User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Decreasing Overutilization of Echocardiograms and Abdominal Imaging in the Evaluation of Children with Fungemia
From the University of Miami, Department of Pediatrics and Department of Medicine, Miami, FL.
Abstract
- Objective: Pediatric fungemia is associated with a low risk of fungal endocarditis and renal infections. The majority of current guidelines do not recommend routine abdominal imaging/echocardiograms in the evaluation of fungemia, but such imaging has been routinely ordered for patients on the pediatric gastroenterology service at our institution. Our goals were to assess the financial impact of this deviation from current clinical guidelines and redefine the standard work to reduce overutilization of abdominal ultrasounds and echocardiograms. Specifically, our goal was to reduce imaging by 50% by 18 months.
- Methods: Root cause analysis showed a lack of familiarity with current evidence. Using this data, countermeasures were implemented, including practitioner education of guidelines and creation of a readily accessible clinical pathway and an electronic order set for pediatric fungemia management. Balancing measures were missed episodes of fungal endocarditis and renal infection.
- Results: During the period January 1, 2016 to November 19, 2017, 18 of 21 episodes of fungemia in our pediatric institution occurred in patients admitted to the pediatric gastroenterology service. Abdominal imaging and echocardiograms were done 100% of the time, with no positive findings and an estimated cost of approximately $58,000. Post-intervention from November 20, 2017 to April 3, 2019, 7 of 13 episodes of fungemia occurred on this service. Frequency of abdominal imaging and echocardiograms decreased to 43% and 57%, respectively. No episodes of fungal endocarditis or renal infection were identified.
- Conclusion: Overutilization of abdominal imaging and echocardiograms in pediatric fungemia evaluation can be safely decreased.
Keywords: guidelines; cost; candidemia; endocarditis.
Practitioners may remain under the impression that routine abdominal ultrasounds (US) and echocardiograms (echo) are indicated in fungemia to evaluate for fungal endocarditis and renal infection, although these conditions are rare and limited to a subset of the population.1-10 Risk factors include prematurity, immunosuppression, prior bacterial endocarditis, abnormal cardiac valves, and previous urogenital surgeries.11
The 2016 Infectious Diseases Society of America (IDSA) guidelines do not recommend routine US or echo but rather provide scenarios in which Candida endocarditis should be suspected, and these include: persistently positive blood cultures, persistent fevers despite appropriate therapy, and clinical signs that may suggest endocarditis, such as a new heart murmur, heart failure, or embolic phenomena.11 IDSA recommends abdominal imaging in neonates with persistently positive blood cultures to evaluate the urogenital system, in addition to the liver and spleen. They also recommend abdominal imaging in symptomatic ascending Candida pyelonephritis beyond the neonatal period and in chronic disseminated candidiasis; the latter is uncommon and seen almost exclusively in patients recovering from neutropenia with a hematologic malignancy.11
We also reviewed guidelines on fungemia originating outside the United States. The 2010 Canadian clinical guidelines on invasive candidiasis do not explicitly recommend routine imaging, but rather state that various imaging studies, including US and echo among others, may be helpful.12 The German Speaking Mycological Society and the Paul-Ehrlich-Society for Chemotherapy published a joint recommendation against routine US and echo in uncomplicated candidemia in 2011.13
The European Society for Clinical Microbiology and Infectious Diseases is the only society that recommends routine echo. Their 2012 guidelines on candidiasis recommend transesophageal echo in adults14 and echocardiography in children,15 as well as abdominal imaging in the diagnosis of chronic disseminated candidiasis in adults with hematological malignancies/hematopoietic stem cell transplantation.16
The 2013 Brazilian guidelines explicitly recommend against routine abdominal imaging and echo because of the low frequency of visceral lesions in adults with candidemia and recommend reserving imaging for those with persistently positive blood cultures or with clinical signs/symptoms suggestive of endocarditis/abdominal infection or clinical deterioration.17 The 2014 Japanese guidelines recommend ruling out chronic disseminated candidiasis in these patients with symptoms during the neutrophil recovery phase, but do not mention routinely imaging other patients. They do not address the role of echocardiography.18
Although physicians in the United Sates typically follow IDSA guidelines, abdominal US and echo were ordered routinely for patients with fungemia on the pediatric gastroenterology service at our institution, leading to higher medical costs and waste of medical resources. Our goals were to assess the current standard work for fungemia evaluation on this service, assess the impact of its deviation from current clinical guidelines, and redefine the standard work by (1) presenting current evidence to practitioners taking care of patients on this service, (2) providing a clinical pathway that allowed for variations where appropriate, and (3) providing a plan for pediatric fungemia management. Our SMART (Specific, Measurable, Attainable, Relevant and Timely) goal was to reduce overutilization of abdominal US and echo in pediatric patients with fungemia on the pediatric gastroenterology service by 50%.
Methods
Study, Setting, and Participants
We executed this quality improvement project at a quaternary care pediatric hospital affiliated with a school of medicine. The project scope consisted of inpatient pediatric patients with fungemia on the pediatric gastroenterology service admitted to the wards or pediatric critical care unit at this institution, along with the practitioners caring for these patients. The project was part of an institutional quality improvement initiative program. The quality improvement team included quality improvement experts from the departments of medicine and pediatrics, a pediatric resident and student, and physicians from the divisions of pediatric infectious disease, pediatric critical care, and pediatric gastroenterology. This study qualified for Institutional Review Board (IRB) exemption based on the University’s IRB stipulations.
Current Condition
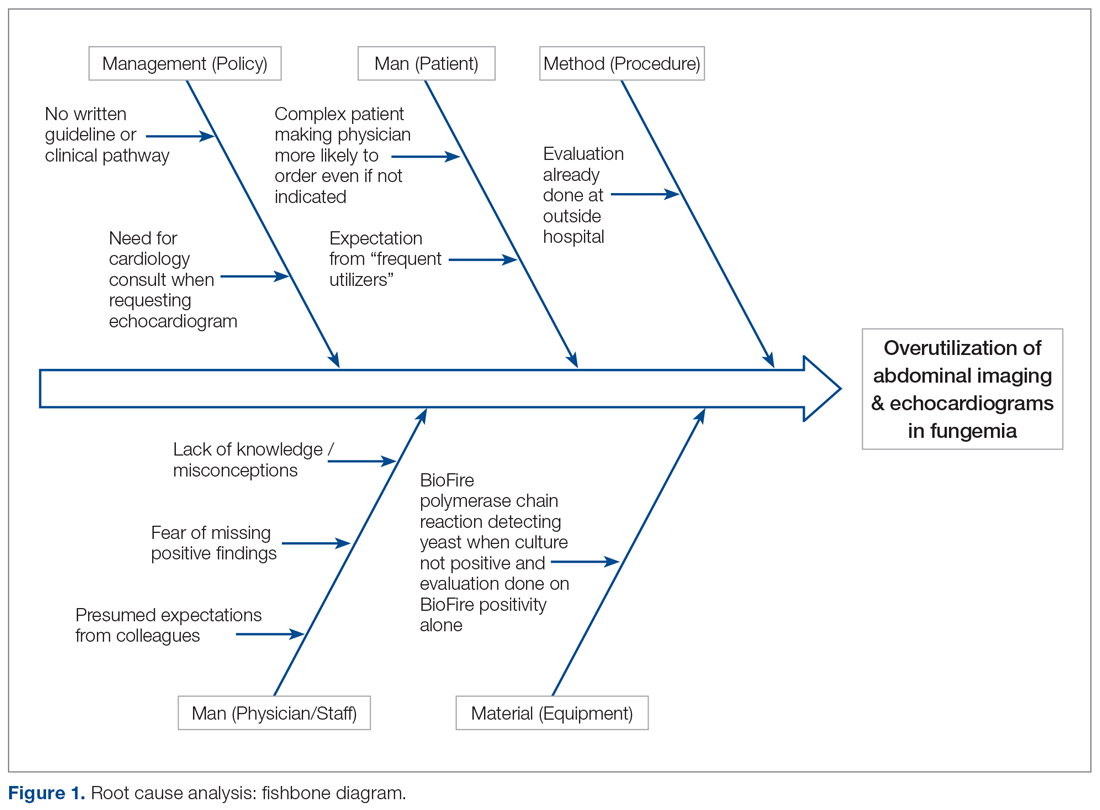
Root cause analysis was performed by creating a process map of the current standard work and a fishbone diagram (Figure 1). We incorporated feedback from voice of the customer in the root cause analysis. In this analysis, the voice of the customer came from the bedside floor nurses, ultrasound clerk and sonographer, echo technician, cardiology fellow, and microbiology medical technician. We got their feedback on our process map, its accuracy and ways to expand, their thoughts on the problem and why we have this problem, and any solutions they could offer to help improve the problem. Some of the key points obtained were: echos were not routinely done on the floors and were not considered urgent as they often did not change management; the sonographer and those from the cardiology department felt imaging was often overutilized because of misconceptions and lack of available hospital guidelines. Suggested solutions included provider education with reference to Duke’s criteria and establishing a clinical pathway approved by all concerned departments.
Prior to education, we surveyed current practices of practitioners on teams caring for these patients, which included physicians of all levels (attendings, fellows, residents) as well as nurse practitioners and medical students from the department of pediatrics and divisions of pediatric gastroenterology, pediatric infectious disease, and pediatric critical care medicine.
Countermeasures
Practitioner Education. In October 2017 practitioners were given a 20-minute presentation on the latest international guidelines on fungemia. Fifty-nine practitioners completed pre- and post-test surveys. Eight respondents were excluded due to incomplete surveys. We compared self-reported frequencies of ordering abdominal imaging and echo before the presentation with intention to order post education. Intention to change clinical practice after the presentation was also surveyed.
Clinical Pathway. Education alone may not result in sustainability, and thus we provided a readily accessible clinical pathway and an electronic order set for pediatric fungemia management. Inter-department buy-in was also necessary for success. It was important to get the input from the various teams (infectious disease, cardiology, gastroenterology, and critical care), which was done by incorporating members from those divisions in the project or getting their feedback through voice of the customer analysis.
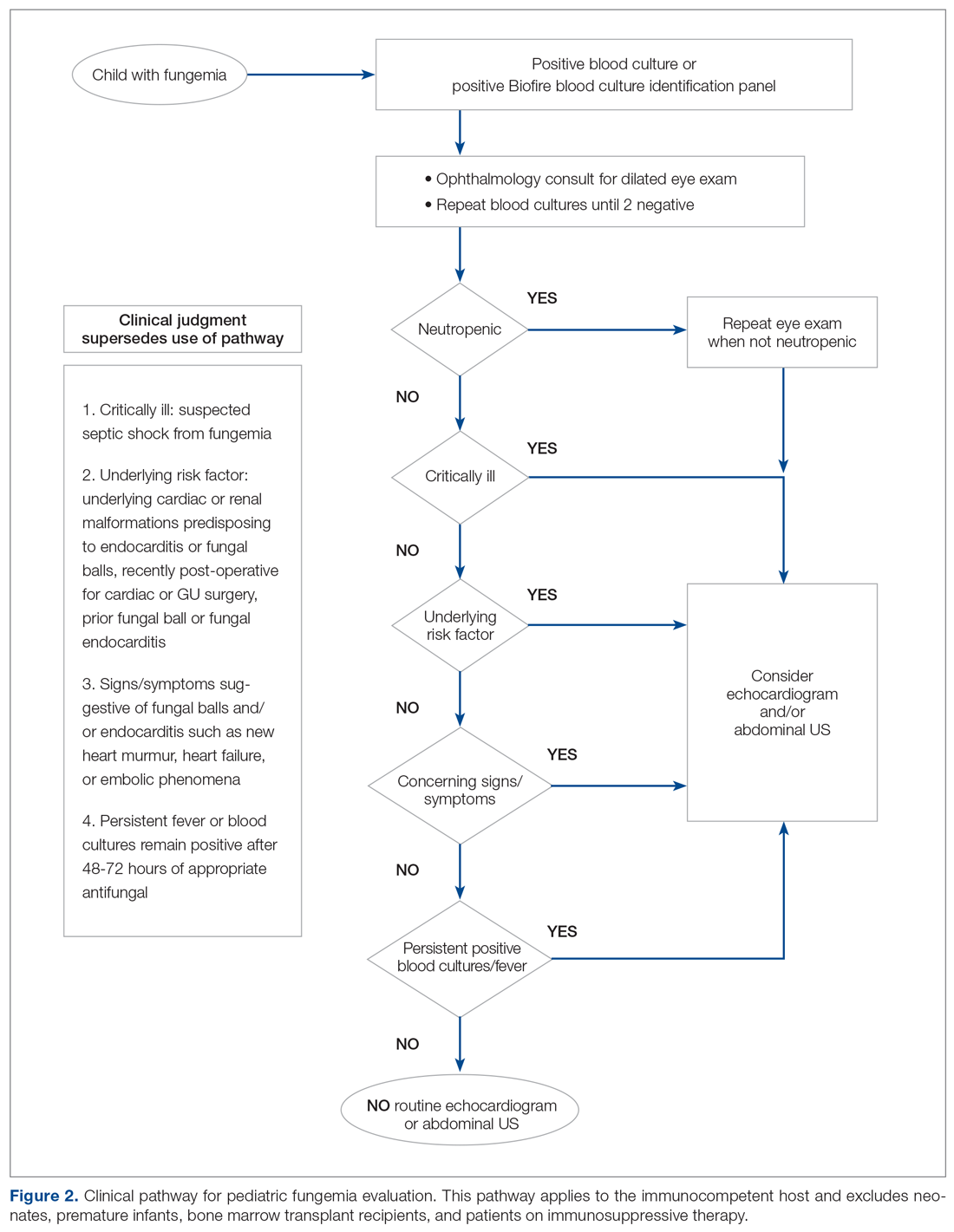
We redefined standard work based on current evidence and created a clinical pathway during March 2018 that included variations when appropriate (Figure 2). We presented the clinical pathway to practitioners and distributed it via email. We also made it available to pediatric residents and fellows on their mobile institutional work resource application.
Electronic Order Set. We created an electronic order set for pediatric fungemia management and made it available in the electronic health record May 2018.
Measurement
Cases of fungemia were identified through the electronic health record pre-intervention (January 1, 2016 through November 19, 2017) and post-intervention (November 20, 2019 through April 3, 2019). An episode of fungemia was defined as an encounter with 1 or more positive blood culture(s) for Candida species or Cryptococcus species. We manually identified patients belonging to the pediatric gastroenterology service and reviewed these charts to determine the presenting complaint, organism isolated, transplant status, central lines status, risk factors, if abdominal imaging or echocardiography were done for the episode of fungemia, and their corresponding results. We calculated overall and per patient medical charges by using the average charges at our institution of US and echocardiography with a cardiology consult. These average charges were provided by patient financial services and the pediatric cardiology department, respectively. To address non-technical expenditures, we calculated the average time taken for transport to and from radiology and the echo suite for each identified patient. We identified missed fungal endocarditis and fungal balls as balancing measures.
Results
Survey
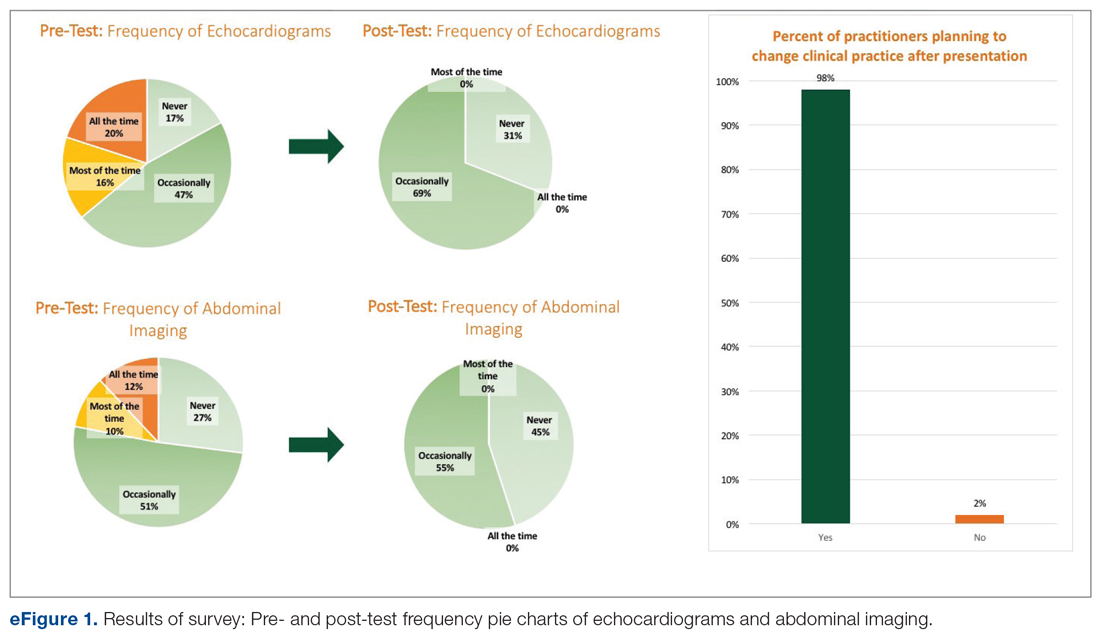
Among the 51 practitioners surveyed, 36% were performing routine echo and 22% self-reported performing routine abdominal imaging. After education, no respondents planned to routinely do echo or abdominal imaging. All but 1 respondent planned to change their practice for evaluation of fungemia patients based on the presentation (eFigure 1).
Baseline Data
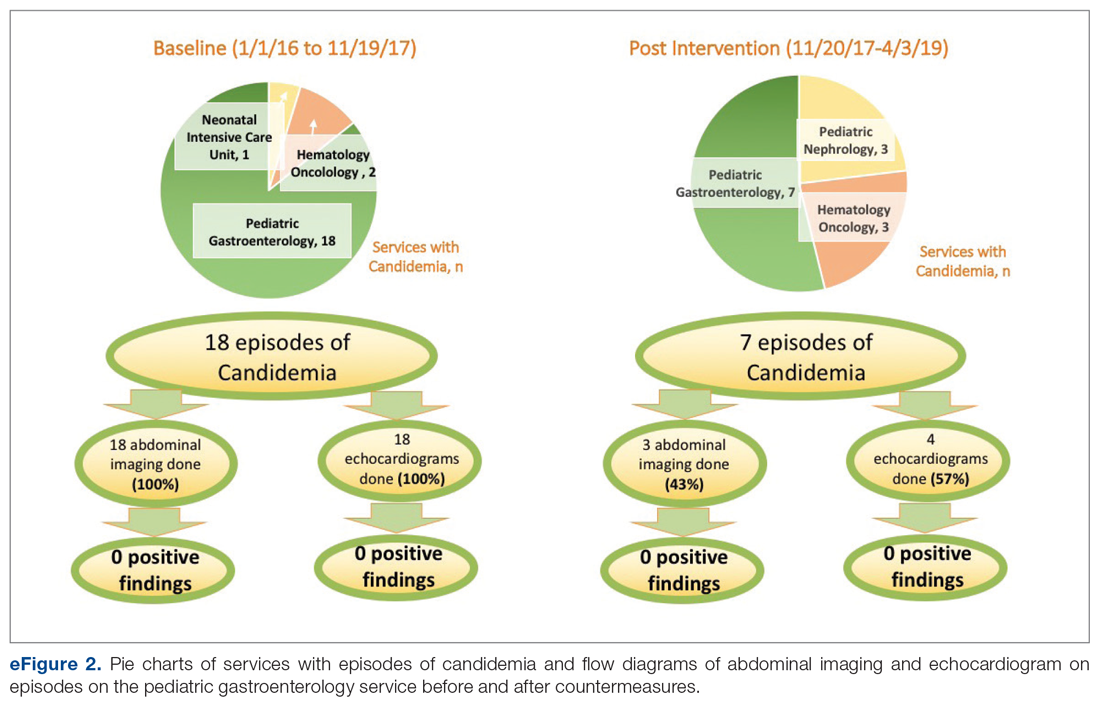
Over the 23-month period from January 1, 2016 to November 19, 2017, there were 21 episodes of fungemia, 18 of which occurred in patients on the pediatric gastroenterology service (2 of the 18 were transplant recipients). For the 18 episodes on this service, abdominal imaging and echo were done 100% of the time, with 0 positive findings (eFigure 2).
Of those 18 episodes, the average age was 4.6 years, with two-thirds of the population being male. There were 3 patients with multiple episodes that accounted for 8 of the episodes (3, 3, and 2 episodes each). Fever was the most common presenting complaint. The most common organism was Candida parapsilosis (6 of the 18 episodes). All episodes but one involved a central line, and all central lines were removed when present except for one case. Of the risk factors, 3 episodes occurred in neutropenic patients, and for 1 episode the patient had a questionable history of fungal endocarditis (and was on fungal prophylaxis). There were no patients with recent cardiac/urogenital surgery or prior fungal balls. No episodes had clinical symptoms suggestive of fungal endocarditis or fungal balls.
Post-Intervention Data
Over the subsequent 17-month period (November 11, 2017 to April 3, 2019), there were 13 episodes of candidemia. There were no episodes of Cryptococcus fungemia. Seven episodes occurred in patients on the pediatric gastroenterology service (2 of the 7 occurred in transplant recipients). Abdominal imaging was done in 3 of these episodes (43%), and in 2 of these 3 episodes, imaging was done at an outside institution prior to arrival, with no positive results (eFigure 2).
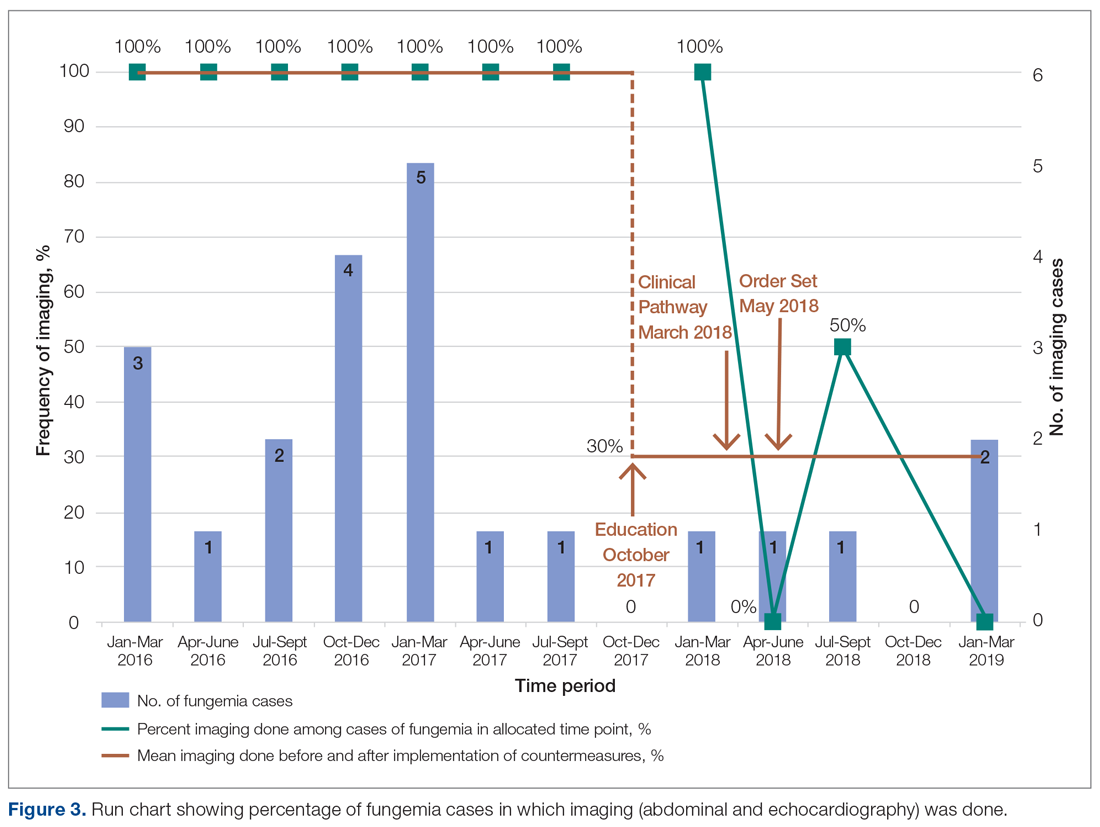
Echocardiography was done 57% of the time (n = 4), with echo being done at an outside institution prior to arrival half of the time (n = 2), with no endocarditis identified. The cases of abdominal imaging and echo done at outside institutions prior to arrival were not impacted by the countermeasures. Excluding those 2 patients who had both abdominal imaging and echocardiography done prior to arrival, the overall rate of imaging (both abdominal imaging and echo) done after countermeasures were instituted was 30% (Figure 3).
Of those 7 episodes, the average age was 6.8 years (57% female). There were no patients with multiple episodes. The most common presenting complaint was fever. The most common organism was Candida albicans (3 of the 7 episodes). All episodes involved a central line, which was removed in all cases except for one. Of the risk factors, 2 episodes were in neutropenic patients, and 1 episode had a history of bacterial endocarditis (not related to fungemia). No episodes occurred in patients with prior fungal renal infection, urogenital malformations, or recent cardiac/urogenital surgery. No episodes had clinical symptoms suggestive of fungal endocarditis or renal infection. No episodes of fungal endocarditis or renal infection were identified.
On average, a patient at our institution undergoing abdominal US and echo with a cardiology consult results in medical waste of approximately $3200 per patient. This cost does not take into account other miscellaneous charges possibly incurred, such as the radiologist interpreting the findings and transportation. Baseline data calculations show that patients waste on average 55 minutes in physical transport, and this does not take into account wait times.
Discussion
Candidemia contributes to 10% of central-line associated blood stream infections (CLABSI).19 Increased usage of indwelling central catheters for administration of parenteral nutrition will inevitably result in practitioners encountering cases of candidemia when caring for this population. As seen from our results, the majority of episodes of candidemia at our institution occurred on the pediatric gastroenterology service, and thus redefining standard work on this service will be impactful.
Candida parapsilosis and Candida albicans were the most common causative agents before and after intervention, respectively, but overall the most common organism was Candida albicans, which is in keeping with that of CLABSI in the literature.19 Growth of Candida parapsilosis has been particularly linked to CLABSI.19 The third most common organism in our study was Candida glabrata, which is the second most common cause of candidemia in CLABSI.19
The cases of positive abdominal imaging in fungemia in the literature are limited to the neonatal population1-4 and chronic disseminated candidiasis in patients with hematologic malignancies/neutropenia/immunosuppression.5,6 In fungal endocarditis, the reported cases were generally in neonates,1,3,7 critically ill patients,8 patients with hematologic malignancies/neutropenia/immunosuppression,6,9 or those with a cardiac history.9,10 This population differs from the patient population on the pediatric gastroenterology service. Patients on this service may not need US or echo. Performing abdominal US and echo in fungemia patients in whom such imaging is not indicated may result in medical waste of approximately $3200 per patient. There is also a waste of medical resources and time.
We found almost all practitioners are willing to change clinical practice once provided with current guidelines. Face-to-face oral presentations allowed for questions and interaction, making this form of information dissemination better than e-mails or handouts.
Though the numbers were small over the short study period, we were able to decrease overutilization of abdominal imaging and echo after implementing countermeasures. Frequency decreased from 100% to 43% and 57% for abdominal imaging and echo, respectively. Imaging that was done after the countermeasures were implemented was mainly attributed to imaging patients underwent prior to presenting to our institution. This reinforces the need for education at other institutions as well. Of the balancing measures assessed, there were no missed cases of fungal balls or fungal endocarditis. Additionally,
The findings from this quality improvement project underscore current recommendations that, despite common misconceptions, routine abdominal US and echo are not indicated in all cases of fungemia. Case-by-case assessment based on the clinical scenario remains key to management of fungemia to avoid unnecessary medical interventions.
Corresponding author: Donna Cheung, MBBS, 200 Hawkins Drive, BT 1120-G, Iowa City, IA 52242; [email protected].
Financial support: None.
1. Benjamin DK Jr, Poole C, Steinbach WJ, et al. Neonatal candidemia and end-organ damage: a critical appraisal of the literature using meta-analytic techniques. Pediatrics. 2003;112:634-640.
2. Wynn JL, Tan S, Gantz MG, et al. Outcomes following candiduria in extremely low birth weight infants. Clin Infect Dis. 2012;54:331-339.
3. Noyola DE, Fernandez M, Moylett EH, et al. Ophthalmologic, visceral, and cardiac involvement in neonates with candidemia. Clin Infect Dis. 2001;32:1018-1023.
4. Phillips JR, Karlowicz MG Prevalence of Candida species in hospital-acquired urinary tract infections in a neonatal intensive care unit. Pediatr Infect Dis J. 1997;16:190-194.
5. Pagano L, Mele L, Fianchi L, et al. Chronic disseminated candidiasis in patients with hematologic malignancies. Clinical features and outcome of 29 episodes. Haematologica. 2002;87:535-541.
6. Zaoutis TE, Greves HM, Lautenbach E, et al. Risk factors for disseminated candidiasis in children with candidemia. Pediatr Infect Dis J. 2004;23:635-641.
7. Levy I, Shalit I, Birk E, et al. Candida endocarditis in neonates: report of five cases and review of the literature. Mycoses. 2006;49:43-48.
8. Aspesberro F, Beghetti M, Oberhansli I, et al. Fungal endocarditis in critically ill children. Eur J Pediatr. 1999;158:275-280.
9. Fernandez-Cruz A, Cruz Menarguez M, Munoz P, et al. The search for endocarditis in patients with candidemia: a systematic recommendation for echocardiography? A prospective cohort. Eur J Clin Microbiol Infect Dis. 2015;34:1543-1549.
10. Hernandez-Torres A, Garcia-Vazquez E, Laso-Ortiz A, et al. [Candida sp endocarditis. Experience in a third-level hospital and review of the literature]. Rev Esp Quimioter. 2013;26:51-55.
11. Pappas PG, Kauffman CA, Andes DR, et al. Clinical practice guideline for the management of candidiasis: 2016 Update by the Infectious Diseases Society of America. Clin Infect Dis. 2016;62:e1-50.
12. Bow EJ, Evans G, Fuller J, et al. Canadian clinical practice guidelines for invasive candidiasis in adults. Can J Infect Dis Med Microbiol. 2010;21:e122-50.
13. Ruhnke M, Rickerts V, Cornely OA, et al. Diagnosis and therapy of Candida infections: joint recommendations of the German Speaking Mycological Society and the Paul-Ehrlich-Society for Chemotherapy. Mycoses. 2011;54:279-310.
14. Cornely OA, Bassetti M, Calandra T, et al. ESCMID* guideline for the diagnosis and management of Candida diseases 2012: non-neutropenic adult patients. Clin Microbiol Infect. 2012;18 Suppl 7:19-37.
15. Hope WW, Castagnola E, Groll AH, et al. ESCMID* guideline for the diagnosis and management of Candida diseases 2012: prevention and management of invasive infections in neonates and children caused by Candida spp. Clin Microbiol Infect. 2012;18 Suppl 7:38-52.
16. Ullmann AJ, Akova M, Herbrecht R, et al. ESCMID* guideline for the diagnosis and management of Candida diseases 2012: adults with haematological malignancies and after haematopoietic stem cell transplantation (HCT). Clin Microbiol Infect. 2012;18 Suppl 7:53-67.
17. Colombo AL, Guimaraes T, Camargo LF, et al. Brazilian guidelines for the management of candidiasis - a joint meeting report of three medical societies: Sociedade Brasileira de Infectologia, Sociedade Paulista de Infectologia and Sociedade Brasileira de Medicina Tropical. Braz J Infect Dis. 2013;17:283-312.
18. Kohno S, Tamura K, Niki Y, et al. Executive Summary of Japanese Domestic guidelines for management of deep-seated mycosis 2014. Med Mycol J. 2016;57:E117-E163.
19. Kojic EM, Darouiche RO. Candida infections of medical devices. Clin Microbiology Rev. 2004;17:255-267.
From the University of Miami, Department of Pediatrics and Department of Medicine, Miami, FL.
Abstract
- Objective: Pediatric fungemia is associated with a low risk of fungal endocarditis and renal infections. The majority of current guidelines do not recommend routine abdominal imaging/echocardiograms in the evaluation of fungemia, but such imaging has been routinely ordered for patients on the pediatric gastroenterology service at our institution. Our goals were to assess the financial impact of this deviation from current clinical guidelines and redefine the standard work to reduce overutilization of abdominal ultrasounds and echocardiograms. Specifically, our goal was to reduce imaging by 50% by 18 months.
- Methods: Root cause analysis showed a lack of familiarity with current evidence. Using this data, countermeasures were implemented, including practitioner education of guidelines and creation of a readily accessible clinical pathway and an electronic order set for pediatric fungemia management. Balancing measures were missed episodes of fungal endocarditis and renal infection.
- Results: During the period January 1, 2016 to November 19, 2017, 18 of 21 episodes of fungemia in our pediatric institution occurred in patients admitted to the pediatric gastroenterology service. Abdominal imaging and echocardiograms were done 100% of the time, with no positive findings and an estimated cost of approximately $58,000. Post-intervention from November 20, 2017 to April 3, 2019, 7 of 13 episodes of fungemia occurred on this service. Frequency of abdominal imaging and echocardiograms decreased to 43% and 57%, respectively. No episodes of fungal endocarditis or renal infection were identified.
- Conclusion: Overutilization of abdominal imaging and echocardiograms in pediatric fungemia evaluation can be safely decreased.
Keywords: guidelines; cost; candidemia; endocarditis.
Practitioners may remain under the impression that routine abdominal ultrasounds (US) and echocardiograms (echo) are indicated in fungemia to evaluate for fungal endocarditis and renal infection, although these conditions are rare and limited to a subset of the population.1-10 Risk factors include prematurity, immunosuppression, prior bacterial endocarditis, abnormal cardiac valves, and previous urogenital surgeries.11
The 2016 Infectious Diseases Society of America (IDSA) guidelines do not recommend routine US or echo but rather provide scenarios in which Candida endocarditis should be suspected, and these include: persistently positive blood cultures, persistent fevers despite appropriate therapy, and clinical signs that may suggest endocarditis, such as a new heart murmur, heart failure, or embolic phenomena.11 IDSA recommends abdominal imaging in neonates with persistently positive blood cultures to evaluate the urogenital system, in addition to the liver and spleen. They also recommend abdominal imaging in symptomatic ascending Candida pyelonephritis beyond the neonatal period and in chronic disseminated candidiasis; the latter is uncommon and seen almost exclusively in patients recovering from neutropenia with a hematologic malignancy.11
We also reviewed guidelines on fungemia originating outside the United States. The 2010 Canadian clinical guidelines on invasive candidiasis do not explicitly recommend routine imaging, but rather state that various imaging studies, including US and echo among others, may be helpful.12 The German Speaking Mycological Society and the Paul-Ehrlich-Society for Chemotherapy published a joint recommendation against routine US and echo in uncomplicated candidemia in 2011.13
The European Society for Clinical Microbiology and Infectious Diseases is the only society that recommends routine echo. Their 2012 guidelines on candidiasis recommend transesophageal echo in adults14 and echocardiography in children,15 as well as abdominal imaging in the diagnosis of chronic disseminated candidiasis in adults with hematological malignancies/hematopoietic stem cell transplantation.16
The 2013 Brazilian guidelines explicitly recommend against routine abdominal imaging and echo because of the low frequency of visceral lesions in adults with candidemia and recommend reserving imaging for those with persistently positive blood cultures or with clinical signs/symptoms suggestive of endocarditis/abdominal infection or clinical deterioration.17 The 2014 Japanese guidelines recommend ruling out chronic disseminated candidiasis in these patients with symptoms during the neutrophil recovery phase, but do not mention routinely imaging other patients. They do not address the role of echocardiography.18
Although physicians in the United Sates typically follow IDSA guidelines, abdominal US and echo were ordered routinely for patients with fungemia on the pediatric gastroenterology service at our institution, leading to higher medical costs and waste of medical resources. Our goals were to assess the current standard work for fungemia evaluation on this service, assess the impact of its deviation from current clinical guidelines, and redefine the standard work by (1) presenting current evidence to practitioners taking care of patients on this service, (2) providing a clinical pathway that allowed for variations where appropriate, and (3) providing a plan for pediatric fungemia management. Our SMART (Specific, Measurable, Attainable, Relevant and Timely) goal was to reduce overutilization of abdominal US and echo in pediatric patients with fungemia on the pediatric gastroenterology service by 50%.
Methods
Study, Setting, and Participants
We executed this quality improvement project at a quaternary care pediatric hospital affiliated with a school of medicine. The project scope consisted of inpatient pediatric patients with fungemia on the pediatric gastroenterology service admitted to the wards or pediatric critical care unit at this institution, along with the practitioners caring for these patients. The project was part of an institutional quality improvement initiative program. The quality improvement team included quality improvement experts from the departments of medicine and pediatrics, a pediatric resident and student, and physicians from the divisions of pediatric infectious disease, pediatric critical care, and pediatric gastroenterology. This study qualified for Institutional Review Board (IRB) exemption based on the University’s IRB stipulations.
Current Condition
Root cause analysis was performed by creating a process map of the current standard work and a fishbone diagram (Figure 1). We incorporated feedback from voice of the customer in the root cause analysis. In this analysis, the voice of the customer came from the bedside floor nurses, ultrasound clerk and sonographer, echo technician, cardiology fellow, and microbiology medical technician. We got their feedback on our process map, its accuracy and ways to expand, their thoughts on the problem and why we have this problem, and any solutions they could offer to help improve the problem. Some of the key points obtained were: echos were not routinely done on the floors and were not considered urgent as they often did not change management; the sonographer and those from the cardiology department felt imaging was often overutilized because of misconceptions and lack of available hospital guidelines. Suggested solutions included provider education with reference to Duke’s criteria and establishing a clinical pathway approved by all concerned departments.

