User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
Flu vaccine induces antibodies that help fight H7N9
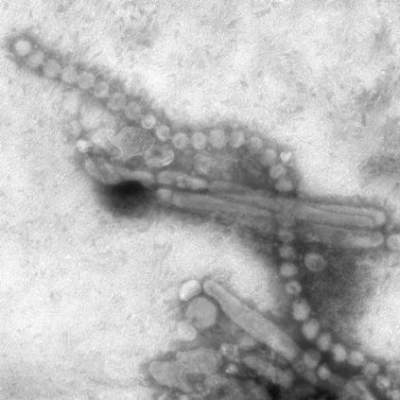
Certain antibodies that develop in the immune system of individuals who have received seasonal flu vaccines appear to broadly neutralize H7 viruses, including the H7N9 avian flu, according to a new study published in the Journal of Clinical Investigation.
In order to determine whether seasonal vaccinations would also induce the production of antibodies against rare flu strains, the team of researchers selected 83 antibodies found in blood samples of 28 vaccinated individuals that reacted with H3N2.
When tested, 7% of these antibodies reacted against rare H7 strains, even though H7 strains were not included in the vaccines the subjects received.
The researchers theorized that the potency of these antibodies could be due to the viral anatomy of the influenza virus – binding to specific sites of the virus allows the antibodies to neutralize a range of influenza strains – although further research will be needed before clinical applications are apparent.
Read the entire article here: J Clin Invest. 2015 (doi:10.1172/JCI74374).
Certain antibodies that develop in the immune system of individuals who have received seasonal flu vaccines appear to broadly neutralize H7 viruses, including the H7N9 avian flu, according to a new study published in the Journal of Clinical Investigation.
In order to determine whether seasonal vaccinations would also induce the production of antibodies against rare flu strains, the team of researchers selected 83 antibodies found in blood samples of 28 vaccinated individuals that reacted with H3N2.
When tested, 7% of these antibodies reacted against rare H7 strains, even though H7 strains were not included in the vaccines the subjects received.
The researchers theorized that the potency of these antibodies could be due to the viral anatomy of the influenza virus – binding to specific sites of the virus allows the antibodies to neutralize a range of influenza strains – although further research will be needed before clinical applications are apparent.
Read the entire article here: J Clin Invest. 2015 (doi:10.1172/JCI74374).
Certain antibodies that develop in the immune system of individuals who have received seasonal flu vaccines appear to broadly neutralize H7 viruses, including the H7N9 avian flu, according to a new study published in the Journal of Clinical Investigation.
In order to determine whether seasonal vaccinations would also induce the production of antibodies against rare flu strains, the team of researchers selected 83 antibodies found in blood samples of 28 vaccinated individuals that reacted with H3N2.
When tested, 7% of these antibodies reacted against rare H7 strains, even though H7 strains were not included in the vaccines the subjects received.
The researchers theorized that the potency of these antibodies could be due to the viral anatomy of the influenza virus – binding to specific sites of the virus allows the antibodies to neutralize a range of influenza strains – although further research will be needed before clinical applications are apparent.
Read the entire article here: J Clin Invest. 2015 (doi:10.1172/JCI74374).
VIDEO: Varenicline may help smokers quit gradually
Is it all or nothing when it comes to quitting smoking?
For many smokers, there may be another approach: A new study showed that a 24-week course of varenicline boosted quit rates among smokers who preferred to gradually reduce their cigarette use (JAMA 2015;313:687-94). That behavioral health reduction strategy may offer 14 million smokers an alternative route to quitting, noted study author Dr. Jon O. Ebbert of the Mayo Clinic, Rochester, Minn.
In a video interview, Dr. Ebbert discusses the strategy and its benefits.
Dr. Ebbert reported receiving grants from Pfizer, JHP Pharmaceuticals, and Orexigen, as well as personal fees from GlaxoSmithKline.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Is it all or nothing when it comes to quitting smoking?
For many smokers, there may be another approach: A new study showed that a 24-week course of varenicline boosted quit rates among smokers who preferred to gradually reduce their cigarette use (JAMA 2015;313:687-94). That behavioral health reduction strategy may offer 14 million smokers an alternative route to quitting, noted study author Dr. Jon O. Ebbert of the Mayo Clinic, Rochester, Minn.
In a video interview, Dr. Ebbert discusses the strategy and its benefits.
Dr. Ebbert reported receiving grants from Pfizer, JHP Pharmaceuticals, and Orexigen, as well as personal fees from GlaxoSmithKline.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Is it all or nothing when it comes to quitting smoking?
For many smokers, there may be another approach: A new study showed that a 24-week course of varenicline boosted quit rates among smokers who preferred to gradually reduce their cigarette use (JAMA 2015;313:687-94). That behavioral health reduction strategy may offer 14 million smokers an alternative route to quitting, noted study author Dr. Jon O. Ebbert of the Mayo Clinic, Rochester, Minn.
In a video interview, Dr. Ebbert discusses the strategy and its benefits.
Dr. Ebbert reported receiving grants from Pfizer, JHP Pharmaceuticals, and Orexigen, as well as personal fees from GlaxoSmithKline.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
FROM JAMA
Varenicline facilitates gradual smoking cessation
A 24-week course of varenicline improved quit rates among smokers who were unwilling or unable to stop smoking abruptly but preferred to gradually reduce their use of cigarettes, according to a report published online Feb. 17 in JAMA.
In an industry-sponsored, randomized, double-blind, controlled trial, participants who were given varenicline showed higher quit rates at the end of treatment as well as 1 year later, compared with those given placebo, said Dr. Jon O. Ebbert of the Mayo Clinic, Rochester, Minn., and his associates.
Current U.S. clinical practice guidelines recommend that smokers set an immediate quit date and quit abruptly, “even though only 8% of smokers report being ready to quit within the next month.” The findings of this study show that a more gradual, reduce-to-quit approach also can be effective, and “would be expected to be of interest to 14 million of the 42 million current smokers in this country,” the investigators noted.
The study was performed at 61 medical centers in 10 countries during a 2-year period. The 1,510 participants would not quit abruptly, as is recommended, but were willing to reduce their smoking and make a quit attempt within the next 3 months. They were asked to reduce their smoking rate by 50% or more by week 4, to further reduce it by 75% or more by week 8, and to quit altogether by week 12.
Study participants, who smoked an average of 10 or more cigarettes per day at baseline, were randomly assigned to receive varenicline (760 patients) or a matching placebo (750 patients) for 24 weeks. All also received written materials and smoking cessation counseling focused on reduction techniques, problem-solving, and skills training. This was provided during 18 clinic visits and 10 telephone sessions of about 10 minutes’ duration.
The primary efficacy endpoint was the continuous abstinence rate during weeks 15-24, which was self-reported by the participants and confirmed using exhaled carbon monoxide measurements. This rate was significantly higher for the varenicline group (32.1%) than for the placebo group (6.9%). The continuous abstinence rate remained significantly higher through 1 year of follow-up for varenicline (27%) than for placebo (9.9%).
In addition, the median time to abstinence was significantly shorter with varenicline (50 days) than with placebo (85 days). Results of sensitivity analyses confirmed those of the primary analysis, the investigators said (JAMA 2015 Feb. 17;313:687-94).
The percentage of participants who reported adverse events was higher with varenicline (82.3% vs. 72.5%), and the difference was largely accounted for by increases in nausea, abnormal dreams, insomnia, constipation, vomiting, and weight gain. Rates of serious adverse events were similar between the two study groups, as were rates of treatment discontinuation (8.4% for varenicline and 7% for placebo). In particular, rates of suicidal ideation or behavior and depression scores were not significantly higher with varenicline.
The study findings indicate that prescribing varenicline “with a recommendation to reduce the number of cigarettes smoked per day, with the eventual goal of quitting, could be a useful therapeutic option for this population of smokers,” Dr. Ebbert and his associates said.
One limitation of this study was that patients were excluded from participating if they had severe psychiatric, pulmonary, cardiovascular, or cerebrovascular disease, which hinders the generalizability of the results to a broader population of smokers. In addition, study participants received significant counseling support that would not necessarily be available to patients in real-world clinical practice, so quit rates would be expected to be lower in actual practice, they added.
This study was funded by Pfizer, maker of varenicline (Chantix). Pfizer also participated in the design and conduct of the study; the collection, analysis, and interpretation of the data; and manuscript preparation. Dr. Ebbert reported receiving grants from Pfizer, JHP Pharmaceuticals, and Orexigen, as well as personal fees from GlaxoSmithKline. His associates reported numerous ties to industry sources.
A 24-week course of varenicline improved quit rates among smokers who were unwilling or unable to stop smoking abruptly but preferred to gradually reduce their use of cigarettes, according to a report published online Feb. 17 in JAMA.
In an industry-sponsored, randomized, double-blind, controlled trial, participants who were given varenicline showed higher quit rates at the end of treatment as well as 1 year later, compared with those given placebo, said Dr. Jon O. Ebbert of the Mayo Clinic, Rochester, Minn., and his associates.
Current U.S. clinical practice guidelines recommend that smokers set an immediate quit date and quit abruptly, “even though only 8% of smokers report being ready to quit within the next month.” The findings of this study show that a more gradual, reduce-to-quit approach also can be effective, and “would be expected to be of interest to 14 million of the 42 million current smokers in this country,” the investigators noted.
The study was performed at 61 medical centers in 10 countries during a 2-year period. The 1,510 participants would not quit abruptly, as is recommended, but were willing to reduce their smoking and make a quit attempt within the next 3 months. They were asked to reduce their smoking rate by 50% or more by week 4, to further reduce it by 75% or more by week 8, and to quit altogether by week 12.
Study participants, who smoked an average of 10 or more cigarettes per day at baseline, were randomly assigned to receive varenicline (760 patients) or a matching placebo (750 patients) for 24 weeks. All also received written materials and smoking cessation counseling focused on reduction techniques, problem-solving, and skills training. This was provided during 18 clinic visits and 10 telephone sessions of about 10 minutes’ duration.
The primary efficacy endpoint was the continuous abstinence rate during weeks 15-24, which was self-reported by the participants and confirmed using exhaled carbon monoxide measurements. This rate was significantly higher for the varenicline group (32.1%) than for the placebo group (6.9%). The continuous abstinence rate remained significantly higher through 1 year of follow-up for varenicline (27%) than for placebo (9.9%).
