User login
Doctor, PA, face lawsuit after patient dies of COVID-19
, claiming the health providers failed to properly test the man and rendered improper care that led to his demise.
Shirley Dimoh of Memphis alleges her husband Peter Dimoh, 66, received inadequate care when he presented with COVID-19 symptoms to May Medical Group in Munford, Tenn., on November 19, 2020. Physician assistant Robert Moody ordered a COVID-19 blood test for Mr. Dimoh, rather than a nasal swab test, which was sent to an Atlanta lab for analysis, according to the lawsuit.
By the time the Dimohs were informed of the positive result on November 23, Mr. Dimoh was seriously ill and showed signs of severe infection, the claim alleges. Family physician David Krapf, DO, then prescribed methocarbamol, a muscle relaxant, which the complaint claims exacerbated Mr. Dimoh’s infection, caused serious adverse reactions, and led to his death on December 16, 2020.
“Mr. Dimoh had multiple comorbid conditions and he had a positive test, he should have been referred to a specialist at a higher level of care,” said Duncan E. Ragsdale, a Memphis-based attorney representing Shirley Dimoh. “This is not something for a family physician or a physician assistant to be attempting to handle. It’s my belief this was a preventable death.”
Shirley Dimoh filed a lawsuit against May Medical Group, Moody, Krapf, and several others on June 23, 2021, in the Circuit Court of Tennessee, 13th Judicial District. She accuses the group of negligence, failure to refer, and failure to supervise, among other claims. She is requesting $5 million in damages.
May Medical Group did not return phone or email messages seeking comment for this story. The group had not issued a reply to the lawsuit as of this article’s deadline. An attorney for the practice is not yet listed in court records.
Wife had COVID-19 first, treated by same PA
Shirley Dimoh was the first of the couple to contract COVID-19. She visited May Medical Group with COVID-19 symptoms on November 9, according to the lawsuit. The same physician assistant allegedly prescribed antibiotics and sent Shirley Dimoh to the health department for a rapid nasal swab test. After a positive test resulted, she was prescribed more antibiotics, steroids, and other medications, according to the claim. She recovered without complications.
The plaintiff alleges the medical practice did not warn Peter Dimoh of the risk of contracting COVID-19 from contact with Shirley Dimoh or offer prophylactics against infection. Peter Dimoh had underlying conditions that included diabetes and kidney failure, according to Mr. Ragsdale.
It’s unclear why Peter Dimoh may have been prescribed methocarbamol after his positive COVID-19 test on November 23. The same day, Shirley Dimoh went to the pharmacy to pick up her husband’s prescription because he was too sick to go himself, the complaint states. Mr. Dimoh took the medication three times a day as prescribed. The methocarbamol was “unreasonably dangerous” for Mr. Dimoh at the time and caused an injury he would have not incurred otherwise, the suit claims.
On November 28, Mr. Dimoh collapsed and was taken by ambulance to Methodist Hospital in Memphis. He was treated for COVID-19 as well as bacterial pneumonia, severe sepsis, and sytemic inflammatory response syndrome, according to the complaint. He died December 16 from respiratory and heart failure.
“What’s interesting about this case is that you’ve got a good example and a bad example together,” Mr. Ragsdale said. “In other words, the good example is they treated her appropriately, she recovered. They didn’t treat him at all, he died. I don’t know how you could have a better counter position established by the same practice.”
Family has sued for malpractice before
Before his death, Peter Dimoh was the plaintiff in another medical negligence suit. In 2013, he sued the Center for Oral and Facial Surgery of Memphis and two oral surgeons for allegedly operating on him improperly and causing an adverse outcome.
Mr. Dimoh underwent oral surgery on February 8, 2012, at the center. The day of his operation, Mr. Dimoh’s A1c and glucose levels were grossly elevated, according to the claim, which was also represented by Mr. Ragsdale. Surgeons concluded, incorrectly, that Mr. Dimoh’s diabetes was under control, the complaint alleges, failed to order preoperative antibiotics, operated on him, and caused a bacterial infection at the operative site. The infection allegedly resulted in osteomyelitis of Mr. Dimoh’s left mandible with a pathologic fracture.
The complaint also alleges that although Mr. Dimoh signed a consent form for the surgery, the defendants failed to obtain his consent because they were unaware his diabetes was not under control and they did not explain the risks of surgery while in such a condition. The suit alleges lack of informed consent, negligence, and gross negligence by the practice and requests $2 million in damages.
Court documents show the suit was voluntarily withdrawn in 2018 without prejudice and reissued in 2019. In January 2021, the court was given notice of Mr. Dimoh’s death, and a motion was made to substitute another plaintiff, according to Shelby County court records.
An attorney for the Center for Oral and Facial Surgery of Memphis did not return a message seeking comment.
A version of this article first appeared on Medscape.com.
, claiming the health providers failed to properly test the man and rendered improper care that led to his demise.
Shirley Dimoh of Memphis alleges her husband Peter Dimoh, 66, received inadequate care when he presented with COVID-19 symptoms to May Medical Group in Munford, Tenn., on November 19, 2020. Physician assistant Robert Moody ordered a COVID-19 blood test for Mr. Dimoh, rather than a nasal swab test, which was sent to an Atlanta lab for analysis, according to the lawsuit.
By the time the Dimohs were informed of the positive result on November 23, Mr. Dimoh was seriously ill and showed signs of severe infection, the claim alleges. Family physician David Krapf, DO, then prescribed methocarbamol, a muscle relaxant, which the complaint claims exacerbated Mr. Dimoh’s infection, caused serious adverse reactions, and led to his death on December 16, 2020.
“Mr. Dimoh had multiple comorbid conditions and he had a positive test, he should have been referred to a specialist at a higher level of care,” said Duncan E. Ragsdale, a Memphis-based attorney representing Shirley Dimoh. “This is not something for a family physician or a physician assistant to be attempting to handle. It’s my belief this was a preventable death.”
Shirley Dimoh filed a lawsuit against May Medical Group, Moody, Krapf, and several others on June 23, 2021, in the Circuit Court of Tennessee, 13th Judicial District. She accuses the group of negligence, failure to refer, and failure to supervise, among other claims. She is requesting $5 million in damages.
May Medical Group did not return phone or email messages seeking comment for this story. The group had not issued a reply to the lawsuit as of this article’s deadline. An attorney for the practice is not yet listed in court records.
Wife had COVID-19 first, treated by same PA
Shirley Dimoh was the first of the couple to contract COVID-19. She visited May Medical Group with COVID-19 symptoms on November 9, according to the lawsuit. The same physician assistant allegedly prescribed antibiotics and sent Shirley Dimoh to the health department for a rapid nasal swab test. After a positive test resulted, she was prescribed more antibiotics, steroids, and other medications, according to the claim. She recovered without complications.
The plaintiff alleges the medical practice did not warn Peter Dimoh of the risk of contracting COVID-19 from contact with Shirley Dimoh or offer prophylactics against infection. Peter Dimoh had underlying conditions that included diabetes and kidney failure, according to Mr. Ragsdale.
It’s unclear why Peter Dimoh may have been prescribed methocarbamol after his positive COVID-19 test on November 23. The same day, Shirley Dimoh went to the pharmacy to pick up her husband’s prescription because he was too sick to go himself, the complaint states. Mr. Dimoh took the medication three times a day as prescribed. The methocarbamol was “unreasonably dangerous” for Mr. Dimoh at the time and caused an injury he would have not incurred otherwise, the suit claims.
On November 28, Mr. Dimoh collapsed and was taken by ambulance to Methodist Hospital in Memphis. He was treated for COVID-19 as well as bacterial pneumonia, severe sepsis, and sytemic inflammatory response syndrome, according to the complaint. He died December 16 from respiratory and heart failure.
“What’s interesting about this case is that you’ve got a good example and a bad example together,” Mr. Ragsdale said. “In other words, the good example is they treated her appropriately, she recovered. They didn’t treat him at all, he died. I don’t know how you could have a better counter position established by the same practice.”
Family has sued for malpractice before
Before his death, Peter Dimoh was the plaintiff in another medical negligence suit. In 2013, he sued the Center for Oral and Facial Surgery of Memphis and two oral surgeons for allegedly operating on him improperly and causing an adverse outcome.
Mr. Dimoh underwent oral surgery on February 8, 2012, at the center. The day of his operation, Mr. Dimoh’s A1c and glucose levels were grossly elevated, according to the claim, which was also represented by Mr. Ragsdale. Surgeons concluded, incorrectly, that Mr. Dimoh’s diabetes was under control, the complaint alleges, failed to order preoperative antibiotics, operated on him, and caused a bacterial infection at the operative site. The infection allegedly resulted in osteomyelitis of Mr. Dimoh’s left mandible with a pathologic fracture.
The complaint also alleges that although Mr. Dimoh signed a consent form for the surgery, the defendants failed to obtain his consent because they were unaware his diabetes was not under control and they did not explain the risks of surgery while in such a condition. The suit alleges lack of informed consent, negligence, and gross negligence by the practice and requests $2 million in damages.
Court documents show the suit was voluntarily withdrawn in 2018 without prejudice and reissued in 2019. In January 2021, the court was given notice of Mr. Dimoh’s death, and a motion was made to substitute another plaintiff, according to Shelby County court records.
An attorney for the Center for Oral and Facial Surgery of Memphis did not return a message seeking comment.
A version of this article first appeared on Medscape.com.
, claiming the health providers failed to properly test the man and rendered improper care that led to his demise.
Shirley Dimoh of Memphis alleges her husband Peter Dimoh, 66, received inadequate care when he presented with COVID-19 symptoms to May Medical Group in Munford, Tenn., on November 19, 2020. Physician assistant Robert Moody ordered a COVID-19 blood test for Mr. Dimoh, rather than a nasal swab test, which was sent to an Atlanta lab for analysis, according to the lawsuit.
By the time the Dimohs were informed of the positive result on November 23, Mr. Dimoh was seriously ill and showed signs of severe infection, the claim alleges. Family physician David Krapf, DO, then prescribed methocarbamol, a muscle relaxant, which the complaint claims exacerbated Mr. Dimoh’s infection, caused serious adverse reactions, and led to his death on December 16, 2020.
“Mr. Dimoh had multiple comorbid conditions and he had a positive test, he should have been referred to a specialist at a higher level of care,” said Duncan E. Ragsdale, a Memphis-based attorney representing Shirley Dimoh. “This is not something for a family physician or a physician assistant to be attempting to handle. It’s my belief this was a preventable death.”
Shirley Dimoh filed a lawsuit against May Medical Group, Moody, Krapf, and several others on June 23, 2021, in the Circuit Court of Tennessee, 13th Judicial District. She accuses the group of negligence, failure to refer, and failure to supervise, among other claims. She is requesting $5 million in damages.
May Medical Group did not return phone or email messages seeking comment for this story. The group had not issued a reply to the lawsuit as of this article’s deadline. An attorney for the practice is not yet listed in court records.
Wife had COVID-19 first, treated by same PA
Shirley Dimoh was the first of the couple to contract COVID-19. She visited May Medical Group with COVID-19 symptoms on November 9, according to the lawsuit. The same physician assistant allegedly prescribed antibiotics and sent Shirley Dimoh to the health department for a rapid nasal swab test. After a positive test resulted, she was prescribed more antibiotics, steroids, and other medications, according to the claim. She recovered without complications.
The plaintiff alleges the medical practice did not warn Peter Dimoh of the risk of contracting COVID-19 from contact with Shirley Dimoh or offer prophylactics against infection. Peter Dimoh had underlying conditions that included diabetes and kidney failure, according to Mr. Ragsdale.
It’s unclear why Peter Dimoh may have been prescribed methocarbamol after his positive COVID-19 test on November 23. The same day, Shirley Dimoh went to the pharmacy to pick up her husband’s prescription because he was too sick to go himself, the complaint states. Mr. Dimoh took the medication three times a day as prescribed. The methocarbamol was “unreasonably dangerous” for Mr. Dimoh at the time and caused an injury he would have not incurred otherwise, the suit claims.
On November 28, Mr. Dimoh collapsed and was taken by ambulance to Methodist Hospital in Memphis. He was treated for COVID-19 as well as bacterial pneumonia, severe sepsis, and sytemic inflammatory response syndrome, according to the complaint. He died December 16 from respiratory and heart failure.
“What’s interesting about this case is that you’ve got a good example and a bad example together,” Mr. Ragsdale said. “In other words, the good example is they treated her appropriately, she recovered. They didn’t treat him at all, he died. I don’t know how you could have a better counter position established by the same practice.”
Family has sued for malpractice before
Before his death, Peter Dimoh was the plaintiff in another medical negligence suit. In 2013, he sued the Center for Oral and Facial Surgery of Memphis and two oral surgeons for allegedly operating on him improperly and causing an adverse outcome.
Mr. Dimoh underwent oral surgery on February 8, 2012, at the center. The day of his operation, Mr. Dimoh’s A1c and glucose levels were grossly elevated, according to the claim, which was also represented by Mr. Ragsdale. Surgeons concluded, incorrectly, that Mr. Dimoh’s diabetes was under control, the complaint alleges, failed to order preoperative antibiotics, operated on him, and caused a bacterial infection at the operative site. The infection allegedly resulted in osteomyelitis of Mr. Dimoh’s left mandible with a pathologic fracture.
The complaint also alleges that although Mr. Dimoh signed a consent form for the surgery, the defendants failed to obtain his consent because they were unaware his diabetes was not under control and they did not explain the risks of surgery while in such a condition. The suit alleges lack of informed consent, negligence, and gross negligence by the practice and requests $2 million in damages.
Court documents show the suit was voluntarily withdrawn in 2018 without prejudice and reissued in 2019. In January 2021, the court was given notice of Mr. Dimoh’s death, and a motion was made to substitute another plaintiff, according to Shelby County court records.
An attorney for the Center for Oral and Facial Surgery of Memphis did not return a message seeking comment.
A version of this article first appeared on Medscape.com.
As common respiratory viruses resurface, children are at serious risk
Younger children may be vulnerable to the reemergence of common respiratory viruses such as influenza and respiratory syncytial virus (RSV) as COVID-19 restrictions wane, experts say. The impact could be detrimental.
