User login
COVID vaccine controversies: How can hospitalists help?
On April 1, Houston Methodist Hospital in Houston, Texas, announced a new policy that all of its staff would need to be vaccinated against COVID-19 by June 7 in order to hold onto their jobs. Most responded positively but an estimated 150 staff members who did not comply either resigned or were terminated. A lawsuit by employees opposed to the vaccine mandate was dismissed by Federal District Court Judge Lynn Hughes in June, although a subsequent lawsuit was filed Aug. 16.
Vaccines have been shown to dramatically reduce both the incidence and the severity of COVID infections. Vaccinations of health care workers, especially those who have direct contact with patients, are demonstrated to be effective strategies to significantly reduce, although not eliminate, the possibility of viral transmissions to patients – or to health care workers themselves – thus saving lives.
Hospitalists, in their central role in the care of hospitalized patients, and often with primary responsibility for managing their hospital’s COVID-19 caseloads, may find themselves encountering conversations about the vaccine, its safety, effectiveness, and mandates with their peers, other hospital staff, patients, and families, and their communities. They can play key roles in advocating for the vaccine, answering questions, clarifying the science, and dispelling misinformation – for those who are willing to listen.
Becker’s Hospital Review, which has kept an ongoing tally of announced vaccine mandate policies in hospitals, health systems, and health departments nationwide, reported on Aug. 13 that 1,850 or 30% of U.S. hospitals, had announced vaccine mandates.1 Often exceptions can be made, such as for medical or religious reasons, or with other declarations or opt-out provisions. But in many settings, mandating COVID vaccinations won’t be easy.
Amith Skandhan, MD, SFHM, FACP, a hospitalist at Southeast Health Medical Center in Dothan, Ala., and a core faculty member in the internal medicine residency program at Alabama College of Osteopathic Medicine, said that implementing vaccine mandates will be more difficult in smaller health systems, in rural communities, and in states with lower vaccination rates and greater vaccine controversy.
Alabama has the lowest vaccination rates in the country, reflected in the recent rise in COVID cases and hospitalizations, even higher than during the surge of late 2020, Dr. Skandhan said. “In June we had one COVID patient in this hospital.” By late August the number was 119 COVID patients and climbing.
But where he works, in a health system where staffing is already spread thin, a vaccine mandate would be challenging. “What if our staff started leaving? It’s only 10 minutes from here to the Florida or Georgia border,” Dr. Skandhan said. Health care workers opposed to vaccinations would have the option of easily seeking work elsewhere.
When contacted for this article, he had been off work for several days but was mentally preparing himself to go back. “I’m not even following the [COVID-19] numbers but I am prepared for the worst. I know it will be mostly COVID. People just don’t realize what goes into this work.”
Dr. Skandhan, who said he was the third or fourth person in Alabama to receive the COVID vaccine, often finds himself feeling frustrated and angry – in the midst of a surge in cases that could have been prevented – that such a beneficial medical advance for bringing the pandemic under control became so politicized. “It is imperative that we find out why this mistrust exists and work to address it. It has to be done.”
Protecting health care professionals
On July 26, the Society of Hospital Medicine joined 50 other health care organizations including the American Medical Association, American Nurses Association, and American Academy of Pediatrics in advocating for all health care employers to require their employees to be vaccinated against COVID, in order to protect the safety of all patients and residents of health care facilities.2
“As an organization, we support vaccinating health care workers, including hospitalists, to help stop the spread of COVID-19 and the increasingly dominant Delta variant,” said SHM’s chief executive officer Eric E. Howell, MD, MHM, in a prepared statement. “We aim to uphold the highest standards among hospitalists and other health care providers to help protect our fellow health care professionals, our patients, and our communities.”
To that end, Dr. Skandhan has started conversations with hospital staff who he knows are not vaccinated. “For some, we’re not able to have a civil conversation, but in most cases I can help to persuade people.” The reasons people give for not getting vaccinated are not based in science, he said. “I am worried about the safety of our hospitalists and staff nurses.” But unvaccinated frontline workers are also putting their patients at risk. “Can we say why they’re hesitating? Can we have an honest discourse? If we can’t do that with our colleagues, how can we blame the patients?”
Dr. Skandhan encourages hospitalists to start simply in their own hospitals, trying to influence their own departments and colleagues. “If you can convince one or two more every week, you can start a chain reaction. Have that conversation. Use your trust.” For some hospitalized patients, the vaccination conversation comes too late, after their infection, but even some of them might consider obtaining it down the road or trying to persuade family members to get vaccinated.
Adult hospitalists, however, may not have received training in how to effectively address vaccine fears and misconceptions among their patients, he said. Because the patients they see in the hospital are already very sick, they don’t get a lot of practice talking about vaccines except, perhaps, for the influenza vaccine.
Pediatric hospitalists have more experience with such conversations involving their patients’ parents, Dr. Skandhan said. “It comes more naturally to them. We need to learn quickly from them about how to talk about vaccines with our patients.”
Pediatric training and experience
Anika Kumar, MD, FHM, FAAP, a pediatric hospitalist at the Cleveland Clinic and the pediatric editor of The Hospitalist, agrees that pediatricians and pediatric hospitalists often have received more training in how to lead vaccination conversations. She often talks about vaccines with the parents of hospitalized children relative to chicken pox, measles, and other diseases of childhood.
Pediatric hospitalists may also ask to administer the hepatitis B vaccine to newborn babies, along with other preventive treatments such as eye drops and vitamin K shots. “I often encourage the influenza vaccine prior to the patient’s hospital discharge, especially for kids with chronic conditions, asthma, diabetes, or premature birth. We talk about how the influenza vaccine isn’t perfect, but it helps to prevent more serious disease,” she said.
“A lot of vaccine hesitancy comes from misunderstandings about the role of vaccines,” she said. People forget that for years children have been getting vaccines before starting school. “Misinformation and opinions about vaccines have existed for decades. What’s new today is the abundance of sources for obtaining these opinions. My job is to inform families of scientific facts and to address their concerns.”
It has become more common recently for parents to say they don’t want their kids to get vaccinated, Dr. Kumar said. Another group is better described as vaccine hesitant and just needs more information. “I may not, by the time they leave the hospital, convince them to allow me to administer the vaccine. But in the discharge summary, I document that I had this conversation. I’ve done my due diligence and tried to start a larger dialogue. I say: ‘I encourage you to continue this discussion with the pediatrician you trust.’ I also communicate with the outpatient team,” she said.
“But it’s our responsibility, because we’re the ones seeing these patients, to do whatever we can to keep our patients from getting sick. A lot of challenging conversations we have with families are just trying to find out where they’re at with the issue – which can lead to productive dialogue.”
Ariel Carpenter, MD, a 4th-year resident in internal medicine and pediatrics at the University of Louisville (Ky.), and a future pediatric hospitalist, agreed that her combined training in med-peds has been helpful preparation for the vaccine conversation. That training has included techniques of motivational interviewing. In pediatrics, she explained, the communication is a little softer. “I try to approach my patients in a family-centered way.”
Dr. Carpenter recently wrote a personal essay for Louisville Medicine magazine from the perspective of growing up homeschooled by a mother who didn’t believe in vaccines.3 As a teenager, she independently obtained the complete childhood vaccine series so that she could do medical shadowing and volunteering. In medical school she became a passionate vaccine advocate, eventually persuading her mother to change her mind on the subject in time for the COVID vaccine.
“There’s not one answer to the vaccination dilemma,” she said. “Different approaches are required because there are so many different reasons for it. Based on my own life experience, I try to approach patients where they are – not from a place of data and science. What worked in my own family, and works with my patients, is first to establish trust. If they trust you, they’re more likely to listen. Simply ask their worries and concerns,” Dr. Carpenter said.
“A lot of them haven’t had the opportunity before to sit down with a physician they trust and have their worries listened to. They don’t feel heard in our medical system. So I remind myself that I need to understand my patients first – before inserting myself into the conversation.”
Many patients she sees are in an information bubble, with a very different understanding of the issue than their doctors. “A lot of well-meaning people feel they are making the safer choice. Very few truly don’t care about protecting others. But they don’t feel the urgency about that and see the vaccine as the scarier option right now.”
Frontline vaccine advocates
Hospitalists are the frontline advocates within their hospital system, in a position to lead, so they need to make vaccines a priority, Dr. Carpenter said. They should also make sure that their hospitals have ready access to the vaccine, so patients who agree to receive it are able to get it quickly. “In our hospital they can get the shot within a few hours if the opportunity arises. We stocked the Johnson & Johnson vaccine so that they wouldn’t have to connect with another health care provider in order to get a second dose.”
Hospitals should also invest in access to vaccine counseling training and personnel. “Fund a nurse clinician who can screen and counsel hospitalized patients for vaccination. If they meet resistance, they can then refer to the dedicated physician of the day to have the conversation,” she said. “But if we don’t mention it, patients will assume we don’t feel strongly about it.”
Because hospitalists are front and center in treating COVID, they need to be the experts and the people offering guidance, said Shyam Odeti, MD, SFHM, FAAFP, section chief for hospital medicine at the Carilion Clinic in Roanoke, Va. “What we’re trying to do is spread awareness. We educated physician groups, learners, and clinical teams during the initial phase, and now mostly patients and their families.” COVID vaccine reluctance is hard to overcome, Dr. Odeti said. People feel the vaccine was developed very quickly. But there are different ways to present it.
“Like most doctors, I thought people would jump on a vaccine to get past the pandemic. I was surprised and then disappointed. Right now, the pandemic is among the unvaccinated. So we face these encounters, and we’re doing our best to overcome the misinformation. My organization is 100% supportive. We talk about these issues every day.”
Carilion, effective Oct. 1, has required unvaccinated employees to get weekly COVID tests and wear an N95 mask while working, and has developed Facebook pages, other social media, and an Internet presence to address these issues. “We’ve gone to the local African-American community with physician leaders active in that community. We had a Spanish language roundtable,” Dr. Odeti said.
Dr. Skandhan reported that the Wiregrass regional chapter of SHM recently organized a successful statewide community educational event aimed at empowering community leaders to address vaccine misinformation and mistrust. “We surveyed religious leaders and pastors regarding the causes of vaccine hesitancy and reached out to physicians active in community awareness.” Based on that input, a presentation by the faith leaders was developed. Legislators from the Alabama State Senate’s Healthcare Policy Committee were also invited to the presentation and discussion.
Trying to stay positive
It’s important to try to stay positive, Dr. Odeti said. “We have to be empathetic with every patient. We have to keep working at this, since there’s no way out of the pandemic except through vaccinations. But it all creates stress for hospitalists. Our job is made significantly more difficult by the vaccine controversy.”
Jennifer Cowart, MD, a hospitalist at Mayo Clinic in Jacksonville, Fla., has been outspoken in her community about vaccination and masking issues, talking to reporters, attending rallies and press conferences, posting on social media, and speaking in favor of mask policies at a local school board meeting. She is part of an informal local group called Doctors Fighting COVID, which meets online to strategize how to share its expertise, including writing a recent letter about masks to Jacksonville’s mayor.
“In July, when we saw the Delta variant surging locally, we held a webinar via local media, taking calls about the vaccine from the community. I’m trying not to make this a political issue, but we are health officials.” Dr. Cowart said she also tries not to raise her voice when speaking with vaccine opponents and tries to remain empathetic. “Even though inwardly I’m screaming, I try to stay calm. The misinformation is real. People are afraid and feeling pressure. I do my best, but I’m human, too.”
Hospitalists need to pull whatever levers they can to help advance understanding of vaccines, Dr. Cowart said. “In the hospital, our biggest issue is time. We often don’t have it, with a long list of patients to see. But every patient encounter is an opportunity to talk to patients, whether they have COVID or something else.” Sometimes, she might go back to a patient’s room after rounds to resume the conversation.
Hospital nurses have been trained and entrusted to do tobacco abatement counseling, she said, so why not mobilize them for vaccine education? “Or respiratory therapists, who do inhaler training, could talk about what it’s like to care for COVID patients. There’s a whole bunch of staff in the hospital who could be mobilized,” she said.
“I feel passionate about vaccines, as a hospitalist, as a medical educator, as a daughter, as a responsible member of society,” said Eileen Barrett, MD, MPH, SFHM, MACP, director of continuing medical education at the University of New Mexico, Albuquerque. “I see this as a personal and societal responsibility. When I speak about the vaccine among groups of doctors, I say we need to stay in our lane regarding our skills at interpreting the science and not undermining it.”
Some health care worker hesitancy is from distrust of pharmaceutical companies, or of federal agencies, she said. “Our research has highlighted to me the widespread inequity issues in our health care system. We should also take a long, hard look at how we teach the scientific method to health professionals. That will be part of a pandemic retrospective.”
Sometimes with people who are vaccine deliberative, whether health care workers or patients, there is a small window of opportunity. “We need to hear people and respond to them as people. Then, if they are willing to get vaccinated, we need to accomplish that as quickly and easily as possible,” Dr. Barrett said. “I see them make a face and say, ‘Well, okay, I’ll do it.’ We need to get the vaccine to them that same day. We should be able to accomplish that.”
References
1. Gamble M. 30% of US hospitals mandate vaccination for employment. Becker’s Hospital Review. 2021 Aug 13. www.beckershospitalreview.com/workforce/covid-19-vaccination-needed-to-work-at-30-of-us-hospitals.html .
2. Society of Hospital Medicine signs on to joint statement in support of health worker COVID-19 vaccine mandates. Press release. 2021 Jul 26. www.hospitalmedicine.org/news-publications/press-releases/society-of-hospital-medicine-signs-on-to-joint-statement-of-support-of-health-worker-covid-19-vaccine-mandates/.
3. Carpenter A. A physician’s lessons from an unvaccinated childhood. Louisville Medicine. 2021 July;69(2):26-7. https://viewer.joomag.com/louisville-medicine-volume-69-issue-2/0045988001624974172?short&.
Lessons for hospitalists from the vaccination controversy
1. Remain up-to-date on information about the COVID infection, its treatment, and vaccination efficacy data.
2. Hospitalists should take advantage of their positions to lead conversations in their facilities about the importance of COVID vaccinations.
3. Other professionals in the hospital, with some additional training and support, could take on the role of providing vaccine education and support – with a physician to back them up on difficult cases.
4. It’s important to listen to people’s concerns, try to build trust, and establish dialogue before starting to convey a lot of information. People need to feel heard.
5. If you are successful in persuading someone to take the vaccine, a shot should be promptly and easily accessible to them.
6. Pediatric hospitalists may have more experience and skill with vaccine discussions, which they should share with their peers who treat adults.
On April 1, Houston Methodist Hospital in Houston, Texas, announced a new policy that all of its staff would need to be vaccinated against COVID-19 by June 7 in order to hold onto their jobs. Most responded positively but an estimated 150 staff members who did not comply either resigned or were terminated. A lawsuit by employees opposed to the vaccine mandate was dismissed by Federal District Court Judge Lynn Hughes in June, although a subsequent lawsuit was filed Aug. 16.
Vaccines have been shown to dramatically reduce both the incidence and the severity of COVID infections. Vaccinations of health care workers, especially those who have direct contact with patients, are demonstrated to be effective strategies to significantly reduce, although not eliminate, the possibility of viral transmissions to patients – or to health care workers themselves – thus saving lives.
Hospitalists, in their central role in the care of hospitalized patients, and often with primary responsibility for managing their hospital’s COVID-19 caseloads, may find themselves encountering conversations about the vaccine, its safety, effectiveness, and mandates with their peers, other hospital staff, patients, and families, and their communities. They can play key roles in advocating for the vaccine, answering questions, clarifying the science, and dispelling misinformation – for those who are willing to listen.
Becker’s Hospital Review, which has kept an ongoing tally of announced vaccine mandate policies in hospitals, health systems, and health departments nationwide, reported on Aug. 13 that 1,850 or 30% of U.S. hospitals, had announced vaccine mandates.1 Often exceptions can be made, such as for medical or religious reasons, or with other declarations or opt-out provisions. But in many settings, mandating COVID vaccinations won’t be easy.
Amith Skandhan, MD, SFHM, FACP, a hospitalist at Southeast Health Medical Center in Dothan, Ala., and a core faculty member in the internal medicine residency program at Alabama College of Osteopathic Medicine, said that implementing vaccine mandates will be more difficult in smaller health systems, in rural communities, and in states with lower vaccination rates and greater vaccine controversy.
Alabama has the lowest vaccination rates in the country, reflected in the recent rise in COVID cases and hospitalizations, even higher than during the surge of late 2020, Dr. Skandhan said. “In June we had one COVID patient in this hospital.” By late August the number was 119 COVID patients and climbing.
But where he works, in a health system where staffing is already spread thin, a vaccine mandate would be challenging. “What if our staff started leaving? It’s only 10 minutes from here to the Florida or Georgia border,” Dr. Skandhan said. Health care workers opposed to vaccinations would have the option of easily seeking work elsewhere.
When contacted for this article, he had been off work for several days but was mentally preparing himself to go back. “I’m not even following the [COVID-19] numbers but I am prepared for the worst. I know it will be mostly COVID. People just don’t realize what goes into this work.”
Dr. Skandhan, who said he was the third or fourth person in Alabama to receive the COVID vaccine, often finds himself feeling frustrated and angry – in the midst of a surge in cases that could have been prevented – that such a beneficial medical advance for bringing the pandemic under control became so politicized. “It is imperative that we find out why this mistrust exists and work to address it. It has to be done.”
Protecting health care professionals
On July 26, the Society of Hospital Medicine joined 50 other health care organizations including the American Medical Association, American Nurses Association, and American Academy of Pediatrics in advocating for all health care employers to require their employees to be vaccinated against COVID, in order to protect the safety of all patients and residents of health care facilities.2
“As an organization, we support vaccinating health care workers, including hospitalists, to help stop the spread of COVID-19 and the increasingly dominant Delta variant,” said SHM’s chief executive officer Eric E. Howell, MD, MHM, in a prepared statement. “We aim to uphold the highest standards among hospitalists and other health care providers to help protect our fellow health care professionals, our patients, and our communities.”
To that end, Dr. Skandhan has started conversations with hospital staff who he knows are not vaccinated. “For some, we’re not able to have a civil conversation, but in most cases I can help to persuade people.” The reasons people give for not getting vaccinated are not based in science, he said. “I am worried about the safety of our hospitalists and staff nurses.” But unvaccinated frontline workers are also putting their patients at risk. “Can we say why they’re hesitating? Can we have an honest discourse? If we can’t do that with our colleagues, how can we blame the patients?”
Dr. Skandhan encourages hospitalists to start simply in their own hospitals, trying to influence their own departments and colleagues. “If you can convince one or two more every week, you can start a chain reaction. Have that conversation. Use your trust.” For some hospitalized patients, the vaccination conversation comes too late, after their infection, but even some of them might consider obtaining it down the road or trying to persuade family members to get vaccinated.
Adult hospitalists, however, may not have received training in how to effectively address vaccine fears and misconceptions among their patients, he said. Because the patients they see in the hospital are already very sick, they don’t get a lot of practice talking about vaccines except, perhaps, for the influenza vaccine.
Pediatric hospitalists have more experience with such conversations involving their patients’ parents, Dr. Skandhan said. “It comes more naturally to them. We need to learn quickly from them about how to talk about vaccines with our patients.”
Pediatric training and experience
Anika Kumar, MD, FHM, FAAP, a pediatric hospitalist at the Cleveland Clinic and the pediatric editor of The Hospitalist, agrees that pediatricians and pediatric hospitalists often have received more training in how to lead vaccination conversations. She often talks about vaccines with the parents of hospitalized children relative to chicken pox, measles, and other diseases of childhood.
Pediatric hospitalists may also ask to administer the hepatitis B vaccine to newborn babies, along with other preventive treatments such as eye drops and vitamin K shots. “I often encourage the influenza vaccine prior to the patient’s hospital discharge, especially for kids with chronic conditions, asthma, diabetes, or premature birth. We talk about how the influenza vaccine isn’t perfect, but it helps to prevent more serious disease,” she said.
“A lot of vaccine hesitancy comes from misunderstandings about the role of vaccines,” she said. People forget that for years children have been getting vaccines before starting school. “Misinformation and opinions about vaccines have existed for decades. What’s new today is the abundance of sources for obtaining these opinions. My job is to inform families of scientific facts and to address their concerns.”
It has become more common recently for parents to say they don’t want their kids to get vaccinated, Dr. Kumar said. Another group is better described as vaccine hesitant and just needs more information. “I may not, by the time they leave the hospital, convince them to allow me to administer the vaccine. But in the discharge summary, I document that I had this conversation. I’ve done my due diligence and tried to start a larger dialogue. I say: ‘I encourage you to continue this discussion with the pediatrician you trust.’ I also communicate with the outpatient team,” she said.
“But it’s our responsibility, because we’re the ones seeing these patients, to do whatever we can to keep our patients from getting sick. A lot of challenging conversations we have with families are just trying to find out where they’re at with the issue – which can lead to productive dialogue.”
Ariel Carpenter, MD, a 4th-year resident in internal medicine and pediatrics at the University of Louisville (Ky.), and a future pediatric hospitalist, agreed that her combined training in med-peds has been helpful preparation for the vaccine conversation. That training has included techniques of motivational interviewing. In pediatrics, she explained, the communication is a little softer. “I try to approach my patients in a family-centered way.”
Dr. Carpenter recently wrote a personal essay for Louisville Medicine magazine from the perspective of growing up homeschooled by a mother who didn’t believe in vaccines.3 As a teenager, she independently obtained the complete childhood vaccine series so that she could do medical shadowing and volunteering. In medical school she became a passionate vaccine advocate, eventually persuading her mother to change her mind on the subject in time for the COVID vaccine.
“There’s not one answer to the vaccination dilemma,” she said. “Different approaches are required because there are so many different reasons for it. Based on my own life experience, I try to approach patients where they are – not from a place of data and science. What worked in my own family, and works with my patients, is first to establish trust. If they trust you, they’re more likely to listen. Simply ask their worries and concerns,” Dr. Carpenter said.
“A lot of them haven’t had the opportunity before to sit down with a physician they trust and have their worries listened to. They don’t feel heard in our medical system. So I remind myself that I need to understand my patients first – before inserting myself into the conversation.”
Many patients she sees are in an information bubble, with a very different understanding of the issue than their doctors. “A lot of well-meaning people feel they are making the safer choice. Very few truly don’t care about protecting others. But they don’t feel the urgency about that and see the vaccine as the scarier option right now.”
Frontline vaccine advocates
Hospitalists are the frontline advocates within their hospital system, in a position to lead, so they need to make vaccines a priority, Dr. Carpenter said. They should also make sure that their hospitals have ready access to the vaccine, so patients who agree to receive it are able to get it quickly. “In our hospital they can get the shot within a few hours if the opportunity arises. We stocked the Johnson & Johnson vaccine so that they wouldn’t have to connect with another health care provider in order to get a second dose.”
Hospitals should also invest in access to vaccine counseling training and personnel. “Fund a nurse clinician who can screen and counsel hospitalized patients for vaccination. If they meet resistance, they can then refer to the dedicated physician of the day to have the conversation,” she said. “But if we don’t mention it, patients will assume we don’t feel strongly about it.”
Because hospitalists are front and center in treating COVID, they need to be the experts and the people offering guidance, said Shyam Odeti, MD, SFHM, FAAFP, section chief for hospital medicine at the Carilion Clinic in Roanoke, Va. “What we’re trying to do is spread awareness. We educated physician groups, learners, and clinical teams during the initial phase, and now mostly patients and their families.” COVID vaccine reluctance is hard to overcome, Dr. Odeti said. People feel the vaccine was developed very quickly. But there are different ways to present it.
“Like most doctors, I thought people would jump on a vaccine to get past the pandemic. I was surprised and then disappointed. Right now, the pandemic is among the unvaccinated. So we face these encounters, and we’re doing our best to overcome the misinformation. My organization is 100% supportive. We talk about these issues every day.”
Carilion, effective Oct. 1, has required unvaccinated employees to get weekly COVID tests and wear an N95 mask while working, and has developed Facebook pages, other social media, and an Internet presence to address these issues. “We’ve gone to the local African-American community with physician leaders active in that community. We had a Spanish language roundtable,” Dr. Odeti said.
Dr. Skandhan reported that the Wiregrass regional chapter of SHM recently organized a successful statewide community educational event aimed at empowering community leaders to address vaccine misinformation and mistrust. “We surveyed religious leaders and pastors regarding the causes of vaccine hesitancy and reached out to physicians active in community awareness.” Based on that input, a presentation by the faith leaders was developed. Legislators from the Alabama State Senate’s Healthcare Policy Committee were also invited to the presentation and discussion.
Trying to stay positive
It’s important to try to stay positive, Dr. Odeti said. “We have to be empathetic with every patient. We have to keep working at this, since there’s no way out of the pandemic except through vaccinations. But it all creates stress for hospitalists. Our job is made significantly more difficult by the vaccine controversy.”
Jennifer Cowart, MD, a hospitalist at Mayo Clinic in Jacksonville, Fla., has been outspoken in her community about vaccination and masking issues, talking to reporters, attending rallies and press conferences, posting on social media, and speaking in favor of mask policies at a local school board meeting. She is part of an informal local group called Doctors Fighting COVID, which meets online to strategize how to share its expertise, including writing a recent letter about masks to Jacksonville’s mayor.
“In July, when we saw the Delta variant surging locally, we held a webinar via local media, taking calls about the vaccine from the community. I’m trying not to make this a political issue, but we are health officials.” Dr. Cowart said she also tries not to raise her voice when speaking with vaccine opponents and tries to remain empathetic. “Even though inwardly I’m screaming, I try to stay calm. The misinformation is real. People are afraid and feeling pressure. I do my best, but I’m human, too.”
Hospitalists need to pull whatever levers they can to help advance understanding of vaccines, Dr. Cowart said. “In the hospital, our biggest issue is time. We often don’t have it, with a long list of patients to see. But every patient encounter is an opportunity to talk to patients, whether they have COVID or something else.” Sometimes, she might go back to a patient’s room after rounds to resume the conversation.
Hospital nurses have been trained and entrusted to do tobacco abatement counseling, she said, so why not mobilize them for vaccine education? “Or respiratory therapists, who do inhaler training, could talk about what it’s like to care for COVID patients. There’s a whole bunch of staff in the hospital who could be mobilized,” she said.
“I feel passionate about vaccines, as a hospitalist, as a medical educator, as a daughter, as a responsible member of society,” said Eileen Barrett, MD, MPH, SFHM, MACP, director of continuing medical education at the University of New Mexico, Albuquerque. “I see this as a personal and societal responsibility. When I speak about the vaccine among groups of doctors, I say we need to stay in our lane regarding our skills at interpreting the science and not undermining it.”
Some health care worker hesitancy is from distrust of pharmaceutical companies, or of federal agencies, she said. “Our research has highlighted to me the widespread inequity issues in our health care system. We should also take a long, hard look at how we teach the scientific method to health professionals. That will be part of a pandemic retrospective.”
Sometimes with people who are vaccine deliberative, whether health care workers or patients, there is a small window of opportunity. “We need to hear people and respond to them as people. Then, if they are willing to get vaccinated, we need to accomplish that as quickly and easily as possible,” Dr. Barrett said. “I see them make a face and say, ‘Well, okay, I’ll do it.’ We need to get the vaccine to them that same day. We should be able to accomplish that.”
References
1. Gamble M. 30% of US hospitals mandate vaccination for employment. Becker’s Hospital Review. 2021 Aug 13. www.beckershospitalreview.com/workforce/covid-19-vaccination-needed-to-work-at-30-of-us-hospitals.html .
2. Society of Hospital Medicine signs on to joint statement in support of health worker COVID-19 vaccine mandates. Press release. 2021 Jul 26. www.hospitalmedicine.org/news-publications/press-releases/society-of-hospital-medicine-signs-on-to-joint-statement-of-support-of-health-worker-covid-19-vaccine-mandates/.
3. Carpenter A. A physician’s lessons from an unvaccinated childhood. Louisville Medicine. 2021 July;69(2):26-7. https://viewer.joomag.com/louisville-medicine-volume-69-issue-2/0045988001624974172?short&.
Lessons for hospitalists from the vaccination controversy
1. Remain up-to-date on information about the COVID infection, its treatment, and vaccination efficacy data.
2. Hospitalists should take advantage of their positions to lead conversations in their facilities about the importance of COVID vaccinations.
3. Other professionals in the hospital, with some additional training and support, could take on the role of providing vaccine education and support – with a physician to back them up on difficult cases.
4. It’s important to listen to people’s concerns, try to build trust, and establish dialogue before starting to convey a lot of information. People need to feel heard.
5. If you are successful in persuading someone to take the vaccine, a shot should be promptly and easily accessible to them.
6. Pediatric hospitalists may have more experience and skill with vaccine discussions, which they should share with their peers who treat adults.
On April 1, Houston Methodist Hospital in Houston, Texas, announced a new policy that all of its staff would need to be vaccinated against COVID-19 by June 7 in order to hold onto their jobs. Most responded positively but an estimated 150 staff members who did not comply either resigned or were terminated. A lawsuit by employees opposed to the vaccine mandate was dismissed by Federal District Court Judge Lynn Hughes in June, although a subsequent lawsuit was filed Aug. 16.
Vaccines have been shown to dramatically reduce both the incidence and the severity of COVID infections. Vaccinations of health care workers, especially those who have direct contact with patients, are demonstrated to be effective strategies to significantly reduce, although not eliminate, the possibility of viral transmissions to patients – or to health care workers themselves – thus saving lives.
