User login
Review finds anti-staphylococcus treatments have little impact on eczema
published in Clinical and Experimental Allergy.
Eczema remains a huge disease burden worldwide, and colonization with S. aureus in eczema patients is common, but no standard intervention exists to relieve symptoms, wrote Nandini Banerjee, MD, of Addenbrooke’s Hospital, Cambridge, England. “While antibiotic treatment of clinically obvious infections such as cellulitis is beneficial, it is not clear whether antibiotic treatment of eczema influences eczema severity,” Dr. Banerjee noted.
The 41 studies included 1,753 participants and 10 treatment categories. Most of the studies were conducted in secondary care centers in Western Europe, North America, and the Far East. Twelve studies included children, four included only adults, 19 included children and adults, and in six studies, the participant age range was unclear. Among the studies with reported ages, the mean age ranged from 1.1 to 34.6 years. Eczema severity ranged from mild to severe, and treatment durations ranged from 10 minutes to 3 months.
The review presented comparisons of topical steroid/antibiotic combinations, oral antibiotics, and bleach baths. In 14 studies that compared topical steroid/antibiotic combinations to topical steroids alone, patients showed slightly greater global improvement in symptoms with the combination, but the impact on quality of life was not significantly different. Severe adverse events, including flare of dermatitis, worsening of eczema, and folliculitis, were reported by the patients who received the combination and the topical steroid–only patients. One study reported similar rates of antibiotic resistance in children treated with steroid only and with an antibiotic/steroid combination at 3 months’ follow-up.
In four studies, oral antibiotics “may make no difference in terms of good or excellent global improvement in infants and children at 14 to 28 days follow-up compared to placebo,” according to the review. The reviewers said that there was likely little or no difference in quality of life for infants and children given oral antibiotics, although they noted the low quality of evidence on this topic.
Five studies evaluated the impact of bleach baths on eczema patients with and without S. aureus infections. These studies showed no difference in global improvement measures compared with placebo and little or no difference in quality of life. Also, patients who underwent bleach baths compared with placebo patients reported similar adverse events of burning/stinging or dry skin at 2 months’ follow-up.
“Low-quality evidence, due to risk of bias, imprecise effect estimates, and heterogeneity, made pooling of results difficult,” Dr. Banerjee wrote. “Topical steroid/antibiotic combinations may be associated with possible small improvements in good or excellent signs/symptoms compared with topical steroid alone. High-quality trials evaluating efficacy, QOL, and antibiotic resistance are required,” she concluded.
In a commentary section after the review, Dr. Banerjee and colleagues noted that the United Kingdom’s NICE guidelines for managing atopic eczema in children younger than 12 years of age, published in March 2021, include evidence from the current updated Cochrane Review. The NICE guidelines emphasize that “in people who are not systemically unwell, clinicians should not routinely offer either a topical or oral antibiotic for secondary bacterial infection of eczema,” the Cochrane authors said. They added in their commentary that the use of antibiotics in cases of nonsevere infections can worsen eczema. Also, “the risk of antimicrobial resistance is high with topical antibiotics, and therefore extended doses of the same antibiotics should be avoided to prevent resistance,” they said. However, the authors acknowledged a role for antibiotics in certain situations. “In patients with systemic signs of infection such as cellulitis, systemic antibiotics have an important role in helping clear infection,” they noted.
Reasons for varying disease severity elude research
The current study is important because of the abundance of preclinical and clinical data that implicate S. aureus in atopic dermatitis pathogenesis, Brian Kim, MD, of the Icahn School of Medicine at Mount Sinai, New York, said in an interview.
Dr. Kim said that he was surprised by some of the study findings but not others. “On the one hand, I thought there would be data supporting antimicrobial therapy, albeit not strong support,” he said. “However, AD is a very complex disease, and understanding what a disease modifier does to it is hard to capture across studies of various different designs,” he said.
“The data supporting antimicrobial therapy for S. aureus in AD is not as clear as our clinical impressions may indicate,” said Dr. Kim. “We need to understand the relationship better, perhaps in particular subsets of patients,” he emphasized. In addition, “We need a better understanding of why some people are colonized with S. aureus, yet with little effect on AD itself, while others experience severe exacerbation of disease,” said Dr. Kim. Therefore, a key research question for future studies is whether the exacerbation is caused by the particular strain of the bug, the host susceptibility, or both, he said.
The review received no outside funding. Dr. Banerjee and Dr. Kim have disclosed that they had no relevant financial relationships.
A version of this article first appeared on Medscape.com.
published in Clinical and Experimental Allergy.
Eczema remains a huge disease burden worldwide, and colonization with S. aureus in eczema patients is common, but no standard intervention exists to relieve symptoms, wrote Nandini Banerjee, MD, of Addenbrooke’s Hospital, Cambridge, England. “While antibiotic treatment of clinically obvious infections such as cellulitis is beneficial, it is not clear whether antibiotic treatment of eczema influences eczema severity,” Dr. Banerjee noted.
The 41 studies included 1,753 participants and 10 treatment categories. Most of the studies were conducted in secondary care centers in Western Europe, North America, and the Far East. Twelve studies included children, four included only adults, 19 included children and adults, and in six studies, the participant age range was unclear. Among the studies with reported ages, the mean age ranged from 1.1 to 34.6 years. Eczema severity ranged from mild to severe, and treatment durations ranged from 10 minutes to 3 months.
The review presented comparisons of topical steroid/antibiotic combinations, oral antibiotics, and bleach baths. In 14 studies that compared topical steroid/antibiotic combinations to topical steroids alone, patients showed slightly greater global improvement in symptoms with the combination, but the impact on quality of life was not significantly different. Severe adverse events, including flare of dermatitis, worsening of eczema, and folliculitis, were reported by the patients who received the combination and the topical steroid–only patients. One study reported similar rates of antibiotic resistance in children treated with steroid only and with an antibiotic/steroid combination at 3 months’ follow-up.
In four studies, oral antibiotics “may make no difference in terms of good or excellent global improvement in infants and children at 14 to 28 days follow-up compared to placebo,” according to the review. The reviewers said that there was likely little or no difference in quality of life for infants and children given oral antibiotics, although they noted the low quality of evidence on this topic.
Five studies evaluated the impact of bleach baths on eczema patients with and without S. aureus infections. These studies showed no difference in global improvement measures compared with placebo and little or no difference in quality of life. Also, patients who underwent bleach baths compared with placebo patients reported similar adverse events of burning/stinging or dry skin at 2 months’ follow-up.
“Low-quality evidence, due to risk of bias, imprecise effect estimates, and heterogeneity, made pooling of results difficult,” Dr. Banerjee wrote. “Topical steroid/antibiotic combinations may be associated with possible small improvements in good or excellent signs/symptoms compared with topical steroid alone. High-quality trials evaluating efficacy, QOL, and antibiotic resistance are required,” she concluded.
In a commentary section after the review, Dr. Banerjee and colleagues noted that the United Kingdom’s NICE guidelines for managing atopic eczema in children younger than 12 years of age, published in March 2021, include evidence from the current updated Cochrane Review. The NICE guidelines emphasize that “in people who are not systemically unwell, clinicians should not routinely offer either a topical or oral antibiotic for secondary bacterial infection of eczema,” the Cochrane authors said. They added in their commentary that the use of antibiotics in cases of nonsevere infections can worsen eczema. Also, “the risk of antimicrobial resistance is high with topical antibiotics, and therefore extended doses of the same antibiotics should be avoided to prevent resistance,” they said. However, the authors acknowledged a role for antibiotics in certain situations. “In patients with systemic signs of infection such as cellulitis, systemic antibiotics have an important role in helping clear infection,” they noted.
Reasons for varying disease severity elude research
The current study is important because of the abundance of preclinical and clinical data that implicate S. aureus in atopic dermatitis pathogenesis, Brian Kim, MD, of the Icahn School of Medicine at Mount Sinai, New York, said in an interview.
Dr. Kim said that he was surprised by some of the study findings but not others. “On the one hand, I thought there would be data supporting antimicrobial therapy, albeit not strong support,” he said. “However, AD is a very complex disease, and understanding what a disease modifier does to it is hard to capture across studies of various different designs,” he said.
“The data supporting antimicrobial therapy for S. aureus in AD is not as clear as our clinical impressions may indicate,” said Dr. Kim. “We need to understand the relationship better, perhaps in particular subsets of patients,” he emphasized. In addition, “We need a better understanding of why some people are colonized with S. aureus, yet with little effect on AD itself, while others experience severe exacerbation of disease,” said Dr. Kim. Therefore, a key research question for future studies is whether the exacerbation is caused by the particular strain of the bug, the host susceptibility, or both, he said.
The review received no outside funding. Dr. Banerjee and Dr. Kim have disclosed that they had no relevant financial relationships.
A version of this article first appeared on Medscape.com.
published in Clinical and Experimental Allergy.
Eczema remains a huge disease burden worldwide, and colonization with S. aureus in eczema patients is common, but no standard intervention exists to relieve symptoms, wrote Nandini Banerjee, MD, of Addenbrooke’s Hospital, Cambridge, England. “While antibiotic treatment of clinically obvious infections such as cellulitis is beneficial, it is not clear whether antibiotic treatment of eczema influences eczema severity,” Dr. Banerjee noted.
The 41 studies included 1,753 participants and 10 treatment categories. Most of the studies were conducted in secondary care centers in Western Europe, North America, and the Far East. Twelve studies included children, four included only adults, 19 included children and adults, and in six studies, the participant age range was unclear. Among the studies with reported ages, the mean age ranged from 1.1 to 34.6 years. Eczema severity ranged from mild to severe, and treatment durations ranged from 10 minutes to 3 months.
The review presented comparisons of topical steroid/antibiotic combinations, oral antibiotics, and bleach baths. In 14 studies that compared topical steroid/antibiotic combinations to topical steroids alone, patients showed slightly greater global improvement in symptoms with the combination, but the impact on quality of life was not significantly different. Severe adverse events, including flare of dermatitis, worsening of eczema, and folliculitis, were reported by the patients who received the combination and the topical steroid–only patients. One study reported similar rates of antibiotic resistance in children treated with steroid only and with an antibiotic/steroid combination at 3 months’ follow-up.
In four studies, oral antibiotics “may make no difference in terms of good or excellent global improvement in infants and children at 14 to 28 days follow-up compared to placebo,” according to the review. The reviewers said that there was likely little or no difference in quality of life for infants and children given oral antibiotics, although they noted the low quality of evidence on this topic.
Five studies evaluated the impact of bleach baths on eczema patients with and without S. aureus infections. These studies showed no difference in global improvement measures compared with placebo and little or no difference in quality of life. Also, patients who underwent bleach baths compared with placebo patients reported similar adverse events of burning/stinging or dry skin at 2 months’ follow-up.
“Low-quality evidence, due to risk of bias, imprecise effect estimates, and heterogeneity, made pooling of results difficult,” Dr. Banerjee wrote. “Topical steroid/antibiotic combinations may be associated with possible small improvements in good or excellent signs/symptoms compared with topical steroid alone. High-quality trials evaluating efficacy, QOL, and antibiotic resistance are required,” she concluded.
In a commentary section after the review, Dr. Banerjee and colleagues noted that the United Kingdom’s NICE guidelines for managing atopic eczema in children younger than 12 years of age, published in March 2021, include evidence from the current updated Cochrane Review. The NICE guidelines emphasize that “in people who are not systemically unwell, clinicians should not routinely offer either a topical or oral antibiotic for secondary bacterial infection of eczema,” the Cochrane authors said. They added in their commentary that the use of antibiotics in cases of nonsevere infections can worsen eczema. Also, “the risk of antimicrobial resistance is high with topical antibiotics, and therefore extended doses of the same antibiotics should be avoided to prevent resistance,” they said. However, the authors acknowledged a role for antibiotics in certain situations. “In patients with systemic signs of infection such as cellulitis, systemic antibiotics have an important role in helping clear infection,” they noted.
Reasons for varying disease severity elude research
The current study is important because of the abundance of preclinical and clinical data that implicate S. aureus in atopic dermatitis pathogenesis, Brian Kim, MD, of the Icahn School of Medicine at Mount Sinai, New York, said in an interview.
Dr. Kim said that he was surprised by some of the study findings but not others. “On the one hand, I thought there would be data supporting antimicrobial therapy, albeit not strong support,” he said. “However, AD is a very complex disease, and understanding what a disease modifier does to it is hard to capture across studies of various different designs,” he said.
“The data supporting antimicrobial therapy for S. aureus in AD is not as clear as our clinical impressions may indicate,” said Dr. Kim. “We need to understand the relationship better, perhaps in particular subsets of patients,” he emphasized. In addition, “We need a better understanding of why some people are colonized with S. aureus, yet with little effect on AD itself, while others experience severe exacerbation of disease,” said Dr. Kim. Therefore, a key research question for future studies is whether the exacerbation is caused by the particular strain of the bug, the host susceptibility, or both, he said.
The review received no outside funding. Dr. Banerjee and Dr. Kim have disclosed that they had no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CLINICAL AND EXPERIMENTAL ALLERGY
Seniors face higher risk of other medical conditions after COVID-19
The findings of the observational study, which were published in the BMJ, show the risk of a new condition being triggered by COVID is more than twice as high in seniors, compared with younger patients. Plus, the researchers observed an even higher risk among those who were hospitalized, with nearly half (46%) of patients having developed new conditions after the acute COVID-19 infection period.
Respiratory failure with shortness of breath was the most common postacute sequela, but a wide range of heart, kidney, lung, liver, cognitive, mental health, and other conditions were diagnosed at least 3 weeks after initial infection and persisted beyond 30 days.
This is one of the first studies to specifically describe the incidence and severity of new conditions triggered by COVID-19 infection in a general sample of older adults, said study author Ken Cohen MD, FACP, executive director of translational research at Optum Labs and national senior medical director at Optum Care.
“Much of what has been published on the postacute sequelae of COVID-19 has been predominantly from a younger population, and many of the patients had been hospitalized,” Dr. Cohen noted. “This was the first study to focus on a large population of seniors, most of whom did not require hospitalization.”
