User login
ACA lawsuits progress through the courts
While Republican efforts to repeal and/or replace the Affordable Care Act are likely to resurface after the congressional summer recess, lawsuits challenging the health law continue to wind their way through the courts.
Judges recently ruled in three ACA cases regarding the contraceptive mandate, risk corridors program, and cost-sharing reduction payments.
Real Alternatives Inc. v. HHS
Secular groups are not entitled to a religious exemption to the ACA’s contraceptive mandate and must offer plans to employees that cover birth control, the United States Court of Appeals for the Third Circuit ruled in an Aug. 4 opinion.
But a majority of appeal judges disagreed, concluding that Real Alternatives is not similar to a religious denomination or one of its nontheistic counterparts, “not in structure, not in aim, not in purpose, and not in function.”
“We do not doubt that Real Alternative’s stance on contraceptives is grounded in sincerely-held moral values, but religion is not generally confined to one question or one moral teaching, it has a broader scope,” Judge Marjorie O. Rendell wrote in the majority opinion. “Real Alternatives is functionally similar not to a church, but to the countless nonreligious nonprofit organizations that take morally informed positions on some discrete set of issues ... While commitment to an anti-abortion platform may be important to the people who hold them, that commitment is not a religion in any legally or theologically accepted sense; and organizations do not become quasi-churches for equal-protection purposes merely by espousing a commitment of that sort.”
Molina Healthcare v. HHS
The federal Heath & Human Services department may have to dish out millions to marketplace insurers after a recent decision by the U.S. Court of Federal Claims. In an Aug. 4 opinion, a federal claims judge ruled the federal government owes Long Beach, Calif.–based insurer Molina Healthcare $52 million in risk corridor payments under the ACA.
The decision stems from a lawsuit by Molina and dozens of other insurers against HHS over the ACA’s risk corridor program. The program requires HHS to collect funds from excessively profitable insurers that offer qualified health plans (QHP) under the exchanges, while paying out funds to QHP insurers with excessive losses. Collections from profitable insurers under the program fell short in 2014 and again in 2015, resulting in HHS paying about 12 cents on the dollar in payments to insurers.
Insurers allege they’ve been shortchanged and that the government must reimburse them full payments for 2014 and 2015. Under the Obama administration, the Department of Justice requested to dismiss the cases, arguing that riders attached to appropriations bills in 2015 and 2016 barred it from making full risk corridor payments.
In his decision, Judge Thomas Wheeler said the ACA mandates that HHS pay insurers what they are owed under the risk corridor program, regardless of the riders.
“The undisputed facts show the government entered into an implied-in-fact contract with Molina and subsequently breached the contract when it failed to make full risk corridor payments,” Judge Wheeler wrote. “Importantly, Molina prevails on its argument of breach of an implied-in-fact contract regardless of the government’s appropriation law defenses – later appropriation restrictions cannot erase a previously created contractual obligation ...The government is liable for its breach of a statutory and contractual obligation to make full annual payments to insurers who participated in the risk corridor program.”
Judge Wheeler ruled similarly in February when he concluded the federal government owes Portland, Ore.–based Moda Health $214 million in risk corridor payments.
The issue is far from over. More than 25 lawsuits have been filed in the Court of Federal Claims by insurers seeking risk corridor money. Several of the cases have moved onto federal appeals courts, while other cases have been dismissed. Analysts say the issue is likely to reach the U.S. Supreme Court. It’s unclear where the Trump administration would get the funds to reimburse insurers if the payback rulings stand. About $19.3 billion in risk corridor payments are at stake for 2014 and 2015 and an estimated $3 billion is in play for 2016, the final risk corridor year.
House v. Price
A U.S. appeals court has allowed 16 states to intervene in a lawsuit over whether subsidy payments made to insurers under the ACA are legal. The decision comes as President Trump recently threatened to cut off the cost sharing reduction (CSR) payments to marketplace insurers, calling them a “bailout.”
On Aug. 1, the U.S. Court of Appeals for the District of Columbia Circuit granted a motion by 18 attorneys general to enter the lawsuit. The attorneys general represent California, New York, Connecticut, Delaware, Hawaii, Illinois, Iowa, Kentucky, Maryland, Massachusetts, Minnesota, New Mexico, North Carolina, Pennsylvania, Vermont, Virginia, Washington, and the District of Columbia.
Under the ACA, the federal government provides CSR payments to insurers to offset the costs for providing discount plans to patients who earn up to 200% of the federal poverty level. Plans on the individual exchanges are required to cover a package of essential benefits with pricing limitations to ensure that out-of-pocket costs are low enough for low-income patients.
Republican members of the House of Representatives sued the HHS over the CSR payments under the Obama administration, claiming the funding was illegal because it was never appropriated by Congress. A court ruled in favor of the House in 2016, but an appeal filed by the Obama administration allowed the CSR payments to continue. President Trump has not indicated whether he plans to drop the appeal or carry on the case. But if he fails to continue the suit, the move would immediately end the CSR payments.
On July 29, President Trump tweeted, “If a new HealthCare Bill is not approved quickly, BAILOUTS for Insurance Companies and BAILOUTS for Members of Congress will end very soon!”
The state attorneys general are ready to defend the ACA and the cost sharing reduction payments, California Attorney General Xavier Becerra said in a statement.
“If Donald Trump won’t defend these vital subsidies for American families, then we will,” Mr. Becerra said in the statement. “This ruling gives my fellow attorneys general and me the ability to stand up for the millions of families who otherwise would lack access to affordable health care. It’s time Americans knew we were working for them, not against them.”
[email protected]
On Twitter @legal_med
While Republican efforts to repeal and/or replace the Affordable Care Act are likely to resurface after the congressional summer recess, lawsuits challenging the health law continue to wind their way through the courts.
Judges recently ruled in three ACA cases regarding the contraceptive mandate, risk corridors program, and cost-sharing reduction payments.
Real Alternatives Inc. v. HHS
Secular groups are not entitled to a religious exemption to the ACA’s contraceptive mandate and must offer plans to employees that cover birth control, the United States Court of Appeals for the Third Circuit ruled in an Aug. 4 opinion.
But a majority of appeal judges disagreed, concluding that Real Alternatives is not similar to a religious denomination or one of its nontheistic counterparts, “not in structure, not in aim, not in purpose, and not in function.”
“We do not doubt that Real Alternative’s stance on contraceptives is grounded in sincerely-held moral values, but religion is not generally confined to one question or one moral teaching, it has a broader scope,” Judge Marjorie O. Rendell wrote in the majority opinion. “Real Alternatives is functionally similar not to a church, but to the countless nonreligious nonprofit organizations that take morally informed positions on some discrete set of issues ... While commitment to an anti-abortion platform may be important to the people who hold them, that commitment is not a religion in any legally or theologically accepted sense; and organizations do not become quasi-churches for equal-protection purposes merely by espousing a commitment of that sort.”
Molina Healthcare v. HHS
The federal Heath & Human Services department may have to dish out millions to marketplace insurers after a recent decision by the U.S. Court of Federal Claims. In an Aug. 4 opinion, a federal claims judge ruled the federal government owes Long Beach, Calif.–based insurer Molina Healthcare $52 million in risk corridor payments under the ACA.
The decision stems from a lawsuit by Molina and dozens of other insurers against HHS over the ACA’s risk corridor program. The program requires HHS to collect funds from excessively profitable insurers that offer qualified health plans (QHP) under the exchanges, while paying out funds to QHP insurers with excessive losses. Collections from profitable insurers under the program fell short in 2014 and again in 2015, resulting in HHS paying about 12 cents on the dollar in payments to insurers.
Insurers allege they’ve been shortchanged and that the government must reimburse them full payments for 2014 and 2015. Under the Obama administration, the Department of Justice requested to dismiss the cases, arguing that riders attached to appropriations bills in 2015 and 2016 barred it from making full risk corridor payments.
In his decision, Judge Thomas Wheeler said the ACA mandates that HHS pay insurers what they are owed under the risk corridor program, regardless of the riders.
“The undisputed facts show the government entered into an implied-in-fact contract with Molina and subsequently breached the contract when it failed to make full risk corridor payments,” Judge Wheeler wrote. “Importantly, Molina prevails on its argument of breach of an implied-in-fact contract regardless of the government’s appropriation law defenses – later appropriation restrictions cannot erase a previously created contractual obligation ...The government is liable for its breach of a statutory and contractual obligation to make full annual payments to insurers who participated in the risk corridor program.”
Judge Wheeler ruled similarly in February when he concluded the federal government owes Portland, Ore.–based Moda Health $214 million in risk corridor payments.
The issue is far from over. More than 25 lawsuits have been filed in the Court of Federal Claims by insurers seeking risk corridor money. Several of the cases have moved onto federal appeals courts, while other cases have been dismissed. Analysts say the issue is likely to reach the U.S. Supreme Court. It’s unclear where the Trump administration would get the funds to reimburse insurers if the payback rulings stand. About $19.3 billion in risk corridor payments are at stake for 2014 and 2015 and an estimated $3 billion is in play for 2016, the final risk corridor year.
House v. Price
A U.S. appeals court has allowed 16 states to intervene in a lawsuit over whether subsidy payments made to insurers under the ACA are legal. The decision comes as President Trump recently threatened to cut off the cost sharing reduction (CSR) payments to marketplace insurers, calling them a “bailout.”
On Aug. 1, the U.S. Court of Appeals for the District of Columbia Circuit granted a motion by 18 attorneys general to enter the lawsuit. The attorneys general represent California, New York, Connecticut, Delaware, Hawaii, Illinois, Iowa, Kentucky, Maryland, Massachusetts, Minnesota, New Mexico, North Carolina, Pennsylvania, Vermont, Virginia, Washington, and the District of Columbia.
Under the ACA, the federal government provides CSR payments to insurers to offset the costs for providing discount plans to patients who earn up to 200% of the federal poverty level. Plans on the individual exchanges are required to cover a package of essential benefits with pricing limitations to ensure that out-of-pocket costs are low enough for low-income patients.
Republican members of the House of Representatives sued the HHS over the CSR payments under the Obama administration, claiming the funding was illegal because it was never appropriated by Congress. A court ruled in favor of the House in 2016, but an appeal filed by the Obama administration allowed the CSR payments to continue. President Trump has not indicated whether he plans to drop the appeal or carry on the case. But if he fails to continue the suit, the move would immediately end the CSR payments.
On July 29, President Trump tweeted, “If a new HealthCare Bill is not approved quickly, BAILOUTS for Insurance Companies and BAILOUTS for Members of Congress will end very soon!”
The state attorneys general are ready to defend the ACA and the cost sharing reduction payments, California Attorney General Xavier Becerra said in a statement.
“If Donald Trump won’t defend these vital subsidies for American families, then we will,” Mr. Becerra said in the statement. “This ruling gives my fellow attorneys general and me the ability to stand up for the millions of families who otherwise would lack access to affordable health care. It’s time Americans knew we were working for them, not against them.”
[email protected]
On Twitter @legal_med
While Republican efforts to repeal and/or replace the Affordable Care Act are likely to resurface after the congressional summer recess, lawsuits challenging the health law continue to wind their way through the courts.
Judges recently ruled in three ACA cases regarding the contraceptive mandate, risk corridors program, and cost-sharing reduction payments.
Real Alternatives Inc. v. HHS
Secular groups are not entitled to a religious exemption to the ACA’s contraceptive mandate and must offer plans to employees that cover birth control, the United States Court of Appeals for the Third Circuit ruled in an Aug. 4 opinion.
But a majority of appeal judges disagreed, concluding that Real Alternatives is not similar to a religious denomination or one of its nontheistic counterparts, “not in structure, not in aim, not in purpose, and not in function.”
“We do not doubt that Real Alternative’s stance on contraceptives is grounded in sincerely-held moral values, but religion is not generally confined to one question or one moral teaching, it has a broader scope,” Judge Marjorie O. Rendell wrote in the majority opinion. “Real Alternatives is functionally similar not to a church, but to the countless nonreligious nonprofit organizations that take morally informed positions on some discrete set of issues ... While commitment to an anti-abortion platform may be important to the people who hold them, that commitment is not a religion in any legally or theologically accepted sense; and organizations do not become quasi-churches for equal-protection purposes merely by espousing a commitment of that sort.”
Molina Healthcare v. HHS
The federal Heath & Human Services department may have to dish out millions to marketplace insurers after a recent decision by the U.S. Court of Federal Claims. In an Aug. 4 opinion, a federal claims judge ruled the federal government owes Long Beach, Calif.–based insurer Molina Healthcare $52 million in risk corridor payments under the ACA.
The decision stems from a lawsuit by Molina and dozens of other insurers against HHS over the ACA’s risk corridor program. The program requires HHS to collect funds from excessively profitable insurers that offer qualified health plans (QHP) under the exchanges, while paying out funds to QHP insurers with excessive losses. Collections from profitable insurers under the program fell short in 2014 and again in 2015, resulting in HHS paying about 12 cents on the dollar in payments to insurers.
Insurers allege they’ve been shortchanged and that the government must reimburse them full payments for 2014 and 2015. Under the Obama administration, the Department of Justice requested to dismiss the cases, arguing that riders attached to appropriations bills in 2015 and 2016 barred it from making full risk corridor payments.
In his decision, Judge Thomas Wheeler said the ACA mandates that HHS pay insurers what they are owed under the risk corridor program, regardless of the riders.
“The undisputed facts show the government entered into an implied-in-fact contract with Molina and subsequently breached the contract when it failed to make full risk corridor payments,” Judge Wheeler wrote. “Importantly, Molina prevails on its argument of breach of an implied-in-fact contract regardless of the government’s appropriation law defenses – later appropriation restrictions cannot erase a previously created contractual obligation ...The government is liable for its breach of a statutory and contractual obligation to make full annual payments to insurers who participated in the risk corridor program.”
Judge Wheeler ruled similarly in February when he concluded the federal government owes Portland, Ore.–based Moda Health $214 million in risk corridor payments.
The issue is far from over. More than 25 lawsuits have been filed in the Court of Federal Claims by insurers seeking risk corridor money. Several of the cases have moved onto federal appeals courts, while other cases have been dismissed. Analysts say the issue is likely to reach the U.S. Supreme Court. It’s unclear where the Trump administration would get the funds to reimburse insurers if the payback rulings stand. About $19.3 billion in risk corridor payments are at stake for 2014 and 2015 and an estimated $3 billion is in play for 2016, the final risk corridor year.
House v. Price
A U.S. appeals court has allowed 16 states to intervene in a lawsuit over whether subsidy payments made to insurers under the ACA are legal. The decision comes as President Trump recently threatened to cut off the cost sharing reduction (CSR) payments to marketplace insurers, calling them a “bailout.”
On Aug. 1, the U.S. Court of Appeals for the District of Columbia Circuit granted a motion by 18 attorneys general to enter the lawsuit. The attorneys general represent California, New York, Connecticut, Delaware, Hawaii, Illinois, Iowa, Kentucky, Maryland, Massachusetts, Minnesota, New Mexico, North Carolina, Pennsylvania, Vermont, Virginia, Washington, and the District of Columbia.
Under the ACA, the federal government provides CSR payments to insurers to offset the costs for providing discount plans to patients who earn up to 200% of the federal poverty level. Plans on the individual exchanges are required to cover a package of essential benefits with pricing limitations to ensure that out-of-pocket costs are low enough for low-income patients.
Republican members of the House of Representatives sued the HHS over the CSR payments under the Obama administration, claiming the funding was illegal because it was never appropriated by Congress. A court ruled in favor of the House in 2016, but an appeal filed by the Obama administration allowed the CSR payments to continue. President Trump has not indicated whether he plans to drop the appeal or carry on the case. But if he fails to continue the suit, the move would immediately end the CSR payments.
On July 29, President Trump tweeted, “If a new HealthCare Bill is not approved quickly, BAILOUTS for Insurance Companies and BAILOUTS for Members of Congress will end very soon!”
The state attorneys general are ready to defend the ACA and the cost sharing reduction payments, California Attorney General Xavier Becerra said in a statement.
“If Donald Trump won’t defend these vital subsidies for American families, then we will,” Mr. Becerra said in the statement. “This ruling gives my fellow attorneys general and me the ability to stand up for the millions of families who otherwise would lack access to affordable health care. It’s time Americans knew we were working for them, not against them.”
[email protected]
On Twitter @legal_med
Practitioner Cognitive Reframing: Working More Effectively in Addictions
I am a behavioral health-licensed clinical social worker, an approved motivational enhancement therapy provider, and the point of contact for substance use disorders at the Berks CBOC of the Lebanon VAMC in Wyomissing, Pennsylvania. Recently, an exasperated primary care provider at the Berks CBOC approached me about working with patients with substance use disorders and asked “How do you work with this challenging population?”
