User login
First case of COVID-19 presenting as Guillain-Barré reported
The patient was a 61-year-old woman returning home from Wuhan during the pandemic.
“GBS is an autoimmune neuropathy, which could be triggered by various infections,” said corresponding author Sheng Chen, MD, PhD, of Shanghai Jiao Tong University School of Medicine in China. However, “Our single case report only suggests a possible association between GBS and SARS-CoV-2 infection. It may or may not have a causal relationship,” Dr. Chen noted.
The case study was published online April 1 in Lancet Neurology.
GBS presentation
The female patient returned from Wuhan on January 19 but denied having any fever, cough, chest pain, or diarrhea. She presented on January 23 with acute weakness in both legs and severe fatigue that progressed.
At presentation, temperature was normal, oxygen saturation was 99% on room air, and the patient’s respiratory rate was 16 breaths per minute. She was not tested for SARS-CoV-2 at that point.
A neurologic examination revealed symmetric weakness (Medical Research Council grade 4/5) and areflexia in both legs and feet. The patient’s symptoms had progressed 3 days after admission, and testing revealed decreased sensation to light touch and pinprick.
Admission laboratory test results indicated a low lymphocyte count and thrombocytopenia. Results of nerve conduction studies performed on day 5 of hospitalization were consistent with demyelinating neuropathy.
She was diagnosed with GBS and given intravenous immunoglobulin. On day 8, she developed a dry cough and fever, and a chest CT showed ground-glass opacities in both lungs. At this point, she was tested for SARS-CoV-2, and the results were positive.
The patient was immediately transferred to an isolation room and received supportive care and antiviral drugs. Her condition improved gradually, and her lymphocyte and thrombocyte counts were normal on day 20.
At discharge on day 30, she had normal muscle strength in both arms and legs, and tendon reflexes in both legs and feet had returned. Her respiratory symptoms had resolved as well. A second SARS-CoV-2 test was negative.
Different pattern from Zika
Two relatives of the patient who had been with her during her hospital stay also tested positive for SARS-CoV-2 and were isolated and treated. All of the hospital staff that cared for the patient, including two neurologists and six nurses, tested negative for SARS-CoV-2.
Given the temporal association, a SARS-CoV-2 infection could be responsible for the development of GBS in this patient, the investigators noted. They added that the onset of GBS symptoms overlapped with the period of SARS-CoV-2 infection.
“Hence Guillain-Barré syndrome associated with SARS-CoV-2 might follow the pattern of a parainfectious profile, instead of the classic postinfectious profile, as reported in Guillain-Barré syndrome associated with Zika virus,” the researchers wrote.
“More cases with epidemiological data are necessary to support a causal relationship” between SARS-CoV-2 infection and GBS, said Dr. Chen.
“However, we still suggest physicians who encounter an acute GBS patient from a pandemic area protect themselves carefully and test [for the] virus on admission. If the result is positive, the patient needs to be isolated,” Dr. Chen said.
This article was first published on Medscape.com.
The patient was a 61-year-old woman returning home from Wuhan during the pandemic.
“GBS is an autoimmune neuropathy, which could be triggered by various infections,” said corresponding author Sheng Chen, MD, PhD, of Shanghai Jiao Tong University School of Medicine in China. However, “Our single case report only suggests a possible association between GBS and SARS-CoV-2 infection. It may or may not have a causal relationship,” Dr. Chen noted.
The case study was published online April 1 in Lancet Neurology.
GBS presentation
The female patient returned from Wuhan on January 19 but denied having any fever, cough, chest pain, or diarrhea. She presented on January 23 with acute weakness in both legs and severe fatigue that progressed.
At presentation, temperature was normal, oxygen saturation was 99% on room air, and the patient’s respiratory rate was 16 breaths per minute. She was not tested for SARS-CoV-2 at that point.
A neurologic examination revealed symmetric weakness (Medical Research Council grade 4/5) and areflexia in both legs and feet. The patient’s symptoms had progressed 3 days after admission, and testing revealed decreased sensation to light touch and pinprick.
Admission laboratory test results indicated a low lymphocyte count and thrombocytopenia. Results of nerve conduction studies performed on day 5 of hospitalization were consistent with demyelinating neuropathy.
She was diagnosed with GBS and given intravenous immunoglobulin. On day 8, she developed a dry cough and fever, and a chest CT showed ground-glass opacities in both lungs. At this point, she was tested for SARS-CoV-2, and the results were positive.
The patient was immediately transferred to an isolation room and received supportive care and antiviral drugs. Her condition improved gradually, and her lymphocyte and thrombocyte counts were normal on day 20.
At discharge on day 30, she had normal muscle strength in both arms and legs, and tendon reflexes in both legs and feet had returned. Her respiratory symptoms had resolved as well. A second SARS-CoV-2 test was negative.
Different pattern from Zika
Two relatives of the patient who had been with her during her hospital stay also tested positive for SARS-CoV-2 and were isolated and treated. All of the hospital staff that cared for the patient, including two neurologists and six nurses, tested negative for SARS-CoV-2.
Given the temporal association, a SARS-CoV-2 infection could be responsible for the development of GBS in this patient, the investigators noted. They added that the onset of GBS symptoms overlapped with the period of SARS-CoV-2 infection.
“Hence Guillain-Barré syndrome associated with SARS-CoV-2 might follow the pattern of a parainfectious profile, instead of the classic postinfectious profile, as reported in Guillain-Barré syndrome associated with Zika virus,” the researchers wrote.
“More cases with epidemiological data are necessary to support a causal relationship” between SARS-CoV-2 infection and GBS, said Dr. Chen.
“However, we still suggest physicians who encounter an acute GBS patient from a pandemic area protect themselves carefully and test [for the] virus on admission. If the result is positive, the patient needs to be isolated,” Dr. Chen said.
This article was first published on Medscape.com.
The patient was a 61-year-old woman returning home from Wuhan during the pandemic.
“GBS is an autoimmune neuropathy, which could be triggered by various infections,” said corresponding author Sheng Chen, MD, PhD, of Shanghai Jiao Tong University School of Medicine in China. However, “Our single case report only suggests a possible association between GBS and SARS-CoV-2 infection. It may or may not have a causal relationship,” Dr. Chen noted.
The case study was published online April 1 in Lancet Neurology.
GBS presentation
The female patient returned from Wuhan on January 19 but denied having any fever, cough, chest pain, or diarrhea. She presented on January 23 with acute weakness in both legs and severe fatigue that progressed.
At presentation, temperature was normal, oxygen saturation was 99% on room air, and the patient’s respiratory rate was 16 breaths per minute. She was not tested for SARS-CoV-2 at that point.
A neurologic examination revealed symmetric weakness (Medical Research Council grade 4/5) and areflexia in both legs and feet. The patient’s symptoms had progressed 3 days after admission, and testing revealed decreased sensation to light touch and pinprick.
Admission laboratory test results indicated a low lymphocyte count and thrombocytopenia. Results of nerve conduction studies performed on day 5 of hospitalization were consistent with demyelinating neuropathy.
She was diagnosed with GBS and given intravenous immunoglobulin. On day 8, she developed a dry cough and fever, and a chest CT showed ground-glass opacities in both lungs. At this point, she was tested for SARS-CoV-2, and the results were positive.
The patient was immediately transferred to an isolation room and received supportive care and antiviral drugs. Her condition improved gradually, and her lymphocyte and thrombocyte counts were normal on day 20.
At discharge on day 30, she had normal muscle strength in both arms and legs, and tendon reflexes in both legs and feet had returned. Her respiratory symptoms had resolved as well. A second SARS-CoV-2 test was negative.
Different pattern from Zika
Two relatives of the patient who had been with her during her hospital stay also tested positive for SARS-CoV-2 and were isolated and treated. All of the hospital staff that cared for the patient, including two neurologists and six nurses, tested negative for SARS-CoV-2.
Given the temporal association, a SARS-CoV-2 infection could be responsible for the development of GBS in this patient, the investigators noted. They added that the onset of GBS symptoms overlapped with the period of SARS-CoV-2 infection.
“Hence Guillain-Barré syndrome associated with SARS-CoV-2 might follow the pattern of a parainfectious profile, instead of the classic postinfectious profile, as reported in Guillain-Barré syndrome associated with Zika virus,” the researchers wrote.
“More cases with epidemiological data are necessary to support a causal relationship” between SARS-CoV-2 infection and GBS, said Dr. Chen.
“However, we still suggest physicians who encounter an acute GBS patient from a pandemic area protect themselves carefully and test [for the] virus on admission. If the result is positive, the patient needs to be isolated,” Dr. Chen said.
This article was first published on Medscape.com.
Abortion is essential health care
In my New Mexico reproductive health clinic one week in early April, I saw more than twice the number of patients usually scheduled, all seeking abortion care. Two-thirds of those patients were from Texas – some came from towns as close as 6 hours away, and at least two drove for more than 11 hours to receive care at our clinic. In addition to the many reasons women pursue abortion care, all of my patients had an overriding concern about the COVID-19 pandemic. Many worried for the safety of their parents and children; some worried about the safety of continuing a pregnancy during the pandemic; and many were worried for themselves because of the risk involved in their employment or their status as the sole breadwinner for their families. One patient chose an abortion for severe fetal anomalies diagnosed in the early second trimester; she had an appointment with a provider in Texas, which was canceled the day the Texas abortion ban was reinstated. New Mexico, more than 10 hours away, was the closest location to receive the care she needed; she traveled by car with her children.
I am fortunate to live in New Mexico. On March 24, New Mexico Secretary of Health Kathyleen “Kathy” Kunkel affirmed reproductive health care as an essential service. The American College of Obstetricians and Gynecologists, the U.S. professional organization for reproductive health care, agrees. In a joint statement with seven other professional organizations, they emphasize the importance of abortion access: “Abortion is an essential component of comprehensive health care. It is also a time-sensitive service. The consequences of being unable to obtain an abortion profoundly impact a person’s life, health, and well-being.”
Anti-abortion politicians are using the crisis as an opportunity to restrict health care access as they have done for my patients who have driven hundreds of miles for essential care they should receive in their home communities. My heart goes out to our patients and the burden they have been forced to take on at a time when our politicians should be protecting and ensuring their safety.
Dr. Espey is an obstetrician and gynecologist in New Mexico. She has no relevant financial disclosures. Dr. Espey is a member of the Ob.Gyn. News Editorial Advisory Board. Email her at [email protected].
In my New Mexico reproductive health clinic one week in early April, I saw more than twice the number of patients usually scheduled, all seeking abortion care. Two-thirds of those patients were from Texas – some came from towns as close as 6 hours away, and at least two drove for more than 11 hours to receive care at our clinic. In addition to the many reasons women pursue abortion care, all of my patients had an overriding concern about the COVID-19 pandemic. Many worried for the safety of their parents and children; some worried about the safety of continuing a pregnancy during the pandemic; and many were worried for themselves because of the risk involved in their employment or their status as the sole breadwinner for their families. One patient chose an abortion for severe fetal anomalies diagnosed in the early second trimester; she had an appointment with a provider in Texas, which was canceled the day the Texas abortion ban was reinstated. New Mexico, more than 10 hours away, was the closest location to receive the care she needed; she traveled by car with her children.
I am fortunate to live in New Mexico. On March 24, New Mexico Secretary of Health Kathyleen “Kathy” Kunkel affirmed reproductive health care as an essential service. The American College of Obstetricians and Gynecologists, the U.S. professional organization for reproductive health care, agrees. In a joint statement with seven other professional organizations, they emphasize the importance of abortion access: “Abortion is an essential component of comprehensive health care. It is also a time-sensitive service. The consequences of being unable to obtain an abortion profoundly impact a person’s life, health, and well-being.”
Anti-abortion politicians are using the crisis as an opportunity to restrict health care access as they have done for my patients who have driven hundreds of miles for essential care they should receive in their home communities. My heart goes out to our patients and the burden they have been forced to take on at a time when our politicians should be protecting and ensuring their safety.
Dr. Espey is an obstetrician and gynecologist in New Mexico. She has no relevant financial disclosures. Dr. Espey is a member of the Ob.Gyn. News Editorial Advisory Board. Email her at [email protected].
In my New Mexico reproductive health clinic one week in early April, I saw more than twice the number of patients usually scheduled, all seeking abortion care. Two-thirds of those patients were from Texas – some came from towns as close as 6 hours away, and at least two drove for more than 11 hours to receive care at our clinic. In addition to the many reasons women pursue abortion care, all of my patients had an overriding concern about the COVID-19 pandemic. Many worried for the safety of their parents and children; some worried about the safety of continuing a pregnancy during the pandemic; and many were worried for themselves because of the risk involved in their employment or their status as the sole breadwinner for their families. One patient chose an abortion for severe fetal anomalies diagnosed in the early second trimester; she had an appointment with a provider in Texas, which was canceled the day the Texas abortion ban was reinstated. New Mexico, more than 10 hours away, was the closest location to receive the care she needed; she traveled by car with her children.
I am fortunate to live in New Mexico. On March 24, New Mexico Secretary of Health Kathyleen “Kathy” Kunkel affirmed reproductive health care as an essential service. The American College of Obstetricians and Gynecologists, the U.S. professional organization for reproductive health care, agrees. In a joint statement with seven other professional organizations, they emphasize the importance of abortion access: “Abortion is an essential component of comprehensive health care. It is also a time-sensitive service. The consequences of being unable to obtain an abortion profoundly impact a person’s life, health, and well-being.”
Anti-abortion politicians are using the crisis as an opportunity to restrict health care access as they have done for my patients who have driven hundreds of miles for essential care they should receive in their home communities. My heart goes out to our patients and the burden they have been forced to take on at a time when our politicians should be protecting and ensuring their safety.
Dr. Espey is an obstetrician and gynecologist in New Mexico. She has no relevant financial disclosures. Dr. Espey is a member of the Ob.Gyn. News Editorial Advisory Board. Email her at [email protected].
Small study links preterm birth, maternal preconception phthalate exposure
Maternal preconception exposure to phthalates was associated with increased risk of preterm birth, according to a study of 420 births to subfertile couples over a 13-year period.
Previous studies have shown increased risk of preterm birth associated with prenatal exposure to phthalates, which are commonly found in a range of household and commercial products as well as medical equipment and some pharmaceuticals.