In addition, the median time to abstinence was significantly shorter with varenicline (50 days) than with placebo (85 days). Results of sensitivity analyses confirmed those of the primary analysis, the investigators said (JAMA 2015 Feb. 17;313:687-94).
The percentage of participants who reported adverse events was higher with varenicline (82.3% vs. 72.5%), and the difference was largely accounted for by increases in nausea, abnormal dreams, insomnia, constipation, vomiting, and weight gain. Rates of serious adverse events were similar between the two study groups, as were rates of treatment discontinuation (8.4% for varenicline and 7% for placebo). In particular, rates of suicidal ideation or behavior and depression scores were not significantly higher with varenicline.
The study findings indicate that prescribing varenicline “with a recommendation to reduce the number of cigarettes smoked per day, with the eventual goal of quitting, could be a useful therapeutic option for this population of smokers,” Dr. Ebbert and his associates said.
One limitation of this study was that patients were excluded from participating if they had severe psychiatric, pulmonary, cardiovascular, or cerebrovascular disease, which hinders the generalizability of the results to a broader population of smokers. In addition, study participants received significant counseling support that would not necessarily be available to patients in real-world clinical practice, so quit rates would be expected to be lower in actual practice, they added.
This study was funded by Pfizer, maker of varenicline (Chantix). Pfizer also participated in the design and conduct of the study; the collection, analysis, and interpretation of the data; and manuscript preparation. Dr. Ebbert reported receiving grants from Pfizer, JHP Pharmaceuticals, and Orexigen, as well as personal fees from GlaxoSmithKline. His associates reported numerous ties to industry sources.
A 24-week course of varenicline improved quit rates among smokers who were unwilling or unable to stop smoking abruptly but preferred to gradually reduce their use of cigarettes, according to a report published online Feb. 17 in JAMA.
In an industry-sponsored, randomized, double-blind, controlled trial, participants who were given varenicline showed higher quit rates at the end of treatment as well as 1 year later, compared with those given placebo, said Dr. Jon O. Ebbert of the Mayo Clinic, Rochester, Minn., and his associates.
Current U.S. clinical practice guidelines recommend that smokers set an immediate quit date and quit abruptly, “even though only 8% of smokers report being ready to quit within the next month.” The findings of this study show that a more gradual, reduce-to-quit approach also can be effective, and “would be expected to be of interest to 14 million of the 42 million current smokers in this country,” the investigators noted.
The study was performed at 61 medical centers in 10 countries during a 2-year period. The 1,510 participants would not quit abruptly, as is recommended, but were willing to reduce their smoking and make a quit attempt within the next 3 months. They were asked to reduce their smoking rate by 50% or more by week 4, to further reduce it by 75% or more by week 8, and to quit altogether by week 12.
Study participants, who smoked an average of 10 or more cigarettes per day at baseline, were randomly assigned to receive varenicline (760 patients) or a matching placebo (750 patients) for 24 weeks. All also received written materials and smoking cessation counseling focused on reduction techniques, problem-solving, and skills training. This was provided during 18 clinic visits and 10 telephone sessions of about 10 minutes’ duration.
The primary efficacy endpoint was the continuous abstinence rate during weeks 15-24, which was self-reported by the participants and confirmed using exhaled carbon monoxide measurements. This rate was significantly higher for the varenicline group (32.1%) than for the placebo group (6.9%). The continuous abstinence rate remained significantly higher through 1 year of follow-up for varenicline (27%) than for placebo (9.9%).
In addition, the median time to abstinence was significantly shorter with varenicline (50 days) than with placebo (85 days). Results of sensitivity analyses confirmed those of the primary analysis, the investigators said (JAMA 2015 Feb. 17;313:687-94).
The percentage of participants who reported adverse events was higher with varenicline (82.3% vs. 72.5%), and the difference was largely accounted for by increases in nausea, abnormal dreams, insomnia, constipation, vomiting, and weight gain. Rates of serious adverse events were similar between the two study groups, as were rates of treatment discontinuation (8.4% for varenicline and 7% for placebo). In particular, rates of suicidal ideation or behavior and depression scores were not significantly higher with varenicline.
The study findings indicate that prescribing varenicline “with a recommendation to reduce the number of cigarettes smoked per day, with the eventual goal of quitting, could be a useful therapeutic option for this population of smokers,” Dr. Ebbert and his associates said.
One limitation of this study was that patients were excluded from participating if they had severe psychiatric, pulmonary, cardiovascular, or cerebrovascular disease, which hinders the generalizability of the results to a broader population of smokers. In addition, study participants received significant counseling support that would not necessarily be available to patients in real-world clinical practice, so quit rates would be expected to be lower in actual practice, they added.
This study was funded by Pfizer, maker of varenicline (Chantix). Pfizer also participated in the design and conduct of the study; the collection, analysis, and interpretation of the data; and manuscript preparation. Dr. Ebbert reported receiving grants from Pfizer, JHP Pharmaceuticals, and Orexigen, as well as personal fees from GlaxoSmithKline. His associates reported numerous ties to industry sources.
Key clinical point: Twenty-four weeks of varenicline therapy aimed at gradually cutting back on cigarette smoking can improve quit rates for smokers during treatment and at 1 year.
Major finding: The primary efficacy endpoint, the continuous abstinence rate during weeks 15-24 of treatment, was significantly higher for the varenicline group (32.1%) than for the placebo group (6.9%).
Data source: An industry-funded, international, randomized, double-blind, controlled trial involving 1,510 adult smokers treated for 24 weeks and followed for 1 year.
Disclosures: This study was funded by Pfizer, maker of varenicline (Chantix). Pfizer also participated in the design and conduct of the study; the collection, analysis, and interpretation of the data; and manuscript preparation. Dr. Ebbert reported receiving grants from Pfizer, JHP Pharmaceuticals, and Orexigen, as well as personal fees from GlaxoSmithKline. His associates reported numerous ties to industry sources.
Methylprednisolone cuts treatment failure in severe CAP
A 5-day course of methylprednisolone reduced the rate of treatment failure in adults with severe community-acquired pneumonia and a high initial inflammatory response, according to a report published online Feb. 17 in JAMA.
“If replicated, these findings would support the use of corticosteroids as adjunctive treatment in this clinical population,” said Dr. Antoni Torres of Institut Clinic del Torax, Hospital Clinic, Barcelona, and his associates.
The use of corticosteroids in community-acquired pneumonia is controversial. Some studies show that corticosteroids decrease radiographic progression of the disease, prevent shock, prevent respiratory failure, decrease length of stay, and reduce mortality. Other studies show no benefit. Dr. Torres and his colleagues studied the use of corticosteroids in the subgroup of patients who present with severe disease and a proinflammatory profile characterized by a serum CRP level over 150 mg/dL – the patients most likely to benefit from anti-inflammatories and least likely to be harmed by steroid-induced superinfection.
In a randomized double-blind trial at three teaching hospitals in Spain, 120 such patients were randomly assigned to receive 5 days of either IV methylprednisolone (61 patients) at a dose of 0.5 mg/kg every 12 hours or a matching placebo (59 patients), in addition to antibiotics. The most common cause of pneumonia in both study groups was Streptococcus pneumoniae, and the most frequent empiric antimicrobial treatment was a combination of ceftriaxone, levofloxacin, and azithromycin.
As expected, CRP and IL-10 levels decreased more in patients who received the corticosteroid than in those who received placebo.
The primary efficacy endpoint was the rate of treatment failure, both within 72 hours (early) and at 72-120 hours (late) after initiation of therapy. This rate was significantly lower in patients who received methylprednisolone (8 patients, or 13%) than in those who received placebo (18, or 31%). This reduction was largely attributed to the prevention of radiographic progression and late septic shock.
However, there were no significant differences in the secondary outcomes of time to clinical stabilization, length of ICU stay, length of hospitalization, and in-hospital mortality, the investigators said (JAMA 2015 Feb. 17 [doi:10.1001/jama.2015.88]). Adverse events were similar between the two study groups and included hyperglycemia (18% with methylprednisolone vs 12% with placebo) and acute kidney injury (13% vs. 14%). One patient taking methylprednisolone developed a superinfection. Other serious adverse events included one case of delirium and one of acute hepatic failure in the methylprednisolone group and one case of GI hemorrhage in the placebo group.
It is likely that the most feared adverse effect of corticosteroid therapy – immunosuppression leading to superinfection – wasn’t an issue in this study because of the short course of treatment and the relatively low dose of methylprednisolone used, the researchers said.
These findings are important because any efficacious adjunctive treatment may help reduce the high mortality associated with severe community-acquired pneumonia. It is estimated that despite effective antibiotic treatment, 12%-36% of patients admitted to an ICU with this disease will die within a short period, Dr. Torres and his associates noted.
They are conducting another study to confirm these results, in part because the small difference between the two study groups in the number of treatment failures – only 10 patients – indicates that replication is needed.
This study was supported by the Sociedad Españolade Neumologia, Societat Catalanade Pneumologia, Fundació Catalanade Pneumologia, Grup de Recercade Qualitatdela Generalitatde Catalunya, Fondode Investigación Sanitaria, Institut d’Investigacions Biomèdiques August Pii Sunyer, and Centrode Investigación Biomédica En Red-Enfermedades Respiratorias. Dr. Torres reported having no financial disclosures; his associates reported ties to GlaxoSmithKline, Dey Pharma, Pfizer, Boehringer Ingelheim, Bayer, Sherin Pharma, AstraZeneca, Cubist, Thermo Diagnostics, and Theravance.
These findings raise the intriguing possibility that corticosteroids block a Jarisch-Herxheimer–like reaction to the initiation of antibiotics in patients who have a high genomic bacterial load. Such a reaction is thought to result from high concentrations of cytokines observed shortly after antibiotics are first administered, possibly through the release of endotoxin or other bacterial mediators in patients with a high bacterial burden.
Richard G. Wunderink, M.D., is at Northwestern University, Chicago. He reported having no financial disclosures. Dr. Wunderink made these remarks in an editorial accompanying Dr. Torres’ report (JAMA 2015;313:673-4).