Prior to education, we surveyed current practices of practitioners on teams caring for these patients, which included physicians of all levels (attendings, fellows, residents) as well as nurse practitioners and medical students from the department of pediatrics and divisions of pediatric gastroenterology, pediatric infectious disease, and pediatric critical care medicine.
Countermeasures
Practitioner Education. In October 2017 practitioners were given a 20-minute presentation on the latest international guidelines on fungemia. Fifty-nine practitioners completed pre- and post-test surveys. Eight respondents were excluded due to incomplete surveys. We compared self-reported frequencies of ordering abdominal imaging and echo before the presentation with intention to order post education. Intention to change clinical practice after the presentation was also surveyed.
Clinical Pathway. Education alone may not result in sustainability, and thus we provided a readily accessible clinical pathway and an electronic order set for pediatric fungemia management. Inter-department buy-in was also necessary for success. It was important to get the input from the various teams (infectious disease, cardiology, gastroenterology, and critical care), which was done by incorporating members from those divisions in the project or getting their feedback through voice of the customer analysis.
We redefined standard work based on current evidence and created a clinical pathway during March 2018 that included variations when appropriate (Figure 2). We presented the clinical pathway to practitioners and distributed it via email. We also made it available to pediatric residents and fellows on their mobile institutional work resource application.

Electronic Order Set. We created an electronic order set for pediatric fungemia management and made it available in the electronic health record May 2018.
Measurement
Cases of fungemia were identified through the electronic health record pre-intervention (January 1, 2016 through November 19, 2017) and post-intervention (November 20, 2019 through April 3, 2019). An episode of fungemia was defined as an encounter with 1 or more positive blood culture(s) for Candida species or Cryptococcus species. We manually identified patients belonging to the pediatric gastroenterology service and reviewed these charts to determine the presenting complaint, organism isolated, transplant status, central lines status, risk factors, if abdominal imaging or echocardiography were done for the episode of fungemia, and their corresponding results. We calculated overall and per patient medical charges by using the average charges at our institution of US and echocardiography with a cardiology consult. These average charges were provided by patient financial services and the pediatric cardiology department, respectively. To address non-technical expenditures, we calculated the average time taken for transport to and from radiology and the echo suite for each identified patient. We identified missed fungal endocarditis and fungal balls as balancing measures.
Results
Survey
Among the 51 practitioners surveyed, 36% were performing routine echo and 22% self-reported performing routine abdominal imaging. After education, no respondents planned to routinely do echo or abdominal imaging. All but 1 respondent planned to change their practice for evaluation of fungemia patients based on the presentation (eFigure 1).

Baseline Data
Over the 23-month period from January 1, 2016 to November 19, 2017, there were 21 episodes of fungemia, 18 of which occurred in patients on the pediatric gastroenterology service (2 of the 18 were transplant recipients). For the 18 episodes on this service, abdominal imaging and echo were done 100% of the time, with 0 positive findings (eFigure 2).

Of those 18 episodes, the average age was 4.6 years, with two-thirds of the population being male. There were 3 patients with multiple episodes that accounted for 8 of the episodes (3, 3, and 2 episodes each). Fever was the most common presenting complaint. The most common organism was Candida parapsilosis (6 of the 18 episodes). All episodes but one involved a central line, and all central lines were removed when present except for one case. Of the risk factors, 3 episodes occurred in neutropenic patients, and for 1 episode the patient had a questionable history of fungal endocarditis (and was on fungal prophylaxis). There were no patients with recent cardiac/urogenital surgery or prior fungal balls. No episodes had clinical symptoms suggestive of fungal endocarditis or fungal balls.
Post-Intervention Data
Over the subsequent 17-month period (November 11, 2017 to April 3, 2019), there were 13 episodes of candidemia. There were no episodes of Cryptococcus fungemia. Seven episodes occurred in patients on the pediatric gastroenterology service (2 of the 7 occurred in transplant recipients). Abdominal imaging was done in 3 of these episodes (43%), and in 2 of these 3 episodes, imaging was done at an outside institution prior to arrival, with no positive results (eFigure 2).
Echocardiography was done 57% of the time (n = 4), with echo being done at an outside institution prior to arrival half of the time (n = 2), with no endocarditis identified. The cases of abdominal imaging and echo done at outside institutions prior to arrival were not impacted by the countermeasures. Excluding those 2 patients who had both abdominal imaging and echocardiography done prior to arrival, the overall rate of imaging (both abdominal imaging and echo) done after countermeasures were instituted was 30% (Figure 3).

Of those 7 episodes, the average age was 6.8 years (57% female). There were no patients with multiple episodes. The most common presenting complaint was fever. The most common organism was Candida albicans (3 of the 7 episodes). All episodes involved a central line, which was removed in all cases except for one. Of the risk factors, 2 episodes were in neutropenic patients, and 1 episode had a history of bacterial endocarditis (not related to fungemia). No episodes occurred in patients with prior fungal renal infection, urogenital malformations, or recent cardiac/urogenital surgery. No episodes had clinical symptoms suggestive of fungal endocarditis or renal infection. No episodes of fungal endocarditis or renal infection were identified.
On average, a patient at our institution undergoing abdominal US and echo with a cardiology consult results in medical waste of approximately $3200 per patient. This cost does not take into account other miscellaneous charges possibly incurred, such as the radiologist interpreting the findings and transportation. Baseline data calculations show that patients waste on average 55 minutes in physical transport, and this does not take into account wait times.
Discussion
Candidemia contributes to 10% of central-line associated blood stream infections (CLABSI).19 Increased usage of indwelling central catheters for administration of parenteral nutrition will inevitably result in practitioners encountering cases of candidemia when caring for this population. As seen from our results, the majority of episodes of candidemia at our institution occurred on the pediatric gastroenterology service, and thus redefining standard work on this service will be impactful.
Candida parapsilosis and Candida albicans were the most common causative agents before and after intervention, respectively, but overall the most common organism was Candida albicans, which is in keeping with that of CLABSI in the literature.19 Growth of Candida parapsilosis has been particularly linked to CLABSI.19 The third most common organism in our study was Candida glabrata, which is the second most common cause of candidemia in CLABSI.19
The cases of positive abdominal imaging in fungemia in the literature are limited to the neonatal population1-4 and chronic disseminated candidiasis in patients with hematologic malignancies/neutropenia/immunosuppression.5,6 In fungal endocarditis, the reported cases were generally in neonates,1,3,7 critically ill patients,8 patients with hematologic malignancies/neutropenia/immunosuppression,6,9 or those with a cardiac history.9,10 This population differs from the patient population on the pediatric gastroenterology service. Patients on this service may not need US or echo. Performing abdominal US and echo in fungemia patients in whom such imaging is not indicated may result in medical waste of approximately $3200 per patient. There is also a waste of medical resources and time.
We found almost all practitioners are willing to change clinical practice once provided with current guidelines. Face-to-face oral presentations allowed for questions and interaction, making this form of information dissemination better than e-mails or handouts.
Though the numbers were small over the short study period, we were able to decrease overutilization of abdominal imaging and echo after implementing countermeasures. Frequency decreased from 100% to 43% and 57% for abdominal imaging and echo, respectively. Imaging that was done after the countermeasures were implemented was mainly attributed to imaging patients underwent prior to presenting to our institution. This reinforces the need for education at other institutions as well. Of the balancing measures assessed, there were no missed cases of fungal balls or fungal endocarditis. Additionally,
The findings from this quality improvement project underscore current recommendations that, despite common misconceptions, routine abdominal US and echo are not indicated in all cases of fungemia. Case-by-case assessment based on the clinical scenario remains key to management of fungemia to avoid unnecessary medical interventions.
Corresponding author: Donna Cheung, MBBS, 200 Hawkins Drive, BT 1120-G, Iowa City, IA 52242; [email protected].
Financial support: None.
From the University of Miami, Department of Pediatrics and Department of Medicine, Miami, FL.
Abstract
- Objective: Pediatric fungemia is associated with a low risk of fungal endocarditis and renal infections. The majority of current guidelines do not recommend routine abdominal imaging/echocardiograms in the evaluation of fungemia, but such imaging has been routinely ordered for patients on the pediatric gastroenterology service at our institution. Our goals were to assess the financial impact of this deviation from current clinical guidelines and redefine the standard work to reduce overutilization of abdominal ultrasounds and echocardiograms. Specifically, our goal was to reduce imaging by 50% by 18 months.
- Methods: Root cause analysis showed a lack of familiarity with current evidence. Using this data, countermeasures were implemented, including practitioner education of guidelines and creation of a readily accessible clinical pathway and an electronic order set for pediatric fungemia management. Balancing measures were missed episodes of fungal endocarditis and renal infection.
- Results: During the period January 1, 2016 to November 19, 2017, 18 of 21 episodes of fungemia in our pediatric institution occurred in patients admitted to the pediatric gastroenterology service. Abdominal imaging and echocardiograms were done 100% of the time, with no positive findings and an estimated cost of approximately $58,000. Post-intervention from November 20, 2017 to April 3, 2019, 7 of 13 episodes of fungemia occurred on this service. Frequency of abdominal imaging and echocardiograms decreased to 43% and 57%, respectively. No episodes of fungal endocarditis or renal infection were identified.
- Conclusion: Overutilization of abdominal imaging and echocardiograms in pediatric fungemia evaluation can be safely decreased.
Keywords: guidelines; cost; candidemia; endocarditis.
Practitioners may remain under the impression that routine abdominal ultrasounds (US) and echocardiograms (echo) are indicated in fungemia to evaluate for fungal endocarditis and renal infection, although these conditions are rare and limited to a subset of the population.1-10 Risk factors include prematurity, immunosuppression, prior bacterial endocarditis, abnormal cardiac valves, and previous urogenital surgeries.11
The 2016 Infectious Diseases Society of America (IDSA) guidelines do not recommend routine US or echo but rather provide scenarios in which Candida endocarditis should be suspected, and these include: persistently positive blood cultures, persistent fevers despite appropriate therapy, and clinical signs that may suggest endocarditis, such as a new heart murmur, heart failure, or embolic phenomena.11 IDSA recommends abdominal imaging in neonates with persistently positive blood cultures to evaluate the urogenital system, in addition to the liver and spleen. They also recommend abdominal imaging in symptomatic ascending Candida pyelonephritis beyond the neonatal period and in chronic disseminated candidiasis; the latter is uncommon and seen almost exclusively in patients recovering from neutropenia with a hematologic malignancy.11
We also reviewed guidelines on fungemia originating outside the United States. The 2010 Canadian clinical guidelines on invasive candidiasis do not explicitly recommend routine imaging, but rather state that various imaging studies, including US and echo among others, may be helpful.12 The German Speaking Mycological Society and the Paul-Ehrlich-Society for Chemotherapy published a joint recommendation against routine US and echo in uncomplicated candidemia in 2011.13
The European Society for Clinical Microbiology and Infectious Diseases is the only society that recommends routine echo. Their 2012 guidelines on candidiasis recommend transesophageal echo in adults14 and echocardiography in children,15 as well as abdominal imaging in the diagnosis of chronic disseminated candidiasis in adults with hematological malignancies/hematopoietic stem cell transplantation.16
The 2013 Brazilian guidelines explicitly recommend against routine abdominal imaging and echo because of the low frequency of visceral lesions in adults with candidemia and recommend reserving imaging for those with persistently positive blood cultures or with clinical signs/symptoms suggestive of endocarditis/abdominal infection or clinical deterioration.17 The 2014 Japanese guidelines recommend ruling out chronic disseminated candidiasis in these patients with symptoms during the neutrophil recovery phase, but do not mention routinely imaging other patients. They do not address the role of echocardiography.18
Although physicians in the United Sates typically follow IDSA guidelines, abdominal US and echo were ordered routinely for patients with fungemia on the pediatric gastroenterology service at our institution, leading to higher medical costs and waste of medical resources. Our goals were to assess the current standard work for fungemia evaluation on this service, assess the impact of its deviation from current clinical guidelines, and redefine the standard work by (1) presenting current evidence to practitioners taking care of patients on this service, (2) providing a clinical pathway that allowed for variations where appropriate, and (3) providing a plan for pediatric fungemia management. Our SMART (Specific, Measurable, Attainable, Relevant and Timely) goal was to reduce overutilization of abdominal US and echo in pediatric patients with fungemia on the pediatric gastroenterology service by 50%.
Methods
Study, Setting, and Participants
We executed this quality improvement project at a quaternary care pediatric hospital affiliated with a school of medicine. The project scope consisted of inpatient pediatric patients with fungemia on the pediatric gastroenterology service admitted to the wards or pediatric critical care unit at this institution, along with the practitioners caring for these patients. The project was part of an institutional quality improvement initiative program. The quality improvement team included quality improvement experts from the departments of medicine and pediatrics, a pediatric resident and student, and physicians from the divisions of pediatric infectious disease, pediatric critical care, and pediatric gastroenterology. This study qualified for Institutional Review Board (IRB) exemption based on the University’s IRB stipulations.
Current Condition
Root cause analysis was performed by creating a process map of the current standard work and a fishbone diagram (Figure 1). We incorporated feedback from voice of the customer in the root cause analysis. In this analysis, the voice of the customer came from the bedside floor nurses, ultrasound clerk and sonographer, echo technician, cardiology fellow, and microbiology medical technician. We got their feedback on our process map, its accuracy and ways to expand, their thoughts on the problem and why we have this problem, and any solutions they could offer to help improve the problem. Some of the key points obtained were: echos were not routinely done on the floors and were not considered urgent as they often did not change management; the sonographer and those from the cardiology department felt imaging was often overutilized because of misconceptions and lack of available hospital guidelines. Suggested solutions included provider education with reference to Duke’s criteria and establishing a clinical pathway approved by all concerned departments.

Prior to education, we surveyed current practices of practitioners on teams caring for these patients, which included physicians of all levels (attendings, fellows, residents) as well as nurse practitioners and medical students from the department of pediatrics and divisions of pediatric gastroenterology, pediatric infectious disease, and pediatric critical care medicine.
Countermeasures
Practitioner Education. In October 2017 practitioners were given a 20-minute presentation on the latest international guidelines on fungemia. Fifty-nine practitioners completed pre- and post-test surveys. Eight respondents were excluded due to incomplete surveys. We compared self-reported frequencies of ordering abdominal imaging and echo before the presentation with intention to order post education. Intention to change clinical practice after the presentation was also surveyed.
Clinical Pathway. Education alone may not result in sustainability, and thus we provided a readily accessible clinical pathway and an electronic order set for pediatric fungemia management. Inter-department buy-in was also necessary for success. It was important to get the input from the various teams (infectious disease, cardiology, gastroenterology, and critical care), which was done by incorporating members from those divisions in the project or getting their feedback through voice of the customer analysis.
We redefined standard work based on current evidence and created a clinical pathway during March 2018 that included variations when appropriate (Figure 2). We presented the clinical pathway to practitioners and distributed it via email. We also made it available to pediatric residents and fellows on their mobile institutional work resource application.

Electronic Order Set. We created an electronic order set for pediatric fungemia management and made it available in the electronic health record May 2018.
Measurement
Cases of fungemia were identified through the electronic health record pre-intervention (January 1, 2016 through November 19, 2017) and post-intervention (November 20, 2019 through April 3, 2019). An episode of fungemia was defined as an encounter with 1 or more positive blood culture(s) for Candida species or Cryptococcus species. We manually identified patients belonging to the pediatric gastroenterology service and reviewed these charts to determine the presenting complaint, organism isolated, transplant status, central lines status, risk factors, if abdominal imaging or echocardiography were done for the episode of fungemia, and their corresponding results. We calculated overall and per patient medical charges by using the average charges at our institution of US and echocardiography with a cardiology consult. These average charges were provided by patient financial services and the pediatric cardiology department, respectively. To address non-technical expenditures, we calculated the average time taken for transport to and from radiology and the echo suite for each identified patient. We identified missed fungal endocarditis and fungal balls as balancing measures.
Results
Survey
Among the 51 practitioners surveyed, 36% were performing routine echo and 22% self-reported performing routine abdominal imaging. After education, no respondents planned to routinely do echo or abdominal imaging. All but 1 respondent planned to change their practice for evaluation of fungemia patients based on the presentation (eFigure 1).

Baseline Data
Over the 23-month period from January 1, 2016 to November 19, 2017, there were 21 episodes of fungemia, 18 of which occurred in patients on the pediatric gastroenterology service (2 of the 18 were transplant recipients). For the 18 episodes on this service, abdominal imaging and echo were done 100% of the time, with 0 positive findings (eFigure 2).

