User login
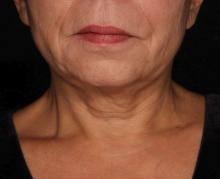
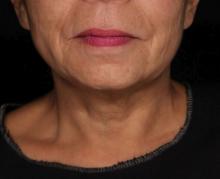
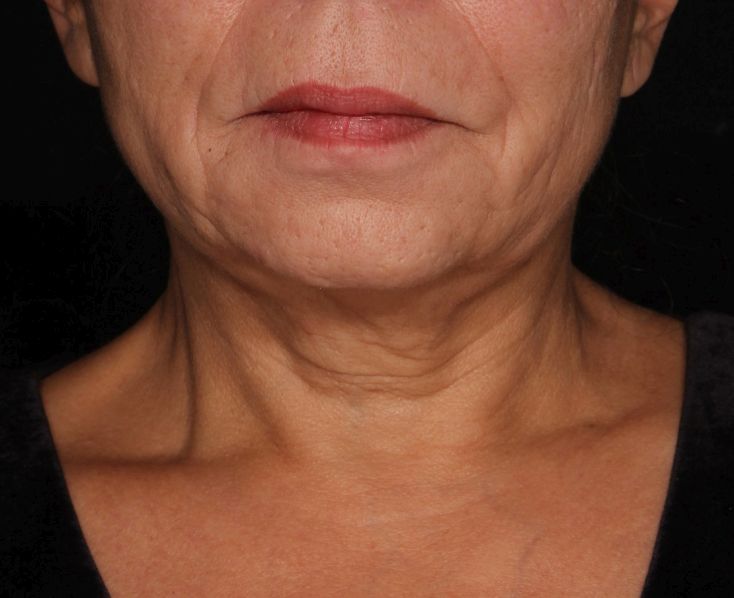
Picosecond 755-nm laser found effective for neck rejuvenation
DALLAS –
“It’s important to note that response was variable, but many patients were satisfied with the treatment,” Hana Jeon, MD said at the annual conference of the American Society for Laser Medicine and Surgery, Inc. “Further studies are needed to identify the clinical characteristics of neck laxity that would most benefit from this treatment.”
The researchers enrolled 25 patients with an average age of 58 years. The laser treatment settings were a 6-mm spot side-delivered at a fluence of 0.71 J/cm2 in a pulse width of 750 picoseconds. The patients were treated five times on the neck every 2-4 weeks, and follow-up visits were scheduled for 1 month and 3 months after the last treatment. Digital photos were taken at each visit. Formal assessment tools included patient and physician satisfaction scores and the Global Aesthetic Improvement Scale. In all, 21 women and 3 men completed the study. The majority (72%) had Fitzpatrick skin type II, while 16% had type III, 8% had type I, and 4% had type IV. An average of 5,042 pulses were delivered during each treatment session. The majority of patients (84%) required no anesthesia, while the rest used topical numbing medicine from 30 minutes to an hour prior to the procedure.
Dr. Jeon reported that the average pain score during the procedure was 4.7 on a 10-point scale. Forced-air cooling was used for comfort, and on average, mild redness following the treatment lasted less than 1 day (a mean of 0.6 days, with a range of 0-5 days). Mild pain also lasted less than 1 day (a mean of 0.1 days, with a range of 0-2 days). No swelling, crusting, bruising, bleeding, infection, blistering, scarring, burn, or dyspigmentation occurred.
On the Global Aesthetic Improvement Scale at 1 and 3 months, physicians described 43% and 23% of cases, respectively, as “improved,” 17% and 18% of cases as “much improved,” and 4% and 9% of cases as “extremely improved.”
At 3 months, 35% of patients said they would be “somewhat likely” to recommend the procedure, and 30% said they would be “extremely likely” to recommend it.
Dr. Jeon reported having no financial disclosures.
DALLAS –
“It’s important to note that response was variable, but many patients were satisfied with the treatment,” Hana Jeon, MD said at the annual conference of the American Society for Laser Medicine and Surgery, Inc. “Further studies are needed to identify the clinical characteristics of neck laxity that would most benefit from this treatment.”
The researchers enrolled 25 patients with an average age of 58 years. The laser treatment settings were a 6-mm spot side-delivered at a fluence of 0.71 J/cm2 in a pulse width of 750 picoseconds. The patients were treated five times on the neck every 2-4 weeks, and follow-up visits were scheduled for 1 month and 3 months after the last treatment. Digital photos were taken at each visit. Formal assessment tools included patient and physician satisfaction scores and the Global Aesthetic Improvement Scale. In all, 21 women and 3 men completed the study. The majority (72%) had Fitzpatrick skin type II, while 16% had type III, 8% had type I, and 4% had type IV. An average of 5,042 pulses were delivered during each treatment session. The majority of patients (84%) required no anesthesia, while the rest used topical numbing medicine from 30 minutes to an hour prior to the procedure.
Dr. Jeon reported that the average pain score during the procedure was 4.7 on a 10-point scale. Forced-air cooling was used for comfort, and on average, mild redness following the treatment lasted less than 1 day (a mean of 0.6 days, with a range of 0-5 days). Mild pain also lasted less than 1 day (a mean of 0.1 days, with a range of 0-2 days). No swelling, crusting, bruising, bleeding, infection, blistering, scarring, burn, or dyspigmentation occurred.
On the Global Aesthetic Improvement Scale at 1 and 3 months, physicians described 43% and 23% of cases, respectively, as “improved,” 17% and 18% of cases as “much improved,” and 4% and 9% of cases as “extremely improved.”
At 3 months, 35% of patients said they would be “somewhat likely” to recommend the procedure, and 30% said they would be “extremely likely” to recommend it.
Dr. Jeon reported having no financial disclosures.
DALLAS –
“It’s important to note that response was variable, but many patients were satisfied with the treatment,” Hana Jeon, MD said at the annual conference of the American Society for Laser Medicine and Surgery, Inc. “Further studies are needed to identify the clinical characteristics of neck laxity that would most benefit from this treatment.”
The researchers enrolled 25 patients with an average age of 58 years. The laser treatment settings were a 6-mm spot side-delivered at a fluence of 0.71 J/cm2 in a pulse width of 750 picoseconds. The patients were treated five times on the neck every 2-4 weeks, and follow-up visits were scheduled for 1 month and 3 months after the last treatment. Digital photos were taken at each visit. Formal assessment tools included patient and physician satisfaction scores and the Global Aesthetic Improvement Scale. In all, 21 women and 3 men completed the study. The majority (72%) had Fitzpatrick skin type II, while 16% had type III, 8% had type I, and 4% had type IV. An average of 5,042 pulses were delivered during each treatment session. The majority of patients (84%) required no anesthesia, while the rest used topical numbing medicine from 30 minutes to an hour prior to the procedure.
Dr. Jeon reported that the average pain score during the procedure was 4.7 on a 10-point scale. Forced-air cooling was used for comfort, and on average, mild redness following the treatment lasted less than 1 day (a mean of 0.6 days, with a range of 0-5 days). Mild pain also lasted less than 1 day (a mean of 0.1 days, with a range of 0-2 days). No swelling, crusting, bruising, bleeding, infection, blistering, scarring, burn, or dyspigmentation occurred.
On the Global Aesthetic Improvement Scale at 1 and 3 months, physicians described 43% and 23% of cases, respectively, as “improved,” 17% and 18% of cases as “much improved,” and 4% and 9% of cases as “extremely improved.”
At 3 months, 35% of patients said they would be “somewhat likely” to recommend the procedure, and 30% said they would be “extremely likely” to recommend it.
Dr. Jeon reported having no financial disclosures.
REPORTING FROM ASLMS 2018
Key clinical point: Response to using a picosecond 755-nm laser with focus lens array for neck rejuvenation was variable.
Major finding: On the Global Aesthetic Improvement Scale at 1 and 3 months, physicians described 43% and 23% of cases, respectively, as “improved.”
Study details: A single-center study of 25 patients treated for neck laxity.
Disclosures: Dr. Jeon reported having no financial disclosures.
Noninvasive 1060-nm diode laser found effective for axillary hyperhidrosis
DALLAS – , a condition that affects an estimated 1.4% people in the United States.
“Newer treatments such as microwave technology, botulinum toxin injections, and lasers have emerged as effective methods,” lead study author Bruce E. Katz, MD, who directs the Juva Skin & Laser Center in New York, said at the annual conference of the American Society for Laser Medicine and Surgery Inc.
In a prospective pilot study, Dr. Katz and his associates examined the use of a noninvasive 1060-nm diode laser (SculpSure, Cynosure) in 10 subjects with moderate to severe hyperhidrosis of the axilla, which was defined as having Hyperhidrosis Disease Severity Scale (HDSS) scores of 3 or 4. The subjects, whose mean age was 42 years, received two laser treatments on the axillary area at 6-week intervals, and the researchers performed starch iodine tests and took two-dimensional photography of the axilla before and after treatment. Subjects were asked to rate their conditions on the HDSS and satisfaction with treatment. Two blinded dermatologist evaluators graded the reduction in sweat production on photos of starch iodine tests. The researchers also performed a retrospective analysis on all subjects to assess safety and efficacy of treatment.
The 1060-nm diode laser used for the study features four flat, nonsuction applicators. Dr. Katz and his associates positioned two applicators on the axilla simultaneously for 25 minutes. “The 1060-nm wavelength has minimal absorption in the melanin, so we can really treat any skin type,” he said. “It has a high affinity for adipose tissue, and we believe that its targeted effect at the level of the eccrine and apocrine glands should help improve hyperhidrosis. It works by elevating the tissue temperature to about 42 to 47 degrees Centigrade, without damaging surrounding tissue. The device has a sapphire cooling plate, so it’s comfortable for the patient.”
The retrospective analysis revealed minimal adverse events such as pain, mild erythema, and edema, which were transient and resolved without intervention in a day or two. To date, no cases have recurred, “which we were surprised by,” Dr. Katz said. “We were surprised that the effect lasted that long.”
SOURCE: Katz et. al. ASLMS 2018.
DALLAS – , a condition that affects an estimated 1.4% people in the United States.
“Newer treatments such as microwave technology, botulinum toxin injections, and lasers have emerged as effective methods,” lead study author Bruce E. Katz, MD, who directs the Juva Skin & Laser Center in New York, said at the annual conference of the American Society for Laser Medicine and Surgery Inc.
In a prospective pilot study, Dr. Katz and his associates examined the use of a noninvasive 1060-nm diode laser (SculpSure, Cynosure) in 10 subjects with moderate to severe hyperhidrosis of the axilla, which was defined as having Hyperhidrosis Disease Severity Scale (HDSS) scores of 3 or 4. The subjects, whose mean age was 42 years, received two laser treatments on the axillary area at 6-week intervals, and the researchers performed starch iodine tests and took two-dimensional photography of the axilla before and after treatment. Subjects were asked to rate their conditions on the HDSS and satisfaction with treatment. Two blinded dermatologist evaluators graded the reduction in sweat production on photos of starch iodine tests. The researchers also performed a retrospective analysis on all subjects to assess safety and efficacy of treatment.
The 1060-nm diode laser used for the study features four flat, nonsuction applicators. Dr. Katz and his associates positioned two applicators on the axilla simultaneously for 25 minutes. “The 1060-nm wavelength has minimal absorption in the melanin, so we can really treat any skin type,” he said. “It has a high affinity for adipose tissue, and we believe that its targeted effect at the level of the eccrine and apocrine glands should help improve hyperhidrosis. It works by elevating the tissue temperature to about 42 to 47 degrees Centigrade, without damaging surrounding tissue. The device has a sapphire cooling plate, so it’s comfortable for the patient.”
The retrospective analysis revealed minimal adverse events such as pain, mild erythema, and edema, which were transient and resolved without intervention in a day or two. To date, no cases have recurred, “which we were surprised by,” Dr. Katz said. “We were surprised that the effect lasted that long.”
SOURCE: Katz et. al. ASLMS 2018.
DALLAS – , a condition that affects an estimated 1.4% people in the United States.
“Newer treatments such as microwave technology, botulinum toxin injections, and lasers have emerged as effective methods,” lead study author Bruce E. Katz, MD, who directs the Juva Skin & Laser Center in New York, said at the annual conference of the American Society for Laser Medicine and Surgery Inc.
In a prospective pilot study, Dr. Katz and his associates examined the use of a noninvasive 1060-nm diode laser (SculpSure, Cynosure) in 10 subjects with moderate to severe hyperhidrosis of the axilla, which was defined as having Hyperhidrosis Disease Severity Scale (HDSS) scores of 3 or 4. The subjects, whose mean age was 42 years, received two laser treatments on the axillary area at 6-week intervals, and the researchers performed starch iodine tests and took two-dimensional photography of the axilla before and after treatment. Subjects were asked to rate their conditions on the HDSS and satisfaction with treatment. Two blinded dermatologist evaluators graded the reduction in sweat production on photos of starch iodine tests. The researchers also performed a retrospective analysis on all subjects to assess safety and efficacy of treatment.
The 1060-nm diode laser used for the study features four flat, nonsuction applicators. Dr. Katz and his associates positioned two applicators on the axilla simultaneously for 25 minutes. “The 1060-nm wavelength has minimal absorption in the melanin, so we can really treat any skin type,” he said. “It has a high affinity for adipose tissue, and we believe that its targeted effect at the level of the eccrine and apocrine glands should help improve hyperhidrosis. It works by elevating the tissue temperature to about 42 to 47 degrees Centigrade, without damaging surrounding tissue. The device has a sapphire cooling plate, so it’s comfortable for the patient.”
The retrospective analysis revealed minimal adverse events such as pain, mild erythema, and edema, which were transient and resolved without intervention in a day or two. To date, no cases have recurred, “which we were surprised by,” Dr. Katz said. “We were surprised that the effect lasted that long.”
SOURCE: Katz et. al. ASLMS 2018.
REPORTING FROM ASLMS 2018
Skin rollers
, but only a few actually have any scientific data or clinical studies supporting their claims. In general, these rollers promise to increase collagen, depuff the skin, lift and firm, increase circulation, increase oxygenation, and decrease inflammation. But no clinically significant results have been reported with most of these over-the-counter devices. Furthermore, not every roller is meant for every skin type – and some should stay within the hands of an experienced professional.
Ice rollers have been used for many years and are very effective to cool the skin for in-office procedures. They are drum-shaped stainless steel rollers that are left in the freezer and cool the epidermis upon application. At-home ice rollers cause immediate vasoconstriction and are a quick fix for periorbital edema or skin erythema. Three-dimensional roller face massagers are simply a massage tool and can be used on any skin type to increase facial circulation; they do not provide any visible clinical benefits. Nanocurrent or vibrating rollers use nanocurrents and vibration alongside a conductor gel to glide across the skin; they massage the skin and help topically applied agents penetrate into the stratum corneum.
Dr. Talakoub and Dr. Wesley are cocontributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Write to them at [email protected]. They had no relevant disclosures.
References
Orentreich DS et al. Dermatol Surg. 1995;21(6):543-9.
Aust MC et al. Plast Reconstr Surg. 2008;21(4):1421-9.Fernandes D et al. Clin Dermatol. 2008 Mar-Apr;26(2):192-9.
Nair PA et al. GMJ. 2014;69:24-7.
, but only a few actually have any scientific data or clinical studies supporting their claims. In general, these rollers promise to increase collagen, depuff the skin, lift and firm, increase circulation, increase oxygenation, and decrease inflammation. But no clinically significant results have been reported with most of these over-the-counter devices. Furthermore, not every roller is meant for every skin type – and some should stay within the hands of an experienced professional.
Ice rollers have been used for many years and are very effective to cool the skin for in-office procedures. They are drum-shaped stainless steel rollers that are left in the freezer and cool the epidermis upon application. At-home ice rollers cause immediate vasoconstriction and are a quick fix for periorbital edema or skin erythema. Three-dimensional roller face massagers are simply a massage tool and can be used on any skin type to increase facial circulation; they do not provide any visible clinical benefits. Nanocurrent or vibrating rollers use nanocurrents and vibration alongside a conductor gel to glide across the skin; they massage the skin and help topically applied agents penetrate into the stratum corneum.
Dr. Talakoub and Dr. Wesley are cocontributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Write to them at [email protected]. They had no relevant disclosures.
References
Orentreich DS et al. Dermatol Surg. 1995;21(6):543-9.
Aust MC et al. Plast Reconstr Surg. 2008;21(4):1421-9.Fernandes D et al. Clin Dermatol. 2008 Mar-Apr;26(2):192-9.
Nair PA et al. GMJ. 2014;69:24-7.
, but only a few actually have any scientific data or clinical studies supporting their claims. In general, these rollers promise to increase collagen, depuff the skin, lift and firm, increase circulation, increase oxygenation, and decrease inflammation. But no clinically significant results have been reported with most of these over-the-counter devices. Furthermore, not every roller is meant for every skin type – and some should stay within the hands of an experienced professional.