The question was cause for some introspection. When I started working in addictions, I had to modify my approach to work effectively with this population. I experienced a paradigm shift in which I no longer assigned myself credit or blame for a veteran’s continued sobriety or relapse: Each patient is responsible for his or her progress in the recovery journey. When a patient has a relapse, I remind myself that statistically relapse is a probability for the majority of those in recovery; even multiple relapses are common. Therefore, another way of viewing relapse is that the relapse itself may bring the veteran a step closer to permanent abstinence.
Cognitive Reframing
Consider a toddler learning to walk. Parents and caregivers of the child expect the child to fall quite a few times before he or she has mastered walking. The parents and caregivers don’t get angry, take the situation personally, or feel manipulated by the child’s “failure.” Instead, the parents offer the child emotional support and encouragement.
Arguably, the practitioner should take a similar stance—emotionally supportive and encouraging—in combination with dialogue guided by motivational interviewing to support change and help the veteran get back on track. Although not inevitable, relapse is a normal part of recovery.
Practitioners should avoid either scolding or praising patients. Scolding doesn’t help in the recovery journey, and if the veteran learns that the practitioner reacts by scolding after learning of the relapse, then it is less likely that he or she will be open about future missteps. In fact, the veteran may not return at all. The goal is to have the patient come back when relapse occurs to help him or her progress to recovery.
Too much praise can cause similar problems. A veteran accustomed to praise when he or she is doing well, may be too embarrassed or ashamed to return to ask for help after a relapse. Instead the practitioner should use affirmations, which are widely discussed in motivational interviewing (MI) literature.
“I want to be clean and sober” or “I want to stop drinking” are vague statements that should raise a red flag for experienced practitioners. Is this patient just telling me what he or she thinks I want to hear to respond? That may be the case but also may be an assumption. Instead these assertions could be regarded as global treatment goals, and the task of the practitioner is to help the veteran develop objectives and interventions in relation to this goal. These broad statements can be a starting point in MI. These words can sound just as foreign to a patient who isn’t sure whether becoming clean and sober is a possibility. The veteran may not have the confidence to reach the goal of being clean and sober, and these statements may seem awkward and out of sync with his or her facial expression and body language.
Showing disbelief in a veteran also can have negative consequences. The veteran might feel that “even my therapist/health care provider doesn’t believe I can become clean and sober.” Instead, I remind myself that we all must manipulate our environment for survival. I find it more valuable to think of the veteran as being resourceful rather than manipulative.
This point may seem self-evident, but it took me a while to catch on: Most of the change in the recovery journey transpires outside the practitioner’s office. I had to embrace this truism and be prepared when the veteran returned for the next session. The task, then, is to determine at this moment where the veteran is in his or her recovery journey instead of continuing the conversation from the previous session. The previous session may be irrelevant. Thinking this way was an adjustment for me. One of my favorite therapy approaches was to consciously continue a conversation from the previous session to demonstrate that I remembered what the patient had said, thereby showing that I care.
In addition, get to know the patient underneath, behind, and before the substance use disorder. Knowing and liking the veteran helps me avoid burnout, bringing me back to my values and the reason I became a therapist. I make efforts in my thinking process to convert “alcoholic” or “drug abuser” into a more helpful “client addicted to alcohol” or “client with a substance use disorder.” The veteran should not be labeled.
Motivational Interviewing
Oftentimes what propels veterans forward in their recovery is the cognitive dissonance created between who they were and how they acted before substance addiction and how they act now. Conversation steered in this direction, fueled by MI, can enhance a veteran’s motivation to change and ready the veteran to change behaviors. Listen to the veteran’s account of loved ones and remember the names of these important family members and friends. Weave into the conversation the names of these loved ones when the veteran makes statements about becoming a better son/father/grandfather or daughter/mother/grandmother. These references make the goal concrete . A loved one can even be a pet; and for some, the desire to be a more competent and reliable pet owner can be a strong motivation. Give the veteran an opportunity to describe his or her strengths and bask in a self-description. In recovery, it is critical to identify strengths that can be built on to sustain recovery.
Veterans who are confronting a substance use disorder may approach the practitioner with a “fix it for me” attitude as a mental inventory is being taken of all of the negative consequences (eg, homelessness, legal issues, or unemployment). Getting the veteran to take ownership of the problem and the solution is key.
I don’t promise to fix things, instead I engage the veteran in problem-solving. I offer to team up with the veteran in this process, and I promise my best efforts but not outcomes. I avoid giving advice and work to empower the veteran to make sound decisions. Veterans who make their own decisions feel as though they have more control over their lives.
Working with the substance use disorder population is challenging but rewarding when a practitioner can embrace some of the paradigms described in this article. Practitioners may need to do some cognitive reframing within their own thinking, as I described in this article, to become more effective in the field and to help avoid burnout.
I am a behavioral health-licensed clinical social worker, an approved motivational enhancement therapy provider, and the point of contact for substance use disorders at the Berks CBOC of the Lebanon VAMC in Wyomissing, Pennsylvania. Recently, an exasperated primary care provider at the Berks CBOC approached me about working with patients with substance use disorders and asked “How do you work with this challenging population?”
The question was cause for some introspection. When I started working in addictions, I had to modify my approach to work effectively with this population. I experienced a paradigm shift in which I no longer assigned myself credit or blame for a veteran’s continued sobriety or relapse: Each patient is responsible for his or her progress in the recovery journey. When a patient has a relapse, I remind myself that statistically relapse is a probability for the majority of those in recovery; even multiple relapses are common. Therefore, another way of viewing relapse is that the relapse itself may bring the veteran a step closer to permanent abstinence.
Cognitive Reframing
Consider a toddler learning to walk. Parents and caregivers of the child expect the child to fall quite a few times before he or she has mastered walking. The parents and caregivers don’t get angry, take the situation personally, or feel manipulated by the child’s “failure.” Instead, the parents offer the child emotional support and encouragement.
Arguably, the practitioner should take a similar stance—emotionally supportive and encouraging—in combination with dialogue guided by motivational interviewing to support change and help the veteran get back on track. Although not inevitable, relapse is a normal part of recovery.
Practitioners should avoid either scolding or praising patients. Scolding doesn’t help in the recovery journey, and if the veteran learns that the practitioner reacts by scolding after learning of the relapse, then it is less likely that he or she will be open about future missteps. In fact, the veteran may not return at all. The goal is to have the patient come back when relapse occurs to help him or her progress to recovery.
Too much praise can cause similar problems. A veteran accustomed to praise when he or she is doing well, may be too embarrassed or ashamed to return to ask for help after a relapse. Instead the practitioner should use affirmations, which are widely discussed in motivational interviewing (MI) literature.
“I want to be clean and sober” or “I want to stop drinking” are vague statements that should raise a red flag for experienced practitioners. Is this patient just telling me what he or she thinks I want to hear to respond? That may be the case but also may be an assumption. Instead these assertions could be regarded as global treatment goals, and the task of the practitioner is to help the veteran develop objectives and interventions in relation to this goal. These broad statements can be a starting point in MI. These words can sound just as foreign to a patient who isn’t sure whether becoming clean and sober is a possibility. The veteran may not have the confidence to reach the goal of being clean and sober, and these statements may seem awkward and out of sync with his or her facial expression and body language.
Showing disbelief in a veteran also can have negative consequences. The veteran might feel that “even my therapist/health care provider doesn’t believe I can become clean and sober.” Instead, I remind myself that we all must manipulate our environment for survival. I find it more valuable to think of the veteran as being resourceful rather than manipulative.
This point may seem self-evident, but it took me a while to catch on: Most of the change in the recovery journey transpires outside the practitioner’s office. I had to embrace this truism and be prepared when the veteran returned for the next session. The task, then, is to determine at this moment where the veteran is in his or her recovery journey instead of continuing the conversation from the previous session. The previous session may be irrelevant. Thinking this way was an adjustment for me. One of my favorite therapy approaches was to consciously continue a conversation from the previous session to demonstrate that I remembered what the patient had said, thereby showing that I care.
In addition, get to know the patient underneath, behind, and before the substance use disorder. Knowing and liking the veteran helps me avoid burnout, bringing me back to my values and the reason I became a therapist. I make efforts in my thinking process to convert “alcoholic” or “drug abuser” into a more helpful “client addicted to alcohol” or “client with a substance use disorder.” The veteran should not be labeled.
Motivational Interviewing
Oftentimes what propels veterans forward in their recovery is the cognitive dissonance created between who they were and how they acted before substance addiction and how they act now. Conversation steered in this direction, fueled by MI, can enhance a veteran’s motivation to change and ready the veteran to change behaviors. Listen to the veteran’s account of loved ones and remember the names of these important family members and friends. Weave into the conversation the names of these loved ones when the veteran makes statements about becoming a better son/father/grandfather or daughter/mother/grandmother. These references make the goal concrete . A loved one can even be a pet; and for some, the desire to be a more competent and reliable pet owner can be a strong motivation. Give the veteran an opportunity to describe his or her strengths and bask in a self-description. In recovery, it is critical to identify strengths that can be built on to sustain recovery.
Veterans who are confronting a substance use disorder may approach the practitioner with a “fix it for me” attitude as a mental inventory is being taken of all of the negative consequences (eg, homelessness, legal issues, or unemployment). Getting the veteran to take ownership of the problem and the solution is key.
I don’t promise to fix things, instead I engage the veteran in problem-solving. I offer to team up with the veteran in this process, and I promise my best efforts but not outcomes. I avoid giving advice and work to empower the veteran to make sound decisions. Veterans who make their own decisions feel as though they have more control over their lives.
Working with the substance use disorder population is challenging but rewarding when a practitioner can embrace some of the paradigms described in this article. Practitioners may need to do some cognitive reframing within their own thinking, as I described in this article, to become more effective in the field and to help avoid burnout.
I am a behavioral health-licensed clinical social worker, an approved motivational enhancement therapy provider, and the point of contact for substance use disorders at the Berks CBOC of the Lebanon VAMC in Wyomissing, Pennsylvania. Recently, an exasperated primary care provider at the Berks CBOC approached me about working with patients with substance use disorders and asked “How do you work with this challenging population?”
The question was cause for some introspection. When I started working in addictions, I had to modify my approach to work effectively with this population. I experienced a paradigm shift in which I no longer assigned myself credit or blame for a veteran’s continued sobriety or relapse: Each patient is responsible for his or her progress in the recovery journey. When a patient has a relapse, I remind myself that statistically relapse is a probability for the majority of those in recovery; even multiple relapses are common. Therefore, another way of viewing relapse is that the relapse itself may bring the veteran a step closer to permanent abstinence.
Cognitive Reframing
Consider a toddler learning to walk. Parents and caregivers of the child expect the child to fall quite a few times before he or she has mastered walking. The parents and caregivers don’t get angry, take the situation personally, or feel manipulated by the child’s “failure.” Instead, the parents offer the child emotional support and encouragement.
Arguably, the practitioner should take a similar stance—emotionally supportive and encouraging—in combination with dialogue guided by motivational interviewing to support change and help the veteran get back on track. Although not inevitable, relapse is a normal part of recovery.
Practitioners should avoid either scolding or praising patients. Scolding doesn’t help in the recovery journey, and if the veteran learns that the practitioner reacts by scolding after learning of the relapse, then it is less likely that he or she will be open about future missteps. In fact, the veteran may not return at all. The goal is to have the patient come back when relapse occurs to help him or her progress to recovery.
Too much praise can cause similar problems. A veteran accustomed to praise when he or she is doing well, may be too embarrassed or ashamed to return to ask for help after a relapse. Instead the practitioner should use affirmations, which are widely discussed in motivational interviewing (MI) literature.
“I want to be clean and sober” or “I want to stop drinking” are vague statements that should raise a red flag for experienced practitioners. Is this patient just telling me what he or she thinks I want to hear to respond? That may be the case but also may be an assumption. Instead these assertions could be regarded as global treatment goals, and the task of the practitioner is to help the veteran develop objectives and interventions in relation to this goal. These broad statements can be a starting point in MI. These words can sound just as foreign to a patient who isn’t sure whether becoming clean and sober is a possibility. The veteran may not have the confidence to reach the goal of being clean and sober, and these statements may seem awkward and out of sync with his or her facial expression and body language.
Showing disbelief in a veteran also can have negative consequences. The veteran might feel that “even my therapist/health care provider doesn’t believe I can become clean and sober.” Instead, I remind myself that we all must manipulate our environment for survival. I find it more valuable to think of the veteran as being resourceful rather than manipulative.
This point may seem self-evident, but it took me a while to catch on: Most of the change in the recovery journey transpires outside the practitioner’s office. I had to embrace this truism and be prepared when the veteran returned for the next session. The task, then, is to determine at this moment where the veteran is in his or her recovery journey instead of continuing the conversation from the previous session. The previous session may be irrelevant. Thinking this way was an adjustment for me. One of my favorite therapy approaches was to consciously continue a conversation from the previous session to demonstrate that I remembered what the patient had said, thereby showing that I care.
In addition, get to know the patient underneath, behind, and before the substance use disorder. Knowing and liking the veteran helps me avoid burnout, bringing me back to my values and the reason I became a therapist. I make efforts in my thinking process to convert “alcoholic” or “drug abuser” into a more helpful “client addicted to alcohol” or “client with a substance use disorder.” The veteran should not be labeled.
Motivational Interviewing
Oftentimes what propels veterans forward in their recovery is the cognitive dissonance created between who they were and how they acted before substance addiction and how they act now. Conversation steered in this direction, fueled by MI, can enhance a veteran’s motivation to change and ready the veteran to change behaviors. Listen to the veteran’s account of loved ones and remember the names of these important family members and friends. Weave into the conversation the names of these loved ones when the veteran makes statements about becoming a better son/father/grandfather or daughter/mother/grandmother. These references make the goal concrete . A loved one can even be a pet; and for some, the desire to be a more competent and reliable pet owner can be a strong motivation. Give the veteran an opportunity to describe his or her strengths and bask in a self-description. In recovery, it is critical to identify strengths that can be built on to sustain recovery.
Veterans who are confronting a substance use disorder may approach the practitioner with a “fix it for me” attitude as a mental inventory is being taken of all of the negative consequences (eg, homelessness, legal issues, or unemployment). Getting the veteran to take ownership of the problem and the solution is key.
I don’t promise to fix things, instead I engage the veteran in problem-solving. I offer to team up with the veteran in this process, and I promise my best efforts but not outcomes. I avoid giving advice and work to empower the veteran to make sound decisions. Veterans who make their own decisions feel as though they have more control over their lives.
Working with the substance use disorder population is challenging but rewarding when a practitioner can embrace some of the paradigms described in this article. Practitioners may need to do some cognitive reframing within their own thinking, as I described in this article, to become more effective in the field and to help avoid burnout.
Preop atrial fib in CABG patients spells trouble
COLORADO SPRINGS – Preoperative atrial fibrillation is present in more than 10% of patients undergoing isolated coronary artery bypass graft (CABG) surgery, and if not subjected to concomitant surgical ablation it’s associated with increased perioperative and long-term major morbidity and mortality, S. Chris Malaisrie, MD, reported at the annual meeting of the Western Thoracic Surgical Association.
The increased early and late risks posed by preoperative atrial fibrillation (AF) that go unaddressed remain significant even after adjusting for the numerous comorbid conditions more prevalent in CABG patients with preoperative AF than in those without the arrhythmia, added Dr. Malaisrie, a cardiac surgeon at Northwestern University in Chicago.
The unadjusted operative mortality rate was 1.8% in the no-AF group and 4.0% in patients with preoperative AF. Unadjusted in-hospital rates of permanent stroke, prolonged ventilation, reoperation, and new renal failure were also significantly higher in the preoperative AF group.
Not surprisingly, the preoperative AF group was older. They also had significantly higher baseline rates of numerous comorbid conditions, including diabetes, peripheral vascular disease, renal failure, and prior stroke, as well as a lower mean left ventricular ejection fraction. However, after adjustment for the many comorbidities in multivariate regression analysis, the risks of all in-hospital adverse outcomes remained significantly higher in the preoperative AF group. For example, their adjusted risk of operative mortality was 1.5-fold greater than in the no-AF patients.
In the long-term follow-up analysis, the unadjusted risk of mortality in the first 5 years after CABG was 2.5-fold greater in the preoperative AF group. Their 5-year risk of stroke or systemic embolization was 1.5-fold greater, too. Upon adjustment for potentially confounding comorbid conditions, preoperative AF was associated with a 1.5-fold increased 5-year risk of mortality and a 1.2-fold increase in stroke or systemic embolism.