Hospitalists, in their central role in the care of hospitalized patients, and often with primary responsibility for managing their hospital’s COVID-19 caseloads, may find themselves encountering conversations about the vaccine, its safety, effectiveness, and mandates with their peers, other hospital staff, patients, and families, and their communities. They can play key roles in advocating for the vaccine, answering questions, clarifying the science, and dispelling misinformation – for those who are willing to listen.
Becker’s Hospital Review, which has kept an ongoing tally of announced vaccine mandate policies in hospitals, health systems, and health departments nationwide, reported on Aug. 13 that 1,850 or 30% of U.S. hospitals, had announced vaccine mandates.1 Often exceptions can be made, such as for medical or religious reasons, or with other declarations or opt-out provisions. But in many settings, mandating COVID vaccinations won’t be easy.
Amith Skandhan, MD, SFHM, FACP, a hospitalist at Southeast Health Medical Center in Dothan, Ala., and a core faculty member in the internal medicine residency program at Alabama College of Osteopathic Medicine, said that implementing vaccine mandates will be more difficult in smaller health systems, in rural communities, and in states with lower vaccination rates and greater vaccine controversy.
Alabama has the lowest vaccination rates in the country, reflected in the recent rise in COVID cases and hospitalizations, even higher than during the surge of late 2020, Dr. Skandhan said. “In June we had one COVID patient in this hospital.” By late August the number was 119 COVID patients and climbing.
But where he works, in a health system where staffing is already spread thin, a vaccine mandate would be challenging. “What if our staff started leaving? It’s only 10 minutes from here to the Florida or Georgia border,” Dr. Skandhan said. Health care workers opposed to vaccinations would have the option of easily seeking work elsewhere.
When contacted for this article, he had been off work for several days but was mentally preparing himself to go back. “I’m not even following the [COVID-19] numbers but I am prepared for the worst. I know it will be mostly COVID. People just don’t realize what goes into this work.”
Dr. Skandhan, who said he was the third or fourth person in Alabama to receive the COVID vaccine, often finds himself feeling frustrated and angry – in the midst of a surge in cases that could have been prevented – that such a beneficial medical advance for bringing the pandemic under control became so politicized. “It is imperative that we find out why this mistrust exists and work to address it. It has to be done.”
Protecting health care professionals
On July 26, the Society of Hospital Medicine joined 50 other health care organizations including the American Medical Association, American Nurses Association, and American Academy of Pediatrics in advocating for all health care employers to require their employees to be vaccinated against COVID, in order to protect the safety of all patients and residents of health care facilities.2
“As an organization, we support vaccinating health care workers, including hospitalists, to help stop the spread of COVID-19 and the increasingly dominant Delta variant,” said SHM’s chief executive officer Eric E. Howell, MD, MHM, in a prepared statement. “We aim to uphold the highest standards among hospitalists and other health care providers to help protect our fellow health care professionals, our patients, and our communities.”
To that end, Dr. Skandhan has started conversations with hospital staff who he knows are not vaccinated. “For some, we’re not able to have a civil conversation, but in most cases I can help to persuade people.” The reasons people give for not getting vaccinated are not based in science, he said. “I am worried about the safety of our hospitalists and staff nurses.” But unvaccinated frontline workers are also putting their patients at risk. “Can we say why they’re hesitating? Can we have an honest discourse? If we can’t do that with our colleagues, how can we blame the patients?”
Dr. Skandhan encourages hospitalists to start simply in their own hospitals, trying to influence their own departments and colleagues. “If you can convince one or two more every week, you can start a chain reaction. Have that conversation. Use your trust.” For some hospitalized patients, the vaccination conversation comes too late, after their infection, but even some of them might consider obtaining it down the road or trying to persuade family members to get vaccinated.
Adult hospitalists, however, may not have received training in how to effectively address vaccine fears and misconceptions among their patients, he said. Because the patients they see in the hospital are already very sick, they don’t get a lot of practice talking about vaccines except, perhaps, for the influenza vaccine.
Pediatric hospitalists have more experience with such conversations involving their patients’ parents, Dr. Skandhan said. “It comes more naturally to them. We need to learn quickly from them about how to talk about vaccines with our patients.”
Pediatric training and experience
Anika Kumar, MD, FHM, FAAP, a pediatric hospitalist at the Cleveland Clinic and the pediatric editor of The Hospitalist, agrees that pediatricians and pediatric hospitalists often have received more training in how to lead vaccination conversations. She often talks about vaccines with the parents of hospitalized children relative to chicken pox, measles, and other diseases of childhood.
Pediatric hospitalists may also ask to administer the hepatitis B vaccine to newborn babies, along with other preventive treatments such as eye drops and vitamin K shots. “I often encourage the influenza vaccine prior to the patient’s hospital discharge, especially for kids with chronic conditions, asthma, diabetes, or premature birth. We talk about how the influenza vaccine isn’t perfect, but it helps to prevent more serious disease,” she said.
“A lot of vaccine hesitancy comes from misunderstandings about the role of vaccines,” she said. People forget that for years children have been getting vaccines before starting school. “Misinformation and opinions about vaccines have existed for decades. What’s new today is the abundance of sources for obtaining these opinions. My job is to inform families of scientific facts and to address their concerns.”
It has become more common recently for parents to say they don’t want their kids to get vaccinated, Dr. Kumar said. Another group is better described as vaccine hesitant and just needs more information. “I may not, by the time they leave the hospital, convince them to allow me to administer the vaccine. But in the discharge summary, I document that I had this conversation. I’ve done my due diligence and tried to start a larger dialogue. I say: ‘I encourage you to continue this discussion with the pediatrician you trust.’ I also communicate with the outpatient team,” she said.
“But it’s our responsibility, because we’re the ones seeing these patients, to do whatever we can to keep our patients from getting sick. A lot of challenging conversations we have with families are just trying to find out where they’re at with the issue – which can lead to productive dialogue.”
Ariel Carpenter, MD, a 4th-year resident in internal medicine and pediatrics at the University of Louisville (Ky.), and a future pediatric hospitalist, agreed that her combined training in med-peds has been helpful preparation for the vaccine conversation. That training has included techniques of motivational interviewing. In pediatrics, she explained, the communication is a little softer. “I try to approach my patients in a family-centered way.”
Dr. Carpenter recently wrote a personal essay for Louisville Medicine magazine from the perspective of growing up homeschooled by a mother who didn’t believe in vaccines.3 As a teenager, she independently obtained the complete childhood vaccine series so that she could do medical shadowing and volunteering. In medical school she became a passionate vaccine advocate, eventually persuading her mother to change her mind on the subject in time for the COVID vaccine.
“There’s not one answer to the vaccination dilemma,” she said. “Different approaches are required because there are so many different reasons for it. Based on my own life experience, I try to approach patients where they are – not from a place of data and science. What worked in my own family, and works with my patients, is first to establish trust. If they trust you, they’re more likely to listen. Simply ask their worries and concerns,” Dr. Carpenter said.
“A lot of them haven’t had the opportunity before to sit down with a physician they trust and have their worries listened to. They don’t feel heard in our medical system. So I remind myself that I need to understand my patients first – before inserting myself into the conversation.”
Many patients she sees are in an information bubble, with a very different understanding of the issue than their doctors. “A lot of well-meaning people feel they are making the safer choice. Very few truly don’t care about protecting others. But they don’t feel the urgency about that and see the vaccine as the scarier option right now.”
Frontline vaccine advocates
Hospitalists are the frontline advocates within their hospital system, in a position to lead, so they need to make vaccines a priority, Dr. Carpenter said. They should also make sure that their hospitals have ready access to the vaccine, so patients who agree to receive it are able to get it quickly. “In our hospital they can get the shot within a few hours if the opportunity arises. We stocked the Johnson & Johnson vaccine so that they wouldn’t have to connect with another health care provider in order to get a second dose.”
Hospitals should also invest in access to vaccine counseling training and personnel. “Fund a nurse clinician who can screen and counsel hospitalized patients for vaccination. If they meet resistance, they can then refer to the dedicated physician of the day to have the conversation,” she said. “But if we don’t mention it, patients will assume we don’t feel strongly about it.”
Because hospitalists are front and center in treating COVID, they need to be the experts and the people offering guidance, said Shyam Odeti, MD, SFHM, FAAFP, section chief for hospital medicine at the Carilion Clinic in Roanoke, Va. “What we’re trying to do is spread awareness. We educated physician groups, learners, and clinical teams during the initial phase, and now mostly patients and their families.” COVID vaccine reluctance is hard to overcome, Dr. Odeti said. People feel the vaccine was developed very quickly. But there are different ways to present it.
“Like most doctors, I thought people would jump on a vaccine to get past the pandemic. I was surprised and then disappointed. Right now, the pandemic is among the unvaccinated. So we face these encounters, and we’re doing our best to overcome the misinformation. My organization is 100% supportive. We talk about these issues every day.”
Carilion, effective Oct. 1, has required unvaccinated employees to get weekly COVID tests and wear an N95 mask while working, and has developed Facebook pages, other social media, and an Internet presence to address these issues. “We’ve gone to the local African-American community with physician leaders active in that community. We had a Spanish language roundtable,” Dr. Odeti said.
Dr. Skandhan reported that the Wiregrass regional chapter of SHM recently organized a successful statewide community educational event aimed at empowering community leaders to address vaccine misinformation and mistrust. “We surveyed religious leaders and pastors regarding the causes of vaccine hesitancy and reached out to physicians active in community awareness.” Based on that input, a presentation by the faith leaders was developed. Legislators from the Alabama State Senate’s Healthcare Policy Committee were also invited to the presentation and discussion.
Trying to stay positive
It’s important to try to stay positive, Dr. Odeti said. “We have to be empathetic with every patient. We have to keep working at this, since there’s no way out of the pandemic except through vaccinations. But it all creates stress for hospitalists. Our job is made significantly more difficult by the vaccine controversy.”
Jennifer Cowart, MD, a hospitalist at Mayo Clinic in Jacksonville, Fla., has been outspoken in her community about vaccination and masking issues, talking to reporters, attending rallies and press conferences, posting on social media, and speaking in favor of mask policies at a local school board meeting. She is part of an informal local group called Doctors Fighting COVID, which meets online to strategize how to share its expertise, including writing a recent letter about masks to Jacksonville’s mayor.
“In July, when we saw the Delta variant surging locally, we held a webinar via local media, taking calls about the vaccine from the community. I’m trying not to make this a political issue, but we are health officials.” Dr. Cowart said she also tries not to raise her voice when speaking with vaccine opponents and tries to remain empathetic. “Even though inwardly I’m screaming, I try to stay calm. The misinformation is real. People are afraid and feeling pressure. I do my best, but I’m human, too.”
Hospitalists need to pull whatever levers they can to help advance understanding of vaccines, Dr. Cowart said. “In the hospital, our biggest issue is time. We often don’t have it, with a long list of patients to see. But every patient encounter is an opportunity to talk to patients, whether they have COVID or something else.” Sometimes, she might go back to a patient’s room after rounds to resume the conversation.
Hospital nurses have been trained and entrusted to do tobacco abatement counseling, she said, so why not mobilize them for vaccine education? “Or respiratory therapists, who do inhaler training, could talk about what it’s like to care for COVID patients. There’s a whole bunch of staff in the hospital who could be mobilized,” she said.
“I feel passionate about vaccines, as a hospitalist, as a medical educator, as a daughter, as a responsible member of society,” said Eileen Barrett, MD, MPH, SFHM, MACP, director of continuing medical education at the University of New Mexico, Albuquerque. “I see this as a personal and societal responsibility. When I speak about the vaccine among groups of doctors, I say we need to stay in our lane regarding our skills at interpreting the science and not undermining it.”
Some health care worker hesitancy is from distrust of pharmaceutical companies, or of federal agencies, she said. “Our research has highlighted to me the widespread inequity issues in our health care system. We should also take a long, hard look at how we teach the scientific method to health professionals. That will be part of a pandemic retrospective.”
Sometimes with people who are vaccine deliberative, whether health care workers or patients, there is a small window of opportunity. “We need to hear people and respond to them as people. Then, if they are willing to get vaccinated, we need to accomplish that as quickly and easily as possible,” Dr. Barrett said. “I see them make a face and say, ‘Well, okay, I’ll do it.’ We need to get the vaccine to them that same day. We should be able to accomplish that.”
References
1. Gamble M. 30% of US hospitals mandate vaccination for employment. Becker’s Hospital Review. 2021 Aug 13. www.beckershospitalreview.com/workforce/covid-19-vaccination-needed-to-work-at-30-of-us-hospitals.html .
2. Society of Hospital Medicine signs on to joint statement in support of health worker COVID-19 vaccine mandates. Press release. 2021 Jul 26. www.hospitalmedicine.org/news-publications/press-releases/society-of-hospital-medicine-signs-on-to-joint-statement-of-support-of-health-worker-covid-19-vaccine-mandates/.
3. Carpenter A. A physician’s lessons from an unvaccinated childhood. Louisville Medicine. 2021 July;69(2):26-7. https://viewer.joomag.com/louisville-medicine-volume-69-issue-2/0045988001624974172?short&.
Lessons for hospitalists from the vaccination controversy
1. Remain up-to-date on information about the COVID infection, its treatment, and vaccination efficacy data.
2. Hospitalists should take advantage of their positions to lead conversations in their facilities about the importance of COVID vaccinations.
3. Other professionals in the hospital, with some additional training and support, could take on the role of providing vaccine education and support – with a physician to back them up on difficult cases.
4. It’s important to listen to people’s concerns, try to build trust, and establish dialogue before starting to convey a lot of information. People need to feel heard.
5. If you are successful in persuading someone to take the vaccine, a shot should be promptly and easily accessible to them.
6. Pediatric hospitalists may have more experience and skill with vaccine discussions, which they should share with their peers who treat adults.
Flu shot highly recommended this year
With the Delta variant of COVID-19 still raging in the United States and ICUs in parts of the country filled with patients with the coronavirus, experts are voicing concern about the added risk of a difficult flu season.
Two mathematical models are predicting a big rebound in the number and severity of flu cases in the 2021-22 season after 2020-2021’s flu season failed to show up when public health measures brought in to control COVID-19 seemed to have the added benefit of stopping the flu.
But both analyses, posted to the medRxiv preprint server and not yet peer reviewed by other experts, have come to the same conclusion: The flu could make a comeback this year.
In the worst-case scenario, the United States could see an extra 300,000-400,000 hospitalizations from the flu – almost double the usual number – according to senior study author Mark Roberts, MD, director of the Public Health Dynamics Laboratory at the University of Pittsburgh. These numbers could be a disaster in areas where hospitals are already filled with COVID-19 patients.
Waning natural immunity in the public because of 2020-2021’s missing flu season could make people, especially young children, more likely to get the virus.
“Usually, a combination of natural immunity and vaccination helps tamp down seasonal influenza,” said Dr. Roberts. “If we don’t have the first part, we’ll have to rely more on the vaccine.”
In a typical year, about half of Americans get the flu shot. The new mathematical models predict that the vaccination rate would need to rise to about 75% to avoid the extra hospitalizations. But even a 10% increase in vaccination rates could reduce hospitalizations by 6%-46%, depending on what strains are dominant.
Usually, the Southern Hemisphere flu season, from February to August, helps show what the Northern Hemisphere can expect over the coming winter. But with strict COVID-19 measures and limits on international travel still in place in countries like Australia and New Zealand and much of South America, it has been another record-low year for flu infections, said Ian Barr, PhD, deputy director of the World Health Organization’s Collaborating Center for Reference and Research on Influenza in Melbourne.
Australia detected only around 500 cases in 2021, compared with about 300,000 in a normal year, and recorded no hospitalizations or deaths from the flu. New Zealand recorded just two cases.
“I’ve never seen anything like this,” Dr. Barr said.
In Australia, the mild flu season led to fewer people getting their flu shot than usual. The rate fell from around 50% to just 33%, said Dr. Barr. “If that happens in the U.S., the population will be even more vulnerable because there has been almost no flu for more than 12 months,” he said.
Both Dr. Roberts and Dr. Barr say it is vital that as many people as possible get vaccinated during the upcoming flu season, especially children who will have almost no natural immunity to the virus.
“The vaccine is our best weapon against the flu, especially for the most at-risk groups,” said Dr. Barr.
Other parts of the world had mixed results. India saw a high number of flu cases, while neighboring Sri Lanka had very few. West Africa also saw quite a high level of circulating virus. Overall, the flu was detected in 45 countries during the Southern Hemisphere season, less than half of what might be expected in a normal year, said Dr. Barr.
Despite the overall low numbers, the WHO saw enough in the data to make two changes to 2022’s Southern Hemisphere vaccine formulation at its meeting on Sept. 24, after changing just one of the strains for the Northern Hemisphere vaccine at its meeting in February.
The CDC recommends that everyone 6 months or older get the flu shot, with few exceptions.
A version of this article first appeared on WebMD.com.
With the Delta variant of COVID-19 still raging in the United States and ICUs in parts of the country filled with patients with the coronavirus, experts are voicing concern about the added risk of a difficult flu season.
Two mathematical models are predicting a big rebound in the number and severity of flu cases in the 2021-22 season after 2020-2021’s flu season failed to show up when public health measures brought in to control COVID-19 seemed to have the added benefit of stopping the flu.
But both analyses, posted to the medRxiv preprint server and not yet peer reviewed by other experts, have come to the same conclusion: The flu could make a comeback this year.
In the worst-case scenario, the United States could see an extra 300,000-400,000 hospitalizations from the flu – almost double the usual number – according to senior study author Mark Roberts, MD, director of the Public Health Dynamics Laboratory at the University of Pittsburgh. These numbers could be a disaster in areas where hospitals are already filled with COVID-19 patients.
Waning natural immunity in the public because of 2020-2021’s missing flu season could make people, especially young children, more likely to get the virus.
“Usually, a combination of natural immunity and vaccination helps tamp down seasonal influenza,” said Dr. Roberts. “If we don’t have the first part, we’ll have to rely more on the vaccine.”
In a typical year, about half of Americans get the flu shot. The new mathematical models predict that the vaccination rate would need to rise to about 75% to avoid the extra hospitalizations. But even a 10% increase in vaccination rates could reduce hospitalizations by 6%-46%, depending on what strains are dominant.
Usually, the Southern Hemisphere flu season, from February to August, helps show what the Northern Hemisphere can expect over the coming winter. But with strict COVID-19 measures and limits on international travel still in place in countries like Australia and New Zealand and much of South America, it has been another record-low year for flu infections, said Ian Barr, PhD, deputy director of the World Health Organization’s Collaborating Center for Reference and Research on Influenza in Melbourne.
Australia detected only around 500 cases in 2021, compared with about 300,000 in a normal year, and recorded no hospitalizations or deaths from the flu. New Zealand recorded just two cases.
“I’ve never seen anything like this,” Dr. Barr said.
In Australia, the mild flu season led to fewer people getting their flu shot than usual. The rate fell from around 50% to just 33%, said Dr. Barr. “If that happens in the U.S., the population will be even more vulnerable because there has been almost no flu for more than 12 months,” he said.
Both Dr. Roberts and Dr. Barr say it is vital that as many people as possible get vaccinated during the upcoming flu season, especially children who will have almost no natural immunity to the virus.
“The vaccine is our best weapon against the flu, especially for the most at-risk groups,” said Dr. Barr.
Other parts of the world had mixed results. India saw a high number of flu cases, while neighboring Sri Lanka had very few. West Africa also saw quite a high level of circulating virus. Overall, the flu was detected in 45 countries during the Southern Hemisphere season, less than half of what might be expected in a normal year, said Dr. Barr.
Despite the overall low numbers, the WHO saw enough in the data to make two changes to 2022’s Southern Hemisphere vaccine formulation at its meeting on Sept. 24, after changing just one of the strains for the Northern Hemisphere vaccine at its meeting in February.
The CDC recommends that everyone 6 months or older get the flu shot, with few exceptions.
A version of this article first appeared on WebMD.com.
With the Delta variant of COVID-19 still raging in the United States and ICUs in parts of the country filled with patients with the coronavirus, experts are voicing concern about the added risk of a difficult flu season.
Two mathematical models are predicting a big rebound in the number and severity of flu cases in the 2021-22 season after 2020-2021’s flu season failed to show up when public health measures brought in to control COVID-19 seemed to have the added benefit of stopping the flu.
But both analyses, posted to the medRxiv preprint server and not yet peer reviewed by other experts, have come to the same conclusion: The flu could make a comeback this year.
In the worst-case scenario, the United States could see an extra 300,000-400,000 hospitalizations from the flu – almost double the usual number – according to senior study author Mark Roberts, MD, director of the Public Health Dynamics Laboratory at the University of Pittsburgh. These numbers could be a disaster in areas where hospitals are already filled with COVID-19 patients.
Waning natural immunity in the public because of 2020-2021’s missing flu season could make people, especially young children, more likely to get the virus.
“Usually, a combination of natural immunity and vaccination helps tamp down seasonal influenza,” said Dr. Roberts. “If we don’t have the first part, we’ll have to rely more on the vaccine.”
In a typical year, about half of Americans get the flu shot. The new mathematical models predict that the vaccination rate would need to rise to about 75% to avoid the extra hospitalizations. But even a 10% increase in vaccination rates could reduce hospitalizations by 6%-46%, depending on what strains are dominant.
Usually, the Southern Hemisphere flu season, from February to August, helps show what the Northern Hemisphere can expect over the coming winter. But with strict COVID-19 measures and limits on international travel still in place in countries like Australia and New Zealand and much of South America, it has been another record-low year for flu infections, said Ian Barr, PhD, deputy director of the World Health Organization’s Collaborating Center for Reference and Research on Influenza in Melbourne.
Australia detected only around 500 cases in 2021, compared with about 300,000 in a normal year, and recorded no hospitalizations or deaths from the flu. New Zealand recorded just two cases.
“I’ve never seen anything like this,” Dr. Barr said.
In Australia, the mild flu season led to fewer people getting their flu shot than usual. The rate fell from around 50% to just 33%, said Dr. Barr. “If that happens in the U.S., the population will be even more vulnerable because there has been almost no flu for more than 12 months,” he said.
Both Dr. Roberts and Dr. Barr say it is vital that as many people as possible get vaccinated during the upcoming flu season, especially children who will have almost no natural immunity to the virus.
“The vaccine is our best weapon against the flu, especially for the most at-risk groups,” said Dr. Barr.
Other parts of the world had mixed results. India saw a high number of flu cases, while neighboring Sri Lanka had very few. West Africa also saw quite a high level of circulating virus. Overall, the flu was detected in 45 countries during the Southern Hemisphere season, less than half of what might be expected in a normal year, said Dr. Barr.
Despite the overall low numbers, the WHO saw enough in the data to make two changes to 2022’s Southern Hemisphere vaccine formulation at its meeting on Sept. 24, after changing just one of the strains for the Northern Hemisphere vaccine at its meeting in February.
The CDC recommends that everyone 6 months or older get the flu shot, with few exceptions.
A version of this article first appeared on WebMD.com.
Worried parents scramble to vaccinate kids despite FDA guidance
One week after reporting promising results from the trial of their COVID-19 vaccine in children ages 5-11, Pfizer and BioNTech announced they’d submitted the data to the Food and Drug Administration. But that hasn’t stopped some parents from discreetly getting their children under age 12 vaccinated.
“The FDA, you never want to get ahead of their judgment,” Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, told MSNBC on Sept. 28. “But I would imagine in the next few weeks, they will examine that data and hopefully they’ll give the okay so that we can start vaccinating children, hopefully before the end of October.”
Lying to vaccinate now
More than half of all parents with children under 12 say they plan to get their kids vaccinated, according to a Gallup poll.
And although the FDA and the American Academy of Pediatrics have warned against it, some parents whose children can pass for 12 have lied to get them vaccinated already.
Dawn G. is a mom of two in southwest Missouri, where less than 45% of the population has been fully vaccinated. Her son turns 12 in early October, but in-person school started in mid-August.
“It was scary, thinking of him going to school for even 2 months,” she said. “Some parents thought their kid had a low chance of getting COVID, and their kid died. Nobody expects it to be them.”
In July, she and her husband took their son to a walk-in clinic and lied about his age.
“So many things can happen, from bullying to school shootings, and now this added pandemic risk,” she said. “I’ll do anything I can to protect my child, and a birthdate seems so arbitrary. He’ll be 12 in a matter of weeks. It seems ridiculous that that date would stop me from protecting him.”
In northern California, Carrie S. had a similar thought. When the vaccine was authorized for children ages 12-15 in May, the older of her two children got the shot right away. But her youngest doesn’t turn 12 until November.
“We were tempted to get the younger one vaccinated in May, but it didn’t seem like a rush. We were willing to wait to get the dosage right,” she ssaid. “But as Delta came through, there were no options for online school, the CDC was dropping mask expectations –it seemed like the world was ready to forget the pandemic was happening. It seemed like the least-bad option to get her vaccinated so she could go back to school, and we could find some balance of risk in our lives.”
Adult vs. pediatric doses
For now, experts advise against getting younger children vaccinated, even those who are the size of an adult, because of the way the human immune system develops.
“It’s not really about size,” said Anne Liu, MD, an immunologist and pediatrics professor at Stanford (Calif.) University. “The immune system behaves differently at different ages. Younger kids tend to have a more exuberant innate immune system, which is the part of the immune system that senses danger, even before it has developed a memory response.”
The adult Pfizer-BioNTech vaccine contains 30 mcg of mRNA, while the pediatric dose is just 10 mcg. That smaller dose produces an immune response similar to what’s seen in adults who receive 30 mcg, according to Pfizer.
“We were one of the sites that was involved in the phase 1 trial, a lot of times that’s called a dose-finding trial,” said Michael Smith, MD, a coinvestigator for the COVID vaccine trials done at Duke University. “And basically, if younger kids got a higher dose, they had more of a reaction, so it hurt more. They had fever, they had more redness and swelling at the site of the injection, and they just felt lousy, more than at the lower doses.”
At this point, with Pfizer’s data showing that younger children need a smaller dose, it doesn’t make sense to lie about your child’s age, said Dr. Smith.
“If my two options were having my child get the infection versus getting the vaccine, I’d get the vaccine. But we’re a few weeks away from getting the lower dose approved in kids,” he said. “It’s certainly safer. I don’t expect major, lifelong side effects from the higher dose, but it’s going to hurt, your kid’s going to have a fever, they’re going to feel lousy for a couple days, and they just don’t need that much antigen.”
A version of this article first appeared on WebMD.com.
One week after reporting promising results from the trial of their COVID-19 vaccine in children ages 5-11, Pfizer and BioNTech announced they’d submitted the data to the Food and Drug Administration. But that hasn’t stopped some parents from discreetly getting their children under age 12 vaccinated.
“The FDA, you never want to get ahead of their judgment,” Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, told MSNBC on Sept. 28. “But I would imagine in the next few weeks, they will examine that data and hopefully they’ll give the okay so that we can start vaccinating children, hopefully before the end of October.”
Lying to vaccinate now
More than half of all parents with children under 12 say they plan to get their kids vaccinated, according to a Gallup poll.
And although the FDA and the American Academy of Pediatrics have warned against it, some parents whose children can pass for 12 have lied to get them vaccinated already.
Dawn G. is a mom of two in southwest Missouri, where less than 45% of the population has been fully vaccinated. Her son turns 12 in early October, but in-person school started in mid-August.
“It was scary, thinking of him going to school for even 2 months,” she said. “Some parents thought their kid had a low chance of getting COVID, and their kid died. Nobody expects it to be them.”
In July, she and her husband took their son to a walk-in clinic and lied about his age.
“So many things can happen, from bullying to school shootings, and now this added pandemic risk,” she said. “I’ll do anything I can to protect my child, and a birthdate seems so arbitrary. He’ll be 12 in a matter of weeks. It seems ridiculous that that date would stop me from protecting him.”
In northern California, Carrie S. had a similar thought. When the vaccine was authorized for children ages 12-15 in May, the older of her two children got the shot right away. But her youngest doesn’t turn 12 until November.
“We were tempted to get the younger one vaccinated in May, but it didn’t seem like a rush. We were willing to wait to get the dosage right,” she ssaid. “But as Delta came through, there were no options for online school, the CDC was dropping mask expectations –it seemed like the world was ready to forget the pandemic was happening. It seemed like the least-bad option to get her vaccinated so she could go back to school, and we could find some balance of risk in our lives.”
Adult vs. pediatric doses
For now, experts advise against getting younger children vaccinated, even those who are the size of an adult, because of the way the human immune system develops.
“It’s not really about size,” said Anne Liu, MD, an immunologist and pediatrics professor at Stanford (Calif.) University. “The immune system behaves differently at different ages. Younger kids tend to have a more exuberant innate immune system, which is the part of the immune system that senses danger, even before it has developed a memory response.”
The adult Pfizer-BioNTech vaccine contains 30 mcg of mRNA, while the pediatric dose is just 10 mcg. That smaller dose produces an immune response similar to what’s seen in adults who receive 30 mcg, according to Pfizer.
“We were one of the sites that was involved in the phase 1 trial, a lot of times that’s called a dose-finding trial,” said Michael Smith, MD, a coinvestigator for the COVID vaccine trials done at Duke University. “And basically, if younger kids got a higher dose, they had more of a reaction, so it hurt more. They had fever, they had more redness and swelling at the site of the injection, and they just felt lousy, more than at the lower doses.”