“Our results suggest that female exposure to [4 di(2-ethylhexyl) phthalate] DEHP before conception might be an unrecognized risk factor for adverse pregnancy outcomes, often overlooked in clinical practice,” wrote Yu Zhang of the department of environmental health at Harvard T.H. Chan School of Public Health, Boston, and colleagues.
The prospective cohort study evaluated preconception urinary levels of phthalates and phthalate substitutes in 419 women and 229 men participating in the Environment and Reproductive Health (EARTH) study, a cohort of couples seeking fertility care at the Massachusetts General Hospital Fertility Center. The study cohort gave birth during 2005-2018. The average gestational age of the 420 singleton children born to this cohort was 39 weeks, with 8% (n = 34) born preterm.
Adjusted models showed that maternal preconception urinary concentrations of phthalates and of cyclohexane-1, 2-dicarboxylic acid monohydroxy isononyl ester (MHiNCH), a metabolite of a nonphthalate plasticizer substitute, were associated with a 50% and 70% increased risk of preterm birth, respectively (P = .01, .11), according to results published in JAMA Network Open .
Sensitivity analysis showed that maternal preconception MHiNCH concentrations above the median were associated with a fourfold increased risk of preterm birth (risk ratio, 4.02; P = .08), Maternal preconception MHiNCH concentrations were associated with an average 2-day reduction in gestational age (P = .02).
Covariate-adjusted models found that paternal urinary phthalate metabolite concentrations were associated with an increased risk of preterm birth (RR, 1.41; P = .09), but this association was attenuated toward zero (RR, 1.06) in models that accounted for maternal preconception phthalate concentrations. Sensitivity analysis of 228 couples found the associations of maternal preconception phthalate metabolite concentrations and preterm birth remained robust in three different models: a twofold increased risk in covariate-adjusted models (P < .001); an almost fivefold increased risk in adjusting for prenatal levels (RR, 4.98; P < .001); and a twofold risk (P = .001) in adjusting for paternal levels. “Couple-based analyses confirmed the results for an association between maternal preconception DEHP concentrations and increased risk of preterm birth,” the investigators said.
“To our knowledge, this is the first study evaluating couples’ exposure to phthalate metabolites during the preconception window and its association with preterm birth,” the researchers wrote. “Our findings support a novel hypothesis: Maternal phthalate exposure during the critical period before conception may be associated with shorter gestation.”
“This study is consistent with several, but not all, prior studies supporting maternal prenatal exposure to phthalates increase preterm birth,” said Mark P. Trolice, MD, professor of obstetrics and gynecology at the University of Central Florida, Orlando. “The uniqueness of the current study was the assessment of couples’ exposures and the outcome, though paternal exposure to phthalates did not demonstrate a significant association.”
Dr. Trolice noted that about 25% of women in the study were smokers, but the study didn’t adjust for tobacco use and phthalate exposure, and 85% of the women were white. He urged caution in applying the study results in practice, adding that the study didn’t adjust for method of conception. “Assisted reproductive technology, multiple gestation, and advanced age are all known risk factors for preterm birth,”
The National Institute of Environmental Health Science funded the study. Two study coauthors received grants from the NIEHS, one coauthor received grants from the National Institutes of Health, and one received a grant from the Canadian Institutes of Health Research. No other disclosures were reported. Dr. Trolice has no financial relationships to disclose.
SOURCE: Zhang Y et al. JAMA Network Open. 2020; doi: 10.1001/jamanetworkopen.2020.2159.
Maternal preconception exposure to phthalates was associated with increased risk of preterm birth, according to a study of 420 births to subfertile couples over a 13-year period.
Previous studies have shown increased risk of preterm birth associated with prenatal exposure to phthalates, which are commonly found in a range of household and commercial products as well as medical equipment and some pharmaceuticals.
“Our results suggest that female exposure to [4 di(2-ethylhexyl) phthalate] DEHP before conception might be an unrecognized risk factor for adverse pregnancy outcomes, often overlooked in clinical practice,” wrote Yu Zhang of the department of environmental health at Harvard T.H. Chan School of Public Health, Boston, and colleagues.
The prospective cohort study evaluated preconception urinary levels of phthalates and phthalate substitutes in 419 women and 229 men participating in the Environment and Reproductive Health (EARTH) study, a cohort of couples seeking fertility care at the Massachusetts General Hospital Fertility Center. The study cohort gave birth during 2005-2018. The average gestational age of the 420 singleton children born to this cohort was 39 weeks, with 8% (n = 34) born preterm.
Adjusted models showed that maternal preconception urinary concentrations of phthalates and of cyclohexane-1, 2-dicarboxylic acid monohydroxy isononyl ester (MHiNCH), a metabolite of a nonphthalate plasticizer substitute, were associated with a 50% and 70% increased risk of preterm birth, respectively (P = .01, .11), according to results published in JAMA Network Open .
Sensitivity analysis showed that maternal preconception MHiNCH concentrations above the median were associated with a fourfold increased risk of preterm birth (risk ratio, 4.02; P = .08), Maternal preconception MHiNCH concentrations were associated with an average 2-day reduction in gestational age (P = .02).
Covariate-adjusted models found that paternal urinary phthalate metabolite concentrations were associated with an increased risk of preterm birth (RR, 1.41; P = .09), but this association was attenuated toward zero (RR, 1.06) in models that accounted for maternal preconception phthalate concentrations. Sensitivity analysis of 228 couples found the associations of maternal preconception phthalate metabolite concentrations and preterm birth remained robust in three different models: a twofold increased risk in covariate-adjusted models (P < .001); an almost fivefold increased risk in adjusting for prenatal levels (RR, 4.98; P < .001); and a twofold risk (P = .001) in adjusting for paternal levels. “Couple-based analyses confirmed the results for an association between maternal preconception DEHP concentrations and increased risk of preterm birth,” the investigators said.
“To our knowledge, this is the first study evaluating couples’ exposure to phthalate metabolites during the preconception window and its association with preterm birth,” the researchers wrote. “Our findings support a novel hypothesis: Maternal phthalate exposure during the critical period before conception may be associated with shorter gestation.”
“This study is consistent with several, but not all, prior studies supporting maternal prenatal exposure to phthalates increase preterm birth,” said Mark P. Trolice, MD, professor of obstetrics and gynecology at the University of Central Florida, Orlando. “The uniqueness of the current study was the assessment of couples’ exposures and the outcome, though paternal exposure to phthalates did not demonstrate a significant association.”
Dr. Trolice noted that about 25% of women in the study were smokers, but the study didn’t adjust for tobacco use and phthalate exposure, and 85% of the women were white. He urged caution in applying the study results in practice, adding that the study didn’t adjust for method of conception. “Assisted reproductive technology, multiple gestation, and advanced age are all known risk factors for preterm birth,”
The National Institute of Environmental Health Science funded the study. Two study coauthors received grants from the NIEHS, one coauthor received grants from the National Institutes of Health, and one received a grant from the Canadian Institutes of Health Research. No other disclosures were reported. Dr. Trolice has no financial relationships to disclose.
SOURCE: Zhang Y et al. JAMA Network Open. 2020; doi: 10.1001/jamanetworkopen.2020.2159.
Maternal preconception exposure to phthalates was associated with increased risk of preterm birth, according to a study of 420 births to subfertile couples over a 13-year period.
Previous studies have shown increased risk of preterm birth associated with prenatal exposure to phthalates, which are commonly found in a range of household and commercial products as well as medical equipment and some pharmaceuticals.
“Our results suggest that female exposure to [4 di(2-ethylhexyl) phthalate] DEHP before conception might be an unrecognized risk factor for adverse pregnancy outcomes, often overlooked in clinical practice,” wrote Yu Zhang of the department of environmental health at Harvard T.H. Chan School of Public Health, Boston, and colleagues.
The prospective cohort study evaluated preconception urinary levels of phthalates and phthalate substitutes in 419 women and 229 men participating in the Environment and Reproductive Health (EARTH) study, a cohort of couples seeking fertility care at the Massachusetts General Hospital Fertility Center. The study cohort gave birth during 2005-2018. The average gestational age of the 420 singleton children born to this cohort was 39 weeks, with 8% (n = 34) born preterm.
Adjusted models showed that maternal preconception urinary concentrations of phthalates and of cyclohexane-1, 2-dicarboxylic acid monohydroxy isononyl ester (MHiNCH), a metabolite of a nonphthalate plasticizer substitute, were associated with a 50% and 70% increased risk of preterm birth, respectively (P = .01, .11), according to results published in JAMA Network Open .
Sensitivity analysis showed that maternal preconception MHiNCH concentrations above the median were associated with a fourfold increased risk of preterm birth (risk ratio, 4.02; P = .08), Maternal preconception MHiNCH concentrations were associated with an average 2-day reduction in gestational age (P = .02).
Covariate-adjusted models found that paternal urinary phthalate metabolite concentrations were associated with an increased risk of preterm birth (RR, 1.41; P = .09), but this association was attenuated toward zero (RR, 1.06) in models that accounted for maternal preconception phthalate concentrations. Sensitivity analysis of 228 couples found the associations of maternal preconception phthalate metabolite concentrations and preterm birth remained robust in three different models: a twofold increased risk in covariate-adjusted models (P < .001); an almost fivefold increased risk in adjusting for prenatal levels (RR, 4.98; P < .001); and a twofold risk (P = .001) in adjusting for paternal levels. “Couple-based analyses confirmed the results for an association between maternal preconception DEHP concentrations and increased risk of preterm birth,” the investigators said.
“To our knowledge, this is the first study evaluating couples’ exposure to phthalate metabolites during the preconception window and its association with preterm birth,” the researchers wrote. “Our findings support a novel hypothesis: Maternal phthalate exposure during the critical period before conception may be associated with shorter gestation.”
“This study is consistent with several, but not all, prior studies supporting maternal prenatal exposure to phthalates increase preterm birth,” said Mark P. Trolice, MD, professor of obstetrics and gynecology at the University of Central Florida, Orlando. “The uniqueness of the current study was the assessment of couples’ exposures and the outcome, though paternal exposure to phthalates did not demonstrate a significant association.”
Dr. Trolice noted that about 25% of women in the study were smokers, but the study didn’t adjust for tobacco use and phthalate exposure, and 85% of the women were white. He urged caution in applying the study results in practice, adding that the study didn’t adjust for method of conception. “Assisted reproductive technology, multiple gestation, and advanced age are all known risk factors for preterm birth,”
The National Institute of Environmental Health Science funded the study. Two study coauthors received grants from the NIEHS, one coauthor received grants from the National Institutes of Health, and one received a grant from the Canadian Institutes of Health Research. No other disclosures were reported. Dr. Trolice has no financial relationships to disclose.
SOURCE: Zhang Y et al. JAMA Network Open. 2020; doi: 10.1001/jamanetworkopen.2020.2159.
FROM JAMA NETWORK OPEN
Reproductive health care in the time of COVID-19
It is often said that a crisis brings out the best and worst in people, and I think we are definitely seeing that when it comes to the responses to reproductive health, family planning, and abortion care during this global pandemic.
Many national and international organizations have published strong statements of support for the importance of continuing reproductive health services. These organizations include the American College of Obstetricians and Gynecologists, American Board of Obstetrics & Gynecology, the American Society for Reproductive Medicine, the Society of Family Planning, the Society for Maternal-Fetal Medicine, Society of Family Planning, American Medical Association, the World Health Organization, and the Ethiopian Society of Obstetricians and Gynecologists. They state the obvious, which is that
We do not have complete knowledge of what the novel coronavirus 2019 does to a developing pregnancy, especially early in pregnancy. Many people who are struggling with all the uncertainty of this time – job, health, housing, food, school – may decide it is not the best moment to be adding to their family.
These concerns apply as well to the need to maintain and prioritize contraceptive services. Stay-at-home orders have put people in close quarters for long periods of time, and we are already getting reports of increased sexual intercourse, as well as increased sexual violence, both of which could result in a need for abortion if contraception is not accessible. Additionally, many women are expressing a concern for whether they will still have a job or have a job again when this first wave of the crisis passes, so they are wanting to access contraception now when they can afford to do so.
I was personally very proud of and grateful to Barbara A. Goff, MD, the chair of my department at the University of Washington, Seattle, for stating clearly in the first email she sent to faculty about canceling elective procedures and visits that family planning and abortion is not elective. My heart goes out to my colleagues and the patients who are in states that are using this opportunity to act poorly and use COVID-19 as another excuse to legislate against abortion and contraception. It demonstrates horrifying gender discrimination during a time when we should really be focusing on keeping everyone healthy.
I predict there will be an increase in the numbers of abortions after this crisis ebbs, and an increase in the numbers of term deliveries. The time to influence that is now.
Dr. Prager is professor of obstetrics and gynecology, chief of the family planning division, and director of the family planning fellowship at the University of Washington, Seattle. She also is professor of obstetrics and gynecology at St. Paul’s Hospital and Millennium Medical College in Addis Ababa, Ethiopia. Dr. Prager is a member of the Ob.Gyn. News editorial advisory board. She said she has no relevant financial disclosures. Email Dr. Prager at [email protected].
It is often said that a crisis brings out the best and worst in people, and I think we are definitely seeing that when it comes to the responses to reproductive health, family planning, and abortion care during this global pandemic.
Many national and international organizations have published strong statements of support for the importance of continuing reproductive health services. These organizations include the American College of Obstetricians and Gynecologists, American Board of Obstetrics & Gynecology, the American Society for Reproductive Medicine, the Society of Family Planning, the Society for Maternal-Fetal Medicine, Society of Family Planning, American Medical Association, the World Health Organization, and the Ethiopian Society of Obstetricians and Gynecologists. They state the obvious, which is that
We do not have complete knowledge of what the novel coronavirus 2019 does to a developing pregnancy, especially early in pregnancy. Many people who are struggling with all the uncertainty of this time – job, health, housing, food, school – may decide it is not the best moment to be adding to their family.
These concerns apply as well to the need to maintain and prioritize contraceptive services. Stay-at-home orders have put people in close quarters for long periods of time, and we are already getting reports of increased sexual intercourse, as well as increased sexual violence, both of which could result in a need for abortion if contraception is not accessible. Additionally, many women are expressing a concern for whether they will still have a job or have a job again when this first wave of the crisis passes, so they are wanting to access contraception now when they can afford to do so.
