User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
Healthy diet could reduce COPD risk
People with a higher score on the Alternate Healthy Eating Index 2010 are at a lower risk of chronic obstructive pulmonary disease, according to Dr. Raphaëlle Varraso and associates.
For both men and women, those in the highest quintile based on AHEI-2010 score were at the lowest risk of COPD, compared with those in the lowest quintile. For women, the third-highest quintile had the highest risk of COPD with a hazard ratio of 1.01, slightly higher than the baseline of 1. For men, the baseline quintile had the highest risk of COPD, but the second-highest quintile had the next-highest hazard ratio at 0.9.
Although efforts to prevent COPD should continue to focus on smoking cessation, these prospective findings support the importance of a healthy diet in multi-interventional programs to prevent COPD, the researchers concluded.
Read the full article at the BMJ (doi:10.1136/bmj.h286).
People with a higher score on the Alternate Healthy Eating Index 2010 are at a lower risk of chronic obstructive pulmonary disease, according to Dr. Raphaëlle Varraso and associates.
For both men and women, those in the highest quintile based on AHEI-2010 score were at the lowest risk of COPD, compared with those in the lowest quintile. For women, the third-highest quintile had the highest risk of COPD with a hazard ratio of 1.01, slightly higher than the baseline of 1. For men, the baseline quintile had the highest risk of COPD, but the second-highest quintile had the next-highest hazard ratio at 0.9.
Although efforts to prevent COPD should continue to focus on smoking cessation, these prospective findings support the importance of a healthy diet in multi-interventional programs to prevent COPD, the researchers concluded.
Read the full article at the BMJ (doi:10.1136/bmj.h286).
People with a higher score on the Alternate Healthy Eating Index 2010 are at a lower risk of chronic obstructive pulmonary disease, according to Dr. Raphaëlle Varraso and associates.
For both men and women, those in the highest quintile based on AHEI-2010 score were at the lowest risk of COPD, compared with those in the lowest quintile. For women, the third-highest quintile had the highest risk of COPD with a hazard ratio of 1.01, slightly higher than the baseline of 1. For men, the baseline quintile had the highest risk of COPD, but the second-highest quintile had the next-highest hazard ratio at 0.9.
Although efforts to prevent COPD should continue to focus on smoking cessation, these prospective findings support the importance of a healthy diet in multi-interventional programs to prevent COPD, the researchers concluded.
Read the full article at the BMJ (doi:10.1136/bmj.h286).
Secondhand smoke remains problematic in the United States
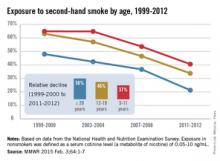
Exposure to and inhalation of secondhand smoke continue to be a significant problem throughout the United States, with cases of secondhand smoking–related illness and morbidity higher in juvenile and African American populations, among others, Dr. Thomas Frieden, director of the Centers for Disease Control and Prevention, said at a press briefing.
“Secondhand smoke is a Class A carcinogen [and] the Surgeon General has concluded that there is no risk-free level of exposure to secondhand smoke,” Dr. Frieden told reporters ahead of the release of this month’s CDC Vital Signs report (MMWR 2015 Feb.3;64:1-7). “People may not fully appreciate that secondhand smoke is not merely a nuisance – it causes disease and death.”
He explained that the Vital Signs report will highlight data from the National Health and Nutrition Examination Survey (NHANES), which was conducted between 1999 and 2012 to examine the health and nutrition of both children and adults across the United States. The study’s findings indicate that 40% of children, or two out of every five, are still exposed to secondhand smoke, and that even though cigarette smoking has decreased while smoke-free laws have increased in recent years, one in four nonsmokers and 58 million people overall are being exposed to secondhand smoke.
Furthermore, certain demographic groups are more likely to suffer exposure to secondhand smoke and, consequently, face several related health problems. In addition to 40.6% of children aged 3-11 years, nearly 70% of African American children in the same age range and 46.8% of all African Americans face a higher risk of secondhand smoke exposure. Mexican Americans and non-Hispanic whites also are highly predisposed to secondhand smoke exposure, with rates of 23.9% and 21.8%, respectively.
Americans living below the poverty line also face a 43% secondhand smoke exposure rate, while 37% of Americans in rental housing also are at risk. In fact, the home is the most significant source of exposure to secondhand smoke for children, although Dr. Frieden credited Americans with doubling the country’s number of smoke-free households over the last 20 years.
“Comprehensive smoke-free laws that prohibit smoking in all indoor areas of bars, restaurants, and workplaces are an important start,” stated Dr. Frieden. “As of today, 26 states and the District of Columbia, plus about 700 communities across the country, have adopted smoke-free laws that cover these three locations [and] we’ve seen tremendous increases in the number of college and university campuses that have gone smoke free.”
According to the CDC, exposure to secondhand smoke has been proven to cause afflictions such as ear infections, respiratory infections, asthma attacks, and sudden infant death syndrome in infants and children, plus stroke, coronary heart disease, heart attack, and lung cancer in nonsmoking adults. Secondhand smoke exposure causes 400 deaths in infants each year and more than 41,000 fatalities among nonsmoking adults per year. CDC figures also state that secondhand smoke exposure costs the United States $5.6 billion annually in lost productivity.
Exposure to and inhalation of secondhand smoke continue to be a significant problem throughout the United States, with cases of secondhand smoking–related illness and morbidity higher in juvenile and African American populations, among others, Dr. Thomas Frieden, director of the Centers for Disease Control and Prevention, said at a press briefing.
“Secondhand smoke is a Class A carcinogen [and] the Surgeon General has concluded that there is no risk-free level of exposure to secondhand smoke,” Dr. Frieden told reporters ahead of the release of this month’s CDC Vital Signs report (MMWR 2015 Feb.3;64:1-7). “People may not fully appreciate that secondhand smoke is not merely a nuisance – it causes disease and death.”
He explained that the Vital Signs report will highlight data from the National Health and Nutrition Examination Survey (NHANES), which was conducted between 1999 and 2012 to examine the health and nutrition of both children and adults across the United States. The study’s findings indicate that 40% of children, or two out of every five, are still exposed to secondhand smoke, and that even though cigarette smoking has decreased while smoke-free laws have increased in recent years, one in four nonsmokers and 58 million people overall are being exposed to secondhand smoke.
Furthermore, certain demographic groups are more likely to suffer exposure to secondhand smoke and, consequently, face several related health problems. In addition to 40.6% of children aged 3-11 years, nearly 70% of African American children in the same age range and 46.8% of all African Americans face a higher risk of secondhand smoke exposure. Mexican Americans and non-Hispanic whites also are highly predisposed to secondhand smoke exposure, with rates of 23.9% and 21.8%, respectively.
Americans living below the poverty line also face a 43% secondhand smoke exposure rate, while 37% of Americans in rental housing also are at risk. In fact, the home is the most significant source of exposure to secondhand smoke for children, although Dr. Frieden credited Americans with doubling the country’s number of smoke-free households over the last 20 years.
“Comprehensive smoke-free laws that prohibit smoking in all indoor areas of bars, restaurants, and workplaces are an important start,” stated Dr. Frieden. “As of today, 26 states and the District of Columbia, plus about 700 communities across the country, have adopted smoke-free laws that cover these three locations [and] we’ve seen tremendous increases in the number of college and university campuses that have gone smoke free.”
According to the CDC, exposure to secondhand smoke has been proven to cause afflictions such as ear infections, respiratory infections, asthma attacks, and sudden infant death syndrome in infants and children, plus stroke, coronary heart disease, heart attack, and lung cancer in nonsmoking adults. Secondhand smoke exposure causes 400 deaths in infants each year and more than 41,000 fatalities among nonsmoking adults per year. CDC figures also state that secondhand smoke exposure costs the United States $5.6 billion annually in lost productivity.
Exposure to and inhalation of secondhand smoke continue to be a significant problem throughout the United States, with cases of secondhand smoking–related illness and morbidity higher in juvenile and African American populations, among others, Dr. Thomas Frieden, director of the Centers for Disease Control and Prevention, said at a press briefing.
“Secondhand smoke is a Class A carcinogen [and] the Surgeon General has concluded that there is no risk-free level of exposure to secondhand smoke,” Dr. Frieden told reporters ahead of the release of this month’s CDC Vital Signs report (MMWR 2015 Feb.3;64:1-7). “People may not fully appreciate that secondhand smoke is not merely a nuisance – it causes disease and death.”
He explained that the Vital Signs report will highlight data from the National Health and Nutrition Examination Survey (NHANES), which was conducted between 1999 and 2012 to examine the health and nutrition of both children and adults across the United States. The study’s findings indicate that 40% of children, or two out of every five, are still exposed to secondhand smoke, and that even though cigarette smoking has decreased while smoke-free laws have increased in recent years, one in four nonsmokers and 58 million people overall are being exposed to secondhand smoke.
Furthermore, certain demographic groups are more likely to suffer exposure to secondhand smoke and, consequently, face several related health problems. In addition to 40.6% of children aged 3-11 years, nearly 70% of African American children in the same age range and 46.8% of all African Americans face a higher risk of secondhand smoke exposure. Mexican Americans and non-Hispanic whites also are highly predisposed to secondhand smoke exposure, with rates of 23.9% and 21.8%, respectively.
Americans living below the poverty line also face a 43% secondhand smoke exposure rate, while 37% of Americans in rental housing also are at risk. In fact, the home is the most significant source of exposure to secondhand smoke for children, although Dr. Frieden credited Americans with doubling the country’s number of smoke-free households over the last 20 years.
“Comprehensive smoke-free laws that prohibit smoking in all indoor areas of bars, restaurants, and workplaces are an important start,” stated Dr. Frieden. “As of today, 26 states and the District of Columbia, plus about 700 communities across the country, have adopted smoke-free laws that cover these three locations [and] we’ve seen tremendous increases in the number of college and university campuses that have gone smoke free.”
