User login
For MD-IQ use only
Student loan management: An introduction for the young gastroenterologist
The young gastroenterologist has no shortage of personal finance topics to juggle, ranging from investments, to life and disability coverage, and planning for retirement. But the elephant in the room is student loan management. Average medical student debt today is approximately $240,000, and debt burdens greater than $300,000 are becoming common.1,2 With this staggering amount of debt, it is understandable why student loans are a major source of anxiety. Here, I will provide a brief introduction to student loan management for gastroenterologists.
Student loans: Basic strategy
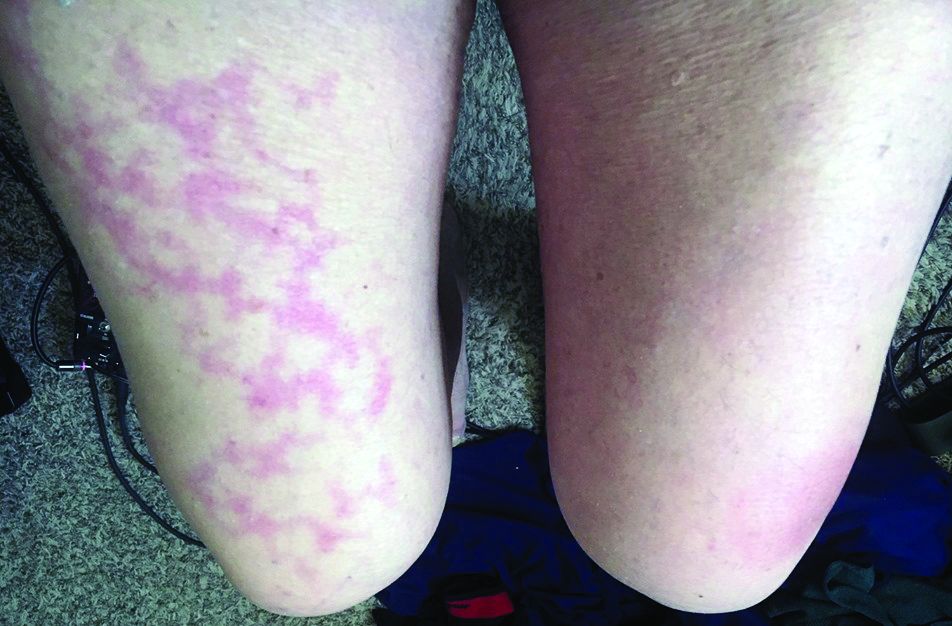
It is important to distinguish between two major types of loans: private student loans and direct federal loans. With private student loans the best strategy in most cases is to refinance to a lower interest rate. For direct federal loans, however, the decision making is more complex. There are two major approaches to these federal loans – either 1) refinance, or 2) go for public service loan forgiveness (PSLF). See Figure 1 for a flowchart summarizing my general approach to student loan management.
Refinance basics
One potential approach is to refinance your federal loans. Most federal loans today are at a relatively high interest rate of 6%-8%.3 Private refinancing can yield rates in the 3%-5% range, depending on the type of loan and other factors. For a loan balance of $200,000, the savings by refinancing could be approximately $2,000-$10,000 per year in interest alone. However, refinancing your loans with a private company eliminates the possibility of PSLF. Hence, you should only refinance federal loans once you are sure that you will not be pursuing PSLF. You may refinance your private loans anytime since they do not qualify for PSLF. There are multiple companies that provide student loan refinancing. The process can be done online, sometimes in as little as 30 minutes. There is generally little or no cost to refinancing, and many companies even provide a small cash-back incentive to refinance.
PSLF basics
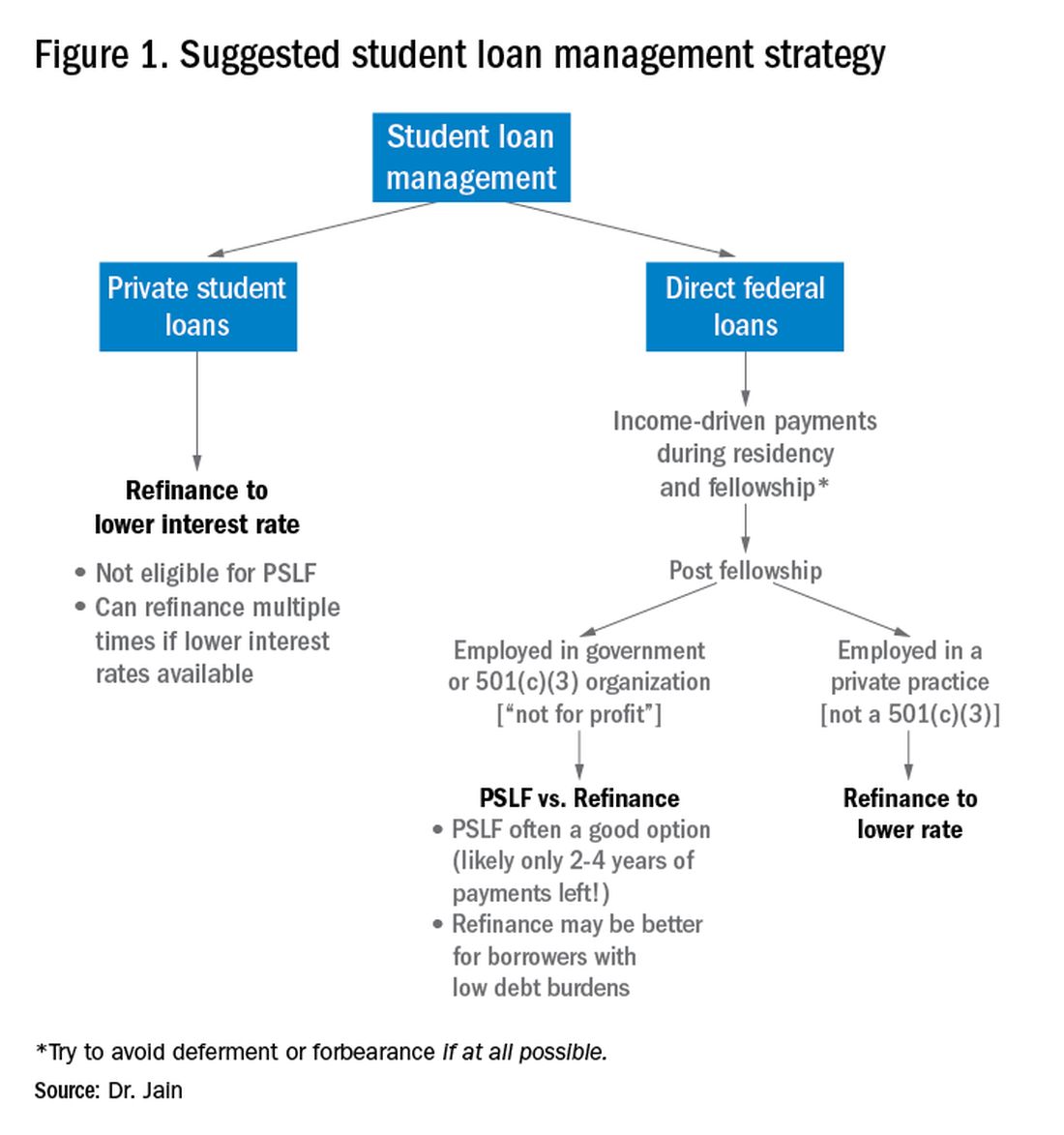
The PSLF program allows borrowers to have the remainder of their direct loans forgiven after 10 years (120 monthly payments) under a qualifying income-driven repayment (IDR) plan.4 Figure 2 shows an overview of the various IDR plans. During the 120 payments, the borrower must work full time for a qualifying employer, which includes a government employer or a not-for-profit 501(c)(3) organization. Loan forgiveness with PSLF is completely tax free. Importantly, the PSLF program only applies to direct federal loans. You can see your federal loan types and balances by visiting https://studentaid.gov/.
To PSLF or not to PSLF?
With direct federal loans, the decision to refinance or go for PSLF is a major fork in the road. PSLF can be a good option for borrowers with long training programs and with high student loan burdens (e.g., loan-to-income ratios of 1:1, 2:1 or higher). By contrast, borrowers with short training programs or relatively small loan burdens may be better off refinancing to a low interest rate and paying off loans quickly. Virtually all institutions that train residents and fellows are qualified government or 501(c)(3) organizations. Hence, a gastroenterology graduate generally will have completed at least 6 out of 10 years of payments by the end of training. Trainees who did a chief resident year or gastroenterology research track may have completed 7 or 8 years of qualifying payments already.
For trainees who are already planning an academic career, PSLF is often a good option. While PSLF can be a nice benefit, I would not advise making a career decision purely based on PSLF. Private practice jobs generally come with substantially higher salaries than academic and government jobs. This salary differential typically more than compensates for the loss of access to PSLF. Hence, I advise trainees to choose the practice setting that is best for their personal and career satisfaction, and then build a student loan management plan around that. The exception may be the trainee who has a very large student loan burden (e.g., loan-to-income ratio of 2:1 or 3:1).
Caveats with PSLF
There have been well publicized concerns about the future of PSLF, including proposals to eliminate or cap the program.5,6 However, most proposed legislation has only recommended changes to PSLF for new borrowers. If you currently have existing federal loans, you would very likely be grandfathered into the existing PSLF terms. All federal master promissory notes since 2007 have cited PSLF as a loan repayment option.7 Hence, eliminating PSLF for existing borrowers seems unlikely since it would be changing the terms of an executed contract.8
There have also been widespread reports of high numbers of borrowers being denied applications for PSLF.9,10 However, the majority of these applicants did not have correct types of loans, had not worked full time for qualifying employers or had not made the full 120 payments.11 Yet some denials have apparently resulted from errors in tracking qualifying payments by FedLoan servicing.12 Therefore it would be prudent to keep your own careful records of all qualifying payments towards PSLF.
The nuclear option: 20- to 25-year IDR-based forgiveness
An additional option allows borrowers to make IDRs for 20-25 years (details in Figure 2) and then having their remaining loan balance forgiven.13 This option is completely independent of PSLF. Borrowers can work full time or part time and can work for any employer, including private employers.
One additional option: NIH loan repayment programs
One additional solution to consider are the NIH Loan Repayment Programs (LRPs). These programs can provide substantial loan repayment (up to $50,000 annually) for trainees and attendings engaged in research that aligns with NIH priorities, including clinical research or health disparities research.14 Notably, the applicant’s research does not have to be NIH sponsored research.
Getting more information
The approach above is a general overview of student loan concepts for gastroenterologists. However, there are countless nuances and tactics that are beyond the scope of this introductory article. I encourage everyone to get additional information and advice when making your own loan management plan. There are many helpful online resources, podcasts, and books discussing the topic. Several companies provide detailed consultation on managing student loans. Such services may cost a few hundred dollars but could potentially save tens of thousands of dollars on student loan costs.
Dr. Jain is assistant professor of medicine, division of gastroenterology & hepatology, department of medicine, University of North Carolina School of Medicine, Chapel Hill. Dr. Jain has no conflicts of interest and no funding source.
References
1. https://nces.ed.gov/programs/digest/d18/tables/dt18_332.45.asp
2. https://www.credible.com/blog/statistics/average-medical-school-debt/
3. https://studentaid.gov/understand-aid/types/loans/interest-rates
4. https://studentaid.gov/manage-loans/forgiveness-cancellation/public-service
5. https://www.forbes.com/sites/robertfarrington/2019/09/24/how-to-get-your-public-service-loan-forgiveness-qualifying-payments-recounted/#18567f061f5d
6. https://www.cbo.gov/budget-options/2018/54721
7. https://static.studentloans.gov/images/ApplicationAndPromissoryNote.pdf
8. https://www.biglawinvestor.com/pslf-promissory-note/
9. https://bostonstudentloanlawyer.com/scary-stats-for-public-service-loan-forgiveness/
10. https://www.marketwatch.com/story/this-government-loan-forgiveness-program-has-rejected-99-of-borrowers-so-far-2018-09-20
11. https://studentaid.gov/data-center/student/loan-forgiveness/pslf-data
12. https://www.nytimes.com/2019/04/12/your-money/public-service-loan-forgiveness.html
13. https://studentaid.gov/manage-loans/repayment/plans/income-driven
14. https://www.lrp.nih.gov/eligibility-programs
The young gastroenterologist has no shortage of personal finance topics to juggle, ranging from investments, to life and disability coverage, and planning for retirement. But the elephant in the room is student loan management. Average medical student debt today is approximately $240,000, and debt burdens greater than $300,000 are becoming common.1,2 With this staggering amount of debt, it is understandable why student loans are a major source of anxiety. Here, I will provide a brief introduction to student loan management for gastroenterologists.
Student loans: Basic strategy
It is important to distinguish between two major types of loans: private student loans and direct federal loans. With private student loans the best strategy in most cases is to refinance to a lower interest rate. For direct federal loans, however, the decision making is more complex. There are two major approaches to these federal loans – either 1) refinance, or 2) go for public service loan forgiveness (PSLF). See Figure 1 for a flowchart summarizing my general approach to student loan management.

Refinance basics
One potential approach is to refinance your federal loans. Most federal loans today are at a relatively high interest rate of 6%-8%.3 Private refinancing can yield rates in the 3%-5% range, depending on the type of loan and other factors. For a loan balance of $200,000, the savings by refinancing could be approximately $2,000-$10,000 per year in interest alone. However, refinancing your loans with a private company eliminates the possibility of PSLF. Hence, you should only refinance federal loans once you are sure that you will not be pursuing PSLF. You may refinance your private loans anytime since they do not qualify for PSLF. There are multiple companies that provide student loan refinancing. The process can be done online, sometimes in as little as 30 minutes. There is generally little or no cost to refinancing, and many companies even provide a small cash-back incentive to refinance.
PSLF basics
The PSLF program allows borrowers to have the remainder of their direct loans forgiven after 10 years (120 monthly payments) under a qualifying income-driven repayment (IDR) plan.4 Figure 2 shows an overview of the various IDR plans. During the 120 payments, the borrower must work full time for a qualifying employer, which includes a government employer or a not-for-profit 501(c)(3) organization. Loan forgiveness with PSLF is completely tax free. Importantly, the PSLF program only applies to direct federal loans. You can see your federal loan types and balances by visiting https://studentaid.gov/.
To PSLF or not to PSLF?