I was personally very proud of and grateful to Barbara A. Goff, MD, the chair of my department at the University of Washington, Seattle, for stating clearly in the first email she sent to faculty about canceling elective procedures and visits that family planning and abortion is not elective. My heart goes out to my colleagues and the patients who are in states that are using this opportunity to act poorly and use COVID-19 as another excuse to legislate against abortion and contraception. It demonstrates horrifying gender discrimination during a time when we should really be focusing on keeping everyone healthy.
I predict there will be an increase in the numbers of abortions after this crisis ebbs, and an increase in the numbers of term deliveries. The time to influence that is now.
Dr. Prager is professor of obstetrics and gynecology, chief of the family planning division, and director of the family planning fellowship at the University of Washington, Seattle. She also is professor of obstetrics and gynecology at St. Paul’s Hospital and Millennium Medical College in Addis Ababa, Ethiopia. Dr. Prager is a member of the Ob.Gyn. News editorial advisory board. She said she has no relevant financial disclosures. Email Dr. Prager at [email protected].
It is often said that a crisis brings out the best and worst in people, and I think we are definitely seeing that when it comes to the responses to reproductive health, family planning, and abortion care during this global pandemic.
Many national and international organizations have published strong statements of support for the importance of continuing reproductive health services. These organizations include the American College of Obstetricians and Gynecologists, American Board of Obstetrics & Gynecology, the American Society for Reproductive Medicine, the Society of Family Planning, the Society for Maternal-Fetal Medicine, Society of Family Planning, American Medical Association, the World Health Organization, and the Ethiopian Society of Obstetricians and Gynecologists. They state the obvious, which is that
We do not have complete knowledge of what the novel coronavirus 2019 does to a developing pregnancy, especially early in pregnancy. Many people who are struggling with all the uncertainty of this time – job, health, housing, food, school – may decide it is not the best moment to be adding to their family.
These concerns apply as well to the need to maintain and prioritize contraceptive services. Stay-at-home orders have put people in close quarters for long periods of time, and we are already getting reports of increased sexual intercourse, as well as increased sexual violence, both of which could result in a need for abortion if contraception is not accessible. Additionally, many women are expressing a concern for whether they will still have a job or have a job again when this first wave of the crisis passes, so they are wanting to access contraception now when they can afford to do so.
I was personally very proud of and grateful to Barbara A. Goff, MD, the chair of my department at the University of Washington, Seattle, for stating clearly in the first email she sent to faculty about canceling elective procedures and visits that family planning and abortion is not elective. My heart goes out to my colleagues and the patients who are in states that are using this opportunity to act poorly and use COVID-19 as another excuse to legislate against abortion and contraception. It demonstrates horrifying gender discrimination during a time when we should really be focusing on keeping everyone healthy.
I predict there will be an increase in the numbers of abortions after this crisis ebbs, and an increase in the numbers of term deliveries. The time to influence that is now.
Dr. Prager is professor of obstetrics and gynecology, chief of the family planning division, and director of the family planning fellowship at the University of Washington, Seattle. She also is professor of obstetrics and gynecology at St. Paul’s Hospital and Millennium Medical College in Addis Ababa, Ethiopia. Dr. Prager is a member of the Ob.Gyn. News editorial advisory board. She said she has no relevant financial disclosures. Email Dr. Prager at [email protected].
Amid coronavirus concerns, researchers urge mental health interventions for patients with dementia
according to a letter published online ahead of print March 30 in Lancet. Consistent with recommendations from Alzheimer’s Disease International and other dementia experts, teams that include mental health professionals, social workers, nursing home administrators, and volunteers should collaborate to provide mental health care for people with dementia. Experts in dementia should lead each team and support team members from other disciplines, wrote Huali Wang, MD, chair of clinical research at Peking University Institute of Mental Health in Beijing, and colleagues.
Interventions could be administered through telehealth, said the authors. Teams led by mental health professionals could use electronic media to provide self-help guidance for reducing stress, such as relaxation or meditation exercise. These teams also could use telephone hotlines to support behavioral management, and psychological counselors could provide online consultations for caregivers in nursing homes or in the community. “We encourage people who have a parent with dementia to have more frequent contact or spend more time with their parent, or to take on some of the caregiving duties so as to give the carer some respite time,” wrote Dr. Wang and colleagues.
Many local authorities are banning visits to nursing home residents to reduce the latter’s risk of COVID-19 infection. As a consequence, these elderly people are becoming more isolated, and anxiety is increasing among nursing home staffs.
In China, five organizations, including the Chinese Society of Geriatric Psychiatry and Alzheimer’s Disease Chinese, responded to the COVID-19 outbreak by publishing recommendations for providing mental health and psychosocial support. Groups of providers from various disciplines offered free counseling services for people with dementia and their caregivers. “These approaches minimized the complex impact of both COVID-19 outbreak and dementia,” wrote the authors.
“China has contained the epidemic, and business is starting to return to normal,” they continued. “We believe that learning lessons from China would empower the world to tackle the COVID-19 pandemic, with little risk of compromising the quality of life of people living with dementia and their carers.”
Dr. Wang has received lecture fees from Eisai China and Lundbeck China. She owns the copyright for the neuropsychiatric symptoms individualized management system. Her coauthors reported serving as advisory board members and receiving fees from companies such as Biogen, Novartis, and Genentech.
SOURCE: Wang H et al. Lancet. 2020 Mar 30. doi: 10.1016/S0140-6736(20)30755-8.
according to a letter published online ahead of print March 30 in Lancet. Consistent with recommendations from Alzheimer’s Disease International and other dementia experts, teams that include mental health professionals, social workers, nursing home administrators, and volunteers should collaborate to provide mental health care for people with dementia. Experts in dementia should lead each team and support team members from other disciplines, wrote Huali Wang, MD, chair of clinical research at Peking University Institute of Mental Health in Beijing, and colleagues.
Interventions could be administered through telehealth, said the authors. Teams led by mental health professionals could use electronic media to provide self-help guidance for reducing stress, such as relaxation or meditation exercise. These teams also could use telephone hotlines to support behavioral management, and psychological counselors could provide online consultations for caregivers in nursing homes or in the community. “We encourage people who have a parent with dementia to have more frequent contact or spend more time with their parent, or to take on some of the caregiving duties so as to give the carer some respite time,” wrote Dr. Wang and colleagues.
Many local authorities are banning visits to nursing home residents to reduce the latter’s risk of COVID-19 infection. As a consequence, these elderly people are becoming more isolated, and anxiety is increasing among nursing home staffs.
In China, five organizations, including the Chinese Society of Geriatric Psychiatry and Alzheimer’s Disease Chinese, responded to the COVID-19 outbreak by publishing recommendations for providing mental health and psychosocial support. Groups of providers from various disciplines offered free counseling services for people with dementia and their caregivers. “These approaches minimized the complex impact of both COVID-19 outbreak and dementia,” wrote the authors.
“China has contained the epidemic, and business is starting to return to normal,” they continued. “We believe that learning lessons from China would empower the world to tackle the COVID-19 pandemic, with little risk of compromising the quality of life of people living with dementia and their carers.”
Dr. Wang has received lecture fees from Eisai China and Lundbeck China. She owns the copyright for the neuropsychiatric symptoms individualized management system. Her coauthors reported serving as advisory board members and receiving fees from companies such as Biogen, Novartis, and Genentech.
SOURCE: Wang H et al. Lancet. 2020 Mar 30. doi: 10.1016/S0140-6736(20)30755-8.
according to a letter published online ahead of print March 30 in Lancet. Consistent with recommendations from Alzheimer’s Disease International and other dementia experts, teams that include mental health professionals, social workers, nursing home administrators, and volunteers should collaborate to provide mental health care for people with dementia. Experts in dementia should lead each team and support team members from other disciplines, wrote Huali Wang, MD, chair of clinical research at Peking University Institute of Mental Health in Beijing, and colleagues.
Interventions could be administered through telehealth, said the authors. Teams led by mental health professionals could use electronic media to provide self-help guidance for reducing stress, such as relaxation or meditation exercise. These teams also could use telephone hotlines to support behavioral management, and psychological counselors could provide online consultations for caregivers in nursing homes or in the community. “We encourage people who have a parent with dementia to have more frequent contact or spend more time with their parent, or to take on some of the caregiving duties so as to give the carer some respite time,” wrote Dr. Wang and colleagues.
Many local authorities are banning visits to nursing home residents to reduce the latter’s risk of COVID-19 infection. As a consequence, these elderly people are becoming more isolated, and anxiety is increasing among nursing home staffs.
In China, five organizations, including the Chinese Society of Geriatric Psychiatry and Alzheimer’s Disease Chinese, responded to the COVID-19 outbreak by publishing recommendations for providing mental health and psychosocial support. Groups of providers from various disciplines offered free counseling services for people with dementia and their caregivers. “These approaches minimized the complex impact of both COVID-19 outbreak and dementia,” wrote the authors.
“China has contained the epidemic, and business is starting to return to normal,” they continued. “We believe that learning lessons from China would empower the world to tackle the COVID-19 pandemic, with little risk of compromising the quality of life of people living with dementia and their carers.”
Dr. Wang has received lecture fees from Eisai China and Lundbeck China. She owns the copyright for the neuropsychiatric symptoms individualized management system. Her coauthors reported serving as advisory board members and receiving fees from companies such as Biogen, Novartis, and Genentech.
SOURCE: Wang H et al. Lancet. 2020 Mar 30. doi: 10.1016/S0140-6736(20)30755-8.
REPORTING FROM THE LANCET
Colorectal cancer: Proposed treatment guidelines for the COVID-19 era
In light of the rapid changes affecting cancer clinics due to the COVID-19 pandemic, Dr. David Kerr and Dr. Rachel Kerr, both specialists in gastrointestinal cancers at the University of Oxford in Oxford, United Kingdom, drafted these guidelines for the use of chemotherapy in colorectal cancer patients. Dr. Kerr and Dr. Kerr are putting forth this guidance as a topic for discussion and debate.
Our aim in developing these recommendations for the care of colorectal cancer patients in areas affected by the COVID-19 outbreak is to reduce the comorbidity of chemotherapy and decrease the risk of patients dying from COVID-19, weighed against the potential benefits of receiving chemotherapy. These recommendations are also designed to reduce the burden on chemotherapy units during a time of great pressure.
We have modified the guidelines in such a way that, we believe, will decrease the total number of patients receiving chemotherapy – particularly in the adjuvant setting – and reduce the overall immune impact of chemotherapy on these patients. Specifically, we suggest changing doublet chemotherapy to single-agent chemotherapy for some groups; changing to combinations involving capecitabine rather than bolus and infusional 5-FU for other patients; and, finally, making reasonable dose reductions upfront to reduce the risk for cycle 1 complications.
By changing from push-and-pump 5-FU to capecitabine for the vast majority of patients, we will both reduce the rates of neutropenia and decrease throughput in chemotherapy outpatient units, reducing requirements for weekly line flushing, pump disconnections, and other routine maintenance.
We continue to recommend the use of ToxNav germline genetic testing as a genetic screen for DPYD/ENOSF1 single-nucleotide polymorphisms (SNPs) to identify patients at high risk for fluoropyrimidine toxicity.
Use of biomarkers to sharpen prognosis should also be considered to refine therapeutic decisions.
Recommendations for stage II-III colorectal cancer
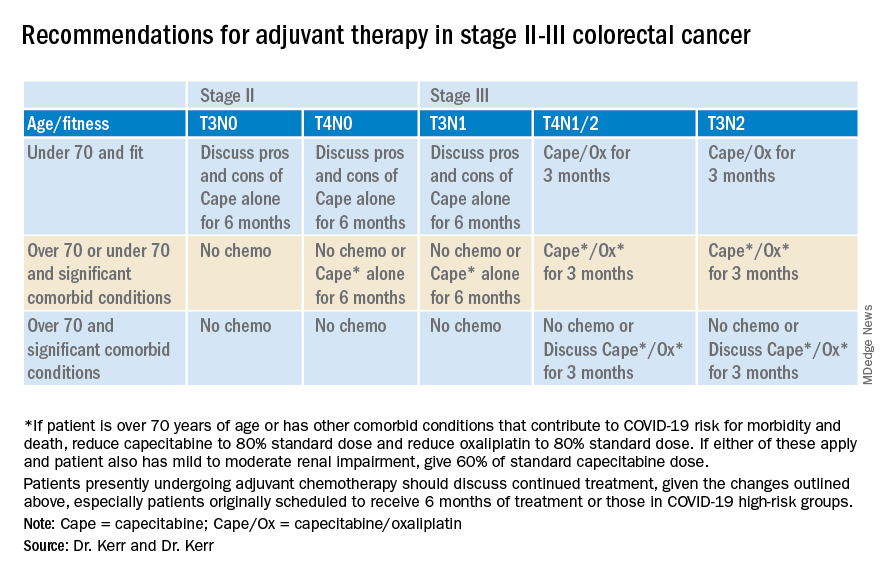
Recommendations for advanced colorectal cancer
Which regimen? Capecitabine/oxaliplatin should be the default backbone chemotherapy (rather than FOLFOX) in order to decrease the stress on infusion units.
Capecitabine plus irinotecan should be considered rather than FOLFIRI. However, in order to increase safety, reduce the dose of the capecitabine and the irinotecan, both to 80%, in all patient groups; and perhaps reduce the capecitabine dose further to 60% in those over the age of 70 or with significant comorbid conditions.
Treatment breaks. Full treatment breaks should be considered after 3 months of treatment in most patients with lower-volume, more indolent disease.
Treatment deintensification to capecitabine alone should be used in those with higher-volume disease (for example, more than 50% of liver replaced by tumor) at the beginning of treatment.
Deferring the start of any chemotherapy. Some older patients, or those with significant other comorbidities (that is, those who will be at increased risk for COVID-19 complications and death); who have low-volume disease, such as a couple of small lung metastases or a single liver metastasis; or who were diagnosed more than 12 months since adjuvant chemotherapy may decide to defer any chemotherapy for a period of time.
