User login
For MD-IQ use only
The apricot tree
My apricot tree has bloomed. It is a foolish tree planted by a foolish man since it blossoms, with beautiful pink then white flowers, at least 3 weeks too early in Northern Kentucky. Nonetheless, I am hopeful that it will produce fruit, maybe this year.
The apricot tree takes me back to my early childhood in Oklahoma City. We had a small apricot tree in the backyard of our rental house, and my dad would talk about how there was nothing finer than a sun ripened apricot. Those were happy times. My dad was a milkman and was home every day by late afternoon, though he was still taking classes at night to try to finish his degree. My mother was at home and my older brother in first grade down the street. My little sister was small and tried to keep up.
My time was unstructured, and I reveled in the backyard. In retrospect, the backyard was an open display of broken and hoped for dreams. There was a junked car my best friend Alvin and I would sit in, there was a huge tree stump we sat on and played around, we had an old slow dog named Pooch, gifted to us when my mom’s sister moved to Alaska. We ran around with no shirts or shoes, played and pretended, and carefully watched the apricot tree.
I remember one time when the apricots finally ripened. My father climbed up and got me one, and it was so sweet I did not notice that the juice ran down my face and my bare chest. It was the sweetest and most wonderful thing I have ever tasted. All the better for having to wait for it.
. I have had four major meetings canceled and though my livelihood and life are at risk, I feel oddly free and happy. I am no longer under those pressures to research, write, and present, and am spending at lot of time at home with my wife and daughter. I think I will clean out the garage (who knows what I will find?) and work in the backyard – and keep a close watch on the apricot tree.
As many of you have, I have awkwardly embraced telemedicine in the past. It is interesting now, how HIPAA regulations and state licensing requirements have finally been tossed aside, making it possible to practice telemedicine. I suspect things will stay that way if it is demonstrated they are unnecessary.
In my office, we are depopulating the waiting room and autoclaving face masks. I am cleaning out the stockroom and donating extra gloves, gowns, and masks to the local hospital. We may shut down altogether. There is little more I can do unless called to man a ventilator. I hope it doesn’t come to that, but I will serve if called.
I suggest you embrace your current unstructured time and use it to let your mind roam. It is a reprieve from today’s hyperconnected, hurly burly world. I also suggest you check COVID-19 news updates only once a day and turn off television news altogether. Other than following the recommendations and guidance of public health authorities, there is nothing you can do to speed up the resolution of this pandemic.
No matter how awful, this will pass. It is a warm spring and it is possible the apricot tree will not be bitten by frost, and we may have fruit this year. We should know in about 2 months. I am going to keep a close watch on it.
Dr. Coldiron is in private practice but maintains a clinical assistant professorship at the University of Cincinnati. He cares for patients, teaches medical students and residents, and has several active clinical research projects. Dr. Coldiron is the author of more than 80 scientific letters, papers, and several book chapters, and he speaks frequently on a variety of topics. He is a past president of the American Academy of Dermatology. Write to him at [email protected].
My apricot tree has bloomed. It is a foolish tree planted by a foolish man since it blossoms, with beautiful pink then white flowers, at least 3 weeks too early in Northern Kentucky. Nonetheless, I am hopeful that it will produce fruit, maybe this year.
The apricot tree takes me back to my early childhood in Oklahoma City. We had a small apricot tree in the backyard of our rental house, and my dad would talk about how there was nothing finer than a sun ripened apricot. Those were happy times. My dad was a milkman and was home every day by late afternoon, though he was still taking classes at night to try to finish his degree. My mother was at home and my older brother in first grade down the street. My little sister was small and tried to keep up.
My time was unstructured, and I reveled in the backyard. In retrospect, the backyard was an open display of broken and hoped for dreams. There was a junked car my best friend Alvin and I would sit in, there was a huge tree stump we sat on and played around, we had an old slow dog named Pooch, gifted to us when my mom’s sister moved to Alaska. We ran around with no shirts or shoes, played and pretended, and carefully watched the apricot tree.
I remember one time when the apricots finally ripened. My father climbed up and got me one, and it was so sweet I did not notice that the juice ran down my face and my bare chest. It was the sweetest and most wonderful thing I have ever tasted. All the better for having to wait for it.
. I have had four major meetings canceled and though my livelihood and life are at risk, I feel oddly free and happy. I am no longer under those pressures to research, write, and present, and am spending at lot of time at home with my wife and daughter. I think I will clean out the garage (who knows what I will find?) and work in the backyard – and keep a close watch on the apricot tree.
As many of you have, I have awkwardly embraced telemedicine in the past. It is interesting now, how HIPAA regulations and state licensing requirements have finally been tossed aside, making it possible to practice telemedicine. I suspect things will stay that way if it is demonstrated they are unnecessary.
In my office, we are depopulating the waiting room and autoclaving face masks. I am cleaning out the stockroom and donating extra gloves, gowns, and masks to the local hospital. We may shut down altogether. There is little more I can do unless called to man a ventilator. I hope it doesn’t come to that, but I will serve if called.
I suggest you embrace your current unstructured time and use it to let your mind roam. It is a reprieve from today’s hyperconnected, hurly burly world. I also suggest you check COVID-19 news updates only once a day and turn off television news altogether. Other than following the recommendations and guidance of public health authorities, there is nothing you can do to speed up the resolution of this pandemic.
No matter how awful, this will pass. It is a warm spring and it is possible the apricot tree will not be bitten by frost, and we may have fruit this year. We should know in about 2 months. I am going to keep a close watch on it.
Dr. Coldiron is in private practice but maintains a clinical assistant professorship at the University of Cincinnati. He cares for patients, teaches medical students and residents, and has several active clinical research projects. Dr. Coldiron is the author of more than 80 scientific letters, papers, and several book chapters, and he speaks frequently on a variety of topics. He is a past president of the American Academy of Dermatology. Write to him at [email protected].
My apricot tree has bloomed. It is a foolish tree planted by a foolish man since it blossoms, with beautiful pink then white flowers, at least 3 weeks too early in Northern Kentucky. Nonetheless, I am hopeful that it will produce fruit, maybe this year.
The apricot tree takes me back to my early childhood in Oklahoma City. We had a small apricot tree in the backyard of our rental house, and my dad would talk about how there was nothing finer than a sun ripened apricot. Those were happy times. My dad was a milkman and was home every day by late afternoon, though he was still taking classes at night to try to finish his degree. My mother was at home and my older brother in first grade down the street. My little sister was small and tried to keep up.
My time was unstructured, and I reveled in the backyard. In retrospect, the backyard was an open display of broken and hoped for dreams. There was a junked car my best friend Alvin and I would sit in, there was a huge tree stump we sat on and played around, we had an old slow dog named Pooch, gifted to us when my mom’s sister moved to Alaska. We ran around with no shirts or shoes, played and pretended, and carefully watched the apricot tree.
I remember one time when the apricots finally ripened. My father climbed up and got me one, and it was so sweet I did not notice that the juice ran down my face and my bare chest. It was the sweetest and most wonderful thing I have ever tasted. All the better for having to wait for it.
. I have had four major meetings canceled and though my livelihood and life are at risk, I feel oddly free and happy. I am no longer under those pressures to research, write, and present, and am spending at lot of time at home with my wife and daughter. I think I will clean out the garage (who knows what I will find?) and work in the backyard – and keep a close watch on the apricot tree.
As many of you have, I have awkwardly embraced telemedicine in the past. It is interesting now, how HIPAA regulations and state licensing requirements have finally been tossed aside, making it possible to practice telemedicine. I suspect things will stay that way if it is demonstrated they are unnecessary.
In my office, we are depopulating the waiting room and autoclaving face masks. I am cleaning out the stockroom and donating extra gloves, gowns, and masks to the local hospital. We may shut down altogether. There is little more I can do unless called to man a ventilator. I hope it doesn’t come to that, but I will serve if called.
I suggest you embrace your current unstructured time and use it to let your mind roam. It is a reprieve from today’s hyperconnected, hurly burly world. I also suggest you check COVID-19 news updates only once a day and turn off television news altogether. Other than following the recommendations and guidance of public health authorities, there is nothing you can do to speed up the resolution of this pandemic.
No matter how awful, this will pass. It is a warm spring and it is possible the apricot tree will not be bitten by frost, and we may have fruit this year. We should know in about 2 months. I am going to keep a close watch on it.
Dr. Coldiron is in private practice but maintains a clinical assistant professorship at the University of Cincinnati. He cares for patients, teaches medical students and residents, and has several active clinical research projects. Dr. Coldiron is the author of more than 80 scientific letters, papers, and several book chapters, and he speaks frequently on a variety of topics. He is a past president of the American Academy of Dermatology. Write to him at [email protected].
Chlamydia trachomatis infections
CASE Pregnant woman with symptoms of genital infection
A 23-year-old primigravid woman at 15 weeks and 2 days’ gestation reported having a 2-week history of increased urinary frequency and vaginal discharge. She said she experienced similar symptoms 6 weeks previously that resolved within a week. The patient has had 3 sexual partners in the past year. Her current partner was experiencing a yellow urethral discharge and dysuria. On the patient’s speculum examination, the clinician noted a yellow-green discharge emanating from the cervix as well as cervical motion tenderness.
What is the most likely diagnosis, and how would you treat this patient?
The culprit was chlamydia
Chlamydia trachomatis is an obligate intracellular bacterium that does not stain with Gram staining. A rigid cell wall encloses its intracellular component. C trachomatis infection begins when the chlamydial elementary body enters a susceptible host cell.
Once ingested, the organism’s surface antigens (major outer membrane protein and lipopolysaccharide antigens) provide intracellular sanctuary for the bacterium by inhibiting phagolysosomal fusion. Subsequently, the elementary body morphs into a reticular body, which replicates through adenosine triphosphate (ATP)–dependent binary fission. After approximately 48 hours of replication, the organism again morphs into an elementary body and is released to infect additional cells and acquire new ATP stores for further replication.
Chlamydia can be transmitted horizontally during oral, vaginal, or anal intercourse or vertically to the infant during vaginal delivery.
The US’s most common notifiable disease
According to the Centers for Disease Control and Prevention (CDC), the incidence of chlamydia infection in the United States increased considerably in recent years: from 976,455 cases in 2005 to 1,758,668 cases in 2018.1 In 2018, rates of chlamydia infection in women were nearly double the rates in men, with an incidence of 688.2 versus 377.5 per 100,000 cases, and a prevalence of 1,150,672 versus 612,020.1
Young adults have a higher frequency of chlamydia infection than any other age group. From 2017 to 2018, reported cases in women aged 15–19 years increased by 1.3%, to 3,306.8 per 100,000; in women aged 20–24 years, cases increased by 0.8%, to 4,064.6 per 100,000. In young men in the same age ranges, reported cases increased by 3.7%, to 959.0 cases per 100,000, and by 3.3%, to 1,784.5 per 100,000 cases, respectively.1
Both the incidence and prevalence of chlamydia infection are higher in African Americans than in whites, while Asians have the lowest rates.1 The prevalence of infection also is increased with incarceration, lower socioeconomic status, and residence in the southern United States.
The prevalence of chlamydia infection in pregnant women is approximately 2% to 3%, but it may be as high as 30% in high-risk populations, such as women who are unmarried, have multiple sex partners, are coinfected with another sexually transmitted disease (STD), have partners with nongonococcal urethritis, have mucopurulent discharge, have acute urethral syndrome, and have late or no prenatal care.2 Since chlamydia infection often is asymptomatic and some infections resolve spontaneously, the true prevalence of infection probably is underreported.
Continue to: Chlamydia infection can cause serious clinical manifestations...
Chlamydia infection can cause serious clinical manifestations
The 15 serotypes of C trachomatis are grouped into 3 categories according to clinical manifestations:
- Serotypes A, B, Ba, and C cause endemic trachoma, characterized by bilateral irritation of the eyelids that progresses to eyelid thickening and scarring, eventually leading to corneal abrasion and blindness.
- Serotypes D–K manifest as conjunctivitis and pneumonia in newborns, proctitis in men (especially in men who have sex with men), and genitourinary infections in women. Reactive arthritis and inclusion conjunctivitis also can occur with D–K serotypes.
- Serotypes L1–L3 cause lymphogranuloma venereum.
About 70% of women with chlamydia infection are asymptomatic. Those who have symptoms often present with endocervicitis or acute urethral syndrome (acute urethritis). Manifestations of these 2 conditions include a frothy yellow-green vaginal and/or urethral discharge, dysuria, and frequency. Women who engage in rectal intercourse also may notice a purulent discharge from the anus. Untreated, C trachomatis organisms may ascend the reproductive tract, causing both endometritis and pelvic inflammatory disease (PID).
While a single episode of PID increases tubal infertility risk by 10%, a second episode increases the risk by 40%.3 Over time, recurrent and/or chronic PID causes scarring and adhesion formation, which may result in chronic pelvic pain. In addition, chronic infection is the single most important risk factor for ectopic pregnancy. Finally, chlamydia infection is a risk factor for Fitz-Hugh-Cutis syndrome (perihepatitis). In this condition, organisms ascend from the site of pelvic infection along the pericolic gutter to ultimately infect the liver capsule.
Specific complications in pregnancy
Chlamydia infection in pregnant women is associated with preterm delivery and preterm premature rupture of membranes. Infants born to mothers with untreated chlamydia infection are at risk for pneumonia, conjunctivitis, and even perinatal death.2 Acquisition of infection occurs at the time of delivery rather than in the antepartum period.
The significant morbidity associated with chlamydia infection underscores the importance of regular screening, especially in pregnant women. The current United States Preventive Service Task Force guidelines recommend annual screening of all sexually active women who are 24 years of age or younger, as well as of older, high-risk women.
The CDC recommends routine screening of all pregnant women for chlamydia at the first prenatal visit. Repeat screening is recommended in the third trimester for all pregnant women younger than 25 years, those at increased risk, and those infected within the past 3 to 6 months or during the first trimester. Those who test positive should be retested 3 weeks after completion of treatment.1
Chlamydia screening strategies
Historically, a chlamydia diagnosis was made by isolating the organisms in tissue culture. In the 1990s, however, that extremely time-consuming and resource-intensive procedure was replaced by nucleic acid amplification testing (NAAT).
NAAT methodology. NAAT is the gold standard for diagnosing C trachomatis infection; this methodology utilizes various assays, including polymerase chain reaction, ligase chain reaction, and transcription-mediated amplification.
Continue to: Compared with previous culture and antigen detection techniques...
Compared with previous culture and antigen detection techniques, NAAT’s advantages include excellent sensitivity and specificity (>90% and ≥99%, respectively), enabling detection of a low inoculum of organisms in a sample obtained by noninvasive methods, such as first-void urine collection or vaginal swab.2,4,5 Furthermore, NAAT does not impose any specific storage regulations on collected specimens, is cost effective, and can jointly test for Neisseria gonorrhoeae, which commonly co-infects with C trachomatis.6
Screening in pregnancy. In 2012, Blatt and colleagues examined testing patterns in nearly 1.3 million obstetric patients and found that only 59% (761,315) of women were tested for chlamydia at least once in pregnancy.7 Only 1 in 3 women were tested during the first prenatal visit, as CDC guidelines recommend. Testing rates declined with increasing age. Of women screened, 3.5% tested positive for chlamydia.7 Of these, 3 of 4 were retested at least once, with almost 20% having at least 1 subsequent positive result.7
Of note, in a study of women who reported receptive anal intercourse (n = 2,818), 292 women tested positive for chlamydia; 10.4% tested positive in genital-only sites, 58.6% in genital and rectal sites, and 20.5% at the rectal site only.8
It is alarming that only 59% of pregnant women are screened for chlamydia given the significant perinatal complications associated with this infection. Barriers to screening pregnant women may include clinician discomfort in discussing STDs and patient refusal of screening. Furthermore, clinicians should routinely ask women about receptive anal sex. Women who report this risk factor should be tested for chlamydia in both the endocervix and rectum.
