User login
Study Finds Isotretinoin Effective for Acne in Transgender Patients on Hormone Rx
TOPLINE:
, but more information is needed on dosing and barriers to treatment.
METHODOLOGY:
- Acne can be a side effect of masculinizing hormone therapy for transmasculine individuals. While isotretinoin is an effective treatment option for acne, its effectiveness and safety in transgender and gender-diverse individuals are not well understood.
- This retrospective case series included 55 patients (mean age, 25.4 years) undergoing masculinizing hormone therapy at four medical centers, who were prescribed isotretinoin for acne associated with treatment.
- Isotretinoin treatment was started a median of 22.1 months after hormone therapy was initiated and continued for a median of 6 months with a median cumulative dose of 132.7 mg/kg.
- Researchers assessed acne improvement, clearance, recurrence, adverse effects, and reasons for treatment discontinuation.
TAKEAWAY:
- Overall, 48 patients (87.3%) experienced improvement, and 26 (47.3%) achieved clearance during treatment. A higher proportion of patients experienced improvement (97% vs 72.7%) and achieved clearance (63.6% vs 22.7%) with cumulative doses of ≥ 120 mg/kg than those who received cumulative doses < 120 mg/kg.
- The risk for recurrence was 20% (in four patients) among 20 patients who achieved clearance and had any subsequent health care encounters, with a mean follow-up time of 734.3 days.
- Common adverse effects included dryness (80%), joint pain (14.5%), and headaches (10.9%). Other adverse effects included nose bleeds (9.1%) and depression (5.5%).
- Of the 22 patients with a cumulative dose < 120 mg/kg, 14 (63.6%) were lost to follow-up; among those not lost to follow-up, 2 patients discontinued treatment because of transfer of care, 1 because of adverse effects, and 1 because of gender-affirming surgery, with concerns about wound healing.
IN PRACTICE:
“Although isotretinoin appears to be an effective treatment option for acne among individuals undergoing masculinizing hormone therapy, further efforts are needed to understand optimal dosing and treatment barriers to improve outcomes in transgender and gender-diverse individuals receiving testosterone,” the authors concluded.
SOURCE:
The study, led by James Choe, BS, Department of Dermatology, Brigham and Women’s Hospital, Harvard Medical School, Boston, was published online in JAMA Dermatology.
LIMITATIONS:
The study population was limited to four centers, and variability in clinician- and patient-reported acne outcomes and missing information could affect the reliability of data. Because of the small sample size, the association of masculinizing hormone therapy regimens with outcomes could not be evaluated.
DISCLOSURES:
One author is supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Three authors reported receiving grants or personal fees from various sources. The other authors declared no conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
, but more information is needed on dosing and barriers to treatment.
METHODOLOGY:
- Acne can be a side effect of masculinizing hormone therapy for transmasculine individuals. While isotretinoin is an effective treatment option for acne, its effectiveness and safety in transgender and gender-diverse individuals are not well understood.
- This retrospective case series included 55 patients (mean age, 25.4 years) undergoing masculinizing hormone therapy at four medical centers, who were prescribed isotretinoin for acne associated with treatment.
- Isotretinoin treatment was started a median of 22.1 months after hormone therapy was initiated and continued for a median of 6 months with a median cumulative dose of 132.7 mg/kg.
- Researchers assessed acne improvement, clearance, recurrence, adverse effects, and reasons for treatment discontinuation.
TAKEAWAY:
- Overall, 48 patients (87.3%) experienced improvement, and 26 (47.3%) achieved clearance during treatment. A higher proportion of patients experienced improvement (97% vs 72.7%) and achieved clearance (63.6% vs 22.7%) with cumulative doses of ≥ 120 mg/kg than those who received cumulative doses < 120 mg/kg.
- The risk for recurrence was 20% (in four patients) among 20 patients who achieved clearance and had any subsequent health care encounters, with a mean follow-up time of 734.3 days.
- Common adverse effects included dryness (80%), joint pain (14.5%), and headaches (10.9%). Other adverse effects included nose bleeds (9.1%) and depression (5.5%).
- Of the 22 patients with a cumulative dose < 120 mg/kg, 14 (63.6%) were lost to follow-up; among those not lost to follow-up, 2 patients discontinued treatment because of transfer of care, 1 because of adverse effects, and 1 because of gender-affirming surgery, with concerns about wound healing.
IN PRACTICE:
“Although isotretinoin appears to be an effective treatment option for acne among individuals undergoing masculinizing hormone therapy, further efforts are needed to understand optimal dosing and treatment barriers to improve outcomes in transgender and gender-diverse individuals receiving testosterone,” the authors concluded.
SOURCE:
The study, led by James Choe, BS, Department of Dermatology, Brigham and Women’s Hospital, Harvard Medical School, Boston, was published online in JAMA Dermatology.
LIMITATIONS:
The study population was limited to four centers, and variability in clinician- and patient-reported acne outcomes and missing information could affect the reliability of data. Because of the small sample size, the association of masculinizing hormone therapy regimens with outcomes could not be evaluated.
DISCLOSURES:
One author is supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Three authors reported receiving grants or personal fees from various sources. The other authors declared no conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
, but more information is needed on dosing and barriers to treatment.
METHODOLOGY:
- Acne can be a side effect of masculinizing hormone therapy for transmasculine individuals. While isotretinoin is an effective treatment option for acne, its effectiveness and safety in transgender and gender-diverse individuals are not well understood.
- This retrospective case series included 55 patients (mean age, 25.4 years) undergoing masculinizing hormone therapy at four medical centers, who were prescribed isotretinoin for acne associated with treatment.
- Isotretinoin treatment was started a median of 22.1 months after hormone therapy was initiated and continued for a median of 6 months with a median cumulative dose of 132.7 mg/kg.
- Researchers assessed acne improvement, clearance, recurrence, adverse effects, and reasons for treatment discontinuation.
TAKEAWAY:
- Overall, 48 patients (87.3%) experienced improvement, and 26 (47.3%) achieved clearance during treatment. A higher proportion of patients experienced improvement (97% vs 72.7%) and achieved clearance (63.6% vs 22.7%) with cumulative doses of ≥ 120 mg/kg than those who received cumulative doses < 120 mg/kg.
- The risk for recurrence was 20% (in four patients) among 20 patients who achieved clearance and had any subsequent health care encounters, with a mean follow-up time of 734.3 days.
- Common adverse effects included dryness (80%), joint pain (14.5%), and headaches (10.9%). Other adverse effects included nose bleeds (9.1%) and depression (5.5%).
- Of the 22 patients with a cumulative dose < 120 mg/kg, 14 (63.6%) were lost to follow-up; among those not lost to follow-up, 2 patients discontinued treatment because of transfer of care, 1 because of adverse effects, and 1 because of gender-affirming surgery, with concerns about wound healing.
IN PRACTICE:
“Although isotretinoin appears to be an effective treatment option for acne among individuals undergoing masculinizing hormone therapy, further efforts are needed to understand optimal dosing and treatment barriers to improve outcomes in transgender and gender-diverse individuals receiving testosterone,” the authors concluded.
SOURCE:
The study, led by James Choe, BS, Department of Dermatology, Brigham and Women’s Hospital, Harvard Medical School, Boston, was published online in JAMA Dermatology.
LIMITATIONS:
The study population was limited to four centers, and variability in clinician- and patient-reported acne outcomes and missing information could affect the reliability of data. Because of the small sample size, the association of masculinizing hormone therapy regimens with outcomes could not be evaluated.
DISCLOSURES:
One author is supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Three authors reported receiving grants or personal fees from various sources. The other authors declared no conflicts of interest.
A version of this article first appeared on Medscape.com.
Hidradenitis Suppurativa: Clinical Outcomes for Bimekizumab Positive in Phase 3 Studies
TOPLINE:
, in two phase 3 studies.
METHODOLOGY:
- To assess the efficacy and safety of bimekizumab, an interleukin (IL)-17A and IL-17F antagonist, 320 mg for HS, researchers conducted two 48-week phase 3 trials BE HEARD I (n = 505) and II (n = 509), which enrolled patients with moderate to severe HS and a history of inadequate response to systemic antibiotics.
- Patients were randomly assigned to one of four groups: Bimekizumab every 2 weeks, bimekizumab every 2 weeks for 16 weeks followed by every 4 weeks of dosing, bimekizumab every 4 weeks, or placebo for 16 weeks followed by bimekizumab every 2 weeks.
- The primary outcome was an HS clinical response of at least 50% (HiSCR50) at week 16, defined as at least a 50% reduction in total abscess and inflammatory nodule count.
TAKEAWAY:
- A higher proportion of patients receiving bimekizumab every 2 weeks vs placebo achieved an HiSCR50 response at week 16 in BE HEARD I (48% vs 29%; odds ratio [OR], 2.23; P = .006) and II (52% vs 32%; OR, 2.29; P = .0032) trials.
- Patients receiving bimekizumab every 4 weeks also achieved a higher HiSCR50 response at week 16 vs placebo in the BE HEARD II trial (54% vs 32%; OR, 2.42; P = .0038).
- At week 16, a higher proportion of patients receiving bimekizumab every 2 weeks vs placebo achieved at least a 75% HiSCR (HiSCR75) in both trials, and a higher proportion of those receiving bimekizumab every 4 weeks achieved HiSCR75 in the BE HEARD II trial.
- At week 48, 45%-68% of patients achieved HiSCR50 in both trials.
- Patients who received bimekizumab vs placebo for the initial 16 weeks had greater improvements in patient-reported outcomes, and bimekizumab was well tolerated with a low number of serious or severe treatment-emergent adverse events.
IN PRACTICE:
“Bimekizumab was well tolerated by patients with hidradenitis suppurativa and produced rapid and deep clinically meaningful responses that were maintained up to 48 weeks,” the authors wrote. “These data support the use of bimekizumab as a promising new therapeutic option for patients with moderate to severe hidradenitis suppurativa.”
SOURCE:
Alexa B. Kimball, MD, MPH, from Beth Israel Deaconess Medical Center and Harvard Medical School, Boston, led this study, which was published online in The Lancet.
LIMITATIONS:
The placebo-controlled part of this trial was relatively short at 16 weeks and may affect the interpretation of later efficacy data, there was a lack of an active comparator group, and the efficacy of treatment was evaluated in the presence of rescue treatment with systemic antibiotics.
DISCLOSURES:
The studies were funded by bimekizumab manufacturer UCB Pharma. Seven authors disclosed being current or former employees of UCB Pharma. Other authors reported several ties with many companies, including UCB Pharma.
A version of this article first appeared on Medscape.com.
TOPLINE:
, in two phase 3 studies.
METHODOLOGY:
- To assess the efficacy and safety of bimekizumab, an interleukin (IL)-17A and IL-17F antagonist, 320 mg for HS, researchers conducted two 48-week phase 3 trials BE HEARD I (n = 505) and II (n = 509), which enrolled patients with moderate to severe HS and a history of inadequate response to systemic antibiotics.
- Patients were randomly assigned to one of four groups: Bimekizumab every 2 weeks, bimekizumab every 2 weeks for 16 weeks followed by every 4 weeks of dosing, bimekizumab every 4 weeks, or placebo for 16 weeks followed by bimekizumab every 2 weeks.
- The primary outcome was an HS clinical response of at least 50% (HiSCR50) at week 16, defined as at least a 50% reduction in total abscess and inflammatory nodule count.
TAKEAWAY:
- A higher proportion of patients receiving bimekizumab every 2 weeks vs placebo achieved an HiSCR50 response at week 16 in BE HEARD I (48% vs 29%; odds ratio [OR], 2.23; P = .006) and II (52% vs 32%; OR, 2.29; P = .0032) trials.
- Patients receiving bimekizumab every 4 weeks also achieved a higher HiSCR50 response at week 16 vs placebo in the BE HEARD II trial (54% vs 32%; OR, 2.42; P = .0038).
- At week 16, a higher proportion of patients receiving bimekizumab every 2 weeks vs placebo achieved at least a 75% HiSCR (HiSCR75) in both trials, and a higher proportion of those receiving bimekizumab every 4 weeks achieved HiSCR75 in the BE HEARD II trial.
- At week 48, 45%-68% of patients achieved HiSCR50 in both trials.
- Patients who received bimekizumab vs placebo for the initial 16 weeks had greater improvements in patient-reported outcomes, and bimekizumab was well tolerated with a low number of serious or severe treatment-emergent adverse events.
IN PRACTICE:
“Bimekizumab was well tolerated by patients with hidradenitis suppurativa and produced rapid and deep clinically meaningful responses that were maintained up to 48 weeks,” the authors wrote. “These data support the use of bimekizumab as a promising new therapeutic option for patients with moderate to severe hidradenitis suppurativa.”
SOURCE:
Alexa B. Kimball, MD, MPH, from Beth Israel Deaconess Medical Center and Harvard Medical School, Boston, led this study, which was published online in The Lancet.
LIMITATIONS:
The placebo-controlled part of this trial was relatively short at 16 weeks and may affect the interpretation of later efficacy data, there was a lack of an active comparator group, and the efficacy of treatment was evaluated in the presence of rescue treatment with systemic antibiotics.
DISCLOSURES:
The studies were funded by bimekizumab manufacturer UCB Pharma. Seven authors disclosed being current or former employees of UCB Pharma. Other authors reported several ties with many companies, including UCB Pharma.
