User login
AMA joins in lawsuit accusing Cigna of underpaying physicians
, the nation’s largest third-party network.
The American Medical Association, the Medical Society of New Jersey, and the Washington State Medical Association on Sept. 12 entered into a legal battle between the giant insurers and patients.
At issue are claims involving the firm MultiPlan as an intermediary. Cigna had not responded to this news organization at press time following multiple requests for comment.
According to the legal complaint AMA and the two state medical societies joined, MultiPlan has contracts with more than 1.2 million clinicians. Under these agreements, medical professionals agree to accept a set percentage of billed charges as payment in full, while not holding patients responsible for the difference between the original billed charges and the discounted rate.
But the complaint alleges that MultiPlan failed to stick with that bargain. In a statement, AMA President Jack Resneck Jr, MD, said the physician groups joined the legal action “to shed light on Cigna’s misconduct and create remedies so that patients and physicians can look forward to getting what they are promised.”
Dr. Resneck said Cigna’s approach “is riddled with conflicts of interest and manipulations that routinely shortchanged payments to MultiPlan Network physicians and interfered with the patient-physician relationship by ignoring the MultiPlan contracts and making incorrect statements to patients about their liability for the unpaid portion of the billed charges.”
According to the complaint, Cigna used a company called Zelis to “unilaterally re-price’’ claims at an amount far lower than that called for by the MultiPlan Contract. The three cases cited in the lawsuit stem from a 2017 spine surgery in Washington and 2018 orthopedic and 2020 breast reconstruction surgeries in New Jersey. The decisions to ignore the previous agreements and cut the reimbursement led the physicians involved to eventually bill patients for some of the money in dispute, according to the complaint.
“The providers were left in a very untenable situation,” D. Brian Hufford, an attorney involved in the case, told this news organization. “Their only choice was to go after the insurance company and sue them or they have to go after the patient. That interferes with the patient-doctor relationship.”
Mr. Hufford, who’s a partner at law firm Zuckerman Spaeder, said that these kinds of cases fall beyond the protections provided by the No Surprises Act. Plaintiffs in these cases were enrolled in what are called self-insured plans provided by employers, through which they were supposed to be allowed to seek out-of-network care.
Highly concerning are the messages that insurers send to patients through explanation of benefits (EOB) statements, Mr. Hufford said. Thus in this case against Cigna, physicians and patients have the “same interest in trying to make sure the insurance companies are paying the appropriate amounts for these services,” he said.
Cigna “is telling the patients that the provider has accepted something, and that the patient does not have to worry about paying for that, when in fact that’s not true,” Mr. Hufford said. “That goes beyond merely not complying with the plan documents, but also engaging in conduct that we believe was inappropriate.”
A version of this article first appeared on Medscape.com.
, the nation’s largest third-party network.
The American Medical Association, the Medical Society of New Jersey, and the Washington State Medical Association on Sept. 12 entered into a legal battle between the giant insurers and patients.
At issue are claims involving the firm MultiPlan as an intermediary. Cigna had not responded to this news organization at press time following multiple requests for comment.
According to the legal complaint AMA and the two state medical societies joined, MultiPlan has contracts with more than 1.2 million clinicians. Under these agreements, medical professionals agree to accept a set percentage of billed charges as payment in full, while not holding patients responsible for the difference between the original billed charges and the discounted rate.
But the complaint alleges that MultiPlan failed to stick with that bargain. In a statement, AMA President Jack Resneck Jr, MD, said the physician groups joined the legal action “to shed light on Cigna’s misconduct and create remedies so that patients and physicians can look forward to getting what they are promised.”
Dr. Resneck said Cigna’s approach “is riddled with conflicts of interest and manipulations that routinely shortchanged payments to MultiPlan Network physicians and interfered with the patient-physician relationship by ignoring the MultiPlan contracts and making incorrect statements to patients about their liability for the unpaid portion of the billed charges.”
According to the complaint, Cigna used a company called Zelis to “unilaterally re-price’’ claims at an amount far lower than that called for by the MultiPlan Contract. The three cases cited in the lawsuit stem from a 2017 spine surgery in Washington and 2018 orthopedic and 2020 breast reconstruction surgeries in New Jersey. The decisions to ignore the previous agreements and cut the reimbursement led the physicians involved to eventually bill patients for some of the money in dispute, according to the complaint.
“The providers were left in a very untenable situation,” D. Brian Hufford, an attorney involved in the case, told this news organization. “Their only choice was to go after the insurance company and sue them or they have to go after the patient. That interferes with the patient-doctor relationship.”
Mr. Hufford, who’s a partner at law firm Zuckerman Spaeder, said that these kinds of cases fall beyond the protections provided by the No Surprises Act. Plaintiffs in these cases were enrolled in what are called self-insured plans provided by employers, through which they were supposed to be allowed to seek out-of-network care.
Highly concerning are the messages that insurers send to patients through explanation of benefits (EOB) statements, Mr. Hufford said. Thus in this case against Cigna, physicians and patients have the “same interest in trying to make sure the insurance companies are paying the appropriate amounts for these services,” he said.
Cigna “is telling the patients that the provider has accepted something, and that the patient does not have to worry about paying for that, when in fact that’s not true,” Mr. Hufford said. “That goes beyond merely not complying with the plan documents, but also engaging in conduct that we believe was inappropriate.”
A version of this article first appeared on Medscape.com.
, the nation’s largest third-party network.
The American Medical Association, the Medical Society of New Jersey, and the Washington State Medical Association on Sept. 12 entered into a legal battle between the giant insurers and patients.
At issue are claims involving the firm MultiPlan as an intermediary. Cigna had not responded to this news organization at press time following multiple requests for comment.
According to the legal complaint AMA and the two state medical societies joined, MultiPlan has contracts with more than 1.2 million clinicians. Under these agreements, medical professionals agree to accept a set percentage of billed charges as payment in full, while not holding patients responsible for the difference between the original billed charges and the discounted rate.
But the complaint alleges that MultiPlan failed to stick with that bargain. In a statement, AMA President Jack Resneck Jr, MD, said the physician groups joined the legal action “to shed light on Cigna’s misconduct and create remedies so that patients and physicians can look forward to getting what they are promised.”
Dr. Resneck said Cigna’s approach “is riddled with conflicts of interest and manipulations that routinely shortchanged payments to MultiPlan Network physicians and interfered with the patient-physician relationship by ignoring the MultiPlan contracts and making incorrect statements to patients about their liability for the unpaid portion of the billed charges.”
According to the complaint, Cigna used a company called Zelis to “unilaterally re-price’’ claims at an amount far lower than that called for by the MultiPlan Contract. The three cases cited in the lawsuit stem from a 2017 spine surgery in Washington and 2018 orthopedic and 2020 breast reconstruction surgeries in New Jersey. The decisions to ignore the previous agreements and cut the reimbursement led the physicians involved to eventually bill patients for some of the money in dispute, according to the complaint.
“The providers were left in a very untenable situation,” D. Brian Hufford, an attorney involved in the case, told this news organization. “Their only choice was to go after the insurance company and sue them or they have to go after the patient. That interferes with the patient-doctor relationship.”
Mr. Hufford, who’s a partner at law firm Zuckerman Spaeder, said that these kinds of cases fall beyond the protections provided by the No Surprises Act. Plaintiffs in these cases were enrolled in what are called self-insured plans provided by employers, through which they were supposed to be allowed to seek out-of-network care.
Highly concerning are the messages that insurers send to patients through explanation of benefits (EOB) statements, Mr. Hufford said. Thus in this case against Cigna, physicians and patients have the “same interest in trying to make sure the insurance companies are paying the appropriate amounts for these services,” he said.
Cigna “is telling the patients that the provider has accepted something, and that the patient does not have to worry about paying for that, when in fact that’s not true,” Mr. Hufford said. “That goes beyond merely not complying with the plan documents, but also engaging in conduct that we believe was inappropriate.”
A version of this article first appeared on Medscape.com.
‘Smoking gun–level’ evidence found linking air pollution with lung cancer
PARIS – Air pollution has been recognized as a risk factor for lung cancer for about 2 decades, and already present in normal lung cells to cause cancer.
Think of it as “smoking gun–level” evidence that may explain why many nonsmokers still develop non–small cell lung cancer, said Charles Swanton, PhD, from the Francis Crick Institute and Cancer Research UK Chief Clinician, London.
“What this work shows is that air pollution is directly causing lung cancer but through a slightly unexpected pathway,” he said at a briefing prior to his presentation of the data in a presidential symposium held earlier this month in Paris at the European Society for Medical Oncology Congress 2022.
Importantly, he and his team also propose a mechanism for blocking the effects of air pollution with monoclonal antibodies directed against the inflammatory cytokine interleukein-1 beta.
Carcinogenesis explored
Lung cancer in never-smokers has a low mutational burden, with about 5- to 10-fold fewer mutations in a nonsmoker, compared with an ever smoker or current smoker, Dr. Swanton noted.
“The other thing to say about never-smokers is that they don’t have a clear environmental carcinogenic signature. So how do you square the circle? You’ve got the problem that you know that air pollution is associated with lung cancer – we don’t know if it causes it – but we also see that we’ve got no DNA mutations due to an environmental carcinogen,” he said during his symposium presentation.
The traditional model proposed to explain how carcinogens cause cancer holds that exposure to a carcinogen causes DNA mutations that lead to clonal expansion and tumor growth.
“But there are some major problems with this model,” Dr. Swanton said.
For example, normal skin contains a “patchwork of mutant clones,” but skin cancer is still uncommon, he said, and in studies in mice, 17 of 20 environmental carcinogens did not induce DNA mutations. He also noted that a common melanoma driver mutation, BRAF V600E, is not induced by exposure to a ultraviolet light.
“Any explanation for never-smoking lung cancer would have to fulfill three criteria: one, you have to explain why geographic variation exists; two, you have to prove causation; and three, you have to explain how cancers can be initiated without directly causing DNA mutations,” he said.
Normal lung tissues in nonsmoking adults can harbor pre-existing mutations, with the number of mutations increasing likely as a consequence of aging. In fact, more than 50% of normal lung biopsy tissues have been shown to harbor driver KRAS and/or EGFR mutations, Dr. Swanton said.
“In our research, these mutations alone only weakly potentiated cancer in laboratory models. However, when lung cells with these mutations were exposed to air pollutants, we saw more cancers and these occurred more quickly than when lung cells with these mutations were not exposed to pollutants, suggesting that air pollution promotes the initiation of lung cancer in cells harboring driver gene mutations. The next step is to discover why some lung cells with mutations become cancerous when exposed to pollutants while others don’t,” he said.
Geographical exposures
Looking at data on 447,932 participants in the UK Biobank, the investigators found that increasing exposure to ambient air particles smaller than 2.5 mcm (PM2.5) was significantly associated with seven cancer types, including lung cancer. They also saw an association between PM2.5 exposure levels and EGFR-mutated lung cancer incidence in the United Kingdom, South Korea, and Taiwan.
And crucially, as Dr. Swanton and associates showed in mouse models, exposure of lung cells bearing somatic EGFR and KRAS mutations to PM2.5 causes recruitment of macrophages that in turn secrete IL-1B, resulting in a transdifferentiation of EGFR-mutated cells into a cancer stem cell state, and tumor formation.
Importantly, pollution-induced tumor formation can be blocked by antibodies directed against IL-1B, Dr. Swanton said.
He pointed to a 2017 study in The Lancet suggesting that anti-inflammatory therapy with the anti–IL-1 antibody canakinumab (Ilaris) could reduce incident lung cancer and lung cancer deaths.
‘Elegant first demonstration’
“This is a very meaningful demonstration, from epidemiological data to preclinical models of the role of PM2.5 air pollutants in the promotion of lung cancer, and it provides us with very important insights into the mechanism through which nonsmokers can get lung cancer,” commented Suzette Delaloge, MD, from the cancer interception program at Institut Goustave Roussy in Villejuif, France, the invited discussant.
“But beyond that, it also has a great impact on our vision of carcinogenesis, with this very elegant first demonstration of the alternative nonmutagenic, carcinogenetic promotion hypothesis for fine particulate matter,” she said.
Questions still to be answered include whether PM2.5 pollutants could also be mutagenic, is the oncogenic pathway ubiquitous in tissue, which components of PM2.5 might drive the effect, how long of an exposure is required to promote lung cancer, and why and how persons without cancer develop specific driver mutations such as EGFR, she said.
“This research is intriguing and exciting as it means that we can ask whether, in the future, it will be possible to use lung scans to look for precancerous lesions in the lungs and try to reverse them with medicines such as interleukin-1B inhibitors,” said Tony Mok, MD, a lung cancer specialist at the Chinese University of Hong Kong, who was not involved in the study.
“We don’t yet know whether it will be possible to use highly sensitive EGFR profiling on blood or other samples to find nonsmokers who are predisposed to lung cancer and may benefit from lung scanning, so discussions are still very speculative,” he said in a statement.
The study was supported by Cancer Research UK, the Lung Cancer Research Foundations, Rosetrees Trust, the Mark Foundation for Cancer Research and the Ruth Strauss Foundation. Dr. Swanton disclosed grants/research support, honoraria, and stock ownership with multiple entities. Dr. Delaloge disclosed institutional financing and research funding from multiple companies. Dr. Mok disclosed stock ownership and honoraria with multiple companies.
PARIS – Air pollution has been recognized as a risk factor for lung cancer for about 2 decades, and already present in normal lung cells to cause cancer.
Think of it as “smoking gun–level” evidence that may explain why many nonsmokers still develop non–small cell lung cancer, said Charles Swanton, PhD, from the Francis Crick Institute and Cancer Research UK Chief Clinician, London.
“What this work shows is that air pollution is directly causing lung cancer but through a slightly unexpected pathway,” he said at a briefing prior to his presentation of the data in a presidential symposium held earlier this month in Paris at the European Society for Medical Oncology Congress 2022.
Importantly, he and his team also propose a mechanism for blocking the effects of air pollution with monoclonal antibodies directed against the inflammatory cytokine interleukein-1 beta.
Carcinogenesis explored
Lung cancer in never-smokers has a low mutational burden, with about 5- to 10-fold fewer mutations in a nonsmoker, compared with an ever smoker or current smoker, Dr. Swanton noted.
“The other thing to say about never-smokers is that they don’t have a clear environmental carcinogenic signature. So how do you square the circle? You’ve got the problem that you know that air pollution is associated with lung cancer – we don’t know if it causes it – but we also see that we’ve got no DNA mutations due to an environmental carcinogen,” he said during his symposium presentation.
The traditional model proposed to explain how carcinogens cause cancer holds that exposure to a carcinogen causes DNA mutations that lead to clonal expansion and tumor growth.
“But there are some major problems with this model,” Dr. Swanton said.
For example, normal skin contains a “patchwork of mutant clones,” but skin cancer is still uncommon, he said, and in studies in mice, 17 of 20 environmental carcinogens did not induce DNA mutations. He also noted that a common melanoma driver mutation, BRAF V600E, is not induced by exposure to a ultraviolet light.
“Any explanation for never-smoking lung cancer would have to fulfill three criteria: one, you have to explain why geographic variation exists; two, you have to prove causation; and three, you have to explain how cancers can be initiated without directly causing DNA mutations,” he said.
Normal lung tissues in nonsmoking adults can harbor pre-existing mutations, with the number of mutations increasing likely as a consequence of aging. In fact, more than 50% of normal lung biopsy tissues have been shown to harbor driver KRAS and/or EGFR mutations, Dr. Swanton said.
“In our research, these mutations alone only weakly potentiated cancer in laboratory models. However, when lung cells with these mutations were exposed to air pollutants, we saw more cancers and these occurred more quickly than when lung cells with these mutations were not exposed to pollutants, suggesting that air pollution promotes the initiation of lung cancer in cells harboring driver gene mutations. The next step is to discover why some lung cells with mutations become cancerous when exposed to pollutants while others don’t,” he said.
Geographical exposures
Looking at data on 447,932 participants in the UK Biobank, the investigators found that increasing exposure to ambient air particles smaller than 2.5 mcm (PM2.5) was significantly associated with seven cancer types, including lung cancer. They also saw an association between PM2.5 exposure levels and EGFR-mutated lung cancer incidence in the United Kingdom, South Korea, and Taiwan.
And crucially, as Dr. Swanton and associates showed in mouse models, exposure of lung cells bearing somatic EGFR and KRAS mutations to PM2.5 causes recruitment of macrophages that in turn secrete IL-1B, resulting in a transdifferentiation of EGFR-mutated cells into a cancer stem cell state, and tumor formation.
Importantly, pollution-induced tumor formation can be blocked by antibodies directed against IL-1B, Dr. Swanton said.
He pointed to a 2017 study in The Lancet suggesting that anti-inflammatory therapy with the anti–IL-1 antibody canakinumab (Ilaris) could reduce incident lung cancer and lung cancer deaths.
‘Elegant first demonstration’
“This is a very meaningful demonstration, from epidemiological data to preclinical models of the role of PM2.5 air pollutants in the promotion of lung cancer, and it provides us with very important insights into the mechanism through which nonsmokers can get lung cancer,” commented Suzette Delaloge, MD, from the cancer interception program at Institut Goustave Roussy in Villejuif, France, the invited discussant.
“But beyond that, it also has a great impact on our vision of carcinogenesis, with this very elegant first demonstration of the alternative nonmutagenic, carcinogenetic promotion hypothesis for fine particulate matter,” she said.
Questions still to be answered include whether PM2.5 pollutants could also be mutagenic, is the oncogenic pathway ubiquitous in tissue, which components of PM2.5 might drive the effect, how long of an exposure is required to promote lung cancer, and why and how persons without cancer develop specific driver mutations such as EGFR, she said.
“This research is intriguing and exciting as it means that we can ask whether, in the future, it will be possible to use lung scans to look for precancerous lesions in the lungs and try to reverse them with medicines such as interleukin-1B inhibitors,” said Tony Mok, MD, a lung cancer specialist at the Chinese University of Hong Kong, who was not involved in the study.
“We don’t yet know whether it will be possible to use highly sensitive EGFR profiling on blood or other samples to find nonsmokers who are predisposed to lung cancer and may benefit from lung scanning, so discussions are still very speculative,” he said in a statement.
The study was supported by Cancer Research UK, the Lung Cancer Research Foundations, Rosetrees Trust, the Mark Foundation for Cancer Research and the Ruth Strauss Foundation. Dr. Swanton disclosed grants/research support, honoraria, and stock ownership with multiple entities. Dr. Delaloge disclosed institutional financing and research funding from multiple companies. Dr. Mok disclosed stock ownership and honoraria with multiple companies.
PARIS – Air pollution has been recognized as a risk factor for lung cancer for about 2 decades, and already present in normal lung cells to cause cancer.
Think of it as “smoking gun–level” evidence that may explain why many nonsmokers still develop non–small cell lung cancer, said Charles Swanton, PhD, from the Francis Crick Institute and Cancer Research UK Chief Clinician, London.
“What this work shows is that air pollution is directly causing lung cancer but through a slightly unexpected pathway,” he said at a briefing prior to his presentation of the data in a presidential symposium held earlier this month in Paris at the European Society for Medical Oncology Congress 2022.
Importantly, he and his team also propose a mechanism for blocking the effects of air pollution with monoclonal antibodies directed against the inflammatory cytokine interleukein-1 beta.
Carcinogenesis explored
Lung cancer in never-smokers has a low mutational burden, with about 5- to 10-fold fewer mutations in a nonsmoker, compared with an ever smoker or current smoker, Dr. Swanton noted.
“The other thing to say about never-smokers is that they don’t have a clear environmental carcinogenic signature. So how do you square the circle? You’ve got the problem that you know that air pollution is associated with lung cancer – we don’t know if it causes it – but we also see that we’ve got no DNA mutations due to an environmental carcinogen,” he said during his symposium presentation.
The traditional model proposed to explain how carcinogens cause cancer holds that exposure to a carcinogen causes DNA mutations that lead to clonal expansion and tumor growth.
“But there are some major problems with this model,” Dr. Swanton said.
For example, normal skin contains a “patchwork of mutant clones,” but skin cancer is still uncommon, he said, and in studies in mice, 17 of 20 environmental carcinogens did not induce DNA mutations. He also noted that a common melanoma driver mutation, BRAF V600E, is not induced by exposure to a ultraviolet light.
“Any explanation for never-smoking lung cancer would have to fulfill three criteria: one, you have to explain why geographic variation exists; two, you have to prove causation; and three, you have to explain how cancers can be initiated without directly causing DNA mutations,” he said.
Normal lung tissues in nonsmoking adults can harbor pre-existing mutations, with the number of mutations increasing likely as a consequence of aging. In fact, more than 50% of normal lung biopsy tissues have been shown to harbor driver KRAS and/or EGFR mutations, Dr. Swanton said.
“In our research, these mutations alone only weakly potentiated cancer in laboratory models. However, when lung cells with these mutations were exposed to air pollutants, we saw more cancers and these occurred more quickly than when lung cells with these mutations were not exposed to pollutants, suggesting that air pollution promotes the initiation of lung cancer in cells harboring driver gene mutations. The next step is to discover why some lung cells with mutations become cancerous when exposed to pollutants while others don’t,” he said.
Geographical exposures
Looking at data on 447,932 participants in the UK Biobank, the investigators found that increasing exposure to ambient air particles smaller than 2.5 mcm (PM2.5) was significantly associated with seven cancer types, including lung cancer. They also saw an association between PM2.5 exposure levels and EGFR-mutated lung cancer incidence in the United Kingdom, South Korea, and Taiwan.
And crucially, as Dr. Swanton and associates showed in mouse models, exposure of lung cells bearing somatic EGFR and KRAS mutations to PM2.5 causes recruitment of macrophages that in turn secrete IL-1B, resulting in a transdifferentiation of EGFR-mutated cells into a cancer stem cell state, and tumor formation.
Importantly, pollution-induced tumor formation can be blocked by antibodies directed against IL-1B, Dr. Swanton said.
He pointed to a 2017 study in The Lancet suggesting that anti-inflammatory therapy with the anti–IL-1 antibody canakinumab (Ilaris) could reduce incident lung cancer and lung cancer deaths.
‘Elegant first demonstration’
“This is a very meaningful demonstration, from epidemiological data to preclinical models of the role of PM2.5 air pollutants in the promotion of lung cancer, and it provides us with very important insights into the mechanism through which nonsmokers can get lung cancer,” commented Suzette Delaloge, MD, from the cancer interception program at Institut Goustave Roussy in Villejuif, France, the invited discussant.