Ice rollers have been used for many years and are very effective to cool the skin for in-office procedures. They are drum-shaped stainless steel rollers that are left in the freezer and cool the epidermis upon application. At-home ice rollers cause immediate vasoconstriction and are a quick fix for periorbital edema or skin erythema. Three-dimensional roller face massagers are simply a massage tool and can be used on any skin type to increase facial circulation; they do not provide any visible clinical benefits. Nanocurrent or vibrating rollers use nanocurrents and vibration alongside a conductor gel to glide across the skin; they massage the skin and help topically applied agents penetrate into the stratum corneum.
Dr. Talakoub and Dr. Wesley are cocontributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Write to them at [email protected]. They had no relevant disclosures.
References
Orentreich DS et al. Dermatol Surg. 1995;21(6):543-9.
Aust MC et al. Plast Reconstr Surg. 2008;21(4):1421-9.Fernandes D et al. Clin Dermatol. 2008 Mar-Apr;26(2):192-9.
Nair PA et al. GMJ. 2014;69:24-7.
Hyaluronic acid filler preferred for infraorbital hollowing
Most patients who responded to surveys reported being satisfied after off-label treatment with Juvéderm Voluma XC hyaluronic acid filler for infraorbital hollowing, a study finds.
Adverse effects were reported in 12% of patients.
The treatment’s “high patient satisfaction profile and a similar safety profile among other soft-tissue fillers make it an excellent adjunct in the plastic surgeon’s armamentarium,” reported Michael B. Hall, MD, and his associates at their private, ambulatory facial plastic and reconstructive surgery practice in Austin, Texas, in JAMA Facial Plastic Surgery.
According to the researchers, the Food and Drug Administration has not approved any soft-tissue fillers for the periorbital complex. At their practice, Dr. Hall and his associates treat infraorbital hollows with Juvéderm Voluma XC, which is approved by the FDA for certain types of cheek augmentation. Other studies have examined Belotero or Restylane as treatments for building volume in the periorbital area, the authors wrote, but research into cosmetic injections of Juvéderm Voluma XC is lacking.
For the new study, the researchers retrospectively analyzed the cases of 101 patients (aged 32-75 years, with average age of 54 years; 89% female; 54% Fitzpatrick skin type II; racial breakdown not reported) who were electively treated with the filler for infraorbital hollowing in 2016 and 2017. The patients received an average 1 mL of the treatment gel.
The patients were photographed and answered surveys, and they were evaluated using the Allergan Infraorbital Hollows Scale. Follow-up time averaged 12 months.
A total of 18 patients (18%) required touch-up within 3 months, and 2 required multiple touch-ups. A total of 12 subjects (12%) had adverse effects (including 3 who had more than one), which included bruising (10%), contour irregularities (2%), edema (3%) and Tyndall effect (1%). Hyaluronidase was required in 3 patients (3%), and 24 patients sought further treatment after 3 months.
The researchers sent two satisfaction surveys to the participants. A total of 41% responded to the FACE-Q Satisfaction With Eyes survey, and 42% responded to the FACE-Q Satisfaction With Decision survey.
Depending on the question, 70%-85% of the respondents to the Satisfaction With Eyes survey said they were “definitely” or “somewhat” satisfied with the treatment outcome.
The highest levels of dissatisfaction came in response to a questions about whether the subjects felt their eyes looked alert (not tired) or youthful. The highest levels of satisfaction were in response to questions about whether the subjects were happy with the shape, attractiveness, and openness of their eyes.
Depending on the question, 73%-85% of the subjects who took the Satisfaction With Decision survey reported that they “definitely” or “somewhat” agree with positive statements about the treatment. While differences were small, they agreed the most with a statement saying the procedure was “worth the time and effort.”
No external funding or remuneration was received. The study authors reported no relevant disclosures.
SOURCE: Hall MB et al. JAMA Facial Plast Surg. 2018 Apr 5. doi:10.1001/jamafacial.2018.0230.
Most patients who responded to surveys reported being satisfied after off-label treatment with Juvéderm Voluma XC hyaluronic acid filler for infraorbital hollowing, a study finds.
Adverse effects were reported in 12% of patients.
The treatment’s “high patient satisfaction profile and a similar safety profile among other soft-tissue fillers make it an excellent adjunct in the plastic surgeon’s armamentarium,” reported Michael B. Hall, MD, and his associates at their private, ambulatory facial plastic and reconstructive surgery practice in Austin, Texas, in JAMA Facial Plastic Surgery.
According to the researchers, the Food and Drug Administration has not approved any soft-tissue fillers for the periorbital complex. At their practice, Dr. Hall and his associates treat infraorbital hollows with Juvéderm Voluma XC, which is approved by the FDA for certain types of cheek augmentation. Other studies have examined Belotero or Restylane as treatments for building volume in the periorbital area, the authors wrote, but research into cosmetic injections of Juvéderm Voluma XC is lacking.
For the new study, the researchers retrospectively analyzed the cases of 101 patients (aged 32-75 years, with average age of 54 years; 89% female; 54% Fitzpatrick skin type II; racial breakdown not reported) who were electively treated with the filler for infraorbital hollowing in 2016 and 2017. The patients received an average 1 mL of the treatment gel.
The patients were photographed and answered surveys, and they were evaluated using the Allergan Infraorbital Hollows Scale. Follow-up time averaged 12 months.
A total of 18 patients (18%) required touch-up within 3 months, and 2 required multiple touch-ups. A total of 12 subjects (12%) had adverse effects (including 3 who had more than one), which included bruising (10%), contour irregularities (2%), edema (3%) and Tyndall effect (1%). Hyaluronidase was required in 3 patients (3%), and 24 patients sought further treatment after 3 months.
The researchers sent two satisfaction surveys to the participants. A total of 41% responded to the FACE-Q Satisfaction With Eyes survey, and 42% responded to the FACE-Q Satisfaction With Decision survey.
Depending on the question, 70%-85% of the respondents to the Satisfaction With Eyes survey said they were “definitely” or “somewhat” satisfied with the treatment outcome.
The highest levels of dissatisfaction came in response to a questions about whether the subjects felt their eyes looked alert (not tired) or youthful. The highest levels of satisfaction were in response to questions about whether the subjects were happy with the shape, attractiveness, and openness of their eyes.
Depending on the question, 73%-85% of the subjects who took the Satisfaction With Decision survey reported that they “definitely” or “somewhat” agree with positive statements about the treatment. While differences were small, they agreed the most with a statement saying the procedure was “worth the time and effort.”
No external funding or remuneration was received. The study authors reported no relevant disclosures.
SOURCE: Hall MB et al. JAMA Facial Plast Surg. 2018 Apr 5. doi:10.1001/jamafacial.2018.0230.
Most patients who responded to surveys reported being satisfied after off-label treatment with Juvéderm Voluma XC hyaluronic acid filler for infraorbital hollowing, a study finds.
Adverse effects were reported in 12% of patients.
The treatment’s “high patient satisfaction profile and a similar safety profile among other soft-tissue fillers make it an excellent adjunct in the plastic surgeon’s armamentarium,” reported Michael B. Hall, MD, and his associates at their private, ambulatory facial plastic and reconstructive surgery practice in Austin, Texas, in JAMA Facial Plastic Surgery.
According to the researchers, the Food and Drug Administration has not approved any soft-tissue fillers for the periorbital complex. At their practice, Dr. Hall and his associates treat infraorbital hollows with Juvéderm Voluma XC, which is approved by the FDA for certain types of cheek augmentation. Other studies have examined Belotero or Restylane as treatments for building volume in the periorbital area, the authors wrote, but research into cosmetic injections of Juvéderm Voluma XC is lacking.
For the new study, the researchers retrospectively analyzed the cases of 101 patients (aged 32-75 years, with average age of 54 years; 89% female; 54% Fitzpatrick skin type II; racial breakdown not reported) who were electively treated with the filler for infraorbital hollowing in 2016 and 2017. The patients received an average 1 mL of the treatment gel.
The patients were photographed and answered surveys, and they were evaluated using the Allergan Infraorbital Hollows Scale. Follow-up time averaged 12 months.
A total of 18 patients (18%) required touch-up within 3 months, and 2 required multiple touch-ups. A total of 12 subjects (12%) had adverse effects (including 3 who had more than one), which included bruising (10%), contour irregularities (2%), edema (3%) and Tyndall effect (1%). Hyaluronidase was required in 3 patients (3%), and 24 patients sought further treatment after 3 months.
The researchers sent two satisfaction surveys to the participants. A total of 41% responded to the FACE-Q Satisfaction With Eyes survey, and 42% responded to the FACE-Q Satisfaction With Decision survey.
Depending on the question, 70%-85% of the respondents to the Satisfaction With Eyes survey said they were “definitely” or “somewhat” satisfied with the treatment outcome.
The highest levels of dissatisfaction came in response to a questions about whether the subjects felt their eyes looked alert (not tired) or youthful. The highest levels of satisfaction were in response to questions about whether the subjects were happy with the shape, attractiveness, and openness of their eyes.
Depending on the question, 73%-85% of the subjects who took the Satisfaction With Decision survey reported that they “definitely” or “somewhat” agree with positive statements about the treatment. While differences were small, they agreed the most with a statement saying the procedure was “worth the time and effort.”
No external funding or remuneration was received. The study authors reported no relevant disclosures.
SOURCE: Hall MB et al. JAMA Facial Plast Surg. 2018 Apr 5. doi:10.1001/jamafacial.2018.0230.
FROM JAMA FACIAL PLASTIC SURGERY
Key clinical point:
Major finding: Adverse effects occurred at a rate of 12%, and most who responded to surveys reported satisfaction postprocedure (70%-85%).
Study details: A retrospective observational study of 101 patients.
Disclosures: No external funding or remuneration was received. The study authors reported no relevant disclosures.
Source: Hall MB et al. JAMA Facial Plast Surg. 2018 Apr 5. doi:10.1001/jamafacial.2018.0230.
Few physicians report adverse events related to body contouring devices
DALLAS – Most physicians do not report adverse events related to noninvasive body contouring devices to the Food and Drug Administration, a database review showed.
“The FDA mandates that manufacturers and device operators disclose medical device reports to monitor suspected injuries and device malfunctions to the Manufacturer and User Facility Device Experience [MAUDE] database,” study author Adam J. Wulkan, MD, said at the annual conference of the American Society for Laser Medicine and Surgery. “Given the rapid growth in the noninvasive fat reduction market, it is essential physicians be aware of associated adverse events.”
“Given that this is the largest database for adverse events, I was surprised how few reports were done by physicians,” Dr. Wulkan said in an interview.
He reported results from 83 of the 98 MDRs. A total of 26 MDRs reported on cryolipolysis and included newly diagnosed or exacerbation of prior umbilical hernias (10), neuropathies (5), paradoxical fat hyperplasias (4), blisters (3), rashes (1), flu-like symptoms (1), gastroenteritis (1), and prolapsed bladder/uterus/rectum (1). Of the 11 MDRs for radio-frequency devices, 10 were burns/blisters and 1 fire.
Dr. Wulkan went on to note that 17 MDRs were reported for 1060-nm laser lipolysis, including burns/blisters (5), nodules (4), pain (3), cellulitis/abscesses (2), excessive swelling (1), neuropathy (1), and vomiting (1). There were four cases of burns on MDRs from focused/pulsed ultrasound procedures. One was on a non–FDA approved body site and another after combination radio-frequency/infrared treatment for cellulite reduction.
There were eight MDRs reported with 1440-nm laser cellulite reduction, including burn (5), unacceptable cosmesis (2), and faulty power supply (1). In addition, there were 12 MDRs reported on high-frequency ultrasound, including burns/blisters (9), subcutaneous nodules (2), and excoriation (1). Finally, vacuum-assisted submission was associated with five MDRs, including festooning/unacceptable cosmesis (2), seroma/hematoma (1), laceration (1), and nodules (1).
Dr. Wulkan acknowledged certain limitations of the study, including the fact that not all MDRs are reported to MAUDE, and that the reliability of patient reporting can be questionable. “We have to figure out if there is a natural causation between the adverse event and the treatment itself,” he said at the meeting. “The number of MDRs for any given device must be correlated to the number of procedures performed before we jump to conclusions about the safety of any given device.”
He reported having no financial disclosures. One study coauthor holds positions on advisory boards of, is a consultant for, and has intellectual property and/or stock options with various industry companies, such as Soliton and Allergan.
SOURCE: Wulkan et. al., ASLMS 2018.
DALLAS – Most physicians do not report adverse events related to noninvasive body contouring devices to the Food and Drug Administration, a database review showed.
“The FDA mandates that manufacturers and device operators disclose medical device reports to monitor suspected injuries and device malfunctions to the Manufacturer and User Facility Device Experience [MAUDE] database,” study author Adam J. Wulkan, MD, said at the annual conference of the American Society for Laser Medicine and Surgery. “Given the rapid growth in the noninvasive fat reduction market, it is essential physicians be aware of associated adverse events.”
“Given that this is the largest database for adverse events, I was surprised how few reports were done by physicians,” Dr. Wulkan said in an interview.
He reported results from 83 of the 98 MDRs. A total of 26 MDRs reported on cryolipolysis and included newly diagnosed or exacerbation of prior umbilical hernias (10), neuropathies (5), paradoxical fat hyperplasias (4), blisters (3), rashes (1), flu-like symptoms (1), gastroenteritis (1), and prolapsed bladder/uterus/rectum (1). Of the 11 MDRs for radio-frequency devices, 10 were burns/blisters and 1 fire.
Dr. Wulkan went on to note that 17 MDRs were reported for 1060-nm laser lipolysis, including burns/blisters (5), nodules (4), pain (3), cellulitis/abscesses (2), excessive swelling (1), neuropathy (1), and vomiting (1). There were four cases of burns on MDRs from focused/pulsed ultrasound procedures. One was on a non–FDA approved body site and another after combination radio-frequency/infrared treatment for cellulite reduction.
There were eight MDRs reported with 1440-nm laser cellulite reduction, including burn (5), unacceptable cosmesis (2), and faulty power supply (1). In addition, there were 12 MDRs reported on high-frequency ultrasound, including burns/blisters (9), subcutaneous nodules (2), and excoriation (1). Finally, vacuum-assisted submission was associated with five MDRs, including festooning/unacceptable cosmesis (2), seroma/hematoma (1), laceration (1), and nodules (1).
Dr. Wulkan acknowledged certain limitations of the study, including the fact that not all MDRs are reported to MAUDE, and that the reliability of patient reporting can be questionable. “We have to figure out if there is a natural causation between the adverse event and the treatment itself,” he said at the meeting. “The number of MDRs for any given device must be correlated to the number of procedures performed before we jump to conclusions about the safety of any given device.”
He reported having no financial disclosures. One study coauthor holds positions on advisory boards of, is a consultant for, and has intellectual property and/or stock options with various industry companies, such as Soliton and Allergan.
SOURCE: Wulkan et. al., ASLMS 2018.
DALLAS – Most physicians do not report adverse events related to noninvasive body contouring devices to the Food and Drug Administration, a database review showed.
“The FDA mandates that manufacturers and device operators disclose medical device reports to monitor suspected injuries and device malfunctions to the Manufacturer and User Facility Device Experience [MAUDE] database,” study author Adam J. Wulkan, MD, said at the annual conference of the American Society for Laser Medicine and Surgery. “Given the rapid growth in the noninvasive fat reduction market, it is essential physicians be aware of associated adverse events.”
“Given that this is the largest database for adverse events, I was surprised how few reports were done by physicians,” Dr. Wulkan said in an interview.
He reported results from 83 of the 98 MDRs. A total of 26 MDRs reported on cryolipolysis and included newly diagnosed or exacerbation of prior umbilical hernias (10), neuropathies (5), paradoxical fat hyperplasias (4), blisters (3), rashes (1), flu-like symptoms (1), gastroenteritis (1), and prolapsed bladder/uterus/rectum (1). Of the 11 MDRs for radio-frequency devices, 10 were burns/blisters and 1 fire.
Dr. Wulkan went on to note that 17 MDRs were reported for 1060-nm laser lipolysis, including burns/blisters (5), nodules (4), pain (3), cellulitis/abscesses (2), excessive swelling (1), neuropathy (1), and vomiting (1). There were four cases of burns on MDRs from focused/pulsed ultrasound procedures. One was on a non–FDA approved body site and another after combination radio-frequency/infrared treatment for cellulite reduction.
There were eight MDRs reported with 1440-nm laser cellulite reduction, including burn (5), unacceptable cosmesis (2), and faulty power supply (1). In addition, there were 12 MDRs reported on high-frequency ultrasound, including burns/blisters (9), subcutaneous nodules (2), and excoriation (1). Finally, vacuum-assisted submission was associated with five MDRs, including festooning/unacceptable cosmesis (2), seroma/hematoma (1), laceration (1), and nodules (1).
Dr. Wulkan acknowledged certain limitations of the study, including the fact that not all MDRs are reported to MAUDE, and that the reliability of patient reporting can be questionable. “We have to figure out if there is a natural causation between the adverse event and the treatment itself,” he said at the meeting. “The number of MDRs for any given device must be correlated to the number of procedures performed before we jump to conclusions about the safety of any given device.”