In these cases, we suggest rescanning at 3 months and discussing further treatment at that point. Some of these patients will be eligible for other interventions, such as resection, ablation, or stereotactic body radiation therapy. However, it will be important to consider the pressures on these other services during this unprecedented time.
Chemotherapy after resection of metastases. Given the lack of evidence and the present extenuating circumstances, we would not recommend any chemotherapy in this setting.
David J. Kerr, MD, CBE, MD, DSc, is a professor of cancer medicine at the University of Oxford. He is recognized internationally for his work in the research and treatment of colorectal cancer, and has founded three university spin-out companies: COBRA Therapeutics, Celleron Therapeutics, and Oxford Cancer Biomarkers. In 2002, he was appointed Commander of the British Empire by Queen Elizabeth. Rachel S. Kerr, MBChB, is a medical oncologist and associate professor of gastrointestinal oncology at the University of Oxford. She holds a UK Department of Health Fellowship, where she is clinical director of phase 3 trials in the oncology clinical trials office.
This article first appeared on Medscape.com.
In light of the rapid changes affecting cancer clinics due to the COVID-19 pandemic, Dr. David Kerr and Dr. Rachel Kerr, both specialists in gastrointestinal cancers at the University of Oxford in Oxford, United Kingdom, drafted these guidelines for the use of chemotherapy in colorectal cancer patients. Dr. Kerr and Dr. Kerr are putting forth this guidance as a topic for discussion and debate.
Our aim in developing these recommendations for the care of colorectal cancer patients in areas affected by the COVID-19 outbreak is to reduce the comorbidity of chemotherapy and decrease the risk of patients dying from COVID-19, weighed against the potential benefits of receiving chemotherapy. These recommendations are also designed to reduce the burden on chemotherapy units during a time of great pressure.
We have modified the guidelines in such a way that, we believe, will decrease the total number of patients receiving chemotherapy – particularly in the adjuvant setting – and reduce the overall immune impact of chemotherapy on these patients. Specifically, we suggest changing doublet chemotherapy to single-agent chemotherapy for some groups; changing to combinations involving capecitabine rather than bolus and infusional 5-FU for other patients; and, finally, making reasonable dose reductions upfront to reduce the risk for cycle 1 complications.
By changing from push-and-pump 5-FU to capecitabine for the vast majority of patients, we will both reduce the rates of neutropenia and decrease throughput in chemotherapy outpatient units, reducing requirements for weekly line flushing, pump disconnections, and other routine maintenance.
We continue to recommend the use of ToxNav germline genetic testing as a genetic screen for DPYD/ENOSF1 single-nucleotide polymorphisms (SNPs) to identify patients at high risk for fluoropyrimidine toxicity.
Use of biomarkers to sharpen prognosis should also be considered to refine therapeutic decisions.
Recommendations for stage II-III colorectal cancer

Recommendations for advanced colorectal cancer
Which regimen? Capecitabine/oxaliplatin should be the default backbone chemotherapy (rather than FOLFOX) in order to decrease the stress on infusion units.
Capecitabine plus irinotecan should be considered rather than FOLFIRI. However, in order to increase safety, reduce the dose of the capecitabine and the irinotecan, both to 80%, in all patient groups; and perhaps reduce the capecitabine dose further to 60% in those over the age of 70 or with significant comorbid conditions.
Treatment breaks. Full treatment breaks should be considered after 3 months of treatment in most patients with lower-volume, more indolent disease.
Treatment deintensification to capecitabine alone should be used in those with higher-volume disease (for example, more than 50% of liver replaced by tumor) at the beginning of treatment.
Deferring the start of any chemotherapy. Some older patients, or those with significant other comorbidities (that is, those who will be at increased risk for COVID-19 complications and death); who have low-volume disease, such as a couple of small lung metastases or a single liver metastasis; or who were diagnosed more than 12 months since adjuvant chemotherapy may decide to defer any chemotherapy for a period of time.
In these cases, we suggest rescanning at 3 months and discussing further treatment at that point. Some of these patients will be eligible for other interventions, such as resection, ablation, or stereotactic body radiation therapy. However, it will be important to consider the pressures on these other services during this unprecedented time.
Chemotherapy after resection of metastases. Given the lack of evidence and the present extenuating circumstances, we would not recommend any chemotherapy in this setting.
David J. Kerr, MD, CBE, MD, DSc, is a professor of cancer medicine at the University of Oxford. He is recognized internationally for his work in the research and treatment of colorectal cancer, and has founded three university spin-out companies: COBRA Therapeutics, Celleron Therapeutics, and Oxford Cancer Biomarkers. In 2002, he was appointed Commander of the British Empire by Queen Elizabeth. Rachel S. Kerr, MBChB, is a medical oncologist and associate professor of gastrointestinal oncology at the University of Oxford. She holds a UK Department of Health Fellowship, where she is clinical director of phase 3 trials in the oncology clinical trials office.
This article first appeared on Medscape.com.
In light of the rapid changes affecting cancer clinics due to the COVID-19 pandemic, Dr. David Kerr and Dr. Rachel Kerr, both specialists in gastrointestinal cancers at the University of Oxford in Oxford, United Kingdom, drafted these guidelines for the use of chemotherapy in colorectal cancer patients. Dr. Kerr and Dr. Kerr are putting forth this guidance as a topic for discussion and debate.
Our aim in developing these recommendations for the care of colorectal cancer patients in areas affected by the COVID-19 outbreak is to reduce the comorbidity of chemotherapy and decrease the risk of patients dying from COVID-19, weighed against the potential benefits of receiving chemotherapy. These recommendations are also designed to reduce the burden on chemotherapy units during a time of great pressure.
We have modified the guidelines in such a way that, we believe, will decrease the total number of patients receiving chemotherapy – particularly in the adjuvant setting – and reduce the overall immune impact of chemotherapy on these patients. Specifically, we suggest changing doublet chemotherapy to single-agent chemotherapy for some groups; changing to combinations involving capecitabine rather than bolus and infusional 5-FU for other patients; and, finally, making reasonable dose reductions upfront to reduce the risk for cycle 1 complications.
By changing from push-and-pump 5-FU to capecitabine for the vast majority of patients, we will both reduce the rates of neutropenia and decrease throughput in chemotherapy outpatient units, reducing requirements for weekly line flushing, pump disconnections, and other routine maintenance.
We continue to recommend the use of ToxNav germline genetic testing as a genetic screen for DPYD/ENOSF1 single-nucleotide polymorphisms (SNPs) to identify patients at high risk for fluoropyrimidine toxicity.
Use of biomarkers to sharpen prognosis should also be considered to refine therapeutic decisions.
Recommendations for stage II-III colorectal cancer

Recommendations for advanced colorectal cancer
Which regimen? Capecitabine/oxaliplatin should be the default backbone chemotherapy (rather than FOLFOX) in order to decrease the stress on infusion units.
Capecitabine plus irinotecan should be considered rather than FOLFIRI. However, in order to increase safety, reduce the dose of the capecitabine and the irinotecan, both to 80%, in all patient groups; and perhaps reduce the capecitabine dose further to 60% in those over the age of 70 or with significant comorbid conditions.
Treatment breaks. Full treatment breaks should be considered after 3 months of treatment in most patients with lower-volume, more indolent disease.
Treatment deintensification to capecitabine alone should be used in those with higher-volume disease (for example, more than 50% of liver replaced by tumor) at the beginning of treatment.
Deferring the start of any chemotherapy. Some older patients, or those with significant other comorbidities (that is, those who will be at increased risk for COVID-19 complications and death); who have low-volume disease, such as a couple of small lung metastases or a single liver metastasis; or who were diagnosed more than 12 months since adjuvant chemotherapy may decide to defer any chemotherapy for a period of time.
In these cases, we suggest rescanning at 3 months and discussing further treatment at that point. Some of these patients will be eligible for other interventions, such as resection, ablation, or stereotactic body radiation therapy. However, it will be important to consider the pressures on these other services during this unprecedented time.
Chemotherapy after resection of metastases. Given the lack of evidence and the present extenuating circumstances, we would not recommend any chemotherapy in this setting.
David J. Kerr, MD, CBE, MD, DSc, is a professor of cancer medicine at the University of Oxford. He is recognized internationally for his work in the research and treatment of colorectal cancer, and has founded three university spin-out companies: COBRA Therapeutics, Celleron Therapeutics, and Oxford Cancer Biomarkers. In 2002, he was appointed Commander of the British Empire by Queen Elizabeth. Rachel S. Kerr, MBChB, is a medical oncologist and associate professor of gastrointestinal oncology at the University of Oxford. She holds a UK Department of Health Fellowship, where she is clinical director of phase 3 trials in the oncology clinical trials office.
This article first appeared on Medscape.com.
When the going gets tough, ophthalmologists call the rheumatologist
MAUI, HAWAII – When a rheumatologist gets a call from an ophthalmologist regarding a patient with an inflamed eye and elevated intraocular pressure unresponsive to the eye specialist’s customary array of topical, systemic, and intraocular implanted corticosteroids, that’s a patient who needs to be seen immediately, Alvin F. Wells, MD, PhD, said at the 2020 Rheumatology Winter Clinical Symposium.
Elevated intraocular pressure due to uveitis or scleritis can result in blindness. Eye specialists call upon rheumatologists here because of their expertise in step-up therapy with methotrexate and other traditional oral disease-modifying antirheumatic drugs as well as biologic agents.
“Here’s my treatment approach to inflammatory eye disease: We’re pulling out all the guns,” declared Dr. Wells, a rheumatologist with a special interest in eye disease. He is director of the Rheumatology and Immunotherapy Center in Franklin, Wisc., with academic appointments to the Karolinska Institute in Stockholm, Duke University, and Marquette University.
Uveitis involves inflammation of the iris, choroid, and ciliary body. A straightforward case of noninfectious anterior uveitis will typically respond to 2 weeks of topical steroid drops, or sometimes even topical NSAID drops.
However, noninfectious posterior, intermediate, or panuveitis is another matter. In those circumstances, he gives the patient 125 mg of methylprednisolone by intramuscular injection and a 20-mg dose of oral methotrexate at that first clinic visit. The patient is sent home with a prescription for oral prednisone, tapering over 2-3 weeks, and another for methotrexate at 15-25 mg/week plus 1-2 mg/day of folic acid. Dr. Wells also gives consideration to add-on azathioprine or mycophenolate mofetil. He views multidrug therapy as having a sound rationale because multiple inflammatory pathways are involved in noninfectious uveitis.
“Ophthalmologists like to push for cyclophosphamide, but there’s no controlled data out there showing it’s effective in inflammatory eye disorders. It’s a pretty toxic regimen, and when you think about all the complications we see in using this drug to treat patients with lupus, I’d rather hold it in reserve for severe cases where we can go to it if we need to,” the rheumatologist explained.
He conducted a literature review to rank rheumatologic medications in terms of their evidence base for treatment of inflammatory ocular disorders. Among oral agents, at the top of the heap is methotrexate, whose efficacy for both noninfectious uveitis and scleritis is supported by multiple randomized, controlled studies. But mycophenolate mofetil is a reasonable alternative first-line corticosteroid-sparing agent, as demonstrated in the 265-patient multicenter FAST (First-line Antimetabolites as Steroid-sparing Treatment) trial sponsored by the National Eye Institute. That trial demonstrated no significant difference in treatment success at 6 months between methotrexate and mycophenolate mofetil.
Oral apremilast (Otezla) is approved for treatment of the oral ulcers of Behçet’s disease, but not for Behçet’s eye disease, where the experience is anecdotal.
Dr. Wells is quick to turn to adalimumab (Humira) when he deems a biologic to be warranted; indeed, it’s the only biologic approved for noninfectious uveitis. Of course, not everyone is a responder.
“Can we extrapolate that high-quality evidence of benefit for adalimumab to other drugs? Probably yes, and if you did that it would be for the IgG monoclonal antibodies that can cross the blood/aqueous barrier,” he said.
Infliximab (Remicade) is the biologic with the second-strongest supporting evidence in noninfectious uveitis. For the uveitis of Behçet’s disease, one of the most common rheumatic causes of inflammatory eye disease, Spanish investigators who conducted a nationwide nonrandomized study reported that both adalimumab and infliximab were effective, although adalimumab had superior outcomes at 1 year.
Uveitis is the most common extra-articular expression of axial spondyloarthritis (axSpA). In the open-label extension of the randomized RAPID-axSpA trial, patients randomized to certolizumab pegol (Cimzia) had a significantly lower incidence of uveitis flares than with placebo through 204 weeks of follow-up.
“The take-home message is we have some post hoc data here to say, ‘Hey, this could work in those patients who have inflammatory eye diseases in the setting of axSpA,’ ” Dr. Wells said.
The interleukin-6 receptor inhibitor tocilizumab (Actemra) “definitely works” for noninfectious uveitis, according to Dr. Wells, pointing to the positive results of the multicenter U.S. STOP-Uveitis study.
“The caveat here is tocilizumab has only been studied in the IV formulation. It’s too bad they didn’t use the [subcutaneous formulation]; you can’t get IV tocilizumab approved by payers in the U.S.,” according to the rheumatologist.
Based upon positive anecdotal case reports, Dr. Wells has a few patients on rituximab (Rituxan) for uveitis, with favorable results. The same for abatacept (Orencia).
It’s imperative that a patient on a biologic for uveitis undergo weekly ophthalmologic examinations. Only after the intraocular pressure is normal and inflammatory cells in the anterior chamber have waned is it appropriate to discontinue the biologic and slowly taper the methotrexate and any other oral disease-modifying antirheumatic drugs. Some experts argue for lifelong therapy in patients who’ve experienced uveitis. Dr. Wells disagrees, preferring to treat acute uveitis flares as they arise, although if underlying disease such as psoriatic arthritis or axSpA is present, some form of background therapy will probably be necessary.
Get to know teprotumumab
Rheumatologists who operate an infusion center are likely to increasingly be called upon by endocrinologists and ophthalmologists to administer intravenous teprotumumab-trbw (Tepezza), a human monoclonal antibody directed against the insulin-like growth factor 1 receptor that was approved earlier this year by the Food and Drug Administration as the first-ever drug for thyroid eye disease, a disfiguring and potentially blinding condition.