According to the CDC, exposure to secondhand smoke has been proven to cause afflictions such as ear infections, respiratory infections, asthma attacks, and sudden infant death syndrome in infants and children, plus stroke, coronary heart disease, heart attack, and lung cancer in nonsmoking adults. Secondhand smoke exposure causes 400 deaths in infants each year and more than 41,000 fatalities among nonsmoking adults per year. CDC figures also state that secondhand smoke exposure costs the United States $5.6 billion annually in lost productivity.
FROM A CDC TELEBRIEFING
Half of patients elect head and neck surgery before meeting surgeon
CORONADO, CALIF. – About half of patients decide to undergo head and neck surgery even before meeting their surgeon, and concerns about cost of the procedure weigh heavily on their minds, results from a pilot study demonstrated.
In an effort to determine which factors influence patient decision making about elective surgery in otolaryngology, lead study author Dr. Maya G. Sardesai and her associates surveyed 48 consecutive adults who underwent head and neck surgery performed by one of six surgeons at Harborview Medical Center, Seattle, between March and September 2014.
The effort “rose from an observation in her clinical practice that, despite similar degrees of disease burden and similar counseling, patients sometimes show widely divergent degrees of enthusiasm for elective procedures,” Dr. Sardesai of the department of otolaryngology-head and neck surgery at the medical center said at the Triological Society’s Combined Sections Meeting. “This prompted the question: What information influences decision making in this setting?”
Current guidelines emphasize discussing the risks and benefits of surgery in the informed consent process, she continued, “but some studies of decision making in this setting have suggested that other factors might also influence decisions, such as family advice, social perception, and cost. There’s limited data in the otolaryngology literature about this, even though there’s a preponderance of quality-of-life surgery with low but potentially significant risks.”
With input from patients and surgeons, the researchers created a 35-question survey and administered it in the surgeon’s office, with questions that centered around the timing of the procedure, advice of others, sources of information, and their approach to decision making. More than half of patients (56%) were undergoing tonsillectomy, followed by a nasal procedure (48%), palate procedure (44%), midline glossectomy (35%), hyoid suspension (4%), genioglossus advancement (4%), laryngeal procedure (2%), and other (6%). (The numbers exceeded 100% because some patients underwent more than one procedure.)
Nearly half of subjects (49%) reported making their decision to pursue surgery even before their surgical consultation or meeting their surgeon. The researchers then divided the cohort into patients who had decided to pursue surgery before or after meeting their surgeon. Among those who made the decision before meeting the surgeon, 64% rated information they received from their primary care provider as very important, while 100% rated information they received from the surgeon as very important. These percentages were similar among patients who made the decision after meeting the surgeon (43% and 96%, respectively).
Patients who made their decision to pursue surgery after meeting their surgeon also were more likely to weigh information received from the Internet as more important, compared with patients who made their decision before meeting their surgeon (38% vs. 20%). “This difference was not statistically significant,” Dr. Sardesai said at the meeting, which was jointly sponsored by the Triological Society and the American College of Surgeons. “All patients felt that Internet information seemed important.”
Patients in both groups weighed concerns about symptoms as very important (in the range of 83%), which rated “highly if not more than concerns about the risks with or without surgery (70%).” Finally, she and her associates found that 49% of patients in both groups considered the cost of medical bills as very important, “which is an interesting finding, because our current consent process doesn’t include much discussion about monetary costs of treatment.”
Overall, the findings suggest that otolaryngologists and head and neck surgeons should reach out to referring providers “to ensure that they are well informed about the indications, benefits, limitation, and risks of head and neck surgeries,” Dr. Sardesai concluded. “This may also enhance opportunities for shared and collaborative decision making. If decisions are being made prior to consultation, it begs the question about whether there are potential surgery candidates who defer surgical consultation altogether, and thus may be missing opportunities for better care. As otolaryngologists, we should also take an active role in providing and curating information from the Internet, since this is currently likely an increasingly important source of information for patients.”
She acknowledged certain limitations of the study, including its small sample size and the potential for recall bias. In addition, the survey “was administered in a surgeon’s office, which might bias patients to overemphasize the role of the surgeon,” she said. “Our future plans are to administer an enhanced version of the survey to broader [practice settings] to better understand these differences.”
Dr. Sardesai reported having no relevant financial conflicts.
On Twitter @dougbrunk
CORONADO, CALIF. – About half of patients decide to undergo head and neck surgery even before meeting their surgeon, and concerns about cost of the procedure weigh heavily on their minds, results from a pilot study demonstrated.
In an effort to determine which factors influence patient decision making about elective surgery in otolaryngology, lead study author Dr. Maya G. Sardesai and her associates surveyed 48 consecutive adults who underwent head and neck surgery performed by one of six surgeons at Harborview Medical Center, Seattle, between March and September 2014.
The effort “rose from an observation in her clinical practice that, despite similar degrees of disease burden and similar counseling, patients sometimes show widely divergent degrees of enthusiasm for elective procedures,” Dr. Sardesai of the department of otolaryngology-head and neck surgery at the medical center said at the Triological Society’s Combined Sections Meeting. “This prompted the question: What information influences decision making in this setting?”
Current guidelines emphasize discussing the risks and benefits of surgery in the informed consent process, she continued, “but some studies of decision making in this setting have suggested that other factors might also influence decisions, such as family advice, social perception, and cost. There’s limited data in the otolaryngology literature about this, even though there’s a preponderance of quality-of-life surgery with low but potentially significant risks.”
With input from patients and surgeons, the researchers created a 35-question survey and administered it in the surgeon’s office, with questions that centered around the timing of the procedure, advice of others, sources of information, and their approach to decision making. More than half of patients (56%) were undergoing tonsillectomy, followed by a nasal procedure (48%), palate procedure (44%), midline glossectomy (35%), hyoid suspension (4%), genioglossus advancement (4%), laryngeal procedure (2%), and other (6%). (The numbers exceeded 100% because some patients underwent more than one procedure.)
Nearly half of subjects (49%) reported making their decision to pursue surgery even before their surgical consultation or meeting their surgeon. The researchers then divided the cohort into patients who had decided to pursue surgery before or after meeting their surgeon. Among those who made the decision before meeting the surgeon, 64% rated information they received from their primary care provider as very important, while 100% rated information they received from the surgeon as very important. These percentages were similar among patients who made the decision after meeting the surgeon (43% and 96%, respectively).
Patients who made their decision to pursue surgery after meeting their surgeon also were more likely to weigh information received from the Internet as more important, compared with patients who made their decision before meeting their surgeon (38% vs. 20%). “This difference was not statistically significant,” Dr. Sardesai said at the meeting, which was jointly sponsored by the Triological Society and the American College of Surgeons. “All patients felt that Internet information seemed important.”
Patients in both groups weighed concerns about symptoms as very important (in the range of 83%), which rated “highly if not more than concerns about the risks with or without surgery (70%).” Finally, she and her associates found that 49% of patients in both groups considered the cost of medical bills as very important, “which is an interesting finding, because our current consent process doesn’t include much discussion about monetary costs of treatment.”
Overall, the findings suggest that otolaryngologists and head and neck surgeons should reach out to referring providers “to ensure that they are well informed about the indications, benefits, limitation, and risks of head and neck surgeries,” Dr. Sardesai concluded. “This may also enhance opportunities for shared and collaborative decision making. If decisions are being made prior to consultation, it begs the question about whether there are potential surgery candidates who defer surgical consultation altogether, and thus may be missing opportunities for better care. As otolaryngologists, we should also take an active role in providing and curating information from the Internet, since this is currently likely an increasingly important source of information for patients.”
She acknowledged certain limitations of the study, including its small sample size and the potential for recall bias. In addition, the survey “was administered in a surgeon’s office, which might bias patients to overemphasize the role of the surgeon,” she said. “Our future plans are to administer an enhanced version of the survey to broader [practice settings] to better understand these differences.”
Dr. Sardesai reported having no relevant financial conflicts.
On Twitter @dougbrunk
CORONADO, CALIF. – About half of patients decide to undergo head and neck surgery even before meeting their surgeon, and concerns about cost of the procedure weigh heavily on their minds, results from a pilot study demonstrated.
In an effort to determine which factors influence patient decision making about elective surgery in otolaryngology, lead study author Dr. Maya G. Sardesai and her associates surveyed 48 consecutive adults who underwent head and neck surgery performed by one of six surgeons at Harborview Medical Center, Seattle, between March and September 2014.
The effort “rose from an observation in her clinical practice that, despite similar degrees of disease burden and similar counseling, patients sometimes show widely divergent degrees of enthusiasm for elective procedures,” Dr. Sardesai of the department of otolaryngology-head and neck surgery at the medical center said at the Triological Society’s Combined Sections Meeting. “This prompted the question: What information influences decision making in this setting?”
Current guidelines emphasize discussing the risks and benefits of surgery in the informed consent process, she continued, “but some studies of decision making in this setting have suggested that other factors might also influence decisions, such as family advice, social perception, and cost. There’s limited data in the otolaryngology literature about this, even though there’s a preponderance of quality-of-life surgery with low but potentially significant risks.”
With input from patients and surgeons, the researchers created a 35-question survey and administered it in the surgeon’s office, with questions that centered around the timing of the procedure, advice of others, sources of information, and their approach to decision making. More than half of patients (56%) were undergoing tonsillectomy, followed by a nasal procedure (48%), palate procedure (44%), midline glossectomy (35%), hyoid suspension (4%), genioglossus advancement (4%), laryngeal procedure (2%), and other (6%). (The numbers exceeded 100% because some patients underwent more than one procedure.)
Nearly half of subjects (49%) reported making their decision to pursue surgery even before their surgical consultation or meeting their surgeon. The researchers then divided the cohort into patients who had decided to pursue surgery before or after meeting their surgeon. Among those who made the decision before meeting the surgeon, 64% rated information they received from their primary care provider as very important, while 100% rated information they received from the surgeon as very important. These percentages were similar among patients who made the decision after meeting the surgeon (43% and 96%, respectively).
Patients who made their decision to pursue surgery after meeting their surgeon also were more likely to weigh information received from the Internet as more important, compared with patients who made their decision before meeting their surgeon (38% vs. 20%). “This difference was not statistically significant,” Dr. Sardesai said at the meeting, which was jointly sponsored by the Triological Society and the American College of Surgeons. “All patients felt that Internet information seemed important.”
