User login
For MD-IQ use only
Oncologists need to advocate for scarce COVID-19 resources: ASCO
both at the institutional and regional level, according to new recommendations from the American Society for Clinical Oncology (ASCO).
“There was a lot of concern from the oncology community that if a patient had cancer, they would be arbitrarily excluded from consideration for critical care resources,” said Jonathan M. Marron, MD, chair-elect of ASCO’s Ethics Committee and lead author of the recommendations.
“The hope is that we’ll never have to make any of these decisions ... but the primary reason for putting together these recommendations was that if such decisions have to be made, we hope to inform them,” he told Medscape Medical News.
Marron, who is a pediatric hematologist at Boston Children’s Hospital, says ASCO’s main recommendation is that decisions about the allocation of resources must be separated from bedside clinical care, meaning that clinicians who are caring for individual patients should not also be the ones making the allocation decisions.
“Those dueling responsibilities are a conflict of interest and make that physician unable to make an unbiased decision,” he said.
“It’s also just an unbearable burden to try and do those two things simultaneously,” he added. “It’s an incredible burden to do them individually, but it’s multifold worse to try to do them both simultaneously.”
He said the vital role of oncologists who provide treatment is to offer the kind of personalized information that triage committees need in order to make appropriate decisions.
“They should be asked – maybe even must be asked – to provide the most high-quality evidence-based data about their patients’ diagnosis and prognosis,” Marron commented. “Because oncology is evolving so rapidly, and cancer is so many different diseases, it’s impossible for someone making these decisions to know everything they would need to know about why this patient is likely to survive their cancer and this patient is not.”
He says that during the COVID-19 pandemic, concerns regarding public health transcend the well-being of individual patients and that consideration must be given to providing the maximum benefit to the greatest number of people.
“That makes perfect sense and is the appropriate and laudable goal during a public health emergency like this ... but one of the challenges is that there is this belief that a diagnosis of cancer is uniformly fatal,” Marron said.
“It’s certainly conceivable that it would be a better use of resources to give the last ventilator to a young, otherwise healthy patient rather than a patient with multiply recurrent progressive metastatic cancer,” he continued. “However, we want to ensure that there is at least a discussion where that information is made available, rather than just saying, ‘She’s got cancer. She’s a lost cause.’ ”
Cancer patients are doing very well
Concerns about cancer misconceptions have been circulating in the oncology community since the start of the pandemic. “It’s really important that people understand that cancer patients are doing very well nowadays, and even with a diagnosis of cancer, they can potentially live for many years,” Anne Chiang, MD, PhD, from the Smilow Cancer Network, New Haven, Connecticut, told Medscape Medical News in a recent interview.
Thus far, even in hard-hit New York City, fears that cancer patients may not be receiving appropriate care have not materialized, according to Mark Robson, MD, a medical oncologist at Memorial Sloan Kettering Cancer Center (MSKCC). “I would emphasize that cancer patients are ABSOLUTELY getting the care they need, including patients with metastatic disease,” he recently tweeted. “NOONE at @sloan_kettering (or anywhere else) is being ‘triaged’ because of advanced cancer. Period.”
Robson told Medscape Medical News that although MSKCC continues to provide oncology care to patients with cancer, “we are [also] treating them if they develop COVID. ... I am trying to help pivot the institution towards care in this setting.”
He said he agrees with Craig Spencer, MD, MPH, director of global health in emergency medicine at the New York–Presbyterian/Columbia University Medical Center, who recently tweeted, “If you need a ventilator, you get a ventilator. Let’s be clear – this isn’t being ‘rationed.’ ”
Marron emphasized that an important safeguard against uninformed decision making is appropriate planning. For hospitalized patients, this means oncologists who provide treatment should offer information even before it is requested. But he said the “duty to plan” begins long before that.
“Clinicians haven’t always been great at talking about death and long-term outcomes with their patients, but this really cranks up the importance of having those conversations, and having them early, even though it’s incredibly hard. If someone has expressed that they would never want to be put on a ventilator, it’s important now even more so that is made clear,” he said.
He said early responses to the ASCO statement suggest that it has calmed some concerns in the oncology community, “but it still remains to be seen whether individual institutions will take this to heart, because this unto itself cannot enforce anything – it is up to individual institutions. I am hopeful this will get to the people it needs to get to.”
This article first appeared on Medscape.com.
both at the institutional and regional level, according to new recommendations from the American Society for Clinical Oncology (ASCO).
“There was a lot of concern from the oncology community that if a patient had cancer, they would be arbitrarily excluded from consideration for critical care resources,” said Jonathan M. Marron, MD, chair-elect of ASCO’s Ethics Committee and lead author of the recommendations.
“The hope is that we’ll never have to make any of these decisions ... but the primary reason for putting together these recommendations was that if such decisions have to be made, we hope to inform them,” he told Medscape Medical News.
Marron, who is a pediatric hematologist at Boston Children’s Hospital, says ASCO’s main recommendation is that decisions about the allocation of resources must be separated from bedside clinical care, meaning that clinicians who are caring for individual patients should not also be the ones making the allocation decisions.
“Those dueling responsibilities are a conflict of interest and make that physician unable to make an unbiased decision,” he said.
“It’s also just an unbearable burden to try and do those two things simultaneously,” he added. “It’s an incredible burden to do them individually, but it’s multifold worse to try to do them both simultaneously.”
He said the vital role of oncologists who provide treatment is to offer the kind of personalized information that triage committees need in order to make appropriate decisions.
“They should be asked – maybe even must be asked – to provide the most high-quality evidence-based data about their patients’ diagnosis and prognosis,” Marron commented. “Because oncology is evolving so rapidly, and cancer is so many different diseases, it’s impossible for someone making these decisions to know everything they would need to know about why this patient is likely to survive their cancer and this patient is not.”
He says that during the COVID-19 pandemic, concerns regarding public health transcend the well-being of individual patients and that consideration must be given to providing the maximum benefit to the greatest number of people.
“That makes perfect sense and is the appropriate and laudable goal during a public health emergency like this ... but one of the challenges is that there is this belief that a diagnosis of cancer is uniformly fatal,” Marron said.
“It’s certainly conceivable that it would be a better use of resources to give the last ventilator to a young, otherwise healthy patient rather than a patient with multiply recurrent progressive metastatic cancer,” he continued. “However, we want to ensure that there is at least a discussion where that information is made available, rather than just saying, ‘She’s got cancer. She’s a lost cause.’ ”
Cancer patients are doing very well
Concerns about cancer misconceptions have been circulating in the oncology community since the start of the pandemic. “It’s really important that people understand that cancer patients are doing very well nowadays, and even with a diagnosis of cancer, they can potentially live for many years,” Anne Chiang, MD, PhD, from the Smilow Cancer Network, New Haven, Connecticut, told Medscape Medical News in a recent interview.
Thus far, even in hard-hit New York City, fears that cancer patients may not be receiving appropriate care have not materialized, according to Mark Robson, MD, a medical oncologist at Memorial Sloan Kettering Cancer Center (MSKCC). “I would emphasize that cancer patients are ABSOLUTELY getting the care they need, including patients with metastatic disease,” he recently tweeted. “NOONE at @sloan_kettering (or anywhere else) is being ‘triaged’ because of advanced cancer. Period.”
Robson told Medscape Medical News that although MSKCC continues to provide oncology care to patients with cancer, “we are [also] treating them if they develop COVID. ... I am trying to help pivot the institution towards care in this setting.”
He said he agrees with Craig Spencer, MD, MPH, director of global health in emergency medicine at the New York–Presbyterian/Columbia University Medical Center, who recently tweeted, “If you need a ventilator, you get a ventilator. Let’s be clear – this isn’t being ‘rationed.’ ”
Marron emphasized that an important safeguard against uninformed decision making is appropriate planning. For hospitalized patients, this means oncologists who provide treatment should offer information even before it is requested. But he said the “duty to plan” begins long before that.
“Clinicians haven’t always been great at talking about death and long-term outcomes with their patients, but this really cranks up the importance of having those conversations, and having them early, even though it’s incredibly hard. If someone has expressed that they would never want to be put on a ventilator, it’s important now even more so that is made clear,” he said.
He said early responses to the ASCO statement suggest that it has calmed some concerns in the oncology community, “but it still remains to be seen whether individual institutions will take this to heart, because this unto itself cannot enforce anything – it is up to individual institutions. I am hopeful this will get to the people it needs to get to.”
This article first appeared on Medscape.com.
both at the institutional and regional level, according to new recommendations from the American Society for Clinical Oncology (ASCO).
“There was a lot of concern from the oncology community that if a patient had cancer, they would be arbitrarily excluded from consideration for critical care resources,” said Jonathan M. Marron, MD, chair-elect of ASCO’s Ethics Committee and lead author of the recommendations.
“The hope is that we’ll never have to make any of these decisions ... but the primary reason for putting together these recommendations was that if such decisions have to be made, we hope to inform them,” he told Medscape Medical News.
Marron, who is a pediatric hematologist at Boston Children’s Hospital, says ASCO’s main recommendation is that decisions about the allocation of resources must be separated from bedside clinical care, meaning that clinicians who are caring for individual patients should not also be the ones making the allocation decisions.
“Those dueling responsibilities are a conflict of interest and make that physician unable to make an unbiased decision,” he said.
“It’s also just an unbearable burden to try and do those two things simultaneously,” he added. “It’s an incredible burden to do them individually, but it’s multifold worse to try to do them both simultaneously.”
He said the vital role of oncologists who provide treatment is to offer the kind of personalized information that triage committees need in order to make appropriate decisions.
“They should be asked – maybe even must be asked – to provide the most high-quality evidence-based data about their patients’ diagnosis and prognosis,” Marron commented. “Because oncology is evolving so rapidly, and cancer is so many different diseases, it’s impossible for someone making these decisions to know everything they would need to know about why this patient is likely to survive their cancer and this patient is not.”
He says that during the COVID-19 pandemic, concerns regarding public health transcend the well-being of individual patients and that consideration must be given to providing the maximum benefit to the greatest number of people.
“That makes perfect sense and is the appropriate and laudable goal during a public health emergency like this ... but one of the challenges is that there is this belief that a diagnosis of cancer is uniformly fatal,” Marron said.
“It’s certainly conceivable that it would be a better use of resources to give the last ventilator to a young, otherwise healthy patient rather than a patient with multiply recurrent progressive metastatic cancer,” he continued. “However, we want to ensure that there is at least a discussion where that information is made available, rather than just saying, ‘She’s got cancer. She’s a lost cause.’ ”
Cancer patients are doing very well
Concerns about cancer misconceptions have been circulating in the oncology community since the start of the pandemic. “It’s really important that people understand that cancer patients are doing very well nowadays, and even with a diagnosis of cancer, they can potentially live for many years,” Anne Chiang, MD, PhD, from the Smilow Cancer Network, New Haven, Connecticut, told Medscape Medical News in a recent interview.
Thus far, even in hard-hit New York City, fears that cancer patients may not be receiving appropriate care have not materialized, according to Mark Robson, MD, a medical oncologist at Memorial Sloan Kettering Cancer Center (MSKCC). “I would emphasize that cancer patients are ABSOLUTELY getting the care they need, including patients with metastatic disease,” he recently tweeted. “NOONE at @sloan_kettering (or anywhere else) is being ‘triaged’ because of advanced cancer. Period.”
Robson told Medscape Medical News that although MSKCC continues to provide oncology care to patients with cancer, “we are [also] treating them if they develop COVID. ... I am trying to help pivot the institution towards care in this setting.”
He said he agrees with Craig Spencer, MD, MPH, director of global health in emergency medicine at the New York–Presbyterian/Columbia University Medical Center, who recently tweeted, “If you need a ventilator, you get a ventilator. Let’s be clear – this isn’t being ‘rationed.’ ”
Marron emphasized that an important safeguard against uninformed decision making is appropriate planning. For hospitalized patients, this means oncologists who provide treatment should offer information even before it is requested. But he said the “duty to plan” begins long before that.
“Clinicians haven’t always been great at talking about death and long-term outcomes with their patients, but this really cranks up the importance of having those conversations, and having them early, even though it’s incredibly hard. If someone has expressed that they would never want to be put on a ventilator, it’s important now even more so that is made clear,” he said.
He said early responses to the ASCO statement suggest that it has calmed some concerns in the oncology community, “but it still remains to be seen whether individual institutions will take this to heart, because this unto itself cannot enforce anything – it is up to individual institutions. I am hopeful this will get to the people it needs to get to.”
This article first appeared on Medscape.com.
ASCO announces its own COVID-19 and cancer registry
Data will not be commercialized, unlike CancerLinQ
The American Society of Clinical Oncology (ASCO) has launched a registry to collect data on cancer patients with COVID-19 and is asking oncology practices across the United States to share information about their patients with the infection for educational purposes.
The new registry joins at least two other cancer and COVID-19 patient registries already underway in the U.S.
In a statement, ASCO President Howard “Skip” Burris III, MD said there is a need to know “how the virus is impacting our patients, their cancer treatment, and outcomes to inform current cancer care” and future care.
The web-based registry, known as the American Society of Clinical Oncology (ASCO) Survey on COVID-19 in Oncology Registry, is open to all U.S. oncology practices. Participating practices will receive an unspecified “nominal” payment for their data entry efforts.
The registry patient information will be stored on ASCO’s “Big Data” platform, known as CancerLinQ, but is being held apart from that pool of data. The registry information will not be available for commercial purposes, ASCO spokesperson Rachel Martin recently told Medscape Medical News.
Separately, CancerLinQ, which is a wholly owned subsidiary of ASCO, will continue to collect data from its participant oncology practices (as usual), including COVID-19 information.
CancerLinQ has been criticized by ethicists for allowing partner companies to sell access to its data (after stripping off patient identifiers), but without asking for patients’ permission, as reported last year by Medscape Medical News.
Eleven practices, including academic enterprises, have so far expressed interested in participating in the ASCO COVID-19 Registry.
Participating practices are requested to send in details about cancer patients with a confirmed COVID-19 diagnosis. As well as a baseline data capture form, they will need to provide details of subsequent status, treatment, and outcomes. Some patient-identifying data, including zip code, date of birth, gender, race, ethnicity, type of cancer, and comorbidities, will be collected for the purposes of analysis.
ASCO hopes to learn about characteristics of patients with cancer most impacted by COVID-19; estimates of disease severity; treatment modifications or delays; implementation of telemedicine in the cancer treatment setting; and clinical outcomes related to both COVID-19 and cancer.
ASCO says it will deliver periodic reports to the cancer community and the broader public on these and other “key learnings.” It also says that the registry is designed to capture point-in-time data as well as longitudinal data on how the virus will impact care and outcomes into 2021.
ASCO is not alone in its data collection efforts.
The COVID-19 and Cancer Consortium is already collecting information from more than 50 cancer centers and organizations on COVID-19 in patients with cancer. The American Society of Hematology (ASH) Research Collaborative COVID-19 Registry for Hematologic Malignancy is doing the same but with a focus on hematologic malignancies.
This article first appeared on Medscape.com.
Data will not be commercialized, unlike CancerLinQ
Data will not be commercialized, unlike CancerLinQ
The American Society of Clinical Oncology (ASCO) has launched a registry to collect data on cancer patients with COVID-19 and is asking oncology practices across the United States to share information about their patients with the infection for educational purposes.
The new registry joins at least two other cancer and COVID-19 patient registries already underway in the U.S.
In a statement, ASCO President Howard “Skip” Burris III, MD said there is a need to know “how the virus is impacting our patients, their cancer treatment, and outcomes to inform current cancer care” and future care.
The web-based registry, known as the American Society of Clinical Oncology (ASCO) Survey on COVID-19 in Oncology Registry, is open to all U.S. oncology practices. Participating practices will receive an unspecified “nominal” payment for their data entry efforts.
The registry patient information will be stored on ASCO’s “Big Data” platform, known as CancerLinQ, but is being held apart from that pool of data. The registry information will not be available for commercial purposes, ASCO spokesperson Rachel Martin recently told Medscape Medical News.
Separately, CancerLinQ, which is a wholly owned subsidiary of ASCO, will continue to collect data from its participant oncology practices (as usual), including COVID-19 information.
CancerLinQ has been criticized by ethicists for allowing partner companies to sell access to its data (after stripping off patient identifiers), but without asking for patients’ permission, as reported last year by Medscape Medical News.
Eleven practices, including academic enterprises, have so far expressed interested in participating in the ASCO COVID-19 Registry.
Participating practices are requested to send in details about cancer patients with a confirmed COVID-19 diagnosis. As well as a baseline data capture form, they will need to provide details of subsequent status, treatment, and outcomes. Some patient-identifying data, including zip code, date of birth, gender, race, ethnicity, type of cancer, and comorbidities, will be collected for the purposes of analysis.
ASCO hopes to learn about characteristics of patients with cancer most impacted by COVID-19; estimates of disease severity; treatment modifications or delays; implementation of telemedicine in the cancer treatment setting; and clinical outcomes related to both COVID-19 and cancer.
ASCO says it will deliver periodic reports to the cancer community and the broader public on these and other “key learnings.” It also says that the registry is designed to capture point-in-time data as well as longitudinal data on how the virus will impact care and outcomes into 2021.
ASCO is not alone in its data collection efforts.
The COVID-19 and Cancer Consortium is already collecting information from more than 50 cancer centers and organizations on COVID-19 in patients with cancer. The American Society of Hematology (ASH) Research Collaborative COVID-19 Registry for Hematologic Malignancy is doing the same but with a focus on hematologic malignancies.
This article first appeared on Medscape.com.
The American Society of Clinical Oncology (ASCO) has launched a registry to collect data on cancer patients with COVID-19 and is asking oncology practices across the United States to share information about their patients with the infection for educational purposes.
The new registry joins at least two other cancer and COVID-19 patient registries already underway in the U.S.
In a statement, ASCO President Howard “Skip” Burris III, MD said there is a need to know “how the virus is impacting our patients, their cancer treatment, and outcomes to inform current cancer care” and future care.
The web-based registry, known as the American Society of Clinical Oncology (ASCO) Survey on COVID-19 in Oncology Registry, is open to all U.S. oncology practices. Participating practices will receive an unspecified “nominal” payment for their data entry efforts.
The registry patient information will be stored on ASCO’s “Big Data” platform, known as CancerLinQ, but is being held apart from that pool of data. The registry information will not be available for commercial purposes, ASCO spokesperson Rachel Martin recently told Medscape Medical News.
Separately, CancerLinQ, which is a wholly owned subsidiary of ASCO, will continue to collect data from its participant oncology practices (as usual), including COVID-19 information.
CancerLinQ has been criticized by ethicists for allowing partner companies to sell access to its data (after stripping off patient identifiers), but without asking for patients’ permission, as reported last year by Medscape Medical News.
Eleven practices, including academic enterprises, have so far expressed interested in participating in the ASCO COVID-19 Registry.
Participating practices are requested to send in details about cancer patients with a confirmed COVID-19 diagnosis. As well as a baseline data capture form, they will need to provide details of subsequent status, treatment, and outcomes. Some patient-identifying data, including zip code, date of birth, gender, race, ethnicity, type of cancer, and comorbidities, will be collected for the purposes of analysis.
ASCO hopes to learn about characteristics of patients with cancer most impacted by COVID-19; estimates of disease severity; treatment modifications or delays; implementation of telemedicine in the cancer treatment setting; and clinical outcomes related to both COVID-19 and cancer.
ASCO says it will deliver periodic reports to the cancer community and the broader public on these and other “key learnings.” It also says that the registry is designed to capture point-in-time data as well as longitudinal data on how the virus will impact care and outcomes into 2021.
ASCO is not alone in its data collection efforts.
The COVID-19 and Cancer Consortium is already collecting information from more than 50 cancer centers and organizations on COVID-19 in patients with cancer. The American Society of Hematology (ASH) Research Collaborative COVID-19 Registry for Hematologic Malignancy is doing the same but with a focus on hematologic malignancies.
This article first appeared on Medscape.com.
May 2020 – ICYMI
Gastroenterology
February 2020
Gastric electrical stimulation reduces refractory vomiting in a randomized crossover trial. Philippe Ducrotte et al. 2020 Feb;158(3):506-14.e2. doi: 10.1053/j.gastro.2019.10.018
Efficacy and safety of vedolizumab subcutaneous formulation in a randomized trial of patients with ulcerative colitis. William J. Sandborn et al. 2020 Feb;158(3)562-72.e12. doi: 10.1053/j.gastro.2019.08.027
AGA Clinical Practice Guidelines on management of gastric intestinal metaplasia. Samir Gupta et al. 2020 Feb;158(3):693-702. doi: 10.1053/j.gastro.2019.12.003
March 2020
Approaches and challenges to management of pediatric and adult patients with eosinophilic esophagitis. Ikuo Hirano, Glenn T. Furuta. 2020 Mar;158(4):840-51. doi: 10.1053/j.gastro.2019.09.052
Uptake of colorectal cancer screening by physicians is associated with greater uptake by their patients. Owen Litwin et al. 2020 Mar;158(4):905-14. doi: 10.1053/j.gastro.2019.10.027
Recommendations for follow-up after colonoscopy and polypectomy: A consensus update by the US Multi-Society Task Force on Colorectal Cancer. Samir Gupta et al. 2020 Mar;158(4):1131-53.e5. doi: 10.1053/j.gastro.2019.10.026
Differences in fecal microbiomes and metabolomes of people with vs without irritable bowel syndrome and bile acid malabsorption. Ian B. Jeffery et al. 2020 Mar;158(4):1016-28.e8. doi: 10.1053/j.gastro.2019.11.301
April 2020
How to set up a successful motility lab. Rena Yadlapati et al. 2020 April;158(5):1202-10. doi: 10.1053/j.gastro.2020.01.030
Mechanisms, evaluation, and management of chronic constipation. Adil E. Bharucha, Brian E. Lacy. 2020 April;158(5):1232-49.e3. doi: 10.1053/j.gastro.2019.12.034.
Incidence of venous thromboembolism in patients with newly diagnosed pancreatic cancer and factors associated with outcomes. Corinne Frere et al. 2020 April;158(5):1346-58.e4. doi: 10.1053/j.gastro.2019.12.009
Clinical Gastroenterology and Hepatology
February 2020
Increased incidence and mortality of gastric cancer in immigrant populations from high to low regions of incidence: A systematic review and meta-analysis. Baldeep S. Pabla et al. 2020 Feb;18(2):347-59.e5. doi: 10.1016/j.cgh.2019.05.032
Risk of gastrointestinal bleeding increases with combinations of antithrombotic agents and patient age. Neena S. Abraham et al. 2020 Feb;18(2):337-46.e19. doi: 10.1016/j.cgh.2019.05.017
Alcohol rehabilitation within 30 days of hospital discharge is associated with reduced readmission, relapse, and death in patients with alcoholic hepatitis. Thoetchai (Bee) Peeraphatdit et al. 2020 Feb;18(2):477-85.e5. doi: 10.1016/j.cgh.2019.04.048
March 2020
Telemedicine in gastroenterology: A value-added service for patients. Theresa Lee, Lawrence Kim. 2020 Mar;18(3):530-3. doi: 10.1016/j.cgh.2019.12.005
Best practices in teaching endoscopy based on a Delphi survey of gastroenterology program directors and experts in endoscopy education. Navin L. Kumar et al. 2020 Mar;18(3):574-9.e1. doi: 10.1016/j.cgh.2019.05.023
Consumption of fish and long-chain n-3 polyunsaturated fatty acids is associated with reduced risk of colorectal cancer in a large European cohort. Elom K. Aglago et al. 2020 Mar;18(3):654-66.e6. doi: 10.1016/j.cgh.2019.06.031
April 2020
Low incidence of aerodigestive cancers in patients with negative results from colonoscopies, regardless of findings from multitarget stool DNA tests. Barry M. Berger et al. 2020 April;18(4):864-71. doi: 10.1016/j.cgh.2019.07.057
Lifetime economic burden of Crohn’s disease and ulcerative colitis by age at diagnosis. Gary R. Lichtenstein et al. 2020 April;18(4):889-97.e10. doi: 10.1016/j.cgh.2019.07.022
Clinical and Molecular Gastroenterology and Hepatology
Etiopathogenetic mechanisms in diverticular disease of the colon. Michael Camilleri et al. 2020;9(1):15-32. doi: 10.1016/j.jcmgh.2019.07.007
Gastroenterology
February 2020
Gastric electrical stimulation reduces refractory vomiting in a randomized crossover trial. Philippe Ducrotte et al. 2020 Feb;158(3):506-14.e2. doi: 10.1053/j.gastro.2019.10.018
Efficacy and safety of vedolizumab subcutaneous formulation in a randomized trial of patients with ulcerative colitis. William J. Sandborn et al. 2020 Feb;158(3)562-72.e12. doi: 10.1053/j.gastro.2019.08.027
AGA Clinical Practice Guidelines on management of gastric intestinal metaplasia. Samir Gupta et al. 2020 Feb;158(3):693-702. doi: 10.1053/j.gastro.2019.12.003
March 2020
Approaches and challenges to management of pediatric and adult patients with eosinophilic esophagitis. Ikuo Hirano, Glenn T. Furuta. 2020 Mar;158(4):840-51. doi: 10.1053/j.gastro.2019.09.052
Uptake of colorectal cancer screening by physicians is associated with greater uptake by their patients. Owen Litwin et al. 2020 Mar;158(4):905-14. doi: 10.1053/j.gastro.2019.10.027
Recommendations for follow-up after colonoscopy and polypectomy: A consensus update by the US Multi-Society Task Force on Colorectal Cancer. Samir Gupta et al. 2020 Mar;158(4):1131-53.e5. doi: 10.1053/j.gastro.2019.10.026
Differences in fecal microbiomes and metabolomes of people with vs without irritable bowel syndrome and bile acid malabsorption. Ian B. Jeffery et al. 2020 Mar;158(4):1016-28.e8. doi: 10.1053/j.gastro.2019.11.301
April 2020
How to set up a successful motility lab. Rena Yadlapati et al. 2020 April;158(5):1202-10. doi: 10.1053/j.gastro.2020.01.030
Mechanisms, evaluation, and management of chronic constipation. Adil E. Bharucha, Brian E. Lacy. 2020 April;158(5):1232-49.e3. doi: 10.1053/j.gastro.2019.12.034.
Incidence of venous thromboembolism in patients with newly diagnosed pancreatic cancer and factors associated with outcomes. Corinne Frere et al. 2020 April;158(5):1346-58.e4. doi: 10.1053/j.gastro.2019.12.009
Clinical Gastroenterology and Hepatology
February 2020
Increased incidence and mortality of gastric cancer in immigrant populations from high to low regions of incidence: A systematic review and meta-analysis. Baldeep S. Pabla et al. 2020 Feb;18(2):347-59.e5. doi: 10.1016/j.cgh.2019.05.032
Risk of gastrointestinal bleeding increases with combinations of antithrombotic agents and patient age. Neena S. Abraham et al. 2020 Feb;18(2):337-46.e19. doi: 10.1016/j.cgh.2019.05.017
Alcohol rehabilitation within 30 days of hospital discharge is associated with reduced readmission, relapse, and death in patients with alcoholic hepatitis. Thoetchai (Bee) Peeraphatdit et al. 2020 Feb;18(2):477-85.e5. doi: 10.1016/j.cgh.2019.04.048
March 2020
Telemedicine in gastroenterology: A value-added service for patients. Theresa Lee, Lawrence Kim. 2020 Mar;18(3):530-3. doi: 10.1016/j.cgh.2019.12.005
Best practices in teaching endoscopy based on a Delphi survey of gastroenterology program directors and experts in endoscopy education. Navin L. Kumar et al. 2020 Mar;18(3):574-9.e1. doi: 10.1016/j.cgh.2019.05.023
Consumption of fish and long-chain n-3 polyunsaturated fatty acids is associated with reduced risk of colorectal cancer in a large European cohort. Elom K. Aglago et al. 2020 Mar;18(3):654-66.e6. doi: 10.1016/j.cgh.2019.06.031
April 2020
Low incidence of aerodigestive cancers in patients with negative results from colonoscopies, regardless of findings from multitarget stool DNA tests. Barry M. Berger et al. 2020 April;18(4):864-71. doi: 10.1016/j.cgh.2019.07.057
Lifetime economic burden of Crohn’s disease and ulcerative colitis by age at diagnosis. Gary R. Lichtenstein et al. 2020 April;18(4):889-97.e10. doi: 10.1016/j.cgh.2019.07.022
Clinical and Molecular Gastroenterology and Hepatology
Etiopathogenetic mechanisms in diverticular disease of the colon. Michael Camilleri et al. 2020;9(1):15-32. doi: 10.1016/j.jcmgh.2019.07.007
Gastroenterology
February 2020
Gastric electrical stimulation reduces refractory vomiting in a randomized crossover trial. Philippe Ducrotte et al. 2020 Feb;158(3):506-14.e2. doi: 10.1053/j.gastro.2019.10.018
Efficacy and safety of vedolizumab subcutaneous formulation in a randomized trial of patients with ulcerative colitis. William J. Sandborn et al. 2020 Feb;158(3)562-72.e12. doi: 10.1053/j.gastro.2019.08.027
AGA Clinical Practice Guidelines on management of gastric intestinal metaplasia. Samir Gupta et al. 2020 Feb;158(3):693-702. doi: 10.1053/j.gastro.2019.12.003
March 2020
Approaches and challenges to management of pediatric and adult patients with eosinophilic esophagitis. Ikuo Hirano, Glenn T. Furuta. 2020 Mar;158(4):840-51. doi: 10.1053/j.gastro.2019.09.052
Uptake of colorectal cancer screening by physicians is associated with greater uptake by their patients. Owen Litwin et al. 2020 Mar;158(4):905-14. doi: 10.1053/j.gastro.2019.10.027
Recommendations for follow-up after colonoscopy and polypectomy: A consensus update by the US Multi-Society Task Force on Colorectal Cancer. Samir Gupta et al. 2020 Mar;158(4):1131-53.e5. doi: 10.1053/j.gastro.2019.10.026
Differences in fecal microbiomes and metabolomes of people with vs without irritable bowel syndrome and bile acid malabsorption. Ian B. Jeffery et al. 2020 Mar;158(4):1016-28.e8. doi: 10.1053/j.gastro.2019.11.301
April 2020
How to set up a successful motility lab. Rena Yadlapati et al. 2020 April;158(5):1202-10. doi: 10.1053/j.gastro.2020.01.030
Mechanisms, evaluation, and management of chronic constipation. Adil E. Bharucha, Brian E. Lacy. 2020 April;158(5):1232-49.e3. doi: 10.1053/j.gastro.2019.12.034.
Incidence of venous thromboembolism in patients with newly diagnosed pancreatic cancer and factors associated with outcomes. Corinne Frere et al. 2020 April;158(5):1346-58.e4. doi: 10.1053/j.gastro.2019.12.009
Clinical Gastroenterology and Hepatology
February 2020
Increased incidence and mortality of gastric cancer in immigrant populations from high to low regions of incidence: A systematic review and meta-analysis. Baldeep S. Pabla et al. 2020 Feb;18(2):347-59.e5. doi: 10.1016/j.cgh.2019.05.032
Risk of gastrointestinal bleeding increases with combinations of antithrombotic agents and patient age. Neena S. Abraham et al. 2020 Feb;18(2):337-46.e19. doi: 10.1016/j.cgh.2019.05.017
Alcohol rehabilitation within 30 days of hospital discharge is associated with reduced readmission, relapse, and death in patients with alcoholic hepatitis. Thoetchai (Bee) Peeraphatdit et al. 2020 Feb;18(2):477-85.e5. doi: 10.1016/j.cgh.2019.04.048
March 2020
Telemedicine in gastroenterology: A value-added service for patients. Theresa Lee, Lawrence Kim. 2020 Mar;18(3):530-3. doi: 10.1016/j.cgh.2019.12.005
Best practices in teaching endoscopy based on a Delphi survey of gastroenterology program directors and experts in endoscopy education. Navin L. Kumar et al. 2020 Mar;18(3):574-9.e1. doi: 10.1016/j.cgh.2019.05.023
Consumption of fish and long-chain n-3 polyunsaturated fatty acids is associated with reduced risk of colorectal cancer in a large European cohort. Elom K. Aglago et al. 2020 Mar;18(3):654-66.e6. doi: 10.1016/j.cgh.2019.06.031
April 2020
Low incidence of aerodigestive cancers in patients with negative results from colonoscopies, regardless of findings from multitarget stool DNA tests. Barry M. Berger et al. 2020 April;18(4):864-71. doi: 10.1016/j.cgh.2019.07.057
Lifetime economic burden of Crohn’s disease and ulcerative colitis by age at diagnosis. Gary R. Lichtenstein et al. 2020 April;18(4):889-97.e10. doi: 10.1016/j.cgh.2019.07.022
Clinical and Molecular Gastroenterology and Hepatology
Etiopathogenetic mechanisms in diverticular disease of the colon. Michael Camilleri et al. 2020;9(1):15-32. doi: 10.1016/j.jcmgh.2019.07.007
Fellowship Burnout: What can we do to identify those at risk and minimize the impact?
Jeff is a high-performing first-year gastroenterology fellow who started with eagerness and enthusiasm. He seemed to enjoy talking to patients, wrote thorough notes, and often participated during case discussions at morning report. He initiated a quality improvement project and joined a hospital committee. Over the past few months, he has interacted less with his peers in the fellow’s office and stayed late to complete his patient encounters. He now frequently arrives late to work, is unprepared for rounds, and forgets to place important orders. One day, you notice him shuffling through several papers when the attending asks him a question about his patient. Later that day, he snapped at a nurse who paged to ask a question about a patient who just had a colonoscopy. When you ask him how he is doing, he becomes tearful and reports that he is under a lot of stress between work and home and does not feel the work he is doing is meaningful.
Introduction
The above scenario is all too familiar. Gastroenterology training can be a stressful period in an individual’s life. Long hours, steep learning curves for new cognitive and mechanical skill sets, as well as managing personal relationships and responsibilities at home all contribute to the stress of training and finding appropriate work-life balance. These stressors can result in burnout. The last decade has brought about a renewed emphasis on mitigating the impact of occupational burnout and improving trainee lifestyle through interventions such as work-hour restrictions, resiliency training, instruction on the importance of sleep, and team-building activities.
The problem
The World Health Organization (WHO) defines occupational burnout as chronic work-related stress, which may be characterized by feelings of energy depletion, mental distance from one’s job or feelings of negativity toward it, and reduced professional efficacy. Occupational burnout has been identified as an increasing problem both in practicing providers and trainees. Surveys in gastroenterologists show rates of burnout ranging between 37% and 50%,1 with trainees and early-career physicians disproportionately affected.1,2Physicians along the entire training spectrum are more likely to report high emotional exhaustion, high depersonalization, and burnout than a population control sample.2
Several individual factors identified for those at increased risk for burnout include younger age, not being married, and being male.2 Individuals spending less than 20% of their time working on activities they find meaningful and productive were more likely to show evidence of burnout.1