“This is really exciting,” Dr. Wells said. “The disease has an acute inflammatory stage, and that’s when you’ll be called on to give this drug. It makes a dramatic difference. Once a patient gets to the scarring phase there’s not a whole lot they can do other than surgery.”
In the pivotal phase 3 randomized trial, 83% of the teprotumumab group achieved the primary endpoint, a reduction in proptosis, or eye bulging, of at least 2 mm at week 24, compared with 10% of placebo-treated controls. The number needed to treat was 1.4. The chief side effects were muscle spasms, hair loss, fatigue, and nausea.
“You might say, ‘two millimeters, that’s nothing.’ But the primary drug used before teprotumumab was IV steroids, and there a 0.6-mm reduction in proptosis was considered improvement,” Dr. Wells observed.
Obtaining payer approval
“I’ve found over the last 10 years that when it comes to eye disease, insurance companies have a little more wiggle room,” he said. “They’re not going to let somebody go blind. You can get the references I’ve mentioned and show them the data. After all, we only have one biologic drug that’s been approved, and not everybody responds to it.
“Titrate your therapy based upon the intraocular pressure, the number of inflammatory cells in the anterior chamber, and any visual changes. You’ve got to be very aggressive with therapy, and don’t take no for an answer from the insurance companies,” he advised.
Dr. Wells reported serving as a member of an advisory board and/or speakers bureau for more than a dozen pharmaceutical companies.
MAUI, HAWAII – When a rheumatologist gets a call from an ophthalmologist regarding a patient with an inflamed eye and elevated intraocular pressure unresponsive to the eye specialist’s customary array of topical, systemic, and intraocular implanted corticosteroids, that’s a patient who needs to be seen immediately, Alvin F. Wells, MD, PhD, said at the 2020 Rheumatology Winter Clinical Symposium.
Elevated intraocular pressure due to uveitis or scleritis can result in blindness. Eye specialists call upon rheumatologists here because of their expertise in step-up therapy with methotrexate and other traditional oral disease-modifying antirheumatic drugs as well as biologic agents.
“Here’s my treatment approach to inflammatory eye disease: We’re pulling out all the guns,” declared Dr. Wells, a rheumatologist with a special interest in eye disease. He is director of the Rheumatology and Immunotherapy Center in Franklin, Wisc., with academic appointments to the Karolinska Institute in Stockholm, Duke University, and Marquette University.
Uveitis involves inflammation of the iris, choroid, and ciliary body. A straightforward case of noninfectious anterior uveitis will typically respond to 2 weeks of topical steroid drops, or sometimes even topical NSAID drops.
However, noninfectious posterior, intermediate, or panuveitis is another matter. In those circumstances, he gives the patient 125 mg of methylprednisolone by intramuscular injection and a 20-mg dose of oral methotrexate at that first clinic visit. The patient is sent home with a prescription for oral prednisone, tapering over 2-3 weeks, and another for methotrexate at 15-25 mg/week plus 1-2 mg/day of folic acid. Dr. Wells also gives consideration to add-on azathioprine or mycophenolate mofetil. He views multidrug therapy as having a sound rationale because multiple inflammatory pathways are involved in noninfectious uveitis.
“Ophthalmologists like to push for cyclophosphamide, but there’s no controlled data out there showing it’s effective in inflammatory eye disorders. It’s a pretty toxic regimen, and when you think about all the complications we see in using this drug to treat patients with lupus, I’d rather hold it in reserve for severe cases where we can go to it if we need to,” the rheumatologist explained.
He conducted a literature review to rank rheumatologic medications in terms of their evidence base for treatment of inflammatory ocular disorders. Among oral agents, at the top of the heap is methotrexate, whose efficacy for both noninfectious uveitis and scleritis is supported by multiple randomized, controlled studies. But mycophenolate mofetil is a reasonable alternative first-line corticosteroid-sparing agent, as demonstrated in the 265-patient multicenter FAST (First-line Antimetabolites as Steroid-sparing Treatment) trial sponsored by the National Eye Institute. That trial demonstrated no significant difference in treatment success at 6 months between methotrexate and mycophenolate mofetil.
Oral apremilast (Otezla) is approved for treatment of the oral ulcers of Behçet’s disease, but not for Behçet’s eye disease, where the experience is anecdotal.
Dr. Wells is quick to turn to adalimumab (Humira) when he deems a biologic to be warranted; indeed, it’s the only biologic approved for noninfectious uveitis. Of course, not everyone is a responder.
“Can we extrapolate that high-quality evidence of benefit for adalimumab to other drugs? Probably yes, and if you did that it would be for the IgG monoclonal antibodies that can cross the blood/aqueous barrier,” he said.
Infliximab (Remicade) is the biologic with the second-strongest supporting evidence in noninfectious uveitis. For the uveitis of Behçet’s disease, one of the most common rheumatic causes of inflammatory eye disease, Spanish investigators who conducted a nationwide nonrandomized study reported that both adalimumab and infliximab were effective, although adalimumab had superior outcomes at 1 year.
Uveitis is the most common extra-articular expression of axial spondyloarthritis (axSpA). In the open-label extension of the randomized RAPID-axSpA trial, patients randomized to certolizumab pegol (Cimzia) had a significantly lower incidence of uveitis flares than with placebo through 204 weeks of follow-up.
“The take-home message is we have some post hoc data here to say, ‘Hey, this could work in those patients who have inflammatory eye diseases in the setting of axSpA,’ ” Dr. Wells said.
The interleukin-6 receptor inhibitor tocilizumab (Actemra) “definitely works” for noninfectious uveitis, according to Dr. Wells, pointing to the positive results of the multicenter U.S. STOP-Uveitis study.
“The caveat here is tocilizumab has only been studied in the IV formulation. It’s too bad they didn’t use the [subcutaneous formulation]; you can’t get IV tocilizumab approved by payers in the U.S.,” according to the rheumatologist.
Based upon positive anecdotal case reports, Dr. Wells has a few patients on rituximab (Rituxan) for uveitis, with favorable results. The same for abatacept (Orencia).
It’s imperative that a patient on a biologic for uveitis undergo weekly ophthalmologic examinations. Only after the intraocular pressure is normal and inflammatory cells in the anterior chamber have waned is it appropriate to discontinue the biologic and slowly taper the methotrexate and any other oral disease-modifying antirheumatic drugs. Some experts argue for lifelong therapy in patients who’ve experienced uveitis. Dr. Wells disagrees, preferring to treat acute uveitis flares as they arise, although if underlying disease such as psoriatic arthritis or axSpA is present, some form of background therapy will probably be necessary.
Get to know teprotumumab
Rheumatologists who operate an infusion center are likely to increasingly be called upon by endocrinologists and ophthalmologists to administer intravenous teprotumumab-trbw (Tepezza), a human monoclonal antibody directed against the insulin-like growth factor 1 receptor that was approved earlier this year by the Food and Drug Administration as the first-ever drug for thyroid eye disease, a disfiguring and potentially blinding condition.
“This is really exciting,” Dr. Wells said. “The disease has an acute inflammatory stage, and that’s when you’ll be called on to give this drug. It makes a dramatic difference. Once a patient gets to the scarring phase there’s not a whole lot they can do other than surgery.”
In the pivotal phase 3 randomized trial, 83% of the teprotumumab group achieved the primary endpoint, a reduction in proptosis, or eye bulging, of at least 2 mm at week 24, compared with 10% of placebo-treated controls. The number needed to treat was 1.4. The chief side effects were muscle spasms, hair loss, fatigue, and nausea.
“You might say, ‘two millimeters, that’s nothing.’ But the primary drug used before teprotumumab was IV steroids, and there a 0.6-mm reduction in proptosis was considered improvement,” Dr. Wells observed.
Obtaining payer approval
“I’ve found over the last 10 years that when it comes to eye disease, insurance companies have a little more wiggle room,” he said. “They’re not going to let somebody go blind. You can get the references I’ve mentioned and show them the data. After all, we only have one biologic drug that’s been approved, and not everybody responds to it.
“Titrate your therapy based upon the intraocular pressure, the number of inflammatory cells in the anterior chamber, and any visual changes. You’ve got to be very aggressive with therapy, and don’t take no for an answer from the insurance companies,” he advised.
Dr. Wells reported serving as a member of an advisory board and/or speakers bureau for more than a dozen pharmaceutical companies.
MAUI, HAWAII – When a rheumatologist gets a call from an ophthalmologist regarding a patient with an inflamed eye and elevated intraocular pressure unresponsive to the eye specialist’s customary array of topical, systemic, and intraocular implanted corticosteroids, that’s a patient who needs to be seen immediately, Alvin F. Wells, MD, PhD, said at the 2020 Rheumatology Winter Clinical Symposium.
Elevated intraocular pressure due to uveitis or scleritis can result in blindness. Eye specialists call upon rheumatologists here because of their expertise in step-up therapy with methotrexate and other traditional oral disease-modifying antirheumatic drugs as well as biologic agents.
“Here’s my treatment approach to inflammatory eye disease: We’re pulling out all the guns,” declared Dr. Wells, a rheumatologist with a special interest in eye disease. He is director of the Rheumatology and Immunotherapy Center in Franklin, Wisc., with academic appointments to the Karolinska Institute in Stockholm, Duke University, and Marquette University.
Uveitis involves inflammation of the iris, choroid, and ciliary body. A straightforward case of noninfectious anterior uveitis will typically respond to 2 weeks of topical steroid drops, or sometimes even topical NSAID drops.
However, noninfectious posterior, intermediate, or panuveitis is another matter. In those circumstances, he gives the patient 125 mg of methylprednisolone by intramuscular injection and a 20-mg dose of oral methotrexate at that first clinic visit. The patient is sent home with a prescription for oral prednisone, tapering over 2-3 weeks, and another for methotrexate at 15-25 mg/week plus 1-2 mg/day of folic acid. Dr. Wells also gives consideration to add-on azathioprine or mycophenolate mofetil. He views multidrug therapy as having a sound rationale because multiple inflammatory pathways are involved in noninfectious uveitis.
“Ophthalmologists like to push for cyclophosphamide, but there’s no controlled data out there showing it’s effective in inflammatory eye disorders. It’s a pretty toxic regimen, and when you think about all the complications we see in using this drug to treat patients with lupus, I’d rather hold it in reserve for severe cases where we can go to it if we need to,” the rheumatologist explained.
He conducted a literature review to rank rheumatologic medications in terms of their evidence base for treatment of inflammatory ocular disorders. Among oral agents, at the top of the heap is methotrexate, whose efficacy for both noninfectious uveitis and scleritis is supported by multiple randomized, controlled studies. But mycophenolate mofetil is a reasonable alternative first-line corticosteroid-sparing agent, as demonstrated in the 265-patient multicenter FAST (First-line Antimetabolites as Steroid-sparing Treatment) trial sponsored by the National Eye Institute. That trial demonstrated no significant difference in treatment success at 6 months between methotrexate and mycophenolate mofetil.
Oral apremilast (Otezla) is approved for treatment of the oral ulcers of Behçet’s disease, but not for Behçet’s eye disease, where the experience is anecdotal.
Dr. Wells is quick to turn to adalimumab (Humira) when he deems a biologic to be warranted; indeed, it’s the only biologic approved for noninfectious uveitis. Of course, not everyone is a responder.
“Can we extrapolate that high-quality evidence of benefit for adalimumab to other drugs? Probably yes, and if you did that it would be for the IgG monoclonal antibodies that can cross the blood/aqueous barrier,” he said.
Infliximab (Remicade) is the biologic with the second-strongest supporting evidence in noninfectious uveitis. For the uveitis of Behçet’s disease, one of the most common rheumatic causes of inflammatory eye disease, Spanish investigators who conducted a nationwide nonrandomized study reported that both adalimumab and infliximab were effective, although adalimumab had superior outcomes at 1 year.
Uveitis is the most common extra-articular expression of axial spondyloarthritis (axSpA). In the open-label extension of the randomized RAPID-axSpA trial, patients randomized to certolizumab pegol (Cimzia) had a significantly lower incidence of uveitis flares than with placebo through 204 weeks of follow-up.
“The take-home message is we have some post hoc data here to say, ‘Hey, this could work in those patients who have inflammatory eye diseases in the setting of axSpA,’ ” Dr. Wells said.
The interleukin-6 receptor inhibitor tocilizumab (Actemra) “definitely works” for noninfectious uveitis, according to Dr. Wells, pointing to the positive results of the multicenter U.S. STOP-Uveitis study.
“The caveat here is tocilizumab has only been studied in the IV formulation. It’s too bad they didn’t use the [subcutaneous formulation]; you can’t get IV tocilizumab approved by payers in the U.S.,” according to the rheumatologist.
Based upon positive anecdotal case reports, Dr. Wells has a few patients on rituximab (Rituxan) for uveitis, with favorable results. The same for abatacept (Orencia).
It’s imperative that a patient on a biologic for uveitis undergo weekly ophthalmologic examinations. Only after the intraocular pressure is normal and inflammatory cells in the anterior chamber have waned is it appropriate to discontinue the biologic and slowly taper the methotrexate and any other oral disease-modifying antirheumatic drugs. Some experts argue for lifelong therapy in patients who’ve experienced uveitis. Dr. Wells disagrees, preferring to treat acute uveitis flares as they arise, although if underlying disease such as psoriatic arthritis or axSpA is present, some form of background therapy will probably be necessary.
Get to know teprotumumab
Rheumatologists who operate an infusion center are likely to increasingly be called upon by endocrinologists and ophthalmologists to administer intravenous teprotumumab-trbw (Tepezza), a human monoclonal antibody directed against the insulin-like growth factor 1 receptor that was approved earlier this year by the Food and Drug Administration as the first-ever drug for thyroid eye disease, a disfiguring and potentially blinding condition.
“This is really exciting,” Dr. Wells said. “The disease has an acute inflammatory stage, and that’s when you’ll be called on to give this drug. It makes a dramatic difference. Once a patient gets to the scarring phase there’s not a whole lot they can do other than surgery.”
In the pivotal phase 3 randomized trial, 83% of the teprotumumab group achieved the primary endpoint, a reduction in proptosis, or eye bulging, of at least 2 mm at week 24, compared with 10% of placebo-treated controls. The number needed to treat was 1.4. The chief side effects were muscle spasms, hair loss, fatigue, and nausea.