Of those 18 episodes, the average age was 4.6 years, with two-thirds of the population being male. There were 3 patients with multiple episodes that accounted for 8 of the episodes (3, 3, and 2 episodes each). Fever was the most common presenting complaint. The most common organism was Candida parapsilosis (6 of the 18 episodes). All episodes but one involved a central line, and all central lines were removed when present except for one case. Of the risk factors, 3 episodes occurred in neutropenic patients, and for 1 episode the patient had a questionable history of fungal endocarditis (and was on fungal prophylaxis). There were no patients with recent cardiac/urogenital surgery or prior fungal balls. No episodes had clinical symptoms suggestive of fungal endocarditis or fungal balls.
Post-Intervention Data
Over the subsequent 17-month period (November 11, 2017 to April 3, 2019), there were 13 episodes of candidemia. There were no episodes of Cryptococcus fungemia. Seven episodes occurred in patients on the pediatric gastroenterology service (2 of the 7 occurred in transplant recipients). Abdominal imaging was done in 3 of these episodes (43%), and in 2 of these 3 episodes, imaging was done at an outside institution prior to arrival, with no positive results (eFigure 2).
Echocardiography was done 57% of the time (n = 4), with echo being done at an outside institution prior to arrival half of the time (n = 2), with no endocarditis identified. The cases of abdominal imaging and echo done at outside institutions prior to arrival were not impacted by the countermeasures. Excluding those 2 patients who had both abdominal imaging and echocardiography done prior to arrival, the overall rate of imaging (both abdominal imaging and echo) done after countermeasures were instituted was 30% (Figure 3).

Of those 7 episodes, the average age was 6.8 years (57% female). There were no patients with multiple episodes. The most common presenting complaint was fever. The most common organism was Candida albicans (3 of the 7 episodes). All episodes involved a central line, which was removed in all cases except for one. Of the risk factors, 2 episodes were in neutropenic patients, and 1 episode had a history of bacterial endocarditis (not related to fungemia). No episodes occurred in patients with prior fungal renal infection, urogenital malformations, or recent cardiac/urogenital surgery. No episodes had clinical symptoms suggestive of fungal endocarditis or renal infection. No episodes of fungal endocarditis or renal infection were identified.
On average, a patient at our institution undergoing abdominal US and echo with a cardiology consult results in medical waste of approximately $3200 per patient. This cost does not take into account other miscellaneous charges possibly incurred, such as the radiologist interpreting the findings and transportation. Baseline data calculations show that patients waste on average 55 minutes in physical transport, and this does not take into account wait times.
Discussion
Candidemia contributes to 10% of central-line associated blood stream infections (CLABSI).19 Increased usage of indwelling central catheters for administration of parenteral nutrition will inevitably result in practitioners encountering cases of candidemia when caring for this population. As seen from our results, the majority of episodes of candidemia at our institution occurred on the pediatric gastroenterology service, and thus redefining standard work on this service will be impactful.
Candida parapsilosis and Candida albicans were the most common causative agents before and after intervention, respectively, but overall the most common organism was Candida albicans, which is in keeping with that of CLABSI in the literature.19 Growth of Candida parapsilosis has been particularly linked to CLABSI.19 The third most common organism in our study was Candida glabrata, which is the second most common cause of candidemia in CLABSI.19
The cases of positive abdominal imaging in fungemia in the literature are limited to the neonatal population1-4 and chronic disseminated candidiasis in patients with hematologic malignancies/neutropenia/immunosuppression.5,6 In fungal endocarditis, the reported cases were generally in neonates,1,3,7 critically ill patients,8 patients with hematologic malignancies/neutropenia/immunosuppression,6,9 or those with a cardiac history.9,10 This population differs from the patient population on the pediatric gastroenterology service. Patients on this service may not need US or echo. Performing abdominal US and echo in fungemia patients in whom such imaging is not indicated may result in medical waste of approximately $3200 per patient. There is also a waste of medical resources and time.
We found almost all practitioners are willing to change clinical practice once provided with current guidelines. Face-to-face oral presentations allowed for questions and interaction, making this form of information dissemination better than e-mails or handouts.
Though the numbers were small over the short study period, we were able to decrease overutilization of abdominal imaging and echo after implementing countermeasures. Frequency decreased from 100% to 43% and 57% for abdominal imaging and echo, respectively. Imaging that was done after the countermeasures were implemented was mainly attributed to imaging patients underwent prior to presenting to our institution. This reinforces the need for education at other institutions as well. Of the balancing measures assessed, there were no missed cases of fungal balls or fungal endocarditis. Additionally,
The findings from this quality improvement project underscore current recommendations that, despite common misconceptions, routine abdominal US and echo are not indicated in all cases of fungemia. Case-by-case assessment based on the clinical scenario remains key to management of fungemia to avoid unnecessary medical interventions.
Corresponding author: Donna Cheung, MBBS, 200 Hawkins Drive, BT 1120-G, Iowa City, IA 52242; [email protected].
Financial support: None.
1. Benjamin DK Jr, Poole C, Steinbach WJ, et al. Neonatal candidemia and end-organ damage: a critical appraisal of the literature using meta-analytic techniques. Pediatrics. 2003;112:634-640.
2. Wynn JL, Tan S, Gantz MG, et al. Outcomes following candiduria in extremely low birth weight infants. Clin Infect Dis. 2012;54:331-339.
3. Noyola DE, Fernandez M, Moylett EH, et al. Ophthalmologic, visceral, and cardiac involvement in neonates with candidemia. Clin Infect Dis. 2001;32:1018-1023.
4. Phillips JR, Karlowicz MG Prevalence of Candida species in hospital-acquired urinary tract infections in a neonatal intensive care unit. Pediatr Infect Dis J. 1997;16:190-194.
5. Pagano L, Mele L, Fianchi L, et al. Chronic disseminated candidiasis in patients with hematologic malignancies. Clinical features and outcome of 29 episodes. Haematologica. 2002;87:535-541.
6. Zaoutis TE, Greves HM, Lautenbach E, et al. Risk factors for disseminated candidiasis in children with candidemia. Pediatr Infect Dis J. 2004;23:635-641.
7. Levy I, Shalit I, Birk E, et al. Candida endocarditis in neonates: report of five cases and review of the literature. Mycoses. 2006;49:43-48.
8. Aspesberro F, Beghetti M, Oberhansli I, et al. Fungal endocarditis in critically ill children. Eur J Pediatr. 1999;158:275-280.
9. Fernandez-Cruz A, Cruz Menarguez M, Munoz P, et al. The search for endocarditis in patients with candidemia: a systematic recommendation for echocardiography? A prospective cohort. Eur J Clin Microbiol Infect Dis. 2015;34:1543-1549.
10. Hernandez-Torres A, Garcia-Vazquez E, Laso-Ortiz A, et al. [Candida sp endocarditis. Experience in a third-level hospital and review of the literature]. Rev Esp Quimioter. 2013;26:51-55.
11. Pappas PG, Kauffman CA, Andes DR, et al. Clinical practice guideline for the management of candidiasis: 2016 Update by the Infectious Diseases Society of America. Clin Infect Dis. 2016;62:e1-50.
12. Bow EJ, Evans G, Fuller J, et al. Canadian clinical practice guidelines for invasive candidiasis in adults. Can J Infect Dis Med Microbiol. 2010;21:e122-50.
13. Ruhnke M, Rickerts V, Cornely OA, et al. Diagnosis and therapy of Candida infections: joint recommendations of the German Speaking Mycological Society and the Paul-Ehrlich-Society for Chemotherapy. Mycoses. 2011;54:279-310.
14. Cornely OA, Bassetti M, Calandra T, et al. ESCMID* guideline for the diagnosis and management of Candida diseases 2012: non-neutropenic adult patients. Clin Microbiol Infect. 2012;18 Suppl 7:19-37.
15. Hope WW, Castagnola E, Groll AH, et al. ESCMID* guideline for the diagnosis and management of Candida diseases 2012: prevention and management of invasive infections in neonates and children caused by Candida spp. Clin Microbiol Infect. 2012;18 Suppl 7:38-52.
16. Ullmann AJ, Akova M, Herbrecht R, et al. ESCMID* guideline for the diagnosis and management of Candida diseases 2012: adults with haematological malignancies and after haematopoietic stem cell transplantation (HCT). Clin Microbiol Infect. 2012;18 Suppl 7:53-67.
17. Colombo AL, Guimaraes T, Camargo LF, et al. Brazilian guidelines for the management of candidiasis - a joint meeting report of three medical societies: Sociedade Brasileira de Infectologia, Sociedade Paulista de Infectologia and Sociedade Brasileira de Medicina Tropical. Braz J Infect Dis. 2013;17:283-312.
18. Kohno S, Tamura K, Niki Y, et al. Executive Summary of Japanese Domestic guidelines for management of deep-seated mycosis 2014. Med Mycol J. 2016;57:E117-E163.
19. Kojic EM, Darouiche RO. Candida infections of medical devices. Clin Microbiology Rev. 2004;17:255-267.
1. Benjamin DK Jr, Poole C, Steinbach WJ, et al. Neonatal candidemia and end-organ damage: a critical appraisal of the literature using meta-analytic techniques. Pediatrics. 2003;112:634-640.
2. Wynn JL, Tan S, Gantz MG, et al. Outcomes following candiduria in extremely low birth weight infants. Clin Infect Dis. 2012;54:331-339.
3. Noyola DE, Fernandez M, Moylett EH, et al. Ophthalmologic, visceral, and cardiac involvement in neonates with candidemia. Clin Infect Dis. 2001;32:1018-1023.
4. Phillips JR, Karlowicz MG Prevalence of Candida species in hospital-acquired urinary tract infections in a neonatal intensive care unit. Pediatr Infect Dis J. 1997;16:190-194.
5. Pagano L, Mele L, Fianchi L, et al. Chronic disseminated candidiasis in patients with hematologic malignancies. Clinical features and outcome of 29 episodes. Haematologica. 2002;87:535-541.
6. Zaoutis TE, Greves HM, Lautenbach E, et al. Risk factors for disseminated candidiasis in children with candidemia. Pediatr Infect Dis J. 2004;23:635-641.
7. Levy I, Shalit I, Birk E, et al. Candida endocarditis in neonates: report of five cases and review of the literature. Mycoses. 2006;49:43-48.
8. Aspesberro F, Beghetti M, Oberhansli I, et al. Fungal endocarditis in critically ill children. Eur J Pediatr. 1999;158:275-280.
9. Fernandez-Cruz A, Cruz Menarguez M, Munoz P, et al. The search for endocarditis in patients with candidemia: a systematic recommendation for echocardiography? A prospective cohort. Eur J Clin Microbiol Infect Dis. 2015;34:1543-1549.
10. Hernandez-Torres A, Garcia-Vazquez E, Laso-Ortiz A, et al. [Candida sp endocarditis. Experience in a third-level hospital and review of the literature]. Rev Esp Quimioter. 2013;26:51-55.
11. Pappas PG, Kauffman CA, Andes DR, et al. Clinical practice guideline for the management of candidiasis: 2016 Update by the Infectious Diseases Society of America. Clin Infect Dis. 2016;62:e1-50.
12. Bow EJ, Evans G, Fuller J, et al. Canadian clinical practice guidelines for invasive candidiasis in adults. Can J Infect Dis Med Microbiol. 2010;21:e122-50.
13. Ruhnke M, Rickerts V, Cornely OA, et al. Diagnosis and therapy of Candida infections: joint recommendations of the German Speaking Mycological Society and the Paul-Ehrlich-Society for Chemotherapy. Mycoses. 2011;54:279-310.
14. Cornely OA, Bassetti M, Calandra T, et al. ESCMID* guideline for the diagnosis and management of Candida diseases 2012: non-neutropenic adult patients. Clin Microbiol Infect. 2012;18 Suppl 7:19-37.
15. Hope WW, Castagnola E, Groll AH, et al. ESCMID* guideline for the diagnosis and management of Candida diseases 2012: prevention and management of invasive infections in neonates and children caused by Candida spp. Clin Microbiol Infect. 2012;18 Suppl 7:38-52.
16. Ullmann AJ, Akova M, Herbrecht R, et al. ESCMID* guideline for the diagnosis and management of Candida diseases 2012: adults with haematological malignancies and after haematopoietic stem cell transplantation (HCT). Clin Microbiol Infect. 2012;18 Suppl 7:53-67.
17. Colombo AL, Guimaraes T, Camargo LF, et al. Brazilian guidelines for the management of candidiasis - a joint meeting report of three medical societies: Sociedade Brasileira de Infectologia, Sociedade Paulista de Infectologia and Sociedade Brasileira de Medicina Tropical. Braz J Infect Dis. 2013;17:283-312.
18. Kohno S, Tamura K, Niki Y, et al. Executive Summary of Japanese Domestic guidelines for management of deep-seated mycosis 2014. Med Mycol J. 2016;57:E117-E163.
19. Kojic EM, Darouiche RO. Candida infections of medical devices. Clin Microbiology Rev. 2004;17:255-267.
Applying Robust Process Improvement Techniques to the Voluntary Inpatient Psychiatry Admission Process
From the Wake Forest School of Medicine (Ms. Newman), and Wake Forest Baptist Health, Department of Psychiatry and Behavioral Medicine (Dr. Kramer), Winston-Salem, NC.
Abstract
- Background: Adults voluntarily admitted to inpatient behavioral health units can ask to sign a Request to Discharge (RTD) form if they would like to be discharged before the treatment team agrees that discharge is appropriate. This gives the team 72 hours to determine whether the patient is safe to discharge or to involuntarily commit the patient to the unit. At 1 medical center, patients who were offered voluntary admission often lacked complete understanding of the “72-hour rule” and the early discharge procedure.
- Methods: Robust Process Improvement® techniques were implemented to improve the admission process. Flow charts, standardized scripts, and pocket cards were distributed to relevant staff. The Request for Voluntary Admission form was revised to emphasize the “72-hour rule” and the process for requesting a RTD form.
- Results: The unit’s average overall Press Ganey score improved from 77.1 to 81.6 (P = 0.003), while the average discharge score improved from 83.0 to 87.5 (P = 0.023) following implementation of the new process.
- Conclusion: Incorporating strategies such as an opportunity to “teach back” important information about the voluntary admission process (ie, what the 72-hour rule is, what the request to discharge form is, and the possibility of involuntary commitment) allows clinicians to assess capacity while simultaneously giving patients realistic expectations of the admission. These changes can lead to improvement in patient satisfaction.
Keywords: behavioral health; communication; patient satisfaction.
Communication is paramount within medical teams to improve outcomes and strengthen rapport with patients, particularly with psychiatric patients in acute crisis. Studies
While some forms of communication are required to protect the safety of patients and others around them, other forms are required to build strong relationships with patients. However, these 2 goals do not have to be mutually exclusive in the psychiatric hospital environment. Hospitals aim to improve patient satisfaction while simultaneously providing effective communication about treatment. Studies have indicated that communication during graduate medical training may decline due to “emotional and physical brutality” associated with residency training programs.4 To ameliorate this and emphasize communication education, accredited psychiatry residency programs require residents to use structured communication tools to achieve a level 2 in the Accreditation Council for Graduate Medical Education milestone project for the category of patient safety and health care team.5 These standardized processes allow all patients to receive the same important information related to their care while minimizing human error. Such communication skills aim to improve health care outcomes and satisfaction for patients while also training better physicians.
For legal and ethical reasons, the adult inpatient behavioral health units at major hospitals are highly regulated. In most states, a patient who is admitted to an adult inpatient behavioral health unit on a voluntary basis can ask to sign a request to discharge (RTD) form if he or she would like to be discharged from the hospital before the treatment team sees fit.6 In most jurisdictions, this action gives the treatment team 72 hours to determine whether the patient is safe to discharge. Within that time frame, the physician must either discharge the patient, or, if it is not safe to do so, involuntarily commit him or her to the unit. In most jurisdictions, this process is commonly referred to as the “72-hour rule.”
In North Carolina, state legislation Chapter 122C, Article 5, Part 2(b) specifies: “In 24-hour facilities the application shall acknowledge that the applicant may be held by the facility for a period of 72 hours after any written request for release that the applicant may make, and shall acknowledge that the 24-hour facility may have the legal right to petition for involuntary commitment of the applicant during that period. At the time of application, the facility shall tell the applicant about procedures for discharge.”7 This requirement can be somewhat confusing for both medical team members and patients alike.
As formerly practiced on the behavioral health unit described in this report, patients offered voluntary admission status to the inpatient behavioral unit often lacked complete understanding of the 72-hour rule and the process for requesting early discharge from the facility. We hypothesized that this led to the observed patient frustration and hostility, lack of trust in the treatment team, poor attendance and participation in group therapy activities, medication refusal, and overall decreased patient satisfaction. To address this issue, this pilot project was conducted to improve the voluntary admission process on the adult inpatient unit of a major academic medical center in North Carolina.
In April 2008, The Joint Commission’s Center for Transforming Healthcare embarked on an enterprise-wide initiative called Robust Process Improvement (RPI). RPI was developed as a blended approach in applying Six Sigma, Lean, and Change Management techniques to improve medical processes and procedures. RPI techniques were applied in this study to better define the problems related to inpatient behavioral health unit admission and discharge by collecting data, obtaining staff involvement, creating a solution, and monitoring for lasting benefit.
Methods
This quality improvement project took place at an 885-bed tertiary care academic medical center with Level 1 Trauma Center designation. Institutional Review Board approval was not required because this was performed as a quality improvement project rather than an experimental clinical trial and was not designed to create new generalizable knowledge.
The techniques used to improve outcomes on the inpatient behavioral health unit included Active Listening, Elevator Speech, Statistics, Cause and Effect Diagrams, development of a Communication Plan, Brainstorming, and Standard Work. Through interviews with physician assistants, nurses, and resident physicians conducted over a 1-month period, it became clear that there was confusion among patients surrounding the voluntary admission process, the process for requesting discharge, and the possibility of a voluntary admission being converted to an involuntary one. Active listening was used to better understand the opportunities for improvement from multiple perspectives through varying stages of the admission—from the consent process in the emergency department, admission to the unit, throughout the hospital stay, to the time of discharge. The following elevator speech was used to highlight the areas of confusion and the importance of implementing change with the team involved in implementing the new admission procedures:
Our project is about improving patient understanding of the voluntary admission process to the Adult Psychiatry Unit and the 72-hour rule. This is important because the present process leads to patient misunderstanding, discontent on the unit, resistance to provided therapies, and low Press Ganey satisfaction scores. Success will look like reduced patient confusion about the 72-hour rule, increased group participation, decreased patient-staff conflict, and improved Press Ganey scores. What we are asking from you is to use a standardized, scripted informed consent process, flow chart, and pocket card during the voluntary admission process.
Additionally, brainstorming sessions were conducted with physician assistants, nurses, and residents to discuss options to improve the process and elicit a list of barriers.
Data Gathering
Several metrics were tracked to further understand the issue. Overall Press Ganey scores, in addition to admission and discharge subsection scores, were tracked for the 8 months prior to implementing the quality improvement procedures (March 2017-October 2017). Additionally, the treatment team members answered survey questions related to perceived patient understanding at the beginning of training sessions in the 5-week period immediately prior to implementing the quality improvement procedures. This survey was administered using a 6-point Likert scale, ranging from 1 (never) to 6 (frequently), and included the following questions:
- How often do you have to explain: (a) Request for Voluntary Admission form, (b) 72-Hour Rule, (c) Request to Discharge form?
- How often is there confusion about the 72-hour rule and RTD form once admitted to the adult inpatient psychiatric unit?
- How often do you need to re-explain the 72-hour rule and RTD form once admitted to the adult inpatient psychiatric unit?
In-depth interviews were conducted with 4 resident physicians, 4 physician assistants, and 3 nurses to identify specific shortcomings of the admission procedure. A key finding from these interviews was that some patients tended not to understand that the treatment team had 72 hours to respond to the RTD application. Instead, several patients had indicated that they thought that they could immediately discharge themselves since they were on the unit “voluntarily” or that they could categorically discharge themselves after 72 hours of being admitted. This feedback was crucial in determining the next steps that could be taken to minimize confusion.
Process Changes
In preparation for this quality improvement project, the language and layout of the Request for Voluntary Admission form was revised and approved internally by the hospital’s Forms Committee to emphasize the 72-hour rule and the process for completing a RTD form. Additionally, these interviews indicated that it was difficult to track patients who were admitted voluntarily versus involuntarily. To rectify this problem, a field was added to the electronic medical record system to include current psychiatric admission status, allowing the selection to be either “Voluntary” or “Involuntary.” This new field in the electronic medical record system gives nurses the ability to easily update legal status daily as appropriate, which minimizes the risk of information not being effectively communicated at shift changes, while also allowing various members of the treatment team to be updated on the admission status of each patient.
After reviewing data and obtaining staff involvement related to the problem, a new psychiatric admission and consent process was created. The new consent and admission process was characterized by a standardized procedure and scripted language to present to candidates for voluntary admission. The standardized procedure begins with the admitting staff member reading scripted consent language from a pocket card that includes 3 key points describing the voluntary admission procedure (see script in Figure 1). The first key point is to describe the 72-hour rule. Next, the staff member describes the purpose of the RTD form and shows an example to the patient. Finally, the staff member responsible for consenting describes the possibility of the patient being required to remain on the unit involuntarily in the event that he or she wants to leave before the treatment team sees fit and is deemed to be a danger to himself or herself or others.