“But beyond that, it also has a great impact on our vision of carcinogenesis, with this very elegant first demonstration of the alternative nonmutagenic, carcinogenetic promotion hypothesis for fine particulate matter,” she said.
Questions still to be answered include whether PM2.5 pollutants could also be mutagenic, is the oncogenic pathway ubiquitous in tissue, which components of PM2.5 might drive the effect, how long of an exposure is required to promote lung cancer, and why and how persons without cancer develop specific driver mutations such as EGFR, she said.
“This research is intriguing and exciting as it means that we can ask whether, in the future, it will be possible to use lung scans to look for precancerous lesions in the lungs and try to reverse them with medicines such as interleukin-1B inhibitors,” said Tony Mok, MD, a lung cancer specialist at the Chinese University of Hong Kong, who was not involved in the study.
“We don’t yet know whether it will be possible to use highly sensitive EGFR profiling on blood or other samples to find nonsmokers who are predisposed to lung cancer and may benefit from lung scanning, so discussions are still very speculative,” he said in a statement.
The study was supported by Cancer Research UK, the Lung Cancer Research Foundations, Rosetrees Trust, the Mark Foundation for Cancer Research and the Ruth Strauss Foundation. Dr. Swanton disclosed grants/research support, honoraria, and stock ownership with multiple entities. Dr. Delaloge disclosed institutional financing and research funding from multiple companies. Dr. Mok disclosed stock ownership and honoraria with multiple companies.
AT ESMO CONGRESS 2022
Optimizing Narrowband UVB Phototherapy: Is It More Challenging for Your Older Patients?
Even with recent pharmacologic treatment advances, narrowband UVB (NB-UVB) phototherapy remains a versatile, safe, and efficacious adjunctive or exclusive treatment for multiple dermatologic conditions, including psoriasis and atopic dermatitis.
In a prior study, Matthews et al13 reported that 96% (50/52) of patients older than 65 years achieved medium to high levels of clearance with NB-UVB phototherapy. Nonetheless, 2 other findings in this study related to the number of treatments required to achieve clearance (ie, clearance rates) and erythema rates prompted further investigation. The first finding was higher-than-expected clearance rates. Older adults had a clearance rate with a mean of 33 treatments compared to prior studies featuring mean clearance rates of 20 to 28 treatments.7,8,14-16 This finding resembled a study in the United Kingdom17 with a median clearance rate in older adults of 30 treatments. In contrast, the median clearance rate from a study in Turkey18 was 42 treatments in older adults. We hypothesized that more photosensitizing medications used in older vs younger adults prompted more dose adjustments with NB-UVB phototherapy to avoid burning (ie, erythema) at baseline and throughout the treatment course. These dose adjustments may have increased the overall clearance rates. If true, we predicted that younger adults treated with the same protocol would have cleared more quickly, either because of age-related differences or because they likely had fewer comorbidities and therefore fewer medications.
The second finding from Matthews et al13 that warranted further investigation was a higher erythema rate compared to the older adult study from the United Kingdom.17 We hypothesized that potentially greater use of photosensitizing medications in the United States could explain the higher erythema rates. Although medication-induced photosensitivity is less likely with NB-UVB phototherapy than with UVA, certain medications can cause UVB photosensitivity, including thiazides, quinidine, calcium channel antagonists, phenothiazines, and nonsteroidal anti-inflammatory drugs.8,19,20 Therefore, photosensitizing medication use either at baseline or during a course of NB-UVB phototherapy could increase the risk for erythema. Age-related skin changes also have been considered as a
This retrospective study aimed to determine if NB-UVB phototherapy is equally effective in both older and younger adults treated with the same protocol; to examine the association between the use of photosensitizing medications and clearance rates in both older and younger adults; and to examine the association between the use of photosensitizing medications and erythema rates in older vs younger adults.
Methods
Study Design and Patients—This retrospective cohort study used billing records to identify patients who received NB-UVB phototherapy at 3 different clinical sites within a large US health care system in Washington (Group Health Cooperative, now Kaiser Permanente Washington), serving more than 600,000 patients between January 1, 2012, and December 31, 2016. The institutional review board of Kaiser Permanente Washington Health Research Institute approved this study (IRB 1498087-4). Younger adults were classified as those 64 years or younger and older adults as those 65 years and older at the start of their phototherapy regimen. A power analysis determined that the optimal sample size for this study was 250 patients.
Individuals were excluded if they had fewer than 6 phototherapy treatments; a diagnosis of vitiligo, photosensitivity dermatitis, morphea, or pityriasis rubra pilaris; and/or treatment of the hands or feet only.
Phototherapy Protocol—Using a 48-lamp NB-UVB unit, trained phototherapy nurses provided all treatments following standardized treatment protocols13 based on previously published phototherapy guidelines.24 Nurses determined each patient’s disease clearance level using a 3-point clearance scale (high, medium, low).13 Each patient’s starting dose was determined based on the estimated MED for their skin phototype.
Statistical Analysis—Data were analyzed using Stata statistical software (StataCorp LLC). Univariate analyses were used to examine the data and identify outliers, bad values, and missing data, as well as to calculate descriptive statistics. Pearson χ2 and Fisher exact statistics were used to calculate differences in categorical variables. Linear multivariate regression models and logistic multivariate models were used to examine statistical relationships between variables. Statistical significance was defined as P≤.05.
Results
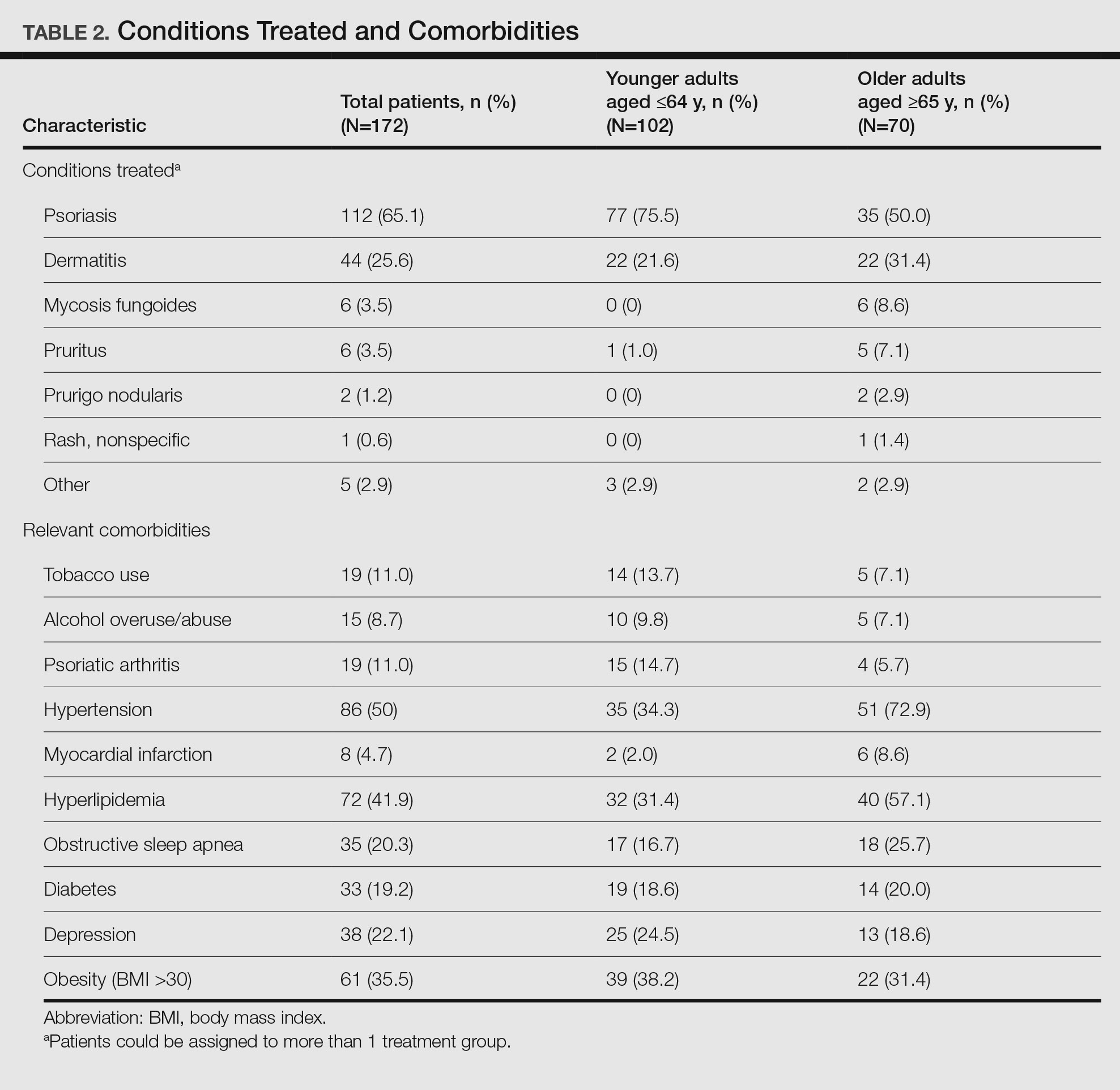
Patient Characteristics—Medical records were reviewed for 172 patients who received phototherapy between 2012 and 2016. Patients ranged in age from 23 to 91 years, with 102 patients 64 years and younger and 70 patients 65 years and older. Tables 1 and 2 outline the patient characteristics and conditions treated.

Phototherapy Effectiveness—
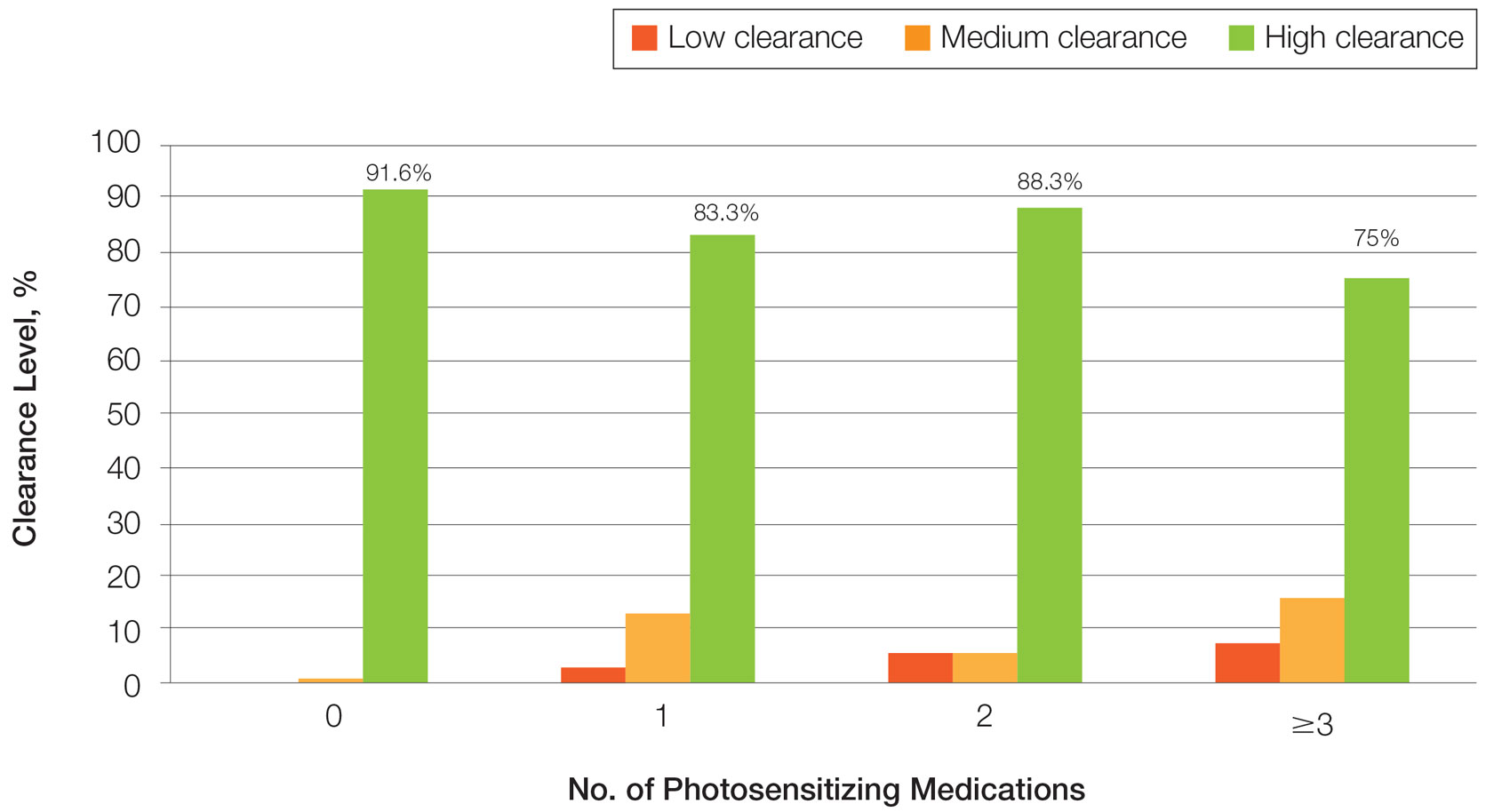
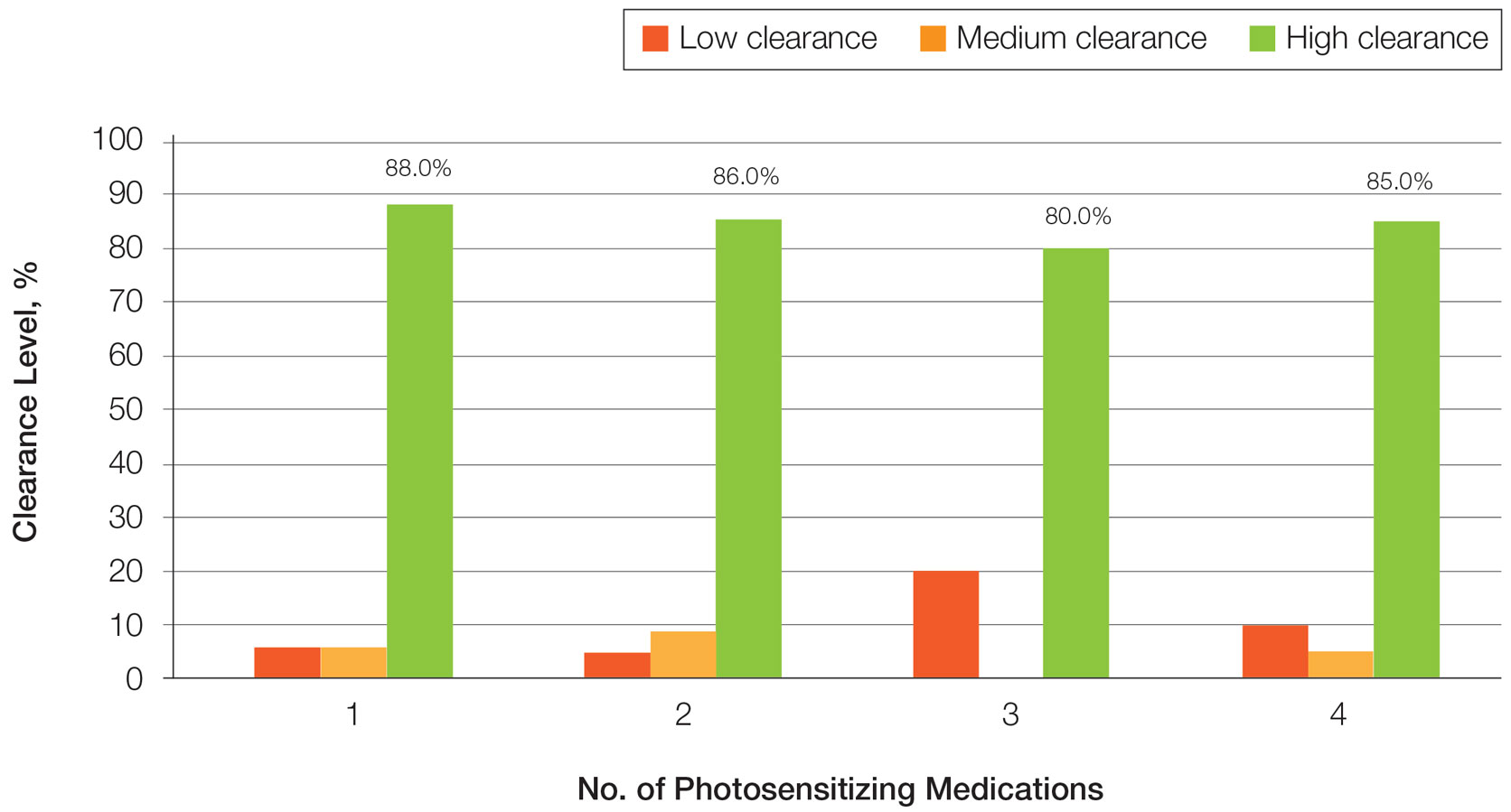
Photosensitizing Medications, Clearance Levels, and Clearance Rates—
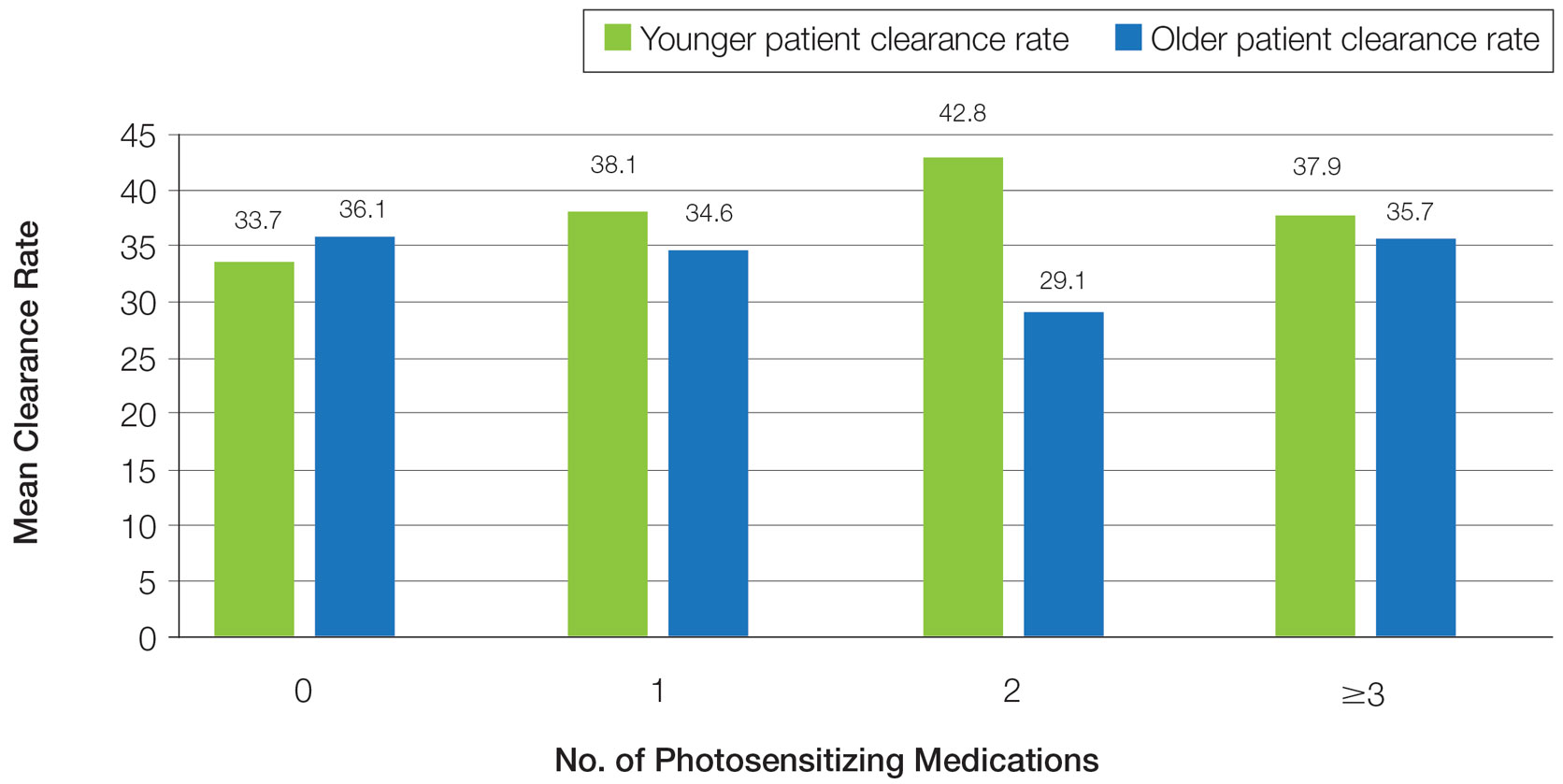
Frequency of Treatments and Clearance Rates—Older adults more consistently completed the recommended frequency of treatments—3 times weekly—compared to younger adults (74.3% vs 58.5%). However, all patients who completed 3 treatments per week required a similar number of treatments to clear (older adults, mean [SD]: 35.7 [21.6]; younger adults, mean [SD]: 34.7 [19.0]; P=.85). Among patients completing 2 or fewer treatments per week, older adults required a mean (SD) of only 31 (9.0) treatments to clear vs 41.5 (21.3) treatments to clear for younger adults, but the difference was not statistically significant (P=.08). However, even those with suboptimal frequency ultimately achieved similar clearance levels.