A version of this article first appeared on Medscape.com.
TOPLINE:
, in two phase 3 studies.
METHODOLOGY:
- To assess the efficacy and safety of bimekizumab, an interleukin (IL)-17A and IL-17F antagonist, 320 mg for HS, researchers conducted two 48-week phase 3 trials BE HEARD I (n = 505) and II (n = 509), which enrolled patients with moderate to severe HS and a history of inadequate response to systemic antibiotics.
- Patients were randomly assigned to one of four groups: Bimekizumab every 2 weeks, bimekizumab every 2 weeks for 16 weeks followed by every 4 weeks of dosing, bimekizumab every 4 weeks, or placebo for 16 weeks followed by bimekizumab every 2 weeks.
- The primary outcome was an HS clinical response of at least 50% (HiSCR50) at week 16, defined as at least a 50% reduction in total abscess and inflammatory nodule count.
TAKEAWAY:
- A higher proportion of patients receiving bimekizumab every 2 weeks vs placebo achieved an HiSCR50 response at week 16 in BE HEARD I (48% vs 29%; odds ratio [OR], 2.23; P = .006) and II (52% vs 32%; OR, 2.29; P = .0032) trials.
- Patients receiving bimekizumab every 4 weeks also achieved a higher HiSCR50 response at week 16 vs placebo in the BE HEARD II trial (54% vs 32%; OR, 2.42; P = .0038).
- At week 16, a higher proportion of patients receiving bimekizumab every 2 weeks vs placebo achieved at least a 75% HiSCR (HiSCR75) in both trials, and a higher proportion of those receiving bimekizumab every 4 weeks achieved HiSCR75 in the BE HEARD II trial.
- At week 48, 45%-68% of patients achieved HiSCR50 in both trials.
- Patients who received bimekizumab vs placebo for the initial 16 weeks had greater improvements in patient-reported outcomes, and bimekizumab was well tolerated with a low number of serious or severe treatment-emergent adverse events.
IN PRACTICE:
“Bimekizumab was well tolerated by patients with hidradenitis suppurativa and produced rapid and deep clinically meaningful responses that were maintained up to 48 weeks,” the authors wrote. “These data support the use of bimekizumab as a promising new therapeutic option for patients with moderate to severe hidradenitis suppurativa.”
SOURCE:
Alexa B. Kimball, MD, MPH, from Beth Israel Deaconess Medical Center and Harvard Medical School, Boston, led this study, which was published online in The Lancet.
LIMITATIONS:
The placebo-controlled part of this trial was relatively short at 16 weeks and may affect the interpretation of later efficacy data, there was a lack of an active comparator group, and the efficacy of treatment was evaluated in the presence of rescue treatment with systemic antibiotics.
DISCLOSURES:
The studies were funded by bimekizumab manufacturer UCB Pharma. Seven authors disclosed being current or former employees of UCB Pharma. Other authors reported several ties with many companies, including UCB Pharma.
A version of this article first appeared on Medscape.com.
Clear Coverage Preference for Humira Over Biosimilars Seen in Most Medicare Part D Plans
Despite the influx of adalimumab biosimilars entering the market in 2023, Humira remains on top.
As of January 2024, both high and low concentrations of Humira, the originator adalimumab product, are nearly universally covered by Medicare Part D plans, while only half of these plans covered adalimumab biosimilars, according to a new research letter published online on June 6, 2024, in JAMA.
Of the plans that covered both, only 1.5% had lower-tier placement for biosimilars.
“This study of formulary coverage helps explain limited uptake of adalimumab biosimilars,” wrote the authors, led by Matthew J. Klebanoff, MD, of the University of Pennsylvania, Philadelphia. “Subpar biosimilar adoption will not only undermine their potential to reduce spending but also may deter investments in biosimilar development.”
The analysis included the formulary and enrollment files for 5609 Medicare Part D plans, representing 44.4 million beneficiaries. Drug list prices and whole acquisition costs (WAC) were pulled from the Red Book database, which provides prices for prescription and over-the-counter drugs as well as medical devices and supplies.
Nearly all (98.9%) of Part D plans covered the high-concentration (100 mg/mL) version of adalimumab with a WAC of $6923. This higher concentration is the most popular formulation of the drug, making up an estimated 85% of prescriptions. By comparison, 26.8% of plans covered the high-concentration version of adalimumab-adaz (Hyrimoz), with a WAC 5% less than the reference product.
The unbranded version of adalimumab-adaz, sold at an 81% discount from the reference product, was covered by 13% of plans. Only 4.6% of plans covered high-concentration adalimumab-bwwd (Hadlima), manufactured by Samsung Bioepis.
In January 2024, no high-concentration adalimumab biosimilar had been granted interchangeability status by the US Food and Drug Administration (FDA). Adalimumab-ryvk (Simlandi) was the first biosimilar to receive this designation and was launched in late May 2024.
Coverage for the lower concentration of adalimumab was nearly universal (98.7% of plans). About half of the plans (50.7%) covered adalimumab-adbm (Cyltezo) at a 5% discount. Adalimumab-adbm (Boehringer Ingelheim) was the first interchangeable Humira biosimilar approved by the FDA, but it is only interchangeable with the less popular, lower concentration formulation of adalimumab.
All other biosimilars were covered by less than 5% of Medicare Part D plans, even with some having a WAC 86% below Humira.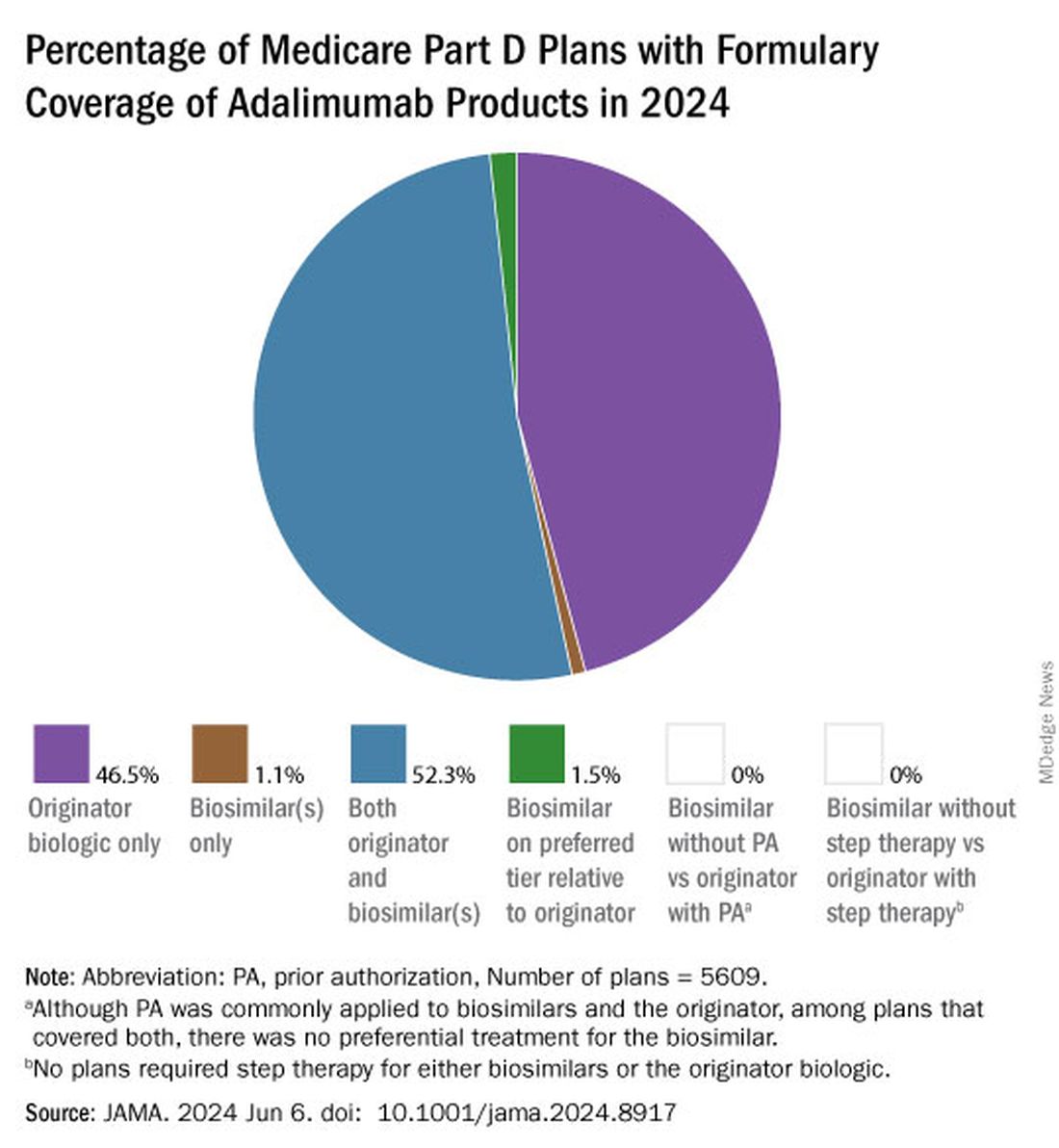
Few plans (1.5%) had biosimilars on preferred tiers compared with the reference product, and no plans used prior authorization to incentivize use of biosimilars. Most plans preferred the higher-priced version of adalimumab biosimilars, which appeals to pharmacy benefit managers who can therefore receive higher rebates, the authors noted.
“Ultimately, biosimilars’ true effect on spending will depend not on their list price but rather on their net price (after rebates) and their influence on originator biologics’ net price,” they wrote. They pointed to the 38% drop in Humira’s annual net price at the end of 2023 compared with the prior year.
“Despite this price decrease, biosimilars offer far greater potential savings: Several adalimumab biosimilars have list prices that are less than half of Humira’s net price,” the authors continued, and encouraged policy makers to mandate coverage for these lower-priced options.
Dr. Klebanoff was supported by a grant from the Health Resources and Services Administration. Two coauthors were supported by a grant from the National Institute on Aging. One author reported receiving consulting fees from AbbVie, which manufactures Humira.
A version of this article appeared on Medscape.com .
Despite the influx of adalimumab biosimilars entering the market in 2023, Humira remains on top.
As of January 2024, both high and low concentrations of Humira, the originator adalimumab product, are nearly universally covered by Medicare Part D plans, while only half of these plans covered adalimumab biosimilars, according to a new research letter published online on June 6, 2024, in JAMA.
Of the plans that covered both, only 1.5% had lower-tier placement for biosimilars.
“This study of formulary coverage helps explain limited uptake of adalimumab biosimilars,” wrote the authors, led by Matthew J. Klebanoff, MD, of the University of Pennsylvania, Philadelphia. “Subpar biosimilar adoption will not only undermine their potential to reduce spending but also may deter investments in biosimilar development.”
The analysis included the formulary and enrollment files for 5609 Medicare Part D plans, representing 44.4 million beneficiaries. Drug list prices and whole acquisition costs (WAC) were pulled from the Red Book database, which provides prices for prescription and over-the-counter drugs as well as medical devices and supplies.
Nearly all (98.9%) of Part D plans covered the high-concentration (100 mg/mL) version of adalimumab with a WAC of $6923. This higher concentration is the most popular formulation of the drug, making up an estimated 85% of prescriptions. By comparison, 26.8% of plans covered the high-concentration version of adalimumab-adaz (Hyrimoz), with a WAC 5% less than the reference product.
The unbranded version of adalimumab-adaz, sold at an 81% discount from the reference product, was covered by 13% of plans. Only 4.6% of plans covered high-concentration adalimumab-bwwd (Hadlima), manufactured by Samsung Bioepis.
In January 2024, no high-concentration adalimumab biosimilar had been granted interchangeability status by the US Food and Drug Administration (FDA). Adalimumab-ryvk (Simlandi) was the first biosimilar to receive this designation and was launched in late May 2024.
Coverage for the lower concentration of adalimumab was nearly universal (98.7% of plans). About half of the plans (50.7%) covered adalimumab-adbm (Cyltezo) at a 5% discount. Adalimumab-adbm (Boehringer Ingelheim) was the first interchangeable Humira biosimilar approved by the FDA, but it is only interchangeable with the less popular, lower concentration formulation of adalimumab.
All other biosimilars were covered by less than 5% of Medicare Part D plans, even with some having a WAC 86% below Humira.
Few plans (1.5%) had biosimilars on preferred tiers compared with the reference product, and no plans used prior authorization to incentivize use of biosimilars. Most plans preferred the higher-priced version of adalimumab biosimilars, which appeals to pharmacy benefit managers who can therefore receive higher rebates, the authors noted.
“Ultimately, biosimilars’ true effect on spending will depend not on their list price but rather on their net price (after rebates) and their influence on originator biologics’ net price,” they wrote. They pointed to the 38% drop in Humira’s annual net price at the end of 2023 compared with the prior year.
“Despite this price decrease, biosimilars offer far greater potential savings: Several adalimumab biosimilars have list prices that are less than half of Humira’s net price,” the authors continued, and encouraged policy makers to mandate coverage for these lower-priced options.
Dr. Klebanoff was supported by a grant from the Health Resources and Services Administration. Two coauthors were supported by a grant from the National Institute on Aging. One author reported receiving consulting fees from AbbVie, which manufactures Humira.
A version of this article appeared on Medscape.com .
