User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
Analysis examines glossectomy as solo treatment for sleep apnea
CORONADO, CALIF. – Results from a new meta-analysis suggest that glossectomy significantly improves sleep outcomes when performed as part of a multi-level surgery for adults with obstructive sleep apnea.
However, “there is insufficient evidence to analyze the role of glossectomy as a stand-alone procedure for the treatment of sleep apnea,” lead study author Dr. Alexander W. Murphey said in an interview in advance of The Triological Society’s Combined Sections meeting, where the work was presented. “The lack of available data for glossectomy as a single treatment was disappointing, and points to the need for further studies in this population.”
Glossectomy for OSA was first reported in 1991 as a salvage surgery after uvulopalatopharyngoplasty, said Dr. Murphey, who is completing a clinical research fellowship in the department of otolaryngology-head and neck surgery at Medical University of South Carolina, Charleston, under the mentoring of Dr. Marion B. Gillespie and Dr. Shaun A. Nguyen. Since that time, “many modifications have been made regarding technique and instrumentation with recent focus on minimally invasive techniques aimed at maximizing tissue reduction while limiting the inherent morbidity associated with glossectomy,” he said. “The aim of our study was to review and analyze all of the available literature on partial glossectomy for OSA in one study. Overall, there is a significant lack of research into glossectomy, and what literature is available include small, case-series that analyze glossectomy as part of complex, multi-level surgeries. This study represents the first large scale meta-analysis on the role of glossectomy, and attempts to determine the role of glossectomy both as part of multi-level surgery, and as a single, stand-alone sleep apnea treatment.”
Dr. Murphey used the PubMed-NCBI literature database to identify studies with 10 or more patients and reported preoperative and postoperative apnea-hypopnea index (AHI) scores. The primary endpoint was change in AHI while secondary endpoints included predefined surgical success rates, and changes in additional reported sleep outcomes such as Epworth Sleep Scores (ESS), Lowest Oxygen Saturation (LSAT), and snoring visual analog scale (VAS). The researchers reported results from 15 articles with 442 patients treated with three glossectomy techniques (midline glossectomy, lingualplasty, and submucosal minimally invasive lingual excision (SMILE). In pooled analyses that compared baseline vs. post-surgery, investigators observed significant reductions in AHI (from 48 to 20); ESS (from 12 to 5), and VAS (from 9 to 3; all with a P of less than .0001). In addition, they observed a significant increase in LSAT (from 77% to 84%; P less than .0001), according to the findings, presented at the meeting, which was jointly sponsored by the Triological Society and the American College of Surgeons.
Surgical success, which was defined as an AHI less than 20 and a greater than 50% reduction in AHI, was achieved in 56% of cases, while complications occurred in 18% of patients. Only 24 patients (5%) were treated with glossectomy as sole therapy for OSA. Among these 24 patients, significant reductions occurred in AHI (from 42 to 25; P=.0345) and ESS (from 12 to 7; P less than .0001).
Dr. Murphey acknowledged certain limitations of the analysis, including “a lack of quality research involving glossectomy, with the majority of available published data drawn from small, case-series without control arms,” he said. “Additionally, studies varied in surgical approach, inclusion criteria, and accompanying procedures in multi-level treatment. This makes it extremely difficult to truly compare treatments.”
The researchers reported having no relevant financial conflicts.
On Twitter @dougbrunk
CORONADO, CALIF. – Results from a new meta-analysis suggest that glossectomy significantly improves sleep outcomes when performed as part of a multi-level surgery for adults with obstructive sleep apnea.
However, “there is insufficient evidence to analyze the role of glossectomy as a stand-alone procedure for the treatment of sleep apnea,” lead study author Dr. Alexander W. Murphey said in an interview in advance of The Triological Society’s Combined Sections meeting, where the work was presented. “The lack of available data for glossectomy as a single treatment was disappointing, and points to the need for further studies in this population.”
Glossectomy for OSA was first reported in 1991 as a salvage surgery after uvulopalatopharyngoplasty, said Dr. Murphey, who is completing a clinical research fellowship in the department of otolaryngology-head and neck surgery at Medical University of South Carolina, Charleston, under the mentoring of Dr. Marion B. Gillespie and Dr. Shaun A. Nguyen. Since that time, “many modifications have been made regarding technique and instrumentation with recent focus on minimally invasive techniques aimed at maximizing tissue reduction while limiting the inherent morbidity associated with glossectomy,” he said. “The aim of our study was to review and analyze all of the available literature on partial glossectomy for OSA in one study. Overall, there is a significant lack of research into glossectomy, and what literature is available include small, case-series that analyze glossectomy as part of complex, multi-level surgeries. This study represents the first large scale meta-analysis on the role of glossectomy, and attempts to determine the role of glossectomy both as part of multi-level surgery, and as a single, stand-alone sleep apnea treatment.”
Dr. Murphey used the PubMed-NCBI literature database to identify studies with 10 or more patients and reported preoperative and postoperative apnea-hypopnea index (AHI) scores. The primary endpoint was change in AHI while secondary endpoints included predefined surgical success rates, and changes in additional reported sleep outcomes such as Epworth Sleep Scores (ESS), Lowest Oxygen Saturation (LSAT), and snoring visual analog scale (VAS). The researchers reported results from 15 articles with 442 patients treated with three glossectomy techniques (midline glossectomy, lingualplasty, and submucosal minimally invasive lingual excision (SMILE). In pooled analyses that compared baseline vs. post-surgery, investigators observed significant reductions in AHI (from 48 to 20); ESS (from 12 to 5), and VAS (from 9 to 3; all with a P of less than .0001). In addition, they observed a significant increase in LSAT (from 77% to 84%; P less than .0001), according to the findings, presented at the meeting, which was jointly sponsored by the Triological Society and the American College of Surgeons.
Surgical success, which was defined as an AHI less than 20 and a greater than 50% reduction in AHI, was achieved in 56% of cases, while complications occurred in 18% of patients. Only 24 patients (5%) were treated with glossectomy as sole therapy for OSA. Among these 24 patients, significant reductions occurred in AHI (from 42 to 25; P=.0345) and ESS (from 12 to 7; P less than .0001).
Dr. Murphey acknowledged certain limitations of the analysis, including “a lack of quality research involving glossectomy, with the majority of available published data drawn from small, case-series without control arms,” he said. “Additionally, studies varied in surgical approach, inclusion criteria, and accompanying procedures in multi-level treatment. This makes it extremely difficult to truly compare treatments.”
The researchers reported having no relevant financial conflicts.
On Twitter @dougbrunk
CORONADO, CALIF. – Results from a new meta-analysis suggest that glossectomy significantly improves sleep outcomes when performed as part of a multi-level surgery for adults with obstructive sleep apnea.
However, “there is insufficient evidence to analyze the role of glossectomy as a stand-alone procedure for the treatment of sleep apnea,” lead study author Dr. Alexander W. Murphey said in an interview in advance of The Triological Society’s Combined Sections meeting, where the work was presented. “The lack of available data for glossectomy as a single treatment was disappointing, and points to the need for further studies in this population.”
Glossectomy for OSA was first reported in 1991 as a salvage surgery after uvulopalatopharyngoplasty, said Dr. Murphey, who is completing a clinical research fellowship in the department of otolaryngology-head and neck surgery at Medical University of South Carolina, Charleston, under the mentoring of Dr. Marion B. Gillespie and Dr. Shaun A. Nguyen. Since that time, “many modifications have been made regarding technique and instrumentation with recent focus on minimally invasive techniques aimed at maximizing tissue reduction while limiting the inherent morbidity associated with glossectomy,” he said. “The aim of our study was to review and analyze all of the available literature on partial glossectomy for OSA in one study. Overall, there is a significant lack of research into glossectomy, and what literature is available include small, case-series that analyze glossectomy as part of complex, multi-level surgeries. This study represents the first large scale meta-analysis on the role of glossectomy, and attempts to determine the role of glossectomy both as part of multi-level surgery, and as a single, stand-alone sleep apnea treatment.”
Dr. Murphey used the PubMed-NCBI literature database to identify studies with 10 or more patients and reported preoperative and postoperative apnea-hypopnea index (AHI) scores. The primary endpoint was change in AHI while secondary endpoints included predefined surgical success rates, and changes in additional reported sleep outcomes such as Epworth Sleep Scores (ESS), Lowest Oxygen Saturation (LSAT), and snoring visual analog scale (VAS). The researchers reported results from 15 articles with 442 patients treated with three glossectomy techniques (midline glossectomy, lingualplasty, and submucosal minimally invasive lingual excision (SMILE). In pooled analyses that compared baseline vs. post-surgery, investigators observed significant reductions in AHI (from 48 to 20); ESS (from 12 to 5), and VAS (from 9 to 3; all with a P of less than .0001). In addition, they observed a significant increase in LSAT (from 77% to 84%; P less than .0001), according to the findings, presented at the meeting, which was jointly sponsored by the Triological Society and the American College of Surgeons.
Surgical success, which was defined as an AHI less than 20 and a greater than 50% reduction in AHI, was achieved in 56% of cases, while complications occurred in 18% of patients. Only 24 patients (5%) were treated with glossectomy as sole therapy for OSA. Among these 24 patients, significant reductions occurred in AHI (from 42 to 25; P=.0345) and ESS (from 12 to 7; P less than .0001).
Dr. Murphey acknowledged certain limitations of the analysis, including “a lack of quality research involving glossectomy, with the majority of available published data drawn from small, case-series without control arms,” he said. “Additionally, studies varied in surgical approach, inclusion criteria, and accompanying procedures in multi-level treatment. This makes it extremely difficult to truly compare treatments.”
The researchers reported having no relevant financial conflicts.
On Twitter @dougbrunk
AT THE COMBINED SECTIONS WINTER MEETING
Key clinical point: Among adults with obstructive sleep apnea, glossectomy significantly improved sleep outcomes when performed as part of a multi-level surgery, though evidence is sparse.
Major finding: There are no available data on the role of glossectomy as a stand-alone procedure for the treatment of OSA.
Data source: A meta-analysis of 15 articles concerning 442 patients treated with three glossectomy techniques (midline glossectomy, lingualplasty, and submucosal minimally invasive lingual excision.
Disclosures: The researchers reported having no financial disclosures.
Asphyxiation by Cake: An Unusual Case of Dyspnea
A 58-year-old man presented to the emergency department (ED) via emergency medical services (EMS) with shortness of breath, lightheadedness, and nausea. Upon arrival to the ED, most of his symptoms had resolved. The patient reported that he had taken a two-hour flight into town the previous day and had spent an uneventful evening at a local hotel. He said that he began experiencing shortness of breath and lightheadedness soon after entering his rental vehicle an hour prior to presentation, explaining that he felt as if he “could not get any air.”
He denied chest pain, leg pain or swelling, abdominal pain, or recent illness. Medical history was significant for hypertension, for which he was taking losartan and amlodipine. He had no drug allergies, surgical history, or smoking history. Of note, when the hotel clerk got in the same rental vehicle to move it, he developed symptoms similar to those of the patient. As with the patient, the clerk’s symptoms quickly resolved after he got out of the vehicle.
The patient’s vital signs at examination included an oral temperature of 97.5°F; pulse, 62 beats/min; respiratory rate (RR), 18 breaths/min; blood pressure, 133/83 mm Hg; and O2 saturation, 100% on room air. He was alert and oriented, in no distress, easily conversational, and without diaphoresis. The lungs were clear to auscultation bilaterally, and there was no calf swelling, tenderness, or palpable cords. The remainder of the physical exam was normal.
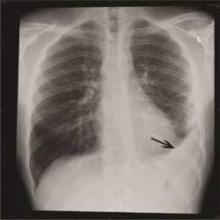
Ancillary studies included a normal chest X-ray. An ECG demonstrated sinus bradycardia with a rate of 56 beats/min but no evidence of ischemia or right heart strain. Complete blood count, troponin I, D-dimer, and creatine phosphokinase (CPK) with MB fraction levels were all within normal limits. A serum chemistry panel was also within normal limits, except for a serum glucose level of 181 mg/dL. Venous co-oximetry showed a carboxyhemoglobin level of 0.0, and methemoglobin level of 0.5 gm% (normal range, 0.4-1.5).
Since both the patient’s and hotel clerk’s symptoms started when each was in the rental car, the patient was questioned about the vehicle and its contents. The car was a late-model rental in good condition per report. The patient informed the treating emergency physician that he worked as a decorative cake salesman and had brought cake samples with him to display at a trade show. He further stated that he had left these samples in the car overnight, packed in dry ice.
What's the solution to this unusual case?
Upon learning this information, EMS was contacted and instructed to return to the hotel and rental vehicle. The hotel room was noted to have normal levels of O2 and carbon monoxide (CO) on measurement. Investigation of the car revealed normal levels of CO but O2 levels too low to read on the sensor. The emergency team concluded that the dry ice (the solid form of carbon dioxide [CO2]) sublimed to CO2 gas overnight. This displaced the O2 in the vehicle, resulting in severe hypoxia and the symptoms of both the patient and hotel clerk.
The patient was initially placed on 15 L of O2 via a nonrebreather mask, then switched to 2 L of O2 via nasal cannula. He was observed for a total of four hours after arrival; as he remained symptom-free, he was discharged home. Postdischarge follow-up information was not obtainable.
DISCUSSION
Carbon dioxide is prevalent in everyday life, from an agent in fire extinguishers and carbonation in beverages to byproducts of cellular metabolism. Similar to CO, it is a colorless and odorless gas.
Carbon dioxide is commonly used in the food industry as dry ice to keep items cold. In its solid state, CO2 can cause severe frostbite with direct contact, similar to a burn. However, when dry ice is warmed and sublimated to a gaseous state, large amounts of CO2 are generated, and this heavy gas can accumulate and displace air (ie, atmospheric O2), especially in confined spaces. In low concentrations, gaseous CO2 appears to have minimal toxicologic effects, but at higher concentrations it can cause tachycardia, tachypnea, dyspnea, visual disturbances, arrhythmias, impaired levels of consciousness, and death.
Carbon dioxide primarily acts as a simple asphyxiant, but it also dissolves in serum as carbonic acid, resulting in a metabolic acidosis. Compensation for this acidosis is accomplished by an increased RR (ie, respiratory alkalosis), which further worsens the intake of CO2.1,2
The normal concentration of CO2 in the atmosphere is approximately 0.04% (396 ppm). The Occupational Safety and Health Administration (OSHA) has set a maximum safe exposure level of CO2 at 0.5% (5,000 ppm) over an eight-hour day.3 Concentrations as low as 1% (10,000 ppm) may cause drowsiness. Exposure to concentrations of 7% to 10% for several minutes to an hour results in headache, tachycardia, dyspnea, and hyperventilation. At levels of 10% to 15%, dizziness, severe muscle twitching, and loss of consciousness can occur after only a few minutes. Death occurs within minutes at concentrations greater than 30%.2
Carbon dioxide also acts as a potent cerebral vasodilator, which may explain symptoms such as headache and dizziness.2 The severity of symptoms is dependent on the concentration of CO2, the length of the exposure, and the underlying health of the patient. Elevated concentrations of CO2 can occur in areas where there is limited or poor ventilation, such as in a mine (where it is known as blackdamp, stythe, or choke damp),4 submarine, grain silo, or a sealed building without mechanical ventilation.
Continue for other case presentations >>
Other case presentations
Similar cases have been described in the literature. In one case, following Hurricane Ivan, a 34-year-old man placed four 25-pound blocks of dry ice wrapped in paper in the front seat of his truck with the windows closed.5 After driving less than one-quarter of a mile, he developed dyspnea and telephoned for help before losing consciousness. Fortunately, he was found in time and recovered soon after the doors to his truck were opened.5
In another case, a 59-year-old man entered a walk-in freezer that contained dry ice wrapped loosely in plastic. He was found inside the freezer 20 minutes later in cardiac arrest; resuscitation efforts were unsuccessful. Investigation of the freezer found an initial O2 concentration of 13% (normal level, 20.93%) and an estimated CO2 level of 40%.5
Similarly, a 35-year-old woman was inadvertently locked in a bank vault while storing receipts. In a bid for help, she pulled the fire alarm, which triggered a CO2-based fire-extinguishing system. The fire department responded and found the woman dead in the vault 30 minutes later. The cause of death was labeled as CO2 intoxication.6
Natural phenomena
There have also been documented cases of CO2 toxicity associated with volcanic eruption and other natural phenomena; for example, the Lake Nyos, Cameroon, West Africa incident in 1986. In this event, a magma pocket underlying the lake saturated the water with CO2 stored as carbonic acid in the water. When a landslide hit the lake, it caused the carbonic acid stored in the depths of the lake to be upheaved to the surface, where it turned back into CO2 and was released into the atmosphere. Since CO2 is heavier than O2, it displaced the O2 near the ground, resulting in the suffocation and death of 1,700 people in the surrounding villages.2
Next page: Differential diagnosis >>
Differential diagnosis
When CO2 toxicity is suspected, other conditions should be considered, as there may be more than one process involved. For example, other causes of coma or dyspnea should be investigated, including trauma, hypoglycemia, CO, methemoglobinemia, or other metabolic processes. In addition, a patient may have a pre-existing condition, such as a trauma or an altered mental status due to drugs or alcohol, all of which can increase his or her susceptibility to the effects of CO2.
Evaluation and treatment
Useful laboratory testing includes arterial blood gas, venous co-oximetry for carboxyhemoglobin, chemistry panels, ethanol testing, and radiographs or CT, as indicated.
Initial management of suspected CO2 toxicity entails first removing the patient from the source of the gas. Rescuers must exercise caution so as to prevent a mass-casualty incident. Once out of the dangerous environment, as long as the patient is conscious and spontaneously breathing, supportive measures are generally all that are necessary. Oxygen should be applied, after which the spontaneously breathing patient without underlying lung disease should rapidly return to normal.
