User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
Reslizumab aces pivotal trials in asthma with eosinophilia
HOUSTON – Reslizumab, a next-generation molecular-based asthma therapy, achieved its primary and secondary endpoints and demonstrated favorable safety in patients with moderate to severe asthma and eosinophilia in two pivotal clinical trials.
“We believe that reslizumab is an effective therapy for controlling asthma in patients with elevated blood eosinophils who are inadequately controlled on medium to high dose inhaled corticosteroid–based regimens,” Dr. Mario Castro concluded in presenting the two phase III results at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
The frequency of clinical asthma exacerbations was reduced by more than half in reslizumab-treated patients, compared with controls in the two year-long studies. In addition, the reslizumab group experienced an early improvement in lung function as expressed in forced expiratory volume in 1 second (FEV1) that was sustained throughout the year-long trials, as well as improvements in other measures of asthma control, including quality of life, reported Dr. Castro, professor of pulmonary and critical care medicine and pediatrics at Washington University in St. Louis.
Elevated blood and sputum levels of eosinophils define an asthma phenotype at increased risk for serious asthma exacerbations. Reslizumab is a humanized monoclonal antibody that binds circulating interleukin-5 and prevents binding to the IL-5 receptor, thereby disrupting eosinophil production and function.
Dr. Castro presented two identically designed phase III, double-blind, placebo-controlled, 12-month studies totaling 953 adolescents and adults. They were randomized to intravenous reslizumab at 3 mg/kg or placebo every 4 weeks for a year.
The primary endpoint was frequency of clinical asthma exacerbations (CAEs), an independently adjudicated composite outcome which required an episode featuring an increase in corticosteroids, an asthma-related ER visit or unscheduled office visit, evidence of asthma worsening in the form of at least a 20% drop from baseline in FEV1 or a 30% reduction in peak expiratory flow rate on 2 consecutive days, and worsening clinical symptoms.
Participants averaged two CAEs during the year prior to enrollment. The placebo-treated controls maintained that event rate during the two year-long studies, while the reslizumab-treated patients experienced 50% and 59% reductions relative to controls (P < .0001).
Reslizumab also increased the time to first CAE. In the two trials, 61% and 73% of reslizumab-treated patients didn’t develop a single CAE during 52 weeks, compared with 44% and 52% of controls.
The more CAEs a patient had in the year prior to enrollment, the greater the magnitude of benefit with reslizumab. While the relative risk reduction was 54%, compared with placebo in the combined studies, it climbed to 64% in patients with four or more CAEs in the previous year.
FEV1 improved after the first dose of reslizumab. The benefit – placebo-subtracted gains of 0.126 L in one trial and 0.09 L in the other – was sustained throughout the 52 weeks.
The reslizumab group also outperformed controls in terms of Asthma Control Questionnaire scores and Asthma Quality of Life Questionnaire scores. For example, 74% and 73% of reslizumab-treated patients in the two studies experienced at least a 0.5-point improvement in the AQLQ, which is considered the minimal clinically important difference, compared with 65% and 62% of controls, Dr. Castro continued.
Study discontinuation due to adverse events occurred in 2% of patients on reslizumab, with worsening asthma the No. 1 reason. Two patients on reslizumab experienced anaphylactoid reactions, neither requiring epinephrine. Three percent of reslizumab-treated patients developed low-titer, generally transient antidrug antibodies that didn’t affect eosinophil levels, which plunged with the first dose of reslizumab and stayed low throughout.
Reslizumab is one of a cluster of novel agents in development for severe or treatment-resistant asthma. These are targeted therapies directed at specific patient phenotypes. Biomarkers such as eosinophilia provide guidance as to the specific asthmatic inflammatory pathways involved.
Recent European Respiratory Society/American Thoracic Society guidelines stress the major unmet need for new treatments for severe asthma (Eur. Respir. J. 2014;43:343-73).
In an interview, Dr. James E. Gern, who wasn’t involved in the reslizumab studies, said the various severe asthma phenotypes account for a relatively small proportion of the total asthma population, but a tremendously disproportionate amount of health care utilization.
“These new medications that are coming out are probably going to be indicated for a relatively small number of people. But for those people, it’ll make a huge difference because of the huge burden that severe asthma has on quality of life,” said Dr. Gern, professor of pediatrics at the University of Wisconsin, Madison.
The two pivotal reslizumab studies were sponsored by Teva. Dr. Castro is on the company’s speakers’ bureau and receives research grants from more than a dozen pharmaceutical and device companies as well as from the National Institutes of Health and Centers for Disease Control and Prevention. Simultaneously with Dr. Castro’s presentation at AAAAI, the study results were published online (Lancet Respir. Med. 2015 [doi.org/10.1016/S2213-2600(15)00042-9]).
HOUSTON – Reslizumab, a next-generation molecular-based asthma therapy, achieved its primary and secondary endpoints and demonstrated favorable safety in patients with moderate to severe asthma and eosinophilia in two pivotal clinical trials.
“We believe that reslizumab is an effective therapy for controlling asthma in patients with elevated blood eosinophils who are inadequately controlled on medium to high dose inhaled corticosteroid–based regimens,” Dr. Mario Castro concluded in presenting the two phase III results at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
The frequency of clinical asthma exacerbations was reduced by more than half in reslizumab-treated patients, compared with controls in the two year-long studies. In addition, the reslizumab group experienced an early improvement in lung function as expressed in forced expiratory volume in 1 second (FEV1) that was sustained throughout the year-long trials, as well as improvements in other measures of asthma control, including quality of life, reported Dr. Castro, professor of pulmonary and critical care medicine and pediatrics at Washington University in St. Louis.
Elevated blood and sputum levels of eosinophils define an asthma phenotype at increased risk for serious asthma exacerbations. Reslizumab is a humanized monoclonal antibody that binds circulating interleukin-5 and prevents binding to the IL-5 receptor, thereby disrupting eosinophil production and function.
Dr. Castro presented two identically designed phase III, double-blind, placebo-controlled, 12-month studies totaling 953 adolescents and adults. They were randomized to intravenous reslizumab at 3 mg/kg or placebo every 4 weeks for a year.
The primary endpoint was frequency of clinical asthma exacerbations (CAEs), an independently adjudicated composite outcome which required an episode featuring an increase in corticosteroids, an asthma-related ER visit or unscheduled office visit, evidence of asthma worsening in the form of at least a 20% drop from baseline in FEV1 or a 30% reduction in peak expiratory flow rate on 2 consecutive days, and worsening clinical symptoms.
Participants averaged two CAEs during the year prior to enrollment. The placebo-treated controls maintained that event rate during the two year-long studies, while the reslizumab-treated patients experienced 50% and 59% reductions relative to controls (P < .0001).
Reslizumab also increased the time to first CAE. In the two trials, 61% and 73% of reslizumab-treated patients didn’t develop a single CAE during 52 weeks, compared with 44% and 52% of controls.
The more CAEs a patient had in the year prior to enrollment, the greater the magnitude of benefit with reslizumab. While the relative risk reduction was 54%, compared with placebo in the combined studies, it climbed to 64% in patients with four or more CAEs in the previous year.
FEV1 improved after the first dose of reslizumab. The benefit – placebo-subtracted gains of 0.126 L in one trial and 0.09 L in the other – was sustained throughout the 52 weeks.
The reslizumab group also outperformed controls in terms of Asthma Control Questionnaire scores and Asthma Quality of Life Questionnaire scores. For example, 74% and 73% of reslizumab-treated patients in the two studies experienced at least a 0.5-point improvement in the AQLQ, which is considered the minimal clinically important difference, compared with 65% and 62% of controls, Dr. Castro continued.
Study discontinuation due to adverse events occurred in 2% of patients on reslizumab, with worsening asthma the No. 1 reason. Two patients on reslizumab experienced anaphylactoid reactions, neither requiring epinephrine. Three percent of reslizumab-treated patients developed low-titer, generally transient antidrug antibodies that didn’t affect eosinophil levels, which plunged with the first dose of reslizumab and stayed low throughout.
Reslizumab is one of a cluster of novel agents in development for severe or treatment-resistant asthma. These are targeted therapies directed at specific patient phenotypes. Biomarkers such as eosinophilia provide guidance as to the specific asthmatic inflammatory pathways involved.
Recent European Respiratory Society/American Thoracic Society guidelines stress the major unmet need for new treatments for severe asthma (Eur. Respir. J. 2014;43:343-73).
In an interview, Dr. James E. Gern, who wasn’t involved in the reslizumab studies, said the various severe asthma phenotypes account for a relatively small proportion of the total asthma population, but a tremendously disproportionate amount of health care utilization.
“These new medications that are coming out are probably going to be indicated for a relatively small number of people. But for those people, it’ll make a huge difference because of the huge burden that severe asthma has on quality of life,” said Dr. Gern, professor of pediatrics at the University of Wisconsin, Madison.
The two pivotal reslizumab studies were sponsored by Teva. Dr. Castro is on the company’s speakers’ bureau and receives research grants from more than a dozen pharmaceutical and device companies as well as from the National Institutes of Health and Centers for Disease Control and Prevention. Simultaneously with Dr. Castro’s presentation at AAAAI, the study results were published online (Lancet Respir. Med. 2015 [doi.org/10.1016/S2213-2600(15)00042-9]).
HOUSTON – Reslizumab, a next-generation molecular-based asthma therapy, achieved its primary and secondary endpoints and demonstrated favorable safety in patients with moderate to severe asthma and eosinophilia in two pivotal clinical trials.
“We believe that reslizumab is an effective therapy for controlling asthma in patients with elevated blood eosinophils who are inadequately controlled on medium to high dose inhaled corticosteroid–based regimens,” Dr. Mario Castro concluded in presenting the two phase III results at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
The frequency of clinical asthma exacerbations was reduced by more than half in reslizumab-treated patients, compared with controls in the two year-long studies. In addition, the reslizumab group experienced an early improvement in lung function as expressed in forced expiratory volume in 1 second (FEV1) that was sustained throughout the year-long trials, as well as improvements in other measures of asthma control, including quality of life, reported Dr. Castro, professor of pulmonary and critical care medicine and pediatrics at Washington University in St. Louis.
Elevated blood and sputum levels of eosinophils define an asthma phenotype at increased risk for serious asthma exacerbations. Reslizumab is a humanized monoclonal antibody that binds circulating interleukin-5 and prevents binding to the IL-5 receptor, thereby disrupting eosinophil production and function.
Dr. Castro presented two identically designed phase III, double-blind, placebo-controlled, 12-month studies totaling 953 adolescents and adults. They were randomized to intravenous reslizumab at 3 mg/kg or placebo every 4 weeks for a year.
The primary endpoint was frequency of clinical asthma exacerbations (CAEs), an independently adjudicated composite outcome which required an episode featuring an increase in corticosteroids, an asthma-related ER visit or unscheduled office visit, evidence of asthma worsening in the form of at least a 20% drop from baseline in FEV1 or a 30% reduction in peak expiratory flow rate on 2 consecutive days, and worsening clinical symptoms.
Participants averaged two CAEs during the year prior to enrollment. The placebo-treated controls maintained that event rate during the two year-long studies, while the reslizumab-treated patients experienced 50% and 59% reductions relative to controls (P < .0001).
Reslizumab also increased the time to first CAE. In the two trials, 61% and 73% of reslizumab-treated patients didn’t develop a single CAE during 52 weeks, compared with 44% and 52% of controls.
The more CAEs a patient had in the year prior to enrollment, the greater the magnitude of benefit with reslizumab. While the relative risk reduction was 54%, compared with placebo in the combined studies, it climbed to 64% in patients with four or more CAEs in the previous year.
FEV1 improved after the first dose of reslizumab. The benefit – placebo-subtracted gains of 0.126 L in one trial and 0.09 L in the other – was sustained throughout the 52 weeks.
The reslizumab group also outperformed controls in terms of Asthma Control Questionnaire scores and Asthma Quality of Life Questionnaire scores. For example, 74% and 73% of reslizumab-treated patients in the two studies experienced at least a 0.5-point improvement in the AQLQ, which is considered the minimal clinically important difference, compared with 65% and 62% of controls, Dr. Castro continued.
Study discontinuation due to adverse events occurred in 2% of patients on reslizumab, with worsening asthma the No. 1 reason. Two patients on reslizumab experienced anaphylactoid reactions, neither requiring epinephrine. Three percent of reslizumab-treated patients developed low-titer, generally transient antidrug antibodies that didn’t affect eosinophil levels, which plunged with the first dose of reslizumab and stayed low throughout.
Reslizumab is one of a cluster of novel agents in development for severe or treatment-resistant asthma. These are targeted therapies directed at specific patient phenotypes. Biomarkers such as eosinophilia provide guidance as to the specific asthmatic inflammatory pathways involved.
Recent European Respiratory Society/American Thoracic Society guidelines stress the major unmet need for new treatments for severe asthma (Eur. Respir. J. 2014;43:343-73).