Despite the influx of adalimumab biosimilars entering the market in 2023, Humira remains on top.
As of January 2024, both high and low concentrations of Humira, the originator adalimumab product, are nearly universally covered by Medicare Part D plans, while only half of these plans covered adalimumab biosimilars, according to a new research letter published online on June 6, 2024, in JAMA.
Of the plans that covered both, only 1.5% had lower-tier placement for biosimilars.
“This study of formulary coverage helps explain limited uptake of adalimumab biosimilars,” wrote the authors, led by Matthew J. Klebanoff, MD, of the University of Pennsylvania, Philadelphia. “Subpar biosimilar adoption will not only undermine their potential to reduce spending but also may deter investments in biosimilar development.”
The analysis included the formulary and enrollment files for 5609 Medicare Part D plans, representing 44.4 million beneficiaries. Drug list prices and whole acquisition costs (WAC) were pulled from the Red Book database, which provides prices for prescription and over-the-counter drugs as well as medical devices and supplies.
Nearly all (98.9%) of Part D plans covered the high-concentration (100 mg/mL) version of adalimumab with a WAC of $6923. This higher concentration is the most popular formulation of the drug, making up an estimated 85% of prescriptions. By comparison, 26.8% of plans covered the high-concentration version of adalimumab-adaz (Hyrimoz), with a WAC 5% less than the reference product.
The unbranded version of adalimumab-adaz, sold at an 81% discount from the reference product, was covered by 13% of plans. Only 4.6% of plans covered high-concentration adalimumab-bwwd (Hadlima), manufactured by Samsung Bioepis.
In January 2024, no high-concentration adalimumab biosimilar had been granted interchangeability status by the US Food and Drug Administration (FDA). Adalimumab-ryvk (Simlandi) was the first biosimilar to receive this designation and was launched in late May 2024.
Coverage for the lower concentration of adalimumab was nearly universal (98.7% of plans). About half of the plans (50.7%) covered adalimumab-adbm (Cyltezo) at a 5% discount. Adalimumab-adbm (Boehringer Ingelheim) was the first interchangeable Humira biosimilar approved by the FDA, but it is only interchangeable with the less popular, lower concentration formulation of adalimumab.
All other biosimilars were covered by less than 5% of Medicare Part D plans, even with some having a WAC 86% below Humira.
Few plans (1.5%) had biosimilars on preferred tiers compared with the reference product, and no plans used prior authorization to incentivize use of biosimilars. Most plans preferred the higher-priced version of adalimumab biosimilars, which appeals to pharmacy benefit managers who can therefore receive higher rebates, the authors noted.
“Ultimately, biosimilars’ true effect on spending will depend not on their list price but rather on their net price (after rebates) and their influence on originator biologics’ net price,” they wrote. They pointed to the 38% drop in Humira’s annual net price at the end of 2023 compared with the prior year.
“Despite this price decrease, biosimilars offer far greater potential savings: Several adalimumab biosimilars have list prices that are less than half of Humira’s net price,” the authors continued, and encouraged policy makers to mandate coverage for these lower-priced options.
Dr. Klebanoff was supported by a grant from the Health Resources and Services Administration. Two coauthors were supported by a grant from the National Institute on Aging. One author reported receiving consulting fees from AbbVie, which manufactures Humira.
A version of this article appeared on Medscape.com .
FROM JAMA
FDA Grants New Pediatric Arthritis Indications for Upadacitinib
Upadacitinib (Rinvoq) is now indicated for patients aged 2 years or older with active polyarticular juvenile idiopathic arthritis (pJIA) and psoriatic arthritis (PsA) who cannot tolerate or achieve adequate disease response with one or more tumor necrosis factor (TNF) blockers, according to a press release from manufacturer AbbVie.
For the youngest patients, upadacitinib is also available as a weight-based oral solution (Rinvoq LQ) in addition to the previously available tablets, according to the company. JIA, which includes pJIA and juvenile PsA, affects nearly 300,000 children and adolescents in the United States, and alternatives to TNF inhibitor (TNFi) therapy are limited, according to the company.
“Pediatric patients with pJIA and PsA can be severely limited in their ability to complete daily physical tasks and participate in everyday activities. Understanding their needs today and knowing the likelihood of disease in adulthood underscores the need for additional treatment options,” Aarat Patel, MD, a pediatric rheumatologist at Bon Secours Rheumatology Center, Richmond, Virginia, said in the press release. “Having a treatment option available for patients who do not respond well to a TNFi addresses a need for the healthcare community, patients, and their families,” he said.
Upadacitinib, a Janus kinase (JAK) inhibitor, is being studied for multiple immune-mediated inflammatory diseases. The new indication was supported by data from adults with rheumatoid arthritis (RA) and PsA, 51 pediatric patients with pJIA and active polyarthritis, and safety data from 83 pediatric patients aged 2 years to younger than 18 years with pJIA and active polyarthritis.
In the studies, the drug’s safety in pediatric patients was similar to the known safety profile in adults, which includes increased risk for serious infections such as tuberculosis, cancer, immune system problems, blood clots, and serious allergic reactions to components of the drug, according to the press release. However, the safety and effectiveness of upadacitinib for pJIA and PsA in patients younger than 2 years are unknown.
“Upadacitinib plasma exposures in pediatric patients with pJIA and PsA at the recommended dosage are predicted to be comparable to those observed in adults with RA and PsA based on population pharmacokinetic modeling and simulation,” according to the press release.
Currently, upadacitinib’s only other pediatric indication is for moderate to severe atopic dermatitis in children aged 12 years or older. Upadacitinib also is indicated for treatment of adults with moderate to severe RA, active PsA, active ankylosing spondylitis, active nonradiographic axial spondyloarthritis, and moderate to severe ulcerative colitis and Crohn’s disease, but safety and efficacy for its use in treatment of these conditions in children and adolescents is unknown.
Upadacitinib also is being studied in phase 3 trials for treatment of conditions including alopecia areata, ankylosing spondylitis, atopic dermatitis, axial spondyloarthritis, Crohn’s disease, giant cell arteritis, hidradenitis suppurativa, psoriatic arthritis, RA, systemic lupus erythematosus, Takayasu arteritis, ulcerative colitis, and vitiligo, according to the press release.
Full prescribing information and safety data for upadacitinib are available here.
A version of this article appeared on Medscape.com.
Upadacitinib (Rinvoq) is now indicated for patients aged 2 years or older with active polyarticular juvenile idiopathic arthritis (pJIA) and psoriatic arthritis (PsA) who cannot tolerate or achieve adequate disease response with one or more tumor necrosis factor (TNF) blockers, according to a press release from manufacturer AbbVie.
For the youngest patients, upadacitinib is also available as a weight-based oral solution (Rinvoq LQ) in addition to the previously available tablets, according to the company. JIA, which includes pJIA and juvenile PsA, affects nearly 300,000 children and adolescents in the United States, and alternatives to TNF inhibitor (TNFi) therapy are limited, according to the company.
“Pediatric patients with pJIA and PsA can be severely limited in their ability to complete daily physical tasks and participate in everyday activities. Understanding their needs today and knowing the likelihood of disease in adulthood underscores the need for additional treatment options,” Aarat Patel, MD, a pediatric rheumatologist at Bon Secours Rheumatology Center, Richmond, Virginia, said in the press release. “Having a treatment option available for patients who do not respond well to a TNFi addresses a need for the healthcare community, patients, and their families,” he said.
Upadacitinib, a Janus kinase (JAK) inhibitor, is being studied for multiple immune-mediated inflammatory diseases. The new indication was supported by data from adults with rheumatoid arthritis (RA) and PsA, 51 pediatric patients with pJIA and active polyarthritis, and safety data from 83 pediatric patients aged 2 years to younger than 18 years with pJIA and active polyarthritis.
In the studies, the drug’s safety in pediatric patients was similar to the known safety profile in adults, which includes increased risk for serious infections such as tuberculosis, cancer, immune system problems, blood clots, and serious allergic reactions to components of the drug, according to the press release. However, the safety and effectiveness of upadacitinib for pJIA and PsA in patients younger than 2 years are unknown.
“Upadacitinib plasma exposures in pediatric patients with pJIA and PsA at the recommended dosage are predicted to be comparable to those observed in adults with RA and PsA based on population pharmacokinetic modeling and simulation,” according to the press release.
Currently, upadacitinib’s only other pediatric indication is for moderate to severe atopic dermatitis in children aged 12 years or older. Upadacitinib also is indicated for treatment of adults with moderate to severe RA, active PsA, active ankylosing spondylitis, active nonradiographic axial spondyloarthritis, and moderate to severe ulcerative colitis and Crohn’s disease, but safety and efficacy for its use in treatment of these conditions in children and adolescents is unknown.
Upadacitinib also is being studied in phase 3 trials for treatment of conditions including alopecia areata, ankylosing spondylitis, atopic dermatitis, axial spondyloarthritis, Crohn’s disease, giant cell arteritis, hidradenitis suppurativa, psoriatic arthritis, RA, systemic lupus erythematosus, Takayasu arteritis, ulcerative colitis, and vitiligo, according to the press release.
Full prescribing information and safety data for upadacitinib are available here.
A version of this article appeared on Medscape.com.
Upadacitinib (Rinvoq) is now indicated for patients aged 2 years or older with active polyarticular juvenile idiopathic arthritis (pJIA) and psoriatic arthritis (PsA) who cannot tolerate or achieve adequate disease response with one or more tumor necrosis factor (TNF) blockers, according to a press release from manufacturer AbbVie.
For the youngest patients, upadacitinib is also available as a weight-based oral solution (Rinvoq LQ) in addition to the previously available tablets, according to the company. JIA, which includes pJIA and juvenile PsA, affects nearly 300,000 children and adolescents in the United States, and alternatives to TNF inhibitor (TNFi) therapy are limited, according to the company.
“Pediatric patients with pJIA and PsA can be severely limited in their ability to complete daily physical tasks and participate in everyday activities. Understanding their needs today and knowing the likelihood of disease in adulthood underscores the need for additional treatment options,” Aarat Patel, MD, a pediatric rheumatologist at Bon Secours Rheumatology Center, Richmond, Virginia, said in the press release. “Having a treatment option available for patients who do not respond well to a TNFi addresses a need for the healthcare community, patients, and their families,” he said.
Upadacitinib, a Janus kinase (JAK) inhibitor, is being studied for multiple immune-mediated inflammatory diseases. The new indication was supported by data from adults with rheumatoid arthritis (RA) and PsA, 51 pediatric patients with pJIA and active polyarthritis, and safety data from 83 pediatric patients aged 2 years to younger than 18 years with pJIA and active polyarthritis.
In the studies, the drug’s safety in pediatric patients was similar to the known safety profile in adults, which includes increased risk for serious infections such as tuberculosis, cancer, immune system problems, blood clots, and serious allergic reactions to components of the drug, according to the press release. However, the safety and effectiveness of upadacitinib for pJIA and PsA in patients younger than 2 years are unknown.
“Upadacitinib plasma exposures in pediatric patients with pJIA and PsA at the recommended dosage are predicted to be comparable to those observed in adults with RA and PsA based on population pharmacokinetic modeling and simulation,” according to the press release.
Currently, upadacitinib’s only other pediatric indication is for moderate to severe atopic dermatitis in children aged 12 years or older. Upadacitinib also is indicated for treatment of adults with moderate to severe RA, active PsA, active ankylosing spondylitis, active nonradiographic axial spondyloarthritis, and moderate to severe ulcerative colitis and Crohn’s disease, but safety and efficacy for its use in treatment of these conditions in children and adolescents is unknown.
Upadacitinib also is being studied in phase 3 trials for treatment of conditions including alopecia areata, ankylosing spondylitis, atopic dermatitis, axial spondyloarthritis, Crohn’s disease, giant cell arteritis, hidradenitis suppurativa, psoriatic arthritis, RA, systemic lupus erythematosus, Takayasu arteritis, ulcerative colitis, and vitiligo, according to the press release.
Full prescribing information and safety data for upadacitinib are available here.
A version of this article appeared on Medscape.com.
High Sodium Intake Linked to Greater Risk for Eczema
In a study of adults, an increase of 1 g in estimated 24-hour urinary sodium excretion was associated with 11% higher odds of an atopic dermatitis (AD) diagnosis, 16% higher odds of having active AD, and 11% higher odds of increased severity of AD.
Those are key findings from a cross-sectional analysis of data from the United Kingdom.
“Excessive dietary sodium, common in fast food, may be associated with AD,” corresponding author Katrina Abuabara, MD, MA, MSCE, and colleagues wrote in the study, which was published online in JAMA Dermatology. They referred to recent research using sodium MRI, which showed that “the majority of the body’s exchangeable sodium is stored in the skin and that skin sodium is associated with autoimmune and chronic inflammatory conditions, including AD.” And in another study published in 2019, lesional skin sodium was 30-fold greater in patients with AD than in healthy controls.
To investigate whether there is an association between higher levels of sodium consumption and AD prevalence, activity, and severity at the population level, Dr. Abuabara, of the program for clinical research in the Department of Dermatology at the University of California, San Francisco, and coauthors drew from the UK Biobank, a population-based cohort of more than 500,000 individuals aged 37-73 years at the time of recruitment by the National Health Service. The primary exposure was 24-hour urinary sodium excretion, which was calculated by using the INTERSALT equation, a sex-specific estimation that incorporates body mass index; age; and urine concentrations of potassium, sodium, and creatinine. The primary study outcome was AD or active AD based on diagnostic and prescription codes from linked electronic medical records. The researchers used multivariable logistic regression models adjusted for age, sex, race and ethnicity, Townsend deprivation index, and education to measure the association.