Retesting and follow-up. After the initial diagnosis of chlamydia, a test of cure 3 weeks after treatment is an important aspect of care. Thus, identifying and overcoming barriers to retesting is important. Clinicians should educate patients about the importance of follow-up. Also consider incorporating the use of home-based, self-obtained vaginal swabs for retesting. Results from 2 randomized trials showed that eliminating a patient’s transportation barriers and providing a home-based alternative to a follow-up visit significantly increased rescreening rates by 33% in STD clinic patients and by 59.2% in family planning clinic patients.9
Reinfection risk. The rate of venereal chlamydia transmission in heterosexual partners is 70%. Since sexually active chlamydia-positive patients are at risk for reinfection by their partner after treatment completion, clinicians should refer the sex partners for evaluation. If the sex partners are reluctant to have testing, it is reasonable to provide empiric antibiotic treatment to decrease the risk of re-infection in the patient.7 Before doing so, however, make certain that state law permits this practice, and be sure to document the prescribed treatment in the patient’s record.
Treatment options
Prompt treatment of C trachomatis infection is essential to decrease the risk of disease sequelae. Nonpregnant adults can be treated with oral doxycycline 100 mg twice daily for 7 days.
In a head-to-head study performed in a controlled environment that ensured treatment adherence, 97% efficacy was achieved with one oral dose of azithromycin (1 g) compared with 100% efficacy with doxycycline.10 However, in the real-world setting, imperfect adherence to the multi-day doxycycline regimen is associated with treatment failures. Thus, a single dose of azithromycin is preferable for patients with questionable compliance.11
In obstetric patients, azithromycin and amoxicillin are preferred as first-line agents for treatment of C trachomatis due to their improved safety profile in this demographic. Amoxicillin 500 mg orally 3 times daily for 7 days has 95% efficacy.2
Women allergic to these agents may be treated with an alternative regimen of erythromycin base, 500 mg orally 4 times daily for 7 days, or erythromycin ethylsuccinate, 800 mg orally 4 times daily for 7 days. Erythromycin should be reserved for second-line therapy because of its lower efficacy (64%) and frequent gastrointestinal adverse effects.2 Doxycycline is contraindicated in pregnancy because of possible teratogenic effects on the teeth and bone of the fetus.
- Centers for Disease Control and Prevention Division of STD Prevention. Sexually transmitted disease surveillance 2018. October 2019. https://www.cdc.gov/std/stats18/default.htm. 2019. Accessed January 4, 2020.
- Duff P. Maternal and fetal infections. In: Creasy RK, Resnik R, Iams JD, et al, eds. Creasy and Resnik’s Maternal-Fetal Medicine: Principles and Practice. 8th ed. Philadelphia, PA: Elsevier Saunders; 2019:869.
- Ljubin-Sternak S, Meštrović T. Chlamydia trachomatis and genital mycoplasmas: pathogens with an impact on human reproductive health. J Pathog. 2014. doi: 10.1155/2014/183167.
- Meyer T. Diagnostic procedures to detect Chlamydia trachomatis infections. Microorganisms. 2016:4(3).
- Centers for Disease Control and Prevention.. Recommendations for the laboratory-based detection of Chlamydia trachomatis and Neisseria gonorrhoeae. MMWR Recomm Rep. 2014;63:1-19.
- Wiesenfeld HC. Screening for Chlamydia trachomatis infections in women. N Engl J Med. 2017;376:765-773.
- Blatt AJ, Lieberman JM, Hoover DR, et al. Chlamydial and gonococcal testing during pregnancy in the United States. Am J Obstet Gynecol. 2012;207:55.e1-8.
- Llata E, Braxton J, Asbel L, et al. Rectal Chlamydia trachomatis and Neisseria gonorrhoeae infections among women reporting anal intercourse. Obstet Gynecol. 2018;132:692-697.
- Xu F, Stoner BP, Taylor SN, et al. Use of home-obtained vaginal swabs to facilitate rescreening for Chlamydia trachomatis infections: two randomized controlled trials. Obstet Gynecol. 2011;118(2 pt 1):231-239.
- Geisler WM, Uniyal A, Lee JY, et al. Azithromycin versus doxycycline for urogenital Chlamydia trachomatis infection. N Engl J Med. 2015;373:2512-2521.
- Quinn TC, Gaydos CA. Treatment for chlamydia infection—doxycycline versus azithromycin. N Engl J Med. 2015;373:2573-2575.
CASE Pregnant woman with symptoms of genital infection
A 23-year-old primigravid woman at 15 weeks and 2 days’ gestation reported having a 2-week history of increased urinary frequency and vaginal discharge. She said she experienced similar symptoms 6 weeks previously that resolved within a week. The patient has had 3 sexual partners in the past year. Her current partner was experiencing a yellow urethral discharge and dysuria. On the patient’s speculum examination, the clinician noted a yellow-green discharge emanating from the cervix as well as cervical motion tenderness.
What is the most likely diagnosis, and how would you treat this patient?
The culprit was chlamydia
Chlamydia trachomatis is an obligate intracellular bacterium that does not stain with Gram staining. A rigid cell wall encloses its intracellular component. C trachomatis infection begins when the chlamydial elementary body enters a susceptible host cell.
Once ingested, the organism’s surface antigens (major outer membrane protein and lipopolysaccharide antigens) provide intracellular sanctuary for the bacterium by inhibiting phagolysosomal fusion. Subsequently, the elementary body morphs into a reticular body, which replicates through adenosine triphosphate (ATP)–dependent binary fission. After approximately 48 hours of replication, the organism again morphs into an elementary body and is released to infect additional cells and acquire new ATP stores for further replication.
Chlamydia can be transmitted horizontally during oral, vaginal, or anal intercourse or vertically to the infant during vaginal delivery.
The US’s most common notifiable disease
According to the Centers for Disease Control and Prevention (CDC), the incidence of chlamydia infection in the United States increased considerably in recent years: from 976,455 cases in 2005 to 1,758,668 cases in 2018.1 In 2018, rates of chlamydia infection in women were nearly double the rates in men, with an incidence of 688.2 versus 377.5 per 100,000 cases, and a prevalence of 1,150,672 versus 612,020.1
Young adults have a higher frequency of chlamydia infection than any other age group. From 2017 to 2018, reported cases in women aged 15–19 years increased by 1.3%, to 3,306.8 per 100,000; in women aged 20–24 years, cases increased by 0.8%, to 4,064.6 per 100,000. In young men in the same age ranges, reported cases increased by 3.7%, to 959.0 cases per 100,000, and by 3.3%, to 1,784.5 per 100,000 cases, respectively.1
Both the incidence and prevalence of chlamydia infection are higher in African Americans than in whites, while Asians have the lowest rates.1 The prevalence of infection also is increased with incarceration, lower socioeconomic status, and residence in the southern United States.
The prevalence of chlamydia infection in pregnant women is approximately 2% to 3%, but it may be as high as 30% in high-risk populations, such as women who are unmarried, have multiple sex partners, are coinfected with another sexually transmitted disease (STD), have partners with nongonococcal urethritis, have mucopurulent discharge, have acute urethral syndrome, and have late or no prenatal care.2 Since chlamydia infection often is asymptomatic and some infections resolve spontaneously, the true prevalence of infection probably is underreported.
Continue to: Chlamydia infection can cause serious clinical manifestations...
Chlamydia infection can cause serious clinical manifestations
The 15 serotypes of C trachomatis are grouped into 3 categories according to clinical manifestations:
- Serotypes A, B, Ba, and C cause endemic trachoma, characterized by bilateral irritation of the eyelids that progresses to eyelid thickening and scarring, eventually leading to corneal abrasion and blindness.
- Serotypes D–K manifest as conjunctivitis and pneumonia in newborns, proctitis in men (especially in men who have sex with men), and genitourinary infections in women. Reactive arthritis and inclusion conjunctivitis also can occur with D–K serotypes.
- Serotypes L1–L3 cause lymphogranuloma venereum.
About 70% of women with chlamydia infection are asymptomatic. Those who have symptoms often present with endocervicitis or acute urethral syndrome (acute urethritis). Manifestations of these 2 conditions include a frothy yellow-green vaginal and/or urethral discharge, dysuria, and frequency. Women who engage in rectal intercourse also may notice a purulent discharge from the anus. Untreated, C trachomatis organisms may ascend the reproductive tract, causing both endometritis and pelvic inflammatory disease (PID).
While a single episode of PID increases tubal infertility risk by 10%, a second episode increases the risk by 40%.3 Over time, recurrent and/or chronic PID causes scarring and adhesion formation, which may result in chronic pelvic pain. In addition, chronic infection is the single most important risk factor for ectopic pregnancy. Finally, chlamydia infection is a risk factor for Fitz-Hugh-Cutis syndrome (perihepatitis). In this condition, organisms ascend from the site of pelvic infection along the pericolic gutter to ultimately infect the liver capsule.
Specific complications in pregnancy
Chlamydia infection in pregnant women is associated with preterm delivery and preterm premature rupture of membranes. Infants born to mothers with untreated chlamydia infection are at risk for pneumonia, conjunctivitis, and even perinatal death.2 Acquisition of infection occurs at the time of delivery rather than in the antepartum period.
The significant morbidity associated with chlamydia infection underscores the importance of regular screening, especially in pregnant women. The current United States Preventive Service Task Force guidelines recommend annual screening of all sexually active women who are 24 years of age or younger, as well as of older, high-risk women.
The CDC recommends routine screening of all pregnant women for chlamydia at the first prenatal visit. Repeat screening is recommended in the third trimester for all pregnant women younger than 25 years, those at increased risk, and those infected within the past 3 to 6 months or during the first trimester. Those who test positive should be retested 3 weeks after completion of treatment.1
Chlamydia screening strategies
Historically, a chlamydia diagnosis was made by isolating the organisms in tissue culture. In the 1990s, however, that extremely time-consuming and resource-intensive procedure was replaced by nucleic acid amplification testing (NAAT).
NAAT methodology. NAAT is the gold standard for diagnosing C trachomatis infection; this methodology utilizes various assays, including polymerase chain reaction, ligase chain reaction, and transcription-mediated amplification.
Continue to: Compared with previous culture and antigen detection techniques...
Compared with previous culture and antigen detection techniques, NAAT’s advantages include excellent sensitivity and specificity (>90% and ≥99%, respectively), enabling detection of a low inoculum of organisms in a sample obtained by noninvasive methods, such as first-void urine collection or vaginal swab.2,4,5 Furthermore, NAAT does not impose any specific storage regulations on collected specimens, is cost effective, and can jointly test for Neisseria gonorrhoeae, which commonly co-infects with C trachomatis.6
Screening in pregnancy. In 2012, Blatt and colleagues examined testing patterns in nearly 1.3 million obstetric patients and found that only 59% (761,315) of women were tested for chlamydia at least once in pregnancy.7 Only 1 in 3 women were tested during the first prenatal visit, as CDC guidelines recommend. Testing rates declined with increasing age. Of women screened, 3.5% tested positive for chlamydia.7 Of these, 3 of 4 were retested at least once, with almost 20% having at least 1 subsequent positive result.7
Of note, in a study of women who reported receptive anal intercourse (n = 2,818), 292 women tested positive for chlamydia; 10.4% tested positive in genital-only sites, 58.6% in genital and rectal sites, and 20.5% at the rectal site only.8
It is alarming that only 59% of pregnant women are screened for chlamydia given the significant perinatal complications associated with this infection. Barriers to screening pregnant women may include clinician discomfort in discussing STDs and patient refusal of screening. Furthermore, clinicians should routinely ask women about receptive anal sex. Women who report this risk factor should be tested for chlamydia in both the endocervix and rectum.
Retesting and follow-up. After the initial diagnosis of chlamydia, a test of cure 3 weeks after treatment is an important aspect of care. Thus, identifying and overcoming barriers to retesting is important. Clinicians should educate patients about the importance of follow-up. Also consider incorporating the use of home-based, self-obtained vaginal swabs for retesting. Results from 2 randomized trials showed that eliminating a patient’s transportation barriers and providing a home-based alternative to a follow-up visit significantly increased rescreening rates by 33% in STD clinic patients and by 59.2% in family planning clinic patients.9
Reinfection risk. The rate of venereal chlamydia transmission in heterosexual partners is 70%. Since sexually active chlamydia-positive patients are at risk for reinfection by their partner after treatment completion, clinicians should refer the sex partners for evaluation. If the sex partners are reluctant to have testing, it is reasonable to provide empiric antibiotic treatment to decrease the risk of re-infection in the patient.7 Before doing so, however, make certain that state law permits this practice, and be sure to document the prescribed treatment in the patient’s record.
Treatment options
Prompt treatment of C trachomatis infection is essential to decrease the risk of disease sequelae. Nonpregnant adults can be treated with oral doxycycline 100 mg twice daily for 7 days.
In a head-to-head study performed in a controlled environment that ensured treatment adherence, 97% efficacy was achieved with one oral dose of azithromycin (1 g) compared with 100% efficacy with doxycycline.10 However, in the real-world setting, imperfect adherence to the multi-day doxycycline regimen is associated with treatment failures. Thus, a single dose of azithromycin is preferable for patients with questionable compliance.11
In obstetric patients, azithromycin and amoxicillin are preferred as first-line agents for treatment of C trachomatis due to their improved safety profile in this demographic. Amoxicillin 500 mg orally 3 times daily for 7 days has 95% efficacy.2
Women allergic to these agents may be treated with an alternative regimen of erythromycin base, 500 mg orally 4 times daily for 7 days, or erythromycin ethylsuccinate, 800 mg orally 4 times daily for 7 days. Erythromycin should be reserved for second-line therapy because of its lower efficacy (64%) and frequent gastrointestinal adverse effects.2 Doxycycline is contraindicated in pregnancy because of possible teratogenic effects on the teeth and bone of the fetus.
CASE Pregnant woman with symptoms of genital infection
A 23-year-old primigravid woman at 15 weeks and 2 days’ gestation reported having a 2-week history of increased urinary frequency and vaginal discharge. She said she experienced similar symptoms 6 weeks previously that resolved within a week. The patient has had 3 sexual partners in the past year. Her current partner was experiencing a yellow urethral discharge and dysuria. On the patient’s speculum examination, the clinician noted a yellow-green discharge emanating from the cervix as well as cervical motion tenderness.
What is the most likely diagnosis, and how would you treat this patient?
The culprit was chlamydia
Chlamydia trachomatis is an obligate intracellular bacterium that does not stain with Gram staining. A rigid cell wall encloses its intracellular component. C trachomatis infection begins when the chlamydial elementary body enters a susceptible host cell.