Patients in both groups weighed concerns about symptoms as very important (in the range of 83%), which rated “highly if not more than concerns about the risks with or without surgery (70%).” Finally, she and her associates found that 49% of patients in both groups considered the cost of medical bills as very important, “which is an interesting finding, because our current consent process doesn’t include much discussion about monetary costs of treatment.”
Overall, the findings suggest that otolaryngologists and head and neck surgeons should reach out to referring providers “to ensure that they are well informed about the indications, benefits, limitation, and risks of head and neck surgeries,” Dr. Sardesai concluded. “This may also enhance opportunities for shared and collaborative decision making. If decisions are being made prior to consultation, it begs the question about whether there are potential surgery candidates who defer surgical consultation altogether, and thus may be missing opportunities for better care. As otolaryngologists, we should also take an active role in providing and curating information from the Internet, since this is currently likely an increasingly important source of information for patients.”
She acknowledged certain limitations of the study, including its small sample size and the potential for recall bias. In addition, the survey “was administered in a surgeon’s office, which might bias patients to overemphasize the role of the surgeon,” she said. “Our future plans are to administer an enhanced version of the survey to broader [practice settings] to better understand these differences.”
Dr. Sardesai reported having no relevant financial conflicts.
On Twitter @dougbrunk
AT THE COMBINED SECTIONS WINTER MEETING
Key clinical point: Otolaryngologists and head and neck surgeons should ensure that primary and referring providers are well educated about elective head and neck procedures.
Major finding: Nearly half of patients opted for elective head and neck surgery even before meeting their surgeon.
Data source: A survey of 48 consecutive adults who underwent elective head and neck surgery between March and September 2014.
Disclosures: Dr. Sardesai reported having no financial disclosures.
Teen screen and electronic use linked to less sleep
Teenagers’ use of electronic devices before bed and their high screen usage during the day both independently increase the likelihood of sleep deprivation, a study found.
“While the frequency of use differed between the various devices, the relation between different types of electronic devices and sleep remained significant,” Mari Hysing of Uni Research Health in Bergen, Norway, and her associates reported online. “This suggests that the established relationship between TV and sleep found in previous studies can be generalized to newer technology,” they wrote (BMJ Open 2015 Feb. 2 [doi:10.1136/bmjopen-2014-006748]).
The researchers gathered data from 9,846 teens in Hordaland County, Norway, aged 16-19 years, regarding their use of electronic devices during daytime and nighttime and regarding their sleep quantity and quality. They asked about six different device types: personal computer (PC), cellphone, MP3 player, tablet, video game console, and television.
They assessed whether the teens used any of these devices in their bedrooms during the hour before they went to sleep, how often they used them during the daytime, and for what reasons they used the devices. The sleep data included typical bedtimes, rise times, time in bed, and total sleep on weekends and weekdays.
Nearly all teens used at least one, and often more than one, electronic device in the hour before bed. More than 80% reported using a PC, more than half reported watching TV, and approximately 90% of girls and 80% of boys had used a cellphone.
Total daily daytime screen use averaged approximately 5.5 hours for girls and more than 6.5 hours for boys.
Teens who used an electronic device in the hour before bed, used screens more than 4 hours total a day, or used any individual electronic device at least 2 hours a day were more likely to have a sleep deficiency of at least 2 hours a day.
Those using a PC or a cellphone in the hour before bed were 52% and 48% more likely, respectively, to take more than 60 minutes to fall asleep. Before-bed PC or cellphone use increased the likelihood of at least 2 hours’ sleep deficit by 53% and 35%, respectively.
Teens were 2.7 times more likely to get less than 5 hours sleep with PC use before bed and 1.85 times more likely with cellphone use before bed. Getting less than 5 hours sleep was 3.6 times more likely with at least 4 hours of total daily screen time.
The research was funded by Uni Research Health and the Norwegian Directorate for Health and Social Affairs. The authors reported no relevant financial disclosures.
Teenagers’ use of electronic devices before bed and their high screen usage during the day both independently increase the likelihood of sleep deprivation, a study found.
“While the frequency of use differed between the various devices, the relation between different types of electronic devices and sleep remained significant,” Mari Hysing of Uni Research Health in Bergen, Norway, and her associates reported online. “This suggests that the established relationship between TV and sleep found in previous studies can be generalized to newer technology,” they wrote (BMJ Open 2015 Feb. 2 [doi:10.1136/bmjopen-2014-006748]).
The researchers gathered data from 9,846 teens in Hordaland County, Norway, aged 16-19 years, regarding their use of electronic devices during daytime and nighttime and regarding their sleep quantity and quality. They asked about six different device types: personal computer (PC), cellphone, MP3 player, tablet, video game console, and television.
They assessed whether the teens used any of these devices in their bedrooms during the hour before they went to sleep, how often they used them during the daytime, and for what reasons they used the devices. The sleep data included typical bedtimes, rise times, time in bed, and total sleep on weekends and weekdays.
Nearly all teens used at least one, and often more than one, electronic device in the hour before bed. More than 80% reported using a PC, more than half reported watching TV, and approximately 90% of girls and 80% of boys had used a cellphone.
Total daily daytime screen use averaged approximately 5.5 hours for girls and more than 6.5 hours for boys.
Teens who used an electronic device in the hour before bed, used screens more than 4 hours total a day, or used any individual electronic device at least 2 hours a day were more likely to have a sleep deficiency of at least 2 hours a day.
Those using a PC or a cellphone in the hour before bed were 52% and 48% more likely, respectively, to take more than 60 minutes to fall asleep. Before-bed PC or cellphone use increased the likelihood of at least 2 hours’ sleep deficit by 53% and 35%, respectively.
Teens were 2.7 times more likely to get less than 5 hours sleep with PC use before bed and 1.85 times more likely with cellphone use before bed. Getting less than 5 hours sleep was 3.6 times more likely with at least 4 hours of total daily screen time.
The research was funded by Uni Research Health and the Norwegian Directorate for Health and Social Affairs. The authors reported no relevant financial disclosures.
Teenagers’ use of electronic devices before bed and their high screen usage during the day both independently increase the likelihood of sleep deprivation, a study found.
“While the frequency of use differed between the various devices, the relation between different types of electronic devices and sleep remained significant,” Mari Hysing of Uni Research Health in Bergen, Norway, and her associates reported online. “This suggests that the established relationship between TV and sleep found in previous studies can be generalized to newer technology,” they wrote (BMJ Open 2015 Feb. 2 [doi:10.1136/bmjopen-2014-006748]).
The researchers gathered data from 9,846 teens in Hordaland County, Norway, aged 16-19 years, regarding their use of electronic devices during daytime and nighttime and regarding their sleep quantity and quality. They asked about six different device types: personal computer (PC), cellphone, MP3 player, tablet, video game console, and television.
They assessed whether the teens used any of these devices in their bedrooms during the hour before they went to sleep, how often they used them during the daytime, and for what reasons they used the devices. The sleep data included typical bedtimes, rise times, time in bed, and total sleep on weekends and weekdays.
Nearly all teens used at least one, and often more than one, electronic device in the hour before bed. More than 80% reported using a PC, more than half reported watching TV, and approximately 90% of girls and 80% of boys had used a cellphone.
Total daily daytime screen use averaged approximately 5.5 hours for girls and more than 6.5 hours for boys.
Teens who used an electronic device in the hour before bed, used screens more than 4 hours total a day, or used any individual electronic device at least 2 hours a day were more likely to have a sleep deficiency of at least 2 hours a day.
Those using a PC or a cellphone in the hour before bed were 52% and 48% more likely, respectively, to take more than 60 minutes to fall asleep. Before-bed PC or cellphone use increased the likelihood of at least 2 hours’ sleep deficit by 53% and 35%, respectively.
Teens were 2.7 times more likely to get less than 5 hours sleep with PC use before bed and 1.85 times more likely with cellphone use before bed. Getting less than 5 hours sleep was 3.6 times more likely with at least 4 hours of total daily screen time.
The research was funded by Uni Research Health and the Norwegian Directorate for Health and Social Affairs. The authors reported no relevant financial disclosures.
FROM BMJ OPEN
Key clinical point: Teens who frequently use electronic devices before bedtime get less sleep.
Major finding: At least 4 hours of total daily screen time and personal computer or cellphone use in the hour before bed increased the risk of getting less than 5 hours sleep 3.6, 2.7, and 1.85 times, respectively.
Data source: The findings are based on a cross-sectional community-based study of 9,846 adolescents aged 16-19 years in Hordaland County, Norway in 2012.
Disclosures: The research was funded by Uni Research Health and the Norwegian Directorate for Health and Social Affairs. The authors reported no relevant financial disclosures.
Guideline clarifies first-line treatment for allergic rhinitis
First-line treatment for allergic rhinitis should include intranasal steroids, as well as less-sedating second-generation oral antihistamines for patients whose primary complaints are sneezing and itching, according to a new clinical practice guideline published online Feb. 2 in Otolaryngology–Head and Neck Surgery.
In contrast, sinonasal imaging should not be routine when patients first present with symptoms consistent with allergic rhinitis, and oral leukotriene receptor antagonists are not recommended as first-line therapy, said Dr. Michael D. Seidman of Henry Ford West Bloomfield (Mich.) Hospital and chair of the guideline working group, and his associates.
Dr. Seidman and a panel of 20 experts in otolaryngology, allergy and immunology, internal medicine, family medicine, pediatrics, sleep medicine, advanced practice nursing, complementary and alternative medicine, and consumer advocacy developed the new practice guideline to enable clinicians in all settings to improve patient care and reduce harmful or unnecessary variations in care for allergic rhinitis.
“The guideline is intended to focus on a limited number of quality improvement opportunities deemed most important by the working group and is not intended to be a comprehensive reference for diagnosing and managing allergic rhinitis,” the authors noted.