Of the 215,832 Biobank participants included in the analysis, 54% were female, their mean age was 57 years, 95% were White, their mean estimated 24-hour urine sodium excretion was 3.01 g/day, and 10,839 (5%) had a diagnosis of AD. The researchers observed that on multivariable logistic regression, a 1-g increase in estimated 24-hour urine sodium excretion was associated with increased odds of AD (adjusted odds ratio [AOR], 1.11; 95% CI, 1.07-1.14), increased odds of active AD (AOR, 1.16; 95% CI, 1.05-1.28), and increased odds of increasing severity of AD (AOR, 1.11; 95% CI, 1.07-1.15).
Validating Results With US Data
To validate the findings, the researchers evaluated a cohort of 13,014 participants from the US-based National Health and Nutrition Examination Survey (NHANES), using pooled data from the 1999-2000, 2001-2002, and 2003-2004 samples. Of the 13,014 participants, 796 reported current AD, and 1493 reported AD in the past year. The mean dietary sodium intake of overall NHANES participants estimated with 24-hour dietary recall questionnaires was 3.45 g, with a mean of 3.47 g for those with current AD and a mean of 3.44 g for those without AD.
The researchers observed that a 1-g/day higher dietary sodium intake was associated with a higher risk for current AD (AOR, 1.22; 95%CI, 1.01-1.47) and a somewhat higher risk for AD in the past year (AOR, 1.14; 95% CI, 0.97-1.35).
“Future work should examine whether variation of sodium intake over time might trigger AD flares and whether it helps to explain heterogeneity in response to new immunomodulatory treatments for AD,” the authors wrote. “Reduced sodium intake was recommended as a treatment for AD more than a century ago, but there have yet to be studies examining the association of dietary sodium reduction with skin sodium concentration or AD severity,” they added. Noting that sodium reduction “has been shown to be a cost-effective intervention for hypertension and other cardiovascular disease outcomes,” they said that their data “support experimental studies of this approach in AD.”
They acknowledged certain limitations of the study, including the fact that a single spot urine sample was used in the UK Biobank cohort, “which only captures dietary intake of the last 24 hours and is not the best measure of usual or long-term intake of sodium.” They also noted that the findings may not be generalizable to other populations and that AD was based on self-report in the NHANES validation cohort.
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was asked to comment on the results, said the study by Dr. Abuabara and colleagues “gives us another reason to avoid salt, showing that 1 g/day of higher salt intake increases the risk of AD in an adult population and more severe AD.”
He added that, “Now, can you say that reducing salt intake will have a therapeutic effect or clinically relevant impact? No. [That is] certainly worth exploring but at a minimum, gives some more credibility to keeping it bland.”
The study was supported by a grant from the Medical Student in Aging Research Program, the National Institute on Aging, and the National Eczema Association. Dr. Abuabara reported receiving research funding for her institution from Pfizer and Cosmetique Internacional/La Roche-Posay and consulting fees from Target RWE, Sanofi, Nektar, and Amgen. No other disclosures were reported. Dr. Friedman had no relevant disclosures.
A version of this article appeared on Medscape.com.
In a study of adults, an increase of 1 g in estimated 24-hour urinary sodium excretion was associated with 11% higher odds of an atopic dermatitis (AD) diagnosis, 16% higher odds of having active AD, and 11% higher odds of increased severity of AD.
Those are key findings from a cross-sectional analysis of data from the United Kingdom.
“Excessive dietary sodium, common in fast food, may be associated with AD,” corresponding author Katrina Abuabara, MD, MA, MSCE, and colleagues wrote in the study, which was published online in JAMA Dermatology. They referred to recent research using sodium MRI, which showed that “the majority of the body’s exchangeable sodium is stored in the skin and that skin sodium is associated with autoimmune and chronic inflammatory conditions, including AD.” And in another study published in 2019, lesional skin sodium was 30-fold greater in patients with AD than in healthy controls.
To investigate whether there is an association between higher levels of sodium consumption and AD prevalence, activity, and severity at the population level, Dr. Abuabara, of the program for clinical research in the Department of Dermatology at the University of California, San Francisco, and coauthors drew from the UK Biobank, a population-based cohort of more than 500,000 individuals aged 37-73 years at the time of recruitment by the National Health Service. The primary exposure was 24-hour urinary sodium excretion, which was calculated by using the INTERSALT equation, a sex-specific estimation that incorporates body mass index; age; and urine concentrations of potassium, sodium, and creatinine. The primary study outcome was AD or active AD based on diagnostic and prescription codes from linked electronic medical records. The researchers used multivariable logistic regression models adjusted for age, sex, race and ethnicity, Townsend deprivation index, and education to measure the association.
Of the 215,832 Biobank participants included in the analysis, 54% were female, their mean age was 57 years, 95% were White, their mean estimated 24-hour urine sodium excretion was 3.01 g/day, and 10,839 (5%) had a diagnosis of AD. The researchers observed that on multivariable logistic regression, a 1-g increase in estimated 24-hour urine sodium excretion was associated with increased odds of AD (adjusted odds ratio [AOR], 1.11; 95% CI, 1.07-1.14), increased odds of active AD (AOR, 1.16; 95% CI, 1.05-1.28), and increased odds of increasing severity of AD (AOR, 1.11; 95% CI, 1.07-1.15).
Validating Results With US Data
To validate the findings, the researchers evaluated a cohort of 13,014 participants from the US-based National Health and Nutrition Examination Survey (NHANES), using pooled data from the 1999-2000, 2001-2002, and 2003-2004 samples. Of the 13,014 participants, 796 reported current AD, and 1493 reported AD in the past year. The mean dietary sodium intake of overall NHANES participants estimated with 24-hour dietary recall questionnaires was 3.45 g, with a mean of 3.47 g for those with current AD and a mean of 3.44 g for those without AD.
The researchers observed that a 1-g/day higher dietary sodium intake was associated with a higher risk for current AD (AOR, 1.22; 95%CI, 1.01-1.47) and a somewhat higher risk for AD in the past year (AOR, 1.14; 95% CI, 0.97-1.35).
“Future work should examine whether variation of sodium intake over time might trigger AD flares and whether it helps to explain heterogeneity in response to new immunomodulatory treatments for AD,” the authors wrote. “Reduced sodium intake was recommended as a treatment for AD more than a century ago, but there have yet to be studies examining the association of dietary sodium reduction with skin sodium concentration or AD severity,” they added. Noting that sodium reduction “has been shown to be a cost-effective intervention for hypertension and other cardiovascular disease outcomes,” they said that their data “support experimental studies of this approach in AD.”
They acknowledged certain limitations of the study, including the fact that a single spot urine sample was used in the UK Biobank cohort, “which only captures dietary intake of the last 24 hours and is not the best measure of usual or long-term intake of sodium.” They also noted that the findings may not be generalizable to other populations and that AD was based on self-report in the NHANES validation cohort.
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was asked to comment on the results, said the study by Dr. Abuabara and colleagues “gives us another reason to avoid salt, showing that 1 g/day of higher salt intake increases the risk of AD in an adult population and more severe AD.”
He added that, “Now, can you say that reducing salt intake will have a therapeutic effect or clinically relevant impact? No. [That is] certainly worth exploring but at a minimum, gives some more credibility to keeping it bland.”
The study was supported by a grant from the Medical Student in Aging Research Program, the National Institute on Aging, and the National Eczema Association. Dr. Abuabara reported receiving research funding for her institution from Pfizer and Cosmetique Internacional/La Roche-Posay and consulting fees from Target RWE, Sanofi, Nektar, and Amgen. No other disclosures were reported. Dr. Friedman had no relevant disclosures.
A version of this article appeared on Medscape.com.
In a study of adults, an increase of 1 g in estimated 24-hour urinary sodium excretion was associated with 11% higher odds of an atopic dermatitis (AD) diagnosis, 16% higher odds of having active AD, and 11% higher odds of increased severity of AD.
Those are key findings from a cross-sectional analysis of data from the United Kingdom.
“Excessive dietary sodium, common in fast food, may be associated with AD,” corresponding author Katrina Abuabara, MD, MA, MSCE, and colleagues wrote in the study, which was published online in JAMA Dermatology. They referred to recent research using sodium MRI, which showed that “the majority of the body’s exchangeable sodium is stored in the skin and that skin sodium is associated with autoimmune and chronic inflammatory conditions, including AD.” And in another study published in 2019, lesional skin sodium was 30-fold greater in patients with AD than in healthy controls.
To investigate whether there is an association between higher levels of sodium consumption and AD prevalence, activity, and severity at the population level, Dr. Abuabara, of the program for clinical research in the Department of Dermatology at the University of California, San Francisco, and coauthors drew from the UK Biobank, a population-based cohort of more than 500,000 individuals aged 37-73 years at the time of recruitment by the National Health Service. The primary exposure was 24-hour urinary sodium excretion, which was calculated by using the INTERSALT equation, a sex-specific estimation that incorporates body mass index; age; and urine concentrations of potassium, sodium, and creatinine. The primary study outcome was AD or active AD based on diagnostic and prescription codes from linked electronic medical records. The researchers used multivariable logistic regression models adjusted for age, sex, race and ethnicity, Townsend deprivation index, and education to measure the association.
Of the 215,832 Biobank participants included in the analysis, 54% were female, their mean age was 57 years, 95% were White, their mean estimated 24-hour urine sodium excretion was 3.01 g/day, and 10,839 (5%) had a diagnosis of AD. The researchers observed that on multivariable logistic regression, a 1-g increase in estimated 24-hour urine sodium excretion was associated with increased odds of AD (adjusted odds ratio [AOR], 1.11; 95% CI, 1.07-1.14), increased odds of active AD (AOR, 1.16; 95% CI, 1.05-1.28), and increased odds of increasing severity of AD (AOR, 1.11; 95% CI, 1.07-1.15).
Validating Results With US Data
To validate the findings, the researchers evaluated a cohort of 13,014 participants from the US-based National Health and Nutrition Examination Survey (NHANES), using pooled data from the 1999-2000, 2001-2002, and 2003-2004 samples. Of the 13,014 participants, 796 reported current AD, and 1493 reported AD in the past year. The mean dietary sodium intake of overall NHANES participants estimated with 24-hour dietary recall questionnaires was 3.45 g, with a mean of 3.47 g for those with current AD and a mean of 3.44 g for those without AD.
The researchers observed that a 1-g/day higher dietary sodium intake was associated with a higher risk for current AD (AOR, 1.22; 95%CI, 1.01-1.47) and a somewhat higher risk for AD in the past year (AOR, 1.14; 95% CI, 0.97-1.35).
“Future work should examine whether variation of sodium intake over time might trigger AD flares and whether it helps to explain heterogeneity in response to new immunomodulatory treatments for AD,” the authors wrote. “Reduced sodium intake was recommended as a treatment for AD more than a century ago, but there have yet to be studies examining the association of dietary sodium reduction with skin sodium concentration or AD severity,” they added. Noting that sodium reduction “has been shown to be a cost-effective intervention for hypertension and other cardiovascular disease outcomes,” they said that their data “support experimental studies of this approach in AD.”
They acknowledged certain limitations of the study, including the fact that a single spot urine sample was used in the UK Biobank cohort, “which only captures dietary intake of the last 24 hours and is not the best measure of usual or long-term intake of sodium.” They also noted that the findings may not be generalizable to other populations and that AD was based on self-report in the NHANES validation cohort.
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was asked to comment on the results, said the study by Dr. Abuabara and colleagues “gives us another reason to avoid salt, showing that 1 g/day of higher salt intake increases the risk of AD in an adult population and more severe AD.”
He added that, “Now, can you say that reducing salt intake will have a therapeutic effect or clinically relevant impact? No. [That is] certainly worth exploring but at a minimum, gives some more credibility to keeping it bland.”
The study was supported by a grant from the Medical Student in Aging Research Program, the National Institute on Aging, and the National Eczema Association. Dr. Abuabara reported receiving research funding for her institution from Pfizer and Cosmetique Internacional/La Roche-Posay and consulting fees from Target RWE, Sanofi, Nektar, and Amgen. No other disclosures were reported. Dr. Friedman had no relevant disclosures.
A version of this article appeared on Medscape.com.
Losing Weight, Decreasing Alcohol, and Improving Sex Life?
Richard* was a master-of-the-universe type. He went to Wharton, ran a large hedge fund, and lived in Greenwich, Connecticut. His three children attended Ivy League schools. He played golf on the weekends and ate three healthy meals per day. There was just one issue: He had gained 90 pounds since the 1990s from consuming six to seven alcoholic beverages per day. He already had one DUI under his belt, and his marriage was on shaky ground. He had tried to address his alcohol abuse disorder on multiple occasions: He went to a yearlong class on alcoholism, saw a psychologist for cognitive-behavioral therapy, and joined Alcoholics Anonymous, all to no avail.
When I met him in December 2023, he had hit rock bottom and was willing to try anything.
At our first visit, I prescribed him weekly tirzepatide (Zepbound) off label, along with a small dose of naltrexone.
