User login
Myocardial infarction in women younger than 50: Lessons to learn
Young women (under 50) are increasingly having heart attacks without doctors really knowing why. This is where the Young Women Presenting Acute Myocardial Infarction in France (WAMIF) study comes in, the results of which were presented in an e-poster at the annual congress of the European Society of Cardiology by Stéphane Manzo-Silberman, MD, Institute of Cardiology, Pitié-Salpétrière, Paris. The results (yet to be published) fight several of the preconceived ideas on the topic, Dr. Manzo-Silberman commented in an interview.
Significantly higher hospital death rates in women
“Cardiovascular disease is the main cause of death in women, killing seven times more than breast cancer,” notes Dr. Manzo-Silberman. The hospital death rate is significantly higher in women and, despite going down, is significantly higher than in men (more than double), particularly in women under 50. What’s more, in addition to the typical risk factors, women present specific risk factors related to hormone changes, high-risk inflammatory profiles, and thrombophilia.”
The WAMIF study was designed to determine the clinical, biological, and morphological features linked to hospital mortality after 12 months in women under 50. The prospective, observational study included all women in this age range from 30 sites in France between May 2017 and June 2019.
90% with retrosternal chest pain
The age of the 314 women enrolled was 44.9 years on average. Nearly two-thirds (192) presented with ST-segment elevation myocardial infarction and the other 122 without. In terms of symptoms, 91.6% of these women presented with typical chest pain, and 59.7% had related symptoms.
“With more than 90% having retrosternal pain, the idea that myocardial infarction presents with atypical symptoms in women has been widely challenged, despite the fact that more than half present with related symptoms and it isn’t known in which order these symptoms occur, Dr. Manzo-Silberman said in an interview. But what we can say is that if at any point a young woman mentions chest pain, even when occurring as part of several other symptoms, MI must be deemed a possibility until it has been ruled out.”
The risk profile revealed that 75.5% were smokers, 35% had a family history of heart disease, 33% had pregnancy complications, and 55% had recently experienced a stressful situation. The analysis also showed that cannabis use and oral contraception were primary risk factors in women younger than 35.
“With regard to risk factors, when designing this study we expected that lots of these young women would have largely atypical autoimmune conditions, with high levels of inflammation. We looked for everything, but this was not actually the case. Instead, we found very many women to have classic risk factors; three-quarters were smokers, a modifiable risk factor, which can largely be prevented. The other aspect concerns contraception, and it’s why I insist that gynecologists must be involved insofar as they must inform their patients how to manage their risk factors and tweak their contraception.”
Coronary angiography findings showed that only 1% received a normal result, 29.3% had vessel damage, and 14.6% had aortic dissection. “We were surprised again here because we expected that with young women we would see lots of heart attacks without obstruction, [in other words] normal coronary arteries, atypical forms of MI,” commented Dr. Manzo-Silberman. “In fact, most presented with atheroma, often obstructive lesions, or even triple-vessel disease, in nearly a third of the cohort. So that’s another misconception dispelled – we can’t just think that because a woman is young, nothing will be found. Coronary catheterization should be considered, and the diagnostic process should be completed in full.”
After 1 year, there had been two cancer-related deaths and 25 patients had undergone several angioplasty procedures. Nevertheless, 90.4% had not experienced any type of CV event, and 72% had not even had any symptoms.
“The final surprise was prognosis,” he said. “Previous studies, especially some authored by Viola Vaccarino, MD, PhD, showed an excess hospital rate in women and we had expected this to be the case here, but no hospital deaths were recorded. However, not far off 10% of women attended (at least once) the emergency department in the year following for recurrent chest pain which was not ischemic – ECG normal, troponin normal – so something was missing in their education as a patient.”
“So, there are improvements to be made in terms of secondary prevention, follow-up, and in the education of these young female patients who have experienced the major event that is a myocardial infarction,” concluded Dr. Manzo-Silberman.
This content was originally published on Medscape French edition.
Young women (under 50) are increasingly having heart attacks without doctors really knowing why. This is where the Young Women Presenting Acute Myocardial Infarction in France (WAMIF) study comes in, the results of which were presented in an e-poster at the annual congress of the European Society of Cardiology by Stéphane Manzo-Silberman, MD, Institute of Cardiology, Pitié-Salpétrière, Paris. The results (yet to be published) fight several of the preconceived ideas on the topic, Dr. Manzo-Silberman commented in an interview.
Significantly higher hospital death rates in women
“Cardiovascular disease is the main cause of death in women, killing seven times more than breast cancer,” notes Dr. Manzo-Silberman. The hospital death rate is significantly higher in women and, despite going down, is significantly higher than in men (more than double), particularly in women under 50. What’s more, in addition to the typical risk factors, women present specific risk factors related to hormone changes, high-risk inflammatory profiles, and thrombophilia.”
The WAMIF study was designed to determine the clinical, biological, and morphological features linked to hospital mortality after 12 months in women under 50. The prospective, observational study included all women in this age range from 30 sites in France between May 2017 and June 2019.
90% with retrosternal chest pain
The age of the 314 women enrolled was 44.9 years on average. Nearly two-thirds (192) presented with ST-segment elevation myocardial infarction and the other 122 without. In terms of symptoms, 91.6% of these women presented with typical chest pain, and 59.7% had related symptoms.
“With more than 90% having retrosternal pain, the idea that myocardial infarction presents with atypical symptoms in women has been widely challenged, despite the fact that more than half present with related symptoms and it isn’t known in which order these symptoms occur, Dr. Manzo-Silberman said in an interview. But what we can say is that if at any point a young woman mentions chest pain, even when occurring as part of several other symptoms, MI must be deemed a possibility until it has been ruled out.”
The risk profile revealed that 75.5% were smokers, 35% had a family history of heart disease, 33% had pregnancy complications, and 55% had recently experienced a stressful situation. The analysis also showed that cannabis use and oral contraception were primary risk factors in women younger than 35.
“With regard to risk factors, when designing this study we expected that lots of these young women would have largely atypical autoimmune conditions, with high levels of inflammation. We looked for everything, but this was not actually the case. Instead, we found very many women to have classic risk factors; three-quarters were smokers, a modifiable risk factor, which can largely be prevented. The other aspect concerns contraception, and it’s why I insist that gynecologists must be involved insofar as they must inform their patients how to manage their risk factors and tweak their contraception.”
Coronary angiography findings showed that only 1% received a normal result, 29.3% had vessel damage, and 14.6% had aortic dissection. “We were surprised again here because we expected that with young women we would see lots of heart attacks without obstruction, [in other words] normal coronary arteries, atypical forms of MI,” commented Dr. Manzo-Silberman. “In fact, most presented with atheroma, often obstructive lesions, or even triple-vessel disease, in nearly a third of the cohort. So that’s another misconception dispelled – we can’t just think that because a woman is young, nothing will be found. Coronary catheterization should be considered, and the diagnostic process should be completed in full.”
After 1 year, there had been two cancer-related deaths and 25 patients had undergone several angioplasty procedures. Nevertheless, 90.4% had not experienced any type of CV event, and 72% had not even had any symptoms.
“The final surprise was prognosis,” he said. “Previous studies, especially some authored by Viola Vaccarino, MD, PhD, showed an excess hospital rate in women and we had expected this to be the case here, but no hospital deaths were recorded. However, not far off 10% of women attended (at least once) the emergency department in the year following for recurrent chest pain which was not ischemic – ECG normal, troponin normal – so something was missing in their education as a patient.”
“So, there are improvements to be made in terms of secondary prevention, follow-up, and in the education of these young female patients who have experienced the major event that is a myocardial infarction,” concluded Dr. Manzo-Silberman.
This content was originally published on Medscape French edition.
Young women (under 50) are increasingly having heart attacks without doctors really knowing why. This is where the Young Women Presenting Acute Myocardial Infarction in France (WAMIF) study comes in, the results of which were presented in an e-poster at the annual congress of the European Society of Cardiology by Stéphane Manzo-Silberman, MD, Institute of Cardiology, Pitié-Salpétrière, Paris. The results (yet to be published) fight several of the preconceived ideas on the topic, Dr. Manzo-Silberman commented in an interview.
Significantly higher hospital death rates in women
“Cardiovascular disease is the main cause of death in women, killing seven times more than breast cancer,” notes Dr. Manzo-Silberman. The hospital death rate is significantly higher in women and, despite going down, is significantly higher than in men (more than double), particularly in women under 50. What’s more, in addition to the typical risk factors, women present specific risk factors related to hormone changes, high-risk inflammatory profiles, and thrombophilia.”
The WAMIF study was designed to determine the clinical, biological, and morphological features linked to hospital mortality after 12 months in women under 50. The prospective, observational study included all women in this age range from 30 sites in France between May 2017 and June 2019.
90% with retrosternal chest pain
The age of the 314 women enrolled was 44.9 years on average. Nearly two-thirds (192) presented with ST-segment elevation myocardial infarction and the other 122 without. In terms of symptoms, 91.6% of these women presented with typical chest pain, and 59.7% had related symptoms.
“With more than 90% having retrosternal pain, the idea that myocardial infarction presents with atypical symptoms in women has been widely challenged, despite the fact that more than half present with related symptoms and it isn’t known in which order these symptoms occur, Dr. Manzo-Silberman said in an interview. But what we can say is that if at any point a young woman mentions chest pain, even when occurring as part of several other symptoms, MI must be deemed a possibility until it has been ruled out.”
The risk profile revealed that 75.5% were smokers, 35% had a family history of heart disease, 33% had pregnancy complications, and 55% had recently experienced a stressful situation. The analysis also showed that cannabis use and oral contraception were primary risk factors in women younger than 35.
“With regard to risk factors, when designing this study we expected that lots of these young women would have largely atypical autoimmune conditions, with high levels of inflammation. We looked for everything, but this was not actually the case. Instead, we found very many women to have classic risk factors; three-quarters were smokers, a modifiable risk factor, which can largely be prevented. The other aspect concerns contraception, and it’s why I insist that gynecologists must be involved insofar as they must inform their patients how to manage their risk factors and tweak their contraception.”
Coronary angiography findings showed that only 1% received a normal result, 29.3% had vessel damage, and 14.6% had aortic dissection. “We were surprised again here because we expected that with young women we would see lots of heart attacks without obstruction, [in other words] normal coronary arteries, atypical forms of MI,” commented Dr. Manzo-Silberman. “In fact, most presented with atheroma, often obstructive lesions, or even triple-vessel disease, in nearly a third of the cohort. So that’s another misconception dispelled – we can’t just think that because a woman is young, nothing will be found. Coronary catheterization should be considered, and the diagnostic process should be completed in full.”
After 1 year, there had been two cancer-related deaths and 25 patients had undergone several angioplasty procedures. Nevertheless, 90.4% had not experienced any type of CV event, and 72% had not even had any symptoms.
“The final surprise was prognosis,” he said. “Previous studies, especially some authored by Viola Vaccarino, MD, PhD, showed an excess hospital rate in women and we had expected this to be the case here, but no hospital deaths were recorded. However, not far off 10% of women attended (at least once) the emergency department in the year following for recurrent chest pain which was not ischemic – ECG normal, troponin normal – so something was missing in their education as a patient.”
“So, there are improvements to be made in terms of secondary prevention, follow-up, and in the education of these young female patients who have experienced the major event that is a myocardial infarction,” concluded Dr. Manzo-Silberman.
This content was originally published on Medscape French edition.
FROM ESC CONGRESS 2022
Polycyclic Scaly Eruption
The Diagnosis: Netherton Syndrome
A punch biopsy from the right lower back supported the clinical diagnosis of ichthyosis linearis circumflexa. The patient underwent genetic testing and was found to have a heterozygous mutation in the serine protease inhibitor Kazal type 5 gene, SPINK5, that was consistent with a diagnosis of Netherton syndrome.
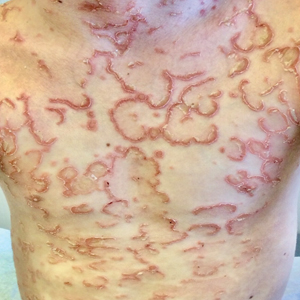
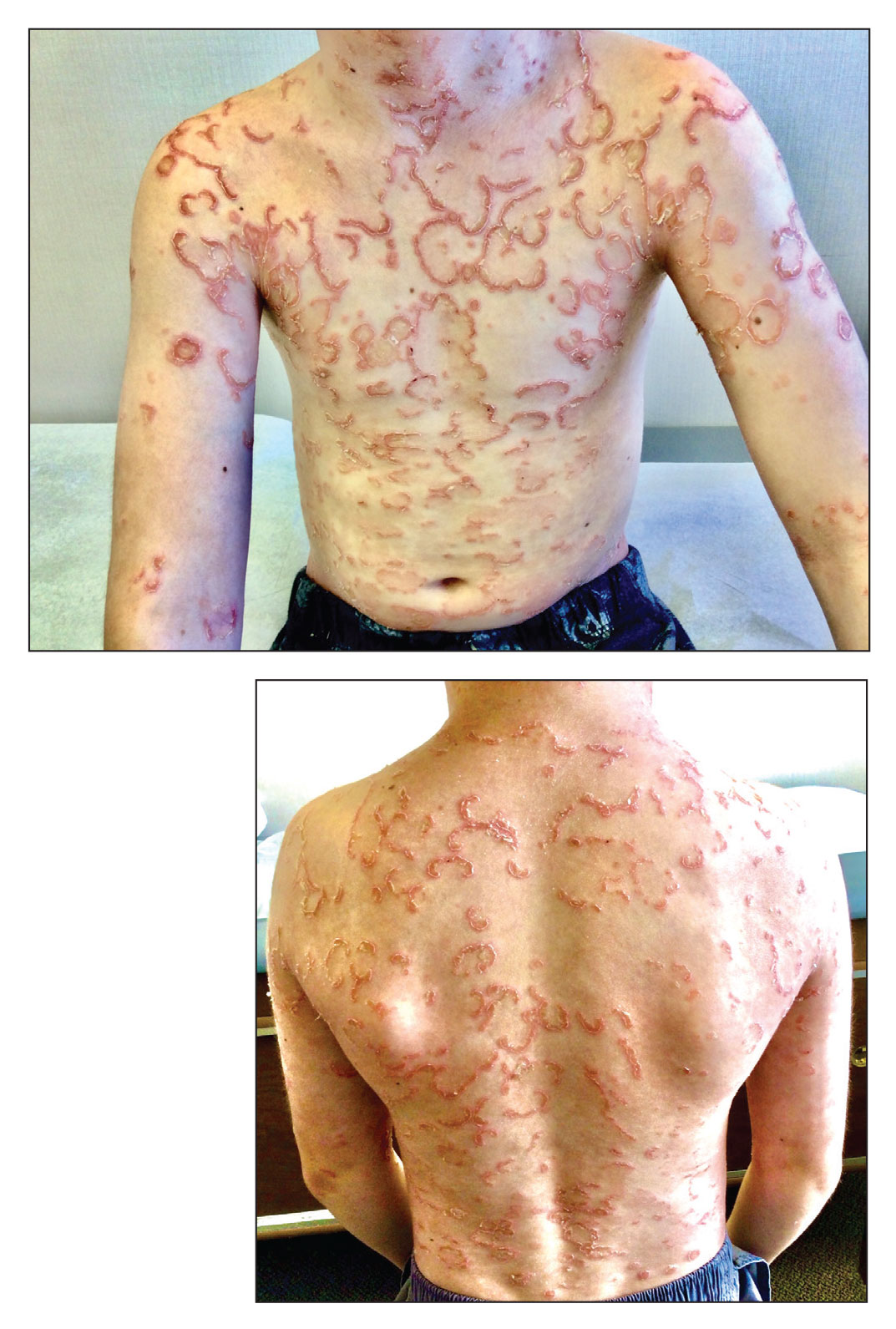
Netherton syndrome is an autosomal-recessive genodermatosis characterized by a triad of congenital ichthyosis, hair shaft abnormalities, and atopic diatheses.1,2 It affects approximately 1 in 200,000 live births2,3; however, it is considered by many to be underdiagnosed due to the variability in the clinical appearance. Therefore, the incidence of Netherton syndrome may actually be closer 1 in 50,000 live births.1 The manifestations of the disease are caused by a germline mutation in the SPINK5 gene, which encodes the serine protease inhibitor LEKTI.1,2 Dysfunctional LEKTI results in increased proteolytic activity of the lipid-processing enzymes in the stratum corneum, resulting in a disruption in the lipid bilayer.1 Dysfunctional LEKTI also results in a loss of the antiinflammatory and antimicrobial function of the stratum corneum. Clinical features of Netherton syndrome usually present at birth or shortly thereafter.1 Congenital ichthyosiform erythroderma, or the continuous peeling of the skin, is a common presentation seen at birth and in the neonatal period.2 As the patient ages, the dermatologic manifestations evolve into serpiginous and circinate, erythematous plaques with a characteristic peripheral, double-edged scaling.1,2 This distinctive finding is termed ichthyosis linearis circumflexa and is pathognomonic for the syndrome.2 Lesions often affect the trunk and extremities and demonstrate an undulating course.1 Because eczematous and lichenified plaques in flexural areas as well as pruritus are common clinical features, this disease often is misdiagnosed as atopic dermatitis,1,3 as was the case in our patient.