If there is marked decrease in mental status or poor respiratory drive despite O2 administration, intubation with mechanical ventilation may be required. A higher-than-normal RR will help remove excessive CO2 in this instance.
If a respiratory acidosis is present, IV sodium bicarbonate should be avoided, as this may increase the level of serum CO2. IV fluids and other supportive measures, including treatment for any concurrent conditions, may be indicated.
REFERENCES
1. Nelson LS, Odujebe OA. Simple asphyxiants and pulmonary irritants. In: Nelson LS, Lewin NA, Howland MA, et al, eds. Goldfrank’s Toxicologic Emergencies, 9th ed. New York, NY: McGraw-Hill; 2011:1644-1645.
2. Langford NJ. Carbon dioxide poisoning. Toxicol Rev. 2005;24(4):229-235.
3. Occupational Health and Safety Standards. Table Z-1, Limits for air contaminants. Occupational Safety and Health Administration Web site. www.osha.gov/pls/oshaweb/owadisp.show_document?p_table=standards&p_id=9992. Accessed January 19, 2015.
4. Hedlund FH. The extreme carbon dioxide outburst at the Menzengraben potash mine 7 July 1953. Safety Sci. 2012;50(3):537-553.
5. Dunford JV, Lucas J, Vent N, et al. Asphyxiation due to dry ice in a walk-in freezer. J Emerg Med. 2009;36(4):353-356.
6. Gill JR, Ely SF, Hua Z. Environmental gas displacement: three accidental deaths in the workplace. Am J Forensic Med Pathol. 2002;23(1):26-30.
A 58-year-old man presented to the emergency department (ED) via emergency medical services (EMS) with shortness of breath, lightheadedness, and nausea. Upon arrival to the ED, most of his symptoms had resolved. The patient reported that he had taken a two-hour flight into town the previous day and had spent an uneventful evening at a local hotel. He said that he began experiencing shortness of breath and lightheadedness soon after entering his rental vehicle an hour prior to presentation, explaining that he felt as if he “could not get any air.”
He denied chest pain, leg pain or swelling, abdominal pain, or recent illness. Medical history was significant for hypertension, for which he was taking losartan and amlodipine. He had no drug allergies, surgical history, or smoking history. Of note, when the hotel clerk got in the same rental vehicle to move it, he developed symptoms similar to those of the patient. As with the patient, the clerk’s symptoms quickly resolved after he got out of the vehicle.
The patient’s vital signs at examination included an oral temperature of 97.5°F; pulse, 62 beats/min; respiratory rate (RR), 18 breaths/min; blood pressure, 133/83 mm Hg; and O2 saturation, 100% on room air. He was alert and oriented, in no distress, easily conversational, and without diaphoresis. The lungs were clear to auscultation bilaterally, and there was no calf swelling, tenderness, or palpable cords. The remainder of the physical exam was normal.
Ancillary studies included a normal chest X-ray. An ECG demonstrated sinus bradycardia with a rate of 56 beats/min but no evidence of ischemia or right heart strain. Complete blood count, troponin I, D-dimer, and creatine phosphokinase (CPK) with MB fraction levels were all within normal limits. A serum chemistry panel was also within normal limits, except for a serum glucose level of 181 mg/dL. Venous co-oximetry showed a carboxyhemoglobin level of 0.0, and methemoglobin level of 0.5 gm% (normal range, 0.4-1.5).
Since both the patient’s and hotel clerk’s symptoms started when each was in the rental car, the patient was questioned about the vehicle and its contents. The car was a late-model rental in good condition per report. The patient informed the treating emergency physician that he worked as a decorative cake salesman and had brought cake samples with him to display at a trade show. He further stated that he had left these samples in the car overnight, packed in dry ice.
What's the solution to this unusual case?
Upon learning this information, EMS was contacted and instructed to return to the hotel and rental vehicle. The hotel room was noted to have normal levels of O2 and carbon monoxide (CO) on measurement. Investigation of the car revealed normal levels of CO but O2 levels too low to read on the sensor. The emergency team concluded that the dry ice (the solid form of carbon dioxide [CO2]) sublimed to CO2 gas overnight. This displaced the O2 in the vehicle, resulting in severe hypoxia and the symptoms of both the patient and hotel clerk.
The patient was initially placed on 15 L of O2 via a nonrebreather mask, then switched to 2 L of O2 via nasal cannula. He was observed for a total of four hours after arrival; as he remained symptom-free, he was discharged home. Postdischarge follow-up information was not obtainable.
DISCUSSION
Carbon dioxide is prevalent in everyday life, from an agent in fire extinguishers and carbonation in beverages to byproducts of cellular metabolism. Similar to CO, it is a colorless and odorless gas.
Carbon dioxide is commonly used in the food industry as dry ice to keep items cold. In its solid state, CO2 can cause severe frostbite with direct contact, similar to a burn. However, when dry ice is warmed and sublimated to a gaseous state, large amounts of CO2 are generated, and this heavy gas can accumulate and displace air (ie, atmospheric O2), especially in confined spaces. In low concentrations, gaseous CO2 appears to have minimal toxicologic effects, but at higher concentrations it can cause tachycardia, tachypnea, dyspnea, visual disturbances, arrhythmias, impaired levels of consciousness, and death.
Carbon dioxide primarily acts as a simple asphyxiant, but it also dissolves in serum as carbonic acid, resulting in a metabolic acidosis. Compensation for this acidosis is accomplished by an increased RR (ie, respiratory alkalosis), which further worsens the intake of CO2.1,2
The normal concentration of CO2 in the atmosphere is approximately 0.04% (396 ppm). The Occupational Safety and Health Administration (OSHA) has set a maximum safe exposure level of CO2 at 0.5% (5,000 ppm) over an eight-hour day.3 Concentrations as low as 1% (10,000 ppm) may cause drowsiness. Exposure to concentrations of 7% to 10% for several minutes to an hour results in headache, tachycardia, dyspnea, and hyperventilation. At levels of 10% to 15%, dizziness, severe muscle twitching, and loss of consciousness can occur after only a few minutes. Death occurs within minutes at concentrations greater than 30%.2
Carbon dioxide also acts as a potent cerebral vasodilator, which may explain symptoms such as headache and dizziness.2 The severity of symptoms is dependent on the concentration of CO2, the length of the exposure, and the underlying health of the patient. Elevated concentrations of CO2 can occur in areas where there is limited or poor ventilation, such as in a mine (where it is known as blackdamp, stythe, or choke damp),4 submarine, grain silo, or a sealed building without mechanical ventilation.
Continue for other case presentations >>
Other case presentations
Similar cases have been described in the literature. In one case, following Hurricane Ivan, a 34-year-old man placed four 25-pound blocks of dry ice wrapped in paper in the front seat of his truck with the windows closed.5 After driving less than one-quarter of a mile, he developed dyspnea and telephoned for help before losing consciousness. Fortunately, he was found in time and recovered soon after the doors to his truck were opened.5
In another case, a 59-year-old man entered a walk-in freezer that contained dry ice wrapped loosely in plastic. He was found inside the freezer 20 minutes later in cardiac arrest; resuscitation efforts were unsuccessful. Investigation of the freezer found an initial O2 concentration of 13% (normal level, 20.93%) and an estimated CO2 level of 40%.5
Similarly, a 35-year-old woman was inadvertently locked in a bank vault while storing receipts. In a bid for help, she pulled the fire alarm, which triggered a CO2-based fire-extinguishing system. The fire department responded and found the woman dead in the vault 30 minutes later. The cause of death was labeled as CO2 intoxication.6
Natural phenomena
There have also been documented cases of CO2 toxicity associated with volcanic eruption and other natural phenomena; for example, the Lake Nyos, Cameroon, West Africa incident in 1986. In this event, a magma pocket underlying the lake saturated the water with CO2 stored as carbonic acid in the water. When a landslide hit the lake, it caused the carbonic acid stored in the depths of the lake to be upheaved to the surface, where it turned back into CO2 and was released into the atmosphere. Since CO2 is heavier than O2, it displaced the O2 near the ground, resulting in the suffocation and death of 1,700 people in the surrounding villages.2
Next page: Differential diagnosis >>
Differential diagnosis
When CO2 toxicity is suspected, other conditions should be considered, as there may be more than one process involved. For example, other causes of coma or dyspnea should be investigated, including trauma, hypoglycemia, CO, methemoglobinemia, or other metabolic processes. In addition, a patient may have a pre-existing condition, such as a trauma or an altered mental status due to drugs or alcohol, all of which can increase his or her susceptibility to the effects of CO2.
Evaluation and treatment
Useful laboratory testing includes arterial blood gas, venous co-oximetry for carboxyhemoglobin, chemistry panels, ethanol testing, and radiographs or CT, as indicated.
Initial management of suspected CO2 toxicity entails first removing the patient from the source of the gas. Rescuers must exercise caution so as to prevent a mass-casualty incident. Once out of the dangerous environment, as long as the patient is conscious and spontaneously breathing, supportive measures are generally all that are necessary. Oxygen should be applied, after which the spontaneously breathing patient without underlying lung disease should rapidly return to normal.
If there is marked decrease in mental status or poor respiratory drive despite O2 administration, intubation with mechanical ventilation may be required. A higher-than-normal RR will help remove excessive CO2 in this instance.
If a respiratory acidosis is present, IV sodium bicarbonate should be avoided, as this may increase the level of serum CO2. IV fluids and other supportive measures, including treatment for any concurrent conditions, may be indicated.
REFERENCES
1. Nelson LS, Odujebe OA. Simple asphyxiants and pulmonary irritants. In: Nelson LS, Lewin NA, Howland MA, et al, eds. Goldfrank’s Toxicologic Emergencies, 9th ed. New York, NY: McGraw-Hill; 2011:1644-1645.
2. Langford NJ. Carbon dioxide poisoning. Toxicol Rev. 2005;24(4):229-235.
3. Occupational Health and Safety Standards. Table Z-1, Limits for air contaminants. Occupational Safety and Health Administration Web site. www.osha.gov/pls/oshaweb/owadisp.show_document?p_table=standards&p_id=9992. Accessed January 19, 2015.
4. Hedlund FH. The extreme carbon dioxide outburst at the Menzengraben potash mine 7 July 1953. Safety Sci. 2012;50(3):537-553.
5. Dunford JV, Lucas J, Vent N, et al. Asphyxiation due to dry ice in a walk-in freezer. J Emerg Med. 2009;36(4):353-356.
6. Gill JR, Ely SF, Hua Z. Environmental gas displacement: three accidental deaths in the workplace. Am J Forensic Med Pathol. 2002;23(1):26-30.
A 58-year-old man presented to the emergency department (ED) via emergency medical services (EMS) with shortness of breath, lightheadedness, and nausea. Upon arrival to the ED, most of his symptoms had resolved. The patient reported that he had taken a two-hour flight into town the previous day and had spent an uneventful evening at a local hotel. He said that he began experiencing shortness of breath and lightheadedness soon after entering his rental vehicle an hour prior to presentation, explaining that he felt as if he “could not get any air.”
He denied chest pain, leg pain or swelling, abdominal pain, or recent illness. Medical history was significant for hypertension, for which he was taking losartan and amlodipine. He had no drug allergies, surgical history, or smoking history. Of note, when the hotel clerk got in the same rental vehicle to move it, he developed symptoms similar to those of the patient. As with the patient, the clerk’s symptoms quickly resolved after he got out of the vehicle.
The patient’s vital signs at examination included an oral temperature of 97.5°F; pulse, 62 beats/min; respiratory rate (RR), 18 breaths/min; blood pressure, 133/83 mm Hg; and O2 saturation, 100% on room air. He was alert and oriented, in no distress, easily conversational, and without diaphoresis. The lungs were clear to auscultation bilaterally, and there was no calf swelling, tenderness, or palpable cords. The remainder of the physical exam was normal.
Ancillary studies included a normal chest X-ray. An ECG demonstrated sinus bradycardia with a rate of 56 beats/min but no evidence of ischemia or right heart strain. Complete blood count, troponin I, D-dimer, and creatine phosphokinase (CPK) with MB fraction levels were all within normal limits. A serum chemistry panel was also within normal limits, except for a serum glucose level of 181 mg/dL. Venous co-oximetry showed a carboxyhemoglobin level of 0.0, and methemoglobin level of 0.5 gm% (normal range, 0.4-1.5).
Since both the patient’s and hotel clerk’s symptoms started when each was in the rental car, the patient was questioned about the vehicle and its contents. The car was a late-model rental in good condition per report. The patient informed the treating emergency physician that he worked as a decorative cake salesman and had brought cake samples with him to display at a trade show. He further stated that he had left these samples in the car overnight, packed in dry ice.
What's the solution to this unusual case?
Upon learning this information, EMS was contacted and instructed to return to the hotel and rental vehicle. The hotel room was noted to have normal levels of O2 and carbon monoxide (CO) on measurement. Investigation of the car revealed normal levels of CO but O2 levels too low to read on the sensor. The emergency team concluded that the dry ice (the solid form of carbon dioxide [CO2]) sublimed to CO2 gas overnight. This displaced the O2 in the vehicle, resulting in severe hypoxia and the symptoms of both the patient and hotel clerk.
The patient was initially placed on 15 L of O2 via a nonrebreather mask, then switched to 2 L of O2 via nasal cannula. He was observed for a total of four hours after arrival; as he remained symptom-free, he was discharged home. Postdischarge follow-up information was not obtainable.
DISCUSSION
Carbon dioxide is prevalent in everyday life, from an agent in fire extinguishers and carbonation in beverages to byproducts of cellular metabolism. Similar to CO, it is a colorless and odorless gas.
Carbon dioxide is commonly used in the food industry as dry ice to keep items cold. In its solid state, CO2 can cause severe frostbite with direct contact, similar to a burn. However, when dry ice is warmed and sublimated to a gaseous state, large amounts of CO2 are generated, and this heavy gas can accumulate and displace air (ie, atmospheric O2), especially in confined spaces. In low concentrations, gaseous CO2 appears to have minimal toxicologic effects, but at higher concentrations it can cause tachycardia, tachypnea, dyspnea, visual disturbances, arrhythmias, impaired levels of consciousness, and death.
Carbon dioxide primarily acts as a simple asphyxiant, but it also dissolves in serum as carbonic acid, resulting in a metabolic acidosis. Compensation for this acidosis is accomplished by an increased RR (ie, respiratory alkalosis), which further worsens the intake of CO2.1,2
The normal concentration of CO2 in the atmosphere is approximately 0.04% (396 ppm). The Occupational Safety and Health Administration (OSHA) has set a maximum safe exposure level of CO2 at 0.5% (5,000 ppm) over an eight-hour day.3 Concentrations as low as 1% (10,000 ppm) may cause drowsiness. Exposure to concentrations of 7% to 10% for several minutes to an hour results in headache, tachycardia, dyspnea, and hyperventilation. At levels of 10% to 15%, dizziness, severe muscle twitching, and loss of consciousness can occur after only a few minutes. Death occurs within minutes at concentrations greater than 30%.2
Carbon dioxide also acts as a potent cerebral vasodilator, which may explain symptoms such as headache and dizziness.2 The severity of symptoms is dependent on the concentration of CO2, the length of the exposure, and the underlying health of the patient. Elevated concentrations of CO2 can occur in areas where there is limited or poor ventilation, such as in a mine (where it is known as blackdamp, stythe, or choke damp),4 submarine, grain silo, or a sealed building without mechanical ventilation.
Continue for other case presentations >>
Other case presentations
Similar cases have been described in the literature. In one case, following Hurricane Ivan, a 34-year-old man placed four 25-pound blocks of dry ice wrapped in paper in the front seat of his truck with the windows closed.5 After driving less than one-quarter of a mile, he developed dyspnea and telephoned for help before losing consciousness. Fortunately, he was found in time and recovered soon after the doors to his truck were opened.5
In another case, a 59-year-old man entered a walk-in freezer that contained dry ice wrapped loosely in plastic. He was found inside the freezer 20 minutes later in cardiac arrest; resuscitation efforts were unsuccessful. Investigation of the freezer found an initial O2 concentration of 13% (normal level, 20.93%) and an estimated CO2 level of 40%.5
Similarly, a 35-year-old woman was inadvertently locked in a bank vault while storing receipts. In a bid for help, she pulled the fire alarm, which triggered a CO2-based fire-extinguishing system. The fire department responded and found the woman dead in the vault 30 minutes later. The cause of death was labeled as CO2 intoxication.6
Natural phenomena
There have also been documented cases of CO2 toxicity associated with volcanic eruption and other natural phenomena; for example, the Lake Nyos, Cameroon, West Africa incident in 1986. In this event, a magma pocket underlying the lake saturated the water with CO2 stored as carbonic acid in the water. When a landslide hit the lake, it caused the carbonic acid stored in the depths of the lake to be upheaved to the surface, where it turned back into CO2 and was released into the atmosphere. Since CO2 is heavier than O2, it displaced the O2 near the ground, resulting in the suffocation and death of 1,700 people in the surrounding villages.2
Next page: Differential diagnosis >>
Differential diagnosis
When CO2 toxicity is suspected, other conditions should be considered, as there may be more than one process involved. For example, other causes of coma or dyspnea should be investigated, including trauma, hypoglycemia, CO, methemoglobinemia, or other metabolic processes. In addition, a patient may have a pre-existing condition, such as a trauma or an altered mental status due to drugs or alcohol, all of which can increase his or her susceptibility to the effects of CO2.
Evaluation and treatment
Useful laboratory testing includes arterial blood gas, venous co-oximetry for carboxyhemoglobin, chemistry panels, ethanol testing, and radiographs or CT, as indicated.
