User login
Third COVID booster benefits cancer patients
though this population still suffers higher risks than those of the general population, according to a new large-scale observational study out of the United Kingdom.
People living with lymphoma and those who underwent recent systemic anti-cancer treatment or radiotherapy are at the highest risk, according to study author Lennard Y.W. Lee, PhD. “Our study is the largest evaluation of a coronavirus third dose vaccine booster effectiveness in people living with cancer in the world. For the first time we have quantified the benefits of boosters for COVID-19 in cancer patients,” said Dr. Lee, UK COVID Cancer program lead and a medical oncologist at the University of Oxford, England.
The research was published in the November issue of the European Journal of Cancer.
Despite the encouraging numbers, those with cancer continue to have a more than threefold increased risk of both hospitalization and death from coronavirus compared to the general population. “More needs to be done to reduce this excess risk, like prophylactic antibody therapies,” Dr. Lee said.
Third dose efficacy was lower among cancer patients who had been diagnosed within the past 12 months, as well as those with lymphoma, and those who had undergone systemic anti-cancer therapy or radiotherapy within the past 12 months.
The increased vulnerability among individuals with cancer is likely due to compromised immune systems. “Patients with cancer often have impaired B and T cell function and this study provides the largest global clinical study showing the definitive meaningful clinical impact of this,” Dr. Lee said. The greater risk among those with lymphoma likely traces to aberrant white cells or immunosuppressant regimens, he said.
“Vaccination probably should be used in combination with new forms of prevention and in Europe the strategy of using prophylactic antibodies is going to provide additional levels of protection,” Dr. Lee said.
Overall, the study reveals the challenges that cancer patients face in a pandemic that remains a critical health concern, one that can seriously affect quality of life. “Many are still shielding, unable to see family or hug loved ones. Furthermore, looking beyond the direct health risks, there is also the mental health impact. Shielding for nearly 3 years is very difficult. It is important to realize that behind this large-scale study, which is the biggest in the world, there are real people. The pandemic still goes on for them as they remain at higher risk from COVID-19 and we must be aware of the impact on them,” Dr. Lee said.
The study included data from the United Kingdom’s third dose booster vaccine program, representing 361,098 individuals who participated from December 2020 through December 2021. It also include results from all coronavirus tests conducted in the United Kingdom during that period. Among the participants, 97.8% got the Pfizer-BioNTech vaccine as a booster, while 1.5% received the Moderna vaccine. Overall, 8,371,139 individuals received a third dose booster, including 230,666 living with cancer. The researchers used a test-negative case-controlled analysis to estimate vaccine efficacy.
The booster shot had a 59.1% efficacy against breakthrough infections, 62.8% efficacy against symptomatic infections, 80.5% efficacy versus coronavirus hospitalization, and 94.5% efficacy against coronavirus death. Patients with solid tumors benefited from higher efficacy versus breakthrough infections 66.0% versus 53.2%) and symptomatic infections (69.6% versus 56.0%).
Patients with lymphoma experienced just a 10.5% efficacy of the primary dose vaccine versus breakthrough infections and 13.6% versus symptomatic infections, and this did not improve with a third dose. The benefit was greater for hospitalization (23.2%) and death (80.1%).
Despite the additional protection of a third dose, patients with cancer had a higher risk than the population control for coronavirus hospitalization (odds ratio, 3.38; P < .000001) and death (odds ratio, 3.01; P < .000001).
Dr. Lee has no relevant financial disclosures.
though this population still suffers higher risks than those of the general population, according to a new large-scale observational study out of the United Kingdom.
People living with lymphoma and those who underwent recent systemic anti-cancer treatment or radiotherapy are at the highest risk, according to study author Lennard Y.W. Lee, PhD. “Our study is the largest evaluation of a coronavirus third dose vaccine booster effectiveness in people living with cancer in the world. For the first time we have quantified the benefits of boosters for COVID-19 in cancer patients,” said Dr. Lee, UK COVID Cancer program lead and a medical oncologist at the University of Oxford, England.
The research was published in the November issue of the European Journal of Cancer.
Despite the encouraging numbers, those with cancer continue to have a more than threefold increased risk of both hospitalization and death from coronavirus compared to the general population. “More needs to be done to reduce this excess risk, like prophylactic antibody therapies,” Dr. Lee said.
Third dose efficacy was lower among cancer patients who had been diagnosed within the past 12 months, as well as those with lymphoma, and those who had undergone systemic anti-cancer therapy or radiotherapy within the past 12 months.
The increased vulnerability among individuals with cancer is likely due to compromised immune systems. “Patients with cancer often have impaired B and T cell function and this study provides the largest global clinical study showing the definitive meaningful clinical impact of this,” Dr. Lee said. The greater risk among those with lymphoma likely traces to aberrant white cells or immunosuppressant regimens, he said.
“Vaccination probably should be used in combination with new forms of prevention and in Europe the strategy of using prophylactic antibodies is going to provide additional levels of protection,” Dr. Lee said.
Overall, the study reveals the challenges that cancer patients face in a pandemic that remains a critical health concern, one that can seriously affect quality of life. “Many are still shielding, unable to see family or hug loved ones. Furthermore, looking beyond the direct health risks, there is also the mental health impact. Shielding for nearly 3 years is very difficult. It is important to realize that behind this large-scale study, which is the biggest in the world, there are real people. The pandemic still goes on for them as they remain at higher risk from COVID-19 and we must be aware of the impact on them,” Dr. Lee said.
The study included data from the United Kingdom’s third dose booster vaccine program, representing 361,098 individuals who participated from December 2020 through December 2021. It also include results from all coronavirus tests conducted in the United Kingdom during that period. Among the participants, 97.8% got the Pfizer-BioNTech vaccine as a booster, while 1.5% received the Moderna vaccine. Overall, 8,371,139 individuals received a third dose booster, including 230,666 living with cancer. The researchers used a test-negative case-controlled analysis to estimate vaccine efficacy.
The booster shot had a 59.1% efficacy against breakthrough infections, 62.8% efficacy against symptomatic infections, 80.5% efficacy versus coronavirus hospitalization, and 94.5% efficacy against coronavirus death. Patients with solid tumors benefited from higher efficacy versus breakthrough infections 66.0% versus 53.2%) and symptomatic infections (69.6% versus 56.0%).
Patients with lymphoma experienced just a 10.5% efficacy of the primary dose vaccine versus breakthrough infections and 13.6% versus symptomatic infections, and this did not improve with a third dose. The benefit was greater for hospitalization (23.2%) and death (80.1%).
Despite the additional protection of a third dose, patients with cancer had a higher risk than the population control for coronavirus hospitalization (odds ratio, 3.38; P < .000001) and death (odds ratio, 3.01; P < .000001).
Dr. Lee has no relevant financial disclosures.
though this population still suffers higher risks than those of the general population, according to a new large-scale observational study out of the United Kingdom.
People living with lymphoma and those who underwent recent systemic anti-cancer treatment or radiotherapy are at the highest risk, according to study author Lennard Y.W. Lee, PhD. “Our study is the largest evaluation of a coronavirus third dose vaccine booster effectiveness in people living with cancer in the world. For the first time we have quantified the benefits of boosters for COVID-19 in cancer patients,” said Dr. Lee, UK COVID Cancer program lead and a medical oncologist at the University of Oxford, England.
The research was published in the November issue of the European Journal of Cancer.
Despite the encouraging numbers, those with cancer continue to have a more than threefold increased risk of both hospitalization and death from coronavirus compared to the general population. “More needs to be done to reduce this excess risk, like prophylactic antibody therapies,” Dr. Lee said.
Third dose efficacy was lower among cancer patients who had been diagnosed within the past 12 months, as well as those with lymphoma, and those who had undergone systemic anti-cancer therapy or radiotherapy within the past 12 months.
The increased vulnerability among individuals with cancer is likely due to compromised immune systems. “Patients with cancer often have impaired B and T cell function and this study provides the largest global clinical study showing the definitive meaningful clinical impact of this,” Dr. Lee said. The greater risk among those with lymphoma likely traces to aberrant white cells or immunosuppressant regimens, he said.
“Vaccination probably should be used in combination with new forms of prevention and in Europe the strategy of using prophylactic antibodies is going to provide additional levels of protection,” Dr. Lee said.
Overall, the study reveals the challenges that cancer patients face in a pandemic that remains a critical health concern, one that can seriously affect quality of life. “Many are still shielding, unable to see family or hug loved ones. Furthermore, looking beyond the direct health risks, there is also the mental health impact. Shielding for nearly 3 years is very difficult. It is important to realize that behind this large-scale study, which is the biggest in the world, there are real people. The pandemic still goes on for them as they remain at higher risk from COVID-19 and we must be aware of the impact on them,” Dr. Lee said.
The study included data from the United Kingdom’s third dose booster vaccine program, representing 361,098 individuals who participated from December 2020 through December 2021. It also include results from all coronavirus tests conducted in the United Kingdom during that period. Among the participants, 97.8% got the Pfizer-BioNTech vaccine as a booster, while 1.5% received the Moderna vaccine. Overall, 8,371,139 individuals received a third dose booster, including 230,666 living with cancer. The researchers used a test-negative case-controlled analysis to estimate vaccine efficacy.
The booster shot had a 59.1% efficacy against breakthrough infections, 62.8% efficacy against symptomatic infections, 80.5% efficacy versus coronavirus hospitalization, and 94.5% efficacy against coronavirus death. Patients with solid tumors benefited from higher efficacy versus breakthrough infections 66.0% versus 53.2%) and symptomatic infections (69.6% versus 56.0%).
Patients with lymphoma experienced just a 10.5% efficacy of the primary dose vaccine versus breakthrough infections and 13.6% versus symptomatic infections, and this did not improve with a third dose. The benefit was greater for hospitalization (23.2%) and death (80.1%).
Despite the additional protection of a third dose, patients with cancer had a higher risk than the population control for coronavirus hospitalization (odds ratio, 3.38; P < .000001) and death (odds ratio, 3.01; P < .000001).
Dr. Lee has no relevant financial disclosures.
FROM THE EUROPEAN JOURNAL OF CANCER
No benefit of rivaroxaban in COVID outpatients: PREVENT-HD
A new U.S. randomized trial has failed to show benefit of a 35-day course of oral anticoagulation with rivaroxaban for the prevention of thrombotic events in outpatients with symptomatic COVID-19.
The PREVENT-HD trial was presented at the American Heart Association scientific sessions by Gregory Piazza, MD, Brigham and Women’s Hospital, Boston.
“With the caveat that the trial was underpowered to provide a definitive conclusion, these data do not support routine antithrombotic prophylaxis in nonhospitalized patients with symptomatic COVID-19,” Dr. Piazza concluded.
PREVENT-HD is the largest randomized study to look at anticoagulation in nonhospitalized COVID-19 patients and joins a long list of smaller trials that have also shown no benefit with this approach.
However, anticoagulation is recommended in patients who are hospitalized with COVID-19.
Dr. Piazza noted that the issue of anticoagulation in COVID-19 has focused mainly on hospitalized patients, but most COVID-19 cases are treated as outpatients, who are also suspected to be at risk for venous and arterial thrombotic events, especially if they have additional risk factors. Histopathological evidence also suggests that at least part of the deterioration in lung function leading to hospitalization may be attributable to in situ pulmonary artery thrombosis.
The PREVENT-HD trial explored the question of whether early initiation of thromboprophylaxis dosing of rivaroxaban in higher-risk outpatients with COVID-19 may lower the incidence of venous and arterial thrombotic events, reduce in situ pulmonary thrombosis and the worsening of pulmonary function that may lead to hospitalization, and reduce all-cause mortality.
The trial included 1,284 outpatients with a positive test for COVID-19 and who were within 14 days of symptom onset. They also had to have at least one of the following additional risk factors: age over 60 years; prior history of venous thromboembolism (VTE), thrombophilia, coronary artery disease, peripheral artery disease, cardiovascular disease or ischemic stroke, cancer, diabetes, heart failure, obesity (body mass index ≥ 35 kg/m2) or D-dimer > upper limit of normal. Around 35% of the study population had two or more of these risk factors.
Patients were randomized to rivaroxaban 10 mg daily for 35 days or placebo.
The primary efficacy endpoint was time to first occurrence of a composite of symptomatic VTE, myocardial infarction, ischemic stroke, acute limb ischemia, non–central nervous system systemic embolization, all-cause hospitalization, and all-cause mortality up to day 35.
The primary safety endpoint was time to first occurrence of International Society on Thrombosis and Hemostasis (ISTH) critical-site and fatal bleeding.
A modified intention-to-treat analysis (all participants taking at least one dose of study intervention) was also planned.
The trial was stopped early in April this year because of a lower than expected event incidence (3.2%), compared with the planned rate (8.5%), giving a very low likelihood of being able to achieve the required number of events.
Dr. Piazza said reasons contributing to the low event rate included a falling COVID-19 death and hospitalization rate nationwide, and increased use of effective vaccines.
Results of the main intention-to-treat analysis (in 1,284 patients) showed no significant difference in the primary efficacy composite endpoint, which occurred in 3.4% of the rivaroxaban group versus 3.0% of the placebo group.
In the modified intention-to-treat analysis (which included 1,197 patients who actually took at least one dose of the study medication) there was shift in the directionality of the point estimate (rivaroxaban 2.0% vs. placebo 2.7%), which Dr. Piazza said was related to a higher number of patients hospitalized before receiving study drug in the rivaroxaban group. However, the difference was still nonsignificant.
The first major secondary outcome of symptomatic VTE, arterial thrombotic events, and all-cause mortality occurred in 0.3% of rivaroxaban patients versus 1.1% of placebo patients, but this difference did not reach statistical significance.
However, a post hoc exploratory analysis did show a significant reduction in the outcome of symptomatic VTE and arterial thrombotic events.
In terms of safety, there were no fatal critical-site bleeding events, and there was no difference in ISTH major bleeding, which occurred in one patient in the rivaroxaban group versus no patients in the placebo group.
There was, however, a significant increase in nonmajor clinically relevant bleeding with rivaroxaban, which occurred in nine patients (1.5%) versus one patient (0.2%) in the placebo group.
Trivial bleeding was also increased in the rivaroxaban group, occurring in 17 patients (2.8%) versus 5 patients (0.8%) in the placebo group.
Discussant for the study, Renato Lopes, MD, Duke University Medical Center, Durham, N.C., noted that the relationship between COVID-19 and thrombosis has been an important issue since the beginning of the pandemic, with many proposed mechanisms to explain the COVID-19–associated coagulopathy, which is a major cause of death and disability.
While observational data at the beginning of the pandemic suggested patients with COVID-19 might benefit from anticoagulation, looking at all the different randomized trials that have tested anticoagulation in COVID-19 outpatients, there is no treatment effect on the various different primary outcomes in those studies and also no effect on all-cause mortality, Dr. Lopes said.
He pointed out that PREVENT-HD was stopped prematurely with only about one-third of the planned number of patients enrolled, “just like every other outpatient COVID-19 trial.”
He also drew attention to the low rates of vaccination in the trial population, which does not reflect the current vaccination rate in the United States, and said the different direction of the results between the main intention-to-treat and modified intention-to-treat analyses deserve further investigation.
However, Dr. Lopes concluded, “The results of this trial, in line with the body of evidence in this field, do not support the routine use of any antithrombotic therapy for outpatients with COVID-19.”
The PREVENT-HD trial was sponsored by Janssen. Dr. Piazza has reported receiving research support from Bristol-Myers Squibb/Pfizer Alliance, Bayer, Janssen, Alexion, Amgen, and Boston Scientific, and consulting fees from Bristol-Myers Squibb/Pfizer Alliance, Boston Scientific, Janssen, NAMSA, Prairie Education and Research Cooperative, Boston Clinical Research Institute, and Amgen.
A version of this article first appeared on Medscape.com.
A new U.S. randomized trial has failed to show benefit of a 35-day course of oral anticoagulation with rivaroxaban for the prevention of thrombotic events in outpatients with symptomatic COVID-19.
The PREVENT-HD trial was presented at the American Heart Association scientific sessions by Gregory Piazza, MD, Brigham and Women’s Hospital, Boston.
“With the caveat that the trial was underpowered to provide a definitive conclusion, these data do not support routine antithrombotic prophylaxis in nonhospitalized patients with symptomatic COVID-19,” Dr. Piazza concluded.
PREVENT-HD is the largest randomized study to look at anticoagulation in nonhospitalized COVID-19 patients and joins a long list of smaller trials that have also shown no benefit with this approach.
However, anticoagulation is recommended in patients who are hospitalized with COVID-19.
Dr. Piazza noted that the issue of anticoagulation in COVID-19 has focused mainly on hospitalized patients, but most COVID-19 cases are treated as outpatients, who are also suspected to be at risk for venous and arterial thrombotic events, especially if they have additional risk factors. Histopathological evidence also suggests that at least part of the deterioration in lung function leading to hospitalization may be attributable to in situ pulmonary artery thrombosis.
The PREVENT-HD trial explored the question of whether early initiation of thromboprophylaxis dosing of rivaroxaban in higher-risk outpatients with COVID-19 may lower the incidence of venous and arterial thrombotic events, reduce in situ pulmonary thrombosis and the worsening of pulmonary function that may lead to hospitalization, and reduce all-cause mortality.
The trial included 1,284 outpatients with a positive test for COVID-19 and who were within 14 days of symptom onset. They also had to have at least one of the following additional risk factors: age over 60 years; prior history of venous thromboembolism (VTE), thrombophilia, coronary artery disease, peripheral artery disease, cardiovascular disease or ischemic stroke, cancer, diabetes, heart failure, obesity (body mass index ≥ 35 kg/m2) or D-dimer > upper limit of normal. Around 35% of the study population had two or more of these risk factors.
Patients were randomized to rivaroxaban 10 mg daily for 35 days or placebo.
The primary efficacy endpoint was time to first occurrence of a composite of symptomatic VTE, myocardial infarction, ischemic stroke, acute limb ischemia, non–central nervous system systemic embolization, all-cause hospitalization, and all-cause mortality up to day 35.
The primary safety endpoint was time to first occurrence of International Society on Thrombosis and Hemostasis (ISTH) critical-site and fatal bleeding.
A modified intention-to-treat analysis (all participants taking at least one dose of study intervention) was also planned.
The trial was stopped early in April this year because of a lower than expected event incidence (3.2%), compared with the planned rate (8.5%), giving a very low likelihood of being able to achieve the required number of events.
Dr. Piazza said reasons contributing to the low event rate included a falling COVID-19 death and hospitalization rate nationwide, and increased use of effective vaccines.
Results of the main intention-to-treat analysis (in 1,284 patients) showed no significant difference in the primary efficacy composite endpoint, which occurred in 3.4% of the rivaroxaban group versus 3.0% of the placebo group.
In the modified intention-to-treat analysis (which included 1,197 patients who actually took at least one dose of the study medication) there was shift in the directionality of the point estimate (rivaroxaban 2.0% vs. placebo 2.7%), which Dr. Piazza said was related to a higher number of patients hospitalized before receiving study drug in the rivaroxaban group. However, the difference was still nonsignificant.
The first major secondary outcome of symptomatic VTE, arterial thrombotic events, and all-cause mortality occurred in 0.3% of rivaroxaban patients versus 1.1% of placebo patients, but this difference did not reach statistical significance.
However, a post hoc exploratory analysis did show a significant reduction in the outcome of symptomatic VTE and arterial thrombotic events.
In terms of safety, there were no fatal critical-site bleeding events, and there was no difference in ISTH major bleeding, which occurred in one patient in the rivaroxaban group versus no patients in the placebo group.
There was, however, a significant increase in nonmajor clinically relevant bleeding with rivaroxaban, which occurred in nine patients (1.5%) versus one patient (0.2%) in the placebo group.
Trivial bleeding was also increased in the rivaroxaban group, occurring in 17 patients (2.8%) versus 5 patients (0.8%) in the placebo group.