“You might say, ‘two millimeters, that’s nothing.’ But the primary drug used before teprotumumab was IV steroids, and there a 0.6-mm reduction in proptosis was considered improvement,” Dr. Wells observed.
Obtaining payer approval
“I’ve found over the last 10 years that when it comes to eye disease, insurance companies have a little more wiggle room,” he said. “They’re not going to let somebody go blind. You can get the references I’ve mentioned and show them the data. After all, we only have one biologic drug that’s been approved, and not everybody responds to it.
“Titrate your therapy based upon the intraocular pressure, the number of inflammatory cells in the anterior chamber, and any visual changes. You’ve got to be very aggressive with therapy, and don’t take no for an answer from the insurance companies,” he advised.
Dr. Wells reported serving as a member of an advisory board and/or speakers bureau for more than a dozen pharmaceutical companies.
REPORTING FROM RWCS 2020
COVID-19: A guide to making telepsychiatry work
Changes prompted by social distancing could last beyond the pandemic
As the coronavirus pandemic persists, insurers and the federal government are making it easier for mental health professionals to deliver safe and effective psychiatric services to patients via Zoom, FaceTime, and other conferencing tools. Many psychiatrists, meanwhile, are embracing telepsychiatry for the first time – in some cases with urgency.
Jay H. Shore, MD, MPH, said in an interview that mental health providers at his medical center have gone entirely virtual in recent weeks.
“The genie is out of the bottle on this,” said Dr. Shore, director of telemedicine at the Helen and Arthur E. Johnson Depression Center and director of telemedicine programming for the department of psychiatry at the University of Colorado at Denver, Aurora. He thinks this is the beginning of a new era that will last beyond the pandemic. “There’s going to be a much wider and diffuse acceptance of telemedicine as we go forward,” he added.
Dr. Shore and several colleagues from across the country offered several tips about factors to consider while learning to use telepsychiatry as a treatment tool.
To start, Dr. Shore advised reviewing the American Psychiatric Association’s Telepsychiatry Practice Guidelines and its Telepsychiatry Toolkit, which include dozens of brief videos about topics such as room lighting and managing the content process.
Another resource is the joint APA–American Academy of Child and Adolescent Psychiatry Telepsychiatry Toolkit, said Shabana Khan, MD, an assistant professor and director of telemedicine for the department of child and adolescent psychiatry at New York University Langone Health.
One of the challenges is managing emergencies long distance. If a patient experiences a mental health emergency in a psychiatrist’s office, the clinician can call 911 or direct staff to seek help. “When they’re at their house,” said Dr. Shore, “it’s a little different.”
Staff members are not present at home offices, for example, and the patient might live in a different city and therefore have a different 911 system. “It’s important to know your protocol about how you plan to handle these emergencies before you start working with the patient,” Dr. Shore said.
Another tip is to ask staff to perform a test session to work out the technical kinks before the first patient appointment. “They can make the connection and make sure there’s a video signal with adequate quality,” Dr. Shore said. Failing to conduct a test run can lead to spending several minutes of a session trying to help patients figure out how to make video conferencing work properly.
“You can spend a lot of time acting as IT support,” he said.
It is important to ensure that virtual visits are not interrupted by technical glitches, Daniel Bristow, MD, said in an interview. If possible, hardwire your laptop or computer to an ethernet cable, said Dr. Bristow, president of the Oregon Psychiatric Physicians Association, the state’s branch of the APA. “This will lead to fewer fluctuations that you could see by using wifi,” said Dr. Bristow, who practices in Portland.
“Initially, I assumed that those with psychotic symptoms might struggle more. But I have been surprised at how well some patients have done,” said Andrew J. McLean, MD, MPH, clinical professor and chair of the department of psychiatry and behavioral science at the University of North Dakota, Grand Forks.
However, it might help to provide additional coaching to those patients, said Dr. Bristow. He offers a warning to these patients: “If you feel like you’re getting messages over the TV, my talking to you may make you feel worse.” However, “in every case, the patient was able to say, ‘I know you’re real.’ One patient even said: ‘I’ve heard these voices from my TV for years. But I know you’re a doctor, and you’re in an office trying to help me.’ ”
Dr. Shore thinks that video meetings have the potential to help psychiatrists and patients form better personal connections than in-person meetings. Patients with anxiety or PTSD, for example, “may feel safer since they’re in their own space, and they have a greater sense of control over the session than being in somebody’s office,” he said.
Dr. Khan agreed. “Some children, such as those with a significant trauma history or with significant anxiety, may feel more comfortable with this modality and may open up more during video sessions,” she said. In addition, “the distance that telepsychiatry provides may also enhance feelings of confidentiality and reduce potential stigma that may be associated with seeking mental health care.”
When it comes to using videoconferencing to treat children, take advantage of interactive features that are available, said Katherine Nguyen Williams, PhD. Zoom’s HIPAA-compliant health care software, for example, offers a “share screen” capability. “It allows for easy interactive activities,” said Dr. Nguyen Williams, director of strategic development and clinical innovation at Rady Children’s Hospital’s department of psychiatry at the University of California, San Diego. “Clinicians can play tic-tac-toe on the screen with the young patients, and they can work on cognitive-behavioral therapy worksheets together on the digital screen. Clinicians can even show a mindfulness video to the patient while actively coaching and giving feedback to the patient as they practice diaphragmatic breathing while viewing the video.
“There are so many more options for making virtual therapy as interactive as face-to-face therapy,” said Dr. Nguyen Williams, who also is an associate clinical professor at the university. “This is the key to getting and keeping the patient engaged in telepsychiatry.”
Despite the many positive aspects of using telepsychiatry as a treatment tool, some negative factors must be considered. “You lose some of the nuances, subtleties in terms of expression, movement, smell, etc.,” said Dr. McLean. “Also, there are rare instances where a part of a physical examination would be appropriate, which also is precluded.”
Videoconferencing software might allow the clinician to zoom in to take a closer look at a patient to look for subtle movements and tremors, Dr. McLean said. And, he added, he has asked nursing staff to check for particular signs and symptoms during visits and to describe them to him. “Still,” Dr. McLean said, “this does not take the place of being there.”
Dr. Shore suggested several other practical considerations. For example, while on a screen, keep the home environment as professional as the office would be, he said. Be clear with family members about the importance of not interrupting and make sure that privacy is maintained. The message should be: “I’m working from home, and I’m not available during these hours,” Dr. Shore said. “You need to be aware that, during this time, I need this for clinical work.”
Dr. Shore reported serving as chief medical officer of AccessCare Services, and receiving royalties from American Psychiatric Association Publishing and Springer. He also is coauthor with Peter Yellowlees, MD, of “Telepsychiatry and Health Technologies: A Guide for Mental Health Professionals” (Arlington, Va.: American Psychiatric Association Publishing, 2018). Dr. Khan and Dr. McLean reported no relevant disclosures. Dr. Bristow reported relationships with MCG Health and Insight + Regroup Telehealth.
For more details about using telepsychiatry in the time of COVID-19, listen to the April 8 Psychcast Masterclass lecture by Dr. Shore.
Changes prompted by social distancing could last beyond the pandemic
Changes prompted by social distancing could last beyond the pandemic
As the coronavirus pandemic persists, insurers and the federal government are making it easier for mental health professionals to deliver safe and effective psychiatric services to patients via Zoom, FaceTime, and other conferencing tools. Many psychiatrists, meanwhile, are embracing telepsychiatry for the first time – in some cases with urgency.
Jay H. Shore, MD, MPH, said in an interview that mental health providers at his medical center have gone entirely virtual in recent weeks.
“The genie is out of the bottle on this,” said Dr. Shore, director of telemedicine at the Helen and Arthur E. Johnson Depression Center and director of telemedicine programming for the department of psychiatry at the University of Colorado at Denver, Aurora. He thinks this is the beginning of a new era that will last beyond the pandemic. “There’s going to be a much wider and diffuse acceptance of telemedicine as we go forward,” he added.
Dr. Shore and several colleagues from across the country offered several tips about factors to consider while learning to use telepsychiatry as a treatment tool.
To start, Dr. Shore advised reviewing the American Psychiatric Association’s Telepsychiatry Practice Guidelines and its Telepsychiatry Toolkit, which include dozens of brief videos about topics such as room lighting and managing the content process.
Another resource is the joint APA–American Academy of Child and Adolescent Psychiatry Telepsychiatry Toolkit, said Shabana Khan, MD, an assistant professor and director of telemedicine for the department of child and adolescent psychiatry at New York University Langone Health.
One of the challenges is managing emergencies long distance. If a patient experiences a mental health emergency in a psychiatrist’s office, the clinician can call 911 or direct staff to seek help. “When they’re at their house,” said Dr. Shore, “it’s a little different.”
Staff members are not present at home offices, for example, and the patient might live in a different city and therefore have a different 911 system. “It’s important to know your protocol about how you plan to handle these emergencies before you start working with the patient,” Dr. Shore said.
Another tip is to ask staff to perform a test session to work out the technical kinks before the first patient appointment. “They can make the connection and make sure there’s a video signal with adequate quality,” Dr. Shore said. Failing to conduct a test run can lead to spending several minutes of a session trying to help patients figure out how to make video conferencing work properly.
“You can spend a lot of time acting as IT support,” he said.
It is important to ensure that virtual visits are not interrupted by technical glitches, Daniel Bristow, MD, said in an interview. If possible, hardwire your laptop or computer to an ethernet cable, said Dr. Bristow, president of the Oregon Psychiatric Physicians Association, the state’s branch of the APA. “This will lead to fewer fluctuations that you could see by using wifi,” said Dr. Bristow, who practices in Portland.
“Initially, I assumed that those with psychotic symptoms might struggle more. But I have been surprised at how well some patients have done,” said Andrew J. McLean, MD, MPH, clinical professor and chair of the department of psychiatry and behavioral science at the University of North Dakota, Grand Forks.
However, it might help to provide additional coaching to those patients, said Dr. Bristow. He offers a warning to these patients: “If you feel like you’re getting messages over the TV, my talking to you may make you feel worse.” However, “in every case, the patient was able to say, ‘I know you’re real.’ One patient even said: ‘I’ve heard these voices from my TV for years. But I know you’re a doctor, and you’re in an office trying to help me.’ ”
Dr. Shore thinks that video meetings have the potential to help psychiatrists and patients form better personal connections than in-person meetings. Patients with anxiety or PTSD, for example, “may feel safer since they’re in their own space, and they have a greater sense of control over the session than being in somebody’s office,” he said.
Dr. Khan agreed. “Some children, such as those with a significant trauma history or with significant anxiety, may feel more comfortable with this modality and may open up more during video sessions,” she said. In addition, “the distance that telepsychiatry provides may also enhance feelings of confidentiality and reduce potential stigma that may be associated with seeking mental health care.”
When it comes to using videoconferencing to treat children, take advantage of interactive features that are available, said Katherine Nguyen Williams, PhD. Zoom’s HIPAA-compliant health care software, for example, offers a “share screen” capability. “It allows for easy interactive activities,” said Dr. Nguyen Williams, director of strategic development and clinical innovation at Rady Children’s Hospital’s department of psychiatry at the University of California, San Diego. “Clinicians can play tic-tac-toe on the screen with the young patients, and they can work on cognitive-behavioral therapy worksheets together on the digital screen. Clinicians can even show a mindfulness video to the patient while actively coaching and giving feedback to the patient as they practice diaphragmatic breathing while viewing the video.
“There are so many more options for making virtual therapy as interactive as face-to-face therapy,” said Dr. Nguyen Williams, who also is an associate clinical professor at the university. “This is the key to getting and keeping the patient engaged in telepsychiatry.”
Despite the many positive aspects of using telepsychiatry as a treatment tool, some negative factors must be considered. “You lose some of the nuances, subtleties in terms of expression, movement, smell, etc.,” said Dr. McLean. “Also, there are rare instances where a part of a physical examination would be appropriate, which also is precluded.”
Videoconferencing software might allow the clinician to zoom in to take a closer look at a patient to look for subtle movements and tremors, Dr. McLean said. And, he added, he has asked nursing staff to check for particular signs and symptoms during visits and to describe them to him. “Still,” Dr. McLean said, “this does not take the place of being there.”
Dr. Shore suggested several other practical considerations. For example, while on a screen, keep the home environment as professional as the office would be, he said. Be clear with family members about the importance of not interrupting and make sure that privacy is maintained. The message should be: “I’m working from home, and I’m not available during these hours,” Dr. Shore said. “You need to be aware that, during this time, I need this for clinical work.”
Dr. Shore reported serving as chief medical officer of AccessCare Services, and receiving royalties from American Psychiatric Association Publishing and Springer. He also is coauthor with Peter Yellowlees, MD, of “Telepsychiatry and Health Technologies: A Guide for Mental Health Professionals” (Arlington, Va.: American Psychiatric Association Publishing, 2018). Dr. Khan and Dr. McLean reported no relevant disclosures. Dr. Bristow reported relationships with MCG Health and Insight + Regroup Telehealth.
For more details about using telepsychiatry in the time of COVID-19, listen to the April 8 Psychcast Masterclass lecture by Dr. Shore.
As the coronavirus pandemic persists, insurers and the federal government are making it easier for mental health professionals to deliver safe and effective psychiatric services to patients via Zoom, FaceTime, and other conferencing tools. Many psychiatrists, meanwhile, are embracing telepsychiatry for the first time – in some cases with urgency.
Jay H. Shore, MD, MPH, said in an interview that mental health providers at his medical center have gone entirely virtual in recent weeks.
“The genie is out of the bottle on this,” said Dr. Shore, director of telemedicine at the Helen and Arthur E. Johnson Depression Center and director of telemedicine programming for the department of psychiatry at the University of Colorado at Denver, Aurora. He thinks this is the beginning of a new era that will last beyond the pandemic. “There’s going to be a much wider and diffuse acceptance of telemedicine as we go forward,” he added.
Dr. Shore and several colleagues from across the country offered several tips about factors to consider while learning to use telepsychiatry as a treatment tool.
To start, Dr. Shore advised reviewing the American Psychiatric Association’s Telepsychiatry Practice Guidelines and its Telepsychiatry Toolkit, which include dozens of brief videos about topics such as room lighting and managing the content process.