In an effort to identify a particularly high-risk group of CABG patients with preoperative AF, Dr. Malaisrie and his coinvestigators stratified the group’s long-term stroke and mortality risks by their CHA2DS2-VASc score at the time of surgery. The results were revealing: the unadjusted 5-year risk of stroke or systemic embolization was 7.9% in those with a CHA2DS2-VASc score of 1-3, 12.2% with a score of 4-6, and 15.4% with a score of 7-9. The 5-year survival rate was 74.8% with a score of 1-3, 56.5% with a score of 4-6, and 41.2% with a score of 7-9.
“That’s really a striking finding,” Dr. Malaisrie observed. “When you consider a patient who’s, say, 72-75 years old, who is undergoing isolated CABG with preoperative atrial fibrillation and who has a high CHA2DS2-VASc score of 7-9, 5-year survival is only 41%, with a 15% risk of stroke or systemic embolization.”
Discussant William T. Caine, MD, found the study results unsettling.
“I was surprised to see that in this day and age, fully two-thirds of the patients who had preoperative atrial fibrillation had no attempt at any ablation procedure to treat their atrial fibrillation,” declared Dr. Caine of Intermountain Medical Center in Salt Lake City.
In reply, Dr. Malaisrie noted that other, smaller studies have also found that only about 30% of CABG patients with preoperative AF undergo surgical AF ablation through a maze procedure or some other method.
“Probably most of us in this room would go ahead and perform surgical ablation, but the STS database represents all isolated CABG procedures done throughout the United States,” Dr. Malaisrie said. “I think this dataset should help convince the other 70% of surgeons out there that there is a high cost for preoperative AF – in particular, in patients with very high CHA2DS2-VASc scores. If you can identify a group of patients at increased risk for stroke and mortality, you’d certainly want to bend their survival curve.”
The maze procedure has been convincingly shown to be very safe, with no associated increased risk of perioperative morbidity and mortality. The downside is cost. But while it’s true that adding surgical ablation to an isolated CABG procedure boosts OR time and procedural costs, a successful ablation is likely to pay dividends through reduced downstream rates of major morbidity and mortality.
“I look forward to the second part of our analysis, where we’ll look at the comparative data for the patients who did in fact have surgical ablation. That dataset is pending from the Duke Clinical Research Institute,” according to Dr. Malaisrie.
He cited as study limitations the inability to complete linkage to the Medicare database in about 37% of CABG patients in the STS database, or more than 200,000 people. Also, the Medicare database is an administrative dataset reliant upon medical record coding. The mortality data are probably quite accurate, but the stroke and systemic embolization rates cited in this analysis likely underestimate the true rates.
He reported serving as a consultant to Edwards Lifesciences, Abbott Vascular, and Baxter, and serving on speakers’ bureaus for Bolton and Abiomed. However, the STS analysis was funded exclusively by philanthropy.
COLORADO SPRINGS – Preoperative atrial fibrillation is present in more than 10% of patients undergoing isolated coronary artery bypass graft (CABG) surgery, and if not subjected to concomitant surgical ablation it’s associated with increased perioperative and long-term major morbidity and mortality, S. Chris Malaisrie, MD, reported at the annual meeting of the Western Thoracic Surgical Association.
The increased early and late risks posed by preoperative atrial fibrillation (AF) that go unaddressed remain significant even after adjusting for the numerous comorbid conditions more prevalent in CABG patients with preoperative AF than in those without the arrhythmia, added Dr. Malaisrie, a cardiac surgeon at Northwestern University in Chicago.
The unadjusted operative mortality rate was 1.8% in the no-AF group and 4.0% in patients with preoperative AF. Unadjusted in-hospital rates of permanent stroke, prolonged ventilation, reoperation, and new renal failure were also significantly higher in the preoperative AF group.
Not surprisingly, the preoperative AF group was older. They also had significantly higher baseline rates of numerous comorbid conditions, including diabetes, peripheral vascular disease, renal failure, and prior stroke, as well as a lower mean left ventricular ejection fraction. However, after adjustment for the many comorbidities in multivariate regression analysis, the risks of all in-hospital adverse outcomes remained significantly higher in the preoperative AF group. For example, their adjusted risk of operative mortality was 1.5-fold greater than in the no-AF patients.
In the long-term follow-up analysis, the unadjusted risk of mortality in the first 5 years after CABG was 2.5-fold greater in the preoperative AF group. Their 5-year risk of stroke or systemic embolization was 1.5-fold greater, too. Upon adjustment for potentially confounding comorbid conditions, preoperative AF was associated with a 1.5-fold increased 5-year risk of mortality and a 1.2-fold increase in stroke or systemic embolism.
In an effort to identify a particularly high-risk group of CABG patients with preoperative AF, Dr. Malaisrie and his coinvestigators stratified the group’s long-term stroke and mortality risks by their CHA2DS2-VASc score at the time of surgery. The results were revealing: the unadjusted 5-year risk of stroke or systemic embolization was 7.9% in those with a CHA2DS2-VASc score of 1-3, 12.2% with a score of 4-6, and 15.4% with a score of 7-9. The 5-year survival rate was 74.8% with a score of 1-3, 56.5% with a score of 4-6, and 41.2% with a score of 7-9.
“That’s really a striking finding,” Dr. Malaisrie observed. “When you consider a patient who’s, say, 72-75 years old, who is undergoing isolated CABG with preoperative atrial fibrillation and who has a high CHA2DS2-VASc score of 7-9, 5-year survival is only 41%, with a 15% risk of stroke or systemic embolization.”
Discussant William T. Caine, MD, found the study results unsettling.
“I was surprised to see that in this day and age, fully two-thirds of the patients who had preoperative atrial fibrillation had no attempt at any ablation procedure to treat their atrial fibrillation,” declared Dr. Caine of Intermountain Medical Center in Salt Lake City.
In reply, Dr. Malaisrie noted that other, smaller studies have also found that only about 30% of CABG patients with preoperative AF undergo surgical AF ablation through a maze procedure or some other method.
“Probably most of us in this room would go ahead and perform surgical ablation, but the STS database represents all isolated CABG procedures done throughout the United States,” Dr. Malaisrie said. “I think this dataset should help convince the other 70% of surgeons out there that there is a high cost for preoperative AF – in particular, in patients with very high CHA2DS2-VASc scores. If you can identify a group of patients at increased risk for stroke and mortality, you’d certainly want to bend their survival curve.”
The maze procedure has been convincingly shown to be very safe, with no associated increased risk of perioperative morbidity and mortality. The downside is cost. But while it’s true that adding surgical ablation to an isolated CABG procedure boosts OR time and procedural costs, a successful ablation is likely to pay dividends through reduced downstream rates of major morbidity and mortality.
“I look forward to the second part of our analysis, where we’ll look at the comparative data for the patients who did in fact have surgical ablation. That dataset is pending from the Duke Clinical Research Institute,” according to Dr. Malaisrie.
He cited as study limitations the inability to complete linkage to the Medicare database in about 37% of CABG patients in the STS database, or more than 200,000 people. Also, the Medicare database is an administrative dataset reliant upon medical record coding. The mortality data are probably quite accurate, but the stroke and systemic embolization rates cited in this analysis likely underestimate the true rates.
He reported serving as a consultant to Edwards Lifesciences, Abbott Vascular, and Baxter, and serving on speakers’ bureaus for Bolton and Abiomed. However, the STS analysis was funded exclusively by philanthropy.
COLORADO SPRINGS – Preoperative atrial fibrillation is present in more than 10% of patients undergoing isolated coronary artery bypass graft (CABG) surgery, and if not subjected to concomitant surgical ablation it’s associated with increased perioperative and long-term major morbidity and mortality, S. Chris Malaisrie, MD, reported at the annual meeting of the Western Thoracic Surgical Association.
The increased early and late risks posed by preoperative atrial fibrillation (AF) that go unaddressed remain significant even after adjusting for the numerous comorbid conditions more prevalent in CABG patients with preoperative AF than in those without the arrhythmia, added Dr. Malaisrie, a cardiac surgeon at Northwestern University in Chicago.
The unadjusted operative mortality rate was 1.8% in the no-AF group and 4.0% in patients with preoperative AF. Unadjusted in-hospital rates of permanent stroke, prolonged ventilation, reoperation, and new renal failure were also significantly higher in the preoperative AF group.
Not surprisingly, the preoperative AF group was older. They also had significantly higher baseline rates of numerous comorbid conditions, including diabetes, peripheral vascular disease, renal failure, and prior stroke, as well as a lower mean left ventricular ejection fraction. However, after adjustment for the many comorbidities in multivariate regression analysis, the risks of all in-hospital adverse outcomes remained significantly higher in the preoperative AF group. For example, their adjusted risk of operative mortality was 1.5-fold greater than in the no-AF patients.
In the long-term follow-up analysis, the unadjusted risk of mortality in the first 5 years after CABG was 2.5-fold greater in the preoperative AF group. Their 5-year risk of stroke or systemic embolization was 1.5-fold greater, too. Upon adjustment for potentially confounding comorbid conditions, preoperative AF was associated with a 1.5-fold increased 5-year risk of mortality and a 1.2-fold increase in stroke or systemic embolism.
In an effort to identify a particularly high-risk group of CABG patients with preoperative AF, Dr. Malaisrie and his coinvestigators stratified the group’s long-term stroke and mortality risks by their CHA2DS2-VASc score at the time of surgery. The results were revealing: the unadjusted 5-year risk of stroke or systemic embolization was 7.9% in those with a CHA2DS2-VASc score of 1-3, 12.2% with a score of 4-6, and 15.4% with a score of 7-9. The 5-year survival rate was 74.8% with a score of 1-3, 56.5% with a score of 4-6, and 41.2% with a score of 7-9.
“That’s really a striking finding,” Dr. Malaisrie observed. “When you consider a patient who’s, say, 72-75 years old, who is undergoing isolated CABG with preoperative atrial fibrillation and who has a high CHA2DS2-VASc score of 7-9, 5-year survival is only 41%, with a 15% risk of stroke or systemic embolization.”
Discussant William T. Caine, MD, found the study results unsettling.
“I was surprised to see that in this day and age, fully two-thirds of the patients who had preoperative atrial fibrillation had no attempt at any ablation procedure to treat their atrial fibrillation,” declared Dr. Caine of Intermountain Medical Center in Salt Lake City.
In reply, Dr. Malaisrie noted that other, smaller studies have also found that only about 30% of CABG patients with preoperative AF undergo surgical AF ablation through a maze procedure or some other method.
“Probably most of us in this room would go ahead and perform surgical ablation, but the STS database represents all isolated CABG procedures done throughout the United States,” Dr. Malaisrie said. “I think this dataset should help convince the other 70% of surgeons out there that there is a high cost for preoperative AF – in particular, in patients with very high CHA2DS2-VASc scores. If you can identify a group of patients at increased risk for stroke and mortality, you’d certainly want to bend their survival curve.”
The maze procedure has been convincingly shown to be very safe, with no associated increased risk of perioperative morbidity and mortality. The downside is cost. But while it’s true that adding surgical ablation to an isolated CABG procedure boosts OR time and procedural costs, a successful ablation is likely to pay dividends through reduced downstream rates of major morbidity and mortality.
“I look forward to the second part of our analysis, where we’ll look at the comparative data for the patients who did in fact have surgical ablation. That dataset is pending from the Duke Clinical Research Institute,” according to Dr. Malaisrie.
He cited as study limitations the inability to complete linkage to the Medicare database in about 37% of CABG patients in the STS database, or more than 200,000 people. Also, the Medicare database is an administrative dataset reliant upon medical record coding. The mortality data are probably quite accurate, but the stroke and systemic embolization rates cited in this analysis likely underestimate the true rates.
He reported serving as a consultant to Edwards Lifesciences, Abbott Vascular, and Baxter, and serving on speakers’ bureaus for Bolton and Abiomed. However, the STS analysis was funded exclusively by philanthropy.
AT THE WTSA ANNUAL MEETING
Key clinical point:
Major finding: Preoperative AF in patients undergoing isolated CABG was tied to an adjusted 45% greater 5-year mortality and 25% increase in stroke and systemic embolization risk, compared with CABG patients without the preoperative arrhythmia.
Data source: This retrospective study compared perioperative and long-term morbidity and mortality in nearly 350,000 patients in the Society of Thoracic Surgeons database who underwent isolated CABG, including more than 24,000 who had preoperative atrial fibrillation that wasn’t addressed surgically.
Disclosures: The study presenter reported serving as a consultant to Edwards Lifesciences, Abbott Vascular, and Baxter, and serving on speakers’ bureaus for Bolton and Abiomed. However, the STS analysis was funded exclusively by philanthropy.
VSTs can treat 5 different viral infections after HSCT
New research suggests virus-specific T cells (VSTs) can protect patients from severe viral infections that sometimes occur after hematopoietic stem cell transplant (HSCT).
The VSTs proved effective against 5 different viruses—Epstein-Barr virus (EBV), adenovirus (AdV), cytomegalovirus (CMV), BK virus (BKV), and human herpesvirus 6 (HHV-6).
Ifigeneia Tzannou, MD, of Baylor College of Medicine in Houston, Texas, and her colleagues reported these findings in the Journal of Clinical Oncology.
“In this study, we continued our previous work . . . in which we showed that patients who had developed an Epstein-Barr virus infection after a transplant . . . could be helped by receiving immune cells specialized in eliminating that particular virus,” Dr Tzannou said. “Then, we and others successfully targeted other viruses—namely, adenoviruses and cytomegalovirus.”
“The novel contribution of this study is that we have targeted additional viruses, the BK virus and the HHV-6 virus, which had not been targeted this way before,” added study author Bilal Omer, MD, of Baylor College of Medicine.
“This is important because the BK virus does not have an effective treatment, and the complications are significant, including severe pain and bleeding. These patients are in the hospital for weeks, months sometimes, and, now, we have a treatment option.”
The researchers tested their VSTs in a phase 2 trial of 38 HSCT recipients with at least 1 of the aforementioned viruses.
“[To prepare the VSTs,] we take blood from healthy donors who have already been exposed to these viruses and who we have confirmed have immune cells that can fight the infections,” Dr Tzannou said.
“We isolate the cells and let them multiply in culture. The final product is a mixture of cells that, together, can target all 5 viruses. We prepared 59 sets of virus-specific cells from different donors following this procedure.”
“Our strategy is to prepare a number of sets of virus-specific cells ahead of time and store them in a freezer, ready to use when a patient needs them,” Dr Omer noted. “To match patient and donor, we use elaborate matching algorithms.”
Patients
The trial included 38 patients who had undergone HSCT to treat acute myeloid leukemia/myelodysplastic syndromes (n=20), acute lymphoblastic leukemia (n=9), lymphoma/myeloma (n=3), or nonmalignant disorders (n=6).
These 38 patients had a total of 45 infections—CMV (n=17), EBV (n=2), AdV (n=7), BKV (n=16), and HHV-6 (n=3).
Response
The researchers monitored virus levels and other clinical responses in the 37 evaluable patients.
Six weeks after the first VST infusion, the overall response rate was 91.9%.
Seventeen patients received VSTs for persistent CMV. Sixteen of these patients (94.1%) responded, 6 with complete responses (CRs) and 10 with partial responses (PRs).
Two patients received VSTs for EBV, and both achieved a virologic CR.
Seven patients received VSTs for persistent AdV. The response rate was 71.4%. Four patients achieved a CR, 1 had a PR, and 2 patients did not respond.
Three patients received VSTs to treat HHV-6 reactivations. The response rate was 67%. Two patients had a PR, and 1 was not evaluable.
Sixteen patients received VSTs for BKV-associated hemorrhagic cystitis (n= 14) or BKV-associated nephritis (n=2).
All 16 patients responded. One had a clinical and virologic CR. Six had a clinical CR but a virologic PR. Seven had a virologic and clinical PR. And 2 patients had only a virologic PR.
A total of 15 patients received a second VST infusion—1 due to lack of response, 7 who had a PR, and 7 due to recurrence. Ten of these patients responded to the second infusion—1 with a CR and 9 with a PR.
Four patients received a third infusion of VSTs. Two achieved a CR, 1 had a PR, and 1 did not respond.
Toxicity
One patient developed an isolated fever within 24 hours of VST infusion, but the researchers did not observe any other immediate toxicities.
One of the patients with BKV-associated hemorrhagic cystitis experienced transient hydronephrosis and a decrease in renal function associated with a concomitant bacterial urinary tract infection.
Nineteen patients had prior grade 2 to 4 graft-versus-host disease (GVHD)—15 with grade 2 and 4 with grade 3. All GVHD was quiescent at the time of VST infusion.
