurse with breast cancer – also HER2-neu positive. She’d had her surgery and finished her adjuvant chemotherapy. One of the things that you do as standard of care, after a year of trastuzumab, is you start them on neratinib. There are studies that show it improves progression-free survival if you give them an extra year of this drug as an adjuvant.
I prescribed the neratinib, but the insurance company denied it because the patient “did not have positive lymph nodes and was not considered high risk.” I told them, “That’s BS, that’s not what the indication is for.” I asked for a peer-to-peer and they said the policy did not allow for peer-to-peer. So, I made a big fuss about it. We appealed, and I finally spoke to a pharmacist who worked for the insurance company. I told him, “Why did you guys deny this? It’s standard of care.” He said, “Oh, I agree with you, this will be approved. And actually, we’re going to change the policy now.”
When that pharmacist told me they were going to change the policy, it was like someone gave me 1 million dollars. Because, you know what? I didn’t just help my patient; now other patients will also get it. The hope is that if you keep fighting for something, they will change it.
I think every doctor wants to do the best for their patients. It’s not like they don’t want to, but really, I am fortunate that I have the means to do it. We’re a big practice and we have dedicated staff who can help.
If you’re a small practice, it’s almost impossible to deal with this. I have two nurse practitioners, and a lot of their work is filling out paperwork for insurance companies.
We had a colleague, a solo practitioner, who would send us his patients with complicated therapies, because he couldn’t afford the time or the effort or the risk of not getting reimbursed. His practice could have paid out $100,000 for drugs and not get a reimbursement for a few months.
Even when an insurance company does give the preauthorization, there’s always this disclaimer that it doesn’t guarantee payment. If they find in the future that your patient didn’t meet the criteria, they can still deny payment.
If the insurers refuse coverage, we really work hard at getting patients free drugs, and most of the time, we manage to do that. We either look to charitable organizations, like the Leukemia and Lymphoma Society, or we look for rare disease societies or we go to the pharmaceutical company.
For really expensive drugs, pharmaceutical companies have a program where you can enroll the patient and they can help copay or even cover the drug. For less expensive drugs, it might not be a big problem, but for a drug that can cost $18,000 to $20,000 a month, that’s a big risk to take.
It’s confusing for patients, too. They get angry and frustrated, and that’s not good for their treatment, because attitude and psychology are very important. Sometimes they yell at us because they think it’s our fault. I encourage them to call their insurance companies themselves, and some of them do.
I don’t do it with every patient, but there are some more educated patients who are advocates, and if their condition is stable, I do encourage them to call their senators or congressmen or congresswomen to complain.
I don’t mind treating complicated patients. I don’t want to say I enjoy it, but I like challenges. That’s my field, that’s medicine, that’s what I’m supposed to do. But it’s really sad and frustrating that, when you want to treat a patient, you first have to look at their insurance to see how much care you can actually give them.
Maen Hussein, MD, is physician director of finance at Florida Cancer Specialists and Research Institute, Fort Myers. He is a board member of the Florida Cancer Specialists Foundation and sits on the board of directors for the Florida Society of Clinical Oncology.
A version of this article first appeared on Medscape.com.
Editor’s note: Prior authorization has been flagged as the biggest payer-related cause of stress for U.S. oncologists. In one survey, 75% said prior authorization was their biggest burden, followed by coverage denials and appeals (62%). Another survey found that practices spent on average 16.4 hours a week dealing with prior authorizations.
Around 5% of my emails every day are from insurance companies denying my patients the treatments I have recommended. A part of every day is spent worrying about how I’m going to cover my patients’ therapy and what I need to order to make sure it doesn’t get delayed.
Many doctors are retiring because they don’t want to deal with this anymore. There are many times that I have thought about quitting for this reason. A partner of mine had a heart attack last year. He’s a few years older than I am – in his mid-50s – and that scared me. I actually had a CT angiogram just to make sure. They told me my heart is fine, but I worry because of all these frustrations every day. And I’m not alone. For every doctor I work with, it’s the same story, and it’s just ridiculous.
For example, I had a patient with a huge breast mass. My nurse got me the prior authorization for an emergency biopsy. I got back the results for estrogen and progesterone receptor status, but not the HER2-neu results because that test required another authorization.
Authorization shouldn’t be required for every single step. I understand if maybe you need to get an authorization to do something outside the standard of care or something that is unique or unheard of, but HER2-neu biopsy is standard of care and should not require additional authorization.
And the sad part is, that patient turned out to be HER2-neu positive. She lost 4 weeks just waiting for an authorization of a test that should be a no-brainer.
We cannot even do a blood count in our office before getting authorization from some insurances. This is a very important test when we give chemotherapy, and it’s very cheap.
And then another nightmare is if you want to give a patient growth factors when you see their blood count is going down. Sometimes the insurance company will say, “When they get neutropenic fever, we’ll allow it with the next cycle.” Why do I have to wait until the patient develops such a problem to start with a treatment that could avoid it? They may end up in the hospital.
I think I’m one of the more conservative doctors; I try to do everything scientifically and only order a test or a treatment if it’s indicated. But sometimes this guidance costs more money. For example, an insurance company may say to order a CT scan first and if you don’t find your answer, then get a PET scan.
So I order a CT scan, knowing it’s not going to help, and then I tell them, “Now I need a PET scan.” That’s another week delay and an extra cost that I don’t want the system to incur.
I’ve even had some issues with lung screening scans for smokers. This screening has reduced mortality by 20%; it should be a no-brainer to encourage smokers to do it because many of them may not even need chemotherapy if you find early-stage lung cancer. And the screening is not expensive, you can do it for $90 to $100. So why do we have to get authorization for that?
Sometimes I push back and request a “peer-to-peer,” where you challenge the decision of the insurance company and speak to one of their doctors. Out of 10 doctors, maybe three or four will do the peer-to-peer. The rest will give up because it’s so frustrating.
In one case, I wanted to modify a standard regimen and give only two out of three drugs because I thought the third would be too toxic. But the insurance company wouldn’t approve the regimen because the guidelines say you have to give three drugs.
Guidelines are guidance, they should not dictate how you treat an individual patient – there should be some allowance in there for a doctor’s discretion. If not, why do we even need doctors? We could just follow treatment regimens dictated by computers. They have to allow me to personalize the care that my patient deserves and make changes so that the treatment can be tolerated.
But then, I get that one patient whom I feel I really helped and I realize, “Okay, I can help more people.”
I had this one patient, a young, 40-year-old nurse with breast cancer – also HER2-neu positive. She’d had her surgery and finished her adjuvant chemotherapy. One of the things that you do as standard of care, after a year of trastuzumab, is you start them on neratinib. There are studies that show it improves progression-free survival if you give them an extra year of this drug as an adjuvant.
I prescribed the neratinib, but the insurance company denied it because the patient “did not have positive lymph nodes and was not considered high risk.” I told them, “That’s BS, that’s not what the indication is for.” I asked for a peer-to-peer and they said the policy did not allow for peer-to-peer. So, I made a big fuss about it. We appealed, and I finally spoke to a pharmacist who worked for the insurance company. I told him, “Why did you guys deny this? It’s standard of care.” He said, “Oh, I agree with you, this will be approved. And actually, we’re going to change the policy now.”
When that pharmacist told me they were going to change the policy, it was like someone gave me 1 million dollars. Because, you know what? I didn’t just help my patient; now other patients will also get it. The hope is that if you keep fighting for something, they will change it.
I think every doctor wants to do the best for their patients. It’s not like they don’t want to, but really, I am fortunate that I have the means to do it. We’re a big practice and we have dedicated staff who can help.
If you’re a small practice, it’s almost impossible to deal with this. I have two nurse practitioners, and a lot of their work is filling out paperwork for insurance companies.
We had a colleague, a solo practitioner, who would send us his patients with complicated therapies, because he couldn’t afford the time or the effort or the risk of not getting reimbursed. His practice could have paid out $100,000 for drugs and not get a reimbursement for a few months.
Even when an insurance company does give the preauthorization, there’s always this disclaimer that it doesn’t guarantee payment. If they find in the future that your patient didn’t meet the criteria, they can still deny payment.
If the insurers refuse coverage, we really work hard at getting patients free drugs, and most of the time, we manage to do that. We either look to charitable organizations, like the Leukemia and Lymphoma Society, or we look for rare disease societies or we go to the pharmaceutical company.
For really expensive drugs, pharmaceutical companies have a program where you can enroll the patient and they can help copay or even cover the drug. For less expensive drugs, it might not be a big problem, but for a drug that can cost $18,000 to $20,000 a month, that’s a big risk to take.
It’s confusing for patients, too. They get angry and frustrated, and that’s not good for their treatment, because attitude and psychology are very important. Sometimes they yell at us because they think it’s our fault. I encourage them to call their insurance companies themselves, and some of them do.
I don’t do it with every patient, but there are some more educated patients who are advocates, and if their condition is stable, I do encourage them to call their senators or congressmen or congresswomen to complain.
I don’t mind treating complicated patients. I don’t want to say I enjoy it, but I like challenges. That’s my field, that’s medicine, that’s what I’m supposed to do. But it’s really sad and frustrating that, when you want to treat a patient, you first have to look at their insurance to see how much care you can actually give them.
Maen Hussein, MD, is physician director of finance at Florida Cancer Specialists and Research Institute, Fort Myers. He is a board member of the Florida Cancer Specialists Foundation and sits on the board of directors for the Florida Society of Clinical Oncology.
A version of this article first appeared on Medscape.com.
Feasibility of Risk Stratification of Patients Presenting to the Emergency Department With Chest Pain Using HEART Score
From the Department of Internal Medicine, Mount Sinai Health System, Icahn School of Medicine at Mount Sinai, New York, NY (Dr. Gandhi), and the School of Medicine, Seth Gordhandas Sunderdas Medical College, and King Edward Memorial Hospital, Mumbai, India (Drs. Gandhi and Tiwari).
Objective: Calculation of HEART score to (1) stratify patients as low-risk, intermediate-risk, high-risk, and to predict the short-term incidence of major adverse cardiovascular events (MACE), and (2) demonstrate feasibility of HEART score in our local settings.
Design: A prospective cohort study of patients with a chief complaint of chest pain concerning for acute coronary syndrome.
Setting: Participants were recruited from the emergency department (ED) of King Edward Memorial Hospital, a tertiary care academic medical center and a resource-limited setting in Mumbai, India.
Participants: We evaluated 141 patients aged 18 years and older presenting to the ED and stratified them using the HEART score. To assess patients’ progress, a follow-up phone call was made within 6 weeks after presentation to the ED.
Measurements: The primary outcomes were a risk stratification, 6-week occurrence of MACE, and performance of unscheduled revascularization or stress testing. The secondary outcomes were discharge or death.
Results: The 141 participants were stratified into low-risk, intermediate-risk, and high-risk groups: 67 (47.52%), 44 (31.21%), and 30 (21.28%), respectively. The 6-week incidence of MACE in each category was 1.49%, 18.18%, and 90%, respectively. An acute myocardial infarction was diagnosed in 24 patients (17.02%), 15 patients (10.64%) underwent percutaneous coronary intervention (PCI), and 4 patients (2.84%) underwent coronary artery bypass graft (CABG). Overall, 98.5% of low-risk patients and 93.33% of high-risk patients had an uneventful recovery following discharge; therefore, extrapolation based on results demonstrated reduced health care utilization. All the survey respondents found the HEART score to be feasible. The patient characteristics and risk profile of the patients with and without MACE demonstrated that: patients with MACE were older and had a higher proportion of males, hypertension, type 2 diabetes mellitus, smoking, hypercholesterolemia, prior history of PCI/CABG, and history of stroke.
Conclusion: The HEART score seems to be a useful tool for risk stratification and a reliable predictor of outcomes in chest pain patients and can therefore be used for triage.
Keywords: chest pain; emergency department; HEART score; acute coronary syndrome; major adverse cardiac events; myocardial infarction; revascularization.
Cardiovascular diseases (CVDs), especially coronary heart disease (CHD), have epidemic proportions worldwide. Globally, in 2012, CVD led to 17.5 million deaths,1,2 with more than 75% of them occurring in developing countries. In contrast to developed countries, where mortality from CHD is rapidly declining, it is increasing in developing countries.1,3 Current estimates from epidemiologic studies from various parts of India indicate the prevalence of CHD in India to be between 7% and 13% in urban populations and 2% and 7% in rural populations.4
Premature mortality in terms of years of life lost because of CVD in India increased by 59% over a 20-year span, from 23.2 million in 1990 to 37 million in 2010.5 Studies conducted in Mumbai (Mumbai Cohort Study) reported very high CVD mortality rates, approaching 500 per 100 000 for men and 250 per 100 000 for women.6,7 However, to the best of our knowledge, in the Indian population, there are minimal data on utilization of a triage score, such as the HEART score, in chest pain patients in the emergency department (ED) in a resource-limited setting.
The most common reason for admitting patients to the ED is chest pain.8 There are various cardiac and noncardiac etiologies of chest pain presentation. Acute coronary syndrome (ACS) needs to be ruled out first in every patient presenting with chest pain. However, 80% of patients with ACS have no clear diagnostic features on presentation.9 The timely diagnosis and treatment of patients with ACS improves their prognosis. Therefore, clinicians tend to start each patient on ACS treatment to reduce the risk, which often leads to increased costs due to unnecessary, time-consuming diagnostic procedures that may place burdens on both the health care system and the patient.10
Several risk-stratifying tools have been developed in the last few years. Both the GRACE and TIMI risk scores have been designed for risk stratification of patients with proven ACS and not for the chest pain population at the ED.11 Some of these tools are applicable to patients with all types of chest pain presenting to the ED, such as the Manchester Triage System. Other, more selective systems are devoted to the risk stratification of suspected ACS in the ED. One is the HEART score.12
The first study on the HEART score—an acronym that stands for History, Electrocardiogram, Age, Risk factors, and Troponin—was done by Backus et al, who proved that the HEART score is an easy, quick, and reliable predictor of outcomes in chest pain patients.10 The HEART score predicts the short-term incidence of major adverse cardiac events (MACE), which allows clinicians to stratify patients as low-risk, intermediate-risk, and high-risk and to guide their clinical decision-making accordingly. It was developed to provide clinicians with a simple, reliable predictor of cardiac risk on the basis of the lowest score of 0 (very low-risk) up to a score of 10 (very high-risk).
We studied the clinical performance of the HEART score in patients with chest pain, focusing on the efficacy and safety of rapidly identifying patients at risk of MACE. We aimed to determine (1) whether the HEART score is a reliable predictor of outcomes of chest pain patients presenting to the ED; (2) whether the score is feasible in our local settings; and (3) whether it describes the risk profile of patients with and without MACE.
Methods
Setting
Participants were recruited from the ED of King Edward Memorial Hospital, a municipal teaching hospital in Mumbai. The study institute is a tertiary care academic medical center located in Parel, Mumbai, Maharashtra, and is a resource-limited setting serving urban, suburban, and rural populations. Participants requiring urgent attention are first seen by a casualty officer and then referred to the emergency ward. Here, the physician on duty evaluates them and decides on admission to the various wards, like the general ward, medical intensive care unit (ICU), coronary care unit (CCU), etc. The specialist’s opinion may also be obtained before admission. Critically ill patients are initially admitted to the emergency ward and stabilized before being shifted to other areas of the hospital.
Participants
Patients aged 18 years and older presenting with symptoms of acute chest pain or suspected ACS were stratified by priority using the chest pain scoring system—the HEART score. Only patients presenting to the ED were eligible for the study. Informed consent from the patient or next of kin was mandatory for participation in the study.
Patients were determined ineligible for the following reasons: a clear cause for chest pain other than ACS (eg, trauma, diagnosed aortic dissection), persisting or recurrent chest pain caused by rheumatic diseases or cancer (a terminal illness), pregnancy, unable or unwilling to provide informed consent, or incomplete data.
Study design
We conducted a
We conducted our study to determine the importance of calculating the HEART score in each patient, which will help to correctly place them into low-, intermediate-, and high-risk groups for clinically important, irreversible adverse cardiac events and guide the clinical decision-making. Patients with low risk will avoid costly tests and hospital admissions, thus decreasing the cost of treatment and ensuring timely discharge from the ED. Patients with high risk will be treated immediately, to possibly prevent a life-threatening, ACS-related incident. Thus, the HEART score will serve as a quick and reliable predictor of outcomes in chest pain patients and help clinicians to make accurate diagnostic and therapeutic choices in uncertain situations.
HEART score
The total number of points for History, Electrocardiogram (ECG), Age, Risk factors, and Troponin was noted as the HEART score (Table 1).
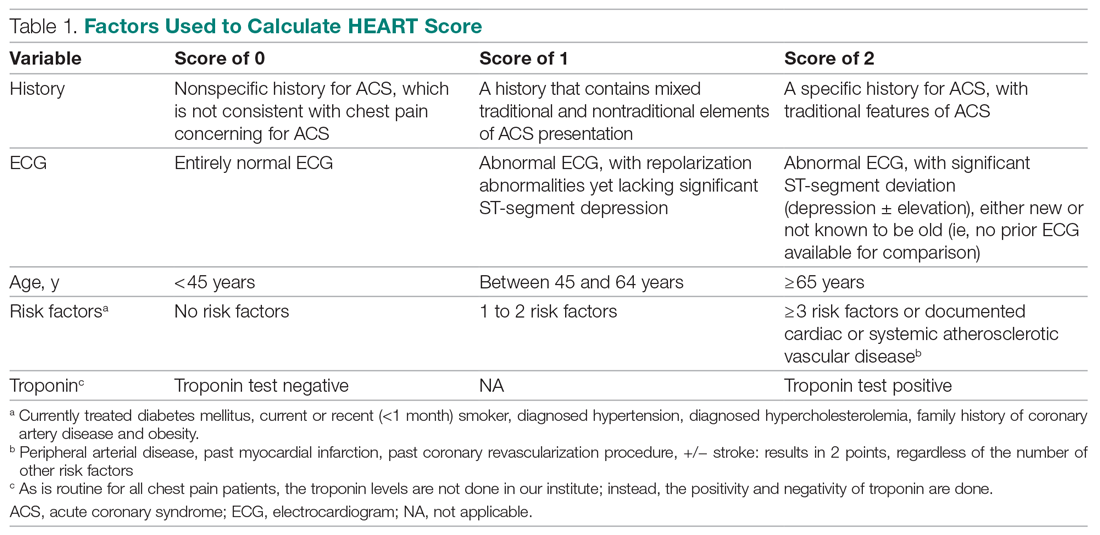
For this study, the patient’s history and ECGs were interpreted by internal medicine attending physicians in the ED. The ECG taken in the emergency room was reviewed and classified, and a copy of the admission ECG was added to the file. The recommendation for patients with a HEART score in a particular range was evaluated. Notably, those with a score of 3 or lower led to a recommendation of reassurance and early discharge. Those with a HEART score in the intermediate range (4-6) were admitted to the hospital for further clinical observation and testing, whereas a high HEART score (7-10) led to admission for intensive monitoring and early intervention. In the analysis of HEART score data, we only used those patients having records for all 5 parameters, excluding patients without an ECG or troponin test.
Results
Myocardial infarction (MI) was defined based on Universal Definition of Myocardial Infarction.13 Coronary revascularization was defined as angioplasty with or without stent placement or coronary artery bypass surgery.14 Percutaneous coronary intervention (PCI) was defined as any therapeutic catheter intervention in the coronary arteries. Coronary artery bypass graft (CABG) surgery was defined as any cardiac surgery in which coronary arteries were operated on.
The primary outcomes in this study were the (1) risk stratification of chest pain patients into low-risk, intermediate-risk, and high-risk categories; (2) incidence of a MACE within 6 weeks of initial presentation. MACE consists of acute myocardial infarction (AMI), PCI, CABG, coronary angiography revealing procedurally correctable stenosis managed conservatively, and death due to any cause.
Our secondary outcomes were discharge or death due to any cause within 6 weeks after presentation.
Follow-up
Within 6 weeks after presentation to the ED, a follow-up phone call was placed to assess the patient’s progress. The follow-up focused on the endpoint of MACE, comprising all-cause death, MI, and revascularization. No patient was lost to follow-up.
Statistical analysis
We aimed to find a difference in the 6-week MACE between low-, intermediate-, and high-risk categories of the HEART score. The prevalence of CHD in India is 10%,4 and assuming an α of 0.05, we needed a sample of 141 patients from the ED patient population. Continuous variables were presented by mean (SD), and categorical variables as percentages. We used t test and the Mann-Whitney U test for comparison of means for continuous variables, χ2 for categorical variables, and Fisher’s exact test for comparison of the categorical variables. Results with P < .05 were considered statistically significant.
We evaluated 141 patients presenting to the ED with chest pain concerning for ACS during the study period, from July 2019 to October 2019.
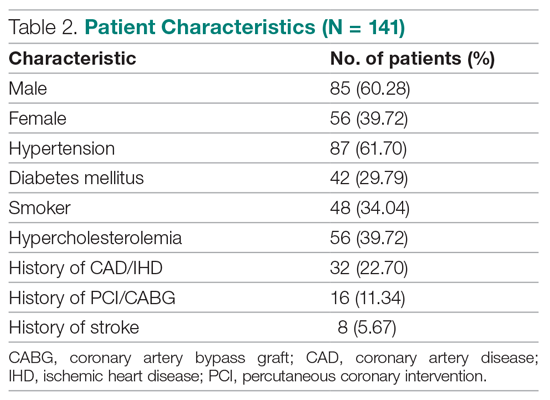
Primary outcomes
The risk stratification of the HEART score in chest pain patients and the incidence of 6-week MACE are outlined in Table 3
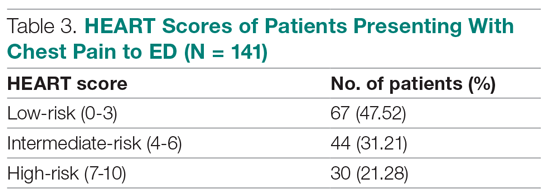
The distribution of the HEART score’s 5 elements in the groups with or without MACE endpoints is shown in Table 5. Notice the significant differences between the groups. A follow-up phone call was made within 6 weeks after the presentation to the ED to assess the patient’s progress. The 6-week follow-up call data are included in Table 6.
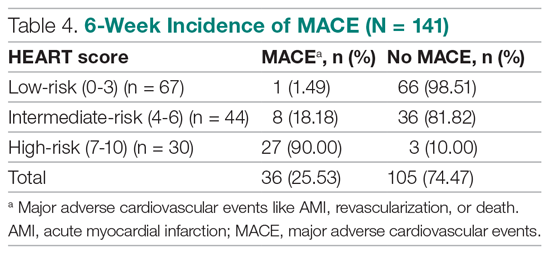
Of 141 patients, 36 patients (25.53%) were diagnosed with MACE within 6 weeks of presentation.
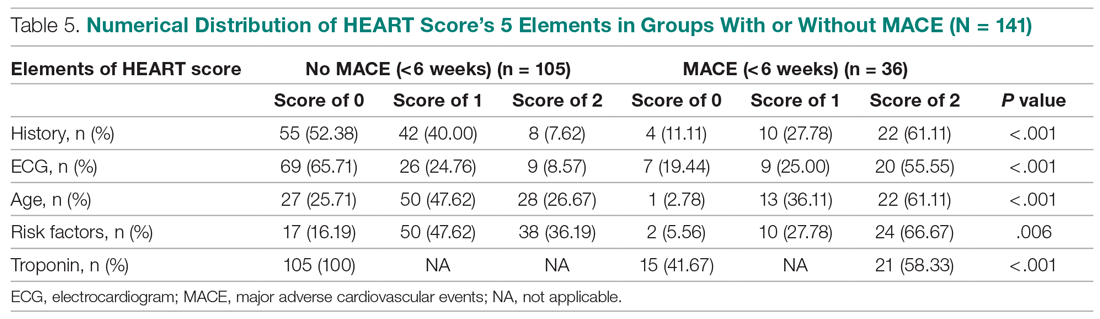
Myocardial infarction—An AMI was diagnosed in 24 of the 141 patients (17.02%). Twenty-one of those already had positive markers on admission (apparently, these AMI had started before their arrival to the emergency room). One AMI occurred 2 days after admission in a 66-year-old male, and another occurred 10 days after discharge. A further AMI occurred 2 weeks after discharge. All 3 patients belonged to the intermediate-risk group.
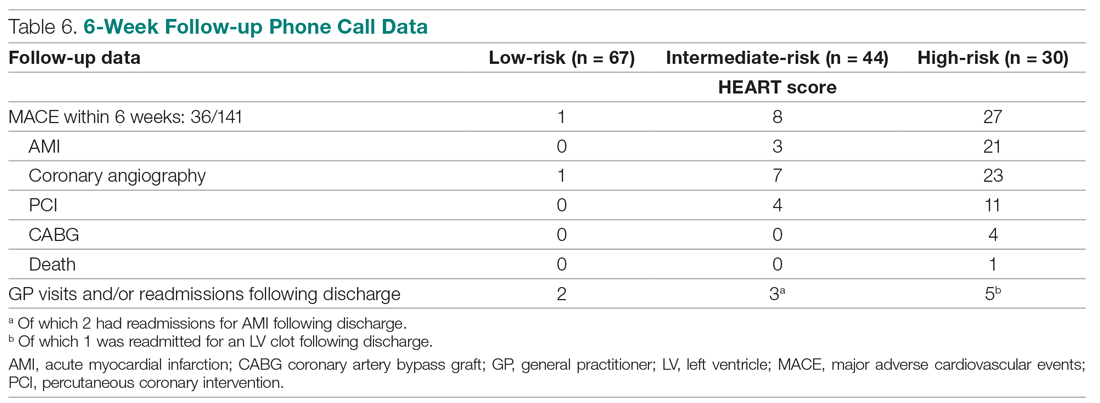
Revascularization—Coronary angiography was performed in 31 of 141 patients (21.99%). Revascularization was performed in 19 patients (13.48%), of which 15 were PCIs (10.64%) and 4 were CABGs (2.84%).