Richard shared some feedback after his first 2 weeks:
The naltrexone works great and is strong ... small dose for me effective ... I haven’t wanted to drink and when I do I can’t finish a glass over 2 hours … went from 25 drinks a week to about 4 … don’t notice other side effects … sleeping better too.
And after 6 weeks:
Some more feedback … on week 6-7 and all going well ... drinking very little alcohol and still on half tab of naltrexone ... that works well and have no side effects ... the Zepbound works well too. I do get hungry a few days after the shot but still don’t crave sugar or bad snacks … weight down 21 pounds since started … 292 to 271.
And finally, after 8 weeks:
Looking at my last text to you I see the progress … been incredible ... now down 35 pounds and at 257 … continue to feel excellent with plenty of energy … want to exercise more ... and no temptation to eat or drink unhealthy stuff ... I’m very happy this has surpassed my expectations on how fast it’s worked and I don’t feel any side effects. Marriage has never been better … all thanks to you.
Tirzepatide contains two hormones, glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP), that are naturally produced by our bodies after meals. Scientists recently learned that the GLP-1 system contributes to the feedback loop of addictive behaviors. Increasing synthetic GLP-1, through medications like tirzepatide, appears to minimize addictive behaviors by limiting their ability to upregulate the brain’s production of dopamine.
Dopamine is a neurotransmitter produced in the brain’s reward center, which regulates how people experience pleasure and control impulses. Dopamine reinforces the pleasure experienced by certain behaviors like drinking, smoking, and eating sweets. These new medications reduce the amount of dopamine released after these activities and thereby lower the motivation to repeat these behaviors.
Contrary to some reports in the news, the vast majority of my male patients using these medications for alcohol abuse disorder experience concurrent increases in testosterone, for two reasons: (1) testosterone increases as body mass index decreases and (2) chronic alcohol use can damage the cells in the testicles that produce testosterone and also decrease the brain’s ability to stimulate the testicles to produce testosterone.
At his most recent checkup last month, Richard’s testosterone had risen from borderline to robust levels, his libido and sleep had improved, and he reported never having felt so healthy or confident. Fingers crossed that the US Food and Drug Administration won’t wait too long before approving this class of medications for more than just diabetes, heart disease, and obesity.
*Patient’s name has been changed.
Dr. Messer is clinical assistant professor, Icahn School of Medicine at Mount Sinai, New York, and associate professor, Zucker School of Medicine at Hofstra University, Hempstead, New York. She has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Richard* was a master-of-the-universe type. He went to Wharton, ran a large hedge fund, and lived in Greenwich, Connecticut. His three children attended Ivy League schools. He played golf on the weekends and ate three healthy meals per day. There was just one issue: He had gained 90 pounds since the 1990s from consuming six to seven alcoholic beverages per day. He already had one DUI under his belt, and his marriage was on shaky ground. He had tried to address his alcohol abuse disorder on multiple occasions: He went to a yearlong class on alcoholism, saw a psychologist for cognitive-behavioral therapy, and joined Alcoholics Anonymous, all to no avail.
When I met him in December 2023, he had hit rock bottom and was willing to try anything.
At our first visit, I prescribed him weekly tirzepatide (Zepbound) off label, along with a small dose of naltrexone.
Richard shared some feedback after his first 2 weeks:
The naltrexone works great and is strong ... small dose for me effective ... I haven’t wanted to drink and when I do I can’t finish a glass over 2 hours … went from 25 drinks a week to about 4 … don’t notice other side effects … sleeping better too.
And after 6 weeks:
Some more feedback … on week 6-7 and all going well ... drinking very little alcohol and still on half tab of naltrexone ... that works well and have no side effects ... the Zepbound works well too. I do get hungry a few days after the shot but still don’t crave sugar or bad snacks … weight down 21 pounds since started … 292 to 271.
And finally, after 8 weeks:
Looking at my last text to you I see the progress … been incredible ... now down 35 pounds and at 257 … continue to feel excellent with plenty of energy … want to exercise more ... and no temptation to eat or drink unhealthy stuff ... I’m very happy this has surpassed my expectations on how fast it’s worked and I don’t feel any side effects. Marriage has never been better … all thanks to you.
Tirzepatide contains two hormones, glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP), that are naturally produced by our bodies after meals. Scientists recently learned that the GLP-1 system contributes to the feedback loop of addictive behaviors. Increasing synthetic GLP-1, through medications like tirzepatide, appears to minimize addictive behaviors by limiting their ability to upregulate the brain’s production of dopamine.
Dopamine is a neurotransmitter produced in the brain’s reward center, which regulates how people experience pleasure and control impulses. Dopamine reinforces the pleasure experienced by certain behaviors like drinking, smoking, and eating sweets. These new medications reduce the amount of dopamine released after these activities and thereby lower the motivation to repeat these behaviors.
Contrary to some reports in the news, the vast majority of my male patients using these medications for alcohol abuse disorder experience concurrent increases in testosterone, for two reasons: (1) testosterone increases as body mass index decreases and (2) chronic alcohol use can damage the cells in the testicles that produce testosterone and also decrease the brain’s ability to stimulate the testicles to produce testosterone.
At his most recent checkup last month, Richard’s testosterone had risen from borderline to robust levels, his libido and sleep had improved, and he reported never having felt so healthy or confident. Fingers crossed that the US Food and Drug Administration won’t wait too long before approving this class of medications for more than just diabetes, heart disease, and obesity.
*Patient’s name has been changed.
Dr. Messer is clinical assistant professor, Icahn School of Medicine at Mount Sinai, New York, and associate professor, Zucker School of Medicine at Hofstra University, Hempstead, New York. She has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Richard* was a master-of-the-universe type. He went to Wharton, ran a large hedge fund, and lived in Greenwich, Connecticut. His three children attended Ivy League schools. He played golf on the weekends and ate three healthy meals per day. There was just one issue: He had gained 90 pounds since the 1990s from consuming six to seven alcoholic beverages per day. He already had one DUI under his belt, and his marriage was on shaky ground. He had tried to address his alcohol abuse disorder on multiple occasions: He went to a yearlong class on alcoholism, saw a psychologist for cognitive-behavioral therapy, and joined Alcoholics Anonymous, all to no avail.
When I met him in December 2023, he had hit rock bottom and was willing to try anything.
At our first visit, I prescribed him weekly tirzepatide (Zepbound) off label, along with a small dose of naltrexone.
Richard shared some feedback after his first 2 weeks:
The naltrexone works great and is strong ... small dose for me effective ... I haven’t wanted to drink and when I do I can’t finish a glass over 2 hours … went from 25 drinks a week to about 4 … don’t notice other side effects … sleeping better too.
And after 6 weeks:
Some more feedback … on week 6-7 and all going well ... drinking very little alcohol and still on half tab of naltrexone ... that works well and have no side effects ... the Zepbound works well too. I do get hungry a few days after the shot but still don’t crave sugar or bad snacks … weight down 21 pounds since started … 292 to 271.
And finally, after 8 weeks:
Looking at my last text to you I see the progress … been incredible ... now down 35 pounds and at 257 … continue to feel excellent with plenty of energy … want to exercise more ... and no temptation to eat or drink unhealthy stuff ... I’m very happy this has surpassed my expectations on how fast it’s worked and I don’t feel any side effects. Marriage has never been better … all thanks to you.
Tirzepatide contains two hormones, glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP), that are naturally produced by our bodies after meals. Scientists recently learned that the GLP-1 system contributes to the feedback loop of addictive behaviors. Increasing synthetic GLP-1, through medications like tirzepatide, appears to minimize addictive behaviors by limiting their ability to upregulate the brain’s production of dopamine.
Dopamine is a neurotransmitter produced in the brain’s reward center, which regulates how people experience pleasure and control impulses. Dopamine reinforces the pleasure experienced by certain behaviors like drinking, smoking, and eating sweets. These new medications reduce the amount of dopamine released after these activities and thereby lower the motivation to repeat these behaviors.
Contrary to some reports in the news, the vast majority of my male patients using these medications for alcohol abuse disorder experience concurrent increases in testosterone, for two reasons: (1) testosterone increases as body mass index decreases and (2) chronic alcohol use can damage the cells in the testicles that produce testosterone and also decrease the brain’s ability to stimulate the testicles to produce testosterone.
At his most recent checkup last month, Richard’s testosterone had risen from borderline to robust levels, his libido and sleep had improved, and he reported never having felt so healthy or confident. Fingers crossed that the US Food and Drug Administration won’t wait too long before approving this class of medications for more than just diabetes, heart disease, and obesity.
*Patient’s name has been changed.
Dr. Messer is clinical assistant professor, Icahn School of Medicine at Mount Sinai, New York, and associate professor, Zucker School of Medicine at Hofstra University, Hempstead, New York. She has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Study Finds Mace Risk Remains High in Patients with Psoriasis, Dyslipidemia
Over a period of 5 years, the, even after adjusting for covariates, results from a large retrospective study showed.
“It is well-established that psoriasis is an independent risk factor for the development of MACE, with cardiometabolic risk factors being more prevalent and incident among patients with psoriasis,” the study’s first author Ana Ormaza Vera, MD, a dermatology research fellow at Eastern Virginia Medical School, Norfolk, said in an interview after the annual meeting of the Society for Investigational Dermatology, where the study was presented during a late-breaking abstract session.
Current guidelines from the joint American Academy of Dermatology/National Psoriasis Foundation and the American Academy of Cardiology/American Heart Association Task Force recommend statins, a lipid-lowering and anti-inflammatory therapy, “for patients with psoriasis who have additional risk-enhancing factors, similar to recommendations made for the general population without psoriasis,” she noted. But how the incidence of MACE differs between patients with and without psoriasis while on statin therapy “has not been explored in real-world settings,” she added.
To address this question, the researchers used real-world data from the TriNetX health research network to identify individuals aged 18-90 years with a diagnosis of both psoriasis and lipid disorders who were undergoing treatment with statins. Those with a prior history of MACE were excluded from the analysis. Patients with lipid disorders on statin therapy, but without psoriatic disease, were matched 1:1 by age, sex, race, ethnicity, common risk factors for MACE, and medications shown to reduce MACE risk. The researchers then assessed the cohorts 5 years following their first statin prescription and used the TriNetX analytics tool to calculate the odds ratio (OR) with 95% CI to evaluate the likelihood of MACE in the presence of statin therapy.
Dr. Ormaza Vera and colleagues identified 20,660 patients with psoriasis and 2,768,429 patients without psoriasis who met the criteria for analysis. After propensity score matching, each cohort included 20,660 patients with a mean age of 60 years. During the 5-year observation period, 2725 patients in the psoriasis cohort experienced MACE compared with 2203 patients in the non-psoriasis cohort (OR, 1.40; 95% CI, 1.317-1.488).
“This was an unexpected outcome that challenges the current understanding and highlights the need for further research into tailored treatments for cardiovascular risk in psoriasis patients,” Dr. Ormaza Vera told this news organization.
She acknowledged certain limitations of the study, including its retrospective design, the inherent limitations of an observational study, and the use of electronic medical record data.
Lawrence J. Green, MD, clinical professor of dermatology, George Washington University, Washington, who was asked to comment on the study results, said that the findings imply that there is more than statin use alone to protect someone with psoriasis from having an increased risk for MACE. “This is not really surprising because statin use alone is only part of a prevention strategy in someone with psoriasis who usually has multiple comorbidities,” Dr. Green said. “On the other hand, the study only went out for 5 years and cardiovascular disease is a long accumulating process, so it could also be too early to demonstrate MACE prevention.”
The study was funded by a grant from the American Skin Association. Dr. Ormaza Vera and her coauthors reported having no relevant disclosures. Dr. Green disclosed that he is a speaker, consultant, or investigator for many pharmaceutical companies.
A version of this article appeared on Medscape.com .
Over a period of 5 years, the, even after adjusting for covariates, results from a large retrospective study showed.
“It is well-established that psoriasis is an independent risk factor for the development of MACE, with cardiometabolic risk factors being more prevalent and incident among patients with psoriasis,” the study’s first author Ana Ormaza Vera, MD, a dermatology research fellow at Eastern Virginia Medical School, Norfolk, said in an interview after the annual meeting of the Society for Investigational Dermatology, where the study was presented during a late-breaking abstract session.
Current guidelines from the joint American Academy of Dermatology/National Psoriasis Foundation and the American Academy of Cardiology/American Heart Association Task Force recommend statins, a lipid-lowering and anti-inflammatory therapy, “for patients with psoriasis who have additional risk-enhancing factors, similar to recommendations made for the general population without psoriasis,” she noted. But how the incidence of MACE differs between patients with and without psoriasis while on statin therapy “has not been explored in real-world settings,” she added.
To address this question, the researchers used real-world data from the TriNetX health research network to identify individuals aged 18-90 years with a diagnosis of both psoriasis and lipid disorders who were undergoing treatment with statins. Those with a prior history of MACE were excluded from the analysis. Patients with lipid disorders on statin therapy, but without psoriatic disease, were matched 1:1 by age, sex, race, ethnicity, common risk factors for MACE, and medications shown to reduce MACE risk. The researchers then assessed the cohorts 5 years following their first statin prescription and used the TriNetX analytics tool to calculate the odds ratio (OR) with 95% CI to evaluate the likelihood of MACE in the presence of statin therapy.
