User login
Living kidney donors should receive money for their costs of donating
This transcript has been edited for clarity.
Hi. I’m Art Caplan. I’m at the division of medical ethics at New York University’s Grossman School of Medicine in New York City.
We try very hard to get organ donation from those who die. That’s a commendable thing to do. I think doctors should always be discussing the opportunity to donate organs upon death, even in primary care settings.
It’s good to find out what people’s attitudes are. Let them learn about organ donation as something they can think about. Let them talk about it with family and friends and partners so that they know their wishes.
However, despite these efforts to encourage organ donation, we still have far fewer organs than we could use to transplant people, many people die on waiting lists because there are no organs to give them, and we’re in a situation where demand for organ transplant is actually increasing.
There is more capacity to do transplants both in the United States and elsewhere, and more people are living longer, so organ failure starts to become more common before, let’s say, terminal illness is really there. Now, we have more people who might benefit from organ transplant in an aging population.
One place to turn to help reduce the shortage of organs is to living donation. At least insofar as kidneys go, kidney donation from living persons has become a prominent source of organs for those who need kidneys – most of whom are surviving on dialysis, by the way, at a very high cost and often with a quality of life that they don’t find particularly easy to accept.
Transplant is far preferred, even though they have to take immunosuppression to keep those organ transplants going, and that has its own risks and side effects. They still get more mobility. They still are able to have a broader diet. They enjoy life far more than they do having to show up for dialysis three times a week for a couple of hours, every week, for every week that they live.
There is an interest in living kidney donation. One battle has been that, well, maybe we could get more kidneys if we just paid people to sell us their kidneys. That has been resisted, and I’ve been resistant to that idea, too, because I worry that it leads to exploitation.
The people who sell their kidneys are poor. They’re often in debt. They feel coerced by their circumstances, so they make a kidney sale. This happens in countries like India, where there are markets underground, and you see that it’s the poorest of the poor who do this, and they don’t really work their way out of debt. They just wind up without a kidney, help relieve their debt a little bit, and pretty soon, because they don’t have a job or an income except that sale of a kidney, they’re not much better off than they were before they started.
Also, people who sell kidneys for money are more likely not to admit to their own health problems, raising risks about the quality of organs. Then, of course, it puts doctors in a position to take out an organ for pay, even though it doesn’t benefit you, so that you can sell it. This raises some questions about whether that’s consistent with medical ethics.
A different idea has emerged. New York State Governor Kathy Hochul just signed legislation that allows living donors to be compensated for legitimate costs. That’s a little different matter. You’re not buying the organ, but you’re saying that if you experience health care problems due to complications from a donation, if you need money for transportation, if you lost money because you did this altruistically and you had to take time off from work and had expenses for a babysitter, restaurants, or other things, the state is going to try to create funds that will compensate you.
That, I think we should agree, is not a bad idea. You’re in a situation there where you don’t want to make people who are heroic, altruistic, and trying to help others by donating a kidney end up financially worse off.
I think there’s a difference between making someone financially whole after the decision to make a kidney available and creating a market where the poorest of the poor come forward to just sell because they see no other choice in terms of how to get rid of debts. I see these situations as not ethically equivalent, so I support efforts to try to compensate people who are our heroes. I don’t think we should ask them to financially suffer.
We’ll watch to see what happens as the New York state law comes into effect. By the way, New York is one of the states that really lags in the supply of organs for transplant, so this measure is particularly important for that state. Many other states should be considering this legislation as well.
It’s one thing to reward, if you will, donors by making sure they don’t suffer financial loss. It’s a very different thing to say, let’s have a free market and we’ll pay whoever it is that’s willing to sell us a kidney to do so. The former seems to me to be humane and just, whereas the latter risks exploitation.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Hi. I’m Art Caplan. I’m at the division of medical ethics at New York University’s Grossman School of Medicine in New York City.
We try very hard to get organ donation from those who die. That’s a commendable thing to do. I think doctors should always be discussing the opportunity to donate organs upon death, even in primary care settings.
It’s good to find out what people’s attitudes are. Let them learn about organ donation as something they can think about. Let them talk about it with family and friends and partners so that they know their wishes.
However, despite these efforts to encourage organ donation, we still have far fewer organs than we could use to transplant people, many people die on waiting lists because there are no organs to give them, and we’re in a situation where demand for organ transplant is actually increasing.
There is more capacity to do transplants both in the United States and elsewhere, and more people are living longer, so organ failure starts to become more common before, let’s say, terminal illness is really there. Now, we have more people who might benefit from organ transplant in an aging population.
One place to turn to help reduce the shortage of organs is to living donation. At least insofar as kidneys go, kidney donation from living persons has become a prominent source of organs for those who need kidneys – most of whom are surviving on dialysis, by the way, at a very high cost and often with a quality of life that they don’t find particularly easy to accept.
Transplant is far preferred, even though they have to take immunosuppression to keep those organ transplants going, and that has its own risks and side effects. They still get more mobility. They still are able to have a broader diet. They enjoy life far more than they do having to show up for dialysis three times a week for a couple of hours, every week, for every week that they live.
There is an interest in living kidney donation. One battle has been that, well, maybe we could get more kidneys if we just paid people to sell us their kidneys. That has been resisted, and I’ve been resistant to that idea, too, because I worry that it leads to exploitation.
The people who sell their kidneys are poor. They’re often in debt. They feel coerced by their circumstances, so they make a kidney sale. This happens in countries like India, where there are markets underground, and you see that it’s the poorest of the poor who do this, and they don’t really work their way out of debt. They just wind up without a kidney, help relieve their debt a little bit, and pretty soon, because they don’t have a job or an income except that sale of a kidney, they’re not much better off than they were before they started.
Also, people who sell kidneys for money are more likely not to admit to their own health problems, raising risks about the quality of organs. Then, of course, it puts doctors in a position to take out an organ for pay, even though it doesn’t benefit you, so that you can sell it. This raises some questions about whether that’s consistent with medical ethics.
A different idea has emerged. New York State Governor Kathy Hochul just signed legislation that allows living donors to be compensated for legitimate costs. That’s a little different matter. You’re not buying the organ, but you’re saying that if you experience health care problems due to complications from a donation, if you need money for transportation, if you lost money because you did this altruistically and you had to take time off from work and had expenses for a babysitter, restaurants, or other things, the state is going to try to create funds that will compensate you.
That, I think we should agree, is not a bad idea. You’re in a situation there where you don’t want to make people who are heroic, altruistic, and trying to help others by donating a kidney end up financially worse off.
I think there’s a difference between making someone financially whole after the decision to make a kidney available and creating a market where the poorest of the poor come forward to just sell because they see no other choice in terms of how to get rid of debts. I see these situations as not ethically equivalent, so I support efforts to try to compensate people who are our heroes. I don’t think we should ask them to financially suffer.
We’ll watch to see what happens as the New York state law comes into effect. By the way, New York is one of the states that really lags in the supply of organs for transplant, so this measure is particularly important for that state. Many other states should be considering this legislation as well.
It’s one thing to reward, if you will, donors by making sure they don’t suffer financial loss. It’s a very different thing to say, let’s have a free market and we’ll pay whoever it is that’s willing to sell us a kidney to do so. The former seems to me to be humane and just, whereas the latter risks exploitation.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Hi. I’m Art Caplan. I’m at the division of medical ethics at New York University’s Grossman School of Medicine in New York City.
We try very hard to get organ donation from those who die. That’s a commendable thing to do. I think doctors should always be discussing the opportunity to donate organs upon death, even in primary care settings.
It’s good to find out what people’s attitudes are. Let them learn about organ donation as something they can think about. Let them talk about it with family and friends and partners so that they know their wishes.
However, despite these efforts to encourage organ donation, we still have far fewer organs than we could use to transplant people, many people die on waiting lists because there are no organs to give them, and we’re in a situation where demand for organ transplant is actually increasing.
There is more capacity to do transplants both in the United States and elsewhere, and more people are living longer, so organ failure starts to become more common before, let’s say, terminal illness is really there. Now, we have more people who might benefit from organ transplant in an aging population.
One place to turn to help reduce the shortage of organs is to living donation. At least insofar as kidneys go, kidney donation from living persons has become a prominent source of organs for those who need kidneys – most of whom are surviving on dialysis, by the way, at a very high cost and often with a quality of life that they don’t find particularly easy to accept.
Transplant is far preferred, even though they have to take immunosuppression to keep those organ transplants going, and that has its own risks and side effects. They still get more mobility. They still are able to have a broader diet. They enjoy life far more than they do having to show up for dialysis three times a week for a couple of hours, every week, for every week that they live.
There is an interest in living kidney donation. One battle has been that, well, maybe we could get more kidneys if we just paid people to sell us their kidneys. That has been resisted, and I’ve been resistant to that idea, too, because I worry that it leads to exploitation.
The people who sell their kidneys are poor. They’re often in debt. They feel coerced by their circumstances, so they make a kidney sale. This happens in countries like India, where there are markets underground, and you see that it’s the poorest of the poor who do this, and they don’t really work their way out of debt. They just wind up without a kidney, help relieve their debt a little bit, and pretty soon, because they don’t have a job or an income except that sale of a kidney, they’re not much better off than they were before they started.
Also, people who sell kidneys for money are more likely not to admit to their own health problems, raising risks about the quality of organs. Then, of course, it puts doctors in a position to take out an organ for pay, even though it doesn’t benefit you, so that you can sell it. This raises some questions about whether that’s consistent with medical ethics.
A different idea has emerged. New York State Governor Kathy Hochul just signed legislation that allows living donors to be compensated for legitimate costs. That’s a little different matter. You’re not buying the organ, but you’re saying that if you experience health care problems due to complications from a donation, if you need money for transportation, if you lost money because you did this altruistically and you had to take time off from work and had expenses for a babysitter, restaurants, or other things, the state is going to try to create funds that will compensate you.
That, I think we should agree, is not a bad idea. You’re in a situation there where you don’t want to make people who are heroic, altruistic, and trying to help others by donating a kidney end up financially worse off.
I think there’s a difference between making someone financially whole after the decision to make a kidney available and creating a market where the poorest of the poor come forward to just sell because they see no other choice in terms of how to get rid of debts. I see these situations as not ethically equivalent, so I support efforts to try to compensate people who are our heroes. I don’t think we should ask them to financially suffer.
We’ll watch to see what happens as the New York state law comes into effect. By the way, New York is one of the states that really lags in the supply of organs for transplant, so this measure is particularly important for that state. Many other states should be considering this legislation as well.
It’s one thing to reward, if you will, donors by making sure they don’t suffer financial loss. It’s a very different thing to say, let’s have a free market and we’ll pay whoever it is that’s willing to sell us a kidney to do so. The former seems to me to be humane and just, whereas the latter risks exploitation.
A version of this article first appeared on Medscape.com.
NOVIDs: Do some have the genes to dodge COVID?
As a field service representative for a slot machine company, Ryan Alexander, 37, of Louisville, Ky., spends his working hours in casinos, covering a large territory including Norfolk, Va., Indianapolis, and Charlotte. Social distancing in the casinos is not the norm. Despite all this up-close contact with people, he said he is still COVID-free, 3 years into the pandemic.
There was one nervous night when his temperature rose to 101° F, and he figured the virus had caught up with him. “I took a test and was fine,” he said, relieved that the result was negative. The fever disappeared, and he was back to normal soon. “Maybe it was just an exhausting day.”
Mr. Alexander is one of those people who have managed – or at least think they have managed – to avoid getting COVID-19.
He is, some say, a NOVID. While some scientists cringe at the term, it’s caught on to describe these virus super-dodgers. Online entrepreneurs offer NOVID-19 T-shirts, masks, and stickers, in case these super-healthy or super-lucky folks want to publicize their good luck. On Twitter, NOVIDs share stories of how they’ve done it.
How many NOVIDs?
As of March 16, according to the CDC, almost 104 million cases of COVID – about one-third of the U.S. population – have been reported, but many cases are known to go unreported. About half of American adults surveyed said they have had COVID, according to a December report by the COVID States Project, a multiuniversity effort to supply pandemic data.
As the numbers settle over time, though, it becomes clearer that some in the U.S. have apparently managed to avoid the virus.
But some scientists bristle at the term NOVIDs. They prefer the term “resisters,” according to Elena Hsieh, MD, associate professor of pediatrics and immunology at the University of Colorado at Denver, Aurora. Currently, she said, there is much more information on who is more susceptible to contracting severe COVID than who is resistant.
Dr. Hsieh is one of the regional coordinators for the COVID Human Genetic Effort, an international consortium of more than 250 researchers and doctors dedicated to discovering the genetic and immunological bases of the forms of SARS-CoV-2 infection. These researchers and others are looking for explanations for why some people get severe COVID while others seem resistant despite repeated exposure.
Resistance research
In determining explanations for resistance to infection, “the needle in the haystack that we are looking for is a change in the genetic code that would allow for you to avoid entry of the virus into the cell,” Dr. Hsieh said. “That is what being resistant to infection is.”
Part of the reason it’s so difficult to study resistance is defining a resister, she said. While many people consider themselves among that group because they’re been exposed multiple times – even with close family members infected and sick, yet they still felt fine – that doesn’t necessarily make them a resister, she said.
Those people could have been infected but remained without symptoms. “Resistance means the virus was inside you, it was near your cell and it did not infect your cell,” Dr. Hsieh said.
“I don’t think we know a lot so far,” Dr. Hsieh said about resisters. “I do believe that, just like there are genetic defects that make someone more susceptible, there are likely to be genetic defects that make somebody less susceptible.’’
“To identify genetic variants that are protective is a really challenging thing to do,” agreed Peter K. Gregersen, MD, professor of genetics at the Feinstein Institutes for Medical Research at Northwell Health in Manhasset, N.Y. Dr. Gregersen is also a regional coordinator for the COVID Human Genetic Effort.
He suspects the number found to be truly resistant to COVID – versus dodging it so far – is going to be very small or not found at all.
“It may exist for COVID or it may not,” he said. Some people may simply have what he calls a robust immune response in the upper part of the throat, perhaps killing off the virus quickly as soon as it enters, so they don’t get a positive test.
Genetic resistance has been found for other diseases, such as HIV.
“For HIV, scientists have been able to identify a specific gene that codes for a protein that can prevent individuals from getting infected,” said Sabrina Assoumou, MD, MPH, professor of medicine at Boston University, who researches HIV.
However, she said, “we haven’t yet found a similar gene or protein that can prevent people from getting infected with SARS-CoV-2.”
What has been found “is that some people might have a mutation in a gene that encodes for what’s called human leukocyte antigen (HLA),” Dr. Assoumou said. HLA, a molecule found on the surface of most cells, has a crucial role in the immune response to foreign substances. “A mutation in HLA can make people less likely to have symptoms if they get infected. Individuals still get infected, but they are less likely to have symptoms.”
Other research has found that those with food allergies are also less likely to be infected. The researchers have speculated that the inflammation characteristic of allergic conditions may reduce levels of a protein called the ACE2 receptor on the surface of airway cells. The SARS-CoV-2 virus uses the receptor to enter the cells, so if levels are low, that could reduce the ability of the virus to infect people.
The COVID Human Genetic Effort continues to search for participants, both those who were admitted to a hospital or repeatedly seen at a hospital because of COVID, as well as those who did not get infected, even after “intense and repeated” exposure.
The number of people likely to be resistant is much smaller, Dr. Hsieh said, than the number of people susceptible to severe disease.
The testing ... or lack thereof factor
The timing of testing and a person’s “infection profile” may be factors in people incorrectly declaring themselves NOVIDs, said Anne Wyllie, PhD, a research scientist in epidemiology at the Yale School of Public Health in New Haven, Conn., and a codeveloper of a saliva PCR test for COVID.
“Infection profiles can vary between individuals,” she said. For some, the infection may start in the lower respiratory tract, others in the higher respiratory tract. “Depending on where the virus takes up residence, that can affect test results.”
Then there’s the following-instructions factor. “It’s very likely that due to tests not being done at the right time, with the right sample, or not repeated if there is ongoing evidence of symptoms, that there are individuals out there who believe they are NOVIDs but just missed catching their infection at the window of opportunity.” Dr. Wyllie said.
Susceptibility research
“The part we have proven is the genetic defect that would make you more susceptible to having severe disease,” Dr. Hsieh said.
Many published papers report that inherited and/or autoimmune deficiencies of type I interferon immunity, important for combating viral infections and modulating the immune response, can be a significant cause of life-threatening COVID pneumonia.
More recently, researchers, including Jean-Laurent Casanova, MD, PhD, professor at Rockefeller University, New York, and cofounder of the COVID Human Genome Effort, reported that deficiencies in a gene that plays a role in built-in immunity (the early response), and a gene involved in signaling within the immune cells, impair interferon production and may be the basis of severe COVID pneumonia.
NOVIDs’ habits run the gamut
As scientists continue their research, the NOVIDs have their own ideas about why they’ve dodged the pandemic bullet, and they have a variety of approaches to handling the pandemic now.
Ryan Alexander, the field rep who travels to casinos, is up to date on his vaccinations and has gotten all the recommended COVID shots. “I was wearing a mask when told to wear masks,” he said.
He still observes the social distance habit but lives life. “I’ve been to three or four concerts in the past couple of years.”
And does he worry his number will eventually be up? “Not at this point, no,” he said.
Joe Asher, 46, said he has not gotten COVID despite being in contact with about 100 people a day, on average. He works as a bartender at an Evansville, Ind., brewery.
“On a Friday night, we can get 500 people,” he said. “I feel like almost everyone at the brewery got it. There’s no way I wasn’t exposed to it all the time.”
However, he said, his coworkers who did get sick were very cautious about not infecting others, partly to help protect a coworker’s family with newborn twins, so that may have helped him stay uninfected, too.
Mr. Asher said he’s in good physical shape, and he’s worked around the public for a long time, so figures maybe that has strengthened his immune system. He’s always been careful about handwashing and said he’s perhaps a bit more conscious of germs than others might be.
Roselyn Mena, 68, a retired teacher in Richmond, Calif., about 16 miles northeast of San Francisco, said she’s managed to avoid the virus even though her husband, Jesus Mena, got infected, as did her two adult children. Now, she remains vigilant about wearing a mask. She tries not to eat inside at restaurants. “I’m super careful,” she said.
Besides her teacher training, Ms. Mena had training as a medical assistant and learned a lot about sanitizing methods. She gets an annual flu shot, washes her hands often, and uses hand sanitizer.
When she shops, she will ask salespeople not wearing masks to please mask. “Only one refused, and she got someone else [to wait on her].”
One reason she is always careful about hygiene, Ms. Mena said, is that “when I get a cold, I get really sick. It last and lasts.” Now, she does worry she might still get it, she said, with the prospect of getting long COVID driving that worry.
In the beginning of the pandemic, Rhonda Fleming, 68, of Los Angeles, lived in a “COVID bubble,” interacting with just a few close family members. As cases went down, she enlarged the bubble. Her two grown daughters got infected, but her granddaughter did not.
She has been vigilant about masking, she said, “and I do still mask in public places.” She has a mask wardrobe, including basic black as well as glittery masks for dressier occasions. “I always carry a mask because inevitably, a cougher surrounds me.”
Now, she will bypass restaurants if she doesn’t feel comfortable with the environment, choosing ones with good air flow. When she flew to Mexico recently, she masked on the plane.
At this point, she said she doesn’t worry about getting infected but remains careful.
Recently, two friends, who have been as diligent as she has about precautions, got infected, “and they don’t know how they got it.”
Bragging rights?
Until researchers separate out the true resisters from those who claim to be, some NOVIDs are simply quietly grateful for their luck, while others mention their COVID-free status to anyone who asks or who will listen, and are proud of it.
And what about those who wear a “NOVID” T-shirt?
“I would think they have a need to convey to the world they are different, perhaps special, because they beat COVID,” said Richard B. Joelson, a New York–based doctor of social work, a psychotherapist, and the author of Help Me! A Psychotherapist’s Tried-and-True Techniques for a Happier Relationship with Yourself and the People You Love. “They didn’t beat COVID, they just didn’t get it.”
Or they may be relieved they didn’t get sick, he said, because they feel defeated when they do. So “it’s a source of pride.” It might be the same people who tell anyone who will listen they never need a doctor or take no medicines, he said.
Even though science may prove many NOVIDs are inaccurate when they call themselves resisters, Dr. Hsieh understands the temptation to talk about it. “It’s kind of cool to think you are supernatural,” she said. “It’s much more attractive than being susceptible. It’s a lot sexier.” ■
A version of this article first appeared on Medscape.com.