Initial management of suspected CO2 toxicity entails first removing the patient from the source of the gas. Rescuers must exercise caution so as to prevent a mass-casualty incident. Once out of the dangerous environment, as long as the patient is conscious and spontaneously breathing, supportive measures are generally all that are necessary. Oxygen should be applied, after which the spontaneously breathing patient without underlying lung disease should rapidly return to normal.
If there is marked decrease in mental status or poor respiratory drive despite O2 administration, intubation with mechanical ventilation may be required. A higher-than-normal RR will help remove excessive CO2 in this instance.
If a respiratory acidosis is present, IV sodium bicarbonate should be avoided, as this may increase the level of serum CO2. IV fluids and other supportive measures, including treatment for any concurrent conditions, may be indicated.
REFERENCES
1. Nelson LS, Odujebe OA. Simple asphyxiants and pulmonary irritants. In: Nelson LS, Lewin NA, Howland MA, et al, eds. Goldfrank’s Toxicologic Emergencies, 9th ed. New York, NY: McGraw-Hill; 2011:1644-1645.
2. Langford NJ. Carbon dioxide poisoning. Toxicol Rev. 2005;24(4):229-235.
3. Occupational Health and Safety Standards. Table Z-1, Limits for air contaminants. Occupational Safety and Health Administration Web site. www.osha.gov/pls/oshaweb/owadisp.show_document?p_table=standards&p_id=9992. Accessed January 19, 2015.
4. Hedlund FH. The extreme carbon dioxide outburst at the Menzengraben potash mine 7 July 1953. Safety Sci. 2012;50(3):537-553.
5. Dunford JV, Lucas J, Vent N, et al. Asphyxiation due to dry ice in a walk-in freezer. J Emerg Med. 2009;36(4):353-356.
6. Gill JR, Ely SF, Hua Z. Environmental gas displacement: three accidental deaths in the workplace. Am J Forensic Med Pathol. 2002;23(1):26-30.
FDA panel backs antifungal for invasive aspergillosis, mucormycosis
SILVER SPRING, MD. – A novel treatment for invasive aspergillosis and invasive mucormycosis gained the support of a Food and Drug Administration advisory panel, although members were more ambivalent about the mucormycosis indication, based on the small amount of data available in those patients.
At a meeting on Jan. 22, the FDA’s Anti-Infective Drugs Advisory Committee voted 11-0 that there was “substantial evidence” that isavuconazonium, an antifungal prodrug, was safe and effective for the treatment of invasive aspergillosis, a life-threatening condition most commonly seen in immunocompromised patients. The drug was studied in a phase III study comparing isavuconazonium to voriconazole, the standard of care, in more than 500 patients.
The panel voted 8-2, with one abstention, that there was substantial evidence it was safe and effective for treating patients with invasive mucormycosis, with panelists voting on both sides of the question citing the study of only 37 patients that used historical controls as an issue. Those supporting approval for mucormycosis said that, if approved, the manufacturer, Astellas, should be required to conduct a phase IV trial further evaluating this treatment for patients with this infection.
Isavuconazonium is a prodrug of isavuconazole, a triazole antifungal, and would be available in an oral capsule formulation and a powder formulation that is reconstituted for intravenous administration (and needs to be administered through an in-line filter in case of particulate formation).
For aspergillosis, “I do believe that this drug provides a reasonable alternative to the current therapies that are available without additional toxicities,” said panelist Dr. Paige Waterman of the Global Emerging Infections Surveillance and Response System at the Walter Reed Army Medical Center, Silver Spring, Md. Other panelists agreed with her recommendations that labeling should make clear that it should not be used in people under age 18 years or in pregnant women, that a filter should be used when administered intravenously, and that labeling should include a statement about the increased risk of hepatotoxicity that also appears in the labeling of other drugs in the same class.
Because it has been associated with a shortened QT interval, she said that screening ECGs should be recommended for patients and that there should be some extra caution when it is prescribed to people who are Asian or of Asian descent, since drug concentrations were slightly higher among Asian patients who received the drug. Other recommendations from panelists included the need to conduct studies of the drug to provide information on therapeutic drug monitoring and in people under 18 years.
For treatment of invasive mucormycosis, an even rarer fungal infection which, in hospitals, is associated with the use of contaminated materials or organ transplantation, the panelists were more hesitant, but those voting in favor of approval cited the significance of the condition and the reasonable efficacy results in the study, stressing that postmarketing studies were critical. Panelists also noted that more clinical data are clearly needed in patients with this infection and the lack of data directly comparing it to amphotericin B – the only FDA-approved drug for this indication – was problematic.
“This drug really does fill an unmet need; I have high hopes that it is at least as good as amphotericin. But I do think we need more data to confirm that,” said Dr. Michael Neely, chair of antimicrobial stewardship at Children’s Hospital, Los Angeles, who was among those voting in favor of this indication. It appears to have a better safety profile and it would be available in both oral and intravenous formulations, he added. Amphotericin B is available only in an intravenous (IV) formulation.
If approved, isavuconazonium would provide an alternative to voriconazole for treating aspergillosis, and the IV formulation does not contain cyclodextrin, unlike the IV formulation of voriconazole, which limits its use in patients with moderate to severe renal dysfunction, according to Astellas. Safety concerns specific to isavuconazonium include QT-segment shortening and particulate formation in the IV formulation, according to the FDA.
The randomized, double-blind, international, noninferiority study compared treatment with isavuconazonium to voriconazole in 516 adults with invasive aspergillosis. In the randomized study, the mean age of patients was 51 years, 60% were men, most were white, 11% were in the United States and Canada, 20% had had an allogeneic bone marrow transplant, and 70% had an uncontrolled malignancy at baseline (Infect. Drug Resist. 2013;6:16374). The primary effectiveness endpoint, all-cause mortality through day 42, was 19% in those on isavuconazonium, compared with 20% in those on voriconazole. The study met the prespecified noninferiority margin. The rates of deaths and serious adverse events were similar to voriconazole, and there were fewer adverse events in those on isavuconazonium requiring discontinuation of the drug (14% vs. 23%).
Differences in adverse events included lower rates of hepatobiliary disorders, including hyperbilirubinemia, abnormal hepatic function, and jaundice (9% vs. 16%); skin and subcutaneous tissue disorders, including rash, erythema, and drug eruption (34% vs 43%) associated with isavuconazonium, compared with voriconazole. Common adverse events included nausea (28%), vomiting (25%), and diarrhea (24%). Decreases in the QT segment occurred in 7.5% of those on isavuconazonium, compared with 4.5% of those on voriconazole, but were not associated with clinical events.
The prospective, open-label, single-arm study evaluated isavuconazonium in 37 patients with proven or probable mucormycosis infections, whose mean age was 49 years; 59% had a hematologic malignancy, and about 40% were neutropenic at baseline. All-cause mortality at day 42 was almost 38%, which was similar to the mortality rate for amphotericin in the literature, according to Astellas.
There are about 12,000 cases of aspergillosis and about 500 cases of mucormycosis in the United States every year, the company said. In addition to voriconazole, other drugs approved for invasive aspergillosis include amphotericin formulations, itraconazole, and caspofungin. The FDA usually follows the recommendations of its advisory panels. The FDA is expected to make a decision by March 8, according to Astellas, which plans to market the drug as Cresemba if approved. It is also under review in Europe for the same indications.
Panelists were cleared of potential conflicts of interest related to the topic of the meeting. In some cases, a panelist may be given a waiver but not at this meeting.
Dr. Daniel Ouellette, FCCP, comments: Serious fungal infections are being increasingly recognized in patients who are immunocompromised or critically ill, but effective treatments with an acceptable side-effect profile have been sparse. Just a few years ago, the mainstay of treatment was amphotericin, an agent whose use has been associated with a variety of complications. However, novel agents have been developed that hold promise. The recent development of isavuconazonium, a pro-drug of isavuconazole, may lead to further augmentation of the clinician's armamentarium in fighting these illnesses.
Dr. Daniel Ouellette, FCCP, comments: Serious fungal infections are being increasingly recognized in patients who are immunocompromised or critically ill, but effective treatments with an acceptable side-effect profile have been sparse. Just a few years ago, the mainstay of treatment was amphotericin, an agent whose use has been associated with a variety of complications. However, novel agents have been developed that hold promise. The recent development of isavuconazonium, a pro-drug of isavuconazole, may lead to further augmentation of the clinician's armamentarium in fighting these illnesses.
Dr. Daniel Ouellette, FCCP, comments: Serious fungal infections are being increasingly recognized in patients who are immunocompromised or critically ill, but effective treatments with an acceptable side-effect profile have been sparse. Just a few years ago, the mainstay of treatment was amphotericin, an agent whose use has been associated with a variety of complications. However, novel agents have been developed that hold promise. The recent development of isavuconazonium, a pro-drug of isavuconazole, may lead to further augmentation of the clinician's armamentarium in fighting these illnesses.
SILVER SPRING, MD. – A novel treatment for invasive aspergillosis and invasive mucormycosis gained the support of a Food and Drug Administration advisory panel, although members were more ambivalent about the mucormycosis indication, based on the small amount of data available in those patients.
At a meeting on Jan. 22, the FDA’s Anti-Infective Drugs Advisory Committee voted 11-0 that there was “substantial evidence” that isavuconazonium, an antifungal prodrug, was safe and effective for the treatment of invasive aspergillosis, a life-threatening condition most commonly seen in immunocompromised patients. The drug was studied in a phase III study comparing isavuconazonium to voriconazole, the standard of care, in more than 500 patients.
The panel voted 8-2, with one abstention, that there was substantial evidence it was safe and effective for treating patients with invasive mucormycosis, with panelists voting on both sides of the question citing the study of only 37 patients that used historical controls as an issue. Those supporting approval for mucormycosis said that, if approved, the manufacturer, Astellas, should be required to conduct a phase IV trial further evaluating this treatment for patients with this infection.
Isavuconazonium is a prodrug of isavuconazole, a triazole antifungal, and would be available in an oral capsule formulation and a powder formulation that is reconstituted for intravenous administration (and needs to be administered through an in-line filter in case of particulate formation).
For aspergillosis, “I do believe that this drug provides a reasonable alternative to the current therapies that are available without additional toxicities,” said panelist Dr. Paige Waterman of the Global Emerging Infections Surveillance and Response System at the Walter Reed Army Medical Center, Silver Spring, Md. Other panelists agreed with her recommendations that labeling should make clear that it should not be used in people under age 18 years or in pregnant women, that a filter should be used when administered intravenously, and that labeling should include a statement about the increased risk of hepatotoxicity that also appears in the labeling of other drugs in the same class.
Because it has been associated with a shortened QT interval, she said that screening ECGs should be recommended for patients and that there should be some extra caution when it is prescribed to people who are Asian or of Asian descent, since drug concentrations were slightly higher among Asian patients who received the drug. Other recommendations from panelists included the need to conduct studies of the drug to provide information on therapeutic drug monitoring and in people under 18 years.
For treatment of invasive mucormycosis, an even rarer fungal infection which, in hospitals, is associated with the use of contaminated materials or organ transplantation, the panelists were more hesitant, but those voting in favor of approval cited the significance of the condition and the reasonable efficacy results in the study, stressing that postmarketing studies were critical. Panelists also noted that more clinical data are clearly needed in patients with this infection and the lack of data directly comparing it to amphotericin B – the only FDA-approved drug for this indication – was problematic.
“This drug really does fill an unmet need; I have high hopes that it is at least as good as amphotericin. But I do think we need more data to confirm that,” said Dr. Michael Neely, chair of antimicrobial stewardship at Children’s Hospital, Los Angeles, who was among those voting in favor of this indication. It appears to have a better safety profile and it would be available in both oral and intravenous formulations, he added. Amphotericin B is available only in an intravenous (IV) formulation.
If approved, isavuconazonium would provide an alternative to voriconazole for treating aspergillosis, and the IV formulation does not contain cyclodextrin, unlike the IV formulation of voriconazole, which limits its use in patients with moderate to severe renal dysfunction, according to Astellas. Safety concerns specific to isavuconazonium include QT-segment shortening and particulate formation in the IV formulation, according to the FDA.
The randomized, double-blind, international, noninferiority study compared treatment with isavuconazonium to voriconazole in 516 adults with invasive aspergillosis. In the randomized study, the mean age of patients was 51 years, 60% were men, most were white, 11% were in the United States and Canada, 20% had had an allogeneic bone marrow transplant, and 70% had an uncontrolled malignancy at baseline (Infect. Drug Resist. 2013;6:16374). The primary effectiveness endpoint, all-cause mortality through day 42, was 19% in those on isavuconazonium, compared with 20% in those on voriconazole. The study met the prespecified noninferiority margin. The rates of deaths and serious adverse events were similar to voriconazole, and there were fewer adverse events in those on isavuconazonium requiring discontinuation of the drug (14% vs. 23%).
Differences in adverse events included lower rates of hepatobiliary disorders, including hyperbilirubinemia, abnormal hepatic function, and jaundice (9% vs. 16%); skin and subcutaneous tissue disorders, including rash, erythema, and drug eruption (34% vs 43%) associated with isavuconazonium, compared with voriconazole. Common adverse events included nausea (28%), vomiting (25%), and diarrhea (24%). Decreases in the QT segment occurred in 7.5% of those on isavuconazonium, compared with 4.5% of those on voriconazole, but were not associated with clinical events.
The prospective, open-label, single-arm study evaluated isavuconazonium in 37 patients with proven or probable mucormycosis infections, whose mean age was 49 years; 59% had a hematologic malignancy, and about 40% were neutropenic at baseline. All-cause mortality at day 42 was almost 38%, which was similar to the mortality rate for amphotericin in the literature, according to Astellas.
There are about 12,000 cases of aspergillosis and about 500 cases of mucormycosis in the United States every year, the company said. In addition to voriconazole, other drugs approved for invasive aspergillosis include amphotericin formulations, itraconazole, and caspofungin. The FDA usually follows the recommendations of its advisory panels. The FDA is expected to make a decision by March 8, according to Astellas, which plans to market the drug as Cresemba if approved. It is also under review in Europe for the same indications.
Panelists were cleared of potential conflicts of interest related to the topic of the meeting. In some cases, a panelist may be given a waiver but not at this meeting.
SILVER SPRING, MD. – A novel treatment for invasive aspergillosis and invasive mucormycosis gained the support of a Food and Drug Administration advisory panel, although members were more ambivalent about the mucormycosis indication, based on the small amount of data available in those patients.
At a meeting on Jan. 22, the FDA’s Anti-Infective Drugs Advisory Committee voted 11-0 that there was “substantial evidence” that isavuconazonium, an antifungal prodrug, was safe and effective for the treatment of invasive aspergillosis, a life-threatening condition most commonly seen in immunocompromised patients. The drug was studied in a phase III study comparing isavuconazonium to voriconazole, the standard of care, in more than 500 patients.
The panel voted 8-2, with one abstention, that there was substantial evidence it was safe and effective for treating patients with invasive mucormycosis, with panelists voting on both sides of the question citing the study of only 37 patients that used historical controls as an issue. Those supporting approval for mucormycosis said that, if approved, the manufacturer, Astellas, should be required to conduct a phase IV trial further evaluating this treatment for patients with this infection.
Isavuconazonium is a prodrug of isavuconazole, a triazole antifungal, and would be available in an oral capsule formulation and a powder formulation that is reconstituted for intravenous administration (and needs to be administered through an in-line filter in case of particulate formation).
For aspergillosis, “I do believe that this drug provides a reasonable alternative to the current therapies that are available without additional toxicities,” said panelist Dr. Paige Waterman of the Global Emerging Infections Surveillance and Response System at the Walter Reed Army Medical Center, Silver Spring, Md. Other panelists agreed with her recommendations that labeling should make clear that it should not be used in people under age 18 years or in pregnant women, that a filter should be used when administered intravenously, and that labeling should include a statement about the increased risk of hepatotoxicity that also appears in the labeling of other drugs in the same class.
Because it has been associated with a shortened QT interval, she said that screening ECGs should be recommended for patients and that there should be some extra caution when it is prescribed to people who are Asian or of Asian descent, since drug concentrations were slightly higher among Asian patients who received the drug. Other recommendations from panelists included the need to conduct studies of the drug to provide information on therapeutic drug monitoring and in people under 18 years.
For treatment of invasive mucormycosis, an even rarer fungal infection which, in hospitals, is associated with the use of contaminated materials or organ transplantation, the panelists were more hesitant, but those voting in favor of approval cited the significance of the condition and the reasonable efficacy results in the study, stressing that postmarketing studies were critical. Panelists also noted that more clinical data are clearly needed in patients with this infection and the lack of data directly comparing it to amphotericin B – the only FDA-approved drug for this indication – was problematic.
“This drug really does fill an unmet need; I have high hopes that it is at least as good as amphotericin. But I do think we need more data to confirm that,” said Dr. Michael Neely, chair of antimicrobial stewardship at Children’s Hospital, Los Angeles, who was among those voting in favor of this indication. It appears to have a better safety profile and it would be available in both oral and intravenous formulations, he added. Amphotericin B is available only in an intravenous (IV) formulation.
If approved, isavuconazonium would provide an alternative to voriconazole for treating aspergillosis, and the IV formulation does not contain cyclodextrin, unlike the IV formulation of voriconazole, which limits its use in patients with moderate to severe renal dysfunction, according to Astellas. Safety concerns specific to isavuconazonium include QT-segment shortening and particulate formation in the IV formulation, according to the FDA.