On the reverse side of the pocket card, an example of the RTD form is available to show to the patient. The subsequent teach-back procedure is summarized using the flow chart in Figure 2, and this was made available to staff who participate in the admission and discharge processes. After reading the consent script, the consenting staff member must ask the patient to recall the 3 key points. For each key point that the patient cannot recall, the relevant section of the scripted language is re-read to the patient, who is asked to explain it again. If the patient recalls the 3 key points, then he or she is deemed to have cognitive capacity and thus can become a candidate for voluntary admission.
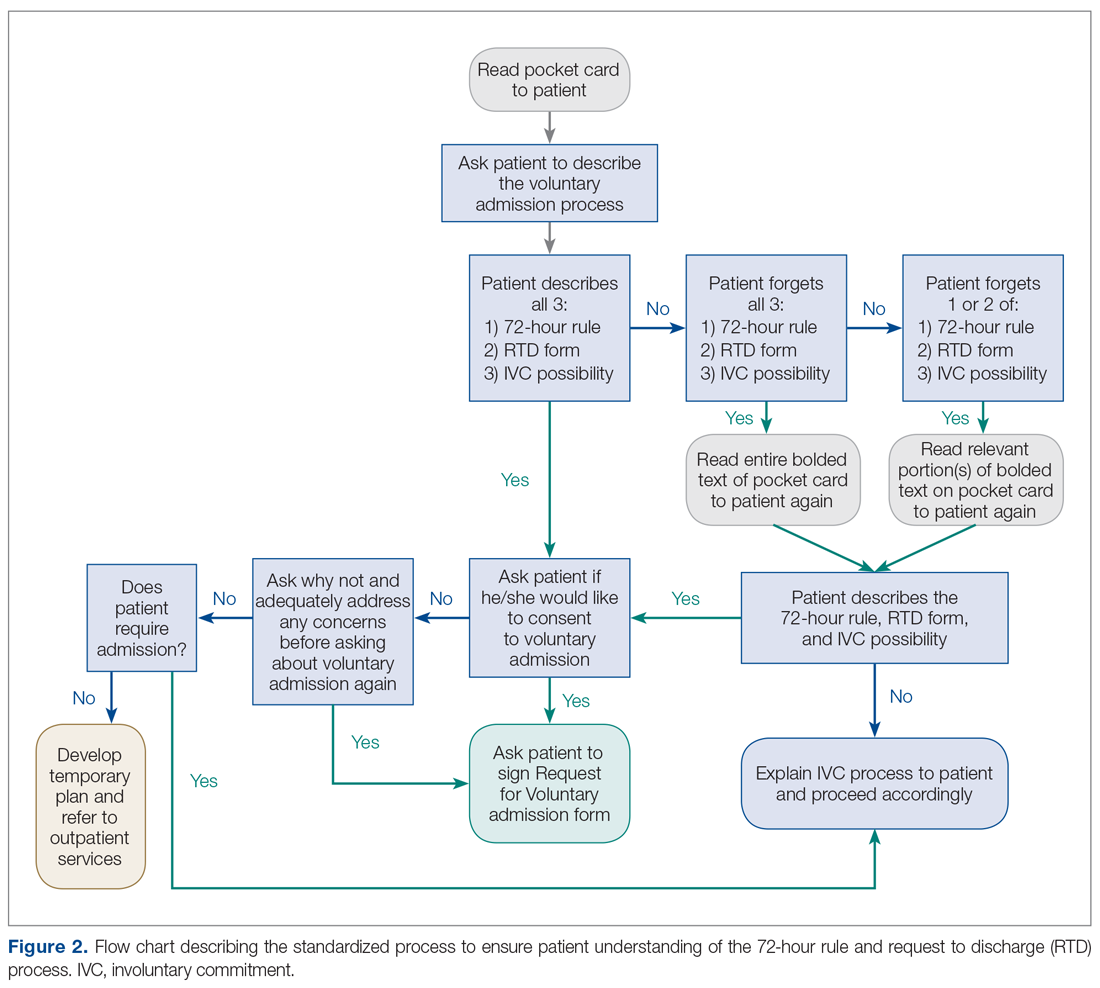
Flow charts, scripts, and pocket cards were created and distributed to relevant physicians, physician assistants, and nurses who participate in the admission or discharge process. Additional copies of pocket cards were made available within the department. In October 2017, an attending psychiatrist and medical student trained psychiatry physician assistants, nurses, and resident physicians who participated in the admission process in the ED or patient care on the unit on how to use the new materials. The new process was first implemented on November 1, 2017.
Measurements
Press Ganey scores were compared for 8 months before and 8 months after implementing the new process to monitor changes from the patients’ perspective. Additionally, the treatment team members answered survey questions related to perceived patient understanding at the beginning of training sessions in mid-September through mid-October and again 5 weeks after the new process was implemented.
Results
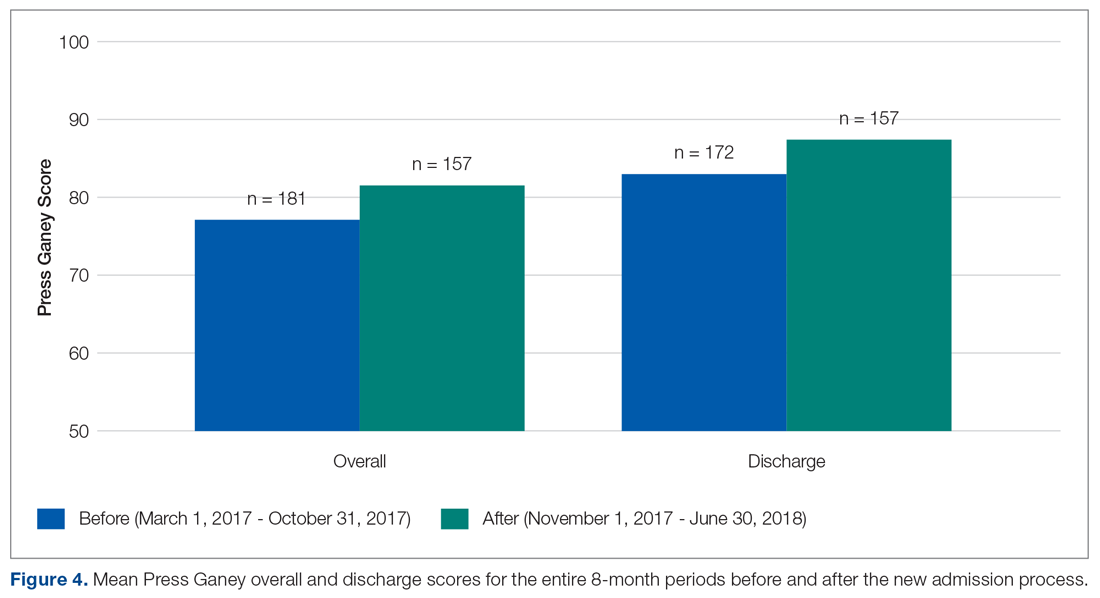
The behavioral health unit’s Press Ganey (overall and discharge) scores increased during the 8-month period following implementation of the quality improvement project (Figure 3). There was a notable upward trend of overall and discharge Press Ganey scores on a month-by-month basis from November through April. In total, 181 Press Ganey score reports were available for the 6-month period prior to the new process versus 157 score reports after (Figure 4). The average overall Press Ganey score for respondents improved from 77.1 to 81.6 (P = 0.003), while the average discharge score improved from 83.0 to 87.5 (P = 0.023).
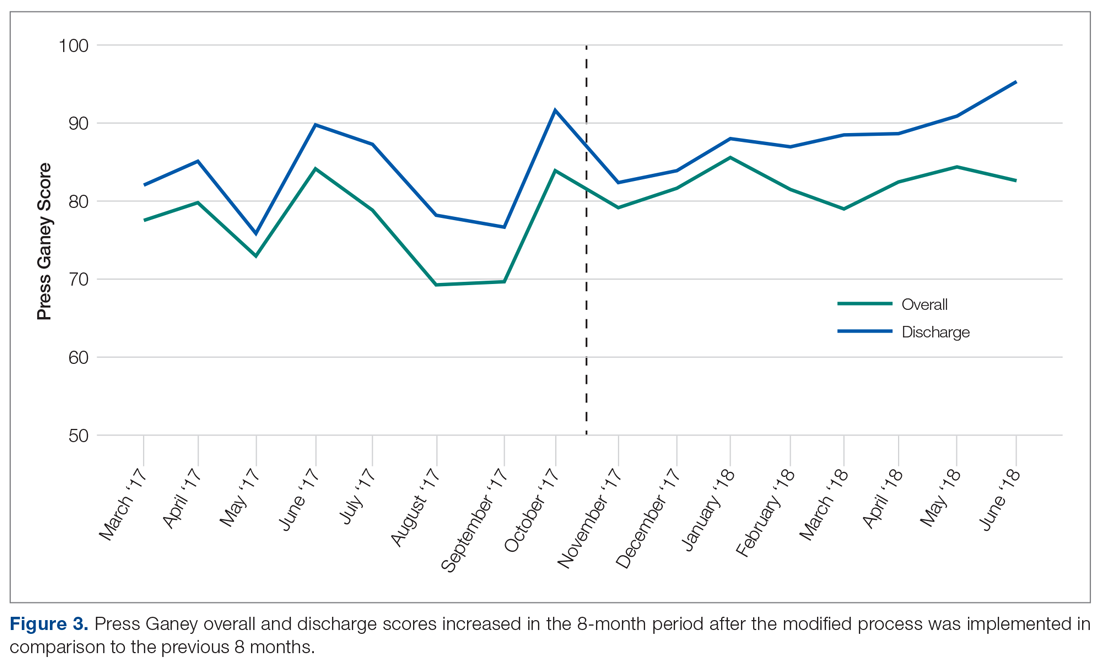
In recent months, the behavioral health discharge satisfaction score has become one of the highest performing aspects of the department according to Press Ganey reports. From April through June 2018, the department has performed in the 98th percentile or higher in “information about patient’s rights” during admission and “discharge instructions if help is needed.”
The survey related to perceived patient satisfaction and confusion also indicated significant improvement. Survey respondents indicated that there was less confusion about the 72-hour rule and RTD form after the quality improvement procedure was implemented (P = 0.039) and that fewer attempts to re-explain these concepts were required as well (P = 0.035).
Discussion
The Press Ganey scores for this unit indicated an improvement in patient satisfaction, in particular with the discharge process. While the overall Press Ganey scores on the inpatient behavioral health unit showed a significant improvement, it remained stagnant, around 80, during the 8-month period after implementing the new standardized admission process. However, the discharge score consistently improved over the same 8 months, from 82 to 95 in the most recent month. Also, the overall and discharge scores indicated a brief spike/improvement in October, immediately preceding the implementation of the new scripted language. Given the timeline, this spike is likely related to the ongoing meetings, trainings, and awareness of the upcoming process improvement.
With hospital and health system reimbursements becoming increasingly tied with patient outcomes, quality improvement efforts to improve patient care and satisfaction are of the utmost importance. In order to develop the rapport with patients needed for a high level of cooperation and excellent outcomes on an inpatient psychiatric unit, it is essential that all patients receive specific information about what the admission entails and what the options are for being discharged from the unit. Since a voluntary admission can be converted to an involuntary admission if a patient is deemed a threat to himself or herself or others despite already signing a RTD form, it is essential that this is not only discussed prior to admission, but that these details are explicitly checked for understanding. This allows the treatment team to assess for capacity and the patient to demonstrate informed consent. Differing expectations or understanding in what the voluntary admission or discharge process entails can lead to patient frustration and hostility, lack of trust in the treatment team, poor attendance and participation in group therapy activities, and medication refusal. Altogether, this can lead to longer inpatient stays, increased costs, decreased outcomes, and decreased patient satisfaction.
These initiatives were relatively easy to implement and are backed by evidence that they ultimately increased patient satisfaction. These findings could be extended to other institutions to improve the voluntary admission process and, ultimately, the patient experience. Additionally, the methods could be applied to other patient care processes within psychiatric facilities, and to improve other aspects of the patient care experience that have room for improvement, as illustrated by the department’s Press Ganey subsection scores, or areas that the treatment team would like to focus on.
Limitations
There are several limitations in the design and evaluation of this project. The assessment of patient understanding, especially in psychiatric patients, is very difficult to quantify. The principal measure of assessing patient understanding was limited to health care professional survey results. This may have led to a slight social desirability bias. An objective assessment of understanding directly from the patients was not readily attainable in our study, but future studies could look at this metric in addition to health care professional survey results.
Additionally, the overall Press Ganey scores may be influenced by factors beyond the admission process and the applied improvement procedures. It is difficult to discern whether there were any other factors that also contributed to the overall increase. However, the discharge score was a more direct measure specifically related to the modified procedures, and the temporal association of the intervention with the increased scores suggests that the intervention was responsible.
Conclusion
Standardization of the consent process ensures that all patients receive the necessary information every time in busy clinical settings. Incorporating an opportunity to “teach back” specific important information about the voluntary admission process, specifically what the 72-hour rule is, what the RTD form is, and the possibility of involuntary commitment, allows clinicians to assess capacity, while simultaneously allowing patients to have realistic expectations of the admission. Concise, standardized answers regarding these points minimizes variation in information being dispersed and decreases the possibility of omitting important information. At a major academic medical center, easy-to-implement quality improvement techniques significantly decreased patient confusion surrounding the 72-hour rule and the RTD form, along with the frequency in which these policies needed to be re-explained on the adult inpatient psychiatric unit. These changes ultimately led to improvement in patient satisfaction, as indicated by significant improvement in both overall and discharge patient satisfaction scores.
Corresponding author: Jennifer F. Newman, 475 Vine St., Winston-Salem, NC, 27101; [email protected].
Financial disclosures: None.
1. Redfern E, Brown R, Vincent C. Improving communication in the emergency department. Emerg Med J. 2009;26:658-661.
2. Payne CE, Stein JM, Leong T, Dressler DD. Avoiding handover fumbles: a controlled trial of a structured handover tool versus traditional handover methods. BMJ Qual Saf. 2012;21:925-932.
3. Weller J, Boyd M, Cumin D. Teams, tribes and patient safety: overcoming barriers to effective teamwork in healthcare. Postgrad Med J. 2014;90:149-154.
4. DiMatteo MR. The role of the physician in the emerging health care environment. Western J Med. 1998;168:328.
5. The Psychiatry Milestone Project. J Grad Med Educ. 2014;6(1 Suppl 1):284-304.
6. Garakani A, Shalenberg E, Burstin SC, et al. Voluntary psychiatric hospitalization and patient-driven requests for discharge: a statutory review and analysis of implications for the capacity to consent to voluntary hospitalization. Harv Rev Psychiatry. 2014;22:241-249.
7. Procedure for Admission and Discharge of Clients Act, 211 § 122C-211(2014).
From the Wake Forest School of Medicine (Ms. Newman), and Wake Forest Baptist Health, Department of Psychiatry and Behavioral Medicine (Dr. Kramer), Winston-Salem, NC.
Abstract
- Background: Adults voluntarily admitted to inpatient behavioral health units can ask to sign a Request to Discharge (RTD) form if they would like to be discharged before the treatment team agrees that discharge is appropriate. This gives the team 72 hours to determine whether the patient is safe to discharge or to involuntarily commit the patient to the unit. At 1 medical center, patients who were offered voluntary admission often lacked complete understanding of the “72-hour rule” and the early discharge procedure.
- Methods: Robust Process Improvement® techniques were implemented to improve the admission process. Flow charts, standardized scripts, and pocket cards were distributed to relevant staff. The Request for Voluntary Admission form was revised to emphasize the “72-hour rule” and the process for requesting a RTD form.
- Results: The unit’s average overall Press Ganey score improved from 77.1 to 81.6 (P = 0.003), while the average discharge score improved from 83.0 to 87.5 (P = 0.023) following implementation of the new process.
- Conclusion: Incorporating strategies such as an opportunity to “teach back” important information about the voluntary admission process (ie, what the 72-hour rule is, what the request to discharge form is, and the possibility of involuntary commitment) allows clinicians to assess capacity while simultaneously giving patients realistic expectations of the admission. These changes can lead to improvement in patient satisfaction.
Keywords: behavioral health; communication; patient satisfaction.
Communication is paramount within medical teams to improve outcomes and strengthen rapport with patients, particularly with psychiatric patients in acute crisis. Studies
While some forms of communication are required to protect the safety of patients and others around them, other forms are required to build strong relationships with patients. However, these 2 goals do not have to be mutually exclusive in the psychiatric hospital environment. Hospitals aim to improve patient satisfaction while simultaneously providing effective communication about treatment. Studies have indicated that communication during graduate medical training may decline due to “emotional and physical brutality” associated with residency training programs.4 To ameliorate this and emphasize communication education, accredited psychiatry residency programs require residents to use structured communication tools to achieve a level 2 in the Accreditation Council for Graduate Medical Education milestone project for the category of patient safety and health care team.5 These standardized processes allow all patients to receive the same important information related to their care while minimizing human error. Such communication skills aim to improve health care outcomes and satisfaction for patients while also training better physicians.
For legal and ethical reasons, the adult inpatient behavioral health units at major hospitals are highly regulated. In most states, a patient who is admitted to an adult inpatient behavioral health unit on a voluntary basis can ask to sign a request to discharge (RTD) form if he or she would like to be discharged from the hospital before the treatment team sees fit.6 In most jurisdictions, this action gives the treatment team 72 hours to determine whether the patient is safe to discharge. Within that time frame, the physician must either discharge the patient, or, if it is not safe to do so, involuntarily commit him or her to the unit. In most jurisdictions, this process is commonly referred to as the “72-hour rule.”
In North Carolina, state legislation Chapter 122C, Article 5, Part 2(b) specifies: “In 24-hour facilities the application shall acknowledge that the applicant may be held by the facility for a period of 72 hours after any written request for release that the applicant may make, and shall acknowledge that the 24-hour facility may have the legal right to petition for involuntary commitment of the applicant during that period. At the time of application, the facility shall tell the applicant about procedures for discharge.”7 This requirement can be somewhat confusing for both medical team members and patients alike.
As formerly practiced on the behavioral health unit described in this report, patients offered voluntary admission status to the inpatient behavioral unit often lacked complete understanding of the 72-hour rule and the process for requesting early discharge from the facility. We hypothesized that this led to the observed patient frustration and hostility, lack of trust in the treatment team, poor attendance and participation in group therapy activities, medication refusal, and overall decreased patient satisfaction. To address this issue, this pilot project was conducted to improve the voluntary admission process on the adult inpatient unit of a major academic medical center in North Carolina.
In April 2008, The Joint Commission’s Center for Transforming Healthcare embarked on an enterprise-wide initiative called Robust Process Improvement (RPI). RPI was developed as a blended approach in applying Six Sigma, Lean, and Change Management techniques to improve medical processes and procedures. RPI techniques were applied in this study to better define the problems related to inpatient behavioral health unit admission and discharge by collecting data, obtaining staff involvement, creating a solution, and monitoring for lasting benefit.
Methods
This quality improvement project took place at an 885-bed tertiary care academic medical center with Level 1 Trauma Center designation. Institutional Review Board approval was not required because this was performed as a quality improvement project rather than an experimental clinical trial and was not designed to create new generalizable knowledge.
The techniques used to improve outcomes on the inpatient behavioral health unit included Active Listening, Elevator Speech, Statistics, Cause and Effect Diagrams, development of a Communication Plan, Brainstorming, and Standard Work. Through interviews with physician assistants, nurses, and resident physicians conducted over a 1-month period, it became clear that there was confusion among patients surrounding the voluntary admission process, the process for requesting discharge, and the possibility of a voluntary admission being converted to an involuntary one. Active listening was used to better understand the opportunities for improvement from multiple perspectives through varying stages of the admission—from the consent process in the emergency department, admission to the unit, throughout the hospital stay, to the time of discharge. The following elevator speech was used to highlight the areas of confusion and the importance of implementing change with the team involved in implementing the new admission procedures:
Our project is about improving patient understanding of the voluntary admission process to the Adult Psychiatry Unit and the 72-hour rule. This is important because the present process leads to patient misunderstanding, discontent on the unit, resistance to provided therapies, and low Press Ganey satisfaction scores. Success will look like reduced patient confusion about the 72-hour rule, increased group participation, decreased patient-staff conflict, and improved Press Ganey scores. What we are asking from you is to use a standardized, scripted informed consent process, flow chart, and pocket card during the voluntary admission process.
Additionally, brainstorming sessions were conducted with physician assistants, nurses, and residents to discuss options to improve the process and elicit a list of barriers.
Data Gathering
Several metrics were tracked to further understand the issue. Overall Press Ganey scores, in addition to admission and discharge subsection scores, were tracked for the 8 months prior to implementing the quality improvement procedures (March 2017-October 2017). Additionally, the treatment team members answered survey questions related to perceived patient understanding at the beginning of training sessions in the 5-week period immediately prior to implementing the quality improvement procedures. This survey was administered using a 6-point Likert scale, ranging from 1 (never) to 6 (frequently), and included the following questions:
- How often do you have to explain: (a) Request for Voluntary Admission form, (b) 72-Hour Rule, (c) Request to Discharge form?
- How often is there confusion about the 72-hour rule and RTD form once admitted to the adult inpatient psychiatric unit?
- How often do you need to re-explain the 72-hour rule and RTD form once admitted to the adult inpatient psychiatric unit?
In-depth interviews were conducted with 4 resident physicians, 4 physician assistants, and 3 nurses to identify specific shortcomings of the admission procedure. A key finding from these interviews was that some patients tended not to understand that the treatment team had 72 hours to respond to the RTD application. Instead, several patients had indicated that they thought that they could immediately discharge themselves since they were on the unit “voluntarily” or that they could categorically discharge themselves after 72 hours of being admitted. This feedback was crucial in determining the next steps that could be taken to minimize confusion.
Process Changes
In preparation for this quality improvement project, the language and layout of the Request for Voluntary Admission form was revised and approved internally by the hospital’s Forms Committee to emphasize the 72-hour rule and the process for completing a RTD form. Additionally, these interviews indicated that it was difficult to track patients who were admitted voluntarily versus involuntarily. To rectify this problem, a field was added to the electronic medical record system to include current psychiatric admission status, allowing the selection to be either “Voluntary” or “Involuntary.” This new field in the electronic medical record system gives nurses the ability to easily update legal status daily as appropriate, which minimizes the risk of information not being effectively communicated at shift changes, while also allowing various members of the treatment team to be updated on the admission status of each patient.
After reviewing data and obtaining staff involvement related to the problem, a new psychiatric admission and consent process was created. The new consent and admission process was characterized by a standardized procedure and scripted language to present to candidates for voluntary admission. The standardized procedure begins with the admitting staff member reading scripted consent language from a pocket card that includes 3 key points describing the voluntary admission procedure (see script in Figure 1). The first key point is to describe the 72-hour rule. Next, the staff member describes the purpose of the RTD form and shows an example to the patient. Finally, the staff member responsible for consenting describes the possibility of the patient being required to remain on the unit involuntarily in the event that he or she wants to leave before the treatment team sees fit and is deemed to be a danger to himself or herself or others.

On the reverse side of the pocket card, an example of the RTD form is available to show to the patient. The subsequent teach-back procedure is summarized using the flow chart in Figure 2, and this was made available to staff who participate in the admission and discharge processes. After reading the consent script, the consenting staff member must ask the patient to recall the 3 key points. For each key point that the patient cannot recall, the relevant section of the scripted language is re-read to the patient, who is asked to explain it again. If the patient recalls the 3 key points, then he or she is deemed to have cognitive capacity and thus can become a candidate for voluntary admission.

Flow charts, scripts, and pocket cards were created and distributed to relevant physicians, physician assistants, and nurses who participate in the admission or discharge process. Additional copies of pocket cards were made available within the department. In October 2017, an attending psychiatrist and medical student trained psychiatry physician assistants, nurses, and resident physicians who participated in the admission process in the ED or patient care on the unit on how to use the new materials. The new process was first implemented on November 1, 2017.
Measurements
Press Ganey scores were compared for 8 months before and 8 months after implementing the new process to monitor changes from the patients’ perspective. Additionally, the treatment team members answered survey questions related to perceived patient understanding at the beginning of training sessions in mid-September through mid-October and again 5 weeks after the new process was implemented.
Results
The behavioral health unit’s Press Ganey (overall and discharge) scores increased during the 8-month period following implementation of the quality improvement project (Figure 3). There was a notable upward trend of overall and discharge Press Ganey scores on a month-by-month basis from November through April. In total, 181 Press Ganey score reports were available for the 6-month period prior to the new process versus 157 score reports after (Figure 4). The average overall Press Ganey score for respondents improved from 77.1 to 81.6 (P = 0.003), while the average discharge score improved from 83.0 to 87.5 (P = 0.023).

In recent months, the behavioral health discharge satisfaction score has become one of the highest performing aspects of the department according to Press Ganey reports. From April through June 2018, the department has performed in the 98th percentile or higher in “information about patient’s rights” during admission and “discharge instructions if help is needed.”

The survey related to perceived patient satisfaction and confusion also indicated significant improvement. Survey respondents indicated that there was less confusion about the 72-hour rule and RTD form after the quality improvement procedure was implemented (P = 0.039) and that fewer attempts to re-explain these concepts were required as well (P = 0.035).
Discussion
The Press Ganey scores for this unit indicated an improvement in patient satisfaction, in particular with the discharge process. While the overall Press Ganey scores on the inpatient behavioral health unit showed a significant improvement, it remained stagnant, around 80, during the 8-month period after implementing the new standardized admission process. However, the discharge score consistently improved over the same 8 months, from 82 to 95 in the most recent month. Also, the overall and discharge scores indicated a brief spike/improvement in October, immediately preceding the implementation of the new scripted language. Given the timeline, this spike is likely related to the ongoing meetings, trainings, and awareness of the upcoming process improvement.
With hospital and health system reimbursements becoming increasingly tied with patient outcomes, quality improvement efforts to improve patient care and satisfaction are of the utmost importance. In order to develop the rapport with patients needed for a high level of cooperation and excellent outcomes on an inpatient psychiatric unit, it is essential that all patients receive specific information about what the admission entails and what the options are for being discharged from the unit. Since a voluntary admission can be converted to an involuntary admission if a patient is deemed a threat to himself or herself or others despite already signing a RTD form, it is essential that this is not only discussed prior to admission, but that these details are explicitly checked for understanding. This allows the treatment team to assess for capacity and the patient to demonstrate informed consent. Differing expectations or understanding in what the voluntary admission or discharge process entails can lead to patient frustration and hostility, lack of trust in the treatment team, poor attendance and participation in group therapy activities, and medication refusal. Altogether, this can lead to longer inpatient stays, increased costs, decreased outcomes, and decreased patient satisfaction.
These initiatives were relatively easy to implement and are backed by evidence that they ultimately increased patient satisfaction. These findings could be extended to other institutions to improve the voluntary admission process and, ultimately, the patient experience. Additionally, the methods could be applied to other patient care processes within psychiatric facilities, and to improve other aspects of the patient care experience that have room for improvement, as illustrated by the department’s Press Ganey subsection scores, or areas that the treatment team would like to focus on.
Limitations
There are several limitations in the design and evaluation of this project. The assessment of patient understanding, especially in psychiatric patients, is very difficult to quantify. The principal measure of assessing patient understanding was limited to health care professional survey results. This may have led to a slight social desirability bias. An objective assessment of understanding directly from the patients was not readily attainable in our study, but future studies could look at this metric in addition to health care professional survey results.
Additionally, the overall Press Ganey scores may be influenced by factors beyond the admission process and the applied improvement procedures. It is difficult to discern whether there were any other factors that also contributed to the overall increase. However, the discharge score was a more direct measure specifically related to the modified procedures, and the temporal association of the intervention with the increased scores suggests that the intervention was responsible.
Conclusion
Standardization of the consent process ensures that all patients receive the necessary information every time in busy clinical settings. Incorporating an opportunity to “teach back” specific important information about the voluntary admission process, specifically what the 72-hour rule is, what the RTD form is, and the possibility of involuntary commitment, allows clinicians to assess capacity, while simultaneously allowing patients to have realistic expectations of the admission. Concise, standardized answers regarding these points minimizes variation in information being dispersed and decreases the possibility of omitting important information. At a major academic medical center, easy-to-implement quality improvement techniques significantly decreased patient confusion surrounding the 72-hour rule and the RTD form, along with the frequency in which these policies needed to be re-explained on the adult inpatient psychiatric unit. These changes ultimately led to improvement in patient satisfaction, as indicated by significant improvement in both overall and discharge patient satisfaction scores.
Corresponding author: Jennifer F. Newman, 475 Vine St., Winston-Salem, NC, 27101; [email protected].
Financial disclosures: None.
From the Wake Forest School of Medicine (Ms. Newman), and Wake Forest Baptist Health, Department of Psychiatry and Behavioral Medicine (Dr. Kramer), Winston-Salem, NC.
Abstract
- Background: Adults voluntarily admitted to inpatient behavioral health units can ask to sign a Request to Discharge (RTD) form if they would like to be discharged before the treatment team agrees that discharge is appropriate. This gives the team 72 hours to determine whether the patient is safe to discharge or to involuntarily commit the patient to the unit. At 1 medical center, patients who were offered voluntary admission often lacked complete understanding of the “72-hour rule” and the early discharge procedure.
- Methods: Robust Process Improvement® techniques were implemented to improve the admission process. Flow charts, standardized scripts, and pocket cards were distributed to relevant staff. The Request for Voluntary Admission form was revised to emphasize the “72-hour rule” and the process for requesting a RTD form.
- Results: The unit’s average overall Press Ganey score improved from 77.1 to 81.6 (P = 0.003), while the average discharge score improved from 83.0 to 87.5 (P = 0.023) following implementation of the new process.
- Conclusion: Incorporating strategies such as an opportunity to “teach back” important information about the voluntary admission process (ie, what the 72-hour rule is, what the request to discharge form is, and the possibility of involuntary commitment) allows clinicians to assess capacity while simultaneously giving patients realistic expectations of the admission. These changes can lead to improvement in patient satisfaction.
Keywords: behavioral health; communication; patient satisfaction.
Communication is paramount within medical teams to improve outcomes and strengthen rapport with patients, particularly with psychiatric patients in acute crisis. Studies
While some forms of communication are required to protect the safety of patients and others around them, other forms are required to build strong relationships with patients. However, these 2 goals do not have to be mutually exclusive in the psychiatric hospital environment. Hospitals aim to improve patient satisfaction while simultaneously providing effective communication about treatment. Studies have indicated that communication during graduate medical training may decline due to “emotional and physical brutality” associated with residency training programs.4 To ameliorate this and emphasize communication education, accredited psychiatry residency programs require residents to use structured communication tools to achieve a level 2 in the Accreditation Council for Graduate Medical Education milestone project for the category of patient safety and health care team.5 These standardized processes allow all patients to receive the same important information related to their care while minimizing human error. Such communication skills aim to improve health care outcomes and satisfaction for patients while also training better physicians.
For legal and ethical reasons, the adult inpatient behavioral health units at major hospitals are highly regulated. In most states, a patient who is admitted to an adult inpatient behavioral health unit on a voluntary basis can ask to sign a request to discharge (RTD) form if he or she would like to be discharged from the hospital before the treatment team sees fit.6 In most jurisdictions, this action gives the treatment team 72 hours to determine whether the patient is safe to discharge. Within that time frame, the physician must either discharge the patient, or, if it is not safe to do so, involuntarily commit him or her to the unit. In most jurisdictions, this process is commonly referred to as the “72-hour rule.”
In North Carolina, state legislation Chapter 122C, Article 5, Part 2(b) specifies: “In 24-hour facilities the application shall acknowledge that the applicant may be held by the facility for a period of 72 hours after any written request for release that the applicant may make, and shall acknowledge that the 24-hour facility may have the legal right to petition for involuntary commitment of the applicant during that period. At the time of application, the facility shall tell the applicant about procedures for discharge.”7 This requirement can be somewhat confusing for both medical team members and patients alike.
As formerly practiced on the behavioral health unit described in this report, patients offered voluntary admission status to the inpatient behavioral unit often lacked complete understanding of the 72-hour rule and the process for requesting early discharge from the facility. We hypothesized that this led to the observed patient frustration and hostility, lack of trust in the treatment team, poor attendance and participation in group therapy activities, medication refusal, and overall decreased patient satisfaction. To address this issue, this pilot project was conducted to improve the voluntary admission process on the adult inpatient unit of a major academic medical center in North Carolina.
In April 2008, The Joint Commission’s Center for Transforming Healthcare embarked on an enterprise-wide initiative called Robust Process Improvement (RPI). RPI was developed as a blended approach in applying Six Sigma, Lean, and Change Management techniques to improve medical processes and procedures. RPI techniques were applied in this study to better define the problems related to inpatient behavioral health unit admission and discharge by collecting data, obtaining staff involvement, creating a solution, and monitoring for lasting benefit.
Methods
This quality improvement project took place at an 885-bed tertiary care academic medical center with Level 1 Trauma Center designation. Institutional Review Board approval was not required because this was performed as a quality improvement project rather than an experimental clinical trial and was not designed to create new generalizable knowledge.
The techniques used to improve outcomes on the inpatient behavioral health unit included Active Listening, Elevator Speech, Statistics, Cause and Effect Diagrams, development of a Communication Plan, Brainstorming, and Standard Work. Through interviews with physician assistants, nurses, and resident physicians conducted over a 1-month period, it became clear that there was confusion among patients surrounding the voluntary admission process, the process for requesting discharge, and the possibility of a voluntary admission being converted to an involuntary one. Active listening was used to better understand the opportunities for improvement from multiple perspectives through varying stages of the admission—from the consent process in the emergency department, admission to the unit, throughout the hospital stay, to the time of discharge. The following elevator speech was used to highlight the areas of confusion and the importance of implementing change with the team involved in implementing the new admission procedures:
Our project is about improving patient understanding of the voluntary admission process to the Adult Psychiatry Unit and the 72-hour rule. This is important because the present process leads to patient misunderstanding, discontent on the unit, resistance to provided therapies, and low Press Ganey satisfaction scores. Success will look like reduced patient confusion about the 72-hour rule, increased group participation, decreased patient-staff conflict, and improved Press Ganey scores. What we are asking from you is to use a standardized, scripted informed consent process, flow chart, and pocket card during the voluntary admission process.
Additionally, brainstorming sessions were conducted with physician assistants, nurses, and residents to discuss options to improve the process and elicit a list of barriers.
Data Gathering
Several metrics were tracked to further understand the issue. Overall Press Ganey scores, in addition to admission and discharge subsection scores, were tracked for the 8 months prior to implementing the quality improvement procedures (March 2017-October 2017). Additionally, the treatment team members answered survey questions related to perceived patient understanding at the beginning of training sessions in the 5-week period immediately prior to implementing the quality improvement procedures. This survey was administered using a 6-point Likert scale, ranging from 1 (never) to 6 (frequently), and included the following questions:
- How often do you have to explain: (a) Request for Voluntary Admission form, (b) 72-Hour Rule, (c) Request to Discharge form?
- How often is there confusion about the 72-hour rule and RTD form once admitted to the adult inpatient psychiatric unit?
- How often do you need to re-explain the 72-hour rule and RTD form once admitted to the adult inpatient psychiatric unit?
In-depth interviews were conducted with 4 resident physicians, 4 physician assistants, and 3 nurses to identify specific shortcomings of the admission procedure. A key finding from these interviews was that some patients tended not to understand that the treatment team had 72 hours to respond to the RTD application. Instead, several patients had indicated that they thought that they could immediately discharge themselves since they were on the unit “voluntarily” or that they could categorically discharge themselves after 72 hours of being admitted. This feedback was crucial in determining the next steps that could be taken to minimize confusion.
Process Changes
In preparation for this quality improvement project, the language and layout of the Request for Voluntary Admission form was revised and approved internally by the hospital’s Forms Committee to emphasize the 72-hour rule and the process for completing a RTD form. Additionally, these interviews indicated that it was difficult to track patients who were admitted voluntarily versus involuntarily. To rectify this problem, a field was added to the electronic medical record system to include current psychiatric admission status, allowing the selection to be either “Voluntary” or “Involuntary.” This new field in the electronic medical record system gives nurses the ability to easily update legal status daily as appropriate, which minimizes the risk of information not being effectively communicated at shift changes, while also allowing various members of the treatment team to be updated on the admission status of each patient.
After reviewing data and obtaining staff involvement related to the problem, a new psychiatric admission and consent process was created. The new consent and admission process was characterized by a standardized procedure and scripted language to present to candidates for voluntary admission. The standardized procedure begins with the admitting staff member reading scripted consent language from a pocket card that includes 3 key points describing the voluntary admission procedure (see script in Figure 1). The first key point is to describe the 72-hour rule. Next, the staff member describes the purpose of the RTD form and shows an example to the patient. Finally, the staff member responsible for consenting describes the possibility of the patient being required to remain on the unit involuntarily in the event that he or she wants to leave before the treatment team sees fit and is deemed to be a danger to himself or herself or others.