At this point, with Pfizer’s data showing that younger children need a smaller dose, it doesn’t make sense to lie about your child’s age, said Dr. Smith.
“If my two options were having my child get the infection versus getting the vaccine, I’d get the vaccine. But we’re a few weeks away from getting the lower dose approved in kids,” he said. “It’s certainly safer. I don’t expect major, lifelong side effects from the higher dose, but it’s going to hurt, your kid’s going to have a fever, they’re going to feel lousy for a couple days, and they just don’t need that much antigen.”
A version of this article first appeared on WebMD.com.
One week after reporting promising results from the trial of their COVID-19 vaccine in children ages 5-11, Pfizer and BioNTech announced they’d submitted the data to the Food and Drug Administration. But that hasn’t stopped some parents from discreetly getting their children under age 12 vaccinated.
“The FDA, you never want to get ahead of their judgment,” Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, told MSNBC on Sept. 28. “But I would imagine in the next few weeks, they will examine that data and hopefully they’ll give the okay so that we can start vaccinating children, hopefully before the end of October.”
Lying to vaccinate now
More than half of all parents with children under 12 say they plan to get their kids vaccinated, according to a Gallup poll.
And although the FDA and the American Academy of Pediatrics have warned against it, some parents whose children can pass for 12 have lied to get them vaccinated already.
Dawn G. is a mom of two in southwest Missouri, where less than 45% of the population has been fully vaccinated. Her son turns 12 in early October, but in-person school started in mid-August.
“It was scary, thinking of him going to school for even 2 months,” she said. “Some parents thought their kid had a low chance of getting COVID, and their kid died. Nobody expects it to be them.”
In July, she and her husband took their son to a walk-in clinic and lied about his age.
“So many things can happen, from bullying to school shootings, and now this added pandemic risk,” she said. “I’ll do anything I can to protect my child, and a birthdate seems so arbitrary. He’ll be 12 in a matter of weeks. It seems ridiculous that that date would stop me from protecting him.”
In northern California, Carrie S. had a similar thought. When the vaccine was authorized for children ages 12-15 in May, the older of her two children got the shot right away. But her youngest doesn’t turn 12 until November.
“We were tempted to get the younger one vaccinated in May, but it didn’t seem like a rush. We were willing to wait to get the dosage right,” she ssaid. “But as Delta came through, there were no options for online school, the CDC was dropping mask expectations –it seemed like the world was ready to forget the pandemic was happening. It seemed like the least-bad option to get her vaccinated so she could go back to school, and we could find some balance of risk in our lives.”
Adult vs. pediatric doses
For now, experts advise against getting younger children vaccinated, even those who are the size of an adult, because of the way the human immune system develops.
“It’s not really about size,” said Anne Liu, MD, an immunologist and pediatrics professor at Stanford (Calif.) University. “The immune system behaves differently at different ages. Younger kids tend to have a more exuberant innate immune system, which is the part of the immune system that senses danger, even before it has developed a memory response.”
The adult Pfizer-BioNTech vaccine contains 30 mcg of mRNA, while the pediatric dose is just 10 mcg. That smaller dose produces an immune response similar to what’s seen in adults who receive 30 mcg, according to Pfizer.
“We were one of the sites that was involved in the phase 1 trial, a lot of times that’s called a dose-finding trial,” said Michael Smith, MD, a coinvestigator for the COVID vaccine trials done at Duke University. “And basically, if younger kids got a higher dose, they had more of a reaction, so it hurt more. They had fever, they had more redness and swelling at the site of the injection, and they just felt lousy, more than at the lower doses.”
At this point, with Pfizer’s data showing that younger children need a smaller dose, it doesn’t make sense to lie about your child’s age, said Dr. Smith.
“If my two options were having my child get the infection versus getting the vaccine, I’d get the vaccine. But we’re a few weeks away from getting the lower dose approved in kids,” he said. “It’s certainly safer. I don’t expect major, lifelong side effects from the higher dose, but it’s going to hurt, your kid’s going to have a fever, they’re going to feel lousy for a couple days, and they just don’t need that much antigen.”
A version of this article first appeared on WebMD.com.
Children and COVID: New cases topped 200,000 after 3 weeks of declines
Weekly COVID-19 cases in children dropped again, but the count remained above 200,000 for the fifth consecutive week, according to the American Academy of Pediatrics and the Children’s Hospital Association.
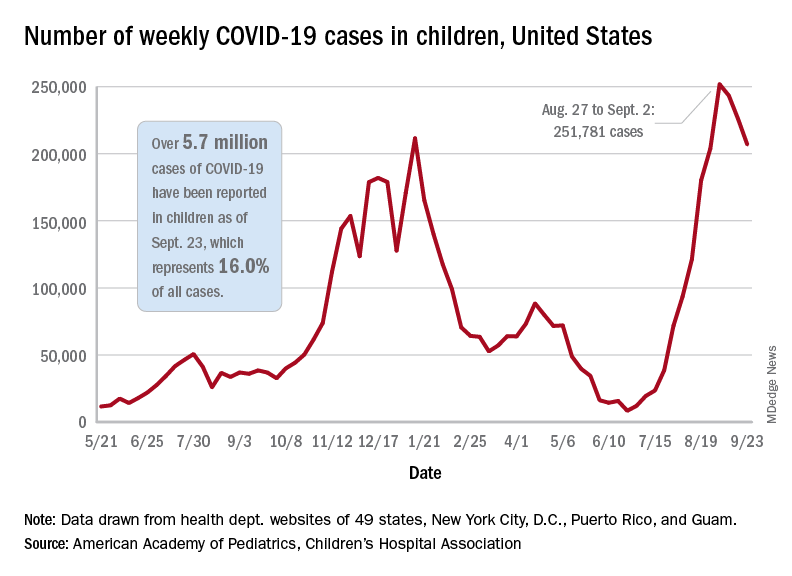
based on the data in the AAP/CHA joint weekly report on COVID in children.
In the most recent week, Sept. 17-23, there were almost 207,000 new cases of COVID-19 in children, which represented 26.7% of all cases reported in the 46 states that are currently posting data by age on their COVID dashboards, the AAP and CHA said. (New York has never reported such data by age, and Alabama, Nebraska, and Texas have not updated their websites since July 29, June 24, and Aug. 26, respectively.)
The decline in new vaccinations among children, however, began before the summer surge in new cases hit its peak – 251,781 during the week of Aug. 27 to Sept. 2 – and has continued for 7 straight weeks in children aged 12-17 years, based on data from the Centers for Disease Control and Prevention.
There were about 172,000 COVID vaccine initiations in children aged 12-17 for the week of Sept. 21-27, the lowest number since April, before it was approved for use in 12- to 15-year-olds. That figure is down by almost a third from the previous week and by more than two-thirds since early August, just before the decline in vaccinations began, according to the CDC’s COVID Data Tracker.
The cumulative vaccine situation looks like this: Just over 13 million children under age 18 years have received at least one dose as of Sept. 27, and almost 10.6 million are fully vaccinated. By age group, 53.9% of 12- to 15-year-olds and 61.6% of 16- to 17-year-olds have received at least one dose, with corresponding figures of 43.3% and 51.3% for full vaccination, the CDC said.
COVID-related hospital admissions also continue to fall after peaking at 0.51 children aged 0-17 per 100,000 population on Sept. 4. The admission rate was down to 0.45 per 100,000 as of Sept. 17, and the latest 7-day average (Sept. 19-25) was 258 admissions, compared with a peak of 371 for the week of Aug. 29 to Sept. 4, the CDC reported.
“Although we have seen slight improvements in COVID-19 volumes in the past week, we are at the beginning of an anticipated increase in” multi-inflammatory syndrome in children, Margaret Rush, MD, president of Monroe Carell Jr. Children’s Hospital at Vanderbilt University, Nashville, Tenn., said at a recent hearing of the House Committee on Energy and Commerce’s Oversight subcommittee. That increase would be expected to produce “a secondary wave of seriously ill children 3-6 weeks after acute infection peaks in the community,” the American Hospital Association said.
Meanwhile, Dr. Rush noted, there are signs that seasonal viruses are coming into play. “With the emergence of the Delta variant, we’ve experienced a steep increase in COVID-19 hospitalizations among children on top of an early surge of [respiratory syncytial virus], a serious respiratory illness we usually see in the winter months,” she said in a prepared statement before her testimony.
Weekly COVID-19 cases in children dropped again, but the count remained above 200,000 for the fifth consecutive week, according to the American Academy of Pediatrics and the Children’s Hospital Association.

based on the data in the AAP/CHA joint weekly report on COVID in children.
In the most recent week, Sept. 17-23, there were almost 207,000 new cases of COVID-19 in children, which represented 26.7% of all cases reported in the 46 states that are currently posting data by age on their COVID dashboards, the AAP and CHA said. (New York has never reported such data by age, and Alabama, Nebraska, and Texas have not updated their websites since July 29, June 24, and Aug. 26, respectively.)
The decline in new vaccinations among children, however, began before the summer surge in new cases hit its peak – 251,781 during the week of Aug. 27 to Sept. 2 – and has continued for 7 straight weeks in children aged 12-17 years, based on data from the Centers for Disease Control and Prevention.
There were about 172,000 COVID vaccine initiations in children aged 12-17 for the week of Sept. 21-27, the lowest number since April, before it was approved for use in 12- to 15-year-olds. That figure is down by almost a third from the previous week and by more than two-thirds since early August, just before the decline in vaccinations began, according to the CDC’s COVID Data Tracker.
The cumulative vaccine situation looks like this: Just over 13 million children under age 18 years have received at least one dose as of Sept. 27, and almost 10.6 million are fully vaccinated. By age group, 53.9% of 12- to 15-year-olds and 61.6% of 16- to 17-year-olds have received at least one dose, with corresponding figures of 43.3% and 51.3% for full vaccination, the CDC said.
COVID-related hospital admissions also continue to fall after peaking at 0.51 children aged 0-17 per 100,000 population on Sept. 4. The admission rate was down to 0.45 per 100,000 as of Sept. 17, and the latest 7-day average (Sept. 19-25) was 258 admissions, compared with a peak of 371 for the week of Aug. 29 to Sept. 4, the CDC reported.
“Although we have seen slight improvements in COVID-19 volumes in the past week, we are at the beginning of an anticipated increase in” multi-inflammatory syndrome in children, Margaret Rush, MD, president of Monroe Carell Jr. Children’s Hospital at Vanderbilt University, Nashville, Tenn., said at a recent hearing of the House Committee on Energy and Commerce’s Oversight subcommittee. That increase would be expected to produce “a secondary wave of seriously ill children 3-6 weeks after acute infection peaks in the community,” the American Hospital Association said.
Meanwhile, Dr. Rush noted, there are signs that seasonal viruses are coming into play. “With the emergence of the Delta variant, we’ve experienced a steep increase in COVID-19 hospitalizations among children on top of an early surge of [respiratory syncytial virus], a serious respiratory illness we usually see in the winter months,” she said in a prepared statement before her testimony.
Weekly COVID-19 cases in children dropped again, but the count remained above 200,000 for the fifth consecutive week, according to the American Academy of Pediatrics and the Children’s Hospital Association.

based on the data in the AAP/CHA joint weekly report on COVID in children.
In the most recent week, Sept. 17-23, there were almost 207,000 new cases of COVID-19 in children, which represented 26.7% of all cases reported in the 46 states that are currently posting data by age on their COVID dashboards, the AAP and CHA said. (New York has never reported such data by age, and Alabama, Nebraska, and Texas have not updated their websites since July 29, June 24, and Aug. 26, respectively.)
The decline in new vaccinations among children, however, began before the summer surge in new cases hit its peak – 251,781 during the week of Aug. 27 to Sept. 2 – and has continued for 7 straight weeks in children aged 12-17 years, based on data from the Centers for Disease Control and Prevention.
There were about 172,000 COVID vaccine initiations in children aged 12-17 for the week of Sept. 21-27, the lowest number since April, before it was approved for use in 12- to 15-year-olds. That figure is down by almost a third from the previous week and by more than two-thirds since early August, just before the decline in vaccinations began, according to the CDC’s COVID Data Tracker.
The cumulative vaccine situation looks like this: Just over 13 million children under age 18 years have received at least one dose as of Sept. 27, and almost 10.6 million are fully vaccinated. By age group, 53.9% of 12- to 15-year-olds and 61.6% of 16- to 17-year-olds have received at least one dose, with corresponding figures of 43.3% and 51.3% for full vaccination, the CDC said.
COVID-related hospital admissions also continue to fall after peaking at 0.51 children aged 0-17 per 100,000 population on Sept. 4. The admission rate was down to 0.45 per 100,000 as of Sept. 17, and the latest 7-day average (Sept. 19-25) was 258 admissions, compared with a peak of 371 for the week of Aug. 29 to Sept. 4, the CDC reported.
“Although we have seen slight improvements in COVID-19 volumes in the past week, we are at the beginning of an anticipated increase in” multi-inflammatory syndrome in children, Margaret Rush, MD, president of Monroe Carell Jr. Children’s Hospital at Vanderbilt University, Nashville, Tenn., said at a recent hearing of the House Committee on Energy and Commerce’s Oversight subcommittee. That increase would be expected to produce “a secondary wave of seriously ill children 3-6 weeks after acute infection peaks in the community,” the American Hospital Association said.
Meanwhile, Dr. Rush noted, there are signs that seasonal viruses are coming into play. “With the emergence of the Delta variant, we’ve experienced a steep increase in COVID-19 hospitalizations among children on top of an early surge of [respiratory syncytial virus], a serious respiratory illness we usually see in the winter months,” she said in a prepared statement before her testimony.
Polyethylene glycol linked to rare allergic reactions seen with mRNA COVID-19 vaccines
A common inert ingredient may be the culprit behind the rare allergic reactions reported among individuals who have received mRNA COVID-19 vaccines, according to investigators at a large regional health center that was among the first to administer the shots.
Blood samples from 10 of 11 individuals with suspected allergic reactions reacted to polyethylene glycol (PEG), a component of both the Pfizer and Moderna mRNA vaccines, according to a report in JAMA Network Open.
In total, only 22 individuals had suspected allergic reactions out of nearly 39,000 mRNA COVID-19 vaccine doses administered, the investigators reported, noting that the reactions were generally mild and all fully resolved.
Those findings should be reassuring to individuals who are reticent to sign up for a COVID-19 vaccine because of fear of an allergic reaction, said study senior author Kari Nadeau, MD, PhD, director of the Parker Center for Allergy and Asthma Research at Stanford (Calif.) University.
“We’re hoping that this word will get out and then that the companies could also think about making vaccines that have other products in them that don’t include polyethylene glycol,” Dr. Nadeau said in an interview.
PEG is a compound used in many products, including pharmaceuticals, cosmetics, and food. In the mRNA COVID-19 vaccines, PEG serves to stabilize the lipid nanoparticles that help protect and transport mRNA. However, its use in this setting has been linked to allergic reactions in this and previous studies.
No immunoglobulin E (IgE) antibodies to PEG were detected among the 22 individuals with suspected allergic reactions to mRNA COVID-19 vaccine, but PEG immunoglobulin G (IgG) was present. That suggests non-IgE mediated allergic reactions to PEG may be implicated for the majority of cases, Dr. Nadeau said.
This case series provides interesting new evidence to confirm previous reports that a mechanism other than the classic IgE-mediated allergic response is behind the suspected allergic reactions that are occurring after mRNA COVID-19 vaccine, said Aleena Banerji, MD, associate professor at Harvard Medical School, Boston, and clinical director of the Drug Allergy Program at Massachusetts General Hospital.
“We need to further understand the mechanism of these reactions, but what we know is that IGE mediated allergy to excipients like PEG is probably not the main cause,” Dr. Banerji, who was not involved in the study, said in an interview.
In a recent research letter published in JAMA Internal Medicine, Dr. Banerji and coauthors reported that all individuals with immediate suspected allergic reactions to mRNA COVID-19 vaccine went on to tolerate the second dose, with mild symptoms reported in the minority of patients (32 out of 159, or about 20%).
“Again, that is very consistent with not having an IgE-mediated allergy, so it seems to all be fitting with that picture,” Dr. Banerji said.
The case series by Dr. Nadeau and coauthors was based on review of nearly 39,000 mRNA COVID-19 vaccine doses administered between December 18, 2020 and January 26, 2021. Most mRNA vaccine recipients were Stanford-affiliated health care workers, according to the report.
Among recipients of those doses, they identified 148 individuals who had anaphylaxis-related ICD-10 codes recorded over the same time period. In a review of medical records, investigators pinpointed 22 individuals as having suspected allergy and invited them to participate in follow-up allergy testing.
A total of 11 individuals underwent skin prick testing, but none of them tested positive to PEG or to polysorbate 80, another excipient that has been linked to vaccine-related allergic reactions. One of the patients tested positive to the same mRNA vaccine they had previously received, according to the report.
Those same 11 individuals also underwent basophil activation testing (BAT). In contrast to the skin testing results, BAT results were positive for PEG in 10 of 11 cases (or 91%) and positive for their administered vaccine in all 11 cases, the report shows.
High levels of IgG to PEG were identified in blood samples of individuals with an allergy to the vaccine. Investigators said it’s possible that the BAT results were activated due to IgG via complement activation–related pseudoallergy, or CARPA, as has been hypothesized by some other investigators.
The negative skin prick testing results for PEG, which contrast with the positive BAT results to PEG, suggest that the former may not be appropriate for use as a predictive marker of potential vaccine allergy, according to Dr. Nadeau.
“The take-home message for doctors is to be careful,” she said. “Don’t assume that just because the person skin-tests negative to PEG or to the vaccine itself that you’re out of the woods, because the skin test would be often negative in those scenarios.”
The study was supported by a grants from the Asthma and Allergic Diseases Cooperative Research Centers, a grant from the National Institutes of Health, the National Institute of Allergy and Infectious Disease SARS Vaccine study, the Parker Foundation, the Crown Foundation, and the Sunshine Foundation. Dr. Nadeau reports numerous conflicts with various sources in the industry. Dr. Banerji has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A common inert ingredient may be the culprit behind the rare allergic reactions reported among individuals who have received mRNA COVID-19 vaccines, according to investigators at a large regional health center that was among the first to administer the shots.
Blood samples from 10 of 11 individuals with suspected allergic reactions reacted to polyethylene glycol (PEG), a component of both the Pfizer and Moderna mRNA vaccines, according to a report in JAMA Network Open.
In total, only 22 individuals had suspected allergic reactions out of nearly 39,000 mRNA COVID-19 vaccine doses administered, the investigators reported, noting that the reactions were generally mild and all fully resolved.
Those findings should be reassuring to individuals who are reticent to sign up for a COVID-19 vaccine because of fear of an allergic reaction, said study senior author Kari Nadeau, MD, PhD, director of the Parker Center for Allergy and Asthma Research at Stanford (Calif.) University.
“We’re hoping that this word will get out and then that the companies could also think about making vaccines that have other products in them that don’t include polyethylene glycol,” Dr. Nadeau said in an interview.
PEG is a compound used in many products, including pharmaceuticals, cosmetics, and food. In the mRNA COVID-19 vaccines, PEG serves to stabilize the lipid nanoparticles that help protect and transport mRNA. However, its use in this setting has been linked to allergic reactions in this and previous studies.
No immunoglobulin E (IgE) antibodies to PEG were detected among the 22 individuals with suspected allergic reactions to mRNA COVID-19 vaccine, but PEG immunoglobulin G (IgG) was present. That suggests non-IgE mediated allergic reactions to PEG may be implicated for the majority of cases, Dr. Nadeau said.
This case series provides interesting new evidence to confirm previous reports that a mechanism other than the classic IgE-mediated allergic response is behind the suspected allergic reactions that are occurring after mRNA COVID-19 vaccine, said Aleena Banerji, MD, associate professor at Harvard Medical School, Boston, and clinical director of the Drug Allergy Program at Massachusetts General Hospital.
“We need to further understand the mechanism of these reactions, but what we know is that IGE mediated allergy to excipients like PEG is probably not the main cause,” Dr. Banerji, who was not involved in the study, said in an interview.
In a recent research letter published in JAMA Internal Medicine, Dr. Banerji and coauthors reported that all individuals with immediate suspected allergic reactions to mRNA COVID-19 vaccine went on to tolerate the second dose, with mild symptoms reported in the minority of patients (32 out of 159, or about 20%).
“Again, that is very consistent with not having an IgE-mediated allergy, so it seems to all be fitting with that picture,” Dr. Banerji said.
The case series by Dr. Nadeau and coauthors was based on review of nearly 39,000 mRNA COVID-19 vaccine doses administered between December 18, 2020 and January 26, 2021. Most mRNA vaccine recipients were Stanford-affiliated health care workers, according to the report.
Among recipients of those doses, they identified 148 individuals who had anaphylaxis-related ICD-10 codes recorded over the same time period. In a review of medical records, investigators pinpointed 22 individuals as having suspected allergy and invited them to participate in follow-up allergy testing.
A total of 11 individuals underwent skin prick testing, but none of them tested positive to PEG or to polysorbate 80, another excipient that has been linked to vaccine-related allergic reactions. One of the patients tested positive to the same mRNA vaccine they had previously received, according to the report.
Those same 11 individuals also underwent basophil activation testing (BAT). In contrast to the skin testing results, BAT results were positive for PEG in 10 of 11 cases (or 91%) and positive for their administered vaccine in all 11 cases, the report shows.
High levels of IgG to PEG were identified in blood samples of individuals with an allergy to the vaccine. Investigators said it’s possible that the BAT results were activated due to IgG via complement activation–related pseudoallergy, or CARPA, as has been hypothesized by some other investigators.
The negative skin prick testing results for PEG, which contrast with the positive BAT results to PEG, suggest that the former may not be appropriate for use as a predictive marker of potential vaccine allergy, according to Dr. Nadeau.
“The take-home message for doctors is to be careful,” she said. “Don’t assume that just because the person skin-tests negative to PEG or to the vaccine itself that you’re out of the woods, because the skin test would be often negative in those scenarios.”
The study was supported by a grants from the Asthma and Allergic Diseases Cooperative Research Centers, a grant from the National Institutes of Health, the National Institute of Allergy and Infectious Disease SARS Vaccine study, the Parker Foundation, the Crown Foundation, and the Sunshine Foundation. Dr. Nadeau reports numerous conflicts with various sources in the industry. Dr. Banerji has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A common inert ingredient may be the culprit behind the rare allergic reactions reported among individuals who have received mRNA COVID-19 vaccines, according to investigators at a large regional health center that was among the first to administer the shots.
Blood samples from 10 of 11 individuals with suspected allergic reactions reacted to polyethylene glycol (PEG), a component of both the Pfizer and Moderna mRNA vaccines, according to a report in JAMA Network Open.
In total, only 22 individuals had suspected allergic reactions out of nearly 39,000 mRNA COVID-19 vaccine doses administered, the investigators reported, noting that the reactions were generally mild and all fully resolved.
Those findings should be reassuring to individuals who are reticent to sign up for a COVID-19 vaccine because of fear of an allergic reaction, said study senior author Kari Nadeau, MD, PhD, director of the Parker Center for Allergy and Asthma Research at Stanford (Calif.) University.
“We’re hoping that this word will get out and then that the companies could also think about making vaccines that have other products in them that don’t include polyethylene glycol,” Dr. Nadeau said in an interview.
PEG is a compound used in many products, including pharmaceuticals, cosmetics, and food. In the mRNA COVID-19 vaccines, PEG serves to stabilize the lipid nanoparticles that help protect and transport mRNA. However, its use in this setting has been linked to allergic reactions in this and previous studies.
No immunoglobulin E (IgE) antibodies to PEG were detected among the 22 individuals with suspected allergic reactions to mRNA COVID-19 vaccine, but PEG immunoglobulin G (IgG) was present. That suggests non-IgE mediated allergic reactions to PEG may be implicated for the majority of cases, Dr. Nadeau said.
This case series provides interesting new evidence to confirm previous reports that a mechanism other than the classic IgE-mediated allergic response is behind the suspected allergic reactions that are occurring after mRNA COVID-19 vaccine, said Aleena Banerji, MD, associate professor at Harvard Medical School, Boston, and clinical director of the Drug Allergy Program at Massachusetts General Hospital.
“We need to further understand the mechanism of these reactions, but what we know is that IGE mediated allergy to excipients like PEG is probably not the main cause,” Dr. Banerji, who was not involved in the study, said in an interview.
In a recent research letter published in JAMA Internal Medicine, Dr. Banerji and coauthors reported that all individuals with immediate suspected allergic reactions to mRNA COVID-19 vaccine went on to tolerate the second dose, with mild symptoms reported in the minority of patients (32 out of 159, or about 20%).
“Again, that is very consistent with not having an IgE-mediated allergy, so it seems to all be fitting with that picture,” Dr. Banerji said.
The case series by Dr. Nadeau and coauthors was based on review of nearly 39,000 mRNA COVID-19 vaccine doses administered between December 18, 2020 and January 26, 2021. Most mRNA vaccine recipients were Stanford-affiliated health care workers, according to the report.
Among recipients of those doses, they identified 148 individuals who had anaphylaxis-related ICD-10 codes recorded over the same time period. In a review of medical records, investigators pinpointed 22 individuals as having suspected allergy and invited them to participate in follow-up allergy testing.
A total of 11 individuals underwent skin prick testing, but none of them tested positive to PEG or to polysorbate 80, another excipient that has been linked to vaccine-related allergic reactions. One of the patients tested positive to the same mRNA vaccine they had previously received, according to the report.
Those same 11 individuals also underwent basophil activation testing (BAT). In contrast to the skin testing results, BAT results were positive for PEG in 10 of 11 cases (or 91%) and positive for their administered vaccine in all 11 cases, the report shows.
High levels of IgG to PEG were identified in blood samples of individuals with an allergy to the vaccine. Investigators said it’s possible that the BAT results were activated due to IgG via complement activation–related pseudoallergy, or CARPA, as has been hypothesized by some other investigators.
The negative skin prick testing results for PEG, which contrast with the positive BAT results to PEG, suggest that the former may not be appropriate for use as a predictive marker of potential vaccine allergy, according to Dr. Nadeau.
“The take-home message for doctors is to be careful,” she said. “Don’t assume that just because the person skin-tests negative to PEG or to the vaccine itself that you’re out of the woods, because the skin test would be often negative in those scenarios.”
The study was supported by a grants from the Asthma and Allergic Diseases Cooperative Research Centers, a grant from the National Institutes of Health, the National Institute of Allergy and Infectious Disease SARS Vaccine study, the Parker Foundation, the Crown Foundation, and the Sunshine Foundation. Dr. Nadeau reports numerous conflicts with various sources in the industry. Dr. Banerji has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Updates to CDC’s STI guidelines relevant to midlife women too
Sexually transmitted infection rates have not increased as dramatically in older women as they have in women in their teens and 20s, but rates of chlamydia and gonorrhea in women over age 35 have seen a steady incline over the past decade, and syphilis rates have climbed steeply, according to data from the Centers for Disease Control and Prevention.
That makes the STI treatment guidelines released by the CDC in July even timelier for practitioners of menopause medicine, according to Michael S. Policar, MD, MPH, a professor emeritus of ob.gyn. and reproductive sciences at the University of California, San Francisco.
Dr. Policar discussed what clinicians need to know about STIs in midlife women at the hybrid annual meeting of the North American Menopause Society. Even the nomenclature change in the guidelines from “sexually transmitted diseases” to “sexually transmitted infections” is important “because they want to acknowledge the fact that a lot of the sexually transmitted infections that we’re treating are asymptomatic, are colonizations, and are not yet diseases,” Dr. Policar said. “We’re trying to be much more expansive in thinking about finding these infections before they actually start causing morbidity in the form of a disease.”
Sexual history
The primary guidelines update for taking sexual history is the recommendation to ask patients about their intentions regarding pregnancy. The “5 Ps” of sexual history are now Partners, Practices, Protection from STIs, Past history of STIs, and Pregnancy intention.
“There should be a sixth P that has to do with pleasure questions,” Policar added. “We ask all the time for patients that we see in the context of perimenopausal and menopausal services, ‘Are you satisfied with your sexual relationship with your partner?’ Hopefully that will make it into the CDC guidelines as the sixth P at some point, but for now, that’s aspirational.”
In asking about partners, instead of asking patients whether they have sex with men, women, or both, clinicians should ask first if the patient is having sex of any kind – oral, vaginal, or anal – with anyone. From there, providers should ask how many sex partners the patient has had, the gender(s) of the partners, and whether they or their partners have other sex partners, using more gender-inclusive language.
When asking about practices, in addition to asking about the type of sexual contact patients have had, additional questions include whether the patient met their partners online or through apps, whether they or any of their partners use drugs, and whether the patient has exchanged sex for any needs, such as money, housing, or drugs. The additional questions can identify those at higher risk for STIs.
After reviewing the CDC’s list of risk factors for gonorrhea and chlamydia screening, Dr. Policar shared the screening list from the California Department of Public Health, which he finds more helpful:
- History of gonorrhea, chlamydia, or pelvic inflammatory disease (PID) in the past 2 years.
- More than 1 sexual partner in the past year.
- New sexual partner within 90 days.
- Reason to believe that a sex partner has had other partners in the past year.
- Exchanging sex for drugs or money within the past year.
- Other factors identified locally, including prevalence of infection in the community.