The randomized, double-blind, international, noninferiority study compared treatment with isavuconazonium to voriconazole in 516 adults with invasive aspergillosis. In the randomized study, the mean age of patients was 51 years, 60% were men, most were white, 11% were in the United States and Canada, 20% had had an allogeneic bone marrow transplant, and 70% had an uncontrolled malignancy at baseline (Infect. Drug Resist. 2013;6:16374). The primary effectiveness endpoint, all-cause mortality through day 42, was 19% in those on isavuconazonium, compared with 20% in those on voriconazole. The study met the prespecified noninferiority margin. The rates of deaths and serious adverse events were similar to voriconazole, and there were fewer adverse events in those on isavuconazonium requiring discontinuation of the drug (14% vs. 23%).
Differences in adverse events included lower rates of hepatobiliary disorders, including hyperbilirubinemia, abnormal hepatic function, and jaundice (9% vs. 16%); skin and subcutaneous tissue disorders, including rash, erythema, and drug eruption (34% vs 43%) associated with isavuconazonium, compared with voriconazole. Common adverse events included nausea (28%), vomiting (25%), and diarrhea (24%). Decreases in the QT segment occurred in 7.5% of those on isavuconazonium, compared with 4.5% of those on voriconazole, but were not associated with clinical events.
The prospective, open-label, single-arm study evaluated isavuconazonium in 37 patients with proven or probable mucormycosis infections, whose mean age was 49 years; 59% had a hematologic malignancy, and about 40% were neutropenic at baseline. All-cause mortality at day 42 was almost 38%, which was similar to the mortality rate for amphotericin in the literature, according to Astellas.
There are about 12,000 cases of aspergillosis and about 500 cases of mucormycosis in the United States every year, the company said. In addition to voriconazole, other drugs approved for invasive aspergillosis include amphotericin formulations, itraconazole, and caspofungin. The FDA usually follows the recommendations of its advisory panels. The FDA is expected to make a decision by March 8, according to Astellas, which plans to market the drug as Cresemba if approved. It is also under review in Europe for the same indications.
Panelists were cleared of potential conflicts of interest related to the topic of the meeting. In some cases, a panelist may be given a waiver but not at this meeting.
AT AN FDA ADVISORY COMMITTEE MEETING
Pertussis persists
The Centers for Disease Control and Prevention suggests that recurring pertussis outbreaks may be the “new normal.” Such outbreaks show that some of what we “know” about pertussis is still correct, but some things are evolving. So in this new year, what do we need to know about patient vulnerability post vaccine as well as the clinical course, diagnosis, and treatment of this stubborn persisting disease?
Vulnerability after acellular pertussis vaccine
The recent large 2014 California outbreak surpassed the record numbers for the previously highest incidence year, 2010 (MMWR 2014;63:1129-32). This is scary because more cases had been reported in California in 2010 than in any prior year since the 1940s. The overall 2014 California pertussis rate (26/100,000 population) was approximately 10 times the national average for the first 9 years of this century, Are there clues as to who is most vulnerable and why?
No age group was spared, but certain age groups did appear more vulnerable. Infants had a startling 174.6/100,000 incidence (six times the rate for the overall population). It is not surprising to any clinician that infants less than 1 year of age were hardest hit. Infants have the most evident symptoms with pertussis. Infants also have 5-7 months of their first year in which they are incompletely immunized. Therefore, many are not expected to be protected until about 7-9 months of age. This vulnerability could be partly remedied if all pregnant women got Tdap boosters as recommended during pregnancy.
Of note, 15-year-olds had an incidence similar to that of infants (137.8/100,000). Ethnically, non-Hispanic whites had the highest incidence among adolescents (166.2/100,000), compared with Hispanics (64.2/100,000), Asian/Pacific Islanders (43.9/100,000), and non-Hispanic blacks (23.7/100,000). Disturbingly, 87% of cases among 15-year-olds had received a prior Tdap booster dose (median time since booster Tdap = 3 years, range = 0-7 years). Previous data from the 2010 outbreak suggested that immunity to pertussis wanes 3-4 years after receipt of the last acellular pertussis (aP)–containing vaccine. This is likely part of the explanation in 2014 as well. However, waning immunity after the booster does not explain why non-Hispanic whites had two to six times the incidence of other ethnicities. Non-Hispanic whites are thought to be the demographic with the most vaccine refusal and vaccine delay in California, so this may partially explain excess cases. Racial differences in access to care or genetic differences in disease susceptibility also may play a role.
Why is this biphasic increase in incidence in California a microcosm of the new epidemiology of pertussis in the United States? A kinder, gentler DTaP vaccine replaced the whole-cell DTP in the late 1990s. This occurred in response to the public’s concern about potential central nervous system adverse effects associated with the whole-cell DTP vaccine. Immunogenicity studies seemed to show equivalent immune responses in infants and toddlers receiving DTaP, compared with those who received DTP. It has only been in the last 5 years that we now know that the new DTaP and Tdap are not working as well as we had hoped.
The two aspects to the lesser protection provided by aP vaccines are pertactin-deficient pertussis strains and quicker waning of aP vaccine–induced immunity. Antibody to pertactin appears to be important in protection against clinical pertussis. New circulating clinical strains of pertussis may not have pertactin (N. Engl. J. Med. 2013;368:583-4). The strains used in our current DTaP and Tdap were designed to protect against pertactin-containing strains and were tested for this. This means that a proportion of the antibodies induced by vaccine strains are not useful against pertactin-deficient strains. The aP vaccine still induces antibody to the pertussis toxin and other pertussis components in the vaccines, so they will likely still reduce the severity of disease. But the vaccines are not likely to prevent infections from pertactin-deficient strains. This is similar to the partial vaccine mismatch that we are seeing with the current seasonal H3N2 influenza vaccine strain.
The second aspect is that protection appears to wane approximately 3-5 years after the last dose of aP-containing vaccine. This contrasts sharply with expectations in the past of 7-10 years of protection from whole cell pertussis–containing vaccines. The less reactive aP vaccine produces fewer adverse effects by not producing as much inflammation as DPT. The problem is that part of the reason the DPT has such good protective responses is the amount of inflammation it produces. So with less aP vaccine–induced inflammation comes less robust antibody and T-cell responses.
Nevertheless, the current acellular pertussis vaccines remain the most effective available tools to reduce pertussis disease (Cochrane Database Syst. Rev. 2014;9:CD001478]). But until we have new versions of pertussis vaccines that address these two issues, we clinicians need to remain vigilant for signs and symptoms of pertussis.
Clinical course
Remember that a whoop is rarely seen in young children and often also not seen when older patients present. The many outbreaks over the last 10 years have confirmed that paroxysmal cough with/without apnea in an infant/toddler should raise our index of suspicion. Likewise, older children, adolescents, and adults with persistent cough beyond 2 weeks are potential pertussis cases. Once the diagnosis is made, treatment is not expected to have a major impact on the clinical course, in part because the diagnosis is usually delayed (more than 10 days into symptoms). This delay allows more injury to the respiratory mucosa and cilia so that healing can require 6-12 weeks after bacterial replication ceases. This prolonged healing process is what is mostly responsible for the syndrome known as the “100-day cough.” So the clinical course of pertussis has not changed in the last 10 years. However, there have been changes in the commonly used diagnostic approach.
Pertussis diagnosis and contagion
During the last 5 years, polymerase chain reaction (PCR) testing has become the preferred technology to detect pertussis. This is due to the sensitivity and quick turnaround time of the assay. The gold standard for pertussis diagnosis remains culture, but it is expensive, cumbersome, and slow (up to a week to provide results). An ongoing debate arose about how long PCR testing remains positive after the onset of symptoms or treatment. This was not the problem when culture was the diagnostic tool of choice. Data from the 1970s and 1980s indicated that cultures were rarely positive after the third week of symptoms even without treatment. Furthermore, macrolides eliminated both contagion and positive culture results of infected patients after 5 days of treatment.
So now that we use PCR most often for diagnosis, what is the outer limit of positivity? A recent prospective cohort study from Salt Lake City suggests that PCR may detect pertussis DNA way beyond 3 weeks after symptom onset (J. Ped. Infect. Dis. 2014;3:347-9). Among patients hospitalized with laboratory-confirmed Bordetella pertussis infection, half had persistently positive pertussis PCR testing more than 50 days after symptom onset, despite antibiotic treatment and clinical improvement. The median (range) for the last day for a positive test after symptom onset was 58 days (4-172 days).
This raises the question as to whether there are viable pertussis organisms in the respiratory tract beyond the traditional 3 weeks defined by culture data. It is likely that DNA persists in the thick mucus of the respiratory tract way beyond viability of the last pertussis organisms. Put another way, PCR likely detects bacterial corpses or components way beyond the time that the patient is contagious. Unfortunately, current PCR data do not tell us how long patients remain contagious with the current strains of pertussis as infecting agents. Some institutions appear to be extending the isolation time for patients treated for pertussis beyond the traditional 5 days post initiation of effective treatment. Until more data are available, we are somewhat in the dark. But I would take comfort in the fact that it is unlikely the “new” data will be much different from those derived from the traditional studies that use culture to define infectivity. The American Academy of Pediatrics Committee on Infectious Diseases Red Book appears to agree.
For hospitalized pertussis patients, the AAP Committee on Infectious Diseases Red Book recommends standard and droplet precautions for 5 days after starting effective therapy, or 3 weeks after cough onset if appropriate antimicrobial therapy has not been given.
In addition, the CDC states: “PCR has optimal sensitivity during the first 3 weeks of cough when bacterial DNA is still present in the nasopharynx. After the fourth week of cough, the amount of bacterial DNA rapidly diminishes, which increases the risk of obtaining falsely negative results.” Later in the same document, the CDC says: “PCR testing following antibiotic therapy also can result in falsely negative findings. The exact duration of positivity following antibiotic use is not well understood, but PCR testing after 5 days of antibiotic use is unlikely to be of benefit and is generally not recommended.”
So what do we know? Not all PCR assays use the same primers, so some variance from the usual experience of up to 4 weeks of positive PCR results may be due to differences in the assays. But this raises concern that the PCR that you order may be positive at times when the patient is no longer contagious.
Pertussis treatment
If strains of pertussis have changed their pertactin antigen, are they changing their antibiotic susceptibility patterns? While there have been reports of macrolide resistance in a few pertussis strains, these still remain rare. The most recent comprehensive review of treatment efficacy was a Cochrane review performed in 2005 and published in 2007 (Cochrane Database Syst. Rev. 2007;3:CD004404). They evaluated 10 trials from 1969 to 2004 in which microbiologic eradication was defined by negative results from repeat pertussis culture. While meta-analysis of microbiologic eradication was not possible because of differences in antibiotic use, the investigators did conclude that antibiotic treatment “is effective in eliminating B. pertussis from patients with the disease to render them noninfectious, but does not alter the subsequent clinical course of the illness.”
Further, they state that “the best regimens for microbiologic clearance, with fewer side effects,” are 3 days of azithromycin (a single 10-mg/kg dose on 3 consecutive days) or 7 days of clarithromycin (7.5-mg/kg dose twice daily).
Another effective regimen is 14 days of erythromycin ethylsuccinate (60 mg/kg per day in 3 divided doses) .
CDC treatment recommendations include azithromycin or erythromycin, with trimethoprim-sulfamethoxazole as a possibility for macrolide-intolerant patients, although there are less data and success rates may not be as high.
Conclusion
So what do we know now about pertussis?
• Outbreaks are ongoing and likely will continue until newer more effective vaccines are produced, including those that circumvent the problem of pertactin-deficient strains.
• Pertussis is likely contagious up to 5 days on effective therapy, and for as long as 3 weeks if effective therapy has not been administered.
• PCR is a sensitive test that may remain positive for many weeks beyond contagion.
• Treatment with macrolides appears to be the most effective way to eradicate replicating pertussis pathogens.
• Treatment is not likely to have a major impact on the clinical course of disease because most of the damage to the respiratory tract is done prior to diagnosis and treatment. Treatment does reduce infectivity and subsequent cases.
• Current aP vaccines currently are our best preventative tools – including use in pregnant women to protect young infants.
As clinicians, our best course is to continue to immunize with the current vaccines, and remain vigilant for symptoms and signs of pertussis infection of patients so that early diagnosis and treatment can prevent further spread.
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics, Kansas City, Mo. Children’s Mercy Hospitals receives funds from GlaxoSmithKline for Dr. Harrison being principal investigator on a multicenter research study of a hexavalent pertussis-containing infant vaccine. E-mail Dr. Harrison at [email protected].
The Centers for Disease Control and Prevention suggests that recurring pertussis outbreaks may be the “new normal.” Such outbreaks show that some of what we “know” about pertussis is still correct, but some things are evolving. So in this new year, what do we need to know about patient vulnerability post vaccine as well as the clinical course, diagnosis, and treatment of this stubborn persisting disease?
Vulnerability after acellular pertussis vaccine
The recent large 2014 California outbreak surpassed the record numbers for the previously highest incidence year, 2010 (MMWR 2014;63:1129-32). This is scary because more cases had been reported in California in 2010 than in any prior year since the 1940s. The overall 2014 California pertussis rate (26/100,000 population) was approximately 10 times the national average for the first 9 years of this century, Are there clues as to who is most vulnerable and why?
No age group was spared, but certain age groups did appear more vulnerable. Infants had a startling 174.6/100,000 incidence (six times the rate for the overall population). It is not surprising to any clinician that infants less than 1 year of age were hardest hit. Infants have the most evident symptoms with pertussis. Infants also have 5-7 months of their first year in which they are incompletely immunized. Therefore, many are not expected to be protected until about 7-9 months of age. This vulnerability could be partly remedied if all pregnant women got Tdap boosters as recommended during pregnancy.
Of note, 15-year-olds had an incidence similar to that of infants (137.8/100,000). Ethnically, non-Hispanic whites had the highest incidence among adolescents (166.2/100,000), compared with Hispanics (64.2/100,000), Asian/Pacific Islanders (43.9/100,000), and non-Hispanic blacks (23.7/100,000). Disturbingly, 87% of cases among 15-year-olds had received a prior Tdap booster dose (median time since booster Tdap = 3 years, range = 0-7 years). Previous data from the 2010 outbreak suggested that immunity to pertussis wanes 3-4 years after receipt of the last acellular pertussis (aP)–containing vaccine. This is likely part of the explanation in 2014 as well. However, waning immunity after the booster does not explain why non-Hispanic whites had two to six times the incidence of other ethnicities. Non-Hispanic whites are thought to be the demographic with the most vaccine refusal and vaccine delay in California, so this may partially explain excess cases. Racial differences in access to care or genetic differences in disease susceptibility also may play a role.
Why is this biphasic increase in incidence in California a microcosm of the new epidemiology of pertussis in the United States? A kinder, gentler DTaP vaccine replaced the whole-cell DTP in the late 1990s. This occurred in response to the public’s concern about potential central nervous system adverse effects associated with the whole-cell DTP vaccine. Immunogenicity studies seemed to show equivalent immune responses in infants and toddlers receiving DTaP, compared with those who received DTP. It has only been in the last 5 years that we now know that the new DTaP and Tdap are not working as well as we had hoped.
The two aspects to the lesser protection provided by aP vaccines are pertactin-deficient pertussis strains and quicker waning of aP vaccine–induced immunity. Antibody to pertactin appears to be important in protection against clinical pertussis. New circulating clinical strains of pertussis may not have pertactin (N. Engl. J. Med. 2013;368:583-4). The strains used in our current DTaP and Tdap were designed to protect against pertactin-containing strains and were tested for this. This means that a proportion of the antibodies induced by vaccine strains are not useful against pertactin-deficient strains. The aP vaccine still induces antibody to the pertussis toxin and other pertussis components in the vaccines, so they will likely still reduce the severity of disease. But the vaccines are not likely to prevent infections from pertactin-deficient strains. This is similar to the partial vaccine mismatch that we are seeing with the current seasonal H3N2 influenza vaccine strain.
The second aspect is that protection appears to wane approximately 3-5 years after the last dose of aP-containing vaccine. This contrasts sharply with expectations in the past of 7-10 years of protection from whole cell pertussis–containing vaccines. The less reactive aP vaccine produces fewer adverse effects by not producing as much inflammation as DPT. The problem is that part of the reason the DPT has such good protective responses is the amount of inflammation it produces. So with less aP vaccine–induced inflammation comes less robust antibody and T-cell responses.
Nevertheless, the current acellular pertussis vaccines remain the most effective available tools to reduce pertussis disease (Cochrane Database Syst. Rev. 2014;9:CD001478]). But until we have new versions of pertussis vaccines that address these two issues, we clinicians need to remain vigilant for signs and symptoms of pertussis.
Clinical course
Remember that a whoop is rarely seen in young children and often also not seen when older patients present. The many outbreaks over the last 10 years have confirmed that paroxysmal cough with/without apnea in an infant/toddler should raise our index of suspicion. Likewise, older children, adolescents, and adults with persistent cough beyond 2 weeks are potential pertussis cases. Once the diagnosis is made, treatment is not expected to have a major impact on the clinical course, in part because the diagnosis is usually delayed (more than 10 days into symptoms). This delay allows more injury to the respiratory mucosa and cilia so that healing can require 6-12 weeks after bacterial replication ceases. This prolonged healing process is what is mostly responsible for the syndrome known as the “100-day cough.” So the clinical course of pertussis has not changed in the last 10 years. However, there have been changes in the commonly used diagnostic approach.
Pertussis diagnosis and contagion
During the last 5 years, polymerase chain reaction (PCR) testing has become the preferred technology to detect pertussis. This is due to the sensitivity and quick turnaround time of the assay. The gold standard for pertussis diagnosis remains culture, but it is expensive, cumbersome, and slow (up to a week to provide results). An ongoing debate arose about how long PCR testing remains positive after the onset of symptoms or treatment. This was not the problem when culture was the diagnostic tool of choice. Data from the 1970s and 1980s indicated that cultures were rarely positive after the third week of symptoms even without treatment. Furthermore, macrolides eliminated both contagion and positive culture results of infected patients after 5 days of treatment.