Patients with Netherton syndrome can present with various hair abnormalities. Trichorrhexis invaginata, known as bamboo hair, is the intussusception of the hair shaft and is characteristic of the disease.3 It develops from a reduced number of disulfide bonds, which results in cortical softening.1 Trichorrhexis invaginata may not be present at birth and often improves with age.1,3 Other hair shaft abnormalities such as pili torti, trichorrhexis nodosa, and helical hair also may be observed in Netherton syndrome.1 Extracutaneous manifestations also are typical. There is immune dysregulation of memory B cells and natural killer cells, which manifests as frequent respiratory and skin infections as well as sepsis.1,2 Patients also may have increased levels of serum IgE and eosinophilia resulting in atopy and allergic reactions to various triggers such as foods.1 The neonatal period also may be complicated by dehydration, electrolyte imbalances, inability to regulate body temperature, and failure to thrive.1,3
When there is an extensive disruption of the skin barrier during the neonatal period, there may be severe electrolyte imbalances and thermoregulatory challenges necessitating treatment in the neonatal intensive care unit. Cutaneous disease can be treated with topical therapies with variable success.1 Topical therapies for symptom management include emollients, corticosteroids, calcineurin inhibitors, calcipotriene, and retinoids; however, utmost caution must be employed with these therapies due to the increased risk for systemic absorption resulting from the disturbance of the skin barrier. When therapy with topical tacrolimus is implemented, monitoring of serum drug levels is required.1 Pruritus may be treated symptomatically with oral antihistamines. Intravenous immunoglobulin has been shown to decrease the frequency of infections and improve skin inflammation. Systemic retinoids have unpredictable effects and result in improvement of disease in some patients but exacerbation in others. Phototherapy with narrowband UVB, psoralen plus UVA, UVA1, and balneophototherapy also are effective treatments for cutaneous disease.1 Dupilumab has been shown to decrease pruritus, improve hair abnormalities, and improve skin disease, thereby demonstrating its effectiveness in treating the atopy and ichthyosis in Netherton syndrome.4
The differential diagnosis includes other figurate erythemas including erythema marginatum and erythrokeratodermia variabilis. Erythema marginatum is a cutaneous manifestation of acute rheumatic fever and is characterized by migratory polycyclic erythematous plaques without overlying scale, usually on the trunk and proximal extremities.5 Erythrokeratodermia variabilis is caused by heterozygous mutations in gap junction protein beta 3, GJB3, and gap junction protein beta 4, GJB4, and is characterized by transient geographic and erythematous patches and stable scaly plaques; however, double-edged scaling is not a feature.1 Acrodermatitis enteropathica is an autosomal-recessive disorder caused by mutations in the zinc transporter SLC39A4. Cutaneous manifestations occur after weaning from breast milk and are characterized by erythematous plaques with erosions, vesicles, and scaling, which characteristically occur in the perioral and perianal locations.6 Neonatal lupus is a form of subacute cutaneous lupus erythematosus. Typical skin lesions are erythematous annular plaques with overlying scaling, which may be present at birth and have a predilection for the face and other sun-exposed areas. Lesions generally resolve after clearance of the pathogenic maternal antibodies.7
- Richard G, Ringpfeil F. Ichthyoses, erythrokeratodermas, and related disorders. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2018:888-923.
- Garza JI, Herz-Ruelas ME, Guerrero-González GA, et al. Netherton syndrome: a diagnostic and therapeutic challenge. J Am Acad Dermatol. 2016;74(suppl 1):AB129.
- Heymann W. Appending the appendages: new perspectives on Netherton syndrome and green nail syndrome. J Am Acad Dermatol. 2020;83:735-736.
- Murase C, Takeichi T, Taki T, et al. Successful dupilumab treatment for ichthyotic and atopic features of Netherton syndrome. J Dermatol Sci. 2021;102:126-129.
- España A. Figurate erythemas. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2018:320-331.
- Noguera-Morel L, McLeish Schaefer S, Hivnor C. Nutritional diseases. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2018:793-809.
- Lee L, Werth V. Lupus erythematosus. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2018:662-680.
The Diagnosis: Netherton Syndrome
A punch biopsy from the right lower back supported the clinical diagnosis of ichthyosis linearis circumflexa. The patient underwent genetic testing and was found to have a heterozygous mutation in the serine protease inhibitor Kazal type 5 gene, SPINK5, that was consistent with a diagnosis of Netherton syndrome.
Netherton syndrome is an autosomal-recessive genodermatosis characterized by a triad of congenital ichthyosis, hair shaft abnormalities, and atopic diatheses.1,2 It affects approximately 1 in 200,000 live births2,3; however, it is considered by many to be underdiagnosed due to the variability in the clinical appearance. Therefore, the incidence of Netherton syndrome may actually be closer 1 in 50,000 live births.1 The manifestations of the disease are caused by a germline mutation in the SPINK5 gene, which encodes the serine protease inhibitor LEKTI.1,2 Dysfunctional LEKTI results in increased proteolytic activity of the lipid-processing enzymes in the stratum corneum, resulting in a disruption in the lipid bilayer.1 Dysfunctional LEKTI also results in a loss of the antiinflammatory and antimicrobial function of the stratum corneum. Clinical features of Netherton syndrome usually present at birth or shortly thereafter.1 Congenital ichthyosiform erythroderma, or the continuous peeling of the skin, is a common presentation seen at birth and in the neonatal period.2 As the patient ages, the dermatologic manifestations evolve into serpiginous and circinate, erythematous plaques with a characteristic peripheral, double-edged scaling.1,2 This distinctive finding is termed ichthyosis linearis circumflexa and is pathognomonic for the syndrome.2 Lesions often affect the trunk and extremities and demonstrate an undulating course.1 Because eczematous and lichenified plaques in flexural areas as well as pruritus are common clinical features, this disease often is misdiagnosed as atopic dermatitis,1,3 as was the case in our patient.
Patients with Netherton syndrome can present with various hair abnormalities. Trichorrhexis invaginata, known as bamboo hair, is the intussusception of the hair shaft and is characteristic of the disease.3 It develops from a reduced number of disulfide bonds, which results in cortical softening.1 Trichorrhexis invaginata may not be present at birth and often improves with age.1,3 Other hair shaft abnormalities such as pili torti, trichorrhexis nodosa, and helical hair also may be observed in Netherton syndrome.1 Extracutaneous manifestations also are typical. There is immune dysregulation of memory B cells and natural killer cells, which manifests as frequent respiratory and skin infections as well as sepsis.1,2 Patients also may have increased levels of serum IgE and eosinophilia resulting in atopy and allergic reactions to various triggers such as foods.1 The neonatal period also may be complicated by dehydration, electrolyte imbalances, inability to regulate body temperature, and failure to thrive.1,3
When there is an extensive disruption of the skin barrier during the neonatal period, there may be severe electrolyte imbalances and thermoregulatory challenges necessitating treatment in the neonatal intensive care unit. Cutaneous disease can be treated with topical therapies with variable success.1 Topical therapies for symptom management include emollients, corticosteroids, calcineurin inhibitors, calcipotriene, and retinoids; however, utmost caution must be employed with these therapies due to the increased risk for systemic absorption resulting from the disturbance of the skin barrier. When therapy with topical tacrolimus is implemented, monitoring of serum drug levels is required.1 Pruritus may be treated symptomatically with oral antihistamines. Intravenous immunoglobulin has been shown to decrease the frequency of infections and improve skin inflammation. Systemic retinoids have unpredictable effects and result in improvement of disease in some patients but exacerbation in others. Phototherapy with narrowband UVB, psoralen plus UVA, UVA1, and balneophototherapy also are effective treatments for cutaneous disease.1 Dupilumab has been shown to decrease pruritus, improve hair abnormalities, and improve skin disease, thereby demonstrating its effectiveness in treating the atopy and ichthyosis in Netherton syndrome.4
The differential diagnosis includes other figurate erythemas including erythema marginatum and erythrokeratodermia variabilis. Erythema marginatum is a cutaneous manifestation of acute rheumatic fever and is characterized by migratory polycyclic erythematous plaques without overlying scale, usually on the trunk and proximal extremities.5 Erythrokeratodermia variabilis is caused by heterozygous mutations in gap junction protein beta 3, GJB3, and gap junction protein beta 4, GJB4, and is characterized by transient geographic and erythematous patches and stable scaly plaques; however, double-edged scaling is not a feature.1 Acrodermatitis enteropathica is an autosomal-recessive disorder caused by mutations in the zinc transporter SLC39A4. Cutaneous manifestations occur after weaning from breast milk and are characterized by erythematous plaques with erosions, vesicles, and scaling, which characteristically occur in the perioral and perianal locations.6 Neonatal lupus is a form of subacute cutaneous lupus erythematosus. Typical skin lesions are erythematous annular plaques with overlying scaling, which may be present at birth and have a predilection for the face and other sun-exposed areas. Lesions generally resolve after clearance of the pathogenic maternal antibodies.7
The Diagnosis: Netherton Syndrome
A punch biopsy from the right lower back supported the clinical diagnosis of ichthyosis linearis circumflexa. The patient underwent genetic testing and was found to have a heterozygous mutation in the serine protease inhibitor Kazal type 5 gene, SPINK5, that was consistent with a diagnosis of Netherton syndrome.
Netherton syndrome is an autosomal-recessive genodermatosis characterized by a triad of congenital ichthyosis, hair shaft abnormalities, and atopic diatheses.1,2 It affects approximately 1 in 200,000 live births2,3; however, it is considered by many to be underdiagnosed due to the variability in the clinical appearance. Therefore, the incidence of Netherton syndrome may actually be closer 1 in 50,000 live births.1 The manifestations of the disease are caused by a germline mutation in the SPINK5 gene, which encodes the serine protease inhibitor LEKTI.1,2 Dysfunctional LEKTI results in increased proteolytic activity of the lipid-processing enzymes in the stratum corneum, resulting in a disruption in the lipid bilayer.1 Dysfunctional LEKTI also results in a loss of the antiinflammatory and antimicrobial function of the stratum corneum. Clinical features of Netherton syndrome usually present at birth or shortly thereafter.1 Congenital ichthyosiform erythroderma, or the continuous peeling of the skin, is a common presentation seen at birth and in the neonatal period.2 As the patient ages, the dermatologic manifestations evolve into serpiginous and circinate, erythematous plaques with a characteristic peripheral, double-edged scaling.1,2 This distinctive finding is termed ichthyosis linearis circumflexa and is pathognomonic for the syndrome.2 Lesions often affect the trunk and extremities and demonstrate an undulating course.1 Because eczematous and lichenified plaques in flexural areas as well as pruritus are common clinical features, this disease often is misdiagnosed as atopic dermatitis,1,3 as was the case in our patient.
Patients with Netherton syndrome can present with various hair abnormalities. Trichorrhexis invaginata, known as bamboo hair, is the intussusception of the hair shaft and is characteristic of the disease.3 It develops from a reduced number of disulfide bonds, which results in cortical softening.1 Trichorrhexis invaginata may not be present at birth and often improves with age.1,3 Other hair shaft abnormalities such as pili torti, trichorrhexis nodosa, and helical hair also may be observed in Netherton syndrome.1 Extracutaneous manifestations also are typical. There is immune dysregulation of memory B cells and natural killer cells, which manifests as frequent respiratory and skin infections as well as sepsis.1,2 Patients also may have increased levels of serum IgE and eosinophilia resulting in atopy and allergic reactions to various triggers such as foods.1 The neonatal period also may be complicated by dehydration, electrolyte imbalances, inability to regulate body temperature, and failure to thrive.1,3
When there is an extensive disruption of the skin barrier during the neonatal period, there may be severe electrolyte imbalances and thermoregulatory challenges necessitating treatment in the neonatal intensive care unit. Cutaneous disease can be treated with topical therapies with variable success.1 Topical therapies for symptom management include emollients, corticosteroids, calcineurin inhibitors, calcipotriene, and retinoids; however, utmost caution must be employed with these therapies due to the increased risk for systemic absorption resulting from the disturbance of the skin barrier. When therapy with topical tacrolimus is implemented, monitoring of serum drug levels is required.1 Pruritus may be treated symptomatically with oral antihistamines. Intravenous immunoglobulin has been shown to decrease the frequency of infections and improve skin inflammation. Systemic retinoids have unpredictable effects and result in improvement of disease in some patients but exacerbation in others. Phototherapy with narrowband UVB, psoralen plus UVA, UVA1, and balneophototherapy also are effective treatments for cutaneous disease.1 Dupilumab has been shown to decrease pruritus, improve hair abnormalities, and improve skin disease, thereby demonstrating its effectiveness in treating the atopy and ichthyosis in Netherton syndrome.4
The differential diagnosis includes other figurate erythemas including erythema marginatum and erythrokeratodermia variabilis. Erythema marginatum is a cutaneous manifestation of acute rheumatic fever and is characterized by migratory polycyclic erythematous plaques without overlying scale, usually on the trunk and proximal extremities.5 Erythrokeratodermia variabilis is caused by heterozygous mutations in gap junction protein beta 3, GJB3, and gap junction protein beta 4, GJB4, and is characterized by transient geographic and erythematous patches and stable scaly plaques; however, double-edged scaling is not a feature.1 Acrodermatitis enteropathica is an autosomal-recessive disorder caused by mutations in the zinc transporter SLC39A4. Cutaneous manifestations occur after weaning from breast milk and are characterized by erythematous plaques with erosions, vesicles, and scaling, which characteristically occur in the perioral and perianal locations.6 Neonatal lupus is a form of subacute cutaneous lupus erythematosus. Typical skin lesions are erythematous annular plaques with overlying scaling, which may be present at birth and have a predilection for the face and other sun-exposed areas. Lesions generally resolve after clearance of the pathogenic maternal antibodies.7
- Richard G, Ringpfeil F. Ichthyoses, erythrokeratodermas, and related disorders. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2018:888-923.
- Garza JI, Herz-Ruelas ME, Guerrero-González GA, et al. Netherton syndrome: a diagnostic and therapeutic challenge. J Am Acad Dermatol. 2016;74(suppl 1):AB129.
- Heymann W. Appending the appendages: new perspectives on Netherton syndrome and green nail syndrome. J Am Acad Dermatol. 2020;83:735-736.
- Murase C, Takeichi T, Taki T, et al. Successful dupilumab treatment for ichthyotic and atopic features of Netherton syndrome. J Dermatol Sci. 2021;102:126-129.
- España A. Figurate erythemas. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2018:320-331.
- Noguera-Morel L, McLeish Schaefer S, Hivnor C. Nutritional diseases. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2018:793-809.
- Lee L, Werth V. Lupus erythematosus. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2018:662-680.