Once ingested, the organism’s surface antigens (major outer membrane protein and lipopolysaccharide antigens) provide intracellular sanctuary for the bacterium by inhibiting phagolysosomal fusion. Subsequently, the elementary body morphs into a reticular body, which replicates through adenosine triphosphate (ATP)–dependent binary fission. After approximately 48 hours of replication, the organism again morphs into an elementary body and is released to infect additional cells and acquire new ATP stores for further replication.
Chlamydia can be transmitted horizontally during oral, vaginal, or anal intercourse or vertically to the infant during vaginal delivery.
The US’s most common notifiable disease
According to the Centers for Disease Control and Prevention (CDC), the incidence of chlamydia infection in the United States increased considerably in recent years: from 976,455 cases in 2005 to 1,758,668 cases in 2018.1 In 2018, rates of chlamydia infection in women were nearly double the rates in men, with an incidence of 688.2 versus 377.5 per 100,000 cases, and a prevalence of 1,150,672 versus 612,020.1
Young adults have a higher frequency of chlamydia infection than any other age group. From 2017 to 2018, reported cases in women aged 15–19 years increased by 1.3%, to 3,306.8 per 100,000; in women aged 20–24 years, cases increased by 0.8%, to 4,064.6 per 100,000. In young men in the same age ranges, reported cases increased by 3.7%, to 959.0 cases per 100,000, and by 3.3%, to 1,784.5 per 100,000 cases, respectively.1
Both the incidence and prevalence of chlamydia infection are higher in African Americans than in whites, while Asians have the lowest rates.1 The prevalence of infection also is increased with incarceration, lower socioeconomic status, and residence in the southern United States.
The prevalence of chlamydia infection in pregnant women is approximately 2% to 3%, but it may be as high as 30% in high-risk populations, such as women who are unmarried, have multiple sex partners, are coinfected with another sexually transmitted disease (STD), have partners with nongonococcal urethritis, have mucopurulent discharge, have acute urethral syndrome, and have late or no prenatal care.2 Since chlamydia infection often is asymptomatic and some infections resolve spontaneously, the true prevalence of infection probably is underreported.
Continue to: Chlamydia infection can cause serious clinical manifestations...
Chlamydia infection can cause serious clinical manifestations
The 15 serotypes of C trachomatis are grouped into 3 categories according to clinical manifestations:
- Serotypes A, B, Ba, and C cause endemic trachoma, characterized by bilateral irritation of the eyelids that progresses to eyelid thickening and scarring, eventually leading to corneal abrasion and blindness.
- Serotypes D–K manifest as conjunctivitis and pneumonia in newborns, proctitis in men (especially in men who have sex with men), and genitourinary infections in women. Reactive arthritis and inclusion conjunctivitis also can occur with D–K serotypes.
- Serotypes L1–L3 cause lymphogranuloma venereum.
About 70% of women with chlamydia infection are asymptomatic. Those who have symptoms often present with endocervicitis or acute urethral syndrome (acute urethritis). Manifestations of these 2 conditions include a frothy yellow-green vaginal and/or urethral discharge, dysuria, and frequency. Women who engage in rectal intercourse also may notice a purulent discharge from the anus. Untreated, C trachomatis organisms may ascend the reproductive tract, causing both endometritis and pelvic inflammatory disease (PID).
While a single episode of PID increases tubal infertility risk by 10%, a second episode increases the risk by 40%.3 Over time, recurrent and/or chronic PID causes scarring and adhesion formation, which may result in chronic pelvic pain. In addition, chronic infection is the single most important risk factor for ectopic pregnancy. Finally, chlamydia infection is a risk factor for Fitz-Hugh-Cutis syndrome (perihepatitis). In this condition, organisms ascend from the site of pelvic infection along the pericolic gutter to ultimately infect the liver capsule.
Specific complications in pregnancy
Chlamydia infection in pregnant women is associated with preterm delivery and preterm premature rupture of membranes. Infants born to mothers with untreated chlamydia infection are at risk for pneumonia, conjunctivitis, and even perinatal death.2 Acquisition of infection occurs at the time of delivery rather than in the antepartum period.
The significant morbidity associated with chlamydia infection underscores the importance of regular screening, especially in pregnant women. The current United States Preventive Service Task Force guidelines recommend annual screening of all sexually active women who are 24 years of age or younger, as well as of older, high-risk women.
The CDC recommends routine screening of all pregnant women for chlamydia at the first prenatal visit. Repeat screening is recommended in the third trimester for all pregnant women younger than 25 years, those at increased risk, and those infected within the past 3 to 6 months or during the first trimester. Those who test positive should be retested 3 weeks after completion of treatment.1
Chlamydia screening strategies
Historically, a chlamydia diagnosis was made by isolating the organisms in tissue culture. In the 1990s, however, that extremely time-consuming and resource-intensive procedure was replaced by nucleic acid amplification testing (NAAT).
NAAT methodology. NAAT is the gold standard for diagnosing C trachomatis infection; this methodology utilizes various assays, including polymerase chain reaction, ligase chain reaction, and transcription-mediated amplification.
Continue to: Compared with previous culture and antigen detection techniques...
Compared with previous culture and antigen detection techniques, NAAT’s advantages include excellent sensitivity and specificity (>90% and ≥99%, respectively), enabling detection of a low inoculum of organisms in a sample obtained by noninvasive methods, such as first-void urine collection or vaginal swab.2,4,5 Furthermore, NAAT does not impose any specific storage regulations on collected specimens, is cost effective, and can jointly test for Neisseria gonorrhoeae, which commonly co-infects with C trachomatis.6
Screening in pregnancy. In 2012, Blatt and colleagues examined testing patterns in nearly 1.3 million obstetric patients and found that only 59% (761,315) of women were tested for chlamydia at least once in pregnancy.7 Only 1 in 3 women were tested during the first prenatal visit, as CDC guidelines recommend. Testing rates declined with increasing age. Of women screened, 3.5% tested positive for chlamydia.7 Of these, 3 of 4 were retested at least once, with almost 20% having at least 1 subsequent positive result.7
Of note, in a study of women who reported receptive anal intercourse (n = 2,818), 292 women tested positive for chlamydia; 10.4% tested positive in genital-only sites, 58.6% in genital and rectal sites, and 20.5% at the rectal site only.8
It is alarming that only 59% of pregnant women are screened for chlamydia given the significant perinatal complications associated with this infection. Barriers to screening pregnant women may include clinician discomfort in discussing STDs and patient refusal of screening. Furthermore, clinicians should routinely ask women about receptive anal sex. Women who report this risk factor should be tested for chlamydia in both the endocervix and rectum.
Retesting and follow-up. After the initial diagnosis of chlamydia, a test of cure 3 weeks after treatment is an important aspect of care. Thus, identifying and overcoming barriers to retesting is important. Clinicians should educate patients about the importance of follow-up. Also consider incorporating the use of home-based, self-obtained vaginal swabs for retesting. Results from 2 randomized trials showed that eliminating a patient’s transportation barriers and providing a home-based alternative to a follow-up visit significantly increased rescreening rates by 33% in STD clinic patients and by 59.2% in family planning clinic patients.9
Reinfection risk. The rate of venereal chlamydia transmission in heterosexual partners is 70%. Since sexually active chlamydia-positive patients are at risk for reinfection by their partner after treatment completion, clinicians should refer the sex partners for evaluation. If the sex partners are reluctant to have testing, it is reasonable to provide empiric antibiotic treatment to decrease the risk of re-infection in the patient.7 Before doing so, however, make certain that state law permits this practice, and be sure to document the prescribed treatment in the patient’s record.
Treatment options
Prompt treatment of C trachomatis infection is essential to decrease the risk of disease sequelae. Nonpregnant adults can be treated with oral doxycycline 100 mg twice daily for 7 days.
In a head-to-head study performed in a controlled environment that ensured treatment adherence, 97% efficacy was achieved with one oral dose of azithromycin (1 g) compared with 100% efficacy with doxycycline.10 However, in the real-world setting, imperfect adherence to the multi-day doxycycline regimen is associated with treatment failures. Thus, a single dose of azithromycin is preferable for patients with questionable compliance.11
In obstetric patients, azithromycin and amoxicillin are preferred as first-line agents for treatment of C trachomatis due to their improved safety profile in this demographic. Amoxicillin 500 mg orally 3 times daily for 7 days has 95% efficacy.2
Women allergic to these agents may be treated with an alternative regimen of erythromycin base, 500 mg orally 4 times daily for 7 days, or erythromycin ethylsuccinate, 800 mg orally 4 times daily for 7 days. Erythromycin should be reserved for second-line therapy because of its lower efficacy (64%) and frequent gastrointestinal adverse effects.2 Doxycycline is contraindicated in pregnancy because of possible teratogenic effects on the teeth and bone of the fetus.
- Centers for Disease Control and Prevention Division of STD Prevention. Sexually transmitted disease surveillance 2018. October 2019. https://www.cdc.gov/std/stats18/default.htm. 2019. Accessed January 4, 2020.
- Duff P. Maternal and fetal infections. In: Creasy RK, Resnik R, Iams JD, et al, eds. Creasy and Resnik’s Maternal-Fetal Medicine: Principles and Practice. 8th ed. Philadelphia, PA: Elsevier Saunders; 2019:869.
- Ljubin-Sternak S, Meštrović T. Chlamydia trachomatis and genital mycoplasmas: pathogens with an impact on human reproductive health. J Pathog. 2014. doi: 10.1155/2014/183167.
- Meyer T. Diagnostic procedures to detect Chlamydia trachomatis infections. Microorganisms. 2016:4(3).
- Centers for Disease Control and Prevention.. Recommendations for the laboratory-based detection of Chlamydia trachomatis and Neisseria gonorrhoeae. MMWR Recomm Rep. 2014;63:1-19.
- Wiesenfeld HC. Screening for Chlamydia trachomatis infections in women. N Engl J Med. 2017;376:765-773.
- Blatt AJ, Lieberman JM, Hoover DR, et al. Chlamydial and gonococcal testing during pregnancy in the United States. Am J Obstet Gynecol. 2012;207:55.e1-8.
- Llata E, Braxton J, Asbel L, et al. Rectal Chlamydia trachomatis and Neisseria gonorrhoeae infections among women reporting anal intercourse. Obstet Gynecol. 2018;132:692-697.
- Xu F, Stoner BP, Taylor SN, et al. Use of home-obtained vaginal swabs to facilitate rescreening for Chlamydia trachomatis infections: two randomized controlled trials. Obstet Gynecol. 2011;118(2 pt 1):231-239.
- Geisler WM, Uniyal A, Lee JY, et al. Azithromycin versus doxycycline for urogenital Chlamydia trachomatis infection. N Engl J Med. 2015;373:2512-2521.
- Quinn TC, Gaydos CA. Treatment for chlamydia infection—doxycycline versus azithromycin. N Engl J Med. 2015;373:2573-2575.
- Centers for Disease Control and Prevention Division of STD Prevention. Sexually transmitted disease surveillance 2018. October 2019. https://www.cdc.gov/std/stats18/default.htm. 2019. Accessed January 4, 2020.
- Duff P. Maternal and fetal infections. In: Creasy RK, Resnik R, Iams JD, et al, eds. Creasy and Resnik’s Maternal-Fetal Medicine: Principles and Practice. 8th ed. Philadelphia, PA: Elsevier Saunders; 2019:869.
- Ljubin-Sternak S, Meštrović T. Chlamydia trachomatis and genital mycoplasmas: pathogens with an impact on human reproductive health. J Pathog. 2014. doi: 10.1155/2014/183167.
- Meyer T. Diagnostic procedures to detect Chlamydia trachomatis infections. Microorganisms. 2016:4(3).
- Centers for Disease Control and Prevention.. Recommendations for the laboratory-based detection of Chlamydia trachomatis and Neisseria gonorrhoeae. MMWR Recomm Rep. 2014;63:1-19.
- Wiesenfeld HC. Screening for Chlamydia trachomatis infections in women. N Engl J Med. 2017;376:765-773.
- Blatt AJ, Lieberman JM, Hoover DR, et al. Chlamydial and gonococcal testing during pregnancy in the United States. Am J Obstet Gynecol. 2012;207:55.e1-8.
- Llata E, Braxton J, Asbel L, et al. Rectal Chlamydia trachomatis and Neisseria gonorrhoeae infections among women reporting anal intercourse. Obstet Gynecol. 2018;132:692-697.
- Xu F, Stoner BP, Taylor SN, et al. Use of home-obtained vaginal swabs to facilitate rescreening for Chlamydia trachomatis infections: two randomized controlled trials. Obstet Gynecol. 2011;118(2 pt 1):231-239.
- Geisler WM, Uniyal A, Lee JY, et al. Azithromycin versus doxycycline for urogenital Chlamydia trachomatis infection. N Engl J Med. 2015;373:2512-2521.
- Quinn TC, Gaydos CA. Treatment for chlamydia infection—doxycycline versus azithromycin. N Engl J Med. 2015;373:2573-2575.
ACP outlines guide for COVID-19 telehealth coding, billing
and for handling clinician and staff absences due to illness or quarantine during the COVID-19 pandemic.
It strongly encourages practices to use telehealth, whenever possible, to mitigate exposure of patients who are sick or at risk because of other underlying conditions and to protect health care workers and the community from the spread of the disease.
The national organization of internists also recommends in the guidance that practices establish protocols and procedures for use by clinicians and all other staff in light of the pandemic.
The billing and coding tips are being offered to help practices deal with the rapidly changing situation surrounding the COVID-19 emergency, according to a statement from the ACP.
The coding-related guidance incorporates changes to a number of telehealth rules for Medicare beneficiaries, announced by the Centers for Medicare and Medicaid Services on March 17.
“Now in a full state of emergency, many Medicare restrictions related to telehealth have been lifted. Patients can be at home, and non-HIPAA compliant technology is allowed. There is no cost sharing for COVID-19 testing. In addition, to encourage use by patients, Medicare is allowing practices to waive cost sharing (copays and deductibles) for all telehealth services,” the organization said in the guidance. It notes, however, that the CMS does not currently reimburse for telephone calls.
The guidance includes details of the new ICD-10 codes, and stresses the importance of using the appropriate codes, given that some service cost-sharing has been waived for COVID-19 testing and treatment.
There is detailed coding guidance for virtual check-in, online evaluation and management, remote monitoring, originating site, and allowed technology and services.
In regard to clinician and staff absence due to illness or quarantine, the ACP says “practices may need to review emergency plans related to telework and to employee and clinician absence.” Among its recommendations are that practices and employers consider temporary adjustments to compensation formulas to accommodate those clinicians who experience a loss of income because they are paid based on production.
The organization emphasizes that, given the rapidly changing availability of testing for COVID-19, practices should contact their local health departments, hospitals, reference labs, or state health authorities to determine the status of their access to testing.
The full list of the ACP’s tips are available here.
Any new guidance for physicians will be posted on the ACP’s COVID-19 resource page.
and for handling clinician and staff absences due to illness or quarantine during the COVID-19 pandemic.
It strongly encourages practices to use telehealth, whenever possible, to mitigate exposure of patients who are sick or at risk because of other underlying conditions and to protect health care workers and the community from the spread of the disease.
The national organization of internists also recommends in the guidance that practices establish protocols and procedures for use by clinicians and all other staff in light of the pandemic.
The billing and coding tips are being offered to help practices deal with the rapidly changing situation surrounding the COVID-19 emergency, according to a statement from the ACP.
