User login
Resource Utilization in Bacterial Meningitis
Bacterial meningitis can be a devastating disease in children. Overall mortality in children in the United States is 4%1 while long‐term morbidity is present in up to 25%2 of surviving children. The introduction of Haemophilus influenzae type B vaccine, heptavalent pneumococcal conjugate vaccine, and the quadrivalent meningococcal conjugate vaccine has altered the epidemiology of bacterial meningitis.24 Currently, little is known about the epidemiology of systemic complications and associated focal infections that occur during episodes of bacterial meningitis in children and how the presence of such complications affects in‐hospital healthcare resource utilization.
In a randomized controlled trial, the administration of adjuvant corticosteroids was associated with lower mortality rates in adults with bacterial meningitis due to all causes, with the greatest reduction in those with pneumococcal meningitis.5 In a post hoc analysis of data from this trial, reductions in systemic complications, such as septic shock, pneumonia, and acute respiratory distress syndrome, rather than neurologic complications were thought to be the underlying reason for the decrease in mortality associated with pneumococcal meningitis among corticosteroid recipients.6 However, children with bacterial meningitis have an overall 4‐fold lower mortality rate than adults with bacterial meningitis. An even greater difference in mortality rates exists between children and adults with pneumococcal meningitis.1, 5 Children do not benefit from adjuvant corticosteroids as adults do.1, 5, 7 Therefore, the pathogenesis of bacterial meningitis may differ in children from adults and account for the difference in response to adjuvant corticosteroids. Understanding the epidemiology of systemic complications and associated focal infections can aid in the understanding of the pathogenesis of the disease in varying age groups of children.
Previous studies in children have documented the frequency of certain bacterial meningitis‐associated conditions such as respiratory failure, pneumonia, endocarditis, and mastoiditis. Researchers have used the presence of such conditions to predict either mortality or neurologic sequelae in children.810 These studies were small and only included a few types of complications associated with bacterial meningitis. In‐hospital healthcare resource utilization, which may be an important indicator of in‐hospital morbidity, was also not considered as an outcome. In‐hospital morbidity may represent aspects of disease burden not captured by mortality rates or markers for long‐term morbidity alone. In future vaccine efficacy trails or novel therapeutics evaluations, consideration of these associated conditions is important.
The quantification of the use of in‐hospital healthcare utilization is also important for hospital planning and resource allocation in children with bacterial meningitis. A child presenting with bacterial meningitis and a systemic complication or an associated focal infection may require additional resource planning initially to expedite care to enhance recovery and decrease hospital length of stay (LOS).
Our goal was to document the frequency of bacterial meningitis‐associated conditions (systemic complications and associated focal infections) in a large cohort of children with bacterial meningitis treated at tertiary care children's hospitals in the United States, and determine how the presence of such conditions impacted in‐hospital healthcare resource utilization.
Patients and Methods
Data Source
Data for this retrospective cohort study was obtained from the Pediatric Health Information System (PHIS), a national administrative database containing data from 36 freestanding, tertiary care children's hospitals. These hospitals are affiliated with the Child Health Corporation of America (Shawnee Mission, KS), a business alliance of children's hospitals. Data quality and reliability are assured through a joint effort between the Child Health Corporation of America and participating hospitals. For the purposes of external benchmarking, participating hospitals provide discharge data including patient demographics, diagnoses, and procedures. Procedures to assure data validity were described previously.1 Total hospital charges are reported in the PHIS database and adjusted for hospital location using the Centers for Medicare and Medicaid price/wage index. A total of 27 participating hospitals also provide resource utilization data for each hospital discharge (ie, pharmaceutical dispensing, imaging, and laboratory studies); patients from these 27 hospitals were eligible for inclusion in this study. The protocol for the conduct of this study was reviewed and approved by The Children's Hospital of Philadelphia Committees for the Protection of Human Subjects.
Patients
Children less than 18 years of age with bacterial meningitis were eligible for this study if they were discharged from any of the 27 hospitals disclosing resource utilization data between January 1, 2001 and December 31, 2006. Study participants discharged with bacterial meningitis as their primary diagnosis were identified in the PHIS database using International Classification of Diseases, 9th revision, (ICD‐9) discharge diagnosis codes. The study population was limited to children without conditions predisposing to meningitis. Therefore, patients with ventricular shunts prior to the episode of bacterial meningitis were excluded using the following ICD‐9 procedure codes: ventricular shunt replacement (02.42); incision of peritoneum (54.95); removal of ventricular shunts (02.43); and the ICD‐9 discharge diagnosis code for mechanical complication of nervous system device, implant, and graft (996.2). Also, children with comorbid conditions that could predispose to meningitis or increase the likelihood of associated complications such as cancer (hematologic and nonhematologic), primary or secondary immunodeficiencies, prematurity, post‐operative infection, congenital cardiac disease, and sickle cell disease, were excluded from the analysis. Race and ethnicity were self‐reported by patients at time of admission.
Study Definitions
Study participants were identified from the PHIS database using ICD‐9 codes for the primary diagnosis of bacterial meningitis (codes 036.0‐036.1; 320.0‐320.3; 320.7; 320.81‐320.82; 320.89; 320.9). The sensitivity and specificity of ICD‐9 codes in identifying children with bacterial meningitis is unknown, however these codes have been used by previous investigators.1113 Bacterial meningitis associated‐conditions were classified as systemic complications (sepsis, systemic inflammatory response syndrome (SIRS), and respiratory failure) and associated focal infections (septic arthritis, mastoiditis, osteomyelitis, pneumonia and endocarditis). These associated conditions were identified by ICD‐9 discharge and procedural codes as listed in the Appendix (Supporting Information). Bone and joint infections were defined by the presence of either osteomyelitis or septic arthritis.
Primary Outcomes
The primary outcomes of interest were total in‐hospital charges and hospital LOS.
Measured Exposures
The primary exposures of interest were the occurrences of systemic complications, focal infections, or both conditions in children with bacterial meningitis.
Statistical Analysis
The data were initially described using frequencies and percentages for categorical variables and mean, median, interquartile ranges (IQRs) and range values for continuous variables. Analyses of bivariate associations between the outcomes (total in‐hospital charges and length of hospital stay) and potential covariates entailed either chi‐square tests or, for rare events with an expected frequency <5, Fishers Exact Test.
Following bivariate analysis, multivariable models were constructed to assess the adjusted impact of systemic complications and focal infection on total in‐hospital charges and hospital LOS. In evaluating total in‐hospital charges, the charge data were logarithmically transformed to account for the skewed distribution of charges. Multivariable linear regression was then performed to analyze the log transformed charges. The resulting beta‐coefficients were transformed to reflect the percent difference in total hospital charges between children with and without specific complications. In evaluating hospital LOS, negative binomial regression models were employed to estimate incidence rate ratios (IRRs) rather than log‐linear models, as to account for overdispersion in the outcome data. The negative binomial model produced a ratio of lengths of stay or IRR, where a ratio >1 indicates that the risk factor was associated with a longer LOS. The results were presented as percentage change to facilitate interpretation of the results.
The multivariable models were adjusted for the following confounders as determined a priori: age category, race, sex, vancomycin receipt, and adjuvant corticosteroid receipt within the first 24 hours of admission. Tests for interaction between systemic complications or focal infections and age were performed for each of these models. To address the possibility of referral bias which would lead us to overestimate the cost of caring for children with bacterial meningitis with an associated condition, the analyses were repeated restricting the sample to those children who had a lumber puncture performed at a PHIS‐participating hospital. The frequency of systemic complications and focal infections in those who were transferred was no different than in children who were not transferred; therefore the entire cohort was used in the final analyses. Sub‐group analyses were also performed for children identified with pneumococcal and meningococcal meningitis.
The standard errors for all estimates of covariate effects including metastatic effects under the above models were adjusted for the hospital to account for the increased variability due to clustering of individuals within hospitals. Two‐tailed P values <0.05 were considered statistically significant. Actual P values and 95% confidence intervals are reported. Data were analyzed using STATA, Version 10 (Stata Corporation, College Station, TX).
Results
Demographics
There were 2780 children admitted with bacterial meningitis during the study period; 461 (17%) children were excluded because of comorbid illness including malignancy (n = 37), congential heart disease (n = 231), prematurity (n = 104), human immunodeficiency virus infection (n = 4), sickle cell disease (n = 17), and post‐operative infection (n = 68). The remaining 2319 children with bacterial meningitis were included in the analyses. The mean age was 3.6 years (median, 1 year; IQR, 0‐6 years). Approximately half of the children were less than 1 year of age, 23% were 1 to 5 years, and 27% were >5 years. A total of 54% of children were white, 19% were black, 22% were Hispanic, and 5% were of other racial groups. Males accounted for 58% of the children. In this cohort of children, 9% received adjuvant corticosteroids within 24 hours of hospitalization.
Bacterial Meningitis‐Associated Conditions
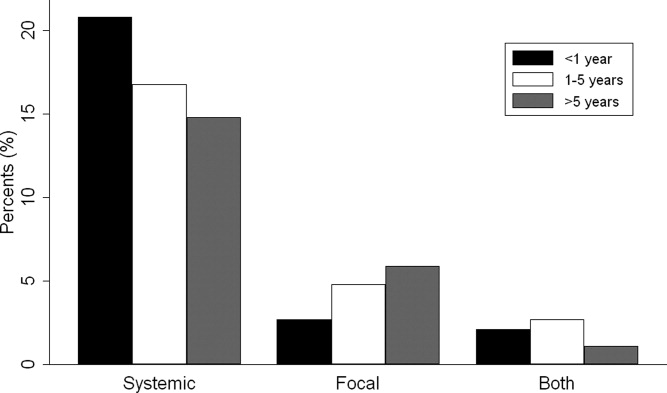
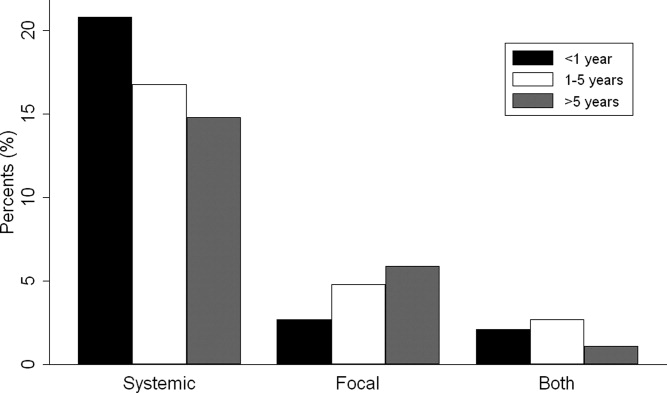
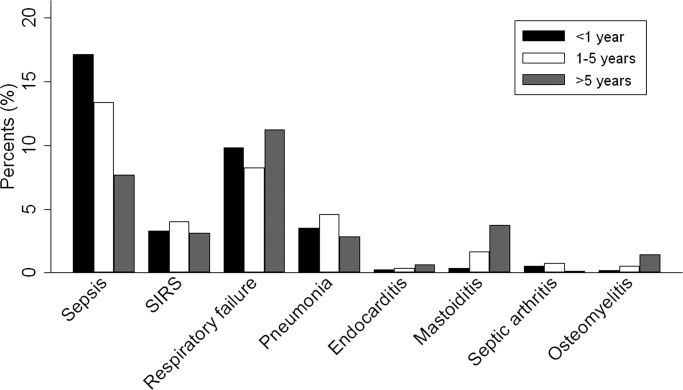
Overall, 574 (25%) of children with bacterial meningitis suffered a systemic complication or an associated focal infection. Figure 1 shows the types of associated condition stratified by age category. Older children had a higher frequency of associated focal infections while younger children had a higher frequency of systemic complications (P = 0.002, chi‐square test for trend). Figure 2 shows the distribution of specific conditions among children in each age category. The frequency of sepsis decreased with age (P < 0.001, chi‐square test) while the frequency of mastoiditis (P < 0.001, Fisher's exact test) and osteomyelitis (P = 0.005, Fisher's exact test) increased with age. There did not appear to be substantial variability in the proportion of patients with SIRS or sepsis across hospitals, suggesting that hospital‐level variability in coding for these conditions was likely minimal. The median proportion of patients with SIRS by hospital was 2.4% (IQR, 1.2‐4.8%) while the median proportion of patients with sepsis by hospital was 13.4% (IQR, 10.0‐16.9%).
Of the 151 children with an associated focal infection, only 3 (2%) of children had more than 1 infection (1 child had mastoiditis and endocarditis, 1 child had pneumonia and osteomyelitis, and 1 child had pneumonia and endocarditis). However, of the 479 children with systemic complications, 116 (24%) had more than 1 systemic disease (Table 1).
| Types of Systemic Complications | Systemic Complications in All Bacterial Meningitis, n (%) | Systemic Complications in Meningococcal Meningitis, n (%) | Systemic Complications in Pneumococcal Meningitis, n (%) |
|---|---|---|---|
| |||
| Sepsis only | 209 (44) | 16 (21) | 69 (54) |
| Respiratory failure only | 139 (29) | 38 (49) | 30 (24) |
| SIRS only | 15 (3) | 9 (12) | 1 (1) |
| Sepsis and respiratory failure | 52 (11) | 4 (5) | 18 (14) |
| SIRS and sepsis | 27 (6) | 2 (3) | 4 (3) |
| SIRS and respiratory failure | 9 (2) | 5 (6) | 0 (0) |
| SIRS and respiratory failure and sepsis | 28 (6) | 3 (4) | 5 (4) |
| Total systemic complications | 479 | 77 | 127 |
In sub‐group analyses, 269 children had meningococcal meningitis and 470 children had pneumococcal meningitis. Of the children with meningococcal meningitis, 31.2% had a meningitis‐associated condition: 26.4% had a systemic complication, 2.6% had a focal infection, and 2.2% had both conditions. The most common associated conditions in children with meningococcal meningitis were respiratory failure (18.6%; n = 50), sepsis (9.3%; n = 25), and SIRS (7.1%; n = 19). In children with pneumococcal meningitis, 32.3% had a meningitis‐associated complication: 24.7% had a systemic complication, 5.3% had a focal infection, and 2.3% had both conditions. The most common associated conditions in children with pneumococcal meningitis were sepsis (20.4%; n = 96), respiratory failure (11.3%; n = 53), and pneumonia (4.7%; n = 22); mastoiditis was present in 2.3% (n = 11) of children with pneumococcal meningitis. Respiratory failure was more common in meningococcal meningitis (18.6%) than in pneumococcal meningitis (11.3%; P = 0.006). In contrast, sepsis was less common in meningococcal meningitis (9.3%) than in pneumococcal meningitis (20.4%; P < 0.001).
Hospital Charges
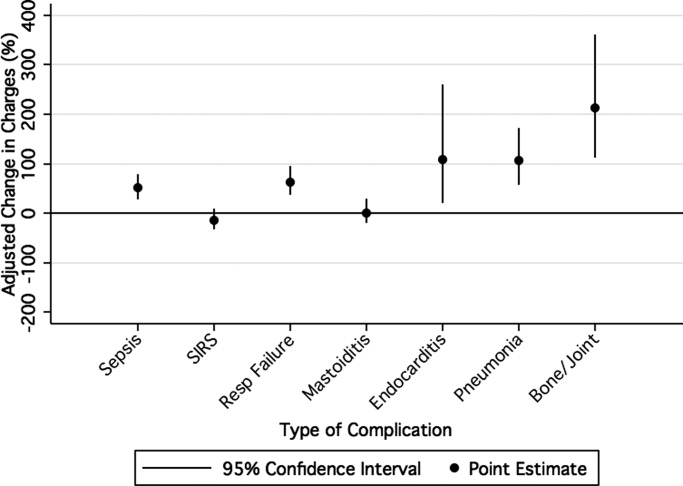
Overall, the median charges per hospital ranged from $20,158 to $53,823. In‐hospital charges for children with bacterial meningitis with and without any identified associated conditions are presented in Table 2. In multivariate analyses, the presence of systemic conditions, associated focal infections, or both conditions was independently associated with significantly higher total in‐hospital charges (Table 2). When conditions were considered individually, bone and joint infections (213% increase; 95% CI, 113‐260%), endocarditis (108% increase; 95% CI, 23‐258%), and pneumonia (107% increase; 95% CI, 58‐171%) were associated with the highest increases in total hospital charges (Figure 3). In contrast, SIRS and mastoiditis were not associated with higher hospital charges (Figure 3).
| Charges | LOS | |||
|---|---|---|---|---|
| Median, $ (IQR) | Adjusted Increase,* % (95% CI) | Median, days (IQR) | Adjusted Increase,* % (95% CI) | |
| ||||
| None (n = 1,745) | $27,110 (15,823‐48,307) | Reference** | 9 (6‐14) | Reference |
| Systemic (n = 423) | $66,690 (39,546136,756) | 136 (108269) | 14 (923) | 72 (5196) |
| Focal Infection (n = 95) | $58,016 (29,056125,813) | 118 (77168) | 13 (928) | 78 (40126) |
| Both (n = 56) | $130,744 (62,397299,288) | 351 (237503) | 21.5 (1245) | 211 (142303) |
LOS
The median LOS was 9 days (IQR, 6‐15 days); 5% of children had a LOS >42 days. Table 2 summarizes difference in LOS by the presence and absence of systemic conditions and focal infections. In multivariate analyses, the presence of systemic conditions, associated focal infections, or both conditions was independently associated with a significantly longer LOS (Table 2). When conditions were considered individually, endocarditis (152% increase; 95% CI, 60‐300%) and pneumonia (136% increase; 95% CI, 85‐201%) were associated with the greatest adjusted increases in LOS (Figure 4); only mastoiditis was not associated with an increased LOS compared with those without complications.

Discussion
To our knowledge, this is the first study to examine bacterial meningitis‐associated conditions in children and their impact on in‐hospital resource utilization. We found that 25% of the cohort of children with bacterial meningitis suffered from at least one focal infection or systemic complication. This represents a significant invasive disease burden among children with bacterial meningitis who do not have underlying comorbid conditions. Younger children were more likely to have systemic complications when compared with older children, specifically due to a higher frequency of sepsis in children <1 year. Older children were more likely to have an associated focal infection, specifically due to an increase in mastoiditis and osteomyelitis in children >1 year. Only 2% of children had more than 1 focal infection, while 24% of children had more than 1 systemic complication.
Importantly, the presence of a systemic complication in a child with bacterial meningitis increased their in‐hospital adjusted charges by 136%. The presence of a focal infection increased in‐hospital adjusted charges by 118%. A child with both a systemic complication and a focal infection and had a 351% increase in in‐hospital adjusted charges.
The presence of systemic complications or associated focal infections was significantly associated with higher in‐hospital charges and longer hospital LOS. Most individual meningitis‐associated conditions included in this study were associated with higher in‐hospital charges with the exception of SIRS and mastoiditis. All individual meningitis‐associated conditions were associated with a longer LOS except mastoiditis. This finding is not surprising as the LOS for children with mastoiditis is typically shorter than for children with bacterial meningitis. Glikich et al.14 reported a mean LOS of approximately 8 days for children with mastoiditis. As meningitis in the context of mastoiditis is likely caused by direct extension of infection, patients with meningitis and mastoiditis likely required extended hospitalization to treat meningitis rather than mastoiditis. In contrast, patients with meningitis occurring in the context of metastatic dissemination of infection (eg, endocarditis, pneumonia) often have hemodynamic instability requiring prolonged intensive care support.
A study of children with sepsis found that increasing severity of illness was associated with greater hospital resource utilization.15 Our study shows that this may also be true in children with bacterial meningitis. We found that in children with bacterial meningitis, having systemic complications or an associated focal infection was associated with greater in‐hospital resource utilization. This finding may therefore indicate greater in‐hospital morbidity among children with a bacterial meningitis‐associated condition. Since mortality rates for bacterial meningitis are low in children, in‐hospital morbidity may be a better indicator of disease burden.
Our data show that, in contrast to adults, bacterial meningitis in children is not typically associated with other focal infections. Some focal complications such as mastoiditis and osteomyelitis disproportionately affect older children. These complications are typically accompanied by overt clinical manifestations. Therefore, we believe that the evaluation for the presence of concomitant focal infections can be guided by clinical examination findings and that routine radiologic evaluation for focal complications may not be necessary. Additionally, focal infections tend to occur in the absence of concomitant systemic complications. Of the 151 children with at least 1 associated focal infection, only 37% had a systemic complication. Bacterial meningitis may lie on a continuum of invasive disease depending on the virulence factors of the invading pathogen as well as specific host factors. Understanding the epidemiology of these associated conditions can enhance our understanding of the pathogenesis of bacterial meningitis in children. Understanding why some children suffer from septicemia rather than bacteremia may help in developing novel therapeutics.
There are several limitations to our study. First, since we identified focal infections and systemic complications using billing charges and ICD‐9 discharge diagnosis codes, it was impossible to determine when these conditions represented true complications of bacterial meningitis and when they represented the primary source of infection. Therefore, some of our primary outcomes may represent the cause of meningitis rather than a direct complication. We attempted to minimize such misclassification by limiting the cohort to those with a primary discharge diagnosis of bacterial meningitis though such misclassification is still possible.
Second, the use of ICD‐9 codes to accurately identify systemic complications and associated focal infections is a potential limitation. For example, respiratory failure, defined as the requirement of endotracheal intubation in our study, may not capture children receiving non‐invasive mechanical ventilation (eg, bilevel positive airway pressure). If use of noninvasive ventilation strategies did not depend exclusively on illness severity, our study would underestimate the frequency of respiratory failure. Furthermore, there may be inconsistencies among pediatric physicians in coding conditions such as SIRS and sepsis. Even in the clinical setting, a uniform definition of SIRS and sepsis is problematic due to physiologic differences between adults and children of varying age groups.16 An international panel of pediatricians proposed age‐specific definitions for sepsis and SIRS, while acknowledging the paucity of evidence to support some of their recommendations.16 None of the proposed definitions could be applied using administrative data. Limitations in the use of ICD‐9 discharge diagnosis codes to identify children with bacterial meningitis were discussed previously.1
Third, only free‐standing children's hospitals were included in the analysis. It is likely that many children with uncomplicated bacterial meningitis are treated at community hospitals or smaller academic centers. Our study may overestimate the rate of bacterial meningitis‐associated focal infections and systemic complications since participating hospitals serve as regional referral centers. To address the potential for such referral bias, we repeated the analysis while restricting the cohort to those children who had a lumbar puncture performed at the treating facility. No difference in frequency of associated conditions or in‐hospital resource utilization was found between children transferred and children not transferred. Finally, the PHIS database reports billed charge data rather than cost data. Billed data may overestimate the actual economic impact of bacterial meningitis‐associated complications since payers often reimburse at lesser rates. Resource utilization may also vary widely between hospitals and geographic locations as previously shown.15
In conclusion, bacterial meningitis remains an important cause of morbidity in children. Systemic complications such as sepsis and respiratory failure are common. Respiratory failure occurred more commonly among patients with meningococcal meningitis while sepsis occurred more commonly among patients with pneumococcal meningitis. While focal complications are uncommon, children >5 years of age are more likely than younger children to have concomitant mastoiditis or osteomyelitis. The presence of both systemic and focal complications is associated with substantially greater resource utilization than either complication alone.
Dr. Shah had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the analysis. Study concept and design: Shah, Mongelluzzo; acquisition of data: Shah, Mohamad; analysis and interpretation of data: Mongelluzzo, Mohamad, Ten Have, Shah; drafting of the manuscript: Mongelluzzo; critical revision of the manuscript for important intellectual content: Mongelluzzo, Mohamad, Ten Have, Shah; statistical analysis: Shah, Mongelluzzo, Ten Have; obtained funding: Shah, Mongelluzzo; administrative, technical, or material support: Shah; study supervision: Shah.
Appendix
Diagnosis Codes:
Endocarditis: 421.0, 421.1, 421.9
Mastoiditis: 383.0, 383.1, 383.2, 383.8, 383.9
Osteomyelitis: 730.0, 730.1, 730.2, 730.3, 730.7, 730.8, 730.9
Septic arthritis: 711.0, 711.1, 711.2, 711.3, 711.4, 711.5, 711.6, 711.7, 711.8, 711.9
Sepsis: 038.0, 038.1, 038.2, 038.3, 038.4, 038.8, 038.9
Systemic Inflammatory Response Syndrome: 995.92
Pneumonia: 480.0, 480.1, 480.2, 480.3, 480.8, 480.9, 481, 482.0, 482.1, 482.2, 482.3, 482.4, 482.8, 482.9, 483.0, 483.1, 483.8, 484.1, 484.3, 484.5, 484.6, 484.7, 484.8, 485, 486
Procedure Codes:
Endotracheal Intubation: 96.04
- , , , .Corticosteroids and mortality in children with bacterial meningitis.JAMA.2008;299:2048–2055.
- , , , et al.Bacterial meningitis in the United States in 1995. Active surveillance team.N Engl J Med.1997;337:970–976.
- Progress toward elimination of Haemophilus influenzae type b disease among infants and children–United States, 1987–1995.MMWR Morb Mortal Wkly Rep.1996;45:901–906.
- , .Bacterial meningitis in children.Lancet.2003;361:2139–2148.
- , .Dexamethasone in adults with bacterial meningitis.N Engl J Med.2002;347:1549–1556.
- , .Dexamethasone and pneumococcal meningitis.Ann Intern Med.2004;141:327.
- , , , et al.Dexamethasone in Vietnamese adolescents and adults with bacterial meningitis.N Engl J Med.2007;357:2431–2440.
- , , , , .Bacterial meningitis in an urban area: etiologic study and prognostic factors.Infection.2007;35:406–413.
- , , .Clinical features and prognostic factors in childhood pneumococcal meningitis.J Microbiol Immunol Infect.2008;41:48–53.
- , , .Clinical presentation and prognostic factors of Streptococcus pneumoniae meningitis according to the focus of infection.BMC Infect Dis.2005;5:93.
- , .Trends in invasive pneumococcal disease‐associated hospitalizations.Clin Infect Dis.2006;42:e1–e5.
- , , .Managing meningococcal disease in the United States: Hospital case characteristics and costs by age.Value Health.2006;9:236–243.
- , , , , , .Population‐based analysis of meningococcal disease mortality in the United States: 1990–2002.Pediatr Infect Dis J.2006;25:191–194.
- , , , .A contemporary analysis of acute mastoiditis.Arch Otolaryngol Head Neck Surg.1996;122:135–139.
- , , .Patient and hospital correlates of clinical outcomes and resource utilization in severe pediatric sepsis.Pediatrics.2007;119:487–494.
- , , .International pediatric sepsis consensus conference: definitions for sepsis and organ dysfunction in pediatrics.Pediatr Crit Care Med.2005;6:2–8.
Bacterial meningitis can be a devastating disease in children. Overall mortality in children in the United States is 4%1 while long‐term morbidity is present in up to 25%2 of surviving children. The introduction of Haemophilus influenzae type B vaccine, heptavalent pneumococcal conjugate vaccine, and the quadrivalent meningococcal conjugate vaccine has altered the epidemiology of bacterial meningitis.24 Currently, little is known about the epidemiology of systemic complications and associated focal infections that occur during episodes of bacterial meningitis in children and how the presence of such complications affects in‐hospital healthcare resource utilization.
In a randomized controlled trial, the administration of adjuvant corticosteroids was associated with lower mortality rates in adults with bacterial meningitis due to all causes, with the greatest reduction in those with pneumococcal meningitis.5 In a post hoc analysis of data from this trial, reductions in systemic complications, such as septic shock, pneumonia, and acute respiratory distress syndrome, rather than neurologic complications were thought to be the underlying reason for the decrease in mortality associated with pneumococcal meningitis among corticosteroid recipients.6 However, children with bacterial meningitis have an overall 4‐fold lower mortality rate than adults with bacterial meningitis. An even greater difference in mortality rates exists between children and adults with pneumococcal meningitis.1, 5 Children do not benefit from adjuvant corticosteroids as adults do.1, 5, 7 Therefore, the pathogenesis of bacterial meningitis may differ in children from adults and account for the difference in response to adjuvant corticosteroids. Understanding the epidemiology of systemic complications and associated focal infections can aid in the understanding of the pathogenesis of the disease in varying age groups of children.
Previous studies in children have documented the frequency of certain bacterial meningitis‐associated conditions such as respiratory failure, pneumonia, endocarditis, and mastoiditis. Researchers have used the presence of such conditions to predict either mortality or neurologic sequelae in children.810 These studies were small and only included a few types of complications associated with bacterial meningitis. In‐hospital healthcare resource utilization, which may be an important indicator of in‐hospital morbidity, was also not considered as an outcome. In‐hospital morbidity may represent aspects of disease burden not captured by mortality rates or markers for long‐term morbidity alone. In future vaccine efficacy trails or novel therapeutics evaluations, consideration of these associated conditions is important.
The quantification of the use of in‐hospital healthcare utilization is also important for hospital planning and resource allocation in children with bacterial meningitis. A child presenting with bacterial meningitis and a systemic complication or an associated focal infection may require additional resource planning initially to expedite care to enhance recovery and decrease hospital length of stay (LOS).
Our goal was to document the frequency of bacterial meningitis‐associated conditions (systemic complications and associated focal infections) in a large cohort of children with bacterial meningitis treated at tertiary care children's hospitals in the United States, and determine how the presence of such conditions impacted in‐hospital healthcare resource utilization.
Patients and Methods
Data Source
Data for this retrospective cohort study was obtained from the Pediatric Health Information System (PHIS), a national administrative database containing data from 36 freestanding, tertiary care children's hospitals. These hospitals are affiliated with the Child Health Corporation of America (Shawnee Mission, KS), a business alliance of children's hospitals. Data quality and reliability are assured through a joint effort between the Child Health Corporation of America and participating hospitals. For the purposes of external benchmarking, participating hospitals provide discharge data including patient demographics, diagnoses, and procedures. Procedures to assure data validity were described previously.1 Total hospital charges are reported in the PHIS database and adjusted for hospital location using the Centers for Medicare and Medicaid price/wage index. A total of 27 participating hospitals also provide resource utilization data for each hospital discharge (ie, pharmaceutical dispensing, imaging, and laboratory studies); patients from these 27 hospitals were eligible for inclusion in this study. The protocol for the conduct of this study was reviewed and approved by The Children's Hospital of Philadelphia Committees for the Protection of Human Subjects.
Patients
Children less than 18 years of age with bacterial meningitis were eligible for this study if they were discharged from any of the 27 hospitals disclosing resource utilization data between January 1, 2001 and December 31, 2006. Study participants discharged with bacterial meningitis as their primary diagnosis were identified in the PHIS database using International Classification of Diseases, 9th revision, (ICD‐9) discharge diagnosis codes. The study population was limited to children without conditions predisposing to meningitis. Therefore, patients with ventricular shunts prior to the episode of bacterial meningitis were excluded using the following ICD‐9 procedure codes: ventricular shunt replacement (02.42); incision of peritoneum (54.95); removal of ventricular shunts (02.43); and the ICD‐9 discharge diagnosis code for mechanical complication of nervous system device, implant, and graft (996.2). Also, children with comorbid conditions that could predispose to meningitis or increase the likelihood of associated complications such as cancer (hematologic and nonhematologic), primary or secondary immunodeficiencies, prematurity, post‐operative infection, congenital cardiac disease, and sickle cell disease, were excluded from the analysis. Race and ethnicity were self‐reported by patients at time of admission.
Study Definitions
Study participants were identified from the PHIS database using ICD‐9 codes for the primary diagnosis of bacterial meningitis (codes 036.0‐036.1; 320.0‐320.3; 320.7; 320.81‐320.82; 320.89; 320.9). The sensitivity and specificity of ICD‐9 codes in identifying children with bacterial meningitis is unknown, however these codes have been used by previous investigators.1113 Bacterial meningitis associated‐conditions were classified as systemic complications (sepsis, systemic inflammatory response syndrome (SIRS), and respiratory failure) and associated focal infections (septic arthritis, mastoiditis, osteomyelitis, pneumonia and endocarditis). These associated conditions were identified by ICD‐9 discharge and procedural codes as listed in the Appendix (Supporting Information). Bone and joint infections were defined by the presence of either osteomyelitis or septic arthritis.
Primary Outcomes
The primary outcomes of interest were total in‐hospital charges and hospital LOS.
Measured Exposures
The primary exposures of interest were the occurrences of systemic complications, focal infections, or both conditions in children with bacterial meningitis.
Statistical Analysis
The data were initially described using frequencies and percentages for categorical variables and mean, median, interquartile ranges (IQRs) and range values for continuous variables. Analyses of bivariate associations between the outcomes (total in‐hospital charges and length of hospital stay) and potential covariates entailed either chi‐square tests or, for rare events with an expected frequency <5, Fishers Exact Test.
Following bivariate analysis, multivariable models were constructed to assess the adjusted impact of systemic complications and focal infection on total in‐hospital charges and hospital LOS. In evaluating total in‐hospital charges, the charge data were logarithmically transformed to account for the skewed distribution of charges. Multivariable linear regression was then performed to analyze the log transformed charges. The resulting beta‐coefficients were transformed to reflect the percent difference in total hospital charges between children with and without specific complications. In evaluating hospital LOS, negative binomial regression models were employed to estimate incidence rate ratios (IRRs) rather than log‐linear models, as to account for overdispersion in the outcome data. The negative binomial model produced a ratio of lengths of stay or IRR, where a ratio >1 indicates that the risk factor was associated with a longer LOS. The results were presented as percentage change to facilitate interpretation of the results.
The multivariable models were adjusted for the following confounders as determined a priori: age category, race, sex, vancomycin receipt, and adjuvant corticosteroid receipt within the first 24 hours of admission. Tests for interaction between systemic complications or focal infections and age were performed for each of these models. To address the possibility of referral bias which would lead us to overestimate the cost of caring for children with bacterial meningitis with an associated condition, the analyses were repeated restricting the sample to those children who had a lumber puncture performed at a PHIS‐participating hospital. The frequency of systemic complications and focal infections in those who were transferred was no different than in children who were not transferred; therefore the entire cohort was used in the final analyses. Sub‐group analyses were also performed for children identified with pneumococcal and meningococcal meningitis.
The standard errors for all estimates of covariate effects including metastatic effects under the above models were adjusted for the hospital to account for the increased variability due to clustering of individuals within hospitals. Two‐tailed P values <0.05 were considered statistically significant. Actual P values and 95% confidence intervals are reported. Data were analyzed using STATA, Version 10 (Stata Corporation, College Station, TX).
Results
Demographics
There were 2780 children admitted with bacterial meningitis during the study period; 461 (17%) children were excluded because of comorbid illness including malignancy (n = 37), congential heart disease (n = 231), prematurity (n = 104), human immunodeficiency virus infection (n = 4), sickle cell disease (n = 17), and post‐operative infection (n = 68). The remaining 2319 children with bacterial meningitis were included in the analyses. The mean age was 3.6 years (median, 1 year; IQR, 0‐6 years). Approximately half of the children were less than 1 year of age, 23% were 1 to 5 years, and 27% were >5 years. A total of 54% of children were white, 19% were black, 22% were Hispanic, and 5% were of other racial groups. Males accounted for 58% of the children. In this cohort of children, 9% received adjuvant corticosteroids within 24 hours of hospitalization.
Bacterial Meningitis‐Associated Conditions
Overall, 574 (25%) of children with bacterial meningitis suffered a systemic complication or an associated focal infection. Figure 1 shows the types of associated condition stratified by age category. Older children had a higher frequency of associated focal infections while younger children had a higher frequency of systemic complications (P = 0.002, chi‐square test for trend). Figure 2 shows the distribution of specific conditions among children in each age category. The frequency of sepsis decreased with age (P < 0.001, chi‐square test) while the frequency of mastoiditis (P < 0.001, Fisher's exact test) and osteomyelitis (P = 0.005, Fisher's exact test) increased with age. There did not appear to be substantial variability in the proportion of patients with SIRS or sepsis across hospitals, suggesting that hospital‐level variability in coding for these conditions was likely minimal. The median proportion of patients with SIRS by hospital was 2.4% (IQR, 1.2‐4.8%) while the median proportion of patients with sepsis by hospital was 13.4% (IQR, 10.0‐16.9%).


Of the 151 children with an associated focal infection, only 3 (2%) of children had more than 1 infection (1 child had mastoiditis and endocarditis, 1 child had pneumonia and osteomyelitis, and 1 child had pneumonia and endocarditis). However, of the 479 children with systemic complications, 116 (24%) had more than 1 systemic disease (Table 1).
| Types of Systemic Complications | Systemic Complications in All Bacterial Meningitis, n (%) | Systemic Complications in Meningococcal Meningitis, n (%) | Systemic Complications in Pneumococcal Meningitis, n (%) |
|---|---|---|---|
| |||
| Sepsis only | 209 (44) | 16 (21) | 69 (54) |
| Respiratory failure only | 139 (29) | 38 (49) | 30 (24) |
| SIRS only | 15 (3) | 9 (12) | 1 (1) |
| Sepsis and respiratory failure | 52 (11) | 4 (5) | 18 (14) |
| SIRS and sepsis | 27 (6) | 2 (3) | 4 (3) |
| SIRS and respiratory failure | 9 (2) | 5 (6) | 0 (0) |
| SIRS and respiratory failure and sepsis | 28 (6) | 3 (4) | 5 (4) |
| Total systemic complications | 479 | 77 | 127 |
In sub‐group analyses, 269 children had meningococcal meningitis and 470 children had pneumococcal meningitis. Of the children with meningococcal meningitis, 31.2% had a meningitis‐associated condition: 26.4% had a systemic complication, 2.6% had a focal infection, and 2.2% had both conditions. The most common associated conditions in children with meningococcal meningitis were respiratory failure (18.6%; n = 50), sepsis (9.3%; n = 25), and SIRS (7.1%; n = 19). In children with pneumococcal meningitis, 32.3% had a meningitis‐associated complication: 24.7% had a systemic complication, 5.3% had a focal infection, and 2.3% had both conditions. The most common associated conditions in children with pneumococcal meningitis were sepsis (20.4%; n = 96), respiratory failure (11.3%; n = 53), and pneumonia (4.7%; n = 22); mastoiditis was present in 2.3% (n = 11) of children with pneumococcal meningitis. Respiratory failure was more common in meningococcal meningitis (18.6%) than in pneumococcal meningitis (11.3%; P = 0.006). In contrast, sepsis was less common in meningococcal meningitis (9.3%) than in pneumococcal meningitis (20.4%; P < 0.001).
Hospital Charges
Overall, the median charges per hospital ranged from $20,158 to $53,823. In‐hospital charges for children with bacterial meningitis with and without any identified associated conditions are presented in Table 2. In multivariate analyses, the presence of systemic conditions, associated focal infections, or both conditions was independently associated with significantly higher total in‐hospital charges (Table 2). When conditions were considered individually, bone and joint infections (213% increase; 95% CI, 113‐260%), endocarditis (108% increase; 95% CI, 23‐258%), and pneumonia (107% increase; 95% CI, 58‐171%) were associated with the highest increases in total hospital charges (Figure 3). In contrast, SIRS and mastoiditis were not associated with higher hospital charges (Figure 3).
| Charges | LOS | |||
|---|---|---|---|---|
| Median, $ (IQR) | Adjusted Increase,* % (95% CI) | Median, days (IQR) | Adjusted Increase,* % (95% CI) | |
| ||||
| None (n = 1,745) | $27,110 (15,823‐48,307) | Reference** | 9 (6‐14) | Reference |
| Systemic (n = 423) | $66,690 (39,546136,756) | 136 (108269) | 14 (923) | 72 (5196) |
| Focal Infection (n = 95) | $58,016 (29,056125,813) | 118 (77168) | 13 (928) | 78 (40126) |
| Both (n = 56) | $130,744 (62,397299,288) | 351 (237503) | 21.5 (1245) | 211 (142303) |

LOS
The median LOS was 9 days (IQR, 6‐15 days); 5% of children had a LOS >42 days. Table 2 summarizes difference in LOS by the presence and absence of systemic conditions and focal infections. In multivariate analyses, the presence of systemic conditions, associated focal infections, or both conditions was independently associated with a significantly longer LOS (Table 2). When conditions were considered individually, endocarditis (152% increase; 95% CI, 60‐300%) and pneumonia (136% increase; 95% CI, 85‐201%) were associated with the greatest adjusted increases in LOS (Figure 4); only mastoiditis was not associated with an increased LOS compared with those without complications.

Discussion
To our knowledge, this is the first study to examine bacterial meningitis‐associated conditions in children and their impact on in‐hospital resource utilization. We found that 25% of the cohort of children with bacterial meningitis suffered from at least one focal infection or systemic complication. This represents a significant invasive disease burden among children with bacterial meningitis who do not have underlying comorbid conditions. Younger children were more likely to have systemic complications when compared with older children, specifically due to a higher frequency of sepsis in children <1 year. Older children were more likely to have an associated focal infection, specifically due to an increase in mastoiditis and osteomyelitis in children >1 year. Only 2% of children had more than 1 focal infection, while 24% of children had more than 1 systemic complication.
Importantly, the presence of a systemic complication in a child with bacterial meningitis increased their in‐hospital adjusted charges by 136%. The presence of a focal infection increased in‐hospital adjusted charges by 118%. A child with both a systemic complication and a focal infection and had a 351% increase in in‐hospital adjusted charges.
The presence of systemic complications or associated focal infections was significantly associated with higher in‐hospital charges and longer hospital LOS. Most individual meningitis‐associated conditions included in this study were associated with higher in‐hospital charges with the exception of SIRS and mastoiditis. All individual meningitis‐associated conditions were associated with a longer LOS except mastoiditis. This finding is not surprising as the LOS for children with mastoiditis is typically shorter than for children with bacterial meningitis. Glikich et al.14 reported a mean LOS of approximately 8 days for children with mastoiditis. As meningitis in the context of mastoiditis is likely caused by direct extension of infection, patients with meningitis and mastoiditis likely required extended hospitalization to treat meningitis rather than mastoiditis. In contrast, patients with meningitis occurring in the context of metastatic dissemination of infection (eg, endocarditis, pneumonia) often have hemodynamic instability requiring prolonged intensive care support.
A study of children with sepsis found that increasing severity of illness was associated with greater hospital resource utilization.15 Our study shows that this may also be true in children with bacterial meningitis. We found that in children with bacterial meningitis, having systemic complications or an associated focal infection was associated with greater in‐hospital resource utilization. This finding may therefore indicate greater in‐hospital morbidity among children with a bacterial meningitis‐associated condition. Since mortality rates for bacterial meningitis are low in children, in‐hospital morbidity may be a better indicator of disease burden.
Our data show that, in contrast to adults, bacterial meningitis in children is not typically associated with other focal infections. Some focal complications such as mastoiditis and osteomyelitis disproportionately affect older children. These complications are typically accompanied by overt clinical manifestations. Therefore, we believe that the evaluation for the presence of concomitant focal infections can be guided by clinical examination findings and that routine radiologic evaluation for focal complications may not be necessary. Additionally, focal infections tend to occur in the absence of concomitant systemic complications. Of the 151 children with at least 1 associated focal infection, only 37% had a systemic complication. Bacterial meningitis may lie on a continuum of invasive disease depending on the virulence factors of the invading pathogen as well as specific host factors. Understanding the epidemiology of these associated conditions can enhance our understanding of the pathogenesis of bacterial meningitis in children. Understanding why some children suffer from septicemia rather than bacteremia may help in developing novel therapeutics.
There are several limitations to our study. First, since we identified focal infections and systemic complications using billing charges and ICD‐9 discharge diagnosis codes, it was impossible to determine when these conditions represented true complications of bacterial meningitis and when they represented the primary source of infection. Therefore, some of our primary outcomes may represent the cause of meningitis rather than a direct complication. We attempted to minimize such misclassification by limiting the cohort to those with a primary discharge diagnosis of bacterial meningitis though such misclassification is still possible.
Second, the use of ICD‐9 codes to accurately identify systemic complications and associated focal infections is a potential limitation. For example, respiratory failure, defined as the requirement of endotracheal intubation in our study, may not capture children receiving non‐invasive mechanical ventilation (eg, bilevel positive airway pressure). If use of noninvasive ventilation strategies did not depend exclusively on illness severity, our study would underestimate the frequency of respiratory failure. Furthermore, there may be inconsistencies among pediatric physicians in coding conditions such as SIRS and sepsis. Even in the clinical setting, a uniform definition of SIRS and sepsis is problematic due to physiologic differences between adults and children of varying age groups.16 An international panel of pediatricians proposed age‐specific definitions for sepsis and SIRS, while acknowledging the paucity of evidence to support some of their recommendations.16 None of the proposed definitions could be applied using administrative data. Limitations in the use of ICD‐9 discharge diagnosis codes to identify children with bacterial meningitis were discussed previously.1
Third, only free‐standing children's hospitals were included in the analysis. It is likely that many children with uncomplicated bacterial meningitis are treated at community hospitals or smaller academic centers. Our study may overestimate the rate of bacterial meningitis‐associated focal infections and systemic complications since participating hospitals serve as regional referral centers. To address the potential for such referral bias, we repeated the analysis while restricting the cohort to those children who had a lumbar puncture performed at the treating facility. No difference in frequency of associated conditions or in‐hospital resource utilization was found between children transferred and children not transferred. Finally, the PHIS database reports billed charge data rather than cost data. Billed data may overestimate the actual economic impact of bacterial meningitis‐associated complications since payers often reimburse at lesser rates. Resource utilization may also vary widely between hospitals and geographic locations as previously shown.15
In conclusion, bacterial meningitis remains an important cause of morbidity in children. Systemic complications such as sepsis and respiratory failure are common. Respiratory failure occurred more commonly among patients with meningococcal meningitis while sepsis occurred more commonly among patients with pneumococcal meningitis. While focal complications are uncommon, children >5 years of age are more likely than younger children to have concomitant mastoiditis or osteomyelitis. The presence of both systemic and focal complications is associated with substantially greater resource utilization than either complication alone.
Dr. Shah had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the analysis. Study concept and design: Shah, Mongelluzzo; acquisition of data: Shah, Mohamad; analysis and interpretation of data: Mongelluzzo, Mohamad, Ten Have, Shah; drafting of the manuscript: Mongelluzzo; critical revision of the manuscript for important intellectual content: Mongelluzzo, Mohamad, Ten Have, Shah; statistical analysis: Shah, Mongelluzzo, Ten Have; obtained funding: Shah, Mongelluzzo; administrative, technical, or material support: Shah; study supervision: Shah.
Appendix
Diagnosis Codes:
Endocarditis: 421.0, 421.1, 421.9
Mastoiditis: 383.0, 383.1, 383.2, 383.8, 383.9
Osteomyelitis: 730.0, 730.1, 730.2, 730.3, 730.7, 730.8, 730.9
Septic arthritis: 711.0, 711.1, 711.2, 711.3, 711.4, 711.5, 711.6, 711.7, 711.8, 711.9
Sepsis: 038.0, 038.1, 038.2, 038.3, 038.4, 038.8, 038.9
Systemic Inflammatory Response Syndrome: 995.92
Pneumonia: 480.0, 480.1, 480.2, 480.3, 480.8, 480.9, 481, 482.0, 482.1, 482.2, 482.3, 482.4, 482.8, 482.9, 483.0, 483.1, 483.8, 484.1, 484.3, 484.5, 484.6, 484.7, 484.8, 485, 486
Procedure Codes:
Endotracheal Intubation: 96.04
Bacterial meningitis can be a devastating disease in children. Overall mortality in children in the United States is 4%1 while long‐term morbidity is present in up to 25%2 of surviving children. The introduction of Haemophilus influenzae type B vaccine, heptavalent pneumococcal conjugate vaccine, and the quadrivalent meningococcal conjugate vaccine has altered the epidemiology of bacterial meningitis.24 Currently, little is known about the epidemiology of systemic complications and associated focal infections that occur during episodes of bacterial meningitis in children and how the presence of such complications affects in‐hospital healthcare resource utilization.
In a randomized controlled trial, the administration of adjuvant corticosteroids was associated with lower mortality rates in adults with bacterial meningitis due to all causes, with the greatest reduction in those with pneumococcal meningitis.5 In a post hoc analysis of data from this trial, reductions in systemic complications, such as septic shock, pneumonia, and acute respiratory distress syndrome, rather than neurologic complications were thought to be the underlying reason for the decrease in mortality associated with pneumococcal meningitis among corticosteroid recipients.6 However, children with bacterial meningitis have an overall 4‐fold lower mortality rate than adults with bacterial meningitis. An even greater difference in mortality rates exists between children and adults with pneumococcal meningitis.1, 5 Children do not benefit from adjuvant corticosteroids as adults do.1, 5, 7 Therefore, the pathogenesis of bacterial meningitis may differ in children from adults and account for the difference in response to adjuvant corticosteroids. Understanding the epidemiology of systemic complications and associated focal infections can aid in the understanding of the pathogenesis of the disease in varying age groups of children.
Previous studies in children have documented the frequency of certain bacterial meningitis‐associated conditions such as respiratory failure, pneumonia, endocarditis, and mastoiditis. Researchers have used the presence of such conditions to predict either mortality or neurologic sequelae in children.810 These studies were small and only included a few types of complications associated with bacterial meningitis. In‐hospital healthcare resource utilization, which may be an important indicator of in‐hospital morbidity, was also not considered as an outcome. In‐hospital morbidity may represent aspects of disease burden not captured by mortality rates or markers for long‐term morbidity alone. In future vaccine efficacy trails or novel therapeutics evaluations, consideration of these associated conditions is important.
The quantification of the use of in‐hospital healthcare utilization is also important for hospital planning and resource allocation in children with bacterial meningitis. A child presenting with bacterial meningitis and a systemic complication or an associated focal infection may require additional resource planning initially to expedite care to enhance recovery and decrease hospital length of stay (LOS).
Our goal was to document the frequency of bacterial meningitis‐associated conditions (systemic complications and associated focal infections) in a large cohort of children with bacterial meningitis treated at tertiary care children's hospitals in the United States, and determine how the presence of such conditions impacted in‐hospital healthcare resource utilization.
Patients and Methods
Data Source
Data for this retrospective cohort study was obtained from the Pediatric Health Information System (PHIS), a national administrative database containing data from 36 freestanding, tertiary care children's hospitals. These hospitals are affiliated with the Child Health Corporation of America (Shawnee Mission, KS), a business alliance of children's hospitals. Data quality and reliability are assured through a joint effort between the Child Health Corporation of America and participating hospitals. For the purposes of external benchmarking, participating hospitals provide discharge data including patient demographics, diagnoses, and procedures. Procedures to assure data validity were described previously.1 Total hospital charges are reported in the PHIS database and adjusted for hospital location using the Centers for Medicare and Medicaid price/wage index. A total of 27 participating hospitals also provide resource utilization data for each hospital discharge (ie, pharmaceutical dispensing, imaging, and laboratory studies); patients from these 27 hospitals were eligible for inclusion in this study. The protocol for the conduct of this study was reviewed and approved by The Children's Hospital of Philadelphia Committees for the Protection of Human Subjects.
Patients
Children less than 18 years of age with bacterial meningitis were eligible for this study if they were discharged from any of the 27 hospitals disclosing resource utilization data between January 1, 2001 and December 31, 2006. Study participants discharged with bacterial meningitis as their primary diagnosis were identified in the PHIS database using International Classification of Diseases, 9th revision, (ICD‐9) discharge diagnosis codes. The study population was limited to children without conditions predisposing to meningitis. Therefore, patients with ventricular shunts prior to the episode of bacterial meningitis were excluded using the following ICD‐9 procedure codes: ventricular shunt replacement (02.42); incision of peritoneum (54.95); removal of ventricular shunts (02.43); and the ICD‐9 discharge diagnosis code for mechanical complication of nervous system device, implant, and graft (996.2). Also, children with comorbid conditions that could predispose to meningitis or increase the likelihood of associated complications such as cancer (hematologic and nonhematologic), primary or secondary immunodeficiencies, prematurity, post‐operative infection, congenital cardiac disease, and sickle cell disease, were excluded from the analysis. Race and ethnicity were self‐reported by patients at time of admission.
Study Definitions
Study participants were identified from the PHIS database using ICD‐9 codes for the primary diagnosis of bacterial meningitis (codes 036.0‐036.1; 320.0‐320.3; 320.7; 320.81‐320.82; 320.89; 320.9). The sensitivity and specificity of ICD‐9 codes in identifying children with bacterial meningitis is unknown, however these codes have been used by previous investigators.1113 Bacterial meningitis associated‐conditions were classified as systemic complications (sepsis, systemic inflammatory response syndrome (SIRS), and respiratory failure) and associated focal infections (septic arthritis, mastoiditis, osteomyelitis, pneumonia and endocarditis). These associated conditions were identified by ICD‐9 discharge and procedural codes as listed in the Appendix (Supporting Information). Bone and joint infections were defined by the presence of either osteomyelitis or septic arthritis.
Primary Outcomes
The primary outcomes of interest were total in‐hospital charges and hospital LOS.
Measured Exposures
The primary exposures of interest were the occurrences of systemic complications, focal infections, or both conditions in children with bacterial meningitis.
Statistical Analysis
The data were initially described using frequencies and percentages for categorical variables and mean, median, interquartile ranges (IQRs) and range values for continuous variables. Analyses of bivariate associations between the outcomes (total in‐hospital charges and length of hospital stay) and potential covariates entailed either chi‐square tests or, for rare events with an expected frequency <5, Fishers Exact Test.
Following bivariate analysis, multivariable models were constructed to assess the adjusted impact of systemic complications and focal infection on total in‐hospital charges and hospital LOS. In evaluating total in‐hospital charges, the charge data were logarithmically transformed to account for the skewed distribution of charges. Multivariable linear regression was then performed to analyze the log transformed charges. The resulting beta‐coefficients were transformed to reflect the percent difference in total hospital charges between children with and without specific complications. In evaluating hospital LOS, negative binomial regression models were employed to estimate incidence rate ratios (IRRs) rather than log‐linear models, as to account for overdispersion in the outcome data. The negative binomial model produced a ratio of lengths of stay or IRR, where a ratio >1 indicates that the risk factor was associated with a longer LOS. The results were presented as percentage change to facilitate interpretation of the results.
The multivariable models were adjusted for the following confounders as determined a priori: age category, race, sex, vancomycin receipt, and adjuvant corticosteroid receipt within the first 24 hours of admission. Tests for interaction between systemic complications or focal infections and age were performed for each of these models. To address the possibility of referral bias which would lead us to overestimate the cost of caring for children with bacterial meningitis with an associated condition, the analyses were repeated restricting the sample to those children who had a lumber puncture performed at a PHIS‐participating hospital. The frequency of systemic complications and focal infections in those who were transferred was no different than in children who were not transferred; therefore the entire cohort was used in the final analyses. Sub‐group analyses were also performed for children identified with pneumococcal and meningococcal meningitis.
The standard errors for all estimates of covariate effects including metastatic effects under the above models were adjusted for the hospital to account for the increased variability due to clustering of individuals within hospitals. Two‐tailed P values <0.05 were considered statistically significant. Actual P values and 95% confidence intervals are reported. Data were analyzed using STATA, Version 10 (Stata Corporation, College Station, TX).
Results
Demographics
There were 2780 children admitted with bacterial meningitis during the study period; 461 (17%) children were excluded because of comorbid illness including malignancy (n = 37), congential heart disease (n = 231), prematurity (n = 104), human immunodeficiency virus infection (n = 4), sickle cell disease (n = 17), and post‐operative infection (n = 68). The remaining 2319 children with bacterial meningitis were included in the analyses. The mean age was 3.6 years (median, 1 year; IQR, 0‐6 years). Approximately half of the children were less than 1 year of age, 23% were 1 to 5 years, and 27% were >5 years. A total of 54% of children were white, 19% were black, 22% were Hispanic, and 5% were of other racial groups. Males accounted for 58% of the children. In this cohort of children, 9% received adjuvant corticosteroids within 24 hours of hospitalization.
Bacterial Meningitis‐Associated Conditions
Overall, 574 (25%) of children with bacterial meningitis suffered a systemic complication or an associated focal infection. Figure 1 shows the types of associated condition stratified by age category. Older children had a higher frequency of associated focal infections while younger children had a higher frequency of systemic complications (P = 0.002, chi‐square test for trend). Figure 2 shows the distribution of specific conditions among children in each age category. The frequency of sepsis decreased with age (P < 0.001, chi‐square test) while the frequency of mastoiditis (P < 0.001, Fisher's exact test) and osteomyelitis (P = 0.005, Fisher's exact test) increased with age. There did not appear to be substantial variability in the proportion of patients with SIRS or sepsis across hospitals, suggesting that hospital‐level variability in coding for these conditions was likely minimal. The median proportion of patients with SIRS by hospital was 2.4% (IQR, 1.2‐4.8%) while the median proportion of patients with sepsis by hospital was 13.4% (IQR, 10.0‐16.9%).


Of the 151 children with an associated focal infection, only 3 (2%) of children had more than 1 infection (1 child had mastoiditis and endocarditis, 1 child had pneumonia and osteomyelitis, and 1 child had pneumonia and endocarditis). However, of the 479 children with systemic complications, 116 (24%) had more than 1 systemic disease (Table 1).
| Types of Systemic Complications | Systemic Complications in All Bacterial Meningitis, n (%) | Systemic Complications in Meningococcal Meningitis, n (%) | Systemic Complications in Pneumococcal Meningitis, n (%) |
|---|---|---|---|
| |||
| Sepsis only | 209 (44) | 16 (21) | 69 (54) |
| Respiratory failure only | 139 (29) | 38 (49) | 30 (24) |
| SIRS only | 15 (3) | 9 (12) | 1 (1) |
| Sepsis and respiratory failure | 52 (11) | 4 (5) | 18 (14) |
| SIRS and sepsis | 27 (6) | 2 (3) | 4 (3) |
| SIRS and respiratory failure | 9 (2) | 5 (6) | 0 (0) |
| SIRS and respiratory failure and sepsis | 28 (6) | 3 (4) | 5 (4) |
| Total systemic complications | 479 | 77 | 127 |
In sub‐group analyses, 269 children had meningococcal meningitis and 470 children had pneumococcal meningitis. Of the children with meningococcal meningitis, 31.2% had a meningitis‐associated condition: 26.4% had a systemic complication, 2.6% had a focal infection, and 2.2% had both conditions. The most common associated conditions in children with meningococcal meningitis were respiratory failure (18.6%; n = 50), sepsis (9.3%; n = 25), and SIRS (7.1%; n = 19). In children with pneumococcal meningitis, 32.3% had a meningitis‐associated complication: 24.7% had a systemic complication, 5.3% had a focal infection, and 2.3% had both conditions. The most common associated conditions in children with pneumococcal meningitis were sepsis (20.4%; n = 96), respiratory failure (11.3%; n = 53), and pneumonia (4.7%; n = 22); mastoiditis was present in 2.3% (n = 11) of children with pneumococcal meningitis. Respiratory failure was more common in meningococcal meningitis (18.6%) than in pneumococcal meningitis (11.3%; P = 0.006). In contrast, sepsis was less common in meningococcal meningitis (9.3%) than in pneumococcal meningitis (20.4%; P < 0.001).
Hospital Charges
Overall, the median charges per hospital ranged from $20,158 to $53,823. In‐hospital charges for children with bacterial meningitis with and without any identified associated conditions are presented in Table 2. In multivariate analyses, the presence of systemic conditions, associated focal infections, or both conditions was independently associated with significantly higher total in‐hospital charges (Table 2). When conditions were considered individually, bone and joint infections (213% increase; 95% CI, 113‐260%), endocarditis (108% increase; 95% CI, 23‐258%), and pneumonia (107% increase; 95% CI, 58‐171%) were associated with the highest increases in total hospital charges (Figure 3). In contrast, SIRS and mastoiditis were not associated with higher hospital charges (Figure 3).
| Charges | LOS | |||
|---|---|---|---|---|
| Median, $ (IQR) | Adjusted Increase,* % (95% CI) | Median, days (IQR) | Adjusted Increase,* % (95% CI) | |
| ||||
| None (n = 1,745) | $27,110 (15,823‐48,307) | Reference** | 9 (6‐14) | Reference |
| Systemic (n = 423) | $66,690 (39,546136,756) | 136 (108269) | 14 (923) | 72 (5196) |
| Focal Infection (n = 95) | $58,016 (29,056125,813) | 118 (77168) | 13 (928) | 78 (40126) |
| Both (n = 56) | $130,744 (62,397299,288) | 351 (237503) | 21.5 (1245) | 211 (142303) |

LOS
The median LOS was 9 days (IQR, 6‐15 days); 5% of children had a LOS >42 days. Table 2 summarizes difference in LOS by the presence and absence of systemic conditions and focal infections. In multivariate analyses, the presence of systemic conditions, associated focal infections, or both conditions was independently associated with a significantly longer LOS (Table 2). When conditions were considered individually, endocarditis (152% increase; 95% CI, 60‐300%) and pneumonia (136% increase; 95% CI, 85‐201%) were associated with the greatest adjusted increases in LOS (Figure 4); only mastoiditis was not associated with an increased LOS compared with those without complications.

