User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
High complete response rate seen with novel CAR-T for myeloma
ORLANDO – A novel chimeric antigen receptor T (CAR T) cell construct is associated with deep clinical responses in patients with multiple myeloma for whom prior lines of therapy – some numbering in the double digits – have failed.
Among 29 patients with multiple myeloma enrolled in a phase 1b/2 trial of JNJ-4528, the overall response rate (ORR) at 6 months median follow-up was 100%, including 69% complete responses, with 27 patients remaining free of disease progression at a median of 6 months, reported Deepu Madduri, MD, of Icahn School of Medicine at Mount Sinai, New York.
“These are very heavily pretreated patients, and so getting early and deep responses is quite amazing,” she said at a briefing prior to presentation of the data at the annual meeting of the American Society of Hematology.
JNJ-4528 is a second-generation CAR T containing two single-domain antibodies targeted against B-cell maturation protein (BCMA). As previously reported, an identical CAR T cell construct showed a high overall response with manageable toxicities in 74 patients with relapsed/refractory multiple myeloma. JNJ-4528 was granted a breakthrough therapy designation for relapsed/refractory multiple myeloma by the Food and Drug Administration on Dec. 6, 2019, and a priority medicines (PRIME) designation by the European Medicines Agency in April 2019.
BCMA was first described in myeloma in 2004 as a mechanism for the growth and survival of malignant plasma cells. Several research groups are currently investigating CAR T cells or monoclonal antibodies targeted to BCMA. The product closest to receiving FDA approval is likely BB2121.
At ASH 2019, Dr. Madduri presented results from the phase 1b portion of the CARTITUDE-1 trial. The investigators enrolled patients with multiple myeloma with measurable diseases as assessed by M-protein or serum free light chain levels who had experienced disease progression on at least 3 prior lines of therapy, or whose disease was refractory to at least two lines of therapy with a proteasome inhibitor, immunomodulatory drug (IMiD), and an anti-CD38 antibody.
Patients underwent apheresis for T-cell collection, with bridging therapy allowed until the expanded T cells could be delivered.
Following T-cell depletion with cyclophosphamide 300 mg/m2 and fludarabine 30 mg/m2 over 3 days, patients received a single weight-based infusion (compared with fixed-dose infusions used with other CAR T cell constructs).
The dose was targeted at 0.75x106 CAR-positive cells/kg, with a target range of 0.5–1.0x106, administered 5-7 days after the start of the conditioning regimen.
A total of 29 patients, median age 60, were evaluable for the safety and efficacy endpoints. One-fourth of the patients had a high-risk cytogenetic profile. The patients had received a median of 5 prior lines of therapy, with one patient receiving 18 prior lines. Of the 29 patients, 25 (86%) had previously undergone autologous transplantation.
As noted before, the ORR after a median follow-up of 6 months was 100%, with 69% completer responses, 17% very good partial responses, and 14% partial responses. The median time to complete response was 1 month (range 1 to 9 months). All but two patients remained free of disease progression at the median 6-month follow-up.
Nearly all patients (27) developed cytokine release syndrome (CRS), and one patient with prolonged grade 4 CRS died from related complications 99 days after infusion.
The median time to onset of CRS was 7 days with more than 90% of cases occurring between days 5 and 9.
Neurotoxicities, specifically immune effector cell–associated neurotoxicity syndrome (ICANS), were infrequent in CRS, and when they did occur were generally low grade, with only 1 grade 3 ICANS event.
Asked in an interview whether the impressive response rates seen with JNJ-4528 might persist over time, Dr. Madduri acknowledged that follow-up is still relatively short.
“This product is unique in that has a CD8 central memory phenotype preferentially, and we’re hoping that this would play a central role in the durability of response because they’re memory cells, but I think at this time we don’t know,” she said.
The CARTITUDE-1 trial is funded by Janssen Research & Development. Dr. Madduri disclosed serving as a consultant to Janssen and to Takeda, Foundation Medicine, AbbVie, and Celgene.
SOURCE: Madduri D et al. ASH 2019. Abstract 577.
ORLANDO – A novel chimeric antigen receptor T (CAR T) cell construct is associated with deep clinical responses in patients with multiple myeloma for whom prior lines of therapy – some numbering in the double digits – have failed.
Among 29 patients with multiple myeloma enrolled in a phase 1b/2 trial of JNJ-4528, the overall response rate (ORR) at 6 months median follow-up was 100%, including 69% complete responses, with 27 patients remaining free of disease progression at a median of 6 months, reported Deepu Madduri, MD, of Icahn School of Medicine at Mount Sinai, New York.
“These are very heavily pretreated patients, and so getting early and deep responses is quite amazing,” she said at a briefing prior to presentation of the data at the annual meeting of the American Society of Hematology.
JNJ-4528 is a second-generation CAR T containing two single-domain antibodies targeted against B-cell maturation protein (BCMA). As previously reported, an identical CAR T cell construct showed a high overall response with manageable toxicities in 74 patients with relapsed/refractory multiple myeloma. JNJ-4528 was granted a breakthrough therapy designation for relapsed/refractory multiple myeloma by the Food and Drug Administration on Dec. 6, 2019, and a priority medicines (PRIME) designation by the European Medicines Agency in April 2019.
BCMA was first described in myeloma in 2004 as a mechanism for the growth and survival of malignant plasma cells. Several research groups are currently investigating CAR T cells or monoclonal antibodies targeted to BCMA. The product closest to receiving FDA approval is likely BB2121.
At ASH 2019, Dr. Madduri presented results from the phase 1b portion of the CARTITUDE-1 trial. The investigators enrolled patients with multiple myeloma with measurable diseases as assessed by M-protein or serum free light chain levels who had experienced disease progression on at least 3 prior lines of therapy, or whose disease was refractory to at least two lines of therapy with a proteasome inhibitor, immunomodulatory drug (IMiD), and an anti-CD38 antibody.
Patients underwent apheresis for T-cell collection, with bridging therapy allowed until the expanded T cells could be delivered.
Following T-cell depletion with cyclophosphamide 300 mg/m2 and fludarabine 30 mg/m2 over 3 days, patients received a single weight-based infusion (compared with fixed-dose infusions used with other CAR T cell constructs).
The dose was targeted at 0.75x106 CAR-positive cells/kg, with a target range of 0.5–1.0x106, administered 5-7 days after the start of the conditioning regimen.
A total of 29 patients, median age 60, were evaluable for the safety and efficacy endpoints. One-fourth of the patients had a high-risk cytogenetic profile. The patients had received a median of 5 prior lines of therapy, with one patient receiving 18 prior lines. Of the 29 patients, 25 (86%) had previously undergone autologous transplantation.
As noted before, the ORR after a median follow-up of 6 months was 100%, with 69% completer responses, 17% very good partial responses, and 14% partial responses. The median time to complete response was 1 month (range 1 to 9 months). All but two patients remained free of disease progression at the median 6-month follow-up.
Nearly all patients (27) developed cytokine release syndrome (CRS), and one patient with prolonged grade 4 CRS died from related complications 99 days after infusion.
The median time to onset of CRS was 7 days with more than 90% of cases occurring between days 5 and 9.
Neurotoxicities, specifically immune effector cell–associated neurotoxicity syndrome (ICANS), were infrequent in CRS, and when they did occur were generally low grade, with only 1 grade 3 ICANS event.
Asked in an interview whether the impressive response rates seen with JNJ-4528 might persist over time, Dr. Madduri acknowledged that follow-up is still relatively short.
“This product is unique in that has a CD8 central memory phenotype preferentially, and we’re hoping that this would play a central role in the durability of response because they’re memory cells, but I think at this time we don’t know,” she said.
The CARTITUDE-1 trial is funded by Janssen Research & Development. Dr. Madduri disclosed serving as a consultant to Janssen and to Takeda, Foundation Medicine, AbbVie, and Celgene.
SOURCE: Madduri D et al. ASH 2019. Abstract 577.
ORLANDO – A novel chimeric antigen receptor T (CAR T) cell construct is associated with deep clinical responses in patients with multiple myeloma for whom prior lines of therapy – some numbering in the double digits – have failed.
Among 29 patients with multiple myeloma enrolled in a phase 1b/2 trial of JNJ-4528, the overall response rate (ORR) at 6 months median follow-up was 100%, including 69% complete responses, with 27 patients remaining free of disease progression at a median of 6 months, reported Deepu Madduri, MD, of Icahn School of Medicine at Mount Sinai, New York.
“These are very heavily pretreated patients, and so getting early and deep responses is quite amazing,” she said at a briefing prior to presentation of the data at the annual meeting of the American Society of Hematology.
JNJ-4528 is a second-generation CAR T containing two single-domain antibodies targeted against B-cell maturation protein (BCMA). As previously reported, an identical CAR T cell construct showed a high overall response with manageable toxicities in 74 patients with relapsed/refractory multiple myeloma. JNJ-4528 was granted a breakthrough therapy designation for relapsed/refractory multiple myeloma by the Food and Drug Administration on Dec. 6, 2019, and a priority medicines (PRIME) designation by the European Medicines Agency in April 2019.
BCMA was first described in myeloma in 2004 as a mechanism for the growth and survival of malignant plasma cells. Several research groups are currently investigating CAR T cells or monoclonal antibodies targeted to BCMA. The product closest to receiving FDA approval is likely BB2121.
At ASH 2019, Dr. Madduri presented results from the phase 1b portion of the CARTITUDE-1 trial. The investigators enrolled patients with multiple myeloma with measurable diseases as assessed by M-protein or serum free light chain levels who had experienced disease progression on at least 3 prior lines of therapy, or whose disease was refractory to at least two lines of therapy with a proteasome inhibitor, immunomodulatory drug (IMiD), and an anti-CD38 antibody.
Patients underwent apheresis for T-cell collection, with bridging therapy allowed until the expanded T cells could be delivered.
Following T-cell depletion with cyclophosphamide 300 mg/m2 and fludarabine 30 mg/m2 over 3 days, patients received a single weight-based infusion (compared with fixed-dose infusions used with other CAR T cell constructs).
The dose was targeted at 0.75x106 CAR-positive cells/kg, with a target range of 0.5–1.0x106, administered 5-7 days after the start of the conditioning regimen.
A total of 29 patients, median age 60, were evaluable for the safety and efficacy endpoints. One-fourth of the patients had a high-risk cytogenetic profile. The patients had received a median of 5 prior lines of therapy, with one patient receiving 18 prior lines. Of the 29 patients, 25 (86%) had previously undergone autologous transplantation.
As noted before, the ORR after a median follow-up of 6 months was 100%, with 69% completer responses, 17% very good partial responses, and 14% partial responses. The median time to complete response was 1 month (range 1 to 9 months). All but two patients remained free of disease progression at the median 6-month follow-up.
Nearly all patients (27) developed cytokine release syndrome (CRS), and one patient with prolonged grade 4 CRS died from related complications 99 days after infusion.
The median time to onset of CRS was 7 days with more than 90% of cases occurring between days 5 and 9.
Neurotoxicities, specifically immune effector cell–associated neurotoxicity syndrome (ICANS), were infrequent in CRS, and when they did occur were generally low grade, with only 1 grade 3 ICANS event.
Asked in an interview whether the impressive response rates seen with JNJ-4528 might persist over time, Dr. Madduri acknowledged that follow-up is still relatively short.
“This product is unique in that has a CD8 central memory phenotype preferentially, and we’re hoping that this would play a central role in the durability of response because they’re memory cells, but I think at this time we don’t know,” she said.
The CARTITUDE-1 trial is funded by Janssen Research & Development. Dr. Madduri disclosed serving as a consultant to Janssen and to Takeda, Foundation Medicine, AbbVie, and Celgene.
SOURCE: Madduri D et al. ASH 2019. Abstract 577.
REPORTING FROM ASH 2019
Influenza already in midseason form
It’s been a decade since flu activity levels were this high this early in the season.
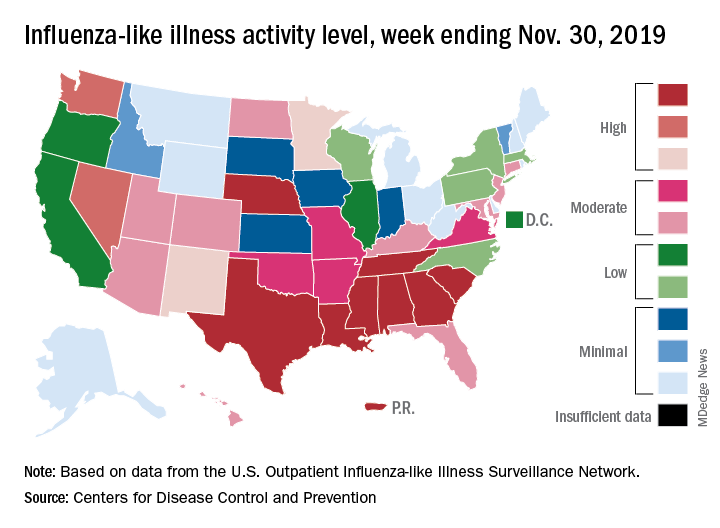
For the week ending Nov. 30, outpatient visits for influenza-like illness reached 3.5% of all visits to health care providers, the Centers for Disease Control and Prevention reported Dec. 6. That is the highest pre-December rate since the pandemic of 2009-2010, when the rate peaked at 7.7% in mid-October, CDC data show.
For the last week of November, eight states and Puerto Rico reported activity levels at the high point of the CDC’s 1-10 scale, which is at least five more states than any of the past five flu seasons. Three of the last five seasons had no states at level 10 this early in the season.
Another 4 states at levels 8 and 9 put a total of 13 jurisdictions in the “high” range of flu activity, with another 14 states in the “moderate” range of levels 6 and 7. Geographically speaking, 24 jurisdictions are experiencing regional or widespread activity, which is up from the 15 reported last week, the CDC’s influenza division said.
The hospitalization rate to date for the 2019-2020 season – 2.7 per 100,000 population – is “similar to what has been seen at this time during other recent seasons,” the CDC said.
One influenza-related pediatric death was reported during the week ending Nov. 30, which brings the total for the season to six, according to the CDC report.
It’s been a decade since flu activity levels were this high this early in the season.

For the week ending Nov. 30, outpatient visits for influenza-like illness reached 3.5% of all visits to health care providers, the Centers for Disease Control and Prevention reported Dec. 6. That is the highest pre-December rate since the pandemic of 2009-2010, when the rate peaked at 7.7% in mid-October, CDC data show.
For the last week of November, eight states and Puerto Rico reported activity levels at the high point of the CDC’s 1-10 scale, which is at least five more states than any of the past five flu seasons. Three of the last five seasons had no states at level 10 this early in the season.
Another 4 states at levels 8 and 9 put a total of 13 jurisdictions in the “high” range of flu activity, with another 14 states in the “moderate” range of levels 6 and 7. Geographically speaking, 24 jurisdictions are experiencing regional or widespread activity, which is up from the 15 reported last week, the CDC’s influenza division said.
The hospitalization rate to date for the 2019-2020 season – 2.7 per 100,000 population – is “similar to what has been seen at this time during other recent seasons,” the CDC said.
One influenza-related pediatric death was reported during the week ending Nov. 30, which brings the total for the season to six, according to the CDC report.
It’s been a decade since flu activity levels were this high this early in the season.

For the week ending Nov. 30, outpatient visits for influenza-like illness reached 3.5% of all visits to health care providers, the Centers for Disease Control and Prevention reported Dec. 6. That is the highest pre-December rate since the pandemic of 2009-2010, when the rate peaked at 7.7% in mid-October, CDC data show.
For the last week of November, eight states and Puerto Rico reported activity levels at the high point of the CDC’s 1-10 scale, which is at least five more states than any of the past five flu seasons. Three of the last five seasons had no states at level 10 this early in the season.
Another 4 states at levels 8 and 9 put a total of 13 jurisdictions in the “high” range of flu activity, with another 14 states in the “moderate” range of levels 6 and 7. Geographically speaking, 24 jurisdictions are experiencing regional or widespread activity, which is up from the 15 reported last week, the CDC’s influenza division said.
The hospitalization rate to date for the 2019-2020 season – 2.7 per 100,000 population – is “similar to what has been seen at this time during other recent seasons,” the CDC said.
One influenza-related pediatric death was reported during the week ending Nov. 30, which brings the total for the season to six, according to the CDC report.
Patient-reported outcomes support first-line pembrolizumab for NSCLC
Patient-reported outcomes support pembrolizumab plus chemotherapy for first-line treatment of metastatic non–small cell lung cancer (NSCLC), based on results from the KEYNOTE-407 trial.
At week 18, patients given pembrolizumab more often reported clinically meaningful health-related quality of life improvements than those in the placebo group, according to lead author Julien Mazieres, MD, PhD, of Paul Sabatier University in Toulouse, France, and colleagues.
Writing in the Journal of Clinical Oncology, the investigators explained that these findings build upon previously published results from KEYNOTE-407, which showed that adding pembrolizumab to chemotherapy in the first line could extend both progression-free and overall survival among patients with NSCLC. The benefits to quality of life associated with pembrolizumab align with similar findings from the KEYNOTE-024 and KEYNOTE-189 trials, they added.
The present analysis involved 559 patients with treatment-naive metastatic NSCLC. Patients were randomized to receive 4 cycles of placebo or pembrolizumab once every 3 weeks with carboplatin-based chemotherapy, followed by pembrolizumab or placebo for an additional 31 cycles. Health-related quality of life was assessed by two measures: the European Organisation for the Research and Treatment of Cancer Quality of Life Questionnaire–Core 30 (QLQ-C30) and QLQ–Lung Cancer Module 13 (QLQ-LC13).
Of the 559 patients enrolled, 554 completed at least one QLQ-C30 assessment and 553 completed at least one QLQ-LC13 assessment. These quality of life scores were compared temporally within treatment groups, from baseline to week 9 and week 18, and between groups. The investigators also analyzed median time to deterioration in chest pain, cough, and dyspnea.
Results showed that patients in the pembrolizumab group had statistically significant improvements in patient-reported outcomes over time and more frequently reported improvements than patients in the placebo group. Specifically, in the pembrolizumab group, least-squares mean score improved from baseline to week 9 (1.8) and week 18 (4.3); in comparison, least-squares mean score deteriorated in the placebo group from baseline to week 9 (–1.8) and week 18 (–0.57). Compared with placebo, treatment with pembrolizumab was associated with a least-squares mean change of 3.6 at week 9 (nominal P = .0337) and 4.9 at week 18 (nominal P = .0060). Stated differently, at week 18, compared with placebo, more patients in the pembrolizumab group reported clinically meaningful improvements in health-related quality of life (36.2% vs. 27.7%), and relatively fewer reported deterioration (22.8% vs. 31.3%). Median time to deterioration in symptoms was not reached in either treatment arm.
“These health-related quality of life findings, along with the improved efficacy (including overall survival benefit) of pembrolizumab plus carboplatin and paclitaxel/nab-paclitaxel, support its use as a first-line treatment of metastatic squamous NSCLC, regardless of programmed death–ligand 1 expression,” the investigators concluded.
The study was funded by Merck. The investigators reported additional relationships with Novartis, Genentech, Pfizer, and others.
SOURCE: Mazieres J et al. J Clin Oncol. 2019 Nov 21. doi: 10.1200/JCO.19.01348.
Patient-reported outcomes support pembrolizumab plus chemotherapy for first-line treatment of metastatic non–small cell lung cancer (NSCLC), based on results from the KEYNOTE-407 trial.
At week 18, patients given pembrolizumab more often reported clinically meaningful health-related quality of life improvements than those in the placebo group, according to lead author Julien Mazieres, MD, PhD, of Paul Sabatier University in Toulouse, France, and colleagues.
Writing in the Journal of Clinical Oncology, the investigators explained that these findings build upon previously published results from KEYNOTE-407, which showed that adding pembrolizumab to chemotherapy in the first line could extend both progression-free and overall survival among patients with NSCLC. The benefits to quality of life associated with pembrolizumab align with similar findings from the KEYNOTE-024 and KEYNOTE-189 trials, they added.
The present analysis involved 559 patients with treatment-naive metastatic NSCLC. Patients were randomized to receive 4 cycles of placebo or pembrolizumab once every 3 weeks with carboplatin-based chemotherapy, followed by pembrolizumab or placebo for an additional 31 cycles. Health-related quality of life was assessed by two measures: the European Organisation for the Research and Treatment of Cancer Quality of Life Questionnaire–Core 30 (QLQ-C30) and QLQ–Lung Cancer Module 13 (QLQ-LC13).