One patient developed recurrent grade 3 gastrointestinal GVHD after VST infusion and rapid corticosteroid taper. Five patients developed recurrent (n=3) or de novo (n=2) grade 1 to 2 skin GVHD, which resolved with topical treatment (n=4) and reinitiation of corticosteroid treatment (n=1).
Two patients had a flare of upper-gastrointestinal GVHD, which resolved after a brief corticosteroid course.
“We didn’t have any significant toxicities,” Dr Tzannou said. “Taken together, the results of this trial suggest that it is reasonable to consider this treatment as an early option for these patients. We hope that the results of a future multicenter, phase 3 clinical trial will help raise awareness in both physicians and patients that this treatment, which is safe and effective, is available.” ![]()
New research suggests virus-specific T cells (VSTs) can protect patients from severe viral infections that sometimes occur after hematopoietic stem cell transplant (HSCT).
The VSTs proved effective against 5 different viruses—Epstein-Barr virus (EBV), adenovirus (AdV), cytomegalovirus (CMV), BK virus (BKV), and human herpesvirus 6 (HHV-6).
Ifigeneia Tzannou, MD, of Baylor College of Medicine in Houston, Texas, and her colleagues reported these findings in the Journal of Clinical Oncology.
“In this study, we continued our previous work . . . in which we showed that patients who had developed an Epstein-Barr virus infection after a transplant . . . could be helped by receiving immune cells specialized in eliminating that particular virus,” Dr Tzannou said. “Then, we and others successfully targeted other viruses—namely, adenoviruses and cytomegalovirus.”
“The novel contribution of this study is that we have targeted additional viruses, the BK virus and the HHV-6 virus, which had not been targeted this way before,” added study author Bilal Omer, MD, of Baylor College of Medicine.
“This is important because the BK virus does not have an effective treatment, and the complications are significant, including severe pain and bleeding. These patients are in the hospital for weeks, months sometimes, and, now, we have a treatment option.”
The researchers tested their VSTs in a phase 2 trial of 38 HSCT recipients with at least 1 of the aforementioned viruses.
“[To prepare the VSTs,] we take blood from healthy donors who have already been exposed to these viruses and who we have confirmed have immune cells that can fight the infections,” Dr Tzannou said.
“We isolate the cells and let them multiply in culture. The final product is a mixture of cells that, together, can target all 5 viruses. We prepared 59 sets of virus-specific cells from different donors following this procedure.”
“Our strategy is to prepare a number of sets of virus-specific cells ahead of time and store them in a freezer, ready to use when a patient needs them,” Dr Omer noted. “To match patient and donor, we use elaborate matching algorithms.”
Patients
The trial included 38 patients who had undergone HSCT to treat acute myeloid leukemia/myelodysplastic syndromes (n=20), acute lymphoblastic leukemia (n=9), lymphoma/myeloma (n=3), or nonmalignant disorders (n=6).
These 38 patients had a total of 45 infections—CMV (n=17), EBV (n=2), AdV (n=7), BKV (n=16), and HHV-6 (n=3).
Response
The researchers monitored virus levels and other clinical responses in the 37 evaluable patients.
Six weeks after the first VST infusion, the overall response rate was 91.9%.
Seventeen patients received VSTs for persistent CMV. Sixteen of these patients (94.1%) responded, 6 with complete responses (CRs) and 10 with partial responses (PRs).
Two patients received VSTs for EBV, and both achieved a virologic CR.
Seven patients received VSTs for persistent AdV. The response rate was 71.4%. Four patients achieved a CR, 1 had a PR, and 2 patients did not respond.
Three patients received VSTs to treat HHV-6 reactivations. The response rate was 67%. Two patients had a PR, and 1 was not evaluable.
Sixteen patients received VSTs for BKV-associated hemorrhagic cystitis (n= 14) or BKV-associated nephritis (n=2).
All 16 patients responded. One had a clinical and virologic CR. Six had a clinical CR but a virologic PR. Seven had a virologic and clinical PR. And 2 patients had only a virologic PR.
A total of 15 patients received a second VST infusion—1 due to lack of response, 7 who had a PR, and 7 due to recurrence. Ten of these patients responded to the second infusion—1 with a CR and 9 with a PR.
Four patients received a third infusion of VSTs. Two achieved a CR, 1 had a PR, and 1 did not respond.
Toxicity
One patient developed an isolated fever within 24 hours of VST infusion, but the researchers did not observe any other immediate toxicities.
One of the patients with BKV-associated hemorrhagic cystitis experienced transient hydronephrosis and a decrease in renal function associated with a concomitant bacterial urinary tract infection.
Nineteen patients had prior grade 2 to 4 graft-versus-host disease (GVHD)—15 with grade 2 and 4 with grade 3. All GVHD was quiescent at the time of VST infusion.
One patient developed recurrent grade 3 gastrointestinal GVHD after VST infusion and rapid corticosteroid taper. Five patients developed recurrent (n=3) or de novo (n=2) grade 1 to 2 skin GVHD, which resolved with topical treatment (n=4) and reinitiation of corticosteroid treatment (n=1).
Two patients had a flare of upper-gastrointestinal GVHD, which resolved after a brief corticosteroid course.
“We didn’t have any significant toxicities,” Dr Tzannou said. “Taken together, the results of this trial suggest that it is reasonable to consider this treatment as an early option for these patients. We hope that the results of a future multicenter, phase 3 clinical trial will help raise awareness in both physicians and patients that this treatment, which is safe and effective, is available.” ![]()
New research suggests virus-specific T cells (VSTs) can protect patients from severe viral infections that sometimes occur after hematopoietic stem cell transplant (HSCT).
The VSTs proved effective against 5 different viruses—Epstein-Barr virus (EBV), adenovirus (AdV), cytomegalovirus (CMV), BK virus (BKV), and human herpesvirus 6 (HHV-6).
Ifigeneia Tzannou, MD, of Baylor College of Medicine in Houston, Texas, and her colleagues reported these findings in the Journal of Clinical Oncology.
“In this study, we continued our previous work . . . in which we showed that patients who had developed an Epstein-Barr virus infection after a transplant . . . could be helped by receiving immune cells specialized in eliminating that particular virus,” Dr Tzannou said. “Then, we and others successfully targeted other viruses—namely, adenoviruses and cytomegalovirus.”
“The novel contribution of this study is that we have targeted additional viruses, the BK virus and the HHV-6 virus, which had not been targeted this way before,” added study author Bilal Omer, MD, of Baylor College of Medicine.
“This is important because the BK virus does not have an effective treatment, and the complications are significant, including severe pain and bleeding. These patients are in the hospital for weeks, months sometimes, and, now, we have a treatment option.”
The researchers tested their VSTs in a phase 2 trial of 38 HSCT recipients with at least 1 of the aforementioned viruses.
“[To prepare the VSTs,] we take blood from healthy donors who have already been exposed to these viruses and who we have confirmed have immune cells that can fight the infections,” Dr Tzannou said.
“We isolate the cells and let them multiply in culture. The final product is a mixture of cells that, together, can target all 5 viruses. We prepared 59 sets of virus-specific cells from different donors following this procedure.”
“Our strategy is to prepare a number of sets of virus-specific cells ahead of time and store them in a freezer, ready to use when a patient needs them,” Dr Omer noted. “To match patient and donor, we use elaborate matching algorithms.”
Patients
The trial included 38 patients who had undergone HSCT to treat acute myeloid leukemia/myelodysplastic syndromes (n=20), acute lymphoblastic leukemia (n=9), lymphoma/myeloma (n=3), or nonmalignant disorders (n=6).
These 38 patients had a total of 45 infections—CMV (n=17), EBV (n=2), AdV (n=7), BKV (n=16), and HHV-6 (n=3).
Response
The researchers monitored virus levels and other clinical responses in the 37 evaluable patients.
Six weeks after the first VST infusion, the overall response rate was 91.9%.
Seventeen patients received VSTs for persistent CMV. Sixteen of these patients (94.1%) responded, 6 with complete responses (CRs) and 10 with partial responses (PRs).
Two patients received VSTs for EBV, and both achieved a virologic CR.
Seven patients received VSTs for persistent AdV. The response rate was 71.4%. Four patients achieved a CR, 1 had a PR, and 2 patients did not respond.
Three patients received VSTs to treat HHV-6 reactivations. The response rate was 67%. Two patients had a PR, and 1 was not evaluable.
Sixteen patients received VSTs for BKV-associated hemorrhagic cystitis (n= 14) or BKV-associated nephritis (n=2).
All 16 patients responded. One had a clinical and virologic CR. Six had a clinical CR but a virologic PR. Seven had a virologic and clinical PR. And 2 patients had only a virologic PR.
A total of 15 patients received a second VST infusion—1 due to lack of response, 7 who had a PR, and 7 due to recurrence. Ten of these patients responded to the second infusion—1 with a CR and 9 with a PR.
Four patients received a third infusion of VSTs. Two achieved a CR, 1 had a PR, and 1 did not respond.
Toxicity
One patient developed an isolated fever within 24 hours of VST infusion, but the researchers did not observe any other immediate toxicities.
One of the patients with BKV-associated hemorrhagic cystitis experienced transient hydronephrosis and a decrease in renal function associated with a concomitant bacterial urinary tract infection.
Nineteen patients had prior grade 2 to 4 graft-versus-host disease (GVHD)—15 with grade 2 and 4 with grade 3. All GVHD was quiescent at the time of VST infusion.
One patient developed recurrent grade 3 gastrointestinal GVHD after VST infusion and rapid corticosteroid taper. Five patients developed recurrent (n=3) or de novo (n=2) grade 1 to 2 skin GVHD, which resolved with topical treatment (n=4) and reinitiation of corticosteroid treatment (n=1).
Two patients had a flare of upper-gastrointestinal GVHD, which resolved after a brief corticosteroid course.
“We didn’t have any significant toxicities,” Dr Tzannou said. “Taken together, the results of this trial suggest that it is reasonable to consider this treatment as an early option for these patients. We hope that the results of a future multicenter, phase 3 clinical trial will help raise awareness in both physicians and patients that this treatment, which is safe and effective, is available.” ![]()
Young & Crusty
1. A 4-year-old boy presents with erythematous, oozing, excoriated plaques on the cheeks and chin (sparing the nose), trunk, and extensor surfaces of the arms and legs. His parents report that the “rash,” which flares on occasion, has been apparent since the boy was 6 months old, and he does anything he can to scratch the itch.
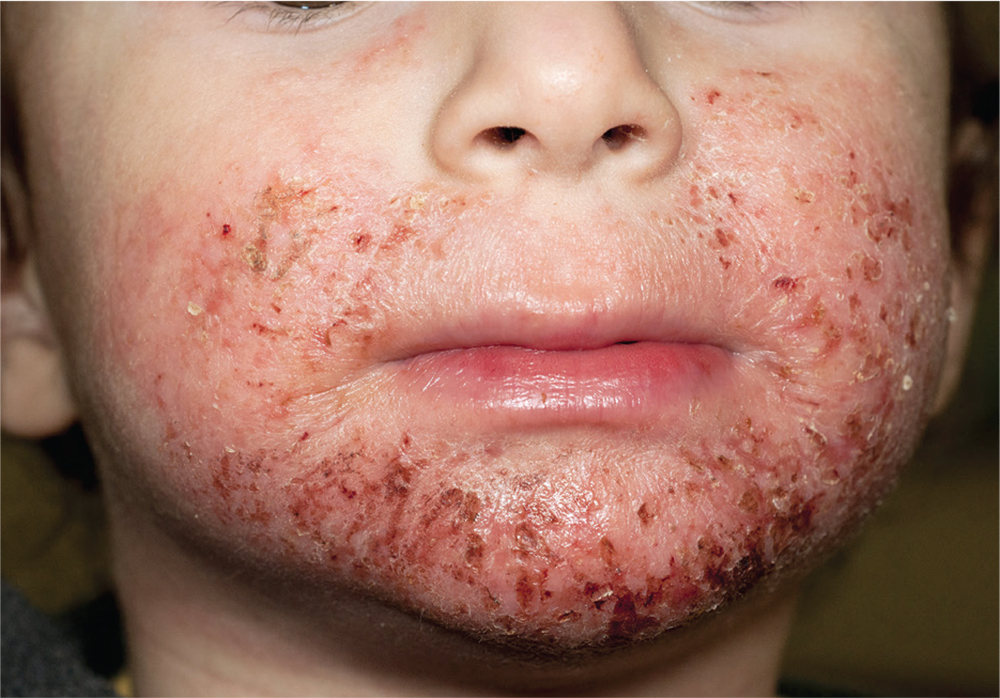
Diagnosis: Diagnosis of atopic dermatitis (AD) is made by age 5 in 85% to 90% of children who develop the disease, and by age 1 in 60% to 65%. AD persists into adulthood in up to one-third of patients. Although the cause is unknown, AD may be triggered by viral infections, food allergens, or weather, and in turn may trigger an inflammatory progression known as atopic march. Other factors affecting AD development include genetics and hygiene.
For more information:
“A Practical Overview of Pediatric Atopic Dermatitis, Part 2: Triggers and Grading.” Cutis. 2016;97(5):326-329.
“A Practical Overview of Pediatric Atopic Dermatitis, Part 3: Differential Diagnosis, Comorbidities, and Measurement of Disease Burden.” Cutis. 2016;97(6):408-412.
2. A child presents with a history of flaccid bullae and encased fluid progressing from clear yellow to turbid and darkish yellow. The pustules have ruptured, resulting in thin, light brown to golden yellow crusts and a collarette of scale at the periphery of the erosion. Itching is mild.
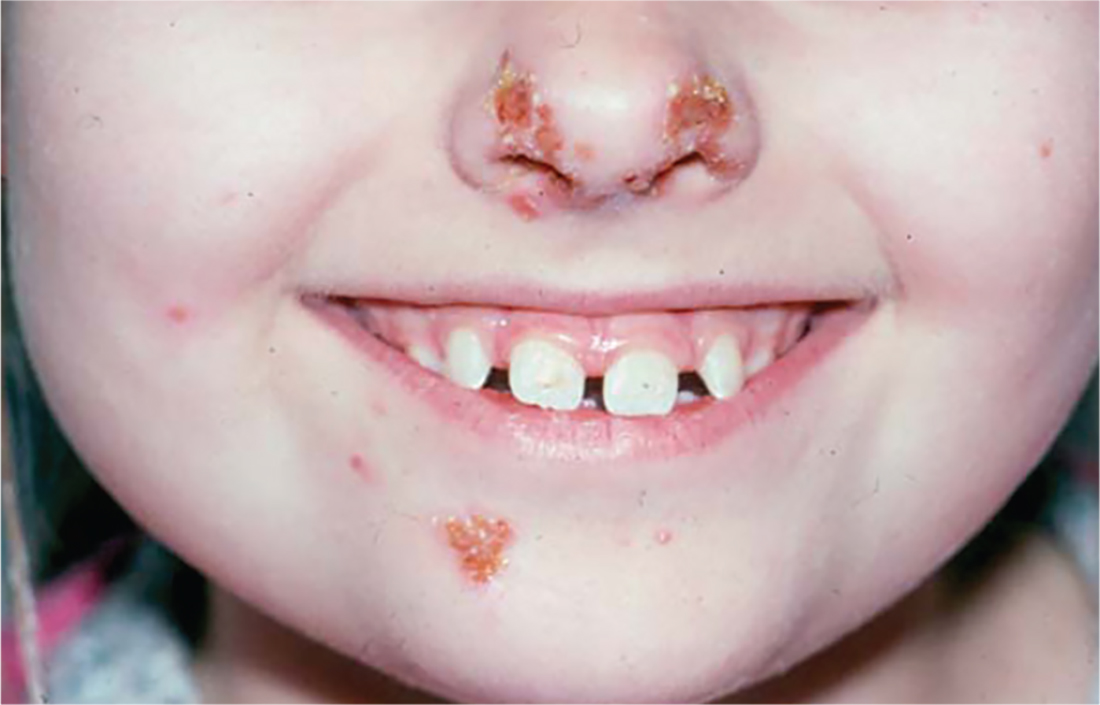
Diagnosis: Bullous impetigo most commonly affects neonates, hence the occasionally used (and inadvisably employed) name pemphigus neonatorum. Bullous impetigo appears to be less contagious than the nonbullous form and is usually sporadic in presentation. Typical areas of occurrence include the trunk and extremities, as well as intertriginous zones (eg, the diaper area, neck folds, and axillae).
For more information, see “Impetigo Update: New Challenges in the Era of Methicillin Resistance.” Cutis. 2010;85(2):65-70.