These findings raise the intriguing possibility that corticosteroids block a Jarisch-Herxheimer–like reaction to the initiation of antibiotics in patients who have a high genomic bacterial load. Such a reaction is thought to result from high concentrations of cytokines observed shortly after antibiotics are first administered, possibly through the release of endotoxin or other bacterial mediators in patients with a high bacterial burden.
Richard G. Wunderink, M.D., is at Northwestern University, Chicago. He reported having no financial disclosures. Dr. Wunderink made these remarks in an editorial accompanying Dr. Torres’ report (JAMA 2015;313:673-4).
These findings raise the intriguing possibility that corticosteroids block a Jarisch-Herxheimer–like reaction to the initiation of antibiotics in patients who have a high genomic bacterial load. Such a reaction is thought to result from high concentrations of cytokines observed shortly after antibiotics are first administered, possibly through the release of endotoxin or other bacterial mediators in patients with a high bacterial burden.
Richard G. Wunderink, M.D., is at Northwestern University, Chicago. He reported having no financial disclosures. Dr. Wunderink made these remarks in an editorial accompanying Dr. Torres’ report (JAMA 2015;313:673-4).
A 5-day course of methylprednisolone reduced the rate of treatment failure in adults with severe community-acquired pneumonia and a high initial inflammatory response, according to a report published online Feb. 17 in JAMA.
“If replicated, these findings would support the use of corticosteroids as adjunctive treatment in this clinical population,” said Dr. Antoni Torres of Institut Clinic del Torax, Hospital Clinic, Barcelona, and his associates.
The use of corticosteroids in community-acquired pneumonia is controversial. Some studies show that corticosteroids decrease radiographic progression of the disease, prevent shock, prevent respiratory failure, decrease length of stay, and reduce mortality. Other studies show no benefit. Dr. Torres and his colleagues studied the use of corticosteroids in the subgroup of patients who present with severe disease and a proinflammatory profile characterized by a serum CRP level over 150 mg/dL – the patients most likely to benefit from anti-inflammatories and least likely to be harmed by steroid-induced superinfection.
In a randomized double-blind trial at three teaching hospitals in Spain, 120 such patients were randomly assigned to receive 5 days of either IV methylprednisolone (61 patients) at a dose of 0.5 mg/kg every 12 hours or a matching placebo (59 patients), in addition to antibiotics. The most common cause of pneumonia in both study groups was Streptococcus pneumoniae, and the most frequent empiric antimicrobial treatment was a combination of ceftriaxone, levofloxacin, and azithromycin.
As expected, CRP and IL-10 levels decreased more in patients who received the corticosteroid than in those who received placebo.
The primary efficacy endpoint was the rate of treatment failure, both within 72 hours (early) and at 72-120 hours (late) after initiation of therapy. This rate was significantly lower in patients who received methylprednisolone (8 patients, or 13%) than in those who received placebo (18, or 31%). This reduction was largely attributed to the prevention of radiographic progression and late septic shock.
However, there were no significant differences in the secondary outcomes of time to clinical stabilization, length of ICU stay, length of hospitalization, and in-hospital mortality, the investigators said (JAMA 2015 Feb. 17 [doi:10.1001/jama.2015.88]). Adverse events were similar between the two study groups and included hyperglycemia (18% with methylprednisolone vs 12% with placebo) and acute kidney injury (13% vs. 14%). One patient taking methylprednisolone developed a superinfection. Other serious adverse events included one case of delirium and one of acute hepatic failure in the methylprednisolone group and one case of GI hemorrhage in the placebo group.
It is likely that the most feared adverse effect of corticosteroid therapy – immunosuppression leading to superinfection – wasn’t an issue in this study because of the short course of treatment and the relatively low dose of methylprednisolone used, the researchers said.
These findings are important because any efficacious adjunctive treatment may help reduce the high mortality associated with severe community-acquired pneumonia. It is estimated that despite effective antibiotic treatment, 12%-36% of patients admitted to an ICU with this disease will die within a short period, Dr. Torres and his associates noted.
They are conducting another study to confirm these results, in part because the small difference between the two study groups in the number of treatment failures – only 10 patients – indicates that replication is needed.
This study was supported by the Sociedad Españolade Neumologia, Societat Catalanade Pneumologia, Fundació Catalanade Pneumologia, Grup de Recercade Qualitatdela Generalitatde Catalunya, Fondode Investigación Sanitaria, Institut d’Investigacions Biomèdiques August Pii Sunyer, and Centrode Investigación Biomédica En Red-Enfermedades Respiratorias. Dr. Torres reported having no financial disclosures; his associates reported ties to GlaxoSmithKline, Dey Pharma, Pfizer, Boehringer Ingelheim, Bayer, Sherin Pharma, AstraZeneca, Cubist, Thermo Diagnostics, and Theravance.
A 5-day course of methylprednisolone reduced the rate of treatment failure in adults with severe community-acquired pneumonia and a high initial inflammatory response, according to a report published online Feb. 17 in JAMA.
“If replicated, these findings would support the use of corticosteroids as adjunctive treatment in this clinical population,” said Dr. Antoni Torres of Institut Clinic del Torax, Hospital Clinic, Barcelona, and his associates.
The use of corticosteroids in community-acquired pneumonia is controversial. Some studies show that corticosteroids decrease radiographic progression of the disease, prevent shock, prevent respiratory failure, decrease length of stay, and reduce mortality. Other studies show no benefit. Dr. Torres and his colleagues studied the use of corticosteroids in the subgroup of patients who present with severe disease and a proinflammatory profile characterized by a serum CRP level over 150 mg/dL – the patients most likely to benefit from anti-inflammatories and least likely to be harmed by steroid-induced superinfection.
In a randomized double-blind trial at three teaching hospitals in Spain, 120 such patients were randomly assigned to receive 5 days of either IV methylprednisolone (61 patients) at a dose of 0.5 mg/kg every 12 hours or a matching placebo (59 patients), in addition to antibiotics. The most common cause of pneumonia in both study groups was Streptococcus pneumoniae, and the most frequent empiric antimicrobial treatment was a combination of ceftriaxone, levofloxacin, and azithromycin.
As expected, CRP and IL-10 levels decreased more in patients who received the corticosteroid than in those who received placebo.
The primary efficacy endpoint was the rate of treatment failure, both within 72 hours (early) and at 72-120 hours (late) after initiation of therapy. This rate was significantly lower in patients who received methylprednisolone (8 patients, or 13%) than in those who received placebo (18, or 31%). This reduction was largely attributed to the prevention of radiographic progression and late septic shock.
However, there were no significant differences in the secondary outcomes of time to clinical stabilization, length of ICU stay, length of hospitalization, and in-hospital mortality, the investigators said (JAMA 2015 Feb. 17 [doi:10.1001/jama.2015.88]). Adverse events were similar between the two study groups and included hyperglycemia (18% with methylprednisolone vs 12% with placebo) and acute kidney injury (13% vs. 14%). One patient taking methylprednisolone developed a superinfection. Other serious adverse events included one case of delirium and one of acute hepatic failure in the methylprednisolone group and one case of GI hemorrhage in the placebo group.
It is likely that the most feared adverse effect of corticosteroid therapy – immunosuppression leading to superinfection – wasn’t an issue in this study because of the short course of treatment and the relatively low dose of methylprednisolone used, the researchers said.
These findings are important because any efficacious adjunctive treatment may help reduce the high mortality associated with severe community-acquired pneumonia. It is estimated that despite effective antibiotic treatment, 12%-36% of patients admitted to an ICU with this disease will die within a short period, Dr. Torres and his associates noted.
They are conducting another study to confirm these results, in part because the small difference between the two study groups in the number of treatment failures – only 10 patients – indicates that replication is needed.
This study was supported by the Sociedad Españolade Neumologia, Societat Catalanade Pneumologia, Fundació Catalanade Pneumologia, Grup de Recercade Qualitatdela Generalitatde Catalunya, Fondode Investigación Sanitaria, Institut d’Investigacions Biomèdiques August Pii Sunyer, and Centrode Investigación Biomédica En Red-Enfermedades Respiratorias. Dr. Torres reported having no financial disclosures; his associates reported ties to GlaxoSmithKline, Dey Pharma, Pfizer, Boehringer Ingelheim, Bayer, Sherin Pharma, AstraZeneca, Cubist, Thermo Diagnostics, and Theravance.
FROM JAMA
Key clinical point: Acute methylprednisolone decreased the rate of treatment failure in severe community-acquired pneumonia with a high initial inflammatory response.
Major finding: The primary efficacy endpoint – the rate of treatment failure – was significantly lower in patients who received methylprednisolone (8, or 13%) than in those who received placebo (18, or 31%).
Data source: A prospective randomized double-bind controlled trial involving 120 adults with severe community-acquired pneumonia treated at three hospitals in Spain.
Disclosures: This study was supported by the Sociedad Españolade Neumologia, Societat Catalanade Pneumologia, Fundació Catalanade Pneumologia, Grup de Recercade Qualitatdela Generalitatde Catalunya, Fondode Investigación Sanitaria, Institut d’Investigacions Biomèdiques August Pii Sunyer, and Centrode Investigación Biomédica En Red-Enfermedades Respiratorias. Dr. Torres reported having no financial disclosures; his associates reported ties to GlaxoSmithKline, Dey Pharma, Pfizer, Boehringer Ingelheim, Bayer, Sherin Pharma, AstraZeneca, Cubist, Thermo Diagnostics, and Theravance.
COPD incidence may be on the decline
Chronic lower airway diseases may have decreased in prevalence in the years between 1987-1994 and 2007-2010, according to Cara N. Halldin, Ph.D., and her associates.
Prevalence of physician-diagnosed chronic obstructive pulmonary disease decreased significantly for males in the second period, while asthma increased only in women. Airflow obstruction also decreased among those with shortness of breath and physician-diagnosed respiratory conditions. COPD outcomes and asthma were highly associated with lower education, smoking, being underweight, obesity, and occupational dust and fume exposure.
“Continued focus on prevention of these diseases through public health interventions is prudent,” the researchers recommended.
Find the full study at Chronic Respiratory Disease (doi:10.1177/1479972314562409).
Chronic lower airway diseases may have decreased in prevalence in the years between 1987-1994 and 2007-2010, according to Cara N. Halldin, Ph.D., and her associates.