During the course of 1 year, the working group reviewed 1,605 randomized, controlled trials, 31 existing clinical practice guidelines, and 390 systematic reviews of the literature regarding allergic rhinitis in adults and children older than age 2 years. They then compiled 14 key recommendations that underwent extensive peer review, which have been published online and as a supplement to the February issue (Otolaryngol. Head Neck Surg. 2015;152:S1-S43).
In addition to the recommendations noted above, the guideline advises:
* Clinicians should diagnose allergic rhinitis when patients present with a history and physical exam consistent with the disorder (including clear rhinorrhea, nasal congestion, pale discoloration of the nasal mucosa, and red, watery eyes) plus symptoms of nasal congestion, runny nose, itchy nose, or sneezing.
* Clinicians should perform and interpret (or refer patients for) specific IgE allergy testing for allergic rhinitis that doesn’t respond to empiric treatment, or when the diagnosis is uncertain, or when identifying the specific causative allergen would allow targeted therapy.
* Clinicians should assess diagnosed patients for associated conditions such as asthma, atopic dermatitis, sleep-disordered breathing, conjunctivitis, rhinosinusitis, and otitis media, and should document that in the medical record.
* Clinicians should offer (or refer patients for) sublingual or subcutaneous immunotherapy when allergic rhinitis doesn’t respond adequately to pharmacologic therapy.
* Clinicians may advise avoidance of known allergens or controlling the patient’s environment by such measures as removing pets, using air filtration systems, using dust-mite–reducing covers for bedding, and using acaricides.
* Clinicians may offer intranasal antihistamines for patients with seasonal, perennial or episodic allergic rhinitis.*
* Clinicians may offer (or refer patients for) reduction of the inferior turbinates for patients who have nasal airway obstruction or enlarged turbinates.
* Clinicians may offer (or refer patient for) acupuncture if they are interested in nonpharmacologic therapy.
*Clinicians may offer combination pharmacologic therapy in patients with allergic rhinitis who have inadequate response to pharmacologic monotherapy.
The working group offered no recommendations concerning herbal therapy for allergic rhinitis, because of the limited literature on those substances and concern about their safety.
The full text of the guideline and its supporting data are available free of charge at www.entnet.org. In addition, an algorithm of the guideline’s action statements and a table of common allergic rhinitis clinical scenarios are available as quick reference guides for clinicians.
The American Academy of Otolaryngology–Head and Neck Surgery Foundation funded the guideline. Dr. Seidman reported being medical director of the Scientific Advisory Board of Visalus, founder of the Body Language Vitamin, and holder of six patents related to dietary supplements, aircraft, and middle ear and brain implants. His associates reported ties to Acclarent/Johnson/Johnson, FirstLine Medical, GlaxoSmithKline, Intersect, MEDA, Medtronic, Merck, Mylan, Novartis, TEVA, Transit of Venus, Sanofi, Sunovion Pharmaceuticals, and WellPoint.
*Correction, 2/18/2015: An earlier version of this story misstated the guideline for the use of intranasal antihistamines.
First-line treatment for allergic rhinitis should include intranasal steroids, as well as less-sedating second-generation oral antihistamines for patients whose primary complaints are sneezing and itching, according to a new clinical practice guideline published online Feb. 2 in Otolaryngology–Head and Neck Surgery.
In contrast, sinonasal imaging should not be routine when patients first present with symptoms consistent with allergic rhinitis, and oral leukotriene receptor antagonists are not recommended as first-line therapy, said Dr. Michael D. Seidman of Henry Ford West Bloomfield (Mich.) Hospital and chair of the guideline working group, and his associates.
Dr. Seidman and a panel of 20 experts in otolaryngology, allergy and immunology, internal medicine, family medicine, pediatrics, sleep medicine, advanced practice nursing, complementary and alternative medicine, and consumer advocacy developed the new practice guideline to enable clinicians in all settings to improve patient care and reduce harmful or unnecessary variations in care for allergic rhinitis.
“The guideline is intended to focus on a limited number of quality improvement opportunities deemed most important by the working group and is not intended to be a comprehensive reference for diagnosing and managing allergic rhinitis,” the authors noted.
During the course of 1 year, the working group reviewed 1,605 randomized, controlled trials, 31 existing clinical practice guidelines, and 390 systematic reviews of the literature regarding allergic rhinitis in adults and children older than age 2 years. They then compiled 14 key recommendations that underwent extensive peer review, which have been published online and as a supplement to the February issue (Otolaryngol. Head Neck Surg. 2015;152:S1-S43).
In addition to the recommendations noted above, the guideline advises:
* Clinicians should diagnose allergic rhinitis when patients present with a history and physical exam consistent with the disorder (including clear rhinorrhea, nasal congestion, pale discoloration of the nasal mucosa, and red, watery eyes) plus symptoms of nasal congestion, runny nose, itchy nose, or sneezing.
* Clinicians should perform and interpret (or refer patients for) specific IgE allergy testing for allergic rhinitis that doesn’t respond to empiric treatment, or when the diagnosis is uncertain, or when identifying the specific causative allergen would allow targeted therapy.
* Clinicians should assess diagnosed patients for associated conditions such as asthma, atopic dermatitis, sleep-disordered breathing, conjunctivitis, rhinosinusitis, and otitis media, and should document that in the medical record.
* Clinicians should offer (or refer patients for) sublingual or subcutaneous immunotherapy when allergic rhinitis doesn’t respond adequately to pharmacologic therapy.
* Clinicians may advise avoidance of known allergens or controlling the patient’s environment by such measures as removing pets, using air filtration systems, using dust-mite–reducing covers for bedding, and using acaricides.
* Clinicians may offer intranasal antihistamines for patients with seasonal, perennial or episodic allergic rhinitis.*
* Clinicians may offer (or refer patients for) reduction of the inferior turbinates for patients who have nasal airway obstruction or enlarged turbinates.
* Clinicians may offer (or refer patient for) acupuncture if they are interested in nonpharmacologic therapy.
*Clinicians may offer combination pharmacologic therapy in patients with allergic rhinitis who have inadequate response to pharmacologic monotherapy.
The working group offered no recommendations concerning herbal therapy for allergic rhinitis, because of the limited literature on those substances and concern about their safety.
The full text of the guideline and its supporting data are available free of charge at www.entnet.org. In addition, an algorithm of the guideline’s action statements and a table of common allergic rhinitis clinical scenarios are available as quick reference guides for clinicians.
The American Academy of Otolaryngology–Head and Neck Surgery Foundation funded the guideline. Dr. Seidman reported being medical director of the Scientific Advisory Board of Visalus, founder of the Body Language Vitamin, and holder of six patents related to dietary supplements, aircraft, and middle ear and brain implants. His associates reported ties to Acclarent/Johnson/Johnson, FirstLine Medical, GlaxoSmithKline, Intersect, MEDA, Medtronic, Merck, Mylan, Novartis, TEVA, Transit of Venus, Sanofi, Sunovion Pharmaceuticals, and WellPoint.
*Correction, 2/18/2015: An earlier version of this story misstated the guideline for the use of intranasal antihistamines.
First-line treatment for allergic rhinitis should include intranasal steroids, as well as less-sedating second-generation oral antihistamines for patients whose primary complaints are sneezing and itching, according to a new clinical practice guideline published online Feb. 2 in Otolaryngology–Head and Neck Surgery.
In contrast, sinonasal imaging should not be routine when patients first present with symptoms consistent with allergic rhinitis, and oral leukotriene receptor antagonists are not recommended as first-line therapy, said Dr. Michael D. Seidman of Henry Ford West Bloomfield (Mich.) Hospital and chair of the guideline working group, and his associates.
Dr. Seidman and a panel of 20 experts in otolaryngology, allergy and immunology, internal medicine, family medicine, pediatrics, sleep medicine, advanced practice nursing, complementary and alternative medicine, and consumer advocacy developed the new practice guideline to enable clinicians in all settings to improve patient care and reduce harmful or unnecessary variations in care for allergic rhinitis.
“The guideline is intended to focus on a limited number of quality improvement opportunities deemed most important by the working group and is not intended to be a comprehensive reference for diagnosing and managing allergic rhinitis,” the authors noted.
During the course of 1 year, the working group reviewed 1,605 randomized, controlled trials, 31 existing clinical practice guidelines, and 390 systematic reviews of the literature regarding allergic rhinitis in adults and children older than age 2 years. They then compiled 14 key recommendations that underwent extensive peer review, which have been published online and as a supplement to the February issue (Otolaryngol. Head Neck Surg. 2015;152:S1-S43).
In addition to the recommendations noted above, the guideline advises:
* Clinicians should diagnose allergic rhinitis when patients present with a history and physical exam consistent with the disorder (including clear rhinorrhea, nasal congestion, pale discoloration of the nasal mucosa, and red, watery eyes) plus symptoms of nasal congestion, runny nose, itchy nose, or sneezing.
* Clinicians should perform and interpret (or refer patients for) specific IgE allergy testing for allergic rhinitis that doesn’t respond to empiric treatment, or when the diagnosis is uncertain, or when identifying the specific causative allergen would allow targeted therapy.
* Clinicians should assess diagnosed patients for associated conditions such as asthma, atopic dermatitis, sleep-disordered breathing, conjunctivitis, rhinosinusitis, and otitis media, and should document that in the medical record.
* Clinicians should offer (or refer patients for) sublingual or subcutaneous immunotherapy when allergic rhinitis doesn’t respond adequately to pharmacologic therapy.
* Clinicians may advise avoidance of known allergens or controlling the patient’s environment by such measures as removing pets, using air filtration systems, using dust-mite–reducing covers for bedding, and using acaricides.
* Clinicians may offer intranasal antihistamines for patients with seasonal, perennial or episodic allergic rhinitis.*
* Clinicians may offer (or refer patients for) reduction of the inferior turbinates for patients who have nasal airway obstruction or enlarged turbinates.
* Clinicians may offer (or refer patient for) acupuncture if they are interested in nonpharmacologic therapy.
*Clinicians may offer combination pharmacologic therapy in patients with allergic rhinitis who have inadequate response to pharmacologic monotherapy.