In an interview, Dr. James E. Gern, who wasn’t involved in the reslizumab studies, said the various severe asthma phenotypes account for a relatively small proportion of the total asthma population, but a tremendously disproportionate amount of health care utilization.
“These new medications that are coming out are probably going to be indicated for a relatively small number of people. But for those people, it’ll make a huge difference because of the huge burden that severe asthma has on quality of life,” said Dr. Gern, professor of pediatrics at the University of Wisconsin, Madison.
The two pivotal reslizumab studies were sponsored by Teva. Dr. Castro is on the company’s speakers’ bureau and receives research grants from more than a dozen pharmaceutical and device companies as well as from the National Institutes of Health and Centers for Disease Control and Prevention. Simultaneously with Dr. Castro’s presentation at AAAAI, the study results were published online (Lancet Respir. Med. 2015 [doi.org/10.1016/S2213-2600(15)00042-9]).
AT 2015 AAAAI ANNUAL MEETING
Key clinical point: A monoclonal antibody against interleukin-5 significantly reduced the frequency of clinical asthma exacerbations in an important subset of asthma patients.
Major finding: Intravenous reslizumab given every 4 weeks reduced the annual rate of clinical asthma exacerbations by 50% and 59%, compared with placebo in two phase III trials.
Data source: The two randomized, double-blind, placebo-controlled, 1-year pivotal trials totaled 953 patients with moderate to severe asthma with elevated blood eosinophils inadequately controlled using inhaled corticosteroid–based regimens.
Disclosures: The studies were sponsored by Teva. The presenter is on the company’s speakers’ bureau and receives research grants from more than a dozen pharmaceutical and device companies.
Respiratory harm reversal seen in asthmatic smokers on e-cigarettes
HOUSTON – Asthmatic smokers who switched to electronic cigarettes showed evidence suggestive of respiratory harm reversal in a retrospective pilot study.
“Electronic cigarette use improves respiratory physiology and subjective asthma outcomes in asthmatic smokers. E-cigarettes are a safer alternative to conventional cigarettes in this vulnerable population,” Dr. Cristina Russo declared at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
She said that her small retrospective study is the first to examine the respiratory health impact of a switch to e-cigarettes by asthmatic smokers.
Every one of the objective and subjective measures of asthma status evaluated in the study showed statistically significant improvement 1 year after patients adopted e-cigarettes, and the e-cigarette users’ consumption of conventional cigarettes dropped precipitously, reported Dr. Russo of the University of Catania (Italy).
She and her colleagues in the university asthma clinic have taken to suggesting the use of battery-powered e-cigarettes to their asthmatic smokers who haven’t benefited from or aren’t interested in trying the more conventional approaches to smoking cessation or reduction, including medications. While abstinence from cigarette smoking is best, the available evidence indicates e-cigarettes are at least 95% less harmful than conventional cigarettes in the general population, she said.
The study included 18 smokers with mild to moderate asthma who switched to e-cigarettes and underwent spirometry and other testing at baseline and 6 and 12 months of follow-up. Ten patients switched over to e-cigarettes exclusively, while the other 8 used both conventional and e-cigarettes.
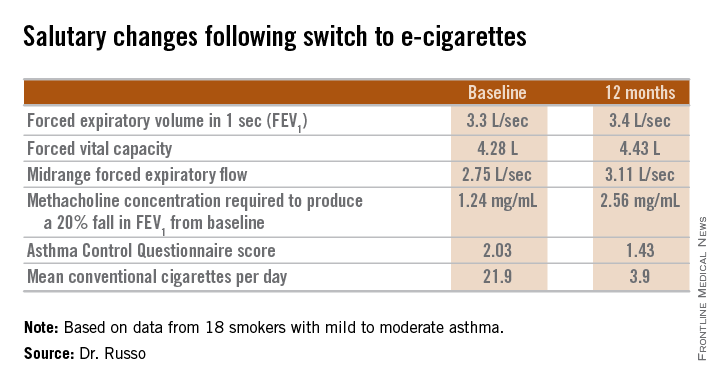
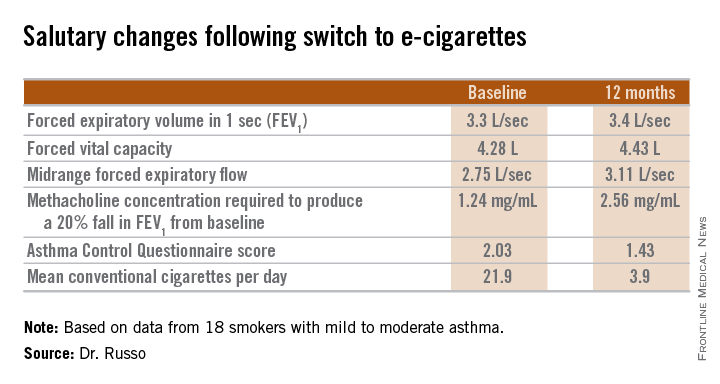
Among the highlights: The mid-range forced expiratory flow (25%-75%) showed a major, clinically important improvement, increasing from 2.75 L/sec to 3.11 L/sec. And patients’ mean self-reported conventional cigarette consumption dropped from 21.9 per day at baseline to 5 at 6 months and 3.9 per day at 12 months.
Among the group at large, no significant change was seen in the frequency of asthma exacerbations resulting in hospitalization. However, among the frequent exacerbators – the six patients with two or more exacerbations during the 6 months prior to baseline – exacerbation frequency was cut in half both 6 and 12 months following the switch to e-cigarettes.
Dr. Russo’s presentation sparked vigorous audience discussion. Several physicians cited a Centers for Disease Control and Prevention warning about the unknowns regarding e-cigarette safety, and one allergist declared he didn’t think physicians should ever encourage patients to smoke anything. But others defended the “lesser of two evils” approach adopted by Dr. Russo and coworkers.
Dr. Russo noted that the prevalence of smoking among asthma patients is similar to that of the general population. She called smoking and asthma “a dangerous liaison.” Smoking accelerates asthma patients’ decline in lung function, worsens persistent airways obstruction, and increases insensitivity to corticosteroids.
Her study was supported by a university grant and the Italian League Against Smoking. She reported having no financial conflicts.
HOUSTON – Asthmatic smokers who switched to electronic cigarettes showed evidence suggestive of respiratory harm reversal in a retrospective pilot study.
“Electronic cigarette use improves respiratory physiology and subjective asthma outcomes in asthmatic smokers. E-cigarettes are a safer alternative to conventional cigarettes in this vulnerable population,” Dr. Cristina Russo declared at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.

She said that her small retrospective study is the first to examine the respiratory health impact of a switch to e-cigarettes by asthmatic smokers.
Every one of the objective and subjective measures of asthma status evaluated in the study showed statistically significant improvement 1 year after patients adopted e-cigarettes, and the e-cigarette users’ consumption of conventional cigarettes dropped precipitously, reported Dr. Russo of the University of Catania (Italy).
She and her colleagues in the university asthma clinic have taken to suggesting the use of battery-powered e-cigarettes to their asthmatic smokers who haven’t benefited from or aren’t interested in trying the more conventional approaches to smoking cessation or reduction, including medications. While abstinence from cigarette smoking is best, the available evidence indicates e-cigarettes are at least 95% less harmful than conventional cigarettes in the general population, she said.
The study included 18 smokers with mild to moderate asthma who switched to e-cigarettes and underwent spirometry and other testing at baseline and 6 and 12 months of follow-up. Ten patients switched over to e-cigarettes exclusively, while the other 8 used both conventional and e-cigarettes.
Among the highlights: The mid-range forced expiratory flow (25%-75%) showed a major, clinically important improvement, increasing from 2.75 L/sec to 3.11 L/sec. And patients’ mean self-reported conventional cigarette consumption dropped from 21.9 per day at baseline to 5 at 6 months and 3.9 per day at 12 months.
Among the group at large, no significant change was seen in the frequency of asthma exacerbations resulting in hospitalization. However, among the frequent exacerbators – the six patients with two or more exacerbations during the 6 months prior to baseline – exacerbation frequency was cut in half both 6 and 12 months following the switch to e-cigarettes.
Dr. Russo’s presentation sparked vigorous audience discussion. Several physicians cited a Centers for Disease Control and Prevention warning about the unknowns regarding e-cigarette safety, and one allergist declared he didn’t think physicians should ever encourage patients to smoke anything. But others defended the “lesser of two evils” approach adopted by Dr. Russo and coworkers.
Dr. Russo noted that the prevalence of smoking among asthma patients is similar to that of the general population. She called smoking and asthma “a dangerous liaison.” Smoking accelerates asthma patients’ decline in lung function, worsens persistent airways obstruction, and increases insensitivity to corticosteroids.
Her study was supported by a university grant and the Italian League Against Smoking. She reported having no financial conflicts.
HOUSTON – Asthmatic smokers who switched to electronic cigarettes showed evidence suggestive of respiratory harm reversal in a retrospective pilot study.
“Electronic cigarette use improves respiratory physiology and subjective asthma outcomes in asthmatic smokers. E-cigarettes are a safer alternative to conventional cigarettes in this vulnerable population,” Dr. Cristina Russo declared at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.

She said that her small retrospective study is the first to examine the respiratory health impact of a switch to e-cigarettes by asthmatic smokers.
Every one of the objective and subjective measures of asthma status evaluated in the study showed statistically significant improvement 1 year after patients adopted e-cigarettes, and the e-cigarette users’ consumption of conventional cigarettes dropped precipitously, reported Dr. Russo of the University of Catania (Italy).
She and her colleagues in the university asthma clinic have taken to suggesting the use of battery-powered e-cigarettes to their asthmatic smokers who haven’t benefited from or aren’t interested in trying the more conventional approaches to smoking cessation or reduction, including medications. While abstinence from cigarette smoking is best, the available evidence indicates e-cigarettes are at least 95% less harmful than conventional cigarettes in the general population, she said.
The study included 18 smokers with mild to moderate asthma who switched to e-cigarettes and underwent spirometry and other testing at baseline and 6 and 12 months of follow-up. Ten patients switched over to e-cigarettes exclusively, while the other 8 used both conventional and e-cigarettes.
Among the highlights: The mid-range forced expiratory flow (25%-75%) showed a major, clinically important improvement, increasing from 2.75 L/sec to 3.11 L/sec. And patients’ mean self-reported conventional cigarette consumption dropped from 21.9 per day at baseline to 5 at 6 months and 3.9 per day at 12 months.
Among the group at large, no significant change was seen in the frequency of asthma exacerbations resulting in hospitalization. However, among the frequent exacerbators – the six patients with two or more exacerbations during the 6 months prior to baseline – exacerbation frequency was cut in half both 6 and 12 months following the switch to e-cigarettes.
Dr. Russo’s presentation sparked vigorous audience discussion. Several physicians cited a Centers for Disease Control and Prevention warning about the unknowns regarding e-cigarette safety, and one allergist declared he didn’t think physicians should ever encourage patients to smoke anything. But others defended the “lesser of two evils” approach adopted by Dr. Russo and coworkers.
Dr. Russo noted that the prevalence of smoking among asthma patients is similar to that of the general population. She called smoking and asthma “a dangerous liaison.” Smoking accelerates asthma patients’ decline in lung function, worsens persistent airways obstruction, and increases insensitivity to corticosteroids.
Her study was supported by a university grant and the Italian League Against Smoking. She reported having no financial conflicts.
AT 2015 AAAAI ANNUAL MEETING
Key clinical point: Asthmatic smokers who adopted physician advice to switch to electronic cigarettes showed significant 1-year improvements in lung function, methacholine-provoked airway hyperresponsiveness, and asthma-related quality of life.
Major finding: Self-reported daily consumption of conventional cigarettes by asthmatic smokers who switched to e-cigarettes fell from a mean of 21.9 per day at baseline to 5.0 at 6 months and 3.9 at 12 months of follow-up.
Data source: This retrospective pilot study included 18 asthmatic smokers who switched to e-cigarettes.
Disclosures: The study was supported by a university grant and the Italian League Against Smoking. The presenter reported having no financial conflicts.
Early consumption of peanuts can induce tolerance in high-risk children
HOUSTON – The introduction of peanuts at an early age to children who are more highly predisposed to having a peanut allergy can induce tolerance of peanuts and thereby significantly decrease the likelihood of developing a sustained allergy.
This finding from the Learning Early about Peanut Allergy (LEAP) study was presented at the annual meeting of the American Academy of Allergy, Asthma, and Immunology and simultaneously published by the New England Journal of Medicine. Investigators enrolled 640 infants aged 4-11 months between December 2006 and May 6, 2009, all of whom were given skin-prick tests at baseline to determine existing peanut allergies, and randomized them into cohorts that would either consume or avoid peanuts until they reached 60 months of age (N. Engl. J. Med. 2015 Feb. 23 [doi:10.1056/NEJMoa1414850]).