Discussion
To our knowledge, this is the first study to examine bacterial meningitis‐associated conditions in children and their impact on in‐hospital resource utilization. We found that 25% of the cohort of children with bacterial meningitis suffered from at least one focal infection or systemic complication. This represents a significant invasive disease burden among children with bacterial meningitis who do not have underlying comorbid conditions. Younger children were more likely to have systemic complications when compared with older children, specifically due to a higher frequency of sepsis in children <1 year. Older children were more likely to have an associated focal infection, specifically due to an increase in mastoiditis and osteomyelitis in children >1 year. Only 2% of children had more than 1 focal infection, while 24% of children had more than 1 systemic complication.
Importantly, the presence of a systemic complication in a child with bacterial meningitis increased their in‐hospital adjusted charges by 136%. The presence of a focal infection increased in‐hospital adjusted charges by 118%. A child with both a systemic complication and a focal infection and had a 351% increase in in‐hospital adjusted charges.
The presence of systemic complications or associated focal infections was significantly associated with higher in‐hospital charges and longer hospital LOS. Most individual meningitis‐associated conditions included in this study were associated with higher in‐hospital charges with the exception of SIRS and mastoiditis. All individual meningitis‐associated conditions were associated with a longer LOS except mastoiditis. This finding is not surprising as the LOS for children with mastoiditis is typically shorter than for children with bacterial meningitis. Glikich et al.14 reported a mean LOS of approximately 8 days for children with mastoiditis. As meningitis in the context of mastoiditis is likely caused by direct extension of infection, patients with meningitis and mastoiditis likely required extended hospitalization to treat meningitis rather than mastoiditis. In contrast, patients with meningitis occurring in the context of metastatic dissemination of infection (eg, endocarditis, pneumonia) often have hemodynamic instability requiring prolonged intensive care support.
A study of children with sepsis found that increasing severity of illness was associated with greater hospital resource utilization.15 Our study shows that this may also be true in children with bacterial meningitis. We found that in children with bacterial meningitis, having systemic complications or an associated focal infection was associated with greater in‐hospital resource utilization. This finding may therefore indicate greater in‐hospital morbidity among children with a bacterial meningitis‐associated condition. Since mortality rates for bacterial meningitis are low in children, in‐hospital morbidity may be a better indicator of disease burden.
Our data show that, in contrast to adults, bacterial meningitis in children is not typically associated with other focal infections. Some focal complications such as mastoiditis and osteomyelitis disproportionately affect older children. These complications are typically accompanied by overt clinical manifestations. Therefore, we believe that the evaluation for the presence of concomitant focal infections can be guided by clinical examination findings and that routine radiologic evaluation for focal complications may not be necessary. Additionally, focal infections tend to occur in the absence of concomitant systemic complications. Of the 151 children with at least 1 associated focal infection, only 37% had a systemic complication. Bacterial meningitis may lie on a continuum of invasive disease depending on the virulence factors of the invading pathogen as well as specific host factors. Understanding the epidemiology of these associated conditions can enhance our understanding of the pathogenesis of bacterial meningitis in children. Understanding why some children suffer from septicemia rather than bacteremia may help in developing novel therapeutics.
There are several limitations to our study. First, since we identified focal infections and systemic complications using billing charges and ICD‐9 discharge diagnosis codes, it was impossible to determine when these conditions represented true complications of bacterial meningitis and when they represented the primary source of infection. Therefore, some of our primary outcomes may represent the cause of meningitis rather than a direct complication. We attempted to minimize such misclassification by limiting the cohort to those with a primary discharge diagnosis of bacterial meningitis though such misclassification is still possible.
Second, the use of ICD‐9 codes to accurately identify systemic complications and associated focal infections is a potential limitation. For example, respiratory failure, defined as the requirement of endotracheal intubation in our study, may not capture children receiving non‐invasive mechanical ventilation (eg, bilevel positive airway pressure). If use of noninvasive ventilation strategies did not depend exclusively on illness severity, our study would underestimate the frequency of respiratory failure. Furthermore, there may be inconsistencies among pediatric physicians in coding conditions such as SIRS and sepsis. Even in the clinical setting, a uniform definition of SIRS and sepsis is problematic due to physiologic differences between adults and children of varying age groups.16 An international panel of pediatricians proposed age‐specific definitions for sepsis and SIRS, while acknowledging the paucity of evidence to support some of their recommendations.16 None of the proposed definitions could be applied using administrative data. Limitations in the use of ICD‐9 discharge diagnosis codes to identify children with bacterial meningitis were discussed previously.1
Third, only free‐standing children's hospitals were included in the analysis. It is likely that many children with uncomplicated bacterial meningitis are treated at community hospitals or smaller academic centers. Our study may overestimate the rate of bacterial meningitis‐associated focal infections and systemic complications since participating hospitals serve as regional referral centers. To address the potential for such referral bias, we repeated the analysis while restricting the cohort to those children who had a lumbar puncture performed at the treating facility. No difference in frequency of associated conditions or in‐hospital resource utilization was found between children transferred and children not transferred. Finally, the PHIS database reports billed charge data rather than cost data. Billed data may overestimate the actual economic impact of bacterial meningitis‐associated complications since payers often reimburse at lesser rates. Resource utilization may also vary widely between hospitals and geographic locations as previously shown.15
In conclusion, bacterial meningitis remains an important cause of morbidity in children. Systemic complications such as sepsis and respiratory failure are common. Respiratory failure occurred more commonly among patients with meningococcal meningitis while sepsis occurred more commonly among patients with pneumococcal meningitis. While focal complications are uncommon, children >5 years of age are more likely than younger children to have concomitant mastoiditis or osteomyelitis. The presence of both systemic and focal complications is associated with substantially greater resource utilization than either complication alone.
Dr. Shah had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the analysis. Study concept and design: Shah, Mongelluzzo; acquisition of data: Shah, Mohamad; analysis and interpretation of data: Mongelluzzo, Mohamad, Ten Have, Shah; drafting of the manuscript: Mongelluzzo; critical revision of the manuscript for important intellectual content: Mongelluzzo, Mohamad, Ten Have, Shah; statistical analysis: Shah, Mongelluzzo, Ten Have; obtained funding: Shah, Mongelluzzo; administrative, technical, or material support: Shah; study supervision: Shah.
Appendix
Diagnosis Codes:
Endocarditis: 421.0, 421.1, 421.9
Mastoiditis: 383.0, 383.1, 383.2, 383.8, 383.9
Osteomyelitis: 730.0, 730.1, 730.2, 730.3, 730.7, 730.8, 730.9
Septic arthritis: 711.0, 711.1, 711.2, 711.3, 711.4, 711.5, 711.6, 711.7, 711.8, 711.9
Sepsis: 038.0, 038.1, 038.2, 038.3, 038.4, 038.8, 038.9
Systemic Inflammatory Response Syndrome: 995.92
Pneumonia: 480.0, 480.1, 480.2, 480.3, 480.8, 480.9, 481, 482.0, 482.1, 482.2, 482.3, 482.4, 482.8, 482.9, 483.0, 483.1, 483.8, 484.1, 484.3, 484.5, 484.6, 484.7, 484.8, 485, 486
Procedure Codes:
Endotracheal Intubation: 96.04
- , , , .Corticosteroids and mortality in children with bacterial meningitis.JAMA.2008;299:2048–2055.
- , , , et al.Bacterial meningitis in the United States in 1995. Active surveillance team.N Engl J Med.1997;337:970–976.
- Progress toward elimination of Haemophilus influenzae type b disease among infants and children–United States, 1987–1995.MMWR Morb Mortal Wkly Rep.1996;45:901–906.
- , .Bacterial meningitis in children.Lancet.2003;361:2139–2148.
- , .Dexamethasone in adults with bacterial meningitis.N Engl J Med.2002;347:1549–1556.
- , .Dexamethasone and pneumococcal meningitis.Ann Intern Med.2004;141:327.
- , , , et al.Dexamethasone in Vietnamese adolescents and adults with bacterial meningitis.N Engl J Med.2007;357:2431–2440.
- , , , , .Bacterial meningitis in an urban area: etiologic study and prognostic factors.Infection.2007;35:406–413.
- , , .Clinical features and prognostic factors in childhood pneumococcal meningitis.J Microbiol Immunol Infect.2008;41:48–53.
- , , .Clinical presentation and prognostic factors of Streptococcus pneumoniae meningitis according to the focus of infection.BMC Infect Dis.2005;5:93.
- , .Trends in invasive pneumococcal disease‐associated hospitalizations.Clin Infect Dis.2006;42:e1–e5.
- , , .Managing meningococcal disease in the United States: Hospital case characteristics and costs by age.Value Health.2006;9:236–243.
- , , , , , .Population‐based analysis of meningococcal disease mortality in the United States: 1990–2002.Pediatr Infect Dis J.2006;25:191–194.
- , , , .A contemporary analysis of acute mastoiditis.Arch Otolaryngol Head Neck Surg.1996;122:135–139.
- , , .Patient and hospital correlates of clinical outcomes and resource utilization in severe pediatric sepsis.Pediatrics.2007;119:487–494.
- , , .International pediatric sepsis consensus conference: definitions for sepsis and organ dysfunction in pediatrics.Pediatr Crit Care Med.2005;6:2–8.
- , , , .Corticosteroids and mortality in children with bacterial meningitis.JAMA.2008;299:2048–2055.
- , , , et al.Bacterial meningitis in the United States in 1995. Active surveillance team.N Engl J Med.1997;337:970–976.
- Progress toward elimination of Haemophilus influenzae type b disease among infants and children–United States, 1987–1995.MMWR Morb Mortal Wkly Rep.1996;45:901–906.
- , .Bacterial meningitis in children.Lancet.2003;361:2139–2148.
- , .Dexamethasone in adults with bacterial meningitis.N Engl J Med.2002;347:1549–1556.
- , .Dexamethasone and pneumococcal meningitis.Ann Intern Med.2004;141:327.
- , , , et al.Dexamethasone in Vietnamese adolescents and adults with bacterial meningitis.N Engl J Med.2007;357:2431–2440.
- , , , , .Bacterial meningitis in an urban area: etiologic study and prognostic factors.Infection.2007;35:406–413.
- , , .Clinical features and prognostic factors in childhood pneumococcal meningitis.J Microbiol Immunol Infect.2008;41:48–53.
- , , .Clinical presentation and prognostic factors of Streptococcus pneumoniae meningitis according to the focus of infection.BMC Infect Dis.2005;5:93.
- , .Trends in invasive pneumococcal disease‐associated hospitalizations.Clin Infect Dis.2006;42:e1–e5.
- , , .Managing meningococcal disease in the United States: Hospital case characteristics and costs by age.Value Health.2006;9:236–243.
- , , , , , .Population‐based analysis of meningococcal disease mortality in the United States: 1990–2002.Pediatr Infect Dis J.2006;25:191–194.
- , , , .A contemporary analysis of acute mastoiditis.Arch Otolaryngol Head Neck Surg.1996;122:135–139.
- , , .Patient and hospital correlates of clinical outcomes and resource utilization in severe pediatric sepsis.Pediatrics.2007;119:487–494.
- , , .International pediatric sepsis consensus conference: definitions for sepsis and organ dysfunction in pediatrics.Pediatr Crit Care Med.2005;6:2–8.
Copyright © 2010 Society of Hospital Medicine
The way to a man's heart is through his liver
A 57‐year‐old man with Hepatitis B and C was scheduled for an outpatient computed tomography (CT)‐guided biopsy of the left lobe of the liver for grading and staging of his liver disease at an outside hospital. Complete metabolic profile (CMP), complete blood count (CBC) and international normalized ratio (INR) were performed and were normal. Following his liver biopsy, the patient became hypotensive and developed shock. He received fluid resuscitation. Clinical exam and an abdominal x‐ray failed to identify a hepatic or intraperitoneal source of bleeding to explain the cause of hypotension. A chest radiograph showed an enlarged cardiac silhouette with clear lung fields. An electrocardiogram (ECG) demonstrated (Figure 1) 1‐mm ST segment elevation in the anterior leads. An emergent echocardiogram showed a significant pericardial effusion with echocardiographic evidence of cardiac tamponade. The patient underwent emergent pericardiocentesis with removal of 400 mL of hemorrhagic fluid. The etiology of the hemorrhagic fluid was thought to be due to myocardial injury secondary to the liver biopsy. Subsequently, the histopathology of the liver biopsy reported (Figure 2) features suggestive of cirrhosis of the liver. Additional tissues from the biopsy included pericardium, myocardium and coronary artery. The patient was transferred to our institution for a cardiac catheterization and coronary angiogram to evaluate his coronary anatomy. The coronary angiogram was normal without any evidence of dye extravasation. A follow‐up echocardiogram and CT scan of the chest showed residual pericardial and pleural fluid. The remainder of the hospital stay was uneventful and he was discharged on day 7 in good condition. He was doing well a month later at the time of his follow‐up visit.0, 0

Discussion
Serious complications of liver biopsy occur in less than 1% of biopsied patients and include intraperitoneal1 or intrahepatic hemorrhage,2, 3 pneumothorax, hemothorax, hemobilia,1 and injury to the gall bladder, colon, kidney and lung. Other rare complications of percutaneous liver biopsy include biliary ascites, bile pleuritis, bile peritonitis, subcutaneous emphysema, pneumoperitoneum, subphrenic abscess, carcinoid crisis, anaphylaxis after biopsy of an echinococcal cyst, pancreatitis due to hemobilia, and breakage of the biopsy needle.1, 4, 5 Bleeding is more common in the elderly and in patients with cirrhosis and liver cancer.1, 6 The most common cause of shock in a patient after liver biopsy is hypovolemia from intrahepatic or intraperitoneal bleeding. The incidence of such complications may be higher than commonly appreciated as these complications are likely underreported.
Ultrasound (US)‐guided and CT‐guided techniques are 2 common methods for performing liver biopsy. US‐guided and CT‐guided liver biopsy each have their own benefits and limitations. The CT‐guided biopsy provides an excellent resolution of the liver architecture and is done from the epigastric region, but uses a static view. The US‐guided liver biopsy, on the other hand, is done from the mid‐axillary intercostal line and is performed with real‐time images.
Our patient's complication was presumably caused by accidental passage of the biopsy needle from the liver into the pericardium during respiratory movements. The coronary artery tissue was most likely not an epicardial vessel as supported by the normal angiogram without evidence of coronary artery perforation.
Our patient had an inadvertent biopsy of cardiac tissue during a liver biopsy resulting in a hemorrhagic pericardial effusion and tamponade. Cardiac tamponade as a complication of liver biopsy and intrathoracic percutaneous procedures has been described, although this has very rarely been reported.7, 8 Fortunately, the patient's coronary artery anatomy was normal and he did well without surgical intervention.
Unusual procedural complications are reported in the literature with the intention of increasing awareness and improving patient safety. Percutaneous liver biopsy is performed to stage and grade liver disease for assessment and treatment. However, as with any invasive procedure, there are potential risks and complications. In patients who present with hypotension and shock following liver biopsy, myocardial injury with cardiac tamponade should be considered in the differential diagnosis.
- , , , .Complications following percutaneous liver biopsy: a multicentre retrospective study on 68,276 biopsies.J Hepatol.1986;2:165–173.
- , , .Intrahepatic hematoma: a complication of percutaneous liver biopsy.Gastroenterology.1974;67:284–289.
- , , , et al.Intrahepatic hematoma resulting in obstructive jaundice. An unusual complication of liver biopsy.Gastroenterology.1978;74(1):124–127.
- , , , .Liver biopsy: its safety and complications as seen at a liver transplant center.Transplantation.1993;55:1087–1090.
- , .Bile peritonitis after liver biopsy: nonsurgical management of a patient with an acute abdomen: a case report with review of the literature.Am J Gastroenterol.1987;82:265–268.
- , .Outcome of patients hospitalized for complications after outpatient liver biopsy.Ann Intern Med.1993;118:96–98.
- , , .Unusual presentation and course of acute cardiac tamponade.J Cardiothorac Vasc Anesth.2007;21:712–714.
- , , .Cardiac tamponade following fine needle aspiration (FNA) of a mediastinal mass.Clin Radiol.1998;53(2):151–152.
A 57‐year‐old man with Hepatitis B and C was scheduled for an outpatient computed tomography (CT)‐guided biopsy of the left lobe of the liver for grading and staging of his liver disease at an outside hospital. Complete metabolic profile (CMP), complete blood count (CBC) and international normalized ratio (INR) were performed and were normal. Following his liver biopsy, the patient became hypotensive and developed shock. He received fluid resuscitation. Clinical exam and an abdominal x‐ray failed to identify a hepatic or intraperitoneal source of bleeding to explain the cause of hypotension. A chest radiograph showed an enlarged cardiac silhouette with clear lung fields. An electrocardiogram (ECG) demonstrated (Figure 1) 1‐mm ST segment elevation in the anterior leads. An emergent echocardiogram showed a significant pericardial effusion with echocardiographic evidence of cardiac tamponade. The patient underwent emergent pericardiocentesis with removal of 400 mL of hemorrhagic fluid. The etiology of the hemorrhagic fluid was thought to be due to myocardial injury secondary to the liver biopsy. Subsequently, the histopathology of the liver biopsy reported (Figure 2) features suggestive of cirrhosis of the liver. Additional tissues from the biopsy included pericardium, myocardium and coronary artery. The patient was transferred to our institution for a cardiac catheterization and coronary angiogram to evaluate his coronary anatomy. The coronary angiogram was normal without any evidence of dye extravasation. A follow‐up echocardiogram and CT scan of the chest showed residual pericardial and pleural fluid. The remainder of the hospital stay was uneventful and he was discharged on day 7 in good condition. He was doing well a month later at the time of his follow‐up visit.0, 0


Discussion
Serious complications of liver biopsy occur in less than 1% of biopsied patients and include intraperitoneal1 or intrahepatic hemorrhage,2, 3 pneumothorax, hemothorax, hemobilia,1 and injury to the gall bladder, colon, kidney and lung. Other rare complications of percutaneous liver biopsy include biliary ascites, bile pleuritis, bile peritonitis, subcutaneous emphysema, pneumoperitoneum, subphrenic abscess, carcinoid crisis, anaphylaxis after biopsy of an echinococcal cyst, pancreatitis due to hemobilia, and breakage of the biopsy needle.1, 4, 5 Bleeding is more common in the elderly and in patients with cirrhosis and liver cancer.1, 6 The most common cause of shock in a patient after liver biopsy is hypovolemia from intrahepatic or intraperitoneal bleeding. The incidence of such complications may be higher than commonly appreciated as these complications are likely underreported.
Ultrasound (US)‐guided and CT‐guided techniques are 2 common methods for performing liver biopsy. US‐guided and CT‐guided liver biopsy each have their own benefits and limitations. The CT‐guided biopsy provides an excellent resolution of the liver architecture and is done from the epigastric region, but uses a static view. The US‐guided liver biopsy, on the other hand, is done from the mid‐axillary intercostal line and is performed with real‐time images.
Our patient's complication was presumably caused by accidental passage of the biopsy needle from the liver into the pericardium during respiratory movements. The coronary artery tissue was most likely not an epicardial vessel as supported by the normal angiogram without evidence of coronary artery perforation.
Our patient had an inadvertent biopsy of cardiac tissue during a liver biopsy resulting in a hemorrhagic pericardial effusion and tamponade. Cardiac tamponade as a complication of liver biopsy and intrathoracic percutaneous procedures has been described, although this has very rarely been reported.7, 8 Fortunately, the patient's coronary artery anatomy was normal and he did well without surgical intervention.
Unusual procedural complications are reported in the literature with the intention of increasing awareness and improving patient safety. Percutaneous liver biopsy is performed to stage and grade liver disease for assessment and treatment. However, as with any invasive procedure, there are potential risks and complications. In patients who present with hypotension and shock following liver biopsy, myocardial injury with cardiac tamponade should be considered in the differential diagnosis.
A 57‐year‐old man with Hepatitis B and C was scheduled for an outpatient computed tomography (CT)‐guided biopsy of the left lobe of the liver for grading and staging of his liver disease at an outside hospital. Complete metabolic profile (CMP), complete blood count (CBC) and international normalized ratio (INR) were performed and were normal. Following his liver biopsy, the patient became hypotensive and developed shock. He received fluid resuscitation. Clinical exam and an abdominal x‐ray failed to identify a hepatic or intraperitoneal source of bleeding to explain the cause of hypotension. A chest radiograph showed an enlarged cardiac silhouette with clear lung fields. An electrocardiogram (ECG) demonstrated (Figure 1) 1‐mm ST segment elevation in the anterior leads. An emergent echocardiogram showed a significant pericardial effusion with echocardiographic evidence of cardiac tamponade. The patient underwent emergent pericardiocentesis with removal of 400 mL of hemorrhagic fluid. The etiology of the hemorrhagic fluid was thought to be due to myocardial injury secondary to the liver biopsy. Subsequently, the histopathology of the liver biopsy reported (Figure 2) features suggestive of cirrhosis of the liver. Additional tissues from the biopsy included pericardium, myocardium and coronary artery. The patient was transferred to our institution for a cardiac catheterization and coronary angiogram to evaluate his coronary anatomy. The coronary angiogram was normal without any evidence of dye extravasation. A follow‐up echocardiogram and CT scan of the chest showed residual pericardial and pleural fluid. The remainder of the hospital stay was uneventful and he was discharged on day 7 in good condition. He was doing well a month later at the time of his follow‐up visit.0, 0


Discussion
Serious complications of liver biopsy occur in less than 1% of biopsied patients and include intraperitoneal1 or intrahepatic hemorrhage,2, 3 pneumothorax, hemothorax, hemobilia,1 and injury to the gall bladder, colon, kidney and lung. Other rare complications of percutaneous liver biopsy include biliary ascites, bile pleuritis, bile peritonitis, subcutaneous emphysema, pneumoperitoneum, subphrenic abscess, carcinoid crisis, anaphylaxis after biopsy of an echinococcal cyst, pancreatitis due to hemobilia, and breakage of the biopsy needle.1, 4, 5 Bleeding is more common in the elderly and in patients with cirrhosis and liver cancer.1, 6 The most common cause of shock in a patient after liver biopsy is hypovolemia from intrahepatic or intraperitoneal bleeding. The incidence of such complications may be higher than commonly appreciated as these complications are likely underreported.
Ultrasound (US)‐guided and CT‐guided techniques are 2 common methods for performing liver biopsy. US‐guided and CT‐guided liver biopsy each have their own benefits and limitations. The CT‐guided biopsy provides an excellent resolution of the liver architecture and is done from the epigastric region, but uses a static view. The US‐guided liver biopsy, on the other hand, is done from the mid‐axillary intercostal line and is performed with real‐time images.
Our patient's complication was presumably caused by accidental passage of the biopsy needle from the liver into the pericardium during respiratory movements. The coronary artery tissue was most likely not an epicardial vessel as supported by the normal angiogram without evidence of coronary artery perforation.
Our patient had an inadvertent biopsy of cardiac tissue during a liver biopsy resulting in a hemorrhagic pericardial effusion and tamponade. Cardiac tamponade as a complication of liver biopsy and intrathoracic percutaneous procedures has been described, although this has very rarely been reported.7, 8 Fortunately, the patient's coronary artery anatomy was normal and he did well without surgical intervention.
Unusual procedural complications are reported in the literature with the intention of increasing awareness and improving patient safety. Percutaneous liver biopsy is performed to stage and grade liver disease for assessment and treatment. However, as with any invasive procedure, there are potential risks and complications. In patients who present with hypotension and shock following liver biopsy, myocardial injury with cardiac tamponade should be considered in the differential diagnosis.
- , , , .Complications following percutaneous liver biopsy: a multicentre retrospective study on 68,276 biopsies.J Hepatol.1986;2:165–173.
- , , .Intrahepatic hematoma: a complication of percutaneous liver biopsy.Gastroenterology.1974;67:284–289.
- , , , et al.Intrahepatic hematoma resulting in obstructive jaundice. An unusual complication of liver biopsy.Gastroenterology.1978;74(1):124–127.
- , , , .Liver biopsy: its safety and complications as seen at a liver transplant center.Transplantation.1993;55:1087–1090.
- , .Bile peritonitis after liver biopsy: nonsurgical management of a patient with an acute abdomen: a case report with review of the literature.Am J Gastroenterol.1987;82:265–268.
- , .Outcome of patients hospitalized for complications after outpatient liver biopsy.Ann Intern Med.1993;118:96–98.
- , , .Unusual presentation and course of acute cardiac tamponade.J Cardiothorac Vasc Anesth.2007;21:712–714.
- , , .Cardiac tamponade following fine needle aspiration (FNA) of a mediastinal mass.Clin Radiol.1998;53(2):151–152.
- , , , .Complications following percutaneous liver biopsy: a multicentre retrospective study on 68,276 biopsies.J Hepatol.1986;2:165–173.
- , , .Intrahepatic hematoma: a complication of percutaneous liver biopsy.Gastroenterology.1974;67:284–289.
- , , , et al.Intrahepatic hematoma resulting in obstructive jaundice. An unusual complication of liver biopsy.Gastroenterology.1978;74(1):124–127.
- , , , .Liver biopsy: its safety and complications as seen at a liver transplant center.Transplantation.1993;55:1087–1090.
- , .Bile peritonitis after liver biopsy: nonsurgical management of a patient with an acute abdomen: a case report with review of the literature.Am J Gastroenterol.1987;82:265–268.
- , .Outcome of patients hospitalized for complications after outpatient liver biopsy.Ann Intern Med.1993;118:96–98.
- , , .Unusual presentation and course of acute cardiac tamponade.J Cardiothorac Vasc Anesth.2007;21:712–714.
- , , .Cardiac tamponade following fine needle aspiration (FNA) of a mediastinal mass.Clin Radiol.1998;53(2):151–152.
Inappropriate Treatment of HCA‐cSSTI
Classically, infections have been categorized as either community‐acquired (CAI) or nosocomial in origin. Until recently, this scheme was thought adequate to capture the differences in the microbiology and outcomes in the corresponding scenarios. However, recent evidence suggests that this distinction may no longer be valid. For example, with the spread and diffusion of healthcare delivery beyond the confines of the hospital along with the increasing use of broad spectrum antibiotics both in and out of the hospital, pathogens such as methicillin‐resistant Staphylococcus aureus (MRSA) and Pseudomonas aeruginosa (PA), traditionally thought to be confined to the hospital, are now seen in patients presenting from the community to the emergency department (ED).1, 2 Reflecting this shift in epidemiology, some national guidelines now recognize healthcare‐associated infection (HCAI) as a distinct entity.3 The concept of HCAI allows the clinician to identify patients who, despite suffering a community onset infection, still may be at risk for a resistant bacterial pathogen. Recent studies in both bloodstream infection and pneumonia have clearly demonstrated that those with HCAI have distinct microbiology and outcomes relative to those with pure CAI.47
Most work focusing on establishing HCAI has not addressed skin and soft tissue infections. These infections, although not often fatal, account for an increasing number of admissions to the hospital.8, 9 In addition, they may be associated with substantial morbidity and cost.8 Given that many pathogens such as S. aureus, which may be resistant to typical antimicrobials used in the ED, are also major culprits in complicated skin and skin structure infections (cSSSI), the HCAI paradigm may apply in cSSSI. Furthermore, because of these patterns of increased resistance, HCA‐cSSSI patients, similar to other HCAI groups, may be at an increased risk of being treated with initially inappropriate antibiotic therapy.7, 10
Since in the setting of other types of infection inappropriate empiric treatment has been shown to be associated with increased mortality and costs,7, 1015 and since indirect evidence suggests a similar impact on healthcare utilization among cSSSI patients,8 we hypothesized that among a cohort of patients hospitalized with a cSSSI, the initial empiric choice of therapy is independently associated with hospital length of stay (LOS). We performed a retrospective cohort study to address this question.
Methods
Study Design
We performed a single‐center retrospective cohort study of patients with cSSSI admitted to the hospital through the ED. All consecutive patients hospitalized between April 2006 and December 2007 meeting predefined inclusion criteria (see below) were enrolled. The study was approved by the Washington University School of Medicine Human Studies Committee, and informed consent was waived. We have previously reported on the characteristics and outcomes of this cohort, including both community‐acquired and HCA‐cSSSI patients.16
Study Cohort
All consecutive patients admitted from the community through the ED between April 2006 and December 2007 at the Barnes‐Jewish Hospital, a 1200‐bed university‐affiliated, urban teaching hospital in St. Louis, MO were included if: (1) they had a diagnosis of a predefined cSSSI (see Appendix Table A1, based on reference 8) and (2) they had a positive microbiology culture obtained within 24 hours of hospital admission. Similar to the work by Edelsberg et al.8 we excluded patients if certain diagnoses and procedures were present (Appendix Table A2). Cases were also excluded if they represented a readmission for the same diagnosis within 30 days of the original hospitalization.
Definitions
HCAI was defined as any cSSSI in a patient with a history of recent hospitalization (within the previous year, consistent with the previous study16), receiving antibiotics prior to admission (previous 90 days), transferring from a nursing home, or needing chronic dialysis. We defined a polymicrobial infection as one with more than one organism, and mixed infection as an infection with both a gram‐positive and a gram‐negative organism. Inappropriate empiric therapy took place if a patient did not receive treatment within 24 hours of the time the culture was obtained with an agent exhibiting in vitro activity against the isolated pathogen(s). In mixed infections, appropriate therapy was treatment within 24 hours of culture being obtained with agent(s) active against all pathogens recovered.
Data Elements
We collected information about multiple baseline demographic and clinical factors including: age, gender, race/ethnicity, comorbidities, the presence of risk factors for HCAI, the presence of bacteremia at admission, and the location of admission (ward vs. intensive care unit [ICU]). Bacteriology data included information on specific bacterium/a recovered from culture, the site of the culture (eg, tissue, blood), susceptibility patterns, and whether the infection was monomicrobial, polymicrobial, or mixed. When blood culture was available and positive, we prioritized this over wound and other cultures and designated the corresponding organism as the culprit in the index infection. Cultures growing our coagulase‐negative S. aureus were excluded as a probable contaminant. Treatment data included information on the choice of the antimicrobial therapy and the timing of its institution relative to the timing of obtaining the culture specimen. The presence of such procedures as incision and drainage (I&D) or debridement was recorded.
Statistical Analyses
Descriptive statistics comparing HCAI patients treated appropriately to those receiving inappropriate empiric coverage based on their clinical, demographic, microbiologic and treatment characteristics were computed. Hospital LOS served as the primary and hospital mortality as the secondary outcomes, comparing patients with HCAI treated appropriately to those treated inappropriately. All continuous variables were compared using Student's t test or the Mann‐Whitney U test as appropriate. All categorical variables were compared using the chi‐square test or Fisher's exact test. To assess the attributable impact of inappropriate therapy in HCAI on the outcomes of interest, general linear models with log transformation were developed to model hospital LOS parameters; all means are presented as geometric means. All potential risk factors significant at the 0.1 level in univariate analyses were entered into the model. All calculations were performed in Stata version 9 (Statacorp, College Station, TX).
Results
Of the 717 patients with culture‐positive cSSSI admitted during the study period, 527 (73.5%) were classified as HCAI. The most common reason for classification as an HCAI was recent hospitalization. Among those with an HCA‐cSSSI, 405 (76.9%) received appropriate empiric treatment, with nearly one‐quarter receiving inappropriate initial coverage. Those receiving inappropriate antibiotic were more likely to be African American, and had a higher likelihood of having end‐stage renal disease (ESRD) than those with appropriate coverage (Table 1). While those patients treated appropriately had higher rates of both cellulitis and abscess as the presenting infection, a substantially higher proportion of those receiving inappropriate initial treatment had a decubitus ulcer (29.5% vs. 10.9%, P <0.001), a device‐associated infection (42.6% vs. 28.6%, P = 0.004), and had evidence of bacteremia (68.9% vs. 57.8%, P = 0.028) than those receiving appropriate empiric coverage (Table 2).
| Inappropriate (n = 122), n (%) | Appropriate (n = 405), n (%) | P Value | |
|---|---|---|---|
| |||
| Age, years | 56.3 18.0 | 53.6 16.7 | 0.147 |
| Gender (F) | 62 (50.8) | 190 (46.9) | 0.449 |
| Race | |||
| Caucasian | 51 (41.8) | 219 (54.1) | 0.048 |
| African American | 68 (55.7) | 178 (43.9) | |
| Other | 3 (2.5) | 8 (2.0) | |
| HCAI risk factors | |||
| Recent hospitalization* | 110 (90.2) | 373 (92.1) | 0.498 |
| Within 90 days | 98 (80.3) | 274 (67.7) | 0.007 |
| >90 and 180 days | 52 (42.6) | 170 (42.0) | 0.899 |
| >180 days and 1 year | 46 (37.7) | 164 (40.5) | 0.581 |
| Prior antibiotics | 26 (21.3) | 90 (22.2) | 0.831 |
| Nursing home resident | 29 (23.8) | 54 (13.3) | 0.006 |
| Hemodialysis | 19 (15.6) | 39 (9.7) | 0.067 |
| Comorbidities | |||
| DM | 40 (37.8) | 128 (31.6) | 0.806 |
| PVD | 5 (4.1) | 15 (3.7) | 0.841 |
| Liver disease | 6 (4.9) | 33 (8.2) | 0.232 |
| Cancer | 21 (17.2) | 85 (21.0) | 0.362 |
| HIV | 1 (0.8) | 12 (3.0) | 0.316 |
| Organ transplant | 2 (1.6) | 8 (2.0) | 1.000 |
| Autoimmune disease | 5 (4.1) | 8 (2.0) | 0.185 |
| ESRD | 22 (18.0) | 46 (11.4) | 0.054 |
| Inappropriate (n = 122), n (%) | Appropriate (n = 405), n (%) | P Value | |
|---|---|---|---|
| |||
| Cellulitis | 28 (23.0) | 171 (42.2) | <0.001 |
| Decubitus ulcer | 36 (29.5) | 44 (10.9) | <0.001 |
| Post‐op wound | 25 (20.5) | 75 (18.5) | 0.626 |
| Device‐associated infection | 52 (42.6) | 116 (28.6) | 0.004 |
| Diabetic foot ulcer | 9 (7.4) | 24 (5.9) | 0.562 |
| Abscess | 22 (18.0) | 108 (26.7) | 0.052 |
| Other* | 2 (1.6) | 17 (4.2) | 0.269 |
| Presence of bacteremia | 84 (68.9) | 234 (57.8) | 0.028 |
The pathogens recovered from the appropriately and inappropriately treated groups are listed in Figure 1. While S. aureus overall was more common among those treated appropriately, the frequency of MRSA did not differ between the groups. Both E. faecalis and E. faecium were recovered more frequently in the inappropriate group, resulting in a similar pattern among the vancomycin‐resistant enterococcal species. Likewise, P. aeruginosa, P. mirabilis, and A. baumannii were all more frequently seen in the group treated inappropriately than in the group getting appropriate empiric coverage. A mixed infection was also more likely to be present among those not exposed (16.5%) than among those exposed (7.5%) to appropriate early therapy (P = 0.001) (Figure 1).
In terms of processes of care and outcomes (Table 3), commensurate with the higher prevalence of abscess in the appropriately treated group, the rate of I&D was significantly higher in this cohort (36.8%) than in the inappropriately treated (23.0%) group (P = 0.005). Need for initial ICU care did not differ as a function of appropriateness of therapy (P = 0.635).
| Inappropriate (n = 122) | Appropriate (n = 405) | P Value | |
|---|---|---|---|
| |||
| I&D/debridement | 28 (23.0%) | 149 (36.8%) | 0.005 |
| I&D in ED | 0 | 7 (1.7) | 0.361 |
| ICU | 9 (7.4%) | 25 (6.2%) | 0.635 |
| Hospital LOS, days | |||
| Median (IQR 25, 75) [Range] | 7.0 (4.2, 13.6) [0.686.6] | 6 (3.3, 10.1) [0.748.3] | 0.026 |
| Hospital mortality | 9 (7.4%) | 26 (6.4%) | 0.710 |
The unadjusted mortality rate was low overall and did not vary based on initial treatment (Table 3). In a generalized linear model with the log‐transformed LOS as the dependent variable, adjusting for multiple potential confounders, initial inappropriate antibiotic therapy had an attributable incremental increase in the hospital LOS of 1.8 days (95% CI, 1.42.3) (Table 4).
| Factor | Attributable LOS (days) | 95% CI | P Value |
|---|---|---|---|
| |||
| Infection type: device | 3.6 | 2.74.8 | <0.001 |
| Infection type: decubitus ulcer | 3.3 | 2.64.2 | <0.001 |
| Infection type: abscess | 2.5 | 1.64.0 | <0.001 |
| Organism: P. mirabilis | 2.2 | 1.43.4 | <0.001 |
| Organism: E. faecalis | 2.1 | 1.72.6 | <0.001 |
| Nursing home resident | 2.1 | 1.62.6 | <0.001 |
| Inappropriate antibiotic | 1.8 | 1.42.3 | <0.001 |
| Race: Non‐Caucasian | 0.31 | 0.240.41 | <0.001 |
| Organism: E. faecium | 0.23 | 0.150.35 | <0.001 |
Because bacteremia is known to be an effect modifier of the relationship between the empiric choice of antibiotic and infection outcomes, we further explored its role in the HCAI cSSSI on the outcomes of interest (Table 5). Similar to the effect detected in the overall cohort, treatment with inappropriate therapy was associated with an increase in the hospital LOS, but not hospital mortality in those with bacteremia, though this phenomenon was observed only among patients with secondary bacteremia, and not among those without (Table 5).
| Bacteremia Present (n = 318) | Bacteremia Absent (n = 209) | |||||
|---|---|---|---|---|---|---|
| I (n = 84) | A (n = 234) | P Value | I (n = 38) | A (n = 171) | P Value | |
| ||||||
| Hospital LOS, days | ||||||
| Mean SD | 14.4 27.5 | 9.8 9.7 | 0.041 | 6.6 6.8 | 6.9 8.2 | 0.761 |
| Median (IQR 25, 75) | 8.8 (5.4, 13.9) | 7.0 (4.3, 11.7) | 4.4 (2.4, 7.7) | 3.9 (2.0, 8.2) | ||
| Hospital mortality | 8 (9.5%) | 24 (10.3%) | 0.848 | 1 (2.6%) | 2 (1.2%) | 0.454 |
Discussion
This retrospective analysis provides evidence that inappropriate empiric antibiotic therapy for HCA‐cSSSI independently prolongs hospital LOS. The impact of inappropriate initial treatment on LOS is independent of many important confounders. In addition, we observed that this effect, while present among patients with secondary bacteremia, is absent among those without a blood stream infection.
To the best of our knowledge, ours is the first cohort study to examine the outcomes associated with inappropriate treatment of a HCAI cSSSI within the context of available microbiology data. Edelsberg et al.8 examined clinical and economic outcomes associated with the failure of the initial treatment of cSSSI. While not specifically focusing on HCAI patients, these authors noted an overall 23% initial therapy failure rate. Among those patients who failed initial therapy, the risk of hospital death was nearly 3‐fold higher (adjusted odds ratio [OR], 2.91; 95% CI, 2.343.62), and they incurred the mean of 5.4 additional hospital days, compared to patients treated successfully with the initial regimen.8 Our study confirms Edelsberg et al.'s8 observation of prolonged hospital LOS in association with treatment failure, and builds upon it by defining the actual LOS increment attributable to inappropriate empiric therapy. It is worth noting that the study by Edelsberg et al.,8 however, lacked explicit definition of the HCAI population and microbiology data, and used treatment failure as a surrogate marker for inappropriate treatment. It is likely these differences between our two studies in the underlying population and exposure definitions that account for the differences in the mortality data between that study and ours.
It is not fundamentally surprising that early exposure to inappropriate empiric therapy alters healthcare resource utilization outcomes for the worse. Others have demonstrated that infection with a resistant organism results in prolongation of hospital LOS and costs. For example, in a large cohort of over 600 surgical hospitalizations requiring treatment for a gram‐negative infection, antibiotic resistance was an independent predictor of increased LOS and costs.15 These authors quantified the incremental burden of early gram‐negative resistance at over $11,000 in hospital costs.15 Unfortunately, the treatment differences for resistant and sensitive organisms were not examined.15 Similarly, Shorr et al. examined risk factors for prolonged hospital LOS and increased costs in a cohort of 291 patients with MRSA sterile site infection.17 Because in this study 23% of the patients received inappropriate empiric therapy, the authors were able to examine the impact of this exposure on utilization outcomes.17 In an adjusted analysis, inappropriate initial treatment was associated with an incremental increase in the LOS of 2.5 days, corresponding to the unadjusted cost differential of nearly $6,000.17 Although focusing on a different population, our results are consistent with these previous observations that antibiotic resistance and early inappropriate therapy affect hospital utilization parameters, in our case by adding nearly 2 days to the hospital LOS.
Our study has a number of limitations. First, as a retrospective cohort study it is prone to various forms of bias, most notably selection bias. To minimize the possibility of such, we established a priori case definitions and enrolled consecutive patients over a specific period of time. Second, as in any observational study, confounding is an issue. We dealt with this statistically by constructing a multivariable regression model; however, the possibility of residual confounding remains. Third, because some of the wound and ulcer cultures likely were obtained with a swab and thus represented colonization, rather than infection, we may have over‐estimated the rate of inappropriate therapy, and this needs to be followed up in future prospective studies. Similarly, we may have over‐estimated the likelihood of inappropriate therapy among polymicrobial and mixed infections as well, given that, for example, a gram‐negative organism may carry a different clinical significance when cultured from blood (infection) than when it is detected in a decubitus ulcer (potential colonization). Fourth, because we limited our cohort to patients without deep‐seated infections, such as necrotizing fasciitis, other procedures were not collected. This omission may have led to either over‐estimation or under‐estimation of the impact of inappropriate therapy on the outcomes of interest.
The fact that our cohort represents a single large urban academic tertiary care medical center may limit the generalizability of our results only to centers that share similar characteristics. Finally, similar to most other studies of this type, ours lacks data on posthospitalization outcomes and for this reason limits itself to hospital outcomes only.
In summary, we have shown that, similar to other populations with HCAI, a substantial proportion (nearly 1/4) of cSSSI patients with HCAI receive inappropriate empiric therapy for their infection, and this early exposure, though not affecting hospital mortality, is associated with a significant prolongation of the hospitalization by as much as 2 days. Studies are needed to refine decision rules for risk‐stratifying patients with cSSSI HCAI in order to determine the probability of infection with a resistant organism. In turn, such instruments at the bedside may assure improved utilization of appropriately targeted empiric therapy that will both optimize individual patient outcomes and reduce the risk of emergence of antimicrobial resistance.
Appendix
| Principal diagnosis code | Description |
|---|---|
| 680 | Carbuncle and furuncle |
| 681 | Cellulitis and abscess of finger and toe |
| 682 | Other cellulitis and abscess |
| 683 | Acute lymphadenitis |
| 685 | Pilonidal cyst with abscess |
| 686 | Other local infections of skin and subcutaneous tissue |
| 707 | Decubitus ulcer |
| 707.1 | Ulcers of lower limbs, except decubitus |
| 707.8 | Chronic ulcer of other specified sites |
| 707.9 | Chronic ulcer of unspecified site |
| 958.3 | Posttraumatic wound infection, not elsewhere classified |
| 996.62 | Infection due to other vascular device, implant, and graft |
| 997.62 | Infection (chronic) of amputation stump |
| 998.5 | Postoperative wound infection |
| Diagnosis code | Description |
|---|---|
| 728.86 | Necrotizing fasciitis |
| 785.4 | Gangrene |
| 686.09 | Ecthyma gangrenosum |
| 730.00730.2 | Osteomyelitis |
| 630677 | Complications of pregnancy, childbirth and puerperium |
| 288.0 | Neutropenia |
| 684 | Impetigo |
| Procedure code | |
| 39.95 | Plasmapheresis |
| 99.71 | Hemoperfusion |
- ,,, et al.Invasive methicillin‐resistant Staphylococcus aureus infections in the United States.JAMA.2007;298:1762–1771.
- ,,, et al.Methicillin‐resistant S. aureus infections among patients in the emergency department.N Engl J Med.2006;17;355:666–674.
- Hospital‐Acquired Pneumonia Guideline Committee of the American Thoracic Society and Infectious Diseases Society of America.Guidelines for the management of adults with hospital‐acquired pneumonia, ventilator‐associated pneumonia, and healthcare‐associated pneumonia.Am J Respir Crit Care Med.2005;171:388–416.
- ,,, et al.Epidemiology and outcomes of health‐care‐associated pneumonia: Results from a large US database of culture‐positive pneumonia.Chest.2005;128:3854–3862.
- ,,, et al.Health care‐associated bloodstream infections in adults: A reason to change the accepted definition of community‐acquired infections.Ann Intern Med.2002;137:791–797.
- ,,,,,.Healthcare‐associated bloodstream infection: A distinct entity? Insights from a large U.S. database.Crit Care Med.2006;34:2588–2595.
- ,,, et al.Health care‐associated pneumonia and community‐acquired pneumonia: a single‐center experience.Antimicrob Agents Chemother.2007;51:3568–3573.
- ,,,,,.Clinical and economic consequences of failure of initial antibiotic therapy for hospitalized patients with complicated skin and skin‐structure infections.Infect Control Hosp Epidemiol.2008;29:160–169.
- ,,, et al.Skin, soft tissue, bone, and joint infections in hospitalized patients: Epidemiology and microbiological, clinical, and economic outcomes.Infect Control Hosp Epidemiol.2007;28:1290–1298.
- ,,, et al.Methicillin‐resistant Staphylococcus aureus sterile‐site infection: The importance of appropriate initial antimicrobial treatment.Crit Care Med.2006;34:2069–2074.
- ,,, et al.The influence of inadequate antimicrobial treatment of bloodstream infections on patient outcomes in the ICU setting.Chest.2000;118:146–155.
- ,.Modification of empiric antibiotic treatment in patients with pneumonia acquired in the intensive care unit.Intensive Care Med.1996;22:387–394.
- ,,, et al.Clinical importance of delays in the initiation of appropriate antibiotic treatment for ventilator‐associated pneumonia.Chest.2002;122:262–268.
- ,,,,.Antimicrobial therapy escalation and hospital mortality among patients with HCAP: A single center experience.Chest.2008;134:963–968.
- ,,, et al.Cost of gram‐negative resistance.Crit Care Med.2007;35:89–95.
- ,,, et al.Epidemiology and outcomes of hospitalizations with complicated skin and skin‐structure infections: implications of healthcare‐associated infection risk factors.Infect Control Hosp Epidemiol.2009;30:1203–1210.
- ,,.Inappropriate therapy for methicillin‐resistant Staphylococcus aureus: resource utilization and cost implications.Crit Care Med.2008;36:2335–2340.
Classically, infections have been categorized as either community‐acquired (CAI) or nosocomial in origin. Until recently, this scheme was thought adequate to capture the differences in the microbiology and outcomes in the corresponding scenarios. However, recent evidence suggests that this distinction may no longer be valid. For example, with the spread and diffusion of healthcare delivery beyond the confines of the hospital along with the increasing use of broad spectrum antibiotics both in and out of the hospital, pathogens such as methicillin‐resistant Staphylococcus aureus (MRSA) and Pseudomonas aeruginosa (PA), traditionally thought to be confined to the hospital, are now seen in patients presenting from the community to the emergency department (ED).1, 2 Reflecting this shift in epidemiology, some national guidelines now recognize healthcare‐associated infection (HCAI) as a distinct entity.3 The concept of HCAI allows the clinician to identify patients who, despite suffering a community onset infection, still may be at risk for a resistant bacterial pathogen. Recent studies in both bloodstream infection and pneumonia have clearly demonstrated that those with HCAI have distinct microbiology and outcomes relative to those with pure CAI.47
Most work focusing on establishing HCAI has not addressed skin and soft tissue infections. These infections, although not often fatal, account for an increasing number of admissions to the hospital.8, 9 In addition, they may be associated with substantial morbidity and cost.8 Given that many pathogens such as S. aureus, which may be resistant to typical antimicrobials used in the ED, are also major culprits in complicated skin and skin structure infections (cSSSI), the HCAI paradigm may apply in cSSSI. Furthermore, because of these patterns of increased resistance, HCA‐cSSSI patients, similar to other HCAI groups, may be at an increased risk of being treated with initially inappropriate antibiotic therapy.7, 10
Since in the setting of other types of infection inappropriate empiric treatment has been shown to be associated with increased mortality and costs,7, 1015 and since indirect evidence suggests a similar impact on healthcare utilization among cSSSI patients,8 we hypothesized that among a cohort of patients hospitalized with a cSSSI, the initial empiric choice of therapy is independently associated with hospital length of stay (LOS). We performed a retrospective cohort study to address this question.
Methods
Study Design
We performed a single‐center retrospective cohort study of patients with cSSSI admitted to the hospital through the ED. All consecutive patients hospitalized between April 2006 and December 2007 meeting predefined inclusion criteria (see below) were enrolled. The study was approved by the Washington University School of Medicine Human Studies Committee, and informed consent was waived. We have previously reported on the characteristics and outcomes of this cohort, including both community‐acquired and HCA‐cSSSI patients.16
Study Cohort
All consecutive patients admitted from the community through the ED between April 2006 and December 2007 at the Barnes‐Jewish Hospital, a 1200‐bed university‐affiliated, urban teaching hospital in St. Louis, MO were included if: (1) they had a diagnosis of a predefined cSSSI (see Appendix Table A1, based on reference 8) and (2) they had a positive microbiology culture obtained within 24 hours of hospital admission. Similar to the work by Edelsberg et al.8 we excluded patients if certain diagnoses and procedures were present (Appendix Table A2). Cases were also excluded if they represented a readmission for the same diagnosis within 30 days of the original hospitalization.
Definitions
HCAI was defined as any cSSSI in a patient with a history of recent hospitalization (within the previous year, consistent with the previous study16), receiving antibiotics prior to admission (previous 90 days), transferring from a nursing home, or needing chronic dialysis. We defined a polymicrobial infection as one with more than one organism, and mixed infection as an infection with both a gram‐positive and a gram‐negative organism. Inappropriate empiric therapy took place if a patient did not receive treatment within 24 hours of the time the culture was obtained with an agent exhibiting in vitro activity against the isolated pathogen(s). In mixed infections, appropriate therapy was treatment within 24 hours of culture being obtained with agent(s) active against all pathogens recovered.
Data Elements
We collected information about multiple baseline demographic and clinical factors including: age, gender, race/ethnicity, comorbidities, the presence of risk factors for HCAI, the presence of bacteremia at admission, and the location of admission (ward vs. intensive care unit [ICU]). Bacteriology data included information on specific bacterium/a recovered from culture, the site of the culture (eg, tissue, blood), susceptibility patterns, and whether the infection was monomicrobial, polymicrobial, or mixed. When blood culture was available and positive, we prioritized this over wound and other cultures and designated the corresponding organism as the culprit in the index infection. Cultures growing our coagulase‐negative S. aureus were excluded as a probable contaminant. Treatment data included information on the choice of the antimicrobial therapy and the timing of its institution relative to the timing of obtaining the culture specimen. The presence of such procedures as incision and drainage (I&D) or debridement was recorded.
Statistical Analyses
Descriptive statistics comparing HCAI patients treated appropriately to those receiving inappropriate empiric coverage based on their clinical, demographic, microbiologic and treatment characteristics were computed. Hospital LOS served as the primary and hospital mortality as the secondary outcomes, comparing patients with HCAI treated appropriately to those treated inappropriately. All continuous variables were compared using Student's t test or the Mann‐Whitney U test as appropriate. All categorical variables were compared using the chi‐square test or Fisher's exact test. To assess the attributable impact of inappropriate therapy in HCAI on the outcomes of interest, general linear models with log transformation were developed to model hospital LOS parameters; all means are presented as geometric means. All potential risk factors significant at the 0.1 level in univariate analyses were entered into the model. All calculations were performed in Stata version 9 (Statacorp, College Station, TX).
Results
Of the 717 patients with culture‐positive cSSSI admitted during the study period, 527 (73.5%) were classified as HCAI. The most common reason for classification as an HCAI was recent hospitalization. Among those with an HCA‐cSSSI, 405 (76.9%) received appropriate empiric treatment, with nearly one‐quarter receiving inappropriate initial coverage. Those receiving inappropriate antibiotic were more likely to be African American, and had a higher likelihood of having end‐stage renal disease (ESRD) than those with appropriate coverage (Table 1). While those patients treated appropriately had higher rates of both cellulitis and abscess as the presenting infection, a substantially higher proportion of those receiving inappropriate initial treatment had a decubitus ulcer (29.5% vs. 10.9%, P <0.001), a device‐associated infection (42.6% vs. 28.6%, P = 0.004), and had evidence of bacteremia (68.9% vs. 57.8%, P = 0.028) than those receiving appropriate empiric coverage (Table 2).
| Inappropriate (n = 122), n (%) | Appropriate (n = 405), n (%) | P Value | |
|---|---|---|---|
| |||
| Age, years | 56.3 18.0 | 53.6 16.7 | 0.147 |
| Gender (F) | 62 (50.8) | 190 (46.9) | 0.449 |
| Race | |||
| Caucasian | 51 (41.8) | 219 (54.1) | 0.048 |
| African American | 68 (55.7) | 178 (43.9) | |
| Other | 3 (2.5) | 8 (2.0) | |
| HCAI risk factors | |||
| Recent hospitalization* | 110 (90.2) | 373 (92.1) | 0.498 |
| Within 90 days | 98 (80.3) | 274 (67.7) | 0.007 |
| >90 and 180 days | 52 (42.6) | 170 (42.0) | 0.899 |
| >180 days and 1 year | 46 (37.7) | 164 (40.5) | 0.581 |
| Prior antibiotics | 26 (21.3) | 90 (22.2) | 0.831 |
| Nursing home resident | 29 (23.8) | 54 (13.3) | 0.006 |
| Hemodialysis | 19 (15.6) | 39 (9.7) | 0.067 |
| Comorbidities | |||
| DM | 40 (37.8) | 128 (31.6) | 0.806 |
| PVD | 5 (4.1) | 15 (3.7) | 0.841 |
| Liver disease | 6 (4.9) | 33 (8.2) | 0.232 |
| Cancer | 21 (17.2) | 85 (21.0) | 0.362 |
| HIV | 1 (0.8) | 12 (3.0) | 0.316 |
| Organ transplant | 2 (1.6) | 8 (2.0) | 1.000 |
| Autoimmune disease | 5 (4.1) | 8 (2.0) | 0.185 |
| ESRD | 22 (18.0) | 46 (11.4) | 0.054 |
| Inappropriate (n = 122), n (%) | Appropriate (n = 405), n (%) | P Value | |
|---|---|---|---|
| |||
| Cellulitis | 28 (23.0) | 171 (42.2) | <0.001 |
| Decubitus ulcer | 36 (29.5) | 44 (10.9) | <0.001 |
| Post‐op wound | 25 (20.5) | 75 (18.5) | 0.626 |
| Device‐associated infection | 52 (42.6) | 116 (28.6) | 0.004 |
| Diabetic foot ulcer | 9 (7.4) | 24 (5.9) | 0.562 |
| Abscess | 22 (18.0) | 108 (26.7) | 0.052 |
| Other* | 2 (1.6) | 17 (4.2) | 0.269 |
| Presence of bacteremia | 84 (68.9) | 234 (57.8) | 0.028 |
The pathogens recovered from the appropriately and inappropriately treated groups are listed in Figure 1. While S. aureus overall was more common among those treated appropriately, the frequency of MRSA did not differ between the groups. Both E. faecalis and E. faecium were recovered more frequently in the inappropriate group, resulting in a similar pattern among the vancomycin‐resistant enterococcal species. Likewise, P. aeruginosa, P. mirabilis, and A. baumannii were all more frequently seen in the group treated inappropriately than in the group getting appropriate empiric coverage. A mixed infection was also more likely to be present among those not exposed (16.5%) than among those exposed (7.5%) to appropriate early therapy (P = 0.001) (Figure 1).