With direct federal loans, the decision to refinance or go for PSLF is a major fork in the road. PSLF can be a good option for borrowers with long training programs and with high student loan burdens (e.g., loan-to-income ratios of 1:1, 2:1 or higher). By contrast, borrowers with short training programs or relatively small loan burdens may be better off refinancing to a low interest rate and paying off loans quickly. Virtually all institutions that train residents and fellows are qualified government or 501(c)(3) organizations. Hence, a gastroenterology graduate generally will have completed at least 6 out of 10 years of payments by the end of training. Trainees who did a chief resident year or gastroenterology research track may have completed 7 or 8 years of qualifying payments already.
For trainees who are already planning an academic career, PSLF is often a good option. While PSLF can be a nice benefit, I would not advise making a career decision purely based on PSLF. Private practice jobs generally come with substantially higher salaries than academic and government jobs. This salary differential typically more than compensates for the loss of access to PSLF. Hence, I advise trainees to choose the practice setting that is best for their personal and career satisfaction, and then build a student loan management plan around that. The exception may be the trainee who has a very large student loan burden (e.g., loan-to-income ratio of 2:1 or 3:1).
Caveats with PSLF
There have been well publicized concerns about the future of PSLF, including proposals to eliminate or cap the program.5,6 However, most proposed legislation has only recommended changes to PSLF for new borrowers. If you currently have existing federal loans, you would very likely be grandfathered into the existing PSLF terms. All federal master promissory notes since 2007 have cited PSLF as a loan repayment option.7 Hence, eliminating PSLF for existing borrowers seems unlikely since it would be changing the terms of an executed contract.8
There have also been widespread reports of high numbers of borrowers being denied applications for PSLF.9,10 However, the majority of these applicants did not have correct types of loans, had not worked full time for qualifying employers or had not made the full 120 payments.11 Yet some denials have apparently resulted from errors in tracking qualifying payments by FedLoan servicing.12 Therefore it would be prudent to keep your own careful records of all qualifying payments towards PSLF.
The nuclear option: 20- to 25-year IDR-based forgiveness
An additional option allows borrowers to make IDRs for 20-25 years (details in Figure 2) and then having their remaining loan balance forgiven.13 This option is completely independent of PSLF. Borrowers can work full time or part time and can work for any employer, including private employers.

One additional option: NIH loan repayment programs
One additional solution to consider are the NIH Loan Repayment Programs (LRPs). These programs can provide substantial loan repayment (up to $50,000 annually) for trainees and attendings engaged in research that aligns with NIH priorities, including clinical research or health disparities research.14 Notably, the applicant’s research does not have to be NIH sponsored research.
Getting more information
The approach above is a general overview of student loan concepts for gastroenterologists. However, there are countless nuances and tactics that are beyond the scope of this introductory article. I encourage everyone to get additional information and advice when making your own loan management plan. There are many helpful online resources, podcasts, and books discussing the topic. Several companies provide detailed consultation on managing student loans. Such services may cost a few hundred dollars but could potentially save tens of thousands of dollars on student loan costs.
Dr. Jain is assistant professor of medicine, division of gastroenterology & hepatology, department of medicine, University of North Carolina School of Medicine, Chapel Hill. Dr. Jain has no conflicts of interest and no funding source.
References
1. https://nces.ed.gov/programs/digest/d18/tables/dt18_332.45.asp
2. https://www.credible.com/blog/statistics/average-medical-school-debt/
3. https://studentaid.gov/understand-aid/types/loans/interest-rates
4. https://studentaid.gov/manage-loans/forgiveness-cancellation/public-service
5. https://www.forbes.com/sites/robertfarrington/2019/09/24/how-to-get-your-public-service-loan-forgiveness-qualifying-payments-recounted/#18567f061f5d
6. https://www.cbo.gov/budget-options/2018/54721
7. https://static.studentloans.gov/images/ApplicationAndPromissoryNote.pdf
8. https://www.biglawinvestor.com/pslf-promissory-note/
9. https://bostonstudentloanlawyer.com/scary-stats-for-public-service-loan-forgiveness/
10. https://www.marketwatch.com/story/this-government-loan-forgiveness-program-has-rejected-99-of-borrowers-so-far-2018-09-20
11. https://studentaid.gov/data-center/student/loan-forgiveness/pslf-data
12. https://www.nytimes.com/2019/04/12/your-money/public-service-loan-forgiveness.html
13. https://studentaid.gov/manage-loans/repayment/plans/income-driven
14. https://www.lrp.nih.gov/eligibility-programs
The young gastroenterologist has no shortage of personal finance topics to juggle, ranging from investments, to life and disability coverage, and planning for retirement. But the elephant in the room is student loan management. Average medical student debt today is approximately $240,000, and debt burdens greater than $300,000 are becoming common.1,2 With this staggering amount of debt, it is understandable why student loans are a major source of anxiety. Here, I will provide a brief introduction to student loan management for gastroenterologists.
Student loans: Basic strategy
It is important to distinguish between two major types of loans: private student loans and direct federal loans. With private student loans the best strategy in most cases is to refinance to a lower interest rate. For direct federal loans, however, the decision making is more complex. There are two major approaches to these federal loans – either 1) refinance, or 2) go for public service loan forgiveness (PSLF). See Figure 1 for a flowchart summarizing my general approach to student loan management.

Refinance basics
One potential approach is to refinance your federal loans. Most federal loans today are at a relatively high interest rate of 6%-8%.3 Private refinancing can yield rates in the 3%-5% range, depending on the type of loan and other factors. For a loan balance of $200,000, the savings by refinancing could be approximately $2,000-$10,000 per year in interest alone. However, refinancing your loans with a private company eliminates the possibility of PSLF. Hence, you should only refinance federal loans once you are sure that you will not be pursuing PSLF. You may refinance your private loans anytime since they do not qualify for PSLF. There are multiple companies that provide student loan refinancing. The process can be done online, sometimes in as little as 30 minutes. There is generally little or no cost to refinancing, and many companies even provide a small cash-back incentive to refinance.
PSLF basics
The PSLF program allows borrowers to have the remainder of their direct loans forgiven after 10 years (120 monthly payments) under a qualifying income-driven repayment (IDR) plan.4 Figure 2 shows an overview of the various IDR plans. During the 120 payments, the borrower must work full time for a qualifying employer, which includes a government employer or a not-for-profit 501(c)(3) organization. Loan forgiveness with PSLF is completely tax free. Importantly, the PSLF program only applies to direct federal loans. You can see your federal loan types and balances by visiting https://studentaid.gov/.
To PSLF or not to PSLF?
With direct federal loans, the decision to refinance or go for PSLF is a major fork in the road. PSLF can be a good option for borrowers with long training programs and with high student loan burdens (e.g., loan-to-income ratios of 1:1, 2:1 or higher). By contrast, borrowers with short training programs or relatively small loan burdens may be better off refinancing to a low interest rate and paying off loans quickly. Virtually all institutions that train residents and fellows are qualified government or 501(c)(3) organizations. Hence, a gastroenterology graduate generally will have completed at least 6 out of 10 years of payments by the end of training. Trainees who did a chief resident year or gastroenterology research track may have completed 7 or 8 years of qualifying payments already.
For trainees who are already planning an academic career, PSLF is often a good option. While PSLF can be a nice benefit, I would not advise making a career decision purely based on PSLF. Private practice jobs generally come with substantially higher salaries than academic and government jobs. This salary differential typically more than compensates for the loss of access to PSLF. Hence, I advise trainees to choose the practice setting that is best for their personal and career satisfaction, and then build a student loan management plan around that. The exception may be the trainee who has a very large student loan burden (e.g., loan-to-income ratio of 2:1 or 3:1).
Caveats with PSLF
There have been well publicized concerns about the future of PSLF, including proposals to eliminate or cap the program.5,6 However, most proposed legislation has only recommended changes to PSLF for new borrowers. If you currently have existing federal loans, you would very likely be grandfathered into the existing PSLF terms. All federal master promissory notes since 2007 have cited PSLF as a loan repayment option.7 Hence, eliminating PSLF for existing borrowers seems unlikely since it would be changing the terms of an executed contract.8
There have also been widespread reports of high numbers of borrowers being denied applications for PSLF.9,10 However, the majority of these applicants did not have correct types of loans, had not worked full time for qualifying employers or had not made the full 120 payments.11 Yet some denials have apparently resulted from errors in tracking qualifying payments by FedLoan servicing.12 Therefore it would be prudent to keep your own careful records of all qualifying payments towards PSLF.
The nuclear option: 20- to 25-year IDR-based forgiveness
An additional option allows borrowers to make IDRs for 20-25 years (details in Figure 2) and then having their remaining loan balance forgiven.13 This option is completely independent of PSLF. Borrowers can work full time or part time and can work for any employer, including private employers.

One additional option: NIH loan repayment programs
One additional solution to consider are the NIH Loan Repayment Programs (LRPs). These programs can provide substantial loan repayment (up to $50,000 annually) for trainees and attendings engaged in research that aligns with NIH priorities, including clinical research or health disparities research.14 Notably, the applicant’s research does not have to be NIH sponsored research.
Getting more information
The approach above is a general overview of student loan concepts for gastroenterologists. However, there are countless nuances and tactics that are beyond the scope of this introductory article. I encourage everyone to get additional information and advice when making your own loan management plan. There are many helpful online resources, podcasts, and books discussing the topic. Several companies provide detailed consultation on managing student loans. Such services may cost a few hundred dollars but could potentially save tens of thousands of dollars on student loan costs.
Dr. Jain is assistant professor of medicine, division of gastroenterology & hepatology, department of medicine, University of North Carolina School of Medicine, Chapel Hill. Dr. Jain has no conflicts of interest and no funding source.
References
1. https://nces.ed.gov/programs/digest/d18/tables/dt18_332.45.asp
2. https://www.credible.com/blog/statistics/average-medical-school-debt/
3. https://studentaid.gov/understand-aid/types/loans/interest-rates
4. https://studentaid.gov/manage-loans/forgiveness-cancellation/public-service
5. https://www.forbes.com/sites/robertfarrington/2019/09/24/how-to-get-your-public-service-loan-forgiveness-qualifying-payments-recounted/#18567f061f5d
6. https://www.cbo.gov/budget-options/2018/54721
7. https://static.studentloans.gov/images/ApplicationAndPromissoryNote.pdf
8. https://www.biglawinvestor.com/pslf-promissory-note/
9. https://bostonstudentloanlawyer.com/scary-stats-for-public-service-loan-forgiveness/
10. https://www.marketwatch.com/story/this-government-loan-forgiveness-program-has-rejected-99-of-borrowers-so-far-2018-09-20
11. https://studentaid.gov/data-center/student/loan-forgiveness/pslf-data
12. https://www.nytimes.com/2019/04/12/your-money/public-service-loan-forgiveness.html
13. https://studentaid.gov/manage-loans/repayment/plans/income-driven
14. https://www.lrp.nih.gov/eligibility-programs
Skin manifestations are emerging in the coronavirus pandemic
Dermatologists there were pulled from their usual duty to help with the pandemic and looked at what was going on with the skin in 148 COVID-19 inpatients. They excluded 60 who had started new drugs within 15 days to rule out acute drug reactions, then reported what they saw (J Eur Acad Dermatol Venereol. 2020 Mar 26. doi: 10.1111/jdv.16387).
Of the 88 COVID-19 patients, 20.5% developed skin manifestations. Eight of the 18 (44%) had skin eruptions at symptom onset, and the rest after hospitalization. Fourteen (78%) had red rashes, three had widespread urticaria, and one had chickenpox-like vesicles. The most commonly affected area was the trunk. Itching was mild or absent, and lesions usually healed up in a few days. Most importantly, skin manifestations did not correlate with disease severity.
These skin manifestations “are similar to cutaneous involvement occurring during common viral infections,” said the author of the report, Sebastiano Recalcati, MD, a dermatologist at Alessandro Manzoni Hospital.
COVID-19 skin manifestations can cloud the diagnosis, according to the authors of another report from Thailand, where the first case of COVID-19 outside of China was reported.
They described a case of a COVID-19 infection in a Bangkok hospital that masqueraded as dengue fever. A person there presented with only a skin rash, petechiae, and a low platelet count, and was diagnosed with Dengue because that’s exactly what it looked like, the authors wrote (J Am Acad Dermatol. 2020 Mar 22. pii: S0190-9622[20]30454-0. doi: 10.1016/j.jaad.2020.03.036).
The correct diagnosis, COVID-19, was made at a tertiary care center after the patient was admitted with respiratory problems.
“There is a possibility that a COVID-19 patient might initially present with a skin rash that can be misdiagnosed as another common disease. ... The practitioner should recognize the possibility that the patient might have only a skin rash” at first, said the lead author of that report, Beuy Joob, PhD, of the Sanitation1 Medical Academic Center, Bangkok, and a coauthor.
There are similar reports in the United States, too. “Many have wondered if COVID-19 presents with any particular skin changes. The answer is yes,” said Randy Jacobs, MD, an assistant clinical professor of dermatology at the University of California, Riverside, who also has a private practice in southern California.
“COVID-19 can feature signs of small blood vessel occlusion. These can be petechiae or tiny bruises, and transient livedoid eruptions,” he said in an interview.
Dr. Jacobs had a 67-year-old patient who presented with a low fever, nasal congestion, postnasal drip, and a wet cough but no shortness of breath. It looked like a common cold. But a week later, the man had a nonpruritic blanching livedoid vascular eruption on his right anterior thigh, and blood in his urine, and he felt weak. The vascular eruption and bloody urine resolved in 24 hours, but the COVID-19 test came back positive and his cough became dry and hacking, and the weakness persisted. He’s in a hospital now and on oxygen, but not ventilated so far.
“Another dermatologist friend of mine also reported a similar transient COVID-19 unilateral livedoid eruption,” Dr. Jacobs said.
It suggests vaso-occlusion. Whether it’s neurogenic, microthrombotic, or immune complex mediated is unknown, but it’s “a skin finding that can help clinicians as they work up their patients with COVID-19 symptoms,” he noted.
Dr. Jacobs and the authors of the studies had no disclosures.
Dermatologists there were pulled from their usual duty to help with the pandemic and looked at what was going on with the skin in 148 COVID-19 inpatients. They excluded 60 who had started new drugs within 15 days to rule out acute drug reactions, then reported what they saw (J Eur Acad Dermatol Venereol. 2020 Mar 26. doi: 10.1111/jdv.16387).