As a field service representative for a slot machine company, Ryan Alexander, 37, of Louisville, Ky., spends his working hours in casinos, covering a large territory including Norfolk, Va., Indianapolis, and Charlotte. Social distancing in the casinos is not the norm. Despite all this up-close contact with people, he said he is still COVID-free, 3 years into the pandemic.
There was one nervous night when his temperature rose to 101° F, and he figured the virus had caught up with him. “I took a test and was fine,” he said, relieved that the result was negative. The fever disappeared, and he was back to normal soon. “Maybe it was just an exhausting day.”
Mr. Alexander is one of those people who have managed – or at least think they have managed – to avoid getting COVID-19.
He is, some say, a NOVID. While some scientists cringe at the term, it’s caught on to describe these virus super-dodgers. Online entrepreneurs offer NOVID-19 T-shirts, masks, and stickers, in case these super-healthy or super-lucky folks want to publicize their good luck. On Twitter, NOVIDs share stories of how they’ve done it.
How many NOVIDs?
As of March 16, according to the CDC, almost 104 million cases of COVID – about one-third of the U.S. population – have been reported, but many cases are known to go unreported. About half of American adults surveyed said they have had COVID, according to a December report by the COVID States Project, a multiuniversity effort to supply pandemic data.
As the numbers settle over time, though, it becomes clearer that some in the U.S. have apparently managed to avoid the virus.
But some scientists bristle at the term NOVIDs. They prefer the term “resisters,” according to Elena Hsieh, MD, associate professor of pediatrics and immunology at the University of Colorado at Denver, Aurora. Currently, she said, there is much more information on who is more susceptible to contracting severe COVID than who is resistant.
Dr. Hsieh is one of the regional coordinators for the COVID Human Genetic Effort, an international consortium of more than 250 researchers and doctors dedicated to discovering the genetic and immunological bases of the forms of SARS-CoV-2 infection. These researchers and others are looking for explanations for why some people get severe COVID while others seem resistant despite repeated exposure.
Resistance research
In determining explanations for resistance to infection, “the needle in the haystack that we are looking for is a change in the genetic code that would allow for you to avoid entry of the virus into the cell,” Dr. Hsieh said. “That is what being resistant to infection is.”
Part of the reason it’s so difficult to study resistance is defining a resister, she said. While many people consider themselves among that group because they’re been exposed multiple times – even with close family members infected and sick, yet they still felt fine – that doesn’t necessarily make them a resister, she said.
Those people could have been infected but remained without symptoms. “Resistance means the virus was inside you, it was near your cell and it did not infect your cell,” Dr. Hsieh said.
“I don’t think we know a lot so far,” Dr. Hsieh said about resisters. “I do believe that, just like there are genetic defects that make someone more susceptible, there are likely to be genetic defects that make somebody less susceptible.’’
“To identify genetic variants that are protective is a really challenging thing to do,” agreed Peter K. Gregersen, MD, professor of genetics at the Feinstein Institutes for Medical Research at Northwell Health in Manhasset, N.Y. Dr. Gregersen is also a regional coordinator for the COVID Human Genetic Effort.
He suspects the number found to be truly resistant to COVID – versus dodging it so far – is going to be very small or not found at all.
“It may exist for COVID or it may not,” he said. Some people may simply have what he calls a robust immune response in the upper part of the throat, perhaps killing off the virus quickly as soon as it enters, so they don’t get a positive test.
Genetic resistance has been found for other diseases, such as HIV.
“For HIV, scientists have been able to identify a specific gene that codes for a protein that can prevent individuals from getting infected,” said Sabrina Assoumou, MD, MPH, professor of medicine at Boston University, who researches HIV.
However, she said, “we haven’t yet found a similar gene or protein that can prevent people from getting infected with SARS-CoV-2.”
What has been found “is that some people might have a mutation in a gene that encodes for what’s called human leukocyte antigen (HLA),” Dr. Assoumou said. HLA, a molecule found on the surface of most cells, has a crucial role in the immune response to foreign substances. “A mutation in HLA can make people less likely to have symptoms if they get infected. Individuals still get infected, but they are less likely to have symptoms.”
Other research has found that those with food allergies are also less likely to be infected. The researchers have speculated that the inflammation characteristic of allergic conditions may reduce levels of a protein called the ACE2 receptor on the surface of airway cells. The SARS-CoV-2 virus uses the receptor to enter the cells, so if levels are low, that could reduce the ability of the virus to infect people.
The COVID Human Genetic Effort continues to search for participants, both those who were admitted to a hospital or repeatedly seen at a hospital because of COVID, as well as those who did not get infected, even after “intense and repeated” exposure.
The number of people likely to be resistant is much smaller, Dr. Hsieh said, than the number of people susceptible to severe disease.
The testing ... or lack thereof factor
The timing of testing and a person’s “infection profile” may be factors in people incorrectly declaring themselves NOVIDs, said Anne Wyllie, PhD, a research scientist in epidemiology at the Yale School of Public Health in New Haven, Conn., and a codeveloper of a saliva PCR test for COVID.
“Infection profiles can vary between individuals,” she said. For some, the infection may start in the lower respiratory tract, others in the higher respiratory tract. “Depending on where the virus takes up residence, that can affect test results.”
Then there’s the following-instructions factor. “It’s very likely that due to tests not being done at the right time, with the right sample, or not repeated if there is ongoing evidence of symptoms, that there are individuals out there who believe they are NOVIDs but just missed catching their infection at the window of opportunity.” Dr. Wyllie said.
Susceptibility research
“The part we have proven is the genetic defect that would make you more susceptible to having severe disease,” Dr. Hsieh said.
Many published papers report that inherited and/or autoimmune deficiencies of type I interferon immunity, important for combating viral infections and modulating the immune response, can be a significant cause of life-threatening COVID pneumonia.
More recently, researchers, including Jean-Laurent Casanova, MD, PhD, professor at Rockefeller University, New York, and cofounder of the COVID Human Genome Effort, reported that deficiencies in a gene that plays a role in built-in immunity (the early response), and a gene involved in signaling within the immune cells, impair interferon production and may be the basis of severe COVID pneumonia.
NOVIDs’ habits run the gamut
As scientists continue their research, the NOVIDs have their own ideas about why they’ve dodged the pandemic bullet, and they have a variety of approaches to handling the pandemic now.
Ryan Alexander, the field rep who travels to casinos, is up to date on his vaccinations and has gotten all the recommended COVID shots. “I was wearing a mask when told to wear masks,” he said.
He still observes the social distance habit but lives life. “I’ve been to three or four concerts in the past couple of years.”
And does he worry his number will eventually be up? “Not at this point, no,” he said.
Joe Asher, 46, said he has not gotten COVID despite being in contact with about 100 people a day, on average. He works as a bartender at an Evansville, Ind., brewery.
“On a Friday night, we can get 500 people,” he said. “I feel like almost everyone at the brewery got it. There’s no way I wasn’t exposed to it all the time.”
However, he said, his coworkers who did get sick were very cautious about not infecting others, partly to help protect a coworker’s family with newborn twins, so that may have helped him stay uninfected, too.
Mr. Asher said he’s in good physical shape, and he’s worked around the public for a long time, so figures maybe that has strengthened his immune system. He’s always been careful about handwashing and said he’s perhaps a bit more conscious of germs than others might be.
Roselyn Mena, 68, a retired teacher in Richmond, Calif., about 16 miles northeast of San Francisco, said she’s managed to avoid the virus even though her husband, Jesus Mena, got infected, as did her two adult children. Now, she remains vigilant about wearing a mask. She tries not to eat inside at restaurants. “I’m super careful,” she said.
Besides her teacher training, Ms. Mena had training as a medical assistant and learned a lot about sanitizing methods. She gets an annual flu shot, washes her hands often, and uses hand sanitizer.
When she shops, she will ask salespeople not wearing masks to please mask. “Only one refused, and she got someone else [to wait on her].”
One reason she is always careful about hygiene, Ms. Mena said, is that “when I get a cold, I get really sick. It last and lasts.” Now, she does worry she might still get it, she said, with the prospect of getting long COVID driving that worry.
In the beginning of the pandemic, Rhonda Fleming, 68, of Los Angeles, lived in a “COVID bubble,” interacting with just a few close family members. As cases went down, she enlarged the bubble. Her two grown daughters got infected, but her granddaughter did not.
She has been vigilant about masking, she said, “and I do still mask in public places.” She has a mask wardrobe, including basic black as well as glittery masks for dressier occasions. “I always carry a mask because inevitably, a cougher surrounds me.”
Now, she will bypass restaurants if she doesn’t feel comfortable with the environment, choosing ones with good air flow. When she flew to Mexico recently, she masked on the plane.
At this point, she said she doesn’t worry about getting infected but remains careful.
Recently, two friends, who have been as diligent as she has about precautions, got infected, “and they don’t know how they got it.”
Bragging rights?
Until researchers separate out the true resisters from those who claim to be, some NOVIDs are simply quietly grateful for their luck, while others mention their COVID-free status to anyone who asks or who will listen, and are proud of it.
And what about those who wear a “NOVID” T-shirt?
“I would think they have a need to convey to the world they are different, perhaps special, because they beat COVID,” said Richard B. Joelson, a New York–based doctor of social work, a psychotherapist, and the author of Help Me! A Psychotherapist’s Tried-and-True Techniques for a Happier Relationship with Yourself and the People You Love. “They didn’t beat COVID, they just didn’t get it.”
Or they may be relieved they didn’t get sick, he said, because they feel defeated when they do. So “it’s a source of pride.” It might be the same people who tell anyone who will listen they never need a doctor or take no medicines, he said.
Even though science may prove many NOVIDs are inaccurate when they call themselves resisters, Dr. Hsieh understands the temptation to talk about it. “It’s kind of cool to think you are supernatural,” she said. “It’s much more attractive than being susceptible. It’s a lot sexier.” ■
A version of this article first appeared on Medscape.com.
As a field service representative for a slot machine company, Ryan Alexander, 37, of Louisville, Ky., spends his working hours in casinos, covering a large territory including Norfolk, Va., Indianapolis, and Charlotte. Social distancing in the casinos is not the norm. Despite all this up-close contact with people, he said he is still COVID-free, 3 years into the pandemic.
There was one nervous night when his temperature rose to 101° F, and he figured the virus had caught up with him. “I took a test and was fine,” he said, relieved that the result was negative. The fever disappeared, and he was back to normal soon. “Maybe it was just an exhausting day.”
Mr. Alexander is one of those people who have managed – or at least think they have managed – to avoid getting COVID-19.
He is, some say, a NOVID. While some scientists cringe at the term, it’s caught on to describe these virus super-dodgers. Online entrepreneurs offer NOVID-19 T-shirts, masks, and stickers, in case these super-healthy or super-lucky folks want to publicize their good luck. On Twitter, NOVIDs share stories of how they’ve done it.
How many NOVIDs?
As of March 16, according to the CDC, almost 104 million cases of COVID – about one-third of the U.S. population – have been reported, but many cases are known to go unreported. About half of American adults surveyed said they have had COVID, according to a December report by the COVID States Project, a multiuniversity effort to supply pandemic data.
As the numbers settle over time, though, it becomes clearer that some in the U.S. have apparently managed to avoid the virus.
But some scientists bristle at the term NOVIDs. They prefer the term “resisters,” according to Elena Hsieh, MD, associate professor of pediatrics and immunology at the University of Colorado at Denver, Aurora. Currently, she said, there is much more information on who is more susceptible to contracting severe COVID than who is resistant.
Dr. Hsieh is one of the regional coordinators for the COVID Human Genetic Effort, an international consortium of more than 250 researchers and doctors dedicated to discovering the genetic and immunological bases of the forms of SARS-CoV-2 infection. These researchers and others are looking for explanations for why some people get severe COVID while others seem resistant despite repeated exposure.
Resistance research
In determining explanations for resistance to infection, “the needle in the haystack that we are looking for is a change in the genetic code that would allow for you to avoid entry of the virus into the cell,” Dr. Hsieh said. “That is what being resistant to infection is.”
Part of the reason it’s so difficult to study resistance is defining a resister, she said. While many people consider themselves among that group because they’re been exposed multiple times – even with close family members infected and sick, yet they still felt fine – that doesn’t necessarily make them a resister, she said.
Those people could have been infected but remained without symptoms. “Resistance means the virus was inside you, it was near your cell and it did not infect your cell,” Dr. Hsieh said.
“I don’t think we know a lot so far,” Dr. Hsieh said about resisters. “I do believe that, just like there are genetic defects that make someone more susceptible, there are likely to be genetic defects that make somebody less susceptible.’’
“To identify genetic variants that are protective is a really challenging thing to do,” agreed Peter K. Gregersen, MD, professor of genetics at the Feinstein Institutes for Medical Research at Northwell Health in Manhasset, N.Y. Dr. Gregersen is also a regional coordinator for the COVID Human Genetic Effort.
He suspects the number found to be truly resistant to COVID – versus dodging it so far – is going to be very small or not found at all.
“It may exist for COVID or it may not,” he said. Some people may simply have what he calls a robust immune response in the upper part of the throat, perhaps killing off the virus quickly as soon as it enters, so they don’t get a positive test.
Genetic resistance has been found for other diseases, such as HIV.
“For HIV, scientists have been able to identify a specific gene that codes for a protein that can prevent individuals from getting infected,” said Sabrina Assoumou, MD, MPH, professor of medicine at Boston University, who researches HIV.
However, she said, “we haven’t yet found a similar gene or protein that can prevent people from getting infected with SARS-CoV-2.”
What has been found “is that some people might have a mutation in a gene that encodes for what’s called human leukocyte antigen (HLA),” Dr. Assoumou said. HLA, a molecule found on the surface of most cells, has a crucial role in the immune response to foreign substances. “A mutation in HLA can make people less likely to have symptoms if they get infected. Individuals still get infected, but they are less likely to have symptoms.”
Other research has found that those with food allergies are also less likely to be infected. The researchers have speculated that the inflammation characteristic of allergic conditions may reduce levels of a protein called the ACE2 receptor on the surface of airway cells. The SARS-CoV-2 virus uses the receptor to enter the cells, so if levels are low, that could reduce the ability of the virus to infect people.
The COVID Human Genetic Effort continues to search for participants, both those who were admitted to a hospital or repeatedly seen at a hospital because of COVID, as well as those who did not get infected, even after “intense and repeated” exposure.
The number of people likely to be resistant is much smaller, Dr. Hsieh said, than the number of people susceptible to severe disease.
The testing ... or lack thereof factor
The timing of testing and a person’s “infection profile” may be factors in people incorrectly declaring themselves NOVIDs, said Anne Wyllie, PhD, a research scientist in epidemiology at the Yale School of Public Health in New Haven, Conn., and a codeveloper of a saliva PCR test for COVID.
“Infection profiles can vary between individuals,” she said. For some, the infection may start in the lower respiratory tract, others in the higher respiratory tract. “Depending on where the virus takes up residence, that can affect test results.”
Then there’s the following-instructions factor. “It’s very likely that due to tests not being done at the right time, with the right sample, or not repeated if there is ongoing evidence of symptoms, that there are individuals out there who believe they are NOVIDs but just missed catching their infection at the window of opportunity.” Dr. Wyllie said.
Susceptibility research
“The part we have proven is the genetic defect that would make you more susceptible to having severe disease,” Dr. Hsieh said.
Many published papers report that inherited and/or autoimmune deficiencies of type I interferon immunity, important for combating viral infections and modulating the immune response, can be a significant cause of life-threatening COVID pneumonia.
More recently, researchers, including Jean-Laurent Casanova, MD, PhD, professor at Rockefeller University, New York, and cofounder of the COVID Human Genome Effort, reported that deficiencies in a gene that plays a role in built-in immunity (the early response), and a gene involved in signaling within the immune cells, impair interferon production and may be the basis of severe COVID pneumonia.
NOVIDs’ habits run the gamut
As scientists continue their research, the NOVIDs have their own ideas about why they’ve dodged the pandemic bullet, and they have a variety of approaches to handling the pandemic now.
Ryan Alexander, the field rep who travels to casinos, is up to date on his vaccinations and has gotten all the recommended COVID shots. “I was wearing a mask when told to wear masks,” he said.
He still observes the social distance habit but lives life. “I’ve been to three or four concerts in the past couple of years.”
And does he worry his number will eventually be up? “Not at this point, no,” he said.
Joe Asher, 46, said he has not gotten COVID despite being in contact with about 100 people a day, on average. He works as a bartender at an Evansville, Ind., brewery.
“On a Friday night, we can get 500 people,” he said. “I feel like almost everyone at the brewery got it. There’s no way I wasn’t exposed to it all the time.”
However, he said, his coworkers who did get sick were very cautious about not infecting others, partly to help protect a coworker’s family with newborn twins, so that may have helped him stay uninfected, too.
Mr. Asher said he’s in good physical shape, and he’s worked around the public for a long time, so figures maybe that has strengthened his immune system. He’s always been careful about handwashing and said he’s perhaps a bit more conscious of germs than others might be.
Roselyn Mena, 68, a retired teacher in Richmond, Calif., about 16 miles northeast of San Francisco, said she’s managed to avoid the virus even though her husband, Jesus Mena, got infected, as did her two adult children. Now, she remains vigilant about wearing a mask. She tries not to eat inside at restaurants. “I’m super careful,” she said.
Besides her teacher training, Ms. Mena had training as a medical assistant and learned a lot about sanitizing methods. She gets an annual flu shot, washes her hands often, and uses hand sanitizer.
When she shops, she will ask salespeople not wearing masks to please mask. “Only one refused, and she got someone else [to wait on her].”
One reason she is always careful about hygiene, Ms. Mena said, is that “when I get a cold, I get really sick. It last and lasts.” Now, she does worry she might still get it, she said, with the prospect of getting long COVID driving that worry.
In the beginning of the pandemic, Rhonda Fleming, 68, of Los Angeles, lived in a “COVID bubble,” interacting with just a few close family members. As cases went down, she enlarged the bubble. Her two grown daughters got infected, but her granddaughter did not.
She has been vigilant about masking, she said, “and I do still mask in public places.” She has a mask wardrobe, including basic black as well as glittery masks for dressier occasions. “I always carry a mask because inevitably, a cougher surrounds me.”
Now, she will bypass restaurants if she doesn’t feel comfortable with the environment, choosing ones with good air flow. When she flew to Mexico recently, she masked on the plane.
At this point, she said she doesn’t worry about getting infected but remains careful.
Recently, two friends, who have been as diligent as she has about precautions, got infected, “and they don’t know how they got it.”
Bragging rights?
Until researchers separate out the true resisters from those who claim to be, some NOVIDs are simply quietly grateful for their luck, while others mention their COVID-free status to anyone who asks or who will listen, and are proud of it.
And what about those who wear a “NOVID” T-shirt?
“I would think they have a need to convey to the world they are different, perhaps special, because they beat COVID,” said Richard B. Joelson, a New York–based doctor of social work, a psychotherapist, and the author of Help Me! A Psychotherapist’s Tried-and-True Techniques for a Happier Relationship with Yourself and the People You Love. “They didn’t beat COVID, they just didn’t get it.”
Or they may be relieved they didn’t get sick, he said, because they feel defeated when they do. So “it’s a source of pride.” It might be the same people who tell anyone who will listen they never need a doctor or take no medicines, he said.
Even though science may prove many NOVIDs are inaccurate when they call themselves resisters, Dr. Hsieh understands the temptation to talk about it. “It’s kind of cool to think you are supernatural,” she said. “It’s much more attractive than being susceptible. It’s a lot sexier.” ■
A version of this article first appeared on Medscape.com.
Factors linked with increased VTE risk in COVID outpatients
Though VTE risk is well studied and significant in those hospitalized with COVID, little is known about the risk in the outpatient setting, said the authors of the new research published online in JAMA Network Open.
The study was conducted at two integrated health care delivery systems in northern and southern California. Data were gathered from the Kaiser Permanente Virtual Data Warehouse and electronic health records.
Nearly 400,000 patients studied
Researchers, led by Margaret Fang, MD, with the division of hospital medicine, University of California, San Francisco, identified 398,530 outpatients with COVID-19 from Jan. 1, 2020, through Jan. 31, 2021.
VTE risk was low overall for ambulatory COVID patients.
“It is a reassuring study,” Dr. Fang said in an interview.