In terms of processes of care and outcomes (Table 3), commensurate with the higher prevalence of abscess in the appropriately treated group, the rate of I&D was significantly higher in this cohort (36.8%) than in the inappropriately treated (23.0%) group (P = 0.005). Need for initial ICU care did not differ as a function of appropriateness of therapy (P = 0.635).
| Inappropriate (n = 122) | Appropriate (n = 405) | P Value | |
|---|---|---|---|
| |||
| I&D/debridement | 28 (23.0%) | 149 (36.8%) | 0.005 |
| I&D in ED | 0 | 7 (1.7) | 0.361 |
| ICU | 9 (7.4%) | 25 (6.2%) | 0.635 |
| Hospital LOS, days | |||
| Median (IQR 25, 75) [Range] | 7.0 (4.2, 13.6) [0.686.6] | 6 (3.3, 10.1) [0.748.3] | 0.026 |
| Hospital mortality | 9 (7.4%) | 26 (6.4%) | 0.710 |
The unadjusted mortality rate was low overall and did not vary based on initial treatment (Table 3). In a generalized linear model with the log‐transformed LOS as the dependent variable, adjusting for multiple potential confounders, initial inappropriate antibiotic therapy had an attributable incremental increase in the hospital LOS of 1.8 days (95% CI, 1.42.3) (Table 4).
| Factor | Attributable LOS (days) | 95% CI | P Value |
|---|---|---|---|
| |||
| Infection type: device | 3.6 | 2.74.8 | <0.001 |
| Infection type: decubitus ulcer | 3.3 | 2.64.2 | <0.001 |
| Infection type: abscess | 2.5 | 1.64.0 | <0.001 |
| Organism: P. mirabilis | 2.2 | 1.43.4 | <0.001 |
| Organism: E. faecalis | 2.1 | 1.72.6 | <0.001 |
| Nursing home resident | 2.1 | 1.62.6 | <0.001 |
| Inappropriate antibiotic | 1.8 | 1.42.3 | <0.001 |
| Race: Non‐Caucasian | 0.31 | 0.240.41 | <0.001 |
| Organism: E. faecium | 0.23 | 0.150.35 | <0.001 |
Because bacteremia is known to be an effect modifier of the relationship between the empiric choice of antibiotic and infection outcomes, we further explored its role in the HCAI cSSSI on the outcomes of interest (Table 5). Similar to the effect detected in the overall cohort, treatment with inappropriate therapy was associated with an increase in the hospital LOS, but not hospital mortality in those with bacteremia, though this phenomenon was observed only among patients with secondary bacteremia, and not among those without (Table 5).
| Bacteremia Present (n = 318) | Bacteremia Absent (n = 209) | |||||
|---|---|---|---|---|---|---|
| I (n = 84) | A (n = 234) | P Value | I (n = 38) | A (n = 171) | P Value | |
| ||||||
| Hospital LOS, days | ||||||
| Mean SD | 14.4 27.5 | 9.8 9.7 | 0.041 | 6.6 6.8 | 6.9 8.2 | 0.761 |
| Median (IQR 25, 75) | 8.8 (5.4, 13.9) | 7.0 (4.3, 11.7) | 4.4 (2.4, 7.7) | 3.9 (2.0, 8.2) | ||
| Hospital mortality | 8 (9.5%) | 24 (10.3%) | 0.848 | 1 (2.6%) | 2 (1.2%) | 0.454 |
Discussion
This retrospective analysis provides evidence that inappropriate empiric antibiotic therapy for HCA‐cSSSI independently prolongs hospital LOS. The impact of inappropriate initial treatment on LOS is independent of many important confounders. In addition, we observed that this effect, while present among patients with secondary bacteremia, is absent among those without a blood stream infection.
To the best of our knowledge, ours is the first cohort study to examine the outcomes associated with inappropriate treatment of a HCAI cSSSI within the context of available microbiology data. Edelsberg et al.8 examined clinical and economic outcomes associated with the failure of the initial treatment of cSSSI. While not specifically focusing on HCAI patients, these authors noted an overall 23% initial therapy failure rate. Among those patients who failed initial therapy, the risk of hospital death was nearly 3‐fold higher (adjusted odds ratio [OR], 2.91; 95% CI, 2.343.62), and they incurred the mean of 5.4 additional hospital days, compared to patients treated successfully with the initial regimen.8 Our study confirms Edelsberg et al.'s8 observation of prolonged hospital LOS in association with treatment failure, and builds upon it by defining the actual LOS increment attributable to inappropriate empiric therapy. It is worth noting that the study by Edelsberg et al.,8 however, lacked explicit definition of the HCAI population and microbiology data, and used treatment failure as a surrogate marker for inappropriate treatment. It is likely these differences between our two studies in the underlying population and exposure definitions that account for the differences in the mortality data between that study and ours.
It is not fundamentally surprising that early exposure to inappropriate empiric therapy alters healthcare resource utilization outcomes for the worse. Others have demonstrated that infection with a resistant organism results in prolongation of hospital LOS and costs. For example, in a large cohort of over 600 surgical hospitalizations requiring treatment for a gram‐negative infection, antibiotic resistance was an independent predictor of increased LOS and costs.15 These authors quantified the incremental burden of early gram‐negative resistance at over $11,000 in hospital costs.15 Unfortunately, the treatment differences for resistant and sensitive organisms were not examined.15 Similarly, Shorr et al. examined risk factors for prolonged hospital LOS and increased costs in a cohort of 291 patients with MRSA sterile site infection.17 Because in this study 23% of the patients received inappropriate empiric therapy, the authors were able to examine the impact of this exposure on utilization outcomes.17 In an adjusted analysis, inappropriate initial treatment was associated with an incremental increase in the LOS of 2.5 days, corresponding to the unadjusted cost differential of nearly $6,000.17 Although focusing on a different population, our results are consistent with these previous observations that antibiotic resistance and early inappropriate therapy affect hospital utilization parameters, in our case by adding nearly 2 days to the hospital LOS.
Our study has a number of limitations. First, as a retrospective cohort study it is prone to various forms of bias, most notably selection bias. To minimize the possibility of such, we established a priori case definitions and enrolled consecutive patients over a specific period of time. Second, as in any observational study, confounding is an issue. We dealt with this statistically by constructing a multivariable regression model; however, the possibility of residual confounding remains. Third, because some of the wound and ulcer cultures likely were obtained with a swab and thus represented colonization, rather than infection, we may have over‐estimated the rate of inappropriate therapy, and this needs to be followed up in future prospective studies. Similarly, we may have over‐estimated the likelihood of inappropriate therapy among polymicrobial and mixed infections as well, given that, for example, a gram‐negative organism may carry a different clinical significance when cultured from blood (infection) than when it is detected in a decubitus ulcer (potential colonization). Fourth, because we limited our cohort to patients without deep‐seated infections, such as necrotizing fasciitis, other procedures were not collected. This omission may have led to either over‐estimation or under‐estimation of the impact of inappropriate therapy on the outcomes of interest.
The fact that our cohort represents a single large urban academic tertiary care medical center may limit the generalizability of our results only to centers that share similar characteristics. Finally, similar to most other studies of this type, ours lacks data on posthospitalization outcomes and for this reason limits itself to hospital outcomes only.
In summary, we have shown that, similar to other populations with HCAI, a substantial proportion (nearly 1/4) of cSSSI patients with HCAI receive inappropriate empiric therapy for their infection, and this early exposure, though not affecting hospital mortality, is associated with a significant prolongation of the hospitalization by as much as 2 days. Studies are needed to refine decision rules for risk‐stratifying patients with cSSSI HCAI in order to determine the probability of infection with a resistant organism. In turn, such instruments at the bedside may assure improved utilization of appropriately targeted empiric therapy that will both optimize individual patient outcomes and reduce the risk of emergence of antimicrobial resistance.
Appendix
| Principal diagnosis code | Description |
|---|---|
| 680 | Carbuncle and furuncle |
| 681 | Cellulitis and abscess of finger and toe |
| 682 | Other cellulitis and abscess |
| 683 | Acute lymphadenitis |
| 685 | Pilonidal cyst with abscess |
| 686 | Other local infections of skin and subcutaneous tissue |
| 707 | Decubitus ulcer |
| 707.1 | Ulcers of lower limbs, except decubitus |
| 707.8 | Chronic ulcer of other specified sites |
| 707.9 | Chronic ulcer of unspecified site |
| 958.3 | Posttraumatic wound infection, not elsewhere classified |
| 996.62 | Infection due to other vascular device, implant, and graft |
| 997.62 | Infection (chronic) of amputation stump |
| 998.5 | Postoperative wound infection |
| Diagnosis code | Description |
|---|---|
| 728.86 | Necrotizing fasciitis |
| 785.4 | Gangrene |
| 686.09 | Ecthyma gangrenosum |
| 730.00730.2 | Osteomyelitis |
| 630677 | Complications of pregnancy, childbirth and puerperium |
| 288.0 | Neutropenia |
| 684 | Impetigo |
| Procedure code | |
| 39.95 | Plasmapheresis |
| 99.71 | Hemoperfusion |
Classically, infections have been categorized as either community‐acquired (CAI) or nosocomial in origin. Until recently, this scheme was thought adequate to capture the differences in the microbiology and outcomes in the corresponding scenarios. However, recent evidence suggests that this distinction may no longer be valid. For example, with the spread and diffusion of healthcare delivery beyond the confines of the hospital along with the increasing use of broad spectrum antibiotics both in and out of the hospital, pathogens such as methicillin‐resistant Staphylococcus aureus (MRSA) and Pseudomonas aeruginosa (PA), traditionally thought to be confined to the hospital, are now seen in patients presenting from the community to the emergency department (ED).1, 2 Reflecting this shift in epidemiology, some national guidelines now recognize healthcare‐associated infection (HCAI) as a distinct entity.3 The concept of HCAI allows the clinician to identify patients who, despite suffering a community onset infection, still may be at risk for a resistant bacterial pathogen. Recent studies in both bloodstream infection and pneumonia have clearly demonstrated that those with HCAI have distinct microbiology and outcomes relative to those with pure CAI.47
Most work focusing on establishing HCAI has not addressed skin and soft tissue infections. These infections, although not often fatal, account for an increasing number of admissions to the hospital.8, 9 In addition, they may be associated with substantial morbidity and cost.8 Given that many pathogens such as S. aureus, which may be resistant to typical antimicrobials used in the ED, are also major culprits in complicated skin and skin structure infections (cSSSI), the HCAI paradigm may apply in cSSSI. Furthermore, because of these patterns of increased resistance, HCA‐cSSSI patients, similar to other HCAI groups, may be at an increased risk of being treated with initially inappropriate antibiotic therapy.7, 10
Since in the setting of other types of infection inappropriate empiric treatment has been shown to be associated with increased mortality and costs,7, 1015 and since indirect evidence suggests a similar impact on healthcare utilization among cSSSI patients,8 we hypothesized that among a cohort of patients hospitalized with a cSSSI, the initial empiric choice of therapy is independently associated with hospital length of stay (LOS). We performed a retrospective cohort study to address this question.
Methods
Study Design
We performed a single‐center retrospective cohort study of patients with cSSSI admitted to the hospital through the ED. All consecutive patients hospitalized between April 2006 and December 2007 meeting predefined inclusion criteria (see below) were enrolled. The study was approved by the Washington University School of Medicine Human Studies Committee, and informed consent was waived. We have previously reported on the characteristics and outcomes of this cohort, including both community‐acquired and HCA‐cSSSI patients.16
Study Cohort
All consecutive patients admitted from the community through the ED between April 2006 and December 2007 at the Barnes‐Jewish Hospital, a 1200‐bed university‐affiliated, urban teaching hospital in St. Louis, MO were included if: (1) they had a diagnosis of a predefined cSSSI (see Appendix Table A1, based on reference 8) and (2) they had a positive microbiology culture obtained within 24 hours of hospital admission. Similar to the work by Edelsberg et al.8 we excluded patients if certain diagnoses and procedures were present (Appendix Table A2). Cases were also excluded if they represented a readmission for the same diagnosis within 30 days of the original hospitalization.
Definitions
HCAI was defined as any cSSSI in a patient with a history of recent hospitalization (within the previous year, consistent with the previous study16), receiving antibiotics prior to admission (previous 90 days), transferring from a nursing home, or needing chronic dialysis. We defined a polymicrobial infection as one with more than one organism, and mixed infection as an infection with both a gram‐positive and a gram‐negative organism. Inappropriate empiric therapy took place if a patient did not receive treatment within 24 hours of the time the culture was obtained with an agent exhibiting in vitro activity against the isolated pathogen(s). In mixed infections, appropriate therapy was treatment within 24 hours of culture being obtained with agent(s) active against all pathogens recovered.
Data Elements
We collected information about multiple baseline demographic and clinical factors including: age, gender, race/ethnicity, comorbidities, the presence of risk factors for HCAI, the presence of bacteremia at admission, and the location of admission (ward vs. intensive care unit [ICU]). Bacteriology data included information on specific bacterium/a recovered from culture, the site of the culture (eg, tissue, blood), susceptibility patterns, and whether the infection was monomicrobial, polymicrobial, or mixed. When blood culture was available and positive, we prioritized this over wound and other cultures and designated the corresponding organism as the culprit in the index infection. Cultures growing our coagulase‐negative S. aureus were excluded as a probable contaminant. Treatment data included information on the choice of the antimicrobial therapy and the timing of its institution relative to the timing of obtaining the culture specimen. The presence of such procedures as incision and drainage (I&D) or debridement was recorded.
Statistical Analyses
Descriptive statistics comparing HCAI patients treated appropriately to those receiving inappropriate empiric coverage based on their clinical, demographic, microbiologic and treatment characteristics were computed. Hospital LOS served as the primary and hospital mortality as the secondary outcomes, comparing patients with HCAI treated appropriately to those treated inappropriately. All continuous variables were compared using Student's t test or the Mann‐Whitney U test as appropriate. All categorical variables were compared using the chi‐square test or Fisher's exact test. To assess the attributable impact of inappropriate therapy in HCAI on the outcomes of interest, general linear models with log transformation were developed to model hospital LOS parameters; all means are presented as geometric means. All potential risk factors significant at the 0.1 level in univariate analyses were entered into the model. All calculations were performed in Stata version 9 (Statacorp, College Station, TX).
Results
Of the 717 patients with culture‐positive cSSSI admitted during the study period, 527 (73.5%) were classified as HCAI. The most common reason for classification as an HCAI was recent hospitalization. Among those with an HCA‐cSSSI, 405 (76.9%) received appropriate empiric treatment, with nearly one‐quarter receiving inappropriate initial coverage. Those receiving inappropriate antibiotic were more likely to be African American, and had a higher likelihood of having end‐stage renal disease (ESRD) than those with appropriate coverage (Table 1). While those patients treated appropriately had higher rates of both cellulitis and abscess as the presenting infection, a substantially higher proportion of those receiving inappropriate initial treatment had a decubitus ulcer (29.5% vs. 10.9%, P <0.001), a device‐associated infection (42.6% vs. 28.6%, P = 0.004), and had evidence of bacteremia (68.9% vs. 57.8%, P = 0.028) than those receiving appropriate empiric coverage (Table 2).
| Inappropriate (n = 122), n (%) | Appropriate (n = 405), n (%) | P Value | |
|---|---|---|---|
| |||
| Age, years | 56.3 18.0 | 53.6 16.7 | 0.147 |
| Gender (F) | 62 (50.8) | 190 (46.9) | 0.449 |
| Race | |||
| Caucasian | 51 (41.8) | 219 (54.1) | 0.048 |
| African American | 68 (55.7) | 178 (43.9) | |
| Other | 3 (2.5) | 8 (2.0) | |
| HCAI risk factors | |||
| Recent hospitalization* | 110 (90.2) | 373 (92.1) | 0.498 |
| Within 90 days | 98 (80.3) | 274 (67.7) | 0.007 |
| >90 and 180 days | 52 (42.6) | 170 (42.0) | 0.899 |
| >180 days and 1 year | 46 (37.7) | 164 (40.5) | 0.581 |
| Prior antibiotics | 26 (21.3) | 90 (22.2) | 0.831 |
| Nursing home resident | 29 (23.8) | 54 (13.3) | 0.006 |
| Hemodialysis | 19 (15.6) | 39 (9.7) | 0.067 |
| Comorbidities | |||
| DM | 40 (37.8) | 128 (31.6) | 0.806 |
| PVD | 5 (4.1) | 15 (3.7) | 0.841 |
| Liver disease | 6 (4.9) | 33 (8.2) | 0.232 |
| Cancer | 21 (17.2) | 85 (21.0) | 0.362 |
| HIV | 1 (0.8) | 12 (3.0) | 0.316 |
| Organ transplant | 2 (1.6) | 8 (2.0) | 1.000 |
| Autoimmune disease | 5 (4.1) | 8 (2.0) | 0.185 |
| ESRD | 22 (18.0) | 46 (11.4) | 0.054 |
| Inappropriate (n = 122), n (%) | Appropriate (n = 405), n (%) | P Value | |
|---|---|---|---|
| |||
| Cellulitis | 28 (23.0) | 171 (42.2) | <0.001 |
| Decubitus ulcer | 36 (29.5) | 44 (10.9) | <0.001 |
| Post‐op wound | 25 (20.5) | 75 (18.5) | 0.626 |
| Device‐associated infection | 52 (42.6) | 116 (28.6) | 0.004 |
| Diabetic foot ulcer | 9 (7.4) | 24 (5.9) | 0.562 |
| Abscess | 22 (18.0) | 108 (26.7) | 0.052 |
| Other* | 2 (1.6) | 17 (4.2) | 0.269 |
| Presence of bacteremia | 84 (68.9) | 234 (57.8) | 0.028 |
The pathogens recovered from the appropriately and inappropriately treated groups are listed in Figure 1. While S. aureus overall was more common among those treated appropriately, the frequency of MRSA did not differ between the groups. Both E. faecalis and E. faecium were recovered more frequently in the inappropriate group, resulting in a similar pattern among the vancomycin‐resistant enterococcal species. Likewise, P. aeruginosa, P. mirabilis, and A. baumannii were all more frequently seen in the group treated inappropriately than in the group getting appropriate empiric coverage. A mixed infection was also more likely to be present among those not exposed (16.5%) than among those exposed (7.5%) to appropriate early therapy (P = 0.001) (Figure 1).