- Richard G, Ringpfeil F. Ichthyoses, erythrokeratodermas, and related disorders. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2018:888-923.
- Garza JI, Herz-Ruelas ME, Guerrero-González GA, et al. Netherton syndrome: a diagnostic and therapeutic challenge. J Am Acad Dermatol. 2016;74(suppl 1):AB129.
- Heymann W. Appending the appendages: new perspectives on Netherton syndrome and green nail syndrome. J Am Acad Dermatol. 2020;83:735-736.
- Murase C, Takeichi T, Taki T, et al. Successful dupilumab treatment for ichthyotic and atopic features of Netherton syndrome. J Dermatol Sci. 2021;102:126-129.
- España A. Figurate erythemas. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2018:320-331.
- Noguera-Morel L, McLeish Schaefer S, Hivnor C. Nutritional diseases. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2018:793-809.
- Lee L, Werth V. Lupus erythematosus. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2018:662-680.
A 9-year-old boy presented to the dermatology clinic with a scaly eruption distributed throughout the body that had been present since birth. He had been diagnosed with atopic dermatitis by multiple dermatologists prior to the current presentation and had been treated with various topical steroids with minimal improvement. He had no family history of similar eruptions and no personal history of asthma or allergies. Physical examination revealed erythematous, serpiginous, polycyclic plaques with peripheral, double-edged scaling. Decreased hair density of the lateral eyebrows also was observed.
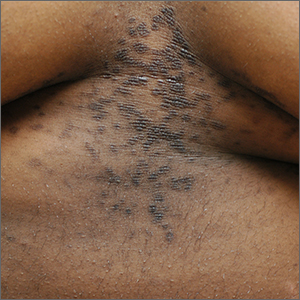
An asymptomatic rash
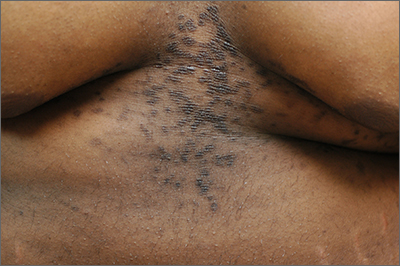
This patient was given a diagnosis of confluent and reticulated papillomatosis (CRP) based on the clinical presentation.
CRP is characterized by centrally confluent and peripherally reticulated scaly brown plaques and papules that are cosmetically disfiguring.1 CRP is usually asymptomatic and primarily impacts young adults—especially teenagers.2,3 It affects both males and females and commonly occurs on the trunk.1-3 CRP is believed to be a disorder of keratinization. Malassezia furfur may induce CRP’s hyperproliferative epidermal changes, but systemic treatment that eliminates this organism does not clear CRP.3
A CRP diagnosis is made based on clinical presentation. The eruption usually begins as verrucous papules in the inframammary or epigastric region that enlarge to 4 to 5 mm in diameter and coalesce to form a confluent plaque with a peripheral reticulated pattern. CRP can extend over the back, chest, and abdomen to the neck, shoulders, and gluteal cleft. CRP does not affect the oral mucosa, and rarely involves flexural areas, which differentiates it from the similar looking acanthosis nigricans.2 Although most cases are asymptomatic, mild pruritus may occur.1,2
A skin biopsy is rarely necessary for making a CRP diagnosis, but histopathologic findings include papillomatosis, hyperkeratosis, variable acanthosis, follicular plugging, and sparse dermal inflammation.1,3
Systemic antibiotics, most commonly minocycline 100 mg bid for 30 days or doxycycline 100 mg bid for 30 days, are safe and effective for CRP.1,4 Sometimes treatment is extended for as long as 6 months. Although CRP usually responds to minocycline or doxycycline, it is believed that this is the result of these drugs’ anti-inflammatory—rather than antibiotic—properties.1,2,4 Azithromycin is an effective alternative therapy.2,4 There is a high rate of recurrence of CRP in patients after systemic antibiotics are discontinued.2 Uniform responses to treatment and retreatment of flares have solidified the belief that antibiotics are an effective suppressive (if not curative) therapy despite a lack of randomized controlled trials.4
This patient was treated with minocycline 100 mg bid. After 1 month, the rash had improved by 70%. At 3 months, it was completely clear, and treatment was discontinued.
This case was adapted from: Sessums MT, Ward KMH, Brodell R. Cutaneous eruption on chest and back. J Fam Pract. 2014;63:467-468.
1. Davis MD, Weenig RH, Camilleri MJ. Confluent and reticulated papillomatosis (Gougerot-Carteaud syndrome): a minocycline-responsive dermatosis without evidence for yeast in pathogenesis. A study of 39 patients and a proposal of diagnostic criteria. Br J Dermatol. 2006;154:287-293. doi: 10.1111/j.1365-2133.2005.06955.x
2. Scheinfeld N. Confluent and reticulated papillomatosis: a review of the literature. Am J Clin Dermatol. 2006;7:305-313. doi: 10.2165/00128071-200607050-00004
3. Tamraz H, Raffoul M, Kurban M, et al. Confluent and reticulated papillomatosis: clinical and histopathological study of 10 cases from Lebanon. J Eur Acad Dermatol Venereol. 2013;27:e119-e123. doi: 10.1111/j.1468-3083.2011.04328.x
4. Jang HS, Oh CK, Cha JH, et al. Six cases of confluent and reticulated papillomatosis alleviated by various antibiotics. J Am Acad Dermatol. 2001;44:652-655. doi: 10.1067/mjd.2001.112577

This patient was given a diagnosis of confluent and reticulated papillomatosis (CRP) based on the clinical presentation.
CRP is characterized by centrally confluent and peripherally reticulated scaly brown plaques and papules that are cosmetically disfiguring.1 CRP is usually asymptomatic and primarily impacts young adults—especially teenagers.2,3 It affects both males and females and commonly occurs on the trunk.1-3 CRP is believed to be a disorder of keratinization. Malassezia furfur may induce CRP’s hyperproliferative epidermal changes, but systemic treatment that eliminates this organism does not clear CRP.3
A CRP diagnosis is made based on clinical presentation. The eruption usually begins as verrucous papules in the inframammary or epigastric region that enlarge to 4 to 5 mm in diameter and coalesce to form a confluent plaque with a peripheral reticulated pattern. CRP can extend over the back, chest, and abdomen to the neck, shoulders, and gluteal cleft. CRP does not affect the oral mucosa, and rarely involves flexural areas, which differentiates it from the similar looking acanthosis nigricans.2 Although most cases are asymptomatic, mild pruritus may occur.1,2
A skin biopsy is rarely necessary for making a CRP diagnosis, but histopathologic findings include papillomatosis, hyperkeratosis, variable acanthosis, follicular plugging, and sparse dermal inflammation.1,3
Systemic antibiotics, most commonly minocycline 100 mg bid for 30 days or doxycycline 100 mg bid for 30 days, are safe and effective for CRP.1,4 Sometimes treatment is extended for as long as 6 months. Although CRP usually responds to minocycline or doxycycline, it is believed that this is the result of these drugs’ anti-inflammatory—rather than antibiotic—properties.1,2,4 Azithromycin is an effective alternative therapy.2,4 There is a high rate of recurrence of CRP in patients after systemic antibiotics are discontinued.2 Uniform responses to treatment and retreatment of flares have solidified the belief that antibiotics are an effective suppressive (if not curative) therapy despite a lack of randomized controlled trials.4
This patient was treated with minocycline 100 mg bid. After 1 month, the rash had improved by 70%. At 3 months, it was completely clear, and treatment was discontinued.
This case was adapted from: Sessums MT, Ward KMH, Brodell R. Cutaneous eruption on chest and back. J Fam Pract. 2014;63:467-468.

This patient was given a diagnosis of confluent and reticulated papillomatosis (CRP) based on the clinical presentation.
CRP is characterized by centrally confluent and peripherally reticulated scaly brown plaques and papules that are cosmetically disfiguring.1 CRP is usually asymptomatic and primarily impacts young adults—especially teenagers.2,3 It affects both males and females and commonly occurs on the trunk.1-3 CRP is believed to be a disorder of keratinization. Malassezia furfur may induce CRP’s hyperproliferative epidermal changes, but systemic treatment that eliminates this organism does not clear CRP.3
A CRP diagnosis is made based on clinical presentation. The eruption usually begins as verrucous papules in the inframammary or epigastric region that enlarge to 4 to 5 mm in diameter and coalesce to form a confluent plaque with a peripheral reticulated pattern. CRP can extend over the back, chest, and abdomen to the neck, shoulders, and gluteal cleft. CRP does not affect the oral mucosa, and rarely involves flexural areas, which differentiates it from the similar looking acanthosis nigricans.2 Although most cases are asymptomatic, mild pruritus may occur.1,2
A skin biopsy is rarely necessary for making a CRP diagnosis, but histopathologic findings include papillomatosis, hyperkeratosis, variable acanthosis, follicular plugging, and sparse dermal inflammation.1,3
Systemic antibiotics, most commonly minocycline 100 mg bid for 30 days or doxycycline 100 mg bid for 30 days, are safe and effective for CRP.1,4 Sometimes treatment is extended for as long as 6 months. Although CRP usually responds to minocycline or doxycycline, it is believed that this is the result of these drugs’ anti-inflammatory—rather than antibiotic—properties.1,2,4 Azithromycin is an effective alternative therapy.2,4 There is a high rate of recurrence of CRP in patients after systemic antibiotics are discontinued.2 Uniform responses to treatment and retreatment of flares have solidified the belief that antibiotics are an effective suppressive (if not curative) therapy despite a lack of randomized controlled trials.4
This patient was treated with minocycline 100 mg bid. After 1 month, the rash had improved by 70%. At 3 months, it was completely clear, and treatment was discontinued.
This case was adapted from: Sessums MT, Ward KMH, Brodell R. Cutaneous eruption on chest and back. J Fam Pract. 2014;63:467-468.
1. Davis MD, Weenig RH, Camilleri MJ. Confluent and reticulated papillomatosis (Gougerot-Carteaud syndrome): a minocycline-responsive dermatosis without evidence for yeast in pathogenesis. A study of 39 patients and a proposal of diagnostic criteria. Br J Dermatol. 2006;154:287-293. doi: 10.1111/j.1365-2133.2005.06955.x
2. Scheinfeld N. Confluent and reticulated papillomatosis: a review of the literature. Am J Clin Dermatol. 2006;7:305-313. doi: 10.2165/00128071-200607050-00004
3. Tamraz H, Raffoul M, Kurban M, et al. Confluent and reticulated papillomatosis: clinical and histopathological study of 10 cases from Lebanon. J Eur Acad Dermatol Venereol. 2013;27:e119-e123. doi: 10.1111/j.1468-3083.2011.04328.x
4. Jang HS, Oh CK, Cha JH, et al. Six cases of confluent and reticulated papillomatosis alleviated by various antibiotics. J Am Acad Dermatol. 2001;44:652-655. doi: 10.1067/mjd.2001.112577
1. Davis MD, Weenig RH, Camilleri MJ. Confluent and reticulated papillomatosis (Gougerot-Carteaud syndrome): a minocycline-responsive dermatosis without evidence for yeast in pathogenesis. A study of 39 patients and a proposal of diagnostic criteria. Br J Dermatol. 2006;154:287-293. doi: 10.1111/j.1365-2133.2005.06955.x
2. Scheinfeld N. Confluent and reticulated papillomatosis: a review of the literature. Am J Clin Dermatol. 2006;7:305-313. doi: 10.2165/00128071-200607050-00004
3. Tamraz H, Raffoul M, Kurban M, et al. Confluent and reticulated papillomatosis: clinical and histopathological study of 10 cases from Lebanon. J Eur Acad Dermatol Venereol. 2013;27:e119-e123. doi: 10.1111/j.1468-3083.2011.04328.x
4. Jang HS, Oh CK, Cha JH, et al. Six cases of confluent and reticulated papillomatosis alleviated by various antibiotics. J Am Acad Dermatol. 2001;44:652-655. doi: 10.1067/mjd.2001.112577
Ketamine promising for rare condition linked to autism
Also known as Helsmoortel–Van Der Aa syndrome, ADNP syndrome is caused by mutations in the ADNP gene. Studies in animal models suggest that low-dose ketamine increases expression of ADNP and is neuroprotective.
Intrigued by the preclinical evidence, Alexander Kolevzon, MD, clinical director of the Seaver Autism Center at Mount Sinai, New York, and colleagues treated 10 children with ADNP syndrome with a single low dose of ketamine (0.5mg/kg) infused intravenously over 40 minutes. The children ranged in ages 6-12 years.
Using parent-report instruments to assess treatment effects, ketamine was associated with “nominally significant” improvement in a variety of domains, including social behavior, attention-deficit and hyperactivity, restricted and repetitive behaviors, and sensory sensitivities.
Parent reports of improvement in these domains aligned with clinician-rated assessments based on the Clinical Global Impressions–Improvement scale.
The results also highlight the potential utility of electrophysiological measurement of auditory steady-state response and eye-tracking to track change with ketamine treatment, the researchers say.
The study was published online in Human Genetics and Genomic (HGG) Advances.
Hypothesis-generating
Ketamine was generally well tolerated. There were no clinically significant abnormalities in laboratory or cardiac monitoring, and there were no serious adverse events (AEs).
Treatment emergent AEs were all mild to moderate and no child required any interventions.
The most common AEs were elation/silliness in five children (50%), all of whom had a history of similar symptoms. Drowsiness and fatigue occurred in four children (40%) and two of them had a history of drowsiness. Aggression was likewise relatively common, reported in four children (40%), all of whom had aggression at baseline.
Decreased appetite emerged as a new AE in three children (30%), increased anxiety occurred in three children (30%), and irritability, nausea/vomiting, and restlessness each occurred in two children (20%).
The researchers caution that the findings are intended to be “hypothesis generating.”
“We are encouraged by these findings, which provide preliminary support for ketamine to help reduce negative effects of this devastating syndrome,” Dr. Kolevzon said in a news release from Mount Sinai.
Ketamine might help ease symptoms of ADNP syndrome “by increasing expression of the ADNP gene or by promoting synaptic plasticity through glutamatergic pathways,” Dr. Kolevzon told this news organization.
The next step, he said, is to get “a larger, placebo-controlled study approved for funding using repeated dosing over a longer duration of time. We are working with the FDA to get the design approved for an investigational new drug application.”
Support for the study was provided by the ADNP Kids Foundation and the Foundation for Mood Disorders. Support for mediKanren was provided by the National Center for Advancing Translational Sciences, and National Institutes of Health through the Biomedical Data Translator Program. Dr. Kolevzon is on the scientific advisory board of Ovid Therapeutics, Ritrova Therapeutics, and Jaguar Therapeutics and consults to Acadia, Alkermes, GW Pharmaceuticals, Neuren Pharmaceuticals, Clinilabs Drug Development Corporation, and Scioto Biosciences.
A version of this article first appeared on Medscape.com.
Also known as Helsmoortel–Van Der Aa syndrome, ADNP syndrome is caused by mutations in the ADNP gene. Studies in animal models suggest that low-dose ketamine increases expression of ADNP and is neuroprotective.
Intrigued by the preclinical evidence, Alexander Kolevzon, MD, clinical director of the Seaver Autism Center at Mount Sinai, New York, and colleagues treated 10 children with ADNP syndrome with a single low dose of ketamine (0.5mg/kg) infused intravenously over 40 minutes. The children ranged in ages 6-12 years.
Using parent-report instruments to assess treatment effects, ketamine was associated with “nominally significant” improvement in a variety of domains, including social behavior, attention-deficit and hyperactivity, restricted and repetitive behaviors, and sensory sensitivities.
Parent reports of improvement in these domains aligned with clinician-rated assessments based on the Clinical Global Impressions–Improvement scale.
The results also highlight the potential utility of electrophysiological measurement of auditory steady-state response and eye-tracking to track change with ketamine treatment, the researchers say.
The study was published online in Human Genetics and Genomic (HGG) Advances.