The researchers found that the risk is highest in the first 30 days after COVID-19 diagnosis (unadjusted rate, 0.58; 95% confidence interval, 0.51-0.67 per 100 person-years vs. 0.09; 95% CI, 0.08-0.11 per 100 person-years after 30 days).
Factors linked with high VTE risk
They also found that several factors were linked with a higher risk of blood clots in the study population, including being at least 55 years old; being male; having a history of blood clots or thrombophilia; and a body mass index (BMI) of at least 30 kg/m2.
The authors write, “These findings may help identify subsets of patients with COVID-19 who could benefit from VTE preventive strategies and more intensive short-term surveillance.”
Are routine anticoagulants justified?
Previously, randomized clinical trials have found that hospitalized patients with moderate COVID-19 may benefit from therapeutically dosed heparin anticoagulants but that therapeutic anticoagulation had no net benefit – and perhaps could even harm – patients who were critically ill with COVID.
“[M]uch less is known about the optimal thromboprophylaxis strategy for people with milder presentations of COVID-19 who do not require hospitalization,” they write.
Mild COVID VTE risk similar to general population
The authors note that rates of blood clots linked with COVID-19 are not much higher than the average blood clot rate in the general population, which is about 0.1-0.2 per 100 person-years.
Therefore, the results don’t justify routine administration of anticoagulation given the costs, inconvenience, and bleeding risks, they acknowledge.
Dr. Fang told this publication that it’s hard to know what to tell patients, given the overall low VTE risk. She said their study wasn’t designed to advise when to give prophylaxis.
Physicians should inform patients of their higher risk
“We should tell our patients who fall into these risk categories that blood clot is a concern after the development of COVID, especially in those first 30 days. And some people might benefit from increased surveillance,” Dr. Fang said.
”I think this study would support ongoing studies that look at whether selected patients benefit from VTE prophylaxis, for example low-dose anticoagulants,” she said.
Dr. Fang said the subgroup factors they found increased risk of blood clots for all patients, not just COVID-19 patients. It’s not clear why factors such as being male may increase blood clot risk, though that is consistent with previous literature, but higher risk with higher BMI might be related to a combination of inflammation or decreased mobility, she said.
Unanswered questions
Robert H. Hopkins Jr., MD, says the study helps answer a couple of important questions – that the VTE risk in nonhospitalized COVID-19 patients is low and when and for which patients risk may be highest.
However, there are several unanswered questions that argue against routine initiation of anticoagulants, notes the professor of internal medicine and pediatrics chief, division of general internal medicine, at University of Arkansas for Medical Sciences, Little Rock.
One is the change in the COVID variant landscape.
“We do not know whether rates of VTE are same or lower or higher with current circulating variants,” Dr. Hopkins said.
The authors acknowledge this as a limitation. Study data predate Omicron and subvariants, which appear to lower clinical severity, so it’s unclear whether VTE risk is different in this Omicron era.
Dr. Hopkins added another unknown: “We do not know whether vaccination affects rates of VTE in ambulatory breakthrough infection.”
Dr. Hopkins and the authors also note the lack of a control group in the study, to better compare risk.
Coauthor Dr. Prasad reports consultant fees from EpiExcellence LLC outside the submitted work. Coauthor Dr. Go reports grants paid to the division of research, Kaiser Permanente Northern California, from CSL Behring, Novartis, Bristol Meyers Squibb/Pfizer Alliance, and Janssen outside the submitted work.
The research was funded through Patient-Centered Outcomes Research Institute.
Dr. Hopkins reports no relevant financial relationships.
Though VTE risk is well studied and significant in those hospitalized with COVID, little is known about the risk in the outpatient setting, said the authors of the new research published online in JAMA Network Open.
The study was conducted at two integrated health care delivery systems in northern and southern California. Data were gathered from the Kaiser Permanente Virtual Data Warehouse and electronic health records.
Nearly 400,000 patients studied
Researchers, led by Margaret Fang, MD, with the division of hospital medicine, University of California, San Francisco, identified 398,530 outpatients with COVID-19 from Jan. 1, 2020, through Jan. 31, 2021.
VTE risk was low overall for ambulatory COVID patients.
“It is a reassuring study,” Dr. Fang said in an interview.
The researchers found that the risk is highest in the first 30 days after COVID-19 diagnosis (unadjusted rate, 0.58; 95% confidence interval, 0.51-0.67 per 100 person-years vs. 0.09; 95% CI, 0.08-0.11 per 100 person-years after 30 days).
Factors linked with high VTE risk
They also found that several factors were linked with a higher risk of blood clots in the study population, including being at least 55 years old; being male; having a history of blood clots or thrombophilia; and a body mass index (BMI) of at least 30 kg/m2.
The authors write, “These findings may help identify subsets of patients with COVID-19 who could benefit from VTE preventive strategies and more intensive short-term surveillance.”
Are routine anticoagulants justified?
Previously, randomized clinical trials have found that hospitalized patients with moderate COVID-19 may benefit from therapeutically dosed heparin anticoagulants but that therapeutic anticoagulation had no net benefit – and perhaps could even harm – patients who were critically ill with COVID.
“[M]uch less is known about the optimal thromboprophylaxis strategy for people with milder presentations of COVID-19 who do not require hospitalization,” they write.
Mild COVID VTE risk similar to general population
The authors note that rates of blood clots linked with COVID-19 are not much higher than the average blood clot rate in the general population, which is about 0.1-0.2 per 100 person-years.
Therefore, the results don’t justify routine administration of anticoagulation given the costs, inconvenience, and bleeding risks, they acknowledge.
Dr. Fang told this publication that it’s hard to know what to tell patients, given the overall low VTE risk. She said their study wasn’t designed to advise when to give prophylaxis.
Physicians should inform patients of their higher risk
“We should tell our patients who fall into these risk categories that blood clot is a concern after the development of COVID, especially in those first 30 days. And some people might benefit from increased surveillance,” Dr. Fang said.
”I think this study would support ongoing studies that look at whether selected patients benefit from VTE prophylaxis, for example low-dose anticoagulants,” she said.
Dr. Fang said the subgroup factors they found increased risk of blood clots for all patients, not just COVID-19 patients. It’s not clear why factors such as being male may increase blood clot risk, though that is consistent with previous literature, but higher risk with higher BMI might be related to a combination of inflammation or decreased mobility, she said.
Unanswered questions
Robert H. Hopkins Jr., MD, says the study helps answer a couple of important questions – that the VTE risk in nonhospitalized COVID-19 patients is low and when and for which patients risk may be highest.
However, there are several unanswered questions that argue against routine initiation of anticoagulants, notes the professor of internal medicine and pediatrics chief, division of general internal medicine, at University of Arkansas for Medical Sciences, Little Rock.
One is the change in the COVID variant landscape.
“We do not know whether rates of VTE are same or lower or higher with current circulating variants,” Dr. Hopkins said.
The authors acknowledge this as a limitation. Study data predate Omicron and subvariants, which appear to lower clinical severity, so it’s unclear whether VTE risk is different in this Omicron era.
Dr. Hopkins added another unknown: “We do not know whether vaccination affects rates of VTE in ambulatory breakthrough infection.”
Dr. Hopkins and the authors also note the lack of a control group in the study, to better compare risk.
Coauthor Dr. Prasad reports consultant fees from EpiExcellence LLC outside the submitted work. Coauthor Dr. Go reports grants paid to the division of research, Kaiser Permanente Northern California, from CSL Behring, Novartis, Bristol Meyers Squibb/Pfizer Alliance, and Janssen outside the submitted work.
The research was funded through Patient-Centered Outcomes Research Institute.
Dr. Hopkins reports no relevant financial relationships.
Though VTE risk is well studied and significant in those hospitalized with COVID, little is known about the risk in the outpatient setting, said the authors of the new research published online in JAMA Network Open.
The study was conducted at two integrated health care delivery systems in northern and southern California. Data were gathered from the Kaiser Permanente Virtual Data Warehouse and electronic health records.
Nearly 400,000 patients studied
Researchers, led by Margaret Fang, MD, with the division of hospital medicine, University of California, San Francisco, identified 398,530 outpatients with COVID-19 from Jan. 1, 2020, through Jan. 31, 2021.
VTE risk was low overall for ambulatory COVID patients.
“It is a reassuring study,” Dr. Fang said in an interview.
The researchers found that the risk is highest in the first 30 days after COVID-19 diagnosis (unadjusted rate, 0.58; 95% confidence interval, 0.51-0.67 per 100 person-years vs. 0.09; 95% CI, 0.08-0.11 per 100 person-years after 30 days).
Factors linked with high VTE risk
They also found that several factors were linked with a higher risk of blood clots in the study population, including being at least 55 years old; being male; having a history of blood clots or thrombophilia; and a body mass index (BMI) of at least 30 kg/m2.
The authors write, “These findings may help identify subsets of patients with COVID-19 who could benefit from VTE preventive strategies and more intensive short-term surveillance.”
Are routine anticoagulants justified?
Previously, randomized clinical trials have found that hospitalized patients with moderate COVID-19 may benefit from therapeutically dosed heparin anticoagulants but that therapeutic anticoagulation had no net benefit – and perhaps could even harm – patients who were critically ill with COVID.
“[M]uch less is known about the optimal thromboprophylaxis strategy for people with milder presentations of COVID-19 who do not require hospitalization,” they write.
Mild COVID VTE risk similar to general population
The authors note that rates of blood clots linked with COVID-19 are not much higher than the average blood clot rate in the general population, which is about 0.1-0.2 per 100 person-years.
Therefore, the results don’t justify routine administration of anticoagulation given the costs, inconvenience, and bleeding risks, they acknowledge.
Dr. Fang told this publication that it’s hard to know what to tell patients, given the overall low VTE risk. She said their study wasn’t designed to advise when to give prophylaxis.
Physicians should inform patients of their higher risk
“We should tell our patients who fall into these risk categories that blood clot is a concern after the development of COVID, especially in those first 30 days. And some people might benefit from increased surveillance,” Dr. Fang said.
”I think this study would support ongoing studies that look at whether selected patients benefit from VTE prophylaxis, for example low-dose anticoagulants,” she said.
Dr. Fang said the subgroup factors they found increased risk of blood clots for all patients, not just COVID-19 patients. It’s not clear why factors such as being male may increase blood clot risk, though that is consistent with previous literature, but higher risk with higher BMI might be related to a combination of inflammation or decreased mobility, she said.
Unanswered questions
Robert H. Hopkins Jr., MD, says the study helps answer a couple of important questions – that the VTE risk in nonhospitalized COVID-19 patients is low and when and for which patients risk may be highest.
However, there are several unanswered questions that argue against routine initiation of anticoagulants, notes the professor of internal medicine and pediatrics chief, division of general internal medicine, at University of Arkansas for Medical Sciences, Little Rock.
One is the change in the COVID variant landscape.
“We do not know whether rates of VTE are same or lower or higher with current circulating variants,” Dr. Hopkins said.
The authors acknowledge this as a limitation. Study data predate Omicron and subvariants, which appear to lower clinical severity, so it’s unclear whether VTE risk is different in this Omicron era.
Dr. Hopkins added another unknown: “We do not know whether vaccination affects rates of VTE in ambulatory breakthrough infection.”
Dr. Hopkins and the authors also note the lack of a control group in the study, to better compare risk.
Coauthor Dr. Prasad reports consultant fees from EpiExcellence LLC outside the submitted work. Coauthor Dr. Go reports grants paid to the division of research, Kaiser Permanente Northern California, from CSL Behring, Novartis, Bristol Meyers Squibb/Pfizer Alliance, and Janssen outside the submitted work.
The research was funded through Patient-Centered Outcomes Research Institute.
Dr. Hopkins reports no relevant financial relationships.
FROM JAMA NETWORK OPEN
Can particles in dairy and beef cause cancer and MS?
In Western diets, dairy and beef are ubiquitous: Milk goes with coffee, melted cheese with pizza, and chili with rice. But what if dairy products and beef contained a new kind of pathogen that could infect you as a child and trigger cancer or multiple sclerosis (MS) 40-70 years later?
However, in two joint statements, the German Federal Institute for Risk Assessment (BfR) and the Max Rubner Institute (MRI) have rejected such theories.
In 2008, Harald zur Hausen, MD, DSc, received the Nobel Prize in Medicine for his discovery that human papillomaviruses cause cervical cancer. His starting point was the observation that sexually abstinent women, such as nuns, rarely develop this cancer. So it was possible to draw the conclusion that pathogens are transmitted during sexual intercourse, explain Dr. zur Hausen and his wife Ethel-Michele de Villiers, PhD, both of DKFZ Heidelberg.
Papillomaviruses, as well as human herpes and Epstein-Barr viruses (EBV), polyomaviruses, and retroviruses, cause cancer in a direct way: by inserting their genes into the DNA of human cells. With a latency of a few years to a few decades, the proteins formed through expression stimulate malignant growth by altering the regulating host gene.
Acid radicals
However, viruses – just like bacteria and parasites – can also indirectly trigger cancer. One mechanism for this triggering is the disruption of immune defenses, as shown by the sometimes drastically increased tumor incidence with AIDS or with immunosuppressants after transplants. Chronic inflammation is a second mechanism that generates acid radicals and thereby causes random mutations in replicating cells. Examples include stomach cancer caused by Helicobacter pylori and liver cancer caused by Schistosoma, liver fluke, and hepatitis B and C viruses.
According to Dr. de Villiers and Dr. zur Hausen, there are good reasons to believe that other pathogens could cause chronic inflammation and thereby lead to cancer. Epidemiologic data suggest that dairy and meat products from European cows (Bos taurus) are a potential source. This is because colon cancer and breast cancer commonly occur in places where these foods are heavily consumed (that is, in North America, Argentina, Europe, and Australia). In contrast, the rate is low in India, where cows are revered as holy animals. Also noteworthy is that women with a lactose intolerance rarely develop breast cancer.
Viral progeny
In fact, the researchers found single-stranded DNA rings that originated in viruses, which they named bovine meat and milk factors (BMMF), in the intestines of patients with colon cancer. They reported, “This new class of pathogen deserves, in our opinion at least, to become the focus of cancer development and further chronic diseases.” They also detected elevated levels of acid radicals in these areas (that is, oxidative stress), which is typical for chronic inflammation.
The researchers assume that infants, whose immune system is not yet fully matured, ingest the BMMF as soon as they have dairy. Therefore, there is no need for adults to avoid dairy or beef because everyone is infected anyway, said Dr. zur Hausen.
‘Breast milk is healthy’
Dr. De Villiers and Dr. zur Hausen outlined more evidence of cancer-triggering pathogens. Mothers who have breastfed are less likely, especially after multiple pregnancies, to develop tumors in various organs or to have MS and type 2 diabetes. The authors attribute the protective effect to oligosaccharides in breast milk, which begin to be formed midway through the pregnancy. They bind to lectin receptors and, in so doing, mask the terminal molecule onto which the viruses need to dock. As a result, their port of entry into the cells is blocked.
The oligosaccharides also protect the baby against life-threatening infections by blocking access by rotaviruses and noroviruses. In this way, especially if breastfeeding lasts a long time – around 1 year – the period of incomplete immunocompetence is bridged.
Colon cancer
To date, it has been assumed that around 20% of all cancerous diseases globally are caused by infections, said the researchers. But if the suspected BMMF cases are included, this figure rises to 50%, even to around 80%, for colon cancer. If the suspicion is confirmed, the consequences for prevention and therapy would be significant.
The voice of a Nobel prize winner undoubtedly carries weight, but at the time, Dr. zur Hausen still had to convince a host of skeptics with his discovery that a viral infection is a major cause of cervical cancer. Nonetheless, some indicators suggest that he and his wife have found a dead end this time.
Institutional skepticism
When his working group made the results public in February 2019, the DKFZ felt the need to give an all-clear signal in response to alarmed press reports. There is no reason to see dairy and meat consumption as something negative. Similarly, in their first joint statement, the BfR and the MRI judged the data to be insufficient and called for further studies. Multiple research teams began to focus on BMMF as a result. In what foods can they be found? Are they more common in patients with cancer than in healthy people? Are they infectious? Do they cause inflammation and cancer?
The findings presented in a second statement by the BfR and MRI at the end of November 2022 contradicted the claims made by the DKFZ scientists across the board. In no way do BMMF represent new pathogens. They are variants of already known DNA sequences. In addition, they are present in numerous animal-based and plant-based foods, including pork, fish, fruit, vegetables, and nuts.
BMMF do not possess the ability to infect human cells, the institutes said. The proof that they are damaging to one’s health was also absent. It is true that the incidence of intestinal tumors correlates positively with the consumption of red and processed meat – which in no way signifies causality – but dairy products are linked to a reduced risk. On the other hand, breast cancer cannot be associated with the consumption of beef or dairy.
Therefore, both institutes recommend continuing to use these products as supplementary diet for infants because of their micronutrients. They further stated that the products are safe for people of all ages.
Association with MS?
Unperturbed, Dr. de Villiers and Dr. zur Hausen went one step further in their current article. They posited that MS is also associated with the consumption of dairy products and beef. Here too geographic distribution prompted the idea to look for BMMF in the brain lesions of patients with MS. The researchers isolated ring-shaped DNA molecules that proved to be closely related to BMMF from dairy and cattle blood. “The result was electrifying for us.”
However, there are several other factors to consider, such as vitamin D3 deficiency. This is because the incidence of MS decreases the further you travel from the poles toward the equator (that is, as solar radiation increases). Also, EBV clearly plays a role because patients with MS display increased titers of EBV antibodies. One study also showed that people in Antarctica excreted reactivated EBV in their saliva during winter and that vitamin D3 stopped the viral secretion.
Under these conditions, the researchers hypothesized that MS is caused by a double infection of brain cells by EBV and BMMF. EBV is reactivated by a lack of vitamin D3, and the BMMF multiply and are eventually converted into proteins. A focal immunoreaction causes the Schwann cells and oligodendrocytes to malfunction, which leads to the destruction of the myelin sheaths around the nerve fibers.
This article was translated from the Medscape German Edition. A version appeared on Medscape.com.
In Western diets, dairy and beef are ubiquitous: Milk goes with coffee, melted cheese with pizza, and chili with rice. But what if dairy products and beef contained a new kind of pathogen that could infect you as a child and trigger cancer or multiple sclerosis (MS) 40-70 years later?
However, in two joint statements, the German Federal Institute for Risk Assessment (BfR) and the Max Rubner Institute (MRI) have rejected such theories.
In 2008, Harald zur Hausen, MD, DSc, received the Nobel Prize in Medicine for his discovery that human papillomaviruses cause cervical cancer. His starting point was the observation that sexually abstinent women, such as nuns, rarely develop this cancer. So it was possible to draw the conclusion that pathogens are transmitted during sexual intercourse, explain Dr. zur Hausen and his wife Ethel-Michele de Villiers, PhD, both of DKFZ Heidelberg.
Papillomaviruses, as well as human herpes and Epstein-Barr viruses (EBV), polyomaviruses, and retroviruses, cause cancer in a direct way: by inserting their genes into the DNA of human cells. With a latency of a few years to a few decades, the proteins formed through expression stimulate malignant growth by altering the regulating host gene.
Acid radicals
However, viruses – just like bacteria and parasites – can also indirectly trigger cancer. One mechanism for this triggering is the disruption of immune defenses, as shown by the sometimes drastically increased tumor incidence with AIDS or with immunosuppressants after transplants. Chronic inflammation is a second mechanism that generates acid radicals and thereby causes random mutations in replicating cells. Examples include stomach cancer caused by Helicobacter pylori and liver cancer caused by Schistosoma, liver fluke, and hepatitis B and C viruses.
According to Dr. de Villiers and Dr. zur Hausen, there are good reasons to believe that other pathogens could cause chronic inflammation and thereby lead to cancer. Epidemiologic data suggest that dairy and meat products from European cows (Bos taurus) are a potential source. This is because colon cancer and breast cancer commonly occur in places where these foods are heavily consumed (that is, in North America, Argentina, Europe, and Australia). In contrast, the rate is low in India, where cows are revered as holy animals. Also noteworthy is that women with a lactose intolerance rarely develop breast cancer.
Viral progeny
In fact, the researchers found single-stranded DNA rings that originated in viruses, which they named bovine meat and milk factors (BMMF), in the intestines of patients with colon cancer. They reported, “This new class of pathogen deserves, in our opinion at least, to become the focus of cancer development and further chronic diseases.” They also detected elevated levels of acid radicals in these areas (that is, oxidative stress), which is typical for chronic inflammation.