In terms of processes of care and outcomes (Table 3), commensurate with the higher prevalence of abscess in the appropriately treated group, the rate of I&D was significantly higher in this cohort (36.8%) than in the inappropriately treated (23.0%) group (P = 0.005). Need for initial ICU care did not differ as a function of appropriateness of therapy (P = 0.635).
| Inappropriate (n = 122) | Appropriate (n = 405) | P Value | |
|---|---|---|---|
| |||
| I&D/debridement | 28 (23.0%) | 149 (36.8%) | 0.005 |
| I&D in ED | 0 | 7 (1.7) | 0.361 |
| ICU | 9 (7.4%) | 25 (6.2%) | 0.635 |
| Hospital LOS, days | |||
| Median (IQR 25, 75) [Range] | 7.0 (4.2, 13.6) [0.686.6] | 6 (3.3, 10.1) [0.748.3] | 0.026 |
| Hospital mortality | 9 (7.4%) | 26 (6.4%) | 0.710 |
The unadjusted mortality rate was low overall and did not vary based on initial treatment (Table 3). In a generalized linear model with the log‐transformed LOS as the dependent variable, adjusting for multiple potential confounders, initial inappropriate antibiotic therapy had an attributable incremental increase in the hospital LOS of 1.8 days (95% CI, 1.42.3) (Table 4).
| Factor | Attributable LOS (days) | 95% CI | P Value |
|---|---|---|---|
| |||
| Infection type: device | 3.6 | 2.74.8 | <0.001 |
| Infection type: decubitus ulcer | 3.3 | 2.64.2 | <0.001 |
| Infection type: abscess | 2.5 | 1.64.0 | <0.001 |
| Organism: P. mirabilis | 2.2 | 1.43.4 | <0.001 |
| Organism: E. faecalis | 2.1 | 1.72.6 | <0.001 |
| Nursing home resident | 2.1 | 1.62.6 | <0.001 |
| Inappropriate antibiotic | 1.8 | 1.42.3 | <0.001 |
| Race: Non‐Caucasian | 0.31 | 0.240.41 | <0.001 |
| Organism: E. faecium | 0.23 | 0.150.35 | <0.001 |
Because bacteremia is known to be an effect modifier of the relationship between the empiric choice of antibiotic and infection outcomes, we further explored its role in the HCAI cSSSI on the outcomes of interest (Table 5). Similar to the effect detected in the overall cohort, treatment with inappropriate therapy was associated with an increase in the hospital LOS, but not hospital mortality in those with bacteremia, though this phenomenon was observed only among patients with secondary bacteremia, and not among those without (Table 5).
| Bacteremia Present (n = 318) | Bacteremia Absent (n = 209) | |||||
|---|---|---|---|---|---|---|
| I (n = 84) | A (n = 234) | P Value | I (n = 38) | A (n = 171) | P Value | |
| ||||||
| Hospital LOS, days | ||||||
| Mean SD | 14.4 27.5 | 9.8 9.7 | 0.041 | 6.6 6.8 | 6.9 8.2 | 0.761 |
| Median (IQR 25, 75) | 8.8 (5.4, 13.9) | 7.0 (4.3, 11.7) | 4.4 (2.4, 7.7) | 3.9 (2.0, 8.2) | ||
| Hospital mortality | 8 (9.5%) | 24 (10.3%) | 0.848 | 1 (2.6%) | 2 (1.2%) | 0.454 |
Discussion
This retrospective analysis provides evidence that inappropriate empiric antibiotic therapy for HCA‐cSSSI independently prolongs hospital LOS. The impact of inappropriate initial treatment on LOS is independent of many important confounders. In addition, we observed that this effect, while present among patients with secondary bacteremia, is absent among those without a blood stream infection.
To the best of our knowledge, ours is the first cohort study to examine the outcomes associated with inappropriate treatment of a HCAI cSSSI within the context of available microbiology data. Edelsberg et al.8 examined clinical and economic outcomes associated with the failure of the initial treatment of cSSSI. While not specifically focusing on HCAI patients, these authors noted an overall 23% initial therapy failure rate. Among those patients who failed initial therapy, the risk of hospital death was nearly 3‐fold higher (adjusted odds ratio [OR], 2.91; 95% CI, 2.343.62), and they incurred the mean of 5.4 additional hospital days, compared to patients treated successfully with the initial regimen.8 Our study confirms Edelsberg et al.'s8 observation of prolonged hospital LOS in association with treatment failure, and builds upon it by defining the actual LOS increment attributable to inappropriate empiric therapy. It is worth noting that the study by Edelsberg et al.,8 however, lacked explicit definition of the HCAI population and microbiology data, and used treatment failure as a surrogate marker for inappropriate treatment. It is likely these differences between our two studies in the underlying population and exposure definitions that account for the differences in the mortality data between that study and ours.
It is not fundamentally surprising that early exposure to inappropriate empiric therapy alters healthcare resource utilization outcomes for the worse. Others have demonstrated that infection with a resistant organism results in prolongation of hospital LOS and costs. For example, in a large cohort of over 600 surgical hospitalizations requiring treatment for a gram‐negative infection, antibiotic resistance was an independent predictor of increased LOS and costs.15 These authors quantified the incremental burden of early gram‐negative resistance at over $11,000 in hospital costs.15 Unfortunately, the treatment differences for resistant and sensitive organisms were not examined.15 Similarly, Shorr et al. examined risk factors for prolonged hospital LOS and increased costs in a cohort of 291 patients with MRSA sterile site infection.17 Because in this study 23% of the patients received inappropriate empiric therapy, the authors were able to examine the impact of this exposure on utilization outcomes.17 In an adjusted analysis, inappropriate initial treatment was associated with an incremental increase in the LOS of 2.5 days, corresponding to the unadjusted cost differential of nearly $6,000.17 Although focusing on a different population, our results are consistent with these previous observations that antibiotic resistance and early inappropriate therapy affect hospital utilization parameters, in our case by adding nearly 2 days to the hospital LOS.
Our study has a number of limitations. First, as a retrospective cohort study it is prone to various forms of bias, most notably selection bias. To minimize the possibility of such, we established a priori case definitions and enrolled consecutive patients over a specific period of time. Second, as in any observational study, confounding is an issue. We dealt with this statistically by constructing a multivariable regression model; however, the possibility of residual confounding remains. Third, because some of the wound and ulcer cultures likely were obtained with a swab and thus represented colonization, rather than infection, we may have over‐estimated the rate of inappropriate therapy, and this needs to be followed up in future prospective studies. Similarly, we may have over‐estimated the likelihood of inappropriate therapy among polymicrobial and mixed infections as well, given that, for example, a gram‐negative organism may carry a different clinical significance when cultured from blood (infection) than when it is detected in a decubitus ulcer (potential colonization). Fourth, because we limited our cohort to patients without deep‐seated infections, such as necrotizing fasciitis, other procedures were not collected. This omission may have led to either over‐estimation or under‐estimation of the impact of inappropriate therapy on the outcomes of interest.
The fact that our cohort represents a single large urban academic tertiary care medical center may limit the generalizability of our results only to centers that share similar characteristics. Finally, similar to most other studies of this type, ours lacks data on posthospitalization outcomes and for this reason limits itself to hospital outcomes only.
In summary, we have shown that, similar to other populations with HCAI, a substantial proportion (nearly 1/4) of cSSSI patients with HCAI receive inappropriate empiric therapy for their infection, and this early exposure, though not affecting hospital mortality, is associated with a significant prolongation of the hospitalization by as much as 2 days. Studies are needed to refine decision rules for risk‐stratifying patients with cSSSI HCAI in order to determine the probability of infection with a resistant organism. In turn, such instruments at the bedside may assure improved utilization of appropriately targeted empiric therapy that will both optimize individual patient outcomes and reduce the risk of emergence of antimicrobial resistance.
Appendix
| Principal diagnosis code | Description |
|---|---|
| 680 | Carbuncle and furuncle |
| 681 | Cellulitis and abscess of finger and toe |
| 682 | Other cellulitis and abscess |
| 683 | Acute lymphadenitis |
| 685 | Pilonidal cyst with abscess |
| 686 | Other local infections of skin and subcutaneous tissue |
| 707 | Decubitus ulcer |
| 707.1 | Ulcers of lower limbs, except decubitus |
| 707.8 | Chronic ulcer of other specified sites |
| 707.9 | Chronic ulcer of unspecified site |
| 958.3 | Posttraumatic wound infection, not elsewhere classified |
| 996.62 | Infection due to other vascular device, implant, and graft |
| 997.62 | Infection (chronic) of amputation stump |
| 998.5 | Postoperative wound infection |
| Diagnosis code | Description |
|---|---|
| 728.86 | Necrotizing fasciitis |
| 785.4 | Gangrene |
| 686.09 | Ecthyma gangrenosum |
| 730.00730.2 | Osteomyelitis |
| 630677 | Complications of pregnancy, childbirth and puerperium |
| 288.0 | Neutropenia |
| 684 | Impetigo |
| Procedure code | |
| 39.95 | Plasmapheresis |
| 99.71 | Hemoperfusion |
- ,,, et al.Invasive methicillin‐resistant Staphylococcus aureus infections in the United States.JAMA.2007;298:1762–1771.
- ,,, et al.Methicillin‐resistant S. aureus infections among patients in the emergency department.N Engl J Med.2006;17;355:666–674.
- Hospital‐Acquired Pneumonia Guideline Committee of the American Thoracic Society and Infectious Diseases Society of America.Guidelines for the management of adults with hospital‐acquired pneumonia, ventilator‐associated pneumonia, and healthcare‐associated pneumonia.Am J Respir Crit Care Med.2005;171:388–416.
- ,,, et al.Epidemiology and outcomes of health‐care‐associated pneumonia: Results from a large US database of culture‐positive pneumonia.Chest.2005;128:3854–3862.
- ,,, et al.Health care‐associated bloodstream infections in adults: A reason to change the accepted definition of community‐acquired infections.Ann Intern Med.2002;137:791–797.
- ,,,,,.Healthcare‐associated bloodstream infection: A distinct entity? Insights from a large U.S. database.Crit Care Med.2006;34:2588–2595.
- ,,, et al.Health care‐associated pneumonia and community‐acquired pneumonia: a single‐center experience.Antimicrob Agents Chemother.2007;51:3568–3573.
- ,,,,,.Clinical and economic consequences of failure of initial antibiotic therapy for hospitalized patients with complicated skin and skin‐structure infections.Infect Control Hosp Epidemiol.2008;29:160–169.
- ,,, et al.Skin, soft tissue, bone, and joint infections in hospitalized patients: Epidemiology and microbiological, clinical, and economic outcomes.Infect Control Hosp Epidemiol.2007;28:1290–1298.
- ,,, et al.Methicillin‐resistant Staphylococcus aureus sterile‐site infection: The importance of appropriate initial antimicrobial treatment.Crit Care Med.2006;34:2069–2074.
- ,,, et al.The influence of inadequate antimicrobial treatment of bloodstream infections on patient outcomes in the ICU setting.Chest.2000;118:146–155.
- ,.Modification of empiric antibiotic treatment in patients with pneumonia acquired in the intensive care unit.Intensive Care Med.1996;22:387–394.
- ,,, et al.Clinical importance of delays in the initiation of appropriate antibiotic treatment for ventilator‐associated pneumonia.Chest.2002;122:262–268.
- ,,,,.Antimicrobial therapy escalation and hospital mortality among patients with HCAP: A single center experience.Chest.2008;134:963–968.
- ,,, et al.Cost of gram‐negative resistance.Crit Care Med.2007;35:89–95.
- ,,, et al.Epidemiology and outcomes of hospitalizations with complicated skin and skin‐structure infections: implications of healthcare‐associated infection risk factors.Infect Control Hosp Epidemiol.2009;30:1203–1210.
- ,,.Inappropriate therapy for methicillin‐resistant Staphylococcus aureus: resource utilization and cost implications.Crit Care Med.2008;36:2335–2340.
- ,,, et al.Invasive methicillin‐resistant Staphylococcus aureus infections in the United States.JAMA.2007;298:1762–1771.
- ,,, et al.Methicillin‐resistant S. aureus infections among patients in the emergency department.N Engl J Med.2006;17;355:666–674.
- Hospital‐Acquired Pneumonia Guideline Committee of the American Thoracic Society and Infectious Diseases Society of America.Guidelines for the management of adults with hospital‐acquired pneumonia, ventilator‐associated pneumonia, and healthcare‐associated pneumonia.Am J Respir Crit Care Med.2005;171:388–416.
- ,,, et al.Epidemiology and outcomes of health‐care‐associated pneumonia: Results from a large US database of culture‐positive pneumonia.Chest.2005;128:3854–3862.
- ,,, et al.Health care‐associated bloodstream infections in adults: A reason to change the accepted definition of community‐acquired infections.Ann Intern Med.2002;137:791–797.
- ,,,,,.Healthcare‐associated bloodstream infection: A distinct entity? Insights from a large U.S. database.Crit Care Med.2006;34:2588–2595.
- ,,, et al.Health care‐associated pneumonia and community‐acquired pneumonia: a single‐center experience.Antimicrob Agents Chemother.2007;51:3568–3573.
- ,,,,,.Clinical and economic consequences of failure of initial antibiotic therapy for hospitalized patients with complicated skin and skin‐structure infections.Infect Control Hosp Epidemiol.2008;29:160–169.
- ,,, et al.Skin, soft tissue, bone, and joint infections in hospitalized patients: Epidemiology and microbiological, clinical, and economic outcomes.Infect Control Hosp Epidemiol.2007;28:1290–1298.
- ,,, et al.Methicillin‐resistant Staphylococcus aureus sterile‐site infection: The importance of appropriate initial antimicrobial treatment.Crit Care Med.2006;34:2069–2074.
- ,,, et al.The influence of inadequate antimicrobial treatment of bloodstream infections on patient outcomes in the ICU setting.Chest.2000;118:146–155.
- ,.Modification of empiric antibiotic treatment in patients with pneumonia acquired in the intensive care unit.Intensive Care Med.1996;22:387–394.
- ,,, et al.Clinical importance of delays in the initiation of appropriate antibiotic treatment for ventilator‐associated pneumonia.Chest.2002;122:262–268.
- ,,,,.Antimicrobial therapy escalation and hospital mortality among patients with HCAP: A single center experience.Chest.2008;134:963–968.
- ,,, et al.Cost of gram‐negative resistance.Crit Care Med.2007;35:89–95.
- ,,, et al.Epidemiology and outcomes of hospitalizations with complicated skin and skin‐structure infections: implications of healthcare‐associated infection risk factors.Infect Control Hosp Epidemiol.2009;30:1203–1210.
- ,,.Inappropriate therapy for methicillin‐resistant Staphylococcus aureus: resource utilization and cost implications.Crit Care Med.2008;36:2335–2340.
Copyright © 2010 Society of Hospital Medicine
Clinical and Economic Outcomes of Community‐Acquired Pneumonia
Staphylococcus aureus (S. aureus) is well recognized as a major cause of nosocomial and healthcare‐associated pneumonia (HCAP). These infections are associated with substantial morbidity and mortality, and generate high medical costs.14 The significance of S. aureus in community‐acquired pneumonia (CAP) is less clear. Historically, S. aureus has not been considered a common pathogen in CAP, usually arising in association with or following influenza or an influenza‐like syndrome.58 However, with the increasing prevalence of methicillin‐resistant S. aureus (MRSA) as a cause of HCAPand most recently, serious community‐acquired non‐pulmonary infectionsinterest in this pathogen and its impact beyond the hospital have been expanding. During the 2003 to 2004 influenza season, 15 cases of influenza‐associated MRSA CAP were identified.6 In January 2007, the US Centers for Disease Control and Prevention (CDC) received reports of an additional 10 cases of severe MRSA CAP, resulting in six deaths, among previously healthy children and adults in Louisiana and Georgia.7 Kallen et al.9 recently reported 51 cases from 19 states between November 2006 and April 2007. To the best of our knowledge, these reports represent the largest case series describing MRSA in CAP.
Earlier studies have suggested a relationship between the outcome of S. aureus infection and the presence of various patient and strain characteristics.1013 Most observations regarding the adverse impact of MRSA on patient outcomes have arisen from analyses of cohorts of patients with either mixed infections, bacteremias, or nosocomial infection. There is little information on outcomes in patients with S. aureus CAP. To address this issue, we conducted a retrospective study of patients with culture‐proven S. aureus pneumonia admitted to a large urban hospital.
Materials and Methods
Data Source
This retrospective study was conducted at Henry Ford Hospital in Detroit, MI, a 903‐bed tertiary care center. (Preliminary findings from the study have been presented at annual meetings of the Infectious Diseases Society of America [IDSA], the American College of Chest Physicians [ACCP], and the Interscience Conference on Antimicrobial Agents and Chemotherapy [ICAAC].) Data were obtained from the Henry Ford CarePlus Electronic Medical Record database, the Henry Ford Infectious Diseases Research Laboratory database, and the cost component of the Corporate Data Store, which is a central repository of data on patient encounters at Henry Ford Hospital and all Henry Ford Health System (HFHS) ambulatory care sites.
Sample Selection
The source population for the study consisted of all admissions to Henry Ford Hospital between January 2005 and May 2008 (study period) Patients were included in the study sample if they had: (1) a discharge diagnosis (principal or secondary) of pneumonia (International Classification of Diseases 9th Edition, Clinical Modification [ICD‐9‐CM] diagnosis codes 481.X 486.X) on their discharge summary or in their medical record; (2) a positive chest x‐ray (ie, infiltrate, consolidation, pleural effusion) within 48 hours of hospital admission; (3) an abnormal temperature (>38.3C [101.0F] or <36C [96.8F]), an abnormal white blood count (WBC) (>12,000/mm3 or <4,000/mm3), or increased sputum production on their day of hospital admission; and (4) a positive blood or respiratory culture for S. aureus within 48 hours of hospital admission.
Among these identified patients, those with probable HCAP were excluded from the study sample, based on evidence of: (1) hospitalization for 2 days during the 90‐day period preceding the index hospitalization; (2) admission to hospital from a nursing home or long‐term care facility; (3) hemodialysis 30 days prior to hospital admission; (4) receipt of cancer chemotherapy, IV antibiotic therapy, or wound care 30 days prior to hospital admission; or (5) receipt of an immunosuppressant at the time of hospital admission. All remaining patients were assumed to have CAP.
Data Extraction
For each admission in the study sample, selected demographic and clinical information was extracted from inpatient and outpatient medical records, beginning 1 year prior to the date of hospital admission and ending 30 days subsequent to the date of hospital discharge or the date of discontinuation of antibiotic therapy, whichever occurred later. All data were extracted by trained medical abstractors, using a set of case‐report forms developed specifically for this study.
Study Measures
Baseline demographic and clinical characteristics of study subjects were examined, including age, sex, race, evidence of positive S. aureus culture during the 1‐year period prior to hospital admission, history of selected disease conditions, and clinical status at admission (eg, comorbidities, vital signs, WBC, platelet count). A Confusion, Urea nitrogen, Respiratory rate, Blood pressure, 65 years of age and older score (CURB‐65) was calculated for each patient, based on clinical information at hospital admission.14 (A CURB‐65 score of 0 indicates a low [<1%] risk of death from pneumonia, while a score of 5 indicates a very high [57%] risk of death.)
Patients were designated as having MRSA or MSSA CAP based on the results of blood or respiratory cultures obtained within 48 hours of hospital admission. MRSA isolates were initially identified using automated dilution testing, in accordance with guidelines of the Clinical and Laboratory Standards Institute.15 The genotypic and phenotypic characteristics of MRSA isolates were examined, including susceptibility to vancomycin and presence of the Panton‐Valentine leukocidin (PVL) gene. Minimum inhibitory concentrations (MICs) for vancomycin were ascertained using the E‐test (AB BIODISK, Solna, Sweden).16 The PVL gene was detected using pulse field gel electrophoresis patterns (PFGE) and polymerase chain reaction. PFGE was performed using the restriction endonuclease SmaI. All S. aureus isolates were entered into a database using Gel Doc 2000 (BioRad) gel documentation system, and PFGE patterns were analyzed using BioNumerics Version 3.5, and grouped into Pulse‐field types using DICE coefficients and 80% relatedness.17
Initial antibiotic therapy was defined to consist of all antibiotics received within the first 48 hours in the hospital, regardless of sequence. Appropriateness of initial antibiotic therapy was ascertained based on susceptibility of the organism to the initial regimen received. Patients with MRSA isolates were designated as having received appropriate initial therapy if they were given vancomycin, linezolid or tigecycline. Those with MSSA isolates were designated as having received appropriate initial therapy if they were given a beta‐lactam, vancomycin, linezolid, or tigecycline. For patients with MRSA or MSSA that received doxycycline, clindamycin, or sulfamethoxazole/trimethoprim, each culture was evaluated individually to determine appropriateness, defined as in vitro susceptibility of the antimicrobial received to the organism. Clinical and economic outcomes of interest included: (1) thoracic surgery for pneumonia any time prior to hospital discharge; (2) receipt of mechanical ventilation any time prior to hospital discharge; (3) admission to an intensive care unit (ICU), irrespective of reason, anytime prior to hospital discharge; (4) length of stay in hospital; (5) total hospital charges for all services provided between hospital admission and hospital discharge; and (6) in‐hospital death (case‐fatality)
Statistical Analyses
We examined the baseline demographic and clinical characteristics of patients in the study sample, on an overall basis and for those with MRSA vs. MSSA isolates respectively. Categorical measures were summarized using frequency distributions and percentages; means, standard deviations (SDs) and medians were employed for continuous measures.
Clinical and economic outcomes were similarly examined, on an overall basis and for patients with MSSA vs. MRSA isolates. For patients with MRSA, we also examined outcomes in relation to selected genotypic and phenotypic characteristics of MRSA isolates, including MIC to vancomycin (1.00 g/mL, 1.50 g/mL, and 2.00 g/mL) and presence of the gene for PVL toxin.
Statistical significance of differences between patients with MRSA vs. MSSA isolates was assessed using t‐tests for continuous measures, and chi‐square tests for categorical measures; statistical significance was similarly assessed for patients with the PVL toxin gene vs. those without it. Because the distribution of vancomycin MICs was highly skewed, statistical significance of differences in outcomes was not assessed in relation to this measure.
All analyses were conducted using SAS Proprietary Software, Release 9.1 (SAS Institute Inc., Cary, NC). Missing and/or incomplete case‐report form data were not imputed, as observations were presumed not to be missing at random.
Results
There were 282 admissions to Henry Ford Hospital between January 2005 and May 2008 of patients with pneumonia who had positive blood or respiratory cultures for S. aureus within 48 hours of admission. (The total number of admissions over this period of patients with a principal diagnosis of pneumonia was 3894). Twelve patients had a negative chest x‐ray or negative clinical findings for pneumonia on the day of hospital admission and were excluded from the analysis. An additional 142 (53%) patients had evidence of HCAP and were excluded from the analysis. The final study sample therefore consisted of 128 patients with S. aureus CAP; their demographic and clinical characteristics are presented in Table 1.
| Characteristic | Overall (n = 128) | MSSA (n = 73) | MRSA (n = 55) | MSSA vs. MRSA P Value |
|---|---|---|---|---|
| ||||
| Age, years | ||||
| 1734 | 10 (7.8) | 6 (8.2) | 4 (7.3) | 1.000 |
| 3549 | 22 (17.2) | 12 (16.4) | 10 (18.2) | 0.817 |
| 5064 | 47 (36.7) | 26 (35.6) | 21 (38.2) | 0.854 |
| 65 | 49 (38.3) | 29 (39.7) | 20 (36.4) | 0.717 |
| Mean (SD) | 59.8 (17.0) | 59.8 (16.6) | 59.8 (17.8) | 0.999 |
| Sex | ||||
| Male | 74 (57.8) | 35 (47.9) | 39 (70.9) | 0.011 |
| Female | 54 (42.2) | 38 (52.1) | 16 (29.1) | |
| Race | ||||
| African American | 86 (67.2) | 46 (63.0) | 40 (72.7) | 0.261 |
| Caucasian | 36 (28.1) | 24 (32.9) | 12 (21.8) | 0.233 |
| Other/unknown | 6 (4.7) | 3 (4.1) | 3 (5.5) | 1.000 |
| Prior positive S. aureus culture* | ||||
| MSSA | 4 (3.1) | 4 (5.5) | 0 (0.0) | 0.134 |
| MRSA | 4 (3.1) | 1 (1.4) | 3 (5.5) | 0.314 |
| Both | 0 (0.0) | 0 (0.0) | 0 (0.0) | |
| Comorbidities | ||||
| Active malignancy | 11 (8.6) | 6 (8.2) | 5 (9.1) | 1.000 |
| Acute renal failure | 60 (46.9) | 31 (42.5) | 29 (52.7) | 0.285 |
| Coronary artery bypass grafting | 7 (5.5) | 5 (6.8) | 2 (3.6) | 0.698 |
| Coronary artery disease | 39 (30.5) | 21 (28.8) | 18 (32.7) | 0.700 |
| Cerebrovascular disease | 23 (18.0) | 15 (20.5) | 8 (14.5) | 0.487 |
| Congestive heart failure | 40 (31.3) | 22 (30.1) | 18 (32.7) | 0.848 |
| Chronic renal failure | 23 (18.0) | 12 (16.4) | 11 (20.0) | 0.647 |
| Chronic obstructive pulmonary disease | 31 (24.2) | 14 (19.2) | 17 (30.9) | 0.147 |
| Diabetes | 38 (29.7) | 19 (26.0) | 19 (34.5) | 0.332 |
| Diabetes mellitus with organ damage | 4 (3.1) | 2 (2.7) | 2 (3.6) | 1.000 |
| End‐stage renal disease with receipt of dialysis | 1 (0.8) | 1 (1.4) | 0 (0.0) | 1.000 |
| HIV/AIDS | 7 (5.5) | 2 (2.7) | 5 (9.1) | 0.138 |
| Myocardial infarction | 27 (21.1) | 15 (20.5) | 12 (21.8) | 1.000 |
| Peripheral vascular disease | 26 (20.3) | 10 (13.7) | 16 (29.1) | 0.045 |
| CURB‐65 | ||||
| 0 | 15 (11.7) | 6 (8.2) | 9 (16.4) | 0.175 |
| 1 | 36 (28.1) | 21 (28.8) | 15 (27.3) | 1.000 |
| 2 | 48 (37.5) | 28 (38.4) | 20 (36.4) | 0.855 |
| 3 | 20 (15.6) | 14 (19.2) | 6 (10.9) | 0.229 |
| 4 | 8 (6.3) | 3 (4.1) | 5 (9.1) | 0.288 |
| 5 | 1 (0.8) | 1 (1.4) | 0 (0.0) | 1.000 |
| Mean(SD) | 1.8 (1.1) | 2 (1.0) | 2 (1.2) | 0.379 |
Mean (SD) age of study subjects was 60 (17.0) years; 72% were nonwhite, and 58% were males. Prevalence of selected comorbidities, including acute renal failure, coronary artery disease, congestive heart failure, chronic obstructive pulmonary disease, diabetes, myocardial infarction, and peripheral vascular disease was high (>20%). Mean (SD) CURB score at admission was 1.8 (1.1); scores did not differ significantly between MRSA vs. MSSA patients. Eight patients (6%) had evidence of positive S. aureus cultures in the year prior to admission.
A total of 55 (43%) patients had positive MRSA cultures within the first 48 hours of admission, while the remainder (n = 73 [57%]) had cultures positive for MSSA. Among the 55 patients with MRSA, 23 (42%) had USA300 MRSA strains that produced PVL toxin. Only 18% of MRSA patients had MICs to vancomycin of 1 g/mL; most remaining patients (73%) had 1.5 g/mL MIC values. Thirty‐one patients (24%) died prior to hospital discharge (Table 2). Eighty‐eight (69%) patients received mechanical ventilation, and 79% were admitted to ICU; 25% of patients underwent thoracic surgery for pneumonia. Mean (SD) length of stay in hospital was 17.0 (15.7) days, and mean (SD) total charges per hospitalization were $127,922 ($154,605). (Mean length of stay in hospital was 5.8 days for all 3894 patients with a principal discharge diagnosis of pneumonia, and mean total charges were $29,448).
| Study Outcomes | Overall (n = 128) | MSSA (n = 73) | MRSA (n = 55) | P Value | PVL Toxin* (n = 23) | No PVL Toxin* (n = 28) | P Value |
|---|---|---|---|---|---|---|---|
| |||||||
| Case fatality | 31 (24.2) | 19 (26.0) | 12 (21.8) | 0.678 | 4 (17.4) | 6 (21.4) | 1.000 |
| Surgery for pneumonia | 32 (25.0) | 19 (26.0) | 13 (23.6) | 0.838 | 7 (30.4) | 5 (17.9) | 0.329 |
| Receipt of mechanical ventilation | 88 (68.8) | 56 (76.7) | 32 (58.2) | 0.034 | 12 (52.2) | 18 (64.3) | 0.576 |
| ICU admission | 101 (78.9) | 60 (82.2) | 41 (74.5) | 0.382 | 15 (65.2) | 22 (78.6) | 0.348 |
| Length of stay, days | |||||||
| Mean (SD) | 17.2 (15.7) | 16.4 (15.0) | 18.2 (16.6) | 0.525 | 25.3 (22.6) | 13.2 (7.7) | 0.020 |
| Median | 13.0 | 13.0 | 13.0 | 16.0 | 12.0 | ||
| Total charges, $ | |||||||
| Mean (SD) | 127,922 (154,605) | 135,784 (170,046) | 117,489 (132,164) | 0.510 | 162,124 (186,923) | 85,215 (57,957) | 0.066 |
| Median | 81,374 | 84,593 | 71,868 | 106,599 | 67,328 | ||
Notwithstanding the fact that patients with MRSA were more than twice as likely to receive inappropriate initial therapy (44% for MRSA vs. 18% for MSSA; odds ratio [OR], 3.57; 95% confidence interval [CI], 1.607.97; P = 0.0015), there were no statistically significant differences in in‐hospital mortality (26% for MSSA vs. 22% for MRSA; P = 0.678), surgery for pneumonia (26% vs. 24%; P = 0.838), or admission to ICU (82% vs. 74%; P = 0.382). Patients with MSSA CAP were more likely to receive mechanical ventilation (77% vs. 58%; P = 0.034). Mean (SD) total charges per admission did not differ between patients with MSSA vs. MRSA CAP ($135,784 [$170,046] vs $117,489 [$132,164], respectively [P = 0.510]). (30‐day mortality following hospital discharge was 24% for MRSA CAP patients and 30% for MSSA CAP patients.)
Among patients with MRSA CAP, there were no notable differences in outcomes between those with strains with the PVL toxin vs. those without it (Table 2). Mean (SD) length of stay in hospital, however, was significantly longer among the former patients (25 [22.6] days vs. 13 [7.7] days for those without PVL toxin; P = 0.020). Reflecting this difference, mean (SD) total charges per admission were $76,909 higher among MRSA patients with PVL‐positive strains vs. those with PVL‐negative strains ($162,124 [$186,923] vs. $85,215 [$57,957] respectively; P = 0.066).
Among MRSA patients with MICs to vancomycin of 1.5 g/mL, 17% died in hospital, 20% underwent surgery for pneumonia, 60% received mechanical ventilation, and 75% were admitted to ICU. Mean (SD) total charges per admission were $104,514 ($112,606) (Table 2).
Discussion
To the best of our knowledge, this is the largest observational study to date of outcomes in patients with S. aureus CAP. Our results indicate that MRSA represents almost one‐half of all such infections, with USA300 strains accounting for a substantial proportion of these cases. In addition, for most outcome measures, there were no significant differences between patients with MSSA vs. MRSA CAP. In patients with MRSA CAP, however, those with PVL‐positive strains had longer stays in the hospital and had higher total costs of hospitalization.
Our study highlights the significant clinical and economic burden of S. aureus CAP. In the US, the mortality rate in patients hospitalized with CAP is about 8%, while mean length of stay is approximately 5 days (it was 5.8 days in our institution among all patients with CAP). In contrast, we found that most patients with S. aureus CAP were admitted to ICU, and that nearly one in four died during hospitalization. One possible explanation for these findings is that patients with S. aureus CAP may often receive inappropriate initial antibiotic therapy, a major predictor of adverse outcome in infection. The overall rate of inappropriate treatment, however, was low in our study (although we note that optimal antibiotic treatment is not clearly defined for the USA300 strain, and as a result, we assumed that vancomycin would be appropriate for all MRSA infections). A more likely explanation for our findings is the severity of CAP associated with S. aureus, whether MRSA or MSSA. Given the nature of S. aureus infections, the need for prevention (and potentially, development of a vaccine) remains crucial.
While MRSA has been studied extensively as a cause of bacteremia, as well as HCAP and VAP, it has not been well studied in CAP. Patients with MRSA bacteremia have been shown to have a higher mortality risk and higher healthcare costs than those with MSSA infections, and a meta‐analysis of 31 studies of patients with S. aureus bloodstream infections demonstrated a significant increase in mortality among patients with MRSA vs. MSSA bacteremia.1820 Outcomes when comparing MRSA and MSSA in VAP are slightly more variable. A higher rate of mortality has been reported in patients with pneumonia caused by MRSA vs. MSSA, but others found no difference after controlling for potential confounders.21, 22 We found that case fatality, surgery for pneumonia, ICU admission, length of stay, and total hospital charges do not differ significantly between patients with MRSA vs. MSSA CAP. This suggests that appreciation of the burden of MRSA can only be done in the context of the syndrome in question, and that conclusions from analyses of bacteremia and VAP may not be generalizable to CAP.
In the US, an increasing number of community‐associated infections are due to MRSA. Skin and skin structure infections comprise the majority of community‐associated MRSA infections and are caused by a single pulsed field type, termed USA300. These strains are believed to have distinctive virulence and epidemiologic characteristics. USA300 isolates typically are resistant only to beta‐lactam and macrolide antimicrobial agents and contain genes for the PVL toxin, which typically are not present in strains of healthcare‐associated MRSA.23 Recent studies of acute pneumonia with animal models and in humans have suggested that the PVL toxinalone or in combination with other virulence factorsis associated with the development of necrotizing pneumonia.2430 In our study, 23 patients with MRSA CAP had strains that contained PVL toxin, and these patients had longer stays in hospital and higher total hospital charges than those with MRSA CAP not containing the PVL toxin. This is an important finding, as length of stay is an important proxy for morbidity and case severity. This also extends the finding of previous case studies reporting that MRSA CAP with PVL toxin is associated with worse outcomes.2430 Moreover, we clearly document that PVL‐positive strains are emerging as a cause of pulmonary infection in broader clinical scenarios. As such, physicians must remain vigilant for this toxin‐producing strain. The full extent of the impact of PVL‐positive strains in our study is unknown, as we cannot ascertain whether morbidity was worse because of the PVL toxin itself, or because most patients were not treated with clindamycin or linezolid, which inhibit toxin production.
We failed to observe a correlation between MICs to vancomycin and the outcomes we studied. The clinical significance of reduced susceptibility of S. aureus to vancomycin remains a controversial issue. In our study, there was no significant association between MICs to vancomycin and mortality, need for surgery, ICU admission, length of stay, or total hospital charges. Sakoulas et al.11 reported that as vancomycin MICs for MRSA isolates rose within the susceptible range from 0.5 mcg/mL to 2.0 mcg/mL, so too did the number of clinical failures among bacteremic patients receiving vancomycin. Moise‐Broder et al.31 reported a similar finding in a study of 63 patients with MRSA bacteremia, of whom 45 failed or were intolerant of vancomycin therapy. In a more recent report, Soriano et al.12 prospectively evaluated 414 episodes of bacteremia. The authors concluded that mortality in patients with MRSA bacteremia is significantly higher when the empiric antimicrobial agent is inappropriate and when vancomycin is used to treat infections involving strains with MICs >1.0 mcg/mL. In pneumonia, Hidayat et al.13 reported that patients with infections due to pathogens with higher MICs to vancomycin had a higher rate of mortality than those with lower MICs. Our findings likely differ from this report because of the proportion of subjects with PVL‐positive strains, and because of the skew in the distribution of vancomycin MICs. More specifically, the majority of patients had higher MIC strains, clustered around 1.5 mg/L, leaving us with few lower MIC strains and fewer strains with MICs of 2.0 mcg/mL for comparison, and therefore limited statistical power to assess the relationship between MICs and outcomes.
There were no clinical features that clearly distinguished patients with MRSA vs. MSSA CAP. This suggests that if physicians hope to ensure that patients with MRSA CAP receive appropriate initial antibiotic therapy, they cannot base therapeutic decision‐making (ie, use of an anti‐MRSA treatment) on clinical criteria. Presently, national guidelines recommend diagnostic testing only if the results might affect clinical decisions (eg, antimicrobial management).32 Furthermore, the IDSA/ATS guidelines recommend sputum cultures along with blood cultures and other diagnostic tests only in select cases (eg, those with severe disease). Since beginning optimal therapy quickly can reduce mortality in pneumonia,33 our results indicate a need for both rapid diagnostic tests to identify patients with MRSA, and reevaluation of current recommendations for diagnostic testing in CAP.
Our study has several important limitations. First, its retrospective design exposes it to many forms of bias. Second, because the diagnosis of pneumonia can be challenging, we required that all patients have both clinical and radiologic evidence of the disease. However, we may have thereby excluded some admissions for S. aureus pneumonia among the over 3000 pneumonia admissions to our hospital during the study period.
Third, we only included patients with culture evidence of infection. This was necessary by design, but as a result we may have failed to identify some patients with S. aureus infections because of the limitations of respiratory culture technology. Patients on general practice units often do not get cultured, and accordingly we may have missed those with milder disease caused by S. aureus.
Fourth, we used only one method of MIC testing to determine vancomycin susceptibility. It would have been of interest to have used automated dilution testing also and to have compared both methods. Comparison of laboratory methods was not among our study objectives, and most of the earlier studies examining epidemiology and outcomes used a single laboratory testing method only.
Finally, since the study was conducted at a large urban teaching hospital in a city with a long history of issues with MRSA and resistance, the generalizability of our results may be limited.
In summary, our study provides further evidence that S. aureus is an important pathogen in CAP. MRSA CAP has few obvious characteristics that differentiate it from MSSA CAP. MRSA and MSSA CAP are both very serious infections that should be treated aggressively to avoid poor outcomes.
- .Staphylococcus aureus: a well‐armed pathogen.Clin Infect Dis.1998;26:1179–1181.
- .Staphylococcus aureus infections.N Engl J Med.1998;339:520–532.
- .The growing menace of community‐acquired methicillin‐resistant Staphylococcus aureus.Ann Intern Med.2006;144:368–370.
- ,,, et al.Predictors of mortality for methicillin‐resistant Staphylococcus aureus health‐care‐associated pneumonia: specific evaluation of vancomycin pharmacokinetic indices.Chest.2006;130:947–955.
- ,,,.Bacterial pneumonia during the Hong Kong influenza epidemic of 1968–1969.Arch Intern Med.1971;127:1037–1041.
- ,,, et al.Severe community‐acquired pneumonia due to Staphylococcus aureus, 2003–04 influenza season.Emerg Infect Dis.2006;12:894–899.
- Centers for Disease Control and Prevention.Severe methicillin‐resistant Staphylococcus aureus community‐acquired pneumonia associated with influenza—Louisiana and Georgia, December 2006‐January 2007.Morb Mortal Wkly Rep.2007;56:325–329.
- ,,.Incidence, characteristics and outcomes of patients with severe community acquired‐MRSA pneumonia.Eur Respir J.2009;34:1148–1158.
- ,,, et al.Staphylococcus aureus community‐acquired pneumonia during the 2006 to 2007 influenza season.Ann Emerg Med.2009;53:358–365.
- ,,, et al.Treatment outcomes for serious infections caused by methicillin‐resistant Staphylococcus aureus with reduced vancomycin susceptibility.Clin Infect Dis.2004;38:521–528.
- ,,, et al.Relationship of MIC and bactericidal activity to efficacy of vancomycin for treatment of methicillin‐resistant Staphylococcus aureus bacteremia.J Clin Microbiol.2004;42:2398–2402.
- ,,, et al.Influence of vancomycin minimum inhibitory concentration on the treatment of Methicillin‐resistant Staphylococcus aureus bacteremia.Clin Infect Dis.2008;46:193–200.
- ,,, et al.High‐dose vancomycin therapy for methicillin‐resistant Staphylococcus aureus infections.Arch Intern Med.2006;166:2138–2144.
- ,,, et al.Defining community acquired pneumonia severity on presentation to hospital: An international derivation and validation study.Thorax.2003;58(5):377–382.
- Clinical and Laboratory Standards Institute (CLSI).Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically: Approved Standard. 8th ed. CLSI document M07‐A8 2008;29(2):1–65. Available at: www.clsi.org/source/orders/free/m07‐a8.pdf.
- ,,,.A multicenter study evaluating the current strategies for isolating Staphylococcus aureus strains with reduced susceptibility to glycopeptides.J Clin Microbiol.2007;45:329–332.
- ,,,,.Application of molecular techniques to the study of hospital infection.Clin Microbiol Rev.2006;19:512–530.
- ,,.Nosocomial methicillin‐resistant Staphylococcus aureus bacteremia: Is it any worse than nosocomial methicillin‐sensitive Staphylococcus aureus bacteremia?Infect Control Hosp Epidemiol.2000;21:645–648.
- ,.Clinical and economic impact of methicillin resistance in patients with Staphylococcus aureus bacteremia.Diagn Microbiol Infect Dis.2005;52:113–122.
- ,,, et al.Comparison of mortality associated with methicillin‐resistant and methicillin‐susceptible Staphylococcus aureus bacteremia: A meta‐analysis.Clin Infect Dis.2003;36:53–59.
- ,,, et al.Ventilator‐associated pneumonia by Staphylococcus aureus: Comparison of methicillin‐resistant and methicillin‐sensitive episodes.Am J Respir Crit Care Med.1994;150:1545–1549.
- ,,, et al.Is methicillin resistance associated with a worse prognosis in Staphylococcus aureus ventilator‐associated pneumonia?Clin Infect Dis.2005;41:1224–1231.
- ,,, et al.Characterization of a strain of community‐associated methicillin‐resistant Staphylococcus aureus widely disseminated in the United States.J Clin Microbiol.2006;44:108–111.
- .Staphylococcus aureus, Panton‐Valentine leukocidin, and necrotising pneumonia.BMJ.2005;331:793–794.
- ,,, et al.Methicillin‐resistant Staphylococcus aureus necrotizing pneumonia.Emerg Infect Dis.2005;11:1647–1648.
- ,.Fatal necrotizing pneumonia due to community‐acquired methicillin‐resistant Staphylococcus aureus (MRSA).Med J Aust.2004;181:228–229.
- ,,.Pleuropulmonary complications of Panton‐Valentine leukocidin‐positive community‐acquired methicillin‐resistant Staphylococcus aureus: Importance of treatment with antimicrobials inhibiting exotoxin production.Chest.2005;128:2732–2738.
- ,,, et al.Severe community‐onset pneumonia in healthy adults caused by methicillin‐resistant Staphylococcus aureus carrying the Panton‐Valentine leukocidin genes.Clin Infect Dis.2005;40:100–107.
- ,,, et al.Staphylococcus aureus Panton‐Valentine leukocidin causes necrotizing pneumonia.Science.2007;315:1130–1133.
- ,,, et al.Association between Staphylococcus aureus strains carrying gene for Panton‐Valentine leukocidin and highly lethal necrotizing pneumonia in young immunocompetent patients.Lancet.2002;359:753–759.
- ,,, et al.Accessory gene regulator group II polymorphism in methicillin‐resistant Staphylococcus aureus is predictive of failure of vancomycin therapy.Clin Infect Dis.2004;38:1700–1705.
- ,,, et al.Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community‐acquired pneumonia in adults.Clin Infect Dis.2007;44(Suppl 2):S27–S72.
- ,,, et al.Antimicrobial therapy escalation and hospital mortality among patients with health‐care‐associated pneumonia: a single center experience.Chest.2008;134:963–968.
Staphylococcus aureus (S. aureus) is well recognized as a major cause of nosocomial and healthcare‐associated pneumonia (HCAP). These infections are associated with substantial morbidity and mortality, and generate high medical costs.14 The significance of S. aureus in community‐acquired pneumonia (CAP) is less clear. Historically, S. aureus has not been considered a common pathogen in CAP, usually arising in association with or following influenza or an influenza‐like syndrome.58 However, with the increasing prevalence of methicillin‐resistant S. aureus (MRSA) as a cause of HCAPand most recently, serious community‐acquired non‐pulmonary infectionsinterest in this pathogen and its impact beyond the hospital have been expanding. During the 2003 to 2004 influenza season, 15 cases of influenza‐associated MRSA CAP were identified.6 In January 2007, the US Centers for Disease Control and Prevention (CDC) received reports of an additional 10 cases of severe MRSA CAP, resulting in six deaths, among previously healthy children and adults in Louisiana and Georgia.7 Kallen et al.9 recently reported 51 cases from 19 states between November 2006 and April 2007. To the best of our knowledge, these reports represent the largest case series describing MRSA in CAP.
Earlier studies have suggested a relationship between the outcome of S. aureus infection and the presence of various patient and strain characteristics.1013 Most observations regarding the adverse impact of MRSA on patient outcomes have arisen from analyses of cohorts of patients with either mixed infections, bacteremias, or nosocomial infection. There is little information on outcomes in patients with S. aureus CAP. To address this issue, we conducted a retrospective study of patients with culture‐proven S. aureus pneumonia admitted to a large urban hospital.
Materials and Methods
Data Source
This retrospective study was conducted at Henry Ford Hospital in Detroit, MI, a 903‐bed tertiary care center. (Preliminary findings from the study have been presented at annual meetings of the Infectious Diseases Society of America [IDSA], the American College of Chest Physicians [ACCP], and the Interscience Conference on Antimicrobial Agents and Chemotherapy [ICAAC].) Data were obtained from the Henry Ford CarePlus Electronic Medical Record database, the Henry Ford Infectious Diseases Research Laboratory database, and the cost component of the Corporate Data Store, which is a central repository of data on patient encounters at Henry Ford Hospital and all Henry Ford Health System (HFHS) ambulatory care sites.
Sample Selection
The source population for the study consisted of all admissions to Henry Ford Hospital between January 2005 and May 2008 (study period) Patients were included in the study sample if they had: (1) a discharge diagnosis (principal or secondary) of pneumonia (International Classification of Diseases 9th Edition, Clinical Modification [ICD‐9‐CM] diagnosis codes 481.X 486.X) on their discharge summary or in their medical record; (2) a positive chest x‐ray (ie, infiltrate, consolidation, pleural effusion) within 48 hours of hospital admission; (3) an abnormal temperature (>38.3C [101.0F] or <36C [96.8F]), an abnormal white blood count (WBC) (>12,000/mm3 or <4,000/mm3), or increased sputum production on their day of hospital admission; and (4) a positive blood or respiratory culture for S. aureus within 48 hours of hospital admission.
Among these identified patients, those with probable HCAP were excluded from the study sample, based on evidence of: (1) hospitalization for 2 days during the 90‐day period preceding the index hospitalization; (2) admission to hospital from a nursing home or long‐term care facility; (3) hemodialysis 30 days prior to hospital admission; (4) receipt of cancer chemotherapy, IV antibiotic therapy, or wound care 30 days prior to hospital admission; or (5) receipt of an immunosuppressant at the time of hospital admission. All remaining patients were assumed to have CAP.
Data Extraction
For each admission in the study sample, selected demographic and clinical information was extracted from inpatient and outpatient medical records, beginning 1 year prior to the date of hospital admission and ending 30 days subsequent to the date of hospital discharge or the date of discontinuation of antibiotic therapy, whichever occurred later. All data were extracted by trained medical abstractors, using a set of case‐report forms developed specifically for this study.
Study Measures
Baseline demographic and clinical characteristics of study subjects were examined, including age, sex, race, evidence of positive S. aureus culture during the 1‐year period prior to hospital admission, history of selected disease conditions, and clinical status at admission (eg, comorbidities, vital signs, WBC, platelet count). A Confusion, Urea nitrogen, Respiratory rate, Blood pressure, 65 years of age and older score (CURB‐65) was calculated for each patient, based on clinical information at hospital admission.14 (A CURB‐65 score of 0 indicates a low [<1%] risk of death from pneumonia, while a score of 5 indicates a very high [57%] risk of death.)
Patients were designated as having MRSA or MSSA CAP based on the results of blood or respiratory cultures obtained within 48 hours of hospital admission. MRSA isolates were initially identified using automated dilution testing, in accordance with guidelines of the Clinical and Laboratory Standards Institute.15 The genotypic and phenotypic characteristics of MRSA isolates were examined, including susceptibility to vancomycin and presence of the Panton‐Valentine leukocidin (PVL) gene. Minimum inhibitory concentrations (MICs) for vancomycin were ascertained using the E‐test (AB BIODISK, Solna, Sweden).16 The PVL gene was detected using pulse field gel electrophoresis patterns (PFGE) and polymerase chain reaction. PFGE was performed using the restriction endonuclease SmaI. All S. aureus isolates were entered into a database using Gel Doc 2000 (BioRad) gel documentation system, and PFGE patterns were analyzed using BioNumerics Version 3.5, and grouped into Pulse‐field types using DICE coefficients and 80% relatedness.17
Initial antibiotic therapy was defined to consist of all antibiotics received within the first 48 hours in the hospital, regardless of sequence. Appropriateness of initial antibiotic therapy was ascertained based on susceptibility of the organism to the initial regimen received. Patients with MRSA isolates were designated as having received appropriate initial therapy if they were given vancomycin, linezolid or tigecycline. Those with MSSA isolates were designated as having received appropriate initial therapy if they were given a beta‐lactam, vancomycin, linezolid, or tigecycline. For patients with MRSA or MSSA that received doxycycline, clindamycin, or sulfamethoxazole/trimethoprim, each culture was evaluated individually to determine appropriateness, defined as in vitro susceptibility of the antimicrobial received to the organism. Clinical and economic outcomes of interest included: (1) thoracic surgery for pneumonia any time prior to hospital discharge; (2) receipt of mechanical ventilation any time prior to hospital discharge; (3) admission to an intensive care unit (ICU), irrespective of reason, anytime prior to hospital discharge; (4) length of stay in hospital; (5) total hospital charges for all services provided between hospital admission and hospital discharge; and (6) in‐hospital death (case‐fatality)
Statistical Analyses
We examined the baseline demographic and clinical characteristics of patients in the study sample, on an overall basis and for those with MRSA vs. MSSA isolates respectively. Categorical measures were summarized using frequency distributions and percentages; means, standard deviations (SDs) and medians were employed for continuous measures.
Clinical and economic outcomes were similarly examined, on an overall basis and for patients with MSSA vs. MRSA isolates. For patients with MRSA, we also examined outcomes in relation to selected genotypic and phenotypic characteristics of MRSA isolates, including MIC to vancomycin (1.00 g/mL, 1.50 g/mL, and 2.00 g/mL) and presence of the gene for PVL toxin.
Statistical significance of differences between patients with MRSA vs. MSSA isolates was assessed using t‐tests for continuous measures, and chi‐square tests for categorical measures; statistical significance was similarly assessed for patients with the PVL toxin gene vs. those without it. Because the distribution of vancomycin MICs was highly skewed, statistical significance of differences in outcomes was not assessed in relation to this measure.
All analyses were conducted using SAS Proprietary Software, Release 9.1 (SAS Institute Inc., Cary, NC). Missing and/or incomplete case‐report form data were not imputed, as observations were presumed not to be missing at random.
Results
There were 282 admissions to Henry Ford Hospital between January 2005 and May 2008 of patients with pneumonia who had positive blood or respiratory cultures for S. aureus within 48 hours of admission. (The total number of admissions over this period of patients with a principal diagnosis of pneumonia was 3894). Twelve patients had a negative chest x‐ray or negative clinical findings for pneumonia on the day of hospital admission and were excluded from the analysis. An additional 142 (53%) patients had evidence of HCAP and were excluded from the analysis. The final study sample therefore consisted of 128 patients with S. aureus CAP; their demographic and clinical characteristics are presented in Table 1.
| Characteristic | Overall (n = 128) | MSSA (n = 73) | MRSA (n = 55) | MSSA vs. MRSA P Value |
|---|---|---|---|---|
| ||||
| Age, years | ||||
| 1734 | 10 (7.8) | 6 (8.2) | 4 (7.3) | 1.000 |
| 3549 | 22 (17.2) | 12 (16.4) | 10 (18.2) | 0.817 |
| 5064 | 47 (36.7) | 26 (35.6) | 21 (38.2) | 0.854 |
| 65 | 49 (38.3) | 29 (39.7) | 20 (36.4) | 0.717 |
| Mean (SD) | 59.8 (17.0) | 59.8 (16.6) | 59.8 (17.8) | 0.999 |
| Sex | ||||
| Male | 74 (57.8) | 35 (47.9) | 39 (70.9) | 0.011 |
| Female | 54 (42.2) | 38 (52.1) | 16 (29.1) | |
| Race | ||||
| African American | 86 (67.2) | 46 (63.0) | 40 (72.7) | 0.261 |
| Caucasian | 36 (28.1) | 24 (32.9) | 12 (21.8) | 0.233 |
| Other/unknown | 6 (4.7) | 3 (4.1) | 3 (5.5) | 1.000 |
| Prior positive S. aureus culture* | ||||
| MSSA | 4 (3.1) | 4 (5.5) | 0 (0.0) | 0.134 |
| MRSA | 4 (3.1) | 1 (1.4) | 3 (5.5) | 0.314 |
| Both | 0 (0.0) | 0 (0.0) | 0 (0.0) | |
| Comorbidities | ||||
| Active malignancy | 11 (8.6) | 6 (8.2) | 5 (9.1) | 1.000 |
| Acute renal failure | 60 (46.9) | 31 (42.5) | 29 (52.7) | 0.285 |
| Coronary artery bypass grafting | 7 (5.5) | 5 (6.8) | 2 (3.6) | 0.698 |
| Coronary artery disease | 39 (30.5) | 21 (28.8) | 18 (32.7) | 0.700 |
| Cerebrovascular disease | 23 (18.0) | 15 (20.5) | 8 (14.5) | 0.487 |
| Congestive heart failure | 40 (31.3) | 22 (30.1) | 18 (32.7) | 0.848 |
| Chronic renal failure | 23 (18.0) | 12 (16.4) | 11 (20.0) | 0.647 |
| Chronic obstructive pulmonary disease | 31 (24.2) | 14 (19.2) | 17 (30.9) | 0.147 |
| Diabetes | 38 (29.7) | 19 (26.0) | 19 (34.5) | 0.332 |
| Diabetes mellitus with organ damage | 4 (3.1) | 2 (2.7) | 2 (3.6) | 1.000 |
| End‐stage renal disease with receipt of dialysis | 1 (0.8) | 1 (1.4) | 0 (0.0) | 1.000 |
| HIV/AIDS | 7 (5.5) | 2 (2.7) | 5 (9.1) | 0.138 |
| Myocardial infarction | 27 (21.1) | 15 (20.5) | 12 (21.8) | 1.000 |
| Peripheral vascular disease | 26 (20.3) | 10 (13.7) | 16 (29.1) | 0.045 |
| CURB‐65 | ||||
| 0 | 15 (11.7) | 6 (8.2) | 9 (16.4) | 0.175 |
| 1 | 36 (28.1) | 21 (28.8) | 15 (27.3) | 1.000 |
| 2 | 48 (37.5) | 28 (38.4) | 20 (36.4) | 0.855 |
| 3 | 20 (15.6) | 14 (19.2) | 6 (10.9) | 0.229 |
| 4 | 8 (6.3) | 3 (4.1) | 5 (9.1) | 0.288 |
| 5 | 1 (0.8) | 1 (1.4) | 0 (0.0) | 1.000 |
| Mean(SD) | 1.8 (1.1) | 2 (1.0) | 2 (1.2) | 0.379 |
Mean (SD) age of study subjects was 60 (17.0) years; 72% were nonwhite, and 58% were males. Prevalence of selected comorbidities, including acute renal failure, coronary artery disease, congestive heart failure, chronic obstructive pulmonary disease, diabetes, myocardial infarction, and peripheral vascular disease was high (>20%). Mean (SD) CURB score at admission was 1.8 (1.1); scores did not differ significantly between MRSA vs. MSSA patients. Eight patients (6%) had evidence of positive S. aureus cultures in the year prior to admission.
A total of 55 (43%) patients had positive MRSA cultures within the first 48 hours of admission, while the remainder (n = 73 [57%]) had cultures positive for MSSA. Among the 55 patients with MRSA, 23 (42%) had USA300 MRSA strains that produced PVL toxin. Only 18% of MRSA patients had MICs to vancomycin of 1 g/mL; most remaining patients (73%) had 1.5 g/mL MIC values. Thirty‐one patients (24%) died prior to hospital discharge (Table 2). Eighty‐eight (69%) patients received mechanical ventilation, and 79% were admitted to ICU; 25% of patients underwent thoracic surgery for pneumonia. Mean (SD) length of stay in hospital was 17.0 (15.7) days, and mean (SD) total charges per hospitalization were $127,922 ($154,605). (Mean length of stay in hospital was 5.8 days for all 3894 patients with a principal discharge diagnosis of pneumonia, and mean total charges were $29,448).
| Study Outcomes | Overall (n = 128) | MSSA (n = 73) | MRSA (n = 55) | P Value | PVL Toxin* (n = 23) | No PVL Toxin* (n = 28) | P Value |
|---|---|---|---|---|---|---|---|
| |||||||
| Case fatality | 31 (24.2) | 19 (26.0) | 12 (21.8) | 0.678 | 4 (17.4) | 6 (21.4) | 1.000 |
| Surgery for pneumonia | 32 (25.0) | 19 (26.0) | 13 (23.6) | 0.838 | 7 (30.4) | 5 (17.9) | 0.329 |
| Receipt of mechanical ventilation | 88 (68.8) | 56 (76.7) | 32 (58.2) | 0.034 | 12 (52.2) | 18 (64.3) | 0.576 |
| ICU admission | 101 (78.9) | 60 (82.2) | 41 (74.5) | 0.382 | 15 (65.2) | 22 (78.6) | 0.348 |
| Length of stay, days | |||||||
| Mean (SD) | 17.2 (15.7) | 16.4 (15.0) | 18.2 (16.6) | 0.525 | 25.3 (22.6) | 13.2 (7.7) | 0.020 |
| Median | 13.0 | 13.0 | 13.0 | 16.0 | 12.0 | ||
| Total charges, $ | |||||||
| Mean (SD) | 127,922 (154,605) | 135,784 (170,046) | 117,489 (132,164) | 0.510 | 162,124 (186,923) | 85,215 (57,957) | 0.066 |
| Median | 81,374 | 84,593 | 71,868 | 106,599 | 67,328 | ||
Notwithstanding the fact that patients with MRSA were more than twice as likely to receive inappropriate initial therapy (44% for MRSA vs. 18% for MSSA; odds ratio [OR], 3.57; 95% confidence interval [CI], 1.607.97; P = 0.0015), there were no statistically significant differences in in‐hospital mortality (26% for MSSA vs. 22% for MRSA; P = 0.678), surgery for pneumonia (26% vs. 24%; P = 0.838), or admission to ICU (82% vs. 74%; P = 0.382). Patients with MSSA CAP were more likely to receive mechanical ventilation (77% vs. 58%; P = 0.034). Mean (SD) total charges per admission did not differ between patients with MSSA vs. MRSA CAP ($135,784 [$170,046] vs $117,489 [$132,164], respectively [P = 0.510]). (30‐day mortality following hospital discharge was 24% for MRSA CAP patients and 30% for MSSA CAP patients.)
Among patients with MRSA CAP, there were no notable differences in outcomes between those with strains with the PVL toxin vs. those without it (Table 2). Mean (SD) length of stay in hospital, however, was significantly longer among the former patients (25 [22.6] days vs. 13 [7.7] days for those without PVL toxin; P = 0.020). Reflecting this difference, mean (SD) total charges per admission were $76,909 higher among MRSA patients with PVL‐positive strains vs. those with PVL‐negative strains ($162,124 [$186,923] vs. $85,215 [$57,957] respectively; P = 0.066).
Among MRSA patients with MICs to vancomycin of 1.5 g/mL, 17% died in hospital, 20% underwent surgery for pneumonia, 60% received mechanical ventilation, and 75% were admitted to ICU. Mean (SD) total charges per admission were $104,514 ($112,606) (Table 2).
Discussion
To the best of our knowledge, this is the largest observational study to date of outcomes in patients with S. aureus CAP. Our results indicate that MRSA represents almost one‐half of all such infections, with USA300 strains accounting for a substantial proportion of these cases. In addition, for most outcome measures, there were no significant differences between patients with MSSA vs. MRSA CAP. In patients with MRSA CAP, however, those with PVL‐positive strains had longer stays in the hospital and had higher total costs of hospitalization.
Our study highlights the significant clinical and economic burden of S. aureus CAP. In the US, the mortality rate in patients hospitalized with CAP is about 8%, while mean length of stay is approximately 5 days (it was 5.8 days in our institution among all patients with CAP). In contrast, we found that most patients with S. aureus CAP were admitted to ICU, and that nearly one in four died during hospitalization. One possible explanation for these findings is that patients with S. aureus CAP may often receive inappropriate initial antibiotic therapy, a major predictor of adverse outcome in infection. The overall rate of inappropriate treatment, however, was low in our study (although we note that optimal antibiotic treatment is not clearly defined for the USA300 strain, and as a result, we assumed that vancomycin would be appropriate for all MRSA infections). A more likely explanation for our findings is the severity of CAP associated with S. aureus, whether MRSA or MSSA. Given the nature of S. aureus infections, the need for prevention (and potentially, development of a vaccine) remains crucial.
While MRSA has been studied extensively as a cause of bacteremia, as well as HCAP and VAP, it has not been well studied in CAP. Patients with MRSA bacteremia have been shown to have a higher mortality risk and higher healthcare costs than those with MSSA infections, and a meta‐analysis of 31 studies of patients with S. aureus bloodstream infections demonstrated a significant increase in mortality among patients with MRSA vs. MSSA bacteremia.1820 Outcomes when comparing MRSA and MSSA in VAP are slightly more variable. A higher rate of mortality has been reported in patients with pneumonia caused by MRSA vs. MSSA, but others found no difference after controlling for potential confounders.21, 22 We found that case fatality, surgery for pneumonia, ICU admission, length of stay, and total hospital charges do not differ significantly between patients with MRSA vs. MSSA CAP. This suggests that appreciation of the burden of MRSA can only be done in the context of the syndrome in question, and that conclusions from analyses of bacteremia and VAP may not be generalizable to CAP.
In the US, an increasing number of community‐associated infections are due to MRSA. Skin and skin structure infections comprise the majority of community‐associated MRSA infections and are caused by a single pulsed field type, termed USA300. These strains are believed to have distinctive virulence and epidemiologic characteristics. USA300 isolates typically are resistant only to beta‐lactam and macrolide antimicrobial agents and contain genes for the PVL toxin, which typically are not present in strains of healthcare‐associated MRSA.23 Recent studies of acute pneumonia with animal models and in humans have suggested that the PVL toxinalone or in combination with other virulence factorsis associated with the development of necrotizing pneumonia.2430 In our study, 23 patients with MRSA CAP had strains that contained PVL toxin, and these patients had longer stays in hospital and higher total hospital charges than those with MRSA CAP not containing the PVL toxin. This is an important finding, as length of stay is an important proxy for morbidity and case severity. This also extends the finding of previous case studies reporting that MRSA CAP with PVL toxin is associated with worse outcomes.2430 Moreover, we clearly document that PVL‐positive strains are emerging as a cause of pulmonary infection in broader clinical scenarios. As such, physicians must remain vigilant for this toxin‐producing strain. The full extent of the impact of PVL‐positive strains in our study is unknown, as we cannot ascertain whether morbidity was worse because of the PVL toxin itself, or because most patients were not treated with clindamycin or linezolid, which inhibit toxin production.
We failed to observe a correlation between MICs to vancomycin and the outcomes we studied. The clinical significance of reduced susceptibility of S. aureus to vancomycin remains a controversial issue. In our study, there was no significant association between MICs to vancomycin and mortality, need for surgery, ICU admission, length of stay, or total hospital charges. Sakoulas et al.11 reported that as vancomycin MICs for MRSA isolates rose within the susceptible range from 0.5 mcg/mL to 2.0 mcg/mL, so too did the number of clinical failures among bacteremic patients receiving vancomycin. Moise‐Broder et al.31 reported a similar finding in a study of 63 patients with MRSA bacteremia, of whom 45 failed or were intolerant of vancomycin therapy. In a more recent report, Soriano et al.12 prospectively evaluated 414 episodes of bacteremia. The authors concluded that mortality in patients with MRSA bacteremia is significantly higher when the empiric antimicrobial agent is inappropriate and when vancomycin is used to treat infections involving strains with MICs >1.0 mcg/mL. In pneumonia, Hidayat et al.13 reported that patients with infections due to pathogens with higher MICs to vancomycin had a higher rate of mortality than those with lower MICs. Our findings likely differ from this report because of the proportion of subjects with PVL‐positive strains, and because of the skew in the distribution of vancomycin MICs. More specifically, the majority of patients had higher MIC strains, clustered around 1.5 mg/L, leaving us with few lower MIC strains and fewer strains with MICs of 2.0 mcg/mL for comparison, and therefore limited statistical power to assess the relationship between MICs and outcomes.
There were no clinical features that clearly distinguished patients with MRSA vs. MSSA CAP. This suggests that if physicians hope to ensure that patients with MRSA CAP receive appropriate initial antibiotic therapy, they cannot base therapeutic decision‐making (ie, use of an anti‐MRSA treatment) on clinical criteria. Presently, national guidelines recommend diagnostic testing only if the results might affect clinical decisions (eg, antimicrobial management).32 Furthermore, the IDSA/ATS guidelines recommend sputum cultures along with blood cultures and other diagnostic tests only in select cases (eg, those with severe disease). Since beginning optimal therapy quickly can reduce mortality in pneumonia,33 our results indicate a need for both rapid diagnostic tests to identify patients with MRSA, and reevaluation of current recommendations for diagnostic testing in CAP.
Our study has several important limitations. First, its retrospective design exposes it to many forms of bias. Second, because the diagnosis of pneumonia can be challenging, we required that all patients have both clinical and radiologic evidence of the disease. However, we may have thereby excluded some admissions for S. aureus pneumonia among the over 3000 pneumonia admissions to our hospital during the study period.
Third, we only included patients with culture evidence of infection. This was necessary by design, but as a result we may have failed to identify some patients with S. aureus infections because of the limitations of respiratory culture technology. Patients on general practice units often do not get cultured, and accordingly we may have missed those with milder disease caused by S. aureus.
Fourth, we used only one method of MIC testing to determine vancomycin susceptibility. It would have been of interest to have used automated dilution testing also and to have compared both methods. Comparison of laboratory methods was not among our study objectives, and most of the earlier studies examining epidemiology and outcomes used a single laboratory testing method only.
Finally, since the study was conducted at a large urban teaching hospital in a city with a long history of issues with MRSA and resistance, the generalizability of our results may be limited.
In summary, our study provides further evidence that S. aureus is an important pathogen in CAP. MRSA CAP has few obvious characteristics that differentiate it from MSSA CAP. MRSA and MSSA CAP are both very serious infections that should be treated aggressively to avoid poor outcomes.
Staphylococcus aureus (S. aureus) is well recognized as a major cause of nosocomial and healthcare‐associated pneumonia (HCAP). These infections are associated with substantial morbidity and mortality, and generate high medical costs.14 The significance of S. aureus in community‐acquired pneumonia (CAP) is less clear. Historically, S. aureus has not been considered a common pathogen in CAP, usually arising in association with or following influenza or an influenza‐like syndrome.58 However, with the increasing prevalence of methicillin‐resistant S. aureus (MRSA) as a cause of HCAPand most recently, serious community‐acquired non‐pulmonary infectionsinterest in this pathogen and its impact beyond the hospital have been expanding. During the 2003 to 2004 influenza season, 15 cases of influenza‐associated MRSA CAP were identified.6 In January 2007, the US Centers for Disease Control and Prevention (CDC) received reports of an additional 10 cases of severe MRSA CAP, resulting in six deaths, among previously healthy children and adults in Louisiana and Georgia.7 Kallen et al.9 recently reported 51 cases from 19 states between November 2006 and April 2007. To the best of our knowledge, these reports represent the largest case series describing MRSA in CAP.
Earlier studies have suggested a relationship between the outcome of S. aureus infection and the presence of various patient and strain characteristics.1013 Most observations regarding the adverse impact of MRSA on patient outcomes have arisen from analyses of cohorts of patients with either mixed infections, bacteremias, or nosocomial infection. There is little information on outcomes in patients with S. aureus CAP. To address this issue, we conducted a retrospective study of patients with culture‐proven S. aureus pneumonia admitted to a large urban hospital.
Materials and Methods
Data Source
This retrospective study was conducted at Henry Ford Hospital in Detroit, MI, a 903‐bed tertiary care center. (Preliminary findings from the study have been presented at annual meetings of the Infectious Diseases Society of America [IDSA], the American College of Chest Physicians [ACCP], and the Interscience Conference on Antimicrobial Agents and Chemotherapy [ICAAC].) Data were obtained from the Henry Ford CarePlus Electronic Medical Record database, the Henry Ford Infectious Diseases Research Laboratory database, and the cost component of the Corporate Data Store, which is a central repository of data on patient encounters at Henry Ford Hospital and all Henry Ford Health System (HFHS) ambulatory care sites.
Sample Selection
The source population for the study consisted of all admissions to Henry Ford Hospital between January 2005 and May 2008 (study period) Patients were included in the study sample if they had: (1) a discharge diagnosis (principal or secondary) of pneumonia (International Classification of Diseases 9th Edition, Clinical Modification [ICD‐9‐CM] diagnosis codes 481.X 486.X) on their discharge summary or in their medical record; (2) a positive chest x‐ray (ie, infiltrate, consolidation, pleural effusion) within 48 hours of hospital admission; (3) an abnormal temperature (>38.3C [101.0F] or <36C [96.8F]), an abnormal white blood count (WBC) (>12,000/mm3 or <4,000/mm3), or increased sputum production on their day of hospital admission; and (4) a positive blood or respiratory culture for S. aureus within 48 hours of hospital admission.
Among these identified patients, those with probable HCAP were excluded from the study sample, based on evidence of: (1) hospitalization for 2 days during the 90‐day period preceding the index hospitalization; (2) admission to hospital from a nursing home or long‐term care facility; (3) hemodialysis 30 days prior to hospital admission; (4) receipt of cancer chemotherapy, IV antibiotic therapy, or wound care 30 days prior to hospital admission; or (5) receipt of an immunosuppressant at the time of hospital admission. All remaining patients were assumed to have CAP.
Data Extraction
For each admission in the study sample, selected demographic and clinical information was extracted from inpatient and outpatient medical records, beginning 1 year prior to the date of hospital admission and ending 30 days subsequent to the date of hospital discharge or the date of discontinuation of antibiotic therapy, whichever occurred later. All data were extracted by trained medical abstractors, using a set of case‐report forms developed specifically for this study.
Study Measures
Baseline demographic and clinical characteristics of study subjects were examined, including age, sex, race, evidence of positive S. aureus culture during the 1‐year period prior to hospital admission, history of selected disease conditions, and clinical status at admission (eg, comorbidities, vital signs, WBC, platelet count). A Confusion, Urea nitrogen, Respiratory rate, Blood pressure, 65 years of age and older score (CURB‐65) was calculated for each patient, based on clinical information at hospital admission.14 (A CURB‐65 score of 0 indicates a low [<1%] risk of death from pneumonia, while a score of 5 indicates a very high [57%] risk of death.)
Patients were designated as having MRSA or MSSA CAP based on the results of blood or respiratory cultures obtained within 48 hours of hospital admission. MRSA isolates were initially identified using automated dilution testing, in accordance with guidelines of the Clinical and Laboratory Standards Institute.15 The genotypic and phenotypic characteristics of MRSA isolates were examined, including susceptibility to vancomycin and presence of the Panton‐Valentine leukocidin (PVL) gene. Minimum inhibitory concentrations (MICs) for vancomycin were ascertained using the E‐test (AB BIODISK, Solna, Sweden).16 The PVL gene was detected using pulse field gel electrophoresis patterns (PFGE) and polymerase chain reaction. PFGE was performed using the restriction endonuclease SmaI. All S. aureus isolates were entered into a database using Gel Doc 2000 (BioRad) gel documentation system, and PFGE patterns were analyzed using BioNumerics Version 3.5, and grouped into Pulse‐field types using DICE coefficients and 80% relatedness.17
Initial antibiotic therapy was defined to consist of all antibiotics received within the first 48 hours in the hospital, regardless of sequence. Appropriateness of initial antibiotic therapy was ascertained based on susceptibility of the organism to the initial regimen received. Patients with MRSA isolates were designated as having received appropriate initial therapy if they were given vancomycin, linezolid or tigecycline. Those with MSSA isolates were designated as having received appropriate initial therapy if they were given a beta‐lactam, vancomycin, linezolid, or tigecycline. For patients with MRSA or MSSA that received doxycycline, clindamycin, or sulfamethoxazole/trimethoprim, each culture was evaluated individually to determine appropriateness, defined as in vitro susceptibility of the antimicrobial received to the organism. Clinical and economic outcomes of interest included: (1) thoracic surgery for pneumonia any time prior to hospital discharge; (2) receipt of mechanical ventilation any time prior to hospital discharge; (3) admission to an intensive care unit (ICU), irrespective of reason, anytime prior to hospital discharge; (4) length of stay in hospital; (5) total hospital charges for all services provided between hospital admission and hospital discharge; and (6) in‐hospital death (case‐fatality)
Statistical Analyses
We examined the baseline demographic and clinical characteristics of patients in the study sample, on an overall basis and for those with MRSA vs. MSSA isolates respectively. Categorical measures were summarized using frequency distributions and percentages; means, standard deviations (SDs) and medians were employed for continuous measures.
Clinical and economic outcomes were similarly examined, on an overall basis and for patients with MSSA vs. MRSA isolates. For patients with MRSA, we also examined outcomes in relation to selected genotypic and phenotypic characteristics of MRSA isolates, including MIC to vancomycin (1.00 g/mL, 1.50 g/mL, and 2.00 g/mL) and presence of the gene for PVL toxin.
Statistical significance of differences between patients with MRSA vs. MSSA isolates was assessed using t‐tests for continuous measures, and chi‐square tests for categorical measures; statistical significance was similarly assessed for patients with the PVL toxin gene vs. those without it. Because the distribution of vancomycin MICs was highly skewed, statistical significance of differences in outcomes was not assessed in relation to this measure.
All analyses were conducted using SAS Proprietary Software, Release 9.1 (SAS Institute Inc., Cary, NC). Missing and/or incomplete case‐report form data were not imputed, as observations were presumed not to be missing at random.
Results
There were 282 admissions to Henry Ford Hospital between January 2005 and May 2008 of patients with pneumonia who had positive blood or respiratory cultures for S. aureus within 48 hours of admission. (The total number of admissions over this period of patients with a principal diagnosis of pneumonia was 3894). Twelve patients had a negative chest x‐ray or negative clinical findings for pneumonia on the day of hospital admission and were excluded from the analysis. An additional 142 (53%) patients had evidence of HCAP and were excluded from the analysis. The final study sample therefore consisted of 128 patients with S. aureus CAP; their demographic and clinical characteristics are presented in Table 1.
| Characteristic | Overall (n = 128) | MSSA (n = 73) | MRSA (n = 55) | MSSA vs. MRSA P Value |
|---|---|---|---|---|
| ||||
| Age, years | ||||
| 1734 | 10 (7.8) | 6 (8.2) | 4 (7.3) | 1.000 |
| 3549 | 22 (17.2) | 12 (16.4) | 10 (18.2) | 0.817 |
| 5064 | 47 (36.7) | 26 (35.6) | 21 (38.2) | 0.854 |
| 65 | 49 (38.3) | 29 (39.7) | 20 (36.4) | 0.717 |
| Mean (SD) | 59.8 (17.0) | 59.8 (16.6) | 59.8 (17.8) | 0.999 |
| Sex | ||||
| Male | 74 (57.8) | 35 (47.9) | 39 (70.9) | 0.011 |
| Female | 54 (42.2) | 38 (52.1) | 16 (29.1) | |
| Race | ||||
| African American | 86 (67.2) | 46 (63.0) | 40 (72.7) | 0.261 |
| Caucasian | 36 (28.1) | 24 (32.9) | 12 (21.8) | 0.233 |
| Other/unknown | 6 (4.7) | 3 (4.1) | 3 (5.5) | 1.000 |
| Prior positive S. aureus culture* | ||||
| MSSA | 4 (3.1) | 4 (5.5) | 0 (0.0) | 0.134 |
| MRSA | 4 (3.1) | 1 (1.4) | 3 (5.5) | 0.314 |
| Both | 0 (0.0) | 0 (0.0) | 0 (0.0) | |
| Comorbidities | ||||
| Active malignancy | 11 (8.6) | 6 (8.2) | 5 (9.1) | 1.000 |
| Acute renal failure | 60 (46.9) | 31 (42.5) | 29 (52.7) | 0.285 |
| Coronary artery bypass grafting | 7 (5.5) | 5 (6.8) | 2 (3.6) | 0.698 |
| Coronary artery disease | 39 (30.5) | 21 (28.8) | 18 (32.7) | 0.700 |
| Cerebrovascular disease | 23 (18.0) | 15 (20.5) | 8 (14.5) | 0.487 |
| Congestive heart failure | 40 (31.3) | 22 (30.1) | 18 (32.7) | 0.848 |
| Chronic renal failure | 23 (18.0) | 12 (16.4) | 11 (20.0) | 0.647 |
| Chronic obstructive pulmonary disease | 31 (24.2) | 14 (19.2) | 17 (30.9) | 0.147 |
| Diabetes | 38 (29.7) | 19 (26.0) | 19 (34.5) | 0.332 |
| Diabetes mellitus with organ damage | 4 (3.1) | 2 (2.7) | 2 (3.6) | 1.000 |
| End‐stage renal disease with receipt of dialysis | 1 (0.8) | 1 (1.4) | 0 (0.0) | 1.000 |
| HIV/AIDS | 7 (5.5) | 2 (2.7) | 5 (9.1) | 0.138 |
| Myocardial infarction | 27 (21.1) | 15 (20.5) | 12 (21.8) | 1.000 |
| Peripheral vascular disease | 26 (20.3) | 10 (13.7) | 16 (29.1) | 0.045 |
| CURB‐65 | ||||
| 0 | 15 (11.7) | 6 (8.2) | 9 (16.4) | 0.175 |
| 1 | 36 (28.1) | 21 (28.8) | 15 (27.3) | 1.000 |
| 2 | 48 (37.5) | 28 (38.4) | 20 (36.4) | 0.855 |
| 3 | 20 (15.6) | 14 (19.2) | 6 (10.9) | 0.229 |
| 4 | 8 (6.3) | 3 (4.1) | 5 (9.1) | 0.288 |
| 5 | 1 (0.8) | 1 (1.4) | 0 (0.0) | 1.000 |
| Mean(SD) | 1.8 (1.1) | 2 (1.0) | 2 (1.2) | 0.379 |
Mean (SD) age of study subjects was 60 (17.0) years; 72% were nonwhite, and 58% were males. Prevalence of selected comorbidities, including acute renal failure, coronary artery disease, congestive heart failure, chronic obstructive pulmonary disease, diabetes, myocardial infarction, and peripheral vascular disease was high (>20%). Mean (SD) CURB score at admission was 1.8 (1.1); scores did not differ significantly between MRSA vs. MSSA patients. Eight patients (6%) had evidence of positive S. aureus cultures in the year prior to admission.
A total of 55 (43%) patients had positive MRSA cultures within the first 48 hours of admission, while the remainder (n = 73 [57%]) had cultures positive for MSSA. Among the 55 patients with MRSA, 23 (42%) had USA300 MRSA strains that produced PVL toxin. Only 18% of MRSA patients had MICs to vancomycin of 1 g/mL; most remaining patients (73%) had 1.5 g/mL MIC values. Thirty‐one patients (24%) died prior to hospital discharge (Table 2). Eighty‐eight (69%) patients received mechanical ventilation, and 79% were admitted to ICU; 25% of patients underwent thoracic surgery for pneumonia. Mean (SD) length of stay in hospital was 17.0 (15.7) days, and mean (SD) total charges per hospitalization were $127,922 ($154,605). (Mean length of stay in hospital was 5.8 days for all 3894 patients with a principal discharge diagnosis of pneumonia, and mean total charges were $29,448).
| Study Outcomes | Overall (n = 128) | MSSA (n = 73) | MRSA (n = 55) | P Value | PVL Toxin* (n = 23) | No PVL Toxin* (n = 28) | P Value |
|---|---|---|---|---|---|---|---|
| |||||||
| Case fatality | 31 (24.2) | 19 (26.0) | 12 (21.8) | 0.678 | 4 (17.4) | 6 (21.4) | 1.000 |
| Surgery for pneumonia | 32 (25.0) | 19 (26.0) | 13 (23.6) | 0.838 | 7 (30.4) | 5 (17.9) | 0.329 |
| Receipt of mechanical ventilation | 88 (68.8) | 56 (76.7) | 32 (58.2) | 0.034 | 12 (52.2) | 18 (64.3) | 0.576 |
| ICU admission | 101 (78.9) | 60 (82.2) | 41 (74.5) | 0.382 | 15 (65.2) | 22 (78.6) | 0.348 |
| Length of stay, days | |||||||
| Mean (SD) | 17.2 (15.7) | 16.4 (15.0) | 18.2 (16.6) | 0.525 | 25.3 (22.6) | 13.2 (7.7) | 0.020 |
| Median | 13.0 | 13.0 | 13.0 | 16.0 | 12.0 | ||
| Total charges, $ | |||||||
| Mean (SD) | 127,922 (154,605) | 135,784 (170,046) | 117,489 (132,164) | 0.510 | 162,124 (186,923) | 85,215 (57,957) | 0.066 |
| Median | 81,374 | 84,593 | 71,868 | 106,599 | 67,328 | ||
Notwithstanding the fact that patients with MRSA were more than twice as likely to receive inappropriate initial therapy (44% for MRSA vs. 18% for MSSA; odds ratio [OR], 3.57; 95% confidence interval [CI], 1.607.97; P = 0.0015), there were no statistically significant differences in in‐hospital mortality (26% for MSSA vs. 22% for MRSA; P = 0.678), surgery for pneumonia (26% vs. 24%; P = 0.838), or admission to ICU (82% vs. 74%; P = 0.382). Patients with MSSA CAP were more likely to receive mechanical ventilation (77% vs. 58%; P = 0.034). Mean (SD) total charges per admission did not differ between patients with MSSA vs. MRSA CAP ($135,784 [$170,046] vs $117,489 [$132,164], respectively [P = 0.510]). (30‐day mortality following hospital discharge was 24% for MRSA CAP patients and 30% for MSSA CAP patients.)
Among patients with MRSA CAP, there were no notable differences in outcomes between those with strains with the PVL toxin vs. those without it (Table 2). Mean (SD) length of stay in hospital, however, was significantly longer among the former patients (25 [22.6] days vs. 13 [7.7] days for those without PVL toxin; P = 0.020). Reflecting this difference, mean (SD) total charges per admission were $76,909 higher among MRSA patients with PVL‐positive strains vs. those with PVL‐negative strains ($162,124 [$186,923] vs. $85,215 [$57,957] respectively; P = 0.066).
Among MRSA patients with MICs to vancomycin of 1.5 g/mL, 17% died in hospital, 20% underwent surgery for pneumonia, 60% received mechanical ventilation, and 75% were admitted to ICU. Mean (SD) total charges per admission were $104,514 ($112,606) (Table 2).
Discussion
To the best of our knowledge, this is the largest observational study to date of outcomes in patients with S. aureus CAP. Our results indicate that MRSA represents almost one‐half of all such infections, with USA300 strains accounting for a substantial proportion of these cases. In addition, for most outcome measures, there were no significant differences between patients with MSSA vs. MRSA CAP. In patients with MRSA CAP, however, those with PVL‐positive strains had longer stays in the hospital and had higher total costs of hospitalization.
Our study highlights the significant clinical and economic burden of S. aureus CAP. In the US, the mortality rate in patients hospitalized with CAP is about 8%, while mean length of stay is approximately 5 days (it was 5.8 days in our institution among all patients with CAP). In contrast, we found that most patients with S. aureus CAP were admitted to ICU, and that nearly one in four died during hospitalization. One possible explanation for these findings is that patients with S. aureus CAP may often receive inappropriate initial antibiotic therapy, a major predictor of adverse outcome in infection. The overall rate of inappropriate treatment, however, was low in our study (although we note that optimal antibiotic treatment is not clearly defined for the USA300 strain, and as a result, we assumed that vancomycin would be appropriate for all MRSA infections). A more likely explanation for our findings is the severity of CAP associated with S. aureus, whether MRSA or MSSA. Given the nature of S. aureus infections, the need for prevention (and potentially, development of a vaccine) remains crucial.
While MRSA has been studied extensively as a cause of bacteremia, as well as HCAP and VAP, it has not been well studied in CAP. Patients with MRSA bacteremia have been shown to have a higher mortality risk and higher healthcare costs than those with MSSA infections, and a meta‐analysis of 31 studies of patients with S. aureus bloodstream infections demonstrated a significant increase in mortality among patients with MRSA vs. MSSA bacteremia.1820 Outcomes when comparing MRSA and MSSA in VAP are slightly more variable. A higher rate of mortality has been reported in patients with pneumonia caused by MRSA vs. MSSA, but others found no difference after controlling for potential confounders.21, 22 We found that case fatality, surgery for pneumonia, ICU admission, length of stay, and total hospital charges do not differ significantly between patients with MRSA vs. MSSA CAP. This suggests that appreciation of the burden of MRSA can only be done in the context of the syndrome in question, and that conclusions from analyses of bacteremia and VAP may not be generalizable to CAP.
In the US, an increasing number of community‐associated infections are due to MRSA. Skin and skin structure infections comprise the majority of community‐associated MRSA infections and are caused by a single pulsed field type, termed USA300. These strains are believed to have distinctive virulence and epidemiologic characteristics. USA300 isolates typically are resistant only to beta‐lactam and macrolide antimicrobial agents and contain genes for the PVL toxin, which typically are not present in strains of healthcare‐associated MRSA.23 Recent studies of acute pneumonia with animal models and in humans have suggested that the PVL toxinalone or in combination with other virulence factorsis associated with the development of necrotizing pneumonia.2430 In our study, 23 patients with MRSA CAP had strains that contained PVL toxin, and these patients had longer stays in hospital and higher total hospital charges than those with MRSA CAP not containing the PVL toxin. This is an important finding, as length of stay is an important proxy for morbidity and case severity. This also extends the finding of previous case studies reporting that MRSA CAP with PVL toxin is associated with worse outcomes.2430 Moreover, we clearly document that PVL‐positive strains are emerging as a cause of pulmonary infection in broader clinical scenarios. As such, physicians must remain vigilant for this toxin‐producing strain. The full extent of the impact of PVL‐positive strains in our study is unknown, as we cannot ascertain whether morbidity was worse because of the PVL toxin itself, or because most patients were not treated with clindamycin or linezolid, which inhibit toxin production.
We failed to observe a correlation between MICs to vancomycin and the outcomes we studied. The clinical significance of reduced susceptibility of S. aureus to vancomycin remains a controversial issue. In our study, there was no significant association between MICs to vancomycin and mortality, need for surgery, ICU admission, length of stay, or total hospital charges. Sakoulas et al.11 reported that as vancomycin MICs for MRSA isolates rose within the susceptible range from 0.5 mcg/mL to 2.0 mcg/mL, so too did the number of clinical failures among bacteremic patients receiving vancomycin. Moise‐Broder et al.31 reported a similar finding in a study of 63 patients with MRSA bacteremia, of whom 45 failed or were intolerant of vancomycin therapy. In a more recent report, Soriano et al.12 prospectively evaluated 414 episodes of bacteremia. The authors concluded that mortality in patients with MRSA bacteremia is significantly higher when the empiric antimicrobial agent is inappropriate and when vancomycin is used to treat infections involving strains with MICs >1.0 mcg/mL. In pneumonia, Hidayat et al.13 reported that patients with infections due to pathogens with higher MICs to vancomycin had a higher rate of mortality than those with lower MICs. Our findings likely differ from this report because of the proportion of subjects with PVL‐positive strains, and because of the skew in the distribution of vancomycin MICs. More specifically, the majority of patients had higher MIC strains, clustered around 1.5 mg/L, leaving us with few lower MIC strains and fewer strains with MICs of 2.0 mcg/mL for comparison, and therefore limited statistical power to assess the relationship between MICs and outcomes.
There were no clinical features that clearly distinguished patients with MRSA vs. MSSA CAP. This suggests that if physicians hope to ensure that patients with MRSA CAP receive appropriate initial antibiotic therapy, they cannot base therapeutic decision‐making (ie, use of an anti‐MRSA treatment) on clinical criteria. Presently, national guidelines recommend diagnostic testing only if the results might affect clinical decisions (eg, antimicrobial management).32 Furthermore, the IDSA/ATS guidelines recommend sputum cultures along with blood cultures and other diagnostic tests only in select cases (eg, those with severe disease). Since beginning optimal therapy quickly can reduce mortality in pneumonia,33 our results indicate a need for both rapid diagnostic tests to identify patients with MRSA, and reevaluation of current recommendations for diagnostic testing in CAP.
Our study has several important limitations. First, its retrospective design exposes it to many forms of bias. Second, because the diagnosis of pneumonia can be challenging, we required that all patients have both clinical and radiologic evidence of the disease. However, we may have thereby excluded some admissions for S. aureus pneumonia among the over 3000 pneumonia admissions to our hospital during the study period.
Third, we only included patients with culture evidence of infection. This was necessary by design, but as a result we may have failed to identify some patients with S. aureus infections because of the limitations of respiratory culture technology. Patients on general practice units often do not get cultured, and accordingly we may have missed those with milder disease caused by S. aureus.
Fourth, we used only one method of MIC testing to determine vancomycin susceptibility. It would have been of interest to have used automated dilution testing also and to have compared both methods. Comparison of laboratory methods was not among our study objectives, and most of the earlier studies examining epidemiology and outcomes used a single laboratory testing method only.
Finally, since the study was conducted at a large urban teaching hospital in a city with a long history of issues with MRSA and resistance, the generalizability of our results may be limited.
In summary, our study provides further evidence that S. aureus is an important pathogen in CAP. MRSA CAP has few obvious characteristics that differentiate it from MSSA CAP. MRSA and MSSA CAP are both very serious infections that should be treated aggressively to avoid poor outcomes.
- .Staphylococcus aureus: a well‐armed pathogen.Clin Infect Dis.1998;26:1179–1181.
- .Staphylococcus aureus infections.N Engl J Med.1998;339:520–532.
- .The growing menace of community‐acquired methicillin‐resistant Staphylococcus aureus.Ann Intern Med.2006;144:368–370.
- ,,, et al.Predictors of mortality for methicillin‐resistant Staphylococcus aureus health‐care‐associated pneumonia: specific evaluation of vancomycin pharmacokinetic indices.Chest.2006;130:947–955.
- ,,,.Bacterial pneumonia during the Hong Kong influenza epidemic of 1968–1969.Arch Intern Med.1971;127:1037–1041.
- ,,, et al.Severe community‐acquired pneumonia due to Staphylococcus aureus, 2003–04 influenza season.Emerg Infect Dis.2006;12:894–899.
- Centers for Disease Control and Prevention.Severe methicillin‐resistant Staphylococcus aureus community‐acquired pneumonia associated with influenza—Louisiana and Georgia, December 2006‐January 2007.Morb Mortal Wkly Rep.2007;56:325–329.
- ,,.Incidence, characteristics and outcomes of patients with severe community acquired‐MRSA pneumonia.Eur Respir J.2009;34:1148–1158.
- ,,, et al.Staphylococcus aureus community‐acquired pneumonia during the 2006 to 2007 influenza season.Ann Emerg Med.2009;53:358–365.
- ,,, et al.Treatment outcomes for serious infections caused by methicillin‐resistant Staphylococcus aureus with reduced vancomycin susceptibility.Clin Infect Dis.2004;38:521–528.
- ,,, et al.Relationship of MIC and bactericidal activity to efficacy of vancomycin for treatment of methicillin‐resistant Staphylococcus aureus bacteremia.J Clin Microbiol.2004;42:2398–2402.
- ,,, et al.Influence of vancomycin minimum inhibitory concentration on the treatment of Methicillin‐resistant Staphylococcus aureus bacteremia.Clin Infect Dis.2008;46:193–200.
- ,,, et al.High‐dose vancomycin therapy for methicillin‐resistant Staphylococcus aureus infections.Arch Intern Med.2006;166:2138–2144.
- ,,, et al.Defining community acquired pneumonia severity on presentation to hospital: An international derivation and validation study.Thorax.2003;58(5):377–382.
- Clinical and Laboratory Standards Institute (CLSI).Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically: Approved Standard. 8th ed. CLSI document M07‐A8 2008;29(2):1–65. Available at: www.clsi.org/source/orders/free/m07‐a8.pdf.
- ,,,.A multicenter study evaluating the current strategies for isolating Staphylococcus aureus strains with reduced susceptibility to glycopeptides.J Clin Microbiol.2007;45:329–332.
- ,,,,.Application of molecular techniques to the study of hospital infection.Clin Microbiol Rev.2006;19:512–530.
- ,,.Nosocomial methicillin‐resistant Staphylococcus aureus bacteremia: Is it any worse than nosocomial methicillin‐sensitive Staphylococcus aureus bacteremia?Infect Control Hosp Epidemiol.2000;21:645–648.
- ,.Clinical and economic impact of methicillin resistance in patients with Staphylococcus aureus bacteremia.Diagn Microbiol Infect Dis.2005;52:113–122.
- ,,, et al.Comparison of mortality associated with methicillin‐resistant and methicillin‐susceptible Staphylococcus aureus bacteremia: A meta‐analysis.Clin Infect Dis.2003;36:53–59.
- ,,, et al.Ventilator‐associated pneumonia by Staphylococcus aureus: Comparison of methicillin‐resistant and methicillin‐sensitive episodes.Am J Respir Crit Care Med.1994;150:1545–1549.
- ,,, et al.Is methicillin resistance associated with a worse prognosis in Staphylococcus aureus ventilator‐associated pneumonia?Clin Infect Dis.2005;41:1224–1231.
- ,,, et al.Characterization of a strain of community‐associated methicillin‐resistant Staphylococcus aureus widely disseminated in the United States.J Clin Microbiol.2006;44:108–111.
- .Staphylococcus aureus, Panton‐Valentine leukocidin, and necrotising pneumonia.BMJ.2005;331:793–794.
- ,,, et al.Methicillin‐resistant Staphylococcus aureus necrotizing pneumonia.Emerg Infect Dis.2005;11:1647–1648.
- ,.Fatal necrotizing pneumonia due to community‐acquired methicillin‐resistant Staphylococcus aureus (MRSA).Med J Aust.2004;181:228–229.
- ,,.Pleuropulmonary complications of Panton‐Valentine leukocidin‐positive community‐acquired methicillin‐resistant Staphylococcus aureus: Importance of treatment with antimicrobials inhibiting exotoxin production.Chest.2005;128:2732–2738.
- ,,, et al.Severe community‐onset pneumonia in healthy adults caused by methicillin‐resistant Staphylococcus aureus carrying the Panton‐Valentine leukocidin genes.Clin Infect Dis.2005;40:100–107.
- ,,, et al.Staphylococcus aureus Panton‐Valentine leukocidin causes necrotizing pneumonia.Science.2007;315:1130–1133.
- ,,, et al.Association between Staphylococcus aureus strains carrying gene for Panton‐Valentine leukocidin and highly lethal necrotizing pneumonia in young immunocompetent patients.Lancet.2002;359:753–759.
- ,,, et al.Accessory gene regulator group II polymorphism in methicillin‐resistant Staphylococcus aureus is predictive of failure of vancomycin therapy.Clin Infect Dis.2004;38:1700–1705.
- ,,, et al.Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community‐acquired pneumonia in adults.Clin Infect Dis.2007;44(Suppl 2):S27–S72.
- ,,, et al.Antimicrobial therapy escalation and hospital mortality among patients with health‐care‐associated pneumonia: a single center experience.Chest.2008;134:963–968.
- .Staphylococcus aureus: a well‐armed pathogen.Clin Infect Dis.1998;26:1179–1181.
- .Staphylococcus aureus infections.N Engl J Med.1998;339:520–532.
- .The growing menace of community‐acquired methicillin‐resistant Staphylococcus aureus.Ann Intern Med.2006;144:368–370.
- ,,, et al.Predictors of mortality for methicillin‐resistant Staphylococcus aureus health‐care‐associated pneumonia: specific evaluation of vancomycin pharmacokinetic indices.Chest.2006;130:947–955.
- ,,,.Bacterial pneumonia during the Hong Kong influenza epidemic of 1968–1969.Arch Intern Med.1971;127:1037–1041.
- ,,, et al.Severe community‐acquired pneumonia due to Staphylococcus aureus, 2003–04 influenza season.Emerg Infect Dis.2006;12:894–899.
- Centers for Disease Control and Prevention.Severe methicillin‐resistant Staphylococcus aureus community‐acquired pneumonia associated with influenza—Louisiana and Georgia, December 2006‐January 2007.Morb Mortal Wkly Rep.2007;56:325–329.
- ,,.Incidence, characteristics and outcomes of patients with severe community acquired‐MRSA pneumonia.Eur Respir J.2009;34:1148–1158.
- ,,, et al.Staphylococcus aureus community‐acquired pneumonia during the 2006 to 2007 influenza season.Ann Emerg Med.2009;53:358–365.
- ,,, et al.Treatment outcomes for serious infections caused by methicillin‐resistant Staphylococcus aureus with reduced vancomycin susceptibility.Clin Infect Dis.2004;38:521–528.
- ,,, et al.Relationship of MIC and bactericidal activity to efficacy of vancomycin for treatment of methicillin‐resistant Staphylococcus aureus bacteremia.J Clin Microbiol.2004;42:2398–2402.
- ,,, et al.Influence of vancomycin minimum inhibitory concentration on the treatment of Methicillin‐resistant Staphylococcus aureus bacteremia.Clin Infect Dis.2008;46:193–200.
- ,,, et al.High‐dose vancomycin therapy for methicillin‐resistant Staphylococcus aureus infections.Arch Intern Med.2006;166:2138–2144.
- ,,, et al.Defining community acquired pneumonia severity on presentation to hospital: An international derivation and validation study.Thorax.2003;58(5):377–382.
- Clinical and Laboratory Standards Institute (CLSI).Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically: Approved Standard. 8th ed. CLSI document M07‐A8 2008;29(2):1–65. Available at: www.clsi.org/source/orders/free/m07‐a8.pdf.
- ,,,.A multicenter study evaluating the current strategies for isolating Staphylococcus aureus strains with reduced susceptibility to glycopeptides.J Clin Microbiol.2007;45:329–332.
- ,,,,.Application of molecular techniques to the study of hospital infection.Clin Microbiol Rev.2006;19:512–530.
- ,,.Nosocomial methicillin‐resistant Staphylococcus aureus bacteremia: Is it any worse than nosocomial methicillin‐sensitive Staphylococcus aureus bacteremia?Infect Control Hosp Epidemiol.2000;21:645–648.
- ,.Clinical and economic impact of methicillin resistance in patients with Staphylococcus aureus bacteremia.Diagn Microbiol Infect Dis.2005;52:113–122.
- ,,, et al.Comparison of mortality associated with methicillin‐resistant and methicillin‐susceptible Staphylococcus aureus bacteremia: A meta‐analysis.Clin Infect Dis.2003;36:53–59.
- ,,, et al.Ventilator‐associated pneumonia by Staphylococcus aureus: Comparison of methicillin‐resistant and methicillin‐sensitive episodes.Am J Respir Crit Care Med.1994;150:1545–1549.
- ,,, et al.Is methicillin resistance associated with a worse prognosis in Staphylococcus aureus ventilator‐associated pneumonia?Clin Infect Dis.2005;41:1224–1231.
- ,,, et al.Characterization of a strain of community‐associated methicillin‐resistant Staphylococcus aureus widely disseminated in the United States.J Clin Microbiol.2006;44:108–111.
- .Staphylococcus aureus, Panton‐Valentine leukocidin, and necrotising pneumonia.BMJ.2005;331:793–794.
- ,,, et al.Methicillin‐resistant Staphylococcus aureus necrotizing pneumonia.Emerg Infect Dis.2005;11:1647–1648.
- ,.Fatal necrotizing pneumonia due to community‐acquired methicillin‐resistant Staphylococcus aureus (MRSA).Med J Aust.2004;181:228–229.
- ,,.Pleuropulmonary complications of Panton‐Valentine leukocidin‐positive community‐acquired methicillin‐resistant Staphylococcus aureus: Importance of treatment with antimicrobials inhibiting exotoxin production.Chest.2005;128:2732–2738.
- ,,, et al.Severe community‐onset pneumonia in healthy adults caused by methicillin‐resistant Staphylococcus aureus carrying the Panton‐Valentine leukocidin genes.Clin Infect Dis.2005;40:100–107.
- ,,, et al.Staphylococcus aureus Panton‐Valentine leukocidin causes necrotizing pneumonia.Science.2007;315:1130–1133.
- ,,, et al.Association between Staphylococcus aureus strains carrying gene for Panton‐Valentine leukocidin and highly lethal necrotizing pneumonia in young immunocompetent patients.Lancet.2002;359:753–759.
- ,,, et al.Accessory gene regulator group II polymorphism in methicillin‐resistant Staphylococcus aureus is predictive of failure of vancomycin therapy.Clin Infect Dis.2004;38:1700–1705.
- ,,, et al.Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community‐acquired pneumonia in adults.Clin Infect Dis.2007;44(Suppl 2):S27–S72.
- ,,, et al.Antimicrobial therapy escalation and hospital mortality among patients with health‐care‐associated pneumonia: a single center experience.Chest.2008;134:963–968.
Copyright © 2010 Society of Hospital Medicine
Fragmentation of Care
There is a potential for discontinuity of care in the management of inpatients by hospitalists since many patients are cared for by more than one physician during their hospitalization. Previous studies have explored the impact of this type of discontinuity of care in residency programs. With the restrictions put in place by the Accreditation Council on Graduate Medical Education (ACGME) in 2003, there has been an increase in the fragmentation of care (FOC) that patients receive in the hospital. Studies have explored the impact of these changes on length of stay (LOS) and quality of care. The results have been mixed, with some studies showing that the increased FOC was associated with prolonged LOS,1, 2 as well as having a negative influence on quality.2, 3 Other studies have shown no change or a reduction in LOS,4 and an improvement in quality measures.46
There have been no prior studies on the impact of hospitalist care as the source of FOC on LOS. We therefore undertook a study to explore the impact of fragmented hospitalist care on LOS. Additionally, there has been some discussion on the impact of admission day of week on both FOC and LOS. Prior studies have mainly looked at day of the week in intensive care unit patients. One study found a modest increase in ICU LOS in patients admitted during the weekend.7 We therefore also looked at the impact of day of the week of admission on FOC in order to determine if fragmentation was just a proxy for admission day as the true indicator for increased LOS.
Methods
Design Overview
Data for this study consisted of clinical and billing information from IPC.
Setting and Participants
Data on LOS and FOC were drawn from the clinical and billing database of IPC The Hospitalist Company, which is a national group practice of hospitalists that at the time of the study had practices in over 230 acute health care facilities in 24 healthcare markets across the United States. Physicians enter clinical and billing data into IPC‐Link, a proprietary web based program.
Patients included in this study were discharged between December 1, 2006 and November 30, 2007 with a diagnosis‐related group (DRG) of 89 Pneumonia with complications or comorbidities (PNA) or a DRG of 127 Heart failure and shock (HF). A total of 10,977 patients with DRG 89 (n = 1951) or DRG 127 (n = 9026) were identified. These patients were admitted to 223 hospitals in 16 states.
Outcome
The outcome measure of interest was LOS.
FOC Measure
The independent variable of interest was the measure of fragmentation of physician care during the hospital stay. The intuitive meaning of fragmentation in physician hospital services is the delivery of care by multiple hospitalist physicians within a single stay. We converted the idea of fragmentation into a quantitative measure, an FOC Index, by calculating the percentage of hospitalist care delivered by physicians other than the primary hospitalist. The primary hospitalist was defined as the physician responsible for the largest number of visits during the hospital stay. Fragmentation was calculated by summing the number of daily billable encounters by IPC hospitalists other than the primary hospitalist and dividing by the total number of billable encounters by all IPC hospitalists. Thus, if three days out of a five‐day stay, the patient was seen by one hospitalist, then 60% of the care would be by the primary hospitalist. The patient was seen by other physicians than the primary hospitalist 2 out of the 5 days, for a FOC Index of 40%. IPC billing data was used to identify each hospitalist visit during the hospital stay by physician, date, and type of visit.
Covariates
The clinical database contained potential covariates: age, gender, day of admission, the number of ICD‐9 diagnosis codes on the discharge summary, and severity of illness (SOI) and risk of mortality (ROM) measures. The 3M DRG Grouper (St. Paul, Minnesota), using clinical data from the IPC‐Link discharge summary, assigned each patient an SOI score, and an ROM score based on age, primary diagnosis, and non‐operating room procedures.8 Both SOI and ROM are scored from 1 to 4, ranging from 1 for minor to 4 for extreme. We created dummy variables for the SOI and ROM categories, that is, variables for each score with a value of 1 if the SOI or ROM is that score and 0, otherwise. In each case, a score of 1 was the referent category. Dummy variables were created for each admission day of the week with Monday as the referent. Demographic variables and variables related to the patient condition were included to reduce any potential confounding arising from differences in patient populations across hospitals and hospitalist practice patterns. Admission day of the week has been shown to influence LOS and may influence FOC due to weekend scheduling within hospitalist practices.
Statistical Analysis
All analyses were performed using SAS (version 9.1). To reduce the influence of outliers on the model, observations with extreme values for LOS (defined as greater than 3 standard deviation [STD] from the mean) were truncated to 30 days. For pneumonia, the mean LOS was 7.85 days (STD 7.44) and all patients with LOS greater than 30 days were truncated to 30 days. For heart failure, the mean LOS was 5.88 days (STD 9.77) and all patients with LOS greater than 35 days were truncated to 35 days. Multivariable regression was performed using the negative binomial distribution. Negative binomial regression used as LOS is not normally distributed, but instead has a positive skew. Forward stepwise selection was performed separately for patients with each DRG using variables from the clinical database described in the Methods section. Categorical dummy variables were entered as a group. The final model contained all variables that were significant (P < 0.05).
Results
Table 1 reviews the demographics of patients with DRG 89 (PNA) and DRG 127 (HF). A large proportion of patients with both DRGs experienced no fragmentation in their physician care during hospitalization; 4152 patients with HF (46.3%) and 685 patients with PNA (35.8%) had visits from only 1 hospitalist throughout their hospital stay, for a FOC Index of 0%. The mean fragmentation level was 21.9% for pneumonia patients, with the mean number of hospitalists seen during the stay at 2.05. For heart failure patients, the mean fragmentation level was 18.3 % and a mean of 1.78 hospitalists seen (Table 1).
| DRG 89 (n = 1950), Mean (STD) | DRG 127 (n = 9026), Mean (STD) | |
|---|---|---|
| ||
| Age, years | 65.8 (18.6) | 71.4 (15.2)* |
| % Female | 49.7 | 51.7 |
| Number of secondary diagnoses | 5.6 (2.5) | 5.0 (2.3)* |
| Severity of Illness | 2.80 (0.57) | 1.943 (0.72)* |
| Risk of Mortality | 2.49 (0.84) | 1.81 (0.75)* |
| Fragmentation: percent of care by non‐primary hospitalist(s) | 21.9% (20.3) | 18.3% (19.9)* |
| Number of physicians seen during hospital stay | 2.05 (1.12) | 1.78 (0.94)* |
Table 2 presents the results of the regression analysis on LOS. The association between fragmentation and LOS, adjusted for demographics, case mix and day of admission, was similar for PNA and HF patients. We found an increase in LOS of 0.39 days for each 10% increase in fragmentation level for pneumonia. Other variables that significantly increased LOS for PNA were larger ROM score, Sunday admission and more discharge diagnoses. Similarly, for heart failure patients, there was an increase in LOS of 0.30 days for each 10% increase in fragmentation level. Other variables associated with a significantly increase in LOS for PNA were larger ROM score, larger SOI category, more discharge diagnoses, and age. Figures 1 and 2 demonstrate the results graphically for LOS in each condition. They show the mean adjusted LOS by fragmentation level. Adjusted LOS was calculated for each patient based on the final model. For example, the average adjusted LOS for PNA patients with a fragmentation level of 20% to 30% is 8.06 days and 9.16 days for patients with a fragmentation level of 30% to 40%.
| Pneumonia (DRG = 89), n = 1950 | Heart Failure (DRG = 127), n = 9026 | |
|---|---|---|
| ||
| Fragmentation | Additional 0.39 day for each 10% increase in fragmentation | Additional 0.30 day for each 10% increase in fragmentation |
| Adjustment variables in the model | Age, number of secondary diagnoses, severity of illness variables, risk of mortality variables | Age, gender, number of secondary diagnoses. severity of illness variables, risk of mortality variables |