Of the 559 patients enrolled, 554 completed at least one QLQ-C30 assessment and 553 completed at least one QLQ-LC13 assessment. These quality of life scores were compared temporally within treatment groups, from baseline to week 9 and week 18, and between groups. The investigators also analyzed median time to deterioration in chest pain, cough, and dyspnea.
Results showed that patients in the pembrolizumab group had statistically significant improvements in patient-reported outcomes over time and more frequently reported improvements than patients in the placebo group. Specifically, in the pembrolizumab group, least-squares mean score improved from baseline to week 9 (1.8) and week 18 (4.3); in comparison, least-squares mean score deteriorated in the placebo group from baseline to week 9 (–1.8) and week 18 (–0.57). Compared with placebo, treatment with pembrolizumab was associated with a least-squares mean change of 3.6 at week 9 (nominal P = .0337) and 4.9 at week 18 (nominal P = .0060). Stated differently, at week 18, compared with placebo, more patients in the pembrolizumab group reported clinically meaningful improvements in health-related quality of life (36.2% vs. 27.7%), and relatively fewer reported deterioration (22.8% vs. 31.3%). Median time to deterioration in symptoms was not reached in either treatment arm.
“These health-related quality of life findings, along with the improved efficacy (including overall survival benefit) of pembrolizumab plus carboplatin and paclitaxel/nab-paclitaxel, support its use as a first-line treatment of metastatic squamous NSCLC, regardless of programmed death–ligand 1 expression,” the investigators concluded.
The study was funded by Merck. The investigators reported additional relationships with Novartis, Genentech, Pfizer, and others.
SOURCE: Mazieres J et al. J Clin Oncol. 2019 Nov 21. doi: 10.1200/JCO.19.01348.
Patient-reported outcomes support pembrolizumab plus chemotherapy for first-line treatment of metastatic non–small cell lung cancer (NSCLC), based on results from the KEYNOTE-407 trial.
At week 18, patients given pembrolizumab more often reported clinically meaningful health-related quality of life improvements than those in the placebo group, according to lead author Julien Mazieres, MD, PhD, of Paul Sabatier University in Toulouse, France, and colleagues.
Writing in the Journal of Clinical Oncology, the investigators explained that these findings build upon previously published results from KEYNOTE-407, which showed that adding pembrolizumab to chemotherapy in the first line could extend both progression-free and overall survival among patients with NSCLC. The benefits to quality of life associated with pembrolizumab align with similar findings from the KEYNOTE-024 and KEYNOTE-189 trials, they added.
The present analysis involved 559 patients with treatment-naive metastatic NSCLC. Patients were randomized to receive 4 cycles of placebo or pembrolizumab once every 3 weeks with carboplatin-based chemotherapy, followed by pembrolizumab or placebo for an additional 31 cycles. Health-related quality of life was assessed by two measures: the European Organisation for the Research and Treatment of Cancer Quality of Life Questionnaire–Core 30 (QLQ-C30) and QLQ–Lung Cancer Module 13 (QLQ-LC13).
Of the 559 patients enrolled, 554 completed at least one QLQ-C30 assessment and 553 completed at least one QLQ-LC13 assessment. These quality of life scores were compared temporally within treatment groups, from baseline to week 9 and week 18, and between groups. The investigators also analyzed median time to deterioration in chest pain, cough, and dyspnea.
Results showed that patients in the pembrolizumab group had statistically significant improvements in patient-reported outcomes over time and more frequently reported improvements than patients in the placebo group. Specifically, in the pembrolizumab group, least-squares mean score improved from baseline to week 9 (1.8) and week 18 (4.3); in comparison, least-squares mean score deteriorated in the placebo group from baseline to week 9 (–1.8) and week 18 (–0.57). Compared with placebo, treatment with pembrolizumab was associated with a least-squares mean change of 3.6 at week 9 (nominal P = .0337) and 4.9 at week 18 (nominal P = .0060). Stated differently, at week 18, compared with placebo, more patients in the pembrolizumab group reported clinically meaningful improvements in health-related quality of life (36.2% vs. 27.7%), and relatively fewer reported deterioration (22.8% vs. 31.3%). Median time to deterioration in symptoms was not reached in either treatment arm.
“These health-related quality of life findings, along with the improved efficacy (including overall survival benefit) of pembrolizumab plus carboplatin and paclitaxel/nab-paclitaxel, support its use as a first-line treatment of metastatic squamous NSCLC, regardless of programmed death–ligand 1 expression,” the investigators concluded.
The study was funded by Merck. The investigators reported additional relationships with Novartis, Genentech, Pfizer, and others.
SOURCE: Mazieres J et al. J Clin Oncol. 2019 Nov 21. doi: 10.1200/JCO.19.01348.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Bilateral mastectomy reduces second breast cancer risk, but not deaths
Bilateral mastectomy significantly decreases the risk for a second contralateral breast cancer, but does not decrease the risk of death, compared with breast-conserving therapy, results of a large retrospective study indicate.
Among 245,418 patients followed for a median of 6.7 years, the risk of death from breast cancer was similar for those who had undergone either breast-conserving therapy or bilateral mastectomy (BLM) but was 20% higher among women who had undergone unilateral mastectomy (ULM) when compared with breast-conserving therapy, reported Allison W. Kurian, MD, MSc, from Stanford (Calif.) University, and colleagues.
“Second breast cancers are rare, and their reduction should be weighed against the harms associated with BLM,” they wrote in a study published online in Cancer.
The investigators extracted data from the Surveillance, Epidemiology, and End Results program on all women diagnosed with American Joint Committee on Cancer stage 0 to stage III unilateral breast cancer in California from 1998 to 2015 who were treated with either BLM versus breast-conserving therapy, including surgery and radiation or unilateral mastectomy.
They calculated the absolute excess risk of contralateral breast cancer as the observed minus expected number of breast cancers in the general population divided by 10,000 person-years at risk.
Of 421,643 women with a first diagnosis of primary breast cancer during the study period, 245,418 met the study criteria. Of this cohort, 7,784 (3.2%) developed a contralateral second breast cancer more than 6 months after diagnosis of the first, after a median 6.7 years of follow-up.
Slightly more than half of the cohort (52.1%) had undergone breast-conserving therapy, 37.5% underwent unilateral mastectomy, and 7.6% had bilateral mastectomy. An additional 2.9% of patients were women aged 70 years and older with stage I hormone receptor–positive, HER2-negative disease who underwent breast-conserving surgery without radiation (percentages exceed 100% because of rounding).
A multivariate-adjusted model showed that, as might be expected, patients who underwent bilateral mastectomy had a 90% reduction in risk of contralateral cancer (hazard ratio, 0.10; P less than .001), compared with breast-conserving therapy. In contrast, patients who underwent unilateral mastectomy had a slight but significant increase in risk for a second contralateral breast cancer (HR, 1.07; P = .008).
The absolute excess risk for second contralateral breast cancer was 5 per 10,000 person-years with breast-conserving therapy, 13.6 per 10,000 person-years with unilateral mastectomy, and –28.6 per 10,000 person-years with bilateral mastectomy.
When they looked at risk for death, however they found that, compared with breast-conserving therapy, breast-conserving surgery alone (HR, 1.36; P = .0001) and unilateral mastectomy (HR, 1.21; P less than .001), but not bilateral mastectomy (HR, 1.03; P = .35) were significantly associated with increased risk for breast cancer death.
The authors noted that their estimates of absolute risk of second contralateral breast cancer jibe with those of earlier studies, and can help clinicians frame the discussion of the benefits versus risks for individual patients.
“What one patient might consider to be a negligible benefit of BLM, weighed against its potential harms of greater pain, recovery time, and impact on body image and employment, might appear worthwhile to another,” they wrote.
The study was funded by the National Cancer Institute, National Institutes of Health, Department of Health & Human Services, Suzanne Pride Bryan Fund for Breast Cancer Research, Jan Weimer Faculty Chair for Breast Oncology, and the BRCA Foundation. Dr. Kurian disclosed institutional research funding from Myriad Genetics.
SOURCE: Kurin AW et al. Cancer. 2019 Nov 21. doi: 10.1002/cncr.32618.
Bilateral mastectomy significantly decreases the risk for a second contralateral breast cancer, but does not decrease the risk of death, compared with breast-conserving therapy, results of a large retrospective study indicate.
Among 245,418 patients followed for a median of 6.7 years, the risk of death from breast cancer was similar for those who had undergone either breast-conserving therapy or bilateral mastectomy (BLM) but was 20% higher among women who had undergone unilateral mastectomy (ULM) when compared with breast-conserving therapy, reported Allison W. Kurian, MD, MSc, from Stanford (Calif.) University, and colleagues.
“Second breast cancers are rare, and their reduction should be weighed against the harms associated with BLM,” they wrote in a study published online in Cancer.
The investigators extracted data from the Surveillance, Epidemiology, and End Results program on all women diagnosed with American Joint Committee on Cancer stage 0 to stage III unilateral breast cancer in California from 1998 to 2015 who were treated with either BLM versus breast-conserving therapy, including surgery and radiation or unilateral mastectomy.
They calculated the absolute excess risk of contralateral breast cancer as the observed minus expected number of breast cancers in the general population divided by 10,000 person-years at risk.
Of 421,643 women with a first diagnosis of primary breast cancer during the study period, 245,418 met the study criteria. Of this cohort, 7,784 (3.2%) developed a contralateral second breast cancer more than 6 months after diagnosis of the first, after a median 6.7 years of follow-up.
Slightly more than half of the cohort (52.1%) had undergone breast-conserving therapy, 37.5% underwent unilateral mastectomy, and 7.6% had bilateral mastectomy. An additional 2.9% of patients were women aged 70 years and older with stage I hormone receptor–positive, HER2-negative disease who underwent breast-conserving surgery without radiation (percentages exceed 100% because of rounding).
A multivariate-adjusted model showed that, as might be expected, patients who underwent bilateral mastectomy had a 90% reduction in risk of contralateral cancer (hazard ratio, 0.10; P less than .001), compared with breast-conserving therapy. In contrast, patients who underwent unilateral mastectomy had a slight but significant increase in risk for a second contralateral breast cancer (HR, 1.07; P = .008).
The absolute excess risk for second contralateral breast cancer was 5 per 10,000 person-years with breast-conserving therapy, 13.6 per 10,000 person-years with unilateral mastectomy, and –28.6 per 10,000 person-years with bilateral mastectomy.
When they looked at risk for death, however they found that, compared with breast-conserving therapy, breast-conserving surgery alone (HR, 1.36; P = .0001) and unilateral mastectomy (HR, 1.21; P less than .001), but not bilateral mastectomy (HR, 1.03; P = .35) were significantly associated with increased risk for breast cancer death.
The authors noted that their estimates of absolute risk of second contralateral breast cancer jibe with those of earlier studies, and can help clinicians frame the discussion of the benefits versus risks for individual patients.
“What one patient might consider to be a negligible benefit of BLM, weighed against its potential harms of greater pain, recovery time, and impact on body image and employment, might appear worthwhile to another,” they wrote.
The study was funded by the National Cancer Institute, National Institutes of Health, Department of Health & Human Services, Suzanne Pride Bryan Fund for Breast Cancer Research, Jan Weimer Faculty Chair for Breast Oncology, and the BRCA Foundation. Dr. Kurian disclosed institutional research funding from Myriad Genetics.
SOURCE: Kurin AW et al. Cancer. 2019 Nov 21. doi: 10.1002/cncr.32618.
Bilateral mastectomy significantly decreases the risk for a second contralateral breast cancer, but does not decrease the risk of death, compared with breast-conserving therapy, results of a large retrospective study indicate.
Among 245,418 patients followed for a median of 6.7 years, the risk of death from breast cancer was similar for those who had undergone either breast-conserving therapy or bilateral mastectomy (BLM) but was 20% higher among women who had undergone unilateral mastectomy (ULM) when compared with breast-conserving therapy, reported Allison W. Kurian, MD, MSc, from Stanford (Calif.) University, and colleagues.
“Second breast cancers are rare, and their reduction should be weighed against the harms associated with BLM,” they wrote in a study published online in Cancer.
The investigators extracted data from the Surveillance, Epidemiology, and End Results program on all women diagnosed with American Joint Committee on Cancer stage 0 to stage III unilateral breast cancer in California from 1998 to 2015 who were treated with either BLM versus breast-conserving therapy, including surgery and radiation or unilateral mastectomy.
They calculated the absolute excess risk of contralateral breast cancer as the observed minus expected number of breast cancers in the general population divided by 10,000 person-years at risk.
Of 421,643 women with a first diagnosis of primary breast cancer during the study period, 245,418 met the study criteria. Of this cohort, 7,784 (3.2%) developed a contralateral second breast cancer more than 6 months after diagnosis of the first, after a median 6.7 years of follow-up.
Slightly more than half of the cohort (52.1%) had undergone breast-conserving therapy, 37.5% underwent unilateral mastectomy, and 7.6% had bilateral mastectomy. An additional 2.9% of patients were women aged 70 years and older with stage I hormone receptor–positive, HER2-negative disease who underwent breast-conserving surgery without radiation (percentages exceed 100% because of rounding).
A multivariate-adjusted model showed that, as might be expected, patients who underwent bilateral mastectomy had a 90% reduction in risk of contralateral cancer (hazard ratio, 0.10; P less than .001), compared with breast-conserving therapy. In contrast, patients who underwent unilateral mastectomy had a slight but significant increase in risk for a second contralateral breast cancer (HR, 1.07; P = .008).
The absolute excess risk for second contralateral breast cancer was 5 per 10,000 person-years with breast-conserving therapy, 13.6 per 10,000 person-years with unilateral mastectomy, and –28.6 per 10,000 person-years with bilateral mastectomy.
When they looked at risk for death, however they found that, compared with breast-conserving therapy, breast-conserving surgery alone (HR, 1.36; P = .0001) and unilateral mastectomy (HR, 1.21; P less than .001), but not bilateral mastectomy (HR, 1.03; P = .35) were significantly associated with increased risk for breast cancer death.
The authors noted that their estimates of absolute risk of second contralateral breast cancer jibe with those of earlier studies, and can help clinicians frame the discussion of the benefits versus risks for individual patients.
“What one patient might consider to be a negligible benefit of BLM, weighed against its potential harms of greater pain, recovery time, and impact on body image and employment, might appear worthwhile to another,” they wrote.
The study was funded by the National Cancer Institute, National Institutes of Health, Department of Health & Human Services, Suzanne Pride Bryan Fund for Breast Cancer Research, Jan Weimer Faculty Chair for Breast Oncology, and the BRCA Foundation. Dr. Kurian disclosed institutional research funding from Myriad Genetics.
SOURCE: Kurin AW et al. Cancer. 2019 Nov 21. doi: 10.1002/cncr.32618.
FROM CANCER
Intensive BP control reduced dementia but increased brain atrophy and hurt cognition
SAN DIEGO – Intensive blood pressure control over 4 years reduced the overall risk of all-cause dementia by 17%, compared with standard care, but in subanalyses of the Systolic Blood Pressure Intervention Trial (SPRINT) it was also associated with significant decreases in cognitive function and total brain volume, researchers said at the Clinical Trials on Alzheimer’s Disease conference.
Whether these between-group differences were clinically meaningful was the topic of some debate, but they were enough to prompt Mary Sano, PhD, to strongly state her reservations.
“The cardiovascular effects of SPRINT were impressive, but I am concerned about minimizing the potentially negative effect on cognition,” said Dr. Sano, professor of psychiatry and director of the Alzheimer’s Disease Research Center at the Icahn School of Medicine at Mount Sinai, New York. “Do I really want to treat a healthy, nonimpaired patient like this if I have to warn them that their cognition might actually get worse? We just cannot minimize this risk. There is very strong evidence that [intensive treatment of blood pressure] might be a step backward in cognition. Would you lower your own blood pressure at a risk of losing some points on your cognition?”
The subanalyses were conducted as part of the SPRINT Memory and Cognition In Decreased Hypertension (SPRINT MIND) substudy, which looked at cardiovascular and mortality outcomes in 9,361 subjects whose hypertension was managed intensively or by standard care (target systolic blood pressure less than 120 mm Hg vs. less than 140 mm Hg). The trial was stopped early because of a 25% reduction in the primary composite cardiovascular disease endpoint and a 27% reduction in all-cause mortality in the intensive-treatment group.
SPRINT MIND examined the risks of incident probable dementia, mild cognitive impairment (MCI), and a composite outcome of both. Intensive control reduced the risk of MCI by 19% and the combined outcome by 15%.
At the conference, SPRINT MIND investigators presented three long-term subanalyses with a median intervention and follow-up time of about 4 years.
Sarah Gaussoin of Wake Forest University, Winston-Salem, N.C., presented unpublished data detailing the effects of intensive control on several dementia subtypes: nonamnestic single domain, nonamnestic multidomain, amnestic single domain, and amnestic multidomain. There were 640 subjects in this analysis.
After a median of 3.3 years of intervention and 5 years of follow-up, there were no differences in the rate of incident probable dementia between the single- and multidomain nonamnestic groups. “We did see a strong 22% decreased risk in single-domain versus multidomain amnestic MCI, however,” she said.
Nicholas Pajewski, PhD, also of Wake Forest University, discussed more detailed cognitive outcomes in SPRINT MIND among 2,900 subjects who had a full battery of cognitive testing at every assessment over 5 years. The outcomes included memory deficit and processing speed.
Dr. Pajewski reported finding no significant difference between the groups in the rates of memory decline in either outcome. But there was a greater rate of decline in processing speed in the intensively treated group, he added. The difference was small but statistically significant.
The difference was largely driven by results of a single cognitive test – the Trail Making Test Part A. “It corresponded to about a 1.25-second increase over 4 years,” in processing speed on this test, Dr. Pajewski said.
There were no between-group differences in any of the other domains explored, including language, executive function, global cognitive function, or the Montreal Cognitive Assessment.
“Obviously, these results are perplexing,” given the overall positive results of SPRINT MIND, he said. “Intensive blood pressure control is a beneficial thing, and we expected to see an effect on memory, or a blunting of decline, and instead we saw some small decrements going the other way. This led us to speculate about what’s going on.”
The trial relied on a narrow definition of MCI that might have affected the outcomes. There was also a very broad range of ages in the study, ranging from 53 to 86 years. More importantly, he said, the original SPRINT study didn’t collect cognitive data at baseline, so there was no way to know how many subjects already might have had MCI when they entered the trial.
Ilya Nasrallah, MD, PhD, of the University of Pennsylvania, Philadelphia, presented MRI data on white-matter lesions, hippocampal volume fractional anisotropy in the cingulum, and cerebral blood flow. The median time between scans was 4 years, with a median treatment time of 3.4 years.
The standard-care group showed a significantly greater increase in white-matter lesion volume at the follow-up scan than did the intensive-treatment group (1.45 cm3 vs. 0.92 cm3). But the intensively treated group had significantly more brain atrophy, losing a median of 30.6 cm3, compared with a loss of 26.9 cm3 in the standard-treatment group.
“It was a very small difference amounting to less than 1% of the total brain volume, but it was still statistically significant,” Dr. Nasrallah said.
Loss of gray-matter volume drove about two-thirds of the difference in the intensively treated group. There was a corresponding increase in cerebrospinal fluid volume that was driven by differences in the ventricles and the subarachnoid space.
However, there were no significant differences in right, left, or total hippocampal volume. There also were no differences in cingulate bundle anisotropy or cerebral blood flow.
SPRINT was funded by the National Institutes of Health. None of the investigators reported having financial conflicts of interest.
SAN DIEGO – Intensive blood pressure control over 4 years reduced the overall risk of all-cause dementia by 17%, compared with standard care, but in subanalyses of the Systolic Blood Pressure Intervention Trial (SPRINT) it was also associated with significant decreases in cognitive function and total brain volume, researchers said at the Clinical Trials on Alzheimer’s Disease conference.
Whether these between-group differences were clinically meaningful was the topic of some debate, but they were enough to prompt Mary Sano, PhD, to strongly state her reservations.