On the reverse side of the pocket card, an example of the RTD form is available to show to the patient. The subsequent teach-back procedure is summarized using the flow chart in Figure 2, and this was made available to staff who participate in the admission and discharge processes. After reading the consent script, the consenting staff member must ask the patient to recall the 3 key points. For each key point that the patient cannot recall, the relevant section of the scripted language is re-read to the patient, who is asked to explain it again. If the patient recalls the 3 key points, then he or she is deemed to have cognitive capacity and thus can become a candidate for voluntary admission.

Flow charts, scripts, and pocket cards were created and distributed to relevant physicians, physician assistants, and nurses who participate in the admission or discharge process. Additional copies of pocket cards were made available within the department. In October 2017, an attending psychiatrist and medical student trained psychiatry physician assistants, nurses, and resident physicians who participated in the admission process in the ED or patient care on the unit on how to use the new materials. The new process was first implemented on November 1, 2017.
Measurements
Press Ganey scores were compared for 8 months before and 8 months after implementing the new process to monitor changes from the patients’ perspective. Additionally, the treatment team members answered survey questions related to perceived patient understanding at the beginning of training sessions in mid-September through mid-October and again 5 weeks after the new process was implemented.
Results
The behavioral health unit’s Press Ganey (overall and discharge) scores increased during the 8-month period following implementation of the quality improvement project (Figure 3). There was a notable upward trend of overall and discharge Press Ganey scores on a month-by-month basis from November through April. In total, 181 Press Ganey score reports were available for the 6-month period prior to the new process versus 157 score reports after (Figure 4). The average overall Press Ganey score for respondents improved from 77.1 to 81.6 (P = 0.003), while the average discharge score improved from 83.0 to 87.5 (P = 0.023).

In recent months, the behavioral health discharge satisfaction score has become one of the highest performing aspects of the department according to Press Ganey reports. From April through June 2018, the department has performed in the 98th percentile or higher in “information about patient’s rights” during admission and “discharge instructions if help is needed.”