STI screening guidelines
For those with a positive gonorrhea/chlamydia (GC/CT) screen, a nucleic acid amplification test (NAAT) vaginal swab is the preferred specimen source, and self-collection is fine for women of any age, Dr. Policar said. In addition, cis-women who received anal intercourse in the preceding year should consider undergoing a rectal GC/CT NAAT, and those who performed oral sex should consider a pharyngeal GC/CT NAAT, based on shared clinical decision-making. A rectal swab requires an insertion of 3-4 cm and a 360-degree twirl of the wrist, not the swab, to ensure you get a sample from the entire circumference. Pharyngeal samples require swabbing both tonsillar pillars while taking care for those who may gag.
For contact testing – asymptomatic people who have had a high-risk sexual exposure – providers should test for gonorrhea, chlamydia, HIV, and syphilis but not for herpes, high-risk HPV, hepatitis B, hepatitis C, or bacterial vaginosis. “Maybe we’ll do a screen for trichomoniasis, and maybe we’ll offer herpes type 2 serology or antibody screening,” Dr. Policar said. Providers should also ask patients requesting contact testing if they have been vaccinated for hepatitis B. If not, “the conversation should be how can we get you vaccinated for hepatitis B,” Dr. Policar said.
HIV screening only needs to occur once between the ages of 15 and 65 for low-risk people and then once annually (or more often if necessary) for those who have a sex partner with HIV, use injectable drugs, engage in commercial sex work, have a new sex partner with unknown HIV status, received care at an STD or TB clinic, or were in a correctional facility or homeless shelter.
Those at increased risk for syphilis include men who have sex with men, men under age 29, and anyone living with HIV or who has a history of incarceration or a history of commercial sex work. In addition, African Americans have the greatest risk for syphilis of racial/ethnic groups, followed by Hispanics. Most adults only require hepatitis C screening with anti-hep C antibody testing once in their lifetime. Periodic hepatitis C screening should occur for people who inject drugs. If the screening is positive, providers should conduct an RNA polymerase chain reaction (PCR) test to determine whether a chronic infection is present.
Trichomoniasis screening should occur annually in women living with HIV or in correctional facilities. Others to consider screening include people with new or multiple sex partners, a history of STIs, inconsistent condom use, a history of sex work, and intravenous drug use. Dr. Policar also noted that several new assays, including NAAT, PCR, and a rapid test, are available for trichomoniasis.
STI treatment guidelines
For women with mucoprurulent cervicitis, the cause could be chlamydia, gonorrhea, herpes, trichomonas, mycoplasma, or even progesterone from pregnancy or contraception, Dr. Policar said. The new preferred treatment is 100 mg of doxycycline. The alternative, albeit less preferred, treatment is 1 g azithromycin.
The preferred treatment for chlamydia is now 100 mg oral doxycycline twice daily, or doxycycline 200 mg delayed-release once daily, for 7 days. Alternative regimens include 1 g oral azithromycin in a single dose or 500 mg oral levofloxacin once daily for 7 days. The switch to recommending doxycycline over azithromycin is based on recent evidence showing that doxycycline has a slightly higher efficacy for urogenital chlamydia and a substantially higher efficacy for rectal chlamydia. In addition, an increasing proportion of gonorrheal infections have shown resistance to azithromycin, particularly beginning in 2014.
Preferred treatment of new, uncomplicated gonorrhea infections of the cervix, urethra, rectum, and pharynx is one 500-mg dose of ceftriaxone for those weighing under 150 kg and 1 g for those weighing 150 kg or more. If ceftriaxone is unavailable, the new alternative recommended treatment for gonorrhea is 800 mg cefixime. For pharyngeal gonorrhea only, the CDC recommends a test-of-cure 7-14 days after treatment.
For gonorrheal infections, the CDC also recommends treatment with doxycycline if chlamydia has not been excluded, but the agency no longer recommends dual therapy with azithromycin unless it’s used in place of doxycycline for those who are pregnant, have an allergy, or may not be compliant with a 7-day doxycycline regimen.
The preferred treatment for bacterial vaginosis has not changed. The new recommended regimen for trichomoniasis is 500 mg oral metronidazole for 7 days, with the alternative being a single 2-g dose of tinidazole. Male partners should receive 2 g oral metronidazole. The CDC also notes that patients taking metronidazole no longer need to abstain from alcohol during treatment.
”Another area where the guidelines changed is in their description of expedited partner therapy, which means that, when we find an index case who has gonorrhea or chlamydia, we always have a discussion with her about getting her partners treated,” Dr. Policar said. “The CDC was quite clear that the responsibility for discussing partner treatment rests with us as the diagnosing provider” since city and county health departments don’t have the time or resources for contact tracing these STIs.
The two main ways to treat partners are to have the patient bring their partner(s) to the appointment with them or to do patient-delivered partner therapy. Ideally, clinicians who dispense their own medications can give the patient enough drugs to give her partner(s) a complete dose as well. Otherwise, providers can prescribe extra doses in the index patients’ name or write prescriptions in the partner’s name.
“In every state of the union now, it is legal for you to to prescribe antibiotics for partners sight unseen, Dr. Policar said.
Margaret Sullivan, MD, an ob.gyn. from rural western North Carolina, noted during the Q&A that an obstacle to partner therapy at her practice has been cost, particularly since many of the men don’t have insurance.
“I have not heard before of prescribing the extra doses for partners under the patient’s name,” Dr. Sullivan said. “I’ve thought about doing it, but [was worried about] it potentially being fraudulent if that patient has Medicaid and we’re prescribing extra doses under her name, so how do you work around that?”
Dr. Policar acknowledged that barrier and recommended that patients use the website/app Goodrx.com to find discounts for out-of-pocket generic medications. He also noted the occasional obstacle of pharmacists balking at filling a double or triple dose.
“What we’ve been suggesting in that circumstance is to literally copy that part of the CDC guidelines, which explains expedited partner therapy or patient-delivered partner therapy and send that off to the pharmacist so they can see that it’s a national recommendation of the CDC,” Dr. Policar said.
Claudia Rodriguez, MD, an ob.gyn. who works at Sherman Hospital in Elgin, Ill., asked about the CDC recommendations for HPV vaccination in older women. Although the CDC permits women over age 26 to receive the HPV vaccine, the agency does not “make a solid recommendation to have this done, which oftentimes makes a big difference in whether or not health insurance will actually pay for vaccination in that circumstance,” Dr. Policar said.
Patients are welcome to request the vaccine after shared decision-making, but “we should never present this as something which is routine,” he said. For women in their 50s, for example, “there’s virtually no data about any additional degree of protection that you would get” from HPV vaccination, Dr. Policar said in response to a similar question from Tara Allmen, MD, an ob.gyn. in New York City. “If you ask me for my personal clinical opinion about it, I would say it’s not going to be worth it,” he said.
Dr Policar had no disclosures. Disclosures were unavailable for attendees who spoke.
Sexually transmitted infection rates have not increased as dramatically in older women as they have in women in their teens and 20s, but rates of chlamydia and gonorrhea in women over age 35 have seen a steady incline over the past decade, and syphilis rates have climbed steeply, according to data from the Centers for Disease Control and Prevention.
That makes the STI treatment guidelines released by the CDC in July even timelier for practitioners of menopause medicine, according to Michael S. Policar, MD, MPH, a professor emeritus of ob.gyn. and reproductive sciences at the University of California, San Francisco.
Dr. Policar discussed what clinicians need to know about STIs in midlife women at the hybrid annual meeting of the North American Menopause Society. Even the nomenclature change in the guidelines from “sexually transmitted diseases” to “sexually transmitted infections” is important “because they want to acknowledge the fact that a lot of the sexually transmitted infections that we’re treating are asymptomatic, are colonizations, and are not yet diseases,” Dr. Policar said. “We’re trying to be much more expansive in thinking about finding these infections before they actually start causing morbidity in the form of a disease.”
Sexual history
The primary guidelines update for taking sexual history is the recommendation to ask patients about their intentions regarding pregnancy. The “5 Ps” of sexual history are now Partners, Practices, Protection from STIs, Past history of STIs, and Pregnancy intention.
“There should be a sixth P that has to do with pleasure questions,” Policar added. “We ask all the time for patients that we see in the context of perimenopausal and menopausal services, ‘Are you satisfied with your sexual relationship with your partner?’ Hopefully that will make it into the CDC guidelines as the sixth P at some point, but for now, that’s aspirational.”
In asking about partners, instead of asking patients whether they have sex with men, women, or both, clinicians should ask first if the patient is having sex of any kind – oral, vaginal, or anal – with anyone. From there, providers should ask how many sex partners the patient has had, the gender(s) of the partners, and whether they or their partners have other sex partners, using more gender-inclusive language.
When asking about practices, in addition to asking about the type of sexual contact patients have had, additional questions include whether the patient met their partners online or through apps, whether they or any of their partners use drugs, and whether the patient has exchanged sex for any needs, such as money, housing, or drugs. The additional questions can identify those at higher risk for STIs.
After reviewing the CDC’s list of risk factors for gonorrhea and chlamydia screening, Dr. Policar shared the screening list from the California Department of Public Health, which he finds more helpful:
- History of gonorrhea, chlamydia, or pelvic inflammatory disease (PID) in the past 2 years.
- More than 1 sexual partner in the past year.
- New sexual partner within 90 days.
- Reason to believe that a sex partner has had other partners in the past year.
- Exchanging sex for drugs or money within the past year.
- Other factors identified locally, including prevalence of infection in the community.
STI screening guidelines
For those with a positive gonorrhea/chlamydia (GC/CT) screen, a nucleic acid amplification test (NAAT) vaginal swab is the preferred specimen source, and self-collection is fine for women of any age, Dr. Policar said. In addition, cis-women who received anal intercourse in the preceding year should consider undergoing a rectal GC/CT NAAT, and those who performed oral sex should consider a pharyngeal GC/CT NAAT, based on shared clinical decision-making. A rectal swab requires an insertion of 3-4 cm and a 360-degree twirl of the wrist, not the swab, to ensure you get a sample from the entire circumference. Pharyngeal samples require swabbing both tonsillar pillars while taking care for those who may gag.
For contact testing – asymptomatic people who have had a high-risk sexual exposure – providers should test for gonorrhea, chlamydia, HIV, and syphilis but not for herpes, high-risk HPV, hepatitis B, hepatitis C, or bacterial vaginosis. “Maybe we’ll do a screen for trichomoniasis, and maybe we’ll offer herpes type 2 serology or antibody screening,” Dr. Policar said. Providers should also ask patients requesting contact testing if they have been vaccinated for hepatitis B. If not, “the conversation should be how can we get you vaccinated for hepatitis B,” Dr. Policar said.
HIV screening only needs to occur once between the ages of 15 and 65 for low-risk people and then once annually (or more often if necessary) for those who have a sex partner with HIV, use injectable drugs, engage in commercial sex work, have a new sex partner with unknown HIV status, received care at an STD or TB clinic, or were in a correctional facility or homeless shelter.
Those at increased risk for syphilis include men who have sex with men, men under age 29, and anyone living with HIV or who has a history of incarceration or a history of commercial sex work. In addition, African Americans have the greatest risk for syphilis of racial/ethnic groups, followed by Hispanics. Most adults only require hepatitis C screening with anti-hep C antibody testing once in their lifetime. Periodic hepatitis C screening should occur for people who inject drugs. If the screening is positive, providers should conduct an RNA polymerase chain reaction (PCR) test to determine whether a chronic infection is present.
Trichomoniasis screening should occur annually in women living with HIV or in correctional facilities. Others to consider screening include people with new or multiple sex partners, a history of STIs, inconsistent condom use, a history of sex work, and intravenous drug use. Dr. Policar also noted that several new assays, including NAAT, PCR, and a rapid test, are available for trichomoniasis.
STI treatment guidelines
For women with mucoprurulent cervicitis, the cause could be chlamydia, gonorrhea, herpes, trichomonas, mycoplasma, or even progesterone from pregnancy or contraception, Dr. Policar said. The new preferred treatment is 100 mg of doxycycline. The alternative, albeit less preferred, treatment is 1 g azithromycin.
The preferred treatment for chlamydia is now 100 mg oral doxycycline twice daily, or doxycycline 200 mg delayed-release once daily, for 7 days. Alternative regimens include 1 g oral azithromycin in a single dose or 500 mg oral levofloxacin once daily for 7 days. The switch to recommending doxycycline over azithromycin is based on recent evidence showing that doxycycline has a slightly higher efficacy for urogenital chlamydia and a substantially higher efficacy for rectal chlamydia. In addition, an increasing proportion of gonorrheal infections have shown resistance to azithromycin, particularly beginning in 2014.
Preferred treatment of new, uncomplicated gonorrhea infections of the cervix, urethra, rectum, and pharynx is one 500-mg dose of ceftriaxone for those weighing under 150 kg and 1 g for those weighing 150 kg or more. If ceftriaxone is unavailable, the new alternative recommended treatment for gonorrhea is 800 mg cefixime. For pharyngeal gonorrhea only, the CDC recommends a test-of-cure 7-14 days after treatment.
For gonorrheal infections, the CDC also recommends treatment with doxycycline if chlamydia has not been excluded, but the agency no longer recommends dual therapy with azithromycin unless it’s used in place of doxycycline for those who are pregnant, have an allergy, or may not be compliant with a 7-day doxycycline regimen.
The preferred treatment for bacterial vaginosis has not changed. The new recommended regimen for trichomoniasis is 500 mg oral metronidazole for 7 days, with the alternative being a single 2-g dose of tinidazole. Male partners should receive 2 g oral metronidazole. The CDC also notes that patients taking metronidazole no longer need to abstain from alcohol during treatment.
”Another area where the guidelines changed is in their description of expedited partner therapy, which means that, when we find an index case who has gonorrhea or chlamydia, we always have a discussion with her about getting her partners treated,” Dr. Policar said. “The CDC was quite clear that the responsibility for discussing partner treatment rests with us as the diagnosing provider” since city and county health departments don’t have the time or resources for contact tracing these STIs.
The two main ways to treat partners are to have the patient bring their partner(s) to the appointment with them or to do patient-delivered partner therapy. Ideally, clinicians who dispense their own medications can give the patient enough drugs to give her partner(s) a complete dose as well. Otherwise, providers can prescribe extra doses in the index patients’ name or write prescriptions in the partner’s name.
“In every state of the union now, it is legal for you to to prescribe antibiotics for partners sight unseen, Dr. Policar said.
Margaret Sullivan, MD, an ob.gyn. from rural western North Carolina, noted during the Q&A that an obstacle to partner therapy at her practice has been cost, particularly since many of the men don’t have insurance.
“I have not heard before of prescribing the extra doses for partners under the patient’s name,” Dr. Sullivan said. “I’ve thought about doing it, but [was worried about] it potentially being fraudulent if that patient has Medicaid and we’re prescribing extra doses under her name, so how do you work around that?”
Dr. Policar acknowledged that barrier and recommended that patients use the website/app Goodrx.com to find discounts for out-of-pocket generic medications. He also noted the occasional obstacle of pharmacists balking at filling a double or triple dose.
“What we’ve been suggesting in that circumstance is to literally copy that part of the CDC guidelines, which explains expedited partner therapy or patient-delivered partner therapy and send that off to the pharmacist so they can see that it’s a national recommendation of the CDC,” Dr. Policar said.
Claudia Rodriguez, MD, an ob.gyn. who works at Sherman Hospital in Elgin, Ill., asked about the CDC recommendations for HPV vaccination in older women. Although the CDC permits women over age 26 to receive the HPV vaccine, the agency does not “make a solid recommendation to have this done, which oftentimes makes a big difference in whether or not health insurance will actually pay for vaccination in that circumstance,” Dr. Policar said.
Patients are welcome to request the vaccine after shared decision-making, but “we should never present this as something which is routine,” he said. For women in their 50s, for example, “there’s virtually no data about any additional degree of protection that you would get” from HPV vaccination, Dr. Policar said in response to a similar question from Tara Allmen, MD, an ob.gyn. in New York City. “If you ask me for my personal clinical opinion about it, I would say it’s not going to be worth it,” he said.
Dr Policar had no disclosures. Disclosures were unavailable for attendees who spoke.
Sexually transmitted infection rates have not increased as dramatically in older women as they have in women in their teens and 20s, but rates of chlamydia and gonorrhea in women over age 35 have seen a steady incline over the past decade, and syphilis rates have climbed steeply, according to data from the Centers for Disease Control and Prevention.
That makes the STI treatment guidelines released by the CDC in July even timelier for practitioners of menopause medicine, according to Michael S. Policar, MD, MPH, a professor emeritus of ob.gyn. and reproductive sciences at the University of California, San Francisco.
Dr. Policar discussed what clinicians need to know about STIs in midlife women at the hybrid annual meeting of the North American Menopause Society. Even the nomenclature change in the guidelines from “sexually transmitted diseases” to “sexually transmitted infections” is important “because they want to acknowledge the fact that a lot of the sexually transmitted infections that we’re treating are asymptomatic, are colonizations, and are not yet diseases,” Dr. Policar said. “We’re trying to be much more expansive in thinking about finding these infections before they actually start causing morbidity in the form of a disease.”
Sexual history
The primary guidelines update for taking sexual history is the recommendation to ask patients about their intentions regarding pregnancy. The “5 Ps” of sexual history are now Partners, Practices, Protection from STIs, Past history of STIs, and Pregnancy intention.
“There should be a sixth P that has to do with pleasure questions,” Policar added. “We ask all the time for patients that we see in the context of perimenopausal and menopausal services, ‘Are you satisfied with your sexual relationship with your partner?’ Hopefully that will make it into the CDC guidelines as the sixth P at some point, but for now, that’s aspirational.”
In asking about partners, instead of asking patients whether they have sex with men, women, or both, clinicians should ask first if the patient is having sex of any kind – oral, vaginal, or anal – with anyone. From there, providers should ask how many sex partners the patient has had, the gender(s) of the partners, and whether they or their partners have other sex partners, using more gender-inclusive language.
When asking about practices, in addition to asking about the type of sexual contact patients have had, additional questions include whether the patient met their partners online or through apps, whether they or any of their partners use drugs, and whether the patient has exchanged sex for any needs, such as money, housing, or drugs. The additional questions can identify those at higher risk for STIs.
After reviewing the CDC’s list of risk factors for gonorrhea and chlamydia screening, Dr. Policar shared the screening list from the California Department of Public Health, which he finds more helpful:
- History of gonorrhea, chlamydia, or pelvic inflammatory disease (PID) in the past 2 years.
- More than 1 sexual partner in the past year.
- New sexual partner within 90 days.
- Reason to believe that a sex partner has had other partners in the past year.
- Exchanging sex for drugs or money within the past year.
- Other factors identified locally, including prevalence of infection in the community.
STI screening guidelines
For those with a positive gonorrhea/chlamydia (GC/CT) screen, a nucleic acid amplification test (NAAT) vaginal swab is the preferred specimen source, and self-collection is fine for women of any age, Dr. Policar said. In addition, cis-women who received anal intercourse in the preceding year should consider undergoing a rectal GC/CT NAAT, and those who performed oral sex should consider a pharyngeal GC/CT NAAT, based on shared clinical decision-making. A rectal swab requires an insertion of 3-4 cm and a 360-degree twirl of the wrist, not the swab, to ensure you get a sample from the entire circumference. Pharyngeal samples require swabbing both tonsillar pillars while taking care for those who may gag.
For contact testing – asymptomatic people who have had a high-risk sexual exposure – providers should test for gonorrhea, chlamydia, HIV, and syphilis but not for herpes, high-risk HPV, hepatitis B, hepatitis C, or bacterial vaginosis. “Maybe we’ll do a screen for trichomoniasis, and maybe we’ll offer herpes type 2 serology or antibody screening,” Dr. Policar said. Providers should also ask patients requesting contact testing if they have been vaccinated for hepatitis B. If not, “the conversation should be how can we get you vaccinated for hepatitis B,” Dr. Policar said.
HIV screening only needs to occur once between the ages of 15 and 65 for low-risk people and then once annually (or more often if necessary) for those who have a sex partner with HIV, use injectable drugs, engage in commercial sex work, have a new sex partner with unknown HIV status, received care at an STD or TB clinic, or were in a correctional facility or homeless shelter.
Those at increased risk for syphilis include men who have sex with men, men under age 29, and anyone living with HIV or who has a history of incarceration or a history of commercial sex work. In addition, African Americans have the greatest risk for syphilis of racial/ethnic groups, followed by Hispanics. Most adults only require hepatitis C screening with anti-hep C antibody testing once in their lifetime. Periodic hepatitis C screening should occur for people who inject drugs. If the screening is positive, providers should conduct an RNA polymerase chain reaction (PCR) test to determine whether a chronic infection is present.
Trichomoniasis screening should occur annually in women living with HIV or in correctional facilities. Others to consider screening include people with new or multiple sex partners, a history of STIs, inconsistent condom use, a history of sex work, and intravenous drug use. Dr. Policar also noted that several new assays, including NAAT, PCR, and a rapid test, are available for trichomoniasis.
STI treatment guidelines
For women with mucoprurulent cervicitis, the cause could be chlamydia, gonorrhea, herpes, trichomonas, mycoplasma, or even progesterone from pregnancy or contraception, Dr. Policar said. The new preferred treatment is 100 mg of doxycycline. The alternative, albeit less preferred, treatment is 1 g azithromycin.
The preferred treatment for chlamydia is now 100 mg oral doxycycline twice daily, or doxycycline 200 mg delayed-release once daily, for 7 days. Alternative regimens include 1 g oral azithromycin in a single dose or 500 mg oral levofloxacin once daily for 7 days. The switch to recommending doxycycline over azithromycin is based on recent evidence showing that doxycycline has a slightly higher efficacy for urogenital chlamydia and a substantially higher efficacy for rectal chlamydia. In addition, an increasing proportion of gonorrheal infections have shown resistance to azithromycin, particularly beginning in 2014.
Preferred treatment of new, uncomplicated gonorrhea infections of the cervix, urethra, rectum, and pharynx is one 500-mg dose of ceftriaxone for those weighing under 150 kg and 1 g for those weighing 150 kg or more. If ceftriaxone is unavailable, the new alternative recommended treatment for gonorrhea is 800 mg cefixime. For pharyngeal gonorrhea only, the CDC recommends a test-of-cure 7-14 days after treatment.
For gonorrheal infections, the CDC also recommends treatment with doxycycline if chlamydia has not been excluded, but the agency no longer recommends dual therapy with azithromycin unless it’s used in place of doxycycline for those who are pregnant, have an allergy, or may not be compliant with a 7-day doxycycline regimen.
The preferred treatment for bacterial vaginosis has not changed. The new recommended regimen for trichomoniasis is 500 mg oral metronidazole for 7 days, with the alternative being a single 2-g dose of tinidazole. Male partners should receive 2 g oral metronidazole. The CDC also notes that patients taking metronidazole no longer need to abstain from alcohol during treatment.
”Another area where the guidelines changed is in their description of expedited partner therapy, which means that, when we find an index case who has gonorrhea or chlamydia, we always have a discussion with her about getting her partners treated,” Dr. Policar said. “The CDC was quite clear that the responsibility for discussing partner treatment rests with us as the diagnosing provider” since city and county health departments don’t have the time or resources for contact tracing these STIs.
The two main ways to treat partners are to have the patient bring their partner(s) to the appointment with them or to do patient-delivered partner therapy. Ideally, clinicians who dispense their own medications can give the patient enough drugs to give her partner(s) a complete dose as well. Otherwise, providers can prescribe extra doses in the index patients’ name or write prescriptions in the partner’s name.
“In every state of the union now, it is legal for you to to prescribe antibiotics for partners sight unseen, Dr. Policar said.
Margaret Sullivan, MD, an ob.gyn. from rural western North Carolina, noted during the Q&A that an obstacle to partner therapy at her practice has been cost, particularly since many of the men don’t have insurance.
“I have not heard before of prescribing the extra doses for partners under the patient’s name,” Dr. Sullivan said. “I’ve thought about doing it, but [was worried about] it potentially being fraudulent if that patient has Medicaid and we’re prescribing extra doses under her name, so how do you work around that?”
Dr. Policar acknowledged that barrier and recommended that patients use the website/app Goodrx.com to find discounts for out-of-pocket generic medications. He also noted the occasional obstacle of pharmacists balking at filling a double or triple dose.
“What we’ve been suggesting in that circumstance is to literally copy that part of the CDC guidelines, which explains expedited partner therapy or patient-delivered partner therapy and send that off to the pharmacist so they can see that it’s a national recommendation of the CDC,” Dr. Policar said.
Claudia Rodriguez, MD, an ob.gyn. who works at Sherman Hospital in Elgin, Ill., asked about the CDC recommendations for HPV vaccination in older women. Although the CDC permits women over age 26 to receive the HPV vaccine, the agency does not “make a solid recommendation to have this done, which oftentimes makes a big difference in whether or not health insurance will actually pay for vaccination in that circumstance,” Dr. Policar said.
Patients are welcome to request the vaccine after shared decision-making, but “we should never present this as something which is routine,” he said. For women in their 50s, for example, “there’s virtually no data about any additional degree of protection that you would get” from HPV vaccination, Dr. Policar said in response to a similar question from Tara Allmen, MD, an ob.gyn. in New York City. “If you ask me for my personal clinical opinion about it, I would say it’s not going to be worth it,” he said.
Dr Policar had no disclosures. Disclosures were unavailable for attendees who spoke.
FROM NAMS 2021
An 80-year-old female developed a painful purulent nodule a day after gardening
. There are more than 100 species of dematiaceous fungi that can cause phaeohyphomycosis, including Alternaria, Exophiala, Phialophora, Wangiella, Bipolaris, Curvularia, and Exserohilum.1,2 The causative fungi are found in plants and soil, so they are commonly seen after activities such as gardening or walking barefoot. Trauma, such as a splinter, typically incites the infection. Infections can present with superficial, cutaneous and subcutaneous involvement.
Sporotrichosis, also called Rose gardener’s disease, is a mycosis caused by Sporothrix schenckii. A typical presentation is when a gardener gets pricked by a rose thorn. Classically, a pustule will develop at the site of inoculation, with additional lesions forming along the path of lymphatic drainage (called a “sporotrichoid” pattern) weeks later. Atypical mycobacterial infections, mainly Mycobacterium marinum, may also present in this way. Histopathology and tissue cultures help to differentiate the two.
An incision and drainage with pathology was performed in the office. Upon opening the nodule, a large wood splinter was extracted. Both the foreign body and a punch biopsy of skin were sent in for examination. Pathology revealed polarizable foreign material in association with suppurative inflammation and dematiaceous fungi. PAS (Periodic-acid Schiff) and GMS (Grocott methenamine silver) stain highlighted fungal forms. Cultures were negative.
Local disease may be treated with excision alone. Oral antifungals, such as itraconazole, fluconazole, or ketoconazole may be used, although may require long treatment courses for months. Amphotericin B and flucytosine may be required in systemic cases. Almost all cases of disseminated disease occur in immunocompromised patients. Our patient’s hand resolved after removal of the causative thorn.
This case and these photos were submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Kradin R. Diagnostic Pathology of Infectious Disease, 1st edition (Saunders, Feb. 2, 2010).
2. Bolognia J et al. Dermatology (St. Louis: Mosby/Elsevier, 2008).
. There are more than 100 species of dematiaceous fungi that can cause phaeohyphomycosis, including Alternaria, Exophiala, Phialophora, Wangiella, Bipolaris, Curvularia, and Exserohilum.1,2 The causative fungi are found in plants and soil, so they are commonly seen after activities such as gardening or walking barefoot. Trauma, such as a splinter, typically incites the infection. Infections can present with superficial, cutaneous and subcutaneous involvement.
Sporotrichosis, also called Rose gardener’s disease, is a mycosis caused by Sporothrix schenckii. A typical presentation is when a gardener gets pricked by a rose thorn. Classically, a pustule will develop at the site of inoculation, with additional lesions forming along the path of lymphatic drainage (called a “sporotrichoid” pattern) weeks later. Atypical mycobacterial infections, mainly Mycobacterium marinum, may also present in this way. Histopathology and tissue cultures help to differentiate the two.
An incision and drainage with pathology was performed in the office. Upon opening the nodule, a large wood splinter was extracted. Both the foreign body and a punch biopsy of skin were sent in for examination. Pathology revealed polarizable foreign material in association with suppurative inflammation and dematiaceous fungi. PAS (Periodic-acid Schiff) and GMS (Grocott methenamine silver) stain highlighted fungal forms. Cultures were negative.
Local disease may be treated with excision alone. Oral antifungals, such as itraconazole, fluconazole, or ketoconazole may be used, although may require long treatment courses for months. Amphotericin B and flucytosine may be required in systemic cases. Almost all cases of disseminated disease occur in immunocompromised patients. Our patient’s hand resolved after removal of the causative thorn.
This case and these photos were submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Kradin R. Diagnostic Pathology of Infectious Disease, 1st edition (Saunders, Feb. 2, 2010).
2. Bolognia J et al. Dermatology (St. Louis: Mosby/Elsevier, 2008).
. There are more than 100 species of dematiaceous fungi that can cause phaeohyphomycosis, including Alternaria, Exophiala, Phialophora, Wangiella, Bipolaris, Curvularia, and Exserohilum.1,2 The causative fungi are found in plants and soil, so they are commonly seen after activities such as gardening or walking barefoot. Trauma, such as a splinter, typically incites the infection. Infections can present with superficial, cutaneous and subcutaneous involvement.