The researchers assume that infants, whose immune system is not yet fully matured, ingest the BMMF as soon as they have dairy. Therefore, there is no need for adults to avoid dairy or beef because everyone is infected anyway, said Dr. zur Hausen.
‘Breast milk is healthy’
Dr. De Villiers and Dr. zur Hausen outlined more evidence of cancer-triggering pathogens. Mothers who have breastfed are less likely, especially after multiple pregnancies, to develop tumors in various organs or to have MS and type 2 diabetes. The authors attribute the protective effect to oligosaccharides in breast milk, which begin to be formed midway through the pregnancy. They bind to lectin receptors and, in so doing, mask the terminal molecule onto which the viruses need to dock. As a result, their port of entry into the cells is blocked.
The oligosaccharides also protect the baby against life-threatening infections by blocking access by rotaviruses and noroviruses. In this way, especially if breastfeeding lasts a long time – around 1 year – the period of incomplete immunocompetence is bridged.
Colon cancer
To date, it has been assumed that around 20% of all cancerous diseases globally are caused by infections, said the researchers. But if the suspected BMMF cases are included, this figure rises to 50%, even to around 80%, for colon cancer. If the suspicion is confirmed, the consequences for prevention and therapy would be significant.
The voice of a Nobel prize winner undoubtedly carries weight, but at the time, Dr. zur Hausen still had to convince a host of skeptics with his discovery that a viral infection is a major cause of cervical cancer. Nonetheless, some indicators suggest that he and his wife have found a dead end this time.
Institutional skepticism
When his working group made the results public in February 2019, the DKFZ felt the need to give an all-clear signal in response to alarmed press reports. There is no reason to see dairy and meat consumption as something negative. Similarly, in their first joint statement, the BfR and the MRI judged the data to be insufficient and called for further studies. Multiple research teams began to focus on BMMF as a result. In what foods can they be found? Are they more common in patients with cancer than in healthy people? Are they infectious? Do they cause inflammation and cancer?
The findings presented in a second statement by the BfR and MRI at the end of November 2022 contradicted the claims made by the DKFZ scientists across the board. In no way do BMMF represent new pathogens. They are variants of already known DNA sequences. In addition, they are present in numerous animal-based and plant-based foods, including pork, fish, fruit, vegetables, and nuts.
BMMF do not possess the ability to infect human cells, the institutes said. The proof that they are damaging to one’s health was also absent. It is true that the incidence of intestinal tumors correlates positively with the consumption of red and processed meat – which in no way signifies causality – but dairy products are linked to a reduced risk. On the other hand, breast cancer cannot be associated with the consumption of beef or dairy.
Therefore, both institutes recommend continuing to use these products as supplementary diet for infants because of their micronutrients. They further stated that the products are safe for people of all ages.
Association with MS?
Unperturbed, Dr. de Villiers and Dr. zur Hausen went one step further in their current article. They posited that MS is also associated with the consumption of dairy products and beef. Here too geographic distribution prompted the idea to look for BMMF in the brain lesions of patients with MS. The researchers isolated ring-shaped DNA molecules that proved to be closely related to BMMF from dairy and cattle blood. “The result was electrifying for us.”
However, there are several other factors to consider, such as vitamin D3 deficiency. This is because the incidence of MS decreases the further you travel from the poles toward the equator (that is, as solar radiation increases). Also, EBV clearly plays a role because patients with MS display increased titers of EBV antibodies. One study also showed that people in Antarctica excreted reactivated EBV in their saliva during winter and that vitamin D3 stopped the viral secretion.
Under these conditions, the researchers hypothesized that MS is caused by a double infection of brain cells by EBV and BMMF. EBV is reactivated by a lack of vitamin D3, and the BMMF multiply and are eventually converted into proteins. A focal immunoreaction causes the Schwann cells and oligodendrocytes to malfunction, which leads to the destruction of the myelin sheaths around the nerve fibers.
This article was translated from the Medscape German Edition. A version appeared on Medscape.com.
In Western diets, dairy and beef are ubiquitous: Milk goes with coffee, melted cheese with pizza, and chili with rice. But what if dairy products and beef contained a new kind of pathogen that could infect you as a child and trigger cancer or multiple sclerosis (MS) 40-70 years later?
However, in two joint statements, the German Federal Institute for Risk Assessment (BfR) and the Max Rubner Institute (MRI) have rejected such theories.
In 2008, Harald zur Hausen, MD, DSc, received the Nobel Prize in Medicine for his discovery that human papillomaviruses cause cervical cancer. His starting point was the observation that sexually abstinent women, such as nuns, rarely develop this cancer. So it was possible to draw the conclusion that pathogens are transmitted during sexual intercourse, explain Dr. zur Hausen and his wife Ethel-Michele de Villiers, PhD, both of DKFZ Heidelberg.
Papillomaviruses, as well as human herpes and Epstein-Barr viruses (EBV), polyomaviruses, and retroviruses, cause cancer in a direct way: by inserting their genes into the DNA of human cells. With a latency of a few years to a few decades, the proteins formed through expression stimulate malignant growth by altering the regulating host gene.
Acid radicals
However, viruses – just like bacteria and parasites – can also indirectly trigger cancer. One mechanism for this triggering is the disruption of immune defenses, as shown by the sometimes drastically increased tumor incidence with AIDS or with immunosuppressants after transplants. Chronic inflammation is a second mechanism that generates acid radicals and thereby causes random mutations in replicating cells. Examples include stomach cancer caused by Helicobacter pylori and liver cancer caused by Schistosoma, liver fluke, and hepatitis B and C viruses.
According to Dr. de Villiers and Dr. zur Hausen, there are good reasons to believe that other pathogens could cause chronic inflammation and thereby lead to cancer. Epidemiologic data suggest that dairy and meat products from European cows (Bos taurus) are a potential source. This is because colon cancer and breast cancer commonly occur in places where these foods are heavily consumed (that is, in North America, Argentina, Europe, and Australia). In contrast, the rate is low in India, where cows are revered as holy animals. Also noteworthy is that women with a lactose intolerance rarely develop breast cancer.
Viral progeny
In fact, the researchers found single-stranded DNA rings that originated in viruses, which they named bovine meat and milk factors (BMMF), in the intestines of patients with colon cancer. They reported, “This new class of pathogen deserves, in our opinion at least, to become the focus of cancer development and further chronic diseases.” They also detected elevated levels of acid radicals in these areas (that is, oxidative stress), which is typical for chronic inflammation.
The researchers assume that infants, whose immune system is not yet fully matured, ingest the BMMF as soon as they have dairy. Therefore, there is no need for adults to avoid dairy or beef because everyone is infected anyway, said Dr. zur Hausen.
‘Breast milk is healthy’
Dr. De Villiers and Dr. zur Hausen outlined more evidence of cancer-triggering pathogens. Mothers who have breastfed are less likely, especially after multiple pregnancies, to develop tumors in various organs or to have MS and type 2 diabetes. The authors attribute the protective effect to oligosaccharides in breast milk, which begin to be formed midway through the pregnancy. They bind to lectin receptors and, in so doing, mask the terminal molecule onto which the viruses need to dock. As a result, their port of entry into the cells is blocked.
The oligosaccharides also protect the baby against life-threatening infections by blocking access by rotaviruses and noroviruses. In this way, especially if breastfeeding lasts a long time – around 1 year – the period of incomplete immunocompetence is bridged.
Colon cancer
To date, it has been assumed that around 20% of all cancerous diseases globally are caused by infections, said the researchers. But if the suspected BMMF cases are included, this figure rises to 50%, even to around 80%, for colon cancer. If the suspicion is confirmed, the consequences for prevention and therapy would be significant.
The voice of a Nobel prize winner undoubtedly carries weight, but at the time, Dr. zur Hausen still had to convince a host of skeptics with his discovery that a viral infection is a major cause of cervical cancer. Nonetheless, some indicators suggest that he and his wife have found a dead end this time.
Institutional skepticism
When his working group made the results public in February 2019, the DKFZ felt the need to give an all-clear signal in response to alarmed press reports. There is no reason to see dairy and meat consumption as something negative. Similarly, in their first joint statement, the BfR and the MRI judged the data to be insufficient and called for further studies. Multiple research teams began to focus on BMMF as a result. In what foods can they be found? Are they more common in patients with cancer than in healthy people? Are they infectious? Do they cause inflammation and cancer?
The findings presented in a second statement by the BfR and MRI at the end of November 2022 contradicted the claims made by the DKFZ scientists across the board. In no way do BMMF represent new pathogens. They are variants of already known DNA sequences. In addition, they are present in numerous animal-based and plant-based foods, including pork, fish, fruit, vegetables, and nuts.
BMMF do not possess the ability to infect human cells, the institutes said. The proof that they are damaging to one’s health was also absent. It is true that the incidence of intestinal tumors correlates positively with the consumption of red and processed meat – which in no way signifies causality – but dairy products are linked to a reduced risk. On the other hand, breast cancer cannot be associated with the consumption of beef or dairy.
Therefore, both institutes recommend continuing to use these products as supplementary diet for infants because of their micronutrients. They further stated that the products are safe for people of all ages.
Association with MS?
Unperturbed, Dr. de Villiers and Dr. zur Hausen went one step further in their current article. They posited that MS is also associated with the consumption of dairy products and beef. Here too geographic distribution prompted the idea to look for BMMF in the brain lesions of patients with MS. The researchers isolated ring-shaped DNA molecules that proved to be closely related to BMMF from dairy and cattle blood. “The result was electrifying for us.”
However, there are several other factors to consider, such as vitamin D3 deficiency. This is because the incidence of MS decreases the further you travel from the poles toward the equator (that is, as solar radiation increases). Also, EBV clearly plays a role because patients with MS display increased titers of EBV antibodies. One study also showed that people in Antarctica excreted reactivated EBV in their saliva during winter and that vitamin D3 stopped the viral secretion.
Under these conditions, the researchers hypothesized that MS is caused by a double infection of brain cells by EBV and BMMF. EBV is reactivated by a lack of vitamin D3, and the BMMF multiply and are eventually converted into proteins. A focal immunoreaction causes the Schwann cells and oligodendrocytes to malfunction, which leads to the destruction of the myelin sheaths around the nerve fibers.
This article was translated from the Medscape German Edition. A version appeared on Medscape.com.
FDA warns about anaphylaxis after false-negative allergen tests
The Food and Drug Administration has issued a warning about the potential for patients to experience anaphylactic reactions after a negative skin test with any allergenic extract used to diagnose food allergies.
The FDA is requiring that an anaphylaxis warning after false-negative food allergen skin test results be added to the labels of these products in light of reports to the FDA’s Adverse Event Reporting System (FAERS), according to a March 3 statement.
The action follows the recognition of an increase in adverse event reports of false-negative test results with specific lots of “ALK-Abello’s Allergenic Extract-Peanut (Arachis hypogaea) – For Diagnostic Use Only.” Some of these reports “were associated with life-threatening anaphylaxis from subsequent exposure to peanut,” according to the statement. “FDA determined that the risk of anaphylaxis following false-negative food allergen skin test results is applicable to all allergenic extracts for the diagnosis of food allergies,” the statement notes.
To date, four lots of allergenic extracts have been voluntarily withdrawn from the market by the manufacturer, in November and December 2022, and should not be used.
Although some allergenic extracts are standardized, those used in the diagnosis of food allergy currently licensed by the FDA for use in the United States are nonstandardized, so potency may vary by lot.
The FDA advises health care professionals to consider confirming a negative skin test with serologic testing for peanut-specific IgE or conducting a medically supervised oral food challenge in patients, “based on the patient’s clinical history and the index of suspicion.”
The FDA also urges patients to discuss negative food allergen skin test results with their health care providers to determine the possible need for additional testing and to review the symptoms of a severe allergic reaction.
Any adverse events or side effects associated with allergenic products should be reported to the FDA via the FDA’s MedWatch Safety Information and Adverse Event Reporting Program.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has issued a warning about the potential for patients to experience anaphylactic reactions after a negative skin test with any allergenic extract used to diagnose food allergies.
The FDA is requiring that an anaphylaxis warning after false-negative food allergen skin test results be added to the labels of these products in light of reports to the FDA’s Adverse Event Reporting System (FAERS), according to a March 3 statement.
The action follows the recognition of an increase in adverse event reports of false-negative test results with specific lots of “ALK-Abello’s Allergenic Extract-Peanut (Arachis hypogaea) – For Diagnostic Use Only.” Some of these reports “were associated with life-threatening anaphylaxis from subsequent exposure to peanut,” according to the statement. “FDA determined that the risk of anaphylaxis following false-negative food allergen skin test results is applicable to all allergenic extracts for the diagnosis of food allergies,” the statement notes.
To date, four lots of allergenic extracts have been voluntarily withdrawn from the market by the manufacturer, in November and December 2022, and should not be used.
Although some allergenic extracts are standardized, those used in the diagnosis of food allergy currently licensed by the FDA for use in the United States are nonstandardized, so potency may vary by lot.
The FDA advises health care professionals to consider confirming a negative skin test with serologic testing for peanut-specific IgE or conducting a medically supervised oral food challenge in patients, “based on the patient’s clinical history and the index of suspicion.”
The FDA also urges patients to discuss negative food allergen skin test results with their health care providers to determine the possible need for additional testing and to review the symptoms of a severe allergic reaction.
Any adverse events or side effects associated with allergenic products should be reported to the FDA via the FDA’s MedWatch Safety Information and Adverse Event Reporting Program.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has issued a warning about the potential for patients to experience anaphylactic reactions after a negative skin test with any allergenic extract used to diagnose food allergies.
The FDA is requiring that an anaphylaxis warning after false-negative food allergen skin test results be added to the labels of these products in light of reports to the FDA’s Adverse Event Reporting System (FAERS), according to a March 3 statement.
The action follows the recognition of an increase in adverse event reports of false-negative test results with specific lots of “ALK-Abello’s Allergenic Extract-Peanut (Arachis hypogaea) – For Diagnostic Use Only.” Some of these reports “were associated with life-threatening anaphylaxis from subsequent exposure to peanut,” according to the statement. “FDA determined that the risk of anaphylaxis following false-negative food allergen skin test results is applicable to all allergenic extracts for the diagnosis of food allergies,” the statement notes.
To date, four lots of allergenic extracts have been voluntarily withdrawn from the market by the manufacturer, in November and December 2022, and should not be used.
Although some allergenic extracts are standardized, those used in the diagnosis of food allergy currently licensed by the FDA for use in the United States are nonstandardized, so potency may vary by lot.
The FDA advises health care professionals to consider confirming a negative skin test with serologic testing for peanut-specific IgE or conducting a medically supervised oral food challenge in patients, “based on the patient’s clinical history and the index of suspicion.”
The FDA also urges patients to discuss negative food allergen skin test results with their health care providers to determine the possible need for additional testing and to review the symptoms of a severe allergic reaction.
Any adverse events or side effects associated with allergenic products should be reported to the FDA via the FDA’s MedWatch Safety Information and Adverse Event Reporting Program.
A version of this article first appeared on Medscape.com.
Are you misdiagnosing IBS? Watch out for this mimic
Josh struggled for more than a decade with what his doctors had told him was irritable bowel syndrome (IBS). But curiously, the 39-year-old’s flare-ups were caused by some foods that aren’t typical IBS triggers.
So, Josh (not his real name) sought the care of New York gastroenterologist Yevgenia Pashinsky, MD. She conducted a comprehensive nutritional assessment and sent him for allergy testing. The results: Josh had a little-known condition called systemic nickel allergy syndrome (SNAS), which can mimic some of the symptoms of IBS.
Dr. Pashinsky, of the department of medicine at Icahn School of Medicine at Mount Sinai, New York, and a partner with New York Gastroenterology Associates, presented Josh’s case as part of a seminar on SNAS and IBS “mimickers” at the Food and Nutrition Conference and Expo in Orlando last October, sponsored by the Academy of Nutrition and Dietetics.
She and two registered dietitians in her practice, Suzie Finkel, MS, RD, CDN, and Tamara Duker Freuman, MS, RD, CDN, told seminar attendees that SNAS is rarely diagnosed and can be mistaken for IBS. They noted that it probably strikes more people than doctors suspect.
“Systemic nickel allergy is present in at least 10% of the U.S. population (and much higher in some subgroups),” Dr. Pashinsky told this news organization. “But its connection to GI symptoms and functional GI disorders is still being learned about.
“I think of nickel allergy and other allergic disorders when, in addition to GI symptoms, the patient reports skin and mucous membrane involvement along with their abdominal reactions,” she said.
For patients like Josh with SNAS, the diagnosis and treatment of this condition are surprisingly simple and effective.
“Josh had these really [unusual] symptoms and nontraditional IBS food triggers,” Ms. Finkel said in an interview. “So, that’s a situation where, as dietitians we say, ‘Hmm, that’s weird; if you have IBS, then peanuts and shrimp shouldn’t really cause an issue here.’ But this might be something physicians might not be attuned to because it’s not part of their training.”
Ms. Finkel said that Josh was referred to an allergist. Josh tested positive for skin sensitization to nickel, and he was started on a low-nickel diet, which improved his symptoms.
“So, that was this happy ending,” she added.
The upshot?
“Doctors who treat IBS patients [who are not responding to treatment] need to consider the possibility that they have SNAS and send them for allergy testing,” Ms. Finkel said. “If they come back positive, simple dietary changes can address it.”
An underrecognized condition
There has been very little research regarding SNAS in patients with IBS, and there are no standard guidelines for diagnosing and treating it.
What’s more, many gastroenterologists aren’t familiar with it. More than a dozen gastroenterologists who were contacted for comment declined to be interviewed because they didn’t know about SNAS – or enough about it to provide useful information for the story.
Ms. Finkel said she’s not surprised that many gastroenterologists don’t know much about how SNAS can mimic IBS, which is why she and her colleagues presented the seminar last October in Orlando. “It’s really an allergy and not a GI disease. It manifests with GI symptoms, but the root is not in the digestive tract; the root is in a true allergy – a clinical allergy – to nickel.”
Complicating the issue is that people who have IBS and those with SNAS typically share some common symptoms.
Like IBS, SNAS can cause GI symptoms – such as cramping, abdominal pain, heartburn, constipation, gaseous distension, and mucus in the stool. It can be triggered by certain fresh, cooked, and canned foods.
But the food triggers that cause SNAS are not usually those that cause IBS symptoms. Rather, SNAS flare-ups are nearly always triggered by foods with high levels of nickel. Examples include apricots, artichokes, asparagus, beans, cauliflower, chickpeas, cocoa/chocolate, figs, lentils, licorice, oats, onions, peas, peanuts, potatoes, spinach, tomatoes, and tea.
According to the American Academy of Allergy, Asthma & Immunology, a distinguishing feature of SNAS is that it can cause allergic contact dermatitis when a person touches something made with nickel. Coins, jewelry, eyeglasses, home fixtures, keys, zippers, dental devices, and even stainless-steel cookware can contain allergy-triggering nickel.
What Ms. Finkel sees the most are skin reactions from touching a surface containing nickel or from ingesting it, she said.
The other immediate symptom is abdominal pain or changes in bowel movements, such as diarrhea, she added.
Christopher Randolph, MD, an allergist based in Connecticut, told this news organization that it’s important for doctors to realize that patients who have a skin reaction to nickel may also have inflammatory GI symptoms.
“We definitely need more controlled studies,” said Dr. Randolph, of the department of allergy and immunology at Yale University, New Haven, Conn. “But the takeaway here is for patients and certainly providers to be mindful that you can have systemic reactions to nickel, even though you implicate only the contact dermatitis.”
Diagnosis and treatment recommendations
Skin patch allergy testing – in which a person’s skin is exposed to nickel – can quickly determine whether a patient with IBS is actually experiencing inflammatory reactions to dietary nickel and would benefit from a low-nickel or no-nickel diet, research shows.
For these patients, Dr. Pashinsky recommends the following:
- Avoiding high-nickel foods.
- Limiting canned foods.
- Using nonstainless cookware, especially for acidic foods.
- Boiling foods for potential nickel reduction, especially grains and vegetables.
- Running the tap before using water to drink or cook with first thing in the morning.
Dr. Pashisky and her team also recommend the following guidelines for doctors:
- Ask patients if symptoms occur immediately after eating certain high-nickel foods or worsen with a low-FODMAP (fermentable oligosaccharides, disaccharides, monosaccharides and polyols) diet.