Hypothesis-generating
Ketamine was generally well tolerated. There were no clinically significant abnormalities in laboratory or cardiac monitoring, and there were no serious adverse events (AEs).
Treatment emergent AEs were all mild to moderate and no child required any interventions.
The most common AEs were elation/silliness in five children (50%), all of whom had a history of similar symptoms. Drowsiness and fatigue occurred in four children (40%) and two of them had a history of drowsiness. Aggression was likewise relatively common, reported in four children (40%), all of whom had aggression at baseline.
Decreased appetite emerged as a new AE in three children (30%), increased anxiety occurred in three children (30%), and irritability, nausea/vomiting, and restlessness each occurred in two children (20%).
The researchers caution that the findings are intended to be “hypothesis generating.”
“We are encouraged by these findings, which provide preliminary support for ketamine to help reduce negative effects of this devastating syndrome,” Dr. Kolevzon said in a news release from Mount Sinai.
Ketamine might help ease symptoms of ADNP syndrome “by increasing expression of the ADNP gene or by promoting synaptic plasticity through glutamatergic pathways,” Dr. Kolevzon told this news organization.
The next step, he said, is to get “a larger, placebo-controlled study approved for funding using repeated dosing over a longer duration of time. We are working with the FDA to get the design approved for an investigational new drug application.”
Support for the study was provided by the ADNP Kids Foundation and the Foundation for Mood Disorders. Support for mediKanren was provided by the National Center for Advancing Translational Sciences, and National Institutes of Health through the Biomedical Data Translator Program. Dr. Kolevzon is on the scientific advisory board of Ovid Therapeutics, Ritrova Therapeutics, and Jaguar Therapeutics and consults to Acadia, Alkermes, GW Pharmaceuticals, Neuren Pharmaceuticals, Clinilabs Drug Development Corporation, and Scioto Biosciences.
A version of this article first appeared on Medscape.com.
Also known as Helsmoortel–Van Der Aa syndrome, ADNP syndrome is caused by mutations in the ADNP gene. Studies in animal models suggest that low-dose ketamine increases expression of ADNP and is neuroprotective.
Intrigued by the preclinical evidence, Alexander Kolevzon, MD, clinical director of the Seaver Autism Center at Mount Sinai, New York, and colleagues treated 10 children with ADNP syndrome with a single low dose of ketamine (0.5mg/kg) infused intravenously over 40 minutes. The children ranged in ages 6-12 years.
Using parent-report instruments to assess treatment effects, ketamine was associated with “nominally significant” improvement in a variety of domains, including social behavior, attention-deficit and hyperactivity, restricted and repetitive behaviors, and sensory sensitivities.
Parent reports of improvement in these domains aligned with clinician-rated assessments based on the Clinical Global Impressions–Improvement scale.
The results also highlight the potential utility of electrophysiological measurement of auditory steady-state response and eye-tracking to track change with ketamine treatment, the researchers say.
The study was published online in Human Genetics and Genomic (HGG) Advances.
Hypothesis-generating
Ketamine was generally well tolerated. There were no clinically significant abnormalities in laboratory or cardiac monitoring, and there were no serious adverse events (AEs).
Treatment emergent AEs were all mild to moderate and no child required any interventions.
The most common AEs were elation/silliness in five children (50%), all of whom had a history of similar symptoms. Drowsiness and fatigue occurred in four children (40%) and two of them had a history of drowsiness. Aggression was likewise relatively common, reported in four children (40%), all of whom had aggression at baseline.
Decreased appetite emerged as a new AE in three children (30%), increased anxiety occurred in three children (30%), and irritability, nausea/vomiting, and restlessness each occurred in two children (20%).
The researchers caution that the findings are intended to be “hypothesis generating.”
“We are encouraged by these findings, which provide preliminary support for ketamine to help reduce negative effects of this devastating syndrome,” Dr. Kolevzon said in a news release from Mount Sinai.
Ketamine might help ease symptoms of ADNP syndrome “by increasing expression of the ADNP gene or by promoting synaptic plasticity through glutamatergic pathways,” Dr. Kolevzon told this news organization.
The next step, he said, is to get “a larger, placebo-controlled study approved for funding using repeated dosing over a longer duration of time. We are working with the FDA to get the design approved for an investigational new drug application.”
Support for the study was provided by the ADNP Kids Foundation and the Foundation for Mood Disorders. Support for mediKanren was provided by the National Center for Advancing Translational Sciences, and National Institutes of Health through the Biomedical Data Translator Program. Dr. Kolevzon is on the scientific advisory board of Ovid Therapeutics, Ritrova Therapeutics, and Jaguar Therapeutics and consults to Acadia, Alkermes, GW Pharmaceuticals, Neuren Pharmaceuticals, Clinilabs Drug Development Corporation, and Scioto Biosciences.
A version of this article first appeared on Medscape.com.
From Human Genetics and Genomic Advances
Possible sex differences found in response to first treatments for early RA
Men with early rheumatoid arthritis who had previously never been treated with disease-modifying antirheumatic drugs (DMARDs) achieved remission significantly more often than women when given the interleukin (IL)-6 inhibitor tocilizumab (Actemra), according to new findings published in The Lancet Rheumatology.
Researchers also found that men had higher rates of remission than women when treated with certolizumab pegol (Cimzia), abatacept (Orencia), or conventional synthetic DMARDs, but the differences were not statistically significant.
The findings are based on a post-hoc analysis of data from the randomized, controlled, phase 4 NORD-STAR trial performed across Scandinavia, Iceland, and the Netherlands that is believed to be the first study on treatment-naive patients to specifically analyze the interaction between sex and treatment using interaction terms. In the study, outcomes for men versus women were compared within each treatment group and also to the conventional treatment arm used as the reference group.
“Our findings could provide guidance about the optimal treatment choice for DMARD-naive men and women with early RA,” said first author Kristina Lend, MSc, research assistant at the Karolinska Institute, Stockholm, and PhD student at Amsterdam University Medical Center.
Researchers enrolled 812 patients between 2012 and 2018 and randomly assigned them to receive:
- Conventional treatment involving methotrexate plus prednisolone tapered from 20 mg per day to 5 mg per day within 9 weeks or methotrexate plus sulfasalazine (2 g per day), hydroxychloroquine (35 mg/kg per week or 200 mg per day), and intra-articular glucocorticoids in the swollen joint (maximally four joints and 80 mg per visit);
- the tumor necrosis factor (TNF) inhibitor certolizumab pegol with methotrexate;
- the T-cell co-stimulation modulator abatacept with methotrexate; or
- tocilizumab with methotrexate.
All of the patients were newly diagnosed, with symptoms for less than 24 months, and they had never taken a DMARD. Researchers used the Clinical Disease Activity Index (CDAI) as the primary tool for assessing remission. Patients started oral methotrexate initially at 10-15 mg per week and escalated within 4 weeks to a target dose of 25 mg per week.
In all groups, men achieved remission after 24 weeks at higher rates than women: 55% compared with 50% in the conventional arm; 57% vs. 52% with certolizumab pegol; 65% vs. 51% with abatacept; and 61% vs. 40% with tocilizumab. But in most cases, the 95% confidence intervals overlapped for men and women, meaning the differences didn’t reach statistical significance.
However, in the tocilizumab group, the difference was significant.
Ms. Lend said it was interesting to see this difference with tocilizumab. The drug is known to reduce acute-phase reactants, such as C-reactive protein (CRP). But the CDAI doesn’t take CRP or other acute phase reactants into account. Both men and women taking tocilizumab had significant reductions in CRP, and yet men ultimately did much better on the drug according to the CDAI, as well as other scales, such as the Disease Activity Score in 28 joints and Simplified Disease Activity Index.
Women in the conventional treatment arm actually achieved remission more often, at least in absolute numbers, than did women taking tocilizumab.
“It was surprising to see that men on tocilizumab treatment achieved higher remission rates than men in conventional treatment while women in tocilizumab treatment achieved lower remission rates than women in conventional treatment,” she said.
Several factors could account for the differences in remission, she said. Subjective components when assessing remission – such as tender joint counts and a patient’s own assessment of their disease activity – tend to be higher for women. Underlying biological mechanisms can play a role as well, with evidence suggesting that gonadal hormone concentrations modulate the immune system and affect pain signaling, influencing how the disease is experienced, she said.
Findings such as these could lead to a redrafting of treatment recommendations, Ms. Lend suggested.
“Conventional treatment is currently recommended over tocilizumab and other biologics for DMARD-naive men and women with early RA,” she said. “We do feel that the overall results of the NORD-STAR trial could lead to a reassessment of these recommendations, and that more personalized treatment decisions will become the standard.”
In an accompanying editorial, Alexandre Sepriano, MD, PhD, a rheumatologist at Hospital Egaz Moniz in Lisbon, and Elena Nikiphorou, MD, consultant rheumatologist at King’s College London, said the analysis was generally well-designed, although perhaps too small.
“The NORD-STAR trial, compared to other studies, comes the closest to answering the question at hand,” they wrote. “A fair conclusion is that (with the exception of tocilizumab) men and women respond similarly to biological DMARDs compared with conventional therapy. If true, this is reassuring news both to patients and clinicians.”
They cautioned that the study was “probably underpowered” to answer the question authoritatively.
“Despite this, the study provides useful insights into sex-driven responses to treatment,” they said. “Differences in methodological and analytical approaches will need to be considered in studies with similar intentions when interpreting the findings.”
Ruth Fritsch-Stork, MD, PhD, professor of rheumatology at Sigmund Freud University in Vienna, who has studied sex and RA treatment in the Austrian BIOREG registry, said the findings are an important contribution to the literature.
“I think it is a very interesting paper, as little literature has been published about sex differences in RA patients regarding therapy,” she said. “And the little that is known is ambiguous. So this paper is a badly needed piece in the puzzle of treatment response in RA.”
She said she wondered how much these findings will be applicable to typical clinical scenarios, in which tocilizumab is usually at least a second-line therapy, after use of conventional synthetic DMARDs – and often after anti-TNF therapy as well. But this study population was DMARD naive.
“Also, the literature usually describes a better outcome in men for anti-TNF, which was not seen here,” she added.
“As the effect of tocilizumab seems to be greater in men not only in remission rates, but also in infection rates, I do believe an effect on the IL-6 signaling and immunological sequelae to be the underlying factor,” Dr. Fritsch-Stork said. “However, I agree with the authors that unknown, noninflammatory, sex-dependent effects on pain sensation might play a role.”
Even though the applicability of the study isn’t clear, she said, “it is important information for future investigations.”
Ms. Lend and Dr. Fritsch-Stork reported no relevant financial disclosures. Dr. Sepriano reported financial relationships with UCB, Novartis, and Lilly. Dr. Nikiphorou reported financial relationships with Pfizer, Gilead, Galapagos, Lilly, and Fresenius.
Men with early rheumatoid arthritis who had previously never been treated with disease-modifying antirheumatic drugs (DMARDs) achieved remission significantly more often than women when given the interleukin (IL)-6 inhibitor tocilizumab (Actemra), according to new findings published in The Lancet Rheumatology.
Researchers also found that men had higher rates of remission than women when treated with certolizumab pegol (Cimzia), abatacept (Orencia), or conventional synthetic DMARDs, but the differences were not statistically significant.
The findings are based on a post-hoc analysis of data from the randomized, controlled, phase 4 NORD-STAR trial performed across Scandinavia, Iceland, and the Netherlands that is believed to be the first study on treatment-naive patients to specifically analyze the interaction between sex and treatment using interaction terms. In the study, outcomes for men versus women were compared within each treatment group and also to the conventional treatment arm used as the reference group.
“Our findings could provide guidance about the optimal treatment choice for DMARD-naive men and women with early RA,” said first author Kristina Lend, MSc, research assistant at the Karolinska Institute, Stockholm, and PhD student at Amsterdam University Medical Center.
Researchers enrolled 812 patients between 2012 and 2018 and randomly assigned them to receive:
- Conventional treatment involving methotrexate plus prednisolone tapered from 20 mg per day to 5 mg per day within 9 weeks or methotrexate plus sulfasalazine (2 g per day), hydroxychloroquine (35 mg/kg per week or 200 mg per day), and intra-articular glucocorticoids in the swollen joint (maximally four joints and 80 mg per visit);
- the tumor necrosis factor (TNF) inhibitor certolizumab pegol with methotrexate;
- the T-cell co-stimulation modulator abatacept with methotrexate; or
- tocilizumab with methotrexate.
All of the patients were newly diagnosed, with symptoms for less than 24 months, and they had never taken a DMARD. Researchers used the Clinical Disease Activity Index (CDAI) as the primary tool for assessing remission. Patients started oral methotrexate initially at 10-15 mg per week and escalated within 4 weeks to a target dose of 25 mg per week.
In all groups, men achieved remission after 24 weeks at higher rates than women: 55% compared with 50% in the conventional arm; 57% vs. 52% with certolizumab pegol; 65% vs. 51% with abatacept; and 61% vs. 40% with tocilizumab. But in most cases, the 95% confidence intervals overlapped for men and women, meaning the differences didn’t reach statistical significance.
However, in the tocilizumab group, the difference was significant.
Ms. Lend said it was interesting to see this difference with tocilizumab. The drug is known to reduce acute-phase reactants, such as C-reactive protein (CRP). But the CDAI doesn’t take CRP or other acute phase reactants into account. Both men and women taking tocilizumab had significant reductions in CRP, and yet men ultimately did much better on the drug according to the CDAI, as well as other scales, such as the Disease Activity Score in 28 joints and Simplified Disease Activity Index.
Women in the conventional treatment arm actually achieved remission more often, at least in absolute numbers, than did women taking tocilizumab.
“It was surprising to see that men on tocilizumab treatment achieved higher remission rates than men in conventional treatment while women in tocilizumab treatment achieved lower remission rates than women in conventional treatment,” she said.
Several factors could account for the differences in remission, she said. Subjective components when assessing remission – such as tender joint counts and a patient’s own assessment of their disease activity – tend to be higher for women. Underlying biological mechanisms can play a role as well, with evidence suggesting that gonadal hormone concentrations modulate the immune system and affect pain signaling, influencing how the disease is experienced, she said.
Findings such as these could lead to a redrafting of treatment recommendations, Ms. Lend suggested.
“Conventional treatment is currently recommended over tocilizumab and other biologics for DMARD-naive men and women with early RA,” she said. “We do feel that the overall results of the NORD-STAR trial could lead to a reassessment of these recommendations, and that more personalized treatment decisions will become the standard.”
In an accompanying editorial, Alexandre Sepriano, MD, PhD, a rheumatologist at Hospital Egaz Moniz in Lisbon, and Elena Nikiphorou, MD, consultant rheumatologist at King’s College London, said the analysis was generally well-designed, although perhaps too small.
“The NORD-STAR trial, compared to other studies, comes the closest to answering the question at hand,” they wrote. “A fair conclusion is that (with the exception of tocilizumab) men and women respond similarly to biological DMARDs compared with conventional therapy. If true, this is reassuring news both to patients and clinicians.”
They cautioned that the study was “probably underpowered” to answer the question authoritatively.
“Despite this, the study provides useful insights into sex-driven responses to treatment,” they said. “Differences in methodological and analytical approaches will need to be considered in studies with similar intentions when interpreting the findings.”
Ruth Fritsch-Stork, MD, PhD, professor of rheumatology at Sigmund Freud University in Vienna, who has studied sex and RA treatment in the Austrian BIOREG registry, said the findings are an important contribution to the literature.
“I think it is a very interesting paper, as little literature has been published about sex differences in RA patients regarding therapy,” she said. “And the little that is known is ambiguous. So this paper is a badly needed piece in the puzzle of treatment response in RA.”
She said she wondered how much these findings will be applicable to typical clinical scenarios, in which tocilizumab is usually at least a second-line therapy, after use of conventional synthetic DMARDs – and often after anti-TNF therapy as well. But this study population was DMARD naive.
“Also, the literature usually describes a better outcome in men for anti-TNF, which was not seen here,” she added.