3. A 16-year-old Hispanic girl seeks consultation for a perioral rash that first appeared two weeks ago, shortly after she used an OTC depilatory agent. The rash has worsened despite application of topical neomycin ointment. The patient reports pruritus and occasional burning. Examination reveals erythema, hyperpigmentation, and excoriation of the skin around the corners of the mouth.
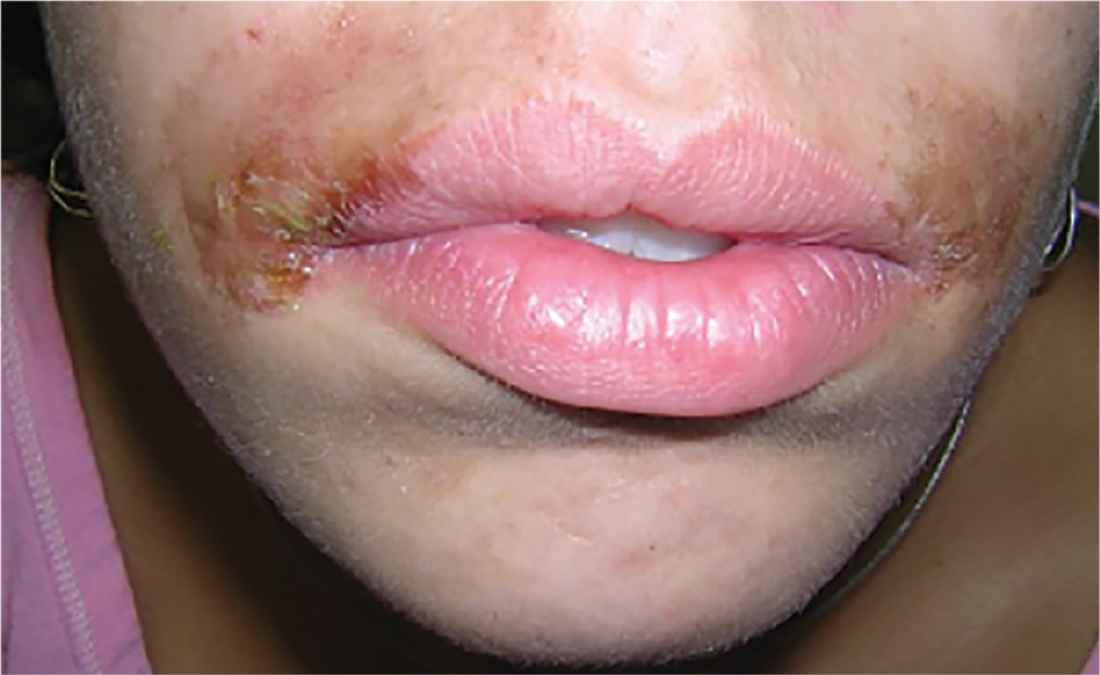
Diagnosis: An allergic contact dermatitis to both the depilatory agent and neomycin was suspected. The patient was advised to discontinue use of both products, and a medium-potency topical steroid was prescribed. She returned for follow-up in 10 days, at which time the condition had improved by about 75%. Therapy was changed to 1% hydrocortisone cream. The patient was instructed to return for patch testing if the dermatitis recurred.
Reprinted with permission from Emergency Medicine. 2010;42(8):19-20. http://www.mdedge.com/emed-journal/article/71585/dermatology/lesion-scrotum
4. The rash on this 5-month-old baby’s hands manifested several weeks ago. It spread to his arms and trunk and is now essentially everywhere except his face. Despite a number of treatment attempts, including oral antibiotics and OTC topical steroid creams, the problem persists.

Diagnosis: The diagnosis of scabies should be confirmed, whenever possible, with microscopic scabetic findings. In addition, treating the whole family and identifying the source of the infestation are crucial. Diagnosis of scabies is difficult in infants, as any part of an infant’s thin, soft, relatively hairless skin is fair game (whereas, in adults, scabies rarely affects skin above the neck). And although infants with scabies undoubtedly itch—probably just as much as adults—they are inept excoriators and even worse historians. In contrast, adults with scabies will scratch and complain continuously while in the exam room.
For more information, see “Baby Has Rash; Parents Feel Itchy.” Clinician Reviews. 2014;24(9):W3.
1. A 4-year-old boy presents with erythematous, oozing, excoriated plaques on the cheeks and chin (sparing the nose), trunk, and extensor surfaces of the arms and legs. His parents report that the “rash,” which flares on occasion, has been apparent since the boy was 6 months old, and he does anything he can to scratch the itch.

Diagnosis: Diagnosis of atopic dermatitis (AD) is made by age 5 in 85% to 90% of children who develop the disease, and by age 1 in 60% to 65%. AD persists into adulthood in up to one-third of patients. Although the cause is unknown, AD may be triggered by viral infections, food allergens, or weather, and in turn may trigger an inflammatory progression known as atopic march. Other factors affecting AD development include genetics and hygiene.
For more information:
“A Practical Overview of Pediatric Atopic Dermatitis, Part 2: Triggers and Grading.” Cutis. 2016;97(5):326-329.
“A Practical Overview of Pediatric Atopic Dermatitis, Part 3: Differential Diagnosis, Comorbidities, and Measurement of Disease Burden.” Cutis. 2016;97(6):408-412.
2. A child presents with a history of flaccid bullae and encased fluid progressing from clear yellow to turbid and darkish yellow. The pustules have ruptured, resulting in thin, light brown to golden yellow crusts and a collarette of scale at the periphery of the erosion. Itching is mild.

Diagnosis: Bullous impetigo most commonly affects neonates, hence the occasionally used (and inadvisably employed) name pemphigus neonatorum. Bullous impetigo appears to be less contagious than the nonbullous form and is usually sporadic in presentation. Typical areas of occurrence include the trunk and extremities, as well as intertriginous zones (eg, the diaper area, neck folds, and axillae).
For more information, see “Impetigo Update: New Challenges in the Era of Methicillin Resistance.” Cutis. 2010;85(2):65-70.
3. A 16-year-old Hispanic girl seeks consultation for a perioral rash that first appeared two weeks ago, shortly after she used an OTC depilatory agent. The rash has worsened despite application of topical neomycin ointment. The patient reports pruritus and occasional burning. Examination reveals erythema, hyperpigmentation, and excoriation of the skin around the corners of the mouth.

Diagnosis: An allergic contact dermatitis to both the depilatory agent and neomycin was suspected. The patient was advised to discontinue use of both products, and a medium-potency topical steroid was prescribed. She returned for follow-up in 10 days, at which time the condition had improved by about 75%. Therapy was changed to 1% hydrocortisone cream. The patient was instructed to return for patch testing if the dermatitis recurred.
Reprinted with permission from Emergency Medicine. 2010;42(8):19-20. http://www.mdedge.com/emed-journal/article/71585/dermatology/lesion-scrotum
4. The rash on this 5-month-old baby’s hands manifested several weeks ago. It spread to his arms and trunk and is now essentially everywhere except his face. Despite a number of treatment attempts, including oral antibiotics and OTC topical steroid creams, the problem persists.

Diagnosis: The diagnosis of scabies should be confirmed, whenever possible, with microscopic scabetic findings. In addition, treating the whole family and identifying the source of the infestation are crucial. Diagnosis of scabies is difficult in infants, as any part of an infant’s thin, soft, relatively hairless skin is fair game (whereas, in adults, scabies rarely affects skin above the neck). And although infants with scabies undoubtedly itch—probably just as much as adults—they are inept excoriators and even worse historians. In contrast, adults with scabies will scratch and complain continuously while in the exam room.
For more information, see “Baby Has Rash; Parents Feel Itchy.” Clinician Reviews. 2014;24(9):W3.
1. A 4-year-old boy presents with erythematous, oozing, excoriated plaques on the cheeks and chin (sparing the nose), trunk, and extensor surfaces of the arms and legs. His parents report that the “rash,” which flares on occasion, has been apparent since the boy was 6 months old, and he does anything he can to scratch the itch.

Diagnosis: Diagnosis of atopic dermatitis (AD) is made by age 5 in 85% to 90% of children who develop the disease, and by age 1 in 60% to 65%. AD persists into adulthood in up to one-third of patients. Although the cause is unknown, AD may be triggered by viral infections, food allergens, or weather, and in turn may trigger an inflammatory progression known as atopic march. Other factors affecting AD development include genetics and hygiene.
For more information:
“A Practical Overview of Pediatric Atopic Dermatitis, Part 2: Triggers and Grading.” Cutis. 2016;97(5):326-329.
“A Practical Overview of Pediatric Atopic Dermatitis, Part 3: Differential Diagnosis, Comorbidities, and Measurement of Disease Burden.” Cutis. 2016;97(6):408-412.
2. A child presents with a history of flaccid bullae and encased fluid progressing from clear yellow to turbid and darkish yellow. The pustules have ruptured, resulting in thin, light brown to golden yellow crusts and a collarette of scale at the periphery of the erosion. Itching is mild.

Diagnosis: Bullous impetigo most commonly affects neonates, hence the occasionally used (and inadvisably employed) name pemphigus neonatorum. Bullous impetigo appears to be less contagious than the nonbullous form and is usually sporadic in presentation. Typical areas of occurrence include the trunk and extremities, as well as intertriginous zones (eg, the diaper area, neck folds, and axillae).
For more information, see “Impetigo Update: New Challenges in the Era of Methicillin Resistance.” Cutis. 2010;85(2):65-70.
3. A 16-year-old Hispanic girl seeks consultation for a perioral rash that first appeared two weeks ago, shortly after she used an OTC depilatory agent. The rash has worsened despite application of topical neomycin ointment. The patient reports pruritus and occasional burning. Examination reveals erythema, hyperpigmentation, and excoriation of the skin around the corners of the mouth.

Diagnosis: An allergic contact dermatitis to both the depilatory agent and neomycin was suspected. The patient was advised to discontinue use of both products, and a medium-potency topical steroid was prescribed. She returned for follow-up in 10 days, at which time the condition had improved by about 75%. Therapy was changed to 1% hydrocortisone cream. The patient was instructed to return for patch testing if the dermatitis recurred.
Reprinted with permission from Emergency Medicine. 2010;42(8):19-20. http://www.mdedge.com/emed-journal/article/71585/dermatology/lesion-scrotum
4. The rash on this 5-month-old baby’s hands manifested several weeks ago. It spread to his arms and trunk and is now essentially everywhere except his face. Despite a number of treatment attempts, including oral antibiotics and OTC topical steroid creams, the problem persists.

Diagnosis: The diagnosis of scabies should be confirmed, whenever possible, with microscopic scabetic findings. In addition, treating the whole family and identifying the source of the infestation are crucial. Diagnosis of scabies is difficult in infants, as any part of an infant’s thin, soft, relatively hairless skin is fair game (whereas, in adults, scabies rarely affects skin above the neck). And although infants with scabies undoubtedly itch—probably just as much as adults—they are inept excoriators and even worse historians. In contrast, adults with scabies will scratch and complain continuously while in the exam room.
For more information, see “Baby Has Rash; Parents Feel Itchy.” Clinician Reviews. 2014;24(9):W3.
From the Editors: Your call is important to us
There they were – dropping like a stone toward the lunar surface some 48 years ago. Buzz Aldrin looked at his onboard computer and it reported Error 1202, and alarms started going off in the Lunar Module. Fortunately for Neil Armstrong and Buzz Aldrin, they had a healthy relationship with their computer systems and IT support. Mission control was only 1.5 seconds away and had a huge team of experts that told them they could ignore the error message. The rest is, of course, history. Human beings, not computer software, landed the Eagle. They used their own judgment and experience and data provided by the computer to make their landing decisions.
At 25,000 feet Mission Control answers. Buzz describes the error code. Mission Control reports that his computer is made by Grumman and that Mission Control uses Lockheed-based software. He is advised to call the Grumman help line. At 15,000 feet Grumman responds. They report that Buzz’s password expired about the time the lunar descent burn occurred. He needs to put in a new password and confirm it. He will receive a confirmation email within the next 24 hours. Neil is getting increasingly restive despite his famously bland emotional responses in crises.
At 10,000 feet Buzz gets a new password confirmation, but Error 1202 remains on the display. The moon is enormous in the windshield. Grumman support responds that the error is likely because Buzz put in the wrong weight for the Lunar Module. Buzz begins again feeding in the data to the onboard computer. Grumman suggests that had Buzz simply created the right template, this problem would not have occurred. Buzz, through gritted teeth, asks how he was supposed to create a template for a problem that no one seemed to expect.
They are down to 150 feet now. Neil tells Buzz what he thinks of the computer systems and is told by Mission Control his microphone is hot and that such comments are not appropriate. At Mission Control, a notation is made on their system that Neil will need to discuss this pilot error with the astronaut office upon his return. Neil simply turns off the onboard computer and lands the Lunar Module with seconds of fuel left as he avoids a large boulder field and finds just the right spot. Tranquility Base reports in to Mission Control. Mankind has landed on the Moon.
The alternative history is what surgeons are experiencing every day because of the unhealthy relationship existing between American health care today and our institutional computer systems. Like Neil and Buzz, these surgeons are heroes who avoid boulder fields despite so many obstacles unrelated to their missions. No wonder burnout (a missile term) is so prevalent. We’ve gone from inconvenient to intolerable. Our health care computer systems must be interoperable to have any meaningful use. Our formats need to be understandable. Surgeons need computers to help them make judgments based on easily accessible data in real time. Surgeons need to “fly” the missions and computer systems need to be our servants, not our masters.
Dr. Hughes is clinical professor in the department of surgery and director of medical education at the Kansas University School of Medicine, Salina Campus, and Co-Editor of ACS Surgery News.
There they were – dropping like a stone toward the lunar surface some 48 years ago. Buzz Aldrin looked at his onboard computer and it reported Error 1202, and alarms started going off in the Lunar Module. Fortunately for Neil Armstrong and Buzz Aldrin, they had a healthy relationship with their computer systems and IT support. Mission control was only 1.5 seconds away and had a huge team of experts that told them they could ignore the error message. The rest is, of course, history. Human beings, not computer software, landed the Eagle. They used their own judgment and experience and data provided by the computer to make their landing decisions.
At 25,000 feet Mission Control answers. Buzz describes the error code. Mission Control reports that his computer is made by Grumman and that Mission Control uses Lockheed-based software. He is advised to call the Grumman help line. At 15,000 feet Grumman responds. They report that Buzz’s password expired about the time the lunar descent burn occurred. He needs to put in a new password and confirm it. He will receive a confirmation email within the next 24 hours. Neil is getting increasingly restive despite his famously bland emotional responses in crises.
At 10,000 feet Buzz gets a new password confirmation, but Error 1202 remains on the display. The moon is enormous in the windshield. Grumman support responds that the error is likely because Buzz put in the wrong weight for the Lunar Module. Buzz begins again feeding in the data to the onboard computer. Grumman suggests that had Buzz simply created the right template, this problem would not have occurred. Buzz, through gritted teeth, asks how he was supposed to create a template for a problem that no one seemed to expect.
They are down to 150 feet now. Neil tells Buzz what he thinks of the computer systems and is told by Mission Control his microphone is hot and that such comments are not appropriate. At Mission Control, a notation is made on their system that Neil will need to discuss this pilot error with the astronaut office upon his return. Neil simply turns off the onboard computer and lands the Lunar Module with seconds of fuel left as he avoids a large boulder field and finds just the right spot. Tranquility Base reports in to Mission Control. Mankind has landed on the Moon.
The alternative history is what surgeons are experiencing every day because of the unhealthy relationship existing between American health care today and our institutional computer systems. Like Neil and Buzz, these surgeons are heroes who avoid boulder fields despite so many obstacles unrelated to their missions. No wonder burnout (a missile term) is so prevalent. We’ve gone from inconvenient to intolerable. Our health care computer systems must be interoperable to have any meaningful use. Our formats need to be understandable. Surgeons need computers to help them make judgments based on easily accessible data in real time. Surgeons need to “fly” the missions and computer systems need to be our servants, not our masters.
Dr. Hughes is clinical professor in the department of surgery and director of medical education at the Kansas University School of Medicine, Salina Campus, and Co-Editor of ACS Surgery News.
There they were – dropping like a stone toward the lunar surface some 48 years ago. Buzz Aldrin looked at his onboard computer and it reported Error 1202, and alarms started going off in the Lunar Module. Fortunately for Neil Armstrong and Buzz Aldrin, they had a healthy relationship with their computer systems and IT support. Mission control was only 1.5 seconds away and had a huge team of experts that told them they could ignore the error message. The rest is, of course, history. Human beings, not computer software, landed the Eagle. They used their own judgment and experience and data provided by the computer to make their landing decisions.