- Determine whether a patient is not responding to typical medical and dietary interventions used to treat IBS.
- Conduct a food/symptom history to identify potential nickel allergy triggers.
- Try a low-nickel dietary intervention to see whether a patient’s symptoms improve in a week or two.
- Refer the patient for additional diagnostic skin-patch testing or treatment.
A multidisciplinary approach
Ms. Finkel said it’s important for doctors, particularly gastroenterologists who treat patients for suspected GI disorders to consider nickel allergy as a cause.
“SNAS is this overlooked condition ... and the research is really in its nascency here,” Ms. Finkel said.
“I would say only give [a low- or no-nickel diet] consideration if the high-nickel foods are a possible trigger,” she said. “It is very specific, looking at their diet history, to have a clear hypothesis based on what their triggers are. It’s not something to try out lightly because it’s a very restrictive diet, so I would never put a patient on a diet that I didn’t think was necessary.”
Ms. Finkel added that treatment of SNAS requires a multidisciplinary approach with a gastroenterologist, an allergist, and a dietitian.
Doctors and dietitians have distinct roles in identifying and treating these patients, Ms. Finkel said.
“If there is a suspicion of IBS symptoms and the patient is not responding to first-line treatments, then it is worth having the input of a dietitian and an allergist,” she said.
A version of this article first appeared on Medscape.com.
Josh struggled for more than a decade with what his doctors had told him was irritable bowel syndrome (IBS). But curiously, the 39-year-old’s flare-ups were caused by some foods that aren’t typical IBS triggers.
So, Josh (not his real name) sought the care of New York gastroenterologist Yevgenia Pashinsky, MD. She conducted a comprehensive nutritional assessment and sent him for allergy testing. The results: Josh had a little-known condition called systemic nickel allergy syndrome (SNAS), which can mimic some of the symptoms of IBS.
Dr. Pashinsky, of the department of medicine at Icahn School of Medicine at Mount Sinai, New York, and a partner with New York Gastroenterology Associates, presented Josh’s case as part of a seminar on SNAS and IBS “mimickers” at the Food and Nutrition Conference and Expo in Orlando last October, sponsored by the Academy of Nutrition and Dietetics.
She and two registered dietitians in her practice, Suzie Finkel, MS, RD, CDN, and Tamara Duker Freuman, MS, RD, CDN, told seminar attendees that SNAS is rarely diagnosed and can be mistaken for IBS. They noted that it probably strikes more people than doctors suspect.
“Systemic nickel allergy is present in at least 10% of the U.S. population (and much higher in some subgroups),” Dr. Pashinsky told this news organization. “But its connection to GI symptoms and functional GI disorders is still being learned about.
“I think of nickel allergy and other allergic disorders when, in addition to GI symptoms, the patient reports skin and mucous membrane involvement along with their abdominal reactions,” she said.
For patients like Josh with SNAS, the diagnosis and treatment of this condition are surprisingly simple and effective.
“Josh had these really [unusual] symptoms and nontraditional IBS food triggers,” Ms. Finkel said in an interview. “So, that’s a situation where, as dietitians we say, ‘Hmm, that’s weird; if you have IBS, then peanuts and shrimp shouldn’t really cause an issue here.’ But this might be something physicians might not be attuned to because it’s not part of their training.”
Ms. Finkel said that Josh was referred to an allergist. Josh tested positive for skin sensitization to nickel, and he was started on a low-nickel diet, which improved his symptoms.
“So, that was this happy ending,” she added.
The upshot?
“Doctors who treat IBS patients [who are not responding to treatment] need to consider the possibility that they have SNAS and send them for allergy testing,” Ms. Finkel said. “If they come back positive, simple dietary changes can address it.”
An underrecognized condition
There has been very little research regarding SNAS in patients with IBS, and there are no standard guidelines for diagnosing and treating it.
What’s more, many gastroenterologists aren’t familiar with it. More than a dozen gastroenterologists who were contacted for comment declined to be interviewed because they didn’t know about SNAS – or enough about it to provide useful information for the story.
Ms. Finkel said she’s not surprised that many gastroenterologists don’t know much about how SNAS can mimic IBS, which is why she and her colleagues presented the seminar last October in Orlando. “It’s really an allergy and not a GI disease. It manifests with GI symptoms, but the root is not in the digestive tract; the root is in a true allergy – a clinical allergy – to nickel.”
Complicating the issue is that people who have IBS and those with SNAS typically share some common symptoms.
Like IBS, SNAS can cause GI symptoms – such as cramping, abdominal pain, heartburn, constipation, gaseous distension, and mucus in the stool. It can be triggered by certain fresh, cooked, and canned foods.
But the food triggers that cause SNAS are not usually those that cause IBS symptoms. Rather, SNAS flare-ups are nearly always triggered by foods with high levels of nickel. Examples include apricots, artichokes, asparagus, beans, cauliflower, chickpeas, cocoa/chocolate, figs, lentils, licorice, oats, onions, peas, peanuts, potatoes, spinach, tomatoes, and tea.
According to the American Academy of Allergy, Asthma & Immunology, a distinguishing feature of SNAS is that it can cause allergic contact dermatitis when a person touches something made with nickel. Coins, jewelry, eyeglasses, home fixtures, keys, zippers, dental devices, and even stainless-steel cookware can contain allergy-triggering nickel.
What Ms. Finkel sees the most are skin reactions from touching a surface containing nickel or from ingesting it, she said.
The other immediate symptom is abdominal pain or changes in bowel movements, such as diarrhea, she added.
Christopher Randolph, MD, an allergist based in Connecticut, told this news organization that it’s important for doctors to realize that patients who have a skin reaction to nickel may also have inflammatory GI symptoms.
“We definitely need more controlled studies,” said Dr. Randolph, of the department of allergy and immunology at Yale University, New Haven, Conn. “But the takeaway here is for patients and certainly providers to be mindful that you can have systemic reactions to nickel, even though you implicate only the contact dermatitis.”
Diagnosis and treatment recommendations
Skin patch allergy testing – in which a person’s skin is exposed to nickel – can quickly determine whether a patient with IBS is actually experiencing inflammatory reactions to dietary nickel and would benefit from a low-nickel or no-nickel diet, research shows.
For these patients, Dr. Pashinsky recommends the following:
- Avoiding high-nickel foods.
- Limiting canned foods.
- Using nonstainless cookware, especially for acidic foods.
- Boiling foods for potential nickel reduction, especially grains and vegetables.
- Running the tap before using water to drink or cook with first thing in the morning.
Dr. Pashisky and her team also recommend the following guidelines for doctors:
- Ask patients if symptoms occur immediately after eating certain high-nickel foods or worsen with a low-FODMAP (fermentable oligosaccharides, disaccharides, monosaccharides and polyols) diet.
- Determine whether a patient is not responding to typical medical and dietary interventions used to treat IBS.
- Conduct a food/symptom history to identify potential nickel allergy triggers.
- Try a low-nickel dietary intervention to see whether a patient’s symptoms improve in a week or two.
- Refer the patient for additional diagnostic skin-patch testing or treatment.
A multidisciplinary approach
Ms. Finkel said it’s important for doctors, particularly gastroenterologists who treat patients for suspected GI disorders to consider nickel allergy as a cause.
“SNAS is this overlooked condition ... and the research is really in its nascency here,” Ms. Finkel said.
“I would say only give [a low- or no-nickel diet] consideration if the high-nickel foods are a possible trigger,” she said. “It is very specific, looking at their diet history, to have a clear hypothesis based on what their triggers are. It’s not something to try out lightly because it’s a very restrictive diet, so I would never put a patient on a diet that I didn’t think was necessary.”
Ms. Finkel added that treatment of SNAS requires a multidisciplinary approach with a gastroenterologist, an allergist, and a dietitian.
Doctors and dietitians have distinct roles in identifying and treating these patients, Ms. Finkel said.
“If there is a suspicion of IBS symptoms and the patient is not responding to first-line treatments, then it is worth having the input of a dietitian and an allergist,” she said.
A version of this article first appeared on Medscape.com.
Josh struggled for more than a decade with what his doctors had told him was irritable bowel syndrome (IBS). But curiously, the 39-year-old’s flare-ups were caused by some foods that aren’t typical IBS triggers.
So, Josh (not his real name) sought the care of New York gastroenterologist Yevgenia Pashinsky, MD. She conducted a comprehensive nutritional assessment and sent him for allergy testing. The results: Josh had a little-known condition called systemic nickel allergy syndrome (SNAS), which can mimic some of the symptoms of IBS.
Dr. Pashinsky, of the department of medicine at Icahn School of Medicine at Mount Sinai, New York, and a partner with New York Gastroenterology Associates, presented Josh’s case as part of a seminar on SNAS and IBS “mimickers” at the Food and Nutrition Conference and Expo in Orlando last October, sponsored by the Academy of Nutrition and Dietetics.
She and two registered dietitians in her practice, Suzie Finkel, MS, RD, CDN, and Tamara Duker Freuman, MS, RD, CDN, told seminar attendees that SNAS is rarely diagnosed and can be mistaken for IBS. They noted that it probably strikes more people than doctors suspect.
“Systemic nickel allergy is present in at least 10% of the U.S. population (and much higher in some subgroups),” Dr. Pashinsky told this news organization. “But its connection to GI symptoms and functional GI disorders is still being learned about.
“I think of nickel allergy and other allergic disorders when, in addition to GI symptoms, the patient reports skin and mucous membrane involvement along with their abdominal reactions,” she said.
For patients like Josh with SNAS, the diagnosis and treatment of this condition are surprisingly simple and effective.
“Josh had these really [unusual] symptoms and nontraditional IBS food triggers,” Ms. Finkel said in an interview. “So, that’s a situation where, as dietitians we say, ‘Hmm, that’s weird; if you have IBS, then peanuts and shrimp shouldn’t really cause an issue here.’ But this might be something physicians might not be attuned to because it’s not part of their training.”
Ms. Finkel said that Josh was referred to an allergist. Josh tested positive for skin sensitization to nickel, and he was started on a low-nickel diet, which improved his symptoms.
“So, that was this happy ending,” she added.
The upshot?
“Doctors who treat IBS patients [who are not responding to treatment] need to consider the possibility that they have SNAS and send them for allergy testing,” Ms. Finkel said. “If they come back positive, simple dietary changes can address it.”
An underrecognized condition
There has been very little research regarding SNAS in patients with IBS, and there are no standard guidelines for diagnosing and treating it.
What’s more, many gastroenterologists aren’t familiar with it. More than a dozen gastroenterologists who were contacted for comment declined to be interviewed because they didn’t know about SNAS – or enough about it to provide useful information for the story.
Ms. Finkel said she’s not surprised that many gastroenterologists don’t know much about how SNAS can mimic IBS, which is why she and her colleagues presented the seminar last October in Orlando. “It’s really an allergy and not a GI disease. It manifests with GI symptoms, but the root is not in the digestive tract; the root is in a true allergy – a clinical allergy – to nickel.”
Complicating the issue is that people who have IBS and those with SNAS typically share some common symptoms.
Like IBS, SNAS can cause GI symptoms – such as cramping, abdominal pain, heartburn, constipation, gaseous distension, and mucus in the stool. It can be triggered by certain fresh, cooked, and canned foods.
But the food triggers that cause SNAS are not usually those that cause IBS symptoms. Rather, SNAS flare-ups are nearly always triggered by foods with high levels of nickel. Examples include apricots, artichokes, asparagus, beans, cauliflower, chickpeas, cocoa/chocolate, figs, lentils, licorice, oats, onions, peas, peanuts, potatoes, spinach, tomatoes, and tea.
According to the American Academy of Allergy, Asthma & Immunology, a distinguishing feature of SNAS is that it can cause allergic contact dermatitis when a person touches something made with nickel. Coins, jewelry, eyeglasses, home fixtures, keys, zippers, dental devices, and even stainless-steel cookware can contain allergy-triggering nickel.
What Ms. Finkel sees the most are skin reactions from touching a surface containing nickel or from ingesting it, she said.
The other immediate symptom is abdominal pain or changes in bowel movements, such as diarrhea, she added.
Christopher Randolph, MD, an allergist based in Connecticut, told this news organization that it’s important for doctors to realize that patients who have a skin reaction to nickel may also have inflammatory GI symptoms.
“We definitely need more controlled studies,” said Dr. Randolph, of the department of allergy and immunology at Yale University, New Haven, Conn. “But the takeaway here is for patients and certainly providers to be mindful that you can have systemic reactions to nickel, even though you implicate only the contact dermatitis.”
Diagnosis and treatment recommendations
Skin patch allergy testing – in which a person’s skin is exposed to nickel – can quickly determine whether a patient with IBS is actually experiencing inflammatory reactions to dietary nickel and would benefit from a low-nickel or no-nickel diet, research shows.
For these patients, Dr. Pashinsky recommends the following:
- Avoiding high-nickel foods.
- Limiting canned foods.
- Using nonstainless cookware, especially for acidic foods.
- Boiling foods for potential nickel reduction, especially grains and vegetables.
- Running the tap before using water to drink or cook with first thing in the morning.
Dr. Pashisky and her team also recommend the following guidelines for doctors:
- Ask patients if symptoms occur immediately after eating certain high-nickel foods or worsen with a low-FODMAP (fermentable oligosaccharides, disaccharides, monosaccharides and polyols) diet.
- Determine whether a patient is not responding to typical medical and dietary interventions used to treat IBS.
- Conduct a food/symptom history to identify potential nickel allergy triggers.
- Try a low-nickel dietary intervention to see whether a patient’s symptoms improve in a week or two.
- Refer the patient for additional diagnostic skin-patch testing or treatment.
A multidisciplinary approach
Ms. Finkel said it’s important for doctors, particularly gastroenterologists who treat patients for suspected GI disorders to consider nickel allergy as a cause.
“SNAS is this overlooked condition ... and the research is really in its nascency here,” Ms. Finkel said.
“I would say only give [a low- or no-nickel diet] consideration if the high-nickel foods are a possible trigger,” she said. “It is very specific, looking at their diet history, to have a clear hypothesis based on what their triggers are. It’s not something to try out lightly because it’s a very restrictive diet, so I would never put a patient on a diet that I didn’t think was necessary.”
Ms. Finkel added that treatment of SNAS requires a multidisciplinary approach with a gastroenterologist, an allergist, and a dietitian.
Doctors and dietitians have distinct roles in identifying and treating these patients, Ms. Finkel said.
“If there is a suspicion of IBS symptoms and the patient is not responding to first-line treatments, then it is worth having the input of a dietitian and an allergist,” she said.
A version of this article first appeared on Medscape.com.
In utero exposure to asthma medication not tied to risks of neurodevelopmental disorders
The drugs included in the study were leukotriene-receptor antagonists (LTRAs), which are often used to treat allergic airway diseases, including asthma and allergic rhinitis.
“Over the years, the U.S. Food and Drug Administration has monitored post-marketing data about the potential harm of neuropsychiatric events (NEs) associated with montelukast, the first type of LTRAs, and issued boxed warnings about serious mental health side effects for montelukast in 2020,” said corresponding author Tsung-Chieh Yao, MD, of Chang Gung Memorial Hospital, Taiwan, in an interview.
However, evidence of a link between NEs and LTRA use has been inconsistent, according to Dr. Yao and colleagues.
“To date, it remains totally unknown whether the exposure to LTRAs during pregnancy is associated with the risk of neuropsychiatric events in offspring,” said Dr. Yao.
To address this question, the researchers used data from National Health Insurance Research Database in Taiwan to identify pregnant women and their offspring from 2009 to 2019. The initial study population included 576,157 mother-offspring pairs, including 1,995 LTRA-exposed and 574,162 nonexposed children.
The women had a diagnosis of asthma or allergic rhinitis; multiple births and children with congenital malformations were excluded. LTRA exposure was defined as any dispensed prescription for LTRAs during pregnancy. Approximately two-thirds of the mothers were aged 30-40 years at the time of delivery.
The findings were published in a research letter in JAMA Network Open.
In the study population at large, the incidence of the three neurodevelopmental disorders ADHD, autism spectrum disorder (ASD), and Tourette syndrome was not significantly different between those children exposed to LTRAs and those not exposed to LTRAs in utero (1.25% vs. 1.32%; 3.31% vs. 4.36%; and 0.45% vs. 0.83%, respectively).
After propensity score matching, the study population included 1,988 LTRA-exposed children and 19,863 nonexposed children. In this group, no significant associations appeared between prenatal LTRA exposure and the risk of attention-deficit/hyperactivity disorder (adjusted hazard ratio, 1.03), autism spectrum disorder (AHR, 1.01), and Tourette syndrome (AHR, 0.63).
Neither duration nor cumulative dose of LTRA use during pregnancy showed an association with ADHD, ASD, or Tourette syndrome in offspring. Duration of LTRA use was categorized as shorter or longer periods of 1-4 weeks vs. more than 4 weeks; cumulative dose was categorized as 1-170 mg vs. 170 mg or higher.
The findings were limited by the lack of randomization, inability to detect long-term risk, and potential lack of generalizability to non-Asian populations, and more research is needed to replicate the results, the researchers noted. However, the current findings were strengthened by the large study population, and suggest that LTRA use in pregnancy does not present a significant risk for NEs in children, which should be reassuring to clinicians and patients, they concluded.
The current study is the first to use the whole of Taiwan population data and extends previous studies by examining the association between LTRA use during pregnancy and risk of neuropsychiatric events in offspring, Dr. Yao said in an interview. “The possibly surprising, but reassuring, finding is that prenatal LTRA exposure did not increase risk of ADHD, ASD, and Tourette syndrome in offspring,” he said.
“Clinicians prescribing LTRAs such as montelukast (Singulair and generics) to pregnant women with asthma or allergic rhinitis may be reassured by our findings,” Dr. Yao added. The results offer real-world evidence to help inform decision-making about the use of LTRAs during pregnancy, although additional research is needed to replicate the study findings in other populations, he said.
The study was supported by the National Health Research Institutes, Taiwan, the Ministry of Science and Technology of Taiwan, the National Science and Technology Council of Taiwan, and the Chang Gung Medical Foundation. The researchers had no financial conflicts to disclose.
The drugs included in the study were leukotriene-receptor antagonists (LTRAs), which are often used to treat allergic airway diseases, including asthma and allergic rhinitis.
“Over the years, the U.S. Food and Drug Administration has monitored post-marketing data about the potential harm of neuropsychiatric events (NEs) associated with montelukast, the first type of LTRAs, and issued boxed warnings about serious mental health side effects for montelukast in 2020,” said corresponding author Tsung-Chieh Yao, MD, of Chang Gung Memorial Hospital, Taiwan, in an interview.
However, evidence of a link between NEs and LTRA use has been inconsistent, according to Dr. Yao and colleagues.
“To date, it remains totally unknown whether the exposure to LTRAs during pregnancy is associated with the risk of neuropsychiatric events in offspring,” said Dr. Yao.
To address this question, the researchers used data from National Health Insurance Research Database in Taiwan to identify pregnant women and their offspring from 2009 to 2019. The initial study population included 576,157 mother-offspring pairs, including 1,995 LTRA-exposed and 574,162 nonexposed children.
The women had a diagnosis of asthma or allergic rhinitis; multiple births and children with congenital malformations were excluded. LTRA exposure was defined as any dispensed prescription for LTRAs during pregnancy. Approximately two-thirds of the mothers were aged 30-40 years at the time of delivery.
The findings were published in a research letter in JAMA Network Open.
In the study population at large, the incidence of the three neurodevelopmental disorders ADHD, autism spectrum disorder (ASD), and Tourette syndrome was not significantly different between those children exposed to LTRAs and those not exposed to LTRAs in utero (1.25% vs. 1.32%; 3.31% vs. 4.36%; and 0.45% vs. 0.83%, respectively).
After propensity score matching, the study population included 1,988 LTRA-exposed children and 19,863 nonexposed children. In this group, no significant associations appeared between prenatal LTRA exposure and the risk of attention-deficit/hyperactivity disorder (adjusted hazard ratio, 1.03), autism spectrum disorder (AHR, 1.01), and Tourette syndrome (AHR, 0.63).
Neither duration nor cumulative dose of LTRA use during pregnancy showed an association with ADHD, ASD, or Tourette syndrome in offspring. Duration of LTRA use was categorized as shorter or longer periods of 1-4 weeks vs. more than 4 weeks; cumulative dose was categorized as 1-170 mg vs. 170 mg or higher.
