User login
Society of Gynecologic Surgeons meeting champions training of future gynecologic surgeons
It was such a pleasure at the 48th Annual Meeting of the Society of Gynecologic Surgeons (SGS) to witness record meeting attendance and strong enthusiasm after 2 depressing years with the COVID-19 pandemic. Evidently, everyone was tired of virtual gatherings and presentations. As a dedicated surgical educator and a passionate vaginal surgeon, SGS President Carl Zimmerman, MD, chose “Gynecologic surgery training: Lessons from the past, looking to the future” as the theme for this year’s meeting. Our keynote speakers, Patricia Turner, MD, MBA, Executive Director of the American College of Surgeons, and Marta Crispens, MD, MBA, Professor and Division Director of Gynecologic Oncology at Vanderbilt, were spot on. They reviewed the current status of surgical training eloquently with convincing statistics. They mapped out the path forward by stressing collaboration and proposing strategies that might produce competent surgeons in all fields.
The meeting featured 2 panel discussions. The first, titled “Innovations in training gynecologic surgeons,” reviewed tracking in residency, use of simulation for surgical proficiency, and European perspective on training. The panelists emphasized the dwindling numbers of surgical procedures, especially vaginal hysterectomies. Cecile Ferrando, MD, suggested that tracking might be part of the answer, based on their experience, which provided a structure for residents to obtain concentrated training in their areas of interest. Douglas Miyazaki, MD, presented the prospects for his innovative, federally funded vaginal surgery simulation model. Oliver Preyer, MD, presented Austrian trainees’ low case volumes, showing that the grass was not actually greener on the other side. Finally, this panel reinvigorated ongoing debate about separating Obstetrics and Gynecology.
The second panel, “Operating room safety and efficiency,” shed light on human and nontechnical factors that might be as critical as surgeons’ skills and experience, and it highlighted an innovative technology that monitored and analyzed all operating room parameters to improve operational processes and surgical technique. Points by Jason Wright, MD, on the relationship between surgical volume and outcomes complemented the meeting theme and the first panel discussion. He underlined how much surgical volume of individual surgeons and hospitals mattered, but he also indicated that restrictive credentialing strategies might lead to unintended consequences.
Importantly, the SGS Women’s Council held a panel on the “Impact of Texas legislation on the physician/patient relationship” to provide a platform for members who had mixed feelings about attending this meeting in Texas.
The SGS meeting also included several popular postgraduate courses on multidisciplinary management of Müllerian anomalies, pelvic fistula treatment, surgical simulation, management modalities for uterine fibroids, and medical innovation and entrepreneurship. In this special section and in the next issue of OBG M
It was such a pleasure at the 48th Annual Meeting of the Society of Gynecologic Surgeons (SGS) to witness record meeting attendance and strong enthusiasm after 2 depressing years with the COVID-19 pandemic. Evidently, everyone was tired of virtual gatherings and presentations. As a dedicated surgical educator and a passionate vaginal surgeon, SGS President Carl Zimmerman, MD, chose “Gynecologic surgery training: Lessons from the past, looking to the future” as the theme for this year’s meeting. Our keynote speakers, Patricia Turner, MD, MBA, Executive Director of the American College of Surgeons, and Marta Crispens, MD, MBA, Professor and Division Director of Gynecologic Oncology at Vanderbilt, were spot on. They reviewed the current status of surgical training eloquently with convincing statistics. They mapped out the path forward by stressing collaboration and proposing strategies that might produce competent surgeons in all fields.
The meeting featured 2 panel discussions. The first, titled “Innovations in training gynecologic surgeons,” reviewed tracking in residency, use of simulation for surgical proficiency, and European perspective on training. The panelists emphasized the dwindling numbers of surgical procedures, especially vaginal hysterectomies. Cecile Ferrando, MD, suggested that tracking might be part of the answer, based on their experience, which provided a structure for residents to obtain concentrated training in their areas of interest. Douglas Miyazaki, MD, presented the prospects for his innovative, federally funded vaginal surgery simulation model. Oliver Preyer, MD, presented Austrian trainees’ low case volumes, showing that the grass was not actually greener on the other side. Finally, this panel reinvigorated ongoing debate about separating Obstetrics and Gynecology.
The second panel, “Operating room safety and efficiency,” shed light on human and nontechnical factors that might be as critical as surgeons’ skills and experience, and it highlighted an innovative technology that monitored and analyzed all operating room parameters to improve operational processes and surgical technique. Points by Jason Wright, MD, on the relationship between surgical volume and outcomes complemented the meeting theme and the first panel discussion. He underlined how much surgical volume of individual surgeons and hospitals mattered, but he also indicated that restrictive credentialing strategies might lead to unintended consequences.
Importantly, the SGS Women’s Council held a panel on the “Impact of Texas legislation on the physician/patient relationship” to provide a platform for members who had mixed feelings about attending this meeting in Texas.
The SGS meeting also included several popular postgraduate courses on multidisciplinary management of Müllerian anomalies, pelvic fistula treatment, surgical simulation, management modalities for uterine fibroids, and medical innovation and entrepreneurship. In this special section and in the next issue of OBG M
It was such a pleasure at the 48th Annual Meeting of the Society of Gynecologic Surgeons (SGS) to witness record meeting attendance and strong enthusiasm after 2 depressing years with the COVID-19 pandemic. Evidently, everyone was tired of virtual gatherings and presentations. As a dedicated surgical educator and a passionate vaginal surgeon, SGS President Carl Zimmerman, MD, chose “Gynecologic surgery training: Lessons from the past, looking to the future” as the theme for this year’s meeting. Our keynote speakers, Patricia Turner, MD, MBA, Executive Director of the American College of Surgeons, and Marta Crispens, MD, MBA, Professor and Division Director of Gynecologic Oncology at Vanderbilt, were spot on. They reviewed the current status of surgical training eloquently with convincing statistics. They mapped out the path forward by stressing collaboration and proposing strategies that might produce competent surgeons in all fields.
The meeting featured 2 panel discussions. The first, titled “Innovations in training gynecologic surgeons,” reviewed tracking in residency, use of simulation for surgical proficiency, and European perspective on training. The panelists emphasized the dwindling numbers of surgical procedures, especially vaginal hysterectomies. Cecile Ferrando, MD, suggested that tracking might be part of the answer, based on their experience, which provided a structure for residents to obtain concentrated training in their areas of interest. Douglas Miyazaki, MD, presented the prospects for his innovative, federally funded vaginal surgery simulation model. Oliver Preyer, MD, presented Austrian trainees’ low case volumes, showing that the grass was not actually greener on the other side. Finally, this panel reinvigorated ongoing debate about separating Obstetrics and Gynecology.
The second panel, “Operating room safety and efficiency,” shed light on human and nontechnical factors that might be as critical as surgeons’ skills and experience, and it highlighted an innovative technology that monitored and analyzed all operating room parameters to improve operational processes and surgical technique. Points by Jason Wright, MD, on the relationship between surgical volume and outcomes complemented the meeting theme and the first panel discussion. He underlined how much surgical volume of individual surgeons and hospitals mattered, but he also indicated that restrictive credentialing strategies might lead to unintended consequences.
Importantly, the SGS Women’s Council held a panel on the “Impact of Texas legislation on the physician/patient relationship” to provide a platform for members who had mixed feelings about attending this meeting in Texas.
The SGS meeting also included several popular postgraduate courses on multidisciplinary management of Müllerian anomalies, pelvic fistula treatment, surgical simulation, management modalities for uterine fibroids, and medical innovation and entrepreneurship. In this special section and in the next issue of OBG M
How to teach vaginal surgery through simulation
Vaginal surgery, including vaginal hysterectomy, is slowly becoming a dying art. According to the National Inpatient Sample and the Nationwide Ambulatory Surgery Sample from 2018, only 11.8% of all hysterectomies were performed vaginally.1 The combination of uterine-sparing surgeries, advances in conservative therapies for benign uterine conditions, and the diversification of minimally invasive routes (laparoscopic and robotic) has resulted in a continued downtrend in vaginal surgical volumes. This shift has led to fewer operative learning opportunities and declining graduating resident surgical volume.2 According to the Accreditation Council for Graduate Medical Education (ACGME), the minimum number of vaginal hysterectomies is 15, which represents only the minimum accepted exposure and does not imply competency.
In response, surgical simulation has been used for skill acquisition and maintenance outside of the operating room in a learning environment that is safe for the learners and does not expose patients to additional risk. Educators are uniquely poised to use simulation to teach residents and to evaluate their procedural competency. Although vaginal surgery, specifically vaginal hysterectomy, continues to decline, it can be resuscitated with the assistance of surgical simulation.
In this article, we provide a broad overview of vaginal surgical simulation. We discuss the basic tenets of simulation, review how to teach and evaluate vaginal surgical skills, and present some of the commonly available vaginal surgery simulation models and their associated resources, cost, setup time, fidelity, and limitations.
Simulation principles relevant for vaginal hysterectomy simulation
Here, we review simulation-based learning principles that will help place specific simulation models into perspective.
One size does not fit all
Simulation, like many educational interventions, does not work via a “one-size-fits-all” approach. While the American College of Obstetricians and Gynecologists (ACOG) Simulations Working Group (SWG) has created a toolkit (available online at https://www.acog.org/education-and-events/simulations/about/curriculum) with many ready-to-use how-to simulation descriptions and lesson plans that cover common topics, what works in one setting may not work in another. The SWG created those modules to help educators save time and resources and to avoid reinventing the wheel for each simulation session. However, these simulations need to be adapted to the local needs of trainees and resources, such as faculty time, space, models, and funding.
Cost vs fidelity
It is important to distinguish between cost and fidelity. “Low cost” is often incorrectly used interchangeably with “low fidelity” when referring to models and simulations. The most basic principle of fidelity is that it is associated with situational realism that in turn, drives learning.3,4 For example, the term high fidelity does apply to a virtual reality robotic surgery simulator, which also is high cost. However, a low-cost beef tongue model of fourth-degree laceration5 is high fidelity, while more expensive commercial models are less realistic, which makes them high cost and low fidelity.6 When selecting simulation models, educators need to consider cost based on their available resources and the level of fidelity needed for their learners.
Continue to: Task breakdown...
Task breakdown
As surgeon-educators, we love to teach! And while educators are passionate about imparting vaginal hysterectomy skills to the next generation of surgeons, it is important to assess where the learners are technically. Vaginal hysterectomy is a high-complexity procedure, with each step involving a unique skill set that is new to residents as learners; this is where the science of learning can help us teach more effectively.7 Focusing on doing the entire procedure all at once is more likely to result in cognitive overload, while a better approach is to break the procedure down into several components and practice those parts until goal proficiency is reached.
Deliberate practice
The idea of deliberate practice was popularized by Malcolm Gladwell in his book titled Outliers, in which he gives examples of how 10,000 hours of practice leads to mastery of complex skills. This concept was deepened by the work of cognitive psychologist Anders Ericsson, who emphasized that not only the duration but also the quality of practice—which involves concentration, analysis, and problem-solving—leads to the most effective training.8
In surgical education, this concept translates into many domains. For example, an individualized learning plan includes frequent low-stakes assessments, video recording for later viewing and analysis, surgical coaching, and detailed planning of future training sessions to incorporate past performance. “Just doing” surgery on a simulator (or in the operating room) results in missed learning opportunities.
Logistics and implementation: Who, where, when
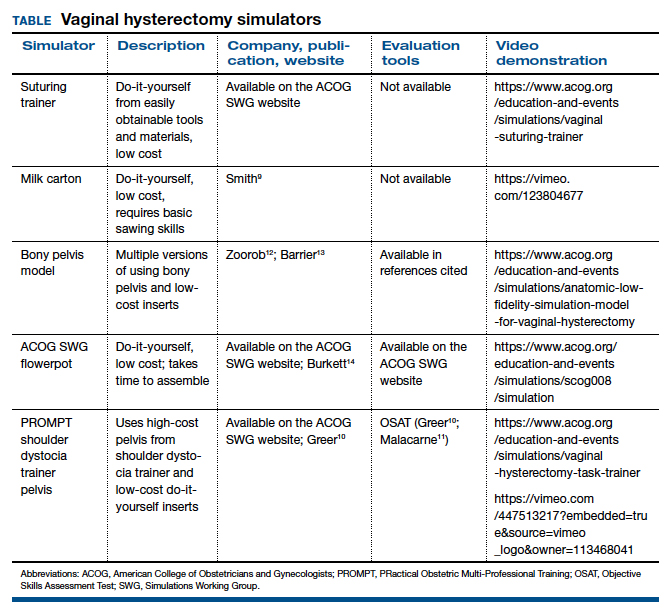
The simulation “formula” takes into account multiple factors but should start with learning objectives and then an assessment of what resources are available to address them. For example, if one surgeon-educator and one resident-learner are available for 30 minutes in between cases in the operating room, and the goal is to teach the resident clamp-and-tie technique on pedicles, the “milk carton” model9 and a few instruments from the vaginal hysterectomy tray are ideal for this training. On the other hand, if it is important to achieve competency for an entire procedure prior to operating room debut and a group of surgeon-educators is available to share the time commitment of 2-hour sessions per each resident, then the PROMPT (PRactical Obstetric Multi-Professional Training) shoulder dystocia model could be used (TABLE).10-14
Learning curves
Ideally, educators would like to know how many simulated training sessions are needed for a learner to reach a proficiency level and become operating room ready. Such information about learning curves, unfortunately, is not available yet for vaginal hysterectomies. The first step in the process is to establish a baseline for performance to know a starting point, with assessment tools specific to each simulator; the next step is to study how many “takes” are needed for learners to move through their learning curve.15 The use of assessment tools can help assess each learner’s progression.
Continue to: Evaluation, assessment, and feedback...
Evaluation, assessment, and feedback
With more emphasis being placed on patient safety and transparency in every aspect of health care, including surgical training, graduate medical education leaders increasingly highlight the importance of objective assessment tools and outcome-based standards for certification of competency in surgery.16,17 Commonly used assessment tools that have reliability and validity evidence include surgical checklists and global rating scales. Checklists for common gynecologic procedures, including vaginal hysterectomy, as well as a global rating scale specifically developed for vaginal surgery (Vaginal Surgical Skills Index, VSSI)18 are accessible on the ACOG Simulations Working Group Surgical Curriculum in Obstetrics and Gynecology website.19
While checklists contain the main steps of each procedure, these lists do not assess for how well each step of the procedure is performed. By contrast, global rating scales, such as the VSSI, can discriminate between surgeons with different skill levels both in the simulation and operating room settings; each metric within the global rating scale (for example, time and motion) does not pertain to the performance of a procedure’s specific step but rather to the overall performance of the entire procedure.18,20 Hence, to provide detailed feedback, especially for formative assessment, both checklists and global rating scales often are used together.
Although standardized, checklists and global rating scales ultimately are still subjective (not objective) assessment tools. Recently, more attention has been to use surgical data science, particularly artificial intelligence methods, to objectively assess surgical performance by analyzing data generated during the performance of surgery, such as instrumental motion and video.21 These methods have been applied to a wide range of surgical techniques, including open, laparoscopic, robotic, endoscopic, and microsurgical approaches. Most of these types of studies have used assessment of surgical skill as the main outcome, with fewer studies correlating skill with clinically relevant metrics, such as patient outcomes.22-25 Although this is an area of active research, these methods are still being developed, and their validity and utility are not well established. For now, educators should continue to use validated checklists and global rating skills to help assess any type of surgical performance, particularly vaginal surgery.
Vaginal surgical simulation models
Vaginal surgery requires a surgeon to operate in a narrow, deep space. This requires ambidexterity, accurate depth perception, understanding of how to handle tissues, and use of movements that are efficient, fluid, and rhythmic. Multiple proposed simulation models are relevant to vaginal surgery, and these vary based on level of fidelity, cost, feasibility, ability to maintain standardization, ease of construction (if required), and generalizability to all of pelvic surgery (that is, procedure specific vs basic skills focused).10,11,13,26-31
Below, we describe various simulation models that are available for teaching vaginal surgical skills.
Vaginal hysterectomy simulation model
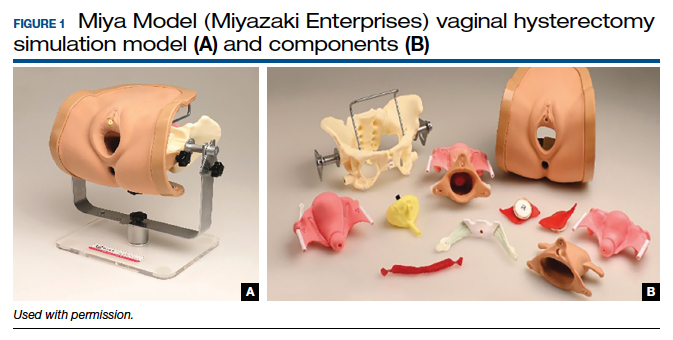
One commercially available simulation model for vaginal hysterectomy (as well as other vaginal surgical procedures, such as midurethral sling and anterior and posterior colporrhaphy) is the Miya Model (Miyazaki Enterprises) (FIGURE 1) and its accompanying MiyaMODEL App. In a multi-institutional study funded by the National Institutes of Health (NIH), the Miya Model, when used with the VSSI, was shown to be a valid assessment tool in terms of ability to differentiate a competent from a noncompetent surgeon.20 Currently, an ongoing NIH-sponsored multi-institutional study is assessing the Miya Model as a teaching tool and whether skills acquired on the Miya Model are transferable to the operating room.
Continue to: Low-cost vaginal hysterectomy models...
Low-cost vaginal hysterectomy models
Multiple low-cost vaginal hysterectomy simulation models are described. Two models developed many years ago include the ACOG SWG flowerpot model14 and the PROMPT shoulder dystocia pelvic trainer model.10,11,14 The former model is low cost as it can be constructed from easily obtained household materials, but its downside is that it takes time and effort to obtain the materials and to assemble them. The latter model is faster to assemble but requires one to use a PROMPT pelvis for shoulder dystocia training, which has a considerable upfront cost. However, it is available in most hospitals with considerable obstetrical volume, and it allows for the most realistic perineum, which is helpful in recreating the feel of vaginal surgery, including retraction and exposure.
Many models created and described in the literature are variations of the models mentioned above, and many use commercially available low-cost bony pelvis models and polyvinyl chloride (PVC) pipes as a foundation for the soft tissue inserts to attach.12,13,31-33 Each model varies on what it “teaches best” regarding realism—for example, teaching anatomy, working in a tight space, dissection, or clamp placement and suture ligature.
Furthermore, since vaginal hysterectomy is a high-complexity procedure in terms of skills (working in confined space, limited view, “upside-down” anatomy, and need to direct assistants for retraction and exposure), task breakdown is important for simulation learning, as it is not efficient to repeat the entire procedure until proficiency is reached. Two trainers have been described to address that need: the milk carton and the vaginal suturing trainer. The latter allows learners to practice clamp placement and pedicle ligation multiple times, including in confined space (FIGURE 2), and the former allows them to do the same in a procedural matter as the clamp placement moves caudad to cephalad during the procedure (FIGURE 3).


Native tissue pelvic floor surgery simulation
While there are few publications regarding surgical simulation models for native tissue pelvic floor surgeries, a low-cost anterior and posterior repair model was developed for the ACOG SWG Simulation Toolkit and published online in 2017, after their peer-review process. The fidelity is moderate for this low-cost model, which costs less than $5 per use. The simulation model requires a new vaginal insert for each learner, which is fast and easy to make and requires only a few components; however, the bony pelvis (for example, the flowerpot model) needs to be purchased or created. The stage of the anterior wall prolapse can be adjusted by the amount of fluid placed in the balloon, which is used to simulate the bladder. The more fluid that is placed in the “bladder,” the more severe the anterior wall prolapse appears. The vaginal caliber can be adjusted, if needed, by increasing or decreasing the size of the components to create the vagina, but the suggested sizes simulate a significantly widened vaginal caliber that would benefit from a posterior repair with perineorrhaphy. Although there is no validity evidence for this model, a skills assessment is available through the ACOG Simulation Surgical Curriculum. Of note, native tissue colpopexy repairs are also possible with this model (or another high-fidelity model, such as the Miya Model), if the sacrospinous ligaments and/or uterosacral ligaments are available on the pelvic model in use. This model’s limitations include the absence of a high-fidelity plane of dissection of the vaginal muscularis, and that no bleeding is encountered, which is the case for many low-cost models.19,34
Fundamentals of Vaginal Surgery (FVS) basic surgical skills simulation
The FVS simulation system, consisting of a task trainer paired with 6 selected surgical tasks, was developed to teach basic skills used in vaginal surgery.35 The FVS task trainer is 3D printed and has 3 main components: a base piece that allows for different surgical materials to be secured, a depth extender, and a width reducer. In addition, it has a mobile phone mount and a window into the system to enable video capture of skills exercises.
The FVS simulator is designed to enable 6 surgical tasks, including one-handed knot tying, two-handed knot tying, running suturing, plication suturing, Heaney transfixion pedicle ligation, and free pedicle ligation (FIGURE 4). In a pilot study, the FVS simulation system was deemed representative of the intended surgical field, useful for inclusion in a training program, and favored as a tool for both training and testing. Additionally, an initial proficiency score of 400 was set, which discriminated between novice and expert surgeons.35
An advantage of this simulation system is that it allows learners to focus on basic skills, rather than on an entire specific procedure. Further, the system is standardized, as it is commercially manufactured; this also allows for easy assembly. The disadvantage of this model is that it cannot be modified to teach specific vaginal procedures, and it must be purchased, rather than constructed on site. Further studies are needed to create generalizable proficiency scores and to assess its use in training and testing. For more information on the FVS simulation model, visit the Arbor Simulation website (http://arborsim.com).
Surgical simulation’s important role
Surgical skills can be learned and improved in the simulation setting in a low-stakes, low-pressure environment. Simulation can enable basic skills development and then higher-level learning of complex procedures. Skill assessment is important to aid in learning (via formative assessments) and for examination or certification (summative assessments).
With decreasing vaginal surgical volumes occurring nationally, it is becoming even more important to use surgical simulation to teach and maintain vaginal surgical skills. In this article, we reviewed various different simulation models that can be used for developing vaginal surgical skills and presented the advantages, limitations, and resources relevant for each simulation model. ●
- Wright JD, Huang Y, Li AH, et al. Nationwide estimates of annual inpatient and outpatient hysterectomies performed in the United States. Obstet Gynecol. 2022;139:446-448.
- Gressel GM, Potts JR 3rd, Cha S, et al. Hysterectomy route and numbers reported by graduating residents in obstetrics and gynecology training programs. Obstet Gynecol. 2020;135:268-273.
- Lioce L, ed. Healthcare Simulation Dictionary. 2nd ed. Rockville, MD; Agency for Healthcare Research and Quality: 2020. AHRQ Publication No. 20-0019.
- Norman G, Dore K, Grierson L. The minimal relationship between simulation fidelity and transfer of learning. Med Educ. 2012;46:636-647.
- Illston JD, Ballard AC, Ellington DR, et al. Modified beef tongue model for fourth-degree laceration repair simulation. Obstet Gynecol. 2017;129:491-496.
- WorldPoint website. 3B Scientific Episiotomy and Suturing Trainer. https://www.worldpoint.com/3b-episiotomy-and-suturing-sim. Accessed April 20, 2022.
- Balafoutas D, Joukhadar R, Kiesel M, et al. The role of deconstructive teaching in the training of laparoscopy. JSLS. 2019;23:e2019.00020.
- Ericsson KA, Harwell KW. Deliberate practice and proposed limits on the effects of practice on the acquisition of expert performance: why the original definition matters and recommendations for future research. Front Psychol. 2019;10:2396.
- Smith TM, Fenner DE. Vaginal hysterectomy teaching model—an educational video. Female Pelvic Med Reconstr Surg. 2012;18:S43. Abstract.
- Greer JA, Segal S, Salva CR, et al. Development and validation of simulation training for vaginal hysterectomy. J Minim Invasive Gynecol. 2014;21:74-82.
- Malacarne DR, Escobar CM, Lam CJ, et al. Teaching vaginal hysterectomy via simulation: creation and validation of the objective skills assessment tool for simulated vaginal hysterectomy on a task trainer and performance among different levels of trainees. Female Pelvic Med Reconstr Surg. 2019;25:298-304.
- Zoorob D, Frenn R, Moffitt M, et al. Multi-institutional validation of a vaginal hysterectomy simulation model for resident training. J Minim Invasive Gynecol. 2021;28:1490-1496.e1.
- Barrier BF, Thompson AB, McCullough MW, et al. A novel and inexpensive vaginal hysterectomy simulator. Simul Healthc. 2012;7:374-379.
- Burkett LS, Makin J, Ackenbom M, et al. Validation of transvaginal hysterectomy surgical model—modification of the flowerpot model to improve vesicovaginal plane simulation. J Minim Invasive Gynecol. 2021;28:1526-1530.
- Escobar C, Malacarne Pape D, Ferrante KL, et al. Who should be teaching vaginal hysterectomy on a task trainer? A multicenter randomized trial of peer versus expert coaching. J Surg Simul. 2020;7:63-72.
- The obstetrics and gynecology milestone project. J Grad Med Educ. 2014;6(1 suppl 1):129-143.
- Nasca TJ, Philibert I, Brigham T, et al. The next GME accreditation system—rationale and benefits. N Engl J Med. 2012;366:1051-1056.
- Chen CCG, Korn A, Klingele C, et al. Objective assessment of vaginal surgical skills. Am J Obstet Gynecol. 2010;203:79.e1-8.
- American College of Obstetricians and Gynecologists. Surgical curriculum in obstetrics and gynecology. https://www.acog.org /education-and-events/simulations/surgical-curriculum-in-ob-gyn.
- Chen CCG, Lockrow EG, DeStephano CC, et al. Establishing validity for a vaginal hysterectomy simulation model for surgical skills assessment. Obstet Gynecol. 2020;136:942-949.
- Vedula SS, Hager GD. Surgical data science: the new knowledge domain. Innov Surg Sci. 2017;2:109-121.
- Witthaus MW, Farooq S, Melnyk R, et al. Incorporation and validation of clinically relevant performance metrics of simulation (CRPMS) into a novel full-immersion simulation platform for nerve-sparing robot-assisted radical prostatectomy (NS-RARP) utilizing three-dimensional printing and hydrogel casting technology. BJU Int. 2020;125:322-332.
- Vedula SS, Malpani A, Ahmidi N, et al. Task-level vs segment-level quantitative metrics for surgical skill assessment. J Surg Educ. 2016;73:482-489.
- Maier-Hein L, Eisenmann M, Sarikaya D, et al. Surgical data science—from concepts toward clinical translation. Med Image Anal. 2022;76:102306.
- Hung AJ, Chen J, Gill IS. Automated performance metrics and machine learning algorithms to measure surgeon performance and anticipate clinical outcomes in robotic surgery. JAMA Surg. 2018;153:770-771.
- Altman K, Chen G, Chou B, et al. Surgical curriculum in obstetrics and gynecology: vaginal hysterectomy simulation. https://cfweb.acog. org/scog/scog008/Simulation.cfm.
- DeLancey JOL. Basic Exercises: Surgical Technique. Davis + Geck; Brooklyn, NY: 1987.
- Geoffrion R, Suen MW, Koenig NA, et al. Teaching vaginal surgery to junior residents: initial validation of 3 novel procedure-specific low-fidelity models. J Surg Educ. 2016;73:157-161.
- Pandey VA, Wolfe JHN, Lindhal AK, et al. Validity of an exam assessment in surgical skill: EBSQ-VASC pilot study. Eur J Vasc Endovasc Surg. 2004;27:341-348.
- Limbs&Things website. Knot Tying Trainer. https://limbsandthings. com/us/products/50050/50050-knot-tying-trainer. Accessed April 20, 2022.
- Vaughan MH, Kim-Fine S, Hullfish KL, et al. Validation of the simulated vaginal hysterectomy trainer. J Minim Invasive Gynecol. 2018;25:1101-1106.
- Braun K, Henley B, Ray C, et al. Teaching vaginal hysterectomy: low fidelity trainer provides effective simulation at low cost. Obstet Gynecol. 2017;130:44S.
- Anand M, Duffy CP, Vragovic O, et al. Surgical anatomy of vaginal hysterectomy—impact of a resident-constructed simulation model. Female Pelvic Med Reconstr Surg. 2018;24:176-182.
- Chen CC, Vaccaro CM. ACOG Simulation Consortium Surgical Curriculum: anterior and posterior repair. 2017. https://cfweb.acog. org/scog/.
- Schmidt PC, Fairchild PS, Fenner DE, et al. The Fundamentals of Vaginal Surgery pilot study: developing, validating, and setting proficiency scores for a vaginal surgical skills simulation system. Am J Obstet Gynecol. 2021;225:558.e1-558.e11.
Vaginal surgery, including vaginal hysterectomy, is slowly becoming a dying art. According to the National Inpatient Sample and the Nationwide Ambulatory Surgery Sample from 2018, only 11.8% of all hysterectomies were performed vaginally.1 The combination of uterine-sparing surgeries, advances in conservative therapies for benign uterine conditions, and the diversification of minimally invasive routes (laparoscopic and robotic) has resulted in a continued downtrend in vaginal surgical volumes. This shift has led to fewer operative learning opportunities and declining graduating resident surgical volume.2 According to the Accreditation Council for Graduate Medical Education (ACGME), the minimum number of vaginal hysterectomies is 15, which represents only the minimum accepted exposure and does not imply competency.
In response, surgical simulation has been used for skill acquisition and maintenance outside of the operating room in a learning environment that is safe for the learners and does not expose patients to additional risk. Educators are uniquely poised to use simulation to teach residents and to evaluate their procedural competency. Although vaginal surgery, specifically vaginal hysterectomy, continues to decline, it can be resuscitated with the assistance of surgical simulation.
In this article, we provide a broad overview of vaginal surgical simulation. We discuss the basic tenets of simulation, review how to teach and evaluate vaginal surgical skills, and present some of the commonly available vaginal surgery simulation models and their associated resources, cost, setup time, fidelity, and limitations.
Simulation principles relevant for vaginal hysterectomy simulation
Here, we review simulation-based learning principles that will help place specific simulation models into perspective.
One size does not fit all
Simulation, like many educational interventions, does not work via a “one-size-fits-all” approach. While the American College of Obstetricians and Gynecologists (ACOG) Simulations Working Group (SWG) has created a toolkit (available online at https://www.acog.org/education-and-events/simulations/about/curriculum) with many ready-to-use how-to simulation descriptions and lesson plans that cover common topics, what works in one setting may not work in another. The SWG created those modules to help educators save time and resources and to avoid reinventing the wheel for each simulation session. However, these simulations need to be adapted to the local needs of trainees and resources, such as faculty time, space, models, and funding.
Cost vs fidelity
It is important to distinguish between cost and fidelity. “Low cost” is often incorrectly used interchangeably with “low fidelity” when referring to models and simulations. The most basic principle of fidelity is that it is associated with situational realism that in turn, drives learning.3,4 For example, the term high fidelity does apply to a virtual reality robotic surgery simulator, which also is high cost. However, a low-cost beef tongue model of fourth-degree laceration5 is high fidelity, while more expensive commercial models are less realistic, which makes them high cost and low fidelity.6 When selecting simulation models, educators need to consider cost based on their available resources and the level of fidelity needed for their learners.
Continue to: Task breakdown...
Task breakdown
As surgeon-educators, we love to teach! And while educators are passionate about imparting vaginal hysterectomy skills to the next generation of surgeons, it is important to assess where the learners are technically. Vaginal hysterectomy is a high-complexity procedure, with each step involving a unique skill set that is new to residents as learners; this is where the science of learning can help us teach more effectively.7 Focusing on doing the entire procedure all at once is more likely to result in cognitive overload, while a better approach is to break the procedure down into several components and practice those parts until goal proficiency is reached.
Deliberate practice
The idea of deliberate practice was popularized by Malcolm Gladwell in his book titled Outliers, in which he gives examples of how 10,000 hours of practice leads to mastery of complex skills. This concept was deepened by the work of cognitive psychologist Anders Ericsson, who emphasized that not only the duration but also the quality of practice—which involves concentration, analysis, and problem-solving—leads to the most effective training.8
In surgical education, this concept translates into many domains. For example, an individualized learning plan includes frequent low-stakes assessments, video recording for later viewing and analysis, surgical coaching, and detailed planning of future training sessions to incorporate past performance. “Just doing” surgery on a simulator (or in the operating room) results in missed learning opportunities.
Logistics and implementation: Who, where, when
The simulation “formula” takes into account multiple factors but should start with learning objectives and then an assessment of what resources are available to address them. For example, if one surgeon-educator and one resident-learner are available for 30 minutes in between cases in the operating room, and the goal is to teach the resident clamp-and-tie technique on pedicles, the “milk carton” model9 and a few instruments from the vaginal hysterectomy tray are ideal for this training. On the other hand, if it is important to achieve competency for an entire procedure prior to operating room debut and a group of surgeon-educators is available to share the time commitment of 2-hour sessions per each resident, then the PROMPT (PRactical Obstetric Multi-Professional Training) shoulder dystocia model could be used (TABLE).10-14

Learning curves
Ideally, educators would like to know how many simulated training sessions are needed for a learner to reach a proficiency level and become operating room ready. Such information about learning curves, unfortunately, is not available yet for vaginal hysterectomies. The first step in the process is to establish a baseline for performance to know a starting point, with assessment tools specific to each simulator; the next step is to study how many “takes” are needed for learners to move through their learning curve.15 The use of assessment tools can help assess each learner’s progression.
Continue to: Evaluation, assessment, and feedback...
Evaluation, assessment, and feedback
With more emphasis being placed on patient safety and transparency in every aspect of health care, including surgical training, graduate medical education leaders increasingly highlight the importance of objective assessment tools and outcome-based standards for certification of competency in surgery.16,17 Commonly used assessment tools that have reliability and validity evidence include surgical checklists and global rating scales. Checklists for common gynecologic procedures, including vaginal hysterectomy, as well as a global rating scale specifically developed for vaginal surgery (Vaginal Surgical Skills Index, VSSI)18 are accessible on the ACOG Simulations Working Group Surgical Curriculum in Obstetrics and Gynecology website.19
While checklists contain the main steps of each procedure, these lists do not assess for how well each step of the procedure is performed. By contrast, global rating scales, such as the VSSI, can discriminate between surgeons with different skill levels both in the simulation and operating room settings; each metric within the global rating scale (for example, time and motion) does not pertain to the performance of a procedure’s specific step but rather to the overall performance of the entire procedure.18,20 Hence, to provide detailed feedback, especially for formative assessment, both checklists and global rating scales often are used together.
Although standardized, checklists and global rating scales ultimately are still subjective (not objective) assessment tools. Recently, more attention has been to use surgical data science, particularly artificial intelligence methods, to objectively assess surgical performance by analyzing data generated during the performance of surgery, such as instrumental motion and video.21 These methods have been applied to a wide range of surgical techniques, including open, laparoscopic, robotic, endoscopic, and microsurgical approaches. Most of these types of studies have used assessment of surgical skill as the main outcome, with fewer studies correlating skill with clinically relevant metrics, such as patient outcomes.22-25 Although this is an area of active research, these methods are still being developed, and their validity and utility are not well established. For now, educators should continue to use validated checklists and global rating skills to help assess any type of surgical performance, particularly vaginal surgery.
Vaginal surgical simulation models
Vaginal surgery requires a surgeon to operate in a narrow, deep space. This requires ambidexterity, accurate depth perception, understanding of how to handle tissues, and use of movements that are efficient, fluid, and rhythmic. Multiple proposed simulation models are relevant to vaginal surgery, and these vary based on level of fidelity, cost, feasibility, ability to maintain standardization, ease of construction (if required), and generalizability to all of pelvic surgery (that is, procedure specific vs basic skills focused).10,11,13,26-31
Below, we describe various simulation models that are available for teaching vaginal surgical skills.
Vaginal hysterectomy simulation model
One commercially available simulation model for vaginal hysterectomy (as well as other vaginal surgical procedures, such as midurethral sling and anterior and posterior colporrhaphy) is the Miya Model (Miyazaki Enterprises) (FIGURE 1) and its accompanying MiyaMODEL App. In a multi-institutional study funded by the National Institutes of Health (NIH), the Miya Model, when used with the VSSI, was shown to be a valid assessment tool in terms of ability to differentiate a competent from a noncompetent surgeon.20 Currently, an ongoing NIH-sponsored multi-institutional study is assessing the Miya Model as a teaching tool and whether skills acquired on the Miya Model are transferable to the operating room.

Continue to: Low-cost vaginal hysterectomy models...
Low-cost vaginal hysterectomy models
Multiple low-cost vaginal hysterectomy simulation models are described. Two models developed many years ago include the ACOG SWG flowerpot model14 and the PROMPT shoulder dystocia pelvic trainer model.10,11,14 The former model is low cost as it can be constructed from easily obtained household materials, but its downside is that it takes time and effort to obtain the materials and to assemble them. The latter model is faster to assemble but requires one to use a PROMPT pelvis for shoulder dystocia training, which has a considerable upfront cost. However, it is available in most hospitals with considerable obstetrical volume, and it allows for the most realistic perineum, which is helpful in recreating the feel of vaginal surgery, including retraction and exposure.
Many models created and described in the literature are variations of the models mentioned above, and many use commercially available low-cost bony pelvis models and polyvinyl chloride (PVC) pipes as a foundation for the soft tissue inserts to attach.12,13,31-33 Each model varies on what it “teaches best” regarding realism—for example, teaching anatomy, working in a tight space, dissection, or clamp placement and suture ligature.
Furthermore, since vaginal hysterectomy is a high-complexity procedure in terms of skills (working in confined space, limited view, “upside-down” anatomy, and need to direct assistants for retraction and exposure), task breakdown is important for simulation learning, as it is not efficient to repeat the entire procedure until proficiency is reached. Two trainers have been described to address that need: the milk carton and the vaginal suturing trainer. The latter allows learners to practice clamp placement and pedicle ligation multiple times, including in confined space (FIGURE 2), and the former allows them to do the same in a procedural matter as the clamp placement moves caudad to cephalad during the procedure (FIGURE 3).