Discussion
This study demonstrated that there was a statistically significant association between FOC by hospitalist physicians and LOS for patients with DRG 89 (PNA) and DRG 127 (HF). As the percentage of fragmentation increased, the LOS increased significantly.
There are many ways in which to define FOC in the context of hospitalist care of inpatients. We chose to use an index similar to the Usual Provider of Care Index (UPC) which is a standard way to measure continuity of care in the primary care setting.9 The UPC measures the proportion of time spent with the primary provider. In the inpatient setting, we defined continuity of care as the proportion of visits by the primary hospitalist (the physician assuming the greatest number of days of the patient's care). The FOC Index is simply 1 minus the continuity of care. There are other potential measures of FOC, such as the number of handoffs or the number of physicians. We selected our measure based on its following strengths: (1) it does not have the simultaneity problem of measures such as the number of doctors or the number of handoffs that leads to endogeneitya correlation between the error term and the independent variable that biases the coefficient. Endogeneity can occur when the outcome variable and the independent variable are jointly determined, in this case, that an additional day in the hospital increases the likelihood of having an additional handoff as well as an additional handoff increasing the likelihood of an additional day; (2) fragmentation patterns that lead to handoffs on the weekend that then return to the primary hospitalist may be very different from fragmentation patterns that lead to new physicians at each transfer; (3) It provides a good comparison to other models of care where community physicians provide care to their own patients and have an effective FOC index of 0. Further research should be performed looking at different measures that may capture other aspects of care fragmentation.
This is one of the first studies to directly explore the relationship between FOC and LOS. Previous studies in the hospitalist literature have explored the impact of variables on LOS in which FOC may have been an indirect influence. For example, one study found a 13% shorter LOS among academic hospitalists at a teaching hospital who worked on a block rotation compared to a group of community hospitalists with a schedule involving more patient handoffs.10 Although there is little literature on FOC among hospitalists, the literature on medical residency programs is informative. The amount of discontinuity and the number of housestaff‐to‐housestaff transfers of responsibility has increased dramatically since the institution of more stringent work‐hour restrictions by the Accreditation Council for Graduate Medical Education (ACGME) in 2003.11 Horwitz et al.12 found that after the institution of work‐hour regulations, there was an 11% increase in number of transfers of care for a hypothetical patient admitted from Monday until Friday. They noted that programs with a night‐float system had a statistically significant increase in transitions of care compared to residencies without night float. Studies on the impact of the ACGME regulations on LOS have been mixed. One study found that LOS was reduced, and that there was improved adherence to quality indicators.4 A recent study found that there was a 44% increase in the median LOS when Short Call admitting teams were involved.2
There are several limitations to the findings of this study. The largest potential limitation is interpreting the direction of causality between FOC and LOS. This was a concurrent control study. Because the study was not randomized, there is a risk of confounding variables. Admission day of the week was 1 variable of concern for confounding due to the existence of a significant relationship between admission day LOS and between admission day and FOC. We included admission day in our modeling process. The coefficient estimates on FOC were stable, changing less than 1% when day of admission was added to the model. The risk of temporal trends was minimized by studying 1‐year's worth of data. Although we looked at variables that were potentially relevant in all hospitalized patients, every hospital and hospitalist practice has its own unique features that may impact both LOS and FOC. For example, for some hospitals, bed capacity may impact these measures. In many hospitals, hospitalist work force shortages may impact these measures by affecting the extent to which patients are cared for by full‐time hospitalists vs. locums tenens physicians. The large number of facilities in the study mitigates the influence of any individual hospital's LOS and FOC tendencies.
If increased FOC does result in prolonged LOS, the question arises as to how this fragmentation can be decreased. Although no physician can guarantee presence continually throughout a patient's inpatient stay, there are scheduling methods that reduce fragmentation and maximize the odds of a patient being followed by a single clinician. For example, the longer the block of days that a hospitalist is scheduled, the fewer hospitalists will need to care for a patient. Many of the top DRGs for patients cared for by hospitalists have lengths of stay between 4 and 5 days.13 Therefore, any schedule that has a hospitalist on for at least four days at a time will increase the likelihood that the same hospitalist will care for many of the patients throughout their stay. In making schedules, the goals of clinical efficiency and physician satisfaction must be weighed against the potential risks to the quality of patient care. For instance, the use of one hospitalist as the admitting physician for all patients may increase efficiency, but will also increase fragmentation.
A factor that may influence the impact of FOC on LOS is the quality of patient handoffs. The Joint Commission instituted this as a national patient safety goal in 2006.14 This goal was based on a Joint Commission analysis that 70% of sentinel events were caused by communication breakdowns, half of these occurring during handoffs.15
In conclusion, this study explored the relationship between FOC in patients cared for by hospitalists and LOS, using an FOC index. For every 10% increase in fragmentation, the LOS went up by 0.39 days for pneumonia and 0.30 days for heart failure. By adjusting for many variables that may impact LOS due to higher severity or complexity of illness, there is an increased likelihood that the FOC may have a causative relationship with the prolonged LOS.
There is a potential for discontinuity of care in the management of inpatients by hospitalists since many patients are cared for by more than one physician during their hospitalization. Previous studies have explored the impact of this type of discontinuity of care in residency programs. With the restrictions put in place by the Accreditation Council on Graduate Medical Education (ACGME) in 2003, there has been an increase in the fragmentation of care (FOC) that patients receive in the hospital. Studies have explored the impact of these changes on length of stay (LOS) and quality of care. The results have been mixed, with some studies showing that the increased FOC was associated with prolonged LOS,1, 2 as well as having a negative influence on quality.2, 3 Other studies have shown no change or a reduction in LOS,4 and an improvement in quality measures.46
There have been no prior studies on the impact of hospitalist care as the source of FOC on LOS. We therefore undertook a study to explore the impact of fragmented hospitalist care on LOS. Additionally, there has been some discussion on the impact of admission day of week on both FOC and LOS. Prior studies have mainly looked at day of the week in intensive care unit patients. One study found a modest increase in ICU LOS in patients admitted during the weekend.7 We therefore also looked at the impact of day of the week of admission on FOC in order to determine if fragmentation was just a proxy for admission day as the true indicator for increased LOS.
Methods
Design Overview
Data for this study consisted of clinical and billing information from IPC.
Setting and Participants
Data on LOS and FOC were drawn from the clinical and billing database of IPC The Hospitalist Company, which is a national group practice of hospitalists that at the time of the study had practices in over 230 acute health care facilities in 24 healthcare markets across the United States. Physicians enter clinical and billing data into IPC‐Link, a proprietary web based program.
Patients included in this study were discharged between December 1, 2006 and November 30, 2007 with a diagnosis‐related group (DRG) of 89 Pneumonia with complications or comorbidities (PNA) or a DRG of 127 Heart failure and shock (HF). A total of 10,977 patients with DRG 89 (n = 1951) or DRG 127 (n = 9026) were identified. These patients were admitted to 223 hospitals in 16 states.
Outcome
The outcome measure of interest was LOS.
FOC Measure
The independent variable of interest was the measure of fragmentation of physician care during the hospital stay. The intuitive meaning of fragmentation in physician hospital services is the delivery of care by multiple hospitalist physicians within a single stay. We converted the idea of fragmentation into a quantitative measure, an FOC Index, by calculating the percentage of hospitalist care delivered by physicians other than the primary hospitalist. The primary hospitalist was defined as the physician responsible for the largest number of visits during the hospital stay. Fragmentation was calculated by summing the number of daily billable encounters by IPC hospitalists other than the primary hospitalist and dividing by the total number of billable encounters by all IPC hospitalists. Thus, if three days out of a five‐day stay, the patient was seen by one hospitalist, then 60% of the care would be by the primary hospitalist. The patient was seen by other physicians than the primary hospitalist 2 out of the 5 days, for a FOC Index of 40%. IPC billing data was used to identify each hospitalist visit during the hospital stay by physician, date, and type of visit.
Covariates
The clinical database contained potential covariates: age, gender, day of admission, the number of ICD‐9 diagnosis codes on the discharge summary, and severity of illness (SOI) and risk of mortality (ROM) measures. The 3M DRG Grouper (St. Paul, Minnesota), using clinical data from the IPC‐Link discharge summary, assigned each patient an SOI score, and an ROM score based on age, primary diagnosis, and non‐operating room procedures.8 Both SOI and ROM are scored from 1 to 4, ranging from 1 for minor to 4 for extreme. We created dummy variables for the SOI and ROM categories, that is, variables for each score with a value of 1 if the SOI or ROM is that score and 0, otherwise. In each case, a score of 1 was the referent category. Dummy variables were created for each admission day of the week with Monday as the referent. Demographic variables and variables related to the patient condition were included to reduce any potential confounding arising from differences in patient populations across hospitals and hospitalist practice patterns. Admission day of the week has been shown to influence LOS and may influence FOC due to weekend scheduling within hospitalist practices.
Statistical Analysis
All analyses were performed using SAS (version 9.1). To reduce the influence of outliers on the model, observations with extreme values for LOS (defined as greater than 3 standard deviation [STD] from the mean) were truncated to 30 days. For pneumonia, the mean LOS was 7.85 days (STD 7.44) and all patients with LOS greater than 30 days were truncated to 30 days. For heart failure, the mean LOS was 5.88 days (STD 9.77) and all patients with LOS greater than 35 days were truncated to 35 days. Multivariable regression was performed using the negative binomial distribution. Negative binomial regression used as LOS is not normally distributed, but instead has a positive skew. Forward stepwise selection was performed separately for patients with each DRG using variables from the clinical database described in the Methods section. Categorical dummy variables were entered as a group. The final model contained all variables that were significant (P < 0.05).
Results
Table 1 reviews the demographics of patients with DRG 89 (PNA) and DRG 127 (HF). A large proportion of patients with both DRGs experienced no fragmentation in their physician care during hospitalization; 4152 patients with HF (46.3%) and 685 patients with PNA (35.8%) had visits from only 1 hospitalist throughout their hospital stay, for a FOC Index of 0%. The mean fragmentation level was 21.9% for pneumonia patients, with the mean number of hospitalists seen during the stay at 2.05. For heart failure patients, the mean fragmentation level was 18.3 % and a mean of 1.78 hospitalists seen (Table 1).
| DRG 89 (n = 1950), Mean (STD) | DRG 127 (n = 9026), Mean (STD) | |
|---|---|---|
| ||
| Age, years | 65.8 (18.6) | 71.4 (15.2)* |
| % Female | 49.7 | 51.7 |
| Number of secondary diagnoses | 5.6 (2.5) | 5.0 (2.3)* |
| Severity of Illness | 2.80 (0.57) | 1.943 (0.72)* |
| Risk of Mortality | 2.49 (0.84) | 1.81 (0.75)* |
| Fragmentation: percent of care by non‐primary hospitalist(s) | 21.9% (20.3) | 18.3% (19.9)* |
| Number of physicians seen during hospital stay | 2.05 (1.12) | 1.78 (0.94)* |
Table 2 presents the results of the regression analysis on LOS. The association between fragmentation and LOS, adjusted for demographics, case mix and day of admission, was similar for PNA and HF patients. We found an increase in LOS of 0.39 days for each 10% increase in fragmentation level for pneumonia. Other variables that significantly increased LOS for PNA were larger ROM score, Sunday admission and more discharge diagnoses. Similarly, for heart failure patients, there was an increase in LOS of 0.30 days for each 10% increase in fragmentation level. Other variables associated with a significantly increase in LOS for PNA were larger ROM score, larger SOI category, more discharge diagnoses, and age. Figures 1 and 2 demonstrate the results graphically for LOS in each condition. They show the mean adjusted LOS by fragmentation level. Adjusted LOS was calculated for each patient based on the final model. For example, the average adjusted LOS for PNA patients with a fragmentation level of 20% to 30% is 8.06 days and 9.16 days for patients with a fragmentation level of 30% to 40%.
| Pneumonia (DRG = 89), n = 1950 | Heart Failure (DRG = 127), n = 9026 | |
|---|---|---|
| ||
| Fragmentation | Additional 0.39 day for each 10% increase in fragmentation | Additional 0.30 day for each 10% increase in fragmentation |
| Adjustment variables in the model | Age, number of secondary diagnoses, severity of illness variables, risk of mortality variables | Age, gender, number of secondary diagnoses. severity of illness variables, risk of mortality variables |


Discussion
This study demonstrated that there was a statistically significant association between FOC by hospitalist physicians and LOS for patients with DRG 89 (PNA) and DRG 127 (HF). As the percentage of fragmentation increased, the LOS increased significantly.
There are many ways in which to define FOC in the context of hospitalist care of inpatients. We chose to use an index similar to the Usual Provider of Care Index (UPC) which is a standard way to measure continuity of care in the primary care setting.9 The UPC measures the proportion of time spent with the primary provider. In the inpatient setting, we defined continuity of care as the proportion of visits by the primary hospitalist (the physician assuming the greatest number of days of the patient's care). The FOC Index is simply 1 minus the continuity of care. There are other potential measures of FOC, such as the number of handoffs or the number of physicians. We selected our measure based on its following strengths: (1) it does not have the simultaneity problem of measures such as the number of doctors or the number of handoffs that leads to endogeneitya correlation between the error term and the independent variable that biases the coefficient. Endogeneity can occur when the outcome variable and the independent variable are jointly determined, in this case, that an additional day in the hospital increases the likelihood of having an additional handoff as well as an additional handoff increasing the likelihood of an additional day; (2) fragmentation patterns that lead to handoffs on the weekend that then return to the primary hospitalist may be very different from fragmentation patterns that lead to new physicians at each transfer; (3) It provides a good comparison to other models of care where community physicians provide care to their own patients and have an effective FOC index of 0. Further research should be performed looking at different measures that may capture other aspects of care fragmentation.
This is one of the first studies to directly explore the relationship between FOC and LOS. Previous studies in the hospitalist literature have explored the impact of variables on LOS in which FOC may have been an indirect influence. For example, one study found a 13% shorter LOS among academic hospitalists at a teaching hospital who worked on a block rotation compared to a group of community hospitalists with a schedule involving more patient handoffs.10 Although there is little literature on FOC among hospitalists, the literature on medical residency programs is informative. The amount of discontinuity and the number of housestaff‐to‐housestaff transfers of responsibility has increased dramatically since the institution of more stringent work‐hour restrictions by the Accreditation Council for Graduate Medical Education (ACGME) in 2003.11 Horwitz et al.12 found that after the institution of work‐hour regulations, there was an 11% increase in number of transfers of care for a hypothetical patient admitted from Monday until Friday. They noted that programs with a night‐float system had a statistically significant increase in transitions of care compared to residencies without night float. Studies on the impact of the ACGME regulations on LOS have been mixed. One study found that LOS was reduced, and that there was improved adherence to quality indicators.4 A recent study found that there was a 44% increase in the median LOS when Short Call admitting teams were involved.2
There are several limitations to the findings of this study. The largest potential limitation is interpreting the direction of causality between FOC and LOS. This was a concurrent control study. Because the study was not randomized, there is a risk of confounding variables. Admission day of the week was 1 variable of concern for confounding due to the existence of a significant relationship between admission day LOS and between admission day and FOC. We included admission day in our modeling process. The coefficient estimates on FOC were stable, changing less than 1% when day of admission was added to the model. The risk of temporal trends was minimized by studying 1‐year's worth of data. Although we looked at variables that were potentially relevant in all hospitalized patients, every hospital and hospitalist practice has its own unique features that may impact both LOS and FOC. For example, for some hospitals, bed capacity may impact these measures. In many hospitals, hospitalist work force shortages may impact these measures by affecting the extent to which patients are cared for by full‐time hospitalists vs. locums tenens physicians. The large number of facilities in the study mitigates the influence of any individual hospital's LOS and FOC tendencies.
If increased FOC does result in prolonged LOS, the question arises as to how this fragmentation can be decreased. Although no physician can guarantee presence continually throughout a patient's inpatient stay, there are scheduling methods that reduce fragmentation and maximize the odds of a patient being followed by a single clinician. For example, the longer the block of days that a hospitalist is scheduled, the fewer hospitalists will need to care for a patient. Many of the top DRGs for patients cared for by hospitalists have lengths of stay between 4 and 5 days.13 Therefore, any schedule that has a hospitalist on for at least four days at a time will increase the likelihood that the same hospitalist will care for many of the patients throughout their stay. In making schedules, the goals of clinical efficiency and physician satisfaction must be weighed against the potential risks to the quality of patient care. For instance, the use of one hospitalist as the admitting physician for all patients may increase efficiency, but will also increase fragmentation.
A factor that may influence the impact of FOC on LOS is the quality of patient handoffs. The Joint Commission instituted this as a national patient safety goal in 2006.14 This goal was based on a Joint Commission analysis that 70% of sentinel events were caused by communication breakdowns, half of these occurring during handoffs.15
In conclusion, this study explored the relationship between FOC in patients cared for by hospitalists and LOS, using an FOC index. For every 10% increase in fragmentation, the LOS went up by 0.39 days for pneumonia and 0.30 days for heart failure. By adjusting for many variables that may impact LOS due to higher severity or complexity of illness, there is an increased likelihood that the FOC may have a causative relationship with the prolonged LOS.
There is a potential for discontinuity of care in the management of inpatients by hospitalists since many patients are cared for by more than one physician during their hospitalization. Previous studies have explored the impact of this type of discontinuity of care in residency programs. With the restrictions put in place by the Accreditation Council on Graduate Medical Education (ACGME) in 2003, there has been an increase in the fragmentation of care (FOC) that patients receive in the hospital. Studies have explored the impact of these changes on length of stay (LOS) and quality of care. The results have been mixed, with some studies showing that the increased FOC was associated with prolonged LOS,1, 2 as well as having a negative influence on quality.2, 3 Other studies have shown no change or a reduction in LOS,4 and an improvement in quality measures.46
There have been no prior studies on the impact of hospitalist care as the source of FOC on LOS. We therefore undertook a study to explore the impact of fragmented hospitalist care on LOS. Additionally, there has been some discussion on the impact of admission day of week on both FOC and LOS. Prior studies have mainly looked at day of the week in intensive care unit patients. One study found a modest increase in ICU LOS in patients admitted during the weekend.7 We therefore also looked at the impact of day of the week of admission on FOC in order to determine if fragmentation was just a proxy for admission day as the true indicator for increased LOS.
Methods
Design Overview
Data for this study consisted of clinical and billing information from IPC.
Setting and Participants
Data on LOS and FOC were drawn from the clinical and billing database of IPC The Hospitalist Company, which is a national group practice of hospitalists that at the time of the study had practices in over 230 acute health care facilities in 24 healthcare markets across the United States. Physicians enter clinical and billing data into IPC‐Link, a proprietary web based program.
Patients included in this study were discharged between December 1, 2006 and November 30, 2007 with a diagnosis‐related group (DRG) of 89 Pneumonia with complications or comorbidities (PNA) or a DRG of 127 Heart failure and shock (HF). A total of 10,977 patients with DRG 89 (n = 1951) or DRG 127 (n = 9026) were identified. These patients were admitted to 223 hospitals in 16 states.
Outcome
The outcome measure of interest was LOS.
FOC Measure
The independent variable of interest was the measure of fragmentation of physician care during the hospital stay. The intuitive meaning of fragmentation in physician hospital services is the delivery of care by multiple hospitalist physicians within a single stay. We converted the idea of fragmentation into a quantitative measure, an FOC Index, by calculating the percentage of hospitalist care delivered by physicians other than the primary hospitalist. The primary hospitalist was defined as the physician responsible for the largest number of visits during the hospital stay. Fragmentation was calculated by summing the number of daily billable encounters by IPC hospitalists other than the primary hospitalist and dividing by the total number of billable encounters by all IPC hospitalists. Thus, if three days out of a five‐day stay, the patient was seen by one hospitalist, then 60% of the care would be by the primary hospitalist. The patient was seen by other physicians than the primary hospitalist 2 out of the 5 days, for a FOC Index of 40%. IPC billing data was used to identify each hospitalist visit during the hospital stay by physician, date, and type of visit.
Covariates
The clinical database contained potential covariates: age, gender, day of admission, the number of ICD‐9 diagnosis codes on the discharge summary, and severity of illness (SOI) and risk of mortality (ROM) measures. The 3M DRG Grouper (St. Paul, Minnesota), using clinical data from the IPC‐Link discharge summary, assigned each patient an SOI score, and an ROM score based on age, primary diagnosis, and non‐operating room procedures.8 Both SOI and ROM are scored from 1 to 4, ranging from 1 for minor to 4 for extreme. We created dummy variables for the SOI and ROM categories, that is, variables for each score with a value of 1 if the SOI or ROM is that score and 0, otherwise. In each case, a score of 1 was the referent category. Dummy variables were created for each admission day of the week with Monday as the referent. Demographic variables and variables related to the patient condition were included to reduce any potential confounding arising from differences in patient populations across hospitals and hospitalist practice patterns. Admission day of the week has been shown to influence LOS and may influence FOC due to weekend scheduling within hospitalist practices.
Statistical Analysis
All analyses were performed using SAS (version 9.1). To reduce the influence of outliers on the model, observations with extreme values for LOS (defined as greater than 3 standard deviation [STD] from the mean) were truncated to 30 days. For pneumonia, the mean LOS was 7.85 days (STD 7.44) and all patients with LOS greater than 30 days were truncated to 30 days. For heart failure, the mean LOS was 5.88 days (STD 9.77) and all patients with LOS greater than 35 days were truncated to 35 days. Multivariable regression was performed using the negative binomial distribution. Negative binomial regression used as LOS is not normally distributed, but instead has a positive skew. Forward stepwise selection was performed separately for patients with each DRG using variables from the clinical database described in the Methods section. Categorical dummy variables were entered as a group. The final model contained all variables that were significant (P < 0.05).
Results
Table 1 reviews the demographics of patients with DRG 89 (PNA) and DRG 127 (HF). A large proportion of patients with both DRGs experienced no fragmentation in their physician care during hospitalization; 4152 patients with HF (46.3%) and 685 patients with PNA (35.8%) had visits from only 1 hospitalist throughout their hospital stay, for a FOC Index of 0%. The mean fragmentation level was 21.9% for pneumonia patients, with the mean number of hospitalists seen during the stay at 2.05. For heart failure patients, the mean fragmentation level was 18.3 % and a mean of 1.78 hospitalists seen (Table 1).
| DRG 89 (n = 1950), Mean (STD) | DRG 127 (n = 9026), Mean (STD) | |
|---|---|---|
| ||
| Age, years | 65.8 (18.6) | 71.4 (15.2)* |
| % Female | 49.7 | 51.7 |
| Number of secondary diagnoses | 5.6 (2.5) | 5.0 (2.3)* |
| Severity of Illness | 2.80 (0.57) | 1.943 (0.72)* |
| Risk of Mortality | 2.49 (0.84) | 1.81 (0.75)* |
| Fragmentation: percent of care by non‐primary hospitalist(s) | 21.9% (20.3) | 18.3% (19.9)* |
| Number of physicians seen during hospital stay | 2.05 (1.12) | 1.78 (0.94)* |
Table 2 presents the results of the regression analysis on LOS. The association between fragmentation and LOS, adjusted for demographics, case mix and day of admission, was similar for PNA and HF patients. We found an increase in LOS of 0.39 days for each 10% increase in fragmentation level for pneumonia. Other variables that significantly increased LOS for PNA were larger ROM score, Sunday admission and more discharge diagnoses. Similarly, for heart failure patients, there was an increase in LOS of 0.30 days for each 10% increase in fragmentation level. Other variables associated with a significantly increase in LOS for PNA were larger ROM score, larger SOI category, more discharge diagnoses, and age. Figures 1 and 2 demonstrate the results graphically for LOS in each condition. They show the mean adjusted LOS by fragmentation level. Adjusted LOS was calculated for each patient based on the final model. For example, the average adjusted LOS for PNA patients with a fragmentation level of 20% to 30% is 8.06 days and 9.16 days for patients with a fragmentation level of 30% to 40%.
| Pneumonia (DRG = 89), n = 1950 | Heart Failure (DRG = 127), n = 9026 | |
|---|---|---|
| ||
| Fragmentation | Additional 0.39 day for each 10% increase in fragmentation | Additional 0.30 day for each 10% increase in fragmentation |
| Adjustment variables in the model | Age, number of secondary diagnoses, severity of illness variables, risk of mortality variables | Age, gender, number of secondary diagnoses. severity of illness variables, risk of mortality variables |