He reported having no financial disclosures. One study coauthor holds positions on advisory boards of, is a consultant for, and has intellectual property and/or stock options with various industry companies, such as Soliton and Allergan.
SOURCE: Wulkan et. al., ASLMS 2018.
REPORTING FROM ASLMS 2018
Key clinical point: Most physicians do not report adverse events related to noninvasive body contouring devices to the FDA .
Major finding: Of 83 medical device reports, 26 were related to cryolipolysis.
Study details: An analysis of 83 complications of noninvasive fat reduction and cellulite reduction devices reported to the FDA.
Disclosures: Dr. Wulkan reported having no financial disclosures. One study coauthor holds positions on advisory boards of, is a consultant for, and has intellectual property and/or stock options with various industry companies, such as Soliton and Allergan.
Source: Wulkan et al. ASLMS 2018.
Carboxytherapy caused small yet transient subcutaneous fat reduction
DALLAS – , a small randomized, controlled trial demonstrated.
“Carboxytherapy has been previously used therapeutically to reduce fat, and studies of carboxytherapy have been done in the past as well,” the study’s senior author, Murad Alam, MD, said in an interview in advance of a poster session at the annual conference of the American Society for Laser Medicine and Surgery Inc. “However, earlier studies have not been controlled trials.”
For the split-body study, he and his associates enrolled 16 adults and randomized one side of their body to receive infusions of 1,000 cc of CO2 every week for 5 weeks in the flank region, while the contralateral side received sham treatments. Outcomes of interest included fat layer thickness using a diagnostic ultrasound, total circumference, and body weight, said Dr. Alam, vice-chair of the department of dermatology and chief of the division of cutaneous and aesthetic surgery at Northwestern University, Chicago. He reported that a significant difference in fat thickness was observed in the carboxytherapy-treated side 1 week after the last treatment (P = .011), but this difference was not maintained at 28 weeks as measured by diagnostic ultrasound. Total circumference decreased nominally but not significantly. Body weights did not significantly change throughout the study.
Dr. Alam acknowledged that the study’s small sample size is a limitation. “We do think we did enough treatments, five per side, to obtain an effect if one were to be seen,” he said. The researchers reported having no financial disclosures.
DALLAS – , a small randomized, controlled trial demonstrated.
“Carboxytherapy has been previously used therapeutically to reduce fat, and studies of carboxytherapy have been done in the past as well,” the study’s senior author, Murad Alam, MD, said in an interview in advance of a poster session at the annual conference of the American Society for Laser Medicine and Surgery Inc. “However, earlier studies have not been controlled trials.”
For the split-body study, he and his associates enrolled 16 adults and randomized one side of their body to receive infusions of 1,000 cc of CO2 every week for 5 weeks in the flank region, while the contralateral side received sham treatments. Outcomes of interest included fat layer thickness using a diagnostic ultrasound, total circumference, and body weight, said Dr. Alam, vice-chair of the department of dermatology and chief of the division of cutaneous and aesthetic surgery at Northwestern University, Chicago. He reported that a significant difference in fat thickness was observed in the carboxytherapy-treated side 1 week after the last treatment (P = .011), but this difference was not maintained at 28 weeks as measured by diagnostic ultrasound. Total circumference decreased nominally but not significantly. Body weights did not significantly change throughout the study.
Dr. Alam acknowledged that the study’s small sample size is a limitation. “We do think we did enough treatments, five per side, to obtain an effect if one were to be seen,” he said. The researchers reported having no financial disclosures.
DALLAS – , a small randomized, controlled trial demonstrated.
“Carboxytherapy has been previously used therapeutically to reduce fat, and studies of carboxytherapy have been done in the past as well,” the study’s senior author, Murad Alam, MD, said in an interview in advance of a poster session at the annual conference of the American Society for Laser Medicine and Surgery Inc. “However, earlier studies have not been controlled trials.”
For the split-body study, he and his associates enrolled 16 adults and randomized one side of their body to receive infusions of 1,000 cc of CO2 every week for 5 weeks in the flank region, while the contralateral side received sham treatments. Outcomes of interest included fat layer thickness using a diagnostic ultrasound, total circumference, and body weight, said Dr. Alam, vice-chair of the department of dermatology and chief of the division of cutaneous and aesthetic surgery at Northwestern University, Chicago. He reported that a significant difference in fat thickness was observed in the carboxytherapy-treated side 1 week after the last treatment (P = .011), but this difference was not maintained at 28 weeks as measured by diagnostic ultrasound. Total circumference decreased nominally but not significantly. Body weights did not significantly change throughout the study.
Dr. Alam acknowledged that the study’s small sample size is a limitation. “We do think we did enough treatments, five per side, to obtain an effect if one were to be seen,” he said. The researchers reported having no financial disclosures.
REPORTING FROM ASLMS 2018
Key clinical point: Carboxytherapy yielded a modest reduction in fat that went away after a few months.
Major finding: A significant difference in fat thickness was observed in the carboxytherapy-treated flank region 1 week after the last treatment (P = .011).
Study details: A randomized, split-body study of 16 adults.
Disclosures: Dr. Alam reported having no financial disclosures.
Recently approved SK treatment spares melanocytes in preclinical study
A study using an ex vivo model to evaluate a seborrheic keratosis (SK) treatment shows that a topical application of 40% hydrogen peroxide is gentler on skin than a 5- or 10-second treatment with liquid nitrogen, particularly with respect to melanocytes, suggesting that the former may be less likely to produce disfiguring damage.
The 40% hydrogen peroxide solution (Eskata), also known as A-101, received Food and Drug Administration approval for the treatment of “seborrheic keratoses that are raised” in December, 2017. The study was published online in the Journal of the American Academy of Dermatology.
Melanocyte damage can lead to significant dyschromia, a poor cosmetic outcome that can have a tremendous impact on quality of life for dark-skinned patients, in whom it produces white spots. “A lot of these destructive approaches, especially liquid nitrogen, can leave more disfigurement upon treatment than the lesion itself,” the study’s lead author Adam Friedman, MD, said in an interview.
Melanocytes are particularly vulnerable to the effects of cold, so the destructive potential of liquid nitrogen is no surprise. But Dr. Friedman of the department of dermatology, George Washington University, Washington, wanted to get a better understanding of the impact of the new treatment on different skin cell types and the toxicity profile, so he approached the manufacturer, Aclaris Therapeutics, to do a study.
His team tested 40% hydrogen peroxide treatment and liquid nitrogen cryosurgery on a validated ex vivo human reconstituted full-thickness model derived from Fitzpatrick V skin, with 5 or 10 seconds of cryosurgery or 1 or 2 mcL of 40% hydrogen peroxide.
Using standard a hematoxylin and eosin stain as well as immunohistochemical staining to examine the architecture and cells types of the skin model following both treatments, the researchers found that 5- and 10-second cryosurgery resulted in significant thinning of the epidermis and increased cell death. There was also separation at the dermal-epidermal junction, which was more prominent in the 10-second cryosurgery group, although present even with a 5-second freeze cycle.
The hydrogen peroxide–treated groups showed acanthosis of the epidermis and mild pallor, but this was less noticeable than in the cryosurgery specimens. There was no epidermal clefting in the hydrogen peroxide–treated samples.
Tunel staining revealed 16.4 (plus or minus 0.6424) apoptotic cells per high-powered field in the 5-second cryosurgery experiment and 20.6 (plus or minus 0.6424) in the 10-second procedure. For the hydrogen peroxide treatment, those numbers were 8.65 (plus or minus 0.4122) in the 1 mcL experiment and 12.4 (plus or minus 0.3728) in the 2 mcL experiment.
As expected, melanocytes fared better with the hydrogen peroxide treatment. In the untreated samples, there were 2.5 melanocytes (plus or minus 0.1987) in the untreated sample and 2.0 (plus or minus 0.5000) melanocytes in the vehicle-treated sample. In the 5-second cryosurgery sample, there were 0.45 melanocytes (plus or minus 0.1535), and in the 10 second cryosurgery sample there were 0.2 (plus or minus 0.0918) melanocytes. In contrast, with the 1-mcL hydrogen peroxide-treated sample, there were 1.95 melanocytes in both the 1-mcL and 2-mcL samples (plus or minus 0.1535 for both groups).
“,” Dr. Friedman said. These results, he added, “offer us a lot of insight in terms of how damaging liquid nitrogen is, and it’s good to be reminded of that so that we don’t cause too much harm.”
The authors noted that a clinical trial evaluating the risk of hypopigmentation and hyperpigmentation with 40% hydrogen peroxide in people with darker skin types is underway. In the study, hydrogen peroxide is used to treat dermatosis papulosa nigra.
The study was funded by Aclaris Therapeutics. Senior author Adam Friedman, MD, is a consultant for Aclaris. Dr. Friedman is on the editorial board of Dermatology News.
SOURCE: Kao S et al. J Am Acad Dermatol. 2018 Mar 27. doi: 10.1016/j.jaad.2018.03.034.
A study using an ex vivo model to evaluate a seborrheic keratosis (SK) treatment shows that a topical application of 40% hydrogen peroxide is gentler on skin than a 5- or 10-second treatment with liquid nitrogen, particularly with respect to melanocytes, suggesting that the former may be less likely to produce disfiguring damage.
The 40% hydrogen peroxide solution (Eskata), also known as A-101, received Food and Drug Administration approval for the treatment of “seborrheic keratoses that are raised” in December, 2017. The study was published online in the Journal of the American Academy of Dermatology.
Melanocyte damage can lead to significant dyschromia, a poor cosmetic outcome that can have a tremendous impact on quality of life for dark-skinned patients, in whom it produces white spots. “A lot of these destructive approaches, especially liquid nitrogen, can leave more disfigurement upon treatment than the lesion itself,” the study’s lead author Adam Friedman, MD, said in an interview.
Melanocytes are particularly vulnerable to the effects of cold, so the destructive potential of liquid nitrogen is no surprise. But Dr. Friedman of the department of dermatology, George Washington University, Washington, wanted to get a better understanding of the impact of the new treatment on different skin cell types and the toxicity profile, so he approached the manufacturer, Aclaris Therapeutics, to do a study.
His team tested 40% hydrogen peroxide treatment and liquid nitrogen cryosurgery on a validated ex vivo human reconstituted full-thickness model derived from Fitzpatrick V skin, with 5 or 10 seconds of cryosurgery or 1 or 2 mcL of 40% hydrogen peroxide.
Using standard a hematoxylin and eosin stain as well as immunohistochemical staining to examine the architecture and cells types of the skin model following both treatments, the researchers found that 5- and 10-second cryosurgery resulted in significant thinning of the epidermis and increased cell death. There was also separation at the dermal-epidermal junction, which was more prominent in the 10-second cryosurgery group, although present even with a 5-second freeze cycle.
The hydrogen peroxide–treated groups showed acanthosis of the epidermis and mild pallor, but this was less noticeable than in the cryosurgery specimens. There was no epidermal clefting in the hydrogen peroxide–treated samples.
Tunel staining revealed 16.4 (plus or minus 0.6424) apoptotic cells per high-powered field in the 5-second cryosurgery experiment and 20.6 (plus or minus 0.6424) in the 10-second procedure. For the hydrogen peroxide treatment, those numbers were 8.65 (plus or minus 0.4122) in the 1 mcL experiment and 12.4 (plus or minus 0.3728) in the 2 mcL experiment.
As expected, melanocytes fared better with the hydrogen peroxide treatment. In the untreated samples, there were 2.5 melanocytes (plus or minus 0.1987) in the untreated sample and 2.0 (plus or minus 0.5000) melanocytes in the vehicle-treated sample. In the 5-second cryosurgery sample, there were 0.45 melanocytes (plus or minus 0.1535), and in the 10 second cryosurgery sample there were 0.2 (plus or minus 0.0918) melanocytes. In contrast, with the 1-mcL hydrogen peroxide-treated sample, there were 1.95 melanocytes in both the 1-mcL and 2-mcL samples (plus or minus 0.1535 for both groups).
“,” Dr. Friedman said. These results, he added, “offer us a lot of insight in terms of how damaging liquid nitrogen is, and it’s good to be reminded of that so that we don’t cause too much harm.”
The authors noted that a clinical trial evaluating the risk of hypopigmentation and hyperpigmentation with 40% hydrogen peroxide in people with darker skin types is underway. In the study, hydrogen peroxide is used to treat dermatosis papulosa nigra.
The study was funded by Aclaris Therapeutics. Senior author Adam Friedman, MD, is a consultant for Aclaris. Dr. Friedman is on the editorial board of Dermatology News.
SOURCE: Kao S et al. J Am Acad Dermatol. 2018 Mar 27. doi: 10.1016/j.jaad.2018.03.034.
A study using an ex vivo model to evaluate a seborrheic keratosis (SK) treatment shows that a topical application of 40% hydrogen peroxide is gentler on skin than a 5- or 10-second treatment with liquid nitrogen, particularly with respect to melanocytes, suggesting that the former may be less likely to produce disfiguring damage.
The 40% hydrogen peroxide solution (Eskata), also known as A-101, received Food and Drug Administration approval for the treatment of “seborrheic keratoses that are raised” in December, 2017. The study was published online in the Journal of the American Academy of Dermatology.
Melanocyte damage can lead to significant dyschromia, a poor cosmetic outcome that can have a tremendous impact on quality of life for dark-skinned patients, in whom it produces white spots. “A lot of these destructive approaches, especially liquid nitrogen, can leave more disfigurement upon treatment than the lesion itself,” the study’s lead author Adam Friedman, MD, said in an interview.
Melanocytes are particularly vulnerable to the effects of cold, so the destructive potential of liquid nitrogen is no surprise. But Dr. Friedman of the department of dermatology, George Washington University, Washington, wanted to get a better understanding of the impact of the new treatment on different skin cell types and the toxicity profile, so he approached the manufacturer, Aclaris Therapeutics, to do a study.
His team tested 40% hydrogen peroxide treatment and liquid nitrogen cryosurgery on a validated ex vivo human reconstituted full-thickness model derived from Fitzpatrick V skin, with 5 or 10 seconds of cryosurgery or 1 or 2 mcL of 40% hydrogen peroxide.
Using standard a hematoxylin and eosin stain as well as immunohistochemical staining to examine the architecture and cells types of the skin model following both treatments, the researchers found that 5- and 10-second cryosurgery resulted in significant thinning of the epidermis and increased cell death. There was also separation at the dermal-epidermal junction, which was more prominent in the 10-second cryosurgery group, although present even with a 5-second freeze cycle.
The hydrogen peroxide–treated groups showed acanthosis of the epidermis and mild pallor, but this was less noticeable than in the cryosurgery specimens. There was no epidermal clefting in the hydrogen peroxide–treated samples.
Tunel staining revealed 16.4 (plus or minus 0.6424) apoptotic cells per high-powered field in the 5-second cryosurgery experiment and 20.6 (plus or minus 0.6424) in the 10-second procedure. For the hydrogen peroxide treatment, those numbers were 8.65 (plus or minus 0.4122) in the 1 mcL experiment and 12.4 (plus or minus 0.3728) in the 2 mcL experiment.
As expected, melanocytes fared better with the hydrogen peroxide treatment. In the untreated samples, there were 2.5 melanocytes (plus or minus 0.1987) in the untreated sample and 2.0 (plus or minus 0.5000) melanocytes in the vehicle-treated sample. In the 5-second cryosurgery sample, there were 0.45 melanocytes (plus or minus 0.1535), and in the 10 second cryosurgery sample there were 0.2 (plus or minus 0.0918) melanocytes. In contrast, with the 1-mcL hydrogen peroxide-treated sample, there were 1.95 melanocytes in both the 1-mcL and 2-mcL samples (plus or minus 0.1535 for both groups).
“,” Dr. Friedman said. These results, he added, “offer us a lot of insight in terms of how damaging liquid nitrogen is, and it’s good to be reminded of that so that we don’t cause too much harm.”
The authors noted that a clinical trial evaluating the risk of hypopigmentation and hyperpigmentation with 40% hydrogen peroxide in people with darker skin types is underway. In the study, hydrogen peroxide is used to treat dermatosis papulosa nigra.
The study was funded by Aclaris Therapeutics. Senior author Adam Friedman, MD, is a consultant for Aclaris. Dr. Friedman is on the editorial board of Dermatology News.
SOURCE: Kao S et al. J Am Acad Dermatol. 2018 Mar 27. doi: 10.1016/j.jaad.2018.03.034.
FROM JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Key clinical point: The results of a preclinical study using an ex vivo model of darker skin found that a 40% hydrogen peroxide solution for treating SKs was less damaging to the skin.
Major finding: The topical hydrogen peroxide treatment left 1.95 melanocytes in place, compared with 0.2-0.4 in the liquid nitrogen samples. As expected, melanocytes fared better with the hydrogen peroxide treatment. In the untreated samples, there were 2.5 melanocytes (plus or minus 0.1987) in the untreated sample and 2.0 (plus or minus 0.5000) melanocytes in the vehicle-treated sample. In the 5-second cryosurgery sample, there were 0.45 melanocytes (plus or minus 0.1535), and in the 10-second cryosurgery sample there were 0.2 (plus or minus 0.0918) melanocytes.