Native tissue pelvic floor surgery simulation
While there are few publications regarding surgical simulation models for native tissue pelvic floor surgeries, a low-cost anterior and posterior repair model was developed for the ACOG SWG Simulation Toolkit and published online in 2017, after their peer-review process. The fidelity is moderate for this low-cost model, which costs less than $5 per use. The simulation model requires a new vaginal insert for each learner, which is fast and easy to make and requires only a few components; however, the bony pelvis (for example, the flowerpot model) needs to be purchased or created. The stage of the anterior wall prolapse can be adjusted by the amount of fluid placed in the balloon, which is used to simulate the bladder. The more fluid that is placed in the “bladder,” the more severe the anterior wall prolapse appears. The vaginal caliber can be adjusted, if needed, by increasing or decreasing the size of the components to create the vagina, but the suggested sizes simulate a significantly widened vaginal caliber that would benefit from a posterior repair with perineorrhaphy. Although there is no validity evidence for this model, a skills assessment is available through the ACOG Simulation Surgical Curriculum. Of note, native tissue colpopexy repairs are also possible with this model (or another high-fidelity model, such as the Miya Model), if the sacrospinous ligaments and/or uterosacral ligaments are available on the pelvic model in use. This model’s limitations include the absence of a high-fidelity plane of dissection of the vaginal muscularis, and that no bleeding is encountered, which is the case for many low-cost models.19,34
Fundamentals of Vaginal Surgery (FVS) basic surgical skills simulation
The FVS simulation system, consisting of a task trainer paired with 6 selected surgical tasks, was developed to teach basic skills used in vaginal surgery.35 The FVS task trainer is 3D printed and has 3 main components: a base piece that allows for different surgical materials to be secured, a depth extender, and a width reducer. In addition, it has a mobile phone mount and a window into the system to enable video capture of skills exercises.
The FVS simulator is designed to enable 6 surgical tasks, including one-handed knot tying, two-handed knot tying, running suturing, plication suturing, Heaney transfixion pedicle ligation, and free pedicle ligation (FIGURE 4). In a pilot study, the FVS simulation system was deemed representative of the intended surgical field, useful for inclusion in a training program, and favored as a tool for both training and testing. Additionally, an initial proficiency score of 400 was set, which discriminated between novice and expert surgeons.35
An advantage of this simulation system is that it allows learners to focus on basic skills, rather than on an entire specific procedure. Further, the system is standardized, as it is commercially manufactured; this also allows for easy assembly. The disadvantage of this model is that it cannot be modified to teach specific vaginal procedures, and it must be purchased, rather than constructed on site. Further studies are needed to create generalizable proficiency scores and to assess its use in training and testing. For more information on the FVS simulation model, visit the Arbor Simulation website (http://arborsim.com).
Surgical simulation’s important role
Surgical skills can be learned and improved in the simulation setting in a low-stakes, low-pressure environment. Simulation can enable basic skills development and then higher-level learning of complex procedures. Skill assessment is important to aid in learning (via formative assessments) and for examination or certification (summative assessments).
With decreasing vaginal surgical volumes occurring nationally, it is becoming even more important to use surgical simulation to teach and maintain vaginal surgical skills. In this article, we reviewed various different simulation models that can be used for developing vaginal surgical skills and presented the advantages, limitations, and resources relevant for each simulation model. ●
Vaginal surgery, including vaginal hysterectomy, is slowly becoming a dying art. According to the National Inpatient Sample and the Nationwide Ambulatory Surgery Sample from 2018, only 11.8% of all hysterectomies were performed vaginally.1 The combination of uterine-sparing surgeries, advances in conservative therapies for benign uterine conditions, and the diversification of minimally invasive routes (laparoscopic and robotic) has resulted in a continued downtrend in vaginal surgical volumes. This shift has led to fewer operative learning opportunities and declining graduating resident surgical volume.2 According to the Accreditation Council for Graduate Medical Education (ACGME), the minimum number of vaginal hysterectomies is 15, which represents only the minimum accepted exposure and does not imply competency.
In response, surgical simulation has been used for skill acquisition and maintenance outside of the operating room in a learning environment that is safe for the learners and does not expose patients to additional risk. Educators are uniquely poised to use simulation to teach residents and to evaluate their procedural competency. Although vaginal surgery, specifically vaginal hysterectomy, continues to decline, it can be resuscitated with the assistance of surgical simulation.
In this article, we provide a broad overview of vaginal surgical simulation. We discuss the basic tenets of simulation, review how to teach and evaluate vaginal surgical skills, and present some of the commonly available vaginal surgery simulation models and their associated resources, cost, setup time, fidelity, and limitations.
Simulation principles relevant for vaginal hysterectomy simulation
Here, we review simulation-based learning principles that will help place specific simulation models into perspective.
One size does not fit all
Simulation, like many educational interventions, does not work via a “one-size-fits-all” approach. While the American College of Obstetricians and Gynecologists (ACOG) Simulations Working Group (SWG) has created a toolkit (available online at https://www.acog.org/education-and-events/simulations/about/curriculum) with many ready-to-use how-to simulation descriptions and lesson plans that cover common topics, what works in one setting may not work in another. The SWG created those modules to help educators save time and resources and to avoid reinventing the wheel for each simulation session. However, these simulations need to be adapted to the local needs of trainees and resources, such as faculty time, space, models, and funding.
Cost vs fidelity
It is important to distinguish between cost and fidelity. “Low cost” is often incorrectly used interchangeably with “low fidelity” when referring to models and simulations. The most basic principle of fidelity is that it is associated with situational realism that in turn, drives learning.3,4 For example, the term high fidelity does apply to a virtual reality robotic surgery simulator, which also is high cost. However, a low-cost beef tongue model of fourth-degree laceration5 is high fidelity, while more expensive commercial models are less realistic, which makes them high cost and low fidelity.6 When selecting simulation models, educators need to consider cost based on their available resources and the level of fidelity needed for their learners.
Continue to: Task breakdown...
Task breakdown
As surgeon-educators, we love to teach! And while educators are passionate about imparting vaginal hysterectomy skills to the next generation of surgeons, it is important to assess where the learners are technically. Vaginal hysterectomy is a high-complexity procedure, with each step involving a unique skill set that is new to residents as learners; this is where the science of learning can help us teach more effectively.7 Focusing on doing the entire procedure all at once is more likely to result in cognitive overload, while a better approach is to break the procedure down into several components and practice those parts until goal proficiency is reached.
Deliberate practice
The idea of deliberate practice was popularized by Malcolm Gladwell in his book titled Outliers, in which he gives examples of how 10,000 hours of practice leads to mastery of complex skills. This concept was deepened by the work of cognitive psychologist Anders Ericsson, who emphasized that not only the duration but also the quality of practice—which involves concentration, analysis, and problem-solving—leads to the most effective training.8
In surgical education, this concept translates into many domains. For example, an individualized learning plan includes frequent low-stakes assessments, video recording for later viewing and analysis, surgical coaching, and detailed planning of future training sessions to incorporate past performance. “Just doing” surgery on a simulator (or in the operating room) results in missed learning opportunities.
Logistics and implementation: Who, where, when
The simulation “formula” takes into account multiple factors but should start with learning objectives and then an assessment of what resources are available to address them. For example, if one surgeon-educator and one resident-learner are available for 30 minutes in between cases in the operating room, and the goal is to teach the resident clamp-and-tie technique on pedicles, the “milk carton” model9 and a few instruments from the vaginal hysterectomy tray are ideal for this training. On the other hand, if it is important to achieve competency for an entire procedure prior to operating room debut and a group of surgeon-educators is available to share the time commitment of 2-hour sessions per each resident, then the PROMPT (PRactical Obstetric Multi-Professional Training) shoulder dystocia model could be used (TABLE).10-14

Learning curves
Ideally, educators would like to know how many simulated training sessions are needed for a learner to reach a proficiency level and become operating room ready. Such information about learning curves, unfortunately, is not available yet for vaginal hysterectomies. The first step in the process is to establish a baseline for performance to know a starting point, with assessment tools specific to each simulator; the next step is to study how many “takes” are needed for learners to move through their learning curve.15 The use of assessment tools can help assess each learner’s progression.
Continue to: Evaluation, assessment, and feedback...
Evaluation, assessment, and feedback
With more emphasis being placed on patient safety and transparency in every aspect of health care, including surgical training, graduate medical education leaders increasingly highlight the importance of objective assessment tools and outcome-based standards for certification of competency in surgery.16,17 Commonly used assessment tools that have reliability and validity evidence include surgical checklists and global rating scales. Checklists for common gynecologic procedures, including vaginal hysterectomy, as well as a global rating scale specifically developed for vaginal surgery (Vaginal Surgical Skills Index, VSSI)18 are accessible on the ACOG Simulations Working Group Surgical Curriculum in Obstetrics and Gynecology website.19
While checklists contain the main steps of each procedure, these lists do not assess for how well each step of the procedure is performed. By contrast, global rating scales, such as the VSSI, can discriminate between surgeons with different skill levels both in the simulation and operating room settings; each metric within the global rating scale (for example, time and motion) does not pertain to the performance of a procedure’s specific step but rather to the overall performance of the entire procedure.18,20 Hence, to provide detailed feedback, especially for formative assessment, both checklists and global rating scales often are used together.
Although standardized, checklists and global rating scales ultimately are still subjective (not objective) assessment tools. Recently, more attention has been to use surgical data science, particularly artificial intelligence methods, to objectively assess surgical performance by analyzing data generated during the performance of surgery, such as instrumental motion and video.21 These methods have been applied to a wide range of surgical techniques, including open, laparoscopic, robotic, endoscopic, and microsurgical approaches. Most of these types of studies have used assessment of surgical skill as the main outcome, with fewer studies correlating skill with clinically relevant metrics, such as patient outcomes.22-25 Although this is an area of active research, these methods are still being developed, and their validity and utility are not well established. For now, educators should continue to use validated checklists and global rating skills to help assess any type of surgical performance, particularly vaginal surgery.
Vaginal surgical simulation models
Vaginal surgery requires a surgeon to operate in a narrow, deep space. This requires ambidexterity, accurate depth perception, understanding of how to handle tissues, and use of movements that are efficient, fluid, and rhythmic. Multiple proposed simulation models are relevant to vaginal surgery, and these vary based on level of fidelity, cost, feasibility, ability to maintain standardization, ease of construction (if required), and generalizability to all of pelvic surgery (that is, procedure specific vs basic skills focused).10,11,13,26-31
Below, we describe various simulation models that are available for teaching vaginal surgical skills.
Vaginal hysterectomy simulation model
One commercially available simulation model for vaginal hysterectomy (as well as other vaginal surgical procedures, such as midurethral sling and anterior and posterior colporrhaphy) is the Miya Model (Miyazaki Enterprises) (FIGURE 1) and its accompanying MiyaMODEL App. In a multi-institutional study funded by the National Institutes of Health (NIH), the Miya Model, when used with the VSSI, was shown to be a valid assessment tool in terms of ability to differentiate a competent from a noncompetent surgeon.20 Currently, an ongoing NIH-sponsored multi-institutional study is assessing the Miya Model as a teaching tool and whether skills acquired on the Miya Model are transferable to the operating room.

Continue to: Low-cost vaginal hysterectomy models...
Low-cost vaginal hysterectomy models
Multiple low-cost vaginal hysterectomy simulation models are described. Two models developed many years ago include the ACOG SWG flowerpot model14 and the PROMPT shoulder dystocia pelvic trainer model.10,11,14 The former model is low cost as it can be constructed from easily obtained household materials, but its downside is that it takes time and effort to obtain the materials and to assemble them. The latter model is faster to assemble but requires one to use a PROMPT pelvis for shoulder dystocia training, which has a considerable upfront cost. However, it is available in most hospitals with considerable obstetrical volume, and it allows for the most realistic perineum, which is helpful in recreating the feel of vaginal surgery, including retraction and exposure.
Many models created and described in the literature are variations of the models mentioned above, and many use commercially available low-cost bony pelvis models and polyvinyl chloride (PVC) pipes as a foundation for the soft tissue inserts to attach.12,13,31-33 Each model varies on what it “teaches best” regarding realism—for example, teaching anatomy, working in a tight space, dissection, or clamp placement and suture ligature.
Furthermore, since vaginal hysterectomy is a high-complexity procedure in terms of skills (working in confined space, limited view, “upside-down” anatomy, and need to direct assistants for retraction and exposure), task breakdown is important for simulation learning, as it is not efficient to repeat the entire procedure until proficiency is reached. Two trainers have been described to address that need: the milk carton and the vaginal suturing trainer. The latter allows learners to practice clamp placement and pedicle ligation multiple times, including in confined space (FIGURE 2), and the former allows them to do the same in a procedural matter as the clamp placement moves caudad to cephalad during the procedure (FIGURE 3).


Native tissue pelvic floor surgery simulation
While there are few publications regarding surgical simulation models for native tissue pelvic floor surgeries, a low-cost anterior and posterior repair model was developed for the ACOG SWG Simulation Toolkit and published online in 2017, after their peer-review process. The fidelity is moderate for this low-cost model, which costs less than $5 per use. The simulation model requires a new vaginal insert for each learner, which is fast and easy to make and requires only a few components; however, the bony pelvis (for example, the flowerpot model) needs to be purchased or created. The stage of the anterior wall prolapse can be adjusted by the amount of fluid placed in the balloon, which is used to simulate the bladder. The more fluid that is placed in the “bladder,” the more severe the anterior wall prolapse appears. The vaginal caliber can be adjusted, if needed, by increasing or decreasing the size of the components to create the vagina, but the suggested sizes simulate a significantly widened vaginal caliber that would benefit from a posterior repair with perineorrhaphy. Although there is no validity evidence for this model, a skills assessment is available through the ACOG Simulation Surgical Curriculum. Of note, native tissue colpopexy repairs are also possible with this model (or another high-fidelity model, such as the Miya Model), if the sacrospinous ligaments and/or uterosacral ligaments are available on the pelvic model in use. This model’s limitations include the absence of a high-fidelity plane of dissection of the vaginal muscularis, and that no bleeding is encountered, which is the case for many low-cost models.19,34
Fundamentals of Vaginal Surgery (FVS) basic surgical skills simulation
The FVS simulation system, consisting of a task trainer paired with 6 selected surgical tasks, was developed to teach basic skills used in vaginal surgery.35 The FVS task trainer is 3D printed and has 3 main components: a base piece that allows for different surgical materials to be secured, a depth extender, and a width reducer. In addition, it has a mobile phone mount and a window into the system to enable video capture of skills exercises.
The FVS simulator is designed to enable 6 surgical tasks, including one-handed knot tying, two-handed knot tying, running suturing, plication suturing, Heaney transfixion pedicle ligation, and free pedicle ligation (FIGURE 4). In a pilot study, the FVS simulation system was deemed representative of the intended surgical field, useful for inclusion in a training program, and favored as a tool for both training and testing. Additionally, an initial proficiency score of 400 was set, which discriminated between novice and expert surgeons.35
An advantage of this simulation system is that it allows learners to focus on basic skills, rather than on an entire specific procedure. Further, the system is standardized, as it is commercially manufactured; this also allows for easy assembly. The disadvantage of this model is that it cannot be modified to teach specific vaginal procedures, and it must be purchased, rather than constructed on site. Further studies are needed to create generalizable proficiency scores and to assess its use in training and testing. For more information on the FVS simulation model, visit the Arbor Simulation website (http://arborsim.com).
Surgical simulation’s important role
Surgical skills can be learned and improved in the simulation setting in a low-stakes, low-pressure environment. Simulation can enable basic skills development and then higher-level learning of complex procedures. Skill assessment is important to aid in learning (via formative assessments) and for examination or certification (summative assessments).
With decreasing vaginal surgical volumes occurring nationally, it is becoming even more important to use surgical simulation to teach and maintain vaginal surgical skills. In this article, we reviewed various different simulation models that can be used for developing vaginal surgical skills and presented the advantages, limitations, and resources relevant for each simulation model. ●
- Wright JD, Huang Y, Li AH, et al. Nationwide estimates of annual inpatient and outpatient hysterectomies performed in the United States. Obstet Gynecol. 2022;139:446-448.
- Gressel GM, Potts JR 3rd, Cha S, et al. Hysterectomy route and numbers reported by graduating residents in obstetrics and gynecology training programs. Obstet Gynecol. 2020;135:268-273.
- Lioce L, ed. Healthcare Simulation Dictionary. 2nd ed. Rockville, MD; Agency for Healthcare Research and Quality: 2020. AHRQ Publication No. 20-0019.
- Norman G, Dore K, Grierson L. The minimal relationship between simulation fidelity and transfer of learning. Med Educ. 2012;46:636-647.
- Illston JD, Ballard AC, Ellington DR, et al. Modified beef tongue model for fourth-degree laceration repair simulation. Obstet Gynecol. 2017;129:491-496.
- WorldPoint website. 3B Scientific Episiotomy and Suturing Trainer. https://www.worldpoint.com/3b-episiotomy-and-suturing-sim. Accessed April 20, 2022.
- Balafoutas D, Joukhadar R, Kiesel M, et al. The role of deconstructive teaching in the training of laparoscopy. JSLS. 2019;23:e2019.00020.
- Ericsson KA, Harwell KW. Deliberate practice and proposed limits on the effects of practice on the acquisition of expert performance: why the original definition matters and recommendations for future research. Front Psychol. 2019;10:2396.
- Smith TM, Fenner DE. Vaginal hysterectomy teaching model—an educational video. Female Pelvic Med Reconstr Surg. 2012;18:S43. Abstract.
- Greer JA, Segal S, Salva CR, et al. Development and validation of simulation training for vaginal hysterectomy. J Minim Invasive Gynecol. 2014;21:74-82.
- Malacarne DR, Escobar CM, Lam CJ, et al. Teaching vaginal hysterectomy via simulation: creation and validation of the objective skills assessment tool for simulated vaginal hysterectomy on a task trainer and performance among different levels of trainees. Female Pelvic Med Reconstr Surg. 2019;25:298-304.
- Zoorob D, Frenn R, Moffitt M, et al. Multi-institutional validation of a vaginal hysterectomy simulation model for resident training. J Minim Invasive Gynecol. 2021;28:1490-1496.e1.
- Barrier BF, Thompson AB, McCullough MW, et al. A novel and inexpensive vaginal hysterectomy simulator. Simul Healthc. 2012;7:374-379.
- Burkett LS, Makin J, Ackenbom M, et al. Validation of transvaginal hysterectomy surgical model—modification of the flowerpot model to improve vesicovaginal plane simulation. J Minim Invasive Gynecol. 2021;28:1526-1530.
- Escobar C, Malacarne Pape D, Ferrante KL, et al. Who should be teaching vaginal hysterectomy on a task trainer? A multicenter randomized trial of peer versus expert coaching. J Surg Simul. 2020;7:63-72.
- The obstetrics and gynecology milestone project. J Grad Med Educ. 2014;6(1 suppl 1):129-143.
- Nasca TJ, Philibert I, Brigham T, et al. The next GME accreditation system—rationale and benefits. N Engl J Med. 2012;366:1051-1056.
- Chen CCG, Korn A, Klingele C, et al. Objective assessment of vaginal surgical skills. Am J Obstet Gynecol. 2010;203:79.e1-8.
- American College of Obstetricians and Gynecologists. Surgical curriculum in obstetrics and gynecology. https://www.acog.org /education-and-events/simulations/surgical-curriculum-in-ob-gyn.
- Chen CCG, Lockrow EG, DeStephano CC, et al. Establishing validity for a vaginal hysterectomy simulation model for surgical skills assessment. Obstet Gynecol. 2020;136:942-949.
- Vedula SS, Hager GD. Surgical data science: the new knowledge domain. Innov Surg Sci. 2017;2:109-121.
- Witthaus MW, Farooq S, Melnyk R, et al. Incorporation and validation of clinically relevant performance metrics of simulation (CRPMS) into a novel full-immersion simulation platform for nerve-sparing robot-assisted radical prostatectomy (NS-RARP) utilizing three-dimensional printing and hydrogel casting technology. BJU Int. 2020;125:322-332.
- Vedula SS, Malpani A, Ahmidi N, et al. Task-level vs segment-level quantitative metrics for surgical skill assessment. J Surg Educ. 2016;73:482-489.
- Maier-Hein L, Eisenmann M, Sarikaya D, et al. Surgical data science—from concepts toward clinical translation. Med Image Anal. 2022;76:102306.
- Hung AJ, Chen J, Gill IS. Automated performance metrics and machine learning algorithms to measure surgeon performance and anticipate clinical outcomes in robotic surgery. JAMA Surg. 2018;153:770-771.
- Altman K, Chen G, Chou B, et al. Surgical curriculum in obstetrics and gynecology: vaginal hysterectomy simulation. https://cfweb.acog. org/scog/scog008/Simulation.cfm.
- DeLancey JOL. Basic Exercises: Surgical Technique. Davis + Geck; Brooklyn, NY: 1987.
- Geoffrion R, Suen MW, Koenig NA, et al. Teaching vaginal surgery to junior residents: initial validation of 3 novel procedure-specific low-fidelity models. J Surg Educ. 2016;73:157-161.
- Pandey VA, Wolfe JHN, Lindhal AK, et al. Validity of an exam assessment in surgical skill: EBSQ-VASC pilot study. Eur J Vasc Endovasc Surg. 2004;27:341-348.
- Limbs&Things website. Knot Tying Trainer. https://limbsandthings. com/us/products/50050/50050-knot-tying-trainer. Accessed April 20, 2022.
- Vaughan MH, Kim-Fine S, Hullfish KL, et al. Validation of the simulated vaginal hysterectomy trainer. J Minim Invasive Gynecol. 2018;25:1101-1106.
- Braun K, Henley B, Ray C, et al. Teaching vaginal hysterectomy: low fidelity trainer provides effective simulation at low cost. Obstet Gynecol. 2017;130:44S.
- Anand M, Duffy CP, Vragovic O, et al. Surgical anatomy of vaginal hysterectomy—impact of a resident-constructed simulation model. Female Pelvic Med Reconstr Surg. 2018;24:176-182.
- Chen CC, Vaccaro CM. ACOG Simulation Consortium Surgical Curriculum: anterior and posterior repair. 2017. https://cfweb.acog. org/scog/.
- Schmidt PC, Fairchild PS, Fenner DE, et al. The Fundamentals of Vaginal Surgery pilot study: developing, validating, and setting proficiency scores for a vaginal surgical skills simulation system. Am J Obstet Gynecol. 2021;225:558.e1-558.e11.
- Wright JD, Huang Y, Li AH, et al. Nationwide estimates of annual inpatient and outpatient hysterectomies performed in the United States. Obstet Gynecol. 2022;139:446-448.
- Gressel GM, Potts JR 3rd, Cha S, et al. Hysterectomy route and numbers reported by graduating residents in obstetrics and gynecology training programs. Obstet Gynecol. 2020;135:268-273.
- Lioce L, ed. Healthcare Simulation Dictionary. 2nd ed. Rockville, MD; Agency for Healthcare Research and Quality: 2020. AHRQ Publication No. 20-0019.
- Norman G, Dore K, Grierson L. The minimal relationship between simulation fidelity and transfer of learning. Med Educ. 2012;46:636-647.
- Illston JD, Ballard AC, Ellington DR, et al. Modified beef tongue model for fourth-degree laceration repair simulation. Obstet Gynecol. 2017;129:491-496.
- WorldPoint website. 3B Scientific Episiotomy and Suturing Trainer. https://www.worldpoint.com/3b-episiotomy-and-suturing-sim. Accessed April 20, 2022.
- Balafoutas D, Joukhadar R, Kiesel M, et al. The role of deconstructive teaching in the training of laparoscopy. JSLS. 2019;23:e2019.00020.
- Ericsson KA, Harwell KW. Deliberate practice and proposed limits on the effects of practice on the acquisition of expert performance: why the original definition matters and recommendations for future research. Front Psychol. 2019;10:2396.
- Smith TM, Fenner DE. Vaginal hysterectomy teaching model—an educational video. Female Pelvic Med Reconstr Surg. 2012;18:S43. Abstract.
- Greer JA, Segal S, Salva CR, et al. Development and validation of simulation training for vaginal hysterectomy. J Minim Invasive Gynecol. 2014;21:74-82.
- Malacarne DR, Escobar CM, Lam CJ, et al. Teaching vaginal hysterectomy via simulation: creation and validation of the objective skills assessment tool for simulated vaginal hysterectomy on a task trainer and performance among different levels of trainees. Female Pelvic Med Reconstr Surg. 2019;25:298-304.
- Zoorob D, Frenn R, Moffitt M, et al. Multi-institutional validation of a vaginal hysterectomy simulation model for resident training. J Minim Invasive Gynecol. 2021;28:1490-1496.e1.
- Barrier BF, Thompson AB, McCullough MW, et al. A novel and inexpensive vaginal hysterectomy simulator. Simul Healthc. 2012;7:374-379.
- Burkett LS, Makin J, Ackenbom M, et al. Validation of transvaginal hysterectomy surgical model—modification of the flowerpot model to improve vesicovaginal plane simulation. J Minim Invasive Gynecol. 2021;28:1526-1530.
- Escobar C, Malacarne Pape D, Ferrante KL, et al. Who should be teaching vaginal hysterectomy on a task trainer? A multicenter randomized trial of peer versus expert coaching. J Surg Simul. 2020;7:63-72.
- The obstetrics and gynecology milestone project. J Grad Med Educ. 2014;6(1 suppl 1):129-143.
- Nasca TJ, Philibert I, Brigham T, et al. The next GME accreditation system—rationale and benefits. N Engl J Med. 2012;366:1051-1056.
- Chen CCG, Korn A, Klingele C, et al. Objective assessment of vaginal surgical skills. Am J Obstet Gynecol. 2010;203:79.e1-8.
- American College of Obstetricians and Gynecologists. Surgical curriculum in obstetrics and gynecology. https://www.acog.org /education-and-events/simulations/surgical-curriculum-in-ob-gyn.
- Chen CCG, Lockrow EG, DeStephano CC, et al. Establishing validity for a vaginal hysterectomy simulation model for surgical skills assessment. Obstet Gynecol. 2020;136:942-949.
- Vedula SS, Hager GD. Surgical data science: the new knowledge domain. Innov Surg Sci. 2017;2:109-121.
- Witthaus MW, Farooq S, Melnyk R, et al. Incorporation and validation of clinically relevant performance metrics of simulation (CRPMS) into a novel full-immersion simulation platform for nerve-sparing robot-assisted radical prostatectomy (NS-RARP) utilizing three-dimensional printing and hydrogel casting technology. BJU Int. 2020;125:322-332.
- Vedula SS, Malpani A, Ahmidi N, et al. Task-level vs segment-level quantitative metrics for surgical skill assessment. J Surg Educ. 2016;73:482-489.
- Maier-Hein L, Eisenmann M, Sarikaya D, et al. Surgical data science—from concepts toward clinical translation. Med Image Anal. 2022;76:102306.
- Hung AJ, Chen J, Gill IS. Automated performance metrics and machine learning algorithms to measure surgeon performance and anticipate clinical outcomes in robotic surgery. JAMA Surg. 2018;153:770-771.
- Altman K, Chen G, Chou B, et al. Surgical curriculum in obstetrics and gynecology: vaginal hysterectomy simulation. https://cfweb.acog. org/scog/scog008/Simulation.cfm.
- DeLancey JOL. Basic Exercises: Surgical Technique. Davis + Geck; Brooklyn, NY: 1987.
- Geoffrion R, Suen MW, Koenig NA, et al. Teaching vaginal surgery to junior residents: initial validation of 3 novel procedure-specific low-fidelity models. J Surg Educ. 2016;73:157-161.
- Pandey VA, Wolfe JHN, Lindhal AK, et al. Validity of an exam assessment in surgical skill: EBSQ-VASC pilot study. Eur J Vasc Endovasc Surg. 2004;27:341-348.
- Limbs&Things website. Knot Tying Trainer. https://limbsandthings. com/us/products/50050/50050-knot-tying-trainer. Accessed April 20, 2022.
- Vaughan MH, Kim-Fine S, Hullfish KL, et al. Validation of the simulated vaginal hysterectomy trainer. J Minim Invasive Gynecol. 2018;25:1101-1106.
- Braun K, Henley B, Ray C, et al. Teaching vaginal hysterectomy: low fidelity trainer provides effective simulation at low cost. Obstet Gynecol. 2017;130:44S.
- Anand M, Duffy CP, Vragovic O, et al. Surgical anatomy of vaginal hysterectomy—impact of a resident-constructed simulation model. Female Pelvic Med Reconstr Surg. 2018;24:176-182.
- Chen CC, Vaccaro CM. ACOG Simulation Consortium Surgical Curriculum: anterior and posterior repair. 2017. https://cfweb.acog. org/scog/.
- Schmidt PC, Fairchild PS, Fenner DE, et al. The Fundamentals of Vaginal Surgery pilot study: developing, validating, and setting proficiency scores for a vaginal surgical skills simulation system. Am J Obstet Gynecol. 2021;225:558.e1-558.e11.
2022 Update on cervical disease
Cervical cancer is an important global health problem with an estimated 604,127 new cases and 341,831 deaths in 2020.1 Nearly 85% of the disease burden affects individuals from low and middle-income countries. The World Health Organization (WHO) set forth the goal for all countries to reach and maintain an incidence rate of below 4 per 100,000 women by 2030 as part of the Global Strategy to Accelerate the Elimination of Cervical Cancer.
Although traditional Pap cytology has been the cornerstone of screening programs, its poor sensitivity of approximately 50% and limitations in accessibility require new strategies to achieve the elimination of cervical cancer.2 The discovery that persistent infection with oncogenic human papillomavirus (HPV) is an essential step in the development of cervical cancer led to the development of diagnostic HPV tests, which have higher sensitivity than cytology (96.1% vs 53.0%) but somewhat lower specificity (90.7% vs 96.3%) for the detection of cervical intraepithelial neoplasia (CIN) 2 or worse lesions.2 Initially, HPV testing was incorporated as a method to triage atypical squamous cells of undetermined significance (ASCUS) cytology results.3 Later, the concept of cotesting with cytology emerged,4,5 and since then, several clinical trials have demonstrated the effectiveness of primary HPV screening.6-9
In 2020, the WHO recommended HPV DNA testing as the primary screening method starting at the age of 30 years, with regular testing every 5 to 10 years, for the general population.10 Currently, primary HPV has been adopted in multiple countries, including Australia, the Netherlands, Turkey, England, and Argentina.
In the United States, there are 3 currently acceptable screening strategies: cytology, cytology plus HPV (cotesting), and primary HPV testing (TABLE). The American Cancer Society (ACS) specifically states that HPV testing alone every 5 years is preferred starting at age 25 years; cotesting every 5 years or cytology alone every 3 years are also acceptable.11 The US Preventive Services Task Force (USPSTF) states that cytology alone every 3 years starting at 21 years and then HPV testing alone or cotesting every 5 years or cytology every 3 years starting at age 30 are all acceptable strategies.12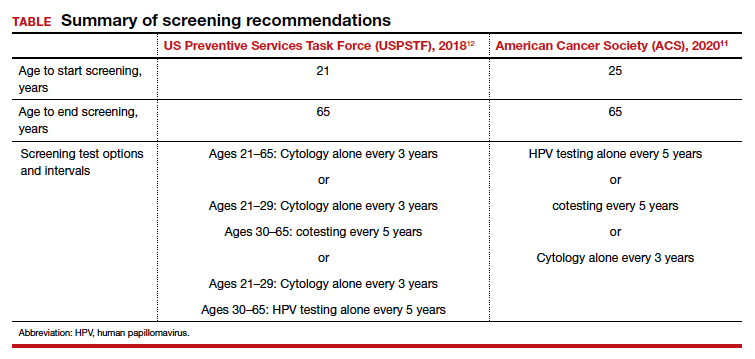
When applying these guidelines, it is important to note that they are intended for the screening of patients with all prior normal results with no symptoms. These routine screening guidelines do not apply to special populations, such as those with a history of abnormal results or treatment, a history of immunosuppression,13 a history of HPV-related vulvar or vaginal dysplasia,14-16 or a history of hysterectomy with removal of the cervix and no prior history of cervical dysplasia.17,18 By contrast, surveillance is interval testing for those who have either an abnormal prior test result or treatment; these may be managed per risk-based estimates provided by the American Society for Colposcopy and Cervical Pathology (ASCCP).18,19 Finally, diagnosis is evaluation (which may include diagnostic cytology) of a patient with abnormal signs and/or symptoms (such as bleeding, pain, discharge, or cervical mass).
In this Update, we present the evidence for primary HPV testing, the management options for a positive result in the United States, and research that will improve uptake of primary HPV testing as well as accessibility.
Change in screening paradigm: Evidence for primary HPV testing
HPV DNA tests are multiplex assays that detect the DNA of targeted high-risk HPV types, using multiple probes, either by direct genomic detection or by amplification of a viral DNA fragment using polymerase chain reaction (PCR).20,21 Alternatively, HPV mRNA-based tests detect the expression of E6 and E7 oncoproteins, a marker of viral integration.20 In examining the data from well-conducted clinical trials, 2 important observations are that different HPV assays were used and that direct comparison may not be valid. In addition, not all tests used in the studies are approved by the US Food and Drug Administration (FDA) for primary HPV testing.
Continue to: FDA-approved HPV tests...
FDA-approved HPV tests
Currently, 2 tests are FDA approved for primary HPV screening. The Cobas HPV test (Roche Molecular Diagnostics) was the first FDA-approved test for primary HPV screening in women aged 25 years and older.6 This test reports pooled results from 12 high-risk (hr) HPV types (31/33/35/39/45/51/52/56/58/59/66/68) with reflex genotyping for HPV 16/18, and thus it provides an immediate triage option for HPV-positive women. Of note, it is also approved for cotesting. The second FDA-approved test is the BD Onclarity HPV assay (Becton, Dickinson and Company) for primary HPV screening.22 It detects 14 hrHPV types, types 16/18/45 specifically as well as types 31/33/35/39/51/52/56/58/59/66/68.
Other HPV tests are FDA approved for cotesting and reflex testing but not for primary HPV testing. The Hybrid Capture test, or HC2 (Qiagen Inc), was the first HPV test to be approved by the FDA in 1997 for reflex testing of women with ASCUS cytology. In 2003, it was approved for cotesting along with cytology in women aged 30 years and older.20,21 In 2009, the Cervista HPV HR test (Hologic Inc) was approved for cotesting. The Aptima HPV assay (Hologic Inc), which is also approved for cotesting, is an RNA-based assay that allows detection of E6/E7 mRNA transcripts of 14 HPV types.23
Comparing HPV testing with cytology
Ronco and colleagues pooled data from 4 European randomized controlled trials (RCTs)—Swedescreen, POBASCAM, NTCC, ARTISTIC—with a total of 176,464 participants randomly assigned to HPV or cytology screening.24 Swedescreen and POBASCAM used GP5/GP6 PCR, while ARTISTIC and NTCC used HC2 for primary HPV screening. The screening interval was 3 years in all except 5 years in POBASCAM. The pooled detection rate of invasive disease was similar in the 2 arms, with pooled rate ratio for cancer detection being 0.79 (95% confidence interval [CI], 0.46–1.36) in the first 2.5 years, but was 0.45 (95% CI, 0.25–0.81), favoring the HPV arm, after 2.5 years. HPV testing was more effective in preventing cases of adenocarcinoma than squamous cell carcinoma (0.31 [95% CI, 0.14–0.69] vs 0.78 [95% CI, 0.49–1.25]). The authors concluded that HPV-based screening from age 30 years provided 60% to 70% better protection than cytology.
The result of the above meta-analysis was confirmed by the HPV FOCAL RCT that investigated the efficacy of HPV testing (HC2) in comparison with cytology.25 The detection rates for CIN 3 lesions supported primary HPV screening, with an absolute difference in incidence rate of 2.67/1,000 (95% CI, 0.53–4.88) at study randomization and 3.22/1,000 (95% CI, 5.12–1.48) at study exit 4 years later.
Cotesting using HPV and cytology: Marginal benefit
Dillner and colleagues were one of the first groups to report on the risk of CIN 3 based on both HPV and cytology status.26 Using pooled analysis of data from multiple countries, these investigators reported that the cumulative incidence rates (CIR) of CIN 3 after 6 years of follow-up increased consistently in HPV-positive subjects, and an HPV-positive result more accurately predicted CIN 3+ at 5 years than cytology alone. Furthermore, HPV negativity provided greater reassurance than cytology alone. At 5 years of follow-up, the rates of CIN 3+ were 0.25% (0.12%–0.41%) for women negative for HPV compared with 0.83% (0.50%–1.13%) for women with negative cytology results. There was little difference in rates for CIN 3+ between women with negative results on both tests and women who were negative for HPV.
The important question is then the marginal benefit of cotesting, which is the most costly screening option. A study of 331,818 women enrolled for cotesting at Kaiser Permanente found that the risk of CIN 3+ predicted by HPV testing alone when compared with cytology was significantly higher at both 3 years (5.0% vs 3.8%; P = .046) and 5 years (7.6% vs 4.7%; P = .001).27 A negative cytology result did not decrease the risk of CIN 3 further for HPV-negative patients (3 years: 0.047% vs 0.063%, P = .6; 5 years: 0.16% vs 0.17%, P = .8). They concluded that a negative HPV test was enough reassurance for low risk of CIN 3+ and that an additional negative cytology result does not provide extra reassurance.
Furthermore, a systematic meta-analysis of 48 studies, including 8 RCTs, found that the addition of cytology to HPV testing raised the sensitivity by 2% for CIN 3 compared with HPV testing alone. This improvement in sensitivity was at the expense of considerable loss of specificity, with a ratio of 0.93 (95% CI, 0.92–0.95) for CIN 3.28 Schiffman and colleagues also assessed the relative contribution of HPV testing and cytology in detection of CIN 3 and cancer.29 The HPV component alone identified a significantly higher proportion of preinvasive and invasive disease than cytology. Only 3.5% of precancers and 5.9% of cancers were preceded by HPV-negative, cytology-positive results. Thus, cytology contributed only 5 cases per million women per year to the sensitivity of the combined test, at the cost of significantly more colposcopies. Hence, the evidence suggests that there is limited benefit of adding cytology to HPV testing.30
Continue to: Triage of a positive HPV result...
Triage of a positive HPV result
An important limitation of HPV testing is its inability to discriminate between transient and persistent infections. Referral of all HPV-positive cases to colposcopy would overburden the system with associated unnecessary procedures. Hence, a triage strategy is essential to identify clinically important infections that truly require colposcopic evaluation. The FIGURE illustrates the management of a primary HPV test result performed for screening.