Sporotrichosis, also called Rose gardener’s disease, is a mycosis caused by Sporothrix schenckii. A typical presentation is when a gardener gets pricked by a rose thorn. Classically, a pustule will develop at the site of inoculation, with additional lesions forming along the path of lymphatic drainage (called a “sporotrichoid” pattern) weeks later. Atypical mycobacterial infections, mainly Mycobacterium marinum, may also present in this way. Histopathology and tissue cultures help to differentiate the two.
An incision and drainage with pathology was performed in the office. Upon opening the nodule, a large wood splinter was extracted. Both the foreign body and a punch biopsy of skin were sent in for examination. Pathology revealed polarizable foreign material in association with suppurative inflammation and dematiaceous fungi. PAS (Periodic-acid Schiff) and GMS (Grocott methenamine silver) stain highlighted fungal forms. Cultures were negative.
Local disease may be treated with excision alone. Oral antifungals, such as itraconazole, fluconazole, or ketoconazole may be used, although may require long treatment courses for months. Amphotericin B and flucytosine may be required in systemic cases. Almost all cases of disseminated disease occur in immunocompromised patients. Our patient’s hand resolved after removal of the causative thorn.
This case and these photos were submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Kradin R. Diagnostic Pathology of Infectious Disease, 1st edition (Saunders, Feb. 2, 2010).
2. Bolognia J et al. Dermatology (St. Louis: Mosby/Elsevier, 2008).
Virtual Visitation: Exploring the Impact on Patients and Families During COVID-19 and Beyond
From Northwell Health, Lake Success, NY.
Objective: Northwell Health, New York’s largest health care organization, rapidly adopted technology solutions to support patient and family communication during the COVID-19 pandemic.
Methods: This case series outlines the pragmatic, interdisciplinary approach Northwell underwent to rapidly implement patient virtual visitation processes during the peak of the initial crisis.
Results: Implementation of large-scale virtual visitation required leadership, technology, and dedicated, empathetic frontline professionals. Patient and family feedback uncovered varied feelings and perspectives, from confusion to gratitude.
Conclusion: Subsequent efforts to obtain direct patient and family perspectives and insights helped Northwell identify areas of strength and ongoing performance improvement.
Keywords: virtual visitation; COVID-19; technology; communication; patient experience.
The power of human connection has become increasingly apparent throughout the COVID-19 pandemic and subsequent recovery phases. Due to the need for social distancing, people worldwide have turned to virtual means of communication, staying in touch with family, friends, and colleagues via digital technology platforms. On March 18, 2020, the New York State Department of Health (NYSDOH) issued a health advisory, suspending all hospital visitation.1 As a result, hospitals rapidly transformed existing in-person visitation practices to meet large-scale virtual programming needs.
Family members often take on various roles—such as advocate, emotional support person, and postdischarge caregiver—for an ill or injured loved one.2 The Institute for Patient- and Family-Centered Care, a nonprofit organization founded in 1992, has been leading a cultural transformation where families are valued as care partners, as opposed to “visitors.”3 Although widely adopted and well-received in specialized units, such as neonatal intensive care units,4 virtual visitation had not been widely implemented across adult care settings. The NYSDOH guidance therefore required organizational leadership, innovation, flexibility, and systems ingenuity to meet the evolving needs of patients, families, and health care professionals. An overarching goal was ensuring patients and families were afforded opportunities to stay connected throughout hospitalization.
Reflecting the impact of COVID-19 surges, hospital environments became increasingly depersonalized, with health care providers wearing extensive personal protective equipment (PPE) and taking remarkable measures to socially distance and minimize exposure. Patients’ room doors were kept primarily closed, while codes and alerts blared in the halls overhead. The lack of families and visitors became increasingly obvious, aiding feelings of isolation and confinement. With fear of nosocomial transmission, impactful modalities (such as sitting at the bedside) and empathetic, therapeutic touch were no longer taking place.
With those scenarios—common to so many health care systems during the pandemic—as a backdrop, comes our experience. Northwell Health is the largest health care system in New York State, geographically spread throughout New York City’s 5 boroughs, Westchester County, and Long Island. With 23 hospitals, approximately 820 medical practices, and over 72 000 employees, Northwell has cared for more than 100 000 COVID-positive patients to date. This case series outlines a pragmatic approach to implementing virtual visitation during the initial peak and obtaining patient and family perspectives to help inform performance improvement and future programming.
Methods
Implementing virtual visitation
Through swift and focused multidisciplinary collaboration, numerous Northwell teams came together to implement large-scale virtual visitation across the organization during the first wave of the COVID crisis. The initial priority involved securing devices that could support patient-family communication. Prior to COVID, each facility had only a handful of tablets that were used primarily during leadership rounding, so once visitation was restricted, we needed a large quantity of devices within a matter of days. Through diligent work from System Procurement and internal Foundation, Northwell was able to acquire nearly 900 devices, including iPads, PadInMotion tablets, and Samsung tablets.
Typically, the benefits of using wireless tablets within a health care setting include long battery life, powerful data processing, advanced operating systems, large screens, and easy end-user navigation.4 During COVID-19 and its associated isolation precautions, tablets offered a lifeline for effective and socially distant communication. With new devices in hand, the system Office of the Chief Information Officer (OCIO) and site-based Information Technology (IT) teams were engaged. They worked tirelessly to streamline connectivity, download necessary apps, test devices on approved WiFi networks, and troubleshoot issues. Once set up, devices were strategically deployed across all Northwell hospitals and post-acute rehabilitation facilities.
Frontline teams quickly realized that a model similar to mobile proning teams, who focus solely on turning and positioning COVID patients to promote optimal respiratory ventilation,5 was needed to support virtual visitation. During the initial COVID wave, elective surgeries were not permissible, as per the NYSDOH. As a result, large numbers of clinical and nonclinical ambulatory surgery employees were redeployed throughout the organization, with many assigned and dedicated to facilitating newly created virtual visitation processes. These employees were primarily responsible for creating unit-based schedules, coordinating logistics, navigating devices on behalf of patients, being present during video calls, and sanitizing the devices between uses. Finally, if necessary, virtual interpretation services were used to overcome language barriers between staff and patients.
What began as an ad hoc function quickly became a valued and meaningful role. Utilizing triage mentality, virtual visitation was first offered during unit-based rounding protocols to those patients with the highest acuity and need to connect with family. We had no formal script; instead, unit-based leaders and frontline team members had open dialogues with patients and families to gauge their interest in virtual visitation. That included patients with an active end-of-life care plan, critically ill patients within intensive care units, and those soon to be intubated or recently extubated. Utilization also occurred within specialty areas such as labor and delivery, pediatrics, inpatient psychiatry, medical units, and long-term rehab facilities. Frontline teams appreciated the supplementary support so they could prioritize ongoing physical assessments and medical interventions. Donned in PPE, virtual visitation team members often served as physical extensions of the patient’s loved ones—holding their hand, offering prayers, and, at times, bearing witness to a last breath. In reflecting on that time, this role required absolute professionalism, empathy, and compassion.
In summer 2020, although demand for virtual visitation was still at an all-time high when ambulatory surgery was reinstated, redeployed staff returned to their responsibilities. To fill this void without interruption to patients and their families, site leaders quickly pivoted and refined processes and protocols utilizing Patient & Customer Experience and Hospitality department team members. Throughout spring 2021, the NYSDOH offered guidance to open in-person visitation, and the institution’s Clinical Advisory Group has been taking a pragmatic approach to doing that in a measured and safe manner across care settings.
Listening to the ‘voice’ of patients and families
Our institution’s mission is grounded in providing “quality service and patient-centered care.” Honoring those tenets, during the initial COVID wave, the system “Voice of the Customer End User Device Workgroup” was created with system and site-based interdisciplinary representation. Despite challenging and unprecedented times, conscious attention and effort was undertaken to assess the use and impact of virtual devices. One of the major work streams was to capture and examine patient and family thoughts, feedback, and the overall experience as it relates to virtual visitation.
The system Office of Patient & Customer Experience (OPCE), led by Sven Gierlinger, SVP Chief Experience Officer, reached out to our colleagues at Press Ganey to add a custom question to patient experience surveys. Beginning on December 1, 2020, discharged inpatients were asked to rate the “Degree to which you were able to stay connected with your family/caregiver during your stay.” Potential answers include the Likert scale responses of Always, Usually, Sometimes, and Never, with “Always” representing the Top Box score. The OPCE team believes these quantitative insights are important to track and trend, particularly since in-person and virtual visitation remain in constant flux.
In an effort to obtain additional, focused, qualitative feedback, OPCE partnered with our institution’s Digital Patient Experience (dPX) colleagues. The approach consisted of voluntary, semistructured, interview-type conversations with patients and family members who engaged in virtual visitation multiple times while the patient was hospitalized. OPCE contacted site-based Patient Experience leads, also known as Culture Leaders, at 3 hospitals, asking them to identify potential participants. This convenience sample excluded instances where the patient passed away during and/or immediately following hospitalization.
The OPCE team phoned potential interview candidates to make a personalized connection, explain the purpose of the interviews, and schedule them, if interested. For consistency, the same Digital Customer Experience Researcher on the dPX team facilitated all sessions, which were 30-minute, semiscripted interviews conducted virtually via Microsoft Teams. The tone was intentionally conversational so that patients and family members would feel comfortable delving into themes that were most impactful during their experience. After some initial ice breakers, such as “What were some of your feelings about being a patient/having a loved one in the hospital during the early days of the COVID-19 pandemic?” we moved on to some more pragmatic, implementation questions and rating scales. These included questions such as “How did you first learn about the option for virtual visitation? Was it something you inquired about or did someone offer it to you? How was it explained to you?” Patients were also asked, on a scale of 1 (easy) to 5 (difficult), to rate their experience with the technology aspect when connecting with their loved ones. They also provided verbal consent to be recorded and were given a $15 gift card upon completion of the interview.
Transcriptions were generated by uploading the interview recordings to a platform called UserTesting. In addition to these transcriptions, this platform also allowed for a keyword mapping tool that organized high-level themes and adjectives into groupings along a sentiment axis from negative to neutral to positive. Transcripts were then read carefully and annotated by the Digital Customer Experience Researcher, which allowed for strengthening of some of the automated themes as well as the emergence of new, more nuanced themes. These themes were organized into those that we could address with design and/or procedure updates (actionable insights), those that came up most frequently overall (frequency), and those that came up across our 3 interview sessions (commonality).
This feedback, along with the responses to the new Press Ganey question, was presented to the system Voice of the Customer End User Device Workgroup. The results led to robust discussion and brainstorming regarding how to improve the process to be more patient-centered. Findings were also shared with our hospital-based Culture Leaders. As many of their local strategic plans focused on patient-family communication, this information was helpful to them in considering plans for expansion and/or sustaining virtual visitation efforts. The process map in the Figure outlines key milestones within this feedback loop.
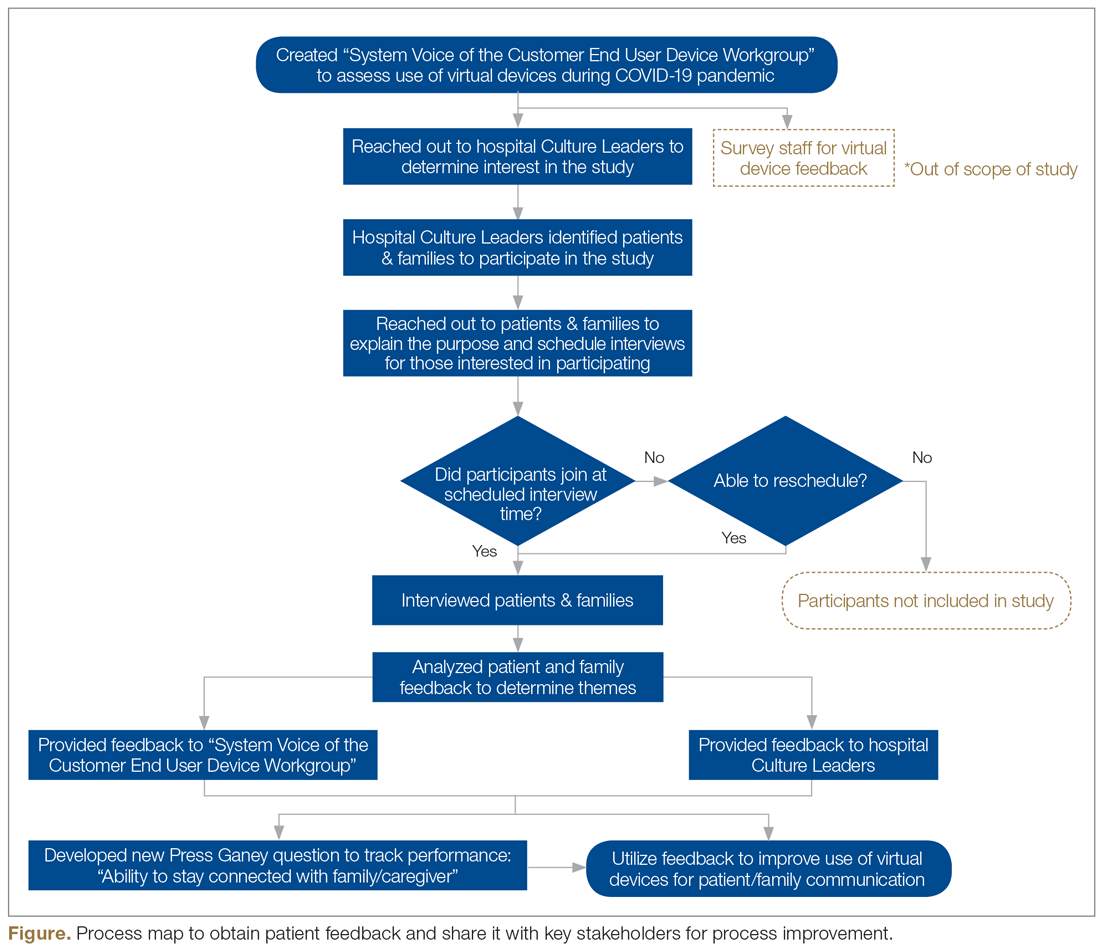
Outcomes
During the height of the initial COVID-19 crisis, virtual visitation was a new and ever-evolving process. Amidst the chaos, mechanisms to capture the quantity and quality of virtual visits were not in place. Based on informal observation, a majority of patients utilized personal devices to connect with loved ones, and staff even offered their own cellular devices to facilitate timely patient-family communication. The technology primarily used included FaceTime, Zoom, and EZCall, as there was much public awareness and comfort with those platforms.
In the first quarter of 2021, our institution overall performed at a Top Box score of 60.2 for our ability to assist patients with staying connected to their family/caregiver during their inpatient visit. With more than 6700 returned surveys during that time period, our hospitals earned Top Box scores ranging between 48.0 and 75.3. At this time, obtaining a national benchmark ranking is not possible, because the question regarding connectedness is unique to Northwell inpatient settings. As other health care organizations adopt this customized question, further peer-to-peer measurements can be established.
Regarding virtual interviews, 25 patients were initially contacted to determine their interest in participating. Of that sample, 17 patients were engaged over the phone, representing a reach rate of 68%. Overall, 10 interviews were scheduled; 7 patients did not show up, resulting in 3 completed interviews. During follow-up, “no-show” participants either gave no response or stated they had a conflict at their originally scheduled time but were not interested in rescheduling due to personal circumstances. Through such conversations, ongoing health complications were found to be a reoccurring barrier to participation.
Each of the participating patients had experienced being placed on a ventilator. They described their hospitalization as a time of “confusion and despair” in the first days after extubation. After we reviewed interview recordings, a reoccurring theme across all interviews was the feeling of gratitude. Patients expressed deep and heartfelt appreciation for being given the opportunity to connect as a family. One patient described virtual visitation sessions as her “only tether to reality when nothing else made sense.”
Interestingly enough, none of the participants knew that virtual visitation was an option and/or thought to inquire about it before a hospital staff member offered to set up a session. Patients recounted how they were weak and physically unable to connect to the sessions without significant assistance. They reported examples of not having the physical strength to hold up the tablet or needing a staff member to facilitate the conversation because the patient could not speak loudly enough and/or they were having difficulty hearing over background medical equipment noises. Participants also described times when a nurse or social worker would stand and hold the tablet for 20 to 30 minutes at a time, further describing mixed feelings of gratitude, guilt for “taking up their time,” and a desire for more privacy to have those precious conversations.
Discussion
Our institution encountered various barriers when establishing, implementing, and sustaining virtual visitation. The acquisition and bulk purchasing of devices, so that each hospital unit and department had adequate par levels during a high-demand time frame, was an initial challenge. Ensuring appropriate safeguards, software programming, and access to WiFi required ingenuity from IT teams. Leaders sought to advocate for the importance of prioritizing virtual visitation alongside clinical interventions. For team members, education was needed to build awareness, learn how to navigate technology, and troubleshoot, in real-time, issues such as poor connectivity. However, despite these organizational struggles, the hospital’s frontline professionals fully recognized and understood the humanistic value of connecting ill patients with their loved ones. Harnessing their teamwork, empathy, and innovative spirits, they forged through such difficulties to create meaningful interactions.
Although virtual visitation occurred prior to the COVID-19 pandemic, particularly in subspecialty areas such as neonatal intensive care units,6 it was not commonplace in most adult inpatient care settings. However, now that virtual means to communication are widely accepted and preferred, our hospital anticipates these offerings will become a broad patient expectation and, therefore, part of standard hospital care and operations. Health care leaders and interdisciplinary teams must therefore prioritize virtual visitation protocols, efforts, and future programming. It is no longer an exception to the rule, but rather a critical approach when ensuring quality communication between patients, families, and care teams.
We strive to continually improve by including user feedback as part of an interactive design process. For a broader, more permanent installation of virtual visitation, health care organizations must proactively promote this capability as a valued option. Considering health literacy and comfort with technology, functionality, and logistics must be carefully explained to patients and their families. This may require additional staff training so that they are knowledgeable, comfortable with, and able to troubleshoot questions/concerns in real time. There needs to be an adequate number of mobile devices available at a unit or departmental level to meet short-term and long-term demands. Additionally, now that we have emerged from our initial crisis-based mentality, it is time to consider alternatives to alleviate the need for staff assistance, such as mounts to hold devices and enabling voice controls.
Conclusion
As an organization grounded in the spirit of innovation, Northwell has been able to quickly pivot, adopting virtual visitation to address emerging and complex communication needs. Taking a best practice established during a crisis period and engraining it into sustainable organizational culture and operations requires visionary leadership, strong teamwork, and an unbridled commitment to patient and family centeredness. Despite unprecedented challenges, our commitment to listening to the “voice” of patients and families never wavered. Using their insights and feedback as critical components to the decision-making process, there is much work ahead within the realm of virtual visitation.
Acknowledgements: The authors would like to acknowledge the Northwell Health providers, frontline health care professionals, and team members who worked tirelessly to care for its community during initial COVID-19 waves and every day thereafter. Heartfelt gratitude to Northwell’s senior leaders for the visionary leadership; the OCIO and hospital-based IT teams for their swift collaboration; and dedicated Culture Leaders, Patient Experience team members, and redeployed staff for their unbridled passion for caring for patients and families. Special thanks to Agnes Barden, DNP, RN, CPXP, Joseph Narvaez, MBA, and Natalie Bashkin, MBA, from the system Office of Patient & Customer Experience, and Carolyne Burgess, MPH, from the Digital Patient Experience teams, for their participation, leadership, and syngeristic partnerships.
Corresponding Author: Nicole Giammarinaro, MSN, RN, CPXP, Director, Patient & Customer Experience, Northwell Health, 2000 Marcus Ave, Lake Success, NY 11042; [email protected].
Financial disclosures: Sven Gierlinger serves on the Speakers Bureau for Northwell Health and as an Executive Board Member for The Beryl Institute.
1. New York State Department of Health. Health advisory: COVID-19 guidance for hospital operators regarding visitation. March 18, 2020. https://coronavirus.health.ny.gov/system/files/documents/2020/03/covid19-hospital-visitation-guidance-3.18.20.pdf
2. Zhang Y. Family functioning in the context of an adult family member with illness: a concept analysis. J Clin Nurs. 2018;27(15-16):3205-3224. doi:10.1111/jocn.14500
3. Institute for Patient- & Family-Centered Care. Better Together: Partnering with Families. https://www.ipfcc.org/bestpractices/better-together-ny.html
4. Marceglia S, Bonacina S, Zaccaria V, et al. How might the iPad change healthcare? J R Soc Med. 2012;105(6):233-241. doi:10.1258/jrsm.2012.110296
5. Short B, Parekh M, Ryan P, et al. Rapid implementation of a mobile prone team during the COVID-19 pandemic. J Crit Care. 2020;60:230-234. doi:10.1016/j.jcrc.2020.08.020
6. Yeo C, Ho SK, Khong K, Lau Y. Virtual visitation in the neonatal intensive care: experience with the use of internet and telemedicine in a tertiary neonatal unit. Perm J. 2011;15(3):32-36.
From Northwell Health, Lake Success, NY.
Objective: Northwell Health, New York’s largest health care organization, rapidly adopted technology solutions to support patient and family communication during the COVID-19 pandemic.
Methods: This case series outlines the pragmatic, interdisciplinary approach Northwell underwent to rapidly implement patient virtual visitation processes during the peak of the initial crisis.
Results: Implementation of large-scale virtual visitation required leadership, technology, and dedicated, empathetic frontline professionals. Patient and family feedback uncovered varied feelings and perspectives, from confusion to gratitude.
Conclusion: Subsequent efforts to obtain direct patient and family perspectives and insights helped Northwell identify areas of strength and ongoing performance improvement.
Keywords: virtual visitation; COVID-19; technology; communication; patient experience.
The power of human connection has become increasingly apparent throughout the COVID-19 pandemic and subsequent recovery phases. Due to the need for social distancing, people worldwide have turned to virtual means of communication, staying in touch with family, friends, and colleagues via digital technology platforms. On March 18, 2020, the New York State Department of Health (NYSDOH) issued a health advisory, suspending all hospital visitation.1 As a result, hospitals rapidly transformed existing in-person visitation practices to meet large-scale virtual programming needs.
Family members often take on various roles—such as advocate, emotional support person, and postdischarge caregiver—for an ill or injured loved one.2 The Institute for Patient- and Family-Centered Care, a nonprofit organization founded in 1992, has been leading a cultural transformation where families are valued as care partners, as opposed to “visitors.”3 Although widely adopted and well-received in specialized units, such as neonatal intensive care units,4 virtual visitation had not been widely implemented across adult care settings. The NYSDOH guidance therefore required organizational leadership, innovation, flexibility, and systems ingenuity to meet the evolving needs of patients, families, and health care professionals. An overarching goal was ensuring patients and families were afforded opportunities to stay connected throughout hospitalization.
Reflecting the impact of COVID-19 surges, hospital environments became increasingly depersonalized, with health care providers wearing extensive personal protective equipment (PPE) and taking remarkable measures to socially distance and minimize exposure. Patients’ room doors were kept primarily closed, while codes and alerts blared in the halls overhead. The lack of families and visitors became increasingly obvious, aiding feelings of isolation and confinement. With fear of nosocomial transmission, impactful modalities (such as sitting at the bedside) and empathetic, therapeutic touch were no longer taking place.
With those scenarios—common to so many health care systems during the pandemic—as a backdrop, comes our experience. Northwell Health is the largest health care system in New York State, geographically spread throughout New York City’s 5 boroughs, Westchester County, and Long Island. With 23 hospitals, approximately 820 medical practices, and over 72 000 employees, Northwell has cared for more than 100 000 COVID-positive patients to date. This case series outlines a pragmatic approach to implementing virtual visitation during the initial peak and obtaining patient and family perspectives to help inform performance improvement and future programming.
Methods
Implementing virtual visitation
Through swift and focused multidisciplinary collaboration, numerous Northwell teams came together to implement large-scale virtual visitation across the organization during the first wave of the COVID crisis. The initial priority involved securing devices that could support patient-family communication. Prior to COVID, each facility had only a handful of tablets that were used primarily during leadership rounding, so once visitation was restricted, we needed a large quantity of devices within a matter of days. Through diligent work from System Procurement and internal Foundation, Northwell was able to acquire nearly 900 devices, including iPads, PadInMotion tablets, and Samsung tablets.
Typically, the benefits of using wireless tablets within a health care setting include long battery life, powerful data processing, advanced operating systems, large screens, and easy end-user navigation.4 During COVID-19 and its associated isolation precautions, tablets offered a lifeline for effective and socially distant communication. With new devices in hand, the system Office of the Chief Information Officer (OCIO) and site-based Information Technology (IT) teams were engaged. They worked tirelessly to streamline connectivity, download necessary apps, test devices on approved WiFi networks, and troubleshoot issues. Once set up, devices were strategically deployed across all Northwell hospitals and post-acute rehabilitation facilities.
Frontline teams quickly realized that a model similar to mobile proning teams, who focus solely on turning and positioning COVID patients to promote optimal respiratory ventilation,5 was needed to support virtual visitation. During the initial COVID wave, elective surgeries were not permissible, as per the NYSDOH. As a result, large numbers of clinical and nonclinical ambulatory surgery employees were redeployed throughout the organization, with many assigned and dedicated to facilitating newly created virtual visitation processes. These employees were primarily responsible for creating unit-based schedules, coordinating logistics, navigating devices on behalf of patients, being present during video calls, and sanitizing the devices between uses. Finally, if necessary, virtual interpretation services were used to overcome language barriers between staff and patients.
What began as an ad hoc function quickly became a valued and meaningful role. Utilizing triage mentality, virtual visitation was first offered during unit-based rounding protocols to those patients with the highest acuity and need to connect with family. We had no formal script; instead, unit-based leaders and frontline team members had open dialogues with patients and families to gauge their interest in virtual visitation. That included patients with an active end-of-life care plan, critically ill patients within intensive care units, and those soon to be intubated or recently extubated. Utilization also occurred within specialty areas such as labor and delivery, pediatrics, inpatient psychiatry, medical units, and long-term rehab facilities. Frontline teams appreciated the supplementary support so they could prioritize ongoing physical assessments and medical interventions. Donned in PPE, virtual visitation team members often served as physical extensions of the patient’s loved ones—holding their hand, offering prayers, and, at times, bearing witness to a last breath. In reflecting on that time, this role required absolute professionalism, empathy, and compassion.
In summer 2020, although demand for virtual visitation was still at an all-time high when ambulatory surgery was reinstated, redeployed staff returned to their responsibilities. To fill this void without interruption to patients and their families, site leaders quickly pivoted and refined processes and protocols utilizing Patient & Customer Experience and Hospitality department team members. Throughout spring 2021, the NYSDOH offered guidance to open in-person visitation, and the institution’s Clinical Advisory Group has been taking a pragmatic approach to doing that in a measured and safe manner across care settings.
Listening to the ‘voice’ of patients and families
Our institution’s mission is grounded in providing “quality service and patient-centered care.” Honoring those tenets, during the initial COVID wave, the system “Voice of the Customer End User Device Workgroup” was created with system and site-based interdisciplinary representation. Despite challenging and unprecedented times, conscious attention and effort was undertaken to assess the use and impact of virtual devices. One of the major work streams was to capture and examine patient and family thoughts, feedback, and the overall experience as it relates to virtual visitation.
The system Office of Patient & Customer Experience (OPCE), led by Sven Gierlinger, SVP Chief Experience Officer, reached out to our colleagues at Press Ganey to add a custom question to patient experience surveys. Beginning on December 1, 2020, discharged inpatients were asked to rate the “Degree to which you were able to stay connected with your family/caregiver during your stay.” Potential answers include the Likert scale responses of Always, Usually, Sometimes, and Never, with “Always” representing the Top Box score. The OPCE team believes these quantitative insights are important to track and trend, particularly since in-person and virtual visitation remain in constant flux.
In an effort to obtain additional, focused, qualitative feedback, OPCE partnered with our institution’s Digital Patient Experience (dPX) colleagues. The approach consisted of voluntary, semistructured, interview-type conversations with patients and family members who engaged in virtual visitation multiple times while the patient was hospitalized. OPCE contacted site-based Patient Experience leads, also known as Culture Leaders, at 3 hospitals, asking them to identify potential participants. This convenience sample excluded instances where the patient passed away during and/or immediately following hospitalization.
The OPCE team phoned potential interview candidates to make a personalized connection, explain the purpose of the interviews, and schedule them, if interested. For consistency, the same Digital Customer Experience Researcher on the dPX team facilitated all sessions, which were 30-minute, semiscripted interviews conducted virtually via Microsoft Teams. The tone was intentionally conversational so that patients and family members would feel comfortable delving into themes that were most impactful during their experience. After some initial ice breakers, such as “What were some of your feelings about being a patient/having a loved one in the hospital during the early days of the COVID-19 pandemic?” we moved on to some more pragmatic, implementation questions and rating scales. These included questions such as “How did you first learn about the option for virtual visitation? Was it something you inquired about or did someone offer it to you? How was it explained to you?” Patients were also asked, on a scale of 1 (easy) to 5 (difficult), to rate their experience with the technology aspect when connecting with their loved ones. They also provided verbal consent to be recorded and were given a $15 gift card upon completion of the interview.
Transcriptions were generated by uploading the interview recordings to a platform called UserTesting. In addition to these transcriptions, this platform also allowed for a keyword mapping tool that organized high-level themes and adjectives into groupings along a sentiment axis from negative to neutral to positive. Transcripts were then read carefully and annotated by the Digital Customer Experience Researcher, which allowed for strengthening of some of the automated themes as well as the emergence of new, more nuanced themes. These themes were organized into those that we could address with design and/or procedure updates (actionable insights), those that came up most frequently overall (frequency), and those that came up across our 3 interview sessions (commonality).