The COVID-19 pandemic and the implementation of preventative measures such as social distancing, travel restrictions, mask use, and shelter in place, reduced the transmission of respiratory viruses, according to the Centers for Disease Control and Prevention. However, because older infants and toddlers have not been exposed to these bugs during the pandemic, they are vulnerable to suffering severe viral infections.
“[We’ve] been in the honeymoon for 18 months,” said Christopher J. Harrison, MD, professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics in Kansas City, Mo. “We are going to be coming out of the honeymoon and the children who didn’t get sick are going to start packing 2 years’ worth of infections into the next 9 months so there’s going to be twice as many as would be normal.”
The CDC issued a health advisory in June for parts of the southern United States, such as Texas, the Carolinas, and Oklahoma, encouraging broader testing for RSV – a virus that usually causes mild, cold-like symptoms and is the most common cause of bronchiolitis and pneumonia in children – among those who test negative for COVID-19. Virtually all children get an RSV infection by the time they are 2 years old, according to the CDC.
In previous years, RSV usually spread during the fall and spring seasons and usually peaked late December to mid-February. However, there’s been an offseason spike in the common illness this year, with nearly 2,000 confirmed cases each week of July.
Richard J. Webby, PhD, of the infectious diseases department at St. Jude Children’s Research Hospital, Memphis, Tenn., said that although RSV transmits more easily during the winter, the virus is able to thrive during this summer because many children have limited immunity and are more vulnerable to catching the virus than before. Population immunity normally limits a virus to circulating under its most favorable conditions, which is usually the winter. However, because there are a few more “susceptible hosts,” it gives the virus the ability to spread during a time when it typically wouldn’t be able to.
“Now we have a wider range of susceptible kids because they haven’t had that exposure over the past 18 months,” said Dr. Webby, who is on the World Health Organization’s Influenza Vaccine Composition Advisory Team. “It gives the virus more chances to transmit during conditions that are less favorable.”
Dr. Harrison said that, if children continue to take preventative measures such as wearing masks and sanitizing, they can delay catching the RSV – which can be severe in infants and young children – until they’re older and symptoms won’t be as severe.
“The swelling that these viruses cause in the trachea and the bronchial tubes is much bigger in proportion to the overall size of the tubes, so it takes less swelling to clog up the trachea or bronchial tube for the 9-month-old than it does of a 9-year-old,” Dr. Harrison said. “So if a 9-year-old was to get RSV, they’re not going to have nearly the same amount symptoms as the 9-month-old.
Dr. Harrison said delaying RSV in children was never an option before because it’s a virus that’s almost impossible to avoid.
“Hopefully, the mask means that if you get exposed, instead of getting a million virus particles from your classmate or your playmate, you may only get a couple thousand,” Dr. Harrison explained. “And maybe that’s enough that you can fight it off or it may be small enough that you get a mild infection instead of a severe infection.”
A summer surge of RSV has also occurred in Australia. A study published in Clinical Infectious Diseases found that Western Australia saw a 98% reduction in RSV cases. This suggests that COVID-19 restrictions also delayed the RSV season.
Dr. Webby said the lax in penetrative measures against COVID-19 may also affect this upcoming flu season. Usually, around 10%-30% of the population gets infected with the flu each year, but that hasn’t happened the past couple of seasons, he said.
“There might be slightly less overall immunity to these viruses,” Dr. Webby said. “When these viruses do come back, there’s a little bit more room for them to take off.”
Although a severe influenza season rebound this winter is a possibility, Australia continues to experience a historically low flu season. Dr. Harrison, who said the northern hemisphere looks at what’s happening in Australia and the rest of the “southern half of the world because their influenza season is during our summer,” hopes this is an indication that the northern hemisphere will also experience a mild season.
However, there’s no indication of how this upcoming flu season will hit the United States and there isn’t any guidance on what could happen because these historically low levels of respiratory viruses have never happened before, Dr. Webby explained.
He said that, if COVID-19’s delta variant continues to circulate during the fall and winter seasons, it will keep other viruses at low levels. This is because there is rarely a peak of activity of different viruses at the same time.
“When you get infected with the virus, your body’s immune response has this nonspecific reaction that protects you from anything else for a short period of time,” Dr. Webby explained. “When you get a lot of one virus circulating, it’s really hard for these other viruses to get into that population and sort of set off an epidemic of their own.”
To prepare for an unsure influenza season, Dr. Harrison suggests making the influenza vaccine available in August as opposed to October.
Dr. Harrison and Dr. Webby reported no conflicts of interest.
Younger children may be vulnerable to the reemergence of common respiratory viruses such as influenza and respiratory syncytial virus (RSV) as COVID-19 restrictions wane, experts say. The impact could be detrimental.
The COVID-19 pandemic and the implementation of preventative measures such as social distancing, travel restrictions, mask use, and shelter in place, reduced the transmission of respiratory viruses, according to the Centers for Disease Control and Prevention. However, because older infants and toddlers have not been exposed to these bugs during the pandemic, they are vulnerable to suffering severe viral infections.
“[We’ve] been in the honeymoon for 18 months,” said Christopher J. Harrison, MD, professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics in Kansas City, Mo. “We are going to be coming out of the honeymoon and the children who didn’t get sick are going to start packing 2 years’ worth of infections into the next 9 months so there’s going to be twice as many as would be normal.”
The CDC issued a health advisory in June for parts of the southern United States, such as Texas, the Carolinas, and Oklahoma, encouraging broader testing for RSV – a virus that usually causes mild, cold-like symptoms and is the most common cause of bronchiolitis and pneumonia in children – among those who test negative for COVID-19. Virtually all children get an RSV infection by the time they are 2 years old, according to the CDC.
In previous years, RSV usually spread during the fall and spring seasons and usually peaked late December to mid-February. However, there’s been an offseason spike in the common illness this year, with nearly 2,000 confirmed cases each week of July.
Richard J. Webby, PhD, of the infectious diseases department at St. Jude Children’s Research Hospital, Memphis, Tenn., said that although RSV transmits more easily during the winter, the virus is able to thrive during this summer because many children have limited immunity and are more vulnerable to catching the virus than before. Population immunity normally limits a virus to circulating under its most favorable conditions, which is usually the winter. However, because there are a few more “susceptible hosts,” it gives the virus the ability to spread during a time when it typically wouldn’t be able to.
“Now we have a wider range of susceptible kids because they haven’t had that exposure over the past 18 months,” said Dr. Webby, who is on the World Health Organization’s Influenza Vaccine Composition Advisory Team. “It gives the virus more chances to transmit during conditions that are less favorable.”
Dr. Harrison said that, if children continue to take preventative measures such as wearing masks and sanitizing, they can delay catching the RSV – which can be severe in infants and young children – until they’re older and symptoms won’t be as severe.
“The swelling that these viruses cause in the trachea and the bronchial tubes is much bigger in proportion to the overall size of the tubes, so it takes less swelling to clog up the trachea or bronchial tube for the 9-month-old than it does of a 9-year-old,” Dr. Harrison said. “So if a 9-year-old was to get RSV, they’re not going to have nearly the same amount symptoms as the 9-month-old.
Dr. Harrison said delaying RSV in children was never an option before because it’s a virus that’s almost impossible to avoid.
“Hopefully, the mask means that if you get exposed, instead of getting a million virus particles from your classmate or your playmate, you may only get a couple thousand,” Dr. Harrison explained. “And maybe that’s enough that you can fight it off or it may be small enough that you get a mild infection instead of a severe infection.”
A summer surge of RSV has also occurred in Australia. A study published in Clinical Infectious Diseases found that Western Australia saw a 98% reduction in RSV cases. This suggests that COVID-19 restrictions also delayed the RSV season.
Dr. Webby said the lax in penetrative measures against COVID-19 may also affect this upcoming flu season. Usually, around 10%-30% of the population gets infected with the flu each year, but that hasn’t happened the past couple of seasons, he said.
“There might be slightly less overall immunity to these viruses,” Dr. Webby said. “When these viruses do come back, there’s a little bit more room for them to take off.”
Although a severe influenza season rebound this winter is a possibility, Australia continues to experience a historically low flu season. Dr. Harrison, who said the northern hemisphere looks at what’s happening in Australia and the rest of the “southern half of the world because their influenza season is during our summer,” hopes this is an indication that the northern hemisphere will also experience a mild season.
However, there’s no indication of how this upcoming flu season will hit the United States and there isn’t any guidance on what could happen because these historically low levels of respiratory viruses have never happened before, Dr. Webby explained.
He said that, if COVID-19’s delta variant continues to circulate during the fall and winter seasons, it will keep other viruses at low levels. This is because there is rarely a peak of activity of different viruses at the same time.
“When you get infected with the virus, your body’s immune response has this nonspecific reaction that protects you from anything else for a short period of time,” Dr. Webby explained. “When you get a lot of one virus circulating, it’s really hard for these other viruses to get into that population and sort of set off an epidemic of their own.”
To prepare for an unsure influenza season, Dr. Harrison suggests making the influenza vaccine available in August as opposed to October.
Dr. Harrison and Dr. Webby reported no conflicts of interest.
Younger children may be vulnerable to the reemergence of common respiratory viruses such as influenza and respiratory syncytial virus (RSV) as COVID-19 restrictions wane, experts say. The impact could be detrimental.
The COVID-19 pandemic and the implementation of preventative measures such as social distancing, travel restrictions, mask use, and shelter in place, reduced the transmission of respiratory viruses, according to the Centers for Disease Control and Prevention. However, because older infants and toddlers have not been exposed to these bugs during the pandemic, they are vulnerable to suffering severe viral infections.
“[We’ve] been in the honeymoon for 18 months,” said Christopher J. Harrison, MD, professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics in Kansas City, Mo. “We are going to be coming out of the honeymoon and the children who didn’t get sick are going to start packing 2 years’ worth of infections into the next 9 months so there’s going to be twice as many as would be normal.”
The CDC issued a health advisory in June for parts of the southern United States, such as Texas, the Carolinas, and Oklahoma, encouraging broader testing for RSV – a virus that usually causes mild, cold-like symptoms and is the most common cause of bronchiolitis and pneumonia in children – among those who test negative for COVID-19. Virtually all children get an RSV infection by the time they are 2 years old, according to the CDC.
In previous years, RSV usually spread during the fall and spring seasons and usually peaked late December to mid-February. However, there’s been an offseason spike in the common illness this year, with nearly 2,000 confirmed cases each week of July.
Richard J. Webby, PhD, of the infectious diseases department at St. Jude Children’s Research Hospital, Memphis, Tenn., said that although RSV transmits more easily during the winter, the virus is able to thrive during this summer because many children have limited immunity and are more vulnerable to catching the virus than before. Population immunity normally limits a virus to circulating under its most favorable conditions, which is usually the winter. However, because there are a few more “susceptible hosts,” it gives the virus the ability to spread during a time when it typically wouldn’t be able to.
“Now we have a wider range of susceptible kids because they haven’t had that exposure over the past 18 months,” said Dr. Webby, who is on the World Health Organization’s Influenza Vaccine Composition Advisory Team. “It gives the virus more chances to transmit during conditions that are less favorable.”
Dr. Harrison said that, if children continue to take preventative measures such as wearing masks and sanitizing, they can delay catching the RSV – which can be severe in infants and young children – until they’re older and symptoms won’t be as severe.
“The swelling that these viruses cause in the trachea and the bronchial tubes is much bigger in proportion to the overall size of the tubes, so it takes less swelling to clog up the trachea or bronchial tube for the 9-month-old than it does of a 9-year-old,” Dr. Harrison said. “So if a 9-year-old was to get RSV, they’re not going to have nearly the same amount symptoms as the 9-month-old.
Dr. Harrison said delaying RSV in children was never an option before because it’s a virus that’s almost impossible to avoid.
“Hopefully, the mask means that if you get exposed, instead of getting a million virus particles from your classmate or your playmate, you may only get a couple thousand,” Dr. Harrison explained. “And maybe that’s enough that you can fight it off or it may be small enough that you get a mild infection instead of a severe infection.”
A summer surge of RSV has also occurred in Australia. A study published in Clinical Infectious Diseases found that Western Australia saw a 98% reduction in RSV cases. This suggests that COVID-19 restrictions also delayed the RSV season.
Dr. Webby said the lax in penetrative measures against COVID-19 may also affect this upcoming flu season. Usually, around 10%-30% of the population gets infected with the flu each year, but that hasn’t happened the past couple of seasons, he said.
“There might be slightly less overall immunity to these viruses,” Dr. Webby said. “When these viruses do come back, there’s a little bit more room for them to take off.”
Although a severe influenza season rebound this winter is a possibility, Australia continues to experience a historically low flu season. Dr. Harrison, who said the northern hemisphere looks at what’s happening in Australia and the rest of the “southern half of the world because their influenza season is during our summer,” hopes this is an indication that the northern hemisphere will also experience a mild season.
However, there’s no indication of how this upcoming flu season will hit the United States and there isn’t any guidance on what could happen because these historically low levels of respiratory viruses have never happened before, Dr. Webby explained.
He said that, if COVID-19’s delta variant continues to circulate during the fall and winter seasons, it will keep other viruses at low levels. This is because there is rarely a peak of activity of different viruses at the same time.
“When you get infected with the virus, your body’s immune response has this nonspecific reaction that protects you from anything else for a short period of time,” Dr. Webby explained. “When you get a lot of one virus circulating, it’s really hard for these other viruses to get into that population and sort of set off an epidemic of their own.”
To prepare for an unsure influenza season, Dr. Harrison suggests making the influenza vaccine available in August as opposed to October.
Dr. Harrison and Dr. Webby reported no conflicts of interest.
Pfizer vaccine protection wanes after 6 months, study finds
, according to a new study.
The July 28 preprint report of the study, which has not been peer reviewed, suggests a gradual “declining trend in vaccine efficacy” over 6 months after two doses of the Pfizer vaccine in more than 45,000 people worldwide.
The study finds overall effectiveness falls from 96% to 84%.
At the same time, a third booster dose of the Pfizer vaccine increases neutralizing antibody levels against the Delta variant by more than five times, compared to levels after just a second dose in people aged 18-55 years, new data from Pfizer shows.
The third-dose immune response appears even more robust – more than 11 times higher than the second shot – among people aged 65-85 years.
The company noted this could mean an estimated 100-fold increase in Delta variant protection after a third dose. These new findings are outlined in a Pfizer second-quarter 2021 earnings report, which notes that the data are submitted for publication in a medical journal.