Of the 530 children who had negative skin-prick test results, the prevalence of peanut allergy after 60 months was 13.7% in the avoidance cohort and 1.9% in the consumption cohort (P < .001). Of 98 children who had positive skin-prick test results, 35% of the avoidance cohort had peanut allergy after 60 months, while only 11% of the consumption cohort had the allergy (P = .004). Several children who were initially enrolled and randomized either dropped out or were excluded because of inadequate adherence to treatment or missing data.
Outcomes were measured by placing children in a double-blind, placebo-controlled study that had them consume a cumulative total of 9.4 g of peanut protein to determine whether or not tolerance to peanuts had been effectively induced. All children consumed peanuts by eating peanut butter or a peanut-based snack called Bamba, which was provided for the study at a discounted rate. Children whose skin-prick tests showed wheals of larger than 4 mm in diameter were excluded for presumed existing peanut allergy.
“If you enroll children after 11 months of age, you get about twice the rate of [already allergic] children, with a steady decline as [age] goes down,” corresponding author Dr. Gideon Lack, head of the department of pediatric allergy at King’s College, London, said in a press conference following presentation of the study. “But of children younger than 5 months of age, none of them were peanut allergic, so that would suggest that timing here is key: There is a narrow window of opportunity to intervene if you want to be effective.”
Investigators also noted that children who consumed peanuts had increased levels of IgG4 antibody, while those told to avoid peanuts had higher levels of IgE antibody. They also found that development of peanut allergy was generally associated with subjects who both displayed larger wheals on the skin-prick test and had a lower ratio of IgG4:IgE antibodies. Skin-prick tests that yielded wheals of 1-2 mm in diameter were considered indicative of early risk for peanut allergy.
There was no significant difference in the rates of adverse events or hospitalizations between the consumption and avoidance cohort, but 99% of subjects in each group did experience at least one adverse event: 4,527 in the consumption cohort and 4,287 in the avoidance cohort (P = .02). Infants who consumed peanuts had higher instances of upper respiratory tract infection, viral skin infection, gastroenteritis, urticaria, and conjunctivitis, but events mostly ranged from mild to moderate and their severity was not significantly different from similar incidents reported in the avoidance cohort.
Hospitalization rates also were low, with 52 children in the avoidance cohort and 50 children who consumed peanuts being admitted over the study interval, or 16.2% and 15.7%, respectively (P = .86). Rates of serious adverse effects also were not significantly different between the two cohorts, with 101 children who avoided peanuts and 89 who consumed peanuts experiencing such events (P = .41).
Adequate adherence to treatment for the children randomized into the peanut-avoidance group was defined as eating less than 0.2 g of peanut protein on any single occasion and no more than 0.5 g over the course of a single week in the first 24 months of life. For children in the consumption cohort, adequate adherence meant consuming at least 2 g of peanut protein on at least one occasion in both the first and second years of life, and at least 3 g of peanut protein weekly for at least half the number of weeks for which data was collected.
“Whether or not these benefits can be sustained, we will find out in 1 year,” Dr. Lack said, adding that “all the children who had been consuming peanut have stopped [for] 1 year, and we will see if peanut allergy persists after 1 year of cessation of consumption.” He predicted that the findings would most likely produce a mixed response, with some children relapsing while others maintain sustained tolerance.
The LEAP study was supported by grants from the National Institute of Allergy and Infectious Diseases, Food Allergy and Research Education, the Medical Research Council and Asthma UK, the UK Department of Health’s National Institute for Health Research, the National Peanut Board, and the UK Food Standards Agency. Dr. Lack disclosed financial affiliations with DBV Technologies, and coauthor Dr. Helen Brough disclosed receiving financial support from Fare and Action Medical Research, along with study materials from Stallergenes, Thermo Scientific, and Meridian Foods.
HOUSTON – The introduction of peanuts at an early age to children who are more highly predisposed to having a peanut allergy can induce tolerance of peanuts and thereby significantly decrease the likelihood of developing a sustained allergy.
This finding from the Learning Early about Peanut Allergy (LEAP) study was presented at the annual meeting of the American Academy of Allergy, Asthma, and Immunology and simultaneously published by the New England Journal of Medicine. Investigators enrolled 640 infants aged 4-11 months between December 2006 and May 6, 2009, all of whom were given skin-prick tests at baseline to determine existing peanut allergies, and randomized them into cohorts that would either consume or avoid peanuts until they reached 60 months of age (N. Engl. J. Med. 2015 Feb. 23 [doi:10.1056/NEJMoa1414850]).
Of the 530 children who had negative skin-prick test results, the prevalence of peanut allergy after 60 months was 13.7% in the avoidance cohort and 1.9% in the consumption cohort (P < .001). Of 98 children who had positive skin-prick test results, 35% of the avoidance cohort had peanut allergy after 60 months, while only 11% of the consumption cohort had the allergy (P = .004). Several children who were initially enrolled and randomized either dropped out or were excluded because of inadequate adherence to treatment or missing data.
Outcomes were measured by placing children in a double-blind, placebo-controlled study that had them consume a cumulative total of 9.4 g of peanut protein to determine whether or not tolerance to peanuts had been effectively induced. All children consumed peanuts by eating peanut butter or a peanut-based snack called Bamba, which was provided for the study at a discounted rate. Children whose skin-prick tests showed wheals of larger than 4 mm in diameter were excluded for presumed existing peanut allergy.
“If you enroll children after 11 months of age, you get about twice the rate of [already allergic] children, with a steady decline as [age] goes down,” corresponding author Dr. Gideon Lack, head of the department of pediatric allergy at King’s College, London, said in a press conference following presentation of the study. “But of children younger than 5 months of age, none of them were peanut allergic, so that would suggest that timing here is key: There is a narrow window of opportunity to intervene if you want to be effective.”
Investigators also noted that children who consumed peanuts had increased levels of IgG4 antibody, while those told to avoid peanuts had higher levels of IgE antibody. They also found that development of peanut allergy was generally associated with subjects who both displayed larger wheals on the skin-prick test and had a lower ratio of IgG4:IgE antibodies. Skin-prick tests that yielded wheals of 1-2 mm in diameter were considered indicative of early risk for peanut allergy.
There was no significant difference in the rates of adverse events or hospitalizations between the consumption and avoidance cohort, but 99% of subjects in each group did experience at least one adverse event: 4,527 in the consumption cohort and 4,287 in the avoidance cohort (P = .02). Infants who consumed peanuts had higher instances of upper respiratory tract infection, viral skin infection, gastroenteritis, urticaria, and conjunctivitis, but events mostly ranged from mild to moderate and their severity was not significantly different from similar incidents reported in the avoidance cohort.
Hospitalization rates also were low, with 52 children in the avoidance cohort and 50 children who consumed peanuts being admitted over the study interval, or 16.2% and 15.7%, respectively (P = .86). Rates of serious adverse effects also were not significantly different between the two cohorts, with 101 children who avoided peanuts and 89 who consumed peanuts experiencing such events (P = .41).
Adequate adherence to treatment for the children randomized into the peanut-avoidance group was defined as eating less than 0.2 g of peanut protein on any single occasion and no more than 0.5 g over the course of a single week in the first 24 months of life. For children in the consumption cohort, adequate adherence meant consuming at least 2 g of peanut protein on at least one occasion in both the first and second years of life, and at least 3 g of peanut protein weekly for at least half the number of weeks for which data was collected.
“Whether or not these benefits can be sustained, we will find out in 1 year,” Dr. Lack said, adding that “all the children who had been consuming peanut have stopped [for] 1 year, and we will see if peanut allergy persists after 1 year of cessation of consumption.” He predicted that the findings would most likely produce a mixed response, with some children relapsing while others maintain sustained tolerance.
The LEAP study was supported by grants from the National Institute of Allergy and Infectious Diseases, Food Allergy and Research Education, the Medical Research Council and Asthma UK, the UK Department of Health’s National Institute for Health Research, the National Peanut Board, and the UK Food Standards Agency. Dr. Lack disclosed financial affiliations with DBV Technologies, and coauthor Dr. Helen Brough disclosed receiving financial support from Fare and Action Medical Research, along with study materials from Stallergenes, Thermo Scientific, and Meridian Foods.
HOUSTON – The introduction of peanuts at an early age to children who are more highly predisposed to having a peanut allergy can induce tolerance of peanuts and thereby significantly decrease the likelihood of developing a sustained allergy.
This finding from the Learning Early about Peanut Allergy (LEAP) study was presented at the annual meeting of the American Academy of Allergy, Asthma, and Immunology and simultaneously published by the New England Journal of Medicine. Investigators enrolled 640 infants aged 4-11 months between December 2006 and May 6, 2009, all of whom were given skin-prick tests at baseline to determine existing peanut allergies, and randomized them into cohorts that would either consume or avoid peanuts until they reached 60 months of age (N. Engl. J. Med. 2015 Feb. 23 [doi:10.1056/NEJMoa1414850]).
Of the 530 children who had negative skin-prick test results, the prevalence of peanut allergy after 60 months was 13.7% in the avoidance cohort and 1.9% in the consumption cohort (P < .001). Of 98 children who had positive skin-prick test results, 35% of the avoidance cohort had peanut allergy after 60 months, while only 11% of the consumption cohort had the allergy (P = .004). Several children who were initially enrolled and randomized either dropped out or were excluded because of inadequate adherence to treatment or missing data.
Outcomes were measured by placing children in a double-blind, placebo-controlled study that had them consume a cumulative total of 9.4 g of peanut protein to determine whether or not tolerance to peanuts had been effectively induced. All children consumed peanuts by eating peanut butter or a peanut-based snack called Bamba, which was provided for the study at a discounted rate. Children whose skin-prick tests showed wheals of larger than 4 mm in diameter were excluded for presumed existing peanut allergy.
“If you enroll children after 11 months of age, you get about twice the rate of [already allergic] children, with a steady decline as [age] goes down,” corresponding author Dr. Gideon Lack, head of the department of pediatric allergy at King’s College, London, said in a press conference following presentation of the study. “But of children younger than 5 months of age, none of them were peanut allergic, so that would suggest that timing here is key: There is a narrow window of opportunity to intervene if you want to be effective.”
Investigators also noted that children who consumed peanuts had increased levels of IgG4 antibody, while those told to avoid peanuts had higher levels of IgE antibody. They also found that development of peanut allergy was generally associated with subjects who both displayed larger wheals on the skin-prick test and had a lower ratio of IgG4:IgE antibodies. Skin-prick tests that yielded wheals of 1-2 mm in diameter were considered indicative of early risk for peanut allergy.
There was no significant difference in the rates of adverse events or hospitalizations between the consumption and avoidance cohort, but 99% of subjects in each group did experience at least one adverse event: 4,527 in the consumption cohort and 4,287 in the avoidance cohort (P = .02). Infants who consumed peanuts had higher instances of upper respiratory tract infection, viral skin infection, gastroenteritis, urticaria, and conjunctivitis, but events mostly ranged from mild to moderate and their severity was not significantly different from similar incidents reported in the avoidance cohort.
Hospitalization rates also were low, with 52 children in the avoidance cohort and 50 children who consumed peanuts being admitted over the study interval, or 16.2% and 15.7%, respectively (P = .86). Rates of serious adverse effects also were not significantly different between the two cohorts, with 101 children who avoided peanuts and 89 who consumed peanuts experiencing such events (P = .41).
Adequate adherence to treatment for the children randomized into the peanut-avoidance group was defined as eating less than 0.2 g of peanut protein on any single occasion and no more than 0.5 g over the course of a single week in the first 24 months of life. For children in the consumption cohort, adequate adherence meant consuming at least 2 g of peanut protein on at least one occasion in both the first and second years of life, and at least 3 g of peanut protein weekly for at least half the number of weeks for which data was collected.
“Whether or not these benefits can be sustained, we will find out in 1 year,” Dr. Lack said, adding that “all the children who had been consuming peanut have stopped [for] 1 year, and we will see if peanut allergy persists after 1 year of cessation of consumption.” He predicted that the findings would most likely produce a mixed response, with some children relapsing while others maintain sustained tolerance.
The LEAP study was supported by grants from the National Institute of Allergy and Infectious Diseases, Food Allergy and Research Education, the Medical Research Council and Asthma UK, the UK Department of Health’s National Institute for Health Research, the National Peanut Board, and the UK Food Standards Agency. Dr. Lack disclosed financial affiliations with DBV Technologies, and coauthor Dr. Helen Brough disclosed receiving financial support from Fare and Action Medical Research, along with study materials from Stallergenes, Thermo Scientific, and Meridian Foods.
AT 2015 AAAAI ANNUAL MEETING
Key clinical point: Early introduction of peanuts into the diets of children at high risk for peanut allergy can significantly decrease the possibility of peanut allergy development.
Major finding: Of 530 infants who initially tested negative for peanut allergy, prevalence of said allergy after 60 months was 13.7% in the avoidance cohort and 1.9% in the consumption cohort (P < .001); of the 98 infants who initially tested positive, prevalence after 60 months was 35% in the avoidance cohort and 11% in the consumption cohort (P = .004).
Data source: Randomized cohort study of 640 children enrolled at ages 4-11 months between December 2006 and May 2009.