Dr. Cohen and colleagues reviewed the health insurance records of more than 133,000 Medicare beneficiaries aged 65 or older who were diagnosed with COVID-19 before April 2020. They also matched individuals by age, race, sex, hospitalization status, and other factors to comparison groups without COVID-19 (one from 2020 and one from 2019), and to a group diagnosed with other lower respiratory tract viral infections before the pandemic.
Risk of developing new conditions was higher in hospitalized
After acute COVID-19 infection, 32% of seniors sought medical care for at least one new medical condition in 2020, compared with 21% of uninfected people in the same year.
The most commonly observed conditions included:
- Respiratory failure (7.55% higher risk).
- Fatigue (5.66% higher risk).
- High blood pressure (4.43% higher risk).
- Memory problems (2.63% higher risk).
- Kidney injury (2.59% higher risk).
- Mental health diagnoses (2.5% higher risk).
- Blood-clotting disorders (1.47 % higher risk).
- Heart rhythm disorders (2.9% higher risk).
The risk of developing new conditions was even higher among those 23,486 who were hospitalized in 2020. Those individuals showed a 23.6% higher risk for developing at least one new condition, compared with uninfected seniors in the same year. Also, patients older than 75 had a higher risk for neurological disorders, including dementia, encephalopathy, and memory problems. The researchers also found that respiratory failure and kidney injury were significantly more likely to affect men and Black patients.
When those who had COVID were compared with the group with other lower respiratory viral infections before the pandemic, only the risks of respiratory failure (2.39% higher), dementia (0.71% higher), and fatigue (0.18% higher) were higher.
Primary care providers can learn from these data to better evaluate and manage their geriatric patients with COVID-19 infection, said Amit Shah, MD, a geriatrician with the Mayo Clinic in Phoenix, in an interview.
“We must assess older patients who have had COVID-19 for more than just improvement from the respiratory symptoms of COVID-19 in post-COVID follow-up visits,” he said. “Older individuals with frailty have vulnerability to subsequent complications from severe illnesses and it is common to see post-illness diagnoses, such as new diagnosis of delirium; dementia; or renal, respiratory, or cardiac issues that is precipitated by the original illness. This study confirms that this is likely the case with COVID-19 as well.
“Primary care physicians should be vigilant for these complications, including attention to the rehabilitation needs of older patients with longer-term postviral fatigue from COVID-19,” Dr. Shah added.
Data predates ‘Omicron wave’
It remains uncertain whether sequelae will differ with the Omicron variant, but the findings remain applicable, Dr. Cohen said.
“We know that illness from the Omicron variant is on average less severe in those that have been vaccinated. However, throughout the Omicron wave, individuals who have not been vaccinated continue to have significant rates of serious illness and hospitalization,” he said.
“Our findings showed that serious illness with hospitalization was associated with a higher rate of sequelae. It can therefore be inferred that the rates of sequelae seen in our study would continue to occur in unvaccinated individuals who contract Omicron, but might occur less frequently in vaccinated individuals who contract Omicron and have less severe illness.”
Dr. Cohen serves as a consultant for Pfizer. Dr. Shah has disclosed no relevant financial relationships.
The findings of the observational study, which were published in the BMJ, show the risk of a new condition being triggered by COVID is more than twice as high in seniors, compared with younger patients. Plus, the researchers observed an even higher risk among those who were hospitalized, with nearly half (46%) of patients having developed new conditions after the acute COVID-19 infection period.
Respiratory failure with shortness of breath was the most common postacute sequela, but a wide range of heart, kidney, lung, liver, cognitive, mental health, and other conditions were diagnosed at least 3 weeks after initial infection and persisted beyond 30 days.
This is one of the first studies to specifically describe the incidence and severity of new conditions triggered by COVID-19 infection in a general sample of older adults, said study author Ken Cohen MD, FACP, executive director of translational research at Optum Labs and national senior medical director at Optum Care.
“Much of what has been published on the postacute sequelae of COVID-19 has been predominantly from a younger population, and many of the patients had been hospitalized,” Dr. Cohen noted. “This was the first study to focus on a large population of seniors, most of whom did not require hospitalization.”
Dr. Cohen and colleagues reviewed the health insurance records of more than 133,000 Medicare beneficiaries aged 65 or older who were diagnosed with COVID-19 before April 2020. They also matched individuals by age, race, sex, hospitalization status, and other factors to comparison groups without COVID-19 (one from 2020 and one from 2019), and to a group diagnosed with other lower respiratory tract viral infections before the pandemic.
Risk of developing new conditions was higher in hospitalized
After acute COVID-19 infection, 32% of seniors sought medical care for at least one new medical condition in 2020, compared with 21% of uninfected people in the same year.
The most commonly observed conditions included:
- Respiratory failure (7.55% higher risk).
- Fatigue (5.66% higher risk).
- High blood pressure (4.43% higher risk).
- Memory problems (2.63% higher risk).
- Kidney injury (2.59% higher risk).
- Mental health diagnoses (2.5% higher risk).
- Blood-clotting disorders (1.47 % higher risk).
- Heart rhythm disorders (2.9% higher risk).
The risk of developing new conditions was even higher among those 23,486 who were hospitalized in 2020. Those individuals showed a 23.6% higher risk for developing at least one new condition, compared with uninfected seniors in the same year. Also, patients older than 75 had a higher risk for neurological disorders, including dementia, encephalopathy, and memory problems. The researchers also found that respiratory failure and kidney injury were significantly more likely to affect men and Black patients.
When those who had COVID were compared with the group with other lower respiratory viral infections before the pandemic, only the risks of respiratory failure (2.39% higher), dementia (0.71% higher), and fatigue (0.18% higher) were higher.
Primary care providers can learn from these data to better evaluate and manage their geriatric patients with COVID-19 infection, said Amit Shah, MD, a geriatrician with the Mayo Clinic in Phoenix, in an interview.
“We must assess older patients who have had COVID-19 for more than just improvement from the respiratory symptoms of COVID-19 in post-COVID follow-up visits,” he said. “Older individuals with frailty have vulnerability to subsequent complications from severe illnesses and it is common to see post-illness diagnoses, such as new diagnosis of delirium; dementia; or renal, respiratory, or cardiac issues that is precipitated by the original illness. This study confirms that this is likely the case with COVID-19 as well.
“Primary care physicians should be vigilant for these complications, including attention to the rehabilitation needs of older patients with longer-term postviral fatigue from COVID-19,” Dr. Shah added.
Data predates ‘Omicron wave’
It remains uncertain whether sequelae will differ with the Omicron variant, but the findings remain applicable, Dr. Cohen said.
“We know that illness from the Omicron variant is on average less severe in those that have been vaccinated. However, throughout the Omicron wave, individuals who have not been vaccinated continue to have significant rates of serious illness and hospitalization,” he said.
“Our findings showed that serious illness with hospitalization was associated with a higher rate of sequelae. It can therefore be inferred that the rates of sequelae seen in our study would continue to occur in unvaccinated individuals who contract Omicron, but might occur less frequently in vaccinated individuals who contract Omicron and have less severe illness.”
Dr. Cohen serves as a consultant for Pfizer. Dr. Shah has disclosed no relevant financial relationships.
The findings of the observational study, which were published in the BMJ, show the risk of a new condition being triggered by COVID is more than twice as high in seniors, compared with younger patients. Plus, the researchers observed an even higher risk among those who were hospitalized, with nearly half (46%) of patients having developed new conditions after the acute COVID-19 infection period.
Respiratory failure with shortness of breath was the most common postacute sequela, but a wide range of heart, kidney, lung, liver, cognitive, mental health, and other conditions were diagnosed at least 3 weeks after initial infection and persisted beyond 30 days.
This is one of the first studies to specifically describe the incidence and severity of new conditions triggered by COVID-19 infection in a general sample of older adults, said study author Ken Cohen MD, FACP, executive director of translational research at Optum Labs and national senior medical director at Optum Care.
“Much of what has been published on the postacute sequelae of COVID-19 has been predominantly from a younger population, and many of the patients had been hospitalized,” Dr. Cohen noted. “This was the first study to focus on a large population of seniors, most of whom did not require hospitalization.”
Dr. Cohen and colleagues reviewed the health insurance records of more than 133,000 Medicare beneficiaries aged 65 or older who were diagnosed with COVID-19 before April 2020. They also matched individuals by age, race, sex, hospitalization status, and other factors to comparison groups without COVID-19 (one from 2020 and one from 2019), and to a group diagnosed with other lower respiratory tract viral infections before the pandemic.
Risk of developing new conditions was higher in hospitalized
After acute COVID-19 infection, 32% of seniors sought medical care for at least one new medical condition in 2020, compared with 21% of uninfected people in the same year.
The most commonly observed conditions included:
- Respiratory failure (7.55% higher risk).
- Fatigue (5.66% higher risk).
- High blood pressure (4.43% higher risk).
- Memory problems (2.63% higher risk).
- Kidney injury (2.59% higher risk).
- Mental health diagnoses (2.5% higher risk).
- Blood-clotting disorders (1.47 % higher risk).
- Heart rhythm disorders (2.9% higher risk).
The risk of developing new conditions was even higher among those 23,486 who were hospitalized in 2020. Those individuals showed a 23.6% higher risk for developing at least one new condition, compared with uninfected seniors in the same year. Also, patients older than 75 had a higher risk for neurological disorders, including dementia, encephalopathy, and memory problems. The researchers also found that respiratory failure and kidney injury were significantly more likely to affect men and Black patients.
When those who had COVID were compared with the group with other lower respiratory viral infections before the pandemic, only the risks of respiratory failure (2.39% higher), dementia (0.71% higher), and fatigue (0.18% higher) were higher.
Primary care providers can learn from these data to better evaluate and manage their geriatric patients with COVID-19 infection, said Amit Shah, MD, a geriatrician with the Mayo Clinic in Phoenix, in an interview.
“We must assess older patients who have had COVID-19 for more than just improvement from the respiratory symptoms of COVID-19 in post-COVID follow-up visits,” he said. “Older individuals with frailty have vulnerability to subsequent complications from severe illnesses and it is common to see post-illness diagnoses, such as new diagnosis of delirium; dementia; or renal, respiratory, or cardiac issues that is precipitated by the original illness. This study confirms that this is likely the case with COVID-19 as well.
“Primary care physicians should be vigilant for these complications, including attention to the rehabilitation needs of older patients with longer-term postviral fatigue from COVID-19,” Dr. Shah added.
Data predates ‘Omicron wave’
It remains uncertain whether sequelae will differ with the Omicron variant, but the findings remain applicable, Dr. Cohen said.
“We know that illness from the Omicron variant is on average less severe in those that have been vaccinated. However, throughout the Omicron wave, individuals who have not been vaccinated continue to have significant rates of serious illness and hospitalization,” he said.
“Our findings showed that serious illness with hospitalization was associated with a higher rate of sequelae. It can therefore be inferred that the rates of sequelae seen in our study would continue to occur in unvaccinated individuals who contract Omicron, but might occur less frequently in vaccinated individuals who contract Omicron and have less severe illness.”
Dr. Cohen serves as a consultant for Pfizer. Dr. Shah has disclosed no relevant financial relationships.
FROM BMJ
C. difficile: New vancomycin-resistant strains raise concerns
Samples from patients in the United States and Kenya show an increasing emergence of previously undetected vancomycin-resistant strains of Clostridioides difficile, sparking concern as recurrences in the treatment of C. difficile infection (CDI) continue to rise.
“Our results may help explain a decreasing effectiveness of antibiotic-based therapy in C. difficile infection, since a significant proportion of patients harboring strains with reduced susceptibility to vancomycin may not respond to treatment,” reported the authors in research published recently in Clinical Infectious Diseases.
The spread of the resistant strains “has serious public health implications, underscoring an urgent need for a comprehensive analysis of the circulating strains to help inform clinical decisions,” they added.
Commenting on the findings, Cornelius J. Clancy, MD, professor of medicine at the University of Pittsburgh, and chief of infectious diseases at the Veterans Affairs Pittsburgh Healthcare System, echoed the concern.
“The casual belief has been that [C. difficile] strains at most centers can be assumed to be vancomycin susceptible,” he told this news organization. “This study shows that this assumption can no longer be taken as a given.”
Dr. Clancy, who was not involved with this research, noted that “based on this study, there might be need for the Infectious Diseases Society of America and other organizations to offer guidance on generating good, quality surveillance data for C. difficile resistance.”
With C. difficile showing the ability to resist multiple antibiotics, drugs in the armamentarium to treat the infection have declined in recent years, and recurrences with the infection are reported in up to 25% of cases.
Oral vancomycin is recommended as the antibiotic of choice by the IDSA and the Society for Healthcare Epidemiology of America for severe as well as nonsevere cases of CDI, and although there are reports of nine vancomycin-resistant gene clusters, most involve Enterococcus.
To take a closer look at the prevalence of vancomycin-resistant C. difficile strains, first author Charles Darkoh, PhD, with the Center for Infectious Diseases at the University of Texas Health Science Center, Houston, and colleagues analyzed stool samples from patients with CDI, including 438 patients in Houston, taken between 2012 and 2017, and 98 in Nairobi, Kenya, taken in 2017.
They found that, among samples from patients in Houston, over the time period, 26% showed vancomycin nonsusceptible C. difficile isolates and 29% had isolates that were metronidazole resistant.
And among samples from the Nairobi patients, 67% harbored vancomycin-resistant isolates and 85% had isolates resistant to metronidazole.
Of note, the proportion of samples containing vancomycin-resistant C. difficile in the Houston patients showed a marked increase over time, from «complete absence» in 2012 to approximately 35% in 2017, the authors reported.
“These nonsusceptibility rates significantly exceeded prior reports from other studies conducted in the United States and Europe from 2011 to 2014, suggesting a lower percentage of resistance to both metronidazole and vancomycin,” the authors wrote.
Further experiments on mouse models infected with one of the vancomycin-resistant isolates showed that treatment with vancomycin failed to eradicate the infection, and 5-day survival was significantly lower after vancomycin treatment in those mice (25%) versus those infected with strains known to be vancomycin sensitive (50%).
Unrecognized genetic strains
Whole-genome sequencing of 10 of the resistant isolates showed no matches with gene clusters that have been previously recognized as being vancomycin resistant, suggesting the emergence of new clusters.