At 25,000 feet Mission Control answers. Buzz describes the error code. Mission Control reports that his computer is made by Grumman and that Mission Control uses Lockheed-based software. He is advised to call the Grumman help line. At 15,000 feet Grumman responds. They report that Buzz’s password expired about the time the lunar descent burn occurred. He needs to put in a new password and confirm it. He will receive a confirmation email within the next 24 hours. Neil is getting increasingly restive despite his famously bland emotional responses in crises.
At 10,000 feet Buzz gets a new password confirmation, but Error 1202 remains on the display. The moon is enormous in the windshield. Grumman support responds that the error is likely because Buzz put in the wrong weight for the Lunar Module. Buzz begins again feeding in the data to the onboard computer. Grumman suggests that had Buzz simply created the right template, this problem would not have occurred. Buzz, through gritted teeth, asks how he was supposed to create a template for a problem that no one seemed to expect.
They are down to 150 feet now. Neil tells Buzz what he thinks of the computer systems and is told by Mission Control his microphone is hot and that such comments are not appropriate. At Mission Control, a notation is made on their system that Neil will need to discuss this pilot error with the astronaut office upon his return. Neil simply turns off the onboard computer and lands the Lunar Module with seconds of fuel left as he avoids a large boulder field and finds just the right spot. Tranquility Base reports in to Mission Control. Mankind has landed on the Moon.
The alternative history is what surgeons are experiencing every day because of the unhealthy relationship existing between American health care today and our institutional computer systems. Like Neil and Buzz, these surgeons are heroes who avoid boulder fields despite so many obstacles unrelated to their missions. No wonder burnout (a missile term) is so prevalent. We’ve gone from inconvenient to intolerable. Our health care computer systems must be interoperable to have any meaningful use. Our formats need to be understandable. Surgeons need computers to help them make judgments based on easily accessible data in real time. Surgeons need to “fly” the missions and computer systems need to be our servants, not our masters.
Dr. Hughes is clinical professor in the department of surgery and director of medical education at the Kansas University School of Medicine, Salina Campus, and Co-Editor of ACS Surgery News.
ACS delegation influences AMA policy at HOD meeting
The American Medical Association (AMA) annual House of Delegates (HOD) meeting took place June 10–14, in Chicago, IL. The agenda included 62 reports from the AMA board and councils and 186 resolutions from state medical associations, specialty societies, and AMA sections.
The uncertain mood of the HOD was shaped by congressional activity on the Affordable Care Act and a competitive election for AMA president-elect. At the start of the meeting’s final day, the HOD paused to remember the lives lost and changed one year earlier at the Pulse nightclub in Orlando, FL. The HOD also noted the value of American College of Surgeons (ACS) Stop the Bleed® training in response to the shooting June 14 at the Republican congressional baseball team practice in Alexandria, VA.
In the AMA House of Delegates, which is composed of 545 members, the College was represented by its six-member delegation.
The meeting has eight reference committees that focus on the following issues: bylaws and medical ethics, health care system, legislation, medical education, public health, medical science and technology, governance, and medical practice. John H. Armstrong, MD, FACS, co-author of this article, chaired Reference Committee A, which reviewed items pertaining to the health care system. This article provides an overview of several issues of relevance to ACS members.
Medical education
The priority issue for the ACS at this meeting was protecting the profession’s autonomy in defining the standards of lifelong learning through Maintenance of Certification (MOC). Five items related to MOC and Continuing Medical Education (CME) resulted in considerable debate. Internists and medical specialists were particularly frustrated by their experiences with their certifying boards. While acknowledging opportunities to improve MOC, the College delegation took the lead in collaborating with other specialty and state societies to argue against referring MOC to the state legislatures and in creating a new solely CME-based certification system.
• Council on Medical Education Report 2, Update on Maintenance of Certification and Osteopathic Continuous Certification, provided an overview of board-specific MOC innovations that increase relevance and reduce cost. The report was amended to have the AMA advocate that physicians who participate in programs related to quality improvement and/or patient safety should receive credit for MOC Part IV.
• Resolution 302, Comprehensive Review of CME Process, directed the AMA, in collaboration with the Accreditation Council for Continuing Medical Education, to conduct a comprehensive review of the CME process on a national level, with the goal of decreasing costs and simplifying the process of providing CME.
• Resolution 316, Action Steps Regarding Maintenance of Certification, sponsored by the largest state delegations in the HOD (Arizona, California, Florida, Georgia, New York, Pennsylvania, and Texas), sought to extend AMA model state legislation against MOC by barring hospitals, health care insurers, and state licensing boards from linking nonparticipation in the American Board of Medical Specialties (ABMS) MOC process to exclusion from credentialing. Further, it sought to create AMA policy that would replace MOC with high-quality CME under the purview of a physician’s specialty society solely as the demonstration of lifelong learning in hospital, insurance, and licensing credentialing. The HOD conversation shifted from restricting how hospitals, licensing boards, and insurers can use MOC, to redefining MOC.
To address this challenge to professional autonomy while emphasizing the need for responsiveness to concerns across boards, the ACS Delegation applied a strategy that involved prepared talking points; caucus presentations by ACS Executive Director David B. Hoyt, MD, FACS, and Dr. Armstrong; reference committee testimony from Dr. Hoyt and Vice-Chair of the ACS Board of Regents Leigh A. Neumayer, MD, FACS, and ACS Regent James Denneny, MD, FACS, executive director, American Academy of Otolaryngology-Head and Neck Surgery; as well as hallway conversations and HOD floor discussion. As a result, the AMA affirmed that lifelong learning is a fundamental obligation of the profession and recognizes that, for a physician, it is best achieved through ongoing participation in a program of high-quality CME appropriate to the physician’s medical practice as determined by the relevant specialty society. The concept of the AMA lobbying hospital associations, health care insurers, and state licensing boards to not use the ABMS-sponsored MOC process with lifelong interval high stakes testing for credentialing, in addition to the idea of the AMA partnering with state medical associations and specialty societies to undertake a study to establish a separate program of certification by 2020, were referred for further consideration. The ACS will continue to emphasize that a core element of professionalism is self-regulation of lifelong learning through standard-setting.
• Resolution 318, Oppose Direct-to-Consumer Advertising of the ABMS MOC Product, asked the AMA to oppose direct-to-consumer marketing of the ABMS MOC product in print media, social media, apps, and websites that specifically target patients and their families, including but not limited to the promotion of false or misleading claims linking MOC participation with improved patient health outcomes and experiences where limited evidence exists. This item was referred for further study.
• Resolution 319, Public Access to Initial Board Certification Status of Time-Limited ABMS Diplomates, was adopted. The AMA now advocates that the initial certification status of time-limited diplomates be listed and publicly available on all ABMS and ABMS member board websites and physician certification databases and that the names and initial certification status of time-limited diplomates remain on ABMS and ABMS member board websites or physician certification databases, even if the diplomate opts out of MOC participation.
Health system
• Council on Medical Service Report 6, Expansion of U.S. Veterans’ Health Care Choices, was adopted with an amendment offered by the ACS delegation. The AMA will encourage the Veterans Health Administration (VHA) to engage with VHA physicians to explore and develop solutions to improve the health care choices of veterans. The AMA also will continue to support efforts to improve the Veterans Choice Program (VCP) and make it permanent, advocate for new funding to support expansion of the VCP, and encourage the acceleration of interoperability of electronic personal and health records and the exchange of medical records between VHA and non-VHA physicians.
• Substitute Resolution 115, Out-of-Network Care, combined four resolutions on the subject of greatest interest in the Reference Committee A hearing—surprise (unanticipated) billing for out-of-network care. It was adopted with eight AMA principles regarding unanticipated out-of-network care and AMA development of model state legislation addressing the coverage of and payment for unexpected unanticipated out-of-network care.
Public health
• Resolution 419, Improving Physicians’ Ability to Discuss Firearm Safety, was adopted with minor amendments. As a result, the AMA is working with appropriate stakeholders to develop state-specific guidance for physicians on how to counsel patients to reduce their risk for firearm-related injury or death by suicide, including guidance on when and how to ask sensitive questions about firearm ownership, access, and use; and clarification on the circumstances under which physicians are permitted or may be required to disclose the content of such conversations to family members, law enforcement, or other third parties.
Medical practice
• Substitute Resolution 706, Concurrent and Overlapping Surgery, was adopted. The AMA will work with interested national medical specialty societies on issues related to concurrent and overlapping surgery.
AMA elections
The June meeting is when AMA officers, trustees, and councilors are elected. Barbara McAneny, MD, an oncologist from New Mexico, was elected AMA president-elect. Three ACS-endorsed candidates were successful in their bids to serve on the Council on Medical Education. Liana Puscas, MD, MHS, FACS, associate professor of surgery, Duke University School of Medicine, Durham, NC, and Luke Selby, MD, a general surgery resident, University of Colorado School of Medicine, Denver, were reelected; Krystal Tomei, MD, assistant professor of neurosurgery, Case Western Reserve University School of Medicine, Cleveland, OH, was elected.
Surgical Caucus
The Surgical Caucus hosted a well-attended educational session, Cultivating and Protecting Your Digital Presence: Do’s and Don’ts of Social Media. Speakers included Deanna Attai, MD, FACS, a breast surgeon and assistant clinical professor of surgery, David Geffen School of Medicine, University of California, Los Angeles, and Ravi Goel, MD, an ophthalmologist in private practice in Cherry Hill, NJ, and delegate from the American Academy of Ophthalmology. Both presentations emphasized that surgeons need to manage their online reputation in practice reviews and other web content. Drs. Attai and Goel provided constructive ideas for personal and professional branding through social media, as well as practical ways for surgeons to build and protect their social media reputation. Both presentations are available on the Surgical Caucus web page at facs.org/advocacy/ama-house-of-delegates/surgical-caucus.
The ACS delegation successfully put forth the College’s priorities at the June AMA HOD meeting. The delegation is now working with the ACS Division of Advocacy and Health Policy, the Health Policy and Advocacy Group, and other committees to develop next steps in shaping AMA policy consistent with College principles.
The next AMA meeting is the Interim Meeting in Honolulu, HI, November 11–14. The College’s delegation welcomes input from Fellows regarding issues of importance to surgeons. Comments and questions should be directed to [email protected].
ACS delegation at the AMA HOD
John H. Armstrong, MD, FACS (Delegation Chair), acute care surgery, Ocala, FL
Brian J. Gavitt, MD, MPH (also Young Physicians Section delegate), general surgery, Cincinnati, OH
Jacob Moalem, MD, FACS, general surgery, Rochester, NY
Leigh A. Neumayer, MD, FACS, general surgery, Tucson, AZ; Vice-Chair, ACS Board of Regents
Naveen F. Sangji, MD, (also Resident and Fellow Section delegate), general surgery resident, Boston, MA
Patricia L. Turner, MD, FACS, general surgery, Chicago, IL; Director, ACS Division of Member Services; Chair, AMA Council on Medical Education
Dr. Armstrong is affiliate associate professor of surgery, University of South Florida Morsani College of Medicine, and former Florida Surgeon General and Secretary of Health (2012–2016). He is a member of the American College of Surgeons (ACS) Health Policy and Advocacy Group, and Past-Chair, ACS Professional Association political action committee (ACSPA-SurgeonsPAC).
Mr. Sutton is Manager, State Affairs, ACS Division of Advocacy and Health Policy.
The American Medical Association (AMA) annual House of Delegates (HOD) meeting took place June 10–14, in Chicago, IL. The agenda included 62 reports from the AMA board and councils and 186 resolutions from state medical associations, specialty societies, and AMA sections.
The uncertain mood of the HOD was shaped by congressional activity on the Affordable Care Act and a competitive election for AMA president-elect. At the start of the meeting’s final day, the HOD paused to remember the lives lost and changed one year earlier at the Pulse nightclub in Orlando, FL. The HOD also noted the value of American College of Surgeons (ACS) Stop the Bleed® training in response to the shooting June 14 at the Republican congressional baseball team practice in Alexandria, VA.
In the AMA House of Delegates, which is composed of 545 members, the College was represented by its six-member delegation.
The meeting has eight reference committees that focus on the following issues: bylaws and medical ethics, health care system, legislation, medical education, public health, medical science and technology, governance, and medical practice. John H. Armstrong, MD, FACS, co-author of this article, chaired Reference Committee A, which reviewed items pertaining to the health care system. This article provides an overview of several issues of relevance to ACS members.
Medical education
The priority issue for the ACS at this meeting was protecting the profession’s autonomy in defining the standards of lifelong learning through Maintenance of Certification (MOC). Five items related to MOC and Continuing Medical Education (CME) resulted in considerable debate. Internists and medical specialists were particularly frustrated by their experiences with their certifying boards. While acknowledging opportunities to improve MOC, the College delegation took the lead in collaborating with other specialty and state societies to argue against referring MOC to the state legislatures and in creating a new solely CME-based certification system.
• Council on Medical Education Report 2, Update on Maintenance of Certification and Osteopathic Continuous Certification, provided an overview of board-specific MOC innovations that increase relevance and reduce cost. The report was amended to have the AMA advocate that physicians who participate in programs related to quality improvement and/or patient safety should receive credit for MOC Part IV.
• Resolution 302, Comprehensive Review of CME Process, directed the AMA, in collaboration with the Accreditation Council for Continuing Medical Education, to conduct a comprehensive review of the CME process on a national level, with the goal of decreasing costs and simplifying the process of providing CME.
• Resolution 316, Action Steps Regarding Maintenance of Certification, sponsored by the largest state delegations in the HOD (Arizona, California, Florida, Georgia, New York, Pennsylvania, and Texas), sought to extend AMA model state legislation against MOC by barring hospitals, health care insurers, and state licensing boards from linking nonparticipation in the American Board of Medical Specialties (ABMS) MOC process to exclusion from credentialing. Further, it sought to create AMA policy that would replace MOC with high-quality CME under the purview of a physician’s specialty society solely as the demonstration of lifelong learning in hospital, insurance, and licensing credentialing. The HOD conversation shifted from restricting how hospitals, licensing boards, and insurers can use MOC, to redefining MOC.
To address this challenge to professional autonomy while emphasizing the need for responsiveness to concerns across boards, the ACS Delegation applied a strategy that involved prepared talking points; caucus presentations by ACS Executive Director David B. Hoyt, MD, FACS, and Dr. Armstrong; reference committee testimony from Dr. Hoyt and Vice-Chair of the ACS Board of Regents Leigh A. Neumayer, MD, FACS, and ACS Regent James Denneny, MD, FACS, executive director, American Academy of Otolaryngology-Head and Neck Surgery; as well as hallway conversations and HOD floor discussion. As a result, the AMA affirmed that lifelong learning is a fundamental obligation of the profession and recognizes that, for a physician, it is best achieved through ongoing participation in a program of high-quality CME appropriate to the physician’s medical practice as determined by the relevant specialty society. The concept of the AMA lobbying hospital associations, health care insurers, and state licensing boards to not use the ABMS-sponsored MOC process with lifelong interval high stakes testing for credentialing, in addition to the idea of the AMA partnering with state medical associations and specialty societies to undertake a study to establish a separate program of certification by 2020, were referred for further consideration. The ACS will continue to emphasize that a core element of professionalism is self-regulation of lifelong learning through standard-setting.
• Resolution 318, Oppose Direct-to-Consumer Advertising of the ABMS MOC Product, asked the AMA to oppose direct-to-consumer marketing of the ABMS MOC product in print media, social media, apps, and websites that specifically target patients and their families, including but not limited to the promotion of false or misleading claims linking MOC participation with improved patient health outcomes and experiences where limited evidence exists. This item was referred for further study.
• Resolution 319, Public Access to Initial Board Certification Status of Time-Limited ABMS Diplomates, was adopted. The AMA now advocates that the initial certification status of time-limited diplomates be listed and publicly available on all ABMS and ABMS member board websites and physician certification databases and that the names and initial certification status of time-limited diplomates remain on ABMS and ABMS member board websites or physician certification databases, even if the diplomate opts out of MOC participation.
Health system
• Council on Medical Service Report 6, Expansion of U.S. Veterans’ Health Care Choices, was adopted with an amendment offered by the ACS delegation. The AMA will encourage the Veterans Health Administration (VHA) to engage with VHA physicians to explore and develop solutions to improve the health care choices of veterans. The AMA also will continue to support efforts to improve the Veterans Choice Program (VCP) and make it permanent, advocate for new funding to support expansion of the VCP, and encourage the acceleration of interoperability of electronic personal and health records and the exchange of medical records between VHA and non-VHA physicians.
• Substitute Resolution 115, Out-of-Network Care, combined four resolutions on the subject of greatest interest in the Reference Committee A hearing—surprise (unanticipated) billing for out-of-network care. It was adopted with eight AMA principles regarding unanticipated out-of-network care and AMA development of model state legislation addressing the coverage of and payment for unexpected unanticipated out-of-network care.