Mortality—One patient died from the study population. He was a 72-year-old male who died 14 days after admission. He had a HEART score of 8.
Among the 67 low-risk patients:
- MACE: Coronary angiography was performed in 1 patient (1.49%). Among the 67 patients in the low-risk category, there was no cases of AMI or deaths. The remaining 66 patients (98.51%) had an uneventful recovery following discharge.
- General practitioner (GP) visits/readmissions following discharge: Two of 67 patients (2.99%) had GP visits following discharge, of which 1 was uneventful. The other patient, a 64-year-old male, was readmitted due to a recurrent history of chest pain and underwent coronary angiography.
Among the 44 intermediate-risk patients:
- MACE: Of the 7 of 44 patients (15.91%) who had coronary angiography, 3 patients (6.82%) had AMI, of which 1 occurred 2 days after admission in a 66-year-old male. Two patients had AMI following discharge. There were no deaths. Overall, 42 of 44 patients (95.55%) had an uneventful recovery following discharge.
- GP visits/readmissions following discharge: Three of 44 patients (6.82%) had repeated visits following discharge. One was a GP visit that was uneventful. The remaining 2 patients were diagnosed with AMI and readmitted following discharge. One AMI occurred 10 days after discharge in a patient with a HEART score of 6; another occurred 2 weeks after discharge in a patient with a HEART score of 5.
Among the 30 high-risk patients:
- MACE: Twenty-three of 30 patients (76.67%) underwent coronary angiography. One patient died 5 days after discharge. The patient had a HEART score of 8. Most patients however, had an uneventful recovery following discharge (28, 93.33%).
- GP visits/readmissions following discharge: Five of 30 patients (16.67%) had repeated visits following discharge. Two were uneventful. Two patients had a history of recurrent chest pain that resolved on Sorbitrate. One patient was readmitted 2 weeks following discharge due to a complication: a left ventricular clot was found. The patient had a HEART score of 10.
Secondary outcome—Overall, 140 of 141 patients were discharged. One patient died: a 72-year-old male with a HEART score of 8.
Feasibility—To determine the ease and feasibility of performing a HEART score in chest pain patients presenting to the ED, a survey was distributed to the internal medicine physicians in the ED. In the survey, the Likert scale was used to rate the ease of utilizing the HEART score and whether the physicians found it feasible to use it for risk stratification of their chest pain patients. A total of 12 of 15 respondents (80%) found it “easy” to use. Of the remaining 3 respondents, 2 (13.33%) rated the HEART score “very easy” to use, while 1 (6.66%) considered it “difficult” to work with. None of the respondents said that it was not feasible to perform a HEART score in the ED.
Risk factors for reaching an endpoint:
We compared risk profiles between the patient groups with and without an endpoint. The group of patients with MACE were older and had a higher proportion of males than the group of patients without MACE. Moreover, they also had a higher prevalence of hypertension, type 2 diabetes mellitus, smoking, hypercholesterolemia, prior history of PCI/CABG, and history of stroke. These also showed a significant association with MACE. Obesity was not included in our risk factors as we did not have data collected to measure body mass index. Results are represented in Table 7.
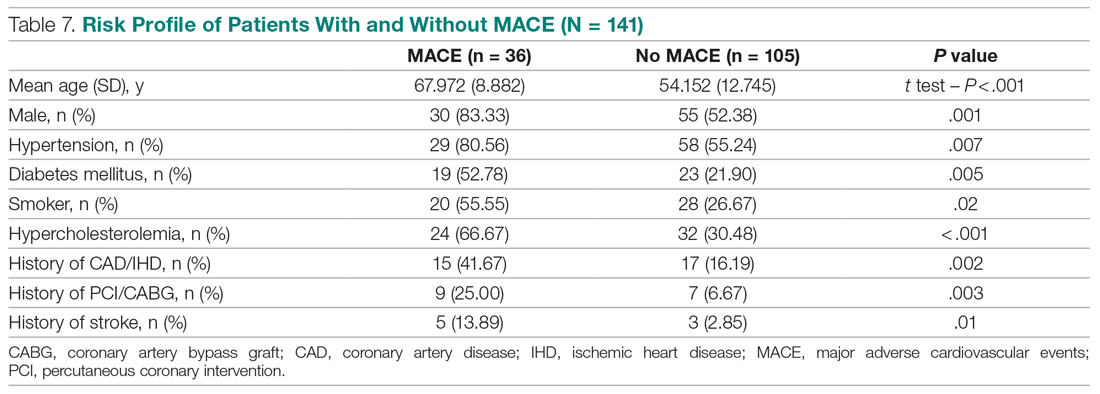
Discussion
Our study described a patient population presenting to an ED with chest pain as their primary complaint. The results of this prospective study confirm that the HEART score is an excellent system to triage chest pain patients. It provides the clinician with a reliable predictor of the outcome (MACE) after the patient’s arrival, based on available clinical data and in a resource-limited setting like ours.
Cardiovascular epidemiology studies indicate that this has become a significant public health problem in India.1 Several risk scores for ACS have been published in European and American guidelines. However, in the Indian population, minimal data are available on utilization of such a triage score (HEART score) in chest pain patients in the ED in a resource-limited setting, to the best of our knowledge. In India, only 1 such study is reported,15 at the Sundaram Medical Foundation, a 170-bed community hospital in Chennai. In this study, 13 of 14 patients (92.86%) with a high HEART score had MACE, indicating a sensitivity of 92.86%; in the 44 patients with a low HEART score, 1 patient (2.22%) had MACE, indicating a specificity of 97.78%; and in the 28 patients with a moderate HEART score, 12 patients (42.86%) had MACE.
In looking for the optimal risk-stratifying system for chest pain patients, we analyzed the HEART score. The first study on the HEART score was done Backus et al, proving that the HEART score is an easy, quick, and reliable predictor of outcomes in chest pain patients.10 The HEART score had good discriminatory power, too. The C statistic for the HEART score for ACS occurrence shows a value of 0.83. This signifies a good-to-excellent ability to stratify all-cause chest pain patients in the ED for their risk of MACE. The application of the HEART score to our patient population demonstrated that the majority of the patients belonged to the low-risk category, as reported in the first cohort study that applied the HEART score.8 The relationship between the HEART score category and occurrence of MACE within 6 weeks showed a curve with 3 different patterns, corresponding to the 3 risk categories defined in the literature.11,12 The risk stratification of chest pain patients using the 3 categories (0-3, 4-6, 7-10) identified MACE with an incidence similar to the multicenter study of Backus et al,10,11 but with a greater risk of MACE in the high-risk category (Figure).
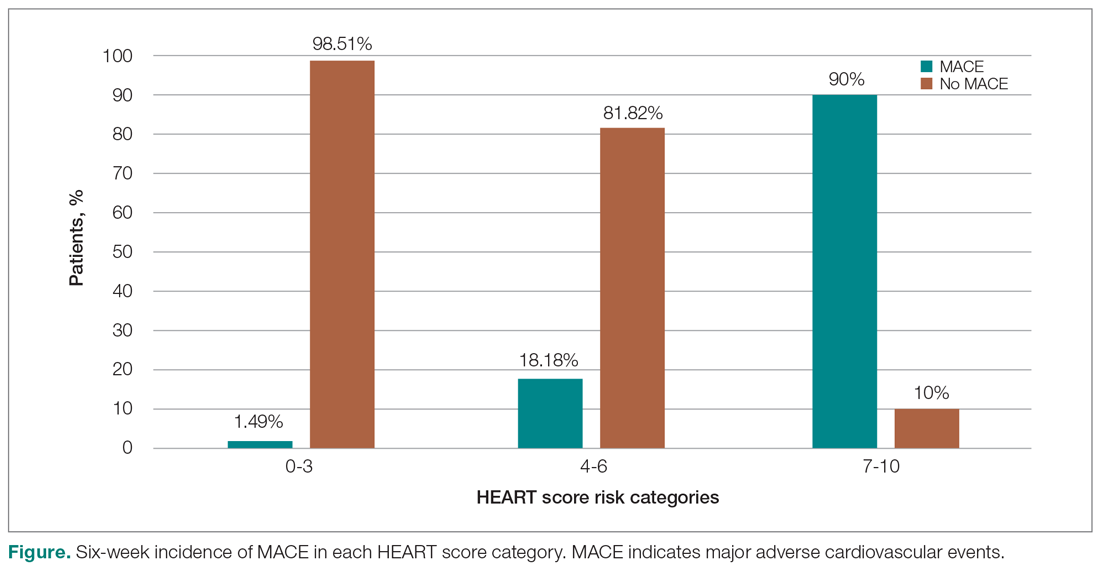
Thus, our study confirmed the utility of the HEART score categories to predict the 6-week incidence of MACE. The sensitivity, specificity, and positive and negative predictive values for the established cut-off scores of 4 and 7 are shown in Table 8. The patients in the low-risk category, corresponding to a score < 4, had a very high negative predictive value, thus identifying a small-risk population. The patients in the high-risk category (score ≥ 7) showed a high positive predictive value, allowing the identification of a high-risk population, even in patients with more atypical presentations. Therefore, the HEART score may help clinicians to make accurate management choices by being a strong predictor of both event-free survival and potentially life-threatening cardiac events.11,12
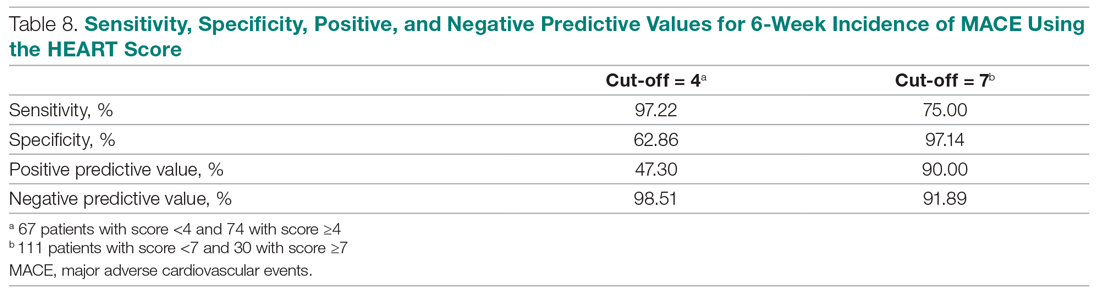
Our study tested the efficacy of the HEART score pathway in helping clinicians make smart diagnostic and therapeutic choices. It confirmed that the HEART score was accurate in predicting the short-term incidence of MACE, thus stratifying patients according to their risk severity. In our study, 67 of 141 patients (47.52%) had low-risk HEART scores, and we found the 6-week incidence of MACE to be 1.49%. We omitted the diagnostic and treatment evaluation for patients in the low-risk category and moved onto discharge. Overall, 66 of 67 patients (98.51%) in the low-risk category had an uneventful recovery following discharge. Only 2 of 67 these patients (2.99%) of patients had health care utilization following discharge. Therefore, extrapolation based on results demonstrates reduced health care utilization. Previous studies have shown similar results.9,12,14,16 For instance, in a prospective study conducted in the Netherlands, low-risk patients representing 36.4% of the total were found to have a low MACE rate (1.7%).9 These low-risk patients were categorized as appropriate and safe for ED discharge without additional cardiac evaluation or inpatient admission.9 Another retrospective study in Portugal,12 and one in Chennai, India,15 found the 6-week incidence of MACE to be 2.00% and 2.22%, respectively. The results of the first HEART Pathway Randomized Control Trial14 showed that the HEART score pathway reduces health care utilization (cardiac testing, hospitalization, and hospital length of stay). The study also showed that these gains occurred without any of the patients that were identified for early discharge, suffering from MACE at 30 days, or secondary increase in cardiac-related hospitalizations. Similar results were obtained by a randomized trial conducted in North Carolina17 that also demonstrated a reduction in objective cardiac testing, a doubling of the rate of early discharge from the ED, and a reduced length of stay by half a day. Another study using a modified HEART score also demonstrated that when low-risk patients are evaluated with cardiac testing, the likelihood for false positives is high.16 Hoffman et al also reported that patients randomized to coronary computed tomographic angiography (CCTA) received > 2.5 times more radiation exposure.16 Thus, low-risk patients may be safely discharged without the need for stress testing or CCTA.
In our study, 30 out of 141 patients (21.28%) had high-risk HEART scores (7-10), and we found the 6-week incidence of MACE to be 90%. Based on the pathway leading to inpatient admission and intensive treatment, 23 of 30 patients (76.67%) patients in our study underwent coronary angiography and further therapeutic treatment. In the high-risk category, 28 of 30 patients (93.33%) patients had an uneventful recovery following discharge. Previous studies have shown similar results. A retrospective study in Portugal showed that 76.9% of the high-risk patients had a 6-week incidence of MACE.12 In a study in the Netherlands,9 72.7% of high-risk patients had a 6-week incidence of MACE. Therefore, a HEART score of ≥ 7 in patients implies early aggressive treatment, including invasive strategies, when necessary, without noninvasive treatment preceding it.8
In terms of intermediate risk, in our study 44 of 141 patients (31.21%) patients had an intermediate-risk HEART score (4-6), and we found the 6-week incidence of MACE to be 18.18%. Based on the pathway, they were kept in the observation ward on admission. In our study, 7 of 44 patients (15.91%) underwent coronary angiography and further treatment; 42 of 44 patients (95.55%) had an uneventful recovery following discharge. In a prospective study in the Netherlands, 46.1% of patients with an intermediate score had a 6-week MACE incidence of 16.6%.10 Similarly, in another retrospective study in Portugal, the incidence of 6-week MACE in intermediate-risk patients (36.7%) was found to be 15.6%.12 Therefore, in patients with a HEART score of 4-6 points, immediate discharge is not an option, as this figure indicates a risk of 18.18% for an adverse outcome. These patients should be admitted for clinical observation, treated as an ACS awaiting final diagnosis, and subjected to noninvasive investigations, such as repeated troponin. Using the HEART score as guidance in the treatment of chest pain patients will benefit patients on both sides of the spectrum.11,12
Our sample presented a male predominance, a wide range of age, and a mean age similar to that of previous studies.12.16 Some risk factors, we found, can increase significantly the odds of chest pain being of cardiovascular origin, such as male gender, smoking, hypertension, type 2 diabetes mellitus, and hypercholesterolemia. Other studies also reported similar findings.8,12,16 Risk factors for premature CHD have been quantified in the case-control INTERHEART study.1 In the INTERHEART study, 8 common risk factors explained > 90% of AMIs in South Asian and Indian patients. The risk factors include dyslipidemia, smoking or tobacco use, known hypertension, known diabetes, abdominal obesity, physical inactivity, low fruit and vegetable intake, and psychosocial stress.1 Regarding the feasibility of treating physicians using the HEART score in the ED, we observed that, based on the Likert scale, 80% of survey respondents found it easy to use, and 100% found it feasible in the ED.
However, there were certain limitations to our study. It involved a single academic medical center and a small sample size, which limit generalizability of the findings. In addition, troponin levels are not calculated at our institution, as it is a resource-limited setting; therefore, we used a positive and negative as +2 and 0, respectively.
Conclusion
The HEART score provides the clinician with a quick and reliable predictor of outcome of patients with chest pain after arrival to the ED and can be used for triage. For patients with low HEART scores (0-3), short-term MACE can be excluded with greater than 98% certainty. In these patients, one may consider reserved treatment and discharge policies that may also reduce health care utilization. In patients with high HEART scores (7-10), the high risk of MACE (90%) may indicate early aggressive treatment, including invasive strategies, when necessary. Therefore, the HEART score may help clinicians make accurate management choices by being a strong predictor of both event-free survival and potentially life-threatening cardiac events. Age, gender, and cardiovascular risk factors may also be considered in the assessment of patients. This study confirmed the utility of the HEART score categories to predict the 6-week incidence of MACE.
Corresponding author: Smrati Bajpai Tiwari, MD, DNB, FAIMER, Department of Medicine, Seth Gordhandas Sunderdas Medical College and King Edward Memorial Hospital, Acharya Donde Marg, Parel, Mumbai 400 012, Maharashtra, India; [email protected].
Financial disclosures: None.
1. Gupta R, Mohan I, Narula J. Trends in coronary heart disease epidemiology in India. Ann Glob Health. 2016;82:307-315.
2. World Health Organization. Global status report on non-communicable diseases 2014. Accessed June 22, 2021. https://apps.who.int/iris/bitstream/handle/10665/148114/9789241564854_eng.pdf
3. Fuster V, Kelly BB, eds. Promoting Cardiovascular Health in the Developing World: A Critical Challenge to Achieve Global Health. Institutes of Medicine; 2010.
4. Krishnan MN. Coronary heart disease and risk factors in India—on the brink of an epidemic. Indian Heart J. 2012;64:364-367.
5. Prabhakaran D, Jeemon P, Roy A. Cardiovascular diseases in India: current epidemiology and future directions. Circulation. 2016;133:1605-1620.
6. Aeri B, Chauhan S. The rising incidence of cardiovascular diseases in India: assessing its economic impact. J Prev Cardiol. 2015;4:735-740.
7. Pednekar M, Gupta R, Gupta PC. Illiteracy, low educational status and cardiovascular mortality in India. BMC Public Health. 2011;11:567.
8. Six AJ, Backus BE, Kelder JC. Chest pain in the emergency room: value of the HEART score. Neth Heart J. 2008;16:191-196.
9. Backus BE, Six AJ, Kelder JC, et al. A prospective validation of the HEART score for chest pain patients at the emergency department. Int J Cardiol. 2013;168;2153-2158.
10. Backus BE, Six AJ, Kelder JC, et al. Chest pain in the emergency room: a multicenter validation of the HEART score. Crit Pathw Cardiol. 2010;9:164-169.
11. Backus BE, Six AJ, Kelder JH, et al. Risk scores for patients with chest pain: evaluation in the emergency department. Curr Cardiol Rev. 2011;7:2-8.
12. Leite L, Baptista R, Leitão J, et al. Chest pain in the emergency department: risk stratification with Manchester triage system and HEART score. BMC Cardiovasc Disord. 2015;15:48.
13. Thygesen K, Alpert JS, Jaffe AS, et al. Fourth Universal Definition of Myocardial Infarction. Circulation. 2018;138:e618-e651.
14. Mahler SA, Riley RF, Hiestand BC, et al. The HEART Pathway randomized trial: identifying emergency department patients with acute chest pain for early discharge. Circ Cardiovasc Qual Outcomes. 2015;8:195-203.
15. Natarajan B, Mallick P, Thangalvadi TA, Rajavelu P. Validation of the HEART score in Indian population. Int J Emerg Med. 2015,8(suppl 1):P5.
16. McCord J, Cabrera R, Lindahl B, et al. Prognostic utility of a modified HEART score in chest pain patients in the emergency department. Circ Cardiovasc Qual Outcomes. 2017;10:e003101.
17. Mahler SA, Miller CD, Hollander JE, et al. Identifying patients for early discharge: performance of decision rules among patients with acute chest pain. Int J Cardiol. 2012;168:795-802.
From the Department of Internal Medicine, Mount Sinai Health System, Icahn School of Medicine at Mount Sinai, New York, NY (Dr. Gandhi), and the School of Medicine, Seth Gordhandas Sunderdas Medical College, and King Edward Memorial Hospital, Mumbai, India (Drs. Gandhi and Tiwari).
Objective: Calculation of HEART score to (1) stratify patients as low-risk, intermediate-risk, high-risk, and to predict the short-term incidence of major adverse cardiovascular events (MACE), and (2) demonstrate feasibility of HEART score in our local settings.
Design: A prospective cohort study of patients with a chief complaint of chest pain concerning for acute coronary syndrome.
Setting: Participants were recruited from the emergency department (ED) of King Edward Memorial Hospital, a tertiary care academic medical center and a resource-limited setting in Mumbai, India.
Participants: We evaluated 141 patients aged 18 years and older presenting to the ED and stratified them using the HEART score. To assess patients’ progress, a follow-up phone call was made within 6 weeks after presentation to the ED.
Measurements: The primary outcomes were a risk stratification, 6-week occurrence of MACE, and performance of unscheduled revascularization or stress testing. The secondary outcomes were discharge or death.
Results: The 141 participants were stratified into low-risk, intermediate-risk, and high-risk groups: 67 (47.52%), 44 (31.21%), and 30 (21.28%), respectively. The 6-week incidence of MACE in each category was 1.49%, 18.18%, and 90%, respectively. An acute myocardial infarction was diagnosed in 24 patients (17.02%), 15 patients (10.64%) underwent percutaneous coronary intervention (PCI), and 4 patients (2.84%) underwent coronary artery bypass graft (CABG). Overall, 98.5% of low-risk patients and 93.33% of high-risk patients had an uneventful recovery following discharge; therefore, extrapolation based on results demonstrated reduced health care utilization. All the survey respondents found the HEART score to be feasible. The patient characteristics and risk profile of the patients with and without MACE demonstrated that: patients with MACE were older and had a higher proportion of males, hypertension, type 2 diabetes mellitus, smoking, hypercholesterolemia, prior history of PCI/CABG, and history of stroke.
Conclusion: The HEART score seems to be a useful tool for risk stratification and a reliable predictor of outcomes in chest pain patients and can therefore be used for triage.
Keywords: chest pain; emergency department; HEART score; acute coronary syndrome; major adverse cardiac events; myocardial infarction; revascularization.
Cardiovascular diseases (CVDs), especially coronary heart disease (CHD), have epidemic proportions worldwide. Globally, in 2012, CVD led to 17.5 million deaths,1,2 with more than 75% of them occurring in developing countries. In contrast to developed countries, where mortality from CHD is rapidly declining, it is increasing in developing countries.1,3 Current estimates from epidemiologic studies from various parts of India indicate the prevalence of CHD in India to be between 7% and 13% in urban populations and 2% and 7% in rural populations.4
Premature mortality in terms of years of life lost because of CVD in India increased by 59% over a 20-year span, from 23.2 million in 1990 to 37 million in 2010.5 Studies conducted in Mumbai (Mumbai Cohort Study) reported very high CVD mortality rates, approaching 500 per 100 000 for men and 250 per 100 000 for women.6,7 However, to the best of our knowledge, in the Indian population, there are minimal data on utilization of a triage score, such as the HEART score, in chest pain patients in the emergency department (ED) in a resource-limited setting.
The most common reason for admitting patients to the ED is chest pain.8 There are various cardiac and noncardiac etiologies of chest pain presentation. Acute coronary syndrome (ACS) needs to be ruled out first in every patient presenting with chest pain. However, 80% of patients with ACS have no clear diagnostic features on presentation.9 The timely diagnosis and treatment of patients with ACS improves their prognosis. Therefore, clinicians tend to start each patient on ACS treatment to reduce the risk, which often leads to increased costs due to unnecessary, time-consuming diagnostic procedures that may place burdens on both the health care system and the patient.10
Several risk-stratifying tools have been developed in the last few years. Both the GRACE and TIMI risk scores have been designed for risk stratification of patients with proven ACS and not for the chest pain population at the ED.11 Some of these tools are applicable to patients with all types of chest pain presenting to the ED, such as the Manchester Triage System. Other, more selective systems are devoted to the risk stratification of suspected ACS in the ED. One is the HEART score.12
The first study on the HEART score—an acronym that stands for History, Electrocardiogram, Age, Risk factors, and Troponin—was done by Backus et al, who proved that the HEART score is an easy, quick, and reliable predictor of outcomes in chest pain patients.10 The HEART score predicts the short-term incidence of major adverse cardiac events (MACE), which allows clinicians to stratify patients as low-risk, intermediate-risk, and high-risk and to guide their clinical decision-making accordingly. It was developed to provide clinicians with a simple, reliable predictor of cardiac risk on the basis of the lowest score of 0 (very low-risk) up to a score of 10 (very high-risk).
We studied the clinical performance of the HEART score in patients with chest pain, focusing on the efficacy and safety of rapidly identifying patients at risk of MACE. We aimed to determine (1) whether the HEART score is a reliable predictor of outcomes of chest pain patients presenting to the ED; (2) whether the score is feasible in our local settings; and (3) whether it describes the risk profile of patients with and without MACE.
Methods
Setting
Participants were recruited from the ED of King Edward Memorial Hospital, a municipal teaching hospital in Mumbai. The study institute is a tertiary care academic medical center located in Parel, Mumbai, Maharashtra, and is a resource-limited setting serving urban, suburban, and rural populations. Participants requiring urgent attention are first seen by a casualty officer and then referred to the emergency ward. Here, the physician on duty evaluates them and decides on admission to the various wards, like the general ward, medical intensive care unit (ICU), coronary care unit (CCU), etc. The specialist’s opinion may also be obtained before admission. Critically ill patients are initially admitted to the emergency ward and stabilized before being shifted to other areas of the hospital.
Participants
Patients aged 18 years and older presenting with symptoms of acute chest pain or suspected ACS were stratified by priority using the chest pain scoring system—the HEART score. Only patients presenting to the ED were eligible for the study. Informed consent from the patient or next of kin was mandatory for participation in the study.
Patients were determined ineligible for the following reasons: a clear cause for chest pain other than ACS (eg, trauma, diagnosed aortic dissection), persisting or recurrent chest pain caused by rheumatic diseases or cancer (a terminal illness), pregnancy, unable or unwilling to provide informed consent, or incomplete data.
Study design
We conducted a
We conducted our study to determine the importance of calculating the HEART score in each patient, which will help to correctly place them into low-, intermediate-, and high-risk groups for clinically important, irreversible adverse cardiac events and guide the clinical decision-making. Patients with low risk will avoid costly tests and hospital admissions, thus decreasing the cost of treatment and ensuring timely discharge from the ED. Patients with high risk will be treated immediately, to possibly prevent a life-threatening, ACS-related incident. Thus, the HEART score will serve as a quick and reliable predictor of outcomes in chest pain patients and help clinicians to make accurate diagnostic and therapeutic choices in uncertain situations.
HEART score
The total number of points for History, Electrocardiogram (ECG), Age, Risk factors, and Troponin was noted as the HEART score (Table 1).