“The cardiovascular effects of SPRINT were impressive, but I am concerned about minimizing the potentially negative effect on cognition,” said Dr. Sano, professor of psychiatry and director of the Alzheimer’s Disease Research Center at the Icahn School of Medicine at Mount Sinai, New York. “Do I really want to treat a healthy, nonimpaired patient like this if I have to warn them that their cognition might actually get worse? We just cannot minimize this risk. There is very strong evidence that [intensive treatment of blood pressure] might be a step backward in cognition. Would you lower your own blood pressure at a risk of losing some points on your cognition?”
The subanalyses were conducted as part of the SPRINT Memory and Cognition In Decreased Hypertension (SPRINT MIND) substudy, which looked at cardiovascular and mortality outcomes in 9,361 subjects whose hypertension was managed intensively or by standard care (target systolic blood pressure less than 120 mm Hg vs. less than 140 mm Hg). The trial was stopped early because of a 25% reduction in the primary composite cardiovascular disease endpoint and a 27% reduction in all-cause mortality in the intensive-treatment group.
SPRINT MIND examined the risks of incident probable dementia, mild cognitive impairment (MCI), and a composite outcome of both. Intensive control reduced the risk of MCI by 19% and the combined outcome by 15%.
At the conference, SPRINT MIND investigators presented three long-term subanalyses with a median intervention and follow-up time of about 4 years.
Sarah Gaussoin of Wake Forest University, Winston-Salem, N.C., presented unpublished data detailing the effects of intensive control on several dementia subtypes: nonamnestic single domain, nonamnestic multidomain, amnestic single domain, and amnestic multidomain. There were 640 subjects in this analysis.
After a median of 3.3 years of intervention and 5 years of follow-up, there were no differences in the rate of incident probable dementia between the single- and multidomain nonamnestic groups. “We did see a strong 22% decreased risk in single-domain versus multidomain amnestic MCI, however,” she said.
Nicholas Pajewski, PhD, also of Wake Forest University, discussed more detailed cognitive outcomes in SPRINT MIND among 2,900 subjects who had a full battery of cognitive testing at every assessment over 5 years. The outcomes included memory deficit and processing speed.
Dr. Pajewski reported finding no significant difference between the groups in the rates of memory decline in either outcome. But there was a greater rate of decline in processing speed in the intensively treated group, he added. The difference was small but statistically significant.
The difference was largely driven by results of a single cognitive test – the Trail Making Test Part A. “It corresponded to about a 1.25-second increase over 4 years,” in processing speed on this test, Dr. Pajewski said.
There were no between-group differences in any of the other domains explored, including language, executive function, global cognitive function, or the Montreal Cognitive Assessment.
“Obviously, these results are perplexing,” given the overall positive results of SPRINT MIND, he said. “Intensive blood pressure control is a beneficial thing, and we expected to see an effect on memory, or a blunting of decline, and instead we saw some small decrements going the other way. This led us to speculate about what’s going on.”
The trial relied on a narrow definition of MCI that might have affected the outcomes. There was also a very broad range of ages in the study, ranging from 53 to 86 years. More importantly, he said, the original SPRINT study didn’t collect cognitive data at baseline, so there was no way to know how many subjects already might have had MCI when they entered the trial.
Ilya Nasrallah, MD, PhD, of the University of Pennsylvania, Philadelphia, presented MRI data on white-matter lesions, hippocampal volume fractional anisotropy in the cingulum, and cerebral blood flow. The median time between scans was 4 years, with a median treatment time of 3.4 years.
The standard-care group showed a significantly greater increase in white-matter lesion volume at the follow-up scan than did the intensive-treatment group (1.45 cm3 vs. 0.92 cm3). But the intensively treated group had significantly more brain atrophy, losing a median of 30.6 cm3, compared with a loss of 26.9 cm3 in the standard-treatment group.
“It was a very small difference amounting to less than 1% of the total brain volume, but it was still statistically significant,” Dr. Nasrallah said.
Loss of gray-matter volume drove about two-thirds of the difference in the intensively treated group. There was a corresponding increase in cerebrospinal fluid volume that was driven by differences in the ventricles and the subarachnoid space.
However, there were no significant differences in right, left, or total hippocampal volume. There also were no differences in cingulate bundle anisotropy or cerebral blood flow.
SPRINT was funded by the National Institutes of Health. None of the investigators reported having financial conflicts of interest.
SAN DIEGO – Intensive blood pressure control over 4 years reduced the overall risk of all-cause dementia by 17%, compared with standard care, but in subanalyses of the Systolic Blood Pressure Intervention Trial (SPRINT) it was also associated with significant decreases in cognitive function and total brain volume, researchers said at the Clinical Trials on Alzheimer’s Disease conference.
Whether these between-group differences were clinically meaningful was the topic of some debate, but they were enough to prompt Mary Sano, PhD, to strongly state her reservations.
“The cardiovascular effects of SPRINT were impressive, but I am concerned about minimizing the potentially negative effect on cognition,” said Dr. Sano, professor of psychiatry and director of the Alzheimer’s Disease Research Center at the Icahn School of Medicine at Mount Sinai, New York. “Do I really want to treat a healthy, nonimpaired patient like this if I have to warn them that their cognition might actually get worse? We just cannot minimize this risk. There is very strong evidence that [intensive treatment of blood pressure] might be a step backward in cognition. Would you lower your own blood pressure at a risk of losing some points on your cognition?”
The subanalyses were conducted as part of the SPRINT Memory and Cognition In Decreased Hypertension (SPRINT MIND) substudy, which looked at cardiovascular and mortality outcomes in 9,361 subjects whose hypertension was managed intensively or by standard care (target systolic blood pressure less than 120 mm Hg vs. less than 140 mm Hg). The trial was stopped early because of a 25% reduction in the primary composite cardiovascular disease endpoint and a 27% reduction in all-cause mortality in the intensive-treatment group.
SPRINT MIND examined the risks of incident probable dementia, mild cognitive impairment (MCI), and a composite outcome of both. Intensive control reduced the risk of MCI by 19% and the combined outcome by 15%.
At the conference, SPRINT MIND investigators presented three long-term subanalyses with a median intervention and follow-up time of about 4 years.
Sarah Gaussoin of Wake Forest University, Winston-Salem, N.C., presented unpublished data detailing the effects of intensive control on several dementia subtypes: nonamnestic single domain, nonamnestic multidomain, amnestic single domain, and amnestic multidomain. There were 640 subjects in this analysis.
After a median of 3.3 years of intervention and 5 years of follow-up, there were no differences in the rate of incident probable dementia between the single- and multidomain nonamnestic groups. “We did see a strong 22% decreased risk in single-domain versus multidomain amnestic MCI, however,” she said.
Nicholas Pajewski, PhD, also of Wake Forest University, discussed more detailed cognitive outcomes in SPRINT MIND among 2,900 subjects who had a full battery of cognitive testing at every assessment over 5 years. The outcomes included memory deficit and processing speed.
Dr. Pajewski reported finding no significant difference between the groups in the rates of memory decline in either outcome. But there was a greater rate of decline in processing speed in the intensively treated group, he added. The difference was small but statistically significant.
The difference was largely driven by results of a single cognitive test – the Trail Making Test Part A. “It corresponded to about a 1.25-second increase over 4 years,” in processing speed on this test, Dr. Pajewski said.
There were no between-group differences in any of the other domains explored, including language, executive function, global cognitive function, or the Montreal Cognitive Assessment.
“Obviously, these results are perplexing,” given the overall positive results of SPRINT MIND, he said. “Intensive blood pressure control is a beneficial thing, and we expected to see an effect on memory, or a blunting of decline, and instead we saw some small decrements going the other way. This led us to speculate about what’s going on.”
The trial relied on a narrow definition of MCI that might have affected the outcomes. There was also a very broad range of ages in the study, ranging from 53 to 86 years. More importantly, he said, the original SPRINT study didn’t collect cognitive data at baseline, so there was no way to know how many subjects already might have had MCI when they entered the trial.
Ilya Nasrallah, MD, PhD, of the University of Pennsylvania, Philadelphia, presented MRI data on white-matter lesions, hippocampal volume fractional anisotropy in the cingulum, and cerebral blood flow. The median time between scans was 4 years, with a median treatment time of 3.4 years.
The standard-care group showed a significantly greater increase in white-matter lesion volume at the follow-up scan than did the intensive-treatment group (1.45 cm3 vs. 0.92 cm3). But the intensively treated group had significantly more brain atrophy, losing a median of 30.6 cm3, compared with a loss of 26.9 cm3 in the standard-treatment group.
“It was a very small difference amounting to less than 1% of the total brain volume, but it was still statistically significant,” Dr. Nasrallah said.
Loss of gray-matter volume drove about two-thirds of the difference in the intensively treated group. There was a corresponding increase in cerebrospinal fluid volume that was driven by differences in the ventricles and the subarachnoid space.
However, there were no significant differences in right, left, or total hippocampal volume. There also were no differences in cingulate bundle anisotropy or cerebral blood flow.
SPRINT was funded by the National Institutes of Health. None of the investigators reported having financial conflicts of interest.
REPORTING FROM CTAD 2019
New opioid recommendations: Pain from most dermatologic procedures should be managed with acetaminophen, ibuprofen
has recommended.
Rotation flaps, interpolation flaps, wedge resections, cartilage alar-batten grafts, and Mustarde flaps were among the 20 procedures that can be managed with up to 10 oral oxycodone 5-mg equivalents, according to the panel. Only the Abbe procedure might warrant dispensing up to 15 oxycodone 5-mg pills, Justin McLawhorn, MD, and colleagues wrote in the Journal of the American Academy of Dermatology. The recommended amount of opioids are in addition to nonopioid analgesics, the guidelines point out.
All the other procedures can – and should – be managed with a combination of acetaminophen and ibuprofen, either alone or in an alternating dose pattern, said Dr. McLawhorn, of the department of dermatology at the University of Oklahoma Health Sciences Center, Oklahoma City, and coauthors.
But limited opioid prescribing is an important part of healing for patients who undergo the most invasive procedures, they wrote. “The management of complications, including adequate pain control, should be tailored to each patient on a case-by-case basis. Moreover, any pain management plan should not strictly adhere to any single guideline, but rather should be formed with consideration of the expected pain from the procedure and/or closure and consider the patient’s expectations for pain control.”
The time is ripe for dermatologists to make a stand in combating the opioid crisis, according to a group email response to questions from Dr. McLawhorn, Thomas Stasko, MD, professor and chair of dermatology at the University of Oklahoma, Oklahoma City, and Lindsey Collins, MD, also of the University of Oklahoma.
“The opioid crisis has reached epidemic proportions. More than 70,000 Americans have died from an opioid overdose in 2017,” they wrote. “Moreover, recent data suggest that nearly 6% of postsurgical, opioid-naive patients become long-term users of opioids. The lack of specific evidence-based recommendations likely contributes to a wide variety in prescribing patterns and a steady supply of unused opioids. Countering the opioid crisis necessitates a restructuring of the opioid prescribing practices that addresses pain in a procedure-specific manner. These recommendations are one tool in the dermatologists’ arsenal that can be used as a reference to help guide opioid management and prevent excessive opioid prescriptions at discharge following dermatologic interventions.”
Unfortunately, they added, dermatologists have inadvertently fueled the opioid abuse fire.
“It is difficult to quantify which providers are responsible for the onslaught of opioids into our communities,” the authors wrote in the email interview. “However, we can deduce, based on recent opioid prescribing patterns, that dermatologists provide approximately 500,000 unused opioid pills to their communities on an annual basis. This is the result of a wide variation in practice patterns and narratives that have been previously circulated in an attempt to mitigate the providers’ perception of the addictive nature of opioid analgesics. Our hope is that by addressing pain in a procedure-specific manner, we can help to limit the excessive number of unused opioid pills that are provided by dermatologists and ultimately decrease the rate of opioid-related complications, including addiction and death.”
Still, patients need and deserve effective pain management after a procedure. In the guidelines, the investigators wrote that a “one-size-fits-all” approach “does not account for the mechanism of pain, the invasiveness of the procedure, or the anatomic structures that are manipulated. As a result, current guidelines cannot accurately predict the quantity of opioids that are necessary to manage postoperative pain.”
The panel brought together experts in general dermatology, dermatologic surgery, cosmetics, and phlebology to develop a consensus on opioid prescribing guidelines for 87 of the most common procedures. Everyone on the panel was a member of the American College of Mohs Surgery, American Academy of Dermatology, or the American Vein and Lymphatic Society. The panel conducted a literature review to determine which procedures might require opioids and which would not. At least 75% of the panel had to agree on a reasonable but effective opioid amount; they were then polled as to whether they might employ that recommendation in their own clinical practice.
The recommendations are aimed at patients who experienced no peri- or postoperative complications.
The panel agreed that acetaminophen and ibuprofen – alone, in combination, or with opioids – were reasonable choices for all the 87 procedures. In such instances, acetaminophen 1 g can be staggered with ibuprofen 400 mg every 4 or 8 hours.
“I think providers will encounter a mixed bag of preconceived notions regarding patients’ expectations for pain control,” Dr. McLawhorn and coauthors wrote in the interview. “The important point for providers to make is to emphasize the noninferiority of acetaminophen and/or ibuprofen in controlling acute pain for patients who are not dependent on opioids for the management of chronic pain. Our experience in caring for many surgical patients has shown that patients are usually receptive to the use of nonopioid analgesics as many are familiar with their addictive potential because of the uptick in the publicity of the opioid-related complications.”
In cases where opioids might be appropriate, the panel unanimously agreed that dose limits be imposed. For 15 of the 87 procedures, the panel recommend a maximum prescription of 10 oxycodone 5-mg equivalents. Only one other – the Abbe flap – might warrant more, with a maximum of 15 oxycodone 5-mg pills at discharge.
Sometimes called a “lip switch,” the Abbe flap is reconstruction for full-thickness lip defects. It is a composite flap that moves skin, muscle, mucosa, and blood supply from the lower lip to reconstruct a defect of the upper lip. This reconstruction attempts to respect the native anatomic landmarks of the lip and allow for a better functional outcome.
“Because of the extensive nature of the repair and the anatomic territories that are manipulated, including the suturing of the lower lip to the upper lip with delayed separation, adequate pain control may require opioid analgesics in the immediate postoperative period,” the team wrote in the interview.
The panel could not agree on pain management strategies for five other procedures: Karapandzic flaps, en bloc nail excisions, facial resurfacing with deep chemical peels, and small- or large-volume liposuction. This was partly because of a lack of personal experience. Only 8 of the 40 panelists performed Karapandzic flaps. The maximum number of 5-mg oxycodone tablets any panelist prescribed for Karapandzic flaps and en bloc nail excisions was 20.
Facial resurfacing was likewise an uncommon procedure for the panel, with just 11 members performing this using deep chemical peels. However, five of those panelists said that opioids were routinely needed for postoperative pain with a maximum of 15 oxycodone 5-mg equivalents. And just four panelists performed liposuction, for which they used a maximum of 15 oxycodone 5-mg equivalents.
“However,” they wrote in the guidelines, “these providers noted that the location where the procedure is performed strongly influences the need for opioid pain management, with small-volume removal in the neck, arms, or flanks being unlikely to require opioids for adequate pain control, whereas large-volume removal in the thighs, knees, and hips may routinely require opioids.”
Addressing patient expectations is a very important part of pain management, the panel noted. “Patients will invariably experience postoperative pain after cutaneous surgeries or other interventions, often peaking within 4 hours after surgery. Wound tension, size and type of repair, anatomical location/nerve innervation, and patient pain tolerance are all factors that contribute to postoperative discomfort and should be considered when developing a postoperative pain management plan.”
Ultimately, according to Dr. McLawhorn and coauthors, the decision to use opioids at discharge for postoperative pain control should be an individual one based on patients’ comorbidities and expectations.
“Admittedly, many of the procedures listed within the recommendations may result in a rather large or complex defect that requires an equally large or complex repair,” they wrote in the interview. “However, proper education of the patient and provider regarding the risks of addiction with the use of opioids even short term should be discussed as part of every preoperative consultation. Furthermore, the patient and the provider must discuss their expectations for postoperative pain interventions for adequate pain control.”
SOURCE: McLawhorn J et al. J Am Acad Dermatol. 2019 Nov 12. doi: 10.1016/j.jaad.2019.09.080.
has recommended.
Rotation flaps, interpolation flaps, wedge resections, cartilage alar-batten grafts, and Mustarde flaps were among the 20 procedures that can be managed with up to 10 oral oxycodone 5-mg equivalents, according to the panel. Only the Abbe procedure might warrant dispensing up to 15 oxycodone 5-mg pills, Justin McLawhorn, MD, and colleagues wrote in the Journal of the American Academy of Dermatology. The recommended amount of opioids are in addition to nonopioid analgesics, the guidelines point out.
All the other procedures can – and should – be managed with a combination of acetaminophen and ibuprofen, either alone or in an alternating dose pattern, said Dr. McLawhorn, of the department of dermatology at the University of Oklahoma Health Sciences Center, Oklahoma City, and coauthors.
But limited opioid prescribing is an important part of healing for patients who undergo the most invasive procedures, they wrote. “The management of complications, including adequate pain control, should be tailored to each patient on a case-by-case basis. Moreover, any pain management plan should not strictly adhere to any single guideline, but rather should be formed with consideration of the expected pain from the procedure and/or closure and consider the patient’s expectations for pain control.”
The time is ripe for dermatologists to make a stand in combating the opioid crisis, according to a group email response to questions from Dr. McLawhorn, Thomas Stasko, MD, professor and chair of dermatology at the University of Oklahoma, Oklahoma City, and Lindsey Collins, MD, also of the University of Oklahoma.
“The opioid crisis has reached epidemic proportions. More than 70,000 Americans have died from an opioid overdose in 2017,” they wrote. “Moreover, recent data suggest that nearly 6% of postsurgical, opioid-naive patients become long-term users of opioids. The lack of specific evidence-based recommendations likely contributes to a wide variety in prescribing patterns and a steady supply of unused opioids. Countering the opioid crisis necessitates a restructuring of the opioid prescribing practices that addresses pain in a procedure-specific manner. These recommendations are one tool in the dermatologists’ arsenal that can be used as a reference to help guide opioid management and prevent excessive opioid prescriptions at discharge following dermatologic interventions.”
Unfortunately, they added, dermatologists have inadvertently fueled the opioid abuse fire.
“It is difficult to quantify which providers are responsible for the onslaught of opioids into our communities,” the authors wrote in the email interview. “However, we can deduce, based on recent opioid prescribing patterns, that dermatologists provide approximately 500,000 unused opioid pills to their communities on an annual basis. This is the result of a wide variation in practice patterns and narratives that have been previously circulated in an attempt to mitigate the providers’ perception of the addictive nature of opioid analgesics. Our hope is that by addressing pain in a procedure-specific manner, we can help to limit the excessive number of unused opioid pills that are provided by dermatologists and ultimately decrease the rate of opioid-related complications, including addiction and death.”
Still, patients need and deserve effective pain management after a procedure. In the guidelines, the investigators wrote that a “one-size-fits-all” approach “does not account for the mechanism of pain, the invasiveness of the procedure, or the anatomic structures that are manipulated. As a result, current guidelines cannot accurately predict the quantity of opioids that are necessary to manage postoperative pain.”
The panel brought together experts in general dermatology, dermatologic surgery, cosmetics, and phlebology to develop a consensus on opioid prescribing guidelines for 87 of the most common procedures. Everyone on the panel was a member of the American College of Mohs Surgery, American Academy of Dermatology, or the American Vein and Lymphatic Society. The panel conducted a literature review to determine which procedures might require opioids and which would not. At least 75% of the panel had to agree on a reasonable but effective opioid amount; they were then polled as to whether they might employ that recommendation in their own clinical practice.
The recommendations are aimed at patients who experienced no peri- or postoperative complications.
The panel agreed that acetaminophen and ibuprofen – alone, in combination, or with opioids – were reasonable choices for all the 87 procedures. In such instances, acetaminophen 1 g can be staggered with ibuprofen 400 mg every 4 or 8 hours.
“I think providers will encounter a mixed bag of preconceived notions regarding patients’ expectations for pain control,” Dr. McLawhorn and coauthors wrote in the interview. “The important point for providers to make is to emphasize the noninferiority of acetaminophen and/or ibuprofen in controlling acute pain for patients who are not dependent on opioids for the management of chronic pain. Our experience in caring for many surgical patients has shown that patients are usually receptive to the use of nonopioid analgesics as many are familiar with their addictive potential because of the uptick in the publicity of the opioid-related complications.”