“Together, these results suggest unknown genetic elements associated with vancomycin nonsusceptibility in isolates circulating in the patient population,” the authors wrote.
Dr. Darkoh told this news organization that the research team is currently working to further investigate the patterns and mechanisms.
“We are currently working on a follow-up study for the next 5 years to find out how widespread this is,” he said. “We want to make sure it’s not necessarily just occurring in the settings we studied, and we also need to establish the mechanism of resistance.”
Further commenting on the results, Dr. Clancy noted that “the extent of resistance caught many in the field a bit off guard, as they are higher than previously reported.”
“The data are also concerning because most centers do not routinely test C. difficile for drug susceptibility.”
Dr. Clancy noted that “another immediately pressing need is to understand mechanisms of resistance. It was quite striking that vancomycin-resistant strains in this study did not carry vanA genes, pointing to previously unrecognized mechanisms of resistance.”
“As is often the case, antibiotic overuse was likely a factor in the resistances, with overtesting often leading to overtreatment of C. difficile,” Dr. Clancy said. “The situation may have been compounded by failure to appreciate how entrenched C. difficile resistance may be at certain hospitals, since widespread susceptibility testing is generally not routinely performed.”
As alternative treatments, Dr. Clancy pointed to the recent IDSA update, which included a stronger endorsement of fidaxomicin.
“Of course, there is also the need to assure that data on resistance to agents like fidaxomicin are generated going forward,” he noted.
The study was supported by was supported by National Institutes of Health, the National Institute of Allergy and Infectious Diseases, the Texas Medical Center Digestive Diseases Center, and the University of Texas Health Science Center. Dr. Darkoh has disclosed no relevant financial relationships. One coauthor received grant support from Merck, Entasis Pharmaceuticals, and MeMed Diagnostics. Dr. Clancy disclosed advisory board, consulting and/or research relationships with Merck, Qpex Biopharma, Shionogi, Astellas, Cidara, Scynexis, and Needham & Associates.
Help your patients understand their C. difficile diagnosis by sharing patient education from the AGA GI Patient Center: www.gastro.org/Cdiff.
A version of this article first appeared on Medscape.com.
Samples from patients in the United States and Kenya show an increasing emergence of previously undetected vancomycin-resistant strains of Clostridioides difficile, sparking concern as recurrences in the treatment of C. difficile infection (CDI) continue to rise.
“Our results may help explain a decreasing effectiveness of antibiotic-based therapy in C. difficile infection, since a significant proportion of patients harboring strains with reduced susceptibility to vancomycin may not respond to treatment,” reported the authors in research published recently in Clinical Infectious Diseases.
The spread of the resistant strains “has serious public health implications, underscoring an urgent need for a comprehensive analysis of the circulating strains to help inform clinical decisions,” they added.
Commenting on the findings, Cornelius J. Clancy, MD, professor of medicine at the University of Pittsburgh, and chief of infectious diseases at the Veterans Affairs Pittsburgh Healthcare System, echoed the concern.
“The casual belief has been that [C. difficile] strains at most centers can be assumed to be vancomycin susceptible,” he told this news organization. “This study shows that this assumption can no longer be taken as a given.”
Dr. Clancy, who was not involved with this research, noted that “based on this study, there might be need for the Infectious Diseases Society of America and other organizations to offer guidance on generating good, quality surveillance data for C. difficile resistance.”
With C. difficile showing the ability to resist multiple antibiotics, drugs in the armamentarium to treat the infection have declined in recent years, and recurrences with the infection are reported in up to 25% of cases.
Oral vancomycin is recommended as the antibiotic of choice by the IDSA and the Society for Healthcare Epidemiology of America for severe as well as nonsevere cases of CDI, and although there are reports of nine vancomycin-resistant gene clusters, most involve Enterococcus.
To take a closer look at the prevalence of vancomycin-resistant C. difficile strains, first author Charles Darkoh, PhD, with the Center for Infectious Diseases at the University of Texas Health Science Center, Houston, and colleagues analyzed stool samples from patients with CDI, including 438 patients in Houston, taken between 2012 and 2017, and 98 in Nairobi, Kenya, taken in 2017.
They found that, among samples from patients in Houston, over the time period, 26% showed vancomycin nonsusceptible C. difficile isolates and 29% had isolates that were metronidazole resistant.
And among samples from the Nairobi patients, 67% harbored vancomycin-resistant isolates and 85% had isolates resistant to metronidazole.
Of note, the proportion of samples containing vancomycin-resistant C. difficile in the Houston patients showed a marked increase over time, from «complete absence» in 2012 to approximately 35% in 2017, the authors reported.
“These nonsusceptibility rates significantly exceeded prior reports from other studies conducted in the United States and Europe from 2011 to 2014, suggesting a lower percentage of resistance to both metronidazole and vancomycin,” the authors wrote.
Further experiments on mouse models infected with one of the vancomycin-resistant isolates showed that treatment with vancomycin failed to eradicate the infection, and 5-day survival was significantly lower after vancomycin treatment in those mice (25%) versus those infected with strains known to be vancomycin sensitive (50%).
Unrecognized genetic strains
Whole-genome sequencing of 10 of the resistant isolates showed no matches with gene clusters that have been previously recognized as being vancomycin resistant, suggesting the emergence of new clusters.
“Together, these results suggest unknown genetic elements associated with vancomycin nonsusceptibility in isolates circulating in the patient population,” the authors wrote.
Dr. Darkoh told this news organization that the research team is currently working to further investigate the patterns and mechanisms.
“We are currently working on a follow-up study for the next 5 years to find out how widespread this is,” he said. “We want to make sure it’s not necessarily just occurring in the settings we studied, and we also need to establish the mechanism of resistance.”
Further commenting on the results, Dr. Clancy noted that “the extent of resistance caught many in the field a bit off guard, as they are higher than previously reported.”
“The data are also concerning because most centers do not routinely test C. difficile for drug susceptibility.”
Dr. Clancy noted that “another immediately pressing need is to understand mechanisms of resistance. It was quite striking that vancomycin-resistant strains in this study did not carry vanA genes, pointing to previously unrecognized mechanisms of resistance.”
“As is often the case, antibiotic overuse was likely a factor in the resistances, with overtesting often leading to overtreatment of C. difficile,” Dr. Clancy said. “The situation may have been compounded by failure to appreciate how entrenched C. difficile resistance may be at certain hospitals, since widespread susceptibility testing is generally not routinely performed.”
As alternative treatments, Dr. Clancy pointed to the recent IDSA update, which included a stronger endorsement of fidaxomicin.
“Of course, there is also the need to assure that data on resistance to agents like fidaxomicin are generated going forward,” he noted.
The study was supported by was supported by National Institutes of Health, the National Institute of Allergy and Infectious Diseases, the Texas Medical Center Digestive Diseases Center, and the University of Texas Health Science Center. Dr. Darkoh has disclosed no relevant financial relationships. One coauthor received grant support from Merck, Entasis Pharmaceuticals, and MeMed Diagnostics. Dr. Clancy disclosed advisory board, consulting and/or research relationships with Merck, Qpex Biopharma, Shionogi, Astellas, Cidara, Scynexis, and Needham & Associates.
Help your patients understand their C. difficile diagnosis by sharing patient education from the AGA GI Patient Center: www.gastro.org/Cdiff.
A version of this article first appeared on Medscape.com.
Samples from patients in the United States and Kenya show an increasing emergence of previously undetected vancomycin-resistant strains of Clostridioides difficile, sparking concern as recurrences in the treatment of C. difficile infection (CDI) continue to rise.
“Our results may help explain a decreasing effectiveness of antibiotic-based therapy in C. difficile infection, since a significant proportion of patients harboring strains with reduced susceptibility to vancomycin may not respond to treatment,” reported the authors in research published recently in Clinical Infectious Diseases.
The spread of the resistant strains “has serious public health implications, underscoring an urgent need for a comprehensive analysis of the circulating strains to help inform clinical decisions,” they added.
Commenting on the findings, Cornelius J. Clancy, MD, professor of medicine at the University of Pittsburgh, and chief of infectious diseases at the Veterans Affairs Pittsburgh Healthcare System, echoed the concern.
“The casual belief has been that [C. difficile] strains at most centers can be assumed to be vancomycin susceptible,” he told this news organization. “This study shows that this assumption can no longer be taken as a given.”
Dr. Clancy, who was not involved with this research, noted that “based on this study, there might be need for the Infectious Diseases Society of America and other organizations to offer guidance on generating good, quality surveillance data for C. difficile resistance.”
With C. difficile showing the ability to resist multiple antibiotics, drugs in the armamentarium to treat the infection have declined in recent years, and recurrences with the infection are reported in up to 25% of cases.
Oral vancomycin is recommended as the antibiotic of choice by the IDSA and the Society for Healthcare Epidemiology of America for severe as well as nonsevere cases of CDI, and although there are reports of nine vancomycin-resistant gene clusters, most involve Enterococcus.
To take a closer look at the prevalence of vancomycin-resistant C. difficile strains, first author Charles Darkoh, PhD, with the Center for Infectious Diseases at the University of Texas Health Science Center, Houston, and colleagues analyzed stool samples from patients with CDI, including 438 patients in Houston, taken between 2012 and 2017, and 98 in Nairobi, Kenya, taken in 2017.
They found that, among samples from patients in Houston, over the time period, 26% showed vancomycin nonsusceptible C. difficile isolates and 29% had isolates that were metronidazole resistant.
And among samples from the Nairobi patients, 67% harbored vancomycin-resistant isolates and 85% had isolates resistant to metronidazole.
Of note, the proportion of samples containing vancomycin-resistant C. difficile in the Houston patients showed a marked increase over time, from «complete absence» in 2012 to approximately 35% in 2017, the authors reported.
“These nonsusceptibility rates significantly exceeded prior reports from other studies conducted in the United States and Europe from 2011 to 2014, suggesting a lower percentage of resistance to both metronidazole and vancomycin,” the authors wrote.
Further experiments on mouse models infected with one of the vancomycin-resistant isolates showed that treatment with vancomycin failed to eradicate the infection, and 5-day survival was significantly lower after vancomycin treatment in those mice (25%) versus those infected with strains known to be vancomycin sensitive (50%).
Unrecognized genetic strains
Whole-genome sequencing of 10 of the resistant isolates showed no matches with gene clusters that have been previously recognized as being vancomycin resistant, suggesting the emergence of new clusters.
“Together, these results suggest unknown genetic elements associated with vancomycin nonsusceptibility in isolates circulating in the patient population,” the authors wrote.
Dr. Darkoh told this news organization that the research team is currently working to further investigate the patterns and mechanisms.
“We are currently working on a follow-up study for the next 5 years to find out how widespread this is,” he said. “We want to make sure it’s not necessarily just occurring in the settings we studied, and we also need to establish the mechanism of resistance.”
Further commenting on the results, Dr. Clancy noted that “the extent of resistance caught many in the field a bit off guard, as they are higher than previously reported.”
“The data are also concerning because most centers do not routinely test C. difficile for drug susceptibility.”
Dr. Clancy noted that “another immediately pressing need is to understand mechanisms of resistance. It was quite striking that vancomycin-resistant strains in this study did not carry vanA genes, pointing to previously unrecognized mechanisms of resistance.”
“As is often the case, antibiotic overuse was likely a factor in the resistances, with overtesting often leading to overtreatment of C. difficile,” Dr. Clancy said. “The situation may have been compounded by failure to appreciate how entrenched C. difficile resistance may be at certain hospitals, since widespread susceptibility testing is generally not routinely performed.”
As alternative treatments, Dr. Clancy pointed to the recent IDSA update, which included a stronger endorsement of fidaxomicin.
“Of course, there is also the need to assure that data on resistance to agents like fidaxomicin are generated going forward,” he noted.
The study was supported by was supported by National Institutes of Health, the National Institute of Allergy and Infectious Diseases, the Texas Medical Center Digestive Diseases Center, and the University of Texas Health Science Center. Dr. Darkoh has disclosed no relevant financial relationships. One coauthor received grant support from Merck, Entasis Pharmaceuticals, and MeMed Diagnostics. Dr. Clancy disclosed advisory board, consulting and/or research relationships with Merck, Qpex Biopharma, Shionogi, Astellas, Cidara, Scynexis, and Needham & Associates.
Help your patients understand their C. difficile diagnosis by sharing patient education from the AGA GI Patient Center: www.gastro.org/Cdiff.
A version of this article first appeared on Medscape.com.
FROM CLINICAL INFECTIOUS DISEASES
Children and COVID: New cases down again, but still ‘extremely high’
The indication of an Omicron decline has become a trend: New cases of COVID-19 in children were down for a second consecutive week in the United States, according to the American Academy of Pediatrics and the Children’s Hospital Association.
but the nearly 632,000 cases reported were down by 22% from the previous week and by 45% from what appears to be the peak of the Omicron surge during the week of Jan. 14-20, the AAP/CHA data show.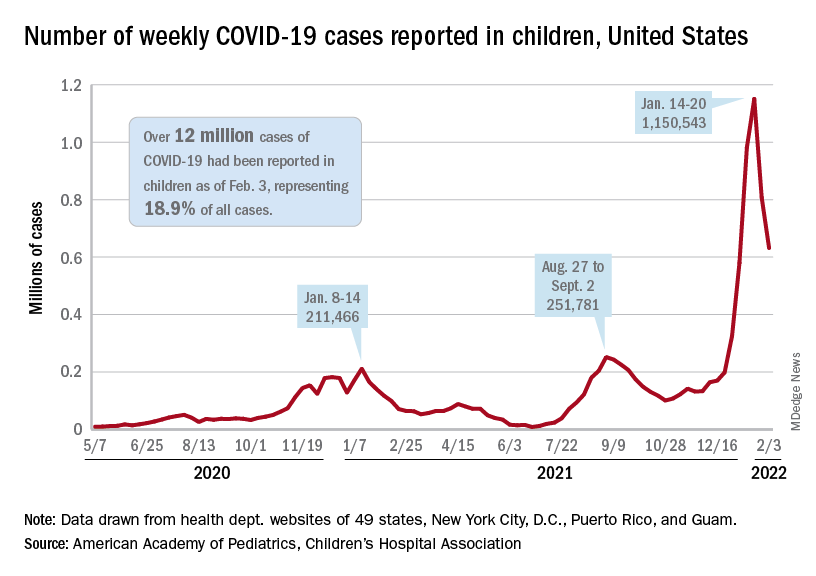
To put the effect of the Delta and Omicron variants into some sort of perspective, the total number of COVID-19 cases among children passed 5 million at the beginning of September 2021, about a year and a half into the pandemic. In the last 5 months, the cumulative count has more than doubled and now stands at 12 million, the AAP and CHA said in their weekly COVID report.