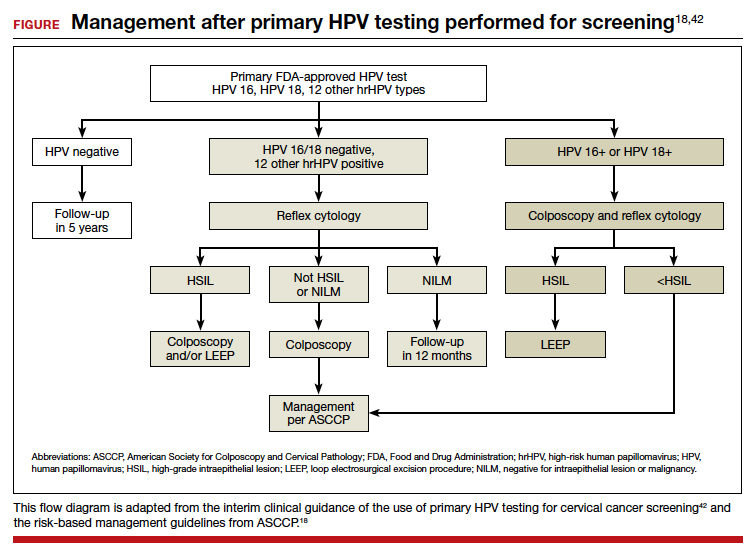
HPV genotyping
One strategy for triaging a positive HPV test result is genotyping. HPV 16 and 18 have the highest risk of persistence and progression and merit immediate referral to colposcopy. In the ATHENA trial, CIN 3 was identified in 17.8% (95% CI, 14.8–20.7%) of HPV 16 positive women at baseline, and the CIR increased to 25.2% (95% CI, 21.7–28.7%) after 3 years. The 3-year CIR of CIN 3 was only 5.4% (95% CI, 4.5–6.3%) in women with HPV genotypes other than 16/18. HPV 18–positive women had a 3-year CIR that was intermediate between women with HPV 16 and women with the 12 other genotypes.6 Hence, HPV 16/18–positive cases should be referred for immediate colposcopy, and negative cases should be followed up with cytology and referred for colposcopy if the cytology is ASCUS or worse.31
In July 2020, extended genotyping was approved by the FDA with individual detection of HPV 31, 51, 52 (in addition to 16, 18, and 45) and pooled detection of 33/58, 35/39/68, and 56/59/66. One study found that individual genotypes HPV 16 and 31 carry baseline risk values for CIN 3+ (8.1% and 7.5%, respectively) that are above the 5-year risk threshold for referral to colposcopy following the ASCCP risk-based management guideline.32
Cytology
The higher specificity of cytology makes it an option for triaging HPV-positive cases, and current management guidelines recommend triage to both genotyping and cytology for all patients who are HPV positive, and especially if they are HPV positive but HPV 16/18 negative. Of note, cytology results remain more subjective than those of primary HPV testing, but the combination of initial HPV testing with reflex to cytology is a reasonable and cost effective next step.18 The VASCAR trial found higher colposcopy referrals in the HPV screening and cytology triage group compared with the cytology alone group (19.36 vs 14.54 per 1,000 women).33 The ATHENA trial investigated various triage strategies for HPV-positive cases and its impact on colposcopy referrals.6 Using HPV genotyping and reflex cytology, if HPV 16/18 was positive, colposcopy was advised, but if any of the other 12 HPV types were positive, reflex cytology was done. If reported as ASCUS or worse, colposcopy was performed; conversely, if it was normal, women were rescreened with cotesting after 1 year. Although this strategy led to a reduction in the number of colposcopies, referrals were still higher in the primary HPV arm (3,769 colposcopies per 294 cases) compared with cytology (1,934 colposcopies per 179 cases) or cotesting (3,097 colposcopies per 240 cases) in women aged 25 years.14
p16/Ki-67 Dual-Stain
Diffused p16 immunohistochemical staining, as opposed to focal staining, is associated with active HPV infection but can be present in low-grade as well as high-grade lesions.34 Ki-67 is a marker of cellular proliferation. Coexpression of p16 and Ki-67 indicates a loss of cell cycle regulation and is a hallmark of neoplastic transformation. When positive, these tests are supportive of active HPV infection and of a high-grade lesion. Incorporation of these stains to cytology alone provides additional objective reassurance to cytology, where there is much inter- and intra-observer variability. These stains can be done by laboratories using the stains alone or they can use the FDA-approved p16/Ki-67 Dual-Stain immunohistochemistry (DS), CINtec PLUS Cytology (Roche Diagnostics). However, DS is not yet formally incorporated into triage algorithms by national guidelines.
The IMPACT trial assessed the performance of DS compared with cytology in the triage of HPV-positive results, with or without HPV 16/18 genotyping.35 This was a prospective observational screening study of 35,263 women aged 25 to 65 years across 32 sites in the United States. Of the 4,927 HPV-positive patients with DS results, the sensitivity of DS for CIN 3+ was 91.9% (95% CI, 86.1%–95.4%) and 86.0% (95% CI, 77.5%–91.6%) in HPV 16/18–positive and in the 12 other genotypes, respectively. Using DS alone to triage HPV-positive results showed significantly higher sensitivity and specificity than HPV 16/18 genotyping with cytology triage of 12 “other” genotypes, and substantially higher sensitivity but lower specificity than using cytology alone. Of note, triage with DS alone would have referred significantly fewer women to colposcopy than HPV 16/18 genotyping with cytology triage for the 12 other genotypes (48.6% vs 56.0%; P< .0001).
Similarly, a retrospective analysis of the ATHENA trial cohort of HPV-positive results of 7,727 patients aged 25 years or older also demonstrated increased sensitivity of DS compared with cytology (74.9% vs 51.9%; P<.0001) and similar specificities (74.1% vs 75%; P = .3198).36 The European PALMS study, which included 27,349 women aged 18 years or older across 5 countries who underwent routine screening with HPV testing, cytology, and DS, confirmed these findings.37 The sensitivity of DS was higher than that of cytology (86.7% vs 68.5%; P<.001) for CIN 3+ with comparable specificities (95.2% vs 95.4%; P = .15).
Challenges and opportunities to improve access to primary HPV screening
The historical success of the Pap test in reducing the incidence of cervical cancer relied on individuals having access to the test. This remains true as screening transitions to primary HPV testing. Limitations of HPV-based screening include provider and patient knowledge; access to tests; cost; need for new laboratory infrastructure; need to leverage the electronic health record to record results, calculate a patient’s risk and determine next steps; and the need to re-educate patients and providers about this new model of care. The American Cancer Society and the Centers for Disease Control and Prevention are currently leading initiatives to help adopt primary HPV screening in the United States and to facilitate new care approaches.
Self-collection and independence from subjective cytology would further improve access. Multiple effectiveness studies and patient acceptability studies have shown that primary HPV screening via self-collection is effective, cost effective, and acceptable to women, especially among underscreened populations.38 Sensitivity is comparable to clinician-obtained samples with polymerase chain reaction–based HPV tests. Furthermore, newer molecular tests that detect methylated target host genes or methylated viral genome can be used to triage HPV-positive cases. Several host methylation markers that identify the specific host genes (for example, CADM1, MAL, and miR-124-2) have been shown to be more specific, reproducible, and can be used in self-collected samples as they are based on molecular methylation analysis.39 The ASCCP monitors these new developments and will incorporate promising tests and approaches once validated and FDA approved into the risk-based management guidelines. An erratum was recently published, and the risk-calculator is also available on the ASCCP website free of charge (https://app.asccp.org).40
In conclusion, transition to primary HPV testing from Pap cytology in cervical cancer screening has many challenges but also opportunities. Learning from the experience of countries that have already adopted primary HPV testing is crucial to successful implementation of this new screening paradigm.41 The evidence supporting primary HPV screening with its improved sensitivity is clear, and the existing triage options and innovations will continue to improve triage of patients with clinically important lesions as well as accessibility. With strong advocacy and sound implementation, the WHO goal of cervical cancer elimination and 70% of women being screened with a high-performance test by age 35 and again by age 45 is achievable. ●
- Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71: 209-249.
- Cuzick J, Clavel C, Petry KU, et al. Overview of the European and North American studies on HPV testing in primary cervical cancer screening. Int J Cancer. 2006;119:1095-1101.
- Wright TC Jr, Massad LS, Dunton CJ, et al. 2006 consensus guidelines for the management of women with abnormal cervical cancer screening tests. Am J Obstet Gynecol. 2007;197:346-355.
- Tota JE, Bentley J, Blake J, et al. Introduction of molecular HPV testing as the primary technology in cervical cancer screening: acting on evidence to change the current paradigm. Prev Med. 2017;98:5-14.
- Ronco G, Giorgi Rossi P. Role of HPV DNA testing in modern gynaecological practice. Best Prac Res Clin Obstet Gynaecol. 2018;47:107-118.
- Wright TC, Stoler MH, Behrens CM, et al. Primary cervical cancer screening with human papillomavirus: end of study results from the ATHENA study using HPV as the first-line screening test. Gynecol Oncol. 2015;136:189-197.
- Mayrand MH, Duarte-Franco E, Rodrigues I, et al. Human papillomavirus DNA versus Papanicolaou screening tests for cervical cancer. N Engl J Med. 2007;357:1579-1588.
- Ronco G, Giorgi-Rossi P, Carozzi F, et al. Efficacy of human papillomavirus testing for the detection of invasive cervical cancers and cervical intraepithelial neoplasia: a randomised controlled trial. Lancet Oncol. 2010;11:249-257.
- Bulkmans NW, Rozendaal L, Snijders PJ, et al. POBASCAM, a population-based randomized controlled trial for implementation of high-risk HPV testing in cervical screening: design, methods and baseline data of 44,102 women. Int J Cancer. 2004;110:94-101.
- World Health Organization. WHO guideline for screening and treatment of cervical pre-cancer lesions for cervical cancer prevention. 2nd edition. Geneva: 2021. https://www .who.int/publications/i/item/9789240030824. Accessed April 28, 2022.
- American Cancer Society. The American Cancer Society guidelines for the prevention and early detection of cervical cancer. American Cancer Society; 2020. https://www.cancer .org/cancer/cervical-cancer/detection-diagnosis-staging /cervical-cancer-screening-guidelines.html. Accessed April 28, 2022.
- US Preventive Services Task Force; Curry SJ, Krist AH, Owens KD, et al. Screening for cervical cancer: US Preventive Services Task Force recommendation statement. JAMA. 2018;320:674-686.
- Moscicki AB, Flowers L, Huchko MJ, et al. Guidelines for cervical cancer screening in immunosuppressed women without HIV infection. J Low Gen Tract Dis. 2019;23:87-101.
- Committee opinion no. 675. Management of vulvar intraepithelial neoplasia. Obstet Gynecol. 2016;128:e178-e182.
- Satmary W, Holschneider CH, Brunette LL, et al. Vulvar intraepithelial neoplasia: risk factors for recurrence. Gynecol Oncol. 2018;148:126-131.
- Preti M, Scurry J, Marchitelli CE, et al. Vulvar intraepithelial neoplasia. Best Pract Res Clin Obstet Gynaecol. 2014;28:10511062.
- Khan MJ, Massad LS, Kinney W, et al. A common clinical dilemma: management of abnormal vaginal cytology and human papillomavirus test results. Gynecol Oncol. 2016;141:364-370.
- Perkins RB, Guido RS, Castle PE, et al. 2019 ASCCP risk-based management consensus guidelines for abnormal cervical cancer screening tests and cancer precursors. J Low Gen Tract Dis. 2020;24:102-131.
- Egemen D, Cheung LC, Chen X, et al. Risk estimates supporting the 2019 ASCCP risk-based management consensus guidelines. J Low Gen Tract Dis. 2020;24:132-143.
- Bhatla N, Singla S, Awasthi D. Human papillomavirus deoxyribonucleic acid testing in developed countries. Best Pract Res Clin Obstet Gynaecol. 2012;26:209-220.
- Meijer CJ, Berkhof J, Castle PE, et al. Guidelines for human papillomavirus DNA test requirements for primary cervical cancer screening in women 30 years and older. Int J Cancer. 2009;124:516-520.
- Ejegod D, Bottari F, Pedersen H, et al. The BD Onclarity HPV assay on samples collected in SurePath medium meets the international guidelines for human papillomavirus test requirements for cervical screening. J Clin Microbiol. 2016;54:2267-2272.
- Richardson LA, Tota J, Franco EL. Optimizing technology for cervical cancer screening in high-resource settings. Expert Rev Obstet Gynecol. 2011;6:343-353.
- Ronco G, Dillner J, Elfström KM, et al. Efficacy of HPV-based screening for prevention of invasive cervical cancer: followup of four European randomised controlled trials. Lancet. 2014;383:524-532.
- Ogilvie GS, van Niekerk D, Krajden M, et al. Effect of screening with primary cervical HPV testing vs cytology testing on high-grade cervical intraepithelial neoplasia at 48 months: the HPV FOCAL randomized clinical trial. JAMA. 2018;320:43-52.
- Dillner J, Rebolj M, Birembaut P, et al. Long term predictive values of cytology and human papillomavirus testing in cervical cancer screening: joint European cohort study. BMJ. 2008;337:a1754.
- Katki HA, Kinney WK, Fetterman B, et al. Cervical cancer risk for women undergoing concurrent testing for human papillomavirus and cervical cytology: a population-based study in routine clinical practice. Lancet Oncol. 2011;12:663-672.
- Arbyn M, Ronco G, Anttila A, et al. Evidence regarding human papillomavirus testing in secondary prevention of cervical cancer. Vaccine. 2012;30(suppl 5):F88-99.
- Schiffman M, Kinney WK, et al. Relative performance of HPV and cytology components of cotesting in cervical screening. J Nat Cancer Inst. 2018;110:501-508.
- Jin XW, Lipold L, Foucher J, et al. Cost-effectiveness of primary HPV testing, cytology and co-testing as cervical cancer screening for women above age 30 years. J Gen Intern Med. 2016;31:1338-1344.
- Tota JE, Bentley J, Blake J, et al. Approaches for triaging women who test positive for human papillomavirus in cervical cancer screening. Prev Med. 2017;98:15-20.
- Stoler MH, Wright TC Jr, Parvu V, et al. Stratified risk of high-grade cervical disease using onclarity HPV extended genotyping in women, ≥25 years of age, with NILM cytology. Gynecol Oncol. 2019;153:26-33.
- Louvanto K, Chevarie-Davis M, Ramanakumar AV, et al. HPV testing with cytology triage for cervical cancer screening in routine practice. Am J Obstet Gynecol. 2014;210:474.e1-7.
- Keating JT, Cviko A, Riethdorf S, et al. Ki-67, cyclin E, and p16INK4 are complimentary surrogate biomarkers for human papilloma virus-related cervical neoplasia. Am J Surg Pathol. 2001;25:884-891.
- Wright TC Jr, Stoler MH, Ranger-Moore J, et al. Clinical validation of p16/Ki-67 dual-stained cytology triage of HPV-positive women: results from the IMPACT trial. Int J Cancer. 2022;150:461-471.
- Wright TC Jr, Behrens CM, Ranger-Moore J, et al. Triaging HPV-positive women with p16/Ki-67 dual-stained cytology: results from a sub-study nested into the ATHENA trial. Gynecol Oncol. 2017;144:51-56.
- Ikenberg H, Bergeron C, Schmidt D, et al. Screening for cervical cancer precursors with p16/Ki-67 dual-stained cytology: results of the PALMS study. J Nat Cancer Inst. 2013;105:15501557.
- Arbyn M, Smith SB, Temin S, et al. Detecting cervical precancer and reaching underscreened women by using HPV testing on self samples: updated meta-analyses. BMJ. 2018;363:k4823.
- Verhoef VMJ, Bosgraaf RP, van Kemenade FJ, et al. Triage by methylation-marker testing versus cytology in women who test HPV-positive on self-collected cervicovaginal specimens (PROHTECT-3): a randomised controlled non-inferiority trial. Lancet Oncol. 2014;15:315-322.
- Perkins RB, Guido RS, Castle PE, et al. Erratum: 2019 ASCCP risk-based management consensus guidelines for abnormal cervical cancer screening tests and cancer precursors. J Low Gen Tract Dis. 2021;25:330-331.
- Hall MT, Simms KT, Lew JB, et al. The projected timeframe until cervical cancer elimination in Australia: a modelling study. Lancet Public Health. 2019;4:e19-e27.
- Huh WK, Ault KA, Chelmow D, et al. Use of primary high-risk human papillomavirus testing for cervical cancer screening: interim clinical guidance. Gynecol Oncol. 2015;136:178-182.
Cervical cancer is an important global health problem with an estimated 604,127 new cases and 341,831 deaths in 2020.1 Nearly 85% of the disease burden affects individuals from low and middle-income countries. The World Health Organization (WHO) set forth the goal for all countries to reach and maintain an incidence rate of below 4 per 100,000 women by 2030 as part of the Global Strategy to Accelerate the Elimination of Cervical Cancer.
Although traditional Pap cytology has been the cornerstone of screening programs, its poor sensitivity of approximately 50% and limitations in accessibility require new strategies to achieve the elimination of cervical cancer.2 The discovery that persistent infection with oncogenic human papillomavirus (HPV) is an essential step in the development of cervical cancer led to the development of diagnostic HPV tests, which have higher sensitivity than cytology (96.1% vs 53.0%) but somewhat lower specificity (90.7% vs 96.3%) for the detection of cervical intraepithelial neoplasia (CIN) 2 or worse lesions.2 Initially, HPV testing was incorporated as a method to triage atypical squamous cells of undetermined significance (ASCUS) cytology results.3 Later, the concept of cotesting with cytology emerged,4,5 and since then, several clinical trials have demonstrated the effectiveness of primary HPV screening.6-9
In 2020, the WHO recommended HPV DNA testing as the primary screening method starting at the age of 30 years, with regular testing every 5 to 10 years, for the general population.10 Currently, primary HPV has been adopted in multiple countries, including Australia, the Netherlands, Turkey, England, and Argentina.
In the United States, there are 3 currently acceptable screening strategies: cytology, cytology plus HPV (cotesting), and primary HPV testing (TABLE). The American Cancer Society (ACS) specifically states that HPV testing alone every 5 years is preferred starting at age 25 years; cotesting every 5 years or cytology alone every 3 years are also acceptable.11 The US Preventive Services Task Force (USPSTF) states that cytology alone every 3 years starting at 21 years and then HPV testing alone or cotesting every 5 years or cytology every 3 years starting at age 30 are all acceptable strategies.12
When applying these guidelines, it is important to note that they are intended for the screening of patients with all prior normal results with no symptoms. These routine screening guidelines do not apply to special populations, such as those with a history of abnormal results or treatment, a history of immunosuppression,13 a history of HPV-related vulvar or vaginal dysplasia,14-16 or a history of hysterectomy with removal of the cervix and no prior history of cervical dysplasia.17,18 By contrast, surveillance is interval testing for those who have either an abnormal prior test result or treatment; these may be managed per risk-based estimates provided by the American Society for Colposcopy and Cervical Pathology (ASCCP).18,19 Finally, diagnosis is evaluation (which may include diagnostic cytology) of a patient with abnormal signs and/or symptoms (such as bleeding, pain, discharge, or cervical mass).
In this Update, we present the evidence for primary HPV testing, the management options for a positive result in the United States, and research that will improve uptake of primary HPV testing as well as accessibility.
Change in screening paradigm: Evidence for primary HPV testing
HPV DNA tests are multiplex assays that detect the DNA of targeted high-risk HPV types, using multiple probes, either by direct genomic detection or by amplification of a viral DNA fragment using polymerase chain reaction (PCR).20,21 Alternatively, HPV mRNA-based tests detect the expression of E6 and E7 oncoproteins, a marker of viral integration.20 In examining the data from well-conducted clinical trials, 2 important observations are that different HPV assays were used and that direct comparison may not be valid. In addition, not all tests used in the studies are approved by the US Food and Drug Administration (FDA) for primary HPV testing.
Continue to: FDA-approved HPV tests...
FDA-approved HPV tests
Currently, 2 tests are FDA approved for primary HPV screening. The Cobas HPV test (Roche Molecular Diagnostics) was the first FDA-approved test for primary HPV screening in women aged 25 years and older.6 This test reports pooled results from 12 high-risk (hr) HPV types (31/33/35/39/45/51/52/56/58/59/66/68) with reflex genotyping for HPV 16/18, and thus it provides an immediate triage option for HPV-positive women. Of note, it is also approved for cotesting. The second FDA-approved test is the BD Onclarity HPV assay (Becton, Dickinson and Company) for primary HPV screening.22 It detects 14 hrHPV types, types 16/18/45 specifically as well as types 31/33/35/39/51/52/56/58/59/66/68.
Other HPV tests are FDA approved for cotesting and reflex testing but not for primary HPV testing. The Hybrid Capture test, or HC2 (Qiagen Inc), was the first HPV test to be approved by the FDA in 1997 for reflex testing of women with ASCUS cytology. In 2003, it was approved for cotesting along with cytology in women aged 30 years and older.20,21 In 2009, the Cervista HPV HR test (Hologic Inc) was approved for cotesting. The Aptima HPV assay (Hologic Inc), which is also approved for cotesting, is an RNA-based assay that allows detection of E6/E7 mRNA transcripts of 14 HPV types.23
Comparing HPV testing with cytology
Ronco and colleagues pooled data from 4 European randomized controlled trials (RCTs)—Swedescreen, POBASCAM, NTCC, ARTISTIC—with a total of 176,464 participants randomly assigned to HPV or cytology screening.24 Swedescreen and POBASCAM used GP5/GP6 PCR, while ARTISTIC and NTCC used HC2 for primary HPV screening. The screening interval was 3 years in all except 5 years in POBASCAM. The pooled detection rate of invasive disease was similar in the 2 arms, with pooled rate ratio for cancer detection being 0.79 (95% confidence interval [CI], 0.46–1.36) in the first 2.5 years, but was 0.45 (95% CI, 0.25–0.81), favoring the HPV arm, after 2.5 years. HPV testing was more effective in preventing cases of adenocarcinoma than squamous cell carcinoma (0.31 [95% CI, 0.14–0.69] vs 0.78 [95% CI, 0.49–1.25]). The authors concluded that HPV-based screening from age 30 years provided 60% to 70% better protection than cytology.
The result of the above meta-analysis was confirmed by the HPV FOCAL RCT that investigated the efficacy of HPV testing (HC2) in comparison with cytology.25 The detection rates for CIN 3 lesions supported primary HPV screening, with an absolute difference in incidence rate of 2.67/1,000 (95% CI, 0.53–4.88) at study randomization and 3.22/1,000 (95% CI, 5.12–1.48) at study exit 4 years later.
Cotesting using HPV and cytology: Marginal benefit
Dillner and colleagues were one of the first groups to report on the risk of CIN 3 based on both HPV and cytology status.26 Using pooled analysis of data from multiple countries, these investigators reported that the cumulative incidence rates (CIR) of CIN 3 after 6 years of follow-up increased consistently in HPV-positive subjects, and an HPV-positive result more accurately predicted CIN 3+ at 5 years than cytology alone. Furthermore, HPV negativity provided greater reassurance than cytology alone. At 5 years of follow-up, the rates of CIN 3+ were 0.25% (0.12%–0.41%) for women negative for HPV compared with 0.83% (0.50%–1.13%) for women with negative cytology results. There was little difference in rates for CIN 3+ between women with negative results on both tests and women who were negative for HPV.
The important question is then the marginal benefit of cotesting, which is the most costly screening option. A study of 331,818 women enrolled for cotesting at Kaiser Permanente found that the risk of CIN 3+ predicted by HPV testing alone when compared with cytology was significantly higher at both 3 years (5.0% vs 3.8%; P = .046) and 5 years (7.6% vs 4.7%; P = .001).27 A negative cytology result did not decrease the risk of CIN 3 further for HPV-negative patients (3 years: 0.047% vs 0.063%, P = .6; 5 years: 0.16% vs 0.17%, P = .8). They concluded that a negative HPV test was enough reassurance for low risk of CIN 3+ and that an additional negative cytology result does not provide extra reassurance.
Furthermore, a systematic meta-analysis of 48 studies, including 8 RCTs, found that the addition of cytology to HPV testing raised the sensitivity by 2% for CIN 3 compared with HPV testing alone. This improvement in sensitivity was at the expense of considerable loss of specificity, with a ratio of 0.93 (95% CI, 0.92–0.95) for CIN 3.28 Schiffman and colleagues also assessed the relative contribution of HPV testing and cytology in detection of CIN 3 and cancer.29 The HPV component alone identified a significantly higher proportion of preinvasive and invasive disease than cytology. Only 3.5% of precancers and 5.9% of cancers were preceded by HPV-negative, cytology-positive results. Thus, cytology contributed only 5 cases per million women per year to the sensitivity of the combined test, at the cost of significantly more colposcopies. Hence, the evidence suggests that there is limited benefit of adding cytology to HPV testing.30
Continue to: Triage of a positive HPV result...
Triage of a positive HPV result
An important limitation of HPV testing is its inability to discriminate between transient and persistent infections. Referral of all HPV-positive cases to colposcopy would overburden the system with associated unnecessary procedures. Hence, a triage strategy is essential to identify clinically important infections that truly require colposcopic evaluation. The FIGURE illustrates the management of a primary HPV test result performed for screening.

HPV genotyping
One strategy for triaging a positive HPV test result is genotyping. HPV 16 and 18 have the highest risk of persistence and progression and merit immediate referral to colposcopy. In the ATHENA trial, CIN 3 was identified in 17.8% (95% CI, 14.8–20.7%) of HPV 16 positive women at baseline, and the CIR increased to 25.2% (95% CI, 21.7–28.7%) after 3 years. The 3-year CIR of CIN 3 was only 5.4% (95% CI, 4.5–6.3%) in women with HPV genotypes other than 16/18. HPV 18–positive women had a 3-year CIR that was intermediate between women with HPV 16 and women with the 12 other genotypes.6 Hence, HPV 16/18–positive cases should be referred for immediate colposcopy, and negative cases should be followed up with cytology and referred for colposcopy if the cytology is ASCUS or worse.31
In July 2020, extended genotyping was approved by the FDA with individual detection of HPV 31, 51, 52 (in addition to 16, 18, and 45) and pooled detection of 33/58, 35/39/68, and 56/59/66. One study found that individual genotypes HPV 16 and 31 carry baseline risk values for CIN 3+ (8.1% and 7.5%, respectively) that are above the 5-year risk threshold for referral to colposcopy following the ASCCP risk-based management guideline.32
Cytology
The higher specificity of cytology makes it an option for triaging HPV-positive cases, and current management guidelines recommend triage to both genotyping and cytology for all patients who are HPV positive, and especially if they are HPV positive but HPV 16/18 negative. Of note, cytology results remain more subjective than those of primary HPV testing, but the combination of initial HPV testing with reflex to cytology is a reasonable and cost effective next step.18 The VASCAR trial found higher colposcopy referrals in the HPV screening and cytology triage group compared with the cytology alone group (19.36 vs 14.54 per 1,000 women).33 The ATHENA trial investigated various triage strategies for HPV-positive cases and its impact on colposcopy referrals.6 Using HPV genotyping and reflex cytology, if HPV 16/18 was positive, colposcopy was advised, but if any of the other 12 HPV types were positive, reflex cytology was done. If reported as ASCUS or worse, colposcopy was performed; conversely, if it was normal, women were rescreened with cotesting after 1 year. Although this strategy led to a reduction in the number of colposcopies, referrals were still higher in the primary HPV arm (3,769 colposcopies per 294 cases) compared with cytology (1,934 colposcopies per 179 cases) or cotesting (3,097 colposcopies per 240 cases) in women aged 25 years.14
p16/Ki-67 Dual-Stain
Diffused p16 immunohistochemical staining, as opposed to focal staining, is associated with active HPV infection but can be present in low-grade as well as high-grade lesions.34 Ki-67 is a marker of cellular proliferation. Coexpression of p16 and Ki-67 indicates a loss of cell cycle regulation and is a hallmark of neoplastic transformation. When positive, these tests are supportive of active HPV infection and of a high-grade lesion. Incorporation of these stains to cytology alone provides additional objective reassurance to cytology, where there is much inter- and intra-observer variability. These stains can be done by laboratories using the stains alone or they can use the FDA-approved p16/Ki-67 Dual-Stain immunohistochemistry (DS), CINtec PLUS Cytology (Roche Diagnostics). However, DS is not yet formally incorporated into triage algorithms by national guidelines.
The IMPACT trial assessed the performance of DS compared with cytology in the triage of HPV-positive results, with or without HPV 16/18 genotyping.35 This was a prospective observational screening study of 35,263 women aged 25 to 65 years across 32 sites in the United States. Of the 4,927 HPV-positive patients with DS results, the sensitivity of DS for CIN 3+ was 91.9% (95% CI, 86.1%–95.4%) and 86.0% (95% CI, 77.5%–91.6%) in HPV 16/18–positive and in the 12 other genotypes, respectively. Using DS alone to triage HPV-positive results showed significantly higher sensitivity and specificity than HPV 16/18 genotyping with cytology triage of 12 “other” genotypes, and substantially higher sensitivity but lower specificity than using cytology alone. Of note, triage with DS alone would have referred significantly fewer women to colposcopy than HPV 16/18 genotyping with cytology triage for the 12 other genotypes (48.6% vs 56.0%; P< .0001).
Similarly, a retrospective analysis of the ATHENA trial cohort of HPV-positive results of 7,727 patients aged 25 years or older also demonstrated increased sensitivity of DS compared with cytology (74.9% vs 51.9%; P<.0001) and similar specificities (74.1% vs 75%; P = .3198).36 The European PALMS study, which included 27,349 women aged 18 years or older across 5 countries who underwent routine screening with HPV testing, cytology, and DS, confirmed these findings.37 The sensitivity of DS was higher than that of cytology (86.7% vs 68.5%; P<.001) for CIN 3+ with comparable specificities (95.2% vs 95.4%; P = .15).
Challenges and opportunities to improve access to primary HPV screening
The historical success of the Pap test in reducing the incidence of cervical cancer relied on individuals having access to the test. This remains true as screening transitions to primary HPV testing. Limitations of HPV-based screening include provider and patient knowledge; access to tests; cost; need for new laboratory infrastructure; need to leverage the electronic health record to record results, calculate a patient’s risk and determine next steps; and the need to re-educate patients and providers about this new model of care. The American Cancer Society and the Centers for Disease Control and Prevention are currently leading initiatives to help adopt primary HPV screening in the United States and to facilitate new care approaches.
Self-collection and independence from subjective cytology would further improve access. Multiple effectiveness studies and patient acceptability studies have shown that primary HPV screening via self-collection is effective, cost effective, and acceptable to women, especially among underscreened populations.38 Sensitivity is comparable to clinician-obtained samples with polymerase chain reaction–based HPV tests. Furthermore, newer molecular tests that detect methylated target host genes or methylated viral genome can be used to triage HPV-positive cases. Several host methylation markers that identify the specific host genes (for example, CADM1, MAL, and miR-124-2) have been shown to be more specific, reproducible, and can be used in self-collected samples as they are based on molecular methylation analysis.39 The ASCCP monitors these new developments and will incorporate promising tests and approaches once validated and FDA approved into the risk-based management guidelines. An erratum was recently published, and the risk-calculator is also available on the ASCCP website free of charge (https://app.asccp.org).40
In conclusion, transition to primary HPV testing from Pap cytology in cervical cancer screening has many challenges but also opportunities. Learning from the experience of countries that have already adopted primary HPV testing is crucial to successful implementation of this new screening paradigm.41 The evidence supporting primary HPV screening with its improved sensitivity is clear, and the existing triage options and innovations will continue to improve triage of patients with clinically important lesions as well as accessibility. With strong advocacy and sound implementation, the WHO goal of cervical cancer elimination and 70% of women being screened with a high-performance test by age 35 and again by age 45 is achievable. ●
Cervical cancer is an important global health problem with an estimated 604,127 new cases and 341,831 deaths in 2020.1 Nearly 85% of the disease burden affects individuals from low and middle-income countries. The World Health Organization (WHO) set forth the goal for all countries to reach and maintain an incidence rate of below 4 per 100,000 women by 2030 as part of the Global Strategy to Accelerate the Elimination of Cervical Cancer.
Although traditional Pap cytology has been the cornerstone of screening programs, its poor sensitivity of approximately 50% and limitations in accessibility require new strategies to achieve the elimination of cervical cancer.2 The discovery that persistent infection with oncogenic human papillomavirus (HPV) is an essential step in the development of cervical cancer led to the development of diagnostic HPV tests, which have higher sensitivity than cytology (96.1% vs 53.0%) but somewhat lower specificity (90.7% vs 96.3%) for the detection of cervical intraepithelial neoplasia (CIN) 2 or worse lesions.2 Initially, HPV testing was incorporated as a method to triage atypical squamous cells of undetermined significance (ASCUS) cytology results.3 Later, the concept of cotesting with cytology emerged,4,5 and since then, several clinical trials have demonstrated the effectiveness of primary HPV screening.6-9
In 2020, the WHO recommended HPV DNA testing as the primary screening method starting at the age of 30 years, with regular testing every 5 to 10 years, for the general population.10 Currently, primary HPV has been adopted in multiple countries, including Australia, the Netherlands, Turkey, England, and Argentina.
In the United States, there are 3 currently acceptable screening strategies: cytology, cytology plus HPV (cotesting), and primary HPV testing (TABLE). The American Cancer Society (ACS) specifically states that HPV testing alone every 5 years is preferred starting at age 25 years; cotesting every 5 years or cytology alone every 3 years are also acceptable.11 The US Preventive Services Task Force (USPSTF) states that cytology alone every 3 years starting at 21 years and then HPV testing alone or cotesting every 5 years or cytology every 3 years starting at age 30 are all acceptable strategies.12
When applying these guidelines, it is important to note that they are intended for the screening of patients with all prior normal results with no symptoms. These routine screening guidelines do not apply to special populations, such as those with a history of abnormal results or treatment, a history of immunosuppression,13 a history of HPV-related vulvar or vaginal dysplasia,14-16 or a history of hysterectomy with removal of the cervix and no prior history of cervical dysplasia.17,18 By contrast, surveillance is interval testing for those who have either an abnormal prior test result or treatment; these may be managed per risk-based estimates provided by the American Society for Colposcopy and Cervical Pathology (ASCCP).18,19 Finally, diagnosis is evaluation (which may include diagnostic cytology) of a patient with abnormal signs and/or symptoms (such as bleeding, pain, discharge, or cervical mass).
In this Update, we present the evidence for primary HPV testing, the management options for a positive result in the United States, and research that will improve uptake of primary HPV testing as well as accessibility.
Change in screening paradigm: Evidence for primary HPV testing
HPV DNA tests are multiplex assays that detect the DNA of targeted high-risk HPV types, using multiple probes, either by direct genomic detection or by amplification of a viral DNA fragment using polymerase chain reaction (PCR).20,21 Alternatively, HPV mRNA-based tests detect the expression of E6 and E7 oncoproteins, a marker of viral integration.20 In examining the data from well-conducted clinical trials, 2 important observations are that different HPV assays were used and that direct comparison may not be valid. In addition, not all tests used in the studies are approved by the US Food and Drug Administration (FDA) for primary HPV testing.
Continue to: FDA-approved HPV tests...
FDA-approved HPV tests
Currently, 2 tests are FDA approved for primary HPV screening. The Cobas HPV test (Roche Molecular Diagnostics) was the first FDA-approved test for primary HPV screening in women aged 25 years and older.6 This test reports pooled results from 12 high-risk (hr) HPV types (31/33/35/39/45/51/52/56/58/59/66/68) with reflex genotyping for HPV 16/18, and thus it provides an immediate triage option for HPV-positive women. Of note, it is also approved for cotesting. The second FDA-approved test is the BD Onclarity HPV assay (Becton, Dickinson and Company) for primary HPV screening.22 It detects 14 hrHPV types, types 16/18/45 specifically as well as types 31/33/35/39/51/52/56/58/59/66/68.
Other HPV tests are FDA approved for cotesting and reflex testing but not for primary HPV testing. The Hybrid Capture test, or HC2 (Qiagen Inc), was the first HPV test to be approved by the FDA in 1997 for reflex testing of women with ASCUS cytology. In 2003, it was approved for cotesting along with cytology in women aged 30 years and older.20,21 In 2009, the Cervista HPV HR test (Hologic Inc) was approved for cotesting. The Aptima HPV assay (Hologic Inc), which is also approved for cotesting, is an RNA-based assay that allows detection of E6/E7 mRNA transcripts of 14 HPV types.23
Comparing HPV testing with cytology
Ronco and colleagues pooled data from 4 European randomized controlled trials (RCTs)—Swedescreen, POBASCAM, NTCC, ARTISTIC—with a total of 176,464 participants randomly assigned to HPV or cytology screening.24 Swedescreen and POBASCAM used GP5/GP6 PCR, while ARTISTIC and NTCC used HC2 for primary HPV screening. The screening interval was 3 years in all except 5 years in POBASCAM. The pooled detection rate of invasive disease was similar in the 2 arms, with pooled rate ratio for cancer detection being 0.79 (95% confidence interval [CI], 0.46–1.36) in the first 2.5 years, but was 0.45 (95% CI, 0.25–0.81), favoring the HPV arm, after 2.5 years. HPV testing was more effective in preventing cases of adenocarcinoma than squamous cell carcinoma (0.31 [95% CI, 0.14–0.69] vs 0.78 [95% CI, 0.49–1.25]). The authors concluded that HPV-based screening from age 30 years provided 60% to 70% better protection than cytology.
The result of the above meta-analysis was confirmed by the HPV FOCAL RCT that investigated the efficacy of HPV testing (HC2) in comparison with cytology.25 The detection rates for CIN 3 lesions supported primary HPV screening, with an absolute difference in incidence rate of 2.67/1,000 (95% CI, 0.53–4.88) at study randomization and 3.22/1,000 (95% CI, 5.12–1.48) at study exit 4 years later.
Cotesting using HPV and cytology: Marginal benefit
Dillner and colleagues were one of the first groups to report on the risk of CIN 3 based on both HPV and cytology status.26 Using pooled analysis of data from multiple countries, these investigators reported that the cumulative incidence rates (CIR) of CIN 3 after 6 years of follow-up increased consistently in HPV-positive subjects, and an HPV-positive result more accurately predicted CIN 3+ at 5 years than cytology alone. Furthermore, HPV negativity provided greater reassurance than cytology alone. At 5 years of follow-up, the rates of CIN 3+ were 0.25% (0.12%–0.41%) for women negative for HPV compared with 0.83% (0.50%–1.13%) for women with negative cytology results. There was little difference in rates for CIN 3+ between women with negative results on both tests and women who were negative for HPV.
The important question is then the marginal benefit of cotesting, which is the most costly screening option. A study of 331,818 women enrolled for cotesting at Kaiser Permanente found that the risk of CIN 3+ predicted by HPV testing alone when compared with cytology was significantly higher at both 3 years (5.0% vs 3.8%; P = .046) and 5 years (7.6% vs 4.7%; P = .001).27 A negative cytology result did not decrease the risk of CIN 3 further for HPV-negative patients (3 years: 0.047% vs 0.063%, P = .6; 5 years: 0.16% vs 0.17%, P = .8). They concluded that a negative HPV test was enough reassurance for low risk of CIN 3+ and that an additional negative cytology result does not provide extra reassurance.