The data come from a relatively small number of people studied. There were 11 people in the 18- to 55-year-old group and 12 people in the 65- to 85-year-old group.
“These preliminary data are very encouraging as Delta continues to spread,” Mikael Dolsten, MD, chief scientific officer and president of the Worldwide Research, Development, and Medical organization at Pfizer, said during prepared remarks on a company earnings call July 28, CNN reported.
Availability of a third dose of any of the current COVID-19 vaccines would require amendment of the Food and Drug Administration’s emergency use authorization, or full FDA approval for the vaccine.
The possibility of a third dose authorization or approval has not been without controversy. For example, when Pfizer announced intentions to file for FDA authorization of a booster dose on July 8, the Centers for Disease Control and Prevention, the FDA, and the National Institutes of Health were quick to issue a joint statement saying they would decide when the timing is right for Americans to have a third immunization. The agencies stated, in part, “We are prepared for booster doses if and when the science demonstrates that they are needed.”
In addition, the World Health Organization said at a media briefing on July 12 that rich countries should prioritize sharing of COVID-19 vaccine supplies to other countries in need worldwide before allocating doses for a booster shot for its own residents.
A version of this article first appeared on WebMD.com.
, according to a new study.
The July 28 preprint report of the study, which has not been peer reviewed, suggests a gradual “declining trend in vaccine efficacy” over 6 months after two doses of the Pfizer vaccine in more than 45,000 people worldwide.
The study finds overall effectiveness falls from 96% to 84%.
At the same time, a third booster dose of the Pfizer vaccine increases neutralizing antibody levels against the Delta variant by more than five times, compared to levels after just a second dose in people aged 18-55 years, new data from Pfizer shows.
The third-dose immune response appears even more robust – more than 11 times higher than the second shot – among people aged 65-85 years.
The company noted this could mean an estimated 100-fold increase in Delta variant protection after a third dose. These new findings are outlined in a Pfizer second-quarter 2021 earnings report, which notes that the data are submitted for publication in a medical journal.
The data come from a relatively small number of people studied. There were 11 people in the 18- to 55-year-old group and 12 people in the 65- to 85-year-old group.
“These preliminary data are very encouraging as Delta continues to spread,” Mikael Dolsten, MD, chief scientific officer and president of the Worldwide Research, Development, and Medical organization at Pfizer, said during prepared remarks on a company earnings call July 28, CNN reported.
Availability of a third dose of any of the current COVID-19 vaccines would require amendment of the Food and Drug Administration’s emergency use authorization, or full FDA approval for the vaccine.
The possibility of a third dose authorization or approval has not been without controversy. For example, when Pfizer announced intentions to file for FDA authorization of a booster dose on July 8, the Centers for Disease Control and Prevention, the FDA, and the National Institutes of Health were quick to issue a joint statement saying they would decide when the timing is right for Americans to have a third immunization. The agencies stated, in part, “We are prepared for booster doses if and when the science demonstrates that they are needed.”
In addition, the World Health Organization said at a media briefing on July 12 that rich countries should prioritize sharing of COVID-19 vaccine supplies to other countries in need worldwide before allocating doses for a booster shot for its own residents.
A version of this article first appeared on WebMD.com.
, according to a new study.
The July 28 preprint report of the study, which has not been peer reviewed, suggests a gradual “declining trend in vaccine efficacy” over 6 months after two doses of the Pfizer vaccine in more than 45,000 people worldwide.
The study finds overall effectiveness falls from 96% to 84%.
At the same time, a third booster dose of the Pfizer vaccine increases neutralizing antibody levels against the Delta variant by more than five times, compared to levels after just a second dose in people aged 18-55 years, new data from Pfizer shows.
The third-dose immune response appears even more robust – more than 11 times higher than the second shot – among people aged 65-85 years.
The company noted this could mean an estimated 100-fold increase in Delta variant protection after a third dose. These new findings are outlined in a Pfizer second-quarter 2021 earnings report, which notes that the data are submitted for publication in a medical journal.
The data come from a relatively small number of people studied. There were 11 people in the 18- to 55-year-old group and 12 people in the 65- to 85-year-old group.
“These preliminary data are very encouraging as Delta continues to spread,” Mikael Dolsten, MD, chief scientific officer and president of the Worldwide Research, Development, and Medical organization at Pfizer, said during prepared remarks on a company earnings call July 28, CNN reported.
Availability of a third dose of any of the current COVID-19 vaccines would require amendment of the Food and Drug Administration’s emergency use authorization, or full FDA approval for the vaccine.
The possibility of a third dose authorization or approval has not been without controversy. For example, when Pfizer announced intentions to file for FDA authorization of a booster dose on July 8, the Centers for Disease Control and Prevention, the FDA, and the National Institutes of Health were quick to issue a joint statement saying they would decide when the timing is right for Americans to have a third immunization. The agencies stated, in part, “We are prepared for booster doses if and when the science demonstrates that they are needed.”
In addition, the World Health Organization said at a media briefing on July 12 that rich countries should prioritize sharing of COVID-19 vaccine supplies to other countries in need worldwide before allocating doses for a booster shot for its own residents.
A version of this article first appeared on WebMD.com.
Many pandemic-driven changes to cancer clinical trials should remain
Many of the changes to cancer clinical trials forced through by the COVID-19 pandemic should remain, as they have made trials “more patient centered and efficient,” according to a group of thought leaders in oncology.
Among the potential improvements were more efficient study enrollment through secure electronic platforms, direct shipment of oral drugs to patients, remote assessment of adverse events, and streamlined data collection.
These changes should be implemented on a permanent basis, the group argues in a commentary published online July 21, 2021, in Cancer Discovery, a journal of the American Association for Cancer Research.
“The ability to distribute oral investigational drugs by mail to patients at their home has probably been the single most impactful change to clinical trial conduct, linked with virtual visits with patients to assess side effects and symptoms,” commented lead author Keith Flaherty, MD, who is director of clinical research at Massachusetts General Hospital, a professor at Harvard Medical School, Boston, and a member of the AACR board of directors.
“This has made it more feasible for patients for whom participation in clinical trials poses a disruption of their ability to work or provide care for family members to participate in trials,” he added in a press statement issued by the AACR.
Pandemic halted many clinical trials
A survey of cancer programs in early 2020 showed that nearly 60% halted screening and/or enrollment for at least some trials because of COVID-19.
“In the spring of 2020, clinical trial conduct halted and then restarted focusing on the bare minimum procedures that first allowed patients continued access to their experimental therapies, and then allowed clinical trial sites and sponsors to collect information on the effects of the therapies,” the authors said.
“The COVID-19–induced changes to clinical trials were a big challenge, probably the largest change in clinical trial conduct since the start of modern oncology clinical testing,” they commented.
“But it also represents an opportunity to rethink the key aspects of clinical trial conduct that are strictly necessary to reach the goal of testing the effectiveness of cancer therapies, and which others are dispensable or provide only minor additional contributions,” they added.
As previously reported at the time by this news organization, efforts to find alternative approaches to conducting trials amid the pandemic led to the emergence of a few “silver linings.”
Key adaptations made to clinical trials and highlighted by the authors include:
- Uptake of remote consenting and telemedicine
- Use of alternative laboratories and imaging centers
- Delivery or administration of investigational drugs at patients’ homes or local clinics
- Commercial attainment of study drugs already approved for other indications
Indeed, the restrictions encountered during the pandemic underscore the importance of designing patient-centered trials versus study site–centered trials, added Antoni Ribas, MD, commentary coauthor and immediate past president of the AACR.
Many of the changes implemented during the pandemic could help increase access for patients living in underserved communities who are underrepresented in clinical trials, he explained.
Harnessing the lessons learned
The authors also recommended the following additional adaptations, which they believe will enhance efficiency and further expand access to clinical trials:
- Incorporating patient-reported outcomes and alternative endpoints in efficacy assessments
- Aiming for 100% remote drug infusions and monitoring
- Increasing funding for clinical trials conducted in underserved communities
- Expanding clinical trial eligibility to include patients with a wide range of comorbidities
- Reducing collection of low-grade adverse events and allowing minor protocol deviations
The group’s recommendations are based on discussions by the AACR COVID-19 and Cancer Task Force, in which they participated.
The American Society of Clinical Oncology is also working to leverage pandemic-related lessons to streamline care and trial planning.
ASCO’s “Road to Recovery” recommendations, published in December 2020, aim to “ensure lessons learned from the COVID-19 experience are used to craft a more equitable, accessible, and efficient clinical research system that protects patient safety, ensures scientific integrity, and maintains data quality,” the authors explained.
Dr. Flaherty and colleagues further underscore the importance of focusing on improvements going forward.
“Guided by lessons learned, many of the remote assessments and trial efficiencies deployed during the pandemic can be preserved and improved upon. We strongly encourage use of these streamlined procedures where appropriate in future prospectively designed cancer clinical trials,” they wrote.
Dr. Flaherty reported receiving personal fees from numerous pharmaceutical companies. Dr. Ribas reported receiving grants from Agilent and Bristol Myers Squibb.
A version of this article first appeared on Medscape.com.
Many of the changes to cancer clinical trials forced through by the COVID-19 pandemic should remain, as they have made trials “more patient centered and efficient,” according to a group of thought leaders in oncology.
Among the potential improvements were more efficient study enrollment through secure electronic platforms, direct shipment of oral drugs to patients, remote assessment of adverse events, and streamlined data collection.
These changes should be implemented on a permanent basis, the group argues in a commentary published online July 21, 2021, in Cancer Discovery, a journal of the American Association for Cancer Research.
“The ability to distribute oral investigational drugs by mail to patients at their home has probably been the single most impactful change to clinical trial conduct, linked with virtual visits with patients to assess side effects and symptoms,” commented lead author Keith Flaherty, MD, who is director of clinical research at Massachusetts General Hospital, a professor at Harvard Medical School, Boston, and a member of the AACR board of directors.
“This has made it more feasible for patients for whom participation in clinical trials poses a disruption of their ability to work or provide care for family members to participate in trials,” he added in a press statement issued by the AACR.
Pandemic halted many clinical trials
A survey of cancer programs in early 2020 showed that nearly 60% halted screening and/or enrollment for at least some trials because of COVID-19.
“In the spring of 2020, clinical trial conduct halted and then restarted focusing on the bare minimum procedures that first allowed patients continued access to their experimental therapies, and then allowed clinical trial sites and sponsors to collect information on the effects of the therapies,” the authors said.
“The COVID-19–induced changes to clinical trials were a big challenge, probably the largest change in clinical trial conduct since the start of modern oncology clinical testing,” they commented.
“But it also represents an opportunity to rethink the key aspects of clinical trial conduct that are strictly necessary to reach the goal of testing the effectiveness of cancer therapies, and which others are dispensable or provide only minor additional contributions,” they added.
As previously reported at the time by this news organization, efforts to find alternative approaches to conducting trials amid the pandemic led to the emergence of a few “silver linings.”
Key adaptations made to clinical trials and highlighted by the authors include:
- Uptake of remote consenting and telemedicine
- Use of alternative laboratories and imaging centers
- Delivery or administration of investigational drugs at patients’ homes or local clinics
- Commercial attainment of study drugs already approved for other indications
Indeed, the restrictions encountered during the pandemic underscore the importance of designing patient-centered trials versus study site–centered trials, added Antoni Ribas, MD, commentary coauthor and immediate past president of the AACR.
Many of the changes implemented during the pandemic could help increase access for patients living in underserved communities who are underrepresented in clinical trials, he explained.
Harnessing the lessons learned
The authors also recommended the following additional adaptations, which they believe will enhance efficiency and further expand access to clinical trials:
- Incorporating patient-reported outcomes and alternative endpoints in efficacy assessments
- Aiming for 100% remote drug infusions and monitoring
- Increasing funding for clinical trials conducted in underserved communities
- Expanding clinical trial eligibility to include patients with a wide range of comorbidities
- Reducing collection of low-grade adverse events and allowing minor protocol deviations
The group’s recommendations are based on discussions by the AACR COVID-19 and Cancer Task Force, in which they participated.
The American Society of Clinical Oncology is also working to leverage pandemic-related lessons to streamline care and trial planning.
ASCO’s “Road to Recovery” recommendations, published in December 2020, aim to “ensure lessons learned from the COVID-19 experience are used to craft a more equitable, accessible, and efficient clinical research system that protects patient safety, ensures scientific integrity, and maintains data quality,” the authors explained.
Dr. Flaherty and colleagues further underscore the importance of focusing on improvements going forward.
“Guided by lessons learned, many of the remote assessments and trial efficiencies deployed during the pandemic can be preserved and improved upon. We strongly encourage use of these streamlined procedures where appropriate in future prospectively designed cancer clinical trials,” they wrote.
Dr. Flaherty reported receiving personal fees from numerous pharmaceutical companies. Dr. Ribas reported receiving grants from Agilent and Bristol Myers Squibb.
A version of this article first appeared on Medscape.com.
Many of the changes to cancer clinical trials forced through by the COVID-19 pandemic should remain, as they have made trials “more patient centered and efficient,” according to a group of thought leaders in oncology.
Among the potential improvements were more efficient study enrollment through secure electronic platforms, direct shipment of oral drugs to patients, remote assessment of adverse events, and streamlined data collection.
These changes should be implemented on a permanent basis, the group argues in a commentary published online July 21, 2021, in Cancer Discovery, a journal of the American Association for Cancer Research.
“The ability to distribute oral investigational drugs by mail to patients at their home has probably been the single most impactful change to clinical trial conduct, linked with virtual visits with patients to assess side effects and symptoms,” commented lead author Keith Flaherty, MD, who is director of clinical research at Massachusetts General Hospital, a professor at Harvard Medical School, Boston, and a member of the AACR board of directors.
“This has made it more feasible for patients for whom participation in clinical trials poses a disruption of their ability to work or provide care for family members to participate in trials,” he added in a press statement issued by the AACR.
Pandemic halted many clinical trials
A survey of cancer programs in early 2020 showed that nearly 60% halted screening and/or enrollment for at least some trials because of COVID-19.
“In the spring of 2020, clinical trial conduct halted and then restarted focusing on the bare minimum procedures that first allowed patients continued access to their experimental therapies, and then allowed clinical trial sites and sponsors to collect information on the effects of the therapies,” the authors said.