For this study, the patient’s history and ECGs were interpreted by internal medicine attending physicians in the ED. The ECG taken in the emergency room was reviewed and classified, and a copy of the admission ECG was added to the file. The recommendation for patients with a HEART score in a particular range was evaluated. Notably, those with a score of 3 or lower led to a recommendation of reassurance and early discharge. Those with a HEART score in the intermediate range (4-6) were admitted to the hospital for further clinical observation and testing, whereas a high HEART score (7-10) led to admission for intensive monitoring and early intervention. In the analysis of HEART score data, we only used those patients having records for all 5 parameters, excluding patients without an ECG or troponin test.
Results
Myocardial infarction (MI) was defined based on Universal Definition of Myocardial Infarction.13 Coronary revascularization was defined as angioplasty with or without stent placement or coronary artery bypass surgery.14 Percutaneous coronary intervention (PCI) was defined as any therapeutic catheter intervention in the coronary arteries. Coronary artery bypass graft (CABG) surgery was defined as any cardiac surgery in which coronary arteries were operated on.
The primary outcomes in this study were the (1) risk stratification of chest pain patients into low-risk, intermediate-risk, and high-risk categories; (2) incidence of a MACE within 6 weeks of initial presentation. MACE consists of acute myocardial infarction (AMI), PCI, CABG, coronary angiography revealing procedurally correctable stenosis managed conservatively, and death due to any cause.
Our secondary outcomes were discharge or death due to any cause within 6 weeks after presentation.
Follow-up
Within 6 weeks after presentation to the ED, a follow-up phone call was placed to assess the patient’s progress. The follow-up focused on the endpoint of MACE, comprising all-cause death, MI, and revascularization. No patient was lost to follow-up.
Statistical analysis
We aimed to find a difference in the 6-week MACE between low-, intermediate-, and high-risk categories of the HEART score. The prevalence of CHD in India is 10%,4 and assuming an α of 0.05, we needed a sample of 141 patients from the ED patient population. Continuous variables were presented by mean (SD), and categorical variables as percentages. We used t test and the Mann-Whitney U test for comparison of means for continuous variables, χ2 for categorical variables, and Fisher’s exact test for comparison of the categorical variables. Results with P < .05 were considered statistically significant.
We evaluated 141 patients presenting to the ED with chest pain concerning for ACS during the study period, from July 2019 to October 2019.