The survey related to perceived patient satisfaction and confusion also indicated significant improvement. Survey respondents indicated that there was less confusion about the 72-hour rule and RTD form after the quality improvement procedure was implemented (P = 0.039) and that fewer attempts to re-explain these concepts were required as well (P = 0.035).
Discussion
The Press Ganey scores for this unit indicated an improvement in patient satisfaction, in particular with the discharge process. While the overall Press Ganey scores on the inpatient behavioral health unit showed a significant improvement, it remained stagnant, around 80, during the 8-month period after implementing the new standardized admission process. However, the discharge score consistently improved over the same 8 months, from 82 to 95 in the most recent month. Also, the overall and discharge scores indicated a brief spike/improvement in October, immediately preceding the implementation of the new scripted language. Given the timeline, this spike is likely related to the ongoing meetings, trainings, and awareness of the upcoming process improvement.
With hospital and health system reimbursements becoming increasingly tied with patient outcomes, quality improvement efforts to improve patient care and satisfaction are of the utmost importance. In order to develop the rapport with patients needed for a high level of cooperation and excellent outcomes on an inpatient psychiatric unit, it is essential that all patients receive specific information about what the admission entails and what the options are for being discharged from the unit. Since a voluntary admission can be converted to an involuntary admission if a patient is deemed a threat to himself or herself or others despite already signing a RTD form, it is essential that this is not only discussed prior to admission, but that these details are explicitly checked for understanding. This allows the treatment team to assess for capacity and the patient to demonstrate informed consent. Differing expectations or understanding in what the voluntary admission or discharge process entails can lead to patient frustration and hostility, lack of trust in the treatment team, poor attendance and participation in group therapy activities, and medication refusal. Altogether, this can lead to longer inpatient stays, increased costs, decreased outcomes, and decreased patient satisfaction.
These initiatives were relatively easy to implement and are backed by evidence that they ultimately increased patient satisfaction. These findings could be extended to other institutions to improve the voluntary admission process and, ultimately, the patient experience. Additionally, the methods could be applied to other patient care processes within psychiatric facilities, and to improve other aspects of the patient care experience that have room for improvement, as illustrated by the department’s Press Ganey subsection scores, or areas that the treatment team would like to focus on.
Limitations
There are several limitations in the design and evaluation of this project. The assessment of patient understanding, especially in psychiatric patients, is very difficult to quantify. The principal measure of assessing patient understanding was limited to health care professional survey results. This may have led to a slight social desirability bias. An objective assessment of understanding directly from the patients was not readily attainable in our study, but future studies could look at this metric in addition to health care professional survey results.
Additionally, the overall Press Ganey scores may be influenced by factors beyond the admission process and the applied improvement procedures. It is difficult to discern whether there were any other factors that also contributed to the overall increase. However, the discharge score was a more direct measure specifically related to the modified procedures, and the temporal association of the intervention with the increased scores suggests that the intervention was responsible.
Conclusion
Standardization of the consent process ensures that all patients receive the necessary information every time in busy clinical settings. Incorporating an opportunity to “teach back” specific important information about the voluntary admission process, specifically what the 72-hour rule is, what the RTD form is, and the possibility of involuntary commitment, allows clinicians to assess capacity, while simultaneously allowing patients to have realistic expectations of the admission. Concise, standardized answers regarding these points minimizes variation in information being dispersed and decreases the possibility of omitting important information. At a major academic medical center, easy-to-implement quality improvement techniques significantly decreased patient confusion surrounding the 72-hour rule and the RTD form, along with the frequency in which these policies needed to be re-explained on the adult inpatient psychiatric unit. These changes ultimately led to improvement in patient satisfaction, as indicated by significant improvement in both overall and discharge patient satisfaction scores.
Corresponding author: Jennifer F. Newman, 475 Vine St., Winston-Salem, NC, 27101; [email protected].
Financial disclosures: None.
1. Redfern E, Brown R, Vincent C. Improving communication in the emergency department. Emerg Med J. 2009;26:658-661.
2. Payne CE, Stein JM, Leong T, Dressler DD. Avoiding handover fumbles: a controlled trial of a structured handover tool versus traditional handover methods. BMJ Qual Saf. 2012;21:925-932.
3. Weller J, Boyd M, Cumin D. Teams, tribes and patient safety: overcoming barriers to effective teamwork in healthcare. Postgrad Med J. 2014;90:149-154.
4. DiMatteo MR. The role of the physician in the emerging health care environment. Western J Med. 1998;168:328.
5. The Psychiatry Milestone Project. J Grad Med Educ. 2014;6(1 Suppl 1):284-304.
6. Garakani A, Shalenberg E, Burstin SC, et al. Voluntary psychiatric hospitalization and patient-driven requests for discharge: a statutory review and analysis of implications for the capacity to consent to voluntary hospitalization. Harv Rev Psychiatry. 2014;22:241-249.
7. Procedure for Admission and Discharge of Clients Act, 211 § 122C-211(2014).
1. Redfern E, Brown R, Vincent C. Improving communication in the emergency department. Emerg Med J. 2009;26:658-661.
2. Payne CE, Stein JM, Leong T, Dressler DD. Avoiding handover fumbles: a controlled trial of a structured handover tool versus traditional handover methods. BMJ Qual Saf. 2012;21:925-932.
3. Weller J, Boyd M, Cumin D. Teams, tribes and patient safety: overcoming barriers to effective teamwork in healthcare. Postgrad Med J. 2014;90:149-154.
4. DiMatteo MR. The role of the physician in the emerging health care environment. Western J Med. 1998;168:328.
5. The Psychiatry Milestone Project. J Grad Med Educ. 2014;6(1 Suppl 1):284-304.
6. Garakani A, Shalenberg E, Burstin SC, et al. Voluntary psychiatric hospitalization and patient-driven requests for discharge: a statutory review and analysis of implications for the capacity to consent to voluntary hospitalization. Harv Rev Psychiatry. 2014;22:241-249.
7. Procedure for Admission and Discharge of Clients Act, 211 § 122C-211(2014).
Cardioprotective Effect of Metformin in Patients with Decreased Renal Function
Study Overview
Objective. To assess whether metformin use is associated with lower risk of fatal or nonfatal major adverse cardiovascular events (MACE) as compared to sulfonylurea use among diabetic patients with reduced kidney function.
Design. Retrospective cohort study of US Veterans receiving care within the Veterans Health Administration, with data supplemented by linkage to Medicare, Medicaid, and National Death Index data from 2001 through 2016.
Setting and participants. A retrospective cohort of Veterans Health Administration (VHA) patients, aged 18 years and older. Pharmacy data included medication, date filled, days supplied, and number of pills dispensed. For Medicare and Medicaid patients, enrollees’ claims files and prescription (Part D) data were obtained. In addition, dates and cause of death were obtained from vital status and the National Death Index files.
Patients with new-onset type 2 diabetes were identified by selecting new users of metformin, glipizide, glyburide, or glimepiride. These patients were followed longitudinally and the date of cohort entry and start of follow-up was the day of reaching a reduced kidney function threshold, defined as either an estimated glomerular filtration rate (eGFR) of less than 60 mL/m
Main outcome measures. Primary outcome was the composite of MACE including hospitalization for acute myocardial infarction (AMI), ischemic or hemorrhagic stroke, transient ischemic attack (TIA), or date of cardiovascular death. The secondary outcome excluded TIA as part of the composite MACE event because not all patients who sustain a TIA are admitted to the hospital.
Main results. From January 1, 2002 through December 30, 2015, 67,749 new metformin users and 28,976 new sulfonylurea users who persisted with treatment were identified. After using propensity score-weighted matching, 24,679 metformin users and 24,799 sulfonylurea users entered the final analysis. Cohort patients were 98% male and 81.8% white. Metformin users were younger than sulfonylurea users, with a median age of 61 years versus 71 years, respectively.
For the main outcome, there were 1048 composite MACE events among metformin patients with reduced kidney function and 1394 MACE events among sulfonylurea patients, yielding 23.0 (95% confidence interval [CI], 21.7-24.4) versus 29.2 (95% CI, 27.7-30.7) events per 1000 person-years of use, respectively, after propensity score-weighting. After covariate adjustment, the cause-specific adjusted hazard ratio (aHR) for MACE was 0.80 (95% CI, 0.75-0.86) among metformin users compared with sulfonylurea users. The adjusted incidence rate difference was 5.8 (95% CI, 4.1-7.3) fewer events per 1000-person years for metformin compared with sulfonylurea users. Results were also consistent for each component of the primary outcome, including cardiovascular hospitalizations (aHR, 0.87; 95% CI, 0.80-0.95) and cardiovascular deaths (aHR, 0.70; 95% CI, 0.63-0.78).
Analysis of secondary outcomes, which included AMI, stroke, and cardiovascular death and excluded TIA, demonstrated similar results, with a cause-specific aHR of 0.78 (95% CI, 0.72-0.84) among metformin users compared with sulfonylurea users. The adjusted incidence rate difference was 5.9 (95% CI, 4.3-7.6) fewer events per 1000-person years for metformin compared with sulfonylurea users.
Conclusion. For patients with diabetes and reduced kidney function, treatment with metformin monotherapy, as compared with a sulfonylurea, was associated with a lower risk of MACE.
Commentary
There are approximately 30 million US adults with a diagnosis of type 2 diabetes (T2DM), of whom 20% also have impaired kidney function or chronic kidney disease (CKD).1 Metformin hydrochloride has remained the preferred first-line treatment for T2DM based on safety and effectiveness, as well as low cost.2 Metformin is eliminated by the kidneys and can accumulate as eGFR declines. Based on the negative clinical experience, the US Food and Drug Administration (FDA) issued a safety warning restricting metformin for patients with serum creatinine levels of 1.5 mg/dL or greater for men or 1.4 mg/dL or greater for women. The FDA recommended against starting metformin therapy in patients with CKD with eGFR between 30 and 45 mL/min/1.73 m2, although patients already taking metformin can continue with caution in that setting.1,3
There are several limitations in conducting observational studies comparing metformin to other glucose-lowering medications. First, metformin trials typically excluded patients with CKD due to the FDA warnings. Second, there is usually a time-lag bias in which patients who initiate glucose-lowering medications other than metformin are at a later stage of disease. Third, there is often an allocation bias, as there are substantial differences in baseline characteristics between metformin and sulfonylurea monotherapy users, with metformin users usually being younger and healthier.4
In this retrospective cohort study by Roumie et al, the authors used propensity score–weighted matching to reduce the impacts on time-lag and allocation bias. However, several major limitations remained in this study. First, the study design excluded those who began diabetes treatment after the onset of reduced kidney function; therefore, this study cannot be generalized to patients who already have reduced eGFR at the time of metformin initiation. Second, cohort entry and the start of follow-up was either an elevated serum creatinine or reduced eGFR less than 60 mL/min/1.73 m2. The cohort may have included some patients with an acute kidney injury event, rather than progression to CKD, who recovered from their acute kidney injury. Third, the study population was mostly elderly white men; together with the lack of dose analysis, this study may not be generalizable to other populations.
Applications for Clinical Practice
The current study demonstrated that metformin use, as compared to sulfonylureas, has a lower risk of fatal or nonfatal major adverse cardiovascular events among patients with reduced kidney function. When clinicians are managing hyperglycemia in patients with type 2 diabetes, it is important to keep in mind that all medications have adverse effects. There are now 11 drug classes for treating diabetes, in addition to multiple insulin options, and the challenge for clinicians is to present clear information to guide patients using shared decision making, based on each patient’s clinical circumstances and preferences, to achieve individualized glycemic target ranges.
–Ka Ming Gordon Ngai, MD, MPH
1. Geiss LS, Kirtland K, Lin J, et al. Changes in diagnosed diabetes, obesity, and physical inactivity prevalence in US counties, 2004-2012. PLoS One. 2017;12:e0173428.
2. Good CB, Pogach LM. Should metformin be first-line therapy for patients with type 2 diabetes and chronic kidney disease? JAMA Intern Med. 2018;178:911-912.
3. US Food and Drug Administration. FDA revises warnings regarding use of the diabetes medicine metformin in certain patients with reduced kidney function. https://www.fda.gov/downloads/Drugs/DrugSafety/UCM494140.pdf. Accessed September 30, 2019.
4. Wexler DJ. Sulfonylureas and cardiovascular safety the final verdict? JAMA. 2019;322:1147-1149.
Study Overview
Objective. To assess whether metformin use is associated with lower risk of fatal or nonfatal major adverse cardiovascular events (MACE) as compared to sulfonylurea use among diabetic patients with reduced kidney function.
Design. Retrospective cohort study of US Veterans receiving care within the Veterans Health Administration, with data supplemented by linkage to Medicare, Medicaid, and National Death Index data from 2001 through 2016.
Setting and participants. A retrospective cohort of Veterans Health Administration (VHA) patients, aged 18 years and older. Pharmacy data included medication, date filled, days supplied, and number of pills dispensed. For Medicare and Medicaid patients, enrollees’ claims files and prescription (Part D) data were obtained. In addition, dates and cause of death were obtained from vital status and the National Death Index files.
Patients with new-onset type 2 diabetes were identified by selecting new users of metformin, glipizide, glyburide, or glimepiride. These patients were followed longitudinally and the date of cohort entry and start of follow-up was the day of reaching a reduced kidney function threshold, defined as either an estimated glomerular filtration rate (eGFR) of less than 60 mL/m
Main outcome measures. Primary outcome was the composite of MACE including hospitalization for acute myocardial infarction (AMI), ischemic or hemorrhagic stroke, transient ischemic attack (TIA), or date of cardiovascular death. The secondary outcome excluded TIA as part of the composite MACE event because not all patients who sustain a TIA are admitted to the hospital.
Main results. From January 1, 2002 through December 30, 2015, 67,749 new metformin users and 28,976 new sulfonylurea users who persisted with treatment were identified. After using propensity score-weighted matching, 24,679 metformin users and 24,799 sulfonylurea users entered the final analysis. Cohort patients were 98% male and 81.8% white. Metformin users were younger than sulfonylurea users, with a median age of 61 years versus 71 years, respectively.
For the main outcome, there were 1048 composite MACE events among metformin patients with reduced kidney function and 1394 MACE events among sulfonylurea patients, yielding 23.0 (95% confidence interval [CI], 21.7-24.4) versus 29.2 (95% CI, 27.7-30.7) events per 1000 person-years of use, respectively, after propensity score-weighting. After covariate adjustment, the cause-specific adjusted hazard ratio (aHR) for MACE was 0.80 (95% CI, 0.75-0.86) among metformin users compared with sulfonylurea users. The adjusted incidence rate difference was 5.8 (95% CI, 4.1-7.3) fewer events per 1000-person years for metformin compared with sulfonylurea users. Results were also consistent for each component of the primary outcome, including cardiovascular hospitalizations (aHR, 0.87; 95% CI, 0.80-0.95) and cardiovascular deaths (aHR, 0.70; 95% CI, 0.63-0.78).
Analysis of secondary outcomes, which included AMI, stroke, and cardiovascular death and excluded TIA, demonstrated similar results, with a cause-specific aHR of 0.78 (95% CI, 0.72-0.84) among metformin users compared with sulfonylurea users. The adjusted incidence rate difference was 5.9 (95% CI, 4.3-7.6) fewer events per 1000-person years for metformin compared with sulfonylurea users.
Conclusion. For patients with diabetes and reduced kidney function, treatment with metformin monotherapy, as compared with a sulfonylurea, was associated with a lower risk of MACE.
Commentary
There are approximately 30 million US adults with a diagnosis of type 2 diabetes (T2DM), of whom 20% also have impaired kidney function or chronic kidney disease (CKD).1 Metformin hydrochloride has remained the preferred first-line treatment for T2DM based on safety and effectiveness, as well as low cost.2 Metformin is eliminated by the kidneys and can accumulate as eGFR declines. Based on the negative clinical experience, the US Food and Drug Administration (FDA) issued a safety warning restricting metformin for patients with serum creatinine levels of 1.5 mg/dL or greater for men or 1.4 mg/dL or greater for women. The FDA recommended against starting metformin therapy in patients with CKD with eGFR between 30 and 45 mL/min/1.73 m2, although patients already taking metformin can continue with caution in that setting.1,3
There are several limitations in conducting observational studies comparing metformin to other glucose-lowering medications. First, metformin trials typically excluded patients with CKD due to the FDA warnings. Second, there is usually a time-lag bias in which patients who initiate glucose-lowering medications other than metformin are at a later stage of disease. Third, there is often an allocation bias, as there are substantial differences in baseline characteristics between metformin and sulfonylurea monotherapy users, with metformin users usually being younger and healthier.4
In this retrospective cohort study by Roumie et al, the authors used propensity score–weighted matching to reduce the impacts on time-lag and allocation bias. However, several major limitations remained in this study. First, the study design excluded those who began diabetes treatment after the onset of reduced kidney function; therefore, this study cannot be generalized to patients who already have reduced eGFR at the time of metformin initiation. Second, cohort entry and the start of follow-up was either an elevated serum creatinine or reduced eGFR less than 60 mL/min/1.73 m2. The cohort may have included some patients with an acute kidney injury event, rather than progression to CKD, who recovered from their acute kidney injury. Third, the study population was mostly elderly white men; together with the lack of dose analysis, this study may not be generalizable to other populations.
Applications for Clinical Practice
The current study demonstrated that metformin use, as compared to sulfonylureas, has a lower risk of fatal or nonfatal major adverse cardiovascular events among patients with reduced kidney function. When clinicians are managing hyperglycemia in patients with type 2 diabetes, it is important to keep in mind that all medications have adverse effects. There are now 11 drug classes for treating diabetes, in addition to multiple insulin options, and the challenge for clinicians is to present clear information to guide patients using shared decision making, based on each patient’s clinical circumstances and preferences, to achieve individualized glycemic target ranges.
–Ka Ming Gordon Ngai, MD, MPH
Study Overview
Objective. To assess whether metformin use is associated with lower risk of fatal or nonfatal major adverse cardiovascular events (MACE) as compared to sulfonylurea use among diabetic patients with reduced kidney function.
Design. Retrospective cohort study of US Veterans receiving care within the Veterans Health Administration, with data supplemented by linkage to Medicare, Medicaid, and National Death Index data from 2001 through 2016.
Setting and participants. A retrospective cohort of Veterans Health Administration (VHA) patients, aged 18 years and older. Pharmacy data included medication, date filled, days supplied, and number of pills dispensed. For Medicare and Medicaid patients, enrollees’ claims files and prescription (Part D) data were obtained. In addition, dates and cause of death were obtained from vital status and the National Death Index files.
Patients with new-onset type 2 diabetes were identified by selecting new users of metformin, glipizide, glyburide, or glimepiride. These patients were followed longitudinally and the date of cohort entry and start of follow-up was the day of reaching a reduced kidney function threshold, defined as either an estimated glomerular filtration rate (eGFR) of less than 60 mL/m
Main outcome measures. Primary outcome was the composite of MACE including hospitalization for acute myocardial infarction (AMI), ischemic or hemorrhagic stroke, transient ischemic attack (TIA), or date of cardiovascular death. The secondary outcome excluded TIA as part of the composite MACE event because not all patients who sustain a TIA are admitted to the hospital.
Main results. From January 1, 2002 through December 30, 2015, 67,749 new metformin users and 28,976 new sulfonylurea users who persisted with treatment were identified. After using propensity score-weighted matching, 24,679 metformin users and 24,799 sulfonylurea users entered the final analysis. Cohort patients were 98% male and 81.8% white. Metformin users were younger than sulfonylurea users, with a median age of 61 years versus 71 years, respectively.
For the main outcome, there were 1048 composite MACE events among metformin patients with reduced kidney function and 1394 MACE events among sulfonylurea patients, yielding 23.0 (95% confidence interval [CI], 21.7-24.4) versus 29.2 (95% CI, 27.7-30.7) events per 1000 person-years of use, respectively, after propensity score-weighting. After covariate adjustment, the cause-specific adjusted hazard ratio (aHR) for MACE was 0.80 (95% CI, 0.75-0.86) among metformin users compared with sulfonylurea users. The adjusted incidence rate difference was 5.8 (95% CI, 4.1-7.3) fewer events per 1000-person years for metformin compared with sulfonylurea users. Results were also consistent for each component of the primary outcome, including cardiovascular hospitalizations (aHR, 0.87; 95% CI, 0.80-0.95) and cardiovascular deaths (aHR, 0.70; 95% CI, 0.63-0.78).
Analysis of secondary outcomes, which included AMI, stroke, and cardiovascular death and excluded TIA, demonstrated similar results, with a cause-specific aHR of 0.78 (95% CI, 0.72-0.84) among metformin users compared with sulfonylurea users. The adjusted incidence rate difference was 5.9 (95% CI, 4.3-7.6) fewer events per 1000-person years for metformin compared with sulfonylurea users.
Conclusion. For patients with diabetes and reduced kidney function, treatment with metformin monotherapy, as compared with a sulfonylurea, was associated with a lower risk of MACE.
Commentary
There are approximately 30 million US adults with a diagnosis of type 2 diabetes (T2DM), of whom 20% also have impaired kidney function or chronic kidney disease (CKD).1 Metformin hydrochloride has remained the preferred first-line treatment for T2DM based on safety and effectiveness, as well as low cost.2 Metformin is eliminated by the kidneys and can accumulate as eGFR declines. Based on the negative clinical experience, the US Food and Drug Administration (FDA) issued a safety warning restricting metformin for patients with serum creatinine levels of 1.5 mg/dL or greater for men or 1.4 mg/dL or greater for women. The FDA recommended against starting metformin therapy in patients with CKD with eGFR between 30 and 45 mL/min/1.73 m2, although patients already taking metformin can continue with caution in that setting.1,3
There are several limitations in conducting observational studies comparing metformin to other glucose-lowering medications. First, metformin trials typically excluded patients with CKD due to the FDA warnings. Second, there is usually a time-lag bias in which patients who initiate glucose-lowering medications other than metformin are at a later stage of disease. Third, there is often an allocation bias, as there are substantial differences in baseline characteristics between metformin and sulfonylurea monotherapy users, with metformin users usually being younger and healthier.4
In this retrospective cohort study by Roumie et al, the authors used propensity score–weighted matching to reduce the impacts on time-lag and allocation bias. However, several major limitations remained in this study. First, the study design excluded those who began diabetes treatment after the onset of reduced kidney function; therefore, this study cannot be generalized to patients who already have reduced eGFR at the time of metformin initiation. Second, cohort entry and the start of follow-up was either an elevated serum creatinine or reduced eGFR less than 60 mL/min/1.73 m2. The cohort may have included some patients with an acute kidney injury event, rather than progression to CKD, who recovered from their acute kidney injury. Third, the study population was mostly elderly white men; together with the lack of dose analysis, this study may not be generalizable to other populations.
Applications for Clinical Practice
The current study demonstrated that metformin use, as compared to sulfonylureas, has a lower risk of fatal or nonfatal major adverse cardiovascular events among patients with reduced kidney function. When clinicians are managing hyperglycemia in patients with type 2 diabetes, it is important to keep in mind that all medications have adverse effects. There are now 11 drug classes for treating diabetes, in addition to multiple insulin options, and the challenge for clinicians is to present clear information to guide patients using shared decision making, based on each patient’s clinical circumstances and preferences, to achieve individualized glycemic target ranges.
–Ka Ming Gordon Ngai, MD, MPH
1. Geiss LS, Kirtland K, Lin J, et al. Changes in diagnosed diabetes, obesity, and physical inactivity prevalence in US counties, 2004-2012. PLoS One. 2017;12:e0173428.
2. Good CB, Pogach LM. Should metformin be first-line therapy for patients with type 2 diabetes and chronic kidney disease? JAMA Intern Med. 2018;178:911-912.
3. US Food and Drug Administration. FDA revises warnings regarding use of the diabetes medicine metformin in certain patients with reduced kidney function. https://www.fda.gov/downloads/Drugs/DrugSafety/UCM494140.pdf. Accessed September 30, 2019.
4. Wexler DJ. Sulfonylureas and cardiovascular safety the final verdict? JAMA. 2019;322:1147-1149.
1. Geiss LS, Kirtland K, Lin J, et al. Changes in diagnosed diabetes, obesity, and physical inactivity prevalence in US counties, 2004-2012. PLoS One. 2017;12:e0173428.
2. Good CB, Pogach LM. Should metformin be first-line therapy for patients with type 2 diabetes and chronic kidney disease? JAMA Intern Med. 2018;178:911-912.
3. US Food and Drug Administration. FDA revises warnings regarding use of the diabetes medicine metformin in certain patients with reduced kidney function. https://www.fda.gov/downloads/Drugs/DrugSafety/UCM494140.pdf. Accessed September 30, 2019.
4. Wexler DJ. Sulfonylureas and cardiovascular safety the final verdict? JAMA. 2019;322:1147-1149.
Prasugrel Superior to Ticagrelor in Acute Coronary Syndromes
Study Overview
Objective. To assess the relative merits of ticagrelor compared to prasugrel in patients with acute coronary syndromes who will undergo invasive evaluation.
Design. Multicenter, open-label, prospective randomized controlled trial.
Setting and participants. A total of 4018 patients who presented with ACS with or without ST-segment elevation.
Intervention. Patients were randomly assigned to receive either ticagrelor or prasugrel.
Main outcome measures. The primary end point was the composite of death, myocardial infarction, or stroke at 1 year. The secondary end point was bleeding.
Main results. At 1 year, a primary end point event occurred in 184 of 2012 patients (9.3%) in the ticagrelor group and 137 of 2006 patients (6.9%) in the prasugrel group (hazard ratio [HR], 1.36; 95% confidence interval [CI], 1.09-1.70; P = 0.006). In the comparison between
Conclusion. In patients who presented with ACS with or without ST-segment elevation, the incidence of death, myocardial infarction, or stroke was significantly lower among those who received prasugrel as compared to those who received ticagrelor, and incidence of major bleeding was not significantly different.
Commentary
Dual antiplatelet therapy combining an adenosine disphosphate (ADP) receptor antagonist and aspirin is standard treatment for patients presenting with ACS. The limitation of clopidogrel has been its modest antiplatelet effect, with substantial interpatient variability. The newer generation thienopyridine prasugrel and the reversible direct-acting oral antagonist of the ADP receptor ticagrelor provide consistent and greater antiplatelet effect compared to clopidogrel. It has been previously reported that these agents are superior in reducing ischemic events when compared to clopidogrel.1,2 Therefore, current guidelines recommend ticagrelor and prasugrel in preference to clopidogrel.3,4 However, there has been no large randomized controlled study comparing the effect of ticagrelor and prasugrel. In this context, Shupke et al investigated this clinical question by performing a well-designed multicenter randomized controlled trial in patients presenting with ACS. At 12-month follow-up, the composite of death, myocardial infarction, and stroke occurred more frequently in the ticagrelor group compared to the prasugrel group (9.3% versus 6.9%; HR, 1.36; 95% CI, 1.09-1.70; P < 0.01). The incidence of major bleeding was not significantly different between the 2 groups (5.4% versus 4.8%; P = 0.46).
The strengths of this current study include the randomized design and the large number of patients enrolled, with adequate power to evaluate for superiority. This was a multicenter trial in Europe with 23 participating centers (21 from Germany). Furthermore, the interventional technique used by the operators reflects more contemporary technique compared to the previous studies comparing each agent to clopidogrel,1,2 with more frequent use of radial access (37%) and drug-eluting stents (90%) and reduced use of GPIIb/IIIa inhibitors (12%).
There are a few important points to consider due to the differences between the 2 agents compared in this study. First, the loading dose of ticagrelor and prasugrel was administered differently in patients presenting with ACS without ST elevation. Ticagrelor was administered as soon as possible prior to the coronary angiogram, but prasugrel was administered after the coronary anatomy was defined prior to the intervention, which is how this agent is administered in current clinical practice. Therefore, this trial was an open-label study that compared not only different medications, but different administration strategies. Second, ticagrelor and prasugrel have different side-effect profiles. The side effects unique to ticagrelor are dyspnea and bradycardia. On the other hand, a contraindication unique to prasugrel is patients with a history of transient ischemic attack or stroke due to increased risk of thrombotic and hemorrhagic stroke.1 In addition, prasugrel has increased bleeding risk in patients older than 75 years of age and those with low body weight (< 60 kg). In this study, the overall medication discontinuation rate was higher in the ticagrelor group specifically due to dyspnea, and the reduced dose of 5 mg of prasugrel was used in patients older than 75 years or with low body weight.
Since the timing of administration of ticagrelor (preloading prior to coronary angiography is recommended) is similar to that of clopidogrel, and given the theoretical benefit of reversible inhibition of the ADP receptor, ticagrelor has been used more commonly in clinical practice than prasugrel, and it has been implemented in the ACS protocol in many hospitals. In light of the results from this first head-to-head comparison utilizing more contemporary interventional techniques, these protocols may need to be adjusted in favor of prasugrel for patients presenting with ACS. However, given the difference in timing of administration and the difference in side-effect profile, operators must also tailor these agents depending on the patient profile.
Applications for Clinical Practice
In patients presenting with ACS, prasugrel was superior to ticagrelor, with a lower composite of death, myocardial infarction, and stroke at 12 months. Prasugrel should be considered as a first-line treatment for ACS.
– Taishi Hirai, MD, and Arun Kumar, MD, University of Missouri, Columbia, MO
1. Wiviott SD, Braunwald E, McCabe CH, et al. Prasugrel versus clopidogrel in patients with acute coronary syndromes. N Engl J Med. 2007;357:2001-2015.
2. Wallentin L, Becker RC, Budaj A, et al. Ticagrelor versus clopidogrel in patients with acute coronary syndromes. N Engl J Med. 2009;361:1045-1057.
3. Ibanez B, James S, Agewall S, et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: The Task Force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Society of Cardiology (ESC). Eur Heart J. 2018;39:119-177.
4. Levine GN, Bates ER, Bittl JA, et al. 2016 ACC/AHA guideline focused update on duration of dual antiplatelet therapy in patients with coronary artery disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2016;68:1082-1115.
Study Overview
Objective. To assess the relative merits of ticagrelor compared to prasugrel in patients with acute coronary syndromes who will undergo invasive evaluation.
Design. Multicenter, open-label, prospective randomized controlled trial.
Setting and participants. A total of 4018 patients who presented with ACS with or without ST-segment elevation.
Intervention. Patients were randomly assigned to receive either ticagrelor or prasugrel.
Main outcome measures. The primary end point was the composite of death, myocardial infarction, or stroke at 1 year. The secondary end point was bleeding.
Main results. At 1 year, a primary end point event occurred in 184 of 2012 patients (9.3%) in the ticagrelor group and 137 of 2006 patients (6.9%) in the prasugrel group (hazard ratio [HR], 1.36; 95% confidence interval [CI], 1.09-1.70; P = 0.006). In the comparison between
Conclusion. In patients who presented with ACS with or without ST-segment elevation, the incidence of death, myocardial infarction, or stroke was significantly lower among those who received prasugrel as compared to those who received ticagrelor, and incidence of major bleeding was not significantly different.
Commentary
Dual antiplatelet therapy combining an adenosine disphosphate (ADP) receptor antagonist and aspirin is standard treatment for patients presenting with ACS. The limitation of clopidogrel has been its modest antiplatelet effect, with substantial interpatient variability. The newer generation thienopyridine prasugrel and the reversible direct-acting oral antagonist of the ADP receptor ticagrelor provide consistent and greater antiplatelet effect compared to clopidogrel. It has been previously reported that these agents are superior in reducing ischemic events when compared to clopidogrel.1,2 Therefore, current guidelines recommend ticagrelor and prasugrel in preference to clopidogrel.3,4 However, there has been no large randomized controlled study comparing the effect of ticagrelor and prasugrel. In this context, Shupke et al investigated this clinical question by performing a well-designed multicenter randomized controlled trial in patients presenting with ACS. At 12-month follow-up, the composite of death, myocardial infarction, and stroke occurred more frequently in the ticagrelor group compared to the prasugrel group (9.3% versus 6.9%; HR, 1.36; 95% CI, 1.09-1.70; P < 0.01). The incidence of major bleeding was not significantly different between the 2 groups (5.4% versus 4.8%; P = 0.46).
The strengths of this current study include the randomized design and the large number of patients enrolled, with adequate power to evaluate for superiority. This was a multicenter trial in Europe with 23 participating centers (21 from Germany). Furthermore, the interventional technique used by the operators reflects more contemporary technique compared to the previous studies comparing each agent to clopidogrel,1,2 with more frequent use of radial access (37%) and drug-eluting stents (90%) and reduced use of GPIIb/IIIa inhibitors (12%).
There are a few important points to consider due to the differences between the 2 agents compared in this study. First, the loading dose of ticagrelor and prasugrel was administered differently in patients presenting with ACS without ST elevation. Ticagrelor was administered as soon as possible prior to the coronary angiogram, but prasugrel was administered after the coronary anatomy was defined prior to the intervention, which is how this agent is administered in current clinical practice. Therefore, this trial was an open-label study that compared not only different medications, but different administration strategies. Second, ticagrelor and prasugrel have different side-effect profiles. The side effects unique to ticagrelor are dyspnea and bradycardia. On the other hand, a contraindication unique to prasugrel is patients with a history of transient ischemic attack or stroke due to increased risk of thrombotic and hemorrhagic stroke.1 In addition, prasugrel has increased bleeding risk in patients older than 75 years of age and those with low body weight (< 60 kg). In this study, the overall medication discontinuation rate was higher in the ticagrelor group specifically due to dyspnea, and the reduced dose of 5 mg of prasugrel was used in patients older than 75 years or with low body weight.
Since the timing of administration of ticagrelor (preloading prior to coronary angiography is recommended) is similar to that of clopidogrel, and given the theoretical benefit of reversible inhibition of the ADP receptor, ticagrelor has been used more commonly in clinical practice than prasugrel, and it has been implemented in the ACS protocol in many hospitals. In light of the results from this first head-to-head comparison utilizing more contemporary interventional techniques, these protocols may need to be adjusted in favor of prasugrel for patients presenting with ACS. However, given the difference in timing of administration and the difference in side-effect profile, operators must also tailor these agents depending on the patient profile.
Applications for Clinical Practice
In patients presenting with ACS, prasugrel was superior to ticagrelor, with a lower composite of death, myocardial infarction, and stroke at 12 months. Prasugrel should be considered as a first-line treatment for ACS.
– Taishi Hirai, MD, and Arun Kumar, MD, University of Missouri, Columbia, MO
Study Overview
Objective. To assess the relative merits of ticagrelor compared to prasugrel in patients with acute coronary syndromes who will undergo invasive evaluation.
Design. Multicenter, open-label, prospective randomized controlled trial.
Setting and participants. A total of 4018 patients who presented with ACS with or without ST-segment elevation.
Intervention. Patients were randomly assigned to receive either ticagrelor or prasugrel.
Main outcome measures. The primary end point was the composite of death, myocardial infarction, or stroke at 1 year. The secondary end point was bleeding.
Main results. At 1 year, a primary end point event occurred in 184 of 2012 patients (9.3%) in the ticagrelor group and 137 of 2006 patients (6.9%) in the prasugrel group (hazard ratio [HR], 1.36; 95% confidence interval [CI], 1.09-1.70; P = 0.006). In the comparison between
Conclusion. In patients who presented with ACS with or without ST-segment elevation, the incidence of death, myocardial infarction, or stroke was significantly lower among those who received prasugrel as compared to those who received ticagrelor, and incidence of major bleeding was not significantly different.
Commentary
Dual antiplatelet therapy combining an adenosine disphosphate (ADP) receptor antagonist and aspirin is standard treatment for patients presenting with ACS. The limitation of clopidogrel has been its modest antiplatelet effect, with substantial interpatient variability. The newer generation thienopyridine prasugrel and the reversible direct-acting oral antagonist of the ADP receptor ticagrelor provide consistent and greater antiplatelet effect compared to clopidogrel. It has been previously reported that these agents are superior in reducing ischemic events when compared to clopidogrel.1,2 Therefore, current guidelines recommend ticagrelor and prasugrel in preference to clopidogrel.3,4 However, there has been no large randomized controlled study comparing the effect of ticagrelor and prasugrel. In this context, Shupke et al investigated this clinical question by performing a well-designed multicenter randomized controlled trial in patients presenting with ACS. At 12-month follow-up, the composite of death, myocardial infarction, and stroke occurred more frequently in the ticagrelor group compared to the prasugrel group (9.3% versus 6.9%; HR, 1.36; 95% CI, 1.09-1.70; P < 0.01). The incidence of major bleeding was not significantly different between the 2 groups (5.4% versus 4.8%; P = 0.46).
The strengths of this current study include the randomized design and the large number of patients enrolled, with adequate power to evaluate for superiority. This was a multicenter trial in Europe with 23 participating centers (21 from Germany). Furthermore, the interventional technique used by the operators reflects more contemporary technique compared to the previous studies comparing each agent to clopidogrel,1,2 with more frequent use of radial access (37%) and drug-eluting stents (90%) and reduced use of GPIIb/IIIa inhibitors (12%).
There are a few important points to consider due to the differences between the 2 agents compared in this study. First, the loading dose of ticagrelor and prasugrel was administered differently in patients presenting with ACS without ST elevation. Ticagrelor was administered as soon as possible prior to the coronary angiogram, but prasugrel was administered after the coronary anatomy was defined prior to the intervention, which is how this agent is administered in current clinical practice. Therefore, this trial was an open-label study that compared not only different medications, but different administration strategies. Second, ticagrelor and prasugrel have different side-effect profiles. The side effects unique to ticagrelor are dyspnea and bradycardia. On the other hand, a contraindication unique to prasugrel is patients with a history of transient ischemic attack or stroke due to increased risk of thrombotic and hemorrhagic stroke.1 In addition, prasugrel has increased bleeding risk in patients older than 75 years of age and those with low body weight (< 60 kg). In this study, the overall medication discontinuation rate was higher in the ticagrelor group specifically due to dyspnea, and the reduced dose of 5 mg of prasugrel was used in patients older than 75 years or with low body weight.
Since the timing of administration of ticagrelor (preloading prior to coronary angiography is recommended) is similar to that of clopidogrel, and given the theoretical benefit of reversible inhibition of the ADP receptor, ticagrelor has been used more commonly in clinical practice than prasugrel, and it has been implemented in the ACS protocol in many hospitals. In light of the results from this first head-to-head comparison utilizing more contemporary interventional techniques, these protocols may need to be adjusted in favor of prasugrel for patients presenting with ACS. However, given the difference in timing of administration and the difference in side-effect profile, operators must also tailor these agents depending on the patient profile.
Applications for Clinical Practice
In patients presenting with ACS, prasugrel was superior to ticagrelor, with a lower composite of death, myocardial infarction, and stroke at 12 months. Prasugrel should be considered as a first-line treatment for ACS.
– Taishi Hirai, MD, and Arun Kumar, MD, University of Missouri, Columbia, MO
1. Wiviott SD, Braunwald E, McCabe CH, et al. Prasugrel versus clopidogrel in patients with acute coronary syndromes. N Engl J Med. 2007;357:2001-2015.
2. Wallentin L, Becker RC, Budaj A, et al. Ticagrelor versus clopidogrel in patients with acute coronary syndromes. N Engl J Med. 2009;361:1045-1057.
3. Ibanez B, James S, Agewall S, et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: The Task Force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Society of Cardiology (ESC). Eur Heart J. 2018;39:119-177.
4. Levine GN, Bates ER, Bittl JA, et al. 2016 ACC/AHA guideline focused update on duration of dual antiplatelet therapy in patients with coronary artery disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2016;68:1082-1115.
1. Wiviott SD, Braunwald E, McCabe CH, et al. Prasugrel versus clopidogrel in patients with acute coronary syndromes. N Engl J Med. 2007;357:2001-2015.
2. Wallentin L, Becker RC, Budaj A, et al. Ticagrelor versus clopidogrel in patients with acute coronary syndromes. N Engl J Med. 2009;361:1045-1057.
3. Ibanez B, James S, Agewall S, et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: The Task Force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Society of Cardiology (ESC). Eur Heart J. 2018;39:119-177.
4. Levine GN, Bates ER, Bittl JA, et al. 2016 ACC/AHA guideline focused update on duration of dual antiplatelet therapy in patients with coronary artery disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2016;68:1082-1115.
Combination Encorafenib, Cetuximab, and Binimetinib Improves Survival in BRAF V600E–Mutated Metastatic Colon Cancer
Study Overview
Objective. To evaluate whether the combination of encorafenib plus cetuximab with or without the MEK inhibitor binimetin
Design. Global, multicenter, randomized, open-label, phase 3 trial.
Intervention. Patients were randomized in a 1:1:1 fashion to 1 of 3 groups: triplet-therapy group (encorafenib 300 mg daily, binimetinib 45 mg twice daily, and cetuximab 400 mg/m2 of body surface area initially, then 250 mg/m2 weekly), doublet-therapy group (encorafenib and cetuximab in same doses and schedule as the triplet-therapy group), and control group (investigators choice of cetuximab and irinotecan or cetuximab and FOLFIRI). The randomization was stratified by performance status and prior irinotecan use. Treatment was given until progression or unacceptable toxicities on a 28-day cycle. No crossover was permitted.
Setting and participants. 665 patients underwent randomization: 224 patients to triplet-therapy, 220 to doublet-therapy, and 221 to the control group. Eligible patients had histologically confirmed metastatic colorectal cancer with a BRAF V600E mutation. Patients all had disease progression after 1 or 2 previous lines of therapy.
Main outcome measures. The primary end point of the study was OS and objective response rate (ORR) in the triplet-therapy group compared with the control group. Secondary endpoints included OS in the doublet-therapy group compared with the control group, as well as progression-free survival (PFS), duration of response (DOR), and safety. Assessments were performed every 6 weeks for the first 24 weeks and then every 12 weeks thereafter.
Results. The baseline characteristics were well balanced between the treatment arms. At the time of data cutoff, the median duration of follow-up was 7.8 months for each group. The median OS was 9 months in the triplet-therapy group and 5.4 months in the control group (hazard ratio [HR] for death, 0.52; 95% confidence interval [CI], 0.39-0.70; P < 0.001). The median OS was 8.4 months for the doublet-therapy group, resulting in a significant reduction in the risk of death compared with the control group (HR, 0.6; 95% CI, 0.45-0.79; P < 0.001). The estimated 6-month survival was 71% for the triplet-therapy group, 65% for the doublet-therapy group, and 47% for the control group. The triplet-therapy group had a higher ORR compared with the control group (26% versus 2%, P < 0.001). The ORR in the doublet-therapy group was also significantly higher than that in the control group (20% versus 2%, P < 0.001). Complete responses were seen in 4% of patients in the triplet-therapy group, 5% of the doublet-therapy group, and no patients in the control group. PFS was significantly longer in both the triplet-therapy and doublet-therapy groups compared with the control group (median PFS: 4.3 months, 4.2 months, 1.5 months, respectively). This translated into a 62% and 60% reduction in the risk for disease progression or death in the triplet-therapy and doublet-therapy groups, respectively, compared to the control group.
The most common adverse event reported in the triplet-therapy group was gastrointestinal (GI) related (diarrhea, nausea, and vomiting), with grade 3 or higher GI toxicity seen in 10% of patients. Skin toxicity in the form of acneiform dermatitis was seen in almost 50% of those in the triplet-therapy arm; however, grade 3 or higher skin toxicity was uncommon (2%). Overall, adverse events grade 3 or higher were observed in 58% of those in the triplet-therapy group, 50% in the doublet-therapy group, and 61% in the control group. Adverse events leading to drug discontinuation occurred in 7% in the triplet-therapy group, 8% in the doublet-therapy group, and 11% in the control group. Three deaths were considered treatment related: 1 in the triplet-therapy group (bowel perforation) and 2 in the control group (anaphylaxis and respiratory failure).
Conclusion. The triplet-combination of encorafenib, binimetinib, and cetuximab as well as the doublet-regimen of encorafenib and cetuximab improved both PFS and OS in patients with metastatic, BRAF V600E–mutated colorectal cancer that has progressed after 1 or 2 lines of therapy.
Commentary
The current interim analysis of the BEACON CRC trial demonstrates improved response rates, PFS, and, importantly, OS with the triplet regimen of encorafenib, binimetinib, and cetuximab in patients with metastatic BRAF V600E–mutated colorectal cancer compared to standard irinotecan-based therapy. Similarly, a doublet-regimen of encorafenib and cetuximab also improved outcomes compared with irinotecan-based chemotherapy, resulting in significantly higher response rates, PFS, and OS.
BRAF mutations are seen in approximately 5% to 15% of colorectal cancers and are more commonly seen in right-sided disease. BRAF-mutated colorectal cancer has a poor prognosis, and the presence of a BRAF mutation is an independent prognostic factor for decreased survival.1 Previous work to improve outcomes in this subset of patients has been largely disappointing. For example, Kopetz and colleagues have previously shown that single-agent BRAF inhibition with vemurafenib in metastatic BRAF-mutated colorectal cancer did not show meaningful clinical activity.2 Preclinical studies have suggested that single-agent BRAF or MEK inhibition alone do not lead to sustained MAPK pathway inhibition. Mechanistically, inhibition of BRAF has been shown to lead to feedback activation of EGFR; thus, inhibition of BRAF alone does not lead to cessation of proliferation.3 In light of this, the combination of EGFR and BRAF inhibition has been an attractive therapeutic strategy. Yaeger and colleagues enrolled 15 patients in a pilot study looking at the efficacy and safety of the BRAF inhibitor vemurafenib and the EGFR antibody panitumumab in patients with BRAF-mutated metastatic colorectal cancer. In this cohort, combined BRAF and EGFR inhibition showed tumor regression in 10 of 12 patients.4 This finding was validated in other subsequent studies.5
The current study is the first phase 3 trial to validate the efficacy of BRAF, MEK, and EGFR inhibition in patients with BRAF-mutant metastatic colorectal cancer. The results of this study represent a very important step forward in treating this patient cohort that has historically had very poor clinical outcomes. The combination of encorafenib, binimetinib, and cetuximab improved OS by 48% compared with standard irinotecan-based chemotherapy. In light of this, we now have a chemotherapy-free targeted combination that improves survival and likely represents the new standard of care in patients with BRAF-mutated colorectal cancer after progression on 1 or 2 prior lines of therapy. Ongoing trials are being pursued to investigate the efficacy of these combinations in the upfront setting, and the results of these trials are eagerly awaited.
Applications for Clinical Practice
The combination of encorafenib, binimetinib, and cetuximab improved OS in patients with BRAF-mutated metastatic colorectal cancer after progression on 1 or 2 prior lines of therapy. This combination represents a potential new standard of care in this patient population.
–Daniel Isaac, DO, MS
1. Souglakos J, Philips J, Wang, R, et al. Prognostic and predictive value of common mutations for treatment response and survival in patients with metastatic colorectal cancer. Br J Cancer. 2009;101:465-472.
2. Kopetz S, Desai J, Chan E, et al. Phase II pilot study of vemurafenib in patients with metastatic BRAF-mutated colorectal cancer. J Clin Oncol. 2015;33:4032-4038.
3. Prahallad A, Sun C, Huang S, et al. Unresponsiveness of colon cancer to BRAF (V600e) inhibition through feedback activation of EGFR. Nature. 2012;483:100-103.
4. Yaeger R, Cercek A, O’Reilly EM, et al. Pilot trial of combined BRAF and EGFR inhibition in BRAF-mutant metastatic colorectal cancer patients. Clin Cancer Res. 2015;21:1313-1320.
5. Van Geel EMJM, Tabernero J, Elez E, et al. A phase Ib dose-escalation study of encorafenib and cetuximab with or without alpelisib in metastatic BRAF-mutant colorectal cancer. Cancer Discov. 2017;7:610-619.
Study Overview
Objective. To evaluate whether the combination of encorafenib plus cetuximab with or without the MEK inhibitor binimetin
Design. Global, multicenter, randomized, open-label, phase 3 trial.
Intervention. Patients were randomized in a 1:1:1 fashion to 1 of 3 groups: triplet-therapy group (encorafenib 300 mg daily, binimetinib 45 mg twice daily, and cetuximab 400 mg/m2 of body surface area initially, then 250 mg/m2 weekly), doublet-therapy group (encorafenib and cetuximab in same doses and schedule as the triplet-therapy group), and control group (investigators choice of cetuximab and irinotecan or cetuximab and FOLFIRI). The randomization was stratified by performance status and prior irinotecan use. Treatment was given until progression or unacceptable toxicities on a 28-day cycle. No crossover was permitted.
Setting and participants. 665 patients underwent randomization: 224 patients to triplet-therapy, 220 to doublet-therapy, and 221 to the control group. Eligible patients had histologically confirmed metastatic colorectal cancer with a BRAF V600E mutation. Patients all had disease progression after 1 or 2 previous lines of therapy.
Main outcome measures. The primary end point of the study was OS and objective response rate (ORR) in the triplet-therapy group compared with the control group. Secondary endpoints included OS in the doublet-therapy group compared with the control group, as well as progression-free survival (PFS), duration of response (DOR), and safety. Assessments were performed every 6 weeks for the first 24 weeks and then every 12 weeks thereafter.
Results. The baseline characteristics were well balanced between the treatment arms. At the time of data cutoff, the median duration of follow-up was 7.8 months for each group. The median OS was 9 months in the triplet-therapy group and 5.4 months in the control group (hazard ratio [HR] for death, 0.52; 95% confidence interval [CI], 0.39-0.70; P < 0.001). The median OS was 8.4 months for the doublet-therapy group, resulting in a significant reduction in the risk of death compared with the control group (HR, 0.6; 95% CI, 0.45-0.79; P < 0.001). The estimated 6-month survival was 71% for the triplet-therapy group, 65% for the doublet-therapy group, and 47% for the control group. The triplet-therapy group had a higher ORR compared with the control group (26% versus 2%, P < 0.001). The ORR in the doublet-therapy group was also significantly higher than that in the control group (20% versus 2%, P < 0.001). Complete responses were seen in 4% of patients in the triplet-therapy group, 5% of the doublet-therapy group, and no patients in the control group. PFS was significantly longer in both the triplet-therapy and doublet-therapy groups compared with the control group (median PFS: 4.3 months, 4.2 months, 1.5 months, respectively). This translated into a 62% and 60% reduction in the risk for disease progression or death in the triplet-therapy and doublet-therapy groups, respectively, compared to the control group.
The most common adverse event reported in the triplet-therapy group was gastrointestinal (GI) related (diarrhea, nausea, and vomiting), with grade 3 or higher GI toxicity seen in 10% of patients. Skin toxicity in the form of acneiform dermatitis was seen in almost 50% of those in the triplet-therapy arm; however, grade 3 or higher skin toxicity was uncommon (2%). Overall, adverse events grade 3 or higher were observed in 58% of those in the triplet-therapy group, 50% in the doublet-therapy group, and 61% in the control group. Adverse events leading to drug discontinuation occurred in 7% in the triplet-therapy group, 8% in the doublet-therapy group, and 11% in the control group. Three deaths were considered treatment related: 1 in the triplet-therapy group (bowel perforation) and 2 in the control group (anaphylaxis and respiratory failure).
Conclusion. The triplet-combination of encorafenib, binimetinib, and cetuximab as well as the doublet-regimen of encorafenib and cetuximab improved both PFS and OS in patients with metastatic, BRAF V600E–mutated colorectal cancer that has progressed after 1 or 2 lines of therapy.
Commentary
The current interim analysis of the BEACON CRC trial demonstrates improved response rates, PFS, and, importantly, OS with the triplet regimen of encorafenib, binimetinib, and cetuximab in patients with metastatic BRAF V600E–mutated colorectal cancer compared to standard irinotecan-based therapy. Similarly, a doublet-regimen of encorafenib and cetuximab also improved outcomes compared with irinotecan-based chemotherapy, resulting in significantly higher response rates, PFS, and OS.
BRAF mutations are seen in approximately 5% to 15% of colorectal cancers and are more commonly seen in right-sided disease. BRAF-mutated colorectal cancer has a poor prognosis, and the presence of a BRAF mutation is an independent prognostic factor for decreased survival.1 Previous work to improve outcomes in this subset of patients has been largely disappointing. For example, Kopetz and colleagues have previously shown that single-agent BRAF inhibition with vemurafenib in metastatic BRAF-mutated colorectal cancer did not show meaningful clinical activity.2 Preclinical studies have suggested that single-agent BRAF or MEK inhibition alone do not lead to sustained MAPK pathway inhibition. Mechanistically, inhibition of BRAF has been shown to lead to feedback activation of EGFR; thus, inhibition of BRAF alone does not lead to cessation of proliferation.3 In light of this, the combination of EGFR and BRAF inhibition has been an attractive therapeutic strategy. Yaeger and colleagues enrolled 15 patients in a pilot study looking at the efficacy and safety of the BRAF inhibitor vemurafenib and the EGFR antibody panitumumab in patients with BRAF-mutated metastatic colorectal cancer. In this cohort, combined BRAF and EGFR inhibition showed tumor regression in 10 of 12 patients.4 This finding was validated in other subsequent studies.5
The current study is the first phase 3 trial to validate the efficacy of BRAF, MEK, and EGFR inhibition in patients with BRAF-mutant metastatic colorectal cancer. The results of this study represent a very important step forward in treating this patient cohort that has historically had very poor clinical outcomes. The combination of encorafenib, binimetinib, and cetuximab improved OS by 48% compared with standard irinotecan-based chemotherapy. In light of this, we now have a chemotherapy-free targeted combination that improves survival and likely represents the new standard of care in patients with BRAF-mutated colorectal cancer after progression on 1 or 2 prior lines of therapy. Ongoing trials are being pursued to investigate the efficacy of these combinations in the upfront setting, and the results of these trials are eagerly awaited.
Applications for Clinical Practice
The combination of encorafenib, binimetinib, and cetuximab improved OS in patients with BRAF-mutated metastatic colorectal cancer after progression on 1 or 2 prior lines of therapy. This combination represents a potential new standard of care in this patient population.
–Daniel Isaac, DO, MS
Study Overview
Objective. To evaluate whether the combination of encorafenib plus cetuximab with or without the MEK inhibitor binimetin
Design. Global, multicenter, randomized, open-label, phase 3 trial.
Intervention. Patients were randomized in a 1:1:1 fashion to 1 of 3 groups: triplet-therapy group (encorafenib 300 mg daily, binimetinib 45 mg twice daily, and cetuximab 400 mg/m2 of body surface area initially, then 250 mg/m2 weekly), doublet-therapy group (encorafenib and cetuximab in same doses and schedule as the triplet-therapy group), and control group (investigators choice of cetuximab and irinotecan or cetuximab and FOLFIRI). The randomization was stratified by performance status and prior irinotecan use. Treatment was given until progression or unacceptable toxicities on a 28-day cycle. No crossover was permitted.
Setting and participants. 665 patients underwent randomization: 224 patients to triplet-therapy, 220 to doublet-therapy, and 221 to the control group. Eligible patients had histologically confirmed metastatic colorectal cancer with a BRAF V600E mutation. Patients all had disease progression after 1 or 2 previous lines of therapy.
Main outcome measures. The primary end point of the study was OS and objective response rate (ORR) in the triplet-therapy group compared with the control group. Secondary endpoints included OS in the doublet-therapy group compared with the control group, as well as progression-free survival (PFS), duration of response (DOR), and safety. Assessments were performed every 6 weeks for the first 24 weeks and then every 12 weeks thereafter.
Results. The baseline characteristics were well balanced between the treatment arms. At the time of data cutoff, the median duration of follow-up was 7.8 months for each group. The median OS was 9 months in the triplet-therapy group and 5.4 months in the control group (hazard ratio [HR] for death, 0.52; 95% confidence interval [CI], 0.39-0.70; P < 0.001). The median OS was 8.4 months for the doublet-therapy group, resulting in a significant reduction in the risk of death compared with the control group (HR, 0.6; 95% CI, 0.45-0.79; P < 0.001). The estimated 6-month survival was 71% for the triplet-therapy group, 65% for the doublet-therapy group, and 47% for the control group. The triplet-therapy group had a higher ORR compared with the control group (26% versus 2%, P < 0.001). The ORR in the doublet-therapy group was also significantly higher than that in the control group (20% versus 2%, P < 0.001). Complete responses were seen in 4% of patients in the triplet-therapy group, 5% of the doublet-therapy group, and no patients in the control group. PFS was significantly longer in both the triplet-therapy and doublet-therapy groups compared with the control group (median PFS: 4.3 months, 4.2 months, 1.5 months, respectively). This translated into a 62% and 60% reduction in the risk for disease progression or death in the triplet-therapy and doublet-therapy groups, respectively, compared to the control group.
The most common adverse event reported in the triplet-therapy group was gastrointestinal (GI) related (diarrhea, nausea, and vomiting), with grade 3 or higher GI toxicity seen in 10% of patients. Skin toxicity in the form of acneiform dermatitis was seen in almost 50% of those in the triplet-therapy arm; however, grade 3 or higher skin toxicity was uncommon (2%). Overall, adverse events grade 3 or higher were observed in 58% of those in the triplet-therapy group, 50% in the doublet-therapy group, and 61% in the control group. Adverse events leading to drug discontinuation occurred in 7% in the triplet-therapy group, 8% in the doublet-therapy group, and 11% in the control group. Three deaths were considered treatment related: 1 in the triplet-therapy group (bowel perforation) and 2 in the control group (anaphylaxis and respiratory failure).
Conclusion. The triplet-combination of encorafenib, binimetinib, and cetuximab as well as the doublet-regimen of encorafenib and cetuximab improved both PFS and OS in patients with metastatic, BRAF V600E–mutated colorectal cancer that has progressed after 1 or 2 lines of therapy.
Commentary
The current interim analysis of the BEACON CRC trial demonstrates improved response rates, PFS, and, importantly, OS with the triplet regimen of encorafenib, binimetinib, and cetuximab in patients with metastatic BRAF V600E–mutated colorectal cancer compared to standard irinotecan-based therapy. Similarly, a doublet-regimen of encorafenib and cetuximab also improved outcomes compared with irinotecan-based chemotherapy, resulting in significantly higher response rates, PFS, and OS.
BRAF mutations are seen in approximately 5% to 15% of colorectal cancers and are more commonly seen in right-sided disease. BRAF-mutated colorectal cancer has a poor prognosis, and the presence of a BRAF mutation is an independent prognostic factor for decreased survival.1 Previous work to improve outcomes in this subset of patients has been largely disappointing. For example, Kopetz and colleagues have previously shown that single-agent BRAF inhibition with vemurafenib in metastatic BRAF-mutated colorectal cancer did not show meaningful clinical activity.2 Preclinical studies have suggested that single-agent BRAF or MEK inhibition alone do not lead to sustained MAPK pathway inhibition. Mechanistically, inhibition of BRAF has been shown to lead to feedback activation of EGFR; thus, inhibition of BRAF alone does not lead to cessation of proliferation.3 In light of this, the combination of EGFR and BRAF inhibition has been an attractive therapeutic strategy. Yaeger and colleagues enrolled 15 patients in a pilot study looking at the efficacy and safety of the BRAF inhibitor vemurafenib and the EGFR antibody panitumumab in patients with BRAF-mutated metastatic colorectal cancer. In this cohort, combined BRAF and EGFR inhibition showed tumor regression in 10 of 12 patients.4 This finding was validated in other subsequent studies.5
The current study is the first phase 3 trial to validate the efficacy of BRAF, MEK, and EGFR inhibition in patients with BRAF-mutant metastatic colorectal cancer. The results of this study represent a very important step forward in treating this patient cohort that has historically had very poor clinical outcomes. The combination of encorafenib, binimetinib, and cetuximab improved OS by 48% compared with standard irinotecan-based chemotherapy. In light of this, we now have a chemotherapy-free targeted combination that improves survival and likely represents the new standard of care in patients with BRAF-mutated colorectal cancer after progression on 1 or 2 prior lines of therapy. Ongoing trials are being pursued to investigate the efficacy of these combinations in the upfront setting, and the results of these trials are eagerly awaited.
Applications for Clinical Practice
The combination of encorafenib, binimetinib, and cetuximab improved OS in patients with BRAF-mutated metastatic colorectal cancer after progression on 1 or 2 prior lines of therapy. This combination represents a potential new standard of care in this patient population.
–Daniel Isaac, DO, MS
1. Souglakos J, Philips J, Wang, R, et al. Prognostic and predictive value of common mutations for treatment response and survival in patients with metastatic colorectal cancer. Br J Cancer. 2009;101:465-472.
2. Kopetz S, Desai J, Chan E, et al. Phase II pilot study of vemurafenib in patients with metastatic BRAF-mutated colorectal cancer. J Clin Oncol. 2015;33:4032-4038.
3. Prahallad A, Sun C, Huang S, et al. Unresponsiveness of colon cancer to BRAF (V600e) inhibition through feedback activation of EGFR. Nature. 2012;483:100-103.
4. Yaeger R, Cercek A, O’Reilly EM, et al. Pilot trial of combined BRAF and EGFR inhibition in BRAF-mutant metastatic colorectal cancer patients. Clin Cancer Res. 2015;21:1313-1320.
5. Van Geel EMJM, Tabernero J, Elez E, et al. A phase Ib dose-escalation study of encorafenib and cetuximab with or without alpelisib in metastatic BRAF-mutant colorectal cancer. Cancer Discov. 2017;7:610-619.
1. Souglakos J, Philips J, Wang, R, et al. Prognostic and predictive value of common mutations for treatment response and survival in patients with metastatic colorectal cancer. Br J Cancer. 2009;101:465-472.
2. Kopetz S, Desai J, Chan E, et al. Phase II pilot study of vemurafenib in patients with metastatic BRAF-mutated colorectal cancer. J Clin Oncol. 2015;33:4032-4038.
3. Prahallad A, Sun C, Huang S, et al. Unresponsiveness of colon cancer to BRAF (V600e) inhibition through feedback activation of EGFR. Nature. 2012;483:100-103.
4. Yaeger R, Cercek A, O’Reilly EM, et al. Pilot trial of combined BRAF and EGFR inhibition in BRAF-mutant metastatic colorectal cancer patients. Clin Cancer Res. 2015;21:1313-1320.
5. Van Geel EMJM, Tabernero J, Elez E, et al. A phase Ib dose-escalation study of encorafenib and cetuximab with or without alpelisib in metastatic BRAF-mutant colorectal cancer. Cancer Discov. 2017;7:610-619.
CDC finds that efforts to reduce new HIV infections have stalled
, according to a Vital Signs report published by the Centers for Disease Control and Prevention based upon a simultaneous MMWR Early Release. The report indicates that many Americans with HIV are not aware of their status or are not receiving effective treatment. Furthermore, the data suggest that few Americans who could benefit from preexposure prophylaxis (PrEP), a daily pill that prevents HIV, are receiving it.
The report “shows that HIV testing, treatment, and prevention have not reached enough Americans, and it emphasizes the continued urgent need to increase these interventions,” said Jay C. Butler, MD, deputy director for infectious diseases at the CDC, at a press conference. “We made a lot of progress in the late ’90s and into the early part of the 21st century in reducing the number of new cases of HIV. But HIV prevention progress has stalled in America since 2013. This stalling underscores the need to increase resources, deploy new technologies, and build expertise, particularly in areas where they’re needed most.”
To achieve these objectives, the CDC has proposed a federal initiative called Ending the HIV Epidemic: A Plan for America. The goal of the initiative is to reduce new HIV infections by 90% by 2030, in part by expanding access to PrEP medications.
Data suggest shortcomings in diagnosis, treatment, and prevention
In its review of data on HIV testing and treatment in 2017, the Vital Signs report found that approximately 154,000 people with HIV (that is, 14% of the total population with HIV) were unaware that they had the virus. These patients consequently could not take advantage of HIV treatment to maintain health, control the virus, and prevent HIV transmission. Young people aged 13-24 years were less likely to know their HIV status than did those aged 25 years and older, according to the report.
Furthermore, approximately two-thirds (63%) of patients who knew that they had HIV had the virus under control through effective treatment. Young people and African Americans were least likely to have the virus under control, according to the report.
The report also examines data about treatment with PrEP in 2018. About 1.2 million Americans could benefit from PrEP, but only 219,700 (18%) of them had received a prescription for the drug. The eligible groups with the lowest rates of coverage were young people, African Americans, and Latinos.
The report presents a conservative estimate of PrEP coverage, however. Researchers examined data from 92% of prescriptions from retail pharmacies in the United States but did not include prescriptions from closed health care systems such as managed care organizations and military health plans. PrEP coverage in 2018 likely was higher than these estimates indicate, according to the CDC.
“There has been a rapid increase in the number of people taking PrEP over the past 3 years, but there is no doubt that PrEP uptake is too low,” said Eugene McCray, MD, director of CDC’s division of HIV/AIDS prevention. “We are working hard to increase access to PrEP, especially among gay and bisexual men, women, transgender people, young people, African Americans, and Latinos.”
The rate of new HIV infections has not decreased, but remained stable, according to the report. The CDC estimates that there were about 38,000 new infections per year from 2013 to 2017.
Proposed initiative focuses on areas of greatest need
The proposed Ending the HIV Epidemic initiative, if it is funded, will target the locations of greatest need throughout the country. Its initial focus will be on 50 areas that account for more than half of new HIV diagnoses, including 48 counties; San Juan, Puerto Rico; and Washington, D.C. It also will direct resources to seven states with high rates of infection in rural areas. In a second phase, the initiative will expand nationwide, provided that additional resources are made available.
The proposed initiative relies on four science-based strategies. First, it will aim to diagnose all Americans with HIV (at least 95% of HIV infections) as early as possible. Second, the initiative will enable people with HIV to receive treatment rapidly and effectively. The CDC’s target is to achieve viral suppression in at least 95% of people with diagnosed HIV. Third, the initiative will use proven interventions such as PrEP and syringe services programs to prevent new HIV transmissions. One related goal is for at least 50% of people who could benefit from PrEP to receive a prescription. Finally, the initiative is intended to respond quickly to potential HIV outbreaks and provide prevention and treatment to those who need them.
The U.S. Department of Health & Human Services already has taken steps to enable the initiative to be implemented quickly if it is funded in 2020. The department has provided funding to Baltimore City, Md.; DeKalb County, Ga.; and East Baton Rouge Parish, La. to begin pursuing parts of the initiative. These communities are encouraged to share the lessons of their experiences with other communities. HHS also has supported local efforts to develop plans under the initiative in all priority geographic areas. These plans draw upon recommendations from the community, HIV-planning bodies, and health care providers.
“Ending the HIV epidemic would be one of the greatest public health triumphs in our nation’s history,” said Dr. McCray.
SOURCES: Centers for Disease Control and Prevention. CDC Vital Signs. 2019 Dec 3. and Harris NS et al. MMWR Morb Mortal Wkly Rep. 2019 Dec 3.
, according to a Vital Signs report published by the Centers for Disease Control and Prevention based upon a simultaneous MMWR Early Release. The report indicates that many Americans with HIV are not aware of their status or are not receiving effective treatment. Furthermore, the data suggest that few Americans who could benefit from preexposure prophylaxis (PrEP), a daily pill that prevents HIV, are receiving it.