Of the 88 COVID-19 patients, 20.5% developed skin manifestations. Eight of the 18 (44%) had skin eruptions at symptom onset, and the rest after hospitalization. Fourteen (78%) had red rashes, three had widespread urticaria, and one had chickenpox-like vesicles. The most commonly affected area was the trunk. Itching was mild or absent, and lesions usually healed up in a few days. Most importantly, skin manifestations did not correlate with disease severity.
These skin manifestations “are similar to cutaneous involvement occurring during common viral infections,” said the author of the report, Sebastiano Recalcati, MD, a dermatologist at Alessandro Manzoni Hospital.
COVID-19 skin manifestations can cloud the diagnosis, according to the authors of another report from Thailand, where the first case of COVID-19 outside of China was reported.
They described a case of a COVID-19 infection in a Bangkok hospital that masqueraded as dengue fever. A person there presented with only a skin rash, petechiae, and a low platelet count, and was diagnosed with Dengue because that’s exactly what it looked like, the authors wrote (J Am Acad Dermatol. 2020 Mar 22. pii: S0190-9622[20]30454-0. doi: 10.1016/j.jaad.2020.03.036).
The correct diagnosis, COVID-19, was made at a tertiary care center after the patient was admitted with respiratory problems.
“There is a possibility that a COVID-19 patient might initially present with a skin rash that can be misdiagnosed as another common disease. ... The practitioner should recognize the possibility that the patient might have only a skin rash” at first, said the lead author of that report, Beuy Joob, PhD, of the Sanitation1 Medical Academic Center, Bangkok, and a coauthor.
There are similar reports in the United States, too. “Many have wondered if COVID-19 presents with any particular skin changes. The answer is yes,” said Randy Jacobs, MD, an assistant clinical professor of dermatology at the University of California, Riverside, who also has a private practice in southern California.
“COVID-19 can feature signs of small blood vessel occlusion. These can be petechiae or tiny bruises, and transient livedoid eruptions,” he said in an interview.
Dr. Jacobs had a 67-year-old patient who presented with a low fever, nasal congestion, postnasal drip, and a wet cough but no shortness of breath. It looked like a common cold. But a week later, the man had a nonpruritic blanching livedoid vascular eruption on his right anterior thigh, and blood in his urine, and he felt weak. The vascular eruption and bloody urine resolved in 24 hours, but the COVID-19 test came back positive and his cough became dry and hacking, and the weakness persisted. He’s in a hospital now and on oxygen, but not ventilated so far.
“Another dermatologist friend of mine also reported a similar transient COVID-19 unilateral livedoid eruption,” Dr. Jacobs said.
It suggests vaso-occlusion. Whether it’s neurogenic, microthrombotic, or immune complex mediated is unknown, but it’s “a skin finding that can help clinicians as they work up their patients with COVID-19 symptoms,” he noted.
Dr. Jacobs and the authors of the studies had no disclosures.
Dermatologists there were pulled from their usual duty to help with the pandemic and looked at what was going on with the skin in 148 COVID-19 inpatients. They excluded 60 who had started new drugs within 15 days to rule out acute drug reactions, then reported what they saw (J Eur Acad Dermatol Venereol. 2020 Mar 26. doi: 10.1111/jdv.16387).
Of the 88 COVID-19 patients, 20.5% developed skin manifestations. Eight of the 18 (44%) had skin eruptions at symptom onset, and the rest after hospitalization. Fourteen (78%) had red rashes, three had widespread urticaria, and one had chickenpox-like vesicles. The most commonly affected area was the trunk. Itching was mild or absent, and lesions usually healed up in a few days. Most importantly, skin manifestations did not correlate with disease severity.
These skin manifestations “are similar to cutaneous involvement occurring during common viral infections,” said the author of the report, Sebastiano Recalcati, MD, a dermatologist at Alessandro Manzoni Hospital.
COVID-19 skin manifestations can cloud the diagnosis, according to the authors of another report from Thailand, where the first case of COVID-19 outside of China was reported.
They described a case of a COVID-19 infection in a Bangkok hospital that masqueraded as dengue fever. A person there presented with only a skin rash, petechiae, and a low platelet count, and was diagnosed with Dengue because that’s exactly what it looked like, the authors wrote (J Am Acad Dermatol. 2020 Mar 22. pii: S0190-9622[20]30454-0. doi: 10.1016/j.jaad.2020.03.036).
The correct diagnosis, COVID-19, was made at a tertiary care center after the patient was admitted with respiratory problems.
“There is a possibility that a COVID-19 patient might initially present with a skin rash that can be misdiagnosed as another common disease. ... The practitioner should recognize the possibility that the patient might have only a skin rash” at first, said the lead author of that report, Beuy Joob, PhD, of the Sanitation1 Medical Academic Center, Bangkok, and a coauthor.
There are similar reports in the United States, too. “Many have wondered if COVID-19 presents with any particular skin changes. The answer is yes,” said Randy Jacobs, MD, an assistant clinical professor of dermatology at the University of California, Riverside, who also has a private practice in southern California.
“COVID-19 can feature signs of small blood vessel occlusion. These can be petechiae or tiny bruises, and transient livedoid eruptions,” he said in an interview.
Dr. Jacobs had a 67-year-old patient who presented with a low fever, nasal congestion, postnasal drip, and a wet cough but no shortness of breath. It looked like a common cold. But a week later, the man had a nonpruritic blanching livedoid vascular eruption on his right anterior thigh, and blood in his urine, and he felt weak. The vascular eruption and bloody urine resolved in 24 hours, but the COVID-19 test came back positive and his cough became dry and hacking, and the weakness persisted. He’s in a hospital now and on oxygen, but not ventilated so far.
“Another dermatologist friend of mine also reported a similar transient COVID-19 unilateral livedoid eruption,” Dr. Jacobs said.
It suggests vaso-occlusion. Whether it’s neurogenic, microthrombotic, or immune complex mediated is unknown, but it’s “a skin finding that can help clinicians as they work up their patients with COVID-19 symptoms,” he noted.
Dr. Jacobs and the authors of the studies had no disclosures.
What Happens When COVID-19 Breaks Out on a Nuclear Aircraft Carrier?
Updated April 2, 2020.
The commander of a US Navy aircraft carrier in the midst of a COVID-19 outbreak was swiftly fired by Acting Secretary of the Navy Thomas Modly following media coverage of the plight of more than 200 COVID-19 positive sailors on the USS Theodore Roosevelt.
In a statement released April 2, Modly announced the removal of Capt. Brett Crozier for writing a memo that was later leaked to the San Francisco Chronicle newspaper. According to Acting Secretary Modly, the memo was sent “outside the chain of command” and his action “made his Sailors, their families, and many in the public believe that his letter was the only reason help from our larger Navy family was forthcoming, which was hardly the case.”
On Monday, March 30, Capt. Crozier, commanding officer of the nuclear aircraft carrier USS Theodore Roosevelt, sent an urgent request for assistance to senior Navy officials: “[I]n combat we are willing to take certain risks that are not acceptable in peacetime. However, we are not at war, and therefore cannot allow a single Sailor to perish as a result of this pandemic unnecessarily. Decisive action is required now in order to comply with CDC and NAVADMIN 083/20 guidance and prevent tragic outcomes.”
Even as a number of cruise ships with ill and dying passengers were—are—waiting to be allowed to dock in Florida and elsewhere, the USS Theodore Roosevelt was also dealing with a COVID-19 outbreak onboard—and awaiting permission to let the crew of more than 4,000 on shore so they could quarantine safely.
Crozier pointed to “lessons learned” from the Diamond Princess—the only comparable situation at the time. He quoted from the abstract to an epidemiological research study: An index case on board the cruise ship was reported in late January; a month later, 619 of 3,700 passengers and crew had tested positive. Without any interventions, the abstract noted, between January 21st and February 19th an estimated 2,920 of the passengers would have been infected. Isolation and quarantine, it concluded, prevented 2,307 cases. Further, an early evacuation would have been associated with 76 infected persons.
The Diamond Princess, Crozier wrote, was able to more effectively isolate people due to a higher percentage of individual and compartmentalized accommodations. However, due to a warship’s “inherent limitations of space,” his crew could not comply with orders to practice social distancing. “With the exceptions of a handful of senior officer staterooms,” he wrote, “none of the berthing onboard a warship is appropriate for quarantine or isolation.” He also pointed to other obstacles: shared bathrooms, shared sleeping quarters, group mealtimes, and ladders and other surfaces touched and possibly contaminated as crew move around the ship.
Moreover, Crozier wrote, “The spread of the disease is ongoing and accelerating.” By Tuesday March 31st, nearly 1,300 sailors had been tested, and hundreds were testing negative, but 243 sailors had tested positive and 87 more were showing symptoms, according to the latest reports. So far, none are showing serious symptoms.
“If we do not act now, we are failing to take care of our most trusted asset—our sailors,” Capt Crozier wrote. At first, no one seemed to be listening, but after the Chronicle broke the story and it began circulating in the media—things changed. “I heard about the letter from Capt. Crozier [Tuesday] morning,” said Acting Secretary Modly in an interview with the Chronicle. “I know that our command organization has been aware of this for about 24 hours and we have been working actually the last 7 days to move those sailors off the ship and get them into accommodations in Guam. The problem is that Guam doesn’t have enough beds right now and we’re having to talk to the government there to see if we can get some hotel space, create tent-type facilities.”
He noted that the situation for the USS Theodore Roosevelt is “a little bit different and unique” in that it has aircraft and armaments on it, fire hazards, and “we have to run a nuclear power plant.” Crozier had proposed that approximately 10% of the crew remain on board to take care of the duties such as tending to the nuclear reactor.
As of April 1, the Navy plans to remove some 2,700 sailors to the hotel rooms government officials on Guam have secured for them. Secretary Modly made no mention of the care or treatment of infected sailors in his April 2nd statement, but offered this reassurance: "You can offer comfort to your fellow citizens who are struggling and fearful here at home by standing the watch, and working your way through this pandemic with courage and optimism and set the example for the nation. We have an obligation to ensure you have everything you need as fast as we can get it there, and you have my commitment that we will not let you down."
Updated April 2, 2020.
The commander of a US Navy aircraft carrier in the midst of a COVID-19 outbreak was swiftly fired by Acting Secretary of the Navy Thomas Modly following media coverage of the plight of more than 200 COVID-19 positive sailors on the USS Theodore Roosevelt.
In a statement released April 2, Modly announced the removal of Capt. Brett Crozier for writing a memo that was later leaked to the San Francisco Chronicle newspaper. According to Acting Secretary Modly, the memo was sent “outside the chain of command” and his action “made his Sailors, their families, and many in the public believe that his letter was the only reason help from our larger Navy family was forthcoming, which was hardly the case.”
On Monday, March 30, Capt. Crozier, commanding officer of the nuclear aircraft carrier USS Theodore Roosevelt, sent an urgent request for assistance to senior Navy officials: “[I]n combat we are willing to take certain risks that are not acceptable in peacetime. However, we are not at war, and therefore cannot allow a single Sailor to perish as a result of this pandemic unnecessarily. Decisive action is required now in order to comply with CDC and NAVADMIN 083/20 guidance and prevent tragic outcomes.”
Even as a number of cruise ships with ill and dying passengers were—are—waiting to be allowed to dock in Florida and elsewhere, the USS Theodore Roosevelt was also dealing with a COVID-19 outbreak onboard—and awaiting permission to let the crew of more than 4,000 on shore so they could quarantine safely.
Crozier pointed to “lessons learned” from the Diamond Princess—the only comparable situation at the time. He quoted from the abstract to an epidemiological research study: An index case on board the cruise ship was reported in late January; a month later, 619 of 3,700 passengers and crew had tested positive. Without any interventions, the abstract noted, between January 21st and February 19th an estimated 2,920 of the passengers would have been infected. Isolation and quarantine, it concluded, prevented 2,307 cases. Further, an early evacuation would have been associated with 76 infected persons.
The Diamond Princess, Crozier wrote, was able to more effectively isolate people due to a higher percentage of individual and compartmentalized accommodations. However, due to a warship’s “inherent limitations of space,” his crew could not comply with orders to practice social distancing. “With the exceptions of a handful of senior officer staterooms,” he wrote, “none of the berthing onboard a warship is appropriate for quarantine or isolation.” He also pointed to other obstacles: shared bathrooms, shared sleeping quarters, group mealtimes, and ladders and other surfaces touched and possibly contaminated as crew move around the ship.
Moreover, Crozier wrote, “The spread of the disease is ongoing and accelerating.” By Tuesday March 31st, nearly 1,300 sailors had been tested, and hundreds were testing negative, but 243 sailors had tested positive and 87 more were showing symptoms, according to the latest reports. So far, none are showing serious symptoms.
“If we do not act now, we are failing to take care of our most trusted asset—our sailors,” Capt Crozier wrote. At first, no one seemed to be listening, but after the Chronicle broke the story and it began circulating in the media—things changed. “I heard about the letter from Capt. Crozier [Tuesday] morning,” said Acting Secretary Modly in an interview with the Chronicle. “I know that our command organization has been aware of this for about 24 hours and we have been working actually the last 7 days to move those sailors off the ship and get them into accommodations in Guam. The problem is that Guam doesn’t have enough beds right now and we’re having to talk to the government there to see if we can get some hotel space, create tent-type facilities.”
He noted that the situation for the USS Theodore Roosevelt is “a little bit different and unique” in that it has aircraft and armaments on it, fire hazards, and “we have to run a nuclear power plant.” Crozier had proposed that approximately 10% of the crew remain on board to take care of the duties such as tending to the nuclear reactor.
As of April 1, the Navy plans to remove some 2,700 sailors to the hotel rooms government officials on Guam have secured for them. Secretary Modly made no mention of the care or treatment of infected sailors in his April 2nd statement, but offered this reassurance: "You can offer comfort to your fellow citizens who are struggling and fearful here at home by standing the watch, and working your way through this pandemic with courage and optimism and set the example for the nation. We have an obligation to ensure you have everything you need as fast as we can get it there, and you have my commitment that we will not let you down."
Updated April 2, 2020.
The commander of a US Navy aircraft carrier in the midst of a COVID-19 outbreak was swiftly fired by Acting Secretary of the Navy Thomas Modly following media coverage of the plight of more than 200 COVID-19 positive sailors on the USS Theodore Roosevelt.