Discussion
This study demonstrated that there was a statistically significant association between FOC by hospitalist physicians and LOS for patients with DRG 89 (PNA) and DRG 127 (HF). As the percentage of fragmentation increased, the LOS increased significantly.
There are many ways in which to define FOC in the context of hospitalist care of inpatients. We chose to use an index similar to the Usual Provider of Care Index (UPC) which is a standard way to measure continuity of care in the primary care setting.9 The UPC measures the proportion of time spent with the primary provider. In the inpatient setting, we defined continuity of care as the proportion of visits by the primary hospitalist (the physician assuming the greatest number of days of the patient's care). The FOC Index is simply 1 minus the continuity of care. There are other potential measures of FOC, such as the number of handoffs or the number of physicians. We selected our measure based on its following strengths: (1) it does not have the simultaneity problem of measures such as the number of doctors or the number of handoffs that leads to endogeneitya correlation between the error term and the independent variable that biases the coefficient. Endogeneity can occur when the outcome variable and the independent variable are jointly determined, in this case, that an additional day in the hospital increases the likelihood of having an additional handoff as well as an additional handoff increasing the likelihood of an additional day; (2) fragmentation patterns that lead to handoffs on the weekend that then return to the primary hospitalist may be very different from fragmentation patterns that lead to new physicians at each transfer; (3) It provides a good comparison to other models of care where community physicians provide care to their own patients and have an effective FOC index of 0. Further research should be performed looking at different measures that may capture other aspects of care fragmentation.
This is one of the first studies to directly explore the relationship between FOC and LOS. Previous studies in the hospitalist literature have explored the impact of variables on LOS in which FOC may have been an indirect influence. For example, one study found a 13% shorter LOS among academic hospitalists at a teaching hospital who worked on a block rotation compared to a group of community hospitalists with a schedule involving more patient handoffs.10 Although there is little literature on FOC among hospitalists, the literature on medical residency programs is informative. The amount of discontinuity and the number of housestaff‐to‐housestaff transfers of responsibility has increased dramatically since the institution of more stringent work‐hour restrictions by the Accreditation Council for Graduate Medical Education (ACGME) in 2003.11 Horwitz et al.12 found that after the institution of work‐hour regulations, there was an 11% increase in number of transfers of care for a hypothetical patient admitted from Monday until Friday. They noted that programs with a night‐float system had a statistically significant increase in transitions of care compared to residencies without night float. Studies on the impact of the ACGME regulations on LOS have been mixed. One study found that LOS was reduced, and that there was improved adherence to quality indicators.4 A recent study found that there was a 44% increase in the median LOS when Short Call admitting teams were involved.2
There are several limitations to the findings of this study. The largest potential limitation is interpreting the direction of causality between FOC and LOS. This was a concurrent control study. Because the study was not randomized, there is a risk of confounding variables. Admission day of the week was 1 variable of concern for confounding due to the existence of a significant relationship between admission day LOS and between admission day and FOC. We included admission day in our modeling process. The coefficient estimates on FOC were stable, changing less than 1% when day of admission was added to the model. The risk of temporal trends was minimized by studying 1‐year's worth of data. Although we looked at variables that were potentially relevant in all hospitalized patients, every hospital and hospitalist practice has its own unique features that may impact both LOS and FOC. For example, for some hospitals, bed capacity may impact these measures. In many hospitals, hospitalist work force shortages may impact these measures by affecting the extent to which patients are cared for by full‐time hospitalists vs. locums tenens physicians. The large number of facilities in the study mitigates the influence of any individual hospital's LOS and FOC tendencies.
If increased FOC does result in prolonged LOS, the question arises as to how this fragmentation can be decreased. Although no physician can guarantee presence continually throughout a patient's inpatient stay, there are scheduling methods that reduce fragmentation and maximize the odds of a patient being followed by a single clinician. For example, the longer the block of days that a hospitalist is scheduled, the fewer hospitalists will need to care for a patient. Many of the top DRGs for patients cared for by hospitalists have lengths of stay between 4 and 5 days.13 Therefore, any schedule that has a hospitalist on for at least four days at a time will increase the likelihood that the same hospitalist will care for many of the patients throughout their stay. In making schedules, the goals of clinical efficiency and physician satisfaction must be weighed against the potential risks to the quality of patient care. For instance, the use of one hospitalist as the admitting physician for all patients may increase efficiency, but will also increase fragmentation.
A factor that may influence the impact of FOC on LOS is the quality of patient handoffs. The Joint Commission instituted this as a national patient safety goal in 2006.14 This goal was based on a Joint Commission analysis that 70% of sentinel events were caused by communication breakdowns, half of these occurring during handoffs.15
In conclusion, this study explored the relationship between FOC in patients cared for by hospitalists and LOS, using an FOC index. For every 10% increase in fragmentation, the LOS went up by 0.39 days for pneumonia and 0.30 days for heart failure. By adjusting for many variables that may impact LOS due to higher severity or complexity of illness, there is an increased likelihood that the FOC may have a causative relationship with the prolonged LOS.
Copyright © 2010 Society of Hospital Medicine
Care Model for ED Boarders
Emergency Department (ED) overcrowding has become an important problem in North American hospitals.13 A national survey identified the prolonged length of stay of admitted patients in the ED as the most frequent reason for overcrowding.4 This complex problem occurs when hospital inpatient census increases and prevents admitted patients from being assigned and transported to hospital beds in a timely manner.5 The practice of holding admitted patients in the ED, known as boarding, is typically defined as the length of stay (LOS) in ED beginning 2 hours after the time of admission to the time of transfer to the wards.
In a study of daily mean ED LOS, Rathlev et al.6 concluded that a 5% increase in hospital occupancy resulted in 14 hours of holding time for all patients in the ED, and an observational study found that when hospital occupancy exceeds a threshold of 90%, the ED LOS for admitted patients correspondingly increased.7 Thus, efforts to decrease overcrowding will need to address both ED and hospital throughput and LOS. Most importantly, overcrowding has important consequences on physician and patient satisfaction and the quality of patient care.811
Between 1995 and 2005, ED visits rose 20% from 96.5 million to 115.3 million visits annually, while the number of hospital EDs decreased from 4176 to 3795, making an overall 7% increase in ED utilization rate.12 Similarly, there was a 12% increase in the total inpatient admissions for all registered hospitals in the United States from 31 million in 1995 to 35.3 million in 2005.13 However, despite this increase in demand of ED utilization and inpatient admissions, there had been a steady decline in the supply of hospital beds, from 874,000 in 1995, to 805,000 in 2006.13 These factors have exacerbated the problem of ED overcrowding and boarding.
Not only does boarding entail additional consumption of space, resources, equipment, and manpower, it also potentially compromises patient safety. Typically, hospitalists and inpatient medical teams are engaged in providing care to patients in the wards, while ED physicians and nurses are busy taking care of newly‐arrived ED patients. Non‐ED physicians may have the false impression that their boarded patients, while in the ED, are receiving continuous care and so may decide to delay seeing these patients, which can jeopardize the quality and timeliness of care. Studies have shown that ED overcrowding may potentially lead to poor patient care and outcomes and increased risk for medical errors.1416 ED overcrowding potentially causes multiple effects, including prolonging patient pain and suffering, long patient waiting time, patient dissatisfaction, ambulance diversions, decreased physician productivity, and increased frustration among medical staff.15 In a report by the Joint Commission Accreditation of Healthcare Organizations,17 ED overcrowding was cited as a significant contributing factor in sentinel event cases of patient death or permanent injury due to delays in treatment. Boarding critically ill patients who are physiologically vulnerable and unstable can allow them to be subjected to treatment delays at a pivotal point when time‐sensitive interventions are necessary, ie, sepsis or cardiogenic shockthe golden hour in trauma.16 Medical errors are usually not caused by individual errors but by complex hospital systems; and ED overcrowding is a prime example of a system problem that creates a high‐risk environment for medical errors and threatens patient safety.18
Our hospital commonly has 5 to 15 boarders and often has 20 to 30 boarders at any time. Approximately 90% of these patients are admitted to the Medical Service. In response to this challenge, our institution has designated a full‐time hospitalist to manage boarded patients. The primary goal of this new role is to ensure patient safety and the delivery of high‐quality care while admitted patients are in the ED (Table 1).
| |
| 1 | Round on all patients admitted to the Department of Medicine located in the ED, including those on the Teaching and Nonteaching Services. Rounds focus on patient safety, such as ensuring vital home and hospital medications are administered and changes in stability are noted. All patient updates are documented in the ED electronic medical records (IBEX). |
| 2 | Identify admitted patients who may be downgraded from telemetry to nontelemetry status. Telemetry and cardiac beds are in high demand, and decreasing utilization facilitates obtaining the appropriate ward bed for ED patients. |
| 3 | Assess admitted patients for possible discharge. The patient's condition may have improved or results may indicate that admission is no longer required. The ED hospitalist communicates with the ED physician and wards teams, facilitates management, implements the discharge, and ensures adequate follow‐up. |
| 4 | Refer patients to an ED social worker as needed. |
| 5 | Facilitate referrals to other medical or surgical specialties if indicated. |
| 6 | Clarify the plan of care with the ED staff and facilitates ED communication with the ward team. Acts as a liaison and a resource for the ED physicians and nursing staff. |
| 7 | Supervise the triage duties of the medical admitting resident. |
| 8 | Provide medical consultation to ED physicians for patients not being admitted to the hospital or who are being admitted to other services (eg, surgery). |
The objectives of the study were to determine: (1) the impact on quality of care by assessing laboratory results acted upon and medication follow‐up by the ED hospitalist, and (2) the impact on throughput by assessing the number of ED discharges and telemetry downgrades.
Methods
Setting
The Mount Sinai Medical Center is a tertiary‐care 1121‐bed acute care teaching hospital located in New York City. The hospital borders East Harlem and the Upper East Side of Manhattan. The Medical Service is composed of a Teaching Service, composed of house staff and attendings, and a non‐Teaching Service, composed of nurse practitioners, physician assistants, and attendings. Hospitalists and private attendings may have patients on either the Teaching or the non‐Teaching Service. In 2007, there were 56,541 patients admitted for a total of 332,368 days. The mean LOS for medical inpatients was 5.89 days. The total ED visit was 79,500 with a total inpatient and critical care admissions of 24,522. The mean and median LOS for all ED patients were 623 minutes and 493 minutes, respectively. There were 11,488 patients who qualified as boarders, averaging 31.5 boarders per 24 hours; with a mean and median LOS per boarder of 288 minutes and 198 minutes, respectively. The ED LOS for admitted patients ranged from 2 minutes to 4074 minutes (2.83 days).
Admission Process
Once an ED attending physician decides that a patient is to be admitted, the patient is placed on a computerized list in the ED's electronic medical record (IBEX software). The Medical Admitting Resident (MAR) evaluates and triages admitted patients, and assigns and gives a verbal report to the appropriate Medicine Service (ie, Teaching, non‐Teaching, cardiac telemetry unit, intensive care, etc.). The Admitting Office searches for and assigns the appropriate unit and bed for the patient. A hospitalist or resident physician performs the patient's initial assessment and evaluation in the ED, and admission orders are placed in the inpatient computerized order entry system (TDS). When the bed is ready, the ED nurse gives a verbal report to the floor nurse, and the patient is transported to the ward.
Responsibilities
The specific responsibilities of the ED hospitalist are listed in Table 1. The primary role is to round on patients admitted to the Medicine Service who are located in the ED. This encompasses a wide array of patients and services, including patients assigned to a hospitalist service attending or who have a private attending, patients admitted to the Teaching or non‐Teaching Service, patients admitted to the intensive care unit, and patients admitted to a general medicine or specialty service (eg, telemetry, oncology, human immunodeficiency virus [HIV]). Rounding includes review of the ED's electronic medical record as well as direct examination of patients. The hospitalist focuses on patients with longer ED LOS and on aspects of care that may lapse while patients remain in the ED for prolonged periods. At our institution, the follow‐up of subsequent tests, laboratory values, and medications for ED boarders is the responsibility of the primary inpatient team, though the ED physicians act on urgent and critical results and continue to deliver all emergency care. Through rounding, the ED hospitalist is able to identify abnormal results in a timely manner, alert the ED physician and primary inpatient team, and address abnormalities. Specific examples of laboratory results acted upon include hypokalemia, hyperglycemia, and elevated cardiac enzymes. The ED hospitalist is also able to determine whether any outpatient medications have not yet been administered (eg, antihypertensives, immune suppressants) and ensure that subsequent doses of medications initiated in the ED (eg, antibiotics) are administered during the appropriate timeframe.
Communication is emphasized, as contact with ED physicians, ward physicians, and often the outpatient primary care physician is required when any change in management is considered. The ED hospitalist also provides the capability of rapid response to changes in patient status (eg, a new complaint or fever). In addition, the hospitalist is available to consult on medical patients who may not require admission and on nonmedical patients for whom an internal medicine consult may be beneficial (eg, preoperative optimization of a surgical patient). The ED hospitalist documents the evaluation in the IBEX system. Bills were submitted for visits in which patients were discharged as these encounters are comprehensive, but not for other encounters.
Data Collection
The ED hospitalist role began March 10, 2008 and is a 10‐hour shift (8 AM to 6 PM) on weekdays. The study period was from March 10, 2008 through June 30, 2008. The study was approved by the hospital's institutional review board.
Data were collected on aspects of care that could have been impacted by the ED hospitalist, including medication and laboratory orders, ED discharges, ED admissions avoided, and telemetry downgrades. Discharges from ED refers to boarded admitted patients in the ED, who by the judgment of the ED hospitalist were ready for discharge. Admissions avoided refers to patients who the ED physician planned to admit but had not yet been admitted, and whose admission was avoided through the recommendations made by the ED hospitalist. The ED LOS was defined as the duration of time from when the patient was admitted to the Medicine Service to the time the patient was transferred to a medical ward. Telemetry downgrades were defined as patients assigned to the cardiac telemetry unit who the hospitalist determined required only telemetry on a general medical unit or did not require telemetry, or patients assigned to telemetry on a general medicine unit who the hospitalist determined no longer required telemetry.
Results were expressed as percentages of patients admitted to a Medicine Service and percentage of patients evaluated by the ED hospitalist, as indicated. 95% confidence intervals (CI) were calculated.
Results
During the study period, there were 4363 patients admitted to the Medicine Service and 3555 patients who qualified as boarders (mean of 29 boarders per 24 hours). The mean boarding time of admitted patients was 440 minutes. A total of 634 patients (17.8% of all boarded patients) were evaluated by the ED hospitalist. The mean daily number of patients seen by the ED hospitalist was 8.0.
The key elements of the delivery of care by the ED hospitalist are summarized in Table 2.
| Elements | Boarders (n = 3555) [n (%)] | Patients Intervened on (n = 634) [n (%)] |
|---|---|---|
| ||
| Laboratory results acted upon | 472 (13.2) | 472 (74.5) |
| Medication follow‐up | 506 (14.2) | 506 (79.8) |
| Discharges from the ED* | 46 (1.3) | 46 (7.3) |
| Admissions avoided | 6 (0.2) | 6 (0.95) |
| Telemetry downgrades | 61 (1.8) | 61 (9.6) |
The care of boarded patients included follow‐up of laboratory tests for 74.5% (95% CI, 71‐78%) and medication orders for 79.8% (95% CI, 77‐83%) of patients. A total of 46 patients were discharged by the ED hospitalist (0.6 discharges/day) and telemetry was discontinued for 61 patients (0.8 downgrades/day). The discharge rate was 7.3% (95% CI, 5‐10%) and telemetry downgrade rate was 9.6% (95% CI, 8‐12%) of those patients assessed by the ED hospitalist. Expressed as a percentage of the total ED boarders (n = 3555), the combined discharge rate and the admissions avoided rate was 1.5%.
Table 3 shows the discharge diagnoses made from the ED. Chest pain was the most common diagnosis, followed by syncope, pneumonia, and chronic obstructive pulmonary disease (COPD).
| Diagnoses | Patients (n = 46) [n (%)] |
|---|---|
| |
| Chest pain | 12 (26) |
| Syncope/dizziness | 7 (15) |
| Pneumonia | 4 (9) |
| COPD | 4 (9) |
| Congestive heart failure | 3 (7) |
| Gastroenteritis | 3 (7) |
| Dermatitis/rash | 3 (7) |
| Alcohol abuse | 3 (7) |
| Abdominal pain | 3 (7) |
| End stage renal disease | 2 (4) |
| Vaginal bleeding | 1 (2) |
| Fall | 1 (2) |
| Asthma | 1 (2) |
Discussion
Our hospital has successfully implemented an innovative strategy utilizing a hospitalist to help provide seamless care to medical patients located in the ED. Other solutions at our hospital had previously been implemented, but had not adequately addressed the problem, including: (1) protocols to monitor length of stay patterns and deviations, (2) discharge planning activities, (3) organized computerized bed tracking, (4) improvement in the timeliness of ancillary services, (5) daily bed briefing among nurse managers, and (6) 24‐hour presence of a MAR to facilitate triage in the ED.
The current study demonstrates the potential for substantial impact on patient care. The substantial number of the assessed boarder patients for whom laboratory tests (74.5%) and medications (79.8%) were ordered by the ED hospitalists suggests that the quality and timeliness of care was enhanced by this initiative. In addition, the considerable number of patients discharged from the ED and downgraded from telemetry (1.5% and 1.8% of all boarder patients, respectively) suggests that an ED hospitalist may have a meaningful impact on bed utilization and thus decrease ED overcrowding. In 2007, there were 11,488 who qualified as boarders; our data suggest that an ED hospitalist would result in approximately 172 boarders not being admitted annually.
Though the ED LOS was higher during the study period compared to 2007, it was lower than the 2 months immediately preceding implementation of the ED hospitalist role. The ED LOS was 732 and 658 minutes for January and February 2008, respectively, which was markedly increased from 2007 (288 minutes), and prompted development of the ED hospitalist role. The ED LOS during the study period subsequently decreased to 440 minutes. Though the wide fluctuations in ED LOS and the short time period with high ED LOS prior to implementation preclude concluding that the ED hospitalist role decreased ED LOS, the data suggest that an ED hospitalist may be able to improve ED throughput.
The majority of the discharges made by the ED hospitalist are patients who had been admitted for chest pain, had improved, and had negative cardiac enzymes and stress tests. Patients with syncope who were discharged were likely patients without any comorbidities. The COPD and pneumonia admissions were likely patients who improved after aggressive treatment in the ED.
The impact of ED overcrowding on the quality of patient care and outcomes may be substantial. Hwang et al.19 found a direct correlation between ED census and time to pain assessment and administration of analgesic medication. A study at an academic medical center found that higher ED volume was associated with less likelihood of antibiotics being administered within 4 hours for patients with community‐acquired pneumonia.20 A comprehensive review of the literature identified 41 studies examining the effects of ED overcrowding on clinical outcomes and the investigators noted that ED overcrowding was associated with increased in‐hospital mortality.8
Causes of poor outcomes during periods of overcrowding may be the high volume of acute patients preventing adequate time and attention for each ED patient, as well as confusion during the transition from ED to ward physicians. For example, a patient may receive their initial dose of antibiotics from the ED physician, but subsequent doses may be overlooked in the transition of care from the ED physician to the inpatient team. In addition, having admitted patients located in the ED for extended periods of time may lead to these patients not being seen as frequently as patients admitted to the inpatient wards. Another potential consequence of prolonged ED stay for admitted patients is delay in inpatient management. Tests done in the ED may prompt further studies that may not be ordered promptly while patients remain in the ED, which subsequently increases LOS. Other potential issues may be an increase in confusion among geriatric patients in a noisy and crowded ED; decreased access to specialized nursing care that may be available on a hospital ward; decreased access to physical therapy and occupational therapy services; and decreased comfort and satisfaction as patients wait in overcrowded EDs for prolonged periods.
Several other potential innovative solutions to ED overcrowding have been proposed, studied, and tested. These measures generally are focused on improving the three interdependent components of ED workflow: INPUT THROUGHPUT OUTPUT.21, 22 However, process redesign and intervention on these 3 interdependent ED workflow components may be difficult to achieve, especially when hospital resources are limited and when inpatient hospital capacity is already maximized. In some institutions, efforts have been reported to successfully streamline the transfer of admitted ED patients to inpatient beds, through transfer‐to‐ward policy interventions (eg, physician coordinators for patient flow and bed management or transfers made within a defined period of time).2326 However, in a study by Quinn et al.,27 implementation of a rapid admission policy resulted in a decrease of only 10.1 minutes in the ED LOS. Several studies have demonstrated the benefits of an acute medical admissions unit in alleviating ED overcrowding.28, 29 Other unconventional solutions by some hospitals include sending admitted patients to the unit's hallways or placing discharged patients in the hallway while waiting for transportation so that the ED bed will be readily available.30
The ED hospitalist is well‐situated to have an impact on several key hospital outcomes. As the ED hospitalist role was shown to affect processes that relate to ED throughput, it is possible that the role will improve ED overcrowding and decrease ED LOS. Specifically, identifying patients who can be discharged and for whom telemetry is no longer indicated decreases unnecessary bed utilization and allows these beds to be available for other ED patients. This initiative also may promote patient satisfaction by assuring patients that their medical and concerns are being fully addressed while they are in the ED. Increased emphasis on hospital reporting will make patient satisfaction a priority for many hospitals, and the ED hospitalist will be in a unique position to meet and greet patients admitted to the Medicine Service and to reassure them that the medical team is present and addressing their concerns. The hospitalist's ability to facilitate diagnostic testing and treatment while patients remain in the ED may also help decrease the total LOS in the hospital. In addition, the ED hospitalist is also in position to recognize social factors at the earliest stage of admission so that they can be immediately addressed. Future studies will need to be done to determine if this model of transitional care impacts these important factors.
Our study has several important limitations. Most notably, the lack of a comparison interval for which a hospitalist was not assigned to this role prevents us from drawing any definitive conclusions on the benefits of the ED hospitalist model. Also, we collected only summary data and do not have demographic data on the patients managed by the ED hospitalist or information on the ED course of patients who were discharged or had telemetry downgraded. This prevents determination of whether discharged patients did not require admission initially or whose condition evolved over a prolonged ED stay. In addition, other key outcomes, such as patient satisfaction and satisfaction of the ED physicians and nursing staff have not yet been formally measured. Future studies will be needed to determine if an ED hospital model can improve important process and clinical outcomes.
The greatest challenge of this initiative was introducing and familiarizing this role to the key stakeholders, including the ED physicians and nursing staff, house staff, and private practice physicians. Though we did not perform structured surveys on satisfaction, through informal discussions we noted that the role was welcomed with enthusiasm by the ED physicians. Notably, several ED physicians expressed appreciation that they were able to focus their care on new ED patients rather than on the boarded ED patients. Through feedback, we noted soon after implementation that ED faculty and nurses needed further clarification about the potential overlapping roles of the ED hospitalist and ED physicians and ward physicians. These concerns were addressed by educational sessions and announcements, including presentations at ED faculty and staff meetings. The hospitalist assigned to the role each month received individualized orientation prior to assuming the role, and an ED Hospitalist Manual was distributed. Possibly due to these focused sessions, the hospitalists assigned to the role became quickly acclimated.
Conclusions
We have found that designating a hospitalist to directly address the care of ED boarders can enhance the quality and timeliness of care and decrease bed and telemetry utilization with the potential to impact ED and hospital LOS. Given the success of the pilot model, the role was expanded at our institution to 10 hours per day, 7 days per week. Hospitals struggling to address the needs of their admitted patients in the ED should consider incorporating an ED hospitalist to enhance clinical care and address issues relating to throughput. A follow‐up study is needed to more precisely describe the impact of the ED hospitalist model.
- , , , et al.Estimating the degree of emergency department overcrowding in academic medical centers: results of the National ED Overcrowding Study (NEDOCS).Acad Emerg Med.2004;11:38–50.
- , , , et al.Frequency, determinants, and impact of overcrowding in emergency departments in Canada: a national survey.Healthc Q.2007;10:32–40.
- , .EMDOC (emergency department overcrowding) internet‐based safety net research.Admin Emerg Med.2008;35:101–107.
- United States General Accounting Office.Hospital Emergency Departments: Crowded Conditions Vary Among Hospitals and Communities. March 2003.Washington, DC:General Accounting Office;2003.
- .Overcrowding in emergency department: increased demand and decreased capacity.Ann Emerg Med.2002;39:430–432.
- , , , et al.Time series analysis of variables associated with daily mean emergency department length of stay.Ann Emerg Med.2007;49:265–271.
- , , , et al.Effect of hospital occupancy on emergency department length of stay and patient disposition.Ann Emerg Med.2003;10:127–133.
- , , , et al.The effect of emergency department crowding on clinically oriented outcomes.Acad Emerg Med.2009;16:1–10.
- , .Emergency department overcrowding: the impact of resource scarcity on physician job satisfaction.J Health Manag.2005;50:327–340.
- , , , , , .The effect of emergency department crowding on patient satisfaction for admitted patients.Acad Emerg Med.2008;15:825–831.
- , .The effect of crowding on access and quality in an academic ED.Am J Emerg Med.2006;24:787–794.
- , , .National Hospital Ambulatory Medical Care Survey: 2005 Emergency Department Summary. Advance Data from Vital and Health Statistics. No. 386.Hyattsville, MD:National Center for Health Statistics;2007.
- American Hospital Association (AHA).Table 1: Historical trends in utilization, personnel, and finances: year 1946–2006.AHA Hospital Statistics.2008 ed.Chicago:Health Forum LLC;2008:3.
- , .Emergency department overcrowding in the US: an emerging threat to patient safety and public health.Emerg Med J.2003;20:402–405.
- , .Overcrowding in the nation's emergency departments: complex causes and disturbing effects.Ann Emerg Med.2000;35:63–68.
- , .Clinical review: emergency department overcrowding and the potential impact on the critically ill.Crit Care.2005;9:291–295.
- Joint Commission on Accreditation of Healthcare Organizations (JCAHO): Sentinel event alert 2002, Issue 26. Available at: http://www.jointcommission.org/SentinelEvents/SentinelEventAlert/sea_26.htm. Accessed October2009.
- , , , et al.Safety net research in emergency medicine: proceedings of the Academic Emergency Consensus Conference on “The Unraveling Safety Net.”Acad Emerg Med.2001;8:1024–1029.
- , , , , , .Emergency department crowding and decreased quality of pain care.Acad Emerg Med.2008;15:1248–1256.
- , , , .Effect of emergency department crowding on time to antibiotics in patients admitted with community‐acquired pneumonia.Ann Emerg Med.2007;50:501–509.
- , , , et al.A conceptual model of emergency department crowding.Ann Emerg Med.2003;42:173–180.
- , , , et al.Emergency department crowding: consensus development of potential measures.Ann Emerg Med.2003;42:824–834.
- , , , et al.Intervention to decrease emergency department crowding: does it have an effect on return visits and hospital readmission?Ann Emerg Med.2003;41:173–185.
- , , , et al.Rapid process design in a university‐based emergency department: decreasing waiting time intervals and improving patient satisfaction.Ann Emerg Med.2002;39:168–177.
- .Emergency department crowding: an action plan.Acad Emerg Med.2001;18:185–187.
- , , .Hospitalists and an innovative emergency department admission process.J Gen Intern Med.2004;19:266–268.
- , , et al.Effects of implementing a rapid admission policy in the ED.Am J Emerg Med.2007;25:559–563.
- , , .Effect of an emergency department managed acute care unit on ED overcrowding and emergency medical services diversion.Acad Emerg Med.2001;8:1085–1100.
- , , , .Emergency department census of patients awaiting admission following reorganization of an admissions process.Emerg Med J.2006;23:363–367.
- .Emergency department flow and the boarded patient: how to get admitted patients upstairs.Ann Emerg Med.2007;49:68–70.
Emergency Department (ED) overcrowding has become an important problem in North American hospitals.13 A national survey identified the prolonged length of stay of admitted patients in the ED as the most frequent reason for overcrowding.4 This complex problem occurs when hospital inpatient census increases and prevents admitted patients from being assigned and transported to hospital beds in a timely manner.5 The practice of holding admitted patients in the ED, known as boarding, is typically defined as the length of stay (LOS) in ED beginning 2 hours after the time of admission to the time of transfer to the wards.
In a study of daily mean ED LOS, Rathlev et al.6 concluded that a 5% increase in hospital occupancy resulted in 14 hours of holding time for all patients in the ED, and an observational study found that when hospital occupancy exceeds a threshold of 90%, the ED LOS for admitted patients correspondingly increased.7 Thus, efforts to decrease overcrowding will need to address both ED and hospital throughput and LOS. Most importantly, overcrowding has important consequences on physician and patient satisfaction and the quality of patient care.811
Between 1995 and 2005, ED visits rose 20% from 96.5 million to 115.3 million visits annually, while the number of hospital EDs decreased from 4176 to 3795, making an overall 7% increase in ED utilization rate.12 Similarly, there was a 12% increase in the total inpatient admissions for all registered hospitals in the United States from 31 million in 1995 to 35.3 million in 2005.13 However, despite this increase in demand of ED utilization and inpatient admissions, there had been a steady decline in the supply of hospital beds, from 874,000 in 1995, to 805,000 in 2006.13 These factors have exacerbated the problem of ED overcrowding and boarding.
Not only does boarding entail additional consumption of space, resources, equipment, and manpower, it also potentially compromises patient safety. Typically, hospitalists and inpatient medical teams are engaged in providing care to patients in the wards, while ED physicians and nurses are busy taking care of newly‐arrived ED patients. Non‐ED physicians may have the false impression that their boarded patients, while in the ED, are receiving continuous care and so may decide to delay seeing these patients, which can jeopardize the quality and timeliness of care. Studies have shown that ED overcrowding may potentially lead to poor patient care and outcomes and increased risk for medical errors.1416 ED overcrowding potentially causes multiple effects, including prolonging patient pain and suffering, long patient waiting time, patient dissatisfaction, ambulance diversions, decreased physician productivity, and increased frustration among medical staff.15 In a report by the Joint Commission Accreditation of Healthcare Organizations,17 ED overcrowding was cited as a significant contributing factor in sentinel event cases of patient death or permanent injury due to delays in treatment. Boarding critically ill patients who are physiologically vulnerable and unstable can allow them to be subjected to treatment delays at a pivotal point when time‐sensitive interventions are necessary, ie, sepsis or cardiogenic shockthe golden hour in trauma.16 Medical errors are usually not caused by individual errors but by complex hospital systems; and ED overcrowding is a prime example of a system problem that creates a high‐risk environment for medical errors and threatens patient safety.18
Our hospital commonly has 5 to 15 boarders and often has 20 to 30 boarders at any time. Approximately 90% of these patients are admitted to the Medical Service. In response to this challenge, our institution has designated a full‐time hospitalist to manage boarded patients. The primary goal of this new role is to ensure patient safety and the delivery of high‐quality care while admitted patients are in the ED (Table 1).
| |
| 1 | Round on all patients admitted to the Department of Medicine located in the ED, including those on the Teaching and Nonteaching Services. Rounds focus on patient safety, such as ensuring vital home and hospital medications are administered and changes in stability are noted. All patient updates are documented in the ED electronic medical records (IBEX). |
| 2 | Identify admitted patients who may be downgraded from telemetry to nontelemetry status. Telemetry and cardiac beds are in high demand, and decreasing utilization facilitates obtaining the appropriate ward bed for ED patients. |
| 3 | Assess admitted patients for possible discharge. The patient's condition may have improved or results may indicate that admission is no longer required. The ED hospitalist communicates with the ED physician and wards teams, facilitates management, implements the discharge, and ensures adequate follow‐up. |
| 4 | Refer patients to an ED social worker as needed. |
| 5 | Facilitate referrals to other medical or surgical specialties if indicated. |
| 6 | Clarify the plan of care with the ED staff and facilitates ED communication with the ward team. Acts as a liaison and a resource for the ED physicians and nursing staff. |
| 7 | Supervise the triage duties of the medical admitting resident. |
| 8 | Provide medical consultation to ED physicians for patients not being admitted to the hospital or who are being admitted to other services (eg, surgery). |
The objectives of the study were to determine: (1) the impact on quality of care by assessing laboratory results acted upon and medication follow‐up by the ED hospitalist, and (2) the impact on throughput by assessing the number of ED discharges and telemetry downgrades.
Methods
Setting
The Mount Sinai Medical Center is a tertiary‐care 1121‐bed acute care teaching hospital located in New York City. The hospital borders East Harlem and the Upper East Side of Manhattan. The Medical Service is composed of a Teaching Service, composed of house staff and attendings, and a non‐Teaching Service, composed of nurse practitioners, physician assistants, and attendings. Hospitalists and private attendings may have patients on either the Teaching or the non‐Teaching Service. In 2007, there were 56,541 patients admitted for a total of 332,368 days. The mean LOS for medical inpatients was 5.89 days. The total ED visit was 79,500 with a total inpatient and critical care admissions of 24,522. The mean and median LOS for all ED patients were 623 minutes and 493 minutes, respectively. There were 11,488 patients who qualified as boarders, averaging 31.5 boarders per 24 hours; with a mean and median LOS per boarder of 288 minutes and 198 minutes, respectively. The ED LOS for admitted patients ranged from 2 minutes to 4074 minutes (2.83 days).
Admission Process
Once an ED attending physician decides that a patient is to be admitted, the patient is placed on a computerized list in the ED's electronic medical record (IBEX software). The Medical Admitting Resident (MAR) evaluates and triages admitted patients, and assigns and gives a verbal report to the appropriate Medicine Service (ie, Teaching, non‐Teaching, cardiac telemetry unit, intensive care, etc.). The Admitting Office searches for and assigns the appropriate unit and bed for the patient. A hospitalist or resident physician performs the patient's initial assessment and evaluation in the ED, and admission orders are placed in the inpatient computerized order entry system (TDS). When the bed is ready, the ED nurse gives a verbal report to the floor nurse, and the patient is transported to the ward.
Responsibilities
The specific responsibilities of the ED hospitalist are listed in Table 1. The primary role is to round on patients admitted to the Medicine Service who are located in the ED. This encompasses a wide array of patients and services, including patients assigned to a hospitalist service attending or who have a private attending, patients admitted to the Teaching or non‐Teaching Service, patients admitted to the intensive care unit, and patients admitted to a general medicine or specialty service (eg, telemetry, oncology, human immunodeficiency virus [HIV]). Rounding includes review of the ED's electronic medical record as well as direct examination of patients. The hospitalist focuses on patients with longer ED LOS and on aspects of care that may lapse while patients remain in the ED for prolonged periods. At our institution, the follow‐up of subsequent tests, laboratory values, and medications for ED boarders is the responsibility of the primary inpatient team, though the ED physicians act on urgent and critical results and continue to deliver all emergency care. Through rounding, the ED hospitalist is able to identify abnormal results in a timely manner, alert the ED physician and primary inpatient team, and address abnormalities. Specific examples of laboratory results acted upon include hypokalemia, hyperglycemia, and elevated cardiac enzymes. The ED hospitalist is also able to determine whether any outpatient medications have not yet been administered (eg, antihypertensives, immune suppressants) and ensure that subsequent doses of medications initiated in the ED (eg, antibiotics) are administered during the appropriate timeframe.
Communication is emphasized, as contact with ED physicians, ward physicians, and often the outpatient primary care physician is required when any change in management is considered. The ED hospitalist also provides the capability of rapid response to changes in patient status (eg, a new complaint or fever). In addition, the hospitalist is available to consult on medical patients who may not require admission and on nonmedical patients for whom an internal medicine consult may be beneficial (eg, preoperative optimization of a surgical patient). The ED hospitalist documents the evaluation in the IBEX system. Bills were submitted for visits in which patients were discharged as these encounters are comprehensive, but not for other encounters.
Data Collection
The ED hospitalist role began March 10, 2008 and is a 10‐hour shift (8 AM to 6 PM) on weekdays. The study period was from March 10, 2008 through June 30, 2008. The study was approved by the hospital's institutional review board.
Data were collected on aspects of care that could have been impacted by the ED hospitalist, including medication and laboratory orders, ED discharges, ED admissions avoided, and telemetry downgrades. Discharges from ED refers to boarded admitted patients in the ED, who by the judgment of the ED hospitalist were ready for discharge. Admissions avoided refers to patients who the ED physician planned to admit but had not yet been admitted, and whose admission was avoided through the recommendations made by the ED hospitalist. The ED LOS was defined as the duration of time from when the patient was admitted to the Medicine Service to the time the patient was transferred to a medical ward. Telemetry downgrades were defined as patients assigned to the cardiac telemetry unit who the hospitalist determined required only telemetry on a general medical unit or did not require telemetry, or patients assigned to telemetry on a general medicine unit who the hospitalist determined no longer required telemetry.
Results were expressed as percentages of patients admitted to a Medicine Service and percentage of patients evaluated by the ED hospitalist, as indicated. 95% confidence intervals (CI) were calculated.
Results
During the study period, there were 4363 patients admitted to the Medicine Service and 3555 patients who qualified as boarders (mean of 29 boarders per 24 hours). The mean boarding time of admitted patients was 440 minutes. A total of 634 patients (17.8% of all boarded patients) were evaluated by the ED hospitalist. The mean daily number of patients seen by the ED hospitalist was 8.0.
The key elements of the delivery of care by the ED hospitalist are summarized in Table 2.
| Elements | Boarders (n = 3555) [n (%)] | Patients Intervened on (n = 634) [n (%)] |
|---|---|---|
| ||
| Laboratory results acted upon | 472 (13.2) | 472 (74.5) |
| Medication follow‐up | 506 (14.2) | 506 (79.8) |
| Discharges from the ED* | 46 (1.3) | 46 (7.3) |
| Admissions avoided | 6 (0.2) | 6 (0.95) |
| Telemetry downgrades | 61 (1.8) | 61 (9.6) |
The care of boarded patients included follow‐up of laboratory tests for 74.5% (95% CI, 71‐78%) and medication orders for 79.8% (95% CI, 77‐83%) of patients. A total of 46 patients were discharged by the ED hospitalist (0.6 discharges/day) and telemetry was discontinued for 61 patients (0.8 downgrades/day). The discharge rate was 7.3% (95% CI, 5‐10%) and telemetry downgrade rate was 9.6% (95% CI, 8‐12%) of those patients assessed by the ED hospitalist. Expressed as a percentage of the total ED boarders (n = 3555), the combined discharge rate and the admissions avoided rate was 1.5%.
Table 3 shows the discharge diagnoses made from the ED. Chest pain was the most common diagnosis, followed by syncope, pneumonia, and chronic obstructive pulmonary disease (COPD).
| Diagnoses | Patients (n = 46) [n (%)] |
|---|---|
| |
| Chest pain | 12 (26) |
| Syncope/dizziness | 7 (15) |
| Pneumonia | 4 (9) |
| COPD | 4 (9) |
| Congestive heart failure | 3 (7) |
| Gastroenteritis | 3 (7) |
| Dermatitis/rash | 3 (7) |
| Alcohol abuse | 3 (7) |
| Abdominal pain | 3 (7) |
| End stage renal disease | 2 (4) |
| Vaginal bleeding | 1 (2) |
| Fall | 1 (2) |
| Asthma | 1 (2) |
Discussion
Our hospital has successfully implemented an innovative strategy utilizing a hospitalist to help provide seamless care to medical patients located in the ED. Other solutions at our hospital had previously been implemented, but had not adequately addressed the problem, including: (1) protocols to monitor length of stay patterns and deviations, (2) discharge planning activities, (3) organized computerized bed tracking, (4) improvement in the timeliness of ancillary services, (5) daily bed briefing among nurse managers, and (6) 24‐hour presence of a MAR to facilitate triage in the ED.
The current study demonstrates the potential for substantial impact on patient care. The substantial number of the assessed boarder patients for whom laboratory tests (74.5%) and medications (79.8%) were ordered by the ED hospitalists suggests that the quality and timeliness of care was enhanced by this initiative. In addition, the considerable number of patients discharged from the ED and downgraded from telemetry (1.5% and 1.8% of all boarder patients, respectively) suggests that an ED hospitalist may have a meaningful impact on bed utilization and thus decrease ED overcrowding. In 2007, there were 11,488 who qualified as boarders; our data suggest that an ED hospitalist would result in approximately 172 boarders not being admitted annually.
Though the ED LOS was higher during the study period compared to 2007, it was lower than the 2 months immediately preceding implementation of the ED hospitalist role. The ED LOS was 732 and 658 minutes for January and February 2008, respectively, which was markedly increased from 2007 (288 minutes), and prompted development of the ED hospitalist role. The ED LOS during the study period subsequently decreased to 440 minutes. Though the wide fluctuations in ED LOS and the short time period with high ED LOS prior to implementation preclude concluding that the ED hospitalist role decreased ED LOS, the data suggest that an ED hospitalist may be able to improve ED throughput.
The majority of the discharges made by the ED hospitalist are patients who had been admitted for chest pain, had improved, and had negative cardiac enzymes and stress tests. Patients with syncope who were discharged were likely patients without any comorbidities. The COPD and pneumonia admissions were likely patients who improved after aggressive treatment in the ED.
The impact of ED overcrowding on the quality of patient care and outcomes may be substantial. Hwang et al.19 found a direct correlation between ED census and time to pain assessment and administration of analgesic medication. A study at an academic medical center found that higher ED volume was associated with less likelihood of antibiotics being administered within 4 hours for patients with community‐acquired pneumonia.20 A comprehensive review of the literature identified 41 studies examining the effects of ED overcrowding on clinical outcomes and the investigators noted that ED overcrowding was associated with increased in‐hospital mortality.8
Causes of poor outcomes during periods of overcrowding may be the high volume of acute patients preventing adequate time and attention for each ED patient, as well as confusion during the transition from ED to ward physicians. For example, a patient may receive their initial dose of antibiotics from the ED physician, but subsequent doses may be overlooked in the transition of care from the ED physician to the inpatient team. In addition, having admitted patients located in the ED for extended periods of time may lead to these patients not being seen as frequently as patients admitted to the inpatient wards. Another potential consequence of prolonged ED stay for admitted patients is delay in inpatient management. Tests done in the ED may prompt further studies that may not be ordered promptly while patients remain in the ED, which subsequently increases LOS. Other potential issues may be an increase in confusion among geriatric patients in a noisy and crowded ED; decreased access to specialized nursing care that may be available on a hospital ward; decreased access to physical therapy and occupational therapy services; and decreased comfort and satisfaction as patients wait in overcrowded EDs for prolonged periods.
Several other potential innovative solutions to ED overcrowding have been proposed, studied, and tested. These measures generally are focused on improving the three interdependent components of ED workflow: INPUT THROUGHPUT OUTPUT.21, 22 However, process redesign and intervention on these 3 interdependent ED workflow components may be difficult to achieve, especially when hospital resources are limited and when inpatient hospital capacity is already maximized. In some institutions, efforts have been reported to successfully streamline the transfer of admitted ED patients to inpatient beds, through transfer‐to‐ward policy interventions (eg, physician coordinators for patient flow and bed management or transfers made within a defined period of time).2326 However, in a study by Quinn et al.,27 implementation of a rapid admission policy resulted in a decrease of only 10.1 minutes in the ED LOS. Several studies have demonstrated the benefits of an acute medical admissions unit in alleviating ED overcrowding.28, 29 Other unconventional solutions by some hospitals include sending admitted patients to the unit's hallways or placing discharged patients in the hallway while waiting for transportation so that the ED bed will be readily available.30
The ED hospitalist is well‐situated to have an impact on several key hospital outcomes. As the ED hospitalist role was shown to affect processes that relate to ED throughput, it is possible that the role will improve ED overcrowding and decrease ED LOS. Specifically, identifying patients who can be discharged and for whom telemetry is no longer indicated decreases unnecessary bed utilization and allows these beds to be available for other ED patients. This initiative also may promote patient satisfaction by assuring patients that their medical and concerns are being fully addressed while they are in the ED. Increased emphasis on hospital reporting will make patient satisfaction a priority for many hospitals, and the ED hospitalist will be in a unique position to meet and greet patients admitted to the Medicine Service and to reassure them that the medical team is present and addressing their concerns. The hospitalist's ability to facilitate diagnostic testing and treatment while patients remain in the ED may also help decrease the total LOS in the hospital. In addition, the ED hospitalist is also in position to recognize social factors at the earliest stage of admission so that they can be immediately addressed. Future studies will need to be done to determine if this model of transitional care impacts these important factors.
Our study has several important limitations. Most notably, the lack of a comparison interval for which a hospitalist was not assigned to this role prevents us from drawing any definitive conclusions on the benefits of the ED hospitalist model. Also, we collected only summary data and do not have demographic data on the patients managed by the ED hospitalist or information on the ED course of patients who were discharged or had telemetry downgraded. This prevents determination of whether discharged patients did not require admission initially or whose condition evolved over a prolonged ED stay. In addition, other key outcomes, such as patient satisfaction and satisfaction of the ED physicians and nursing staff have not yet been formally measured. Future studies will be needed to determine if an ED hospital model can improve important process and clinical outcomes.
The greatest challenge of this initiative was introducing and familiarizing this role to the key stakeholders, including the ED physicians and nursing staff, house staff, and private practice physicians. Though we did not perform structured surveys on satisfaction, through informal discussions we noted that the role was welcomed with enthusiasm by the ED physicians. Notably, several ED physicians expressed appreciation that they were able to focus their care on new ED patients rather than on the boarded ED patients. Through feedback, we noted soon after implementation that ED faculty and nurses needed further clarification about the potential overlapping roles of the ED hospitalist and ED physicians and ward physicians. These concerns were addressed by educational sessions and announcements, including presentations at ED faculty and staff meetings. The hospitalist assigned to the role each month received individualized orientation prior to assuming the role, and an ED Hospitalist Manual was distributed. Possibly due to these focused sessions, the hospitalists assigned to the role became quickly acclimated.
Conclusions
We have found that designating a hospitalist to directly address the care of ED boarders can enhance the quality and timeliness of care and decrease bed and telemetry utilization with the potential to impact ED and hospital LOS. Given the success of the pilot model, the role was expanded at our institution to 10 hours per day, 7 days per week. Hospitals struggling to address the needs of their admitted patients in the ED should consider incorporating an ED hospitalist to enhance clinical care and address issues relating to throughput. A follow‐up study is needed to more precisely describe the impact of the ED hospitalist model.
Emergency Department (ED) overcrowding has become an important problem in North American hospitals.13 A national survey identified the prolonged length of stay of admitted patients in the ED as the most frequent reason for overcrowding.4 This complex problem occurs when hospital inpatient census increases and prevents admitted patients from being assigned and transported to hospital beds in a timely manner.5 The practice of holding admitted patients in the ED, known as boarding, is typically defined as the length of stay (LOS) in ED beginning 2 hours after the time of admission to the time of transfer to the wards.
In a study of daily mean ED LOS, Rathlev et al.6 concluded that a 5% increase in hospital occupancy resulted in 14 hours of holding time for all patients in the ED, and an observational study found that when hospital occupancy exceeds a threshold of 90%, the ED LOS for admitted patients correspondingly increased.7 Thus, efforts to decrease overcrowding will need to address both ED and hospital throughput and LOS. Most importantly, overcrowding has important consequences on physician and patient satisfaction and the quality of patient care.811
Between 1995 and 2005, ED visits rose 20% from 96.5 million to 115.3 million visits annually, while the number of hospital EDs decreased from 4176 to 3795, making an overall 7% increase in ED utilization rate.12 Similarly, there was a 12% increase in the total inpatient admissions for all registered hospitals in the United States from 31 million in 1995 to 35.3 million in 2005.13 However, despite this increase in demand of ED utilization and inpatient admissions, there had been a steady decline in the supply of hospital beds, from 874,000 in 1995, to 805,000 in 2006.13 These factors have exacerbated the problem of ED overcrowding and boarding.
Not only does boarding entail additional consumption of space, resources, equipment, and manpower, it also potentially compromises patient safety. Typically, hospitalists and inpatient medical teams are engaged in providing care to patients in the wards, while ED physicians and nurses are busy taking care of newly‐arrived ED patients. Non‐ED physicians may have the false impression that their boarded patients, while in the ED, are receiving continuous care and so may decide to delay seeing these patients, which can jeopardize the quality and timeliness of care. Studies have shown that ED overcrowding may potentially lead to poor patient care and outcomes and increased risk for medical errors.1416 ED overcrowding potentially causes multiple effects, including prolonging patient pain and suffering, long patient waiting time, patient dissatisfaction, ambulance diversions, decreased physician productivity, and increased frustration among medical staff.15 In a report by the Joint Commission Accreditation of Healthcare Organizations,17 ED overcrowding was cited as a significant contributing factor in sentinel event cases of patient death or permanent injury due to delays in treatment. Boarding critically ill patients who are physiologically vulnerable and unstable can allow them to be subjected to treatment delays at a pivotal point when time‐sensitive interventions are necessary, ie, sepsis or cardiogenic shockthe golden hour in trauma.16 Medical errors are usually not caused by individual errors but by complex hospital systems; and ED overcrowding is a prime example of a system problem that creates a high‐risk environment for medical errors and threatens patient safety.18
Our hospital commonly has 5 to 15 boarders and often has 20 to 30 boarders at any time. Approximately 90% of these patients are admitted to the Medical Service. In response to this challenge, our institution has designated a full‐time hospitalist to manage boarded patients. The primary goal of this new role is to ensure patient safety and the delivery of high‐quality care while admitted patients are in the ED (Table 1).
| |
| 1 | Round on all patients admitted to the Department of Medicine located in the ED, including those on the Teaching and Nonteaching Services. Rounds focus on patient safety, such as ensuring vital home and hospital medications are administered and changes in stability are noted. All patient updates are documented in the ED electronic medical records (IBEX). |
| 2 | Identify admitted patients who may be downgraded from telemetry to nontelemetry status. Telemetry and cardiac beds are in high demand, and decreasing utilization facilitates obtaining the appropriate ward bed for ED patients. |
| 3 | Assess admitted patients for possible discharge. The patient's condition may have improved or results may indicate that admission is no longer required. The ED hospitalist communicates with the ED physician and wards teams, facilitates management, implements the discharge, and ensures adequate follow‐up. |
| 4 | Refer patients to an ED social worker as needed. |
| 5 | Facilitate referrals to other medical or surgical specialties if indicated. |
| 6 | Clarify the plan of care with the ED staff and facilitates ED communication with the ward team. Acts as a liaison and a resource for the ED physicians and nursing staff. |
| 7 | Supervise the triage duties of the medical admitting resident. |
| 8 | Provide medical consultation to ED physicians for patients not being admitted to the hospital or who are being admitted to other services (eg, surgery). |
The objectives of the study were to determine: (1) the impact on quality of care by assessing laboratory results acted upon and medication follow‐up by the ED hospitalist, and (2) the impact on throughput by assessing the number of ED discharges and telemetry downgrades.
Methods
Setting
The Mount Sinai Medical Center is a tertiary‐care 1121‐bed acute care teaching hospital located in New York City. The hospital borders East Harlem and the Upper East Side of Manhattan. The Medical Service is composed of a Teaching Service, composed of house staff and attendings, and a non‐Teaching Service, composed of nurse practitioners, physician assistants, and attendings. Hospitalists and private attendings may have patients on either the Teaching or the non‐Teaching Service. In 2007, there were 56,541 patients admitted for a total of 332,368 days. The mean LOS for medical inpatients was 5.89 days. The total ED visit was 79,500 with a total inpatient and critical care admissions of 24,522. The mean and median LOS for all ED patients were 623 minutes and 493 minutes, respectively. There were 11,488 patients who qualified as boarders, averaging 31.5 boarders per 24 hours; with a mean and median LOS per boarder of 288 minutes and 198 minutes, respectively. The ED LOS for admitted patients ranged from 2 minutes to 4074 minutes (2.83 days).
Admission Process
Once an ED attending physician decides that a patient is to be admitted, the patient is placed on a computerized list in the ED's electronic medical record (IBEX software). The Medical Admitting Resident (MAR) evaluates and triages admitted patients, and assigns and gives a verbal report to the appropriate Medicine Service (ie, Teaching, non‐Teaching, cardiac telemetry unit, intensive care, etc.). The Admitting Office searches for and assigns the appropriate unit and bed for the patient. A hospitalist or resident physician performs the patient's initial assessment and evaluation in the ED, and admission orders are placed in the inpatient computerized order entry system (TDS). When the bed is ready, the ED nurse gives a verbal report to the floor nurse, and the patient is transported to the ward.
Responsibilities
The specific responsibilities of the ED hospitalist are listed in Table 1. The primary role is to round on patients admitted to the Medicine Service who are located in the ED. This encompasses a wide array of patients and services, including patients assigned to a hospitalist service attending or who have a private attending, patients admitted to the Teaching or non‐Teaching Service, patients admitted to the intensive care unit, and patients admitted to a general medicine or specialty service (eg, telemetry, oncology, human immunodeficiency virus [HIV]). Rounding includes review of the ED's electronic medical record as well as direct examination of patients. The hospitalist focuses on patients with longer ED LOS and on aspects of care that may lapse while patients remain in the ED for prolonged periods. At our institution, the follow‐up of subsequent tests, laboratory values, and medications for ED boarders is the responsibility of the primary inpatient team, though the ED physicians act on urgent and critical results and continue to deliver all emergency care. Through rounding, the ED hospitalist is able to identify abnormal results in a timely manner, alert the ED physician and primary inpatient team, and address abnormalities. Specific examples of laboratory results acted upon include hypokalemia, hyperglycemia, and elevated cardiac enzymes. The ED hospitalist is also able to determine whether any outpatient medications have not yet been administered (eg, antihypertensives, immune suppressants) and ensure that subsequent doses of medications initiated in the ED (eg, antibiotics) are administered during the appropriate timeframe.
Communication is emphasized, as contact with ED physicians, ward physicians, and often the outpatient primary care physician is required when any change in management is considered. The ED hospitalist also provides the capability of rapid response to changes in patient status (eg, a new complaint or fever). In addition, the hospitalist is available to consult on medical patients who may not require admission and on nonmedical patients for whom an internal medicine consult may be beneficial (eg, preoperative optimization of a surgical patient). The ED hospitalist documents the evaluation in the IBEX system. Bills were submitted for visits in which patients were discharged as these encounters are comprehensive, but not for other encounters.
Data Collection
The ED hospitalist role began March 10, 2008 and is a 10‐hour shift (8 AM to 6 PM) on weekdays. The study period was from March 10, 2008 through June 30, 2008. The study was approved by the hospital's institutional review board.
Data were collected on aspects of care that could have been impacted by the ED hospitalist, including medication and laboratory orders, ED discharges, ED admissions avoided, and telemetry downgrades. Discharges from ED refers to boarded admitted patients in the ED, who by the judgment of the ED hospitalist were ready for discharge. Admissions avoided refers to patients who the ED physician planned to admit but had not yet been admitted, and whose admission was avoided through the recommendations made by the ED hospitalist. The ED LOS was defined as the duration of time from when the patient was admitted to the Medicine Service to the time the patient was transferred to a medical ward. Telemetry downgrades were defined as patients assigned to the cardiac telemetry unit who the hospitalist determined required only telemetry on a general medical unit or did not require telemetry, or patients assigned to telemetry on a general medicine unit who the hospitalist determined no longer required telemetry.
Results were expressed as percentages of patients admitted to a Medicine Service and percentage of patients evaluated by the ED hospitalist, as indicated. 95% confidence intervals (CI) were calculated.
Results
During the study period, there were 4363 patients admitted to the Medicine Service and 3555 patients who qualified as boarders (mean of 29 boarders per 24 hours). The mean boarding time of admitted patients was 440 minutes. A total of 634 patients (17.8% of all boarded patients) were evaluated by the ED hospitalist. The mean daily number of patients seen by the ED hospitalist was 8.0.
The key elements of the delivery of care by the ED hospitalist are summarized in Table 2.
| Elements | Boarders (n = 3555) [n (%)] | Patients Intervened on (n = 634) [n (%)] |
|---|---|---|
| ||
| Laboratory results acted upon | 472 (13.2) | 472 (74.5) |
| Medication follow‐up | 506 (14.2) | 506 (79.8) |
| Discharges from the ED* | 46 (1.3) | 46 (7.3) |
| Admissions avoided | 6 (0.2) | 6 (0.95) |
| Telemetry downgrades | 61 (1.8) | 61 (9.6) |
The care of boarded patients included follow‐up of laboratory tests for 74.5% (95% CI, 71‐78%) and medication orders for 79.8% (95% CI, 77‐83%) of patients. A total of 46 patients were discharged by the ED hospitalist (0.6 discharges/day) and telemetry was discontinued for 61 patients (0.8 downgrades/day). The discharge rate was 7.3% (95% CI, 5‐10%) and telemetry downgrade rate was 9.6% (95% CI, 8‐12%) of those patients assessed by the ED hospitalist. Expressed as a percentage of the total ED boarders (n = 3555), the combined discharge rate and the admissions avoided rate was 1.5%.
Table 3 shows the discharge diagnoses made from the ED. Chest pain was the most common diagnosis, followed by syncope, pneumonia, and chronic obstructive pulmonary disease (COPD).
| Diagnoses | Patients (n = 46) [n (%)] |
|---|---|
| |
| Chest pain | 12 (26) |
| Syncope/dizziness | 7 (15) |
| Pneumonia | 4 (9) |
| COPD | 4 (9) |
| Congestive heart failure | 3 (7) |
| Gastroenteritis | 3 (7) |
| Dermatitis/rash | 3 (7) |
| Alcohol abuse | 3 (7) |
| Abdominal pain | 3 (7) |
| End stage renal disease | 2 (4) |
| Vaginal bleeding | 1 (2) |
| Fall | 1 (2) |
| Asthma | 1 (2) |
Discussion
Our hospital has successfully implemented an innovative strategy utilizing a hospitalist to help provide seamless care to medical patients located in the ED. Other solutions at our hospital had previously been implemented, but had not adequately addressed the problem, including: (1) protocols to monitor length of stay patterns and deviations, (2) discharge planning activities, (3) organized computerized bed tracking, (4) improvement in the timeliness of ancillary services, (5) daily bed briefing among nurse managers, and (6) 24‐hour presence of a MAR to facilitate triage in the ED.
The current study demonstrates the potential for substantial impact on patient care. The substantial number of the assessed boarder patients for whom laboratory tests (74.5%) and medications (79.8%) were ordered by the ED hospitalists suggests that the quality and timeliness of care was enhanced by this initiative. In addition, the considerable number of patients discharged from the ED and downgraded from telemetry (1.5% and 1.8% of all boarder patients, respectively) suggests that an ED hospitalist may have a meaningful impact on bed utilization and thus decrease ED overcrowding. In 2007, there were 11,488 who qualified as boarders; our data suggest that an ED hospitalist would result in approximately 172 boarders not being admitted annually.
Though the ED LOS was higher during the study period compared to 2007, it was lower than the 2 months immediately preceding implementation of the ED hospitalist role. The ED LOS was 732 and 658 minutes for January and February 2008, respectively, which was markedly increased from 2007 (288 minutes), and prompted development of the ED hospitalist role. The ED LOS during the study period subsequently decreased to 440 minutes. Though the wide fluctuations in ED LOS and the short time period with high ED LOS prior to implementation preclude concluding that the ED hospitalist role decreased ED LOS, the data suggest that an ED hospitalist may be able to improve ED throughput.
The majority of the discharges made by the ED hospitalist are patients who had been admitted for chest pain, had improved, and had negative cardiac enzymes and stress tests. Patients with syncope who were discharged were likely patients without any comorbidities. The COPD and pneumonia admissions were likely patients who improved after aggressive treatment in the ED.
The impact of ED overcrowding on the quality of patient care and outcomes may be substantial. Hwang et al.19 found a direct correlation between ED census and time to pain assessment and administration of analgesic medication. A study at an academic medical center found that higher ED volume was associated with less likelihood of antibiotics being administered within 4 hours for patients with community‐acquired pneumonia.20 A comprehensive review of the literature identified 41 studies examining the effects of ED overcrowding on clinical outcomes and the investigators noted that ED overcrowding was associated with increased in‐hospital mortality.8
Causes of poor outcomes during periods of overcrowding may be the high volume of acute patients preventing adequate time and attention for each ED patient, as well as confusion during the transition from ED to ward physicians. For example, a patient may receive their initial dose of antibiotics from the ED physician, but subsequent doses may be overlooked in the transition of care from the ED physician to the inpatient team. In addition, having admitted patients located in the ED for extended periods of time may lead to these patients not being seen as frequently as patients admitted to the inpatient wards. Another potential consequence of prolonged ED stay for admitted patients is delay in inpatient management. Tests done in the ED may prompt further studies that may not be ordered promptly while patients remain in the ED, which subsequently increases LOS. Other potential issues may be an increase in confusion among geriatric patients in a noisy and crowded ED; decreased access to specialized nursing care that may be available on a hospital ward; decreased access to physical therapy and occupational therapy services; and decreased comfort and satisfaction as patients wait in overcrowded EDs for prolonged periods.
Several other potential innovative solutions to ED overcrowding have been proposed, studied, and tested. These measures generally are focused on improving the three interdependent components of ED workflow: INPUT THROUGHPUT OUTPUT.21, 22 However, process redesign and intervention on these 3 interdependent ED workflow components may be difficult to achieve, especially when hospital resources are limited and when inpatient hospital capacity is already maximized. In some institutions, efforts have been reported to successfully streamline the transfer of admitted ED patients to inpatient beds, through transfer‐to‐ward policy interventions (eg, physician coordinators for patient flow and bed management or transfers made within a defined period of time).2326 However, in a study by Quinn et al.,27 implementation of a rapid admission policy resulted in a decrease of only 10.1 minutes in the ED LOS. Several studies have demonstrated the benefits of an acute medical admissions unit in alleviating ED overcrowding.28, 29 Other unconventional solutions by some hospitals include sending admitted patients to the unit's hallways or placing discharged patients in the hallway while waiting for transportation so that the ED bed will be readily available.30
The ED hospitalist is well‐situated to have an impact on several key hospital outcomes. As the ED hospitalist role was shown to affect processes that relate to ED throughput, it is possible that the role will improve ED overcrowding and decrease ED LOS. Specifically, identifying patients who can be discharged and for whom telemetry is no longer indicated decreases unnecessary bed utilization and allows these beds to be available for other ED patients. This initiative also may promote patient satisfaction by assuring patients that their medical and concerns are being fully addressed while they are in the ED. Increased emphasis on hospital reporting will make patient satisfaction a priority for many hospitals, and the ED hospitalist will be in a unique position to meet and greet patients admitted to the Medicine Service and to reassure them that the medical team is present and addressing their concerns. The hospitalist's ability to facilitate diagnostic testing and treatment while patients remain in the ED may also help decrease the total LOS in the hospital. In addition, the ED hospitalist is also in position to recognize social factors at the earliest stage of admission so that they can be immediately addressed. Future studies will need to be done to determine if this model of transitional care impacts these important factors.
Our study has several important limitations. Most notably, the lack of a comparison interval for which a hospitalist was not assigned to this role prevents us from drawing any definitive conclusions on the benefits of the ED hospitalist model. Also, we collected only summary data and do not have demographic data on the patients managed by the ED hospitalist or information on the ED course of patients who were discharged or had telemetry downgraded. This prevents determination of whether discharged patients did not require admission initially or whose condition evolved over a prolonged ED stay. In addition, other key outcomes, such as patient satisfaction and satisfaction of the ED physicians and nursing staff have not yet been formally measured. Future studies will be needed to determine if an ED hospital model can improve important process and clinical outcomes.
The greatest challenge of this initiative was introducing and familiarizing this role to the key stakeholders, including the ED physicians and nursing staff, house staff, and private practice physicians. Though we did not perform structured surveys on satisfaction, through informal discussions we noted that the role was welcomed with enthusiasm by the ED physicians. Notably, several ED physicians expressed appreciation that they were able to focus their care on new ED patients rather than on the boarded ED patients. Through feedback, we noted soon after implementation that ED faculty and nurses needed further clarification about the potential overlapping roles of the ED hospitalist and ED physicians and ward physicians. These concerns were addressed by educational sessions and announcements, including presentations at ED faculty and staff meetings. The hospitalist assigned to the role each month received individualized orientation prior to assuming the role, and an ED Hospitalist Manual was distributed. Possibly due to these focused sessions, the hospitalists assigned to the role became quickly acclimated.
Conclusions
We have found that designating a hospitalist to directly address the care of ED boarders can enhance the quality and timeliness of care and decrease bed and telemetry utilization with the potential to impact ED and hospital LOS. Given the success of the pilot model, the role was expanded at our institution to 10 hours per day, 7 days per week. Hospitals struggling to address the needs of their admitted patients in the ED should consider incorporating an ED hospitalist to enhance clinical care and address issues relating to throughput. A follow‐up study is needed to more precisely describe the impact of the ED hospitalist model.
- , , , et al.Estimating the degree of emergency department overcrowding in academic medical centers: results of the National ED Overcrowding Study (NEDOCS).Acad Emerg Med.2004;11:38–50.
- , , , et al.Frequency, determinants, and impact of overcrowding in emergency departments in Canada: a national survey.Healthc Q.2007;10:32–40.
- , .EMDOC (emergency department overcrowding) internet‐based safety net research.Admin Emerg Med.2008;35:101–107.
- United States General Accounting Office.Hospital Emergency Departments: Crowded Conditions Vary Among Hospitals and Communities. March 2003.Washington, DC:General Accounting Office;2003.
- .Overcrowding in emergency department: increased demand and decreased capacity.Ann Emerg Med.2002;39:430–432.
- , , , et al.Time series analysis of variables associated with daily mean emergency department length of stay.Ann Emerg Med.2007;49:265–271.
- , , , et al.Effect of hospital occupancy on emergency department length of stay and patient disposition.Ann Emerg Med.2003;10:127–133.
- , , , et al.The effect of emergency department crowding on clinically oriented outcomes.Acad Emerg Med.2009;16:1–10.
- , .Emergency department overcrowding: the impact of resource scarcity on physician job satisfaction.J Health Manag.2005;50:327–340.
- , , , , , .The effect of emergency department crowding on patient satisfaction for admitted patients.Acad Emerg Med.2008;15:825–831.
- , .The effect of crowding on access and quality in an academic ED.Am J Emerg Med.2006;24:787–794.
- , , .National Hospital Ambulatory Medical Care Survey: 2005 Emergency Department Summary. Advance Data from Vital and Health Statistics. No. 386.Hyattsville, MD:National Center for Health Statistics;2007.
- American Hospital Association (AHA).Table 1: Historical trends in utilization, personnel, and finances: year 1946–2006.AHA Hospital Statistics.2008 ed.Chicago:Health Forum LLC;2008:3.
- , .Emergency department overcrowding in the US: an emerging threat to patient safety and public health.Emerg Med J.2003;20:402–405.
- , .Overcrowding in the nation's emergency departments: complex causes and disturbing effects.Ann Emerg Med.2000;35:63–68.
- , .Clinical review: emergency department overcrowding and the potential impact on the critically ill.Crit Care.2005;9:291–295.
- Joint Commission on Accreditation of Healthcare Organizations (JCAHO): Sentinel event alert 2002, Issue 26. Available at: http://www.jointcommission.org/SentinelEvents/SentinelEventAlert/sea_26.htm. Accessed October2009.
- , , , et al.Safety net research in emergency medicine: proceedings of the Academic Emergency Consensus Conference on “The Unraveling Safety Net.”Acad Emerg Med.2001;8:1024–1029.
- , , , , , .Emergency department crowding and decreased quality of pain care.Acad Emerg Med.2008;15:1248–1256.
- , , , .Effect of emergency department crowding on time to antibiotics in patients admitted with community‐acquired pneumonia.Ann Emerg Med.2007;50:501–509.
- , , , et al.A conceptual model of emergency department crowding.Ann Emerg Med.2003;42:173–180.
- , , , et al.Emergency department crowding: consensus development of potential measures.Ann Emerg Med.2003;42:824–834.
- , , , et al.Intervention to decrease emergency department crowding: does it have an effect on return visits and hospital readmission?Ann Emerg Med.2003;41:173–185.
- , , , et al.Rapid process design in a university‐based emergency department: decreasing waiting time intervals and improving patient satisfaction.Ann Emerg Med.2002;39:168–177.
- .Emergency department crowding: an action plan.Acad Emerg Med.2001;18:185–187.
- , , .Hospitalists and an innovative emergency department admission process.J Gen Intern Med.2004;19:266–268.
- , , et al.Effects of implementing a rapid admission policy in the ED.Am J Emerg Med.2007;25:559–563.
- , , .Effect of an emergency department managed acute care unit on ED overcrowding and emergency medical services diversion.Acad Emerg Med.2001;8:1085–1100.
- , , , .Emergency department census of patients awaiting admission following reorganization of an admissions process.Emerg Med J.2006;23:363–367.
- .Emergency department flow and the boarded patient: how to get admitted patients upstairs.Ann Emerg Med.2007;49:68–70.
- , , , et al.Estimating the degree of emergency department overcrowding in academic medical centers: results of the National ED Overcrowding Study (NEDOCS).Acad Emerg Med.2004;11:38–50.
- , , , et al.Frequency, determinants, and impact of overcrowding in emergency departments in Canada: a national survey.Healthc Q.2007;10:32–40.
- , .EMDOC (emergency department overcrowding) internet‐based safety net research.Admin Emerg Med.2008;35:101–107.
- United States General Accounting Office.Hospital Emergency Departments: Crowded Conditions Vary Among Hospitals and Communities. March 2003.Washington, DC:General Accounting Office;2003.
- .Overcrowding in emergency department: increased demand and decreased capacity.Ann Emerg Med.2002;39:430–432.
- , , , et al.Time series analysis of variables associated with daily mean emergency department length of stay.Ann Emerg Med.2007;49:265–271.
- , , , et al.Effect of hospital occupancy on emergency department length of stay and patient disposition.Ann Emerg Med.2003;10:127–133.
- , , , et al.The effect of emergency department crowding on clinically oriented outcomes.Acad Emerg Med.2009;16:1–10.
- , .Emergency department overcrowding: the impact of resource scarcity on physician job satisfaction.J Health Manag.2005;50:327–340.
- , , , , , .The effect of emergency department crowding on patient satisfaction for admitted patients.Acad Emerg Med.2008;15:825–831.
- , .The effect of crowding on access and quality in an academic ED.Am J Emerg Med.2006;24:787–794.
- , , .National Hospital Ambulatory Medical Care Survey: 2005 Emergency Department Summary. Advance Data from Vital and Health Statistics. No. 386.Hyattsville, MD:National Center for Health Statistics;2007.
- American Hospital Association (AHA).Table 1: Historical trends in utilization, personnel, and finances: year 1946–2006.AHA Hospital Statistics.2008 ed.Chicago:Health Forum LLC;2008:3.
- , .Emergency department overcrowding in the US: an emerging threat to patient safety and public health.Emerg Med J.2003;20:402–405.
- , .Overcrowding in the nation's emergency departments: complex causes and disturbing effects.Ann Emerg Med.2000;35:63–68.
- , .Clinical review: emergency department overcrowding and the potential impact on the critically ill.Crit Care.2005;9:291–295.
- Joint Commission on Accreditation of Healthcare Organizations (JCAHO): Sentinel event alert 2002, Issue 26. Available at: http://www.jointcommission.org/SentinelEvents/SentinelEventAlert/sea_26.htm. Accessed October2009.
- , , , et al.Safety net research in emergency medicine: proceedings of the Academic Emergency Consensus Conference on “The Unraveling Safety Net.”Acad Emerg Med.2001;8:1024–1029.
- , , , , , .Emergency department crowding and decreased quality of pain care.Acad Emerg Med.2008;15:1248–1256.
- , , , .Effect of emergency department crowding on time to antibiotics in patients admitted with community‐acquired pneumonia.Ann Emerg Med.2007;50:501–509.
- , , , et al.A conceptual model of emergency department crowding.Ann Emerg Med.2003;42:173–180.
- , , , et al.Emergency department crowding: consensus development of potential measures.Ann Emerg Med.2003;42:824–834.
- , , , et al.Intervention to decrease emergency department crowding: does it have an effect on return visits and hospital readmission?Ann Emerg Med.2003;41:173–185.
- , , , et al.Rapid process design in a university‐based emergency department: decreasing waiting time intervals and improving patient satisfaction.Ann Emerg Med.2002;39:168–177.
- .Emergency department crowding: an action plan.Acad Emerg Med.2001;18:185–187.
- , , .Hospitalists and an innovative emergency department admission process.J Gen Intern Med.2004;19:266–268.
- , , et al.Effects of implementing a rapid admission policy in the ED.Am J Emerg Med.2007;25:559–563.
- , , .Effect of an emergency department managed acute care unit on ED overcrowding and emergency medical services diversion.Acad Emerg Med.2001;8:1085–1100.
- , , , .Emergency department census of patients awaiting admission following reorganization of an admissions process.Emerg Med J.2006;23:363–367.
- .Emergency department flow and the boarded patient: how to get admitted patients upstairs.Ann Emerg Med.2007;49:68–70.
Copyright © 2009 Society of Hospital Medicine
Effectiveness of Course to Teach Handoffs
Communication failures are well‐recognized as causes of medical errors.1, 2 Specifically, handoffs of patient care responsibilities, which are increasingly prevalent in academic medical centers,3 have been cited as the most frequent cause of teamwork breakdown resulting in the harmful medical errors found in malpractice claims.1 The Institute of Medicine has recently identified patient handoffs as the moment where patient care errors are most likely to occur.4 A survey of 125 U.S. medical schools, however, found that only 8% specifically taught students how to hand off patient care.3
In July 2003, the American Council of Graduate Medical Education (ACGME) mandated that residency programs decrease resident work hours to improve patient care and safety by reducing fatigue,5 and a recent Institute of Medicine report suggests that they be decreased even further.4 Studies examining outcomes during the first 2 years after reducing duty hours did not find reductions in risk‐adjusted mortality.68 One proposed explanation for this lack of improvement is that the reduction in fatigue‐related medical errors is being offset by discontinuity of care with due to the increased number of patient handoffs resulting from shortened duty hours,911 one recent study found that omission of key information during patient sign outs frequently resulted in adverse patient care outcomes.12
In 2007, the Joint Commission developed a new National Patient Safety Goal that requires organizations to improve communication between caregivers.13 We recently developed an approach by which Internal Medicine residents hand off patient care using a structured process, written and verbal templates, formal training about handoffs, and direct attending supervision.14 Because fourth‐year medical students perform the duties of interns when working as subinterns, we recognized that education about handoffs should occur prior to the time students became interns. Accordingly, we developed a course designed to teach patient handoffs to medical students at the transition between their third and fourth years of training.
Setting
The Handoff Selective was developed by faculty of Denver Health and the University of Colorado Denver School of Medicine.
Program Description
The Selective was first offered in April 2007 as part of an Integrated Clinician's Course (ICC), a 2‐week course for students beginning their fourth year, which starts in April at the University of Colorado. The ICC includes both mandatory and selective sessions that are focused on developing clinical skills and preparing them for their subinternships. The Handoff Selective was conducted in a computerized teaching laboratory, lasted a total of 2 hours and consisted of 2 parts. Each of the 5 Denver Health Hospital Medicine faculty members versed in handoff education taught 2 sessions of 6 to 8 students.
Part 1: Didactic
During the first hour of class, the faculty presented a lecture that summarized the relevant literature on handoffs and explained the importance of the topic. The objectives of the didactic were to: (1) understand the importance of handoffs; (2) explore different communication elements and structures; (3) gain exposure to handoffs outside of healthcare; and (4) learn a structure for handoffs of patient care in hospitalized patients.
We used 3 video clips of handoffs from 2 football games to demonstrate the importance of practice, training, and 2‐way communications in handoffs. The first video clip showed a runner trying to make a spontaneous handoff while being tackled. The receiver was not expecting the handoff and was preoccupied with blocking another player. This attempted handoff resulted in a fumble, which we related to an adverse patient event.
The next 2 video clips showed 2 complex, seldom used, but well‐known football handoffsthe hook and lateral and the Statue of Liberty. Both handoffs were successfully executed presumably as a result of education, practice and the active participation of both players (handing off and receiving) in the process. We then related the teaching and practicing of complex communication to the Joint Commission on Accreditation of Healthcare Organizations (JCAHO; now simply the Joint Commission) data suggesting that most sentinel events have their root cause in communication and training failures.2
Basic communication elements and process structures were then explored using scenarios from everyday life and evidence from fields outside of medicine. We emphasized that structures for communication (modes, vehicles, and settings) must be chosen according to the occasion and that handoffs are common and important in all occupations. In discussing modes (verbal, written, or nonverbal), vehicles (paper, telephone, or e‐mail), and settings (face‐to face, virtual, or disconnected), we emphasized that the most effective structures for communication (verbal, face‐to face meetings, with written materials and other visual aids at the patient's bedside) were also the most time‐consuming (Figure 1). While our standard for resident handoffs is a face‐to‐face verbal interaction with preprinted written materials as an aid, we also emphasized that for complex patients (eg, mental status changes, concern for an acute abdomen) more robust communication is often needed. Accordingly, a more time‐consuming bedside handoff with simultaneous, focused physical exam and history‐taking by both oncoming and off‐going providers may be most appropriate.
As real‐life examples, we asked our students to communicate a happy birthday wish to their mother, who lives in another state. Almost uniformly, in addition to a written aid (birthday card), they choose the telephone as a vehicle for their verbal mode in a virtual setting with 2‐way communication possible. In contrast, when asked to propose marriage to a significant other in another state, students felt that a face‐to‐face meeting with verbal and nonverbal (ie, ring) modes was appropriate. This time‐consuming mode of communication was felt to be necessary to create a sentiment of importance and avert any possible miscommunication.
The didactic session concluded by demonstrating how to use standardized written and verbal templates for handoffs of the care of a hospitalized patient. We explore the differentiation between written and verbal handoffs in our discussion below.
Part 2: Practicum
The second hour was devoted to practicing handoffs as a group. The faculty developed 6 case scenarios that differed with respect to diagnosis, length of stay, active medical issues, and anticipated discharge (Table 1). The scenarios included extensive admission information as well as evolving issues for each patient that were specific to the day of the intended handoff. Students were given Microsoft Word table‐based handoff templates to use when creating written sign‐outs for their patients. Verbal handoffs were performed between students and sign‐outs were exchanged. The faculty then role‐played cross‐cover calls that were specific for each scenario to test the students' inclusion of integral information in their handoffs and their ability to create contingency plans.
| Diagnosis | LOS | Active Issues | Cross‐Cover |
|---|---|---|---|
| |||
| CP | 1 | CP, HTN, DM | CP, HTN, headache |
| GIB | 1 | GIB, alcohol withdrawal | Poor response to red call transfusion, coagulopathy |
| Acute pancreatitis | 2 | Pain, possible pancreatic abscess | Fever, agitation, hypoxia |
| CHF | 2 | CHF, DM, nausea | Lack of diuresis, CP, hypoglycemia |
| Acute kidney injury | 3 | None, ready for discharge | HTN, hyperglycemia |
| Community acquired pneumonia | 3 | Anxiety, discharge pending | Confusion, emesis with hypoxia |
Program Evaluation
We developed a 2‐part survey to evaluate the effectiveness of the Selective and to solicit feedback about the didactic and practicum portions of the course. The first part of the survey (Table 2) contained 16 items to assess the students' knowledge of, and attitudes toward handing off patient care, along with their comfort with the handoff process. Responses to this section were scored using a 5‐point Likert scale with 1 indicating strongly disagree and 5 indicating strongly agree. This part of the survey was administered both prior to and after the Selective.
| Competency | Selective | |
|---|---|---|
| Before | After | |
| ||
| I know how to hand off patients | 2.3 0.8 | 4.2 0.6* |
| I know how to make contingency plans for my patients | 2.1 0.8 | 3.9 0.7* |
| I know what a read‐back is | 2.3 1.3 | 4.4 0.9* |
| I know how to perform a read‐back | 2.0 1.2 | 4.2 0.9* |
| I know when to perform a read‐back | 1.6 0.8 | 4.1 1.0* |
| I am efficient at communicating patient information | 2.2 0.9 | 3.6 0.7* |
| I am effective at communicating patient information | 2.2 0.8 | 3.8 0.6* |
| I know a standard written structure for handoffs | 2.1 1.1 | 4.4 0.6* |
| I know a standard verbal structure for handoffs | 2.0 1.1 | 4.2 0.6* |
| I can choose appropriate modes of communication | 2.7 1.1 | 4.4 0.6* |
| I can choose appropriate vehicles of communication | 2.6 1.1 | 4.5 0.6* |
| I can choose appropriate settings for communication | 2.9 1.1 | 4.4 0.6* |
| Handoffs are well taught in my medical school | 1.6 0.8 | 3.5 1.0* |
| Standardization is important in handoffs | 4.3 0.9 | 4.6 0.5 |
| Handoffs are safer with attending supervision | 3.7 1.0 | 3.9 0.8 |
| I feel comfortable cross‐covering on patients | 1.6 0.7 | 3.0 1.0* |
The second part (Table 3) contained 12 items and was designed to evaluate the perceived usefulness of the different components of the class. This section was only administered at the end of the Selective. It utilized a 4‐point Likert scale with 1 indicating that the component was not useful at all, and 4 indicating that it was extremely useful. The first 6 items of the second section allowed students to evaluate the didactic portion of the handoff. The second 6 items allowed students to evaluate the practicum. Responses to all 12 items were then combined to determine an overall composite usefulness for the Selective.
| Useful [n (%)] | |
|---|---|
| |
| Overall composite usefulness | 578 (92) |
| Didactic composite usefulness | 254 (84) |
| Using fumble video clips for discussing handoffs | 32 (64)* |
| Discussion of modes of communication | 46 (88) |
| Discussion of vehicles of communication | 46 (88) |
| Discussion of settings of communication | 48 (96) |
| Choosing handoff structures for nonhealthcare handoffs | 37 (71)* |
| Discussing handoffs in industries outside of healthcare | 45 (94) |
| Practicum composite usefulness | 324(100) |
| Role playing | 54 (100) |
| Patient handoff scenarios | 54 (100) |
| Completing computerized templates | 54 (100) |
| Delivering handoffs to peer | 54 (100) |
| Receiving handoffs from peer | 54 (100) |
| Cross‐cover questions and discussion | 54 (100) |
The Selective was also evaluated qualitatively through the use of open‐ended, written comments that were solicited at the end of the survey. All surveys were administered anonymously.
Data Analysis
Student paired t test was used to compare continuous variables recorded before and after the Selective. A chi‐square test was used to assess the students' perception of the usefulness of the didactic vs. the practicum methods of teaching handoffs.
All analyses were performed using SAS (version 8.1; SAS Institute, Inc., Cary, NC). Bonferroni corrections were used for multiple comparisons such that P values of <0.003 and <0.004 were considered to be significant for continuous and categorical variables, respectively. All data are reported as mean standard deviation (SD).
The survey was approved by our local Institutional Review Board.
Results
More students chose the Selective than we had capacity to accommodate (60 of a class of 150). The pre‐ and postcourse survey response rate was 56 of 60 (93%) and 58 of 60 (97%), respectively. After the Selective, the mean score in response to whether handoffs are well taught in medical school increased from 1.6 to 3.5 (P < 0.003). Our students' self‐perceived skills and knowledge about handoffs improved after the Selective (Table 2). The greatest changes in perceived knowledge occurred in questions regarding the what, how, and when of read‐backs, and the knowledge of standard verbal and written handoff structures. The responses to the survey elements which assessed our students' attitudes regarding the importance of standardization and whether they felt handoffs were safer with faculty supervision did not change after the Selective (Table 2).
A total of 92% of the students felt that the course was extremely useful or useful. The role‐playing activity was thought to be more helpful than the didactic, but 84% of the students still rated the didactic portion as useful or extremely useful (Table 3). The element which was the least well received in the didactic portion was the use of video clips to demonstrate successful and unsuccessful (fumbled) college football handoffs, although the majority (64%) of students still found it useful.
The major theme generated from the comments section of the survey was that the Selective should be a required course.
Discussion
We know of no previously published literature that has addressed teaching handoffs to medical students. Horwitz et al.15 developed a sign‐out curriculum for Internal Medicine residents and found that none of their house‐staff had any previous training in handoffs during medical school, consistent with the finding that only 8% of U.S. medical schools provided formal instruction on handoffs.3 Prior to taking the Selective, our students had no knowledge of verbal or written templates for patient handoffs, although both before and after the course they felt that standardization was an important component of the process.
A number of verbal structures for handing off patient care have been described in the literature and there is not a consensus as to which functions best. Perhaps the most cited verbal communication format is SBAR (ie, situation, background, assessment and recommendation).16, 17 This tool was developed by Leonard et al.18 specifically for use by nurses to provide 1‐way communication to physicians pertaining to a change in patient status. We considered teaching the SBAR approach to the students but felt that it did not provide a suitable structure for handoffs because the transfer of care is not generally an event‐based situation and the literature on handoffs indicates that an optimal verbal system includes 2‐way communication.
Additional mnemonics for handoffs found in the literature include SIGNOUT (ie, Sick or DNR, Identifying information, General hospital course, New events of the day, Overall health status, Upcoming possibilities with plan, and Tasks to complete),14 I PASS the BATON (ie, Introduction, Patient, Assessment, Situation, Safety, Background, Actions, Timing, Ownership, Next)19 and the SAIF‐IR system (see boxed text).14
Verbal Structure for Patient Handoffs: SAIF‐IR
Off‐going provider performs a SAIF handoff:
Summary statement(s)
Active issues
If‐then contingency planning
Follow‐up activities
On‐coming provider makes the handoff SAIF‐IR:
Interactive questioning
Read‐backs
We developed the SAIF‐IR mnemonic to maximize efficiency and effectiveness while differentiating the verbal portion of the handoff from the written and incorporating 2‐way communication into its structure. In the Summary statement, we emphasize that this is not a history of present illness. We ask our students to summarize, in 1 to 3 sentences, the patient's presentation and working diagnosis. When discussing patient issues, we ask our students to only verbalize Active issues, although the written template has inactive, chronic issues listed. Here, we also ask our students to express their level of concern for the active issues and patient in general. If‐then's and Follow‐ups are usually verbalized together. Based on the offgoing provider's knowledge of the patient, we encourage the offgoing provider to anticipate potential problems and advise the oncoming provider on potential responses. Much of this advice is difficult to express in the written format and thus may not be found on the written handoff when the verbal handoff occurs. We encourage oncoming providers to take notes on the preprinted handoff sheet as part of the handoff process.
Through Interactive questioning and Read‐backs, we train our students and house‐staff to use the active listening techniques used outside of healthcare, in settings such as nuclear power plants and National Aeronautics and Space Administration mission control, where poor handoff communication may also result in safety concerns and adverse events.20 Interactive questioning allows the oncoming provider to correct or clarify any information given by the off‐going provider. Read‐backs are a method of confirming follow‐up activity or contingency plans. Together, the SAIF‐IR mnemonic builds a 2‐way communication structure into the patient handoff with both offgoing and oncoming providers having predefined roles.
Much of the information on our written handoff (patient identifying information, medications, language preference, code status, admission date) is not verbalized unless it is part of the active issues or the if‐then, follow‐ups (ie, medication titration for a patient admitted with an acute coronary syndrome or cor status in a patient newly made comfort care). By not reading extraneous information, we seek to emphasize the Active issues as well as the If‐then, Follow‐ups. We feel this emphasis maximizes the effectiveness of the handoff, while the purposeful nonverbalization of written materials such as identifying information maximizes its efficiency. Future work may examine which verbal and written structures for patient handoffs most benefit patient care and workflow through standard communication.
While our students found the Handoff Selective to be useful and to improve their self‐perceived ability to perform handoffs, we were not able to determine whether our program affected downstream outcomes such as adverse events relating to failures in handoff communication. Additionally, since we only taught and evaluated our Selective at the University of Colorado Denver School of Medicine, the response of our students may not generalize to other medical schools. Multicentered, prospective, randomized controlled trials may determine whether handoff education programs are successful in reducing patient adverse events related to transfers of care.
While handoffs occur frequently and are increasingly recognized as a vulnerable time in patient care, little is known about how to effectively teach handoffs to medical students during their clinical years. We developed a formal course to teach the importance of handoffs and how the process should be conducted. Our students reported that the Handoff Selective we developed improved their knowledge about the process and their perception of their ability to perform handoffs in a time‐appropriate and effective manner. In response to the feedback we received from our students, the Handoff Selective is the only course in the ICC that has been made mandatory for all students.
- , , .Communication failures: an insidious contributor to medical mishaps.Acad Med.2004;79:186–194.
- Root causes of sentinel events. The Joint Commission. Available at: http://www.jointcommission.org/NR/rdonlyres/FA465646‐5F5F‐4543‐AC8F‐E8AF6571E372/0/root_cause_se.jpg Accessed October2009.
- , , , et al.Lost in translation: challenges‐to‐physician communication during patient handoffs.Acad Med.2005;80:1094–1099.
- Institute of Medicine.Resident Duty Hours: Enhancing Sleep, Supervision and Safety.Washington, DC:National Academies Press;2008.
- ACGME duty hours. Accreditation Council for Graduate Medical Education. http://www.acgme.org/acWebsite/dutyHours/dh_ComProgrRequirmentsDutyHours0707.pdf. Accessed October2009.
- , , , et al.Mortality among hospitalized Medicare beneficiaries in the first 2 years following ACGME duty hour reform.JAMA.2007;298(9):975–983.
- , , , et al.Mortality among patient in VA hospitals in the first 2 years following ACGME duty hour reform.JAMA.2007;298(9):984–992.
- , , , .Changes in outcomes for internal medicine patients after work‐hour regulations.Ann Intern Med.2007;147(2):1–7.
- , , , et al.Transfers of patient care between house staff on internal medicine wards.Arch Intern Med.2006;166:1173–1177.
- , , , .Medical errors involving trainees.Arch Intern Med.2007;167(19):2030–2036.
- .Reducing resident work hours: unproven assumptions and unforeseen outcomes.Ann Intern Med.2006;140:814–815.
- , , et al.Consequences of inadequate sign‐out for patient care.Arch Intern Med.2008;168(16):1755–1760.
- JCAHO Handoff Communication. National patient safety goal. The Joint Commission. http://www.jointcommission.org/GeneralPublic/NPSG/07_npsgs.htm. Accessed October2009.
- , , , et al.A structured handoff program for interns.Acad Med.2009;84:347–352.
- , , .Development and implementation of an oral sign out skills curriculum.J Gen Intern Med.2007;22(10):1470–1474.
- , , , et al.Managing discontinuity in academic medical centers: strategies for a safe and effective sign out.J Hosp Med.2006;1:257–266.
- , , , .A theoretical framework and competency based approach to improving handoffs.Qual Saf Health Care.2008;17:11–14.
- , , .The human factor: the critical importance of effective teamwork in providing safe care.Qual Saf Health Care.2004;13(suppl 1):i85–i90.
- University HealthSystem Consortium Best Practice Recommendation: Patient Handoff Communication. White Paper. May 2006.Oak Brook, IL:University HealthSystem Consortium;2006.
- , , , , .Handoff strategies in settings with high consequences for failure: lessons for health care operations.Int J Qual Health Care.2004;16(2):125–132.
Communication failures are well‐recognized as causes of medical errors.1, 2 Specifically, handoffs of patient care responsibilities, which are increasingly prevalent in academic medical centers,3 have been cited as the most frequent cause of teamwork breakdown resulting in the harmful medical errors found in malpractice claims.1 The Institute of Medicine has recently identified patient handoffs as the moment where patient care errors are most likely to occur.4 A survey of 125 U.S. medical schools, however, found that only 8% specifically taught students how to hand off patient care.3
In July 2003, the American Council of Graduate Medical Education (ACGME) mandated that residency programs decrease resident work hours to improve patient care and safety by reducing fatigue,5 and a recent Institute of Medicine report suggests that they be decreased even further.4 Studies examining outcomes during the first 2 years after reducing duty hours did not find reductions in risk‐adjusted mortality.68 One proposed explanation for this lack of improvement is that the reduction in fatigue‐related medical errors is being offset by discontinuity of care with due to the increased number of patient handoffs resulting from shortened duty hours,911 one recent study found that omission of key information during patient sign outs frequently resulted in adverse patient care outcomes.12
In 2007, the Joint Commission developed a new National Patient Safety Goal that requires organizations to improve communication between caregivers.13 We recently developed an approach by which Internal Medicine residents hand off patient care using a structured process, written and verbal templates, formal training about handoffs, and direct attending supervision.14 Because fourth‐year medical students perform the duties of interns when working as subinterns, we recognized that education about handoffs should occur prior to the time students became interns. Accordingly, we developed a course designed to teach patient handoffs to medical students at the transition between their third and fourth years of training.
Setting
The Handoff Selective was developed by faculty of Denver Health and the University of Colorado Denver School of Medicine.
Program Description
The Selective was first offered in April 2007 as part of an Integrated Clinician's Course (ICC), a 2‐week course for students beginning their fourth year, which starts in April at the University of Colorado. The ICC includes both mandatory and selective sessions that are focused on developing clinical skills and preparing them for their subinternships. The Handoff Selective was conducted in a computerized teaching laboratory, lasted a total of 2 hours and consisted of 2 parts. Each of the 5 Denver Health Hospital Medicine faculty members versed in handoff education taught 2 sessions of 6 to 8 students.
Part 1: Didactic
During the first hour of class, the faculty presented a lecture that summarized the relevant literature on handoffs and explained the importance of the topic. The objectives of the didactic were to: (1) understand the importance of handoffs; (2) explore different communication elements and structures; (3) gain exposure to handoffs outside of healthcare; and (4) learn a structure for handoffs of patient care in hospitalized patients.
We used 3 video clips of handoffs from 2 football games to demonstrate the importance of practice, training, and 2‐way communications in handoffs. The first video clip showed a runner trying to make a spontaneous handoff while being tackled. The receiver was not expecting the handoff and was preoccupied with blocking another player. This attempted handoff resulted in a fumble, which we related to an adverse patient event.
The next 2 video clips showed 2 complex, seldom used, but well‐known football handoffsthe hook and lateral and the Statue of Liberty. Both handoffs were successfully executed presumably as a result of education, practice and the active participation of both players (handing off and receiving) in the process. We then related the teaching and practicing of complex communication to the Joint Commission on Accreditation of Healthcare Organizations (JCAHO; now simply the Joint Commission) data suggesting that most sentinel events have their root cause in communication and training failures.2
Basic communication elements and process structures were then explored using scenarios from everyday life and evidence from fields outside of medicine. We emphasized that structures for communication (modes, vehicles, and settings) must be chosen according to the occasion and that handoffs are common and important in all occupations. In discussing modes (verbal, written, or nonverbal), vehicles (paper, telephone, or e‐mail), and settings (face‐to face, virtual, or disconnected), we emphasized that the most effective structures for communication (verbal, face‐to face meetings, with written materials and other visual aids at the patient's bedside) were also the most time‐consuming (Figure 1). While our standard for resident handoffs is a face‐to‐face verbal interaction with preprinted written materials as an aid, we also emphasized that for complex patients (eg, mental status changes, concern for an acute abdomen) more robust communication is often needed. Accordingly, a more time‐consuming bedside handoff with simultaneous, focused physical exam and history‐taking by both oncoming and off‐going providers may be most appropriate.