In cases where opioids might be appropriate, the panel unanimously agreed that dose limits be imposed. For 15 of the 87 procedures, the panel recommend a maximum prescription of 10 oxycodone 5-mg equivalents. Only one other – the Abbe flap – might warrant more, with a maximum of 15 oxycodone 5-mg pills at discharge.
Sometimes called a “lip switch,” the Abbe flap is reconstruction for full-thickness lip defects. It is a composite flap that moves skin, muscle, mucosa, and blood supply from the lower lip to reconstruct a defect of the upper lip. This reconstruction attempts to respect the native anatomic landmarks of the lip and allow for a better functional outcome.
“Because of the extensive nature of the repair and the anatomic territories that are manipulated, including the suturing of the lower lip to the upper lip with delayed separation, adequate pain control may require opioid analgesics in the immediate postoperative period,” the team wrote in the interview.
The panel could not agree on pain management strategies for five other procedures: Karapandzic flaps, en bloc nail excisions, facial resurfacing with deep chemical peels, and small- or large-volume liposuction. This was partly because of a lack of personal experience. Only 8 of the 40 panelists performed Karapandzic flaps. The maximum number of 5-mg oxycodone tablets any panelist prescribed for Karapandzic flaps and en bloc nail excisions was 20.
Facial resurfacing was likewise an uncommon procedure for the panel, with just 11 members performing this using deep chemical peels. However, five of those panelists said that opioids were routinely needed for postoperative pain with a maximum of 15 oxycodone 5-mg equivalents. And just four panelists performed liposuction, for which they used a maximum of 15 oxycodone 5-mg equivalents.
“However,” they wrote in the guidelines, “these providers noted that the location where the procedure is performed strongly influences the need for opioid pain management, with small-volume removal in the neck, arms, or flanks being unlikely to require opioids for adequate pain control, whereas large-volume removal in the thighs, knees, and hips may routinely require opioids.”
Addressing patient expectations is a very important part of pain management, the panel noted. “Patients will invariably experience postoperative pain after cutaneous surgeries or other interventions, often peaking within 4 hours after surgery. Wound tension, size and type of repair, anatomical location/nerve innervation, and patient pain tolerance are all factors that contribute to postoperative discomfort and should be considered when developing a postoperative pain management plan.”
Ultimately, according to Dr. McLawhorn and coauthors, the decision to use opioids at discharge for postoperative pain control should be an individual one based on patients’ comorbidities and expectations.
“Admittedly, many of the procedures listed within the recommendations may result in a rather large or complex defect that requires an equally large or complex repair,” they wrote in the interview. “However, proper education of the patient and provider regarding the risks of addiction with the use of opioids even short term should be discussed as part of every preoperative consultation. Furthermore, the patient and the provider must discuss their expectations for postoperative pain interventions for adequate pain control.”
SOURCE: McLawhorn J et al. J Am Acad Dermatol. 2019 Nov 12. doi: 10.1016/j.jaad.2019.09.080.
has recommended.
Rotation flaps, interpolation flaps, wedge resections, cartilage alar-batten grafts, and Mustarde flaps were among the 20 procedures that can be managed with up to 10 oral oxycodone 5-mg equivalents, according to the panel. Only the Abbe procedure might warrant dispensing up to 15 oxycodone 5-mg pills, Justin McLawhorn, MD, and colleagues wrote in the Journal of the American Academy of Dermatology. The recommended amount of opioids are in addition to nonopioid analgesics, the guidelines point out.
All the other procedures can – and should – be managed with a combination of acetaminophen and ibuprofen, either alone or in an alternating dose pattern, said Dr. McLawhorn, of the department of dermatology at the University of Oklahoma Health Sciences Center, Oklahoma City, and coauthors.
But limited opioid prescribing is an important part of healing for patients who undergo the most invasive procedures, they wrote. “The management of complications, including adequate pain control, should be tailored to each patient on a case-by-case basis. Moreover, any pain management plan should not strictly adhere to any single guideline, but rather should be formed with consideration of the expected pain from the procedure and/or closure and consider the patient’s expectations for pain control.”
The time is ripe for dermatologists to make a stand in combating the opioid crisis, according to a group email response to questions from Dr. McLawhorn, Thomas Stasko, MD, professor and chair of dermatology at the University of Oklahoma, Oklahoma City, and Lindsey Collins, MD, also of the University of Oklahoma.
“The opioid crisis has reached epidemic proportions. More than 70,000 Americans have died from an opioid overdose in 2017,” they wrote. “Moreover, recent data suggest that nearly 6% of postsurgical, opioid-naive patients become long-term users of opioids. The lack of specific evidence-based recommendations likely contributes to a wide variety in prescribing patterns and a steady supply of unused opioids. Countering the opioid crisis necessitates a restructuring of the opioid prescribing practices that addresses pain in a procedure-specific manner. These recommendations are one tool in the dermatologists’ arsenal that can be used as a reference to help guide opioid management and prevent excessive opioid prescriptions at discharge following dermatologic interventions.”
Unfortunately, they added, dermatologists have inadvertently fueled the opioid abuse fire.
“It is difficult to quantify which providers are responsible for the onslaught of opioids into our communities,” the authors wrote in the email interview. “However, we can deduce, based on recent opioid prescribing patterns, that dermatologists provide approximately 500,000 unused opioid pills to their communities on an annual basis. This is the result of a wide variation in practice patterns and narratives that have been previously circulated in an attempt to mitigate the providers’ perception of the addictive nature of opioid analgesics. Our hope is that by addressing pain in a procedure-specific manner, we can help to limit the excessive number of unused opioid pills that are provided by dermatologists and ultimately decrease the rate of opioid-related complications, including addiction and death.”
Still, patients need and deserve effective pain management after a procedure. In the guidelines, the investigators wrote that a “one-size-fits-all” approach “does not account for the mechanism of pain, the invasiveness of the procedure, or the anatomic structures that are manipulated. As a result, current guidelines cannot accurately predict the quantity of opioids that are necessary to manage postoperative pain.”
The panel brought together experts in general dermatology, dermatologic surgery, cosmetics, and phlebology to develop a consensus on opioid prescribing guidelines for 87 of the most common procedures. Everyone on the panel was a member of the American College of Mohs Surgery, American Academy of Dermatology, or the American Vein and Lymphatic Society. The panel conducted a literature review to determine which procedures might require opioids and which would not. At least 75% of the panel had to agree on a reasonable but effective opioid amount; they were then polled as to whether they might employ that recommendation in their own clinical practice.
The recommendations are aimed at patients who experienced no peri- or postoperative complications.
The panel agreed that acetaminophen and ibuprofen – alone, in combination, or with opioids – were reasonable choices for all the 87 procedures. In such instances, acetaminophen 1 g can be staggered with ibuprofen 400 mg every 4 or 8 hours.
“I think providers will encounter a mixed bag of preconceived notions regarding patients’ expectations for pain control,” Dr. McLawhorn and coauthors wrote in the interview. “The important point for providers to make is to emphasize the noninferiority of acetaminophen and/or ibuprofen in controlling acute pain for patients who are not dependent on opioids for the management of chronic pain. Our experience in caring for many surgical patients has shown that patients are usually receptive to the use of nonopioid analgesics as many are familiar with their addictive potential because of the uptick in the publicity of the opioid-related complications.”
In cases where opioids might be appropriate, the panel unanimously agreed that dose limits be imposed. For 15 of the 87 procedures, the panel recommend a maximum prescription of 10 oxycodone 5-mg equivalents. Only one other – the Abbe flap – might warrant more, with a maximum of 15 oxycodone 5-mg pills at discharge.
Sometimes called a “lip switch,” the Abbe flap is reconstruction for full-thickness lip defects. It is a composite flap that moves skin, muscle, mucosa, and blood supply from the lower lip to reconstruct a defect of the upper lip. This reconstruction attempts to respect the native anatomic landmarks of the lip and allow for a better functional outcome.
“Because of the extensive nature of the repair and the anatomic territories that are manipulated, including the suturing of the lower lip to the upper lip with delayed separation, adequate pain control may require opioid analgesics in the immediate postoperative period,” the team wrote in the interview.
The panel could not agree on pain management strategies for five other procedures: Karapandzic flaps, en bloc nail excisions, facial resurfacing with deep chemical peels, and small- or large-volume liposuction. This was partly because of a lack of personal experience. Only 8 of the 40 panelists performed Karapandzic flaps. The maximum number of 5-mg oxycodone tablets any panelist prescribed for Karapandzic flaps and en bloc nail excisions was 20.
Facial resurfacing was likewise an uncommon procedure for the panel, with just 11 members performing this using deep chemical peels. However, five of those panelists said that opioids were routinely needed for postoperative pain with a maximum of 15 oxycodone 5-mg equivalents. And just four panelists performed liposuction, for which they used a maximum of 15 oxycodone 5-mg equivalents.
“However,” they wrote in the guidelines, “these providers noted that the location where the procedure is performed strongly influences the need for opioid pain management, with small-volume removal in the neck, arms, or flanks being unlikely to require opioids for adequate pain control, whereas large-volume removal in the thighs, knees, and hips may routinely require opioids.”
Addressing patient expectations is a very important part of pain management, the panel noted. “Patients will invariably experience postoperative pain after cutaneous surgeries or other interventions, often peaking within 4 hours after surgery. Wound tension, size and type of repair, anatomical location/nerve innervation, and patient pain tolerance are all factors that contribute to postoperative discomfort and should be considered when developing a postoperative pain management plan.”
Ultimately, according to Dr. McLawhorn and coauthors, the decision to use opioids at discharge for postoperative pain control should be an individual one based on patients’ comorbidities and expectations.
“Admittedly, many of the procedures listed within the recommendations may result in a rather large or complex defect that requires an equally large or complex repair,” they wrote in the interview. “However, proper education of the patient and provider regarding the risks of addiction with the use of opioids even short term should be discussed as part of every preoperative consultation. Furthermore, the patient and the provider must discuss their expectations for postoperative pain interventions for adequate pain control.”
SOURCE: McLawhorn J et al. J Am Acad Dermatol. 2019 Nov 12. doi: 10.1016/j.jaad.2019.09.080.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Bariatric surgery should be considered in individuals with class 1 obesity
Mitchel L. Zoler’s article on Abstract A105, presented at Obesity Week 2019, addresses an important health concern and is timely.
Over the past 4 decades we have seen a rise in the prevalence of obesity and associated health complications, not just in the United States but across the world. The incidence of obesity (having a BMI greater than 30) was 35% for women and 31% for men in the United States, and associated deaths and disability were primarily attributed to diabetes and cardiovascular disease resulting from obesity.
This article references the benefits of bariatric/metabolic surgery in individuals with class 1 obesity. In the United States, more than half of those who meet the criteria for obesity come under the class 1 category (BMI, 30-34.9). Those in this class of obesity are at increased risk of developing diabetes, hypertension, hyperlipidemia, coronary artery disease, cerebrovascular disease, obstructive sleep apnea, polycystic ovarian syndrome, and bone and joint disorders.
There are several studies that document the significant reduction in incidence of the above cardiometabolic risks with sustained weight loss. Nonsurgical interventions in individuals with class 1 obesity through lifestyle modifications and pharmacotherapy have not demonstrated success in providing persistent weight loss or metabolic benefits. The data presented in this article are of great significance to patients and physicians alike as they highlight the long-term benefits and reversal of metabolic disorders.
Current guidelines for bariatric surgery for individuals with a BMI greater than 35 were published in 1991. Since then several safe surgical options including laparoscopic procedures, sleeve gastrectomy, and adjustable gastric banding have been developed with decreased surgical risks, morbidity, and mortality.
The International Federation for the Surgery of Obesity and Metabolic Disorders, the International Diabetes Federation, and the National Institute for Health and Care Excellence of the United Kingdom, have supported the option of bariatric surgery in class 1 obese individuals with metabolic disorders.
While lifestyle modifications with medications should be the first-line treatment for class 1 obesity, as a primary care physician I believe that, given the major changes in the surgical options, the proven long-term benefits, and the rising incidences of obesity and metabolic syndrome, it is time for the health care community, insurers, patients, and all other stakeholders to consider bariatric surgery in class 1 obese individuals as a potential and viable option.
Noel N. Deep, MD, is a general internist in a multispecialty group practice with Aspirus Antigo (Wis.) Clinic and the chief medical officer and a staff physician at Aspirus Langlade Hospital in Antigo. He is also assistant clinical professor at the Medical College of Wisconsin, Central Wisconsin Campus, Wausau, and the governor of the Wisconsin chapter of the American College of Physicians. Dr. Deep serves on the editorial advisory board of Internal Medicine News.
He made these comments in response to questions from MDedge and had no relevant disclosures.
Mitchel L. Zoler’s article on Abstract A105, presented at Obesity Week 2019, addresses an important health concern and is timely.
Over the past 4 decades we have seen a rise in the prevalence of obesity and associated health complications, not just in the United States but across the world. The incidence of obesity (having a BMI greater than 30) was 35% for women and 31% for men in the United States, and associated deaths and disability were primarily attributed to diabetes and cardiovascular disease resulting from obesity.
This article references the benefits of bariatric/metabolic surgery in individuals with class 1 obesity. In the United States, more than half of those who meet the criteria for obesity come under the class 1 category (BMI, 30-34.9). Those in this class of obesity are at increased risk of developing diabetes, hypertension, hyperlipidemia, coronary artery disease, cerebrovascular disease, obstructive sleep apnea, polycystic ovarian syndrome, and bone and joint disorders.
There are several studies that document the significant reduction in incidence of the above cardiometabolic risks with sustained weight loss. Nonsurgical interventions in individuals with class 1 obesity through lifestyle modifications and pharmacotherapy have not demonstrated success in providing persistent weight loss or metabolic benefits. The data presented in this article are of great significance to patients and physicians alike as they highlight the long-term benefits and reversal of metabolic disorders.
Current guidelines for bariatric surgery for individuals with a BMI greater than 35 were published in 1991. Since then several safe surgical options including laparoscopic procedures, sleeve gastrectomy, and adjustable gastric banding have been developed with decreased surgical risks, morbidity, and mortality.
The International Federation for the Surgery of Obesity and Metabolic Disorders, the International Diabetes Federation, and the National Institute for Health and Care Excellence of the United Kingdom, have supported the option of bariatric surgery in class 1 obese individuals with metabolic disorders.
While lifestyle modifications with medications should be the first-line treatment for class 1 obesity, as a primary care physician I believe that, given the major changes in the surgical options, the proven long-term benefits, and the rising incidences of obesity and metabolic syndrome, it is time for the health care community, insurers, patients, and all other stakeholders to consider bariatric surgery in class 1 obese individuals as a potential and viable option.
Noel N. Deep, MD, is a general internist in a multispecialty group practice with Aspirus Antigo (Wis.) Clinic and the chief medical officer and a staff physician at Aspirus Langlade Hospital in Antigo. He is also assistant clinical professor at the Medical College of Wisconsin, Central Wisconsin Campus, Wausau, and the governor of the Wisconsin chapter of the American College of Physicians. Dr. Deep serves on the editorial advisory board of Internal Medicine News.
He made these comments in response to questions from MDedge and had no relevant disclosures.
Mitchel L. Zoler’s article on Abstract A105, presented at Obesity Week 2019, addresses an important health concern and is timely.
Over the past 4 decades we have seen a rise in the prevalence of obesity and associated health complications, not just in the United States but across the world. The incidence of obesity (having a BMI greater than 30) was 35% for women and 31% for men in the United States, and associated deaths and disability were primarily attributed to diabetes and cardiovascular disease resulting from obesity.
This article references the benefits of bariatric/metabolic surgery in individuals with class 1 obesity. In the United States, more than half of those who meet the criteria for obesity come under the class 1 category (BMI, 30-34.9). Those in this class of obesity are at increased risk of developing diabetes, hypertension, hyperlipidemia, coronary artery disease, cerebrovascular disease, obstructive sleep apnea, polycystic ovarian syndrome, and bone and joint disorders.
There are several studies that document the significant reduction in incidence of the above cardiometabolic risks with sustained weight loss. Nonsurgical interventions in individuals with class 1 obesity through lifestyle modifications and pharmacotherapy have not demonstrated success in providing persistent weight loss or metabolic benefits. The data presented in this article are of great significance to patients and physicians alike as they highlight the long-term benefits and reversal of metabolic disorders.
Current guidelines for bariatric surgery for individuals with a BMI greater than 35 were published in 1991. Since then several safe surgical options including laparoscopic procedures, sleeve gastrectomy, and adjustable gastric banding have been developed with decreased surgical risks, morbidity, and mortality.
The International Federation for the Surgery of Obesity and Metabolic Disorders, the International Diabetes Federation, and the National Institute for Health and Care Excellence of the United Kingdom, have supported the option of bariatric surgery in class 1 obese individuals with metabolic disorders.
While lifestyle modifications with medications should be the first-line treatment for class 1 obesity, as a primary care physician I believe that, given the major changes in the surgical options, the proven long-term benefits, and the rising incidences of obesity and metabolic syndrome, it is time for the health care community, insurers, patients, and all other stakeholders to consider bariatric surgery in class 1 obese individuals as a potential and viable option.
Noel N. Deep, MD, is a general internist in a multispecialty group practice with Aspirus Antigo (Wis.) Clinic and the chief medical officer and a staff physician at Aspirus Langlade Hospital in Antigo. He is also assistant clinical professor at the Medical College of Wisconsin, Central Wisconsin Campus, Wausau, and the governor of the Wisconsin chapter of the American College of Physicians. Dr. Deep serves on the editorial advisory board of Internal Medicine News.
He made these comments in response to questions from MDedge and had no relevant disclosures.
FDA expands use of Toujeo to childhood type 1 diabetes
The Food and Drug Administration has expanded the indication for Toujeo (insulin glargine 300 units/mL injection; Sanofi) to include children as young as 6 years of age with type 1 diabetes.
The FDA first approved Toujeo in 2015 for adults with type 1 and type 2 diabetes, designed as a more potent follow-up to Sanofi’s top-selling insulin glargine (Lantus).
Last month, Sanofi reported positive results from the phase 3 EDITION JUNIOR trial of Toujeo in children and adolescents with type 1 diabetes. These were presented at the International Society for Pediatric and Adolescent Diabetes 45th Annual Conference in Boston.
In the trial, 463 children and adolescents (aged 6-17 years) treated for type 1 diabetes for at least 1 year and with A1c between 7.5% and 11.0% at screening were randomized to Toujeo or insulin glargine 100 units/mL (Gla-100); participants continued to take their existing mealtime insulin.
The primary endpoint was noninferior reduction in A1c after 26 weeks.
The study met its primary endpoint, confirming a noninferior reduction in A1c with Toujeo versus Gla-100 after 26 weeks (mean reduction, 0.4% vs. 0.4%; difference, 0.004%; 95% confidence interval, –0.17 to 0.18; upper bound was below the prespecified noninferiority margin of 0.3%).
Over 26 weeks, a comparable number of patients in each group experienced one or more hypoglycemic events documented at anytime over 24 hours. Numerically fewer patients taking Toujeo experienced severe hypoglycemia or experienced one or more episodes of hyperglycemia with ketosis compared with those taking Gla-100.
No unexpected safety concerns were reported based on the established profiles of both products, the company said.
In October 2019, the European Medicines Agency Committee for Medicinal Products for Human Use recommended approval of Toujeo for children age 6 years and older with diabetes.
For more diabetes and endocrinology news, follow us on Twitter and Facebook.
This story first appeared on Medscape.com.
The Food and Drug Administration has expanded the indication for Toujeo (insulin glargine 300 units/mL injection; Sanofi) to include children as young as 6 years of age with type 1 diabetes.
The FDA first approved Toujeo in 2015 for adults with type 1 and type 2 diabetes, designed as a more potent follow-up to Sanofi’s top-selling insulin glargine (Lantus).
Last month, Sanofi reported positive results from the phase 3 EDITION JUNIOR trial of Toujeo in children and adolescents with type 1 diabetes. These were presented at the International Society for Pediatric and Adolescent Diabetes 45th Annual Conference in Boston.
In the trial, 463 children and adolescents (aged 6-17 years) treated for type 1 diabetes for at least 1 year and with A1c between 7.5% and 11.0% at screening were randomized to Toujeo or insulin glargine 100 units/mL (Gla-100); participants continued to take their existing mealtime insulin.