In a statement released April 2, Modly announced the removal of Capt. Brett Crozier for writing a memo that was later leaked to the San Francisco Chronicle newspaper. According to Acting Secretary Modly, the memo was sent “outside the chain of command” and his action “made his Sailors, their families, and many in the public believe that his letter was the only reason help from our larger Navy family was forthcoming, which was hardly the case.”
On Monday, March 30, Capt. Crozier, commanding officer of the nuclear aircraft carrier USS Theodore Roosevelt, sent an urgent request for assistance to senior Navy officials: “[I]n combat we are willing to take certain risks that are not acceptable in peacetime. However, we are not at war, and therefore cannot allow a single Sailor to perish as a result of this pandemic unnecessarily. Decisive action is required now in order to comply with CDC and NAVADMIN 083/20 guidance and prevent tragic outcomes.”
Even as a number of cruise ships with ill and dying passengers were—are—waiting to be allowed to dock in Florida and elsewhere, the USS Theodore Roosevelt was also dealing with a COVID-19 outbreak onboard—and awaiting permission to let the crew of more than 4,000 on shore so they could quarantine safely.
Crozier pointed to “lessons learned” from the Diamond Princess—the only comparable situation at the time. He quoted from the abstract to an epidemiological research study: An index case on board the cruise ship was reported in late January; a month later, 619 of 3,700 passengers and crew had tested positive. Without any interventions, the abstract noted, between January 21st and February 19th an estimated 2,920 of the passengers would have been infected. Isolation and quarantine, it concluded, prevented 2,307 cases. Further, an early evacuation would have been associated with 76 infected persons.
The Diamond Princess, Crozier wrote, was able to more effectively isolate people due to a higher percentage of individual and compartmentalized accommodations. However, due to a warship’s “inherent limitations of space,” his crew could not comply with orders to practice social distancing. “With the exceptions of a handful of senior officer staterooms,” he wrote, “none of the berthing onboard a warship is appropriate for quarantine or isolation.” He also pointed to other obstacles: shared bathrooms, shared sleeping quarters, group mealtimes, and ladders and other surfaces touched and possibly contaminated as crew move around the ship.
Moreover, Crozier wrote, “The spread of the disease is ongoing and accelerating.” By Tuesday March 31st, nearly 1,300 sailors had been tested, and hundreds were testing negative, but 243 sailors had tested positive and 87 more were showing symptoms, according to the latest reports. So far, none are showing serious symptoms.
“If we do not act now, we are failing to take care of our most trusted asset—our sailors,” Capt Crozier wrote. At first, no one seemed to be listening, but after the Chronicle broke the story and it began circulating in the media—things changed. “I heard about the letter from Capt. Crozier [Tuesday] morning,” said Acting Secretary Modly in an interview with the Chronicle. “I know that our command organization has been aware of this for about 24 hours and we have been working actually the last 7 days to move those sailors off the ship and get them into accommodations in Guam. The problem is that Guam doesn’t have enough beds right now and we’re having to talk to the government there to see if we can get some hotel space, create tent-type facilities.”
He noted that the situation for the USS Theodore Roosevelt is “a little bit different and unique” in that it has aircraft and armaments on it, fire hazards, and “we have to run a nuclear power plant.” Crozier had proposed that approximately 10% of the crew remain on board to take care of the duties such as tending to the nuclear reactor.
As of April 1, the Navy plans to remove some 2,700 sailors to the hotel rooms government officials on Guam have secured for them. Secretary Modly made no mention of the care or treatment of infected sailors in his April 2nd statement, but offered this reassurance: "You can offer comfort to your fellow citizens who are struggling and fearful here at home by standing the watch, and working your way through this pandemic with courage and optimism and set the example for the nation. We have an obligation to ensure you have everything you need as fast as we can get it there, and you have my commitment that we will not let you down."
Failure is not an option
Gene Kranz was the NASA Flight Director during the Gemini and Apollo space flights, including Apollo 11 (moon landing) and Apollo 13. He has written: “When bad things happened, we just calmly laid out all the options and failure was not one of them. We never panicked and we never gave up on finding a solution.”
2019-nCoV (coronavirus, COVID-19) will define this generation of health care providers. First identified in Wuhan, China, in December 2019 and first appearing in the United States on January 19, 2020 (NEJM 2020;382:929). COVID-19 is a coronavirus similar to SARS and MERS. The U.S. Surgeon General and multiple endoscopy societies have recommended (strongly) that elective surgical and endoscopy procedures be deferred. Availability of testing has been slow, but many centers now have developed testing capabilities (including drive-through testing) with 6-hour result turnaround. We do not know the full pathophysiology, R0 (number of secondary cases attributed to an index infection), ease of community transmission, risk to providers, definition of people at high risk (for both acquisition and complications), and much key information n to base recommendations.
Health system and practice leaders do not yet have sufficient information to know which patients to defer, which patients should still be seen, visitor policies, how to segregate waiting rooms, or how to protect providers. Despite a lack of definitive knowledge, we must make critical decisions and know that recommendations can change hourly. As the Chief Clinical Officer at Michigan Medicine, I am spending 16 hours a day immersed in these decisions and find that one of my critical jobs is to keep people from panicking.
Schools, bars, restaurants, churches and other public gathering places are closing. Three countries (to date) have instituted complete quarantine. Digestive Disease Week® has been cancelled. We are entering a time in which normal life is altered and many changes will open up our thinking about long-term alternative processes.
COVID-19 will define this generation. The public will understand the real need for science, policies based on real facts, robust public health systems, and leaders who inspire confidence based on expert guidance. And, I believe we will see gastroenterologists, health systems, hospitals, and practices all showing us what our “finest hours” look like.
John I. Allen, MD, MBA, AGAF
Editor in Chief
Gene Kranz was the NASA Flight Director during the Gemini and Apollo space flights, including Apollo 11 (moon landing) and Apollo 13. He has written: “When bad things happened, we just calmly laid out all the options and failure was not one of them. We never panicked and we never gave up on finding a solution.”
2019-nCoV (coronavirus, COVID-19) will define this generation of health care providers. First identified in Wuhan, China, in December 2019 and first appearing in the United States on January 19, 2020 (NEJM 2020;382:929). COVID-19 is a coronavirus similar to SARS and MERS. The U.S. Surgeon General and multiple endoscopy societies have recommended (strongly) that elective surgical and endoscopy procedures be deferred. Availability of testing has been slow, but many centers now have developed testing capabilities (including drive-through testing) with 6-hour result turnaround. We do not know the full pathophysiology, R0 (number of secondary cases attributed to an index infection), ease of community transmission, risk to providers, definition of people at high risk (for both acquisition and complications), and much key information n to base recommendations.
Health system and practice leaders do not yet have sufficient information to know which patients to defer, which patients should still be seen, visitor policies, how to segregate waiting rooms, or how to protect providers. Despite a lack of definitive knowledge, we must make critical decisions and know that recommendations can change hourly. As the Chief Clinical Officer at Michigan Medicine, I am spending 16 hours a day immersed in these decisions and find that one of my critical jobs is to keep people from panicking.
Schools, bars, restaurants, churches and other public gathering places are closing. Three countries (to date) have instituted complete quarantine. Digestive Disease Week® has been cancelled. We are entering a time in which normal life is altered and many changes will open up our thinking about long-term alternative processes.
COVID-19 will define this generation. The public will understand the real need for science, policies based on real facts, robust public health systems, and leaders who inspire confidence based on expert guidance. And, I believe we will see gastroenterologists, health systems, hospitals, and practices all showing us what our “finest hours” look like.
John I. Allen, MD, MBA, AGAF
Editor in Chief
Gene Kranz was the NASA Flight Director during the Gemini and Apollo space flights, including Apollo 11 (moon landing) and Apollo 13. He has written: “When bad things happened, we just calmly laid out all the options and failure was not one of them. We never panicked and we never gave up on finding a solution.”
2019-nCoV (coronavirus, COVID-19) will define this generation of health care providers. First identified in Wuhan, China, in December 2019 and first appearing in the United States on January 19, 2020 (NEJM 2020;382:929). COVID-19 is a coronavirus similar to SARS and MERS. The U.S. Surgeon General and multiple endoscopy societies have recommended (strongly) that elective surgical and endoscopy procedures be deferred. Availability of testing has been slow, but many centers now have developed testing capabilities (including drive-through testing) with 6-hour result turnaround. We do not know the full pathophysiology, R0 (number of secondary cases attributed to an index infection), ease of community transmission, risk to providers, definition of people at high risk (for both acquisition and complications), and much key information n to base recommendations.
Health system and practice leaders do not yet have sufficient information to know which patients to defer, which patients should still be seen, visitor policies, how to segregate waiting rooms, or how to protect providers. Despite a lack of definitive knowledge, we must make critical decisions and know that recommendations can change hourly. As the Chief Clinical Officer at Michigan Medicine, I am spending 16 hours a day immersed in these decisions and find that one of my critical jobs is to keep people from panicking.
Schools, bars, restaurants, churches and other public gathering places are closing. Three countries (to date) have instituted complete quarantine. Digestive Disease Week® has been cancelled. We are entering a time in which normal life is altered and many changes will open up our thinking about long-term alternative processes.
COVID-19 will define this generation. The public will understand the real need for science, policies based on real facts, robust public health systems, and leaders who inspire confidence based on expert guidance. And, I believe we will see gastroenterologists, health systems, hospitals, and practices all showing us what our “finest hours” look like.
John I. Allen, MD, MBA, AGAF
Editor in Chief
Blood test might detect multiple cancer types, study suggests
Investigators led by Minetta C. Liu, MD, a medical oncologist with the Mayo Clinic, Rochester, Minn., studied 6,689 participants – 2,482 with cancers of more than 50 types and 4,207 without cancer – drawn from the Circulating Cell-free Genome Atlas Study and the STRIVE Study populations.
The investigators performed bisulfite sequencing that targeted informative methylation regions of plasma cell-free DNA (cfDNA), and developed and validated a molecular classifier using methylation patterns to detect cancer and determine its tissue of origin.
Test performance was assessed both for cancer overall and for a prespecified set of 12 cancers (anus, bladder, colon/rectum, esophagus, head and neck, liver/bile duct, lung, lymphoma, ovary, pancreas, plasma cell neoplasm, stomach) that account for about 63% of U.S. cancer deaths annually.
Results reported this week in the Annals of Oncology showed that the test had a specificity of 99.3% in the validation cohort, corresponding to a false-positive rate of just 0.7%.
Sensitivity for detecting stage I-III disease was 43.9% for cancer overall and 67.3% for the prespecified set of cancers accounting for the majority of U.S. cancer deaths.
Test sensitivity increased with stage both for cancer overall (18%, 43%, 81%, and 93% for stage I, II, III, and IV disease, respectively) and for the prespecified set of cancers (39%, 69%, 83%, and 92%, respectively).
The test was able to predict a tissue of origin in 96% of samples in which a cancerlike signal was detected, and in 93% of cases, that prediction was accurate.
Some of the patients who had cancer were symptomatic and therefore would not be considered a screening population, Dr. Liu and coinvestigators acknowledged. Also, the test’s potential for reducing mortality remains unknown, and 1-year follow-up to verify cancer-free status was not yet available for all of the individuals without cancer.
“Together, these data provide compelling evidence that targeted methylation analysis of cfDNA can detect and localize a broad range of nonmetastatic and metastatic cancer types including many common and deadly cancers that lack effective screening strategies,” they maintained. The test’s “specificity and sensitivity performance approach ... the goal for population-level screening.”
“Considering the potential value of early detection in deadly malignancies, further evaluation of this test is justified in prospective population-level studies,” the investigators conclude. “Clinical validation in intended use populations is ongoing ... and a study has been initiated that is returning results to health care providers and patients ....”
Dr. Liu disclosed that the Mayo Clinic was compensated for her advisory board activities for GRAIL Inc. The study was supported by GRAIL, and by Princess Margaret Cancer Centre’s McCain Genitourinary BioBank in the department of surgical oncology.
SOURCE: Liu MC et al. Ann Oncol. 2020 Mar 31. doi: 10.1016/j.annonc.2020.02.011.
Investigators led by Minetta C. Liu, MD, a medical oncologist with the Mayo Clinic, Rochester, Minn., studied 6,689 participants – 2,482 with cancers of more than 50 types and 4,207 without cancer – drawn from the Circulating Cell-free Genome Atlas Study and the STRIVE Study populations.
The investigators performed bisulfite sequencing that targeted informative methylation regions of plasma cell-free DNA (cfDNA), and developed and validated a molecular classifier using methylation patterns to detect cancer and determine its tissue of origin.
Test performance was assessed both for cancer overall and for a prespecified set of 12 cancers (anus, bladder, colon/rectum, esophagus, head and neck, liver/bile duct, lung, lymphoma, ovary, pancreas, plasma cell neoplasm, stomach) that account for about 63% of U.S. cancer deaths annually.
Results reported this week in the Annals of Oncology showed that the test had a specificity of 99.3% in the validation cohort, corresponding to a false-positive rate of just 0.7%.
Sensitivity for detecting stage I-III disease was 43.9% for cancer overall and 67.3% for the prespecified set of cancers accounting for the majority of U.S. cancer deaths.
Test sensitivity increased with stage both for cancer overall (18%, 43%, 81%, and 93% for stage I, II, III, and IV disease, respectively) and for the prespecified set of cancers (39%, 69%, 83%, and 92%, respectively).
The test was able to predict a tissue of origin in 96% of samples in which a cancerlike signal was detected, and in 93% of cases, that prediction was accurate.
Some of the patients who had cancer were symptomatic and therefore would not be considered a screening population, Dr. Liu and coinvestigators acknowledged. Also, the test’s potential for reducing mortality remains unknown, and 1-year follow-up to verify cancer-free status was not yet available for all of the individuals without cancer.
“Together, these data provide compelling evidence that targeted methylation analysis of cfDNA can detect and localize a broad range of nonmetastatic and metastatic cancer types including many common and deadly cancers that lack effective screening strategies,” they maintained. The test’s “specificity and sensitivity performance approach ... the goal for population-level screening.”
“Considering the potential value of early detection in deadly malignancies, further evaluation of this test is justified in prospective population-level studies,” the investigators conclude. “Clinical validation in intended use populations is ongoing ... and a study has been initiated that is returning results to health care providers and patients ....”