Photosensitizing Medications and Erythema Rates—
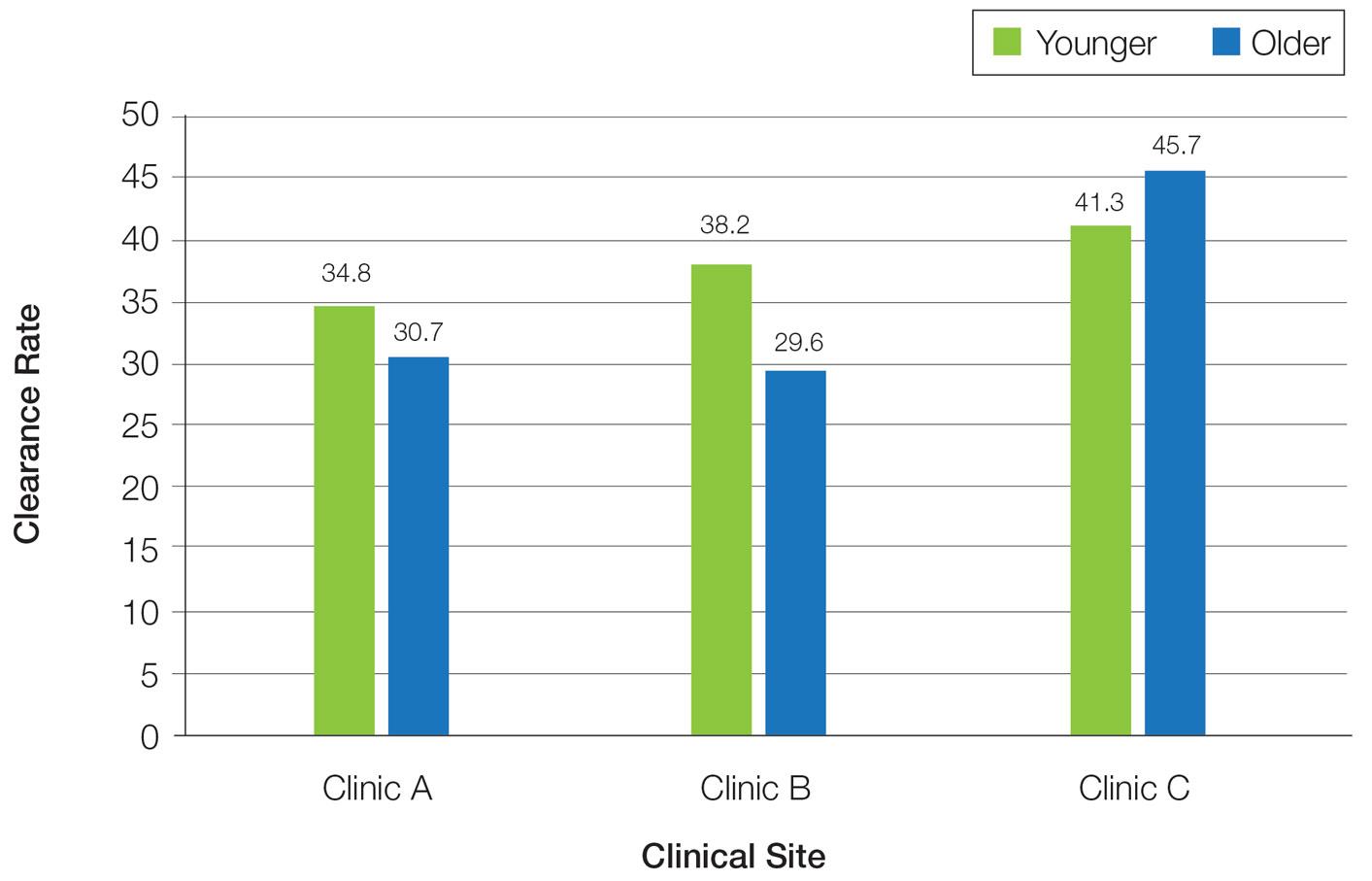
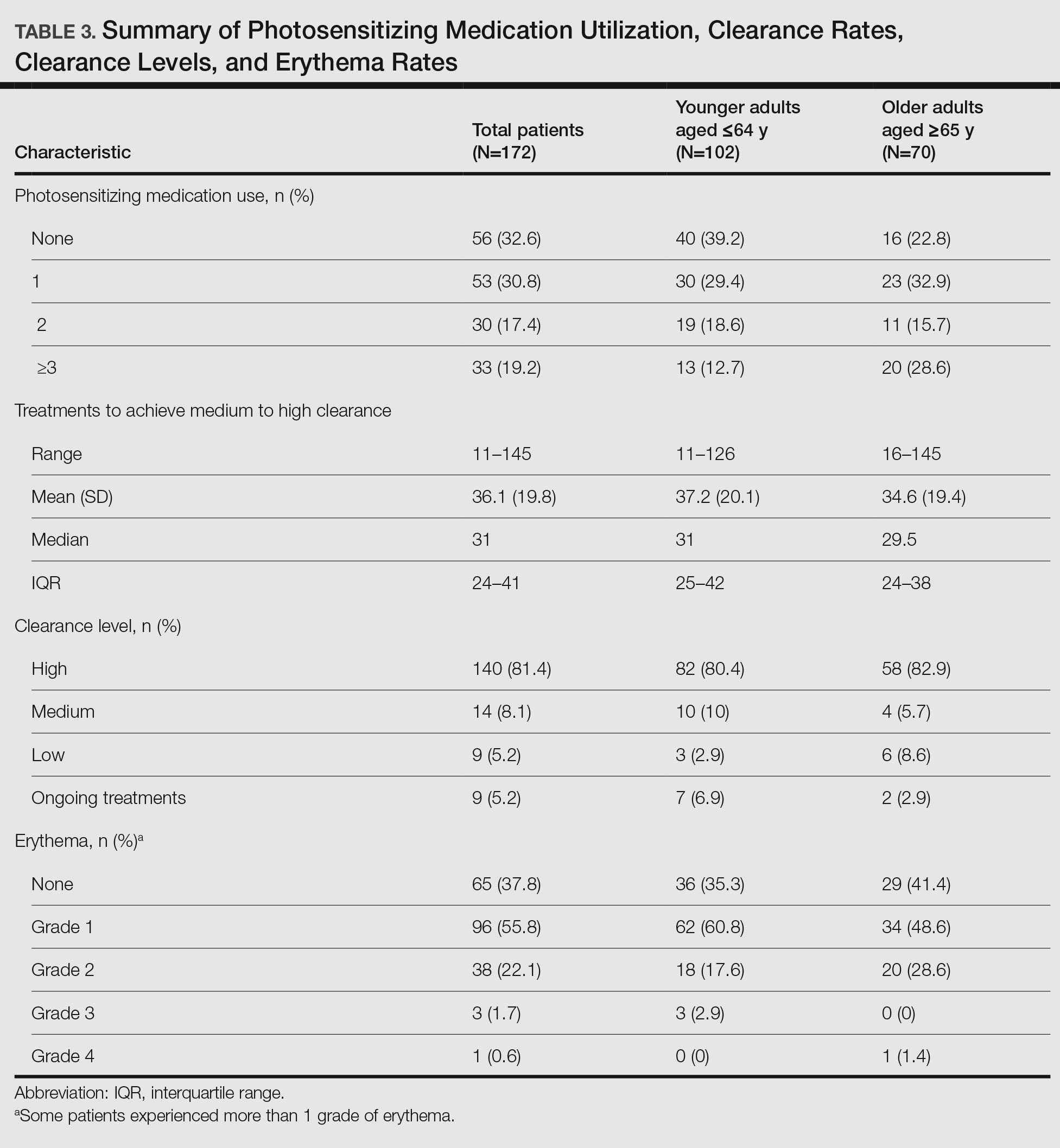
Overall, phototherapy nurses adjusted the starting dose according to the phototype-based protocol an average of 69% of the time for patients on medications with photosensitivity listed as a potential side effect. However, the frequency depended significantly on the clinic (clinic A, 24%; clinic B, 92%; clinic C, 87%)(P≤.001). Nurses across all clinics consistently decreased the treatment dose when patients reported starting new photosensitizing medications. Patients with adjusted starting doses had slightly but not significantly higher clearance rates compared to those without (mean, 37.8 vs 35.5; t(104)=0.58; P=.56).
Comment
Impact of Photosensitizing Medications on Clearance—Photosensitizing medications and treatment frequency were 2 factors that might explain the slower clearance rates in younger adults. In this study, both groups of patients used similar numbers of photosensitizing medications, but more older adults were taking 3 or more medications (Table 3). We found no statistically significant relationship between taking photosensitizing medications and either the clearance rates or the level of clearance achieved in either age group.
Impact of Treatment Frequency—Weekly treatment frequency also was examined. One prior study demonstrated that treatments 3 times weekly led to a faster clearance time and higher clearance levels compared with twice-weekly treatment.7 When patients completed treatments twice weekly, it took an average of 1.5 times more days to clear, which impacted cost and clinical resource availability. The patients ranged in age from 17 to 80 years, but outcomes in older patients were not described separately.7 Interestingly, our study seemed to find a difference between age groups when the impact of treatment frequency was examined. Older adults completed nearly 4 fewer mean treatments to clear when treating less often, with more than 80% achieving high levels of clearance, whereas the younger adults required almost 7 more treatments to clear when they came in less frequently, with approximately 80% achieving a high level of clearance. As a result, our study found that in both age groups, slowing the treatment frequency extended the treatment time to clearance—more for the younger adults than the older adults—but did not significantly change the percentage of individuals reaching full clearance in either group.
Erythema Rates—There was no association between photosensitizing medications and erythema rates except when patients were taking at least 3 medications. Most medications that listed photosensitivity as a possible side effect did not specify their relevant range of UV radiation; therefore, all such medications were examined during this analysis. Prior research has shown UVB range photosensitizing medications include thiazides, quinidine, calcium channel antagonists, phenothiazines, and nonsteroidal anti-inflammatory drugs.19 A sensitivity analysis that focused only on these medications found no association between them and any particular grade of erythema. However, patients taking 3 or more of any medications listing photosensitivity as a side effect had an increased risk for grade 2 erythema.
Erythema rates in this study were consistent with a 2013 systematic review that reported 57% of patients with asymptomatic grade 1 erythema.25 In the 2 other comparative older adult studies, erythema rates varied widely: 35% in a study from Turkey18compared to only1.89% in a study from the United Kingdom.17
The starting dose for NB-UVB may drive erythema rates. The current study’s protocols were based on an estimated MED that is subjectively determined by the dermatology provider’s assessment of the patient’s skin sensitivity via examination and questions to the patient about their response to environmental sun exposure (ie, burning and tanning)26 and is frequently used to determine the starting dose and subsequent dose escalation. Certain medications have been found to increase photosensitivity and erythema,20 which can change an individual’s MED. If photosensitizing medications are started prior to or during a course of NB-UVB without a pretreatment MED, they might increase the risk for erythema. This study did not identify specific erythema-inducing medications but did find that taking 3 or more photosensitizing medications was associated with increased episodes of grade 2 erythema. Similarly, Harrop et al8 found that patients who were taking photosensitizing medications were more likely to have grade 2 or higher erythema, despite baseline MED testing, which is an established safety mechanism to reduce the risk and severity of erythema.14,20,27 The authors of a recent study of older adults in Taiwan specifically recommended MED testing due to the unpredictable influence of polypharmacy on MED calculations in this population.28 Therefore, this study’s use of an estimated MED in older adults may have influenced the starting dose as well as the incidence and severity of erythemic events. Age-related skin changes likely are ruled out as a consideration for mild erythema by the similarity of grade 1 erythema rates in both older and younger adults. Other studies have identified differences between the age groups, where older patients experienced more intense erythema in the late phase of UVB treatments.22,23 This phenomenon could increase the risk for a grade 2 erythema, which may correspond with this study’s findings.
Other potential causes of erythema were ruled out during our study, including erythema related to missed treatments and shielding mishaps. Other factors, however, may impact the level of sensitivity each patient has to phototherapy, including genetics, epigenetics, and cumulative sun damage. With NB-UVB, near-erythemogenic doses are optimal to achieve effective treatments but require a delicate balance to achieve, which may be more problematic for older adults, especially those taking several medications.
Study Limitations—Our study design made it difficult to draw conclusions about rarer dermatologic conditions. Some patients received treatments over years that were not included in the study period. Finally, power calculations suggested that our actual sample size was too small, with approximately one-third of the required sample missing.
Practical Implications—The goals of phototherapy are to achieve a high level of disease clearance with the fewest number of treatments possible and minimal side effects.
The extra staff training and patient monitoring required for MED testing likely is to add value and preserve resources if faster clearance rates could be achieved and may warrant further investigation. Phototherapy centers require standardized treatment protocols, diligent well-trained staff, and program monitoring to ensure consistent care to all patients. This study highlighted the ongoing opportunity for health care organizations to conduct evidence-based practice inquiries to continually optimize care for their patients.
- Fernández-Guarino M, Aboin-Gonzalez S, Barchino L, et al. Treatment of moderate and severe adult chronic atopic dermatitis with narrow-band UVB and the combination of narrow-band UVB/UVA phototherapy. Dermatol Ther. 2016;29:19-23.
- Foerster J, Boswell K, West J, et al. Narrowband UVB treatment is highly effective and causes a strong reduction in the use of steroid and other creams in psoriasis patients in clinical practice. PLoS One. 2017;12:e0181813.
- Gambichler T, Breuckmann F, Boms S, et al. Narrowband UVB phototherapy in skin conditions beyond psoriasis. J Am Acad Dermatol. 2005;52:660-670.
- Ryu HH, Choe YS, Jo S, et al. Remission period in psoriasis after multiple cycles of narrowband ultraviolet B phototherapy. J Dermatol. 2014;41:622-627.
- Schneider LA, Hinrichs R, Scharffetter-Kochanek K. Phototherapy and photochemotherapy. Clin Dermatol. 2008;26:464-476.
- Tintle S, Shemer A, Suárez-Fariñas M, et al. Reversal of atopic dermatitis with narrow-band UVB phototherapy and biomarkers for therapeutic response. J Allergy Clin Immunol. 2011;128:583-593.e581-584.
- Cameron H, Dawe RS, Yule S, et al. A randomized, observer-blinded trial of twice vs. three times weekly narrowband ultraviolet B phototherapy for chronic plaque psoriasis. Br J Dermatol. 2002;147:973-978.
- Harrop G, Dawe RS, Ibbotson S. Are photosensitizing medications associated with increased risk of important erythemal reactions during ultraviolet B phototherapy? Br J Dermatol. 2018;179:1184-1185.
- Torres AE, Lyons AB, Hamzavi IH, et al. Role of phototherapy in the era of biologics. J Am Acad Dermatol. 2021;84:479-485.
- Bukvic´ć Mokos Z, Jovic´ A, Cˇeovic´ R, et al. Therapeutic challenges in the mature patient. Clin Dermatol. 2018;36:128-139.
- Di Lernia V, Goldust M. An overview of the efficacy and safety of systemic treatments for psoriasis in the elderly. Expert Opin Biol Ther. 2018;18:897-903.
- Oliveira C, Torres T. More than skin deep: the systemic nature of atopic dermatitis. Eur J Dermatol. 2019;29:250-258.
- Matthews S, Pike K, Chien A. Phototherapy: safe and effective for challenging skin conditions in older adults. Cutis. 2021;108:E15-E21.
- Rodríguez-Granados MT, Estany-Gestal A, Pousa-Martínez M, et al. Is it useful to calculate minimal erythema dose before narrowband UV-B phototherapy? Actas Dermosifiliogr. 2017;108:852-858.
- Parlak N, Kundakci N, Parlak A, et al. Narrowband ultraviolet B phototherapy starting and incremental dose in patients with psoriasis: comparison of percentage dose and fixed dose protocols. Photodermatol Photoimmunol Photomed. 2015;31:90-97.
- Kleinpenning MM, Smits T, Boezeman J, et al. Narrowband ultraviolet B therapy in psoriasis: randomized double-blind comparison of high-dose and low-dose irradiation regimens. Br J Dermatol. 2009;161:1351-1356.
- Powell JB, Gach JE. Phototherapy in the elderly. Clin Exp Dermatol. 2015;40:605-610.
- Bulur I, Erdogan HK, Aksu AE, et al. The efficacy and safety of phototherapy in geriatric patients: a retrospective study. An Bras Dermatol. 2018;93:33-38.
- Dawe RS, Ibbotson SH. Drug-induced photosensitivity. Dermatol Clin. 2014;32:363-368, ix.
- Cameron H, Dawe RS. Photosensitizing drugs may lower the narrow-band ultraviolet B (TL-01) minimal erythema dose. Br J Dermatol. 2000;142:389-390.
- Elmets CA, Lim HW, Stoff B, et al. Joint American Academy of Dermatology-National Psoriasis Foundation guidelines of care for the management and treatment of psoriasis with phototherapy. J Am Acad Dermatol. 2019;81:775-804.
- Gloor M, Scherotzke A. Age dependence of ultraviolet light-induced erythema following narrow-band UVB exposure. Photodermatol Photoimmunol Photomed. 2002;18:121-126.
- Cox NH, Diffey BL, Farr PM. The relationship between chronological age and the erythemal response to ultraviolet B radiation. Br J Dermatol. 1992;126:315-319.
- Morrison W. Phototherapy and Photochemotherapy for Skin Disease. 2nd ed. Informa Healthcare; 2005.
- Almutawa F, Alnomair N, Wang Y, et al. Systematic review of UV-based therapy for psoriasis. Am J Clin Dermatol. 2013;14:87-109.
- Trakatelli M, Bylaite-Bucinskiene M, Correia O, et al. Clinical assessment of skin phototypes: watch your words! Eur J Dermatol. 2017;27:615-619.
- Kwon IH, Kwon HH, Na SJ, et al. Could colorimetric method replace the individual minimal erythemal dose (MED) measurements in determining the initial dose of narrow-band UVB treatment for psoriasis patients with skin phototype III-V? J Eur Acad Dermatol Venereol. 2013;27:494-498.
- Chen WA, Chang CM. The minimal erythema dose of narrowband ultraviolet B in elderly Taiwanese [published online September 1, 2021]. Photodermatol Photoimmunol Photomed. doi:10.1111/phpp.12730
Even with recent pharmacologic treatment advances, narrowband UVB (NB-UVB) phototherapy remains a versatile, safe, and efficacious adjunctive or exclusive treatment for multiple dermatologic conditions, including psoriasis and atopic dermatitis.
In a prior study, Matthews et al13 reported that 96% (50/52) of patients older than 65 years achieved medium to high levels of clearance with NB-UVB phototherapy. Nonetheless, 2 other findings in this study related to the number of treatments required to achieve clearance (ie, clearance rates) and erythema rates prompted further investigation. The first finding was higher-than-expected clearance rates. Older adults had a clearance rate with a mean of 33 treatments compared to prior studies featuring mean clearance rates of 20 to 28 treatments.7,8,14-16 This finding resembled a study in the United Kingdom17 with a median clearance rate in older adults of 30 treatments. In contrast, the median clearance rate from a study in Turkey18 was 42 treatments in older adults. We hypothesized that more photosensitizing medications used in older vs younger adults prompted more dose adjustments with NB-UVB phototherapy to avoid burning (ie, erythema) at baseline and throughout the treatment course. These dose adjustments may have increased the overall clearance rates. If true, we predicted that younger adults treated with the same protocol would have cleared more quickly, either because of age-related differences or because they likely had fewer comorbidities and therefore fewer medications.
The second finding from Matthews et al13 that warranted further investigation was a higher erythema rate compared to the older adult study from the United Kingdom.17 We hypothesized that potentially greater use of photosensitizing medications in the United States could explain the higher erythema rates. Although medication-induced photosensitivity is less likely with NB-UVB phototherapy than with UVA, certain medications can cause UVB photosensitivity, including thiazides, quinidine, calcium channel antagonists, phenothiazines, and nonsteroidal anti-inflammatory drugs.8,19,20 Therefore, photosensitizing medication use either at baseline or during a course of NB-UVB phototherapy could increase the risk for erythema. Age-related skin changes also have been considered as a
This retrospective study aimed to determine if NB-UVB phototherapy is equally effective in both older and younger adults treated with the same protocol; to examine the association between the use of photosensitizing medications and clearance rates in both older and younger adults; and to examine the association between the use of photosensitizing medications and erythema rates in older vs younger adults.
Methods
Study Design and Patients—This retrospective cohort study used billing records to identify patients who received NB-UVB phototherapy at 3 different clinical sites within a large US health care system in Washington (Group Health Cooperative, now Kaiser Permanente Washington), serving more than 600,000 patients between January 1, 2012, and December 31, 2016. The institutional review board of Kaiser Permanente Washington Health Research Institute approved this study (IRB 1498087-4). Younger adults were classified as those 64 years or younger and older adults as those 65 years and older at the start of their phototherapy regimen. A power analysis determined that the optimal sample size for this study was 250 patients.
Individuals were excluded if they had fewer than 6 phototherapy treatments; a diagnosis of vitiligo, photosensitivity dermatitis, morphea, or pityriasis rubra pilaris; and/or treatment of the hands or feet only.
Phototherapy Protocol—Using a 48-lamp NB-UVB unit, trained phototherapy nurses provided all treatments following standardized treatment protocols13 based on previously published phototherapy guidelines.24 Nurses determined each patient’s disease clearance level using a 3-point clearance scale (high, medium, low).13 Each patient’s starting dose was determined based on the estimated MED for their skin phototype.
Statistical Analysis—Data were analyzed using Stata statistical software (StataCorp LLC). Univariate analyses were used to examine the data and identify outliers, bad values, and missing data, as well as to calculate descriptive statistics. Pearson χ2 and Fisher exact statistics were used to calculate differences in categorical variables. Linear multivariate regression models and logistic multivariate models were used to examine statistical relationships between variables. Statistical significance was defined as P≤.05.
Results
Patient Characteristics—Medical records were reviewed for 172 patients who received phototherapy between 2012 and 2016. Patients ranged in age from 23 to 91 years, with 102 patients 64 years and younger and 70 patients 65 years and older. Tables 1 and 2 outline the patient characteristics and conditions treated.

Phototherapy Effectiveness—

Photosensitizing Medications, Clearance Levels, and Clearance Rates—

Frequency of Treatments and Clearance Rates—Older adults more consistently completed the recommended frequency of treatments—3 times weekly—compared to younger adults (74.3% vs 58.5%). However, all patients who completed 3 treatments per week required a similar number of treatments to clear (older adults, mean [SD]: 35.7 [21.6]; younger adults, mean [SD]: 34.7 [19.0]; P=.85). Among patients completing 2 or fewer treatments per week, older adults required a mean (SD) of only 31 (9.0) treatments to clear vs 41.5 (21.3) treatments to clear for younger adults, but the difference was not statistically significant (P=.08). However, even those with suboptimal frequency ultimately achieved similar clearance levels.


Photosensitizing Medications and Erythema Rates—

Overall, phototherapy nurses adjusted the starting dose according to the phototype-based protocol an average of 69% of the time for patients on medications with photosensitivity listed as a potential side effect. However, the frequency depended significantly on the clinic (clinic A, 24%; clinic B, 92%; clinic C, 87%)(P≤.001). Nurses across all clinics consistently decreased the treatment dose when patients reported starting new photosensitizing medications. Patients with adjusted starting doses had slightly but not significantly higher clearance rates compared to those without (mean, 37.8 vs 35.5; t(104)=0.58; P=.56).