The primary endpoint was noninferior reduction in A1c after 26 weeks.
The study met its primary endpoint, confirming a noninferior reduction in A1c with Toujeo versus Gla-100 after 26 weeks (mean reduction, 0.4% vs. 0.4%; difference, 0.004%; 95% confidence interval, –0.17 to 0.18; upper bound was below the prespecified noninferiority margin of 0.3%).
Over 26 weeks, a comparable number of patients in each group experienced one or more hypoglycemic events documented at anytime over 24 hours. Numerically fewer patients taking Toujeo experienced severe hypoglycemia or experienced one or more episodes of hyperglycemia with ketosis compared with those taking Gla-100.
No unexpected safety concerns were reported based on the established profiles of both products, the company said.
In October 2019, the European Medicines Agency Committee for Medicinal Products for Human Use recommended approval of Toujeo for children age 6 years and older with diabetes.
For more diabetes and endocrinology news, follow us on Twitter and Facebook.
This story first appeared on Medscape.com.
The Food and Drug Administration has expanded the indication for Toujeo (insulin glargine 300 units/mL injection; Sanofi) to include children as young as 6 years of age with type 1 diabetes.
The FDA first approved Toujeo in 2015 for adults with type 1 and type 2 diabetes, designed as a more potent follow-up to Sanofi’s top-selling insulin glargine (Lantus).
Last month, Sanofi reported positive results from the phase 3 EDITION JUNIOR trial of Toujeo in children and adolescents with type 1 diabetes. These were presented at the International Society for Pediatric and Adolescent Diabetes 45th Annual Conference in Boston.
In the trial, 463 children and adolescents (aged 6-17 years) treated for type 1 diabetes for at least 1 year and with A1c between 7.5% and 11.0% at screening were randomized to Toujeo or insulin glargine 100 units/mL (Gla-100); participants continued to take their existing mealtime insulin.
The primary endpoint was noninferior reduction in A1c after 26 weeks.
The study met its primary endpoint, confirming a noninferior reduction in A1c with Toujeo versus Gla-100 after 26 weeks (mean reduction, 0.4% vs. 0.4%; difference, 0.004%; 95% confidence interval, –0.17 to 0.18; upper bound was below the prespecified noninferiority margin of 0.3%).
Over 26 weeks, a comparable number of patients in each group experienced one or more hypoglycemic events documented at anytime over 24 hours. Numerically fewer patients taking Toujeo experienced severe hypoglycemia or experienced one or more episodes of hyperglycemia with ketosis compared with those taking Gla-100.
No unexpected safety concerns were reported based on the established profiles of both products, the company said.
In October 2019, the European Medicines Agency Committee for Medicinal Products for Human Use recommended approval of Toujeo for children age 6 years and older with diabetes.
For more diabetes and endocrinology news, follow us on Twitter and Facebook.
This story first appeared on Medscape.com.
Dual vs Triple Therapy Following ACS or PCI in Patients with Atrial Fibrillation
Study Overview
Objective. To compare the benefit of apixaban with a vitamin K antagonist and compare aspirin with placebo in patients with atrial fibrillation who had acute coronary syndrome or underwent percutaneous coronary intervention (PCI) and were planning to take a P2Y12 inhibitor.
Design. Multicenter, international, open-label, prospective randomized controlled trial with a 2-by-2 factorial design.
Setting and participants. 4614 patients who had an acute coronary syndrome or had undergone PCI and were planning to take a P2Y12 inhibitor.
Intervention. Patients were assigned by means of an interactive voice-response system to receive apixaban or a vitamin K antagonist and to receive aspirin or matching placebo for 6 months.
Main outcome measures. The primary outcome was major or clinically relevant nonmajor bleeding. Secondary outcomes included death or hospitalization and a composite of ischemic events.
Main results. At 6 months, major or clinically relevant nonmajor bleeding had occurred in 10.5% of the patients receiving apixaban, as compared to 14.7% of those receiving a vitamin K antagonist (hazard ratio [HR], 0.69; 95% confidence interval [CI], 0.58-0.81, P < 0.001 for both noninferiority and superiority), and in 16.1% of the patients receiving aspirin, as compared with 9.0% of those receiving placebo (HR 1.89; 95% CI, 1.59-2.24; P < 0.001). Patients in the apixaban group had a lower incidence of death or hospitalization than those in the vitamin K antagonist group (23.5% versus 27.4%; HR 0.83; 95% CI, 0.74-0.93; P = 0.002) and similar incidence of ischemic events.
Conclusion. Among patients with atrial fibrillation and recent acute coronary syndrome or PCI treated with a P2Y12 inhibitor, an antithrombotic regimen that included apixaban without aspirin resulted in less bleeding and fewer hospitalizations without significant differences in the incidence of ischemic events than the regimens that included a vitamin K antagonist, aspirin, or both.
Commentary
PCI is performed in about 20% of patients with atrial fibrillation. These patients require dual antiplatelet therapy to prevent ischemic events, combined with long-term anticoagulation to prevent stroke due to atrial fibrillation. Because the combination of anticoagulation and antiplatelet therapy is associated with a higher risk of bleeding, balancing the risk and benefit of dual antiplatelet therapy and anticoagulation in this population is crucial.
Previous studies have assessed the risk and benefit associated with anticoagulation and antiplatelet therapy. When warfarin plus clopidogrel (double therapy) was compared with warfarin, aspirin, and clopidogrel (triple therapy) in patients with acute coronary syndromes and stable ischemic coronary disease undergoing PCI, use of clopidogrel without aspirin (double therapy) was associated with a significant reduction in bleeding complications (19.4% versus 44.4%, HR, 0.36; 95% CI, 0.26-0.20; P < 0.0001) without increasing thrombotic events.1 Recent studies have compared triple therapy with warfarin to double therapy using direct oral anticoagulants (DOAC). The PIONEER AF-PCI study, which compared low-dose rivaroxaban (15 mg once daily) plus a P2Y12 inhibitor to vitamin K antagonist plus dual antiplatelet therapy, found that the rates of clinically significant bleeding were lower in the low-dose rivaroxaban group compared to the triple-therapy group with a vitamin K antagonist (16.8% versus 26.7%; HR, 0.59; 95% CI, 0.47-0.76; P < 0.001).2 Similarly, the RE-DUAL PCI studied dabigatran and showed that the dual therapy group with dabigatran had a lower incidence of major or clinically relevant nonmajor bleeding events during follow-up compared to triple therapy including a vitamin K antagonist (15.4% versus 26.9%; HR, 0.52; 95% CI, 0.42-0.63; P < 0.001).3
In this context, Lopes at al investigated the clinical question of dual therapy versus triple therapy by performing a well-designed randomized clinical trial. In this trial with a 2-by-2 factorial design, the authors studied the effect of apixaban compared to vitamin K antagonist and the effect of aspirin compared to placebo. Major or clinically relevant nonmajor bleeding occurred in 10.5% of patients receiving apixaban, as compared to 14.7% of those receiving a vitamin K antagonist (HR 0.69; 95% CI, 0.58-0.81; P < 0.001). The incidence of major or clinically relevant nonmajor bleeding was higher in patients receiving aspirin than in those receiving placebo (16.1% versus 9.0%; HR, 1.89; 95% CI, 1.59-2.24; P < 0.001). Patients in the apixaban group had a lower incidence of death or hospitalization than those in the vitamin K antagonist group (23.5% versus 27.4%; HR, 0.83; 95% CI, 0.74-0.93; P = 0.002). The incidence of ischemic events was similar between the apixaban group and vitamin K antagonist group and between the aspirin group and placebo group.
The strengths of this current study include the large number of patients it enrolled. Taking the results from the PIONEER-AF, RE-DUAL PCI, and AUGUSTUS trials, it is clear that DOAC reduces the risk of bleeding compared to vitamin K antagonist. In addition, the AUGUSTUS trial was the first that evaluated the effect of aspirin in patients treated with DOAC and antiplatelet therapy. Aspirin was associated with increased risk of bleeding, with a similar rate of ischemic events compared to placebo.
The AUGUSTUS trial has several limitations. Although the incidence of ischemic events was similar between the apixaban group and the vitamin K antagonist group, the study was not powered to evaluate for individual ischemic outcomes. However, there was no clear evidence of an increase in harm. Since more than 90% of P2Y12 inhibitors used were clopidogrel, the safety and efficacy of combining apixaban with ticagrelor or prasugrel will require further study.
Applications for Clinical Practice
In patients with atrial fibrillation and a recent acute coronary syndrome or PCI treated with a P2Y12 inhibitor, dual therapy with a P2Y12 inhibitor and DOAC should be favored over a regimen that includes a vitamin K antagonist and/or aspirin.
—Taishi Hirai, MD, University of Missouri Medical Center, and John Blair, MD, University of Chicago Medical Center
1. Dewilde WJM, Oirbans T, Verheugt FWA, et al. Use of clopidogrel with or without aspirin in patients taking oral anticoagulant therapy and undergoing percutaneous coronary intervention: an open-label, randomised, controlled trial. Lancet. 2013;381(9872):1107-1115.
2. Gibson CM, Mehran R, Bode C, et al. Prevention of bleeding in patients with atrial fibrillation undergoing PCI. N Engl J Med. 2016;375:2423-2434.
3. Cannon CP, Bhatt DL, Oldgren J, et al. Dual antithrombotic therapy with dabigatran after PCI in atrial fibrillation. N Engl J Med. 2017;377:1513-1524.
Study Overview
Objective. To compare the benefit of apixaban with a vitamin K antagonist and compare aspirin with placebo in patients with atrial fibrillation who had acute coronary syndrome or underwent percutaneous coronary intervention (PCI) and were planning to take a P2Y12 inhibitor.
Design. Multicenter, international, open-label, prospective randomized controlled trial with a 2-by-2 factorial design.
Setting and participants. 4614 patients who had an acute coronary syndrome or had undergone PCI and were planning to take a P2Y12 inhibitor.
Intervention. Patients were assigned by means of an interactive voice-response system to receive apixaban or a vitamin K antagonist and to receive aspirin or matching placebo for 6 months.
Main outcome measures. The primary outcome was major or clinically relevant nonmajor bleeding. Secondary outcomes included death or hospitalization and a composite of ischemic events.
Main results. At 6 months, major or clinically relevant nonmajor bleeding had occurred in 10.5% of the patients receiving apixaban, as compared to 14.7% of those receiving a vitamin K antagonist (hazard ratio [HR], 0.69; 95% confidence interval [CI], 0.58-0.81, P < 0.001 for both noninferiority and superiority), and in 16.1% of the patients receiving aspirin, as compared with 9.0% of those receiving placebo (HR 1.89; 95% CI, 1.59-2.24; P < 0.001). Patients in the apixaban group had a lower incidence of death or hospitalization than those in the vitamin K antagonist group (23.5% versus 27.4%; HR 0.83; 95% CI, 0.74-0.93; P = 0.002) and similar incidence of ischemic events.
Conclusion. Among patients with atrial fibrillation and recent acute coronary syndrome or PCI treated with a P2Y12 inhibitor, an antithrombotic regimen that included apixaban without aspirin resulted in less bleeding and fewer hospitalizations without significant differences in the incidence of ischemic events than the regimens that included a vitamin K antagonist, aspirin, or both.
Commentary
PCI is performed in about 20% of patients with atrial fibrillation. These patients require dual antiplatelet therapy to prevent ischemic events, combined with long-term anticoagulation to prevent stroke due to atrial fibrillation. Because the combination of anticoagulation and antiplatelet therapy is associated with a higher risk of bleeding, balancing the risk and benefit of dual antiplatelet therapy and anticoagulation in this population is crucial.
Previous studies have assessed the risk and benefit associated with anticoagulation and antiplatelet therapy. When warfarin plus clopidogrel (double therapy) was compared with warfarin, aspirin, and clopidogrel (triple therapy) in patients with acute coronary syndromes and stable ischemic coronary disease undergoing PCI, use of clopidogrel without aspirin (double therapy) was associated with a significant reduction in bleeding complications (19.4% versus 44.4%, HR, 0.36; 95% CI, 0.26-0.20; P < 0.0001) without increasing thrombotic events.1 Recent studies have compared triple therapy with warfarin to double therapy using direct oral anticoagulants (DOAC). The PIONEER AF-PCI study, which compared low-dose rivaroxaban (15 mg once daily) plus a P2Y12 inhibitor to vitamin K antagonist plus dual antiplatelet therapy, found that the rates of clinically significant bleeding were lower in the low-dose rivaroxaban group compared to the triple-therapy group with a vitamin K antagonist (16.8% versus 26.7%; HR, 0.59; 95% CI, 0.47-0.76; P < 0.001).2 Similarly, the RE-DUAL PCI studied dabigatran and showed that the dual therapy group with dabigatran had a lower incidence of major or clinically relevant nonmajor bleeding events during follow-up compared to triple therapy including a vitamin K antagonist (15.4% versus 26.9%; HR, 0.52; 95% CI, 0.42-0.63; P < 0.001).3
In this context, Lopes at al investigated the clinical question of dual therapy versus triple therapy by performing a well-designed randomized clinical trial. In this trial with a 2-by-2 factorial design, the authors studied the effect of apixaban compared to vitamin K antagonist and the effect of aspirin compared to placebo. Major or clinically relevant nonmajor bleeding occurred in 10.5% of patients receiving apixaban, as compared to 14.7% of those receiving a vitamin K antagonist (HR 0.69; 95% CI, 0.58-0.81; P < 0.001). The incidence of major or clinically relevant nonmajor bleeding was higher in patients receiving aspirin than in those receiving placebo (16.1% versus 9.0%; HR, 1.89; 95% CI, 1.59-2.24; P < 0.001). Patients in the apixaban group had a lower incidence of death or hospitalization than those in the vitamin K antagonist group (23.5% versus 27.4%; HR, 0.83; 95% CI, 0.74-0.93; P = 0.002). The incidence of ischemic events was similar between the apixaban group and vitamin K antagonist group and between the aspirin group and placebo group.
The strengths of this current study include the large number of patients it enrolled. Taking the results from the PIONEER-AF, RE-DUAL PCI, and AUGUSTUS trials, it is clear that DOAC reduces the risk of bleeding compared to vitamin K antagonist. In addition, the AUGUSTUS trial was the first that evaluated the effect of aspirin in patients treated with DOAC and antiplatelet therapy. Aspirin was associated with increased risk of bleeding, with a similar rate of ischemic events compared to placebo.
The AUGUSTUS trial has several limitations. Although the incidence of ischemic events was similar between the apixaban group and the vitamin K antagonist group, the study was not powered to evaluate for individual ischemic outcomes. However, there was no clear evidence of an increase in harm. Since more than 90% of P2Y12 inhibitors used were clopidogrel, the safety and efficacy of combining apixaban with ticagrelor or prasugrel will require further study.
Applications for Clinical Practice
In patients with atrial fibrillation and a recent acute coronary syndrome or PCI treated with a P2Y12 inhibitor, dual therapy with a P2Y12 inhibitor and DOAC should be favored over a regimen that includes a vitamin K antagonist and/or aspirin.
—Taishi Hirai, MD, University of Missouri Medical Center, and John Blair, MD, University of Chicago Medical Center
Study Overview
Objective. To compare the benefit of apixaban with a vitamin K antagonist and compare aspirin with placebo in patients with atrial fibrillation who had acute coronary syndrome or underwent percutaneous coronary intervention (PCI) and were planning to take a P2Y12 inhibitor.
Design. Multicenter, international, open-label, prospective randomized controlled trial with a 2-by-2 factorial design.
Setting and participants. 4614 patients who had an acute coronary syndrome or had undergone PCI and were planning to take a P2Y12 inhibitor.
Intervention. Patients were assigned by means of an interactive voice-response system to receive apixaban or a vitamin K antagonist and to receive aspirin or matching placebo for 6 months.
Main outcome measures. The primary outcome was major or clinically relevant nonmajor bleeding. Secondary outcomes included death or hospitalization and a composite of ischemic events.
Main results. At 6 months, major or clinically relevant nonmajor bleeding had occurred in 10.5% of the patients receiving apixaban, as compared to 14.7% of those receiving a vitamin K antagonist (hazard ratio [HR], 0.69; 95% confidence interval [CI], 0.58-0.81, P < 0.001 for both noninferiority and superiority), and in 16.1% of the patients receiving aspirin, as compared with 9.0% of those receiving placebo (HR 1.89; 95% CI, 1.59-2.24; P < 0.001). Patients in the apixaban group had a lower incidence of death or hospitalization than those in the vitamin K antagonist group (23.5% versus 27.4%; HR 0.83; 95% CI, 0.74-0.93; P = 0.002) and similar incidence of ischemic events.
Conclusion. Among patients with atrial fibrillation and recent acute coronary syndrome or PCI treated with a P2Y12 inhibitor, an antithrombotic regimen that included apixaban without aspirin resulted in less bleeding and fewer hospitalizations without significant differences in the incidence of ischemic events than the regimens that included a vitamin K antagonist, aspirin, or both.
Commentary
PCI is performed in about 20% of patients with atrial fibrillation. These patients require dual antiplatelet therapy to prevent ischemic events, combined with long-term anticoagulation to prevent stroke due to atrial fibrillation. Because the combination of anticoagulation and antiplatelet therapy is associated with a higher risk of bleeding, balancing the risk and benefit of dual antiplatelet therapy and anticoagulation in this population is crucial.
Previous studies have assessed the risk and benefit associated with anticoagulation and antiplatelet therapy. When warfarin plus clopidogrel (double therapy) was compared with warfarin, aspirin, and clopidogrel (triple therapy) in patients with acute coronary syndromes and stable ischemic coronary disease undergoing PCI, use of clopidogrel without aspirin (double therapy) was associated with a significant reduction in bleeding complications (19.4% versus 44.4%, HR, 0.36; 95% CI, 0.26-0.20; P < 0.0001) without increasing thrombotic events.1 Recent studies have compared triple therapy with warfarin to double therapy using direct oral anticoagulants (DOAC). The PIONEER AF-PCI study, which compared low-dose rivaroxaban (15 mg once daily) plus a P2Y12 inhibitor to vitamin K antagonist plus dual antiplatelet therapy, found that the rates of clinically significant bleeding were lower in the low-dose rivaroxaban group compared to the triple-therapy group with a vitamin K antagonist (16.8% versus 26.7%; HR, 0.59; 95% CI, 0.47-0.76; P < 0.001).2 Similarly, the RE-DUAL PCI studied dabigatran and showed that the dual therapy group with dabigatran had a lower incidence of major or clinically relevant nonmajor bleeding events during follow-up compared to triple therapy including a vitamin K antagonist (15.4% versus 26.9%; HR, 0.52; 95% CI, 0.42-0.63; P < 0.001).3
In this context, Lopes at al investigated the clinical question of dual therapy versus triple therapy by performing a well-designed randomized clinical trial. In this trial with a 2-by-2 factorial design, the authors studied the effect of apixaban compared to vitamin K antagonist and the effect of aspirin compared to placebo. Major or clinically relevant nonmajor bleeding occurred in 10.5% of patients receiving apixaban, as compared to 14.7% of those receiving a vitamin K antagonist (HR 0.69; 95% CI, 0.58-0.81; P < 0.001). The incidence of major or clinically relevant nonmajor bleeding was higher in patients receiving aspirin than in those receiving placebo (16.1% versus 9.0%; HR, 1.89; 95% CI, 1.59-2.24; P < 0.001). Patients in the apixaban group had a lower incidence of death or hospitalization than those in the vitamin K antagonist group (23.5% versus 27.4%; HR, 0.83; 95% CI, 0.74-0.93; P = 0.002). The incidence of ischemic events was similar between the apixaban group and vitamin K antagonist group and between the aspirin group and placebo group.
The strengths of this current study include the large number of patients it enrolled. Taking the results from the PIONEER-AF, RE-DUAL PCI, and AUGUSTUS trials, it is clear that DOAC reduces the risk of bleeding compared to vitamin K antagonist. In addition, the AUGUSTUS trial was the first that evaluated the effect of aspirin in patients treated with DOAC and antiplatelet therapy. Aspirin was associated with increased risk of bleeding, with a similar rate of ischemic events compared to placebo.