Symptoms of burnout can have a profound impact on trainees’ work performance, personal interactions, and the learning environment as a whole. The Accreditation Council for Graduate Medical Education (ACGME) annual survey of trainees asks them how strongly they agree or disagree on various components of burnout such as how meaningful they find their work, if they have enough time to think and reflect, if they feel emotionally drained at work, and if they feel worn out and weary after work. The intent of these questions is to provide anonymous feedback to training programs to help identify year to year trends and intervene early to prevent occupational burnout from becoming an increasing issue.
The solution
Considerations for any intervention should take several factors into account: the impact it may have on training and the development of a competent physician in their individualized specialty, the sustainability of the intervention, and whether it is something that will be accepted by the invested parties.

One method proposed for preventing burnout during fellowship has been designated as the three R’s: relaxation, reflection, and regrouping.3
- Relax. In order to relax, trainees need ways to decompress. Activities such as exercise and social events can be helpful. Within our own program the fellows have started their own group exercise program, playing wallyball weekly before clinical duties. We also encourage use of vacation days and build comradery by organizing potluck dinners for major holidays, graduation parties at the program director’s house, and an end-of-the-year golf outing in which trainees play against staff followed by a discussion regarding the state of the program. More recently we have added one half-day per a quarter for morale and team building. During this first year, the activities in which trainees have collectively decided to participate include an escape room, a rock-climbing facility, and laser tag. The addition of more team-building days has been well received by our program’s trainees and the simple addition of these team-building days has resulted in the trainees interacting more together outside of work, particularly in the form of group dinners.