Comment
Impact of Photosensitizing Medications on Clearance—Photosensitizing medications and treatment frequency were 2 factors that might explain the slower clearance rates in younger adults. In this study, both groups of patients used similar numbers of photosensitizing medications, but more older adults were taking 3 or more medications (Table 3). We found no statistically significant relationship between taking photosensitizing medications and either the clearance rates or the level of clearance achieved in either age group.
Impact of Treatment Frequency—Weekly treatment frequency also was examined. One prior study demonstrated that treatments 3 times weekly led to a faster clearance time and higher clearance levels compared with twice-weekly treatment.7 When patients completed treatments twice weekly, it took an average of 1.5 times more days to clear, which impacted cost and clinical resource availability. The patients ranged in age from 17 to 80 years, but outcomes in older patients were not described separately.7 Interestingly, our study seemed to find a difference between age groups when the impact of treatment frequency was examined. Older adults completed nearly 4 fewer mean treatments to clear when treating less often, with more than 80% achieving high levels of clearance, whereas the younger adults required almost 7 more treatments to clear when they came in less frequently, with approximately 80% achieving a high level of clearance. As a result, our study found that in both age groups, slowing the treatment frequency extended the treatment time to clearance—more for the younger adults than the older adults—but did not significantly change the percentage of individuals reaching full clearance in either group.
Erythema Rates—There was no association between photosensitizing medications and erythema rates except when patients were taking at least 3 medications. Most medications that listed photosensitivity as a possible side effect did not specify their relevant range of UV radiation; therefore, all such medications were examined during this analysis. Prior research has shown UVB range photosensitizing medications include thiazides, quinidine, calcium channel antagonists, phenothiazines, and nonsteroidal anti-inflammatory drugs.19 A sensitivity analysis that focused only on these medications found no association between them and any particular grade of erythema. However, patients taking 3 or more of any medications listing photosensitivity as a side effect had an increased risk for grade 2 erythema.
Erythema rates in this study were consistent with a 2013 systematic review that reported 57% of patients with asymptomatic grade 1 erythema.25 In the 2 other comparative older adult studies, erythema rates varied widely: 35% in a study from Turkey18compared to only1.89% in a study from the United Kingdom.17
The starting dose for NB-UVB may drive erythema rates. The current study’s protocols were based on an estimated MED that is subjectively determined by the dermatology provider’s assessment of the patient’s skin sensitivity via examination and questions to the patient about their response to environmental sun exposure (ie, burning and tanning)26 and is frequently used to determine the starting dose and subsequent dose escalation. Certain medications have been found to increase photosensitivity and erythema,20 which can change an individual’s MED. If photosensitizing medications are started prior to or during a course of NB-UVB without a pretreatment MED, they might increase the risk for erythema. This study did not identify specific erythema-inducing medications but did find that taking 3 or more photosensitizing medications was associated with increased episodes of grade 2 erythema. Similarly, Harrop et al8 found that patients who were taking photosensitizing medications were more likely to have grade 2 or higher erythema, despite baseline MED testing, which is an established safety mechanism to reduce the risk and severity of erythema.14,20,27 The authors of a recent study of older adults in Taiwan specifically recommended MED testing due to the unpredictable influence of polypharmacy on MED calculations in this population.28 Therefore, this study’s use of an estimated MED in older adults may have influenced the starting dose as well as the incidence and severity of erythemic events. Age-related skin changes likely are ruled out as a consideration for mild erythema by the similarity of grade 1 erythema rates in both older and younger adults. Other studies have identified differences between the age groups, where older patients experienced more intense erythema in the late phase of UVB treatments.22,23 This phenomenon could increase the risk for a grade 2 erythema, which may correspond with this study’s findings.
Other potential causes of erythema were ruled out during our study, including erythema related to missed treatments and shielding mishaps. Other factors, however, may impact the level of sensitivity each patient has to phototherapy, including genetics, epigenetics, and cumulative sun damage. With NB-UVB, near-erythemogenic doses are optimal to achieve effective treatments but require a delicate balance to achieve, which may be more problematic for older adults, especially those taking several medications.
Study Limitations—Our study design made it difficult to draw conclusions about rarer dermatologic conditions. Some patients received treatments over years that were not included in the study period. Finally, power calculations suggested that our actual sample size was too small, with approximately one-third of the required sample missing.
Practical Implications—The goals of phototherapy are to achieve a high level of disease clearance with the fewest number of treatments possible and minimal side effects.
The extra staff training and patient monitoring required for MED testing likely is to add value and preserve resources if faster clearance rates could be achieved and may warrant further investigation. Phototherapy centers require standardized treatment protocols, diligent well-trained staff, and program monitoring to ensure consistent care to all patients. This study highlighted the ongoing opportunity for health care organizations to conduct evidence-based practice inquiries to continually optimize care for their patients.
Even with recent pharmacologic treatment advances, narrowband UVB (NB-UVB) phototherapy remains a versatile, safe, and efficacious adjunctive or exclusive treatment for multiple dermatologic conditions, including psoriasis and atopic dermatitis.
In a prior study, Matthews et al13 reported that 96% (50/52) of patients older than 65 years achieved medium to high levels of clearance with NB-UVB phototherapy. Nonetheless, 2 other findings in this study related to the number of treatments required to achieve clearance (ie, clearance rates) and erythema rates prompted further investigation. The first finding was higher-than-expected clearance rates. Older adults had a clearance rate with a mean of 33 treatments compared to prior studies featuring mean clearance rates of 20 to 28 treatments.7,8,14-16 This finding resembled a study in the United Kingdom17 with a median clearance rate in older adults of 30 treatments. In contrast, the median clearance rate from a study in Turkey18 was 42 treatments in older adults. We hypothesized that more photosensitizing medications used in older vs younger adults prompted more dose adjustments with NB-UVB phototherapy to avoid burning (ie, erythema) at baseline and throughout the treatment course. These dose adjustments may have increased the overall clearance rates. If true, we predicted that younger adults treated with the same protocol would have cleared more quickly, either because of age-related differences or because they likely had fewer comorbidities and therefore fewer medications.
The second finding from Matthews et al13 that warranted further investigation was a higher erythema rate compared to the older adult study from the United Kingdom.17 We hypothesized that potentially greater use of photosensitizing medications in the United States could explain the higher erythema rates. Although medication-induced photosensitivity is less likely with NB-UVB phototherapy than with UVA, certain medications can cause UVB photosensitivity, including thiazides, quinidine, calcium channel antagonists, phenothiazines, and nonsteroidal anti-inflammatory drugs.8,19,20 Therefore, photosensitizing medication use either at baseline or during a course of NB-UVB phototherapy could increase the risk for erythema. Age-related skin changes also have been considered as a
This retrospective study aimed to determine if NB-UVB phototherapy is equally effective in both older and younger adults treated with the same protocol; to examine the association between the use of photosensitizing medications and clearance rates in both older and younger adults; and to examine the association between the use of photosensitizing medications and erythema rates in older vs younger adults.
Methods
Study Design and Patients—This retrospective cohort study used billing records to identify patients who received NB-UVB phototherapy at 3 different clinical sites within a large US health care system in Washington (Group Health Cooperative, now Kaiser Permanente Washington), serving more than 600,000 patients between January 1, 2012, and December 31, 2016. The institutional review board of Kaiser Permanente Washington Health Research Institute approved this study (IRB 1498087-4). Younger adults were classified as those 64 years or younger and older adults as those 65 years and older at the start of their phototherapy regimen. A power analysis determined that the optimal sample size for this study was 250 patients.
Individuals were excluded if they had fewer than 6 phototherapy treatments; a diagnosis of vitiligo, photosensitivity dermatitis, morphea, or pityriasis rubra pilaris; and/or treatment of the hands or feet only.
Phototherapy Protocol—Using a 48-lamp NB-UVB unit, trained phototherapy nurses provided all treatments following standardized treatment protocols13 based on previously published phototherapy guidelines.24 Nurses determined each patient’s disease clearance level using a 3-point clearance scale (high, medium, low).13 Each patient’s starting dose was determined based on the estimated MED for their skin phototype.
Statistical Analysis—Data were analyzed using Stata statistical software (StataCorp LLC). Univariate analyses were used to examine the data and identify outliers, bad values, and missing data, as well as to calculate descriptive statistics. Pearson χ2 and Fisher exact statistics were used to calculate differences in categorical variables. Linear multivariate regression models and logistic multivariate models were used to examine statistical relationships between variables. Statistical significance was defined as P≤.05.
Results
Patient Characteristics—Medical records were reviewed for 172 patients who received phototherapy between 2012 and 2016. Patients ranged in age from 23 to 91 years, with 102 patients 64 years and younger and 70 patients 65 years and older. Tables 1 and 2 outline the patient characteristics and conditions treated.

Phototherapy Effectiveness—

Photosensitizing Medications, Clearance Levels, and Clearance Rates—

Frequency of Treatments and Clearance Rates—Older adults more consistently completed the recommended frequency of treatments—3 times weekly—compared to younger adults (74.3% vs 58.5%). However, all patients who completed 3 treatments per week required a similar number of treatments to clear (older adults, mean [SD]: 35.7 [21.6]; younger adults, mean [SD]: 34.7 [19.0]; P=.85). Among patients completing 2 or fewer treatments per week, older adults required a mean (SD) of only 31 (9.0) treatments to clear vs 41.5 (21.3) treatments to clear for younger adults, but the difference was not statistically significant (P=.08). However, even those with suboptimal frequency ultimately achieved similar clearance levels.


Photosensitizing Medications and Erythema Rates—

Overall, phototherapy nurses adjusted the starting dose according to the phototype-based protocol an average of 69% of the time for patients on medications with photosensitivity listed as a potential side effect. However, the frequency depended significantly on the clinic (clinic A, 24%; clinic B, 92%; clinic C, 87%)(P≤.001). Nurses across all clinics consistently decreased the treatment dose when patients reported starting new photosensitizing medications. Patients with adjusted starting doses had slightly but not significantly higher clearance rates compared to those without (mean, 37.8 vs 35.5; t(104)=0.58; P=.56).

Comment
Impact of Photosensitizing Medications on Clearance—Photosensitizing medications and treatment frequency were 2 factors that might explain the slower clearance rates in younger adults. In this study, both groups of patients used similar numbers of photosensitizing medications, but more older adults were taking 3 or more medications (Table 3). We found no statistically significant relationship between taking photosensitizing medications and either the clearance rates or the level of clearance achieved in either age group.
Impact of Treatment Frequency—Weekly treatment frequency also was examined. One prior study demonstrated that treatments 3 times weekly led to a faster clearance time and higher clearance levels compared with twice-weekly treatment.7 When patients completed treatments twice weekly, it took an average of 1.5 times more days to clear, which impacted cost and clinical resource availability. The patients ranged in age from 17 to 80 years, but outcomes in older patients were not described separately.7 Interestingly, our study seemed to find a difference between age groups when the impact of treatment frequency was examined. Older adults completed nearly 4 fewer mean treatments to clear when treating less often, with more than 80% achieving high levels of clearance, whereas the younger adults required almost 7 more treatments to clear when they came in less frequently, with approximately 80% achieving a high level of clearance. As a result, our study found that in both age groups, slowing the treatment frequency extended the treatment time to clearance—more for the younger adults than the older adults—but did not significantly change the percentage of individuals reaching full clearance in either group.
Erythema Rates—There was no association between photosensitizing medications and erythema rates except when patients were taking at least 3 medications. Most medications that listed photosensitivity as a possible side effect did not specify their relevant range of UV radiation; therefore, all such medications were examined during this analysis. Prior research has shown UVB range photosensitizing medications include thiazides, quinidine, calcium channel antagonists, phenothiazines, and nonsteroidal anti-inflammatory drugs.19 A sensitivity analysis that focused only on these medications found no association between them and any particular grade of erythema. However, patients taking 3 or more of any medications listing photosensitivity as a side effect had an increased risk for grade 2 erythema.
Erythema rates in this study were consistent with a 2013 systematic review that reported 57% of patients with asymptomatic grade 1 erythema.25 In the 2 other comparative older adult studies, erythema rates varied widely: 35% in a study from Turkey18compared to only1.89% in a study from the United Kingdom.17
The starting dose for NB-UVB may drive erythema rates. The current study’s protocols were based on an estimated MED that is subjectively determined by the dermatology provider’s assessment of the patient’s skin sensitivity via examination and questions to the patient about their response to environmental sun exposure (ie, burning and tanning)26 and is frequently used to determine the starting dose and subsequent dose escalation. Certain medications have been found to increase photosensitivity and erythema,20 which can change an individual’s MED. If photosensitizing medications are started prior to or during a course of NB-UVB without a pretreatment MED, they might increase the risk for erythema. This study did not identify specific erythema-inducing medications but did find that taking 3 or more photosensitizing medications was associated with increased episodes of grade 2 erythema. Similarly, Harrop et al8 found that patients who were taking photosensitizing medications were more likely to have grade 2 or higher erythema, despite baseline MED testing, which is an established safety mechanism to reduce the risk and severity of erythema.14,20,27 The authors of a recent study of older adults in Taiwan specifically recommended MED testing due to the unpredictable influence of polypharmacy on MED calculations in this population.28 Therefore, this study’s use of an estimated MED in older adults may have influenced the starting dose as well as the incidence and severity of erythemic events. Age-related skin changes likely are ruled out as a consideration for mild erythema by the similarity of grade 1 erythema rates in both older and younger adults. Other studies have identified differences between the age groups, where older patients experienced more intense erythema in the late phase of UVB treatments.22,23 This phenomenon could increase the risk for a grade 2 erythema, which may correspond with this study’s findings.
Other potential causes of erythema were ruled out during our study, including erythema related to missed treatments and shielding mishaps. Other factors, however, may impact the level of sensitivity each patient has to phototherapy, including genetics, epigenetics, and cumulative sun damage. With NB-UVB, near-erythemogenic doses are optimal to achieve effective treatments but require a delicate balance to achieve, which may be more problematic for older adults, especially those taking several medications.
Study Limitations—Our study design made it difficult to draw conclusions about rarer dermatologic conditions. Some patients received treatments over years that were not included in the study period. Finally, power calculations suggested that our actual sample size was too small, with approximately one-third of the required sample missing.
Practical Implications—The goals of phototherapy are to achieve a high level of disease clearance with the fewest number of treatments possible and minimal side effects.
The extra staff training and patient monitoring required for MED testing likely is to add value and preserve resources if faster clearance rates could be achieved and may warrant further investigation. Phototherapy centers require standardized treatment protocols, diligent well-trained staff, and program monitoring to ensure consistent care to all patients. This study highlighted the ongoing opportunity for health care organizations to conduct evidence-based practice inquiries to continually optimize care for their patients.
- Fernández-Guarino M, Aboin-Gonzalez S, Barchino L, et al. Treatment of moderate and severe adult chronic atopic dermatitis with narrow-band UVB and the combination of narrow-band UVB/UVA phototherapy. Dermatol Ther. 2016;29:19-23.
- Foerster J, Boswell K, West J, et al. Narrowband UVB treatment is highly effective and causes a strong reduction in the use of steroid and other creams in psoriasis patients in clinical practice. PLoS One. 2017;12:e0181813.
- Gambichler T, Breuckmann F, Boms S, et al. Narrowband UVB phototherapy in skin conditions beyond psoriasis. J Am Acad Dermatol. 2005;52:660-670.
- Ryu HH, Choe YS, Jo S, et al. Remission period in psoriasis after multiple cycles of narrowband ultraviolet B phototherapy. J Dermatol. 2014;41:622-627.
- Schneider LA, Hinrichs R, Scharffetter-Kochanek K. Phototherapy and photochemotherapy. Clin Dermatol. 2008;26:464-476.
- Tintle S, Shemer A, Suárez-Fariñas M, et al. Reversal of atopic dermatitis with narrow-band UVB phototherapy and biomarkers for therapeutic response. J Allergy Clin Immunol. 2011;128:583-593.e581-584.
- Cameron H, Dawe RS, Yule S, et al. A randomized, observer-blinded trial of twice vs. three times weekly narrowband ultraviolet B phototherapy for chronic plaque psoriasis. Br J Dermatol. 2002;147:973-978.
- Harrop G, Dawe RS, Ibbotson S. Are photosensitizing medications associated with increased risk of important erythemal reactions during ultraviolet B phototherapy? Br J Dermatol. 2018;179:1184-1185.
- Torres AE, Lyons AB, Hamzavi IH, et al. Role of phototherapy in the era of biologics. J Am Acad Dermatol. 2021;84:479-485.
- Bukvic´ć Mokos Z, Jovic´ A, Cˇeovic´ R, et al. Therapeutic challenges in the mature patient. Clin Dermatol. 2018;36:128-139.
- Di Lernia V, Goldust M. An overview of the efficacy and safety of systemic treatments for psoriasis in the elderly. Expert Opin Biol Ther. 2018;18:897-903.
- Oliveira C, Torres T. More than skin deep: the systemic nature of atopic dermatitis. Eur J Dermatol. 2019;29:250-258.
- Matthews S, Pike K, Chien A. Phototherapy: safe and effective for challenging skin conditions in older adults. Cutis. 2021;108:E15-E21.
- Rodríguez-Granados MT, Estany-Gestal A, Pousa-Martínez M, et al. Is it useful to calculate minimal erythema dose before narrowband UV-B phototherapy? Actas Dermosifiliogr. 2017;108:852-858.
- Parlak N, Kundakci N, Parlak A, et al. Narrowband ultraviolet B phototherapy starting and incremental dose in patients with psoriasis: comparison of percentage dose and fixed dose protocols. Photodermatol Photoimmunol Photomed. 2015;31:90-97.
- Kleinpenning MM, Smits T, Boezeman J, et al. Narrowband ultraviolet B therapy in psoriasis: randomized double-blind comparison of high-dose and low-dose irradiation regimens. Br J Dermatol. 2009;161:1351-1356.
- Powell JB, Gach JE. Phototherapy in the elderly. Clin Exp Dermatol. 2015;40:605-610.
- Bulur I, Erdogan HK, Aksu AE, et al. The efficacy and safety of phototherapy in geriatric patients: a retrospective study. An Bras Dermatol. 2018;93:33-38.
- Dawe RS, Ibbotson SH. Drug-induced photosensitivity. Dermatol Clin. 2014;32:363-368, ix.
- Cameron H, Dawe RS. Photosensitizing drugs may lower the narrow-band ultraviolet B (TL-01) minimal erythema dose. Br J Dermatol. 2000;142:389-390.
- Elmets CA, Lim HW, Stoff B, et al. Joint American Academy of Dermatology-National Psoriasis Foundation guidelines of care for the management and treatment of psoriasis with phototherapy. J Am Acad Dermatol. 2019;81:775-804.
- Gloor M, Scherotzke A. Age dependence of ultraviolet light-induced erythema following narrow-band UVB exposure. Photodermatol Photoimmunol Photomed. 2002;18:121-126.
- Cox NH, Diffey BL, Farr PM. The relationship between chronological age and the erythemal response to ultraviolet B radiation. Br J Dermatol. 1992;126:315-319.
- Morrison W. Phototherapy and Photochemotherapy for Skin Disease. 2nd ed. Informa Healthcare; 2005.
- Almutawa F, Alnomair N, Wang Y, et al. Systematic review of UV-based therapy for psoriasis. Am J Clin Dermatol. 2013;14:87-109.
- Trakatelli M, Bylaite-Bucinskiene M, Correia O, et al. Clinical assessment of skin phototypes: watch your words! Eur J Dermatol. 2017;27:615-619.
- Kwon IH, Kwon HH, Na SJ, et al. Could colorimetric method replace the individual minimal erythemal dose (MED) measurements in determining the initial dose of narrow-band UVB treatment for psoriasis patients with skin phototype III-V? J Eur Acad Dermatol Venereol. 2013;27:494-498.
- Chen WA, Chang CM. The minimal erythema dose of narrowband ultraviolet B in elderly Taiwanese [published online September 1, 2021]. Photodermatol Photoimmunol Photomed. doi:10.1111/phpp.12730
- Fernández-Guarino M, Aboin-Gonzalez S, Barchino L, et al. Treatment of moderate and severe adult chronic atopic dermatitis with narrow-band UVB and the combination of narrow-band UVB/UVA phototherapy. Dermatol Ther. 2016;29:19-23.
- Foerster J, Boswell K, West J, et al. Narrowband UVB treatment is highly effective and causes a strong reduction in the use of steroid and other creams in psoriasis patients in clinical practice. PLoS One. 2017;12:e0181813.
- Gambichler T, Breuckmann F, Boms S, et al. Narrowband UVB phototherapy in skin conditions beyond psoriasis. J Am Acad Dermatol. 2005;52:660-670.
- Ryu HH, Choe YS, Jo S, et al. Remission period in psoriasis after multiple cycles of narrowband ultraviolet B phototherapy. J Dermatol. 2014;41:622-627.
- Schneider LA, Hinrichs R, Scharffetter-Kochanek K. Phototherapy and photochemotherapy. Clin Dermatol. 2008;26:464-476.
- Tintle S, Shemer A, Suárez-Fariñas M, et al. Reversal of atopic dermatitis with narrow-band UVB phototherapy and biomarkers for therapeutic response. J Allergy Clin Immunol. 2011;128:583-593.e581-584.
- Cameron H, Dawe RS, Yule S, et al. A randomized, observer-blinded trial of twice vs. three times weekly narrowband ultraviolet B phototherapy for chronic plaque psoriasis. Br J Dermatol. 2002;147:973-978.
- Harrop G, Dawe RS, Ibbotson S. Are photosensitizing medications associated with increased risk of important erythemal reactions during ultraviolet B phototherapy? Br J Dermatol. 2018;179:1184-1185.
- Torres AE, Lyons AB, Hamzavi IH, et al. Role of phototherapy in the era of biologics. J Am Acad Dermatol. 2021;84:479-485.
- Bukvic´ć Mokos Z, Jovic´ A, Cˇeovic´ R, et al. Therapeutic challenges in the mature patient. Clin Dermatol. 2018;36:128-139.
- Di Lernia V, Goldust M. An overview of the efficacy and safety of systemic treatments for psoriasis in the elderly. Expert Opin Biol Ther. 2018;18:897-903.
- Oliveira C, Torres T. More than skin deep: the systemic nature of atopic dermatitis. Eur J Dermatol. 2019;29:250-258.
- Matthews S, Pike K, Chien A. Phototherapy: safe and effective for challenging skin conditions in older adults. Cutis. 2021;108:E15-E21.
- Rodríguez-Granados MT, Estany-Gestal A, Pousa-Martínez M, et al. Is it useful to calculate minimal erythema dose before narrowband UV-B phototherapy? Actas Dermosifiliogr. 2017;108:852-858.