Furthermore, a systematic meta-analysis of 48 studies, including 8 RCTs, found that the addition of cytology to HPV testing raised the sensitivity by 2% for CIN 3 compared with HPV testing alone. This improvement in sensitivity was at the expense of considerable loss of specificity, with a ratio of 0.93 (95% CI, 0.92–0.95) for CIN 3.28 Schiffman and colleagues also assessed the relative contribution of HPV testing and cytology in detection of CIN 3 and cancer.29 The HPV component alone identified a significantly higher proportion of preinvasive and invasive disease than cytology. Only 3.5% of precancers and 5.9% of cancers were preceded by HPV-negative, cytology-positive results. Thus, cytology contributed only 5 cases per million women per year to the sensitivity of the combined test, at the cost of significantly more colposcopies. Hence, the evidence suggests that there is limited benefit of adding cytology to HPV testing.30
Continue to: Triage of a positive HPV result...
Triage of a positive HPV result
An important limitation of HPV testing is its inability to discriminate between transient and persistent infections. Referral of all HPV-positive cases to colposcopy would overburden the system with associated unnecessary procedures. Hence, a triage strategy is essential to identify clinically important infections that truly require colposcopic evaluation. The FIGURE illustrates the management of a primary HPV test result performed for screening.

HPV genotyping
One strategy for triaging a positive HPV test result is genotyping. HPV 16 and 18 have the highest risk of persistence and progression and merit immediate referral to colposcopy. In the ATHENA trial, CIN 3 was identified in 17.8% (95% CI, 14.8–20.7%) of HPV 16 positive women at baseline, and the CIR increased to 25.2% (95% CI, 21.7–28.7%) after 3 years. The 3-year CIR of CIN 3 was only 5.4% (95% CI, 4.5–6.3%) in women with HPV genotypes other than 16/18. HPV 18–positive women had a 3-year CIR that was intermediate between women with HPV 16 and women with the 12 other genotypes.6 Hence, HPV 16/18–positive cases should be referred for immediate colposcopy, and negative cases should be followed up with cytology and referred for colposcopy if the cytology is ASCUS or worse.31
In July 2020, extended genotyping was approved by the FDA with individual detection of HPV 31, 51, 52 (in addition to 16, 18, and 45) and pooled detection of 33/58, 35/39/68, and 56/59/66. One study found that individual genotypes HPV 16 and 31 carry baseline risk values for CIN 3+ (8.1% and 7.5%, respectively) that are above the 5-year risk threshold for referral to colposcopy following the ASCCP risk-based management guideline.32
Cytology
The higher specificity of cytology makes it an option for triaging HPV-positive cases, and current management guidelines recommend triage to both genotyping and cytology for all patients who are HPV positive, and especially if they are HPV positive but HPV 16/18 negative. Of note, cytology results remain more subjective than those of primary HPV testing, but the combination of initial HPV testing with reflex to cytology is a reasonable and cost effective next step.18 The VASCAR trial found higher colposcopy referrals in the HPV screening and cytology triage group compared with the cytology alone group (19.36 vs 14.54 per 1,000 women).33 The ATHENA trial investigated various triage strategies for HPV-positive cases and its impact on colposcopy referrals.6 Using HPV genotyping and reflex cytology, if HPV 16/18 was positive, colposcopy was advised, but if any of the other 12 HPV types were positive, reflex cytology was done. If reported as ASCUS or worse, colposcopy was performed; conversely, if it was normal, women were rescreened with cotesting after 1 year. Although this strategy led to a reduction in the number of colposcopies, referrals were still higher in the primary HPV arm (3,769 colposcopies per 294 cases) compared with cytology (1,934 colposcopies per 179 cases) or cotesting (3,097 colposcopies per 240 cases) in women aged 25 years.14
p16/Ki-67 Dual-Stain
Diffused p16 immunohistochemical staining, as opposed to focal staining, is associated with active HPV infection but can be present in low-grade as well as high-grade lesions.34 Ki-67 is a marker of cellular proliferation. Coexpression of p16 and Ki-67 indicates a loss of cell cycle regulation and is a hallmark of neoplastic transformation. When positive, these tests are supportive of active HPV infection and of a high-grade lesion. Incorporation of these stains to cytology alone provides additional objective reassurance to cytology, where there is much inter- and intra-observer variability. These stains can be done by laboratories using the stains alone or they can use the FDA-approved p16/Ki-67 Dual-Stain immunohistochemistry (DS), CINtec PLUS Cytology (Roche Diagnostics). However, DS is not yet formally incorporated into triage algorithms by national guidelines.
The IMPACT trial assessed the performance of DS compared with cytology in the triage of HPV-positive results, with or without HPV 16/18 genotyping.35 This was a prospective observational screening study of 35,263 women aged 25 to 65 years across 32 sites in the United States. Of the 4,927 HPV-positive patients with DS results, the sensitivity of DS for CIN 3+ was 91.9% (95% CI, 86.1%–95.4%) and 86.0% (95% CI, 77.5%–91.6%) in HPV 16/18–positive and in the 12 other genotypes, respectively. Using DS alone to triage HPV-positive results showed significantly higher sensitivity and specificity than HPV 16/18 genotyping with cytology triage of 12 “other” genotypes, and substantially higher sensitivity but lower specificity than using cytology alone. Of note, triage with DS alone would have referred significantly fewer women to colposcopy than HPV 16/18 genotyping with cytology triage for the 12 other genotypes (48.6% vs 56.0%; P< .0001).
Similarly, a retrospective analysis of the ATHENA trial cohort of HPV-positive results of 7,727 patients aged 25 years or older also demonstrated increased sensitivity of DS compared with cytology (74.9% vs 51.9%; P<.0001) and similar specificities (74.1% vs 75%; P = .3198).36 The European PALMS study, which included 27,349 women aged 18 years or older across 5 countries who underwent routine screening with HPV testing, cytology, and DS, confirmed these findings.37 The sensitivity of DS was higher than that of cytology (86.7% vs 68.5%; P<.001) for CIN 3+ with comparable specificities (95.2% vs 95.4%; P = .15).
Challenges and opportunities to improve access to primary HPV screening
The historical success of the Pap test in reducing the incidence of cervical cancer relied on individuals having access to the test. This remains true as screening transitions to primary HPV testing. Limitations of HPV-based screening include provider and patient knowledge; access to tests; cost; need for new laboratory infrastructure; need to leverage the electronic health record to record results, calculate a patient’s risk and determine next steps; and the need to re-educate patients and providers about this new model of care. The American Cancer Society and the Centers for Disease Control and Prevention are currently leading initiatives to help adopt primary HPV screening in the United States and to facilitate new care approaches.
Self-collection and independence from subjective cytology would further improve access. Multiple effectiveness studies and patient acceptability studies have shown that primary HPV screening via self-collection is effective, cost effective, and acceptable to women, especially among underscreened populations.38 Sensitivity is comparable to clinician-obtained samples with polymerase chain reaction–based HPV tests. Furthermore, newer molecular tests that detect methylated target host genes or methylated viral genome can be used to triage HPV-positive cases. Several host methylation markers that identify the specific host genes (for example, CADM1, MAL, and miR-124-2) have been shown to be more specific, reproducible, and can be used in self-collected samples as they are based on molecular methylation analysis.39 The ASCCP monitors these new developments and will incorporate promising tests and approaches once validated and FDA approved into the risk-based management guidelines. An erratum was recently published, and the risk-calculator is also available on the ASCCP website free of charge (https://app.asccp.org).40
In conclusion, transition to primary HPV testing from Pap cytology in cervical cancer screening has many challenges but also opportunities. Learning from the experience of countries that have already adopted primary HPV testing is crucial to successful implementation of this new screening paradigm.41 The evidence supporting primary HPV screening with its improved sensitivity is clear, and the existing triage options and innovations will continue to improve triage of patients with clinically important lesions as well as accessibility. With strong advocacy and sound implementation, the WHO goal of cervical cancer elimination and 70% of women being screened with a high-performance test by age 35 and again by age 45 is achievable. ●
- Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71: 209-249.
- Cuzick J, Clavel C, Petry KU, et al. Overview of the European and North American studies on HPV testing in primary cervical cancer screening. Int J Cancer. 2006;119:1095-1101.
- Wright TC Jr, Massad LS, Dunton CJ, et al. 2006 consensus guidelines for the management of women with abnormal cervical cancer screening tests. Am J Obstet Gynecol. 2007;197:346-355.
- Tota JE, Bentley J, Blake J, et al. Introduction of molecular HPV testing as the primary technology in cervical cancer screening: acting on evidence to change the current paradigm. Prev Med. 2017;98:5-14.
- Ronco G, Giorgi Rossi P. Role of HPV DNA testing in modern gynaecological practice. Best Prac Res Clin Obstet Gynaecol. 2018;47:107-118.
- Wright TC, Stoler MH, Behrens CM, et al. Primary cervical cancer screening with human papillomavirus: end of study results from the ATHENA study using HPV as the first-line screening test. Gynecol Oncol. 2015;136:189-197.
- Mayrand MH, Duarte-Franco E, Rodrigues I, et al. Human papillomavirus DNA versus Papanicolaou screening tests for cervical cancer. N Engl J Med. 2007;357:1579-1588.
- Ronco G, Giorgi-Rossi P, Carozzi F, et al. Efficacy of human papillomavirus testing for the detection of invasive cervical cancers and cervical intraepithelial neoplasia: a randomised controlled trial. Lancet Oncol. 2010;11:249-257.
- Bulkmans NW, Rozendaal L, Snijders PJ, et al. POBASCAM, a population-based randomized controlled trial for implementation of high-risk HPV testing in cervical screening: design, methods and baseline data of 44,102 women. Int J Cancer. 2004;110:94-101.
- World Health Organization. WHO guideline for screening and treatment of cervical pre-cancer lesions for cervical cancer prevention. 2nd edition. Geneva: 2021. https://www .who.int/publications/i/item/9789240030824. Accessed April 28, 2022.
- American Cancer Society. The American Cancer Society guidelines for the prevention and early detection of cervical cancer. American Cancer Society; 2020. https://www.cancer .org/cancer/cervical-cancer/detection-diagnosis-staging /cervical-cancer-screening-guidelines.html. Accessed April 28, 2022.
- US Preventive Services Task Force; Curry SJ, Krist AH, Owens KD, et al. Screening for cervical cancer: US Preventive Services Task Force recommendation statement. JAMA. 2018;320:674-686.
- Moscicki AB, Flowers L, Huchko MJ, et al. Guidelines for cervical cancer screening in immunosuppressed women without HIV infection. J Low Gen Tract Dis. 2019;23:87-101.
- Committee opinion no. 675. Management of vulvar intraepithelial neoplasia. Obstet Gynecol. 2016;128:e178-e182.
- Satmary W, Holschneider CH, Brunette LL, et al. Vulvar intraepithelial neoplasia: risk factors for recurrence. Gynecol Oncol. 2018;148:126-131.
- Preti M, Scurry J, Marchitelli CE, et al. Vulvar intraepithelial neoplasia. Best Pract Res Clin Obstet Gynaecol. 2014;28:10511062.
- Khan MJ, Massad LS, Kinney W, et al. A common clinical dilemma: management of abnormal vaginal cytology and human papillomavirus test results. Gynecol Oncol. 2016;141:364-370.
- Perkins RB, Guido RS, Castle PE, et al. 2019 ASCCP risk-based management consensus guidelines for abnormal cervical cancer screening tests and cancer precursors. J Low Gen Tract Dis. 2020;24:102-131.
- Egemen D, Cheung LC, Chen X, et al. Risk estimates supporting the 2019 ASCCP risk-based management consensus guidelines. J Low Gen Tract Dis. 2020;24:132-143.
- Bhatla N, Singla S, Awasthi D. Human papillomavirus deoxyribonucleic acid testing in developed countries. Best Pract Res Clin Obstet Gynaecol. 2012;26:209-220.
- Meijer CJ, Berkhof J, Castle PE, et al. Guidelines for human papillomavirus DNA test requirements for primary cervical cancer screening in women 30 years and older. Int J Cancer. 2009;124:516-520.
- Ejegod D, Bottari F, Pedersen H, et al. The BD Onclarity HPV assay on samples collected in SurePath medium meets the international guidelines for human papillomavirus test requirements for cervical screening. J Clin Microbiol. 2016;54:2267-2272.
- Richardson LA, Tota J, Franco EL. Optimizing technology for cervical cancer screening in high-resource settings. Expert Rev Obstet Gynecol. 2011;6:343-353.
- Ronco G, Dillner J, Elfström KM, et al. Efficacy of HPV-based screening for prevention of invasive cervical cancer: followup of four European randomised controlled trials. Lancet. 2014;383:524-532.
- Ogilvie GS, van Niekerk D, Krajden M, et al. Effect of screening with primary cervical HPV testing vs cytology testing on high-grade cervical intraepithelial neoplasia at 48 months: the HPV FOCAL randomized clinical trial. JAMA. 2018;320:43-52.
- Dillner J, Rebolj M, Birembaut P, et al. Long term predictive values of cytology and human papillomavirus testing in cervical cancer screening: joint European cohort study. BMJ. 2008;337:a1754.
- Katki HA, Kinney WK, Fetterman B, et al. Cervical cancer risk for women undergoing concurrent testing for human papillomavirus and cervical cytology: a population-based study in routine clinical practice. Lancet Oncol. 2011;12:663-672.
- Arbyn M, Ronco G, Anttila A, et al. Evidence regarding human papillomavirus testing in secondary prevention of cervical cancer. Vaccine. 2012;30(suppl 5):F88-99.
- Schiffman M, Kinney WK, et al. Relative performance of HPV and cytology components of cotesting in cervical screening. J Nat Cancer Inst. 2018;110:501-508.
- Jin XW, Lipold L, Foucher J, et al. Cost-effectiveness of primary HPV testing, cytology and co-testing as cervical cancer screening for women above age 30 years. J Gen Intern Med. 2016;31:1338-1344.
- Tota JE, Bentley J, Blake J, et al. Approaches for triaging women who test positive for human papillomavirus in cervical cancer screening. Prev Med. 2017;98:15-20.
- Stoler MH, Wright TC Jr, Parvu V, et al. Stratified risk of high-grade cervical disease using onclarity HPV extended genotyping in women, ≥25 years of age, with NILM cytology. Gynecol Oncol. 2019;153:26-33.
- Louvanto K, Chevarie-Davis M, Ramanakumar AV, et al. HPV testing with cytology triage for cervical cancer screening in routine practice. Am J Obstet Gynecol. 2014;210:474.e1-7.
- Keating JT, Cviko A, Riethdorf S, et al. Ki-67, cyclin E, and p16INK4 are complimentary surrogate biomarkers for human papilloma virus-related cervical neoplasia. Am J Surg Pathol. 2001;25:884-891.
- Wright TC Jr, Stoler MH, Ranger-Moore J, et al. Clinical validation of p16/Ki-67 dual-stained cytology triage of HPV-positive women: results from the IMPACT trial. Int J Cancer. 2022;150:461-471.
- Wright TC Jr, Behrens CM, Ranger-Moore J, et al. Triaging HPV-positive women with p16/Ki-67 dual-stained cytology: results from a sub-study nested into the ATHENA trial. Gynecol Oncol. 2017;144:51-56.
- Ikenberg H, Bergeron C, Schmidt D, et al. Screening for cervical cancer precursors with p16/Ki-67 dual-stained cytology: results of the PALMS study. J Nat Cancer Inst. 2013;105:15501557.
- Arbyn M, Smith SB, Temin S, et al. Detecting cervical precancer and reaching underscreened women by using HPV testing on self samples: updated meta-analyses. BMJ. 2018;363:k4823.
- Verhoef VMJ, Bosgraaf RP, van Kemenade FJ, et al. Triage by methylation-marker testing versus cytology in women who test HPV-positive on self-collected cervicovaginal specimens (PROHTECT-3): a randomised controlled non-inferiority trial. Lancet Oncol. 2014;15:315-322.
- Perkins RB, Guido RS, Castle PE, et al. Erratum: 2019 ASCCP risk-based management consensus guidelines for abnormal cervical cancer screening tests and cancer precursors. J Low Gen Tract Dis. 2021;25:330-331.
- Hall MT, Simms KT, Lew JB, et al. The projected timeframe until cervical cancer elimination in Australia: a modelling study. Lancet Public Health. 2019;4:e19-e27.
- Huh WK, Ault KA, Chelmow D, et al. Use of primary high-risk human papillomavirus testing for cervical cancer screening: interim clinical guidance. Gynecol Oncol. 2015;136:178-182.
- Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71: 209-249.
- Cuzick J, Clavel C, Petry KU, et al. Overview of the European and North American studies on HPV testing in primary cervical cancer screening. Int J Cancer. 2006;119:1095-1101.
- Wright TC Jr, Massad LS, Dunton CJ, et al. 2006 consensus guidelines for the management of women with abnormal cervical cancer screening tests. Am J Obstet Gynecol. 2007;197:346-355.
- Tota JE, Bentley J, Blake J, et al. Introduction of molecular HPV testing as the primary technology in cervical cancer screening: acting on evidence to change the current paradigm. Prev Med. 2017;98:5-14.
- Ronco G, Giorgi Rossi P. Role of HPV DNA testing in modern gynaecological practice. Best Prac Res Clin Obstet Gynaecol. 2018;47:107-118.
- Wright TC, Stoler MH, Behrens CM, et al. Primary cervical cancer screening with human papillomavirus: end of study results from the ATHENA study using HPV as the first-line screening test. Gynecol Oncol. 2015;136:189-197.
- Mayrand MH, Duarte-Franco E, Rodrigues I, et al. Human papillomavirus DNA versus Papanicolaou screening tests for cervical cancer. N Engl J Med. 2007;357:1579-1588.
- Ronco G, Giorgi-Rossi P, Carozzi F, et al. Efficacy of human papillomavirus testing for the detection of invasive cervical cancers and cervical intraepithelial neoplasia: a randomised controlled trial. Lancet Oncol. 2010;11:249-257.
- Bulkmans NW, Rozendaal L, Snijders PJ, et al. POBASCAM, a population-based randomized controlled trial for implementation of high-risk HPV testing in cervical screening: design, methods and baseline data of 44,102 women. Int J Cancer. 2004;110:94-101.
- World Health Organization. WHO guideline for screening and treatment of cervical pre-cancer lesions for cervical cancer prevention. 2nd edition. Geneva: 2021. https://www .who.int/publications/i/item/9789240030824. Accessed April 28, 2022.
- American Cancer Society. The American Cancer Society guidelines for the prevention and early detection of cervical cancer. American Cancer Society; 2020. https://www.cancer .org/cancer/cervical-cancer/detection-diagnosis-staging /cervical-cancer-screening-guidelines.html. Accessed April 28, 2022.
- US Preventive Services Task Force; Curry SJ, Krist AH, Owens KD, et al. Screening for cervical cancer: US Preventive Services Task Force recommendation statement. JAMA. 2018;320:674-686.
- Moscicki AB, Flowers L, Huchko MJ, et al. Guidelines for cervical cancer screening in immunosuppressed women without HIV infection. J Low Gen Tract Dis. 2019;23:87-101.
- Committee opinion no. 675. Management of vulvar intraepithelial neoplasia. Obstet Gynecol. 2016;128:e178-e182.
- Satmary W, Holschneider CH, Brunette LL, et al. Vulvar intraepithelial neoplasia: risk factors for recurrence. Gynecol Oncol. 2018;148:126-131.
- Preti M, Scurry J, Marchitelli CE, et al. Vulvar intraepithelial neoplasia. Best Pract Res Clin Obstet Gynaecol. 2014;28:10511062.
- Khan MJ, Massad LS, Kinney W, et al. A common clinical dilemma: management of abnormal vaginal cytology and human papillomavirus test results. Gynecol Oncol. 2016;141:364-370.
- Perkins RB, Guido RS, Castle PE, et al. 2019 ASCCP risk-based management consensus guidelines for abnormal cervical cancer screening tests and cancer precursors. J Low Gen Tract Dis. 2020;24:102-131.
- Egemen D, Cheung LC, Chen X, et al. Risk estimates supporting the 2019 ASCCP risk-based management consensus guidelines. J Low Gen Tract Dis. 2020;24:132-143.
- Bhatla N, Singla S, Awasthi D. Human papillomavirus deoxyribonucleic acid testing in developed countries. Best Pract Res Clin Obstet Gynaecol. 2012;26:209-220.
- Meijer CJ, Berkhof J, Castle PE, et al. Guidelines for human papillomavirus DNA test requirements for primary cervical cancer screening in women 30 years and older. Int J Cancer. 2009;124:516-520.
- Ejegod D, Bottari F, Pedersen H, et al. The BD Onclarity HPV assay on samples collected in SurePath medium meets the international guidelines for human papillomavirus test requirements for cervical screening. J Clin Microbiol. 2016;54:2267-2272.
- Richardson LA, Tota J, Franco EL. Optimizing technology for cervical cancer screening in high-resource settings. Expert Rev Obstet Gynecol. 2011;6:343-353.
- Ronco G, Dillner J, Elfström KM, et al. Efficacy of HPV-based screening for prevention of invasive cervical cancer: followup of four European randomised controlled trials. Lancet. 2014;383:524-532.
- Ogilvie GS, van Niekerk D, Krajden M, et al. Effect of screening with primary cervical HPV testing vs cytology testing on high-grade cervical intraepithelial neoplasia at 48 months: the HPV FOCAL randomized clinical trial. JAMA. 2018;320:43-52.
- Dillner J, Rebolj M, Birembaut P, et al. Long term predictive values of cytology and human papillomavirus testing in cervical cancer screening: joint European cohort study. BMJ. 2008;337:a1754.
- Katki HA, Kinney WK, Fetterman B, et al. Cervical cancer risk for women undergoing concurrent testing for human papillomavirus and cervical cytology: a population-based study in routine clinical practice. Lancet Oncol. 2011;12:663-672.
- Arbyn M, Ronco G, Anttila A, et al. Evidence regarding human papillomavirus testing in secondary prevention of cervical cancer. Vaccine. 2012;30(suppl 5):F88-99.
- Schiffman M, Kinney WK, et al. Relative performance of HPV and cytology components of cotesting in cervical screening. J Nat Cancer Inst. 2018;110:501-508.
- Jin XW, Lipold L, Foucher J, et al. Cost-effectiveness of primary HPV testing, cytology and co-testing as cervical cancer screening for women above age 30 years. J Gen Intern Med. 2016;31:1338-1344.
- Tota JE, Bentley J, Blake J, et al. Approaches for triaging women who test positive for human papillomavirus in cervical cancer screening. Prev Med. 2017;98:15-20.
- Stoler MH, Wright TC Jr, Parvu V, et al. Stratified risk of high-grade cervical disease using onclarity HPV extended genotyping in women, ≥25 years of age, with NILM cytology. Gynecol Oncol. 2019;153:26-33.
- Louvanto K, Chevarie-Davis M, Ramanakumar AV, et al. HPV testing with cytology triage for cervical cancer screening in routine practice. Am J Obstet Gynecol. 2014;210:474.e1-7.
- Keating JT, Cviko A, Riethdorf S, et al. Ki-67, cyclin E, and p16INK4 are complimentary surrogate biomarkers for human papilloma virus-related cervical neoplasia. Am J Surg Pathol. 2001;25:884-891.
- Wright TC Jr, Stoler MH, Ranger-Moore J, et al. Clinical validation of p16/Ki-67 dual-stained cytology triage of HPV-positive women: results from the IMPACT trial. Int J Cancer. 2022;150:461-471.
- Wright TC Jr, Behrens CM, Ranger-Moore J, et al. Triaging HPV-positive women with p16/Ki-67 dual-stained cytology: results from a sub-study nested into the ATHENA trial. Gynecol Oncol. 2017;144:51-56.
- Ikenberg H, Bergeron C, Schmidt D, et al. Screening for cervical cancer precursors with p16/Ki-67 dual-stained cytology: results of the PALMS study. J Nat Cancer Inst. 2013;105:15501557.
- Arbyn M, Smith SB, Temin S, et al. Detecting cervical precancer and reaching underscreened women by using HPV testing on self samples: updated meta-analyses. BMJ. 2018;363:k4823.
- Verhoef VMJ, Bosgraaf RP, van Kemenade FJ, et al. Triage by methylation-marker testing versus cytology in women who test HPV-positive on self-collected cervicovaginal specimens (PROHTECT-3): a randomised controlled non-inferiority trial. Lancet Oncol. 2014;15:315-322.
- Perkins RB, Guido RS, Castle PE, et al. Erratum: 2019 ASCCP risk-based management consensus guidelines for abnormal cervical cancer screening tests and cancer precursors. J Low Gen Tract Dis. 2021;25:330-331.
- Hall MT, Simms KT, Lew JB, et al. The projected timeframe until cervical cancer elimination in Australia: a modelling study. Lancet Public Health. 2019;4:e19-e27.
- Huh WK, Ault KA, Chelmow D, et al. Use of primary high-risk human papillomavirus testing for cervical cancer screening: interim clinical guidance. Gynecol Oncol. 2015;136:178-182.
Cervical cancer: A path to eradication
David G. Mutch, MD: The cervical cancer screening guidelines, using Pap testing, have changed significantly since the times of yearly Paps and exams. Coupled with vaccination and new management guidelines (recommending HPV testing, etc), we actually hope that we are on the way to eradicating cervical cancer from our environment.
Screening: Current recommendations
Dr. Mutch: Warner, the American Society of Colposcopy and Cervical Pathology (ASCCP)1 endorses the cervical cancer screening guidelines for several professional organizations, including the American Cancer Society (ACS),2 the US Preventive Services Task Force (USPSTF),3 and the American College of Obstetricians and Gynecologists (ACOG).4 What are the current screening recommendations, as these organizations have disparate views?
Warner Huh, MD: There was a time, around 2012-2013, when for the first time ever, we had significant harmonization of the guidelines between ACOG and the USPSTF and ACS. But in the last 10 years there has been an explosion of data in terms of how to best screen patients.
The move to primary HPV testing. The USPSTF3 initially had recommended looking into primary HPV screening, which is just using HPV testing by itself as the screening modality. But there was a lot of pushback at that time. I think a lot of people thought that we were not prepared to make that leap. Therefore, they endorsed screening with cytology by itself as well as HPV testing by itself, as well as co-testing; but in their recommendations, they made it very clear that they were leaning toward primary HPV screening.
A new patient age to begin screening. In 2020, the ACS put out their new guidelines,2 which are a significant departure from what we are used to—they are recommending that we start screening at 25 years of age. Like you said, Dr. Mutch, it doesn’t seem that long ago when we were screening people at age 18, or within 3 years of sexual intercourse. But the reason for it is that the rate of cervical cancer is extremely low under age 25, and other countries like the United Kingdom already do this.
The other major departure in the ACS guidelines is that they really are asking clinicians and screeners to focus on primary HPV screening. Overall, they have sort of doubled down on why they think primary HPV screening is so important.
ACOG sits sort of in the middle of the other recommendations. ACOG understands the value of primary HPV screening, but I don’t think that they are quite ready to recommend screening at age 25. If you look at their updated guidelines from April 2021,4 they state that we should continue a screening-starting age of 21 years. So there are some disparate views, but I am confident, Dr. Mutch, that in the next 2 to 3 years, there will be greater harmonization of these guidelines and less confusion for our providers. The greatest barrier is understanding the science and the comfort level of clinicians to go with just an HPV test, since for the last 40 years the Pap test has anchored gynecologic care in this country. And it took at least 10 years to get to what I consider to be widespread adoption to co-testing. The other thing that readers should recognize is that the Task Force is actually revisiting their cervical cancer screening guidance now, so expect another major revision.
Reimbursement and access are barriers. Reimbursement is a further real issue. We are now using one less test, but insurance companies may not reimburse when just the HPV test is used. The other issue is access to labs that can do the HPV testing.
Dr. Mutch: We used to see patients yearly and picked up a lot of adjunctive or additional illnesses. Now they are not being seen yearly it could impact negatively their overall health care. We need to understand that cervical cancer screening is simply a test, which should not eliminate other health care.
Dr. Huh: Yes, I think the extended interval between recommended HPV screenings scares people. I have been involved in these screening guidelines (and I can only speak for myself, not for my colleagues), but even I do think we made a leap to a longer interval way too quickly in this country. Screening changes are slow, and sometimes a glacial process. I think it can worry providers when we make rapid changes.
But this is a test that should not anchor the yearly visit. There are plenty of other reasons—and ACOG actually states this4—why patients should come for a wellness exam on a yearly basis. So I think our ObGyns in the United States need to recognize that, but I understand there are underlying concerns that if you extend intervals too long, (a) will patients come back, and (b), as a consequence, is the interval going to miss something in between? Those are real legitimate concerns.
Continue to: Management guidelines...
Management guidelines: The latest
Dr. Mutch: The ASCCP issued new management guidelines in 2019.5 Can you address what you feel are the most important updates?
Dr. Huh: Going back to 2002, we have revised these guidelines every 5 years. For this one, the revision came out a little bit later for various reasons, but the reason we revised it is because we collect new data that we think markedly changes our understanding of the disease process and natural history and the interventions for women that have preinvasive disease of the cervix.
Briefly, I think the biggest changes based on what we were hearing from our providers and users of our apps and algorithms was that our algorithms were becoming way too complicated, and they were. If you look over the last 10 years, the number of branch points on our algorithms basically quadrupled. If we incorporated the new data this time, the algorithms would be unworkable, and you could not use them on your phone because they would be too complicated.
So, we created a system where, in essence, providers have 5 choices for patients:
- treatment
- colposcopy
- follow-up in 1 year
- follow-up in 3 years
- follow-up in 5 years.
Those recommendations are based on what we call “clinically actionable thresholds”—basically, the percent chance of developing immediate CIN3 or worse. That threshold will probably change over time, but what we did is create a system that (a) makes it easier for the provider, (although they have to trust the system—and they can look under the hood and understand how we did this) and (b) allows us to create a foundation where we can add future technologies that use the same rubric or paradigm so that they still wind up getting the same result without having to go to another algorithm.
This new system is probably the most marked change in the history of the ASCCP management guidelines, but we did it to make it ultimately easier for providers going forward for the next 10 to 20 years. There are real opportunities, Dr. Mutch, in terms of how do we integrate this into the electronic medical record (EMR), and how do we pull data so clinicians don’t have to manually enter it.
The other difference is now there is a web-based application. Back in 2012, there were a lot of people that were not using EMRs. Now the majority of the country is, and so they actually are on a browser more than they are on their phone. We actually have an equally robust web platform that allows them to get the information that they need.
Dr. Mutch: I think that is really important—the utility of utilizing a mobile app, if you will, for triaging your patient with a specific test result so that patients are followed up at the proper interval, and that ultimately becomes cost-effective.
Dr. Huh: Yes, the app now is very different than the app that I think people are used to using for the last almost 10 years. You don’t put inputs, pull up the algorithm, and look at the outcome. This is different. You enter the patient’s age. You add their cytology, their HPV results, the clinical scenario that you are in, and then it puts out a recommendation of what to do next. Over time, we want to get away from an algorithm and for our providers to understand what the risk is and how that risk calculation then translates into a clinical recommendation.
Dr. Mutch: I think to utilize an app is almost necessary given the complexity of the triaging process so that it does become, in fact, the most cost-effective way to screen patients.
Dr. Huh: I would agree with that. There is a learning curve for whenever you see new technology. There was a learning curve for even ASCCP leadership as they tried to educate providers. I think people will ultimately see that this is a much better way of managing patients with cervical abnormalities, and I am hoping actually that we will use a similar platform for many other diseases that we manage in women’s health.
Continue to: Chipping away of the yearly exam...
Chipping away of the yearly exam
Dr. Mutch: With this moving away from the yearly exam and Pap test, women may not get yearly examinations. Do you feel that this could affect a stage migration to a higher stage at diagnosis, for instance, of a cervical cancer? Or that it might adversely impact other health issues?
Dr. Huh: I think that’s a good question. I am worried about the interval—I think 5 years is a bit long. I am more worried that patients will miss out on visits because they may think that they need to only come back for their Pap, even though they should be re-educated on that.
COVID-19 has made this a little hard for us to analyze because, clearly, we have had access to care issues. But I am a little concerned that we could see an uptick in invasive cancer rates in this country, including an uptick in the stage and more locally advanced cervical cancer because of the changes in the screening paradigms. But we don’t know that to be the case.
As with all screenings, the bottom line is you have to worry about what the false-negative and false-positive rates of screening are, and that affects everything. I want the readers to know that primary HPV should be used for screenings. It is not perfect, but it is much better than cytology alone. We need to think about how to better adapt screening in the age that we live in.
HPV self-sampling
Dr. Mutch: Could self-sampling for HPV testing, which obviously would be easier for the patient, and certainly useful in terms of screening, address some health care disparities with regard to cervical cancer?
Dr. Huh: The short answer is, yes. Self-sampling is not US Food and Drug Administration-approved in this country. It’s not being widely used without that approval. But there are multiple countries, including the United States, that have done lots of studies on this topic. There are many public health experts and champions for HPV self-sampling. I think we have learned, based on some studies, that the sensitivity is reasonable.6
I live in a part of the country that is woefully underserved; where you are there are pockets in Missouri that are woefully underserved as well. So the issue is, can we reduce these disparities and access to care with something like self-sampling? My personal feeling is I think that we can make a dent in that, and it is never going to fully replace screening, but it at least will allow us to reallocate our resources and attention to those women that are at highest risk for developing cervical cancer or precancer based on the self-sampling result.
I don’t think it will ever replace screening per se, but if we have an abnormal self-sampling test, we might say to that patient, “You really do need to come in to get re-tested or to get re-evaluated.” So it could be a better resource and use of our health care dollars and investments in terms of trying to reduce the incidence of cervical cancer. Of course the verdict is out, but I think there are a lot of people who would love to see this scenario.
If we screen and treat perfectly in this country, we would not even need the HPV vaccine when it comes to cervical cancer. That is how effective screening is. But, up to 50% to 60% of women in this country now still are underscreened or unscreened. We were talking about that number almost 25-30 years ago, Dr. Mutch. So access to screening is a big problem, but the other problem is how do you get patients in to be seen if they have an abnormal screening test? It’s not just about screening. It’s about screening, evaluation, and treatment; all 3 components are really important.
Continue to: Where do we stand with HPV vaccination?...
Where do we stand with HPV vaccination?
Dr. Mutch: Those are great points.
You brought up vaccination. We have a long way to go with regard to that, certainly in the United States, because of the various factions opposed to vaccination and so on. But do you think that vaccination has allowed us to decrease the incidence of cervical cancer?
Dr. Huh: Yes. There is clear evidence from the Nordic countries.7 There is emerging evidence from Australia.8 There is emerging evidence from other industrialized nations that clearly demonstrate vaccination’s positive effect in reducing the incidence of cervical cancer. None of this should be a surprise. Every population-based study that has been published with the HPV vaccine in populations that have a low frequency of vaccination have demonstrated substantial reductions in things like genital warts, abnormal Pap tests, precancer, and now evidence that there is a downward trend in terms of the incidence of cervical cancer.9
I don’t think that there is any debate anymore that vaccination is the way to go. Our challenge is about implementation and getting the vaccine to people. We still have a long way to go with that. There are parts of the world that are so affected by invasive cervical cancers; we need to get the vaccine to those parts of the world.
Dr. Mutch: What are the barriers to vaccination? How can we overcome those barriers?
Dr. Huh: There is a lot of criticism that we are not vaccinating more in the United States. However, the rates of vaccination are going up every single year. The pandemic may have blunted that rise a bit, but if you look at the vaccination curves, they are going up, not down. We need to continue to educate patients, parents, and pediatricians on the importance of vaccination.
Boys still get vaccinated less frequently than girls, so we have some work to do there. I think globally it is the issue of getting the vaccine to people, making sure that vaccine is available. The thing that I think will be the game-changer going forward is whether or not we will have evidence to indicate that 1 dose is as effective as 2 doses or 3 doses. If we can vaccinate boys and girls with just 1 dose, then in the next generation or two, we seriously might eradicate not just cervical cancer but a lot of HPV-related malignancies worldwide.
Educating patients, clinicians is key
Dr. Mutch: So it seems education, education, education, with regard to screening guidelines, with regard to the need for continued examinations, and that HPV testing is only a test, it does not supplant overall care. Finally, education regarding eradication of cervical cancer through vaccination.
Dr. Huh: That summarizes it well. We are still going to screen for cervical cancer. We are still going to vaccinate, and providers are still going to manage abnormal Pap tests. It is confusing because we are changing it up it seems every year or 2, so this conversation you and I are having is particularly important for clinicians to understand the basis of that. There has been an explosion of data that has come out in this area in the last decade.
Dr. Mutch: Thank you, Dr. Huh. I really appreciate your thoughts on this. As you all know, Dr. Huh has been President of the ASCCP and is instrumental in writing and disseminating these guidelines, so we are very grateful that he has consented to agree to come and talk with us today.
Dr. Huh: My pleasure. Thank you for inviting me. This was fun, and I have really enjoyed talking to you and participating. ●
- ASCCP Screening Guidelines. https://www.asccp.org /screening-guidelines. Accessed April 25, 2021.
- Fontham ET, Wolf AM, Church TR, et al. Cervical cancer screening for individuals at average risk: 2020 guidelines update from the American Cancer Society. CA Cancer J Clin. 2020;70:321-346. doi:10.3322/caac.21628.
- US Preventive Services Task Force. Screening for cervical cancer. US Preventive Services Task Force Recommendation Statement. JAMA. 2018;320:674-686. doi:10.1001/jama .2018.10897.
- American College of Obstetricians and Gynecologists. Practice Advisory: Updated cervical cancer screening guidelines. April 2022. https://www.acog.org/clinical/clinical-guidance /practice-advisory/articles/2021/04/updated-cervical -cancer-screening-guidelines. Accessed April 25, 2022.
- Perkins RB, Guido RS, Castle PE, et al; 2019 ASCCP RiskBased Management Consensus Guidelines Committee. 2019 ASCCP risk-based management consensus guidelines for abnormal cervical cancer screening tests and cancer precursors. J Low Genit Tract Dis. 2020;24:102-131. doi: 10 34 .1097/LGT.0000000000000525.
- Yeh PT, Kennedy CE, de Vuyst H, et al. Self-sampling for human papillomavirus (HPV) testing: a systematic review and meta-analysis. BMJ Global Health. 2019;4:e001351. doi:10.1136/ bmjgh-2018-001351.