Study details: The study compared the cytotoxic effects and impact on melanocytes of liquid nitrogen and 40% hydrogen peroxide solution using ex vivo human reconstituted full-thickness model.
Disclosures: The study was funded by Aclaris Therapeutics. Senior author Adam Friedman, MD, is a consultant for Aclaris.
Source: Kao S. et al. J Am Acad Dermatol. 2018 Mar 27. doi: 10.1016/j.jaad.2018.03.034.
Liability claims from cutaneous laser surgery continue to rise
DALLAS – , and the average indemnity claim is close to $320,000, results from an analysis of national data have shown.
“Physicians seeking to undertake laser surgery in their practice should ensure they have proper training in the physics, safety, and techniques of each procedure,” study author Saami Khalifian, MD, said in an interview in advance of the annual meeting of the American Society for Laser Medicine and Surgery. “Legal precedent has demonstrated that nonphysicians performing laser surgery will likely be held to a standard of care and training corresponding to a physician with appropriate training. Therefore, physicians who delegate laser operations to their nonphysician agents are ultimately responsible for the actions of their agents.”
For the period between June 2012 and December 2017, Dr. Khalifian and his colleagues identified 32 cases with public decision related to injury secondary to cutaneous laser surgery. The incidence of litigation related to laser surgery demonstrates an increasing trend since 1985, with peak occurrence in 2010 (total of 22 cases, which represents 10.7% of 206 cases reported in the entire data set since 1985). Laser hair removal was the most common litigated procedure prior to 2012, but since then has been surpassed by laser rejuvenation, which includes ablative and nonablative resurfacing, fractionated lasers, and intense pulsed light (47% of cases since mid-2012). Nonphysician operators accounted for a substantial subset of these cases (66% of all cases since mid-2012), with their physician supervisors named as defendants, despite not performing the procedure.
Dr. Khalifian noted that medical spas in the study “uniformly had unsupervised nonphysician operators in nonmedical office settings,” and therefore “had one of the highest litigation rates and number of cases. What is interesting about this, however, is that medical spas that are run by nonphysicians are also less likely to have medical liability insurance to satisfy a potential malpractice claim and therefore would be less likely to be pursued by a plaintiff’s attorney. Despite this, they had one of the highest patient injury rates, which is likely underrepresented based on the lack of malpractice insurance to be pursued in a legal claim.”
The researchers also found that delegated procedures were most frequently litigated against regardless of subspecialty of supervising physician (66%). Of the preventable causes of action, the most common was failure to obtain an informed consent (34%) and negligence (50%). Of the 32 cases with public decisions, 17 (53.1%) resulted in decisions in favor of the plaintiff. The mean indemnity payment was $318,832.
“Our current work evaluating data from 2012-2017 is consistent with our prior work dating back to 1985 and demonstrates again that nonphysician operators account for the majority of cases of legal action,” Dr. Khalifian said. “In fact, there is an increasing trend since about 2008 for a higher proportion of cases being performed by physician extenders and nonphysician operators. Despite physicians acting as supervisors and not the actual laser operator they are nonetheless named as defendants in legal claims,” he added.
The data on allegations and causes of legal action “should serve as a guide to help improve patient safety and decrease professional liability,” he pointed out. “It is critical that physicians take steps to mitigate risk to patients by ensuring robust and adequate training for their agents and by directly supervising procedures. However, the data clearly show that the safest treatments are performed directly by physicians who have had such dedicated training in laser and light sources.”
Dr. Khalifian acknowledged certain limitations of the study, including the fact that the data were extracted from only one legal database and did not include cases settled outside of the justice system through third-party arbitration or other means. “It is also limited by search terms in our query,” he said. “It is possible that some cases did not include the terms we used. Finally, only incomplete information was available for some cases even when supplemented through other resources and additional research.”
He reported having no financial disclosures. Study coauthor Mathew M. Avram, MD, JD, serves on the medical advisory board of Sciton and on the scientific advisory boards of Sienna Biopharmaceuticals, Cytrellis, and Allergan. He is also consultant for Merz Aesthetics, Allergan, Soliton, Invasix, and Revance, and has intellectual property with Cytrellis. He also holds stock options with Cytrellis, Invasix, and Zalea.
SOURCE: Khalifian et al. ASLMS 2018.
DALLAS – , and the average indemnity claim is close to $320,000, results from an analysis of national data have shown.
“Physicians seeking to undertake laser surgery in their practice should ensure they have proper training in the physics, safety, and techniques of each procedure,” study author Saami Khalifian, MD, said in an interview in advance of the annual meeting of the American Society for Laser Medicine and Surgery. “Legal precedent has demonstrated that nonphysicians performing laser surgery will likely be held to a standard of care and training corresponding to a physician with appropriate training. Therefore, physicians who delegate laser operations to their nonphysician agents are ultimately responsible for the actions of their agents.”
For the period between June 2012 and December 2017, Dr. Khalifian and his colleagues identified 32 cases with public decision related to injury secondary to cutaneous laser surgery. The incidence of litigation related to laser surgery demonstrates an increasing trend since 1985, with peak occurrence in 2010 (total of 22 cases, which represents 10.7% of 206 cases reported in the entire data set since 1985). Laser hair removal was the most common litigated procedure prior to 2012, but since then has been surpassed by laser rejuvenation, which includes ablative and nonablative resurfacing, fractionated lasers, and intense pulsed light (47% of cases since mid-2012). Nonphysician operators accounted for a substantial subset of these cases (66% of all cases since mid-2012), with their physician supervisors named as defendants, despite not performing the procedure.
Dr. Khalifian noted that medical spas in the study “uniformly had unsupervised nonphysician operators in nonmedical office settings,” and therefore “had one of the highest litigation rates and number of cases. What is interesting about this, however, is that medical spas that are run by nonphysicians are also less likely to have medical liability insurance to satisfy a potential malpractice claim and therefore would be less likely to be pursued by a plaintiff’s attorney. Despite this, they had one of the highest patient injury rates, which is likely underrepresented based on the lack of malpractice insurance to be pursued in a legal claim.”
The researchers also found that delegated procedures were most frequently litigated against regardless of subspecialty of supervising physician (66%). Of the preventable causes of action, the most common was failure to obtain an informed consent (34%) and negligence (50%). Of the 32 cases with public decisions, 17 (53.1%) resulted in decisions in favor of the plaintiff. The mean indemnity payment was $318,832.
“Our current work evaluating data from 2012-2017 is consistent with our prior work dating back to 1985 and demonstrates again that nonphysician operators account for the majority of cases of legal action,” Dr. Khalifian said. “In fact, there is an increasing trend since about 2008 for a higher proportion of cases being performed by physician extenders and nonphysician operators. Despite physicians acting as supervisors and not the actual laser operator they are nonetheless named as defendants in legal claims,” he added.
The data on allegations and causes of legal action “should serve as a guide to help improve patient safety and decrease professional liability,” he pointed out. “It is critical that physicians take steps to mitigate risk to patients by ensuring robust and adequate training for their agents and by directly supervising procedures. However, the data clearly show that the safest treatments are performed directly by physicians who have had such dedicated training in laser and light sources.”
Dr. Khalifian acknowledged certain limitations of the study, including the fact that the data were extracted from only one legal database and did not include cases settled outside of the justice system through third-party arbitration or other means. “It is also limited by search terms in our query,” he said. “It is possible that some cases did not include the terms we used. Finally, only incomplete information was available for some cases even when supplemented through other resources and additional research.”
He reported having no financial disclosures. Study coauthor Mathew M. Avram, MD, JD, serves on the medical advisory board of Sciton and on the scientific advisory boards of Sienna Biopharmaceuticals, Cytrellis, and Allergan. He is also consultant for Merz Aesthetics, Allergan, Soliton, Invasix, and Revance, and has intellectual property with Cytrellis. He also holds stock options with Cytrellis, Invasix, and Zalea.
SOURCE: Khalifian et al. ASLMS 2018.
DALLAS – , and the average indemnity claim is close to $320,000, results from an analysis of national data have shown.
“Physicians seeking to undertake laser surgery in their practice should ensure they have proper training in the physics, safety, and techniques of each procedure,” study author Saami Khalifian, MD, said in an interview in advance of the annual meeting of the American Society for Laser Medicine and Surgery. “Legal precedent has demonstrated that nonphysicians performing laser surgery will likely be held to a standard of care and training corresponding to a physician with appropriate training. Therefore, physicians who delegate laser operations to their nonphysician agents are ultimately responsible for the actions of their agents.”
For the period between June 2012 and December 2017, Dr. Khalifian and his colleagues identified 32 cases with public decision related to injury secondary to cutaneous laser surgery. The incidence of litigation related to laser surgery demonstrates an increasing trend since 1985, with peak occurrence in 2010 (total of 22 cases, which represents 10.7% of 206 cases reported in the entire data set since 1985). Laser hair removal was the most common litigated procedure prior to 2012, but since then has been surpassed by laser rejuvenation, which includes ablative and nonablative resurfacing, fractionated lasers, and intense pulsed light (47% of cases since mid-2012). Nonphysician operators accounted for a substantial subset of these cases (66% of all cases since mid-2012), with their physician supervisors named as defendants, despite not performing the procedure.
Dr. Khalifian noted that medical spas in the study “uniformly had unsupervised nonphysician operators in nonmedical office settings,” and therefore “had one of the highest litigation rates and number of cases. What is interesting about this, however, is that medical spas that are run by nonphysicians are also less likely to have medical liability insurance to satisfy a potential malpractice claim and therefore would be less likely to be pursued by a plaintiff’s attorney. Despite this, they had one of the highest patient injury rates, which is likely underrepresented based on the lack of malpractice insurance to be pursued in a legal claim.”
The researchers also found that delegated procedures were most frequently litigated against regardless of subspecialty of supervising physician (66%). Of the preventable causes of action, the most common was failure to obtain an informed consent (34%) and negligence (50%). Of the 32 cases with public decisions, 17 (53.1%) resulted in decisions in favor of the plaintiff. The mean indemnity payment was $318,832.
“Our current work evaluating data from 2012-2017 is consistent with our prior work dating back to 1985 and demonstrates again that nonphysician operators account for the majority of cases of legal action,” Dr. Khalifian said. “In fact, there is an increasing trend since about 2008 for a higher proportion of cases being performed by physician extenders and nonphysician operators. Despite physicians acting as supervisors and not the actual laser operator they are nonetheless named as defendants in legal claims,” he added.
The data on allegations and causes of legal action “should serve as a guide to help improve patient safety and decrease professional liability,” he pointed out. “It is critical that physicians take steps to mitigate risk to patients by ensuring robust and adequate training for their agents and by directly supervising procedures. However, the data clearly show that the safest treatments are performed directly by physicians who have had such dedicated training in laser and light sources.”
Dr. Khalifian acknowledged certain limitations of the study, including the fact that the data were extracted from only one legal database and did not include cases settled outside of the justice system through third-party arbitration or other means. “It is also limited by search terms in our query,” he said. “It is possible that some cases did not include the terms we used. Finally, only incomplete information was available for some cases even when supplemented through other resources and additional research.”
He reported having no financial disclosures. Study coauthor Mathew M. Avram, MD, JD, serves on the medical advisory board of Sciton and on the scientific advisory boards of Sienna Biopharmaceuticals, Cytrellis, and Allergan. He is also consultant for Merz Aesthetics, Allergan, Soliton, Invasix, and Revance, and has intellectual property with Cytrellis. He also holds stock options with Cytrellis, Invasix, and Zalea.
SOURCE: Khalifian et al. ASLMS 2018.
REPORTING FROM ASLMS 2018
Key clinical point: Claims related to cutaneous laser surgery are steadily increasing, as are the average indemnity payments.
Major finding: Among cases with public decision related to injury secondary to cutaneous laser surgery, nearly half since mid-2012 (47%) have been due to laser rejuvenation.
Study details: An analysis of 32 medical professional liability claims related to cutaneous laser surgery June 2012 through December 2017.
Disclosures: Dr. Khalifian reported having no financial disclosures. Study coauthor Mathew Avram, MD, serves on the medical advisory board of Sciton and on the scientific advisory boards of Sienna Biopharmaceuticals, Cytrellis, and Allergan. He is also consultant for Merz Aesthetics, Allergan, Soliton, Invasix, and Revance, and has intellectual property with Cytrellis. He also holds stock options with Cytrellis, Invasix, and Zalea.
Source: Khalifian et al. ASLMS 2018.
Update on Noninvasive Body Contouring Techniques
In today’s society there is a ubiquitous pressure to lose weight, reduce fat, and rejuvenate the skin that stems not only from images of idealized bodies in the media but also from our growing knowledge of the detrimental effects of obesity. Along with diet and exercise, it has become popular to use noninvasive devices to attain these goals by means of body contouring—the optimization of the definition, smoothness, and shape of the human physique.1 In fact, body contouring currently is the fastest-growing area of cosmetic dermatology.2
Previously, body contouring primarily involved invasive procedures (eg, liposuction) that are associated with various adverse effects, financial costs, and lengthy downtime.3 More recently, a growing demand for safer and less painful procedures for adipose tissue reduction and skin tightening have led to the development of several novel modalities for noninvasive body contouring. Although the results achieved using these new technologies may be less dramatic than invasive techniques and are not immediate, they do not carry the risks and adverse effects that are associated with surgical procedures and therefore are increasingly requested by cosmetic patients.4,5 New noninvasive techniques primarily target the physical properties of fat, resulting in an efflux of triglycerides from fat cells, causing either reduced size, necrosis, or apoptosis of adipocytes.3,6 Of these modalities, cold-induced adipocyte apoptosis has been commercially available the longest and has been the most researched; however, other noninvasive body contouring techniques have been increasingly explored by researchers since the first reports of human adipose tissue explants exhibiting features of apoptosis after heat injury became available.7,8
There currently are 4 leading modalities used for noninvasive body contouring: cryolipolysis, radiofrequency (RF), high-intensity focused ultrasound (HIFU), and laser therapy (Table). Although no procedure has yet been accepted as the gold standard, investigators are working to determine which technique is the most effective.9 In this article, we provide an overview of these techniques to help dermatologists choose appropriate modalities for their cosmetic patients.
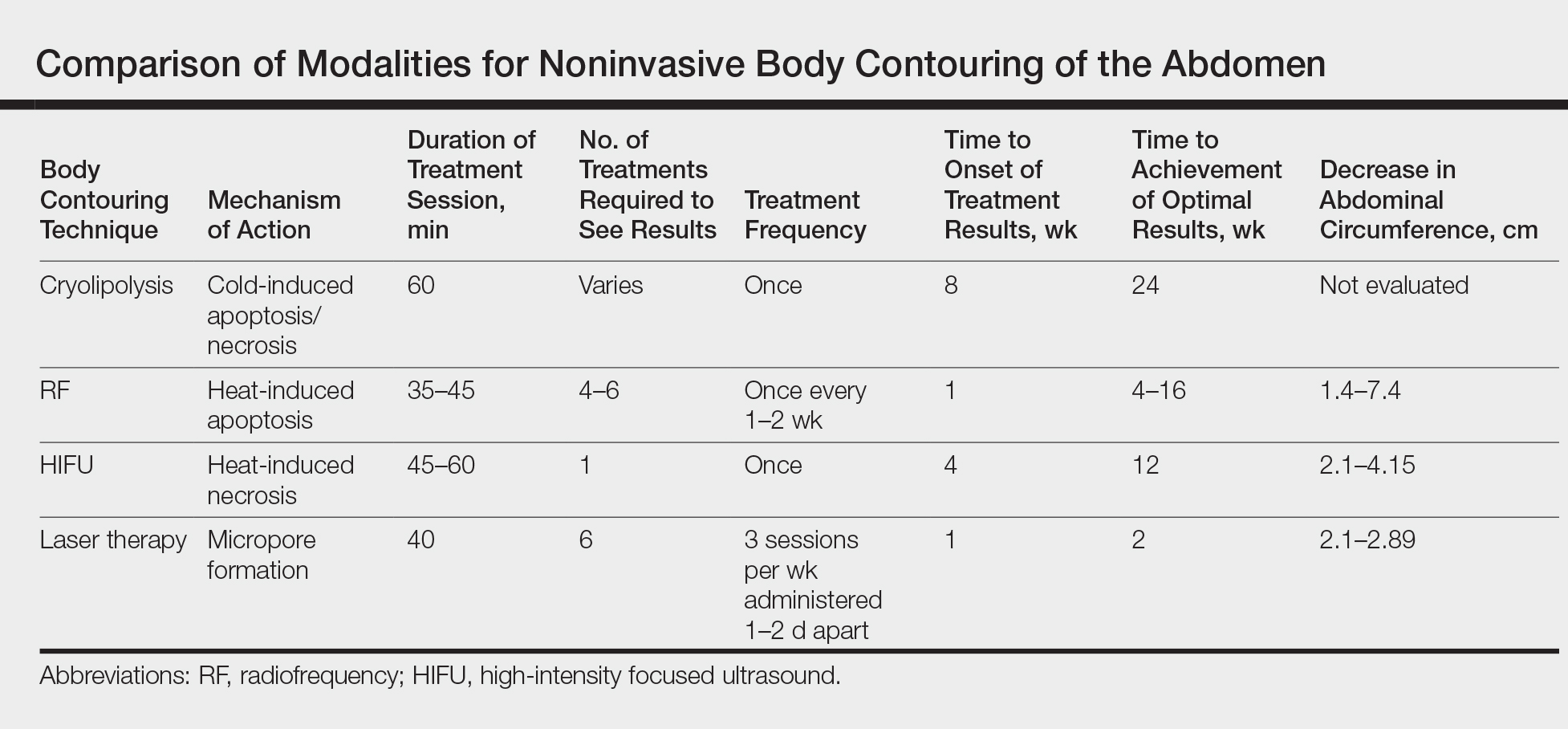
Cryolipolysis
Cryolipolysis is unique in that it employs the principle that lipid-rich adipocytes are more susceptible to freezing than surrounding water-rich cells, allowing selective apoptosis while preserving the adjacent structures. As macrophages digest the apoptotic adipocytes, patients experience a decrease in subcutaneous fat volume over the subsequent 2 to 3 months.10-13 Cryolipolysis has been gaining popularity since 2010, when it was first approved by the US Food and Drug Administration (FDA) for fat reduction in the flank areas; it was later approved for the abdomen in 2012, thighs in 2014, and submental area in 2015.14 Most recently, cryolipolysis was approved for fat reduction in the arms, back, and buttocks in 2016.