- Parlak N, Kundakci N, Parlak A, et al. Narrowband ultraviolet B phototherapy starting and incremental dose in patients with psoriasis: comparison of percentage dose and fixed dose protocols. Photodermatol Photoimmunol Photomed. 2015;31:90-97.
- Kleinpenning MM, Smits T, Boezeman J, et al. Narrowband ultraviolet B therapy in psoriasis: randomized double-blind comparison of high-dose and low-dose irradiation regimens. Br J Dermatol. 2009;161:1351-1356.
- Powell JB, Gach JE. Phototherapy in the elderly. Clin Exp Dermatol. 2015;40:605-610.
- Bulur I, Erdogan HK, Aksu AE, et al. The efficacy and safety of phototherapy in geriatric patients: a retrospective study. An Bras Dermatol. 2018;93:33-38.
- Dawe RS, Ibbotson SH. Drug-induced photosensitivity. Dermatol Clin. 2014;32:363-368, ix.
- Cameron H, Dawe RS. Photosensitizing drugs may lower the narrow-band ultraviolet B (TL-01) minimal erythema dose. Br J Dermatol. 2000;142:389-390.
- Elmets CA, Lim HW, Stoff B, et al. Joint American Academy of Dermatology-National Psoriasis Foundation guidelines of care for the management and treatment of psoriasis with phototherapy. J Am Acad Dermatol. 2019;81:775-804.
- Gloor M, Scherotzke A. Age dependence of ultraviolet light-induced erythema following narrow-band UVB exposure. Photodermatol Photoimmunol Photomed. 2002;18:121-126.
- Cox NH, Diffey BL, Farr PM. The relationship between chronological age and the erythemal response to ultraviolet B radiation. Br J Dermatol. 1992;126:315-319.
- Morrison W. Phototherapy and Photochemotherapy for Skin Disease. 2nd ed. Informa Healthcare; 2005.
- Almutawa F, Alnomair N, Wang Y, et al. Systematic review of UV-based therapy for psoriasis. Am J Clin Dermatol. 2013;14:87-109.
- Trakatelli M, Bylaite-Bucinskiene M, Correia O, et al. Clinical assessment of skin phototypes: watch your words! Eur J Dermatol. 2017;27:615-619.
- Kwon IH, Kwon HH, Na SJ, et al. Could colorimetric method replace the individual minimal erythemal dose (MED) measurements in determining the initial dose of narrow-band UVB treatment for psoriasis patients with skin phototype III-V? J Eur Acad Dermatol Venereol. 2013;27:494-498.
- Chen WA, Chang CM. The minimal erythema dose of narrowband ultraviolet B in elderly Taiwanese [published online September 1, 2021]. Photodermatol Photoimmunol Photomed. doi:10.1111/phpp.12730
Practice Points
- Narrowband UVB (NB-UVB) phototherapy remains a safe and efficacious nonpharmacologic treatment for dermatologic conditions in older and younger adults.
- Compared to younger adults, older adults using the same protocols need similar or even fewer treatments to achieve high levels of clearance.
- Individuals taking 3 or more photosensitizing medications, regardless of age, may be at higher risk for substantial erythema with NB-UVB phototherapy.
- Phototherapy program monitoring is important to ensure quality care and investigate opportunities for care optimization.
Heparin pretreatment may safely open arteries before STEMI cath
, suggests a large registry study.
An open infarct-related artery (IRA) at angiography on cath-lab arrival presents STEMI patients an opportunity for earlier reperfusion and a chance, in theory at least, for smaller infarcts and maybe improved clinical outcomes.
In the new analysis, which covers more than 40,000 patients with STEMI in Sweden, the 38% who received heparin before cath-lab arrival were 11% less likely to show IRA occlusion at angiography prior to direct percutaneous coronary intervention (PCI). They also showed a 13% lower 30-day mortality compared with patients who were started on heparin in the cath lab. Importantly, their risk of major bleeding in the hospital did not increase.
The “early reperfusion” associated with IRA patency at angiography “could have long-term benefit due to smaller infarct size,” potentially explaining the observed 30-day survival gain in the pretreatment group, Oskar Love Emilsson, Lund (Sweden) University, said in an interview.
Mr. Emilsson, a third-year medical student, reported the analysis at the annual congress of the European Society of Cardiology, and is lead author on its same-day publication in the journal EuroIntervention.
He mentioned a few cautions in interpreting the study, which is based primarily on data from the Swedish Coronary Angiography and Angioplasty Registry (SCAAR). It included several sensitivity analyses that continued to back pretreatment heparin as a significant predictor of an unoccluded IRA but didn’t consistently support the 30-day mortality benefit seen in the primary analysis.
And, although the pretreatment group overall didn’t have more major bleeds, the risk did go up significantly for those older than 75 or those who weighed less than 60 kg (132 pounds) or underwent catheterization with an access route other than the radial artery. Extra caution should be exercised in such patients who receive heparin before cath-lab arrival for PCI, Mr. Emilsson observed.
“Our results suggest that heparin pretreatment might be a good option to improve patency of infarct related arteries in STEMI,” and potentially clinical outcomes, he said. “However, a definite answer would require a randomized controlled trial.”
Meanwhile, the current study may be the largest yet to look at clinical outcomes after pretreatment with unfractionated heparin before PCI for acute STEMI, the report states. There have been some observational studies, subanalyses of STEMI trials, and even a few limited randomized trials – including the HEAP trial published in 2000 – to weigh in on the subject. Some have supported the strategy, others have not.
“With rapid door-to-balloon times in STEMI, it can be challenging to show a significant difference between a prehospital heparin approach and heparin given in the lab,” observed Sunil V. Rao, MD, NYU Langone Health System, New York, who is not connected with the current study.
Many EDs in the United States have “a STEMI protocol that calls for an IV bolus of heparin. It would be tougher in the U.S. to give it in the ambulance but again, it’s not clear how much advantage that would really provide,” he told this news organization.
Support from randomized trials would be needed before the practice could be formally recommended. “The SCAAR registries have set the standard for how registries should be conducted,” Dr. Rao said. “This is a very well done observational study, but it is observational.”
The priority for STEMI patients, he added, “really should be to get them to the lab as fast as possible. If the ED protocol includes heparin before the cath lab, that’s great, but I don’t think we should delay getting these patients to the lab to accommodate pre–cath-lab heparin.”
The current analysis covered 41,631 patients with STEMI from 2008 through to 2016, of whom 38% were pretreated with heparin in an ambulance or the ED. The remaining 62% initiated heparin in the cath lab.
About one-third of the group had an open IRA at angiography. The adjusted risk ratio (RR) for IRA occlusion at angiography for patients pretreated vs. not pretreated with heparin was 0.89 (95% confidence interval [CI], 0.87-0.90).
The corresponding RR for death within 30 days was 0.87 (95% CI, 0.77-0.99), and for major in-hospital bleeding it was 1.01 (95% CI, 0.86-1.18).
The analysis was adjusted for other medications received before cath-lab arrival, especially a long list of antiplatelets and non-heparin antithrombins. That strengthens the case for heparin pretreatment as an independent predictor of an open IRA at initial angiography, Mr. Emilsson said.
Comparisons of propensity-score–matched subgroups of the total cohort, conducted separately for the IRA-occlusion endpoint and the endpoints of 30-day mortality and major bleeding, produced similar results.
Some observational data suggest that antiplatelet pretreatment with a P2Y12 inhibitor may promote IRA patency on angiography after cath lab arrival, Dr. Rao observed. “This indicates that there probably is a role of earlier antithrombotic therapy in STEMI patients, but the randomized trials have not shown a consistent benefit,” he said, referring in particular to the ATLANTIC trial.
Mr. Emilsson and Dr. Rao disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, suggests a large registry study.
An open infarct-related artery (IRA) at angiography on cath-lab arrival presents STEMI patients an opportunity for earlier reperfusion and a chance, in theory at least, for smaller infarcts and maybe improved clinical outcomes.
In the new analysis, which covers more than 40,000 patients with STEMI in Sweden, the 38% who received heparin before cath-lab arrival were 11% less likely to show IRA occlusion at angiography prior to direct percutaneous coronary intervention (PCI). They also showed a 13% lower 30-day mortality compared with patients who were started on heparin in the cath lab. Importantly, their risk of major bleeding in the hospital did not increase.
The “early reperfusion” associated with IRA patency at angiography “could have long-term benefit due to smaller infarct size,” potentially explaining the observed 30-day survival gain in the pretreatment group, Oskar Love Emilsson, Lund (Sweden) University, said in an interview.
Mr. Emilsson, a third-year medical student, reported the analysis at the annual congress of the European Society of Cardiology, and is lead author on its same-day publication in the journal EuroIntervention.
He mentioned a few cautions in interpreting the study, which is based primarily on data from the Swedish Coronary Angiography and Angioplasty Registry (SCAAR). It included several sensitivity analyses that continued to back pretreatment heparin as a significant predictor of an unoccluded IRA but didn’t consistently support the 30-day mortality benefit seen in the primary analysis.
And, although the pretreatment group overall didn’t have more major bleeds, the risk did go up significantly for those older than 75 or those who weighed less than 60 kg (132 pounds) or underwent catheterization with an access route other than the radial artery. Extra caution should be exercised in such patients who receive heparin before cath-lab arrival for PCI, Mr. Emilsson observed.
“Our results suggest that heparin pretreatment might be a good option to improve patency of infarct related arteries in STEMI,” and potentially clinical outcomes, he said. “However, a definite answer would require a randomized controlled trial.”
Meanwhile, the current study may be the largest yet to look at clinical outcomes after pretreatment with unfractionated heparin before PCI for acute STEMI, the report states. There have been some observational studies, subanalyses of STEMI trials, and even a few limited randomized trials – including the HEAP trial published in 2000 – to weigh in on the subject. Some have supported the strategy, others have not.
“With rapid door-to-balloon times in STEMI, it can be challenging to show a significant difference between a prehospital heparin approach and heparin given in the lab,” observed Sunil V. Rao, MD, NYU Langone Health System, New York, who is not connected with the current study.
Many EDs in the United States have “a STEMI protocol that calls for an IV bolus of heparin. It would be tougher in the U.S. to give it in the ambulance but again, it’s not clear how much advantage that would really provide,” he told this news organization.
Support from randomized trials would be needed before the practice could be formally recommended. “The SCAAR registries have set the standard for how registries should be conducted,” Dr. Rao said. “This is a very well done observational study, but it is observational.”
The priority for STEMI patients, he added, “really should be to get them to the lab as fast as possible. If the ED protocol includes heparin before the cath lab, that’s great, but I don’t think we should delay getting these patients to the lab to accommodate pre–cath-lab heparin.”
The current analysis covered 41,631 patients with STEMI from 2008 through to 2016, of whom 38% were pretreated with heparin in an ambulance or the ED. The remaining 62% initiated heparin in the cath lab.
About one-third of the group had an open IRA at angiography. The adjusted risk ratio (RR) for IRA occlusion at angiography for patients pretreated vs. not pretreated with heparin was 0.89 (95% confidence interval [CI], 0.87-0.90).
The corresponding RR for death within 30 days was 0.87 (95% CI, 0.77-0.99), and for major in-hospital bleeding it was 1.01 (95% CI, 0.86-1.18).
The analysis was adjusted for other medications received before cath-lab arrival, especially a long list of antiplatelets and non-heparin antithrombins. That strengthens the case for heparin pretreatment as an independent predictor of an open IRA at initial angiography, Mr. Emilsson said.
Comparisons of propensity-score–matched subgroups of the total cohort, conducted separately for the IRA-occlusion endpoint and the endpoints of 30-day mortality and major bleeding, produced similar results.
Some observational data suggest that antiplatelet pretreatment with a P2Y12 inhibitor may promote IRA patency on angiography after cath lab arrival, Dr. Rao observed. “This indicates that there probably is a role of earlier antithrombotic therapy in STEMI patients, but the randomized trials have not shown a consistent benefit,” he said, referring in particular to the ATLANTIC trial.
Mr. Emilsson and Dr. Rao disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, suggests a large registry study.
An open infarct-related artery (IRA) at angiography on cath-lab arrival presents STEMI patients an opportunity for earlier reperfusion and a chance, in theory at least, for smaller infarcts and maybe improved clinical outcomes.
In the new analysis, which covers more than 40,000 patients with STEMI in Sweden, the 38% who received heparin before cath-lab arrival were 11% less likely to show IRA occlusion at angiography prior to direct percutaneous coronary intervention (PCI). They also showed a 13% lower 30-day mortality compared with patients who were started on heparin in the cath lab. Importantly, their risk of major bleeding in the hospital did not increase.
The “early reperfusion” associated with IRA patency at angiography “could have long-term benefit due to smaller infarct size,” potentially explaining the observed 30-day survival gain in the pretreatment group, Oskar Love Emilsson, Lund (Sweden) University, said in an interview.
Mr. Emilsson, a third-year medical student, reported the analysis at the annual congress of the European Society of Cardiology, and is lead author on its same-day publication in the journal EuroIntervention.
He mentioned a few cautions in interpreting the study, which is based primarily on data from the Swedish Coronary Angiography and Angioplasty Registry (SCAAR). It included several sensitivity analyses that continued to back pretreatment heparin as a significant predictor of an unoccluded IRA but didn’t consistently support the 30-day mortality benefit seen in the primary analysis.
And, although the pretreatment group overall didn’t have more major bleeds, the risk did go up significantly for those older than 75 or those who weighed less than 60 kg (132 pounds) or underwent catheterization with an access route other than the radial artery. Extra caution should be exercised in such patients who receive heparin before cath-lab arrival for PCI, Mr. Emilsson observed.
“Our results suggest that heparin pretreatment might be a good option to improve patency of infarct related arteries in STEMI,” and potentially clinical outcomes, he said. “However, a definite answer would require a randomized controlled trial.”
Meanwhile, the current study may be the largest yet to look at clinical outcomes after pretreatment with unfractionated heparin before PCI for acute STEMI, the report states. There have been some observational studies, subanalyses of STEMI trials, and even a few limited randomized trials – including the HEAP trial published in 2000 – to weigh in on the subject. Some have supported the strategy, others have not.
“With rapid door-to-balloon times in STEMI, it can be challenging to show a significant difference between a prehospital heparin approach and heparin given in the lab,” observed Sunil V. Rao, MD, NYU Langone Health System, New York, who is not connected with the current study.
Many EDs in the United States have “a STEMI protocol that calls for an IV bolus of heparin. It would be tougher in the U.S. to give it in the ambulance but again, it’s not clear how much advantage that would really provide,” he told this news organization.
Support from randomized trials would be needed before the practice could be formally recommended. “The SCAAR registries have set the standard for how registries should be conducted,” Dr. Rao said. “This is a very well done observational study, but it is observational.”
The priority for STEMI patients, he added, “really should be to get them to the lab as fast as possible. If the ED protocol includes heparin before the cath lab, that’s great, but I don’t think we should delay getting these patients to the lab to accommodate pre–cath-lab heparin.”
The current analysis covered 41,631 patients with STEMI from 2008 through to 2016, of whom 38% were pretreated with heparin in an ambulance or the ED. The remaining 62% initiated heparin in the cath lab.
About one-third of the group had an open IRA at angiography. The adjusted risk ratio (RR) for IRA occlusion at angiography for patients pretreated vs. not pretreated with heparin was 0.89 (95% confidence interval [CI], 0.87-0.90).
The corresponding RR for death within 30 days was 0.87 (95% CI, 0.77-0.99), and for major in-hospital bleeding it was 1.01 (95% CI, 0.86-1.18).
The analysis was adjusted for other medications received before cath-lab arrival, especially a long list of antiplatelets and non-heparin antithrombins. That strengthens the case for heparin pretreatment as an independent predictor of an open IRA at initial angiography, Mr. Emilsson said.
Comparisons of propensity-score–matched subgroups of the total cohort, conducted separately for the IRA-occlusion endpoint and the endpoints of 30-day mortality and major bleeding, produced similar results.
Some observational data suggest that antiplatelet pretreatment with a P2Y12 inhibitor may promote IRA patency on angiography after cath lab arrival, Dr. Rao observed. “This indicates that there probably is a role of earlier antithrombotic therapy in STEMI patients, but the randomized trials have not shown a consistent benefit,” he said, referring in particular to the ATLANTIC trial.
Mr. Emilsson and Dr. Rao disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ESC CONGRESS 2022
FDA warns of cancer risk in scar tissue around breast implants
.
The FDA safety communication is based on several dozen reports of these cancers occurring in the capsule or scar tissue around breast implants. This issue differs from breast implant–associated anaplastic large-cell lymphoma (BIA-ALCL) – a known risk among implant recipients.
“After preliminary review of published literature as part of our ongoing monitoring of the safety of breast implants, the FDA is aware of less than 20 cases of SCC and less than 30 cases of various lymphomas in the capsule around the breast implant,” the agency’s alert explains.
One avenue through which the FDA has identified cases is via medical device reports. As of Sept. 1, the FDA has received 10 medical device reports about SCC related to breast implants and 12 about various lymphomas.
The incidence rate and risk factors for these events are currently unknown, but reports of SCC and various lymphomas in the capsule around the breast implants have been reported for both textured and smooth breast implants, as well as for both saline and silicone breast implants. In some cases, the cancers were diagnosed years after breast implant surgery.
Reported signs and symptoms included swelling, pain, lumps, or skin changes.
Although the risks of SCC and lymphomas in the tissue around breast implants appears rare, “when safety risks with medical devices are identified, we wanted to provide clear and understandable information to the public as quickly as possible,” Binita Ashar, MD, director of the Office of Surgical and Infection Control Devices, FDA Center for Devices and Radiological Health, explained in a press release.
Patients and providers are strongly encouraged to report breast implant–related problems and cases of SCC or lymphoma of the breast implant capsule to MedWatch, the FDA’s adverse event reporting program.
The FDA plans to complete “a thorough literature review” as well as “identify ways to collect more detailed information regarding patient cases.”
A version of this article first appeared on Medscape.com.
.
The FDA safety communication is based on several dozen reports of these cancers occurring in the capsule or scar tissue around breast implants. This issue differs from breast implant–associated anaplastic large-cell lymphoma (BIA-ALCL) – a known risk among implant recipients.
“After preliminary review of published literature as part of our ongoing monitoring of the safety of breast implants, the FDA is aware of less than 20 cases of SCC and less than 30 cases of various lymphomas in the capsule around the breast implant,” the agency’s alert explains.
One avenue through which the FDA has identified cases is via medical device reports. As of Sept. 1, the FDA has received 10 medical device reports about SCC related to breast implants and 12 about various lymphomas.
The incidence rate and risk factors for these events are currently unknown, but reports of SCC and various lymphomas in the capsule around the breast implants have been reported for both textured and smooth breast implants, as well as for both saline and silicone breast implants. In some cases, the cancers were diagnosed years after breast implant surgery.
Reported signs and symptoms included swelling, pain, lumps, or skin changes.
Although the risks of SCC and lymphomas in the tissue around breast implants appears rare, “when safety risks with medical devices are identified, we wanted to provide clear and understandable information to the public as quickly as possible,” Binita Ashar, MD, director of the Office of Surgical and Infection Control Devices, FDA Center for Devices and Radiological Health, explained in a press release.
Patients and providers are strongly encouraged to report breast implant–related problems and cases of SCC or lymphoma of the breast implant capsule to MedWatch, the FDA’s adverse event reporting program.
The FDA plans to complete “a thorough literature review” as well as “identify ways to collect more detailed information regarding patient cases.”
A version of this article first appeared on Medscape.com.
.
The FDA safety communication is based on several dozen reports of these cancers occurring in the capsule or scar tissue around breast implants. This issue differs from breast implant–associated anaplastic large-cell lymphoma (BIA-ALCL) – a known risk among implant recipients.
“After preliminary review of published literature as part of our ongoing monitoring of the safety of breast implants, the FDA is aware of less than 20 cases of SCC and less than 30 cases of various lymphomas in the capsule around the breast implant,” the agency’s alert explains.
One avenue through which the FDA has identified cases is via medical device reports. As of Sept. 1, the FDA has received 10 medical device reports about SCC related to breast implants and 12 about various lymphomas.
The incidence rate and risk factors for these events are currently unknown, but reports of SCC and various lymphomas in the capsule around the breast implants have been reported for both textured and smooth breast implants, as well as for both saline and silicone breast implants. In some cases, the cancers were diagnosed years after breast implant surgery.
Reported signs and symptoms included swelling, pain, lumps, or skin changes.
Although the risks of SCC and lymphomas in the tissue around breast implants appears rare, “when safety risks with medical devices are identified, we wanted to provide clear and understandable information to the public as quickly as possible,” Binita Ashar, MD, director of the Office of Surgical and Infection Control Devices, FDA Center for Devices and Radiological Health, explained in a press release.
Patients and providers are strongly encouraged to report breast implant–related problems and cases of SCC or lymphoma of the breast implant capsule to MedWatch, the FDA’s adverse event reporting program.
The FDA plans to complete “a thorough literature review” as well as “identify ways to collect more detailed information regarding patient cases.”
A version of this article first appeared on Medscape.com.
Gene mutations may drive lung cancer in never-smokers
Small cell lung cancer has traditionally been attributed almost exclusively to tobacco exposure, but some recent studies have suggested a higher than expected prevalence among nonsmokers. indicating that the subgroups may have unique disease characteristics. Key differences included a lower frequency of TP53 gene mutations and a higher frequency of epidermal growth factor receptor (EGFR) alterations in never smokers.
About 6.9% of small cell lung cancer patients in the CASPIAN study were nonsmokers, as were 3.0% in the IMpower133 study.
“Given that the pathogenesis of small cell lung cancer is often tied to the damaging effects of tobacco, we hypothesized that small cell lung cancer in never-smokers would possess distinct molecular attributes. Our data does not provide any solid evidence for any treatment implications, though it does raise therapeutic questions which we believe deserve further exploration,” said Michael Oh, MD, during a presentation of the study results at the annual meeting of the European Society for Medical Oncology. Dr. Oh is a fellow at the University of California, Los Angeles.
The topic is important clinically, according to Antonio Passaro, MD, PhD, who served as a discussant during the session. He noted that small cell lung cancer in never-smokers is the seventh-most common cause of cancer-related mortality worldwide. In non–small cell lung cancer, rates of tobacco-associated disease have been decreasing, but there are increases in diagnoses among never smokers. Nonsmoking small cell lung cancer patients do not have better prognoses, and novel therapies and advances like immunotherapy and low-dose CT lung cancer screening disproportionately benefit current or former smokers.