The coding-related guidance incorporates changes to a number of telehealth rules for Medicare beneficiaries, announced by the Centers for Medicare and Medicaid Services on March 17.
“Now in a full state of emergency, many Medicare restrictions related to telehealth have been lifted. Patients can be at home, and non-HIPAA compliant technology is allowed. There is no cost sharing for COVID-19 testing. In addition, to encourage use by patients, Medicare is allowing practices to waive cost sharing (copays and deductibles) for all telehealth services,” the organization said in the guidance. It notes, however, that the CMS does not currently reimburse for telephone calls.
The guidance includes details of the new ICD-10 codes, and stresses the importance of using the appropriate codes, given that some service cost-sharing has been waived for COVID-19 testing and treatment.
There is detailed coding guidance for virtual check-in, online evaluation and management, remote monitoring, originating site, and allowed technology and services.
In regard to clinician and staff absence due to illness or quarantine, the ACP says “practices may need to review emergency plans related to telework and to employee and clinician absence.” Among its recommendations are that practices and employers consider temporary adjustments to compensation formulas to accommodate those clinicians who experience a loss of income because they are paid based on production.
The organization emphasizes that, given the rapidly changing availability of testing for COVID-19, practices should contact their local health departments, hospitals, reference labs, or state health authorities to determine the status of their access to testing.
The full list of the ACP’s tips are available here.
Any new guidance for physicians will be posted on the ACP’s COVID-19 resource page.
and for handling clinician and staff absences due to illness or quarantine during the COVID-19 pandemic.
It strongly encourages practices to use telehealth, whenever possible, to mitigate exposure of patients who are sick or at risk because of other underlying conditions and to protect health care workers and the community from the spread of the disease.
The national organization of internists also recommends in the guidance that practices establish protocols and procedures for use by clinicians and all other staff in light of the pandemic.
The billing and coding tips are being offered to help practices deal with the rapidly changing situation surrounding the COVID-19 emergency, according to a statement from the ACP.
The coding-related guidance incorporates changes to a number of telehealth rules for Medicare beneficiaries, announced by the Centers for Medicare and Medicaid Services on March 17.
“Now in a full state of emergency, many Medicare restrictions related to telehealth have been lifted. Patients can be at home, and non-HIPAA compliant technology is allowed. There is no cost sharing for COVID-19 testing. In addition, to encourage use by patients, Medicare is allowing practices to waive cost sharing (copays and deductibles) for all telehealth services,” the organization said in the guidance. It notes, however, that the CMS does not currently reimburse for telephone calls.
The guidance includes details of the new ICD-10 codes, and stresses the importance of using the appropriate codes, given that some service cost-sharing has been waived for COVID-19 testing and treatment.
There is detailed coding guidance for virtual check-in, online evaluation and management, remote monitoring, originating site, and allowed technology and services.
In regard to clinician and staff absence due to illness or quarantine, the ACP says “practices may need to review emergency plans related to telework and to employee and clinician absence.” Among its recommendations are that practices and employers consider temporary adjustments to compensation formulas to accommodate those clinicians who experience a loss of income because they are paid based on production.
The organization emphasizes that, given the rapidly changing availability of testing for COVID-19, practices should contact their local health departments, hospitals, reference labs, or state health authorities to determine the status of their access to testing.
The full list of the ACP’s tips are available here.
Any new guidance for physicians will be posted on the ACP’s COVID-19 resource page.
Clinicians petition government for national quarantine
Clinicians across the United States are petitioning the federal government to follow the lead of South Korea, China, and other nations by imposing an immediate nationwide quarantine to slow the inevitable spread of COVID-19. Without federal action, the creators say, their lives and the lives of their colleagues, patients, and families are being put at increased risk.
In addition to the quarantine, the petition, posted on the website Change.org, calls on U.S. leaders to institute emergency production and distribution of personal protective equipment for healthcare workers and to rapidly increase access to testing.
The petition – which garnered more than 40,000 signatures in just 12 hours and as of this writing was approaching 94,000 – was started by an apolitical Facebook group to focus attention on what members see as the most critical issues for clinicians: slowing the spread of the virus through a coast-to-coast quarantine, protection of medical personnel with adequate supplies of essential equipment, and widespread testing.
“We started this group last Friday out of the realization that clinicians needed information about the outbreak and weren’t getting it,” said coadministrator Jessica McIntyre, MD, a pediatric hospitalist at Elliot Hospital in Manchester, N.H.
“We wanted to get ahead of it and connect with people before we were in the trenches experiencing it and to see what other programs were doing. From a local perspective, it has been really hard to see what people are doing in other states, especially when the protocols in our own states are changing every single day as we collect more information,” she said in an interview.
The Horse Has Bolted
A family medicine physician in Illinois helped launch the Facebook group. She asked that her name not be used but said in an interview that earlier actions may have prevented or at least delayed the need for the more draconian measures that her group is recommending.
“Clearly South Korea is one of the superstars as far as response has gone, but the concern we have in the United States is that we’re well beyond that point – we needed to be testing people over a month ago, in the hope of preventing a quarantine,” she said in an interview.
According to National Public Radio, as of March 13, South Korea had conducted 3,600 tests per million population, compared with five per million in the United States.
“I think the most concerning part is to see where Italy is now and where we are in comparison. Our ICUs have not yet overflowed, but I think we’re definitely looking at that in the next few weeks – hopefully longer, but I suspect that it will happen shortly,” she continued.
She cited work by Harvard University biostatistician Xihong Lin, PhD, that shows that when health authorities in Wuhan, China – widely cited as the epicenter of the global pandemic – cordoned off the city, the infection rate dropped from one person infecting 3.8 others to one infecting 1.25, thereby significantly slowing the rate of transmission.
“This is absolutely what we need to be doing,” she said.
Real News
Within 3 days of its creation, the online group had accrued more than 80,000 members with advanced medical training, including MDs, DOs, physician assistants, nurse practitioners, and certified registered nurse anesthetists.
“A lot of us were already very busy with our day-to-day work outside of COVID-19, and I think a lot of us felt unsure about where to get the best information,” said coadministrator David Janssen, MD, a family medicine physician in group practice in Sioux Center, Iowa,
“If you turn on the TV, there’s a lot of politicizing of the issue, and there’s a lot of good information, but also a lot of bad information. When health care providers talk to other health care providers, that’s often how we get our information and how we learn,” he said in an interview.
The COVID-19 U.S. Physicians/APP Facebook group includes 20 volunteer moderators who handle hundreds of posts per hour from persons seeking information on the novel coronavirus, what to tell patients, and how to protect themselves.
“It’s been wonderful to see how providers have been helping other providers sort through issues. Teaching hospitals have their hands on the latest research, but a lot of people like myself are at small community hospitals, critical-access hospitals, where we may have a lot of questions but don’t necessarily have the answers readily available to us,” Dr. Janssen said.
Dr. Janssen said that his community of about 8,000 residents initially had only four COVID-19 testing kits, or one for every 2,000 people. The situation has since improved, and more tests are now available, he added.
Dr. McIntyre, Dr. Janssen, and the Illinois family physician have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Clinicians across the United States are petitioning the federal government to follow the lead of South Korea, China, and other nations by imposing an immediate nationwide quarantine to slow the inevitable spread of COVID-19. Without federal action, the creators say, their lives and the lives of their colleagues, patients, and families are being put at increased risk.
In addition to the quarantine, the petition, posted on the website Change.org, calls on U.S. leaders to institute emergency production and distribution of personal protective equipment for healthcare workers and to rapidly increase access to testing.
The petition – which garnered more than 40,000 signatures in just 12 hours and as of this writing was approaching 94,000 – was started by an apolitical Facebook group to focus attention on what members see as the most critical issues for clinicians: slowing the spread of the virus through a coast-to-coast quarantine, protection of medical personnel with adequate supplies of essential equipment, and widespread testing.
“We started this group last Friday out of the realization that clinicians needed information about the outbreak and weren’t getting it,” said coadministrator Jessica McIntyre, MD, a pediatric hospitalist at Elliot Hospital in Manchester, N.H.
“We wanted to get ahead of it and connect with people before we were in the trenches experiencing it and to see what other programs were doing. From a local perspective, it has been really hard to see what people are doing in other states, especially when the protocols in our own states are changing every single day as we collect more information,” she said in an interview.
The Horse Has Bolted
A family medicine physician in Illinois helped launch the Facebook group. She asked that her name not be used but said in an interview that earlier actions may have prevented or at least delayed the need for the more draconian measures that her group is recommending.
“Clearly South Korea is one of the superstars as far as response has gone, but the concern we have in the United States is that we’re well beyond that point – we needed to be testing people over a month ago, in the hope of preventing a quarantine,” she said in an interview.
According to National Public Radio, as of March 13, South Korea had conducted 3,600 tests per million population, compared with five per million in the United States.
“I think the most concerning part is to see where Italy is now and where we are in comparison. Our ICUs have not yet overflowed, but I think we’re definitely looking at that in the next few weeks – hopefully longer, but I suspect that it will happen shortly,” she continued.
She cited work by Harvard University biostatistician Xihong Lin, PhD, that shows that when health authorities in Wuhan, China – widely cited as the epicenter of the global pandemic – cordoned off the city, the infection rate dropped from one person infecting 3.8 others to one infecting 1.25, thereby significantly slowing the rate of transmission.
“This is absolutely what we need to be doing,” she said.
Real News
Within 3 days of its creation, the online group had accrued more than 80,000 members with advanced medical training, including MDs, DOs, physician assistants, nurse practitioners, and certified registered nurse anesthetists.
“A lot of us were already very busy with our day-to-day work outside of COVID-19, and I think a lot of us felt unsure about where to get the best information,” said coadministrator David Janssen, MD, a family medicine physician in group practice in Sioux Center, Iowa,
“If you turn on the TV, there’s a lot of politicizing of the issue, and there’s a lot of good information, but also a lot of bad information. When health care providers talk to other health care providers, that’s often how we get our information and how we learn,” he said in an interview.
The COVID-19 U.S. Physicians/APP Facebook group includes 20 volunteer moderators who handle hundreds of posts per hour from persons seeking information on the novel coronavirus, what to tell patients, and how to protect themselves.
“It’s been wonderful to see how providers have been helping other providers sort through issues. Teaching hospitals have their hands on the latest research, but a lot of people like myself are at small community hospitals, critical-access hospitals, where we may have a lot of questions but don’t necessarily have the answers readily available to us,” Dr. Janssen said.
Dr. Janssen said that his community of about 8,000 residents initially had only four COVID-19 testing kits, or one for every 2,000 people. The situation has since improved, and more tests are now available, he added.
Dr. McIntyre, Dr. Janssen, and the Illinois family physician have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Clinicians across the United States are petitioning the federal government to follow the lead of South Korea, China, and other nations by imposing an immediate nationwide quarantine to slow the inevitable spread of COVID-19. Without federal action, the creators say, their lives and the lives of their colleagues, patients, and families are being put at increased risk.
In addition to the quarantine, the petition, posted on the website Change.org, calls on U.S. leaders to institute emergency production and distribution of personal protective equipment for healthcare workers and to rapidly increase access to testing.
The petition – which garnered more than 40,000 signatures in just 12 hours and as of this writing was approaching 94,000 – was started by an apolitical Facebook group to focus attention on what members see as the most critical issues for clinicians: slowing the spread of the virus through a coast-to-coast quarantine, protection of medical personnel with adequate supplies of essential equipment, and widespread testing.
“We started this group last Friday out of the realization that clinicians needed information about the outbreak and weren’t getting it,” said coadministrator Jessica McIntyre, MD, a pediatric hospitalist at Elliot Hospital in Manchester, N.H.
“We wanted to get ahead of it and connect with people before we were in the trenches experiencing it and to see what other programs were doing. From a local perspective, it has been really hard to see what people are doing in other states, especially when the protocols in our own states are changing every single day as we collect more information,” she said in an interview.
The Horse Has Bolted
A family medicine physician in Illinois helped launch the Facebook group. She asked that her name not be used but said in an interview that earlier actions may have prevented or at least delayed the need for the more draconian measures that her group is recommending.
“Clearly South Korea is one of the superstars as far as response has gone, but the concern we have in the United States is that we’re well beyond that point – we needed to be testing people over a month ago, in the hope of preventing a quarantine,” she said in an interview.
According to National Public Radio, as of March 13, South Korea had conducted 3,600 tests per million population, compared with five per million in the United States.
“I think the most concerning part is to see where Italy is now and where we are in comparison. Our ICUs have not yet overflowed, but I think we’re definitely looking at that in the next few weeks – hopefully longer, but I suspect that it will happen shortly,” she continued.
She cited work by Harvard University biostatistician Xihong Lin, PhD, that shows that when health authorities in Wuhan, China – widely cited as the epicenter of the global pandemic – cordoned off the city, the infection rate dropped from one person infecting 3.8 others to one infecting 1.25, thereby significantly slowing the rate of transmission.
“This is absolutely what we need to be doing,” she said.
Real News
Within 3 days of its creation, the online group had accrued more than 80,000 members with advanced medical training, including MDs, DOs, physician assistants, nurse practitioners, and certified registered nurse anesthetists.
“A lot of us were already very busy with our day-to-day work outside of COVID-19, and I think a lot of us felt unsure about where to get the best information,” said coadministrator David Janssen, MD, a family medicine physician in group practice in Sioux Center, Iowa,
“If you turn on the TV, there’s a lot of politicizing of the issue, and there’s a lot of good information, but also a lot of bad information. When health care providers talk to other health care providers, that’s often how we get our information and how we learn,” he said in an interview.
The COVID-19 U.S. Physicians/APP Facebook group includes 20 volunteer moderators who handle hundreds of posts per hour from persons seeking information on the novel coronavirus, what to tell patients, and how to protect themselves.
“It’s been wonderful to see how providers have been helping other providers sort through issues. Teaching hospitals have their hands on the latest research, but a lot of people like myself are at small community hospitals, critical-access hospitals, where we may have a lot of questions but don’t necessarily have the answers readily available to us,” Dr. Janssen said.
Dr. Janssen said that his community of about 8,000 residents initially had only four COVID-19 testing kits, or one for every 2,000 people. The situation has since improved, and more tests are now available, he added.
Dr. McIntyre, Dr. Janssen, and the Illinois family physician have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Coronavirus stays in aerosols for hours, on surfaces for days
according to a new study.
The data indicate that the stability of the new virus is similar to that of SARS-CoV-1, which caused the SARS epidemic, researchers report in an article published on the medRxivpreprint server. (The posted article has been submitted for journal publication but has not been peer reviewed.)
Transmission of SARS-CoV-2, which causes COVID-19, has quickly outstripped the pace of the 2003 SARS epidemic. “Superspread” of the earlier disease arose from infection during medical procedures, in which a single infected individual seeded many secondary cases. In contrast, the novel coronavirus appears to be spread more through human-to-human transmission in a variety of settings.
However, it’s not yet known the extent to which asymptomatic or presymptomatic individuals spread the new virus through daily routine.
To investigate how long SARS-CoV-2 remains infective in the environment, Neeltje van Doremalen, PhD, of the Laboratory of Virology, Division of Intramural Research, National Institute of Allergy and Infectious Diseases, in Hamilton, Montana, and colleagues conducted simulation experiments in which they compared the viability of SARS-CoV-2 with that of SARS-CoV-1 in aerosols and on surfaces.