“As the effect of tocilizumab seems to be greater in men not only in remission rates, but also in infection rates, I do believe an effect on the IL-6 signaling and immunological sequelae to be the underlying factor,” Dr. Fritsch-Stork said. “However, I agree with the authors that unknown, noninflammatory, sex-dependent effects on pain sensation might play a role.”
Even though the applicability of the study isn’t clear, she said, “it is important information for future investigations.”
Ms. Lend and Dr. Fritsch-Stork reported no relevant financial disclosures. Dr. Sepriano reported financial relationships with UCB, Novartis, and Lilly. Dr. Nikiphorou reported financial relationships with Pfizer, Gilead, Galapagos, Lilly, and Fresenius.
Men with early rheumatoid arthritis who had previously never been treated with disease-modifying antirheumatic drugs (DMARDs) achieved remission significantly more often than women when given the interleukin (IL)-6 inhibitor tocilizumab (Actemra), according to new findings published in The Lancet Rheumatology.
Researchers also found that men had higher rates of remission than women when treated with certolizumab pegol (Cimzia), abatacept (Orencia), or conventional synthetic DMARDs, but the differences were not statistically significant.
The findings are based on a post-hoc analysis of data from the randomized, controlled, phase 4 NORD-STAR trial performed across Scandinavia, Iceland, and the Netherlands that is believed to be the first study on treatment-naive patients to specifically analyze the interaction between sex and treatment using interaction terms. In the study, outcomes for men versus women were compared within each treatment group and also to the conventional treatment arm used as the reference group.
“Our findings could provide guidance about the optimal treatment choice for DMARD-naive men and women with early RA,” said first author Kristina Lend, MSc, research assistant at the Karolinska Institute, Stockholm, and PhD student at Amsterdam University Medical Center.
Researchers enrolled 812 patients between 2012 and 2018 and randomly assigned them to receive:
- Conventional treatment involving methotrexate plus prednisolone tapered from 20 mg per day to 5 mg per day within 9 weeks or methotrexate plus sulfasalazine (2 g per day), hydroxychloroquine (35 mg/kg per week or 200 mg per day), and intra-articular glucocorticoids in the swollen joint (maximally four joints and 80 mg per visit);
- the tumor necrosis factor (TNF) inhibitor certolizumab pegol with methotrexate;
- the T-cell co-stimulation modulator abatacept with methotrexate; or
- tocilizumab with methotrexate.
All of the patients were newly diagnosed, with symptoms for less than 24 months, and they had never taken a DMARD. Researchers used the Clinical Disease Activity Index (CDAI) as the primary tool for assessing remission. Patients started oral methotrexate initially at 10-15 mg per week and escalated within 4 weeks to a target dose of 25 mg per week.
In all groups, men achieved remission after 24 weeks at higher rates than women: 55% compared with 50% in the conventional arm; 57% vs. 52% with certolizumab pegol; 65% vs. 51% with abatacept; and 61% vs. 40% with tocilizumab. But in most cases, the 95% confidence intervals overlapped for men and women, meaning the differences didn’t reach statistical significance.
However, in the tocilizumab group, the difference was significant.
Ms. Lend said it was interesting to see this difference with tocilizumab. The drug is known to reduce acute-phase reactants, such as C-reactive protein (CRP). But the CDAI doesn’t take CRP or other acute phase reactants into account. Both men and women taking tocilizumab had significant reductions in CRP, and yet men ultimately did much better on the drug according to the CDAI, as well as other scales, such as the Disease Activity Score in 28 joints and Simplified Disease Activity Index.
Women in the conventional treatment arm actually achieved remission more often, at least in absolute numbers, than did women taking tocilizumab.
“It was surprising to see that men on tocilizumab treatment achieved higher remission rates than men in conventional treatment while women in tocilizumab treatment achieved lower remission rates than women in conventional treatment,” she said.
Several factors could account for the differences in remission, she said. Subjective components when assessing remission – such as tender joint counts and a patient’s own assessment of their disease activity – tend to be higher for women. Underlying biological mechanisms can play a role as well, with evidence suggesting that gonadal hormone concentrations modulate the immune system and affect pain signaling, influencing how the disease is experienced, she said.
Findings such as these could lead to a redrafting of treatment recommendations, Ms. Lend suggested.
“Conventional treatment is currently recommended over tocilizumab and other biologics for DMARD-naive men and women with early RA,” she said. “We do feel that the overall results of the NORD-STAR trial could lead to a reassessment of these recommendations, and that more personalized treatment decisions will become the standard.”
In an accompanying editorial, Alexandre Sepriano, MD, PhD, a rheumatologist at Hospital Egaz Moniz in Lisbon, and Elena Nikiphorou, MD, consultant rheumatologist at King’s College London, said the analysis was generally well-designed, although perhaps too small.
“The NORD-STAR trial, compared to other studies, comes the closest to answering the question at hand,” they wrote. “A fair conclusion is that (with the exception of tocilizumab) men and women respond similarly to biological DMARDs compared with conventional therapy. If true, this is reassuring news both to patients and clinicians.”
They cautioned that the study was “probably underpowered” to answer the question authoritatively.
“Despite this, the study provides useful insights into sex-driven responses to treatment,” they said. “Differences in methodological and analytical approaches will need to be considered in studies with similar intentions when interpreting the findings.”
Ruth Fritsch-Stork, MD, PhD, professor of rheumatology at Sigmund Freud University in Vienna, who has studied sex and RA treatment in the Austrian BIOREG registry, said the findings are an important contribution to the literature.
“I think it is a very interesting paper, as little literature has been published about sex differences in RA patients regarding therapy,” she said. “And the little that is known is ambiguous. So this paper is a badly needed piece in the puzzle of treatment response in RA.”
She said she wondered how much these findings will be applicable to typical clinical scenarios, in which tocilizumab is usually at least a second-line therapy, after use of conventional synthetic DMARDs – and often after anti-TNF therapy as well. But this study population was DMARD naive.
“Also, the literature usually describes a better outcome in men for anti-TNF, which was not seen here,” she added.
“As the effect of tocilizumab seems to be greater in men not only in remission rates, but also in infection rates, I do believe an effect on the IL-6 signaling and immunological sequelae to be the underlying factor,” Dr. Fritsch-Stork said. “However, I agree with the authors that unknown, noninflammatory, sex-dependent effects on pain sensation might play a role.”
Even though the applicability of the study isn’t clear, she said, “it is important information for future investigations.”
Ms. Lend and Dr. Fritsch-Stork reported no relevant financial disclosures. Dr. Sepriano reported financial relationships with UCB, Novartis, and Lilly. Dr. Nikiphorou reported financial relationships with Pfizer, Gilead, Galapagos, Lilly, and Fresenius.
FROM THE LANCET RHEUMATOLOGY
Dietary change tops for reducing CVD risk in stage 1 hypertension
Healthy lifestyle changes to reduce systolic blood pressure to below 130 mm Hg may prevent 26,000 heart attacks and strokes and reduce health care costs over the next 10 years, a new simulation study suggests.
Among the various lifestyle changes, adopting the Dietary Approaches to Stop Hypertension diet, known as the DASH diet, may have the greatest impact for young and middle-aged adults with stage 1 hypertension.
“This research reveals that we should look to feasible ways our food system could make healthy eating the default option,” Kendra Sims, PhD, MPH, postdoctoral fellow at University of California, San Francisco, told this news organization.
“Above all, it means collaborating with the patient about nourishing choices that fit best into their culture and lifestyle,” Dr. Sims said.
Be proactive
“What is important is that people not wait until they have hypertension to start thinking about healthful diets,” commented Taylor Wallace, PhD, department of nutrition and food studies, George Mason University, Fairfax, Va., who was not involved in the study.
“It’s all about prevention in my mind. Whether you are hypertensive or are perfectly healthy, the DASH diet or any other dietary pattern that emphasizes consumption of fruits, vegetables, whole grains, lean meats, seafood, nuts/seeds, and low/non-fat dairy and decreased intake of saturated fats, added sugars, and sodium is a good idea,” Dr. Wallace said in an interview.
The study was presented at the American Heart Association Hypertension Scientific Sessions 2022 in San Diego.
Dr. Sims and colleagues used U.S. statistics from multiple sources to simulate CVD events, mortality, and health care costs between 2018 and 2027 in adults aged 35-64 years with untreated stage 1 hypertension, defined as systolic BP of 130 to 139 mm Hg.
The researchers estimate that 8.8 million U.S. adults (5.5 million women) aged 35-64 years have untreated stage 1 hypertension and would be recommended for lifestyle change, such as physical activity, weight loss, moderating alcohol intake, and adoption of the DASH diet.
Controlling blood pressure to less than 130 mm Hg in this population could prevent 26,000 CVD events, avoid 2,900 deaths, and lead to $1.6 billion saved in associated health care costs, the researchers calculate.
The largest benefit would come from adoption of the DASH diet, with an estimated 15,000 CVD events prevented among men and 11,000 among women.
Even small changes can help
“Young and middle-aged adults with stage 1 hypertension aren’t as low risk as you – or even your doctor – might think,” Dr. Sims told this news organization.
“Millions of working-aged people are walking around with elevated blood pressure, which is symptomless but is also a leading preventable cause of disability and death. Most do not follow the recommended DASH diet,” Dr. Sims said.
“Unfortunately, the availability and affordability of healthy food sources does not easily allow people to follow the DASH diet,” Dr. Sims adds in a conference news release.
“Clinicians should consider whether their patients live in food deserts or places with limited walkability. Health counseling should include addressing these specific challenges to blood pressure control,” Dr. Sims says.
Dr. Wallace noted that diet changes don’t have to be drastic.
“Honestly, just increasing fruit and vegetable intake has been shown to displace calories from saturated fats, added sugars, and sodium,” he told this news organization.
“It’s hard for people to stick to ‘diets’ long-term, so shifting toward healthier dietary patterns without having to read a book on the DASH diet or count calories and carbs seems like a more practical solution for the general population, although I have no issues with the DASH diet and think it is a great dietary pattern for heart health,” Dr. Wallace said.
The study had no funding. Dr. Sims reports no relevant financial relationships. Dr. Wallace is principal and CEO of Think Healthy Group; chief food and nutrition scientist with Produce for Better Health Foundation; editor, Journal of Dietary Supplements; deputy editor, Journal of the American College of Nutrition; nutrition section editor, Annals of Medicine; and advisory board member with Forbes Health.
A version of this article first appeared on Medscape.com.
Healthy lifestyle changes to reduce systolic blood pressure to below 130 mm Hg may prevent 26,000 heart attacks and strokes and reduce health care costs over the next 10 years, a new simulation study suggests.
Among the various lifestyle changes, adopting the Dietary Approaches to Stop Hypertension diet, known as the DASH diet, may have the greatest impact for young and middle-aged adults with stage 1 hypertension.
“This research reveals that we should look to feasible ways our food system could make healthy eating the default option,” Kendra Sims, PhD, MPH, postdoctoral fellow at University of California, San Francisco, told this news organization.
“Above all, it means collaborating with the patient about nourishing choices that fit best into their culture and lifestyle,” Dr. Sims said.
Be proactive
“What is important is that people not wait until they have hypertension to start thinking about healthful diets,” commented Taylor Wallace, PhD, department of nutrition and food studies, George Mason University, Fairfax, Va., who was not involved in the study.
“It’s all about prevention in my mind. Whether you are hypertensive or are perfectly healthy, the DASH diet or any other dietary pattern that emphasizes consumption of fruits, vegetables, whole grains, lean meats, seafood, nuts/seeds, and low/non-fat dairy and decreased intake of saturated fats, added sugars, and sodium is a good idea,” Dr. Wallace said in an interview.
The study was presented at the American Heart Association Hypertension Scientific Sessions 2022 in San Diego.
Dr. Sims and colleagues used U.S. statistics from multiple sources to simulate CVD events, mortality, and health care costs between 2018 and 2027 in adults aged 35-64 years with untreated stage 1 hypertension, defined as systolic BP of 130 to 139 mm Hg.
The researchers estimate that 8.8 million U.S. adults (5.5 million women) aged 35-64 years have untreated stage 1 hypertension and would be recommended for lifestyle change, such as physical activity, weight loss, moderating alcohol intake, and adoption of the DASH diet.
Controlling blood pressure to less than 130 mm Hg in this population could prevent 26,000 CVD events, avoid 2,900 deaths, and lead to $1.6 billion saved in associated health care costs, the researchers calculate.
The largest benefit would come from adoption of the DASH diet, with an estimated 15,000 CVD events prevented among men and 11,000 among women.
Even small changes can help
“Young and middle-aged adults with stage 1 hypertension aren’t as low risk as you – or even your doctor – might think,” Dr. Sims told this news organization.
“Millions of working-aged people are walking around with elevated blood pressure, which is symptomless but is also a leading preventable cause of disability and death. Most do not follow the recommended DASH diet,” Dr. Sims said.
“Unfortunately, the availability and affordability of healthy food sources does not easily allow people to follow the DASH diet,” Dr. Sims adds in a conference news release.
“Clinicians should consider whether their patients live in food deserts or places with limited walkability. Health counseling should include addressing these specific challenges to blood pressure control,” Dr. Sims says.
Dr. Wallace noted that diet changes don’t have to be drastic.
“Honestly, just increasing fruit and vegetable intake has been shown to displace calories from saturated fats, added sugars, and sodium,” he told this news organization.
“It’s hard for people to stick to ‘diets’ long-term, so shifting toward healthier dietary patterns without having to read a book on the DASH diet or count calories and carbs seems like a more practical solution for the general population, although I have no issues with the DASH diet and think it is a great dietary pattern for heart health,” Dr. Wallace said.
The study had no funding. Dr. Sims reports no relevant financial relationships. Dr. Wallace is principal and CEO of Think Healthy Group; chief food and nutrition scientist with Produce for Better Health Foundation; editor, Journal of Dietary Supplements; deputy editor, Journal of the American College of Nutrition; nutrition section editor, Annals of Medicine; and advisory board member with Forbes Health.
A version of this article first appeared on Medscape.com.
Healthy lifestyle changes to reduce systolic blood pressure to below 130 mm Hg may prevent 26,000 heart attacks and strokes and reduce health care costs over the next 10 years, a new simulation study suggests.
Among the various lifestyle changes, adopting the Dietary Approaches to Stop Hypertension diet, known as the DASH diet, may have the greatest impact for young and middle-aged adults with stage 1 hypertension.
“This research reveals that we should look to feasible ways our food system could make healthy eating the default option,” Kendra Sims, PhD, MPH, postdoctoral fellow at University of California, San Francisco, told this news organization.
“Above all, it means collaborating with the patient about nourishing choices that fit best into their culture and lifestyle,” Dr. Sims said.
Be proactive
“What is important is that people not wait until they have hypertension to start thinking about healthful diets,” commented Taylor Wallace, PhD, department of nutrition and food studies, George Mason University, Fairfax, Va., who was not involved in the study.
“It’s all about prevention in my mind. Whether you are hypertensive or are perfectly healthy, the DASH diet or any other dietary pattern that emphasizes consumption of fruits, vegetables, whole grains, lean meats, seafood, nuts/seeds, and low/non-fat dairy and decreased intake of saturated fats, added sugars, and sodium is a good idea,” Dr. Wallace said in an interview.
The study was presented at the American Heart Association Hypertension Scientific Sessions 2022 in San Diego.
Dr. Sims and colleagues used U.S. statistics from multiple sources to simulate CVD events, mortality, and health care costs between 2018 and 2027 in adults aged 35-64 years with untreated stage 1 hypertension, defined as systolic BP of 130 to 139 mm Hg.
The researchers estimate that 8.8 million U.S. adults (5.5 million women) aged 35-64 years have untreated stage 1 hypertension and would be recommended for lifestyle change, such as physical activity, weight loss, moderating alcohol intake, and adoption of the DASH diet.
Controlling blood pressure to less than 130 mm Hg in this population could prevent 26,000 CVD events, avoid 2,900 deaths, and lead to $1.6 billion saved in associated health care costs, the researchers calculate.