Dr. Liu disclosed that the Mayo Clinic was compensated for her advisory board activities for GRAIL Inc. The study was supported by GRAIL, and by Princess Margaret Cancer Centre’s McCain Genitourinary BioBank in the department of surgical oncology.
SOURCE: Liu MC et al. Ann Oncol. 2020 Mar 31. doi: 10.1016/j.annonc.2020.02.011.
Investigators led by Minetta C. Liu, MD, a medical oncologist with the Mayo Clinic, Rochester, Minn., studied 6,689 participants – 2,482 with cancers of more than 50 types and 4,207 without cancer – drawn from the Circulating Cell-free Genome Atlas Study and the STRIVE Study populations.
The investigators performed bisulfite sequencing that targeted informative methylation regions of plasma cell-free DNA (cfDNA), and developed and validated a molecular classifier using methylation patterns to detect cancer and determine its tissue of origin.
Test performance was assessed both for cancer overall and for a prespecified set of 12 cancers (anus, bladder, colon/rectum, esophagus, head and neck, liver/bile duct, lung, lymphoma, ovary, pancreas, plasma cell neoplasm, stomach) that account for about 63% of U.S. cancer deaths annually.
Results reported this week in the Annals of Oncology showed that the test had a specificity of 99.3% in the validation cohort, corresponding to a false-positive rate of just 0.7%.
Sensitivity for detecting stage I-III disease was 43.9% for cancer overall and 67.3% for the prespecified set of cancers accounting for the majority of U.S. cancer deaths.
Test sensitivity increased with stage both for cancer overall (18%, 43%, 81%, and 93% for stage I, II, III, and IV disease, respectively) and for the prespecified set of cancers (39%, 69%, 83%, and 92%, respectively).
The test was able to predict a tissue of origin in 96% of samples in which a cancerlike signal was detected, and in 93% of cases, that prediction was accurate.
Some of the patients who had cancer were symptomatic and therefore would not be considered a screening population, Dr. Liu and coinvestigators acknowledged. Also, the test’s potential for reducing mortality remains unknown, and 1-year follow-up to verify cancer-free status was not yet available for all of the individuals without cancer.
“Together, these data provide compelling evidence that targeted methylation analysis of cfDNA can detect and localize a broad range of nonmetastatic and metastatic cancer types including many common and deadly cancers that lack effective screening strategies,” they maintained. The test’s “specificity and sensitivity performance approach ... the goal for population-level screening.”
“Considering the potential value of early detection in deadly malignancies, further evaluation of this test is justified in prospective population-level studies,” the investigators conclude. “Clinical validation in intended use populations is ongoing ... and a study has been initiated that is returning results to health care providers and patients ....”
Dr. Liu disclosed that the Mayo Clinic was compensated for her advisory board activities for GRAIL Inc. The study was supported by GRAIL, and by Princess Margaret Cancer Centre’s McCain Genitourinary BioBank in the department of surgical oncology.
SOURCE: Liu MC et al. Ann Oncol. 2020 Mar 31. doi: 10.1016/j.annonc.2020.02.011.
FROM ANNALS OF ONCOLOGY
New guidance on management of acute CVD during COVID-19
The Chinese Society of Cardiology (CSC) has issued a consensus statement on the management of cardiac emergencies during the COVID-19 pandemic.
The document first appeared in the Chinese Journal of Cardiology, and a translated version was published in Circulation. The consensus statement was developed by 125 medical experts in the fields of cardiovascular disease and infectious disease. This included 23 experts currently working in Wuhan, China.
Three overarching principles guided their recommendations.
- The highest priority is prevention and control of transmission (including protecting staff).
- Patients should be assessed both for COVID-19 and for cardiovascular issues.
- At all times, all interventions and therapies provided should be in concordance with directives of infection control authorities.
“Considering that some asymptomatic patients may be a source of infection and transmission, all patients with severe emergent cardiovascular diseases should be managed as suspected cases of COVID-19 in Hubei Province,” noted writing chair and cardiologist Yaling Han, MD, of the General Hospital of Northern Theater Command in Shenyang, China.
In areas outside Hubei Province, where COVID-19 was less prevalent, this “infected until proven otherwise” approach was also recommended, although not as strictly.
Diagnosing CVD and COVID-19 simultaneously
In patients with emergent cardiovascular needs in whom COVID-19 has not been ruled out, quarantine in a single-bed room is needed, they wrote. The patient should be monitored for clinical manifestations of the disease, and undergo COVID-19 nucleic acid testing as soon as possible.
After infection control is considered, including limiting risk for infection to health care workers, risk assessment that weighs the relative advantages and disadvantages of treating the cardiovascular disease while preventing transmission can be considered, the investigators wrote.
At all times, transfers to different areas of the hospital and between hospitals should be minimized to reduce the risk for infection transmission.
The authors also recommended the use of “select laboratory tests with definitive sensitivity and specificity for disease diagnosis or assessment.”
For patients with acute aortic syndrome or acute pulmonary embolism, this means CT angiography. When acute pulmonary embolism is suspected, D-dimer testing and deep vein ultrasound can be employed, and for patients with acute coronary syndrome, ordinary electrocardiography and standard biomarkers for cardiac injury are preferred.
In addition, “all patients should undergo lung CT examination to evaluate for imaging features typical of COVID-19. ... Chest x-ray is not recommended because of a high rate of false negative diagnosis,” the authors wrote.
Intervene with caution
Medical therapy should be optimized in patients with emergent cardiovascular issues, with invasive strategies for diagnosis and therapy used “with caution,” according to the Chinese experts.
Conditions for which conservative medical treatment is recommended during COVID-19 pandemic include ST-segment elevation MI (STEMI) where thrombolytic therapy is indicated, STEMI when the optimal window for revascularization has passed, high-risk non-STEMI (NSTEMI), patients with uncomplicated Stanford type B aortic dissection, acute pulmonary embolism, acute exacerbation of heart failure, and hypertensive emergency.
“Vigilance should be paid to avoid misdiagnosing patients with pulmonary infarction as COVID-19 pneumonia,” they noted.
Diagnoses warranting invasive intervention are limited to STEMI with hemodynamic instability, life-threatening NSTEMI, Stanford type A or complex type B acute aortic dissection, bradyarrhythmia complicated by syncope or unstable hemodynamics mandating implantation of a device, and pulmonary embolism with hemodynamic instability for whom intravenous thrombolytics are too risky.
Interventions should be done in a cath lab or operating room with negative-pressure ventilation, with strict periprocedural disinfection. Personal protective equipment should also be of the strictest level.
In patients for whom COVID-19 cannot be ruled out presenting in a region with low incidence of COVID-19, interventions should only be considered for more severe cases and undertaken in a cath lab, electrophysiology lab, or operating room “with more than standard disinfection procedures that fulfill regulatory mandates for infection control.”
If negative-pressure ventilation is not available, air conditioning (for example, laminar flow and ventilation) should be stopped.
Establish plans now
“We operationalized all of these strategies at Beth Israel Deaconess Medical Center several weeks ago, since Boston had that early outbreak with the Biogen conference, but I suspect many institutions nationally are still formulating plans,” said Dhruv Kazi, MD, MSc, in an interview.
Although COVID-19 is “primarily a single-organ disease – it destroys the lungs” – transmission of infection to cardiology providers was an early problem that needed to be addressed, said Dr. Kazi. “We now know that a cardiologist seeing a patient who reports shortness of breath and then leans in to carefully auscultate the lungs and heart can get exposed if not provided adequate personal protective equipment; hence the cancellation of elective procedures, conversion of most elective visits to telemedicine, if possible, and the use of surgical/N95 masks in clinic and on rounds.”
Regarding the CSC recommendation to consider medical over invasive management, Dr. Kazi noteed that this works better in a setting where rapid testing is available. “Where that is not the case – as in the U.S. – resorting to conservative therapy for all COVID suspect cases will result in suboptimal care, particularly when nine out of every 10 COVID suspects will eventually rule out.”
One of his biggest worries now is that patients simply won’t come. Afraid of being exposed to COVID-19, patients with MIs and strokes may avoid or delay coming to the hospital.
“There is some evidence that this occurred in Wuhan, and I’m starting to see anecdotal evidence of this in Boston,” said Dr. Kazi. “We need to remind our patients that, if they experience symptoms of a heart attack or stroke, they deserve the same lifesaving treatment we offered before this pandemic set in. They should not try and sit it out.”
A version of this article originally appeared on Medscape.com.
The Chinese Society of Cardiology (CSC) has issued a consensus statement on the management of cardiac emergencies during the COVID-19 pandemic.
The document first appeared in the Chinese Journal of Cardiology, and a translated version was published in Circulation. The consensus statement was developed by 125 medical experts in the fields of cardiovascular disease and infectious disease. This included 23 experts currently working in Wuhan, China.
Three overarching principles guided their recommendations.
- The highest priority is prevention and control of transmission (including protecting staff).
- Patients should be assessed both for COVID-19 and for cardiovascular issues.
- At all times, all interventions and therapies provided should be in concordance with directives of infection control authorities.
“Considering that some asymptomatic patients may be a source of infection and transmission, all patients with severe emergent cardiovascular diseases should be managed as suspected cases of COVID-19 in Hubei Province,” noted writing chair and cardiologist Yaling Han, MD, of the General Hospital of Northern Theater Command in Shenyang, China.
In areas outside Hubei Province, where COVID-19 was less prevalent, this “infected until proven otherwise” approach was also recommended, although not as strictly.
Diagnosing CVD and COVID-19 simultaneously
In patients with emergent cardiovascular needs in whom COVID-19 has not been ruled out, quarantine in a single-bed room is needed, they wrote. The patient should be monitored for clinical manifestations of the disease, and undergo COVID-19 nucleic acid testing as soon as possible.
After infection control is considered, including limiting risk for infection to health care workers, risk assessment that weighs the relative advantages and disadvantages of treating the cardiovascular disease while preventing transmission can be considered, the investigators wrote.
At all times, transfers to different areas of the hospital and between hospitals should be minimized to reduce the risk for infection transmission.
The authors also recommended the use of “select laboratory tests with definitive sensitivity and specificity for disease diagnosis or assessment.”
For patients with acute aortic syndrome or acute pulmonary embolism, this means CT angiography. When acute pulmonary embolism is suspected, D-dimer testing and deep vein ultrasound can be employed, and for patients with acute coronary syndrome, ordinary electrocardiography and standard biomarkers for cardiac injury are preferred.
In addition, “all patients should undergo lung CT examination to evaluate for imaging features typical of COVID-19. ... Chest x-ray is not recommended because of a high rate of false negative diagnosis,” the authors wrote.
Intervene with caution
Medical therapy should be optimized in patients with emergent cardiovascular issues, with invasive strategies for diagnosis and therapy used “with caution,” according to the Chinese experts.
Conditions for which conservative medical treatment is recommended during COVID-19 pandemic include ST-segment elevation MI (STEMI) where thrombolytic therapy is indicated, STEMI when the optimal window for revascularization has passed, high-risk non-STEMI (NSTEMI), patients with uncomplicated Stanford type B aortic dissection, acute pulmonary embolism, acute exacerbation of heart failure, and hypertensive emergency.
“Vigilance should be paid to avoid misdiagnosing patients with pulmonary infarction as COVID-19 pneumonia,” they noted.
Diagnoses warranting invasive intervention are limited to STEMI with hemodynamic instability, life-threatening NSTEMI, Stanford type A or complex type B acute aortic dissection, bradyarrhythmia complicated by syncope or unstable hemodynamics mandating implantation of a device, and pulmonary embolism with hemodynamic instability for whom intravenous thrombolytics are too risky.
Interventions should be done in a cath lab or operating room with negative-pressure ventilation, with strict periprocedural disinfection. Personal protective equipment should also be of the strictest level.
In patients for whom COVID-19 cannot be ruled out presenting in a region with low incidence of COVID-19, interventions should only be considered for more severe cases and undertaken in a cath lab, electrophysiology lab, or operating room “with more than standard disinfection procedures that fulfill regulatory mandates for infection control.”
If negative-pressure ventilation is not available, air conditioning (for example, laminar flow and ventilation) should be stopped.
Establish plans now
“We operationalized all of these strategies at Beth Israel Deaconess Medical Center several weeks ago, since Boston had that early outbreak with the Biogen conference, but I suspect many institutions nationally are still formulating plans,” said Dhruv Kazi, MD, MSc, in an interview.
Although COVID-19 is “primarily a single-organ disease – it destroys the lungs” – transmission of infection to cardiology providers was an early problem that needed to be addressed, said Dr. Kazi. “We now know that a cardiologist seeing a patient who reports shortness of breath and then leans in to carefully auscultate the lungs and heart can get exposed if not provided adequate personal protective equipment; hence the cancellation of elective procedures, conversion of most elective visits to telemedicine, if possible, and the use of surgical/N95 masks in clinic and on rounds.”
Regarding the CSC recommendation to consider medical over invasive management, Dr. Kazi noteed that this works better in a setting where rapid testing is available. “Where that is not the case – as in the U.S. – resorting to conservative therapy for all COVID suspect cases will result in suboptimal care, particularly when nine out of every 10 COVID suspects will eventually rule out.”
One of his biggest worries now is that patients simply won’t come. Afraid of being exposed to COVID-19, patients with MIs and strokes may avoid or delay coming to the hospital.
“There is some evidence that this occurred in Wuhan, and I’m starting to see anecdotal evidence of this in Boston,” said Dr. Kazi. “We need to remind our patients that, if they experience symptoms of a heart attack or stroke, they deserve the same lifesaving treatment we offered before this pandemic set in. They should not try and sit it out.”
A version of this article originally appeared on Medscape.com.
The Chinese Society of Cardiology (CSC) has issued a consensus statement on the management of cardiac emergencies during the COVID-19 pandemic.
The document first appeared in the Chinese Journal of Cardiology, and a translated version was published in Circulation. The consensus statement was developed by 125 medical experts in the fields of cardiovascular disease and infectious disease. This included 23 experts currently working in Wuhan, China.
Three overarching principles guided their recommendations.
- The highest priority is prevention and control of transmission (including protecting staff).
- Patients should be assessed both for COVID-19 and for cardiovascular issues.
- At all times, all interventions and therapies provided should be in concordance with directives of infection control authorities.
“Considering that some asymptomatic patients may be a source of infection and transmission, all patients with severe emergent cardiovascular diseases should be managed as suspected cases of COVID-19 in Hubei Province,” noted writing chair and cardiologist Yaling Han, MD, of the General Hospital of Northern Theater Command in Shenyang, China.