Another resource is the joint APA–American Academy of Child and Adolescent Psychiatry Telepsychiatry Toolkit, said Shabana Khan, MD, an assistant professor and director of telemedicine for the department of child and adolescent psychiatry at New York University Langone Health.
One of the challenges is managing emergencies long distance. If a patient experiences a mental health emergency in a psychiatrist’s office, the clinician can call 911 or direct staff to seek help. “When they’re at their house,” said Dr. Shore, “it’s a little different.”
Staff members are not present at home offices, for example, and the patient might live in a different city and therefore have a different 911 system. “It’s important to know your protocol about how you plan to handle these emergencies before you start working with the patient,” Dr. Shore said.
Another tip is to ask staff to perform a test session to work out the technical kinks before the first patient appointment. “They can make the connection and make sure there’s a video signal with adequate quality,” Dr. Shore said. Failing to conduct a test run can lead to spending several minutes of a session trying to help patients figure out how to make video conferencing work properly.
“You can spend a lot of time acting as IT support,” he said.
It is important to ensure that virtual visits are not interrupted by technical glitches, Daniel Bristow, MD, said in an interview. If possible, hardwire your laptop or computer to an ethernet cable, said Dr. Bristow, president of the Oregon Psychiatric Physicians Association, the state’s branch of the APA. “This will lead to fewer fluctuations that you could see by using wifi,” said Dr. Bristow, who practices in Portland.
“Initially, I assumed that those with psychotic symptoms might struggle more. But I have been surprised at how well some patients have done,” said Andrew J. McLean, MD, MPH, clinical professor and chair of the department of psychiatry and behavioral science at the University of North Dakota, Grand Forks.
However, it might help to provide additional coaching to those patients, said Dr. Bristow. He offers a warning to these patients: “If you feel like you’re getting messages over the TV, my talking to you may make you feel worse.” However, “in every case, the patient was able to say, ‘I know you’re real.’ One patient even said: ‘I’ve heard these voices from my TV for years. But I know you’re a doctor, and you’re in an office trying to help me.’ ”
Dr. Shore thinks that video meetings have the potential to help psychiatrists and patients form better personal connections than in-person meetings. Patients with anxiety or PTSD, for example, “may feel safer since they’re in their own space, and they have a greater sense of control over the session than being in somebody’s office,” he said.
Dr. Khan agreed. “Some children, such as those with a significant trauma history or with significant anxiety, may feel more comfortable with this modality and may open up more during video sessions,” she said. In addition, “the distance that telepsychiatry provides may also enhance feelings of confidentiality and reduce potential stigma that may be associated with seeking mental health care.”
When it comes to using videoconferencing to treat children, take advantage of interactive features that are available, said Katherine Nguyen Williams, PhD. Zoom’s HIPAA-compliant health care software, for example, offers a “share screen” capability. “It allows for easy interactive activities,” said Dr. Nguyen Williams, director of strategic development and clinical innovation at Rady Children’s Hospital’s department of psychiatry at the University of California, San Diego. “Clinicians can play tic-tac-toe on the screen with the young patients, and they can work on cognitive-behavioral therapy worksheets together on the digital screen. Clinicians can even show a mindfulness video to the patient while actively coaching and giving feedback to the patient as they practice diaphragmatic breathing while viewing the video.
“There are so many more options for making virtual therapy as interactive as face-to-face therapy,” said Dr. Nguyen Williams, who also is an associate clinical professor at the university. “This is the key to getting and keeping the patient engaged in telepsychiatry.”
Despite the many positive aspects of using telepsychiatry as a treatment tool, some negative factors must be considered. “You lose some of the nuances, subtleties in terms of expression, movement, smell, etc.,” said Dr. McLean. “Also, there are rare instances where a part of a physical examination would be appropriate, which also is precluded.”
Videoconferencing software might allow the clinician to zoom in to take a closer look at a patient to look for subtle movements and tremors, Dr. McLean said. And, he added, he has asked nursing staff to check for particular signs and symptoms during visits and to describe them to him. “Still,” Dr. McLean said, “this does not take the place of being there.”
Dr. Shore suggested several other practical considerations. For example, while on a screen, keep the home environment as professional as the office would be, he said. Be clear with family members about the importance of not interrupting and make sure that privacy is maintained. The message should be: “I’m working from home, and I’m not available during these hours,” Dr. Shore said. “You need to be aware that, during this time, I need this for clinical work.”
Dr. Shore reported serving as chief medical officer of AccessCare Services, and receiving royalties from American Psychiatric Association Publishing and Springer. He also is coauthor with Peter Yellowlees, MD, of “Telepsychiatry and Health Technologies: A Guide for Mental Health Professionals” (Arlington, Va.: American Psychiatric Association Publishing, 2018). Dr. Khan and Dr. McLean reported no relevant disclosures. Dr. Bristow reported relationships with MCG Health and Insight + Regroup Telehealth.
For more details about using telepsychiatry in the time of COVID-19, listen to the April 8 Psychcast Masterclass lecture by Dr. Shore.
Combo elicits responses in advanced cervical cancer
regardless of programmed death–ligand 1 (PD-L1) expression, according to preliminary findings from a phase 2 study.
Apatinib, an inhibitor of vascular endothelial growth factor receptor-2, and camrelizumab, an anti-PD-1 monoclonal antibody, produced an objective response rate of 60% in evaluable patients.
Chunyan Lan, MD, of Sun Yat-sen University Cancer Center in Guangzhou, China, and colleagues reported these results in an abstract that had been slated for presentation at the Society of Gynecologic Oncology’s Annual Meeting on Women’s Cancer. The meeting was canceled because of the COVID-19 pandemic. The following data differ somewhat from the abstract.
Dr. Lan and colleagues reported results in 45 patients who had progressed after at least one line of systemic chemotherapy for metastatic, recurrent, or persistent cervical cancer, and had measurable disease. Patients had a median age of 51 years and an Eastern Cooperative Oncology Group performance status of 0-1. They were enrolled at four centers in China between Jan. 21 and Aug. 1, 2019.
Treatment consisted of oral apatinib at a dose of 250 mg once daily and intravenous camrelizumab at a dose of 200 mg every 2 weeks until disease progression, unacceptable toxicity, or consent withdrawal.
As of Jan. 22, 2020, 25 of 42 efficacy-evaluable patients had achieved a response. Two patients had a complete response, 23 had a partial response, and 12 had stable disease.
“We saw responses in patients regardless of PD-L1 expression,” Dr. Lan said. “In our study, 34% were PD-L1 negative, and the response rate is 65% in PD-L1-positive and 50% in PD-L1-negative [patients].”
The median duration of response was not reached, she added.
The median follow-up was 9.2 months, with the last patient enrolled having 6 months of follow-up. At the data cutoff, 19 patients had disease progression, and 8 had died of their disease.
The median overall survival also was not reached, Dr. Lan said, but overall survival at 9 months was 80%. The median progression-free survival was 7.6 months, and the 6-month progression-free survival rate was 58%.
Grade 3 or greater treatment-related adverse events occurred in 68.9% of patients; adverse events occurring in at least 5% of patients included hypertension (22.2%), fatigue (15.6%), anemia (13.3%), and thrombocytopenia (6.7%).
“Nineteen patients were still on treatment at the data cutoff date, and 26 patients discontinued the study,” Dr. Lan said. “The most common reason to discontinue was disease progression, and three patients discontinued the study due to adverse events.”
“These preliminary results are very encouraging,” Dr. Lan said. “As we know, pembrolizumab is approved as the second-line therapy in recurrent cervical cancer [in] PD-L1-positive patients, and the objective response rate with pembrolizumab monotherapy for recurrent cervical cancer is only 17%, as reported in KEYNOTE-028 [J Clin Oncol. 2017 Dec 20;35(36):4035-41].”
Furthermore, apatinib monotherapy has been studied with only modest results.
“But in our study, this combination is really effective in recurrent cervical cancer, and we see a very durable response,” she said, again emphasizing that those responses occurred regardless of PD-L1 expression. “So this is important. ... We think our findings expand the opportunity of patients with recurrent cervical cancer to receive immune therapy.”
Study participants will be followed for 2 years, Dr. Lan noted.
She reported having no disclosures. The study is sponsored by Sun Yat-sen University.
[email protected]
SOURCE: Lan C et al. SGO 2020, Abstract 55.
regardless of programmed death–ligand 1 (PD-L1) expression, according to preliminary findings from a phase 2 study.
Apatinib, an inhibitor of vascular endothelial growth factor receptor-2, and camrelizumab, an anti-PD-1 monoclonal antibody, produced an objective response rate of 60% in evaluable patients.
Chunyan Lan, MD, of Sun Yat-sen University Cancer Center in Guangzhou, China, and colleagues reported these results in an abstract that had been slated for presentation at the Society of Gynecologic Oncology’s Annual Meeting on Women’s Cancer. The meeting was canceled because of the COVID-19 pandemic. The following data differ somewhat from the abstract.
Dr. Lan and colleagues reported results in 45 patients who had progressed after at least one line of systemic chemotherapy for metastatic, recurrent, or persistent cervical cancer, and had measurable disease. Patients had a median age of 51 years and an Eastern Cooperative Oncology Group performance status of 0-1. They were enrolled at four centers in China between Jan. 21 and Aug. 1, 2019.
Treatment consisted of oral apatinib at a dose of 250 mg once daily and intravenous camrelizumab at a dose of 200 mg every 2 weeks until disease progression, unacceptable toxicity, or consent withdrawal.
As of Jan. 22, 2020, 25 of 42 efficacy-evaluable patients had achieved a response. Two patients had a complete response, 23 had a partial response, and 12 had stable disease.
“We saw responses in patients regardless of PD-L1 expression,” Dr. Lan said. “In our study, 34% were PD-L1 negative, and the response rate is 65% in PD-L1-positive and 50% in PD-L1-negative [patients].”
The median duration of response was not reached, she added.
The median follow-up was 9.2 months, with the last patient enrolled having 6 months of follow-up. At the data cutoff, 19 patients had disease progression, and 8 had died of their disease.
The median overall survival also was not reached, Dr. Lan said, but overall survival at 9 months was 80%. The median progression-free survival was 7.6 months, and the 6-month progression-free survival rate was 58%.
Grade 3 or greater treatment-related adverse events occurred in 68.9% of patients; adverse events occurring in at least 5% of patients included hypertension (22.2%), fatigue (15.6%), anemia (13.3%), and thrombocytopenia (6.7%).
“Nineteen patients were still on treatment at the data cutoff date, and 26 patients discontinued the study,” Dr. Lan said. “The most common reason to discontinue was disease progression, and three patients discontinued the study due to adverse events.”
“These preliminary results are very encouraging,” Dr. Lan said. “As we know, pembrolizumab is approved as the second-line therapy in recurrent cervical cancer [in] PD-L1-positive patients, and the objective response rate with pembrolizumab monotherapy for recurrent cervical cancer is only 17%, as reported in KEYNOTE-028 [J Clin Oncol. 2017 Dec 20;35(36):4035-41].”
Furthermore, apatinib monotherapy has been studied with only modest results.
“But in our study, this combination is really effective in recurrent cervical cancer, and we see a very durable response,” she said, again emphasizing that those responses occurred regardless of PD-L1 expression. “So this is important. ... We think our findings expand the opportunity of patients with recurrent cervical cancer to receive immune therapy.”
Study participants will be followed for 2 years, Dr. Lan noted.
She reported having no disclosures. The study is sponsored by Sun Yat-sen University.
[email protected]
SOURCE: Lan C et al. SGO 2020, Abstract 55.
regardless of programmed death–ligand 1 (PD-L1) expression, according to preliminary findings from a phase 2 study.
Apatinib, an inhibitor of vascular endothelial growth factor receptor-2, and camrelizumab, an anti-PD-1 monoclonal antibody, produced an objective response rate of 60% in evaluable patients.
Chunyan Lan, MD, of Sun Yat-sen University Cancer Center in Guangzhou, China, and colleagues reported these results in an abstract that had been slated for presentation at the Society of Gynecologic Oncology’s Annual Meeting on Women’s Cancer. The meeting was canceled because of the COVID-19 pandemic. The following data differ somewhat from the abstract.
Dr. Lan and colleagues reported results in 45 patients who had progressed after at least one line of systemic chemotherapy for metastatic, recurrent, or persistent cervical cancer, and had measurable disease. Patients had a median age of 51 years and an Eastern Cooperative Oncology Group performance status of 0-1. They were enrolled at four centers in China between Jan. 21 and Aug. 1, 2019.
Treatment consisted of oral apatinib at a dose of 250 mg once daily and intravenous camrelizumab at a dose of 200 mg every 2 weeks until disease progression, unacceptable toxicity, or consent withdrawal.
As of Jan. 22, 2020, 25 of 42 efficacy-evaluable patients had achieved a response. Two patients had a complete response, 23 had a partial response, and 12 had stable disease.
“We saw responses in patients regardless of PD-L1 expression,” Dr. Lan said. “In our study, 34% were PD-L1 negative, and the response rate is 65% in PD-L1-positive and 50% in PD-L1-negative [patients].”
The median duration of response was not reached, she added.
The median follow-up was 9.2 months, with the last patient enrolled having 6 months of follow-up. At the data cutoff, 19 patients had disease progression, and 8 had died of their disease.
The median overall survival also was not reached, Dr. Lan said, but overall survival at 9 months was 80%. The median progression-free survival was 7.6 months, and the 6-month progression-free survival rate was 58%.
Grade 3 or greater treatment-related adverse events occurred in 68.9% of patients; adverse events occurring in at least 5% of patients included hypertension (22.2%), fatigue (15.6%), anemia (13.3%), and thrombocytopenia (6.7%).
“Nineteen patients were still on treatment at the data cutoff date, and 26 patients discontinued the study,” Dr. Lan said. “The most common reason to discontinue was disease progression, and three patients discontinued the study due to adverse events.”
“These preliminary results are very encouraging,” Dr. Lan said. “As we know, pembrolizumab is approved as the second-line therapy in recurrent cervical cancer [in] PD-L1-positive patients, and the objective response rate with pembrolizumab monotherapy for recurrent cervical cancer is only 17%, as reported in KEYNOTE-028 [J Clin Oncol. 2017 Dec 20;35(36):4035-41].”