The report “shows that HIV testing, treatment, and prevention have not reached enough Americans, and it emphasizes the continued urgent need to increase these interventions,” said Jay C. Butler, MD, deputy director for infectious diseases at the CDC, at a press conference. “We made a lot of progress in the late ’90s and into the early part of the 21st century in reducing the number of new cases of HIV. But HIV prevention progress has stalled in America since 2013. This stalling underscores the need to increase resources, deploy new technologies, and build expertise, particularly in areas where they’re needed most.”
To achieve these objectives, the CDC has proposed a federal initiative called Ending the HIV Epidemic: A Plan for America. The goal of the initiative is to reduce new HIV infections by 90% by 2030, in part by expanding access to PrEP medications.
Data suggest shortcomings in diagnosis, treatment, and prevention
In its review of data on HIV testing and treatment in 2017, the Vital Signs report found that approximately 154,000 people with HIV (that is, 14% of the total population with HIV) were unaware that they had the virus. These patients consequently could not take advantage of HIV treatment to maintain health, control the virus, and prevent HIV transmission. Young people aged 13-24 years were less likely to know their HIV status than did those aged 25 years and older, according to the report.
Furthermore, approximately two-thirds (63%) of patients who knew that they had HIV had the virus under control through effective treatment. Young people and African Americans were least likely to have the virus under control, according to the report.
The report also examines data about treatment with PrEP in 2018. About 1.2 million Americans could benefit from PrEP, but only 219,700 (18%) of them had received a prescription for the drug. The eligible groups with the lowest rates of coverage were young people, African Americans, and Latinos.
The report presents a conservative estimate of PrEP coverage, however. Researchers examined data from 92% of prescriptions from retail pharmacies in the United States but did not include prescriptions from closed health care systems such as managed care organizations and military health plans. PrEP coverage in 2018 likely was higher than these estimates indicate, according to the CDC.
“There has been a rapid increase in the number of people taking PrEP over the past 3 years, but there is no doubt that PrEP uptake is too low,” said Eugene McCray, MD, director of CDC’s division of HIV/AIDS prevention. “We are working hard to increase access to PrEP, especially among gay and bisexual men, women, transgender people, young people, African Americans, and Latinos.”
The rate of new HIV infections has not decreased, but remained stable, according to the report. The CDC estimates that there were about 38,000 new infections per year from 2013 to 2017.
Proposed initiative focuses on areas of greatest need
The proposed Ending the HIV Epidemic initiative, if it is funded, will target the locations of greatest need throughout the country. Its initial focus will be on 50 areas that account for more than half of new HIV diagnoses, including 48 counties; San Juan, Puerto Rico; and Washington, D.C. It also will direct resources to seven states with high rates of infection in rural areas. In a second phase, the initiative will expand nationwide, provided that additional resources are made available.
The proposed initiative relies on four science-based strategies. First, it will aim to diagnose all Americans with HIV (at least 95% of HIV infections) as early as possible. Second, the initiative will enable people with HIV to receive treatment rapidly and effectively. The CDC’s target is to achieve viral suppression in at least 95% of people with diagnosed HIV. Third, the initiative will use proven interventions such as PrEP and syringe services programs to prevent new HIV transmissions. One related goal is for at least 50% of people who could benefit from PrEP to receive a prescription. Finally, the initiative is intended to respond quickly to potential HIV outbreaks and provide prevention and treatment to those who need them.
The U.S. Department of Health & Human Services already has taken steps to enable the initiative to be implemented quickly if it is funded in 2020. The department has provided funding to Baltimore City, Md.; DeKalb County, Ga.; and East Baton Rouge Parish, La. to begin pursuing parts of the initiative. These communities are encouraged to share the lessons of their experiences with other communities. HHS also has supported local efforts to develop plans under the initiative in all priority geographic areas. These plans draw upon recommendations from the community, HIV-planning bodies, and health care providers.
“Ending the HIV epidemic would be one of the greatest public health triumphs in our nation’s history,” said Dr. McCray.
SOURCES: Centers for Disease Control and Prevention. CDC Vital Signs. 2019 Dec 3. and Harris NS et al. MMWR Morb Mortal Wkly Rep. 2019 Dec 3.
, according to a Vital Signs report published by the Centers for Disease Control and Prevention based upon a simultaneous MMWR Early Release. The report indicates that many Americans with HIV are not aware of their status or are not receiving effective treatment. Furthermore, the data suggest that few Americans who could benefit from preexposure prophylaxis (PrEP), a daily pill that prevents HIV, are receiving it.