In areas outside Hubei Province, where COVID-19 was less prevalent, this “infected until proven otherwise” approach was also recommended, although not as strictly.
Diagnosing CVD and COVID-19 simultaneously
In patients with emergent cardiovascular needs in whom COVID-19 has not been ruled out, quarantine in a single-bed room is needed, they wrote. The patient should be monitored for clinical manifestations of the disease, and undergo COVID-19 nucleic acid testing as soon as possible.
After infection control is considered, including limiting risk for infection to health care workers, risk assessment that weighs the relative advantages and disadvantages of treating the cardiovascular disease while preventing transmission can be considered, the investigators wrote.
At all times, transfers to different areas of the hospital and between hospitals should be minimized to reduce the risk for infection transmission.
The authors also recommended the use of “select laboratory tests with definitive sensitivity and specificity for disease diagnosis or assessment.”
For patients with acute aortic syndrome or acute pulmonary embolism, this means CT angiography. When acute pulmonary embolism is suspected, D-dimer testing and deep vein ultrasound can be employed, and for patients with acute coronary syndrome, ordinary electrocardiography and standard biomarkers for cardiac injury are preferred.
In addition, “all patients should undergo lung CT examination to evaluate for imaging features typical of COVID-19. ... Chest x-ray is not recommended because of a high rate of false negative diagnosis,” the authors wrote.
Intervene with caution
Medical therapy should be optimized in patients with emergent cardiovascular issues, with invasive strategies for diagnosis and therapy used “with caution,” according to the Chinese experts.
Conditions for which conservative medical treatment is recommended during COVID-19 pandemic include ST-segment elevation MI (STEMI) where thrombolytic therapy is indicated, STEMI when the optimal window for revascularization has passed, high-risk non-STEMI (NSTEMI), patients with uncomplicated Stanford type B aortic dissection, acute pulmonary embolism, acute exacerbation of heart failure, and hypertensive emergency.
“Vigilance should be paid to avoid misdiagnosing patients with pulmonary infarction as COVID-19 pneumonia,” they noted.
Diagnoses warranting invasive intervention are limited to STEMI with hemodynamic instability, life-threatening NSTEMI, Stanford type A or complex type B acute aortic dissection, bradyarrhythmia complicated by syncope or unstable hemodynamics mandating implantation of a device, and pulmonary embolism with hemodynamic instability for whom intravenous thrombolytics are too risky.
Interventions should be done in a cath lab or operating room with negative-pressure ventilation, with strict periprocedural disinfection. Personal protective equipment should also be of the strictest level.
In patients for whom COVID-19 cannot be ruled out presenting in a region with low incidence of COVID-19, interventions should only be considered for more severe cases and undertaken in a cath lab, electrophysiology lab, or operating room “with more than standard disinfection procedures that fulfill regulatory mandates for infection control.”
If negative-pressure ventilation is not available, air conditioning (for example, laminar flow and ventilation) should be stopped.
Establish plans now
“We operationalized all of these strategies at Beth Israel Deaconess Medical Center several weeks ago, since Boston had that early outbreak with the Biogen conference, but I suspect many institutions nationally are still formulating plans,” said Dhruv Kazi, MD, MSc, in an interview.
Although COVID-19 is “primarily a single-organ disease – it destroys the lungs” – transmission of infection to cardiology providers was an early problem that needed to be addressed, said Dr. Kazi. “We now know that a cardiologist seeing a patient who reports shortness of breath and then leans in to carefully auscultate the lungs and heart can get exposed if not provided adequate personal protective equipment; hence the cancellation of elective procedures, conversion of most elective visits to telemedicine, if possible, and the use of surgical/N95 masks in clinic and on rounds.”
Regarding the CSC recommendation to consider medical over invasive management, Dr. Kazi noteed that this works better in a setting where rapid testing is available. “Where that is not the case – as in the U.S. – resorting to conservative therapy for all COVID suspect cases will result in suboptimal care, particularly when nine out of every 10 COVID suspects will eventually rule out.”
One of his biggest worries now is that patients simply won’t come. Afraid of being exposed to COVID-19, patients with MIs and strokes may avoid or delay coming to the hospital.
“There is some evidence that this occurred in Wuhan, and I’m starting to see anecdotal evidence of this in Boston,” said Dr. Kazi. “We need to remind our patients that, if they experience symptoms of a heart attack or stroke, they deserve the same lifesaving treatment we offered before this pandemic set in. They should not try and sit it out.”
A version of this article originally appeared on Medscape.com.
Stage I mycosis fungoides is the general dermatologist’s bailiwick
LAHAINA, HAWAII – without bringing in a medical oncologist, Trilokraj Tejasvi, MBBS, said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
This approach is in the best interest of patients with stage I mycosis fungoides, the skin-limited, patch/plaque form of the disease that generally responds well to skin-directed therapies without needing to resort to the medical oncologist’s arsenal of toxic treatments.
“For many medical oncologists, a lymphoma is a lymphoma. The first thing they give is CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone), and all the variants of CHOP,” cautioned Dr. Tejasvi, a dermatologist who is director of the cutaneous lymphoma program at the University of Michigan, Ann Arbor, and chief of the dermatology service at the Ann Arbor Veteran Affairs Hospital.
Stage IA mycosis fungoides is defined under the TNMB (tumor, node, metastasis, blood) classification as patches and/or plaques covering less than 10% of body surface area along with negative nodes, no metastases, and no or low burden of disease in the blood. Stage IB differs only in that it features 10% or greater body surface area involvement. The extent of body surface area involvement can be estimated by hands-on measurement in which the area of one of the patient’s hands – palm plus fingers – is considered equivalent to 1% of that individual’s total body surface area.
The first question patients newly diagnosed with a cutaneous T-cell lymphoma ask concerns their prognosis. For those with stage IA or IB mycosis fungoides, the news is very good, as highlighted in a retrospective study of nearly 1,400 patients with mycosis fungoides, 71% of whom presented with patch/plaque stage disease (J Clin Oncol. 2010 Nov 1;28[31]:4730-9).
The median overall survival was 35.5 years in patients with stage IA disease and 21.5 years in those with stage IB disease.
“I tell patients with stage IA disease that whether we treat it or not will not change the course of their life,” Dr. Tejasvri said.
His message to patients with stage IB disease is that, because of their 38% risk of disease progression, he wants to see them in follow-up annually for the rest of their life.
Stage IIA disease – that is, patches and/or plaques with lymph node involvement with no effacement – is a tipping point at which serious consideration should be given to possible referral to a specialized multidisciplinary lymphoma center, in his view. That’s because the 10-year overall survival rate is only 52%.
Topical therapies
Topical corticosteroids remain the time-honored first-line skin-directed treatment. The mechanism of benefit involves induction of apoptosis and inhibition of lymphocyte binding. In one prospective study, clobetasol propionate achieved a 94% overall response rate in patients with stage IA or B disease, with minimal toxicity.
Alternatives include topical 5% imiquimod (Aldara), with an overall response rate of 80% and complete response rate of 45% in a 20-patient study. A newer formulation of mechlorethamine gel (Valchlor), is reported to have a 59% overall response rate and a sustained response in 86% of initial responders. For refractory skin lesions, 1% bexarotene gel (Targretin) is an option, with overall response rates of 44%-63% reported in prospective trials.
“I like it if the patient’s insurance covers it. Otherwise, it’s like buying a Prius: it’s $30,000 for a 45-g tube, which is insane,” Dr. Tejasvi commented.
Narrow-band UVB phototherapy is an effective modality for thin plaques and patches, as is PUVA for thicker ones. Dr. Tejasvi typically treats with topical steroids and/or phototherapy for at least 3 months before tapering.
When to suspect mycosis fungoides
“Mycosis fungoides is a great masquerader,” the dermatologist observed. For that reason, it deserves to be included in the differential diagnosis of an atypical psoriasiform or eczematoid rash, any new-onset rash in an elderly patient, or a rash with fever, night sweats, and unintended weight loss in a patient of any age. Generalized erythema with severe itching is another red flag.
“This pruritus is so severe that the only other condition which in my clinical practice would match it is Norwegian scabies,” according to Dr. Tejasvi.
Polychromatic patches or plaques in skin of color warrant further investigation as possible mycosis fungoides, he added.
Dr. Tejasvi reported having no financial conflicts of interest regarding his presentation.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – without bringing in a medical oncologist, Trilokraj Tejasvi, MBBS, said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
This approach is in the best interest of patients with stage I mycosis fungoides, the skin-limited, patch/plaque form of the disease that generally responds well to skin-directed therapies without needing to resort to the medical oncologist’s arsenal of toxic treatments.
“For many medical oncologists, a lymphoma is a lymphoma. The first thing they give is CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone), and all the variants of CHOP,” cautioned Dr. Tejasvi, a dermatologist who is director of the cutaneous lymphoma program at the University of Michigan, Ann Arbor, and chief of the dermatology service at the Ann Arbor Veteran Affairs Hospital.
Stage IA mycosis fungoides is defined under the TNMB (tumor, node, metastasis, blood) classification as patches and/or plaques covering less than 10% of body surface area along with negative nodes, no metastases, and no or low burden of disease in the blood. Stage IB differs only in that it features 10% or greater body surface area involvement. The extent of body surface area involvement can be estimated by hands-on measurement in which the area of one of the patient’s hands – palm plus fingers – is considered equivalent to 1% of that individual’s total body surface area.
The first question patients newly diagnosed with a cutaneous T-cell lymphoma ask concerns their prognosis. For those with stage IA or IB mycosis fungoides, the news is very good, as highlighted in a retrospective study of nearly 1,400 patients with mycosis fungoides, 71% of whom presented with patch/plaque stage disease (J Clin Oncol. 2010 Nov 1;28[31]:4730-9).
The median overall survival was 35.5 years in patients with stage IA disease and 21.5 years in those with stage IB disease.
“I tell patients with stage IA disease that whether we treat it or not will not change the course of their life,” Dr. Tejasvri said.
His message to patients with stage IB disease is that, because of their 38% risk of disease progression, he wants to see them in follow-up annually for the rest of their life.
Stage IIA disease – that is, patches and/or plaques with lymph node involvement with no effacement – is a tipping point at which serious consideration should be given to possible referral to a specialized multidisciplinary lymphoma center, in his view. That’s because the 10-year overall survival rate is only 52%.
Topical therapies
Topical corticosteroids remain the time-honored first-line skin-directed treatment. The mechanism of benefit involves induction of apoptosis and inhibition of lymphocyte binding. In one prospective study, clobetasol propionate achieved a 94% overall response rate in patients with stage IA or B disease, with minimal toxicity.
Alternatives include topical 5% imiquimod (Aldara), with an overall response rate of 80% and complete response rate of 45% in a 20-patient study. A newer formulation of mechlorethamine gel (Valchlor), is reported to have a 59% overall response rate and a sustained response in 86% of initial responders. For refractory skin lesions, 1% bexarotene gel (Targretin) is an option, with overall response rates of 44%-63% reported in prospective trials.
“I like it if the patient’s insurance covers it. Otherwise, it’s like buying a Prius: it’s $30,000 for a 45-g tube, which is insane,” Dr. Tejasvi commented.
Narrow-band UVB phototherapy is an effective modality for thin plaques and patches, as is PUVA for thicker ones. Dr. Tejasvi typically treats with topical steroids and/or phototherapy for at least 3 months before tapering.
When to suspect mycosis fungoides
“Mycosis fungoides is a great masquerader,” the dermatologist observed. For that reason, it deserves to be included in the differential diagnosis of an atypical psoriasiform or eczematoid rash, any new-onset rash in an elderly patient, or a rash with fever, night sweats, and unintended weight loss in a patient of any age. Generalized erythema with severe itching is another red flag.
“This pruritus is so severe that the only other condition which in my clinical practice would match it is Norwegian scabies,” according to Dr. Tejasvi.
Polychromatic patches or plaques in skin of color warrant further investigation as possible mycosis fungoides, he added.
Dr. Tejasvi reported having no financial conflicts of interest regarding his presentation.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – without bringing in a medical oncologist, Trilokraj Tejasvi, MBBS, said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
This approach is in the best interest of patients with stage I mycosis fungoides, the skin-limited, patch/plaque form of the disease that generally responds well to skin-directed therapies without needing to resort to the medical oncologist’s arsenal of toxic treatments.
“For many medical oncologists, a lymphoma is a lymphoma. The first thing they give is CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone), and all the variants of CHOP,” cautioned Dr. Tejasvi, a dermatologist who is director of the cutaneous lymphoma program at the University of Michigan, Ann Arbor, and chief of the dermatology service at the Ann Arbor Veteran Affairs Hospital.
Stage IA mycosis fungoides is defined under the TNMB (tumor, node, metastasis, blood) classification as patches and/or plaques covering less than 10% of body surface area along with negative nodes, no metastases, and no or low burden of disease in the blood. Stage IB differs only in that it features 10% or greater body surface area involvement. The extent of body surface area involvement can be estimated by hands-on measurement in which the area of one of the patient’s hands – palm plus fingers – is considered equivalent to 1% of that individual’s total body surface area.
The first question patients newly diagnosed with a cutaneous T-cell lymphoma ask concerns their prognosis. For those with stage IA or IB mycosis fungoides, the news is very good, as highlighted in a retrospective study of nearly 1,400 patients with mycosis fungoides, 71% of whom presented with patch/plaque stage disease (J Clin Oncol. 2010 Nov 1;28[31]:4730-9).
The median overall survival was 35.5 years in patients with stage IA disease and 21.5 years in those with stage IB disease.
“I tell patients with stage IA disease that whether we treat it or not will not change the course of their life,” Dr. Tejasvri said.
His message to patients with stage IB disease is that, because of their 38% risk of disease progression, he wants to see them in follow-up annually for the rest of their life.
Stage IIA disease – that is, patches and/or plaques with lymph node involvement with no effacement – is a tipping point at which serious consideration should be given to possible referral to a specialized multidisciplinary lymphoma center, in his view. That’s because the 10-year overall survival rate is only 52%.
Topical therapies
Topical corticosteroids remain the time-honored first-line skin-directed treatment. The mechanism of benefit involves induction of apoptosis and inhibition of lymphocyte binding. In one prospective study, clobetasol propionate achieved a 94% overall response rate in patients with stage IA or B disease, with minimal toxicity.