This feedback, along with the responses to the new Press Ganey question, was presented to the system Voice of the Customer End User Device Workgroup. The results led to robust discussion and brainstorming regarding how to improve the process to be more patient-centered. Findings were also shared with our hospital-based Culture Leaders. As many of their local strategic plans focused on patient-family communication, this information was helpful to them in considering plans for expansion and/or sustaining virtual visitation efforts. The process map in the Figure outlines key milestones within this feedback loop.

Outcomes
During the height of the initial COVID-19 crisis, virtual visitation was a new and ever-evolving process. Amidst the chaos, mechanisms to capture the quantity and quality of virtual visits were not in place. Based on informal observation, a majority of patients utilized personal devices to connect with loved ones, and staff even offered their own cellular devices to facilitate timely patient-family communication. The technology primarily used included FaceTime, Zoom, and EZCall, as there was much public awareness and comfort with those platforms.
In the first quarter of 2021, our institution overall performed at a Top Box score of 60.2 for our ability to assist patients with staying connected to their family/caregiver during their inpatient visit. With more than 6700 returned surveys during that time period, our hospitals earned Top Box scores ranging between 48.0 and 75.3. At this time, obtaining a national benchmark ranking is not possible, because the question regarding connectedness is unique to Northwell inpatient settings. As other health care organizations adopt this customized question, further peer-to-peer measurements can be established.
Regarding virtual interviews, 25 patients were initially contacted to determine their interest in participating. Of that sample, 17 patients were engaged over the phone, representing a reach rate of 68%. Overall, 10 interviews were scheduled; 7 patients did not show up, resulting in 3 completed interviews. During follow-up, “no-show” participants either gave no response or stated they had a conflict at their originally scheduled time but were not interested in rescheduling due to personal circumstances. Through such conversations, ongoing health complications were found to be a reoccurring barrier to participation.
Each of the participating patients had experienced being placed on a ventilator. They described their hospitalization as a time of “confusion and despair” in the first days after extubation. After we reviewed interview recordings, a reoccurring theme across all interviews was the feeling of gratitude. Patients expressed deep and heartfelt appreciation for being given the opportunity to connect as a family. One patient described virtual visitation sessions as her “only tether to reality when nothing else made sense.”
Interestingly enough, none of the participants knew that virtual visitation was an option and/or thought to inquire about it before a hospital staff member offered to set up a session. Patients recounted how they were weak and physically unable to connect to the sessions without significant assistance. They reported examples of not having the physical strength to hold up the tablet or needing a staff member to facilitate the conversation because the patient could not speak loudly enough and/or they were having difficulty hearing over background medical equipment noises. Participants also described times when a nurse or social worker would stand and hold the tablet for 20 to 30 minutes at a time, further describing mixed feelings of gratitude, guilt for “taking up their time,” and a desire for more privacy to have those precious conversations.
Discussion
Our institution encountered various barriers when establishing, implementing, and sustaining virtual visitation. The acquisition and bulk purchasing of devices, so that each hospital unit and department had adequate par levels during a high-demand time frame, was an initial challenge. Ensuring appropriate safeguards, software programming, and access to WiFi required ingenuity from IT teams. Leaders sought to advocate for the importance of prioritizing virtual visitation alongside clinical interventions. For team members, education was needed to build awareness, learn how to navigate technology, and troubleshoot, in real-time, issues such as poor connectivity. However, despite these organizational struggles, the hospital’s frontline professionals fully recognized and understood the humanistic value of connecting ill patients with their loved ones. Harnessing their teamwork, empathy, and innovative spirits, they forged through such difficulties to create meaningful interactions.
Although virtual visitation occurred prior to the COVID-19 pandemic, particularly in subspecialty areas such as neonatal intensive care units,6 it was not commonplace in most adult inpatient care settings. However, now that virtual means to communication are widely accepted and preferred, our hospital anticipates these offerings will become a broad patient expectation and, therefore, part of standard hospital care and operations. Health care leaders and interdisciplinary teams must therefore prioritize virtual visitation protocols, efforts, and future programming. It is no longer an exception to the rule, but rather a critical approach when ensuring quality communication between patients, families, and care teams.
We strive to continually improve by including user feedback as part of an interactive design process. For a broader, more permanent installation of virtual visitation, health care organizations must proactively promote this capability as a valued option. Considering health literacy and comfort with technology, functionality, and logistics must be carefully explained to patients and their families. This may require additional staff training so that they are knowledgeable, comfortable with, and able to troubleshoot questions/concerns in real time. There needs to be an adequate number of mobile devices available at a unit or departmental level to meet short-term and long-term demands. Additionally, now that we have emerged from our initial crisis-based mentality, it is time to consider alternatives to alleviate the need for staff assistance, such as mounts to hold devices and enabling voice controls.
Conclusion
As an organization grounded in the spirit of innovation, Northwell has been able to quickly pivot, adopting virtual visitation to address emerging and complex communication needs. Taking a best practice established during a crisis period and engraining it into sustainable organizational culture and operations requires visionary leadership, strong teamwork, and an unbridled commitment to patient and family centeredness. Despite unprecedented challenges, our commitment to listening to the “voice” of patients and families never wavered. Using their insights and feedback as critical components to the decision-making process, there is much work ahead within the realm of virtual visitation.
Acknowledgements: The authors would like to acknowledge the Northwell Health providers, frontline health care professionals, and team members who worked tirelessly to care for its community during initial COVID-19 waves and every day thereafter. Heartfelt gratitude to Northwell’s senior leaders for the visionary leadership; the OCIO and hospital-based IT teams for their swift collaboration; and dedicated Culture Leaders, Patient Experience team members, and redeployed staff for their unbridled passion for caring for patients and families. Special thanks to Agnes Barden, DNP, RN, CPXP, Joseph Narvaez, MBA, and Natalie Bashkin, MBA, from the system Office of Patient & Customer Experience, and Carolyne Burgess, MPH, from the Digital Patient Experience teams, for their participation, leadership, and syngeristic partnerships.
Corresponding Author: Nicole Giammarinaro, MSN, RN, CPXP, Director, Patient & Customer Experience, Northwell Health, 2000 Marcus Ave, Lake Success, NY 11042; [email protected].
Financial disclosures: Sven Gierlinger serves on the Speakers Bureau for Northwell Health and as an Executive Board Member for The Beryl Institute.
From Northwell Health, Lake Success, NY.
Objective: Northwell Health, New York’s largest health care organization, rapidly adopted technology solutions to support patient and family communication during the COVID-19 pandemic.
Methods: This case series outlines the pragmatic, interdisciplinary approach Northwell underwent to rapidly implement patient virtual visitation processes during the peak of the initial crisis.
Results: Implementation of large-scale virtual visitation required leadership, technology, and dedicated, empathetic frontline professionals. Patient and family feedback uncovered varied feelings and perspectives, from confusion to gratitude.
Conclusion: Subsequent efforts to obtain direct patient and family perspectives and insights helped Northwell identify areas of strength and ongoing performance improvement.
Keywords: virtual visitation; COVID-19; technology; communication; patient experience.
The power of human connection has become increasingly apparent throughout the COVID-19 pandemic and subsequent recovery phases. Due to the need for social distancing, people worldwide have turned to virtual means of communication, staying in touch with family, friends, and colleagues via digital technology platforms. On March 18, 2020, the New York State Department of Health (NYSDOH) issued a health advisory, suspending all hospital visitation.1 As a result, hospitals rapidly transformed existing in-person visitation practices to meet large-scale virtual programming needs.
Family members often take on various roles—such as advocate, emotional support person, and postdischarge caregiver—for an ill or injured loved one.2 The Institute for Patient- and Family-Centered Care, a nonprofit organization founded in 1992, has been leading a cultural transformation where families are valued as care partners, as opposed to “visitors.”3 Although widely adopted and well-received in specialized units, such as neonatal intensive care units,4 virtual visitation had not been widely implemented across adult care settings. The NYSDOH guidance therefore required organizational leadership, innovation, flexibility, and systems ingenuity to meet the evolving needs of patients, families, and health care professionals. An overarching goal was ensuring patients and families were afforded opportunities to stay connected throughout hospitalization.
Reflecting the impact of COVID-19 surges, hospital environments became increasingly depersonalized, with health care providers wearing extensive personal protective equipment (PPE) and taking remarkable measures to socially distance and minimize exposure. Patients’ room doors were kept primarily closed, while codes and alerts blared in the halls overhead. The lack of families and visitors became increasingly obvious, aiding feelings of isolation and confinement. With fear of nosocomial transmission, impactful modalities (such as sitting at the bedside) and empathetic, therapeutic touch were no longer taking place.
With those scenarios—common to so many health care systems during the pandemic—as a backdrop, comes our experience. Northwell Health is the largest health care system in New York State, geographically spread throughout New York City’s 5 boroughs, Westchester County, and Long Island. With 23 hospitals, approximately 820 medical practices, and over 72 000 employees, Northwell has cared for more than 100 000 COVID-positive patients to date. This case series outlines a pragmatic approach to implementing virtual visitation during the initial peak and obtaining patient and family perspectives to help inform performance improvement and future programming.
Methods
Implementing virtual visitation
Through swift and focused multidisciplinary collaboration, numerous Northwell teams came together to implement large-scale virtual visitation across the organization during the first wave of the COVID crisis. The initial priority involved securing devices that could support patient-family communication. Prior to COVID, each facility had only a handful of tablets that were used primarily during leadership rounding, so once visitation was restricted, we needed a large quantity of devices within a matter of days. Through diligent work from System Procurement and internal Foundation, Northwell was able to acquire nearly 900 devices, including iPads, PadInMotion tablets, and Samsung tablets.
Typically, the benefits of using wireless tablets within a health care setting include long battery life, powerful data processing, advanced operating systems, large screens, and easy end-user navigation.4 During COVID-19 and its associated isolation precautions, tablets offered a lifeline for effective and socially distant communication. With new devices in hand, the system Office of the Chief Information Officer (OCIO) and site-based Information Technology (IT) teams were engaged. They worked tirelessly to streamline connectivity, download necessary apps, test devices on approved WiFi networks, and troubleshoot issues. Once set up, devices were strategically deployed across all Northwell hospitals and post-acute rehabilitation facilities.
Frontline teams quickly realized that a model similar to mobile proning teams, who focus solely on turning and positioning COVID patients to promote optimal respiratory ventilation,5 was needed to support virtual visitation. During the initial COVID wave, elective surgeries were not permissible, as per the NYSDOH. As a result, large numbers of clinical and nonclinical ambulatory surgery employees were redeployed throughout the organization, with many assigned and dedicated to facilitating newly created virtual visitation processes. These employees were primarily responsible for creating unit-based schedules, coordinating logistics, navigating devices on behalf of patients, being present during video calls, and sanitizing the devices between uses. Finally, if necessary, virtual interpretation services were used to overcome language barriers between staff and patients.
What began as an ad hoc function quickly became a valued and meaningful role. Utilizing triage mentality, virtual visitation was first offered during unit-based rounding protocols to those patients with the highest acuity and need to connect with family. We had no formal script; instead, unit-based leaders and frontline team members had open dialogues with patients and families to gauge their interest in virtual visitation. That included patients with an active end-of-life care plan, critically ill patients within intensive care units, and those soon to be intubated or recently extubated. Utilization also occurred within specialty areas such as labor and delivery, pediatrics, inpatient psychiatry, medical units, and long-term rehab facilities. Frontline teams appreciated the supplementary support so they could prioritize ongoing physical assessments and medical interventions. Donned in PPE, virtual visitation team members often served as physical extensions of the patient’s loved ones—holding their hand, offering prayers, and, at times, bearing witness to a last breath. In reflecting on that time, this role required absolute professionalism, empathy, and compassion.
In summer 2020, although demand for virtual visitation was still at an all-time high when ambulatory surgery was reinstated, redeployed staff returned to their responsibilities. To fill this void without interruption to patients and their families, site leaders quickly pivoted and refined processes and protocols utilizing Patient & Customer Experience and Hospitality department team members. Throughout spring 2021, the NYSDOH offered guidance to open in-person visitation, and the institution’s Clinical Advisory Group has been taking a pragmatic approach to doing that in a measured and safe manner across care settings.
Listening to the ‘voice’ of patients and families
Our institution’s mission is grounded in providing “quality service and patient-centered care.” Honoring those tenets, during the initial COVID wave, the system “Voice of the Customer End User Device Workgroup” was created with system and site-based interdisciplinary representation. Despite challenging and unprecedented times, conscious attention and effort was undertaken to assess the use and impact of virtual devices. One of the major work streams was to capture and examine patient and family thoughts, feedback, and the overall experience as it relates to virtual visitation.
The system Office of Patient & Customer Experience (OPCE), led by Sven Gierlinger, SVP Chief Experience Officer, reached out to our colleagues at Press Ganey to add a custom question to patient experience surveys. Beginning on December 1, 2020, discharged inpatients were asked to rate the “Degree to which you were able to stay connected with your family/caregiver during your stay.” Potential answers include the Likert scale responses of Always, Usually, Sometimes, and Never, with “Always” representing the Top Box score. The OPCE team believes these quantitative insights are important to track and trend, particularly since in-person and virtual visitation remain in constant flux.
In an effort to obtain additional, focused, qualitative feedback, OPCE partnered with our institution’s Digital Patient Experience (dPX) colleagues. The approach consisted of voluntary, semistructured, interview-type conversations with patients and family members who engaged in virtual visitation multiple times while the patient was hospitalized. OPCE contacted site-based Patient Experience leads, also known as Culture Leaders, at 3 hospitals, asking them to identify potential participants. This convenience sample excluded instances where the patient passed away during and/or immediately following hospitalization.
The OPCE team phoned potential interview candidates to make a personalized connection, explain the purpose of the interviews, and schedule them, if interested. For consistency, the same Digital Customer Experience Researcher on the dPX team facilitated all sessions, which were 30-minute, semiscripted interviews conducted virtually via Microsoft Teams. The tone was intentionally conversational so that patients and family members would feel comfortable delving into themes that were most impactful during their experience. After some initial ice breakers, such as “What were some of your feelings about being a patient/having a loved one in the hospital during the early days of the COVID-19 pandemic?” we moved on to some more pragmatic, implementation questions and rating scales. These included questions such as “How did you first learn about the option for virtual visitation? Was it something you inquired about or did someone offer it to you? How was it explained to you?” Patients were also asked, on a scale of 1 (easy) to 5 (difficult), to rate their experience with the technology aspect when connecting with their loved ones. They also provided verbal consent to be recorded and were given a $15 gift card upon completion of the interview.
Transcriptions were generated by uploading the interview recordings to a platform called UserTesting. In addition to these transcriptions, this platform also allowed for a keyword mapping tool that organized high-level themes and adjectives into groupings along a sentiment axis from negative to neutral to positive. Transcripts were then read carefully and annotated by the Digital Customer Experience Researcher, which allowed for strengthening of some of the automated themes as well as the emergence of new, more nuanced themes. These themes were organized into those that we could address with design and/or procedure updates (actionable insights), those that came up most frequently overall (frequency), and those that came up across our 3 interview sessions (commonality).
This feedback, along with the responses to the new Press Ganey question, was presented to the system Voice of the Customer End User Device Workgroup. The results led to robust discussion and brainstorming regarding how to improve the process to be more patient-centered. Findings were also shared with our hospital-based Culture Leaders. As many of their local strategic plans focused on patient-family communication, this information was helpful to them in considering plans for expansion and/or sustaining virtual visitation efforts. The process map in the Figure outlines key milestones within this feedback loop.

Outcomes
During the height of the initial COVID-19 crisis, virtual visitation was a new and ever-evolving process. Amidst the chaos, mechanisms to capture the quantity and quality of virtual visits were not in place. Based on informal observation, a majority of patients utilized personal devices to connect with loved ones, and staff even offered their own cellular devices to facilitate timely patient-family communication. The technology primarily used included FaceTime, Zoom, and EZCall, as there was much public awareness and comfort with those platforms.
In the first quarter of 2021, our institution overall performed at a Top Box score of 60.2 for our ability to assist patients with staying connected to their family/caregiver during their inpatient visit. With more than 6700 returned surveys during that time period, our hospitals earned Top Box scores ranging between 48.0 and 75.3. At this time, obtaining a national benchmark ranking is not possible, because the question regarding connectedness is unique to Northwell inpatient settings. As other health care organizations adopt this customized question, further peer-to-peer measurements can be established.
Regarding virtual interviews, 25 patients were initially contacted to determine their interest in participating. Of that sample, 17 patients were engaged over the phone, representing a reach rate of 68%. Overall, 10 interviews were scheduled; 7 patients did not show up, resulting in 3 completed interviews. During follow-up, “no-show” participants either gave no response or stated they had a conflict at their originally scheduled time but were not interested in rescheduling due to personal circumstances. Through such conversations, ongoing health complications were found to be a reoccurring barrier to participation.
Each of the participating patients had experienced being placed on a ventilator. They described their hospitalization as a time of “confusion and despair” in the first days after extubation. After we reviewed interview recordings, a reoccurring theme across all interviews was the feeling of gratitude. Patients expressed deep and heartfelt appreciation for being given the opportunity to connect as a family. One patient described virtual visitation sessions as her “only tether to reality when nothing else made sense.”
Interestingly enough, none of the participants knew that virtual visitation was an option and/or thought to inquire about it before a hospital staff member offered to set up a session. Patients recounted how they were weak and physically unable to connect to the sessions without significant assistance. They reported examples of not having the physical strength to hold up the tablet or needing a staff member to facilitate the conversation because the patient could not speak loudly enough and/or they were having difficulty hearing over background medical equipment noises. Participants also described times when a nurse or social worker would stand and hold the tablet for 20 to 30 minutes at a time, further describing mixed feelings of gratitude, guilt for “taking up their time,” and a desire for more privacy to have those precious conversations.
Discussion
Our institution encountered various barriers when establishing, implementing, and sustaining virtual visitation. The acquisition and bulk purchasing of devices, so that each hospital unit and department had adequate par levels during a high-demand time frame, was an initial challenge. Ensuring appropriate safeguards, software programming, and access to WiFi required ingenuity from IT teams. Leaders sought to advocate for the importance of prioritizing virtual visitation alongside clinical interventions. For team members, education was needed to build awareness, learn how to navigate technology, and troubleshoot, in real-time, issues such as poor connectivity. However, despite these organizational struggles, the hospital’s frontline professionals fully recognized and understood the humanistic value of connecting ill patients with their loved ones. Harnessing their teamwork, empathy, and innovative spirits, they forged through such difficulties to create meaningful interactions.
Although virtual visitation occurred prior to the COVID-19 pandemic, particularly in subspecialty areas such as neonatal intensive care units,6 it was not commonplace in most adult inpatient care settings. However, now that virtual means to communication are widely accepted and preferred, our hospital anticipates these offerings will become a broad patient expectation and, therefore, part of standard hospital care and operations. Health care leaders and interdisciplinary teams must therefore prioritize virtual visitation protocols, efforts, and future programming. It is no longer an exception to the rule, but rather a critical approach when ensuring quality communication between patients, families, and care teams.
We strive to continually improve by including user feedback as part of an interactive design process. For a broader, more permanent installation of virtual visitation, health care organizations must proactively promote this capability as a valued option. Considering health literacy and comfort with technology, functionality, and logistics must be carefully explained to patients and their families. This may require additional staff training so that they are knowledgeable, comfortable with, and able to troubleshoot questions/concerns in real time. There needs to be an adequate number of mobile devices available at a unit or departmental level to meet short-term and long-term demands. Additionally, now that we have emerged from our initial crisis-based mentality, it is time to consider alternatives to alleviate the need for staff assistance, such as mounts to hold devices and enabling voice controls.
Conclusion
As an organization grounded in the spirit of innovation, Northwell has been able to quickly pivot, adopting virtual visitation to address emerging and complex communication needs. Taking a best practice established during a crisis period and engraining it into sustainable organizational culture and operations requires visionary leadership, strong teamwork, and an unbridled commitment to patient and family centeredness. Despite unprecedented challenges, our commitment to listening to the “voice” of patients and families never wavered. Using their insights and feedback as critical components to the decision-making process, there is much work ahead within the realm of virtual visitation.
Acknowledgements: The authors would like to acknowledge the Northwell Health providers, frontline health care professionals, and team members who worked tirelessly to care for its community during initial COVID-19 waves and every day thereafter. Heartfelt gratitude to Northwell’s senior leaders for the visionary leadership; the OCIO and hospital-based IT teams for their swift collaboration; and dedicated Culture Leaders, Patient Experience team members, and redeployed staff for their unbridled passion for caring for patients and families. Special thanks to Agnes Barden, DNP, RN, CPXP, Joseph Narvaez, MBA, and Natalie Bashkin, MBA, from the system Office of Patient & Customer Experience, and Carolyne Burgess, MPH, from the Digital Patient Experience teams, for their participation, leadership, and syngeristic partnerships.
Corresponding Author: Nicole Giammarinaro, MSN, RN, CPXP, Director, Patient & Customer Experience, Northwell Health, 2000 Marcus Ave, Lake Success, NY 11042; [email protected].
Financial disclosures: Sven Gierlinger serves on the Speakers Bureau for Northwell Health and as an Executive Board Member for The Beryl Institute.
1. New York State Department of Health. Health advisory: COVID-19 guidance for hospital operators regarding visitation. March 18, 2020. https://coronavirus.health.ny.gov/system/files/documents/2020/03/covid19-hospital-visitation-guidance-3.18.20.pdf
2. Zhang Y. Family functioning in the context of an adult family member with illness: a concept analysis. J Clin Nurs. 2018;27(15-16):3205-3224. doi:10.1111/jocn.14500
3. Institute for Patient- & Family-Centered Care. Better Together: Partnering with Families. https://www.ipfcc.org/bestpractices/better-together-ny.html
4. Marceglia S, Bonacina S, Zaccaria V, et al. How might the iPad change healthcare? J R Soc Med. 2012;105(6):233-241. doi:10.1258/jrsm.2012.110296
5. Short B, Parekh M, Ryan P, et al. Rapid implementation of a mobile prone team during the COVID-19 pandemic. J Crit Care. 2020;60:230-234. doi:10.1016/j.jcrc.2020.08.020
6. Yeo C, Ho SK, Khong K, Lau Y. Virtual visitation in the neonatal intensive care: experience with the use of internet and telemedicine in a tertiary neonatal unit. Perm J. 2011;15(3):32-36.
1. New York State Department of Health. Health advisory: COVID-19 guidance for hospital operators regarding visitation. March 18, 2020. https://coronavirus.health.ny.gov/system/files/documents/2020/03/covid19-hospital-visitation-guidance-3.18.20.pdf
2. Zhang Y. Family functioning in the context of an adult family member with illness: a concept analysis. J Clin Nurs. 2018;27(15-16):3205-3224. doi:10.1111/jocn.14500
3. Institute for Patient- & Family-Centered Care. Better Together: Partnering with Families. https://www.ipfcc.org/bestpractices/better-together-ny.html
4. Marceglia S, Bonacina S, Zaccaria V, et al. How might the iPad change healthcare? J R Soc Med. 2012;105(6):233-241. doi:10.1258/jrsm.2012.110296
5. Short B, Parekh M, Ryan P, et al. Rapid implementation of a mobile prone team during the COVID-19 pandemic. J Crit Care. 2020;60:230-234. doi:10.1016/j.jcrc.2020.08.020
6. Yeo C, Ho SK, Khong K, Lau Y. Virtual visitation in the neonatal intensive care: experience with the use of internet and telemedicine in a tertiary neonatal unit. Perm J. 2011;15(3):32-36.
Feasibility of a Saliva-Based COVID-19 Screening Program in Abu Dhabi Primary Schools
From Health Center, New York University Abu Dhabi, Abu Dhabi, United Arab Emirates (Dr. Virji and Aisha Al Hamiz), Public Health, Abu Dhabi Public Health Center, Abu Dhabi, United Arab Emirates (Drs. Al Hajeri, Al Shehhi, Al Memari, and Ahlam Al Maskari), College of Medicine and Health Sciences, Khalifa University, Abu Dhabi, United Arab Emirates, Department of Medicine, Sheikh Shakhbout Medical City, Abu Dhabi, United Arab Emirates (Dr. Alhajri), Public Health Research Center, New York University Abu Dhabi, Abu Dhabi, United Arab Emirates, Oxford University Hospitals NHS Foundation Trust, Oxford, England, and the MRC Epidemiology Unit, University of Cambridge, Cambridge, England (Dr. Ali).
Objective: The pandemic has forced closures of primary schools, resulting in loss of learning time on a global scale. In addition to face coverings, social distancing, and hand hygiene, an efficient testing method is important to mitigate the spread of COVID-19 in schools. We evaluated the feasibility of a saliva-based SARS-CoV-2 polymerase chain reaction testing program among 18 primary schools in the Emirate of Abu Dhabi, United Arab Emirates. Qualitative results show that children 4 to 5 years old had difficulty producing an adequate saliva specimen compared to those 6 to 12 years old.
Methods: A short training video on saliva collection beforehand helps demystify the process for students and parents alike. Informed consent was challenging yet should be done beforehand by school health nurses or other medical professionals to reassure parents and maximize participation.
Results: Telephone interviews with school administrators resulted in an 83% response rate. Overall, 93% of school administrators had a positive experience with saliva testing and felt the program improved the safety of their schools. The ongoing use of saliva testing for SARS-CoV-2 was supported by 73% of respondents.
Conclusion: On-campus saliva testing is a feasible option for primary schools to screen for COVID-19 in their student population to help keep their campuses safe and open for learning.
Keywords: COVID-19; saliva testing; mitigation; primary school.
The COVID-19 pandemic is a leading cause of morbidity and mortality worldwide and continues to exhaust health care resources on a large scale.1 Efficient testing is critical to identify cases early and to help mitigate the deleterious effects of the pandemic.2 Saliva polymerase chain reaction (PCR) nucleic acid amplification testing (NAAT) is more comfortable than nasopharyngeal (NP) NAAT and has been validated as a test for SARS-CoV-2.1 Although children are less susceptible to severe disease, primary schools are considered a vector for transmission and community spread.3 Efficient and scalable methods of routine testing are needed globally to help keep schools open. Saliva testing has proven a useful resource for this population.4,5
Abu Dhabi is the largest Emirate in the United Arab Emirates (UAE), with an estimated population of 2.5 million.6 The first case of COVID-19 was discovered in the UAE on January 29, 2020.7 The UAE has been recognized worldwide for its robust pandemic response. Along with the coordinated and swift application of public health measures, the country has one of the highest COVID-19 testing rates per capita and one of the highest vaccination rates worldwide.8,9 The Abu Dhabi Public Health Center (ADPHC) works alongside the Ministry of Education (MOE) to establish testing, quarantine, and general safety guidelines for primary schools. In December 2020, the ADPHC partnered with a local, accredited diagnostic laboratory to test the feasibility of a saliva-based screening program for COVID-19 directly on school campuses for 18 primary schools in the Emirate.
Saliva-based PCR testing for COVID-19 was approved for use in schools in the UAE on January 24, 2021.10 As part of a greater mitigation strategy to reduce both school-based transmission and, hence, community spread, the ADPHC focused its on-site testing program on children aged 4 to 12 years. The program required collaboration among medical professionals, school administrators and teachers, students, and parents. Our study evaluates the feasibility of implementing a saliva-based COVID-19 screening program directly on primary school campuses involving children as young as 4 years of age.
Methods
The ADPHC, in collaboration with G42 Biogenix Labs, conducted a saliva SARS-CoV-2 NAAT testing program in 18 primary schools in the Emirate. Schools were selected based on outbreak prevalence at the time and focused on “hot spot” areas. The school on-site saliva testing program included children aged 4 to 12 years old in a “bubble” attendance model during the school day. This model involved children being assigned to groups or “pods.” This allowed us to limit a potential outbreak to a single pod, as opposed to risk exposing the entire school, should a single student test positive. The well-established SalivaDirect protocol developed at Yale University was used for testing and included an RNA extraction-free, RT-qPCR method for SARS-CoV-2 detection.11
We conducted a qualitative study involving telephone interviews of school administrators to evaluate their experience with the ADPHC testing program at their schools. In addition, we interviewed the G42 Biogenix Lab providers to understand the logistics that supported on-campus collection of saliva specimens for this age group. We also gathered the attitudes of school children before and after testing. This study was reviewed and approved by the Abu Dhabi Health Research and Technology Committee and the Institutional Review Board (IRB), New York University Abu Dhabi (NYUAD).
Sample and recruitment
The original sample collection of saliva specimens was performed by the ADPHC in collaboration with G42 Biogenix Lab providers on school campuses between December 6 and December 10, 2020. During this time, schools operated in a hybrid teaching model, where learning took place both online and in person. Infection control measures were deployed based on ADPHC standards and guidelines. Nurses utilized appropriate patient protective equipment, frequent hand hygiene, and social distancing during the collection process. Inclusion criteria included asymptomatic students aged 4 to 12 years attending in-person classes on campus. Students with respiratory symptoms who were asked to stay home or those not attending in-person classes were excluded.
Data collection
Data with regard to school children’s attitudes before and after testing were compiled through an online survey sent randomly to participants postintervention. Data from school administrators were collected through video and telephone interviews between April 14 and April 29, 2021. We first interviewed G42 Biogenix Lab providers to obtain previously acquired qualitative and quantitative data, which were collected during the intervention itself. After obtaining this information, we designed a questionnaire and proceeded with a structured interview process for school officials.