Primary outcomes
The risk stratification of the HEART score in chest pain patients and the incidence of 6-week MACE are outlined in Table 3

The distribution of the HEART score’s 5 elements in the groups with or without MACE endpoints is shown in Table 5. Notice the significant differences between the groups. A follow-up phone call was made within 6 weeks after the presentation to the ED to assess the patient’s progress. The 6-week follow-up call data are included in Table 6.

Of 141 patients, 36 patients (25.53%) were diagnosed with MACE within 6 weeks of presentation.

Myocardial infarction—An AMI was diagnosed in 24 of the 141 patients (17.02%). Twenty-one of those already had positive markers on admission (apparently, these AMI had started before their arrival to the emergency room). One AMI occurred 2 days after admission in a 66-year-old male, and another occurred 10 days after discharge. A further AMI occurred 2 weeks after discharge. All 3 patients belonged to the intermediate-risk group.

Revascularization—Coronary angiography was performed in 31 of 141 patients (21.99%). Revascularization was performed in 19 patients (13.48%), of which 15 were PCIs (10.64%) and 4 were CABGs (2.84%).
Mortality—One patient died from the study population. He was a 72-year-old male who died 14 days after admission. He had a HEART score of 8.
Among the 67 low-risk patients:
- MACE: Coronary angiography was performed in 1 patient (1.49%). Among the 67 patients in the low-risk category, there was no cases of AMI or deaths. The remaining 66 patients (98.51%) had an uneventful recovery following discharge.
- General practitioner (GP) visits/readmissions following discharge: Two of 67 patients (2.99%) had GP visits following discharge, of which 1 was uneventful. The other patient, a 64-year-old male, was readmitted due to a recurrent history of chest pain and underwent coronary angiography.
Among the 44 intermediate-risk patients:
- MACE: Of the 7 of 44 patients (15.91%) who had coronary angiography, 3 patients (6.82%) had AMI, of which 1 occurred 2 days after admission in a 66-year-old male. Two patients had AMI following discharge. There were no deaths. Overall, 42 of 44 patients (95.55%) had an uneventful recovery following discharge.
- GP visits/readmissions following discharge: Three of 44 patients (6.82%) had repeated visits following discharge. One was a GP visit that was uneventful. The remaining 2 patients were diagnosed with AMI and readmitted following discharge. One AMI occurred 10 days after discharge in a patient with a HEART score of 6; another occurred 2 weeks after discharge in a patient with a HEART score of 5.
Among the 30 high-risk patients:
- MACE: Twenty-three of 30 patients (76.67%) underwent coronary angiography. One patient died 5 days after discharge. The patient had a HEART score of 8. Most patients however, had an uneventful recovery following discharge (28, 93.33%).
- GP visits/readmissions following discharge: Five of 30 patients (16.67%) had repeated visits following discharge. Two were uneventful. Two patients had a history of recurrent chest pain that resolved on Sorbitrate. One patient was readmitted 2 weeks following discharge due to a complication: a left ventricular clot was found. The patient had a HEART score of 10.
Secondary outcome—Overall, 140 of 141 patients were discharged. One patient died: a 72-year-old male with a HEART score of 8.
Feasibility—To determine the ease and feasibility of performing a HEART score in chest pain patients presenting to the ED, a survey was distributed to the internal medicine physicians in the ED. In the survey, the Likert scale was used to rate the ease of utilizing the HEART score and whether the physicians found it feasible to use it for risk stratification of their chest pain patients. A total of 12 of 15 respondents (80%) found it “easy” to use. Of the remaining 3 respondents, 2 (13.33%) rated the HEART score “very easy” to use, while 1 (6.66%) considered it “difficult” to work with. None of the respondents said that it was not feasible to perform a HEART score in the ED.
Risk factors for reaching an endpoint:
We compared risk profiles between the patient groups with and without an endpoint. The group of patients with MACE were older and had a higher proportion of males than the group of patients without MACE. Moreover, they also had a higher prevalence of hypertension, type 2 diabetes mellitus, smoking, hypercholesterolemia, prior history of PCI/CABG, and history of stroke. These also showed a significant association with MACE. Obesity was not included in our risk factors as we did not have data collected to measure body mass index. Results are represented in Table 7.

Discussion
Our study described a patient population presenting to an ED with chest pain as their primary complaint. The results of this prospective study confirm that the HEART score is an excellent system to triage chest pain patients. It provides the clinician with a reliable predictor of the outcome (MACE) after the patient’s arrival, based on available clinical data and in a resource-limited setting like ours.
Cardiovascular epidemiology studies indicate that this has become a significant public health problem in India.1 Several risk scores for ACS have been published in European and American guidelines. However, in the Indian population, minimal data are available on utilization of such a triage score (HEART score) in chest pain patients in the ED in a resource-limited setting, to the best of our knowledge. In India, only 1 such study is reported,15 at the Sundaram Medical Foundation, a 170-bed community hospital in Chennai. In this study, 13 of 14 patients (92.86%) with a high HEART score had MACE, indicating a sensitivity of 92.86%; in the 44 patients with a low HEART score, 1 patient (2.22%) had MACE, indicating a specificity of 97.78%; and in the 28 patients with a moderate HEART score, 12 patients (42.86%) had MACE.
In looking for the optimal risk-stratifying system for chest pain patients, we analyzed the HEART score. The first study on the HEART score was done Backus et al, proving that the HEART score is an easy, quick, and reliable predictor of outcomes in chest pain patients.10 The HEART score had good discriminatory power, too. The C statistic for the HEART score for ACS occurrence shows a value of 0.83. This signifies a good-to-excellent ability to stratify all-cause chest pain patients in the ED for their risk of MACE. The application of the HEART score to our patient population demonstrated that the majority of the patients belonged to the low-risk category, as reported in the first cohort study that applied the HEART score.8 The relationship between the HEART score category and occurrence of MACE within 6 weeks showed a curve with 3 different patterns, corresponding to the 3 risk categories defined in the literature.11,12 The risk stratification of chest pain patients using the 3 categories (0-3, 4-6, 7-10) identified MACE with an incidence similar to the multicenter study of Backus et al,10,11 but with a greater risk of MACE in the high-risk category (Figure).

Thus, our study confirmed the utility of the HEART score categories to predict the 6-week incidence of MACE. The sensitivity, specificity, and positive and negative predictive values for the established cut-off scores of 4 and 7 are shown in Table 8. The patients in the low-risk category, corresponding to a score < 4, had a very high negative predictive value, thus identifying a small-risk population. The patients in the high-risk category (score ≥ 7) showed a high positive predictive value, allowing the identification of a high-risk population, even in patients with more atypical presentations. Therefore, the HEART score may help clinicians to make accurate management choices by being a strong predictor of both event-free survival and potentially life-threatening cardiac events.11,12