The AUGUSTUS trial has several limitations. Although the incidence of ischemic events was similar between the apixaban group and the vitamin K antagonist group, the study was not powered to evaluate for individual ischemic outcomes. However, there was no clear evidence of an increase in harm. Since more than 90% of P2Y12 inhibitors used were clopidogrel, the safety and efficacy of combining apixaban with ticagrelor or prasugrel will require further study.
Applications for Clinical Practice
In patients with atrial fibrillation and a recent acute coronary syndrome or PCI treated with a P2Y12 inhibitor, dual therapy with a P2Y12 inhibitor and DOAC should be favored over a regimen that includes a vitamin K antagonist and/or aspirin.
—Taishi Hirai, MD, University of Missouri Medical Center, and John Blair, MD, University of Chicago Medical Center
1. Dewilde WJM, Oirbans T, Verheugt FWA, et al. Use of clopidogrel with or without aspirin in patients taking oral anticoagulant therapy and undergoing percutaneous coronary intervention: an open-label, randomised, controlled trial. Lancet. 2013;381(9872):1107-1115.
2. Gibson CM, Mehran R, Bode C, et al. Prevention of bleeding in patients with atrial fibrillation undergoing PCI. N Engl J Med. 2016;375:2423-2434.
3. Cannon CP, Bhatt DL, Oldgren J, et al. Dual antithrombotic therapy with dabigatran after PCI in atrial fibrillation. N Engl J Med. 2017;377:1513-1524.
1. Dewilde WJM, Oirbans T, Verheugt FWA, et al. Use of clopidogrel with or without aspirin in patients taking oral anticoagulant therapy and undergoing percutaneous coronary intervention: an open-label, randomised, controlled trial. Lancet. 2013;381(9872):1107-1115.
2. Gibson CM, Mehran R, Bode C, et al. Prevention of bleeding in patients with atrial fibrillation undergoing PCI. N Engl J Med. 2016;375:2423-2434.
3. Cannon CP, Bhatt DL, Oldgren J, et al. Dual antithrombotic therapy with dabigatran after PCI in atrial fibrillation. N Engl J Med. 2017;377:1513-1524.
Patient-Reported Outcomes in Multiple Sclerosis: An Overview
From the Dartmouth Institute for Health Policy & Clinical Practice, Geisel School of Medicine, Hanover, NH (Ms. Manohar and Dr. Oliver), the Department of Community & Family Medicine, Dartmouth-Hitchcock Medical Center, Lebanon, NH (Ms. Perkins, Ms. Laurion, and Dr. Oliver), and the Multiple Sclerosis Specialty Care Program, Concord Hospital, Concord, NH (Dr. Oliver).
Abstract
- Background: Patient-reported outcomes (PROs), including patient-reported outcome measures (PROMs) and patient-reported experience measures (PREMs), can be used to assess perceived health status, functioning, quality of life, and experience of care. Complex chronic illnesses such as multiple sclerosis (MS) affect multiple aspects of health, and PROs can be applied in assessment and decision-making in MS care as well as in research pertaining to MS.
- Objective: To provide a general review of PROs, with a specific focus on implications for MS care.
- Methods: Evidence synthesis of available literature on PROs in MS care.
- Results: PROs (including PROMs and PREMs) have historically been utilized in research and are now being applied in clinical, improvement, and population health settings using learning health system approaches in many disease populations, including MS. Many challenges complicate the use of PROs in MS care, including reliability, validity, and interpretability of PROMs, as well as feasibility barriers due to time and financial constraints in clinical settings.
- Conclusion: PROs have the potential to better inform clinical care, empower patient-centered care, inform health care improvement efforts, and create the conditions for coproduction of health care services.
Keywords: PRO; PROM; patient-reported outcome measure; patient-reported experience measure; quality of life; patient-centered care.
Multiple sclerosis (MS) is a disabling, complex, chronic, immune-mediated disorder of the central nervous system (CNS). MS causes inflammatory and degenerative damage in the CNS, which disrupts signaling pathways.1 It is most commonly diagnosed in young adults and affects 2.3 million people worldwide.2 People with MS experience very different disease courses and a wide range of neurological symptoms, including visual, somatic, mental health, sensory, motor, and cognitive problems.1-3 Relapsing-remitting MS, the most common form, affects 85% of those with MS and is characterized by periods of relapse (exacerbation) and remission.1 Other forms of MS (primary progressive and secondary progressive MS) are characterized by progressive deterioration and worsening symptom severity without exacerbations. Disease-modifying therapies (DMTs) can reduce the frequency of exacerbations and disability progression, but unfortunately there is no cure for MS. Treatment is focused on increasing quality of life, minimizing disability, and maximizing wellness.
Patient-reported outcomes (PROs) describe the perceived health status, function, and/or experience of a person as obtained by direct self-report. Patient-reported outcome measures (PROMs) are validated PROs that can be used to inform clinical care,4 and have demonstrated effectiveness in improving patient-provider communication and decision-making.5-7 PROMs are currently used in some MS clinical trials to determine the impact of experimental interventions,8-10 and are also being used to inform and improve clinical care in some settings. Especially for persons with MS, they can provide individualized perspectives about health experience and outcomes.11 In more advanced applications, PROMs can be used to improve face-to-face collaborations between clinicians and patients and to inform patient-centered systems of care.12-14 PROMs can also be used to inform systems-level improvement for entire patient populations.15,16
In this article, we review current applications of PROs and PROMs in the care of persons with MS, as well as current limitations and barriers to their use.
At a recent visit to her neurologist, Marion reviews her health diary, in which she has been tracking her fatigue levels throughout the day and when she has to visit the bathroom. The PRO diary also helps her remember details that she might not otherwise be able to recall at the time of her clinic visit. They review the diary entrees together to develop a shared understanding of what Marion has been experiencing and identify trends in the PRO data. They discuss symptom management and use the PRO information from the diary to help guide adjustments to her physical therapy routine and medication regimen.
Part of Marion’s “PRO package” includes the Center for Epidemiologic Studies Depression Scale (CES-D), a validated depression screening and symptom severity questionnaire that she completes every 3 months. Although she denies being depressed, she has noticed that her CES-D scores in recent months have been consistently increasing. This prompts a discussion about mental health in MS and a referral to work on depression with the MS mental health specialist. Marion and the mental health specialist use CES-D measures at baseline and during treatment to set a remission target and to track progress during treatment. Marion finds this helpful because she says it is hard for her to “wrap my hands around depression… it’s not something that there is a blood test or a MRI for.” Marion is encouraged by being able to see her CES-D scores change as her depression severity decreases, and this helps motivate her to keep engaged in treatment.
PROs and PROMs: General Applications
PROs are measures obtained directly from an individual without a priori interpretation by a clinician.9,17 PROs capture individual perspectives on symptoms, capability, disability, and health-related quality of life.9 With increasing emphasis on patient-centered care,18 individual perspectives and preferences elicited using PROMs may be able to inform better quality of care and patient-centered disease treatment and management.19-21
PROMs are standardized, validated questionnaires used to assess PROs and can be generic or condition-specific. Generic PROMs can be used in any patient population. The SF-3622 is a set of quality of life measures that assess perceived ability to complete physical tasks and routine activities, general health status, fatigue, social functioning, pain, and emotional and mental health.23 Condition-specific PROMs can be used for particular patient populations and are helpful in identifying changes in health status for a specific disease, disability, or surgery. For example, the PDQ-39 assesses 8 dimensions of daily living, functioning, and well-being for people with Parkinson’s disease.24
PROMs have been used in some MS clinical trials and research studies to determine the effectiveness of experimental treatments from the viewpoint of study participants.9,25,26 PROs can also be utilized in clinical care to facilitate communication of needs and track health outcomes,27 and can inform improvement in outcomes for health systems and populations. They can also be used to assess experience of care,28 encouraging a focus on high-quality outcomes through PRO-connected reimbursement mechanisms,29 and provide aggregate data to evaluate clinical practice, population health outcomes, and the effectiveness of public policies.27
Patient-reported experience measures (PREMs) assess patient satisfaction and experience of health care.30,31 CollaboRATE32 is a PREM that assesses the degree of shared decision-making occurring between patients and clinicians during clinical care. PREMs are currently used for assessing self-efficacy and in shared decision-making and health care improvement applications. PREMs have yet to be developed specifically for persons with MS.
PROMs in MS Care
Generic PROMs have shown that persons with MS are disproportionately burdened by poor quality of life.33-35 Other generic PROMs, like the SF-36,36 the Sickness Impact Profile,37 and versions of the Health Utilities Index,38 can be used to gather information on dysfunction and to determine quality and duration of life modified by MS-related dysfunction and disability. MS-specific PROMs are used to assess MS impairments, including pain, fatigue, cognition, sexual dysfunction, and depression.12,39-42 PROMs have also been used in MS clinical trials, including the Multiple Sclerosis Impact Scale-29 (MSIS-29),43,44 the Leeds MS QoL (LMSQoL),45,46 the Functional Assessment of MS (FAMS),47 the Hamburg Quality of Life Questionnaire in MS (HAQUAMS),48 the MS Quality of Life-54 (MSQoL-54),49 the MS International QoL (MUSIQoL),50 and the Patient-Reported Indices for MS Activity Limitations Scale (PRIMUS).51
Condition-specific PROMs are more sensitive to changes in health status and functioning for persons with MS compared to generic PROMs. They are also more reliable during MS remission and relapse periods.44,52 For example, the SF-36 has floor and ceiling effects in MS populations—a high proportion of persons with MS are scored at the maximum or minimum levels of the scale, limiting discriminant capability.22 As a result, a “combined approach” using both generic and MS-specific measures is often recommended.53 Some MS PROMs (eg, MSQoL-54) include generic questions found in the SF-36 as well as additional MS-specific questions or scales.
The variety of PROMs available (see Table for a selected listing) introduces a significant challenge to using them—limited generalizability and difficulty comparing PROs across MS studies. Efforts to establish common PROMs have been undertaken to address this problem.54 The National Institute of Neurologicical Disorders and Stroke (NINDS) sponsored the development of a neurological quality of life battery, the Neuro-QOL.55 Neuro-QOL measures the physical, mental, and social effects of neurological conditions in adults and children with neurological disorders and has the capability to facilitate comparisons across different neurological conditions. Additionally, the Patient-Reported Outcomes Measure Information System (PROMIS) has been developed to assess physical, mental, and social effects of chronic disease. PROMIS has a hybrid design that includes generic and MS-specific measures (such as PROMIS FatigueMS).56 PROMIS can be used to assess persons with MS as well as to compare the MS population with other populations with chronic illness.
PROMs have varying levels of reliability and validity. The Evaluating the Measure of Patient-Reported Outcomes57 study evaluated the development process of MS PROMs,43 and found that the MSIS-29 and LMSQoL had the highest overall reliability among the most common MS PROMs. However, both scored poorly on validity due to lack of patient involvement during development. This questions the overall capability of existing MS PROMs to accurately and consistently assess PROs in persons with MS.
“Feed-Forward” PROMs
Oliver and colleagues16 have described “feed-forward” PROM applications in MS care in a community hospital setting using a learning health system approach. This MS clinic uses feed-forward PROs to inform clinical care—PRO data are gathered before the clinic visit and analyzed ahead of or during the clinic visit by the clinician. Patients are asked to arrive early and complete a questionnaire comprised of PROMs measuring disability, functioning, quality of life, cognitive ability, pain, fatigue, sleep quality, anxiety, and depression. Clinicians score the PROMs and input scores into the electronic health record before the clinical encounter. During the clinic visit, PROM data is visually displayed so that the clinician and patient can discuss results and use the data to better inform decision-making. The visual data display contains longitudinal information, displaying trends in health status across multiple domains, and includes specified thresholds for clinically active symptom levels (Figure).16 Longitudinal monitoring of PROM data allows for real-time assessment of goal-related progress throughout treatment. As illustrated previously by Marion’s case study, the use of real-time feed-forward PROM data can strengthen the partnership between patient and clinician as well as improve empowerment, engagement, self-monitoring, and adherence.
PRO Dashboards
Performance dashboards are increasingly used in health care to visually display clinical and PRO data for individual patients, systems, and populations over time. Dashboards display a parsimonious group of critically important measures to give clinicians and patients a longitudinal view of PRO status. They can inform decision-making in clinical care, operations, health care improvement efforts, and population health initiatives.58 Effective dashboards allow for user customization with meaningful measures, knowledge discovery for analysis of health problems, accessibility of health information, clear visualization, alerts for unexpected data values, and system connectivity.59,60 Appropriate development of PRO dashboards requires meaningful patient and clinician involvement via focus groups and key informant interviews, Delphi process approaches to prioritize and finalize selection of priority measures, iterative building of the interface with design input from key informants and stakeholders (co-design), and pilot testing to assess feasibility and acceptability of use.61-63
Other Applications of PROs/PROMs in MS
Learning Health Systems
The National Quality Forum (NQF) and the Centers for Medicaid and Medicare Services have adopted PROs for use in quality measurement.64-66 This includes a movement towards the use of LHS, defined as a health system in which information from patients and clinicians is systematically collected and synthesized with external evidence to inform clinical care, improvement, and research.67-70 Often a LHS is undertaken as a collaborative effort between multiple health care centers to improve quality and outcomes of care.70 The MS Continuous Quality Improvement Collaborative (MS-CQI), the first multi-center systems-level health care improvement research collaborative for MS,71 as well as IBD Qorus and the Cystic Fibrosis Care Center Network utilize LHS approaches.72-77
IBD Qorus is a LHS developed by the Crohn’s and Colitis Foundation that uses performance dashboards to better inform clinical care for people with inflammatory bowel disease. It also employs system-level dashboards for performance benchmarking in quality improvement initiatives and aggregate-level dashboards to assess population health status.78,79 MS-CQI uses a LHS approach to inform the improvement of MS care across multiple centers using a comprehensive dashboard, including PROMs, for benchmarking and to monitor system and population health status. MS-CQI collects PROMs using a secure online platform that can be accessed by persons with MS and their clinicians and also includes a journaling feature for collecting qualitative information and for reference and self-monitoring.71
MS Research
PROMs are used in clinical and epidemiological research to evaluate many aspects of MS, including the FAMS, the PDSS, the Fatigue Impact Scale (FIS), and others.80-82 For example, the PROMIS FatigueMS and the Fatigue Performance Scale have been used to assess the impact of MS-related fatigue on social participation.83 Generic and MS-specific PROMs have been used to assess pain levels for people with MS,84-87 and multiple MS-specific PROMs, like PRIMUS and MSQoL-54,43 as well as the SF-3639 include pain assessment scales. PROMs have also been used to assess MS-related bladder, bowel, and sexual dysfunction. Urgency, frequency, and incontinence affect up to 75% of patients with MS,88 and many PROMs, such as the LMSQoL, MUSIQoL, and the MSQoL-54, are able to evaluate bladder control and sexual functioning.43,89
PROMs are employed in MS clinical trials to help assess the tolerability and effectiveness of DMTs.90,91 PROs have been used as secondary endpoints to understand the global experience of a DMT from the patient perspective.92-94 There are 15 FDA-approved DMTs for MS, and clinical trials for 6 of these have used PROMs as an effectiveness end point.54,91,95,96 However, most DMT clinical trials are powered for MRI, relapse rate, or disease progression primary outcomes rather than PROMs, often resulting in underpowered PROM analyses.97 In addition, many PROMs are not appropriate for use in DMT clinical trials.98,99
In order to bridge the gap between clinical research and practice, some industry entities are championing “patient-focused drug development” approaches. The Accelerated Cure Project for MS has launched iConquerMS, which collects PROMs from persons with MS to further PRO research in MS and follows 4700 individuals with MS worldwide.100 In 2018, the American College of Physicians announced a collaboration with an industry partner to share data to inform DMT clinical trials and develop and validate PROMs specifically designed for DMT clinical trials.101
Population Health
Registries following large cohorts of people with MS have the potential to develop knowledge about disease progression, treatment patterns, and outcomes.102 The Swedish EIMS study has identified associations between pre-disease body mass index and MS prognosis,102 alcohol and tobacco consumption affecting MS risk,103,104 and exposure to shift work at a young age and increased MS risk.105 The North American Research Committee on MS83,106,107 and iConquerMS registries are “PROM-driven” and have been useful in identifying reductions in disease progression in people using DMTs.107,108 The New York State MS Consortium has identified important demographic characteristics that influence MS progression.109,110 PROs can also be used to determine risk of MS-related mortality111 and decline in quality of life.112,113 Limitations of these approaches include use of different PROMs, inconsistencies in data collection processes, and different follow-up intervals used across registries.102
Patient-Centered Care
The Institute of Medicine defines patient-centeredness as “care that is respectful and responsive to individual patient preferences, needs, and values and ensures that patient values guide all clinical decisions.”114 PROs are useful for identifying a patient’s individual health concerns and preferences, something that is needed when treating a highly variable chronic health condition like MS. The use of PROs can help clinicicans visualize the lived experience of persons with MS and identify personal preferences,115 as well as improve self-monitoring, self-management, self-efficacy, adherence, wellness, and coping ability.116 At the system level, PROs can inform improvement initiatives and patient-centered care design efforts.117-120
Selecting PROMs
Initiatives from groups like the COnsensus-based Standards for the Selection of health Measurement INstruments (COSMIN)121 and the International Society for Quality of Life Research (ISOQOL)108 offer guidance on selecting PROs. The NINDS has promoted common data collection between clinical studies of the brain and nervous system.122 General guidance from these sources recommends first considering the outcome and target population, selecting PROMs to measure the outcome through a synthesis of the available evidence, assessing validity and reliability of selected PROMs, and using standard measures that can be compared across studies or populations.108,121 Other factors include feasibility, acceptability, and burden of use for patients, clinicians, and systems, as well as literacy, cultural, and linguistic factors.123
The NQF recommends that consideration be given to individual patient needs, insurance factors, clinical setting constraints, and available resources when selecting PROMs.124 To maximize response rate, PROMs that are sensitive, reliable, valid, and developed in a comparative demographic of patients are advised.125 ISOQOL has released a User’s Guide and several companion guides on implementing and utilizing PROMs.108,126,127 Finally, PRO-Performance Measures (PRO-PMs) are sometimes used to assess whether PROMs are appropriately contributing to performance improvement and accountability.124
The Cons of PROs
PROs can disrupt busy clinical care environments and overextend clinical staff.125 Online collection of PROs outside of clinical encounters can relieve PRO-related burden, but this requires finding and funding appropriate secure online networks to effectively collect PROs.128 In 2015, only 60% of people seen for primary care visits could access or view their records online, and of those, only 57% used messaging for medical questions or concerns.129 Ideally, online patient portal or mobile health apps could synchronize directly to electronic health records or virtual scribes to transfer patient communications into clinical documentation.130 There has been limited success with this approach in European countries131 and with some chronic illness conditions in the United States.74
Electronic health technologies, including mobile health (mHealth) solutions, have improved the self-monitoring and self-management capability of patients with MS via information sharing in patient networks, assistive technologies, smartphone applications, and wearable devices.132,133 A recent study found that communication modes included secure online patient portal use (29%) and email use (21%), and among those who owned tablets or smartphones, 46% used mHealth apps.134 Social media use has been associated with increased peer/social/emotional support and increased access to health information, as well as clinical monitoring and behavior change.134,135 Individuals using mHealth apps are younger, have comorbidities, and have higher socioeconomic and education levels,135,136 suggesting that inequities in mHealth access exist.
Questionnaires can be time-consuming and cause mental distress if not appropriately facilitated.137 Decreasing questionnaire length and providing the option for PROMs to be delivered and completed online or outside of the clinic context can reduce burden.138 Additionally, while some people are consistent in sharing their PROs, others struggle with using computers, especially while experiencing severe symptoms, forget to complete PROMs, or simply do not have internet access due to financial or geographic constraints.139 A group of disabled and elderly persons with MS reported barriers to internet use due to visual deficits, small website font sizes, and distracting color schemes.140
Interpretability
Interpreting PROMs and displays of longitudinal PROM data can be a challenge for persons with MS and their clinicians. There is little standardization in how PROMs are scored and presented, and there is often confusion about thresholds for clinical significance and how PROM scores can be compared to other PROMs.141,142 While guidelines exist for implementing PRO scores in clinical settings,126,143 there are few that aid PROM interpretation. As a result, clinicians often seek research evidence for PROMs used in other similar patient populations as a benchmark,142-144 or compare them to other patients seen in their clinical practice.