The findings were limited by the lack of randomization, inability to detect long-term risk, and potential lack of generalizability to non-Asian populations, and more research is needed to replicate the results, the researchers noted. However, the current findings were strengthened by the large study population, and suggest that LTRA use in pregnancy does not present a significant risk for NEs in children, which should be reassuring to clinicians and patients, they concluded.
The current study is the first to use the whole of Taiwan population data and extends previous studies by examining the association between LTRA use during pregnancy and risk of neuropsychiatric events in offspring, Dr. Yao said in an interview. “The possibly surprising, but reassuring, finding is that prenatal LTRA exposure did not increase risk of ADHD, ASD, and Tourette syndrome in offspring,” he said.
“Clinicians prescribing LTRAs such as montelukast (Singulair and generics) to pregnant women with asthma or allergic rhinitis may be reassured by our findings,” Dr. Yao added. The results offer real-world evidence to help inform decision-making about the use of LTRAs during pregnancy, although additional research is needed to replicate the study findings in other populations, he said.
The study was supported by the National Health Research Institutes, Taiwan, the Ministry of Science and Technology of Taiwan, the National Science and Technology Council of Taiwan, and the Chang Gung Medical Foundation. The researchers had no financial conflicts to disclose.
The drugs included in the study were leukotriene-receptor antagonists (LTRAs), which are often used to treat allergic airway diseases, including asthma and allergic rhinitis.
“Over the years, the U.S. Food and Drug Administration has monitored post-marketing data about the potential harm of neuropsychiatric events (NEs) associated with montelukast, the first type of LTRAs, and issued boxed warnings about serious mental health side effects for montelukast in 2020,” said corresponding author Tsung-Chieh Yao, MD, of Chang Gung Memorial Hospital, Taiwan, in an interview.
However, evidence of a link between NEs and LTRA use has been inconsistent, according to Dr. Yao and colleagues.
“To date, it remains totally unknown whether the exposure to LTRAs during pregnancy is associated with the risk of neuropsychiatric events in offspring,” said Dr. Yao.
To address this question, the researchers used data from National Health Insurance Research Database in Taiwan to identify pregnant women and their offspring from 2009 to 2019. The initial study population included 576,157 mother-offspring pairs, including 1,995 LTRA-exposed and 574,162 nonexposed children.
The women had a diagnosis of asthma or allergic rhinitis; multiple births and children with congenital malformations were excluded. LTRA exposure was defined as any dispensed prescription for LTRAs during pregnancy. Approximately two-thirds of the mothers were aged 30-40 years at the time of delivery.
The findings were published in a research letter in JAMA Network Open.
In the study population at large, the incidence of the three neurodevelopmental disorders ADHD, autism spectrum disorder (ASD), and Tourette syndrome was not significantly different between those children exposed to LTRAs and those not exposed to LTRAs in utero (1.25% vs. 1.32%; 3.31% vs. 4.36%; and 0.45% vs. 0.83%, respectively).
After propensity score matching, the study population included 1,988 LTRA-exposed children and 19,863 nonexposed children. In this group, no significant associations appeared between prenatal LTRA exposure and the risk of attention-deficit/hyperactivity disorder (adjusted hazard ratio, 1.03), autism spectrum disorder (AHR, 1.01), and Tourette syndrome (AHR, 0.63).
Neither duration nor cumulative dose of LTRA use during pregnancy showed an association with ADHD, ASD, or Tourette syndrome in offspring. Duration of LTRA use was categorized as shorter or longer periods of 1-4 weeks vs. more than 4 weeks; cumulative dose was categorized as 1-170 mg vs. 170 mg or higher.
The findings were limited by the lack of randomization, inability to detect long-term risk, and potential lack of generalizability to non-Asian populations, and more research is needed to replicate the results, the researchers noted. However, the current findings were strengthened by the large study population, and suggest that LTRA use in pregnancy does not present a significant risk for NEs in children, which should be reassuring to clinicians and patients, they concluded.
The current study is the first to use the whole of Taiwan population data and extends previous studies by examining the association between LTRA use during pregnancy and risk of neuropsychiatric events in offspring, Dr. Yao said in an interview. “The possibly surprising, but reassuring, finding is that prenatal LTRA exposure did not increase risk of ADHD, ASD, and Tourette syndrome in offspring,” he said.
“Clinicians prescribing LTRAs such as montelukast (Singulair and generics) to pregnant women with asthma or allergic rhinitis may be reassured by our findings,” Dr. Yao added. The results offer real-world evidence to help inform decision-making about the use of LTRAs during pregnancy, although additional research is needed to replicate the study findings in other populations, he said.
The study was supported by the National Health Research Institutes, Taiwan, the Ministry of Science and Technology of Taiwan, the National Science and Technology Council of Taiwan, and the Chang Gung Medical Foundation. The researchers had no financial conflicts to disclose.
FROM JAMA NETWORK OPEN
Painful blue fingers
A 48-year-old woman with a history of systemic lupus erythematosus (SLE) presented to the emergency department from the rheumatology clinic for digital ischemia. The clinical manifestations of her SLE consisted predominantly of arthralgias, which had been previously well controlled on hydroxychloroquine 300 mg/d PO. On presentation, she denied oral ulcers, alopecia, shortness of breath, chest pain/pressure, and history of blood clots or miscarriages.
On exam, the patient was afebrile and had a heart rate of 74 bpm; blood pressure, 140/77 mm Hg; and respiratory rate, 18 breaths/min. The fingertips on her left hand were tender and cool to the touch, and the fingertips of her second through fifth digits were blue (FIGURE).
Laboratory workup was notable for the following: hemoglobin, 9.3 g/dL (normal range, 11.6-15.2 g/dL) and erythrocyte sedimentation rate, 44 mm/h (normal range, ≤ 25 mm/h). Double-stranded DNA and complement levels were normal.
Transthoracic echocardiogram did not show any valvular vegetations, and blood cultures from admission were negative. Computed tomography angiography (CTA) with contrast of her left upper extremity showed a filling defect in the origin of the left subclavian artery. Digital plethysmography showed dampened flow signals in the second through fifth digits of the left hand.
Tests for antiphospholipid antibodies were positive for lupus anticoagulant; there were also high titers of anti-β-2-glycoprotein immunoglobulin (Ig) G (58 SGU; normal, ≤ 20 SGU) and anticardiolipin IgG (242.4 CU; normal, ≤ 20 CU).
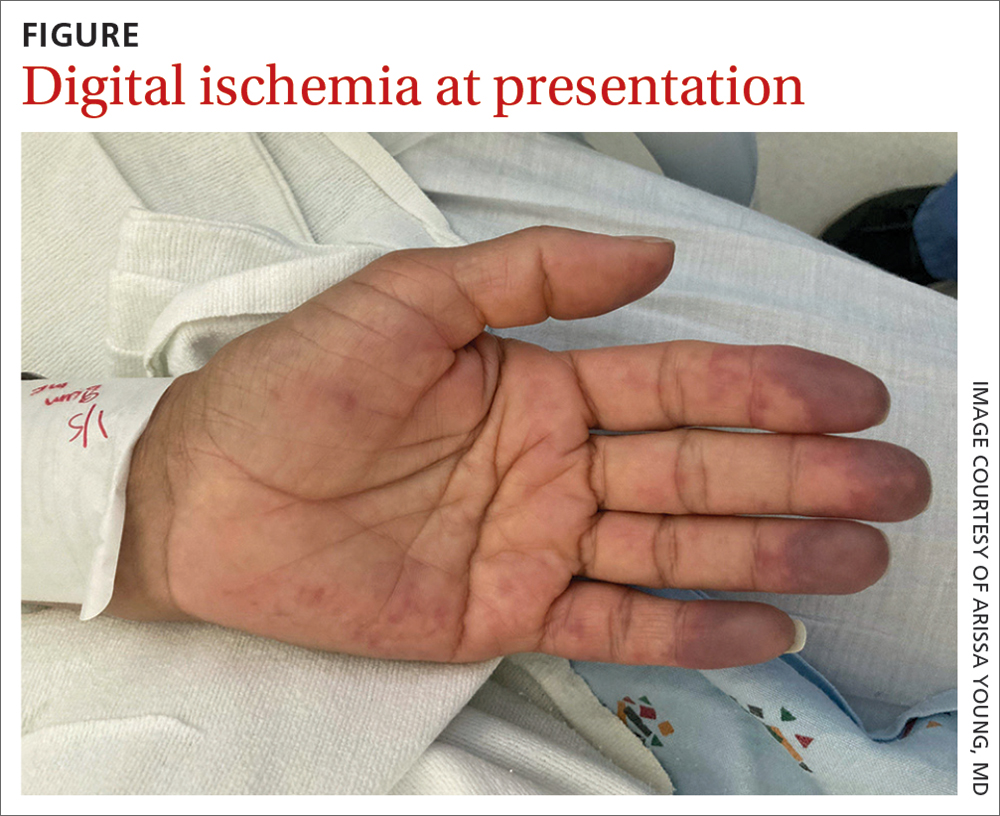
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Antiphospholipid syndrome
Given our patient’s SLE, left subclavian artery thrombosis, digital ischemia, and high-titer antiphospholipid antibodies, we had significant concern for antiphospholipid syndrome (APS). The diagnosis of APS is most often based on the fulfillment of the revised Sapporo classification criteria. These criteria include both clinical criteria (vascular thrombosis or pregnancy morbidity) and laboratory criteria (the presence of antiphospholipid antibodies on at least 2 separate occasions separated by 12 weeks).1 Our patient met clinical criteria given the evidence of subclavian artery thrombosis on CTA as well as digital plethysmography findings consistent with digital emboli. To meet laboratory criteria, she would have needed to have persistent high-titer antiphospholipid antibodies 12 weeks apart.
APS is an autoimmune disease in which the presence of antiphospholipid antibodies is associated with thrombosis; it can be divided into primary and secondary APS. The estimated prevalence of APS is 50 per 100,000 people in the United States.2 Primary APS occurs in the absence of an underlying autoimmune disease, while secondary APS occurs in the presence of an underlying autoimmune disease.
The autoimmune disease most often associated with APS is SLE.3 Among patients with SLE, 15% to 34% have positive lupus anticoagulant and 12% to 30% have anticardiolipin antibodies.4-6 This is compared with young healthy subjects among whom only 1% to 4% have positive lupus anticoagulant and 1% to 5% have anticardiolipin antibodies.7 Previous studies have estimated that 30% to 50% of patients with SLE who test positive for antiphospholipid antibodies will develop thrombosis.5,7
Differential includes Raynaud phenomenon, vasculitis
The differential diagnosis for digital ischemia in a patient with SLE includes APS, Raynaud phenomenon, vasculitis, and septic emboli.
Raynaud phenomenon can manifest in patients with SLE, but the presence of thrombosis on CTA and high-titer positive antiphospholipid antibodies make the diagnosis of APS more likely. Additionally, Raynaud phenomenon is typically temperature dependent with vasospasm in the digital arteries, occurring in cold temperatures and resolving with warming.
Systemic vasculitis may develop in patients with SLE, but in our case was less likely given the patient did not have any evidence of vasculitis on CTA, such as blood vessel wall thickening and/or enhancement.8
Septic emboli from endocarditis can cause digital ischemia but is typically associated with positive blood cultures, fever, and other systemic signs of infection, and/or vegetations on an echocardiogram.
Continue to: Thrombosis determines intensity of lifelong antiocagulation Tx
Thrombosis determines intensity of lifelong anticoagulation Tx
The mainstay of therapy for patients with APS is lifelong anticoagulation with a vitamin K antagonist. The intensity of anticoagulation is determined based on the presence of venous or arterial thrombosis. In patients who present with arterial thrombosis, a higher intensity vitamin K antagonist (ie, international normalized ratio [INR] goal > 3) or the addition of low-dose aspirin should be considered.9,10
Factor Xa inhibitors are generally not recommended at this time due to the lack of evidence to support their use.10 Additionally, a randomized clinical trial comparing rivaroxaban and warfarin in patients with triple antiphospholipid antibody positivity was terminated prematurely due to increased thromboembolic events in the rivaroxaban arm.11
For patients with secondary APS in the setting of SLE, hydroxychloroquine in combination with a vitamin K antagonist has been shown to decrease the risk for recurrent thrombosis compared with treatment with a vitamin K antagonist alone.12
Our patient was started on a heparin drip and transitioned to an oral vitamin K antagonist with an INR goal of 2 to 3. Lifelong anticoagulation was planned. The pain and discoloration in her hands improved on anticoagulation and had nearly resolved by the time of discharge. Given her history of arterial thrombosis, the addition of aspirin was also considered, but this decision was ultimately deferred to her outpatient rheumatologist and hematologist.
1. Miyakis S, Lockshin MD, Atsumi T, et al. International consensus statement on an update of the classification criteria for definite antiphospholipid syndrome (APS). J Thromb Haemost. 2006;4:295-306. doi: 10.1111/j.1538-7836.2006.01753.x
2. Duarte-García A, Pham MM, Crowson CS, et al. The epidemiology of antiphospholipid syndrome: a population-based study. Arthritis Rheumatol. 2019;71:1545-1552. doi: 10.1002/art.40901
3. Levine JS, Branch DW, Rauch J. The antiphospholipid syndrome. N Engl J Med. 2002;346:752-763. doi: 10.1056/NEJMra002974
4. Cervera R, Khamashta MA, Font J, et al. Systemic lupus erythematosus: clinical and immunologic patterns of disease expression in a cohort of 1,000 patients. Medicine (Baltimore). 1993;72:113-124.
5. Love PE, Santoro SA. Antiphospholipid antibodies: anticardiolipin and the lupus anticoagulant in systemic lupus erythematosus (SLE) and in non-SLE disorders: prevalence and clinical significance. Ann Intern Med. 1990;112:682-698. doi: 10.7326/0003-4819-112-9-682
6. Merkel PA, Chang YC, Pierangeli SS, et al. The prevalence and clinical associations of anticardiolipin antibodies in a large inception cohort of patients with connective tissue diseases. Am J Med. 1996;101:576-583. doi: 10.1016/s0002-9343(96)00335-x
7. Petri M. Epidemiology of the antiphospholipid antibody syndrome. J Autoimmun. 2000;15:145-151. doi: 10.1006/jaut. 2000.0409
8. Bozlar U, Ogur T, Khaja MS, et al. CT angiography of the upper extremity arterial system: Part 2—Clinical applications beyond trauma patients. AJR Am J Roentgenol. 2013;201:753-763. doi: 10.2214/AJR.13.11208
9. Ruiz-Irastorza G, Hunt BJ, Khamashta MA. A systematic review of secondary thromboprophylaxis in patients with antiphospholipid antibodies. Arthritis Rheum. 2007;7:1487-1495. doi: 10.1002/art.23109
10. Tektonidou MG, Andreoli L, Limper M, et al. EULAR recommendations for the management of antiphospholipid syndrome in adults. Ann Rheum Dis. 2019;78:1296-1304. doi: 10.1136/annrheumdis-2019-215213
Efficacy and safety of rivaroxaban vs warfarin in high-risk patients with antiphospholipid syndrome: rationale and design of the Trial on Rivaroxaban in AntiPhospholipid Syndrome (TRAPS) trial. Lupus. 2016;25:301-306. doi: 10.1177/0961203315611495
12. Schmidt-Tanguy A, Voswinkel J, Henrion D, et al. Antithrombotic effects of hydroxychloroquine in primary antiphospholipid syndrome patients. J Thromb Haemost. 2013;11:1927-1929. doi: 10.1111/jth.12363
A 48-year-old woman with a history of systemic lupus erythematosus (SLE) presented to the emergency department from the rheumatology clinic for digital ischemia. The clinical manifestations of her SLE consisted predominantly of arthralgias, which had been previously well controlled on hydroxychloroquine 300 mg/d PO. On presentation, she denied oral ulcers, alopecia, shortness of breath, chest pain/pressure, and history of blood clots or miscarriages.
On exam, the patient was afebrile and had a heart rate of 74 bpm; blood pressure, 140/77 mm Hg; and respiratory rate, 18 breaths/min. The fingertips on her left hand were tender and cool to the touch, and the fingertips of her second through fifth digits were blue (FIGURE).
Laboratory workup was notable for the following: hemoglobin, 9.3 g/dL (normal range, 11.6-15.2 g/dL) and erythrocyte sedimentation rate, 44 mm/h (normal range, ≤ 25 mm/h). Double-stranded DNA and complement levels were normal.
Transthoracic echocardiogram did not show any valvular vegetations, and blood cultures from admission were negative. Computed tomography angiography (CTA) with contrast of her left upper extremity showed a filling defect in the origin of the left subclavian artery. Digital plethysmography showed dampened flow signals in the second through fifth digits of the left hand.
Tests for antiphospholipid antibodies were positive for lupus anticoagulant; there were also high titers of anti-β-2-glycoprotein immunoglobulin (Ig) G (58 SGU; normal, ≤ 20 SGU) and anticardiolipin IgG (242.4 CU; normal, ≤ 20 CU).

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Antiphospholipid syndrome
Given our patient’s SLE, left subclavian artery thrombosis, digital ischemia, and high-titer antiphospholipid antibodies, we had significant concern for antiphospholipid syndrome (APS). The diagnosis of APS is most often based on the fulfillment of the revised Sapporo classification criteria. These criteria include both clinical criteria (vascular thrombosis or pregnancy morbidity) and laboratory criteria (the presence of antiphospholipid antibodies on at least 2 separate occasions separated by 12 weeks).1 Our patient met clinical criteria given the evidence of subclavian artery thrombosis on CTA as well as digital plethysmography findings consistent with digital emboli. To meet laboratory criteria, she would have needed to have persistent high-titer antiphospholipid antibodies 12 weeks apart.
APS is an autoimmune disease in which the presence of antiphospholipid antibodies is associated with thrombosis; it can be divided into primary and secondary APS. The estimated prevalence of APS is 50 per 100,000 people in the United States.2 Primary APS occurs in the absence of an underlying autoimmune disease, while secondary APS occurs in the presence of an underlying autoimmune disease.
The autoimmune disease most often associated with APS is SLE.3 Among patients with SLE, 15% to 34% have positive lupus anticoagulant and 12% to 30% have anticardiolipin antibodies.4-6 This is compared with young healthy subjects among whom only 1% to 4% have positive lupus anticoagulant and 1% to 5% have anticardiolipin antibodies.7 Previous studies have estimated that 30% to 50% of patients with SLE who test positive for antiphospholipid antibodies will develop thrombosis.5,7
Differential includes Raynaud phenomenon, vasculitis
The differential diagnosis for digital ischemia in a patient with SLE includes APS, Raynaud phenomenon, vasculitis, and septic emboli.
Raynaud phenomenon can manifest in patients with SLE, but the presence of thrombosis on CTA and high-titer positive antiphospholipid antibodies make the diagnosis of APS more likely. Additionally, Raynaud phenomenon is typically temperature dependent with vasospasm in the digital arteries, occurring in cold temperatures and resolving with warming.
Systemic vasculitis may develop in patients with SLE, but in our case was less likely given the patient did not have any evidence of vasculitis on CTA, such as blood vessel wall thickening and/or enhancement.8
Septic emboli from endocarditis can cause digital ischemia but is typically associated with positive blood cultures, fever, and other systemic signs of infection, and/or vegetations on an echocardiogram.
Continue to: Thrombosis determines intensity of lifelong antiocagulation Tx
Thrombosis determines intensity of lifelong anticoagulation Tx
The mainstay of therapy for patients with APS is lifelong anticoagulation with a vitamin K antagonist. The intensity of anticoagulation is determined based on the presence of venous or arterial thrombosis. In patients who present with arterial thrombosis, a higher intensity vitamin K antagonist (ie, international normalized ratio [INR] goal > 3) or the addition of low-dose aspirin should be considered.9,10
Factor Xa inhibitors are generally not recommended at this time due to the lack of evidence to support their use.10 Additionally, a randomized clinical trial comparing rivaroxaban and warfarin in patients with triple antiphospholipid antibody positivity was terminated prematurely due to increased thromboembolic events in the rivaroxaban arm.11
For patients with secondary APS in the setting of SLE, hydroxychloroquine in combination with a vitamin K antagonist has been shown to decrease the risk for recurrent thrombosis compared with treatment with a vitamin K antagonist alone.12
Our patient was started on a heparin drip and transitioned to an oral vitamin K antagonist with an INR goal of 2 to 3. Lifelong anticoagulation was planned. The pain and discoloration in her hands improved on anticoagulation and had nearly resolved by the time of discharge. Given her history of arterial thrombosis, the addition of aspirin was also considered, but this decision was ultimately deferred to her outpatient rheumatologist and hematologist.