Walter Reed National Military Medical Center fellows gathering for wallyball. - Reflect. They describe reflection as a necessary checkpoint which typically occurs every 6 months.3 These “checkpoints” provide an opportunity to provide feedback to the fellow as well as check in on their well-being and receive feedback about the program. We give frequent feedback to fellows in the form of spot, rotational, and mid-/end-of-year feedback. Additionally, we have developed a unique feedback system in which the trainees meet at the end of the year to discuss collective feedback for the staff and the program. This feedback is collated by the chief fellow and given to the program director as anonymous feedback, which is then passed to the individual staff.
- Regroup. Finally, regrouping to form new strategies.3 This regrouping provides an opportunity to improve on areas in which the trainee may have a deficiency and build on their strengths. To facilitate regrouping, we identify a mentor within the department and occasionally in other departments to meet regularly with the trainee. A successful mentor ensures effective regrouping and can help the trainee avoid pitfalls that they may have experienced in similar situations.
Moving forward
Occupational burnout is a systemic problem within the medical field, with trainees disproportionately affected. It is imperative that training programs continue to work toward creating a culture that prevents development of burnout. Along with the ideas presented here, the ACGME has launched AWARE, which is a suite of resources directed specifically at the GME community, with a goal of mitigating stress and preventing burnout. No one approach will be universally applicable but continued awareness and efforts to address this on an individual and programmatic level should be encouraged.
Dr. Ordway is a chief fellow, Dr. Tritsch and Dr. Singla are associate program directors, and Dr. Torres the program director, division of gastroenterology and hepatology, Walter Reed National Military Medical Center, Bethesda, Md.
References
1. Barnes EL et al. Dig Dis Sci. 2019;64(2):302-6.
2. Dyrbye LN et al. Acad Med. 2014;89(3):443-51.
3. Waldo OA. J Am Coll Cardiol. 2015;66(11):1303-6.
Jeff is a high-performing first-year gastroenterology fellow who started with eagerness and enthusiasm. He seemed to enjoy talking to patients, wrote thorough notes, and often participated during case discussions at morning report. He initiated a quality improvement project and joined a hospital committee. Over the past few months, he has interacted less with his peers in the fellow’s office and stayed late to complete his patient encounters. He now frequently arrives late to work, is unprepared for rounds, and forgets to place important orders. One day, you notice him shuffling through several papers when the attending asks him a question about his patient. Later that day, he snapped at a nurse who paged to ask a question about a patient who just had a colonoscopy. When you ask him how he is doing, he becomes tearful and reports that he is under a lot of stress between work and home and does not feel the work he is doing is meaningful.
Introduction
The above scenario is all too familiar. Gastroenterology training can be a stressful period in an individual’s life. Long hours, steep learning curves for new cognitive and mechanical skill sets, as well as managing personal relationships and responsibilities at home all contribute to the stress of training and finding appropriate work-life balance. These stressors can result in burnout. The last decade has brought about a renewed emphasis on mitigating the impact of occupational burnout and improving trainee lifestyle through interventions such as work-hour restrictions, resiliency training, instruction on the importance of sleep, and team-building activities.
The problem
The World Health Organization (WHO) defines occupational burnout as chronic work-related stress, which may be characterized by feelings of energy depletion, mental distance from one’s job or feelings of negativity toward it, and reduced professional efficacy. Occupational burnout has been identified as an increasing problem both in practicing providers and trainees. Surveys in gastroenterologists show rates of burnout ranging between 37% and 50%,1 with trainees and early-career physicians disproportionately affected.1,2Physicians along the entire training spectrum are more likely to report high emotional exhaustion, high depersonalization, and burnout than a population control sample.2
Several individual factors identified for those at increased risk for burnout include younger age, not being married, and being male.2 Individuals spending less than 20% of their time working on activities they find meaningful and productive were more likely to show evidence of burnout.1

Symptoms of burnout can have a profound impact on trainees’ work performance, personal interactions, and the learning environment as a whole. The Accreditation Council for Graduate Medical Education (ACGME) annual survey of trainees asks them how strongly they agree or disagree on various components of burnout such as how meaningful they find their work, if they have enough time to think and reflect, if they feel emotionally drained at work, and if they feel worn out and weary after work. The intent of these questions is to provide anonymous feedback to training programs to help identify year to year trends and intervene early to prevent occupational burnout from becoming an increasing issue.
The solution
Considerations for any intervention should take several factors into account: the impact it may have on training and the development of a competent physician in their individualized specialty, the sustainability of the intervention, and whether it is something that will be accepted by the invested parties.

One method proposed for preventing burnout during fellowship has been designated as the three R’s: relaxation, reflection, and regrouping.3
- Relax. In order to relax, trainees need ways to decompress. Activities such as exercise and social events can be helpful. Within our own program the fellows have started their own group exercise program, playing wallyball weekly before clinical duties. We also encourage use of vacation days and build comradery by organizing potluck dinners for major holidays, graduation parties at the program director’s house, and an end-of-the-year golf outing in which trainees play against staff followed by a discussion regarding the state of the program. More recently we have added one half-day per a quarter for morale and team building. During this first year, the activities in which trainees have collectively decided to participate include an escape room, a rock-climbing facility, and laser tag. The addition of more team-building days has been well received by our program’s trainees and the simple addition of these team-building days has resulted in the trainees interacting more together outside of work, particularly in the form of group dinners.

Walter Reed National Military Medical Center fellows gathering for wallyball. - Reflect. They describe reflection as a necessary checkpoint which typically occurs every 6 months.3 These “checkpoints” provide an opportunity to provide feedback to the fellow as well as check in on their well-being and receive feedback about the program. We give frequent feedback to fellows in the form of spot, rotational, and mid-/end-of-year feedback. Additionally, we have developed a unique feedback system in which the trainees meet at the end of the year to discuss collective feedback for the staff and the program. This feedback is collated by the chief fellow and given to the program director as anonymous feedback, which is then passed to the individual staff.
- Regroup. Finally, regrouping to form new strategies.3 This regrouping provides an opportunity to improve on areas in which the trainee may have a deficiency and build on their strengths. To facilitate regrouping, we identify a mentor within the department and occasionally in other departments to meet regularly with the trainee. A successful mentor ensures effective regrouping and can help the trainee avoid pitfalls that they may have experienced in similar situations.
Moving forward
Occupational burnout is a systemic problem within the medical field, with trainees disproportionately affected. It is imperative that training programs continue to work toward creating a culture that prevents development of burnout. Along with the ideas presented here, the ACGME has launched AWARE, which is a suite of resources directed specifically at the GME community, with a goal of mitigating stress and preventing burnout. No one approach will be universally applicable but continued awareness and efforts to address this on an individual and programmatic level should be encouraged.
Dr. Ordway is a chief fellow, Dr. Tritsch and Dr. Singla are associate program directors, and Dr. Torres the program director, division of gastroenterology and hepatology, Walter Reed National Military Medical Center, Bethesda, Md.
References
1. Barnes EL et al. Dig Dis Sci. 2019;64(2):302-6.
2. Dyrbye LN et al. Acad Med. 2014;89(3):443-51.
3. Waldo OA. J Am Coll Cardiol. 2015;66(11):1303-6.
Jeff is a high-performing first-year gastroenterology fellow who started with eagerness and enthusiasm. He seemed to enjoy talking to patients, wrote thorough notes, and often participated during case discussions at morning report. He initiated a quality improvement project and joined a hospital committee. Over the past few months, he has interacted less with his peers in the fellow’s office and stayed late to complete his patient encounters. He now frequently arrives late to work, is unprepared for rounds, and forgets to place important orders. One day, you notice him shuffling through several papers when the attending asks him a question about his patient. Later that day, he snapped at a nurse who paged to ask a question about a patient who just had a colonoscopy. When you ask him how he is doing, he becomes tearful and reports that he is under a lot of stress between work and home and does not feel the work he is doing is meaningful.
Introduction
The above scenario is all too familiar. Gastroenterology training can be a stressful period in an individual’s life. Long hours, steep learning curves for new cognitive and mechanical skill sets, as well as managing personal relationships and responsibilities at home all contribute to the stress of training and finding appropriate work-life balance. These stressors can result in burnout. The last decade has brought about a renewed emphasis on mitigating the impact of occupational burnout and improving trainee lifestyle through interventions such as work-hour restrictions, resiliency training, instruction on the importance of sleep, and team-building activities.
The problem
The World Health Organization (WHO) defines occupational burnout as chronic work-related stress, which may be characterized by feelings of energy depletion, mental distance from one’s job or feelings of negativity toward it, and reduced professional efficacy. Occupational burnout has been identified as an increasing problem both in practicing providers and trainees. Surveys in gastroenterologists show rates of burnout ranging between 37% and 50%,1 with trainees and early-career physicians disproportionately affected.1,2Physicians along the entire training spectrum are more likely to report high emotional exhaustion, high depersonalization, and burnout than a population control sample.2
Several individual factors identified for those at increased risk for burnout include younger age, not being married, and being male.2 Individuals spending less than 20% of their time working on activities they find meaningful and productive were more likely to show evidence of burnout.1

Symptoms of burnout can have a profound impact on trainees’ work performance, personal interactions, and the learning environment as a whole. The Accreditation Council for Graduate Medical Education (ACGME) annual survey of trainees asks them how strongly they agree or disagree on various components of burnout such as how meaningful they find their work, if they have enough time to think and reflect, if they feel emotionally drained at work, and if they feel worn out and weary after work. The intent of these questions is to provide anonymous feedback to training programs to help identify year to year trends and intervene early to prevent occupational burnout from becoming an increasing issue.
The solution
Considerations for any intervention should take several factors into account: the impact it may have on training and the development of a competent physician in their individualized specialty, the sustainability of the intervention, and whether it is something that will be accepted by the invested parties.

One method proposed for preventing burnout during fellowship has been designated as the three R’s: relaxation, reflection, and regrouping.3
- Relax. In order to relax, trainees need ways to decompress. Activities such as exercise and social events can be helpful. Within our own program the fellows have started their own group exercise program, playing wallyball weekly before clinical duties. We also encourage use of vacation days and build comradery by organizing potluck dinners for major holidays, graduation parties at the program director’s house, and an end-of-the-year golf outing in which trainees play against staff followed by a discussion regarding the state of the program. More recently we have added one half-day per a quarter for morale and team building. During this first year, the activities in which trainees have collectively decided to participate include an escape room, a rock-climbing facility, and laser tag. The addition of more team-building days has been well received by our program’s trainees and the simple addition of these team-building days has resulted in the trainees interacting more together outside of work, particularly in the form of group dinners.