The report “shows that HIV testing, treatment, and prevention have not reached enough Americans, and it emphasizes the continued urgent need to increase these interventions,” said Jay C. Butler, MD, deputy director for infectious diseases at the CDC, at a press conference. “We made a lot of progress in the late ’90s and into the early part of the 21st century in reducing the number of new cases of HIV. But HIV prevention progress has stalled in America since 2013. This stalling underscores the need to increase resources, deploy new technologies, and build expertise, particularly in areas where they’re needed most.”
To achieve these objectives, the CDC has proposed a federal initiative called Ending the HIV Epidemic: A Plan for America. The goal of the initiative is to reduce new HIV infections by 90% by 2030, in part by expanding access to PrEP medications.
Data suggest shortcomings in diagnosis, treatment, and prevention
In its review of data on HIV testing and treatment in 2017, the Vital Signs report found that approximately 154,000 people with HIV (that is, 14% of the total population with HIV) were unaware that they had the virus. These patients consequently could not take advantage of HIV treatment to maintain health, control the virus, and prevent HIV transmission. Young people aged 13-24 years were less likely to know their HIV status than did those aged 25 years and older, according to the report.
Furthermore, approximately two-thirds (63%) of patients who knew that they had HIV had the virus under control through effective treatment. Young people and African Americans were least likely to have the virus under control, according to the report.
The report also examines data about treatment with PrEP in 2018. About 1.2 million Americans could benefit from PrEP, but only 219,700 (18%) of them had received a prescription for the drug. The eligible groups with the lowest rates of coverage were young people, African Americans, and Latinos.
The report presents a conservative estimate of PrEP coverage, however. Researchers examined data from 92% of prescriptions from retail pharmacies in the United States but did not include prescriptions from closed health care systems such as managed care organizations and military health plans. PrEP coverage in 2018 likely was higher than these estimates indicate, according to the CDC.
“There has been a rapid increase in the number of people taking PrEP over the past 3 years, but there is no doubt that PrEP uptake is too low,” said Eugene McCray, MD, director of CDC’s division of HIV/AIDS prevention. “We are working hard to increase access to PrEP, especially among gay and bisexual men, women, transgender people, young people, African Americans, and Latinos.”
The rate of new HIV infections has not decreased, but remained stable, according to the report. The CDC estimates that there were about 38,000 new infections per year from 2013 to 2017.
Proposed initiative focuses on areas of greatest need
The proposed Ending the HIV Epidemic initiative, if it is funded, will target the locations of greatest need throughout the country. Its initial focus will be on 50 areas that account for more than half of new HIV diagnoses, including 48 counties; San Juan, Puerto Rico; and Washington, D.C. It also will direct resources to seven states with high rates of infection in rural areas. In a second phase, the initiative will expand nationwide, provided that additional resources are made available.
The proposed initiative relies on four science-based strategies. First, it will aim to diagnose all Americans with HIV (at least 95% of HIV infections) as early as possible. Second, the initiative will enable people with HIV to receive treatment rapidly and effectively. The CDC’s target is to achieve viral suppression in at least 95% of people with diagnosed HIV. Third, the initiative will use proven interventions such as PrEP and syringe services programs to prevent new HIV transmissions. One related goal is for at least 50% of people who could benefit from PrEP to receive a prescription. Finally, the initiative is intended to respond quickly to potential HIV outbreaks and provide prevention and treatment to those who need them.
The U.S. Department of Health & Human Services already has taken steps to enable the initiative to be implemented quickly if it is funded in 2020. The department has provided funding to Baltimore City, Md.; DeKalb County, Ga.; and East Baton Rouge Parish, La. to begin pursuing parts of the initiative. These communities are encouraged to share the lessons of their experiences with other communities. HHS also has supported local efforts to develop plans under the initiative in all priority geographic areas. These plans draw upon recommendations from the community, HIV-planning bodies, and health care providers.
“Ending the HIV epidemic would be one of the greatest public health triumphs in our nation’s history,” said Dr. McCray.
SOURCES: Centers for Disease Control and Prevention. CDC Vital Signs. 2019 Dec 3. and Harris NS et al. MMWR Morb Mortal Wkly Rep. 2019 Dec 3.
FROM THE CDC
Antibiotic use may increase the risk of Parkinson’s disease
according to a report published in Movement Disorders. Associations were found for broad-spectrum antibiotics and those that act against anaerobic bacteria and fungi. The timing of antibiotic exposure also seemed to matter.
In a nationwide case-control study, Finnish researchers compared data on antibiotic use in 13,976 individuals diagnosed with Parkinson’s disease between 1998 and 2014 with antibiotic-use data from 40,697 controls. The strongest connection with Parkinson’s disease risk was found for oral exposure to macrolides and lincosamides (adjusted odds ratio up to 1.416). After correction for multiple comparisons, exposure to antianaerobics and tetracyclines 10-15 years before the index date, and antifungal medications 1-5 years before the index date were positively associated with Parkinson’s disease risk. In post hoc analyses, further positive associations were found for broad-spectrum antibiotics.
Tuomas H. Mertsalmi, MD, from the Helsinki University Hospital and coauthors reported that this was the first study to explore a possible connection between antimicrobial use and Parkinson’s disease.
“In Parkinson’s disease, several studies have described alterations of gut microbiota composition, and changes in fecal microbiota abundance have been found to be associated with gastrointestinal and motor symptoms,” they wrote.
Commenting on the delay between the exposure and diagnosis for the most strongly associated antimicrobials, the authors noted that this 10-15 year lag was comparable with what has been found between the peripheral initiation of Parkinson’s disease and its motor manifestation.
“This would also explain the lack of association between antibiotic exposure 1-5 years before index date – if antibiotic exposure could induce or contribute to the pathogenesis of Parkinson’s disease in the gastrointestinal tract, it would probably take several years before the clinical manifestation of Parkinson’s disease,” they wrote.
With regards to the association seen for sulfonamides and trimethoprim – which was 1-5 years before the index date – they speculated this could reflect treatment for urinary tract infections, which individuals with Parkinson’s disease might be more susceptible to in the prodromal phase of the disease.
The authors noted that infectious disease has also been associated with Parkinson’s disease, and that their analysis did not include information about why the antimicrobial agents were prescribed. However, they pointed out that the associations were only for certain antibiotic classes, which makes it unlikely that the association was related to greater burden of infectious disease among individuals with Parkinson’s disease.
The pattern of associations supports the hypothesis that effects on gut microbiota could link antibiotics to Parkinson’s disease. “The link between antibiotic exposure and Parkinson’s disease fits the current view that in a significant proportion of patients the pathology of Parkinson’s disease may originate in the gut, possibly related to microbial changes, years before the onset of typical Parkinson’s disease motor symptoms such as slowness, muscle stiffness, and shaking of the extremities. It was known that bacterial composition of the intestine in patients with Parkinson’s disease is abnormal, but the cause is unclear. Our results suggest that some commonly used antibiotics, which are known to strongly influence the gut microbiota, could be a predisposing factor,” said lead investigator Filip Scheperjans, MD, PhD, from the department of neurology at Helsinki University Hospital.
The findings may have implications for antibiotic prescribing practices in the future, said Dr. Scheperjans. “In addition to the problem of antibiotic resistance, antimicrobial prescribing should also take into account their potentially long-lasting effects on the gut microbiome and the development of certain diseases.”
The study was funded by the Finnish Parkinson Foundation, the Finnish Medical Foundation, the Maire Taponen Foundation, and the Academy of Finland. One author declared relevant patents and his position as founder and chief executive of a private company. No other conflicts of interest were declared.
SOURCE: Mertsalmi TH et al. Mov Disord. 2019 Nov 18. doi: 10.1002/mds.27924.
according to a report published in Movement Disorders. Associations were found for broad-spectrum antibiotics and those that act against anaerobic bacteria and fungi. The timing of antibiotic exposure also seemed to matter.
In a nationwide case-control study, Finnish researchers compared data on antibiotic use in 13,976 individuals diagnosed with Parkinson’s disease between 1998 and 2014 with antibiotic-use data from 40,697 controls. The strongest connection with Parkinson’s disease risk was found for oral exposure to macrolides and lincosamides (adjusted odds ratio up to 1.416). After correction for multiple comparisons, exposure to antianaerobics and tetracyclines 10-15 years before the index date, and antifungal medications 1-5 years before the index date were positively associated with Parkinson’s disease risk. In post hoc analyses, further positive associations were found for broad-spectrum antibiotics.
Tuomas H. Mertsalmi, MD, from the Helsinki University Hospital and coauthors reported that this was the first study to explore a possible connection between antimicrobial use and Parkinson’s disease.
“In Parkinson’s disease, several studies have described alterations of gut microbiota composition, and changes in fecal microbiota abundance have been found to be associated with gastrointestinal and motor symptoms,” they wrote.
Commenting on the delay between the exposure and diagnosis for the most strongly associated antimicrobials, the authors noted that this 10-15 year lag was comparable with what has been found between the peripheral initiation of Parkinson’s disease and its motor manifestation.
“This would also explain the lack of association between antibiotic exposure 1-5 years before index date – if antibiotic exposure could induce or contribute to the pathogenesis of Parkinson’s disease in the gastrointestinal tract, it would probably take several years before the clinical manifestation of Parkinson’s disease,” they wrote.
With regards to the association seen for sulfonamides and trimethoprim – which was 1-5 years before the index date – they speculated this could reflect treatment for urinary tract infections, which individuals with Parkinson’s disease might be more susceptible to in the prodromal phase of the disease.
The authors noted that infectious disease has also been associated with Parkinson’s disease, and that their analysis did not include information about why the antimicrobial agents were prescribed. However, they pointed out that the associations were only for certain antibiotic classes, which makes it unlikely that the association was related to greater burden of infectious disease among individuals with Parkinson’s disease.
The pattern of associations supports the hypothesis that effects on gut microbiota could link antibiotics to Parkinson’s disease. “The link between antibiotic exposure and Parkinson’s disease fits the current view that in a significant proportion of patients the pathology of Parkinson’s disease may originate in the gut, possibly related to microbial changes, years before the onset of typical Parkinson’s disease motor symptoms such as slowness, muscle stiffness, and shaking of the extremities. It was known that bacterial composition of the intestine in patients with Parkinson’s disease is abnormal, but the cause is unclear. Our results suggest that some commonly used antibiotics, which are known to strongly influence the gut microbiota, could be a predisposing factor,” said lead investigator Filip Scheperjans, MD, PhD, from the department of neurology at Helsinki University Hospital.
The findings may have implications for antibiotic prescribing practices in the future, said Dr. Scheperjans. “In addition to the problem of antibiotic resistance, antimicrobial prescribing should also take into account their potentially long-lasting effects on the gut microbiome and the development of certain diseases.”
The study was funded by the Finnish Parkinson Foundation, the Finnish Medical Foundation, the Maire Taponen Foundation, and the Academy of Finland. One author declared relevant patents and his position as founder and chief executive of a private company. No other conflicts of interest were declared.
SOURCE: Mertsalmi TH et al. Mov Disord. 2019 Nov 18. doi: 10.1002/mds.27924.
according to a report published in Movement Disorders. Associations were found for broad-spectrum antibiotics and those that act against anaerobic bacteria and fungi. The timing of antibiotic exposure also seemed to matter.
In a nationwide case-control study, Finnish researchers compared data on antibiotic use in 13,976 individuals diagnosed with Parkinson’s disease between 1998 and 2014 with antibiotic-use data from 40,697 controls. The strongest connection with Parkinson’s disease risk was found for oral exposure to macrolides and lincosamides (adjusted odds ratio up to 1.416). After correction for multiple comparisons, exposure to antianaerobics and tetracyclines 10-15 years before the index date, and antifungal medications 1-5 years before the index date were positively associated with Parkinson’s disease risk. In post hoc analyses, further positive associations were found for broad-spectrum antibiotics.
Tuomas H. Mertsalmi, MD, from the Helsinki University Hospital and coauthors reported that this was the first study to explore a possible connection between antimicrobial use and Parkinson’s disease.
“In Parkinson’s disease, several studies have described alterations of gut microbiota composition, and changes in fecal microbiota abundance have been found to be associated with gastrointestinal and motor symptoms,” they wrote.
Commenting on the delay between the exposure and diagnosis for the most strongly associated antimicrobials, the authors noted that this 10-15 year lag was comparable with what has been found between the peripheral initiation of Parkinson’s disease and its motor manifestation.
“This would also explain the lack of association between antibiotic exposure 1-5 years before index date – if antibiotic exposure could induce or contribute to the pathogenesis of Parkinson’s disease in the gastrointestinal tract, it would probably take several years before the clinical manifestation of Parkinson’s disease,” they wrote.
With regards to the association seen for sulfonamides and trimethoprim – which was 1-5 years before the index date – they speculated this could reflect treatment for urinary tract infections, which individuals with Parkinson’s disease might be more susceptible to in the prodromal phase of the disease.
The authors noted that infectious disease has also been associated with Parkinson’s disease, and that their analysis did not include information about why the antimicrobial agents were prescribed. However, they pointed out that the associations were only for certain antibiotic classes, which makes it unlikely that the association was related to greater burden of infectious disease among individuals with Parkinson’s disease.
The pattern of associations supports the hypothesis that effects on gut microbiota could link antibiotics to Parkinson’s disease. “The link between antibiotic exposure and Parkinson’s disease fits the current view that in a significant proportion of patients the pathology of Parkinson’s disease may originate in the gut, possibly related to microbial changes, years before the onset of typical Parkinson’s disease motor symptoms such as slowness, muscle stiffness, and shaking of the extremities. It was known that bacterial composition of the intestine in patients with Parkinson’s disease is abnormal, but the cause is unclear. Our results suggest that some commonly used antibiotics, which are known to strongly influence the gut microbiota, could be a predisposing factor,” said lead investigator Filip Scheperjans, MD, PhD, from the department of neurology at Helsinki University Hospital.
The findings may have implications for antibiotic prescribing practices in the future, said Dr. Scheperjans. “In addition to the problem of antibiotic resistance, antimicrobial prescribing should also take into account their potentially long-lasting effects on the gut microbiome and the development of certain diseases.”
The study was funded by the Finnish Parkinson Foundation, the Finnish Medical Foundation, the Maire Taponen Foundation, and the Academy of Finland. One author declared relevant patents and his position as founder and chief executive of a private company. No other conflicts of interest were declared.
SOURCE: Mertsalmi TH et al. Mov Disord. 2019 Nov 18. doi: 10.1002/mds.27924.
FROM MOVEMENT DISORDERS
2019-2020 flu season starts off full throttle
For the week ending Nov. 23, there were five states, along with Puerto Rico, at the highest level of the Centers for Disease Control and Prevention’s 1-10 scale of flu activity. That’s more than any year since 2012, including the pandemic season of 2017-2018, according to CDC data, and may suggest either an early peak or the beginning of a particularly bad winter.
“Nationally, ILI [influenza-like illness] activity has been at or above baseline for 3 weeks; however, the amount of influenza activity across the country varies with the south and parts of the west seeing elevated activity while other parts of the country are still seeing low activity,” the CDC’s influenza division said in its weekly FluView report.
The five highest-activity states – Alabama, Georgia, Louisiana, Mississippi, and Texas – are all at level 10, and they join two others – South Carolina and Tennessee, which are at level 8 – in the “high” range from 8-10 on the ILI activity scale; Puerto Rico also is at level 10. ILI is defined as “fever (temperature of 100° F [37.8° C] or greater) and a cough and/or a sore throat without a known cause other than influenza,” the CDC said.
The activity scale is based on the percentage of outpatient visits for ILI in each state, which is reported to the CDC’s Outpatient Influenza-like Illness Surveillance Network (ILINet) each week. The national rate for the week ending Nov. 23 was 2.9%, which is above the new-for-this-season baseline rate of 2.4%. For the three previous flu seasons, the national baseline was 2.2%, having been raised from its previous level of 2.1% in 2015-2016, CDC data show.
The peak month of flu activity occurs most often in February – 15 times from 1982-1983 to 2017-2018 – but there were seven peaks in December and six each in January and March over that time period, along with one peak each in October and November, the CDC said. The October peak occurred during the H1N1 pandemic year of 2009, when the national outpatient ILI rate climbed to just over 7.7%.
For the week ending Nov. 23, there were five states, along with Puerto Rico, at the highest level of the Centers for Disease Control and Prevention’s 1-10 scale of flu activity. That’s more than any year since 2012, including the pandemic season of 2017-2018, according to CDC data, and may suggest either an early peak or the beginning of a particularly bad winter.
“Nationally, ILI [influenza-like illness] activity has been at or above baseline for 3 weeks; however, the amount of influenza activity across the country varies with the south and parts of the west seeing elevated activity while other parts of the country are still seeing low activity,” the CDC’s influenza division said in its weekly FluView report.
The five highest-activity states – Alabama, Georgia, Louisiana, Mississippi, and Texas – are all at level 10, and they join two others – South Carolina and Tennessee, which are at level 8 – in the “high” range from 8-10 on the ILI activity scale; Puerto Rico also is at level 10. ILI is defined as “fever (temperature of 100° F [37.8° C] or greater) and a cough and/or a sore throat without a known cause other than influenza,” the CDC said.
The activity scale is based on the percentage of outpatient visits for ILI in each state, which is reported to the CDC’s Outpatient Influenza-like Illness Surveillance Network (ILINet) each week. The national rate for the week ending Nov. 23 was 2.9%, which is above the new-for-this-season baseline rate of 2.4%. For the three previous flu seasons, the national baseline was 2.2%, having been raised from its previous level of 2.1% in 2015-2016, CDC data show.
The peak month of flu activity occurs most often in February – 15 times from 1982-1983 to 2017-2018 – but there were seven peaks in December and six each in January and March over that time period, along with one peak each in October and November, the CDC said. The October peak occurred during the H1N1 pandemic year of 2009, when the national outpatient ILI rate climbed to just over 7.7%.
For the week ending Nov. 23, there were five states, along with Puerto Rico, at the highest level of the Centers for Disease Control and Prevention’s 1-10 scale of flu activity. That’s more than any year since 2012, including the pandemic season of 2017-2018, according to CDC data, and may suggest either an early peak or the beginning of a particularly bad winter.
“Nationally, ILI [influenza-like illness] activity has been at or above baseline for 3 weeks; however, the amount of influenza activity across the country varies with the south and parts of the west seeing elevated activity while other parts of the country are still seeing low activity,” the CDC’s influenza division said in its weekly FluView report.
The five highest-activity states – Alabama, Georgia, Louisiana, Mississippi, and Texas – are all at level 10, and they join two others – South Carolina and Tennessee, which are at level 8 – in the “high” range from 8-10 on the ILI activity scale; Puerto Rico also is at level 10. ILI is defined as “fever (temperature of 100° F [37.8° C] or greater) and a cough and/or a sore throat without a known cause other than influenza,” the CDC said.
The activity scale is based on the percentage of outpatient visits for ILI in each state, which is reported to the CDC’s Outpatient Influenza-like Illness Surveillance Network (ILINet) each week. The national rate for the week ending Nov. 23 was 2.9%, which is above the new-for-this-season baseline rate of 2.4%. For the three previous flu seasons, the national baseline was 2.2%, having been raised from its previous level of 2.1% in 2015-2016, CDC data show.
The peak month of flu activity occurs most often in February – 15 times from 1982-1983 to 2017-2018 – but there were seven peaks in December and six each in January and March over that time period, along with one peak each in October and November, the CDC said. The October peak occurred during the H1N1 pandemic year of 2009, when the national outpatient ILI rate climbed to just over 7.7%.
Simple prevention strategies can lessen postoperative delirium after orthopedic surgery
A new study has found that and a prevention program can help improve staff education and outcomes.
“In an aging society, it is very important to develop and implement a strategy for POD prevention to ensure that aging patients are treated as safely and effectively as possible,” wrote Jung-Yeon Choi of Seoul (South Korea) National University Bundang Hospital and coauthors. The study was published in BMC Geriatrics.
To determine how to better identify and treat high-risk patients for POD after orthopedic surgery, the researchers led a retrospective cohort study that included an intervention group of participants who were aged at least 65 years (n = 275) and a control group from a year prior (n = 274). Patients in the intervention group had their risk of delirium assessed and categorized using a simple screening tool, and those deemed at risk were entered into a multicomponent delirium prevention program.
Of the 275 patients in the intervention group, 144 required screening for delirium. Ninety-nine were classified as low risk, 29 were classified as high risk, and 16 missed the screening. Fifty-three additional patients were classified as high risk because they were aged 80 years or older. During the study, 17 participants experienced POD, 16 of whom were classified as high risk. In regard to estimating POD risk, the sensitivity and specificity of the delirium screening tool were 94.1% and 72.7%, respectively. Incidence rates of POD were 10.2% in the control group and 6.2% in the intervention group.
The authors noted their study’s limitations, including its design as a retrospective review of medical records rather than a prospective randomized controlled trial. In addition, because it was conducted in just one teaching hospital, they deemed it “not possible to determine the generalizability and long-term effect of our findings.”
The authors reported no conflicts of interest.
SOURCE: Choi JY et al. BMC Geriatr. 2019 Oct 26. doi: 10.1186/s12877-019-1303-z.
A new study has found that and a prevention program can help improve staff education and outcomes.
“In an aging society, it is very important to develop and implement a strategy for POD prevention to ensure that aging patients are treated as safely and effectively as possible,” wrote Jung-Yeon Choi of Seoul (South Korea) National University Bundang Hospital and coauthors. The study was published in BMC Geriatrics.
To determine how to better identify and treat high-risk patients for POD after orthopedic surgery, the researchers led a retrospective cohort study that included an intervention group of participants who were aged at least 65 years (n = 275) and a control group from a year prior (n = 274). Patients in the intervention group had their risk of delirium assessed and categorized using a simple screening tool, and those deemed at risk were entered into a multicomponent delirium prevention program.
Of the 275 patients in the intervention group, 144 required screening for delirium. Ninety-nine were classified as low risk, 29 were classified as high risk, and 16 missed the screening. Fifty-three additional patients were classified as high risk because they were aged 80 years or older. During the study, 17 participants experienced POD, 16 of whom were classified as high risk. In regard to estimating POD risk, the sensitivity and specificity of the delirium screening tool were 94.1% and 72.7%, respectively. Incidence rates of POD were 10.2% in the control group and 6.2% in the intervention group.
The authors noted their study’s limitations, including its design as a retrospective review of medical records rather than a prospective randomized controlled trial. In addition, because it was conducted in just one teaching hospital, they deemed it “not possible to determine the generalizability and long-term effect of our findings.”
The authors reported no conflicts of interest.
SOURCE: Choi JY et al. BMC Geriatr. 2019 Oct 26. doi: 10.1186/s12877-019-1303-z.
A new study has found that and a prevention program can help improve staff education and outcomes.
“In an aging society, it is very important to develop and implement a strategy for POD prevention to ensure that aging patients are treated as safely and effectively as possible,” wrote Jung-Yeon Choi of Seoul (South Korea) National University Bundang Hospital and coauthors. The study was published in BMC Geriatrics.
To determine how to better identify and treat high-risk patients for POD after orthopedic surgery, the researchers led a retrospective cohort study that included an intervention group of participants who were aged at least 65 years (n = 275) and a control group from a year prior (n = 274). Patients in the intervention group had their risk of delirium assessed and categorized using a simple screening tool, and those deemed at risk were entered into a multicomponent delirium prevention program.
Of the 275 patients in the intervention group, 144 required screening for delirium. Ninety-nine were classified as low risk, 29 were classified as high risk, and 16 missed the screening. Fifty-three additional patients were classified as high risk because they were aged 80 years or older. During the study, 17 participants experienced POD, 16 of whom were classified as high risk. In regard to estimating POD risk, the sensitivity and specificity of the delirium screening tool were 94.1% and 72.7%, respectively. Incidence rates of POD were 10.2% in the control group and 6.2% in the intervention group.
The authors noted their study’s limitations, including its design as a retrospective review of medical records rather than a prospective randomized controlled trial. In addition, because it was conducted in just one teaching hospital, they deemed it “not possible to determine the generalizability and long-term effect of our findings.”
The authors reported no conflicts of interest.
SOURCE: Choi JY et al. BMC Geriatr. 2019 Oct 26. doi: 10.1186/s12877-019-1303-z.
FROM BMC GERIATRICS
Nearly one in five U.S. adolescents have prediabetes
with a higher prevalence among males, a study has found.
Linda J. Andes, PhD, from the Centers for Disease Control and Prevention and coauthors reported in JAMA Pediatrics their analysis of data from 2,606 adolescent (12-18 years) and 3,180 young adult (19-34 years) participants in the 2005-2016 National Health and Nutrition Examination Surveys.
This found that the percentage with prediabetes – defined as either impaired fasting glucose (IFG), impaired glucose tolerance (IGT) or increased hemoglobin A1c (HbA1c) level – was 18% among adolescents and 24% among young adults.
The most common condition was IFG, which was seen in 11% of adolescents and 16% of young adults. The rate of IGT was 4% in adolescents and 6% of young adults, while elevated HbA1c levels were seen in 5% of adolescents and 8% of young adults.
This information is important because “In adults, these three phenotypes increase the risk of developing type 2 diabetes as well as cardiovascular diseases,” Dr. Andes and coauthors wrote. “In 2011-2012, the overall prevalence of prediabetes among U.S. adults, defined as the presence of any of the three glucose metabolism dysregulation phenotypes, was 38% and it increased to about 50% in persons 65 years and older.”
Dr. Andes and associates noted that isolated IFG was the most common glucose dysregulation seen in both adolescents and young adults. “While individuals with IFG are at increased risk for type 2 diabetes, few primary prevention trials have included individuals selected for the presence of IFG and none have been conducted in adolescents with IFG or IGT to our knowledge.”
The study saw some key gender differences in prevalence. For example, the prevalence of IFG was significantly lower in adolescent girls than in boys (7% vs. 15%; P less than .001), and in young women, compared with young men (10% vs. 22%; P less than .001).
“These findings are consistent with those of other studies in adults; however, the underlying mechanisms for explaining this discrepancy are still unclear,” Dr. Andes and coauthors wrote.
Ethnicity also appeared to influence risk, with the prevalence of IFG significantly lower in non-Hispanic black adolescents, compared with Hispanic adolescents. However, increased HbA1c levels were significantly more prevalent in non-Hispanic black adolescents, compared with Hispanic or non-Hispanic white adolescents.
“These findings highlight the need for additional studies on the long-term consequences and preventive strategies of abnormal glucose metabolism as measured by HbA1c levels in adolescents and young adults, especially of minority racial/ethnic groups,” the authors wrote.
Adolescents with prediabetes had significantly higher systolic blood pressure, non-HDL cholesterol, waist-to-height ratio, higher body mass index, and lower insulin sensitivity, compared with those with normal glucose tolerance. Among young adults with prediabetes, there was significantly higher systolic blood pressure and non-HDL cholesterol, compared with individuals with normal glucose tolerance.
No funding or conflicts of interest were declared.
SOURCE: Andes LJ et al. JAMA Pediatr. 2019 Dec 2. doi: 10.1001/jamapediatrics.2019.4498.
with a higher prevalence among males, a study has found.
Linda J. Andes, PhD, from the Centers for Disease Control and Prevention and coauthors reported in JAMA Pediatrics their analysis of data from 2,606 adolescent (12-18 years) and 3,180 young adult (19-34 years) participants in the 2005-2016 National Health and Nutrition Examination Surveys.
This found that the percentage with prediabetes – defined as either impaired fasting glucose (IFG), impaired glucose tolerance (IGT) or increased hemoglobin A1c (HbA1c) level – was 18% among adolescents and 24% among young adults.
The most common condition was IFG, which was seen in 11% of adolescents and 16% of young adults. The rate of IGT was 4% in adolescents and 6% of young adults, while elevated HbA1c levels were seen in 5% of adolescents and 8% of young adults.
This information is important because “In adults, these three phenotypes increase the risk of developing type 2 diabetes as well as cardiovascular diseases,” Dr. Andes and coauthors wrote. “In 2011-2012, the overall prevalence of prediabetes among U.S. adults, defined as the presence of any of the three glucose metabolism dysregulation phenotypes, was 38% and it increased to about 50% in persons 65 years and older.”
Dr. Andes and associates noted that isolated IFG was the most common glucose dysregulation seen in both adolescents and young adults. “While individuals with IFG are at increased risk for type 2 diabetes, few primary prevention trials have included individuals selected for the presence of IFG and none have been conducted in adolescents with IFG or IGT to our knowledge.”
The study saw some key gender differences in prevalence. For example, the prevalence of IFG was significantly lower in adolescent girls than in boys (7% vs. 15%; P less than .001), and in young women, compared with young men (10% vs. 22%; P less than .001).
“These findings are consistent with those of other studies in adults; however, the underlying mechanisms for explaining this discrepancy are still unclear,” Dr. Andes and coauthors wrote.
Ethnicity also appeared to influence risk, with the prevalence of IFG significantly lower in non-Hispanic black adolescents, compared with Hispanic adolescents. However, increased HbA1c levels were significantly more prevalent in non-Hispanic black adolescents, compared with Hispanic or non-Hispanic white adolescents.
“These findings highlight the need for additional studies on the long-term consequences and preventive strategies of abnormal glucose metabolism as measured by HbA1c levels in adolescents and young adults, especially of minority racial/ethnic groups,” the authors wrote.
Adolescents with prediabetes had significantly higher systolic blood pressure, non-HDL cholesterol, waist-to-height ratio, higher body mass index, and lower insulin sensitivity, compared with those with normal glucose tolerance. Among young adults with prediabetes, there was significantly higher systolic blood pressure and non-HDL cholesterol, compared with individuals with normal glucose tolerance.
No funding or conflicts of interest were declared.
SOURCE: Andes LJ et al. JAMA Pediatr. 2019 Dec 2. doi: 10.1001/jamapediatrics.2019.4498.
with a higher prevalence among males, a study has found.
Linda J. Andes, PhD, from the Centers for Disease Control and Prevention and coauthors reported in JAMA Pediatrics their analysis of data from 2,606 adolescent (12-18 years) and 3,180 young adult (19-34 years) participants in the 2005-2016 National Health and Nutrition Examination Surveys.
This found that the percentage with prediabetes – defined as either impaired fasting glucose (IFG), impaired glucose tolerance (IGT) or increased hemoglobin A1c (HbA1c) level – was 18% among adolescents and 24% among young adults.
The most common condition was IFG, which was seen in 11% of adolescents and 16% of young adults. The rate of IGT was 4% in adolescents and 6% of young adults, while elevated HbA1c levels were seen in 5% of adolescents and 8% of young adults.
This information is important because “In adults, these three phenotypes increase the risk of developing type 2 diabetes as well as cardiovascular diseases,” Dr. Andes and coauthors wrote. “In 2011-2012, the overall prevalence of prediabetes among U.S. adults, defined as the presence of any of the three glucose metabolism dysregulation phenotypes, was 38% and it increased to about 50% in persons 65 years and older.”
Dr. Andes and associates noted that isolated IFG was the most common glucose dysregulation seen in both adolescents and young adults. “While individuals with IFG are at increased risk for type 2 diabetes, few primary prevention trials have included individuals selected for the presence of IFG and none have been conducted in adolescents with IFG or IGT to our knowledge.”
The study saw some key gender differences in prevalence. For example, the prevalence of IFG was significantly lower in adolescent girls than in boys (7% vs. 15%; P less than .001), and in young women, compared with young men (10% vs. 22%; P less than .001).
“These findings are consistent with those of other studies in adults; however, the underlying mechanisms for explaining this discrepancy are still unclear,” Dr. Andes and coauthors wrote.
Ethnicity also appeared to influence risk, with the prevalence of IFG significantly lower in non-Hispanic black adolescents, compared with Hispanic adolescents. However, increased HbA1c levels were significantly more prevalent in non-Hispanic black adolescents, compared with Hispanic or non-Hispanic white adolescents.
“These findings highlight the need for additional studies on the long-term consequences and preventive strategies of abnormal glucose metabolism as measured by HbA1c levels in adolescents and young adults, especially of minority racial/ethnic groups,” the authors wrote.
Adolescents with prediabetes had significantly higher systolic blood pressure, non-HDL cholesterol, waist-to-height ratio, higher body mass index, and lower insulin sensitivity, compared with those with normal glucose tolerance. Among young adults with prediabetes, there was significantly higher systolic blood pressure and non-HDL cholesterol, compared with individuals with normal glucose tolerance.
No funding or conflicts of interest were declared.
SOURCE: Andes LJ et al. JAMA Pediatr. 2019 Dec 2. doi: 10.1001/jamapediatrics.2019.4498.
FROM JAMA PEDIATRICS