As real‐life examples, we asked our students to communicate a happy birthday wish to their mother, who lives in another state. Almost uniformly, in addition to a written aid (birthday card), they choose the telephone as a vehicle for their verbal mode in a virtual setting with 2‐way communication possible. In contrast, when asked to propose marriage to a significant other in another state, students felt that a face‐to‐face meeting with verbal and nonverbal (ie, ring) modes was appropriate. This time‐consuming mode of communication was felt to be necessary to create a sentiment of importance and avert any possible miscommunication.
The didactic session concluded by demonstrating how to use standardized written and verbal templates for handoffs of the care of a hospitalized patient. We explore the differentiation between written and verbal handoffs in our discussion below.
Part 2: Practicum
The second hour was devoted to practicing handoffs as a group. The faculty developed 6 case scenarios that differed with respect to diagnosis, length of stay, active medical issues, and anticipated discharge (Table 1). The scenarios included extensive admission information as well as evolving issues for each patient that were specific to the day of the intended handoff. Students were given Microsoft Word table‐based handoff templates to use when creating written sign‐outs for their patients. Verbal handoffs were performed between students and sign‐outs were exchanged. The faculty then role‐played cross‐cover calls that were specific for each scenario to test the students' inclusion of integral information in their handoffs and their ability to create contingency plans.
| Diagnosis | LOS | Active Issues | Cross‐Cover |
|---|---|---|---|
| |||
| CP | 1 | CP, HTN, DM | CP, HTN, headache |
| GIB | 1 | GIB, alcohol withdrawal | Poor response to red call transfusion, coagulopathy |
| Acute pancreatitis | 2 | Pain, possible pancreatic abscess | Fever, agitation, hypoxia |
| CHF | 2 | CHF, DM, nausea | Lack of diuresis, CP, hypoglycemia |
| Acute kidney injury | 3 | None, ready for discharge | HTN, hyperglycemia |
| Community acquired pneumonia | 3 | Anxiety, discharge pending | Confusion, emesis with hypoxia |
Program Evaluation
We developed a 2‐part survey to evaluate the effectiveness of the Selective and to solicit feedback about the didactic and practicum portions of the course. The first part of the survey (Table 2) contained 16 items to assess the students' knowledge of, and attitudes toward handing off patient care, along with their comfort with the handoff process. Responses to this section were scored using a 5‐point Likert scale with 1 indicating strongly disagree and 5 indicating strongly agree. This part of the survey was administered both prior to and after the Selective.
| Competency | Selective | |
|---|---|---|
| Before | After | |
| ||
| I know how to hand off patients | 2.3 0.8 | 4.2 0.6* |
| I know how to make contingency plans for my patients | 2.1 0.8 | 3.9 0.7* |
| I know what a read‐back is | 2.3 1.3 | 4.4 0.9* |
| I know how to perform a read‐back | 2.0 1.2 | 4.2 0.9* |
| I know when to perform a read‐back | 1.6 0.8 | 4.1 1.0* |
| I am efficient at communicating patient information | 2.2 0.9 | 3.6 0.7* |
| I am effective at communicating patient information | 2.2 0.8 | 3.8 0.6* |
| I know a standard written structure for handoffs | 2.1 1.1 | 4.4 0.6* |
| I know a standard verbal structure for handoffs | 2.0 1.1 | 4.2 0.6* |
| I can choose appropriate modes of communication | 2.7 1.1 | 4.4 0.6* |
| I can choose appropriate vehicles of communication | 2.6 1.1 | 4.5 0.6* |
| I can choose appropriate settings for communication | 2.9 1.1 | 4.4 0.6* |
| Handoffs are well taught in my medical school | 1.6 0.8 | 3.5 1.0* |
| Standardization is important in handoffs | 4.3 0.9 | 4.6 0.5 |
| Handoffs are safer with attending supervision | 3.7 1.0 | 3.9 0.8 |
| I feel comfortable cross‐covering on patients | 1.6 0.7 | 3.0 1.0* |
The second part (Table 3) contained 12 items and was designed to evaluate the perceived usefulness of the different components of the class. This section was only administered at the end of the Selective. It utilized a 4‐point Likert scale with 1 indicating that the component was not useful at all, and 4 indicating that it was extremely useful. The first 6 items of the second section allowed students to evaluate the didactic portion of the handoff. The second 6 items allowed students to evaluate the practicum. Responses to all 12 items were then combined to determine an overall composite usefulness for the Selective.
| Useful [n (%)] | |
|---|---|
| |
| Overall composite usefulness | 578 (92) |
| Didactic composite usefulness | 254 (84) |
| Using fumble video clips for discussing handoffs | 32 (64)* |
| Discussion of modes of communication | 46 (88) |
| Discussion of vehicles of communication | 46 (88) |
| Discussion of settings of communication | 48 (96) |
| Choosing handoff structures for nonhealthcare handoffs | 37 (71)* |
| Discussing handoffs in industries outside of healthcare | 45 (94) |
| Practicum composite usefulness | 324(100) |
| Role playing | 54 (100) |
| Patient handoff scenarios | 54 (100) |
| Completing computerized templates | 54 (100) |
| Delivering handoffs to peer | 54 (100) |
| Receiving handoffs from peer | 54 (100) |
| Cross‐cover questions and discussion | 54 (100) |
The Selective was also evaluated qualitatively through the use of open‐ended, written comments that were solicited at the end of the survey. All surveys were administered anonymously.
Data Analysis
Student paired t test was used to compare continuous variables recorded before and after the Selective. A chi‐square test was used to assess the students' perception of the usefulness of the didactic vs. the practicum methods of teaching handoffs.
All analyses were performed using SAS (version 8.1; SAS Institute, Inc., Cary, NC). Bonferroni corrections were used for multiple comparisons such that P values of <0.003 and <0.004 were considered to be significant for continuous and categorical variables, respectively. All data are reported as mean standard deviation (SD).
The survey was approved by our local Institutional Review Board.
Results
More students chose the Selective than we had capacity to accommodate (60 of a class of 150). The pre‐ and postcourse survey response rate was 56 of 60 (93%) and 58 of 60 (97%), respectively. After the Selective, the mean score in response to whether handoffs are well taught in medical school increased from 1.6 to 3.5 (P < 0.003). Our students' self‐perceived skills and knowledge about handoffs improved after the Selective (Table 2). The greatest changes in perceived knowledge occurred in questions regarding the what, how, and when of read‐backs, and the knowledge of standard verbal and written handoff structures. The responses to the survey elements which assessed our students' attitudes regarding the importance of standardization and whether they felt handoffs were safer with faculty supervision did not change after the Selective (Table 2).
A total of 92% of the students felt that the course was extremely useful or useful. The role‐playing activity was thought to be more helpful than the didactic, but 84% of the students still rated the didactic portion as useful or extremely useful (Table 3). The element which was the least well received in the didactic portion was the use of video clips to demonstrate successful and unsuccessful (fumbled) college football handoffs, although the majority (64%) of students still found it useful.
The major theme generated from the comments section of the survey was that the Selective should be a required course.
Discussion
We know of no previously published literature that has addressed teaching handoffs to medical students. Horwitz et al.15 developed a sign‐out curriculum for Internal Medicine residents and found that none of their house‐staff had any previous training in handoffs during medical school, consistent with the finding that only 8% of U.S. medical schools provided formal instruction on handoffs.3 Prior to taking the Selective, our students had no knowledge of verbal or written templates for patient handoffs, although both before and after the course they felt that standardization was an important component of the process.
A number of verbal structures for handing off patient care have been described in the literature and there is not a consensus as to which functions best. Perhaps the most cited verbal communication format is SBAR (ie, situation, background, assessment and recommendation).16, 17 This tool was developed by Leonard et al.18 specifically for use by nurses to provide 1‐way communication to physicians pertaining to a change in patient status. We considered teaching the SBAR approach to the students but felt that it did not provide a suitable structure for handoffs because the transfer of care is not generally an event‐based situation and the literature on handoffs indicates that an optimal verbal system includes 2‐way communication.
Additional mnemonics for handoffs found in the literature include SIGNOUT (ie, Sick or DNR, Identifying information, General hospital course, New events of the day, Overall health status, Upcoming possibilities with plan, and Tasks to complete),14 I PASS the BATON (ie, Introduction, Patient, Assessment, Situation, Safety, Background, Actions, Timing, Ownership, Next)19 and the SAIF‐IR system (see boxed text).14
Verbal Structure for Patient Handoffs: SAIF‐IR
Off‐going provider performs a SAIF handoff:
Summary statement(s)
Active issues
If‐then contingency planning
Follow‐up activities
On‐coming provider makes the handoff SAIF‐IR:
Interactive questioning
Read‐backs
We developed the SAIF‐IR mnemonic to maximize efficiency and effectiveness while differentiating the verbal portion of the handoff from the written and incorporating 2‐way communication into its structure. In the Summary statement, we emphasize that this is not a history of present illness. We ask our students to summarize, in 1 to 3 sentences, the patient's presentation and working diagnosis. When discussing patient issues, we ask our students to only verbalize Active issues, although the written template has inactive, chronic issues listed. Here, we also ask our students to express their level of concern for the active issues and patient in general. If‐then's and Follow‐ups are usually verbalized together. Based on the offgoing provider's knowledge of the patient, we encourage the offgoing provider to anticipate potential problems and advise the oncoming provider on potential responses. Much of this advice is difficult to express in the written format and thus may not be found on the written handoff when the verbal handoff occurs. We encourage oncoming providers to take notes on the preprinted handoff sheet as part of the handoff process.
Through Interactive questioning and Read‐backs, we train our students and house‐staff to use the active listening techniques used outside of healthcare, in settings such as nuclear power plants and National Aeronautics and Space Administration mission control, where poor handoff communication may also result in safety concerns and adverse events.20 Interactive questioning allows the oncoming provider to correct or clarify any information given by the off‐going provider. Read‐backs are a method of confirming follow‐up activity or contingency plans. Together, the SAIF‐IR mnemonic builds a 2‐way communication structure into the patient handoff with both offgoing and oncoming providers having predefined roles.
Much of the information on our written handoff (patient identifying information, medications, language preference, code status, admission date) is not verbalized unless it is part of the active issues or the if‐then, follow‐ups (ie, medication titration for a patient admitted with an acute coronary syndrome or cor status in a patient newly made comfort care). By not reading extraneous information, we seek to emphasize the Active issues as well as the If‐then, Follow‐ups. We feel this emphasis maximizes the effectiveness of the handoff, while the purposeful nonverbalization of written materials such as identifying information maximizes its efficiency. Future work may examine which verbal and written structures for patient handoffs most benefit patient care and workflow through standard communication.
While our students found the Handoff Selective to be useful and to improve their self‐perceived ability to perform handoffs, we were not able to determine whether our program affected downstream outcomes such as adverse events relating to failures in handoff communication. Additionally, since we only taught and evaluated our Selective at the University of Colorado Denver School of Medicine, the response of our students may not generalize to other medical schools. Multicentered, prospective, randomized controlled trials may determine whether handoff education programs are successful in reducing patient adverse events related to transfers of care.
While handoffs occur frequently and are increasingly recognized as a vulnerable time in patient care, little is known about how to effectively teach handoffs to medical students during their clinical years. We developed a formal course to teach the importance of handoffs and how the process should be conducted. Our students reported that the Handoff Selective we developed improved their knowledge about the process and their perception of their ability to perform handoffs in a time‐appropriate and effective manner. In response to the feedback we received from our students, the Handoff Selective is the only course in the ICC that has been made mandatory for all students.
Communication failures are well‐recognized as causes of medical errors.1, 2 Specifically, handoffs of patient care responsibilities, which are increasingly prevalent in academic medical centers,3 have been cited as the most frequent cause of teamwork breakdown resulting in the harmful medical errors found in malpractice claims.1 The Institute of Medicine has recently identified patient handoffs as the moment where patient care errors are most likely to occur.4 A survey of 125 U.S. medical schools, however, found that only 8% specifically taught students how to hand off patient care.3
In July 2003, the American Council of Graduate Medical Education (ACGME) mandated that residency programs decrease resident work hours to improve patient care and safety by reducing fatigue,5 and a recent Institute of Medicine report suggests that they be decreased even further.4 Studies examining outcomes during the first 2 years after reducing duty hours did not find reductions in risk‐adjusted mortality.68 One proposed explanation for this lack of improvement is that the reduction in fatigue‐related medical errors is being offset by discontinuity of care with due to the increased number of patient handoffs resulting from shortened duty hours,911 one recent study found that omission of key information during patient sign outs frequently resulted in adverse patient care outcomes.12
In 2007, the Joint Commission developed a new National Patient Safety Goal that requires organizations to improve communication between caregivers.13 We recently developed an approach by which Internal Medicine residents hand off patient care using a structured process, written and verbal templates, formal training about handoffs, and direct attending supervision.14 Because fourth‐year medical students perform the duties of interns when working as subinterns, we recognized that education about handoffs should occur prior to the time students became interns. Accordingly, we developed a course designed to teach patient handoffs to medical students at the transition between their third and fourth years of training.
Setting
The Handoff Selective was developed by faculty of Denver Health and the University of Colorado Denver School of Medicine.
Program Description
The Selective was first offered in April 2007 as part of an Integrated Clinician's Course (ICC), a 2‐week course for students beginning their fourth year, which starts in April at the University of Colorado. The ICC includes both mandatory and selective sessions that are focused on developing clinical skills and preparing them for their subinternships. The Handoff Selective was conducted in a computerized teaching laboratory, lasted a total of 2 hours and consisted of 2 parts. Each of the 5 Denver Health Hospital Medicine faculty members versed in handoff education taught 2 sessions of 6 to 8 students.
Part 1: Didactic
During the first hour of class, the faculty presented a lecture that summarized the relevant literature on handoffs and explained the importance of the topic. The objectives of the didactic were to: (1) understand the importance of handoffs; (2) explore different communication elements and structures; (3) gain exposure to handoffs outside of healthcare; and (4) learn a structure for handoffs of patient care in hospitalized patients.
We used 3 video clips of handoffs from 2 football games to demonstrate the importance of practice, training, and 2‐way communications in handoffs. The first video clip showed a runner trying to make a spontaneous handoff while being tackled. The receiver was not expecting the handoff and was preoccupied with blocking another player. This attempted handoff resulted in a fumble, which we related to an adverse patient event.
The next 2 video clips showed 2 complex, seldom used, but well‐known football handoffsthe hook and lateral and the Statue of Liberty. Both handoffs were successfully executed presumably as a result of education, practice and the active participation of both players (handing off and receiving) in the process. We then related the teaching and practicing of complex communication to the Joint Commission on Accreditation of Healthcare Organizations (JCAHO; now simply the Joint Commission) data suggesting that most sentinel events have their root cause in communication and training failures.2
Basic communication elements and process structures were then explored using scenarios from everyday life and evidence from fields outside of medicine. We emphasized that structures for communication (modes, vehicles, and settings) must be chosen according to the occasion and that handoffs are common and important in all occupations. In discussing modes (verbal, written, or nonverbal), vehicles (paper, telephone, or e‐mail), and settings (face‐to face, virtual, or disconnected), we emphasized that the most effective structures for communication (verbal, face‐to face meetings, with written materials and other visual aids at the patient's bedside) were also the most time‐consuming (Figure 1). While our standard for resident handoffs is a face‐to‐face verbal interaction with preprinted written materials as an aid, we also emphasized that for complex patients (eg, mental status changes, concern for an acute abdomen) more robust communication is often needed. Accordingly, a more time‐consuming bedside handoff with simultaneous, focused physical exam and history‐taking by both oncoming and off‐going providers may be most appropriate.