Disclosures: The LEAP study was funded by several health care agencies in the United States and United Kingdom. Corresponding author Dr. Gideon Lack disclosed financial affiliations with DBV Technologies, and coauthor Dr. Helen Brough disclosed receiving financial support from Food Allergy and Research Education and Action Medical Research, along with study materials from Stallergenes, Thermo Scientific, and Meridian Foods.
Lung cancer screening criteria may need changes
Fewer patients diagnosed with lung cancer are meeting the U.S. Preventive Services Task Force criteria for screening with low-dose computed tomography (LDCT).
Screening criteria may need to be changed, since fewer American adults now have a smoking history of 30-pack years and have quit within the last 15 years, according to Dr. Ping Yang of the division of epidemiology at the Mayo Clinic, Rochester, Minn., and her colleagues.
A retrospective analysis of patients with pathologically confirmed lung cancer in Olmsted County, Minn., between 1984 and 2011 found that in 1984-1990, 57% of patients with diagnosed lung cancer met USPSTF screening guidelines. By 2005-2011, the figure had dropped significantly to 43%.
The proportion of women who would have been eligible under the criteria decreased from 52% to 37% in the same time period; the proportion of men had dropped from 60% to 50%, they found.
“More sensitive screening criteria may need to be identified while balancing the potential harm from computed tomography,” Dr. Yang and her colleagues wrote (JAMA 2015 Feb. 24;313:853-5).
Lung cancer incidence trends in this study were comparable with Surveillance, Epidemiology and End Results data, but may not be generalizable to the U.S. population, they added.The study was funded by the National Institutes of Health and the Mayo Clinic Foundation. The authors reported no relevant conflicts of interest.
Fewer patients diagnosed with lung cancer are meeting the U.S. Preventive Services Task Force criteria for screening with low-dose computed tomography (LDCT).
Screening criteria may need to be changed, since fewer American adults now have a smoking history of 30-pack years and have quit within the last 15 years, according to Dr. Ping Yang of the division of epidemiology at the Mayo Clinic, Rochester, Minn., and her colleagues.
A retrospective analysis of patients with pathologically confirmed lung cancer in Olmsted County, Minn., between 1984 and 2011 found that in 1984-1990, 57% of patients with diagnosed lung cancer met USPSTF screening guidelines. By 2005-2011, the figure had dropped significantly to 43%.
The proportion of women who would have been eligible under the criteria decreased from 52% to 37% in the same time period; the proportion of men had dropped from 60% to 50%, they found.
“More sensitive screening criteria may need to be identified while balancing the potential harm from computed tomography,” Dr. Yang and her colleagues wrote (JAMA 2015 Feb. 24;313:853-5).
Lung cancer incidence trends in this study were comparable with Surveillance, Epidemiology and End Results data, but may not be generalizable to the U.S. population, they added.The study was funded by the National Institutes of Health and the Mayo Clinic Foundation. The authors reported no relevant conflicts of interest.
Fewer patients diagnosed with lung cancer are meeting the U.S. Preventive Services Task Force criteria for screening with low-dose computed tomography (LDCT).
Screening criteria may need to be changed, since fewer American adults now have a smoking history of 30-pack years and have quit within the last 15 years, according to Dr. Ping Yang of the division of epidemiology at the Mayo Clinic, Rochester, Minn., and her colleagues.
A retrospective analysis of patients with pathologically confirmed lung cancer in Olmsted County, Minn., between 1984 and 2011 found that in 1984-1990, 57% of patients with diagnosed lung cancer met USPSTF screening guidelines. By 2005-2011, the figure had dropped significantly to 43%.
The proportion of women who would have been eligible under the criteria decreased from 52% to 37% in the same time period; the proportion of men had dropped from 60% to 50%, they found.
“More sensitive screening criteria may need to be identified while balancing the potential harm from computed tomography,” Dr. Yang and her colleagues wrote (JAMA 2015 Feb. 24;313:853-5).
Lung cancer incidence trends in this study were comparable with Surveillance, Epidemiology and End Results data, but may not be generalizable to the U.S. population, they added.The study was funded by the National Institutes of Health and the Mayo Clinic Foundation. The authors reported no relevant conflicts of interest.
FROM JAMA
Key clinical point: Changes in smoking habits may call for a revision of USPSTF lung cancer screening criteria.
Main finding: Between 2005 and 2011 only 37% of females and 50% of males diagnosed with lung cancer would have met the criteria for screening with LDCT.
Source: Retrospective analysis of 1,351 patients with lung cancer in Olmsted County, Minn., between 1984 and 2011.
Disclosures: The study was funded by NIH and the Mayo Clinic Foundation. The authors reported no relevant conflicts of interest.
Breastfeeding-related changes in gut bacteria protect against childhood allergy
HOUSTON – Findings from a cohort study of mothers and babies point to breastfeeding as influencing infants’ gut microbiome in a way that protects them from developing allergic disease.
In findings presented at the annual meeting of the American Academy of Allergy, Asthma, and Immunology, epidemiologist Christine Cole Johnson, Ph.D., of the Henry Ford Health System in Detroit described a correlation between certain maternal and birth characteristics that had previously been shown to relate to allergic response, and measurable differences in the bacterial profiles of the study infants’ stools.
Using data from the WHEALS (Wayne County [Michigan] Health, Environment, Allergy, and Asthma Longitudinal Study) cohort, Dr. Johnson and her colleagues looked at stool samples from 298 children at 1 and 6 months of age. The investigators also collected dust samples from the infants’ homes, obtained medical records, and conducted interviews with the families (J. Allergy Clin. Immunol. 2015 [http://dx.doi.org/10.1016/j.jaci.2014.12.1443]).
The presence of household pets, the body mass index of mothers before delivery, the mode of delivery, household smoke exposure, marital status, income, race, and maternal education were all found to be significantly correlated to different gut bacterial profiles.
“Environmental and lifestyle variables that we’ve been working on related to childhood asthma and allergy seem to be associated – at least in our study – with the child’s gut microbiome,” Dr. Johnson said. “These factors vary a lot by whether those stool samples were collected at 1 or 6 months,” she said, noting that the infant gut microbiome is shaped rapidly in the first year.
But, at both 1 and 6 months of age, breastfeeding was seen as the dominant factor influencing gut bacterial composition.
At 6 months, breastfed infants had bacterial profiles showing overwhelming dominance of Bifidobacteriaceae, but vastly lower levels of other families of bacteria, notably Lachnospiraceae, which were prominent in the guts of non-breastfed babies.
In a related study that used the same cohort to explore whether the influence of breastfeeding on gut bacterial composition correlated to the development of allergic symptoms at 4 years old, Alexandra R. Sitarik, also of the Henry Ford Health System in Detroit, reported that babies being breastfed at 1 month of age had a significantly lower risk of developing allergiclike symptoms to pets by age 4 years (P = .028) (J. Allergy Clin. Immunol. 2015 [http://dx.doi.org/10.1016/j.jaci.2014.12.1444]).
Both breastfeeding and allergiclike response to pets were significantly related to compositional variation in gut microbiome (P < .001 and P = .023, respectively), Ms. Sitarik reported.
Of the 109 types of bacteria significantly associated with both breastfeeding and allergiclike response to pets, 71% were negatively associated with breastfeeding but positively associated with allergiclike response to pets.
This subset of risk-increasing bacteria suppressed by breastfeeding were predominantly members of the family Lachnospiraceae, the researchers found.
Lachnospiraceae are common adult gut colonizers, Ms. Sitarik said, and as people age the relative abundance of Lachnospiraceae increases. “What we think might be happening in terms of the gut microbiome is that maybe breastfeeding is preventing this premature shift into adulthood,” she said.
Dr. Johnson and Ms. Sitarik reported having no financial disclosures.
HOUSTON – Findings from a cohort study of mothers and babies point to breastfeeding as influencing infants’ gut microbiome in a way that protects them from developing allergic disease.
In findings presented at the annual meeting of the American Academy of Allergy, Asthma, and Immunology, epidemiologist Christine Cole Johnson, Ph.D., of the Henry Ford Health System in Detroit described a correlation between certain maternal and birth characteristics that had previously been shown to relate to allergic response, and measurable differences in the bacterial profiles of the study infants’ stools.
Using data from the WHEALS (Wayne County [Michigan] Health, Environment, Allergy, and Asthma Longitudinal Study) cohort, Dr. Johnson and her colleagues looked at stool samples from 298 children at 1 and 6 months of age. The investigators also collected dust samples from the infants’ homes, obtained medical records, and conducted interviews with the families (J. Allergy Clin. Immunol. 2015 [http://dx.doi.org/10.1016/j.jaci.2014.12.1443]).
The presence of household pets, the body mass index of mothers before delivery, the mode of delivery, household smoke exposure, marital status, income, race, and maternal education were all found to be significantly correlated to different gut bacterial profiles.
“Environmental and lifestyle variables that we’ve been working on related to childhood asthma and allergy seem to be associated – at least in our study – with the child’s gut microbiome,” Dr. Johnson said. “These factors vary a lot by whether those stool samples were collected at 1 or 6 months,” she said, noting that the infant gut microbiome is shaped rapidly in the first year.
But, at both 1 and 6 months of age, breastfeeding was seen as the dominant factor influencing gut bacterial composition.
At 6 months, breastfed infants had bacterial profiles showing overwhelming dominance of Bifidobacteriaceae, but vastly lower levels of other families of bacteria, notably Lachnospiraceae, which were prominent in the guts of non-breastfed babies.
In a related study that used the same cohort to explore whether the influence of breastfeeding on gut bacterial composition correlated to the development of allergic symptoms at 4 years old, Alexandra R. Sitarik, also of the Henry Ford Health System in Detroit, reported that babies being breastfed at 1 month of age had a significantly lower risk of developing allergiclike symptoms to pets by age 4 years (P = .028) (J. Allergy Clin. Immunol. 2015 [http://dx.doi.org/10.1016/j.jaci.2014.12.1444]).
Both breastfeeding and allergiclike response to pets were significantly related to compositional variation in gut microbiome (P < .001 and P = .023, respectively), Ms. Sitarik reported.
Of the 109 types of bacteria significantly associated with both breastfeeding and allergiclike response to pets, 71% were negatively associated with breastfeeding but positively associated with allergiclike response to pets.
This subset of risk-increasing bacteria suppressed by breastfeeding were predominantly members of the family Lachnospiraceae, the researchers found.
Lachnospiraceae are common adult gut colonizers, Ms. Sitarik said, and as people age the relative abundance of Lachnospiraceae increases. “What we think might be happening in terms of the gut microbiome is that maybe breastfeeding is preventing this premature shift into adulthood,” she said.
Dr. Johnson and Ms. Sitarik reported having no financial disclosures.
HOUSTON – Findings from a cohort study of mothers and babies point to breastfeeding as influencing infants’ gut microbiome in a way that protects them from developing allergic disease.
In findings presented at the annual meeting of the American Academy of Allergy, Asthma, and Immunology, epidemiologist Christine Cole Johnson, Ph.D., of the Henry Ford Health System in Detroit described a correlation between certain maternal and birth characteristics that had previously been shown to relate to allergic response, and measurable differences in the bacterial profiles of the study infants’ stools.
Using data from the WHEALS (Wayne County [Michigan] Health, Environment, Allergy, and Asthma Longitudinal Study) cohort, Dr. Johnson and her colleagues looked at stool samples from 298 children at 1 and 6 months of age. The investigators also collected dust samples from the infants’ homes, obtained medical records, and conducted interviews with the families (J. Allergy Clin. Immunol. 2015 [http://dx.doi.org/10.1016/j.jaci.2014.12.1443]).
The presence of household pets, the body mass index of mothers before delivery, the mode of delivery, household smoke exposure, marital status, income, race, and maternal education were all found to be significantly correlated to different gut bacterial profiles.
“Environmental and lifestyle variables that we’ve been working on related to childhood asthma and allergy seem to be associated – at least in our study – with the child’s gut microbiome,” Dr. Johnson said. “These factors vary a lot by whether those stool samples were collected at 1 or 6 months,” she said, noting that the infant gut microbiome is shaped rapidly in the first year.
But, at both 1 and 6 months of age, breastfeeding was seen as the dominant factor influencing gut bacterial composition.
At 6 months, breastfed infants had bacterial profiles showing overwhelming dominance of Bifidobacteriaceae, but vastly lower levels of other families of bacteria, notably Lachnospiraceae, which were prominent in the guts of non-breastfed babies.
In a related study that used the same cohort to explore whether the influence of breastfeeding on gut bacterial composition correlated to the development of allergic symptoms at 4 years old, Alexandra R. Sitarik, also of the Henry Ford Health System in Detroit, reported that babies being breastfed at 1 month of age had a significantly lower risk of developing allergiclike symptoms to pets by age 4 years (P = .028) (J. Allergy Clin. Immunol. 2015 [http://dx.doi.org/10.1016/j.jaci.2014.12.1444]).