Our study tested the efficacy of the HEART score pathway in helping clinicians make smart diagnostic and therapeutic choices. It confirmed that the HEART score was accurate in predicting the short-term incidence of MACE, thus stratifying patients according to their risk severity. In our study, 67 of 141 patients (47.52%) had low-risk HEART scores, and we found the 6-week incidence of MACE to be 1.49%. We omitted the diagnostic and treatment evaluation for patients in the low-risk category and moved onto discharge. Overall, 66 of 67 patients (98.51%) in the low-risk category had an uneventful recovery following discharge. Only 2 of 67 these patients (2.99%) of patients had health care utilization following discharge. Therefore, extrapolation based on results demonstrates reduced health care utilization. Previous studies have shown similar results.9,12,14,16 For instance, in a prospective study conducted in the Netherlands, low-risk patients representing 36.4% of the total were found to have a low MACE rate (1.7%).9 These low-risk patients were categorized as appropriate and safe for ED discharge without additional cardiac evaluation or inpatient admission.9 Another retrospective study in Portugal,12 and one in Chennai, India,15 found the 6-week incidence of MACE to be 2.00% and 2.22%, respectively. The results of the first HEART Pathway Randomized Control Trial14 showed that the HEART score pathway reduces health care utilization (cardiac testing, hospitalization, and hospital length of stay). The study also showed that these gains occurred without any of the patients that were identified for early discharge, suffering from MACE at 30 days, or secondary increase in cardiac-related hospitalizations. Similar results were obtained by a randomized trial conducted in North Carolina17 that also demonstrated a reduction in objective cardiac testing, a doubling of the rate of early discharge from the ED, and a reduced length of stay by half a day. Another study using a modified HEART score also demonstrated that when low-risk patients are evaluated with cardiac testing, the likelihood for false positives is high.16 Hoffman et al also reported that patients randomized to coronary computed tomographic angiography (CCTA) received > 2.5 times more radiation exposure.16 Thus, low-risk patients may be safely discharged without the need for stress testing or CCTA.
In our study, 30 out of 141 patients (21.28%) had high-risk HEART scores (7-10), and we found the 6-week incidence of MACE to be 90%. Based on the pathway leading to inpatient admission and intensive treatment, 23 of 30 patients (76.67%) patients in our study underwent coronary angiography and further therapeutic treatment. In the high-risk category, 28 of 30 patients (93.33%) patients had an uneventful recovery following discharge. Previous studies have shown similar results. A retrospective study in Portugal showed that 76.9% of the high-risk patients had a 6-week incidence of MACE.12 In a study in the Netherlands,9 72.7% of high-risk patients had a 6-week incidence of MACE. Therefore, a HEART score of ≥ 7 in patients implies early aggressive treatment, including invasive strategies, when necessary, without noninvasive treatment preceding it.8
In terms of intermediate risk, in our study 44 of 141 patients (31.21%) patients had an intermediate-risk HEART score (4-6), and we found the 6-week incidence of MACE to be 18.18%. Based on the pathway, they were kept in the observation ward on admission. In our study, 7 of 44 patients (15.91%) underwent coronary angiography and further treatment; 42 of 44 patients (95.55%) had an uneventful recovery following discharge. In a prospective study in the Netherlands, 46.1% of patients with an intermediate score had a 6-week MACE incidence of 16.6%.10 Similarly, in another retrospective study in Portugal, the incidence of 6-week MACE in intermediate-risk patients (36.7%) was found to be 15.6%.12 Therefore, in patients with a HEART score of 4-6 points, immediate discharge is not an option, as this figure indicates a risk of 18.18% for an adverse outcome. These patients should be admitted for clinical observation, treated as an ACS awaiting final diagnosis, and subjected to noninvasive investigations, such as repeated troponin. Using the HEART score as guidance in the treatment of chest pain patients will benefit patients on both sides of the spectrum.11,12
Our sample presented a male predominance, a wide range of age, and a mean age similar to that of previous studies.12.16 Some risk factors, we found, can increase significantly the odds of chest pain being of cardiovascular origin, such as male gender, smoking, hypertension, type 2 diabetes mellitus, and hypercholesterolemia. Other studies also reported similar findings.8,12,16 Risk factors for premature CHD have been quantified in the case-control INTERHEART study.1 In the INTERHEART study, 8 common risk factors explained > 90% of AMIs in South Asian and Indian patients. The risk factors include dyslipidemia, smoking or tobacco use, known hypertension, known diabetes, abdominal obesity, physical inactivity, low fruit and vegetable intake, and psychosocial stress.1 Regarding the feasibility of treating physicians using the HEART score in the ED, we observed that, based on the Likert scale, 80% of survey respondents found it easy to use, and 100% found it feasible in the ED.
However, there were certain limitations to our study. It involved a single academic medical center and a small sample size, which limit generalizability of the findings. In addition, troponin levels are not calculated at our institution, as it is a resource-limited setting; therefore, we used a positive and negative as +2 and 0, respectively.
Conclusion
The HEART score provides the clinician with a quick and reliable predictor of outcome of patients with chest pain after arrival to the ED and can be used for triage. For patients with low HEART scores (0-3), short-term MACE can be excluded with greater than 98% certainty. In these patients, one may consider reserved treatment and discharge policies that may also reduce health care utilization. In patients with high HEART scores (7-10), the high risk of MACE (90%) may indicate early aggressive treatment, including invasive strategies, when necessary. Therefore, the HEART score may help clinicians make accurate management choices by being a strong predictor of both event-free survival and potentially life-threatening cardiac events. Age, gender, and cardiovascular risk factors may also be considered in the assessment of patients. This study confirmed the utility of the HEART score categories to predict the 6-week incidence of MACE.
Corresponding author: Smrati Bajpai Tiwari, MD, DNB, FAIMER, Department of Medicine, Seth Gordhandas Sunderdas Medical College and King Edward Memorial Hospital, Acharya Donde Marg, Parel, Mumbai 400 012, Maharashtra, India; [email protected].
Financial disclosures: None.
From the Department of Internal Medicine, Mount Sinai Health System, Icahn School of Medicine at Mount Sinai, New York, NY (Dr. Gandhi), and the School of Medicine, Seth Gordhandas Sunderdas Medical College, and King Edward Memorial Hospital, Mumbai, India (Drs. Gandhi and Tiwari).
Objective: Calculation of HEART score to (1) stratify patients as low-risk, intermediate-risk, high-risk, and to predict the short-term incidence of major adverse cardiovascular events (MACE), and (2) demonstrate feasibility of HEART score in our local settings.
Design: A prospective cohort study of patients with a chief complaint of chest pain concerning for acute coronary syndrome.
Setting: Participants were recruited from the emergency department (ED) of King Edward Memorial Hospital, a tertiary care academic medical center and a resource-limited setting in Mumbai, India.
Participants: We evaluated 141 patients aged 18 years and older presenting to the ED and stratified them using the HEART score. To assess patients’ progress, a follow-up phone call was made within 6 weeks after presentation to the ED.
Measurements: The primary outcomes were a risk stratification, 6-week occurrence of MACE, and performance of unscheduled revascularization or stress testing. The secondary outcomes were discharge or death.
Results: The 141 participants were stratified into low-risk, intermediate-risk, and high-risk groups: 67 (47.52%), 44 (31.21%), and 30 (21.28%), respectively. The 6-week incidence of MACE in each category was 1.49%, 18.18%, and 90%, respectively. An acute myocardial infarction was diagnosed in 24 patients (17.02%), 15 patients (10.64%) underwent percutaneous coronary intervention (PCI), and 4 patients (2.84%) underwent coronary artery bypass graft (CABG). Overall, 98.5% of low-risk patients and 93.33% of high-risk patients had an uneventful recovery following discharge; therefore, extrapolation based on results demonstrated reduced health care utilization. All the survey respondents found the HEART score to be feasible. The patient characteristics and risk profile of the patients with and without MACE demonstrated that: patients with MACE were older and had a higher proportion of males, hypertension, type 2 diabetes mellitus, smoking, hypercholesterolemia, prior history of PCI/CABG, and history of stroke.
Conclusion: The HEART score seems to be a useful tool for risk stratification and a reliable predictor of outcomes in chest pain patients and can therefore be used for triage.
Keywords: chest pain; emergency department; HEART score; acute coronary syndrome; major adverse cardiac events; myocardial infarction; revascularization.
Cardiovascular diseases (CVDs), especially coronary heart disease (CHD), have epidemic proportions worldwide. Globally, in 2012, CVD led to 17.5 million deaths,1,2 with more than 75% of them occurring in developing countries. In contrast to developed countries, where mortality from CHD is rapidly declining, it is increasing in developing countries.1,3 Current estimates from epidemiologic studies from various parts of India indicate the prevalence of CHD in India to be between 7% and 13% in urban populations and 2% and 7% in rural populations.4
Premature mortality in terms of years of life lost because of CVD in India increased by 59% over a 20-year span, from 23.2 million in 1990 to 37 million in 2010.5 Studies conducted in Mumbai (Mumbai Cohort Study) reported very high CVD mortality rates, approaching 500 per 100 000 for men and 250 per 100 000 for women.6,7 However, to the best of our knowledge, in the Indian population, there are minimal data on utilization of a triage score, such as the HEART score, in chest pain patients in the emergency department (ED) in a resource-limited setting.
The most common reason for admitting patients to the ED is chest pain.8 There are various cardiac and noncardiac etiologies of chest pain presentation. Acute coronary syndrome (ACS) needs to be ruled out first in every patient presenting with chest pain. However, 80% of patients with ACS have no clear diagnostic features on presentation.9 The timely diagnosis and treatment of patients with ACS improves their prognosis. Therefore, clinicians tend to start each patient on ACS treatment to reduce the risk, which often leads to increased costs due to unnecessary, time-consuming diagnostic procedures that may place burdens on both the health care system and the patient.10
Several risk-stratifying tools have been developed in the last few years. Both the GRACE and TIMI risk scores have been designed for risk stratification of patients with proven ACS and not for the chest pain population at the ED.11 Some of these tools are applicable to patients with all types of chest pain presenting to the ED, such as the Manchester Triage System. Other, more selective systems are devoted to the risk stratification of suspected ACS in the ED. One is the HEART score.12
The first study on the HEART score—an acronym that stands for History, Electrocardiogram, Age, Risk factors, and Troponin—was done by Backus et al, who proved that the HEART score is an easy, quick, and reliable predictor of outcomes in chest pain patients.10 The HEART score predicts the short-term incidence of major adverse cardiac events (MACE), which allows clinicians to stratify patients as low-risk, intermediate-risk, and high-risk and to guide their clinical decision-making accordingly. It was developed to provide clinicians with a simple, reliable predictor of cardiac risk on the basis of the lowest score of 0 (very low-risk) up to a score of 10 (very high-risk).
We studied the clinical performance of the HEART score in patients with chest pain, focusing on the efficacy and safety of rapidly identifying patients at risk of MACE. We aimed to determine (1) whether the HEART score is a reliable predictor of outcomes of chest pain patients presenting to the ED; (2) whether the score is feasible in our local settings; and (3) whether it describes the risk profile of patients with and without MACE.
Methods
Setting
Participants were recruited from the ED of King Edward Memorial Hospital, a municipal teaching hospital in Mumbai. The study institute is a tertiary care academic medical center located in Parel, Mumbai, Maharashtra, and is a resource-limited setting serving urban, suburban, and rural populations. Participants requiring urgent attention are first seen by a casualty officer and then referred to the emergency ward. Here, the physician on duty evaluates them and decides on admission to the various wards, like the general ward, medical intensive care unit (ICU), coronary care unit (CCU), etc. The specialist’s opinion may also be obtained before admission. Critically ill patients are initially admitted to the emergency ward and stabilized before being shifted to other areas of the hospital.
Participants
Patients aged 18 years and older presenting with symptoms of acute chest pain or suspected ACS were stratified by priority using the chest pain scoring system—the HEART score. Only patients presenting to the ED were eligible for the study. Informed consent from the patient or next of kin was mandatory for participation in the study.
Patients were determined ineligible for the following reasons: a clear cause for chest pain other than ACS (eg, trauma, diagnosed aortic dissection), persisting or recurrent chest pain caused by rheumatic diseases or cancer (a terminal illness), pregnancy, unable or unwilling to provide informed consent, or incomplete data.
Study design
We conducted a
We conducted our study to determine the importance of calculating the HEART score in each patient, which will help to correctly place them into low-, intermediate-, and high-risk groups for clinically important, irreversible adverse cardiac events and guide the clinical decision-making. Patients with low risk will avoid costly tests and hospital admissions, thus decreasing the cost of treatment and ensuring timely discharge from the ED. Patients with high risk will be treated immediately, to possibly prevent a life-threatening, ACS-related incident. Thus, the HEART score will serve as a quick and reliable predictor of outcomes in chest pain patients and help clinicians to make accurate diagnostic and therapeutic choices in uncertain situations.
HEART score
The total number of points for History, Electrocardiogram (ECG), Age, Risk factors, and Troponin was noted as the HEART score (Table 1).

For this study, the patient’s history and ECGs were interpreted by internal medicine attending physicians in the ED. The ECG taken in the emergency room was reviewed and classified, and a copy of the admission ECG was added to the file. The recommendation for patients with a HEART score in a particular range was evaluated. Notably, those with a score of 3 or lower led to a recommendation of reassurance and early discharge. Those with a HEART score in the intermediate range (4-6) were admitted to the hospital for further clinical observation and testing, whereas a high HEART score (7-10) led to admission for intensive monitoring and early intervention. In the analysis of HEART score data, we only used those patients having records for all 5 parameters, excluding patients without an ECG or troponin test.
Results
Myocardial infarction (MI) was defined based on Universal Definition of Myocardial Infarction.13 Coronary revascularization was defined as angioplasty with or without stent placement or coronary artery bypass surgery.14 Percutaneous coronary intervention (PCI) was defined as any therapeutic catheter intervention in the coronary arteries. Coronary artery bypass graft (CABG) surgery was defined as any cardiac surgery in which coronary arteries were operated on.
The primary outcomes in this study were the (1) risk stratification of chest pain patients into low-risk, intermediate-risk, and high-risk categories; (2) incidence of a MACE within 6 weeks of initial presentation. MACE consists of acute myocardial infarction (AMI), PCI, CABG, coronary angiography revealing procedurally correctable stenosis managed conservatively, and death due to any cause.
Our secondary outcomes were discharge or death due to any cause within 6 weeks after presentation.
Follow-up
Within 6 weeks after presentation to the ED, a follow-up phone call was placed to assess the patient’s progress. The follow-up focused on the endpoint of MACE, comprising all-cause death, MI, and revascularization. No patient was lost to follow-up.
Statistical analysis
We aimed to find a difference in the 6-week MACE between low-, intermediate-, and high-risk categories of the HEART score. The prevalence of CHD in India is 10%,4 and assuming an α of 0.05, we needed a sample of 141 patients from the ED patient population. Continuous variables were presented by mean (SD), and categorical variables as percentages. We used t test and the Mann-Whitney U test for comparison of means for continuous variables, χ2 for categorical variables, and Fisher’s exact test for comparison of the categorical variables. Results with P < .05 were considered statistically significant.
We evaluated 141 patients presenting to the ED with chest pain concerning for ACS during the study period, from July 2019 to October 2019.

Primary outcomes
The risk stratification of the HEART score in chest pain patients and the incidence of 6-week MACE are outlined in Table 3

The distribution of the HEART score’s 5 elements in the groups with or without MACE endpoints is shown in Table 5. Notice the significant differences between the groups. A follow-up phone call was made within 6 weeks after the presentation to the ED to assess the patient’s progress. The 6-week follow-up call data are included in Table 6.

Of 141 patients, 36 patients (25.53%) were diagnosed with MACE within 6 weeks of presentation.

Myocardial infarction—An AMI was diagnosed in 24 of the 141 patients (17.02%). Twenty-one of those already had positive markers on admission (apparently, these AMI had started before their arrival to the emergency room). One AMI occurred 2 days after admission in a 66-year-old male, and another occurred 10 days after discharge. A further AMI occurred 2 weeks after discharge. All 3 patients belonged to the intermediate-risk group.

Revascularization—Coronary angiography was performed in 31 of 141 patients (21.99%). Revascularization was performed in 19 patients (13.48%), of which 15 were PCIs (10.64%) and 4 were CABGs (2.84%).
Mortality—One patient died from the study population. He was a 72-year-old male who died 14 days after admission. He had a HEART score of 8.
Among the 67 low-risk patients:
- MACE: Coronary angiography was performed in 1 patient (1.49%). Among the 67 patients in the low-risk category, there was no cases of AMI or deaths. The remaining 66 patients (98.51%) had an uneventful recovery following discharge.
- General practitioner (GP) visits/readmissions following discharge: Two of 67 patients (2.99%) had GP visits following discharge, of which 1 was uneventful. The other patient, a 64-year-old male, was readmitted due to a recurrent history of chest pain and underwent coronary angiography.
Among the 44 intermediate-risk patients:
- MACE: Of the 7 of 44 patients (15.91%) who had coronary angiography, 3 patients (6.82%) had AMI, of which 1 occurred 2 days after admission in a 66-year-old male. Two patients had AMI following discharge. There were no deaths. Overall, 42 of 44 patients (95.55%) had an uneventful recovery following discharge.
- GP visits/readmissions following discharge: Three of 44 patients (6.82%) had repeated visits following discharge. One was a GP visit that was uneventful. The remaining 2 patients were diagnosed with AMI and readmitted following discharge. One AMI occurred 10 days after discharge in a patient with a HEART score of 6; another occurred 2 weeks after discharge in a patient with a HEART score of 5.
Among the 30 high-risk patients:
- MACE: Twenty-three of 30 patients (76.67%) underwent coronary angiography. One patient died 5 days after discharge. The patient had a HEART score of 8. Most patients however, had an uneventful recovery following discharge (28, 93.33%).
- GP visits/readmissions following discharge: Five of 30 patients (16.67%) had repeated visits following discharge. Two were uneventful. Two patients had a history of recurrent chest pain that resolved on Sorbitrate. One patient was readmitted 2 weeks following discharge due to a complication: a left ventricular clot was found. The patient had a HEART score of 10.
Secondary outcome—Overall, 140 of 141 patients were discharged. One patient died: a 72-year-old male with a HEART score of 8.
Feasibility—To determine the ease and feasibility of performing a HEART score in chest pain patients presenting to the ED, a survey was distributed to the internal medicine physicians in the ED. In the survey, the Likert scale was used to rate the ease of utilizing the HEART score and whether the physicians found it feasible to use it for risk stratification of their chest pain patients. A total of 12 of 15 respondents (80%) found it “easy” to use. Of the remaining 3 respondents, 2 (13.33%) rated the HEART score “very easy” to use, while 1 (6.66%) considered it “difficult” to work with. None of the respondents said that it was not feasible to perform a HEART score in the ED.
Risk factors for reaching an endpoint:
We compared risk profiles between the patient groups with and without an endpoint. The group of patients with MACE were older and had a higher proportion of males than the group of patients without MACE. Moreover, they also had a higher prevalence of hypertension, type 2 diabetes mellitus, smoking, hypercholesterolemia, prior history of PCI/CABG, and history of stroke. These also showed a significant association with MACE. Obesity was not included in our risk factors as we did not have data collected to measure body mass index. Results are represented in Table 7.

Discussion
Our study described a patient population presenting to an ED with chest pain as their primary complaint. The results of this prospective study confirm that the HEART score is an excellent system to triage chest pain patients. It provides the clinician with a reliable predictor of the outcome (MACE) after the patient’s arrival, based on available clinical data and in a resource-limited setting like ours.
Cardiovascular epidemiology studies indicate that this has become a significant public health problem in India.1 Several risk scores for ACS have been published in European and American guidelines. However, in the Indian population, minimal data are available on utilization of such a triage score (HEART score) in chest pain patients in the ED in a resource-limited setting, to the best of our knowledge. In India, only 1 such study is reported,15 at the Sundaram Medical Foundation, a 170-bed community hospital in Chennai. In this study, 13 of 14 patients (92.86%) with a high HEART score had MACE, indicating a sensitivity of 92.86%; in the 44 patients with a low HEART score, 1 patient (2.22%) had MACE, indicating a specificity of 97.78%; and in the 28 patients with a moderate HEART score, 12 patients (42.86%) had MACE.
In looking for the optimal risk-stratifying system for chest pain patients, we analyzed the HEART score. The first study on the HEART score was done Backus et al, proving that the HEART score is an easy, quick, and reliable predictor of outcomes in chest pain patients.10 The HEART score had good discriminatory power, too. The C statistic for the HEART score for ACS occurrence shows a value of 0.83. This signifies a good-to-excellent ability to stratify all-cause chest pain patients in the ED for their risk of MACE. The application of the HEART score to our patient population demonstrated that the majority of the patients belonged to the low-risk category, as reported in the first cohort study that applied the HEART score.8 The relationship between the HEART score category and occurrence of MACE within 6 weeks showed a curve with 3 different patterns, corresponding to the 3 risk categories defined in the literature.11,12 The risk stratification of chest pain patients using the 3 categories (0-3, 4-6, 7-10) identified MACE with an incidence similar to the multicenter study of Backus et al,10,11 but with a greater risk of MACE in the high-risk category (Figure).

Thus, our study confirmed the utility of the HEART score categories to predict the 6-week incidence of MACE. The sensitivity, specificity, and positive and negative predictive values for the established cut-off scores of 4 and 7 are shown in Table 8. The patients in the low-risk category, corresponding to a score < 4, had a very high negative predictive value, thus identifying a small-risk population. The patients in the high-risk category (score ≥ 7) showed a high positive predictive value, allowing the identification of a high-risk population, even in patients with more atypical presentations. Therefore, the HEART score may help clinicians to make accurate management choices by being a strong predictor of both event-free survival and potentially life-threatening cardiac events.11,12