Furthermore, apatinib monotherapy has been studied with only modest results.
“But in our study, this combination is really effective in recurrent cervical cancer, and we see a very durable response,” she said, again emphasizing that those responses occurred regardless of PD-L1 expression. “So this is important. ... We think our findings expand the opportunity of patients with recurrent cervical cancer to receive immune therapy.”
Study participants will be followed for 2 years, Dr. Lan noted.
She reported having no disclosures. The study is sponsored by Sun Yat-sen University.
[email protected]
SOURCE: Lan C et al. SGO 2020, Abstract 55.
FROM SGO 2020
When to suspect calciphylaxis and what to do about it
If the shoe fits a presumptive clinical diagnosis of calciphylaxis, wear it – and don’t assume that ordering imaging studies or histology will make for a better fit or is even necessary.
That was the key message of Karl M. Saardi, MD, during his video presentation at a virtual meeting held by the George Washington University department of dermatology. The virtual meeting included presentations that had been slated for the annual meeting of the American Academy of Dermatology, which was canceled because of the COVID-19 pandemic.
“ said Dr. Saardi, a dermatology resident at Georgetown University in Washington, D.C.
He presented a single-center, retrospective study that underscored the diagnostic challenges posed by calciphylaxis, a condition for which there are no generally accepted clinical, radiographic, or histologic diagnostic criteria.
The rare skin condition is characterized by calcium deposition in small arterioles and capillaries in the skin and subcutaneous tissue. It’s most common in patients with end-stage renal disease who are on dialysis; however, there is also an increasingly recognized nonuremic variant that’s associated with the use of warfarin, chronic steroids, obesity, and possibly with being antiphospholipid antibody positive.
Calciphylaxis is an extremely painful condition – the pain is ischemic in nature – and it’s associated with substantial morbidity as well as a mortality rate that in many series exceeds 50%. Affected individuals typically present with progressive, painful retiform purpura on the legs, belly, buttocks, and other fatty body sites.
Dr. Saardi’s study entailed a retrospective look at the medical records and pathologic reports of 57 patients who underwent skin biopsy for suspected calciphylaxis. Of the 57, 18 had no antecedent imaging studies done during the preceding 3 months; 8 of those 18 (40%), had a confirmatory positive biopsy. A total of 39 patients did have imaging studies, deemed positive for calciphylaxis in 11 cases, which in only 5 of the 11 imaging-positive cases (45%) were subsequently confirmed by positive biopsy.
And finally, of the 28 patients with negative imaging studies, 10 (36%), had a positive biopsy. Those positive biopsy rates, ranging from 36% to 45%, did not differ statistically. Thus, whether an imaging study was positive or negative, or wasn’t even done, made no difference in terms of the ultimate diagnosis.
“You may not need imaging studies, because imaging has often been done before the consultation is requested because people are looking for things like arterial thrombus, cellulitis, [deep vein thrombosis] or something like that,” Dr. Saardi noted. “In our series, the indication was never calciphylaxis, it was always something like pain, infection, swelling, suspected [deep vein thrombosis], things like that.”
The classic signature of calciphylaxis on plain x-ray is net-like calcifications in skin and subcutaneous tissue. In one study, this often-subtle finding was associated with a 830% increased likelihood of a positive biopsy, with a specificity of 90%; however, these x-ray changes were only found in 13 of 29 patients with biopsy-confirmed calciphylaxis.
“It’s really important when you request plain films in these patients to review the images yourself or together with the radiologist because oftentimes the indication for imaging will be very different from what we’re looking for. Radiologists often won’t know to look for this specifically,” Dr. Saardi said.
The classic histopathologic finding is calcification of the small- and medium-sized vessels in the dermis and subcutaneous soft tissue. However, sometimes all that’s present are small intravascular inflammatory thrombi with intimal hyperplasia.
Skin biopsies are not infrequently falsely negative or nondiagnostic. To maximize the utility of the procedure, it’s important to go deep and gather a tissue sample that extends into subcutaneous tissue.
“You need to do a very deep punch or double-punch biopsy,” he said. “Another key is to avoid biopsy if the pretest probability of calciphylaxis is high because a negative biopsy shouldn’t necessarily reassure you or cause you to withhold treatment. And with the concern about pathergy or Koebnerization of the area causing a wound that’s never going to heal, sometimes a biopsy is not needed if the pretest suspicion is high enough.”
Other investigators have shown that the likelihood of an informative biopsy is enhanced by using a calcium stain on the specimen and having an experienced dermatopathologist do the evaluation. Also, the use of a radiographically guided core needle biopsy to ensure that the physician is getting sufficiently deep into subcutaneous fat is now under evaluation.
In addition to plain radiographs, other imaging methods that are sometimes used to evaluate soft-tissue sites for suspected calciphylaxis included CT and ultrasound. Dr. Saardi is particularly intrigued by reports from investigators at Harvard University regarding the utility of nuclear bone scintigraphy; in one study, this form of imaging was positive in 16 of 18 patients with clinically diagnosed calciphylaxis, versus just 1 of 31 controls with end-stage renal disease.
“We’ve started doing this in situations where biopsy is not desirable or feasible at that moment,” he said.
Dr. Saardi reported having no financial conflicts regarding his presentation.
If the shoe fits a presumptive clinical diagnosis of calciphylaxis, wear it – and don’t assume that ordering imaging studies or histology will make for a better fit or is even necessary.
That was the key message of Karl M. Saardi, MD, during his video presentation at a virtual meeting held by the George Washington University department of dermatology. The virtual meeting included presentations that had been slated for the annual meeting of the American Academy of Dermatology, which was canceled because of the COVID-19 pandemic.
“ said Dr. Saardi, a dermatology resident at Georgetown University in Washington, D.C.
He presented a single-center, retrospective study that underscored the diagnostic challenges posed by calciphylaxis, a condition for which there are no generally accepted clinical, radiographic, or histologic diagnostic criteria.
The rare skin condition is characterized by calcium deposition in small arterioles and capillaries in the skin and subcutaneous tissue. It’s most common in patients with end-stage renal disease who are on dialysis; however, there is also an increasingly recognized nonuremic variant that’s associated with the use of warfarin, chronic steroids, obesity, and possibly with being antiphospholipid antibody positive.
Calciphylaxis is an extremely painful condition – the pain is ischemic in nature – and it’s associated with substantial morbidity as well as a mortality rate that in many series exceeds 50%. Affected individuals typically present with progressive, painful retiform purpura on the legs, belly, buttocks, and other fatty body sites.
Dr. Saardi’s study entailed a retrospective look at the medical records and pathologic reports of 57 patients who underwent skin biopsy for suspected calciphylaxis. Of the 57, 18 had no antecedent imaging studies done during the preceding 3 months; 8 of those 18 (40%), had a confirmatory positive biopsy. A total of 39 patients did have imaging studies, deemed positive for calciphylaxis in 11 cases, which in only 5 of the 11 imaging-positive cases (45%) were subsequently confirmed by positive biopsy.
And finally, of the 28 patients with negative imaging studies, 10 (36%), had a positive biopsy. Those positive biopsy rates, ranging from 36% to 45%, did not differ statistically. Thus, whether an imaging study was positive or negative, or wasn’t even done, made no difference in terms of the ultimate diagnosis.
“You may not need imaging studies, because imaging has often been done before the consultation is requested because people are looking for things like arterial thrombus, cellulitis, [deep vein thrombosis] or something like that,” Dr. Saardi noted. “In our series, the indication was never calciphylaxis, it was always something like pain, infection, swelling, suspected [deep vein thrombosis], things like that.”
The classic signature of calciphylaxis on plain x-ray is net-like calcifications in skin and subcutaneous tissue. In one study, this often-subtle finding was associated with a 830% increased likelihood of a positive biopsy, with a specificity of 90%; however, these x-ray changes were only found in 13 of 29 patients with biopsy-confirmed calciphylaxis.
“It’s really important when you request plain films in these patients to review the images yourself or together with the radiologist because oftentimes the indication for imaging will be very different from what we’re looking for. Radiologists often won’t know to look for this specifically,” Dr. Saardi said.
The classic histopathologic finding is calcification of the small- and medium-sized vessels in the dermis and subcutaneous soft tissue. However, sometimes all that’s present are small intravascular inflammatory thrombi with intimal hyperplasia.
Skin biopsies are not infrequently falsely negative or nondiagnostic. To maximize the utility of the procedure, it’s important to go deep and gather a tissue sample that extends into subcutaneous tissue.
“You need to do a very deep punch or double-punch biopsy,” he said. “Another key is to avoid biopsy if the pretest probability of calciphylaxis is high because a negative biopsy shouldn’t necessarily reassure you or cause you to withhold treatment. And with the concern about pathergy or Koebnerization of the area causing a wound that’s never going to heal, sometimes a biopsy is not needed if the pretest suspicion is high enough.”
Other investigators have shown that the likelihood of an informative biopsy is enhanced by using a calcium stain on the specimen and having an experienced dermatopathologist do the evaluation. Also, the use of a radiographically guided core needle biopsy to ensure that the physician is getting sufficiently deep into subcutaneous fat is now under evaluation.
In addition to plain radiographs, other imaging methods that are sometimes used to evaluate soft-tissue sites for suspected calciphylaxis included CT and ultrasound. Dr. Saardi is particularly intrigued by reports from investigators at Harvard University regarding the utility of nuclear bone scintigraphy; in one study, this form of imaging was positive in 16 of 18 patients with clinically diagnosed calciphylaxis, versus just 1 of 31 controls with end-stage renal disease.
“We’ve started doing this in situations where biopsy is not desirable or feasible at that moment,” he said.
Dr. Saardi reported having no financial conflicts regarding his presentation.
If the shoe fits a presumptive clinical diagnosis of calciphylaxis, wear it – and don’t assume that ordering imaging studies or histology will make for a better fit or is even necessary.
That was the key message of Karl M. Saardi, MD, during his video presentation at a virtual meeting held by the George Washington University department of dermatology. The virtual meeting included presentations that had been slated for the annual meeting of the American Academy of Dermatology, which was canceled because of the COVID-19 pandemic.
“ said Dr. Saardi, a dermatology resident at Georgetown University in Washington, D.C.
He presented a single-center, retrospective study that underscored the diagnostic challenges posed by calciphylaxis, a condition for which there are no generally accepted clinical, radiographic, or histologic diagnostic criteria.
The rare skin condition is characterized by calcium deposition in small arterioles and capillaries in the skin and subcutaneous tissue. It’s most common in patients with end-stage renal disease who are on dialysis; however, there is also an increasingly recognized nonuremic variant that’s associated with the use of warfarin, chronic steroids, obesity, and possibly with being antiphospholipid antibody positive.
Calciphylaxis is an extremely painful condition – the pain is ischemic in nature – and it’s associated with substantial morbidity as well as a mortality rate that in many series exceeds 50%. Affected individuals typically present with progressive, painful retiform purpura on the legs, belly, buttocks, and other fatty body sites.
Dr. Saardi’s study entailed a retrospective look at the medical records and pathologic reports of 57 patients who underwent skin biopsy for suspected calciphylaxis. Of the 57, 18 had no antecedent imaging studies done during the preceding 3 months; 8 of those 18 (40%), had a confirmatory positive biopsy. A total of 39 patients did have imaging studies, deemed positive for calciphylaxis in 11 cases, which in only 5 of the 11 imaging-positive cases (45%) were subsequently confirmed by positive biopsy.
And finally, of the 28 patients with negative imaging studies, 10 (36%), had a positive biopsy. Those positive biopsy rates, ranging from 36% to 45%, did not differ statistically. Thus, whether an imaging study was positive or negative, or wasn’t even done, made no difference in terms of the ultimate diagnosis.
“You may not need imaging studies, because imaging has often been done before the consultation is requested because people are looking for things like arterial thrombus, cellulitis, [deep vein thrombosis] or something like that,” Dr. Saardi noted. “In our series, the indication was never calciphylaxis, it was always something like pain, infection, swelling, suspected [deep vein thrombosis], things like that.”
The classic signature of calciphylaxis on plain x-ray is net-like calcifications in skin and subcutaneous tissue. In one study, this often-subtle finding was associated with a 830% increased likelihood of a positive biopsy, with a specificity of 90%; however, these x-ray changes were only found in 13 of 29 patients with biopsy-confirmed calciphylaxis.
“It’s really important when you request plain films in these patients to review the images yourself or together with the radiologist because oftentimes the indication for imaging will be very different from what we’re looking for. Radiologists often won’t know to look for this specifically,” Dr. Saardi said.
The classic histopathologic finding is calcification of the small- and medium-sized vessels in the dermis and subcutaneous soft tissue. However, sometimes all that’s present are small intravascular inflammatory thrombi with intimal hyperplasia.
Skin biopsies are not infrequently falsely negative or nondiagnostic. To maximize the utility of the procedure, it’s important to go deep and gather a tissue sample that extends into subcutaneous tissue.
“You need to do a very deep punch or double-punch biopsy,” he said. “Another key is to avoid biopsy if the pretest probability of calciphylaxis is high because a negative biopsy shouldn’t necessarily reassure you or cause you to withhold treatment. And with the concern about pathergy or Koebnerization of the area causing a wound that’s never going to heal, sometimes a biopsy is not needed if the pretest suspicion is high enough.”
Other investigators have shown that the likelihood of an informative biopsy is enhanced by using a calcium stain on the specimen and having an experienced dermatopathologist do the evaluation. Also, the use of a radiographically guided core needle biopsy to ensure that the physician is getting sufficiently deep into subcutaneous fat is now under evaluation.
In addition to plain radiographs, other imaging methods that are sometimes used to evaluate soft-tissue sites for suspected calciphylaxis included CT and ultrasound. Dr. Saardi is particularly intrigued by reports from investigators at Harvard University regarding the utility of nuclear bone scintigraphy; in one study, this form of imaging was positive in 16 of 18 patients with clinically diagnosed calciphylaxis, versus just 1 of 31 controls with end-stage renal disease.
“We’ve started doing this in situations where biopsy is not desirable or feasible at that moment,” he said.
Dr. Saardi reported having no financial conflicts regarding his presentation.