Discussant for the study, Renato Lopes, MD, Duke University Medical Center, Durham, N.C., noted that the relationship between COVID-19 and thrombosis has been an important issue since the beginning of the pandemic, with many proposed mechanisms to explain the COVID-19–associated coagulopathy, which is a major cause of death and disability.
While observational data at the beginning of the pandemic suggested patients with COVID-19 might benefit from anticoagulation, looking at all the different randomized trials that have tested anticoagulation in COVID-19 outpatients, there is no treatment effect on the various different primary outcomes in those studies and also no effect on all-cause mortality, Dr. Lopes said.
He pointed out that PREVENT-HD was stopped prematurely with only about one-third of the planned number of patients enrolled, “just like every other outpatient COVID-19 trial.”
He also drew attention to the low rates of vaccination in the trial population, which does not reflect the current vaccination rate in the United States, and said the different direction of the results between the main intention-to-treat and modified intention-to-treat analyses deserve further investigation.
However, Dr. Lopes concluded, “The results of this trial, in line with the body of evidence in this field, do not support the routine use of any antithrombotic therapy for outpatients with COVID-19.”
The PREVENT-HD trial was sponsored by Janssen. Dr. Piazza has reported receiving research support from Bristol-Myers Squibb/Pfizer Alliance, Bayer, Janssen, Alexion, Amgen, and Boston Scientific, and consulting fees from Bristol-Myers Squibb/Pfizer Alliance, Boston Scientific, Janssen, NAMSA, Prairie Education and Research Cooperative, Boston Clinical Research Institute, and Amgen.
A version of this article first appeared on Medscape.com.
A new U.S. randomized trial has failed to show benefit of a 35-day course of oral anticoagulation with rivaroxaban for the prevention of thrombotic events in outpatients with symptomatic COVID-19.
The PREVENT-HD trial was presented at the American Heart Association scientific sessions by Gregory Piazza, MD, Brigham and Women’s Hospital, Boston.
“With the caveat that the trial was underpowered to provide a definitive conclusion, these data do not support routine antithrombotic prophylaxis in nonhospitalized patients with symptomatic COVID-19,” Dr. Piazza concluded.
PREVENT-HD is the largest randomized study to look at anticoagulation in nonhospitalized COVID-19 patients and joins a long list of smaller trials that have also shown no benefit with this approach.
However, anticoagulation is recommended in patients who are hospitalized with COVID-19.
Dr. Piazza noted that the issue of anticoagulation in COVID-19 has focused mainly on hospitalized patients, but most COVID-19 cases are treated as outpatients, who are also suspected to be at risk for venous and arterial thrombotic events, especially if they have additional risk factors. Histopathological evidence also suggests that at least part of the deterioration in lung function leading to hospitalization may be attributable to in situ pulmonary artery thrombosis.
The PREVENT-HD trial explored the question of whether early initiation of thromboprophylaxis dosing of rivaroxaban in higher-risk outpatients with COVID-19 may lower the incidence of venous and arterial thrombotic events, reduce in situ pulmonary thrombosis and the worsening of pulmonary function that may lead to hospitalization, and reduce all-cause mortality.
The trial included 1,284 outpatients with a positive test for COVID-19 and who were within 14 days of symptom onset. They also had to have at least one of the following additional risk factors: age over 60 years; prior history of venous thromboembolism (VTE), thrombophilia, coronary artery disease, peripheral artery disease, cardiovascular disease or ischemic stroke, cancer, diabetes, heart failure, obesity (body mass index ≥ 35 kg/m2) or D-dimer > upper limit of normal. Around 35% of the study population had two or more of these risk factors.
Patients were randomized to rivaroxaban 10 mg daily for 35 days or placebo.
The primary efficacy endpoint was time to first occurrence of a composite of symptomatic VTE, myocardial infarction, ischemic stroke, acute limb ischemia, non–central nervous system systemic embolization, all-cause hospitalization, and all-cause mortality up to day 35.
The primary safety endpoint was time to first occurrence of International Society on Thrombosis and Hemostasis (ISTH) critical-site and fatal bleeding.
A modified intention-to-treat analysis (all participants taking at least one dose of study intervention) was also planned.
The trial was stopped early in April this year because of a lower than expected event incidence (3.2%), compared with the planned rate (8.5%), giving a very low likelihood of being able to achieve the required number of events.
Dr. Piazza said reasons contributing to the low event rate included a falling COVID-19 death and hospitalization rate nationwide, and increased use of effective vaccines.
Results of the main intention-to-treat analysis (in 1,284 patients) showed no significant difference in the primary efficacy composite endpoint, which occurred in 3.4% of the rivaroxaban group versus 3.0% of the placebo group.
In the modified intention-to-treat analysis (which included 1,197 patients who actually took at least one dose of the study medication) there was shift in the directionality of the point estimate (rivaroxaban 2.0% vs. placebo 2.7%), which Dr. Piazza said was related to a higher number of patients hospitalized before receiving study drug in the rivaroxaban group. However, the difference was still nonsignificant.
The first major secondary outcome of symptomatic VTE, arterial thrombotic events, and all-cause mortality occurred in 0.3% of rivaroxaban patients versus 1.1% of placebo patients, but this difference did not reach statistical significance.
However, a post hoc exploratory analysis did show a significant reduction in the outcome of symptomatic VTE and arterial thrombotic events.
In terms of safety, there were no fatal critical-site bleeding events, and there was no difference in ISTH major bleeding, which occurred in one patient in the rivaroxaban group versus no patients in the placebo group.
There was, however, a significant increase in nonmajor clinically relevant bleeding with rivaroxaban, which occurred in nine patients (1.5%) versus one patient (0.2%) in the placebo group.
Trivial bleeding was also increased in the rivaroxaban group, occurring in 17 patients (2.8%) versus 5 patients (0.8%) in the placebo group.
Discussant for the study, Renato Lopes, MD, Duke University Medical Center, Durham, N.C., noted that the relationship between COVID-19 and thrombosis has been an important issue since the beginning of the pandemic, with many proposed mechanisms to explain the COVID-19–associated coagulopathy, which is a major cause of death and disability.
While observational data at the beginning of the pandemic suggested patients with COVID-19 might benefit from anticoagulation, looking at all the different randomized trials that have tested anticoagulation in COVID-19 outpatients, there is no treatment effect on the various different primary outcomes in those studies and also no effect on all-cause mortality, Dr. Lopes said.
He pointed out that PREVENT-HD was stopped prematurely with only about one-third of the planned number of patients enrolled, “just like every other outpatient COVID-19 trial.”
He also drew attention to the low rates of vaccination in the trial population, which does not reflect the current vaccination rate in the United States, and said the different direction of the results between the main intention-to-treat and modified intention-to-treat analyses deserve further investigation.
However, Dr. Lopes concluded, “The results of this trial, in line with the body of evidence in this field, do not support the routine use of any antithrombotic therapy for outpatients with COVID-19.”
The PREVENT-HD trial was sponsored by Janssen. Dr. Piazza has reported receiving research support from Bristol-Myers Squibb/Pfizer Alliance, Bayer, Janssen, Alexion, Amgen, and Boston Scientific, and consulting fees from Bristol-Myers Squibb/Pfizer Alliance, Boston Scientific, Janssen, NAMSA, Prairie Education and Research Cooperative, Boston Clinical Research Institute, and Amgen.
A version of this article first appeared on Medscape.com.
FROM AHA 2022
More Than a Health Fair: Preventive Health Care During COVID-19 Vaccine Events
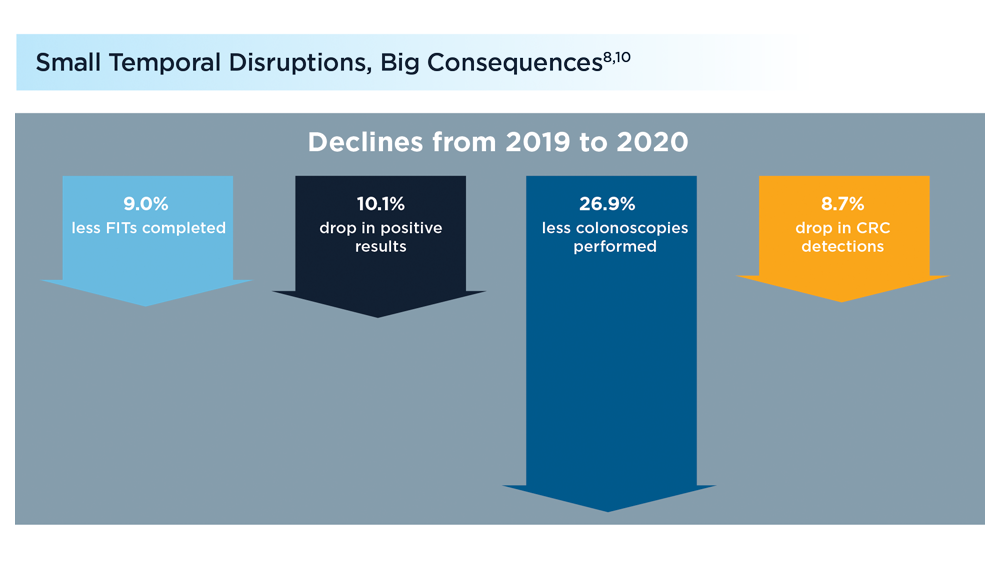
Shortly into the COVID-19 pandemic, Dr. Robert Califf, the commissioner of the US Food and Drug Administration, warned of a coming tsunami of chronic diseases, exacerbated by missed care during the pandemic.1 According to a Centers for Disease Control and Prevention (CDC) survey, more than 30% of adults reported delaying or avoiding routine medical care in the first 6 months of 2020. This rate was highest in people with comorbidities.2 Multiple studies demonstrated declines in hypertension care, hemoglobin A1c testing, mammography, and colon cancer screening.3-5 There has been a resultant increase in colon cancer complications, wounds, and amputations.6,7 The United Kingdom is expected to have a 7.9% to 16.6% increase in future deaths due to breast and colorectal cancer (CRC).8 The World Health Organization estimates an excess 14.9 million people died in 2020 and 2021, either directly from or indirectly related to COVID-19.9
Due to the large-scale conversion from face-to-face care to telehealth modalities, COVID-19 vaccination events offered a unique opportunity to perform preventive health care that requires in-person visits, since most US adults have sought vaccination. However, vaccine events may not reach people most at risk for COVID-19 or chronic disease. Groups of Americans with lower vaccination rates were concerned about driving times and missing work to get the vaccine.10
Distance and travel time may be a particular challenge in Hawaii. Oahu is considered rural by the US Department of Veterans Affairs (VA); some communities are 80 minutes away from the VA Pacific Islands Health Care System (VAPIHCS) main facility. Oahu has approximately 150 veterans experiencing homelessness who may not have transportation to vaccine events. Additionally, VAPIHCS serves veterans that may be at higher risk of not receiving COVID-19 vaccination. Racial and ethnic minority residents have lower vaccination rates, yet are at a higher risk of COVID-19 infection and complications, and through the pandemic, this vaccination gap worsened.11,12 More than 10% of the population of Hawaii is Native Hawaiian or Pacific Islander, and this population is at elevated risk for diabetes mellitus, hypertension, and COVID-19 mortality.13-16
Health Fair Program
The VA provides clinical reminders in its electronic health record (EHR) that are specified by age, gender assigned at birth, and comorbidities. The clinical reminder program is intended to provide clinically relevant reminders for preventive care at the point of care. Veterans with overdue clinical reminders can be identified by name and address, allowing for the creation of health fair events that were directed towards communities with veterans with clinical reminders, including COVID-19 vaccination need. A team of health care professionals from VAPIHCS conceived of a health fair program to increase the reach of vaccine events and include preventive care in partnership with the VAPIHCS Vet Center Program, local communities, U.S.VETS, and the Hawaii Institute of Health Services (HIHS). We sought to determine which services could be offered in community settings; large vaccine events; and at homeless emergency, transitional, or permanent housing. We tracked veterans who received care in the different locations of the directed health fair.
This project was determined to be a quality improvement initiative by the VAPIHCS Office of Research and Development. It was jointly planned by the VAPIHCS pharmacy, infectious diseases, Vet Center Program, and homeless team to make the COVID-19 vaccines available to more rural and to veterans experiencing homelessness, and in response to a decline in facility face-to-face visits. Monthly meetings were held to select sites within zip codes with higher numbers of open clinical reminders and lower vaccination uptake. Informatics developed a list of clinical reminders by zip code for care performed at face-to-face visits.
Partners
The Vet Center Program, suicide prevention coordinator, and the homeless outreach team have a mandate to perform outreach events.17,18 These services collaborate with community partners to locate sites for events. The team was able to leverage these contacts to set up sites for events. The Vet Center Program readjustment counselor and the suicide prevention coordinator provide mental health counseling. The Vet Center counsels on veteran benefits. They supplied a mobile van with WiFi, counseling and examination spaces, and refrigeration, which became the mobile clinic for the preventive care offered at events. The homeless program works with multiple community partners. They contract with HIHS and U.S.VETS to provide emergency and permanent housing for veterans. Each event is reviewed with HIHS and U.S.VETS staff for permission to be on site. The suicide prevention coordinator or the Vet Center readjustment counselor and the homeless team became regular attendees of events. The homeless team provided resources for housing or food insecurity.
Preventive Health Measures
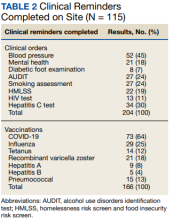
The VA clinical reminder system supports caregivers for both preventive health care and chronic condition management.19 Clinical reminders appear as due in the EHR, and reminder reports can be run by clinical informatics to determine groups of patients who have not had a reminder completed. The following reminders were completed: vaccinations (including COVID-19), CRC screening, diabetic foot check and teaching of foot care, diabetic retinal consultations, laboratory studies (lipids, hemoglobin A1c, microalbumin), mammogram and pap smear referrals, mental health reminders, homeless and food insecurity screening, HIV and hepatitis C testing, and blood pressure (BP) measurement. Health records were reviewed 3 months after each event to determine whether they were completed by the veteran. Additionally, we determined whether BP was controlled (< 130/80 mm Hg).
Settings
Large urban event. The first setting for the health fair was a large vaccination event near the VAPIHCS center in April 2021. Attendance was solicited by VEText, phone calls, and social media advertisements. At check-in, veterans with relevant open clinical reminders were invited to receive preventive health care during the 15-minute monitoring period after the COVID-19 vaccine. The Vet Center Program stationed the mobile van outside the vaccination event, where a physician and a clinical pharmacy specialist (CPS) did assessments, completed reminders, and entered follow-up requests for about 4 hours. A medical support assistant registered veterans who had never signed up for VA health care.
Community Settings. Nine events occurred at least monthly between March and September 2021 at 4 different sites in Oahu. Texts and phone calls were used to solicit attendance; there was no prior publicity on social media. Community events required scheduling resources; this required about 30 hours of medical staff assistant time. Seven sites were visited for about 3 hours each. A physician, pharmacy technician, and CPS conducted assessments, completed reminders, and entered follow-up requests. A medical support assistant registered veterans who had never signed up for VA health care.
Homeless veteran outreach. Five events occurred at 2 homeless veteran housing sites between August 2021 and January 2022. These sites were emergency housing sites (2 events) and transitional and permanent housing (2 events). U.S.VETS and HIHS contacted veterans living in those settings to promote the event. A physician, registered nurse, licensed practical nurse, and CPS conducted assessments, completed reminders, and entered follow-up requests. A medical support assistant registered veterans that had never signed up for VA health care. Each event lasted approximate 3 hours.
Process Quality Improvement
After the CDC changed recommendations to allow concurrent vaccination with the COVID-19 vaccine, we added other vaccinations to the events. This occurred during the course of community events. In June of 2021, there was a health advisory concerning hepatitis A among people experiencing homelessness in Oahu, so hepatitis vaccinations were added for events for veterans.20
Veterans Served
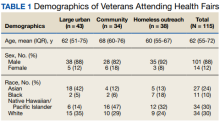
The EHR was used to determine demographics, open clinical reminders, and attendance at follow-up. Simple descriptive statistics were performed in Microsoft Excel. A total of 115 veterans were seen for preventive health visits, and 404 clinical reminders were completed. Seven hundred veterans attended the large centrally located vaccine event and 43 agreed to have a preventive health visit. Thirty-eight veterans had a preventive health visit at homeless outreach events and 34 veterans had a preventive health visit at the community events. Veterans at community
Of the 166 vaccines given, 73 were for COVID-19. Besides vaccination,
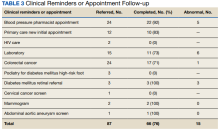
Veteran follow-up or completion
Discussion
This program provided evidence that adding preventive screenings to vaccine events may help reach veterans who may have missed important preventive care due to the COVID-19 pandemic. The involvement of clinical informatics service allowed the outreach to be targeted to communities with incomplete clinical reminders. Interventions that could not be completed at the event had high levels of follow-up by veterans with important findings. The presence of a physician or nurse and a CPS allowed for point-of-care testing, as well as entering orders for medication, laboratory tests, and consultations. The attendance by representatives from the Vet Center, suicide prevention, and homeless services allowed counseling regarding benefits, and mental health follow-up. We believe that we were able to reach communities of veterans with unmet preventive needs and had higher risk of severe COVID-19, given the high numbers with open clinical reminders, the number of vaccines provided, and the high percentage of racial and ethnic minority veterans at events in the community. Our program experience provides some evidence that mobile and pop-up vaccination clinics may be beneficial for screening and managing chronic diseases, as proposed elsewhere.21-24
Strengths of this intervention include that we were able to show a high level of follow-up for recommended medical care as well as the results of our interventions. We have found no similar articles that provide data on completion of follow-up appointments after a health fair. A prior study showed only 23% to 63% of participants at a health fair reported having a recommended follow-up discussion with doctors, but the study reported no outcome of completed cancer screenings.25
Limitations
Weaknesses include the fact that health fair events may reach only healthy people, since attendees generally report better health and better health behaviors than nonattendees.26,27 We felt this was more problematic for the large-scale urban event and that offering rural events and events in homeless housing improved the reach. Future efforts will involve the use of social media and mailings to solicit attendance. To improve follow-up, future work will include adding to the events: phlebotomy or expanded point-of-care testing; specialty care telehealth capability; cervical cancer screen self-collection; and tele-retinal services.
Conclusions
This program provided evidence that directed, preventive screening can be performed in outreach settings paired with vaccine events. These vaccination events in rural and homeless settings reached communities with demonstrable COVID-19 vaccination and other preventive care needs. This approach could be used to help veterans catch up on needed preventive care.
Acknowledgments
Veterans Affairs Pacific Islands Health Care System: Anthony Chance, LCSW; Nicholas Chang, PharmD; Andrew Dahlburg, LCSW; Wilminia G. Ellorimo-Gil, RN; Paul Guillory, RN; Wendy D. Joy; Arthur Minor, LCSW; Avalua Smith; Jessica Spurrier, RN. Veterans Health Administration Vet Center Program: Rolly O. Alvarado; Edmond G. DeGuzman; Richard T. Teel. Hawaii Institute for Human Services. U.S.VETS.