Prevalence of physician-diagnosed chronic obstructive pulmonary disease decreased significantly for males in the second period, while asthma increased only in women. Airflow obstruction also decreased among those with shortness of breath and physician-diagnosed respiratory conditions. COPD outcomes and asthma were highly associated with lower education, smoking, being underweight, obesity, and occupational dust and fume exposure.
“Continued focus on prevention of these diseases through public health interventions is prudent,” the researchers recommended.
Find the full study at Chronic Respiratory Disease (doi:10.1177/1479972314562409).
Chronic lower airway diseases may have decreased in prevalence in the years between 1987-1994 and 2007-2010, according to Cara N. Halldin, Ph.D., and her associates.
Prevalence of physician-diagnosed chronic obstructive pulmonary disease decreased significantly for males in the second period, while asthma increased only in women. Airflow obstruction also decreased among those with shortness of breath and physician-diagnosed respiratory conditions. COPD outcomes and asthma were highly associated with lower education, smoking, being underweight, obesity, and occupational dust and fume exposure.
“Continued focus on prevention of these diseases through public health interventions is prudent,” the researchers recommended.
Find the full study at Chronic Respiratory Disease (doi:10.1177/1479972314562409).
Pneumonia risk in COPD rises with inhaled fluticasone furoate/vilanterol
The incidence of pneumonia increased with the use of inhaled fluticasone furoate and vilanterol in patients with chronic obstructive pulmonary disease, reported Dr. Courtney Crim of GSK in Research Triangle Park, N.C., and her colleagues.
Investigators randomized 3,255 COPD patients to receive either vilanterol once a day or vilanterol combined with fluticasone furoate. Chest radiographs showed pneumonia incidence in 3% of patients with vilanterol alone, compared with 7% in those who received both vilanterol and 200 mcg fluticasone furoate. A total of 205 pneumonia events occurred in 181 patients, the authors reported.
“Modifiable pneumonia risk factors should be considered when attempting to optimize COPD management,” Dr. Crim and her coauthors wrote.
Read the full article at the Annals of the American Thoracic Society [2015;12:27-34 [doi:10.1513/AnnalsATS.201409-413OC]).
The incidence of pneumonia increased with the use of inhaled fluticasone furoate and vilanterol in patients with chronic obstructive pulmonary disease, reported Dr. Courtney Crim of GSK in Research Triangle Park, N.C., and her colleagues.
Investigators randomized 3,255 COPD patients to receive either vilanterol once a day or vilanterol combined with fluticasone furoate. Chest radiographs showed pneumonia incidence in 3% of patients with vilanterol alone, compared with 7% in those who received both vilanterol and 200 mcg fluticasone furoate. A total of 205 pneumonia events occurred in 181 patients, the authors reported.
“Modifiable pneumonia risk factors should be considered when attempting to optimize COPD management,” Dr. Crim and her coauthors wrote.
Read the full article at the Annals of the American Thoracic Society [2015;12:27-34 [doi:10.1513/AnnalsATS.201409-413OC]).
The incidence of pneumonia increased with the use of inhaled fluticasone furoate and vilanterol in patients with chronic obstructive pulmonary disease, reported Dr. Courtney Crim of GSK in Research Triangle Park, N.C., and her colleagues.
Investigators randomized 3,255 COPD patients to receive either vilanterol once a day or vilanterol combined with fluticasone furoate. Chest radiographs showed pneumonia incidence in 3% of patients with vilanterol alone, compared with 7% in those who received both vilanterol and 200 mcg fluticasone furoate. A total of 205 pneumonia events occurred in 181 patients, the authors reported.
“Modifiable pneumonia risk factors should be considered when attempting to optimize COPD management,” Dr. Crim and her coauthors wrote.
Read the full article at the Annals of the American Thoracic Society [2015;12:27-34 [doi:10.1513/AnnalsATS.201409-413OC]).
Smoking linked to many ‘new’ causes of death
Smoking causes death from many diseases that until now have not been linked officially to tobacco use, including digestive disorders, liver cirrhosis, infections, renal failure, and breast and prostate cancers, according to a report published online Feb. 11 in the New England Journal of Medicine.
“Our results suggest that the number of persons in the United States who die each year as a result of smoking cigarettes may be substantially greater than currently estimated,” said Brian D. Carter of the epidemiology research program, American Cancer Society, Atlanta, and his associates.
The 2014 Surgeon General’s report estimated that smoking causes more than 480,000 deaths every year in the United States, based on mortality figures from 21 diseases that have been formally established as caused by smoking: 12 types of cancer, 6 types of cardiovascular disease, diabetes, chronic obstructive pulmonary disease, and pneumonia. Mr. Carter and his associates pooled data from five large cohort studies to examine possible associations between smoking and an additional 31 cause-of-death categories. They now estimate that an additional 60,000-120,000 deaths each year can be attributed to smoking.
For their study, the investigators assessed 421,378 men and 532,651 women aged 55 years and older at baseline whose smoking status was carefully recorded and who were followed from 2000 to 2011 in the Cancer Prevention Study II Nutrition Cohort, the Nurses’ Health Study I, the Health Professionals Follow-up Study, the Women’s Health Initiative, and the National Institutes of Health-AARP Diet and Health Study.
As expected, smokers had a twofold to threefold higher mortality from any cause, compared with nonsmokers. Smokers also had a markedly higher risk of death than nonsmokers from all 21 causes already established as attributable to tobacco use, such as lung cancer, oral cancer, ischemic heart disease, atherosclerosis, and stroke. But approximately 17% of smokers’ excess mortality was accounted for by several diseases that previously have not been attributable to tobacco use.
For example, the risk of death due to intestinal ischemia was approximately six times higher among smokers than among nonsmokers, a remarkably strong association that was also reported in the Million Women Study. “Smoking acutely reduces blood flow to the intestines, and evidence suggests that smoking causes risk factors that can often lead to intestinal ischemia, including atherosclerosis, platelet aggregation, and congestive heart failure,” Mr. Carter and his associates said (N. Engl. J. Med. 2015 Feb. 12 [doi:10.1056/NEJMsa140721]). In this study, smoking also more than doubled the risk of dying from other digestive diseases. Previous studies have suggested a link between smoking and digestive disorders such as Crohn’s disease, peptic ulcers, acute pancreatitis, paralytic ileus, bowel obstruction, choletlithiasis, diverticulitis, and gastrointestinal hemorrhage. “Although these diseases are not common causes of death, they account for millions of hospitalizations each year,” the investigators noted.
The mortality risk from liver cirrhosis, after the data were adjusted to account for alcohol consumption, was more than three times higher in smokers than in nonsmokers. Even smokers who did not drink alcohol were at significantly increased risk of cirrhosis, compared with nonsmokers.
The risk of death due to infection was 2.3 times higher in smokers than in nonsmokers. This strong association was dose-dependent, as infection-related mortality rose with increasing smoking intensity. And among study participants who had quit smoking, infection-related mortality declined as the number of years since cessation increased.
The rate of death due to renal failure was twice as high among smokers as among nonsmokers. And the rate of death due to hypertensive heart disease, the only category of heart disease not already established as smoking related, was 2.4 times higher in smokers. The latter association “is relevant for assessing the public health burden of smoking, since a considerable number of deaths in the United States are attributable to hypertensive heart disease,” according to Mr. Carter and his associates.
Smoking also was strongly associated with “multiple diseases too uncommon to examine individually.” This included all rare cancers combined, rare digestive diseases, and respiratory diseases other than those already known to stem from smoking.
In women, smoking raised breast cancer mortality, with a relative risk of 1.3. This association was strongly dose dependent. In men, smoking raised prostate cancer mortality, with a relative risk of 1.4.
This study was limited in that most of the participants were white and better educated than the general population, which may affect the applicability of the results to other populations.
The findings reported by Carter et al. substantiate that until now, estimates of premature deaths due to smoking have markedly underestimated the burden of tobacco use on U.S. society. Attributing 60,000-120,000 additional deaths each year to smoking would have an important public health impact, since it is comparable to the number of annual deaths attributed to excess alcohol intake.
Many people see the war against smoking as one that we have already won, given that rates have declined so much since the 1960s. But these findings show that although we have had some major victories, the war on smoking is far from over.
Dr. Graham A. Colditz is at Washington University, St. Louis. He reported having no financial disclosures. Dr. Colditz made these remarks in an editorial accompanying Mr. Carter’s report (N. Engl. J. Med. 2015 Feb. 12 [doi:10.1056/NEJMe1414318]).
The findings reported by Carter et al. substantiate that until now, estimates of premature deaths due to smoking have markedly underestimated the burden of tobacco use on U.S. society. Attributing 60,000-120,000 additional deaths each year to smoking would have an important public health impact, since it is comparable to the number of annual deaths attributed to excess alcohol intake.
Many people see the war against smoking as one that we have already won, given that rates have declined so much since the 1960s. But these findings show that although we have had some major victories, the war on smoking is far from over.
Dr. Graham A. Colditz is at Washington University, St. Louis. He reported having no financial disclosures. Dr. Colditz made these remarks in an editorial accompanying Mr. Carter’s report (N. Engl. J. Med. 2015 Feb. 12 [doi:10.1056/NEJMe1414318]).
The findings reported by Carter et al. substantiate that until now, estimates of premature deaths due to smoking have markedly underestimated the burden of tobacco use on U.S. society. Attributing 60,000-120,000 additional deaths each year to smoking would have an important public health impact, since it is comparable to the number of annual deaths attributed to excess alcohol intake.
Many people see the war against smoking as one that we have already won, given that rates have declined so much since the 1960s. But these findings show that although we have had some major victories, the war on smoking is far from over.
Dr. Graham A. Colditz is at Washington University, St. Louis. He reported having no financial disclosures. Dr. Colditz made these remarks in an editorial accompanying Mr. Carter’s report (N. Engl. J. Med. 2015 Feb. 12 [doi:10.1056/NEJMe1414318]).
Smoking causes death from many diseases that until now have not been linked officially to tobacco use, including digestive disorders, liver cirrhosis, infections, renal failure, and breast and prostate cancers, according to a report published online Feb. 11 in the New England Journal of Medicine.