A 48-year-old woman with a history of systemic lupus erythematosus (SLE) presented to the emergency department from the rheumatology clinic for digital ischemia. The clinical manifestations of her SLE consisted predominantly of arthralgias, which had been previously well controlled on hydroxychloroquine 300 mg/d PO. On presentation, she denied oral ulcers, alopecia, shortness of breath, chest pain/pressure, and history of blood clots or miscarriages.
On exam, the patient was afebrile and had a heart rate of 74 bpm; blood pressure, 140/77 mm Hg; and respiratory rate, 18 breaths/min. The fingertips on her left hand were tender and cool to the touch, and the fingertips of her second through fifth digits were blue (FIGURE).
Laboratory workup was notable for the following: hemoglobin, 9.3 g/dL (normal range, 11.6-15.2 g/dL) and erythrocyte sedimentation rate, 44 mm/h (normal range, ≤ 25 mm/h). Double-stranded DNA and complement levels were normal.
Transthoracic echocardiogram did not show any valvular vegetations, and blood cultures from admission were negative. Computed tomography angiography (CTA) with contrast of her left upper extremity showed a filling defect in the origin of the left subclavian artery. Digital plethysmography showed dampened flow signals in the second through fifth digits of the left hand.
Tests for antiphospholipid antibodies were positive for lupus anticoagulant; there were also high titers of anti-β-2-glycoprotein immunoglobulin (Ig) G (58 SGU; normal, ≤ 20 SGU) and anticardiolipin IgG (242.4 CU; normal, ≤ 20 CU).

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Antiphospholipid syndrome
Given our patient’s SLE, left subclavian artery thrombosis, digital ischemia, and high-titer antiphospholipid antibodies, we had significant concern for antiphospholipid syndrome (APS). The diagnosis of APS is most often based on the fulfillment of the revised Sapporo classification criteria. These criteria include both clinical criteria (vascular thrombosis or pregnancy morbidity) and laboratory criteria (the presence of antiphospholipid antibodies on at least 2 separate occasions separated by 12 weeks).1 Our patient met clinical criteria given the evidence of subclavian artery thrombosis on CTA as well as digital plethysmography findings consistent with digital emboli. To meet laboratory criteria, she would have needed to have persistent high-titer antiphospholipid antibodies 12 weeks apart.
APS is an autoimmune disease in which the presence of antiphospholipid antibodies is associated with thrombosis; it can be divided into primary and secondary APS. The estimated prevalence of APS is 50 per 100,000 people in the United States.2 Primary APS occurs in the absence of an underlying autoimmune disease, while secondary APS occurs in the presence of an underlying autoimmune disease.
The autoimmune disease most often associated with APS is SLE.3 Among patients with SLE, 15% to 34% have positive lupus anticoagulant and 12% to 30% have anticardiolipin antibodies.4-6 This is compared with young healthy subjects among whom only 1% to 4% have positive lupus anticoagulant and 1% to 5% have anticardiolipin antibodies.7 Previous studies have estimated that 30% to 50% of patients with SLE who test positive for antiphospholipid antibodies will develop thrombosis.5,7
Differential includes Raynaud phenomenon, vasculitis
The differential diagnosis for digital ischemia in a patient with SLE includes APS, Raynaud phenomenon, vasculitis, and septic emboli.
Raynaud phenomenon can manifest in patients with SLE, but the presence of thrombosis on CTA and high-titer positive antiphospholipid antibodies make the diagnosis of APS more likely. Additionally, Raynaud phenomenon is typically temperature dependent with vasospasm in the digital arteries, occurring in cold temperatures and resolving with warming.
Systemic vasculitis may develop in patients with SLE, but in our case was less likely given the patient did not have any evidence of vasculitis on CTA, such as blood vessel wall thickening and/or enhancement.8
Septic emboli from endocarditis can cause digital ischemia but is typically associated with positive blood cultures, fever, and other systemic signs of infection, and/or vegetations on an echocardiogram.
Continue to: Thrombosis determines intensity of lifelong antiocagulation Tx
Thrombosis determines intensity of lifelong anticoagulation Tx
The mainstay of therapy for patients with APS is lifelong anticoagulation with a vitamin K antagonist. The intensity of anticoagulation is determined based on the presence of venous or arterial thrombosis. In patients who present with arterial thrombosis, a higher intensity vitamin K antagonist (ie, international normalized ratio [INR] goal > 3) or the addition of low-dose aspirin should be considered.9,10
Factor Xa inhibitors are generally not recommended at this time due to the lack of evidence to support their use.10 Additionally, a randomized clinical trial comparing rivaroxaban and warfarin in patients with triple antiphospholipid antibody positivity was terminated prematurely due to increased thromboembolic events in the rivaroxaban arm.11
For patients with secondary APS in the setting of SLE, hydroxychloroquine in combination with a vitamin K antagonist has been shown to decrease the risk for recurrent thrombosis compared with treatment with a vitamin K antagonist alone.12
Our patient was started on a heparin drip and transitioned to an oral vitamin K antagonist with an INR goal of 2 to 3. Lifelong anticoagulation was planned. The pain and discoloration in her hands improved on anticoagulation and had nearly resolved by the time of discharge. Given her history of arterial thrombosis, the addition of aspirin was also considered, but this decision was ultimately deferred to her outpatient rheumatologist and hematologist.
1. Miyakis S, Lockshin MD, Atsumi T, et al. International consensus statement on an update of the classification criteria for definite antiphospholipid syndrome (APS). J Thromb Haemost. 2006;4:295-306. doi: 10.1111/j.1538-7836.2006.01753.x
2. Duarte-García A, Pham MM, Crowson CS, et al. The epidemiology of antiphospholipid syndrome: a population-based study. Arthritis Rheumatol. 2019;71:1545-1552. doi: 10.1002/art.40901
3. Levine JS, Branch DW, Rauch J. The antiphospholipid syndrome. N Engl J Med. 2002;346:752-763. doi: 10.1056/NEJMra002974
4. Cervera R, Khamashta MA, Font J, et al. Systemic lupus erythematosus: clinical and immunologic patterns of disease expression in a cohort of 1,000 patients. Medicine (Baltimore). 1993;72:113-124.
5. Love PE, Santoro SA. Antiphospholipid antibodies: anticardiolipin and the lupus anticoagulant in systemic lupus erythematosus (SLE) and in non-SLE disorders: prevalence and clinical significance. Ann Intern Med. 1990;112:682-698. doi: 10.7326/0003-4819-112-9-682
6. Merkel PA, Chang YC, Pierangeli SS, et al. The prevalence and clinical associations of anticardiolipin antibodies in a large inception cohort of patients with connective tissue diseases. Am J Med. 1996;101:576-583. doi: 10.1016/s0002-9343(96)00335-x
7. Petri M. Epidemiology of the antiphospholipid antibody syndrome. J Autoimmun. 2000;15:145-151. doi: 10.1006/jaut. 2000.0409
8. Bozlar U, Ogur T, Khaja MS, et al. CT angiography of the upper extremity arterial system: Part 2—Clinical applications beyond trauma patients. AJR Am J Roentgenol. 2013;201:753-763. doi: 10.2214/AJR.13.11208
9. Ruiz-Irastorza G, Hunt BJ, Khamashta MA. A systematic review of secondary thromboprophylaxis in patients with antiphospholipid antibodies. Arthritis Rheum. 2007;7:1487-1495. doi: 10.1002/art.23109
10. Tektonidou MG, Andreoli L, Limper M, et al. EULAR recommendations for the management of antiphospholipid syndrome in adults. Ann Rheum Dis. 2019;78:1296-1304. doi: 10.1136/annrheumdis-2019-215213
Efficacy and safety of rivaroxaban vs warfarin in high-risk patients with antiphospholipid syndrome: rationale and design of the Trial on Rivaroxaban in AntiPhospholipid Syndrome (TRAPS) trial. Lupus. 2016;25:301-306. doi: 10.1177/0961203315611495
12. Schmidt-Tanguy A, Voswinkel J, Henrion D, et al. Antithrombotic effects of hydroxychloroquine in primary antiphospholipid syndrome patients. J Thromb Haemost. 2013;11:1927-1929. doi: 10.1111/jth.12363
1. Miyakis S, Lockshin MD, Atsumi T, et al. International consensus statement on an update of the classification criteria for definite antiphospholipid syndrome (APS). J Thromb Haemost. 2006;4:295-306. doi: 10.1111/j.1538-7836.2006.01753.x
2. Duarte-García A, Pham MM, Crowson CS, et al. The epidemiology of antiphospholipid syndrome: a population-based study. Arthritis Rheumatol. 2019;71:1545-1552. doi: 10.1002/art.40901
3. Levine JS, Branch DW, Rauch J. The antiphospholipid syndrome. N Engl J Med. 2002;346:752-763. doi: 10.1056/NEJMra002974
4. Cervera R, Khamashta MA, Font J, et al. Systemic lupus erythematosus: clinical and immunologic patterns of disease expression in a cohort of 1,000 patients. Medicine (Baltimore). 1993;72:113-124.
5. Love PE, Santoro SA. Antiphospholipid antibodies: anticardiolipin and the lupus anticoagulant in systemic lupus erythematosus (SLE) and in non-SLE disorders: prevalence and clinical significance. Ann Intern Med. 1990;112:682-698. doi: 10.7326/0003-4819-112-9-682
6. Merkel PA, Chang YC, Pierangeli SS, et al. The prevalence and clinical associations of anticardiolipin antibodies in a large inception cohort of patients with connective tissue diseases. Am J Med. 1996;101:576-583. doi: 10.1016/s0002-9343(96)00335-x
7. Petri M. Epidemiology of the antiphospholipid antibody syndrome. J Autoimmun. 2000;15:145-151. doi: 10.1006/jaut. 2000.0409
8. Bozlar U, Ogur T, Khaja MS, et al. CT angiography of the upper extremity arterial system: Part 2—Clinical applications beyond trauma patients. AJR Am J Roentgenol. 2013;201:753-763. doi: 10.2214/AJR.13.11208
9. Ruiz-Irastorza G, Hunt BJ, Khamashta MA. A systematic review of secondary thromboprophylaxis in patients with antiphospholipid antibodies. Arthritis Rheum. 2007;7:1487-1495. doi: 10.1002/art.23109
10. Tektonidou MG, Andreoli L, Limper M, et al. EULAR recommendations for the management of antiphospholipid syndrome in adults. Ann Rheum Dis. 2019;78:1296-1304. doi: 10.1136/annrheumdis-2019-215213
Efficacy and safety of rivaroxaban vs warfarin in high-risk patients with antiphospholipid syndrome: rationale and design of the Trial on Rivaroxaban in AntiPhospholipid Syndrome (TRAPS) trial. Lupus. 2016;25:301-306. doi: 10.1177/0961203315611495
12. Schmidt-Tanguy A, Voswinkel J, Henrion D, et al. Antithrombotic effects of hydroxychloroquine in primary antiphospholipid syndrome patients. J Thromb Haemost. 2013;11:1927-1929. doi: 10.1111/jth.12363
How to help pediatricians apply peanut allergy guidelines
Despite the profound shift in guidelines for preventing peanut allergies in infants after the landmark LEAP study, national surveys in 2021 showed that 70% of parents and caregivers said that they hadn’t heard the new recommendations, and fewer than one-third of pediatricians were following them.
Now, in a 5-year National Institutes of Health–funded study called iREACH, researchers are testing whether a two-part intervention, which includes training videos and a clinical decision support tool, helps pediatricians follow the guidelines and ultimately reduces peanut allergy.
Early results from iREACH, presented at the American Academy of Allergy, Asthma, and Immunology 2023 annual meeting in San Antonio, showed mixed results with a sharp rise in clinician knowledge of the guidelines but only a modest increase in their real-world implementation with high-risk infants.
Raising a food-allergic child while working as a pediatrician herself, Ruchi Gupta, MD, MPH, director of the Center for Food Allergy and Asthma Research at Northwestern University, Chicago, understands the importance and challenge of translating published findings into practice.
During a typical 4- to 6-month well-child visit, pediatricians must check the baby’s growth, perform a physical exam, discuss milestones, field questions about sleep and poop and colic and – if they’re up on the latest guidelines – explain why it’s important to feed peanuts early and often.
“Pediatricians get stuff from every single specialty, and guidelines are always changing,” she told this news organization.
The current feeding guidelines, published in 2017 after the landmark LEAP study, switched from “ ‘don’t introduce peanuts until age 3’ to ‘introduce peanuts now,’ ” said Dr. Gupta.
But the recommendations aren’t entirely straightforward. They require pediatricians to make an assessment when the baby is around 4 months old. If the child is high-risk (has severe eczema or an egg allergy), they need a peanut-specific immunoglobulin E (IgE) test. If the test is negative, the pediatrician should encourage peanut introduction. If positive, they should refer the child to an allergist.
“It’s a little complicated,” Dr. Gupta said.
To boost understanding and adherence, Dr. Gupta’s team created the intervention tested in the iREACH study. It includes a set of training videos, a clinical decision support tool that embeds into the electronic health record (EHR) with pop-ups reminding the physician to discuss early introduction, menus for ordering peanut IgE tests or referring to an allergist if needed, and a caregiver handout that explains how to add peanuts to the baby’s diet. (These resources can be found here.)
The study enrolled 290 pediatric clinicians at 30 local practices, examining 18,460 babies from diverse backgrounds, about one-quarter of whom were from families on public insurance. About half of the clinicians received the intervention, whereas the other half served as the control arm.
The training videos seemed effective. Clinicians’ knowledge of the guidelines rose from 72.6% at baseline to 94.5% after the intervention, and their ability to identify severe eczema went up from 63.4% to 97.6%. This translated to 70.4% success with applying the guidelines when presented various clinical scenarios, up from 29% at baseline. These results are in press at JAMA Network Open.
The next set of analyses, preliminary and unpublished, monitored real-world adherence using natural language processing to pull EHR data from 4- and 6-month well-check visits. It was “AI [artificial intelligence] for notes,” Dr. Gupta said.
For low-risk infants, the training and EHR-embedded support tool greatly improved clinician adherence. Eighty percent of clinicians in the intervention arm followed the guidelines, compared with 26% in the control group.
In high-risk infants, the impact was much weaker. Even after the video-based training, only 17% of pediatric clinicians followed the guidelines – that is, ordered a peanut IgE test or referred to an allergist – compared with 8% in the control group.
Why such a low uptake?
Pediatricians are time-pressed. “How do you add [early introduction] to the other 10 or 15 things you want to talk to a parent about at the 4-month visit?” said Jonathan Necheles, MD, MPH, a pediatrician at Children’s Healthcare Associates in Chicago.
It can also be hard to tell if a baby’s eczema is “severe” or “mild to moderate.” The EHR-integrated support tool included a scorecard for judging eczema severity across a range of skin tones. The condition can be hard to recognize in patients of color. “You don’t get the redness in the same way,” said Dr. Necheles, who worked with Dr. Gupta to develop the iREACH intervention.
Curiously, even though the AI analysis found that less than one-fifth of pediatricians put the guidelines into action for high-risk infants, 69% of them recommended peanut introduction.
One interpretation is that busy pediatricians may be “doing the minimum” – introducing the concept of early introduction and telling parents to try it “but not giving any additional sort of guidance as far as who’s high risk, who’s low risk, who should see the allergist, who should get screened,” said Edwin Kim, MD, allergist-immunologist and director of the Food Allergy Initiative at the University of North Carolina at Chapel Hill.
The ultimate impact of iREACH has yet to be seen. “The end goal is, if pediatricians recommend, will parents follow, and will we reduce peanut allergy?” Dr. Gupta said.
Dr. Gupta consults or serves as an advisor for Genentech, Novartis, Aimmune, Allergenis, and Food Allergy Research & Education; receives research funding from Novartis, Genentech, FARE, Melchiorre Family Foundation, and Sunshine Charitable Foundation; and reports ownership interest from Yobee Care. Dr. Necheles reports no financial disclosures. Dr. Kim reports consultancy with Allergy Therapeutics, Belhaven Biopharma, Duke Clinical Research Institute, Genentech, Nutricia, and Revolo; advisory board membership with ALK, Kenota Health, and Ukko; and grant support from the National Institute of Allergy and Infectious Diseases, Immune Tolerance Network, and Food Allergy Research and Education.
A version of this article first appeared on Medscape.com.
Despite the profound shift in guidelines for preventing peanut allergies in infants after the landmark LEAP study, national surveys in 2021 showed that 70% of parents and caregivers said that they hadn’t heard the new recommendations, and fewer than one-third of pediatricians were following them.
Now, in a 5-year National Institutes of Health–funded study called iREACH, researchers are testing whether a two-part intervention, which includes training videos and a clinical decision support tool, helps pediatricians follow the guidelines and ultimately reduces peanut allergy.
Early results from iREACH, presented at the American Academy of Allergy, Asthma, and Immunology 2023 annual meeting in San Antonio, showed mixed results with a sharp rise in clinician knowledge of the guidelines but only a modest increase in their real-world implementation with high-risk infants.
Raising a food-allergic child while working as a pediatrician herself, Ruchi Gupta, MD, MPH, director of the Center for Food Allergy and Asthma Research at Northwestern University, Chicago, understands the importance and challenge of translating published findings into practice.
During a typical 4- to 6-month well-child visit, pediatricians must check the baby’s growth, perform a physical exam, discuss milestones, field questions about sleep and poop and colic and – if they’re up on the latest guidelines – explain why it’s important to feed peanuts early and often.
“Pediatricians get stuff from every single specialty, and guidelines are always changing,” she told this news organization.
The current feeding guidelines, published in 2017 after the landmark LEAP study, switched from “ ‘don’t introduce peanuts until age 3’ to ‘introduce peanuts now,’ ” said Dr. Gupta.
But the recommendations aren’t entirely straightforward. They require pediatricians to make an assessment when the baby is around 4 months old. If the child is high-risk (has severe eczema or an egg allergy), they need a peanut-specific immunoglobulin E (IgE) test. If the test is negative, the pediatrician should encourage peanut introduction. If positive, they should refer the child to an allergist.
“It’s a little complicated,” Dr. Gupta said.
To boost understanding and adherence, Dr. Gupta’s team created the intervention tested in the iREACH study. It includes a set of training videos, a clinical decision support tool that embeds into the electronic health record (EHR) with pop-ups reminding the physician to discuss early introduction, menus for ordering peanut IgE tests or referring to an allergist if needed, and a caregiver handout that explains how to add peanuts to the baby’s diet. (These resources can be found here.)
The study enrolled 290 pediatric clinicians at 30 local practices, examining 18,460 babies from diverse backgrounds, about one-quarter of whom were from families on public insurance. About half of the clinicians received the intervention, whereas the other half served as the control arm.
The training videos seemed effective. Clinicians’ knowledge of the guidelines rose from 72.6% at baseline to 94.5% after the intervention, and their ability to identify severe eczema went up from 63.4% to 97.6%. This translated to 70.4% success with applying the guidelines when presented various clinical scenarios, up from 29% at baseline. These results are in press at JAMA Network Open.
The next set of analyses, preliminary and unpublished, monitored real-world adherence using natural language processing to pull EHR data from 4- and 6-month well-check visits. It was “AI [artificial intelligence] for notes,” Dr. Gupta said.
For low-risk infants, the training and EHR-embedded support tool greatly improved clinician adherence. Eighty percent of clinicians in the intervention arm followed the guidelines, compared with 26% in the control group.
In high-risk infants, the impact was much weaker. Even after the video-based training, only 17% of pediatric clinicians followed the guidelines – that is, ordered a peanut IgE test or referred to an allergist – compared with 8% in the control group.
Why such a low uptake?
Pediatricians are time-pressed. “How do you add [early introduction] to the other 10 or 15 things you want to talk to a parent about at the 4-month visit?” said Jonathan Necheles, MD, MPH, a pediatrician at Children’s Healthcare Associates in Chicago.
It can also be hard to tell if a baby’s eczema is “severe” or “mild to moderate.” The EHR-integrated support tool included a scorecard for judging eczema severity across a range of skin tones. The condition can be hard to recognize in patients of color. “You don’t get the redness in the same way,” said Dr. Necheles, who worked with Dr. Gupta to develop the iREACH intervention.