As real‐life examples, we asked our students to communicate a happy birthday wish to their mother, who lives in another state. Almost uniformly, in addition to a written aid (birthday card), they choose the telephone as a vehicle for their verbal mode in a virtual setting with 2‐way communication possible. In contrast, when asked to propose marriage to a significant other in another state, students felt that a face‐to‐face meeting with verbal and nonverbal (ie, ring) modes was appropriate. This time‐consuming mode of communication was felt to be necessary to create a sentiment of importance and avert any possible miscommunication.
The didactic session concluded by demonstrating how to use standardized written and verbal templates for handoffs of the care of a hospitalized patient. We explore the differentiation between written and verbal handoffs in our discussion below.
Part 2: Practicum
The second hour was devoted to practicing handoffs as a group. The faculty developed 6 case scenarios that differed with respect to diagnosis, length of stay, active medical issues, and anticipated discharge (Table 1). The scenarios included extensive admission information as well as evolving issues for each patient that were specific to the day of the intended handoff. Students were given Microsoft Word table‐based handoff templates to use when creating written sign‐outs for their patients. Verbal handoffs were performed between students and sign‐outs were exchanged. The faculty then role‐played cross‐cover calls that were specific for each scenario to test the students' inclusion of integral information in their handoffs and their ability to create contingency plans.
| Diagnosis | LOS | Active Issues | Cross‐Cover |
|---|---|---|---|
| |||
| CP | 1 | CP, HTN, DM | CP, HTN, headache |
| GIB | 1 | GIB, alcohol withdrawal | Poor response to red call transfusion, coagulopathy |
| Acute pancreatitis | 2 | Pain, possible pancreatic abscess | Fever, agitation, hypoxia |
| CHF | 2 | CHF, DM, nausea | Lack of diuresis, CP, hypoglycemia |
| Acute kidney injury | 3 | None, ready for discharge | HTN, hyperglycemia |
| Community acquired pneumonia | 3 | Anxiety, discharge pending | Confusion, emesis with hypoxia |
Program Evaluation
We developed a 2‐part survey to evaluate the effectiveness of the Selective and to solicit feedback about the didactic and practicum portions of the course. The first part of the survey (Table 2) contained 16 items to assess the students' knowledge of, and attitudes toward handing off patient care, along with their comfort with the handoff process. Responses to this section were scored using a 5‐point Likert scale with 1 indicating strongly disagree and 5 indicating strongly agree. This part of the survey was administered both prior to and after the Selective.
| Competency | Selective | |
|---|---|---|
| Before | After | |
| ||
| I know how to hand off patients | 2.3 0.8 | 4.2 0.6* |
| I know how to make contingency plans for my patients | 2.1 0.8 | 3.9 0.7* |
| I know what a read‐back is | 2.3 1.3 | 4.4 0.9* |
| I know how to perform a read‐back | 2.0 1.2 | 4.2 0.9* |
| I know when to perform a read‐back | 1.6 0.8 | 4.1 1.0* |
| I am efficient at communicating patient information | 2.2 0.9 | 3.6 0.7* |
| I am effective at communicating patient information | 2.2 0.8 | 3.8 0.6* |
| I know a standard written structure for handoffs | 2.1 1.1 | 4.4 0.6* |
| I know a standard verbal structure for handoffs | 2.0 1.1 | 4.2 0.6* |
| I can choose appropriate modes of communication | 2.7 1.1 | 4.4 0.6* |
| I can choose appropriate vehicles of communication | 2.6 1.1 | 4.5 0.6* |
| I can choose appropriate settings for communication | 2.9 1.1 | 4.4 0.6* |
| Handoffs are well taught in my medical school | 1.6 0.8 | 3.5 1.0* |
| Standardization is important in handoffs | 4.3 0.9 | 4.6 0.5 |
| Handoffs are safer with attending supervision | 3.7 1.0 | 3.9 0.8 |
| I feel comfortable cross‐covering on patients | 1.6 0.7 | 3.0 1.0* |
The second part (Table 3) contained 12 items and was designed to evaluate the perceived usefulness of the different components of the class. This section was only administered at the end of the Selective. It utilized a 4‐point Likert scale with 1 indicating that the component was not useful at all, and 4 indicating that it was extremely useful. The first 6 items of the second section allowed students to evaluate the didactic portion of the handoff. The second 6 items allowed students to evaluate the practicum. Responses to all 12 items were then combined to determine an overall composite usefulness for the Selective.
| Useful [n (%)] | |
|---|---|
| |
| Overall composite usefulness | 578 (92) |
| Didactic composite usefulness | 254 (84) |
| Using fumble video clips for discussing handoffs | 32 (64)* |
| Discussion of modes of communication | 46 (88) |
| Discussion of vehicles of communication | 46 (88) |
| Discussion of settings of communication | 48 (96) |
| Choosing handoff structures for nonhealthcare handoffs | 37 (71)* |
| Discussing handoffs in industries outside of healthcare | 45 (94) |
| Practicum composite usefulness | 324(100) |
| Role playing | 54 (100) |
| Patient handoff scenarios | 54 (100) |
| Completing computerized templates | 54 (100) |
| Delivering handoffs to peer | 54 (100) |
| Receiving handoffs from peer | 54 (100) |
| Cross‐cover questions and discussion | 54 (100) |
The Selective was also evaluated qualitatively through the use of open‐ended, written comments that were solicited at the end of the survey. All surveys were administered anonymously.
Data Analysis
Student paired t test was used to compare continuous variables recorded before and after the Selective. A chi‐square test was used to assess the students' perception of the usefulness of the didactic vs. the practicum methods of teaching handoffs.
All analyses were performed using SAS (version 8.1; SAS Institute, Inc., Cary, NC). Bonferroni corrections were used for multiple comparisons such that P values of <0.003 and <0.004 were considered to be significant for continuous and categorical variables, respectively. All data are reported as mean standard deviation (SD).
The survey was approved by our local Institutional Review Board.
Results
More students chose the Selective than we had capacity to accommodate (60 of a class of 150). The pre‐ and postcourse survey response rate was 56 of 60 (93%) and 58 of 60 (97%), respectively. After the Selective, the mean score in response to whether handoffs are well taught in medical school increased from 1.6 to 3.5 (P < 0.003). Our students' self‐perceived skills and knowledge about handoffs improved after the Selective (Table 2). The greatest changes in perceived knowledge occurred in questions regarding the what, how, and when of read‐backs, and the knowledge of standard verbal and written handoff structures. The responses to the survey elements which assessed our students' attitudes regarding the importance of standardization and whether they felt handoffs were safer with faculty supervision did not change after the Selective (Table 2).
A total of 92% of the students felt that the course was extremely useful or useful. The role‐playing activity was thought to be more helpful than the didactic, but 84% of the students still rated the didactic portion as useful or extremely useful (Table 3). The element which was the least well received in the didactic portion was the use of video clips to demonstrate successful and unsuccessful (fumbled) college football handoffs, although the majority (64%) of students still found it useful.
The major theme generated from the comments section of the survey was that the Selective should be a required course.
Discussion
We know of no previously published literature that has addressed teaching handoffs to medical students. Horwitz et al.15 developed a sign‐out curriculum for Internal Medicine residents and found that none of their house‐staff had any previous training in handoffs during medical school, consistent with the finding that only 8% of U.S. medical schools provided formal instruction on handoffs.3 Prior to taking the Selective, our students had no knowledge of verbal or written templates for patient handoffs, although both before and after the course they felt that standardization was an important component of the process.
A number of verbal structures for handing off patient care have been described in the literature and there is not a consensus as to which functions best. Perhaps the most cited verbal communication format is SBAR (ie, situation, background, assessment and recommendation).16, 17 This tool was developed by Leonard et al.18 specifically for use by nurses to provide 1‐way communication to physicians pertaining to a change in patient status. We considered teaching the SBAR approach to the students but felt that it did not provide a suitable structure for handoffs because the transfer of care is not generally an event‐based situation and the literature on handoffs indicates that an optimal verbal system includes 2‐way communication.
Additional mnemonics for handoffs found in the literature include SIGNOUT (ie, Sick or DNR, Identifying information, General hospital course, New events of the day, Overall health status, Upcoming possibilities with plan, and Tasks to complete),14 I PASS the BATON (ie, Introduction, Patient, Assessment, Situation, Safety, Background, Actions, Timing, Ownership, Next)19 and the SAIF‐IR system (see boxed text).14
Verbal Structure for Patient Handoffs: SAIF‐IR
Off‐going provider performs a SAIF handoff:
Summary statement(s)
Active issues
If‐then contingency planning
Follow‐up activities
On‐coming provider makes the handoff SAIF‐IR:
Interactive questioning
Read‐backs
We developed the SAIF‐IR mnemonic to maximize efficiency and effectiveness while differentiating the verbal portion of the handoff from the written and incorporating 2‐way communication into its structure. In the Summary statement, we emphasize that this is not a history of present illness. We ask our students to summarize, in 1 to 3 sentences, the patient's presentation and working diagnosis. When discussing patient issues, we ask our students to only verbalize Active issues, although the written template has inactive, chronic issues listed. Here, we also ask our students to express their level of concern for the active issues and patient in general. If‐then's and Follow‐ups are usually verbalized together. Based on the offgoing provider's knowledge of the patient, we encourage the offgoing provider to anticipate potential problems and advise the oncoming provider on potential responses. Much of this advice is difficult to express in the written format and thus may not be found on the written handoff when the verbal handoff occurs. We encourage oncoming providers to take notes on the preprinted handoff sheet as part of the handoff process.
Through Interactive questioning and Read‐backs, we train our students and house‐staff to use the active listening techniques used outside of healthcare, in settings such as nuclear power plants and National Aeronautics and Space Administration mission control, where poor handoff communication may also result in safety concerns and adverse events.20 Interactive questioning allows the oncoming provider to correct or clarify any information given by the off‐going provider. Read‐backs are a method of confirming follow‐up activity or contingency plans. Together, the SAIF‐IR mnemonic builds a 2‐way communication structure into the patient handoff with both offgoing and oncoming providers having predefined roles.
Much of the information on our written handoff (patient identifying information, medications, language preference, code status, admission date) is not verbalized unless it is part of the active issues or the if‐then, follow‐ups (ie, medication titration for a patient admitted with an acute coronary syndrome or cor status in a patient newly made comfort care). By not reading extraneous information, we seek to emphasize the Active issues as well as the If‐then, Follow‐ups. We feel this emphasis maximizes the effectiveness of the handoff, while the purposeful nonverbalization of written materials such as identifying information maximizes its efficiency. Future work may examine which verbal and written structures for patient handoffs most benefit patient care and workflow through standard communication.
While our students found the Handoff Selective to be useful and to improve their self‐perceived ability to perform handoffs, we were not able to determine whether our program affected downstream outcomes such as adverse events relating to failures in handoff communication. Additionally, since we only taught and evaluated our Selective at the University of Colorado Denver School of Medicine, the response of our students may not generalize to other medical schools. Multicentered, prospective, randomized controlled trials may determine whether handoff education programs are successful in reducing patient adverse events related to transfers of care.
While handoffs occur frequently and are increasingly recognized as a vulnerable time in patient care, little is known about how to effectively teach handoffs to medical students during their clinical years. We developed a formal course to teach the importance of handoffs and how the process should be conducted. Our students reported that the Handoff Selective we developed improved their knowledge about the process and their perception of their ability to perform handoffs in a time‐appropriate and effective manner. In response to the feedback we received from our students, the Handoff Selective is the only course in the ICC that has been made mandatory for all students.
- , , .Communication failures: an insidious contributor to medical mishaps.Acad Med.2004;79:186–194.
- Root causes of sentinel events. The Joint Commission. Available at: http://www.jointcommission.org/NR/rdonlyres/FA465646‐5F5F‐4543‐AC8F‐E8AF6571E372/0/root_cause_se.jpg Accessed October2009.
- , , , et al.Lost in translation: challenges‐to‐physician communication during patient handoffs.Acad Med.2005;80:1094–1099.
- Institute of Medicine.Resident Duty Hours: Enhancing Sleep, Supervision and Safety.Washington, DC:National Academies Press;2008.
- ACGME duty hours. Accreditation Council for Graduate Medical Education. http://www.acgme.org/acWebsite/dutyHours/dh_ComProgrRequirmentsDutyHours0707.pdf. Accessed October2009.
- , , , et al.Mortality among hospitalized Medicare beneficiaries in the first 2 years following ACGME duty hour reform.JAMA.2007;298(9):975–983.
- , , , et al.Mortality among patient in VA hospitals in the first 2 years following ACGME duty hour reform.JAMA.2007;298(9):984–992.
- , , , .Changes in outcomes for internal medicine patients after work‐hour regulations.Ann Intern Med.2007;147(2):1–7.
- , , , et al.Transfers of patient care between house staff on internal medicine wards.Arch Intern Med.2006;166:1173–1177.
- , , , .Medical errors involving trainees.Arch Intern Med.2007;167(19):2030–2036.
- .Reducing resident work hours: unproven assumptions and unforeseen outcomes.Ann Intern Med.2006;140:814–815.
- , , et al.Consequences of inadequate sign‐out for patient care.Arch Intern Med.2008;168(16):1755–1760.
- JCAHO Handoff Communication. National patient safety goal. The Joint Commission. http://www.jointcommission.org/GeneralPublic/NPSG/07_npsgs.htm. Accessed October2009.
- , , , et al.A structured handoff program for interns.Acad Med.2009;84:347–352.
- , , .Development and implementation of an oral sign out skills curriculum.J Gen Intern Med.2007;22(10):1470–1474.
- , , , et al.Managing discontinuity in academic medical centers: strategies for a safe and effective sign out.J Hosp Med.2006;1:257–266.
- , , , .A theoretical framework and competency based approach to improving handoffs.Qual Saf Health Care.2008;17:11–14.
- , , .The human factor: the critical importance of effective teamwork in providing safe care.Qual Saf Health Care.2004;13(suppl 1):i85–i90.
- University HealthSystem Consortium Best Practice Recommendation: Patient Handoff Communication. White Paper. May 2006.Oak Brook, IL:University HealthSystem Consortium;2006.
- , , , , .Handoff strategies in settings with high consequences for failure: lessons for health care operations.Int J Qual Health Care.2004;16(2):125–132.
- , , .Communication failures: an insidious contributor to medical mishaps.Acad Med.2004;79:186–194.
- Root causes of sentinel events. The Joint Commission. Available at: http://www.jointcommission.org/NR/rdonlyres/FA465646‐5F5F‐4543‐AC8F‐E8AF6571E372/0/root_cause_se.jpg Accessed October2009.
- , , , et al.Lost in translation: challenges‐to‐physician communication during patient handoffs.Acad Med.2005;80:1094–1099.
- Institute of Medicine.Resident Duty Hours: Enhancing Sleep, Supervision and Safety.Washington, DC:National Academies Press;2008.
- ACGME duty hours. Accreditation Council for Graduate Medical Education. http://www.acgme.org/acWebsite/dutyHours/dh_ComProgrRequirmentsDutyHours0707.pdf. Accessed October2009.
- , , , et al.Mortality among hospitalized Medicare beneficiaries in the first 2 years following ACGME duty hour reform.JAMA.2007;298(9):975–983.
- , , , et al.Mortality among patient in VA hospitals in the first 2 years following ACGME duty hour reform.JAMA.2007;298(9):984–992.
- , , , .Changes in outcomes for internal medicine patients after work‐hour regulations.Ann Intern Med.2007;147(2):1–7.
- , , , et al.Transfers of patient care between house staff on internal medicine wards.Arch Intern Med.2006;166:1173–1177.
- , , , .Medical errors involving trainees.Arch Intern Med.2007;167(19):2030–2036.
- .Reducing resident work hours: unproven assumptions and unforeseen outcomes.Ann Intern Med.2006;140:814–815.
- , , et al.Consequences of inadequate sign‐out for patient care.Arch Intern Med.2008;168(16):1755–1760.
- JCAHO Handoff Communication. National patient safety goal. The Joint Commission. http://www.jointcommission.org/GeneralPublic/NPSG/07_npsgs.htm. Accessed October2009.
- , , , et al.A structured handoff program for interns.Acad Med.2009;84:347–352.
- , , .Development and implementation of an oral sign out skills curriculum.J Gen Intern Med.2007;22(10):1470–1474.
- , , , et al.Managing discontinuity in academic medical centers: strategies for a safe and effective sign out.J Hosp Med.2006;1:257–266.
- , , , .A theoretical framework and competency based approach to improving handoffs.Qual Saf Health Care.2008;17:11–14.
- , , .The human factor: the critical importance of effective teamwork in providing safe care.Qual Saf Health Care.2004;13(suppl 1):i85–i90.
- University HealthSystem Consortium Best Practice Recommendation: Patient Handoff Communication. White Paper. May 2006.Oak Brook, IL:University HealthSystem Consortium;2006.
- , , , , .Handoff strategies in settings with high consequences for failure: lessons for health care operations.Int J Qual Health Care.2004;16(2):125–132.
Copyright © 2010 Society of Hospital Medicine
Continuing Medical Education Program in
If you wish to receive credit for this activity, which begins on the next page, please refer to the website:
Accreditation and Designation Statement
Blackwell Futura Media Services designates this educational activity for a 1 AMA PRA Category 1 Credit. Physicians should only claim credit commensurate with the extent of their participation in the activity.
Blackwell Futura Media Services is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Educational Objectives
Continuous participation in the Journal of Hospital Medicine CME program will enable learners to be better able to:
-
Interpret clinical guidelines and their applications for higher quality and more efficient care for all hospitalized patients.
-
Describe the standard of care for common illnesses and conditions treated in the hospital; such as pneumonia, COPD exacerbation, acute coronary syndrome, HF exacerbation, glycemic control, venous thromboembolic disease, stroke, etc.
-
Discuss evidence‐based recommendations involving transitions of care, including the hospital discharge process.
-
Gain insights into the roles of hospitalists as medical educators, researchers, medical ethicists, palliative care providers, and hospital‐based geriatricians.
-
Incorporate best practices for hospitalist administration, including quality improvement, patient safety, practice management, leadership, and demonstrating hospitalist value.
-
Identify evidence‐based best practices and trends for both adult and pediatric hospital medicine.
Instructions on Receiving Credit
For information on applicability and acceptance of continuing medical education credit for this activity, please consult your professional licensing board.
This activity is designed to be completed within the time designated on the title page; physicians should claim only those credits that reflect the time actually spent in the activity. To successfully earn credit, participants must complete the activity during the valid credit period that is noted on the title page.
Follow these steps to earn credit:
-
Log on to
www.blackwellpublishing.com/cme . -
Read the target audience, learning objectives, and author disclosures.
-
Read the article in print or online format.
-
Reflect on the article.
-
Access the CME Exam, and choose the best answer to each question.
-
Complete the required evaluation component of the activity.
If you wish to receive credit for this activity, which begins on the next page, please refer to the website:
Accreditation and Designation Statement
Blackwell Futura Media Services designates this educational activity for a 1 AMA PRA Category 1 Credit. Physicians should only claim credit commensurate with the extent of their participation in the activity.
Blackwell Futura Media Services is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Educational Objectives
Continuous participation in the Journal of Hospital Medicine CME program will enable learners to be better able to:
-
Interpret clinical guidelines and their applications for higher quality and more efficient care for all hospitalized patients.
-
Describe the standard of care for common illnesses and conditions treated in the hospital; such as pneumonia, COPD exacerbation, acute coronary syndrome, HF exacerbation, glycemic control, venous thromboembolic disease, stroke, etc.
-
Discuss evidence‐based recommendations involving transitions of care, including the hospital discharge process.
-
Gain insights into the roles of hospitalists as medical educators, researchers, medical ethicists, palliative care providers, and hospital‐based geriatricians.
-
Incorporate best practices for hospitalist administration, including quality improvement, patient safety, practice management, leadership, and demonstrating hospitalist value.
-
Identify evidence‐based best practices and trends for both adult and pediatric hospital medicine.
Instructions on Receiving Credit
For information on applicability and acceptance of continuing medical education credit for this activity, please consult your professional licensing board.
This activity is designed to be completed within the time designated on the title page; physicians should claim only those credits that reflect the time actually spent in the activity. To successfully earn credit, participants must complete the activity during the valid credit period that is noted on the title page.
Follow these steps to earn credit:
-
Log on to
www.blackwellpublishing.com/cme . -
Read the target audience, learning objectives, and author disclosures.
-
Read the article in print or online format.
-
Reflect on the article.
-
Access the CME Exam, and choose the best answer to each question.
-
Complete the required evaluation component of the activity.
If you wish to receive credit for this activity, which begins on the next page, please refer to the website:
Accreditation and Designation Statement
Blackwell Futura Media Services designates this educational activity for a 1 AMA PRA Category 1 Credit. Physicians should only claim credit commensurate with the extent of their participation in the activity.
Blackwell Futura Media Services is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Educational Objectives
Continuous participation in the Journal of Hospital Medicine CME program will enable learners to be better able to:
-
Interpret clinical guidelines and their applications for higher quality and more efficient care for all hospitalized patients.
-
Describe the standard of care for common illnesses and conditions treated in the hospital; such as pneumonia, COPD exacerbation, acute coronary syndrome, HF exacerbation, glycemic control, venous thromboembolic disease, stroke, etc.
-
Discuss evidence‐based recommendations involving transitions of care, including the hospital discharge process.
-
Gain insights into the roles of hospitalists as medical educators, researchers, medical ethicists, palliative care providers, and hospital‐based geriatricians.
-
Incorporate best practices for hospitalist administration, including quality improvement, patient safety, practice management, leadership, and demonstrating hospitalist value.
-
Identify evidence‐based best practices and trends for both adult and pediatric hospital medicine.
Instructions on Receiving Credit
For information on applicability and acceptance of continuing medical education credit for this activity, please consult your professional licensing board.
This activity is designed to be completed within the time designated on the title page; physicians should claim only those credits that reflect the time actually spent in the activity. To successfully earn credit, participants must complete the activity during the valid credit period that is noted on the title page.
Follow these steps to earn credit:
-
Log on to
www.blackwellpublishing.com/cme . -
Read the target audience, learning objectives, and author disclosures.
-
Read the article in print or online format.
-
Reflect on the article.
-
Access the CME Exam, and choose the best answer to each question.
-
Complete the required evaluation component of the activity.
Author Responsibilities and Disclosures
Since its founding in 2006,1 the editors of the Journal of Hospital Medicine (JHM), strongly supported the ethical guidelines and uniform requirements for manuscripts established by the International Committee of Medical Journal Editors (ICMJE).2 These guidelines require authors to verify that they have followed appropriate standards in the conduct of research, meet criteria for authorship, disclose potential conflicts of interest, and respect existing copyrights. With recent publication of editorials in leading medical journals affirming this responsibility for all authors submitting their scholarly work,38 the editors of the Journal echo the importance of following these ethical standards, and wish to update authors and readers on our policies related to authorship and plagiarism.
Disclosure of Competing Interests
Scientific publications commonly require that authors disclose relationships, financial or otherwise, with commercial entities that might have an interest in the subject matter of the article. Historically, biomedical journals varied in the content and format of the information they requested from authors,9 yielding inconsistent reporting by authors depending on the journal. Lack of clarity regarding what relationships authors should report contributed to this variable reporting. For example, an author might submit an article on headache management, and not believe it necessary to report honoraria received from pharmaceutical firms for giving lectures on antibiotic management of pneumonia. Thus, many believed that only funding related to the subject matter in a manuscript needed to be disclosed. While general advice has been to err on the side of disclosure, many authors hesitated to do so.
To clarify and standardize reporting requirements, the ICMJE recommended a uniform format for disclosure of competing interests,3 which was updated recently.10 The document, available online at
JHM strongly supports the ICMJE uniform requirements for manuscripts and has adopted the new form for disclosure of competing interests. Effective immediately, this documentation will be required for all types of manuscripts submitted to JHM. To help reduce the paperwork burden for authors, this documentation will be required only when authors are invited to revise and resubmit their work, after completion of the initial round of reviews. Typically at this stage, JHM also requests each author complete a Copyright Transfer Agreement (CTA). Thus, when a revision is requested by the Journal, we recommend that the corresponding author have each coauthor concurrently complete the CTA and disclosure of competing interests, and return all of the materials to JHM at the same time.
Criteria for Authorship
Authorship of scientific articles has important professional implications. In a field such as Hospital Medicine which explicitly values teamwork, it can sometimes be unclear which members of a team qualify for authorship on an article that may result from the group's work. The ICMJE provides the following guidance:2
-
Authorship credit should be based on (1) substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; (2) drafting the article or revising it critically for important intellectual content; and (3) final approval of the version to be published. Authors should meet conditions 1, 2, and 3.
The ICMJE notes that authorship is not justified for individuals who simply obtain or provide funding, participate in data collection or general supervision of the research, or serve as head of the group. Members of the team who play roles such as these are more appropriately acknowledged, and their specific contributions noted. The corresponding author should obtain written permission as such acknowledgements may imply endorsement of the work or its conclusions.
Authors, too, should make note of their individual contributions to manuscripts submitted to JHM. The Journal will begin publishing these specific contributions with each article, as do other medical journals.11
Plagiarism
Perhaps the most serious ethical violation that journals confront is plagiarism of copyrighted work. In its 5 years, JHM has detected 4 episodes of plagiarism. Thankfully, the Committee on Publication Ethics (
We recognize that other journals have needed to handle similar episodes of plagiarism,1215 and that self‐plagiarism (recycling of one's own published text) is also a concern.16, 17 Many methods exist to detect these practices.18 One powerful approach gaining popularity among medical journals utilizes CrossCheck. The CrossCheck service has 2 components: (1) a large, full‐text database of scholarly work from leading publishers, maintained by CrossRef (
Any form of plagiarism is inexcusable, and, if detected, is immediately addressed. Additionally, any author who submits plagiarized work will be banned from submitting manuscripts to JHM in the future, and will not be allowed to serve the Journal as a reviewer or in any other capacity. Our notification in selected cases of the individual's supervisor or department chair may elicit additional adverse consequences.
Summary
As the Journal of Hospital Medicine continues to grow and evolve, we are extraordinarily grateful when authors choose to submit their scholarly work to us. But growth does not come without challenges and responsibilities, such as a requirement to uphold ethical standards of biomedical publishing. We believe that the uniform disclosure of competing interests, clear reporting of contributions for authorship, and monitoring for plagiarism will help JHM maintain the standards that its readership and contributing authors deserve. We look forward to your contributions during our next 5 years, and beyond.
- .Hospital medicine's evolution—the next steps.J Hosp Med.2006;1(1):1–2.
- International Committee of Medical Journal Editors. Uniform requirements for manuscripts submitted to biomedical journals. Available at: http://www.icmje.org/. Accessed February 22,2010.
- , , , et al.Uniform format for disclosure of competing interests in ICMJE journals.N Engl J Med.2009;361(19):1896–1897.
- , , , et al.Uniform format for disclosure of competing interests in ICMJE journals.JAMA.2010;303(1):75–76.
- , , , et al.Uniform format for disclosure of competing interests in ICMJE journals.Ann Intern Med.2010;152(2):125–126.
- , , , et al.Uniform format for disclosure of competing interests in ICMJE journals.Lancet.2009;374(9699):1395–1396.
- , , , et al.Disclosure of competing interests.BMJ.2009;339:b4144.
- , , , et al.Uniform format for disclosure of competing interests in ICMJE journals.CMAJ.2009;181(9):565.
- , , , .Requirements and definitions in conflict of interest policies of medical journals.JAMA.2009;302(20):2230–2234.
- , , , , et al.Toward more uniform conflict disclosures ‐ the updated ICMJE conflict of interest reporting form.N Engl J Med.2010;363(2):188–189.
- , , , .Authorship criteria and disclosure of contributions: comparison of 3 general medical journals with different author contribution forms.JAMA.2004;292(1):86–88.
- , .Investigating allegations of scientific misconduct.BMJ.2005;331(7511):245–246.
- .Misconduct in medical research: does it exist in Britain?BMJ.1998;297:1531–1535.
- , .Research misconduct, retraction, and cleansing the medical literature: lessons from the Poehlman case.Annals of Internal Medicine.2006;144(8):609–613.
- .Report from the Scientific Integrity Advisor: issues arising in 2005 and 2006.Neurology.2007;68(21):1841–1842.
- Anonymous.Self‐plagiarism: unintentional, harmless, or fraud?Lancet.2009;374(9691):664.
- .Re‐using text from one's own previously published papers: an exploratory study of potential self‐plagiarism.Psychological Reports.2005;97(1):43–49.
- .Policing plagiarism.BMJ.2007;335(7627):963–964.
Since its founding in 2006,1 the editors of the Journal of Hospital Medicine (JHM), strongly supported the ethical guidelines and uniform requirements for manuscripts established by the International Committee of Medical Journal Editors (ICMJE).2 These guidelines require authors to verify that they have followed appropriate standards in the conduct of research, meet criteria for authorship, disclose potential conflicts of interest, and respect existing copyrights. With recent publication of editorials in leading medical journals affirming this responsibility for all authors submitting their scholarly work,38 the editors of the Journal echo the importance of following these ethical standards, and wish to update authors and readers on our policies related to authorship and plagiarism.
Disclosure of Competing Interests
Scientific publications commonly require that authors disclose relationships, financial or otherwise, with commercial entities that might have an interest in the subject matter of the article. Historically, biomedical journals varied in the content and format of the information they requested from authors,9 yielding inconsistent reporting by authors depending on the journal. Lack of clarity regarding what relationships authors should report contributed to this variable reporting. For example, an author might submit an article on headache management, and not believe it necessary to report honoraria received from pharmaceutical firms for giving lectures on antibiotic management of pneumonia. Thus, many believed that only funding related to the subject matter in a manuscript needed to be disclosed. While general advice has been to err on the side of disclosure, many authors hesitated to do so.
To clarify and standardize reporting requirements, the ICMJE recommended a uniform format for disclosure of competing interests,3 which was updated recently.10 The document, available online at
JHM strongly supports the ICMJE uniform requirements for manuscripts and has adopted the new form for disclosure of competing interests. Effective immediately, this documentation will be required for all types of manuscripts submitted to JHM. To help reduce the paperwork burden for authors, this documentation will be required only when authors are invited to revise and resubmit their work, after completion of the initial round of reviews. Typically at this stage, JHM also requests each author complete a Copyright Transfer Agreement (CTA). Thus, when a revision is requested by the Journal, we recommend that the corresponding author have each coauthor concurrently complete the CTA and disclosure of competing interests, and return all of the materials to JHM at the same time.
Criteria for Authorship
Authorship of scientific articles has important professional implications. In a field such as Hospital Medicine which explicitly values teamwork, it can sometimes be unclear which members of a team qualify for authorship on an article that may result from the group's work. The ICMJE provides the following guidance:2
-
Authorship credit should be based on (1) substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; (2) drafting the article or revising it critically for important intellectual content; and (3) final approval of the version to be published. Authors should meet conditions 1, 2, and 3.
The ICMJE notes that authorship is not justified for individuals who simply obtain or provide funding, participate in data collection or general supervision of the research, or serve as head of the group. Members of the team who play roles such as these are more appropriately acknowledged, and their specific contributions noted. The corresponding author should obtain written permission as such acknowledgements may imply endorsement of the work or its conclusions.
Authors, too, should make note of their individual contributions to manuscripts submitted to JHM. The Journal will begin publishing these specific contributions with each article, as do other medical journals.11
Plagiarism
Perhaps the most serious ethical violation that journals confront is plagiarism of copyrighted work. In its 5 years, JHM has detected 4 episodes of plagiarism. Thankfully, the Committee on Publication Ethics (
We recognize that other journals have needed to handle similar episodes of plagiarism,1215 and that self‐plagiarism (recycling of one's own published text) is also a concern.16, 17 Many methods exist to detect these practices.18 One powerful approach gaining popularity among medical journals utilizes CrossCheck. The CrossCheck service has 2 components: (1) a large, full‐text database of scholarly work from leading publishers, maintained by CrossRef (
Any form of plagiarism is inexcusable, and, if detected, is immediately addressed. Additionally, any author who submits plagiarized work will be banned from submitting manuscripts to JHM in the future, and will not be allowed to serve the Journal as a reviewer or in any other capacity. Our notification in selected cases of the individual's supervisor or department chair may elicit additional adverse consequences.
Summary
As the Journal of Hospital Medicine continues to grow and evolve, we are extraordinarily grateful when authors choose to submit their scholarly work to us. But growth does not come without challenges and responsibilities, such as a requirement to uphold ethical standards of biomedical publishing. We believe that the uniform disclosure of competing interests, clear reporting of contributions for authorship, and monitoring for plagiarism will help JHM maintain the standards that its readership and contributing authors deserve. We look forward to your contributions during our next 5 years, and beyond.
Since its founding in 2006,1 the editors of the Journal of Hospital Medicine (JHM), strongly supported the ethical guidelines and uniform requirements for manuscripts established by the International Committee of Medical Journal Editors (ICMJE).2 These guidelines require authors to verify that they have followed appropriate standards in the conduct of research, meet criteria for authorship, disclose potential conflicts of interest, and respect existing copyrights. With recent publication of editorials in leading medical journals affirming this responsibility for all authors submitting their scholarly work,38 the editors of the Journal echo the importance of following these ethical standards, and wish to update authors and readers on our policies related to authorship and plagiarism.
Disclosure of Competing Interests
Scientific publications commonly require that authors disclose relationships, financial or otherwise, with commercial entities that might have an interest in the subject matter of the article. Historically, biomedical journals varied in the content and format of the information they requested from authors,9 yielding inconsistent reporting by authors depending on the journal. Lack of clarity regarding what relationships authors should report contributed to this variable reporting. For example, an author might submit an article on headache management, and not believe it necessary to report honoraria received from pharmaceutical firms for giving lectures on antibiotic management of pneumonia. Thus, many believed that only funding related to the subject matter in a manuscript needed to be disclosed. While general advice has been to err on the side of disclosure, many authors hesitated to do so.
To clarify and standardize reporting requirements, the ICMJE recommended a uniform format for disclosure of competing interests,3 which was updated recently.10 The document, available online at
JHM strongly supports the ICMJE uniform requirements for manuscripts and has adopted the new form for disclosure of competing interests. Effective immediately, this documentation will be required for all types of manuscripts submitted to JHM. To help reduce the paperwork burden for authors, this documentation will be required only when authors are invited to revise and resubmit their work, after completion of the initial round of reviews. Typically at this stage, JHM also requests each author complete a Copyright Transfer Agreement (CTA). Thus, when a revision is requested by the Journal, we recommend that the corresponding author have each coauthor concurrently complete the CTA and disclosure of competing interests, and return all of the materials to JHM at the same time.
Criteria for Authorship
Authorship of scientific articles has important professional implications. In a field such as Hospital Medicine which explicitly values teamwork, it can sometimes be unclear which members of a team qualify for authorship on an article that may result from the group's work. The ICMJE provides the following guidance:2
-
Authorship credit should be based on (1) substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; (2) drafting the article or revising it critically for important intellectual content; and (3) final approval of the version to be published. Authors should meet conditions 1, 2, and 3.
The ICMJE notes that authorship is not justified for individuals who simply obtain or provide funding, participate in data collection or general supervision of the research, or serve as head of the group. Members of the team who play roles such as these are more appropriately acknowledged, and their specific contributions noted. The corresponding author should obtain written permission as such acknowledgements may imply endorsement of the work or its conclusions.
Authors, too, should make note of their individual contributions to manuscripts submitted to JHM. The Journal will begin publishing these specific contributions with each article, as do other medical journals.11
Plagiarism
Perhaps the most serious ethical violation that journals confront is plagiarism of copyrighted work. In its 5 years, JHM has detected 4 episodes of plagiarism. Thankfully, the Committee on Publication Ethics (
We recognize that other journals have needed to handle similar episodes of plagiarism,1215 and that self‐plagiarism (recycling of one's own published text) is also a concern.16, 17 Many methods exist to detect these practices.18 One powerful approach gaining popularity among medical journals utilizes CrossCheck. The CrossCheck service has 2 components: (1) a large, full‐text database of scholarly work from leading publishers, maintained by CrossRef (
Any form of plagiarism is inexcusable, and, if detected, is immediately addressed. Additionally, any author who submits plagiarized work will be banned from submitting manuscripts to JHM in the future, and will not be allowed to serve the Journal as a reviewer or in any other capacity. Our notification in selected cases of the individual's supervisor or department chair may elicit additional adverse consequences.
Summary
As the Journal of Hospital Medicine continues to grow and evolve, we are extraordinarily grateful when authors choose to submit their scholarly work to us. But growth does not come without challenges and responsibilities, such as a requirement to uphold ethical standards of biomedical publishing. We believe that the uniform disclosure of competing interests, clear reporting of contributions for authorship, and monitoring for plagiarism will help JHM maintain the standards that its readership and contributing authors deserve. We look forward to your contributions during our next 5 years, and beyond.
- .Hospital medicine's evolution—the next steps.J Hosp Med.2006;1(1):1–2.
- International Committee of Medical Journal Editors. Uniform requirements for manuscripts submitted to biomedical journals. Available at: http://www.icmje.org/. Accessed February 22,2010.
- , , , et al.Uniform format for disclosure of competing interests in ICMJE journals.N Engl J Med.2009;361(19):1896–1897.
- , , , et al.Uniform format for disclosure of competing interests in ICMJE journals.JAMA.2010;303(1):75–76.
- , , , et al.Uniform format for disclosure of competing interests in ICMJE journals.Ann Intern Med.2010;152(2):125–126.
- , , , et al.Uniform format for disclosure of competing interests in ICMJE journals.Lancet.2009;374(9699):1395–1396.
- , , , et al.Disclosure of competing interests.BMJ.2009;339:b4144.
- , , , et al.Uniform format for disclosure of competing interests in ICMJE journals.CMAJ.2009;181(9):565.
- , , , .Requirements and definitions in conflict of interest policies of medical journals.JAMA.2009;302(20):2230–2234.
- , , , , et al.Toward more uniform conflict disclosures ‐ the updated ICMJE conflict of interest reporting form.N Engl J Med.2010;363(2):188–189.
- , , , .Authorship criteria and disclosure of contributions: comparison of 3 general medical journals with different author contribution forms.JAMA.2004;292(1):86–88.
- , .Investigating allegations of scientific misconduct.BMJ.2005;331(7511):245–246.
- .Misconduct in medical research: does it exist in Britain?BMJ.1998;297:1531–1535.
- , .Research misconduct, retraction, and cleansing the medical literature: lessons from the Poehlman case.Annals of Internal Medicine.2006;144(8):609–613.
- .Report from the Scientific Integrity Advisor: issues arising in 2005 and 2006.Neurology.2007;68(21):1841–1842.
- Anonymous.Self‐plagiarism: unintentional, harmless, or fraud?Lancet.2009;374(9691):664.
- .Re‐using text from one's own previously published papers: an exploratory study of potential self‐plagiarism.Psychological Reports.2005;97(1):43–49.
- .Policing plagiarism.BMJ.2007;335(7627):963–964.
- .Hospital medicine's evolution—the next steps.J Hosp Med.2006;1(1):1–2.
- International Committee of Medical Journal Editors. Uniform requirements for manuscripts submitted to biomedical journals. Available at: http://www.icmje.org/. Accessed February 22,2010.
- , , , et al.Uniform format for disclosure of competing interests in ICMJE journals.N Engl J Med.2009;361(19):1896–1897.
- , , , et al.Uniform format for disclosure of competing interests in ICMJE journals.JAMA.2010;303(1):75–76.
- , , , et al.Uniform format for disclosure of competing interests in ICMJE journals.Ann Intern Med.2010;152(2):125–126.
- , , , et al.Uniform format for disclosure of competing interests in ICMJE journals.Lancet.2009;374(9699):1395–1396.
- , , , et al.Disclosure of competing interests.BMJ.2009;339:b4144.
- , , , et al.Uniform format for disclosure of competing interests in ICMJE journals.CMAJ.2009;181(9):565.
- , , , .Requirements and definitions in conflict of interest policies of medical journals.JAMA.2009;302(20):2230–2234.
- , , , , et al.Toward more uniform conflict disclosures ‐ the updated ICMJE conflict of interest reporting form.N Engl J Med.2010;363(2):188–189.
- , , , .Authorship criteria and disclosure of contributions: comparison of 3 general medical journals with different author contribution forms.JAMA.2004;292(1):86–88.
- , .Investigating allegations of scientific misconduct.BMJ.2005;331(7511):245–246.
- .Misconduct in medical research: does it exist in Britain?BMJ.1998;297:1531–1535.
- , .Research misconduct, retraction, and cleansing the medical literature: lessons from the Poehlman case.Annals of Internal Medicine.2006;144(8):609–613.
- .Report from the Scientific Integrity Advisor: issues arising in 2005 and 2006.Neurology.2007;68(21):1841–1842.
- Anonymous.Self‐plagiarism: unintentional, harmless, or fraud?Lancet.2009;374(9691):664.
- .Re‐using text from one's own previously published papers: an exploratory study of potential self‐plagiarism.Psychological Reports.2005;97(1):43–49.
- .Policing plagiarism.BMJ.2007;335(7627):963–964.
Hospitalist Time Motion Study
Hospital Medicine represents the fastest‐growing specialty in the history of medicine in the United States, with approximately 28,000 hospitalists now working in over half of American hospitals.1 Hospitalists increasingly fill the gap between demand for care of hospitalized patients and the deficit of physicians previously availableprimary care physicians in community hospitals and residents in teaching hospitals.2, 3 This growth has also been driven by hospitalists' ability to increase clinical efficiency. Research consistently demonstrates a reduction in overall costs and length of stay with the use of hospitalists.47 Additionally, many teaching hospitals have implemented nonteaching hospitalist services in an effort to comply with the Accreditation Council for Graduate Medicine Education (ACGME) program requirements regarding resident duty hours.8 Given the potential for improved clinical efficiency and the need to comply with revised ACGME program requirements, the Hospital Medicine Service at Northwestern Memorial Hospital (NMH) was established in 2003. Today, this service cares for more than half of hospitalized medical patients at NMH.
Although extensive research documents that implementation of a hospitalist program improves the efficiency of hospital care delivery,4, 6 there is little data to explain how hospitalists achieve this level of efficiency or how efficiency might be increased further. Several authors have suggested potential explanations for hospitalists' efficiency gains, but none has yet received strong empirical validation.5, 7 The only previously published study to directly observe more than a small portion of the activities of hospitalists was conducted at NMH in 2006.9 O'Leary et al. used time‐motion methodology to study ten hospitalists for 75 hours total. They found that hospitalists spend a large amount of time on communication when compared to nonhospitalist physicians. However, the study only reported partial information about how and with whom this communications was performed. Similarly, the authors reported that documentation occupied about a quarter of hospitalists' time, but did not report more detailed information about what was being documented and how. Additionally, they noted that hospitalists spent 21% of their time multitasking, but did not report what types of activities were performed during these episodes. Finally, at the time of that study hospitalists at NMH saw about 40% fewer patients per day than they do now. Increasing the number of patients each physician sees in a day is an obvious way to increase productivity, but it is unclear how this affects hospitalist workflow and time spent in various clinical activities.
Another important trend in hospital care delivery is the implementation of electronic medical records (EMR).10 NMH was just transitioning to a fully integrated EMR and computerized physician order entry (CPOE) system when the previous time‐motion study was performed. Now that the system is in place, a significant proportion of hospitalists' time has shifted from using a paper‐based record to sitting in front of a computer. However, we do not know exactly how hospitalists interact with the EMR and how this alters workflow; an increasingly important issue as hospitals across the U.S. implement EMRs at the behest of the federal government and aiming to improve patient safety.11
To better understand the workflow of hospitalists and validate the findings of the O'Leary study in a larger sample of hospitalists, we undertook this study seeking to collect data continuously for complete shifts, rather than sampling just a few hours at a time. We hypothesized that this would reduce observer effects and provide us with a more complete and accurate assessment of a day in the life of a hospitalist.
Methods
Study Site
The study was conducted at NMH, an 897‐bed tertiary care teaching hospital in Chicago, IL, and was approved by the Institutional Review Board of Northwestern University. Patients are admitted to the Hospital Medicine Service from the Emergency Department or directly from physicians' offices based on bed availability in a quasi‐randomized fashion. Hospitalists included in the study cared for patients without the assistance of housestaff physicians and worked 7 consecutive days while on service, usually followed by 7 consecutive days off service. During weeks on service, hospitalist shifts started at 7 AM and ended between 5 PM and 7 PM.
Data Collection Tool Development
To facilitate collection of detailed information sought for this study, we developed an electronic data collection tool. A systematic review of the medical literature on time studies performed by our research group indicated a lack of methodological standardization and dissimilar activity categorizations across studies.12 We attempted to develop a standardized method and data collection instrument for future studies, and first created a data dictionary consisting of a list of hospitalist activities and their descriptions. The initial components were drawn from prior time‐motion studies9, 13, 14 and input from experienced hospitalists (KJO and MVW). The activity list was then refined after a preliminary observation period in which five hospitalists were followed for a total of 6 shifts. Observers noted the specific activities being performed by the hospitalists and asked for explanations and clarification when necessary. In order for an activity to be included in the final list, the activity had to be easily observable and identifiable without subjective interpretation from the observer. The preliminary observation period ended once we were satisfied that no new activities were emerging.
The compiled list of activities was then broken down into related groups and separated into additional subcategories to increase the specificity of data collection. The final list of activities was reviewed by several experienced hospitalists to ensure completeness. The data dictionary was then loaded onto a Palm Pilot Tx using WorkStudy+ Plus software. The final activity list consisted of 8 main categories, 32 secondary categories, and 53 tertiary categories (See Appendix). To facilitate comparisons with prior studies, we followed the convention of including the categories of direct and indirect patient care. We defined direct patient care as those activities involving face‐to‐face interaction between the hospitalist and the patient. The more general indirect care category encompassed other categories of activity relevant to the patient's care but not performed in the presence of the patient (ie, professional communication, interaction with the EMR, and other patient related activities like searching for medical knowledge on the Internet or reading telemetry monitors).
Pilot Testing
We trained 6 observers in the use of the data collection tool. Each observer practiced shadowing for more than 20 hours with the tool before collecting study data. During this pilot testing phase we optimized the layout of the tool to facilitate rapid documentation of hospitalist activities and multitasking. Interobserver reliability was confirmed by having 2 observers shadow the same hospitalist for a three hour time period. In all cases, the observers obtained an average interclass correlation coefficient of at least 0.95 with a 95% confidence interval of .85 to 1.0 prior to collecting study data.
Study Design
Data collection occurred between July and September of 2008. A total of 24 hospitalists were observed, each for 2 complete weekday shifts starting at 7 AM and ending between 5 PM and 7 PM. Of note, we only observed hospitalists who were directly caring for patients and not part of a teaching service. Each hospitalist was contacted about the project at least a week prior to any observations and informed consent was obtained. A single observer shadowed a single hospitalist continuously, trading off with a new observer every 3 hours to avoid fatigue. To minimize any observation effect our data collectors were instructed not to initiate and to minimize conversation with the hospitalists. At the end of the hospitalist's shift the following data were tallied: the number of patients in the hospitalist's care at the beginning of the day, the number of patients discharged during the day, and the number of admissions. Patient load was determined by adding the number of admissions to the number of patients at the beginning of the day.
Data Analysis
Minutes were tallied for each of the categories and subcategories. Data is reported as percentages of total duration of observed activities (ie, including multitasking) unless otherwise specified. To explore the effect of patient volume on hospitalist workflow we performed t‐tests comparing the number of minutes hospitalists spent per patient in various activities on days with below average patient volume as compared to those with above average volume. Additionally, we performed a Wilcoxon two‐samples test to check for a difference in length of shift between these 2 groups.
Results
A total of 24 hospitalists were shadowed for a total of approximately 494 hours. For 43 of these hours a hospitalist was observed performing 2 tasks simultaneously, bringing the total duration of observed activities to 537 hours with multitasking. The hospitalists were a mean 34 1.1 years of age and 12 (50%) were female. Twenty (83%) had completed residency 2 or more years prior to the study, 2 (8%) had a year of hospitalist experience since residency, and the remaining 2 (8%) had just completed residency. Sixteen (67%) hospitalists were Asian or Pacific Islanders, 6 (25%) were White, and 2 (8%) were Black. The hospitalists cared for an average of 13.2 0.6 patients per shift and an average shift lasted 10 hours and 19 minutes 52 minutes.
Table 1 lists the mean percentage of time hospitalists spent on the various activities. Subjects spent the most time (34.1%) interacting with the EMR. Communication and direct care were the next most frequent activities at 25.9% and 17.4% of each shift respectively, followed by professional development (6.5%), travel (6.2%), personal time (5.6%), other indirect care (3.9%), and waiting (0.4%). The 3 subcategories included in indirect care time accounted for about 64% of all recorded activities.
| Main Category | % Total Observed Activities | (95% CI)* | Subcategory | % Main Category | (95% CI)* |
|---|---|---|---|---|---|
| |||||
| EMR* | 34.1 | (32.435.9) | |||
| Writing | 58.4 | (55.761.0) | |||
| Orders | 20.2 | (18.521.9) | |||
| Reading/reviewing | 19.4 | (17.321.5) | |||
| Other | 2.1 | (1.82.5) | |||
| Communication* | 25.9 | (24.427.4) | |||
| Outgoing call | 36.9 | (33.640.2) | |||
| Face to face | 28.1 | (25.231.0) | |||
| Incoming call | 14.4 | (12.616.3) | |||
| Sending page | 8.6 | (7.79.4) | |||
| Rounds | 3.8 | (1.85.8) | |||
| Receiving page | 3.4 | (2.94.0) | |||
| E‐mail | 2.9 | (1.83.9) | |||
| Reviewing page | 1.8 | (1.32.3) | |||
| Fax | 0.1 | (0.00.2) | |||
| Direct care | 17.4 | (15.918.9) | |||
| Professional Development | 6.5 | (4.48.5) | |||
| Travel | 6.2 | (5.66.7) | |||
| Personal | 5.7 | (4.17.2) | |||
| Other indirect care* | 3.9 | (3.44.4) | |||
| Wait | 0.4 | (0.20.5) | |||
Of the nearly 4 hours (233 minutes) per shift hospitalists spent using the EMR, the majority (58.4%) was spent documenting (See Table 1). Placing orders and reading/reviewing notes were nearly equal at 20.2% and 19.4% respectively, and other EMR activities took 2.1% of EMR time. Over half of the time (54.1%) hospitalists spent documenting in the EMR system was dedicated to progress notes. The remainder of effort was expended on writing histories and physicals (15.3%), discharge instructions (14.7%), discharge summaries (7.9%), sign‐outs (6.8%), and performing medication reconciliation (1.4%). Of the time spent reading and reviewing documents on the EMR, most was spent reviewing lab results (45.4%) or notes from the current admission (40.4%). Reviewing imaging studies occupied 8.1%, and notes from past encounters accounted for 6.2% of this category's time.
Various modes of communication were used during the nearly three hours (176 minutes) per shift dedicated to communication. Phone calls took up approximately half of the hospitalists' communication time, with 36.8% spent on outgoing calls and 14.2% incoming calls. Face‐to‐face communication was the next most common mode, accounting for 28.2% of the total. Time spent sending pages (8.8%), receiving pages (3.4%), and reviewing pages (1.8%) consumed 14% of all communication time. E‐mail and fax were used sparingly, at 3.1% and 0.1% of communication time, respectively. Finally, meetings involving other hospital staff (interdisciplinary rounds) occupied 3.4% of communication time.
The amount of time hospitalists spent communicating with specific types of individuals is shown in Table 2. Hospitalists spent the most time communicating with other physicians (44.5%) and nurses (18.1%). They spent less time communicating with people from the remaining categories; utilization staff (5.7%), patients' family members (5.6%), case managers (4.2%), primary care physicians (3.4%), ancillary staff (3.1%), and pharmacists (0.6%). Communication with other individuals that did not fit in the above categories accounted for 8.8%, and 5.3% of communication could not be clearly categorized, generally because the hospitalist was communicating by phone or text page and ascertaining with whom would have required significant interruption.
| Subcategory | % Main Category | (95% CI)* |
|---|---|---|
| ||
| Inpatient physician | 44.5 | (41.747.2) |
| Nursing staff | 18.0 | (16.019.9) |
| Other | 8.5 | (6.810.2) |
| Family | 5.8 | (4.07.7) |
| Utilization staff | 5.8 | (4.67.0) |
| Uncategorized | 5.7 | (3.77.6) |
| PCC | 4.0 | (2.35.7) |
| PCP | 3.6 | (2.74.5) |
| Ancillary staff | 2.9 | (2.23.7) |
| Pharmacy | 1.4 | (0.82.0) |
We found that 16% of all recorded activities occurred when another activity was also ongoing. This means that hospitalists were performing more than one activity for approximately 54 minutes per day, or about 9% of the average 10.3‐hour shift. Instances of multitasking occurred frequently, but were usually brief; the hospitalists performed 2 activities simultaneously an average of 75 times per day, but 79% of these occurrences lasted less than 1 minute. Of the 86 hours of multitasking activities recorded, 41% was communication time and another 41% was EMR use. This means that a second activity was being performed during 19% of the time hospitalists spent using the EMR and 26% of the time they spent communicating. Of the time spent on critical documentation activities like writing prescriptions and orders, 24% was recorded during a multitasking event.
The amount of time hospitalists spent per patient on days with above average patient volume as compared to those with below average patient volume is shown in Table 3. Hospitalists with above average patient numbers spent about 3 minutes less per patient interacting with the EMR (a 17% reduction; P < 0.01), and about 2 minutes less per patient communicating (a 14% reduction; P < 0.01). The average length of shift increased by 12 minutes on days when patient volume was above average; P < 0.05.
| Subcategory | Minutes: Below Average Census | (95% CI)* | Minutes: Above Average Census | (95% CI)* | Pr > |t| |
|---|---|---|---|---|---|
| |||||
| EMR | 19.12 | (17.5020.75) | 15.83 | (14.1717.49) | <.001 |
| Communication | 14.28 | (12.8615.71) | 12.21 | (11.0713.36) | 0.002 |
| Direct care | 9.30 | (8.1810.42) | 8.59 | (7.279.91) | 0.293 |
| Professional development | 4.09 | (2.365.81) | 2.57 | (1.263.89) | 0.026 |
| Personal | 3.52 | (2.394.65) | 2.05 | (1.292.82) | 0.032 |
| Travel | 3.32 | (2.863.79) | 2.93 | (2.643.22) | 0.566 |
| Other indirect care | 2.37 | (1.902.84) | 1.65 | (1.321.98) | 0.292 |
| Wait | 0.25 | (0.080.41) | 0.14 | (0.040.25) | 0.881 |
Discussion
To our knowledge, this study represents the largest time‐motion evaluation of hospitalist activities ever undertaken, and provides the most detailed assessment of hospitalists' activities when caring for patients without residents or medical students. We confirmed that hospitalists spend the majority of their time (64%) undertaking care activities away from the patient's bedside, and are involved in direct patient care contact only 17% of their time, averaging about 9 minutes per patient. The hospitalists spent about a quarter (26%) of their time communicating with others. Compared to other physicians, this is an unusually large amount of time. For example, Hollingsworth et al.15 found that emergency medicine physicians spent just half as much (13%) of their time on communication with other providers and staff. This may reflect hospitalists' central role in the coordination of consulting specialists. The other significant portion of hospitalists' effort focuses on documentation in the electronic medical record, with 22% of their time required for CPOE and note writing, and overall a third of their time (34.1%) committed to interacting with the EMR.
In many respects, our results confirm the findings of O'Leary et al.'s previous work. While this current study more precisely identified how hospitalists spend their time, the general proportions of times were similar. Both studies found that indirect care activities occupied about two‐thirds of hospitalists' time (64% in this study and 69% in the previous study). We also documented similar portions of total time for direct patient care (17% vs. 18%) and communication (26% vs. 24%). Interestingly, with complete implementation of the EMR system, the percentage of time spent on documentation appeared to decrease. O'Leary et al. reported that documentation accounted for 26% of hospitalists' time, while the equivalent activities (writing in the EMR or paper prescriptions) accounted for only 21% in the current study. Unfortunately, the significance of this finding is difficult to determine given the concurrent changes in patient volumes and the varying extent of EMR implementation during the earlier study.
Over half of hospitalists' communication time is spent either making or receiving phone calls. This suggests that efforts to facilitate communication (eg, use of mobile phone systems and voicemail) might enhance efficiency. Additionally, we found that nearly half of our hospitalists' communication was with other physicians. Not surprisingly, our study confirmed that an important part of hospitalists' work involves organizing and collaborating with a variety of specialists to provide optimal care for their patients.
Hospitalists spent a great deal of time multitasking. We found that multitasking time accounted for nearly 1 of every 10 minutes during the day. The most common combination of activities involved communication that occurred during a period of EMR use. These interruptions could have serious consequences should physicians lose track of what they are doing while ordering procedures or prescribing medications.
We documented a smaller portion of multitasking time than O'Leary's earlier study. This could be due to differences in how multitasking was defined or recorded in the 2 studies. Our electronic data collection tool allowed us to capture rapid task switching and multitasking to the second, rather than to the minute, as was done with the stopwatch and paper form used in the previous study. This precision was important, especially considering that nearly 80% of the recorded instances of multitasking lasted less than 1 minute.
Our data also suggests that patient census has significant effects on certain parts of hospitalist workflow. Patient volume for our subjects ranged from 10 to 19 patients per shift, with a mean of 13.2 patients. The amount of time our hospitalists spent with each patient did not differ significantly between above and below average census days. However, EMR time per patient was significantly reduced on above average census days. Anecdotally, several of our hospitalists suggested that on high census days they put off less time‐sensitive documentation activities like discharge summaries until after they leave the hospital and complete the work from home or on the following day. Thus, our study likely underestimates the total additional effort on high volume days, but unfortunately we had no direct way of quantifying work performed outside of the hospital or on subsequent days. Communication time was also significantly reduced when patient volumes were above average, suggesting that hospitalists had less time to confer with consultants or answer the questions of nurses and patient family members.
Several factors limit the interpretation and application of our findings. First, our study was conducted at a single urban, academic hospital, which may limit its applicability for hospitalists working at community hospitals. Given that more than 90% of hospital care in the U.S. occurs in the community hospital setting, research to confirm these findings in such hospitals is needed.16 Nonclinical research assistants collected all of the data, so the results may be limited by the accuracy of their interpretations. However, our extensive training and documentation of their accuracy serves as a strength of the study. Finally, we focused exclusively on daytime, weekday activities of hospitalists. Notably, 3 hospitalists work through the night at our facility, and 24‐hour coverage by hospitalists is increasingly common across the U.S. We expect weekend and night shift workflow to be somewhat different from standard day shifts due to the decreased availability of other medical providers for testing, consults, and procedures. Future research should focus on potential differences in activities on nights and weekends compared to weekdays.
This extensive, comprehensive analysis of hospitalist activities and workflow provides a foundation for future research and confirms much of O'Leary et al.'s original study. O'Leary's simpler approach of observing smaller blocks of time rather than full shifts proved effective; the two methodologies produced markedly similar results. The current study also offers some insight into matters of efficiency. We found that hospitalists with higher patient loads cut down on EMR and communication time. We also confirmed that hospitalists spend the largest portion of their time interacting with the EMR. A more efficient EMR system could therefore be especially helpful in providing more time for direct patient care and the communication necessary to coordinate care. Given that most hospitals provide financial support for hospital medicine programs (an average of $95,000 per hospitalist full‐time equivalent (FTE)1), hospital administrators have a keen interest in understanding how hospitalists might be more efficient. For example, if hospitalists could evaluate and manage two additional patients each day by exchanging time focused on medical record documentation for direct care activities, the cost of a hospitalist drops substantively. By understanding current hospitalist activities, efforts at redesigning their workflow can be more successful at addressing issues related to scheduling, communication, and compensation, thus improving the overall model of practice as well as the quality of patient care.17
Acknowledgements
We thank Caitlin Lawes and Stephen Williams for help with data collection, and all the hospitalists who participated in this study.
- Society of Hospital Medicine. About SHM.2008; http://www.hospitalmedicine.org/AM/Template.cfm?Section=About_SHM. Accessed April 2010.
- , .The evolution and future of hospital medicine.Mt Sinai J Med.2008;75(5):418–423.
- , , .Rehospitalizations among patients in the Fee‐for‐Service Medicare Program.N Engl J Med.2009;360(14):1418–1428.
- , , , , , .Outcomes of care by hospitalists, general internists, and family physicians.[see comment].N Engl J Med.2007;357(25):2589–2600.
- , .The hospitalist movement 5 years later.JAMA.2002;287:487–494.
- , .The impact of hospitalists on the cost and quality of inpatient care in the United States: a research synthesis.Med Care Res Rev.2005;62:379–406.
- .Hospitalists and the hospital medicine system of care are good for patient care.Arch Intern Med.2008;168(12):1254–1256; discussion 1259–1260.
- , .Hospitalists in teaching hospitals: opportunities but not without danger.J Gen Intern Med.2004;19:392–393.
- , , .How hospitalists spend their time: insights on efficiency and safety.J Hosp Med.2006;1(2):88–93.
- , , , et al.Use of electronic health records in U.S. hospitals.N Engl J Med.2009;360.
- .Electronic medical records at a crossroads: impetus for change or missed opportunity?JAMA.2009;302(10):1109–1111.
- , , , , .Systematic review of time studies evaluating physicians in the hospital setting.J Hosp Med.2010;5(6):000–000.
- , , , .All in a day's work: an observational study to quantify how and with whom doctors on hospital wards spend their time.Med J Aust.2008;188(9):506–509.
- , , , .Emergency department workplace interruptions: are emergency physicians “interrupt‐driven” and “multitasking”?Acad Emerg Med.2000;7:1239–1243.
- , , , , .How do physicians and nurses spend their time in the emergency department?Ann Emerg Med.1998;31(1):87–91.
- , , , , .The ecology of medical care revisited.N Engl J Med.2001;344(26):2021–2025.
- , .Organizing a hospitalist program: an overview of fundamental concepts.Med Clin North Am.2002;86(4):887–909.
Hospital Medicine represents the fastest‐growing specialty in the history of medicine in the United States, with approximately 28,000 hospitalists now working in over half of American hospitals.1 Hospitalists increasingly fill the gap between demand for care of hospitalized patients and the deficit of physicians previously availableprimary care physicians in community hospitals and residents in teaching hospitals.2, 3 This growth has also been driven by hospitalists' ability to increase clinical efficiency. Research consistently demonstrates a reduction in overall costs and length of stay with the use of hospitalists.47 Additionally, many teaching hospitals have implemented nonteaching hospitalist services in an effort to comply with the Accreditation Council for Graduate Medicine Education (ACGME) program requirements regarding resident duty hours.8 Given the potential for improved clinical efficiency and the need to comply with revised ACGME program requirements, the Hospital Medicine Service at Northwestern Memorial Hospital (NMH) was established in 2003. Today, this service cares for more than half of hospitalized medical patients at NMH.
Although extensive research documents that implementation of a hospitalist program improves the efficiency of hospital care delivery,4, 6 there is little data to explain how hospitalists achieve this level of efficiency or how efficiency might be increased further. Several authors have suggested potential explanations for hospitalists' efficiency gains, but none has yet received strong empirical validation.5, 7 The only previously published study to directly observe more than a small portion of the activities of hospitalists was conducted at NMH in 2006.9 O'Leary et al. used time‐motion methodology to study ten hospitalists for 75 hours total. They found that hospitalists spend a large amount of time on communication when compared to nonhospitalist physicians. However, the study only reported partial information about how and with whom this communications was performed. Similarly, the authors reported that documentation occupied about a quarter of hospitalists' time, but did not report more detailed information about what was being documented and how. Additionally, they noted that hospitalists spent 21% of their time multitasking, but did not report what types of activities were performed during these episodes. Finally, at the time of that study hospitalists at NMH saw about 40% fewer patients per day than they do now. Increasing the number of patients each physician sees in a day is an obvious way to increase productivity, but it is unclear how this affects hospitalist workflow and time spent in various clinical activities.
Another important trend in hospital care delivery is the implementation of electronic medical records (EMR).10 NMH was just transitioning to a fully integrated EMR and computerized physician order entry (CPOE) system when the previous time‐motion study was performed. Now that the system is in place, a significant proportion of hospitalists' time has shifted from using a paper‐based record to sitting in front of a computer. However, we do not know exactly how hospitalists interact with the EMR and how this alters workflow; an increasingly important issue as hospitals across the U.S. implement EMRs at the behest of the federal government and aiming to improve patient safety.11
To better understand the workflow of hospitalists and validate the findings of the O'Leary study in a larger sample of hospitalists, we undertook this study seeking to collect data continuously for complete shifts, rather than sampling just a few hours at a time. We hypothesized that this would reduce observer effects and provide us with a more complete and accurate assessment of a day in the life of a hospitalist.
Methods
Study Site
The study was conducted at NMH, an 897‐bed tertiary care teaching hospital in Chicago, IL, and was approved by the Institutional Review Board of Northwestern University. Patients are admitted to the Hospital Medicine Service from the Emergency Department or directly from physicians' offices based on bed availability in a quasi‐randomized fashion. Hospitalists included in the study cared for patients without the assistance of housestaff physicians and worked 7 consecutive days while on service, usually followed by 7 consecutive days off service. During weeks on service, hospitalist shifts started at 7 AM and ended between 5 PM and 7 PM.
Data Collection Tool Development
To facilitate collection of detailed information sought for this study, we developed an electronic data collection tool. A systematic review of the medical literature on time studies performed by our research group indicated a lack of methodological standardization and dissimilar activity categorizations across studies.12 We attempted to develop a standardized method and data collection instrument for future studies, and first created a data dictionary consisting of a list of hospitalist activities and their descriptions. The initial components were drawn from prior time‐motion studies9, 13, 14 and input from experienced hospitalists (KJO and MVW). The activity list was then refined after a preliminary observation period in which five hospitalists were followed for a total of 6 shifts. Observers noted the specific activities being performed by the hospitalists and asked for explanations and clarification when necessary. In order for an activity to be included in the final list, the activity had to be easily observable and identifiable without subjective interpretation from the observer. The preliminary observation period ended once we were satisfied that no new activities were emerging.
The compiled list of activities was then broken down into related groups and separated into additional subcategories to increase the specificity of data collection. The final list of activities was reviewed by several experienced hospitalists to ensure completeness. The data dictionary was then loaded onto a Palm Pilot Tx using WorkStudy+ Plus software. The final activity list consisted of 8 main categories, 32 secondary categories, and 53 tertiary categories (See Appendix). To facilitate comparisons with prior studies, we followed the convention of including the categories of direct and indirect patient care. We defined direct patient care as those activities involving face‐to‐face interaction between the hospitalist and the patient. The more general indirect care category encompassed other categories of activity relevant to the patient's care but not performed in the presence of the patient (ie, professional communication, interaction with the EMR, and other patient related activities like searching for medical knowledge on the Internet or reading telemetry monitors).
Pilot Testing
We trained 6 observers in the use of the data collection tool. Each observer practiced shadowing for more than 20 hours with the tool before collecting study data. During this pilot testing phase we optimized the layout of the tool to facilitate rapid documentation of hospitalist activities and multitasking. Interobserver reliability was confirmed by having 2 observers shadow the same hospitalist for a three hour time period. In all cases, the observers obtained an average interclass correlation coefficient of at least 0.95 with a 95% confidence interval of .85 to 1.0 prior to collecting study data.
Study Design
Data collection occurred between July and September of 2008. A total of 24 hospitalists were observed, each for 2 complete weekday shifts starting at 7 AM and ending between 5 PM and 7 PM. Of note, we only observed hospitalists who were directly caring for patients and not part of a teaching service. Each hospitalist was contacted about the project at least a week prior to any observations and informed consent was obtained. A single observer shadowed a single hospitalist continuously, trading off with a new observer every 3 hours to avoid fatigue. To minimize any observation effect our data collectors were instructed not to initiate and to minimize conversation with the hospitalists. At the end of the hospitalist's shift the following data were tallied: the number of patients in the hospitalist's care at the beginning of the day, the number of patients discharged during the day, and the number of admissions. Patient load was determined by adding the number of admissions to the number of patients at the beginning of the day.
Data Analysis
Minutes were tallied for each of the categories and subcategories. Data is reported as percentages of total duration of observed activities (ie, including multitasking) unless otherwise specified. To explore the effect of patient volume on hospitalist workflow we performed t‐tests comparing the number of minutes hospitalists spent per patient in various activities on days with below average patient volume as compared to those with above average volume. Additionally, we performed a Wilcoxon two‐samples test to check for a difference in length of shift between these 2 groups.
Results
A total of 24 hospitalists were shadowed for a total of approximately 494 hours. For 43 of these hours a hospitalist was observed performing 2 tasks simultaneously, bringing the total duration of observed activities to 537 hours with multitasking. The hospitalists were a mean 34 1.1 years of age and 12 (50%) were female. Twenty (83%) had completed residency 2 or more years prior to the study, 2 (8%) had a year of hospitalist experience since residency, and the remaining 2 (8%) had just completed residency. Sixteen (67%) hospitalists were Asian or Pacific Islanders, 6 (25%) were White, and 2 (8%) were Black. The hospitalists cared for an average of 13.2 0.6 patients per shift and an average shift lasted 10 hours and 19 minutes 52 minutes.
Table 1 lists the mean percentage of time hospitalists spent on the various activities. Subjects spent the most time (34.1%) interacting with the EMR. Communication and direct care were the next most frequent activities at 25.9% and 17.4% of each shift respectively, followed by professional development (6.5%), travel (6.2%), personal time (5.6%), other indirect care (3.9%), and waiting (0.4%). The 3 subcategories included in indirect care time accounted for about 64% of all recorded activities.
| Main Category | % Total Observed Activities | (95% CI)* | Subcategory | % Main Category | (95% CI)* |
|---|---|---|---|---|---|
| |||||
| EMR* | 34.1 | (32.435.9) | |||
| Writing | 58.4 | (55.761.0) | |||
| Orders | 20.2 | (18.521.9) | |||
| Reading/reviewing | 19.4 | (17.321.5) | |||
| Other | 2.1 | (1.82.5) | |||
| Communication* | 25.9 | (24.427.4) | |||
| Outgoing call | 36.9 | (33.640.2) | |||
| Face to face | 28.1 | (25.231.0) | |||
| Incoming call | 14.4 | (12.616.3) | |||
| Sending page | 8.6 | (7.79.4) | |||
| Rounds | 3.8 | (1.85.8) | |||
| Receiving page | 3.4 | (2.94.0) | |||
| E‐mail | 2.9 | (1.83.9) | |||
| Reviewing page | 1.8 | (1.32.3) | |||
| Fax | 0.1 | (0.00.2) | |||
| Direct care | 17.4 | (15.918.9) | |||
| Professional Development | 6.5 | (4.48.5) | |||
| Travel | 6.2 | (5.66.7) | |||
| Personal | 5.7 | (4.17.2) | |||
| Other indirect care* | 3.9 | (3.44.4) | |||
| Wait | 0.4 | (0.20.5) | |||
Of the nearly 4 hours (233 minutes) per shift hospitalists spent using the EMR, the majority (58.4%) was spent documenting (See Table 1). Placing orders and reading/reviewing notes were nearly equal at 20.2% and 19.4% respectively, and other EMR activities took 2.1% of EMR time. Over half of the time (54.1%) hospitalists spent documenting in the EMR system was dedicated to progress notes. The remainder of effort was expended on writing histories and physicals (15.3%), discharge instructions (14.7%), discharge summaries (7.9%), sign‐outs (6.8%), and performing medication reconciliation (1.4%). Of the time spent reading and reviewing documents on the EMR, most was spent reviewing lab results (45.4%) or notes from the current admission (40.4%). Reviewing imaging studies occupied 8.1%, and notes from past encounters accounted for 6.2% of this category's time.
Various modes of communication were used during the nearly three hours (176 minutes) per shift dedicated to communication. Phone calls took up approximately half of the hospitalists' communication time, with 36.8% spent on outgoing calls and 14.2% incoming calls. Face‐to‐face communication was the next most common mode, accounting for 28.2% of the total. Time spent sending pages (8.8%), receiving pages (3.4%), and reviewing pages (1.8%) consumed 14% of all communication time. E‐mail and fax were used sparingly, at 3.1% and 0.1% of communication time, respectively. Finally, meetings involving other hospital staff (interdisciplinary rounds) occupied 3.4% of communication time.
The amount of time hospitalists spent communicating with specific types of individuals is shown in Table 2. Hospitalists spent the most time communicating with other physicians (44.5%) and nurses (18.1%). They spent less time communicating with people from the remaining categories; utilization staff (5.7%), patients' family members (5.6%), case managers (4.2%), primary care physicians (3.4%), ancillary staff (3.1%), and pharmacists (0.6%). Communication with other individuals that did not fit in the above categories accounted for 8.8%, and 5.3% of communication could not be clearly categorized, generally because the hospitalist was communicating by phone or text page and ascertaining with whom would have required significant interruption.
| Subcategory | % Main Category | (95% CI)* |
|---|---|---|
| ||
| Inpatient physician | 44.5 | (41.747.2) |
| Nursing staff | 18.0 | (16.019.9) |
| Other | 8.5 | (6.810.2) |
| Family | 5.8 | (4.07.7) |
| Utilization staff | 5.8 | (4.67.0) |
| Uncategorized | 5.7 | (3.77.6) |
| PCC | 4.0 | (2.35.7) |
| PCP | 3.6 | (2.74.5) |
| Ancillary staff | 2.9 | (2.23.7) |
| Pharmacy | 1.4 | (0.82.0) |
We found that 16% of all recorded activities occurred when another activity was also ongoing. This means that hospitalists were performing more than one activity for approximately 54 minutes per day, or about 9% of the average 10.3‐hour shift. Instances of multitasking occurred frequently, but were usually brief; the hospitalists performed 2 activities simultaneously an average of 75 times per day, but 79% of these occurrences lasted less than 1 minute. Of the 86 hours of multitasking activities recorded, 41% was communication time and another 41% was EMR use. This means that a second activity was being performed during 19% of the time hospitalists spent using the EMR and 26% of the time they spent communicating. Of the time spent on critical documentation activities like writing prescriptions and orders, 24% was recorded during a multitasking event.
The amount of time hospitalists spent per patient on days with above average patient volume as compared to those with below average patient volume is shown in Table 3. Hospitalists with above average patient numbers spent about 3 minutes less per patient interacting with the EMR (a 17% reduction; P < 0.01), and about 2 minutes less per patient communicating (a 14% reduction; P < 0.01). The average length of shift increased by 12 minutes on days when patient volume was above average; P < 0.05.
| Subcategory | Minutes: Below Average Census | (95% CI)* | Minutes: Above Average Census | (95% CI)* | Pr > |t| |
|---|---|---|---|---|---|
| |||||
| EMR | 19.12 | (17.5020.75) | 15.83 | (14.1717.49) | <.001 |
| Communication | 14.28 | (12.8615.71) | 12.21 | (11.0713.36) | 0.002 |
| Direct care | 9.30 | (8.1810.42) | 8.59 | (7.279.91) | 0.293 |
| Professional development | 4.09 | (2.365.81) | 2.57 | (1.263.89) | 0.026 |
| Personal | 3.52 | (2.394.65) | 2.05 | (1.292.82) | 0.032 |
| Travel | 3.32 | (2.863.79) | 2.93 | (2.643.22) | 0.566 |
| Other indirect care | 2.37 | (1.902.84) | 1.65 | (1.321.98) | 0.292 |
| Wait | 0.25 | (0.080.41) | 0.14 | (0.040.25) | 0.881 |
Discussion
To our knowledge, this study represents the largest time‐motion evaluation of hospitalist activities ever undertaken, and provides the most detailed assessment of hospitalists' activities when caring for patients without residents or medical students. We confirmed that hospitalists spend the majority of their time (64%) undertaking care activities away from the patient's bedside, and are involved in direct patient care contact only 17% of their time, averaging about 9 minutes per patient. The hospitalists spent about a quarter (26%) of their time communicating with others. Compared to other physicians, this is an unusually large amount of time. For example, Hollingsworth et al.15 found that emergency medicine physicians spent just half as much (13%) of their time on communication with other providers and staff. This may reflect hospitalists' central role in the coordination of consulting specialists. The other significant portion of hospitalists' effort focuses on documentation in the electronic medical record, with 22% of their time required for CPOE and note writing, and overall a third of their time (34.1%) committed to interacting with the EMR.
In many respects, our results confirm the findings of O'Leary et al.'s previous work. While this current study more precisely identified how hospitalists spend their time, the general proportions of times were similar. Both studies found that indirect care activities occupied about two‐thirds of hospitalists' time (64% in this study and 69% in the previous study). We also documented similar portions of total time for direct patient care (17% vs. 18%) and communication (26% vs. 24%). Interestingly, with complete implementation of the EMR system, the percentage of time spent on documentation appeared to decrease. O'Leary et al. reported that documentation accounted for 26% of hospitalists' time, while the equivalent activities (writing in the EMR or paper prescriptions) accounted for only 21% in the current study. Unfortunately, the significance of this finding is difficult to determine given the concurrent changes in patient volumes and the varying extent of EMR implementation during the earlier study.
Over half of hospitalists' communication time is spent either making or receiving phone calls. This suggests that efforts to facilitate communication (eg, use of mobile phone systems and voicemail) might enhance efficiency. Additionally, we found that nearly half of our hospitalists' communication was with other physicians. Not surprisingly, our study confirmed that an important part of hospitalists' work involves organizing and collaborating with a variety of specialists to provide optimal care for their patients.
Hospitalists spent a great deal of time multitasking. We found that multitasking time accounted for nearly 1 of every 10 minutes during the day. The most common combination of activities involved communication that occurred during a period of EMR use. These interruptions could have serious consequences should physicians lose track of what they are doing while ordering procedures or prescribing medications.
We documented a smaller portion of multitasking time than O'Leary's earlier study. This could be due to differences in how multitasking was defined or recorded in the 2 studies. Our electronic data collection tool allowed us to capture rapid task switching and multitasking to the second, rather than to the minute, as was done with the stopwatch and paper form used in the previous study. This precision was important, especially considering that nearly 80% of the recorded instances of multitasking lasted less than 1 minute.
Our data also suggests that patient census has significant effects on certain parts of hospitalist workflow. Patient volume for our subjects ranged from 10 to 19 patients per shift, with a mean of 13.2 patients. The amount of time our hospitalists spent with each patient did not differ significantly between above and below average census days. However, EMR time per patient was significantly reduced on above average census days. Anecdotally, several of our hospitalists suggested that on high census days they put off less time‐sensitive documentation activities like discharge summaries until after they leave the hospital and complete the work from home or on the following day. Thus, our study likely underestimates the total additional effort on high volume days, but unfortunately we had no direct way of quantifying work performed outside of the hospital or on subsequent days. Communication time was also significantly reduced when patient volumes were above average, suggesting that hospitalists had less time to confer with consultants or answer the questions of nurses and patient family members.
Several factors limit the interpretation and application of our findings. First, our study was conducted at a single urban, academic hospital, which may limit its applicability for hospitalists working at community hospitals. Given that more than 90% of hospital care in the U.S. occurs in the community hospital setting, research to confirm these findings in such hospitals is needed.16 Nonclinical research assistants collected all of the data, so the results may be limited by the accuracy of their interpretations. However, our extensive training and documentation of their accuracy serves as a strength of the study. Finally, we focused exclusively on daytime, weekday activities of hospitalists. Notably, 3 hospitalists work through the night at our facility, and 24‐hour coverage by hospitalists is increasingly common across the U.S. We expect weekend and night shift workflow to be somewhat different from standard day shifts due to the decreased availability of other medical providers for testing, consults, and procedures. Future research should focus on potential differences in activities on nights and weekends compared to weekdays.
This extensive, comprehensive analysis of hospitalist activities and workflow provides a foundation for future research and confirms much of O'Leary et al.'s original study. O'Leary's simpler approach of observing smaller blocks of time rather than full shifts proved effective; the two methodologies produced markedly similar results. The current study also offers some insight into matters of efficiency. We found that hospitalists with higher patient loads cut down on EMR and communication time. We also confirmed that hospitalists spend the largest portion of their time interacting with the EMR. A more efficient EMR system could therefore be especially helpful in providing more time for direct patient care and the communication necessary to coordinate care. Given that most hospitals provide financial support for hospital medicine programs (an average of $95,000 per hospitalist full‐time equivalent (FTE)1), hospital administrators have a keen interest in understanding how hospitalists might be more efficient. For example, if hospitalists could evaluate and manage two additional patients each day by exchanging time focused on medical record documentation for direct care activities, the cost of a hospitalist drops substantively. By understanding current hospitalist activities, efforts at redesigning their workflow can be more successful at addressing issues related to scheduling, communication, and compensation, thus improving the overall model of practice as well as the quality of patient care.17
Acknowledgements
We thank Caitlin Lawes and Stephen Williams for help with data collection, and all the hospitalists who participated in this study.
Hospital Medicine represents the fastest‐growing specialty in the history of medicine in the United States, with approximately 28,000 hospitalists now working in over half of American hospitals.1 Hospitalists increasingly fill the gap between demand for care of hospitalized patients and the deficit of physicians previously availableprimary care physicians in community hospitals and residents in teaching hospitals.2, 3 This growth has also been driven by hospitalists' ability to increase clinical efficiency. Research consistently demonstrates a reduction in overall costs and length of stay with the use of hospitalists.47 Additionally, many teaching hospitals have implemented nonteaching hospitalist services in an effort to comply with the Accreditation Council for Graduate Medicine Education (ACGME) program requirements regarding resident duty hours.8 Given the potential for improved clinical efficiency and the need to comply with revised ACGME program requirements, the Hospital Medicine Service at Northwestern Memorial Hospital (NMH) was established in 2003. Today, this service cares for more than half of hospitalized medical patients at NMH.
Although extensive research documents that implementation of a hospitalist program improves the efficiency of hospital care delivery,4, 6 there is little data to explain how hospitalists achieve this level of efficiency or how efficiency might be increased further. Several authors have suggested potential explanations for hospitalists' efficiency gains, but none has yet received strong empirical validation.5, 7 The only previously published study to directly observe more than a small portion of the activities of hospitalists was conducted at NMH in 2006.9 O'Leary et al. used time‐motion methodology to study ten hospitalists for 75 hours total. They found that hospitalists spend a large amount of time on communication when compared to nonhospitalist physicians. However, the study only reported partial information about how and with whom this communications was performed. Similarly, the authors reported that documentation occupied about a quarter of hospitalists' time, but did not report more detailed information about what was being documented and how. Additionally, they noted that hospitalists spent 21% of their time multitasking, but did not report what types of activities were performed during these episodes. Finally, at the time of that study hospitalists at NMH saw about 40% fewer patients per day than they do now. Increasing the number of patients each physician sees in a day is an obvious way to increase productivity, but it is unclear how this affects hospitalist workflow and time spent in various clinical activities.
Another important trend in hospital care delivery is the implementation of electronic medical records (EMR).10 NMH was just transitioning to a fully integrated EMR and computerized physician order entry (CPOE) system when the previous time‐motion study was performed. Now that the system is in place, a significant proportion of hospitalists' time has shifted from using a paper‐based record to sitting in front of a computer. However, we do not know exactly how hospitalists interact with the EMR and how this alters workflow; an increasingly important issue as hospitals across the U.S. implement EMRs at the behest of the federal government and aiming to improve patient safety.11
To better understand the workflow of hospitalists and validate the findings of the O'Leary study in a larger sample of hospitalists, we undertook this study seeking to collect data continuously for complete shifts, rather than sampling just a few hours at a time. We hypothesized that this would reduce observer effects and provide us with a more complete and accurate assessment of a day in the life of a hospitalist.
Methods
Study Site
The study was conducted at NMH, an 897‐bed tertiary care teaching hospital in Chicago, IL, and was approved by the Institutional Review Board of Northwestern University. Patients are admitted to the Hospital Medicine Service from the Emergency Department or directly from physicians' offices based on bed availability in a quasi‐randomized fashion. Hospitalists included in the study cared for patients without the assistance of housestaff physicians and worked 7 consecutive days while on service, usually followed by 7 consecutive days off service. During weeks on service, hospitalist shifts started at 7 AM and ended between 5 PM and 7 PM.
Data Collection Tool Development
To facilitate collection of detailed information sought for this study, we developed an electronic data collection tool. A systematic review of the medical literature on time studies performed by our research group indicated a lack of methodological standardization and dissimilar activity categorizations across studies.12 We attempted to develop a standardized method and data collection instrument for future studies, and first created a data dictionary consisting of a list of hospitalist activities and their descriptions. The initial components were drawn from prior time‐motion studies9, 13, 14 and input from experienced hospitalists (KJO and MVW). The activity list was then refined after a preliminary observation period in which five hospitalists were followed for a total of 6 shifts. Observers noted the specific activities being performed by the hospitalists and asked for explanations and clarification when necessary. In order for an activity to be included in the final list, the activity had to be easily observable and identifiable without subjective interpretation from the observer. The preliminary observation period ended once we were satisfied that no new activities were emerging.
The compiled list of activities was then broken down into related groups and separated into additional subcategories to increase the specificity of data collection. The final list of activities was reviewed by several experienced hospitalists to ensure completeness. The data dictionary was then loaded onto a Palm Pilot Tx using WorkStudy+ Plus software. The final activity list consisted of 8 main categories, 32 secondary categories, and 53 tertiary categories (See Appendix). To facilitate comparisons with prior studies, we followed the convention of including the categories of direct and indirect patient care. We defined direct patient care as those activities involving face‐to‐face interaction between the hospitalist and the patient. The more general indirect care category encompassed other categories of activity relevant to the patient's care but not performed in the presence of the patient (ie, professional communication, interaction with the EMR, and other patient related activities like searching for medical knowledge on the Internet or reading telemetry monitors).
Pilot Testing
We trained 6 observers in the use of the data collection tool. Each observer practiced shadowing for more than 20 hours with the tool before collecting study data. During this pilot testing phase we optimized the layout of the tool to facilitate rapid documentation of hospitalist activities and multitasking. Interobserver reliability was confirmed by having 2 observers shadow the same hospitalist for a three hour time period. In all cases, the observers obtained an average interclass correlation coefficient of at least 0.95 with a 95% confidence interval of .85 to 1.0 prior to collecting study data.
Study Design
Data collection occurred between July and September of 2008. A total of 24 hospitalists were observed, each for 2 complete weekday shifts starting at 7 AM and ending between 5 PM and 7 PM. Of note, we only observed hospitalists who were directly caring for patients and not part of a teaching service. Each hospitalist was contacted about the project at least a week prior to any observations and informed consent was obtained. A single observer shadowed a single hospitalist continuously, trading off with a new observer every 3 hours to avoid fatigue. To minimize any observation effect our data collectors were instructed not to initiate and to minimize conversation with the hospitalists. At the end of the hospitalist's shift the following data were tallied: the number of patients in the hospitalist's care at the beginning of the day, the number of patients discharged during the day, and the number of admissions. Patient load was determined by adding the number of admissions to the number of patients at the beginning of the day.
Data Analysis
Minutes were tallied for each of the categories and subcategories. Data is reported as percentages of total duration of observed activities (ie, including multitasking) unless otherwise specified. To explore the effect of patient volume on hospitalist workflow we performed t‐tests comparing the number of minutes hospitalists spent per patient in various activities on days with below average patient volume as compared to those with above average volume. Additionally, we performed a Wilcoxon two‐samples test to check for a difference in length of shift between these 2 groups.
Results
A total of 24 hospitalists were shadowed for a total of approximately 494 hours. For 43 of these hours a hospitalist was observed performing 2 tasks simultaneously, bringing the total duration of observed activities to 537 hours with multitasking. The hospitalists were a mean 34 1.1 years of age and 12 (50%) were female. Twenty (83%) had completed residency 2 or more years prior to the study, 2 (8%) had a year of hospitalist experience since residency, and the remaining 2 (8%) had just completed residency. Sixteen (67%) hospitalists were Asian or Pacific Islanders, 6 (25%) were White, and 2 (8%) were Black. The hospitalists cared for an average of 13.2 0.6 patients per shift and an average shift lasted 10 hours and 19 minutes 52 minutes.
Table 1 lists the mean percentage of time hospitalists spent on the various activities. Subjects spent the most time (34.1%) interacting with the EMR. Communication and direct care were the next most frequent activities at 25.9% and 17.4% of each shift respectively, followed by professional development (6.5%), travel (6.2%), personal time (5.6%), other indirect care (3.9%), and waiting (0.4%). The 3 subcategories included in indirect care time accounted for about 64% of all recorded activities.
| Main Category | % Total Observed Activities | (95% CI)* | Subcategory | % Main Category | (95% CI)* |
|---|---|---|---|---|---|
| |||||
| EMR* | 34.1 | (32.435.9) | |||
| Writing | 58.4 | (55.761.0) | |||
| Orders | 20.2 | (18.521.9) | |||
| Reading/reviewing | 19.4 | (17.321.5) | |||
| Other | 2.1 | (1.82.5) | |||
| Communication* | 25.9 | (24.427.4) | |||
| Outgoing call | 36.9 | (33.640.2) | |||
| Face to face | 28.1 | (25.231.0) | |||
| Incoming call | 14.4 | (12.616.3) | |||
| Sending page | 8.6 | (7.79.4) | |||
| Rounds | 3.8 | (1.85.8) | |||
| Receiving page | 3.4 | (2.94.0) | |||
| E‐mail | 2.9 | (1.83.9) | |||
| Reviewing page | 1.8 | (1.32.3) | |||
| Fax | 0.1 | (0.00.2) | |||
| Direct care | 17.4 | (15.918.9) | |||
| Professional Development | 6.5 | (4.48.5) | |||
| Travel | 6.2 | (5.66.7) | |||
| Personal | 5.7 | (4.17.2) | |||
| Other indirect care* | 3.9 | (3.44.4) | |||
| Wait | 0.4 | (0.20.5) | |||
Of the nearly 4 hours (233 minutes) per shift hospitalists spent using the EMR, the majority (58.4%) was spent documenting (See Table 1). Placing orders and reading/reviewing notes were nearly equal at 20.2% and 19.4% respectively, and other EMR activities took 2.1% of EMR time. Over half of the time (54.1%) hospitalists spent documenting in the EMR system was dedicated to progress notes. The remainder of effort was expended on writing histories and physicals (15.3%), discharge instructions (14.7%), discharge summaries (7.9%), sign‐outs (6.8%), and performing medication reconciliation (1.4%). Of the time spent reading and reviewing documents on the EMR, most was spent reviewing lab results (45.4%) or notes from the current admission (40.4%). Reviewing imaging studies occupied 8.1%, and notes from past encounters accounted for 6.2% of this category's time.
Various modes of communication were used during the nearly three hours (176 minutes) per shift dedicated to communication. Phone calls took up approximately half of the hospitalists' communication time, with 36.8% spent on outgoing calls and 14.2% incoming calls. Face‐to‐face communication was the next most common mode, accounting for 28.2% of the total. Time spent sending pages (8.8%), receiving pages (3.4%), and reviewing pages (1.8%) consumed 14% of all communication time. E‐mail and fax were used sparingly, at 3.1% and 0.1% of communication time, respectively. Finally, meetings involving other hospital staff (interdisciplinary rounds) occupied 3.4% of communication time.
The amount of time hospitalists spent communicating with specific types of individuals is shown in Table 2. Hospitalists spent the most time communicating with other physicians (44.5%) and nurses (18.1%). They spent less time communicating with people from the remaining categories; utilization staff (5.7%), patients' family members (5.6%), case managers (4.2%), primary care physicians (3.4%), ancillary staff (3.1%), and pharmacists (0.6%). Communication with other individuals that did not fit in the above categories accounted for 8.8%, and 5.3% of communication could not be clearly categorized, generally because the hospitalist was communicating by phone or text page and ascertaining with whom would have required significant interruption.
| Subcategory | % Main Category | (95% CI)* |
|---|---|---|
| ||
| Inpatient physician | 44.5 | (41.747.2) |
| Nursing staff | 18.0 | (16.019.9) |
| Other | 8.5 | (6.810.2) |
| Family | 5.8 | (4.07.7) |
| Utilization staff | 5.8 | (4.67.0) |
| Uncategorized | 5.7 | (3.77.6) |
| PCC | 4.0 | (2.35.7) |
| PCP | 3.6 | (2.74.5) |
| Ancillary staff | 2.9 | (2.23.7) |
| Pharmacy | 1.4 | (0.82.0) |
We found that 16% of all recorded activities occurred when another activity was also ongoing. This means that hospitalists were performing more than one activity for approximately 54 minutes per day, or about 9% of the average 10.3‐hour shift. Instances of multitasking occurred frequently, but were usually brief; the hospitalists performed 2 activities simultaneously an average of 75 times per day, but 79% of these occurrences lasted less than 1 minute. Of the 86 hours of multitasking activities recorded, 41% was communication time and another 41% was EMR use. This means that a second activity was being performed during 19% of the time hospitalists spent using the EMR and 26% of the time they spent communicating. Of the time spent on critical documentation activities like writing prescriptions and orders, 24% was recorded during a multitasking event.
The amount of time hospitalists spent per patient on days with above average patient volume as compared to those with below average patient volume is shown in Table 3. Hospitalists with above average patient numbers spent about 3 minutes less per patient interacting with the EMR (a 17% reduction; P < 0.01), and about 2 minutes less per patient communicating (a 14% reduction; P < 0.01). The average length of shift increased by 12 minutes on days when patient volume was above average; P < 0.05.
| Subcategory | Minutes: Below Average Census | (95% CI)* | Minutes: Above Average Census | (95% CI)* | Pr > |t| |
|---|---|---|---|---|---|
| |||||
| EMR | 19.12 | (17.5020.75) | 15.83 | (14.1717.49) | <.001 |
| Communication | 14.28 | (12.8615.71) | 12.21 | (11.0713.36) | 0.002 |
| Direct care | 9.30 | (8.1810.42) | 8.59 | (7.279.91) | 0.293 |
| Professional development | 4.09 | (2.365.81) | 2.57 | (1.263.89) | 0.026 |
| Personal | 3.52 | (2.394.65) | 2.05 | (1.292.82) | 0.032 |
| Travel | 3.32 | (2.863.79) | 2.93 | (2.643.22) | 0.566 |
| Other indirect care | 2.37 | (1.902.84) | 1.65 | (1.321.98) | 0.292 |
| Wait | 0.25 | (0.080.41) | 0.14 | (0.040.25) | 0.881 |
Discussion
To our knowledge, this study represents the largest time‐motion evaluation of hospitalist activities ever undertaken, and provides the most detailed assessment of hospitalists' activities when caring for patients without residents or medical students. We confirmed that hospitalists spend the majority of their time (64%) undertaking care activities away from the patient's bedside, and are involved in direct patient care contact only 17% of their time, averaging about 9 minutes per patient. The hospitalists spent about a quarter (26%) of their time communicating with others. Compared to other physicians, this is an unusually large amount of time. For example, Hollingsworth et al.15 found that emergency medicine physicians spent just half as much (13%) of their time on communication with other providers and staff. This may reflect hospitalists' central role in the coordination of consulting specialists. The other significant portion of hospitalists' effort focuses on documentation in the electronic medical record, with 22% of their time required for CPOE and note writing, and overall a third of their time (34.1%) committed to interacting with the EMR.
In many respects, our results confirm the findings of O'Leary et al.'s previous work. While this current study more precisely identified how hospitalists spend their time, the general proportions of times were similar. Both studies found that indirect care activities occupied about two‐thirds of hospitalists' time (64% in this study and 69% in the previous study). We also documented similar portions of total time for direct patient care (17% vs. 18%) and communication (26% vs. 24%). Interestingly, with complete implementation of the EMR system, the percentage of time spent on documentation appeared to decrease. O'Leary et al. reported that documentation accounted for 26% of hospitalists' time, while the equivalent activities (writing in the EMR or paper prescriptions) accounted for only 21% in the current study. Unfortunately, the significance of this finding is difficult to determine given the concurrent changes in patient volumes and the varying extent of EMR implementation during the earlier study.
Over half of hospitalists' communication time is spent either making or receiving phone calls. This suggests that efforts to facilitate communication (eg, use of mobile phone systems and voicemail) might enhance efficiency. Additionally, we found that nearly half of our hospitalists' communication was with other physicians. Not surprisingly, our study confirmed that an important part of hospitalists' work involves organizing and collaborating with a variety of specialists to provide optimal care for their patients.
Hospitalists spent a great deal of time multitasking. We found that multitasking time accounted for nearly 1 of every 10 minutes during the day. The most common combination of activities involved communication that occurred during a period of EMR use. These interruptions could have serious consequences should physicians lose track of what they are doing while ordering procedures or prescribing medications.
We documented a smaller portion of multitasking time than O'Leary's earlier study. This could be due to differences in how multitasking was defined or recorded in the 2 studies. Our electronic data collection tool allowed us to capture rapid task switching and multitasking to the second, rather than to the minute, as was done with the stopwatch and paper form used in the previous study. This precision was important, especially considering that nearly 80% of the recorded instances of multitasking lasted less than 1 minute.
Our data also suggests that patient census has significant effects on certain parts of hospitalist workflow. Patient volume for our subjects ranged from 10 to 19 patients per shift, with a mean of 13.2 patients. The amount of time our hospitalists spent with each patient did not differ significantly between above and below average census days. However, EMR time per patient was significantly reduced on above average census days. Anecdotally, several of our hospitalists suggested that on high census days they put off less time‐sensitive documentation activities like discharge summaries until after they leave the hospital and complete the work from home or on the following day. Thus, our study likely underestimates the total additional effort on high volume days, but unfortunately we had no direct way of quantifying work performed outside of the hospital or on subsequent days. Communication time was also significantly reduced when patient volumes were above average, suggesting that hospitalists had less time to confer with consultants or answer the questions of nurses and patient family members.
Several factors limit the interpretation and application of our findings. First, our study was conducted at a single urban, academic hospital, which may limit its applicability for hospitalists working at community hospitals. Given that more than 90% of hospital care in the U.S. occurs in the community hospital setting, research to confirm these findings in such hospitals is needed.16 Nonclinical research assistants collected all of the data, so the results may be limited by the accuracy of their interpretations. However, our extensive training and documentation of their accuracy serves as a strength of the study. Finally, we focused exclusively on daytime, weekday activities of hospitalists. Notably, 3 hospitalists work through the night at our facility, and 24‐hour coverage by hospitalists is increasingly common across the U.S. We expect weekend and night shift workflow to be somewhat different from standard day shifts due to the decreased availability of other medical providers for testing, consults, and procedures. Future research should focus on potential differences in activities on nights and weekends compared to weekdays.
This extensive, comprehensive analysis of hospitalist activities and workflow provides a foundation for future research and confirms much of O'Leary et al.'s original study. O'Leary's simpler approach of observing smaller blocks of time rather than full shifts proved effective; the two methodologies produced markedly similar results. The current study also offers some insight into matters of efficiency. We found that hospitalists with higher patient loads cut down on EMR and communication time. We also confirmed that hospitalists spend the largest portion of their time interacting with the EMR. A more efficient EMR system could therefore be especially helpful in providing more time for direct patient care and the communication necessary to coordinate care. Given that most hospitals provide financial support for hospital medicine programs (an average of $95,000 per hospitalist full‐time equivalent (FTE)1), hospital administrators have a keen interest in understanding how hospitalists might be more efficient. For example, if hospitalists could evaluate and manage two additional patients each day by exchanging time focused on medical record documentation for direct care activities, the cost of a hospitalist drops substantively. By understanding current hospitalist activities, efforts at redesigning their workflow can be more successful at addressing issues related to scheduling, communication, and compensation, thus improving the overall model of practice as well as the quality of patient care.17
Acknowledgements
We thank Caitlin Lawes and Stephen Williams for help with data collection, and all the hospitalists who participated in this study.
- Society of Hospital Medicine. About SHM.2008; http://www.hospitalmedicine.org/AM/Template.cfm?Section=About_SHM. Accessed April 2010.
- , .The evolution and future of hospital medicine.Mt Sinai J Med.2008;75(5):418–423.
- , , .Rehospitalizations among patients in the Fee‐for‐Service Medicare Program.N Engl J Med.2009;360(14):1418–1428.
- , , , , , .Outcomes of care by hospitalists, general internists, and family physicians.[see comment].N Engl J Med.2007;357(25):2589–2600.
- , .The hospitalist movement 5 years later.JAMA.2002;287:487–494.
- , .The impact of hospitalists on the cost and quality of inpatient care in the United States: a research synthesis.Med Care Res Rev.2005;62:379–406.
- .Hospitalists and the hospital medicine system of care are good for patient care.Arch Intern Med.2008;168(12):1254–1256; discussion 1259–1260.
- , .Hospitalists in teaching hospitals: opportunities but not without danger.J Gen Intern Med.2004;19:392–393.
- , , .How hospitalists spend their time: insights on efficiency and safety.J Hosp Med.2006;1(2):88–93.
- , , , et al.Use of electronic health records in U.S. hospitals.N Engl J Med.2009;360.
- .Electronic medical records at a crossroads: impetus for change or missed opportunity?JAMA.2009;302(10):1109–1111.
- , , , , .Systematic review of time studies evaluating physicians in the hospital setting.J Hosp Med.2010;5(6):000–000.
- , , , .All in a day's work: an observational study to quantify how and with whom doctors on hospital wards spend their time.Med J Aust.2008;188(9):506–509.
- , , , .Emergency department workplace interruptions: are emergency physicians “interrupt‐driven” and “multitasking”?Acad Emerg Med.2000;7:1239–1243.
- , , , , .How do physicians and nurses spend their time in the emergency department?Ann Emerg Med.1998;31(1):87–91.
- , , , , .The ecology of medical care revisited.N Engl J Med.2001;344(26):2021–2025.
- , .Organizing a hospitalist program: an overview of fundamental concepts.Med Clin North Am.2002;86(4):887–909.
- Society of Hospital Medicine. About SHM.2008; http://www.hospitalmedicine.org/AM/Template.cfm?Section=About_SHM. Accessed April 2010.
- , .The evolution and future of hospital medicine.Mt Sinai J Med.2008;75(5):418–423.
- , , .Rehospitalizations among patients in the Fee‐for‐Service Medicare Program.N Engl J Med.2009;360(14):1418–1428.
- , , , , , .Outcomes of care by hospitalists, general internists, and family physicians.[see comment].N Engl J Med.2007;357(25):2589–2600.
- , .The hospitalist movement 5 years later.JAMA.2002;287:487–494.
- , .The impact of hospitalists on the cost and quality of inpatient care in the United States: a research synthesis.Med Care Res Rev.2005;62:379–406.
- .Hospitalists and the hospital medicine system of care are good for patient care.Arch Intern Med.2008;168(12):1254–1256; discussion 1259–1260.
- , .Hospitalists in teaching hospitals: opportunities but not without danger.J Gen Intern Med.2004;19:392–393.
- , , .How hospitalists spend their time: insights on efficiency and safety.J Hosp Med.2006;1(2):88–93.
- , , , et al.Use of electronic health records in U.S. hospitals.N Engl J Med.2009;360.
- .Electronic medical records at a crossroads: impetus for change or missed opportunity?JAMA.2009;302(10):1109–1111.
- , , , , .Systematic review of time studies evaluating physicians in the hospital setting.J Hosp Med.2010;5(6):000–000.
- , , , .All in a day's work: an observational study to quantify how and with whom doctors on hospital wards spend their time.Med J Aust.2008;188(9):506–509.
- , , , .Emergency department workplace interruptions: are emergency physicians “interrupt‐driven” and “multitasking”?Acad Emerg Med.2000;7:1239–1243.
- , , , , .How do physicians and nurses spend their time in the emergency department?Ann Emerg Med.1998;31(1):87–91.
- , , , , .The ecology of medical care revisited.N Engl J Med.2001;344(26):2021–2025.
- , .Organizing a hospitalist program: an overview of fundamental concepts.Med Clin North Am.2002;86(4):887–909.
Copyright © 2010 Society of Hospital Medicine