Walter Reed National Military Medical Center fellows gathering for wallyball. - Reflect. They describe reflection as a necessary checkpoint which typically occurs every 6 months.3 These “checkpoints” provide an opportunity to provide feedback to the fellow as well as check in on their well-being and receive feedback about the program. We give frequent feedback to fellows in the form of spot, rotational, and mid-/end-of-year feedback. Additionally, we have developed a unique feedback system in which the trainees meet at the end of the year to discuss collective feedback for the staff and the program. This feedback is collated by the chief fellow and given to the program director as anonymous feedback, which is then passed to the individual staff.
- Regroup. Finally, regrouping to form new strategies.3 This regrouping provides an opportunity to improve on areas in which the trainee may have a deficiency and build on their strengths. To facilitate regrouping, we identify a mentor within the department and occasionally in other departments to meet regularly with the trainee. A successful mentor ensures effective regrouping and can help the trainee avoid pitfalls that they may have experienced in similar situations.
Moving forward
Occupational burnout is a systemic problem within the medical field, with trainees disproportionately affected. It is imperative that training programs continue to work toward creating a culture that prevents development of burnout. Along with the ideas presented here, the ACGME has launched AWARE, which is a suite of resources directed specifically at the GME community, with a goal of mitigating stress and preventing burnout. No one approach will be universally applicable but continued awareness and efforts to address this on an individual and programmatic level should be encouraged.
Dr. Ordway is a chief fellow, Dr. Tritsch and Dr. Singla are associate program directors, and Dr. Torres the program director, division of gastroenterology and hepatology, Walter Reed National Military Medical Center, Bethesda, Md.
References
1. Barnes EL et al. Dig Dis Sci. 2019;64(2):302-6.
2. Dyrbye LN et al. Acad Med. 2014;89(3):443-51.
3. Waldo OA. J Am Coll Cardiol. 2015;66(11):1303-6.
The wide-ranging impact of hospital closures
Clinicians struggle to balance priorities
On June 26, 2019, American Academic Health System and Philadelphia Academic Health System announced that Hahnemann University Hospital, a 496-bed tertiary care center in North Philadelphia in operation for over 170 years, would close that September.
The emergency department closed 52 days after the announcement, leaving little time for physicians and staff to coordinate care for patients and secure new employment. The announcement was also made right at the beginning of the new academic year, which meant residents and fellows were forced to find new training programs. In total, 2,500 workers at Hahnemann, including more than 570 hospitalists and physicians training as residents and fellows, were displaced as the hospital closed – the largest such closing in U.S. history.
For most of its existence, Hahnemann was a teaching hospital. While trainees were all eventually placed in new programs thanks to efforts from the Accreditation Council for Graduate Medical Education (ACGME), some of the permanent staff at Hahnemann weren’t so lucky. A month after the announcement, Drexel University’s president told university employees that 40% of the staff who worked at Hahnemann would be cut as a result of the closing. Drexel, also based in Philadelphia, had long had an academic affiliation agreement for training Drexel’s medical school students as a primary academic partner. Overall, Drexel’s entire clinical staff at Hahnemann was let go, and Tower Health Medical Group is expected to hire about 60% of the former Hahnemann staff.
Kevin D’Mello, MD, FACP, FHM, a hospitalist and assistant professor of medicine at Drexel University, said residents during Hahnemann’s closure were essentially teaching themselves how to swim. “There were just no laws, no rules,” he said.
The vast majority of programs accepting applications from residents at Hahnemann were sympathetic and accommodating, he said, but a few programs applied “pressure tactics” to some of the residents offered a transfer position, despite graduate medical education rules in place to prevent such a situation from happening. “The resident says: ‘Oh, well, I’m waiting to hear from this other program,’ ” said Dr. D’Mello. “They’d say: ‘Okay, well, we’re giving you a position now. You have 12 hours to answer.’ ”
Decision makers at the hospital also were not very forthcoming with information to residents, fellows and program directors, according to a recent paper written by Thomas J. Nasca, MD, current president and CEO of ACGME, and colleagues in the journal Academic Medicine (Nasca T et al. Acad Med. 2019 Dec 17. doi: 10.1097/ACM.0000000000003133). When Dr. Nasca and colleagues went to investigate the situation at Hahnemann firsthand, “the team found that residents, fellows, and program directors alike considered their voices to have been ignored in decision making and deemed themselves ‘out of the loop’ of important information that would affect their career transitions.”
While the hospital closed in September 2019, the effects are still being felt. In Pennsylvania, the Medical Care Availability and Reduction of Error Act requires that hospitals and providers have malpractice insurance, including tail insurance for when a doctor’s insurance policy expires. American Academic announced it would not be paying tail insurance for claims made while physicians were at Hahnemann. This meant residents, fellows and physicians who worked at Hahnemann during the closure would be on the hook for paying their own malpractice insurance.
“On one hand, the risk is very low for the house staff. Lawsuits that come up later for house staff are generally dropped at some point,” said William W. Pinsky, MD, FAAP, FACC, president and CEO of the Educational Commission for Foreign Medical Graduates (ECFMG). “But who wants to take that risk going forward? It’s an issue that’s still not resolved.”
The American Medical Association, Association of American Medical Colleges (AAMC), the Philadelphia County Medical Society, and other medical societies have collectively put pressure on Hahnemann’s owners to pay for tail coverage. Beyond a Feb. 10, 2020 deadline, former Hahnemann physicians were still expected to cover their own tail insurance.
To further complicate matters, American Academic attempted to auction more than 570 residency slots at Hahnemann. The slots were sold to a consortium of six health systems in the area – Thomas Jefferson University Hospitals, Einstein Healthcare Network, Temple University Health System, Main Line Health, Cooper University Health Care, and Christiana Care Health System – for $55 million. The Centers for Medicare & Medicaid Services opposed the sale, arguing that the slots are a contract that hospitals enter into with CMS, rather than an asset to be sold. An appeal is currently pending.
The case is being watched by former physicians at Hahnemann. “American Academic said, ‘If we don’t get this $55 million, we’re not going to be able to cover this tail insurance.’ They’re kind of linking the two things,” said Dr. D’Mello. “To me, it’s almost like putting pressure to allow the sale to happen.”
Urban hospital closures disrupt health system balance
When an urban hospital like Hahnemann University Hospital closes, there is a major disruption to patient care. Patients need to relocate to other nearby centers, and they may not always be able to follow their physician to the next health center.
If patients have comorbidities, are being tracked across multiple care points, or change physicians during a hospital closure, details can be missed and care can become more complicated for physicians who end up seeing the patient at a new center. For example, a patient receiving obstetrics care at a hospital that closes will have to reschedule their delivery at another health center, noted Dr. Pinsky.
“Where patients get lost is when there’s not a physician or an individual can keep track of all that, coordinate, and help to be sure that the patient follows through,” he said.
Patients at a closing hospital need to go somewhere else for care, and patient volume naturally increases at other nearby centers, potentially causing problems for systems without the resources to handle the spike in traffic.
“I’m a service director of quality improvement and patient safety for Drexel internal medicine. I know that those sort of jumps and volumes are what increases medical errors and potentially could create some adverse outcomes,” said Dr. D’Mello. “That’s something I’m particularly worried about.”
Physicians are also reconciling their own personal situations during a hospital closure, attempting to figure out their next step while at the same time helping patients figure out theirs. In the case of international medical graduates on J-1 or H1-B visas, who are dependent on hospital positions and training programs to remain in the United States, the situation can be even more dire.
During Hahnemann’s closure, Dr. Pinsky said that the ECFMG, which represents 11,000 individuals with J-1 visas across the country, reached out to the 55 individuals on J-1 visas at the hospital and offered them assistance, including working with the Department of State to ensure they aren’t in jeopardy of deportation before they secure another training program position.
The ECFMG, AMA, AAMC, and ACGME also offered funding to help J-1 visa holders who needed to relocate outside Philadelphia. “Many of them spent a lot of their money or all their money just coming over here,” said Dr. Pinsky. “This was a way to help defray some immediate costs that they might have.”
Education and research, of which hospitalists and residents play a large role, are likewise affected during a hospital closure, Dr. Pinsky said. “Education and research in the hospital is an important contributor to the community, health care and medical education nationally overall. When it’s not considered, there can be a significant asset that is lost in the process, which is hard to ever regain.
“The hospitalists have an integral role in medical education. In most hospitals where there is graduate medical education, particularly in internal medicine or pediatrics, and where there is a hospitalist program, it’s the hospitalists that do the majority of the in-hospital or inpatient training and education,” he added.
Rural hospital closures affect access to care
Since 2005, 163 rural hospitals have closed in the United States. When rural hospitals close, the situation for hospitalists and other physicians is different. In communities where a larger health system owns a hospital, such as when Vidant Health closed Pungo District Hospital in Belhaven, N.C., in 2014 before reopening a nonemergency clinic in the area in 2016, health care services for the community may have limited interruption.
However, if there isn’t a nearby system to join, many doctors will end up leaving the area. More than half of rural hospitals that close end up not providing any kind of supplementary health care service, according to the NC Rural Health Research Program.
“A lot of the hospitals that have closed have not been owned by a system,” said George H. Pink, PhD, deputy director of the NC Rural Health Research Program at the University of North Carolina at Chapel Hill. “They’ve been independent, freestanding, and that perhaps is one of the reasons why they’re closing, is because they haven’t been able to find a system that would buy them out and inject capital into the community.”
This can also have an effect on the number of health care providers in the area, Dr. Pink said. “Their ability to refer patients and treat patients locally may be affected. That’s why, in many towns where hospitals have closed, we see a drop in the number of providers, particularly primary care doctors who actually live in the community.”
Politicians and federal entities have proposed a number of solutions to help protect rural hospitals from closure. Sen. Charles Grassley (R-Iowa), Sen. Amy Klobuchar (D-Minn.), and Sen. Cory Gardener (R-Colo.) have sponsored bills in the Senate, while Rep. Sam Graves (R-Mo.) has introduced legislation in the House. The Medicare Payment Advisory Commission has proposed two models of rural hospital care, and there are additional models proposed by the Kansas Hospital Association. A pilot program in Pennsylvania, the Pennsylvania Rural Health Model, is testing how a global budget by CMS for all inpatient and hospital-based outcomes might help rural hospitals.
“What we haven’t had a lot of action on is actually testing these models out and seeing whether they will work, and in what kinds of communities they will work,” Dr. Pink said.
Hospitalists as community advocates
Dr. D’Mello, who wrote an article for the Journal of Hospital Medicine on Hahnemann’s ownership by a private equity firm (doi: 10.12788/jhm.3378), said that the inherent nature of a for-profit entity trying to make a hospital profitable is a bad sign for a hospital and not necessarily what is in the best interest for an academic institution or for doctors who train there.
“I don’t know if I could blame the private equity firm completely, but in retrospect, the private equity firms stepping in was like the death knell of the hospital,” he said of Hahnemann’s closure.
“I think what the community needs to know – what the health care community, patient community, the hospitalist community need to know – is that there’s got to be more attention paid to these types of issues during mergers and acquisitions to prevent this from happening,” Dr. Pinsky said.
One larger issue was Hahnemann’s position as a safety net hospital, which partly played into American Academic’s lack of success in making the hospital as profitable as they wanted it to be, Dr. D’Mello noted. Hahnemann’s patient population consisted mostly of minority patients on Medicare, Medicaid, and charity care insurance, while recent studies have shown that hospitals are more likely to succeed when they have a larger proportion of patients with private insurance.
“Studies show that, to [make more] money from private insurance, you really have to have this huge footprint, because then you’ve got a better ability to negotiate with these private insurance companies,” Dr. D’Mello said. “Whether that’s actually good for health care is a different issue.”
Despite their own situations, it is not unusual for hospitalists and hospital physicians to step up during a hospital closure and advocate for their patients on behalf of the community, Dr. Pink said.
“When hospitals are in financial difficulty and there’s the risk of closure, typically, the medical staff are among the first to step up and warn the community: ‘We’re at risk of losing our service. We need some help,’ ” he said. “Generally speaking, the local physicians have been at the forefront of helping to keep access to hospital care available in some of these small communities – unfortunately, not always successfully.”
Dr. D’Mello, Dr. Pinsky, and Dr. Pink report no relevant conflicts of interest.
Clinicians struggle to balance priorities
Clinicians struggle to balance priorities
On June 26, 2019, American Academic Health System and Philadelphia Academic Health System announced that Hahnemann University Hospital, a 496-bed tertiary care center in North Philadelphia in operation for over 170 years, would close that September.
The emergency department closed 52 days after the announcement, leaving little time for physicians and staff to coordinate care for patients and secure new employment. The announcement was also made right at the beginning of the new academic year, which meant residents and fellows were forced to find new training programs. In total, 2,500 workers at Hahnemann, including more than 570 hospitalists and physicians training as residents and fellows, were displaced as the hospital closed – the largest such closing in U.S. history.
For most of its existence, Hahnemann was a teaching hospital. While trainees were all eventually placed in new programs thanks to efforts from the Accreditation Council for Graduate Medical Education (ACGME), some of the permanent staff at Hahnemann weren’t so lucky. A month after the announcement, Drexel University’s president told university employees that 40% of the staff who worked at Hahnemann would be cut as a result of the closing. Drexel, also based in Philadelphia, had long had an academic affiliation agreement for training Drexel’s medical school students as a primary academic partner. Overall, Drexel’s entire clinical staff at Hahnemann was let go, and Tower Health Medical Group is expected to hire about 60% of the former Hahnemann staff.
Kevin D’Mello, MD, FACP, FHM, a hospitalist and assistant professor of medicine at Drexel University, said residents during Hahnemann’s closure were essentially teaching themselves how to swim. “There were just no laws, no rules,” he said.
The vast majority of programs accepting applications from residents at Hahnemann were sympathetic and accommodating, he said, but a few programs applied “pressure tactics” to some of the residents offered a transfer position, despite graduate medical education rules in place to prevent such a situation from happening. “The resident says: ‘Oh, well, I’m waiting to hear from this other program,’ ” said Dr. D’Mello. “They’d say: ‘Okay, well, we’re giving you a position now. You have 12 hours to answer.’ ”
Decision makers at the hospital also were not very forthcoming with information to residents, fellows and program directors, according to a recent paper written by Thomas J. Nasca, MD, current president and CEO of ACGME, and colleagues in the journal Academic Medicine (Nasca T et al. Acad Med. 2019 Dec 17. doi: 10.1097/ACM.0000000000003133). When Dr. Nasca and colleagues went to investigate the situation at Hahnemann firsthand, “the team found that residents, fellows, and program directors alike considered their voices to have been ignored in decision making and deemed themselves ‘out of the loop’ of important information that would affect their career transitions.”
While the hospital closed in September 2019, the effects are still being felt. In Pennsylvania, the Medical Care Availability and Reduction of Error Act requires that hospitals and providers have malpractice insurance, including tail insurance for when a doctor’s insurance policy expires. American Academic announced it would not be paying tail insurance for claims made while physicians were at Hahnemann. This meant residents, fellows and physicians who worked at Hahnemann during the closure would be on the hook for paying their own malpractice insurance.
“On one hand, the risk is very low for the house staff. Lawsuits that come up later for house staff are generally dropped at some point,” said William W. Pinsky, MD, FAAP, FACC, president and CEO of the Educational Commission for Foreign Medical Graduates (ECFMG). “But who wants to take that risk going forward? It’s an issue that’s still not resolved.”
The American Medical Association, Association of American Medical Colleges (AAMC), the Philadelphia County Medical Society, and other medical societies have collectively put pressure on Hahnemann’s owners to pay for tail coverage. Beyond a Feb. 10, 2020 deadline, former Hahnemann physicians were still expected to cover their own tail insurance.
To further complicate matters, American Academic attempted to auction more than 570 residency slots at Hahnemann. The slots were sold to a consortium of six health systems in the area – Thomas Jefferson University Hospitals, Einstein Healthcare Network, Temple University Health System, Main Line Health, Cooper University Health Care, and Christiana Care Health System – for $55 million. The Centers for Medicare & Medicaid Services opposed the sale, arguing that the slots are a contract that hospitals enter into with CMS, rather than an asset to be sold. An appeal is currently pending.
The case is being watched by former physicians at Hahnemann. “American Academic said, ‘If we don’t get this $55 million, we’re not going to be able to cover this tail insurance.’ They’re kind of linking the two things,” said Dr. D’Mello. “To me, it’s almost like putting pressure to allow the sale to happen.”
Urban hospital closures disrupt health system balance
When an urban hospital like Hahnemann University Hospital closes, there is a major disruption to patient care. Patients need to relocate to other nearby centers, and they may not always be able to follow their physician to the next health center.
If patients have comorbidities, are being tracked across multiple care points, or change physicians during a hospital closure, details can be missed and care can become more complicated for physicians who end up seeing the patient at a new center. For example, a patient receiving obstetrics care at a hospital that closes will have to reschedule their delivery at another health center, noted Dr. Pinsky.
“Where patients get lost is when there’s not a physician or an individual can keep track of all that, coordinate, and help to be sure that the patient follows through,” he said.
Patients at a closing hospital need to go somewhere else for care, and patient volume naturally increases at other nearby centers, potentially causing problems for systems without the resources to handle the spike in traffic.
“I’m a service director of quality improvement and patient safety for Drexel internal medicine. I know that those sort of jumps and volumes are what increases medical errors and potentially could create some adverse outcomes,” said Dr. D’Mello. “That’s something I’m particularly worried about.”
Physicians are also reconciling their own personal situations during a hospital closure, attempting to figure out their next step while at the same time helping patients figure out theirs. In the case of international medical graduates on J-1 or H1-B visas, who are dependent on hospital positions and training programs to remain in the United States, the situation can be even more dire.
During Hahnemann’s closure, Dr. Pinsky said that the ECFMG, which represents 11,000 individuals with J-1 visas across the country, reached out to the 55 individuals on J-1 visas at the hospital and offered them assistance, including working with the Department of State to ensure they aren’t in jeopardy of deportation before they secure another training program position.
The ECFMG, AMA, AAMC, and ACGME also offered funding to help J-1 visa holders who needed to relocate outside Philadelphia. “Many of them spent a lot of their money or all their money just coming over here,” said Dr. Pinsky. “This was a way to help defray some immediate costs that they might have.”
Education and research, of which hospitalists and residents play a large role, are likewise affected during a hospital closure, Dr. Pinsky said. “Education and research in the hospital is an important contributor to the community, health care and medical education nationally overall. When it’s not considered, there can be a significant asset that is lost in the process, which is hard to ever regain.
“The hospitalists have an integral role in medical education. In most hospitals where there is graduate medical education, particularly in internal medicine or pediatrics, and where there is a hospitalist program, it’s the hospitalists that do the majority of the in-hospital or inpatient training and education,” he added.
Rural hospital closures affect access to care
Since 2005, 163 rural hospitals have closed in the United States. When rural hospitals close, the situation for hospitalists and other physicians is different. In communities where a larger health system owns a hospital, such as when Vidant Health closed Pungo District Hospital in Belhaven, N.C., in 2014 before reopening a nonemergency clinic in the area in 2016, health care services for the community may have limited interruption.
However, if there isn’t a nearby system to join, many doctors will end up leaving the area. More than half of rural hospitals that close end up not providing any kind of supplementary health care service, according to the NC Rural Health Research Program.
“A lot of the hospitals that have closed have not been owned by a system,” said George H. Pink, PhD, deputy director of the NC Rural Health Research Program at the University of North Carolina at Chapel Hill. “They’ve been independent, freestanding, and that perhaps is one of the reasons why they’re closing, is because they haven’t been able to find a system that would buy them out and inject capital into the community.”
This can also have an effect on the number of health care providers in the area, Dr. Pink said. “Their ability to refer patients and treat patients locally may be affected. That’s why, in many towns where hospitals have closed, we see a drop in the number of providers, particularly primary care doctors who actually live in the community.”
Politicians and federal entities have proposed a number of solutions to help protect rural hospitals from closure. Sen. Charles Grassley (R-Iowa), Sen. Amy Klobuchar (D-Minn.), and Sen. Cory Gardener (R-Colo.) have sponsored bills in the Senate, while Rep. Sam Graves (R-Mo.) has introduced legislation in the House. The Medicare Payment Advisory Commission has proposed two models of rural hospital care, and there are additional models proposed by the Kansas Hospital Association. A pilot program in Pennsylvania, the Pennsylvania Rural Health Model, is testing how a global budget by CMS for all inpatient and hospital-based outcomes might help rural hospitals.
“What we haven’t had a lot of action on is actually testing these models out and seeing whether they will work, and in what kinds of communities they will work,” Dr. Pink said.
Hospitalists as community advocates
Dr. D’Mello, who wrote an article for the Journal of Hospital Medicine on Hahnemann’s ownership by a private equity firm (doi: 10.12788/jhm.3378), said that the inherent nature of a for-profit entity trying to make a hospital profitable is a bad sign for a hospital and not necessarily what is in the best interest for an academic institution or for doctors who train there.
“I don’t know if I could blame the private equity firm completely, but in retrospect, the private equity firms stepping in was like the death knell of the hospital,” he said of Hahnemann’s closure.
“I think what the community needs to know – what the health care community, patient community, the hospitalist community need to know – is that there’s got to be more attention paid to these types of issues during mergers and acquisitions to prevent this from happening,” Dr. Pinsky said.
One larger issue was Hahnemann’s position as a safety net hospital, which partly played into American Academic’s lack of success in making the hospital as profitable as they wanted it to be, Dr. D’Mello noted. Hahnemann’s patient population consisted mostly of minority patients on Medicare, Medicaid, and charity care insurance, while recent studies have shown that hospitals are more likely to succeed when they have a larger proportion of patients with private insurance.
“Studies show that, to [make more] money from private insurance, you really have to have this huge footprint, because then you’ve got a better ability to negotiate with these private insurance companies,” Dr. D’Mello said. “Whether that’s actually good for health care is a different issue.”
Despite their own situations, it is not unusual for hospitalists and hospital physicians to step up during a hospital closure and advocate for their patients on behalf of the community, Dr. Pink said.
“When hospitals are in financial difficulty and there’s the risk of closure, typically, the medical staff are among the first to step up and warn the community: ‘We’re at risk of losing our service. We need some help,’ ” he said. “Generally speaking, the local physicians have been at the forefront of helping to keep access to hospital care available in some of these small communities – unfortunately, not always successfully.”
Dr. D’Mello, Dr. Pinsky, and Dr. Pink report no relevant conflicts of interest.
On June 26, 2019, American Academic Health System and Philadelphia Academic Health System announced that Hahnemann University Hospital, a 496-bed tertiary care center in North Philadelphia in operation for over 170 years, would close that September.
The emergency department closed 52 days after the announcement, leaving little time for physicians and staff to coordinate care for patients and secure new employment. The announcement was also made right at the beginning of the new academic year, which meant residents and fellows were forced to find new training programs. In total, 2,500 workers at Hahnemann, including more than 570 hospitalists and physicians training as residents and fellows, were displaced as the hospital closed – the largest such closing in U.S. history.
For most of its existence, Hahnemann was a teaching hospital. While trainees were all eventually placed in new programs thanks to efforts from the Accreditation Council for Graduate Medical Education (ACGME), some of the permanent staff at Hahnemann weren’t so lucky. A month after the announcement, Drexel University’s president told university employees that 40% of the staff who worked at Hahnemann would be cut as a result of the closing. Drexel, also based in Philadelphia, had long had an academic affiliation agreement for training Drexel’s medical school students as a primary academic partner. Overall, Drexel’s entire clinical staff at Hahnemann was let go, and Tower Health Medical Group is expected to hire about 60% of the former Hahnemann staff.
Kevin D’Mello, MD, FACP, FHM, a hospitalist and assistant professor of medicine at Drexel University, said residents during Hahnemann’s closure were essentially teaching themselves how to swim. “There were just no laws, no rules,” he said.
The vast majority of programs accepting applications from residents at Hahnemann were sympathetic and accommodating, he said, but a few programs applied “pressure tactics” to some of the residents offered a transfer position, despite graduate medical education rules in place to prevent such a situation from happening. “The resident says: ‘Oh, well, I’m waiting to hear from this other program,’ ” said Dr. D’Mello. “They’d say: ‘Okay, well, we’re giving you a position now. You have 12 hours to answer.’ ”
Decision makers at the hospital also were not very forthcoming with information to residents, fellows and program directors, according to a recent paper written by Thomas J. Nasca, MD, current president and CEO of ACGME, and colleagues in the journal Academic Medicine (Nasca T et al. Acad Med. 2019 Dec 17. doi: 10.1097/ACM.0000000000003133). When Dr. Nasca and colleagues went to investigate the situation at Hahnemann firsthand, “the team found that residents, fellows, and program directors alike considered their voices to have been ignored in decision making and deemed themselves ‘out of the loop’ of important information that would affect their career transitions.”
While the hospital closed in September 2019, the effects are still being felt. In Pennsylvania, the Medical Care Availability and Reduction of Error Act requires that hospitals and providers have malpractice insurance, including tail insurance for when a doctor’s insurance policy expires. American Academic announced it would not be paying tail insurance for claims made while physicians were at Hahnemann. This meant residents, fellows and physicians who worked at Hahnemann during the closure would be on the hook for paying their own malpractice insurance.
“On one hand, the risk is very low for the house staff. Lawsuits that come up later for house staff are generally dropped at some point,” said William W. Pinsky, MD, FAAP, FACC, president and CEO of the Educational Commission for Foreign Medical Graduates (ECFMG). “But who wants to take that risk going forward? It’s an issue that’s still not resolved.”
The American Medical Association, Association of American Medical Colleges (AAMC), the Philadelphia County Medical Society, and other medical societies have collectively put pressure on Hahnemann’s owners to pay for tail coverage. Beyond a Feb. 10, 2020 deadline, former Hahnemann physicians were still expected to cover their own tail insurance.
To further complicate matters, American Academic attempted to auction more than 570 residency slots at Hahnemann. The slots were sold to a consortium of six health systems in the area – Thomas Jefferson University Hospitals, Einstein Healthcare Network, Temple University Health System, Main Line Health, Cooper University Health Care, and Christiana Care Health System – for $55 million. The Centers for Medicare & Medicaid Services opposed the sale, arguing that the slots are a contract that hospitals enter into with CMS, rather than an asset to be sold. An appeal is currently pending.
The case is being watched by former physicians at Hahnemann. “American Academic said, ‘If we don’t get this $55 million, we’re not going to be able to cover this tail insurance.’ They’re kind of linking the two things,” said Dr. D’Mello. “To me, it’s almost like putting pressure to allow the sale to happen.”
Urban hospital closures disrupt health system balance
When an urban hospital like Hahnemann University Hospital closes, there is a major disruption to patient care. Patients need to relocate to other nearby centers, and they may not always be able to follow their physician to the next health center.
If patients have comorbidities, are being tracked across multiple care points, or change physicians during a hospital closure, details can be missed and care can become more complicated for physicians who end up seeing the patient at a new center. For example, a patient receiving obstetrics care at a hospital that closes will have to reschedule their delivery at another health center, noted Dr. Pinsky.
“Where patients get lost is when there’s not a physician or an individual can keep track of all that, coordinate, and help to be sure that the patient follows through,” he said.
Patients at a closing hospital need to go somewhere else for care, and patient volume naturally increases at other nearby centers, potentially causing problems for systems without the resources to handle the spike in traffic.
“I’m a service director of quality improvement and patient safety for Drexel internal medicine. I know that those sort of jumps and volumes are what increases medical errors and potentially could create some adverse outcomes,” said Dr. D’Mello. “That’s something I’m particularly worried about.”
Physicians are also reconciling their own personal situations during a hospital closure, attempting to figure out their next step while at the same time helping patients figure out theirs. In the case of international medical graduates on J-1 or H1-B visas, who are dependent on hospital positions and training programs to remain in the United States, the situation can be even more dire.
During Hahnemann’s closure, Dr. Pinsky said that the ECFMG, which represents 11,000 individuals with J-1 visas across the country, reached out to the 55 individuals on J-1 visas at the hospital and offered them assistance, including working with the Department of State to ensure they aren’t in jeopardy of deportation before they secure another training program position.
The ECFMG, AMA, AAMC, and ACGME also offered funding to help J-1 visa holders who needed to relocate outside Philadelphia. “Many of them spent a lot of their money or all their money just coming over here,” said Dr. Pinsky. “This was a way to help defray some immediate costs that they might have.”
Education and research, of which hospitalists and residents play a large role, are likewise affected during a hospital closure, Dr. Pinsky said. “Education and research in the hospital is an important contributor to the community, health care and medical education nationally overall. When it’s not considered, there can be a significant asset that is lost in the process, which is hard to ever regain.
“The hospitalists have an integral role in medical education. In most hospitals where there is graduate medical education, particularly in internal medicine or pediatrics, and where there is a hospitalist program, it’s the hospitalists that do the majority of the in-hospital or inpatient training and education,” he added.
Rural hospital closures affect access to care
Since 2005, 163 rural hospitals have closed in the United States. When rural hospitals close, the situation for hospitalists and other physicians is different. In communities where a larger health system owns a hospital, such as when Vidant Health closed Pungo District Hospital in Belhaven, N.C., in 2014 before reopening a nonemergency clinic in the area in 2016, health care services for the community may have limited interruption.
However, if there isn’t a nearby system to join, many doctors will end up leaving the area. More than half of rural hospitals that close end up not providing any kind of supplementary health care service, according to the NC Rural Health Research Program.
“A lot of the hospitals that have closed have not been owned by a system,” said George H. Pink, PhD, deputy director of the NC Rural Health Research Program at the University of North Carolina at Chapel Hill. “They’ve been independent, freestanding, and that perhaps is one of the reasons why they’re closing, is because they haven’t been able to find a system that would buy them out and inject capital into the community.”
This can also have an effect on the number of health care providers in the area, Dr. Pink said. “Their ability to refer patients and treat patients locally may be affected. That’s why, in many towns where hospitals have closed, we see a drop in the number of providers, particularly primary care doctors who actually live in the community.”
Politicians and federal entities have proposed a number of solutions to help protect rural hospitals from closure. Sen. Charles Grassley (R-Iowa), Sen. Amy Klobuchar (D-Minn.), and Sen. Cory Gardener (R-Colo.) have sponsored bills in the Senate, while Rep. Sam Graves (R-Mo.) has introduced legislation in the House. The Medicare Payment Advisory Commission has proposed two models of rural hospital care, and there are additional models proposed by the Kansas Hospital Association. A pilot program in Pennsylvania, the Pennsylvania Rural Health Model, is testing how a global budget by CMS for all inpatient and hospital-based outcomes might help rural hospitals.
“What we haven’t had a lot of action on is actually testing these models out and seeing whether they will work, and in what kinds of communities they will work,” Dr. Pink said.
Hospitalists as community advocates
Dr. D’Mello, who wrote an article for the Journal of Hospital Medicine on Hahnemann’s ownership by a private equity firm (doi: 10.12788/jhm.3378), said that the inherent nature of a for-profit entity trying to make a hospital profitable is a bad sign for a hospital and not necessarily what is in the best interest for an academic institution or for doctors who train there.
“I don’t know if I could blame the private equity firm completely, but in retrospect, the private equity firms stepping in was like the death knell of the hospital,” he said of Hahnemann’s closure.
“I think what the community needs to know – what the health care community, patient community, the hospitalist community need to know – is that there’s got to be more attention paid to these types of issues during mergers and acquisitions to prevent this from happening,” Dr. Pinsky said.
One larger issue was Hahnemann’s position as a safety net hospital, which partly played into American Academic’s lack of success in making the hospital as profitable as they wanted it to be, Dr. D’Mello noted. Hahnemann’s patient population consisted mostly of minority patients on Medicare, Medicaid, and charity care insurance, while recent studies have shown that hospitals are more likely to succeed when they have a larger proportion of patients with private insurance.
“Studies show that, to [make more] money from private insurance, you really have to have this huge footprint, because then you’ve got a better ability to negotiate with these private insurance companies,” Dr. D’Mello said. “Whether that’s actually good for health care is a different issue.”
Despite their own situations, it is not unusual for hospitalists and hospital physicians to step up during a hospital closure and advocate for their patients on behalf of the community, Dr. Pink said.
“When hospitals are in financial difficulty and there’s the risk of closure, typically, the medical staff are among the first to step up and warn the community: ‘We’re at risk of losing our service. We need some help,’ ” he said. “Generally speaking, the local physicians have been at the forefront of helping to keep access to hospital care available in some of these small communities – unfortunately, not always successfully.”
Dr. D’Mello, Dr. Pinsky, and Dr. Pink report no relevant conflicts of interest.
D.C.-area blacks face increased risk of mortality from SJS/TEN
(TEN), compared with nonblack patients, results from a single-center study showed.