Among patients infected with SARS-CoV-2, viral loads in the upper respiratory tract are high; as a consequence, respiratory secretion in the form of aerosols (<5 μm) or droplets (>5 mcm) is likely, the authors note.
van Doremalen and colleagues used nebulizers to generate aerosols. Samples of SARS-CoV-1 and SARS-CoV-2 were collecting at 0, 30, 60, 120, and 180 minutes on a gelatin filter. The researchers then tested the infectivity of the viruses on Vero cells grown in culture.
They found that SARS-CoV-2 was largely stable through the full 180-minute test, with only a slight decline at 3 hours. This time course is similar to that of SARS-CoV-1; both viruses have a median half-life in aerosols of 2.7 hours (range, 1.65 hr for SARS-CoV-1, vs 7.24 hr for SARS-CoV-2).
The researchers then tested the viruses on a variety of surfaces for up to 7 days, using humidity values and temperatures designed to mimic “a variety of household and hospital situations.” The volumes of viral exposures that the team used were consistent with amounts found in the human upper and lower respiratory tracts.
For example, they applied 50 mcL of virus-containing solution to a piece of cardboard and then swabbed the surface, at different times, with an additional 1 mcL of medium. Each surface assay was replicated three times.
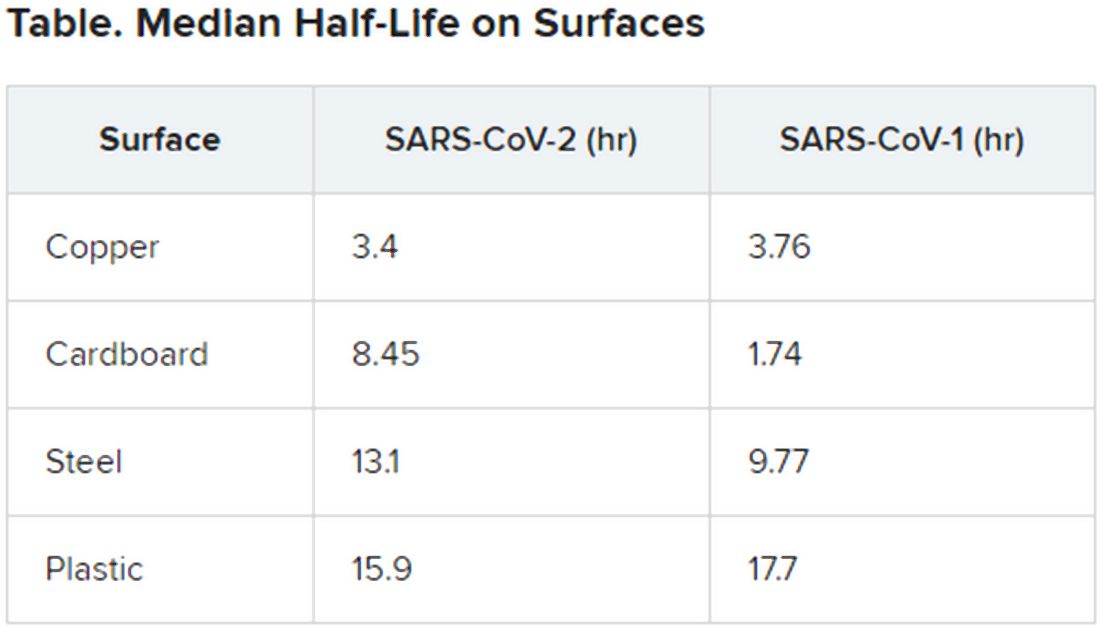
The novel coronavirus was most stable on plastic and stainless steel, with some virus remaining viable up to 72 hours. However, by that time the viral load had fallen by about three orders of magnitude, indicating exponential decay. This profile was remarkably similar to that of SARS-CoV-1, according to the authors.
However, the two viruses differed in staying power on copper and cardboard. No viable SARS-CoV-2 was detectable on copper after 4 hours or on cardboard after 24 hours. In contrast, SARS-CoV-1 was not viable beyond 8 hours for either copper or cardboard.
“Taken together, our results indicate that aerosol and fomite transmission of HCoV-19 [SARS-CoV-2] are plausible, as the virus can remain viable in aerosols for multiple hours and on surfaces up to days,” the authors conclude.
Andrew Pekosz, PhD, codirector of the Center of Excellence in Influenza Research and Surveillance and director of the Center for Emerging Viruses and Infectious Diseases at the Johns Hopkins Center for Global Health, Baltimore, Maryland, applauds the real-world value of the experiments.
“The PCR [polymerase chain reaction] test used [in other studies] to detect SARS-CoV-2 just detects the virus genome. It doesn’t tell you if the virus was still infectious, or ‘viable.’ That’s why this study is interesting,” Pekosz said. “It focuses on infectious virus, which is the virus that has the potential to transmit and infect another person. What we don’t know yet is how much infectious (viable) virus is needed to initiate infection in another person.”
He suggests that further investigations evaluate other types of environmental surfaces, including lacquered wood that is made into desks and ceramic tiles found in bathrooms and kitchens.
One limitation of the study is that the data for experiments on cardboard were more variable than the data for other surfaces tested.
The investigators and Pekosz have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
according to a new study.

The data indicate that the stability of the new virus is similar to that of SARS-CoV-1, which caused the SARS epidemic, researchers report in an article published on the medRxivpreprint server. (The posted article has been submitted for journal publication but has not been peer reviewed.)
Transmission of SARS-CoV-2, which causes COVID-19, has quickly outstripped the pace of the 2003 SARS epidemic. “Superspread” of the earlier disease arose from infection during medical procedures, in which a single infected individual seeded many secondary cases. In contrast, the novel coronavirus appears to be spread more through human-to-human transmission in a variety of settings.
However, it’s not yet known the extent to which asymptomatic or presymptomatic individuals spread the new virus through daily routine.
To investigate how long SARS-CoV-2 remains infective in the environment, Neeltje van Doremalen, PhD, of the Laboratory of Virology, Division of Intramural Research, National Institute of Allergy and Infectious Diseases, in Hamilton, Montana, and colleagues conducted simulation experiments in which they compared the viability of SARS-CoV-2 with that of SARS-CoV-1 in aerosols and on surfaces.
Among patients infected with SARS-CoV-2, viral loads in the upper respiratory tract are high; as a consequence, respiratory secretion in the form of aerosols (<5 μm) or droplets (>5 mcm) is likely, the authors note.
van Doremalen and colleagues used nebulizers to generate aerosols. Samples of SARS-CoV-1 and SARS-CoV-2 were collecting at 0, 30, 60, 120, and 180 minutes on a gelatin filter. The researchers then tested the infectivity of the viruses on Vero cells grown in culture.
They found that SARS-CoV-2 was largely stable through the full 180-minute test, with only a slight decline at 3 hours. This time course is similar to that of SARS-CoV-1; both viruses have a median half-life in aerosols of 2.7 hours (range, 1.65 hr for SARS-CoV-1, vs 7.24 hr for SARS-CoV-2).
The researchers then tested the viruses on a variety of surfaces for up to 7 days, using humidity values and temperatures designed to mimic “a variety of household and hospital situations.” The volumes of viral exposures that the team used were consistent with amounts found in the human upper and lower respiratory tracts.
For example, they applied 50 mcL of virus-containing solution to a piece of cardboard and then swabbed the surface, at different times, with an additional 1 mcL of medium. Each surface assay was replicated three times.
The novel coronavirus was most stable on plastic and stainless steel, with some virus remaining viable up to 72 hours. However, by that time the viral load had fallen by about three orders of magnitude, indicating exponential decay. This profile was remarkably similar to that of SARS-CoV-1, according to the authors.
However, the two viruses differed in staying power on copper and cardboard. No viable SARS-CoV-2 was detectable on copper after 4 hours or on cardboard after 24 hours. In contrast, SARS-CoV-1 was not viable beyond 8 hours for either copper or cardboard.
“Taken together, our results indicate that aerosol and fomite transmission of HCoV-19 [SARS-CoV-2] are plausible, as the virus can remain viable in aerosols for multiple hours and on surfaces up to days,” the authors conclude.
Andrew Pekosz, PhD, codirector of the Center of Excellence in Influenza Research and Surveillance and director of the Center for Emerging Viruses and Infectious Diseases at the Johns Hopkins Center for Global Health, Baltimore, Maryland, applauds the real-world value of the experiments.
“The PCR [polymerase chain reaction] test used [in other studies] to detect SARS-CoV-2 just detects the virus genome. It doesn’t tell you if the virus was still infectious, or ‘viable.’ That’s why this study is interesting,” Pekosz said. “It focuses on infectious virus, which is the virus that has the potential to transmit and infect another person. What we don’t know yet is how much infectious (viable) virus is needed to initiate infection in another person.”
He suggests that further investigations evaluate other types of environmental surfaces, including lacquered wood that is made into desks and ceramic tiles found in bathrooms and kitchens.
One limitation of the study is that the data for experiments on cardboard were more variable than the data for other surfaces tested.
The investigators and Pekosz have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
according to a new study.

The data indicate that the stability of the new virus is similar to that of SARS-CoV-1, which caused the SARS epidemic, researchers report in an article published on the medRxivpreprint server. (The posted article has been submitted for journal publication but has not been peer reviewed.)
Transmission of SARS-CoV-2, which causes COVID-19, has quickly outstripped the pace of the 2003 SARS epidemic. “Superspread” of the earlier disease arose from infection during medical procedures, in which a single infected individual seeded many secondary cases. In contrast, the novel coronavirus appears to be spread more through human-to-human transmission in a variety of settings.
However, it’s not yet known the extent to which asymptomatic or presymptomatic individuals spread the new virus through daily routine.
To investigate how long SARS-CoV-2 remains infective in the environment, Neeltje van Doremalen, PhD, of the Laboratory of Virology, Division of Intramural Research, National Institute of Allergy and Infectious Diseases, in Hamilton, Montana, and colleagues conducted simulation experiments in which they compared the viability of SARS-CoV-2 with that of SARS-CoV-1 in aerosols and on surfaces.
Among patients infected with SARS-CoV-2, viral loads in the upper respiratory tract are high; as a consequence, respiratory secretion in the form of aerosols (<5 μm) or droplets (>5 mcm) is likely, the authors note.
van Doremalen and colleagues used nebulizers to generate aerosols. Samples of SARS-CoV-1 and SARS-CoV-2 were collecting at 0, 30, 60, 120, and 180 minutes on a gelatin filter. The researchers then tested the infectivity of the viruses on Vero cells grown in culture.
They found that SARS-CoV-2 was largely stable through the full 180-minute test, with only a slight decline at 3 hours. This time course is similar to that of SARS-CoV-1; both viruses have a median half-life in aerosols of 2.7 hours (range, 1.65 hr for SARS-CoV-1, vs 7.24 hr for SARS-CoV-2).
The researchers then tested the viruses on a variety of surfaces for up to 7 days, using humidity values and temperatures designed to mimic “a variety of household and hospital situations.” The volumes of viral exposures that the team used were consistent with amounts found in the human upper and lower respiratory tracts.
For example, they applied 50 mcL of virus-containing solution to a piece of cardboard and then swabbed the surface, at different times, with an additional 1 mcL of medium. Each surface assay was replicated three times.
The novel coronavirus was most stable on plastic and stainless steel, with some virus remaining viable up to 72 hours. However, by that time the viral load had fallen by about three orders of magnitude, indicating exponential decay. This profile was remarkably similar to that of SARS-CoV-1, according to the authors.
However, the two viruses differed in staying power on copper and cardboard. No viable SARS-CoV-2 was detectable on copper after 4 hours or on cardboard after 24 hours. In contrast, SARS-CoV-1 was not viable beyond 8 hours for either copper or cardboard.
“Taken together, our results indicate that aerosol and fomite transmission of HCoV-19 [SARS-CoV-2] are plausible, as the virus can remain viable in aerosols for multiple hours and on surfaces up to days,” the authors conclude.
Andrew Pekosz, PhD, codirector of the Center of Excellence in Influenza Research and Surveillance and director of the Center for Emerging Viruses and Infectious Diseases at the Johns Hopkins Center for Global Health, Baltimore, Maryland, applauds the real-world value of the experiments.
“The PCR [polymerase chain reaction] test used [in other studies] to detect SARS-CoV-2 just detects the virus genome. It doesn’t tell you if the virus was still infectious, or ‘viable.’ That’s why this study is interesting,” Pekosz said. “It focuses on infectious virus, which is the virus that has the potential to transmit and infect another person. What we don’t know yet is how much infectious (viable) virus is needed to initiate infection in another person.”
He suggests that further investigations evaluate other types of environmental surfaces, including lacquered wood that is made into desks and ceramic tiles found in bathrooms and kitchens.
One limitation of the study is that the data for experiments on cardboard were more variable than the data for other surfaces tested.
The investigators and Pekosz have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Flattening the curve: Viral graphic shows COVID-19 containment needs
Editor’s note: Find the latest COVID-19 news and guidance in Medscape’s Coronavirus Resource Center.
The “Flattening the Curve” graphic, which has, to not use the term lightly, gone viral on social media, visually explains the best currently available strategy to stop the COVID-19 spread, experts told Medscape Medical News.
The height of the curve is the number of potential cases in the United States; along the horizontal X axis, or the breadth, is the amount of time. The line across the middle represents the point at which too many cases in too short a time overwhelm the healthcare system.
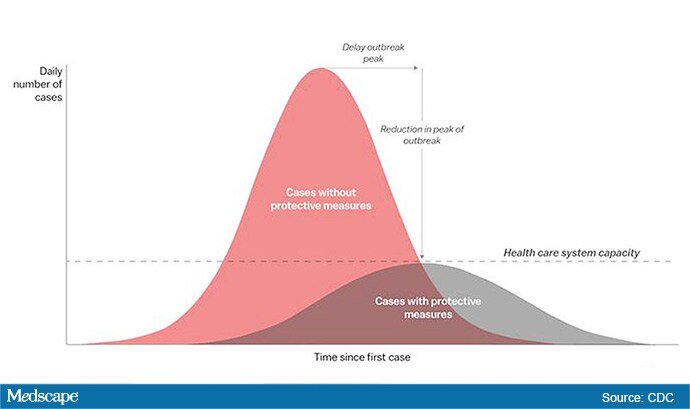
Jeanne Marrazzo, MD, MPH, director of the Division of Infectious Diseases at the University of Alabama at Birmingham’s School of Medicine explained.
“Not only are you spreading out the new cases but the rate at which people recover,” she told Medscape Medical News. “You have time to get people out of the hospital so you can get new people in and clear out those beds.”
The strategy, with its own Twitter hashtag, #Flattenthecurve, “is about all we have,” without a vaccine, Marrazzo said.
Anthony Fauci, director of the National Institute of Allergy and Infectious Diseases, said avoiding spikes in cases could mean fewer deaths.
“If you look at the curves of outbreaks, you know, they go big peaks, and then they come down. What we need to do is flatten that down,” Fauci said March 10 in a White House briefing. “You do that by trying to interfere with the natural flow of the outbreak.”
Wuhan, China, at the epicenter of the pandemic, “had an explosive curve” and quickly got overwhelmed without early containment measures, Marrazzo noted. “If you look at Italy right now, it’s clearly in the same situation.”