Hospital admissions and emergency department visits followed the same downward trend over the last week. The rate of new hospitalizations fell to 0.81 per 100,000 children aged 0-17 years as of Feb. 2 (down from a peak of 1.25 per 100,000 on Jan. 15), and ED visits with diagnosed COVID-19 dropped to 1.8% (peak was 14.1%), 1.9% (peak was 14.3%), and 3.4% (peak was 14%) of all visits for children aged 16-17, 12-15, and 0-11 years, respectively, the Centers for Disease Control and Prevention reported.
The vaccination response
The surge of infections brought about by the Omicron variant, however, did not translate into increased vaccination, at least for the youngest eligible children. Vaccine initiation rose slightly among children aged 5-11 in early and mid-January but, by early February, new vaccinations had declined to their lowest point since approval in early November of 2021, the AAP said in its weekly COVID vaccination report.
As a result, the 5- to 11-year-olds are well behind the pace set by those aged 12-15 for the first 3 months of their vaccination experience. Through the first 13 weeks after the COVID vaccine was approved for children aged 12-15 in early May, 44.5% had received at least one dose and 32.3% were fully vaccinated. Among children aged 5-11, the corresponding figures through 13 weeks were 31% and 22.5%, according to CDC data.
The vaccination reaction to Omicron was somewhat more robust for children aged 12-17, compared with the younger group, but initiations dropped at the same time that new cases began to decline. In terms of total volume, the response among 12- to 17-year-olds was much smaller than that seen in July and August of 2021 as the Delta surge was hitting the United States, the AAP vaccination report shows.
All those vaccinations add up to this: Over 16.8 million children aged 12-17 and almost 9 million aged 5-11 had received at least one dose of vaccine as of Feb. 7, which works out to 66.6% of the older group and 31.2% of the younger cohort. Almost 14.3 million (56.4%) of those aged 12-17 are fully vaccinated, as are 6.6 million (22.9%) of the 5- to 11-year-olds, the CDC said on its COVID Data Tracker.
The indication of an Omicron decline has become a trend: New cases of COVID-19 in children were down for a second consecutive week in the United States, according to the American Academy of Pediatrics and the Children’s Hospital Association.
but the nearly 632,000 cases reported were down by 22% from the previous week and by 45% from what appears to be the peak of the Omicron surge during the week of Jan. 14-20, the AAP/CHA data show.
To put the effect of the Delta and Omicron variants into some sort of perspective, the total number of COVID-19 cases among children passed 5 million at the beginning of September 2021, about a year and a half into the pandemic. In the last 5 months, the cumulative count has more than doubled and now stands at 12 million, the AAP and CHA said in their weekly COVID report.
Hospital admissions and emergency department visits followed the same downward trend over the last week. The rate of new hospitalizations fell to 0.81 per 100,000 children aged 0-17 years as of Feb. 2 (down from a peak of 1.25 per 100,000 on Jan. 15), and ED visits with diagnosed COVID-19 dropped to 1.8% (peak was 14.1%), 1.9% (peak was 14.3%), and 3.4% (peak was 14%) of all visits for children aged 16-17, 12-15, and 0-11 years, respectively, the Centers for Disease Control and Prevention reported.
The vaccination response
The surge of infections brought about by the Omicron variant, however, did not translate into increased vaccination, at least for the youngest eligible children. Vaccine initiation rose slightly among children aged 5-11 in early and mid-January but, by early February, new vaccinations had declined to their lowest point since approval in early November of 2021, the AAP said in its weekly COVID vaccination report.
As a result, the 5- to 11-year-olds are well behind the pace set by those aged 12-15 for the first 3 months of their vaccination experience. Through the first 13 weeks after the COVID vaccine was approved for children aged 12-15 in early May, 44.5% had received at least one dose and 32.3% were fully vaccinated. Among children aged 5-11, the corresponding figures through 13 weeks were 31% and 22.5%, according to CDC data.
The vaccination reaction to Omicron was somewhat more robust for children aged 12-17, compared with the younger group, but initiations dropped at the same time that new cases began to decline. In terms of total volume, the response among 12- to 17-year-olds was much smaller than that seen in July and August of 2021 as the Delta surge was hitting the United States, the AAP vaccination report shows.
All those vaccinations add up to this: Over 16.8 million children aged 12-17 and almost 9 million aged 5-11 had received at least one dose of vaccine as of Feb. 7, which works out to 66.6% of the older group and 31.2% of the younger cohort. Almost 14.3 million (56.4%) of those aged 12-17 are fully vaccinated, as are 6.6 million (22.9%) of the 5- to 11-year-olds, the CDC said on its COVID Data Tracker.
The indication of an Omicron decline has become a trend: New cases of COVID-19 in children were down for a second consecutive week in the United States, according to the American Academy of Pediatrics and the Children’s Hospital Association.
but the nearly 632,000 cases reported were down by 22% from the previous week and by 45% from what appears to be the peak of the Omicron surge during the week of Jan. 14-20, the AAP/CHA data show.
To put the effect of the Delta and Omicron variants into some sort of perspective, the total number of COVID-19 cases among children passed 5 million at the beginning of September 2021, about a year and a half into the pandemic. In the last 5 months, the cumulative count has more than doubled and now stands at 12 million, the AAP and CHA said in their weekly COVID report.
Hospital admissions and emergency department visits followed the same downward trend over the last week. The rate of new hospitalizations fell to 0.81 per 100,000 children aged 0-17 years as of Feb. 2 (down from a peak of 1.25 per 100,000 on Jan. 15), and ED visits with diagnosed COVID-19 dropped to 1.8% (peak was 14.1%), 1.9% (peak was 14.3%), and 3.4% (peak was 14%) of all visits for children aged 16-17, 12-15, and 0-11 years, respectively, the Centers for Disease Control and Prevention reported.
The vaccination response
The surge of infections brought about by the Omicron variant, however, did not translate into increased vaccination, at least for the youngest eligible children. Vaccine initiation rose slightly among children aged 5-11 in early and mid-January but, by early February, new vaccinations had declined to their lowest point since approval in early November of 2021, the AAP said in its weekly COVID vaccination report.
As a result, the 5- to 11-year-olds are well behind the pace set by those aged 12-15 for the first 3 months of their vaccination experience. Through the first 13 weeks after the COVID vaccine was approved for children aged 12-15 in early May, 44.5% had received at least one dose and 32.3% were fully vaccinated. Among children aged 5-11, the corresponding figures through 13 weeks were 31% and 22.5%, according to CDC data.
The vaccination reaction to Omicron was somewhat more robust for children aged 12-17, compared with the younger group, but initiations dropped at the same time that new cases began to decline. In terms of total volume, the response among 12- to 17-year-olds was much smaller than that seen in July and August of 2021 as the Delta surge was hitting the United States, the AAP vaccination report shows.
All those vaccinations add up to this: Over 16.8 million children aged 12-17 and almost 9 million aged 5-11 had received at least one dose of vaccine as of Feb. 7, which works out to 66.6% of the older group and 31.2% of the younger cohort. Almost 14.3 million (56.4%) of those aged 12-17 are fully vaccinated, as are 6.6 million (22.9%) of the 5- to 11-year-olds, the CDC said on its COVID Data Tracker.
C. difficile: New vancomycin-resistant strains raise concerns
Samples from patients in the United States and Kenya show an increasing emergence of previously undetected vancomycin-resistant strains of Clostridioides difficile, sparking concern as recurrences in the treatment of C. difficile infection (CDI) continue to rise.
“Our results may help explain a decreasing effectiveness of antibiotic-based therapy in C. difficile infection, since a significant proportion of patients harboring strains with reduced susceptibility to vancomycin may not respond to treatment,” reported the authors in research published recently in Clinical Infectious Diseases.
The spread of the resistant strains “has serious public health implications, underscoring an urgent need for a comprehensive analysis of the circulating strains to help inform clinical decisions,” they added.
Commenting on the findings, Cornelius J. Clancy, MD, professor of medicine at the University of Pittsburgh, and chief of infectious diseases at the Veterans Affairs Pittsburgh Healthcare System, echoed the concern.
“The casual belief has been that [C. difficile] strains at most centers can be assumed to be vancomycin susceptible,” he told this news organization. “This study shows that this assumption can no longer be taken as a given.”
Dr. Clancy, who was not involved with this research, noted that “based on this study, there might be need for the Infectious Diseases Society of America and other organizations to offer guidance on generating good, quality surveillance data for C. difficile resistance.”
With C. difficile showing the ability to resist multiple antibiotics, drugs in the armamentarium to treat the infection have declined in recent years, and recurrences with the infection are reported in up to 25% of cases.
Oral vancomycin is recommended as the antibiotic of choice by the IDSA and the Society for Healthcare Epidemiology of America for severe as well as nonsevere cases of CDI, and although there are reports of nine vancomycin-resistant gene clusters, most involve Enterococcus.
To take a closer look at the prevalence of vancomycin-resistant C. difficile strains, first author Charles Darkoh, PhD, with the Center for Infectious Diseases at the University of Texas Health Science Center, Houston, and colleagues analyzed stool samples from patients with CDI, including 438 patients in Houston, taken between 2012 and 2017, and 98 in Nairobi, Kenya, taken in 2017.
They found that, among samples from patients in Houston, over the time period, 26% showed vancomycin nonsusceptible C. difficile isolates and 29% had isolates that were metronidazole resistant.
And among samples from the Nairobi patients, 67% harbored vancomycin-resistant isolates and 85% had isolates resistant to metronidazole.
Of note, the proportion of samples containing vancomycin-resistant C. difficile in the Houston patients showed a marked increase over time, from «complete absence» in 2012 to approximately 35% in 2017, the authors reported.
“These nonsusceptibility rates significantly exceeded prior reports from other studies conducted in the United States and Europe from 2011 to 2014, suggesting a lower percentage of resistance to both metronidazole and vancomycin,” the authors wrote.
Further experiments on mouse models infected with one of the vancomycin-resistant isolates showed that treatment with vancomycin failed to eradicate the infection, and 5-day survival was significantly lower after vancomycin treatment in those mice (25%) versus those infected with strains known to be vancomycin sensitive (50%).
Unrecognized genetic strains
Whole-genome sequencing of 10 of the resistant isolates showed no matches with gene clusters that have been previously recognized as being vancomycin resistant, suggesting the emergence of new clusters.
“Together, these results suggest unknown genetic elements associated with vancomycin nonsusceptibility in isolates circulating in the patient population,” the authors wrote.
Dr. Darkoh told this news organization that the research team is currently working to further investigate the patterns and mechanisms.
“We are currently working on a follow-up study for the next 5 years to find out how widespread this is,” he said. “We want to make sure it’s not necessarily just occurring in the settings we studied, and we also need to establish the mechanism of resistance.”
Further commenting on the results, Dr. Clancy noted that “the extent of resistance caught many in the field a bit off guard, as they are higher than previously reported.”
“The data are also concerning because most centers do not routinely test C. difficile for drug susceptibility.”
Dr. Clancy noted that “another immediately pressing need is to understand mechanisms of resistance. It was quite striking that vancomycin-resistant strains in this study did not carry vanA genes, pointing to previously unrecognized mechanisms of resistance.”
“As is often the case, antibiotic overuse was likely a factor in the resistances, with overtesting often leading to overtreatment of C. difficile,” Dr. Clancy said. “The situation may have been compounded by failure to appreciate how entrenched C. difficile resistance may be at certain hospitals, since widespread susceptibility testing is generally not routinely performed.”
As alternative treatments, Dr. Clancy pointed to the recent IDSA update, which included a stronger endorsement of fidaxomicin.
“Of course, there is also the need to assure that data on resistance to agents like fidaxomicin are generated going forward,” he noted.
The study was supported by was supported by National Institutes of Health, the National Institute of Allergy and Infectious Diseases, the Texas Medical Center Digestive Diseases Center, and the University of Texas Health Science Center. Dr. Darkoh has disclosed no relevant financial relationships. One coauthor received grant support from Merck, Entasis Pharmaceuticals, and MeMed Diagnostics. Dr. Clancy disclosed advisory board, consulting and/or research relationships with Merck, Qpex Biopharma, Shionogi, Astellas, Cidara, Scynexis, and Needham & Associates.
A version of this article first appeared on Medscape.com.
Samples from patients in the United States and Kenya show an increasing emergence of previously undetected vancomycin-resistant strains of Clostridioides difficile, sparking concern as recurrences in the treatment of C. difficile infection (CDI) continue to rise.
“Our results may help explain a decreasing effectiveness of antibiotic-based therapy in C. difficile infection, since a significant proportion of patients harboring strains with reduced susceptibility to vancomycin may not respond to treatment,” reported the authors in research published recently in Clinical Infectious Diseases.
The spread of the resistant strains “has serious public health implications, underscoring an urgent need for a comprehensive analysis of the circulating strains to help inform clinical decisions,” they added.
Commenting on the findings, Cornelius J. Clancy, MD, professor of medicine at the University of Pittsburgh, and chief of infectious diseases at the Veterans Affairs Pittsburgh Healthcare System, echoed the concern.
“The casual belief has been that [C. difficile] strains at most centers can be assumed to be vancomycin susceptible,” he told this news organization. “This study shows that this assumption can no longer be taken as a given.”
Dr. Clancy, who was not involved with this research, noted that “based on this study, there might be need for the Infectious Diseases Society of America and other organizations to offer guidance on generating good, quality surveillance data for C. difficile resistance.”
With C. difficile showing the ability to resist multiple antibiotics, drugs in the armamentarium to treat the infection have declined in recent years, and recurrences with the infection are reported in up to 25% of cases.