Adam Swigost, MD, presented data on behalf of the study’s principal investigator, Helena B. Pasieka, MD, and associates at MedStar Health Georgetown University in Washington in a video presentation during a virtual meeting held by the George Washington University department of dermatology. The virtual meeting included presentations slated for the annual meeting of the American Academy of Dermatology, which was canceled because of the COVID-19 pandemic.
According to the 2009-2012 Nationwide Inpatient Survey, there were 12,195 cases of SJS, 2,373 cases of SJS/TEN overlap, and 2,675 cases of TEN. In 2016, researchers led by Derek Y. Hsu, MD, of Northwestern University, Chicago, found that SJS/TEN was associated with nonwhite race, particularly Asians (odds ratio, 3.27) and blacks (OR, 2.01) (J Invest Dermatol. 2016;136[7]:1387-97).
“This led Dr. Pasieka and our team to ask the question: Are there differences in SJS/TEN outcomes in self-reported blacks in the U.S.?” said Dr. Swigost, a resident in the department of dermatology at MedStar Health Georgetown University.
To find out, he and his colleagues retrospectively analyzed records from 74 patients with SJS/TEN who were treated at Washington Hospital Center in Washington, D.C., from 2009 to 2019. They drew data from clinical diagnoses with histopathologic evaluation, when available, and performed a multivariate analysis adjusted for age, HIV status, black race, and offending drug category.
Of the 75 patients, 43 were female, 45 were black, 16 were white, 6 were Asian, 5 were Indian, 1 was Native American, and 1 was South Asian. Multivariate analysis revealed that black race was the only significant variable associated with an elevated risk of mortality from SJS/TEN (OR, 4.81; P = .04).
Of the 45 black patients in the study, 33 were HIV negative and 12 were HIV positive. “While this variable was not statistically significant, it did seem to have an elevated risk for mortality in HIV-positive patients [4 of 12; 33%], compared with 8 of 33 HIV-negative patients [25%],” Dr. Swigost said.
Next, the researchers investigated the culprit medications in the black patients. As a reference, they compared their data with a 2015 study that set out to document the clinical profile, etiologies, and outcomes of SJS and TEN in hospitals in four sub-Saharan African countries (Int J Dermatol. 2013 May;52[5]:575-9). In the 2015 study, sulfonamides were the most-used drugs (38%) followed by the antiretroviral drug nevirapine (20%) and tuberculosis drugs (6%). In the study by Dr. Swigost and colleagues, the most frequently implicated drugs were sulfonamides (24%), followed by other antibiotics (24%), and anticonvulsants (17%).
“Our patients at MedStar Washington Hospital Center are going to have different comorbidities and medical problems that dictate different medications being used in different proportions,” Dr. Swigost explained.
Delayed detection is one possible reason for the increased mortality observed in black patients. “Dermatology education on a national level is biased most commonly toward white skin,” he said. “Often, diseases can be missed in skin of color. It’s possible that the diagnoses are being delayed and so treatment is being delayed.”
Socioeconomics and access to health care could also play a role in the poor outcome we observed. “Those are variables we want to further analyze in this data,” Dr. Swigost said. “Other things to consider are genetic variations between African and American black patient populations, because in the U.S. our black population is likely more heterogeneous than African patient populations are. It’s possible that there are HLA [human leukocyte antigen] differences that are contributing. Lastly, further characterization and stratification of SJS/TEN risk factors are required.”
Dr. Swigost and Dr. Pasieka reported having no disclosures.
(TEN), compared with nonblack patients, results from a single-center study showed.

Adam Swigost, MD, presented data on behalf of the study’s principal investigator, Helena B. Pasieka, MD, and associates at MedStar Health Georgetown University in Washington in a video presentation during a virtual meeting held by the George Washington University department of dermatology. The virtual meeting included presentations slated for the annual meeting of the American Academy of Dermatology, which was canceled because of the COVID-19 pandemic.
According to the 2009-2012 Nationwide Inpatient Survey, there were 12,195 cases of SJS, 2,373 cases of SJS/TEN overlap, and 2,675 cases of TEN. In 2016, researchers led by Derek Y. Hsu, MD, of Northwestern University, Chicago, found that SJS/TEN was associated with nonwhite race, particularly Asians (odds ratio, 3.27) and blacks (OR, 2.01) (J Invest Dermatol. 2016;136[7]:1387-97).
“This led Dr. Pasieka and our team to ask the question: Are there differences in SJS/TEN outcomes in self-reported blacks in the U.S.?” said Dr. Swigost, a resident in the department of dermatology at MedStar Health Georgetown University.
To find out, he and his colleagues retrospectively analyzed records from 74 patients with SJS/TEN who were treated at Washington Hospital Center in Washington, D.C., from 2009 to 2019. They drew data from clinical diagnoses with histopathologic evaluation, when available, and performed a multivariate analysis adjusted for age, HIV status, black race, and offending drug category.
Of the 75 patients, 43 were female, 45 were black, 16 were white, 6 were Asian, 5 were Indian, 1 was Native American, and 1 was South Asian. Multivariate analysis revealed that black race was the only significant variable associated with an elevated risk of mortality from SJS/TEN (OR, 4.81; P = .04).
Of the 45 black patients in the study, 33 were HIV negative and 12 were HIV positive. “While this variable was not statistically significant, it did seem to have an elevated risk for mortality in HIV-positive patients [4 of 12; 33%], compared with 8 of 33 HIV-negative patients [25%],” Dr. Swigost said.
Next, the researchers investigated the culprit medications in the black patients. As a reference, they compared their data with a 2015 study that set out to document the clinical profile, etiologies, and outcomes of SJS and TEN in hospitals in four sub-Saharan African countries (Int J Dermatol. 2013 May;52[5]:575-9). In the 2015 study, sulfonamides were the most-used drugs (38%) followed by the antiretroviral drug nevirapine (20%) and tuberculosis drugs (6%). In the study by Dr. Swigost and colleagues, the most frequently implicated drugs were sulfonamides (24%), followed by other antibiotics (24%), and anticonvulsants (17%).
“Our patients at MedStar Washington Hospital Center are going to have different comorbidities and medical problems that dictate different medications being used in different proportions,” Dr. Swigost explained.
Delayed detection is one possible reason for the increased mortality observed in black patients. “Dermatology education on a national level is biased most commonly toward white skin,” he said. “Often, diseases can be missed in skin of color. It’s possible that the diagnoses are being delayed and so treatment is being delayed.”
Socioeconomics and access to health care could also play a role in the poor outcome we observed. “Those are variables we want to further analyze in this data,” Dr. Swigost said. “Other things to consider are genetic variations between African and American black patient populations, because in the U.S. our black population is likely more heterogeneous than African patient populations are. It’s possible that there are HLA [human leukocyte antigen] differences that are contributing. Lastly, further characterization and stratification of SJS/TEN risk factors are required.”
Dr. Swigost and Dr. Pasieka reported having no disclosures.
(TEN), compared with nonblack patients, results from a single-center study showed.

Adam Swigost, MD, presented data on behalf of the study’s principal investigator, Helena B. Pasieka, MD, and associates at MedStar Health Georgetown University in Washington in a video presentation during a virtual meeting held by the George Washington University department of dermatology. The virtual meeting included presentations slated for the annual meeting of the American Academy of Dermatology, which was canceled because of the COVID-19 pandemic.
According to the 2009-2012 Nationwide Inpatient Survey, there were 12,195 cases of SJS, 2,373 cases of SJS/TEN overlap, and 2,675 cases of TEN. In 2016, researchers led by Derek Y. Hsu, MD, of Northwestern University, Chicago, found that SJS/TEN was associated with nonwhite race, particularly Asians (odds ratio, 3.27) and blacks (OR, 2.01) (J Invest Dermatol. 2016;136[7]:1387-97).
“This led Dr. Pasieka and our team to ask the question: Are there differences in SJS/TEN outcomes in self-reported blacks in the U.S.?” said Dr. Swigost, a resident in the department of dermatology at MedStar Health Georgetown University.
To find out, he and his colleagues retrospectively analyzed records from 74 patients with SJS/TEN who were treated at Washington Hospital Center in Washington, D.C., from 2009 to 2019. They drew data from clinical diagnoses with histopathologic evaluation, when available, and performed a multivariate analysis adjusted for age, HIV status, black race, and offending drug category.
Of the 75 patients, 43 were female, 45 were black, 16 were white, 6 were Asian, 5 were Indian, 1 was Native American, and 1 was South Asian. Multivariate analysis revealed that black race was the only significant variable associated with an elevated risk of mortality from SJS/TEN (OR, 4.81; P = .04).
Of the 45 black patients in the study, 33 were HIV negative and 12 were HIV positive. “While this variable was not statistically significant, it did seem to have an elevated risk for mortality in HIV-positive patients [4 of 12; 33%], compared with 8 of 33 HIV-negative patients [25%],” Dr. Swigost said.
Next, the researchers investigated the culprit medications in the black patients. As a reference, they compared their data with a 2015 study that set out to document the clinical profile, etiologies, and outcomes of SJS and TEN in hospitals in four sub-Saharan African countries (Int J Dermatol. 2013 May;52[5]:575-9). In the 2015 study, sulfonamides were the most-used drugs (38%) followed by the antiretroviral drug nevirapine (20%) and tuberculosis drugs (6%). In the study by Dr. Swigost and colleagues, the most frequently implicated drugs were sulfonamides (24%), followed by other antibiotics (24%), and anticonvulsants (17%).
“Our patients at MedStar Washington Hospital Center are going to have different comorbidities and medical problems that dictate different medications being used in different proportions,” Dr. Swigost explained.
Delayed detection is one possible reason for the increased mortality observed in black patients. “Dermatology education on a national level is biased most commonly toward white skin,” he said. “Often, diseases can be missed in skin of color. It’s possible that the diagnoses are being delayed and so treatment is being delayed.”
Socioeconomics and access to health care could also play a role in the poor outcome we observed. “Those are variables we want to further analyze in this data,” Dr. Swigost said. “Other things to consider are genetic variations between African and American black patient populations, because in the U.S. our black population is likely more heterogeneous than African patient populations are. It’s possible that there are HLA [human leukocyte antigen] differences that are contributing. Lastly, further characterization and stratification of SJS/TEN risk factors are required.”
Dr. Swigost and Dr. Pasieka reported having no disclosures.
Ergonomics 101 for trainees
To the early trainee, often the goal of performing a colonoscopy is to reach the cecum using whatever technique necessary. Although the recommended amount of colonoscopies for safe independent practice is 140 (with some sources stating more than 500), this only relates to the safety of the patient.1 We receive scant education on how to form good procedural habits to preserve our own safety and efficiency over the course of our career. Here are some tips on how to prevent injury:
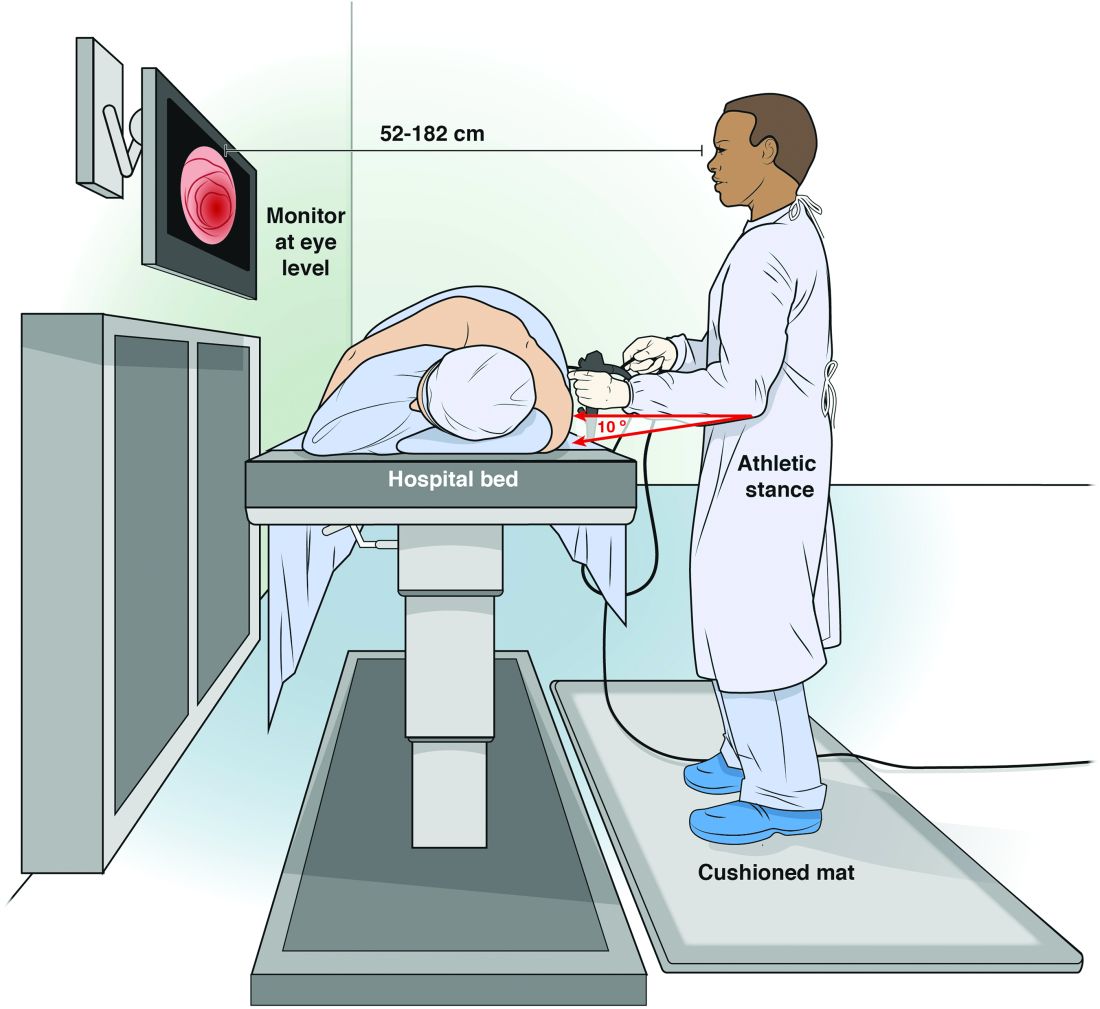
Maintain an appropriate stance. The optimal stance during endoscopy is an athletic stance: chest out, shoulders back to facilitate ease of neck movements, and a slight bend in the knees to facilitate good blood return and distribute weight. Feet should be hip width apart with toes pointed at the endoscopy screen to allow for easy pivoting of the hips and torque of upper body if needed. Ideally, this stance is complemented by the use of proper footwear and a cushioned mat to facilitate weight distribution while standing. An athletic stance facilitates a fluidity for movements from head to toe and an ability to use larger muscles groups to accomplish fine movements.
Handle the endoscope properly. Preserve energy by understanding your equipment and how to manipulate it. Orienting the endoscope directly in front of the endoscopist for upper endoscopy, and at a 45-degree angle for colonoscopy, places the instrument at optimal location to complete the procedure.5 Reviewing how to perform common techniques such as retroflexion, scope reduction, and instrumentation can also facilitate improved ergonomics and adjustment of incorrect techniques at an early stage of endoscopic training. An area of particular concern for most early trainees is the amount of rotational force placed on the right wrist with administration of torque to the endoscope. This is a foreign movement for most endoscopists and requires use of smaller muscle groups of the forearms. We suggest attempting torque with internal and external rotation of the left shoulder to utilize larger muscle groups. We can also combat fatigue during the procedure with the use of microrests intermittently to reduce prolonged muscle contraction. A common way to utilize microrests is by pinning the scope to the patient’s bed with the endoscopist’s hip to provide stability of endoscope and allow removal and relaxation of the right hand. This can be done periodically throughout the procedure to provide the ability to regroup mentally and physically.
Seek feedback. Because it is difficult to focus on ergonomics while performing a diagnostic procedure, utilize your team of observers to facilitate proper form during procedure. This includes your attending gastroenterologists, nurses, and technicians who can observe posture and technique to help detect incorrect positioning early and make corrections. A common practice is to discuss areas of desired improvement before procedures to facilitate a more vigilant observation of areas for improvement.
Assess and adjust often. As early trainees, these endoscopists perform all endoscopies under the direct supervision and often with significant assistance from a supervising gastroenterologist. This can lead to a sharp differential in psychological size; it can be hard to adjust a room to your needs when you have an intimidating and demanding attending physician who has different needs. Despite this disparity, we strongly encourage all trainees to be vigilant about adjusting the room (monitors and beds) to their own needs rather than their attendings’. A great way to head off potential conflict is to discuss the ergonomic positioning of the room before you start endoscopy with your attending, nurse, and technicians so that everyone is in agreement.
Conclusion
We offer this article as a guide for the novice endoscopist to make small changes early to prevent injuries later. Reaching competency with our skills is difficult, and we hope it can be achieved safely with our health in mind.
Dr. Magee, first-year fellow, NCC Gastroenterology; Dr. Singla, associate program director, NCC Gastroenterology, and gastroenterology service, department of internal medicine, Walter Reed National Military Medical Center, Bethesda, Md.
References
1. Spier B et al. Colonoscopy training in gastroenterology fellowships: determining competence. Gastrointest Endosc. 2010 Feb;71(2):319-24G.
2. Malmström EM et al. A slouched body posture decreases arm mobility and changes muscle recruitment in the neck and shoulder region. Eur J Appl Physiol. 2015;115(12):2491-503.
3. Singla M et al. Training the endo-athlete: an update in ergonomics in endoscopy. Clin Gastroenterol Hepatol. 2018 Jul;16(7):1003-6.
4. Bexander CS, et al. Effect of gaze direction on neck muscle activity during cervical rotation. Exp Brain Res. 2005 Dec;167(3):422-32.
5. Soetikno R et al. Holding and manipulating the endoscope: A user’s guide. Techn Gastrointest Endosc. 2019;21:124-32.
To the early trainee, often the goal of performing a colonoscopy is to reach the cecum using whatever technique necessary. Although the recommended amount of colonoscopies for safe independent practice is 140 (with some sources stating more than 500), this only relates to the safety of the patient.1 We receive scant education on how to form good procedural habits to preserve our own safety and efficiency over the course of our career. Here are some tips on how to prevent injury:

Maintain an appropriate stance. The optimal stance during endoscopy is an athletic stance: chest out, shoulders back to facilitate ease of neck movements, and a slight bend in the knees to facilitate good blood return and distribute weight. Feet should be hip width apart with toes pointed at the endoscopy screen to allow for easy pivoting of the hips and torque of upper body if needed. Ideally, this stance is complemented by the use of proper footwear and a cushioned mat to facilitate weight distribution while standing. An athletic stance facilitates a fluidity for movements from head to toe and an ability to use larger muscles groups to accomplish fine movements.
Handle the endoscope properly. Preserve energy by understanding your equipment and how to manipulate it. Orienting the endoscope directly in front of the endoscopist for upper endoscopy, and at a 45-degree angle for colonoscopy, places the instrument at optimal location to complete the procedure.5 Reviewing how to perform common techniques such as retroflexion, scope reduction, and instrumentation can also facilitate improved ergonomics and adjustment of incorrect techniques at an early stage of endoscopic training. An area of particular concern for most early trainees is the amount of rotational force placed on the right wrist with administration of torque to the endoscope. This is a foreign movement for most endoscopists and requires use of smaller muscle groups of the forearms. We suggest attempting torque with internal and external rotation of the left shoulder to utilize larger muscle groups. We can also combat fatigue during the procedure with the use of microrests intermittently to reduce prolonged muscle contraction. A common way to utilize microrests is by pinning the scope to the patient’s bed with the endoscopist’s hip to provide stability of endoscope and allow removal and relaxation of the right hand. This can be done periodically throughout the procedure to provide the ability to regroup mentally and physically.
Seek feedback. Because it is difficult to focus on ergonomics while performing a diagnostic procedure, utilize your team of observers to facilitate proper form during procedure. This includes your attending gastroenterologists, nurses, and technicians who can observe posture and technique to help detect incorrect positioning early and make corrections. A common practice is to discuss areas of desired improvement before procedures to facilitate a more vigilant observation of areas for improvement.
Assess and adjust often. As early trainees, these endoscopists perform all endoscopies under the direct supervision and often with significant assistance from a supervising gastroenterologist. This can lead to a sharp differential in psychological size; it can be hard to adjust a room to your needs when you have an intimidating and demanding attending physician who has different needs. Despite this disparity, we strongly encourage all trainees to be vigilant about adjusting the room (monitors and beds) to their own needs rather than their attendings’. A great way to head off potential conflict is to discuss the ergonomic positioning of the room before you start endoscopy with your attending, nurse, and technicians so that everyone is in agreement.
Conclusion
We offer this article as a guide for the novice endoscopist to make small changes early to prevent injuries later. Reaching competency with our skills is difficult, and we hope it can be achieved safely with our health in mind.
Dr. Magee, first-year fellow, NCC Gastroenterology; Dr. Singla, associate program director, NCC Gastroenterology, and gastroenterology service, department of internal medicine, Walter Reed National Military Medical Center, Bethesda, Md.
References
1. Spier B et al. Colonoscopy training in gastroenterology fellowships: determining competence. Gastrointest Endosc. 2010 Feb;71(2):319-24G.
2. Malmström EM et al. A slouched body posture decreases arm mobility and changes muscle recruitment in the neck and shoulder region. Eur J Appl Physiol. 2015;115(12):2491-503.
3. Singla M et al. Training the endo-athlete: an update in ergonomics in endoscopy. Clin Gastroenterol Hepatol. 2018 Jul;16(7):1003-6.
4. Bexander CS, et al. Effect of gaze direction on neck muscle activity during cervical rotation. Exp Brain Res. 2005 Dec;167(3):422-32.
5. Soetikno R et al. Holding and manipulating the endoscope: A user’s guide. Techn Gastrointest Endosc. 2019;21:124-32.
To the early trainee, often the goal of performing a colonoscopy is to reach the cecum using whatever technique necessary. Although the recommended amount of colonoscopies for safe independent practice is 140 (with some sources stating more than 500), this only relates to the safety of the patient.1 We receive scant education on how to form good procedural habits to preserve our own safety and efficiency over the course of our career. Here are some tips on how to prevent injury:

Maintain an appropriate stance. The optimal stance during endoscopy is an athletic stance: chest out, shoulders back to facilitate ease of neck movements, and a slight bend in the knees to facilitate good blood return and distribute weight. Feet should be hip width apart with toes pointed at the endoscopy screen to allow for easy pivoting of the hips and torque of upper body if needed. Ideally, this stance is complemented by the use of proper footwear and a cushioned mat to facilitate weight distribution while standing. An athletic stance facilitates a fluidity for movements from head to toe and an ability to use larger muscles groups to accomplish fine movements.
Handle the endoscope properly. Preserve energy by understanding your equipment and how to manipulate it. Orienting the endoscope directly in front of the endoscopist for upper endoscopy, and at a 45-degree angle for colonoscopy, places the instrument at optimal location to complete the procedure.5 Reviewing how to perform common techniques such as retroflexion, scope reduction, and instrumentation can also facilitate improved ergonomics and adjustment of incorrect techniques at an early stage of endoscopic training. An area of particular concern for most early trainees is the amount of rotational force placed on the right wrist with administration of torque to the endoscope. This is a foreign movement for most endoscopists and requires use of smaller muscle groups of the forearms. We suggest attempting torque with internal and external rotation of the left shoulder to utilize larger muscle groups. We can also combat fatigue during the procedure with the use of microrests intermittently to reduce prolonged muscle contraction. A common way to utilize microrests is by pinning the scope to the patient’s bed with the endoscopist’s hip to provide stability of endoscope and allow removal and relaxation of the right hand. This can be done periodically throughout the procedure to provide the ability to regroup mentally and physically.
Seek feedback. Because it is difficult to focus on ergonomics while performing a diagnostic procedure, utilize your team of observers to facilitate proper form during procedure. This includes your attending gastroenterologists, nurses, and technicians who can observe posture and technique to help detect incorrect positioning early and make corrections. A common practice is to discuss areas of desired improvement before procedures to facilitate a more vigilant observation of areas for improvement.
Assess and adjust often. As early trainees, these endoscopists perform all endoscopies under the direct supervision and often with significant assistance from a supervising gastroenterologist. This can lead to a sharp differential in psychological size; it can be hard to adjust a room to your needs when you have an intimidating and demanding attending physician who has different needs. Despite this disparity, we strongly encourage all trainees to be vigilant about adjusting the room (monitors and beds) to their own needs rather than their attendings’. A great way to head off potential conflict is to discuss the ergonomic positioning of the room before you start endoscopy with your attending, nurse, and technicians so that everyone is in agreement.
Conclusion
We offer this article as a guide for the novice endoscopist to make small changes early to prevent injuries later. Reaching competency with our skills is difficult, and we hope it can be achieved safely with our health in mind.
Dr. Magee, first-year fellow, NCC Gastroenterology; Dr. Singla, associate program director, NCC Gastroenterology, and gastroenterology service, department of internal medicine, Walter Reed National Military Medical Center, Bethesda, Md.
References
1. Spier B et al. Colonoscopy training in gastroenterology fellowships: determining competence. Gastrointest Endosc. 2010 Feb;71(2):319-24G.
2. Malmström EM et al. A slouched body posture decreases arm mobility and changes muscle recruitment in the neck and shoulder region. Eur J Appl Physiol. 2015;115(12):2491-503.
3. Singla M et al. Training the endo-athlete: an update in ergonomics in endoscopy. Clin Gastroenterol Hepatol. 2018 Jul;16(7):1003-6.
4. Bexander CS, et al. Effect of gaze direction on neck muscle activity during cervical rotation. Exp Brain Res. 2005 Dec;167(3):422-32.
5. Soetikno R et al. Holding and manipulating the endoscope: A user’s guide. Techn Gastrointest Endosc. 2019;21:124-32.
JAK inhibitors may increase risk of herpes zoster
For patients with inflammatory bowel disease or other immune-mediated inflammatory diseases, Janus kinase (JAK) inhibitors appear generally safe, though they may increase the risk of herpes zoster infection, according to a large-scale systematic review and meta-analysis.
Data from more than 66,000 patients revealed no significant links between JAK inhibitors and risks of serious infections, malignancy, or major adverse cardiovascular events, reported lead author Pablo Olivera, MD, of Centro de Educación Médica e Investigación Clínica (CEMIC) in Buenos Aires and colleagues.
“To the best of our knowledge, this is the first systematic review evaluating the risk profile of JAK inhibitors in a wide spectrum of immune-mediated inflammatory diseases,” they wrote in Gastroenterology.
The investigators drew studies from the Cochrane Central Register of Controlled Trials, MEDLINE, and EMBASE from 1990 to 2019 and from conference databases from 2012 to 2018. Out of 973 studies identified, 82 were included in the final analysis, of which two-thirds were randomized clinical trials. In total, 101,925 subjects were included, of whom a majority had rheumatoid arthritis (n = 86,308), followed by psoriasis (n = 9,311), inflammatory bowel disease (n = 5,987), and ankylosing spondylitis (n = 319).
Meta-analysis of JAK inhibitor usage involved 66,159 patients. Four JAK inhibitors were included: tofacitinib, filgotinib, baricitinib, and upadacitinib. The primary outcomes were the incidence rates of adverse events and serious adverse events. The investigators also estimated incidence rates of herpes zoster infection, serious infections, mortality, malignancy, and major adverse cardiovascular events. These rates were compared with those of patients who received placebo or an active comparator in clinical trials.
Analysis showed that almost 9 out of 10 patients (87.16%) who were exposed to a JAK inhibitor received tofacitinib. The investigators described high variability in treatment duration and baseline characteristics of participants. Rates of adverse events and serious adverse events also fell across a broad spectrum, from 10% to 82% and from 0% to 29%, respectively.
“Most [adverse events] were mild, and included worsening of the underlying condition, probably showing lack of efficacy,” the investigators wrote.
Rates of mortality and most adverse events were not significantly associated with JAK inhibitor exposure. In contrast, relative risk of herpes zoster infection was 57% higher in patients who received a JAK inhibitor than in those who received a placebo or comparator (RR, 1.57; 95% confidence interval, 1.01-2.37).
“Regarding the risk of herpes zoster with JAK inhibitors, the largest evidence comes from the use of tofacitinib, but it appears to be a class effect, with a clear dose-dependent effect,” the investigators wrote.
Although risks of herpes zoster may be carried across the drug class, they may not be evenly distributed given that a subgroup analysis revealed that some JAK inhibitors may bring higher risks than others; specifically, tofacitinib and baricitinib were associated with higher relative risks of herpes zoster than were upadacitinib and filgotinib.
“Although this is merely a qualitative comparison, this difference could be related to the fact that both filgotinib and upadacitinib are selective JAK1 inhibitors, whereas tofacitinib is a JAK1/JAK3 inhibitor and baricitinib a JAK1/JAK2 inhibitor,” the investigators wrote. “Further studies are needed to determine if JAK isoform selectivity affects the risk of herpes zoster.”
The investigators emphasized this need for more research. While the present findings help illuminate the safety profile of JAK inhibitors, they are clouded by various other factors, such as disease-specific considerations, a lack of real-world data, and studies that are likely too short to accurately determine risk of malignancy, the investigators wrote.
“More studies with long follow-up and in the real world setting, in different conditions, will be needed to fully elucidate the safety profile of the different JAK inhibitors,” the investigators concluded.
The investigators disclosed relationships with AbbVie, Takeda, Pfizer, and others.
SOURCE: Olivera P et al. Gastroenterology. 2020 Jan 8. doi: 10.1053/j.gastro.2020.01.001.
The multiple different cytokines contributing to intestinal inflammation in IBD patients have been a major challenge in the design of therapies. Because the JAK signaling pathway (comprised of JAK1, JAK2, JAK3, and TYK2) is required for responses to a broad range of cytokines, therapies that inhibit JAK signaling have been an active area of interest. A simultaneous and important concern, however, has been the potential for adverse consequences when inhibiting the breadth of immune and hematopoietic molecules that depend on JAK family members for their functions. This meta-analysis by Olivera et al. examined adverse outcomes of four different JAK inhibitors in clinical trials across four immune-mediated diseases (rheumatoid arthritis, IBD, psoriasis, and ankylosing spondylitis), finding that herpes zoster infection was significantly increased (relative risk, 1.57). In contrast, patients treated with JAK inhibitors were not at a significantly increased risk for various other adverse events.
Reduced dosing of JAK inhibitors has been implemented as a means of improving safety profiles in select immune-mediated diseases. Another approach is more selective JAK inhibition, although it is unclear whether this will eliminate the risk of herpes zoster infection. In the current meta-analysis, about 87% of the studies had evaluated tofacitinib treatment, which inhibits both JAK1 and JAK3; more selective JAK inhibitors could not be evaluated in an equivalent manner. Of note, JAK1 is required for signaling by various cytokines that participate in the response to viruses, including type I IFNs and gamma c family members (such as IL-2 and IL-15); therefore, even the more selective JAK1 inhibitors do not leave this immune function fully intact. However, simply reducing the number of JAK family members inhibited simultaneously may be sufficient to reduce risk.
JAK inhibitors warrant further evaluation as additional infectious challenges arise, particularly with respect to viruses. In addition, more selective targeting of JAK inhibition of intestinal tissues may ultimately reduce systemic effects, including the risk of herpes zoster.
Clara Abraham, MD, professor of medicine, section of digestive diseases, Yale University, New Haven, Conn.
The multiple different cytokines contributing to intestinal inflammation in IBD patients have been a major challenge in the design of therapies. Because the JAK signaling pathway (comprised of JAK1, JAK2, JAK3, and TYK2) is required for responses to a broad range of cytokines, therapies that inhibit JAK signaling have been an active area of interest. A simultaneous and important concern, however, has been the potential for adverse consequences when inhibiting the breadth of immune and hematopoietic molecules that depend on JAK family members for their functions. This meta-analysis by Olivera et al. examined adverse outcomes of four different JAK inhibitors in clinical trials across four immune-mediated diseases (rheumatoid arthritis, IBD, psoriasis, and ankylosing spondylitis), finding that herpes zoster infection was significantly increased (relative risk, 1.57). In contrast, patients treated with JAK inhibitors were not at a significantly increased risk for various other adverse events.
Reduced dosing of JAK inhibitors has been implemented as a means of improving safety profiles in select immune-mediated diseases. Another approach is more selective JAK inhibition, although it is unclear whether this will eliminate the risk of herpes zoster infection. In the current meta-analysis, about 87% of the studies had evaluated tofacitinib treatment, which inhibits both JAK1 and JAK3; more selective JAK inhibitors could not be evaluated in an equivalent manner. Of note, JAK1 is required for signaling by various cytokines that participate in the response to viruses, including type I IFNs and gamma c family members (such as IL-2 and IL-15); therefore, even the more selective JAK1 inhibitors do not leave this immune function fully intact. However, simply reducing the number of JAK family members inhibited simultaneously may be sufficient to reduce risk.
JAK inhibitors warrant further evaluation as additional infectious challenges arise, particularly with respect to viruses. In addition, more selective targeting of JAK inhibition of intestinal tissues may ultimately reduce systemic effects, including the risk of herpes zoster.
Clara Abraham, MD, professor of medicine, section of digestive diseases, Yale University, New Haven, Conn.
The multiple different cytokines contributing to intestinal inflammation in IBD patients have been a major challenge in the design of therapies. Because the JAK signaling pathway (comprised of JAK1, JAK2, JAK3, and TYK2) is required for responses to a broad range of cytokines, therapies that inhibit JAK signaling have been an active area of interest. A simultaneous and important concern, however, has been the potential for adverse consequences when inhibiting the breadth of immune and hematopoietic molecules that depend on JAK family members for their functions. This meta-analysis by Olivera et al. examined adverse outcomes of four different JAK inhibitors in clinical trials across four immune-mediated diseases (rheumatoid arthritis, IBD, psoriasis, and ankylosing spondylitis), finding that herpes zoster infection was significantly increased (relative risk, 1.57). In contrast, patients treated with JAK inhibitors were not at a significantly increased risk for various other adverse events.
Reduced dosing of JAK inhibitors has been implemented as a means of improving safety profiles in select immune-mediated diseases. Another approach is more selective JAK inhibition, although it is unclear whether this will eliminate the risk of herpes zoster infection. In the current meta-analysis, about 87% of the studies had evaluated tofacitinib treatment, which inhibits both JAK1 and JAK3; more selective JAK inhibitors could not be evaluated in an equivalent manner. Of note, JAK1 is required for signaling by various cytokines that participate in the response to viruses, including type I IFNs and gamma c family members (such as IL-2 and IL-15); therefore, even the more selective JAK1 inhibitors do not leave this immune function fully intact. However, simply reducing the number of JAK family members inhibited simultaneously may be sufficient to reduce risk.
JAK inhibitors warrant further evaluation as additional infectious challenges arise, particularly with respect to viruses. In addition, more selective targeting of JAK inhibition of intestinal tissues may ultimately reduce systemic effects, including the risk of herpes zoster.
Clara Abraham, MD, professor of medicine, section of digestive diseases, Yale University, New Haven, Conn.
For patients with inflammatory bowel disease or other immune-mediated inflammatory diseases, Janus kinase (JAK) inhibitors appear generally safe, though they may increase the risk of herpes zoster infection, according to a large-scale systematic review and meta-analysis.
Data from more than 66,000 patients revealed no significant links between JAK inhibitors and risks of serious infections, malignancy, or major adverse cardiovascular events, reported lead author Pablo Olivera, MD, of Centro de Educación Médica e Investigación Clínica (CEMIC) in Buenos Aires and colleagues.
“To the best of our knowledge, this is the first systematic review evaluating the risk profile of JAK inhibitors in a wide spectrum of immune-mediated inflammatory diseases,” they wrote in Gastroenterology.
The investigators drew studies from the Cochrane Central Register of Controlled Trials, MEDLINE, and EMBASE from 1990 to 2019 and from conference databases from 2012 to 2018. Out of 973 studies identified, 82 were included in the final analysis, of which two-thirds were randomized clinical trials. In total, 101,925 subjects were included, of whom a majority had rheumatoid arthritis (n = 86,308), followed by psoriasis (n = 9,311), inflammatory bowel disease (n = 5,987), and ankylosing spondylitis (n = 319).
Meta-analysis of JAK inhibitor usage involved 66,159 patients. Four JAK inhibitors were included: tofacitinib, filgotinib, baricitinib, and upadacitinib. The primary outcomes were the incidence rates of adverse events and serious adverse events. The investigators also estimated incidence rates of herpes zoster infection, serious infections, mortality, malignancy, and major adverse cardiovascular events. These rates were compared with those of patients who received placebo or an active comparator in clinical trials.
Analysis showed that almost 9 out of 10 patients (87.16%) who were exposed to a JAK inhibitor received tofacitinib. The investigators described high variability in treatment duration and baseline characteristics of participants. Rates of adverse events and serious adverse events also fell across a broad spectrum, from 10% to 82% and from 0% to 29%, respectively.
“Most [adverse events] were mild, and included worsening of the underlying condition, probably showing lack of efficacy,” the investigators wrote.
Rates of mortality and most adverse events were not significantly associated with JAK inhibitor exposure. In contrast, relative risk of herpes zoster infection was 57% higher in patients who received a JAK inhibitor than in those who received a placebo or comparator (RR, 1.57; 95% confidence interval, 1.01-2.37).
“Regarding the risk of herpes zoster with JAK inhibitors, the largest evidence comes from the use of tofacitinib, but it appears to be a class effect, with a clear dose-dependent effect,” the investigators wrote.
Although risks of herpes zoster may be carried across the drug class, they may not be evenly distributed given that a subgroup analysis revealed that some JAK inhibitors may bring higher risks than others; specifically, tofacitinib and baricitinib were associated with higher relative risks of herpes zoster than were upadacitinib and filgotinib.
“Although this is merely a qualitative comparison, this difference could be related to the fact that both filgotinib and upadacitinib are selective JAK1 inhibitors, whereas tofacitinib is a JAK1/JAK3 inhibitor and baricitinib a JAK1/JAK2 inhibitor,” the investigators wrote. “Further studies are needed to determine if JAK isoform selectivity affects the risk of herpes zoster.”
The investigators emphasized this need for more research. While the present findings help illuminate the safety profile of JAK inhibitors, they are clouded by various other factors, such as disease-specific considerations, a lack of real-world data, and studies that are likely too short to accurately determine risk of malignancy, the investigators wrote.
“More studies with long follow-up and in the real world setting, in different conditions, will be needed to fully elucidate the safety profile of the different JAK inhibitors,” the investigators concluded.
The investigators disclosed relationships with AbbVie, Takeda, Pfizer, and others.
SOURCE: Olivera P et al. Gastroenterology. 2020 Jan 8. doi: 10.1053/j.gastro.2020.01.001.
For patients with inflammatory bowel disease or other immune-mediated inflammatory diseases, Janus kinase (JAK) inhibitors appear generally safe, though they may increase the risk of herpes zoster infection, according to a large-scale systematic review and meta-analysis.
Data from more than 66,000 patients revealed no significant links between JAK inhibitors and risks of serious infections, malignancy, or major adverse cardiovascular events, reported lead author Pablo Olivera, MD, of Centro de Educación Médica e Investigación Clínica (CEMIC) in Buenos Aires and colleagues.
“To the best of our knowledge, this is the first systematic review evaluating the risk profile of JAK inhibitors in a wide spectrum of immune-mediated inflammatory diseases,” they wrote in Gastroenterology.
The investigators drew studies from the Cochrane Central Register of Controlled Trials, MEDLINE, and EMBASE from 1990 to 2019 and from conference databases from 2012 to 2018. Out of 973 studies identified, 82 were included in the final analysis, of which two-thirds were randomized clinical trials. In total, 101,925 subjects were included, of whom a majority had rheumatoid arthritis (n = 86,308), followed by psoriasis (n = 9,311), inflammatory bowel disease (n = 5,987), and ankylosing spondylitis (n = 319).
Meta-analysis of JAK inhibitor usage involved 66,159 patients. Four JAK inhibitors were included: tofacitinib, filgotinib, baricitinib, and upadacitinib. The primary outcomes were the incidence rates of adverse events and serious adverse events. The investigators also estimated incidence rates of herpes zoster infection, serious infections, mortality, malignancy, and major adverse cardiovascular events. These rates were compared with those of patients who received placebo or an active comparator in clinical trials.
Analysis showed that almost 9 out of 10 patients (87.16%) who were exposed to a JAK inhibitor received tofacitinib. The investigators described high variability in treatment duration and baseline characteristics of participants. Rates of adverse events and serious adverse events also fell across a broad spectrum, from 10% to 82% and from 0% to 29%, respectively.
“Most [adverse events] were mild, and included worsening of the underlying condition, probably showing lack of efficacy,” the investigators wrote.
Rates of mortality and most adverse events were not significantly associated with JAK inhibitor exposure. In contrast, relative risk of herpes zoster infection was 57% higher in patients who received a JAK inhibitor than in those who received a placebo or comparator (RR, 1.57; 95% confidence interval, 1.01-2.37).
“Regarding the risk of herpes zoster with JAK inhibitors, the largest evidence comes from the use of tofacitinib, but it appears to be a class effect, with a clear dose-dependent effect,” the investigators wrote.
Although risks of herpes zoster may be carried across the drug class, they may not be evenly distributed given that a subgroup analysis revealed that some JAK inhibitors may bring higher risks than others; specifically, tofacitinib and baricitinib were associated with higher relative risks of herpes zoster than were upadacitinib and filgotinib.
“Although this is merely a qualitative comparison, this difference could be related to the fact that both filgotinib and upadacitinib are selective JAK1 inhibitors, whereas tofacitinib is a JAK1/JAK3 inhibitor and baricitinib a JAK1/JAK2 inhibitor,” the investigators wrote. “Further studies are needed to determine if JAK isoform selectivity affects the risk of herpes zoster.”
The investigators emphasized this need for more research. While the present findings help illuminate the safety profile of JAK inhibitors, they are clouded by various other factors, such as disease-specific considerations, a lack of real-world data, and studies that are likely too short to accurately determine risk of malignancy, the investigators wrote.
“More studies with long follow-up and in the real world setting, in different conditions, will be needed to fully elucidate the safety profile of the different JAK inhibitors,” the investigators concluded.
The investigators disclosed relationships with AbbVie, Takeda, Pfizer, and others.
SOURCE: Olivera P et al. Gastroenterology. 2020 Jan 8. doi: 10.1053/j.gastro.2020.01.001.
FROM GASTROENTEROLOGY
HM20 canceled: SHM explains why
COVID-19 made holding meeting impossible
In mid-March, the Society of Hospital Medicine board of directors concluded that it was impossible for SHM to move forward with Hospital Medicine 2020 because of the continued spread of virus that causes Coronavirus Disease 2019 (COVID-19).
Given the most recent information available from the Centers for Disease Control & Prevention and the World Health Organization about the evolving global pandemic and the number of institutions that had travel bans in place, SHM leadership concluded that canceling the Annual Conference was the only path forward.
“Canceling the conference during this unprecedented time is the right thing to do,” said Benji K. Mathews, MD, SFHM, CLHM, course director for HM20. “With the evolving circumstances out of our control, there were risks to our community as it would have gathered, communities we connect with on our travels, and our home communities and hospitals – canceling was the best way to mitigate these risks. Through it all, I couldn’t have asked for a better leadership team and the larger SHM community for their support.”
Because hospitalists are on the front lines of patient care at their institutions, they will be needed more than ever as the pandemic continues to grow in order to manage care of hospitalized patients with COVID-19 and other illnesses. As the only medical society dedicated to hospital medicine, SHM will continue to support hospitalists with resources and research specific to COVID-19 and its impact on the practice of hospital medicine.
SHM is aware that this necessary cancellation impacts many from both a financial and logistical perspective. As such, SHM will refund all conference registration fees for HM20 in full. SHM is also providing the opportunity to defer your HM20 registration to HM21, taking place May 4-7, 2021 in Las Vegas, or Pediatric Hospital Medicine 2020, taking place July 23-26, 2020 in Lake Buena Vista, Fla.
For accommodation or travel cancellations, SHM requests that individuals please refer to their respective hotel or carrier’s customer service team and related cancellation policies.
To provide the world-class education that conference attendees have come to expect from SHM over the years, the SHM team is exploring virtual options to offer select content originally anticipated at HM20. SHM also offers online education via the SHM Learning Portal and the new SHM Education app.
Visit shmannualconference.org/faqs for a full list of FAQs. For additional questions, please contact [email protected].
SHM will continue to monitor the COVID-19 pandemic and provide hospitalists with useful resources in this time of need at hospitalmedicine.org/coronavirus. For news coverage of COVID-19, visit https://www.the-hospitalist.org/hospitalist/coronavirus-updates.
COVID-19 made holding meeting impossible
COVID-19 made holding meeting impossible
In mid-March, the Society of Hospital Medicine board of directors concluded that it was impossible for SHM to move forward with Hospital Medicine 2020 because of the continued spread of virus that causes Coronavirus Disease 2019 (COVID-19).
Given the most recent information available from the Centers for Disease Control & Prevention and the World Health Organization about the evolving global pandemic and the number of institutions that had travel bans in place, SHM leadership concluded that canceling the Annual Conference was the only path forward.
“Canceling the conference during this unprecedented time is the right thing to do,” said Benji K. Mathews, MD, SFHM, CLHM, course director for HM20. “With the evolving circumstances out of our control, there were risks to our community as it would have gathered, communities we connect with on our travels, and our home communities and hospitals – canceling was the best way to mitigate these risks. Through it all, I couldn’t have asked for a better leadership team and the larger SHM community for their support.”
Because hospitalists are on the front lines of patient care at their institutions, they will be needed more than ever as the pandemic continues to grow in order to manage care of hospitalized patients with COVID-19 and other illnesses. As the only medical society dedicated to hospital medicine, SHM will continue to support hospitalists with resources and research specific to COVID-19 and its impact on the practice of hospital medicine.
SHM is aware that this necessary cancellation impacts many from both a financial and logistical perspective. As such, SHM will refund all conference registration fees for HM20 in full. SHM is also providing the opportunity to defer your HM20 registration to HM21, taking place May 4-7, 2021 in Las Vegas, or Pediatric Hospital Medicine 2020, taking place July 23-26, 2020 in Lake Buena Vista, Fla.
For accommodation or travel cancellations, SHM requests that individuals please refer to their respective hotel or carrier’s customer service team and related cancellation policies.
To provide the world-class education that conference attendees have come to expect from SHM over the years, the SHM team is exploring virtual options to offer select content originally anticipated at HM20. SHM also offers online education via the SHM Learning Portal and the new SHM Education app.
Visit shmannualconference.org/faqs for a full list of FAQs. For additional questions, please contact [email protected].
SHM will continue to monitor the COVID-19 pandemic and provide hospitalists with useful resources in this time of need at hospitalmedicine.org/coronavirus. For news coverage of COVID-19, visit https://www.the-hospitalist.org/hospitalist/coronavirus-updates.
In mid-March, the Society of Hospital Medicine board of directors concluded that it was impossible for SHM to move forward with Hospital Medicine 2020 because of the continued spread of virus that causes Coronavirus Disease 2019 (COVID-19).
Given the most recent information available from the Centers for Disease Control & Prevention and the World Health Organization about the evolving global pandemic and the number of institutions that had travel bans in place, SHM leadership concluded that canceling the Annual Conference was the only path forward.
“Canceling the conference during this unprecedented time is the right thing to do,” said Benji K. Mathews, MD, SFHM, CLHM, course director for HM20. “With the evolving circumstances out of our control, there were risks to our community as it would have gathered, communities we connect with on our travels, and our home communities and hospitals – canceling was the best way to mitigate these risks. Through it all, I couldn’t have asked for a better leadership team and the larger SHM community for their support.”
Because hospitalists are on the front lines of patient care at their institutions, they will be needed more than ever as the pandemic continues to grow in order to manage care of hospitalized patients with COVID-19 and other illnesses. As the only medical society dedicated to hospital medicine, SHM will continue to support hospitalists with resources and research specific to COVID-19 and its impact on the practice of hospital medicine.
SHM is aware that this necessary cancellation impacts many from both a financial and logistical perspective. As such, SHM will refund all conference registration fees for HM20 in full. SHM is also providing the opportunity to defer your HM20 registration to HM21, taking place May 4-7, 2021 in Las Vegas, or Pediatric Hospital Medicine 2020, taking place July 23-26, 2020 in Lake Buena Vista, Fla.
For accommodation or travel cancellations, SHM requests that individuals please refer to their respective hotel or carrier’s customer service team and related cancellation policies.
To provide the world-class education that conference attendees have come to expect from SHM over the years, the SHM team is exploring virtual options to offer select content originally anticipated at HM20. SHM also offers online education via the SHM Learning Portal and the new SHM Education app.
Visit shmannualconference.org/faqs for a full list of FAQs. For additional questions, please contact [email protected].
SHM will continue to monitor the COVID-19 pandemic and provide hospitalists with useful resources in this time of need at hospitalmedicine.org/coronavirus. For news coverage of COVID-19, visit https://www.the-hospitalist.org/hospitalist/coronavirus-updates.
Translucent Periorbital Papules
The Diagnosis: Apocrine Hidrocystoma
Histopathologic examination of one of the papules revealed cystic cavities located within the dermis (Figure 1) lined by a cuboidal epithelium demonstrating decapitation secretion (Figure 2), confirming the diagnosis of apocrine hidrocystomas. The presence of multiple lesions prompted further examination for an underlying genetic disorder; however, the patient's hair, nails, and teeth were normal. There also was no evidence of palmoplantar keratoderma or blaschkoid dermatosis.
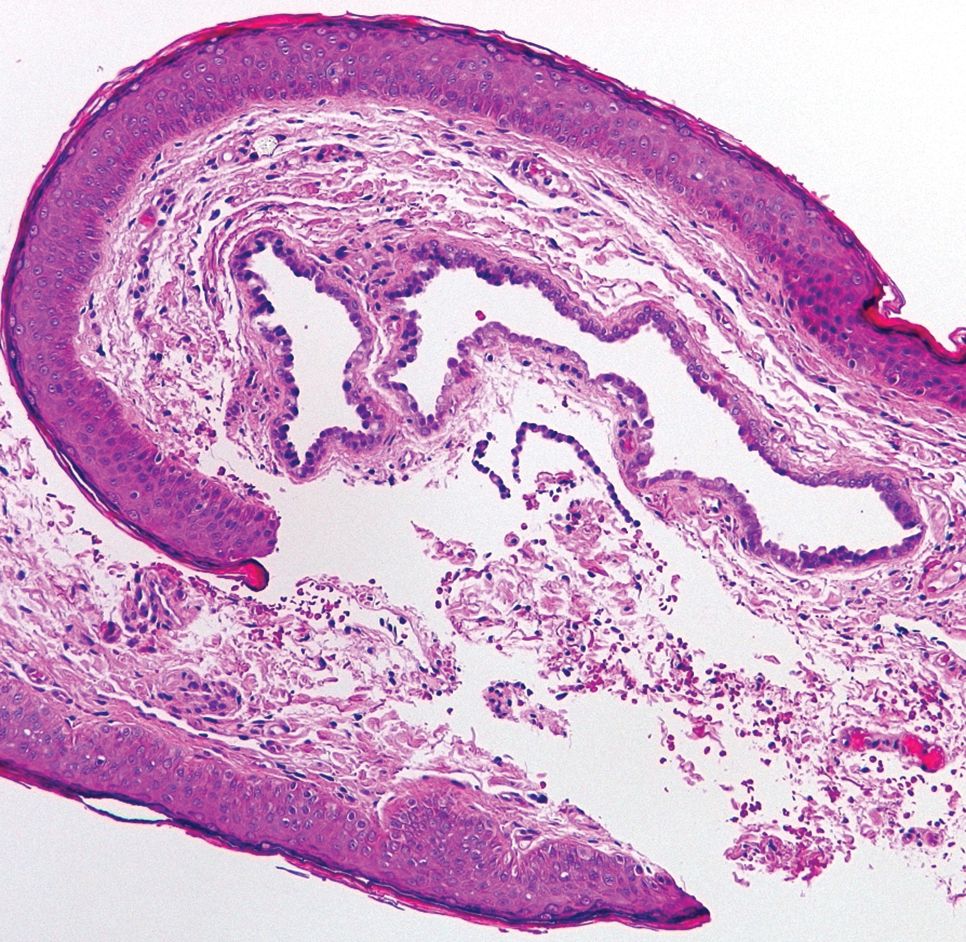
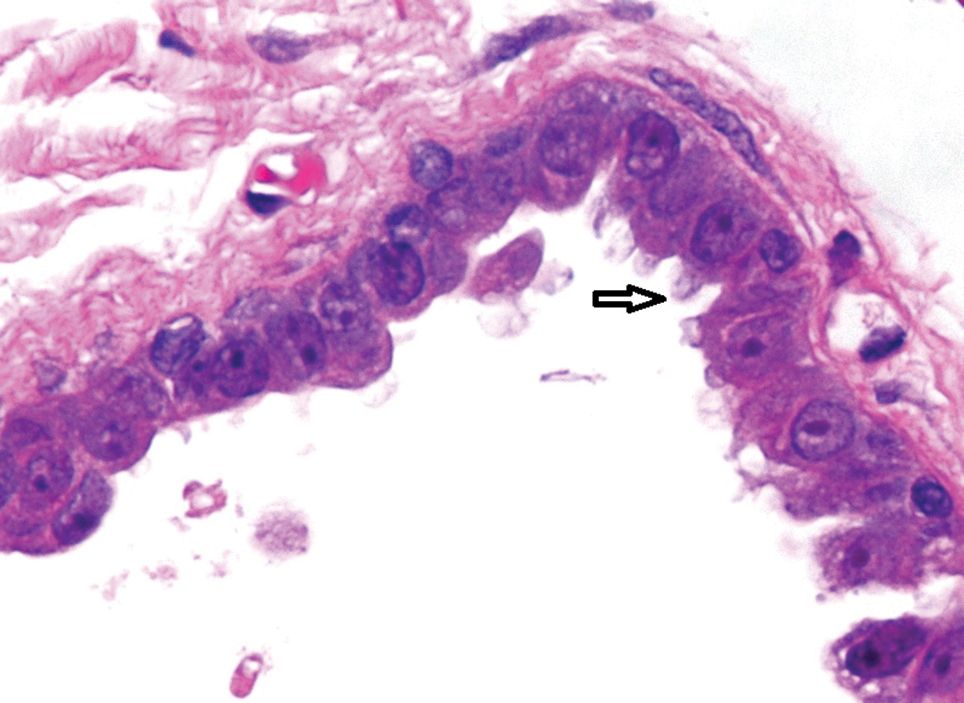
Hidrocystomas are benign cysts of the sudoriferous apparatus that can be subdivided based on histogenesis (apocrine vs eccrine) or lesion count (single vs multiple).1 Multiple lesions may be associated with disorders of ectodermal dysplasia, including Goltz syndrome and Schopf-Schulz-Passarge syndrome. Apocrine hidrocystomas tend to present as solitary, translucent, flesh-colored to bluish facial papules, and the occurrence of multiple lesions is rare in contrast to its eccrine counterpart.2 Various extrafacial sites have been described including the trunk, axillae, umbilicus, genitalia, and digits.3 Apocrine hidrocystomas do not demonstrate aggravation with exposure to heat, unlike their eccrine counterparts.2
A review of 107 patients with 215 histologically proven hidrocystomas demonstrated a preponderance for women in their mid 50s; 74.8% of patients had unilateral disease, and 69.8% of all lesions affected either the lower eyelid or lateral canthus. Recurrence following conventional surgical excision was observed in 2.3% of lesions.1
A review from Japan recounted an incidence of 5 cases per year from 1999 to 2003.4 Patients ranged in age from 30 to 70 years, but there was no gender predilection. Individual apocrine hidrocystomas were mostly less than 2 cm and varied from flesh colored to light red, brown, blue, or purple; 61% of lesions arose periorbitally. Within their cohort, patients with multiple lesions were uncommon, with only 2 cases presenting with 2 lesions simultaneously.4
Apocrine hidrocystomas are thought to result from a cystic proliferation of the secretory component of apocrine sweat glands, though the exact pathogenesis still is unclear.3 Histologic features include a unilocular or multilocular cystic cavity within the dermis lined by columnar cells demonstrating decapitation secretion, followed by a peripheral rim of flattened myoepithelial cells.
Treatment of apocrine hidrocystomas includes topical anticholinergics, surgical excision, electrodesiccation, 1450-nm diode or CO2 lasers, and trichloroacetic acid.2 The novel use of cryotherapy,5 botulinum toxin,2 and intralesional injections of 50% glucose (as a sclerosant)6 also have been reported. Caution should be exercised when managing digital lesions, as digital papillary carcinoma has been described as a clinical and histopathologic mimicker.7
Lipoid proteinosis is a rare autosomal-recessive disorder. Cutaneous lesions manifest in 2 overlapping stages, typically within the first 2 years of life. The first stage consists of vesicles and hemorrhagic crusts on the face and extremities and intraorally, which may heal with scarring. In the second stage, the skin becomes diffusely thickened and waxy, with the appearance of papules, nodules, or plaques along the eyelid margins (moniliform blepharosis), face, axillae, or scrotum. Verrucous lesions also may develop on the knee or elbow extensors.8
Lymphangioma circumscriptum represents microcystic lymphatic malformations that can arise anywhere on the skin or oral mucosa. They present as clusters of clear or hemorrhagic vesicles of variable size and number favoring the proximal extremities and chest. Histologically, dilated lymphatic channels are seen in the upper dermis.8
Syringomas are common benign tumors of the sweat ducts characterized histologically by superficial dermal proliferations of small comma-shaped ducts set in a fibrotic stroma. Clinically, syringomas appear as small, firm, flesh-colored papules with a predilection for the periorbital area. An eruptive onset may be observed, most commonly affecting the trunk. Syringomas may be associated with Down syndrome, while the clear cell variant may be associated with diabetes mellitus.8
Primary systemic amyloidosis may present with a variety of systemic manifestations. Skin involvement can present as waxy, translucent, or purpuric papulonodules or plaques characteristically affecting the periorbital region. Other mucocutaneous signs include macroglossia with or without translucent to hemorrhagic papulovesicles; bruising, especially on the eyelids, neck, axillae, or anogenital area; vesiculobullous skin lesions; or diffuse cutaneous infiltration imparting a sclerodermoid appearance.8
- Maeng M, Petrakos P, Zhou M, et al. Bi-institutional retrospective study on the demographics and basic clinical presentation of hidrocystomas. Orbit. 2017;36:433-435.
- Bordelon JR, Tang N, Elston D, et al. Multiple apocrine hidrocystomas successfully treated with botulinum toxin A. Br J Dermatol. 2017;176:488-490.
- Hafsi W, Badri T. Apocrine hidrocystoma. StatPearls. Treasure Island, FL: StatPearls Publishing; 2017.
- Anzai S, Goto M, Fujiwara S, et al. Apocrine hidrocystoma: a case report and analysis of 167 Japanese cases. Int J Dermatol. 2005;44:702-703.
- Panagiotopoulos A, Vasalou V, Sgontzou T, et al. Multiple apocrine hidrocystomas successfully treated with cryotherapy. Dermatol Surg. 2017;43:993-995.
- Osaki TH, Osaki MH, Osaki T, et al. A minimally invasive approach for apocrine hidrocystomas of the eyelid. Dermatol Surg. 2016;42:134-136.
- Molina-Ruiz AM, Llamas-Velasco M, Rütten A, et al. 'Apocrine hidrocystoma and cystadenoma'-like tumor of the digits or toes: a potential diagnostic pitfall of digital papillary adenocarcinoma. Am J Surg Pathol. 2016;40:410-418.
- Bolognia JL, Jorizzo JL, Schaffer JV, eds. Dermatology. Philadelphia, PA: Elsevier Saunders; 2012.
The Diagnosis: Apocrine Hidrocystoma
Histopathologic examination of one of the papules revealed cystic cavities located within the dermis (Figure 1) lined by a cuboidal epithelium demonstrating decapitation secretion (Figure 2), confirming the diagnosis of apocrine hidrocystomas. The presence of multiple lesions prompted further examination for an underlying genetic disorder; however, the patient's hair, nails, and teeth were normal. There also was no evidence of palmoplantar keratoderma or blaschkoid dermatosis.