“Our results suggest that the number of persons in the United States who die each year as a result of smoking cigarettes may be substantially greater than currently estimated,” said Brian D. Carter of the epidemiology research program, American Cancer Society, Atlanta, and his associates.
The 2014 Surgeon General’s report estimated that smoking causes more than 480,000 deaths every year in the United States, based on mortality figures from 21 diseases that have been formally established as caused by smoking: 12 types of cancer, 6 types of cardiovascular disease, diabetes, chronic obstructive pulmonary disease, and pneumonia. Mr. Carter and his associates pooled data from five large cohort studies to examine possible associations between smoking and an additional 31 cause-of-death categories. They now estimate that an additional 60,000-120,000 deaths each year can be attributed to smoking.
For their study, the investigators assessed 421,378 men and 532,651 women aged 55 years and older at baseline whose smoking status was carefully recorded and who were followed from 2000 to 2011 in the Cancer Prevention Study II Nutrition Cohort, the Nurses’ Health Study I, the Health Professionals Follow-up Study, the Women’s Health Initiative, and the National Institutes of Health-AARP Diet and Health Study.
As expected, smokers had a twofold to threefold higher mortality from any cause, compared with nonsmokers. Smokers also had a markedly higher risk of death than nonsmokers from all 21 causes already established as attributable to tobacco use, such as lung cancer, oral cancer, ischemic heart disease, atherosclerosis, and stroke. But approximately 17% of smokers’ excess mortality was accounted for by several diseases that previously have not been attributable to tobacco use.
For example, the risk of death due to intestinal ischemia was approximately six times higher among smokers than among nonsmokers, a remarkably strong association that was also reported in the Million Women Study. “Smoking acutely reduces blood flow to the intestines, and evidence suggests that smoking causes risk factors that can often lead to intestinal ischemia, including atherosclerosis, platelet aggregation, and congestive heart failure,” Mr. Carter and his associates said (N. Engl. J. Med. 2015 Feb. 12 [doi:10.1056/NEJMsa140721]). In this study, smoking also more than doubled the risk of dying from other digestive diseases. Previous studies have suggested a link between smoking and digestive disorders such as Crohn’s disease, peptic ulcers, acute pancreatitis, paralytic ileus, bowel obstruction, choletlithiasis, diverticulitis, and gastrointestinal hemorrhage. “Although these diseases are not common causes of death, they account for millions of hospitalizations each year,” the investigators noted.
The mortality risk from liver cirrhosis, after the data were adjusted to account for alcohol consumption, was more than three times higher in smokers than in nonsmokers. Even smokers who did not drink alcohol were at significantly increased risk of cirrhosis, compared with nonsmokers.
The risk of death due to infection was 2.3 times higher in smokers than in nonsmokers. This strong association was dose-dependent, as infection-related mortality rose with increasing smoking intensity. And among study participants who had quit smoking, infection-related mortality declined as the number of years since cessation increased.
The rate of death due to renal failure was twice as high among smokers as among nonsmokers. And the rate of death due to hypertensive heart disease, the only category of heart disease not already established as smoking related, was 2.4 times higher in smokers. The latter association “is relevant for assessing the public health burden of smoking, since a considerable number of deaths in the United States are attributable to hypertensive heart disease,” according to Mr. Carter and his associates.
Smoking also was strongly associated with “multiple diseases too uncommon to examine individually.” This included all rare cancers combined, rare digestive diseases, and respiratory diseases other than those already known to stem from smoking.
In women, smoking raised breast cancer mortality, with a relative risk of 1.3. This association was strongly dose dependent. In men, smoking raised prostate cancer mortality, with a relative risk of 1.4.
This study was limited in that most of the participants were white and better educated than the general population, which may affect the applicability of the results to other populations.
Smoking causes death from many diseases that until now have not been linked officially to tobacco use, including digestive disorders, liver cirrhosis, infections, renal failure, and breast and prostate cancers, according to a report published online Feb. 11 in the New England Journal of Medicine.
“Our results suggest that the number of persons in the United States who die each year as a result of smoking cigarettes may be substantially greater than currently estimated,” said Brian D. Carter of the epidemiology research program, American Cancer Society, Atlanta, and his associates.
The 2014 Surgeon General’s report estimated that smoking causes more than 480,000 deaths every year in the United States, based on mortality figures from 21 diseases that have been formally established as caused by smoking: 12 types of cancer, 6 types of cardiovascular disease, diabetes, chronic obstructive pulmonary disease, and pneumonia. Mr. Carter and his associates pooled data from five large cohort studies to examine possible associations between smoking and an additional 31 cause-of-death categories. They now estimate that an additional 60,000-120,000 deaths each year can be attributed to smoking.
For their study, the investigators assessed 421,378 men and 532,651 women aged 55 years and older at baseline whose smoking status was carefully recorded and who were followed from 2000 to 2011 in the Cancer Prevention Study II Nutrition Cohort, the Nurses’ Health Study I, the Health Professionals Follow-up Study, the Women’s Health Initiative, and the National Institutes of Health-AARP Diet and Health Study.
As expected, smokers had a twofold to threefold higher mortality from any cause, compared with nonsmokers. Smokers also had a markedly higher risk of death than nonsmokers from all 21 causes already established as attributable to tobacco use, such as lung cancer, oral cancer, ischemic heart disease, atherosclerosis, and stroke. But approximately 17% of smokers’ excess mortality was accounted for by several diseases that previously have not been attributable to tobacco use.
For example, the risk of death due to intestinal ischemia was approximately six times higher among smokers than among nonsmokers, a remarkably strong association that was also reported in the Million Women Study. “Smoking acutely reduces blood flow to the intestines, and evidence suggests that smoking causes risk factors that can often lead to intestinal ischemia, including atherosclerosis, platelet aggregation, and congestive heart failure,” Mr. Carter and his associates said (N. Engl. J. Med. 2015 Feb. 12 [doi:10.1056/NEJMsa140721]). In this study, smoking also more than doubled the risk of dying from other digestive diseases. Previous studies have suggested a link between smoking and digestive disorders such as Crohn’s disease, peptic ulcers, acute pancreatitis, paralytic ileus, bowel obstruction, choletlithiasis, diverticulitis, and gastrointestinal hemorrhage. “Although these diseases are not common causes of death, they account for millions of hospitalizations each year,” the investigators noted.
The mortality risk from liver cirrhosis, after the data were adjusted to account for alcohol consumption, was more than three times higher in smokers than in nonsmokers. Even smokers who did not drink alcohol were at significantly increased risk of cirrhosis, compared with nonsmokers.
The risk of death due to infection was 2.3 times higher in smokers than in nonsmokers. This strong association was dose-dependent, as infection-related mortality rose with increasing smoking intensity. And among study participants who had quit smoking, infection-related mortality declined as the number of years since cessation increased.
The rate of death due to renal failure was twice as high among smokers as among nonsmokers. And the rate of death due to hypertensive heart disease, the only category of heart disease not already established as smoking related, was 2.4 times higher in smokers. The latter association “is relevant for assessing the public health burden of smoking, since a considerable number of deaths in the United States are attributable to hypertensive heart disease,” according to Mr. Carter and his associates.
Smoking also was strongly associated with “multiple diseases too uncommon to examine individually.” This included all rare cancers combined, rare digestive diseases, and respiratory diseases other than those already known to stem from smoking.
In women, smoking raised breast cancer mortality, with a relative risk of 1.3. This association was strongly dose dependent. In men, smoking raised prostate cancer mortality, with a relative risk of 1.4.
This study was limited in that most of the participants were white and better educated than the general population, which may affect the applicability of the results to other populations.
Key clinical point: Smoking is now thought to cause many deaths from infections; renal failure; liver cirrhosis; digestive diseases; and breast, prostate, and other cancers.
Major finding: Several diseases that previously have not been attributable to tobacco use accounted for 17% of smokers’ excess mortality.
Data source: An analysis of pooled data from five large cohort studies involving 954,029 people aged 55 years and older followed for 12 years to examine associations between smoking and 52 possible causes of death.
Disclosures: This study was supported by the American Cancer Society. Mr. Carter reported having no financial disclosures; one of his associates reported receiving grant support from Novo Nordisk.
Sleep-disordered breathing in pregnancy linked to cardiometabolic morbidity
SAN DIEGO – The prevalence of sleep-disordered breathing during pregnancy is low, but a large prospective study links it to both hypertensive disorders of pregnancy and gestational diabetes.
In an analysis of 3,702 nulliparous women, researchers found that the prevalence of sleep-disordered breathing (SDB) was 3.3% during early pregnancy (weeks 6-15) and 8.1% in midpregnancy (weeks 22-31).
SDB in midpregnancy was significantly associated with hypertensive disorders of pregnancy, while having SDB in both early and midpregnncy was significantly associated with gestational diabetes mellitus (GDM).
While the majority of sleep-disordered breathing cases in the study were mild, “clinical experience tells us that these women are not routinely being diagnosed and treated for SDB,” Dr. Francesca L. Facco said at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine. “Our data demonstrate that even modest elevations of apnea-hypopnea index scores in pregnancy are associated with cardiometabolic morbidity. These findings are important because SDB, unlike many other risk factors, is potentially modifiable.”
In a study that Dr. Facco presented on behalf of the National Institute of Child Health and Human Development NuMoM2b Network, researchers at eight clinical sites in the United States set out to determine if SDB during pregnancy is a risk factor for adverse pregnancy outcomes.
Obstructive sleep apnea is the most common type of sleep disorder, said Dr. Facco of the department of obstetrics and gynecology at Magee-Women’s Hospital at the University of Pittsburgh Medical Center. In adults, an apnea-hypopnea index (AHI) of greater than or equal to 5 is the minimum criterion for establishing a sleep-disordered breathing diagnosis, while severity is classified by the number of events per hour.
“The prevalence, incidence, and severity of SDB and its impact on pregnancy remain undetermined,” Dr. Facco said. “This is despite the fact that pregnant women may be particularly disposed to SDB, given the physiologic changes associated with the gravid state, such as rapid weight gain and edema.”
Researchers recruited 3,702 women from a prospective cohort of 10,000 nulliparous women. The subjects underwent overnight in-home assessments of SDB during early pregnancy and midpregnancy. All studies were scored by a central sleep reading center.