The most popular cryolipolysis device applies suction to the treatment area and vacuums the tissue between 2 cooling panels for 30 to 60 minutes.9 Clinical studies investigating the safety and efficacy of cryolipolysis have reported a high degree of patient satisfaction with the procedure and only minimal side effects.4,6,15,16 Common complications of cryolipolysis include erythema, swelling, and sensitivity at the treatment site followed by a lesser incidence of pain, tingling, and bruising, all of which generally resolve within a few weeks of treatment.6 With the removal of adipocytes, there has been concern regarding elevations in blood lipid levels and liver enzymes; however, these laboratory values have been reported to remain within normal limits during and after cryolipolysis.17,18 Of note, patients should be advised of the risk of paradoxical adipose hyperplasia, a rare side effect of cryolipolysis in which a large, demarcated, tender fat mass develops at the treatment site 2 to 3 months after treatment, with an estimated incidence of 1 in 20,000.19 However, the incidence of paradoxical adipose hyperplasia may be underestimated, as a single practice reported an incidence of 0.47% in 422 cryolipolysis treatments.20 This complication has not been associated with any of the heat-induced fat reduction modalities.
Cryolipolysis has been found to be safe for all skin types with no reported pigmentary changes.16 It should not be performed in patients with cold-induced conditions (eg, cryoglobulinemia, cold urticaria) or in those with severe varicose veins or atopic dermatitis.21,22 Patients benefitting most from this procedure are those who require only small or moderate amounts of adipose tissue and cellulite removal with separate fat bulges.12,17 Interestingly, cryolipolysis also has been used off label to treat pseudogynecomastia in male patients.23
Radiofrequency
Radiofrequency has become an important and frequently used modality in cosmetic dermatology.24 This modality differs from cryolipolysis in that it relies on exploiting the difference in water content and impedance between tissues: the skin has low impedance, whereas fat tissue has high impedance. Radiofrequency induces thermal injury to targeted tissue layers, rather than the cold-induced damage seen in cryolipolysis, through devices that focus thermal energy on tissues with high impedance, inducing apoptosis of cells in the subcutaneous adipose tissue with minimal risk of damaging the epidermis, dermis, and muscle.9,25 Ultimately, thermal exposure to 43°C to 45°C over several minutes results in a delayed adipocyte death response.4 In addition to adipocyte death, RF has been shown to cause denaturation of collagen fibrils, leading to subsequent remodeling, neocollagenesis, and skin tightening.26
Radiofrequency devices can be broadly classified as monopolar or bipolar.24,27 Bipolar devices generally require more frequent treatments, whereas monopolar devices tend to require fewer treatment sessions with superior circumference and fat reduction.28
Overall, RF devices have a favorable side effect profile. The most common side effects are erythema and edema at the treatment site lasting less than 24 hours after the procedure.25 The absence of complications such as abdominal discomfort, erythema, and burning during treatment have been reported,27 with the exception of 1 case of hyperesthesia on the abdomen that lasted for 3 days after a treatment session.5 Although RF has beneficial effects on circumference reduction in the abdomen and thighs and can improve the appearance of cellulite, an increase in body weight may occur during treatment. When a localized area of fat such as the thigh is targeted for treatment but the remaining fat cells in the body are not affected, the remaining cells can continue to grow and expand; for instance, although fat cells destroyed with RF will not continue to expand, fat cells in untreated areas may continue to grow due to continued weight gain (eg, from excessive eating), leading to overall weight gain. Thus, patients must understand that weight gain is not an indication of treatment failure after RF or any other method of irreversible fat destruction.5
High-intensity Focused Ultrasound
High-intensity focused ultrasound recently was introduced as a new treatment modality for body contouring, specifically for skin tightening and rejuvenation.5 The mechanism of HIFU is similar to that of RF in that it also relies on heat to cause adipocyte apoptosis; however, it utilizes acoustic energy rather than electric energy. High-intensity focused ultrasound devices can deliver energy to the deep dermis, subdermal connective tissue, and fibromuscular layers in precise microcoagulation zones without damage to the epidermis. The focused energy induces a high temperature (>65°C) within 1 to 3 seconds, causing cell protein coagulation in the targeted area. In addition to its thermal effects, HIFU induces a mechanical effect that disrupts cell membranes immediately, which contributes to the coagulation necrosis process, further promoting necrosis and apoptosis. The effects of these devices can be visualized, as there always is a sharp demarcation between the targeted and untargeted tissue.29 Additionally, microcoagulation is thought to cause gradual skin tightening through collagen contraction and remodeling.30
High-intensity focused ultrasound first received FDA approval for eyebrow lifting and has been used safely and effectively to treat facial and neck skin in a variety of skin types as well as to improve the clinical appearance of the abdomen and thighs.31 This technique is best suited for patients with mild to moderate laxity of the skin or soft tissue who have a body mass index less than 30 kg/m2 and are seeking mild body contouring.32 The ideal patient is young with normal wound healing, since the clinical response to treatment is partly dependent on new collagen synthesis.33 Older patients with extensive photoaging or severe skin laxity are not good candidates for HIFU.
There are a variety of available HIFU devices,34 which utilize special transducers that direct ultrasound energy to a small focal point in the subcutaneous tissues that harmlessly passes through the skin.35 By using newly developed transducers with different energy outputs and focal depths, dermatologists can tailor HIFU treatment to meet the unique physical characteristics of each patient.31
Adverse effects of HIFU are limited to transient pain in most patients and occasional erythema and ecchymosis in some cases.31 In general, most adverse effects resolve spontaneously within 4 weeks and all by 12 weeks posttreatment. Studies also have reported hard subcutaneous nodules, discomfort, burning sensation, mild blisters, and one case of purpuric lesions, all at the treatment site.36-39 There is no evidence that HIFU can cause abnormalities in serum lipids or liver function tests.
Lasers
Laser technology is a rapidly growing modality in noninvasive body contouring. A novel device recently emerged as the first and only FDA-cleared hyperthermic laser for fat reduction and noninvasive body contouring of the abdomen, flanks, back, inner and outer thighs, and submental area.40,41 The device is a 1060-nm diode laser that uses thermal energy to destroy adipose tissue, leading to permanent reduction in stubborn fat without surgery or downtime through the use of a flat, nonsuction applicator that is designed for consistent, natural-looking results. The device includes a contact cooling system that helps to limit thermal discomfort and prevent damage to the surface of the skin during the procedure. Initial improvement can be seen as quickly as 6 weeks posttreatment, and optimal results usually occur in as few as 12 weeks. This device was found to have an excellent safety profile and was well tolerated among patients, with only mild pain reported.42,43
Prior to the development of this new 1060-nm diode laser, the initial application of lasers for noninvasive body contouring involved low-level laser therapy (LLLT), also known as cold laser therapy.40 One device has 5 rotating diode laser heads that work at a wavelength of 635 nm. Treatment sessions last up to 30 minutes, and 6 to 8 sessions are required to obtain optimal results. Low-level laser therapy is a unique modality that is not based on thermal tissue damage, but rather on producing transient microscopic pores in adipocytes that allow lipids to leak out, leading to fat reduction.34 Because LLLT causes immediate emptying of targeted adipocytes, results are noticeable as soon as treatment is completed; however, there is no necrosis or apoptosis of adipocytes, so the recurrence of fat deposition is believed to be greater when compared to the other modalities. Because the results are temporary, long-term or permanent results should not be expected with LLLT. Depending on the patient’s goals, the temporary nature of the results can be either an advantage or disadvantage: some may prefer immediate results despite gradual diminishment over subsequent months, whereas others may prefer results that progressively increase over time and are more permanent, as seen with cryolipolysis, HIFU, and RF.3
Complications of LLLT generally are fewer and more mild than with all other body contouring procedures, with several studies reporting no adverse effects.44-48 Others reported swelling or erythema at the treatment area, pain or tingling during treatment, and increased urination, all of which were temporary and resolved spontaneously.49 Additionally, although the lipids released from treatment are cleared through the lymphatic system, LLLT has not been shown to increase serum lipid levels.50
Conclusion
The field of noninvasive body contouring is undoubtedly growing and will likely continue to rise in popularity as the efficacy and safety of these treatments improve. Although the available technologies vary by mechanism and side effect profiles, several devices have been revealed to be safe and effective in reducing subcutaneous fat tissue and improving skin laxity.1 However, additional studies are needed to evaluate these devices in a standardized manner, especially considering the high costs associated with treatment.32 Current studies investigating these devices vary in treatment protocol, treatment area, number and timing of follow-up sessions, and outcome measures, making it challenging to compare the results objectively.3 Dermatologists offering body contouring treatments need to be intimately familiar with the available devices and determine which treatment is appropriate for each patient in order to provide the highest quality care. Most importantly, patients and physicians must discuss individual goals when choosing a body-contouring method in order to maximize patient satisfaction.
- Jalian HR, Avram MM. Body contouring: the skinny on noninvasive fat removal. Semin Cutan Med Surg. 2012;31:121-125.
- Ho D, Jagdeo J. A systematic review of paradoxical adipose hyperplasia (PAH) post-cryolipolysis. J Drugs Dermatol. 2017;16:62-67.
- Kennedy J, Verne S, Griffith R, et al. Non-invasive subcutaneous fat reduction: a review. J Eur Acad Dermatol Venereol. 2015;29:1679-1688.
- Krueger N, Mai SV, Luebberding S, et al. Cryolipolysis for noninvasive body contouring: clinical efficacy and patient satisfaction. Clin Cosmet Investig Dermatol. 2014;7:201-205.
- Suh DH, Kim CM, Lee SJ, et al. Safety and efficacy of a non-contact radiofrequency device for body contouring in Asians. J Cosmet Laser Ther. 2017;19:89-92.
- Ingargiola MJ, Motakef S, Chung MT, et al. Cryolipolysis for fat reduction and body contouring: safety and efficacy of current treatment paradigms. Plast Reconstr Surg. 2015;135:1581-1590.
- Prins JB, Walker NI, Winterford CM, et al. Apoptosis of human adipocytes in vitro. Biochem Biophys Res Commun. 1994;201:500-507.
- Sorisky A, Magun R, Gagnon AM. Adipose cell apoptosis: death in the energy depot. Int J Obes Relat Metab Disord. 2000;24(suppl 4):S3-S7.
- Chilukuri S, Mueller G. “Hands-free” noninvasive body contouring devices: review of effectiveness and patient satisfaction. J Drugs Dermatol. 2016;15:1402-1406.
- Manstein D, Laubach H, Watanabe K, et al. Selective cryolysis: a novel method of non-invasive fat removal. Lasers Surg Med. 2008;40:595-604.
- Zelickson B, Egbert BM, Preciado J, et al. Cryolipolysis for noninvasive fat cell destruction: initial results from a pig model. Dermatol Surg. 2009;35:1462-1470.
- Nelson AA, Wasserman D, Avram MM. Cryolipolysis for reduction of excess adipose tissue. Semin Cutan Med Surg. 2009;28:244-249.
- Avram MM, Harry RS. Cryolipolysis for subcutaneous fat layer reduction. Lasers Surg Med. 2009;41:703-708.
- Klein KB, Bachelor EP, Becker EV, et al. Multiple same day cryolipolysis treatments for the reduction of subcutaneous fat are safe and do not affect serum lipid levels or liver function tests. Lasers Surg Med. 2017;49:640-644.
- Dierickx CC, Mazer JM, Sand M, et al. Safety, tolerance, and patient satisfaction with noninvasive cryolipolysis. Dermatol Surg. 2013;39:1209-1216.
- Stevens WG, Pietrzak LK, Spring MA. Broad overview of a clinical and commercial experience with CoolSculpting. Aesthet Surg J. 2013;33:835-846.
- Ferraro GA, De Francesco F, Cataldo C, et al. Synergistic effects of cryolipolysis and shock waves for noninvasive body contouring. Aesthetic Plast Surg. 2012;36:666-679.
- Lee KR. Clinical efficacy of fat reduction on the thigh of Korean women through cryolipolysis. J Obes Weight Loss Ther. 2013;3:203.
- Jalian HR, Avram MM, Garibyan L, et al. Paradoxical adipose hyperplasia after cryolipolysis. JAMA Dermatol. 2014;150:317-319.
- Singh SM, Geddes ER, Boutrous SG, et al. Paradoxical adipose hyperplasia secondary to cryolipolysis: an underreported entity? Lasers Surg Med. 2015;47:476-478.
- Pinto H, Arredondo E, Ricart-Jane D. Evaluation of adipocytic changes after a simil-lipocryolysis stimulus. Cryo Letters. 2013;34:100-105.
- Pinto HR, Garcia-Cruz E, Melamed GE. A study to evaluate the action of lipocryolysis. Cryo Letters. 2012;33:177-181.
- Singh B, Keaney T, Rossi AM. Male body contouring. J Drugs Dermatol. 2015;14:1052-1059.
- Beasley KL, Weiss RA. Radiofrequency in cosmetic dermatology. Dermatol Clin. 2014;32:79-90.
- Weiss R, Weiss M, Beasley K, et al. Operator independent focused high frequency ISM band for fat reduction: porcine model. Lasers Surg Med. 2013;45:235-239.
- Hantash BM, Ubeid AA, Chang H, et al. Bipolar fractional radiofrequency treatment induces neoelastogenesis and neocollagenesis. Lasers Surg Med. 2009;41:1-9.
- Harth Y. Painless, safe, and efficacious noninvasive skin tightening, body contouring, and cellulite reduction using multisource 3DEEP radiofrequency. J Cosmet Dermatol. 2015;14:70-75.
- Nassab R. The evidence behind noninvasive body contouring devices. Aesthet Surg J. 2015;35:279-293.
- Luo W, Zhou X, Gong X, et al. Study of sequential histopathologic changes, apoptosis, and cell proliferation in rabbit livers after high-intensity focused ultrasound ablation. J Ultrasound Med. 2007;26:477-485.
- Minkis K, Alam M. Ultrasound skin tightening. Dermatol Clin. 2014;32:71-77.
- Ko EJ, Hong JY, Kwon TR, et al. Efficacy and safety of non-invasive body tightening with high-intensity focused ultrasound (HIFU). Skin Res Technol. 2017;23:558-562.
- Sklar LR, El Tal AK, Kerwin LY. Use of transcutaneous ultrasound for lipolysis and skin tightening: a review. Aesthetic Plast Surg. 2014;38:429-441.
- MacGregor JL, Tanzi EL. Microfocused ultrasound for skin tightening. Semin Cutan Med Surg. 2013;32:18-25.
- Alizadeh Z, Halabchi F, Mazaheri R, et al. Review of the mechanisms and effects of noninvasive body contouring devices on cellulite and subcutaneous fat. Int J Endocrinol Metab. 2016;14:E36727 .
- Fabi SG. Noninvasive skin tightening: focus on new ultrasound techniques. Clin Cosmet Investig Dermatol. 2015;8:47-52.
- Fatemi A. High-intensity focused ultrasound effectively reduces adipose tissue. Semin Cutan Med Surg. 2009;28:257-262.
- Teitelbaum SA, Burns JL, Kubota J, et al. Noninvasive body contouring by focused ultrasound: safety and efficacy of the Contour I device in a multicenter, controlled, clinical study. Plast Reconstr Surg. 2007;120:779-789.
- Hotta TA. Nonsurgical body contouring with focused ultrasound. Plast Surg Nurs. 2010;30:77-82; quiz 83-84.
- Fatemi A, Kane MA. High-intensity focused ultrasound effectively reduces waist circumference by ablating adipose tissue from the abdomen and flanks: a retrospective case series. Aesthetic Plast Surg. 2010;34:577-582.