The largest benefit would come from adoption of the DASH diet, with an estimated 15,000 CVD events prevented among men and 11,000 among women.
Even small changes can help
“Young and middle-aged adults with stage 1 hypertension aren’t as low risk as you – or even your doctor – might think,” Dr. Sims told this news organization.
“Millions of working-aged people are walking around with elevated blood pressure, which is symptomless but is also a leading preventable cause of disability and death. Most do not follow the recommended DASH diet,” Dr. Sims said.
“Unfortunately, the availability and affordability of healthy food sources does not easily allow people to follow the DASH diet,” Dr. Sims adds in a conference news release.
“Clinicians should consider whether their patients live in food deserts or places with limited walkability. Health counseling should include addressing these specific challenges to blood pressure control,” Dr. Sims says.
Dr. Wallace noted that diet changes don’t have to be drastic.
“Honestly, just increasing fruit and vegetable intake has been shown to displace calories from saturated fats, added sugars, and sodium,” he told this news organization.
“It’s hard for people to stick to ‘diets’ long-term, so shifting toward healthier dietary patterns without having to read a book on the DASH diet or count calories and carbs seems like a more practical solution for the general population, although I have no issues with the DASH diet and think it is a great dietary pattern for heart health,” Dr. Wallace said.
The study had no funding. Dr. Sims reports no relevant financial relationships. Dr. Wallace is principal and CEO of Think Healthy Group; chief food and nutrition scientist with Produce for Better Health Foundation; editor, Journal of Dietary Supplements; deputy editor, Journal of the American College of Nutrition; nutrition section editor, Annals of Medicine; and advisory board member with Forbes Health.
A version of this article first appeared on Medscape.com.
Children and COVID: New cases took a downturn in September
After 2 weeks of increases in the number of new COVID-19 cases in children – a trend that just happened to coincide with the start of a new school year – there were fewer cases reported during the first full week of September, according to the American Academy of Pediatrics and the Children’s Hospital Association.
, the AAP and CHA said in their weekly COVID-19 report, noting also that seven states and the District of Columbia no longer update their online dashboards while others publish new data less often than every week.
The drop in new cases was accompanied by declines in emergency department visits and hospital admissions, both of which had shown some signs of resurgence in mid- to late August. The brief rise in ED visits seemed to be age-related, occurring in those aged 12 years and older but not in younger children, whose ED visit rate fell steadily through August. Through the first week of September, however, 7-day averages were down for both those aged 12-15 and for 16- to 17-year-olds, the Centers for Disease Control and Prevention reported.
The rate of new hospital admissions of children with confirmed COVID-19, available only for ages 0-17 years, has declined every day since Aug. 28, when it reached 0.44 per 100,000 population after a week of climbing, the CDC said on its COVID Data Tracker.
Cumulatively, about 156,000 children were hospitalized with COVID from Aug. 1, 2020 to Sept. 10, 2022, according to the CDC, which puts the total number of pediatric cases at just over 15 million and deaths at 1,778. Those last two figures represent 17.4% and about 0.4% of all U.S. cases and deaths. The AAP and CHA estimate that about 14.6 million child cases have been reported so far, which is 18.4% of cases in all ages.
Vaccinations are slowly adding up
On the prevention side of the health care system’s response to COVID, the CDC’s cumulative numbers looked like this as of Sept. 6:
- 1.1 million children under age 5 (about 5.8% of the age group) had received at least one dose of vaccine, and 280,000 (1.4%) were fully vaccinated.
- Almost 11 million (38.2%) children aged 5-11 had gotten one dose, and 8.9 million (31.1%) were fully vaccinated.
- 17.9 million (70.8%) children aged 12-17 had received at least one dose, and 15.3 million (60.5%) were fully vaccinated.
Over the 14 days ending Sept. 7, children aged 2-4 years made up the largest group (21.4%) of Americans getting their first vaccine doses, while those aged 5-11 years were the third largest age group at 16.7% of all vaccinees (25- to 49-year-olds were second). The situation was reversed for vaccine completion over the last 2 weeks: Those aged 5-11 were first at 24.7%, and the 2- to 4-year-olds were third at 16.7% (those aged 25-49 were second again), according to the COVID Data Tracker.
After 2 weeks of increases in the number of new COVID-19 cases in children – a trend that just happened to coincide with the start of a new school year – there were fewer cases reported during the first full week of September, according to the American Academy of Pediatrics and the Children’s Hospital Association.
, the AAP and CHA said in their weekly COVID-19 report, noting also that seven states and the District of Columbia no longer update their online dashboards while others publish new data less often than every week.
The drop in new cases was accompanied by declines in emergency department visits and hospital admissions, both of which had shown some signs of resurgence in mid- to late August. The brief rise in ED visits seemed to be age-related, occurring in those aged 12 years and older but not in younger children, whose ED visit rate fell steadily through August. Through the first week of September, however, 7-day averages were down for both those aged 12-15 and for 16- to 17-year-olds, the Centers for Disease Control and Prevention reported.
The rate of new hospital admissions of children with confirmed COVID-19, available only for ages 0-17 years, has declined every day since Aug. 28, when it reached 0.44 per 100,000 population after a week of climbing, the CDC said on its COVID Data Tracker.
Cumulatively, about 156,000 children were hospitalized with COVID from Aug. 1, 2020 to Sept. 10, 2022, according to the CDC, which puts the total number of pediatric cases at just over 15 million and deaths at 1,778. Those last two figures represent 17.4% and about 0.4% of all U.S. cases and deaths. The AAP and CHA estimate that about 14.6 million child cases have been reported so far, which is 18.4% of cases in all ages.
Vaccinations are slowly adding up
On the prevention side of the health care system’s response to COVID, the CDC’s cumulative numbers looked like this as of Sept. 6:
- 1.1 million children under age 5 (about 5.8% of the age group) had received at least one dose of vaccine, and 280,000 (1.4%) were fully vaccinated.
- Almost 11 million (38.2%) children aged 5-11 had gotten one dose, and 8.9 million (31.1%) were fully vaccinated.
- 17.9 million (70.8%) children aged 12-17 had received at least one dose, and 15.3 million (60.5%) were fully vaccinated.
Over the 14 days ending Sept. 7, children aged 2-4 years made up the largest group (21.4%) of Americans getting their first vaccine doses, while those aged 5-11 years were the third largest age group at 16.7% of all vaccinees (25- to 49-year-olds were second). The situation was reversed for vaccine completion over the last 2 weeks: Those aged 5-11 were first at 24.7%, and the 2- to 4-year-olds were third at 16.7% (those aged 25-49 were second again), according to the COVID Data Tracker.
After 2 weeks of increases in the number of new COVID-19 cases in children – a trend that just happened to coincide with the start of a new school year – there were fewer cases reported during the first full week of September, according to the American Academy of Pediatrics and the Children’s Hospital Association.
, the AAP and CHA said in their weekly COVID-19 report, noting also that seven states and the District of Columbia no longer update their online dashboards while others publish new data less often than every week.
The drop in new cases was accompanied by declines in emergency department visits and hospital admissions, both of which had shown some signs of resurgence in mid- to late August. The brief rise in ED visits seemed to be age-related, occurring in those aged 12 years and older but not in younger children, whose ED visit rate fell steadily through August. Through the first week of September, however, 7-day averages were down for both those aged 12-15 and for 16- to 17-year-olds, the Centers for Disease Control and Prevention reported.
The rate of new hospital admissions of children with confirmed COVID-19, available only for ages 0-17 years, has declined every day since Aug. 28, when it reached 0.44 per 100,000 population after a week of climbing, the CDC said on its COVID Data Tracker.
Cumulatively, about 156,000 children were hospitalized with COVID from Aug. 1, 2020 to Sept. 10, 2022, according to the CDC, which puts the total number of pediatric cases at just over 15 million and deaths at 1,778. Those last two figures represent 17.4% and about 0.4% of all U.S. cases and deaths. The AAP and CHA estimate that about 14.6 million child cases have been reported so far, which is 18.4% of cases in all ages.
Vaccinations are slowly adding up
On the prevention side of the health care system’s response to COVID, the CDC’s cumulative numbers looked like this as of Sept. 6:
- 1.1 million children under age 5 (about 5.8% of the age group) had received at least one dose of vaccine, and 280,000 (1.4%) were fully vaccinated.
- Almost 11 million (38.2%) children aged 5-11 had gotten one dose, and 8.9 million (31.1%) were fully vaccinated.
- 17.9 million (70.8%) children aged 12-17 had received at least one dose, and 15.3 million (60.5%) were fully vaccinated.
Over the 14 days ending Sept. 7, children aged 2-4 years made up the largest group (21.4%) of Americans getting their first vaccine doses, while those aged 5-11 years were the third largest age group at 16.7% of all vaccinees (25- to 49-year-olds were second). The situation was reversed for vaccine completion over the last 2 weeks: Those aged 5-11 were first at 24.7%, and the 2- to 4-year-olds were third at 16.7% (those aged 25-49 were second again), according to the COVID Data Tracker.
Sleep Medicine Network
Nonrespiratory Sleep Section
Sleep in cancer patients
Sleep disturbance is among the most common symptoms in patients with cancer with an estimated prevalence of up to two out of three patients experiencing sleep disruption during their cancer journey.1,23,4
Common sleep disorders in cancer patients:
Insomnia: Cancer patients have at least a two-fold higher incidence of insomnia compared with the general population.5,6 Predisposing factors may include age, the presence of hyper-arousability,a prior history of insomnia, or a preexisting psychiatric disorder. Cancer-related factors include surgery, hospitalization, chemotherapy, hormonal therapy, radiation therapy, and use of steroids.7 If sedative-hypnotics are considered, they should be used in conjunction with cognitive and behavioral therapy for insomnia (CBT-I). Recent meta-analyses provide data to support a strong recommendation to utilize CBT-I to treat insomnia in cancer patients.6,8,9
Hypersomnolence: Hypersomnolence or excessive daytime sleepiness is a common symptom noted among cancer patients.10 Hypersomnia related to cancer can be often classified as either hypersomnia due to a medical condition or hypersomnia due to a drug or substance, especially for those patients taking opioid or other sedative medications.
Movement Disorders: Sleep movement disorders occur in patients with cancer and may be primary or attributable to chemotherapy-related neuropathy from therapy regimens, including platinum compounds, taxanes, vinca alkaloids, proteasome inhibitors, or thalidomide-based agents.11,12
Obstructive sleep apnea (OSA): OSA occurs in patients with cancer and may be increased in patients with specific cancers such as head and neck tumors.13 Patients with sleep apnea have a five-fold increased risk of cancer-related mortality, and several studies show an increased incidence of cancer in those with sleep apnea.14-16There is an increasing realization that not only sleep apnea, but sleep disturbance, in general, may be oncogenic based on increased autonomic tone, chronic stress, variation in the pituitary-hypothalamic axis, as well as circadian mechanisms.17
Early recognition/treatment of sleep issues is essential to improve quality of life in cancer patients.
Diwakar Balachandran, MD, FCCP
Member-at-Large
References
1. Balachandran DD, Miller MA, Faiz SA, Yennurajalingam S, Innominato PF. Evaluation and management of sleep and circadian rhythm disturbance in cancer. Curr Treat Options Oncol. 2021;22(9):81.
2. Yennurajalingam S, Balachandran D, Pedraza Cardozo SL, et al. Patient-reported sleep disturbance in advanced cancer: frequency, predictors and screening performance of the Edmonton Symptom Assessment System sleep item. BMJ Support Palliat Care. 2017;7(3):274-80.
3. Harris B, Ross J, Sanchez-Reilly S. Sleeping in the arms of cancer: A review of sleeping disorders among patients with cancer. Cancer J. 2014;20(5):299-305.
4. Charalambous A, Berger AM, Matthews E, Balachandran DD, Papastavrou E, Palesh O. Cancer-related fatigue and sleep deficiency in cancer care continuum: concepts, assessment, clusters, and management. Support Care Cancer. 2019;27(7):2747-53.
5. Palesh OG, Roscoe JA, Mustian KM, et al. Prevalence, demographics, and psychological associations of sleep disruption in patients with cancer: University of Rochester Cancer Center-Community Clinical Oncology Program. J Clin Oncol. 2010;28(2):292-8.
6. Savard J, Simard S, Blanchet J, Ivers H, Morin CM. Prevalence, clinical characteristics, and risk factors for insomnia in the context of breast cancer. Sleep. 2001;24(5):583-90.
7. Savard J, Morin CM. Insomnia in the context of cancer: a review of a neglected problem. J Clin Oncol. 2001;19(3):895-908.
8. Garland SN, Johnson JA, Savard J, et al. Sleeping well with cancer: a systematic review of cognitive behavioral therapy for insomnia in cancer patients. Neuropsychiatr Dis Treat. 2014;10:1113-24.
9. Johnson JA, Rash JA, Campbell TS, et al. A systematic review and meta-analysis of randomized controlled trials of cognitive behavior therapy for insomnia (CBT-I) in cancer survivors. Sleep Med Rev. 2016;27:20-8.
10. Jaumally BA, Das A, Cassell NC, et al. Excessive daytime sleepiness in cancer patients. Sleep Breath. 2021;25(2):1063-7.
11. Gewandter JS, Kleckner AS, Marshall JH, et al. Chemotherapy-induced peripheral neuropathy (CIPN) and its treatment: an NIH Collaboratory study of claims data. Support Care Cancer. 2020;28(6):2553-62.
12. St Germain DC, O’Mara AM, Robinson JL, Torres AD, Minasian LM. Chemotherapy-induced peripheral neuropathy: Identifying the research gaps and associated changes to clinical trial design. Cancer. 2020;126(20):4602-13.
13. Faiz SA, Balachandran D, Hessel AC, et al. Sleep-related breathing disorders in patients with tumors in the head and neck region. Oncologist. 2014;19(11):1200-6.
14. Campos-Rodriguez F, Martinez-Garcia MA, Martinez M, et al. Association between obstructive sleep apnea and cancer incidence in a large multicenter Spanish cohort. Am J Respir Crit Care Med. 2013;187(1):99-105.
15. Martinez-Garcia MA, Campos-Rodriguez F, Duran-Cantolla J, et al. Obstructive sleep apnea is associated with cancer mortality in younger patients. Sleep Med. 2014;15(7):742-8.
16. Martinez-Garcia MA, Campos-Rodriguez F, Barbe F. Cancer and OSA: Current evidence from human studies. Chest. 2016;150(2):451-63.
17. Gozal D, Farre R, Nieto FJ. Putative links between sleep apnea and cancer: From hypotheses to evolving evidence. Chest. 2015;148(5):1140-7.
Nonrespiratory Sleep Section
Sleep in cancer patients
Sleep disturbance is among the most common symptoms in patients with cancer with an estimated prevalence of up to two out of three patients experiencing sleep disruption during their cancer journey.1,23,4
Common sleep disorders in cancer patients:
Insomnia: Cancer patients have at least a two-fold higher incidence of insomnia compared with the general population.5,6 Predisposing factors may include age, the presence of hyper-arousability,a prior history of insomnia, or a preexisting psychiatric disorder. Cancer-related factors include surgery, hospitalization, chemotherapy, hormonal therapy, radiation therapy, and use of steroids.7 If sedative-hypnotics are considered, they should be used in conjunction with cognitive and behavioral therapy for insomnia (CBT-I). Recent meta-analyses provide data to support a strong recommendation to utilize CBT-I to treat insomnia in cancer patients.6,8,9
Hypersomnolence: Hypersomnolence or excessive daytime sleepiness is a common symptom noted among cancer patients.10 Hypersomnia related to cancer can be often classified as either hypersomnia due to a medical condition or hypersomnia due to a drug or substance, especially for those patients taking opioid or other sedative medications.