Both breastfeeding and allergiclike response to pets were significantly related to compositional variation in gut microbiome (P < .001 and P = .023, respectively), Ms. Sitarik reported.
Of the 109 types of bacteria significantly associated with both breastfeeding and allergiclike response to pets, 71% were negatively associated with breastfeeding but positively associated with allergiclike response to pets.
This subset of risk-increasing bacteria suppressed by breastfeeding were predominantly members of the family Lachnospiraceae, the researchers found.
Lachnospiraceae are common adult gut colonizers, Ms. Sitarik said, and as people age the relative abundance of Lachnospiraceae increases. “What we think might be happening in terms of the gut microbiome is that maybe breastfeeding is preventing this premature shift into adulthood,” she said.
Dr. Johnson and Ms. Sitarik reported having no financial disclosures.
AT THE 2015 AAAAI ANNUAL MEETING
Key clinical point: Breastfeeding helps to alter the gut microbiome of infants, protecting them from pet allergies in childhood.
Major finding: Infants breastfed at 1 month of age had a significantly lower risk of being allergic to household pets at age 4 years (P =.028).
Data source: Data and stool samples from 298 infants enrolled in the Wayne County [Michigan] Health, Environment, Allergy, and Asthma Longitudinal Study (WHEALS) study.
Disclosures: The investigators reported having no financial disclosures.
Skin patch therapy for peanut allergy wins plaudits
HOUSTON – A peanut protein–bearing skin patch showed considerable promise in the largest randomized trial to date of any potential treatment for peanut allergy.
One year of treatment with the investigational proprietary Viaskin Peanut skin patch raised the threshold of exposure to peanut protein required to elicit an allergic reaction to a level that patients and families would consider life changing, Dr. Hugh A. Sampson said at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
“One of the biggest problems for the family of a patient with peanut allergy is the fear of getting contamination. If they could rest assured that they could tolerate something like a gram’s worth of contamination with peanut protein, much of that concern would go away. They could go to restaurants and birthday parties. They still need to be vigilant and tell people they’re peanut allergic, but the chance of these low-level contaminations that lead to most of the reactions we see would largely be taken care of,” said Dr. Sampson, professor of pediatrics, allergy, and immunology and dean for translational biomedical research at Icahn School of Medicine at Mount Sinai, New York.
He presented the findings of a phase IIb randomized, double-blind, multinational, placebo-controlled trial of what is being called epicutaneous immunotherapy. The trial involved 221 subjects with documented peanut allergy. Roughly half were aged 6-11 years; the rest were adolescents and adults up to age 55. They were randomized to one of four study arms: the skin patch at a dose of 50, 100, or 250 mcg or a placebo patch. As a requirement for study participation, all subjects had to have a peanut protein reaction–eliciting dose of 300 mg or less on baseline formal oral peanut challenge testing; half of the children reacted to 30 mg or less.
The primary endpoint was the ability to tolerate on a repeat oral challenge at 1 year either at least 1 g of peanut protein or 10 times the amount they tolerated on baseline testing. In the overall study population, the 250-mcg patch was the top performer. It was the only patch dose significantly better than placebo: 50% of all patients on the 250-mcg patch met the primary endpoint, compared with 25% of placebo-treated controls.
Peanut allergy typically starts early in life, so it’s noteworthy that the children in the study had a markedly more robust response to epicutaneous immunotherapy than the older patients. Indeed, among the 6- to 11-year-olds, all three patch doses outperformed placebo. The mean cumulative dose of peanut protein required to elicit a reaction on structured oral challenge testing was 1,121 mg greater at 1 year than at baseline among children on the 250-mcg patch, 570 mg greater with the 100-mcg patch, and 471 mg greater than at baseline among those using the 50-mcg patch.
Similarly, a robust dose-dependent increase was seen in protective peanut-specific IgG4 levels in response to 1 year of epicutaneous immunotherapy. The median level of this biomarker of therapeutic benefit climbed 19-fold over baseline in children on the 250-mcg patch, 7-fold with the 100-mcg patch, and 5.5-fold with the 50-mcg patch.
The compliance rate with patch therapy was greater than 96%. Only 2 of the 221 participants dropped out of the study due to adverse events, both because of a flare of atopic dermatitis around the patch site. There were no serious adverse events in the study.
The trial is being extended for a second year of immunotherapy. “My anticipation based on what we see with other forms of immunotherapy is that we might very well see even more protection,” according to Dr. Sampson.
In an interview, he said he’d like to see the skin patch studied at doses above 250 mcg in adolescents and adults.
“I would also love to see epicutaneous immunotherapy looked at in very young children,” he added. “You get a much better response, and it may be longer lasting because we think that the immune system in an infant is much more plastic than it is once they get older. Trying to reverse a set response is much more difficult.”
The skin patch is about the size of a small, round Band-Aid. It is placed over the back or inner upper arm and changed daily. The concept is that as humidity develops under the patch, a small amount of peanut protein permeates the outer skin. Langerhans cells then pick it up and transport it to regional lymph nodes, where T cells can activate desensitization.
“I think this concept of using a low amount of protein in a convenient, safe way could change the way we do immunotherapy,” Dr. Sampson predicted.
Asked to comment, peanut allergy researcher Dr. Brian P. Vickery of the University of North Carolina, Chapel Hill, agreed with Dr. Sampson’s assessment that patch therapy could be a game changer, especially given that there is no FDA-approved treatment for peanut allergy.
“Right now the standard of care is avoidance. So anything that improves upon that to allow a margin of safety that lets a patient get along in the world with the reassurance that a contamination event up to, say, a gram would be well tolerated would be transformative,” he said.
HOUSTON – A peanut protein–bearing skin patch showed considerable promise in the largest randomized trial to date of any potential treatment for peanut allergy.
One year of treatment with the investigational proprietary Viaskin Peanut skin patch raised the threshold of exposure to peanut protein required to elicit an allergic reaction to a level that patients and families would consider life changing, Dr. Hugh A. Sampson said at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
“One of the biggest problems for the family of a patient with peanut allergy is the fear of getting contamination. If they could rest assured that they could tolerate something like a gram’s worth of contamination with peanut protein, much of that concern would go away. They could go to restaurants and birthday parties. They still need to be vigilant and tell people they’re peanut allergic, but the chance of these low-level contaminations that lead to most of the reactions we see would largely be taken care of,” said Dr. Sampson, professor of pediatrics, allergy, and immunology and dean for translational biomedical research at Icahn School of Medicine at Mount Sinai, New York.
He presented the findings of a phase IIb randomized, double-blind, multinational, placebo-controlled trial of what is being called epicutaneous immunotherapy. The trial involved 221 subjects with documented peanut allergy. Roughly half were aged 6-11 years; the rest were adolescents and adults up to age 55. They were randomized to one of four study arms: the skin patch at a dose of 50, 100, or 250 mcg or a placebo patch. As a requirement for study participation, all subjects had to have a peanut protein reaction–eliciting dose of 300 mg or less on baseline formal oral peanut challenge testing; half of the children reacted to 30 mg or less.
The primary endpoint was the ability to tolerate on a repeat oral challenge at 1 year either at least 1 g of peanut protein or 10 times the amount they tolerated on baseline testing. In the overall study population, the 250-mcg patch was the top performer. It was the only patch dose significantly better than placebo: 50% of all patients on the 250-mcg patch met the primary endpoint, compared with 25% of placebo-treated controls.
Peanut allergy typically starts early in life, so it’s noteworthy that the children in the study had a markedly more robust response to epicutaneous immunotherapy than the older patients. Indeed, among the 6- to 11-year-olds, all three patch doses outperformed placebo. The mean cumulative dose of peanut protein required to elicit a reaction on structured oral challenge testing was 1,121 mg greater at 1 year than at baseline among children on the 250-mcg patch, 570 mg greater with the 100-mcg patch, and 471 mg greater than at baseline among those using the 50-mcg patch.
Similarly, a robust dose-dependent increase was seen in protective peanut-specific IgG4 levels in response to 1 year of epicutaneous immunotherapy. The median level of this biomarker of therapeutic benefit climbed 19-fold over baseline in children on the 250-mcg patch, 7-fold with the 100-mcg patch, and 5.5-fold with the 50-mcg patch.
The compliance rate with patch therapy was greater than 96%. Only 2 of the 221 participants dropped out of the study due to adverse events, both because of a flare of atopic dermatitis around the patch site. There were no serious adverse events in the study.
The trial is being extended for a second year of immunotherapy. “My anticipation based on what we see with other forms of immunotherapy is that we might very well see even more protection,” according to Dr. Sampson.
In an interview, he said he’d like to see the skin patch studied at doses above 250 mcg in adolescents and adults.
“I would also love to see epicutaneous immunotherapy looked at in very young children,” he added. “You get a much better response, and it may be longer lasting because we think that the immune system in an infant is much more plastic than it is once they get older. Trying to reverse a set response is much more difficult.”
The skin patch is about the size of a small, round Band-Aid. It is placed over the back or inner upper arm and changed daily. The concept is that as humidity develops under the patch, a small amount of peanut protein permeates the outer skin. Langerhans cells then pick it up and transport it to regional lymph nodes, where T cells can activate desensitization.
“I think this concept of using a low amount of protein in a convenient, safe way could change the way we do immunotherapy,” Dr. Sampson predicted.
Asked to comment, peanut allergy researcher Dr. Brian P. Vickery of the University of North Carolina, Chapel Hill, agreed with Dr. Sampson’s assessment that patch therapy could be a game changer, especially given that there is no FDA-approved treatment for peanut allergy.
“Right now the standard of care is avoidance. So anything that improves upon that to allow a margin of safety that lets a patient get along in the world with the reassurance that a contamination event up to, say, a gram would be well tolerated would be transformative,” he said.
HOUSTON – A peanut protein–bearing skin patch showed considerable promise in the largest randomized trial to date of any potential treatment for peanut allergy.
One year of treatment with the investigational proprietary Viaskin Peanut skin patch raised the threshold of exposure to peanut protein required to elicit an allergic reaction to a level that patients and families would consider life changing, Dr. Hugh A. Sampson said at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
“One of the biggest problems for the family of a patient with peanut allergy is the fear of getting contamination. If they could rest assured that they could tolerate something like a gram’s worth of contamination with peanut protein, much of that concern would go away. They could go to restaurants and birthday parties. They still need to be vigilant and tell people they’re peanut allergic, but the chance of these low-level contaminations that lead to most of the reactions we see would largely be taken care of,” said Dr. Sampson, professor of pediatrics, allergy, and immunology and dean for translational biomedical research at Icahn School of Medicine at Mount Sinai, New York.
He presented the findings of a phase IIb randomized, double-blind, multinational, placebo-controlled trial of what is being called epicutaneous immunotherapy. The trial involved 221 subjects with documented peanut allergy. Roughly half were aged 6-11 years; the rest were adolescents and adults up to age 55. They were randomized to one of four study arms: the skin patch at a dose of 50, 100, or 250 mcg or a placebo patch. As a requirement for study participation, all subjects had to have a peanut protein reaction–eliciting dose of 300 mg or less on baseline formal oral peanut challenge testing; half of the children reacted to 30 mg or less.
The primary endpoint was the ability to tolerate on a repeat oral challenge at 1 year either at least 1 g of peanut protein or 10 times the amount they tolerated on baseline testing. In the overall study population, the 250-mcg patch was the top performer. It was the only patch dose significantly better than placebo: 50% of all patients on the 250-mcg patch met the primary endpoint, compared with 25% of placebo-treated controls.
Peanut allergy typically starts early in life, so it’s noteworthy that the children in the study had a markedly more robust response to epicutaneous immunotherapy than the older patients. Indeed, among the 6- to 11-year-olds, all three patch doses outperformed placebo. The mean cumulative dose of peanut protein required to elicit a reaction on structured oral challenge testing was 1,121 mg greater at 1 year than at baseline among children on the 250-mcg patch, 570 mg greater with the 100-mcg patch, and 471 mg greater than at baseline among those using the 50-mcg patch.
Similarly, a robust dose-dependent increase was seen in protective peanut-specific IgG4 levels in response to 1 year of epicutaneous immunotherapy. The median level of this biomarker of therapeutic benefit climbed 19-fold over baseline in children on the 250-mcg patch, 7-fold with the 100-mcg patch, and 5.5-fold with the 50-mcg patch.
The compliance rate with patch therapy was greater than 96%. Only 2 of the 221 participants dropped out of the study due to adverse events, both because of a flare of atopic dermatitis around the patch site. There were no serious adverse events in the study.
The trial is being extended for a second year of immunotherapy. “My anticipation based on what we see with other forms of immunotherapy is that we might very well see even more protection,” according to Dr. Sampson.
In an interview, he said he’d like to see the skin patch studied at doses above 250 mcg in adolescents and adults.
“I would also love to see epicutaneous immunotherapy looked at in very young children,” he added. “You get a much better response, and it may be longer lasting because we think that the immune system in an infant is much more plastic than it is once they get older. Trying to reverse a set response is much more difficult.”