- Schilling L, Saedi N, Weiss R. 1060 nm diode hyperthermic laser lipolysis: the latest in non-invasive body contouring. J Drugs Dermatol. 2017;16:48-52.
- Body contouring. CynoSure website. https://www.cynosure.com/treatment/body-contouring/SculpSure. Accessed March 28, 2018.
- Decorato JW, Chen B, Sierra R. Subcutaneous adipose tissue response to a non-invasive hyperthermic treatment using a 1,060 nm laser. Lasers Surg Med. 2017;49:480-489.
- Weiss R, McDaniel D, Doherty S. Clinical evaluation of fat reduction treatment of the flanks and abdomen with a non-invasive 1060 nm diode laser: a multicenter study. Paper presented at: 2016 Annual American Society for Laser Medicine and Surgery Conference; March 30–April 3, 2016; Boston, MA.
- Caruso-Davis MK, Guillot TS, Podichetty VK, et al. Efficacy of low-level laser therapy for body contouring and spot fat reduction. Obes Surg. 2011;21:722-729.
- McRae E, Boris J. Independent evaluation of low-level laser therapy at 635 nm for non-invasive body contouring of the waist, hips, and thighs. Lasers Surg Med. 2013;45:1-7.
- Nestor MS, Newburger J, Zarraga MB. Body contouring using 635-nm low level laser therapy. Semin Cutan Med Surg. 2013;32:35-40.
- Jackson RF, Stern FA, Neira R, et al. Application of low-level laser therapy for noninvasive body contouring. Lasers Surg Med. 2012;44:211-217.
- Jackson RF, Dedo DD, Roche GC, et al. Low-level laser therapy as a non-invasive approach for body contouring: a randomized, controlled study. Lasers Surg Med. 2009;41:799-809.
- Gold MH, Khatri KA, Hails K, et al. Reduction in thigh circumference and improvement in the appearance of cellulite with dual-wavelength, low-level laser energy and massage. J Cosmet Laser Ther. 2011;13:13-20.
- Avci P, Nyame TT, Gupta GK, et al. Low-level laser therapy for fat layer reduction: a comprehensive review. Lasers Surg Med. 2013;45:349-357.
In today’s society there is a ubiquitous pressure to lose weight, reduce fat, and rejuvenate the skin that stems not only from images of idealized bodies in the media but also from our growing knowledge of the detrimental effects of obesity. Along with diet and exercise, it has become popular to use noninvasive devices to attain these goals by means of body contouring—the optimization of the definition, smoothness, and shape of the human physique.1 In fact, body contouring currently is the fastest-growing area of cosmetic dermatology.2
Previously, body contouring primarily involved invasive procedures (eg, liposuction) that are associated with various adverse effects, financial costs, and lengthy downtime.3 More recently, a growing demand for safer and less painful procedures for adipose tissue reduction and skin tightening have led to the development of several novel modalities for noninvasive body contouring. Although the results achieved using these new technologies may be less dramatic than invasive techniques and are not immediate, they do not carry the risks and adverse effects that are associated with surgical procedures and therefore are increasingly requested by cosmetic patients.4,5 New noninvasive techniques primarily target the physical properties of fat, resulting in an efflux of triglycerides from fat cells, causing either reduced size, necrosis, or apoptosis of adipocytes.3,6 Of these modalities, cold-induced adipocyte apoptosis has been commercially available the longest and has been the most researched; however, other noninvasive body contouring techniques have been increasingly explored by researchers since the first reports of human adipose tissue explants exhibiting features of apoptosis after heat injury became available.7,8
There currently are 4 leading modalities used for noninvasive body contouring: cryolipolysis, radiofrequency (RF), high-intensity focused ultrasound (HIFU), and laser therapy (Table). Although no procedure has yet been accepted as the gold standard, investigators are working to determine which technique is the most effective.9 In this article, we provide an overview of these techniques to help dermatologists choose appropriate modalities for their cosmetic patients.

Cryolipolysis
Cryolipolysis is unique in that it employs the principle that lipid-rich adipocytes are more susceptible to freezing than surrounding water-rich cells, allowing selective apoptosis while preserving the adjacent structures. As macrophages digest the apoptotic adipocytes, patients experience a decrease in subcutaneous fat volume over the subsequent 2 to 3 months.10-13 Cryolipolysis has been gaining popularity since 2010, when it was first approved by the US Food and Drug Administration (FDA) for fat reduction in the flank areas; it was later approved for the abdomen in 2012, thighs in 2014, and submental area in 2015.14 Most recently, cryolipolysis was approved for fat reduction in the arms, back, and buttocks in 2016.
The most popular cryolipolysis device applies suction to the treatment area and vacuums the tissue between 2 cooling panels for 30 to 60 minutes.9 Clinical studies investigating the safety and efficacy of cryolipolysis have reported a high degree of patient satisfaction with the procedure and only minimal side effects.4,6,15,16 Common complications of cryolipolysis include erythema, swelling, and sensitivity at the treatment site followed by a lesser incidence of pain, tingling, and bruising, all of which generally resolve within a few weeks of treatment.6 With the removal of adipocytes, there has been concern regarding elevations in blood lipid levels and liver enzymes; however, these laboratory values have been reported to remain within normal limits during and after cryolipolysis.17,18 Of note, patients should be advised of the risk of paradoxical adipose hyperplasia, a rare side effect of cryolipolysis in which a large, demarcated, tender fat mass develops at the treatment site 2 to 3 months after treatment, with an estimated incidence of 1 in 20,000.19 However, the incidence of paradoxical adipose hyperplasia may be underestimated, as a single practice reported an incidence of 0.47% in 422 cryolipolysis treatments.20 This complication has not been associated with any of the heat-induced fat reduction modalities.
Cryolipolysis has been found to be safe for all skin types with no reported pigmentary changes.16 It should not be performed in patients with cold-induced conditions (eg, cryoglobulinemia, cold urticaria) or in those with severe varicose veins or atopic dermatitis.21,22 Patients benefitting most from this procedure are those who require only small or moderate amounts of adipose tissue and cellulite removal with separate fat bulges.12,17 Interestingly, cryolipolysis also has been used off label to treat pseudogynecomastia in male patients.23
Radiofrequency
Radiofrequency has become an important and frequently used modality in cosmetic dermatology.24 This modality differs from cryolipolysis in that it relies on exploiting the difference in water content and impedance between tissues: the skin has low impedance, whereas fat tissue has high impedance. Radiofrequency induces thermal injury to targeted tissue layers, rather than the cold-induced damage seen in cryolipolysis, through devices that focus thermal energy on tissues with high impedance, inducing apoptosis of cells in the subcutaneous adipose tissue with minimal risk of damaging the epidermis, dermis, and muscle.9,25 Ultimately, thermal exposure to 43°C to 45°C over several minutes results in a delayed adipocyte death response.4 In addition to adipocyte death, RF has been shown to cause denaturation of collagen fibrils, leading to subsequent remodeling, neocollagenesis, and skin tightening.26
Radiofrequency devices can be broadly classified as monopolar or bipolar.24,27 Bipolar devices generally require more frequent treatments, whereas monopolar devices tend to require fewer treatment sessions with superior circumference and fat reduction.28
Overall, RF devices have a favorable side effect profile. The most common side effects are erythema and edema at the treatment site lasting less than 24 hours after the procedure.25 The absence of complications such as abdominal discomfort, erythema, and burning during treatment have been reported,27 with the exception of 1 case of hyperesthesia on the abdomen that lasted for 3 days after a treatment session.5 Although RF has beneficial effects on circumference reduction in the abdomen and thighs and can improve the appearance of cellulite, an increase in body weight may occur during treatment. When a localized area of fat such as the thigh is targeted for treatment but the remaining fat cells in the body are not affected, the remaining cells can continue to grow and expand; for instance, although fat cells destroyed with RF will not continue to expand, fat cells in untreated areas may continue to grow due to continued weight gain (eg, from excessive eating), leading to overall weight gain. Thus, patients must understand that weight gain is not an indication of treatment failure after RF or any other method of irreversible fat destruction.5
High-intensity Focused Ultrasound
High-intensity focused ultrasound recently was introduced as a new treatment modality for body contouring, specifically for skin tightening and rejuvenation.5 The mechanism of HIFU is similar to that of RF in that it also relies on heat to cause adipocyte apoptosis; however, it utilizes acoustic energy rather than electric energy. High-intensity focused ultrasound devices can deliver energy to the deep dermis, subdermal connective tissue, and fibromuscular layers in precise microcoagulation zones without damage to the epidermis. The focused energy induces a high temperature (>65°C) within 1 to 3 seconds, causing cell protein coagulation in the targeted area. In addition to its thermal effects, HIFU induces a mechanical effect that disrupts cell membranes immediately, which contributes to the coagulation necrosis process, further promoting necrosis and apoptosis. The effects of these devices can be visualized, as there always is a sharp demarcation between the targeted and untargeted tissue.29 Additionally, microcoagulation is thought to cause gradual skin tightening through collagen contraction and remodeling.30
High-intensity focused ultrasound first received FDA approval for eyebrow lifting and has been used safely and effectively to treat facial and neck skin in a variety of skin types as well as to improve the clinical appearance of the abdomen and thighs.31 This technique is best suited for patients with mild to moderate laxity of the skin or soft tissue who have a body mass index less than 30 kg/m2 and are seeking mild body contouring.32 The ideal patient is young with normal wound healing, since the clinical response to treatment is partly dependent on new collagen synthesis.33 Older patients with extensive photoaging or severe skin laxity are not good candidates for HIFU.
There are a variety of available HIFU devices,34 which utilize special transducers that direct ultrasound energy to a small focal point in the subcutaneous tissues that harmlessly passes through the skin.35 By using newly developed transducers with different energy outputs and focal depths, dermatologists can tailor HIFU treatment to meet the unique physical characteristics of each patient.31
Adverse effects of HIFU are limited to transient pain in most patients and occasional erythema and ecchymosis in some cases.31 In general, most adverse effects resolve spontaneously within 4 weeks and all by 12 weeks posttreatment. Studies also have reported hard subcutaneous nodules, discomfort, burning sensation, mild blisters, and one case of purpuric lesions, all at the treatment site.36-39 There is no evidence that HIFU can cause abnormalities in serum lipids or liver function tests.
Lasers
Laser technology is a rapidly growing modality in noninvasive body contouring. A novel device recently emerged as the first and only FDA-cleared hyperthermic laser for fat reduction and noninvasive body contouring of the abdomen, flanks, back, inner and outer thighs, and submental area.40,41 The device is a 1060-nm diode laser that uses thermal energy to destroy adipose tissue, leading to permanent reduction in stubborn fat without surgery or downtime through the use of a flat, nonsuction applicator that is designed for consistent, natural-looking results. The device includes a contact cooling system that helps to limit thermal discomfort and prevent damage to the surface of the skin during the procedure. Initial improvement can be seen as quickly as 6 weeks posttreatment, and optimal results usually occur in as few as 12 weeks. This device was found to have an excellent safety profile and was well tolerated among patients, with only mild pain reported.42,43
Prior to the development of this new 1060-nm diode laser, the initial application of lasers for noninvasive body contouring involved low-level laser therapy (LLLT), also known as cold laser therapy.40 One device has 5 rotating diode laser heads that work at a wavelength of 635 nm. Treatment sessions last up to 30 minutes, and 6 to 8 sessions are required to obtain optimal results. Low-level laser therapy is a unique modality that is not based on thermal tissue damage, but rather on producing transient microscopic pores in adipocytes that allow lipids to leak out, leading to fat reduction.34 Because LLLT causes immediate emptying of targeted adipocytes, results are noticeable as soon as treatment is completed; however, there is no necrosis or apoptosis of adipocytes, so the recurrence of fat deposition is believed to be greater when compared to the other modalities. Because the results are temporary, long-term or permanent results should not be expected with LLLT. Depending on the patient’s goals, the temporary nature of the results can be either an advantage or disadvantage: some may prefer immediate results despite gradual diminishment over subsequent months, whereas others may prefer results that progressively increase over time and are more permanent, as seen with cryolipolysis, HIFU, and RF.3
Complications of LLLT generally are fewer and more mild than with all other body contouring procedures, with several studies reporting no adverse effects.44-48 Others reported swelling or erythema at the treatment area, pain or tingling during treatment, and increased urination, all of which were temporary and resolved spontaneously.49 Additionally, although the lipids released from treatment are cleared through the lymphatic system, LLLT has not been shown to increase serum lipid levels.50
Conclusion
The field of noninvasive body contouring is undoubtedly growing and will likely continue to rise in popularity as the efficacy and safety of these treatments improve. Although the available technologies vary by mechanism and side effect profiles, several devices have been revealed to be safe and effective in reducing subcutaneous fat tissue and improving skin laxity.1 However, additional studies are needed to evaluate these devices in a standardized manner, especially considering the high costs associated with treatment.32 Current studies investigating these devices vary in treatment protocol, treatment area, number and timing of follow-up sessions, and outcome measures, making it challenging to compare the results objectively.3 Dermatologists offering body contouring treatments need to be intimately familiar with the available devices and determine which treatment is appropriate for each patient in order to provide the highest quality care. Most importantly, patients and physicians must discuss individual goals when choosing a body-contouring method in order to maximize patient satisfaction.
In today’s society there is a ubiquitous pressure to lose weight, reduce fat, and rejuvenate the skin that stems not only from images of idealized bodies in the media but also from our growing knowledge of the detrimental effects of obesity. Along with diet and exercise, it has become popular to use noninvasive devices to attain these goals by means of body contouring—the optimization of the definition, smoothness, and shape of the human physique.1 In fact, body contouring currently is the fastest-growing area of cosmetic dermatology.2
Previously, body contouring primarily involved invasive procedures (eg, liposuction) that are associated with various adverse effects, financial costs, and lengthy downtime.3 More recently, a growing demand for safer and less painful procedures for adipose tissue reduction and skin tightening have led to the development of several novel modalities for noninvasive body contouring. Although the results achieved using these new technologies may be less dramatic than invasive techniques and are not immediate, they do not carry the risks and adverse effects that are associated with surgical procedures and therefore are increasingly requested by cosmetic patients.4,5 New noninvasive techniques primarily target the physical properties of fat, resulting in an efflux of triglycerides from fat cells, causing either reduced size, necrosis, or apoptosis of adipocytes.3,6 Of these modalities, cold-induced adipocyte apoptosis has been commercially available the longest and has been the most researched; however, other noninvasive body contouring techniques have been increasingly explored by researchers since the first reports of human adipose tissue explants exhibiting features of apoptosis after heat injury became available.7,8
There currently are 4 leading modalities used for noninvasive body contouring: cryolipolysis, radiofrequency (RF), high-intensity focused ultrasound (HIFU), and laser therapy (Table). Although no procedure has yet been accepted as the gold standard, investigators are working to determine which technique is the most effective.9 In this article, we provide an overview of these techniques to help dermatologists choose appropriate modalities for their cosmetic patients.

Cryolipolysis
Cryolipolysis is unique in that it employs the principle that lipid-rich adipocytes are more susceptible to freezing than surrounding water-rich cells, allowing selective apoptosis while preserving the adjacent structures. As macrophages digest the apoptotic adipocytes, patients experience a decrease in subcutaneous fat volume over the subsequent 2 to 3 months.10-13 Cryolipolysis has been gaining popularity since 2010, when it was first approved by the US Food and Drug Administration (FDA) for fat reduction in the flank areas; it was later approved for the abdomen in 2012, thighs in 2014, and submental area in 2015.14 Most recently, cryolipolysis was approved for fat reduction in the arms, back, and buttocks in 2016.
The most popular cryolipolysis device applies suction to the treatment area and vacuums the tissue between 2 cooling panels for 30 to 60 minutes.9 Clinical studies investigating the safety and efficacy of cryolipolysis have reported a high degree of patient satisfaction with the procedure and only minimal side effects.4,6,15,16 Common complications of cryolipolysis include erythema, swelling, and sensitivity at the treatment site followed by a lesser incidence of pain, tingling, and bruising, all of which generally resolve within a few weeks of treatment.6 With the removal of adipocytes, there has been concern regarding elevations in blood lipid levels and liver enzymes; however, these laboratory values have been reported to remain within normal limits during and after cryolipolysis.17,18 Of note, patients should be advised of the risk of paradoxical adipose hyperplasia, a rare side effect of cryolipolysis in which a large, demarcated, tender fat mass develops at the treatment site 2 to 3 months after treatment, with an estimated incidence of 1 in 20,000.19 However, the incidence of paradoxical adipose hyperplasia may be underestimated, as a single practice reported an incidence of 0.47% in 422 cryolipolysis treatments.20 This complication has not been associated with any of the heat-induced fat reduction modalities.