We interviewed school principals and administrators to collect their overall experiences with the saliva testing program. Before starting each interview, we established the interviewees preferred language, either English or Arabic. We then introduced the meeting attendees and provided study details, aims, and objectives, and described collaborating entities. We obtained verbal informed consent from a script approved by the NYUAD IRB and then proceeded with the interview, which included 4 questions. The first 3 questions were answered on a 5-point Likert scale model that consisted of 5 answer options: 5 being completely agree, 4 agree, 3 somewhat agree, 2 somewhat disagree, and 1 completely disagree. The fourth question invited open-ended feedback and comments on the following statements:
- I believe the COVID-19 saliva testing program improved the safety for my school campus.
- Our community had an overall positive experience with the COVID saliva testing.
- We would like to continue a saliva-based COVID testing program on our school campus.
- Please provide any additional comments you feel important about the program.
During the interview, we transcribed the answers as the interviewee was answering. We then translated those in Arabic into English and collected the data in 1 Excel spreadsheet. School interviewees and school names were de-identified in the collection and storage process.
Results
A total of 2011 saliva samples were collected from 18 different primary school campuses. Samples were sent the same day to G42 Biogenix Labs in Abu Dhabi for COVID PCR testing. A team consisting of 5 doctors providing general oversight, along with 2 to 6 nurses per site, were able to manage the collection process for all 18 school campuses. Samples were collected between 8
Sample stations were set up in either the school auditorium or gymnasium to ensure appropriate crowd control and ventilation. Teachers and other school staff, including public safety, were able to manage lines and the shuttling of students back and forth from classes to testing stations, which allowed medical staff to focus on sample collection.
Informed consent was obtained by prior electronic communication to parents from school staff, asking them to agree to allow their child to participate in the testing program. Informed consent was identified as a challenge: Getting parents to understand that saliva testing was more comfortable than NP testing, and that the results were only being used to help keep the school safe, took time. School staff are used to obtaining consent from parents for field trips, but this was clearly more challenging for them.
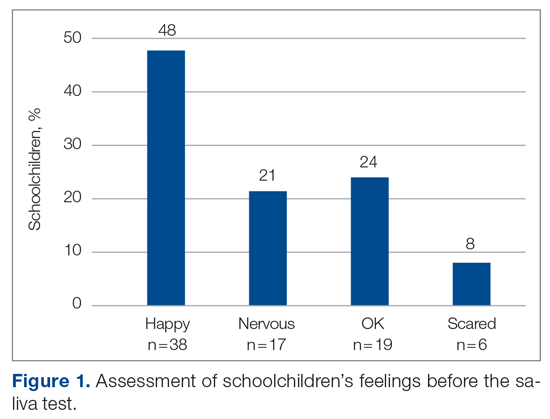
The saliva collection process per child took more time than expected. Children fasted for 45 minutes before saliva collection. We used an active drool technique, which required children to pool saliva in their mouth then express it into a collection tube. Adults can generally do this on command, but we found it took 10 to 12 minutes per child. Saliva production was cued by asking the children to think about food, and by showing them pictures and TV commercials depicting food. Children 4 to 5 years old had more difficulty with the process despite active cueing, while those 6 to 12 years old had an easier time with the process. We collected data on a cohort of 80 children regarding their attitudes pre (Figure 1) and post collection (Figure 2). Children felt happier, less nervous, and less scared after collection than before collection. This trend reassured us that future collections would be easier for students.
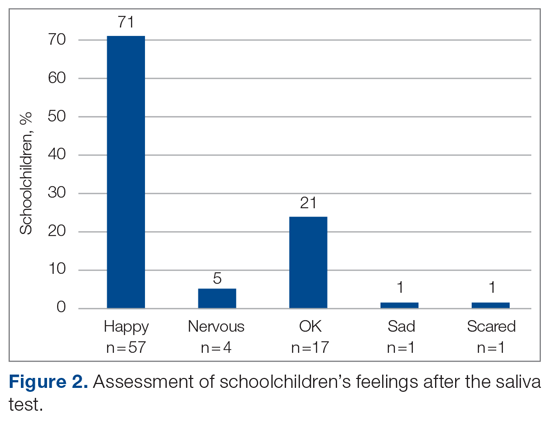
A total of 15 of 18 school principals completed the telephone interview, yielding a response rate of 83%. Overall, 93% of the school principals agreed or completely agreed that the COVID-19 saliva testing program improved school safety; 93% agreed or completely agreed that they had an overall positive experience with the program; and 73% supported the ongoing use of saliva testing in their schools (Table 1). Administrators’ open-ended comments on their experience were positive overall (Table 2).
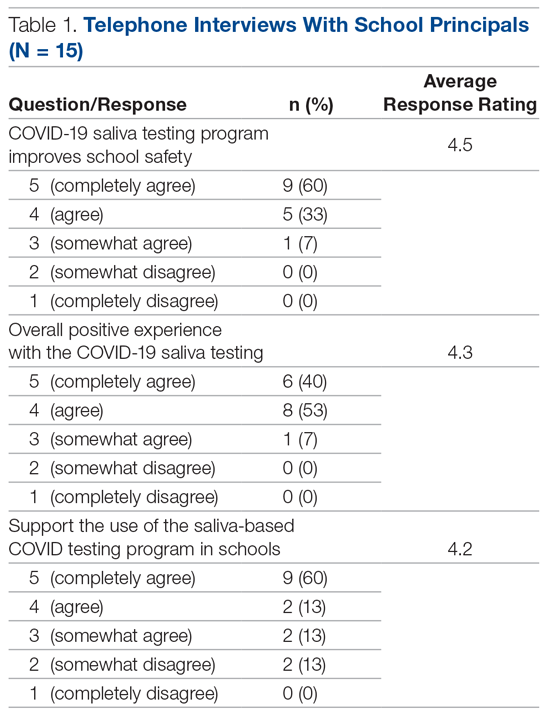
Discussion
By March 2020, many kindergarten to grade 12 public and private schools suspended in-person classes due to the pandemic and turned to online learning platforms. The negative impact of school closures on academic achievement is projected to be significant.7,12,13 Ensuring schools can stay open and run operations safely will require routine SARS-CoV-2 testing. Our study investigated the feasibility of routine saliva testing on children aged 4 to 12 years on their school campuses. The ADPHC school on-site saliva testing program involved bringing lab providers onto 18 primary school campuses and required cooperation among parents, students, school administrators, and health care professionals.

Children younger than 6 years had difficulty producing an adequate saliva specimen, whereas those 6 to 12 years did so with relative ease when cued by thoughts or pictures of food while waiting in line for collection. Schools considering on-site testing programs should consider the age range of 6 to 12 years as a viable age range for saliva screening. Children should fast for a minimum of 45 minutes prior to saliva collection and should be cued by thoughts of food, food pictures, or food commercials. Setting up a sampling station close to the cafeteria where students can smell meal preparation may also help.14,15 Sampling before breakfast or lunch, when children are potentially at their hungriest, should also be considered.
The greatest challenge was obtaining informed consent from parents who were not yet familiar with the reliability of saliva testing as a tool for SARS-CoV-2 screening or with the saliva collection process as a whole. Informed consent was initially done electronically, lacking direct human interaction to answer parents’ questions. Parents who refused had a follow-up call from the school nurse to further explain the logistics and rationale for saliva screening. Having medical professionals directly answer parents’ questions was helpful. Parents were reassured that the process was painless, confidential, and only to be used for school safety purposes. Despite school administrators being experienced in obtaining consent from parents for field trips, obtaining informed consent for a medical testing procedure is more complicated, and parents aren’t accustomed to providing such consent in a school environment. Schools considering on-site testing should ensure that their school nurse or other health care providers are on the front line obtaining informed consent and allaying parents’ fears.
School staff were able to effectively provide crowd control for testing, and children felt at ease being in a familiar environment. Teachers and public safety officers are well-equipped at managing the shuttling of students to class, to lunch, to physical education, and, finally, to dismissal. They were equally equipped at handling the logistics of students to and from testing, including minimizing crowds and helping students feel at ease during the process. This effective collaboration allowed the lab personnel to focus on sample collection and storage, while school staff managed all other aspects of the children’s safety and care.
Conclusion
Overall, school administrators had a positive experience with the testing program, felt the program improved the safety of their schools, and supported the ongoing use of saliva testing for SARS-CoV-2 on their school campuses. Children aged 6 years and older were able to provide adequate saliva samples, and children felt happier and less nervous after the process, indicating repeatability. Our findings highlight the feasibility of an integrated on-site saliva testing model for primary school campuses. Further research is needed to determine the scalability of such a model and whether the added compliance and safety of on-site testing compensates for the potential loss of learning time that testing during school hours would require.
Corresponding author: Ayaz Virji, MD, New York University Abu Dhabi, PO Box 129188, Abu Dhabi, United Arab Emirates; [email protected].
Financial disclosures: None.
1. Kuehn BM. Despite improvements, COVID-19’s health care disruptions persist. JAMA. 2021;325(23):2335. doi:10.1001/jama.2021.9134
2. National Institute on Aging. Why COVID-19 testing is the key to getting back to normal. September 4, 2020. Accessed September 8, 2021. https://www.nia.nih.gov/news/why-covid-19-testing-key-getting-back-normal
3. Centers for Disease Control and Prevention. Science brief: Transmission of SARS-CoV-2 in K-12 schools. Updated July 9, 2021. Accessed September 8, 2021. https://www.cdc.gov/coronavirus/2019-ncov/science/science-briefs/transmission_k_12_schools.html
4. Butler-Laporte G, Lawandi A, Schiller I, et al. Comparison of saliva and nasopharyngeal swab nucleic acid amplification testing for detection of SARS-CoV-2: a systematic review and meta-analysis. JAMA Intern Med. 2021;181(3):353-360. doi:10.1001/jamainternmed.2020.8876
5. Al Suwaidi H, Senok A, Varghese R, et al. Saliva for molecular detection of SARS-CoV-2 in school-age children. Clin Microbiol Infect. 2021;27(9):1330-1335. doi:10.1016/j.cmi.2021.02.009
6. Abu Dhabi. Accessed September 8, 2021. https://u.ae/en/about-the-uae/the-seven-emirates/abu-dhabi
7. Alsuwaidi AR, Al Hosani FI, Al Memari S, et al. Seroprevalence of COVID-19 infection in the Emirate of Abu Dhabi, United Arab Emirates: a population-based cross-sectional study. Int J Epidemiol. 2021;50(4):1077-1090. doi:10.1093/ije/dyab077
8. Al Hosany F, Ganesan S, Al Memari S, et al. Response to COVID-19 pandemic in the UAE: a public health perspective. J Glob Health. 2021;11:03050. doi:10.7189/jogh.11.03050
9. Bremmer I. The best global responses to the COVID-19 pandemic, 1 year later. Time Magazine. Updated February 23, 2021. Accessed September 8, 2021. https://time.com/5851633/best-global-responses-covid-19/
10. Department of Health, Abu Dhabi. Laboratory diagnostic test for COVID-19: update regarding saliva-based testing using RT-PCR test. 2021.
11. Vogels C, Brackney DE, Kalinich CC, et al. SalivaDirect: RNA extraction-free SARS-CoV-2 diagnostics. Protocols.io. Accessed September 8, 2021. https://www.protocols.io/view/salivadirect-rna-extraction-free-sars-cov-2-diagno-bh6jj9cn?version_warning=no
12. Education Endowment Foundation. Impact of school closures on the attainment gap: rapid evidence assessment. June 2020. Accessed September 8, 2021. https://www.researchgate.net/publication/342501263_EEF_2020_-_Impact_of_School_Closures_on_the_Attainment_Gap
13. United Nations. Policy brief: Education during COVID-19 and beyond. Accessed September 8, 2021. https://www.un.org/development/desa/dspd/wp-content/uploads/sites/22/2020/08/sg_policy_brief_covid-19_and_education_august_2020.pdf
14. Schiffman SS, Miletic ID. Effect of taste and smell on secretion rate of salivary IgA in elderly and young persons. J Nutr Health Aging. 1999;3(3):158-164.
15. Lee VM, Linden RW. The effect of odours on stimulated parotid salivary flow in humans. Physiol Behav. 1992;52(6):1121-1125. doi:10.1016/0031-9384(92)90470-m
From Health Center, New York University Abu Dhabi, Abu Dhabi, United Arab Emirates (Dr. Virji and Aisha Al Hamiz), Public Health, Abu Dhabi Public Health Center, Abu Dhabi, United Arab Emirates (Drs. Al Hajeri, Al Shehhi, Al Memari, and Ahlam Al Maskari), College of Medicine and Health Sciences, Khalifa University, Abu Dhabi, United Arab Emirates, Department of Medicine, Sheikh Shakhbout Medical City, Abu Dhabi, United Arab Emirates (Dr. Alhajri), Public Health Research Center, New York University Abu Dhabi, Abu Dhabi, United Arab Emirates, Oxford University Hospitals NHS Foundation Trust, Oxford, England, and the MRC Epidemiology Unit, University of Cambridge, Cambridge, England (Dr. Ali).
Objective: The pandemic has forced closures of primary schools, resulting in loss of learning time on a global scale. In addition to face coverings, social distancing, and hand hygiene, an efficient testing method is important to mitigate the spread of COVID-19 in schools. We evaluated the feasibility of a saliva-based SARS-CoV-2 polymerase chain reaction testing program among 18 primary schools in the Emirate of Abu Dhabi, United Arab Emirates. Qualitative results show that children 4 to 5 years old had difficulty producing an adequate saliva specimen compared to those 6 to 12 years old.
Methods: A short training video on saliva collection beforehand helps demystify the process for students and parents alike. Informed consent was challenging yet should be done beforehand by school health nurses or other medical professionals to reassure parents and maximize participation.
Results: Telephone interviews with school administrators resulted in an 83% response rate. Overall, 93% of school administrators had a positive experience with saliva testing and felt the program improved the safety of their schools. The ongoing use of saliva testing for SARS-CoV-2 was supported by 73% of respondents.
Conclusion: On-campus saliva testing is a feasible option for primary schools to screen for COVID-19 in their student population to help keep their campuses safe and open for learning.
Keywords: COVID-19; saliva testing; mitigation; primary school.
The COVID-19 pandemic is a leading cause of morbidity and mortality worldwide and continues to exhaust health care resources on a large scale.1 Efficient testing is critical to identify cases early and to help mitigate the deleterious effects of the pandemic.2 Saliva polymerase chain reaction (PCR) nucleic acid amplification testing (NAAT) is more comfortable than nasopharyngeal (NP) NAAT and has been validated as a test for SARS-CoV-2.1 Although children are less susceptible to severe disease, primary schools are considered a vector for transmission and community spread.3 Efficient and scalable methods of routine testing are needed globally to help keep schools open. Saliva testing has proven a useful resource for this population.4,5
Abu Dhabi is the largest Emirate in the United Arab Emirates (UAE), with an estimated population of 2.5 million.6 The first case of COVID-19 was discovered in the UAE on January 29, 2020.7 The UAE has been recognized worldwide for its robust pandemic response. Along with the coordinated and swift application of public health measures, the country has one of the highest COVID-19 testing rates per capita and one of the highest vaccination rates worldwide.8,9 The Abu Dhabi Public Health Center (ADPHC) works alongside the Ministry of Education (MOE) to establish testing, quarantine, and general safety guidelines for primary schools. In December 2020, the ADPHC partnered with a local, accredited diagnostic laboratory to test the feasibility of a saliva-based screening program for COVID-19 directly on school campuses for 18 primary schools in the Emirate.
Saliva-based PCR testing for COVID-19 was approved for use in schools in the UAE on January 24, 2021.10 As part of a greater mitigation strategy to reduce both school-based transmission and, hence, community spread, the ADPHC focused its on-site testing program on children aged 4 to 12 years. The program required collaboration among medical professionals, school administrators and teachers, students, and parents. Our study evaluates the feasibility of implementing a saliva-based COVID-19 screening program directly on primary school campuses involving children as young as 4 years of age.
Methods
The ADPHC, in collaboration with G42 Biogenix Labs, conducted a saliva SARS-CoV-2 NAAT testing program in 18 primary schools in the Emirate. Schools were selected based on outbreak prevalence at the time and focused on “hot spot” areas. The school on-site saliva testing program included children aged 4 to 12 years old in a “bubble” attendance model during the school day. This model involved children being assigned to groups or “pods.” This allowed us to limit a potential outbreak to a single pod, as opposed to risk exposing the entire school, should a single student test positive. The well-established SalivaDirect protocol developed at Yale University was used for testing and included an RNA extraction-free, RT-qPCR method for SARS-CoV-2 detection.11
We conducted a qualitative study involving telephone interviews of school administrators to evaluate their experience with the ADPHC testing program at their schools. In addition, we interviewed the G42 Biogenix Lab providers to understand the logistics that supported on-campus collection of saliva specimens for this age group. We also gathered the attitudes of school children before and after testing. This study was reviewed and approved by the Abu Dhabi Health Research and Technology Committee and the Institutional Review Board (IRB), New York University Abu Dhabi (NYUAD).
Sample and recruitment
The original sample collection of saliva specimens was performed by the ADPHC in collaboration with G42 Biogenix Lab providers on school campuses between December 6 and December 10, 2020. During this time, schools operated in a hybrid teaching model, where learning took place both online and in person. Infection control measures were deployed based on ADPHC standards and guidelines. Nurses utilized appropriate patient protective equipment, frequent hand hygiene, and social distancing during the collection process. Inclusion criteria included asymptomatic students aged 4 to 12 years attending in-person classes on campus. Students with respiratory symptoms who were asked to stay home or those not attending in-person classes were excluded.
Data collection
Data with regard to school children’s attitudes before and after testing were compiled through an online survey sent randomly to participants postintervention. Data from school administrators were collected through video and telephone interviews between April 14 and April 29, 2021. We first interviewed G42 Biogenix Lab providers to obtain previously acquired qualitative and quantitative data, which were collected during the intervention itself. After obtaining this information, we designed a questionnaire and proceeded with a structured interview process for school officials.
We interviewed school principals and administrators to collect their overall experiences with the saliva testing program. Before starting each interview, we established the interviewees preferred language, either English or Arabic. We then introduced the meeting attendees and provided study details, aims, and objectives, and described collaborating entities. We obtained verbal informed consent from a script approved by the NYUAD IRB and then proceeded with the interview, which included 4 questions. The first 3 questions were answered on a 5-point Likert scale model that consisted of 5 answer options: 5 being completely agree, 4 agree, 3 somewhat agree, 2 somewhat disagree, and 1 completely disagree. The fourth question invited open-ended feedback and comments on the following statements:
- I believe the COVID-19 saliva testing program improved the safety for my school campus.
- Our community had an overall positive experience with the COVID saliva testing.
- We would like to continue a saliva-based COVID testing program on our school campus.
- Please provide any additional comments you feel important about the program.
During the interview, we transcribed the answers as the interviewee was answering. We then translated those in Arabic into English and collected the data in 1 Excel spreadsheet. School interviewees and school names were de-identified in the collection and storage process.
Results
A total of 2011 saliva samples were collected from 18 different primary school campuses. Samples were sent the same day to G42 Biogenix Labs in Abu Dhabi for COVID PCR testing. A team consisting of 5 doctors providing general oversight, along with 2 to 6 nurses per site, were able to manage the collection process for all 18 school campuses. Samples were collected between 8
Sample stations were set up in either the school auditorium or gymnasium to ensure appropriate crowd control and ventilation. Teachers and other school staff, including public safety, were able to manage lines and the shuttling of students back and forth from classes to testing stations, which allowed medical staff to focus on sample collection.
Informed consent was obtained by prior electronic communication to parents from school staff, asking them to agree to allow their child to participate in the testing program. Informed consent was identified as a challenge: Getting parents to understand that saliva testing was more comfortable than NP testing, and that the results were only being used to help keep the school safe, took time. School staff are used to obtaining consent from parents for field trips, but this was clearly more challenging for them.

The saliva collection process per child took more time than expected. Children fasted for 45 minutes before saliva collection. We used an active drool technique, which required children to pool saliva in their mouth then express it into a collection tube. Adults can generally do this on command, but we found it took 10 to 12 minutes per child. Saliva production was cued by asking the children to think about food, and by showing them pictures and TV commercials depicting food. Children 4 to 5 years old had more difficulty with the process despite active cueing, while those 6 to 12 years old had an easier time with the process. We collected data on a cohort of 80 children regarding their attitudes pre (Figure 1) and post collection (Figure 2). Children felt happier, less nervous, and less scared after collection than before collection. This trend reassured us that future collections would be easier for students.

A total of 15 of 18 school principals completed the telephone interview, yielding a response rate of 83%. Overall, 93% of the school principals agreed or completely agreed that the COVID-19 saliva testing program improved school safety; 93% agreed or completely agreed that they had an overall positive experience with the program; and 73% supported the ongoing use of saliva testing in their schools (Table 1). Administrators’ open-ended comments on their experience were positive overall (Table 2).

Discussion
By March 2020, many kindergarten to grade 12 public and private schools suspended in-person classes due to the pandemic and turned to online learning platforms. The negative impact of school closures on academic achievement is projected to be significant.7,12,13 Ensuring schools can stay open and run operations safely will require routine SARS-CoV-2 testing. Our study investigated the feasibility of routine saliva testing on children aged 4 to 12 years on their school campuses. The ADPHC school on-site saliva testing program involved bringing lab providers onto 18 primary school campuses and required cooperation among parents, students, school administrators, and health care professionals.

Children younger than 6 years had difficulty producing an adequate saliva specimen, whereas those 6 to 12 years did so with relative ease when cued by thoughts or pictures of food while waiting in line for collection. Schools considering on-site testing programs should consider the age range of 6 to 12 years as a viable age range for saliva screening. Children should fast for a minimum of 45 minutes prior to saliva collection and should be cued by thoughts of food, food pictures, or food commercials. Setting up a sampling station close to the cafeteria where students can smell meal preparation may also help.14,15 Sampling before breakfast or lunch, when children are potentially at their hungriest, should also be considered.
The greatest challenge was obtaining informed consent from parents who were not yet familiar with the reliability of saliva testing as a tool for SARS-CoV-2 screening or with the saliva collection process as a whole. Informed consent was initially done electronically, lacking direct human interaction to answer parents’ questions. Parents who refused had a follow-up call from the school nurse to further explain the logistics and rationale for saliva screening. Having medical professionals directly answer parents’ questions was helpful. Parents were reassured that the process was painless, confidential, and only to be used for school safety purposes. Despite school administrators being experienced in obtaining consent from parents for field trips, obtaining informed consent for a medical testing procedure is more complicated, and parents aren’t accustomed to providing such consent in a school environment. Schools considering on-site testing should ensure that their school nurse or other health care providers are on the front line obtaining informed consent and allaying parents’ fears.
School staff were able to effectively provide crowd control for testing, and children felt at ease being in a familiar environment. Teachers and public safety officers are well-equipped at managing the shuttling of students to class, to lunch, to physical education, and, finally, to dismissal. They were equally equipped at handling the logistics of students to and from testing, including minimizing crowds and helping students feel at ease during the process. This effective collaboration allowed the lab personnel to focus on sample collection and storage, while school staff managed all other aspects of the children’s safety and care.
Conclusion
Overall, school administrators had a positive experience with the testing program, felt the program improved the safety of their schools, and supported the ongoing use of saliva testing for SARS-CoV-2 on their school campuses. Children aged 6 years and older were able to provide adequate saliva samples, and children felt happier and less nervous after the process, indicating repeatability. Our findings highlight the feasibility of an integrated on-site saliva testing model for primary school campuses. Further research is needed to determine the scalability of such a model and whether the added compliance and safety of on-site testing compensates for the potential loss of learning time that testing during school hours would require.
Corresponding author: Ayaz Virji, MD, New York University Abu Dhabi, PO Box 129188, Abu Dhabi, United Arab Emirates; [email protected].
Financial disclosures: None.
From Health Center, New York University Abu Dhabi, Abu Dhabi, United Arab Emirates (Dr. Virji and Aisha Al Hamiz), Public Health, Abu Dhabi Public Health Center, Abu Dhabi, United Arab Emirates (Drs. Al Hajeri, Al Shehhi, Al Memari, and Ahlam Al Maskari), College of Medicine and Health Sciences, Khalifa University, Abu Dhabi, United Arab Emirates, Department of Medicine, Sheikh Shakhbout Medical City, Abu Dhabi, United Arab Emirates (Dr. Alhajri), Public Health Research Center, New York University Abu Dhabi, Abu Dhabi, United Arab Emirates, Oxford University Hospitals NHS Foundation Trust, Oxford, England, and the MRC Epidemiology Unit, University of Cambridge, Cambridge, England (Dr. Ali).
Objective: The pandemic has forced closures of primary schools, resulting in loss of learning time on a global scale. In addition to face coverings, social distancing, and hand hygiene, an efficient testing method is important to mitigate the spread of COVID-19 in schools. We evaluated the feasibility of a saliva-based SARS-CoV-2 polymerase chain reaction testing program among 18 primary schools in the Emirate of Abu Dhabi, United Arab Emirates. Qualitative results show that children 4 to 5 years old had difficulty producing an adequate saliva specimen compared to those 6 to 12 years old.
Methods: A short training video on saliva collection beforehand helps demystify the process for students and parents alike. Informed consent was challenging yet should be done beforehand by school health nurses or other medical professionals to reassure parents and maximize participation.
Results: Telephone interviews with school administrators resulted in an 83% response rate. Overall, 93% of school administrators had a positive experience with saliva testing and felt the program improved the safety of their schools. The ongoing use of saliva testing for SARS-CoV-2 was supported by 73% of respondents.
Conclusion: On-campus saliva testing is a feasible option for primary schools to screen for COVID-19 in their student population to help keep their campuses safe and open for learning.
Keywords: COVID-19; saliva testing; mitigation; primary school.
The COVID-19 pandemic is a leading cause of morbidity and mortality worldwide and continues to exhaust health care resources on a large scale.1 Efficient testing is critical to identify cases early and to help mitigate the deleterious effects of the pandemic.2 Saliva polymerase chain reaction (PCR) nucleic acid amplification testing (NAAT) is more comfortable than nasopharyngeal (NP) NAAT and has been validated as a test for SARS-CoV-2.1 Although children are less susceptible to severe disease, primary schools are considered a vector for transmission and community spread.3 Efficient and scalable methods of routine testing are needed globally to help keep schools open. Saliva testing has proven a useful resource for this population.4,5
Abu Dhabi is the largest Emirate in the United Arab Emirates (UAE), with an estimated population of 2.5 million.6 The first case of COVID-19 was discovered in the UAE on January 29, 2020.7 The UAE has been recognized worldwide for its robust pandemic response. Along with the coordinated and swift application of public health measures, the country has one of the highest COVID-19 testing rates per capita and one of the highest vaccination rates worldwide.8,9 The Abu Dhabi Public Health Center (ADPHC) works alongside the Ministry of Education (MOE) to establish testing, quarantine, and general safety guidelines for primary schools. In December 2020, the ADPHC partnered with a local, accredited diagnostic laboratory to test the feasibility of a saliva-based screening program for COVID-19 directly on school campuses for 18 primary schools in the Emirate.
Saliva-based PCR testing for COVID-19 was approved for use in schools in the UAE on January 24, 2021.10 As part of a greater mitigation strategy to reduce both school-based transmission and, hence, community spread, the ADPHC focused its on-site testing program on children aged 4 to 12 years. The program required collaboration among medical professionals, school administrators and teachers, students, and parents. Our study evaluates the feasibility of implementing a saliva-based COVID-19 screening program directly on primary school campuses involving children as young as 4 years of age.
Methods
The ADPHC, in collaboration with G42 Biogenix Labs, conducted a saliva SARS-CoV-2 NAAT testing program in 18 primary schools in the Emirate. Schools were selected based on outbreak prevalence at the time and focused on “hot spot” areas. The school on-site saliva testing program included children aged 4 to 12 years old in a “bubble” attendance model during the school day. This model involved children being assigned to groups or “pods.” This allowed us to limit a potential outbreak to a single pod, as opposed to risk exposing the entire school, should a single student test positive. The well-established SalivaDirect protocol developed at Yale University was used for testing and included an RNA extraction-free, RT-qPCR method for SARS-CoV-2 detection.11
We conducted a qualitative study involving telephone interviews of school administrators to evaluate their experience with the ADPHC testing program at their schools. In addition, we interviewed the G42 Biogenix Lab providers to understand the logistics that supported on-campus collection of saliva specimens for this age group. We also gathered the attitudes of school children before and after testing. This study was reviewed and approved by the Abu Dhabi Health Research and Technology Committee and the Institutional Review Board (IRB), New York University Abu Dhabi (NYUAD).
Sample and recruitment
The original sample collection of saliva specimens was performed by the ADPHC in collaboration with G42 Biogenix Lab providers on school campuses between December 6 and December 10, 2020. During this time, schools operated in a hybrid teaching model, where learning took place both online and in person. Infection control measures were deployed based on ADPHC standards and guidelines. Nurses utilized appropriate patient protective equipment, frequent hand hygiene, and social distancing during the collection process. Inclusion criteria included asymptomatic students aged 4 to 12 years attending in-person classes on campus. Students with respiratory symptoms who were asked to stay home or those not attending in-person classes were excluded.