The skin patch is about the size of a small, round Band-Aid. It is placed over the back or inner upper arm and changed daily. The concept is that as humidity develops under the patch, a small amount of peanut protein permeates the outer skin. Langerhans cells then pick it up and transport it to regional lymph nodes, where T cells can activate desensitization.
“I think this concept of using a low amount of protein in a convenient, safe way could change the way we do immunotherapy,” Dr. Sampson predicted.
Asked to comment, peanut allergy researcher Dr. Brian P. Vickery of the University of North Carolina, Chapel Hill, agreed with Dr. Sampson’s assessment that patch therapy could be a game changer, especially given that there is no FDA-approved treatment for peanut allergy.
“Right now the standard of care is avoidance. So anything that improves upon that to allow a margin of safety that lets a patient get along in the world with the reassurance that a contamination event up to, say, a gram would be well tolerated would be transformative,” he said.
AT 2015 AAAAI ANNUAL MEETING
Key clinical point: Epicutaneous immunotherapy in patients with peanut allergy is safe, well-tolerated, and shows dose- and age-dependent efficacy.
Major finding: Half of peanut-allergic patients who wore a proprietary skin patch containing 250 mcg of peanut protein experienced a clinically meaningful increase in the threshold of exposure required to elicit a reaction, compared with 25% of controls using a placebo patch. The results were more dramatic in children than older subjects.
Data source: This was a 12-month, randomized, double-blind, placebo-controlled, multinational phase IIb study involving 221 peanut-allergic patients aged 6-55 years.
Disclosures: The study was funded by DBV Technologies. The presenter reported serving as an unpaid member of the company’s scientific advisory board.
Get the baby a dog: Infants’ gut biota seen to affect allergy risk
HOUSTON – The specific bacterial composition of infants’ gut biomes may be key to their developing or not developing allergic disease in early childhood, supporting what researchers called “a gut-airway axis” for allergic disease.
In preliminary, unpublished findings, Susan V. Lynch, Ph.D., of the department of medicine at the University of California, San Francisco, and her colleagues looked at stool samples from 298 infants aged between 0 and 11 months. They measured gut bacterial diversity, distribution, and richness at 1, 3, 6, 9, and 12 months. Gut bacteria diversified rapidly and steadily during this time, the researchers found.
"The dramatic diversification that occurs during the first year of life is the peak period of plasticity of microbial accumulation," Dr. Lynch said at the annual meeting of the American Academy of Allergy, Asthma, and Immunology. The composition of the gut microbiomes was significantly related to the age of the infants.
Dr. Lynch's group found that in infants less than 6 months of age gut biomes were dominated by Bifidobacteriaceae (70 infants), Enterobacteriaceae (49), or codominated by both (11). Gut composition related to the types of microbial exposures in the home and was affected by the presence or absence of furred pets, particularly dogs, Dr. Lynch said.
Infants without pets in the home were most likely to have a codominated biome, with a relative risk of multiple sensitization of 2.94 when compared with infants with a Bifidobacteriaeae biome and 2.06 compared with those with a Enterobacteriaceae-dominated biome.
“We asked is there a clinical correlate? Yes. That codominated community runs the highest risk of allergic disease development at age 2, having a significantly increased risk ratio, compared with either of the singly dominated communities,” Dr. Lynch said.
Moreover, the different bacterial profiles were responsible for observed differences in gut function. “We find very dramatic differences in carbohydrate and fatty acid metabolism in these communities for which atopy risk is highest,” Dr. Lynch said. “Perhaps the foundation for atopic development in childhood is associated with a compositionally distinct and highly dysfunctional neonatal gut.”
The potential for intervention is highest in the first year of life when the microbiome is most plastic, Dr. Lynch said; however, she declined to speculate what such interventions might amount to, noting that it was too early.
At the same talk, Dr. Erika Von Mutius, medical director of the asthma and allergy clinic at the University of Munich in Germany, described her work with children growing up on European farms, whom decades worth of studies have shown to have both higher diversity and frequency of microbial exposure and also lower risk of asthma and allergies than children raised in non-farm environments.
Dr. Von Mutius reported that her research group is beginning to pinpoint the protective microbes detected in these children’s’ environments and also their respiratory tracts.
A handful of organisms appear to work in concert as a microbial cocktail that predicts lower asthma and allergy rates. “What we’re seeing here is not the needle in the haystack, it’s not the one thing that will protect [from disease]. The question is, is the more the better, or is it like a soup, in which certain components are more important?’ We think it’s more like a soup,” she said.
The way in which these microbes work in the body to become protective is unknown, said Dr. Von Mutius, whose current research is looking at nose and throat samples from children. Dr. Von Mutius and her colleagues found more than fourfold more microbial diversity in farm children’s noses than their throats, though, she said, they did not yet understand why.
“The more diversity in the nose, the less asthma these children had,” she said, adding that it remained to be learned “whether you can do something in the nose to affect asthma” or whether lower respiratory and/or gut processes were key to mediating asthma risk. Dr. Lynch has received support or other funding from Second Genome, Janssen, Regeneron, and KaloBios. Dr. Von Mutius has received fees or support from ALK, Nestle, Airsonett, Protectimmun, and Novartis.
HOUSTON – The specific bacterial composition of infants’ gut biomes may be key to their developing or not developing allergic disease in early childhood, supporting what researchers called “a gut-airway axis” for allergic disease.
In preliminary, unpublished findings, Susan V. Lynch, Ph.D., of the department of medicine at the University of California, San Francisco, and her colleagues looked at stool samples from 298 infants aged between 0 and 11 months. They measured gut bacterial diversity, distribution, and richness at 1, 3, 6, 9, and 12 months. Gut bacteria diversified rapidly and steadily during this time, the researchers found.
"The dramatic diversification that occurs during the first year of life is the peak period of plasticity of microbial accumulation," Dr. Lynch said at the annual meeting of the American Academy of Allergy, Asthma, and Immunology. The composition of the gut microbiomes was significantly related to the age of the infants.
Dr. Lynch's group found that in infants less than 6 months of age gut biomes were dominated by Bifidobacteriaceae (70 infants), Enterobacteriaceae (49), or codominated by both (11). Gut composition related to the types of microbial exposures in the home and was affected by the presence or absence of furred pets, particularly dogs, Dr. Lynch said.
Infants without pets in the home were most likely to have a codominated biome, with a relative risk of multiple sensitization of 2.94 when compared with infants with a Bifidobacteriaeae biome and 2.06 compared with those with a Enterobacteriaceae-dominated biome.
“We asked is there a clinical correlate? Yes. That codominated community runs the highest risk of allergic disease development at age 2, having a significantly increased risk ratio, compared with either of the singly dominated communities,” Dr. Lynch said.
Moreover, the different bacterial profiles were responsible for observed differences in gut function. “We find very dramatic differences in carbohydrate and fatty acid metabolism in these communities for which atopy risk is highest,” Dr. Lynch said. “Perhaps the foundation for atopic development in childhood is associated with a compositionally distinct and highly dysfunctional neonatal gut.”
The potential for intervention is highest in the first year of life when the microbiome is most plastic, Dr. Lynch said; however, she declined to speculate what such interventions might amount to, noting that it was too early.
At the same talk, Dr. Erika Von Mutius, medical director of the asthma and allergy clinic at the University of Munich in Germany, described her work with children growing up on European farms, whom decades worth of studies have shown to have both higher diversity and frequency of microbial exposure and also lower risk of asthma and allergies than children raised in non-farm environments.
Dr. Von Mutius reported that her research group is beginning to pinpoint the protective microbes detected in these children’s’ environments and also their respiratory tracts.
A handful of organisms appear to work in concert as a microbial cocktail that predicts lower asthma and allergy rates. “What we’re seeing here is not the needle in the haystack, it’s not the one thing that will protect [from disease]. The question is, is the more the better, or is it like a soup, in which certain components are more important?’ We think it’s more like a soup,” she said.
The way in which these microbes work in the body to become protective is unknown, said Dr. Von Mutius, whose current research is looking at nose and throat samples from children. Dr. Von Mutius and her colleagues found more than fourfold more microbial diversity in farm children’s noses than their throats, though, she said, they did not yet understand why.
“The more diversity in the nose, the less asthma these children had,” she said, adding that it remained to be learned “whether you can do something in the nose to affect asthma” or whether lower respiratory and/or gut processes were key to mediating asthma risk. Dr. Lynch has received support or other funding from Second Genome, Janssen, Regeneron, and KaloBios. Dr. Von Mutius has received fees or support from ALK, Nestle, Airsonett, Protectimmun, and Novartis.
HOUSTON – The specific bacterial composition of infants’ gut biomes may be key to their developing or not developing allergic disease in early childhood, supporting what researchers called “a gut-airway axis” for allergic disease.
In preliminary, unpublished findings, Susan V. Lynch, Ph.D., of the department of medicine at the University of California, San Francisco, and her colleagues looked at stool samples from 298 infants aged between 0 and 11 months. They measured gut bacterial diversity, distribution, and richness at 1, 3, 6, 9, and 12 months. Gut bacteria diversified rapidly and steadily during this time, the researchers found.
"The dramatic diversification that occurs during the first year of life is the peak period of plasticity of microbial accumulation," Dr. Lynch said at the annual meeting of the American Academy of Allergy, Asthma, and Immunology. The composition of the gut microbiomes was significantly related to the age of the infants.
Dr. Lynch's group found that in infants less than 6 months of age gut biomes were dominated by Bifidobacteriaceae (70 infants), Enterobacteriaceae (49), or codominated by both (11). Gut composition related to the types of microbial exposures in the home and was affected by the presence or absence of furred pets, particularly dogs, Dr. Lynch said.
Infants without pets in the home were most likely to have a codominated biome, with a relative risk of multiple sensitization of 2.94 when compared with infants with a Bifidobacteriaeae biome and 2.06 compared with those with a Enterobacteriaceae-dominated biome.
“We asked is there a clinical correlate? Yes. That codominated community runs the highest risk of allergic disease development at age 2, having a significantly increased risk ratio, compared with either of the singly dominated communities,” Dr. Lynch said.
Moreover, the different bacterial profiles were responsible for observed differences in gut function. “We find very dramatic differences in carbohydrate and fatty acid metabolism in these communities for which atopy risk is highest,” Dr. Lynch said. “Perhaps the foundation for atopic development in childhood is associated with a compositionally distinct and highly dysfunctional neonatal gut.”
The potential for intervention is highest in the first year of life when the microbiome is most plastic, Dr. Lynch said; however, she declined to speculate what such interventions might amount to, noting that it was too early.
At the same talk, Dr. Erika Von Mutius, medical director of the asthma and allergy clinic at the University of Munich in Germany, described her work with children growing up on European farms, whom decades worth of studies have shown to have both higher diversity and frequency of microbial exposure and also lower risk of asthma and allergies than children raised in non-farm environments.
Dr. Von Mutius reported that her research group is beginning to pinpoint the protective microbes detected in these children’s’ environments and also their respiratory tracts.
A handful of organisms appear to work in concert as a microbial cocktail that predicts lower asthma and allergy rates. “What we’re seeing here is not the needle in the haystack, it’s not the one thing that will protect [from disease]. The question is, is the more the better, or is it like a soup, in which certain components are more important?’ We think it’s more like a soup,” she said.
The way in which these microbes work in the body to become protective is unknown, said Dr. Von Mutius, whose current research is looking at nose and throat samples from children. Dr. Von Mutius and her colleagues found more than fourfold more microbial diversity in farm children’s noses than their throats, though, she said, they did not yet understand why.
“The more diversity in the nose, the less asthma these children had,” she said, adding that it remained to be learned “whether you can do something in the nose to affect asthma” or whether lower respiratory and/or gut processes were key to mediating asthma risk. Dr. Lynch has received support or other funding from Second Genome, Janssen, Regeneron, and KaloBios. Dr. Von Mutius has received fees or support from ALK, Nestle, Airsonett, Protectimmun, and Novartis.
AT 2015 AAAAI ANNUAL MEETING
It’s time to take a stand against vaccine refusers
The challenges in primary care are many, and one of increasing importance is what to say to vaccine refusers. After much debate and thoughtful discussion, my medical partner, Dr. Janet Casey at Legacy Pediatrics, decided that the practice would refuse to care for the refusers.
Over the years, I have accepted such patients into my practice and worked with them to gain their confidence and debunk the many myths about the safety of vaccination that are so visible on the Internet. The approach worked well, and by the time the children were 1 year of age, I cannot remember but a handful of parents who did not come around to realize that it was best to vaccinate. However, with the recent measles outbreak at Disneyland in California, pertussis at epidemic proportions in pockets of the United States and elsewhere in the world, and the antivaccine voices gaining more and more attention, I agree, it is time to take a stand.