So now that we use PCR most often for diagnosis, what is the outer limit of positivity? A recent prospective cohort study from Salt Lake City suggests that PCR may detect pertussis DNA way beyond 3 weeks after symptom onset (J. Ped. Infect. Dis. 2014;3:347-9). Among patients hospitalized with laboratory-confirmed Bordetella pertussis infection, half had persistently positive pertussis PCR testing more than 50 days after symptom onset, despite antibiotic treatment and clinical improvement. The median (range) for the last day for a positive test after symptom onset was 58 days (4-172 days).
This raises the question as to whether there are viable pertussis organisms in the respiratory tract beyond the traditional 3 weeks defined by culture data. It is likely that DNA persists in the thick mucus of the respiratory tract way beyond viability of the last pertussis organisms. Put another way, PCR likely detects bacterial corpses or components way beyond the time that the patient is contagious. Unfortunately, current PCR data do not tell us how long patients remain contagious with the current strains of pertussis as infecting agents. Some institutions appear to be extending the isolation time for patients treated for pertussis beyond the traditional 5 days post initiation of effective treatment. Until more data are available, we are somewhat in the dark. But I would take comfort in the fact that it is unlikely the “new” data will be much different from those derived from the traditional studies that use culture to define infectivity. The American Academy of Pediatrics Committee on Infectious Diseases Red Book appears to agree.
For hospitalized pertussis patients, the AAP Committee on Infectious Diseases Red Book recommends standard and droplet precautions for 5 days after starting effective therapy, or 3 weeks after cough onset if appropriate antimicrobial therapy has not been given.
In addition, the CDC states: “PCR has optimal sensitivity during the first 3 weeks of cough when bacterial DNA is still present in the nasopharynx. After the fourth week of cough, the amount of bacterial DNA rapidly diminishes, which increases the risk of obtaining falsely negative results.” Later in the same document, the CDC says: “PCR testing following antibiotic therapy also can result in falsely negative findings. The exact duration of positivity following antibiotic use is not well understood, but PCR testing after 5 days of antibiotic use is unlikely to be of benefit and is generally not recommended.”
So what do we know? Not all PCR assays use the same primers, so some variance from the usual experience of up to 4 weeks of positive PCR results may be due to differences in the assays. But this raises concern that the PCR that you order may be positive at times when the patient is no longer contagious.
Pertussis treatment
If strains of pertussis have changed their pertactin antigen, are they changing their antibiotic susceptibility patterns? While there have been reports of macrolide resistance in a few pertussis strains, these still remain rare. The most recent comprehensive review of treatment efficacy was a Cochrane review performed in 2005 and published in 2007 (Cochrane Database Syst. Rev. 2007;3:CD004404). They evaluated 10 trials from 1969 to 2004 in which microbiologic eradication was defined by negative results from repeat pertussis culture. While meta-analysis of microbiologic eradication was not possible because of differences in antibiotic use, the investigators did conclude that antibiotic treatment “is effective in eliminating B. pertussis from patients with the disease to render them noninfectious, but does not alter the subsequent clinical course of the illness.”
Further, they state that “the best regimens for microbiologic clearance, with fewer side effects,” are 3 days of azithromycin (a single 10-mg/kg dose on 3 consecutive days) or 7 days of clarithromycin (7.5-mg/kg dose twice daily).
Another effective regimen is 14 days of erythromycin ethylsuccinate (60 mg/kg per day in 3 divided doses) .
CDC treatment recommendations include azithromycin or erythromycin, with trimethoprim-sulfamethoxazole as a possibility for macrolide-intolerant patients, although there are less data and success rates may not be as high.
Conclusion
So what do we know now about pertussis?
• Outbreaks are ongoing and likely will continue until newer more effective vaccines are produced, including those that circumvent the problem of pertactin-deficient strains.
• Pertussis is likely contagious up to 5 days on effective therapy, and for as long as 3 weeks if effective therapy has not been administered.
• PCR is a sensitive test that may remain positive for many weeks beyond contagion.
• Treatment with macrolides appears to be the most effective way to eradicate replicating pertussis pathogens.
• Treatment is not likely to have a major impact on the clinical course of disease because most of the damage to the respiratory tract is done prior to diagnosis and treatment. Treatment does reduce infectivity and subsequent cases.
• Current aP vaccines currently are our best preventative tools – including use in pregnant women to protect young infants.
As clinicians, our best course is to continue to immunize with the current vaccines, and remain vigilant for symptoms and signs of pertussis infection of patients so that early diagnosis and treatment can prevent further spread.
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics, Kansas City, Mo. Children’s Mercy Hospitals receives funds from GlaxoSmithKline for Dr. Harrison being principal investigator on a multicenter research study of a hexavalent pertussis-containing infant vaccine. E-mail Dr. Harrison at [email protected].
The Centers for Disease Control and Prevention suggests that recurring pertussis outbreaks may be the “new normal.” Such outbreaks show that some of what we “know” about pertussis is still correct, but some things are evolving. So in this new year, what do we need to know about patient vulnerability post vaccine as well as the clinical course, diagnosis, and treatment of this stubborn persisting disease?
Vulnerability after acellular pertussis vaccine
The recent large 2014 California outbreak surpassed the record numbers for the previously highest incidence year, 2010 (MMWR 2014;63:1129-32). This is scary because more cases had been reported in California in 2010 than in any prior year since the 1940s. The overall 2014 California pertussis rate (26/100,000 population) was approximately 10 times the national average for the first 9 years of this century, Are there clues as to who is most vulnerable and why?
No age group was spared, but certain age groups did appear more vulnerable. Infants had a startling 174.6/100,000 incidence (six times the rate for the overall population). It is not surprising to any clinician that infants less than 1 year of age were hardest hit. Infants have the most evident symptoms with pertussis. Infants also have 5-7 months of their first year in which they are incompletely immunized. Therefore, many are not expected to be protected until about 7-9 months of age. This vulnerability could be partly remedied if all pregnant women got Tdap boosters as recommended during pregnancy.
Of note, 15-year-olds had an incidence similar to that of infants (137.8/100,000). Ethnically, non-Hispanic whites had the highest incidence among adolescents (166.2/100,000), compared with Hispanics (64.2/100,000), Asian/Pacific Islanders (43.9/100,000), and non-Hispanic blacks (23.7/100,000). Disturbingly, 87% of cases among 15-year-olds had received a prior Tdap booster dose (median time since booster Tdap = 3 years, range = 0-7 years). Previous data from the 2010 outbreak suggested that immunity to pertussis wanes 3-4 years after receipt of the last acellular pertussis (aP)–containing vaccine. This is likely part of the explanation in 2014 as well. However, waning immunity after the booster does not explain why non-Hispanic whites had two to six times the incidence of other ethnicities. Non-Hispanic whites are thought to be the demographic with the most vaccine refusal and vaccine delay in California, so this may partially explain excess cases. Racial differences in access to care or genetic differences in disease susceptibility also may play a role.
Why is this biphasic increase in incidence in California a microcosm of the new epidemiology of pertussis in the United States? A kinder, gentler DTaP vaccine replaced the whole-cell DTP in the late 1990s. This occurred in response to the public’s concern about potential central nervous system adverse effects associated with the whole-cell DTP vaccine. Immunogenicity studies seemed to show equivalent immune responses in infants and toddlers receiving DTaP, compared with those who received DTP. It has only been in the last 5 years that we now know that the new DTaP and Tdap are not working as well as we had hoped.
The two aspects to the lesser protection provided by aP vaccines are pertactin-deficient pertussis strains and quicker waning of aP vaccine–induced immunity. Antibody to pertactin appears to be important in protection against clinical pertussis. New circulating clinical strains of pertussis may not have pertactin (N. Engl. J. Med. 2013;368:583-4). The strains used in our current DTaP and Tdap were designed to protect against pertactin-containing strains and were tested for this. This means that a proportion of the antibodies induced by vaccine strains are not useful against pertactin-deficient strains. The aP vaccine still induces antibody to the pertussis toxin and other pertussis components in the vaccines, so they will likely still reduce the severity of disease. But the vaccines are not likely to prevent infections from pertactin-deficient strains. This is similar to the partial vaccine mismatch that we are seeing with the current seasonal H3N2 influenza vaccine strain.
The second aspect is that protection appears to wane approximately 3-5 years after the last dose of aP-containing vaccine. This contrasts sharply with expectations in the past of 7-10 years of protection from whole cell pertussis–containing vaccines. The less reactive aP vaccine produces fewer adverse effects by not producing as much inflammation as DPT. The problem is that part of the reason the DPT has such good protective responses is the amount of inflammation it produces. So with less aP vaccine–induced inflammation comes less robust antibody and T-cell responses.
Nevertheless, the current acellular pertussis vaccines remain the most effective available tools to reduce pertussis disease (Cochrane Database Syst. Rev. 2014;9:CD001478]). But until we have new versions of pertussis vaccines that address these two issues, we clinicians need to remain vigilant for signs and symptoms of pertussis.
Clinical course
Remember that a whoop is rarely seen in young children and often also not seen when older patients present. The many outbreaks over the last 10 years have confirmed that paroxysmal cough with/without apnea in an infant/toddler should raise our index of suspicion. Likewise, older children, adolescents, and adults with persistent cough beyond 2 weeks are potential pertussis cases. Once the diagnosis is made, treatment is not expected to have a major impact on the clinical course, in part because the diagnosis is usually delayed (more than 10 days into symptoms). This delay allows more injury to the respiratory mucosa and cilia so that healing can require 6-12 weeks after bacterial replication ceases. This prolonged healing process is what is mostly responsible for the syndrome known as the “100-day cough.” So the clinical course of pertussis has not changed in the last 10 years. However, there have been changes in the commonly used diagnostic approach.
Pertussis diagnosis and contagion
During the last 5 years, polymerase chain reaction (PCR) testing has become the preferred technology to detect pertussis. This is due to the sensitivity and quick turnaround time of the assay. The gold standard for pertussis diagnosis remains culture, but it is expensive, cumbersome, and slow (up to a week to provide results). An ongoing debate arose about how long PCR testing remains positive after the onset of symptoms or treatment. This was not the problem when culture was the diagnostic tool of choice. Data from the 1970s and 1980s indicated that cultures were rarely positive after the third week of symptoms even without treatment. Furthermore, macrolides eliminated both contagion and positive culture results of infected patients after 5 days of treatment.
So now that we use PCR most often for diagnosis, what is the outer limit of positivity? A recent prospective cohort study from Salt Lake City suggests that PCR may detect pertussis DNA way beyond 3 weeks after symptom onset (J. Ped. Infect. Dis. 2014;3:347-9). Among patients hospitalized with laboratory-confirmed Bordetella pertussis infection, half had persistently positive pertussis PCR testing more than 50 days after symptom onset, despite antibiotic treatment and clinical improvement. The median (range) for the last day for a positive test after symptom onset was 58 days (4-172 days).
This raises the question as to whether there are viable pertussis organisms in the respiratory tract beyond the traditional 3 weeks defined by culture data. It is likely that DNA persists in the thick mucus of the respiratory tract way beyond viability of the last pertussis organisms. Put another way, PCR likely detects bacterial corpses or components way beyond the time that the patient is contagious. Unfortunately, current PCR data do not tell us how long patients remain contagious with the current strains of pertussis as infecting agents. Some institutions appear to be extending the isolation time for patients treated for pertussis beyond the traditional 5 days post initiation of effective treatment. Until more data are available, we are somewhat in the dark. But I would take comfort in the fact that it is unlikely the “new” data will be much different from those derived from the traditional studies that use culture to define infectivity. The American Academy of Pediatrics Committee on Infectious Diseases Red Book appears to agree.
For hospitalized pertussis patients, the AAP Committee on Infectious Diseases Red Book recommends standard and droplet precautions for 5 days after starting effective therapy, or 3 weeks after cough onset if appropriate antimicrobial therapy has not been given.
In addition, the CDC states: “PCR has optimal sensitivity during the first 3 weeks of cough when bacterial DNA is still present in the nasopharynx. After the fourth week of cough, the amount of bacterial DNA rapidly diminishes, which increases the risk of obtaining falsely negative results.” Later in the same document, the CDC says: “PCR testing following antibiotic therapy also can result in falsely negative findings. The exact duration of positivity following antibiotic use is not well understood, but PCR testing after 5 days of antibiotic use is unlikely to be of benefit and is generally not recommended.”
So what do we know? Not all PCR assays use the same primers, so some variance from the usual experience of up to 4 weeks of positive PCR results may be due to differences in the assays. But this raises concern that the PCR that you order may be positive at times when the patient is no longer contagious.
Pertussis treatment
If strains of pertussis have changed their pertactin antigen, are they changing their antibiotic susceptibility patterns? While there have been reports of macrolide resistance in a few pertussis strains, these still remain rare. The most recent comprehensive review of treatment efficacy was a Cochrane review performed in 2005 and published in 2007 (Cochrane Database Syst. Rev. 2007;3:CD004404). They evaluated 10 trials from 1969 to 2004 in which microbiologic eradication was defined by negative results from repeat pertussis culture. While meta-analysis of microbiologic eradication was not possible because of differences in antibiotic use, the investigators did conclude that antibiotic treatment “is effective in eliminating B. pertussis from patients with the disease to render them noninfectious, but does not alter the subsequent clinical course of the illness.”
Further, they state that “the best regimens for microbiologic clearance, with fewer side effects,” are 3 days of azithromycin (a single 10-mg/kg dose on 3 consecutive days) or 7 days of clarithromycin (7.5-mg/kg dose twice daily).
Another effective regimen is 14 days of erythromycin ethylsuccinate (60 mg/kg per day in 3 divided doses) .
CDC treatment recommendations include azithromycin or erythromycin, with trimethoprim-sulfamethoxazole as a possibility for macrolide-intolerant patients, although there are less data and success rates may not be as high.
Conclusion
So what do we know now about pertussis?
• Outbreaks are ongoing and likely will continue until newer more effective vaccines are produced, including those that circumvent the problem of pertactin-deficient strains.
• Pertussis is likely contagious up to 5 days on effective therapy, and for as long as 3 weeks if effective therapy has not been administered.
• PCR is a sensitive test that may remain positive for many weeks beyond contagion.
• Treatment with macrolides appears to be the most effective way to eradicate replicating pertussis pathogens.
• Treatment is not likely to have a major impact on the clinical course of disease because most of the damage to the respiratory tract is done prior to diagnosis and treatment. Treatment does reduce infectivity and subsequent cases.
• Current aP vaccines currently are our best preventative tools – including use in pregnant women to protect young infants.
As clinicians, our best course is to continue to immunize with the current vaccines, and remain vigilant for symptoms and signs of pertussis infection of patients so that early diagnosis and treatment can prevent further spread.
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics, Kansas City, Mo. Children’s Mercy Hospitals receives funds from GlaxoSmithKline for Dr. Harrison being principal investigator on a multicenter research study of a hexavalent pertussis-containing infant vaccine. E-mail Dr. Harrison at [email protected].
Lighter ventilator sedation protocol no better than usual care in the PICU
PHOENIX – A pediatric ICU protocol to minimize sedation during intubation did not reduce ventilator days among children with acute respiratory failure, compared with usual care, in a randomized trial of 2,449 children across 31 ICUs.
The results were published online Jan. 20 in JAMA.
Seventeen pediatric ICUs (PICUs) followed a protocol that included sedation adjustment at least every 8 hours based on phase of illness; arousal assessments during titration and weaning; daily extubation readiness testing when spontaneously breathing; and weaning off opioids and benzodiazepines if patients were on them for 5 or more days, instead of simply discontinuing them.
The 1,225 children on the protocol were ventilated a median of 6.5 days, the same length as the 1,224 children in the 14 usual-care PICUs. There were no differences in sedation-related adverse events, including inadequate pain and sedation control, clinically significant drug withdrawal symptoms, and unplanned endotracheal tube removal (JAMA 2015 [doi:10.1001/jama.2014.18399])
It’s become clear in recent years that intubated adults do better with lighter sedation, and the PICU findings don’t necessarily mean that children are somehow different. A more likely explanation of the findings is that the control PICUs were using a lighter touch, too; prior to randomization, all the PICUs implemented the same pain, sedation, and withdrawal scales.
In any case, the study does show that “targeting a sedation goal of patients who are calm, easily aroused, and readily evaluated is attainable and safe in children,” wrote registered nurse Martha Curley, Ph.D., of the University of Pennsylvania, Philadelphia, and her associates . The study results were published to coincide with Dr. Curley’s presentation at the Critical Care Congress, sponsored by the Society for Critical Care Medicine.
The protocol patients had a median 9 days of opioids, versus 10 in the usual-care group, and they were awake and calm while intubated a median of 86% of days, versus 75% in the control group. However, intervention children had pain during about half of their days, while usual-care children reported pain during about quarter of their days. Intervention patients also had more agitation, and postextubation stridor, but fewer pressure sores. There were no differences in mortality between the groups.
Morphine was the primary protocol opioid, while fentanyl was used most often in the control group. Morphine was selected for the protocol because it has a longer duration of action, plus some sedative properties and less development of tolerance. The primary sedative in both groups was midazolam; about a quarter of protocol patients received dexmedetomidine, versus about half in the usual-care group.
Baseline patient characteristics were well matched between the two groups, except that the intervention group enrolled more patients younger than 2 years and more patients with bronchiolitis, both of whom are harder to sedate. Pneumonia, bronchiolitis, and acute respiratory failure due to sepsis were the most common diagnoses in both groups. The mean age of the children was 4.7 years.