Our study tested the efficacy of the HEART score pathway in helping clinicians make smart diagnostic and therapeutic choices. It confirmed that the HEART score was accurate in predicting the short-term incidence of MACE, thus stratifying patients according to their risk severity. In our study, 67 of 141 patients (47.52%) had low-risk HEART scores, and we found the 6-week incidence of MACE to be 1.49%. We omitted the diagnostic and treatment evaluation for patients in the low-risk category and moved onto discharge. Overall, 66 of 67 patients (98.51%) in the low-risk category had an uneventful recovery following discharge. Only 2 of 67 these patients (2.99%) of patients had health care utilization following discharge. Therefore, extrapolation based on results demonstrates reduced health care utilization. Previous studies have shown similar results.9,12,14,16 For instance, in a prospective study conducted in the Netherlands, low-risk patients representing 36.4% of the total were found to have a low MACE rate (1.7%).9 These low-risk patients were categorized as appropriate and safe for ED discharge without additional cardiac evaluation or inpatient admission.9 Another retrospective study in Portugal,12 and one in Chennai, India,15 found the 6-week incidence of MACE to be 2.00% and 2.22%, respectively. The results of the first HEART Pathway Randomized Control Trial14 showed that the HEART score pathway reduces health care utilization (cardiac testing, hospitalization, and hospital length of stay). The study also showed that these gains occurred without any of the patients that were identified for early discharge, suffering from MACE at 30 days, or secondary increase in cardiac-related hospitalizations. Similar results were obtained by a randomized trial conducted in North Carolina17 that also demonstrated a reduction in objective cardiac testing, a doubling of the rate of early discharge from the ED, and a reduced length of stay by half a day. Another study using a modified HEART score also demonstrated that when low-risk patients are evaluated with cardiac testing, the likelihood for false positives is high.16 Hoffman et al also reported that patients randomized to coronary computed tomographic angiography (CCTA) received > 2.5 times more radiation exposure.16 Thus, low-risk patients may be safely discharged without the need for stress testing or CCTA.
In our study, 30 out of 141 patients (21.28%) had high-risk HEART scores (7-10), and we found the 6-week incidence of MACE to be 90%. Based on the pathway leading to inpatient admission and intensive treatment, 23 of 30 patients (76.67%) patients in our study underwent coronary angiography and further therapeutic treatment. In the high-risk category, 28 of 30 patients (93.33%) patients had an uneventful recovery following discharge. Previous studies have shown similar results. A retrospective study in Portugal showed that 76.9% of the high-risk patients had a 6-week incidence of MACE.12 In a study in the Netherlands,9 72.7% of high-risk patients had a 6-week incidence of MACE. Therefore, a HEART score of ≥ 7 in patients implies early aggressive treatment, including invasive strategies, when necessary, without noninvasive treatment preceding it.8
In terms of intermediate risk, in our study 44 of 141 patients (31.21%) patients had an intermediate-risk HEART score (4-6), and we found the 6-week incidence of MACE to be 18.18%. Based on the pathway, they were kept in the observation ward on admission. In our study, 7 of 44 patients (15.91%) underwent coronary angiography and further treatment; 42 of 44 patients (95.55%) had an uneventful recovery following discharge. In a prospective study in the Netherlands, 46.1% of patients with an intermediate score had a 6-week MACE incidence of 16.6%.10 Similarly, in another retrospective study in Portugal, the incidence of 6-week MACE in intermediate-risk patients (36.7%) was found to be 15.6%.12 Therefore, in patients with a HEART score of 4-6 points, immediate discharge is not an option, as this figure indicates a risk of 18.18% for an adverse outcome. These patients should be admitted for clinical observation, treated as an ACS awaiting final diagnosis, and subjected to noninvasive investigations, such as repeated troponin. Using the HEART score as guidance in the treatment of chest pain patients will benefit patients on both sides of the spectrum.11,12
Our sample presented a male predominance, a wide range of age, and a mean age similar to that of previous studies.12.16 Some risk factors, we found, can increase significantly the odds of chest pain being of cardiovascular origin, such as male gender, smoking, hypertension, type 2 diabetes mellitus, and hypercholesterolemia. Other studies also reported similar findings.8,12,16 Risk factors for premature CHD have been quantified in the case-control INTERHEART study.1 In the INTERHEART study, 8 common risk factors explained > 90% of AMIs in South Asian and Indian patients. The risk factors include dyslipidemia, smoking or tobacco use, known hypertension, known diabetes, abdominal obesity, physical inactivity, low fruit and vegetable intake, and psychosocial stress.1 Regarding the feasibility of treating physicians using the HEART score in the ED, we observed that, based on the Likert scale, 80% of survey respondents found it easy to use, and 100% found it feasible in the ED.
However, there were certain limitations to our study. It involved a single academic medical center and a small sample size, which limit generalizability of the findings. In addition, troponin levels are not calculated at our institution, as it is a resource-limited setting; therefore, we used a positive and negative as +2 and 0, respectively.
Conclusion
The HEART score provides the clinician with a quick and reliable predictor of outcome of patients with chest pain after arrival to the ED and can be used for triage. For patients with low HEART scores (0-3), short-term MACE can be excluded with greater than 98% certainty. In these patients, one may consider reserved treatment and discharge policies that may also reduce health care utilization. In patients with high HEART scores (7-10), the high risk of MACE (90%) may indicate early aggressive treatment, including invasive strategies, when necessary. Therefore, the HEART score may help clinicians make accurate management choices by being a strong predictor of both event-free survival and potentially life-threatening cardiac events. Age, gender, and cardiovascular risk factors may also be considered in the assessment of patients. This study confirmed the utility of the HEART score categories to predict the 6-week incidence of MACE.
Corresponding author: Smrati Bajpai Tiwari, MD, DNB, FAIMER, Department of Medicine, Seth Gordhandas Sunderdas Medical College and King Edward Memorial Hospital, Acharya Donde Marg, Parel, Mumbai 400 012, Maharashtra, India; [email protected].
Financial disclosures: None.
1. Gupta R, Mohan I, Narula J. Trends in coronary heart disease epidemiology in India. Ann Glob Health. 2016;82:307-315.
2. World Health Organization. Global status report on non-communicable diseases 2014. Accessed June 22, 2021. https://apps.who.int/iris/bitstream/handle/10665/148114/9789241564854_eng.pdf
3. Fuster V, Kelly BB, eds. Promoting Cardiovascular Health in the Developing World: A Critical Challenge to Achieve Global Health. Institutes of Medicine; 2010.
4. Krishnan MN. Coronary heart disease and risk factors in India—on the brink of an epidemic. Indian Heart J. 2012;64:364-367.
5. Prabhakaran D, Jeemon P, Roy A. Cardiovascular diseases in India: current epidemiology and future directions. Circulation. 2016;133:1605-1620.
6. Aeri B, Chauhan S. The rising incidence of cardiovascular diseases in India: assessing its economic impact. J Prev Cardiol. 2015;4:735-740.
7. Pednekar M, Gupta R, Gupta PC. Illiteracy, low educational status and cardiovascular mortality in India. BMC Public Health. 2011;11:567.
8. Six AJ, Backus BE, Kelder JC. Chest pain in the emergency room: value of the HEART score. Neth Heart J. 2008;16:191-196.
9. Backus BE, Six AJ, Kelder JC, et al. A prospective validation of the HEART score for chest pain patients at the emergency department. Int J Cardiol. 2013;168;2153-2158.
10. Backus BE, Six AJ, Kelder JC, et al. Chest pain in the emergency room: a multicenter validation of the HEART score. Crit Pathw Cardiol. 2010;9:164-169.
11. Backus BE, Six AJ, Kelder JH, et al. Risk scores for patients with chest pain: evaluation in the emergency department. Curr Cardiol Rev. 2011;7:2-8.
12. Leite L, Baptista R, Leitão J, et al. Chest pain in the emergency department: risk stratification with Manchester triage system and HEART score. BMC Cardiovasc Disord. 2015;15:48.
13. Thygesen K, Alpert JS, Jaffe AS, et al. Fourth Universal Definition of Myocardial Infarction. Circulation. 2018;138:e618-e651.
14. Mahler SA, Riley RF, Hiestand BC, et al. The HEART Pathway randomized trial: identifying emergency department patients with acute chest pain for early discharge. Circ Cardiovasc Qual Outcomes. 2015;8:195-203.
15. Natarajan B, Mallick P, Thangalvadi TA, Rajavelu P. Validation of the HEART score in Indian population. Int J Emerg Med. 2015,8(suppl 1):P5.
16. McCord J, Cabrera R, Lindahl B, et al. Prognostic utility of a modified HEART score in chest pain patients in the emergency department. Circ Cardiovasc Qual Outcomes. 2017;10:e003101.
17. Mahler SA, Miller CD, Hollander JE, et al. Identifying patients for early discharge: performance of decision rules among patients with acute chest pain. Int J Cardiol. 2012;168:795-802.
1. Gupta R, Mohan I, Narula J. Trends in coronary heart disease epidemiology in India. Ann Glob Health. 2016;82:307-315.
2. World Health Organization. Global status report on non-communicable diseases 2014. Accessed June 22, 2021. https://apps.who.int/iris/bitstream/handle/10665/148114/9789241564854_eng.pdf
3. Fuster V, Kelly BB, eds. Promoting Cardiovascular Health in the Developing World: A Critical Challenge to Achieve Global Health. Institutes of Medicine; 2010.
4. Krishnan MN. Coronary heart disease and risk factors in India—on the brink of an epidemic. Indian Heart J. 2012;64:364-367.
5. Prabhakaran D, Jeemon P, Roy A. Cardiovascular diseases in India: current epidemiology and future directions. Circulation. 2016;133:1605-1620.
6. Aeri B, Chauhan S. The rising incidence of cardiovascular diseases in India: assessing its economic impact. J Prev Cardiol. 2015;4:735-740.
7. Pednekar M, Gupta R, Gupta PC. Illiteracy, low educational status and cardiovascular mortality in India. BMC Public Health. 2011;11:567.
8. Six AJ, Backus BE, Kelder JC. Chest pain in the emergency room: value of the HEART score. Neth Heart J. 2008;16:191-196.
9. Backus BE, Six AJ, Kelder JC, et al. A prospective validation of the HEART score for chest pain patients at the emergency department. Int J Cardiol. 2013;168;2153-2158.
10. Backus BE, Six AJ, Kelder JC, et al. Chest pain in the emergency room: a multicenter validation of the HEART score. Crit Pathw Cardiol. 2010;9:164-169.
11. Backus BE, Six AJ, Kelder JH, et al. Risk scores for patients with chest pain: evaluation in the emergency department. Curr Cardiol Rev. 2011;7:2-8.
12. Leite L, Baptista R, Leitão J, et al. Chest pain in the emergency department: risk stratification with Manchester triage system and HEART score. BMC Cardiovasc Disord. 2015;15:48.
13. Thygesen K, Alpert JS, Jaffe AS, et al. Fourth Universal Definition of Myocardial Infarction. Circulation. 2018;138:e618-e651.
14. Mahler SA, Riley RF, Hiestand BC, et al. The HEART Pathway randomized trial: identifying emergency department patients with acute chest pain for early discharge. Circ Cardiovasc Qual Outcomes. 2015;8:195-203.
15. Natarajan B, Mallick P, Thangalvadi TA, Rajavelu P. Validation of the HEART score in Indian population. Int J Emerg Med. 2015,8(suppl 1):P5.
16. McCord J, Cabrera R, Lindahl B, et al. Prognostic utility of a modified HEART score in chest pain patients in the emergency department. Circ Cardiovasc Qual Outcomes. 2017;10:e003101.
17. Mahler SA, Miller CD, Hollander JE, et al. Identifying patients for early discharge: performance of decision rules among patients with acute chest pain. Int J Cardiol. 2012;168:795-802.
Physicians wearing white coats rated more experienced
Physicians wearing white coats were rated as significantly more experienced and professional than peers wearing casual attire. Regardless of their attire, however, female physicians were more likely to be judged as appearing less professional and were more likely to be misidentified as medical technicians, physician assistants, or nurses, found research published in JAMA Network Open.
“A white coat with scrubs attire was most preferred for surgeons (mean preference index, 1.3), whereas a white coat with business attire was preferred for family physicians and dermatologists (mean preference indexes, 1.6 and 1.2, respectively; P < .001),” Helen Xun, MD, Johns Hopkins University, Baltimore, and colleagues wrote. “A male model wearing business inner wear with a white coat, fleece jacket, or softshell jacket was perceived as significantly more professional than a female model wearing the same attire (mean professionalism score: male, 65.8; female, 56.2; mean difference in professionalism score: white coat, 12.06; fleece, 7.89; softshell, 8.82; P < .001). ... A male model wearing hospital scrubs or fashion scrubs alone was also perceived as more professional than a female model in the same attire.”
While casual attire, such as fleece or softshell jackets emblazoned with the names of the institution and wearer, has become more popular attire for physicians in recent years, the researchers noted theirs is the first published research to identify associations between gender, attire, and how people distinguish between various health care roles. The study authors launched their web-based survey from May to June 2020 and asked people aged 18 years and older to rate a series of photographs of deidentified models wearing health care attire. Inner wear choices were business attire versus scrubs with and without outer wear options of a long white coat, gray fleece jacket, or black softshell jackets. Survey respondents ranked the images on a 6-point Likert scale with 1 being the least experienced, professional, and friendly and 6 being the most experienced, professional, and friendly. Survey respondents also viewed individual images of male or female models and were asked to rate their professionalism on a scale of 0-100 – with 100 as the “most professional” as well as to identify their profession as either physician, surgeon, nurse, medical technician, or physician assistant.
The study team included 487 (93.3%) of 522 completed surveys in their analyses. Respondents’ mean age was 36.2 years; 260 (53.4%) were female; 372 (76.4%) were White; 33 (6.8%) were Black or African American. Younger respondents and those living in the Western United States who had more exposure to physician casual attire appeared more accepting of it, the authors wrote.
“I remember attending my white-coat ceremony as a medical student, and the symbolism of it all representing me entering the profession. It felt very emotional and heavy and I felt very proud to be there. I also remember taking a ‘selfie’ in my long white coat as a doctor for the first time before my first shift as a resident. But, I’ve also been wearing that same white coat, and a large badge with a ‘DOCTOR’ label on it, and been mistaken by a patient or parent for something other than the physician,” Alexandra M. Sims, a pediatrician and health equity researcher in Cincinnati, said in an interview. “So, I’d really hope that the take-home here is not simply that we must wear our white coats to be considered more professional. I think we have to unpack and dismantle how we’ve even built this notion of ‘professionalism’ in the first place. Women, people of color, and other marginalized groups were certainly not a part of the defining, but we must be a part of the reimagining of an equitable health care profession in this new era.”
As sartorial trends usher in more casual attire, clinicians should redouble efforts to build rapport and enhance communication with patients, such as clarifying team members’ roles when introducing themselves. Dr. Xun and coauthors noted that addressing gender bias is important for all clinicians – not just women – and point to the need for institutional and organizational support for disciplines where gender bias is “especially prevalent,” like surgery. “This responsibility should not be undertaken only by the individuals that experience the biases, which may result in additional cumulative career disadvantages. The promotion of equality and diversity begins with recognition, characterization, and evidence-supported interventions and is a community operation,” Dr. Xun and colleagues concluded.
“I do not equate attire to professionalism or experience, nor is it connected to my satisfaction with the physician. For myself and my daughter, it is the experience of care that ultimately influences our perceptions regarding the professionalism of the physician,” Hala H. Durrah, MTA, parent to a chronically ill child with special health care needs and a Patient and Family Engagement Consultant, said in an interview. “My respect for a physician will ultimately be determined by how my daughter and I were treated, not just from a clinical perspective, but how we felt during those interactions.”
Physicians wearing white coats were rated as significantly more experienced and professional than peers wearing casual attire. Regardless of their attire, however, female physicians were more likely to be judged as appearing less professional and were more likely to be misidentified as medical technicians, physician assistants, or nurses, found research published in JAMA Network Open.
“A white coat with scrubs attire was most preferred for surgeons (mean preference index, 1.3), whereas a white coat with business attire was preferred for family physicians and dermatologists (mean preference indexes, 1.6 and 1.2, respectively; P < .001),” Helen Xun, MD, Johns Hopkins University, Baltimore, and colleagues wrote. “A male model wearing business inner wear with a white coat, fleece jacket, or softshell jacket was perceived as significantly more professional than a female model wearing the same attire (mean professionalism score: male, 65.8; female, 56.2; mean difference in professionalism score: white coat, 12.06; fleece, 7.89; softshell, 8.82; P < .001). ... A male model wearing hospital scrubs or fashion scrubs alone was also perceived as more professional than a female model in the same attire.”
While casual attire, such as fleece or softshell jackets emblazoned with the names of the institution and wearer, has become more popular attire for physicians in recent years, the researchers noted theirs is the first published research to identify associations between gender, attire, and how people distinguish between various health care roles. The study authors launched their web-based survey from May to June 2020 and asked people aged 18 years and older to rate a series of photographs of deidentified models wearing health care attire. Inner wear choices were business attire versus scrubs with and without outer wear options of a long white coat, gray fleece jacket, or black softshell jackets. Survey respondents ranked the images on a 6-point Likert scale with 1 being the least experienced, professional, and friendly and 6 being the most experienced, professional, and friendly. Survey respondents also viewed individual images of male or female models and were asked to rate their professionalism on a scale of 0-100 – with 100 as the “most professional” as well as to identify their profession as either physician, surgeon, nurse, medical technician, or physician assistant.
The study team included 487 (93.3%) of 522 completed surveys in their analyses. Respondents’ mean age was 36.2 years; 260 (53.4%) were female; 372 (76.4%) were White; 33 (6.8%) were Black or African American. Younger respondents and those living in the Western United States who had more exposure to physician casual attire appeared more accepting of it, the authors wrote.
“I remember attending my white-coat ceremony as a medical student, and the symbolism of it all representing me entering the profession. It felt very emotional and heavy and I felt very proud to be there. I also remember taking a ‘selfie’ in my long white coat as a doctor for the first time before my first shift as a resident. But, I’ve also been wearing that same white coat, and a large badge with a ‘DOCTOR’ label on it, and been mistaken by a patient or parent for something other than the physician,” Alexandra M. Sims, a pediatrician and health equity researcher in Cincinnati, said in an interview. “So, I’d really hope that the take-home here is not simply that we must wear our white coats to be considered more professional. I think we have to unpack and dismantle how we’ve even built this notion of ‘professionalism’ in the first place. Women, people of color, and other marginalized groups were certainly not a part of the defining, but we must be a part of the reimagining of an equitable health care profession in this new era.”
As sartorial trends usher in more casual attire, clinicians should redouble efforts to build rapport and enhance communication with patients, such as clarifying team members’ roles when introducing themselves. Dr. Xun and coauthors noted that addressing gender bias is important for all clinicians – not just women – and point to the need for institutional and organizational support for disciplines where gender bias is “especially prevalent,” like surgery. “This responsibility should not be undertaken only by the individuals that experience the biases, which may result in additional cumulative career disadvantages. The promotion of equality and diversity begins with recognition, characterization, and evidence-supported interventions and is a community operation,” Dr. Xun and colleagues concluded.
“I do not equate attire to professionalism or experience, nor is it connected to my satisfaction with the physician. For myself and my daughter, it is the experience of care that ultimately influences our perceptions regarding the professionalism of the physician,” Hala H. Durrah, MTA, parent to a chronically ill child with special health care needs and a Patient and Family Engagement Consultant, said in an interview. “My respect for a physician will ultimately be determined by how my daughter and I were treated, not just from a clinical perspective, but how we felt during those interactions.”
Physicians wearing white coats were rated as significantly more experienced and professional than peers wearing casual attire. Regardless of their attire, however, female physicians were more likely to be judged as appearing less professional and were more likely to be misidentified as medical technicians, physician assistants, or nurses, found research published in JAMA Network Open.
“A white coat with scrubs attire was most preferred for surgeons (mean preference index, 1.3), whereas a white coat with business attire was preferred for family physicians and dermatologists (mean preference indexes, 1.6 and 1.2, respectively; P < .001),” Helen Xun, MD, Johns Hopkins University, Baltimore, and colleagues wrote. “A male model wearing business inner wear with a white coat, fleece jacket, or softshell jacket was perceived as significantly more professional than a female model wearing the same attire (mean professionalism score: male, 65.8; female, 56.2; mean difference in professionalism score: white coat, 12.06; fleece, 7.89; softshell, 8.82; P < .001). ... A male model wearing hospital scrubs or fashion scrubs alone was also perceived as more professional than a female model in the same attire.”
While casual attire, such as fleece or softshell jackets emblazoned with the names of the institution and wearer, has become more popular attire for physicians in recent years, the researchers noted theirs is the first published research to identify associations between gender, attire, and how people distinguish between various health care roles. The study authors launched their web-based survey from May to June 2020 and asked people aged 18 years and older to rate a series of photographs of deidentified models wearing health care attire. Inner wear choices were business attire versus scrubs with and without outer wear options of a long white coat, gray fleece jacket, or black softshell jackets. Survey respondents ranked the images on a 6-point Likert scale with 1 being the least experienced, professional, and friendly and 6 being the most experienced, professional, and friendly. Survey respondents also viewed individual images of male or female models and were asked to rate their professionalism on a scale of 0-100 – with 100 as the “most professional” as well as to identify their profession as either physician, surgeon, nurse, medical technician, or physician assistant.
The study team included 487 (93.3%) of 522 completed surveys in their analyses. Respondents’ mean age was 36.2 years; 260 (53.4%) were female; 372 (76.4%) were White; 33 (6.8%) were Black or African American. Younger respondents and those living in the Western United States who had more exposure to physician casual attire appeared more accepting of it, the authors wrote.
“I remember attending my white-coat ceremony as a medical student, and the symbolism of it all representing me entering the profession. It felt very emotional and heavy and I felt very proud to be there. I also remember taking a ‘selfie’ in my long white coat as a doctor for the first time before my first shift as a resident. But, I’ve also been wearing that same white coat, and a large badge with a ‘DOCTOR’ label on it, and been mistaken by a patient or parent for something other than the physician,” Alexandra M. Sims, a pediatrician and health equity researcher in Cincinnati, said in an interview. “So, I’d really hope that the take-home here is not simply that we must wear our white coats to be considered more professional. I think we have to unpack and dismantle how we’ve even built this notion of ‘professionalism’ in the first place. Women, people of color, and other marginalized groups were certainly not a part of the defining, but we must be a part of the reimagining of an equitable health care profession in this new era.”
As sartorial trends usher in more casual attire, clinicians should redouble efforts to build rapport and enhance communication with patients, such as clarifying team members’ roles when introducing themselves. Dr. Xun and coauthors noted that addressing gender bias is important for all clinicians – not just women – and point to the need for institutional and organizational support for disciplines where gender bias is “especially prevalent,” like surgery. “This responsibility should not be undertaken only by the individuals that experience the biases, which may result in additional cumulative career disadvantages. The promotion of equality and diversity begins with recognition, characterization, and evidence-supported interventions and is a community operation,” Dr. Xun and colleagues concluded.
“I do not equate attire to professionalism or experience, nor is it connected to my satisfaction with the physician. For myself and my daughter, it is the experience of care that ultimately influences our perceptions regarding the professionalism of the physician,” Hala H. Durrah, MTA, parent to a chronically ill child with special health care needs and a Patient and Family Engagement Consultant, said in an interview. “My respect for a physician will ultimately be determined by how my daughter and I were treated, not just from a clinical perspective, but how we felt during those interactions.”
FROM JAMA NETWORK OPEN
Surgeon marks ‘right’ instead of ‘left’ testicle, then operates
Wrong-site surgery
Florida regulators have imposed a fine and other measures on a Tampa doctor who made a crucial error prior to his patient’s testicular surgery, as a story in the Miami Herald, among other news sites, reports.
On Sept. 10, 2019, a patient referred to in state documents as “C.F.” showed up for a procedure – a varicocelectomy – that would remove the enlarged veins in his left testicle. His doctor that day was Raul Fernandez-Crespo, MD, a urologist who had been licensed to practice in Florida since April of the same year. Dr. Fernandez-Crespo completed his urology residency at the University of Puerto Rico in 2019.
Following a conversation with C.F., Dr. Fernandez-Crespo designated what he believed was the proper surgical site – his patient’s right testicle.
He then proceeded to operate, but at some point during the procedure – news accounts don’t make clear when or how he became aware of his error – he realized C.F. had actually consented to a left-testicle varicocelectomy. With his patient still sedated, Dr. Fernandez-Crespo also completed the second procedure.
His mistake came to the attention of the Department of Health, which filed an administrative complaint against the surgeon. On June 17, 2021, the department’s medical licensing body, the Florida Board of Medicine, handed down its final order about the case.
In addition to imposing a $2,500 fine on Dr. Fernandez-Crespo and issuing “a letter of concern” – a public document that can be used as evidence in any relevant future disciplinary action against him – regulators said the surgeon must reimburse $2,045.56 to the department for its case-related administrative costs; take a 5-hour CME course in risk management or attend 8 hours of board disciplinary hearings; and, finally, give a 1-hour lecture on wrong-site surgeries at a board-approved medical facility.
Before this, Dr. Fernandez-Crespo had no previous disciplinary history with the Florida Board of Medicine.
Huge judgment after fertility procedure goes wrong
A Connecticut couple whose fertility and prenatal care at a state university health center proved disastrous will receive millions of dollars in damages, according to a report in the Hartford Courant.
In 2014, Jean-Marie Monroe-Lynch and her husband, Aaron Lynch, went to UConn Health, in Farmington, for treatment of Jean-Marie’s infertility. Her care was overseen by the Center for Advanced Reproductive Services (CARS), a private company then under contract with UConn Health. (The contract, which ended in 2014, obligated UConn to provide CARS providers with medical malpractice coverage.)
There, Jean-Marie was inseminated with sperm from a donor who turned out to be a carrier for cytomegalovirus (CMV), the herpes virus that can cause severe birth defects, or fetal death, when contracted by a pregnant woman. The insemination resulted in a twin pregnancy, a boy and a girl. The girl, Shay, died in utero after several of her organs became infected with CMV; the boy, Joshua, was born with severe mental and physical disabilities.
In their suit, Ms. Monroe-Lynch and her husband alleged that they were never cautioned about the risks associated with using a sperm donor whose blood had tested positive for CMV antibodies. Their suit further alleged that, at the 20-week ultrasound, UConn’s prenatal team failed to detect evidence of congenital CMV infection and again failed, at the 22-week ultrasound, to properly recognize and respond to abnormal findings.
“They totally dropped the ball,” said the couple’s attorney. “If you’re a pregnant woman and contract the virus for the first time, the results can be devastating.”
CARS disputes this conclusion, arguing that the plaintiffs failed to prove as a “matter of scientific fact” that Ms. Monroe-Lynch was infected with CMV as the result of her intrauterine insemination.
But Superior Court Judge Mark H. Taylor disagreed. In his 107-page ruling, he said that the court “agrees with the vast majority of superior courts, concluding that a physician providing obstetric care owes a direct duty to a mother to prevent harm to her child during gestation and delivery.”
Jean-Marie Monroe-Lynch and Aaron Lynch received a $37.6 million award, consisting of $24.1 million in economic damages and $13.5 million in noneconomic damages.
Their surviving child, Joshua, will reportedly require a lifetime of medical and other care. In the meantime, UConn Health vows to appeal the Superior Court’s decision.
COVID patient’s relative demands justice for fatal outcome
An Indiana man whose grandfather recently died after suffering a stroke is calling on state lawmakers to rethink legislation passed earlier this year to protect health care providers during the COVID-19 pandemic, according to a story reported by CBS4Indy.
Late last year, Daniel Enlow’s 83-year-old grandfather, Edward Rigney, was checked into Eskenazi Hospital, in Indianapolis. Mr. Rigney suffered from COPD and had also been diagnosed with COVID-19.
At some point during his hospitalization, medical staff attempted to place what seems to have been an arterial line in order to monitor his condition. During the procedure, or at some point shortly thereafter, an “iatrogenic air embolus” was released into his veins and caused a stroke, according to medical records and Mr. Rigney’s death certificate.
“I started asking for medical records because I wanted to know what was happening leading up to it in black and white in front of me,” said Mr. Enlow, who wished to present his evidence to a medical review panel, as required by Indiana law. The first step in this process would have been to consult with a medical malpractice attorney, but several declined to take his case.
Why? Because a pair of bills passed by Indiana legislators in early 2021 make COVID-19–related suits – even tangentially related ones – potentially difficult to take to court.
The bills raised the bar to file a medical malpractice claim in COVID-19 cases and to allow only those that involve “gross negligence or willful or wanton misconduct.”
“In the vast majority of cases, it’s impossible to prove that,” said Fred Schultz, immediate past president of the Indiana Trial Lawyers Association, who lobbied against the legislation.
The bills were never designed to offer “blanket freedom,” said GOP State Senator Aaron Freeman, sponsor of one of the bills. “If something is being used in a way that it is a complete bar to certain claims, then maybe we need to go back and look at it and open that up a little bit and make it less restrictive. I’m certainly open to having those conversations.”
Meanwhile, Mr. Enlow has vowed to keep pushing in the name of his late grandfather. The hospital’s parent company, Eskenazi Health, has declined to comment.
A version of this article first appeared on Medscape.com.
Wrong-site surgery
Florida regulators have imposed a fine and other measures on a Tampa doctor who made a crucial error prior to his patient’s testicular surgery, as a story in the Miami Herald, among other news sites, reports.
On Sept. 10, 2019, a patient referred to in state documents as “C.F.” showed up for a procedure – a varicocelectomy – that would remove the enlarged veins in his left testicle. His doctor that day was Raul Fernandez-Crespo, MD, a urologist who had been licensed to practice in Florida since April of the same year. Dr. Fernandez-Crespo completed his urology residency at the University of Puerto Rico in 2019.
Following a conversation with C.F., Dr. Fernandez-Crespo designated what he believed was the proper surgical site – his patient’s right testicle.
He then proceeded to operate, but at some point during the procedure – news accounts don’t make clear when or how he became aware of his error – he realized C.F. had actually consented to a left-testicle varicocelectomy. With his patient still sedated, Dr. Fernandez-Crespo also completed the second procedure.
His mistake came to the attention of the Department of Health, which filed an administrative complaint against the surgeon. On June 17, 2021, the department’s medical licensing body, the Florida Board of Medicine, handed down its final order about the case.
In addition to imposing a $2,500 fine on Dr. Fernandez-Crespo and issuing “a letter of concern” – a public document that can be used as evidence in any relevant future disciplinary action against him – regulators said the surgeon must reimburse $2,045.56 to the department for its case-related administrative costs; take a 5-hour CME course in risk management or attend 8 hours of board disciplinary hearings; and, finally, give a 1-hour lecture on wrong-site surgeries at a board-approved medical facility.
Before this, Dr. Fernandez-Crespo had no previous disciplinary history with the Florida Board of Medicine.
Huge judgment after fertility procedure goes wrong
A Connecticut couple whose fertility and prenatal care at a state university health center proved disastrous will receive millions of dollars in damages, according to a report in the Hartford Courant.
In 2014, Jean-Marie Monroe-Lynch and her husband, Aaron Lynch, went to UConn Health, in Farmington, for treatment of Jean-Marie’s infertility. Her care was overseen by the Center for Advanced Reproductive Services (CARS), a private company then under contract with UConn Health. (The contract, which ended in 2014, obligated UConn to provide CARS providers with medical malpractice coverage.)
There, Jean-Marie was inseminated with sperm from a donor who turned out to be a carrier for cytomegalovirus (CMV), the herpes virus that can cause severe birth defects, or fetal death, when contracted by a pregnant woman. The insemination resulted in a twin pregnancy, a boy and a girl. The girl, Shay, died in utero after several of her organs became infected with CMV; the boy, Joshua, was born with severe mental and physical disabilities.
In their suit, Ms. Monroe-Lynch and her husband alleged that they were never cautioned about the risks associated with using a sperm donor whose blood had tested positive for CMV antibodies. Their suit further alleged that, at the 20-week ultrasound, UConn’s prenatal team failed to detect evidence of congenital CMV infection and again failed, at the 22-week ultrasound, to properly recognize and respond to abnormal findings.
“They totally dropped the ball,” said the couple’s attorney. “If you’re a pregnant woman and contract the virus for the first time, the results can be devastating.”
CARS disputes this conclusion, arguing that the plaintiffs failed to prove as a “matter of scientific fact” that Ms. Monroe-Lynch was infected with CMV as the result of her intrauterine insemination.
But Superior Court Judge Mark H. Taylor disagreed. In his 107-page ruling, he said that the court “agrees with the vast majority of superior courts, concluding that a physician providing obstetric care owes a direct duty to a mother to prevent harm to her child during gestation and delivery.”
Jean-Marie Monroe-Lynch and Aaron Lynch received a $37.6 million award, consisting of $24.1 million in economic damages and $13.5 million in noneconomic damages.