“Currently there are no pregnancy-specific guidelines for SDB treatment and no data on which to base fetal and maternal parameters for treatment,” Dr. Facco noted. “Given the clinical equipoise that surrounds the issue, all participants and care providers were blinded to the sleep study results unless a study was identified as an urgent alert study.”
The primary outcome measures were hypertensive disorders of pregnancy and gestational diabetes. Hypertensive disorders included mild, severe, or superimposed preeclampsia, eclampsia, and gestational hypertension diagnosed in the antepartum period. The Embletta Portable Diagnostic System was used to perform the home sleep test.
Data were analyzed using multivariable logistic regression adjusted for age, body mass index, and the presence of chronic hypertension in early pregnancy analyses, as well as weight gain between visits for the midpregnancy analyses. All of the women included in the study had singleton pregnancies.
The researchers obtained complete data from 3,261 women in early pregnancy and from 2,511 women in midpregnancy. Among the 3,132 women with hypertension data, there was a 12.4% incidence of hypertensive disorders at baseline, with a 5.5% incidence of preeclampsia. Among 3,076 women with gestational diabetes mellitus data (cases of pregestational diabetes were excluded), the rate of GDM was 3.9%.
The mean age of study participants was 27 years; 60% were non-Hispanic white, 18% were Hispanic, 14% were non-Hispanic black, and the remainder were from other ethnic groups. At baseline, about half (49%) were normal weight, 25% were overweight, 23% were obese or morbidly obese, and the remainder were underweight.
SDB in midpregnancy was significantly associated with hypertensive disorders of pregnancy (an adjusted odds ratio of 1.62; P = .0156). A similar trend was seen in early pregnancy, but the association did not reach statistical significance in adjusted analyses (aOR, 1.44; P = .1434), according to Dr. Facco.
SDB in both early and midpregnancy was significantly associated with GDM (aOR, 3.62; P < .0001 and aOR, 2.79; P = .0002, respectively).
The study was funded by the NICHD. Dr. Facco reported having no relevant financial conflicts.
On Twitter @dougbrunk
SAN DIEGO – The prevalence of sleep-disordered breathing during pregnancy is low, but a large prospective study links it to both hypertensive disorders of pregnancy and gestational diabetes.
In an analysis of 3,702 nulliparous women, researchers found that the prevalence of sleep-disordered breathing (SDB) was 3.3% during early pregnancy (weeks 6-15) and 8.1% in midpregnancy (weeks 22-31).
SDB in midpregnancy was significantly associated with hypertensive disorders of pregnancy, while having SDB in both early and midpregnncy was significantly associated with gestational diabetes mellitus (GDM).
While the majority of sleep-disordered breathing cases in the study were mild, “clinical experience tells us that these women are not routinely being diagnosed and treated for SDB,” Dr. Francesca L. Facco said at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine. “Our data demonstrate that even modest elevations of apnea-hypopnea index scores in pregnancy are associated with cardiometabolic morbidity. These findings are important because SDB, unlike many other risk factors, is potentially modifiable.”
In a study that Dr. Facco presented on behalf of the National Institute of Child Health and Human Development NuMoM2b Network, researchers at eight clinical sites in the United States set out to determine if SDB during pregnancy is a risk factor for adverse pregnancy outcomes.
Obstructive sleep apnea is the most common type of sleep disorder, said Dr. Facco of the department of obstetrics and gynecology at Magee-Women’s Hospital at the University of Pittsburgh Medical Center. In adults, an apnea-hypopnea index (AHI) of greater than or equal to 5 is the minimum criterion for establishing a sleep-disordered breathing diagnosis, while severity is classified by the number of events per hour.
“The prevalence, incidence, and severity of SDB and its impact on pregnancy remain undetermined,” Dr. Facco said. “This is despite the fact that pregnant women may be particularly disposed to SDB, given the physiologic changes associated with the gravid state, such as rapid weight gain and edema.”
Researchers recruited 3,702 women from a prospective cohort of 10,000 nulliparous women. The subjects underwent overnight in-home assessments of SDB during early pregnancy and midpregnancy. All studies were scored by a central sleep reading center.
“Currently there are no pregnancy-specific guidelines for SDB treatment and no data on which to base fetal and maternal parameters for treatment,” Dr. Facco noted. “Given the clinical equipoise that surrounds the issue, all participants and care providers were blinded to the sleep study results unless a study was identified as an urgent alert study.”
The primary outcome measures were hypertensive disorders of pregnancy and gestational diabetes. Hypertensive disorders included mild, severe, or superimposed preeclampsia, eclampsia, and gestational hypertension diagnosed in the antepartum period. The Embletta Portable Diagnostic System was used to perform the home sleep test.
Data were analyzed using multivariable logistic regression adjusted for age, body mass index, and the presence of chronic hypertension in early pregnancy analyses, as well as weight gain between visits for the midpregnancy analyses. All of the women included in the study had singleton pregnancies.
The researchers obtained complete data from 3,261 women in early pregnancy and from 2,511 women in midpregnancy. Among the 3,132 women with hypertension data, there was a 12.4% incidence of hypertensive disorders at baseline, with a 5.5% incidence of preeclampsia. Among 3,076 women with gestational diabetes mellitus data (cases of pregestational diabetes were excluded), the rate of GDM was 3.9%.
The mean age of study participants was 27 years; 60% were non-Hispanic white, 18% were Hispanic, 14% were non-Hispanic black, and the remainder were from other ethnic groups. At baseline, about half (49%) were normal weight, 25% were overweight, 23% were obese or morbidly obese, and the remainder were underweight.
SDB in midpregnancy was significantly associated with hypertensive disorders of pregnancy (an adjusted odds ratio of 1.62; P = .0156). A similar trend was seen in early pregnancy, but the association did not reach statistical significance in adjusted analyses (aOR, 1.44; P = .1434), according to Dr. Facco.
SDB in both early and midpregnancy was significantly associated with GDM (aOR, 3.62; P < .0001 and aOR, 2.79; P = .0002, respectively).
The study was funded by the NICHD. Dr. Facco reported having no relevant financial conflicts.
On Twitter @dougbrunk
SAN DIEGO – The prevalence of sleep-disordered breathing during pregnancy is low, but a large prospective study links it to both hypertensive disorders of pregnancy and gestational diabetes.
In an analysis of 3,702 nulliparous women, researchers found that the prevalence of sleep-disordered breathing (SDB) was 3.3% during early pregnancy (weeks 6-15) and 8.1% in midpregnancy (weeks 22-31).
SDB in midpregnancy was significantly associated with hypertensive disorders of pregnancy, while having SDB in both early and midpregnncy was significantly associated with gestational diabetes mellitus (GDM).
While the majority of sleep-disordered breathing cases in the study were mild, “clinical experience tells us that these women are not routinely being diagnosed and treated for SDB,” Dr. Francesca L. Facco said at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine. “Our data demonstrate that even modest elevations of apnea-hypopnea index scores in pregnancy are associated with cardiometabolic morbidity. These findings are important because SDB, unlike many other risk factors, is potentially modifiable.”
In a study that Dr. Facco presented on behalf of the National Institute of Child Health and Human Development NuMoM2b Network, researchers at eight clinical sites in the United States set out to determine if SDB during pregnancy is a risk factor for adverse pregnancy outcomes.
Obstructive sleep apnea is the most common type of sleep disorder, said Dr. Facco of the department of obstetrics and gynecology at Magee-Women’s Hospital at the University of Pittsburgh Medical Center. In adults, an apnea-hypopnea index (AHI) of greater than or equal to 5 is the minimum criterion for establishing a sleep-disordered breathing diagnosis, while severity is classified by the number of events per hour.
“The prevalence, incidence, and severity of SDB and its impact on pregnancy remain undetermined,” Dr. Facco said. “This is despite the fact that pregnant women may be particularly disposed to SDB, given the physiologic changes associated with the gravid state, such as rapid weight gain and edema.”
Researchers recruited 3,702 women from a prospective cohort of 10,000 nulliparous women. The subjects underwent overnight in-home assessments of SDB during early pregnancy and midpregnancy. All studies were scored by a central sleep reading center.
“Currently there are no pregnancy-specific guidelines for SDB treatment and no data on which to base fetal and maternal parameters for treatment,” Dr. Facco noted. “Given the clinical equipoise that surrounds the issue, all participants and care providers were blinded to the sleep study results unless a study was identified as an urgent alert study.”
The primary outcome measures were hypertensive disorders of pregnancy and gestational diabetes. Hypertensive disorders included mild, severe, or superimposed preeclampsia, eclampsia, and gestational hypertension diagnosed in the antepartum period. The Embletta Portable Diagnostic System was used to perform the home sleep test.
Data were analyzed using multivariable logistic regression adjusted for age, body mass index, and the presence of chronic hypertension in early pregnancy analyses, as well as weight gain between visits for the midpregnancy analyses. All of the women included in the study had singleton pregnancies.
The researchers obtained complete data from 3,261 women in early pregnancy and from 2,511 women in midpregnancy. Among the 3,132 women with hypertension data, there was a 12.4% incidence of hypertensive disorders at baseline, with a 5.5% incidence of preeclampsia. Among 3,076 women with gestational diabetes mellitus data (cases of pregestational diabetes were excluded), the rate of GDM was 3.9%.
The mean age of study participants was 27 years; 60% were non-Hispanic white, 18% were Hispanic, 14% were non-Hispanic black, and the remainder were from other ethnic groups. At baseline, about half (49%) were normal weight, 25% were overweight, 23% were obese or morbidly obese, and the remainder were underweight.
SDB in midpregnancy was significantly associated with hypertensive disorders of pregnancy (an adjusted odds ratio of 1.62; P = .0156). A similar trend was seen in early pregnancy, but the association did not reach statistical significance in adjusted analyses (aOR, 1.44; P = .1434), according to Dr. Facco.
SDB in both early and midpregnancy was significantly associated with GDM (aOR, 3.62; P < .0001 and aOR, 2.79; P = .0002, respectively).
The study was funded by the NICHD. Dr. Facco reported having no relevant financial conflicts.
On Twitter @dougbrunk
AT THE PREGNANCY MEETING
Key clinical point: Sleep-disordered breathing in pregnancy is associated with hypertensive disorders of pregnancy and gestational diabetes mellitus.