- Kjaer SK, Nygard M, Dillner J, et al. A 12-year follow-up on the long-term effectiveness of the quadrivalent human papillomavirus vaccine in 4 Nordic countries. Clin Infect Dis. 2018;66:339-345. doi: 10.1093/cid/cix797.
- Patel C, Brotherton JM, Pillsbury A, et al. The impact of 10 years of human papillomavirus (HPV) vaccination in Australia: what additional disease burden will a nonvalent vaccine prevent? Euro Surveill. 2018;23:1700737. doi: 10 .2807/1560-7917.
- Falcaro M, Castanon A, Ndlela B, et al. The effects of the national HPV vaccination programme in England, UK, on cervical cancer and grade 3 cervical intraepithelial neoplasia incidence: a register-based observational study. 2021;398:2084-2092. doi.org/10.1016/S0140-6736(21) 02178-4.

David G. Mutch, MD: The cervical cancer screening guidelines, using Pap testing, have changed significantly since the times of yearly Paps and exams. Coupled with vaccination and new management guidelines (recommending HPV testing, etc), we actually hope that we are on the way to eradicating cervical cancer from our environment.
Screening: Current recommendations
Dr. Mutch: Warner, the American Society of Colposcopy and Cervical Pathology (ASCCP)1 endorses the cervical cancer screening guidelines for several professional organizations, including the American Cancer Society (ACS),2 the US Preventive Services Task Force (USPSTF),3 and the American College of Obstetricians and Gynecologists (ACOG).4 What are the current screening recommendations, as these organizations have disparate views?
Warner Huh, MD: There was a time, around 2012-2013, when for the first time ever, we had significant harmonization of the guidelines between ACOG and the USPSTF and ACS. But in the last 10 years there has been an explosion of data in terms of how to best screen patients.
The move to primary HPV testing. The USPSTF3 initially had recommended looking into primary HPV screening, which is just using HPV testing by itself as the screening modality. But there was a lot of pushback at that time. I think a lot of people thought that we were not prepared to make that leap. Therefore, they endorsed screening with cytology by itself as well as HPV testing by itself, as well as co-testing; but in their recommendations, they made it very clear that they were leaning toward primary HPV screening.
A new patient age to begin screening. In 2020, the ACS put out their new guidelines,2 which are a significant departure from what we are used to—they are recommending that we start screening at 25 years of age. Like you said, Dr. Mutch, it doesn’t seem that long ago when we were screening people at age 18, or within 3 years of sexual intercourse. But the reason for it is that the rate of cervical cancer is extremely low under age 25, and other countries like the United Kingdom already do this.
The other major departure in the ACS guidelines is that they really are asking clinicians and screeners to focus on primary HPV screening. Overall, they have sort of doubled down on why they think primary HPV screening is so important.
ACOG sits sort of in the middle of the other recommendations. ACOG understands the value of primary HPV screening, but I don’t think that they are quite ready to recommend screening at age 25. If you look at their updated guidelines from April 2021,4 they state that we should continue a screening-starting age of 21 years. So there are some disparate views, but I am confident, Dr. Mutch, that in the next 2 to 3 years, there will be greater harmonization of these guidelines and less confusion for our providers. The greatest barrier is understanding the science and the comfort level of clinicians to go with just an HPV test, since for the last 40 years the Pap test has anchored gynecologic care in this country. And it took at least 10 years to get to what I consider to be widespread adoption to co-testing. The other thing that readers should recognize is that the Task Force is actually revisiting their cervical cancer screening guidance now, so expect another major revision.
Reimbursement and access are barriers. Reimbursement is a further real issue. We are now using one less test, but insurance companies may not reimburse when just the HPV test is used. The other issue is access to labs that can do the HPV testing.
Dr. Mutch: We used to see patients yearly and picked up a lot of adjunctive or additional illnesses. Now they are not being seen yearly it could impact negatively their overall health care. We need to understand that cervical cancer screening is simply a test, which should not eliminate other health care.
Dr. Huh: Yes, I think the extended interval between recommended HPV screenings scares people. I have been involved in these screening guidelines (and I can only speak for myself, not for my colleagues), but even I do think we made a leap to a longer interval way too quickly in this country. Screening changes are slow, and sometimes a glacial process. I think it can worry providers when we make rapid changes.
But this is a test that should not anchor the yearly visit. There are plenty of other reasons—and ACOG actually states this4—why patients should come for a wellness exam on a yearly basis. So I think our ObGyns in the United States need to recognize that, but I understand there are underlying concerns that if you extend intervals too long, (a) will patients come back, and (b), as a consequence, is the interval going to miss something in between? Those are real legitimate concerns.
Continue to: Management guidelines...
Management guidelines: The latest
Dr. Mutch: The ASCCP issued new management guidelines in 2019.5 Can you address what you feel are the most important updates?
Dr. Huh: Going back to 2002, we have revised these guidelines every 5 years. For this one, the revision came out a little bit later for various reasons, but the reason we revised it is because we collect new data that we think markedly changes our understanding of the disease process and natural history and the interventions for women that have preinvasive disease of the cervix.
Briefly, I think the biggest changes based on what we were hearing from our providers and users of our apps and algorithms was that our algorithms were becoming way too complicated, and they were. If you look over the last 10 years, the number of branch points on our algorithms basically quadrupled. If we incorporated the new data this time, the algorithms would be unworkable, and you could not use them on your phone because they would be too complicated.
So, we created a system where, in essence, providers have 5 choices for patients:
- treatment
- colposcopy
- follow-up in 1 year
- follow-up in 3 years
- follow-up in 5 years.
Those recommendations are based on what we call “clinically actionable thresholds”—basically, the percent chance of developing immediate CIN3 or worse. That threshold will probably change over time, but what we did is create a system that (a) makes it easier for the provider, (although they have to trust the system—and they can look under the hood and understand how we did this) and (b) allows us to create a foundation where we can add future technologies that use the same rubric or paradigm so that they still wind up getting the same result without having to go to another algorithm.
This new system is probably the most marked change in the history of the ASCCP management guidelines, but we did it to make it ultimately easier for providers going forward for the next 10 to 20 years. There are real opportunities, Dr. Mutch, in terms of how do we integrate this into the electronic medical record (EMR), and how do we pull data so clinicians don’t have to manually enter it.
The other difference is now there is a web-based application. Back in 2012, there were a lot of people that were not using EMRs. Now the majority of the country is, and so they actually are on a browser more than they are on their phone. We actually have an equally robust web platform that allows them to get the information that they need.
Dr. Mutch: I think that is really important—the utility of utilizing a mobile app, if you will, for triaging your patient with a specific test result so that patients are followed up at the proper interval, and that ultimately becomes cost-effective.
Dr. Huh: Yes, the app now is very different than the app that I think people are used to using for the last almost 10 years. You don’t put inputs, pull up the algorithm, and look at the outcome. This is different. You enter the patient’s age. You add their cytology, their HPV results, the clinical scenario that you are in, and then it puts out a recommendation of what to do next. Over time, we want to get away from an algorithm and for our providers to understand what the risk is and how that risk calculation then translates into a clinical recommendation.
Dr. Mutch: I think to utilize an app is almost necessary given the complexity of the triaging process so that it does become, in fact, the most cost-effective way to screen patients.
Dr. Huh: I would agree with that. There is a learning curve for whenever you see new technology. There was a learning curve for even ASCCP leadership as they tried to educate providers. I think people will ultimately see that this is a much better way of managing patients with cervical abnormalities, and I am hoping actually that we will use a similar platform for many other diseases that we manage in women’s health.
Continue to: Chipping away of the yearly exam...
Chipping away of the yearly exam
Dr. Mutch: With this moving away from the yearly exam and Pap test, women may not get yearly examinations. Do you feel that this could affect a stage migration to a higher stage at diagnosis, for instance, of a cervical cancer? Or that it might adversely impact other health issues?
Dr. Huh: I think that’s a good question. I am worried about the interval—I think 5 years is a bit long. I am more worried that patients will miss out on visits because they may think that they need to only come back for their Pap, even though they should be re-educated on that.
COVID-19 has made this a little hard for us to analyze because, clearly, we have had access to care issues. But I am a little concerned that we could see an uptick in invasive cancer rates in this country, including an uptick in the stage and more locally advanced cervical cancer because of the changes in the screening paradigms. But we don’t know that to be the case.
As with all screenings, the bottom line is you have to worry about what the false-negative and false-positive rates of screening are, and that affects everything. I want the readers to know that primary HPV should be used for screenings. It is not perfect, but it is much better than cytology alone. We need to think about how to better adapt screening in the age that we live in.
HPV self-sampling
Dr. Mutch: Could self-sampling for HPV testing, which obviously would be easier for the patient, and certainly useful in terms of screening, address some health care disparities with regard to cervical cancer?
Dr. Huh: The short answer is, yes. Self-sampling is not US Food and Drug Administration-approved in this country. It’s not being widely used without that approval. But there are multiple countries, including the United States, that have done lots of studies on this topic. There are many public health experts and champions for HPV self-sampling. I think we have learned, based on some studies, that the sensitivity is reasonable.6
I live in a part of the country that is woefully underserved; where you are there are pockets in Missouri that are woefully underserved as well. So the issue is, can we reduce these disparities and access to care with something like self-sampling? My personal feeling is I think that we can make a dent in that, and it is never going to fully replace screening, but it at least will allow us to reallocate our resources and attention to those women that are at highest risk for developing cervical cancer or precancer based on the self-sampling result.
I don’t think it will ever replace screening per se, but if we have an abnormal self-sampling test, we might say to that patient, “You really do need to come in to get re-tested or to get re-evaluated.” So it could be a better resource and use of our health care dollars and investments in terms of trying to reduce the incidence of cervical cancer. Of course the verdict is out, but I think there are a lot of people who would love to see this scenario.
If we screen and treat perfectly in this country, we would not even need the HPV vaccine when it comes to cervical cancer. That is how effective screening is. But, up to 50% to 60% of women in this country now still are underscreened or unscreened. We were talking about that number almost 25-30 years ago, Dr. Mutch. So access to screening is a big problem, but the other problem is how do you get patients in to be seen if they have an abnormal screening test? It’s not just about screening. It’s about screening, evaluation, and treatment; all 3 components are really important.
Continue to: Where do we stand with HPV vaccination?...
Where do we stand with HPV vaccination?
Dr. Mutch: Those are great points.
You brought up vaccination. We have a long way to go with regard to that, certainly in the United States, because of the various factions opposed to vaccination and so on. But do you think that vaccination has allowed us to decrease the incidence of cervical cancer?
Dr. Huh: Yes. There is clear evidence from the Nordic countries.7 There is emerging evidence from Australia.8 There is emerging evidence from other industrialized nations that clearly demonstrate vaccination’s positive effect in reducing the incidence of cervical cancer. None of this should be a surprise. Every population-based study that has been published with the HPV vaccine in populations that have a low frequency of vaccination have demonstrated substantial reductions in things like genital warts, abnormal Pap tests, precancer, and now evidence that there is a downward trend in terms of the incidence of cervical cancer.9
I don’t think that there is any debate anymore that vaccination is the way to go. Our challenge is about implementation and getting the vaccine to people. We still have a long way to go with that. There are parts of the world that are so affected by invasive cervical cancers; we need to get the vaccine to those parts of the world.
Dr. Mutch: What are the barriers to vaccination? How can we overcome those barriers?
Dr. Huh: There is a lot of criticism that we are not vaccinating more in the United States. However, the rates of vaccination are going up every single year. The pandemic may have blunted that rise a bit, but if you look at the vaccination curves, they are going up, not down. We need to continue to educate patients, parents, and pediatricians on the importance of vaccination.
Boys still get vaccinated less frequently than girls, so we have some work to do there. I think globally it is the issue of getting the vaccine to people, making sure that vaccine is available. The thing that I think will be the game-changer going forward is whether or not we will have evidence to indicate that 1 dose is as effective as 2 doses or 3 doses. If we can vaccinate boys and girls with just 1 dose, then in the next generation or two, we seriously might eradicate not just cervical cancer but a lot of HPV-related malignancies worldwide.
Educating patients, clinicians is key
Dr. Mutch: So it seems education, education, education, with regard to screening guidelines, with regard to the need for continued examinations, and that HPV testing is only a test, it does not supplant overall care. Finally, education regarding eradication of cervical cancer through vaccination.
Dr. Huh: That summarizes it well. We are still going to screen for cervical cancer. We are still going to vaccinate, and providers are still going to manage abnormal Pap tests. It is confusing because we are changing it up it seems every year or 2, so this conversation you and I are having is particularly important for clinicians to understand the basis of that. There has been an explosion of data that has come out in this area in the last decade.
Dr. Mutch: Thank you, Dr. Huh. I really appreciate your thoughts on this. As you all know, Dr. Huh has been President of the ASCCP and is instrumental in writing and disseminating these guidelines, so we are very grateful that he has consented to agree to come and talk with us today.
Dr. Huh: My pleasure. Thank you for inviting me. This was fun, and I have really enjoyed talking to you and participating. ●

David G. Mutch, MD: The cervical cancer screening guidelines, using Pap testing, have changed significantly since the times of yearly Paps and exams. Coupled with vaccination and new management guidelines (recommending HPV testing, etc), we actually hope that we are on the way to eradicating cervical cancer from our environment.
Screening: Current recommendations
Dr. Mutch: Warner, the American Society of Colposcopy and Cervical Pathology (ASCCP)1 endorses the cervical cancer screening guidelines for several professional organizations, including the American Cancer Society (ACS),2 the US Preventive Services Task Force (USPSTF),3 and the American College of Obstetricians and Gynecologists (ACOG).4 What are the current screening recommendations, as these organizations have disparate views?
Warner Huh, MD: There was a time, around 2012-2013, when for the first time ever, we had significant harmonization of the guidelines between ACOG and the USPSTF and ACS. But in the last 10 years there has been an explosion of data in terms of how to best screen patients.
The move to primary HPV testing. The USPSTF3 initially had recommended looking into primary HPV screening, which is just using HPV testing by itself as the screening modality. But there was a lot of pushback at that time. I think a lot of people thought that we were not prepared to make that leap. Therefore, they endorsed screening with cytology by itself as well as HPV testing by itself, as well as co-testing; but in their recommendations, they made it very clear that they were leaning toward primary HPV screening.
A new patient age to begin screening. In 2020, the ACS put out their new guidelines,2 which are a significant departure from what we are used to—they are recommending that we start screening at 25 years of age. Like you said, Dr. Mutch, it doesn’t seem that long ago when we were screening people at age 18, or within 3 years of sexual intercourse. But the reason for it is that the rate of cervical cancer is extremely low under age 25, and other countries like the United Kingdom already do this.
The other major departure in the ACS guidelines is that they really are asking clinicians and screeners to focus on primary HPV screening. Overall, they have sort of doubled down on why they think primary HPV screening is so important.
ACOG sits sort of in the middle of the other recommendations. ACOG understands the value of primary HPV screening, but I don’t think that they are quite ready to recommend screening at age 25. If you look at their updated guidelines from April 2021,4 they state that we should continue a screening-starting age of 21 years. So there are some disparate views, but I am confident, Dr. Mutch, that in the next 2 to 3 years, there will be greater harmonization of these guidelines and less confusion for our providers. The greatest barrier is understanding the science and the comfort level of clinicians to go with just an HPV test, since for the last 40 years the Pap test has anchored gynecologic care in this country. And it took at least 10 years to get to what I consider to be widespread adoption to co-testing. The other thing that readers should recognize is that the Task Force is actually revisiting their cervical cancer screening guidance now, so expect another major revision.
Reimbursement and access are barriers. Reimbursement is a further real issue. We are now using one less test, but insurance companies may not reimburse when just the HPV test is used. The other issue is access to labs that can do the HPV testing.
Dr. Mutch: We used to see patients yearly and picked up a lot of adjunctive or additional illnesses. Now they are not being seen yearly it could impact negatively their overall health care. We need to understand that cervical cancer screening is simply a test, which should not eliminate other health care.
Dr. Huh: Yes, I think the extended interval between recommended HPV screenings scares people. I have been involved in these screening guidelines (and I can only speak for myself, not for my colleagues), but even I do think we made a leap to a longer interval way too quickly in this country. Screening changes are slow, and sometimes a glacial process. I think it can worry providers when we make rapid changes.
But this is a test that should not anchor the yearly visit. There are plenty of other reasons—and ACOG actually states this4—why patients should come for a wellness exam on a yearly basis. So I think our ObGyns in the United States need to recognize that, but I understand there are underlying concerns that if you extend intervals too long, (a) will patients come back, and (b), as a consequence, is the interval going to miss something in between? Those are real legitimate concerns.
Continue to: Management guidelines...
Management guidelines: The latest
Dr. Mutch: The ASCCP issued new management guidelines in 2019.5 Can you address what you feel are the most important updates?
Dr. Huh: Going back to 2002, we have revised these guidelines every 5 years. For this one, the revision came out a little bit later for various reasons, but the reason we revised it is because we collect new data that we think markedly changes our understanding of the disease process and natural history and the interventions for women that have preinvasive disease of the cervix.
Briefly, I think the biggest changes based on what we were hearing from our providers and users of our apps and algorithms was that our algorithms were becoming way too complicated, and they were. If you look over the last 10 years, the number of branch points on our algorithms basically quadrupled. If we incorporated the new data this time, the algorithms would be unworkable, and you could not use them on your phone because they would be too complicated.
So, we created a system where, in essence, providers have 5 choices for patients:
- treatment
- colposcopy
- follow-up in 1 year
- follow-up in 3 years
- follow-up in 5 years.
Those recommendations are based on what we call “clinically actionable thresholds”—basically, the percent chance of developing immediate CIN3 or worse. That threshold will probably change over time, but what we did is create a system that (a) makes it easier for the provider, (although they have to trust the system—and they can look under the hood and understand how we did this) and (b) allows us to create a foundation where we can add future technologies that use the same rubric or paradigm so that they still wind up getting the same result without having to go to another algorithm.
This new system is probably the most marked change in the history of the ASCCP management guidelines, but we did it to make it ultimately easier for providers going forward for the next 10 to 20 years. There are real opportunities, Dr. Mutch, in terms of how do we integrate this into the electronic medical record (EMR), and how do we pull data so clinicians don’t have to manually enter it.
The other difference is now there is a web-based application. Back in 2012, there were a lot of people that were not using EMRs. Now the majority of the country is, and so they actually are on a browser more than they are on their phone. We actually have an equally robust web platform that allows them to get the information that they need.
Dr. Mutch: I think that is really important—the utility of utilizing a mobile app, if you will, for triaging your patient with a specific test result so that patients are followed up at the proper interval, and that ultimately becomes cost-effective.
Dr. Huh: Yes, the app now is very different than the app that I think people are used to using for the last almost 10 years. You don’t put inputs, pull up the algorithm, and look at the outcome. This is different. You enter the patient’s age. You add their cytology, their HPV results, the clinical scenario that you are in, and then it puts out a recommendation of what to do next. Over time, we want to get away from an algorithm and for our providers to understand what the risk is and how that risk calculation then translates into a clinical recommendation.
Dr. Mutch: I think to utilize an app is almost necessary given the complexity of the triaging process so that it does become, in fact, the most cost-effective way to screen patients.
Dr. Huh: I would agree with that. There is a learning curve for whenever you see new technology. There was a learning curve for even ASCCP leadership as they tried to educate providers. I think people will ultimately see that this is a much better way of managing patients with cervical abnormalities, and I am hoping actually that we will use a similar platform for many other diseases that we manage in women’s health.
Continue to: Chipping away of the yearly exam...
Chipping away of the yearly exam
Dr. Mutch: With this moving away from the yearly exam and Pap test, women may not get yearly examinations. Do you feel that this could affect a stage migration to a higher stage at diagnosis, for instance, of a cervical cancer? Or that it might adversely impact other health issues?
Dr. Huh: I think that’s a good question. I am worried about the interval—I think 5 years is a bit long. I am more worried that patients will miss out on visits because they may think that they need to only come back for their Pap, even though they should be re-educated on that.
COVID-19 has made this a little hard for us to analyze because, clearly, we have had access to care issues. But I am a little concerned that we could see an uptick in invasive cancer rates in this country, including an uptick in the stage and more locally advanced cervical cancer because of the changes in the screening paradigms. But we don’t know that to be the case.
As with all screenings, the bottom line is you have to worry about what the false-negative and false-positive rates of screening are, and that affects everything. I want the readers to know that primary HPV should be used for screenings. It is not perfect, but it is much better than cytology alone. We need to think about how to better adapt screening in the age that we live in.
HPV self-sampling
Dr. Mutch: Could self-sampling for HPV testing, which obviously would be easier for the patient, and certainly useful in terms of screening, address some health care disparities with regard to cervical cancer?
Dr. Huh: The short answer is, yes. Self-sampling is not US Food and Drug Administration-approved in this country. It’s not being widely used without that approval. But there are multiple countries, including the United States, that have done lots of studies on this topic. There are many public health experts and champions for HPV self-sampling. I think we have learned, based on some studies, that the sensitivity is reasonable.6
I live in a part of the country that is woefully underserved; where you are there are pockets in Missouri that are woefully underserved as well. So the issue is, can we reduce these disparities and access to care with something like self-sampling? My personal feeling is I think that we can make a dent in that, and it is never going to fully replace screening, but it at least will allow us to reallocate our resources and attention to those women that are at highest risk for developing cervical cancer or precancer based on the self-sampling result.
I don’t think it will ever replace screening per se, but if we have an abnormal self-sampling test, we might say to that patient, “You really do need to come in to get re-tested or to get re-evaluated.” So it could be a better resource and use of our health care dollars and investments in terms of trying to reduce the incidence of cervical cancer. Of course the verdict is out, but I think there are a lot of people who would love to see this scenario.
If we screen and treat perfectly in this country, we would not even need the HPV vaccine when it comes to cervical cancer. That is how effective screening is. But, up to 50% to 60% of women in this country now still are underscreened or unscreened. We were talking about that number almost 25-30 years ago, Dr. Mutch. So access to screening is a big problem, but the other problem is how do you get patients in to be seen if they have an abnormal screening test? It’s not just about screening. It’s about screening, evaluation, and treatment; all 3 components are really important.
Continue to: Where do we stand with HPV vaccination?...
Where do we stand with HPV vaccination?
Dr. Mutch: Those are great points.
You brought up vaccination. We have a long way to go with regard to that, certainly in the United States, because of the various factions opposed to vaccination and so on. But do you think that vaccination has allowed us to decrease the incidence of cervical cancer?
Dr. Huh: Yes. There is clear evidence from the Nordic countries.7 There is emerging evidence from Australia.8 There is emerging evidence from other industrialized nations that clearly demonstrate vaccination’s positive effect in reducing the incidence of cervical cancer. None of this should be a surprise. Every population-based study that has been published with the HPV vaccine in populations that have a low frequency of vaccination have demonstrated substantial reductions in things like genital warts, abnormal Pap tests, precancer, and now evidence that there is a downward trend in terms of the incidence of cervical cancer.9
I don’t think that there is any debate anymore that vaccination is the way to go. Our challenge is about implementation and getting the vaccine to people. We still have a long way to go with that. There are parts of the world that are so affected by invasive cervical cancers; we need to get the vaccine to those parts of the world.
Dr. Mutch: What are the barriers to vaccination? How can we overcome those barriers?
Dr. Huh: There is a lot of criticism that we are not vaccinating more in the United States. However, the rates of vaccination are going up every single year. The pandemic may have blunted that rise a bit, but if you look at the vaccination curves, they are going up, not down. We need to continue to educate patients, parents, and pediatricians on the importance of vaccination.
Boys still get vaccinated less frequently than girls, so we have some work to do there. I think globally it is the issue of getting the vaccine to people, making sure that vaccine is available. The thing that I think will be the game-changer going forward is whether or not we will have evidence to indicate that 1 dose is as effective as 2 doses or 3 doses. If we can vaccinate boys and girls with just 1 dose, then in the next generation or two, we seriously might eradicate not just cervical cancer but a lot of HPV-related malignancies worldwide.
Educating patients, clinicians is key
Dr. Mutch: So it seems education, education, education, with regard to screening guidelines, with regard to the need for continued examinations, and that HPV testing is only a test, it does not supplant overall care. Finally, education regarding eradication of cervical cancer through vaccination.
Dr. Huh: That summarizes it well. We are still going to screen for cervical cancer. We are still going to vaccinate, and providers are still going to manage abnormal Pap tests. It is confusing because we are changing it up it seems every year or 2, so this conversation you and I are having is particularly important for clinicians to understand the basis of that. There has been an explosion of data that has come out in this area in the last decade.
Dr. Mutch: Thank you, Dr. Huh. I really appreciate your thoughts on this. As you all know, Dr. Huh has been President of the ASCCP and is instrumental in writing and disseminating these guidelines, so we are very grateful that he has consented to agree to come and talk with us today.
Dr. Huh: My pleasure. Thank you for inviting me. This was fun, and I have really enjoyed talking to you and participating. ●
- ASCCP Screening Guidelines. https://www.asccp.org /screening-guidelines. Accessed April 25, 2021.
- Fontham ET, Wolf AM, Church TR, et al. Cervical cancer screening for individuals at average risk: 2020 guidelines update from the American Cancer Society. CA Cancer J Clin. 2020;70:321-346. doi:10.3322/caac.21628.
- US Preventive Services Task Force. Screening for cervical cancer. US Preventive Services Task Force Recommendation Statement. JAMA. 2018;320:674-686. doi:10.1001/jama .2018.10897.
- American College of Obstetricians and Gynecologists. Practice Advisory: Updated cervical cancer screening guidelines. April 2022. https://www.acog.org/clinical/clinical-guidance /practice-advisory/articles/2021/04/updated-cervical -cancer-screening-guidelines. Accessed April 25, 2022.
- Perkins RB, Guido RS, Castle PE, et al; 2019 ASCCP RiskBased Management Consensus Guidelines Committee. 2019 ASCCP risk-based management consensus guidelines for abnormal cervical cancer screening tests and cancer precursors. J Low Genit Tract Dis. 2020;24:102-131. doi: 10 34 .1097/LGT.0000000000000525.
- Yeh PT, Kennedy CE, de Vuyst H, et al. Self-sampling for human papillomavirus (HPV) testing: a systematic review and meta-analysis. BMJ Global Health. 2019;4:e001351. doi:10.1136/ bmjgh-2018-001351.
- Kjaer SK, Nygard M, Dillner J, et al. A 12-year follow-up on the long-term effectiveness of the quadrivalent human papillomavirus vaccine in 4 Nordic countries. Clin Infect Dis. 2018;66:339-345. doi: 10.1093/cid/cix797.
- Patel C, Brotherton JM, Pillsbury A, et al. The impact of 10 years of human papillomavirus (HPV) vaccination in Australia: what additional disease burden will a nonvalent vaccine prevent? Euro Surveill. 2018;23:1700737. doi: 10 .2807/1560-7917.
- Falcaro M, Castanon A, Ndlela B, et al. The effects of the national HPV vaccination programme in England, UK, on cervical cancer and grade 3 cervical intraepithelial neoplasia incidence: a register-based observational study. 2021;398:2084-2092. doi.org/10.1016/S0140-6736(21) 02178-4.
- ASCCP Screening Guidelines. https://www.asccp.org /screening-guidelines. Accessed April 25, 2021.
- Fontham ET, Wolf AM, Church TR, et al. Cervical cancer screening for individuals at average risk: 2020 guidelines update from the American Cancer Society. CA Cancer J Clin. 2020;70:321-346. doi:10.3322/caac.21628.
- US Preventive Services Task Force. Screening for cervical cancer. US Preventive Services Task Force Recommendation Statement. JAMA. 2018;320:674-686. doi:10.1001/jama .2018.10897.
- American College of Obstetricians and Gynecologists. Practice Advisory: Updated cervical cancer screening guidelines. April 2022. https://www.acog.org/clinical/clinical-guidance /practice-advisory/articles/2021/04/updated-cervical -cancer-screening-guidelines. Accessed April 25, 2022.
- Perkins RB, Guido RS, Castle PE, et al; 2019 ASCCP RiskBased Management Consensus Guidelines Committee. 2019 ASCCP risk-based management consensus guidelines for abnormal cervical cancer screening tests and cancer precursors. J Low Genit Tract Dis. 2020;24:102-131. doi: 10 34 .1097/LGT.0000000000000525.
- Yeh PT, Kennedy CE, de Vuyst H, et al. Self-sampling for human papillomavirus (HPV) testing: a systematic review and meta-analysis. BMJ Global Health. 2019;4:e001351. doi:10.1136/ bmjgh-2018-001351.
- Kjaer SK, Nygard M, Dillner J, et al. A 12-year follow-up on the long-term effectiveness of the quadrivalent human papillomavirus vaccine in 4 Nordic countries. Clin Infect Dis. 2018;66:339-345. doi: 10.1093/cid/cix797.
- Patel C, Brotherton JM, Pillsbury A, et al. The impact of 10 years of human papillomavirus (HPV) vaccination in Australia: what additional disease burden will a nonvalent vaccine prevent? Euro Surveill. 2018;23:1700737. doi: 10 .2807/1560-7917.
- Falcaro M, Castanon A, Ndlela B, et al. The effects of the national HPV vaccination programme in England, UK, on cervical cancer and grade 3 cervical intraepithelial neoplasia incidence: a register-based observational study. 2021;398:2084-2092. doi.org/10.1016/S0140-6736(21) 02178-4.
Commonly used antibiotics in ObGyn practice
In this article, I provide a simplified, practical review of the principal antibiotics that we use on a daily basis to treat bacterial infections. The antibiotics are listed in alphabetical order, either individually or by group. I focus first on the mechanism of action and spectrum of activity of the drugs used against the usual pelvic pathogens (TABLE).1 I then review their principal adverse effects, relative cost (categorized as low, intermediate, and high), and the key indications for these drugs in obstetrics and gynecology. In a forthcoming 2-part companion article, I will review how to select specific antibiotics and their dosing regimens for the most commonly encountered bacterial infections in our clinical practice.

Aminoglycoside antibiotics
The aminoglycosides include amikacin, gentamicin, plazomicin, and tobramycin.2,3 The 2 agents most commonly used in our specialty are amikacin and gentamicin. The drugs may be administered intramuscularly or intravenously, and they specifically target aerobic gram-negative bacilli. They also provide coverage against staphylococci and gonococci. Ototoxicity and nephrotoxicity are their principal adverse effects.
Aminoglycosides are used primarily as single agents to treat pyelonephritis caused by highly resistant bacteria and in combination with agents such as clindamycin and metronidazole to treat polymicrobial infections, including chorioamnionitis, puerperal endometritis, and pelvic inflammatory disease. Of all the aminoglycosides, gentamicin is clearly the least expensive.
Carbapenems
The original carbapenem widely introduced into clinical practice was imipenem-cilastatin. Imipenem, the active antibiotic, inhibits bacterial cell wall synthesis. Cilastatin inhibits renal dehydropeptidase I and, thereby, slows the metabolism of imipenem by the kidney. Other carbapenems include meropenem and ertapenem.
The carbapenems have the widest spectrum of activity against the pelvic pathogens of any antibiotic. They provide excellent coverage of aerobic and anaerobic gram-positive cocci and aerobic and anaerobic gram-negative bacilli. They do not cover methicillin-resistant Staphylococcus aureus (MRSA) and the enterococci very well.
A major adverse effect of the carbapenems is an allergic reaction, including anaphylaxis and Stevens-Johnson syndrome, and there is some minimal cross-sensitivity with the β-lactam antibiotics. Other important, but fortunately rare, adverse effects include neurotoxicity, hepatotoxicity, and Clostridium difficile colitis.4
As a group, the carbapenems are relatively more expensive than most other agents. Their principal application in our specialty is for single-agent treatment of serious polymicrobial infections, such as puerperal endometritis, pelvic cellulitis, and pelvic abscess, especially in patients who have a contraindication to the use of combination antibiotic regimens that include an aminoglycoside.1,2
Cephalosporins
The cephalosporins are β-lactam antibiotics that act by disrupting the synthesis of the bacterial cell wall. They may be administered orally, intramuscularly, and intravenously. The most common adverse effects associated with these agents are an allergic reaction, which can range from a mild rash to anaphylaxis and the Stevens-Johnson syndrome; central nervous system toxicity; and antibiotic-induced diarrhea, including C difficile colitis.1,2,4
This group of antibiotics can be confusing because it includes so many agents, and their spectrum of activity varies. I find it helpful to think about the coverage of these agents as limited spectrum versus intermediate spectrum versus extended spectrum.
The limited-spectrum cephalosporin prototypes are cephalexin (oral administration) and cefazolin (parenteral administration). This group of cephalosporins provides excellent coverage of aerobic and anaerobic gram-positive cocci. They are excellent against staphylococci, except for MRSA. Coverage is moderate for aerobic gram-negative bacilli but only limited for anaerobic gram-negative bacilli. They do not cover the enterococci. In our specialty, their principal application is for treatment of mastitis, urinary tract infections (UTIs), and wound infections and for prophylaxis against group B streptococcus (GBS) infection and post-cesarean infection.2,5 The cost of these drugs is relatively low.
The prototypes of the intermediate-spectrum cephalosporins are cefixime (oral) and ceftriaxone (parenteral). Both drugs have strong activity against aerobic and anaerobic streptococci, Neisseria gonorrhoeae, most aerobic gram-negative bacilli, and Treponema pallidum (principally, ceftriaxone). They are not consistently effective against staphylococci, particularly MRSA, and enterococci. Their key indications in obstetrics and gynecology are treatment of gonorrhea, syphilis (in penicillin-allergic patients), and acute pyelonephritis. Compared with the limited-spectrum cephalosporins, these antibiotics are moderately expensive.1,2
The 3 extended-spectrum cephalosporins used most commonly in our specialty are cefepime, cefotetan, and cefoxitin. These agents are administered intramuscularly and intravenously, and they provide very good coverage against aerobic and anaerobic gram-positive cocci, with the exception of staphylococci and enterococci. They have very good coverage against most gram-negative aerobic bacilli and excellent coverage against anerobic microorganisms. Their primary application in our specialty is for single-agent treatment of polymicrobial infections, such as puerperal endometritis and pelvic cellulitis. When used in combination with doxycycline, they are valuable in treating pelvic inflammatory disease. These drugs are more expensive than the limited-spectrum or intermediate-spectrum agents. They should not be used routinely as prophylaxis for pelvic surgery.1,2,5
Continue to: Fluorinated quinolones...
Fluorinated quinolones
The fluorinated quinolones include several agents, but the 3 most commonly used in our specialty are ciprofloxacin, ofloxacin, and levofloxacin. All 3 drugs can be administered orally; ciprofloxacin and levofloxacin also are available in intravenous formulations. These drugs interfere with bacterial protein synthesis by targeting DNA gyrase, an enzyme that introduces negative supertwists into DNA and separates interlocked DNA molecules.
These drugs provide excellent coverage against gram-negative bacilli, including Haemophilus influenzae; gram-negative cocci, such as N gonorrhoeae, Neisseria meningitidis, and Moraxella catarrhalis; and many staphylococci species. Levofloxacin, but not the other 2 drugs, provides moderate coverage against anaerobes. Ofloxacin and levofloxacin are active against chlamydia. Levofloxacin also covers the mycoplasma organisms that are responsible for atypical pneumonia.
As a group, the fluorinated quinolones are moderately expensive. The most likely adverse effects with these agents are gastrointestinal (GI) upset, headache, agitation, and sleep disturbance. Allergic reactions are rare. These drugs are of primary value in our specialty in treating gonorrhea, chlamydia, complicated UTIs, and respiratory tract infections.1,2,6
The penicillins
Penicillin
Penicillin, a β-lactam antibiotic, was one of the first antibiotics developed and employed in clinical practice. It may be administered orally, intramuscularly, and intravenously. Penicillin exerts its effect by interfering with bacterial cell wall synthesis. Its principal spectrum of activity is against aerobic streptococci, such as group A and B streptococcus; most anaerobic gram-positive cocci that are present in the vaginal flora; some anaerobic gram-negative bacilli; and T pallidum. Penicillin is not effective against the majority of staphylococci species, enterococci, or aerobic gram-negative bacilli, such as Escherichia coli.
Penicillin’s major adverse effect is an allergic reaction, experienced by less than 10% of recipients.7 Most reactions are mild and are characterized by a morbilliform skin rash. However, some reactions are severe and take the form of an urticarial skin eruption, laryngospasm, bronchospasm, and overt anaphylaxis. The cost of both oral and parenteral penicillin formulations is very low. In obstetrics and gynecology, penicillin is used primarily for the treatment of group A and B streptococci infections, clostridial infections, and syphilis.1,2
Ampicillin and amoxicillin
The β-lactam antibiotics ampicillin and amoxicillin also act by interfering with bacterial cell wall synthesis. Amoxicillin is administered orally; ampicillin may be administered orally, intramuscularly, and intravenously. Their spectrum of activity includes group A and B streptococci, enterococci, most anaerobic gram-positive cocci, some anaerobic gram-negative bacilli, many aerobic gram-negative bacilli, and clostridial organisms.
Like penicillin, ampicillin and amoxicillin may cause allergic reactions that range from mild rashes to anaphylaxis. Unlike the more narrow-spectrum penicillin, they may cause antibiotic-associated diarrhea, including C difficile colitis,4 and they may eliminate part of the normal vaginal flora and stimulate an overgrowth of yeast organisms in the vagina. The cost of ampicillin and amoxicillin is very low. These 2 agents are used primarily for treatment of group A and B streptococci infections and some UTIs, particularly those caused by enterococci.1,2
Dicloxacillin sodium
This penicillin derivative disrupts bacterial cell wall synthesis and targets primarily aerobic gram-positive cocci, particularly staphylococci species. The antibiotic is not active against MRSA. The principal adverse effects of dicloxacillin sodium are an allergic reaction and GI upset. The drug is very inexpensive.
The key application for dicloxacillin sodium in our specialty is for treatment of puerperal mastitis.1
Continue to: Extended-spectrum penicillins...
Extended-spectrum penicillins
Three interesting combination extended-spectrum penicillins are used widely in our specialty. They are ampicillin/sulbactam, amoxicillin/clavulanate, and piperacillin/tazobactam. Ampicillin/sulbactam may be administered intramuscularly and intravenously. Piperacillin/tazobactam is administered intravenously; amoxicillin/clavulanate is administered orally.