1. Califf RM. Avoiding the coming tsunami of common, chronic disease: What the lessons of the COVID-19 pandemic can teach us. Circulation. 2021;143(19):1831-1834. doi:10.1161/CIRCULATIONAHA.121.053461
2. Czeisler MÉ, Marynak K, Clarke KEN, et al. Delay or avoidance of medical care because of COVID-19-related concerns - United States, June 2020. MMWR Morb Mortal Wkly Rep. 2020;69(36):1250-1257. doi:10.15585/mmwr.mm6936a4
3. European Society of Hypertension Corona-virus Disease 19 Task Force. The corona-virus disease 2019 pandemic compromised routine care for hypertension: a survey conducted among excellence centers of the European Society of Hypertension. J Hypertens. 2021;39(1):190-195. doi:10.1097/HJH.0000000000002703
4. Whaley CM, Pera MF, Cantor J, et al. Changes in health services use among commercially insured US populations during the COVID-19 pandemic. JAMA Netw Open. 2020;3(11):e2024984. doi:10.1001/jamanetworkopen.2020.24984
5. Song H, Bergman A, Chen AT, et al. Disruptions in preventive care: mammograms during the COVID-19 pandemic. Health Serv Res. 2021;56(1):95-101. doi:10.1111/1475-6773.13596
6. Shinkwin M, Silva L, Vogel I, et al. COVID-19 and the emergency presentation of colorectal cancer. Colorectal Dis. 2021;23(8):2014-2019. doi:10.1111/codi.15662
7. Rogers LC, Snyder RJ, Joseph WS. Diabetes-related amputations: a pandemic within a pandemic. J Am Podiatr Med Assoc. 2020;20-248. doi:10.7547/20-248
8. Maringe C, Spicer J, Morris M, et al. The impact of the COVID-19 pandemic on cancer deaths due to delays in diagnosis in England, UK: a national, population-based, modelling study. Lancet Oncol. 2020;21(8):1023-1034. doi:10.1016/S1470-2045(20)30388-0
9. World Health Organization. 14.9 million excess deaths associated with the COVID-19 pandemic in 2020 and 2021. May 5, 2022. Accessed August 31, 2022. https://www.who.int/news/item/05-05-2022-14.9-million-excess-deaths-were-associated-with-the-covid-19-pandemic-in-2020-and-2021
10. Padamsee TJ, Bond RM, Dixon GN, et al. Changes in COVID-19 vaccine hesitancy among Black and White individuals in the US. JAMA Netw Open. 2022;5(1):e2144470. doi:10.1001/jamanetworkopen.2021.44470
11. Barry V, Dasgupta S, Weller DL, et al. Patterns in COVID-19 vaccination coverage, by social vulnerability and urbanicity - United States, December 14, 2020-May 1, 2021. MMWR Morb Mortal Wkly Rep. 2021;70(22):818-824. doi:10.15585/mmwr.mm7022e1
12. Baack BN, Abad N, Yankey D, et al. COVID-19 vaccination coverage and intent among adults aged 18-39 years - United States, March-May 2021. MMWR Morb Mortal Wkly Rep. 2021;70(25):928-933. doi:10.15585/mmwr.mm7025e2
13. United States Census Bureau. QuickFacts Hawaii. July 7, 2021. Accessed August 31, 2022. https://www.census.gov/quickfacts/HI
14. Hawaii Health Data Warehouse. Diabetes - Adult. November 23, 2021. Updated July 31, 2022. Accessed August 31, 2022. https://hhdw.org/report/indicator/summary/DXDiabetesAA.html
15. Hawaii Health Data Warehouse. High Blood Pressure, Adult. November 23, 2021. Accessed August 31, 2022. https://hhdw.org/report/indicator/summary/DXBPHighAA.html
16. Penaia CS, Morey BN, Thomas KB, et al. Disparities in Native Hawaiian and Pacific Islander COVID-19 mortality: a community-driven data response. Am J Public Health. 2021;111(S2):S49-S52. doi:10.2105/AJPH.2021.306370
17. US Department of Veterans Affairs, Veterans Health Administration. VHA Handbook 1500.02 Readjustment Counseling Services (RCS) Vet Center Program. January 26, 2021. Accessed September 7, 2022. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=9168
18. US Department of Veterans Affairs, Veterans Health Administration. VHA Directive 1162.08 Health Care for Veterans Homeless Outreach Services. February 18, 2022. Accessed September 7, 2022. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=9673
19. US Department of Veterans Affairs. Clinical Reminders Version 2.0. Clinician Guide. October 2006. Accessed August 31, 2022. https://www.va.gov/vdl/documents/clinical/cprs-clinical_reminders/pxrm_2_4_um.pdf
20. Hawaii Department of Health. Hepatitis A Cases on Oahu and Maui. June 21, 2021. Accessed August 31, 2022. https://health.hawaii.gov/docd/files/2021/06/Medical-Advisory-HepA-June-21-2021.pdf
21. Hamel L, Lopes L, Sparks G, et al. KFF COVID-19 vaccine monitor: January 2022. January 28, 2022. Accessed August 31, 2022. https://www.kff.org/coronavirus-covid-19/poll-finding/kff-covid-19-vaccine-monitor-january-2022
22. Mast C, Munoz del Rio A. Delayed cancer screenings—a second look. Epic Research Network. July 17, 2020. Accessed August 31, 2022. https://epicresearch.org/articles/delayed-cancer-screenings-a-second-look
23. Shaukat A, Church T. Colorectal cancer screening in the USA in the wake of COVID-19. Lancet Gastroenterol Hepatol. 2020;5(8):726-727. doi:10.1016/S2468-1253(20)30191-6
24. Crespo J, Lazarus JV, Iruzubieta P, García F, García-Samaniego J; Alliance for the elimination of viral hepatitis in Spain. Let’s leverage SARS-CoV2 vaccination to screen for hepatitis C in Spain, in Europe, around the world. J Hepatol. 2021;75(1):224-226. doi:10.1016/j.jhep.2021.03.009
25. Escoffery C, Liang S, Rodgers K, et al. Process evaluation of health fairs promoting cancer screenings. BMC Cancer. 2017;17(1):865. doi:10.1186/s12885-017-3867-3
26. Waller PR, Crow C, Sands D, Becker H. Health related attitudes and health promoting behaviors: differences between health fair attenders and a community group. Am J Health Promot. 1988;3(1):17-32. doi:10.4278/0890-1171-3.1.17
27. Price JH, O’Connell J, Kukulka G. Preventive health behaviors related to the ten leading causes of mortality of health-fair attenders and nonattenders. Psychol Rep. 1985;56(1):131-135. doi:10.2466/pr0.1985.56.1.131
Shortly into the COVID-19 pandemic, Dr. Robert Califf, the commissioner of the US Food and Drug Administration, warned of a coming tsunami of chronic diseases, exacerbated by missed care during the pandemic.1 According to a Centers for Disease Control and Prevention (CDC) survey, more than 30% of adults reported delaying or avoiding routine medical care in the first 6 months of 2020. This rate was highest in people with comorbidities.2 Multiple studies demonstrated declines in hypertension care, hemoglobin A1c testing, mammography, and colon cancer screening.3-5 There has been a resultant increase in colon cancer complications, wounds, and amputations.6,7 The United Kingdom is expected to have a 7.9% to 16.6% increase in future deaths due to breast and colorectal cancer (CRC).8 The World Health Organization estimates an excess 14.9 million people died in 2020 and 2021, either directly from or indirectly related to COVID-19.9
Due to the large-scale conversion from face-to-face care to telehealth modalities, COVID-19 vaccination events offered a unique opportunity to perform preventive health care that requires in-person visits, since most US adults have sought vaccination. However, vaccine events may not reach people most at risk for COVID-19 or chronic disease. Groups of Americans with lower vaccination rates were concerned about driving times and missing work to get the vaccine.10
Distance and travel time may be a particular challenge in Hawaii. Oahu is considered rural by the US Department of Veterans Affairs (VA); some communities are 80 minutes away from the VA Pacific Islands Health Care System (VAPIHCS) main facility. Oahu has approximately 150 veterans experiencing homelessness who may not have transportation to vaccine events. Additionally, VAPIHCS serves veterans that may be at higher risk of not receiving COVID-19 vaccination. Racial and ethnic minority residents have lower vaccination rates, yet are at a higher risk of COVID-19 infection and complications, and through the pandemic, this vaccination gap worsened.11,12 More than 10% of the population of Hawaii is Native Hawaiian or Pacific Islander, and this population is at elevated risk for diabetes mellitus, hypertension, and COVID-19 mortality.13-16
Health Fair Program
The VA provides clinical reminders in its electronic health record (EHR) that are specified by age, gender assigned at birth, and comorbidities. The clinical reminder program is intended to provide clinically relevant reminders for preventive care at the point of care. Veterans with overdue clinical reminders can be identified by name and address, allowing for the creation of health fair events that were directed towards communities with veterans with clinical reminders, including COVID-19 vaccination need. A team of health care professionals from VAPIHCS conceived of a health fair program to increase the reach of vaccine events and include preventive care in partnership with the VAPIHCS Vet Center Program, local communities, U.S.VETS, and the Hawaii Institute of Health Services (HIHS). We sought to determine which services could be offered in community settings; large vaccine events; and at homeless emergency, transitional, or permanent housing. We tracked veterans who received care in the different locations of the directed health fair.
This project was determined to be a quality improvement initiative by the VAPIHCS Office of Research and Development. It was jointly planned by the VAPIHCS pharmacy, infectious diseases, Vet Center Program, and homeless team to make the COVID-19 vaccines available to more rural and to veterans experiencing homelessness, and in response to a decline in facility face-to-face visits. Monthly meetings were held to select sites within zip codes with higher numbers of open clinical reminders and lower vaccination uptake. Informatics developed a list of clinical reminders by zip code for care performed at face-to-face visits.
Partners
The Vet Center Program, suicide prevention coordinator, and the homeless outreach team have a mandate to perform outreach events.17,18 These services collaborate with community partners to locate sites for events. The team was able to leverage these contacts to set up sites for events. The Vet Center Program readjustment counselor and the suicide prevention coordinator provide mental health counseling. The Vet Center counsels on veteran benefits. They supplied a mobile van with WiFi, counseling and examination spaces, and refrigeration, which became the mobile clinic for the preventive care offered at events. The homeless program works with multiple community partners. They contract with HIHS and U.S.VETS to provide emergency and permanent housing for veterans. Each event is reviewed with HIHS and U.S.VETS staff for permission to be on site. The suicide prevention coordinator or the Vet Center readjustment counselor and the homeless team became regular attendees of events. The homeless team provided resources for housing or food insecurity.
Preventive Health Measures
The VA clinical reminder system supports caregivers for both preventive health care and chronic condition management.19 Clinical reminders appear as due in the EHR, and reminder reports can be run by clinical informatics to determine groups of patients who have not had a reminder completed. The following reminders were completed: vaccinations (including COVID-19), CRC screening, diabetic foot check and teaching of foot care, diabetic retinal consultations, laboratory studies (lipids, hemoglobin A1c, microalbumin), mammogram and pap smear referrals, mental health reminders, homeless and food insecurity screening, HIV and hepatitis C testing, and blood pressure (BP) measurement. Health records were reviewed 3 months after each event to determine whether they were completed by the veteran. Additionally, we determined whether BP was controlled (< 130/80 mm Hg).
Settings
Large urban event. The first setting for the health fair was a large vaccination event near the VAPIHCS center in April 2021. Attendance was solicited by VEText, phone calls, and social media advertisements. At check-in, veterans with relevant open clinical reminders were invited to receive preventive health care during the 15-minute monitoring period after the COVID-19 vaccine. The Vet Center Program stationed the mobile van outside the vaccination event, where a physician and a clinical pharmacy specialist (CPS) did assessments, completed reminders, and entered follow-up requests for about 4 hours. A medical support assistant registered veterans who had never signed up for VA health care.
Community Settings. Nine events occurred at least monthly between March and September 2021 at 4 different sites in Oahu. Texts and phone calls were used to solicit attendance; there was no prior publicity on social media. Community events required scheduling resources; this required about 30 hours of medical staff assistant time. Seven sites were visited for about 3 hours each. A physician, pharmacy technician, and CPS conducted assessments, completed reminders, and entered follow-up requests. A medical support assistant registered veterans who had never signed up for VA health care.
Homeless veteran outreach. Five events occurred at 2 homeless veteran housing sites between August 2021 and January 2022. These sites were emergency housing sites (2 events) and transitional and permanent housing (2 events). U.S.VETS and HIHS contacted veterans living in those settings to promote the event. A physician, registered nurse, licensed practical nurse, and CPS conducted assessments, completed reminders, and entered follow-up requests. A medical support assistant registered veterans that had never signed up for VA health care. Each event lasted approximate 3 hours.
Process Quality Improvement
After the CDC changed recommendations to allow concurrent vaccination with the COVID-19 vaccine, we added other vaccinations to the events. This occurred during the course of community events. In June of 2021, there was a health advisory concerning hepatitis A among people experiencing homelessness in Oahu, so hepatitis vaccinations were added for events for veterans.20
Veterans Served
The EHR was used to determine demographics, open clinical reminders, and attendance at follow-up. Simple descriptive statistics were performed in Microsoft Excel. A total of 115 veterans were seen for preventive health visits, and 404 clinical reminders were completed. Seven hundred veterans attended the large centrally located vaccine event and 43 agreed to have a preventive health visit. Thirty-eight veterans had a preventive health visit at homeless outreach events and 34 veterans had a preventive health visit at the community events. Veterans at community
Of the 166 vaccines given, 73 were for COVID-19. Besides vaccination,
Veteran follow-up or completion
Discussion
This program provided evidence that adding preventive screenings to vaccine events may help reach veterans who may have missed important preventive care due to the COVID-19 pandemic. The involvement of clinical informatics service allowed the outreach to be targeted to communities with incomplete clinical reminders. Interventions that could not be completed at the event had high levels of follow-up by veterans with important findings. The presence of a physician or nurse and a CPS allowed for point-of-care testing, as well as entering orders for medication, laboratory tests, and consultations. The attendance by representatives from the Vet Center, suicide prevention, and homeless services allowed counseling regarding benefits, and mental health follow-up. We believe that we were able to reach communities of veterans with unmet preventive needs and had higher risk of severe COVID-19, given the high numbers with open clinical reminders, the number of vaccines provided, and the high percentage of racial and ethnic minority veterans at events in the community. Our program experience provides some evidence that mobile and pop-up vaccination clinics may be beneficial for screening and managing chronic diseases, as proposed elsewhere.21-24
Strengths of this intervention include that we were able to show a high level of follow-up for recommended medical care as well as the results of our interventions. We have found no similar articles that provide data on completion of follow-up appointments after a health fair. A prior study showed only 23% to 63% of participants at a health fair reported having a recommended follow-up discussion with doctors, but the study reported no outcome of completed cancer screenings.25
Limitations
Weaknesses include the fact that health fair events may reach only healthy people, since attendees generally report better health and better health behaviors than nonattendees.26,27 We felt this was more problematic for the large-scale urban event and that offering rural events and events in homeless housing improved the reach. Future efforts will involve the use of social media and mailings to solicit attendance. To improve follow-up, future work will include adding to the events: phlebotomy or expanded point-of-care testing; specialty care telehealth capability; cervical cancer screen self-collection; and tele-retinal services.
Conclusions
This program provided evidence that directed, preventive screening can be performed in outreach settings paired with vaccine events. These vaccination events in rural and homeless settings reached communities with demonstrable COVID-19 vaccination and other preventive care needs. This approach could be used to help veterans catch up on needed preventive care.
Acknowledgments
Veterans Affairs Pacific Islands Health Care System: Anthony Chance, LCSW; Nicholas Chang, PharmD; Andrew Dahlburg, LCSW; Wilminia G. Ellorimo-Gil, RN; Paul Guillory, RN; Wendy D. Joy; Arthur Minor, LCSW; Avalua Smith; Jessica Spurrier, RN. Veterans Health Administration Vet Center Program: Rolly O. Alvarado; Edmond G. DeGuzman; Richard T. Teel. Hawaii Institute for Human Services. U.S.VETS.
Shortly into the COVID-19 pandemic, Dr. Robert Califf, the commissioner of the US Food and Drug Administration, warned of a coming tsunami of chronic diseases, exacerbated by missed care during the pandemic.1 According to a Centers for Disease Control and Prevention (CDC) survey, more than 30% of adults reported delaying or avoiding routine medical care in the first 6 months of 2020. This rate was highest in people with comorbidities.2 Multiple studies demonstrated declines in hypertension care, hemoglobin A1c testing, mammography, and colon cancer screening.3-5 There has been a resultant increase in colon cancer complications, wounds, and amputations.6,7 The United Kingdom is expected to have a 7.9% to 16.6% increase in future deaths due to breast and colorectal cancer (CRC).8 The World Health Organization estimates an excess 14.9 million people died in 2020 and 2021, either directly from or indirectly related to COVID-19.9
Due to the large-scale conversion from face-to-face care to telehealth modalities, COVID-19 vaccination events offered a unique opportunity to perform preventive health care that requires in-person visits, since most US adults have sought vaccination. However, vaccine events may not reach people most at risk for COVID-19 or chronic disease. Groups of Americans with lower vaccination rates were concerned about driving times and missing work to get the vaccine.10
Distance and travel time may be a particular challenge in Hawaii. Oahu is considered rural by the US Department of Veterans Affairs (VA); some communities are 80 minutes away from the VA Pacific Islands Health Care System (VAPIHCS) main facility. Oahu has approximately 150 veterans experiencing homelessness who may not have transportation to vaccine events. Additionally, VAPIHCS serves veterans that may be at higher risk of not receiving COVID-19 vaccination. Racial and ethnic minority residents have lower vaccination rates, yet are at a higher risk of COVID-19 infection and complications, and through the pandemic, this vaccination gap worsened.11,12 More than 10% of the population of Hawaii is Native Hawaiian or Pacific Islander, and this population is at elevated risk for diabetes mellitus, hypertension, and COVID-19 mortality.13-16
Health Fair Program
The VA provides clinical reminders in its electronic health record (EHR) that are specified by age, gender assigned at birth, and comorbidities. The clinical reminder program is intended to provide clinically relevant reminders for preventive care at the point of care. Veterans with overdue clinical reminders can be identified by name and address, allowing for the creation of health fair events that were directed towards communities with veterans with clinical reminders, including COVID-19 vaccination need. A team of health care professionals from VAPIHCS conceived of a health fair program to increase the reach of vaccine events and include preventive care in partnership with the VAPIHCS Vet Center Program, local communities, U.S.VETS, and the Hawaii Institute of Health Services (HIHS). We sought to determine which services could be offered in community settings; large vaccine events; and at homeless emergency, transitional, or permanent housing. We tracked veterans who received care in the different locations of the directed health fair.
This project was determined to be a quality improvement initiative by the VAPIHCS Office of Research and Development. It was jointly planned by the VAPIHCS pharmacy, infectious diseases, Vet Center Program, and homeless team to make the COVID-19 vaccines available to more rural and to veterans experiencing homelessness, and in response to a decline in facility face-to-face visits. Monthly meetings were held to select sites within zip codes with higher numbers of open clinical reminders and lower vaccination uptake. Informatics developed a list of clinical reminders by zip code for care performed at face-to-face visits.
Partners
The Vet Center Program, suicide prevention coordinator, and the homeless outreach team have a mandate to perform outreach events.17,18 These services collaborate with community partners to locate sites for events. The team was able to leverage these contacts to set up sites for events. The Vet Center Program readjustment counselor and the suicide prevention coordinator provide mental health counseling. The Vet Center counsels on veteran benefits. They supplied a mobile van with WiFi, counseling and examination spaces, and refrigeration, which became the mobile clinic for the preventive care offered at events. The homeless program works with multiple community partners. They contract with HIHS and U.S.VETS to provide emergency and permanent housing for veterans. Each event is reviewed with HIHS and U.S.VETS staff for permission to be on site. The suicide prevention coordinator or the Vet Center readjustment counselor and the homeless team became regular attendees of events. The homeless team provided resources for housing or food insecurity.
Preventive Health Measures
The VA clinical reminder system supports caregivers for both preventive health care and chronic condition management.19 Clinical reminders appear as due in the EHR, and reminder reports can be run by clinical informatics to determine groups of patients who have not had a reminder completed. The following reminders were completed: vaccinations (including COVID-19), CRC screening, diabetic foot check and teaching of foot care, diabetic retinal consultations, laboratory studies (lipids, hemoglobin A1c, microalbumin), mammogram and pap smear referrals, mental health reminders, homeless and food insecurity screening, HIV and hepatitis C testing, and blood pressure (BP) measurement. Health records were reviewed 3 months after each event to determine whether they were completed by the veteran. Additionally, we determined whether BP was controlled (< 130/80 mm Hg).