The Race Is On to Interrupt the Spread
The race is on in the US to interrupt the transmission of the virus and slow the spread, meaning containment measures have increasingly higher and wider stakes.
Closing down Broadway shows and some theme parks and massive sporting events; the escalating numbers of people working from home; and businesses cutting hours or closing all demonstrate the level of US confidence that “social distancing” will work, Marrazzo said.
“We’re clearly ready to disrupt the economy and social infrastructure,” she said.
That appears to have made a difference in Wuhan, Marrazzo said, as the new infections are coming down.
The question, she said, is “we’re not China – so are Americans really going to take to this? Americans greatly value their liberty and there’s some skepticism about public health and its directives. People have never seen a pandemic like this before.”
Dena Grayson, MD, PhD, a Florida-based expert in Ebola and other pandemic threats, told Medscape Medical News that EvergreenHealth in Kirkland, Washington, is a good example of what it means when a virus overwhelms healthcare operations.
The New York Times reported that supplies were so strained at the facility that staff were using sanitary napkins to pad protective helmets.
As of March 11, 65 people who had come into the hospital have tested positive for the virus, and 15 of them had died.
Grayson points out that the COVID-19 cases come on top of a severe flu season and the usual cases hospitals see, so the bar on the graphic is even lower than it usually would be.
“We have a relatively limited capacity with ICU beds to begin with,” she said.
So far, closures, postponements, and cancellations are woefully inadequate, Grayson said.
“We can’t stop this virus. We can hope to contain it and slow down the rate of infection,” she said.
“We need to right now shut down all the schools, preschools, and universities,” Grayson said. “We need to look at shutting down public transportation. We need people to stay home – and not for a day but for a couple of weeks.”
The graphic was developed by visual-data journalist Rosamund Pearce, based on a graphic that had appeared in a Centers for Disease Control and Prevention (CDC) article titled “Community Mitigation Guidelines to Prevent Pandemic Influenza,” the Times reports.
Marrazzo and Grayson have disclosed no relevant financial relationships.
This story first appeared on Medscape.com .
Editor’s note: Find the latest COVID-19 news and guidance in Medscape’s Coronavirus Resource Center.
The “Flattening the Curve” graphic, which has, to not use the term lightly, gone viral on social media, visually explains the best currently available strategy to stop the COVID-19 spread, experts told Medscape Medical News.
The height of the curve is the number of potential cases in the United States; along the horizontal X axis, or the breadth, is the amount of time. The line across the middle represents the point at which too many cases in too short a time overwhelm the healthcare system.

Jeanne Marrazzo, MD, MPH, director of the Division of Infectious Diseases at the University of Alabama at Birmingham’s School of Medicine explained.
“Not only are you spreading out the new cases but the rate at which people recover,” she told Medscape Medical News. “You have time to get people out of the hospital so you can get new people in and clear out those beds.”
The strategy, with its own Twitter hashtag, #Flattenthecurve, “is about all we have,” without a vaccine, Marrazzo said.
Anthony Fauci, director of the National Institute of Allergy and Infectious Diseases, said avoiding spikes in cases could mean fewer deaths.
“If you look at the curves of outbreaks, you know, they go big peaks, and then they come down. What we need to do is flatten that down,” Fauci said March 10 in a White House briefing. “You do that by trying to interfere with the natural flow of the outbreak.”
Wuhan, China, at the epicenter of the pandemic, “had an explosive curve” and quickly got overwhelmed without early containment measures, Marrazzo noted. “If you look at Italy right now, it’s clearly in the same situation.”
The Race Is On to Interrupt the Spread
The race is on in the US to interrupt the transmission of the virus and slow the spread, meaning containment measures have increasingly higher and wider stakes.
Closing down Broadway shows and some theme parks and massive sporting events; the escalating numbers of people working from home; and businesses cutting hours or closing all demonstrate the level of US confidence that “social distancing” will work, Marrazzo said.
“We’re clearly ready to disrupt the economy and social infrastructure,” she said.
That appears to have made a difference in Wuhan, Marrazzo said, as the new infections are coming down.
The question, she said, is “we’re not China – so are Americans really going to take to this? Americans greatly value their liberty and there’s some skepticism about public health and its directives. People have never seen a pandemic like this before.”
Dena Grayson, MD, PhD, a Florida-based expert in Ebola and other pandemic threats, told Medscape Medical News that EvergreenHealth in Kirkland, Washington, is a good example of what it means when a virus overwhelms healthcare operations.
The New York Times reported that supplies were so strained at the facility that staff were using sanitary napkins to pad protective helmets.
As of March 11, 65 people who had come into the hospital have tested positive for the virus, and 15 of them had died.
Grayson points out that the COVID-19 cases come on top of a severe flu season and the usual cases hospitals see, so the bar on the graphic is even lower than it usually would be.
“We have a relatively limited capacity with ICU beds to begin with,” she said.
So far, closures, postponements, and cancellations are woefully inadequate, Grayson said.
“We can’t stop this virus. We can hope to contain it and slow down the rate of infection,” she said.
“We need to right now shut down all the schools, preschools, and universities,” Grayson said. “We need to look at shutting down public transportation. We need people to stay home – and not for a day but for a couple of weeks.”
The graphic was developed by visual-data journalist Rosamund Pearce, based on a graphic that had appeared in a Centers for Disease Control and Prevention (CDC) article titled “Community Mitigation Guidelines to Prevent Pandemic Influenza,” the Times reports.
Marrazzo and Grayson have disclosed no relevant financial relationships.
This story first appeared on Medscape.com .
Editor’s note: Find the latest COVID-19 news and guidance in Medscape’s Coronavirus Resource Center.
The “Flattening the Curve” graphic, which has, to not use the term lightly, gone viral on social media, visually explains the best currently available strategy to stop the COVID-19 spread, experts told Medscape Medical News.
The height of the curve is the number of potential cases in the United States; along the horizontal X axis, or the breadth, is the amount of time. The line across the middle represents the point at which too many cases in too short a time overwhelm the healthcare system.

Jeanne Marrazzo, MD, MPH, director of the Division of Infectious Diseases at the University of Alabama at Birmingham’s School of Medicine explained.
“Not only are you spreading out the new cases but the rate at which people recover,” she told Medscape Medical News. “You have time to get people out of the hospital so you can get new people in and clear out those beds.”
The strategy, with its own Twitter hashtag, #Flattenthecurve, “is about all we have,” without a vaccine, Marrazzo said.
Anthony Fauci, director of the National Institute of Allergy and Infectious Diseases, said avoiding spikes in cases could mean fewer deaths.
“If you look at the curves of outbreaks, you know, they go big peaks, and then they come down. What we need to do is flatten that down,” Fauci said March 10 in a White House briefing. “You do that by trying to interfere with the natural flow of the outbreak.”
Wuhan, China, at the epicenter of the pandemic, “had an explosive curve” and quickly got overwhelmed without early containment measures, Marrazzo noted. “If you look at Italy right now, it’s clearly in the same situation.”
The Race Is On to Interrupt the Spread
The race is on in the US to interrupt the transmission of the virus and slow the spread, meaning containment measures have increasingly higher and wider stakes.
Closing down Broadway shows and some theme parks and massive sporting events; the escalating numbers of people working from home; and businesses cutting hours or closing all demonstrate the level of US confidence that “social distancing” will work, Marrazzo said.
“We’re clearly ready to disrupt the economy and social infrastructure,” she said.
That appears to have made a difference in Wuhan, Marrazzo said, as the new infections are coming down.
The question, she said, is “we’re not China – so are Americans really going to take to this? Americans greatly value their liberty and there’s some skepticism about public health and its directives. People have never seen a pandemic like this before.”
Dena Grayson, MD, PhD, a Florida-based expert in Ebola and other pandemic threats, told Medscape Medical News that EvergreenHealth in Kirkland, Washington, is a good example of what it means when a virus overwhelms healthcare operations.
The New York Times reported that supplies were so strained at the facility that staff were using sanitary napkins to pad protective helmets.
As of March 11, 65 people who had come into the hospital have tested positive for the virus, and 15 of them had died.
Grayson points out that the COVID-19 cases come on top of a severe flu season and the usual cases hospitals see, so the bar on the graphic is even lower than it usually would be.
“We have a relatively limited capacity with ICU beds to begin with,” she said.
So far, closures, postponements, and cancellations are woefully inadequate, Grayson said.
“We can’t stop this virus. We can hope to contain it and slow down the rate of infection,” she said.
“We need to right now shut down all the schools, preschools, and universities,” Grayson said. “We need to look at shutting down public transportation. We need people to stay home – and not for a day but for a couple of weeks.”
The graphic was developed by visual-data journalist Rosamund Pearce, based on a graphic that had appeared in a Centers for Disease Control and Prevention (CDC) article titled “Community Mitigation Guidelines to Prevent Pandemic Influenza,” the Times reports.
Marrazzo and Grayson have disclosed no relevant financial relationships.
This story first appeared on Medscape.com .
So you have a COVID-19 patient: How do you treat them?
Editor’s note: Find the latest COVID-19 news and guidance in Medscape’s Coronavirus Resource Center.
Clinicians are working out how to manage patients with or suspected of having COVID-19.
“Over the past couple of weeks, we’ve been preparing for the oncoming onslaught of patients,” said Lillian Wu, MD, of the HealthPoint network in the Seattle area of greater King County and president elect of the Washington Academy of Family Physicians.
Step One: Triage
The first step, Wu says, is careful triage.
When patients call one of the 17 clinics in the HealthPoint system, nurses gauge how sick they are. High fever? Shortness of breath? Do they have a chronic illness, such as diabetes, cardiovascular disease, or a lung condition, that increases risk for infection and complications?
“If a patient has mild symptoms, we ask them to stay home or to check back in 24 hours, or we’ll reach out to them. For moderate symptoms, we ask them to come in, and [we] clearly mark on the schedule that it is a respiratory patient, who will be sent to a separate area. If the patient is severe, we don’t even see them and send them directly to the hospital to the ER,” Wu told Medscape Medical News.
These categories parallel the World Health Organization’s designations of uncomplicated illness, mild pneumonia, severe pneumonia, acute respiratory distress syndrome, sepsis, and septic shock. The Centers for Disease Control and Prevention (CDC) advises case by case regarding decisions as to outpatient or inpatient assignment.
“Patients who pass the initial phone triage are given masks, separated, and sent to different parts of the clinic or are required to wait in their cars until it’s time to be seen,” Wu said.
Step 2: Hospital Arrival
Once at the hospital, the CDC’s interim guidance kicks in.
“Any patient with fever, cough, and shortness of breath presenting with a history of travel to countries with high ongoing transmission or a credible history of exposure should be promptly evaluated for COVID-19,” said Raghavendra Tirupathi, MD, medical director, Keystone Infectious Diseases/HIV; chair in infection prevention, Summit Health; and clinical assistant professor of medicine, Penn State School of Medicine, Hershey, Pennsylvania.
“We recommend obtaining baseline CBC with differential, basic metabolic panel, liver function tests, and procalcitonin. Clues for COVID-19 include leukopenia, seen in 30% to 45% of patients, and lymphocytopenia, seen in 85% of the patients in the case series from China,” Tirupathi said. He uses a respiratory virus polymerase chain reaction panel to rule out other pathogens.
Wu concurs. “This is the one time we are grateful when someone tests positive for the flu! If flu is negative and other common respiratory infections are negative, then we do a COVID-19 test,” she said.
But test results may be delayed. “At the University of Washington, it takes 8 hours, but commercial labs take up to 4 days,” Wu said. All patients with respiratory symptoms are treated as persons under investigation, for whom isolation precautions are required. In addition, for these patients, use of personal protective equipment by caregivers is required.
For suspected pneumonia, the American College of Radiography recommends chest CT to identify peripheral basal ground-glass opacities characteristic of COVID-19.
However, diagnosis should be based on detection of SARS-CoV-2, because chest images for COVID-19 are nonspecific – associated signs can also be seen in H1N1 influenza, SARS, and MERS.
Step 3: Supportive Care
Once a patient is admitted, supportive care entails “maintaining fluid status and nutrition and supporting physiological functions until we heal. It’s treating complications and organ support, whether that means providing supplementary oxygen all the way to ventilator support, and just waiting it out. If a patient progresses to acute respiratory distress syndrome, it becomes tougher,” said David Liebers, MD, chief medical officer and an infectious disease specialist at Ellis Medicine in Schenectady, New York.
Efforts are ramping up to develop therapeutics. Remdesivir, an investigational antiviral drug developed to treat Ebola and Marburg hemorrhagic fevers, shows activity against SARS-CoV-2 in vitro.
Remdesivir has been used in a few patients on a compassionate-use basis outside of a clinical trial setting. “It’s a nucleotide analogue, and like other drugs of that class, it disrupts nucleic acid production. Some data suggest that it might have some efficacy,” Liebers said.
Antibiotics are reserved for patients suspected of having concomitant bacterial or fungal infections. Liebers said clinicians should be alerted to “the big three” signs of secondary infection – fever, elevated white blood cell count, and lactic acidosis. Immunosuppressed patients are at elevated risk for secondary infection.
Step 4: Managing Complications
Patients do die of COVID-19, mostly through an inability to ventilate, even when supported with oxygen, Liebers told Medscape Medical News. (According to Tirupathi, “The studies from China indicate that from 6%-10% of patients needed ventilators.”)
Liebers continued, “Others may develop sepsis or a syndrome of multisystem organ failure with renal and endothelial collapse, making it difficult to maintain blood pressure. Like with so many pathologies, it is a vicious circle in which everything gets overworked. Off-and-on treatments can sometimes break the cycle: supplementary oxygen, giving red blood cells, dialysis. We support those functions while waiting for healing to occur.”
A facility’s airborne-infection isolation rooms may become filled to capacity, but that isn’t critical, Liebers said. “Airborne precautions are standard to contain measles, tuberculosis, chickenpox, and herpes zoster, in which very small particles spread in the air,” he said.
Consensus is growing that SARS-CoV-2 spreads in large droplets, he added. Private rooms and closed doors may suffice.
Step 5: Discharge
Liebers said that as of now, the million-dollar question regards criteria for discharge.
Patients who clinically improve are sent home with instructions to remain in isolation. They may be tested again for virus before or after discharge.
Liebers and Wu pointed to the experience at EvergreenHealth Medical Center, in Kirkland, Washington, as guidance from the trenches. “They’re the ones who are learning firsthand and passing the experience along to everyone else,” Wu said.
“The situation is unprecedented,” said Liebers, who, like many others, has barely slept these past weeks. “We’re swimming in murky water right now.”
The epidemic in the United States is still months from peaking, Wu emphasized. “There is no vaccine, and many cases are subclinical. COVID-19 has to spread through the country before it infects a critical mass of people who will develop immunity. It’s too late to contain.”
Added Liebers, “It’s a constantly changing situation, and we are still being surprised – not that this wasn’t predicted.”
This article first appeared on Medscape.com.
Editor’s note: Find the latest COVID-19 news and guidance in Medscape’s Coronavirus Resource Center.
Clinicians are working out how to manage patients with or suspected of having COVID-19.
“Over the past couple of weeks, we’ve been preparing for the oncoming onslaught of patients,” said Lillian Wu, MD, of the HealthPoint network in the Seattle area of greater King County and president elect of the Washington Academy of Family Physicians.