Major finding: SDB in midpregnancy was significantly associated with hypertensive disorders of pregnancy (an adjusted odds ratio of 1.62; P = .0156). SDB in both early and midpregnancy was significantly associated with GDM (aOR, 3.62; P < .0001 and aOR, 2.79; P = .0002, respectively).
Data source: An analysis of 3,702 women from a prospective cohort of 10,000 nulliparous women.
Disclosures: The study was funded by the National Institute of Child Health and Human Development. Dr. Facco reported having no relevant financial conflicts.
Upper airway stimulation an option in some patients
CORONADO, CALIF. – Some patients with treatment-refractory obstructive sleep apnea may be candidates for upper airway stimulation, though those with complete concentric palatal collapse may not respond, according to Dr. Marion Boyd Gillespie.
“There’s been sound research showing that patients with obstructive sleep apnea have reduced neural tone, so it may not be due to fat in the tongue; some of it may have to do with reduced neural tone,” Dr. Gillespie, who directs the snoring clinics at the Medical University of South Carolina, Charleston, said at the Triological Society’s Combined Sections Meeting. “During these apneic events, there’s a reduction in the neural tone of the genioglossus muscle, which is the main dilator of the upper airway. With upper airway stimulation, we’re trying to account for that loss of neural tone by providing more neural impulse to these muscle groups that perform the dilator functions.”
In 2014 the Food and Drug Administration cleared an upper airway stimulation system manufactured by Inspire Medical Systems, a pacemakerlike device that’s implanted in the subclavicular space. The system features a stimulator lead that attaches to the right hypoglossal nerve and a sensing lead that goes between the external and internal intercostal muscles to detect breathing. “That allows the device to know when in the phase of respiration to fire,” said Dr. Gillespie, professor of otolaryngology–head and neck surgery at the university. “The sensing lead detects the respiratory wave, and the stimulatory lead starts stimulation at the end of expiration, because that’s when the airway is in its most collapsible state. It continues about two-thirds of the way through the inspiratory cycle to keep the airway open.”
Titration of the device is very similar to continuous positive airway pressure, he continued. Once implanted, the patient “will go back to the sleep lab where a tech who’s trained in the device will ramp up stimulation until observed apneas and hypopneas are adequately reduced. You would think that isolated stimulation of the hypoglossal nerve would only open up the airway at the level of the tongue. However, our initial investigation showed that there is dilation at the velopharynx as well,” Dr. Gillespie said. By moving the tongue out of the posterior airway, “you’re moving the dorsum of the tongue away from the velopharynx. You’re also getting active traction on the palatoglossal fold,” he added.
Results of the initial trial of the system in 126 patients with a mean body mass index of 28.4 kg/m2 were published last year (N. Engl. J. Med. 2014;370:139-49). At 12 months of follow-up, patients experienced a 68% overall reduction in their apnea-hypopnea index (AHI) score, from a preoperative mean of 29 to a postoperative mean of 9. In addition, patients had a 70% overall reduction in their oxygen desaturation index (ODI). The researchers also observed normalization of patient-based outcomes, with improvement in the Functional Outcomes of Sleep Questionnaire score and reduction of the Epworth Sleepiness Scale score to a level of 10 on average. “We also saw a reduction of snoring,” said Dr. Gillespie, who was a member of the research team. “Snoring went from 72% of patients having severe, annoying snoring to the point where a bed partner leaves the room, to 15% postoperatively.” Even so, 96% of patients who had a previous history of uvulopalatopharyngoplasty (UPPP) or laser-assisted uvulopalatoplasty (LAUP) still had tongue-based collapse after 12 months of follow-up. “But we found that their response to this therapy was just as good as people who had never had a UPPP or LAUP,” Dr. Gillespie said at the meeting, jointly sponsored by the Triological Society and the American College of Surgeons. “So it seems like patients who have failed UPPP are still good candidates for upper airway stimulation therapy.”
Dr. Gillespie noted that selection criteria for the trial were limited to patients with a BMI of less than 32 kg/m2 and to those who did not have complete circumferential collapse at the level of the soft palate on preoperative drug-induced endoscopy. These criteria were based on an earlier pilot study that showed that patients with complete circumferential collapse at the level of the soft palate did not respond to upper airway stimulation (J. Clin. Sleep Med. 2013;9:433-8).
Dr. Gillespie disclosed that he has received research support from Inspire Medical Systems, Olympus, and Surgical Specialties. He is also a consultant for those companies as well as for Medtronic.
On Twitter @dougbrunk
Dr. David Schulman, FCCP, comments: The data presented by Dr. Gillespie add to the growing body of literature showing the benefits of stimulation of the upper airway muscles during sleep in a selected subgroup of obstructive sleep apnea (OSA) patients, demonstrating improvements in both physiologic and functional parameters. Given the well-described issues with continuous positive airway pressure (CPAP) adherence and the lesser efficacy of currently available CPAP alternatives, patients with obstructive sleep apnea and their providers have long awaited access to hypoglossal nerve stimulators to add to the armamentarium of options for management of the disorder.
While early data continue to show promise for this treatment, a number of physiologic and anatomic characteristics serve as relative contraindications, limiting the generalizability of study results to some patient populations (such as those with body mass index greater than 32 kg/m2 or those with concentric collapse of the soft palate). While upper airway stimulation is not likely to be the first-line OSA treatment for the majority of patients, it is an important step forward for those unwilling or unable to use CPAP.
Dr. David Schulman, FCCP, comments: The data presented by Dr. Gillespie add to the growing body of literature showing the benefits of stimulation of the upper airway muscles during sleep in a selected subgroup of obstructive sleep apnea (OSA) patients, demonstrating improvements in both physiologic and functional parameters. Given the well-described issues with continuous positive airway pressure (CPAP) adherence and the lesser efficacy of currently available CPAP alternatives, patients with obstructive sleep apnea and their providers have long awaited access to hypoglossal nerve stimulators to add to the armamentarium of options for management of the disorder.
While early data continue to show promise for this treatment, a number of physiologic and anatomic characteristics serve as relative contraindications, limiting the generalizability of study results to some patient populations (such as those with body mass index greater than 32 kg/m2 or those with concentric collapse of the soft palate). While upper airway stimulation is not likely to be the first-line OSA treatment for the majority of patients, it is an important step forward for those unwilling or unable to use CPAP.
Dr. David Schulman, FCCP, comments: The data presented by Dr. Gillespie add to the growing body of literature showing the benefits of stimulation of the upper airway muscles during sleep in a selected subgroup of obstructive sleep apnea (OSA) patients, demonstrating improvements in both physiologic and functional parameters. Given the well-described issues with continuous positive airway pressure (CPAP) adherence and the lesser efficacy of currently available CPAP alternatives, patients with obstructive sleep apnea and their providers have long awaited access to hypoglossal nerve stimulators to add to the armamentarium of options for management of the disorder.
While early data continue to show promise for this treatment, a number of physiologic and anatomic characteristics serve as relative contraindications, limiting the generalizability of study results to some patient populations (such as those with body mass index greater than 32 kg/m2 or those with concentric collapse of the soft palate). While upper airway stimulation is not likely to be the first-line OSA treatment for the majority of patients, it is an important step forward for those unwilling or unable to use CPAP.
CORONADO, CALIF. – Some patients with treatment-refractory obstructive sleep apnea may be candidates for upper airway stimulation, though those with complete concentric palatal collapse may not respond, according to Dr. Marion Boyd Gillespie.
“There’s been sound research showing that patients with obstructive sleep apnea have reduced neural tone, so it may not be due to fat in the tongue; some of it may have to do with reduced neural tone,” Dr. Gillespie, who directs the snoring clinics at the Medical University of South Carolina, Charleston, said at the Triological Society’s Combined Sections Meeting. “During these apneic events, there’s a reduction in the neural tone of the genioglossus muscle, which is the main dilator of the upper airway. With upper airway stimulation, we’re trying to account for that loss of neural tone by providing more neural impulse to these muscle groups that perform the dilator functions.”
In 2014 the Food and Drug Administration cleared an upper airway stimulation system manufactured by Inspire Medical Systems, a pacemakerlike device that’s implanted in the subclavicular space. The system features a stimulator lead that attaches to the right hypoglossal nerve and a sensing lead that goes between the external and internal intercostal muscles to detect breathing. “That allows the device to know when in the phase of respiration to fire,” said Dr. Gillespie, professor of otolaryngology–head and neck surgery at the university. “The sensing lead detects the respiratory wave, and the stimulatory lead starts stimulation at the end of expiration, because that’s when the airway is in its most collapsible state. It continues about two-thirds of the way through the inspiratory cycle to keep the airway open.”
Titration of the device is very similar to continuous positive airway pressure, he continued. Once implanted, the patient “will go back to the sleep lab where a tech who’s trained in the device will ramp up stimulation until observed apneas and hypopneas are adequately reduced. You would think that isolated stimulation of the hypoglossal nerve would only open up the airway at the level of the tongue. However, our initial investigation showed that there is dilation at the velopharynx as well,” Dr. Gillespie said. By moving the tongue out of the posterior airway, “you’re moving the dorsum of the tongue away from the velopharynx. You’re also getting active traction on the palatoglossal fold,” he added.
Results of the initial trial of the system in 126 patients with a mean body mass index of 28.4 kg/m2 were published last year (N. Engl. J. Med. 2014;370:139-49). At 12 months of follow-up, patients experienced a 68% overall reduction in their apnea-hypopnea index (AHI) score, from a preoperative mean of 29 to a postoperative mean of 9. In addition, patients had a 70% overall reduction in their oxygen desaturation index (ODI). The researchers also observed normalization of patient-based outcomes, with improvement in the Functional Outcomes of Sleep Questionnaire score and reduction of the Epworth Sleepiness Scale score to a level of 10 on average. “We also saw a reduction of snoring,” said Dr. Gillespie, who was a member of the research team. “Snoring went from 72% of patients having severe, annoying snoring to the point where a bed partner leaves the room, to 15% postoperatively.” Even so, 96% of patients who had a previous history of uvulopalatopharyngoplasty (UPPP) or laser-assisted uvulopalatoplasty (LAUP) still had tongue-based collapse after 12 months of follow-up. “But we found that their response to this therapy was just as good as people who had never had a UPPP or LAUP,” Dr. Gillespie said at the meeting, jointly sponsored by the Triological Society and the American College of Surgeons. “So it seems like patients who have failed UPPP are still good candidates for upper airway stimulation therapy.”