Oral vancomycin is recommended as the antibiotic of choice by the IDSA and the Society for Healthcare Epidemiology of America for severe as well as nonsevere cases of CDI, and although there are reports of nine vancomycin-resistant gene clusters, most involve Enterococcus.
To take a closer look at the prevalence of vancomycin-resistant C. difficile strains, first author Charles Darkoh, PhD, with the Center for Infectious Diseases at the University of Texas Health Science Center, Houston, and colleagues analyzed stool samples from patients with CDI, including 438 patients in Houston, taken between 2012 and 2017, and 98 in Nairobi, Kenya, taken in 2017.
They found that, among samples from patients in Houston, over the time period, 26% showed vancomycin nonsusceptible C. difficile isolates and 29% had isolates that were metronidazole resistant.
And among samples from the Nairobi patients, 67% harbored vancomycin-resistant isolates and 85% had isolates resistant to metronidazole.
Of note, the proportion of samples containing vancomycin-resistant C. difficile in the Houston patients showed a marked increase over time, from «complete absence» in 2012 to approximately 35% in 2017, the authors reported.
“These nonsusceptibility rates significantly exceeded prior reports from other studies conducted in the United States and Europe from 2011 to 2014, suggesting a lower percentage of resistance to both metronidazole and vancomycin,” the authors wrote.
Further experiments on mouse models infected with one of the vancomycin-resistant isolates showed that treatment with vancomycin failed to eradicate the infection, and 5-day survival was significantly lower after vancomycin treatment in those mice (25%) versus those infected with strains known to be vancomycin sensitive (50%).
Unrecognized genetic strains
Whole-genome sequencing of 10 of the resistant isolates showed no matches with gene clusters that have been previously recognized as being vancomycin resistant, suggesting the emergence of new clusters.
“Together, these results suggest unknown genetic elements associated with vancomycin nonsusceptibility in isolates circulating in the patient population,” the authors wrote.
Dr. Darkoh told this news organization that the research team is currently working to further investigate the patterns and mechanisms.
“We are currently working on a follow-up study for the next 5 years to find out how widespread this is,” he said. “We want to make sure it’s not necessarily just occurring in the settings we studied, and we also need to establish the mechanism of resistance.”
Further commenting on the results, Dr. Clancy noted that “the extent of resistance caught many in the field a bit off guard, as they are higher than previously reported.”
“The data are also concerning because most centers do not routinely test C. difficile for drug susceptibility.”
Dr. Clancy noted that “another immediately pressing need is to understand mechanisms of resistance. It was quite striking that vancomycin-resistant strains in this study did not carry vanA genes, pointing to previously unrecognized mechanisms of resistance.”
“As is often the case, antibiotic overuse was likely a factor in the resistances, with overtesting often leading to overtreatment of C. difficile,” Dr. Clancy said. “The situation may have been compounded by failure to appreciate how entrenched C. difficile resistance may be at certain hospitals, since widespread susceptibility testing is generally not routinely performed.”
As alternative treatments, Dr. Clancy pointed to the recent IDSA update, which included a stronger endorsement of fidaxomicin.
“Of course, there is also the need to assure that data on resistance to agents like fidaxomicin are generated going forward,” he noted.
The study was supported by was supported by National Institutes of Health, the National Institute of Allergy and Infectious Diseases, the Texas Medical Center Digestive Diseases Center, and the University of Texas Health Science Center. Dr. Darkoh has disclosed no relevant financial relationships. One coauthor received grant support from Merck, Entasis Pharmaceuticals, and MeMed Diagnostics. Dr. Clancy disclosed advisory board, consulting and/or research relationships with Merck, Qpex Biopharma, Shionogi, Astellas, Cidara, Scynexis, and Needham & Associates.
A version of this article first appeared on Medscape.com.
Samples from patients in the United States and Kenya show an increasing emergence of previously undetected vancomycin-resistant strains of Clostridioides difficile, sparking concern as recurrences in the treatment of C. difficile infection (CDI) continue to rise.
“Our results may help explain a decreasing effectiveness of antibiotic-based therapy in C. difficile infection, since a significant proportion of patients harboring strains with reduced susceptibility to vancomycin may not respond to treatment,” reported the authors in research published recently in Clinical Infectious Diseases.
The spread of the resistant strains “has serious public health implications, underscoring an urgent need for a comprehensive analysis of the circulating strains to help inform clinical decisions,” they added.
Commenting on the findings, Cornelius J. Clancy, MD, professor of medicine at the University of Pittsburgh, and chief of infectious diseases at the Veterans Affairs Pittsburgh Healthcare System, echoed the concern.
“The casual belief has been that [C. difficile] strains at most centers can be assumed to be vancomycin susceptible,” he told this news organization. “This study shows that this assumption can no longer be taken as a given.”
Dr. Clancy, who was not involved with this research, noted that “based on this study, there might be need for the Infectious Diseases Society of America and other organizations to offer guidance on generating good, quality surveillance data for C. difficile resistance.”
With C. difficile showing the ability to resist multiple antibiotics, drugs in the armamentarium to treat the infection have declined in recent years, and recurrences with the infection are reported in up to 25% of cases.
Oral vancomycin is recommended as the antibiotic of choice by the IDSA and the Society for Healthcare Epidemiology of America for severe as well as nonsevere cases of CDI, and although there are reports of nine vancomycin-resistant gene clusters, most involve Enterococcus.
To take a closer look at the prevalence of vancomycin-resistant C. difficile strains, first author Charles Darkoh, PhD, with the Center for Infectious Diseases at the University of Texas Health Science Center, Houston, and colleagues analyzed stool samples from patients with CDI, including 438 patients in Houston, taken between 2012 and 2017, and 98 in Nairobi, Kenya, taken in 2017.
They found that, among samples from patients in Houston, over the time period, 26% showed vancomycin nonsusceptible C. difficile isolates and 29% had isolates that were metronidazole resistant.
And among samples from the Nairobi patients, 67% harbored vancomycin-resistant isolates and 85% had isolates resistant to metronidazole.
Of note, the proportion of samples containing vancomycin-resistant C. difficile in the Houston patients showed a marked increase over time, from «complete absence» in 2012 to approximately 35% in 2017, the authors reported.
“These nonsusceptibility rates significantly exceeded prior reports from other studies conducted in the United States and Europe from 2011 to 2014, suggesting a lower percentage of resistance to both metronidazole and vancomycin,” the authors wrote.
Further experiments on mouse models infected with one of the vancomycin-resistant isolates showed that treatment with vancomycin failed to eradicate the infection, and 5-day survival was significantly lower after vancomycin treatment in those mice (25%) versus those infected with strains known to be vancomycin sensitive (50%).
Unrecognized genetic strains
Whole-genome sequencing of 10 of the resistant isolates showed no matches with gene clusters that have been previously recognized as being vancomycin resistant, suggesting the emergence of new clusters.
“Together, these results suggest unknown genetic elements associated with vancomycin nonsusceptibility in isolates circulating in the patient population,” the authors wrote.
Dr. Darkoh told this news organization that the research team is currently working to further investigate the patterns and mechanisms.
“We are currently working on a follow-up study for the next 5 years to find out how widespread this is,” he said. “We want to make sure it’s not necessarily just occurring in the settings we studied, and we also need to establish the mechanism of resistance.”
Further commenting on the results, Dr. Clancy noted that “the extent of resistance caught many in the field a bit off guard, as they are higher than previously reported.”
“The data are also concerning because most centers do not routinely test C. difficile for drug susceptibility.”
Dr. Clancy noted that “another immediately pressing need is to understand mechanisms of resistance. It was quite striking that vancomycin-resistant strains in this study did not carry vanA genes, pointing to previously unrecognized mechanisms of resistance.”
“As is often the case, antibiotic overuse was likely a factor in the resistances, with overtesting often leading to overtreatment of C. difficile,” Dr. Clancy said. “The situation may have been compounded by failure to appreciate how entrenched C. difficile resistance may be at certain hospitals, since widespread susceptibility testing is generally not routinely performed.”
As alternative treatments, Dr. Clancy pointed to the recent IDSA update, which included a stronger endorsement of fidaxomicin.
“Of course, there is also the need to assure that data on resistance to agents like fidaxomicin are generated going forward,” he noted.
The study was supported by was supported by National Institutes of Health, the National Institute of Allergy and Infectious Diseases, the Texas Medical Center Digestive Diseases Center, and the University of Texas Health Science Center. Dr. Darkoh has disclosed no relevant financial relationships. One coauthor received grant support from Merck, Entasis Pharmaceuticals, and MeMed Diagnostics. Dr. Clancy disclosed advisory board, consulting and/or research relationships with Merck, Qpex Biopharma, Shionogi, Astellas, Cidara, Scynexis, and Needham & Associates.
A version of this article first appeared on Medscape.com.
FROM CLINICAL INFECTIOUS DISEASES
Infant bronchiolitis subtype may predict asthma risk
Bronchiolitis is the leading cause of infant hospitalizations in the United States and Europe, and almost one-third of these patients go on to develop asthma later in childhood.
But a multinational team of researchers has presented evidence that could avoid that outcome. They identified four different subtypes of bronchiolitis along with a decision tree that can determine which infants are most likely to develop asthma as they get older.
Reporting in the journal eClinical Medicine, Michimasa Fujiogi, MD, of Massachusetts General Hospital and Harvard University, Boston, and colleagues analyzed three multicenter prospective cohort studies that included a combined 3,081 infants hospitalized with severe bronchiolitis.
“This study added a base for the early identification of high-risk patients during early infancy,” Dr. Fujiogi said in an interview. “Using the prediction rule of this study, it is possible to identify groups at high risk of asthma during a critical period of airway development – early infancy.”
The researchers identified four clinically distinct and reproducible profiles of infants hospitalized for bronchiolitis:
- A: characterized by a history of breathing problems and eczema, rhinovirus infection, and low prevalence of respiratory syncytial virus (RSV) infection.
- B: characterized by the classic symptoms of wheezing and cough at presentation, a low prevalence of previous breathing problems and rhinovirus infection, and a high likelihood of RSV infection.
- C: the most severe group, characterized by inadequate oral intake, severe retraction at presentation, and longer hospital stays.
- D: the least ill group, with little history of breathing problems but inadequate oral intake with no or mild retraction.
Infants with profile A had the highest risk for developing asthma – more than 250% greater than with typical bronchiolitis. They were also older and were more likely to have parents who had asthma – and none had solo-RSV infection. In the overall analysis, the risk for developing asthma by age 6 or 7 was 23%.
The researchers stated that the decision tree accurately predicts the high-risk profile with high degrees of sensitivity and specificity. The decision tree used four predictors that together defined infants with profile A: RSV infection status, previous breathing problems, eczema, and parental asthma.
“Our data would facilitate the development of profile-specific prevention strategies for asthma – for example, modification of host response, prophylaxis for severe viral infection – by identifying asthma risk groups early in infancy,” Dr. Fujiogi said.
The study received funding from the National Institutes of Health. Dr. Fujiogi and coauthors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Bronchiolitis is the leading cause of infant hospitalizations in the United States and Europe, and almost one-third of these patients go on to develop asthma later in childhood.
But a multinational team of researchers has presented evidence that could avoid that outcome. They identified four different subtypes of bronchiolitis along with a decision tree that can determine which infants are most likely to develop asthma as they get older.
Reporting in the journal eClinical Medicine, Michimasa Fujiogi, MD, of Massachusetts General Hospital and Harvard University, Boston, and colleagues analyzed three multicenter prospective cohort studies that included a combined 3,081 infants hospitalized with severe bronchiolitis.
“This study added a base for the early identification of high-risk patients during early infancy,” Dr. Fujiogi said in an interview. “Using the prediction rule of this study, it is possible to identify groups at high risk of asthma during a critical period of airway development – early infancy.”
The researchers identified four clinically distinct and reproducible profiles of infants hospitalized for bronchiolitis:
- A: characterized by a history of breathing problems and eczema, rhinovirus infection, and low prevalence of respiratory syncytial virus (RSV) infection.
- B: characterized by the classic symptoms of wheezing and cough at presentation, a low prevalence of previous breathing problems and rhinovirus infection, and a high likelihood of RSV infection.
- C: the most severe group, characterized by inadequate oral intake, severe retraction at presentation, and longer hospital stays.
- D: the least ill group, with little history of breathing problems but inadequate oral intake with no or mild retraction.
Infants with profile A had the highest risk for developing asthma – more than 250% greater than with typical bronchiolitis. They were also older and were more likely to have parents who had asthma – and none had solo-RSV infection. In the overall analysis, the risk for developing asthma by age 6 or 7 was 23%.
The researchers stated that the decision tree accurately predicts the high-risk profile with high degrees of sensitivity and specificity. The decision tree used four predictors that together defined infants with profile A: RSV infection status, previous breathing problems, eczema, and parental asthma.
“Our data would facilitate the development of profile-specific prevention strategies for asthma – for example, modification of host response, prophylaxis for severe viral infection – by identifying asthma risk groups early in infancy,” Dr. Fujiogi said.
The study received funding from the National Institutes of Health. Dr. Fujiogi and coauthors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Bronchiolitis is the leading cause of infant hospitalizations in the United States and Europe, and almost one-third of these patients go on to develop asthma later in childhood.
But a multinational team of researchers has presented evidence that could avoid that outcome. They identified four different subtypes of bronchiolitis along with a decision tree that can determine which infants are most likely to develop asthma as they get older.
Reporting in the journal eClinical Medicine, Michimasa Fujiogi, MD, of Massachusetts General Hospital and Harvard University, Boston, and colleagues analyzed three multicenter prospective cohort studies that included a combined 3,081 infants hospitalized with severe bronchiolitis.
“This study added a base for the early identification of high-risk patients during early infancy,” Dr. Fujiogi said in an interview. “Using the prediction rule of this study, it is possible to identify groups at high risk of asthma during a critical period of airway development – early infancy.”
The researchers identified four clinically distinct and reproducible profiles of infants hospitalized for bronchiolitis:
- A: characterized by a history of breathing problems and eczema, rhinovirus infection, and low prevalence of respiratory syncytial virus (RSV) infection.
- B: characterized by the classic symptoms of wheezing and cough at presentation, a low prevalence of previous breathing problems and rhinovirus infection, and a high likelihood of RSV infection.