Step One: Triage
The first step, Wu says, is careful triage.
When patients call one of the 17 clinics in the HealthPoint system, nurses gauge how sick they are. High fever? Shortness of breath? Do they have a chronic illness, such as diabetes, cardiovascular disease, or a lung condition, that increases risk for infection and complications?
“If a patient has mild symptoms, we ask them to stay home or to check back in 24 hours, or we’ll reach out to them. For moderate symptoms, we ask them to come in, and [we] clearly mark on the schedule that it is a respiratory patient, who will be sent to a separate area. If the patient is severe, we don’t even see them and send them directly to the hospital to the ER,” Wu told Medscape Medical News.
These categories parallel the World Health Organization’s designations of uncomplicated illness, mild pneumonia, severe pneumonia, acute respiratory distress syndrome, sepsis, and septic shock. The Centers for Disease Control and Prevention (CDC) advises case by case regarding decisions as to outpatient or inpatient assignment.
“Patients who pass the initial phone triage are given masks, separated, and sent to different parts of the clinic or are required to wait in their cars until it’s time to be seen,” Wu said.
Step 2: Hospital Arrival
Once at the hospital, the CDC’s interim guidance kicks in.
“Any patient with fever, cough, and shortness of breath presenting with a history of travel to countries with high ongoing transmission or a credible history of exposure should be promptly evaluated for COVID-19,” said Raghavendra Tirupathi, MD, medical director, Keystone Infectious Diseases/HIV; chair in infection prevention, Summit Health; and clinical assistant professor of medicine, Penn State School of Medicine, Hershey, Pennsylvania.
“We recommend obtaining baseline CBC with differential, basic metabolic panel, liver function tests, and procalcitonin. Clues for COVID-19 include leukopenia, seen in 30% to 45% of patients, and lymphocytopenia, seen in 85% of the patients in the case series from China,” Tirupathi said. He uses a respiratory virus polymerase chain reaction panel to rule out other pathogens.
Wu concurs. “This is the one time we are grateful when someone tests positive for the flu! If flu is negative and other common respiratory infections are negative, then we do a COVID-19 test,” she said.
But test results may be delayed. “At the University of Washington, it takes 8 hours, but commercial labs take up to 4 days,” Wu said. All patients with respiratory symptoms are treated as persons under investigation, for whom isolation precautions are required. In addition, for these patients, use of personal protective equipment by caregivers is required.
For suspected pneumonia, the American College of Radiography recommends chest CT to identify peripheral basal ground-glass opacities characteristic of COVID-19.
However, diagnosis should be based on detection of SARS-CoV-2, because chest images for COVID-19 are nonspecific – associated signs can also be seen in H1N1 influenza, SARS, and MERS.
Step 3: Supportive Care
Once a patient is admitted, supportive care entails “maintaining fluid status and nutrition and supporting physiological functions until we heal. It’s treating complications and organ support, whether that means providing supplementary oxygen all the way to ventilator support, and just waiting it out. If a patient progresses to acute respiratory distress syndrome, it becomes tougher,” said David Liebers, MD, chief medical officer and an infectious disease specialist at Ellis Medicine in Schenectady, New York.
Efforts are ramping up to develop therapeutics. Remdesivir, an investigational antiviral drug developed to treat Ebola and Marburg hemorrhagic fevers, shows activity against SARS-CoV-2 in vitro.
Remdesivir has been used in a few patients on a compassionate-use basis outside of a clinical trial setting. “It’s a nucleotide analogue, and like other drugs of that class, it disrupts nucleic acid production. Some data suggest that it might have some efficacy,” Liebers said.
Antibiotics are reserved for patients suspected of having concomitant bacterial or fungal infections. Liebers said clinicians should be alerted to “the big three” signs of secondary infection – fever, elevated white blood cell count, and lactic acidosis. Immunosuppressed patients are at elevated risk for secondary infection.
Step 4: Managing Complications
Patients do die of COVID-19, mostly through an inability to ventilate, even when supported with oxygen, Liebers told Medscape Medical News. (According to Tirupathi, “The studies from China indicate that from 6%-10% of patients needed ventilators.”)
Liebers continued, “Others may develop sepsis or a syndrome of multisystem organ failure with renal and endothelial collapse, making it difficult to maintain blood pressure. Like with so many pathologies, it is a vicious circle in which everything gets overworked. Off-and-on treatments can sometimes break the cycle: supplementary oxygen, giving red blood cells, dialysis. We support those functions while waiting for healing to occur.”
A facility’s airborne-infection isolation rooms may become filled to capacity, but that isn’t critical, Liebers said. “Airborne precautions are standard to contain measles, tuberculosis, chickenpox, and herpes zoster, in which very small particles spread in the air,” he said.
Consensus is growing that SARS-CoV-2 spreads in large droplets, he added. Private rooms and closed doors may suffice.
Step 5: Discharge
Liebers said that as of now, the million-dollar question regards criteria for discharge.
Patients who clinically improve are sent home with instructions to remain in isolation. They may be tested again for virus before or after discharge.
Liebers and Wu pointed to the experience at EvergreenHealth Medical Center, in Kirkland, Washington, as guidance from the trenches. “They’re the ones who are learning firsthand and passing the experience along to everyone else,” Wu said.
“The situation is unprecedented,” said Liebers, who, like many others, has barely slept these past weeks. “We’re swimming in murky water right now.”
The epidemic in the United States is still months from peaking, Wu emphasized. “There is no vaccine, and many cases are subclinical. COVID-19 has to spread through the country before it infects a critical mass of people who will develop immunity. It’s too late to contain.”
Added Liebers, “It’s a constantly changing situation, and we are still being surprised – not that this wasn’t predicted.”
This article first appeared on Medscape.com.
Editor’s note: Find the latest COVID-19 news and guidance in Medscape’s Coronavirus Resource Center.
Clinicians are working out how to manage patients with or suspected of having COVID-19.
“Over the past couple of weeks, we’ve been preparing for the oncoming onslaught of patients,” said Lillian Wu, MD, of the HealthPoint network in the Seattle area of greater King County and president elect of the Washington Academy of Family Physicians.
Step One: Triage
The first step, Wu says, is careful triage.
When patients call one of the 17 clinics in the HealthPoint system, nurses gauge how sick they are. High fever? Shortness of breath? Do they have a chronic illness, such as diabetes, cardiovascular disease, or a lung condition, that increases risk for infection and complications?
“If a patient has mild symptoms, we ask them to stay home or to check back in 24 hours, or we’ll reach out to them. For moderate symptoms, we ask them to come in, and [we] clearly mark on the schedule that it is a respiratory patient, who will be sent to a separate area. If the patient is severe, we don’t even see them and send them directly to the hospital to the ER,” Wu told Medscape Medical News.
These categories parallel the World Health Organization’s designations of uncomplicated illness, mild pneumonia, severe pneumonia, acute respiratory distress syndrome, sepsis, and septic shock. The Centers for Disease Control and Prevention (CDC) advises case by case regarding decisions as to outpatient or inpatient assignment.
“Patients who pass the initial phone triage are given masks, separated, and sent to different parts of the clinic or are required to wait in their cars until it’s time to be seen,” Wu said.
Step 2: Hospital Arrival
Once at the hospital, the CDC’s interim guidance kicks in.
“Any patient with fever, cough, and shortness of breath presenting with a history of travel to countries with high ongoing transmission or a credible history of exposure should be promptly evaluated for COVID-19,” said Raghavendra Tirupathi, MD, medical director, Keystone Infectious Diseases/HIV; chair in infection prevention, Summit Health; and clinical assistant professor of medicine, Penn State School of Medicine, Hershey, Pennsylvania.
“We recommend obtaining baseline CBC with differential, basic metabolic panel, liver function tests, and procalcitonin. Clues for COVID-19 include leukopenia, seen in 30% to 45% of patients, and lymphocytopenia, seen in 85% of the patients in the case series from China,” Tirupathi said. He uses a respiratory virus polymerase chain reaction panel to rule out other pathogens.
Wu concurs. “This is the one time we are grateful when someone tests positive for the flu! If flu is negative and other common respiratory infections are negative, then we do a COVID-19 test,” she said.
But test results may be delayed. “At the University of Washington, it takes 8 hours, but commercial labs take up to 4 days,” Wu said. All patients with respiratory symptoms are treated as persons under investigation, for whom isolation precautions are required. In addition, for these patients, use of personal protective equipment by caregivers is required.
For suspected pneumonia, the American College of Radiography recommends chest CT to identify peripheral basal ground-glass opacities characteristic of COVID-19.
However, diagnosis should be based on detection of SARS-CoV-2, because chest images for COVID-19 are nonspecific – associated signs can also be seen in H1N1 influenza, SARS, and MERS.
Step 3: Supportive Care
Once a patient is admitted, supportive care entails “maintaining fluid status and nutrition and supporting physiological functions until we heal. It’s treating complications and organ support, whether that means providing supplementary oxygen all the way to ventilator support, and just waiting it out. If a patient progresses to acute respiratory distress syndrome, it becomes tougher,” said David Liebers, MD, chief medical officer and an infectious disease specialist at Ellis Medicine in Schenectady, New York.
Efforts are ramping up to develop therapeutics. Remdesivir, an investigational antiviral drug developed to treat Ebola and Marburg hemorrhagic fevers, shows activity against SARS-CoV-2 in vitro.
Remdesivir has been used in a few patients on a compassionate-use basis outside of a clinical trial setting. “It’s a nucleotide analogue, and like other drugs of that class, it disrupts nucleic acid production. Some data suggest that it might have some efficacy,” Liebers said.
Antibiotics are reserved for patients suspected of having concomitant bacterial or fungal infections. Liebers said clinicians should be alerted to “the big three” signs of secondary infection – fever, elevated white blood cell count, and lactic acidosis. Immunosuppressed patients are at elevated risk for secondary infection.
Step 4: Managing Complications
Patients do die of COVID-19, mostly through an inability to ventilate, even when supported with oxygen, Liebers told Medscape Medical News. (According to Tirupathi, “The studies from China indicate that from 6%-10% of patients needed ventilators.”)
Liebers continued, “Others may develop sepsis or a syndrome of multisystem organ failure with renal and endothelial collapse, making it difficult to maintain blood pressure. Like with so many pathologies, it is a vicious circle in which everything gets overworked. Off-and-on treatments can sometimes break the cycle: supplementary oxygen, giving red blood cells, dialysis. We support those functions while waiting for healing to occur.”
A facility’s airborne-infection isolation rooms may become filled to capacity, but that isn’t critical, Liebers said. “Airborne precautions are standard to contain measles, tuberculosis, chickenpox, and herpes zoster, in which very small particles spread in the air,” he said.
Consensus is growing that SARS-CoV-2 spreads in large droplets, he added. Private rooms and closed doors may suffice.
Step 5: Discharge
Liebers said that as of now, the million-dollar question regards criteria for discharge.
Patients who clinically improve are sent home with instructions to remain in isolation. They may be tested again for virus before or after discharge.
Liebers and Wu pointed to the experience at EvergreenHealth Medical Center, in Kirkland, Washington, as guidance from the trenches. “They’re the ones who are learning firsthand and passing the experience along to everyone else,” Wu said.
“The situation is unprecedented,” said Liebers, who, like many others, has barely slept these past weeks. “We’re swimming in murky water right now.”
The epidemic in the United States is still months from peaking, Wu emphasized. “There is no vaccine, and many cases are subclinical. COVID-19 has to spread through the country before it infects a critical mass of people who will develop immunity. It’s too late to contain.”
Added Liebers, “It’s a constantly changing situation, and we are still being surprised – not that this wasn’t predicted.”
This article first appeared on Medscape.com.
Review highlights shortage of data on elderly cancer patients
Phase 3 clinical trials for cancer are underreporting safety and efficacy data for elderly patients, according to a systematic review of 159 articles.
Roughly 40% of articles reporting efficacy data and 9% of articles reporting safety data had results stratified by age, Karlynn BrintzenhofeSzoc, PhD, of the University of Cincinnati, and colleagues noted in the Journal of Geriatric Oncology.
“Results of our systematic review suggest that there is inadequate reporting of treatment efficacy and adverse events as well as discrepancies as to how older age is defined, considered, and reported,” the investigators wrote. “This sparse and varied reporting critically limits the evidence base for treating older patients with cancer.”
This study was inspired by the American Society of Clinical Oncology, which turned a spotlight on the age-specific data shortage in 2015, when it published a statement that called for inclusion of more elderly patients in cancer trials (J Clin Oncol. 2015 Nov 10;33[32]:3826-33).
According to Tammy Hshieh, MD, a geriatrician at Dana-Farber Cancer Institute in Boston, data for elderly patients with cancer are needed more than ever.
“Cancer care has become, increasingly, a field where precision medicine is at its strongest,” Dr. Hshieh said in an interview. “[Oncologists] have a lot of data on patients that allow them to tailor their care to each individual patient’s profile, and so the fact that there is not a lot of evidence looking at toxicities and side effects for older patients makes it basically harder for oncologists to practice evidence-based medicine for this vulnerable but growing population.” This leads to poorer and more variable outcomes, Dr. Hshieh said. When data aren’t available, clinicians must rely on experience and recognize that patient age isn’t as simple as date of birth.
“Oncologists looking at older patients really have to trust their gestalt and their experience in determining how to provide the best care for their older patients,” she said. “They have to look at the chronological age of the patient and try to determine whether that actually matches more of what we’re saying is the physiological age of the patient and use that to guide their treatment.”
Study details
The study included phase 3 clinical trials of adult cancer patients that were conducted from mid-2016 through mid-2017. After identifying 929 manuscripts, the investigators removed duplicates and those that did not meet criteria. This left 159 articles published in 36 journals and covering 25 cancer types.
Of the 159 articles, 73.6% included age-specific medians (in addition to age means), and 47.2% had data stratified by age.
Efficacy was often reported (96.2%), but only 39.9% of articles specified age when describing effectiveness. Although most articles (84.9%) included safety data, only 8.9% had safety findings stratified by age.
In article discussion sections, age was mentioned infrequently in relation to treatment efficacy (13.8%) and rarely in relation to complications and adverse events (5.7%).Beyond underreporting of age-specific data, the investigators found that age categories themselves may be an area in need of improvement.“When outcomes pertaining to older adults were reported, the results were inconsistent as evidenced by the array of age distributions and varying categorization of ‘older adults,’” the investigators wrote. “There is a significant and timely need to design all clinical trials to include older adults and utilize a broad array of geriatric-specific outcomes.” Dr. Hshieh said these findings are concerning, but the study itself suggests the medical community is making efforts to correct the data shortage.“It was actually an important study, even though the results are a little discouraging,” Dr. Hshieh said. “What I’m hoping is that [these findings], combined with all the other literature that’s starting to come out about the need for more research in older patients with cancer, is going to be an impetus for us to do more research, and to be more open to treating older patients, and not to be afraid to confront this head on.”When asked about strategies for managing elderly patients, Dr. Hshieh first recommended the 2018 ASCO Guideline for Geriatric Oncology (J Clin Oncol. 2018 Aug 1;36[22]:2326-47).
“It’s very well written,” she said. “It is clear and user-friendly.”