Curiously, even though the AI analysis found that less than one-fifth of pediatricians put the guidelines into action for high-risk infants, 69% of them recommended peanut introduction.
One interpretation is that busy pediatricians may be “doing the minimum” – introducing the concept of early introduction and telling parents to try it “but not giving any additional sort of guidance as far as who’s high risk, who’s low risk, who should see the allergist, who should get screened,” said Edwin Kim, MD, allergist-immunologist and director of the Food Allergy Initiative at the University of North Carolina at Chapel Hill.
The ultimate impact of iREACH has yet to be seen. “The end goal is, if pediatricians recommend, will parents follow, and will we reduce peanut allergy?” Dr. Gupta said.
Dr. Gupta consults or serves as an advisor for Genentech, Novartis, Aimmune, Allergenis, and Food Allergy Research & Education; receives research funding from Novartis, Genentech, FARE, Melchiorre Family Foundation, and Sunshine Charitable Foundation; and reports ownership interest from Yobee Care. Dr. Necheles reports no financial disclosures. Dr. Kim reports consultancy with Allergy Therapeutics, Belhaven Biopharma, Duke Clinical Research Institute, Genentech, Nutricia, and Revolo; advisory board membership with ALK, Kenota Health, and Ukko; and grant support from the National Institute of Allergy and Infectious Diseases, Immune Tolerance Network, and Food Allergy Research and Education.
A version of this article first appeared on Medscape.com.
Despite the profound shift in guidelines for preventing peanut allergies in infants after the landmark LEAP study, national surveys in 2021 showed that 70% of parents and caregivers said that they hadn’t heard the new recommendations, and fewer than one-third of pediatricians were following them.
Now, in a 5-year National Institutes of Health–funded study called iREACH, researchers are testing whether a two-part intervention, which includes training videos and a clinical decision support tool, helps pediatricians follow the guidelines and ultimately reduces peanut allergy.
Early results from iREACH, presented at the American Academy of Allergy, Asthma, and Immunology 2023 annual meeting in San Antonio, showed mixed results with a sharp rise in clinician knowledge of the guidelines but only a modest increase in their real-world implementation with high-risk infants.
Raising a food-allergic child while working as a pediatrician herself, Ruchi Gupta, MD, MPH, director of the Center for Food Allergy and Asthma Research at Northwestern University, Chicago, understands the importance and challenge of translating published findings into practice.
During a typical 4- to 6-month well-child visit, pediatricians must check the baby’s growth, perform a physical exam, discuss milestones, field questions about sleep and poop and colic and – if they’re up on the latest guidelines – explain why it’s important to feed peanuts early and often.
“Pediatricians get stuff from every single specialty, and guidelines are always changing,” she told this news organization.
The current feeding guidelines, published in 2017 after the landmark LEAP study, switched from “ ‘don’t introduce peanuts until age 3’ to ‘introduce peanuts now,’ ” said Dr. Gupta.
But the recommendations aren’t entirely straightforward. They require pediatricians to make an assessment when the baby is around 4 months old. If the child is high-risk (has severe eczema or an egg allergy), they need a peanut-specific immunoglobulin E (IgE) test. If the test is negative, the pediatrician should encourage peanut introduction. If positive, they should refer the child to an allergist.
“It’s a little complicated,” Dr. Gupta said.
To boost understanding and adherence, Dr. Gupta’s team created the intervention tested in the iREACH study. It includes a set of training videos, a clinical decision support tool that embeds into the electronic health record (EHR) with pop-ups reminding the physician to discuss early introduction, menus for ordering peanut IgE tests or referring to an allergist if needed, and a caregiver handout that explains how to add peanuts to the baby’s diet. (These resources can be found here.)
The study enrolled 290 pediatric clinicians at 30 local practices, examining 18,460 babies from diverse backgrounds, about one-quarter of whom were from families on public insurance. About half of the clinicians received the intervention, whereas the other half served as the control arm.
The training videos seemed effective. Clinicians’ knowledge of the guidelines rose from 72.6% at baseline to 94.5% after the intervention, and their ability to identify severe eczema went up from 63.4% to 97.6%. This translated to 70.4% success with applying the guidelines when presented various clinical scenarios, up from 29% at baseline. These results are in press at JAMA Network Open.
The next set of analyses, preliminary and unpublished, monitored real-world adherence using natural language processing to pull EHR data from 4- and 6-month well-check visits. It was “AI [artificial intelligence] for notes,” Dr. Gupta said.
For low-risk infants, the training and EHR-embedded support tool greatly improved clinician adherence. Eighty percent of clinicians in the intervention arm followed the guidelines, compared with 26% in the control group.
In high-risk infants, the impact was much weaker. Even after the video-based training, only 17% of pediatric clinicians followed the guidelines – that is, ordered a peanut IgE test or referred to an allergist – compared with 8% in the control group.
Why such a low uptake?
Pediatricians are time-pressed. “How do you add [early introduction] to the other 10 or 15 things you want to talk to a parent about at the 4-month visit?” said Jonathan Necheles, MD, MPH, a pediatrician at Children’s Healthcare Associates in Chicago.
It can also be hard to tell if a baby’s eczema is “severe” or “mild to moderate.” The EHR-integrated support tool included a scorecard for judging eczema severity across a range of skin tones. The condition can be hard to recognize in patients of color. “You don’t get the redness in the same way,” said Dr. Necheles, who worked with Dr. Gupta to develop the iREACH intervention.
Curiously, even though the AI analysis found that less than one-fifth of pediatricians put the guidelines into action for high-risk infants, 69% of them recommended peanut introduction.
One interpretation is that busy pediatricians may be “doing the minimum” – introducing the concept of early introduction and telling parents to try it “but not giving any additional sort of guidance as far as who’s high risk, who’s low risk, who should see the allergist, who should get screened,” said Edwin Kim, MD, allergist-immunologist and director of the Food Allergy Initiative at the University of North Carolina at Chapel Hill.
The ultimate impact of iREACH has yet to be seen. “The end goal is, if pediatricians recommend, will parents follow, and will we reduce peanut allergy?” Dr. Gupta said.
Dr. Gupta consults or serves as an advisor for Genentech, Novartis, Aimmune, Allergenis, and Food Allergy Research & Education; receives research funding from Novartis, Genentech, FARE, Melchiorre Family Foundation, and Sunshine Charitable Foundation; and reports ownership interest from Yobee Care. Dr. Necheles reports no financial disclosures. Dr. Kim reports consultancy with Allergy Therapeutics, Belhaven Biopharma, Duke Clinical Research Institute, Genentech, Nutricia, and Revolo; advisory board membership with ALK, Kenota Health, and Ukko; and grant support from the National Institute of Allergy and Infectious Diseases, Immune Tolerance Network, and Food Allergy Research and Education.
A version of this article first appeared on Medscape.com.
FROM AAAAI 2023
New data strengthen case for oral immunotherapy in tots
The buzz surrounding OIT – which involves ingesting daily doses of the culprit food to raise the threshold that would trigger a reaction – grew with the approval, by the U.S. Food and Drug Administration of Palforzia, of the peanut OIT pill in January 2020. Yet many allergists remained wary about the treatment, a monthslong regimen that can itself trigger allergic reactions.
Now, accumulating research points to “a possible window of opportunity early in life, less than 3 years of age, for more successful disease remission,” Justin Schwartz, MD, PhD, an allergist at Cincinnati Children’s Hospital, told a crowd at the annual meeting of the American Academy of Allergy, Asthma, & Immunology.
His presentation about OIT in toddlers kicked off a 3-hour clinical practice course, one of several dozen conference offerings highlighting this emerging approach.
Several AAAAI posters add to prior studies (for example, DEVIL and IMPACT) suggesting that OIT proceeds more smoothly and faster during a child’s earliest years – a season fraught with accidental exposures and reactions.
One poster described a retrospective study of 73 children younger than 4 years who underwent OIT at the Cleveland Clinic Food Allergy Center. Sixty-four were treated for peanut allergies, and seven patients received OIT for multiple foods, including tree nuts, milk, wheat, and sesame.
Of the 80 total OIT courses, 76 (95%) reached maintenance – meaning the child tolerated a small amount (for example, 1-2 peanuts) without reacting – in a median of 104 days (~3.4 months).
That is “quite impressive,” said allergist Hugh Windom, MD, whose clinic in Sarasota, Fla., has offered OIT since 2012.
Older children typically have 80%-90% success and take longer (6-8 months) to reach maintenance because of busier schedules and reactions that slow them down, he said. In his clinic’s larger retrospective analysis of preschool-aged OIT patients, presented at the 2022 AAAAI meeting, 89% of patients with peanut allergies and 72% of children with multiple food allergies achieved maintenance.
In the Cleveland Clinic study, children with favorable lab test results after receiving the maintenance dose for 6 months were offered an oral food challenge. Of 24 patients who completed the challenge, 75% “passed with a normal serving size of the treated food (for example, two tablespoons of peanut butter),” Sarah Johnson, MD, lead author and Cleveland Clinic allergy/immunology fellow, said in an interview.
Plus, OIT seemed safer for toddlers. Although 41% of the children had reactions during clinic updosing and 48% had reactions at home, only ~3% of toddler OIT courses required epinephrine. By comparison, ~11% of treatments required epinephrine in a large OIT study of older children.
When a child reacts, “you might keep them on the dose or go a little slower,” said Johnson, who worked with allergist Jaclyn Bjelac, MD, on the study. These setbacks occurred less frequently in toddlers, allowing their OIT to “go a lot faster” than in older children. And so far, Dr. Johnson said, none of the toddlers have shown signs of eosinophilic esophagitis, a rare complication that can develop during OIT.
A smaller analysis of real-world outcomes in an academic clinical setting also found that OIT was well tolerated at very young ages. Since 2020, this ongoing study at UVA Children’s Hospital in Charlottesville has enrolled 22 peanut-allergic children (aged 6 months to 3 years) for OIT. Three patients have dropped out, four are in the buildup phrase, and 15 have reached maintenance dosing. None have reported having to use epinephrine.
Three patients have completed 1 year of maintenance therapy, and another patient accidentally consumed ~3,000 mg of peanut protein (equivalent to ~10 peanuts) after 5 months of maintenance. All four “now incorporate peanut into their diets ad lib,” according to lead author and allergist Jonathan Hemler, MD, who directs the UVA pediatric food allergy program.
These findings are “really reassuring – because even if you may not offer OIT, you’re still going to get questions about it,” said Ama Alexis, MD, an allergist/immunologist in private practice in New York and a clinical assistant professor at NYU Grossmann School of Medicine, commenting on the Cleveland Clinic study.
“It’s great that we’re hearing and seeing so much about OIT,” she added. While training as an allergy/immunology fellow 15 years ago, many saw the treatment as dangerous – “an absolute no-no,” she said.
The AAAAI still considers OIT “investigational,” yet this year’s annual meeting featured 22 posters – plus a course, workshop, seminar, and oral abstract session – on the approach.
The “thought process has shifted,” Dr. Alexis said. “It’s good to see all these numbers, these results. I think once you’re comfortable, you should embrace new therapies.”
Dr. Schwartz has consulted for Shire/Takeda and has received research funding from Knopp Biosciences. Dr. Alexis consults for AbbVie, serves on advisory boards for Jansen and Eli Lilli, and is a member of Pfizer’s advisory board and speaker’s bureau. Dr. Johnson, Dr. Windom, and Dr. Hemler report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The buzz surrounding OIT – which involves ingesting daily doses of the culprit food to raise the threshold that would trigger a reaction – grew with the approval, by the U.S. Food and Drug Administration of Palforzia, of the peanut OIT pill in January 2020. Yet many allergists remained wary about the treatment, a monthslong regimen that can itself trigger allergic reactions.
Now, accumulating research points to “a possible window of opportunity early in life, less than 3 years of age, for more successful disease remission,” Justin Schwartz, MD, PhD, an allergist at Cincinnati Children’s Hospital, told a crowd at the annual meeting of the American Academy of Allergy, Asthma, & Immunology.
His presentation about OIT in toddlers kicked off a 3-hour clinical practice course, one of several dozen conference offerings highlighting this emerging approach.
Several AAAAI posters add to prior studies (for example, DEVIL and IMPACT) suggesting that OIT proceeds more smoothly and faster during a child’s earliest years – a season fraught with accidental exposures and reactions.
One poster described a retrospective study of 73 children younger than 4 years who underwent OIT at the Cleveland Clinic Food Allergy Center. Sixty-four were treated for peanut allergies, and seven patients received OIT for multiple foods, including tree nuts, milk, wheat, and sesame.
Of the 80 total OIT courses, 76 (95%) reached maintenance – meaning the child tolerated a small amount (for example, 1-2 peanuts) without reacting – in a median of 104 days (~3.4 months).
That is “quite impressive,” said allergist Hugh Windom, MD, whose clinic in Sarasota, Fla., has offered OIT since 2012.
Older children typically have 80%-90% success and take longer (6-8 months) to reach maintenance because of busier schedules and reactions that slow them down, he said. In his clinic’s larger retrospective analysis of preschool-aged OIT patients, presented at the 2022 AAAAI meeting, 89% of patients with peanut allergies and 72% of children with multiple food allergies achieved maintenance.
In the Cleveland Clinic study, children with favorable lab test results after receiving the maintenance dose for 6 months were offered an oral food challenge. Of 24 patients who completed the challenge, 75% “passed with a normal serving size of the treated food (for example, two tablespoons of peanut butter),” Sarah Johnson, MD, lead author and Cleveland Clinic allergy/immunology fellow, said in an interview.
Plus, OIT seemed safer for toddlers. Although 41% of the children had reactions during clinic updosing and 48% had reactions at home, only ~3% of toddler OIT courses required epinephrine. By comparison, ~11% of treatments required epinephrine in a large OIT study of older children.
When a child reacts, “you might keep them on the dose or go a little slower,” said Johnson, who worked with allergist Jaclyn Bjelac, MD, on the study. These setbacks occurred less frequently in toddlers, allowing their OIT to “go a lot faster” than in older children. And so far, Dr. Johnson said, none of the toddlers have shown signs of eosinophilic esophagitis, a rare complication that can develop during OIT.
A smaller analysis of real-world outcomes in an academic clinical setting also found that OIT was well tolerated at very young ages. Since 2020, this ongoing study at UVA Children’s Hospital in Charlottesville has enrolled 22 peanut-allergic children (aged 6 months to 3 years) for OIT. Three patients have dropped out, four are in the buildup phrase, and 15 have reached maintenance dosing. None have reported having to use epinephrine.
Three patients have completed 1 year of maintenance therapy, and another patient accidentally consumed ~3,000 mg of peanut protein (equivalent to ~10 peanuts) after 5 months of maintenance. All four “now incorporate peanut into their diets ad lib,” according to lead author and allergist Jonathan Hemler, MD, who directs the UVA pediatric food allergy program.
These findings are “really reassuring – because even if you may not offer OIT, you’re still going to get questions about it,” said Ama Alexis, MD, an allergist/immunologist in private practice in New York and a clinical assistant professor at NYU Grossmann School of Medicine, commenting on the Cleveland Clinic study.
“It’s great that we’re hearing and seeing so much about OIT,” she added. While training as an allergy/immunology fellow 15 years ago, many saw the treatment as dangerous – “an absolute no-no,” she said.
The AAAAI still considers OIT “investigational,” yet this year’s annual meeting featured 22 posters – plus a course, workshop, seminar, and oral abstract session – on the approach.
The “thought process has shifted,” Dr. Alexis said. “It’s good to see all these numbers, these results. I think once you’re comfortable, you should embrace new therapies.”
Dr. Schwartz has consulted for Shire/Takeda and has received research funding from Knopp Biosciences. Dr. Alexis consults for AbbVie, serves on advisory boards for Jansen and Eli Lilli, and is a member of Pfizer’s advisory board and speaker’s bureau. Dr. Johnson, Dr. Windom, and Dr. Hemler report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The buzz surrounding OIT – which involves ingesting daily doses of the culprit food to raise the threshold that would trigger a reaction – grew with the approval, by the U.S. Food and Drug Administration of Palforzia, of the peanut OIT pill in January 2020. Yet many allergists remained wary about the treatment, a monthslong regimen that can itself trigger allergic reactions.
Now, accumulating research points to “a possible window of opportunity early in life, less than 3 years of age, for more successful disease remission,” Justin Schwartz, MD, PhD, an allergist at Cincinnati Children’s Hospital, told a crowd at the annual meeting of the American Academy of Allergy, Asthma, & Immunology.
His presentation about OIT in toddlers kicked off a 3-hour clinical practice course, one of several dozen conference offerings highlighting this emerging approach.
Several AAAAI posters add to prior studies (for example, DEVIL and IMPACT) suggesting that OIT proceeds more smoothly and faster during a child’s earliest years – a season fraught with accidental exposures and reactions.
One poster described a retrospective study of 73 children younger than 4 years who underwent OIT at the Cleveland Clinic Food Allergy Center. Sixty-four were treated for peanut allergies, and seven patients received OIT for multiple foods, including tree nuts, milk, wheat, and sesame.
Of the 80 total OIT courses, 76 (95%) reached maintenance – meaning the child tolerated a small amount (for example, 1-2 peanuts) without reacting – in a median of 104 days (~3.4 months).
That is “quite impressive,” said allergist Hugh Windom, MD, whose clinic in Sarasota, Fla., has offered OIT since 2012.
Older children typically have 80%-90% success and take longer (6-8 months) to reach maintenance because of busier schedules and reactions that slow them down, he said. In his clinic’s larger retrospective analysis of preschool-aged OIT patients, presented at the 2022 AAAAI meeting, 89% of patients with peanut allergies and 72% of children with multiple food allergies achieved maintenance.
In the Cleveland Clinic study, children with favorable lab test results after receiving the maintenance dose for 6 months were offered an oral food challenge. Of 24 patients who completed the challenge, 75% “passed with a normal serving size of the treated food (for example, two tablespoons of peanut butter),” Sarah Johnson, MD, lead author and Cleveland Clinic allergy/immunology fellow, said in an interview.
Plus, OIT seemed safer for toddlers. Although 41% of the children had reactions during clinic updosing and 48% had reactions at home, only ~3% of toddler OIT courses required epinephrine. By comparison, ~11% of treatments required epinephrine in a large OIT study of older children.
When a child reacts, “you might keep them on the dose or go a little slower,” said Johnson, who worked with allergist Jaclyn Bjelac, MD, on the study. These setbacks occurred less frequently in toddlers, allowing their OIT to “go a lot faster” than in older children. And so far, Dr. Johnson said, none of the toddlers have shown signs of eosinophilic esophagitis, a rare complication that can develop during OIT.
A smaller analysis of real-world outcomes in an academic clinical setting also found that OIT was well tolerated at very young ages. Since 2020, this ongoing study at UVA Children’s Hospital in Charlottesville has enrolled 22 peanut-allergic children (aged 6 months to 3 years) for OIT. Three patients have dropped out, four are in the buildup phrase, and 15 have reached maintenance dosing. None have reported having to use epinephrine.
Three patients have completed 1 year of maintenance therapy, and another patient accidentally consumed ~3,000 mg of peanut protein (equivalent to ~10 peanuts) after 5 months of maintenance. All four “now incorporate peanut into their diets ad lib,” according to lead author and allergist Jonathan Hemler, MD, who directs the UVA pediatric food allergy program.
These findings are “really reassuring – because even if you may not offer OIT, you’re still going to get questions about it,” said Ama Alexis, MD, an allergist/immunologist in private practice in New York and a clinical assistant professor at NYU Grossmann School of Medicine, commenting on the Cleveland Clinic study.
“It’s great that we’re hearing and seeing so much about OIT,” she added. While training as an allergy/immunology fellow 15 years ago, many saw the treatment as dangerous – “an absolute no-no,” she said.
The AAAAI still considers OIT “investigational,” yet this year’s annual meeting featured 22 posters – plus a course, workshop, seminar, and oral abstract session – on the approach.
The “thought process has shifted,” Dr. Alexis said. “It’s good to see all these numbers, these results. I think once you’re comfortable, you should embrace new therapies.”
Dr. Schwartz has consulted for Shire/Takeda and has received research funding from Knopp Biosciences. Dr. Alexis consults for AbbVie, serves on advisory boards for Jansen and Eli Lilli, and is a member of Pfizer’s advisory board and speaker’s bureau. Dr. Johnson, Dr. Windom, and Dr. Hemler report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM AAAAI 2023