“The COVID-19–induced changes to clinical trials were a big challenge, probably the largest change in clinical trial conduct since the start of modern oncology clinical testing,” they commented.
“But it also represents an opportunity to rethink the key aspects of clinical trial conduct that are strictly necessary to reach the goal of testing the effectiveness of cancer therapies, and which others are dispensable or provide only minor additional contributions,” they added.
As previously reported at the time by this news organization, efforts to find alternative approaches to conducting trials amid the pandemic led to the emergence of a few “silver linings.”
Key adaptations made to clinical trials and highlighted by the authors include:
- Uptake of remote consenting and telemedicine
- Use of alternative laboratories and imaging centers
- Delivery or administration of investigational drugs at patients’ homes or local clinics
- Commercial attainment of study drugs already approved for other indications
Indeed, the restrictions encountered during the pandemic underscore the importance of designing patient-centered trials versus study site–centered trials, added Antoni Ribas, MD, commentary coauthor and immediate past president of the AACR.
Many of the changes implemented during the pandemic could help increase access for patients living in underserved communities who are underrepresented in clinical trials, he explained.
Harnessing the lessons learned
The authors also recommended the following additional adaptations, which they believe will enhance efficiency and further expand access to clinical trials:
- Incorporating patient-reported outcomes and alternative endpoints in efficacy assessments
- Aiming for 100% remote drug infusions and monitoring
- Increasing funding for clinical trials conducted in underserved communities
- Expanding clinical trial eligibility to include patients with a wide range of comorbidities
- Reducing collection of low-grade adverse events and allowing minor protocol deviations
The group’s recommendations are based on discussions by the AACR COVID-19 and Cancer Task Force, in which they participated.
The American Society of Clinical Oncology is also working to leverage pandemic-related lessons to streamline care and trial planning.
ASCO’s “Road to Recovery” recommendations, published in December 2020, aim to “ensure lessons learned from the COVID-19 experience are used to craft a more equitable, accessible, and efficient clinical research system that protects patient safety, ensures scientific integrity, and maintains data quality,” the authors explained.
Dr. Flaherty and colleagues further underscore the importance of focusing on improvements going forward.
“Guided by lessons learned, many of the remote assessments and trial efficiencies deployed during the pandemic can be preserved and improved upon. We strongly encourage use of these streamlined procedures where appropriate in future prospectively designed cancer clinical trials,” they wrote.
Dr. Flaherty reported receiving personal fees from numerous pharmaceutical companies. Dr. Ribas reported receiving grants from Agilent and Bristol Myers Squibb.
A version of this article first appeared on Medscape.com.
CDC calls for masks in schools, hard-hit areas, even if vaccinated
The agency has called for masks in K-12 school settings and in areas of the United States experiencing high or substantial SARS-CoV-2 transmission, even for the fully vaccinated.
The move reverses a controversial announcement the agency made in May 2021 that fully vaccinated Americans could skip wearing a mask in most settings.
Unlike the increasing vaccination rates and decreasing case numbers reported in May, however, some regions of the United States are now reporting large jumps in COVID-19 case numbers. And the Delta variant as well as new evidence of transmission from breakthrough cases are largely driving these changes.
“Today we have new science related to the [D]elta variant that requires us to update the guidance on what you can do when you are fully vaccinated,” CDC Director Rochelle Walensky, MD, MPH, said during a media briefing July 27.
New evidence has emerged on breakthrough-case transmission risk, for example. “Information on the [D]elta variant from several states and other countries indicates that in rare cases, some people infected with the [D]elta variant after vaccination may be contagious and spread virus to others,” Dr. Walensky said, adding that the viral loads appear to be about the same in vaccinated and unvaccinated individuals.
“This new science is worrisome,” she said.
Even though unvaccinated people represent the vast majority of cases of transmission, Dr. Walensky said, “we thought it was important for [vaccinated] people to understand they have the potential to transmit the virus to others.”
As a result, in addition to continuing to strongly encourage everyone to get vaccinated, the CDC recommends that fully vaccinated people wear masks in public indoor settings to help prevent the spread of the Delta variant in areas with substantial or high transmission, Dr. Walensky said. “This includes schools.”
Masks in schools
The CDC is now recommending universal indoor masking for all teachers, staff, students, and visitors to K-12 schools, regardless of vaccination status. Their goal is to optimize safety and allow children to return to full-time in-person learning in the fall.
The CDC tracks substantial and high transmission rates through the agency’s COVID Data Tracker site. Substantial transmission means between 50 and 100 cases per 100,000 people reported over 7 days and high means more than 100 cases per 100,000 people.
The B.1.617.2, or Delta, variant is believed to be responsible for COVID-19 cases increasing more than 300% nationally from June 19 to July 23, 2021.
“A prudent move”
“I think it’s a prudent move. Given the dominance of the [D]elta variant and the caseloads that we are seeing rising in many locations across the United States, including in my backyard here in San Francisco,” Joe DeRisi, PhD, copresident of the Chan Zuckerberg Biohub and professor of biochemistry and biophysics at the University of California San Francisco, said in an interview.
Dr. DeRisi said he was not surprised that vaccinated people with breakthrough infections could be capable of transmitting the virus. He added that clinical testing done by the Biohub and UCSF produced a lot of data on viral load levels, “and they cover an enormous range.”
What was unexpected to him was the rapid rise of the dominant variant. “The rise of the [D]elta strain is astonishing. It’s happened so fast,” he said.
“I know it’s difficult”
Reacting to the news, Colleen Kraft, MD, said, “One of the things that we’re learning is that if we’re going to have low vaccine uptake or we have a number of people that can’t be vaccinated yet, such as children, that we really need to go back to stopping transmission, which involves mask wearing.”
“I know that it’s very difficult and people feel like we’re sliding backward,” Dr. Kraft said during a media briefing sponsored by Emory University held shortly after the CDC announcement.
She added that the CDC updated guidance seems appropriate. “I don’t think any of us really want to be in this position or want to go back to masking but…we’re finding ourselves in the same place we were a year ago, in July 2020.
“In general we just don’t want anybody to be infected even if there’s a small chance for you to be infected and there’s a small chance for you to transmit it,” said Dr. Kraft, who’s an assistant professor in the department of pathology and associate professor in the department of medicine, division of infectious diseases at Emory University School of Medicine in Atlanta.
Breakthrough transmissions
“The good news is you’re still unlikely to get critically ill if you’re vaccinated. But what has changed with the [D]elta variant is instead of being 90% plus protected from getting the virus at all, you’re probably more in the 70% to 80% range,” James T. McDeavitt, MD, told this news organization.
“So we’re seeing breakthrough infections,” said Dr. McDeavitt, executive vice president and dean of clinical affairs at Baylor College of Medicine in Houston. “We are starting to see [such people] are potentially infectious.” Even if a vaccinated person is individually much less likely to experience serious COVID-19 outcomes, “they can spread it to someone else who spreads it to someone else who is more vulnerable. It puts the more at-risk populations at further risk.”
It breaks down to individual and public health concerns. “I am fully vaccinated. I am very confident I am not going to end up in a hospital,” he said. “Now if I were unvaccinated, with the prevalence of the virus around the country, I’m probably in more danger than I’ve ever been in the course of the pandemic. The unvaccinated are really at risk right now.”
IDSA and AMA support mask change
The Infectious Diseases Society of America (IDSA) has released a statement supporting the new CDC recommendations. “To stay ahead of the spread of the highly transmissible Delta variant, IDSA also urges that in communities with moderate transmission rates, all individuals, even those who are vaccinated, wear masks in indoor public places,” stated IDSA President Barbara D. Alexander, MD, MHS.
“IDSA also supports CDC’s guidance recommending universal indoor masking for all teachers, staff, students, and visitors to K-12 schools, regardless of vaccination status, until vaccines are authorized and widely available to all children and vaccination rates are sufficient to control transmission.”
“Mask wearing will help reduce infections, prevent serious illnesses and death, limit strain on local hospitals and stave off the development of even more troubling variants,” she added.
The American Medical Association (AMA) also released a statement supporting the CDC’s policy changes.
“According to the CDC, emerging data indicates that vaccinated individuals infected with the Delta variant have similar viral loads as those who are unvaccinated and are capable of transmission,” AMA President Gerald E. Harmon, MD said in the statement.
“However, the science remains clear, the authorized vaccines remain safe and effective in preventing severe complications from COVID-19, including hospitalization and death,” he stated. “We strongly support the updated recommendations, which call for universal masking in areas of high or substantial COVID-19 transmission and in K-12 schools, to help reduce transmission of the virus. Wearing a mask is a small but important protective measure that can help us all stay safer.”
“The highest spread of cases and [most] severe outcomes are happening in places with low vaccination rates and among unvaccinated people,” Dr. Walensky said. “With the [D]elta variant, vaccinating more Americans now is more urgent than ever.”
“This moment, and the associated suffering, illness, and death, could have been avoided with higher vaccination coverage in this country,” she said.
A version of this article first appeared on Medscape.com.
The agency has called for masks in K-12 school settings and in areas of the United States experiencing high or substantial SARS-CoV-2 transmission, even for the fully vaccinated.
The move reverses a controversial announcement the agency made in May 2021 that fully vaccinated Americans could skip wearing a mask in most settings.
Unlike the increasing vaccination rates and decreasing case numbers reported in May, however, some regions of the United States are now reporting large jumps in COVID-19 case numbers. And the Delta variant as well as new evidence of transmission from breakthrough cases are largely driving these changes.
“Today we have new science related to the [D]elta variant that requires us to update the guidance on what you can do when you are fully vaccinated,” CDC Director Rochelle Walensky, MD, MPH, said during a media briefing July 27.
New evidence has emerged on breakthrough-case transmission risk, for example. “Information on the [D]elta variant from several states and other countries indicates that in rare cases, some people infected with the [D]elta variant after vaccination may be contagious and spread virus to others,” Dr. Walensky said, adding that the viral loads appear to be about the same in vaccinated and unvaccinated individuals.
“This new science is worrisome,” she said.
Even though unvaccinated people represent the vast majority of cases of transmission, Dr. Walensky said, “we thought it was important for [vaccinated] people to understand they have the potential to transmit the virus to others.”
As a result, in addition to continuing to strongly encourage everyone to get vaccinated, the CDC recommends that fully vaccinated people wear masks in public indoor settings to help prevent the spread of the Delta variant in areas with substantial or high transmission, Dr. Walensky said. “This includes schools.”
Masks in schools
The CDC is now recommending universal indoor masking for all teachers, staff, students, and visitors to K-12 schools, regardless of vaccination status. Their goal is to optimize safety and allow children to return to full-time in-person learning in the fall.
The CDC tracks substantial and high transmission rates through the agency’s COVID Data Tracker site. Substantial transmission means between 50 and 100 cases per 100,000 people reported over 7 days and high means more than 100 cases per 100,000 people.
The B.1.617.2, or Delta, variant is believed to be responsible for COVID-19 cases increasing more than 300% nationally from June 19 to July 23, 2021.
“A prudent move”
“I think it’s a prudent move. Given the dominance of the [D]elta variant and the caseloads that we are seeing rising in many locations across the United States, including in my backyard here in San Francisco,” Joe DeRisi, PhD, copresident of the Chan Zuckerberg Biohub and professor of biochemistry and biophysics at the University of California San Francisco, said in an interview.
Dr. DeRisi said he was not surprised that vaccinated people with breakthrough infections could be capable of transmitting the virus. He added that clinical testing done by the Biohub and UCSF produced a lot of data on viral load levels, “and they cover an enormous range.”
What was unexpected to him was the rapid rise of the dominant variant. “The rise of the [D]elta strain is astonishing. It’s happened so fast,” he said.
“I know it’s difficult”
Reacting to the news, Colleen Kraft, MD, said, “One of the things that we’re learning is that if we’re going to have low vaccine uptake or we have a number of people that can’t be vaccinated yet, such as children, that we really need to go back to stopping transmission, which involves mask wearing.”
“I know that it’s very difficult and people feel like we’re sliding backward,” Dr. Kraft said during a media briefing sponsored by Emory University held shortly after the CDC announcement.
She added that the CDC updated guidance seems appropriate. “I don’t think any of us really want to be in this position or want to go back to masking but…we’re finding ourselves in the same place we were a year ago, in July 2020.
“In general we just don’t want anybody to be infected even if there’s a small chance for you to be infected and there’s a small chance for you to transmit it,” said Dr. Kraft, who’s an assistant professor in the department of pathology and associate professor in the department of medicine, division of infectious diseases at Emory University School of Medicine in Atlanta.
Breakthrough transmissions
“The good news is you’re still unlikely to get critically ill if you’re vaccinated. But what has changed with the [D]elta variant is instead of being 90% plus protected from getting the virus at all, you’re probably more in the 70% to 80% range,” James T. McDeavitt, MD, told this news organization.
“So we’re seeing breakthrough infections,” said Dr. McDeavitt, executive vice president and dean of clinical affairs at Baylor College of Medicine in Houston. “We are starting to see [such people] are potentially infectious.” Even if a vaccinated person is individually much less likely to experience serious COVID-19 outcomes, “they can spread it to someone else who spreads it to someone else who is more vulnerable. It puts the more at-risk populations at further risk.”
It breaks down to individual and public health concerns. “I am fully vaccinated. I am very confident I am not going to end up in a hospital,” he said. “Now if I were unvaccinated, with the prevalence of the virus around the country, I’m probably in more danger than I’ve ever been in the course of the pandemic. The unvaccinated are really at risk right now.”
IDSA and AMA support mask change
The Infectious Diseases Society of America (IDSA) has released a statement supporting the new CDC recommendations. “To stay ahead of the spread of the highly transmissible Delta variant, IDSA also urges that in communities with moderate transmission rates, all individuals, even those who are vaccinated, wear masks in indoor public places,” stated IDSA President Barbara D. Alexander, MD, MHS.
“IDSA also supports CDC’s guidance recommending universal indoor masking for all teachers, staff, students, and visitors to K-12 schools, regardless of vaccination status, until vaccines are authorized and widely available to all children and vaccination rates are sufficient to control transmission.”
“Mask wearing will help reduce infections, prevent serious illnesses and death, limit strain on local hospitals and stave off the development of even more troubling variants,” she added.
The American Medical Association (AMA) also released a statement supporting the CDC’s policy changes.