Hidrocystomas are benign cysts of the sudoriferous apparatus that can be subdivided based on histogenesis (apocrine vs eccrine) or lesion count (single vs multiple).1 Multiple lesions may be associated with disorders of ectodermal dysplasia, including Goltz syndrome and Schopf-Schulz-Passarge syndrome. Apocrine hidrocystomas tend to present as solitary, translucent, flesh-colored to bluish facial papules, and the occurrence of multiple lesions is rare in contrast to its eccrine counterpart.2 Various extrafacial sites have been described including the trunk, axillae, umbilicus, genitalia, and digits.3 Apocrine hidrocystomas do not demonstrate aggravation with exposure to heat, unlike their eccrine counterparts.2
A review of 107 patients with 215 histologically proven hidrocystomas demonstrated a preponderance for women in their mid 50s; 74.8% of patients had unilateral disease, and 69.8% of all lesions affected either the lower eyelid or lateral canthus. Recurrence following conventional surgical excision was observed in 2.3% of lesions.1
A review from Japan recounted an incidence of 5 cases per year from 1999 to 2003.4 Patients ranged in age from 30 to 70 years, but there was no gender predilection. Individual apocrine hidrocystomas were mostly less than 2 cm and varied from flesh colored to light red, brown, blue, or purple; 61% of lesions arose periorbitally. Within their cohort, patients with multiple lesions were uncommon, with only 2 cases presenting with 2 lesions simultaneously.4
Apocrine hidrocystomas are thought to result from a cystic proliferation of the secretory component of apocrine sweat glands, though the exact pathogenesis still is unclear.3 Histologic features include a unilocular or multilocular cystic cavity within the dermis lined by columnar cells demonstrating decapitation secretion, followed by a peripheral rim of flattened myoepithelial cells.
Treatment of apocrine hidrocystomas includes topical anticholinergics, surgical excision, electrodesiccation, 1450-nm diode or CO2 lasers, and trichloroacetic acid.2 The novel use of cryotherapy,5 botulinum toxin,2 and intralesional injections of 50% glucose (as a sclerosant)6 also have been reported. Caution should be exercised when managing digital lesions, as digital papillary carcinoma has been described as a clinical and histopathologic mimicker.7
Lipoid proteinosis is a rare autosomal-recessive disorder. Cutaneous lesions manifest in 2 overlapping stages, typically within the first 2 years of life. The first stage consists of vesicles and hemorrhagic crusts on the face and extremities and intraorally, which may heal with scarring. In the second stage, the skin becomes diffusely thickened and waxy, with the appearance of papules, nodules, or plaques along the eyelid margins (moniliform blepharosis), face, axillae, or scrotum. Verrucous lesions also may develop on the knee or elbow extensors.8
Lymphangioma circumscriptum represents microcystic lymphatic malformations that can arise anywhere on the skin or oral mucosa. They present as clusters of clear or hemorrhagic vesicles of variable size and number favoring the proximal extremities and chest. Histologically, dilated lymphatic channels are seen in the upper dermis.8
Syringomas are common benign tumors of the sweat ducts characterized histologically by superficial dermal proliferations of small comma-shaped ducts set in a fibrotic stroma. Clinically, syringomas appear as small, firm, flesh-colored papules with a predilection for the periorbital area. An eruptive onset may be observed, most commonly affecting the trunk. Syringomas may be associated with Down syndrome, while the clear cell variant may be associated with diabetes mellitus.8
Primary systemic amyloidosis may present with a variety of systemic manifestations. Skin involvement can present as waxy, translucent, or purpuric papulonodules or plaques characteristically affecting the periorbital region. Other mucocutaneous signs include macroglossia with or without translucent to hemorrhagic papulovesicles; bruising, especially on the eyelids, neck, axillae, or anogenital area; vesiculobullous skin lesions; or diffuse cutaneous infiltration imparting a sclerodermoid appearance.8
The Diagnosis: Apocrine Hidrocystoma
Histopathologic examination of one of the papules revealed cystic cavities located within the dermis (Figure 1) lined by a cuboidal epithelium demonstrating decapitation secretion (Figure 2), confirming the diagnosis of apocrine hidrocystomas. The presence of multiple lesions prompted further examination for an underlying genetic disorder; however, the patient's hair, nails, and teeth were normal. There also was no evidence of palmoplantar keratoderma or blaschkoid dermatosis.


Hidrocystomas are benign cysts of the sudoriferous apparatus that can be subdivided based on histogenesis (apocrine vs eccrine) or lesion count (single vs multiple).1 Multiple lesions may be associated with disorders of ectodermal dysplasia, including Goltz syndrome and Schopf-Schulz-Passarge syndrome. Apocrine hidrocystomas tend to present as solitary, translucent, flesh-colored to bluish facial papules, and the occurrence of multiple lesions is rare in contrast to its eccrine counterpart.2 Various extrafacial sites have been described including the trunk, axillae, umbilicus, genitalia, and digits.3 Apocrine hidrocystomas do not demonstrate aggravation with exposure to heat, unlike their eccrine counterparts.2
A review of 107 patients with 215 histologically proven hidrocystomas demonstrated a preponderance for women in their mid 50s; 74.8% of patients had unilateral disease, and 69.8% of all lesions affected either the lower eyelid or lateral canthus. Recurrence following conventional surgical excision was observed in 2.3% of lesions.1
A review from Japan recounted an incidence of 5 cases per year from 1999 to 2003.4 Patients ranged in age from 30 to 70 years, but there was no gender predilection. Individual apocrine hidrocystomas were mostly less than 2 cm and varied from flesh colored to light red, brown, blue, or purple; 61% of lesions arose periorbitally. Within their cohort, patients with multiple lesions were uncommon, with only 2 cases presenting with 2 lesions simultaneously.4
Apocrine hidrocystomas are thought to result from a cystic proliferation of the secretory component of apocrine sweat glands, though the exact pathogenesis still is unclear.3 Histologic features include a unilocular or multilocular cystic cavity within the dermis lined by columnar cells demonstrating decapitation secretion, followed by a peripheral rim of flattened myoepithelial cells.
Treatment of apocrine hidrocystomas includes topical anticholinergics, surgical excision, electrodesiccation, 1450-nm diode or CO2 lasers, and trichloroacetic acid.2 The novel use of cryotherapy,5 botulinum toxin,2 and intralesional injections of 50% glucose (as a sclerosant)6 also have been reported. Caution should be exercised when managing digital lesions, as digital papillary carcinoma has been described as a clinical and histopathologic mimicker.7
Lipoid proteinosis is a rare autosomal-recessive disorder. Cutaneous lesions manifest in 2 overlapping stages, typically within the first 2 years of life. The first stage consists of vesicles and hemorrhagic crusts on the face and extremities and intraorally, which may heal with scarring. In the second stage, the skin becomes diffusely thickened and waxy, with the appearance of papules, nodules, or plaques along the eyelid margins (moniliform blepharosis), face, axillae, or scrotum. Verrucous lesions also may develop on the knee or elbow extensors.8
Lymphangioma circumscriptum represents microcystic lymphatic malformations that can arise anywhere on the skin or oral mucosa. They present as clusters of clear or hemorrhagic vesicles of variable size and number favoring the proximal extremities and chest. Histologically, dilated lymphatic channels are seen in the upper dermis.8
Syringomas are common benign tumors of the sweat ducts characterized histologically by superficial dermal proliferations of small comma-shaped ducts set in a fibrotic stroma. Clinically, syringomas appear as small, firm, flesh-colored papules with a predilection for the periorbital area. An eruptive onset may be observed, most commonly affecting the trunk. Syringomas may be associated with Down syndrome, while the clear cell variant may be associated with diabetes mellitus.8
Primary systemic amyloidosis may present with a variety of systemic manifestations. Skin involvement can present as waxy, translucent, or purpuric papulonodules or plaques characteristically affecting the periorbital region. Other mucocutaneous signs include macroglossia with or without translucent to hemorrhagic papulovesicles; bruising, especially on the eyelids, neck, axillae, or anogenital area; vesiculobullous skin lesions; or diffuse cutaneous infiltration imparting a sclerodermoid appearance.8
- Maeng M, Petrakos P, Zhou M, et al. Bi-institutional retrospective study on the demographics and basic clinical presentation of hidrocystomas. Orbit. 2017;36:433-435.
- Bordelon JR, Tang N, Elston D, et al. Multiple apocrine hidrocystomas successfully treated with botulinum toxin A. Br J Dermatol. 2017;176:488-490.
- Hafsi W, Badri T. Apocrine hidrocystoma. StatPearls. Treasure Island, FL: StatPearls Publishing; 2017.
- Anzai S, Goto M, Fujiwara S, et al. Apocrine hidrocystoma: a case report and analysis of 167 Japanese cases. Int J Dermatol. 2005;44:702-703.
- Panagiotopoulos A, Vasalou V, Sgontzou T, et al. Multiple apocrine hidrocystomas successfully treated with cryotherapy. Dermatol Surg. 2017;43:993-995.
- Osaki TH, Osaki MH, Osaki T, et al. A minimally invasive approach for apocrine hidrocystomas of the eyelid. Dermatol Surg. 2016;42:134-136.
- Molina-Ruiz AM, Llamas-Velasco M, Rütten A, et al. 'Apocrine hidrocystoma and cystadenoma'-like tumor of the digits or toes: a potential diagnostic pitfall of digital papillary adenocarcinoma. Am J Surg Pathol. 2016;40:410-418.
- Bolognia JL, Jorizzo JL, Schaffer JV, eds. Dermatology. Philadelphia, PA: Elsevier Saunders; 2012.
- Maeng M, Petrakos P, Zhou M, et al. Bi-institutional retrospective study on the demographics and basic clinical presentation of hidrocystomas. Orbit. 2017;36:433-435.
- Bordelon JR, Tang N, Elston D, et al. Multiple apocrine hidrocystomas successfully treated with botulinum toxin A. Br J Dermatol. 2017;176:488-490.
- Hafsi W, Badri T. Apocrine hidrocystoma. StatPearls. Treasure Island, FL: StatPearls Publishing; 2017.
- Anzai S, Goto M, Fujiwara S, et al. Apocrine hidrocystoma: a case report and analysis of 167 Japanese cases. Int J Dermatol. 2005;44:702-703.
- Panagiotopoulos A, Vasalou V, Sgontzou T, et al. Multiple apocrine hidrocystomas successfully treated with cryotherapy. Dermatol Surg. 2017;43:993-995.
- Osaki TH, Osaki MH, Osaki T, et al. A minimally invasive approach for apocrine hidrocystomas of the eyelid. Dermatol Surg. 2016;42:134-136.
- Molina-Ruiz AM, Llamas-Velasco M, Rütten A, et al. 'Apocrine hidrocystoma and cystadenoma'-like tumor of the digits or toes: a potential diagnostic pitfall of digital papillary adenocarcinoma. Am J Surg Pathol. 2016;40:410-418.
- Bolognia JL, Jorizzo JL, Schaffer JV, eds. Dermatology. Philadelphia, PA: Elsevier Saunders; 2012.
A 40-year-old woman was referred to dermatology for evaluation of occasionally pruritic periorbital papules that had gradually increased in size and number over the last 7 to 8 months (top). She had a similar solitary lesion on the left lower eyelid that was removed twice: 10 years and 10 months prior. She was taking an oral contraceptive (desogestrel) but otherwise had no notable medical history or drug allergies. Physical examination revealed individual and clustered translucent papules along the eyelid margins, left medial canthus, and both lateral canthi (bottom).