Data collection
Data with regard to school children’s attitudes before and after testing were compiled through an online survey sent randomly to participants postintervention. Data from school administrators were collected through video and telephone interviews between April 14 and April 29, 2021. We first interviewed G42 Biogenix Lab providers to obtain previously acquired qualitative and quantitative data, which were collected during the intervention itself. After obtaining this information, we designed a questionnaire and proceeded with a structured interview process for school officials.
We interviewed school principals and administrators to collect their overall experiences with the saliva testing program. Before starting each interview, we established the interviewees preferred language, either English or Arabic. We then introduced the meeting attendees and provided study details, aims, and objectives, and described collaborating entities. We obtained verbal informed consent from a script approved by the NYUAD IRB and then proceeded with the interview, which included 4 questions. The first 3 questions were answered on a 5-point Likert scale model that consisted of 5 answer options: 5 being completely agree, 4 agree, 3 somewhat agree, 2 somewhat disagree, and 1 completely disagree. The fourth question invited open-ended feedback and comments on the following statements:
- I believe the COVID-19 saliva testing program improved the safety for my school campus.
- Our community had an overall positive experience with the COVID saliva testing.
- We would like to continue a saliva-based COVID testing program on our school campus.
- Please provide any additional comments you feel important about the program.
During the interview, we transcribed the answers as the interviewee was answering. We then translated those in Arabic into English and collected the data in 1 Excel spreadsheet. School interviewees and school names were de-identified in the collection and storage process.
Results
A total of 2011 saliva samples were collected from 18 different primary school campuses. Samples were sent the same day to G42 Biogenix Labs in Abu Dhabi for COVID PCR testing. A team consisting of 5 doctors providing general oversight, along with 2 to 6 nurses per site, were able to manage the collection process for all 18 school campuses. Samples were collected between 8
Sample stations were set up in either the school auditorium or gymnasium to ensure appropriate crowd control and ventilation. Teachers and other school staff, including public safety, were able to manage lines and the shuttling of students back and forth from classes to testing stations, which allowed medical staff to focus on sample collection.
Informed consent was obtained by prior electronic communication to parents from school staff, asking them to agree to allow their child to participate in the testing program. Informed consent was identified as a challenge: Getting parents to understand that saliva testing was more comfortable than NP testing, and that the results were only being used to help keep the school safe, took time. School staff are used to obtaining consent from parents for field trips, but this was clearly more challenging for them.

The saliva collection process per child took more time than expected. Children fasted for 45 minutes before saliva collection. We used an active drool technique, which required children to pool saliva in their mouth then express it into a collection tube. Adults can generally do this on command, but we found it took 10 to 12 minutes per child. Saliva production was cued by asking the children to think about food, and by showing them pictures and TV commercials depicting food. Children 4 to 5 years old had more difficulty with the process despite active cueing, while those 6 to 12 years old had an easier time with the process. We collected data on a cohort of 80 children regarding their attitudes pre (Figure 1) and post collection (Figure 2). Children felt happier, less nervous, and less scared after collection than before collection. This trend reassured us that future collections would be easier for students.

A total of 15 of 18 school principals completed the telephone interview, yielding a response rate of 83%. Overall, 93% of the school principals agreed or completely agreed that the COVID-19 saliva testing program improved school safety; 93% agreed or completely agreed that they had an overall positive experience with the program; and 73% supported the ongoing use of saliva testing in their schools (Table 1). Administrators’ open-ended comments on their experience were positive overall (Table 2).

Discussion
By March 2020, many kindergarten to grade 12 public and private schools suspended in-person classes due to the pandemic and turned to online learning platforms. The negative impact of school closures on academic achievement is projected to be significant.7,12,13 Ensuring schools can stay open and run operations safely will require routine SARS-CoV-2 testing. Our study investigated the feasibility of routine saliva testing on children aged 4 to 12 years on their school campuses. The ADPHC school on-site saliva testing program involved bringing lab providers onto 18 primary school campuses and required cooperation among parents, students, school administrators, and health care professionals.

Children younger than 6 years had difficulty producing an adequate saliva specimen, whereas those 6 to 12 years did so with relative ease when cued by thoughts or pictures of food while waiting in line for collection. Schools considering on-site testing programs should consider the age range of 6 to 12 years as a viable age range for saliva screening. Children should fast for a minimum of 45 minutes prior to saliva collection and should be cued by thoughts of food, food pictures, or food commercials. Setting up a sampling station close to the cafeteria where students can smell meal preparation may also help.14,15 Sampling before breakfast or lunch, when children are potentially at their hungriest, should also be considered.
The greatest challenge was obtaining informed consent from parents who were not yet familiar with the reliability of saliva testing as a tool for SARS-CoV-2 screening or with the saliva collection process as a whole. Informed consent was initially done electronically, lacking direct human interaction to answer parents’ questions. Parents who refused had a follow-up call from the school nurse to further explain the logistics and rationale for saliva screening. Having medical professionals directly answer parents’ questions was helpful. Parents were reassured that the process was painless, confidential, and only to be used for school safety purposes. Despite school administrators being experienced in obtaining consent from parents for field trips, obtaining informed consent for a medical testing procedure is more complicated, and parents aren’t accustomed to providing such consent in a school environment. Schools considering on-site testing should ensure that their school nurse or other health care providers are on the front line obtaining informed consent and allaying parents’ fears.
School staff were able to effectively provide crowd control for testing, and children felt at ease being in a familiar environment. Teachers and public safety officers are well-equipped at managing the shuttling of students to class, to lunch, to physical education, and, finally, to dismissal. They were equally equipped at handling the logistics of students to and from testing, including minimizing crowds and helping students feel at ease during the process. This effective collaboration allowed the lab personnel to focus on sample collection and storage, while school staff managed all other aspects of the children’s safety and care.
Conclusion
Overall, school administrators had a positive experience with the testing program, felt the program improved the safety of their schools, and supported the ongoing use of saliva testing for SARS-CoV-2 on their school campuses. Children aged 6 years and older were able to provide adequate saliva samples, and children felt happier and less nervous after the process, indicating repeatability. Our findings highlight the feasibility of an integrated on-site saliva testing model for primary school campuses. Further research is needed to determine the scalability of such a model and whether the added compliance and safety of on-site testing compensates for the potential loss of learning time that testing during school hours would require.
Corresponding author: Ayaz Virji, MD, New York University Abu Dhabi, PO Box 129188, Abu Dhabi, United Arab Emirates; [email protected].
Financial disclosures: None.
1. Kuehn BM. Despite improvements, COVID-19’s health care disruptions persist. JAMA. 2021;325(23):2335. doi:10.1001/jama.2021.9134
2. National Institute on Aging. Why COVID-19 testing is the key to getting back to normal. September 4, 2020. Accessed September 8, 2021. https://www.nia.nih.gov/news/why-covid-19-testing-key-getting-back-normal
3. Centers for Disease Control and Prevention. Science brief: Transmission of SARS-CoV-2 in K-12 schools. Updated July 9, 2021. Accessed September 8, 2021. https://www.cdc.gov/coronavirus/2019-ncov/science/science-briefs/transmission_k_12_schools.html
4. Butler-Laporte G, Lawandi A, Schiller I, et al. Comparison of saliva and nasopharyngeal swab nucleic acid amplification testing for detection of SARS-CoV-2: a systematic review and meta-analysis. JAMA Intern Med. 2021;181(3):353-360. doi:10.1001/jamainternmed.2020.8876
5. Al Suwaidi H, Senok A, Varghese R, et al. Saliva for molecular detection of SARS-CoV-2 in school-age children. Clin Microbiol Infect. 2021;27(9):1330-1335. doi:10.1016/j.cmi.2021.02.009
6. Abu Dhabi. Accessed September 8, 2021. https://u.ae/en/about-the-uae/the-seven-emirates/abu-dhabi
7. Alsuwaidi AR, Al Hosani FI, Al Memari S, et al. Seroprevalence of COVID-19 infection in the Emirate of Abu Dhabi, United Arab Emirates: a population-based cross-sectional study. Int J Epidemiol. 2021;50(4):1077-1090. doi:10.1093/ije/dyab077
8. Al Hosany F, Ganesan S, Al Memari S, et al. Response to COVID-19 pandemic in the UAE: a public health perspective. J Glob Health. 2021;11:03050. doi:10.7189/jogh.11.03050
9. Bremmer I. The best global responses to the COVID-19 pandemic, 1 year later. Time Magazine. Updated February 23, 2021. Accessed September 8, 2021. https://time.com/5851633/best-global-responses-covid-19/
10. Department of Health, Abu Dhabi. Laboratory diagnostic test for COVID-19: update regarding saliva-based testing using RT-PCR test. 2021.
11. Vogels C, Brackney DE, Kalinich CC, et al. SalivaDirect: RNA extraction-free SARS-CoV-2 diagnostics. Protocols.io. Accessed September 8, 2021. https://www.protocols.io/view/salivadirect-rna-extraction-free-sars-cov-2-diagno-bh6jj9cn?version_warning=no
12. Education Endowment Foundation. Impact of school closures on the attainment gap: rapid evidence assessment. June 2020. Accessed September 8, 2021. https://www.researchgate.net/publication/342501263_EEF_2020_-_Impact_of_School_Closures_on_the_Attainment_Gap
13. United Nations. Policy brief: Education during COVID-19 and beyond. Accessed September 8, 2021. https://www.un.org/development/desa/dspd/wp-content/uploads/sites/22/2020/08/sg_policy_brief_covid-19_and_education_august_2020.pdf
14. Schiffman SS, Miletic ID. Effect of taste and smell on secretion rate of salivary IgA in elderly and young persons. J Nutr Health Aging. 1999;3(3):158-164.
15. Lee VM, Linden RW. The effect of odours on stimulated parotid salivary flow in humans. Physiol Behav. 1992;52(6):1121-1125. doi:10.1016/0031-9384(92)90470-m
1. Kuehn BM. Despite improvements, COVID-19’s health care disruptions persist. JAMA. 2021;325(23):2335. doi:10.1001/jama.2021.9134
2. National Institute on Aging. Why COVID-19 testing is the key to getting back to normal. September 4, 2020. Accessed September 8, 2021. https://www.nia.nih.gov/news/why-covid-19-testing-key-getting-back-normal
3. Centers for Disease Control and Prevention. Science brief: Transmission of SARS-CoV-2 in K-12 schools. Updated July 9, 2021. Accessed September 8, 2021. https://www.cdc.gov/coronavirus/2019-ncov/science/science-briefs/transmission_k_12_schools.html
4. Butler-Laporte G, Lawandi A, Schiller I, et al. Comparison of saliva and nasopharyngeal swab nucleic acid amplification testing for detection of SARS-CoV-2: a systematic review and meta-analysis. JAMA Intern Med. 2021;181(3):353-360. doi:10.1001/jamainternmed.2020.8876
5. Al Suwaidi H, Senok A, Varghese R, et al. Saliva for molecular detection of SARS-CoV-2 in school-age children. Clin Microbiol Infect. 2021;27(9):1330-1335. doi:10.1016/j.cmi.2021.02.009
6. Abu Dhabi. Accessed September 8, 2021. https://u.ae/en/about-the-uae/the-seven-emirates/abu-dhabi
7. Alsuwaidi AR, Al Hosani FI, Al Memari S, et al. Seroprevalence of COVID-19 infection in the Emirate of Abu Dhabi, United Arab Emirates: a population-based cross-sectional study. Int J Epidemiol. 2021;50(4):1077-1090. doi:10.1093/ije/dyab077
8. Al Hosany F, Ganesan S, Al Memari S, et al. Response to COVID-19 pandemic in the UAE: a public health perspective. J Glob Health. 2021;11:03050. doi:10.7189/jogh.11.03050
9. Bremmer I. The best global responses to the COVID-19 pandemic, 1 year later. Time Magazine. Updated February 23, 2021. Accessed September 8, 2021. https://time.com/5851633/best-global-responses-covid-19/
10. Department of Health, Abu Dhabi. Laboratory diagnostic test for COVID-19: update regarding saliva-based testing using RT-PCR test. 2021.
11. Vogels C, Brackney DE, Kalinich CC, et al. SalivaDirect: RNA extraction-free SARS-CoV-2 diagnostics. Protocols.io. Accessed September 8, 2021. https://www.protocols.io/view/salivadirect-rna-extraction-free-sars-cov-2-diagno-bh6jj9cn?version_warning=no
12. Education Endowment Foundation. Impact of school closures on the attainment gap: rapid evidence assessment. June 2020. Accessed September 8, 2021. https://www.researchgate.net/publication/342501263_EEF_2020_-_Impact_of_School_Closures_on_the_Attainment_Gap
13. United Nations. Policy brief: Education during COVID-19 and beyond. Accessed September 8, 2021. https://www.un.org/development/desa/dspd/wp-content/uploads/sites/22/2020/08/sg_policy_brief_covid-19_and_education_august_2020.pdf
14. Schiffman SS, Miletic ID. Effect of taste and smell on secretion rate of salivary IgA in elderly and young persons. J Nutr Health Aging. 1999;3(3):158-164.
15. Lee VM, Linden RW. The effect of odours on stimulated parotid salivary flow in humans. Physiol Behav. 1992;52(6):1121-1125. doi:10.1016/0031-9384(92)90470-m
What I Learned About Change From Practicing During the COVID-19 Surge
While sick at home with a 26-day symptomatic course of COVID-19 in March 2020, I watched the surge unfold in my state and the hospital where I work as an inpatient adult medicine physician. Although the preponderance of my professional life is dedicated to leading teams in implementing delivery system transformation, the hat I wore in that moment involved living through and keeping up with the changes around me. Once I recovered and returned to the arena as a COVID doctor, I adapted to and made changes during constant shifts in how we provided care.
Looking back on those months during the worst of the COVID-19 hospital surge in my region, I reflect on the factors that helped me, as a frontline and shift-work clinician, adapt to and make those changes. In reflecting on the elements that were meaningful to me during the crisis, I recognize a set of change-enabling factors that have broad relevance for those of us who work to improve outcomes for patients and populations.
Confidence engendered by liberating data
In the early days of the surge, there was much uncertainty, and unfortunately, some seriously imperfect messaging. Trust was broken or badly bruised for many frontline clinicians. I share this painful phase not to criticize, but rather reflect on what mattered to me during that crisis of confidence. It was data. Raw, unadjusted, best-available data. Produced and pushed out. Available, trended over time, telling the story of where we are, now. Counts of tests, beds, and ventilators. The consistent, transparent availability of relevant and straightforward data provided an active antidote to a sense of uncertainty during a crisis of confidence.
Personal practice change stimulated by relevance and urgency
For half a decade, I have been encouraging interdisciplinary inpatient teams to identify and actively engage the family and/or care partner as a member of the care team. Despite even the American Association of Retired Persons mobilizing an impressive regulatory approach in 32 states to require that family and/or care partners are included as such, the practice change efforts continued on a slow and steady path. Why? We just didn’t believe it was of urgent, relevant, mission-critical importance to our daily practice to do so. That all changed in March 2020.
Without needing to be told, educated, or incentivized, my first night as a COVID doctor found me calling every single patient’s family upon admission, regardless of what time it was. It was critical to review the diagnosis, transparently discuss the uncertainty regarding the upcoming hours and days, review the potential contingencies, and ask, right there and then, whether intubation is consistent with goals of care. It was that urgent and relevant. Without exception, families were grateful for the effort and candor.
The significance of this practice—undoubtedly adopted by every inpatient provider who has worked a COVID surge—is rooted in decades of academic deliberation on which is the “right” doctor to have these discussions. None of that mattered. Historical opinions changed due to what was urgent and relevant given the situation at hand and the job we had to do. Imagine, for example, what we could do and how we could change if we now consider it urgent and relevant to identify and mobilize enhanced services and supports to patients who experience inequities because we believe it to be mission-critical to the job we show up to do every day.
Change fostered by a creative problem-solving ecosystem
Embracing personal practice change was made easier and implicitly affirmed by the creative problem solving that occurred everywhere. Tents, drive-throughs, and even college field houses were now settings of care. Primary care physicians, cardiologists, and gastrointestinal (GI) and postanesthesia care nurses staffed the COVID floors. Rolling stands held iPads so staff could communicate with patients without entering the room. This creative ecosystem fostered individual practice change. No debates were needed to recognize that standard processes were inadequate. No single role or service of any discipline was singularly asked to change to meet the needs of the moment. Because of this ecosystem of creative, active change, there was a much greater flexibility among individuals, role types, departments, and disciplines to change. This is particularly poignant to me in light of the work I lead to improve care for patients who experience systemic inequities in our health care system. When we ask a single role type or discipline to change, it can be met with resistance; far more success is achieved when we engage an interdisciplinary and interdepartmental approach to change. When surrounded by others making change, it makes us more willing to change, too.
Change catalyzed teamwork
It is so often invoked that health care is a team sport. In practicality, while we may aspire to work as a team, health care delivery is still all too often comprised of a set of individual actors with individualized responsibilities trying to communicate the best they can with each other.
What I experienced during the surge at my hospital was the very best version of teamwork I have ever been a part of in health care: empathetic, mutually interdependent strangers coming together during daily changes in staffing, processes, and resources. I will never forget nights walking into the pediatric floor or day surgery recovery area—now repurposed as a COVID unit—to entirely new faces comprised of GI suite nurses, outpatient doctors, and moonlighting intensivists.
We were all new to each other, all new to working in this setting, and all new to whatever the newest changes of the day brought. I will never forget how we greeted each other and introduced ourselves. We asked each other where we were “from,” and held a genuine appreciation to each other for being there. Imagine how this impacted how we worked together. Looking back on those night shifts, I remember us as a truly interdependent team. I will endeavor to bring that sense of mutual regard and interdependency into my work to foster effective interdisciplinary and cross-continuum teamwork.
Takeaways
As a student and practitioner of delivery system transformation, I am often in conversations about imperfect data, incomplete evidence, and role-specific and organizational resistance to change. As an acute care provider during the COVID-19 hospital surge in my region, the experiences I had as a participant in the COVID-related delivery system change will stay with me as I lead value-based delivery system change. What worked in an infectious disease crisis holds great relevance to our pressing, urgent, relevant work to create a more person-centered, equitable, and value-based delivery system.
I am confident that if those of us seeking to improve outcomes use visible and accessible data to engender confidence, clearly link practice change to the relevant and urgent issue at hand, promote broadly visible creative problem solving to foster an ecosystem of change, and cultivate empathy and mutual interdependence to promote the teamwork we aspire to have, that we will foster meaningful progress in our efforts to improve care for patients and populations.
Corresponding author: Amy Boutwell, MD, MPP, President, Collaborative Healthcare Strategies, Lexington, MA; [email protected].
Financial disclosures: None.
While sick at home with a 26-day symptomatic course of COVID-19 in March 2020, I watched the surge unfold in my state and the hospital where I work as an inpatient adult medicine physician. Although the preponderance of my professional life is dedicated to leading teams in implementing delivery system transformation, the hat I wore in that moment involved living through and keeping up with the changes around me. Once I recovered and returned to the arena as a COVID doctor, I adapted to and made changes during constant shifts in how we provided care.
Looking back on those months during the worst of the COVID-19 hospital surge in my region, I reflect on the factors that helped me, as a frontline and shift-work clinician, adapt to and make those changes. In reflecting on the elements that were meaningful to me during the crisis, I recognize a set of change-enabling factors that have broad relevance for those of us who work to improve outcomes for patients and populations.
Confidence engendered by liberating data
In the early days of the surge, there was much uncertainty, and unfortunately, some seriously imperfect messaging. Trust was broken or badly bruised for many frontline clinicians. I share this painful phase not to criticize, but rather reflect on what mattered to me during that crisis of confidence. It was data. Raw, unadjusted, best-available data. Produced and pushed out. Available, trended over time, telling the story of where we are, now. Counts of tests, beds, and ventilators. The consistent, transparent availability of relevant and straightforward data provided an active antidote to a sense of uncertainty during a crisis of confidence.
Personal practice change stimulated by relevance and urgency
For half a decade, I have been encouraging interdisciplinary inpatient teams to identify and actively engage the family and/or care partner as a member of the care team. Despite even the American Association of Retired Persons mobilizing an impressive regulatory approach in 32 states to require that family and/or care partners are included as such, the practice change efforts continued on a slow and steady path. Why? We just didn’t believe it was of urgent, relevant, mission-critical importance to our daily practice to do so. That all changed in March 2020.
Without needing to be told, educated, or incentivized, my first night as a COVID doctor found me calling every single patient’s family upon admission, regardless of what time it was. It was critical to review the diagnosis, transparently discuss the uncertainty regarding the upcoming hours and days, review the potential contingencies, and ask, right there and then, whether intubation is consistent with goals of care. It was that urgent and relevant. Without exception, families were grateful for the effort and candor.
The significance of this practice—undoubtedly adopted by every inpatient provider who has worked a COVID surge—is rooted in decades of academic deliberation on which is the “right” doctor to have these discussions. None of that mattered. Historical opinions changed due to what was urgent and relevant given the situation at hand and the job we had to do. Imagine, for example, what we could do and how we could change if we now consider it urgent and relevant to identify and mobilize enhanced services and supports to patients who experience inequities because we believe it to be mission-critical to the job we show up to do every day.
Change fostered by a creative problem-solving ecosystem
Embracing personal practice change was made easier and implicitly affirmed by the creative problem solving that occurred everywhere. Tents, drive-throughs, and even college field houses were now settings of care. Primary care physicians, cardiologists, and gastrointestinal (GI) and postanesthesia care nurses staffed the COVID floors. Rolling stands held iPads so staff could communicate with patients without entering the room. This creative ecosystem fostered individual practice change. No debates were needed to recognize that standard processes were inadequate. No single role or service of any discipline was singularly asked to change to meet the needs of the moment. Because of this ecosystem of creative, active change, there was a much greater flexibility among individuals, role types, departments, and disciplines to change. This is particularly poignant to me in light of the work I lead to improve care for patients who experience systemic inequities in our health care system. When we ask a single role type or discipline to change, it can be met with resistance; far more success is achieved when we engage an interdisciplinary and interdepartmental approach to change. When surrounded by others making change, it makes us more willing to change, too.
Change catalyzed teamwork
It is so often invoked that health care is a team sport. In practicality, while we may aspire to work as a team, health care delivery is still all too often comprised of a set of individual actors with individualized responsibilities trying to communicate the best they can with each other.
What I experienced during the surge at my hospital was the very best version of teamwork I have ever been a part of in health care: empathetic, mutually interdependent strangers coming together during daily changes in staffing, processes, and resources. I will never forget nights walking into the pediatric floor or day surgery recovery area—now repurposed as a COVID unit—to entirely new faces comprised of GI suite nurses, outpatient doctors, and moonlighting intensivists.
We were all new to each other, all new to working in this setting, and all new to whatever the newest changes of the day brought. I will never forget how we greeted each other and introduced ourselves. We asked each other where we were “from,” and held a genuine appreciation to each other for being there. Imagine how this impacted how we worked together. Looking back on those night shifts, I remember us as a truly interdependent team. I will endeavor to bring that sense of mutual regard and interdependency into my work to foster effective interdisciplinary and cross-continuum teamwork.
Takeaways
As a student and practitioner of delivery system transformation, I am often in conversations about imperfect data, incomplete evidence, and role-specific and organizational resistance to change. As an acute care provider during the COVID-19 hospital surge in my region, the experiences I had as a participant in the COVID-related delivery system change will stay with me as I lead value-based delivery system change. What worked in an infectious disease crisis holds great relevance to our pressing, urgent, relevant work to create a more person-centered, equitable, and value-based delivery system.
I am confident that if those of us seeking to improve outcomes use visible and accessible data to engender confidence, clearly link practice change to the relevant and urgent issue at hand, promote broadly visible creative problem solving to foster an ecosystem of change, and cultivate empathy and mutual interdependence to promote the teamwork we aspire to have, that we will foster meaningful progress in our efforts to improve care for patients and populations.
Corresponding author: Amy Boutwell, MD, MPP, President, Collaborative Healthcare Strategies, Lexington, MA; [email protected].
Financial disclosures: None.
While sick at home with a 26-day symptomatic course of COVID-19 in March 2020, I watched the surge unfold in my state and the hospital where I work as an inpatient adult medicine physician. Although the preponderance of my professional life is dedicated to leading teams in implementing delivery system transformation, the hat I wore in that moment involved living through and keeping up with the changes around me. Once I recovered and returned to the arena as a COVID doctor, I adapted to and made changes during constant shifts in how we provided care.
Looking back on those months during the worst of the COVID-19 hospital surge in my region, I reflect on the factors that helped me, as a frontline and shift-work clinician, adapt to and make those changes. In reflecting on the elements that were meaningful to me during the crisis, I recognize a set of change-enabling factors that have broad relevance for those of us who work to improve outcomes for patients and populations.
Confidence engendered by liberating data
In the early days of the surge, there was much uncertainty, and unfortunately, some seriously imperfect messaging. Trust was broken or badly bruised for many frontline clinicians. I share this painful phase not to criticize, but rather reflect on what mattered to me during that crisis of confidence. It was data. Raw, unadjusted, best-available data. Produced and pushed out. Available, trended over time, telling the story of where we are, now. Counts of tests, beds, and ventilators. The consistent, transparent availability of relevant and straightforward data provided an active antidote to a sense of uncertainty during a crisis of confidence.
Personal practice change stimulated by relevance and urgency
For half a decade, I have been encouraging interdisciplinary inpatient teams to identify and actively engage the family and/or care partner as a member of the care team. Despite even the American Association of Retired Persons mobilizing an impressive regulatory approach in 32 states to require that family and/or care partners are included as such, the practice change efforts continued on a slow and steady path. Why? We just didn’t believe it was of urgent, relevant, mission-critical importance to our daily practice to do so. That all changed in March 2020.
Without needing to be told, educated, or incentivized, my first night as a COVID doctor found me calling every single patient’s family upon admission, regardless of what time it was. It was critical to review the diagnosis, transparently discuss the uncertainty regarding the upcoming hours and days, review the potential contingencies, and ask, right there and then, whether intubation is consistent with goals of care. It was that urgent and relevant. Without exception, families were grateful for the effort and candor.
The significance of this practice—undoubtedly adopted by every inpatient provider who has worked a COVID surge—is rooted in decades of academic deliberation on which is the “right” doctor to have these discussions. None of that mattered. Historical opinions changed due to what was urgent and relevant given the situation at hand and the job we had to do. Imagine, for example, what we could do and how we could change if we now consider it urgent and relevant to identify and mobilize enhanced services and supports to patients who experience inequities because we believe it to be mission-critical to the job we show up to do every day.
Change fostered by a creative problem-solving ecosystem
Embracing personal practice change was made easier and implicitly affirmed by the creative problem solving that occurred everywhere. Tents, drive-throughs, and even college field houses were now settings of care. Primary care physicians, cardiologists, and gastrointestinal (GI) and postanesthesia care nurses staffed the COVID floors. Rolling stands held iPads so staff could communicate with patients without entering the room. This creative ecosystem fostered individual practice change. No debates were needed to recognize that standard processes were inadequate. No single role or service of any discipline was singularly asked to change to meet the needs of the moment. Because of this ecosystem of creative, active change, there was a much greater flexibility among individuals, role types, departments, and disciplines to change. This is particularly poignant to me in light of the work I lead to improve care for patients who experience systemic inequities in our health care system. When we ask a single role type or discipline to change, it can be met with resistance; far more success is achieved when we engage an interdisciplinary and interdepartmental approach to change. When surrounded by others making change, it makes us more willing to change, too.
Change catalyzed teamwork
It is so often invoked that health care is a team sport. In practicality, while we may aspire to work as a team, health care delivery is still all too often comprised of a set of individual actors with individualized responsibilities trying to communicate the best they can with each other.
What I experienced during the surge at my hospital was the very best version of teamwork I have ever been a part of in health care: empathetic, mutually interdependent strangers coming together during daily changes in staffing, processes, and resources. I will never forget nights walking into the pediatric floor or day surgery recovery area—now repurposed as a COVID unit—to entirely new faces comprised of GI suite nurses, outpatient doctors, and moonlighting intensivists.
We were all new to each other, all new to working in this setting, and all new to whatever the newest changes of the day brought. I will never forget how we greeted each other and introduced ourselves. We asked each other where we were “from,” and held a genuine appreciation to each other for being there. Imagine how this impacted how we worked together. Looking back on those night shifts, I remember us as a truly interdependent team. I will endeavor to bring that sense of mutual regard and interdependency into my work to foster effective interdisciplinary and cross-continuum teamwork.
Takeaways
As a student and practitioner of delivery system transformation, I am often in conversations about imperfect data, incomplete evidence, and role-specific and organizational resistance to change. As an acute care provider during the COVID-19 hospital surge in my region, the experiences I had as a participant in the COVID-related delivery system change will stay with me as I lead value-based delivery system change. What worked in an infectious disease crisis holds great relevance to our pressing, urgent, relevant work to create a more person-centered, equitable, and value-based delivery system.
I am confident that if those of us seeking to improve outcomes use visible and accessible data to engender confidence, clearly link practice change to the relevant and urgent issue at hand, promote broadly visible creative problem solving to foster an ecosystem of change, and cultivate empathy and mutual interdependence to promote the teamwork we aspire to have, that we will foster meaningful progress in our efforts to improve care for patients and populations.
Corresponding author: Amy Boutwell, MD, MPP, President, Collaborative Healthcare Strategies, Lexington, MA; [email protected].
Financial disclosures: None.