Longitudinal PRO data are usually displayed in simple line graphs.145,146 Overall, line graphs have been found to have the highest ease of understanding by both patients and clinicians, but sometimes can be confusing.147 For example, upward trending lines are usually viewed as improvement and downward trending lines as decline; however, upward trending scores on a PROM can indicate decline, such as increasing fatigue severity. Annotation of visual displays can help. Patients and clinicians find that employing thresholds and color coding is useful, and better than “stoplight” red-yellow-green shading schemes or red-circle formats to indicate data that warrant attention.142
PROs are not free of risk for error, especially if they are used independently of other information sources, such as clinical interview, examination, and diagnostic testing, or if they are utilized too frequently, too infrequently, or are duplicated in practice. If a PRO instrument is employed too frequently, score changes may reflect learning effects rather than actual clinical status. Conversely, if used too infrequently, PRO information will not be timely enough to inform real-time clinical practice. Duplication of PRO assessments (eg, multiple measures of the same PRO for the same patient on the same day) or use of multiple PRO measures to assess the same aspect (eg, 2 measures used to assess fatigue) could introduce unnecessary complexity and confusion to interpretation of PRO results.
PRO measures also can be biased or modified by clinical status and/or perceptions of people with MS at the time of assessment. For example, cognitive impairment, whether at baseline state or due to a cognitive MS relapse event, could impact patients’ ability to understand and respond to PRO assessments, producing erroneous results. However, when used appropriately, PROs targeting cognitive dysfunction may be able to detect onset of cognitive events or help to measure recovery from them. Finally, PROs measure perceived (self-reported) status, which may not be an accurate depiction of actual status.
All of these potential pitfalls support the argument that PROs should be utilized to augment the clinical interview, examination, and diagnostic (objective) testing aspects of comprehensive MS care. In this way, PROs can be correlated with other information sources to deepen the shared understanding of health status between a person with MS and her clinician, increasing the potential to make better treatment decisions and care plans together in partnership.
Value and Cost
National groups such as the Patient-Centered Outcomes Research Institute (PCORI) are working with regulatory bodies, funding agencies, insurance providers, patient advocacy groups, researchers, providers, and specialty groups to investigate how PROMs can be implemented into value-based health care reforms, including value-based reimbursement.148 However, practical PRO implementation requires considerable time and resources, and many methodological and operational questions must be addressed before widespread adoption and reimbursement for PROMs will be feasible.148,149
Summary
PROs can generate valuable information about perceived health status, function, quality of life, and experience of care using self-reported sources. Validated PRO assessment tools include PROMs and PREMs. PROs are currently utilized in research settings (especially PROMs) but are also being used in clinical practice, quality improvement initiatives, and population health applications using LHS approaches. PROs have the advantages of empowering and informing persons with MS and clinicians to optimize patient-centered care, improve systems of care, and study population health outcomes. Barriers include PROM validity, reliability, comparability, specificity, interpretability, equity, time, and cost. Generic PROMs and PREMs, and some MS-specific PROMs, can be used for persons with MS. Unfortunately, no PREMs have been developed specifically for persons with MS, and this is an area for future research. With appropriate development and utilization in LHS applications, PROs can inform patient-centered clinical care, system-level improvement initiatives, and population health research, and have the potential to help facilitate coproduction of health care services.
Acknowledgments: The authors thank Ann Cabot, DO, of the MS Specialty Care Program at Concord Hospital and (especially) peer mentors from a peer outreach wellness program for people with MS (who have asked to remain anonymous) for interviews conducted with their permission to inform the case study described in this article. The case study used in this manuscript has been de-identified, with some aspects modified from actual, and the person in the case study is fictitiously named.
Corresponding author: Brant Oliver, PhD, MS, MPH, APRN-BC, Department of Community & Family Medicine, Dartmouth-Hitchcock Medical Center, Lebanon, NH 03756; [email protected].
Financial disclosures: None.
1. Goldenberg MM. Multiple sclerosis review. PT. 2012;37:175-184.
2. Doshi A, Chataway J. Multiple sclerosis, a treatable disease. Clin Med. 2016;16(Suppl 6):s53-s9.
3. Perrin Ross A. Management of multiple sclerosis. Am J Managed Care. 2013;19(16 Suppl):s301-s306.
4. Batalden M, Batalden P, Margolis P, et al. Coproduction of healthcare service. BMJ Qual Saf. 2016;25:509-517.
5. Chen J, Ou L, Hollis SJ. A systematic review of the impact of routine collection of patient reported outcome measures on patients, providers and health organisations in an oncologic setting. BMC Health Serv Res. 2013;13:211.
6. Knaup C, Koesters M, Schoefer D, et al. Effect of feedback of treatment outcome in specialist mental healthcare: meta-analysis. Br J Psychiatr. 2009;195:15-22.
7. Dudgeon D. The impact of measuring patient-reported outcome measures on quality of and access to palliative care. J Palliat Med. 2018;21(S1):S76-s80.
8. Snyder CF, Herman JM, White SM, et al. When using patient-reported outcomes in clinical practice, the measure matters: a randomized controlled trial. J Oncol Pract. 2014;10:e299-e306.
9. Guidance for industry: patient-reported outcome measures: use in medical product development to support labeling claims: draft guidance. Health Qual Life Outcomes. 2006;4:79.
10. de Jong MJ, Huibregtse R, Masclee AAM, et al. Patient-reported outcome measures for use in clinical trials and clinical practice in inflammatory bowel diseases: a systematic review. Clin Gastroenterol Hepatol. 2018;16:648-663.
11. Roman D, Osborne-Stafsnes J, Amy CH, et al. Early lessons from four ‘aligning forces for quality’ communities bolster the case for patient-centered care. Health Affairs. 2013;32:232-241.
12. Ehde DM, Alschuler KN, Sullivan MD, et al. Improving the quality of depression and pain care in multiple sclerosis using collaborative care: The MS-care trial protocol. Contemporary Clinical Trials. 2018;64:219-229.
13. Lavallee DC, Chenok KE, Love RM, et al. Incorporating patient-reported outcomes into health care to engage patients and enhance care. Health Affairs. 2016;35:575-582.
14. Schick-Makaroff K, Thummapol O, Thompson S, et al. Strategies for incorporating patient-reported outcomes in the care of people with chronic kidney disease (PRO kidney): a protocol for a realist synthesis. Clin Gastroenterol Hepatol. 2018;16:648-663.
15. Nelson EC, Dixon-Woods M, Batalden PB, et al. Patient focused registries can improve health, care, and science. BMJ. 2016;354:i3319.
16. Oliver BJ, Nelson EC, Kerrigan CL. Turning feed-forward and feedback processes on patient-reported data into intelligent action and informed decision-making: case studies and principles. Med Care. 2019;57 Suppl 1:S31-s37.
17. Deshpande PR, Rajan S, Sudeepthi BL, Abdul Nazir CP. Patient-reported outcomes: A new era in clinical research. Perspect Clin Res. 2011;2:137-144.
18. Epstein RM, Street RL, Jr. The values and value of patient-centered care. Ann Fam Med. 2011;9:100-103.
19. Payne SA. A study of quality of life in cancer patients receiving palliative chemotherapy. Soc Sci Med. 1992;35:1505-1509.
20. Borgaonkar MR, Irvine EJ. Quality of life measurement in gastrointestinal and liver disorders. Gut. 2000;47:444-454.
21. Macdonell R, Nagels G, Laplaud DA, et al. Improved patient-reported health impact of multiple sclerosis: The ENABLE study of PR-fampridine. Multiple Sclerosis. 2016;22:944-954.
22. Hobart J, Freeman J, Lamping D, et al. The SF-36 in multiple sclerosis: why basic assumptions must be tested. J Neurol Neurosurg Psychiatry. 2001;71:363-370.
23. Lins L, Carvalho FM. SF-36 total score as a single measure of health-related quality of life: Scoping review. SAGE Open Med. 2016;4:2050312116671725.
24. Parkinson’s Disease Society of the United Kingdom. The Parkinson’s Disease Questionnaire (PDQ-39). https://www.parkinsons.org.uk/professionals/resources/parkinsons-disease-questionnaire-pdq-39. Accessed October 20, 2019.
25. Basch E. The missing voice of patients in drug-safety reporting. N Engl J Med. 2010;362:865-869.
26. Calvert M, Brundage M, Jacobsen PB, et al. The CONSORT Patient-Reported Outcome (PRO) extension: implications for clinical trials and practice. Health Qual Life Outcomes. 2013;11:184.
27. Black N. Patient reported outcome measures could help transform healthcare. BMJ. 2013;346:f167.
28. Franks P, Fiscella K, Shields CG, et al. Are patients’ ratings of their physicians related to health outcomes? Ann Fam Med. 2005;3:229-234.
29. Hostetter M, Klein S. Using patient-reported outcomes to improve health care quality. The Commonwealth Fund 2019. www.commonwealthfund.org/publications/newsletter-article/using-patient-reported-outcomes-improve-health-care-quality. Accessed October 24, 2019.
30. Charlotte Kingsley SP. Patient-reported outcome measures and patient-reported experience measures. BJA Education. 2017;17:137-144.
31. Weldring T, Smith SM. Patient-Reported outcomes (PROs) and patient-reported outcome measures (PROMs). Health Services Insights. 2013;6:61-68.
32. Elwyn G, Barr PJ, Grande SW, et al. Developing CollaboRATE: A fast and frugal patient-reported measure of shared decision making in clinical encounters. Patient Educ Couns. 2013;93:102-107.
33. Kurtzke JF. Rating neurologic impairment in multiple sclerosis: an expanded disability status scale (EDSS). Neurology. 1983;33:1444-1452.
34. Rudick RA, Miller D, Clough JD, et al. Quality of life in multiple sclerosis. Comparison with inflammatory bowel disease and rheumatoid arthritis. Arch Neurol. 1992;49:1237-1242.
35. Benito-Leon J, Morales JM, Rivera-Navarro J, Mitchell A. A review about the impact of multiple sclerosis on health-related quality of life. Disabil Rehabil. 2003;25:1291-1303.
36. RAND Corporation. 36-Item Short Form Survey (SF-36). [https://www.rand.org/health-care/surveys_tools/mos/36-item-short-form.html. Accessed October 20, 2019.
37. Bergner M, Bobbitt RA, Carter WB, Gilson BS. The sickness impact profile: development and final revision of a health status measure. Medical Care. 1981;19:787-805.
38. Horsman J, Furlong W, Feeny D, Torrance G. The Health Utilities Index (HUI): concepts, measurement properties and applications. Health Qual Life Outcomes. 2003;1:54.
39. Kinkel RP, Laforet G, You X. Disease-related determinants of quality of life 10 years after clinically isolated syndrome. Int J MS Care. 2015;17:26-34.
40. Baumstarck-Barrau K, Simeoni MC, Reuter F, et al. Cognitive function and quality of life in multiple sclerosis patients: a cross-sectional study. BMC Neurology. 2011;11:17.
41. Samartzis L, Gavala E, Zoukos Y, et al. Perceived cognitive decline in multiple sclerosis impacts quality of life independently of depression. Rehabil Res Pract. 2014;2014:128751.
42. Vitkova M, Rosenberger J, Krokavcova M, et al. Health-related quality of life in multiple sclerosis patients with bladder, bowel and sexual dysfunction. Disabil Rehabil. 2014;36:987-992.
43. Khurana V, Sharma H, Afroz N, et al. Patient-reported outcomes in multiple sclerosis: a systematic comparison of available measures. Eur J Neurol. 2017;24:1099-1107.
44. Hobart J, Lamping D, Fitzpatrick R, et al. The Multiple Sclerosis Impact Scale (MSIS-29): a new patient-based outcome measure. Brain. 2001;124(Pt 5):962-973.
45. Lily O, McFadden E, Hensor E, et al. Disease-specific quality of life in multiple sclerosis: the effect of disease modifying treatment. Multiple Sclerosis. 2006;12:808-813.
46. Ford HL, Gerry E, Tennant A, et al. Developing a disease-specific quality of life measure for people with multiple sclerosis. Clin Rehabil. 2001;15:247-258.
47. Cella DF, Dineen K, Arnason B, et al. Validation of the functional assessment of multiple sclerosis quality of life instrument. Neurology. 1996;47:129-139.
48. Gold SM, Heesen C, Schulz H, et al. Disease specific quality of life instruments in multiple sclerosis: validation of the Hamburg Quality of Life Questionnaire in Multiple Sclerosis (HAQUAMS). Multiple Sclerosis. 2001;7:119-130.
49. Vickrey BG, Hays RD, Harooni R, et al. A health-related quality of life measure for multiple sclerosis. Qual Life Res. 1995;4:187-206.
50. Simeoni M, Auquier P, Fernandez O, et al. Validation of the Multiple Sclerosis International Quality of Life questionnaire. Multiple Sclerosis. 2008;14:219-230.
51. Doward LC, McKenna SP, Meads DM, et al. The development of patient-reported outcome indices for multiple sclerosis (PRIMUS). Multiple Sclerosis. 2009;15:1092-1102.
52. Ozakbas S, Akdede BB, Kosehasanogullari G, et al. Difference between generic and multiple sclerosis-specific quality of life instruments regarding the assessment of treatment efficacy. J Neurol Sci. 2007;256:30-34.
53. Cella DF, Wiklund I, Shumaker SA, Aaronson NK. Integrating health-related quality of life into cross-national clinical trials. Qual Life Res. 1993;2:433-440.
54. Nowinski CJ, Miller DM, Cella D. Evolution of patient-reported outcomes and their role in multiple sclerosis clinical trials. Neurotherapeutics. 2017;14:934-944.
55. Cella D, Nowinski C, Peterman A, et al. The neurology quality-of-life measurement initiative. Arch Phys Med Rehabil. 2011;92(10 Suppl):S28-S36.
56. HealthMeasures: PROMIS. http://www.healthmeasures.net/explore-measurement-systems/promis. Accessed October 20, 2019.
57. Valderas JM, Ferrer M, Mendivil J, et al. Development of EMPRO: a tool for the standardized assessment of patient-reported outcome measures. Value Health. 2008;11:700-708.
58. Bach K, Martling C, Mork PJ, et al. Design of a clinician dashboard to facilitate co-decision making in the management of non-specific low back pain. J Intelligent Helath Syst. 2019;52:269-284.
59. Karami M, Safdari R, Rahimi A. Effective radiology dashboards: key research findings. Radiology Management. 2013;35:42-45.
60. Dowding D, Randell R, Gardner P, et al. Dashboards for improving patient care: review of the literature. Int J Med Informatic. 2015;84:87-100.
61. Mlaver E, Schnipper JL, Boxer RB, et al. User-centered collaborative design and development of an inpatient safety dashboard. Jt Comm J Qual Patient Saf. 2017;43:676-685.
62. Dowding D, Merrill J, Russell D. Using feedback intervention theory to guide clinical dashboard design. AMIA Annu Symp Proc. 2018;2018:395-403.
63. Hartzler AL, Izard JP, Dalkin BL, et al. Design and feasibility of integrating personalized PRO dashboards into prostate cancer care. J Am Med Inform Assoc. 2016;23:38-47.
64. Lavallee DC, Chenok KE, Love RM, et al. Incorporating patient-reported outcomes into health care to engage patients and enhance care. Health Affairs. 2016;35:575-582.
65. U.S. Centers for Medicare & Medicaid Services. Comprehensive Care for Joint Replacement Model. https://innovation.cms.gov/initiatives/CJR. Accessed August 3, 2019.
66. National Quality Forum. Patient-reported outcomes: Accessed August 3, 2019. http://www.qualityforum.org/Patient-Reported_Outcomes.aspx. Accessed October 20, 2019.
67. Institute of Medicine Roundtable on Evidence-Based Medicine. The National Academies Collection: Reports funded by National Institutes of Health. In: Olsen LA, Aisner D, McGinnis JM, eds. The Learning Healthcare System: Workshop Summary. Washington (DC): National Academies Press (US).National Academy of Sciences; 2007.
68. About Learning Health Systems: Agency for Healthcare Research and Quality. https://www.ahrq.gov/learning-health-systems/about.html. Accessed August 3, 2019.
69. Best Care at Lower Cost: The Path to Continuously Learning Health Care in America. Washington (DC): National Academies Press; 2013.
70. Committee on Quality of Health Care in America. Crossing the Quality Chasm: A New Health System for the 21st Century. Washington, DC: National Academies Press; 2001.
71. Oliver BJ, for the MS-CQI Investigators. The Multiple Sclerosis Continuous improvement collaborative: the first coproduction learning health system improvement science research collaborative for multiple sclerosis. The International Society for Quality in Health Care (ISQua) Annual International Conference; October 2019; Cape Town, South Africa.
72. Britto MT, Fuller SC, Kaplan HC, et al. Using a network organisational architecture to support the development of Learning Healthcare Systems. BMJ Qual Saf. 2018;27:937-946.
73. Care Centers: Cystic Fibrosis Foundation. https://www.cff.org/Care/Care-Centers/. Accessed August 3, 2019.
74. Schechter MS, Fink AK, Homa K, Goss CH. The Cystic Fibrosis Foundation patient registry as a tool for use in quality improvement. BMJ Qual Saf. 2014;23(Suppl 1):i9-i14.
75. Mogayzel PJ, Dunitz J, Marrow LC, Hazle LA. Improving chronic care delivery and outcomes: the impact of the cystic fibrosis Care Center Network. BMJ Qual Saf. 2014;23(Suppl 1):i3-i8.
76. Godfrey MM, Oliver BJ. Accelerating the rate of improvement in cystic fibrosis care: contributions and insights of the learning and leadership collaborative. BMJ Qual Saf. 2014;23(Suppl 1):i23-i32.
77. Nelson EC, Godfrey MM. Quality by Design: A Clinical Microsystems Approach. San Francisco, California: Jossey-Bass; 2007.
78. Crohn’s & Colitis Foundation.Quality of Care: IBD Qorus: https://www.crohnscolitisfoundation.org/research/ibd-qorus. Accessed October 23, 2019.
79. Johnson LC, Melmed GY, Nelson EC, et al. Fostering collaboration through creation of an ibd learning health system. Am J Gastroenterol. 2017;112:406-468.
80. Fernandez-Munoz JJ, Moron-Verdasco A, Cigaran-Mendez M, et al. Disability, quality of life, personality, cognitive and psychological variables associated with fatigue in patients with multiple sclerosis. Acta Neurologica Scandinavica. 2015;132:118-124.
81. Flachenecker P, Kumpfel T, Kallmann B, et al. Fatigue in multiple sclerosis: a comparison of different rating scales and correlation to clinical parameters. Multiple Sclerosis. 2002;8:523-526.
82. Tellez N, Rio J, Tintore M, et al. Does the Modified Fatigue Impact Scale offer a more comprehensive assessment of fatigue in MS? Multiple Sclerosis. 2005;11:198-202.
83. Salter A, Fox RJ, Tyry T, et al. The association of fatigue and social participation in multiple sclerosis as assessed using two different instruments. Mult Scler Relat Disord. 2019;31:165-72.
84. Solaro C, Trabucco E, Messmer Uccelli M. Pain and multiple sclerosis: pathophysiology and treatment. Curr Neurol Neurosci Rep. 2013;13:320.
85. Jawahar R, Oh U, Yang S, Lapane KL. A systematic review of pharmacological pain management in multiple sclerosis. Drugs. 2013;73:1711-1722.
86. Rintala A, Hakkinen A, Paltamaa J. Ten-year follow-up of health-related quality of life among ambulatory persons with multiple sclerosis at baseline. Qual Life Res. 2016;25:3119-3127.
87. Tepavcevic DK, Pekmezovic T, Stojsavljevic N, et al. Change in quality of life and predictors of change among patients with multiple sclerosis: a prospective cohort study. Qual Life Res. 2014;23:1027-1037.
88. DasGupta R, Fowler CJ. Bladder, bowel and sexual dysfunction in multiple sclerosis: management strategies. Drugs. 2003;63:153-166.
89. Marck CH, Jelinek PL, Weiland TJ, et al. Sexual function in multiple sclerosis and associations with demographic, disease and lifestyle characteristics: an international cross-sectional study. BMC Neurology. 2016;16:210.
90. Gajofatto A, Benedetti MD. Treatment strategies for multiple sclerosis: When to start, when to change, when to stop? World J Clinical Cases. 2015;3:545-555.