Clavulanate, sulbactam, and tazobactam are β-lactamase inhibitors. When added to the parent antibiotic (amoxicillin, ampicillin, and piperacillin, respectively), they significantly enhance the parent drug’s spectrum of activity. These agents interfere with bacterial cell wall synthesis. They provide excellent coverage of aerobic gram-positive cocci, including enterococci; anaerobic gram-positive cocci; anaerobic gram-negative bacilli; and aerobic gram-negative bacilli. Their principal adverse effects include allergic reactions and antibiotic-associated diarrhea. They are moderately expensive.
The principal application of ampicillin/sulbactam and piperacillin/tazobactam in our specialty is as single agents for treatment of puerperal endometritis, postoperative pelvic cellulitis, and pyelonephritis. The usual role for amoxicillin/clavulanate is for oral treatment of complicated UTIs, including pyelonephritis in early pregnancy, and for outpatient therapy of mild to moderately severe endometritis following delivery or pregnancy termination.
Macrolides, monobactams, and additional antibiotics
Azithromycin
Azithromycin is a macrolide antibiotic that is in the same class as erythromycin and clindamycin. In our specialty, it has largely replaced erythromycin because of its more convenient dosage schedule and its better tolerability. It inhibits bacterial protein synthesis, and it is available in both an oral and intravenous formulation.
Azithromycin has an excellent spectrum of activity against the 3 major microorganisms that cause otitis media, sinusitis, and bronchitis: Streptococcus pneumoniae, H influenzae, and M catarrhalis. It also provides excellent coverage of Chlamydia trachomatis, Mycoplasma pneumoniae, and genital mycoplasmas; in high doses it provides modest coverage against gonorrhea.8 Unlike erythromycin, it has minimal GI toxicity and is usually very well tolerated by most patients. One unusual, but very important, adverse effect of the drug is prolongation of the Q-T interval.9
Azithromycin is now available in generic form and is relatively inexpensive. As a single agent, its principal applications in our specialty are for treatment of respiratory tract infections such as otitis media, sinusitis, and acute bronchitis and for treatment of chlamydia urethritis and endocervicitis.8,10 In combination with ampicillin, azithromycin is used as prophylaxis in patients with preterm premature rupture of membranes (PPROM), and, in combination with cefazolin, it is used for prophylaxis in patients undergoing cesarean delivery.1,2,5
Aztreonam
Aztreonam is a monobactam antibiotic. Like the cephalosporins and penicillins, aztreonam inhibits bacterial cell wall synthesis. It may be administered intramuscularly and intravenously, and its principal spectrum of activity is against aerobic gram-negative bacilli, which is similar to the aminoglycosides’ spectrum.
Aztreonam’s most likely adverse effects include phlebitis at the injection site, allergy, GI upset, and diarrhea. The drug is moderately expensive. In our specialty, aztreonam could be used as a single agent, in lieu of an aminoglycoside, for treatment of pyelonephritis caused by an unusually resistant organism. It also could be used in combination with clindamycin or metronidazole plus ampicillin for treatment of polymicrobial infections, such as chorioamnionitis, puerperal endometritis, and pelvic cellulitis.1,2
Continue to: Clindamycin...
Clindamycin
A macrolide antibiotic, clindamycin exerts its antibacterial effect by interfering with bacterial protein synthesis. It can be administered orally and intravenously. Its key spectrum of activity in our specialty includes GBS, staphylococci, and anaerobes. However, clindamycin is not active against enterococci or aerobic gram-negative bacilli. GI upset and antibiotic-induced diarrhea are its principal adverse effects, and clindamycin is one of the most important causes of C difficile colitis. Although it is available in a generic formulation, this drug is still relatively expensive.
Clindamycin’s principal application in our specialty is for treating staphylococcal infections, such as wound infections and mastitis. It is particularly effective against MRSA infections. When used in combination with an aminoglycoside such as gentamicin, clindamycin provides excellent treatment for chorioamnionitis, puerperal endometritis, and pelvic inflammatory disease. In fact, for many years, the combination of clindamycin plus gentamicin has been considered the gold standard for the treatment of polymicrobial, mixed aerobic-anaerobic pelvic infections.1,2
Doxycycline
Doxycycline, a tetracycline, exerts its antibacterial effect by inhibiting bacterial protein synthesis. The drug targets a broad range of pelvic pathogens, including C trachomatis and N gonorrhoeae, and it may be administered both orally and intravenously. Doxycycline’s principal adverse effects include headache, GI upset, and photosensitivity. By disrupting the normal bowel and vaginal flora, the drug also can cause diarrhea and vulvovaginal moniliasis. In addition, it can cause permanent discoloration of the teeth, and, for this reason, doxycycline should not be used in pregnant or lactating women or in young children.
Although doxycycline has been available in generic formulation for many years, it remains relatively expensive. As a single agent, its principal application in our specialty is for treatment of chlamydia infection. It may be used as prophylaxis for surgical procedures, such as hysterectomy and pregnancy terminations. In combination with an extended-spectrum cephalosporin, it also may be used to treat pelvic inflammatory disease.2,8,10
Metronidazole
Metronidazole, a nitroimidazole derivative, exerts its antibacterial effect by disrupting bacterial protein synthesis. The drug may be administered topically, orally, and intravenously. Its primary spectrum of activity is against anerobic microorganisms. It is also active against Giardia and Trichomonas vaginalis.
Metronidazole’s most common adverse effects are GI upset, a metallic taste in the mouth, and a disulfiram-like effect when taken with alcohol. The cost of oral and intravenous metronidazole is relatively low; ironically, the cost of topical metronidazole is relatively high. In our specialty, the principal applications of oral metronidazole are as a single agent for treatment of bacterial vaginosis and trichomoniasis. When combined with ampicillin plus an aminoglycoside, intravenous metronidazole provides excellent coverage against the diverse anaerobic microorganisms that cause chorioamnionitis, puerperal endometritis, and pelvic cellulitis.1,2
Trimethoprim-sulfamethoxazole (TMP-SMX)
This antibiotic combination (an antifolate and a sulfonamide) inhibits sequential steps in the synthesis of folic acid, an essential nutrient in bacterial metabolism. It is available in both an intravenous and oral formulation. TMP-SMX has a broad spectrum of activity against the aerobic gram-negative bacilli that cause UTIs in women. In addition, it provides excellent coverage against staphylococci, including MRSA; Pneumocystis jirovecii; and Toxoplasma gondii.
The medication’s principal toxicity is an allergic reaction. Some reactions are quite severe, such as the Stevens-Johnson syndrome. TMP-SMX is relatively inexpensive, particularly the oral formulation. The most common indications for TMP-SMX in our specialty are for treatment of UTIs, mastitis, and wound infections.1,2,11 In HIV-infected patients, the drug provides excellent prophylaxis against recurrent Pneumocystis and Toxoplasma infections. TMP-SMX should not be used in the first trimester of pregnancy because it has been linked to several birth defects, including neural tube defects, heart defects, choanal atresia, and diaphragmatic hernia.12
Nitrofurantoin
Usually administered orally as nitrofurantoin monohydrate macrocrystals, nitrofurantoin exerts its antibacterial effect primarily by inhibiting protein synthesis. Its principal spectrum of activity is against the aerobic gram-negative bacilli, with the exception of Proteus species. Nitrofurantoin’s most common adverse effects are GI upset, headache, vertigo, drowsiness, and allergic reactions. The drug is relatively inexpensive.
Nitrofurantoin is an excellent agent for the treatment of lower UTIs.11 It is not well concentrated in the renal parenchyma or blood, however, so it should not be used to treat pyelonephritis. As a general rule, nitrofurantoin should not be used in the first trimester of pregnancy because it has been associated with eye, heart, and facial cleft defects in the fetus.12
Vancomycin
Vancomycin exerts its antibacterial effect by inhibiting cell wall synthesis. It may be administered both orally and intravenously, and it specifically targets aerobic gram-positive cocci, particularly methicillin-sensitive and methicillin-resistant staphylococci. Vancomycin’s most important adverse effects include GI upset, nephrotoxicity, ototoxicity, and severe allergic reactions, such as anaphylaxis, Stevens-Johnson syndrome, and exfoliative dermatitis (the “red man” syndrome). The drug is moderately expensive.13
In its oral formulation, vancomycin’s principal application in our discipline is for treating C difficile colitis. In its intravenous formulation, it is used primarily as a single agent for GBS prophylaxis in penicillin-allergic patients, and it is used in combination with other antibiotics, such as clindamycin plus gentamicin, for treating patients with deep-seated incisional (wound) infections.1,2,13,14 ●
- Duff P. Maternal and perinatal infection in pregnancy: bacterial. In: Landon MB, Galan HL, Jauniaux ERM, et al, eds. Gabbe’s Obstetrics: Normal and Problem Pregnancies, 8th ed. Elsevier; 2020: chapter 58.
- Duff P. Antibiotic selection in obstetrics: making cost-effective choices. Clin Obstet Gynecol. 2002;45:59-72.
- Wagenlehner FME, Cloutier DJ, Komirenko AS, et al; EPIC Study Group. Once-daily plazomicin for complicated urinary tract infections. N Engl J Med. 2019;380:729-740.
- Leffler DA, Lamont JT. Clostridium difficile infection. N Engl J Med. 2015;372:1539-1548.
- Duff P. Prevention of infection after cesarean delivery. Clin Obstet Gynecol. 2019;62:758-770.
- Hooper DC, Wolfson JS. Fluoroquinolone antimicrobial agents. N Engl J Med. 1991;324:384-394.
- Castells M, Khan DA, Phillips EJ. Penicillin allergy. N Engl J Med. 2019 381:2338-2351.
- St Cyr S, Barbee L, Workowski KA, et al. Update to CDC’s treatment guidelines for gonococcal infection, 2020. MMWR Morbid Mortal Wkly Rep. 2020;69:1911-1916.
- Ray WA, Murray KT, Hall K, et al. Azithromycin and the risk of cardiovascular death. N Engl J Med. 2012;366:1881-1890.
- Workowski KA, Bolan GA. Sexually transmitted disease treatment guidelines, 2015. MMWR Morbid Mortal Wkly Rep. 2015;64(RR3):1-137.
- Duff P. UTIs in pregnancy: managing urethritis, asymptomatic bacteriuria, cystitis, and pyelonephritis. OBG Manag. 2022;34(1):42-46.
- Crider KS, Cleves MA, Reefhuis J, et al. Antibacterial medication use during pregnancy and risk of birth defects prevalence study. Arch Pediatr Adolesc Med. 2009;163:978985.
- Alvarez-Arango S, Ogunwole SM, Sequist TD, et al. Vancomycin infusion reaction—moving beyond “red man syndrome.” N Engl J Med. 2021;384:1283-1286.
- Finley TA, Duff P. Antibiotics for treatment of staphylococcal infections in the obstetric patient. Clin Obstet Gynecol. 2019;62:790-803.
In this article, I provide a simplified, practical review of the principal antibiotics that we use on a daily basis to treat bacterial infections. The antibiotics are listed in alphabetical order, either individually or by group. I focus first on the mechanism of action and spectrum of activity of the drugs used against the usual pelvic pathogens (TABLE).1 I then review their principal adverse effects, relative cost (categorized as low, intermediate, and high), and the key indications for these drugs in obstetrics and gynecology. In a forthcoming 2-part companion article, I will review how to select specific antibiotics and their dosing regimens for the most commonly encountered bacterial infections in our clinical practice.

Aminoglycoside antibiotics
The aminoglycosides include amikacin, gentamicin, plazomicin, and tobramycin.2,3 The 2 agents most commonly used in our specialty are amikacin and gentamicin. The drugs may be administered intramuscularly or intravenously, and they specifically target aerobic gram-negative bacilli. They also provide coverage against staphylococci and gonococci. Ototoxicity and nephrotoxicity are their principal adverse effects.
Aminoglycosides are used primarily as single agents to treat pyelonephritis caused by highly resistant bacteria and in combination with agents such as clindamycin and metronidazole to treat polymicrobial infections, including chorioamnionitis, puerperal endometritis, and pelvic inflammatory disease. Of all the aminoglycosides, gentamicin is clearly the least expensive.
Carbapenems
The original carbapenem widely introduced into clinical practice was imipenem-cilastatin. Imipenem, the active antibiotic, inhibits bacterial cell wall synthesis. Cilastatin inhibits renal dehydropeptidase I and, thereby, slows the metabolism of imipenem by the kidney. Other carbapenems include meropenem and ertapenem.
The carbapenems have the widest spectrum of activity against the pelvic pathogens of any antibiotic. They provide excellent coverage of aerobic and anaerobic gram-positive cocci and aerobic and anaerobic gram-negative bacilli. They do not cover methicillin-resistant Staphylococcus aureus (MRSA) and the enterococci very well.
A major adverse effect of the carbapenems is an allergic reaction, including anaphylaxis and Stevens-Johnson syndrome, and there is some minimal cross-sensitivity with the β-lactam antibiotics. Other important, but fortunately rare, adverse effects include neurotoxicity, hepatotoxicity, and Clostridium difficile colitis.4
As a group, the carbapenems are relatively more expensive than most other agents. Their principal application in our specialty is for single-agent treatment of serious polymicrobial infections, such as puerperal endometritis, pelvic cellulitis, and pelvic abscess, especially in patients who have a contraindication to the use of combination antibiotic regimens that include an aminoglycoside.1,2
Cephalosporins
The cephalosporins are β-lactam antibiotics that act by disrupting the synthesis of the bacterial cell wall. They may be administered orally, intramuscularly, and intravenously. The most common adverse effects associated with these agents are an allergic reaction, which can range from a mild rash to anaphylaxis and the Stevens-Johnson syndrome; central nervous system toxicity; and antibiotic-induced diarrhea, including C difficile colitis.1,2,4
This group of antibiotics can be confusing because it includes so many agents, and their spectrum of activity varies. I find it helpful to think about the coverage of these agents as limited spectrum versus intermediate spectrum versus extended spectrum.
The limited-spectrum cephalosporin prototypes are cephalexin (oral administration) and cefazolin (parenteral administration). This group of cephalosporins provides excellent coverage of aerobic and anaerobic gram-positive cocci. They are excellent against staphylococci, except for MRSA. Coverage is moderate for aerobic gram-negative bacilli but only limited for anaerobic gram-negative bacilli. They do not cover the enterococci. In our specialty, their principal application is for treatment of mastitis, urinary tract infections (UTIs), and wound infections and for prophylaxis against group B streptococcus (GBS) infection and post-cesarean infection.2,5 The cost of these drugs is relatively low.
The prototypes of the intermediate-spectrum cephalosporins are cefixime (oral) and ceftriaxone (parenteral). Both drugs have strong activity against aerobic and anaerobic streptococci, Neisseria gonorrhoeae, most aerobic gram-negative bacilli, and Treponema pallidum (principally, ceftriaxone). They are not consistently effective against staphylococci, particularly MRSA, and enterococci. Their key indications in obstetrics and gynecology are treatment of gonorrhea, syphilis (in penicillin-allergic patients), and acute pyelonephritis. Compared with the limited-spectrum cephalosporins, these antibiotics are moderately expensive.1,2
The 3 extended-spectrum cephalosporins used most commonly in our specialty are cefepime, cefotetan, and cefoxitin. These agents are administered intramuscularly and intravenously, and they provide very good coverage against aerobic and anaerobic gram-positive cocci, with the exception of staphylococci and enterococci. They have very good coverage against most gram-negative aerobic bacilli and excellent coverage against anerobic microorganisms. Their primary application in our specialty is for single-agent treatment of polymicrobial infections, such as puerperal endometritis and pelvic cellulitis. When used in combination with doxycycline, they are valuable in treating pelvic inflammatory disease. These drugs are more expensive than the limited-spectrum or intermediate-spectrum agents. They should not be used routinely as prophylaxis for pelvic surgery.1,2,5
Continue to: Fluorinated quinolones...
Fluorinated quinolones
The fluorinated quinolones include several agents, but the 3 most commonly used in our specialty are ciprofloxacin, ofloxacin, and levofloxacin. All 3 drugs can be administered orally; ciprofloxacin and levofloxacin also are available in intravenous formulations. These drugs interfere with bacterial protein synthesis by targeting DNA gyrase, an enzyme that introduces negative supertwists into DNA and separates interlocked DNA molecules.
These drugs provide excellent coverage against gram-negative bacilli, including Haemophilus influenzae; gram-negative cocci, such as N gonorrhoeae, Neisseria meningitidis, and Moraxella catarrhalis; and many staphylococci species. Levofloxacin, but not the other 2 drugs, provides moderate coverage against anaerobes. Ofloxacin and levofloxacin are active against chlamydia. Levofloxacin also covers the mycoplasma organisms that are responsible for atypical pneumonia.
As a group, the fluorinated quinolones are moderately expensive. The most likely adverse effects with these agents are gastrointestinal (GI) upset, headache, agitation, and sleep disturbance. Allergic reactions are rare. These drugs are of primary value in our specialty in treating gonorrhea, chlamydia, complicated UTIs, and respiratory tract infections.1,2,6
The penicillins
Penicillin
Penicillin, a β-lactam antibiotic, was one of the first antibiotics developed and employed in clinical practice. It may be administered orally, intramuscularly, and intravenously. Penicillin exerts its effect by interfering with bacterial cell wall synthesis. Its principal spectrum of activity is against aerobic streptococci, such as group A and B streptococcus; most anaerobic gram-positive cocci that are present in the vaginal flora; some anaerobic gram-negative bacilli; and T pallidum. Penicillin is not effective against the majority of staphylococci species, enterococci, or aerobic gram-negative bacilli, such as Escherichia coli.
Penicillin’s major adverse effect is an allergic reaction, experienced by less than 10% of recipients.7 Most reactions are mild and are characterized by a morbilliform skin rash. However, some reactions are severe and take the form of an urticarial skin eruption, laryngospasm, bronchospasm, and overt anaphylaxis. The cost of both oral and parenteral penicillin formulations is very low. In obstetrics and gynecology, penicillin is used primarily for the treatment of group A and B streptococci infections, clostridial infections, and syphilis.1,2
Ampicillin and amoxicillin
The β-lactam antibiotics ampicillin and amoxicillin also act by interfering with bacterial cell wall synthesis. Amoxicillin is administered orally; ampicillin may be administered orally, intramuscularly, and intravenously. Their spectrum of activity includes group A and B streptococci, enterococci, most anaerobic gram-positive cocci, some anaerobic gram-negative bacilli, many aerobic gram-negative bacilli, and clostridial organisms.
Like penicillin, ampicillin and amoxicillin may cause allergic reactions that range from mild rashes to anaphylaxis. Unlike the more narrow-spectrum penicillin, they may cause antibiotic-associated diarrhea, including C difficile colitis,4 and they may eliminate part of the normal vaginal flora and stimulate an overgrowth of yeast organisms in the vagina. The cost of ampicillin and amoxicillin is very low. These 2 agents are used primarily for treatment of group A and B streptococci infections and some UTIs, particularly those caused by enterococci.1,2
Dicloxacillin sodium
This penicillin derivative disrupts bacterial cell wall synthesis and targets primarily aerobic gram-positive cocci, particularly staphylococci species. The antibiotic is not active against MRSA. The principal adverse effects of dicloxacillin sodium are an allergic reaction and GI upset. The drug is very inexpensive.
The key application for dicloxacillin sodium in our specialty is for treatment of puerperal mastitis.1
Continue to: Extended-spectrum penicillins...
Extended-spectrum penicillins
Three interesting combination extended-spectrum penicillins are used widely in our specialty. They are ampicillin/sulbactam, amoxicillin/clavulanate, and piperacillin/tazobactam. Ampicillin/sulbactam may be administered intramuscularly and intravenously. Piperacillin/tazobactam is administered intravenously; amoxicillin/clavulanate is administered orally.
Clavulanate, sulbactam, and tazobactam are β-lactamase inhibitors. When added to the parent antibiotic (amoxicillin, ampicillin, and piperacillin, respectively), they significantly enhance the parent drug’s spectrum of activity. These agents interfere with bacterial cell wall synthesis. They provide excellent coverage of aerobic gram-positive cocci, including enterococci; anaerobic gram-positive cocci; anaerobic gram-negative bacilli; and aerobic gram-negative bacilli. Their principal adverse effects include allergic reactions and antibiotic-associated diarrhea. They are moderately expensive.
The principal application of ampicillin/sulbactam and piperacillin/tazobactam in our specialty is as single agents for treatment of puerperal endometritis, postoperative pelvic cellulitis, and pyelonephritis. The usual role for amoxicillin/clavulanate is for oral treatment of complicated UTIs, including pyelonephritis in early pregnancy, and for outpatient therapy of mild to moderately severe endometritis following delivery or pregnancy termination.
Macrolides, monobactams, and additional antibiotics
Azithromycin
Azithromycin is a macrolide antibiotic that is in the same class as erythromycin and clindamycin. In our specialty, it has largely replaced erythromycin because of its more convenient dosage schedule and its better tolerability. It inhibits bacterial protein synthesis, and it is available in both an oral and intravenous formulation.
Azithromycin has an excellent spectrum of activity against the 3 major microorganisms that cause otitis media, sinusitis, and bronchitis: Streptococcus pneumoniae, H influenzae, and M catarrhalis. It also provides excellent coverage of Chlamydia trachomatis, Mycoplasma pneumoniae, and genital mycoplasmas; in high doses it provides modest coverage against gonorrhea.8 Unlike erythromycin, it has minimal GI toxicity and is usually very well tolerated by most patients. One unusual, but very important, adverse effect of the drug is prolongation of the Q-T interval.9
Azithromycin is now available in generic form and is relatively inexpensive. As a single agent, its principal applications in our specialty are for treatment of respiratory tract infections such as otitis media, sinusitis, and acute bronchitis and for treatment of chlamydia urethritis and endocervicitis.8,10 In combination with ampicillin, azithromycin is used as prophylaxis in patients with preterm premature rupture of membranes (PPROM), and, in combination with cefazolin, it is used for prophylaxis in patients undergoing cesarean delivery.1,2,5
Aztreonam
Aztreonam is a monobactam antibiotic. Like the cephalosporins and penicillins, aztreonam inhibits bacterial cell wall synthesis. It may be administered intramuscularly and intravenously, and its principal spectrum of activity is against aerobic gram-negative bacilli, which is similar to the aminoglycosides’ spectrum.
Aztreonam’s most likely adverse effects include phlebitis at the injection site, allergy, GI upset, and diarrhea. The drug is moderately expensive. In our specialty, aztreonam could be used as a single agent, in lieu of an aminoglycoside, for treatment of pyelonephritis caused by an unusually resistant organism. It also could be used in combination with clindamycin or metronidazole plus ampicillin for treatment of polymicrobial infections, such as chorioamnionitis, puerperal endometritis, and pelvic cellulitis.1,2
Continue to: Clindamycin...
Clindamycin
A macrolide antibiotic, clindamycin exerts its antibacterial effect by interfering with bacterial protein synthesis. It can be administered orally and intravenously. Its key spectrum of activity in our specialty includes GBS, staphylococci, and anaerobes. However, clindamycin is not active against enterococci or aerobic gram-negative bacilli. GI upset and antibiotic-induced diarrhea are its principal adverse effects, and clindamycin is one of the most important causes of C difficile colitis. Although it is available in a generic formulation, this drug is still relatively expensive.
Clindamycin’s principal application in our specialty is for treating staphylococcal infections, such as wound infections and mastitis. It is particularly effective against MRSA infections. When used in combination with an aminoglycoside such as gentamicin, clindamycin provides excellent treatment for chorioamnionitis, puerperal endometritis, and pelvic inflammatory disease. In fact, for many years, the combination of clindamycin plus gentamicin has been considered the gold standard for the treatment of polymicrobial, mixed aerobic-anaerobic pelvic infections.1,2
Doxycycline
Doxycycline, a tetracycline, exerts its antibacterial effect by inhibiting bacterial protein synthesis. The drug targets a broad range of pelvic pathogens, including C trachomatis and N gonorrhoeae, and it may be administered both orally and intravenously. Doxycycline’s principal adverse effects include headache, GI upset, and photosensitivity. By disrupting the normal bowel and vaginal flora, the drug also can cause diarrhea and vulvovaginal moniliasis. In addition, it can cause permanent discoloration of the teeth, and, for this reason, doxycycline should not be used in pregnant or lactating women or in young children.
Although doxycycline has been available in generic formulation for many years, it remains relatively expensive. As a single agent, its principal application in our specialty is for treatment of chlamydia infection. It may be used as prophylaxis for surgical procedures, such as hysterectomy and pregnancy terminations. In combination with an extended-spectrum cephalosporin, it also may be used to treat pelvic inflammatory disease.2,8,10
Metronidazole
Metronidazole, a nitroimidazole derivative, exerts its antibacterial effect by disrupting bacterial protein synthesis. The drug may be administered topically, orally, and intravenously. Its primary spectrum of activity is against anerobic microorganisms. It is also active against Giardia and Trichomonas vaginalis.
Metronidazole’s most common adverse effects are GI upset, a metallic taste in the mouth, and a disulfiram-like effect when taken with alcohol. The cost of oral and intravenous metronidazole is relatively low; ironically, the cost of topical metronidazole is relatively high. In our specialty, the principal applications of oral metronidazole are as a single agent for treatment of bacterial vaginosis and trichomoniasis. When combined with ampicillin plus an aminoglycoside, intravenous metronidazole provides excellent coverage against the diverse anaerobic microorganisms that cause chorioamnionitis, puerperal endometritis, and pelvic cellulitis.1,2
Trimethoprim-sulfamethoxazole (TMP-SMX)
This antibiotic combination (an antifolate and a sulfonamide) inhibits sequential steps in the synthesis of folic acid, an essential nutrient in bacterial metabolism. It is available in both an intravenous and oral formulation. TMP-SMX has a broad spectrum of activity against the aerobic gram-negative bacilli that cause UTIs in women. In addition, it provides excellent coverage against staphylococci, including MRSA; Pneumocystis jirovecii; and Toxoplasma gondii.
The medication’s principal toxicity is an allergic reaction. Some reactions are quite severe, such as the Stevens-Johnson syndrome. TMP-SMX is relatively inexpensive, particularly the oral formulation. The most common indications for TMP-SMX in our specialty are for treatment of UTIs, mastitis, and wound infections.1,2,11 In HIV-infected patients, the drug provides excellent prophylaxis against recurrent Pneumocystis and Toxoplasma infections. TMP-SMX should not be used in the first trimester of pregnancy because it has been linked to several birth defects, including neural tube defects, heart defects, choanal atresia, and diaphragmatic hernia.12
Nitrofurantoin
Usually administered orally as nitrofurantoin monohydrate macrocrystals, nitrofurantoin exerts its antibacterial effect primarily by inhibiting protein synthesis. Its principal spectrum of activity is against the aerobic gram-negative bacilli, with the exception of Proteus species. Nitrofurantoin’s most common adverse effects are GI upset, headache, vertigo, drowsiness, and allergic reactions. The drug is relatively inexpensive.
Nitrofurantoin is an excellent agent for the treatment of lower UTIs.11 It is not well concentrated in the renal parenchyma or blood, however, so it should not be used to treat pyelonephritis. As a general rule, nitrofurantoin should not be used in the first trimester of pregnancy because it has been associated with eye, heart, and facial cleft defects in the fetus.12
Vancomycin
Vancomycin exerts its antibacterial effect by inhibiting cell wall synthesis. It may be administered both orally and intravenously, and it specifically targets aerobic gram-positive cocci, particularly methicillin-sensitive and methicillin-resistant staphylococci. Vancomycin’s most important adverse effects include GI upset, nephrotoxicity, ototoxicity, and severe allergic reactions, such as anaphylaxis, Stevens-Johnson syndrome, and exfoliative dermatitis (the “red man” syndrome). The drug is moderately expensive.13
In its oral formulation, vancomycin’s principal application in our discipline is for treating C difficile colitis. In its intravenous formulation, it is used primarily as a single agent for GBS prophylaxis in penicillin-allergic patients, and it is used in combination with other antibiotics, such as clindamycin plus gentamicin, for treating patients with deep-seated incisional (wound) infections.1,2,13,14 ●
In this article, I provide a simplified, practical review of the principal antibiotics that we use on a daily basis to treat bacterial infections. The antibiotics are listed in alphabetical order, either individually or by group. I focus first on the mechanism of action and spectrum of activity of the drugs used against the usual pelvic pathogens (TABLE).1 I then review their principal adverse effects, relative cost (categorized as low, intermediate, and high), and the key indications for these drugs in obstetrics and gynecology. In a forthcoming 2-part companion article, I will review how to select specific antibiotics and their dosing regimens for the most commonly encountered bacterial infections in our clinical practice.

Aminoglycoside antibiotics
The aminoglycosides include amikacin, gentamicin, plazomicin, and tobramycin.2,3 The 2 agents most commonly used in our specialty are amikacin and gentamicin. The drugs may be administered intramuscularly or intravenously, and they specifically target aerobic gram-negative bacilli. They also provide coverage against staphylococci and gonococci. Ototoxicity and nephrotoxicity are their principal adverse effects.
Aminoglycosides are used primarily as single agents to treat pyelonephritis caused by highly resistant bacteria and in combination with agents such as clindamycin and metronidazole to treat polymicrobial infections, including chorioamnionitis, puerperal endometritis, and pelvic inflammatory disease. Of all the aminoglycosides, gentamicin is clearly the least expensive.
Carbapenems
The original carbapenem widely introduced into clinical practice was imipenem-cilastatin. Imipenem, the active antibiotic, inhibits bacterial cell wall synthesis. Cilastatin inhibits renal dehydropeptidase I and, thereby, slows the metabolism of imipenem by the kidney. Other carbapenems include meropenem and ertapenem.
The carbapenems have the widest spectrum of activity against the pelvic pathogens of any antibiotic. They provide excellent coverage of aerobic and anaerobic gram-positive cocci and aerobic and anaerobic gram-negative bacilli. They do not cover methicillin-resistant Staphylococcus aureus (MRSA) and the enterococci very well.
A major adverse effect of the carbapenems is an allergic reaction, including anaphylaxis and Stevens-Johnson syndrome, and there is some minimal cross-sensitivity with the β-lactam antibiotics. Other important, but fortunately rare, adverse effects include neurotoxicity, hepatotoxicity, and Clostridium difficile colitis.4
As a group, the carbapenems are relatively more expensive than most other agents. Their principal application in our specialty is for single-agent treatment of serious polymicrobial infections, such as puerperal endometritis, pelvic cellulitis, and pelvic abscess, especially in patients who have a contraindication to the use of combination antibiotic regimens that include an aminoglycoside.1,2
Cephalosporins
The cephalosporins are β-lactam antibiotics that act by disrupting the synthesis of the bacterial cell wall. They may be administered orally, intramuscularly, and intravenously. The most common adverse effects associated with these agents are an allergic reaction, which can range from a mild rash to anaphylaxis and the Stevens-Johnson syndrome; central nervous system toxicity; and antibiotic-induced diarrhea, including C difficile colitis.1,2,4
This group of antibiotics can be confusing because it includes so many agents, and their spectrum of activity varies. I find it helpful to think about the coverage of these agents as limited spectrum versus intermediate spectrum versus extended spectrum.
The limited-spectrum cephalosporin prototypes are cephalexin (oral administration) and cefazolin (parenteral administration). This group of cephalosporins provides excellent coverage of aerobic and anaerobic gram-positive cocci. They are excellent against staphylococci, except for MRSA. Coverage is moderate for aerobic gram-negative bacilli but only limited for anaerobic gram-negative bacilli. They do not cover the enterococci. In our specialty, their principal application is for treatment of mastitis, urinary tract infections (UTIs), and wound infections and for prophylaxis against group B streptococcus (GBS) infection and post-cesarean infection.2,5 The cost of these drugs is relatively low.
The prototypes of the intermediate-spectrum cephalosporins are cefixime (oral) and ceftriaxone (parenteral). Both drugs have strong activity against aerobic and anaerobic streptococci, Neisseria gonorrhoeae, most aerobic gram-negative bacilli, and Treponema pallidum (principally, ceftriaxone). They are not consistently effective against staphylococci, particularly MRSA, and enterococci. Their key indications in obstetrics and gynecology are treatment of gonorrhea, syphilis (in penicillin-allergic patients), and acute pyelonephritis. Compared with the limited-spectrum cephalosporins, these antibiotics are moderately expensive.1,2
The 3 extended-spectrum cephalosporins used most commonly in our specialty are cefepime, cefotetan, and cefoxitin. These agents are administered intramuscularly and intravenously, and they provide very good coverage against aerobic and anaerobic gram-positive cocci, with the exception of staphylococci and enterococci. They have very good coverage against most gram-negative aerobic bacilli and excellent coverage against anerobic microorganisms. Their primary application in our specialty is for single-agent treatment of polymicrobial infections, such as puerperal endometritis and pelvic cellulitis. When used in combination with doxycycline, they are valuable in treating pelvic inflammatory disease. These drugs are more expensive than the limited-spectrum or intermediate-spectrum agents. They should not be used routinely as prophylaxis for pelvic surgery.1,2,5
Continue to: Fluorinated quinolones...
Fluorinated quinolones
The fluorinated quinolones include several agents, but the 3 most commonly used in our specialty are ciprofloxacin, ofloxacin, and levofloxacin. All 3 drugs can be administered orally; ciprofloxacin and levofloxacin also are available in intravenous formulations. These drugs interfere with bacterial protein synthesis by targeting DNA gyrase, an enzyme that introduces negative supertwists into DNA and separates interlocked DNA molecules.
These drugs provide excellent coverage against gram-negative bacilli, including Haemophilus influenzae; gram-negative cocci, such as N gonorrhoeae, Neisseria meningitidis, and Moraxella catarrhalis; and many staphylococci species. Levofloxacin, but not the other 2 drugs, provides moderate coverage against anaerobes. Ofloxacin and levofloxacin are active against chlamydia. Levofloxacin also covers the mycoplasma organisms that are responsible for atypical pneumonia.
As a group, the fluorinated quinolones are moderately expensive. The most likely adverse effects with these agents are gastrointestinal (GI) upset, headache, agitation, and sleep disturbance. Allergic reactions are rare. These drugs are of primary value in our specialty in treating gonorrhea, chlamydia, complicated UTIs, and respiratory tract infections.1,2,6
The penicillins
Penicillin
Penicillin, a β-lactam antibiotic, was one of the first antibiotics developed and employed in clinical practice. It may be administered orally, intramuscularly, and intravenously. Penicillin exerts its effect by interfering with bacterial cell wall synthesis. Its principal spectrum of activity is against aerobic streptococci, such as group A and B streptococcus; most anaerobic gram-positive cocci that are present in the vaginal flora; some anaerobic gram-negative bacilli; and T pallidum. Penicillin is not effective against the majority of staphylococci species, enterococci, or aerobic gram-negative bacilli, such as Escherichia coli.
Penicillin’s major adverse effect is an allergic reaction, experienced by less than 10% of recipients.7 Most reactions are mild and are characterized by a morbilliform skin rash. However, some reactions are severe and take the form of an urticarial skin eruption, laryngospasm, bronchospasm, and overt anaphylaxis. The cost of both oral and parenteral penicillin formulations is very low. In obstetrics and gynecology, penicillin is used primarily for the treatment of group A and B streptococci infections, clostridial infections, and syphilis.1,2
Ampicillin and amoxicillin
The β-lactam antibiotics ampicillin and amoxicillin also act by interfering with bacterial cell wall synthesis. Amoxicillin is administered orally; ampicillin may be administered orally, intramuscularly, and intravenously. Their spectrum of activity includes group A and B streptococci, enterococci, most anaerobic gram-positive cocci, some anaerobic gram-negative bacilli, many aerobic gram-negative bacilli, and clostridial organisms.
Like penicillin, ampicillin and amoxicillin may cause allergic reactions that range from mild rashes to anaphylaxis. Unlike the more narrow-spectrum penicillin, they may cause antibiotic-associated diarrhea, including C difficile colitis,4 and they may eliminate part of the normal vaginal flora and stimulate an overgrowth of yeast organisms in the vagina. The cost of ampicillin and amoxicillin is very low. These 2 agents are used primarily for treatment of group A and B streptococci infections and some UTIs, particularly those caused by enterococci.1,2
Dicloxacillin sodium
This penicillin derivative disrupts bacterial cell wall synthesis and targets primarily aerobic gram-positive cocci, particularly staphylococci species. The antibiotic is not active against MRSA. The principal adverse effects of dicloxacillin sodium are an allergic reaction and GI upset. The drug is very inexpensive.
The key application for dicloxacillin sodium in our specialty is for treatment of puerperal mastitis.1
Continue to: Extended-spectrum penicillins...
Extended-spectrum penicillins
Three interesting combination extended-spectrum penicillins are used widely in our specialty. They are ampicillin/sulbactam, amoxicillin/clavulanate, and piperacillin/tazobactam. Ampicillin/sulbactam may be administered intramuscularly and intravenously. Piperacillin/tazobactam is administered intravenously; amoxicillin/clavulanate is administered orally.
Clavulanate, sulbactam, and tazobactam are β-lactamase inhibitors. When added to the parent antibiotic (amoxicillin, ampicillin, and piperacillin, respectively), they significantly enhance the parent drug’s spectrum of activity. These agents interfere with bacterial cell wall synthesis. They provide excellent coverage of aerobic gram-positive cocci, including enterococci; anaerobic gram-positive cocci; anaerobic gram-negative bacilli; and aerobic gram-negative bacilli. Their principal adverse effects include allergic reactions and antibiotic-associated diarrhea. They are moderately expensive.
The principal application of ampicillin/sulbactam and piperacillin/tazobactam in our specialty is as single agents for treatment of puerperal endometritis, postoperative pelvic cellulitis, and pyelonephritis. The usual role for amoxicillin/clavulanate is for oral treatment of complicated UTIs, including pyelonephritis in early pregnancy, and for outpatient therapy of mild to moderately severe endometritis following delivery or pregnancy termination.
Macrolides, monobactams, and additional antibiotics
Azithromycin
Azithromycin is a macrolide antibiotic that is in the same class as erythromycin and clindamycin. In our specialty, it has largely replaced erythromycin because of its more convenient dosage schedule and its better tolerability. It inhibits bacterial protein synthesis, and it is available in both an oral and intravenous formulation.