Dr. Ormaza Vera and colleagues identified 20,660 patients with psoriasis and 2,768,429 patients without psoriasis who met the criteria for analysis. After propensity score matching, each cohort included 20,660 patients with a mean age of 60 years. During the 5-year observation period, 2725 patients in the psoriasis cohort experienced MACE compared with 2203 patients in the non-psoriasis cohort (OR, 1.40; 95% CI, 1.317-1.488).
“This was an unexpected outcome that challenges the current understanding and highlights the need for further research into tailored treatments for cardiovascular risk in psoriasis patients,” Dr. Ormaza Vera told this news organization.
She acknowledged certain limitations of the study, including its retrospective design, the inherent limitations of an observational study, and the use of electronic medical record data.
Lawrence J. Green, MD, clinical professor of dermatology, George Washington University, Washington, who was asked to comment on the study results, said that the findings imply that there is more than statin use alone to protect someone with psoriasis from having an increased risk for MACE. “This is not really surprising because statin use alone is only part of a prevention strategy in someone with psoriasis who usually has multiple comorbidities,” Dr. Green said. “On the other hand, the study only went out for 5 years and cardiovascular disease is a long accumulating process, so it could also be too early to demonstrate MACE prevention.”
The study was funded by a grant from the American Skin Association. Dr. Ormaza Vera and her coauthors reported having no relevant disclosures. Dr. Green disclosed that he is a speaker, consultant, or investigator for many pharmaceutical companies.
A version of this article appeared on Medscape.com .
Over a period of 5 years, the, even after adjusting for covariates, results from a large retrospective study showed.
“It is well-established that psoriasis is an independent risk factor for the development of MACE, with cardiometabolic risk factors being more prevalent and incident among patients with psoriasis,” the study’s first author Ana Ormaza Vera, MD, a dermatology research fellow at Eastern Virginia Medical School, Norfolk, said in an interview after the annual meeting of the Society for Investigational Dermatology, where the study was presented during a late-breaking abstract session.
Current guidelines from the joint American Academy of Dermatology/National Psoriasis Foundation and the American Academy of Cardiology/American Heart Association Task Force recommend statins, a lipid-lowering and anti-inflammatory therapy, “for patients with psoriasis who have additional risk-enhancing factors, similar to recommendations made for the general population without psoriasis,” she noted. But how the incidence of MACE differs between patients with and without psoriasis while on statin therapy “has not been explored in real-world settings,” she added.
To address this question, the researchers used real-world data from the TriNetX health research network to identify individuals aged 18-90 years with a diagnosis of both psoriasis and lipid disorders who were undergoing treatment with statins. Those with a prior history of MACE were excluded from the analysis. Patients with lipid disorders on statin therapy, but without psoriatic disease, were matched 1:1 by age, sex, race, ethnicity, common risk factors for MACE, and medications shown to reduce MACE risk. The researchers then assessed the cohorts 5 years following their first statin prescription and used the TriNetX analytics tool to calculate the odds ratio (OR) with 95% CI to evaluate the likelihood of MACE in the presence of statin therapy.
Dr. Ormaza Vera and colleagues identified 20,660 patients with psoriasis and 2,768,429 patients without psoriasis who met the criteria for analysis. After propensity score matching, each cohort included 20,660 patients with a mean age of 60 years. During the 5-year observation period, 2725 patients in the psoriasis cohort experienced MACE compared with 2203 patients in the non-psoriasis cohort (OR, 1.40; 95% CI, 1.317-1.488).
“This was an unexpected outcome that challenges the current understanding and highlights the need for further research into tailored treatments for cardiovascular risk in psoriasis patients,” Dr. Ormaza Vera told this news organization.
She acknowledged certain limitations of the study, including its retrospective design, the inherent limitations of an observational study, and the use of electronic medical record data.
Lawrence J. Green, MD, clinical professor of dermatology, George Washington University, Washington, who was asked to comment on the study results, said that the findings imply that there is more than statin use alone to protect someone with psoriasis from having an increased risk for MACE. “This is not really surprising because statin use alone is only part of a prevention strategy in someone with psoriasis who usually has multiple comorbidities,” Dr. Green said. “On the other hand, the study only went out for 5 years and cardiovascular disease is a long accumulating process, so it could also be too early to demonstrate MACE prevention.”
The study was funded by a grant from the American Skin Association. Dr. Ormaza Vera and her coauthors reported having no relevant disclosures. Dr. Green disclosed that he is a speaker, consultant, or investigator for many pharmaceutical companies.
A version of this article appeared on Medscape.com .
FROM SID 2024
Dupilumab Evaluated as Treatment for Pediatric Alopecia Areata
showed.
“We might be opening a new avenue for a safe, long-term treatment for our children with AA,” the study’s lead investigator, Emma Guttman-Yassky, MD, PhD, professor and chair of dermatology at the Icahn School of Medicine at Mount Sinai, New York City, said in an interview during the annual meeting of the Society for Investigative Dermatology (SID), where the results were presented during a poster session. “I think AA is likely joining the atopic march, which may allow us to adapt some treatments from the atopy world to AA.”
When the original phase 2 and phase 3 trials of dupilumab for patients with moderate to severe AD were being conducted, Dr. Guttman-Yassky, one of the investigators, recalled observing that some patients who also had patch alopecia experienced hair regrowth. “I was scratching my head because, at the time, AA was considered to be only a Th1-driven disease,” she said. “I asked myself, ‘How can this happen?’ I looked in the literature and found many publications linking atopy in general to alopecia areata. The largest of the dermatologic publications showed that eczema and atopy in general are the highest comorbidities in alopecia areata.”
“This and other findings such as IL [interleukin]-13 genetic linkage with AA and high IgE in patients with AA link AA with Th2 immune skewing, particularly in the setting of atopy,” she continued. In addition, she said, in a large biomarker study involving the scalp and blood of patients with AA, “we found increases in Th2 biomarkers that were associated with alopecia severity.”
Case Series of 20 Pediatric Patients
As part of a case series of children with both AD and AA, Dr. Guttman-Yassky and colleagues evaluated hair regrowth using the Severity of Alopecia Tool (SALT) in 20 pediatric patients (mean age, 10.8 years) who were being treated at Mount Sinai. They collected patient demographics, atopic history, immunoglobulin E (IgE) levels, and SALT scores at follow-up visits every 12-16 weeks for more than 72 weeks and performed Spearman correlations between clinical scores, demographics, and IgE levels.
At baseline, the mean SALT score was 54.4, the mean IgE level was 1567.7 IU/mL, and 75% of patients also had a family history of atopy. The mean follow-up was 67.6 weeks. The researchers observed a significant reduction in SALT scores at week 48 compared with baseline (a mean score of 20.4; P < .01) and continued improvement up to at least 72 weeks (P < .01 vs baseline). They also noted that patients who achieved a treatment response at week 24 had baseline IgE levels > 200 IU/mL.
In other findings, baseline IgE positively correlated with improvement in SALT scores at week 36 (P < .05), while baseline SALT scores positively correlated with disease duration (P < .01) and negatively correlated with improvement in SALT scores at weeks 24, 36, and 48 (P < .005). “The robustness of the response surprised me,” Dr. Guttman-Yassky said in the interview. “Dupilumab for AA takes time to work, but once it kicks in, it kicks in. It takes anywhere from 6 to 12 months to see hair regrowth.”
She acknowledged certain limitations of the analysis, including its small sample size and the fact that it was not a standardized trial. “But, based on our data and the adult data, we are very encouraged about the potential of using dupilumab for children with AA,” she said.
Mount Sinai recently announced that the National Institutes of Health awarded a $6.6 million, 5-year grant to Dr. Guttman-Yassky to further investigate dupilumab as a treatment for children with AA. She will lead a multicenter controlled trial of 76 children with alopecia affecting at least 30% of the scalp, who will be randomized 2:1 (dupilumab:placebo) for 48 weeks, followed by 48 weeks of open-label dupilumab for all participants, with 16 weeks of follow-up, for a total of 112 weeks. Participating sites include Mount Sinai, Yale University, Northwestern University, and the University of California, Irvine.
Dr. Guttman-Yassky disclosed that she is a consultant to many pharmaceutical companies, including dupilumab manufacturers Sanofi and Regeneron.
A version of this article appeared on Medscape.com.
showed.
“We might be opening a new avenue for a safe, long-term treatment for our children with AA,” the study’s lead investigator, Emma Guttman-Yassky, MD, PhD, professor and chair of dermatology at the Icahn School of Medicine at Mount Sinai, New York City, said in an interview during the annual meeting of the Society for Investigative Dermatology (SID), where the results were presented during a poster session. “I think AA is likely joining the atopic march, which may allow us to adapt some treatments from the atopy world to AA.”
When the original phase 2 and phase 3 trials of dupilumab for patients with moderate to severe AD were being conducted, Dr. Guttman-Yassky, one of the investigators, recalled observing that some patients who also had patch alopecia experienced hair regrowth. “I was scratching my head because, at the time, AA was considered to be only a Th1-driven disease,” she said. “I asked myself, ‘How can this happen?’ I looked in the literature and found many publications linking atopy in general to alopecia areata. The largest of the dermatologic publications showed that eczema and atopy in general are the highest comorbidities in alopecia areata.”
“This and other findings such as IL [interleukin]-13 genetic linkage with AA and high IgE in patients with AA link AA with Th2 immune skewing, particularly in the setting of atopy,” she continued. In addition, she said, in a large biomarker study involving the scalp and blood of patients with AA, “we found increases in Th2 biomarkers that were associated with alopecia severity.”
Case Series of 20 Pediatric Patients
As part of a case series of children with both AD and AA, Dr. Guttman-Yassky and colleagues evaluated hair regrowth using the Severity of Alopecia Tool (SALT) in 20 pediatric patients (mean age, 10.8 years) who were being treated at Mount Sinai. They collected patient demographics, atopic history, immunoglobulin E (IgE) levels, and SALT scores at follow-up visits every 12-16 weeks for more than 72 weeks and performed Spearman correlations between clinical scores, demographics, and IgE levels.
At baseline, the mean SALT score was 54.4, the mean IgE level was 1567.7 IU/mL, and 75% of patients also had a family history of atopy. The mean follow-up was 67.6 weeks. The researchers observed a significant reduction in SALT scores at week 48 compared with baseline (a mean score of 20.4; P < .01) and continued improvement up to at least 72 weeks (P < .01 vs baseline). They also noted that patients who achieved a treatment response at week 24 had baseline IgE levels > 200 IU/mL.
In other findings, baseline IgE positively correlated with improvement in SALT scores at week 36 (P < .05), while baseline SALT scores positively correlated with disease duration (P < .01) and negatively correlated with improvement in SALT scores at weeks 24, 36, and 48 (P < .005). “The robustness of the response surprised me,” Dr. Guttman-Yassky said in the interview. “Dupilumab for AA takes time to work, but once it kicks in, it kicks in. It takes anywhere from 6 to 12 months to see hair regrowth.”
She acknowledged certain limitations of the analysis, including its small sample size and the fact that it was not a standardized trial. “But, based on our data and the adult data, we are very encouraged about the potential of using dupilumab for children with AA,” she said.
Mount Sinai recently announced that the National Institutes of Health awarded a $6.6 million, 5-year grant to Dr. Guttman-Yassky to further investigate dupilumab as a treatment for children with AA. She will lead a multicenter controlled trial of 76 children with alopecia affecting at least 30% of the scalp, who will be randomized 2:1 (dupilumab:placebo) for 48 weeks, followed by 48 weeks of open-label dupilumab for all participants, with 16 weeks of follow-up, for a total of 112 weeks. Participating sites include Mount Sinai, Yale University, Northwestern University, and the University of California, Irvine.
Dr. Guttman-Yassky disclosed that she is a consultant to many pharmaceutical companies, including dupilumab manufacturers Sanofi and Regeneron.
A version of this article appeared on Medscape.com.
showed.
“We might be opening a new avenue for a safe, long-term treatment for our children with AA,” the study’s lead investigator, Emma Guttman-Yassky, MD, PhD, professor and chair of dermatology at the Icahn School of Medicine at Mount Sinai, New York City, said in an interview during the annual meeting of the Society for Investigative Dermatology (SID), where the results were presented during a poster session. “I think AA is likely joining the atopic march, which may allow us to adapt some treatments from the atopy world to AA.”
When the original phase 2 and phase 3 trials of dupilumab for patients with moderate to severe AD were being conducted, Dr. Guttman-Yassky, one of the investigators, recalled observing that some patients who also had patch alopecia experienced hair regrowth. “I was scratching my head because, at the time, AA was considered to be only a Th1-driven disease,” she said. “I asked myself, ‘How can this happen?’ I looked in the literature and found many publications linking atopy in general to alopecia areata. The largest of the dermatologic publications showed that eczema and atopy in general are the highest comorbidities in alopecia areata.”
“This and other findings such as IL [interleukin]-13 genetic linkage with AA and high IgE in patients with AA link AA with Th2 immune skewing, particularly in the setting of atopy,” she continued. In addition, she said, in a large biomarker study involving the scalp and blood of patients with AA, “we found increases in Th2 biomarkers that were associated with alopecia severity.”