“According to the CDC, emerging data indicates that vaccinated individuals infected with the Delta variant have similar viral loads as those who are unvaccinated and are capable of transmission,” AMA President Gerald E. Harmon, MD said in the statement.
“However, the science remains clear, the authorized vaccines remain safe and effective in preventing severe complications from COVID-19, including hospitalization and death,” he stated. “We strongly support the updated recommendations, which call for universal masking in areas of high or substantial COVID-19 transmission and in K-12 schools, to help reduce transmission of the virus. Wearing a mask is a small but important protective measure that can help us all stay safer.”
“The highest spread of cases and [most] severe outcomes are happening in places with low vaccination rates and among unvaccinated people,” Dr. Walensky said. “With the [D]elta variant, vaccinating more Americans now is more urgent than ever.”
“This moment, and the associated suffering, illness, and death, could have been avoided with higher vaccination coverage in this country,” she said.
A version of this article first appeared on Medscape.com.
The agency has called for masks in K-12 school settings and in areas of the United States experiencing high or substantial SARS-CoV-2 transmission, even for the fully vaccinated.
The move reverses a controversial announcement the agency made in May 2021 that fully vaccinated Americans could skip wearing a mask in most settings.
Unlike the increasing vaccination rates and decreasing case numbers reported in May, however, some regions of the United States are now reporting large jumps in COVID-19 case numbers. And the Delta variant as well as new evidence of transmission from breakthrough cases are largely driving these changes.
“Today we have new science related to the [D]elta variant that requires us to update the guidance on what you can do when you are fully vaccinated,” CDC Director Rochelle Walensky, MD, MPH, said during a media briefing July 27.
New evidence has emerged on breakthrough-case transmission risk, for example. “Information on the [D]elta variant from several states and other countries indicates that in rare cases, some people infected with the [D]elta variant after vaccination may be contagious and spread virus to others,” Dr. Walensky said, adding that the viral loads appear to be about the same in vaccinated and unvaccinated individuals.
“This new science is worrisome,” she said.
Even though unvaccinated people represent the vast majority of cases of transmission, Dr. Walensky said, “we thought it was important for [vaccinated] people to understand they have the potential to transmit the virus to others.”
As a result, in addition to continuing to strongly encourage everyone to get vaccinated, the CDC recommends that fully vaccinated people wear masks in public indoor settings to help prevent the spread of the Delta variant in areas with substantial or high transmission, Dr. Walensky said. “This includes schools.”
Masks in schools
The CDC is now recommending universal indoor masking for all teachers, staff, students, and visitors to K-12 schools, regardless of vaccination status. Their goal is to optimize safety and allow children to return to full-time in-person learning in the fall.
The CDC tracks substantial and high transmission rates through the agency’s COVID Data Tracker site. Substantial transmission means between 50 and 100 cases per 100,000 people reported over 7 days and high means more than 100 cases per 100,000 people.
The B.1.617.2, or Delta, variant is believed to be responsible for COVID-19 cases increasing more than 300% nationally from June 19 to July 23, 2021.
“A prudent move”
“I think it’s a prudent move. Given the dominance of the [D]elta variant and the caseloads that we are seeing rising in many locations across the United States, including in my backyard here in San Francisco,” Joe DeRisi, PhD, copresident of the Chan Zuckerberg Biohub and professor of biochemistry and biophysics at the University of California San Francisco, said in an interview.
Dr. DeRisi said he was not surprised that vaccinated people with breakthrough infections could be capable of transmitting the virus. He added that clinical testing done by the Biohub and UCSF produced a lot of data on viral load levels, “and they cover an enormous range.”
What was unexpected to him was the rapid rise of the dominant variant. “The rise of the [D]elta strain is astonishing. It’s happened so fast,” he said.
“I know it’s difficult”
Reacting to the news, Colleen Kraft, MD, said, “One of the things that we’re learning is that if we’re going to have low vaccine uptake or we have a number of people that can’t be vaccinated yet, such as children, that we really need to go back to stopping transmission, which involves mask wearing.”
“I know that it’s very difficult and people feel like we’re sliding backward,” Dr. Kraft said during a media briefing sponsored by Emory University held shortly after the CDC announcement.
She added that the CDC updated guidance seems appropriate. “I don’t think any of us really want to be in this position or want to go back to masking but…we’re finding ourselves in the same place we were a year ago, in July 2020.
“In general we just don’t want anybody to be infected even if there’s a small chance for you to be infected and there’s a small chance for you to transmit it,” said Dr. Kraft, who’s an assistant professor in the department of pathology and associate professor in the department of medicine, division of infectious diseases at Emory University School of Medicine in Atlanta.
Breakthrough transmissions
“The good news is you’re still unlikely to get critically ill if you’re vaccinated. But what has changed with the [D]elta variant is instead of being 90% plus protected from getting the virus at all, you’re probably more in the 70% to 80% range,” James T. McDeavitt, MD, told this news organization.
“So we’re seeing breakthrough infections,” said Dr. McDeavitt, executive vice president and dean of clinical affairs at Baylor College of Medicine in Houston. “We are starting to see [such people] are potentially infectious.” Even if a vaccinated person is individually much less likely to experience serious COVID-19 outcomes, “they can spread it to someone else who spreads it to someone else who is more vulnerable. It puts the more at-risk populations at further risk.”
It breaks down to individual and public health concerns. “I am fully vaccinated. I am very confident I am not going to end up in a hospital,” he said. “Now if I were unvaccinated, with the prevalence of the virus around the country, I’m probably in more danger than I’ve ever been in the course of the pandemic. The unvaccinated are really at risk right now.”
IDSA and AMA support mask change
The Infectious Diseases Society of America (IDSA) has released a statement supporting the new CDC recommendations. “To stay ahead of the spread of the highly transmissible Delta variant, IDSA also urges that in communities with moderate transmission rates, all individuals, even those who are vaccinated, wear masks in indoor public places,” stated IDSA President Barbara D. Alexander, MD, MHS.
“IDSA also supports CDC’s guidance recommending universal indoor masking for all teachers, staff, students, and visitors to K-12 schools, regardless of vaccination status, until vaccines are authorized and widely available to all children and vaccination rates are sufficient to control transmission.”
“Mask wearing will help reduce infections, prevent serious illnesses and death, limit strain on local hospitals and stave off the development of even more troubling variants,” she added.
The American Medical Association (AMA) also released a statement supporting the CDC’s policy changes.
“According to the CDC, emerging data indicates that vaccinated individuals infected with the Delta variant have similar viral loads as those who are unvaccinated and are capable of transmission,” AMA President Gerald E. Harmon, MD said in the statement.
“However, the science remains clear, the authorized vaccines remain safe and effective in preventing severe complications from COVID-19, including hospitalization and death,” he stated. “We strongly support the updated recommendations, which call for universal masking in areas of high or substantial COVID-19 transmission and in K-12 schools, to help reduce transmission of the virus. Wearing a mask is a small but important protective measure that can help us all stay safer.”
“The highest spread of cases and [most] severe outcomes are happening in places with low vaccination rates and among unvaccinated people,” Dr. Walensky said. “With the [D]elta variant, vaccinating more Americans now is more urgent than ever.”
“This moment, and the associated suffering, illness, and death, could have been avoided with higher vaccination coverage in this country,” she said.
A version of this article first appeared on Medscape.com.
The VA, California, and NYC requiring employee vaccinations
-- or, in the case of California and New York City, undergo regular testing.
The VA becomes the first federal agency to mandate COVID vaccinations for workers. In a news release, VA Secretary Denis McDonough said the mandate is “the best way to keep Veterans safe, especially as the Delta variant spreads across the country.”
VA health care personnel -- including doctors, dentists, podiatrists, optometrists, registered nurses, physician assistants, and chiropractors -- have 8 weeks to become fully vaccinated, the news release said. The New York Times reported that about 115,000 workers will be affected.
The trifecta of federal-state-municipal vaccine requirements arrived as the nation searches for ways to get more people vaccinated to tamp down the Delta variant.
Some organizations, including the military, have already said vaccinations will be required as soon as the Food and Drug Administration formally approves the vaccines, which are now given under emergency use authorizations. The FDA has said the Pfizer vaccine could receive full approval within months.
California Gov. Gavin Newsom said the requirements he announced July 27 were the first in the nation on the state level.
“As the state’s largest employer, we are leading by example and requiring all state and health care workers to show proof of vaccination or be tested regularly, and we are encouraging local governments and businesses to do the same,” he said in a news release.
California employees must provide proof of vaccination or get tested at least once a week. The policy starts Aug. 2 for state employees and Aug. 9 for state health care workers and employees of congregate facilities, such as jails or homeless shelters.
California, especially the southern part of the state, is grappling with a COVID-19 surge. The state’s daily case rate more than quadrupled, from a low of 1.9 cases per 100,000 in May to at least 9.5 cases per 100,000 today, the release said.
In New York City, Mayor Bill de Blasio had previously announced that city health and hospital employees and those working in Department of Health and Mental Hygiene clinical settings would be required to provide proof of vaccination or have regular testing.
On July 27 he expanded the rule to cover all city employees, with a Sept. 13 deadline for most of them, according to a news release.
“This is what it takes to continue our recovery for all of us while fighting back the Delta variant,” Mayor de Blasio said. “It’s going to take all of us to finally end the fight against COVID-19.”
“We have a moral responsibility to take every precaution possible to ensure we keep ourselves, our colleagues and loved ones safe,” NYC Health + Hospitals President and CEO Mitchell Katz, MD, said in the release. “Our city’s new testing requirement for city workers provides more [peace] of mind until more people get their safe and effective COVID-19 vaccine.”
NBC News reported the plan would affect about 340,000 employees.
A version of this article first appeared on WebMD.com.
-- or, in the case of California and New York City, undergo regular testing.
The VA becomes the first federal agency to mandate COVID vaccinations for workers. In a news release, VA Secretary Denis McDonough said the mandate is “the best way to keep Veterans safe, especially as the Delta variant spreads across the country.”
VA health care personnel -- including doctors, dentists, podiatrists, optometrists, registered nurses, physician assistants, and chiropractors -- have 8 weeks to become fully vaccinated, the news release said. The New York Times reported that about 115,000 workers will be affected.
The trifecta of federal-state-municipal vaccine requirements arrived as the nation searches for ways to get more people vaccinated to tamp down the Delta variant.
Some organizations, including the military, have already said vaccinations will be required as soon as the Food and Drug Administration formally approves the vaccines, which are now given under emergency use authorizations. The FDA has said the Pfizer vaccine could receive full approval within months.
California Gov. Gavin Newsom said the requirements he announced July 27 were the first in the nation on the state level.
“As the state’s largest employer, we are leading by example and requiring all state and health care workers to show proof of vaccination or be tested regularly, and we are encouraging local governments and businesses to do the same,” he said in a news release.
California employees must provide proof of vaccination or get tested at least once a week. The policy starts Aug. 2 for state employees and Aug. 9 for state health care workers and employees of congregate facilities, such as jails or homeless shelters.
California, especially the southern part of the state, is grappling with a COVID-19 surge. The state’s daily case rate more than quadrupled, from a low of 1.9 cases per 100,000 in May to at least 9.5 cases per 100,000 today, the release said.
In New York City, Mayor Bill de Blasio had previously announced that city health and hospital employees and those working in Department of Health and Mental Hygiene clinical settings would be required to provide proof of vaccination or have regular testing.
On July 27 he expanded the rule to cover all city employees, with a Sept. 13 deadline for most of them, according to a news release.
“This is what it takes to continue our recovery for all of us while fighting back the Delta variant,” Mayor de Blasio said. “It’s going to take all of us to finally end the fight against COVID-19.”
“We have a moral responsibility to take every precaution possible to ensure we keep ourselves, our colleagues and loved ones safe,” NYC Health + Hospitals President and CEO Mitchell Katz, MD, said in the release. “Our city’s new testing requirement for city workers provides more [peace] of mind until more people get their safe and effective COVID-19 vaccine.”
NBC News reported the plan would affect about 340,000 employees.
A version of this article first appeared on WebMD.com.
-- or, in the case of California and New York City, undergo regular testing.
The VA becomes the first federal agency to mandate COVID vaccinations for workers. In a news release, VA Secretary Denis McDonough said the mandate is “the best way to keep Veterans safe, especially as the Delta variant spreads across the country.”
VA health care personnel -- including doctors, dentists, podiatrists, optometrists, registered nurses, physician assistants, and chiropractors -- have 8 weeks to become fully vaccinated, the news release said. The New York Times reported that about 115,000 workers will be affected.
The trifecta of federal-state-municipal vaccine requirements arrived as the nation searches for ways to get more people vaccinated to tamp down the Delta variant.
Some organizations, including the military, have already said vaccinations will be required as soon as the Food and Drug Administration formally approves the vaccines, which are now given under emergency use authorizations. The FDA has said the Pfizer vaccine could receive full approval within months.
California Gov. Gavin Newsom said the requirements he announced July 27 were the first in the nation on the state level.
“As the state’s largest employer, we are leading by example and requiring all state and health care workers to show proof of vaccination or be tested regularly, and we are encouraging local governments and businesses to do the same,” he said in a news release.
California employees must provide proof of vaccination or get tested at least once a week. The policy starts Aug. 2 for state employees and Aug. 9 for state health care workers and employees of congregate facilities, such as jails or homeless shelters.
California, especially the southern part of the state, is grappling with a COVID-19 surge. The state’s daily case rate more than quadrupled, from a low of 1.9 cases per 100,000 in May to at least 9.5 cases per 100,000 today, the release said.
In New York City, Mayor Bill de Blasio had previously announced that city health and hospital employees and those working in Department of Health and Mental Hygiene clinical settings would be required to provide proof of vaccination or have regular testing.
On July 27 he expanded the rule to cover all city employees, with a Sept. 13 deadline for most of them, according to a news release.
“This is what it takes to continue our recovery for all of us while fighting back the Delta variant,” Mayor de Blasio said. “It’s going to take all of us to finally end the fight against COVID-19.”
“We have a moral responsibility to take every precaution possible to ensure we keep ourselves, our colleagues and loved ones safe,” NYC Health + Hospitals President and CEO Mitchell Katz, MD, said in the release. “Our city’s new testing requirement for city workers provides more [peace] of mind until more people get their safe and effective COVID-19 vaccine.”
NBC News reported the plan would affect about 340,000 employees.