Movement Disorders: Sleep movement disorders occur in patients with cancer and may be primary or attributable to chemotherapy-related neuropathy from therapy regimens, including platinum compounds, taxanes, vinca alkaloids, proteasome inhibitors, or thalidomide-based agents.11,12
Obstructive sleep apnea (OSA): OSA occurs in patients with cancer and may be increased in patients with specific cancers such as head and neck tumors.13 Patients with sleep apnea have a five-fold increased risk of cancer-related mortality, and several studies show an increased incidence of cancer in those with sleep apnea.14-16There is an increasing realization that not only sleep apnea, but sleep disturbance, in general, may be oncogenic based on increased autonomic tone, chronic stress, variation in the pituitary-hypothalamic axis, as well as circadian mechanisms.17
Early recognition/treatment of sleep issues is essential to improve quality of life in cancer patients.
Diwakar Balachandran, MD, FCCP
Member-at-Large
References
1. Balachandran DD, Miller MA, Faiz SA, Yennurajalingam S, Innominato PF. Evaluation and management of sleep and circadian rhythm disturbance in cancer. Curr Treat Options Oncol. 2021;22(9):81.
2. Yennurajalingam S, Balachandran D, Pedraza Cardozo SL, et al. Patient-reported sleep disturbance in advanced cancer: frequency, predictors and screening performance of the Edmonton Symptom Assessment System sleep item. BMJ Support Palliat Care. 2017;7(3):274-80.
3. Harris B, Ross J, Sanchez-Reilly S. Sleeping in the arms of cancer: A review of sleeping disorders among patients with cancer. Cancer J. 2014;20(5):299-305.
4. Charalambous A, Berger AM, Matthews E, Balachandran DD, Papastavrou E, Palesh O. Cancer-related fatigue and sleep deficiency in cancer care continuum: concepts, assessment, clusters, and management. Support Care Cancer. 2019;27(7):2747-53.
5. Palesh OG, Roscoe JA, Mustian KM, et al. Prevalence, demographics, and psychological associations of sleep disruption in patients with cancer: University of Rochester Cancer Center-Community Clinical Oncology Program. J Clin Oncol. 2010;28(2):292-8.
6. Savard J, Simard S, Blanchet J, Ivers H, Morin CM. Prevalence, clinical characteristics, and risk factors for insomnia in the context of breast cancer. Sleep. 2001;24(5):583-90.
7. Savard J, Morin CM. Insomnia in the context of cancer: a review of a neglected problem. J Clin Oncol. 2001;19(3):895-908.
8. Garland SN, Johnson JA, Savard J, et al. Sleeping well with cancer: a systematic review of cognitive behavioral therapy for insomnia in cancer patients. Neuropsychiatr Dis Treat. 2014;10:1113-24.
9. Johnson JA, Rash JA, Campbell TS, et al. A systematic review and meta-analysis of randomized controlled trials of cognitive behavior therapy for insomnia (CBT-I) in cancer survivors. Sleep Med Rev. 2016;27:20-8.
10. Jaumally BA, Das A, Cassell NC, et al. Excessive daytime sleepiness in cancer patients. Sleep Breath. 2021;25(2):1063-7.
11. Gewandter JS, Kleckner AS, Marshall JH, et al. Chemotherapy-induced peripheral neuropathy (CIPN) and its treatment: an NIH Collaboratory study of claims data. Support Care Cancer. 2020;28(6):2553-62.
12. St Germain DC, O’Mara AM, Robinson JL, Torres AD, Minasian LM. Chemotherapy-induced peripheral neuropathy: Identifying the research gaps and associated changes to clinical trial design. Cancer. 2020;126(20):4602-13.
13. Faiz SA, Balachandran D, Hessel AC, et al. Sleep-related breathing disorders in patients with tumors in the head and neck region. Oncologist. 2014;19(11):1200-6.
14. Campos-Rodriguez F, Martinez-Garcia MA, Martinez M, et al. Association between obstructive sleep apnea and cancer incidence in a large multicenter Spanish cohort. Am J Respir Crit Care Med. 2013;187(1):99-105.
15. Martinez-Garcia MA, Campos-Rodriguez F, Duran-Cantolla J, et al. Obstructive sleep apnea is associated with cancer mortality in younger patients. Sleep Med. 2014;15(7):742-8.
16. Martinez-Garcia MA, Campos-Rodriguez F, Barbe F. Cancer and OSA: Current evidence from human studies. Chest. 2016;150(2):451-63.
17. Gozal D, Farre R, Nieto FJ. Putative links between sleep apnea and cancer: From hypotheses to evolving evidence. Chest. 2015;148(5):1140-7.
Nonrespiratory Sleep Section
Sleep in cancer patients
Sleep disturbance is among the most common symptoms in patients with cancer with an estimated prevalence of up to two out of three patients experiencing sleep disruption during their cancer journey.1,23,4
Common sleep disorders in cancer patients:
Insomnia: Cancer patients have at least a two-fold higher incidence of insomnia compared with the general population.5,6 Predisposing factors may include age, the presence of hyper-arousability,a prior history of insomnia, or a preexisting psychiatric disorder. Cancer-related factors include surgery, hospitalization, chemotherapy, hormonal therapy, radiation therapy, and use of steroids.7 If sedative-hypnotics are considered, they should be used in conjunction with cognitive and behavioral therapy for insomnia (CBT-I). Recent meta-analyses provide data to support a strong recommendation to utilize CBT-I to treat insomnia in cancer patients.6,8,9
Hypersomnolence: Hypersomnolence or excessive daytime sleepiness is a common symptom noted among cancer patients.10 Hypersomnia related to cancer can be often classified as either hypersomnia due to a medical condition or hypersomnia due to a drug or substance, especially for those patients taking opioid or other sedative medications.
Movement Disorders: Sleep movement disorders occur in patients with cancer and may be primary or attributable to chemotherapy-related neuropathy from therapy regimens, including platinum compounds, taxanes, vinca alkaloids, proteasome inhibitors, or thalidomide-based agents.11,12
Obstructive sleep apnea (OSA): OSA occurs in patients with cancer and may be increased in patients with specific cancers such as head and neck tumors.13 Patients with sleep apnea have a five-fold increased risk of cancer-related mortality, and several studies show an increased incidence of cancer in those with sleep apnea.14-16There is an increasing realization that not only sleep apnea, but sleep disturbance, in general, may be oncogenic based on increased autonomic tone, chronic stress, variation in the pituitary-hypothalamic axis, as well as circadian mechanisms.17
Early recognition/treatment of sleep issues is essential to improve quality of life in cancer patients.
Diwakar Balachandran, MD, FCCP
Member-at-Large
References
1. Balachandran DD, Miller MA, Faiz SA, Yennurajalingam S, Innominato PF. Evaluation and management of sleep and circadian rhythm disturbance in cancer. Curr Treat Options Oncol. 2021;22(9):81.
2. Yennurajalingam S, Balachandran D, Pedraza Cardozo SL, et al. Patient-reported sleep disturbance in advanced cancer: frequency, predictors and screening performance of the Edmonton Symptom Assessment System sleep item. BMJ Support Palliat Care. 2017;7(3):274-80.
3. Harris B, Ross J, Sanchez-Reilly S. Sleeping in the arms of cancer: A review of sleeping disorders among patients with cancer. Cancer J. 2014;20(5):299-305.
4. Charalambous A, Berger AM, Matthews E, Balachandran DD, Papastavrou E, Palesh O. Cancer-related fatigue and sleep deficiency in cancer care continuum: concepts, assessment, clusters, and management. Support Care Cancer. 2019;27(7):2747-53.
5. Palesh OG, Roscoe JA, Mustian KM, et al. Prevalence, demographics, and psychological associations of sleep disruption in patients with cancer: University of Rochester Cancer Center-Community Clinical Oncology Program. J Clin Oncol. 2010;28(2):292-8.
6. Savard J, Simard S, Blanchet J, Ivers H, Morin CM. Prevalence, clinical characteristics, and risk factors for insomnia in the context of breast cancer. Sleep. 2001;24(5):583-90.
7. Savard J, Morin CM. Insomnia in the context of cancer: a review of a neglected problem. J Clin Oncol. 2001;19(3):895-908.
8. Garland SN, Johnson JA, Savard J, et al. Sleeping well with cancer: a systematic review of cognitive behavioral therapy for insomnia in cancer patients. Neuropsychiatr Dis Treat. 2014;10:1113-24.
9. Johnson JA, Rash JA, Campbell TS, et al. A systematic review and meta-analysis of randomized controlled trials of cognitive behavior therapy for insomnia (CBT-I) in cancer survivors. Sleep Med Rev. 2016;27:20-8.
10. Jaumally BA, Das A, Cassell NC, et al. Excessive daytime sleepiness in cancer patients. Sleep Breath. 2021;25(2):1063-7.
11. Gewandter JS, Kleckner AS, Marshall JH, et al. Chemotherapy-induced peripheral neuropathy (CIPN) and its treatment: an NIH Collaboratory study of claims data. Support Care Cancer. 2020;28(6):2553-62.
12. St Germain DC, O’Mara AM, Robinson JL, Torres AD, Minasian LM. Chemotherapy-induced peripheral neuropathy: Identifying the research gaps and associated changes to clinical trial design. Cancer. 2020;126(20):4602-13.
13. Faiz SA, Balachandran D, Hessel AC, et al. Sleep-related breathing disorders in patients with tumors in the head and neck region. Oncologist. 2014;19(11):1200-6.
14. Campos-Rodriguez F, Martinez-Garcia MA, Martinez M, et al. Association between obstructive sleep apnea and cancer incidence in a large multicenter Spanish cohort. Am J Respir Crit Care Med. 2013;187(1):99-105.
15. Martinez-Garcia MA, Campos-Rodriguez F, Duran-Cantolla J, et al. Obstructive sleep apnea is associated with cancer mortality in younger patients. Sleep Med. 2014;15(7):742-8.
16. Martinez-Garcia MA, Campos-Rodriguez F, Barbe F. Cancer and OSA: Current evidence from human studies. Chest. 2016;150(2):451-63.
17. Gozal D, Farre R, Nieto FJ. Putative links between sleep apnea and cancer: From hypotheses to evolving evidence. Chest. 2015;148(5):1140-7.
Integrase inhibitors and gestational weight gain: Should women worry?
In recent years, increased use of integrase strand transferase inhibitor (INSTI) antiviral treatment (ART) has raised concerns about weight gain and adverse outcomes in patients with HIV. This is especially true regarding possible excessive gestational weight gain, which in women without HIV has been associated with maternal gestational diabetes, hypertensive and liver conditions, as well as related risks for preterm birth, fetal macrosomia, and higher weight after birth.
Unfortunately, few studies in pregnant women with HIV have moved out of the controlled environment into real-world settings, potentially limiting current knowledge about the impact of gestational weight gain – as well as strategies to both prevent it and the associated adverse outcomes.
That is what a team of infectious disease specialists at the Hospital Federal dos Servidores do Estado in Rio de Janeiro recently sought to answer among a cohort of INSTI-experienced and INSTI-naive women with BMIs less than 25 kg/m2 (underweight/normal weight) and higher than 25 kg/m2.
Surprising findings
The investigators determined that rates of excessive weight gain were significantly higher in INSTI-naive women with BMI less than 25 who experienced rates as high as 31.6%, compared with approximately 12% of women who conceived while on INSTIs, regardless of BMI values at baseline (P = .004).
However, rates of unfavorable pregnancy outcomes (for example, small for gestational age, preterm birth, stillbirth, death) appeared to be low overall and similar among all the study groups.
“We had some discussions when we were working on this and thought that the weight gain might have adverse effects,” Trevon Fuller, PhD, lead author and a postdoctoral student at the Hospital Federal dos Servidores do Estado, told this news organization.
“But it looked like the weight gain might actually be good, to the extent that we didn’t see any harm to the mom or the baby of those underweight or normal weight women who were naive to INSTIs,” he explained.
Dr. Fuller and his team enrolled 198 pregnant women living with HIV who sought care at the Hospital Federal dos Servidores do Estado – a national reference center for USAID’s Prevention of Mother to Child Transmission strategic program – between October 2014 and October 2021.
Participants were divided into two primary cohorts: BMI less than 25 at enrollment (n = 74) or BMI of 25 or higher (n = 124), then further divided by timing of INSTI-based combined ART:
- INSTI-naive: women using INSTI-based ART (raltegravir [Isentress] 400 mg twice per day or dolutegravir [Tivicay] 50 mg/day plus 2 non-nucleoside reverse transcriptase inhibitors – lamivudine plus tenofovir disoproxil fumarate or lamivudine plus zidovudine) for 4 weeks between baseline and near delivery.
- INSTI-experienced: women who became pregnant while using INSTIs for at least 6 months before conception.
Among underweight/normal weight participants, 77% (n = 57) were INSTI-naive and 23% (n = 17) INSTI-experienced, and among overweight/obese participants, 81.5% (n = 101) were INSTI-naive, and 18.5% (n = 23) were experienced.
Maternal age, which did not differ significantly by BMI or treatment experience, was a median of 28 years, and most participants were non-White. All participants were virally suppressed near delivery.
Study findings, which were published online in HIV Medicine, highlighted that median weight near delivery in participants who were overweight/obese at baseline was similar regardless of whether they were treatment-experienced (90 kg [198 lb]) or treatment-naive (82.3 kg [181 lb]), P = .026.
However, participants who were underweight/normal weight who were INSTI-naive had significantly higher rates of gestational weight gain (31.5%, 18/57), compared with those of underweight/normal weight who were INSTI-experienced (11.8%, 2/17), P = .004. Notably, this gain was significant in all categories of change (that is, low < 0.18 kg/week, normal 0.18-0.59 kg/week), and high > 0.59 kg/week).
“One of the things that we took away was that this weight gain is primarily happening with women who are starting INSTIs,” said Dr. Fuller.
“The data suggest that [it] might be temporary in the sense that there’s not going to be continuous weight gain but that it will probably approach some type of horizontal asymptote,” he added.
Although obstetric and neonatal outcomes were secondary measures, the investigators did not observe any significantly different outcomes when comparing the groups, and there were no stillbirths, neonatal deaths, or macrosomia.
Preterm delivery rates in underweight/normal weight participants who were INSTI-experienced (11.8%, 2/17) and INSTI-naive (5.3%, 3/57) were similar to overweight/obese participants who were INSTI-experienced (13%, 3/23) and INSTI-naive (6.9%, 7/101).
The same was true for low birthweight.
Still, the study appears to raise more questions than it answers, Sigal Yawetz, MD, an infectious disease specialist at Brigham and Women’s Hospital, Boston, said in an interview – a factor that she said is common also in some of the more recent randomized controlled studies, such as IMPAACT PROMISE.
Dr. Yawetz, who was not involved in the study, also noted, “The groups were small, so comparisons within the groups are difficult, and so many people were excluded that it’s hard to know if there were adverse outcomes related to this ... It’s very confounded.”
The World Health Organization estimates that there are roughly 1.3 million pregnant women with HIV, 81% of whom are on antiretroviral therapy. Although the literature continues to evolve, data suggest that in general, Black women are at greater risk for gestational weight gain.
“We have to remember that women who gain excess weight in pregnancy are still going to be with this weight following pregnancy as well,” Dr. Yawetz said. “So, it might impact their pregnancy but also their health after delivery and for subsequent pregnancies, which we don’t have data for yet.”
Dr. Fuller agrees that more data are needed and mentioned that the team plans to study this further, ideally with larger sample sizes.
Yet, despite the lingering questions, there is a silver lining, one that Dr. Yawetz was emphatic about.
“I really welcome people doing studies on this because we really need the data. By far, integrase inhibitors are the first-line regimen all over the world for pregnant women, and if you look at the gestalt or full picture, this is the best regimen to give pregnant women,” she said.
Dr. Fuller and Dr. Yawetz report no relevant financial relationships. The study was independently supported.
A version of this article first appeared on Medscape.com.
In recent years, increased use of integrase strand transferase inhibitor (INSTI) antiviral treatment (ART) has raised concerns about weight gain and adverse outcomes in patients with HIV. This is especially true regarding possible excessive gestational weight gain, which in women without HIV has been associated with maternal gestational diabetes, hypertensive and liver conditions, as well as related risks for preterm birth, fetal macrosomia, and higher weight after birth.