Azithromycin has an excellent spectrum of activity against the 3 major microorganisms that cause otitis media, sinusitis, and bronchitis: Streptococcus pneumoniae, H influenzae, and M catarrhalis. It also provides excellent coverage of Chlamydia trachomatis, Mycoplasma pneumoniae, and genital mycoplasmas; in high doses it provides modest coverage against gonorrhea.8 Unlike erythromycin, it has minimal GI toxicity and is usually very well tolerated by most patients. One unusual, but very important, adverse effect of the drug is prolongation of the Q-T interval.9
Azithromycin is now available in generic form and is relatively inexpensive. As a single agent, its principal applications in our specialty are for treatment of respiratory tract infections such as otitis media, sinusitis, and acute bronchitis and for treatment of chlamydia urethritis and endocervicitis.8,10 In combination with ampicillin, azithromycin is used as prophylaxis in patients with preterm premature rupture of membranes (PPROM), and, in combination with cefazolin, it is used for prophylaxis in patients undergoing cesarean delivery.1,2,5
Aztreonam
Aztreonam is a monobactam antibiotic. Like the cephalosporins and penicillins, aztreonam inhibits bacterial cell wall synthesis. It may be administered intramuscularly and intravenously, and its principal spectrum of activity is against aerobic gram-negative bacilli, which is similar to the aminoglycosides’ spectrum.
Aztreonam’s most likely adverse effects include phlebitis at the injection site, allergy, GI upset, and diarrhea. The drug is moderately expensive. In our specialty, aztreonam could be used as a single agent, in lieu of an aminoglycoside, for treatment of pyelonephritis caused by an unusually resistant organism. It also could be used in combination with clindamycin or metronidazole plus ampicillin for treatment of polymicrobial infections, such as chorioamnionitis, puerperal endometritis, and pelvic cellulitis.1,2
Continue to: Clindamycin...
Clindamycin
A macrolide antibiotic, clindamycin exerts its antibacterial effect by interfering with bacterial protein synthesis. It can be administered orally and intravenously. Its key spectrum of activity in our specialty includes GBS, staphylococci, and anaerobes. However, clindamycin is not active against enterococci or aerobic gram-negative bacilli. GI upset and antibiotic-induced diarrhea are its principal adverse effects, and clindamycin is one of the most important causes of C difficile colitis. Although it is available in a generic formulation, this drug is still relatively expensive.
Clindamycin’s principal application in our specialty is for treating staphylococcal infections, such as wound infections and mastitis. It is particularly effective against MRSA infections. When used in combination with an aminoglycoside such as gentamicin, clindamycin provides excellent treatment for chorioamnionitis, puerperal endometritis, and pelvic inflammatory disease. In fact, for many years, the combination of clindamycin plus gentamicin has been considered the gold standard for the treatment of polymicrobial, mixed aerobic-anaerobic pelvic infections.1,2
Doxycycline
Doxycycline, a tetracycline, exerts its antibacterial effect by inhibiting bacterial protein synthesis. The drug targets a broad range of pelvic pathogens, including C trachomatis and N gonorrhoeae, and it may be administered both orally and intravenously. Doxycycline’s principal adverse effects include headache, GI upset, and photosensitivity. By disrupting the normal bowel and vaginal flora, the drug also can cause diarrhea and vulvovaginal moniliasis. In addition, it can cause permanent discoloration of the teeth, and, for this reason, doxycycline should not be used in pregnant or lactating women or in young children.
Although doxycycline has been available in generic formulation for many years, it remains relatively expensive. As a single agent, its principal application in our specialty is for treatment of chlamydia infection. It may be used as prophylaxis for surgical procedures, such as hysterectomy and pregnancy terminations. In combination with an extended-spectrum cephalosporin, it also may be used to treat pelvic inflammatory disease.2,8,10
Metronidazole
Metronidazole, a nitroimidazole derivative, exerts its antibacterial effect by disrupting bacterial protein synthesis. The drug may be administered topically, orally, and intravenously. Its primary spectrum of activity is against anerobic microorganisms. It is also active against Giardia and Trichomonas vaginalis.
Metronidazole’s most common adverse effects are GI upset, a metallic taste in the mouth, and a disulfiram-like effect when taken with alcohol. The cost of oral and intravenous metronidazole is relatively low; ironically, the cost of topical metronidazole is relatively high. In our specialty, the principal applications of oral metronidazole are as a single agent for treatment of bacterial vaginosis and trichomoniasis. When combined with ampicillin plus an aminoglycoside, intravenous metronidazole provides excellent coverage against the diverse anaerobic microorganisms that cause chorioamnionitis, puerperal endometritis, and pelvic cellulitis.1,2
Trimethoprim-sulfamethoxazole (TMP-SMX)
This antibiotic combination (an antifolate and a sulfonamide) inhibits sequential steps in the synthesis of folic acid, an essential nutrient in bacterial metabolism. It is available in both an intravenous and oral formulation. TMP-SMX has a broad spectrum of activity against the aerobic gram-negative bacilli that cause UTIs in women. In addition, it provides excellent coverage against staphylococci, including MRSA; Pneumocystis jirovecii; and Toxoplasma gondii.
The medication’s principal toxicity is an allergic reaction. Some reactions are quite severe, such as the Stevens-Johnson syndrome. TMP-SMX is relatively inexpensive, particularly the oral formulation. The most common indications for TMP-SMX in our specialty are for treatment of UTIs, mastitis, and wound infections.1,2,11 In HIV-infected patients, the drug provides excellent prophylaxis against recurrent Pneumocystis and Toxoplasma infections. TMP-SMX should not be used in the first trimester of pregnancy because it has been linked to several birth defects, including neural tube defects, heart defects, choanal atresia, and diaphragmatic hernia.12
Nitrofurantoin
Usually administered orally as nitrofurantoin monohydrate macrocrystals, nitrofurantoin exerts its antibacterial effect primarily by inhibiting protein synthesis. Its principal spectrum of activity is against the aerobic gram-negative bacilli, with the exception of Proteus species. Nitrofurantoin’s most common adverse effects are GI upset, headache, vertigo, drowsiness, and allergic reactions. The drug is relatively inexpensive.
Nitrofurantoin is an excellent agent for the treatment of lower UTIs.11 It is not well concentrated in the renal parenchyma or blood, however, so it should not be used to treat pyelonephritis. As a general rule, nitrofurantoin should not be used in the first trimester of pregnancy because it has been associated with eye, heart, and facial cleft defects in the fetus.12
Vancomycin
Vancomycin exerts its antibacterial effect by inhibiting cell wall synthesis. It may be administered both orally and intravenously, and it specifically targets aerobic gram-positive cocci, particularly methicillin-sensitive and methicillin-resistant staphylococci. Vancomycin’s most important adverse effects include GI upset, nephrotoxicity, ototoxicity, and severe allergic reactions, such as anaphylaxis, Stevens-Johnson syndrome, and exfoliative dermatitis (the “red man” syndrome). The drug is moderately expensive.13
In its oral formulation, vancomycin’s principal application in our discipline is for treating C difficile colitis. In its intravenous formulation, it is used primarily as a single agent for GBS prophylaxis in penicillin-allergic patients, and it is used in combination with other antibiotics, such as clindamycin plus gentamicin, for treating patients with deep-seated incisional (wound) infections.1,2,13,14 ●
- Duff P. Maternal and perinatal infection in pregnancy: bacterial. In: Landon MB, Galan HL, Jauniaux ERM, et al, eds. Gabbe’s Obstetrics: Normal and Problem Pregnancies, 8th ed. Elsevier; 2020: chapter 58.
- Duff P. Antibiotic selection in obstetrics: making cost-effective choices. Clin Obstet Gynecol. 2002;45:59-72.
- Wagenlehner FME, Cloutier DJ, Komirenko AS, et al; EPIC Study Group. Once-daily plazomicin for complicated urinary tract infections. N Engl J Med. 2019;380:729-740.
- Leffler DA, Lamont JT. Clostridium difficile infection. N Engl J Med. 2015;372:1539-1548.
- Duff P. Prevention of infection after cesarean delivery. Clin Obstet Gynecol. 2019;62:758-770.
- Hooper DC, Wolfson JS. Fluoroquinolone antimicrobial agents. N Engl J Med. 1991;324:384-394.
- Castells M, Khan DA, Phillips EJ. Penicillin allergy. N Engl J Med. 2019 381:2338-2351.
- St Cyr S, Barbee L, Workowski KA, et al. Update to CDC’s treatment guidelines for gonococcal infection, 2020. MMWR Morbid Mortal Wkly Rep. 2020;69:1911-1916.
- Ray WA, Murray KT, Hall K, et al. Azithromycin and the risk of cardiovascular death. N Engl J Med. 2012;366:1881-1890.
- Workowski KA, Bolan GA. Sexually transmitted disease treatment guidelines, 2015. MMWR Morbid Mortal Wkly Rep. 2015;64(RR3):1-137.
- Duff P. UTIs in pregnancy: managing urethritis, asymptomatic bacteriuria, cystitis, and pyelonephritis. OBG Manag. 2022;34(1):42-46.
- Crider KS, Cleves MA, Reefhuis J, et al. Antibacterial medication use during pregnancy and risk of birth defects prevalence study. Arch Pediatr Adolesc Med. 2009;163:978985.
- Alvarez-Arango S, Ogunwole SM, Sequist TD, et al. Vancomycin infusion reaction—moving beyond “red man syndrome.” N Engl J Med. 2021;384:1283-1286.
- Finley TA, Duff P. Antibiotics for treatment of staphylococcal infections in the obstetric patient. Clin Obstet Gynecol. 2019;62:790-803.
- Duff P. Maternal and perinatal infection in pregnancy: bacterial. In: Landon MB, Galan HL, Jauniaux ERM, et al, eds. Gabbe’s Obstetrics: Normal and Problem Pregnancies, 8th ed. Elsevier; 2020: chapter 58.
- Duff P. Antibiotic selection in obstetrics: making cost-effective choices. Clin Obstet Gynecol. 2002;45:59-72.
- Wagenlehner FME, Cloutier DJ, Komirenko AS, et al; EPIC Study Group. Once-daily plazomicin for complicated urinary tract infections. N Engl J Med. 2019;380:729-740.
- Leffler DA, Lamont JT. Clostridium difficile infection. N Engl J Med. 2015;372:1539-1548.
- Duff P. Prevention of infection after cesarean delivery. Clin Obstet Gynecol. 2019;62:758-770.
- Hooper DC, Wolfson JS. Fluoroquinolone antimicrobial agents. N Engl J Med. 1991;324:384-394.
- Castells M, Khan DA, Phillips EJ. Penicillin allergy. N Engl J Med. 2019 381:2338-2351.
- St Cyr S, Barbee L, Workowski KA, et al. Update to CDC’s treatment guidelines for gonococcal infection, 2020. MMWR Morbid Mortal Wkly Rep. 2020;69:1911-1916.
- Ray WA, Murray KT, Hall K, et al. Azithromycin and the risk of cardiovascular death. N Engl J Med. 2012;366:1881-1890.
- Workowski KA, Bolan GA. Sexually transmitted disease treatment guidelines, 2015. MMWR Morbid Mortal Wkly Rep. 2015;64(RR3):1-137.
- Duff P. UTIs in pregnancy: managing urethritis, asymptomatic bacteriuria, cystitis, and pyelonephritis. OBG Manag. 2022;34(1):42-46.
- Crider KS, Cleves MA, Reefhuis J, et al. Antibacterial medication use during pregnancy and risk of birth defects prevalence study. Arch Pediatr Adolesc Med. 2009;163:978985.
- Alvarez-Arango S, Ogunwole SM, Sequist TD, et al. Vancomycin infusion reaction—moving beyond “red man syndrome.” N Engl J Med. 2021;384:1283-1286.
- Finley TA, Duff P. Antibiotics for treatment of staphylococcal infections in the obstetric patient. Clin Obstet Gynecol. 2019;62:790-803.
Can US “pattern recognition” of classic adnexal lesions reduce surgery, and even referrals for other imaging, in average-risk women?
Gupta A, Jha P, Baran TM, et al. Ovarian cancer detection in average-risk women: classic- versus nonclassic-appearing adnexal lesions at US. Radiology. 2022;212338. doi: 10.1148/radiol.212338.
Expert commentary
Gupta and colleagues conducted a multicenter, retrospective review of 970 adnexal lesions among 878 women—75% were premenopausal and 25% were postmenopausal.
Imaging details
The lesions were characterized by pattern recognition as “classic” (simple cysts, endometriomas, hemorrhagic cysts, or dermoids) or “nonclassic.” Out of 673 classic lesions, there were 4 malignancies (0.6%), of which 1 was an endometrioma and 3 were classified as simple cysts. However, out of 297 nonclassic lesions (multilocular, unilocular with solid areas or wall irregularity, or mostly solid), 32% (33/103) were malignant when vascularity was present, while 8% (16/184) were malignant when no intralesional vascularity was appreciated.
The authors pointed out that, especially because their study was retrospective, there was no standardization of scan technique or equipment employed. However, this point adds credibility to the “real world” nature of such imaging.
Other data corroborate findings
Other studies have looked at pattern recognition in efforts to optimize a conservative approach to benign masses and referral to oncology for suspected malignant masses, as described above. This was the main cornerstone of the International Consensus Conference,2 which also identified next steps for indeterminate masses, including evidence-based risk assessment algorithms and referral (to an expert imager or gynecologic oncologist). A multicenter trial in Europe3 found that ultrasound experience substantially impacts on diagnostic performance when adnexal masses are classified using pattern recognition. This occurred in a stepwise fashion with increasing accuracy directly related to the level of expertise. Shetty and colleagues4 found that pattern recognition performed better than the risk of malignancy index (sensitivities of 95% and 79%, respectively). ●
While the concept of pattern recognition for some “classic” benign ovarian masses has been around for some time, this is the first time a large United States–based study (albeit retrospective) has corroborated that when ultrasonography reveals a classic, or “almost certainly benign” finding, patients can be reassured that the lesion is benign, thereby avoiding extensive further workup. When a lesion is “nonclassic” in appearance and without any blood flow, further imaging with follow-up magnetic resonance imaging or repeat ultrasound could be considered. In women with a nonclassic lesion with blood flow, particularly in older women, referral to a gynecologic oncologic surgeon will help ensure expeditious treatment of possible ovarian cancer.
- Boll D, Geomini PM, Brölmann HA. The pre-operative assessment of the adnexal mass: the accuracy of clinical estimates versus clinical prediction rules. BJOG. 2003;110:519-523.
- Glanc P, Benacerraf B, Bourne T, et al. First International Consensus Report on adnexal masses: management recommendations. J Ultrasound Med. 2017;36:849-863. doi: 10.1002/jum.14197.
- Van Holsbeke C, Daemen A, Yazbek J, et al. Ultrasound experience substantially impacts on diagnostic performance and confidence when adnexal masses are classified using pattern recognition. Gynecol Obstet Invest. 2010;69:160-168. doi: 10.1159/000265012.
- Shetty J, Reddy G, Pandey D. Role of sonographic grayscale pattern recognition in the diagnosis of adnexal masses. J Clin Diagn Res. 2017;11:QC12-QC15. doi: 10.7860 /JCDR/2017/28533.10614.
Gupta A, Jha P, Baran TM, et al. Ovarian cancer detection in average-risk women: classic- versus nonclassic-appearing adnexal lesions at US. Radiology. 2022;212338. doi: 10.1148/radiol.212338.
Expert commentary
Gupta and colleagues conducted a multicenter, retrospective review of 970 adnexal lesions among 878 women—75% were premenopausal and 25% were postmenopausal.
Imaging details
The lesions were characterized by pattern recognition as “classic” (simple cysts, endometriomas, hemorrhagic cysts, or dermoids) or “nonclassic.” Out of 673 classic lesions, there were 4 malignancies (0.6%), of which 1 was an endometrioma and 3 were classified as simple cysts. However, out of 297 nonclassic lesions (multilocular, unilocular with solid areas or wall irregularity, or mostly solid), 32% (33/103) were malignant when vascularity was present, while 8% (16/184) were malignant when no intralesional vascularity was appreciated.
The authors pointed out that, especially because their study was retrospective, there was no standardization of scan technique or equipment employed. However, this point adds credibility to the “real world” nature of such imaging.
Other data corroborate findings
Other studies have looked at pattern recognition in efforts to optimize a conservative approach to benign masses and referral to oncology for suspected malignant masses, as described above. This was the main cornerstone of the International Consensus Conference,2 which also identified next steps for indeterminate masses, including evidence-based risk assessment algorithms and referral (to an expert imager or gynecologic oncologist). A multicenter trial in Europe3 found that ultrasound experience substantially impacts on diagnostic performance when adnexal masses are classified using pattern recognition. This occurred in a stepwise fashion with increasing accuracy directly related to the level of expertise. Shetty and colleagues4 found that pattern recognition performed better than the risk of malignancy index (sensitivities of 95% and 79%, respectively). ●
While the concept of pattern recognition for some “classic” benign ovarian masses has been around for some time, this is the first time a large United States–based study (albeit retrospective) has corroborated that when ultrasonography reveals a classic, or “almost certainly benign” finding, patients can be reassured that the lesion is benign, thereby avoiding extensive further workup. When a lesion is “nonclassic” in appearance and without any blood flow, further imaging with follow-up magnetic resonance imaging or repeat ultrasound could be considered. In women with a nonclassic lesion with blood flow, particularly in older women, referral to a gynecologic oncologic surgeon will help ensure expeditious treatment of possible ovarian cancer.
Gupta A, Jha P, Baran TM, et al. Ovarian cancer detection in average-risk women: classic- versus nonclassic-appearing adnexal lesions at US. Radiology. 2022;212338. doi: 10.1148/radiol.212338.
Expert commentary
Gupta and colleagues conducted a multicenter, retrospective review of 970 adnexal lesions among 878 women—75% were premenopausal and 25% were postmenopausal.
Imaging details
The lesions were characterized by pattern recognition as “classic” (simple cysts, endometriomas, hemorrhagic cysts, or dermoids) or “nonclassic.” Out of 673 classic lesions, there were 4 malignancies (0.6%), of which 1 was an endometrioma and 3 were classified as simple cysts. However, out of 297 nonclassic lesions (multilocular, unilocular with solid areas or wall irregularity, or mostly solid), 32% (33/103) were malignant when vascularity was present, while 8% (16/184) were malignant when no intralesional vascularity was appreciated.
The authors pointed out that, especially because their study was retrospective, there was no standardization of scan technique or equipment employed. However, this point adds credibility to the “real world” nature of such imaging.
Other data corroborate findings
Other studies have looked at pattern recognition in efforts to optimize a conservative approach to benign masses and referral to oncology for suspected malignant masses, as described above. This was the main cornerstone of the International Consensus Conference,2 which also identified next steps for indeterminate masses, including evidence-based risk assessment algorithms and referral (to an expert imager or gynecologic oncologist). A multicenter trial in Europe3 found that ultrasound experience substantially impacts on diagnostic performance when adnexal masses are classified using pattern recognition. This occurred in a stepwise fashion with increasing accuracy directly related to the level of expertise. Shetty and colleagues4 found that pattern recognition performed better than the risk of malignancy index (sensitivities of 95% and 79%, respectively). ●
While the concept of pattern recognition for some “classic” benign ovarian masses has been around for some time, this is the first time a large United States–based study (albeit retrospective) has corroborated that when ultrasonography reveals a classic, or “almost certainly benign” finding, patients can be reassured that the lesion is benign, thereby avoiding extensive further workup. When a lesion is “nonclassic” in appearance and without any blood flow, further imaging with follow-up magnetic resonance imaging or repeat ultrasound could be considered. In women with a nonclassic lesion with blood flow, particularly in older women, referral to a gynecologic oncologic surgeon will help ensure expeditious treatment of possible ovarian cancer.
- Boll D, Geomini PM, Brölmann HA. The pre-operative assessment of the adnexal mass: the accuracy of clinical estimates versus clinical prediction rules. BJOG. 2003;110:519-523.
- Glanc P, Benacerraf B, Bourne T, et al. First International Consensus Report on adnexal masses: management recommendations. J Ultrasound Med. 2017;36:849-863. doi: 10.1002/jum.14197.
- Van Holsbeke C, Daemen A, Yazbek J, et al. Ultrasound experience substantially impacts on diagnostic performance and confidence when adnexal masses are classified using pattern recognition. Gynecol Obstet Invest. 2010;69:160-168. doi: 10.1159/000265012.
- Shetty J, Reddy G, Pandey D. Role of sonographic grayscale pattern recognition in the diagnosis of adnexal masses. J Clin Diagn Res. 2017;11:QC12-QC15. doi: 10.7860 /JCDR/2017/28533.10614.
- Boll D, Geomini PM, Brölmann HA. The pre-operative assessment of the adnexal mass: the accuracy of clinical estimates versus clinical prediction rules. BJOG. 2003;110:519-523.
- Glanc P, Benacerraf B, Bourne T, et al. First International Consensus Report on adnexal masses: management recommendations. J Ultrasound Med. 2017;36:849-863. doi: 10.1002/jum.14197.
- Van Holsbeke C, Daemen A, Yazbek J, et al. Ultrasound experience substantially impacts on diagnostic performance and confidence when adnexal masses are classified using pattern recognition. Gynecol Obstet Invest. 2010;69:160-168. doi: 10.1159/000265012.
- Shetty J, Reddy G, Pandey D. Role of sonographic grayscale pattern recognition in the diagnosis of adnexal masses. J Clin Diagn Res. 2017;11:QC12-QC15. doi: 10.7860 /JCDR/2017/28533.10614.
Optimize detection and treatment of iron deficiency in pregnancy
During pregnancy, anemia and iron deficiency are prevalent because the fetus depletes maternal iron stores. Iron deficiency and iron deficiency anemia are not synonymous. Effective screening for iron deficiency in the first trimester of pregnancy requires the measurement of a sensitive and specific biomarker of iron deficiency, such as ferritin. Limiting the measurement of ferritin to the subset of patients with anemia will result in missing many cases of iron deficiency. By the time iron deficiency causes anemia, a severe deficiency is present. Detecting iron deficiency in pregnancy and promptly treating the deficiency will reduce the number of women with anemia in the third trimester and at birth.
Diagnosis of anemia
Anemia in pregnancy is diagnosed by a hemoglobin level and hematocrit concentration below 11 g/dL and 33%, respectively, in the first and third trimesters and below 10.5 g/dL and 32%, respectively, in the second trimester.1 The prevalence of anemia in the first, second, and third trimesters is approximately 3%, 2%, and 11%, respectively.2 At a hemoglobin concentration <11 g/dL, severe maternal morbidity rises significantly.3 The laboratory evaluation of pregnant women with anemia may require assessment of iron stores, measurement of folate and cobalamin (vitamin B12), and hemoglobin electrophoresis, if indicated.
Diagnosis of iron deficiency
Iron deficiency anemia is diagnosed by a ferritin level below 30 ng/mL.4,5 Normal iron stores and iron insufficiency are indicated by ferritin levels 45 to 150 ng/mL and 30 to 44 ng/mL, respectively.4,5 Ferritin is an acute phase reactant, and patients with inflammation or chronic illnesses may have iron deficiency and a normal ferritin level. For these patients, a transferrin saturation (TSAT) <16% would support a diagnosis of iron deficiency.6 TSAT is calculated from measurement of serum iron and total iron binding capacity. TSAT saturation may be elevated by iron supplements, which increase serum iron. If measurement of TSAT is necessary, interference with the measurement accuracy can be minimized by not taking an iron supplement on the day of testing.
Iron deficiency is present in approximately 50% of pregnant women.7,8 The greatest prevalence of iron deficiency in pregnancy is observed in non-Hispanic Black females, followed by Hispanic females. Non-Hispanic White females had the lowest prevalence of iron deficiency.2
Fetal needs for iron often cause the depletion of maternal iron stores. Many pregnant women who have a normal ferritin level in the first trimester will develop iron deficiency in the third trimester, even with the usual recommended daily oral iron supplementation. We recommend measuring ferritin and hemoglobin at the first prenatal visit and again between 24 and 28 weeks’ gestation.
Impact of maternal anemia on maternal and newborn health
Iron plays a critical role in maternal health and fetal development independent of its role in red blood cell formation. Many proteins critical to maternal health and fetal development contain iron, including hemoglobin, myoglobin, cytochromes, ribonucleotide reductase, peroxidases, lipoxygenases, and cyclooxygenases. In the fetus, iron plays an important role in myelination of nerves, dendrite arborization, and synthesis of monoamine neurotransmitters.9
Many studies report that maternal anemia is associated with severe maternal morbidity and adverse newborn outcomes. The current literature must be interpreted with caution because socioeconomic factors influence iron stores. Iron deficiency and anemia is more common among economically and socially disadvantaged populations.10-12 It is possible that repleting iron stores, alone, without addressing social determinants of health, including food and housing insecurity, may be insufficient to improve maternal and newborn health.
Maternal anemia is a risk factor for severe maternal morbidity and adverse newborn outcomes.3,13-18 In a study of 515,270 live births in British Columbia between 2004 and 2016, maternal anemia was diagnosed in 12.8% of mothers.15 Maternal morbidity at birth was increased among patients with mild anemia (hemoglobin concentration of 9 to 10.9 g/dL), including higher rates of intrapartum transfusion (adjusted odds ratio [OR], 2.45; 95% confidence interval [CI], 1.74-3.45), cesarean birth (aOR, 1.17; 95% CI, 1.14-1.19), and chorioamnionitis (aOR, 1.35; 95% CI, 1.27-1.44). Newborn morbidity was also increased among newborns of mothers with mild anemia (hemoglobin concentrations of 9 to 10.9 g/dL), including birth before 37 weeks’ gestation (aOR, 1.09; 95% CI, 1.05-1.12), birth before 32 weeks’ gestation (aOR, 1.30; 95% CI, 1.21-1.39), admission to the intensive care unit (aOR, 1.21; 95% CI, 1.17-1.25), and respiratory distress syndrome (aOR, 1.35; 95% CI, 1.24-1.46).15 Adverse maternal and newborn outcomes were more prevalent among mothers with moderate (hemoglobin concentrations of 7 to 8.9 g/dL) or severe anemia (hemoglobin concentrations of <7 g/dL), compared with mild anemia. For example, compared with mothers with no anemia, mothers with moderate anemia had an increased risk of birth <37 weeks (aOR, 2.26) and birth <32 weeks (aOR, 3.95).15
In a study of 166,566 US pregnant patients, 6.1% were diagnosed with anemia.18 Patients with anemia were more likely to have antepartum thrombosis, preeclampsia, eclampsia, a cesarean birth, postpartum hemorrhage, a blood transfusion, and postpartum thrombosis.18 In this study, the newborns of mothers with anemia were more likely to have a diagnosis of antenatal or intrapartum fetal distress, a 5-minute Apgar score <7, and an admission to the neonatal intensive care unit.
Continue to: Maternal anemia and neurodevelopmental disorders in children...
Maternal anemia and neurodevelopmental disorders in children
Some experts, but not all, believe that iron deficiency during pregnancy may adversely impact fetal neurodevelopment and result in childhood behavior issues. All experts agree that more research is needed to understand if maternal anemia causes mental health issues in newborns. In one meta-analysis, among 20 studies of the association of maternal iron deficiency and newborn neurodevelopment, approximately half the studies reported that low maternal ferritin levels were associated with lower childhood performance on standardized tests of cognitive, motor, verbal, and memory function.19 Another systematic review concluded that the evidence linking maternal iron deficiency and child neurodevelopment is equivocal.20
In a study of 532,232 nonadoptive children born in Sweden from 1987 to 2010, maternal anemia was associated with an increased risk of autism spectrum disorder (ASD), attention-deficit/hyperactivity disorder (ADHD), and intellectual disability (ID).21 In Sweden maternal hemoglobin concentration is measured at 10, 25, and 37 weeks of gestation, permitting comparisons of anemia diagnosed early and late in pregnancy with neurodevelopmental outcomes. The association between anemia and neurodevelopmental disorders was greatest if anemia was diagnosed within the first 30 weeks of pregnancy. Compared with mothers without anemia, maternal anemia diagnosed within the first 30 weeks of pregnancy was associated with higher childhood rates of ASD (4.9% vs 3.5%), ADHD (9.3% vs 7.1%), and ID (3.1% vs 1.3%).21 The differences persisted in analyses that controlled for socioeconomic, maternal, and pregnancy-related factors. In a matched sibling comparison, the diagnosis of maternal anemia within the first 30 weeks of gestation was associated with an increased risk of ASD (OR, 2.25; 95% CI, 1.24-4.11) and ID (OR, 2.59; 95% CI, 1.08-6.22) but not ADHD.21 Other studies have also reported a relationship between maternal anemia and intellectual disability.22,23
Measurement of hemoglobin will identify anemia, but hemoglobin measurement is not sufficiently sensitive to identify most cases of iron deficiency. Measuring ferritin can help to identify cases of iron deficiency before the onset of anemia, permitting early treatment of the nutrient deficiency. In pregnancy, iron deficiency is the prelude to developing anemia. Waiting until anemia occurs to diagnose and treat iron deficiency is suboptimal and may miss a critical window of fetal development that is dependent on maternal iron stores. During pregnancy, ferritin levels decrease as much as 80% between the first and third trimesters, as the fetus utilizes maternal iron stores for its growth.24 We recommend the measurement of ferritin and hemoglobin at the first prenatal visit and again at 24 to 28 weeks’ gestation to optimize early detection and treatment of iron deficiency and reduce the frequency of anemia prior to birth. ●
- American College of Obstetricians and Gynecologists. Anemia in pregnancy. ACOG Practice Bulletin No 233. Obstet Gynecol. 2021;138:e55-64.
- Mei Z, Cogswell ME, Looker AC, et al. Assessment of iron status in US pregnant women from the National Health and Nutrition Examination Survey (NHANES), 1996-2006. Am J Clin Nutr. 2011;93:1312-1320.
- Ray JG, Davidson AJF, Berger H, et al. Haemoglobin levels in early pregnancy and severe maternal morbidity: population-based cohort study. BJOG. 2020;127:1154-1164.
- Mast AE, Blinder MA, Gronowski AM, et al. Clinical utility of the soluble transferrin receptor and comparison with serum ferritin in several populations. Clin Chem. 1998;44:45-51.
- Parvord S, Daru J, Prasannan N, et al. UK Guidelines on the management of iron deficiency in pregnancy. Br J Haematol. 2020;188:819-830.
- Camaschell C. Iron-deficiency anemia. N Engl J Med. 2015;372:1832-1843.
- Auerbach M, Abernathy J, Juul S, et al. Prevalence of iron deficiency in first trimester, nonanemic pregnant women. J Matern Fetal Neonatal Med. 2021;34:1002-1005.
- Teichman J, Nisenbaum R, Lausman A, et al. Suboptimal iron deficiency screening in pregnancy and the impact of socioeconomic status in high-resource setting. Blood Adv. 2021;5:4666-4673.
- Georgieff MK. Long-term brain and behavioral consequences of early iron deficiency. Nutr Rev. 2011;69(suppl 1):S43-S48.
- Bodnar LM, Scanlon KS, Freedman DS, et al. High prevalence of postpartum anemia among low-income women in the United States. Am J Obstet Gynecol. 2001;185:438-443.
- Dondi A, PIccinno V, Morigi F, et al. Food insecurity and major diet-related morbidities in migrating children: a systematic review. Nutrients. 2020;12:379.
- Bodnar LM, Cogswell ME, Scanlon KS. Low income postpartum women are at risk of iron deficiency. J Nutr. 2002;132:2298-2302.
- Drukker L, Hants Y, Farkash R, et al. Iron deficiency anemia at admission for labor and delivery is associated with an increased risk for cesarean section and adverse maternal and neonatal outcomes. Transfusion. 2015;55:2799-2806.
- Rahman MM, Abe SK, Rahman S, et al. Maternal anemia and risk of adverse birth and health outcomes in low- and middle-income countries: systematic review and meta-analysis. Am J Clin Nutr. 2016;103:495-504.
- Smith C, Teng F, Branch E, et al. Maternal and perinatal morbidity and mortality associated with anemia in pregnancy. Obstet Gynecol. 2019;134:1234-1244.
- Parks S, Hoffman MK, Goudar SS, et al. Maternal anaemia and maternal, fetal and neonatal outcomes in a prospective cohort study in India and Pakistan. BJOG. 2019;126:737-743.
- Guignard J, Deneux-Tharaux C, Seco A, et al. Gestational anemia and severe acute maternal morbidity: a population based study. Anesthesia. 2021;76:61-71.
- Harrison RK, Lauhon SR, Colvin ZA, et al. Maternal anemia and severe maternal mortality in a US cohort. Am J Obstet Gynecol MFM. 2021;3:100395.
- Quesada-Pinedo HG, Cassel F, Duijts L, et al. Maternal iron status in pregnancy and child health outcomes after birth: a systematic review and meta-analysis. Nutrients. 2021;13:2221.
- McCann S, Perapoch Amado M, Moore SE. The role of iron in brain development: a systematic review. Nutrients. 2020;12:2001.
- Wiegersma AM, Dalman C, Lee BK, et al. Association of prenatal maternal anemia with neurodevelopmental disorders. JAMA Psychiatry. 2019;76:1294-1304.
- Leonard H, de Klerk N, Bourke J, et al. Maternal health in pregnancy and intellectual disability in the offspring: a population-based study. Ann Epidemiol. 2006;16:448-454.
- Drassinower D, Lavery JA, Friedman AM, et al. The effect of maternal hematocrit on offspring IQ at 4 and 7 years of age: a secondary analysis. BJOG. 2016;123:2087-2093.
- Horton KD, Adetona O, Aguilar-Villalobos M, et al. Changes in the concentration of biochemical indicators of diet and nutritional status of pregnant women across pregnancy trimesters in Trujillo, Peru 2004-2005. Nutrition J. 2013;12:80.
During pregnancy, anemia and iron deficiency are prevalent because the fetus depletes maternal iron stores. Iron deficiency and iron deficiency anemia are not synonymous. Effective screening for iron deficiency in the first trimester of pregnancy requires the measurement of a sensitive and specific biomarker of iron deficiency, such as ferritin. Limiting the measurement of ferritin to the subset of patients with anemia will result in missing many cases of iron deficiency. By the time iron deficiency causes anemia, a severe deficiency is present. Detecting iron deficiency in pregnancy and promptly treating the deficiency will reduce the number of women with anemia in the third trimester and at birth.
Diagnosis of anemia
Anemia in pregnancy is diagnosed by a hemoglobin level and hematocrit concentration below 11 g/dL and 33%, respectively, in the first and third trimesters and below 10.5 g/dL and 32%, respectively, in the second trimester.1 The prevalence of anemia in the first, second, and third trimesters is approximately 3%, 2%, and 11%, respectively.2 At a hemoglobin concentration <11 g/dL, severe maternal morbidity rises significantly.3 The laboratory evaluation of pregnant women with anemia may require assessment of iron stores, measurement of folate and cobalamin (vitamin B12), and hemoglobin electrophoresis, if indicated.
Diagnosis of iron deficiency
Iron deficiency anemia is diagnosed by a ferritin level below 30 ng/mL.4,5 Normal iron stores and iron insufficiency are indicated by ferritin levels 45 to 150 ng/mL and 30 to 44 ng/mL, respectively.4,5 Ferritin is an acute phase reactant, and patients with inflammation or chronic illnesses may have iron deficiency and a normal ferritin level. For these patients, a transferrin saturation (TSAT) <16% would support a diagnosis of iron deficiency.6 TSAT is calculated from measurement of serum iron and total iron binding capacity. TSAT saturation may be elevated by iron supplements, which increase serum iron. If measurement of TSAT is necessary, interference with the measurement accuracy can be minimized by not taking an iron supplement on the day of testing.
Iron deficiency is present in approximately 50% of pregnant women.7,8 The greatest prevalence of iron deficiency in pregnancy is observed in non-Hispanic Black females, followed by Hispanic females. Non-Hispanic White females had the lowest prevalence of iron deficiency.2
Fetal needs for iron often cause the depletion of maternal iron stores. Many pregnant women who have a normal ferritin level in the first trimester will develop iron deficiency in the third trimester, even with the usual recommended daily oral iron supplementation. We recommend measuring ferritin and hemoglobin at the first prenatal visit and again between 24 and 28 weeks’ gestation.
Impact of maternal anemia on maternal and newborn health
Iron plays a critical role in maternal health and fetal development independent of its role in red blood cell formation. Many proteins critical to maternal health and fetal development contain iron, including hemoglobin, myoglobin, cytochromes, ribonucleotide reductase, peroxidases, lipoxygenases, and cyclooxygenases. In the fetus, iron plays an important role in myelination of nerves, dendrite arborization, and synthesis of monoamine neurotransmitters.9
Many studies report that maternal anemia is associated with severe maternal morbidity and adverse newborn outcomes. The current literature must be interpreted with caution because socioeconomic factors influence iron stores. Iron deficiency and anemia is more common among economically and socially disadvantaged populations.10-12 It is possible that repleting iron stores, alone, without addressing social determinants of health, including food and housing insecurity, may be insufficient to improve maternal and newborn health.
Maternal anemia is a risk factor for severe maternal morbidity and adverse newborn outcomes.3,13-18 In a study of 515,270 live births in British Columbia between 2004 and 2016, maternal anemia was diagnosed in 12.8% of mothers.15 Maternal morbidity at birth was increased among patients with mild anemia (hemoglobin concentration of 9 to 10.9 g/dL), including higher rates of intrapartum transfusion (adjusted odds ratio [OR], 2.45; 95% confidence interval [CI], 1.74-3.45), cesarean birth (aOR, 1.17; 95% CI, 1.14-1.19), and chorioamnionitis (aOR, 1.35; 95% CI, 1.27-1.44). Newborn morbidity was also increased among newborns of mothers with mild anemia (hemoglobin concentrations of 9 to 10.9 g/dL), including birth before 37 weeks’ gestation (aOR, 1.09; 95% CI, 1.05-1.12), birth before 32 weeks’ gestation (aOR, 1.30; 95% CI, 1.21-1.39), admission to the intensive care unit (aOR, 1.21; 95% CI, 1.17-1.25), and respiratory distress syndrome (aOR, 1.35; 95% CI, 1.24-1.46).15 Adverse maternal and newborn outcomes were more prevalent among mothers with moderate (hemoglobin concentrations of 7 to 8.9 g/dL) or severe anemia (hemoglobin concentrations of <7 g/dL), compared with mild anemia. For example, compared with mothers with no anemia, mothers with moderate anemia had an increased risk of birth <37 weeks (aOR, 2.26) and birth <32 weeks (aOR, 3.95).15
In a study of 166,566 US pregnant patients, 6.1% were diagnosed with anemia.18 Patients with anemia were more likely to have antepartum thrombosis, preeclampsia, eclampsia, a cesarean birth, postpartum hemorrhage, a blood transfusion, and postpartum thrombosis.18 In this study, the newborns of mothers with anemia were more likely to have a diagnosis of antenatal or intrapartum fetal distress, a 5-minute Apgar score <7, and an admission to the neonatal intensive care unit.
Continue to: Maternal anemia and neurodevelopmental disorders in children...