A version of this article first appeared on WebMD.com.
Children and COVID: Vaccinations, new cases both rising
COVID-19 vaccine initiations rose in U.S. children for the second consecutive week, but new pediatric cases jumped by 64% in just 1 week, according to new data.
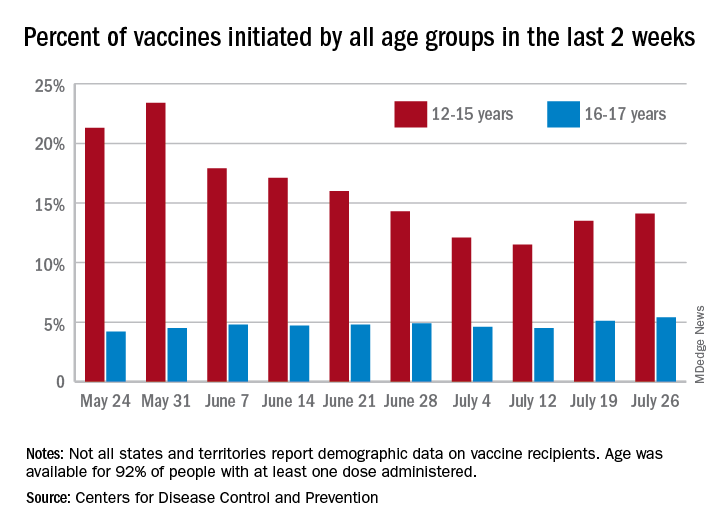
the American Academy of Pediatrics and the Children’s Hospital Association said in their weekly COVID-19 report.
“After decreases in weekly reported cases over the past couple of months, in July we have seen steady increases in cases added to the cumulative total,” the AAP noted. In this latest reversal of COVID fortunes, the steady increase in new cases is in its fourth consecutive week since hitting a low of 8,447 in late June.
As of July 22, the total number of reported cases was over 4.12 million in 49 states, the District of Columbia, New York City, Puerto Rico, and Guam, and there have been 349 deaths in children in the 46 jurisdictions reporting age distributions of COVID-19 deaths, the AAP and CHA said in their report.
Meanwhile, over 9.3 million children received at least one dose of COVID vaccine as of July 26, according to the Centers for Disease Control and Prevention.
Vaccine initiation rose for the second week in a row after falling for several weeks as 301,000 children aged 12-15 years and almost 115,000 children aged 16-17 got their first dose during the week ending July 26. Children aged 12-15 represented 14.1% (up from 13.5% a week before) of all first vaccinations and 16- to 17-year-olds were 5.4% (up from 5.1%) of all vaccine initiators, according to the CDC’s COVID Data Tracker.
Just over 37% of all 12- to 15-year-olds have received at least one dose of the Pfizer-BioNTech vaccine since the CDC approved its use for children under age 16 in May, and almost 28% are fully vaccinated. Use in children aged 16-17 started earlier (December 2020), and 48% of that age group have received a first dose and over 39% have completed the vaccine regimen, the CDC said.
COVID-19 vaccine initiations rose in U.S. children for the second consecutive week, but new pediatric cases jumped by 64% in just 1 week, according to new data.

the American Academy of Pediatrics and the Children’s Hospital Association said in their weekly COVID-19 report.
“After decreases in weekly reported cases over the past couple of months, in July we have seen steady increases in cases added to the cumulative total,” the AAP noted. In this latest reversal of COVID fortunes, the steady increase in new cases is in its fourth consecutive week since hitting a low of 8,447 in late June.
As of July 22, the total number of reported cases was over 4.12 million in 49 states, the District of Columbia, New York City, Puerto Rico, and Guam, and there have been 349 deaths in children in the 46 jurisdictions reporting age distributions of COVID-19 deaths, the AAP and CHA said in their report.
Meanwhile, over 9.3 million children received at least one dose of COVID vaccine as of July 26, according to the Centers for Disease Control and Prevention.
Vaccine initiation rose for the second week in a row after falling for several weeks as 301,000 children aged 12-15 years and almost 115,000 children aged 16-17 got their first dose during the week ending July 26. Children aged 12-15 represented 14.1% (up from 13.5% a week before) of all first vaccinations and 16- to 17-year-olds were 5.4% (up from 5.1%) of all vaccine initiators, according to the CDC’s COVID Data Tracker.
Just over 37% of all 12- to 15-year-olds have received at least one dose of the Pfizer-BioNTech vaccine since the CDC approved its use for children under age 16 in May, and almost 28% are fully vaccinated. Use in children aged 16-17 started earlier (December 2020), and 48% of that age group have received a first dose and over 39% have completed the vaccine regimen, the CDC said.
COVID-19 vaccine initiations rose in U.S. children for the second consecutive week, but new pediatric cases jumped by 64% in just 1 week, according to new data.

the American Academy of Pediatrics and the Children’s Hospital Association said in their weekly COVID-19 report.
“After decreases in weekly reported cases over the past couple of months, in July we have seen steady increases in cases added to the cumulative total,” the AAP noted. In this latest reversal of COVID fortunes, the steady increase in new cases is in its fourth consecutive week since hitting a low of 8,447 in late June.
As of July 22, the total number of reported cases was over 4.12 million in 49 states, the District of Columbia, New York City, Puerto Rico, and Guam, and there have been 349 deaths in children in the 46 jurisdictions reporting age distributions of COVID-19 deaths, the AAP and CHA said in their report.
Meanwhile, over 9.3 million children received at least one dose of COVID vaccine as of July 26, according to the Centers for Disease Control and Prevention.
Vaccine initiation rose for the second week in a row after falling for several weeks as 301,000 children aged 12-15 years and almost 115,000 children aged 16-17 got their first dose during the week ending July 26. Children aged 12-15 represented 14.1% (up from 13.5% a week before) of all first vaccinations and 16- to 17-year-olds were 5.4% (up from 5.1%) of all vaccine initiators, according to the CDC’s COVID Data Tracker.
Just over 37% of all 12- to 15-year-olds have received at least one dose of the Pfizer-BioNTech vaccine since the CDC approved its use for children under age 16 in May, and almost 28% are fully vaccinated. Use in children aged 16-17 started earlier (December 2020), and 48% of that age group have received a first dose and over 39% have completed the vaccine regimen, the CDC said.
COVID-19 vaccination does not increase risk of flare in patients with lupus
COVID-19 vaccinations appear to be well tolerated in patients with systemic lupus erythematosus (SLE) and come with a low risk of flare, according to the results of a global, web-based survey.
“Disseminating these reassuring data might prove crucial to increasing vaccine coverage in patients with SLE,” wrote lead author Renaud Felten, MD, of Strasbourg (France) University Hospital. Their results were published as a comment in Lancet Rheumatology.
To assess vaccine tolerability among lupus patients, the cross-sectional Tolerance and Consequences of Vaccination Against COVID-19 in Lupus Patients (VACOLUP) study analyzed a 43-question survey of 696 participants with a self-reported, medically confirmed diagnosis of SLE from 30 countries between March 22, 2021, and May 17, 2021. The cohort was 96% women, and their median age was 42 (interquartile range, 34-51). Nearly 36% of respondents were from Italy, 27% were from Chile, 13% were from France, and just under 9% were Americans. All participants received at least one dose of COVID-19 vaccine, and 49% received a second dose. The most common vaccines were Pfizer-BioNTech (57%), Sinovac (22%), AstraZeneca (10%), and Moderna (8%).
Only 21 participants (3%) reported a medically confirmed SLE flare after a median of 3 days (IQR, 0-29) post COVID vaccination, with most experiencing musculoskeletal symptoms (90%) and fatigue (86%). Of the 21 cases, 15 reported a subsequent change in SLE treatment and 4 were admitted to the hospital. A previous flare that occurred within a year before vaccination was associated with an increased risk of flare post vaccination (relative risk, 5.52; 95% confidence interval, 2.17-14.03; P < .0001).
Side effects – including swelling, soreness, fever, chills, fatigue, joint and muscle pain, nausea, and headache – were reported in 45% of participants (n = 316) after their first dose and in 53% of the 343 participants who received a second dose. There was no notable difference in the likelihood of side effects across gender and age or in patients who received mRNA vaccines, compared with vaccines with other modes of action. Patients who reported side effects after the first dose were more likely to also report them after the second, compared with those who reported none (109 [81%] of 135 vs. 72 [35%] of 205; RR, 2.30; 95% CI, 1.88-2.82; P < .0001).
In the majority of cases (2,232 of 2,683), the side effects were of minor or moderate intensity and did not affect the participants’ ability to perform daily tasks. The study found no significant association between side effects and a SLE flare and SLE medications or previous SLE disease manifestations.
When asked to comment on the study, Amit Saxena, MD, of the Lupus Center at New York University Langone Health, said: “What we are seeing is pretty mild to moderate in terms of follow-up side effects or lupus-related activity. Several studies have shown this amongst our autoimmune rheumatology cohort, as well as what I’ve seen clinically in my own patients. More than anything else, numbers are the most important, and this is a large study.”
He acknowledged the benefits of going directly to patients to gauge their responses and reactions, giving them the opportunity to share concerns that physicians may not think about.
“As rheumatologists, we tend to focus on certain things that might not necessarily be what the patients themselves focus on,” he said. “I think the fact that this questionnaire dealt with a lot of what people complain about – fatigue, sore arm, things that we know are part of getting the vaccine – they aren’t necessarily things we capture with tools that screen for lupus flares, for example.”
More than anything, Dr. Saxena commended the study’s timeliness. “Patients are constantly asking us about the vaccine, and there’s so much misinformation,” he said. “People say, ‘Because I have lupus, I was told not to get vaccinated.’ I don’t know where they get that information from; we are telling everyone to get it, especially our lupus patients.”
The authors recognized their study’s main limitation as the self-reported and subjective nature of the survey, which they attempted to mitigate by asking for medically confirmed flares only. They noted, however, that the short median time between vaccination and flare onset could be caused by patients confusing expected side effects for something more serious, meaning the 3% figure “could be an overestimation of the actual flare rate.”
“Vaccination is recommended for patients with rheumatic and musculoskeletal diseases according to the American College of Rheumatology,” they added, “irrespective of disease activity and severity.”
Several authors reported potential conflicts of interest, including receiving consultancy fees and grants from Pfizer, GlaxoSmithKline, AbbVie, and Janssen, all unrelated to the study.
COVID-19 vaccinations appear to be well tolerated in patients with systemic lupus erythematosus (SLE) and come with a low risk of flare, according to the results of a global, web-based survey.
“Disseminating these reassuring data might prove crucial to increasing vaccine coverage in patients with SLE,” wrote lead author Renaud Felten, MD, of Strasbourg (France) University Hospital. Their results were published as a comment in Lancet Rheumatology.
To assess vaccine tolerability among lupus patients, the cross-sectional Tolerance and Consequences of Vaccination Against COVID-19 in Lupus Patients (VACOLUP) study analyzed a 43-question survey of 696 participants with a self-reported, medically confirmed diagnosis of SLE from 30 countries between March 22, 2021, and May 17, 2021. The cohort was 96% women, and their median age was 42 (interquartile range, 34-51). Nearly 36% of respondents were from Italy, 27% were from Chile, 13% were from France, and just under 9% were Americans. All participants received at least one dose of COVID-19 vaccine, and 49% received a second dose. The most common vaccines were Pfizer-BioNTech (57%), Sinovac (22%), AstraZeneca (10%), and Moderna (8%).
Only 21 participants (3%) reported a medically confirmed SLE flare after a median of 3 days (IQR, 0-29) post COVID vaccination, with most experiencing musculoskeletal symptoms (90%) and fatigue (86%). Of the 21 cases, 15 reported a subsequent change in SLE treatment and 4 were admitted to the hospital. A previous flare that occurred within a year before vaccination was associated with an increased risk of flare post vaccination (relative risk, 5.52; 95% confidence interval, 2.17-14.03; P < .0001).
Side effects – including swelling, soreness, fever, chills, fatigue, joint and muscle pain, nausea, and headache – were reported in 45% of participants (n = 316) after their first dose and in 53% of the 343 participants who received a second dose. There was no notable difference in the likelihood of side effects across gender and age or in patients who received mRNA vaccines, compared with vaccines with other modes of action. Patients who reported side effects after the first dose were more likely to also report them after the second, compared with those who reported none (109 [81%] of 135 vs. 72 [35%] of 205; RR, 2.30; 95% CI, 1.88-2.82; P < .0001).
In the majority of cases (2,232 of 2,683), the side effects were of minor or moderate intensity and did not affect the participants’ ability to perform daily tasks. The study found no significant association between side effects and a SLE flare and SLE medications or previous SLE disease manifestations.
When asked to comment on the study, Amit Saxena, MD, of the Lupus Center at New York University Langone Health, said: “What we are seeing is pretty mild to moderate in terms of follow-up side effects or lupus-related activity. Several studies have shown this amongst our autoimmune rheumatology cohort, as well as what I’ve seen clinically in my own patients. More than anything else, numbers are the most important, and this is a large study.”
He acknowledged the benefits of going directly to patients to gauge their responses and reactions, giving them the opportunity to share concerns that physicians may not think about.
“As rheumatologists, we tend to focus on certain things that might not necessarily be what the patients themselves focus on,” he said. “I think the fact that this questionnaire dealt with a lot of what people complain about – fatigue, sore arm, things that we know are part of getting the vaccine – they aren’t necessarily things we capture with tools that screen for lupus flares, for example.”
More than anything, Dr. Saxena commended the study’s timeliness. “Patients are constantly asking us about the vaccine, and there’s so much misinformation,” he said. “People say, ‘Because I have lupus, I was told not to get vaccinated.’ I don’t know where they get that information from; we are telling everyone to get it, especially our lupus patients.”
The authors recognized their study’s main limitation as the self-reported and subjective nature of the survey, which they attempted to mitigate by asking for medically confirmed flares only. They noted, however, that the short median time between vaccination and flare onset could be caused by patients confusing expected side effects for something more serious, meaning the 3% figure “could be an overestimation of the actual flare rate.”
“Vaccination is recommended for patients with rheumatic and musculoskeletal diseases according to the American College of Rheumatology,” they added, “irrespective of disease activity and severity.”
Several authors reported potential conflicts of interest, including receiving consultancy fees and grants from Pfizer, GlaxoSmithKline, AbbVie, and Janssen, all unrelated to the study.