- C: the most severe group, characterized by inadequate oral intake, severe retraction at presentation, and longer hospital stays.
- D: the least ill group, with little history of breathing problems but inadequate oral intake with no or mild retraction.
Infants with profile A had the highest risk for developing asthma – more than 250% greater than with typical bronchiolitis. They were also older and were more likely to have parents who had asthma – and none had solo-RSV infection. In the overall analysis, the risk for developing asthma by age 6 or 7 was 23%.
The researchers stated that the decision tree accurately predicts the high-risk profile with high degrees of sensitivity and specificity. The decision tree used four predictors that together defined infants with profile A: RSV infection status, previous breathing problems, eczema, and parental asthma.
“Our data would facilitate the development of profile-specific prevention strategies for asthma – for example, modification of host response, prophylaxis for severe viral infection – by identifying asthma risk groups early in infancy,” Dr. Fujiogi said.
The study received funding from the National Institutes of Health. Dr. Fujiogi and coauthors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Routine vaginal cleansing seen ineffective for unscheduled cesareans
Vaginal cleansing showed no reduction in morbidity when performed before unscheduled cesarean deliveries, researchers reported at the 2022 Pregnancy Meeting of the Society for Maternal-Fetal Medicine.
Several studies have evaluated vaginal cleansing prior to cesarean delivery, with mixed results. The American College of Obstetricians and Gynecologists recommends clinicians consider cleansing prior to unscheduled cesareans, but that advice appears not to be widely heeded.
The new findings, from what the researchers called the single largest study of vaginal cleansing prior to cesarean delivery in the United States, showed no difference in post-cesarean infections when the vagina was cleansed with povidone-iodine prior to unscheduled cesarean delivery.
“These findings do not support routine vaginal cleansing prior to unscheduled cesarean deliveries,” lead author Lorene Atkins Temming, MD, medical director of labor and delivery at Atrium Health Wake Forest School of Medicine, Charlotte, North Carolina, told this news organization. The research was conducted at and sponsored by Washington University School of Medicine, St. Louis, where Dr. Temming did her fellowship.
Dr. Temming’s group compared vaginal cleansing with povidone-iodine in addition to routine abdominal cleansing to abdominal cleansing alone. Among the primary outcomes of the study was the effect of cleansing on post-cesarean infectious morbidity.
“There is a higher risk of infectious complications after cesarean delivery than other gynecologic surgeries,” Dr. Temming told this news organization. “While the reason for this isn’t entirely clear, it is thought to be because cesareans are often performed after a patient’s cervix is dilated. This dilation can allow normal bacteria that live in the vagina to ascend into the uterus and can increase the risk of infections.”
Patients undergoing cesarean delivery after labor were randomly assigned to undergo preoperative abdominal cleansing only (n = 304) or preoperative abdominal cleansing plus vaginal cleansing with povidone-iodine (n = 304). Women were included in the analysis if they underwent cesareans after regular contractions and any cervical dilation, if their membranes ruptured, or if they had the procedure performed when they were more than 4 cm dilated.
The primary outcome was composite infectious morbidity, a catchall that included surgical-site infection, maternal fever, endometritis, and wound complications within 30 days after cesarean delivery. The secondary outcomes were hospital readmission, visits to the emergency department, and treatment for neonatal sepsis.
The researchers observed no significant difference in the primary composite outcome between the two groups (11.7% vs. 11.7%, P = .98; 95% confidence interval, 0.6-1.5). “Vaginal cleansing appears to be unnecessary when preoperative antibiotics and skin antisepsis are performed,” Dr. Temming said.
Jennifer L. Lew, MD, an ob/gyn at Northwestern Medicine Kishwaukee Hospital in Dekalb, Illinois, said current practice regarding preparation for unscheduled cesarean surgery includes chlorhexidine on the abdomen and povidone-iodine for introducing a Foley catheter into the urethra.
“Many patients may already have a catheter in place due to labor and epidural, so they would not need” vaginal prep, Dr. Lew said. “Currently, the standard does not require doing a vaginal prep for any cesarean sections, those in labor or not.”
The researchers have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Vaginal cleansing showed no reduction in morbidity when performed before unscheduled cesarean deliveries, researchers reported at the 2022 Pregnancy Meeting of the Society for Maternal-Fetal Medicine.
Several studies have evaluated vaginal cleansing prior to cesarean delivery, with mixed results. The American College of Obstetricians and Gynecologists recommends clinicians consider cleansing prior to unscheduled cesareans, but that advice appears not to be widely heeded.
The new findings, from what the researchers called the single largest study of vaginal cleansing prior to cesarean delivery in the United States, showed no difference in post-cesarean infections when the vagina was cleansed with povidone-iodine prior to unscheduled cesarean delivery.
“These findings do not support routine vaginal cleansing prior to unscheduled cesarean deliveries,” lead author Lorene Atkins Temming, MD, medical director of labor and delivery at Atrium Health Wake Forest School of Medicine, Charlotte, North Carolina, told this news organization. The research was conducted at and sponsored by Washington University School of Medicine, St. Louis, where Dr. Temming did her fellowship.
Dr. Temming’s group compared vaginal cleansing with povidone-iodine in addition to routine abdominal cleansing to abdominal cleansing alone. Among the primary outcomes of the study was the effect of cleansing on post-cesarean infectious morbidity.
“There is a higher risk of infectious complications after cesarean delivery than other gynecologic surgeries,” Dr. Temming told this news organization. “While the reason for this isn’t entirely clear, it is thought to be because cesareans are often performed after a patient’s cervix is dilated. This dilation can allow normal bacteria that live in the vagina to ascend into the uterus and can increase the risk of infections.”
Patients undergoing cesarean delivery after labor were randomly assigned to undergo preoperative abdominal cleansing only (n = 304) or preoperative abdominal cleansing plus vaginal cleansing with povidone-iodine (n = 304). Women were included in the analysis if they underwent cesareans after regular contractions and any cervical dilation, if their membranes ruptured, or if they had the procedure performed when they were more than 4 cm dilated.
The primary outcome was composite infectious morbidity, a catchall that included surgical-site infection, maternal fever, endometritis, and wound complications within 30 days after cesarean delivery. The secondary outcomes were hospital readmission, visits to the emergency department, and treatment for neonatal sepsis.
The researchers observed no significant difference in the primary composite outcome between the two groups (11.7% vs. 11.7%, P = .98; 95% confidence interval, 0.6-1.5). “Vaginal cleansing appears to be unnecessary when preoperative antibiotics and skin antisepsis are performed,” Dr. Temming said.
Jennifer L. Lew, MD, an ob/gyn at Northwestern Medicine Kishwaukee Hospital in Dekalb, Illinois, said current practice regarding preparation for unscheduled cesarean surgery includes chlorhexidine on the abdomen and povidone-iodine for introducing a Foley catheter into the urethra.
“Many patients may already have a catheter in place due to labor and epidural, so they would not need” vaginal prep, Dr. Lew said. “Currently, the standard does not require doing a vaginal prep for any cesarean sections, those in labor or not.”
The researchers have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Vaginal cleansing showed no reduction in morbidity when performed before unscheduled cesarean deliveries, researchers reported at the 2022 Pregnancy Meeting of the Society for Maternal-Fetal Medicine.
Several studies have evaluated vaginal cleansing prior to cesarean delivery, with mixed results. The American College of Obstetricians and Gynecologists recommends clinicians consider cleansing prior to unscheduled cesareans, but that advice appears not to be widely heeded.
The new findings, from what the researchers called the single largest study of vaginal cleansing prior to cesarean delivery in the United States, showed no difference in post-cesarean infections when the vagina was cleansed with povidone-iodine prior to unscheduled cesarean delivery.
“These findings do not support routine vaginal cleansing prior to unscheduled cesarean deliveries,” lead author Lorene Atkins Temming, MD, medical director of labor and delivery at Atrium Health Wake Forest School of Medicine, Charlotte, North Carolina, told this news organization. The research was conducted at and sponsored by Washington University School of Medicine, St. Louis, where Dr. Temming did her fellowship.
Dr. Temming’s group compared vaginal cleansing with povidone-iodine in addition to routine abdominal cleansing to abdominal cleansing alone. Among the primary outcomes of the study was the effect of cleansing on post-cesarean infectious morbidity.
“There is a higher risk of infectious complications after cesarean delivery than other gynecologic surgeries,” Dr. Temming told this news organization. “While the reason for this isn’t entirely clear, it is thought to be because cesareans are often performed after a patient’s cervix is dilated. This dilation can allow normal bacteria that live in the vagina to ascend into the uterus and can increase the risk of infections.”
Patients undergoing cesarean delivery after labor were randomly assigned to undergo preoperative abdominal cleansing only (n = 304) or preoperative abdominal cleansing plus vaginal cleansing with povidone-iodine (n = 304). Women were included in the analysis if they underwent cesareans after regular contractions and any cervical dilation, if their membranes ruptured, or if they had the procedure performed when they were more than 4 cm dilated.
The primary outcome was composite infectious morbidity, a catchall that included surgical-site infection, maternal fever, endometritis, and wound complications within 30 days after cesarean delivery. The secondary outcomes were hospital readmission, visits to the emergency department, and treatment for neonatal sepsis.
The researchers observed no significant difference in the primary composite outcome between the two groups (11.7% vs. 11.7%, P = .98; 95% confidence interval, 0.6-1.5). “Vaginal cleansing appears to be unnecessary when preoperative antibiotics and skin antisepsis are performed,” Dr. Temming said.
Jennifer L. Lew, MD, an ob/gyn at Northwestern Medicine Kishwaukee Hospital in Dekalb, Illinois, said current practice regarding preparation for unscheduled cesarean surgery includes chlorhexidine on the abdomen and povidone-iodine for introducing a Foley catheter into the urethra.
“Many patients may already have a catheter in place due to labor and epidural, so they would not need” vaginal prep, Dr. Lew said. “Currently, the standard does not require doing a vaginal prep for any cesarean sections, those in labor or not.”
The researchers have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Indurated Violaceous Lesions on the Face, Trunk, and Legs
The Diagnosis: Kaposi Sarcoma
A punch biopsy of a lesion on the right side of the back revealed a diffuse, poorly circumscribed, spindle cell neoplasm of the papillary and reticular dermis with associated vascular and pseudovascular spaces distended by erythrocytes (Figure 1). Immunostaining was positive for human herpesvirus 8 (HHV-8)(Figure 2), ETS-related gene, CD31, and CD34 and negative for pan cytokeratin, confirming the diagnosis of Kaposi sarcoma (KS). Bacterial, fungal, and mycobacterial tissue cultures were negative. The patient was tested for HIV and referred to infectious disease and oncology. He subsequently was found to have HIV with a viral load greater than 1 million copies. He was started on antiretroviral therapy and Pneumocystis jirovecii pneumonia prophylaxis. Computed tomography of the chest, abdomen, and pelvis showed bilateral, multifocal, perihilar, flame-shaped consolidations suggestive of KS. The patient later disclosed having an intermittent dry cough of more than a year’s duration with occasional bright red blood per rectum after bowel movements. After workup, the patient was found to have cytomegalovirus esophagitis/gastritis and candidal esophagitis that were treated with valganciclovir and fluconazole, respectively.
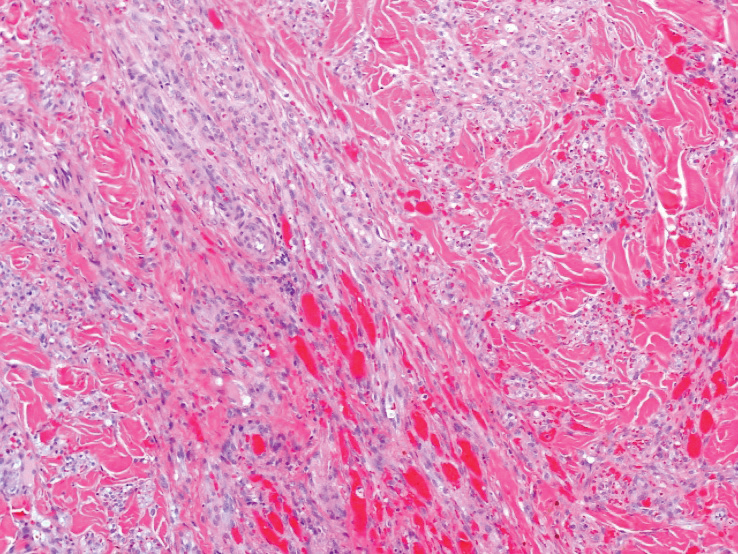
Kaposi sarcoma is an angioproliferative, AIDSdefining disease associated with HHV-8. There are 4 types of KS as defined by the populations they affect. AIDS-associated KS occurs in individuals with HIV, as seen in our patient. It often is accompanied by extensive mucocutaneous and visceral lesions, as well as systemic symptoms such as fever, weight loss, and diarrhea.1 Classic KS is a variant that presents in older men of Mediterranean, Eastern European, and South American descent. Cutaneous lesions typically are distributed on the lower extremities.2,3 Endemic (African) KS is seen in HIV-negative children and young adults in equatorial Africa. It most commonly affects the lower extremities or lymph nodes and usually follows a more aggressive course.2 Lastly, iatrogenic KS is associated with immunosuppressive medications or conditions, such as organ transplantation, chemotherapy, and rheumatologic disorders.3,4
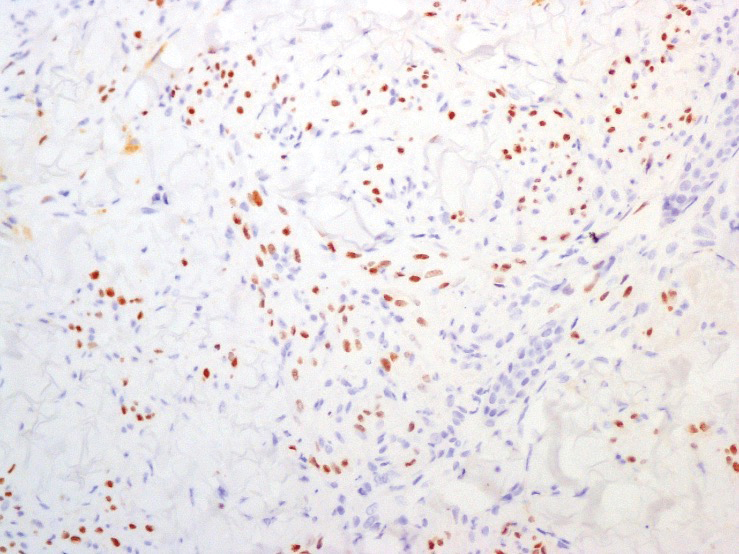
Kaposi sarcoma commonly presents as violaceous or dark red macules, patches, papules, plaques, and nodules on various parts of the body (Figure 3). Lesions typically begin as macules and progress into plaques or nodules. Our patient presented as a deceptively healthy young man with lesions at various stages of development. In addition to the skin and oral mucosa, the lungs, lymph nodes, and gastrointestinal tract commonly are involved in AIDS-associated KS.5 Patients may experience symptoms of internal involvement, including bleeding, hematochezia, odynophagia, or dyspnea.