Maternal anemia and neurodevelopmental disorders in children
Some experts, but not all, believe that iron deficiency during pregnancy may adversely impact fetal neurodevelopment and result in childhood behavior issues. All experts agree that more research is needed to understand if maternal anemia causes mental health issues in newborns. In one meta-analysis, among 20 studies of the association of maternal iron deficiency and newborn neurodevelopment, approximately half the studies reported that low maternal ferritin levels were associated with lower childhood performance on standardized tests of cognitive, motor, verbal, and memory function.19 Another systematic review concluded that the evidence linking maternal iron deficiency and child neurodevelopment is equivocal.20
In a study of 532,232 nonadoptive children born in Sweden from 1987 to 2010, maternal anemia was associated with an increased risk of autism spectrum disorder (ASD), attention-deficit/hyperactivity disorder (ADHD), and intellectual disability (ID).21 In Sweden maternal hemoglobin concentration is measured at 10, 25, and 37 weeks of gestation, permitting comparisons of anemia diagnosed early and late in pregnancy with neurodevelopmental outcomes. The association between anemia and neurodevelopmental disorders was greatest if anemia was diagnosed within the first 30 weeks of pregnancy. Compared with mothers without anemia, maternal anemia diagnosed within the first 30 weeks of pregnancy was associated with higher childhood rates of ASD (4.9% vs 3.5%), ADHD (9.3% vs 7.1%), and ID (3.1% vs 1.3%).21 The differences persisted in analyses that controlled for socioeconomic, maternal, and pregnancy-related factors. In a matched sibling comparison, the diagnosis of maternal anemia within the first 30 weeks of gestation was associated with an increased risk of ASD (OR, 2.25; 95% CI, 1.24-4.11) and ID (OR, 2.59; 95% CI, 1.08-6.22) but not ADHD.21 Other studies have also reported a relationship between maternal anemia and intellectual disability.22,23
Measurement of hemoglobin will identify anemia, but hemoglobin measurement is not sufficiently sensitive to identify most cases of iron deficiency. Measuring ferritin can help to identify cases of iron deficiency before the onset of anemia, permitting early treatment of the nutrient deficiency. In pregnancy, iron deficiency is the prelude to developing anemia. Waiting until anemia occurs to diagnose and treat iron deficiency is suboptimal and may miss a critical window of fetal development that is dependent on maternal iron stores. During pregnancy, ferritin levels decrease as much as 80% between the first and third trimesters, as the fetus utilizes maternal iron stores for its growth.24 We recommend the measurement of ferritin and hemoglobin at the first prenatal visit and again at 24 to 28 weeks’ gestation to optimize early detection and treatment of iron deficiency and reduce the frequency of anemia prior to birth. ●
During pregnancy, anemia and iron deficiency are prevalent because the fetus depletes maternal iron stores. Iron deficiency and iron deficiency anemia are not synonymous. Effective screening for iron deficiency in the first trimester of pregnancy requires the measurement of a sensitive and specific biomarker of iron deficiency, such as ferritin. Limiting the measurement of ferritin to the subset of patients with anemia will result in missing many cases of iron deficiency. By the time iron deficiency causes anemia, a severe deficiency is present. Detecting iron deficiency in pregnancy and promptly treating the deficiency will reduce the number of women with anemia in the third trimester and at birth.
Diagnosis of anemia
Anemia in pregnancy is diagnosed by a hemoglobin level and hematocrit concentration below 11 g/dL and 33%, respectively, in the first and third trimesters and below 10.5 g/dL and 32%, respectively, in the second trimester.1 The prevalence of anemia in the first, second, and third trimesters is approximately 3%, 2%, and 11%, respectively.2 At a hemoglobin concentration <11 g/dL, severe maternal morbidity rises significantly.3 The laboratory evaluation of pregnant women with anemia may require assessment of iron stores, measurement of folate and cobalamin (vitamin B12), and hemoglobin electrophoresis, if indicated.
Diagnosis of iron deficiency
Iron deficiency anemia is diagnosed by a ferritin level below 30 ng/mL.4,5 Normal iron stores and iron insufficiency are indicated by ferritin levels 45 to 150 ng/mL and 30 to 44 ng/mL, respectively.4,5 Ferritin is an acute phase reactant, and patients with inflammation or chronic illnesses may have iron deficiency and a normal ferritin level. For these patients, a transferrin saturation (TSAT) <16% would support a diagnosis of iron deficiency.6 TSAT is calculated from measurement of serum iron and total iron binding capacity. TSAT saturation may be elevated by iron supplements, which increase serum iron. If measurement of TSAT is necessary, interference with the measurement accuracy can be minimized by not taking an iron supplement on the day of testing.
Iron deficiency is present in approximately 50% of pregnant women.7,8 The greatest prevalence of iron deficiency in pregnancy is observed in non-Hispanic Black females, followed by Hispanic females. Non-Hispanic White females had the lowest prevalence of iron deficiency.2
Fetal needs for iron often cause the depletion of maternal iron stores. Many pregnant women who have a normal ferritin level in the first trimester will develop iron deficiency in the third trimester, even with the usual recommended daily oral iron supplementation. We recommend measuring ferritin and hemoglobin at the first prenatal visit and again between 24 and 28 weeks’ gestation.
Impact of maternal anemia on maternal and newborn health
Iron plays a critical role in maternal health and fetal development independent of its role in red blood cell formation. Many proteins critical to maternal health and fetal development contain iron, including hemoglobin, myoglobin, cytochromes, ribonucleotide reductase, peroxidases, lipoxygenases, and cyclooxygenases. In the fetus, iron plays an important role in myelination of nerves, dendrite arborization, and synthesis of monoamine neurotransmitters.9
Many studies report that maternal anemia is associated with severe maternal morbidity and adverse newborn outcomes. The current literature must be interpreted with caution because socioeconomic factors influence iron stores. Iron deficiency and anemia is more common among economically and socially disadvantaged populations.10-12 It is possible that repleting iron stores, alone, without addressing social determinants of health, including food and housing insecurity, may be insufficient to improve maternal and newborn health.
Maternal anemia is a risk factor for severe maternal morbidity and adverse newborn outcomes.3,13-18 In a study of 515,270 live births in British Columbia between 2004 and 2016, maternal anemia was diagnosed in 12.8% of mothers.15 Maternal morbidity at birth was increased among patients with mild anemia (hemoglobin concentration of 9 to 10.9 g/dL), including higher rates of intrapartum transfusion (adjusted odds ratio [OR], 2.45; 95% confidence interval [CI], 1.74-3.45), cesarean birth (aOR, 1.17; 95% CI, 1.14-1.19), and chorioamnionitis (aOR, 1.35; 95% CI, 1.27-1.44). Newborn morbidity was also increased among newborns of mothers with mild anemia (hemoglobin concentrations of 9 to 10.9 g/dL), including birth before 37 weeks’ gestation (aOR, 1.09; 95% CI, 1.05-1.12), birth before 32 weeks’ gestation (aOR, 1.30; 95% CI, 1.21-1.39), admission to the intensive care unit (aOR, 1.21; 95% CI, 1.17-1.25), and respiratory distress syndrome (aOR, 1.35; 95% CI, 1.24-1.46).15 Adverse maternal and newborn outcomes were more prevalent among mothers with moderate (hemoglobin concentrations of 7 to 8.9 g/dL) or severe anemia (hemoglobin concentrations of <7 g/dL), compared with mild anemia. For example, compared with mothers with no anemia, mothers with moderate anemia had an increased risk of birth <37 weeks (aOR, 2.26) and birth <32 weeks (aOR, 3.95).15
In a study of 166,566 US pregnant patients, 6.1% were diagnosed with anemia.18 Patients with anemia were more likely to have antepartum thrombosis, preeclampsia, eclampsia, a cesarean birth, postpartum hemorrhage, a blood transfusion, and postpartum thrombosis.18 In this study, the newborns of mothers with anemia were more likely to have a diagnosis of antenatal or intrapartum fetal distress, a 5-minute Apgar score <7, and an admission to the neonatal intensive care unit.
Continue to: Maternal anemia and neurodevelopmental disorders in children...
Maternal anemia and neurodevelopmental disorders in children
Some experts, but not all, believe that iron deficiency during pregnancy may adversely impact fetal neurodevelopment and result in childhood behavior issues. All experts agree that more research is needed to understand if maternal anemia causes mental health issues in newborns. In one meta-analysis, among 20 studies of the association of maternal iron deficiency and newborn neurodevelopment, approximately half the studies reported that low maternal ferritin levels were associated with lower childhood performance on standardized tests of cognitive, motor, verbal, and memory function.19 Another systematic review concluded that the evidence linking maternal iron deficiency and child neurodevelopment is equivocal.20
In a study of 532,232 nonadoptive children born in Sweden from 1987 to 2010, maternal anemia was associated with an increased risk of autism spectrum disorder (ASD), attention-deficit/hyperactivity disorder (ADHD), and intellectual disability (ID).21 In Sweden maternal hemoglobin concentration is measured at 10, 25, and 37 weeks of gestation, permitting comparisons of anemia diagnosed early and late in pregnancy with neurodevelopmental outcomes. The association between anemia and neurodevelopmental disorders was greatest if anemia was diagnosed within the first 30 weeks of pregnancy. Compared with mothers without anemia, maternal anemia diagnosed within the first 30 weeks of pregnancy was associated with higher childhood rates of ASD (4.9% vs 3.5%), ADHD (9.3% vs 7.1%), and ID (3.1% vs 1.3%).21 The differences persisted in analyses that controlled for socioeconomic, maternal, and pregnancy-related factors. In a matched sibling comparison, the diagnosis of maternal anemia within the first 30 weeks of gestation was associated with an increased risk of ASD (OR, 2.25; 95% CI, 1.24-4.11) and ID (OR, 2.59; 95% CI, 1.08-6.22) but not ADHD.21 Other studies have also reported a relationship between maternal anemia and intellectual disability.22,23
Measurement of hemoglobin will identify anemia, but hemoglobin measurement is not sufficiently sensitive to identify most cases of iron deficiency. Measuring ferritin can help to identify cases of iron deficiency before the onset of anemia, permitting early treatment of the nutrient deficiency. In pregnancy, iron deficiency is the prelude to developing anemia. Waiting until anemia occurs to diagnose and treat iron deficiency is suboptimal and may miss a critical window of fetal development that is dependent on maternal iron stores. During pregnancy, ferritin levels decrease as much as 80% between the first and third trimesters, as the fetus utilizes maternal iron stores for its growth.24 We recommend the measurement of ferritin and hemoglobin at the first prenatal visit and again at 24 to 28 weeks’ gestation to optimize early detection and treatment of iron deficiency and reduce the frequency of anemia prior to birth. ●
- American College of Obstetricians and Gynecologists. Anemia in pregnancy. ACOG Practice Bulletin No 233. Obstet Gynecol. 2021;138:e55-64.
- Mei Z, Cogswell ME, Looker AC, et al. Assessment of iron status in US pregnant women from the National Health and Nutrition Examination Survey (NHANES), 1996-2006. Am J Clin Nutr. 2011;93:1312-1320.
- Ray JG, Davidson AJF, Berger H, et al. Haemoglobin levels in early pregnancy and severe maternal morbidity: population-based cohort study. BJOG. 2020;127:1154-1164.
- Mast AE, Blinder MA, Gronowski AM, et al. Clinical utility of the soluble transferrin receptor and comparison with serum ferritin in several populations. Clin Chem. 1998;44:45-51.
- Parvord S, Daru J, Prasannan N, et al. UK Guidelines on the management of iron deficiency in pregnancy. Br J Haematol. 2020;188:819-830.
- Camaschell C. Iron-deficiency anemia. N Engl J Med. 2015;372:1832-1843.
- Auerbach M, Abernathy J, Juul S, et al. Prevalence of iron deficiency in first trimester, nonanemic pregnant women. J Matern Fetal Neonatal Med. 2021;34:1002-1005.
- Teichman J, Nisenbaum R, Lausman A, et al. Suboptimal iron deficiency screening in pregnancy and the impact of socioeconomic status in high-resource setting. Blood Adv. 2021;5:4666-4673.
- Georgieff MK. Long-term brain and behavioral consequences of early iron deficiency. Nutr Rev. 2011;69(suppl 1):S43-S48.
- Bodnar LM, Scanlon KS, Freedman DS, et al. High prevalence of postpartum anemia among low-income women in the United States. Am J Obstet Gynecol. 2001;185:438-443.
- Dondi A, PIccinno V, Morigi F, et al. Food insecurity and major diet-related morbidities in migrating children: a systematic review. Nutrients. 2020;12:379.
- Bodnar LM, Cogswell ME, Scanlon KS. Low income postpartum women are at risk of iron deficiency. J Nutr. 2002;132:2298-2302.
- Drukker L, Hants Y, Farkash R, et al. Iron deficiency anemia at admission for labor and delivery is associated with an increased risk for cesarean section and adverse maternal and neonatal outcomes. Transfusion. 2015;55:2799-2806.
- Rahman MM, Abe SK, Rahman S, et al. Maternal anemia and risk of adverse birth and health outcomes in low- and middle-income countries: systematic review and meta-analysis. Am J Clin Nutr. 2016;103:495-504.
- Smith C, Teng F, Branch E, et al. Maternal and perinatal morbidity and mortality associated with anemia in pregnancy. Obstet Gynecol. 2019;134:1234-1244.
- Parks S, Hoffman MK, Goudar SS, et al. Maternal anaemia and maternal, fetal and neonatal outcomes in a prospective cohort study in India and Pakistan. BJOG. 2019;126:737-743.
- Guignard J, Deneux-Tharaux C, Seco A, et al. Gestational anemia and severe acute maternal morbidity: a population based study. Anesthesia. 2021;76:61-71.
- Harrison RK, Lauhon SR, Colvin ZA, et al. Maternal anemia and severe maternal mortality in a US cohort. Am J Obstet Gynecol MFM. 2021;3:100395.
- Quesada-Pinedo HG, Cassel F, Duijts L, et al. Maternal iron status in pregnancy and child health outcomes after birth: a systematic review and meta-analysis. Nutrients. 2021;13:2221.
- McCann S, Perapoch Amado M, Moore SE. The role of iron in brain development: a systematic review. Nutrients. 2020;12:2001.
- Wiegersma AM, Dalman C, Lee BK, et al. Association of prenatal maternal anemia with neurodevelopmental disorders. JAMA Psychiatry. 2019;76:1294-1304.
- Leonard H, de Klerk N, Bourke J, et al. Maternal health in pregnancy and intellectual disability in the offspring: a population-based study. Ann Epidemiol. 2006;16:448-454.
- Drassinower D, Lavery JA, Friedman AM, et al. The effect of maternal hematocrit on offspring IQ at 4 and 7 years of age: a secondary analysis. BJOG. 2016;123:2087-2093.
- Horton KD, Adetona O, Aguilar-Villalobos M, et al. Changes in the concentration of biochemical indicators of diet and nutritional status of pregnant women across pregnancy trimesters in Trujillo, Peru 2004-2005. Nutrition J. 2013;12:80.
- American College of Obstetricians and Gynecologists. Anemia in pregnancy. ACOG Practice Bulletin No 233. Obstet Gynecol. 2021;138:e55-64.
- Mei Z, Cogswell ME, Looker AC, et al. Assessment of iron status in US pregnant women from the National Health and Nutrition Examination Survey (NHANES), 1996-2006. Am J Clin Nutr. 2011;93:1312-1320.
- Ray JG, Davidson AJF, Berger H, et al. Haemoglobin levels in early pregnancy and severe maternal morbidity: population-based cohort study. BJOG. 2020;127:1154-1164.
- Mast AE, Blinder MA, Gronowski AM, et al. Clinical utility of the soluble transferrin receptor and comparison with serum ferritin in several populations. Clin Chem. 1998;44:45-51.
- Parvord S, Daru J, Prasannan N, et al. UK Guidelines on the management of iron deficiency in pregnancy. Br J Haematol. 2020;188:819-830.
- Camaschell C. Iron-deficiency anemia. N Engl J Med. 2015;372:1832-1843.
- Auerbach M, Abernathy J, Juul S, et al. Prevalence of iron deficiency in first trimester, nonanemic pregnant women. J Matern Fetal Neonatal Med. 2021;34:1002-1005.
- Teichman J, Nisenbaum R, Lausman A, et al. Suboptimal iron deficiency screening in pregnancy and the impact of socioeconomic status in high-resource setting. Blood Adv. 2021;5:4666-4673.
- Georgieff MK. Long-term brain and behavioral consequences of early iron deficiency. Nutr Rev. 2011;69(suppl 1):S43-S48.
- Bodnar LM, Scanlon KS, Freedman DS, et al. High prevalence of postpartum anemia among low-income women in the United States. Am J Obstet Gynecol. 2001;185:438-443.
- Dondi A, PIccinno V, Morigi F, et al. Food insecurity and major diet-related morbidities in migrating children: a systematic review. Nutrients. 2020;12:379.
- Bodnar LM, Cogswell ME, Scanlon KS. Low income postpartum women are at risk of iron deficiency. J Nutr. 2002;132:2298-2302.
- Drukker L, Hants Y, Farkash R, et al. Iron deficiency anemia at admission for labor and delivery is associated with an increased risk for cesarean section and adverse maternal and neonatal outcomes. Transfusion. 2015;55:2799-2806.
- Rahman MM, Abe SK, Rahman S, et al. Maternal anemia and risk of adverse birth and health outcomes in low- and middle-income countries: systematic review and meta-analysis. Am J Clin Nutr. 2016;103:495-504.
- Smith C, Teng F, Branch E, et al. Maternal and perinatal morbidity and mortality associated with anemia in pregnancy. Obstet Gynecol. 2019;134:1234-1244.
- Parks S, Hoffman MK, Goudar SS, et al. Maternal anaemia and maternal, fetal and neonatal outcomes in a prospective cohort study in India and Pakistan. BJOG. 2019;126:737-743.
- Guignard J, Deneux-Tharaux C, Seco A, et al. Gestational anemia and severe acute maternal morbidity: a population based study. Anesthesia. 2021;76:61-71.
- Harrison RK, Lauhon SR, Colvin ZA, et al. Maternal anemia and severe maternal mortality in a US cohort. Am J Obstet Gynecol MFM. 2021;3:100395.
- Quesada-Pinedo HG, Cassel F, Duijts L, et al. Maternal iron status in pregnancy and child health outcomes after birth: a systematic review and meta-analysis. Nutrients. 2021;13:2221.
- McCann S, Perapoch Amado M, Moore SE. The role of iron in brain development: a systematic review. Nutrients. 2020;12:2001.
- Wiegersma AM, Dalman C, Lee BK, et al. Association of prenatal maternal anemia with neurodevelopmental disorders. JAMA Psychiatry. 2019;76:1294-1304.
- Leonard H, de Klerk N, Bourke J, et al. Maternal health in pregnancy and intellectual disability in the offspring: a population-based study. Ann Epidemiol. 2006;16:448-454.
- Drassinower D, Lavery JA, Friedman AM, et al. The effect of maternal hematocrit on offspring IQ at 4 and 7 years of age: a secondary analysis. BJOG. 2016;123:2087-2093.
- Horton KD, Adetona O, Aguilar-Villalobos M, et al. Changes in the concentration of biochemical indicators of diet and nutritional status of pregnant women across pregnancy trimesters in Trujillo, Peru 2004-2005. Nutrition J. 2013;12:80.
TNF blockers beat newer biologics in Crohn’s disease: Meta-analysis
Tumor necrosis factor (TNF)–alpha inhibitors achieve better endoscopic healing than the newer biologic drugs vedolizumab (Entyvio) and ustekinumab (Stelara) in moderate to severe Crohn’s disease, a new meta-analysis suggests.
The advantage for the TNF blockers infliximab (Remicade) and adalimumab (Humira) came in treating larger ileal ulcers and colonic disease.
This finding could help physicians choose among the four biologic drugs approved in recent years in the United States, Canada, and Western Europe to treat this disease. None of these drugs has emerged as clearly superior to all the others.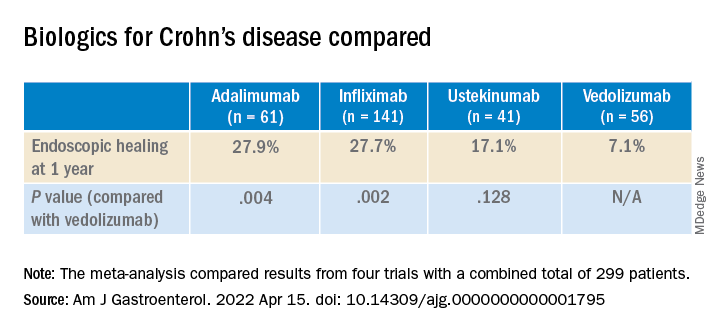
“For patients with high-risk or difficult-to-treat disease, such as those with larger ileal ulcers, the use of anti-TNF may be preferable as a first-line option,” said lead author Neeraj Narula, MD, MPH, of the department of medicine at McMaster University in Toronto, in an email to this news organization.
The study was published online in the American Journal of Gastroenterology.
Few head-to-head trials
In contrast to the TNF blockers infliximab and adalimumab, ustekinumab blocks interleukin-12 and interleukin-23, and vedolizumab blocks integrin–alpha4-beta7.
Only one trial, SEAVUE, has compared any of these drugs head to head for the treatment of Crohn’s disease. This trial found no difference between ustekinumab and adalimumab in rates of clinical remission or endoscopic healing. However, the patients in the trial had a relatively low baseline Simple Endoscopic Score for Crohn’s disease (SES-CD).
In the VARSITY trial, vedolizumab showed better results than adalimumab in clinical remission and endoscopic improvement, but that trial involved patients with ulcerative colitis.
“None of these medications are clearly head and shoulders above the rest; they all work in similar ways,” said Simon Hong, MD, of the Inflammatory Bowel Disease Center at New York University Langone Health, who was not involved in the study. “It’s not clear, at least from a rigorous scientific standpoint, which is better.”
Four biologic drugs compared
In their meta-analysis, Dr. Narula and colleagues compared results from four previous trials, which combined had a total of 299 patients. The investigators assessed the difference in results for specific ileocolonic segments. They focused on endoscopic healing because it is believed to be a more reliable indicator of long-term health than symptoms, which are more susceptible to the placebo effect.
Although the rates of endoscopic healing were low overall, they were significantly better for the TNF blockers than with the newer drugs. The difference between ustekinumab and vedolizumab was not statistically significant.
Among patients with a baseline ileal SES-CD of 3 or greater, the researchers found no significant differences between biologics for 1-year ileal endoscopic healing.
But in patients with ileal ulcers larger than 0.5 cm, the ulcers disappeared after a year in 40.8% of patients who took infliximab vs. 30% of those who took adalimumab, 17.7% of those who took ustekinumab, and 8.7% of those who took vedolizumab. Compared to vedolizumab, the difference was statistically significant for infliximab (P = .045) but not for adalimumab (P = .077) or ustekinumab (P = .259).
Among those patients who had at least one colonic segment with an SES-CD of 3 or greater, the patients taking adalimumab did the best, with 62.5% achieving endoscopic healing of the colon. The rate with infliximab was 52.4%. For vedolizumab, the rate was 31.3%, and for ustekinumab, it was 29.0%. Only the differences between the TNF blockers and the newer biologics were statistically significant for this comparison.
In general, the ileum does not heal as well as the colon, Dr. Narula and colleagues note.
“This confirms, or at least supports, our experience,” Dr. Hong told this news organization. The explanation for the greater efficacy of the TNF blockers could be their more systemic mechanism of action, he said.
The study authors acknowledge that their meta-analysis cannot take the place of true head-to-head trials.
“Safety, convenience, and cost of therapy all are relevant factors that impact decision-making, and the availability of biosimilar TNF-alpha antagonist therapies in routine practice adds additional consideration for cost-effectiveness in population health decisions,” Dr. Narula said.
The study was self-funded. Dr. Narula has received honoraria from Janssen, AbbVie, Takeda, Pfizer, Merck, Sandoz, Novartis, and Ferring. Dr. Hong reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Tumor necrosis factor (TNF)–alpha inhibitors achieve better endoscopic healing than the newer biologic drugs vedolizumab (Entyvio) and ustekinumab (Stelara) in moderate to severe Crohn’s disease, a new meta-analysis suggests.
The advantage for the TNF blockers infliximab (Remicade) and adalimumab (Humira) came in treating larger ileal ulcers and colonic disease.
This finding could help physicians choose among the four biologic drugs approved in recent years in the United States, Canada, and Western Europe to treat this disease. None of these drugs has emerged as clearly superior to all the others.
“For patients with high-risk or difficult-to-treat disease, such as those with larger ileal ulcers, the use of anti-TNF may be preferable as a first-line option,” said lead author Neeraj Narula, MD, MPH, of the department of medicine at McMaster University in Toronto, in an email to this news organization.
The study was published online in the American Journal of Gastroenterology.
Few head-to-head trials
In contrast to the TNF blockers infliximab and adalimumab, ustekinumab blocks interleukin-12 and interleukin-23, and vedolizumab blocks integrin–alpha4-beta7.
Only one trial, SEAVUE, has compared any of these drugs head to head for the treatment of Crohn’s disease. This trial found no difference between ustekinumab and adalimumab in rates of clinical remission or endoscopic healing. However, the patients in the trial had a relatively low baseline Simple Endoscopic Score for Crohn’s disease (SES-CD).
In the VARSITY trial, vedolizumab showed better results than adalimumab in clinical remission and endoscopic improvement, but that trial involved patients with ulcerative colitis.
“None of these medications are clearly head and shoulders above the rest; they all work in similar ways,” said Simon Hong, MD, of the Inflammatory Bowel Disease Center at New York University Langone Health, who was not involved in the study. “It’s not clear, at least from a rigorous scientific standpoint, which is better.”
Four biologic drugs compared
In their meta-analysis, Dr. Narula and colleagues compared results from four previous trials, which combined had a total of 299 patients. The investigators assessed the difference in results for specific ileocolonic segments. They focused on endoscopic healing because it is believed to be a more reliable indicator of long-term health than symptoms, which are more susceptible to the placebo effect.
Although the rates of endoscopic healing were low overall, they were significantly better for the TNF blockers than with the newer drugs. The difference between ustekinumab and vedolizumab was not statistically significant.
Among patients with a baseline ileal SES-CD of 3 or greater, the researchers found no significant differences between biologics for 1-year ileal endoscopic healing.
But in patients with ileal ulcers larger than 0.5 cm, the ulcers disappeared after a year in 40.8% of patients who took infliximab vs. 30% of those who took adalimumab, 17.7% of those who took ustekinumab, and 8.7% of those who took vedolizumab. Compared to vedolizumab, the difference was statistically significant for infliximab (P = .045) but not for adalimumab (P = .077) or ustekinumab (P = .259).
Among those patients who had at least one colonic segment with an SES-CD of 3 or greater, the patients taking adalimumab did the best, with 62.5% achieving endoscopic healing of the colon. The rate with infliximab was 52.4%. For vedolizumab, the rate was 31.3%, and for ustekinumab, it was 29.0%. Only the differences between the TNF blockers and the newer biologics were statistically significant for this comparison.
In general, the ileum does not heal as well as the colon, Dr. Narula and colleagues note.
“This confirms, or at least supports, our experience,” Dr. Hong told this news organization. The explanation for the greater efficacy of the TNF blockers could be their more systemic mechanism of action, he said.
The study authors acknowledge that their meta-analysis cannot take the place of true head-to-head trials.
“Safety, convenience, and cost of therapy all are relevant factors that impact decision-making, and the availability of biosimilar TNF-alpha antagonist therapies in routine practice adds additional consideration for cost-effectiveness in population health decisions,” Dr. Narula said.
The study was self-funded. Dr. Narula has received honoraria from Janssen, AbbVie, Takeda, Pfizer, Merck, Sandoz, Novartis, and Ferring. Dr. Hong reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Tumor necrosis factor (TNF)–alpha inhibitors achieve better endoscopic healing than the newer biologic drugs vedolizumab (Entyvio) and ustekinumab (Stelara) in moderate to severe Crohn’s disease, a new meta-analysis suggests.
The advantage for the TNF blockers infliximab (Remicade) and adalimumab (Humira) came in treating larger ileal ulcers and colonic disease.
This finding could help physicians choose among the four biologic drugs approved in recent years in the United States, Canada, and Western Europe to treat this disease. None of these drugs has emerged as clearly superior to all the others.
“For patients with high-risk or difficult-to-treat disease, such as those with larger ileal ulcers, the use of anti-TNF may be preferable as a first-line option,” said lead author Neeraj Narula, MD, MPH, of the department of medicine at McMaster University in Toronto, in an email to this news organization.
The study was published online in the American Journal of Gastroenterology.
Few head-to-head trials
In contrast to the TNF blockers infliximab and adalimumab, ustekinumab blocks interleukin-12 and interleukin-23, and vedolizumab blocks integrin–alpha4-beta7.
Only one trial, SEAVUE, has compared any of these drugs head to head for the treatment of Crohn’s disease. This trial found no difference between ustekinumab and adalimumab in rates of clinical remission or endoscopic healing. However, the patients in the trial had a relatively low baseline Simple Endoscopic Score for Crohn’s disease (SES-CD).
In the VARSITY trial, vedolizumab showed better results than adalimumab in clinical remission and endoscopic improvement, but that trial involved patients with ulcerative colitis.
“None of these medications are clearly head and shoulders above the rest; they all work in similar ways,” said Simon Hong, MD, of the Inflammatory Bowel Disease Center at New York University Langone Health, who was not involved in the study. “It’s not clear, at least from a rigorous scientific standpoint, which is better.”
Four biologic drugs compared
In their meta-analysis, Dr. Narula and colleagues compared results from four previous trials, which combined had a total of 299 patients. The investigators assessed the difference in results for specific ileocolonic segments. They focused on endoscopic healing because it is believed to be a more reliable indicator of long-term health than symptoms, which are more susceptible to the placebo effect.
Although the rates of endoscopic healing were low overall, they were significantly better for the TNF blockers than with the newer drugs. The difference between ustekinumab and vedolizumab was not statistically significant.
Among patients with a baseline ileal SES-CD of 3 or greater, the researchers found no significant differences between biologics for 1-year ileal endoscopic healing.
But in patients with ileal ulcers larger than 0.5 cm, the ulcers disappeared after a year in 40.8% of patients who took infliximab vs. 30% of those who took adalimumab, 17.7% of those who took ustekinumab, and 8.7% of those who took vedolizumab. Compared to vedolizumab, the difference was statistically significant for infliximab (P = .045) but not for adalimumab (P = .077) or ustekinumab (P = .259).
Among those patients who had at least one colonic segment with an SES-CD of 3 or greater, the patients taking adalimumab did the best, with 62.5% achieving endoscopic healing of the colon. The rate with infliximab was 52.4%. For vedolizumab, the rate was 31.3%, and for ustekinumab, it was 29.0%. Only the differences between the TNF blockers and the newer biologics were statistically significant for this comparison.
In general, the ileum does not heal as well as the colon, Dr. Narula and colleagues note.
“This confirms, or at least supports, our experience,” Dr. Hong told this news organization. The explanation for the greater efficacy of the TNF blockers could be their more systemic mechanism of action, he said.
The study authors acknowledge that their meta-analysis cannot take the place of true head-to-head trials.
“Safety, convenience, and cost of therapy all are relevant factors that impact decision-making, and the availability of biosimilar TNF-alpha antagonist therapies in routine practice adds additional consideration for cost-effectiveness in population health decisions,” Dr. Narula said.
The study was self-funded. Dr. Narula has received honoraria from Janssen, AbbVie, Takeda, Pfizer, Merck, Sandoz, Novartis, and Ferring. Dr. Hong reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE AMERICAN JOURNAL OF GASTROENTEROLOGY
DOJ complaint flags HCV drug denials for people with addiction
A complaint filed with the U.S. Department of Justice (DOJ) alleges that Alabama’s Medicaid program is illegally denying curative drug treatment for hepatitis C virus (HCV) infection to people with substance use disorder.
The complaint was filed May 9 by the Center for Health Law and Policy Innovation (CHLPI) of Harvard Law School, in partnership with AIDS Alabama.
It alleges that Alabama Medicaid has a policy of denying HCV treatment to people who have used illegal drugs or alcohol in the past 6 months.
CHLPI and AIDS Alabama argue that these restrictions violate the Americans With Disabilities Act, which protects people who are disabled because of substance use disorder.
“Forced sobriety policies don’t just unfairly prevent people with substance use disorder from accessing life-saving treatment; they also severely hamper public health efforts to stop the spread of the disease,” Kevin Costello, CHLPI’s litigation director, said in a statement.
“These policies are rooted in stigma, not science, and they violate antidiscrimination provisions of the Americans With Disabilities Act,” Mr. Costello said.
Filing an administrative complaint against Alabama is “an important milestone in fighting sobriety restrictions,” he added.
Morally wrong
Kathie Hiers, CEO of AIDS Alabama, noted that Alabama’s health outcomes are among the worst in the nation.
“Policies that prevent adequate medical care from being provided must end. HCV now has a cure, and withholding that cure from Alabamians based on a moral judgment is wrong and certainly doesn’t follow the science,” Ms. Hiers added.
Direct-acting antiviral (DAA) therapy can cure up to 99% of people living with HCV.
The complaint against Alabama Medicaid builds on CHLPI’s successful policy advocacy and litigation campaigns to expand access to DAA therapy in state Medicaid programs across the country.
Since 2017, 19 states have removed treatment restrictions that were based on drug or alcohol use. In other states, however, “severe, illegal sobriety restrictions remain,” according to CHLPI.
Alabama, Mississippi, Arkansas, South Carolina, and South Dakota still require Medicaid enrollees with HCV to prove they have not used drugs or alcohol for 6 months before they can receive treatment. Iowa, North Dakota, and West Virginia have a 3-month abstinence requirement.
The American Association for the Study of Liver Diseases and the Infectious Diseases Society of America recommend DAA therapy for all patients with chronic HCV infection, regardless of drug or alcohol use.
CHLPI intends to expand this “enforcement campaign” to all states where sobriety restrictions persist.
A version of this article first appeared on Medscape.com.
A complaint filed with the U.S. Department of Justice (DOJ) alleges that Alabama’s Medicaid program is illegally denying curative drug treatment for hepatitis C virus (HCV) infection to people with substance use disorder.
The complaint was filed May 9 by the Center for Health Law and Policy Innovation (CHLPI) of Harvard Law School, in partnership with AIDS Alabama.
It alleges that Alabama Medicaid has a policy of denying HCV treatment to people who have used illegal drugs or alcohol in the past 6 months.
CHLPI and AIDS Alabama argue that these restrictions violate the Americans With Disabilities Act, which protects people who are disabled because of substance use disorder.
“Forced sobriety policies don’t just unfairly prevent people with substance use disorder from accessing life-saving treatment; they also severely hamper public health efforts to stop the spread of the disease,” Kevin Costello, CHLPI’s litigation director, said in a statement.
“These policies are rooted in stigma, not science, and they violate antidiscrimination provisions of the Americans With Disabilities Act,” Mr. Costello said.
Filing an administrative complaint against Alabama is “an important milestone in fighting sobriety restrictions,” he added.
Morally wrong
Kathie Hiers, CEO of AIDS Alabama, noted that Alabama’s health outcomes are among the worst in the nation.
“Policies that prevent adequate medical care from being provided must end. HCV now has a cure, and withholding that cure from Alabamians based on a moral judgment is wrong and certainly doesn’t follow the science,” Ms. Hiers added.
Direct-acting antiviral (DAA) therapy can cure up to 99% of people living with HCV.
The complaint against Alabama Medicaid builds on CHLPI’s successful policy advocacy and litigation campaigns to expand access to DAA therapy in state Medicaid programs across the country.
Since 2017, 19 states have removed treatment restrictions that were based on drug or alcohol use. In other states, however, “severe, illegal sobriety restrictions remain,” according to CHLPI.
Alabama, Mississippi, Arkansas, South Carolina, and South Dakota still require Medicaid enrollees with HCV to prove they have not used drugs or alcohol for 6 months before they can receive treatment. Iowa, North Dakota, and West Virginia have a 3-month abstinence requirement.
The American Association for the Study of Liver Diseases and the Infectious Diseases Society of America recommend DAA therapy for all patients with chronic HCV infection, regardless of drug or alcohol use.
CHLPI intends to expand this “enforcement campaign” to all states where sobriety restrictions persist.
A version of this article first appeared on Medscape.com.
A complaint filed with the U.S. Department of Justice (DOJ) alleges that Alabama’s Medicaid program is illegally denying curative drug treatment for hepatitis C virus (HCV) infection to people with substance use disorder.
The complaint was filed May 9 by the Center for Health Law and Policy Innovation (CHLPI) of Harvard Law School, in partnership with AIDS Alabama.
It alleges that Alabama Medicaid has a policy of denying HCV treatment to people who have used illegal drugs or alcohol in the past 6 months.
CHLPI and AIDS Alabama argue that these restrictions violate the Americans With Disabilities Act, which protects people who are disabled because of substance use disorder.
“Forced sobriety policies don’t just unfairly prevent people with substance use disorder from accessing life-saving treatment; they also severely hamper public health efforts to stop the spread of the disease,” Kevin Costello, CHLPI’s litigation director, said in a statement.
“These policies are rooted in stigma, not science, and they violate antidiscrimination provisions of the Americans With Disabilities Act,” Mr. Costello said.
Filing an administrative complaint against Alabama is “an important milestone in fighting sobriety restrictions,” he added.
Morally wrong
Kathie Hiers, CEO of AIDS Alabama, noted that Alabama’s health outcomes are among the worst in the nation.
“Policies that prevent adequate medical care from being provided must end. HCV now has a cure, and withholding that cure from Alabamians based on a moral judgment is wrong and certainly doesn’t follow the science,” Ms. Hiers added.
Direct-acting antiviral (DAA) therapy can cure up to 99% of people living with HCV.
The complaint against Alabama Medicaid builds on CHLPI’s successful policy advocacy and litigation campaigns to expand access to DAA therapy in state Medicaid programs across the country.
Since 2017, 19 states have removed treatment restrictions that were based on drug or alcohol use. In other states, however, “severe, illegal sobriety restrictions remain,” according to CHLPI.
Alabama, Mississippi, Arkansas, South Carolina, and South Dakota still require Medicaid enrollees with HCV to prove they have not used drugs or alcohol for 6 months before they can receive treatment. Iowa, North Dakota, and West Virginia have a 3-month abstinence requirement.
The American Association for the Study of Liver Diseases and the Infectious Diseases Society of America recommend DAA therapy for all patients with chronic HCV infection, regardless of drug or alcohol use.
CHLPI intends to expand this “enforcement campaign” to all states where sobriety restrictions persist.
A version of this article first appeared on Medscape.com.