Their surviving child, Joshua, will reportedly require a lifetime of medical and other care. In the meantime, UConn Health vows to appeal the Superior Court’s decision.
COVID patient’s relative demands justice for fatal outcome
An Indiana man whose grandfather recently died after suffering a stroke is calling on state lawmakers to rethink legislation passed earlier this year to protect health care providers during the COVID-19 pandemic, according to a story reported by CBS4Indy.
Late last year, Daniel Enlow’s 83-year-old grandfather, Edward Rigney, was checked into Eskenazi Hospital, in Indianapolis. Mr. Rigney suffered from COPD and had also been diagnosed with COVID-19.
At some point during his hospitalization, medical staff attempted to place what seems to have been an arterial line in order to monitor his condition. During the procedure, or at some point shortly thereafter, an “iatrogenic air embolus” was released into his veins and caused a stroke, according to medical records and Mr. Rigney’s death certificate.
“I started asking for medical records because I wanted to know what was happening leading up to it in black and white in front of me,” said Mr. Enlow, who wished to present his evidence to a medical review panel, as required by Indiana law. The first step in this process would have been to consult with a medical malpractice attorney, but several declined to take his case.
Why? Because a pair of bills passed by Indiana legislators in early 2021 make COVID-19–related suits – even tangentially related ones – potentially difficult to take to court.
The bills raised the bar to file a medical malpractice claim in COVID-19 cases and to allow only those that involve “gross negligence or willful or wanton misconduct.”
“In the vast majority of cases, it’s impossible to prove that,” said Fred Schultz, immediate past president of the Indiana Trial Lawyers Association, who lobbied against the legislation.
The bills were never designed to offer “blanket freedom,” said GOP State Senator Aaron Freeman, sponsor of one of the bills. “If something is being used in a way that it is a complete bar to certain claims, then maybe we need to go back and look at it and open that up a little bit and make it less restrictive. I’m certainly open to having those conversations.”
Meanwhile, Mr. Enlow has vowed to keep pushing in the name of his late grandfather. The hospital’s parent company, Eskenazi Health, has declined to comment.
A version of this article first appeared on Medscape.com.
Wrong-site surgery
Florida regulators have imposed a fine and other measures on a Tampa doctor who made a crucial error prior to his patient’s testicular surgery, as a story in the Miami Herald, among other news sites, reports.
On Sept. 10, 2019, a patient referred to in state documents as “C.F.” showed up for a procedure – a varicocelectomy – that would remove the enlarged veins in his left testicle. His doctor that day was Raul Fernandez-Crespo, MD, a urologist who had been licensed to practice in Florida since April of the same year. Dr. Fernandez-Crespo completed his urology residency at the University of Puerto Rico in 2019.
Following a conversation with C.F., Dr. Fernandez-Crespo designated what he believed was the proper surgical site – his patient’s right testicle.
He then proceeded to operate, but at some point during the procedure – news accounts don’t make clear when or how he became aware of his error – he realized C.F. had actually consented to a left-testicle varicocelectomy. With his patient still sedated, Dr. Fernandez-Crespo also completed the second procedure.
His mistake came to the attention of the Department of Health, which filed an administrative complaint against the surgeon. On June 17, 2021, the department’s medical licensing body, the Florida Board of Medicine, handed down its final order about the case.
In addition to imposing a $2,500 fine on Dr. Fernandez-Crespo and issuing “a letter of concern” – a public document that can be used as evidence in any relevant future disciplinary action against him – regulators said the surgeon must reimburse $2,045.56 to the department for its case-related administrative costs; take a 5-hour CME course in risk management or attend 8 hours of board disciplinary hearings; and, finally, give a 1-hour lecture on wrong-site surgeries at a board-approved medical facility.
Before this, Dr. Fernandez-Crespo had no previous disciplinary history with the Florida Board of Medicine.
Huge judgment after fertility procedure goes wrong
A Connecticut couple whose fertility and prenatal care at a state university health center proved disastrous will receive millions of dollars in damages, according to a report in the Hartford Courant.
In 2014, Jean-Marie Monroe-Lynch and her husband, Aaron Lynch, went to UConn Health, in Farmington, for treatment of Jean-Marie’s infertility. Her care was overseen by the Center for Advanced Reproductive Services (CARS), a private company then under contract with UConn Health. (The contract, which ended in 2014, obligated UConn to provide CARS providers with medical malpractice coverage.)
There, Jean-Marie was inseminated with sperm from a donor who turned out to be a carrier for cytomegalovirus (CMV), the herpes virus that can cause severe birth defects, or fetal death, when contracted by a pregnant woman. The insemination resulted in a twin pregnancy, a boy and a girl. The girl, Shay, died in utero after several of her organs became infected with CMV; the boy, Joshua, was born with severe mental and physical disabilities.
In their suit, Ms. Monroe-Lynch and her husband alleged that they were never cautioned about the risks associated with using a sperm donor whose blood had tested positive for CMV antibodies. Their suit further alleged that, at the 20-week ultrasound, UConn’s prenatal team failed to detect evidence of congenital CMV infection and again failed, at the 22-week ultrasound, to properly recognize and respond to abnormal findings.
“They totally dropped the ball,” said the couple’s attorney. “If you’re a pregnant woman and contract the virus for the first time, the results can be devastating.”
CARS disputes this conclusion, arguing that the plaintiffs failed to prove as a “matter of scientific fact” that Ms. Monroe-Lynch was infected with CMV as the result of her intrauterine insemination.
But Superior Court Judge Mark H. Taylor disagreed. In his 107-page ruling, he said that the court “agrees with the vast majority of superior courts, concluding that a physician providing obstetric care owes a direct duty to a mother to prevent harm to her child during gestation and delivery.”
Jean-Marie Monroe-Lynch and Aaron Lynch received a $37.6 million award, consisting of $24.1 million in economic damages and $13.5 million in noneconomic damages.
Their surviving child, Joshua, will reportedly require a lifetime of medical and other care. In the meantime, UConn Health vows to appeal the Superior Court’s decision.
COVID patient’s relative demands justice for fatal outcome
An Indiana man whose grandfather recently died after suffering a stroke is calling on state lawmakers to rethink legislation passed earlier this year to protect health care providers during the COVID-19 pandemic, according to a story reported by CBS4Indy.
Late last year, Daniel Enlow’s 83-year-old grandfather, Edward Rigney, was checked into Eskenazi Hospital, in Indianapolis. Mr. Rigney suffered from COPD and had also been diagnosed with COVID-19.
At some point during his hospitalization, medical staff attempted to place what seems to have been an arterial line in order to monitor his condition. During the procedure, or at some point shortly thereafter, an “iatrogenic air embolus” was released into his veins and caused a stroke, according to medical records and Mr. Rigney’s death certificate.
“I started asking for medical records because I wanted to know what was happening leading up to it in black and white in front of me,” said Mr. Enlow, who wished to present his evidence to a medical review panel, as required by Indiana law. The first step in this process would have been to consult with a medical malpractice attorney, but several declined to take his case.
Why? Because a pair of bills passed by Indiana legislators in early 2021 make COVID-19–related suits – even tangentially related ones – potentially difficult to take to court.
The bills raised the bar to file a medical malpractice claim in COVID-19 cases and to allow only those that involve “gross negligence or willful or wanton misconduct.”
“In the vast majority of cases, it’s impossible to prove that,” said Fred Schultz, immediate past president of the Indiana Trial Lawyers Association, who lobbied against the legislation.
The bills were never designed to offer “blanket freedom,” said GOP State Senator Aaron Freeman, sponsor of one of the bills. “If something is being used in a way that it is a complete bar to certain claims, then maybe we need to go back and look at it and open that up a little bit and make it less restrictive. I’m certainly open to having those conversations.”
Meanwhile, Mr. Enlow has vowed to keep pushing in the name of his late grandfather. The hospital’s parent company, Eskenazi Health, has declined to comment.
A version of this article first appeared on Medscape.com.
COVID-19 leaves wake of medical debt among U.S. adults
Despite the passage of four major relief bills in 2020 and 2021 and federal efforts to offset pandemic- and job-related coverage loss, many people continued to face financial challenges, especially those with a low income and those who are Black or Latino.
The survey, which included responses from 5,450 adults, revealed that 10% of adults aged 19-64 were uninsured during the first half of 2021, a rate lower than what was recorded in 2020 and 2019 in both federal and private surveys. However, uninsured rates were highest among those with low income, those younger than 50 years old, and Black and Latino adults.
For most adults who lost employee health insurance, the coverage gap was relatively brief, with 54% saying their coverage gap lasted 3-4 months. Only 16% of adults said coverage gaps lasted a year or longer.
“The good news is that this survey is suggesting that the coverage losses during the pandemic may have been offset by federal efforts to help people get and maintain health insurance coverage,” lead author Sara Collins, PhD, Commonwealth Fund vice president for health care coverage, access, and tracking, said in an interview.
“The bad news is that a third of Americans continue to struggle with medical bills and medical debt, even among those who have health insurance coverage,” Dr. Collins added.
Indeed, the survey found that about one-third of insured adults reported a medical bill problem or that they were paying off medical debt, as did approximately half of those who were uninsured. Medical debt caused 35% of respondents to use up most or all of their savings to pay it off.
Meanwhile, 27% of adults said medical bills left them unable to pay for necessities such as food, heat, or rent. What surprised Dr. Collins was that 43% of adults said they received a lower credit rating as a result of their medical debt, and 35% said they had taken on more credit card debt to pay off these bills.
“The fact that it’s bleeding over into people’s financial security in terms of their credit scores, I think is something that really needs to be looked at by policymakers,” Dr. Collins said.
When analyzed by race/ethnicity, the researchers found that 55% of Black adults and 44% of Latino/Hispanic adults reported medical bills and debt problems, compared with 32% of White adults. In addition, 47% of those living below the poverty line also reported problems with medical bills.
According to the survey, 45% of respondents were directly affected by the pandemic in at least one of three ways – testing positive or getting sick from COVID-19, losing income, or losing employer coverage – with Black and Latinx adults and those with lower incomes at greater risk.
George Abraham, MD, president of the American College of Physicians, said the Commonwealth Fund’s findings were not surprising because it has always been known that underrepresented populations struggle for access to care because of socioeconomic factors. He said these populations were more vulnerable in terms of more severe infections and disease burden during the pandemic.
“[This study] validates what primary care physicians have been saying all along in regard to our patients’ access to care and their ability to cover health care costs,” said Dr. Abraham, who was not involved with the study. “This will hopefully be an eye-opener and wake-up call that reiterates that we still do not have equitable access to care and vulnerable populations are disproportionately affected.”
He believes that, although people are insured, many of them may contend with medical debt when they fall ill because they can’t afford the premiums.
“Even though they may have been registered for health coverage, they may not have active coverage at the time of illness simply because they weren’t able to make their last premium payments because they’ve been down, because they lost their job, or whatever else,” Dr. Abraham explained. “On paper, they appear to have health care coverage. But in reality, clearly, that coverage does not match their needs or it’s not affordable.”
For Dr. Abraham, the study emphasizes the need to continue support for health care reform, including pricing it so that insurance is available for those with fewer socioeconomic resources.
Yalda Jabbarpour, MD, medical director of the Robert Graham Center for Policy Studies, Washington, said high-deductible health plans need to be “reined in” because they can lead to greater debt, particularly among vulnerable populations.
“Hopefully this will encourage policymakers to look more closely at the problem of medical debt as a contributing factor to financial instability,” Dr. Jabbarpour said. “Federal relief is important, so is expanding access to comprehensive, affordable health care coverage.”
Dr. Collins said there should also be a way to raise awareness of the health care marketplace and coverage options so that people have an easier time getting insured.
A version of this article first appeared on Medscape.com.
Despite the passage of four major relief bills in 2020 and 2021 and federal efforts to offset pandemic- and job-related coverage loss, many people continued to face financial challenges, especially those with a low income and those who are Black or Latino.
The survey, which included responses from 5,450 adults, revealed that 10% of adults aged 19-64 were uninsured during the first half of 2021, a rate lower than what was recorded in 2020 and 2019 in both federal and private surveys. However, uninsured rates were highest among those with low income, those younger than 50 years old, and Black and Latino adults.
For most adults who lost employee health insurance, the coverage gap was relatively brief, with 54% saying their coverage gap lasted 3-4 months. Only 16% of adults said coverage gaps lasted a year or longer.
“The good news is that this survey is suggesting that the coverage losses during the pandemic may have been offset by federal efforts to help people get and maintain health insurance coverage,” lead author Sara Collins, PhD, Commonwealth Fund vice president for health care coverage, access, and tracking, said in an interview.
“The bad news is that a third of Americans continue to struggle with medical bills and medical debt, even among those who have health insurance coverage,” Dr. Collins added.
Indeed, the survey found that about one-third of insured adults reported a medical bill problem or that they were paying off medical debt, as did approximately half of those who were uninsured. Medical debt caused 35% of respondents to use up most or all of their savings to pay it off.
Meanwhile, 27% of adults said medical bills left them unable to pay for necessities such as food, heat, or rent. What surprised Dr. Collins was that 43% of adults said they received a lower credit rating as a result of their medical debt, and 35% said they had taken on more credit card debt to pay off these bills.
“The fact that it’s bleeding over into people’s financial security in terms of their credit scores, I think is something that really needs to be looked at by policymakers,” Dr. Collins said.
When analyzed by race/ethnicity, the researchers found that 55% of Black adults and 44% of Latino/Hispanic adults reported medical bills and debt problems, compared with 32% of White adults. In addition, 47% of those living below the poverty line also reported problems with medical bills.
According to the survey, 45% of respondents were directly affected by the pandemic in at least one of three ways – testing positive or getting sick from COVID-19, losing income, or losing employer coverage – with Black and Latinx adults and those with lower incomes at greater risk.
George Abraham, MD, president of the American College of Physicians, said the Commonwealth Fund’s findings were not surprising because it has always been known that underrepresented populations struggle for access to care because of socioeconomic factors. He said these populations were more vulnerable in terms of more severe infections and disease burden during the pandemic.
“[This study] validates what primary care physicians have been saying all along in regard to our patients’ access to care and their ability to cover health care costs,” said Dr. Abraham, who was not involved with the study. “This will hopefully be an eye-opener and wake-up call that reiterates that we still do not have equitable access to care and vulnerable populations are disproportionately affected.”
He believes that, although people are insured, many of them may contend with medical debt when they fall ill because they can’t afford the premiums.
“Even though they may have been registered for health coverage, they may not have active coverage at the time of illness simply because they weren’t able to make their last premium payments because they’ve been down, because they lost their job, or whatever else,” Dr. Abraham explained. “On paper, they appear to have health care coverage. But in reality, clearly, that coverage does not match their needs or it’s not affordable.”
For Dr. Abraham, the study emphasizes the need to continue support for health care reform, including pricing it so that insurance is available for those with fewer socioeconomic resources.
Yalda Jabbarpour, MD, medical director of the Robert Graham Center for Policy Studies, Washington, said high-deductible health plans need to be “reined in” because they can lead to greater debt, particularly among vulnerable populations.
“Hopefully this will encourage policymakers to look more closely at the problem of medical debt as a contributing factor to financial instability,” Dr. Jabbarpour said. “Federal relief is important, so is expanding access to comprehensive, affordable health care coverage.”
Dr. Collins said there should also be a way to raise awareness of the health care marketplace and coverage options so that people have an easier time getting insured.
A version of this article first appeared on Medscape.com.
Despite the passage of four major relief bills in 2020 and 2021 and federal efforts to offset pandemic- and job-related coverage loss, many people continued to face financial challenges, especially those with a low income and those who are Black or Latino.
The survey, which included responses from 5,450 adults, revealed that 10% of adults aged 19-64 were uninsured during the first half of 2021, a rate lower than what was recorded in 2020 and 2019 in both federal and private surveys. However, uninsured rates were highest among those with low income, those younger than 50 years old, and Black and Latino adults.
For most adults who lost employee health insurance, the coverage gap was relatively brief, with 54% saying their coverage gap lasted 3-4 months. Only 16% of adults said coverage gaps lasted a year or longer.
“The good news is that this survey is suggesting that the coverage losses during the pandemic may have been offset by federal efforts to help people get and maintain health insurance coverage,” lead author Sara Collins, PhD, Commonwealth Fund vice president for health care coverage, access, and tracking, said in an interview.
“The bad news is that a third of Americans continue to struggle with medical bills and medical debt, even among those who have health insurance coverage,” Dr. Collins added.
Indeed, the survey found that about one-third of insured adults reported a medical bill problem or that they were paying off medical debt, as did approximately half of those who were uninsured. Medical debt caused 35% of respondents to use up most or all of their savings to pay it off.
Meanwhile, 27% of adults said medical bills left them unable to pay for necessities such as food, heat, or rent. What surprised Dr. Collins was that 43% of adults said they received a lower credit rating as a result of their medical debt, and 35% said they had taken on more credit card debt to pay off these bills.
“The fact that it’s bleeding over into people’s financial security in terms of their credit scores, I think is something that really needs to be looked at by policymakers,” Dr. Collins said.
When analyzed by race/ethnicity, the researchers found that 55% of Black adults and 44% of Latino/Hispanic adults reported medical bills and debt problems, compared with 32% of White adults. In addition, 47% of those living below the poverty line also reported problems with medical bills.
According to the survey, 45% of respondents were directly affected by the pandemic in at least one of three ways – testing positive or getting sick from COVID-19, losing income, or losing employer coverage – with Black and Latinx adults and those with lower incomes at greater risk.
George Abraham, MD, president of the American College of Physicians, said the Commonwealth Fund’s findings were not surprising because it has always been known that underrepresented populations struggle for access to care because of socioeconomic factors. He said these populations were more vulnerable in terms of more severe infections and disease burden during the pandemic.
“[This study] validates what primary care physicians have been saying all along in regard to our patients’ access to care and their ability to cover health care costs,” said Dr. Abraham, who was not involved with the study. “This will hopefully be an eye-opener and wake-up call that reiterates that we still do not have equitable access to care and vulnerable populations are disproportionately affected.”
He believes that, although people are insured, many of them may contend with medical debt when they fall ill because they can’t afford the premiums.
“Even though they may have been registered for health coverage, they may not have active coverage at the time of illness simply because they weren’t able to make their last premium payments because they’ve been down, because they lost their job, or whatever else,” Dr. Abraham explained. “On paper, they appear to have health care coverage. But in reality, clearly, that coverage does not match their needs or it’s not affordable.”
For Dr. Abraham, the study emphasizes the need to continue support for health care reform, including pricing it so that insurance is available for those with fewer socioeconomic resources.
Yalda Jabbarpour, MD, medical director of the Robert Graham Center for Policy Studies, Washington, said high-deductible health plans need to be “reined in” because they can lead to greater debt, particularly among vulnerable populations.
“Hopefully this will encourage policymakers to look more closely at the problem of medical debt as a contributing factor to financial instability,” Dr. Jabbarpour said. “Federal relief is important, so is expanding access to comprehensive, affordable health care coverage.”
Dr. Collins said there should also be a way to raise awareness of the health care marketplace and coverage options so that people have an easier time getting insured.
A version of this article first appeared on Medscape.com.
Traumatic Fractures Should Trigger Osteoporosis Assessment in Postmenopausal Women
Study Overview
Objective. To compare the risk of subsequent fractures after an initial traumatic or nontraumatic fracture in postmenopausal women.
Design. A prospective observational study utilizing data from the Women’s Health Initiative (WHI) Study, WHI Clinical Trials (WHI-CT), and WHI Bone Density Substudy to evaluate rates at which patients who suffered a traumatic fracture vs nontraumatic fracture develop a subsequent fracture.
Setting and participants. The WHI study, implemented at 40 United States clinical sites, enrolled 161 808 postmenopausal women aged 50 to 79 years at baseline between 1993 and 1998. The study cohort consisted of 75 335 patients who had self-reported fractures from September 1994 to December 1998 that were confirmed by the WHI Bone Density Substudy and WHI-CT. Of these participants, 253 (0.3%) were excluded because of a lack of follow-up information regarding incident fractures, and 8208 (10.9%) were excluded due to incomplete information on covariates, thus resulting in an analytic sample of 66 874 (88.8%) participants. Prospective fracture ascertainment with participants was conducted at least annually and the mechanism of fracture was assessed to differentiate traumatic vs nontraumatic incident fractures. Traumatic fractures were defined as fractures caused by motor vehicle collisions, falls from a height, falls downstairs, or sports injury. Nontraumatic fractures were defined as fractures caused by a trip and fall.
Main outcome measures. The primary outcome was an incident fracture at an anatomically distinct body part. Fractures were classified as upper extremity (carpal, elbow, lower or upper end of humerus, shaft of humerus, upper radius/ulna, or radius/ulna), lower extremity (ankle, hip, patella, pelvis, shaft of femur, tibia/fibula, or tibial plateau), or spine (lumbar and/or thoracic spine). Self-reported fractures were verified via medical chart review by WHI study physicians; hip fractures were confirmed by review of written reports of radiographic studies; and nonhip fractures were confirmed by review of radiography reports or clinical documentations.
Main results. In total, 66 874 women in the study (mean [SD] age) 63.1 (7.0) years without clinical fracture and 65.3 (7.2) years with clinical fracture at baseline were followed for 8.1 (1.6) years. Of these participants, 7142 (10.7%) experienced incident fracture during the study follow-up period (13.9 per 1000 person-years), and 721 (10.1%) of whom had a subsequent fracture. The adjusted hazard ratio (aHR) of subsequent fracture after an initial fracture was 1.49 (95% CI, 1.38-1.61, P < .001). Covariates adjusted were age, race, ethnicity, body mass index, treated diabetes, frequency of falls in the previous year, and physical function and activity. In women with initial traumatic fracture, the association between initial and subsequent fracture was increased (aHR, 1.25; 95% CI, 1.06-1.48, P = .01). Among women with initial nontraumatic fracture, the association between initial and subsequent fracture was also increased (aHR, 1.52; 95% CI, 1.37-1.68, P < .001). The confidence intervals for the 2 preceding associations for traumatic and nontraumatic initial fracture strata were overlapping.
Conclusion. Fractures, regardless of mechanism of injury, are similarly associated with an increased risk of subsequent fractures in postmenopausal women aged 50 years and older. Findings from this study provide evidence to support reevaluation of current clinical guidelines to include traumatic fracture as a trigger for osteoporosis screening.
Commentary
Osteoporosis is one of the most common age-associated disease that affects 1 in 4 women and 1 in 20 men over the age of 65.1 It increases the risk of fracture, and its clinical sequelae include reduced mobility, health decline, and increased all-cause mortality. The high prevalence of osteoporosis poses a clinical challenge as the global population continues to age. Pharmacological treatments such as bisphosphonates are highly effective in preventing or slowing bone mineral density (BMD) loss and reducing risk of fragility fractures (eg, nontraumatic fractures of the vertebra, hip, and femur) and are commonly used to mitigate adverse effects of degenerative bone changes secondary to osteoporosis.1
The high prevalence of osteoporosis and effectiveness of bisphosphonates raises the question of how to optimally identify adults at risk for osteoporosis so that pharmacologic therapy can be promptly initiated to prevent disease progression. Multiple osteoporosis screening guidelines, including those from the United States Preventive Services Task Force (USPSTF), American Association of Family Physicians, and National Osteoporosis Foundation, are widely used in the clinical setting to address this important clinical question. In general, the prevailing wisdom is to screen osteoporosis in postmenopausal women over the age of 65, women under the age of 65 who have a significant 10-year fracture risk, or women over the age of 50 who have experienced a fragility fracture.1 In the study reported by Crandall et al, it was shown that the risks of having subsequent fractures were similar after an initial traumatic or nontraumatic (fragility) fracture in postmenopausal women aged 50 years and older.2 This finding brings into question whether traumatic fractures should be viewed any differently than nontraumatic fractures in women over the age of 50 in light of evaluation for osteoporosis. Furthermore, these results suggest that most fractures in postmenopausal women may indicate decreased bone integrity, thus adding to the rationale that osteoporosis screening needs to be considered and expanded to include postmenopausal women under the age of 65 who endured a traumatic fracture.
Per current guidelines, a woman under the age of 65 is recommended for osteoporosis screening only if she has an increased 10-year fracture risk compared to women aged 65 years and older. This risk is calculated based on the World Health Organization fracture-risk algorithm (WHO FRAX) tool which uses multiple factors such as age, weight, and history of fragility fractures to predict whether an individual is at risk of developing a fracture in the next 10 years. The WHO FRAX tool does not include traumatic fractures in its risk calculation and current clinical guidelines do not account for traumatic fractures as a red flag to initiate osteoporosis screening. Therefore, postmenopausal women under the age of 65 are less likely to be screened for osteoporosis when they experience a traumatic fracture compared to a fragility fracture, despite being at a demonstrably higher risk for subsequent fracture. As an unintended consequence, this may lead to the under diagnosis of osteoporosis in postmenopausal women under the age of 65. Thus, Crandall et al conclude that a fracture due to any cause warrants follow up evaluation for osteoporosis including BMD testing in women older than 50 years of age.
Older men constitute another population who are commonly under screened for osteoporosis. The current USPSTF guidelines indicate that there is an insufficient body of evidence to screen men for osteoporosis given its lower prevalence.1 However, it is important to note that men have significantly increased mortality after a hip fracture, are less likely to be on pharmacological treatment for osteoporosis, and are under diagnosed for osteoporosis.3 Consistent with findings from the current study, Leslie et al showed that high-trauma and low-trauma fractures have similarly elevated subsequent fracture risk in both men and women over the age of 40 in a Canadian study.4 Moreover, in the same study, BMD was decreased in both men and women who suffered a fracture regardless of the injury mechanism. This finding further underscores a need to consider traumatic fractures as a risk factor for osteoporosis. Taken together, given that men are under screened and treated for osteoporosis but have increased mortality post-fracture, considerations to initiate osteoporosis evaluation should be similarly given to men who endured a traumatic fracture.
The study conducted by Crandall et al has several strengths. It is noteworthy for the large size of the WHI cohort with participants from across the United States which enables the capture of a wider range of age groups as women under the age of 65 are not common participants of osteoporosis studies. Additionally, data ascertainment and outcome adjudication utilizing medical records and physician review assure data quality. A limitation of the study is that the study cohort consists exclusively of women and therefore the findings are not generalizable to men. However, findings from this study echo those from other studies that investigate the relationship between fracture mechanisms and subsequent fracture risk in men and women.3,4 Collectively, these comparable findings highlight the need for additional research to validate traumatic fracture as a risk factor for osteoporosis and to incorporate it into clinical guidelines for osteoporosis screening.
Applications for Clinical Practice
The findings from the current study indicate that traumatic and fragility fractures may be more alike than previously recognized in regards to bone health and subsequent fracture prevention in postmenopausal women. If validated, these results may lead to changes in clinical practice whereby all fractures in postmenopausal women could trigger osteoporosis screening, assessment, and treatment if indicated for the secondary prevention of fractures.
1. US Preventive Services Task Force, Curry SJ, Krist Ah, et al. Screening for Osteoporosis to Prevent Fractures: US Preventive Services Task Force Recommendation Statement. JAMA. 2018;319(24):2521–2531. doi:10.1001/jama.2018.7498
2. Crandall CJ, Larson JC, LaCroix AZ, et al. Risk of Subsequent Fractures in Postmenopausal Women After Nontraumatic vs Traumatic Fractures. JAMA Intern Med. Published online June 7, 2021. doi:10.1001/jamainternmed.2021.2617
3. Mackey DC, Lui L, Cawthon PM, et al. High-Trauma Fractures and Low Bone Mineral Density in Older Women and Men. JAMA. 2007;298(20):2381–2388. doi:10.1001/jama.298.20.2381
4. Leslie WD, Schousboe JT, Morin SN, et al. Fracture risk following high-trauma versus low-trauma fracture: a registry-based cohort study. Osteoporos Int. 2020;31(6):1059–1067. doi:10.1007/s00198-019-05274-2
Study Overview
Objective. To compare the risk of subsequent fractures after an initial traumatic or nontraumatic fracture in postmenopausal women.
Design. A prospective observational study utilizing data from the Women’s Health Initiative (WHI) Study, WHI Clinical Trials (WHI-CT), and WHI Bone Density Substudy to evaluate rates at which patients who suffered a traumatic fracture vs nontraumatic fracture develop a subsequent fracture.
Setting and participants. The WHI study, implemented at 40 United States clinical sites, enrolled 161 808 postmenopausal women aged 50 to 79 years at baseline between 1993 and 1998. The study cohort consisted of 75 335 patients who had self-reported fractures from September 1994 to December 1998 that were confirmed by the WHI Bone Density Substudy and WHI-CT. Of these participants, 253 (0.3%) were excluded because of a lack of follow-up information regarding incident fractures, and 8208 (10.9%) were excluded due to incomplete information on covariates, thus resulting in an analytic sample of 66 874 (88.8%) participants. Prospective fracture ascertainment with participants was conducted at least annually and the mechanism of fracture was assessed to differentiate traumatic vs nontraumatic incident fractures. Traumatic fractures were defined as fractures caused by motor vehicle collisions, falls from a height, falls downstairs, or sports injury. Nontraumatic fractures were defined as fractures caused by a trip and fall.
Main outcome measures. The primary outcome was an incident fracture at an anatomically distinct body part. Fractures were classified as upper extremity (carpal, elbow, lower or upper end of humerus, shaft of humerus, upper radius/ulna, or radius/ulna), lower extremity (ankle, hip, patella, pelvis, shaft of femur, tibia/fibula, or tibial plateau), or spine (lumbar and/or thoracic spine). Self-reported fractures were verified via medical chart review by WHI study physicians; hip fractures were confirmed by review of written reports of radiographic studies; and nonhip fractures were confirmed by review of radiography reports or clinical documentations.
Main results. In total, 66 874 women in the study (mean [SD] age) 63.1 (7.0) years without clinical fracture and 65.3 (7.2) years with clinical fracture at baseline were followed for 8.1 (1.6) years. Of these participants, 7142 (10.7%) experienced incident fracture during the study follow-up period (13.9 per 1000 person-years), and 721 (10.1%) of whom had a subsequent fracture. The adjusted hazard ratio (aHR) of subsequent fracture after an initial fracture was 1.49 (95% CI, 1.38-1.61, P < .001). Covariates adjusted were age, race, ethnicity, body mass index, treated diabetes, frequency of falls in the previous year, and physical function and activity. In women with initial traumatic fracture, the association between initial and subsequent fracture was increased (aHR, 1.25; 95% CI, 1.06-1.48, P = .01). Among women with initial nontraumatic fracture, the association between initial and subsequent fracture was also increased (aHR, 1.52; 95% CI, 1.37-1.68, P < .001). The confidence intervals for the 2 preceding associations for traumatic and nontraumatic initial fracture strata were overlapping.
Conclusion. Fractures, regardless of mechanism of injury, are similarly associated with an increased risk of subsequent fractures in postmenopausal women aged 50 years and older. Findings from this study provide evidence to support reevaluation of current clinical guidelines to include traumatic fracture as a trigger for osteoporosis screening.
Commentary
Osteoporosis is one of the most common age-associated disease that affects 1 in 4 women and 1 in 20 men over the age of 65.1 It increases the risk of fracture, and its clinical sequelae include reduced mobility, health decline, and increased all-cause mortality. The high prevalence of osteoporosis poses a clinical challenge as the global population continues to age. Pharmacological treatments such as bisphosphonates are highly effective in preventing or slowing bone mineral density (BMD) loss and reducing risk of fragility fractures (eg, nontraumatic fractures of the vertebra, hip, and femur) and are commonly used to mitigate adverse effects of degenerative bone changes secondary to osteoporosis.1