Potential risk factors for never-smokers include environmental exposures like radon gas, cooking oil vapors, indoor and outdoor wood burning, and genetic and viral factors. “At the present time we do not have the knowledge to identify the most important factor in development of lung cancer in never-smoking [patients],” said Dr. Passaro, who is a medical oncologist at the European Institute of Oncology in Milan.
He added that the current study results are interesting but need much more follow-up, such as “longitudinal studies combining detailed clinical annotation with tissue and blood sampling. Here there is a need for collaborative efforts.” Key questions include the roles of the genomic landscape in normal lung tissue may play, the lung micro-environment, genetic factors, and environmental exposures.
One key possibility is air pollution. “We know that lung cancer in never-smokers is frequent in some countries, for example in Asian countries and it is more frequent in the United States than in Europe, but to find an explanation to this kind of data is difficult at the present time,” Dr. Passaro said.
The researchers retrospectively analyzed data from 608 current or former smokers and 54 never-smokers with small cell lung cancer, with the latter making up 8% of the total population. 70.4% of never-smokers and 55.1% of current or former smokers were female (P = .031). There was no significant between-group difference with respect to age at diagnosis or race.
Somatic mutations were similar to what has been found in previous studies for current or former smokers. 85.2% had changes in TP53, compared with just 59.3% of never-smokers (Q < .001). Changes to EGFR were more common in never-smokers, occurring in 25.9% versus 2.6% (Q < .001). PIK3CA alterations were also more common in never-smokers (14.8% vs. 3.6%; Q = 0.022). There was no significant difference between the two groups with respect to changes in RB1.
Never smokers had tumors with less immune cell infiltration (P = .008), including fewer CD4+ T cells, CD8+ T cells, and macrophages. Their tumor mutation burden was also lower (median, 2.59 vs. 4.99; P < .001).
Dr. Oh has no relevant financial disclosures. Dr. Passaro has consulted, advised, and received research funding from a wide range of pharmaceutical companies.
Small cell lung cancer has traditionally been attributed almost exclusively to tobacco exposure, but some recent studies have suggested a higher than expected prevalence among nonsmokers. indicating that the subgroups may have unique disease characteristics. Key differences included a lower frequency of TP53 gene mutations and a higher frequency of epidermal growth factor receptor (EGFR) alterations in never smokers.
About 6.9% of small cell lung cancer patients in the CASPIAN study were nonsmokers, as were 3.0% in the IMpower133 study.
“Given that the pathogenesis of small cell lung cancer is often tied to the damaging effects of tobacco, we hypothesized that small cell lung cancer in never-smokers would possess distinct molecular attributes. Our data does not provide any solid evidence for any treatment implications, though it does raise therapeutic questions which we believe deserve further exploration,” said Michael Oh, MD, during a presentation of the study results at the annual meeting of the European Society for Medical Oncology. Dr. Oh is a fellow at the University of California, Los Angeles.
The topic is important clinically, according to Antonio Passaro, MD, PhD, who served as a discussant during the session. He noted that small cell lung cancer in never-smokers is the seventh-most common cause of cancer-related mortality worldwide. In non–small cell lung cancer, rates of tobacco-associated disease have been decreasing, but there are increases in diagnoses among never smokers. Nonsmoking small cell lung cancer patients do not have better prognoses, and novel therapies and advances like immunotherapy and low-dose CT lung cancer screening disproportionately benefit current or former smokers.
Potential risk factors for never-smokers include environmental exposures like radon gas, cooking oil vapors, indoor and outdoor wood burning, and genetic and viral factors. “At the present time we do not have the knowledge to identify the most important factor in development of lung cancer in never-smoking [patients],” said Dr. Passaro, who is a medical oncologist at the European Institute of Oncology in Milan.
He added that the current study results are interesting but need much more follow-up, such as “longitudinal studies combining detailed clinical annotation with tissue and blood sampling. Here there is a need for collaborative efforts.” Key questions include the roles of the genomic landscape in normal lung tissue may play, the lung micro-environment, genetic factors, and environmental exposures.
One key possibility is air pollution. “We know that lung cancer in never-smokers is frequent in some countries, for example in Asian countries and it is more frequent in the United States than in Europe, but to find an explanation to this kind of data is difficult at the present time,” Dr. Passaro said.
The researchers retrospectively analyzed data from 608 current or former smokers and 54 never-smokers with small cell lung cancer, with the latter making up 8% of the total population. 70.4% of never-smokers and 55.1% of current or former smokers were female (P = .031). There was no significant between-group difference with respect to age at diagnosis or race.
Somatic mutations were similar to what has been found in previous studies for current or former smokers. 85.2% had changes in TP53, compared with just 59.3% of never-smokers (Q < .001). Changes to EGFR were more common in never-smokers, occurring in 25.9% versus 2.6% (Q < .001). PIK3CA alterations were also more common in never-smokers (14.8% vs. 3.6%; Q = 0.022). There was no significant difference between the two groups with respect to changes in RB1.
Never smokers had tumors with less immune cell infiltration (P = .008), including fewer CD4+ T cells, CD8+ T cells, and macrophages. Their tumor mutation burden was also lower (median, 2.59 vs. 4.99; P < .001).
Dr. Oh has no relevant financial disclosures. Dr. Passaro has consulted, advised, and received research funding from a wide range of pharmaceutical companies.
Small cell lung cancer has traditionally been attributed almost exclusively to tobacco exposure, but some recent studies have suggested a higher than expected prevalence among nonsmokers. indicating that the subgroups may have unique disease characteristics. Key differences included a lower frequency of TP53 gene mutations and a higher frequency of epidermal growth factor receptor (EGFR) alterations in never smokers.
About 6.9% of small cell lung cancer patients in the CASPIAN study were nonsmokers, as were 3.0% in the IMpower133 study.
“Given that the pathogenesis of small cell lung cancer is often tied to the damaging effects of tobacco, we hypothesized that small cell lung cancer in never-smokers would possess distinct molecular attributes. Our data does not provide any solid evidence for any treatment implications, though it does raise therapeutic questions which we believe deserve further exploration,” said Michael Oh, MD, during a presentation of the study results at the annual meeting of the European Society for Medical Oncology. Dr. Oh is a fellow at the University of California, Los Angeles.
The topic is important clinically, according to Antonio Passaro, MD, PhD, who served as a discussant during the session. He noted that small cell lung cancer in never-smokers is the seventh-most common cause of cancer-related mortality worldwide. In non–small cell lung cancer, rates of tobacco-associated disease have been decreasing, but there are increases in diagnoses among never smokers. Nonsmoking small cell lung cancer patients do not have better prognoses, and novel therapies and advances like immunotherapy and low-dose CT lung cancer screening disproportionately benefit current or former smokers.
Potential risk factors for never-smokers include environmental exposures like radon gas, cooking oil vapors, indoor and outdoor wood burning, and genetic and viral factors. “At the present time we do not have the knowledge to identify the most important factor in development of lung cancer in never-smoking [patients],” said Dr. Passaro, who is a medical oncologist at the European Institute of Oncology in Milan.
He added that the current study results are interesting but need much more follow-up, such as “longitudinal studies combining detailed clinical annotation with tissue and blood sampling. Here there is a need for collaborative efforts.” Key questions include the roles of the genomic landscape in normal lung tissue may play, the lung micro-environment, genetic factors, and environmental exposures.
One key possibility is air pollution. “We know that lung cancer in never-smokers is frequent in some countries, for example in Asian countries and it is more frequent in the United States than in Europe, but to find an explanation to this kind of data is difficult at the present time,” Dr. Passaro said.
The researchers retrospectively analyzed data from 608 current or former smokers and 54 never-smokers with small cell lung cancer, with the latter making up 8% of the total population. 70.4% of never-smokers and 55.1% of current or former smokers were female (P = .031). There was no significant between-group difference with respect to age at diagnosis or race.
Somatic mutations were similar to what has been found in previous studies for current or former smokers. 85.2% had changes in TP53, compared with just 59.3% of never-smokers (Q < .001). Changes to EGFR were more common in never-smokers, occurring in 25.9% versus 2.6% (Q < .001). PIK3CA alterations were also more common in never-smokers (14.8% vs. 3.6%; Q = 0.022). There was no significant difference between the two groups with respect to changes in RB1.
Never smokers had tumors with less immune cell infiltration (P = .008), including fewer CD4+ T cells, CD8+ T cells, and macrophages. Their tumor mutation burden was also lower (median, 2.59 vs. 4.99; P < .001).
Dr. Oh has no relevant financial disclosures. Dr. Passaro has consulted, advised, and received research funding from a wide range of pharmaceutical companies.
AT ESMO CONGRESS 2022
Ages and Stages Questionnaire a first step to find developmental delays
The commonly used but sometimes debated Ages and Stages Questionnaire (ASQ), has modest utility for identifying developmental delays in young children, an Australian review and meta-analysis found.
On this easily administered parent-completed screening tool, scores of more than 2 standard deviations below the mean in more than one of five domains had moderate sensitivity and specificity to predict any delay, severe delay, motor delay, and cognitive delay, according to neonatologist Shripada Rao, PhD, a clinical associate professor in the neonatal intensive care unit at Perth Hospital and the University of Western Australia, also in Perth, and colleagues.
If a child of 12-60 months passes all ASQ domains, there is a moderate probability that child does not have severe developmental delay, the researchers concluded. If a child in that age range fails the motor or cognitive domain, there is a moderate probability that some motor or cognitive delay is present. The authors say the tool may work best as a screening test to identify children in need of more formal assessment.
“Our meta-analysis found that ASQ was somewhat more predictive in older children (older than 24 months), compared with younger age groups of 12-24 months,” Dr. Rao said in an interview. “However, the sample size for these comparisons was too small to reach definite conclusions, and we have called for future studies to evaluate ASQ separately for different age groups.”
Early identification of developmental delay in children is essential to enable timely intervention,” Dr. Rao and associates wrote in JAMA Pediatrics.
While formal assessments such as the Bayley Scales of Infant and Toddler Development are the gold standard, they are time-consuming and expensive, need the physical attendance of both the child and caregivers, and “thus may not be feasible in resource-limited settings or in pandemic conditions.”
According to Barbara J. Howard, MD, commenting on a recent update to the Center for Disease Control and Prevention’s developmental milestones guide, Learn the Signs. Act Early, fewer than 25% of children with delays or disabilities receive intervention before age 3 and most with emotional, behavioral, and developmental condition, other than autism spectrum disorder receive no intervention before age 5.
The ASQ
As an accessible alternative, the ASQ consists of questions on communication (language), gross-motor, fine-motor, problem-solving (cognitive), and personal-adaptive skills. The survey requires only 10-15 minutes, is relatively inexpensive, and also establishes a sense of parental involvement, the authors noted.
“Based on the generally accepted interpretation of LR [likelihood ratio] values, if a child passes ASQ-2SD, there is a moderate probability that the child does not have severe delay,” the investigators concluded.
The analysis
The final meta-analysis reviewed 36 eligible ASQ studies published from 1997 to 2022. Looking at the four indicators of pooled sensitivity, specificity, and positive and negative likelihood ratios, the following respective predictive values emerged for scores of more than 2 SDs below the mean across several domains: sensitivity of 0.77 (95% confidence interval, 0.64-0.86), specificity of 0.81 (95% CI 0.75-0.86), positive likelihood ratio of 4.10 (95% CI 3.17-5.30), and a negative likelihood ratio of 0.28 (95% CI, 0.18-0.44)
They cautioned, however, that the certainty of evidence from the reviewed studies was low or very low and given the small sample sizes for comparing domains, clinicians should be circumspect in interpreting the results.
An initial step
Commenting on the paper but not involved in it, David G. Fagan, MD, vice chairman of pediatric ambulatory administration in the department of pediatrics at Cohen Children’s Medical Center, New York, agreed that screening tools such as the ASQ have a place in clinical practice. “However, the purpose of a screening tool is not to make the diagnosis but to identify children at risk for developmental delays,” he said in an interview. “The meta-analysis highlights the fact that no screening is 100% accurate and that results need to be interpreted in context.
“Before screening tools were widely used, pediatricians trusted their gut,” Dr. Fagan continued. “‘I know it when I see it,’ which obviously resulted in tremendous variability based on experience.”
He added that, even if a child passes this validated questionnaire, any concern on the part of a parent or pediatrician about developmental delay should be addressed with further assessment.
The future
According to Dr. Rao, clinicians should continue to screen for developmental delays in young children using the ASQ. “Given the long wait times to see a developmental pediatrician or a clinical psychologist, a screening tool such as ASQ will enable appropriate triaging.”
Going forward, however, studies should evaluate this questionnaire separately for different age groups such as less than 12 months, 12-23 months, and at least 24 months. They should also be prospective in design and entail a low risk of bias, as well as report raw numbers for true and false positives and negatives. “Even if they use their own cutoff ASQ scores, they should also give results for the conventional cutoff scores to enable comparison with other studies,” the authors wrote.
The authors disclosed no specific funding for this study and no competing interests. Dr. Fagan disclosed no competing interests with regard to his comments.
The commonly used but sometimes debated Ages and Stages Questionnaire (ASQ), has modest utility for identifying developmental delays in young children, an Australian review and meta-analysis found.
On this easily administered parent-completed screening tool, scores of more than 2 standard deviations below the mean in more than one of five domains had moderate sensitivity and specificity to predict any delay, severe delay, motor delay, and cognitive delay, according to neonatologist Shripada Rao, PhD, a clinical associate professor in the neonatal intensive care unit at Perth Hospital and the University of Western Australia, also in Perth, and colleagues.
If a child of 12-60 months passes all ASQ domains, there is a moderate probability that child does not have severe developmental delay, the researchers concluded. If a child in that age range fails the motor or cognitive domain, there is a moderate probability that some motor or cognitive delay is present. The authors say the tool may work best as a screening test to identify children in need of more formal assessment.
“Our meta-analysis found that ASQ was somewhat more predictive in older children (older than 24 months), compared with younger age groups of 12-24 months,” Dr. Rao said in an interview. “However, the sample size for these comparisons was too small to reach definite conclusions, and we have called for future studies to evaluate ASQ separately for different age groups.”
Early identification of developmental delay in children is essential to enable timely intervention,” Dr. Rao and associates wrote in JAMA Pediatrics.
While formal assessments such as the Bayley Scales of Infant and Toddler Development are the gold standard, they are time-consuming and expensive, need the physical attendance of both the child and caregivers, and “thus may not be feasible in resource-limited settings or in pandemic conditions.”
According to Barbara J. Howard, MD, commenting on a recent update to the Center for Disease Control and Prevention’s developmental milestones guide, Learn the Signs. Act Early, fewer than 25% of children with delays or disabilities receive intervention before age 3 and most with emotional, behavioral, and developmental condition, other than autism spectrum disorder receive no intervention before age 5.
The ASQ
As an accessible alternative, the ASQ consists of questions on communication (language), gross-motor, fine-motor, problem-solving (cognitive), and personal-adaptive skills. The survey requires only 10-15 minutes, is relatively inexpensive, and also establishes a sense of parental involvement, the authors noted.
“Based on the generally accepted interpretation of LR [likelihood ratio] values, if a child passes ASQ-2SD, there is a moderate probability that the child does not have severe delay,” the investigators concluded.
The analysis
The final meta-analysis reviewed 36 eligible ASQ studies published from 1997 to 2022. Looking at the four indicators of pooled sensitivity, specificity, and positive and negative likelihood ratios, the following respective predictive values emerged for scores of more than 2 SDs below the mean across several domains: sensitivity of 0.77 (95% confidence interval, 0.64-0.86), specificity of 0.81 (95% CI 0.75-0.86), positive likelihood ratio of 4.10 (95% CI 3.17-5.30), and a negative likelihood ratio of 0.28 (95% CI, 0.18-0.44)
They cautioned, however, that the certainty of evidence from the reviewed studies was low or very low and given the small sample sizes for comparing domains, clinicians should be circumspect in interpreting the results.
An initial step
Commenting on the paper but not involved in it, David G. Fagan, MD, vice chairman of pediatric ambulatory administration in the department of pediatrics at Cohen Children’s Medical Center, New York, agreed that screening tools such as the ASQ have a place in clinical practice. “However, the purpose of a screening tool is not to make the diagnosis but to identify children at risk for developmental delays,” he said in an interview. “The meta-analysis highlights the fact that no screening is 100% accurate and that results need to be interpreted in context.
“Before screening tools were widely used, pediatricians trusted their gut,” Dr. Fagan continued. “‘I know it when I see it,’ which obviously resulted in tremendous variability based on experience.”
He added that, even if a child passes this validated questionnaire, any concern on the part of a parent or pediatrician about developmental delay should be addressed with further assessment.
The future
According to Dr. Rao, clinicians should continue to screen for developmental delays in young children using the ASQ. “Given the long wait times to see a developmental pediatrician or a clinical psychologist, a screening tool such as ASQ will enable appropriate triaging.”
Going forward, however, studies should evaluate this questionnaire separately for different age groups such as less than 12 months, 12-23 months, and at least 24 months. They should also be prospective in design and entail a low risk of bias, as well as report raw numbers for true and false positives and negatives. “Even if they use their own cutoff ASQ scores, they should also give results for the conventional cutoff scores to enable comparison with other studies,” the authors wrote.
The authors disclosed no specific funding for this study and no competing interests. Dr. Fagan disclosed no competing interests with regard to his comments.
The commonly used but sometimes debated Ages and Stages Questionnaire (ASQ), has modest utility for identifying developmental delays in young children, an Australian review and meta-analysis found.
On this easily administered parent-completed screening tool, scores of more than 2 standard deviations below the mean in more than one of five domains had moderate sensitivity and specificity to predict any delay, severe delay, motor delay, and cognitive delay, according to neonatologist Shripada Rao, PhD, a clinical associate professor in the neonatal intensive care unit at Perth Hospital and the University of Western Australia, also in Perth, and colleagues.
If a child of 12-60 months passes all ASQ domains, there is a moderate probability that child does not have severe developmental delay, the researchers concluded. If a child in that age range fails the motor or cognitive domain, there is a moderate probability that some motor or cognitive delay is present. The authors say the tool may work best as a screening test to identify children in need of more formal assessment.
“Our meta-analysis found that ASQ was somewhat more predictive in older children (older than 24 months), compared with younger age groups of 12-24 months,” Dr. Rao said in an interview. “However, the sample size for these comparisons was too small to reach definite conclusions, and we have called for future studies to evaluate ASQ separately for different age groups.”
Early identification of developmental delay in children is essential to enable timely intervention,” Dr. Rao and associates wrote in JAMA Pediatrics.
While formal assessments such as the Bayley Scales of Infant and Toddler Development are the gold standard, they are time-consuming and expensive, need the physical attendance of both the child and caregivers, and “thus may not be feasible in resource-limited settings or in pandemic conditions.”
According to Barbara J. Howard, MD, commenting on a recent update to the Center for Disease Control and Prevention’s developmental milestones guide, Learn the Signs. Act Early, fewer than 25% of children with delays or disabilities receive intervention before age 3 and most with emotional, behavioral, and developmental condition, other than autism spectrum disorder receive no intervention before age 5.
The ASQ
As an accessible alternative, the ASQ consists of questions on communication (language), gross-motor, fine-motor, problem-solving (cognitive), and personal-adaptive skills. The survey requires only 10-15 minutes, is relatively inexpensive, and also establishes a sense of parental involvement, the authors noted.
“Based on the generally accepted interpretation of LR [likelihood ratio] values, if a child passes ASQ-2SD, there is a moderate probability that the child does not have severe delay,” the investigators concluded.
The analysis
The final meta-analysis reviewed 36 eligible ASQ studies published from 1997 to 2022. Looking at the four indicators of pooled sensitivity, specificity, and positive and negative likelihood ratios, the following respective predictive values emerged for scores of more than 2 SDs below the mean across several domains: sensitivity of 0.77 (95% confidence interval, 0.64-0.86), specificity of 0.81 (95% CI 0.75-0.86), positive likelihood ratio of 4.10 (95% CI 3.17-5.30), and a negative likelihood ratio of 0.28 (95% CI, 0.18-0.44)
They cautioned, however, that the certainty of evidence from the reviewed studies was low or very low and given the small sample sizes for comparing domains, clinicians should be circumspect in interpreting the results.
An initial step
Commenting on the paper but not involved in it, David G. Fagan, MD, vice chairman of pediatric ambulatory administration in the department of pediatrics at Cohen Children’s Medical Center, New York, agreed that screening tools such as the ASQ have a place in clinical practice. “However, the purpose of a screening tool is not to make the diagnosis but to identify children at risk for developmental delays,” he said in an interview. “The meta-analysis highlights the fact that no screening is 100% accurate and that results need to be interpreted in context.
“Before screening tools were widely used, pediatricians trusted their gut,” Dr. Fagan continued. “‘I know it when I see it,’ which obviously resulted in tremendous variability based on experience.”
He added that, even if a child passes this validated questionnaire, any concern on the part of a parent or pediatrician about developmental delay should be addressed with further assessment.
The future
According to Dr. Rao, clinicians should continue to screen for developmental delays in young children using the ASQ. “Given the long wait times to see a developmental pediatrician or a clinical psychologist, a screening tool such as ASQ will enable appropriate triaging.”
Going forward, however, studies should evaluate this questionnaire separately for different age groups such as less than 12 months, 12-23 months, and at least 24 months. They should also be prospective in design and entail a low risk of bias, as well as report raw numbers for true and false positives and negatives. “Even if they use their own cutoff ASQ scores, they should also give results for the conventional cutoff scores to enable comparison with other studies,” the authors wrote.
The authors disclosed no specific funding for this study and no competing interests. Dr. Fagan disclosed no competing interests with regard to his comments.
FROM JAMA PEDIATRICS
`Wonder Woman’ launches myelofibrosis research foundation
When Lynda Carter talks about her late husband Robert Altman, you can sense right away that this was a love affair for the ages.
“As I’ve often said, if you were a friend of Robert’s, you were one of the luckiest people in the world,” said Ms. Carter, the singer-songwriter and actor best known for her role as Wonder Woman in the 1970s TV series, who married Mr. Altman, an attorney, in 1984.
For Ms. Carter, Mr. Altman, and their children, Jessica and James, everything changed in 2017, when Mr. Altman was diagnosed with myelofibrosis, a rare bone marrow disorder – about one case is reported per 100,000 Americans each year – that was found during routine blood work.
“Robert was never sick a day in his life,” she said in an interview. “He skied and swam, and in many ways we were in the prime of our lives together. When he was initially diagnosed, we weren’t even clear what he had. The buzzword was that he had a rare blood disorder, not cancer.”