Alternatives include topical 5% imiquimod (Aldara), with an overall response rate of 80% and complete response rate of 45% in a 20-patient study. A newer formulation of mechlorethamine gel (Valchlor), is reported to have a 59% overall response rate and a sustained response in 86% of initial responders. For refractory skin lesions, 1% bexarotene gel (Targretin) is an option, with overall response rates of 44%-63% reported in prospective trials.
“I like it if the patient’s insurance covers it. Otherwise, it’s like buying a Prius: it’s $30,000 for a 45-g tube, which is insane,” Dr. Tejasvi commented.
Narrow-band UVB phototherapy is an effective modality for thin plaques and patches, as is PUVA for thicker ones. Dr. Tejasvi typically treats with topical steroids and/or phototherapy for at least 3 months before tapering.
When to suspect mycosis fungoides
“Mycosis fungoides is a great masquerader,” the dermatologist observed. For that reason, it deserves to be included in the differential diagnosis of an atypical psoriasiform or eczematoid rash, any new-onset rash in an elderly patient, or a rash with fever, night sweats, and unintended weight loss in a patient of any age. Generalized erythema with severe itching is another red flag.
“This pruritus is so severe that the only other condition which in my clinical practice would match it is Norwegian scabies,” according to Dr. Tejasvi.
Polychromatic patches or plaques in skin of color warrant further investigation as possible mycosis fungoides, he added.
Dr. Tejasvi reported having no financial conflicts of interest regarding his presentation.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
REPORTING FROM SDEF HAWAII DERMATOLOGY SEMINAR
FMT appears safe and effective for IBD patients with recurrent C. difficile
Fecal microbiota transplantation (FMT) appears safe and effective for treating recurrent Clostridioides difficile infection in patients with inflammatory bowel disease (IBD), according to an ongoing prospective trial.
Most patients were cured of C. difficile after one fecal transplant, reported Jessica Allegretti, MD, associate director of the Crohn’s and Colitis Center at Brigham and Women’s Hospital in Boston.
“[For patients without IBD], fecal microbiota transplantation has been shown to be very effective for the treatment of recurrent C. diff,” Dr. Allegretti said at the annual Gut Microbiota for Health World Summit.
But similar data for patients with IBD are scarce, and this knowledge gap has high clinical relevance, Dr. Allegretti said. She noted that C. difficile infections are eight times more common among patients with IBD, and risk of recurrence is increased 4.5-fold.
According to Dr. Allegretti, three small clinical trials have tested FMT for treating recurrent C. difficile infections in patients with IBD.
“[These studies were] somewhat prospective, but [data] mainly retrospectively collected, as they relied heavily on chart review for the assessment of IBD disease activity,” she said at the meeting sponsored by the American Gastroenterological Association and the European Society for Neurogastroenterology and Motility..
Across the trials, C. difficile infection cure rates were comparable with non-IBD cohorts; but disease flare rates ranged from 17.9% to 54%, which raised concern that FMT may trigger inflammation.
To investigate further, Dr. Allegretti and her colleagues designed a prospective trial that is set to enroll 50 patients with IBD. Among 37 patients treated to date, a slight majority were women (56.8%), about one-third had Crohn’s disease (37.8%), and two-thirds had ulcerative colitis (62.2%). The average baseline calprotectin level, which measures inflammation in the intestines, was 1,804.8 microg/g of feces, which is far above the upper limit of 50 microg/g.
“This is a very inflamed patient population,” Dr. Allegretti said.
Out of these 37 patients, 34 (92%) were cured of C. difficile infection after only one fecal transplant, and the remaining three patients were cured after a second FMT.
“They all did very well,” Dr. Allegretti said.
Concerning IBD clinical scores, all patients with Crohn’s disease either had unchanged or improved disease. Among those with ulcerative colitis, almost all had unchanged or improved disease, except for one patient who had a de novo flare.
Early microbiome analyses showed patients had increased alpha diversity and richness after FMT that was sustained through week 12. Because only three patients had recurrence, numbers were too small to generate predictive data based on relative abundance.
Dr. Allegretti continued her presentation with a review of FMT for IBD in general.
“For Crohn’s disease, the role [of microbiome manipulation] seems a bit more clear,” Dr. Allegretti said, considering multiple effective treatments that alter gut flora, such as antibiotics.
In contrast, the role for microbiome manipulation in treating ulcerative colitis “has remained a bit unclear,” she said. Although some probiotics appear effective for treating mild disease, other microbiome-altering treatments, such as diversion of fecal stream, antibiotics, and bowel rest, have fallen short.
Still, pooled data from four randomized clinical trials showed that FMT led to remission in 28% of patients with ulcerative colitis, compared with 9% who receive placebo.
“You may be thinking that seems a bit underwhelming compared to the 90% or so cure rate we get for C. diff trials,” Dr. Allegretti said. “However, if you look at our other biologic trials in IBD, 28% puts FMT on par with our other IBD therapies.”
According to Dr. Allegretti, at least three stool-based, FMT-like therapeutics are poised to become commercially available in the next few years for the treatment of C. difficile infection, including broad- and narrow-spectrum enema bags and oral capsules.
“I certainly think we will start to see off-label usage in our IBD patients, and we will start to have an easier and more systemic way of utilizing these microbiome-based therapies,” Dr. Allegretti said. “They will be coming to market, and when they do, whether or not we are allowed to still do traditional FMT in its current form remains unseen. The FDA may not allow us to do that in the future when we have an FDA-approved product.”Dr. Allegretti disclosed relationships with Merck, Openbiome, Finch Therapeutics, and others.
Fecal microbiota transplantation (FMT) appears safe and effective for treating recurrent Clostridioides difficile infection in patients with inflammatory bowel disease (IBD), according to an ongoing prospective trial.
Most patients were cured of C. difficile after one fecal transplant, reported Jessica Allegretti, MD, associate director of the Crohn’s and Colitis Center at Brigham and Women’s Hospital in Boston.
“[For patients without IBD], fecal microbiota transplantation has been shown to be very effective for the treatment of recurrent C. diff,” Dr. Allegretti said at the annual Gut Microbiota for Health World Summit.
But similar data for patients with IBD are scarce, and this knowledge gap has high clinical relevance, Dr. Allegretti said. She noted that C. difficile infections are eight times more common among patients with IBD, and risk of recurrence is increased 4.5-fold.
According to Dr. Allegretti, three small clinical trials have tested FMT for treating recurrent C. difficile infections in patients with IBD.
“[These studies were] somewhat prospective, but [data] mainly retrospectively collected, as they relied heavily on chart review for the assessment of IBD disease activity,” she said at the meeting sponsored by the American Gastroenterological Association and the European Society for Neurogastroenterology and Motility..
Across the trials, C. difficile infection cure rates were comparable with non-IBD cohorts; but disease flare rates ranged from 17.9% to 54%, which raised concern that FMT may trigger inflammation.
To investigate further, Dr. Allegretti and her colleagues designed a prospective trial that is set to enroll 50 patients with IBD. Among 37 patients treated to date, a slight majority were women (56.8%), about one-third had Crohn’s disease (37.8%), and two-thirds had ulcerative colitis (62.2%). The average baseline calprotectin level, which measures inflammation in the intestines, was 1,804.8 microg/g of feces, which is far above the upper limit of 50 microg/g.
“This is a very inflamed patient population,” Dr. Allegretti said.
Out of these 37 patients, 34 (92%) were cured of C. difficile infection after only one fecal transplant, and the remaining three patients were cured after a second FMT.
“They all did very well,” Dr. Allegretti said.
Concerning IBD clinical scores, all patients with Crohn’s disease either had unchanged or improved disease. Among those with ulcerative colitis, almost all had unchanged or improved disease, except for one patient who had a de novo flare.
Early microbiome analyses showed patients had increased alpha diversity and richness after FMT that was sustained through week 12. Because only three patients had recurrence, numbers were too small to generate predictive data based on relative abundance.
Dr. Allegretti continued her presentation with a review of FMT for IBD in general.
“For Crohn’s disease, the role [of microbiome manipulation] seems a bit more clear,” Dr. Allegretti said, considering multiple effective treatments that alter gut flora, such as antibiotics.
In contrast, the role for microbiome manipulation in treating ulcerative colitis “has remained a bit unclear,” she said. Although some probiotics appear effective for treating mild disease, other microbiome-altering treatments, such as diversion of fecal stream, antibiotics, and bowel rest, have fallen short.
Still, pooled data from four randomized clinical trials showed that FMT led to remission in 28% of patients with ulcerative colitis, compared with 9% who receive placebo.
“You may be thinking that seems a bit underwhelming compared to the 90% or so cure rate we get for C. diff trials,” Dr. Allegretti said. “However, if you look at our other biologic trials in IBD, 28% puts FMT on par with our other IBD therapies.”
According to Dr. Allegretti, at least three stool-based, FMT-like therapeutics are poised to become commercially available in the next few years for the treatment of C. difficile infection, including broad- and narrow-spectrum enema bags and oral capsules.
“I certainly think we will start to see off-label usage in our IBD patients, and we will start to have an easier and more systemic way of utilizing these microbiome-based therapies,” Dr. Allegretti said. “They will be coming to market, and when they do, whether or not we are allowed to still do traditional FMT in its current form remains unseen. The FDA may not allow us to do that in the future when we have an FDA-approved product.”Dr. Allegretti disclosed relationships with Merck, Openbiome, Finch Therapeutics, and others.
Fecal microbiota transplantation (FMT) appears safe and effective for treating recurrent Clostridioides difficile infection in patients with inflammatory bowel disease (IBD), according to an ongoing prospective trial.
Most patients were cured of C. difficile after one fecal transplant, reported Jessica Allegretti, MD, associate director of the Crohn’s and Colitis Center at Brigham and Women’s Hospital in Boston.
“[For patients without IBD], fecal microbiota transplantation has been shown to be very effective for the treatment of recurrent C. diff,” Dr. Allegretti said at the annual Gut Microbiota for Health World Summit.
But similar data for patients with IBD are scarce, and this knowledge gap has high clinical relevance, Dr. Allegretti said. She noted that C. difficile infections are eight times more common among patients with IBD, and risk of recurrence is increased 4.5-fold.
According to Dr. Allegretti, three small clinical trials have tested FMT for treating recurrent C. difficile infections in patients with IBD.
“[These studies were] somewhat prospective, but [data] mainly retrospectively collected, as they relied heavily on chart review for the assessment of IBD disease activity,” she said at the meeting sponsored by the American Gastroenterological Association and the European Society for Neurogastroenterology and Motility..
Across the trials, C. difficile infection cure rates were comparable with non-IBD cohorts; but disease flare rates ranged from 17.9% to 54%, which raised concern that FMT may trigger inflammation.
To investigate further, Dr. Allegretti and her colleagues designed a prospective trial that is set to enroll 50 patients with IBD. Among 37 patients treated to date, a slight majority were women (56.8%), about one-third had Crohn’s disease (37.8%), and two-thirds had ulcerative colitis (62.2%). The average baseline calprotectin level, which measures inflammation in the intestines, was 1,804.8 microg/g of feces, which is far above the upper limit of 50 microg/g.
“This is a very inflamed patient population,” Dr. Allegretti said.
Out of these 37 patients, 34 (92%) were cured of C. difficile infection after only one fecal transplant, and the remaining three patients were cured after a second FMT.
“They all did very well,” Dr. Allegretti said.
Concerning IBD clinical scores, all patients with Crohn’s disease either had unchanged or improved disease. Among those with ulcerative colitis, almost all had unchanged or improved disease, except for one patient who had a de novo flare.
Early microbiome analyses showed patients had increased alpha diversity and richness after FMT that was sustained through week 12. Because only three patients had recurrence, numbers were too small to generate predictive data based on relative abundance.
Dr. Allegretti continued her presentation with a review of FMT for IBD in general.
“For Crohn’s disease, the role [of microbiome manipulation] seems a bit more clear,” Dr. Allegretti said, considering multiple effective treatments that alter gut flora, such as antibiotics.
In contrast, the role for microbiome manipulation in treating ulcerative colitis “has remained a bit unclear,” she said. Although some probiotics appear effective for treating mild disease, other microbiome-altering treatments, such as diversion of fecal stream, antibiotics, and bowel rest, have fallen short.
Still, pooled data from four randomized clinical trials showed that FMT led to remission in 28% of patients with ulcerative colitis, compared with 9% who receive placebo.
“You may be thinking that seems a bit underwhelming compared to the 90% or so cure rate we get for C. diff trials,” Dr. Allegretti said. “However, if you look at our other biologic trials in IBD, 28% puts FMT on par with our other IBD therapies.”
According to Dr. Allegretti, at least three stool-based, FMT-like therapeutics are poised to become commercially available in the next few years for the treatment of C. difficile infection, including broad- and narrow-spectrum enema bags and oral capsules.
“I certainly think we will start to see off-label usage in our IBD patients, and we will start to have an easier and more systemic way of utilizing these microbiome-based therapies,” Dr. Allegretti said. “They will be coming to market, and when they do, whether or not we are allowed to still do traditional FMT in its current form remains unseen. The FDA may not allow us to do that in the future when we have an FDA-approved product.”Dr. Allegretti disclosed relationships with Merck, Openbiome, Finch Therapeutics, and others.
FROM GMFH 2020
‘Larger-than-life’ physician Stephen Schwartz dies of COVID-19 at 78
Stephen M. Schwartz, MD, PhD, a pioneer in the field of vascular biology and a longtime professor of pathology at the University of Washington, Seattle, died March 17, 2020, after being hospitalized with COVID-19. He was 78.
“This has become all too real,” UW president Ana Mari Cauce said on Facebook, where she described Dr. Schwartz as “larger than life,” and superimposed a photo of him in front of Mount Rainier, according to a report in the Seattle Times.
Dr. Schwartz is “rightfully considered a giant among investigators of the biology of smooth muscle cells and the structure of blood vessels,” Paul Ramsey, MD, CEO of UW Medicine, said in a statement. He will be remembered for his “vigorous advocacy for research and for the field of vascular biology as well as for his many trainees who have gone on to great success as independent investigators in the field of vascular pathobiology,” Dr. Ramsey said.
Dr. Schwartz received a BA in biology from Harvard University in 1963 and an MD from Boston University in 1967. Dr. Schwartz started a residency in the UW department of pathology in 1967 and received his PhD from the institution in 1973. From 1974 to 1979, he was an assistant professor of pathology and became a full professor in 1984.