The work, dubbed the Randomized Evaluation of Sedation Titration for Respiratory Failure (RESTORE) study, was supported by grants from the National Heart, Lung, and Blood Institute and the National Institute of Nursing Research. The lead author had no disclosures. One coauthor is an advisor for Philips, and another reported consultant and other fees from Cerus, Quark Pharmaceuticals, Biogen, GlaxoSmithKline, and other companies.
“While it is disappointing that this trial showed no advantage of a complex sedation management strategy, it is reassuring that the overall clinical outcomes related to ‘usual care’ in the 14 control PICUs were not significantly different than protocolized sedation in the intervention PICUs.”
It’s likely “patients in the control group also were managed with goal-directed minimal sedation despite the lack of a formal protocol. ... Pediatric clinicians are certainly aware of the benefits of goal-directed sedation and are likely practicing it.
“The algorithm used in the study was complex, potentially affecting adherence. ... Perhaps a simpler, less structured algorithm would have been associated with positive results.
“Another question is whether adjustment of sedative doses every 8 hours is adequate and whether more frequent titration would have resulted in lower sedative exposure in the intervention group. Deepest sedation [was] in the acute phase [with] progressive lightening of sedation as the patient approached extubation. However, deep sedation in the early phase of illness may not be necessary and may be detrimental. ... It is possible that the intervention group was adversely affected by the deep sedation.”
Dr. Sangeeta Mehta is a critical care physician and respirologist at Mount Sinai Hospital in Toronto. She had no conflicts of interest, and wrote this editorial in response to Dr. Curley’s article (JAMA 2015 [doi:10.1001/jama.2015.1]).
“While it is disappointing that this trial showed no advantage of a complex sedation management strategy, it is reassuring that the overall clinical outcomes related to ‘usual care’ in the 14 control PICUs were not significantly different than protocolized sedation in the intervention PICUs.”
It’s likely “patients in the control group also were managed with goal-directed minimal sedation despite the lack of a formal protocol. ... Pediatric clinicians are certainly aware of the benefits of goal-directed sedation and are likely practicing it.
“The algorithm used in the study was complex, potentially affecting adherence. ... Perhaps a simpler, less structured algorithm would have been associated with positive results.
“Another question is whether adjustment of sedative doses every 8 hours is adequate and whether more frequent titration would have resulted in lower sedative exposure in the intervention group. Deepest sedation [was] in the acute phase [with] progressive lightening of sedation as the patient approached extubation. However, deep sedation in the early phase of illness may not be necessary and may be detrimental. ... It is possible that the intervention group was adversely affected by the deep sedation.”
Dr. Sangeeta Mehta is a critical care physician and respirologist at Mount Sinai Hospital in Toronto. She had no conflicts of interest, and wrote this editorial in response to Dr. Curley’s article (JAMA 2015 [doi:10.1001/jama.2015.1]).
“While it is disappointing that this trial showed no advantage of a complex sedation management strategy, it is reassuring that the overall clinical outcomes related to ‘usual care’ in the 14 control PICUs were not significantly different than protocolized sedation in the intervention PICUs.”
It’s likely “patients in the control group also were managed with goal-directed minimal sedation despite the lack of a formal protocol. ... Pediatric clinicians are certainly aware of the benefits of goal-directed sedation and are likely practicing it.
“The algorithm used in the study was complex, potentially affecting adherence. ... Perhaps a simpler, less structured algorithm would have been associated with positive results.
“Another question is whether adjustment of sedative doses every 8 hours is adequate and whether more frequent titration would have resulted in lower sedative exposure in the intervention group. Deepest sedation [was] in the acute phase [with] progressive lightening of sedation as the patient approached extubation. However, deep sedation in the early phase of illness may not be necessary and may be detrimental. ... It is possible that the intervention group was adversely affected by the deep sedation.”
Dr. Sangeeta Mehta is a critical care physician and respirologist at Mount Sinai Hospital in Toronto. She had no conflicts of interest, and wrote this editorial in response to Dr. Curley’s article (JAMA 2015 [doi:10.1001/jama.2015.1]).
PHOENIX – A pediatric ICU protocol to minimize sedation during intubation did not reduce ventilator days among children with acute respiratory failure, compared with usual care, in a randomized trial of 2,449 children across 31 ICUs.
The results were published online Jan. 20 in JAMA.
Seventeen pediatric ICUs (PICUs) followed a protocol that included sedation adjustment at least every 8 hours based on phase of illness; arousal assessments during titration and weaning; daily extubation readiness testing when spontaneously breathing; and weaning off opioids and benzodiazepines if patients were on them for 5 or more days, instead of simply discontinuing them.
The 1,225 children on the protocol were ventilated a median of 6.5 days, the same length as the 1,224 children in the 14 usual-care PICUs. There were no differences in sedation-related adverse events, including inadequate pain and sedation control, clinically significant drug withdrawal symptoms, and unplanned endotracheal tube removal (JAMA 2015 [doi:10.1001/jama.2014.18399])
It’s become clear in recent years that intubated adults do better with lighter sedation, and the PICU findings don’t necessarily mean that children are somehow different. A more likely explanation of the findings is that the control PICUs were using a lighter touch, too; prior to randomization, all the PICUs implemented the same pain, sedation, and withdrawal scales.
In any case, the study does show that “targeting a sedation goal of patients who are calm, easily aroused, and readily evaluated is attainable and safe in children,” wrote registered nurse Martha Curley, Ph.D., of the University of Pennsylvania, Philadelphia, and her associates . The study results were published to coincide with Dr. Curley’s presentation at the Critical Care Congress, sponsored by the Society for Critical Care Medicine.
The protocol patients had a median 9 days of opioids, versus 10 in the usual-care group, and they were awake and calm while intubated a median of 86% of days, versus 75% in the control group. However, intervention children had pain during about half of their days, while usual-care children reported pain during about quarter of their days. Intervention patients also had more agitation, and postextubation stridor, but fewer pressure sores. There were no differences in mortality between the groups.
Morphine was the primary protocol opioid, while fentanyl was used most often in the control group. Morphine was selected for the protocol because it has a longer duration of action, plus some sedative properties and less development of tolerance. The primary sedative in both groups was midazolam; about a quarter of protocol patients received dexmedetomidine, versus about half in the usual-care group.
Baseline patient characteristics were well matched between the two groups, except that the intervention group enrolled more patients younger than 2 years and more patients with bronchiolitis, both of whom are harder to sedate. Pneumonia, bronchiolitis, and acute respiratory failure due to sepsis were the most common diagnoses in both groups. The mean age of the children was 4.7 years.
The work, dubbed the Randomized Evaluation of Sedation Titration for Respiratory Failure (RESTORE) study, was supported by grants from the National Heart, Lung, and Blood Institute and the National Institute of Nursing Research. The lead author had no disclosures. One coauthor is an advisor for Philips, and another reported consultant and other fees from Cerus, Quark Pharmaceuticals, Biogen, GlaxoSmithKline, and other companies.
PHOENIX – A pediatric ICU protocol to minimize sedation during intubation did not reduce ventilator days among children with acute respiratory failure, compared with usual care, in a randomized trial of 2,449 children across 31 ICUs.
The results were published online Jan. 20 in JAMA.
Seventeen pediatric ICUs (PICUs) followed a protocol that included sedation adjustment at least every 8 hours based on phase of illness; arousal assessments during titration and weaning; daily extubation readiness testing when spontaneously breathing; and weaning off opioids and benzodiazepines if patients were on them for 5 or more days, instead of simply discontinuing them.
The 1,225 children on the protocol were ventilated a median of 6.5 days, the same length as the 1,224 children in the 14 usual-care PICUs. There were no differences in sedation-related adverse events, including inadequate pain and sedation control, clinically significant drug withdrawal symptoms, and unplanned endotracheal tube removal (JAMA 2015 [doi:10.1001/jama.2014.18399])
It’s become clear in recent years that intubated adults do better with lighter sedation, and the PICU findings don’t necessarily mean that children are somehow different. A more likely explanation of the findings is that the control PICUs were using a lighter touch, too; prior to randomization, all the PICUs implemented the same pain, sedation, and withdrawal scales.
In any case, the study does show that “targeting a sedation goal of patients who are calm, easily aroused, and readily evaluated is attainable and safe in children,” wrote registered nurse Martha Curley, Ph.D., of the University of Pennsylvania, Philadelphia, and her associates . The study results were published to coincide with Dr. Curley’s presentation at the Critical Care Congress, sponsored by the Society for Critical Care Medicine.
The protocol patients had a median 9 days of opioids, versus 10 in the usual-care group, and they were awake and calm while intubated a median of 86% of days, versus 75% in the control group. However, intervention children had pain during about half of their days, while usual-care children reported pain during about quarter of their days. Intervention patients also had more agitation, and postextubation stridor, but fewer pressure sores. There were no differences in mortality between the groups.
Morphine was the primary protocol opioid, while fentanyl was used most often in the control group. Morphine was selected for the protocol because it has a longer duration of action, plus some sedative properties and less development of tolerance. The primary sedative in both groups was midazolam; about a quarter of protocol patients received dexmedetomidine, versus about half in the usual-care group.
Baseline patient characteristics were well matched between the two groups, except that the intervention group enrolled more patients younger than 2 years and more patients with bronchiolitis, both of whom are harder to sedate. Pneumonia, bronchiolitis, and acute respiratory failure due to sepsis were the most common diagnoses in both groups. The mean age of the children was 4.7 years.
The work, dubbed the Randomized Evaluation of Sedation Titration for Respiratory Failure (RESTORE) study, was supported by grants from the National Heart, Lung, and Blood Institute and the National Institute of Nursing Research. The lead author had no disclosures. One coauthor is an advisor for Philips, and another reported consultant and other fees from Cerus, Quark Pharmaceuticals, Biogen, GlaxoSmithKline, and other companies.
AT THE CRITICAL CARE CONGRESS
Key clinical point: A standardized approach to minimize sedation is possible for intubated children.
Major finding: Children spent a median of 6.5 days on the ventilator, whether or not they were on a minimum sedation protocol.
Data source: Randomized, unblinded trial of 2,449 children across 31 PICUs.
Disclosures: The National Heart, Lung, and Blood Institute and the National Institute of Nursing Research funded the study. The lead author has no disclosures. One coauthor is an adviser for Philips, and another disclosed ties with Cerus, Quark Pharmaceuticals, Biogen, GlaxoSmithKline, and other companies.
Hospitalization for pneumonia raises CVD risk
Among older adults, hospitalization for pneumonia raises the short-term (1-month) and long-term (10-year) risk of cardiovascular disease events to a degree comparable to those of smoking, diabetes, and hypertension, according to a report published online Jan. 20 in JAMA.
“Our findings suggest that hospitalization for pneumonia should be considered an independent cardiovascular risk factor” and “should prompt clinical trials to test targeted strategies”’ to prevent the disease in this patient population, said Dr. Vicente F. Corrales-Medina of the University of Ottawa and the Ottawa Hospital Research Institute.
The risk of cardiovascular disease (CVD) events within 30 days of severe infections, mainly those involving the respiratory tract, is well established, but the more lasting effects are uncertain. In what the researchers described as “the first study to document the temporal variation in the long-term risk of CVD ... using rigorous methods to adjust for many potential confounders,” they analyzed data from two multicenter population-based cohorts that were followed for 21 years.
The 591 participants in the Cardiovascular Health Study were 65 years of age or older and had been hospitalized for pneumonia. When researchers compared their outcomes to 1,182 matched controls without pneumonia, they found that in the pneumonia group the rate of CVD events was fourfold higher at 30 days, dropped to twofold higher throughout the rest of the first year, and leveled out at 1.5-fold higher for the remainder of the decade.
Researchers then verified this risk pattern in a cohort of 680 pneumonia patients aged 45-64 years and 1,360 matched controls in the Atherosclerosis Risk in Communities study. The increased risk conferred by hospitalization for pneumonia persisted after the data were adjusted to account for demographic traits, preexisting CVD risk factors, and measures of patient frailty; it also was robust to numerous sensitivity analyses, the researchers said (JAMA 2015 Jan. 20 [doi:10.1001/jama.2014.18229]).
Moreover, the magnitude of risk conferred by hospitalization for pneumonia “was similar or higher, compared with the risk of CVD associated with traditional risk factors such as smoking, diabetes, and hypertension,” wrote Dr. Corrales-Medina and his associates.
The Ottawa Hospital, the Ottawa Hospital Research Institute, and the Canadian National Institute of General Medical Sciences funded the study. Dr. Corrales-Medina reported having no financial disclosures; an associate reported ties to Biogen Idec, Bristol-Myers Squibb/Pfizer, Boehringer Ingelheim, CardioNet, Daiichi Sankyo, diaDexus, Janssen, Merck/Organon, and Sanofi.
Among older adults, hospitalization for pneumonia raises the short-term (1-month) and long-term (10-year) risk of cardiovascular disease events to a degree comparable to those of smoking, diabetes, and hypertension, according to a report published online Jan. 20 in JAMA.
“Our findings suggest that hospitalization for pneumonia should be considered an independent cardiovascular risk factor” and “should prompt clinical trials to test targeted strategies”’ to prevent the disease in this patient population, said Dr. Vicente F. Corrales-Medina of the University of Ottawa and the Ottawa Hospital Research Institute.
The risk of cardiovascular disease (CVD) events within 30 days of severe infections, mainly those involving the respiratory tract, is well established, but the more lasting effects are uncertain. In what the researchers described as “the first study to document the temporal variation in the long-term risk of CVD ... using rigorous methods to adjust for many potential confounders,” they analyzed data from two multicenter population-based cohorts that were followed for 21 years.
The 591 participants in the Cardiovascular Health Study were 65 years of age or older and had been hospitalized for pneumonia. When researchers compared their outcomes to 1,182 matched controls without pneumonia, they found that in the pneumonia group the rate of CVD events was fourfold higher at 30 days, dropped to twofold higher throughout the rest of the first year, and leveled out at 1.5-fold higher for the remainder of the decade.
Researchers then verified this risk pattern in a cohort of 680 pneumonia patients aged 45-64 years and 1,360 matched controls in the Atherosclerosis Risk in Communities study. The increased risk conferred by hospitalization for pneumonia persisted after the data were adjusted to account for demographic traits, preexisting CVD risk factors, and measures of patient frailty; it also was robust to numerous sensitivity analyses, the researchers said (JAMA 2015 Jan. 20 [doi:10.1001/jama.2014.18229]).
Moreover, the magnitude of risk conferred by hospitalization for pneumonia “was similar or higher, compared with the risk of CVD associated with traditional risk factors such as smoking, diabetes, and hypertension,” wrote Dr. Corrales-Medina and his associates.
The Ottawa Hospital, the Ottawa Hospital Research Institute, and the Canadian National Institute of General Medical Sciences funded the study. Dr. Corrales-Medina reported having no financial disclosures; an associate reported ties to Biogen Idec, Bristol-Myers Squibb/Pfizer, Boehringer Ingelheim, CardioNet, Daiichi Sankyo, diaDexus, Janssen, Merck/Organon, and Sanofi.
Among older adults, hospitalization for pneumonia raises the short-term (1-month) and long-term (10-year) risk of cardiovascular disease events to a degree comparable to those of smoking, diabetes, and hypertension, according to a report published online Jan. 20 in JAMA.
“Our findings suggest that hospitalization for pneumonia should be considered an independent cardiovascular risk factor” and “should prompt clinical trials to test targeted strategies”’ to prevent the disease in this patient population, said Dr. Vicente F. Corrales-Medina of the University of Ottawa and the Ottawa Hospital Research Institute.
The risk of cardiovascular disease (CVD) events within 30 days of severe infections, mainly those involving the respiratory tract, is well established, but the more lasting effects are uncertain. In what the researchers described as “the first study to document the temporal variation in the long-term risk of CVD ... using rigorous methods to adjust for many potential confounders,” they analyzed data from two multicenter population-based cohorts that were followed for 21 years.
The 591 participants in the Cardiovascular Health Study were 65 years of age or older and had been hospitalized for pneumonia. When researchers compared their outcomes to 1,182 matched controls without pneumonia, they found that in the pneumonia group the rate of CVD events was fourfold higher at 30 days, dropped to twofold higher throughout the rest of the first year, and leveled out at 1.5-fold higher for the remainder of the decade.
Researchers then verified this risk pattern in a cohort of 680 pneumonia patients aged 45-64 years and 1,360 matched controls in the Atherosclerosis Risk in Communities study. The increased risk conferred by hospitalization for pneumonia persisted after the data were adjusted to account for demographic traits, preexisting CVD risk factors, and measures of patient frailty; it also was robust to numerous sensitivity analyses, the researchers said (JAMA 2015 Jan. 20 [doi:10.1001/jama.2014.18229]).
Moreover, the magnitude of risk conferred by hospitalization for pneumonia “was similar or higher, compared with the risk of CVD associated with traditional risk factors such as smoking, diabetes, and hypertension,” wrote Dr. Corrales-Medina and his associates.
The Ottawa Hospital, the Ottawa Hospital Research Institute, and the Canadian National Institute of General Medical Sciences funded the study. Dr. Corrales-Medina reported having no financial disclosures; an associate reported ties to Biogen Idec, Bristol-Myers Squibb/Pfizer, Boehringer Ingelheim, CardioNet, Daiichi Sankyo, diaDexus, Janssen, Merck/Organon, and Sanofi.
FROM JAMA
Key clinical point: Among older adults, hospitalization for pneumonia raises both short-term and long-term risks of a CVD event.