Settings
Large urban event. The first setting for the health fair was a large vaccination event near the VAPIHCS center in April 2021. Attendance was solicited by VEText, phone calls, and social media advertisements. At check-in, veterans with relevant open clinical reminders were invited to receive preventive health care during the 15-minute monitoring period after the COVID-19 vaccine. The Vet Center Program stationed the mobile van outside the vaccination event, where a physician and a clinical pharmacy specialist (CPS) did assessments, completed reminders, and entered follow-up requests for about 4 hours. A medical support assistant registered veterans who had never signed up for VA health care.
Community Settings. Nine events occurred at least monthly between March and September 2021 at 4 different sites in Oahu. Texts and phone calls were used to solicit attendance; there was no prior publicity on social media. Community events required scheduling resources; this required about 30 hours of medical staff assistant time. Seven sites were visited for about 3 hours each. A physician, pharmacy technician, and CPS conducted assessments, completed reminders, and entered follow-up requests. A medical support assistant registered veterans who had never signed up for VA health care.
Homeless veteran outreach. Five events occurred at 2 homeless veteran housing sites between August 2021 and January 2022. These sites were emergency housing sites (2 events) and transitional and permanent housing (2 events). U.S.VETS and HIHS contacted veterans living in those settings to promote the event. A physician, registered nurse, licensed practical nurse, and CPS conducted assessments, completed reminders, and entered follow-up requests. A medical support assistant registered veterans that had never signed up for VA health care. Each event lasted approximate 3 hours.
Process Quality Improvement
After the CDC changed recommendations to allow concurrent vaccination with the COVID-19 vaccine, we added other vaccinations to the events. This occurred during the course of community events. In June of 2021, there was a health advisory concerning hepatitis A among people experiencing homelessness in Oahu, so hepatitis vaccinations were added for events for veterans.20
Veterans Served
The EHR was used to determine demographics, open clinical reminders, and attendance at follow-up. Simple descriptive statistics were performed in Microsoft Excel. A total of 115 veterans were seen for preventive health visits, and 404 clinical reminders were completed. Seven hundred veterans attended the large centrally located vaccine event and 43 agreed to have a preventive health visit. Thirty-eight veterans had a preventive health visit at homeless outreach events and 34 veterans had a preventive health visit at the community events. Veterans at community
Of the 166 vaccines given, 73 were for COVID-19. Besides vaccination,
Veteran follow-up or completion
Discussion
This program provided evidence that adding preventive screenings to vaccine events may help reach veterans who may have missed important preventive care due to the COVID-19 pandemic. The involvement of clinical informatics service allowed the outreach to be targeted to communities with incomplete clinical reminders. Interventions that could not be completed at the event had high levels of follow-up by veterans with important findings. The presence of a physician or nurse and a CPS allowed for point-of-care testing, as well as entering orders for medication, laboratory tests, and consultations. The attendance by representatives from the Vet Center, suicide prevention, and homeless services allowed counseling regarding benefits, and mental health follow-up. We believe that we were able to reach communities of veterans with unmet preventive needs and had higher risk of severe COVID-19, given the high numbers with open clinical reminders, the number of vaccines provided, and the high percentage of racial and ethnic minority veterans at events in the community. Our program experience provides some evidence that mobile and pop-up vaccination clinics may be beneficial for screening and managing chronic diseases, as proposed elsewhere.21-24
Strengths of this intervention include that we were able to show a high level of follow-up for recommended medical care as well as the results of our interventions. We have found no similar articles that provide data on completion of follow-up appointments after a health fair. A prior study showed only 23% to 63% of participants at a health fair reported having a recommended follow-up discussion with doctors, but the study reported no outcome of completed cancer screenings.25
Limitations
Weaknesses include the fact that health fair events may reach only healthy people, since attendees generally report better health and better health behaviors than nonattendees.26,27 We felt this was more problematic for the large-scale urban event and that offering rural events and events in homeless housing improved the reach. Future efforts will involve the use of social media and mailings to solicit attendance. To improve follow-up, future work will include adding to the events: phlebotomy or expanded point-of-care testing; specialty care telehealth capability; cervical cancer screen self-collection; and tele-retinal services.
Conclusions
This program provided evidence that directed, preventive screening can be performed in outreach settings paired with vaccine events. These vaccination events in rural and homeless settings reached communities with demonstrable COVID-19 vaccination and other preventive care needs. This approach could be used to help veterans catch up on needed preventive care.
Acknowledgments
Veterans Affairs Pacific Islands Health Care System: Anthony Chance, LCSW; Nicholas Chang, PharmD; Andrew Dahlburg, LCSW; Wilminia G. Ellorimo-Gil, RN; Paul Guillory, RN; Wendy D. Joy; Arthur Minor, LCSW; Avalua Smith; Jessica Spurrier, RN. Veterans Health Administration Vet Center Program: Rolly O. Alvarado; Edmond G. DeGuzman; Richard T. Teel. Hawaii Institute for Human Services. U.S.VETS.
1. Califf RM. Avoiding the coming tsunami of common, chronic disease: What the lessons of the COVID-19 pandemic can teach us. Circulation. 2021;143(19):1831-1834. doi:10.1161/CIRCULATIONAHA.121.053461
2. Czeisler MÉ, Marynak K, Clarke KEN, et al. Delay or avoidance of medical care because of COVID-19-related concerns - United States, June 2020. MMWR Morb Mortal Wkly Rep. 2020;69(36):1250-1257. doi:10.15585/mmwr.mm6936a4
3. European Society of Hypertension Corona-virus Disease 19 Task Force. The corona-virus disease 2019 pandemic compromised routine care for hypertension: a survey conducted among excellence centers of the European Society of Hypertension. J Hypertens. 2021;39(1):190-195. doi:10.1097/HJH.0000000000002703
4. Whaley CM, Pera MF, Cantor J, et al. Changes in health services use among commercially insured US populations during the COVID-19 pandemic. JAMA Netw Open. 2020;3(11):e2024984. doi:10.1001/jamanetworkopen.2020.24984
5. Song H, Bergman A, Chen AT, et al. Disruptions in preventive care: mammograms during the COVID-19 pandemic. Health Serv Res. 2021;56(1):95-101. doi:10.1111/1475-6773.13596
6. Shinkwin M, Silva L, Vogel I, et al. COVID-19 and the emergency presentation of colorectal cancer. Colorectal Dis. 2021;23(8):2014-2019. doi:10.1111/codi.15662
7. Rogers LC, Snyder RJ, Joseph WS. Diabetes-related amputations: a pandemic within a pandemic. J Am Podiatr Med Assoc. 2020;20-248. doi:10.7547/20-248
8. Maringe C, Spicer J, Morris M, et al. The impact of the COVID-19 pandemic on cancer deaths due to delays in diagnosis in England, UK: a national, population-based, modelling study. Lancet Oncol. 2020;21(8):1023-1034. doi:10.1016/S1470-2045(20)30388-0
9. World Health Organization. 14.9 million excess deaths associated with the COVID-19 pandemic in 2020 and 2021. May 5, 2022. Accessed August 31, 2022. https://www.who.int/news/item/05-05-2022-14.9-million-excess-deaths-were-associated-with-the-covid-19-pandemic-in-2020-and-2021
10. Padamsee TJ, Bond RM, Dixon GN, et al. Changes in COVID-19 vaccine hesitancy among Black and White individuals in the US. JAMA Netw Open. 2022;5(1):e2144470. doi:10.1001/jamanetworkopen.2021.44470
11. Barry V, Dasgupta S, Weller DL, et al. Patterns in COVID-19 vaccination coverage, by social vulnerability and urbanicity - United States, December 14, 2020-May 1, 2021. MMWR Morb Mortal Wkly Rep. 2021;70(22):818-824. doi:10.15585/mmwr.mm7022e1
12. Baack BN, Abad N, Yankey D, et al. COVID-19 vaccination coverage and intent among adults aged 18-39 years - United States, March-May 2021. MMWR Morb Mortal Wkly Rep. 2021;70(25):928-933. doi:10.15585/mmwr.mm7025e2
13. United States Census Bureau. QuickFacts Hawaii. July 7, 2021. Accessed August 31, 2022. https://www.census.gov/quickfacts/HI
14. Hawaii Health Data Warehouse. Diabetes - Adult. November 23, 2021. Updated July 31, 2022. Accessed August 31, 2022. https://hhdw.org/report/indicator/summary/DXDiabetesAA.html
15. Hawaii Health Data Warehouse. High Blood Pressure, Adult. November 23, 2021. Accessed August 31, 2022. https://hhdw.org/report/indicator/summary/DXBPHighAA.html
16. Penaia CS, Morey BN, Thomas KB, et al. Disparities in Native Hawaiian and Pacific Islander COVID-19 mortality: a community-driven data response. Am J Public Health. 2021;111(S2):S49-S52. doi:10.2105/AJPH.2021.306370
17. US Department of Veterans Affairs, Veterans Health Administration. VHA Handbook 1500.02 Readjustment Counseling Services (RCS) Vet Center Program. January 26, 2021. Accessed September 7, 2022. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=9168
18. US Department of Veterans Affairs, Veterans Health Administration. VHA Directive 1162.08 Health Care for Veterans Homeless Outreach Services. February 18, 2022. Accessed September 7, 2022. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=9673
19. US Department of Veterans Affairs. Clinical Reminders Version 2.0. Clinician Guide. October 2006. Accessed August 31, 2022. https://www.va.gov/vdl/documents/clinical/cprs-clinical_reminders/pxrm_2_4_um.pdf
20. Hawaii Department of Health. Hepatitis A Cases on Oahu and Maui. June 21, 2021. Accessed August 31, 2022. https://health.hawaii.gov/docd/files/2021/06/Medical-Advisory-HepA-June-21-2021.pdf
21. Hamel L, Lopes L, Sparks G, et al. KFF COVID-19 vaccine monitor: January 2022. January 28, 2022. Accessed August 31, 2022. https://www.kff.org/coronavirus-covid-19/poll-finding/kff-covid-19-vaccine-monitor-january-2022
22. Mast C, Munoz del Rio A. Delayed cancer screenings—a second look. Epic Research Network. July 17, 2020. Accessed August 31, 2022. https://epicresearch.org/articles/delayed-cancer-screenings-a-second-look
23. Shaukat A, Church T. Colorectal cancer screening in the USA in the wake of COVID-19. Lancet Gastroenterol Hepatol. 2020;5(8):726-727. doi:10.1016/S2468-1253(20)30191-6
24. Crespo J, Lazarus JV, Iruzubieta P, García F, García-Samaniego J; Alliance for the elimination of viral hepatitis in Spain. Let’s leverage SARS-CoV2 vaccination to screen for hepatitis C in Spain, in Europe, around the world. J Hepatol. 2021;75(1):224-226. doi:10.1016/j.jhep.2021.03.009
25. Escoffery C, Liang S, Rodgers K, et al. Process evaluation of health fairs promoting cancer screenings. BMC Cancer. 2017;17(1):865. doi:10.1186/s12885-017-3867-3
26. Waller PR, Crow C, Sands D, Becker H. Health related attitudes and health promoting behaviors: differences between health fair attenders and a community group. Am J Health Promot. 1988;3(1):17-32. doi:10.4278/0890-1171-3.1.17
27. Price JH, O’Connell J, Kukulka G. Preventive health behaviors related to the ten leading causes of mortality of health-fair attenders and nonattenders. Psychol Rep. 1985;56(1):131-135. doi:10.2466/pr0.1985.56.1.131
1. Califf RM. Avoiding the coming tsunami of common, chronic disease: What the lessons of the COVID-19 pandemic can teach us. Circulation. 2021;143(19):1831-1834. doi:10.1161/CIRCULATIONAHA.121.053461
2. Czeisler MÉ, Marynak K, Clarke KEN, et al. Delay or avoidance of medical care because of COVID-19-related concerns - United States, June 2020. MMWR Morb Mortal Wkly Rep. 2020;69(36):1250-1257. doi:10.15585/mmwr.mm6936a4
3. European Society of Hypertension Corona-virus Disease 19 Task Force. The corona-virus disease 2019 pandemic compromised routine care for hypertension: a survey conducted among excellence centers of the European Society of Hypertension. J Hypertens. 2021;39(1):190-195. doi:10.1097/HJH.0000000000002703
4. Whaley CM, Pera MF, Cantor J, et al. Changes in health services use among commercially insured US populations during the COVID-19 pandemic. JAMA Netw Open. 2020;3(11):e2024984. doi:10.1001/jamanetworkopen.2020.24984
5. Song H, Bergman A, Chen AT, et al. Disruptions in preventive care: mammograms during the COVID-19 pandemic. Health Serv Res. 2021;56(1):95-101. doi:10.1111/1475-6773.13596
6. Shinkwin M, Silva L, Vogel I, et al. COVID-19 and the emergency presentation of colorectal cancer. Colorectal Dis. 2021;23(8):2014-2019. doi:10.1111/codi.15662
7. Rogers LC, Snyder RJ, Joseph WS. Diabetes-related amputations: a pandemic within a pandemic. J Am Podiatr Med Assoc. 2020;20-248. doi:10.7547/20-248
8. Maringe C, Spicer J, Morris M, et al. The impact of the COVID-19 pandemic on cancer deaths due to delays in diagnosis in England, UK: a national, population-based, modelling study. Lancet Oncol. 2020;21(8):1023-1034. doi:10.1016/S1470-2045(20)30388-0
9. World Health Organization. 14.9 million excess deaths associated with the COVID-19 pandemic in 2020 and 2021. May 5, 2022. Accessed August 31, 2022. https://www.who.int/news/item/05-05-2022-14.9-million-excess-deaths-were-associated-with-the-covid-19-pandemic-in-2020-and-2021
10. Padamsee TJ, Bond RM, Dixon GN, et al. Changes in COVID-19 vaccine hesitancy among Black and White individuals in the US. JAMA Netw Open. 2022;5(1):e2144470. doi:10.1001/jamanetworkopen.2021.44470
11. Barry V, Dasgupta S, Weller DL, et al. Patterns in COVID-19 vaccination coverage, by social vulnerability and urbanicity - United States, December 14, 2020-May 1, 2021. MMWR Morb Mortal Wkly Rep. 2021;70(22):818-824. doi:10.15585/mmwr.mm7022e1
12. Baack BN, Abad N, Yankey D, et al. COVID-19 vaccination coverage and intent among adults aged 18-39 years - United States, March-May 2021. MMWR Morb Mortal Wkly Rep. 2021;70(25):928-933. doi:10.15585/mmwr.mm7025e2
13. United States Census Bureau. QuickFacts Hawaii. July 7, 2021. Accessed August 31, 2022. https://www.census.gov/quickfacts/HI
14. Hawaii Health Data Warehouse. Diabetes - Adult. November 23, 2021. Updated July 31, 2022. Accessed August 31, 2022. https://hhdw.org/report/indicator/summary/DXDiabetesAA.html
15. Hawaii Health Data Warehouse. High Blood Pressure, Adult. November 23, 2021. Accessed August 31, 2022. https://hhdw.org/report/indicator/summary/DXBPHighAA.html
16. Penaia CS, Morey BN, Thomas KB, et al. Disparities in Native Hawaiian and Pacific Islander COVID-19 mortality: a community-driven data response. Am J Public Health. 2021;111(S2):S49-S52. doi:10.2105/AJPH.2021.306370
17. US Department of Veterans Affairs, Veterans Health Administration. VHA Handbook 1500.02 Readjustment Counseling Services (RCS) Vet Center Program. January 26, 2021. Accessed September 7, 2022. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=9168
18. US Department of Veterans Affairs, Veterans Health Administration. VHA Directive 1162.08 Health Care for Veterans Homeless Outreach Services. February 18, 2022. Accessed September 7, 2022. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=9673
19. US Department of Veterans Affairs. Clinical Reminders Version 2.0. Clinician Guide. October 2006. Accessed August 31, 2022. https://www.va.gov/vdl/documents/clinical/cprs-clinical_reminders/pxrm_2_4_um.pdf
20. Hawaii Department of Health. Hepatitis A Cases on Oahu and Maui. June 21, 2021. Accessed August 31, 2022. https://health.hawaii.gov/docd/files/2021/06/Medical-Advisory-HepA-June-21-2021.pdf
21. Hamel L, Lopes L, Sparks G, et al. KFF COVID-19 vaccine monitor: January 2022. January 28, 2022. Accessed August 31, 2022. https://www.kff.org/coronavirus-covid-19/poll-finding/kff-covid-19-vaccine-monitor-january-2022
22. Mast C, Munoz del Rio A. Delayed cancer screenings—a second look. Epic Research Network. July 17, 2020. Accessed August 31, 2022. https://epicresearch.org/articles/delayed-cancer-screenings-a-second-look
23. Shaukat A, Church T. Colorectal cancer screening in the USA in the wake of COVID-19. Lancet Gastroenterol Hepatol. 2020;5(8):726-727. doi:10.1016/S2468-1253(20)30191-6
24. Crespo J, Lazarus JV, Iruzubieta P, García F, García-Samaniego J; Alliance for the elimination of viral hepatitis in Spain. Let’s leverage SARS-CoV2 vaccination to screen for hepatitis C in Spain, in Europe, around the world. J Hepatol. 2021;75(1):224-226. doi:10.1016/j.jhep.2021.03.009
25. Escoffery C, Liang S, Rodgers K, et al. Process evaluation of health fairs promoting cancer screenings. BMC Cancer. 2017;17(1):865. doi:10.1186/s12885-017-3867-3
26. Waller PR, Crow C, Sands D, Becker H. Health related attitudes and health promoting behaviors: differences between health fair attenders and a community group. Am J Health Promot. 1988;3(1):17-32. doi:10.4278/0890-1171-3.1.17
27. Price JH, O’Connell J, Kukulka G. Preventive health behaviors related to the ten leading causes of mortality of health-fair attenders and nonattenders. Psychol Rep. 1985;56(1):131-135. doi:10.2466/pr0.1985.56.1.131
Liver disease-related deaths rise during pandemic
according to new findings presented at the annual meeting of the American Association for the Study of Liver Diseases.
Between 2019 and 2021, ALD-related deaths increased by 17.6% and NAFLD-related deaths increased by 14.5%, Yee Hui Yeo, MD, a resident physician and hepatology-focused investigator at Cedars-Sinai Medical Center in Los Angeles, said at a preconference press briefing.
“Even before the pandemic, the mortality rates for these two diseases have been increasing, with NAFLD having an even steeper increasing trend,” he said. “During the pandemic, these two diseases had a significant surge.”
Recent U.S. liver disease death rates
Dr. Yeo and colleagues analyzed data from the Center for Disease Control and Prevention’s National Vital Statistic System to estimate the age-standardized mortality rates (ASMR) of liver disease between 2010 and 2021, including ALD, NAFLD, hepatitis B, and hepatitis C. Using prediction modeling analyses based on trends from 2010 to 2019, they predicted mortality rates for 2020-2021 and compared them with the observed rates to quantify the differences related to the pandemic.
Between 2010 and 2021, there were about 626,000 chronic liver disease–related deaths, including about 343,000 ALD-related deaths, 204,000 hepatitis C–related deaths, 58,000 NAFLD-related deaths, and 21,000 hepatitis B–related deaths.
For ALD-related deaths, the annual percentage change was 3.5% for 2010-2019 and 17.6% for 2019-2021. The observed ASMR in 2020 was significantly higher than predicted, at 15.7 deaths per 100,000 people versus 13.0 predicted from the 2010-2019 rate. The trend continued in 2021, with 17.4 deaths per 100,000 people versus 13.4 in the previous decade.
The highest numbers of ALD-related deaths during the COVID-19 pandemic occurred in Alaska, Montana, Wyoming, Colorado, New Mexico, and South Dakota.
For NAFLD-related deaths, the annual percentage change was 7.6% for 2010-2014, 11.8% for 2014-2019, and 14.5% for 2019-2021. The observed ASMR was also higher than predicted, at 3.1 deaths per 100,000 people versus 2.6 in 2020, as well as 3.4 versus 2.8 in 2021.