The working group offered no recommendations concerning herbal therapy for allergic rhinitis, because of the limited literature on those substances and concern about their safety.
The full text of the guideline and its supporting data are available free of charge at www.entnet.org. In addition, an algorithm of the guideline’s action statements and a table of common allergic rhinitis clinical scenarios are available as quick reference guides for clinicians.
The American Academy of Otolaryngology–Head and Neck Surgery Foundation funded the guideline. Dr. Seidman reported being medical director of the Scientific Advisory Board of Visalus, founder of the Body Language Vitamin, and holder of six patents related to dietary supplements, aircraft, and middle ear and brain implants. His associates reported ties to Acclarent/Johnson/Johnson, FirstLine Medical, GlaxoSmithKline, Intersect, MEDA, Medtronic, Merck, Mylan, Novartis, TEVA, Transit of Venus, Sanofi, Sunovion Pharmaceuticals, and WellPoint.
*Correction, 2/18/2015: An earlier version of this story misstated the guideline for the use of intranasal antihistamines.
FROM OTOLARYNGOLOGY–HEAD AND NECK SURGERY
Key clinical point: First-line treatment for allergic rhinitis should include intranasal steroids and second-generation oral antihistamines, and should not include leukotriene receptor antagonists or sinonasal imaging studies.
Major finding: A panel of 20 experts took 1 year to review the literature and develop action items focusing on a limited number of quality improvement opportunities they deemed most important to improve patient care.
Data source: A review of 1,605 randomized, controlled trials, 31 sets of practice guidelines, and 390 systematic reviews regarding allergic rhinitis, and a compilation of 14 recommendations for managing the disorder.
Disclosures: The American Academy of Otolaryngology–Head and Neck Surgery Foundation funded the guideline. Dr. Seidman reported being medical director of the Scientific Advisory Board of Visalus, founder of the Body Language Vitamin, and holder of six patents related to dietary supplements, aircraft, and middle ear and brain implants. His associates reported ties to Acclarent/Johnson/Johnson, FirstLine Medical, GlaxoSmithKline, Intersect, MEDA, Medtronic, Merck, Mylan, Novartis, TEVA, Transit of Venus, Sanofi, Sunovion Pharmaceuticals, and WellPoint.
Detailed history, nonsedating antihistamines improve management of pediatric urticaria
ORLANDO – Acute urticaria in children is most often caused by infection, food, or medication, and a detailed history is imperative for improving the likelihood of identifying the culprit, according to Dr. Adam Friedman.
Mycoplasma is a particularly common infectious cause in children, but adenovirus, enterovirus, rotavirus, respiratory syncytial virus, Epstein-Barr virus, and cytomegalovirus also have been implicated in urticaria cases, Dr. Friedman, director of dermatologic research at the Albert Einstein College of Medicine, New York, said at the Orlando Dermatology Aesthetic and Clinical Conference.
With respect to foods, ask about intake of milk, eggs, peanuts, wheat, and soy. When it comes to prescription medications, antibiotics are an especially common cause.
“Nonsteroidal anti-inflammatory drugs are also a very important one. If you have a patient with a history of urticaria or a pediatric patient who has a history of urticaria of the mucosa, definitely educate the parents not to give them NSAIDs,” Dr. Friedman said.
Nonimmunologic direct mast cell activation also can be a source of acute urticaria, resulting from exposure to numerous products. These include, but are not limited to, polymyxin B, radiocontrast media, opiates, muscle relaxants, salicylates, and NSAIDs, all of which can “potentially induce urticaria in almost anyone,” he said.
Identifying the cause is more likely in acute case than in chronic cases, for which the etiology is unknown about 70% of time. Regardless, the good news is that about two-thirds of cases are self-limiting; it’s the other third that poses the greatest challenge, he said.
The best bet for nailing down a diagnosis and identifying the cause is an extensive history and physical evaluation.
“Ask a million and one questions to really get to the root of it. In some cases, chronic urticaria is really a diagnosis of exclusion,” he said.
Asking the patient to keep a diary to help identify any unusual exposures just prior to the episode, and having the patient or parent take pictures of the skin are shortcuts that can help.
Extensive laboratory testing is rarely helpful, but certain tests may be warranted. New European guidelines call for erythrocyte sedimentation rate/C-reactive protein and blood differential testing for chronic spontaneous urticaria, but others – like liver function tests, hepatitis B, antinuclear antibody, stool, urinalysis, thyroid function, and antithyroid antibodies – should be directed by the history. A complement panel may be useful in cases involving angioedema, and allergy skin testing may be warranted if a specific trigger can be implicated, but “don’t just order for the sake of ordering,” he said.
Biopsies are not typically useful except in suspected neutrophilic urticaria, which may indicate an association with autoimmune disease, as well as offer some insight into whether dapsone treatment would be helpful over other third-line therapies. Persistent cases of urticaria (lasting over 24-48 hours in one location) may suggest urticarial vasculitis, which would warrant a biopsy.
The therapeutic approach to urticaria involves educating patients about avoiding triggers and identifying and addressing underlying conditions, and using medications that address the pathophysiology of the disease (mast cells, histamine, etc).
In children, the guidelines are generally similar to those in adults, but there is a real push to avoid systemic steroids, Dr. Friedman said, noting that the only time he uses systemic steroids is as a bridge to get to the point where other therapies are beginning to take effect.
Other key concepts for managing urticaria in children, as published in 2013 (Acta. Derm. Venereol. 2013;93:500-8), include using second-generation histamine1 antihistamines for symptom relief, avoiding first-generation H1 antihistamines (due mainly to sedation), and using other therapeutic interventions only after carefully weighing risks and benefits, as evidence in children is lacking.
Nonsedating antihistamines are preferable, as sending kids to school on sedating medications can impact learning as well as social interaction, ultimately resulting in developmental delay.
Keep in mind that standard doses of such medications often are inadequate, and it is acceptable to work up to four times the dose, even in children, he said.
Triple-drug therapy, including H1 and H2 antagonists plus leukotriene blockers may be necessary.
“This is a very complicated and still important disease. The key is history – sometimes – and climbing the therapeutic ladder. These patients are really uncomfortable, and they will love you if you get this disease under control,” Dr. Friedman said.
One thing that might provide some comfort to the patients and their parents is the fact that urticaria in children does seem to have a point of remission. Parents often fear that their children will be plagued with urticaria for life, but a recent study of 92 patients showed that the remission rates at 1, 3, and 5 years were 18.5%, 54%, and 67.7% (J. Am. Acad. Dermatol. 2014;71:663-8).
The median duration of chronic urticaria was 4.3 years.
“That doesn’t sound great, but in considering one’s entire lifetime, it keeps things in perspective,” he said.
Dr. Friedman reported having no relevant disclosures.
ORLANDO – Acute urticaria in children is most often caused by infection, food, or medication, and a detailed history is imperative for improving the likelihood of identifying the culprit, according to Dr. Adam Friedman.
Mycoplasma is a particularly common infectious cause in children, but adenovirus, enterovirus, rotavirus, respiratory syncytial virus, Epstein-Barr virus, and cytomegalovirus also have been implicated in urticaria cases, Dr. Friedman, director of dermatologic research at the Albert Einstein College of Medicine, New York, said at the Orlando Dermatology Aesthetic and Clinical Conference.
With respect to foods, ask about intake of milk, eggs, peanuts, wheat, and soy. When it comes to prescription medications, antibiotics are an especially common cause.
“Nonsteroidal anti-inflammatory drugs are also a very important one. If you have a patient with a history of urticaria or a pediatric patient who has a history of urticaria of the mucosa, definitely educate the parents not to give them NSAIDs,” Dr. Friedman said.
Nonimmunologic direct mast cell activation also can be a source of acute urticaria, resulting from exposure to numerous products. These include, but are not limited to, polymyxin B, radiocontrast media, opiates, muscle relaxants, salicylates, and NSAIDs, all of which can “potentially induce urticaria in almost anyone,” he said.
Identifying the cause is more likely in acute case than in chronic cases, for which the etiology is unknown about 70% of time. Regardless, the good news is that about two-thirds of cases are self-limiting; it’s the other third that poses the greatest challenge, he said.
The best bet for nailing down a diagnosis and identifying the cause is an extensive history and physical evaluation.
“Ask a million and one questions to really get to the root of it. In some cases, chronic urticaria is really a diagnosis of exclusion,” he said.
Asking the patient to keep a diary to help identify any unusual exposures just prior to the episode, and having the patient or parent take pictures of the skin are shortcuts that can help.
Extensive laboratory testing is rarely helpful, but certain tests may be warranted. New European guidelines call for erythrocyte sedimentation rate/C-reactive protein and blood differential testing for chronic spontaneous urticaria, but others – like liver function tests, hepatitis B, antinuclear antibody, stool, urinalysis, thyroid function, and antithyroid antibodies – should be directed by the history. A complement panel may be useful in cases involving angioedema, and allergy skin testing may be warranted if a specific trigger can be implicated, but “don’t just order for the sake of ordering,” he said.
Biopsies are not typically useful except in suspected neutrophilic urticaria, which may indicate an association with autoimmune disease, as well as offer some insight into whether dapsone treatment would be helpful over other third-line therapies. Persistent cases of urticaria (lasting over 24-48 hours in one location) may suggest urticarial vasculitis, which would warrant a biopsy.
The therapeutic approach to urticaria involves educating patients about avoiding triggers and identifying and addressing underlying conditions, and using medications that address the pathophysiology of the disease (mast cells, histamine, etc).
In children, the guidelines are generally similar to those in adults, but there is a real push to avoid systemic steroids, Dr. Friedman said, noting that the only time he uses systemic steroids is as a bridge to get to the point where other therapies are beginning to take effect.
Other key concepts for managing urticaria in children, as published in 2013 (Acta. Derm. Venereol. 2013;93:500-8), include using second-generation histamine1 antihistamines for symptom relief, avoiding first-generation H1 antihistamines (due mainly to sedation), and using other therapeutic interventions only after carefully weighing risks and benefits, as evidence in children is lacking.