Major finding: The rate of CVD events was fourfold higher at 30 days, dropped to twofold higher throughout the rest of the first year, and leveled out at 1.5-fold higher for the remainder of the decade.
Data source: A secondary analysis of data from two multicenter population-based cohorts, of 5,888 and 15,792 older adults, followed for 21 years.
Disclosures: The Ottawa Hospital, the Ottawa Hospital Research Institute, and the Canadian National Institute of General Medical Sciences funded the study. Dr. Corrales-Medina reported having no financial disclosures; an associate reported ties to Biogen Idec, Bristol-Myers Squibb/Pfizer, Boehringer Ingelheim, CardioNet, Daiichi Sankyo, diaDexus, Janssen, Merck/Organon, and Sanofi.
IPAH patients’ nonresponse to vasodilator challenge linked to poor recruitment of capillary surface
Patients with idiopathic pulmonary arterial hypertension did not respond to treatment with an acute vasodilator challenge because they were unable to recruit functional capillary surface area to accommodate increased blood flow to the lungs, according to the results of a study published Jan. 19 in Annals of Internal Medicine.
Dr. David Langleben of McGill University, Montreal, and Dr. Stylianos E. Orfanos of Attikon Hospital, Athens, hypothesized that the increased pulmonary vascular resistance (PVR) noted in responders would be based on vascular tone rather than cellular obstruction, and therefore responders should be capable of recruiting functional capillary surface area (CSA) during a vasodilator challenge.
“Despite sharing the baseline hemodynamic profile of IPAH, responders and nonresponders [may] represent different vascular phenotypes and perhaps different diseases,” they wrote.
The investigators recruited 14 patients (12 female, 2 male) with IPAH, none of whom had previously received IPAH-specific therapy or had saline contrast evidence of intracardiac shunting on echocardiography. Each subject underwent a standardized right heart catheterization followed by insertion of a femoral artery sheath. After a 30-minute rest period, investigators measured baseline hemodynamics and pulmonary capillary, endothelium-bound, angiotensin-converting enzyme (PCEB-ACE) metabolic activity.
Subsequently, the researchers performed an acute vasodilator challenge on each patient using epoprostenol (Flolan, GlaxoSmithKline Canada), after which PCEB-ACE metabolic activity and hemodynamics were measured again at the peak epoprostenol dose – determined “individually by maximum hemodynamic response or adverse effects.”
Functional CSA was calculated and normalized to body surface area. Values among nonresponders before the vasodilator challenge and at the peak epoprostenol dose were compared using paired t-tests. Of the 14 subjects, 12 were found to be nonresponders to vasodilator challenge and 2 (1 female, 1 male) did respond to vasodilator challenge treatment. In nonresponders overall, mean pulmonary arterial pressure (mPAP) did not change significantly – 59.5 mm Hg to 58.1 mm Hg – but cardiac output increased and PVR decreased. In the two subjects who responded, cardiac output and PVR increased, but mPAP decreased 43.5 mm Hg to 31.5 mm Hg.
“These data show the inability of nonresponders with IPAH to open occluded arterioles and thus recruit more downstream capillaries to accommodate the observed increased lung blood volumes that should accompany vasodilator administration,” the researchers concluded.
Funding for this study was provided by the William and Ida Pencer Family Foundation, the Dimitrios Banousis Foundation, the Bank of Montreal Center for the Study of Heart Disease in Women, and the Jewish General Hospital Annual Walk for Pulmonary Hypertension, all at Jewish General Hospital. The authors disclosed no conflicts of interest.
Patients with idiopathic pulmonary arterial hypertension did not respond to treatment with an acute vasodilator challenge because they were unable to recruit functional capillary surface area to accommodate increased blood flow to the lungs, according to the results of a study published Jan. 19 in Annals of Internal Medicine.
Dr. David Langleben of McGill University, Montreal, and Dr. Stylianos E. Orfanos of Attikon Hospital, Athens, hypothesized that the increased pulmonary vascular resistance (PVR) noted in responders would be based on vascular tone rather than cellular obstruction, and therefore responders should be capable of recruiting functional capillary surface area (CSA) during a vasodilator challenge.
“Despite sharing the baseline hemodynamic profile of IPAH, responders and nonresponders [may] represent different vascular phenotypes and perhaps different diseases,” they wrote.
The investigators recruited 14 patients (12 female, 2 male) with IPAH, none of whom had previously received IPAH-specific therapy or had saline contrast evidence of intracardiac shunting on echocardiography. Each subject underwent a standardized right heart catheterization followed by insertion of a femoral artery sheath. After a 30-minute rest period, investigators measured baseline hemodynamics and pulmonary capillary, endothelium-bound, angiotensin-converting enzyme (PCEB-ACE) metabolic activity.
Subsequently, the researchers performed an acute vasodilator challenge on each patient using epoprostenol (Flolan, GlaxoSmithKline Canada), after which PCEB-ACE metabolic activity and hemodynamics were measured again at the peak epoprostenol dose – determined “individually by maximum hemodynamic response or adverse effects.”
Functional CSA was calculated and normalized to body surface area. Values among nonresponders before the vasodilator challenge and at the peak epoprostenol dose were compared using paired t-tests. Of the 14 subjects, 12 were found to be nonresponders to vasodilator challenge and 2 (1 female, 1 male) did respond to vasodilator challenge treatment. In nonresponders overall, mean pulmonary arterial pressure (mPAP) did not change significantly – 59.5 mm Hg to 58.1 mm Hg – but cardiac output increased and PVR decreased. In the two subjects who responded, cardiac output and PVR increased, but mPAP decreased 43.5 mm Hg to 31.5 mm Hg.
“These data show the inability of nonresponders with IPAH to open occluded arterioles and thus recruit more downstream capillaries to accommodate the observed increased lung blood volumes that should accompany vasodilator administration,” the researchers concluded.
Funding for this study was provided by the William and Ida Pencer Family Foundation, the Dimitrios Banousis Foundation, the Bank of Montreal Center for the Study of Heart Disease in Women, and the Jewish General Hospital Annual Walk for Pulmonary Hypertension, all at Jewish General Hospital. The authors disclosed no conflicts of interest.
Patients with idiopathic pulmonary arterial hypertension did not respond to treatment with an acute vasodilator challenge because they were unable to recruit functional capillary surface area to accommodate increased blood flow to the lungs, according to the results of a study published Jan. 19 in Annals of Internal Medicine.
Dr. David Langleben of McGill University, Montreal, and Dr. Stylianos E. Orfanos of Attikon Hospital, Athens, hypothesized that the increased pulmonary vascular resistance (PVR) noted in responders would be based on vascular tone rather than cellular obstruction, and therefore responders should be capable of recruiting functional capillary surface area (CSA) during a vasodilator challenge.
“Despite sharing the baseline hemodynamic profile of IPAH, responders and nonresponders [may] represent different vascular phenotypes and perhaps different diseases,” they wrote.
The investigators recruited 14 patients (12 female, 2 male) with IPAH, none of whom had previously received IPAH-specific therapy or had saline contrast evidence of intracardiac shunting on echocardiography. Each subject underwent a standardized right heart catheterization followed by insertion of a femoral artery sheath. After a 30-minute rest period, investigators measured baseline hemodynamics and pulmonary capillary, endothelium-bound, angiotensin-converting enzyme (PCEB-ACE) metabolic activity.
Subsequently, the researchers performed an acute vasodilator challenge on each patient using epoprostenol (Flolan, GlaxoSmithKline Canada), after which PCEB-ACE metabolic activity and hemodynamics were measured again at the peak epoprostenol dose – determined “individually by maximum hemodynamic response or adverse effects.”
Functional CSA was calculated and normalized to body surface area. Values among nonresponders before the vasodilator challenge and at the peak epoprostenol dose were compared using paired t-tests. Of the 14 subjects, 12 were found to be nonresponders to vasodilator challenge and 2 (1 female, 1 male) did respond to vasodilator challenge treatment. In nonresponders overall, mean pulmonary arterial pressure (mPAP) did not change significantly – 59.5 mm Hg to 58.1 mm Hg – but cardiac output increased and PVR decreased. In the two subjects who responded, cardiac output and PVR increased, but mPAP decreased 43.5 mm Hg to 31.5 mm Hg.
“These data show the inability of nonresponders with IPAH to open occluded arterioles and thus recruit more downstream capillaries to accommodate the observed increased lung blood volumes that should accompany vasodilator administration,” the researchers concluded.
Funding for this study was provided by the William and Ida Pencer Family Foundation, the Dimitrios Banousis Foundation, the Bank of Montreal Center for the Study of Heart Disease in Women, and the Jewish General Hospital Annual Walk for Pulmonary Hypertension, all at Jewish General Hospital. The authors disclosed no conflicts of interest.
FROM ANNALS OF INTERNAL MEDICINE
Key clinical point: Differential responses by patients with idiopathic pulmonary arterial hypertension (IPAH) to acute vasodilator challenge suggests that responders and nonresponders represent different vascular phenotypes and different diseases.
Major finding: In nonresponders to vasodilator challenge, mPAP increased and PVR decreased, while those who did respond saw both levels decrease; both cohorts experienced an increase in cardiac output.
Data source: Cohort study of 14 individuals with IPAH.
Disclosures: Funding provided by the William and Ida Pencer Family Foundation, the Dimitrios Banousis Foundation, the Bank of Montreal Center for the Study of Heart Disease in Women, and the Jewish General Hospital Annual Walk for Pulmonary Hypertension, all at Jewish General Hospital. The authors disclosed no conflicts of interest.
So far, flu vaccine only 23% effective
So far this flu season, more than two-thirds of influenza A (H3N2) viruses differ from the components of the 2014-2015 influenza vaccine. In addition, the overall estimated effectiveness of the 2014-2015 influenza vaccine for preventing laboratory-confirmed influenza infection is only 23%.
Those are key findings from an analysis of 2,321 children and adults who presented to one of five study sites in the United States with acute respiratory illness between Nov. 10, 2014, and Jan. 2, 2015.
“Although influenza vaccines are the best tool for prevention of influenza currently available, more effective vaccines are needed,” wrote Brendan Flannery, Ph.D., of the influenza division at the CDC’s National Center for Immunization and Respiratory Disease, and his associates. The report is in the Jan. 16, 2015, issue of the CDC’s Morbidity and Mortality Weekly Report (MMWR 2015 Jan. 16;64:10-15). “Antiviral medications are an important adjunct in the treatment and control of influenza for the 2014-2015 season and should be used as recommended, regardless of patient vaccination status.”
Targeted groups for antiviral treatment include any patient with suspected or confirmed influenza who is hospitalized, has severe or progressive illness, or is at high risk for complications from influenza, even if the illness seems mild. “Persons at high risk include young children (especially those younger than age 2), pregnant women, persons with chronic medical conditions like asthma, diabetes, or heart disease, and adults aged 65 years and older,” the researchers wrote. “Ideally, antiviral treatment should be initiated within 48 hours of symptom onset, when treatment is most effective.”
Though spot shortages of Tamiflu and other antiviral drugs have been reported in the United States, the CDC investigators noted that it may be necessary for physicians and patients to contact more than one pharmacy to fill a prescription. Updates on the supply of antiviral drugs can be found here.
Dr. Flannery and his associates acknowledged certain limitations of their analysis, including the fact that future estimates could differ as more data become available and that the current estimates “are limited to the prevention of outpatient medical visits, rather than more severe illness outcomes, such as hospitalization or death.”
The researchers reported having no relevant financial conflicts.
On Twitter @dougbrunk
So far this flu season, more than two-thirds of influenza A (H3N2) viruses differ from the components of the 2014-2015 influenza vaccine. In addition, the overall estimated effectiveness of the 2014-2015 influenza vaccine for preventing laboratory-confirmed influenza infection is only 23%.
Those are key findings from an analysis of 2,321 children and adults who presented to one of five study sites in the United States with acute respiratory illness between Nov. 10, 2014, and Jan. 2, 2015.
“Although influenza vaccines are the best tool for prevention of influenza currently available, more effective vaccines are needed,” wrote Brendan Flannery, Ph.D., of the influenza division at the CDC’s National Center for Immunization and Respiratory Disease, and his associates. The report is in the Jan. 16, 2015, issue of the CDC’s Morbidity and Mortality Weekly Report (MMWR 2015 Jan. 16;64:10-15). “Antiviral medications are an important adjunct in the treatment and control of influenza for the 2014-2015 season and should be used as recommended, regardless of patient vaccination status.”
Targeted groups for antiviral treatment include any patient with suspected or confirmed influenza who is hospitalized, has severe or progressive illness, or is at high risk for complications from influenza, even if the illness seems mild. “Persons at high risk include young children (especially those younger than age 2), pregnant women, persons with chronic medical conditions like asthma, diabetes, or heart disease, and adults aged 65 years and older,” the researchers wrote. “Ideally, antiviral treatment should be initiated within 48 hours of symptom onset, when treatment is most effective.”
Though spot shortages of Tamiflu and other antiviral drugs have been reported in the United States, the CDC investigators noted that it may be necessary for physicians and patients to contact more than one pharmacy to fill a prescription. Updates on the supply of antiviral drugs can be found here.
Dr. Flannery and his associates acknowledged certain limitations of their analysis, including the fact that future estimates could differ as more data become available and that the current estimates “are limited to the prevention of outpatient medical visits, rather than more severe illness outcomes, such as hospitalization or death.”
The researchers reported having no relevant financial conflicts.
On Twitter @dougbrunk
So far this flu season, more than two-thirds of influenza A (H3N2) viruses differ from the components of the 2014-2015 influenza vaccine. In addition, the overall estimated effectiveness of the 2014-2015 influenza vaccine for preventing laboratory-confirmed influenza infection is only 23%.
Those are key findings from an analysis of 2,321 children and adults who presented to one of five study sites in the United States with acute respiratory illness between Nov. 10, 2014, and Jan. 2, 2015.
“Although influenza vaccines are the best tool for prevention of influenza currently available, more effective vaccines are needed,” wrote Brendan Flannery, Ph.D., of the influenza division at the CDC’s National Center for Immunization and Respiratory Disease, and his associates. The report is in the Jan. 16, 2015, issue of the CDC’s Morbidity and Mortality Weekly Report (MMWR 2015 Jan. 16;64:10-15). “Antiviral medications are an important adjunct in the treatment and control of influenza for the 2014-2015 season and should be used as recommended, regardless of patient vaccination status.”
Targeted groups for antiviral treatment include any patient with suspected or confirmed influenza who is hospitalized, has severe or progressive illness, or is at high risk for complications from influenza, even if the illness seems mild. “Persons at high risk include young children (especially those younger than age 2), pregnant women, persons with chronic medical conditions like asthma, diabetes, or heart disease, and adults aged 65 years and older,” the researchers wrote. “Ideally, antiviral treatment should be initiated within 48 hours of symptom onset, when treatment is most effective.”
Though spot shortages of Tamiflu and other antiviral drugs have been reported in the United States, the CDC investigators noted that it may be necessary for physicians and patients to contact more than one pharmacy to fill a prescription. Updates on the supply of antiviral drugs can be found here.
Dr. Flannery and his associates acknowledged certain limitations of their analysis, including the fact that future estimates could differ as more data become available and that the current estimates “are limited to the prevention of outpatient medical visits, rather than more severe illness outcomes, such as hospitalization or death.”
The researchers reported having no relevant financial conflicts.
On Twitter @dougbrunk
FROM MORBIDITY AND MORTALITY WEEKLY REPORT
CDC: Flu Remains Widespread; Antivirals Underutilized
The approximate midpoint of the 2014-2015 flu season has been reached, disease remains widespread, and antiviral flu medications remain underutilized, according to the Centers for Disease Control and Prevention.
As predicted, this season is proving particularly severe due to the predominance of antigenically drifted H3N2 virus strains, and the CDC is urging clinicians to maintain a high index of suspicion for flu and to prescribe antivirals earlier and more aggressively for patients presenting with flulike illness – especially young children, those over age 65 years, and those with underlying conditions, even if their symptoms are mild, CDC Director Thomas R. Frieden said during a press briefing.
A health advisory on the topic was issued Jan. 9, he noted.
“The CDC has recommended the use of antiviral drugs as an adjunct to vaccination. They are the only medicines that can specifically treat influenza illness, and in the context of an H3N2 predominant season with a less effective vaccine, treatment with antiflu drugs is even more important than usual,” he said, referring to the two neuraminidase inhibitors currently approved for treating influenza: oseltamivir and zanamivir.
About two-thirds of the H3N2 viruses analyzed this season are different from the H3N2 virus included in this year’s vaccine, which is why the vaccine is expected to have reduced effectiveness, he explained.
This makes early antiviral treatment all the more important, he added, noting that treatment within 2 days of symptom onset is optimal, but later treatment can also offer benefit.
“CDC scientists have looked very carefully at the use of influenza drugs in the clinical setting, and the conclusion is clear: They work, but they aren’t being used nearly enough. They can reduce symptoms, shorten duration of illness, and prevent serious complications,” he said.
Continue for early treatments >>
Early treatment may help patients avoid hospitalization and could be life saving, he added.
Many patients are unaware that effective prescription treatments exist, and many doctors are not using the treatments as recommended. In one study, fewer than one in five eligible high-risk patients received treatment.
“So we’re expanding our efforts to reach clinicians about reminders about the importance of these drugs,” he said, noting that if high-risk patients with underlying conditions such as asthma, sickle cell disease, renal disease, or diabetes were treated, “tens of thousands of hospitalizations, and thousands of deaths could potentially be prevented.”
Dr. Frieden suggested that clinicians consider phone triage lines for high-risk patients to discuss symptoms over the phone and to “facilitate early initiation of treatment.”
An antiviral prescription can be provided without testing and before an office visit, he added.