The highest numbers of NAFLD-related deaths during the COVID-19 pandemic occurred in Oklahoma, Indiana, Kentucky, Tennessee, and West Virginia.
Hepatitis B and C gains lost in pandemic
In contrast, the annual percentage change in was –1.9% for hepatitis B and –2.8% for hepatitis C. After new treatment for hepatitis C emerged in 2013-2014, mortality rates were –7.8% for 2014-2019, Dr. Yeo noted.
“However, during the pandemic, we saw that this decrease has become a nonsignificant change,” he said. “That means our progress of the past 5 or 6 years has already stopped during the pandemic.”
By race and ethnicity, the increase in ALD-related mortality was most pronounced in non-Hispanic White, non-Hispanic Black, and Alaska Native/American Indian populations, Dr. Yeo said. Alaska Natives and American Indians had the highest annual percentage change, at 18%, followed by non-Hispanic Whites at 11.7% and non-Hispanic Blacks at 10.8%. There were no significant differences in race and ethnicity for NAFLD-related deaths, although all groups had major increases in recent years.
Biggest rise in young adults
By age, the increase in ALD-related mortality was particularly severe for ages 25-44, with an annual percentage change of 34.6% in 2019-2021, as compared with 13.7% for ages 45-64 and 12.6% for ages 65 and older.
For NAFLD-related deaths, another major increase was observed among ages 25-44, with an annual percentage change of 28.1% for 2019-2021, as compared with 12% for ages 65 and older and 7.4% for ages 45-64.
By sex, the ASMR increase in NAFLD-related mortality was steady throughout 2010-2021 for both men and women. In contrast, ALD-related death increased sharply between 2019 and 2021, with an annual percentage change of 19.1% for women and 16.7% for men.
“The increasing trend in mortality rates for ALD and NAFLD has been quite alarming, with disparities in age, race, and ethnicity,” Dr. Yeo said.
The study received no funding support. Some authors disclosed research funding, advisory board roles, and consulting fees with various pharmaceutical companies.
according to new findings presented at the annual meeting of the American Association for the Study of Liver Diseases.
Between 2019 and 2021, ALD-related deaths increased by 17.6% and NAFLD-related deaths increased by 14.5%, Yee Hui Yeo, MD, a resident physician and hepatology-focused investigator at Cedars-Sinai Medical Center in Los Angeles, said at a preconference press briefing.
“Even before the pandemic, the mortality rates for these two diseases have been increasing, with NAFLD having an even steeper increasing trend,” he said. “During the pandemic, these two diseases had a significant surge.”
Recent U.S. liver disease death rates
Dr. Yeo and colleagues analyzed data from the Center for Disease Control and Prevention’s National Vital Statistic System to estimate the age-standardized mortality rates (ASMR) of liver disease between 2010 and 2021, including ALD, NAFLD, hepatitis B, and hepatitis C. Using prediction modeling analyses based on trends from 2010 to 2019, they predicted mortality rates for 2020-2021 and compared them with the observed rates to quantify the differences related to the pandemic.
Between 2010 and 2021, there were about 626,000 chronic liver disease–related deaths, including about 343,000 ALD-related deaths, 204,000 hepatitis C–related deaths, 58,000 NAFLD-related deaths, and 21,000 hepatitis B–related deaths.
For ALD-related deaths, the annual percentage change was 3.5% for 2010-2019 and 17.6% for 2019-2021. The observed ASMR in 2020 was significantly higher than predicted, at 15.7 deaths per 100,000 people versus 13.0 predicted from the 2010-2019 rate. The trend continued in 2021, with 17.4 deaths per 100,000 people versus 13.4 in the previous decade.
The highest numbers of ALD-related deaths during the COVID-19 pandemic occurred in Alaska, Montana, Wyoming, Colorado, New Mexico, and South Dakota.
For NAFLD-related deaths, the annual percentage change was 7.6% for 2010-2014, 11.8% for 2014-2019, and 14.5% for 2019-2021. The observed ASMR was also higher than predicted, at 3.1 deaths per 100,000 people versus 2.6 in 2020, as well as 3.4 versus 2.8 in 2021.
The highest numbers of NAFLD-related deaths during the COVID-19 pandemic occurred in Oklahoma, Indiana, Kentucky, Tennessee, and West Virginia.
Hepatitis B and C gains lost in pandemic
In contrast, the annual percentage change in was –1.9% for hepatitis B and –2.8% for hepatitis C. After new treatment for hepatitis C emerged in 2013-2014, mortality rates were –7.8% for 2014-2019, Dr. Yeo noted.
“However, during the pandemic, we saw that this decrease has become a nonsignificant change,” he said. “That means our progress of the past 5 or 6 years has already stopped during the pandemic.”
By race and ethnicity, the increase in ALD-related mortality was most pronounced in non-Hispanic White, non-Hispanic Black, and Alaska Native/American Indian populations, Dr. Yeo said. Alaska Natives and American Indians had the highest annual percentage change, at 18%, followed by non-Hispanic Whites at 11.7% and non-Hispanic Blacks at 10.8%. There were no significant differences in race and ethnicity for NAFLD-related deaths, although all groups had major increases in recent years.
Biggest rise in young adults
By age, the increase in ALD-related mortality was particularly severe for ages 25-44, with an annual percentage change of 34.6% in 2019-2021, as compared with 13.7% for ages 45-64 and 12.6% for ages 65 and older.
For NAFLD-related deaths, another major increase was observed among ages 25-44, with an annual percentage change of 28.1% for 2019-2021, as compared with 12% for ages 65 and older and 7.4% for ages 45-64.
By sex, the ASMR increase in NAFLD-related mortality was steady throughout 2010-2021 for both men and women. In contrast, ALD-related death increased sharply between 2019 and 2021, with an annual percentage change of 19.1% for women and 16.7% for men.
“The increasing trend in mortality rates for ALD and NAFLD has been quite alarming, with disparities in age, race, and ethnicity,” Dr. Yeo said.
The study received no funding support. Some authors disclosed research funding, advisory board roles, and consulting fees with various pharmaceutical companies.
according to new findings presented at the annual meeting of the American Association for the Study of Liver Diseases.
Between 2019 and 2021, ALD-related deaths increased by 17.6% and NAFLD-related deaths increased by 14.5%, Yee Hui Yeo, MD, a resident physician and hepatology-focused investigator at Cedars-Sinai Medical Center in Los Angeles, said at a preconference press briefing.
“Even before the pandemic, the mortality rates for these two diseases have been increasing, with NAFLD having an even steeper increasing trend,” he said. “During the pandemic, these two diseases had a significant surge.”
Recent U.S. liver disease death rates
Dr. Yeo and colleagues analyzed data from the Center for Disease Control and Prevention’s National Vital Statistic System to estimate the age-standardized mortality rates (ASMR) of liver disease between 2010 and 2021, including ALD, NAFLD, hepatitis B, and hepatitis C. Using prediction modeling analyses based on trends from 2010 to 2019, they predicted mortality rates for 2020-2021 and compared them with the observed rates to quantify the differences related to the pandemic.
Between 2010 and 2021, there were about 626,000 chronic liver disease–related deaths, including about 343,000 ALD-related deaths, 204,000 hepatitis C–related deaths, 58,000 NAFLD-related deaths, and 21,000 hepatitis B–related deaths.
For ALD-related deaths, the annual percentage change was 3.5% for 2010-2019 and 17.6% for 2019-2021. The observed ASMR in 2020 was significantly higher than predicted, at 15.7 deaths per 100,000 people versus 13.0 predicted from the 2010-2019 rate. The trend continued in 2021, with 17.4 deaths per 100,000 people versus 13.4 in the previous decade.
The highest numbers of ALD-related deaths during the COVID-19 pandemic occurred in Alaska, Montana, Wyoming, Colorado, New Mexico, and South Dakota.
For NAFLD-related deaths, the annual percentage change was 7.6% for 2010-2014, 11.8% for 2014-2019, and 14.5% for 2019-2021. The observed ASMR was also higher than predicted, at 3.1 deaths per 100,000 people versus 2.6 in 2020, as well as 3.4 versus 2.8 in 2021.
The highest numbers of NAFLD-related deaths during the COVID-19 pandemic occurred in Oklahoma, Indiana, Kentucky, Tennessee, and West Virginia.
Hepatitis B and C gains lost in pandemic
In contrast, the annual percentage change in was –1.9% for hepatitis B and –2.8% for hepatitis C. After new treatment for hepatitis C emerged in 2013-2014, mortality rates were –7.8% for 2014-2019, Dr. Yeo noted.
“However, during the pandemic, we saw that this decrease has become a nonsignificant change,” he said. “That means our progress of the past 5 or 6 years has already stopped during the pandemic.”
By race and ethnicity, the increase in ALD-related mortality was most pronounced in non-Hispanic White, non-Hispanic Black, and Alaska Native/American Indian populations, Dr. Yeo said. Alaska Natives and American Indians had the highest annual percentage change, at 18%, followed by non-Hispanic Whites at 11.7% and non-Hispanic Blacks at 10.8%. There were no significant differences in race and ethnicity for NAFLD-related deaths, although all groups had major increases in recent years.
Biggest rise in young adults
By age, the increase in ALD-related mortality was particularly severe for ages 25-44, with an annual percentage change of 34.6% in 2019-2021, as compared with 13.7% for ages 45-64 and 12.6% for ages 65 and older.
For NAFLD-related deaths, another major increase was observed among ages 25-44, with an annual percentage change of 28.1% for 2019-2021, as compared with 12% for ages 65 and older and 7.4% for ages 45-64.
By sex, the ASMR increase in NAFLD-related mortality was steady throughout 2010-2021 for both men and women. In contrast, ALD-related death increased sharply between 2019 and 2021, with an annual percentage change of 19.1% for women and 16.7% for men.
“The increasing trend in mortality rates for ALD and NAFLD has been quite alarming, with disparities in age, race, and ethnicity,” Dr. Yeo said.
The study received no funding support. Some authors disclosed research funding, advisory board roles, and consulting fees with various pharmaceutical companies.
FROM THE LIVER MEETING
Have you heard the one about the emergency dept. that called 911?
Who watches the ED staff?
We heard a really great joke recently, one we simply have to share.
A man in Seattle went to a therapist. “I’m depressed,” he says. “Depressed, overworked, and lonely.”
“Oh dear, that sounds quite serious,” the therapist replies. “Tell me all about it.”
“Life just seems so harsh and cruel,” the man explains. “The pandemic has caused 300,000 health care workers across the country to leave the industry.”
“Such as the doctor typically filling this role in the joke,” the therapist, who is not licensed to prescribe medicine, nods.
“Exactly! And with so many respiratory viruses circulating and COVID still hanging around, emergency departments all over the country are facing massive backups. People are waiting outside the hospital for hours, hoping a bed will open up. Things got so bad at a hospital near Seattle in October that a nurse called 911 on her own ED. Told the 911 operator to send the fire department to help out, since they were ‘drowning’ and ‘in dire straits.’ They had 45 patients waiting and only five nurses to take care of them.”
“That is quite serious,” the therapist says, scribbling down unseen notes.
“The fire chief did send a crew out, and they cleaned rooms, changed beds, and took vitals for 90 minutes until the crisis passed,” the man says. “But it’s only a matter of time before it happens again. The hospital president said they have 300 open positions, and literally no one has applied to work in the emergency department. Not one person.”
“And how does all this make you feel?” the therapist asks.
“I feel all alone,” the man says. “This world feels so threatening, like no one cares, and I have no idea what will come next. It’s so vague and uncertain.”
“Ah, I think I have a solution for you,” the therapist says. “Go to the emergency department at St. Michael Medical Center in Silverdale, near Seattle. They’ll get your bad mood all settled, and they’ll prescribe you the medicine you need to relax.”
The man bursts into tears. “You don’t understand,” he says. “I am the emergency department at St. Michael Medical Center.”
Good joke. Everybody laugh. Roll on snare drum. Curtains.
Myth buster: Supplements for cholesterol lowering
When it comes to that nasty low-density lipoprotein cholesterol, some people swear by supplements over statins as a holistic approach. Well, we’re busting the myth that those heart-healthy supplements are even effective in comparison.
Which supplements are we talking about? These six are always on sale at the pharmacy: fish oil, cinnamon, garlic, turmeric, plant sterols, and red yeast rice.
In a study presented at the recent American Heart Association scientific sessions, researchers compared these supplements’ effectiveness in lowering LDL cholesterol with low-dose rosuvastatin or placebo among 199 adults aged 40-75 years who didn’t have a personal history of cardiovascular disease.
Participants who took the statin for 28 days had an average of 24% decrease in total cholesterol and a 38% reduction in LDL cholesterol, while 28 days’ worth of the supplements did no better than the placebo in either measure. Compared with placebo, the plant sterols supplement notably lowered HDL cholesterol and the garlic supplement notably increased LDL cholesterol.
Even though there are other studies showing the validity of plant sterols and red yeast rice to lower LDL cholesterol, author Luke J. Laffin, MD, of the Cleveland Clinic noted that this study shows how supplement results can vary and that more research is needed to see the effect they truly have on cholesterol over time.
So, should you stop taking or recommending supplements for heart health or healthy cholesterol levels? Well, we’re not going to come to your house and raid your medicine cabinet, but the authors of this study are definitely not saying that you should rely on them.
Consider this myth mostly busted.
COVID dept. of unintended consequences, part 2
The surveillance testing programs conducted in the first year of the pandemic were, in theory, meant to keep everyone safer. Someone, apparently, forgot to explain that to the students of the University of Wyoming and the University of Idaho.
We’re all familiar with the drill: Students at the two schools had to undergo frequent COVID screening to keep the virus from spreading, thereby making everyone safer. Duck your head now, because here comes the unintended consequence.
The students who didn’t get COVID eventually, and perhaps not so surprisingly, “perceived that the mandatory testing policy decreased their risk of contracting COVID-19, and … this perception led to higher participation in COVID-risky events,” Chian Jones Ritten, PhD, and associates said in PNAS Nexus.
They surveyed 757 students from the Univ. of Washington and 517 from the Univ. of Idaho and found that those who were tested more frequently perceived that they were less likely to contract the virus. Those respondents also more frequently attended indoor gatherings, both small and large, and spent more time in restaurants and bars.
The investigators did not mince words: “From a public health standpoint, such behavior is problematic.”
Current parents/participants in the workforce might have other ideas about an appropriate response to COVID.
At this point, we probably should mention that appropriation is the second-most sincere form of flattery.
Who watches the ED staff?
We heard a really great joke recently, one we simply have to share.
A man in Seattle went to a therapist. “I’m depressed,” he says. “Depressed, overworked, and lonely.”
“Oh dear, that sounds quite serious,” the therapist replies. “Tell me all about it.”
“Life just seems so harsh and cruel,” the man explains. “The pandemic has caused 300,000 health care workers across the country to leave the industry.”
“Such as the doctor typically filling this role in the joke,” the therapist, who is not licensed to prescribe medicine, nods.
“Exactly! And with so many respiratory viruses circulating and COVID still hanging around, emergency departments all over the country are facing massive backups. People are waiting outside the hospital for hours, hoping a bed will open up. Things got so bad at a hospital near Seattle in October that a nurse called 911 on her own ED. Told the 911 operator to send the fire department to help out, since they were ‘drowning’ and ‘in dire straits.’ They had 45 patients waiting and only five nurses to take care of them.”
“That is quite serious,” the therapist says, scribbling down unseen notes.
“The fire chief did send a crew out, and they cleaned rooms, changed beds, and took vitals for 90 minutes until the crisis passed,” the man says. “But it’s only a matter of time before it happens again. The hospital president said they have 300 open positions, and literally no one has applied to work in the emergency department. Not one person.”
“And how does all this make you feel?” the therapist asks.
“I feel all alone,” the man says. “This world feels so threatening, like no one cares, and I have no idea what will come next. It’s so vague and uncertain.”
“Ah, I think I have a solution for you,” the therapist says. “Go to the emergency department at St. Michael Medical Center in Silverdale, near Seattle. They’ll get your bad mood all settled, and they’ll prescribe you the medicine you need to relax.”
The man bursts into tears. “You don’t understand,” he says. “I am the emergency department at St. Michael Medical Center.”
Good joke. Everybody laugh. Roll on snare drum. Curtains.
Myth buster: Supplements for cholesterol lowering
When it comes to that nasty low-density lipoprotein cholesterol, some people swear by supplements over statins as a holistic approach. Well, we’re busting the myth that those heart-healthy supplements are even effective in comparison.
Which supplements are we talking about? These six are always on sale at the pharmacy: fish oil, cinnamon, garlic, turmeric, plant sterols, and red yeast rice.
In a study presented at the recent American Heart Association scientific sessions, researchers compared these supplements’ effectiveness in lowering LDL cholesterol with low-dose rosuvastatin or placebo among 199 adults aged 40-75 years who didn’t have a personal history of cardiovascular disease.
Participants who took the statin for 28 days had an average of 24% decrease in total cholesterol and a 38% reduction in LDL cholesterol, while 28 days’ worth of the supplements did no better than the placebo in either measure. Compared with placebo, the plant sterols supplement notably lowered HDL cholesterol and the garlic supplement notably increased LDL cholesterol.
Even though there are other studies showing the validity of plant sterols and red yeast rice to lower LDL cholesterol, author Luke J. Laffin, MD, of the Cleveland Clinic noted that this study shows how supplement results can vary and that more research is needed to see the effect they truly have on cholesterol over time.
So, should you stop taking or recommending supplements for heart health or healthy cholesterol levels? Well, we’re not going to come to your house and raid your medicine cabinet, but the authors of this study are definitely not saying that you should rely on them.
Consider this myth mostly busted.
COVID dept. of unintended consequences, part 2
The surveillance testing programs conducted in the first year of the pandemic were, in theory, meant to keep everyone safer. Someone, apparently, forgot to explain that to the students of the University of Wyoming and the University of Idaho.
We’re all familiar with the drill: Students at the two schools had to undergo frequent COVID screening to keep the virus from spreading, thereby making everyone safer. Duck your head now, because here comes the unintended consequence.
The students who didn’t get COVID eventually, and perhaps not so surprisingly, “perceived that the mandatory testing policy decreased their risk of contracting COVID-19, and … this perception led to higher participation in COVID-risky events,” Chian Jones Ritten, PhD, and associates said in PNAS Nexus.
They surveyed 757 students from the Univ. of Washington and 517 from the Univ. of Idaho and found that those who were tested more frequently perceived that they were less likely to contract the virus. Those respondents also more frequently attended indoor gatherings, both small and large, and spent more time in restaurants and bars.
The investigators did not mince words: “From a public health standpoint, such behavior is problematic.”
Current parents/participants in the workforce might have other ideas about an appropriate response to COVID.
At this point, we probably should mention that appropriation is the second-most sincere form of flattery.
Who watches the ED staff?
We heard a really great joke recently, one we simply have to share.
A man in Seattle went to a therapist. “I’m depressed,” he says. “Depressed, overworked, and lonely.”
“Oh dear, that sounds quite serious,” the therapist replies. “Tell me all about it.”
“Life just seems so harsh and cruel,” the man explains. “The pandemic has caused 300,000 health care workers across the country to leave the industry.”
“Such as the doctor typically filling this role in the joke,” the therapist, who is not licensed to prescribe medicine, nods.
“Exactly! And with so many respiratory viruses circulating and COVID still hanging around, emergency departments all over the country are facing massive backups. People are waiting outside the hospital for hours, hoping a bed will open up. Things got so bad at a hospital near Seattle in October that a nurse called 911 on her own ED. Told the 911 operator to send the fire department to help out, since they were ‘drowning’ and ‘in dire straits.’ They had 45 patients waiting and only five nurses to take care of them.”
“That is quite serious,” the therapist says, scribbling down unseen notes.
“The fire chief did send a crew out, and they cleaned rooms, changed beds, and took vitals for 90 minutes until the crisis passed,” the man says. “But it’s only a matter of time before it happens again. The hospital president said they have 300 open positions, and literally no one has applied to work in the emergency department. Not one person.”