COVID-19 vaccinations appear to be well tolerated in patients with systemic lupus erythematosus (SLE) and come with a low risk of flare, according to the results of a global, web-based survey.
“Disseminating these reassuring data might prove crucial to increasing vaccine coverage in patients with SLE,” wrote lead author Renaud Felten, MD, of Strasbourg (France) University Hospital. Their results were published as a comment in Lancet Rheumatology.
To assess vaccine tolerability among lupus patients, the cross-sectional Tolerance and Consequences of Vaccination Against COVID-19 in Lupus Patients (VACOLUP) study analyzed a 43-question survey of 696 participants with a self-reported, medically confirmed diagnosis of SLE from 30 countries between March 22, 2021, and May 17, 2021. The cohort was 96% women, and their median age was 42 (interquartile range, 34-51). Nearly 36% of respondents were from Italy, 27% were from Chile, 13% were from France, and just under 9% were Americans. All participants received at least one dose of COVID-19 vaccine, and 49% received a second dose. The most common vaccines were Pfizer-BioNTech (57%), Sinovac (22%), AstraZeneca (10%), and Moderna (8%).
Only 21 participants (3%) reported a medically confirmed SLE flare after a median of 3 days (IQR, 0-29) post COVID vaccination, with most experiencing musculoskeletal symptoms (90%) and fatigue (86%). Of the 21 cases, 15 reported a subsequent change in SLE treatment and 4 were admitted to the hospital. A previous flare that occurred within a year before vaccination was associated with an increased risk of flare post vaccination (relative risk, 5.52; 95% confidence interval, 2.17-14.03; P < .0001).
Side effects – including swelling, soreness, fever, chills, fatigue, joint and muscle pain, nausea, and headache – were reported in 45% of participants (n = 316) after their first dose and in 53% of the 343 participants who received a second dose. There was no notable difference in the likelihood of side effects across gender and age or in patients who received mRNA vaccines, compared with vaccines with other modes of action. Patients who reported side effects after the first dose were more likely to also report them after the second, compared with those who reported none (109 [81%] of 135 vs. 72 [35%] of 205; RR, 2.30; 95% CI, 1.88-2.82; P < .0001).
In the majority of cases (2,232 of 2,683), the side effects were of minor or moderate intensity and did not affect the participants’ ability to perform daily tasks. The study found no significant association between side effects and a SLE flare and SLE medications or previous SLE disease manifestations.
When asked to comment on the study, Amit Saxena, MD, of the Lupus Center at New York University Langone Health, said: “What we are seeing is pretty mild to moderate in terms of follow-up side effects or lupus-related activity. Several studies have shown this amongst our autoimmune rheumatology cohort, as well as what I’ve seen clinically in my own patients. More than anything else, numbers are the most important, and this is a large study.”
He acknowledged the benefits of going directly to patients to gauge their responses and reactions, giving them the opportunity to share concerns that physicians may not think about.
“As rheumatologists, we tend to focus on certain things that might not necessarily be what the patients themselves focus on,” he said. “I think the fact that this questionnaire dealt with a lot of what people complain about – fatigue, sore arm, things that we know are part of getting the vaccine – they aren’t necessarily things we capture with tools that screen for lupus flares, for example.”
More than anything, Dr. Saxena commended the study’s timeliness. “Patients are constantly asking us about the vaccine, and there’s so much misinformation,” he said. “People say, ‘Because I have lupus, I was told not to get vaccinated.’ I don’t know where they get that information from; we are telling everyone to get it, especially our lupus patients.”
The authors recognized their study’s main limitation as the self-reported and subjective nature of the survey, which they attempted to mitigate by asking for medically confirmed flares only. They noted, however, that the short median time between vaccination and flare onset could be caused by patients confusing expected side effects for something more serious, meaning the 3% figure “could be an overestimation of the actual flare rate.”
“Vaccination is recommended for patients with rheumatic and musculoskeletal diseases according to the American College of Rheumatology,” they added, “irrespective of disease activity and severity.”
Several authors reported potential conflicts of interest, including receiving consultancy fees and grants from Pfizer, GlaxoSmithKline, AbbVie, and Janssen, all unrelated to the study.
FROM THE LANCET RHEUMATOLOGY
AMA, 55 other groups urge health care vax mandate
As COVID-19 cases, hospitalizations, and deaths mount again across the country, the American Medical Association (AMA), the American Nursing Association, and 54 other
This injunction, issued July 26, covers everyone in healthcare, Emanuel Ezekiel, MD, PhD, chair of the department of medical ethics and health policy at the University of Pennsylvania, Philadelphia, and the organizer of the joint statement, said in an interview.
That includes not only hospitals, but also physician offices, ambulatory surgery centers, home care agencies, skilled nursing facilities, pharmacies, laboratories, and imaging centers, he said.
The exhortation to get vaccinated also extends to federal and state healthcare facilities, including those of the military health system — TRICARE and the Department of Veterans Affairs — which instituted a mandate the same day.
The American Hospital Association (AHA) and other hospital groups recently said they supported hospitals and health systems that required their personnel to get vaccinated. Several dozen healthcare organizations have already done so, including some of the nation’s largest health systems.
A substantial fraction of U.S. healthcare workers have not yet gotten vaccinated, although how many are unvaccinated is unclear. An analysis by WebMD and Medscape Medical News estimated that 25% of hospital workers who had contact with patients were unvaccinated at the end of May.
More than 38% of nursing workers were not fully vaccinated by July 11, according to an analysis of Centers for Medicare & Medicaid Services data by LeadingAge, which was cited by the Washington Post. And more than 40% of nursing home employees have not been fully vaccinated, according to the Centers for Disease Control and Prevention.
The joint statement did not give any indication of how many employees of physician practices have failed to get COVID shots. However, a recent AMA survey shows that 96% of physicians have been fully vaccinated.
Ethical commitment
The main reason for vaccine mandates, according to the healthcare associations’ statement, is “the ethical commitment to put patients as well as residents of long-term care facilities first and take all steps necessary to ensure their health and well-being.”
In addition, the statement noted, vaccination can protect healthcare workers and their families from getting COVID-19.
The statement also pointed out that many healthcare and long-term care organizations already require vaccinations for influenza, hepatitis B, and pertussis.
Workers who have certain medical conditions should be exempt from the vaccination mandates, the statement added.
While recognizing the “historical mistrust of health care institutions” among some healthcare workers, the statement said, “We must continue to address workers’ concerns, engage with marginalized populations, and work with trusted messengers to improve vaccine acceptance.”
There has been some skepticism about the legality of requiring healthcare workers to get vaccinated as a condition of employment, partly because the U.S. Food and Drug Administration has not yet fully authorized any of the COVID-19 vaccines.
But in June, a federal judge turned down a legal challenge to Houston Methodist’s vaccination mandate.
“It is critical that all people in the health care workforce get vaccinated against COVID-19 for the safety of our patients and our colleagues. With more than 300 million doses administered in the United States and nearly 4 billion doses administered worldwide, we know the vaccines are safe and highly effective at preventing severe illness and death from COVID-19.
“Increased vaccinations among health care personnel will not only reduce the spread of COVID-19 but also reduce the harmful toll this virus is taking within the health care workforce and those we are striving to serve,” Susan Bailey, MD, immediate past president of the AMA, said in a news release.
A version of this article first appeared on Medscape.com.
As COVID-19 cases, hospitalizations, and deaths mount again across the country, the American Medical Association (AMA), the American Nursing Association, and 54 other
This injunction, issued July 26, covers everyone in healthcare, Emanuel Ezekiel, MD, PhD, chair of the department of medical ethics and health policy at the University of Pennsylvania, Philadelphia, and the organizer of the joint statement, said in an interview.
That includes not only hospitals, but also physician offices, ambulatory surgery centers, home care agencies, skilled nursing facilities, pharmacies, laboratories, and imaging centers, he said.
The exhortation to get vaccinated also extends to federal and state healthcare facilities, including those of the military health system — TRICARE and the Department of Veterans Affairs — which instituted a mandate the same day.
The American Hospital Association (AHA) and other hospital groups recently said they supported hospitals and health systems that required their personnel to get vaccinated. Several dozen healthcare organizations have already done so, including some of the nation’s largest health systems.
A substantial fraction of U.S. healthcare workers have not yet gotten vaccinated, although how many are unvaccinated is unclear. An analysis by WebMD and Medscape Medical News estimated that 25% of hospital workers who had contact with patients were unvaccinated at the end of May.
More than 38% of nursing workers were not fully vaccinated by July 11, according to an analysis of Centers for Medicare & Medicaid Services data by LeadingAge, which was cited by the Washington Post. And more than 40% of nursing home employees have not been fully vaccinated, according to the Centers for Disease Control and Prevention.
The joint statement did not give any indication of how many employees of physician practices have failed to get COVID shots. However, a recent AMA survey shows that 96% of physicians have been fully vaccinated.
Ethical commitment
The main reason for vaccine mandates, according to the healthcare associations’ statement, is “the ethical commitment to put patients as well as residents of long-term care facilities first and take all steps necessary to ensure their health and well-being.”
In addition, the statement noted, vaccination can protect healthcare workers and their families from getting COVID-19.
The statement also pointed out that many healthcare and long-term care organizations already require vaccinations for influenza, hepatitis B, and pertussis.
Workers who have certain medical conditions should be exempt from the vaccination mandates, the statement added.
While recognizing the “historical mistrust of health care institutions” among some healthcare workers, the statement said, “We must continue to address workers’ concerns, engage with marginalized populations, and work with trusted messengers to improve vaccine acceptance.”
There has been some skepticism about the legality of requiring healthcare workers to get vaccinated as a condition of employment, partly because the U.S. Food and Drug Administration has not yet fully authorized any of the COVID-19 vaccines.
But in June, a federal judge turned down a legal challenge to Houston Methodist’s vaccination mandate.
“It is critical that all people in the health care workforce get vaccinated against COVID-19 for the safety of our patients and our colleagues. With more than 300 million doses administered in the United States and nearly 4 billion doses administered worldwide, we know the vaccines are safe and highly effective at preventing severe illness and death from COVID-19.
“Increased vaccinations among health care personnel will not only reduce the spread of COVID-19 but also reduce the harmful toll this virus is taking within the health care workforce and those we are striving to serve,” Susan Bailey, MD, immediate past president of the AMA, said in a news release.
A version of this article first appeared on Medscape.com.
As COVID-19 cases, hospitalizations, and deaths mount again across the country, the American Medical Association (AMA), the American Nursing Association, and 54 other
This injunction, issued July 26, covers everyone in healthcare, Emanuel Ezekiel, MD, PhD, chair of the department of medical ethics and health policy at the University of Pennsylvania, Philadelphia, and the organizer of the joint statement, said in an interview.
That includes not only hospitals, but also physician offices, ambulatory surgery centers, home care agencies, skilled nursing facilities, pharmacies, laboratories, and imaging centers, he said.
The exhortation to get vaccinated also extends to federal and state healthcare facilities, including those of the military health system — TRICARE and the Department of Veterans Affairs — which instituted a mandate the same day.
The American Hospital Association (AHA) and other hospital groups recently said they supported hospitals and health systems that required their personnel to get vaccinated. Several dozen healthcare organizations have already done so, including some of the nation’s largest health systems.
A substantial fraction of U.S. healthcare workers have not yet gotten vaccinated, although how many are unvaccinated is unclear. An analysis by WebMD and Medscape Medical News estimated that 25% of hospital workers who had contact with patients were unvaccinated at the end of May.
More than 38% of nursing workers were not fully vaccinated by July 11, according to an analysis of Centers for Medicare & Medicaid Services data by LeadingAge, which was cited by the Washington Post. And more than 40% of nursing home employees have not been fully vaccinated, according to the Centers for Disease Control and Prevention.
The joint statement did not give any indication of how many employees of physician practices have failed to get COVID shots. However, a recent AMA survey shows that 96% of physicians have been fully vaccinated.
Ethical commitment
The main reason for vaccine mandates, according to the healthcare associations’ statement, is “the ethical commitment to put patients as well as residents of long-term care facilities first and take all steps necessary to ensure their health and well-being.”
In addition, the statement noted, vaccination can protect healthcare workers and their families from getting COVID-19.
The statement also pointed out that many healthcare and long-term care organizations already require vaccinations for influenza, hepatitis B, and pertussis.
Workers who have certain medical conditions should be exempt from the vaccination mandates, the statement added.
While recognizing the “historical mistrust of health care institutions” among some healthcare workers, the statement said, “We must continue to address workers’ concerns, engage with marginalized populations, and work with trusted messengers to improve vaccine acceptance.”
There has been some skepticism about the legality of requiring healthcare workers to get vaccinated as a condition of employment, partly because the U.S. Food and Drug Administration has not yet fully authorized any of the COVID-19 vaccines.
But in June, a federal judge turned down a legal challenge to Houston Methodist’s vaccination mandate.
“It is critical that all people in the health care workforce get vaccinated against COVID-19 for the safety of our patients and our colleagues. With more than 300 million doses administered in the United States and nearly 4 billion doses administered worldwide, we know the vaccines are safe and highly effective at preventing severe illness and death from COVID-19.
“Increased vaccinations among health care personnel will not only reduce the spread of COVID-19 but also reduce the harmful toll this virus is taking within the health care workforce and those we are striving to serve,” Susan Bailey, MD, immediate past president of the AMA, said in a news release.
A version of this article first appeared on Medscape.com.
Vaccine breakthrough cases rising with Delta: Here’s what that means
At a recent town hall meeting in Cincinnati, President Joe Biden was asked about COVID-19 cases, hospitalizations, and deaths rising in response to the Delta variant.
Touting the importance of vaccination, “We have a pandemic for those who haven’t gotten a vaccination. It’s that basic, that simple,” President Biden said at the event, which was broadcast live on CNN.
“If you’re vaccinated, you’re not going to be hospitalized, not going to the ICU unit, and not going to die,” he said, adding “you’re not going to get COVID if you have these vaccinations.”
Unfortunately, it’s not so simple. Fully vaccinated people continue to be well protected against severe disease and death, even with Delta, Because of that, many experts continue to advise caution, even if fully vaccinated.
“I was disappointed,” Leana Wen, MD, MSc, an emergency physician and visiting professor of health policy and management at George Washington University’s Milken School of Public Health in Washington, told CNN in response to the president’s statement.