Case Series of 20 Pediatric Patients
As part of a case series of children with both AD and AA, Dr. Guttman-Yassky and colleagues evaluated hair regrowth using the Severity of Alopecia Tool (SALT) in 20 pediatric patients (mean age, 10.8 years) who were being treated at Mount Sinai. They collected patient demographics, atopic history, immunoglobulin E (IgE) levels, and SALT scores at follow-up visits every 12-16 weeks for more than 72 weeks and performed Spearman correlations between clinical scores, demographics, and IgE levels.
At baseline, the mean SALT score was 54.4, the mean IgE level was 1567.7 IU/mL, and 75% of patients also had a family history of atopy. The mean follow-up was 67.6 weeks. The researchers observed a significant reduction in SALT scores at week 48 compared with baseline (a mean score of 20.4; P < .01) and continued improvement up to at least 72 weeks (P < .01 vs baseline). They also noted that patients who achieved a treatment response at week 24 had baseline IgE levels > 200 IU/mL.
In other findings, baseline IgE positively correlated with improvement in SALT scores at week 36 (P < .05), while baseline SALT scores positively correlated with disease duration (P < .01) and negatively correlated with improvement in SALT scores at weeks 24, 36, and 48 (P < .005). “The robustness of the response surprised me,” Dr. Guttman-Yassky said in the interview. “Dupilumab for AA takes time to work, but once it kicks in, it kicks in. It takes anywhere from 6 to 12 months to see hair regrowth.”
She acknowledged certain limitations of the analysis, including its small sample size and the fact that it was not a standardized trial. “But, based on our data and the adult data, we are very encouraged about the potential of using dupilumab for children with AA,” she said.
Mount Sinai recently announced that the National Institutes of Health awarded a $6.6 million, 5-year grant to Dr. Guttman-Yassky to further investigate dupilumab as a treatment for children with AA. She will lead a multicenter controlled trial of 76 children with alopecia affecting at least 30% of the scalp, who will be randomized 2:1 (dupilumab:placebo) for 48 weeks, followed by 48 weeks of open-label dupilumab for all participants, with 16 weeks of follow-up, for a total of 112 weeks. Participating sites include Mount Sinai, Yale University, Northwestern University, and the University of California, Irvine.
Dr. Guttman-Yassky disclosed that she is a consultant to many pharmaceutical companies, including dupilumab manufacturers Sanofi and Regeneron.
A version of this article appeared on Medscape.com.
FROM SID 2024
Analysis Finds Minority of Chronic Wounds Treated by Dermatologists
. However, fewer than 8% of chronic wounds were managed by dermatologists during this time.
Those are among key findings from an analysis of National Ambulatory Medical Care Survey (NAMCS) data between 2011 and 2019 presented as a late-breaking abstract at the annual meeting of the Society for Investigative Dermatology. “Cutaneous wounds were estimated to account for 28.1 to 96.1 billion dollars in US health care costs in 2014,” one of the study authors, Rithi Chandy, MD, MS, a research fellow at the Center for Dermatology Research at Wake Forest University School of Medicine, Winston-Salem, North Carolina, said in an interview following the meeting. “By examining national trends in patient visits and treatment, we may be able to better inform health care utilization for cutaneous wounds.”
Dr. Chandy and colleagues analyzed de-identified patient data from the 2011 to 2019 NAMCS for acute and chronic wound diagnoses, medications prescribed, and physician specialty categories. During the time studied, 5.76 billion patient visits were made, including 45.1 million visits for cutaneous wounds. Of these, the most common diagnoses were open wounds of the thumb without nail damage (7.96%), the lower leg (5.75%), nonpressure chronic ulcers of other parts of the foot (5.08%), and open wounds of the ear (5%).
Among all visits for cutaneous wounds, about one third were chronic cutaneous wounds, with the following descriptions: “Nonpressure chronic ulcer of other part of foot” (17.8%); “nonpressure chronic ulcer of skin, not elsewhere classified” (9.38%); and “ulcer of lower limbs, excluding decubitus, unspecified” (8.72%). “The frequency of patient visits per year during the study period remained stable for both acute and chronic wounds,” Dr. Chandy said. The number of visits for which antimicrobials were used was stable over time for both acute and chronic cutaneous wounds, with the exception of increased use of antivirals for chronic cutaneous wounds, he added.
Specifically, prescriptions were issued in 156 million visits over the time studied, most commonly cephalexin (4.22%), topical silver sulfadiazine (1.59%), topical mupirocin (1.12%), and miscellaneous antibiotics (1.18%).
“Our data shows that topical mupirocin is the most commonly used topical antimicrobial for cutaneous wounds,” Dr. Chandy said. “However, there are reports of emerging bacterial resistance to mupirocin. Our data can inform ongoing efforts to promote antimicrobial stewardship and drug development to provide alternative options that are less likely to induce antimicrobial resistance.”
In findings limited to specialty-specific NAMCS data available from 2011 and from 2013 to 2016, dermatologists managed 3.85% of overall cutaneous wounds, 2.35% of acute wounds, and 7.39% of chronic wounds. By contrast, Dr. Chandy said, 21.1% of chronic wounds were managed by general/family practice physicians, 20.7% by internists, 6.84% by general surgeons, and 5.65% by orthopedic surgeons.
“As dermatologists are experts in the structure and function of the skin and are trained to manage cutaneous disorders including wound healing, we [believe that] dermatologists are equipped with the skill set” for managing wounds, especially for chronic ulcers, he said. The decline in dermatologists who specialize in wound care, he added, “underscores the need for structured dermatology fellowship programs to prepare next-generation dermatologists to address this shortage and ensure dermatology leadership in cutaneous wound healing.”
Dr. Chandy acknowledged certain limitations of the study, including the potential for misclassification of diagnoses or medications prescribed and the fact that the NAMCS database is unable to provide insight into individual patient experiences such as continual cutaneous wound management for the same patient over time.
In the opinion of Shari R. Lipner, MD, PhD, associate professor of clinical dermatology and director of the Nail Division at Weill Cornell Medicine, New York, who was asked to comment on the study, the most interesting finding was that dermatologists cared for a small minority of patients with cutaneous wounds. “It would be interesting to know whether this is due to dermatologist shortages or knowledge gaps on the part of primary care physicians or patients that dermatologists are trained to care for wounds,” Dr. Lipner told this news organization. Other unanswered questions, she noted, “are patient demographics, geographic locations, and comorbidities.”
One of the study authors, Steven R. Feldman, MD, PhD, professor of dermatology at Wake Forest University, disclosed that he has received research, speaking and/or consulting support from numerous pharmaceutical companies. No other authors reported having relevant disclosures. Dr. Lipner reported having no disclosures.
A version of this article appeared on Medscape.com .
. However, fewer than 8% of chronic wounds were managed by dermatologists during this time.
Those are among key findings from an analysis of National Ambulatory Medical Care Survey (NAMCS) data between 2011 and 2019 presented as a late-breaking abstract at the annual meeting of the Society for Investigative Dermatology. “Cutaneous wounds were estimated to account for 28.1 to 96.1 billion dollars in US health care costs in 2014,” one of the study authors, Rithi Chandy, MD, MS, a research fellow at the Center for Dermatology Research at Wake Forest University School of Medicine, Winston-Salem, North Carolina, said in an interview following the meeting. “By examining national trends in patient visits and treatment, we may be able to better inform health care utilization for cutaneous wounds.”
Dr. Chandy and colleagues analyzed de-identified patient data from the 2011 to 2019 NAMCS for acute and chronic wound diagnoses, medications prescribed, and physician specialty categories. During the time studied, 5.76 billion patient visits were made, including 45.1 million visits for cutaneous wounds. Of these, the most common diagnoses were open wounds of the thumb without nail damage (7.96%), the lower leg (5.75%), nonpressure chronic ulcers of other parts of the foot (5.08%), and open wounds of the ear (5%).
Among all visits for cutaneous wounds, about one third were chronic cutaneous wounds, with the following descriptions: “Nonpressure chronic ulcer of other part of foot” (17.8%); “nonpressure chronic ulcer of skin, not elsewhere classified” (9.38%); and “ulcer of lower limbs, excluding decubitus, unspecified” (8.72%). “The frequency of patient visits per year during the study period remained stable for both acute and chronic wounds,” Dr. Chandy said. The number of visits for which antimicrobials were used was stable over time for both acute and chronic cutaneous wounds, with the exception of increased use of antivirals for chronic cutaneous wounds, he added.
Specifically, prescriptions were issued in 156 million visits over the time studied, most commonly cephalexin (4.22%), topical silver sulfadiazine (1.59%), topical mupirocin (1.12%), and miscellaneous antibiotics (1.18%).
“Our data shows that topical mupirocin is the most commonly used topical antimicrobial for cutaneous wounds,” Dr. Chandy said. “However, there are reports of emerging bacterial resistance to mupirocin. Our data can inform ongoing efforts to promote antimicrobial stewardship and drug development to provide alternative options that are less likely to induce antimicrobial resistance.”
In findings limited to specialty-specific NAMCS data available from 2011 and from 2013 to 2016, dermatologists managed 3.85% of overall cutaneous wounds, 2.35% of acute wounds, and 7.39% of chronic wounds. By contrast, Dr. Chandy said, 21.1% of chronic wounds were managed by general/family practice physicians, 20.7% by internists, 6.84% by general surgeons, and 5.65% by orthopedic surgeons.
“As dermatologists are experts in the structure and function of the skin and are trained to manage cutaneous disorders including wound healing, we [believe that] dermatologists are equipped with the skill set” for managing wounds, especially for chronic ulcers, he said. The decline in dermatologists who specialize in wound care, he added, “underscores the need for structured dermatology fellowship programs to prepare next-generation dermatologists to address this shortage and ensure dermatology leadership in cutaneous wound healing.”
Dr. Chandy acknowledged certain limitations of the study, including the potential for misclassification of diagnoses or medications prescribed and the fact that the NAMCS database is unable to provide insight into individual patient experiences such as continual cutaneous wound management for the same patient over time.
In the opinion of Shari R. Lipner, MD, PhD, associate professor of clinical dermatology and director of the Nail Division at Weill Cornell Medicine, New York, who was asked to comment on the study, the most interesting finding was that dermatologists cared for a small minority of patients with cutaneous wounds. “It would be interesting to know whether this is due to dermatologist shortages or knowledge gaps on the part of primary care physicians or patients that dermatologists are trained to care for wounds,” Dr. Lipner told this news organization. Other unanswered questions, she noted, “are patient demographics, geographic locations, and comorbidities.”
One of the study authors, Steven R. Feldman, MD, PhD, professor of dermatology at Wake Forest University, disclosed that he has received research, speaking and/or consulting support from numerous pharmaceutical companies. No other authors reported having relevant disclosures. Dr. Lipner reported having no disclosures.
A version of this article appeared on Medscape.com .
. However, fewer than 8% of chronic wounds were managed by dermatologists during this time.
Those are among key findings from an analysis of National Ambulatory Medical Care Survey (NAMCS) data between 2011 and 2019 presented as a late-breaking abstract at the annual meeting of the Society for Investigative Dermatology. “Cutaneous wounds were estimated to account for 28.1 to 96.1 billion dollars in US health care costs in 2014,” one of the study authors, Rithi Chandy, MD, MS, a research fellow at the Center for Dermatology Research at Wake Forest University School of Medicine, Winston-Salem, North Carolina, said in an interview following the meeting. “By examining national trends in patient visits and treatment, we may be able to better inform health care utilization for cutaneous wounds.”
Dr. Chandy and colleagues analyzed de-identified patient data from the 2011 to 2019 NAMCS for acute and chronic wound diagnoses, medications prescribed, and physician specialty categories. During the time studied, 5.76 billion patient visits were made, including 45.1 million visits for cutaneous wounds. Of these, the most common diagnoses were open wounds of the thumb without nail damage (7.96%), the lower leg (5.75%), nonpressure chronic ulcers of other parts of the foot (5.08%), and open wounds of the ear (5%).
Among all visits for cutaneous wounds, about one third were chronic cutaneous wounds, with the following descriptions: “Nonpressure chronic ulcer of other part of foot” (17.8%); “nonpressure chronic ulcer of skin, not elsewhere classified” (9.38%); and “ulcer of lower limbs, excluding decubitus, unspecified” (8.72%). “The frequency of patient visits per year during the study period remained stable for both acute and chronic wounds,” Dr. Chandy said. The number of visits for which antimicrobials were used was stable over time for both acute and chronic cutaneous wounds, with the exception of increased use of antivirals for chronic cutaneous wounds, he added.
Specifically, prescriptions were issued in 156 million visits over the time studied, most commonly cephalexin (4.22%), topical silver sulfadiazine (1.59%), topical mupirocin (1.12%), and miscellaneous antibiotics (1.18%).
“Our data shows that topical mupirocin is the most commonly used topical antimicrobial for cutaneous wounds,” Dr. Chandy said. “However, there are reports of emerging bacterial resistance to mupirocin. Our data can inform ongoing efforts to promote antimicrobial stewardship and drug development to provide alternative options that are less likely to induce antimicrobial resistance.”
In findings limited to specialty-specific NAMCS data available from 2011 and from 2013 to 2016, dermatologists managed 3.85% of overall cutaneous wounds, 2.35% of acute wounds, and 7.39% of chronic wounds. By contrast, Dr. Chandy said, 21.1% of chronic wounds were managed by general/family practice physicians, 20.7% by internists, 6.84% by general surgeons, and 5.65% by orthopedic surgeons.