Dr. Schwartz was also an adjunct professor in the UW departments of bioengineering and medicine, “reflective of his many collaborative relationships with faculty in other departments in our medical school and in the world,” Dr. Ramsey said.
“Dr. Schwartz left a lasting imprint on the UW School of Medicine and the broader scientific community. He will be greatly missed,” he added.
‘A great loss’
Dr. Schwartz chaired numerous national and international meetings in the field of vascular biology. He was the founding chair of the Gordon Research Conference on Vascular Biology and a cofounder and second president of the North American Vascular Biology Organization (NAVBO). He created NAVBO’s flagship summer course, Vasculata.
“The NAVBO community has suffered a great loss,” Bernadette Englert, executive officer for the organization, said in a statement. He will be “sorely missed by generations of vascular biologists and pathologists.”
News of Dr. Schwartz’s passing lit up Twitter. Here are just a few comments:
UW lost to COVID-19 “beloved professor Stephen Schwartz, a pioneer in vascular biology and a larger-than-life scientist. Steve, you will be missed!” Rong Tian, MD, PhD, with the bioengineering department, wrote in a tweet.
“Stephen Schwartz was a giant in vascular biology and a mentor to countless faculty and trainees, including myself. He will be deeply missed,” said Kelly Stevens, PhD, also from the bioengineering department.
A version of this article originally appeared on Medscape.com.
Stephen M. Schwartz, MD, PhD, a pioneer in the field of vascular biology and a longtime professor of pathology at the University of Washington, Seattle, died March 17, 2020, after being hospitalized with COVID-19. He was 78.
“This has become all too real,” UW president Ana Mari Cauce said on Facebook, where she described Dr. Schwartz as “larger than life,” and superimposed a photo of him in front of Mount Rainier, according to a report in the Seattle Times.
Dr. Schwartz is “rightfully considered a giant among investigators of the biology of smooth muscle cells and the structure of blood vessels,” Paul Ramsey, MD, CEO of UW Medicine, said in a statement. He will be remembered for his “vigorous advocacy for research and for the field of vascular biology as well as for his many trainees who have gone on to great success as independent investigators in the field of vascular pathobiology,” Dr. Ramsey said.
Dr. Schwartz received a BA in biology from Harvard University in 1963 and an MD from Boston University in 1967. Dr. Schwartz started a residency in the UW department of pathology in 1967 and received his PhD from the institution in 1973. From 1974 to 1979, he was an assistant professor of pathology and became a full professor in 1984.
Dr. Schwartz was also an adjunct professor in the UW departments of bioengineering and medicine, “reflective of his many collaborative relationships with faculty in other departments in our medical school and in the world,” Dr. Ramsey said.
“Dr. Schwartz left a lasting imprint on the UW School of Medicine and the broader scientific community. He will be greatly missed,” he added.
‘A great loss’
Dr. Schwartz chaired numerous national and international meetings in the field of vascular biology. He was the founding chair of the Gordon Research Conference on Vascular Biology and a cofounder and second president of the North American Vascular Biology Organization (NAVBO). He created NAVBO’s flagship summer course, Vasculata.
“The NAVBO community has suffered a great loss,” Bernadette Englert, executive officer for the organization, said in a statement. He will be “sorely missed by generations of vascular biologists and pathologists.”
News of Dr. Schwartz’s passing lit up Twitter. Here are just a few comments:
UW lost to COVID-19 “beloved professor Stephen Schwartz, a pioneer in vascular biology and a larger-than-life scientist. Steve, you will be missed!” Rong Tian, MD, PhD, with the bioengineering department, wrote in a tweet.
“Stephen Schwartz was a giant in vascular biology and a mentor to countless faculty and trainees, including myself. He will be deeply missed,” said Kelly Stevens, PhD, also from the bioengineering department.
A version of this article originally appeared on Medscape.com.
Stephen M. Schwartz, MD, PhD, a pioneer in the field of vascular biology and a longtime professor of pathology at the University of Washington, Seattle, died March 17, 2020, after being hospitalized with COVID-19. He was 78.
“This has become all too real,” UW president Ana Mari Cauce said on Facebook, where she described Dr. Schwartz as “larger than life,” and superimposed a photo of him in front of Mount Rainier, according to a report in the Seattle Times.
Dr. Schwartz is “rightfully considered a giant among investigators of the biology of smooth muscle cells and the structure of blood vessels,” Paul Ramsey, MD, CEO of UW Medicine, said in a statement. He will be remembered for his “vigorous advocacy for research and for the field of vascular biology as well as for his many trainees who have gone on to great success as independent investigators in the field of vascular pathobiology,” Dr. Ramsey said.
Dr. Schwartz received a BA in biology from Harvard University in 1963 and an MD from Boston University in 1967. Dr. Schwartz started a residency in the UW department of pathology in 1967 and received his PhD from the institution in 1973. From 1974 to 1979, he was an assistant professor of pathology and became a full professor in 1984.
Dr. Schwartz was also an adjunct professor in the UW departments of bioengineering and medicine, “reflective of his many collaborative relationships with faculty in other departments in our medical school and in the world,” Dr. Ramsey said.
“Dr. Schwartz left a lasting imprint on the UW School of Medicine and the broader scientific community. He will be greatly missed,” he added.
‘A great loss’
Dr. Schwartz chaired numerous national and international meetings in the field of vascular biology. He was the founding chair of the Gordon Research Conference on Vascular Biology and a cofounder and second president of the North American Vascular Biology Organization (NAVBO). He created NAVBO’s flagship summer course, Vasculata.
“The NAVBO community has suffered a great loss,” Bernadette Englert, executive officer for the organization, said in a statement. He will be “sorely missed by generations of vascular biologists and pathologists.”
News of Dr. Schwartz’s passing lit up Twitter. Here are just a few comments:
UW lost to COVID-19 “beloved professor Stephen Schwartz, a pioneer in vascular biology and a larger-than-life scientist. Steve, you will be missed!” Rong Tian, MD, PhD, with the bioengineering department, wrote in a tweet.
“Stephen Schwartz was a giant in vascular biology and a mentor to countless faculty and trainees, including myself. He will be deeply missed,” said Kelly Stevens, PhD, also from the bioengineering department.
A version of this article originally appeared on Medscape.com.
Get out the inpatient vote
Disenfranchisement undeniably remains a major problem across the United States. While it is challenging for health care providers to find time to vote, hospitalized patients are an underrecognized vulnerable group, often unable to exercise this constitutional right. With the 2020 election approaching, voting is as important as ever.
On morning rounds after the 2018 election, we discussed the impact of a changing majority in the House of Representatives and its potential impact on health care in America. We discussed where, when, and how we voted, and then suddenly considered a question that we were unable to answer: How do our hospitalized patients vote and did any of them vote in this important election?
Inpatients rarely know when or how long they will be hospitalized. They often have no chance to prepare by paying bills, arranging care for loved ones, or finding coverage for employment responsibilities. The sickest patients can do little more than wonder about anything other than their short-term health. As a result of restricted voting laws, they, like too many others, are effectively disenfranchised.
We asked administrators in multiple hospitals across New York City how to help our patients vote. Unfortunately, the process is overwhelmingly complex and varies by state. Absentee ballots, which are easily accessible in New York if it they are requested no later than 7 days before the election, are harder to come by on the same day. Most people struggle to vote in general – with only 61% voting in the 2016 election.1 To combat this, individual hospitals have created initiatives such as Penn Votes, which has helped 65 hospitalized Pennsylvania residents vote in the last three elections2 – a success, but still leaving so many without a voice.
With health care being a major policy issue for the 2020 election, voting has never been more important for patients. With nearly 1 million hospital beds in America,3 hospitalized patients represent a significant number of potential voters who are functionally disenfranchised. Most importantly, these patients are directly under our care, and we are their strongest advocates. Therefore, we ask our fellow health care providers to start planning today how we will help our patients exercise their voices, participate in our health care policy debate, and choose the future leaders of our country.
Dr. Rosenblatt is assistant professor of medicine, Division of Gastroenterology and Hepatology, at Weill Cornell Medicine, New York. Dr. Verna is assistant professor of medicine, Department of Surgery, at Columbia University Irving Medical School, New York. Dr. Rosenblatt and Dr. Verna reported having no relevant conflicts of interest.
References
1. File T. Voting in America: A Look at the 2016 Presidential Election [Internet]. 2017 [cited 2020 Jan 7];Available from: https://www.census.gov/newsroom/blogs/random-samplings/2017/05/voting_in_america.html.
2. Vigodner S. Penn students are helping hospitalized patients cast emergency ballots for Tuesday’s election [Internet]. Dly. Pennsylvanian. 2018;Available from: https://www.thedp.com/article/2018/11/penn-med-votes-emergency-hospital-patients-upenn-philadelphia-elections.
3. Association AH. Fast facts on US hospitals [Internet]. 2019 [cited 2020 Jan 7];Available from: https://www.aha.org/statistics/fast-facts-us-hospitals.
Disenfranchisement undeniably remains a major problem across the United States. While it is challenging for health care providers to find time to vote, hospitalized patients are an underrecognized vulnerable group, often unable to exercise this constitutional right. With the 2020 election approaching, voting is as important as ever.
On morning rounds after the 2018 election, we discussed the impact of a changing majority in the House of Representatives and its potential impact on health care in America. We discussed where, when, and how we voted, and then suddenly considered a question that we were unable to answer: How do our hospitalized patients vote and did any of them vote in this important election?
Inpatients rarely know when or how long they will be hospitalized. They often have no chance to prepare by paying bills, arranging care for loved ones, or finding coverage for employment responsibilities. The sickest patients can do little more than wonder about anything other than their short-term health. As a result of restricted voting laws, they, like too many others, are effectively disenfranchised.
We asked administrators in multiple hospitals across New York City how to help our patients vote. Unfortunately, the process is overwhelmingly complex and varies by state. Absentee ballots, which are easily accessible in New York if it they are requested no later than 7 days before the election, are harder to come by on the same day. Most people struggle to vote in general – with only 61% voting in the 2016 election.1 To combat this, individual hospitals have created initiatives such as Penn Votes, which has helped 65 hospitalized Pennsylvania residents vote in the last three elections2 – a success, but still leaving so many without a voice.
With health care being a major policy issue for the 2020 election, voting has never been more important for patients. With nearly 1 million hospital beds in America,3 hospitalized patients represent a significant number of potential voters who are functionally disenfranchised. Most importantly, these patients are directly under our care, and we are their strongest advocates. Therefore, we ask our fellow health care providers to start planning today how we will help our patients exercise their voices, participate in our health care policy debate, and choose the future leaders of our country.
Dr. Rosenblatt is assistant professor of medicine, Division of Gastroenterology and Hepatology, at Weill Cornell Medicine, New York. Dr. Verna is assistant professor of medicine, Department of Surgery, at Columbia University Irving Medical School, New York. Dr. Rosenblatt and Dr. Verna reported having no relevant conflicts of interest.
References
1. File T. Voting in America: A Look at the 2016 Presidential Election [Internet]. 2017 [cited 2020 Jan 7];Available from: https://www.census.gov/newsroom/blogs/random-samplings/2017/05/voting_in_america.html.
2. Vigodner S. Penn students are helping hospitalized patients cast emergency ballots for Tuesday’s election [Internet]. Dly. Pennsylvanian. 2018;Available from: https://www.thedp.com/article/2018/11/penn-med-votes-emergency-hospital-patients-upenn-philadelphia-elections.
3. Association AH. Fast facts on US hospitals [Internet]. 2019 [cited 2020 Jan 7];Available from: https://www.aha.org/statistics/fast-facts-us-hospitals.
Disenfranchisement undeniably remains a major problem across the United States. While it is challenging for health care providers to find time to vote, hospitalized patients are an underrecognized vulnerable group, often unable to exercise this constitutional right. With the 2020 election approaching, voting is as important as ever.
On morning rounds after the 2018 election, we discussed the impact of a changing majority in the House of Representatives and its potential impact on health care in America. We discussed where, when, and how we voted, and then suddenly considered a question that we were unable to answer: How do our hospitalized patients vote and did any of them vote in this important election?
Inpatients rarely know when or how long they will be hospitalized. They often have no chance to prepare by paying bills, arranging care for loved ones, or finding coverage for employment responsibilities. The sickest patients can do little more than wonder about anything other than their short-term health. As a result of restricted voting laws, they, like too many others, are effectively disenfranchised.
We asked administrators in multiple hospitals across New York City how to help our patients vote. Unfortunately, the process is overwhelmingly complex and varies by state. Absentee ballots, which are easily accessible in New York if it they are requested no later than 7 days before the election, are harder to come by on the same day. Most people struggle to vote in general – with only 61% voting in the 2016 election.1 To combat this, individual hospitals have created initiatives such as Penn Votes, which has helped 65 hospitalized Pennsylvania residents vote in the last three elections2 – a success, but still leaving so many without a voice.
With health care being a major policy issue for the 2020 election, voting has never been more important for patients. With nearly 1 million hospital beds in America,3 hospitalized patients represent a significant number of potential voters who are functionally disenfranchised. Most importantly, these patients are directly under our care, and we are their strongest advocates. Therefore, we ask our fellow health care providers to start planning today how we will help our patients exercise their voices, participate in our health care policy debate, and choose the future leaders of our country.
Dr. Rosenblatt is assistant professor of medicine, Division of Gastroenterology and Hepatology, at Weill Cornell Medicine, New York. Dr. Verna is assistant professor of medicine, Department of Surgery, at Columbia University Irving Medical School, New York. Dr. Rosenblatt and Dr. Verna reported having no relevant conflicts of interest.
References
1. File T. Voting in America: A Look at the 2016 Presidential Election [Internet]. 2017 [cited 2020 Jan 7];Available from: https://www.census.gov/newsroom/blogs/random-samplings/2017/05/voting_in_america.html.
2. Vigodner S. Penn students are helping hospitalized patients cast emergency ballots for Tuesday’s election [Internet]. Dly. Pennsylvanian. 2018;Available from: https://www.thedp.com/article/2018/11/penn-med-votes-emergency-hospital-patients-upenn-philadelphia-elections.
3. Association AH. Fast facts on US hospitals [Internet]. 2019 [cited 2020 Jan 7];Available from: https://www.aha.org/statistics/fast-facts-us-hospitals.