Nonsedating antihistamines are preferable, as sending kids to school on sedating medications can impact learning as well as social interaction, ultimately resulting in developmental delay.
Keep in mind that standard doses of such medications often are inadequate, and it is acceptable to work up to four times the dose, even in children, he said.
Triple-drug therapy, including H1 and H2 antagonists plus leukotriene blockers may be necessary.
“This is a very complicated and still important disease. The key is history – sometimes – and climbing the therapeutic ladder. These patients are really uncomfortable, and they will love you if you get this disease under control,” Dr. Friedman said.
One thing that might provide some comfort to the patients and their parents is the fact that urticaria in children does seem to have a point of remission. Parents often fear that their children will be plagued with urticaria for life, but a recent study of 92 patients showed that the remission rates at 1, 3, and 5 years were 18.5%, 54%, and 67.7% (J. Am. Acad. Dermatol. 2014;71:663-8).
The median duration of chronic urticaria was 4.3 years.
“That doesn’t sound great, but in considering one’s entire lifetime, it keeps things in perspective,” he said.
Dr. Friedman reported having no relevant disclosures.
ORLANDO – Acute urticaria in children is most often caused by infection, food, or medication, and a detailed history is imperative for improving the likelihood of identifying the culprit, according to Dr. Adam Friedman.
Mycoplasma is a particularly common infectious cause in children, but adenovirus, enterovirus, rotavirus, respiratory syncytial virus, Epstein-Barr virus, and cytomegalovirus also have been implicated in urticaria cases, Dr. Friedman, director of dermatologic research at the Albert Einstein College of Medicine, New York, said at the Orlando Dermatology Aesthetic and Clinical Conference.
With respect to foods, ask about intake of milk, eggs, peanuts, wheat, and soy. When it comes to prescription medications, antibiotics are an especially common cause.
“Nonsteroidal anti-inflammatory drugs are also a very important one. If you have a patient with a history of urticaria or a pediatric patient who has a history of urticaria of the mucosa, definitely educate the parents not to give them NSAIDs,” Dr. Friedman said.
Nonimmunologic direct mast cell activation also can be a source of acute urticaria, resulting from exposure to numerous products. These include, but are not limited to, polymyxin B, radiocontrast media, opiates, muscle relaxants, salicylates, and NSAIDs, all of which can “potentially induce urticaria in almost anyone,” he said.
Identifying the cause is more likely in acute case than in chronic cases, for which the etiology is unknown about 70% of time. Regardless, the good news is that about two-thirds of cases are self-limiting; it’s the other third that poses the greatest challenge, he said.
The best bet for nailing down a diagnosis and identifying the cause is an extensive history and physical evaluation.
“Ask a million and one questions to really get to the root of it. In some cases, chronic urticaria is really a diagnosis of exclusion,” he said.
Asking the patient to keep a diary to help identify any unusual exposures just prior to the episode, and having the patient or parent take pictures of the skin are shortcuts that can help.
Extensive laboratory testing is rarely helpful, but certain tests may be warranted. New European guidelines call for erythrocyte sedimentation rate/C-reactive protein and blood differential testing for chronic spontaneous urticaria, but others – like liver function tests, hepatitis B, antinuclear antibody, stool, urinalysis, thyroid function, and antithyroid antibodies – should be directed by the history. A complement panel may be useful in cases involving angioedema, and allergy skin testing may be warranted if a specific trigger can be implicated, but “don’t just order for the sake of ordering,” he said.
Biopsies are not typically useful except in suspected neutrophilic urticaria, which may indicate an association with autoimmune disease, as well as offer some insight into whether dapsone treatment would be helpful over other third-line therapies. Persistent cases of urticaria (lasting over 24-48 hours in one location) may suggest urticarial vasculitis, which would warrant a biopsy.
The therapeutic approach to urticaria involves educating patients about avoiding triggers and identifying and addressing underlying conditions, and using medications that address the pathophysiology of the disease (mast cells, histamine, etc).
In children, the guidelines are generally similar to those in adults, but there is a real push to avoid systemic steroids, Dr. Friedman said, noting that the only time he uses systemic steroids is as a bridge to get to the point where other therapies are beginning to take effect.
Other key concepts for managing urticaria in children, as published in 2013 (Acta. Derm. Venereol. 2013;93:500-8), include using second-generation histamine1 antihistamines for symptom relief, avoiding first-generation H1 antihistamines (due mainly to sedation), and using other therapeutic interventions only after carefully weighing risks and benefits, as evidence in children is lacking.
Nonsedating antihistamines are preferable, as sending kids to school on sedating medications can impact learning as well as social interaction, ultimately resulting in developmental delay.
Keep in mind that standard doses of such medications often are inadequate, and it is acceptable to work up to four times the dose, even in children, he said.
Triple-drug therapy, including H1 and H2 antagonists plus leukotriene blockers may be necessary.
“This is a very complicated and still important disease. The key is history – sometimes – and climbing the therapeutic ladder. These patients are really uncomfortable, and they will love you if you get this disease under control,” Dr. Friedman said.
One thing that might provide some comfort to the patients and their parents is the fact that urticaria in children does seem to have a point of remission. Parents often fear that their children will be plagued with urticaria for life, but a recent study of 92 patients showed that the remission rates at 1, 3, and 5 years were 18.5%, 54%, and 67.7% (J. Am. Acad. Dermatol. 2014;71:663-8).
The median duration of chronic urticaria was 4.3 years.
“That doesn’t sound great, but in considering one’s entire lifetime, it keeps things in perspective,” he said.
Dr. Friedman reported having no relevant disclosures.
EXPERT ANALYSIS FROM THE ODAC CONFERENCE
Meta-analysis: Oseltamivir shortens time to flu relief
Oseltamivir treatment in adults with influenza shortens the time to clinical symptom alleviation by about 1 day and substantially reduces the risk of lower respiratory tract complications and hospitalization, according to findings from a meta-analysis of nine randomized, controlled trials involving 4,328 adult patients.
The findings of the study, which is the first to use individual patient data to evaluate the neuraminidase inhibitor, should put to rest persistent doubts its efficacy and safety, according to Dr. Arnold S. Monto of the University of Michigan School of Public Health, Ann Arbor, who was lead investigator, and his colleagues.
The intention-to-treat population of 1,591 patients with confirmed influenza had a significant 21% shorter time to clinical symptom alleviation, compared with the intention-to-treat infected population of 1,302 patients who received placebo (97.5 hours vs. 122.7 hours, difference of 25.2 hours; time ratio, 0.79), the investigators reported online Jan. 30 in the Lancet.
The effects were somewhat attenuated in the 2,402 treated patients in the overall intention-to-treat population – with a 15% reduction in time to symptom alleviation – compared with the 1,926 placebo patients in that population. But the difference remained significant (median of 17.8 hours; time ratio, 0.85), the investigators said (Lancet 2015 Jan. 30 [doi:10.106/S0140-6736(14)62449-1]).
“In the intention-to-treat noninfected population, the estimated time ratio was close to unity (time ratio, 0.99), so only participants identified as influenza infected benefited from oseltamivir,” the study authors wrote. That suggests efficacy is “confined to the antiviral activity of the drug.”
In addition, treatment significantly reduced the risk of lower respiratory tract complications occurring more than 48 hours after randomization by 44% in the intention-to-treat infected population. Only 4.2% of oseltamivir-treated patients requiring such treatment, compared with 8.7% of those who received placebo (risk ratio, 0.56). The risk of hospitalization for any cause was significantly reduced by 63% (0.6% vs. 1.7% in the groups, respectively; RR, 0.37).
The risk ratios for lower respiratory tract complications and hospitalization were 0.62 and 0.61, respectively, in the overall intention-to-treat population, but the difference in hospitalization between the groups was no longer statistically significant.
The benefits of treatment came at the cost of increased risk of nausea and vomiting (RR, 1.60 and 2.43, respectively), but no effect was seen with respect to neurologic or psychiatric disorders or serious adverse events in either of the groups.
To overcome previous concerns regarding potential publication bias, the investigators included all published and unpublished Roche-sponsored randomized, placebo-controlled, double-blind trials of the standard prescribed oseltamivir dose of 75 mg twice daily in adults, as well other applicable trials of the Roche drug for the treatment of naturally occurring influenzalike illness. The trials were conducted between 1997 and 2001, and included patients who were within 36 hours of feeling unwell, and who had a fever and at least one respiratory symptom and one additional constitutional symptom.
Treatment was administered at 12-hour intervals for 5 days, and patients were followed for 21 days, during which time virus cultures were performed in most cases.
“The safety and effectiveness of oseltamivir has been hotly debated, with some researchers claiming there is little evidence that oseltamivir works,” Dr. Monto said in a press statement. “Our meta-analysis provides compelling evidence that oseltamivir therapy reduces by 1 day the typical length of illness in adults infected with influenza and also prevents complications and reduces the number of people needing hospital treatment. Whether the magnitude of these benefits outweighs the harms of nausea and vomiting needs careful consideration.”
The Multiparty Group for Advice on Science funded the study and assembled a multidisciplinary team to examine available data. The group obtained an unrestricted grant from Roche to cover the costs.
Dr. Monto reported receiving fees from BioCryst and Roche outside of the submitted work. His coauthor, Dr. Richard J. Whitely, reported receiving fees as a board member of Gilead Sciences and travel funding from Roche. The remaining authors reported having no disclosures.
Oseltamivir treatment in adults with influenza shortens the time to clinical symptom alleviation by about 1 day and substantially reduces the risk of lower respiratory tract complications and hospitalization, according to findings from a meta-analysis of nine randomized, controlled trials involving 4,328 adult patients.
The findings of the study, which is the first to use individual patient data to evaluate the neuraminidase inhibitor, should put to rest persistent doubts its efficacy and safety, according to Dr. Arnold S. Monto of the University of Michigan School of Public Health, Ann Arbor, who was lead investigator, and his colleagues.