The family was told to wait and see if the disease would get worse, which it did, unfortunately, at the exact time COVID-19 hit.
This condition can progress from myelofibrosis to secondary acute myeloid leukemia, a rare blood cancer, said Michael Caligiuri, MD, a leading researcher in immunology, lymphoma, and leukemia and president of City of Hope National Medical Center, Duarte, Calif., one of the largest cancer research and treatment organizations in the United States.
“This disease is chronic and slow changing, but when it progresses more acutely to a form of leukemia, it can advance rapidly,” he said.
At the acute phase, there’s not much that can be done for the patient.
“This becomes very much a life-and-death situation,” he said. “You want to hope for the best, but there needs to be an expectation of the worst in terms of trying to prepare the patient and the family for what may come so that they can start to psychologically and legally put the person’s life in order.”
Despite every effort, Mr. Altman died in February 2021 at the age of 73.
The goal: To speed up critical research that will improve early detection and survival for this hard-to-treat blood cancer.
“I’m excited to be a part of this team and to know that I may play a small part in helping other families facing this same diagnosis,” Ms. Carter said. “It’s thrilling seeing the progress these scientists are making, from genomic research into a universe of trillions of codes that might actually become a drug therapy someday.”
With the creation of the foundation, there will be funding to develop better diagnostics and better treatments.
“In many instances, this research will shed light on other related disorders, too,” said Dr. Caligiuri. “Cancer is a disease of the genes, and in most cases, we’re not inheriting from our mothers or fathers, but the DNA gets switched around in one of the trillion cells in our body, the way a word is misspelled.”
What happens next is that the cell doesn’t die.
“Instead, it undergoes a nuclear reaction and grows and grows,” he said. “In this case, the first evidence of a problem was myelofibrosis. That ticking time bomb continued until it exploded into leukemia.”
Dr. Caligiuri said the goal of their research will be to develop a device that can rearrange that DNA or block the DNA changes so the disease doesn’t progress to leukemia or, if it does, so “that we can turn it into a chronic condition, not an acute one that’s life-threatening.”
For Ms. Carter, this foundation is one very heartfelt way that she can honor her husband’s legacy.
“When I lost Robert, I was left with so many questions,” she said. “I wanted to understand why rare cancers are so difficult to treat and what research or treatment advances were being made to change that. Robert was never one for self-aggrandizement, but I think he would like this. I think Robert would really be touched by this.”
A version of this article first appeared on WebMD.com.
When Lynda Carter talks about her late husband Robert Altman, you can sense right away that this was a love affair for the ages.
“As I’ve often said, if you were a friend of Robert’s, you were one of the luckiest people in the world,” said Ms. Carter, the singer-songwriter and actor best known for her role as Wonder Woman in the 1970s TV series, who married Mr. Altman, an attorney, in 1984.
For Ms. Carter, Mr. Altman, and their children, Jessica and James, everything changed in 2017, when Mr. Altman was diagnosed with myelofibrosis, a rare bone marrow disorder – about one case is reported per 100,000 Americans each year – that was found during routine blood work.
“Robert was never sick a day in his life,” she said in an interview. “He skied and swam, and in many ways we were in the prime of our lives together. When he was initially diagnosed, we weren’t even clear what he had. The buzzword was that he had a rare blood disorder, not cancer.”
The family was told to wait and see if the disease would get worse, which it did, unfortunately, at the exact time COVID-19 hit.
This condition can progress from myelofibrosis to secondary acute myeloid leukemia, a rare blood cancer, said Michael Caligiuri, MD, a leading researcher in immunology, lymphoma, and leukemia and president of City of Hope National Medical Center, Duarte, Calif., one of the largest cancer research and treatment organizations in the United States.
“This disease is chronic and slow changing, but when it progresses more acutely to a form of leukemia, it can advance rapidly,” he said.
At the acute phase, there’s not much that can be done for the patient.
“This becomes very much a life-and-death situation,” he said. “You want to hope for the best, but there needs to be an expectation of the worst in terms of trying to prepare the patient and the family for what may come so that they can start to psychologically and legally put the person’s life in order.”
Despite every effort, Mr. Altman died in February 2021 at the age of 73.
The goal: To speed up critical research that will improve early detection and survival for this hard-to-treat blood cancer.
“I’m excited to be a part of this team and to know that I may play a small part in helping other families facing this same diagnosis,” Ms. Carter said. “It’s thrilling seeing the progress these scientists are making, from genomic research into a universe of trillions of codes that might actually become a drug therapy someday.”
With the creation of the foundation, there will be funding to develop better diagnostics and better treatments.
“In many instances, this research will shed light on other related disorders, too,” said Dr. Caligiuri. “Cancer is a disease of the genes, and in most cases, we’re not inheriting from our mothers or fathers, but the DNA gets switched around in one of the trillion cells in our body, the way a word is misspelled.”
What happens next is that the cell doesn’t die.
“Instead, it undergoes a nuclear reaction and grows and grows,” he said. “In this case, the first evidence of a problem was myelofibrosis. That ticking time bomb continued until it exploded into leukemia.”
Dr. Caligiuri said the goal of their research will be to develop a device that can rearrange that DNA or block the DNA changes so the disease doesn’t progress to leukemia or, if it does, so “that we can turn it into a chronic condition, not an acute one that’s life-threatening.”
For Ms. Carter, this foundation is one very heartfelt way that she can honor her husband’s legacy.
“When I lost Robert, I was left with so many questions,” she said. “I wanted to understand why rare cancers are so difficult to treat and what research or treatment advances were being made to change that. Robert was never one for self-aggrandizement, but I think he would like this. I think Robert would really be touched by this.”
A version of this article first appeared on WebMD.com.
When Lynda Carter talks about her late husband Robert Altman, you can sense right away that this was a love affair for the ages.
“As I’ve often said, if you were a friend of Robert’s, you were one of the luckiest people in the world,” said Ms. Carter, the singer-songwriter and actor best known for her role as Wonder Woman in the 1970s TV series, who married Mr. Altman, an attorney, in 1984.
For Ms. Carter, Mr. Altman, and their children, Jessica and James, everything changed in 2017, when Mr. Altman was diagnosed with myelofibrosis, a rare bone marrow disorder – about one case is reported per 100,000 Americans each year – that was found during routine blood work.
“Robert was never sick a day in his life,” she said in an interview. “He skied and swam, and in many ways we were in the prime of our lives together. When he was initially diagnosed, we weren’t even clear what he had. The buzzword was that he had a rare blood disorder, not cancer.”
The family was told to wait and see if the disease would get worse, which it did, unfortunately, at the exact time COVID-19 hit.
This condition can progress from myelofibrosis to secondary acute myeloid leukemia, a rare blood cancer, said Michael Caligiuri, MD, a leading researcher in immunology, lymphoma, and leukemia and president of City of Hope National Medical Center, Duarte, Calif., one of the largest cancer research and treatment organizations in the United States.
“This disease is chronic and slow changing, but when it progresses more acutely to a form of leukemia, it can advance rapidly,” he said.
At the acute phase, there’s not much that can be done for the patient.
“This becomes very much a life-and-death situation,” he said. “You want to hope for the best, but there needs to be an expectation of the worst in terms of trying to prepare the patient and the family for what may come so that they can start to psychologically and legally put the person’s life in order.”
Despite every effort, Mr. Altman died in February 2021 at the age of 73.
The goal: To speed up critical research that will improve early detection and survival for this hard-to-treat blood cancer.
“I’m excited to be a part of this team and to know that I may play a small part in helping other families facing this same diagnosis,” Ms. Carter said. “It’s thrilling seeing the progress these scientists are making, from genomic research into a universe of trillions of codes that might actually become a drug therapy someday.”
With the creation of the foundation, there will be funding to develop better diagnostics and better treatments.
“In many instances, this research will shed light on other related disorders, too,” said Dr. Caligiuri. “Cancer is a disease of the genes, and in most cases, we’re not inheriting from our mothers or fathers, but the DNA gets switched around in one of the trillion cells in our body, the way a word is misspelled.”
What happens next is that the cell doesn’t die.
“Instead, it undergoes a nuclear reaction and grows and grows,” he said. “In this case, the first evidence of a problem was myelofibrosis. That ticking time bomb continued until it exploded into leukemia.”
Dr. Caligiuri said the goal of their research will be to develop a device that can rearrange that DNA or block the DNA changes so the disease doesn’t progress to leukemia or, if it does, so “that we can turn it into a chronic condition, not an acute one that’s life-threatening.”
For Ms. Carter, this foundation is one very heartfelt way that she can honor her husband’s legacy.
“When I lost Robert, I was left with so many questions,” she said. “I wanted to understand why rare cancers are so difficult to treat and what research or treatment advances were being made to change that. Robert was never one for self-aggrandizement, but I think he would like this. I think Robert would really be touched by this.”
A version of this article first appeared on WebMD.com.
PARADISE-MI results obscured as post hoc analysis finds flaws
A post hoc analysis of the PARADISE-MI trial, although not intended to alter the conclusions generated by the published data, suggests that clinically relevant benefits were obscured, providing the basis for recommending different analyses for future studies that are more suited to capture the most clinically significant endpoints.
“What these data show us is that we need clinical trial designs moving towards more pragmatic information that better reflect clinical practice,” reported Otavio Berwanger, MD, PhD, director of the Academic Research Organization at Hospital Israelita Albert Einstein, São Paulo, Brazil.
The reevaluation of the PARADISE-MI data, presented at the annual congress of the European Society of Cardiology in Barcelona, was based on a win ratio analysis and on the inclusion of investigator-reported endpoints, not just adjudicated events. Both appear to reveal clinically meaningful benefits not reflected in the published study, according to Dr. Berwanger.
In PARADISE-MI, which was published in the New England Journal of Medicine last year, more than 5,500 patients were randomized to the angiotensin receptor neprilysin inhibitor (ARNI) sacubitril/valsartan or the ACE inhibitor ramipril after a myocardial infarction. A reduced left ventricular ejection fraction (LVEF), pulmonary congestion, or both were required for enrollment.
For the primary composite outcomes of death from cardiovascular (CV) causes or incident heart failure, the ARNI had a 10% numerical advantage, but it did not reach statistical significance (hazard ratio [HR], 0.90; P = .17).
“PARADISE-MI was a neutral trial. This post hoc analysis will not change that result,” Dr. Berwanger emphasized. However, the post hoc analysis does provide a basis for exploring why conventional trial designs might not be providing answers that are relevant and helpful for clinical practice.
New analysis provides positive trial result
When the data from PARADISE-MI are reevaluated in a hierarchical win ratio analysis with CV death serving as the most severe and important outcome, the principal conclusion changes. Whether events are reevaluated in this format by the clinical events committee (CEC) or by investigators, there is a greater number of total wins than total losses for the ARNI. Combined, sacubitril/valsartan was associated with a win ratio of 1.17 (95% confidence interval, 1.03-1.33; P = 0.015) over ramipril.
Using a sports analogy, Dr. Berwanger explained that the win ratio analysis divides the total number of wins to the total number of losses to provide a much more clinically relevant approach to keeping score. It also used a hierarchical analysis so that the most serious and important events are considered first.
In addition to CV death, this analysis included first hospitalization for heart failure and first outpatient heart failure events. CEC-defined events and events reported by investigators were evaluated separately.
The ARNI had more wins than losses in every category for all outcomes, whether CEC adjudicated or investigator reported, but most of this benefit was generated by the endpoint of CEC-adjudicated CV deaths. This accounted for 36.9% of all events (investigator-documented CV death accounted for 0.7%). This is important because PARADISE-MI, like many standard trials, was conducted on a time-to-primary event basis.
“In this type of analysis, the first event is what counts. Usually time-to-first-event analyses are dominated by nonfatal events,” Dr. Berwanger explained. He believes that placing more weight on the most serious events results in an emphasis on what outcomes are of greatest clinical interest.
In addition, Dr. Berwanger argued that it is important to consider investigator-reported events, not just CEC-adjudicated events. While adjudicated events improve the rigor of the data, Dr. Berwanger suggested it omits outcomes with which clinicians are most concerned.
Investigator, adjudicated outcomes differ
Again, using PARADISE-MI as an example, he reevaluated the primary outcome based on investigator reports. When investigator-reported events are included, the number of events increased in both the ARNI (443 vs. 338) and ramipril (516 vs. 373) arms, but the advantage of the ARNI over the ACE inhibitors now reached statistical significance (HR, 0.85; P = .01).
“The data suggest that maybe we should find definitions for adjudication that are closer to clinical judgment in the real world and clinical practice,” Dr. Berwanger said.
One possible explanation for the neutral result in PARADISE-MI is that benefit of an ARNI over an ACE inhibitor would only be expected in those at risk for progressive left ventricular dysfunction, and it is likely that a substantial proportion of patients enrolled in this trial recovered, according to Johann Bauersachs, MD, PhD, professor and head of cardiology at Hannover (Germany) Medical School.
“You cannot predict which patients with reduced LV function following an MI will go on to chronic remodeling and which will recover,” said Dr. Bauersachs, who was an ESC-invited discussant of Dr. Berwanger’s post hoc analysis.
He agreed that Dr. Berwanger has raised several important issues in standard trial design that might have prevented PARADISE-MI from showing a benefit from an ARNI, but he pointed out that there are other potential issues, such as the low use of mineralocorticoid antagonists in PARADISE-MI, that may have skewed results.
However, he agreed generally with the premise that there is a need for trial design likely to generate more clinically useful information.
“We have now seen the win-ratio approach used in several studies,” said Dr. Bauersachs, citing in particular the EMPULSE trial presented at the 2022 meeting of the American College of Cardiology. “It is a very useful tool, and I think we will be seeing it used more in the future.”
However, he indicated that the issues raised by Dr. Berwanger are not necessarily easily resolved. Dr. Bauersachs endorsed the effort to consider trial designs that generate data that are more immediately clinically applicable but suggested that different types of designs may be required for different types of clinical questions.
Dr. Berwanger reports financial relationships with Amgen, AstraZeneca, Bayer, Bristol-Myers Squibb, Pfizer, Servier, and Novartis, which provided funding for the PARADISE-MI trial. Dr. Bauersachs reports financial relationships with Amgen, AstraZeneca, Bayer, Bristol-Myers Squibb, Boehringer Ingelheim, Cardior, Corvia, CVRx, Novartis, Pfizer, Vifor, and Zoll.
A post hoc analysis of the PARADISE-MI trial, although not intended to alter the conclusions generated by the published data, suggests that clinically relevant benefits were obscured, providing the basis for recommending different analyses for future studies that are more suited to capture the most clinically significant endpoints.
“What these data show us is that we need clinical trial designs moving towards more pragmatic information that better reflect clinical practice,” reported Otavio Berwanger, MD, PhD, director of the Academic Research Organization at Hospital Israelita Albert Einstein, São Paulo, Brazil.
The reevaluation of the PARADISE-MI data, presented at the annual congress of the European Society of Cardiology in Barcelona, was based on a win ratio analysis and on the inclusion of investigator-reported endpoints, not just adjudicated events. Both appear to reveal clinically meaningful benefits not reflected in the published study, according to Dr. Berwanger.
In PARADISE-MI, which was published in the New England Journal of Medicine last year, more than 5,500 patients were randomized to the angiotensin receptor neprilysin inhibitor (ARNI) sacubitril/valsartan or the ACE inhibitor ramipril after a myocardial infarction. A reduced left ventricular ejection fraction (LVEF), pulmonary congestion, or both were required for enrollment.
For the primary composite outcomes of death from cardiovascular (CV) causes or incident heart failure, the ARNI had a 10% numerical advantage, but it did not reach statistical significance (hazard ratio [HR], 0.90; P = .17).
“PARADISE-MI was a neutral trial. This post hoc analysis will not change that result,” Dr. Berwanger emphasized. However, the post hoc analysis does provide a basis for exploring why conventional trial designs might not be providing answers that are relevant and helpful for clinical practice.
New analysis provides positive trial result
When the data from PARADISE-MI are reevaluated in a hierarchical win ratio analysis with CV death serving as the most severe and important outcome, the principal conclusion changes. Whether events are reevaluated in this format by the clinical events committee (CEC) or by investigators, there is a greater number of total wins than total losses for the ARNI. Combined, sacubitril/valsartan was associated with a win ratio of 1.17 (95% confidence interval, 1.03-1.33; P = 0.015) over ramipril.
Using a sports analogy, Dr. Berwanger explained that the win ratio analysis divides the total number of wins to the total number of losses to provide a much more clinically relevant approach to keeping score. It also used a hierarchical analysis so that the most serious and important events are considered first.
In addition to CV death, this analysis included first hospitalization for heart failure and first outpatient heart failure events. CEC-defined events and events reported by investigators were evaluated separately.
The ARNI had more wins than losses in every category for all outcomes, whether CEC adjudicated or investigator reported, but most of this benefit was generated by the endpoint of CEC-adjudicated CV deaths. This accounted for 36.9% of all events (investigator-documented CV death accounted for 0.7%). This is important because PARADISE-MI, like many standard trials, was conducted on a time-to-primary event basis.
“In this type of analysis, the first event is what counts. Usually time-to-first-event analyses are dominated by nonfatal events,” Dr. Berwanger explained. He believes that placing more weight on the most serious events results in an emphasis on what outcomes are of greatest clinical interest.
In addition, Dr. Berwanger argued that it is important to consider investigator-reported events, not just CEC-adjudicated events. While adjudicated events improve the rigor of the data, Dr. Berwanger suggested it omits outcomes with which clinicians are most concerned.
Investigator, adjudicated outcomes differ
Again, using PARADISE-MI as an example, he reevaluated the primary outcome based on investigator reports. When investigator-reported events are included, the number of events increased in both the ARNI (443 vs. 338) and ramipril (516 vs. 373) arms, but the advantage of the ARNI over the ACE inhibitors now reached statistical significance (HR, 0.85; P = .01).
“The data suggest that maybe we should find definitions for adjudication that are closer to clinical judgment in the real world and clinical practice,” Dr. Berwanger said.
One possible explanation for the neutral result in PARADISE-MI is that benefit of an ARNI over an ACE inhibitor would only be expected in those at risk for progressive left ventricular dysfunction, and it is likely that a substantial proportion of patients enrolled in this trial recovered, according to Johann Bauersachs, MD, PhD, professor and head of cardiology at Hannover (Germany) Medical School.
“You cannot predict which patients with reduced LV function following an MI will go on to chronic remodeling and which will recover,” said Dr. Bauersachs, who was an ESC-invited discussant of Dr. Berwanger’s post hoc analysis.
He agreed that Dr. Berwanger has raised several important issues in standard trial design that might have prevented PARADISE-MI from showing a benefit from an ARNI, but he pointed out that there are other potential issues, such as the low use of mineralocorticoid antagonists in PARADISE-MI, that may have skewed results.
However, he agreed generally with the premise that there is a need for trial design likely to generate more clinically useful information.
“We have now seen the win-ratio approach used in several studies,” said Dr. Bauersachs, citing in particular the EMPULSE trial presented at the 2022 meeting of the American College of Cardiology. “It is a very useful tool, and I think we will be seeing it used more in the future.”
However, he indicated that the issues raised by Dr. Berwanger are not necessarily easily resolved. Dr. Bauersachs endorsed the effort to consider trial designs that generate data that are more immediately clinically applicable but suggested that different types of designs may be required for different types of clinical questions.
Dr. Berwanger reports financial relationships with Amgen, AstraZeneca, Bayer, Bristol-Myers Squibb, Pfizer, Servier, and Novartis, which provided funding for the PARADISE-MI trial. Dr. Bauersachs reports financial relationships with Amgen, AstraZeneca, Bayer, Bristol-Myers Squibb, Boehringer Ingelheim, Cardior, Corvia, CVRx, Novartis, Pfizer, Vifor, and Zoll.
A post hoc analysis of the PARADISE-MI trial, although not intended to alter the conclusions generated by the published data, suggests that clinically relevant benefits were obscured, providing the basis for recommending different analyses for future studies that are more suited to capture the most clinically significant endpoints.
“What these data show us is that we need clinical trial designs moving towards more pragmatic information that better reflect clinical practice,” reported Otavio Berwanger, MD, PhD, director of the Academic Research Organization at Hospital Israelita Albert Einstein, São Paulo, Brazil.
The reevaluation of the PARADISE-MI data, presented at the annual congress of the European Society of Cardiology in Barcelona, was based on a win ratio analysis and on the inclusion of investigator-reported endpoints, not just adjudicated events. Both appear to reveal clinically meaningful benefits not reflected in the published study, according to Dr. Berwanger.
In PARADISE-MI, which was published in the New England Journal of Medicine last year, more than 5,500 patients were randomized to the angiotensin receptor neprilysin inhibitor (ARNI) sacubitril/valsartan or the ACE inhibitor ramipril after a myocardial infarction. A reduced left ventricular ejection fraction (LVEF), pulmonary congestion, or both were required for enrollment.
For the primary composite outcomes of death from cardiovascular (CV) causes or incident heart failure, the ARNI had a 10% numerical advantage, but it did not reach statistical significance (hazard ratio [HR], 0.90; P = .17).
“PARADISE-MI was a neutral trial. This post hoc analysis will not change that result,” Dr. Berwanger emphasized. However, the post hoc analysis does provide a basis for exploring why conventional trial designs might not be providing answers that are relevant and helpful for clinical practice.
New analysis provides positive trial result
When the data from PARADISE-MI are reevaluated in a hierarchical win ratio analysis with CV death serving as the most severe and important outcome, the principal conclusion changes. Whether events are reevaluated in this format by the clinical events committee (CEC) or by investigators, there is a greater number of total wins than total losses for the ARNI. Combined, sacubitril/valsartan was associated with a win ratio of 1.17 (95% confidence interval, 1.03-1.33; P = 0.015) over ramipril.
Using a sports analogy, Dr. Berwanger explained that the win ratio analysis divides the total number of wins to the total number of losses to provide a much more clinically relevant approach to keeping score. It also used a hierarchical analysis so that the most serious and important events are considered first.
In addition to CV death, this analysis included first hospitalization for heart failure and first outpatient heart failure events. CEC-defined events and events reported by investigators were evaluated separately.
The ARNI had more wins than losses in every category for all outcomes, whether CEC adjudicated or investigator reported, but most of this benefit was generated by the endpoint of CEC-adjudicated CV deaths. This accounted for 36.9% of all events (investigator-documented CV death accounted for 0.7%). This is important because PARADISE-MI, like many standard trials, was conducted on a time-to-primary event basis.
“In this type of analysis, the first event is what counts. Usually time-to-first-event analyses are dominated by nonfatal events,” Dr. Berwanger explained. He believes that placing more weight on the most serious events results in an emphasis on what outcomes are of greatest clinical interest.