The differential diagnosis includes conditions that can mimic KS, including bacillary angiomatosis, angioinvasive fungal disease, sarcoid, and other malignancies. A skin biopsy is the gold standard for definitive diagnosis of KS. Histopathology shows a vascular proliferation in the dermis and spindle cell proliferation.6 Kaposi sarcoma stains positively for factor VIII–related antigen, CD31, and CD34.2 Additionally, staining for HHV-8 gene products, such as latency-associated nuclear antigen 1, is helpful in differentiating KS from other conditions.7
In HIV-associated KS, the mainstay of treatment is initiation of highly active antiretroviral therapy. Typically, as the CD4 count rises with treatment, the tumor burden classic KS, effective treatment options include recurrent cryotherapy or intralesional chemotherapeutics, such as vincristine, for localized lesions; for widespread disease, pegylated liposomal doxorubicin or radiation have been found to be effective options. Lastly, for patients with iatrogenic KS, reducing immunosuppressive medications is a reasonable first step in management. If this does not yield adequate improvement, transitioning from calcineurin inhibitors (eg, cyclosporine) to proliferation signal inhibitors (eg, sirolimus) may lead to resolution.7
- Friedman-Kien AE, Saltzman BR. Clinical manifestations of classical, endemic African, and epidemic AIDS-associated Kaposi’s sarcoma. J Am Acad Dermatol. 1990;22:1237-1250.
- Radu O, Pantanowitz L. Kaposi sarcoma. Arch Pathol Lab Med. 2013;137:289-294.
- Vangipuram R, Tyring SK. Epidemiology of Kaposi sarcoma: review and description of the nonepidemic variant. Int J Dermatol. 2019;58:538-542.
- Klepp O, Dahl O, Stenwig JT. Association of Kaposi’s sarcoma and prior immunosuppressive therapy. a 5‐year material of Kaposi’s sarcoma in Norway. Cancer. 1978;42:2626-2630.
- Lemlich G, Schwam L, Lebwohl M. Kaposi’s sarcoma and acquired immunodeficiency syndrome: postmortem findings in twenty-four cases. J Am Acad Dermatol. 1987;16:319-325.
- Kaposi sarcoma. Nat Rev Dis Primers. 2019;5:10.
- Curtiss P, Strazzulla LC, Friedman-Kien AE. An update on Kaposi’s sarcoma: epidemiology, pathogenesis and treatment. Dermatol Ther. 2016;6:465-470.
The Diagnosis: Kaposi Sarcoma
A punch biopsy of a lesion on the right side of the back revealed a diffuse, poorly circumscribed, spindle cell neoplasm of the papillary and reticular dermis with associated vascular and pseudovascular spaces distended by erythrocytes (Figure 1). Immunostaining was positive for human herpesvirus 8 (HHV-8)(Figure 2), ETS-related gene, CD31, and CD34 and negative for pan cytokeratin, confirming the diagnosis of Kaposi sarcoma (KS). Bacterial, fungal, and mycobacterial tissue cultures were negative. The patient was tested for HIV and referred to infectious disease and oncology. He subsequently was found to have HIV with a viral load greater than 1 million copies. He was started on antiretroviral therapy and Pneumocystis jirovecii pneumonia prophylaxis. Computed tomography of the chest, abdomen, and pelvis showed bilateral, multifocal, perihilar, flame-shaped consolidations suggestive of KS. The patient later disclosed having an intermittent dry cough of more than a year’s duration with occasional bright red blood per rectum after bowel movements. After workup, the patient was found to have cytomegalovirus esophagitis/gastritis and candidal esophagitis that were treated with valganciclovir and fluconazole, respectively.

Kaposi sarcoma is an angioproliferative, AIDSdefining disease associated with HHV-8. There are 4 types of KS as defined by the populations they affect. AIDS-associated KS occurs in individuals with HIV, as seen in our patient. It often is accompanied by extensive mucocutaneous and visceral lesions, as well as systemic symptoms such as fever, weight loss, and diarrhea.1 Classic KS is a variant that presents in older men of Mediterranean, Eastern European, and South American descent. Cutaneous lesions typically are distributed on the lower extremities.2,3 Endemic (African) KS is seen in HIV-negative children and young adults in equatorial Africa. It most commonly affects the lower extremities or lymph nodes and usually follows a more aggressive course.2 Lastly, iatrogenic KS is associated with immunosuppressive medications or conditions, such as organ transplantation, chemotherapy, and rheumatologic disorders.3,4

Kaposi sarcoma commonly presents as violaceous or dark red macules, patches, papules, plaques, and nodules on various parts of the body (Figure 3). Lesions typically begin as macules and progress into plaques or nodules. Our patient presented as a deceptively healthy young man with lesions at various stages of development. In addition to the skin and oral mucosa, the lungs, lymph nodes, and gastrointestinal tract commonly are involved in AIDS-associated KS.5 Patients may experience symptoms of internal involvement, including bleeding, hematochezia, odynophagia, or dyspnea.

The differential diagnosis includes conditions that can mimic KS, including bacillary angiomatosis, angioinvasive fungal disease, sarcoid, and other malignancies. A skin biopsy is the gold standard for definitive diagnosis of KS. Histopathology shows a vascular proliferation in the dermis and spindle cell proliferation.6 Kaposi sarcoma stains positively for factor VIII–related antigen, CD31, and CD34.2 Additionally, staining for HHV-8 gene products, such as latency-associated nuclear antigen 1, is helpful in differentiating KS from other conditions.7
In HIV-associated KS, the mainstay of treatment is initiation of highly active antiretroviral therapy. Typically, as the CD4 count rises with treatment, the tumor burden classic KS, effective treatment options include recurrent cryotherapy or intralesional chemotherapeutics, such as vincristine, for localized lesions; for widespread disease, pegylated liposomal doxorubicin or radiation have been found to be effective options. Lastly, for patients with iatrogenic KS, reducing immunosuppressive medications is a reasonable first step in management. If this does not yield adequate improvement, transitioning from calcineurin inhibitors (eg, cyclosporine) to proliferation signal inhibitors (eg, sirolimus) may lead to resolution.7
The Diagnosis: Kaposi Sarcoma
A punch biopsy of a lesion on the right side of the back revealed a diffuse, poorly circumscribed, spindle cell neoplasm of the papillary and reticular dermis with associated vascular and pseudovascular spaces distended by erythrocytes (Figure 1). Immunostaining was positive for human herpesvirus 8 (HHV-8)(Figure 2), ETS-related gene, CD31, and CD34 and negative for pan cytokeratin, confirming the diagnosis of Kaposi sarcoma (KS). Bacterial, fungal, and mycobacterial tissue cultures were negative. The patient was tested for HIV and referred to infectious disease and oncology. He subsequently was found to have HIV with a viral load greater than 1 million copies. He was started on antiretroviral therapy and Pneumocystis jirovecii pneumonia prophylaxis. Computed tomography of the chest, abdomen, and pelvis showed bilateral, multifocal, perihilar, flame-shaped consolidations suggestive of KS. The patient later disclosed having an intermittent dry cough of more than a year’s duration with occasional bright red blood per rectum after bowel movements. After workup, the patient was found to have cytomegalovirus esophagitis/gastritis and candidal esophagitis that were treated with valganciclovir and fluconazole, respectively.

Kaposi sarcoma is an angioproliferative, AIDSdefining disease associated with HHV-8. There are 4 types of KS as defined by the populations they affect. AIDS-associated KS occurs in individuals with HIV, as seen in our patient. It often is accompanied by extensive mucocutaneous and visceral lesions, as well as systemic symptoms such as fever, weight loss, and diarrhea.1 Classic KS is a variant that presents in older men of Mediterranean, Eastern European, and South American descent. Cutaneous lesions typically are distributed on the lower extremities.2,3 Endemic (African) KS is seen in HIV-negative children and young adults in equatorial Africa. It most commonly affects the lower extremities or lymph nodes and usually follows a more aggressive course.2 Lastly, iatrogenic KS is associated with immunosuppressive medications or conditions, such as organ transplantation, chemotherapy, and rheumatologic disorders.3,4

Kaposi sarcoma commonly presents as violaceous or dark red macules, patches, papules, plaques, and nodules on various parts of the body (Figure 3). Lesions typically begin as macules and progress into plaques or nodules. Our patient presented as a deceptively healthy young man with lesions at various stages of development. In addition to the skin and oral mucosa, the lungs, lymph nodes, and gastrointestinal tract commonly are involved in AIDS-associated KS.5 Patients may experience symptoms of internal involvement, including bleeding, hematochezia, odynophagia, or dyspnea.

The differential diagnosis includes conditions that can mimic KS, including bacillary angiomatosis, angioinvasive fungal disease, sarcoid, and other malignancies. A skin biopsy is the gold standard for definitive diagnosis of KS. Histopathology shows a vascular proliferation in the dermis and spindle cell proliferation.6 Kaposi sarcoma stains positively for factor VIII–related antigen, CD31, and CD34.2 Additionally, staining for HHV-8 gene products, such as latency-associated nuclear antigen 1, is helpful in differentiating KS from other conditions.7
In HIV-associated KS, the mainstay of treatment is initiation of highly active antiretroviral therapy. Typically, as the CD4 count rises with treatment, the tumor burden classic KS, effective treatment options include recurrent cryotherapy or intralesional chemotherapeutics, such as vincristine, for localized lesions; for widespread disease, pegylated liposomal doxorubicin or radiation have been found to be effective options. Lastly, for patients with iatrogenic KS, reducing immunosuppressive medications is a reasonable first step in management. If this does not yield adequate improvement, transitioning from calcineurin inhibitors (eg, cyclosporine) to proliferation signal inhibitors (eg, sirolimus) may lead to resolution.7
- Friedman-Kien AE, Saltzman BR. Clinical manifestations of classical, endemic African, and epidemic AIDS-associated Kaposi’s sarcoma. J Am Acad Dermatol. 1990;22:1237-1250.
- Radu O, Pantanowitz L. Kaposi sarcoma. Arch Pathol Lab Med. 2013;137:289-294.
- Vangipuram R, Tyring SK. Epidemiology of Kaposi sarcoma: review and description of the nonepidemic variant. Int J Dermatol. 2019;58:538-542.
- Klepp O, Dahl O, Stenwig JT. Association of Kaposi’s sarcoma and prior immunosuppressive therapy. a 5‐year material of Kaposi’s sarcoma in Norway. Cancer. 1978;42:2626-2630.
- Lemlich G, Schwam L, Lebwohl M. Kaposi’s sarcoma and acquired immunodeficiency syndrome: postmortem findings in twenty-four cases. J Am Acad Dermatol. 1987;16:319-325.
- Kaposi sarcoma. Nat Rev Dis Primers. 2019;5:10.
- Curtiss P, Strazzulla LC, Friedman-Kien AE. An update on Kaposi’s sarcoma: epidemiology, pathogenesis and treatment. Dermatol Ther. 2016;6:465-470.
- Friedman-Kien AE, Saltzman BR. Clinical manifestations of classical, endemic African, and epidemic AIDS-associated Kaposi’s sarcoma. J Am Acad Dermatol. 1990;22:1237-1250.
- Radu O, Pantanowitz L. Kaposi sarcoma. Arch Pathol Lab Med. 2013;137:289-294.
- Vangipuram R, Tyring SK. Epidemiology of Kaposi sarcoma: review and description of the nonepidemic variant. Int J Dermatol. 2019;58:538-542.
- Klepp O, Dahl O, Stenwig JT. Association of Kaposi’s sarcoma and prior immunosuppressive therapy. a 5‐year material of Kaposi’s sarcoma in Norway. Cancer. 1978;42:2626-2630.
- Lemlich G, Schwam L, Lebwohl M. Kaposi’s sarcoma and acquired immunodeficiency syndrome: postmortem findings in twenty-four cases. J Am Acad Dermatol. 1987;16:319-325.
- Kaposi sarcoma. Nat Rev Dis Primers. 2019;5:10.
- Curtiss P, Strazzulla LC, Friedman-Kien AE. An update on Kaposi’s sarcoma: epidemiology, pathogenesis and treatment. Dermatol Ther. 2016;6:465-470.
A 25-year-old man with no notable medical history presented to the dermatology clinic with growing selfdescribed cysts on the face, trunk, and legs of 6 months’ duration. The lesions started as bruiselike discolorations and progressed to become firm nodules and inflamed masses. Some were minimally itchy and sensitive to touch, but there was no history of bleeding or drainage. The patient denied any new or recent environmental or animal exposures, use of illicit drugs, or travel correlating with the rash onset. He denied any prior treatments. He reported being in his normal state of health and was not taking any medications. Physical examination revealed indurated, violaceous, purpuric subcutaneous nodules, plaques, and masses on the forehead, cheek (top), jaw, flank, axillae (bottom), and back.
Mosquito nets do prevent malaria, longitudinal study confirms
It seems obvious that increased use of mosquito bed nets in sub-Saharan Africa would decrease the incidence of malaria, but a lingering question remained:
Malaria from Plasmodium falciparum infection exacts a significant toll in sub-Saharan Africa. According to the World Health Organization, there were about 228 million cases and 602,000 deaths from malaria in 2020 alone. About 80% of those deaths were in children less than 5 years old. In some areas, as many as 5% of children die from malaria by age 5.