The intention-to-treat population of 1,591 patients with confirmed influenza had a significant 21% shorter time to clinical symptom alleviation, compared with the intention-to-treat infected population of 1,302 patients who received placebo (97.5 hours vs. 122.7 hours, difference of 25.2 hours; time ratio, 0.79), the investigators reported online Jan. 30 in the Lancet.
The effects were somewhat attenuated in the 2,402 treated patients in the overall intention-to-treat population – with a 15% reduction in time to symptom alleviation – compared with the 1,926 placebo patients in that population. But the difference remained significant (median of 17.8 hours; time ratio, 0.85), the investigators said (Lancet 2015 Jan. 30 [doi:10.106/S0140-6736(14)62449-1]).
“In the intention-to-treat noninfected population, the estimated time ratio was close to unity (time ratio, 0.99), so only participants identified as influenza infected benefited from oseltamivir,” the study authors wrote. That suggests efficacy is “confined to the antiviral activity of the drug.”
In addition, treatment significantly reduced the risk of lower respiratory tract complications occurring more than 48 hours after randomization by 44% in the intention-to-treat infected population. Only 4.2% of oseltamivir-treated patients requiring such treatment, compared with 8.7% of those who received placebo (risk ratio, 0.56). The risk of hospitalization for any cause was significantly reduced by 63% (0.6% vs. 1.7% in the groups, respectively; RR, 0.37).
The risk ratios for lower respiratory tract complications and hospitalization were 0.62 and 0.61, respectively, in the overall intention-to-treat population, but the difference in hospitalization between the groups was no longer statistically significant.
The benefits of treatment came at the cost of increased risk of nausea and vomiting (RR, 1.60 and 2.43, respectively), but no effect was seen with respect to neurologic or psychiatric disorders or serious adverse events in either of the groups.
To overcome previous concerns regarding potential publication bias, the investigators included all published and unpublished Roche-sponsored randomized, placebo-controlled, double-blind trials of the standard prescribed oseltamivir dose of 75 mg twice daily in adults, as well other applicable trials of the Roche drug for the treatment of naturally occurring influenzalike illness. The trials were conducted between 1997 and 2001, and included patients who were within 36 hours of feeling unwell, and who had a fever and at least one respiratory symptom and one additional constitutional symptom.
Treatment was administered at 12-hour intervals for 5 days, and patients were followed for 21 days, during which time virus cultures were performed in most cases.
“The safety and effectiveness of oseltamivir has been hotly debated, with some researchers claiming there is little evidence that oseltamivir works,” Dr. Monto said in a press statement. “Our meta-analysis provides compelling evidence that oseltamivir therapy reduces by 1 day the typical length of illness in adults infected with influenza and also prevents complications and reduces the number of people needing hospital treatment. Whether the magnitude of these benefits outweighs the harms of nausea and vomiting needs careful consideration.”
The Multiparty Group for Advice on Science funded the study and assembled a multidisciplinary team to examine available data. The group obtained an unrestricted grant from Roche to cover the costs.
Dr. Monto reported receiving fees from BioCryst and Roche outside of the submitted work. His coauthor, Dr. Richard J. Whitely, reported receiving fees as a board member of Gilead Sciences and travel funding from Roche. The remaining authors reported having no disclosures.
Oseltamivir treatment in adults with influenza shortens the time to clinical symptom alleviation by about 1 day and substantially reduces the risk of lower respiratory tract complications and hospitalization, according to findings from a meta-analysis of nine randomized, controlled trials involving 4,328 adult patients.
The findings of the study, which is the first to use individual patient data to evaluate the neuraminidase inhibitor, should put to rest persistent doubts its efficacy and safety, according to Dr. Arnold S. Monto of the University of Michigan School of Public Health, Ann Arbor, who was lead investigator, and his colleagues.
The intention-to-treat population of 1,591 patients with confirmed influenza had a significant 21% shorter time to clinical symptom alleviation, compared with the intention-to-treat infected population of 1,302 patients who received placebo (97.5 hours vs. 122.7 hours, difference of 25.2 hours; time ratio, 0.79), the investigators reported online Jan. 30 in the Lancet.
The effects were somewhat attenuated in the 2,402 treated patients in the overall intention-to-treat population – with a 15% reduction in time to symptom alleviation – compared with the 1,926 placebo patients in that population. But the difference remained significant (median of 17.8 hours; time ratio, 0.85), the investigators said (Lancet 2015 Jan. 30 [doi:10.106/S0140-6736(14)62449-1]).
“In the intention-to-treat noninfected population, the estimated time ratio was close to unity (time ratio, 0.99), so only participants identified as influenza infected benefited from oseltamivir,” the study authors wrote. That suggests efficacy is “confined to the antiviral activity of the drug.”
In addition, treatment significantly reduced the risk of lower respiratory tract complications occurring more than 48 hours after randomization by 44% in the intention-to-treat infected population. Only 4.2% of oseltamivir-treated patients requiring such treatment, compared with 8.7% of those who received placebo (risk ratio, 0.56). The risk of hospitalization for any cause was significantly reduced by 63% (0.6% vs. 1.7% in the groups, respectively; RR, 0.37).
The risk ratios for lower respiratory tract complications and hospitalization were 0.62 and 0.61, respectively, in the overall intention-to-treat population, but the difference in hospitalization between the groups was no longer statistically significant.
The benefits of treatment came at the cost of increased risk of nausea and vomiting (RR, 1.60 and 2.43, respectively), but no effect was seen with respect to neurologic or psychiatric disorders or serious adverse events in either of the groups.
To overcome previous concerns regarding potential publication bias, the investigators included all published and unpublished Roche-sponsored randomized, placebo-controlled, double-blind trials of the standard prescribed oseltamivir dose of 75 mg twice daily in adults, as well other applicable trials of the Roche drug for the treatment of naturally occurring influenzalike illness. The trials were conducted between 1997 and 2001, and included patients who were within 36 hours of feeling unwell, and who had a fever and at least one respiratory symptom and one additional constitutional symptom.
Treatment was administered at 12-hour intervals for 5 days, and patients were followed for 21 days, during which time virus cultures were performed in most cases.
“The safety and effectiveness of oseltamivir has been hotly debated, with some researchers claiming there is little evidence that oseltamivir works,” Dr. Monto said in a press statement. “Our meta-analysis provides compelling evidence that oseltamivir therapy reduces by 1 day the typical length of illness in adults infected with influenza and also prevents complications and reduces the number of people needing hospital treatment. Whether the magnitude of these benefits outweighs the harms of nausea and vomiting needs careful consideration.”
The Multiparty Group for Advice on Science funded the study and assembled a multidisciplinary team to examine available data. The group obtained an unrestricted grant from Roche to cover the costs.
Dr. Monto reported receiving fees from BioCryst and Roche outside of the submitted work. His coauthor, Dr. Richard J. Whitely, reported receiving fees as a board member of Gilead Sciences and travel funding from Roche. The remaining authors reported having no disclosures.
FROM THE LANCET
Key clinical point: Oseltamivir shortens the time to flu symptom alleviation, but nausea and vomiting are risks.
Major finding: The time to clinical symptom alleviation was about 25 hours shorter (21%) with oseltamivir vs. placebo.
Data source: An individual patient data meta-analysis encompassing nine studies involving 4,328 patients.
Disclosures: The Multiparty Group for Advice on Science funded the study and assembled a multidisciplinary team to examine available data. The group obtained an unrestricted grant from Roche to cover the costs. Dr. Monto reported receiving fees from Biocryst and Roche outside of the submitted work. His coauthor, Dr. Richard J. Whitely, reported receiving fees as a board member of Gilead Sciences and travel funding from Roche. The remaining authors reported having no disclosures.
U.S. measles cases reached postelimination high in 2014
In 2014, the number of measles cases in the United States reached its highest level since the disease was declared eliminated in 2000, the Centers for Disease Control and Prevention reported.
Provisional data show that 644 cases of measles from 27 states in 23 outbreaks were reported to the CDC’s National Center for Immunization and Respiratory Diseases in 2014. The previous postelimination high came in 2011, when 220 cases were reported. The CDC reported that most people that contracted measles were unvaccinated and that travelers with measles continue to bring the infectious disease to the United States.
Meanwhile, 2015 seems to have started off with a bang as California officials have confirmed 59 cases of measles in the state since the end of 2014. Of those cases, “42 have been linked to an initial exposure in December at Disneyland or Disney California Adventure Park in Anaheim,” the California Department of Public Health said in a statement. Measles cases with epidemiologic links to those in Disney include ones in Arizona, Colorado, Mexico, Nebraska, Oregon, Utah, and Washington.

In 2014, the number of measles cases in the United States reached its highest level since the disease was declared eliminated in 2000, the Centers for Disease Control and Prevention reported.
Provisional data show that 644 cases of measles from 27 states in 23 outbreaks were reported to the CDC’s National Center for Immunization and Respiratory Diseases in 2014. The previous postelimination high came in 2011, when 220 cases were reported. The CDC reported that most people that contracted measles were unvaccinated and that travelers with measles continue to bring the infectious disease to the United States.
Meanwhile, 2015 seems to have started off with a bang as California officials have confirmed 59 cases of measles in the state since the end of 2014. Of those cases, “42 have been linked to an initial exposure in December at Disneyland or Disney California Adventure Park in Anaheim,” the California Department of Public Health said in a statement. Measles cases with epidemiologic links to those in Disney include ones in Arizona, Colorado, Mexico, Nebraska, Oregon, Utah, and Washington.

In 2014, the number of measles cases in the United States reached its highest level since the disease was declared eliminated in 2000, the Centers for Disease Control and Prevention reported.
Provisional data show that 644 cases of measles from 27 states in 23 outbreaks were reported to the CDC’s National Center for Immunization and Respiratory Diseases in 2014. The previous postelimination high came in 2011, when 220 cases were reported. The CDC reported that most people that contracted measles were unvaccinated and that travelers with measles continue to bring the infectious disease to the United States.
Meanwhile, 2015 seems to have started off with a bang as California officials have confirmed 59 cases of measles in the state since the end of 2014. Of those cases, “42 have been linked to an initial exposure in December at Disneyland or Disney California Adventure Park in Anaheim,” the California Department of Public Health said in a statement. Measles cases with epidemiologic links to those in Disney include ones in Arizona, Colorado, Mexico, Nebraska, Oregon, Utah, and Washington.