In addition, Dr. Berwanger argued that it is important to consider investigator-reported events, not just CEC-adjudicated events. While adjudicated events improve the rigor of the data, Dr. Berwanger suggested it omits outcomes with which clinicians are most concerned.
Investigator, adjudicated outcomes differ
Again, using PARADISE-MI as an example, he reevaluated the primary outcome based on investigator reports. When investigator-reported events are included, the number of events increased in both the ARNI (443 vs. 338) and ramipril (516 vs. 373) arms, but the advantage of the ARNI over the ACE inhibitors now reached statistical significance (HR, 0.85; P = .01).
“The data suggest that maybe we should find definitions for adjudication that are closer to clinical judgment in the real world and clinical practice,” Dr. Berwanger said.
One possible explanation for the neutral result in PARADISE-MI is that benefit of an ARNI over an ACE inhibitor would only be expected in those at risk for progressive left ventricular dysfunction, and it is likely that a substantial proportion of patients enrolled in this trial recovered, according to Johann Bauersachs, MD, PhD, professor and head of cardiology at Hannover (Germany) Medical School.
“You cannot predict which patients with reduced LV function following an MI will go on to chronic remodeling and which will recover,” said Dr. Bauersachs, who was an ESC-invited discussant of Dr. Berwanger’s post hoc analysis.
He agreed that Dr. Berwanger has raised several important issues in standard trial design that might have prevented PARADISE-MI from showing a benefit from an ARNI, but he pointed out that there are other potential issues, such as the low use of mineralocorticoid antagonists in PARADISE-MI, that may have skewed results.
However, he agreed generally with the premise that there is a need for trial design likely to generate more clinically useful information.
“We have now seen the win-ratio approach used in several studies,” said Dr. Bauersachs, citing in particular the EMPULSE trial presented at the 2022 meeting of the American College of Cardiology. “It is a very useful tool, and I think we will be seeing it used more in the future.”
However, he indicated that the issues raised by Dr. Berwanger are not necessarily easily resolved. Dr. Bauersachs endorsed the effort to consider trial designs that generate data that are more immediately clinically applicable but suggested that different types of designs may be required for different types of clinical questions.
Dr. Berwanger reports financial relationships with Amgen, AstraZeneca, Bayer, Bristol-Myers Squibb, Pfizer, Servier, and Novartis, which provided funding for the PARADISE-MI trial. Dr. Bauersachs reports financial relationships with Amgen, AstraZeneca, Bayer, Bristol-Myers Squibb, Boehringer Ingelheim, Cardior, Corvia, CVRx, Novartis, Pfizer, Vifor, and Zoll.
FROM ESC CONGRESS 2022
Fish oil pills do not reduce fractures in healthy seniors: VITAL
Omega-3 supplements did not reduce fractures during a median 5.3-year follow-up in the more than 25,000 generally healthy men and women (≥ age 50 and ≥ age 55, respectively) in the Vitamin D and Omega-3 Trial (VITAL).
The large randomized controlled trial tested whether omega-3 fatty acid or vitamin D supplements prevented cardiovascular disease or cancer in a representative sample of midlife and older adults from 50 U.S. states – which they did not. In a further analysis of VITAL, vitamin D supplements (cholecalciferol, 2,000 IU/day) did not lower the risk of incident total, nonvertebral, and hip fractures, compared with placebo.
Now this new analysis shows that omega-3 fatty acid supplements (1 g/day of fish oil) did not reduce the risk of such fractures in the VITAL population either. Meryl S. LeBoff, MD, presented the latest findings during an oral session at the annual meeting of the American Society for Bone and Mineral Research.
“In this, the largest randomized controlled trial in the world, we did not find an effect of omega-3 fatty acid supplements on fractures,” Dr. LeBoff, from Brigham and Women’s Hospital and Harvard Medical School, both in Boston, told this news organization.
The current analysis did “unexpectedly” show that among participants who received the omega-3 fatty acid supplements, there was an increase in fractures in men, and fracture risk was higher in people with a normal or low body mass index and lower in people with higher BMI.
However, these subgroup findings need to be interpreted with caution and may be caused by chance, Dr. LeBoff warned. The researchers will be investigating these findings in further analyses.
Should patients take omega-3 supplements or not?
Asked whether, in the meantime, patients should start or keep taking fish oil supplements for possible health benefits, she noted that certain individuals might benefit.
For example, in VITAL, participants who ate less than 1.5 servings of fish per week and received omega-3 fatty acid supplements had a decrease in the combined cardiovascular endpoint, and Black participants who took fish oil supplements had a substantially reduced risk of the outcome, regardless of fish intake.
“I think everybody needs to review [the study findings] with clinicians and make a decision in terms of what would be best for them,” she said.
Session comoderator Bente Langdahl, MD, PhD, commented that “many people take omega-3 because they think it will help” knee, hip, or other joint pain.
Perhaps men are more prone to joint pain because of osteoarthritis and the supplements lessen the pain, so these men became more physically active and more prone to fractures, she speculated.
The current study shows that, “so far, we haven’t been able to demonstrate a reduced rate of fractures with fish oil supplements in clinical randomized trials” conducted in relatively healthy and not the oldest patients, she summarized. “We’re not talking about 80-year-olds.”
In this “well-conducted study, they were not able to see any difference” with omega-3 fatty acid supplements versus placebo, but apparently, there are no harms associated with taking these supplements, she said.
To patients who ask her about such supplements, Dr. Langdahl advised: “Try it out for 3 months. If it really helps you, if it takes away your joint pain or whatever, then that might work for you. But then remember to stop again because it might just be a temporary effect.”
Could fish oil supplements protect against fractures?
An estimated 22% of U.S. adults aged 60 and older take omega-3 fatty acid supplements, Dr. LeBoff noted.
Preclinical studies have shown that omega-3 fatty acids reduce bone resorption and have anti-inflammatory effects, but observational studies have reported conflicting findings.
The researchers conducted this ancillary study of VITAL to fill these knowledge gaps.
VITAL enrolled a national sample of 25,871 U.S. men and women, including 5,106 Black participants, with a mean age of 67 and a mean BMI of 28 kg/m2.
Importantly, participants were not recruited by low bone density, fractures, or vitamin D deficiency. Prior to entry, participants were required to stop taking omega-3 supplements and limit nonstudy vitamin D and calcium supplements.
The omega-3 fatty acid supplements used in the study contained eicosapentaenoic acid and docosahexaenoic acid in a 1.2:1 ratio.
VITAL had a 2x2 factorial design whereby 6,463 participants were randomized to receive the omega-3 fatty acid supplement and 6,474 were randomized to placebo. (Remaining participants were randomized to receive vitamin D or placebo.)
Participants in the omega-3 fatty acid and placebo groups had similar baseline characteristics. For example, about half (50.5%) were women, and on average, they ate 1.1 servings of dark-meat fish (such as salmon) per week.
Participants completed detailed questionnaires at baseline and each year.
Plasma omega-3 levels were measured at baseline and, in 1,583 participants, at 1 year of follow-up. The mean omega-3 index rose 54.7% in the omega-3 fatty acid group and changed less than 2% in the placebo group at 1 year.
Study pill adherence was 87.0% at 2 years and 85.7% at 5 years.
Fractures were self-reported on annual questionnaires and centrally adjudicated in medical record review.
No clinically meaningful effect of omega-3 fatty acids on fractures
During a median 5.3-year follow-up, researchers adjudicated 2,133 total fractures and confirmed 1,991 fractures (93%) in 1551 participants.
Incidences of total, nonvertebral, and hip fractures were similar in both groups.
Compared with placebo, omega-3 fatty acid supplements had no significant effect on risk of total fractures (hazard ratio, 1.02; 95% confidence interval, 0.92-1.13), nonvertebral fractures (HR, 1.01; 95% CI, 0.91-1.12), or hip fractures (HR, 0.89; 95% CI, 0.61-1.30), all adjusted for age, sex, and race.
The “confidence intervals were narrow, likely excluding a clinically meaningful effect,” Dr. LeBoff noted.
Among men, those who received fish oil supplements had a greater risk of fracture than those who received placebo (HR, 1.27; 95% CI, 1.07-1.51), but this result “was not corrected for multiple hypothesis testing,” Dr. LeBoff cautioned.
In the overall population, participants with a BMI less than 25 who received fish oil versus placebo had an increased risk of fracture, and those with a BMI of at least 30 who received fish oil versus placebo had a decreased risk of fracture, but the limits of the confidence intervals crossed 1.00.
After excluding digit, skull, and pathologic fractures, there was no significant reduction in total fractures (HR, 1.02; 95% CI, 0.92-1.14), nonvertebral fractures (HR, 1.02; 95% CI, 0.92-1.14), or hip fractures (HR, 0.90; 95% CI, 0.61-1.33), with omega-3 supplements versus placebo.
Similarly, there was no significant reduction in risk of major osteoporotic fractures (hip, wrist, humerus, and clinical spine fractures) or wrist fractures with omega-3 supplements versus placebo.
VITAL only studied one dose of omega-3 fatty acid supplements, and results may not be generalizable to younger adults, or older adults living in residential communities, Dr. LeBoff noted.
The study was supported by grants from the National Institute of Arthritis Musculoskeletal and Skin Diseases. VITAL was funded by the National Cancer Institute and the National Heart, Lung, and Blood Institute. Dr. LeBoff and Dr. Langdahl have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Omega-3 supplements did not reduce fractures during a median 5.3-year follow-up in the more than 25,000 generally healthy men and women (≥ age 50 and ≥ age 55, respectively) in the Vitamin D and Omega-3 Trial (VITAL).
The large randomized controlled trial tested whether omega-3 fatty acid or vitamin D supplements prevented cardiovascular disease or cancer in a representative sample of midlife and older adults from 50 U.S. states – which they did not. In a further analysis of VITAL, vitamin D supplements (cholecalciferol, 2,000 IU/day) did not lower the risk of incident total, nonvertebral, and hip fractures, compared with placebo.
Now this new analysis shows that omega-3 fatty acid supplements (1 g/day of fish oil) did not reduce the risk of such fractures in the VITAL population either. Meryl S. LeBoff, MD, presented the latest findings during an oral session at the annual meeting of the American Society for Bone and Mineral Research.
“In this, the largest randomized controlled trial in the world, we did not find an effect of omega-3 fatty acid supplements on fractures,” Dr. LeBoff, from Brigham and Women’s Hospital and Harvard Medical School, both in Boston, told this news organization.
The current analysis did “unexpectedly” show that among participants who received the omega-3 fatty acid supplements, there was an increase in fractures in men, and fracture risk was higher in people with a normal or low body mass index and lower in people with higher BMI.
However, these subgroup findings need to be interpreted with caution and may be caused by chance, Dr. LeBoff warned. The researchers will be investigating these findings in further analyses.
Should patients take omega-3 supplements or not?
Asked whether, in the meantime, patients should start or keep taking fish oil supplements for possible health benefits, she noted that certain individuals might benefit.
For example, in VITAL, participants who ate less than 1.5 servings of fish per week and received omega-3 fatty acid supplements had a decrease in the combined cardiovascular endpoint, and Black participants who took fish oil supplements had a substantially reduced risk of the outcome, regardless of fish intake.
“I think everybody needs to review [the study findings] with clinicians and make a decision in terms of what would be best for them,” she said.
Session comoderator Bente Langdahl, MD, PhD, commented that “many people take omega-3 because they think it will help” knee, hip, or other joint pain.
Perhaps men are more prone to joint pain because of osteoarthritis and the supplements lessen the pain, so these men became more physically active and more prone to fractures, she speculated.
The current study shows that, “so far, we haven’t been able to demonstrate a reduced rate of fractures with fish oil supplements in clinical randomized trials” conducted in relatively healthy and not the oldest patients, she summarized. “We’re not talking about 80-year-olds.”
In this “well-conducted study, they were not able to see any difference” with omega-3 fatty acid supplements versus placebo, but apparently, there are no harms associated with taking these supplements, she said.
To patients who ask her about such supplements, Dr. Langdahl advised: “Try it out for 3 months. If it really helps you, if it takes away your joint pain or whatever, then that might work for you. But then remember to stop again because it might just be a temporary effect.”
Could fish oil supplements protect against fractures?
An estimated 22% of U.S. adults aged 60 and older take omega-3 fatty acid supplements, Dr. LeBoff noted.
Preclinical studies have shown that omega-3 fatty acids reduce bone resorption and have anti-inflammatory effects, but observational studies have reported conflicting findings.
The researchers conducted this ancillary study of VITAL to fill these knowledge gaps.
VITAL enrolled a national sample of 25,871 U.S. men and women, including 5,106 Black participants, with a mean age of 67 and a mean BMI of 28 kg/m2.
Importantly, participants were not recruited by low bone density, fractures, or vitamin D deficiency. Prior to entry, participants were required to stop taking omega-3 supplements and limit nonstudy vitamin D and calcium supplements.
The omega-3 fatty acid supplements used in the study contained eicosapentaenoic acid and docosahexaenoic acid in a 1.2:1 ratio.
VITAL had a 2x2 factorial design whereby 6,463 participants were randomized to receive the omega-3 fatty acid supplement and 6,474 were randomized to placebo. (Remaining participants were randomized to receive vitamin D or placebo.)
Participants in the omega-3 fatty acid and placebo groups had similar baseline characteristics. For example, about half (50.5%) were women, and on average, they ate 1.1 servings of dark-meat fish (such as salmon) per week.
Participants completed detailed questionnaires at baseline and each year.
Plasma omega-3 levels were measured at baseline and, in 1,583 participants, at 1 year of follow-up. The mean omega-3 index rose 54.7% in the omega-3 fatty acid group and changed less than 2% in the placebo group at 1 year.
Study pill adherence was 87.0% at 2 years and 85.7% at 5 years.
Fractures were self-reported on annual questionnaires and centrally adjudicated in medical record review.
No clinically meaningful effect of omega-3 fatty acids on fractures
During a median 5.3-year follow-up, researchers adjudicated 2,133 total fractures and confirmed 1,991 fractures (93%) in 1551 participants.
Incidences of total, nonvertebral, and hip fractures were similar in both groups.
Compared with placebo, omega-3 fatty acid supplements had no significant effect on risk of total fractures (hazard ratio, 1.02; 95% confidence interval, 0.92-1.13), nonvertebral fractures (HR, 1.01; 95% CI, 0.91-1.12), or hip fractures (HR, 0.89; 95% CI, 0.61-1.30), all adjusted for age, sex, and race.
The “confidence intervals were narrow, likely excluding a clinically meaningful effect,” Dr. LeBoff noted.
Among men, those who received fish oil supplements had a greater risk of fracture than those who received placebo (HR, 1.27; 95% CI, 1.07-1.51), but this result “was not corrected for multiple hypothesis testing,” Dr. LeBoff cautioned.
In the overall population, participants with a BMI less than 25 who received fish oil versus placebo had an increased risk of fracture, and those with a BMI of at least 30 who received fish oil versus placebo had a decreased risk of fracture, but the limits of the confidence intervals crossed 1.00.
After excluding digit, skull, and pathologic fractures, there was no significant reduction in total fractures (HR, 1.02; 95% CI, 0.92-1.14), nonvertebral fractures (HR, 1.02; 95% CI, 0.92-1.14), or hip fractures (HR, 0.90; 95% CI, 0.61-1.33), with omega-3 supplements versus placebo.
Similarly, there was no significant reduction in risk of major osteoporotic fractures (hip, wrist, humerus, and clinical spine fractures) or wrist fractures with omega-3 supplements versus placebo.
VITAL only studied one dose of omega-3 fatty acid supplements, and results may not be generalizable to younger adults, or older adults living in residential communities, Dr. LeBoff noted.
The study was supported by grants from the National Institute of Arthritis Musculoskeletal and Skin Diseases. VITAL was funded by the National Cancer Institute and the National Heart, Lung, and Blood Institute. Dr. LeBoff and Dr. Langdahl have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Omega-3 supplements did not reduce fractures during a median 5.3-year follow-up in the more than 25,000 generally healthy men and women (≥ age 50 and ≥ age 55, respectively) in the Vitamin D and Omega-3 Trial (VITAL).
The large randomized controlled trial tested whether omega-3 fatty acid or vitamin D supplements prevented cardiovascular disease or cancer in a representative sample of midlife and older adults from 50 U.S. states – which they did not. In a further analysis of VITAL, vitamin D supplements (cholecalciferol, 2,000 IU/day) did not lower the risk of incident total, nonvertebral, and hip fractures, compared with placebo.
Now this new analysis shows that omega-3 fatty acid supplements (1 g/day of fish oil) did not reduce the risk of such fractures in the VITAL population either. Meryl S. LeBoff, MD, presented the latest findings during an oral session at the annual meeting of the American Society for Bone and Mineral Research.
“In this, the largest randomized controlled trial in the world, we did not find an effect of omega-3 fatty acid supplements on fractures,” Dr. LeBoff, from Brigham and Women’s Hospital and Harvard Medical School, both in Boston, told this news organization.
The current analysis did “unexpectedly” show that among participants who received the omega-3 fatty acid supplements, there was an increase in fractures in men, and fracture risk was higher in people with a normal or low body mass index and lower in people with higher BMI.
However, these subgroup findings need to be interpreted with caution and may be caused by chance, Dr. LeBoff warned. The researchers will be investigating these findings in further analyses.
Should patients take omega-3 supplements or not?
Asked whether, in the meantime, patients should start or keep taking fish oil supplements for possible health benefits, she noted that certain individuals might benefit.
For example, in VITAL, participants who ate less than 1.5 servings of fish per week and received omega-3 fatty acid supplements had a decrease in the combined cardiovascular endpoint, and Black participants who took fish oil supplements had a substantially reduced risk of the outcome, regardless of fish intake.
“I think everybody needs to review [the study findings] with clinicians and make a decision in terms of what would be best for them,” she said.
Session comoderator Bente Langdahl, MD, PhD, commented that “many people take omega-3 because they think it will help” knee, hip, or other joint pain.
Perhaps men are more prone to joint pain because of osteoarthritis and the supplements lessen the pain, so these men became more physically active and more prone to fractures, she speculated.
The current study shows that, “so far, we haven’t been able to demonstrate a reduced rate of fractures with fish oil supplements in clinical randomized trials” conducted in relatively healthy and not the oldest patients, she summarized. “We’re not talking about 80-year-olds.”
In this “well-conducted study, they were not able to see any difference” with omega-3 fatty acid supplements versus placebo, but apparently, there are no harms associated with taking these supplements, she said.
To patients who ask her about such supplements, Dr. Langdahl advised: “Try it out for 3 months. If it really helps you, if it takes away your joint pain or whatever, then that might work for you. But then remember to stop again because it might just be a temporary effect.”
Could fish oil supplements protect against fractures?
An estimated 22% of U.S. adults aged 60 and older take omega-3 fatty acid supplements, Dr. LeBoff noted.
Preclinical studies have shown that omega-3 fatty acids reduce bone resorption and have anti-inflammatory effects, but observational studies have reported conflicting findings.
The researchers conducted this ancillary study of VITAL to fill these knowledge gaps.
VITAL enrolled a national sample of 25,871 U.S. men and women, including 5,106 Black participants, with a mean age of 67 and a mean BMI of 28 kg/m2.
Importantly, participants were not recruited by low bone density, fractures, or vitamin D deficiency. Prior to entry, participants were required to stop taking omega-3 supplements and limit nonstudy vitamin D and calcium supplements.
The omega-3 fatty acid supplements used in the study contained eicosapentaenoic acid and docosahexaenoic acid in a 1.2:1 ratio.
VITAL had a 2x2 factorial design whereby 6,463 participants were randomized to receive the omega-3 fatty acid supplement and 6,474 were randomized to placebo. (Remaining participants were randomized to receive vitamin D or placebo.)
Participants in the omega-3 fatty acid and placebo groups had similar baseline characteristics. For example, about half (50.5%) were women, and on average, they ate 1.1 servings of dark-meat fish (such as salmon) per week.
Participants completed detailed questionnaires at baseline and each year.
Plasma omega-3 levels were measured at baseline and, in 1,583 participants, at 1 year of follow-up. The mean omega-3 index rose 54.7% in the omega-3 fatty acid group and changed less than 2% in the placebo group at 1 year.
Study pill adherence was 87.0% at 2 years and 85.7% at 5 years.
Fractures were self-reported on annual questionnaires and centrally adjudicated in medical record review.
No clinically meaningful effect of omega-3 fatty acids on fractures
During a median 5.3-year follow-up, researchers adjudicated 2,133 total fractures and confirmed 1,991 fractures (93%) in 1551 participants.
Incidences of total, nonvertebral, and hip fractures were similar in both groups.
Compared with placebo, omega-3 fatty acid supplements had no significant effect on risk of total fractures (hazard ratio, 1.02; 95% confidence interval, 0.92-1.13), nonvertebral fractures (HR, 1.01; 95% CI, 0.91-1.12), or hip fractures (HR, 0.89; 95% CI, 0.61-1.30), all adjusted for age, sex, and race.
The “confidence intervals were narrow, likely excluding a clinically meaningful effect,” Dr. LeBoff noted.
Among men, those who received fish oil supplements had a greater risk of fracture than those who received placebo (HR, 1.27; 95% CI, 1.07-1.51), but this result “was not corrected for multiple hypothesis testing,” Dr. LeBoff cautioned.
In the overall population, participants with a BMI less than 25 who received fish oil versus placebo had an increased risk of fracture, and those with a BMI of at least 30 who received fish oil versus placebo had a decreased risk of fracture, but the limits of the confidence intervals crossed 1.00.
After excluding digit, skull, and pathologic fractures, there was no significant reduction in total fractures (HR, 1.02; 95% CI, 0.92-1.14), nonvertebral fractures (HR, 1.02; 95% CI, 0.92-1.14), or hip fractures (HR, 0.90; 95% CI, 0.61-1.33), with omega-3 supplements versus placebo.
Similarly, there was no significant reduction in risk of major osteoporotic fractures (hip, wrist, humerus, and clinical spine fractures) or wrist fractures with omega-3 supplements versus placebo.
VITAL only studied one dose of omega-3 fatty acid supplements, and results may not be generalizable to younger adults, or older adults living in residential communities, Dr. LeBoff noted.
The study was supported by grants from the National Institute of Arthritis Musculoskeletal and Skin Diseases. VITAL was funded by the National Cancer Institute and the National Heart, Lung, and Blood Institute. Dr. LeBoff and Dr. Langdahl have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ASMBR 2022