When a family brings their unvaccinated or undervaccinated child into the waiting room of a physician’s practice, that family is potentially exposing others in that waiting room to serious infectious diseases – that is not fair. In the waiting room may well be a patient who is on chemotherapy or immunotherapy or otherwise immunocompromised, and he relies on the “herd immunity” achieved by vaccinations of those who can safely be vaccinated for individual protection and public health. Those patients who have weakened immune systems did not choose to have their medical condition, whereas the vaccine refusers are choosing not to vaccinate their child (or typically themselves as well). And the reasons they are choosing not to vaccinate are based on misrepresentation of medical facts, fabrications of safety concerns, long ago disproven speculations by well-meaning and not so well-meaning physicians and scientists, pseudoscience published in pseudoscientific journals, and/or general distrust of the federal government that mandates vaccinations for the good of the public health.
My personal experience with vaccine scares dates back to a time when whole-cell pertussis vaccine was the only pertussis vaccine available. I was a medical student, resident, and then an infectious diseases fellow during the escalating debate about the significant side effects of vaccines. I joined in the chorus of voices questioning the need for clear data on the problem, and then the pursuit of a safer acellular pertussis vaccine. The physician community and the public were ready for change, and the National Institutes of Health took the lead in organizing multiple studies and clinical trials leading to eventual replacement of the whole cell pertussis vaccine with the current acellular vaccines.
Much more recently, at the request of National Institutes of Health, I led studies of the safety of thimerosal preservative in multidose vaccine vials that appeared in the Lancet (2002;360:1737-41); Pediatrics (2008;121:e208-14) and the Journal of Pediatrics (2009;155:495-9). Using the data from those three studies, the World Health Organization (WHO), the United Nations, the Institute of Medicine, and other organizations were able to see that the metabolism and elimination from the body of ethylmercury in thimerosal was dramatically faster, compared with methylmercury in fish. Therefore, the presumption of possible accumulation of mercury in the body of infants receiving vaccines from multidose vials when such vaccines were closely spaced was disproven by scientific data.
In plain language, there was never a known risk from thimerosal, but a premature, hurried decision was made to mandate removal of thimerosal from vaccines given to children in the United States and western Europe; thereby the myth lives on that thimerosal is not safe. Yet thimerosal is safe, and the WHO continues to advocate use of thimerosal in multidose vaccine vials. Nevertheless, I have been criticized personally on the Internet for this work. The accusation is that I, the rest of the scientists who participated in the study, and the NIH oversight were biased because our academic institutions had previously received funding from vaccine companies to perform clinical and translational research. I received many hate e-mails and even a death threat.
To close this column with a sense of humor, I suggest you Google the responses by U.S. presidential hopefuls on their stand with regard to vaccine refusers. The comments, then the reversal and “corrections” to their comments is amusing. The presidential hopefuls quickly recognized that the right to choose may not be the best policy for the public health of American citizens. Refusing to vaccinate a child potentially harms the child and may harm others!
Dr. Pichichero, a specialist in pediatric infectious diseases, is director of the Research Institute, Rochester (N.Y.) General Hospital. He is also a pediatrician at Legacy Pediatrics in Rochester. Dr. Pichichero said he had no relevant financial disclosures. E-mail him at [email protected].
The challenges in primary care are many, and one of increasing importance is what to say to vaccine refusers. After much debate and thoughtful discussion, my medical partner, Dr. Janet Casey at Legacy Pediatrics, decided that the practice would refuse to care for the refusers.
Over the years, I have accepted such patients into my practice and worked with them to gain their confidence and debunk the many myths about the safety of vaccination that are so visible on the Internet. The approach worked well, and by the time the children were 1 year of age, I cannot remember but a handful of parents who did not come around to realize that it was best to vaccinate. However, with the recent measles outbreak at Disneyland in California, pertussis at epidemic proportions in pockets of the United States and elsewhere in the world, and the antivaccine voices gaining more and more attention, I agree, it is time to take a stand.
When a family brings their unvaccinated or undervaccinated child into the waiting room of a physician’s practice, that family is potentially exposing others in that waiting room to serious infectious diseases – that is not fair. In the waiting room may well be a patient who is on chemotherapy or immunotherapy or otherwise immunocompromised, and he relies on the “herd immunity” achieved by vaccinations of those who can safely be vaccinated for individual protection and public health. Those patients who have weakened immune systems did not choose to have their medical condition, whereas the vaccine refusers are choosing not to vaccinate their child (or typically themselves as well). And the reasons they are choosing not to vaccinate are based on misrepresentation of medical facts, fabrications of safety concerns, long ago disproven speculations by well-meaning and not so well-meaning physicians and scientists, pseudoscience published in pseudoscientific journals, and/or general distrust of the federal government that mandates vaccinations for the good of the public health.
My personal experience with vaccine scares dates back to a time when whole-cell pertussis vaccine was the only pertussis vaccine available. I was a medical student, resident, and then an infectious diseases fellow during the escalating debate about the significant side effects of vaccines. I joined in the chorus of voices questioning the need for clear data on the problem, and then the pursuit of a safer acellular pertussis vaccine. The physician community and the public were ready for change, and the National Institutes of Health took the lead in organizing multiple studies and clinical trials leading to eventual replacement of the whole cell pertussis vaccine with the current acellular vaccines.
Much more recently, at the request of National Institutes of Health, I led studies of the safety of thimerosal preservative in multidose vaccine vials that appeared in the Lancet (2002;360:1737-41); Pediatrics (2008;121:e208-14) and the Journal of Pediatrics (2009;155:495-9). Using the data from those three studies, the World Health Organization (WHO), the United Nations, the Institute of Medicine, and other organizations were able to see that the metabolism and elimination from the body of ethylmercury in thimerosal was dramatically faster, compared with methylmercury in fish. Therefore, the presumption of possible accumulation of mercury in the body of infants receiving vaccines from multidose vials when such vaccines were closely spaced was disproven by scientific data.
In plain language, there was never a known risk from thimerosal, but a premature, hurried decision was made to mandate removal of thimerosal from vaccines given to children in the United States and western Europe; thereby the myth lives on that thimerosal is not safe. Yet thimerosal is safe, and the WHO continues to advocate use of thimerosal in multidose vaccine vials. Nevertheless, I have been criticized personally on the Internet for this work. The accusation is that I, the rest of the scientists who participated in the study, and the NIH oversight were biased because our academic institutions had previously received funding from vaccine companies to perform clinical and translational research. I received many hate e-mails and even a death threat.
To close this column with a sense of humor, I suggest you Google the responses by U.S. presidential hopefuls on their stand with regard to vaccine refusers. The comments, then the reversal and “corrections” to their comments is amusing. The presidential hopefuls quickly recognized that the right to choose may not be the best policy for the public health of American citizens. Refusing to vaccinate a child potentially harms the child and may harm others!
Dr. Pichichero, a specialist in pediatric infectious diseases, is director of the Research Institute, Rochester (N.Y.) General Hospital. He is also a pediatrician at Legacy Pediatrics in Rochester. Dr. Pichichero said he had no relevant financial disclosures. E-mail him at [email protected].
The challenges in primary care are many, and one of increasing importance is what to say to vaccine refusers. After much debate and thoughtful discussion, my medical partner, Dr. Janet Casey at Legacy Pediatrics, decided that the practice would refuse to care for the refusers.
Over the years, I have accepted such patients into my practice and worked with them to gain their confidence and debunk the many myths about the safety of vaccination that are so visible on the Internet. The approach worked well, and by the time the children were 1 year of age, I cannot remember but a handful of parents who did not come around to realize that it was best to vaccinate. However, with the recent measles outbreak at Disneyland in California, pertussis at epidemic proportions in pockets of the United States and elsewhere in the world, and the antivaccine voices gaining more and more attention, I agree, it is time to take a stand.
When a family brings their unvaccinated or undervaccinated child into the waiting room of a physician’s practice, that family is potentially exposing others in that waiting room to serious infectious diseases – that is not fair. In the waiting room may well be a patient who is on chemotherapy or immunotherapy or otherwise immunocompromised, and he relies on the “herd immunity” achieved by vaccinations of those who can safely be vaccinated for individual protection and public health. Those patients who have weakened immune systems did not choose to have their medical condition, whereas the vaccine refusers are choosing not to vaccinate their child (or typically themselves as well). And the reasons they are choosing not to vaccinate are based on misrepresentation of medical facts, fabrications of safety concerns, long ago disproven speculations by well-meaning and not so well-meaning physicians and scientists, pseudoscience published in pseudoscientific journals, and/or general distrust of the federal government that mandates vaccinations for the good of the public health.
My personal experience with vaccine scares dates back to a time when whole-cell pertussis vaccine was the only pertussis vaccine available. I was a medical student, resident, and then an infectious diseases fellow during the escalating debate about the significant side effects of vaccines. I joined in the chorus of voices questioning the need for clear data on the problem, and then the pursuit of a safer acellular pertussis vaccine. The physician community and the public were ready for change, and the National Institutes of Health took the lead in organizing multiple studies and clinical trials leading to eventual replacement of the whole cell pertussis vaccine with the current acellular vaccines.
Much more recently, at the request of National Institutes of Health, I led studies of the safety of thimerosal preservative in multidose vaccine vials that appeared in the Lancet (2002;360:1737-41); Pediatrics (2008;121:e208-14) and the Journal of Pediatrics (2009;155:495-9). Using the data from those three studies, the World Health Organization (WHO), the United Nations, the Institute of Medicine, and other organizations were able to see that the metabolism and elimination from the body of ethylmercury in thimerosal was dramatically faster, compared with methylmercury in fish. Therefore, the presumption of possible accumulation of mercury in the body of infants receiving vaccines from multidose vials when such vaccines were closely spaced was disproven by scientific data.
In plain language, there was never a known risk from thimerosal, but a premature, hurried decision was made to mandate removal of thimerosal from vaccines given to children in the United States and western Europe; thereby the myth lives on that thimerosal is not safe. Yet thimerosal is safe, and the WHO continues to advocate use of thimerosal in multidose vaccine vials. Nevertheless, I have been criticized personally on the Internet for this work. The accusation is that I, the rest of the scientists who participated in the study, and the NIH oversight were biased because our academic institutions had previously received funding from vaccine companies to perform clinical and translational research. I received many hate e-mails and even a death threat.
To close this column with a sense of humor, I suggest you Google the responses by U.S. presidential hopefuls on their stand with regard to vaccine refusers. The comments, then the reversal and “corrections” to their comments is amusing. The presidential hopefuls quickly recognized that the right to choose may not be the best policy for the public health of American citizens. Refusing to vaccinate a child potentially harms the child and may harm others!
Dr. Pichichero, a specialist in pediatric infectious diseases, is director of the Research Institute, Rochester (N.Y.) General Hospital. He is also a pediatrician at Legacy Pediatrics in Rochester. Dr. Pichichero said he had no relevant financial disclosures. E-mail him at [email protected].
Ventilator driving pressure may predict mortality in ARDS
One variable involved with mechanical ventilation – driving pressure – may predict mortality in adult respiratory distress syndrome, according to a report published online Feb. 19 in the New England Journal of Medicine.
Research suggests that scaling ventilator tidal volumes (VT) to patients’ body weight will minimize ventilator-induced lung injury. But patients with ARDS have a marked decrease in the proportion of lung available for ventilation, as is indicated by their lower respiratory-system compliance (CRS), which is not related to their body weight. “Therefore, we hypothesized that normalizing VT to CRS and using the ratio as an index indicating the ‘functional’ size of the lung would provide a better predictor of outcomes in patients with ARDS than VT alone,” said Dr. Marcelo B. P. Amato of the cardiopulmonary department, University of Sao Paulo (Brazil) Heart Institute, and his associates.
This ratio, also known as the driving pressure, is easily calculated at the bedside. The investigators explored whether driving pressure or other variables related to mechanical ventilation, including variables set by the ventilator operator, could be statistically linked to survival outcomes and therefore serve as a risk predictor.
They first devised a survival-prediction model using data from a cohort of 336 ARDS patients participating in four randomized clinical trials examining different ventilation strategies. They then tested their findings using a validation cohort of 861 patients from a single large trial, then tested them again in a more recent validation cohort of 2,365 patients participating in four more randomized trials comparing different ventilation strategies.
Driving pressure was the only ventilation variable found to be strongly associated with survival. Higher driving pressures strongly predicted higher mortality: Every 1-SD increase in driving pressure was related to increased mortality, with a relative risk of 1.41. Even in patients receiving lung-protective plateau pressures and low tidal volumes, higher driving pressure was associated with increased mortality, with a relative risk of 1.36, Dr. Amato and his associates said (N. Engl. J. Med. 2015 Feb. 19 [doi:10.1056/NEJMsa1410639]).
These findings can only suggest that driving pressure is a critical mediator of various ventilator strategies, since they are derived from a post hoc observational statistical analysis and cannot establish causality. Now, prospective clinical trials are needed to determine whether adjusting ventilator settings to lower driving pressure will improve survival in ARDS; in other words, “whether our observations can be translated into changes that may be implemented at the bedside,” the investigators noted.
We strongly urge caution against accepting the idea that clinicians should now set ventilators to limit driving pressure in patients with ARDS, even though that is an appealing concept.