“I actually thought he was answering questions as if it were a month ago. He’s not really meeting the realities of what’s happening on the ground,” she said. “I think he may have led people astray.”
Vaccines still work
Recent cases support Dr. Wen’s claim. Fully vaccinated Olympic athletes, wedding guests, healthcare workers, and even White House staff have recently tested positive. So what gives?
The vast majority of these illnesses are mild, and public health officials say they are to be expected.
“The vaccines were designed to keep us out of the hospital and to keep us from dying. That was the whole purpose of the vaccine and they’re even more successful than we anticipated,” says William Schaffner, MD, an infectious disease expert at Vanderbilt University in Nashville.
As good as they are, these shots aren’t perfect. Their protection differs from person to person depending on age and underlying health. People with immune function that’s weakened because of age or a health condition can still become seriously ill, and, in very rare cases, die after vaccination.
When people are infected with Delta, they carry approximately 1,000 times more virus compared with previous versions of the virus, according to a recent study. All that virus can overwhelm even the strong protection from the vaccines.
“Three months ago, breakthroughs didn’t occur nearly at this rate because there was just so much less virus exposure in the community,” said Michael Osterholm, PhD, director of the Center for Infectious Disease Research and Policy at the University of Minnesota in Minneapolis.
Breakthroughs by the numbers
In Los Angeles County, where 69% of residents over age 12 have been fully vaccinated, COVID-19 cases are rising, and so, too, are cases that break through the protection of the vaccine.
In June, fully vaccinated people accounted for 20%, or 1 in 5, COVID cases in the county, which is the most populous in the United States. The increase mirrors Delta’s rise. The proportion of breakthrough cases is up from 11% in May, 5% in April, and 2% in March, according to the Los Angeles County Department of Public Health.
In the United Kingdom, which is collecting the best information on infections caused by variants, the estimated effectiveness of the vaccines to prevent an illness that causes symptoms dropped by about 10 points against Delta compared with Alpha (or B.1.1.7).
After two doses, vaccines prevent symptomatic infection about 79% of the time against Delta, according to data compiled by Public Health England. They are still highly effective at preventing hospitalization, 96% after two doses.
Out of 229,218 COVID infections in the United Kingdom between February and July 19, 28,773 — or 12.5% — were in fully vaccinated people. Of those breakthrough infections, 1,101, or 3.8%, required a visit to an emergency room, according to Public Health England. Just 474, or 2.9%, of fully vaccinated people required hospital admission, and 229, or less than 1%, died.
Unanswered questions
One of the biggest questions about breakthrough cases is how often people who have it may pass the virus to others.
“We know the vaccine reduces the likelihood of carrying the virus and the amount of virus you would carry,” Dr. Wen told CNN. But we don’t yet know whether a vaccinated person with a breakthrough infection may still be contagious to others.
For that reason, the Centers for Disease Control and Prevention says that fully vaccinated people still need to be tested if they have symptoms and shouldn’t be out in public for at least 10 days after a positive test.
How should fully vaccinated people behave? That depends a lot on their underlying health and whether or not they have vulnerable people around them.
If you’re older or immunocompromised, Dr. Schaffner recommends what he calls the “belt-and-suspenders approach,” in other words, do everything you can to stay safe.
“Get vaccinated for sure, but since we can’t be absolutely certain that the vaccines are going to be optimally protective and you are particularly susceptible to serious disease, you would be well advised to adopt at least one and perhaps more of the other mitigation measures,” he said.
These include wearing a mask, social distancing, making sure your spaces are well ventilated, and not spending prolonged periods of time indoors in crowded places.
Taking young children to visit vaccinated, elderly grandparents demands extra caution, again, with Delta circulating, particularly as they go back to school and start mixing with other kids.
Dr. Schaffner recommends explaining the ground rules before the visit: Hugs around the waist. No kissing. Wearing a mask while indoors with them.
Other important unanswered questions are whether breakthrough infections can lead to prolonged symptoms, or “long covid.” Most experts think that’s less likely in vaccinated people.
And Dr. Osterholm said it will be important to see whether there’s anything unusual about the breakthrough cases happening in the community.
“I think some of us have been challenged by the number of clusters that we’ve seen,” he said. “I think that really needs to be examined more.”
A version of this article first appeared on Medscape.com.
At a recent town hall meeting in Cincinnati, President Joe Biden was asked about COVID-19 cases, hospitalizations, and deaths rising in response to the Delta variant.
Touting the importance of vaccination, “We have a pandemic for those who haven’t gotten a vaccination. It’s that basic, that simple,” President Biden said at the event, which was broadcast live on CNN.
“If you’re vaccinated, you’re not going to be hospitalized, not going to the ICU unit, and not going to die,” he said, adding “you’re not going to get COVID if you have these vaccinations.”
Unfortunately, it’s not so simple. Fully vaccinated people continue to be well protected against severe disease and death, even with Delta, Because of that, many experts continue to advise caution, even if fully vaccinated.
“I was disappointed,” Leana Wen, MD, MSc, an emergency physician and visiting professor of health policy and management at George Washington University’s Milken School of Public Health in Washington, told CNN in response to the president’s statement.
“I actually thought he was answering questions as if it were a month ago. He’s not really meeting the realities of what’s happening on the ground,” she said. “I think he may have led people astray.”
Vaccines still work
Recent cases support Dr. Wen’s claim. Fully vaccinated Olympic athletes, wedding guests, healthcare workers, and even White House staff have recently tested positive. So what gives?
The vast majority of these illnesses are mild, and public health officials say they are to be expected.
“The vaccines were designed to keep us out of the hospital and to keep us from dying. That was the whole purpose of the vaccine and they’re even more successful than we anticipated,” says William Schaffner, MD, an infectious disease expert at Vanderbilt University in Nashville.
As good as they are, these shots aren’t perfect. Their protection differs from person to person depending on age and underlying health. People with immune function that’s weakened because of age or a health condition can still become seriously ill, and, in very rare cases, die after vaccination.
When people are infected with Delta, they carry approximately 1,000 times more virus compared with previous versions of the virus, according to a recent study. All that virus can overwhelm even the strong protection from the vaccines.
“Three months ago, breakthroughs didn’t occur nearly at this rate because there was just so much less virus exposure in the community,” said Michael Osterholm, PhD, director of the Center for Infectious Disease Research and Policy at the University of Minnesota in Minneapolis.
Breakthroughs by the numbers
In Los Angeles County, where 69% of residents over age 12 have been fully vaccinated, COVID-19 cases are rising, and so, too, are cases that break through the protection of the vaccine.
In June, fully vaccinated people accounted for 20%, or 1 in 5, COVID cases in the county, which is the most populous in the United States. The increase mirrors Delta’s rise. The proportion of breakthrough cases is up from 11% in May, 5% in April, and 2% in March, according to the Los Angeles County Department of Public Health.
In the United Kingdom, which is collecting the best information on infections caused by variants, the estimated effectiveness of the vaccines to prevent an illness that causes symptoms dropped by about 10 points against Delta compared with Alpha (or B.1.1.7).
After two doses, vaccines prevent symptomatic infection about 79% of the time against Delta, according to data compiled by Public Health England. They are still highly effective at preventing hospitalization, 96% after two doses.
Out of 229,218 COVID infections in the United Kingdom between February and July 19, 28,773 — or 12.5% — were in fully vaccinated people. Of those breakthrough infections, 1,101, or 3.8%, required a visit to an emergency room, according to Public Health England. Just 474, or 2.9%, of fully vaccinated people required hospital admission, and 229, or less than 1%, died.
Unanswered questions
One of the biggest questions about breakthrough cases is how often people who have it may pass the virus to others.
“We know the vaccine reduces the likelihood of carrying the virus and the amount of virus you would carry,” Dr. Wen told CNN. But we don’t yet know whether a vaccinated person with a breakthrough infection may still be contagious to others.
For that reason, the Centers for Disease Control and Prevention says that fully vaccinated people still need to be tested if they have symptoms and shouldn’t be out in public for at least 10 days after a positive test.
How should fully vaccinated people behave? That depends a lot on their underlying health and whether or not they have vulnerable people around them.
If you’re older or immunocompromised, Dr. Schaffner recommends what he calls the “belt-and-suspenders approach,” in other words, do everything you can to stay safe.
“Get vaccinated for sure, but since we can’t be absolutely certain that the vaccines are going to be optimally protective and you are particularly susceptible to serious disease, you would be well advised to adopt at least one and perhaps more of the other mitigation measures,” he said.
These include wearing a mask, social distancing, making sure your spaces are well ventilated, and not spending prolonged periods of time indoors in crowded places.
Taking young children to visit vaccinated, elderly grandparents demands extra caution, again, with Delta circulating, particularly as they go back to school and start mixing with other kids.
Dr. Schaffner recommends explaining the ground rules before the visit: Hugs around the waist. No kissing. Wearing a mask while indoors with them.
Other important unanswered questions are whether breakthrough infections can lead to prolonged symptoms, or “long covid.” Most experts think that’s less likely in vaccinated people.
And Dr. Osterholm said it will be important to see whether there’s anything unusual about the breakthrough cases happening in the community.
“I think some of us have been challenged by the number of clusters that we’ve seen,” he said. “I think that really needs to be examined more.”
A version of this article first appeared on Medscape.com.
At a recent town hall meeting in Cincinnati, President Joe Biden was asked about COVID-19 cases, hospitalizations, and deaths rising in response to the Delta variant.
Touting the importance of vaccination, “We have a pandemic for those who haven’t gotten a vaccination. It’s that basic, that simple,” President Biden said at the event, which was broadcast live on CNN.
“If you’re vaccinated, you’re not going to be hospitalized, not going to the ICU unit, and not going to die,” he said, adding “you’re not going to get COVID if you have these vaccinations.”
Unfortunately, it’s not so simple. Fully vaccinated people continue to be well protected against severe disease and death, even with Delta, Because of that, many experts continue to advise caution, even if fully vaccinated.
“I was disappointed,” Leana Wen, MD, MSc, an emergency physician and visiting professor of health policy and management at George Washington University’s Milken School of Public Health in Washington, told CNN in response to the president’s statement.
“I actually thought he was answering questions as if it were a month ago. He’s not really meeting the realities of what’s happening on the ground,” she said. “I think he may have led people astray.”
Vaccines still work
Recent cases support Dr. Wen’s claim. Fully vaccinated Olympic athletes, wedding guests, healthcare workers, and even White House staff have recently tested positive. So what gives?
The vast majority of these illnesses are mild, and public health officials say they are to be expected.
“The vaccines were designed to keep us out of the hospital and to keep us from dying. That was the whole purpose of the vaccine and they’re even more successful than we anticipated,” says William Schaffner, MD, an infectious disease expert at Vanderbilt University in Nashville.
As good as they are, these shots aren’t perfect. Their protection differs from person to person depending on age and underlying health. People with immune function that’s weakened because of age or a health condition can still become seriously ill, and, in very rare cases, die after vaccination.
When people are infected with Delta, they carry approximately 1,000 times more virus compared with previous versions of the virus, according to a recent study. All that virus can overwhelm even the strong protection from the vaccines.
“Three months ago, breakthroughs didn’t occur nearly at this rate because there was just so much less virus exposure in the community,” said Michael Osterholm, PhD, director of the Center for Infectious Disease Research and Policy at the University of Minnesota in Minneapolis.
Breakthroughs by the numbers
In Los Angeles County, where 69% of residents over age 12 have been fully vaccinated, COVID-19 cases are rising, and so, too, are cases that break through the protection of the vaccine.
In June, fully vaccinated people accounted for 20%, or 1 in 5, COVID cases in the county, which is the most populous in the United States. The increase mirrors Delta’s rise. The proportion of breakthrough cases is up from 11% in May, 5% in April, and 2% in March, according to the Los Angeles County Department of Public Health.
In the United Kingdom, which is collecting the best information on infections caused by variants, the estimated effectiveness of the vaccines to prevent an illness that causes symptoms dropped by about 10 points against Delta compared with Alpha (or B.1.1.7).
After two doses, vaccines prevent symptomatic infection about 79% of the time against Delta, according to data compiled by Public Health England. They are still highly effective at preventing hospitalization, 96% after two doses.
Out of 229,218 COVID infections in the United Kingdom between February and July 19, 28,773 — or 12.5% — were in fully vaccinated people. Of those breakthrough infections, 1,101, or 3.8%, required a visit to an emergency room, according to Public Health England. Just 474, or 2.9%, of fully vaccinated people required hospital admission, and 229, or less than 1%, died.
Unanswered questions
One of the biggest questions about breakthrough cases is how often people who have it may pass the virus to others.
“We know the vaccine reduces the likelihood of carrying the virus and the amount of virus you would carry,” Dr. Wen told CNN. But we don’t yet know whether a vaccinated person with a breakthrough infection may still be contagious to others.
For that reason, the Centers for Disease Control and Prevention says that fully vaccinated people still need to be tested if they have symptoms and shouldn’t be out in public for at least 10 days after a positive test.
How should fully vaccinated people behave? That depends a lot on their underlying health and whether or not they have vulnerable people around them.
If you’re older or immunocompromised, Dr. Schaffner recommends what he calls the “belt-and-suspenders approach,” in other words, do everything you can to stay safe.
“Get vaccinated for sure, but since we can’t be absolutely certain that the vaccines are going to be optimally protective and you are particularly susceptible to serious disease, you would be well advised to adopt at least one and perhaps more of the other mitigation measures,” he said.
These include wearing a mask, social distancing, making sure your spaces are well ventilated, and not spending prolonged periods of time indoors in crowded places.
Taking young children to visit vaccinated, elderly grandparents demands extra caution, again, with Delta circulating, particularly as they go back to school and start mixing with other kids.
Dr. Schaffner recommends explaining the ground rules before the visit: Hugs around the waist. No kissing. Wearing a mask while indoors with them.
Other important unanswered questions are whether breakthrough infections can lead to prolonged symptoms, or “long covid.” Most experts think that’s less likely in vaccinated people.
And Dr. Osterholm said it will be important to see whether there’s anything unusual about the breakthrough cases happening in the community.
“I think some of us have been challenged by the number of clusters that we’ve seen,” he said. “I think that really needs to be examined more.”
A version of this article first appeared on Medscape.com.