The high prevalence of osteoporosis and effectiveness of bisphosphonates raises the question of how to optimally identify adults at risk for osteoporosis so that pharmacologic therapy can be promptly initiated to prevent disease progression. Multiple osteoporosis screening guidelines, including those from the United States Preventive Services Task Force (USPSTF), American Association of Family Physicians, and National Osteoporosis Foundation, are widely used in the clinical setting to address this important clinical question. In general, the prevailing wisdom is to screen osteoporosis in postmenopausal women over the age of 65, women under the age of 65 who have a significant 10-year fracture risk, or women over the age of 50 who have experienced a fragility fracture.1 In the study reported by Crandall et al, it was shown that the risks of having subsequent fractures were similar after an initial traumatic or nontraumatic (fragility) fracture in postmenopausal women aged 50 years and older.2 This finding brings into question whether traumatic fractures should be viewed any differently than nontraumatic fractures in women over the age of 50 in light of evaluation for osteoporosis. Furthermore, these results suggest that most fractures in postmenopausal women may indicate decreased bone integrity, thus adding to the rationale that osteoporosis screening needs to be considered and expanded to include postmenopausal women under the age of 65 who endured a traumatic fracture.
Per current guidelines, a woman under the age of 65 is recommended for osteoporosis screening only if she has an increased 10-year fracture risk compared to women aged 65 years and older. This risk is calculated based on the World Health Organization fracture-risk algorithm (WHO FRAX) tool which uses multiple factors such as age, weight, and history of fragility fractures to predict whether an individual is at risk of developing a fracture in the next 10 years. The WHO FRAX tool does not include traumatic fractures in its risk calculation and current clinical guidelines do not account for traumatic fractures as a red flag to initiate osteoporosis screening. Therefore, postmenopausal women under the age of 65 are less likely to be screened for osteoporosis when they experience a traumatic fracture compared to a fragility fracture, despite being at a demonstrably higher risk for subsequent fracture. As an unintended consequence, this may lead to the under diagnosis of osteoporosis in postmenopausal women under the age of 65. Thus, Crandall et al conclude that a fracture due to any cause warrants follow up evaluation for osteoporosis including BMD testing in women older than 50 years of age.
Older men constitute another population who are commonly under screened for osteoporosis. The current USPSTF guidelines indicate that there is an insufficient body of evidence to screen men for osteoporosis given its lower prevalence.1 However, it is important to note that men have significantly increased mortality after a hip fracture, are less likely to be on pharmacological treatment for osteoporosis, and are under diagnosed for osteoporosis.3 Consistent with findings from the current study, Leslie et al showed that high-trauma and low-trauma fractures have similarly elevated subsequent fracture risk in both men and women over the age of 40 in a Canadian study.4 Moreover, in the same study, BMD was decreased in both men and women who suffered a fracture regardless of the injury mechanism. This finding further underscores a need to consider traumatic fractures as a risk factor for osteoporosis. Taken together, given that men are under screened and treated for osteoporosis but have increased mortality post-fracture, considerations to initiate osteoporosis evaluation should be similarly given to men who endured a traumatic fracture.
The study conducted by Crandall et al has several strengths. It is noteworthy for the large size of the WHI cohort with participants from across the United States which enables the capture of a wider range of age groups as women under the age of 65 are not common participants of osteoporosis studies. Additionally, data ascertainment and outcome adjudication utilizing medical records and physician review assure data quality. A limitation of the study is that the study cohort consists exclusively of women and therefore the findings are not generalizable to men. However, findings from this study echo those from other studies that investigate the relationship between fracture mechanisms and subsequent fracture risk in men and women.3,4 Collectively, these comparable findings highlight the need for additional research to validate traumatic fracture as a risk factor for osteoporosis and to incorporate it into clinical guidelines for osteoporosis screening.
Applications for Clinical Practice
The findings from the current study indicate that traumatic and fragility fractures may be more alike than previously recognized in regards to bone health and subsequent fracture prevention in postmenopausal women. If validated, these results may lead to changes in clinical practice whereby all fractures in postmenopausal women could trigger osteoporosis screening, assessment, and treatment if indicated for the secondary prevention of fractures.
Study Overview
Objective. To compare the risk of subsequent fractures after an initial traumatic or nontraumatic fracture in postmenopausal women.
Design. A prospective observational study utilizing data from the Women’s Health Initiative (WHI) Study, WHI Clinical Trials (WHI-CT), and WHI Bone Density Substudy to evaluate rates at which patients who suffered a traumatic fracture vs nontraumatic fracture develop a subsequent fracture.
Setting and participants. The WHI study, implemented at 40 United States clinical sites, enrolled 161 808 postmenopausal women aged 50 to 79 years at baseline between 1993 and 1998. The study cohort consisted of 75 335 patients who had self-reported fractures from September 1994 to December 1998 that were confirmed by the WHI Bone Density Substudy and WHI-CT. Of these participants, 253 (0.3%) were excluded because of a lack of follow-up information regarding incident fractures, and 8208 (10.9%) were excluded due to incomplete information on covariates, thus resulting in an analytic sample of 66 874 (88.8%) participants. Prospective fracture ascertainment with participants was conducted at least annually and the mechanism of fracture was assessed to differentiate traumatic vs nontraumatic incident fractures. Traumatic fractures were defined as fractures caused by motor vehicle collisions, falls from a height, falls downstairs, or sports injury. Nontraumatic fractures were defined as fractures caused by a trip and fall.
Main outcome measures. The primary outcome was an incident fracture at an anatomically distinct body part. Fractures were classified as upper extremity (carpal, elbow, lower or upper end of humerus, shaft of humerus, upper radius/ulna, or radius/ulna), lower extremity (ankle, hip, patella, pelvis, shaft of femur, tibia/fibula, or tibial plateau), or spine (lumbar and/or thoracic spine). Self-reported fractures were verified via medical chart review by WHI study physicians; hip fractures were confirmed by review of written reports of radiographic studies; and nonhip fractures were confirmed by review of radiography reports or clinical documentations.
Main results. In total, 66 874 women in the study (mean [SD] age) 63.1 (7.0) years without clinical fracture and 65.3 (7.2) years with clinical fracture at baseline were followed for 8.1 (1.6) years. Of these participants, 7142 (10.7%) experienced incident fracture during the study follow-up period (13.9 per 1000 person-years), and 721 (10.1%) of whom had a subsequent fracture. The adjusted hazard ratio (aHR) of subsequent fracture after an initial fracture was 1.49 (95% CI, 1.38-1.61, P < .001). Covariates adjusted were age, race, ethnicity, body mass index, treated diabetes, frequency of falls in the previous year, and physical function and activity. In women with initial traumatic fracture, the association between initial and subsequent fracture was increased (aHR, 1.25; 95% CI, 1.06-1.48, P = .01). Among women with initial nontraumatic fracture, the association between initial and subsequent fracture was also increased (aHR, 1.52; 95% CI, 1.37-1.68, P < .001). The confidence intervals for the 2 preceding associations for traumatic and nontraumatic initial fracture strata were overlapping.
Conclusion. Fractures, regardless of mechanism of injury, are similarly associated with an increased risk of subsequent fractures in postmenopausal women aged 50 years and older. Findings from this study provide evidence to support reevaluation of current clinical guidelines to include traumatic fracture as a trigger for osteoporosis screening.
Commentary
Osteoporosis is one of the most common age-associated disease that affects 1 in 4 women and 1 in 20 men over the age of 65.1 It increases the risk of fracture, and its clinical sequelae include reduced mobility, health decline, and increased all-cause mortality. The high prevalence of osteoporosis poses a clinical challenge as the global population continues to age. Pharmacological treatments such as bisphosphonates are highly effective in preventing or slowing bone mineral density (BMD) loss and reducing risk of fragility fractures (eg, nontraumatic fractures of the vertebra, hip, and femur) and are commonly used to mitigate adverse effects of degenerative bone changes secondary to osteoporosis.1
The high prevalence of osteoporosis and effectiveness of bisphosphonates raises the question of how to optimally identify adults at risk for osteoporosis so that pharmacologic therapy can be promptly initiated to prevent disease progression. Multiple osteoporosis screening guidelines, including those from the United States Preventive Services Task Force (USPSTF), American Association of Family Physicians, and National Osteoporosis Foundation, are widely used in the clinical setting to address this important clinical question. In general, the prevailing wisdom is to screen osteoporosis in postmenopausal women over the age of 65, women under the age of 65 who have a significant 10-year fracture risk, or women over the age of 50 who have experienced a fragility fracture.1 In the study reported by Crandall et al, it was shown that the risks of having subsequent fractures were similar after an initial traumatic or nontraumatic (fragility) fracture in postmenopausal women aged 50 years and older.2 This finding brings into question whether traumatic fractures should be viewed any differently than nontraumatic fractures in women over the age of 50 in light of evaluation for osteoporosis. Furthermore, these results suggest that most fractures in postmenopausal women may indicate decreased bone integrity, thus adding to the rationale that osteoporosis screening needs to be considered and expanded to include postmenopausal women under the age of 65 who endured a traumatic fracture.
Per current guidelines, a woman under the age of 65 is recommended for osteoporosis screening only if she has an increased 10-year fracture risk compared to women aged 65 years and older. This risk is calculated based on the World Health Organization fracture-risk algorithm (WHO FRAX) tool which uses multiple factors such as age, weight, and history of fragility fractures to predict whether an individual is at risk of developing a fracture in the next 10 years. The WHO FRAX tool does not include traumatic fractures in its risk calculation and current clinical guidelines do not account for traumatic fractures as a red flag to initiate osteoporosis screening. Therefore, postmenopausal women under the age of 65 are less likely to be screened for osteoporosis when they experience a traumatic fracture compared to a fragility fracture, despite being at a demonstrably higher risk for subsequent fracture. As an unintended consequence, this may lead to the under diagnosis of osteoporosis in postmenopausal women under the age of 65. Thus, Crandall et al conclude that a fracture due to any cause warrants follow up evaluation for osteoporosis including BMD testing in women older than 50 years of age.
Older men constitute another population who are commonly under screened for osteoporosis. The current USPSTF guidelines indicate that there is an insufficient body of evidence to screen men for osteoporosis given its lower prevalence.1 However, it is important to note that men have significantly increased mortality after a hip fracture, are less likely to be on pharmacological treatment for osteoporosis, and are under diagnosed for osteoporosis.3 Consistent with findings from the current study, Leslie et al showed that high-trauma and low-trauma fractures have similarly elevated subsequent fracture risk in both men and women over the age of 40 in a Canadian study.4 Moreover, in the same study, BMD was decreased in both men and women who suffered a fracture regardless of the injury mechanism. This finding further underscores a need to consider traumatic fractures as a risk factor for osteoporosis. Taken together, given that men are under screened and treated for osteoporosis but have increased mortality post-fracture, considerations to initiate osteoporosis evaluation should be similarly given to men who endured a traumatic fracture.
The study conducted by Crandall et al has several strengths. It is noteworthy for the large size of the WHI cohort with participants from across the United States which enables the capture of a wider range of age groups as women under the age of 65 are not common participants of osteoporosis studies. Additionally, data ascertainment and outcome adjudication utilizing medical records and physician review assure data quality. A limitation of the study is that the study cohort consists exclusively of women and therefore the findings are not generalizable to men. However, findings from this study echo those from other studies that investigate the relationship between fracture mechanisms and subsequent fracture risk in men and women.3,4 Collectively, these comparable findings highlight the need for additional research to validate traumatic fracture as a risk factor for osteoporosis and to incorporate it into clinical guidelines for osteoporosis screening.
Applications for Clinical Practice
The findings from the current study indicate that traumatic and fragility fractures may be more alike than previously recognized in regards to bone health and subsequent fracture prevention in postmenopausal women. If validated, these results may lead to changes in clinical practice whereby all fractures in postmenopausal women could trigger osteoporosis screening, assessment, and treatment if indicated for the secondary prevention of fractures.
1. US Preventive Services Task Force, Curry SJ, Krist Ah, et al. Screening for Osteoporosis to Prevent Fractures: US Preventive Services Task Force Recommendation Statement. JAMA. 2018;319(24):2521–2531. doi:10.1001/jama.2018.7498
2. Crandall CJ, Larson JC, LaCroix AZ, et al. Risk of Subsequent Fractures in Postmenopausal Women After Nontraumatic vs Traumatic Fractures. JAMA Intern Med. Published online June 7, 2021. doi:10.1001/jamainternmed.2021.2617
3. Mackey DC, Lui L, Cawthon PM, et al. High-Trauma Fractures and Low Bone Mineral Density in Older Women and Men. JAMA. 2007;298(20):2381–2388. doi:10.1001/jama.298.20.2381
4. Leslie WD, Schousboe JT, Morin SN, et al. Fracture risk following high-trauma versus low-trauma fracture: a registry-based cohort study. Osteoporos Int. 2020;31(6):1059–1067. doi:10.1007/s00198-019-05274-2
1. US Preventive Services Task Force, Curry SJ, Krist Ah, et al. Screening for Osteoporosis to Prevent Fractures: US Preventive Services Task Force Recommendation Statement. JAMA. 2018;319(24):2521–2531. doi:10.1001/jama.2018.7498
2. Crandall CJ, Larson JC, LaCroix AZ, et al. Risk of Subsequent Fractures in Postmenopausal Women After Nontraumatic vs Traumatic Fractures. JAMA Intern Med. Published online June 7, 2021. doi:10.1001/jamainternmed.2021.2617
3. Mackey DC, Lui L, Cawthon PM, et al. High-Trauma Fractures and Low Bone Mineral Density in Older Women and Men. JAMA. 2007;298(20):2381–2388. doi:10.1001/jama.298.20.2381
4. Leslie WD, Schousboe JT, Morin SN, et al. Fracture risk following high-trauma versus low-trauma fracture: a registry-based cohort study. Osteoporos Int. 2020;31(6):1059–1067. doi:10.1007/s00198-019-05274-2
I Never Wanted To Be a Hero
I have been in the business of medicine for more than 15 years and I will never forget the initial surge of the COVID-19 pandemic in Massachusetts.
As a hospitalist, I admitted patients infected with COVID-19, followed them on the floor, and, since I had some experience working in an intensive care unit (ICU), was assigned to cover a “COVID ICU.” This wing of the hospital used to be a fancy orthopedic floor that our institution was lucky enough to have. So began the most life-changing experience in my career as a physician.
In this role, we witness death more than any of us would care to discuss. It comes with the territory, and we never expected this to change once COVID hit. However, so many patients succumbed to this disease, especially during the first surge, which made it difficult to handle emotionally. Patients that fell ill initially stayed isolated at home, optimistic they would turn the corner only to enter the hospital a week later after their conditioned worsened. After requiring a couple of liters of supplemental oxygen in the emergency room, they eventually ended up on a high flow nasal cannula in just a matter of hours.
Patients slowly got sicker and felt more helpless as the days passed, leading us to prescribe drugs that eventually proved to have no benefit. We checked countless inflammatory markers, most of which we were not even sure what to do with. Many times, we hosted a family meeting via FaceTime, holding a patient’s hand in one hand and an iPad in the other to discuss goals of care. Too often, a dark cloud hung over these discussions, a realization that there was not much else we could do.
I have always felt that helping someone have a decent and peaceful death is important, especially when the prognosis is grim, and that patient is suffering. But the sheer number of times this happened during the initial surge of the pandemic was difficult to handle. It felt like I had more of those discussions in 3 months than I did during my entire career as a hospitalist.
We helped plenty of people get better, with some heading home in a week. They thanked us, painted rocks and the sidewalks in front of the hospital displaying messages of gratitude, and sent lunches. Others, though, left the hospital 2 months later with a tube in their stomach so they could receive some form of nutrition and another in their neck to help them breathe.
These struggles were by no means special to me; other hospitalists around the world faced similar situations at one point or another during the pandemic. Working overtime, coming home late, exhausted, undressing in the garage, trying to be there for my 3 kids who were full of energy after a whole day of Zoom and doing the usual kid stuff. My house used to have strict rules about screen time. No more.
The summer months provided a bit of a COVID break, with only 1 or 2 infected patients entering my care. We went to outdoor restaurants and tried to get our lives back to “normal.” As the weather turned cold, however, things went south again. This time no more hydroxychloroquine, a drug used to fight malaria but also treat other autoimmune diseases, as it was proven eventually over many studies that it is not helpful and was potentially harmful. We instead shifted our focus to remdesivir—an antiviral drug that displayed some benefits—tocilizumab, and dexamethasone, anti-inflammatory drugs with the latter providing some positive outcomes on mortality.
Patient survival rates improved slightly, likely due to a combination of factors. We were more experienced at fighting the disease, which led to things in the hospital not being as chaotic and more time available to spend with the patients. Personal protective equipment (PPE) and tests were more readily available, and the population getting hit by the disease changed slightly with fewer elderly people from nursing homes falling ill because of social distancing, other safety measures, or having already fought the disease. Our attention turned instead to more young people that had returned to work and their social lives.
The arrival of the vaccines brought considerable relief. I remember a few decades ago debating and sometimes fighting with friends and family over who was better: Iron Man or Spider-Man. Now I found myself having the same conversation about the Pfizer and Moderna COVID vaccines.
Summer 2021 holds significantly more promise. Most of the adult population is getting vaccinated, and I am very hopeful that we are approaching the end of this nightmare. In June, our office received word that we could remove our masks if we were fully vaccinated. It felt weird, but represented another sign that things are improving. I took my kids to the mall and removed my mask. It felt odd considering how that little blue thing became part of me during the pandemic. It also felt strange to not prescribe a single dose of remdesivir for an entire month.
It feels good—and normal—to care for the patients that we neglected for a year. It has been a needed boost to see patients return to their health care providers for their colonoscopy screenings, mammograms, and managing chronic problems like coronary artery disease, congestive heart failure, or receiving chemotherapy.
I learned plenty from this pandemic and hope I am not alone. I learned to be humble. We started with a drug that was harmful, moved on to a drug that is probably neutral and eventually were able to come up with a drug that seems to decrease mortality at least in some COVID patients. I learned it is fine to try new therapies based on the best data in the hope they result in positive clinical outcomes. However, it is critical that we all keep an eye on the rapidly evolving literature and adjust our behavior accordingly.
I also learned, or relearned, that if people are desperate enough, they will drink bleach to see if it works. Others are convinced that the purpose of vaccination is to inject a microchip allowing ourselves to be tracked by some higher power. I learned that we must take the first step to prepare for the next pandemic by having a decent reserve of PPE.
It is clear synthetic messenger RNA (mRNA) technology is here to stay, and I believe it has a huge potential to change many areas of medicine. mRNA vaccines proved to be much faster to develop and probably much easier to change as the pathogen, in this case coronavirus, changes.
The technology could be used against a variety of infectious diseases to make vaccines against malaria, tuberculosis, HIV, or hepatitis. It can also be very useful for faster vaccine development needed in future possible pandemics such as influenza, Ebola, or severe acute respiratory syndrome. It may also be used for cancer treatment.
As John P. Cooke, MD, PhD, the medical director for the Center of RNA Therapeutics Program at the Houston Methodist Research Institute, said, “Most vaccines today are still viral vaccines – they are inactivated virus, so it’s potentially infectious and you have to have virus on hand. With mRNA, you’re just writing code which is going to tell the cell to make a viral protein – one part of a viral protein to stimulate an immune response. And, here’s the wonderful thing, you don’t even need the virus in hand, just its DNA code.”1
Corresponding author: Dragos Vesbianu, MD, Attending Hospitalist, Newton-Wellesley Hospital, 2014 Washington St, Newton, MA 02462; [email protected].
Financial dislosures: None.
1. Houston Methodist. Messenger RNA – the Therapy of the Future. Newswise. November 16, 2020. Accessed June 25, 2021. https://www.newswise.com/coronavirus/messenger-rna-the-therapy-of-the-future/
I have been in the business of medicine for more than 15 years and I will never forget the initial surge of the COVID-19 pandemic in Massachusetts.
As a hospitalist, I admitted patients infected with COVID-19, followed them on the floor, and, since I had some experience working in an intensive care unit (ICU), was assigned to cover a “COVID ICU.” This wing of the hospital used to be a fancy orthopedic floor that our institution was lucky enough to have. So began the most life-changing experience in my career as a physician.
In this role, we witness death more than any of us would care to discuss. It comes with the territory, and we never expected this to change once COVID hit. However, so many patients succumbed to this disease, especially during the first surge, which made it difficult to handle emotionally. Patients that fell ill initially stayed isolated at home, optimistic they would turn the corner only to enter the hospital a week later after their conditioned worsened. After requiring a couple of liters of supplemental oxygen in the emergency room, they eventually ended up on a high flow nasal cannula in just a matter of hours.
Patients slowly got sicker and felt more helpless as the days passed, leading us to prescribe drugs that eventually proved to have no benefit. We checked countless inflammatory markers, most of which we were not even sure what to do with. Many times, we hosted a family meeting via FaceTime, holding a patient’s hand in one hand and an iPad in the other to discuss goals of care. Too often, a dark cloud hung over these discussions, a realization that there was not much else we could do.
I have always felt that helping someone have a decent and peaceful death is important, especially when the prognosis is grim, and that patient is suffering. But the sheer number of times this happened during the initial surge of the pandemic was difficult to handle. It felt like I had more of those discussions in 3 months than I did during my entire career as a hospitalist.
We helped plenty of people get better, with some heading home in a week. They thanked us, painted rocks and the sidewalks in front of the hospital displaying messages of gratitude, and sent lunches. Others, though, left the hospital 2 months later with a tube in their stomach so they could receive some form of nutrition and another in their neck to help them breathe.
These struggles were by no means special to me; other hospitalists around the world faced similar situations at one point or another during the pandemic. Working overtime, coming home late, exhausted, undressing in the garage, trying to be there for my 3 kids who were full of energy after a whole day of Zoom and doing the usual kid stuff. My house used to have strict rules about screen time. No more.
The summer months provided a bit of a COVID break, with only 1 or 2 infected patients entering my care. We went to outdoor restaurants and tried to get our lives back to “normal.” As the weather turned cold, however, things went south again. This time no more hydroxychloroquine, a drug used to fight malaria but also treat other autoimmune diseases, as it was proven eventually over many studies that it is not helpful and was potentially harmful. We instead shifted our focus to remdesivir—an antiviral drug that displayed some benefits—tocilizumab, and dexamethasone, anti-inflammatory drugs with the latter providing some positive outcomes on mortality.
Patient survival rates improved slightly, likely due to a combination of factors. We were more experienced at fighting the disease, which led to things in the hospital not being as chaotic and more time available to spend with the patients. Personal protective equipment (PPE) and tests were more readily available, and the population getting hit by the disease changed slightly with fewer elderly people from nursing homes falling ill because of social distancing, other safety measures, or having already fought the disease. Our attention turned instead to more young people that had returned to work and their social lives.
The arrival of the vaccines brought considerable relief. I remember a few decades ago debating and sometimes fighting with friends and family over who was better: Iron Man or Spider-Man. Now I found myself having the same conversation about the Pfizer and Moderna COVID vaccines.
Summer 2021 holds significantly more promise. Most of the adult population is getting vaccinated, and I am very hopeful that we are approaching the end of this nightmare. In June, our office received word that we could remove our masks if we were fully vaccinated. It felt weird, but represented another sign that things are improving. I took my kids to the mall and removed my mask. It felt odd considering how that little blue thing became part of me during the pandemic. It also felt strange to not prescribe a single dose of remdesivir for an entire month.
It feels good—and normal—to care for the patients that we neglected for a year. It has been a needed boost to see patients return to their health care providers for their colonoscopy screenings, mammograms, and managing chronic problems like coronary artery disease, congestive heart failure, or receiving chemotherapy.
I learned plenty from this pandemic and hope I am not alone. I learned to be humble. We started with a drug that was harmful, moved on to a drug that is probably neutral and eventually were able to come up with a drug that seems to decrease mortality at least in some COVID patients. I learned it is fine to try new therapies based on the best data in the hope they result in positive clinical outcomes. However, it is critical that we all keep an eye on the rapidly evolving literature and adjust our behavior accordingly.
I also learned, or relearned, that if people are desperate enough, they will drink bleach to see if it works. Others are convinced that the purpose of vaccination is to inject a microchip allowing ourselves to be tracked by some higher power. I learned that we must take the first step to prepare for the next pandemic by having a decent reserve of PPE.
It is clear synthetic messenger RNA (mRNA) technology is here to stay, and I believe it has a huge potential to change many areas of medicine. mRNA vaccines proved to be much faster to develop and probably much easier to change as the pathogen, in this case coronavirus, changes.
The technology could be used against a variety of infectious diseases to make vaccines against malaria, tuberculosis, HIV, or hepatitis. It can also be very useful for faster vaccine development needed in future possible pandemics such as influenza, Ebola, or severe acute respiratory syndrome. It may also be used for cancer treatment.
As John P. Cooke, MD, PhD, the medical director for the Center of RNA Therapeutics Program at the Houston Methodist Research Institute, said, “Most vaccines today are still viral vaccines – they are inactivated virus, so it’s potentially infectious and you have to have virus on hand. With mRNA, you’re just writing code which is going to tell the cell to make a viral protein – one part of a viral protein to stimulate an immune response. And, here’s the wonderful thing, you don’t even need the virus in hand, just its DNA code.”1
Corresponding author: Dragos Vesbianu, MD, Attending Hospitalist, Newton-Wellesley Hospital, 2014 Washington St, Newton, MA 02462; [email protected].
Financial dislosures: None.
I have been in the business of medicine for more than 15 years and I will never forget the initial surge of the COVID-19 pandemic in Massachusetts.
As a hospitalist, I admitted patients infected with COVID-19, followed them on the floor, and, since I had some experience working in an intensive care unit (ICU), was assigned to cover a “COVID ICU.” This wing of the hospital used to be a fancy orthopedic floor that our institution was lucky enough to have. So began the most life-changing experience in my career as a physician.
In this role, we witness death more than any of us would care to discuss. It comes with the territory, and we never expected this to change once COVID hit. However, so many patients succumbed to this disease, especially during the first surge, which made it difficult to handle emotionally. Patients that fell ill initially stayed isolated at home, optimistic they would turn the corner only to enter the hospital a week later after their conditioned worsened. After requiring a couple of liters of supplemental oxygen in the emergency room, they eventually ended up on a high flow nasal cannula in just a matter of hours.
Patients slowly got sicker and felt more helpless as the days passed, leading us to prescribe drugs that eventually proved to have no benefit. We checked countless inflammatory markers, most of which we were not even sure what to do with. Many times, we hosted a family meeting via FaceTime, holding a patient’s hand in one hand and an iPad in the other to discuss goals of care. Too often, a dark cloud hung over these discussions, a realization that there was not much else we could do.
I have always felt that helping someone have a decent and peaceful death is important, especially when the prognosis is grim, and that patient is suffering. But the sheer number of times this happened during the initial surge of the pandemic was difficult to handle. It felt like I had more of those discussions in 3 months than I did during my entire career as a hospitalist.
We helped plenty of people get better, with some heading home in a week. They thanked us, painted rocks and the sidewalks in front of the hospital displaying messages of gratitude, and sent lunches. Others, though, left the hospital 2 months later with a tube in their stomach so they could receive some form of nutrition and another in their neck to help them breathe.
These struggles were by no means special to me; other hospitalists around the world faced similar situations at one point or another during the pandemic. Working overtime, coming home late, exhausted, undressing in the garage, trying to be there for my 3 kids who were full of energy after a whole day of Zoom and doing the usual kid stuff. My house used to have strict rules about screen time. No more.
The summer months provided a bit of a COVID break, with only 1 or 2 infected patients entering my care. We went to outdoor restaurants and tried to get our lives back to “normal.” As the weather turned cold, however, things went south again. This time no more hydroxychloroquine, a drug used to fight malaria but also treat other autoimmune diseases, as it was proven eventually over many studies that it is not helpful and was potentially harmful. We instead shifted our focus to remdesivir—an antiviral drug that displayed some benefits—tocilizumab, and dexamethasone, anti-inflammatory drugs with the latter providing some positive outcomes on mortality.
Patient survival rates improved slightly, likely due to a combination of factors. We were more experienced at fighting the disease, which led to things in the hospital not being as chaotic and more time available to spend with the patients. Personal protective equipment (PPE) and tests were more readily available, and the population getting hit by the disease changed slightly with fewer elderly people from nursing homes falling ill because of social distancing, other safety measures, or having already fought the disease. Our attention turned instead to more young people that had returned to work and their social lives.
The arrival of the vaccines brought considerable relief. I remember a few decades ago debating and sometimes fighting with friends and family over who was better: Iron Man or Spider-Man. Now I found myself having the same conversation about the Pfizer and Moderna COVID vaccines.
Summer 2021 holds significantly more promise. Most of the adult population is getting vaccinated, and I am very hopeful that we are approaching the end of this nightmare. In June, our office received word that we could remove our masks if we were fully vaccinated. It felt weird, but represented another sign that things are improving. I took my kids to the mall and removed my mask. It felt odd considering how that little blue thing became part of me during the pandemic. It also felt strange to not prescribe a single dose of remdesivir for an entire month.
It feels good—and normal—to care for the patients that we neglected for a year. It has been a needed boost to see patients return to their health care providers for their colonoscopy screenings, mammograms, and managing chronic problems like coronary artery disease, congestive heart failure, or receiving chemotherapy.
I learned plenty from this pandemic and hope I am not alone. I learned to be humble. We started with a drug that was harmful, moved on to a drug that is probably neutral and eventually were able to come up with a drug that seems to decrease mortality at least in some COVID patients. I learned it is fine to try new therapies based on the best data in the hope they result in positive clinical outcomes. However, it is critical that we all keep an eye on the rapidly evolving literature and adjust our behavior accordingly.
I also learned, or relearned, that if people are desperate enough, they will drink bleach to see if it works. Others are convinced that the purpose of vaccination is to inject a microchip allowing ourselves to be tracked by some higher power. I learned that we must take the first step to prepare for the next pandemic by having a decent reserve of PPE.
It is clear synthetic messenger RNA (mRNA) technology is here to stay, and I believe it has a huge potential to change many areas of medicine. mRNA vaccines proved to be much faster to develop and probably much easier to change as the pathogen, in this case coronavirus, changes.
The technology could be used against a variety of infectious diseases to make vaccines against malaria, tuberculosis, HIV, or hepatitis. It can also be very useful for faster vaccine development needed in future possible pandemics such as influenza, Ebola, or severe acute respiratory syndrome. It may also be used for cancer treatment.
As John P. Cooke, MD, PhD, the medical director for the Center of RNA Therapeutics Program at the Houston Methodist Research Institute, said, “Most vaccines today are still viral vaccines – they are inactivated virus, so it’s potentially infectious and you have to have virus on hand. With mRNA, you’re just writing code which is going to tell the cell to make a viral protein – one part of a viral protein to stimulate an immune response. And, here’s the wonderful thing, you don’t even need the virus in hand, just its DNA code.”1
Corresponding author: Dragos Vesbianu, MD, Attending Hospitalist, Newton-Wellesley Hospital, 2014 Washington St, Newton, MA 02462; [email protected].
Financial dislosures: None.
1. Houston Methodist. Messenger RNA – the Therapy of the Future. Newswise. November 16, 2020. Accessed June 25, 2021. https://www.newswise.com/coronavirus/messenger-rna-the-therapy-of-the-future/
1. Houston Methodist. Messenger RNA – the Therapy of the Future. Newswise. November 16, 2020. Accessed June 25, 2021. https://www.newswise.com/coronavirus/messenger-rna-the-therapy-of-the-future/