Public health
• Resolution 419, Improving Physicians’ Ability to Discuss Firearm Safety, was adopted with minor amendments. As a result, the AMA is working with appropriate stakeholders to develop state-specific guidance for physicians on how to counsel patients to reduce their risk for firearm-related injury or death by suicide, including guidance on when and how to ask sensitive questions about firearm ownership, access, and use; and clarification on the circumstances under which physicians are permitted or may be required to disclose the content of such conversations to family members, law enforcement, or other third parties.
Medical practice
• Substitute Resolution 706, Concurrent and Overlapping Surgery, was adopted. The AMA will work with interested national medical specialty societies on issues related to concurrent and overlapping surgery.
AMA elections
The June meeting is when AMA officers, trustees, and councilors are elected. Barbara McAneny, MD, an oncologist from New Mexico, was elected AMA president-elect. Three ACS-endorsed candidates were successful in their bids to serve on the Council on Medical Education. Liana Puscas, MD, MHS, FACS, associate professor of surgery, Duke University School of Medicine, Durham, NC, and Luke Selby, MD, a general surgery resident, University of Colorado School of Medicine, Denver, were reelected; Krystal Tomei, MD, assistant professor of neurosurgery, Case Western Reserve University School of Medicine, Cleveland, OH, was elected.
Surgical Caucus
The Surgical Caucus hosted a well-attended educational session, Cultivating and Protecting Your Digital Presence: Do’s and Don’ts of Social Media. Speakers included Deanna Attai, MD, FACS, a breast surgeon and assistant clinical professor of surgery, David Geffen School of Medicine, University of California, Los Angeles, and Ravi Goel, MD, an ophthalmologist in private practice in Cherry Hill, NJ, and delegate from the American Academy of Ophthalmology. Both presentations emphasized that surgeons need to manage their online reputation in practice reviews and other web content. Drs. Attai and Goel provided constructive ideas for personal and professional branding through social media, as well as practical ways for surgeons to build and protect their social media reputation. Both presentations are available on the Surgical Caucus web page at facs.org/advocacy/ama-house-of-delegates/surgical-caucus.
The ACS delegation successfully put forth the College’s priorities at the June AMA HOD meeting. The delegation is now working with the ACS Division of Advocacy and Health Policy, the Health Policy and Advocacy Group, and other committees to develop next steps in shaping AMA policy consistent with College principles.
The next AMA meeting is the Interim Meeting in Honolulu, HI, November 11–14. The College’s delegation welcomes input from Fellows regarding issues of importance to surgeons. Comments and questions should be directed to [email protected].
ACS delegation at the AMA HOD
John H. Armstrong, MD, FACS (Delegation Chair), acute care surgery, Ocala, FL
Brian J. Gavitt, MD, MPH (also Young Physicians Section delegate), general surgery, Cincinnati, OH
Jacob Moalem, MD, FACS, general surgery, Rochester, NY
Leigh A. Neumayer, MD, FACS, general surgery, Tucson, AZ; Vice-Chair, ACS Board of Regents
Naveen F. Sangji, MD, (also Resident and Fellow Section delegate), general surgery resident, Boston, MA
Patricia L. Turner, MD, FACS, general surgery, Chicago, IL; Director, ACS Division of Member Services; Chair, AMA Council on Medical Education
Dr. Armstrong is affiliate associate professor of surgery, University of South Florida Morsani College of Medicine, and former Florida Surgeon General and Secretary of Health (2012–2016). He is a member of the American College of Surgeons (ACS) Health Policy and Advocacy Group, and Past-Chair, ACS Professional Association political action committee (ACSPA-SurgeonsPAC).
Mr. Sutton is Manager, State Affairs, ACS Division of Advocacy and Health Policy.
The American Medical Association (AMA) annual House of Delegates (HOD) meeting took place June 10–14, in Chicago, IL. The agenda included 62 reports from the AMA board and councils and 186 resolutions from state medical associations, specialty societies, and AMA sections.
The uncertain mood of the HOD was shaped by congressional activity on the Affordable Care Act and a competitive election for AMA president-elect. At the start of the meeting’s final day, the HOD paused to remember the lives lost and changed one year earlier at the Pulse nightclub in Orlando, FL. The HOD also noted the value of American College of Surgeons (ACS) Stop the Bleed® training in response to the shooting June 14 at the Republican congressional baseball team practice in Alexandria, VA.
In the AMA House of Delegates, which is composed of 545 members, the College was represented by its six-member delegation.
The meeting has eight reference committees that focus on the following issues: bylaws and medical ethics, health care system, legislation, medical education, public health, medical science and technology, governance, and medical practice. John H. Armstrong, MD, FACS, co-author of this article, chaired Reference Committee A, which reviewed items pertaining to the health care system. This article provides an overview of several issues of relevance to ACS members.
Medical education
The priority issue for the ACS at this meeting was protecting the profession’s autonomy in defining the standards of lifelong learning through Maintenance of Certification (MOC). Five items related to MOC and Continuing Medical Education (CME) resulted in considerable debate. Internists and medical specialists were particularly frustrated by their experiences with their certifying boards. While acknowledging opportunities to improve MOC, the College delegation took the lead in collaborating with other specialty and state societies to argue against referring MOC to the state legislatures and in creating a new solely CME-based certification system.
• Council on Medical Education Report 2, Update on Maintenance of Certification and Osteopathic Continuous Certification, provided an overview of board-specific MOC innovations that increase relevance and reduce cost. The report was amended to have the AMA advocate that physicians who participate in programs related to quality improvement and/or patient safety should receive credit for MOC Part IV.
• Resolution 302, Comprehensive Review of CME Process, directed the AMA, in collaboration with the Accreditation Council for Continuing Medical Education, to conduct a comprehensive review of the CME process on a national level, with the goal of decreasing costs and simplifying the process of providing CME.
• Resolution 316, Action Steps Regarding Maintenance of Certification, sponsored by the largest state delegations in the HOD (Arizona, California, Florida, Georgia, New York, Pennsylvania, and Texas), sought to extend AMA model state legislation against MOC by barring hospitals, health care insurers, and state licensing boards from linking nonparticipation in the American Board of Medical Specialties (ABMS) MOC process to exclusion from credentialing. Further, it sought to create AMA policy that would replace MOC with high-quality CME under the purview of a physician’s specialty society solely as the demonstration of lifelong learning in hospital, insurance, and licensing credentialing. The HOD conversation shifted from restricting how hospitals, licensing boards, and insurers can use MOC, to redefining MOC.
To address this challenge to professional autonomy while emphasizing the need for responsiveness to concerns across boards, the ACS Delegation applied a strategy that involved prepared talking points; caucus presentations by ACS Executive Director David B. Hoyt, MD, FACS, and Dr. Armstrong; reference committee testimony from Dr. Hoyt and Vice-Chair of the ACS Board of Regents Leigh A. Neumayer, MD, FACS, and ACS Regent James Denneny, MD, FACS, executive director, American Academy of Otolaryngology-Head and Neck Surgery; as well as hallway conversations and HOD floor discussion. As a result, the AMA affirmed that lifelong learning is a fundamental obligation of the profession and recognizes that, for a physician, it is best achieved through ongoing participation in a program of high-quality CME appropriate to the physician’s medical practice as determined by the relevant specialty society. The concept of the AMA lobbying hospital associations, health care insurers, and state licensing boards to not use the ABMS-sponsored MOC process with lifelong interval high stakes testing for credentialing, in addition to the idea of the AMA partnering with state medical associations and specialty societies to undertake a study to establish a separate program of certification by 2020, were referred for further consideration. The ACS will continue to emphasize that a core element of professionalism is self-regulation of lifelong learning through standard-setting.
• Resolution 318, Oppose Direct-to-Consumer Advertising of the ABMS MOC Product, asked the AMA to oppose direct-to-consumer marketing of the ABMS MOC product in print media, social media, apps, and websites that specifically target patients and their families, including but not limited to the promotion of false or misleading claims linking MOC participation with improved patient health outcomes and experiences where limited evidence exists. This item was referred for further study.
• Resolution 319, Public Access to Initial Board Certification Status of Time-Limited ABMS Diplomates, was adopted. The AMA now advocates that the initial certification status of time-limited diplomates be listed and publicly available on all ABMS and ABMS member board websites and physician certification databases and that the names and initial certification status of time-limited diplomates remain on ABMS and ABMS member board websites or physician certification databases, even if the diplomate opts out of MOC participation.
Health system
• Council on Medical Service Report 6, Expansion of U.S. Veterans’ Health Care Choices, was adopted with an amendment offered by the ACS delegation. The AMA will encourage the Veterans Health Administration (VHA) to engage with VHA physicians to explore and develop solutions to improve the health care choices of veterans. The AMA also will continue to support efforts to improve the Veterans Choice Program (VCP) and make it permanent, advocate for new funding to support expansion of the VCP, and encourage the acceleration of interoperability of electronic personal and health records and the exchange of medical records between VHA and non-VHA physicians.
• Substitute Resolution 115, Out-of-Network Care, combined four resolutions on the subject of greatest interest in the Reference Committee A hearing—surprise (unanticipated) billing for out-of-network care. It was adopted with eight AMA principles regarding unanticipated out-of-network care and AMA development of model state legislation addressing the coverage of and payment for unexpected unanticipated out-of-network care.
Public health
• Resolution 419, Improving Physicians’ Ability to Discuss Firearm Safety, was adopted with minor amendments. As a result, the AMA is working with appropriate stakeholders to develop state-specific guidance for physicians on how to counsel patients to reduce their risk for firearm-related injury or death by suicide, including guidance on when and how to ask sensitive questions about firearm ownership, access, and use; and clarification on the circumstances under which physicians are permitted or may be required to disclose the content of such conversations to family members, law enforcement, or other third parties.
Medical practice
• Substitute Resolution 706, Concurrent and Overlapping Surgery, was adopted. The AMA will work with interested national medical specialty societies on issues related to concurrent and overlapping surgery.
AMA elections
The June meeting is when AMA officers, trustees, and councilors are elected. Barbara McAneny, MD, an oncologist from New Mexico, was elected AMA president-elect. Three ACS-endorsed candidates were successful in their bids to serve on the Council on Medical Education. Liana Puscas, MD, MHS, FACS, associate professor of surgery, Duke University School of Medicine, Durham, NC, and Luke Selby, MD, a general surgery resident, University of Colorado School of Medicine, Denver, were reelected; Krystal Tomei, MD, assistant professor of neurosurgery, Case Western Reserve University School of Medicine, Cleveland, OH, was elected.
Surgical Caucus
The Surgical Caucus hosted a well-attended educational session, Cultivating and Protecting Your Digital Presence: Do’s and Don’ts of Social Media. Speakers included Deanna Attai, MD, FACS, a breast surgeon and assistant clinical professor of surgery, David Geffen School of Medicine, University of California, Los Angeles, and Ravi Goel, MD, an ophthalmologist in private practice in Cherry Hill, NJ, and delegate from the American Academy of Ophthalmology. Both presentations emphasized that surgeons need to manage their online reputation in practice reviews and other web content. Drs. Attai and Goel provided constructive ideas for personal and professional branding through social media, as well as practical ways for surgeons to build and protect their social media reputation. Both presentations are available on the Surgical Caucus web page at facs.org/advocacy/ama-house-of-delegates/surgical-caucus.
The ACS delegation successfully put forth the College’s priorities at the June AMA HOD meeting. The delegation is now working with the ACS Division of Advocacy and Health Policy, the Health Policy and Advocacy Group, and other committees to develop next steps in shaping AMA policy consistent with College principles.
The next AMA meeting is the Interim Meeting in Honolulu, HI, November 11–14. The College’s delegation welcomes input from Fellows regarding issues of importance to surgeons. Comments and questions should be directed to [email protected].
ACS delegation at the AMA HOD
John H. Armstrong, MD, FACS (Delegation Chair), acute care surgery, Ocala, FL
Brian J. Gavitt, MD, MPH (also Young Physicians Section delegate), general surgery, Cincinnati, OH
Jacob Moalem, MD, FACS, general surgery, Rochester, NY
Leigh A. Neumayer, MD, FACS, general surgery, Tucson, AZ; Vice-Chair, ACS Board of Regents
Naveen F. Sangji, MD, (also Resident and Fellow Section delegate), general surgery resident, Boston, MA
Patricia L. Turner, MD, FACS, general surgery, Chicago, IL; Director, ACS Division of Member Services; Chair, AMA Council on Medical Education
Dr. Armstrong is affiliate associate professor of surgery, University of South Florida Morsani College of Medicine, and former Florida Surgeon General and Secretary of Health (2012–2016). He is a member of the American College of Surgeons (ACS) Health Policy and Advocacy Group, and Past-Chair, ACS Professional Association political action committee (ACSPA-SurgeonsPAC).
Mr. Sutton is Manager, State Affairs, ACS Division of Advocacy and Health Policy.
Timothy A. M. Chuter, MD, FACS, receives 2017 ACS Jacobson Innovation Award
The American College of Surgeons (ACS) presented the 2017 Jacobson Innovation Award to Timothy A. M. Chuter, MB, BS, DM, FACS, at a dinner in his honor June 9 in Chicago. Dr. Chuter is professor of surgery at the University of California, San Francisco (UCSF), where he practices vascular surgery with a focus on the endovascular reconstruction of aneurysms involving the aortic arch and thoracoabdominal aorta.
The Jacobson Innovation Award honors living surgeons who have developed innovative devices or techniques in any field of surgery and is made possible through a gift from Julius H. Jacobson II, MD, FACS, and his wife Joan. Dr. Jacobson is a general vascular surgeon known for his pioneering work in the development of microsurgery.
Leading the way in endovascular aneurysm repair, Dr. Chuter was recognized for his role in the development of endovascular aneurysm repair. He was the first individual to design and implant bifurcated stent grafts to treat abdominal aortic aneurysms, based on the idea that if an aneurysm has branches—at the aortic arch or the bifurcation of the common iliac artery, for example—the endovascular prosthesis also should have branches. Because the most common site for aortic aneurysm involves the distal abdominal aorta, bifurcated endovascular repair has become the most accepted method of aneurysm repair worldwide.
In the years between 1993 and 2000, the scope of endovascular repair rapidly expanded, with several firsts in the field, such as the first bifurcated stent grafts in 1993, the first endovascular repair of a ruptured aortic aneurysm in 1994, the first fenestrated stent grafts for aneurysms of the pararenal aorta in 1998, and the first branched stent grafts for the thoracoabdominal aorta in 2000. Dr. Chuter played a role in many of these developments, though none was the work of a single inventor. Dr. Chuter has said that he is proud to have contributed to several advances in endovascular aneurysm repair, not only by inventing new forms of repair, but also by mentoring surgical residents, fellows, and faculty.
In addition to his noted surgical skill, Dr. Chuter has been lauded for inventing and patenting the stent grafts that facilitate his work. Dr. Chuter’s devices and surgical techniques allow aneurysm repair in patients who otherwise might have no other chance of receiving effective treatment. He holds more than 40 patents, including 23 related to endovascular aortic stent-graft devices, stents, attachment systems, delivery systems, and component junctions.
Worldwide recognition
Dr. Chuter is the author or co-author of at least 145 peer-reviewed articles and 23 books or book chapters in the field. Other organizations have recognized his role in the development of endovascular aneurysm repair as well, including the Royal College of Surgeons, through their Kinmonth Medal in 1995; the Society for Vascular Surgery, through their Medal for Innovation in Vascular Surgery in 2008; and the Society for Endovascular Therapy in 2009.
Read more about Dr. Chuter and the Jacobson Innovation Award in the ACS press release at facs.org/media/press-releases/2017/jacobson061217. For a list of previous Jacobson Innovation Award winners, visit the ACS website at facs.org/about-acs/governance/acs-committees/honors-committee/jacobson-list.
The American College of Surgeons (ACS) presented the 2017 Jacobson Innovation Award to Timothy A. M. Chuter, MB, BS, DM, FACS, at a dinner in his honor June 9 in Chicago. Dr. Chuter is professor of surgery at the University of California, San Francisco (UCSF), where he practices vascular surgery with a focus on the endovascular reconstruction of aneurysms involving the aortic arch and thoracoabdominal aorta.
The Jacobson Innovation Award honors living surgeons who have developed innovative devices or techniques in any field of surgery and is made possible through a gift from Julius H. Jacobson II, MD, FACS, and his wife Joan. Dr. Jacobson is a general vascular surgeon known for his pioneering work in the development of microsurgery.