“As dermatologists are experts in the structure and function of the skin and are trained to manage cutaneous disorders including wound healing, we [believe that] dermatologists are equipped with the skill set” for managing wounds, especially for chronic ulcers, he said. The decline in dermatologists who specialize in wound care, he added, “underscores the need for structured dermatology fellowship programs to prepare next-generation dermatologists to address this shortage and ensure dermatology leadership in cutaneous wound healing.”
Dr. Chandy acknowledged certain limitations of the study, including the potential for misclassification of diagnoses or medications prescribed and the fact that the NAMCS database is unable to provide insight into individual patient experiences such as continual cutaneous wound management for the same patient over time.
In the opinion of Shari R. Lipner, MD, PhD, associate professor of clinical dermatology and director of the Nail Division at Weill Cornell Medicine, New York, who was asked to comment on the study, the most interesting finding was that dermatologists cared for a small minority of patients with cutaneous wounds. “It would be interesting to know whether this is due to dermatologist shortages or knowledge gaps on the part of primary care physicians or patients that dermatologists are trained to care for wounds,” Dr. Lipner told this news organization. Other unanswered questions, she noted, “are patient demographics, geographic locations, and comorbidities.”
One of the study authors, Steven R. Feldman, MD, PhD, professor of dermatology at Wake Forest University, disclosed that he has received research, speaking and/or consulting support from numerous pharmaceutical companies. No other authors reported having relevant disclosures. Dr. Lipner reported having no disclosures.
A version of this article appeared on Medscape.com .
FROM SID 2024
Prenatal Antibiotics May Increase Seborrheic Dermatitis Risk in Babies
, but this association was not as strong for childhood-onset SD.
The findings come from a large analysis of data from the United Kingdom that was presented during a late-breaking abstract session at the annual meeting of the Society for Investigative Dermatology.
SD is a common skin disease “that shares similarities with atopic dermatitis or atopic eczema as both are prevalent inflammatory skin diseases that can present with a chronic relapsing, remitting course,” the study’s corresponding author Zelma C. Chiesa Fuxench, MD, MSCE, assistant professor of dermatology at the University of Pennsylvania, Philadelphia, said in an interview. “Like atopic dermatitis, the pathophysiology of seborrheic dermatitis is thought to be complex and involves an interplay between genetics, immune dysregulation, and alterations in lipid composition and the skin microbiome, among others.”
In a previous study, she and colleagues showed that exposure to antibiotics both in utero and during the first 90 days of life increases the risk for atopic dermatitis (AD) in children, with risk being highest with exposure to penicillin even among children whose mothers did not have a history of AD.
For the current study, the researchers drew from a large electronic medical records database in the United Kingdom to perform a prospective cohort analysis of mother-child pairs that used proportional hazards models to examine the association between maternal in utero antibiotic exposure and SD in the child. The population included 1,023,140 children with linked maternal data who were followed for a mean of 10.2 years, which amounts to more than 10-million-person years of data. At baseline, the mean age of mothers was 28 years, 3% had SD, 14% had AD, and 51% of the children were male.
In unadjusted analyses, mothers with SD were more likely to receive an antibiotic during pregnancy than were those who did not have SD (odds ratio [OR], 1.42; 95% CI, 1.39-1.46). In addition, maternal in utero exposure to any antibiotic was associated with an increased risk for infantile SD (OR, 1.70; 95% CI, 1.65-1.76) but less for childhood-onset SD (OR, 1.26; 95% CI, 1.20-1.32). “This effect changed little after adjustment and was still observed if mothers with SD and their babies were excluded,” the authors wrote in their poster abstract.
Any penicillin exposure during pregnancy increased the likelihood of a child having SD (OR, 1.54; 95% CI, 1.50-1.59), with the greater risk for infantile SD (OR, 1.70; 95% CI, 1.65-1.76) than for childhood-onset SD (OR, 1.25; 95% CI, 1.18-1.32). “The trimester of the in utero penicillin exposure did not seem to affect the association with SD,” the authors wrote. The risk was also increased with cephalosporin exposure but was less for sulfonamides and not for childhood-onset SD.
“We observed that antibiotic exposure in utero was primarily associated with an increased risk of infantile SD regardless of the mother’s history of SD, but this association was not as strong for childhood-onset SD,” Dr. Chiesa Fuxench said. “This would suggest that in utero exposure to antibiotics, particularly penicillin, may have its greatest effect on the colonization of skin microbiota in the newborn period leading to the development of infantile SD. Aside from seeking to improve our understanding of the pathophysiology of SD, our findings also suggest that infantile SD and childhood-onset SD may be separate entities with different risk factors, a hypothesis that needs to be further studied.”
She acknowledged certain limitations of the analysis, including the potential for unrecorded diagnoses of SD or misclassified cases in the database. For example, AD and psoriasis “may appear clinically like SD,” she said, although they performed sensitivity analysis excluding patients with these diagnoses and found similar results. In addition, there is the possibility that not all antibiotic exposures were captured in this database, and data on antibiotic exposure may be missing, she added.
Dr. Chiesa Fuxench disclosed that she received research grants from Lilly, LEO Pharma, Regeneron, Sanofi, Tioga, Vanda, and Incyte for work related to AD and from Menlo Therapeutics and Galderma for work related to prurigo nodularis. She has served as a consultant for the Asthma and Allergy Foundation of America, National Eczema Association, AbbVie, Incyte Corporation, and Pfizer and received honoraria for CME work in AD sponsored by education grants from Regeneron/Sanofi and Pfizer and from Beiersdorf for work related to skin cancer and sun protection.
A version of this article appeared on Medscape.com .
, but this association was not as strong for childhood-onset SD.
The findings come from a large analysis of data from the United Kingdom that was presented during a late-breaking abstract session at the annual meeting of the Society for Investigative Dermatology.
SD is a common skin disease “that shares similarities with atopic dermatitis or atopic eczema as both are prevalent inflammatory skin diseases that can present with a chronic relapsing, remitting course,” the study’s corresponding author Zelma C. Chiesa Fuxench, MD, MSCE, assistant professor of dermatology at the University of Pennsylvania, Philadelphia, said in an interview. “Like atopic dermatitis, the pathophysiology of seborrheic dermatitis is thought to be complex and involves an interplay between genetics, immune dysregulation, and alterations in lipid composition and the skin microbiome, among others.”
In a previous study, she and colleagues showed that exposure to antibiotics both in utero and during the first 90 days of life increases the risk for atopic dermatitis (AD) in children, with risk being highest with exposure to penicillin even among children whose mothers did not have a history of AD.
For the current study, the researchers drew from a large electronic medical records database in the United Kingdom to perform a prospective cohort analysis of mother-child pairs that used proportional hazards models to examine the association between maternal in utero antibiotic exposure and SD in the child. The population included 1,023,140 children with linked maternal data who were followed for a mean of 10.2 years, which amounts to more than 10-million-person years of data. At baseline, the mean age of mothers was 28 years, 3% had SD, 14% had AD, and 51% of the children were male.
In unadjusted analyses, mothers with SD were more likely to receive an antibiotic during pregnancy than were those who did not have SD (odds ratio [OR], 1.42; 95% CI, 1.39-1.46). In addition, maternal in utero exposure to any antibiotic was associated with an increased risk for infantile SD (OR, 1.70; 95% CI, 1.65-1.76) but less for childhood-onset SD (OR, 1.26; 95% CI, 1.20-1.32). “This effect changed little after adjustment and was still observed if mothers with SD and their babies were excluded,” the authors wrote in their poster abstract.
Any penicillin exposure during pregnancy increased the likelihood of a child having SD (OR, 1.54; 95% CI, 1.50-1.59), with the greater risk for infantile SD (OR, 1.70; 95% CI, 1.65-1.76) than for childhood-onset SD (OR, 1.25; 95% CI, 1.18-1.32). “The trimester of the in utero penicillin exposure did not seem to affect the association with SD,” the authors wrote. The risk was also increased with cephalosporin exposure but was less for sulfonamides and not for childhood-onset SD.
“We observed that antibiotic exposure in utero was primarily associated with an increased risk of infantile SD regardless of the mother’s history of SD, but this association was not as strong for childhood-onset SD,” Dr. Chiesa Fuxench said. “This would suggest that in utero exposure to antibiotics, particularly penicillin, may have its greatest effect on the colonization of skin microbiota in the newborn period leading to the development of infantile SD. Aside from seeking to improve our understanding of the pathophysiology of SD, our findings also suggest that infantile SD and childhood-onset SD may be separate entities with different risk factors, a hypothesis that needs to be further studied.”
She acknowledged certain limitations of the analysis, including the potential for unrecorded diagnoses of SD or misclassified cases in the database. For example, AD and psoriasis “may appear clinically like SD,” she said, although they performed sensitivity analysis excluding patients with these diagnoses and found similar results. In addition, there is the possibility that not all antibiotic exposures were captured in this database, and data on antibiotic exposure may be missing, she added.
Dr. Chiesa Fuxench disclosed that she received research grants from Lilly, LEO Pharma, Regeneron, Sanofi, Tioga, Vanda, and Incyte for work related to AD and from Menlo Therapeutics and Galderma for work related to prurigo nodularis. She has served as a consultant for the Asthma and Allergy Foundation of America, National Eczema Association, AbbVie, Incyte Corporation, and Pfizer and received honoraria for CME work in AD sponsored by education grants from Regeneron/Sanofi and Pfizer and from Beiersdorf for work related to skin cancer and sun protection.
A version of this article appeared on Medscape.com .
, but this association was not as strong for childhood-onset SD.
The findings come from a large analysis of data from the United Kingdom that was presented during a late-breaking abstract session at the annual meeting of the Society for Investigative Dermatology.
SD is a common skin disease “that shares similarities with atopic dermatitis or atopic eczema as both are prevalent inflammatory skin diseases that can present with a chronic relapsing, remitting course,” the study’s corresponding author Zelma C. Chiesa Fuxench, MD, MSCE, assistant professor of dermatology at the University of Pennsylvania, Philadelphia, said in an interview. “Like atopic dermatitis, the pathophysiology of seborrheic dermatitis is thought to be complex and involves an interplay between genetics, immune dysregulation, and alterations in lipid composition and the skin microbiome, among others.”
In a previous study, she and colleagues showed that exposure to antibiotics both in utero and during the first 90 days of life increases the risk for atopic dermatitis (AD) in children, with risk being highest with exposure to penicillin even among children whose mothers did not have a history of AD.
For the current study, the researchers drew from a large electronic medical records database in the United Kingdom to perform a prospective cohort analysis of mother-child pairs that used proportional hazards models to examine the association between maternal in utero antibiotic exposure and SD in the child. The population included 1,023,140 children with linked maternal data who were followed for a mean of 10.2 years, which amounts to more than 10-million-person years of data. At baseline, the mean age of mothers was 28 years, 3% had SD, 14% had AD, and 51% of the children were male.
In unadjusted analyses, mothers with SD were more likely to receive an antibiotic during pregnancy than were those who did not have SD (odds ratio [OR], 1.42; 95% CI, 1.39-1.46). In addition, maternal in utero exposure to any antibiotic was associated with an increased risk for infantile SD (OR, 1.70; 95% CI, 1.65-1.76) but less for childhood-onset SD (OR, 1.26; 95% CI, 1.20-1.32). “This effect changed little after adjustment and was still observed if mothers with SD and their babies were excluded,” the authors wrote in their poster abstract.
Any penicillin exposure during pregnancy increased the likelihood of a child having SD (OR, 1.54; 95% CI, 1.50-1.59), with the greater risk for infantile SD (OR, 1.70; 95% CI, 1.65-1.76) than for childhood-onset SD (OR, 1.25; 95% CI, 1.18-1.32). “The trimester of the in utero penicillin exposure did not seem to affect the association with SD,” the authors wrote. The risk was also increased with cephalosporin exposure but was less for sulfonamides and not for childhood-onset SD.
“We observed that antibiotic exposure in utero was primarily associated with an increased risk of infantile SD regardless of the mother’s history of SD, but this association was not as strong for childhood-onset SD,” Dr. Chiesa Fuxench said. “This would suggest that in utero exposure to antibiotics, particularly penicillin, may have its greatest effect on the colonization of skin microbiota in the newborn period leading to the development of infantile SD. Aside from seeking to improve our understanding of the pathophysiology of SD, our findings also suggest that infantile SD and childhood-onset SD may be separate entities with different risk factors, a hypothesis that needs to be further studied.”
She acknowledged certain limitations of the analysis, including the potential for unrecorded diagnoses of SD or misclassified cases in the database. For example, AD and psoriasis “may appear clinically like SD,” she said, although they performed sensitivity analysis excluding patients with these diagnoses and found similar results. In addition, there is the possibility that not all antibiotic exposures were captured in this database, and data on antibiotic exposure may be missing, she added.
Dr. Chiesa Fuxench disclosed that she received research grants from Lilly, LEO Pharma, Regeneron, Sanofi, Tioga, Vanda, and Incyte for work related to AD and from Menlo Therapeutics and Galderma for work related to prurigo nodularis. She has served as a consultant for the Asthma and Allergy Foundation of America, National Eczema Association, AbbVie, Incyte Corporation, and Pfizer and received honoraria for CME work in AD sponsored by education grants from Regeneron/Sanofi and Pfizer and from Beiersdorf for work related to skin cancer and sun protection.
A version of this article appeared on Medscape.com .
FROM SID 2024