Dr. Gillespie noted that selection criteria for the trial were limited to patients with a BMI of less than 32 kg/m2 and to those who did not have complete circumferential collapse at the level of the soft palate on preoperative drug-induced endoscopy. These criteria were based on an earlier pilot study that showed that patients with complete circumferential collapse at the level of the soft palate did not respond to upper airway stimulation (J. Clin. Sleep Med. 2013;9:433-8).
Dr. Gillespie disclosed that he has received research support from Inspire Medical Systems, Olympus, and Surgical Specialties. He is also a consultant for those companies as well as for Medtronic.
On Twitter @dougbrunk
CORONADO, CALIF. – Some patients with treatment-refractory obstructive sleep apnea may be candidates for upper airway stimulation, though those with complete concentric palatal collapse may not respond, according to Dr. Marion Boyd Gillespie.
“There’s been sound research showing that patients with obstructive sleep apnea have reduced neural tone, so it may not be due to fat in the tongue; some of it may have to do with reduced neural tone,” Dr. Gillespie, who directs the snoring clinics at the Medical University of South Carolina, Charleston, said at the Triological Society’s Combined Sections Meeting. “During these apneic events, there’s a reduction in the neural tone of the genioglossus muscle, which is the main dilator of the upper airway. With upper airway stimulation, we’re trying to account for that loss of neural tone by providing more neural impulse to these muscle groups that perform the dilator functions.”
In 2014 the Food and Drug Administration cleared an upper airway stimulation system manufactured by Inspire Medical Systems, a pacemakerlike device that’s implanted in the subclavicular space. The system features a stimulator lead that attaches to the right hypoglossal nerve and a sensing lead that goes between the external and internal intercostal muscles to detect breathing. “That allows the device to know when in the phase of respiration to fire,” said Dr. Gillespie, professor of otolaryngology–head and neck surgery at the university. “The sensing lead detects the respiratory wave, and the stimulatory lead starts stimulation at the end of expiration, because that’s when the airway is in its most collapsible state. It continues about two-thirds of the way through the inspiratory cycle to keep the airway open.”
Titration of the device is very similar to continuous positive airway pressure, he continued. Once implanted, the patient “will go back to the sleep lab where a tech who’s trained in the device will ramp up stimulation until observed apneas and hypopneas are adequately reduced. You would think that isolated stimulation of the hypoglossal nerve would only open up the airway at the level of the tongue. However, our initial investigation showed that there is dilation at the velopharynx as well,” Dr. Gillespie said. By moving the tongue out of the posterior airway, “you’re moving the dorsum of the tongue away from the velopharynx. You’re also getting active traction on the palatoglossal fold,” he added.
Results of the initial trial of the system in 126 patients with a mean body mass index of 28.4 kg/m2 were published last year (N. Engl. J. Med. 2014;370:139-49). At 12 months of follow-up, patients experienced a 68% overall reduction in their apnea-hypopnea index (AHI) score, from a preoperative mean of 29 to a postoperative mean of 9. In addition, patients had a 70% overall reduction in their oxygen desaturation index (ODI). The researchers also observed normalization of patient-based outcomes, with improvement in the Functional Outcomes of Sleep Questionnaire score and reduction of the Epworth Sleepiness Scale score to a level of 10 on average. “We also saw a reduction of snoring,” said Dr. Gillespie, who was a member of the research team. “Snoring went from 72% of patients having severe, annoying snoring to the point where a bed partner leaves the room, to 15% postoperatively.” Even so, 96% of patients who had a previous history of uvulopalatopharyngoplasty (UPPP) or laser-assisted uvulopalatoplasty (LAUP) still had tongue-based collapse after 12 months of follow-up. “But we found that their response to this therapy was just as good as people who had never had a UPPP or LAUP,” Dr. Gillespie said at the meeting, jointly sponsored by the Triological Society and the American College of Surgeons. “So it seems like patients who have failed UPPP are still good candidates for upper airway stimulation therapy.”
Dr. Gillespie noted that selection criteria for the trial were limited to patients with a BMI of less than 32 kg/m2 and to those who did not have complete circumferential collapse at the level of the soft palate on preoperative drug-induced endoscopy. These criteria were based on an earlier pilot study that showed that patients with complete circumferential collapse at the level of the soft palate did not respond to upper airway stimulation (J. Clin. Sleep Med. 2013;9:433-8).
Dr. Gillespie disclosed that he has received research support from Inspire Medical Systems, Olympus, and Surgical Specialties. He is also a consultant for those companies as well as for Medtronic.
On Twitter @dougbrunk
EXPERT ANALYSIS AT THE COMBINED SECTIONS WINTER MEETING
Medicare now covers low-dose CT screen for lung cancer
Effective immediately, Medicare will cover annual lung cancer screening with low-dose CT for certain beneficiaries, according to a Feb. 5 national coverage determination.
To qualify, beneficiaries must be 55-77 years old, have a smoking history of at least a 30 pack-years, exhibit no signs or symptoms of lung cancer, and currently smoke or have quit within 15 years. They also must have a written screening order from their provider.
Coverage includes a counseling visit for shared decision-making, so patients know beforehand the “benefits and harms of screening, follow-up diagnostic testing, over-diagnosis, false positive[s], and total radiation exposure,” among other things, according to the decision memo from the Centers for Medicare & Medicaid Services.
Imaging centers are required to collect data on each screening and submit it to a CMS-approved registry.
The decision has been in the works for a while. In 2011, the National Cancer Institute–sponsored National Lung Screening Trial (NLST) showed that people aged 55-74 years with a history of heavy smoking are 20% less likely to die from lung cancer if they are screened with low-dose helical CT instead of standard chest x-ray (N. Engl. J. Med. 2011;365:395-409). Previous studies had shown that screening with standard chest x-rays does not reduce mortality from lung cancer.
Results from NLST and other studies prompted the U.S. Preventive Services Task Force in 2013 to recommend annual low-dose CT screening for adults aged 55-80 years with a 30 pack-year history; the recommendation in turn led to formal requests to CMS for coverage. In proposing coverage, the task force received almost 500 public comments “generally supportive of [expanding] Medicare coverage to include lung cancer screening,” it said.
Low-dose CT is performed at acquisition settings to minimize radiation exposure. For lung cancer screening, CMS is requiring a volumetric CT dose index (CTDIvol) of ≤ 3.0 mGy for standard-size patients – defined to be 5 feet 7 inches tall and approximately 155 pounds – with appropriate reductions or increases for smaller or larger patients.
Effective immediately, Medicare will cover annual lung cancer screening with low-dose CT for certain beneficiaries, according to a Feb. 5 national coverage determination.
To qualify, beneficiaries must be 55-77 years old, have a smoking history of at least a 30 pack-years, exhibit no signs or symptoms of lung cancer, and currently smoke or have quit within 15 years. They also must have a written screening order from their provider.
Coverage includes a counseling visit for shared decision-making, so patients know beforehand the “benefits and harms of screening, follow-up diagnostic testing, over-diagnosis, false positive[s], and total radiation exposure,” among other things, according to the decision memo from the Centers for Medicare & Medicaid Services.
Imaging centers are required to collect data on each screening and submit it to a CMS-approved registry.
The decision has been in the works for a while. In 2011, the National Cancer Institute–sponsored National Lung Screening Trial (NLST) showed that people aged 55-74 years with a history of heavy smoking are 20% less likely to die from lung cancer if they are screened with low-dose helical CT instead of standard chest x-ray (N. Engl. J. Med. 2011;365:395-409). Previous studies had shown that screening with standard chest x-rays does not reduce mortality from lung cancer.
Results from NLST and other studies prompted the U.S. Preventive Services Task Force in 2013 to recommend annual low-dose CT screening for adults aged 55-80 years with a 30 pack-year history; the recommendation in turn led to formal requests to CMS for coverage. In proposing coverage, the task force received almost 500 public comments “generally supportive of [expanding] Medicare coverage to include lung cancer screening,” it said.
Low-dose CT is performed at acquisition settings to minimize radiation exposure. For lung cancer screening, CMS is requiring a volumetric CT dose index (CTDIvol) of ≤ 3.0 mGy for standard-size patients – defined to be 5 feet 7 inches tall and approximately 155 pounds – with appropriate reductions or increases for smaller or larger patients.
Effective immediately, Medicare will cover annual lung cancer screening with low-dose CT for certain beneficiaries, according to a Feb. 5 national coverage determination.
To qualify, beneficiaries must be 55-77 years old, have a smoking history of at least a 30 pack-years, exhibit no signs or symptoms of lung cancer, and currently smoke or have quit within 15 years. They also must have a written screening order from their provider.
Coverage includes a counseling visit for shared decision-making, so patients know beforehand the “benefits and harms of screening, follow-up diagnostic testing, over-diagnosis, false positive[s], and total radiation exposure,” among other things, according to the decision memo from the Centers for Medicare & Medicaid Services.
Imaging centers are required to collect data on each screening and submit it to a CMS-approved registry.
The decision has been in the works for a while. In 2011, the National Cancer Institute–sponsored National Lung Screening Trial (NLST) showed that people aged 55-74 years with a history of heavy smoking are 20% less likely to die from lung cancer if they are screened with low-dose helical CT instead of standard chest x-ray (N. Engl. J. Med. 2011;365:395-409). Previous studies had shown that screening with standard chest x-rays does not reduce mortality from lung cancer.
Results from NLST and other studies prompted the U.S. Preventive Services Task Force in 2013 to recommend annual low-dose CT screening for adults aged 55-80 years with a 30 pack-year history; the recommendation in turn led to formal requests to CMS for coverage. In proposing coverage, the task force received almost 500 public comments “generally supportive of [expanding] Medicare coverage to include lung cancer screening,” it said.
Low-dose CT is performed at acquisition settings to minimize radiation exposure. For lung cancer screening, CMS is requiring a volumetric CT dose index (CTDIvol) of ≤ 3.0 mGy for standard-size patients – defined to be 5 feet 7 inches tall and approximately 155 pounds – with appropriate reductions or increases for smaller or larger patients.