“And how does all this make you feel?” the therapist asks.
“I feel all alone,” the man says. “This world feels so threatening, like no one cares, and I have no idea what will come next. It’s so vague and uncertain.”
“Ah, I think I have a solution for you,” the therapist says. “Go to the emergency department at St. Michael Medical Center in Silverdale, near Seattle. They’ll get your bad mood all settled, and they’ll prescribe you the medicine you need to relax.”
The man bursts into tears. “You don’t understand,” he says. “I am the emergency department at St. Michael Medical Center.”
Good joke. Everybody laugh. Roll on snare drum. Curtains.
Myth buster: Supplements for cholesterol lowering
When it comes to that nasty low-density lipoprotein cholesterol, some people swear by supplements over statins as a holistic approach. Well, we’re busting the myth that those heart-healthy supplements are even effective in comparison.
Which supplements are we talking about? These six are always on sale at the pharmacy: fish oil, cinnamon, garlic, turmeric, plant sterols, and red yeast rice.
In a study presented at the recent American Heart Association scientific sessions, researchers compared these supplements’ effectiveness in lowering LDL cholesterol with low-dose rosuvastatin or placebo among 199 adults aged 40-75 years who didn’t have a personal history of cardiovascular disease.
Participants who took the statin for 28 days had an average of 24% decrease in total cholesterol and a 38% reduction in LDL cholesterol, while 28 days’ worth of the supplements did no better than the placebo in either measure. Compared with placebo, the plant sterols supplement notably lowered HDL cholesterol and the garlic supplement notably increased LDL cholesterol.
Even though there are other studies showing the validity of plant sterols and red yeast rice to lower LDL cholesterol, author Luke J. Laffin, MD, of the Cleveland Clinic noted that this study shows how supplement results can vary and that more research is needed to see the effect they truly have on cholesterol over time.
So, should you stop taking or recommending supplements for heart health or healthy cholesterol levels? Well, we’re not going to come to your house and raid your medicine cabinet, but the authors of this study are definitely not saying that you should rely on them.
Consider this myth mostly busted.
COVID dept. of unintended consequences, part 2
The surveillance testing programs conducted in the first year of the pandemic were, in theory, meant to keep everyone safer. Someone, apparently, forgot to explain that to the students of the University of Wyoming and the University of Idaho.
We’re all familiar with the drill: Students at the two schools had to undergo frequent COVID screening to keep the virus from spreading, thereby making everyone safer. Duck your head now, because here comes the unintended consequence.
The students who didn’t get COVID eventually, and perhaps not so surprisingly, “perceived that the mandatory testing policy decreased their risk of contracting COVID-19, and … this perception led to higher participation in COVID-risky events,” Chian Jones Ritten, PhD, and associates said in PNAS Nexus.
They surveyed 757 students from the Univ. of Washington and 517 from the Univ. of Idaho and found that those who were tested more frequently perceived that they were less likely to contract the virus. Those respondents also more frequently attended indoor gatherings, both small and large, and spent more time in restaurants and bars.
The investigators did not mince words: “From a public health standpoint, such behavior is problematic.”
Current parents/participants in the workforce might have other ideas about an appropriate response to COVID.
At this point, we probably should mention that appropriation is the second-most sincere form of flattery.
Disaster Preparedness in Dermatology Residency Programs
In an age of changing climate and emerging global pandemics, the ability of residency programs to prepare for and adapt to potential disasters may be paramount in preserving the training of physicians. The current literature regarding residency program disaster preparedness, which focuses predominantly on hurricanes and COVID-19,1-8 is lacking in recommendations specific to dermatology residency programs. Likewise, the Accreditation Council for Graduate Medical Education (ACGME) guidelines9 do not address dermatology-specific concerns in disaster preparedness or response. Herein, we propose recommendations to mitigate the impact of various types of disasters on dermatology residency programs and their trainees with regard to resident safety and wellness, resident education, and patient care (Table).
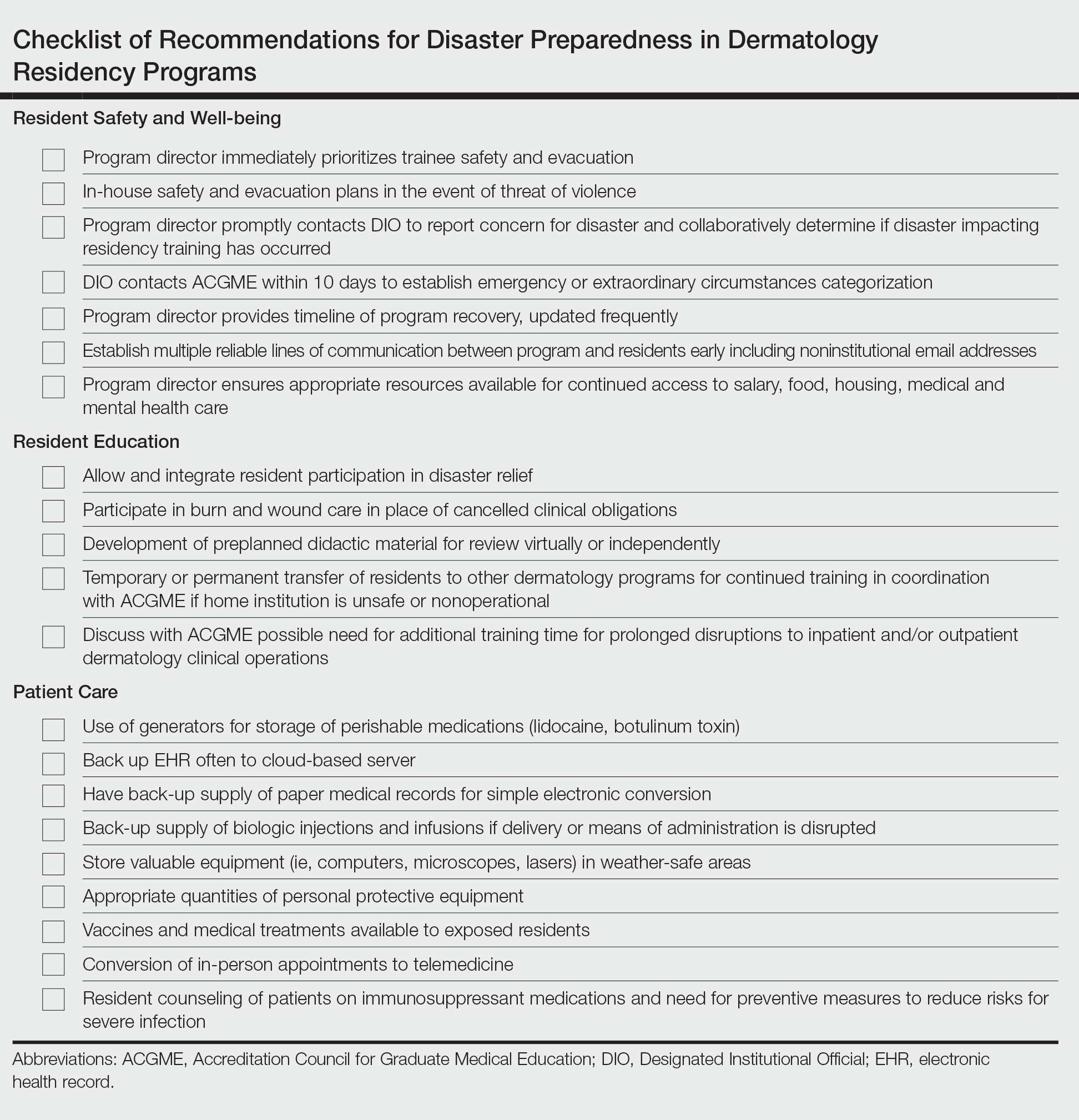
Resident Safety and Wellness
Role of the Program Director—The role of the program director is critical, serving as a figure of structure and reassurance.4,7,10 Once concern of disaster arises, the program director should contact the Designated Institutional Official (DIO) to express concerns about possible disruptions to resident training. The DIO should then contact the ACGME within 10 days to report the disaster and submit a request for emergency (eg, pandemic) or extraordinary circumstances (eg, natural disaster) categorization.4,9 Program directors should promptly prepare plans for program reconfiguration and resident transfers in alignment with ACGME requirements to maintain evaluation and completion of core competencies of training during disasters.9 Program directors should prioritize the safety of trainees during the immediate threat with clear guidelines on sheltering, evacuations, or quarantines; a timeline of program recovery based on communication with residents, faculty, and administration should then be established.10,11
Communication—Establishing a strong line of communication between program directors and residents is paramount. Collection of emergency noninstitutional contact information, establishment of a centralized website for information dissemination, use of noninstitutional email and proxy servers outside of the location of impact, social media updates, on-site use of 2-way radios, and program-wide conference calls when possible should be strongly considered as part of the disaster response.2-4,12,13
Resident Accommodations and Mental Health—If training is disrupted, residents should be reassured of continued access to salary, housing, food, or other resources as necessary.3,4,11 There should be clear contingency plans if residents need to leave the program for extended periods of time due to injury, illness, or personal circumstances. Although relevant in all types of disasters, resident mental health and response to trauma also must be addressed. Access to counseling, morale-building opportunities (eg, resident social events), and screening for depression or posttraumatic stress disorder may help promote well-being among residents following traumatic events.14
Resident Education
Participation in Disaster Relief—Residents may seek to aid in the disaster response, which may prove challenging in the setting of programs with high patient volume.4 In coordination with the ACGME and graduate medical education governing bodies, program directors should consider how residents may fulfill dermatology training requirements in conjunction with disaster relief efforts, such as working in an inpatient setting or providing wound care.10
Continued Didactic Education—The use of online learning and conference calls for continuing the dermatology curriculum is an efficient means to maintaining resident education when meeting in person poses risks to residents.15 Projections of microscopy images, clinical photographs, or other instructional materials allow for continued instruction on resident examination, histopathology, and diagnostic skills.
Continued Clinical Training—If the home institution cannot support the operation of dermatology clinics, residents should be guaranteed continued training at other institutions. Agreements with other dermatology programs, community hospitals, or private dermatology practices should be established in advance, with consideration given to the number of residents a program can support, funding transfers, and credentialing requirements.2,4,5
Prolonged Disruptions—Nonessential departments of medical institutions may cease to function during war or mass casualty disasters, and it may be unsafe to send dermatology residents to other institutions or clinical areas. If the threat is prolonged, programs may need to consider allowing current residents a longer duration of training despite potential overlap with incoming dermatology residents.7
Patient Care
Disruptions to Clinic Operations—Regarding threats of violence, dangerous exposures, or natural disasters, there should be clear guidelines on sheltering in the clinical setting or stabilizing patients during a procedure.11 Equipment used by residents such as laptops, microscopes, and treatment devices (eg, lasers) should be stored in weather-safe locations that would not be notably impacted by moisture or structural damage to the clinic building. If electricity or internet access are compromised, paper medical records should be available to residents to continue clinical operations. Electronic health records used by residents should regularly be backed up on remote servers or cloud storage to allow continued access to patient health information if on-site servers are not functional.12 If disruptions are prolonged, residency program administration should coordinate with the institution to ensure there is adequate supply and storage of medications (eg, lidocaine, botulinum toxin) as well as a continued means of delivering biologic medications to patients and an ability to obtain laboratory or dermatopathology services.
In-Person Appointments vs Telemedicine—There are benefits to both residency training and patient care when physicians are able to perform in-person examinations, biopsies, and in-office treatments.16 Programs should ensure an adequate supply of personal protective equipment to continue in-office appointments, vaccinations, and medical care if a resident or other members of the team are exposed to an infectious disease.7 If in-person appointments are limited or impossible, telemedicine capabilities may still allow residents to meet program requirements.7,10,15 However, reduced patient volume due to decreased elective visits or procedures may complicate the fulfillment of clinical requirements, which may need to be adjusted in the wake of a disaster.7
Use of Immunosuppressive Therapies—Residency programs should address the risks of prescribing immunosuppressive therapies (eg, biologics) during an infectious threat with their residents and encourage trainees to counsel patients on the importance of preventative measures to reduce risks for severe infection.17
Final Thoughts
- Davis W. Hurricane Katrina: the challenge to graduate medical education. Ochsner J. 2006;6:39.
- Cefalu CA, Schwartz RS. Salvaging a geriatric medicine academic program in disaster mode—the LSU training program post-Katrina.J Natl Med Assoc. 2007;99:590-596.
- Ayyala R. Lessons from Katrina: a program director’s perspective. Ophthalmology. 2007;114:1425-1426.
- Wiese JG. Leadership in graduate medical education: eleven steps instrumental in recovering residency programs after a disaster. Am J Med Sci. 2008;336:168-173.
- Griffies WS. Post-Katrina stabilization of the LSU/Ochsner Psychiatry Residency Program: caveats for disaster preparedness. Acad Psychiatry. 2009;33:418-422.
- Kearns DG, Chat VS, Uppal S, et al. Applying to dermatology residency during the COVID-19 pandemic. J Am Acad Dermatol. 2020;83:1214-1215.
- Matthews JB, Blair PG, Ellison EC, et al. Checklist framework for surgical education disaster plans. J Am Coll Surg. 2021;233:557-563.
- Litchman GH, Marson JW, Rigel DS. The continuing impact of COVID-19 on dermatology practice: office workflow, economics, and future implications. J Am Acad Dermatol. 2021;84:576-579.
- Accreditation Council for Graduate Medical Education. Sponsoring institution emergency categorization. Accessed October 20, 2022. https://www.acgme.org/covid-19/sponsoring-institution-emergency-categorization/
- Li YM, Galimberti F, Abrouk M, et al. US dermatology resident responses about the COVID-19 pandemic: results from a nationwide survey. South Med J. 2020;113:462-465.
- Newman B, Gallion C. Hurricane Harvey: firsthand perspectives for disaster preparedness in graduate medical education. Acad Med. 2019;94:1267-1269.
- Pero CD, Pou AM, Arriaga MA, et al. Post-Katrina: study in crisis-related program adaptability. Otolaryngol Head Neck Surg. 2008;138:394-397.
- Hattaway R, Singh N, Rais-Bahrami S, et al. Adaptations of dermatology residency programs to changes in medical education amid the COVID-19 pandemic: virtual opportunities and social media. SKIN. 2021;5:94-100.
- Hillier K, Paskaradevan J, Wilkes JK, et al. Disaster plans: resident involvement and well-being during Hurricane Harvey. J Grad Med Educ. 2019;11:129-131.
- Samimi S, Choi J, Rosman IS, et al. Impact of COVID-19 on dermatology residency. Dermatol Clin. 2021;39:609-618.
- Bastola M, Locatis C, Fontelo P. Diagnostic reliability of in-person versus remote dermatology: a meta-analysis. Telemed J E Health. 2021;27:247-250.
- Bashyam AM, Feldman SR. Should patients stop their biologic treatment during the COVID-19 pandemic? J Dermatolog Treat. 2020;31:317-318.
In an age of changing climate and emerging global pandemics, the ability of residency programs to prepare for and adapt to potential disasters may be paramount in preserving the training of physicians. The current literature regarding residency program disaster preparedness, which focuses predominantly on hurricanes and COVID-19,1-8 is lacking in recommendations specific to dermatology residency programs. Likewise, the Accreditation Council for Graduate Medical Education (ACGME) guidelines9 do not address dermatology-specific concerns in disaster preparedness or response. Herein, we propose recommendations to mitigate the impact of various types of disasters on dermatology residency programs and their trainees with regard to resident safety and wellness, resident education, and patient care (Table).

Resident Safety and Wellness
Role of the Program Director—The role of the program director is critical, serving as a figure of structure and reassurance.4,7,10 Once concern of disaster arises, the program director should contact the Designated Institutional Official (DIO) to express concerns about possible disruptions to resident training. The DIO should then contact the ACGME within 10 days to report the disaster and submit a request for emergency (eg, pandemic) or extraordinary circumstances (eg, natural disaster) categorization.4,9 Program directors should promptly prepare plans for program reconfiguration and resident transfers in alignment with ACGME requirements to maintain evaluation and completion of core competencies of training during disasters.9 Program directors should prioritize the safety of trainees during the immediate threat with clear guidelines on sheltering, evacuations, or quarantines; a timeline of program recovery based on communication with residents, faculty, and administration should then be established.10,11
Communication—Establishing a strong line of communication between program directors and residents is paramount. Collection of emergency noninstitutional contact information, establishment of a centralized website for information dissemination, use of noninstitutional email and proxy servers outside of the location of impact, social media updates, on-site use of 2-way radios, and program-wide conference calls when possible should be strongly considered as part of the disaster response.2-4,12,13
Resident Accommodations and Mental Health—If training is disrupted, residents should be reassured of continued access to salary, housing, food, or other resources as necessary.3,4,11 There should be clear contingency plans if residents need to leave the program for extended periods of time due to injury, illness, or personal circumstances. Although relevant in all types of disasters, resident mental health and response to trauma also must be addressed. Access to counseling, morale-building opportunities (eg, resident social events), and screening for depression or posttraumatic stress disorder may help promote well-being among residents following traumatic events.14
Resident Education
Participation in Disaster Relief—Residents may seek to aid in the disaster response, which may prove challenging in the setting of programs with high patient volume.4 In coordination with the ACGME and graduate medical education governing bodies, program directors should consider how residents may fulfill dermatology training requirements in conjunction with disaster relief efforts, such as working in an inpatient setting or providing wound care.10
Continued Didactic Education—The use of online learning and conference calls for continuing the dermatology curriculum is an efficient means to maintaining resident education when meeting in person poses risks to residents.15 Projections of microscopy images, clinical photographs, or other instructional materials allow for continued instruction on resident examination, histopathology, and diagnostic skills.
Continued Clinical Training—If the home institution cannot support the operation of dermatology clinics, residents should be guaranteed continued training at other institutions. Agreements with other dermatology programs, community hospitals, or private dermatology practices should be established in advance, with consideration given to the number of residents a program can support, funding transfers, and credentialing requirements.2,4,5
Prolonged Disruptions—Nonessential departments of medical institutions may cease to function during war or mass casualty disasters, and it may be unsafe to send dermatology residents to other institutions or clinical areas. If the threat is prolonged, programs may need to consider allowing current residents a longer duration of training despite potential overlap with incoming dermatology residents.7
Patient Care
Disruptions to Clinic Operations—Regarding threats of violence, dangerous exposures, or natural disasters, there should be clear guidelines on sheltering in the clinical setting or stabilizing patients during a procedure.11 Equipment used by residents such as laptops, microscopes, and treatment devices (eg, lasers) should be stored in weather-safe locations that would not be notably impacted by moisture or structural damage to the clinic building. If electricity or internet access are compromised, paper medical records should be available to residents to continue clinical operations. Electronic health records used by residents should regularly be backed up on remote servers or cloud storage to allow continued access to patient health information if on-site servers are not functional.12 If disruptions are prolonged, residency program administration should coordinate with the institution to ensure there is adequate supply and storage of medications (eg, lidocaine, botulinum toxin) as well as a continued means of delivering biologic medications to patients and an ability to obtain laboratory or dermatopathology services.
In-Person Appointments vs Telemedicine—There are benefits to both residency training and patient care when physicians are able to perform in-person examinations, biopsies, and in-office treatments.16 Programs should ensure an adequate supply of personal protective equipment to continue in-office appointments, vaccinations, and medical care if a resident or other members of the team are exposed to an infectious disease.7 If in-person appointments are limited or impossible, telemedicine capabilities may still allow residents to meet program requirements.7,10,15 However, reduced patient volume due to decreased elective visits or procedures may complicate the fulfillment of clinical requirements, which may need to be adjusted in the wake of a disaster.7
Use of Immunosuppressive Therapies—Residency programs should address the risks of prescribing immunosuppressive therapies (eg, biologics) during an infectious threat with their residents and encourage trainees to counsel patients on the importance of preventative measures to reduce risks for severe infection.17
Final Thoughts
In an age of changing climate and emerging global pandemics, the ability of residency programs to prepare for and adapt to potential disasters may be paramount in preserving the training of physicians. The current literature regarding residency program disaster preparedness, which focuses predominantly on hurricanes and COVID-19,1-8 is lacking in recommendations specific to dermatology residency programs. Likewise, the Accreditation Council for Graduate Medical Education (ACGME) guidelines9 do not address dermatology-specific concerns in disaster preparedness or response. Herein, we propose recommendations to mitigate the impact of various types of disasters on dermatology residency programs and their trainees with regard to resident safety and wellness, resident education, and patient care (Table).