Leading the way in endovascular aneurysm repair, Dr. Chuter was recognized for his role in the development of endovascular aneurysm repair. He was the first individual to design and implant bifurcated stent grafts to treat abdominal aortic aneurysms, based on the idea that if an aneurysm has branches—at the aortic arch or the bifurcation of the common iliac artery, for example—the endovascular prosthesis also should have branches. Because the most common site for aortic aneurysm involves the distal abdominal aorta, bifurcated endovascular repair has become the most accepted method of aneurysm repair worldwide.
In the years between 1993 and 2000, the scope of endovascular repair rapidly expanded, with several firsts in the field, such as the first bifurcated stent grafts in 1993, the first endovascular repair of a ruptured aortic aneurysm in 1994, the first fenestrated stent grafts for aneurysms of the pararenal aorta in 1998, and the first branched stent grafts for the thoracoabdominal aorta in 2000. Dr. Chuter played a role in many of these developments, though none was the work of a single inventor. Dr. Chuter has said that he is proud to have contributed to several advances in endovascular aneurysm repair, not only by inventing new forms of repair, but also by mentoring surgical residents, fellows, and faculty.
In addition to his noted surgical skill, Dr. Chuter has been lauded for inventing and patenting the stent grafts that facilitate his work. Dr. Chuter’s devices and surgical techniques allow aneurysm repair in patients who otherwise might have no other chance of receiving effective treatment. He holds more than 40 patents, including 23 related to endovascular aortic stent-graft devices, stents, attachment systems, delivery systems, and component junctions.
Worldwide recognition
Dr. Chuter is the author or co-author of at least 145 peer-reviewed articles and 23 books or book chapters in the field. Other organizations have recognized his role in the development of endovascular aneurysm repair as well, including the Royal College of Surgeons, through their Kinmonth Medal in 1995; the Society for Vascular Surgery, through their Medal for Innovation in Vascular Surgery in 2008; and the Society for Endovascular Therapy in 2009.
Read more about Dr. Chuter and the Jacobson Innovation Award in the ACS press release at facs.org/media/press-releases/2017/jacobson061217. For a list of previous Jacobson Innovation Award winners, visit the ACS website at facs.org/about-acs/governance/acs-committees/honors-committee/jacobson-list.
The American College of Surgeons (ACS) presented the 2017 Jacobson Innovation Award to Timothy A. M. Chuter, MB, BS, DM, FACS, at a dinner in his honor June 9 in Chicago. Dr. Chuter is professor of surgery at the University of California, San Francisco (UCSF), where he practices vascular surgery with a focus on the endovascular reconstruction of aneurysms involving the aortic arch and thoracoabdominal aorta.
The Jacobson Innovation Award honors living surgeons who have developed innovative devices or techniques in any field of surgery and is made possible through a gift from Julius H. Jacobson II, MD, FACS, and his wife Joan. Dr. Jacobson is a general vascular surgeon known for his pioneering work in the development of microsurgery.
Leading the way in endovascular aneurysm repair, Dr. Chuter was recognized for his role in the development of endovascular aneurysm repair. He was the first individual to design and implant bifurcated stent grafts to treat abdominal aortic aneurysms, based on the idea that if an aneurysm has branches—at the aortic arch or the bifurcation of the common iliac artery, for example—the endovascular prosthesis also should have branches. Because the most common site for aortic aneurysm involves the distal abdominal aorta, bifurcated endovascular repair has become the most accepted method of aneurysm repair worldwide.
In the years between 1993 and 2000, the scope of endovascular repair rapidly expanded, with several firsts in the field, such as the first bifurcated stent grafts in 1993, the first endovascular repair of a ruptured aortic aneurysm in 1994, the first fenestrated stent grafts for aneurysms of the pararenal aorta in 1998, and the first branched stent grafts for the thoracoabdominal aorta in 2000. Dr. Chuter played a role in many of these developments, though none was the work of a single inventor. Dr. Chuter has said that he is proud to have contributed to several advances in endovascular aneurysm repair, not only by inventing new forms of repair, but also by mentoring surgical residents, fellows, and faculty.
In addition to his noted surgical skill, Dr. Chuter has been lauded for inventing and patenting the stent grafts that facilitate his work. Dr. Chuter’s devices and surgical techniques allow aneurysm repair in patients who otherwise might have no other chance of receiving effective treatment. He holds more than 40 patents, including 23 related to endovascular aortic stent-graft devices, stents, attachment systems, delivery systems, and component junctions.
Worldwide recognition
Dr. Chuter is the author or co-author of at least 145 peer-reviewed articles and 23 books or book chapters in the field. Other organizations have recognized his role in the development of endovascular aneurysm repair as well, including the Royal College of Surgeons, through their Kinmonth Medal in 1995; the Society for Vascular Surgery, through their Medal for Innovation in Vascular Surgery in 2008; and the Society for Endovascular Therapy in 2009.
Read more about Dr. Chuter and the Jacobson Innovation Award in the ACS press release at facs.org/media/press-releases/2017/jacobson061217. For a list of previous Jacobson Innovation Award winners, visit the ACS website at facs.org/about-acs/governance/acs-committees/honors-committee/jacobson-list.
From the Washington Office: The Mission Zero Act
Surgeons are well familiar with the statistic from the Centers for Disease Control and Prevention (CDC) identifying trauma as the leading cause of death for children and adults under age 44. More Americans lose their lives each year to trauma than to AIDS and stroke combined. Unfortunately, nearly 45 million Americans live in areas more than an hour away from either a Level I or II trauma center. Ensuring access to trauma care requires many crucial components including trauma centers and appropriately trained physicians and nurses, all of which must dedicate extensive resources around the clock so that seriously injured patients have the best possible chance for survival.
It has long been a top legislative priority of the ACS to establish and maintain adequate funding for high-quality trauma systems throughout the United States, including those systems operated by our armed forces.The ACS was a sponsor of the National Academy of Medicine (NAM) report entitled, A National Trauma Care System: Integrating Military and Civilian Trauma Systems to Achieve Zero Preventable Deaths After Injury. This report, released in June of 2016, outlines the steps necessary to secure a national trauma system and sets the goal of achieving zero preventable traumatic deaths.
In an effort to facilitate the achievement of the goals laid out in the report, The Mission Zero Act (H.R. 880) was introduced in the House of Representatives by Chairman of the House Energy and Commerce Health Subcommittee, Michael Burgess, MD (R-TX), Representatives Cathy Castor (D-FL), Gene Green (D-TX), and Richard Hudson (R-NC). Identical companion legislation was introduced in the Senate (S.1022) by Senators Johnny Isakson (R-GA), John Cornyn (R-TX), and Tammy Duckworth (D-IL). The Mission Zero Act creates a grant program to assist civilian trauma centers in partnering with military trauma professionals to establish a pathway to provide patients with the highest quality of trauma care in times of peace and war, thus taking a step in the direction of the NAM report recommendations.
Specifically, the legislation provides for:
• $40 million in grants to fund military trauma teams and providers to embed into civilian trauma facilities.
o Trauma centers are eligible for a $1 million grant to host military trauma teams at eligible high-acuity level 1 trauma centers
o Trauma centers are also eligible for grants to host individual providers ($100,000 for physician or $50,000 for non-physician providers) at eligible level I, II, or III trauma centers
As of today, the House and Senate versions of the Mission Zero Act have 25co-sponsors and 2 co-sponsors respectively. The ACS would very much like to build some momentum for the Mission Zero Act going into the fall when it is expected that there will be several large “must pass” pieces of legislation working their way through Congress to which the Mission Zero Act could potentially be attached. Accordingly, I respectfully ask all Fellows to take a few moments to visit the SurgeonsVoice website at www.surgeonsvoice.org, click on the Take Action tab on the right side of the page and send a message to their individual representatives and senators seeking support for this important legislation.
Until next month …
Dr. Bailey is a pediatric surgeon and Medical Director, Advocacy, for the Division of Advocacy and Health Policy in the ACS offices in Washington, DC.
Surgeons are well familiar with the statistic from the Centers for Disease Control and Prevention (CDC) identifying trauma as the leading cause of death for children and adults under age 44. More Americans lose their lives each year to trauma than to AIDS and stroke combined. Unfortunately, nearly 45 million Americans live in areas more than an hour away from either a Level I or II trauma center. Ensuring access to trauma care requires many crucial components including trauma centers and appropriately trained physicians and nurses, all of which must dedicate extensive resources around the clock so that seriously injured patients have the best possible chance for survival.
It has long been a top legislative priority of the ACS to establish and maintain adequate funding for high-quality trauma systems throughout the United States, including those systems operated by our armed forces.The ACS was a sponsor of the National Academy of Medicine (NAM) report entitled, A National Trauma Care System: Integrating Military and Civilian Trauma Systems to Achieve Zero Preventable Deaths After Injury. This report, released in June of 2016, outlines the steps necessary to secure a national trauma system and sets the goal of achieving zero preventable traumatic deaths.
In an effort to facilitate the achievement of the goals laid out in the report, The Mission Zero Act (H.R. 880) was introduced in the House of Representatives by Chairman of the House Energy and Commerce Health Subcommittee, Michael Burgess, MD (R-TX), Representatives Cathy Castor (D-FL), Gene Green (D-TX), and Richard Hudson (R-NC). Identical companion legislation was introduced in the Senate (S.1022) by Senators Johnny Isakson (R-GA), John Cornyn (R-TX), and Tammy Duckworth (D-IL). The Mission Zero Act creates a grant program to assist civilian trauma centers in partnering with military trauma professionals to establish a pathway to provide patients with the highest quality of trauma care in times of peace and war, thus taking a step in the direction of the NAM report recommendations.
Specifically, the legislation provides for:
• $40 million in grants to fund military trauma teams and providers to embed into civilian trauma facilities.
o Trauma centers are eligible for a $1 million grant to host military trauma teams at eligible high-acuity level 1 trauma centers
o Trauma centers are also eligible for grants to host individual providers ($100,000 for physician or $50,000 for non-physician providers) at eligible level I, II, or III trauma centers
As of today, the House and Senate versions of the Mission Zero Act have 25co-sponsors and 2 co-sponsors respectively. The ACS would very much like to build some momentum for the Mission Zero Act going into the fall when it is expected that there will be several large “must pass” pieces of legislation working their way through Congress to which the Mission Zero Act could potentially be attached. Accordingly, I respectfully ask all Fellows to take a few moments to visit the SurgeonsVoice website at www.surgeonsvoice.org, click on the Take Action tab on the right side of the page and send a message to their individual representatives and senators seeking support for this important legislation.
Until next month …
Dr. Bailey is a pediatric surgeon and Medical Director, Advocacy, for the Division of Advocacy and Health Policy in the ACS offices in Washington, DC.
Surgeons are well familiar with the statistic from the Centers for Disease Control and Prevention (CDC) identifying trauma as the leading cause of death for children and adults under age 44. More Americans lose their lives each year to trauma than to AIDS and stroke combined. Unfortunately, nearly 45 million Americans live in areas more than an hour away from either a Level I or II trauma center. Ensuring access to trauma care requires many crucial components including trauma centers and appropriately trained physicians and nurses, all of which must dedicate extensive resources around the clock so that seriously injured patients have the best possible chance for survival.
It has long been a top legislative priority of the ACS to establish and maintain adequate funding for high-quality trauma systems throughout the United States, including those systems operated by our armed forces.The ACS was a sponsor of the National Academy of Medicine (NAM) report entitled, A National Trauma Care System: Integrating Military and Civilian Trauma Systems to Achieve Zero Preventable Deaths After Injury. This report, released in June of 2016, outlines the steps necessary to secure a national trauma system and sets the goal of achieving zero preventable traumatic deaths.
In an effort to facilitate the achievement of the goals laid out in the report, The Mission Zero Act (H.R. 880) was introduced in the House of Representatives by Chairman of the House Energy and Commerce Health Subcommittee, Michael Burgess, MD (R-TX), Representatives Cathy Castor (D-FL), Gene Green (D-TX), and Richard Hudson (R-NC). Identical companion legislation was introduced in the Senate (S.1022) by Senators Johnny Isakson (R-GA), John Cornyn (R-TX), and Tammy Duckworth (D-IL). The Mission Zero Act creates a grant program to assist civilian trauma centers in partnering with military trauma professionals to establish a pathway to provide patients with the highest quality of trauma care in times of peace and war, thus taking a step in the direction of the NAM report recommendations.
Specifically, the legislation provides for:
• $40 million in grants to fund military trauma teams and providers to embed into civilian trauma facilities.
o Trauma centers are eligible for a $1 million grant to host military trauma teams at eligible high-acuity level 1 trauma centers
o Trauma centers are also eligible for grants to host individual providers ($100,000 for physician or $50,000 for non-physician providers) at eligible level I, II, or III trauma centers
As of today, the House and Senate versions of the Mission Zero Act have 25co-sponsors and 2 co-sponsors respectively. The ACS would very much like to build some momentum for the Mission Zero Act going into the fall when it is expected that there will be several large “must pass” pieces of legislation working their way through Congress to which the Mission Zero Act could potentially be attached. Accordingly, I respectfully ask all Fellows to take a few moments to visit the SurgeonsVoice website at www.surgeonsvoice.org, click on the Take Action tab on the right side of the page and send a message to their individual representatives and senators seeking support for this important legislation.
Until next month …
Dr. Bailey is a pediatric surgeon and Medical Director, Advocacy, for the Division of Advocacy and Health Policy in the ACS offices in Washington, DC.
ACS Committee on Trauma series highlights effort to complete nation’s trauma system
Today in America, more people survive serious injury than any time in the past. Yet trauma remains the leading killer of young people and military service members during combat. Trauma has been characterized as the “neglected epidemic of our time.”
A 2016 report by the National Academies of Science, Engineering and Medicine (NASEM) has added new momentum to the effort to complete the nation’s trauma system. It found one in five trauma deaths could be prevented through stronger Federal leadership, greater research funding to improve outcomes, strengthening prehospital care, distributing trauma centers based on need and integrating military and civilian trauma care into one national trauma system.
In a five-part series of stories, “Putting the Pieces Together: A National Effort to Complete the U.S. Trauma System,” the American College of Surgeons Committee on Trauma will share steps being taken to complete the nation’s trauma system. Stories will look at the history of trauma care and the trauma system in America, partnerships with the military to translate battlefield lessons to the home front and back again, the challenges facing America’s trauma systems, and the steps leading experts are taking to fill the gaps in the system and achieve the goal of zero preventable deaths and disability from injury.
Read the series online at www.facs.org/trauma or follow the ACS COT on Twitter @ACSTrauma.
Today in America, more people survive serious injury than any time in the past. Yet trauma remains the leading killer of young people and military service members during combat. Trauma has been characterized as the “neglected epidemic of our time.”
A 2016 report by the National Academies of Science, Engineering and Medicine (NASEM) has added new momentum to the effort to complete the nation’s trauma system. It found one in five trauma deaths could be prevented through stronger Federal leadership, greater research funding to improve outcomes, strengthening prehospital care, distributing trauma centers based on need and integrating military and civilian trauma care into one national trauma system.
In a five-part series of stories, “Putting the Pieces Together: A National Effort to Complete the U.S. Trauma System,” the American College of Surgeons Committee on Trauma will share steps being taken to complete the nation’s trauma system. Stories will look at the history of trauma care and the trauma system in America, partnerships with the military to translate battlefield lessons to the home front and back again, the challenges facing America’s trauma systems, and the steps leading experts are taking to fill the gaps in the system and achieve the goal of zero preventable deaths and disability from injury.
Read the series online at www.facs.org/trauma or follow the ACS COT on Twitter @ACSTrauma.
Today in America, more people survive serious injury than any time in the past. Yet trauma remains the leading killer of young people and military service members during combat. Trauma has been characterized as the “neglected epidemic of our time.”
A 2016 report by the National Academies of Science, Engineering and Medicine (NASEM) has added new momentum to the effort to complete the nation’s trauma system. It found one in five trauma deaths could be prevented through stronger Federal leadership, greater research funding to improve outcomes, strengthening prehospital care, distributing trauma centers based on need and integrating military and civilian trauma care into one national trauma system.
In a five-part series of stories, “Putting the Pieces Together: A National Effort to Complete the U.S. Trauma System,” the American College of Surgeons Committee on Trauma will share steps being taken to complete the nation’s trauma system. Stories will look at the history of trauma care and the trauma system in America, partnerships with the military to translate battlefield lessons to the home front and back again, the challenges facing America’s trauma systems, and the steps leading experts are taking to fill the gaps in the system and achieve the goal of zero preventable deaths and disability from injury.
Read the series online at www.facs.org/trauma or follow the ACS COT on Twitter @ACSTrauma.