Dr. Hshieh also offered some simple principles that may help guide clinical decision making.“I’m thinking of three things that an oncologist in the community would want to look at when they see an older patient and they’re trying to determine their treatment plan,” she said. “I would say [the oncologist] should look at [the patient’s] function; their psychosocial status, which includes mood and the support that they have in the community; and cognition.”
Dr. Hshieh and the study authors reported no conflicts of interest.
SOURCE: BrintzenhofeSzoc K et al. J Geriatr Oncol. 2020 Jan 10. pii: S1879-4068(19)30501-6.
Phase 3 clinical trials for cancer are underreporting safety and efficacy data for elderly patients, according to a systematic review of 159 articles.
Roughly 40% of articles reporting efficacy data and 9% of articles reporting safety data had results stratified by age, Karlynn BrintzenhofeSzoc, PhD, of the University of Cincinnati, and colleagues noted in the Journal of Geriatric Oncology.
“Results of our systematic review suggest that there is inadequate reporting of treatment efficacy and adverse events as well as discrepancies as to how older age is defined, considered, and reported,” the investigators wrote. “This sparse and varied reporting critically limits the evidence base for treating older patients with cancer.”
This study was inspired by the American Society of Clinical Oncology, which turned a spotlight on the age-specific data shortage in 2015, when it published a statement that called for inclusion of more elderly patients in cancer trials (J Clin Oncol. 2015 Nov 10;33[32]:3826-33).
According to Tammy Hshieh, MD, a geriatrician at Dana-Farber Cancer Institute in Boston, data for elderly patients with cancer are needed more than ever.
“Cancer care has become, increasingly, a field where precision medicine is at its strongest,” Dr. Hshieh said in an interview. “[Oncologists] have a lot of data on patients that allow them to tailor their care to each individual patient’s profile, and so the fact that there is not a lot of evidence looking at toxicities and side effects for older patients makes it basically harder for oncologists to practice evidence-based medicine for this vulnerable but growing population.” This leads to poorer and more variable outcomes, Dr. Hshieh said. When data aren’t available, clinicians must rely on experience and recognize that patient age isn’t as simple as date of birth.
“Oncologists looking at older patients really have to trust their gestalt and their experience in determining how to provide the best care for their older patients,” she said. “They have to look at the chronological age of the patient and try to determine whether that actually matches more of what we’re saying is the physiological age of the patient and use that to guide their treatment.”
Study details
The study included phase 3 clinical trials of adult cancer patients that were conducted from mid-2016 through mid-2017. After identifying 929 manuscripts, the investigators removed duplicates and those that did not meet criteria. This left 159 articles published in 36 journals and covering 25 cancer types.
Of the 159 articles, 73.6% included age-specific medians (in addition to age means), and 47.2% had data stratified by age.
Efficacy was often reported (96.2%), but only 39.9% of articles specified age when describing effectiveness. Although most articles (84.9%) included safety data, only 8.9% had safety findings stratified by age.
In article discussion sections, age was mentioned infrequently in relation to treatment efficacy (13.8%) and rarely in relation to complications and adverse events (5.7%).Beyond underreporting of age-specific data, the investigators found that age categories themselves may be an area in need of improvement.“When outcomes pertaining to older adults were reported, the results were inconsistent as evidenced by the array of age distributions and varying categorization of ‘older adults,’” the investigators wrote. “There is a significant and timely need to design all clinical trials to include older adults and utilize a broad array of geriatric-specific outcomes.” Dr. Hshieh said these findings are concerning, but the study itself suggests the medical community is making efforts to correct the data shortage.“It was actually an important study, even though the results are a little discouraging,” Dr. Hshieh said. “What I’m hoping is that [these findings], combined with all the other literature that’s starting to come out about the need for more research in older patients with cancer, is going to be an impetus for us to do more research, and to be more open to treating older patients, and not to be afraid to confront this head on.”When asked about strategies for managing elderly patients, Dr. Hshieh first recommended the 2018 ASCO Guideline for Geriatric Oncology (J Clin Oncol. 2018 Aug 1;36[22]:2326-47).
“It’s very well written,” she said. “It is clear and user-friendly.”
Dr. Hshieh also offered some simple principles that may help guide clinical decision making.“I’m thinking of three things that an oncologist in the community would want to look at when they see an older patient and they’re trying to determine their treatment plan,” she said. “I would say [the oncologist] should look at [the patient’s] function; their psychosocial status, which includes mood and the support that they have in the community; and cognition.”
Dr. Hshieh and the study authors reported no conflicts of interest.
SOURCE: BrintzenhofeSzoc K et al. J Geriatr Oncol. 2020 Jan 10. pii: S1879-4068(19)30501-6.
Phase 3 clinical trials for cancer are underreporting safety and efficacy data for elderly patients, according to a systematic review of 159 articles.
Roughly 40% of articles reporting efficacy data and 9% of articles reporting safety data had results stratified by age, Karlynn BrintzenhofeSzoc, PhD, of the University of Cincinnati, and colleagues noted in the Journal of Geriatric Oncology.
“Results of our systematic review suggest that there is inadequate reporting of treatment efficacy and adverse events as well as discrepancies as to how older age is defined, considered, and reported,” the investigators wrote. “This sparse and varied reporting critically limits the evidence base for treating older patients with cancer.”
This study was inspired by the American Society of Clinical Oncology, which turned a spotlight on the age-specific data shortage in 2015, when it published a statement that called for inclusion of more elderly patients in cancer trials (J Clin Oncol. 2015 Nov 10;33[32]:3826-33).
According to Tammy Hshieh, MD, a geriatrician at Dana-Farber Cancer Institute in Boston, data for elderly patients with cancer are needed more than ever.
“Cancer care has become, increasingly, a field where precision medicine is at its strongest,” Dr. Hshieh said in an interview. “[Oncologists] have a lot of data on patients that allow them to tailor their care to each individual patient’s profile, and so the fact that there is not a lot of evidence looking at toxicities and side effects for older patients makes it basically harder for oncologists to practice evidence-based medicine for this vulnerable but growing population.” This leads to poorer and more variable outcomes, Dr. Hshieh said. When data aren’t available, clinicians must rely on experience and recognize that patient age isn’t as simple as date of birth.
“Oncologists looking at older patients really have to trust their gestalt and their experience in determining how to provide the best care for their older patients,” she said. “They have to look at the chronological age of the patient and try to determine whether that actually matches more of what we’re saying is the physiological age of the patient and use that to guide their treatment.”
Study details
The study included phase 3 clinical trials of adult cancer patients that were conducted from mid-2016 through mid-2017. After identifying 929 manuscripts, the investigators removed duplicates and those that did not meet criteria. This left 159 articles published in 36 journals and covering 25 cancer types.
Of the 159 articles, 73.6% included age-specific medians (in addition to age means), and 47.2% had data stratified by age.
Efficacy was often reported (96.2%), but only 39.9% of articles specified age when describing effectiveness. Although most articles (84.9%) included safety data, only 8.9% had safety findings stratified by age.
In article discussion sections, age was mentioned infrequently in relation to treatment efficacy (13.8%) and rarely in relation to complications and adverse events (5.7%).Beyond underreporting of age-specific data, the investigators found that age categories themselves may be an area in need of improvement.“When outcomes pertaining to older adults were reported, the results were inconsistent as evidenced by the array of age distributions and varying categorization of ‘older adults,’” the investigators wrote. “There is a significant and timely need to design all clinical trials to include older adults and utilize a broad array of geriatric-specific outcomes.” Dr. Hshieh said these findings are concerning, but the study itself suggests the medical community is making efforts to correct the data shortage.“It was actually an important study, even though the results are a little discouraging,” Dr. Hshieh said. “What I’m hoping is that [these findings], combined with all the other literature that’s starting to come out about the need for more research in older patients with cancer, is going to be an impetus for us to do more research, and to be more open to treating older patients, and not to be afraid to confront this head on.”When asked about strategies for managing elderly patients, Dr. Hshieh first recommended the 2018 ASCO Guideline for Geriatric Oncology (J Clin Oncol. 2018 Aug 1;36[22]:2326-47).
“It’s very well written,” she said. “It is clear and user-friendly.”
Dr. Hshieh also offered some simple principles that may help guide clinical decision making.“I’m thinking of three things that an oncologist in the community would want to look at when they see an older patient and they’re trying to determine their treatment plan,” she said. “I would say [the oncologist] should look at [the patient’s] function; their psychosocial status, which includes mood and the support that they have in the community; and cognition.”
Dr. Hshieh and the study authors reported no conflicts of interest.
SOURCE: BrintzenhofeSzoc K et al. J Geriatr Oncol. 2020 Jan 10. pii: S1879-4068(19)30501-6.
FROM JOURNAL OF GERIATRIC ONCOLOGY
Society of Hospital Medicine cancels 2020 Annual Conference
The Society of Hospital Medicine (SHM) has canceled its annual conference, scheduled for mid-April, joining a growing list of events shuttered by coronavirus (COVID-19) concerns.
In a March 13 announcement, SHM said it would be impossible for the society to host the Hospital Medicine 2020 conference amid the escalating health concerns regarding the global COVID-19 outbreak. For more information about the cancellation and the society’s refund policies, see the SHM website for a list of frequently answered questions.
The Society of Hospital Medicine (SHM) has canceled its annual conference, scheduled for mid-April, joining a growing list of events shuttered by coronavirus (COVID-19) concerns.
In a March 13 announcement, SHM said it would be impossible for the society to host the Hospital Medicine 2020 conference amid the escalating health concerns regarding the global COVID-19 outbreak. For more information about the cancellation and the society’s refund policies, see the SHM website for a list of frequently answered questions.
The Society of Hospital Medicine (SHM) has canceled its annual conference, scheduled for mid-April, joining a growing list of events shuttered by coronavirus (COVID-19) concerns.
In a March 13 announcement, SHM said it would be impossible for the society to host the Hospital Medicine 2020 conference amid the escalating health concerns regarding the global COVID-19 outbreak. For more information about the cancellation and the society’s refund policies, see the SHM website for a list of frequently answered questions.
Accelerated fetal growth in boys associated with development of AML
Accelerated fetal growth was associated with acute myeloid leukemia (AML), especially in infant boys and those with minimally differentiated leukemia, according to researchers from the Childhood Leukemia International Consortium (CLIC).
They assessed data from 22 studies involving a total of 3,564 cases to determine if there was an association between fetal growth and AML. The researchers also examined whether this association might vary by age, sex and disease subtype, according to their report published in the European Journal of Cancer.
The researchers calculated pooled estimates by age, sex and overall for harmonized fetal growth markers in association with AML. They used data from 17 International Fetal and Newborn Growth Consortium for the 21st Century Project studies and performed meta-analyses on 5 more studies. They also did subanalyses based on AML subtype.
They found a nearly 50% increased risk of AML among large-for-gestational-age infant boys (odds ratio [OR]: 1.49, 95% confidence interval [CI]: 1.03-2.14), reduced to 34% in boys aged less than 2 years (OR: 1.34, 95% CI: 1.05-1.71) and 25% in boys aged 0-14 years (OR: 1.25, 95% CI: 1.06-1.46). The association of large for gestational age was stronger in boys with the M0/M1 subtype (OR: 1.80, 95% CI: 1.15-2.83). In addition, large birth length for gestational age was also positively associated with AML (OR: 1.38, 95% CI: 1.00-1.92) in boys. By contrast, there, none of these factors were associated with AML in girls, nor were there associates for girls with respect to decelerated fetal growth markers.
“Although the absolute risk seems to be low at a population level, given the rarity of childhood AML, it would be worth exploring whether modifiable factors leading to macrosomia may also affect AML risk to stimulate future monitoring and preventive interventions before and during pregnancy,” the researchers suggested.
The authors reported that they had no conflicts of interest.
SOURCE: Karalexi MA et al. Eur J Canc. 2020;130:1-11.
Accelerated fetal growth was associated with acute myeloid leukemia (AML), especially in infant boys and those with minimally differentiated leukemia, according to researchers from the Childhood Leukemia International Consortium (CLIC).
They assessed data from 22 studies involving a total of 3,564 cases to determine if there was an association between fetal growth and AML. The researchers also examined whether this association might vary by age, sex and disease subtype, according to their report published in the European Journal of Cancer.
The researchers calculated pooled estimates by age, sex and overall for harmonized fetal growth markers in association with AML. They used data from 17 International Fetal and Newborn Growth Consortium for the 21st Century Project studies and performed meta-analyses on 5 more studies. They also did subanalyses based on AML subtype.
They found a nearly 50% increased risk of AML among large-for-gestational-age infant boys (odds ratio [OR]: 1.49, 95% confidence interval [CI]: 1.03-2.14), reduced to 34% in boys aged less than 2 years (OR: 1.34, 95% CI: 1.05-1.71) and 25% in boys aged 0-14 years (OR: 1.25, 95% CI: 1.06-1.46). The association of large for gestational age was stronger in boys with the M0/M1 subtype (OR: 1.80, 95% CI: 1.15-2.83). In addition, large birth length for gestational age was also positively associated with AML (OR: 1.38, 95% CI: 1.00-1.92) in boys. By contrast, there, none of these factors were associated with AML in girls, nor were there associates for girls with respect to decelerated fetal growth markers.
“Although the absolute risk seems to be low at a population level, given the rarity of childhood AML, it would be worth exploring whether modifiable factors leading to macrosomia may also affect AML risk to stimulate future monitoring and preventive interventions before and during pregnancy,” the researchers suggested.
The authors reported that they had no conflicts of interest.
SOURCE: Karalexi MA et al. Eur J Canc. 2020;130:1-11.
Accelerated fetal growth was associated with acute myeloid leukemia (AML), especially in infant boys and those with minimally differentiated leukemia, according to researchers from the Childhood Leukemia International Consortium (CLIC).
They assessed data from 22 studies involving a total of 3,564 cases to determine if there was an association between fetal growth and AML. The researchers also examined whether this association might vary by age, sex and disease subtype, according to their report published in the European Journal of Cancer.
The researchers calculated pooled estimates by age, sex and overall for harmonized fetal growth markers in association with AML. They used data from 17 International Fetal and Newborn Growth Consortium for the 21st Century Project studies and performed meta-analyses on 5 more studies. They also did subanalyses based on AML subtype.
They found a nearly 50% increased risk of AML among large-for-gestational-age infant boys (odds ratio [OR]: 1.49, 95% confidence interval [CI]: 1.03-2.14), reduced to 34% in boys aged less than 2 years (OR: 1.34, 95% CI: 1.05-1.71) and 25% in boys aged 0-14 years (OR: 1.25, 95% CI: 1.06-1.46). The association of large for gestational age was stronger in boys with the M0/M1 subtype (OR: 1.80, 95% CI: 1.15-2.83). In addition, large birth length for gestational age was also positively associated with AML (OR: 1.38, 95% CI: 1.00-1.92) in boys. By contrast, there, none of these factors were associated with AML in girls, nor were there associates for girls with respect to decelerated fetal growth markers.
“Although the absolute risk seems to be low at a population level, given the rarity of childhood AML, it would be worth exploring whether modifiable factors leading to macrosomia may also affect AML risk to stimulate future monitoring and preventive interventions before and during pregnancy,” the researchers suggested.
The authors reported that they had no conflicts of interest.
SOURCE: Karalexi MA et al. Eur J Canc. 2020;130:1-11.
FROM THE EUROPEAN JOURNAL OF CANCER