Resident Safety and Wellness
Role of the Program Director—The role of the program director is critical, serving as a figure of structure and reassurance.4,7,10 Once concern of disaster arises, the program director should contact the Designated Institutional Official (DIO) to express concerns about possible disruptions to resident training. The DIO should then contact the ACGME within 10 days to report the disaster and submit a request for emergency (eg, pandemic) or extraordinary circumstances (eg, natural disaster) categorization.4,9 Program directors should promptly prepare plans for program reconfiguration and resident transfers in alignment with ACGME requirements to maintain evaluation and completion of core competencies of training during disasters.9 Program directors should prioritize the safety of trainees during the immediate threat with clear guidelines on sheltering, evacuations, or quarantines; a timeline of program recovery based on communication with residents, faculty, and administration should then be established.10,11
Communication—Establishing a strong line of communication between program directors and residents is paramount. Collection of emergency noninstitutional contact information, establishment of a centralized website for information dissemination, use of noninstitutional email and proxy servers outside of the location of impact, social media updates, on-site use of 2-way radios, and program-wide conference calls when possible should be strongly considered as part of the disaster response.2-4,12,13
Resident Accommodations and Mental Health—If training is disrupted, residents should be reassured of continued access to salary, housing, food, or other resources as necessary.3,4,11 There should be clear contingency plans if residents need to leave the program for extended periods of time due to injury, illness, or personal circumstances. Although relevant in all types of disasters, resident mental health and response to trauma also must be addressed. Access to counseling, morale-building opportunities (eg, resident social events), and screening for depression or posttraumatic stress disorder may help promote well-being among residents following traumatic events.14
Resident Education
Participation in Disaster Relief—Residents may seek to aid in the disaster response, which may prove challenging in the setting of programs with high patient volume.4 In coordination with the ACGME and graduate medical education governing bodies, program directors should consider how residents may fulfill dermatology training requirements in conjunction with disaster relief efforts, such as working in an inpatient setting or providing wound care.10
Continued Didactic Education—The use of online learning and conference calls for continuing the dermatology curriculum is an efficient means to maintaining resident education when meeting in person poses risks to residents.15 Projections of microscopy images, clinical photographs, or other instructional materials allow for continued instruction on resident examination, histopathology, and diagnostic skills.
Continued Clinical Training—If the home institution cannot support the operation of dermatology clinics, residents should be guaranteed continued training at other institutions. Agreements with other dermatology programs, community hospitals, or private dermatology practices should be established in advance, with consideration given to the number of residents a program can support, funding transfers, and credentialing requirements.2,4,5
Prolonged Disruptions—Nonessential departments of medical institutions may cease to function during war or mass casualty disasters, and it may be unsafe to send dermatology residents to other institutions or clinical areas. If the threat is prolonged, programs may need to consider allowing current residents a longer duration of training despite potential overlap with incoming dermatology residents.7
Patient Care
Disruptions to Clinic Operations—Regarding threats of violence, dangerous exposures, or natural disasters, there should be clear guidelines on sheltering in the clinical setting or stabilizing patients during a procedure.11 Equipment used by residents such as laptops, microscopes, and treatment devices (eg, lasers) should be stored in weather-safe locations that would not be notably impacted by moisture or structural damage to the clinic building. If electricity or internet access are compromised, paper medical records should be available to residents to continue clinical operations. Electronic health records used by residents should regularly be backed up on remote servers or cloud storage to allow continued access to patient health information if on-site servers are not functional.12 If disruptions are prolonged, residency program administration should coordinate with the institution to ensure there is adequate supply and storage of medications (eg, lidocaine, botulinum toxin) as well as a continued means of delivering biologic medications to patients and an ability to obtain laboratory or dermatopathology services.
In-Person Appointments vs Telemedicine—There are benefits to both residency training and patient care when physicians are able to perform in-person examinations, biopsies, and in-office treatments.16 Programs should ensure an adequate supply of personal protective equipment to continue in-office appointments, vaccinations, and medical care if a resident or other members of the team are exposed to an infectious disease.7 If in-person appointments are limited or impossible, telemedicine capabilities may still allow residents to meet program requirements.7,10,15 However, reduced patient volume due to decreased elective visits or procedures may complicate the fulfillment of clinical requirements, which may need to be adjusted in the wake of a disaster.7
Use of Immunosuppressive Therapies—Residency programs should address the risks of prescribing immunosuppressive therapies (eg, biologics) during an infectious threat with their residents and encourage trainees to counsel patients on the importance of preventative measures to reduce risks for severe infection.17
Final Thoughts
- Davis W. Hurricane Katrina: the challenge to graduate medical education. Ochsner J. 2006;6:39.
- Cefalu CA, Schwartz RS. Salvaging a geriatric medicine academic program in disaster mode—the LSU training program post-Katrina.J Natl Med Assoc. 2007;99:590-596.
- Ayyala R. Lessons from Katrina: a program director’s perspective. Ophthalmology. 2007;114:1425-1426.
- Wiese JG. Leadership in graduate medical education: eleven steps instrumental in recovering residency programs after a disaster. Am J Med Sci. 2008;336:168-173.
- Griffies WS. Post-Katrina stabilization of the LSU/Ochsner Psychiatry Residency Program: caveats for disaster preparedness. Acad Psychiatry. 2009;33:418-422.
- Kearns DG, Chat VS, Uppal S, et al. Applying to dermatology residency during the COVID-19 pandemic. J Am Acad Dermatol. 2020;83:1214-1215.
- Matthews JB, Blair PG, Ellison EC, et al. Checklist framework for surgical education disaster plans. J Am Coll Surg. 2021;233:557-563.
- Litchman GH, Marson JW, Rigel DS. The continuing impact of COVID-19 on dermatology practice: office workflow, economics, and future implications. J Am Acad Dermatol. 2021;84:576-579.
- Accreditation Council for Graduate Medical Education. Sponsoring institution emergency categorization. Accessed October 20, 2022. https://www.acgme.org/covid-19/sponsoring-institution-emergency-categorization/
- Li YM, Galimberti F, Abrouk M, et al. US dermatology resident responses about the COVID-19 pandemic: results from a nationwide survey. South Med J. 2020;113:462-465.
- Newman B, Gallion C. Hurricane Harvey: firsthand perspectives for disaster preparedness in graduate medical education. Acad Med. 2019;94:1267-1269.
- Pero CD, Pou AM, Arriaga MA, et al. Post-Katrina: study in crisis-related program adaptability. Otolaryngol Head Neck Surg. 2008;138:394-397.
- Hattaway R, Singh N, Rais-Bahrami S, et al. Adaptations of dermatology residency programs to changes in medical education amid the COVID-19 pandemic: virtual opportunities and social media. SKIN. 2021;5:94-100.
- Hillier K, Paskaradevan J, Wilkes JK, et al. Disaster plans: resident involvement and well-being during Hurricane Harvey. J Grad Med Educ. 2019;11:129-131.
- Samimi S, Choi J, Rosman IS, et al. Impact of COVID-19 on dermatology residency. Dermatol Clin. 2021;39:609-618.
- Bastola M, Locatis C, Fontelo P. Diagnostic reliability of in-person versus remote dermatology: a meta-analysis. Telemed J E Health. 2021;27:247-250.
- Bashyam AM, Feldman SR. Should patients stop their biologic treatment during the COVID-19 pandemic? J Dermatolog Treat. 2020;31:317-318.
- Davis W. Hurricane Katrina: the challenge to graduate medical education. Ochsner J. 2006;6:39.
- Cefalu CA, Schwartz RS. Salvaging a geriatric medicine academic program in disaster mode—the LSU training program post-Katrina.J Natl Med Assoc. 2007;99:590-596.
- Ayyala R. Lessons from Katrina: a program director’s perspective. Ophthalmology. 2007;114:1425-1426.
- Wiese JG. Leadership in graduate medical education: eleven steps instrumental in recovering residency programs after a disaster. Am J Med Sci. 2008;336:168-173.
- Griffies WS. Post-Katrina stabilization of the LSU/Ochsner Psychiatry Residency Program: caveats for disaster preparedness. Acad Psychiatry. 2009;33:418-422.
- Kearns DG, Chat VS, Uppal S, et al. Applying to dermatology residency during the COVID-19 pandemic. J Am Acad Dermatol. 2020;83:1214-1215.
- Matthews JB, Blair PG, Ellison EC, et al. Checklist framework for surgical education disaster plans. J Am Coll Surg. 2021;233:557-563.
- Litchman GH, Marson JW, Rigel DS. The continuing impact of COVID-19 on dermatology practice: office workflow, economics, and future implications. J Am Acad Dermatol. 2021;84:576-579.
- Accreditation Council for Graduate Medical Education. Sponsoring institution emergency categorization. Accessed October 20, 2022. https://www.acgme.org/covid-19/sponsoring-institution-emergency-categorization/
- Li YM, Galimberti F, Abrouk M, et al. US dermatology resident responses about the COVID-19 pandemic: results from a nationwide survey. South Med J. 2020;113:462-465.
- Newman B, Gallion C. Hurricane Harvey: firsthand perspectives for disaster preparedness in graduate medical education. Acad Med. 2019;94:1267-1269.
- Pero CD, Pou AM, Arriaga MA, et al. Post-Katrina: study in crisis-related program adaptability. Otolaryngol Head Neck Surg. 2008;138:394-397.
- Hattaway R, Singh N, Rais-Bahrami S, et al. Adaptations of dermatology residency programs to changes in medical education amid the COVID-19 pandemic: virtual opportunities and social media. SKIN. 2021;5:94-100.
- Hillier K, Paskaradevan J, Wilkes JK, et al. Disaster plans: resident involvement and well-being during Hurricane Harvey. J Grad Med Educ. 2019;11:129-131.
- Samimi S, Choi J, Rosman IS, et al. Impact of COVID-19 on dermatology residency. Dermatol Clin. 2021;39:609-618.
- Bastola M, Locatis C, Fontelo P. Diagnostic reliability of in-person versus remote dermatology: a meta-analysis. Telemed J E Health. 2021;27:247-250.
- Bashyam AM, Feldman SR. Should patients stop their biologic treatment during the COVID-19 pandemic? J Dermatolog Treat. 2020;31:317-318.
Practice Points
- Dermatology residency programs should prioritize the development of disaster preparedness plans prior to the onset of disasters.
- Comprehensive disaster preparedness addresses many possible disruptions to dermatology resident training and clinic operations, including natural and manmade disasters and threats of widespread infectious disease.
- Safety being paramount, dermatology residency programs may be tasked with maintaining resident wellness, continuing resident education—potentially in unconventional ways—and adapting clinical operations to continue patient care.
Children and COVID: New cases increase for second straight week
New COVID-19 cases rose among U.S. children for the second consecutive week, while hospitals saw signs of renewed activity on the part of SARS-CoV-2.
, when the count fell to its lowest level in more than a year, the American Academy of Pediatrics and the Children’s Hospital Association said in their joint report.
The 7-day average for ED visits with diagnosed COVID was down to just 0.6% of all ED visits for 12- to 15-year-olds as late as Oct. 23 but has moved up to 0.7% since then. Among those aged 16-17 years, the 7-day average was also down to 0.6% for just one day, Oct. 19, but was up to 0.8% as of Nov. 4. So far, though, a similar increase has not yet occurred for ED visits among children aged 0-11 years, the CDC said on its COVID Data Tracker.
The trend is discernible, however, when looking at hospitalizations of children with confirmed COVID. The rate of new admissions of children aged 0-17 years was 0.16 per 100,000 population as late as Oct. 23 but ticked up a notch after that and has been 0.17 per 100,000 since, according to the CDC. As with the ED rate, hospitalizations had been steadily declining since late August.
Vaccine initiation continues to slow
During the week of Oct. 27 to Nov. 2, about 30,000 children under 5 years of age received their initial COVID vaccination. A month earlier (Sept. 29 to Oct. 5), that number was about 40,000. A month before that, about 53,000 children aged 0-5 years received their initial dose, the AAP said in a separate vaccination report based on CDC data.
All of that reduced interest adds up to 7.4% of the age group having received at least one dose and just 3.2% being fully vaccinated as of Nov. 2. Among children aged 5-11 years, the corresponding vaccination rates are 38.9% and 31.8%, while those aged 12-17 years are at 71.3% and 61.1%, the CDC said.
Looking at just the first 20 weeks of the vaccination experience for each age group shows that 1.6 million children under 5 years of age had received at least an initial dose, compared with 8.1 million children aged 5-11 years and 8.1 million children aged 12-15, the AAP said.
New COVID-19 cases rose among U.S. children for the second consecutive week, while hospitals saw signs of renewed activity on the part of SARS-CoV-2.
, when the count fell to its lowest level in more than a year, the American Academy of Pediatrics and the Children’s Hospital Association said in their joint report.
The 7-day average for ED visits with diagnosed COVID was down to just 0.6% of all ED visits for 12- to 15-year-olds as late as Oct. 23 but has moved up to 0.7% since then. Among those aged 16-17 years, the 7-day average was also down to 0.6% for just one day, Oct. 19, but was up to 0.8% as of Nov. 4. So far, though, a similar increase has not yet occurred for ED visits among children aged 0-11 years, the CDC said on its COVID Data Tracker.
The trend is discernible, however, when looking at hospitalizations of children with confirmed COVID. The rate of new admissions of children aged 0-17 years was 0.16 per 100,000 population as late as Oct. 23 but ticked up a notch after that and has been 0.17 per 100,000 since, according to the CDC. As with the ED rate, hospitalizations had been steadily declining since late August.
Vaccine initiation continues to slow
During the week of Oct. 27 to Nov. 2, about 30,000 children under 5 years of age received their initial COVID vaccination. A month earlier (Sept. 29 to Oct. 5), that number was about 40,000. A month before that, about 53,000 children aged 0-5 years received their initial dose, the AAP said in a separate vaccination report based on CDC data.
All of that reduced interest adds up to 7.4% of the age group having received at least one dose and just 3.2% being fully vaccinated as of Nov. 2. Among children aged 5-11 years, the corresponding vaccination rates are 38.9% and 31.8%, while those aged 12-17 years are at 71.3% and 61.1%, the CDC said.
Looking at just the first 20 weeks of the vaccination experience for each age group shows that 1.6 million children under 5 years of age had received at least an initial dose, compared with 8.1 million children aged 5-11 years and 8.1 million children aged 12-15, the AAP said.
New COVID-19 cases rose among U.S. children for the second consecutive week, while hospitals saw signs of renewed activity on the part of SARS-CoV-2.
, when the count fell to its lowest level in more than a year, the American Academy of Pediatrics and the Children’s Hospital Association said in their joint report.
The 7-day average for ED visits with diagnosed COVID was down to just 0.6% of all ED visits for 12- to 15-year-olds as late as Oct. 23 but has moved up to 0.7% since then. Among those aged 16-17 years, the 7-day average was also down to 0.6% for just one day, Oct. 19, but was up to 0.8% as of Nov. 4. So far, though, a similar increase has not yet occurred for ED visits among children aged 0-11 years, the CDC said on its COVID Data Tracker.
The trend is discernible, however, when looking at hospitalizations of children with confirmed COVID. The rate of new admissions of children aged 0-17 years was 0.16 per 100,000 population as late as Oct. 23 but ticked up a notch after that and has been 0.17 per 100,000 since, according to the CDC. As with the ED rate, hospitalizations had been steadily declining since late August.
Vaccine initiation continues to slow
During the week of Oct. 27 to Nov. 2, about 30,000 children under 5 years of age received their initial COVID vaccination. A month earlier (Sept. 29 to Oct. 5), that number was about 40,000. A month before that, about 53,000 children aged 0-5 years received their initial dose, the AAP said in a separate vaccination report based on CDC data.
All of that reduced interest adds up to 7.4% of the age group having received at least one dose and just 3.2% being fully vaccinated as of Nov. 2. Among children aged 5-11 years, the corresponding vaccination rates are 38.9% and 31.8%, while those aged 12-17 years are at 71.3% and 61.1%, the CDC said.
Looking at just the first 20 weeks of the vaccination experience for each age group shows that 1.6 million children under 5 years of age had received at least an initial dose, compared with 8.1 million children aged 5-11 years and 8.1 million children aged 12-15, the AAP said.