Cryolipolysis has been found to be safe for all skin types with no reported pigmentary changes.16 It should not be performed in patients with cold-induced conditions (eg, cryoglobulinemia, cold urticaria) or in those with severe varicose veins or atopic dermatitis.21,22 Patients benefitting most from this procedure are those who require only small or moderate amounts of adipose tissue and cellulite removal with separate fat bulges.12,17 Interestingly, cryolipolysis also has been used off label to treat pseudogynecomastia in male patients.23
Radiofrequency
Radiofrequency has become an important and frequently used modality in cosmetic dermatology.24 This modality differs from cryolipolysis in that it relies on exploiting the difference in water content and impedance between tissues: the skin has low impedance, whereas fat tissue has high impedance. Radiofrequency induces thermal injury to targeted tissue layers, rather than the cold-induced damage seen in cryolipolysis, through devices that focus thermal energy on tissues with high impedance, inducing apoptosis of cells in the subcutaneous adipose tissue with minimal risk of damaging the epidermis, dermis, and muscle.9,25 Ultimately, thermal exposure to 43°C to 45°C over several minutes results in a delayed adipocyte death response.4 In addition to adipocyte death, RF has been shown to cause denaturation of collagen fibrils, leading to subsequent remodeling, neocollagenesis, and skin tightening.26
Radiofrequency devices can be broadly classified as monopolar or bipolar.24,27 Bipolar devices generally require more frequent treatments, whereas monopolar devices tend to require fewer treatment sessions with superior circumference and fat reduction.28
Overall, RF devices have a favorable side effect profile. The most common side effects are erythema and edema at the treatment site lasting less than 24 hours after the procedure.25 The absence of complications such as abdominal discomfort, erythema, and burning during treatment have been reported,27 with the exception of 1 case of hyperesthesia on the abdomen that lasted for 3 days after a treatment session.5 Although RF has beneficial effects on circumference reduction in the abdomen and thighs and can improve the appearance of cellulite, an increase in body weight may occur during treatment. When a localized area of fat such as the thigh is targeted for treatment but the remaining fat cells in the body are not affected, the remaining cells can continue to grow and expand; for instance, although fat cells destroyed with RF will not continue to expand, fat cells in untreated areas may continue to grow due to continued weight gain (eg, from excessive eating), leading to overall weight gain. Thus, patients must understand that weight gain is not an indication of treatment failure after RF or any other method of irreversible fat destruction.5
High-intensity Focused Ultrasound
High-intensity focused ultrasound recently was introduced as a new treatment modality for body contouring, specifically for skin tightening and rejuvenation.5 The mechanism of HIFU is similar to that of RF in that it also relies on heat to cause adipocyte apoptosis; however, it utilizes acoustic energy rather than electric energy. High-intensity focused ultrasound devices can deliver energy to the deep dermis, subdermal connective tissue, and fibromuscular layers in precise microcoagulation zones without damage to the epidermis. The focused energy induces a high temperature (>65°C) within 1 to 3 seconds, causing cell protein coagulation in the targeted area. In addition to its thermal effects, HIFU induces a mechanical effect that disrupts cell membranes immediately, which contributes to the coagulation necrosis process, further promoting necrosis and apoptosis. The effects of these devices can be visualized, as there always is a sharp demarcation between the targeted and untargeted tissue.29 Additionally, microcoagulation is thought to cause gradual skin tightening through collagen contraction and remodeling.30
High-intensity focused ultrasound first received FDA approval for eyebrow lifting and has been used safely and effectively to treat facial and neck skin in a variety of skin types as well as to improve the clinical appearance of the abdomen and thighs.31 This technique is best suited for patients with mild to moderate laxity of the skin or soft tissue who have a body mass index less than 30 kg/m2 and are seeking mild body contouring.32 The ideal patient is young with normal wound healing, since the clinical response to treatment is partly dependent on new collagen synthesis.33 Older patients with extensive photoaging or severe skin laxity are not good candidates for HIFU.
There are a variety of available HIFU devices,34 which utilize special transducers that direct ultrasound energy to a small focal point in the subcutaneous tissues that harmlessly passes through the skin.35 By using newly developed transducers with different energy outputs and focal depths, dermatologists can tailor HIFU treatment to meet the unique physical characteristics of each patient.31
Adverse effects of HIFU are limited to transient pain in most patients and occasional erythema and ecchymosis in some cases.31 In general, most adverse effects resolve spontaneously within 4 weeks and all by 12 weeks posttreatment. Studies also have reported hard subcutaneous nodules, discomfort, burning sensation, mild blisters, and one case of purpuric lesions, all at the treatment site.36-39 There is no evidence that HIFU can cause abnormalities in serum lipids or liver function tests.
Lasers
Laser technology is a rapidly growing modality in noninvasive body contouring. A novel device recently emerged as the first and only FDA-cleared hyperthermic laser for fat reduction and noninvasive body contouring of the abdomen, flanks, back, inner and outer thighs, and submental area.40,41 The device is a 1060-nm diode laser that uses thermal energy to destroy adipose tissue, leading to permanent reduction in stubborn fat without surgery or downtime through the use of a flat, nonsuction applicator that is designed for consistent, natural-looking results. The device includes a contact cooling system that helps to limit thermal discomfort and prevent damage to the surface of the skin during the procedure. Initial improvement can be seen as quickly as 6 weeks posttreatment, and optimal results usually occur in as few as 12 weeks. This device was found to have an excellent safety profile and was well tolerated among patients, with only mild pain reported.42,43
Prior to the development of this new 1060-nm diode laser, the initial application of lasers for noninvasive body contouring involved low-level laser therapy (LLLT), also known as cold laser therapy.40 One device has 5 rotating diode laser heads that work at a wavelength of 635 nm. Treatment sessions last up to 30 minutes, and 6 to 8 sessions are required to obtain optimal results. Low-level laser therapy is a unique modality that is not based on thermal tissue damage, but rather on producing transient microscopic pores in adipocytes that allow lipids to leak out, leading to fat reduction.34 Because LLLT causes immediate emptying of targeted adipocytes, results are noticeable as soon as treatment is completed; however, there is no necrosis or apoptosis of adipocytes, so the recurrence of fat deposition is believed to be greater when compared to the other modalities. Because the results are temporary, long-term or permanent results should not be expected with LLLT. Depending on the patient’s goals, the temporary nature of the results can be either an advantage or disadvantage: some may prefer immediate results despite gradual diminishment over subsequent months, whereas others may prefer results that progressively increase over time and are more permanent, as seen with cryolipolysis, HIFU, and RF.3
Complications of LLLT generally are fewer and more mild than with all other body contouring procedures, with several studies reporting no adverse effects.44-48 Others reported swelling or erythema at the treatment area, pain or tingling during treatment, and increased urination, all of which were temporary and resolved spontaneously.49 Additionally, although the lipids released from treatment are cleared through the lymphatic system, LLLT has not been shown to increase serum lipid levels.50
Conclusion
The field of noninvasive body contouring is undoubtedly growing and will likely continue to rise in popularity as the efficacy and safety of these treatments improve. Although the available technologies vary by mechanism and side effect profiles, several devices have been revealed to be safe and effective in reducing subcutaneous fat tissue and improving skin laxity.1 However, additional studies are needed to evaluate these devices in a standardized manner, especially considering the high costs associated with treatment.32 Current studies investigating these devices vary in treatment protocol, treatment area, number and timing of follow-up sessions, and outcome measures, making it challenging to compare the results objectively.3 Dermatologists offering body contouring treatments need to be intimately familiar with the available devices and determine which treatment is appropriate for each patient in order to provide the highest quality care. Most importantly, patients and physicians must discuss individual goals when choosing a body-contouring method in order to maximize patient satisfaction.
- Jalian HR, Avram MM. Body contouring: the skinny on noninvasive fat removal. Semin Cutan Med Surg. 2012;31:121-125.
- Ho D, Jagdeo J. A systematic review of paradoxical adipose hyperplasia (PAH) post-cryolipolysis. J Drugs Dermatol. 2017;16:62-67.
- Kennedy J, Verne S, Griffith R, et al. Non-invasive subcutaneous fat reduction: a review. J Eur Acad Dermatol Venereol. 2015;29:1679-1688.
- Krueger N, Mai SV, Luebberding S, et al. Cryolipolysis for noninvasive body contouring: clinical efficacy and patient satisfaction. Clin Cosmet Investig Dermatol. 2014;7:201-205.
- Suh DH, Kim CM, Lee SJ, et al. Safety and efficacy of a non-contact radiofrequency device for body contouring in Asians. J Cosmet Laser Ther. 2017;19:89-92.
- Ingargiola MJ, Motakef S, Chung MT, et al. Cryolipolysis for fat reduction and body contouring: safety and efficacy of current treatment paradigms. Plast Reconstr Surg. 2015;135:1581-1590.
- Prins JB, Walker NI, Winterford CM, et al. Apoptosis of human adipocytes in vitro. Biochem Biophys Res Commun. 1994;201:500-507.
- Sorisky A, Magun R, Gagnon AM. Adipose cell apoptosis: death in the energy depot. Int J Obes Relat Metab Disord. 2000;24(suppl 4):S3-S7.
- Chilukuri S, Mueller G. “Hands-free” noninvasive body contouring devices: review of effectiveness and patient satisfaction. J Drugs Dermatol. 2016;15:1402-1406.
- Manstein D, Laubach H, Watanabe K, et al. Selective cryolysis: a novel method of non-invasive fat removal. Lasers Surg Med. 2008;40:595-604.
- Zelickson B, Egbert BM, Preciado J, et al. Cryolipolysis for noninvasive fat cell destruction: initial results from a pig model. Dermatol Surg. 2009;35:1462-1470.
- Nelson AA, Wasserman D, Avram MM. Cryolipolysis for reduction of excess adipose tissue. Semin Cutan Med Surg. 2009;28:244-249.
- Avram MM, Harry RS. Cryolipolysis for subcutaneous fat layer reduction. Lasers Surg Med. 2009;41:703-708.
- Klein KB, Bachelor EP, Becker EV, et al. Multiple same day cryolipolysis treatments for the reduction of subcutaneous fat are safe and do not affect serum lipid levels or liver function tests. Lasers Surg Med. 2017;49:640-644.
- Dierickx CC, Mazer JM, Sand M, et al. Safety, tolerance, and patient satisfaction with noninvasive cryolipolysis. Dermatol Surg. 2013;39:1209-1216.
- Stevens WG, Pietrzak LK, Spring MA. Broad overview of a clinical and commercial experience with CoolSculpting. Aesthet Surg J. 2013;33:835-846.
- Ferraro GA, De Francesco F, Cataldo C, et al. Synergistic effects of cryolipolysis and shock waves for noninvasive body contouring. Aesthetic Plast Surg. 2012;36:666-679.
- Lee KR. Clinical efficacy of fat reduction on the thigh of Korean women through cryolipolysis. J Obes Weight Loss Ther. 2013;3:203.
- Jalian HR, Avram MM, Garibyan L, et al. Paradoxical adipose hyperplasia after cryolipolysis. JAMA Dermatol. 2014;150:317-319.
- Singh SM, Geddes ER, Boutrous SG, et al. Paradoxical adipose hyperplasia secondary to cryolipolysis: an underreported entity? Lasers Surg Med. 2015;47:476-478.
- Pinto H, Arredondo E, Ricart-Jane D. Evaluation of adipocytic changes after a simil-lipocryolysis stimulus. Cryo Letters. 2013;34:100-105.
- Pinto HR, Garcia-Cruz E, Melamed GE. A study to evaluate the action of lipocryolysis. Cryo Letters. 2012;33:177-181.
- Singh B, Keaney T, Rossi AM. Male body contouring. J Drugs Dermatol. 2015;14:1052-1059.
- Beasley KL, Weiss RA. Radiofrequency in cosmetic dermatology. Dermatol Clin. 2014;32:79-90.
- Weiss R, Weiss M, Beasley K, et al. Operator independent focused high frequency ISM band for fat reduction: porcine model. Lasers Surg Med. 2013;45:235-239.
- Hantash BM, Ubeid AA, Chang H, et al. Bipolar fractional radiofrequency treatment induces neoelastogenesis and neocollagenesis. Lasers Surg Med. 2009;41:1-9.
- Harth Y. Painless, safe, and efficacious noninvasive skin tightening, body contouring, and cellulite reduction using multisource 3DEEP radiofrequency. J Cosmet Dermatol. 2015;14:70-75.
- Nassab R. The evidence behind noninvasive body contouring devices. Aesthet Surg J. 2015;35:279-293.
- Luo W, Zhou X, Gong X, et al. Study of sequential histopathologic changes, apoptosis, and cell proliferation in rabbit livers after high-intensity focused ultrasound ablation. J Ultrasound Med. 2007;26:477-485.
- Minkis K, Alam M. Ultrasound skin tightening. Dermatol Clin. 2014;32:71-77.
- Ko EJ, Hong JY, Kwon TR, et al. Efficacy and safety of non-invasive body tightening with high-intensity focused ultrasound (HIFU). Skin Res Technol. 2017;23:558-562.
- Sklar LR, El Tal AK, Kerwin LY. Use of transcutaneous ultrasound for lipolysis and skin tightening: a review. Aesthetic Plast Surg. 2014;38:429-441.
- MacGregor JL, Tanzi EL. Microfocused ultrasound for skin tightening. Semin Cutan Med Surg. 2013;32:18-25.
- Alizadeh Z, Halabchi F, Mazaheri R, et al. Review of the mechanisms and effects of noninvasive body contouring devices on cellulite and subcutaneous fat. Int J Endocrinol Metab. 2016;14:E36727 .
- Fabi SG. Noninvasive skin tightening: focus on new ultrasound techniques. Clin Cosmet Investig Dermatol. 2015;8:47-52.
- Fatemi A. High-intensity focused ultrasound effectively reduces adipose tissue. Semin Cutan Med Surg. 2009;28:257-262.
- Teitelbaum SA, Burns JL, Kubota J, et al. Noninvasive body contouring by focused ultrasound: safety and efficacy of the Contour I device in a multicenter, controlled, clinical study. Plast Reconstr Surg. 2007;120:779-789.
- Hotta TA. Nonsurgical body contouring with focused ultrasound. Plast Surg Nurs. 2010;30:77-82; quiz 83-84.
- Fatemi A, Kane MA. High-intensity focused ultrasound effectively reduces waist circumference by ablating adipose tissue from the abdomen and flanks: a retrospective case series. Aesthetic Plast Surg. 2010;34:577-582.
- Schilling L, Saedi N, Weiss R. 1060 nm diode hyperthermic laser lipolysis: the latest in non-invasive body contouring. J Drugs Dermatol. 2017;16:48-52.
- Body contouring. CynoSure website. https://www.cynosure.com/treatment/body-contouring/SculpSure. Accessed March 28, 2018.
- Decorato JW, Chen B, Sierra R. Subcutaneous adipose tissue response to a non-invasive hyperthermic treatment using a 1,060 nm laser. Lasers Surg Med. 2017;49:480-489.
- Weiss R, McDaniel D, Doherty S. Clinical evaluation of fat reduction treatment of the flanks and abdomen with a non-invasive 1060 nm diode laser: a multicenter study. Paper presented at: 2016 Annual American Society for Laser Medicine and Surgery Conference; March 30–April 3, 2016; Boston, MA.
- Caruso-Davis MK, Guillot TS, Podichetty VK, et al. Efficacy of low-level laser therapy for body contouring and spot fat reduction. Obes Surg. 2011;21:722-729.
- McRae E, Boris J. Independent evaluation of low-level laser therapy at 635 nm for non-invasive body contouring of the waist, hips, and thighs. Lasers Surg Med. 2013;45:1-7.
- Nestor MS, Newburger J, Zarraga MB. Body contouring using 635-nm low level laser therapy. Semin Cutan Med Surg. 2013;32:35-40.
- Jackson RF, Stern FA, Neira R, et al. Application of low-level laser therapy for noninvasive body contouring. Lasers Surg Med. 2012;44:211-217.
- Jackson RF, Dedo DD, Roche GC, et al. Low-level laser therapy as a non-invasive approach for body contouring: a randomized, controlled study. Lasers Surg Med. 2009;41:799-809.
- Gold MH, Khatri KA, Hails K, et al. Reduction in thigh circumference and improvement in the appearance of cellulite with dual-wavelength, low-level laser energy and massage. J Cosmet Laser Ther. 2011;13:13-20.
- Avci P, Nyame TT, Gupta GK, et al. Low-level laser therapy for fat layer reduction: a comprehensive review. Lasers Surg Med. 2013;45:349-357.
- Jalian HR, Avram MM. Body contouring: the skinny on noninvasive fat removal. Semin Cutan Med Surg. 2012;31:121-125.
- Ho D, Jagdeo J. A systematic review of paradoxical adipose hyperplasia (PAH) post-cryolipolysis. J Drugs Dermatol. 2017;16:62-67.
- Kennedy J, Verne S, Griffith R, et al. Non-invasive subcutaneous fat reduction: a review. J Eur Acad Dermatol Venereol. 2015;29:1679-1688.
- Krueger N, Mai SV, Luebberding S, et al. Cryolipolysis for noninvasive body contouring: clinical efficacy and patient satisfaction. Clin Cosmet Investig Dermatol. 2014;7:201-205.
- Suh DH, Kim CM, Lee SJ, et al. Safety and efficacy of a non-contact radiofrequency device for body contouring in Asians. J Cosmet Laser Ther. 2017;19:89-92.