The findings of Amato et al. derive from a meta-analysis, not from prospective randomized controlled trials. Their results should form the basis for a robust debate regarding the design of future trials so that, before we take action, we first ensure that limiting driving pressure will actually be beneficial.
Dr. Stephen H. Loring is in the department of anesthesia, critical care, and pain medicine at Beth Israel Deaconess Medical Center and at Harvard, both in Boston. Dr. Atul Malhotra is in the division of pulmonary critical care and sleep medicine at the University of California, San Diego, in La Jolla. Both Dr. Loring and Dr. Malhotra reported having no financial disclosures. They made these remarks in an editorial accompanying Dr. Amato’s report (N. Engl. J. Med. 2015 Feb. 19 [doi:10.1056/NEJMe1414218]).
We strongly urge caution against accepting the idea that clinicians should now set ventilators to limit driving pressure in patients with ARDS, even though that is an appealing concept.
The findings of Amato et al. derive from a meta-analysis, not from prospective randomized controlled trials. Their results should form the basis for a robust debate regarding the design of future trials so that, before we take action, we first ensure that limiting driving pressure will actually be beneficial.
Dr. Stephen H. Loring is in the department of anesthesia, critical care, and pain medicine at Beth Israel Deaconess Medical Center and at Harvard, both in Boston. Dr. Atul Malhotra is in the division of pulmonary critical care and sleep medicine at the University of California, San Diego, in La Jolla. Both Dr. Loring and Dr. Malhotra reported having no financial disclosures. They made these remarks in an editorial accompanying Dr. Amato’s report (N. Engl. J. Med. 2015 Feb. 19 [doi:10.1056/NEJMe1414218]).
We strongly urge caution against accepting the idea that clinicians should now set ventilators to limit driving pressure in patients with ARDS, even though that is an appealing concept.
The findings of Amato et al. derive from a meta-analysis, not from prospective randomized controlled trials. Their results should form the basis for a robust debate regarding the design of future trials so that, before we take action, we first ensure that limiting driving pressure will actually be beneficial.
Dr. Stephen H. Loring is in the department of anesthesia, critical care, and pain medicine at Beth Israel Deaconess Medical Center and at Harvard, both in Boston. Dr. Atul Malhotra is in the division of pulmonary critical care and sleep medicine at the University of California, San Diego, in La Jolla. Both Dr. Loring and Dr. Malhotra reported having no financial disclosures. They made these remarks in an editorial accompanying Dr. Amato’s report (N. Engl. J. Med. 2015 Feb. 19 [doi:10.1056/NEJMe1414218]).
One variable involved with mechanical ventilation – driving pressure – may predict mortality in adult respiratory distress syndrome, according to a report published online Feb. 19 in the New England Journal of Medicine.
Research suggests that scaling ventilator tidal volumes (VT) to patients’ body weight will minimize ventilator-induced lung injury. But patients with ARDS have a marked decrease in the proportion of lung available for ventilation, as is indicated by their lower respiratory-system compliance (CRS), which is not related to their body weight. “Therefore, we hypothesized that normalizing VT to CRS and using the ratio as an index indicating the ‘functional’ size of the lung would provide a better predictor of outcomes in patients with ARDS than VT alone,” said Dr. Marcelo B. P. Amato of the cardiopulmonary department, University of Sao Paulo (Brazil) Heart Institute, and his associates.
This ratio, also known as the driving pressure, is easily calculated at the bedside. The investigators explored whether driving pressure or other variables related to mechanical ventilation, including variables set by the ventilator operator, could be statistically linked to survival outcomes and therefore serve as a risk predictor.
They first devised a survival-prediction model using data from a cohort of 336 ARDS patients participating in four randomized clinical trials examining different ventilation strategies. They then tested their findings using a validation cohort of 861 patients from a single large trial, then tested them again in a more recent validation cohort of 2,365 patients participating in four more randomized trials comparing different ventilation strategies.
Driving pressure was the only ventilation variable found to be strongly associated with survival. Higher driving pressures strongly predicted higher mortality: Every 1-SD increase in driving pressure was related to increased mortality, with a relative risk of 1.41. Even in patients receiving lung-protective plateau pressures and low tidal volumes, higher driving pressure was associated with increased mortality, with a relative risk of 1.36, Dr. Amato and his associates said (N. Engl. J. Med. 2015 Feb. 19 [doi:10.1056/NEJMsa1410639]).
These findings can only suggest that driving pressure is a critical mediator of various ventilator strategies, since they are derived from a post hoc observational statistical analysis and cannot establish causality. Now, prospective clinical trials are needed to determine whether adjusting ventilator settings to lower driving pressure will improve survival in ARDS; in other words, “whether our observations can be translated into changes that may be implemented at the bedside,” the investigators noted.
One variable involved with mechanical ventilation – driving pressure – may predict mortality in adult respiratory distress syndrome, according to a report published online Feb. 19 in the New England Journal of Medicine.
Research suggests that scaling ventilator tidal volumes (VT) to patients’ body weight will minimize ventilator-induced lung injury. But patients with ARDS have a marked decrease in the proportion of lung available for ventilation, as is indicated by their lower respiratory-system compliance (CRS), which is not related to their body weight. “Therefore, we hypothesized that normalizing VT to CRS and using the ratio as an index indicating the ‘functional’ size of the lung would provide a better predictor of outcomes in patients with ARDS than VT alone,” said Dr. Marcelo B. P. Amato of the cardiopulmonary department, University of Sao Paulo (Brazil) Heart Institute, and his associates.
This ratio, also known as the driving pressure, is easily calculated at the bedside. The investigators explored whether driving pressure or other variables related to mechanical ventilation, including variables set by the ventilator operator, could be statistically linked to survival outcomes and therefore serve as a risk predictor.
They first devised a survival-prediction model using data from a cohort of 336 ARDS patients participating in four randomized clinical trials examining different ventilation strategies. They then tested their findings using a validation cohort of 861 patients from a single large trial, then tested them again in a more recent validation cohort of 2,365 patients participating in four more randomized trials comparing different ventilation strategies.
Driving pressure was the only ventilation variable found to be strongly associated with survival. Higher driving pressures strongly predicted higher mortality: Every 1-SD increase in driving pressure was related to increased mortality, with a relative risk of 1.41. Even in patients receiving lung-protective plateau pressures and low tidal volumes, higher driving pressure was associated with increased mortality, with a relative risk of 1.36, Dr. Amato and his associates said (N. Engl. J. Med. 2015 Feb. 19 [doi:10.1056/NEJMsa1410639]).
These findings can only suggest that driving pressure is a critical mediator of various ventilator strategies, since they are derived from a post hoc observational statistical analysis and cannot establish causality. Now, prospective clinical trials are needed to determine whether adjusting ventilator settings to lower driving pressure will improve survival in ARDS; in other words, “whether our observations can be translated into changes that may be implemented at the bedside,” the investigators noted.
Key clinical point: One variable associated with mechanical ventilation – driving pressure – may predict mortality risk in acute respiratory distress syndrome.
Major finding: Higher driving pressures strongly predicted higher mortality, such that every 1-SD increase in driving pressure was related to increased mortality with a relative risk of 1.41.
Data source: A post hoc observational statistical mediation analysis of data from 3,562 ARDS patients enrolled in nine previously reported randomized trials.
Disclosures: This study was supported by Fundacao de Amparo e Pesquisa do Estado de Sao Paulo, Conselho Nacional de Pesquisa e Desenvolvimento, and Financiadora de Estudos e Projectos. Dr. Amato reported having no financial disclosures; his associates reported ties to numerous industry sources.
Napping may compromise night sleep in children 2 years and older
Children older than 2 years may not benefit from daytime napping, according to results froma new meta-analysis of sleep studiesin children 5 years and younger.
After 2 years, napping was consistently associated with later onset of sleep during the night and lesser quality and duration of sleep, according to the findings published online Feb. 17 in Archives of Disease in Childhood (doi:10.1136/archdischild-2014-307241).
This was the finding in 26 nonrandomized studies of napping in children 5 years and younger measuring various outcomes including endpoints related to night sleep, behavior, and cognition, despite the low quality of studies evaluated and their heterogeneous endpoints, reported Karen Thorpe, Ph.D., of Queensland University of Technology in Australia, and her associates.
Correlation of daytime napping with nighttime sleep patterns ranged from small to moderate effect sizes in the studies. Whether the observed trends translate into other developmental and health outcomes is uncertain, the researchers said, and better-designed studies are needed establish this. Current findings related to child sleep and developmental and behavioral outcomes tend to measure either night sleep only or total sleep in a 24-hour period, and findings from such studies “have been extrapolated to infer the value of promoting napping, in both home and child-care contexts.” However, there may be differences in day and in night sleep, they said, citing studies on napping and night sleep that have revealed “differences in sleep architecture,circadian timing,and developmental salience.”
Dr. Thorpe and her colleagues wrote that one possible explanation for their findings is that “napping acts to dissipate homeostatic sleep pressure,”with this mechanism evidenced by increased sleep latency and later sleep onset.
“Together, the evidence suggests that, beyond the age of 2 years, napping can influence night sleep patterning.” For clinicians treating sleep problems among preschool-age children, “the investigation of napping patterns is indicated.”
Dr. Thorpe and her colleagues disclosed no relevant conflicts of interest.
Children older than 2 years may not benefit from daytime napping, according to results froma new meta-analysis of sleep studiesin children 5 years and younger.
After 2 years, napping was consistently associated with later onset of sleep during the night and lesser quality and duration of sleep, according to the findings published online Feb. 17 in Archives of Disease in Childhood (doi:10.1136/archdischild-2014-307241).
This was the finding in 26 nonrandomized studies of napping in children 5 years and younger measuring various outcomes including endpoints related to night sleep, behavior, and cognition, despite the low quality of studies evaluated and their heterogeneous endpoints, reported Karen Thorpe, Ph.D., of Queensland University of Technology in Australia, and her associates.
Correlation of daytime napping with nighttime sleep patterns ranged from small to moderate effect sizes in the studies. Whether the observed trends translate into other developmental and health outcomes is uncertain, the researchers said, and better-designed studies are needed establish this. Current findings related to child sleep and developmental and behavioral outcomes tend to measure either night sleep only or total sleep in a 24-hour period, and findings from such studies “have been extrapolated to infer the value of promoting napping, in both home and child-care contexts.” However, there may be differences in day and in night sleep, they said, citing studies on napping and night sleep that have revealed “differences in sleep architecture,circadian timing,and developmental salience.”
Dr. Thorpe and her colleagues wrote that one possible explanation for their findings is that “napping acts to dissipate homeostatic sleep pressure,”with this mechanism evidenced by increased sleep latency and later sleep onset.
“Together, the evidence suggests that, beyond the age of 2 years, napping can influence night sleep patterning.” For clinicians treating sleep problems among preschool-age children, “the investigation of napping patterns is indicated.”
Dr. Thorpe and her colleagues disclosed no relevant conflicts of interest.
Children older than 2 years may not benefit from daytime napping, according to results froma new meta-analysis of sleep studiesin children 5 years and younger.
After 2 years, napping was consistently associated with later onset of sleep during the night and lesser quality and duration of sleep, according to the findings published online Feb. 17 in Archives of Disease in Childhood (doi:10.1136/archdischild-2014-307241).
This was the finding in 26 nonrandomized studies of napping in children 5 years and younger measuring various outcomes including endpoints related to night sleep, behavior, and cognition, despite the low quality of studies evaluated and their heterogeneous endpoints, reported Karen Thorpe, Ph.D., of Queensland University of Technology in Australia, and her associates.
Correlation of daytime napping with nighttime sleep patterns ranged from small to moderate effect sizes in the studies. Whether the observed trends translate into other developmental and health outcomes is uncertain, the researchers said, and better-designed studies are needed establish this. Current findings related to child sleep and developmental and behavioral outcomes tend to measure either night sleep only or total sleep in a 24-hour period, and findings from such studies “have been extrapolated to infer the value of promoting napping, in both home and child-care contexts.” However, there may be differences in day and in night sleep, they said, citing studies on napping and night sleep that have revealed “differences in sleep architecture,circadian timing,and developmental salience.”
Dr. Thorpe and her colleagues wrote that one possible explanation for their findings is that “napping acts to dissipate homeostatic sleep pressure,”with this mechanism evidenced by increased sleep latency and later sleep onset.
“Together, the evidence suggests that, beyond the age of 2 years, napping can influence night sleep patterning.” For clinicians treating sleep problems among preschool-age children, “the investigation of napping patterns is indicated.”
Dr. Thorpe and her colleagues disclosed no relevant conflicts of interest.
Key clinical point: For clinicians treating sleep problems among preschool-age children, investigation of napping patterns may be prudent.
Major finding: Daytime napping may affect the duration and quality of night sleep in children over 2 years.
Data source: A meta-analysis of 26 observational studies on napping in children, with 9 of these looking at effects on nighttime sleep.
Disclosures: Dr. Thorpe and her colleagues disclosed no relevant conflicts of interest.