The Impact of COVID-19 on Colorectal Cancer Screening Programs
- Siegel RL, Miller KD, Goding Sauer A, et al. Colorectal cancer statistics, 2020. CA Cancer J Clin. 2020;70(3):145-164. doi:10.3322/caac.21601
- Issaka RB, Somsouk M. Colorectal cancer screening and prevention in the COVID-19 era. JAMA Health Forum. 2020;1(5):e200588. doi:10.1001/jamahealthforum.2020.0588
- Balzora S, Issaka RB, Anyane-Yeboa A, Gray DM 2nd, May FP. Impact of COVID-19 on colorectal cancer disparities and the way forward. Gastrointest Endosc. 2020;92(4):946-950. doi:10.1016/j.gie.2020.06.042
- Truman BI, Chang MH, Moonesinghe R. Provisional COVID-19 age-adjusted death rates, by race and ethnicity – United States, 2020–2021. MMWR Morb Mortal Wkly Rep. 2022;71(17):601-605. doi:10.15585/mmwr.mm7117e2
- Czeisler MÉ, Marynak K, Clarke KEN, et al. Delay or avoidance of medical care because of COVID-19-related concerns – United States, June 2020. MMWR Morb Mortal Wkly Rep. 2020;69(36):1250-1257. doi:10.15585/mmwr.mm6936a4
- Inadomi JM, Vijan S, Janz NK, et al. Adherence to colorectal cancer screening: a randomized clinical trial of competing strategies. Arch Intern Med. 2012;172(7):575-582. doi:10.1001/archinternmed.2012.332
- Fedewa SA, Star J, Bandi P, et al. Changes in cancer screening in the US during the COVID-19 pandemic. JAMA Netw Open. 2022;5(6):e2215490. doi:10.1001/jamanetworkopen.2022.15490
- Levin TR, Corley DA, Jensen CD, et al. Effects of organized colorectal cancer screening on cancer incidence and mortality in a large community-based population. Gastroenterology. 2018;155(5):1383-1391.e5. doi:10.1053/j.gastro.2018.07.017
- Doubeni CA, Corley DA, Zhao W, Lau Y, Jensen CD, Levin TR. Association between improved colorectal screening and racial disparities. N Engl J Med. 2022;386(8):796-798. doi:10.1056/NEJMc2112409
- Lee JK, Lam AY, Jensen CD, et al. Impact of the COVID-19 pandemic on fecal immunochemical testing, colonoscopy services, and colorectal neoplasia detection in a large United States community-based population. Gastroenterology. 2022;S0016-5085(22)00503-0. doi:10.1053/j.gastro.2022.05.014
- Issaka RB, Taylor P, Baxi A, Inadomi JM, Ramsey SD, Roth J. Model-based estimation of colorectal cancer screening and outcomes during the COVID-19 pandemic. JAMA Netw Open. 2021;4(4):e216454. doi:10.1001/jamanetworkopen.2021.6454
- Gupta S, Coronado GD, Argenbright K, et al. Mailed fecal immunochemical test outreach for colorectal cancer screening: summary of a Centers for Disease Control and Prevention–sponsored summit. CA Cancer J Clin. 2020;70(4):283-298. doi:10.3322/caac.21615
- Zorzi M, Battagello J, Selby K, et al. Non-compliance with colonoscopy after a positive faecal immunochemical test doubles the risk of dying from colorectal cancer. Gut. 2022;71(3):561-567. doi:10.1136/gutjnl-2020-322192
- Lieberman D, Ladabaum U, Brill JV, et al. Reducing the burden of colorectal cancer: AGA position statements. Gastroenterology. 2022;163(2):520-526. doi:10.1053/j.gastro.2022.05.011
- Bell-Brown A, Chew L, Weiner BJ, et al. Operationalizing a rideshare intervention for colonoscopy completion: barriers, facilitators, and process recommendations. Front Health Serv. 2022;1:799816. doi:10.3389/frhs.2021.799816
- Siegel RL, Miller KD, Goding Sauer A, et al. Colorectal cancer statistics, 2020. CA Cancer J Clin. 2020;70(3):145-164. doi:10.3322/caac.21601
- Issaka RB, Somsouk M. Colorectal cancer screening and prevention in the COVID-19 era. JAMA Health Forum. 2020;1(5):e200588. doi:10.1001/jamahealthforum.2020.0588
- Balzora S, Issaka RB, Anyane-Yeboa A, Gray DM 2nd, May FP. Impact of COVID-19 on colorectal cancer disparities and the way forward. Gastrointest Endosc. 2020;92(4):946-950. doi:10.1016/j.gie.2020.06.042
- Truman BI, Chang MH, Moonesinghe R. Provisional COVID-19 age-adjusted death rates, by race and ethnicity – United States, 2020–2021. MMWR Morb Mortal Wkly Rep. 2022;71(17):601-605. doi:10.15585/mmwr.mm7117e2
- Czeisler MÉ, Marynak K, Clarke KEN, et al. Delay or avoidance of medical care because of COVID-19-related concerns – United States, June 2020. MMWR Morb Mortal Wkly Rep. 2020;69(36):1250-1257. doi:10.15585/mmwr.mm6936a4
- Inadomi JM, Vijan S, Janz NK, et al. Adherence to colorectal cancer screening: a randomized clinical trial of competing strategies. Arch Intern Med. 2012;172(7):575-582. doi:10.1001/archinternmed.2012.332
- Fedewa SA, Star J, Bandi P, et al. Changes in cancer screening in the US during the COVID-19 pandemic. JAMA Netw Open. 2022;5(6):e2215490. doi:10.1001/jamanetworkopen.2022.15490
- Levin TR, Corley DA, Jensen CD, et al. Effects of organized colorectal cancer screening on cancer incidence and mortality in a large community-based population. Gastroenterology. 2018;155(5):1383-1391.e5. doi:10.1053/j.gastro.2018.07.017
- Doubeni CA, Corley DA, Zhao W, Lau Y, Jensen CD, Levin TR. Association between improved colorectal screening and racial disparities. N Engl J Med. 2022;386(8):796-798. doi:10.1056/NEJMc2112409
- Lee JK, Lam AY, Jensen CD, et al. Impact of the COVID-19 pandemic on fecal immunochemical testing, colonoscopy services, and colorectal neoplasia detection in a large United States community-based population. Gastroenterology. 2022;S0016-5085(22)00503-0. doi:10.1053/j.gastro.2022.05.014
- Issaka RB, Taylor P, Baxi A, Inadomi JM, Ramsey SD, Roth J. Model-based estimation of colorectal cancer screening and outcomes during the COVID-19 pandemic. JAMA Netw Open. 2021;4(4):e216454. doi:10.1001/jamanetworkopen.2021.6454
- Gupta S, Coronado GD, Argenbright K, et al. Mailed fecal immunochemical test outreach for colorectal cancer screening: summary of a Centers for Disease Control and Prevention–sponsored summit. CA Cancer J Clin. 2020;70(4):283-298. doi:10.3322/caac.21615
- Zorzi M, Battagello J, Selby K, et al. Non-compliance with colonoscopy after a positive faecal immunochemical test doubles the risk of dying from colorectal cancer. Gut. 2022;71(3):561-567. doi:10.1136/gutjnl-2020-322192
- Lieberman D, Ladabaum U, Brill JV, et al. Reducing the burden of colorectal cancer: AGA position statements. Gastroenterology. 2022;163(2):520-526. doi:10.1053/j.gastro.2022.05.011
- Bell-Brown A, Chew L, Weiner BJ, et al. Operationalizing a rideshare intervention for colonoscopy completion: barriers, facilitators, and process recommendations. Front Health Serv. 2022;1:799816. doi:10.3389/frhs.2021.799816
- Siegel RL, Miller KD, Goding Sauer A, et al. Colorectal cancer statistics, 2020. CA Cancer J Clin. 2020;70(3):145-164. doi:10.3322/caac.21601
- Issaka RB, Somsouk M. Colorectal cancer screening and prevention in the COVID-19 era. JAMA Health Forum. 2020;1(5):e200588. doi:10.1001/jamahealthforum.2020.0588
- Balzora S, Issaka RB, Anyane-Yeboa A, Gray DM 2nd, May FP. Impact of COVID-19 on colorectal cancer disparities and the way forward. Gastrointest Endosc. 2020;92(4):946-950. doi:10.1016/j.gie.2020.06.042
- Truman BI, Chang MH, Moonesinghe R. Provisional COVID-19 age-adjusted death rates, by race and ethnicity – United States, 2020–2021. MMWR Morb Mortal Wkly Rep. 2022;71(17):601-605. doi:10.15585/mmwr.mm7117e2
- Czeisler MÉ, Marynak K, Clarke KEN, et al. Delay or avoidance of medical care because of COVID-19-related concerns – United States, June 2020. MMWR Morb Mortal Wkly Rep. 2020;69(36):1250-1257. doi:10.15585/mmwr.mm6936a4
- Inadomi JM, Vijan S, Janz NK, et al. Adherence to colorectal cancer screening: a randomized clinical trial of competing strategies. Arch Intern Med. 2012;172(7):575-582. doi:10.1001/archinternmed.2012.332
- Fedewa SA, Star J, Bandi P, et al. Changes in cancer screening in the US during the COVID-19 pandemic. JAMA Netw Open. 2022;5(6):e2215490. doi:10.1001/jamanetworkopen.2022.15490
- Levin TR, Corley DA, Jensen CD, et al. Effects of organized colorectal cancer screening on cancer incidence and mortality in a large community-based population. Gastroenterology. 2018;155(5):1383-1391.e5. doi:10.1053/j.gastro.2018.07.017
- Doubeni CA, Corley DA, Zhao W, Lau Y, Jensen CD, Levin TR. Association between improved colorectal screening and racial disparities. N Engl J Med. 2022;386(8):796-798. doi:10.1056/NEJMc2112409
- Lee JK, Lam AY, Jensen CD, et al. Impact of the COVID-19 pandemic on fecal immunochemical testing, colonoscopy services, and colorectal neoplasia detection in a large United States community-based population. Gastroenterology. 2022;S0016-5085(22)00503-0. doi:10.1053/j.gastro.2022.05.014
- Issaka RB, Taylor P, Baxi A, Inadomi JM, Ramsey SD, Roth J. Model-based estimation of colorectal cancer screening and outcomes during the COVID-19 pandemic. JAMA Netw Open. 2021;4(4):e216454. doi:10.1001/jamanetworkopen.2021.6454
- Gupta S, Coronado GD, Argenbright K, et al. Mailed fecal immunochemical test outreach for colorectal cancer screening: summary of a Centers for Disease Control and Prevention–sponsored summit. CA Cancer J Clin. 2020;70(4):283-298. doi:10.3322/caac.21615
- Zorzi M, Battagello J, Selby K, et al. Non-compliance with colonoscopy after a positive faecal immunochemical test doubles the risk of dying from colorectal cancer. Gut. 2022;71(3):561-567. doi:10.1136/gutjnl-2020-322192
- Lieberman D, Ladabaum U, Brill JV, et al. Reducing the burden of colorectal cancer: AGA position statements. Gastroenterology. 2022;163(2):520-526. doi:10.1053/j.gastro.2022.05.011
- Bell-Brown A, Chew L, Weiner BJ, et al. Operationalizing a rideshare intervention for colonoscopy completion: barriers, facilitators, and process recommendations. Front Health Serv. 2022;1:799816. doi:10.3389/frhs.2021.799816
Post-COVID-19 Effects
- Centers for Disease Control and Prevention. COVID data tracker. Updated August 19, 2022. Accessed August 22, 2022. https://covid.cdc.gov/covid-data-tracker
- Nalbandian A, Sehgal K, Gupta A, et al. Post-acute COVID-19 syndrome. Nat Med. 2021;27(4):601-615. doi:10.1038/s41591-021-01283-z
- Centers for Disease Control and Prevention. Long COVID or post-COVID conditions. Updated May 5, 2022. Accessed June 6, 2022. https://www.cdc.gov/coronavirus/2019-ncov/long-termeffects/index.html
- Ghazanfar H, Kandhi S, Shin D, et al. Impact of COVID-19 on the gastrointestinal tract: a clinical review. Cureus. 2022;14(3):e23333. doi:10.7759/cureus.23333
- Khan SM, Shilen A, Heslin KM, et al. SARS-CoV-2 infection and subsequent changes in the menstrual cycle among participants in the Arizona CoVHORT study. Am J Obstet Gynecol. 2022;226(2):270-273. doi:10.1016/j.ajog.2021.09.016
- Chopra V, Flanders SA, O’Malley M, Malani AN, Prescott HC. Sixty-day outcomes among patients hospitalized with COVID-19. Ann Intern Med. 2021;174(4):576-578. doi:10.7326/M20-5661
- Jiang DH, McCoy RG. Planning for the post-COVID syndrome: how payers can mitigate long-term complications of the pandemic. J Gen Intern Med. 2020;35(10):3036-3039. doi:10.1007/s11606-020-06042-3
- Centers for Disease Control and Prevention. COVID data tracker. Updated August 19, 2022. Accessed August 22, 2022. https://covid.cdc.gov/covid-data-tracker
- Nalbandian A, Sehgal K, Gupta A, et al. Post-acute COVID-19 syndrome. Nat Med. 2021;27(4):601-615. doi:10.1038/s41591-021-01283-z
- Centers for Disease Control and Prevention. Long COVID or post-COVID conditions. Updated May 5, 2022. Accessed June 6, 2022. https://www.cdc.gov/coronavirus/2019-ncov/long-termeffects/index.html
- Ghazanfar H, Kandhi S, Shin D, et al. Impact of COVID-19 on the gastrointestinal tract: a clinical review. Cureus. 2022;14(3):e23333. doi:10.7759/cureus.23333
- Khan SM, Shilen A, Heslin KM, et al. SARS-CoV-2 infection and subsequent changes in the menstrual cycle among participants in the Arizona CoVHORT study. Am J Obstet Gynecol. 2022;226(2):270-273. doi:10.1016/j.ajog.2021.09.016
- Chopra V, Flanders SA, O’Malley M, Malani AN, Prescott HC. Sixty-day outcomes among patients hospitalized with COVID-19. Ann Intern Med. 2021;174(4):576-578. doi:10.7326/M20-5661
- Jiang DH, McCoy RG. Planning for the post-COVID syndrome: how payers can mitigate long-term complications of the pandemic. J Gen Intern Med. 2020;35(10):3036-3039. doi:10.1007/s11606-020-06042-3
- Centers for Disease Control and Prevention. COVID data tracker. Updated August 19, 2022. Accessed August 22, 2022. https://covid.cdc.gov/covid-data-tracker
- Nalbandian A, Sehgal K, Gupta A, et al. Post-acute COVID-19 syndrome. Nat Med. 2021;27(4):601-615. doi:10.1038/s41591-021-01283-z
- Centers for Disease Control and Prevention. Long COVID or post-COVID conditions. Updated May 5, 2022. Accessed June 6, 2022. https://www.cdc.gov/coronavirus/2019-ncov/long-termeffects/index.html
- Ghazanfar H, Kandhi S, Shin D, et al. Impact of COVID-19 on the gastrointestinal tract: a clinical review. Cureus. 2022;14(3):e23333. doi:10.7759/cureus.23333
- Khan SM, Shilen A, Heslin KM, et al. SARS-CoV-2 infection and subsequent changes in the menstrual cycle among participants in the Arizona CoVHORT study. Am J Obstet Gynecol. 2022;226(2):270-273. doi:10.1016/j.ajog.2021.09.016
- Chopra V, Flanders SA, O’Malley M, Malani AN, Prescott HC. Sixty-day outcomes among patients hospitalized with COVID-19. Ann Intern Med. 2021;174(4):576-578. doi:10.7326/M20-5661
- Jiang DH, McCoy RG. Planning for the post-COVID syndrome: how payers can mitigate long-term complications of the pandemic. J Gen Intern Med. 2020;35(10):3036-3039. doi:10.1007/s11606-020-06042-3
CDC warns of early uptick in respiratory disease
The Centers for Disease Control and Prevention is warning of an early surge in respiratory disease caused by multiple viruses. As influenza viruses, respiratory syncytial virus (RSV), SARS-CoV-2, and rhinovirus/enterovirus simultaneously circulate, the agency cautioned that this confluence of viral activity could strain the health care system, according to a CDC Health Network Alert advisory issued Nov. 4.
“This early increase in disease incidence highlights the importance of optimizing respiratory virus prevention and treatment measures, including prompt vaccination and antiviral treatment,” the alert stated.
The CDC reports that RSV activity is increasing nationally, but in some areas – such as the South and Mountain West – cases appear to be trending downward.
Influenza cases continue to climb, with the virus activity being the highest in the South, Mid-Atlantic, and the south-central West Coast, according to CDC data. “In fact, we’re seeing the highest influenza hospitalization rates going back a decade,” said José Romero, MD, director of the CDC’s National Center for Immunization and Respiratory Diseases, during a press briefing. The agency estimates that there have been 1.6 million illnesses, 13,000 hospitalizations, and 730 deaths from the flu so far this season. As of Nov. 4, there have been two pediatric deaths.
COVID-19 cases appear to have plateaued in the past three weeks, Dr. Romero said; however, the CDC expects that there will be “high-level circulation of SARS-CoV-2 this fall and winter,” the health alert stated.
The CDC advised that all eligible individuals aged 6-months or older should be vaccinated against COVID-19 and influenza. To protect against RSV-hospitalization, high-risk children should receive the monoclonal antibody drug palivizumab (Synagis). High-risk children include infants born before 29 weeks, children younger than age 2 with chronic lung disease or hemodynamically significant congenital heart disease, and children with suppressed immune systems or neuromuscular disorders.
Any patient with confirmed or suspected flu who is hospitalized, at higher risk for influenza complications, or who has a severe or progressive illness should be treated as early as possible with antivirals, such as oral oseltamivir (Tamiflu).
Patients with confirmed SARS-CoV-2 infection with increased risk of complications should also be treated with antivirals, such as nirmatrelvir and ritonavir (Paxlovid) or remdesivir (Veklury).
Patients should also be reminded to wash their hands frequently, cover coughs and sneezes, stay home when sick, and avoid close contact with people who are sick, the CDC advised.
“There’s no doubt that we will face some challenges this winter,” said Dawn O’Connell, HHS Assistant Secretary for Preparedness and Response, “but it’s important to remember that RSV and flu are not new, and we have safe and effective vaccines for COVID-19 and the flu.”
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention is warning of an early surge in respiratory disease caused by multiple viruses. As influenza viruses, respiratory syncytial virus (RSV), SARS-CoV-2, and rhinovirus/enterovirus simultaneously circulate, the agency cautioned that this confluence of viral activity could strain the health care system, according to a CDC Health Network Alert advisory issued Nov. 4.
“This early increase in disease incidence highlights the importance of optimizing respiratory virus prevention and treatment measures, including prompt vaccination and antiviral treatment,” the alert stated.
The CDC reports that RSV activity is increasing nationally, but in some areas – such as the South and Mountain West – cases appear to be trending downward.
Influenza cases continue to climb, with the virus activity being the highest in the South, Mid-Atlantic, and the south-central West Coast, according to CDC data. “In fact, we’re seeing the highest influenza hospitalization rates going back a decade,” said José Romero, MD, director of the CDC’s National Center for Immunization and Respiratory Diseases, during a press briefing. The agency estimates that there have been 1.6 million illnesses, 13,000 hospitalizations, and 730 deaths from the flu so far this season. As of Nov. 4, there have been two pediatric deaths.
COVID-19 cases appear to have plateaued in the past three weeks, Dr. Romero said; however, the CDC expects that there will be “high-level circulation of SARS-CoV-2 this fall and winter,” the health alert stated.
The CDC advised that all eligible individuals aged 6-months or older should be vaccinated against COVID-19 and influenza. To protect against RSV-hospitalization, high-risk children should receive the monoclonal antibody drug palivizumab (Synagis). High-risk children include infants born before 29 weeks, children younger than age 2 with chronic lung disease or hemodynamically significant congenital heart disease, and children with suppressed immune systems or neuromuscular disorders.
Any patient with confirmed or suspected flu who is hospitalized, at higher risk for influenza complications, or who has a severe or progressive illness should be treated as early as possible with antivirals, such as oral oseltamivir (Tamiflu).
Patients with confirmed SARS-CoV-2 infection with increased risk of complications should also be treated with antivirals, such as nirmatrelvir and ritonavir (Paxlovid) or remdesivir (Veklury).
Patients should also be reminded to wash their hands frequently, cover coughs and sneezes, stay home when sick, and avoid close contact with people who are sick, the CDC advised.
“There’s no doubt that we will face some challenges this winter,” said Dawn O’Connell, HHS Assistant Secretary for Preparedness and Response, “but it’s important to remember that RSV and flu are not new, and we have safe and effective vaccines for COVID-19 and the flu.”
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention is warning of an early surge in respiratory disease caused by multiple viruses. As influenza viruses, respiratory syncytial virus (RSV), SARS-CoV-2, and rhinovirus/enterovirus simultaneously circulate, the agency cautioned that this confluence of viral activity could strain the health care system, according to a CDC Health Network Alert advisory issued Nov. 4.
“This early increase in disease incidence highlights the importance of optimizing respiratory virus prevention and treatment measures, including prompt vaccination and antiviral treatment,” the alert stated.
The CDC reports that RSV activity is increasing nationally, but in some areas – such as the South and Mountain West – cases appear to be trending downward.
Influenza cases continue to climb, with the virus activity being the highest in the South, Mid-Atlantic, and the south-central West Coast, according to CDC data. “In fact, we’re seeing the highest influenza hospitalization rates going back a decade,” said José Romero, MD, director of the CDC’s National Center for Immunization and Respiratory Diseases, during a press briefing. The agency estimates that there have been 1.6 million illnesses, 13,000 hospitalizations, and 730 deaths from the flu so far this season. As of Nov. 4, there have been two pediatric deaths.
COVID-19 cases appear to have plateaued in the past three weeks, Dr. Romero said; however, the CDC expects that there will be “high-level circulation of SARS-CoV-2 this fall and winter,” the health alert stated.
The CDC advised that all eligible individuals aged 6-months or older should be vaccinated against COVID-19 and influenza. To protect against RSV-hospitalization, high-risk children should receive the monoclonal antibody drug palivizumab (Synagis). High-risk children include infants born before 29 weeks, children younger than age 2 with chronic lung disease or hemodynamically significant congenital heart disease, and children with suppressed immune systems or neuromuscular disorders.
Any patient with confirmed or suspected flu who is hospitalized, at higher risk for influenza complications, or who has a severe or progressive illness should be treated as early as possible with antivirals, such as oral oseltamivir (Tamiflu).
Patients with confirmed SARS-CoV-2 infection with increased risk of complications should also be treated with antivirals, such as nirmatrelvir and ritonavir (Paxlovid) or remdesivir (Veklury).
Patients should also be reminded to wash their hands frequently, cover coughs and sneezes, stay home when sick, and avoid close contact with people who are sick, the CDC advised.
“There’s no doubt that we will face some challenges this winter,” said Dawn O’Connell, HHS Assistant Secretary for Preparedness and Response, “but it’s important to remember that RSV and flu are not new, and we have safe and effective vaccines for COVID-19 and the flu.”
A version of this article first appeared on Medscape.com.