- Ingargiola MJ, Motakef S, Chung MT, et al. Cryolipolysis for fat reduction and body contouring: safety and efficacy of current treatment paradigms. Plast Reconstr Surg. 2015;135:1581-1590.
- Prins JB, Walker NI, Winterford CM, et al. Apoptosis of human adipocytes in vitro. Biochem Biophys Res Commun. 1994;201:500-507.
- Sorisky A, Magun R, Gagnon AM. Adipose cell apoptosis: death in the energy depot. Int J Obes Relat Metab Disord. 2000;24(suppl 4):S3-S7.
- Chilukuri S, Mueller G. “Hands-free” noninvasive body contouring devices: review of effectiveness and patient satisfaction. J Drugs Dermatol. 2016;15:1402-1406.
- Manstein D, Laubach H, Watanabe K, et al. Selective cryolysis: a novel method of non-invasive fat removal. Lasers Surg Med. 2008;40:595-604.
- Zelickson B, Egbert BM, Preciado J, et al. Cryolipolysis for noninvasive fat cell destruction: initial results from a pig model. Dermatol Surg. 2009;35:1462-1470.
- Nelson AA, Wasserman D, Avram MM. Cryolipolysis for reduction of excess adipose tissue. Semin Cutan Med Surg. 2009;28:244-249.
- Avram MM, Harry RS. Cryolipolysis for subcutaneous fat layer reduction. Lasers Surg Med. 2009;41:703-708.
- Klein KB, Bachelor EP, Becker EV, et al. Multiple same day cryolipolysis treatments for the reduction of subcutaneous fat are safe and do not affect serum lipid levels or liver function tests. Lasers Surg Med. 2017;49:640-644.
- Dierickx CC, Mazer JM, Sand M, et al. Safety, tolerance, and patient satisfaction with noninvasive cryolipolysis. Dermatol Surg. 2013;39:1209-1216.
- Stevens WG, Pietrzak LK, Spring MA. Broad overview of a clinical and commercial experience with CoolSculpting. Aesthet Surg J. 2013;33:835-846.
- Ferraro GA, De Francesco F, Cataldo C, et al. Synergistic effects of cryolipolysis and shock waves for noninvasive body contouring. Aesthetic Plast Surg. 2012;36:666-679.
- Lee KR. Clinical efficacy of fat reduction on the thigh of Korean women through cryolipolysis. J Obes Weight Loss Ther. 2013;3:203.
- Jalian HR, Avram MM, Garibyan L, et al. Paradoxical adipose hyperplasia after cryolipolysis. JAMA Dermatol. 2014;150:317-319.
- Singh SM, Geddes ER, Boutrous SG, et al. Paradoxical adipose hyperplasia secondary to cryolipolysis: an underreported entity? Lasers Surg Med. 2015;47:476-478.
- Pinto H, Arredondo E, Ricart-Jane D. Evaluation of adipocytic changes after a simil-lipocryolysis stimulus. Cryo Letters. 2013;34:100-105.
- Pinto HR, Garcia-Cruz E, Melamed GE. A study to evaluate the action of lipocryolysis. Cryo Letters. 2012;33:177-181.
- Singh B, Keaney T, Rossi AM. Male body contouring. J Drugs Dermatol. 2015;14:1052-1059.
- Beasley KL, Weiss RA. Radiofrequency in cosmetic dermatology. Dermatol Clin. 2014;32:79-90.
- Weiss R, Weiss M, Beasley K, et al. Operator independent focused high frequency ISM band for fat reduction: porcine model. Lasers Surg Med. 2013;45:235-239.
- Hantash BM, Ubeid AA, Chang H, et al. Bipolar fractional radiofrequency treatment induces neoelastogenesis and neocollagenesis. Lasers Surg Med. 2009;41:1-9.
- Harth Y. Painless, safe, and efficacious noninvasive skin tightening, body contouring, and cellulite reduction using multisource 3DEEP radiofrequency. J Cosmet Dermatol. 2015;14:70-75.
- Nassab R. The evidence behind noninvasive body contouring devices. Aesthet Surg J. 2015;35:279-293.
- Luo W, Zhou X, Gong X, et al. Study of sequential histopathologic changes, apoptosis, and cell proliferation in rabbit livers after high-intensity focused ultrasound ablation. J Ultrasound Med. 2007;26:477-485.
- Minkis K, Alam M. Ultrasound skin tightening. Dermatol Clin. 2014;32:71-77.
- Ko EJ, Hong JY, Kwon TR, et al. Efficacy and safety of non-invasive body tightening with high-intensity focused ultrasound (HIFU). Skin Res Technol. 2017;23:558-562.
- Sklar LR, El Tal AK, Kerwin LY. Use of transcutaneous ultrasound for lipolysis and skin tightening: a review. Aesthetic Plast Surg. 2014;38:429-441.
- MacGregor JL, Tanzi EL. Microfocused ultrasound for skin tightening. Semin Cutan Med Surg. 2013;32:18-25.
- Alizadeh Z, Halabchi F, Mazaheri R, et al. Review of the mechanisms and effects of noninvasive body contouring devices on cellulite and subcutaneous fat. Int J Endocrinol Metab. 2016;14:E36727 .
- Fabi SG. Noninvasive skin tightening: focus on new ultrasound techniques. Clin Cosmet Investig Dermatol. 2015;8:47-52.
- Fatemi A. High-intensity focused ultrasound effectively reduces adipose tissue. Semin Cutan Med Surg. 2009;28:257-262.
- Teitelbaum SA, Burns JL, Kubota J, et al. Noninvasive body contouring by focused ultrasound: safety and efficacy of the Contour I device in a multicenter, controlled, clinical study. Plast Reconstr Surg. 2007;120:779-789.
- Hotta TA. Nonsurgical body contouring with focused ultrasound. Plast Surg Nurs. 2010;30:77-82; quiz 83-84.
- Fatemi A, Kane MA. High-intensity focused ultrasound effectively reduces waist circumference by ablating adipose tissue from the abdomen and flanks: a retrospective case series. Aesthetic Plast Surg. 2010;34:577-582.
- Schilling L, Saedi N, Weiss R. 1060 nm diode hyperthermic laser lipolysis: the latest in non-invasive body contouring. J Drugs Dermatol. 2017;16:48-52.
- Body contouring. CynoSure website. https://www.cynosure.com/treatment/body-contouring/SculpSure. Accessed March 28, 2018.
- Decorato JW, Chen B, Sierra R. Subcutaneous adipose tissue response to a non-invasive hyperthermic treatment using a 1,060 nm laser. Lasers Surg Med. 2017;49:480-489.
- Weiss R, McDaniel D, Doherty S. Clinical evaluation of fat reduction treatment of the flanks and abdomen with a non-invasive 1060 nm diode laser: a multicenter study. Paper presented at: 2016 Annual American Society for Laser Medicine and Surgery Conference; March 30–April 3, 2016; Boston, MA.
- Caruso-Davis MK, Guillot TS, Podichetty VK, et al. Efficacy of low-level laser therapy for body contouring and spot fat reduction. Obes Surg. 2011;21:722-729.
- McRae E, Boris J. Independent evaluation of low-level laser therapy at 635 nm for non-invasive body contouring of the waist, hips, and thighs. Lasers Surg Med. 2013;45:1-7.
- Nestor MS, Newburger J, Zarraga MB. Body contouring using 635-nm low level laser therapy. Semin Cutan Med Surg. 2013;32:35-40.
- Jackson RF, Stern FA, Neira R, et al. Application of low-level laser therapy for noninvasive body contouring. Lasers Surg Med. 2012;44:211-217.
- Jackson RF, Dedo DD, Roche GC, et al. Low-level laser therapy as a non-invasive approach for body contouring: a randomized, controlled study. Lasers Surg Med. 2009;41:799-809.
- Gold MH, Khatri KA, Hails K, et al. Reduction in thigh circumference and improvement in the appearance of cellulite with dual-wavelength, low-level laser energy and massage. J Cosmet Laser Ther. 2011;13:13-20.
- Avci P, Nyame TT, Gupta GK, et al. Low-level laser therapy for fat layer reduction: a comprehensive review. Lasers Surg Med. 2013;45:349-357.
Practice Points
- There currently are 4 leading modalities used for noninvasive body contouring: cryolipolysis, radiofrequency, high-intensity focused ultrasound, and laser therapy.
- Devices utilizing these 4 modalities have been found to be safe and effective in reducing subcutaneous fat tissue and improving skin laxity.
- Dermatologists utilizing body contouring treatments need to be familiar with available devices to determine which treatment is appropriate for each patient.
Microneedling improved acne scars in small study of patients with darker skin
, and did not contribute to more pigmentation, the study authors reported.
Most patients were pleased with the results. “Microneedling is an effective and safe treatment for acne scars associated with pigmentation in dark-skinned patients, without adding any risk of causing worsening of pigmentation,” the study’s lead author, Firas Al Qarqaz, MD said in an interview.
The study was published online in the Journal of Cosmetic Dermatology.
Dr. Al Qarqaz, of the department of dermatology, Jordan University of Science and Technology, Irbid, Jordan, pointed out that patients with darker skin and acne scars pose a unique challenge because some current treatments “can improve the scars but carry a risk of worsening the pigmentation and making skin/scars darker, which can be as troublesome to patients as their original scars.” Indeed, a review of microneedling as a treatment for dermatologic conditions in patients with darker skin noted that conventional resurfacing procedures can be limited in this patient population, because of concerns of adverse effects, including dyspigmentation (J Am Acad Dermatol. 2016 Feb;74[2]:348-55).
The situation is especially complex because “the current assessment methods for evaluating acne scars are not addressing clearly the important aspect of pigmentation that is associated with such scars, especially in darker skin, which can make objective assessment for improvement lacking,” Dr. Al Qarqaz noted.
He and a coauthor conducted the new study to determine whether microneedling can safely and effectively improve both acne scars and related hyperpigmentation in patients with darker skin. The study of 39 patients with postacne scarring comprised 31 women and 8 men aged 18-43 years (mean age, 27); their skin colors ranged from Fitzpatrick skin types type III to V. Most (27) were type IV.
The patients were treated with an electronic microneedling device and were evaluated at 2 weeks, and at least 4 weeks after their initial assessment (range, 4-14 weeks) for the final evaluation. The researchers found statistically significant improvement in two measures: The Postacne Hyperpigmentation Index improved from a mean score of 13 at baseline to a mean of 10 post procedure (P = .0035), and the Goodman-Baron acne scarring scale improved from a mean of 18 at baseline to a mean of 12 post procedure (P = .008).
Side effects were mild and temporary. “This treatment seems to be safe apart from short-lived erythema and occasional small hematoma following procedure,” the researchers concluded.
Nearly 80% of patients said they were satisfied with the procedure; the rest were not satisfied with the results (no reasons were cited). “Additional studies focusing on postacne scarring with hyperpigmentation are needed,” in addition to “assessment tools designed for this particular patient group,” the authors noted. They added that more treatments may be needed in some patients for hyperpigmentation.
Jordan University of Science and Technology funded the study. The study authors reported no relevant disclosures.
SOURCE: Al-Qarqaz F et al. J Cosmet Dermatol. 2018 Mar 15. (doi: 10.1111/jocd.12520.)
, and did not contribute to more pigmentation, the study authors reported.
Most patients were pleased with the results. “Microneedling is an effective and safe treatment for acne scars associated with pigmentation in dark-skinned patients, without adding any risk of causing worsening of pigmentation,” the study’s lead author, Firas Al Qarqaz, MD said in an interview.
The study was published online in the Journal of Cosmetic Dermatology.
Dr. Al Qarqaz, of the department of dermatology, Jordan University of Science and Technology, Irbid, Jordan, pointed out that patients with darker skin and acne scars pose a unique challenge because some current treatments “can improve the scars but carry a risk of worsening the pigmentation and making skin/scars darker, which can be as troublesome to patients as their original scars.” Indeed, a review of microneedling as a treatment for dermatologic conditions in patients with darker skin noted that conventional resurfacing procedures can be limited in this patient population, because of concerns of adverse effects, including dyspigmentation (J Am Acad Dermatol. 2016 Feb;74[2]:348-55).
The situation is especially complex because “the current assessment methods for evaluating acne scars are not addressing clearly the important aspect of pigmentation that is associated with such scars, especially in darker skin, which can make objective assessment for improvement lacking,” Dr. Al Qarqaz noted.
He and a coauthor conducted the new study to determine whether microneedling can safely and effectively improve both acne scars and related hyperpigmentation in patients with darker skin. The study of 39 patients with postacne scarring comprised 31 women and 8 men aged 18-43 years (mean age, 27); their skin colors ranged from Fitzpatrick skin types type III to V. Most (27) were type IV.
The patients were treated with an electronic microneedling device and were evaluated at 2 weeks, and at least 4 weeks after their initial assessment (range, 4-14 weeks) for the final evaluation. The researchers found statistically significant improvement in two measures: The Postacne Hyperpigmentation Index improved from a mean score of 13 at baseline to a mean of 10 post procedure (P = .0035), and the Goodman-Baron acne scarring scale improved from a mean of 18 at baseline to a mean of 12 post procedure (P = .008).
Side effects were mild and temporary. “This treatment seems to be safe apart from short-lived erythema and occasional small hematoma following procedure,” the researchers concluded.
Nearly 80% of patients said they were satisfied with the procedure; the rest were not satisfied with the results (no reasons were cited). “Additional studies focusing on postacne scarring with hyperpigmentation are needed,” in addition to “assessment tools designed for this particular patient group,” the authors noted. They added that more treatments may be needed in some patients for hyperpigmentation.
Jordan University of Science and Technology funded the study. The study authors reported no relevant disclosures.
SOURCE: Al-Qarqaz F et al. J Cosmet Dermatol. 2018 Mar 15. (doi: 10.1111/jocd.12520.)
, and did not contribute to more pigmentation, the study authors reported.
Most patients were pleased with the results. “Microneedling is an effective and safe treatment for acne scars associated with pigmentation in dark-skinned patients, without adding any risk of causing worsening of pigmentation,” the study’s lead author, Firas Al Qarqaz, MD said in an interview.
The study was published online in the Journal of Cosmetic Dermatology.
Dr. Al Qarqaz, of the department of dermatology, Jordan University of Science and Technology, Irbid, Jordan, pointed out that patients with darker skin and acne scars pose a unique challenge because some current treatments “can improve the scars but carry a risk of worsening the pigmentation and making skin/scars darker, which can be as troublesome to patients as their original scars.” Indeed, a review of microneedling as a treatment for dermatologic conditions in patients with darker skin noted that conventional resurfacing procedures can be limited in this patient population, because of concerns of adverse effects, including dyspigmentation (J Am Acad Dermatol. 2016 Feb;74[2]:348-55).
The situation is especially complex because “the current assessment methods for evaluating acne scars are not addressing clearly the important aspect of pigmentation that is associated with such scars, especially in darker skin, which can make objective assessment for improvement lacking,” Dr. Al Qarqaz noted.
He and a coauthor conducted the new study to determine whether microneedling can safely and effectively improve both acne scars and related hyperpigmentation in patients with darker skin. The study of 39 patients with postacne scarring comprised 31 women and 8 men aged 18-43 years (mean age, 27); their skin colors ranged from Fitzpatrick skin types type III to V. Most (27) were type IV.
The patients were treated with an electronic microneedling device and were evaluated at 2 weeks, and at least 4 weeks after their initial assessment (range, 4-14 weeks) for the final evaluation. The researchers found statistically significant improvement in two measures: The Postacne Hyperpigmentation Index improved from a mean score of 13 at baseline to a mean of 10 post procedure (P = .0035), and the Goodman-Baron acne scarring scale improved from a mean of 18 at baseline to a mean of 12 post procedure (P = .008).
Side effects were mild and temporary. “This treatment seems to be safe apart from short-lived erythema and occasional small hematoma following procedure,” the researchers concluded.
Nearly 80% of patients said they were satisfied with the procedure; the rest were not satisfied with the results (no reasons were cited). “Additional studies focusing on postacne scarring with hyperpigmentation are needed,” in addition to “assessment tools designed for this particular patient group,” the authors noted. They added that more treatments may be needed in some patients for hyperpigmentation.
Jordan University of Science and Technology funded the study. The study authors reported no relevant disclosures.
SOURCE: Al-Qarqaz F et al. J Cosmet Dermatol. 2018 Mar 15. (doi: 10.1111/jocd.12520.)
FROM THE JOURNAL OF COSMETIC DERMATOLOGY
Key clinical point: Microneedling produced statistically significant improvements without worsening pigmentation in darker-skinned patients with postacne scarring.
Major finding: The postacne hyperpigmentation index score improved from a mean of 13 to 10 (P = .0035) and the acne scarring scale improved from 18 to 12 (P = .008).
Study details: Microneedling was used to treat postacne scarring in 31 women and 8 men with Fitzpatrick skin types III-V.
Disclosures: Jordan University of Science and Technology funded the study. The study authors reported no relevant disclosures.
Source: Al Qarqaz F et al. J Cosmet Dermatol. 2018 Mar 15. doi: 10.1111/jocd.12520.