Ibuprofen superior to morphine following pediatric tonsillectomy
Ibuprofen appears to be an effective and safer pain reliever than morphine in children undergoing tonsillectomies, according to a recent study.
Although the two medications, administered with acetaminophen, treated pain about equally, morphine showed a greater risk for oxygen desaturation the night after surgery.
“Given the unpredictable posttonsillectomy respiratory response to opioids (codeine, morphine, and hydrocodone) and the analgesic effectiveness of ibuprofen, perhaps the time has come to question the postoperative use of all opioids in this population,” wrote Lauren Kelly, Ph.D., of Western University in London, Ontario, and her associates. The study was published online Jan. 26 (Pediatrics 2014 Jan. 26 [doi: 10.1542/peds.2014-1906].
The researchers randomized 91 children aged 1-10 years to acetaminophen (10-15 mg/kg per dose every 4 hours) with either an age appropriate dose of 0.2-0.5 mg/kg oral morphine every 4 hours or 10 mg/kg of oral ibuprofen every 6 hours after the children underwent a tonsillectomy with or without an adenoidectomy to treat sleep disordered breathing. The study ran from September 2012 to January 2014.
Parents put pulse oximeters on their children the nights before and after surgery to monitor oxygen saturation and apnea events.
The first evening after the surgery, 68% of children receiving ibuprofen showed improvement in oxygen desaturations, compared with 14% of children receiving morphine. The children receiving ibuprofen experienced an average 1.79 fewer desaturation events per hour, compared to an average 11.17 more desaturation events per hour in the morphine group.
No differences in average postsurgical oxygen saturation, pain relieving effectiveness, tonsillar bleeding or drug adverse events were identified.
“The results of this study support effective posttonsillectomy analgesia in children by using ibuprofen in combination with acetaminophen,” Dr. Kelly and her team wrote.
Ibuprofen appears to be an effective and safer pain reliever than morphine in children undergoing tonsillectomies, according to a recent study.
Although the two medications, administered with acetaminophen, treated pain about equally, morphine showed a greater risk for oxygen desaturation the night after surgery.
“Given the unpredictable posttonsillectomy respiratory response to opioids (codeine, morphine, and hydrocodone) and the analgesic effectiveness of ibuprofen, perhaps the time has come to question the postoperative use of all opioids in this population,” wrote Lauren Kelly, Ph.D., of Western University in London, Ontario, and her associates. The study was published online Jan. 26 (Pediatrics 2014 Jan. 26 [doi: 10.1542/peds.2014-1906].
The researchers randomized 91 children aged 1-10 years to acetaminophen (10-15 mg/kg per dose every 4 hours) with either an age appropriate dose of 0.2-0.5 mg/kg oral morphine every 4 hours or 10 mg/kg of oral ibuprofen every 6 hours after the children underwent a tonsillectomy with or without an adenoidectomy to treat sleep disordered breathing. The study ran from September 2012 to January 2014.
Parents put pulse oximeters on their children the nights before and after surgery to monitor oxygen saturation and apnea events.
The first evening after the surgery, 68% of children receiving ibuprofen showed improvement in oxygen desaturations, compared with 14% of children receiving morphine. The children receiving ibuprofen experienced an average 1.79 fewer desaturation events per hour, compared to an average 11.17 more desaturation events per hour in the morphine group.
No differences in average postsurgical oxygen saturation, pain relieving effectiveness, tonsillar bleeding or drug adverse events were identified.
“The results of this study support effective posttonsillectomy analgesia in children by using ibuprofen in combination with acetaminophen,” Dr. Kelly and her team wrote.
Ibuprofen appears to be an effective and safer pain reliever than morphine in children undergoing tonsillectomies, according to a recent study.
Although the two medications, administered with acetaminophen, treated pain about equally, morphine showed a greater risk for oxygen desaturation the night after surgery.
“Given the unpredictable posttonsillectomy respiratory response to opioids (codeine, morphine, and hydrocodone) and the analgesic effectiveness of ibuprofen, perhaps the time has come to question the postoperative use of all opioids in this population,” wrote Lauren Kelly, Ph.D., of Western University in London, Ontario, and her associates. The study was published online Jan. 26 (Pediatrics 2014 Jan. 26 [doi: 10.1542/peds.2014-1906].
The researchers randomized 91 children aged 1-10 years to acetaminophen (10-15 mg/kg per dose every 4 hours) with either an age appropriate dose of 0.2-0.5 mg/kg oral morphine every 4 hours or 10 mg/kg of oral ibuprofen every 6 hours after the children underwent a tonsillectomy with or without an adenoidectomy to treat sleep disordered breathing. The study ran from September 2012 to January 2014.
Parents put pulse oximeters on their children the nights before and after surgery to monitor oxygen saturation and apnea events.
The first evening after the surgery, 68% of children receiving ibuprofen showed improvement in oxygen desaturations, compared with 14% of children receiving morphine. The children receiving ibuprofen experienced an average 1.79 fewer desaturation events per hour, compared to an average 11.17 more desaturation events per hour in the morphine group.
No differences in average postsurgical oxygen saturation, pain relieving effectiveness, tonsillar bleeding or drug adverse events were identified.
“The results of this study support effective posttonsillectomy analgesia in children by using ibuprofen in combination with acetaminophen,” Dr. Kelly and her team wrote.
FROM PEDIATRICS
Key clinical point: Ibuprofen safely and effectively replaces morphine for children’s pain relief following tonsillectomy.
Major finding: Among children receiving ibuprofen, 68% improved the first night post surgery, compared with 14% of children receiving morphine.
Data source: A prospective randomized clinical trial of 91 children, aged 1-10 years, assigned to receive 10-15 mg/kg acetaminophen with either 0.2-0.5 mg/kg oral morphine or 10 mg/kg of oral ibuprofen following a tonsillectomy with or without adenoidectomy.
Disclosures: The Canadian Institutes for Health Research Drug Safety and Effectiveness Network funded the study. The authors reported no relevant financial disclosures.
New immunization schedule moving online, but changes are minor
The 2015 childhood and adolescent immunization schedule will be published online only for the first time, according to the American Academy of Pediatrics and the Centers for Disease Control and Prevention.
Moving the schedule online will ensure that providers have the most current recommendations, according to the AAP’s Committee on Infectious Diseases (Pediatrics 2015 [doi:10.1542/peds.2014-3955]). The schedule will be updated when new vaccines are released, new recommendations are published, or when a current recommendation is changed. The website also will include tables to clarify recommended use of certain vaccines, such as vaccines containing pertussis.
Changes to the 2015 schedule are relatively minor. A new column highlights availability of the inactivated and live-attenuated influenza vaccine for children 2-8 years old, along with a recommendation for two doses for some children; a second column indicates when two doses are no longer needed. In addition, a purple bar has been added for young children aged 6 months to less than 12 months traveling outside the United States and who will need the MMR vaccine, the committee said.
Small clarifying changes were added to many vaccinations in the catch-up schedule, and minor changes were made in the footnotes to the DTaP and pneumococcal conjugate vaccines. The influenza footnote now includes new contraindications and precautions for the live-attenuated vaccine. Extensive changes were made to the meningococcal footnote, which clarify dosing schedules for children and infants at high risk, the committee reported.
The 2015 schedule can be found on the CDC website and on the AAP website.
The 2015 childhood and adolescent immunization schedule will be published online only for the first time, according to the American Academy of Pediatrics and the Centers for Disease Control and Prevention.
Moving the schedule online will ensure that providers have the most current recommendations, according to the AAP’s Committee on Infectious Diseases (Pediatrics 2015 [doi:10.1542/peds.2014-3955]). The schedule will be updated when new vaccines are released, new recommendations are published, or when a current recommendation is changed. The website also will include tables to clarify recommended use of certain vaccines, such as vaccines containing pertussis.
Changes to the 2015 schedule are relatively minor. A new column highlights availability of the inactivated and live-attenuated influenza vaccine for children 2-8 years old, along with a recommendation for two doses for some children; a second column indicates when two doses are no longer needed. In addition, a purple bar has been added for young children aged 6 months to less than 12 months traveling outside the United States and who will need the MMR vaccine, the committee said.
Small clarifying changes were added to many vaccinations in the catch-up schedule, and minor changes were made in the footnotes to the DTaP and pneumococcal conjugate vaccines. The influenza footnote now includes new contraindications and precautions for the live-attenuated vaccine. Extensive changes were made to the meningococcal footnote, which clarify dosing schedules for children and infants at high risk, the committee reported.
The 2015 schedule can be found on the CDC website and on the AAP website.
The 2015 childhood and adolescent immunization schedule will be published online only for the first time, according to the American Academy of Pediatrics and the Centers for Disease Control and Prevention.
Moving the schedule online will ensure that providers have the most current recommendations, according to the AAP’s Committee on Infectious Diseases (Pediatrics 2015 [doi:10.1542/peds.2014-3955]). The schedule will be updated when new vaccines are released, new recommendations are published, or when a current recommendation is changed. The website also will include tables to clarify recommended use of certain vaccines, such as vaccines containing pertussis.
Changes to the 2015 schedule are relatively minor. A new column highlights availability of the inactivated and live-attenuated influenza vaccine for children 2-8 years old, along with a recommendation for two doses for some children; a second column indicates when two doses are no longer needed. In addition, a purple bar has been added for young children aged 6 months to less than 12 months traveling outside the United States and who will need the MMR vaccine, the committee said.
Small clarifying changes were added to many vaccinations in the catch-up schedule, and minor changes were made in the footnotes to the DTaP and pneumococcal conjugate vaccines. The influenza footnote now includes new contraindications and precautions for the live-attenuated vaccine. Extensive changes were made to the meningococcal footnote, which clarify dosing schedules for children and infants at high risk, the committee reported.
The 2015 schedule can be found on the CDC website and on the AAP website.
FROM PEDIATRICS