The typical flu season lasts about 13 weeks on average, and the season began about 7 weeks ago. The continued high rate of H3N2 disease has had the greatest impact on older adults; that is typical of such seasons, which often involve a higher rate of hospitalizations and deaths.
In fact, hospitalization rates in the over 65 age group are rising sharply, Dr. Frieden said.
Last week the rate was 52 per 100,000 population, and this week the rate was 92 per 100,000 population. The cumulative rate 2 years ago when H3N2 viruses last predominated was 183 per 100,000, he noted.
“We wouldn’t be surprised to see something very similar this year,” he said.
Young children are also severely affected; thus far in the season there have been 26 reported pediatric flu-related deaths.
However, there are early signs that infection rates are declining in areas where the season started earlier, such as the Southeast, and the number of patients presenting to a doctor with flulike symptoms has also declined slightly, but it is too soon to tell whether disease activity has peaked, he said, stressing that “we still have several weeks of flu activity ahead.”
It’s not too late to get vaccinated, and despite the reduced efficacy of this year’s vaccine against the circulating H3N2 strains, vaccination may still offer some benefit.
“There are other strains out there as well,” he said, noting that a proportion of cases from other strains, such as influenza B, tend to occur late in the season, and this year’s vaccine offers a good match for influenza B viruses.
Pneumococcal vaccination is also important, particularly for older adults and those at high risk , he said.
Last September the CDC announced a new recommendation that all adults over age 65 years should get two different pneumococcal vaccines. Such vaccination may help prevent flu-related pneumonia.
Patients with the flu or flulike illness should be advised to cover their cough and to stay home from work or school to protect those who are most vulnerable to the flu, he said.
The approximate midpoint of the 2014-2015 flu season has been reached, disease remains widespread, and antiviral flu medications remain underutilized, according to the Centers for Disease Control and Prevention.
As predicted, this season is proving particularly severe due to the predominance of antigenically drifted H3N2 virus strains, and the CDC is urging clinicians to maintain a high index of suspicion for flu and to prescribe antivirals earlier and more aggressively for patients presenting with flulike illness – especially young children, those over age 65 years, and those with underlying conditions, even if their symptoms are mild, CDC Director Thomas R. Frieden said during a press briefing.
A health advisory on the topic was issued Jan. 9, he noted.
“The CDC has recommended the use of antiviral drugs as an adjunct to vaccination. They are the only medicines that can specifically treat influenza illness, and in the context of an H3N2 predominant season with a less effective vaccine, treatment with antiflu drugs is even more important than usual,” he said, referring to the two neuraminidase inhibitors currently approved for treating influenza: oseltamivir and zanamivir.
About two-thirds of the H3N2 viruses analyzed this season are different from the H3N2 virus included in this year’s vaccine, which is why the vaccine is expected to have reduced effectiveness, he explained.
This makes early antiviral treatment all the more important, he added, noting that treatment within 2 days of symptom onset is optimal, but later treatment can also offer benefit.
“CDC scientists have looked very carefully at the use of influenza drugs in the clinical setting, and the conclusion is clear: They work, but they aren’t being used nearly enough. They can reduce symptoms, shorten duration of illness, and prevent serious complications,” he said.
Continue for early treatments >>
Early treatment may help patients avoid hospitalization and could be life saving, he added.
Many patients are unaware that effective prescription treatments exist, and many doctors are not using the treatments as recommended. In one study, fewer than one in five eligible high-risk patients received treatment.
“So we’re expanding our efforts to reach clinicians about reminders about the importance of these drugs,” he said, noting that if high-risk patients with underlying conditions such as asthma, sickle cell disease, renal disease, or diabetes were treated, “tens of thousands of hospitalizations, and thousands of deaths could potentially be prevented.”
Dr. Frieden suggested that clinicians consider phone triage lines for high-risk patients to discuss symptoms over the phone and to “facilitate early initiation of treatment.”
An antiviral prescription can be provided without testing and before an office visit, he added.
The typical flu season lasts about 13 weeks on average, and the season began about 7 weeks ago. The continued high rate of H3N2 disease has had the greatest impact on older adults; that is typical of such seasons, which often involve a higher rate of hospitalizations and deaths.
In fact, hospitalization rates in the over 65 age group are rising sharply, Dr. Frieden said.
Last week the rate was 52 per 100,000 population, and this week the rate was 92 per 100,000 population. The cumulative rate 2 years ago when H3N2 viruses last predominated was 183 per 100,000, he noted.
“We wouldn’t be surprised to see something very similar this year,” he said.
Young children are also severely affected; thus far in the season there have been 26 reported pediatric flu-related deaths.
However, there are early signs that infection rates are declining in areas where the season started earlier, such as the Southeast, and the number of patients presenting to a doctor with flulike symptoms has also declined slightly, but it is too soon to tell whether disease activity has peaked, he said, stressing that “we still have several weeks of flu activity ahead.”
It’s not too late to get vaccinated, and despite the reduced efficacy of this year’s vaccine against the circulating H3N2 strains, vaccination may still offer some benefit.
“There are other strains out there as well,” he said, noting that a proportion of cases from other strains, such as influenza B, tend to occur late in the season, and this year’s vaccine offers a good match for influenza B viruses.
Pneumococcal vaccination is also important, particularly for older adults and those at high risk , he said.
Last September the CDC announced a new recommendation that all adults over age 65 years should get two different pneumococcal vaccines. Such vaccination may help prevent flu-related pneumonia.
Patients with the flu or flulike illness should be advised to cover their cough and to stay home from work or school to protect those who are most vulnerable to the flu, he said.
The approximate midpoint of the 2014-2015 flu season has been reached, disease remains widespread, and antiviral flu medications remain underutilized, according to the Centers for Disease Control and Prevention.
As predicted, this season is proving particularly severe due to the predominance of antigenically drifted H3N2 virus strains, and the CDC is urging clinicians to maintain a high index of suspicion for flu and to prescribe antivirals earlier and more aggressively for patients presenting with flulike illness – especially young children, those over age 65 years, and those with underlying conditions, even if their symptoms are mild, CDC Director Thomas R. Frieden said during a press briefing.
A health advisory on the topic was issued Jan. 9, he noted.
“The CDC has recommended the use of antiviral drugs as an adjunct to vaccination. They are the only medicines that can specifically treat influenza illness, and in the context of an H3N2 predominant season with a less effective vaccine, treatment with antiflu drugs is even more important than usual,” he said, referring to the two neuraminidase inhibitors currently approved for treating influenza: oseltamivir and zanamivir.
About two-thirds of the H3N2 viruses analyzed this season are different from the H3N2 virus included in this year’s vaccine, which is why the vaccine is expected to have reduced effectiveness, he explained.
This makes early antiviral treatment all the more important, he added, noting that treatment within 2 days of symptom onset is optimal, but later treatment can also offer benefit.
“CDC scientists have looked very carefully at the use of influenza drugs in the clinical setting, and the conclusion is clear: They work, but they aren’t being used nearly enough. They can reduce symptoms, shorten duration of illness, and prevent serious complications,” he said.
Continue for early treatments >>
Early treatment may help patients avoid hospitalization and could be life saving, he added.
Many patients are unaware that effective prescription treatments exist, and many doctors are not using the treatments as recommended. In one study, fewer than one in five eligible high-risk patients received treatment.
“So we’re expanding our efforts to reach clinicians about reminders about the importance of these drugs,” he said, noting that if high-risk patients with underlying conditions such as asthma, sickle cell disease, renal disease, or diabetes were treated, “tens of thousands of hospitalizations, and thousands of deaths could potentially be prevented.”
Dr. Frieden suggested that clinicians consider phone triage lines for high-risk patients to discuss symptoms over the phone and to “facilitate early initiation of treatment.”
An antiviral prescription can be provided without testing and before an office visit, he added.
The typical flu season lasts about 13 weeks on average, and the season began about 7 weeks ago. The continued high rate of H3N2 disease has had the greatest impact on older adults; that is typical of such seasons, which often involve a higher rate of hospitalizations and deaths.
In fact, hospitalization rates in the over 65 age group are rising sharply, Dr. Frieden said.
Last week the rate was 52 per 100,000 population, and this week the rate was 92 per 100,000 population. The cumulative rate 2 years ago when H3N2 viruses last predominated was 183 per 100,000, he noted.
“We wouldn’t be surprised to see something very similar this year,” he said.
Young children are also severely affected; thus far in the season there have been 26 reported pediatric flu-related deaths.
However, there are early signs that infection rates are declining in areas where the season started earlier, such as the Southeast, and the number of patients presenting to a doctor with flulike symptoms has also declined slightly, but it is too soon to tell whether disease activity has peaked, he said, stressing that “we still have several weeks of flu activity ahead.”
It’s not too late to get vaccinated, and despite the reduced efficacy of this year’s vaccine against the circulating H3N2 strains, vaccination may still offer some benefit.
“There are other strains out there as well,” he said, noting that a proportion of cases from other strains, such as influenza B, tend to occur late in the season, and this year’s vaccine offers a good match for influenza B viruses.
Pneumococcal vaccination is also important, particularly for older adults and those at high risk , he said.
Last September the CDC announced a new recommendation that all adults over age 65 years should get two different pneumococcal vaccines. Such vaccination may help prevent flu-related pneumonia.
Patients with the flu or flulike illness should be advised to cover their cough and to stay home from work or school to protect those who are most vulnerable to the flu, he said.
Asthma raises adults’ risk of sleep apnea
Adults who have asthma are at significantly greater risk than are those who don’t for developing obstructive sleep apnea, according to a report published Jan. 13 in JAMA.
“Accumulating evidence suggests a bidirectional relationship between asthma and obstructive sleep apnea, whereby each disorder deleteriously influences the other,” said Dr. Mihaela Teodorescu of the University of Wisconsin, Madison, and her colleagues.
To understand the “initiating processes of a potentially self-enforcing asthma-apnea cycle,” the investigators analyzed data from the Wisconsin Sleep Cohort Study, a population-based longitudinal epidemiologic investigation of the natural history of obstructive sleep apnea that began in 1988 and followed participants at 4-year intervals for up to 24 years. For their analysis, Dr. Teodorescu and her colleagues focused on a subset of 547 participants.
In their first 4-year follow-up intervals, 22 of 81 participants with preexisting asthma (27%) developed incident obstructive sleep apnea, compared with 75 of 466 participants who didn’t have asthma (16%). When all the interval observations were considered, there were 45 cases of obstructive sleep apnea among 167 possible intervals in participants who had preexisting asthma, for a rate of 27%, and 160 cases among 938 possible intervals in participants without asthma, for a rate of 17%. Compared with people who didn’t have asthma, those who did showed a relative risk of 1.39 for developing apnea, regardless of potentially confounding factors such as body mass index, the investigators said (JAMA 2015 Jan. 13 [doi:10.1001/jama.2014.17822]).
This association showed a dose-response pattern when cases were categorized according to the duration of asthma, with the highest risk for obstructive sleep apnea occurring among people who had asthma of 10 or more years’ duration. The association also remained robust in numerous additional analyses, including one involving only the subset of 220 participants who had spirometry results, another involving only participants who used asthma controller medications, and a third that accounted for participants’ neck girth, waist girth, and waist-to-hip ratio.
The mechanism(s) by which asthma may predispose patients to obstructive sleep apnea is not known, but could pertain to the disease itself, its treatment, or its common comorbidities. Nighttime asthma attacks could increase pressure in pharyngeal airway tissues, or pharyngeal airway stiffness could decrease because asthma causes abrupt drops in lung volumes during sleep, or a “spillover” of systemic inflammation could weaken the respiratory muscles or trigger CNS inflammatory responses. Corticosteroid therapy could alter the architecture of the pharyngeal airway by raising or redistributing pressures in the tissues there, or it could diminish the contractility of the dilator muscles. And coexisting gastroesophageal reflux could trigger pharyngeal spasms and induce mucosal exudative neurogenic inflammation, Dr. Teodorescu and her associates said.
Adults who have asthma are at significantly greater risk than are those who don’t for developing obstructive sleep apnea, according to a report published Jan. 13 in JAMA.
“Accumulating evidence suggests a bidirectional relationship between asthma and obstructive sleep apnea, whereby each disorder deleteriously influences the other,” said Dr. Mihaela Teodorescu of the University of Wisconsin, Madison, and her colleagues.
To understand the “initiating processes of a potentially self-enforcing asthma-apnea cycle,” the investigators analyzed data from the Wisconsin Sleep Cohort Study, a population-based longitudinal epidemiologic investigation of the natural history of obstructive sleep apnea that began in 1988 and followed participants at 4-year intervals for up to 24 years. For their analysis, Dr. Teodorescu and her colleagues focused on a subset of 547 participants.
In their first 4-year follow-up intervals, 22 of 81 participants with preexisting asthma (27%) developed incident obstructive sleep apnea, compared with 75 of 466 participants who didn’t have asthma (16%). When all the interval observations were considered, there were 45 cases of obstructive sleep apnea among 167 possible intervals in participants who had preexisting asthma, for a rate of 27%, and 160 cases among 938 possible intervals in participants without asthma, for a rate of 17%. Compared with people who didn’t have asthma, those who did showed a relative risk of 1.39 for developing apnea, regardless of potentially confounding factors such as body mass index, the investigators said (JAMA 2015 Jan. 13 [doi:10.1001/jama.2014.17822]).
This association showed a dose-response pattern when cases were categorized according to the duration of asthma, with the highest risk for obstructive sleep apnea occurring among people who had asthma of 10 or more years’ duration. The association also remained robust in numerous additional analyses, including one involving only the subset of 220 participants who had spirometry results, another involving only participants who used asthma controller medications, and a third that accounted for participants’ neck girth, waist girth, and waist-to-hip ratio.
The mechanism(s) by which asthma may predispose patients to obstructive sleep apnea is not known, but could pertain to the disease itself, its treatment, or its common comorbidities. Nighttime asthma attacks could increase pressure in pharyngeal airway tissues, or pharyngeal airway stiffness could decrease because asthma causes abrupt drops in lung volumes during sleep, or a “spillover” of systemic inflammation could weaken the respiratory muscles or trigger CNS inflammatory responses. Corticosteroid therapy could alter the architecture of the pharyngeal airway by raising or redistributing pressures in the tissues there, or it could diminish the contractility of the dilator muscles. And coexisting gastroesophageal reflux could trigger pharyngeal spasms and induce mucosal exudative neurogenic inflammation, Dr. Teodorescu and her associates said.
Adults who have asthma are at significantly greater risk than are those who don’t for developing obstructive sleep apnea, according to a report published Jan. 13 in JAMA.
“Accumulating evidence suggests a bidirectional relationship between asthma and obstructive sleep apnea, whereby each disorder deleteriously influences the other,” said Dr. Mihaela Teodorescu of the University of Wisconsin, Madison, and her colleagues.
To understand the “initiating processes of a potentially self-enforcing asthma-apnea cycle,” the investigators analyzed data from the Wisconsin Sleep Cohort Study, a population-based longitudinal epidemiologic investigation of the natural history of obstructive sleep apnea that began in 1988 and followed participants at 4-year intervals for up to 24 years. For their analysis, Dr. Teodorescu and her colleagues focused on a subset of 547 participants.
In their first 4-year follow-up intervals, 22 of 81 participants with preexisting asthma (27%) developed incident obstructive sleep apnea, compared with 75 of 466 participants who didn’t have asthma (16%). When all the interval observations were considered, there were 45 cases of obstructive sleep apnea among 167 possible intervals in participants who had preexisting asthma, for a rate of 27%, and 160 cases among 938 possible intervals in participants without asthma, for a rate of 17%. Compared with people who didn’t have asthma, those who did showed a relative risk of 1.39 for developing apnea, regardless of potentially confounding factors such as body mass index, the investigators said (JAMA 2015 Jan. 13 [doi:10.1001/jama.2014.17822]).
This association showed a dose-response pattern when cases were categorized according to the duration of asthma, with the highest risk for obstructive sleep apnea occurring among people who had asthma of 10 or more years’ duration. The association also remained robust in numerous additional analyses, including one involving only the subset of 220 participants who had spirometry results, another involving only participants who used asthma controller medications, and a third that accounted for participants’ neck girth, waist girth, and waist-to-hip ratio.
The mechanism(s) by which asthma may predispose patients to obstructive sleep apnea is not known, but could pertain to the disease itself, its treatment, or its common comorbidities. Nighttime asthma attacks could increase pressure in pharyngeal airway tissues, or pharyngeal airway stiffness could decrease because asthma causes abrupt drops in lung volumes during sleep, or a “spillover” of systemic inflammation could weaken the respiratory muscles or trigger CNS inflammatory responses. Corticosteroid therapy could alter the architecture of the pharyngeal airway by raising or redistributing pressures in the tissues there, or it could diminish the contractility of the dilator muscles. And coexisting gastroesophageal reflux could trigger pharyngeal spasms and induce mucosal exudative neurogenic inflammation, Dr. Teodorescu and her associates said.
Key clinical point: Adults with asthma are at higher risk for developing obstructive sleep apnea than are those without asthma.
Major finding: Twenty-two of 81 participants with preexisting asthma (27%) developed incident obstructive sleep apnea during initial 4-year follow-up, compared with 75 of 466 participants who didn’t have asthma (16%).
Data source: A secondary analysis of data in the Wisconsin Sleep Cohort Study (a prospective, longitudinal epidemiologic study) involving 547 randomly selected state employees aged 30-60 years at its inception in 1988 who were followed at 4-year intervals until 2013.
Disclosures: The National Heart, Lung, and Blood Institute, the National Institute on Aging, the National Center for Research Resources, and the William S. Middleton Memorial VA Hospital funded the study. Dr. Teodorescu and her colleagues reported having no conflicts of interest.