Efforts to reduce the burden of malaria have been ongoing for decades. In the 1990s, insecticide-treated nets were shown to reduce illness and deaths from malaria in children.
As a result, the use of bed nets has grown significantly. In 2000, only 5% of households in sub-Saharan Africa had a net in the house. By 2020, that number had risen to 65%. From 2004 to 2019 about 1.9 billion nets were distributed in this region. The nets are estimated to have prevented more than 663 million malaria cases between 2000 and 2015.
As described in the NEJM report, public health researchers conducted a 22-year prospective longitudinal cohort study in rural southern Tanzania following 6,706 children born between 1998 and 2000. Initially, home visits were made every 4 months from May 1998 to April 2003. Remarkably, in 2019, they were able to verify the status of fully 89% of those people by reaching out to families and community/village leaders.
Günther Fink, PhD, associate professor of epidemiology and household economics, University of Basel (Switzerland), explained the approach and primary findings to this news organization. The analysis looked at three main groups – children whose parents said they always slept under treated nets, those who slept protected most of the time, and those who spent less than half the time under bed nets. The hazard ratio for death was 0.57 (95% confidence interval, 0.45-0.72) for the first two groups, compared with the least protected. The corresponding hazard ratio between age 5 and adulthood was 0.93 (95% CI, 0.58-1.49).
The findings confirmed what they had suspected. Dr. Fink summarized simply, “If you always slept under a net, you did much better than if you never slept under the net. If you slept [under a net] more than half of the time, it was much better than if you slept [under a net] less than half the time.” So the more time children slept under bed nets, the less likely they were to acquire malaria. Dr. Fink stressed that the findings showing protective efficacy persisted into adulthood. “It seems just having a healthier early life actually makes you more resilient against other future infections.”
One of the theoretical concerns was that using nets would delay developing functional immunity and that there might be an increase in mortality seen later. This study showed that did not happen.
An accompanying commentary noted that there was some potential that families receiving nets were better off than those that didn’t but concluded that such confounding had been accounted for in other analyses.
Mark Wilson, ScD, professor emeritus of epidemiology, University of Michigan, Ann Arbor, concurred. He told this news organization that the study was “very well designed,” and the researchers “did a fantastic job” in tracking patients 20 years later.
“This is astounding!” he added. “It’s very rare to find this amount of follow-up.”
Dr. Fink’s conclusion? “Bed nets protect you in the short run, and being protected in the short run is also beneficial in the long run. There is no evidence that protecting kids in early childhood is weakening them in any way. So we should keep doing this.”
Dr. Fink and Dr. Wilson report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
It seems obvious that increased use of mosquito bed nets in sub-Saharan Africa would decrease the incidence of malaria, but a lingering question remained:
Malaria from Plasmodium falciparum infection exacts a significant toll in sub-Saharan Africa. According to the World Health Organization, there were about 228 million cases and 602,000 deaths from malaria in 2020 alone. About 80% of those deaths were in children less than 5 years old. In some areas, as many as 5% of children die from malaria by age 5.
Efforts to reduce the burden of malaria have been ongoing for decades. In the 1990s, insecticide-treated nets were shown to reduce illness and deaths from malaria in children.
As a result, the use of bed nets has grown significantly. In 2000, only 5% of households in sub-Saharan Africa had a net in the house. By 2020, that number had risen to 65%. From 2004 to 2019 about 1.9 billion nets were distributed in this region. The nets are estimated to have prevented more than 663 million malaria cases between 2000 and 2015.
As described in the NEJM report, public health researchers conducted a 22-year prospective longitudinal cohort study in rural southern Tanzania following 6,706 children born between 1998 and 2000. Initially, home visits were made every 4 months from May 1998 to April 2003. Remarkably, in 2019, they were able to verify the status of fully 89% of those people by reaching out to families and community/village leaders.
Günther Fink, PhD, associate professor of epidemiology and household economics, University of Basel (Switzerland), explained the approach and primary findings to this news organization. The analysis looked at three main groups – children whose parents said they always slept under treated nets, those who slept protected most of the time, and those who spent less than half the time under bed nets. The hazard ratio for death was 0.57 (95% confidence interval, 0.45-0.72) for the first two groups, compared with the least protected. The corresponding hazard ratio between age 5 and adulthood was 0.93 (95% CI, 0.58-1.49).
The findings confirmed what they had suspected. Dr. Fink summarized simply, “If you always slept under a net, you did much better than if you never slept under the net. If you slept [under a net] more than half of the time, it was much better than if you slept [under a net] less than half the time.” So the more time children slept under bed nets, the less likely they were to acquire malaria. Dr. Fink stressed that the findings showing protective efficacy persisted into adulthood. “It seems just having a healthier early life actually makes you more resilient against other future infections.”
One of the theoretical concerns was that using nets would delay developing functional immunity and that there might be an increase in mortality seen later. This study showed that did not happen.
An accompanying commentary noted that there was some potential that families receiving nets were better off than those that didn’t but concluded that such confounding had been accounted for in other analyses.
Mark Wilson, ScD, professor emeritus of epidemiology, University of Michigan, Ann Arbor, concurred. He told this news organization that the study was “very well designed,” and the researchers “did a fantastic job” in tracking patients 20 years later.
“This is astounding!” he added. “It’s very rare to find this amount of follow-up.”
Dr. Fink’s conclusion? “Bed nets protect you in the short run, and being protected in the short run is also beneficial in the long run. There is no evidence that protecting kids in early childhood is weakening them in any way. So we should keep doing this.”
Dr. Fink and Dr. Wilson report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
It seems obvious that increased use of mosquito bed nets in sub-Saharan Africa would decrease the incidence of malaria, but a lingering question remained:
Malaria from Plasmodium falciparum infection exacts a significant toll in sub-Saharan Africa. According to the World Health Organization, there were about 228 million cases and 602,000 deaths from malaria in 2020 alone. About 80% of those deaths were in children less than 5 years old. In some areas, as many as 5% of children die from malaria by age 5.
Efforts to reduce the burden of malaria have been ongoing for decades. In the 1990s, insecticide-treated nets were shown to reduce illness and deaths from malaria in children.
As a result, the use of bed nets has grown significantly. In 2000, only 5% of households in sub-Saharan Africa had a net in the house. By 2020, that number had risen to 65%. From 2004 to 2019 about 1.9 billion nets were distributed in this region. The nets are estimated to have prevented more than 663 million malaria cases between 2000 and 2015.
As described in the NEJM report, public health researchers conducted a 22-year prospective longitudinal cohort study in rural southern Tanzania following 6,706 children born between 1998 and 2000. Initially, home visits were made every 4 months from May 1998 to April 2003. Remarkably, in 2019, they were able to verify the status of fully 89% of those people by reaching out to families and community/village leaders.
Günther Fink, PhD, associate professor of epidemiology and household economics, University of Basel (Switzerland), explained the approach and primary findings to this news organization. The analysis looked at three main groups – children whose parents said they always slept under treated nets, those who slept protected most of the time, and those who spent less than half the time under bed nets. The hazard ratio for death was 0.57 (95% confidence interval, 0.45-0.72) for the first two groups, compared with the least protected. The corresponding hazard ratio between age 5 and adulthood was 0.93 (95% CI, 0.58-1.49).
The findings confirmed what they had suspected. Dr. Fink summarized simply, “If you always slept under a net, you did much better than if you never slept under the net. If you slept [under a net] more than half of the time, it was much better than if you slept [under a net] less than half the time.” So the more time children slept under bed nets, the less likely they were to acquire malaria. Dr. Fink stressed that the findings showing protective efficacy persisted into adulthood. “It seems just having a healthier early life actually makes you more resilient against other future infections.”
One of the theoretical concerns was that using nets would delay developing functional immunity and that there might be an increase in mortality seen later. This study showed that did not happen.
An accompanying commentary noted that there was some potential that families receiving nets were better off than those that didn’t but concluded that such confounding had been accounted for in other analyses.
Mark Wilson, ScD, professor emeritus of epidemiology, University of Michigan, Ann Arbor, concurred. He told this news organization that the study was “very well designed,” and the researchers “did a fantastic job” in tracking patients 20 years later.
“This is astounding!” he added. “It’s very rare to find this amount of follow-up.”
Dr. Fink’s conclusion? “Bed nets protect you in the short run, and being protected in the short run is also beneficial in the long run. There is no evidence that protecting kids in early childhood is weakening them in any way. So we should keep doing this.”
Dr. Fink and Dr. Wilson report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM NEW ENGLAND JOURNAL OF MEDICINE
FDA approves 2-month dosing of injectable HIV drug Cabenuva
Cabenuva was first approved by the FDA in January 2021 to be administered once monthly to treat HIV-1 infection in virologically suppressed adults. The medication was the first injectable complete antiretroviral regimen approved by the FDA.
Cabenuva can replace a current treatment in virologically suppressed adults on a stable antiretroviral regimen with no history of treatment failure and no known or suspected resistance to rilpivirine and cabotegravir, the Janssen Pharmaceutical Companies of Johnson & Johnson said in a press release. Janssen and ViiV Healthcare codeveloped the injectable antiretroviral medication Cabenuva.
The expanded label approval “marks an important step forward in advancing the treatment landscape for people living with HIV,” said Candice Long, the president of infectious diseases and vaccines at Janssen Therapeutics, in a Feb. 1 press release. “With this milestone, adults living with HIV have a treatment option that further reduces the frequency of medication.”
This expanded approval was based on global clinical trial of 1,045 adults with HIV-1, which found Cabenuva administered every 8 weeks (3 mL dose of both cabotegravir and rilpivirine) to be noninferior to the 4-week regimen (2 mL dose of both medicines). At week 48 of the trial, the proportion of participants with viral loads above 50 copies per milliliter was 1.7% in the 2-month arm and 1.0% in the 1-month arm. The study found that rates of virological suppression were similar for both the 1-month and 2-month regimens (93.5% and 94.3%, respectively).
The most common side effects were injection site reactions, pyrexia, fatigue, headache, musculoskeletal pain, nausea, sleep disorders, dizziness, and rash. Adverse reactions reported in individuals receiving the regimen every 2 months or once monthly were similar. Cabenuva is contraindicated for patients with a hypersensitivity reaction to cabotegravir or rilpivirine or for those receiving carbamazepine, oxcarbazepine, phenobarbital, phenytoin, rifabutin, rifampin, rifapentine, St. John’s wort, and more than one dose of systemic dexamethasone.
A version of this article first appeared on Medscape.com.
Cabenuva was first approved by the FDA in January 2021 to be administered once monthly to treat HIV-1 infection in virologically suppressed adults. The medication was the first injectable complete antiretroviral regimen approved by the FDA.
Cabenuva can replace a current treatment in virologically suppressed adults on a stable antiretroviral regimen with no history of treatment failure and no known or suspected resistance to rilpivirine and cabotegravir, the Janssen Pharmaceutical Companies of Johnson & Johnson said in a press release. Janssen and ViiV Healthcare codeveloped the injectable antiretroviral medication Cabenuva.
The expanded label approval “marks an important step forward in advancing the treatment landscape for people living with HIV,” said Candice Long, the president of infectious diseases and vaccines at Janssen Therapeutics, in a Feb. 1 press release. “With this milestone, adults living with HIV have a treatment option that further reduces the frequency of medication.”
This expanded approval was based on global clinical trial of 1,045 adults with HIV-1, which found Cabenuva administered every 8 weeks (3 mL dose of both cabotegravir and rilpivirine) to be noninferior to the 4-week regimen (2 mL dose of both medicines). At week 48 of the trial, the proportion of participants with viral loads above 50 copies per milliliter was 1.7% in the 2-month arm and 1.0% in the 1-month arm. The study found that rates of virological suppression were similar for both the 1-month and 2-month regimens (93.5% and 94.3%, respectively).
The most common side effects were injection site reactions, pyrexia, fatigue, headache, musculoskeletal pain, nausea, sleep disorders, dizziness, and rash. Adverse reactions reported in individuals receiving the regimen every 2 months or once monthly were similar. Cabenuva is contraindicated for patients with a hypersensitivity reaction to cabotegravir or rilpivirine or for those receiving carbamazepine, oxcarbazepine, phenobarbital, phenytoin, rifabutin, rifampin, rifapentine, St. John’s wort, and more than one dose of systemic dexamethasone.
A version of this article first appeared on Medscape.com.
Cabenuva was first approved by the FDA in January 2021 to be administered once monthly to treat HIV-1 infection in virologically suppressed adults. The medication was the first injectable complete antiretroviral regimen approved by the FDA.
Cabenuva can replace a current treatment in virologically suppressed adults on a stable antiretroviral regimen with no history of treatment failure and no known or suspected resistance to rilpivirine and cabotegravir, the Janssen Pharmaceutical Companies of Johnson & Johnson said in a press release. Janssen and ViiV Healthcare codeveloped the injectable antiretroviral medication Cabenuva.
The expanded label approval “marks an important step forward in advancing the treatment landscape for people living with HIV,” said Candice Long, the president of infectious diseases and vaccines at Janssen Therapeutics, in a Feb. 1 press release. “With this milestone, adults living with HIV have a treatment option that further reduces the frequency of medication.”
This expanded approval was based on global clinical trial of 1,045 adults with HIV-1, which found Cabenuva administered every 8 weeks (3 mL dose of both cabotegravir and rilpivirine) to be noninferior to the 4-week regimen (2 mL dose of both medicines). At week 48 of the trial, the proportion of participants with viral loads above 50 copies per milliliter was 1.7% in the 2-month arm and 1.0% in the 1-month arm. The study found that rates of virological suppression were similar for both the 1-month and 2-month regimens (93.5% and 94.3%, respectively).
The most common side effects were injection site reactions, pyrexia, fatigue, headache, musculoskeletal pain, nausea, sleep disorders, dizziness, and rash. Adverse reactions reported in individuals receiving the regimen every 2 months or once monthly were similar. Cabenuva is contraindicated for patients with a hypersensitivity reaction to cabotegravir or rilpivirine or for those receiving carbamazepine, oxcarbazepine, phenobarbital, phenytoin, rifabutin, rifampin, rifapentine, St. John’s wort, and more than one dose of systemic dexamethasone.
A version of this article first appeared on Medscape.com.