Unfortunately, few studies in pregnant women with HIV have moved out of the controlled environment into real-world settings, potentially limiting current knowledge about the impact of gestational weight gain – as well as strategies to both prevent it and the associated adverse outcomes.
That is what a team of infectious disease specialists at the Hospital Federal dos Servidores do Estado in Rio de Janeiro recently sought to answer among a cohort of INSTI-experienced and INSTI-naive women with BMIs less than 25 kg/m2 (underweight/normal weight) and higher than 25 kg/m2.
Surprising findings
The investigators determined that rates of excessive weight gain were significantly higher in INSTI-naive women with BMI less than 25 who experienced rates as high as 31.6%, compared with approximately 12% of women who conceived while on INSTIs, regardless of BMI values at baseline (P = .004).
However, rates of unfavorable pregnancy outcomes (for example, small for gestational age, preterm birth, stillbirth, death) appeared to be low overall and similar among all the study groups.
“We had some discussions when we were working on this and thought that the weight gain might have adverse effects,” Trevon Fuller, PhD, lead author and a postdoctoral student at the Hospital Federal dos Servidores do Estado, told this news organization.
“But it looked like the weight gain might actually be good, to the extent that we didn’t see any harm to the mom or the baby of those underweight or normal weight women who were naive to INSTIs,” he explained.
Dr. Fuller and his team enrolled 198 pregnant women living with HIV who sought care at the Hospital Federal dos Servidores do Estado – a national reference center for USAID’s Prevention of Mother to Child Transmission strategic program – between October 2014 and October 2021.
Participants were divided into two primary cohorts: BMI less than 25 at enrollment (n = 74) or BMI of 25 or higher (n = 124), then further divided by timing of INSTI-based combined ART:
- INSTI-naive: women using INSTI-based ART (raltegravir [Isentress] 400 mg twice per day or dolutegravir [Tivicay] 50 mg/day plus 2 non-nucleoside reverse transcriptase inhibitors – lamivudine plus tenofovir disoproxil fumarate or lamivudine plus zidovudine) for 4 weeks between baseline and near delivery.
- INSTI-experienced: women who became pregnant while using INSTIs for at least 6 months before conception.
Among underweight/normal weight participants, 77% (n = 57) were INSTI-naive and 23% (n = 17) INSTI-experienced, and among overweight/obese participants, 81.5% (n = 101) were INSTI-naive, and 18.5% (n = 23) were experienced.
Maternal age, which did not differ significantly by BMI or treatment experience, was a median of 28 years, and most participants were non-White. All participants were virally suppressed near delivery.
Study findings, which were published online in HIV Medicine, highlighted that median weight near delivery in participants who were overweight/obese at baseline was similar regardless of whether they were treatment-experienced (90 kg [198 lb]) or treatment-naive (82.3 kg [181 lb]), P = .026.
However, participants who were underweight/normal weight who were INSTI-naive had significantly higher rates of gestational weight gain (31.5%, 18/57), compared with those of underweight/normal weight who were INSTI-experienced (11.8%, 2/17), P = .004. Notably, this gain was significant in all categories of change (that is, low < 0.18 kg/week, normal 0.18-0.59 kg/week), and high > 0.59 kg/week).
“One of the things that we took away was that this weight gain is primarily happening with women who are starting INSTIs,” said Dr. Fuller.
“The data suggest that [it] might be temporary in the sense that there’s not going to be continuous weight gain but that it will probably approach some type of horizontal asymptote,” he added.
Although obstetric and neonatal outcomes were secondary measures, the investigators did not observe any significantly different outcomes when comparing the groups, and there were no stillbirths, neonatal deaths, or macrosomia.
Preterm delivery rates in underweight/normal weight participants who were INSTI-experienced (11.8%, 2/17) and INSTI-naive (5.3%, 3/57) were similar to overweight/obese participants who were INSTI-experienced (13%, 3/23) and INSTI-naive (6.9%, 7/101).
The same was true for low birthweight.
Still, the study appears to raise more questions than it answers, Sigal Yawetz, MD, an infectious disease specialist at Brigham and Women’s Hospital, Boston, said in an interview – a factor that she said is common also in some of the more recent randomized controlled studies, such as IMPAACT PROMISE.
Dr. Yawetz, who was not involved in the study, also noted, “The groups were small, so comparisons within the groups are difficult, and so many people were excluded that it’s hard to know if there were adverse outcomes related to this ... It’s very confounded.”
The World Health Organization estimates that there are roughly 1.3 million pregnant women with HIV, 81% of whom are on antiretroviral therapy. Although the literature continues to evolve, data suggest that in general, Black women are at greater risk for gestational weight gain.
“We have to remember that women who gain excess weight in pregnancy are still going to be with this weight following pregnancy as well,” Dr. Yawetz said. “So, it might impact their pregnancy but also their health after delivery and for subsequent pregnancies, which we don’t have data for yet.”
Dr. Fuller agrees that more data are needed and mentioned that the team plans to study this further, ideally with larger sample sizes.
Yet, despite the lingering questions, there is a silver lining, one that Dr. Yawetz was emphatic about.
“I really welcome people doing studies on this because we really need the data. By far, integrase inhibitors are the first-line regimen all over the world for pregnant women, and if you look at the gestalt or full picture, this is the best regimen to give pregnant women,” she said.
Dr. Fuller and Dr. Yawetz report no relevant financial relationships. The study was independently supported.
A version of this article first appeared on Medscape.com.
In recent years, increased use of integrase strand transferase inhibitor (INSTI) antiviral treatment (ART) has raised concerns about weight gain and adverse outcomes in patients with HIV. This is especially true regarding possible excessive gestational weight gain, which in women without HIV has been associated with maternal gestational diabetes, hypertensive and liver conditions, as well as related risks for preterm birth, fetal macrosomia, and higher weight after birth.
Unfortunately, few studies in pregnant women with HIV have moved out of the controlled environment into real-world settings, potentially limiting current knowledge about the impact of gestational weight gain – as well as strategies to both prevent it and the associated adverse outcomes.
That is what a team of infectious disease specialists at the Hospital Federal dos Servidores do Estado in Rio de Janeiro recently sought to answer among a cohort of INSTI-experienced and INSTI-naive women with BMIs less than 25 kg/m2 (underweight/normal weight) and higher than 25 kg/m2.
Surprising findings
The investigators determined that rates of excessive weight gain were significantly higher in INSTI-naive women with BMI less than 25 who experienced rates as high as 31.6%, compared with approximately 12% of women who conceived while on INSTIs, regardless of BMI values at baseline (P = .004).
However, rates of unfavorable pregnancy outcomes (for example, small for gestational age, preterm birth, stillbirth, death) appeared to be low overall and similar among all the study groups.
“We had some discussions when we were working on this and thought that the weight gain might have adverse effects,” Trevon Fuller, PhD, lead author and a postdoctoral student at the Hospital Federal dos Servidores do Estado, told this news organization.
“But it looked like the weight gain might actually be good, to the extent that we didn’t see any harm to the mom or the baby of those underweight or normal weight women who were naive to INSTIs,” he explained.
Dr. Fuller and his team enrolled 198 pregnant women living with HIV who sought care at the Hospital Federal dos Servidores do Estado – a national reference center for USAID’s Prevention of Mother to Child Transmission strategic program – between October 2014 and October 2021.
Participants were divided into two primary cohorts: BMI less than 25 at enrollment (n = 74) or BMI of 25 or higher (n = 124), then further divided by timing of INSTI-based combined ART:
- INSTI-naive: women using INSTI-based ART (raltegravir [Isentress] 400 mg twice per day or dolutegravir [Tivicay] 50 mg/day plus 2 non-nucleoside reverse transcriptase inhibitors – lamivudine plus tenofovir disoproxil fumarate or lamivudine plus zidovudine) for 4 weeks between baseline and near delivery.
- INSTI-experienced: women who became pregnant while using INSTIs for at least 6 months before conception.
Among underweight/normal weight participants, 77% (n = 57) were INSTI-naive and 23% (n = 17) INSTI-experienced, and among overweight/obese participants, 81.5% (n = 101) were INSTI-naive, and 18.5% (n = 23) were experienced.
Maternal age, which did not differ significantly by BMI or treatment experience, was a median of 28 years, and most participants were non-White. All participants were virally suppressed near delivery.
Study findings, which were published online in HIV Medicine, highlighted that median weight near delivery in participants who were overweight/obese at baseline was similar regardless of whether they were treatment-experienced (90 kg [198 lb]) or treatment-naive (82.3 kg [181 lb]), P = .026.
However, participants who were underweight/normal weight who were INSTI-naive had significantly higher rates of gestational weight gain (31.5%, 18/57), compared with those of underweight/normal weight who were INSTI-experienced (11.8%, 2/17), P = .004. Notably, this gain was significant in all categories of change (that is, low < 0.18 kg/week, normal 0.18-0.59 kg/week), and high > 0.59 kg/week).
“One of the things that we took away was that this weight gain is primarily happening with women who are starting INSTIs,” said Dr. Fuller.
“The data suggest that [it] might be temporary in the sense that there’s not going to be continuous weight gain but that it will probably approach some type of horizontal asymptote,” he added.
Although obstetric and neonatal outcomes were secondary measures, the investigators did not observe any significantly different outcomes when comparing the groups, and there were no stillbirths, neonatal deaths, or macrosomia.
Preterm delivery rates in underweight/normal weight participants who were INSTI-experienced (11.8%, 2/17) and INSTI-naive (5.3%, 3/57) were similar to overweight/obese participants who were INSTI-experienced (13%, 3/23) and INSTI-naive (6.9%, 7/101).
The same was true for low birthweight.
Still, the study appears to raise more questions than it answers, Sigal Yawetz, MD, an infectious disease specialist at Brigham and Women’s Hospital, Boston, said in an interview – a factor that she said is common also in some of the more recent randomized controlled studies, such as IMPAACT PROMISE.
Dr. Yawetz, who was not involved in the study, also noted, “The groups were small, so comparisons within the groups are difficult, and so many people were excluded that it’s hard to know if there were adverse outcomes related to this ... It’s very confounded.”
The World Health Organization estimates that there are roughly 1.3 million pregnant women with HIV, 81% of whom are on antiretroviral therapy. Although the literature continues to evolve, data suggest that in general, Black women are at greater risk for gestational weight gain.
“We have to remember that women who gain excess weight in pregnancy are still going to be with this weight following pregnancy as well,” Dr. Yawetz said. “So, it might impact their pregnancy but also their health after delivery and for subsequent pregnancies, which we don’t have data for yet.”
Dr. Fuller agrees that more data are needed and mentioned that the team plans to study this further, ideally with larger sample sizes.
Yet, despite the lingering questions, there is a silver lining, one that Dr. Yawetz was emphatic about.
“I really welcome people doing studies on this because we really need the data. By far, integrase inhibitors are the first-line regimen all over the world for pregnant women, and if you look at the gestalt or full picture, this is the best regimen to give pregnant women,” she said.
Dr. Fuller and Dr. Yawetz report no relevant financial relationships. The study was independently supported.
A version of this article first appeared on Medscape.com.
The doctor circuit
A long time ago, as a fourth-year medical student, I did a neurology rotation at a large academic center.
One of the attendings was talking to me about reading, and how, once learned, it became innate: a function that, like breathing, couldn’t be turned off.
He was right, as is obvious to anyone. Driving down the road, walking past a newsstand, even opening a fridge covered with magnets from various other medical businesses ... it’s impossible NOT to process the letters into words and words into meanings, even if just for a second. Advertisers and headline-writers figured this out long ago. The key is to make those few words something that grabs our attention and interest, so we’ll either want to read more or retain it.
So too is being a doctor. Once that switch is on, you can’t flip it off.
Recently Queen Elizabeth II died. In reading the news stories, without intending to, I found my mind trying to pick out details about her medical condition, formulate a differential ... after all these years of being in medicine it’s second nature to do that.
Of course, it’s none of my business, and I greatly respect personal privacy. But the point is there. At some point, like reading, we can’t turn off the doctor circuit (for lack of a better term). We do it all the time, analyzing gait patterns and arm swings as people go by. Noticing facial asymmetries, tremors, speech patterns. It may be turned down a few notches from when we’re in the office or hospital, but it’s still there.
It becomes second nature, a part of who we are.
It’s not just doctors. Architects casually notice building details that no one else would. Software engineers off-handedly see program features (good and bad) that the rest of us wouldn’t. Teachers and editors pick up on grammatical errors even when they’re not trying to.
None of these (aside from basic observation) are things that brains originally started out to do. But through training and experience we’ve adapted them to do this. We never stop observing, collecting data, and processing it, in ways peculiar to our backgrounds.
Which, if you think about it, is pretty remarkable.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
A long time ago, as a fourth-year medical student, I did a neurology rotation at a large academic center.
One of the attendings was talking to me about reading, and how, once learned, it became innate: a function that, like breathing, couldn’t be turned off.
He was right, as is obvious to anyone. Driving down the road, walking past a newsstand, even opening a fridge covered with magnets from various other medical businesses ... it’s impossible NOT to process the letters into words and words into meanings, even if just for a second. Advertisers and headline-writers figured this out long ago. The key is to make those few words something that grabs our attention and interest, so we’ll either want to read more or retain it.
So too is being a doctor. Once that switch is on, you can’t flip it off.
Recently Queen Elizabeth II died. In reading the news stories, without intending to, I found my mind trying to pick out details about her medical condition, formulate a differential ... after all these years of being in medicine it’s second nature to do that.
Of course, it’s none of my business, and I greatly respect personal privacy. But the point is there. At some point, like reading, we can’t turn off the doctor circuit (for lack of a better term). We do it all the time, analyzing gait patterns and arm swings as people go by. Noticing facial asymmetries, tremors, speech patterns. It may be turned down a few notches from when we’re in the office or hospital, but it’s still there.
It becomes second nature, a part of who we are.
It’s not just doctors. Architects casually notice building details that no one else would. Software engineers off-handedly see program features (good and bad) that the rest of us wouldn’t. Teachers and editors pick up on grammatical errors even when they’re not trying to.
None of these (aside from basic observation) are things that brains originally started out to do. But through training and experience we’ve adapted them to do this. We never stop observing, collecting data, and processing it, in ways peculiar to our backgrounds.
Which, if you think about it, is pretty remarkable.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
A long time ago, as a fourth-year medical student, I did a neurology rotation at a large academic center.
One of the attendings was talking to me about reading, and how, once learned, it became innate: a function that, like breathing, couldn’t be turned off.
He was right, as is obvious to anyone. Driving down the road, walking past a newsstand, even opening a fridge covered with magnets from various other medical businesses ... it’s impossible NOT to process the letters into words and words into meanings, even if just for a second. Advertisers and headline-writers figured this out long ago. The key is to make those few words something that grabs our attention and interest, so we’ll either want to read more or retain it.
So too is being a doctor. Once that switch is on, you can’t flip it off.
Recently Queen Elizabeth II died. In reading the news stories, without intending to, I found my mind trying to pick out details about her medical condition, formulate a differential ... after all these years of being in medicine it’s second nature to do that.
Of course, it’s none of my business, and I greatly respect personal privacy. But the point is there. At some point, like reading, we can’t turn off the doctor circuit (for lack of a better term). We do it all the time, analyzing gait patterns and arm swings as people go by. Noticing facial asymmetries, tremors, speech patterns. It may be turned down a few notches from when we’re in the office or hospital, but it’s still there.
It becomes second nature, a part of who we are.
It’s not just doctors. Architects casually notice building details that no one else would. Software engineers off-handedly see program features (good and bad) that the rest of us wouldn’t. Teachers and editors pick up on grammatical errors even when they’re not trying to.
None of these (aside from basic observation) are things that brains originally started out to do. But through training and experience we’ve adapted them to do this. We never stop observing, collecting data, and processing it, in ways peculiar to our backgrounds.
Which, if you think about it, is pretty remarkable.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.