91. National Multiple Sclerosis Society. Disease-modifying therapies for MS. 2018.
92. Rudick RA, Panzara MA. Natalizumab for the treatment of relapsing multiple sclerosis. Biologics. 2008;2:189-199.
93. Cohen JA, Cutter GR, Fischer JS, et al. Benefit of interferon beta-1a on MSFC progression in secondary progressive MS. Neurology. 2002;59:679-687.
94. Patti F, Pappalardo A, Montanari E, et al. Interferon-beta-1a treatment has a positive effect on quality of life of relapsing-remitting multiple sclerosis: results from a longitudinal study. J Neurol Sci. 2014;337:180-185.
95. Novartis. Exploring the efficacy and safety of Siponimod in patients with secondary progressive multiple sclerosis (EXPAND). Available from clinicaltrials.gov/ct2/show/NCT01665144. NLM identifier: NCT01665144.
96. Singal AG, Higgins PD, Waljee AK. A primer on effectiveness and efficacy trials. Clin Transl Gastroenterol. 2014;5:e45.
97. Jongen PJ. Health-related quality of life in patients with multiple sclerosis: impact of disease-modifying drugs. CNS Drugs. 2017;31:585-602.
98. Coretti S, Ruggeri M, McNamee P. The minimum clinically important difference for EQ-5D index: a critical review. Expert Rev Pharmacoecon Outcomes Res. 2014;14:221-233.
99. Clinical Review Report: Ocrelizumab (Ocrevus). CADTH Common Drug Review. 2018:Appendix 4, Validity of Outcome Measures.
100. iCounquer MS; Empowering patients: how individuals with ms are contributing to the fight to find a cure: 2018. https://www.iconquerms.org/empowering-patients-how-individuals-ms-are-contributing-fight-find-cure. Accessed October 23, 2019.
101. K M. Accelerated Cure Project Announces Collaboration with EMD Serono to Advance Patient-Focused Drug Development in Multiple Sclerosis2018. https://www.iconquerms.org/accelerated-cure-project-announces-collaboration-emd-serono-advance-patient-focused-drug-development. Accessed October 24, 2019.
102. Bebo BF Jr, Fox RJ, Lee K, et al. Landscape of MS patient cohorts and registries: Recommendations for maximizing impact. Multiple Sclerosis. 2018;24:579-586.
103. Hedstrom AK, Hillert J, Olsson T, Alfredsson L. Alcohol as a modifiable lifestyle factor affecting multiple sclerosis risk. JAMA Neurology. 2014;71:300-305.
104. Hedstrom AK, Baarnhielm M, Olsson T, Alfredsson L. Tobacco smoking, but not Swedish snuff use, increases the risk of multiple sclerosis. Neurology. 2009;73:696-701.
105. Hedstrom AK, Akerstedt T, Hillert J, et al. Shift work at young age is associated with increased risk for multiple sclerosis. Ann Neurol. 2011;70:733-741.
106. Hadjimichael O, Vollmer T, Oleen-Burkey M. Fatigue characteristics in multiple sclerosis: the North American Research Committee on Multiple Sclerosis (NARCOMS) survey. Health Qual Life Outcomes. 2008;6:100.
107. Fox RJ, Salter AR, Tyry T, et al. Treatment discontinuation and disease progression with injectable disease-modifying therapies: findings from the North American research committee on multiple sclerosis database. Int J MS Care. 2013;15:194-201.
108. Reeve BB, Wyrwich KW, Wu AW, et al. ISOQOL recommends minimum standards for patient-reported outcome measures used in patient-centered outcomes and comparative effectiveness research. Qual Life Res. 2013;22:1889-1905.
109. Jacobs LD, Wende KE, Brownscheidle CM, et al. A profile of multiple sclerosis: the New York State Multiple Sclerosis Consortium. Multiple Sclerosis. 1999;5:369-376.
110. Weinstock-Guttman B, Jacobs LD, Brownscheidle CM, et al. Multiple sclerosis characteristics in African American patients in the New York State Multiple Sclerosis Consortium. Multiple Sclerosis. 2003;9:293-298.
111. Raffel J, Wallace A, Gveric D, et al. Patient-reported outcomes and survival in multiple sclerosis: A 10-year retrospective cohort study using the Multiple Sclerosis Impact Scale-29. PLoS Med. 2017;14:e1002346.
112. Vaughn CB, Kavak KS, Dwyer MG, et al. Fatigue at enrollment predicts EDSS worsening in the New York State Multiple Sclerosis Consortium. Multiple Sclerosis. 2018:1352458518816619.
113. Alroughani RA, Akhtar S, Ahmed SF, Al-Hashel JY. Clinical predictors of disease progression in multiple sclerosis patients with relapsing onset in a nation-wide cohort. Int J Neuroscience. 2015;125:831-837.
114. Dolan JG, Veazie PJ, Russ AJ. Development and initial evaluation of a treatment decision dashboard. BMC Med Inform Decision Mak. 2013;13:51.
115. Stuifbergen AK, Becker H, Blozis S, et al. A randomized clinical trial of a wellness intervention for women with multiple sclerosis. Arch Phys Med Rehabil. 2003;84:467-476.
116. Finkelstein J, Martin C, Bhushan A, et al. Feasibility of computer-assisted education in patients with multiple sclerosis. In: Proceedings of the IEEE Symposium on Computer-Based Medical Systems. Bethesda, MD; 2004.
117. Batalden P. Getting more health from healthcare: quality improvement must acknowledge patient coproduction—an essay by Paul Batalden. BMJ. 2018;362:k3617.
118. Batalden PB, Davidoff F. What is “quality improvement” and how can it transform healthcare? Qual Saf Health Care. 2007;16:2-3.
119. Ovretveit J, Zubkoff L, Nelson EC, et al. Using patient-reported outcome measurement to improve patient care. Int J Qual Health Care. 2017;29:874-879.
120. Prodinger B, Taylor P. Improving quality of care through patient-reported outcome measures (PROMs): expert interviews using the NHS PROMs Programme and the Swedish quality registers for knee and hip arthroplasty as examples. BMC Health Services Res. 2018;18:87.
121. Prinsen CA, Vohra S, Rose MR, et al. How to select outcome measurement instruments for outcomes included in a “Core Outcome Set” – a practical guideline. Trials. 2016;17:449.
122. NINDS. NINDS Common Data Elements: Streamline Your Neuroscience Clinical Research National Institute of Health. https://www.commondataelements.ninds.nih.gov/#page=Default. Accessed October 23, 2019.
123. Francis DO, McPheeters ML, Noud M, et al. Checklist to operationalize measurement characteristics of patient-reported outcome measures. Systematic Rev. 2016;5:129.
124. National Quality Forum. Patient-reported outcomes (PROs) in performance measurement. 2013. https://www.qualityforum.org/Publications/2012/12/Patient-Reported_Outcomes_in_Performance_Measurement.aspx. Accessed October 15, 2019.
125. Dawson J, Doll H, Fitzpatrick R, et al. The routine use of patient reported outcome measures in healthcare settings. BMJ. 2010;340:c186.
126. Aaronson N ET, Greenlhalgh J, Halyard M, et al. User’s guide to implementation of patient reported outcomes assessment in clinical practice. International Society for Quality of Life. 2015.
127. Chan EKH, Edwards TC, Haywood K, et al. Implementing patient-reported outcomes mesaures in clinical practice: a companion guide to the ISOQOL user’s guide. Qual Life Res. 2018;28:621-627.
128. Snyder CF, Wu AW, Miller RS, et al. The role of informatics in promoting patient-centered care. Cancer J. 2011;17:211-218
129. Osborn R, Moulds D, Schneider EC, et al. Primary care physicians in ten countries report challenges caring for patients with complex health needs. Health Affairs. 2015;34:2104-2112.
130. Wu AW, Kharrazi H, Boulware LE, Snyder CF. Measure once, cut 0wice--adding patient-reported outcome measures to the electronic health record for comparative effectiveness research. J Clin Epidemiol. 2013;66(8 Suppl):S12-S20.
131. Trojano M, Bergamaschi R, Amato MP, et al. The Italian multiple sclerosis register. Neurol Sci. 2019;40:155-165.
132. Moorhead SA, Hazlett DE, Harrison L, et al. A new dimension of health care: systematic review of the uses, benefits, and limitations of social media for health communication. J Med Internet Res. 2013;15:e85.
133. Luigi L, Francesco B, Marcello M, et al. e-Health and multiple sclerosis: An update. Multiple Sclerosis J. 2018;24:1657-1664.
134. Marrie RA, Leung S, Tyry T, et al. Use of eHealth and mHealth technology by persons with multiple sclerosis. Mult Scler Relat Dis. 2019;27:13-19.
135. Giunti G, Kool J, Rivera Romero O, Dorronzoro Zubiete E. Exploring the specific needs of persons with multiple sclerosis for mhealth solutions for physical activity: mixed-methods study. JMIR Mhealth Uhealth. 2018;6:e37.
136. Spooner KK, Salemi JL, Salihu HM, Zoorob RJ. eHealth patient-provider communication in the United States: interest, inequalities, and predictors. J Am Med Inform Assoc. 2017;24(e1):e18-e27.
137. Safdar N, Abbo LM, Knobloch MJ, Seo SK. Research methods in healthcare epidemiology: survey and qualitative research. Infect Control Hosp Epidemiol. 2016;37:1272-1277.
138. Rolstad S, Adler J, Rydén A. Response burden and questionnaire length: is shorter better? A review and meta-analysis. Value Health. 2011;14:1101-1108.
139. Chiu C, Bishop M, Pionke JJ, et al. Barriers to the accessibility and continuity of health-care services in people with multiple sclerosis: a literature review. Int J MS Care. 2017;19:313-321.
140. Atreja A, Mehta N, Miller D, et al. One size does not fit all: using qualitative methods to inform the development of an Internet portal for multiple sclerosis patients. AMIA Annu Symp Proc Symp. 2005:16-20.
141. Snyder CF, Blackford AL, Brahmer JR, et al. Needs assessments can identify scores on HRQOL questionnaires that represent problems for patients: an illustration with the Supportive Care Needs Survey and the QLQ-C30. Qual Life Res. 2010;19:837-845.
142. de Vet HC, Terwee CB. The minimal detectable change should not replace the minimal important difference. J Clin Epidemiol. 2010;63:804-805.
143. Snyder CF, Aaronson NK, Choucair AK, et al. Implementing patient-reported outcomes assessment in clinical practice: a review of the options and considerations. Qual Life. 2012;21:1305-1314.
144. Myla DG, Melanie DW, Robert WM, et al. Identification and validation of clinically meaningful benchmarks in the 12-item Multiple Sclerosis Walking Scale. Multiple Sclerosis J. 2017;23:1405-1414.
145. van Munster CEP, Uitdehaag BMJ. Outcome measures in clinical trials for multiple sclerosis. CNS Drugs. 2017;31:217-236.
146. Snyder CF, Jensen R, Courtin SO, Wu AW. PatientViewpoint: a website for patient-reported outcomes assessment. Quality Life Res. 2009;18:793-800.
147. Snyder CF, Smith KC, Bantug ET, et al. What do these scores mean? Presenting patient-reported outcomes data to patients and clinicians to improve interpretability. Cancer. 2017;123:1848-1859.
148. Squitieri L, Bozic KJ, Pusic AL. The role of patient-reported outcome measures in value-based payment reform. Value Health. 2017;20:834-836.
149. Boyce MB, Browne JP. The effectiveness of providing peer benchmarked feedback to hip replacement surgeons based on patient-reported outcome measures—results from the PROFILE trial: a cluster randomised controlled study. BMJ Open. 2015;5:e008325.
From the Dartmouth Institute for Health Policy & Clinical Practice, Geisel School of Medicine, Hanover, NH (Ms. Manohar and Dr. Oliver), the Department of Community & Family Medicine, Dartmouth-Hitchcock Medical Center, Lebanon, NH (Ms. Perkins, Ms. Laurion, and Dr. Oliver), and the Multiple Sclerosis Specialty Care Program, Concord Hospital, Concord, NH (Dr. Oliver).
Abstract
- Background: Patient-reported outcomes (PROs), including patient-reported outcome measures (PROMs) and patient-reported experience measures (PREMs), can be used to assess perceived health status, functioning, quality of life, and experience of care. Complex chronic illnesses such as multiple sclerosis (MS) affect multiple aspects of health, and PROs can be applied in assessment and decision-making in MS care as well as in research pertaining to MS.
- Objective: To provide a general review of PROs, with a specific focus on implications for MS care.
- Methods: Evidence synthesis of available literature on PROs in MS care.
- Results: PROs (including PROMs and PREMs) have historically been utilized in research and are now being applied in clinical, improvement, and population health settings using learning health system approaches in many disease populations, including MS. Many challenges complicate the use of PROs in MS care, including reliability, validity, and interpretability of PROMs, as well as feasibility barriers due to time and financial constraints in clinical settings.
- Conclusion: PROs have the potential to better inform clinical care, empower patient-centered care, inform health care improvement efforts, and create the conditions for coproduction of health care services.
Keywords: PRO; PROM; patient-reported outcome measure; patient-reported experience measure; quality of life; patient-centered care.
Multiple sclerosis (MS) is a disabling, complex, chronic, immune-mediated disorder of the central nervous system (CNS). MS causes inflammatory and degenerative damage in the CNS, which disrupts signaling pathways.1 It is most commonly diagnosed in young adults and affects 2.3 million people worldwide.2 People with MS experience very different disease courses and a wide range of neurological symptoms, including visual, somatic, mental health, sensory, motor, and cognitive problems.1-3 Relapsing-remitting MS, the most common form, affects 85% of those with MS and is characterized by periods of relapse (exacerbation) and remission.1 Other forms of MS (primary progressive and secondary progressive MS) are characterized by progressive deterioration and worsening symptom severity without exacerbations. Disease-modifying therapies (DMTs) can reduce the frequency of exacerbations and disability progression, but unfortunately there is no cure for MS. Treatment is focused on increasing quality of life, minimizing disability, and maximizing wellness.
Patient-reported outcomes (PROs) describe the perceived health status, function, and/or experience of a person as obtained by direct self-report. Patient-reported outcome measures (PROMs) are validated PROs that can be used to inform clinical care,4 and have demonstrated effectiveness in improving patient-provider communication and decision-making.5-7 PROMs are currently used in some MS clinical trials to determine the impact of experimental interventions,8-10 and are also being used to inform and improve clinical care in some settings. Especially for persons with MS, they can provide individualized perspectives about health experience and outcomes.11 In more advanced applications, PROMs can be used to improve face-to-face collaborations between clinicians and patients and to inform patient-centered systems of care.12-14 PROMs can also be used to inform systems-level improvement for entire patient populations.15,16
In this article, we review current applications of PROs and PROMs in the care of persons with MS, as well as current limitations and barriers to their use.
At a recent visit to her neurologist, Marion reviews her health diary, in which she has been tracking her fatigue levels throughout the day and when she has to visit the bathroom. The PRO diary also helps her remember details that she might not otherwise be able to recall at the time of her clinic visit. They review the diary entrees together to develop a shared understanding of what Marion has been experiencing and identify trends in the PRO data. They discuss symptom management and use the PRO information from the diary to help guide adjustments to her physical therapy routine and medication regimen.
Part of Marion’s “PRO package” includes the Center for Epidemiologic Studies Depression Scale (CES-D), a validated depression screening and symptom severity questionnaire that she completes every 3 months. Although she denies being depressed, she has noticed that her CES-D scores in recent months have been consistently increasing. This prompts a discussion about mental health in MS and a referral to work on depression with the MS mental health specialist. Marion and the mental health specialist use CES-D measures at baseline and during treatment to set a remission target and to track progress during treatment. Marion finds this helpful because she says it is hard for her to “wrap my hands around depression… it’s not something that there is a blood test or a MRI for.” Marion is encouraged by being able to see her CES-D scores change as her depression severity decreases, and this helps motivate her to keep engaged in treatment.
PROs and PROMs: General Applications
PROs are measures obtained directly from an individual without a priori interpretation by a clinician.9,17 PROs capture individual perspectives on symptoms, capability, disability, and health-related quality of life.9 With increasing emphasis on patient-centered care,18 individual perspectives and preferences elicited using PROMs may be able to inform better quality of care and patient-centered disease treatment and management.19-21
PROMs are standardized, validated questionnaires used to assess PROs and can be generic or condition-specific. Generic PROMs can be used in any patient population. The SF-3622 is a set of quality of life measures that assess perceived ability to complete physical tasks and routine activities, general health status, fatigue, social functioning, pain, and emotional and mental health.23 Condition-specific PROMs can be used for particular patient populations and are helpful in identifying changes in health status for a specific disease, disability, or surgery. For example, the PDQ-39 assesses 8 dimensions of daily living, functioning, and well-being for people with Parkinson’s disease.24
PROMs have been used in some MS clinical trials and research studies to determine the effectiveness of experimental treatments from the viewpoint of study participants.9,25,26 PROs can also be utilized in clinical care to facilitate communication of needs and track health outcomes,27 and can inform improvement in outcomes for health systems and populations. They can also be used to assess experience of care,28 encouraging a focus on high-quality outcomes through PRO-connected reimbursement mechanisms,29 and provide aggregate data to evaluate clinical practice, population health outcomes, and the effectiveness of public policies.27
Patient-reported experience measures (PREMs) assess patient satisfaction and experience of health care.30,31 CollaboRATE32 is a PREM that assesses the degree of shared decision-making occurring between patients and clinicians during clinical care. PREMs are currently used for assessing self-efficacy and in shared decision-making and health care improvement applications. PREMs have yet to be developed specifically for persons with MS.
PROMs in MS Care
Generic PROMs have shown that persons with MS are disproportionately burdened by poor quality of life.33-35 Other generic PROMs, like the SF-36,36 the Sickness Impact Profile,37 and versions of the Health Utilities Index,38 can be used to gather information on dysfunction and to determine quality and duration of life modified by MS-related dysfunction and disability. MS-specific PROMs are used to assess MS impairments, including pain, fatigue, cognition, sexual dysfunction, and depression.12,39-42 PROMs have also been used in MS clinical trials, including the Multiple Sclerosis Impact Scale-29 (MSIS-29),43,44 the Leeds MS QoL (LMSQoL),45,46 the Functional Assessment of MS (FAMS),47 the Hamburg Quality of Life Questionnaire in MS (HAQUAMS),48 the MS Quality of Life-54 (MSQoL-54),49 the MS International QoL (MUSIQoL),50 and the Patient-Reported Indices for MS Activity Limitations Scale (PRIMUS).51
Condition-specific PROMs are more sensitive to changes in health status and functioning for persons with MS compared to generic PROMs. They are also more reliable during MS remission and relapse periods.44,52 For example, the SF-36 has floor and ceiling effects in MS populations—a high proportion of persons with MS are scored at the maximum or minimum levels of the scale, limiting discriminant capability.22 As a result, a “combined approach” using both generic and MS-specific measures is often recommended.53 Some MS PROMs (eg, MSQoL-54) include generic questions found in the SF-36 as well as additional MS-specific questions or scales.
The variety of PROMs available (see Table for a selected listing) introduces a significant challenge to using them—limited generalizability and difficulty comparing PROs across MS studies. Efforts to establish common PROMs have been undertaken to address this problem.54 The National Institute of Neurologicical Disorders and Stroke (NINDS) sponsored the development of a neurological quality of life battery, the Neuro-QOL.55 Neuro-QOL measures the physical, mental, and social effects of neurological conditions in adults and children with neurological disorders and has the capability to facilitate comparisons across different neurological conditions. Additionally, the Patient-Reported Outcomes Measure Information System (PROMIS) has been developed to assess physical, mental, and social effects of chronic disease. PROMIS has a hybrid design that includes generic and MS-specific measures (such as PROMIS FatigueMS).56 PROMIS can be used to assess persons with MS as well as to compare the MS population with other populations with chronic illness.

PROMs have varying levels of reliability and validity. The Evaluating the Measure of Patient-Reported Outcomes57 study evaluated the development process of MS PROMs,43 and found that the MSIS-29 and LMSQoL had the highest overall reliability among the most common MS PROMs. However, both scored poorly on validity due to lack of patient involvement during development. This questions the overall capability of existing MS PROMs to accurately and consistently assess PROs in persons with MS.
“Feed-Forward” PROMs
Oliver and colleagues16 have described “feed-forward” PROM applications in MS care in a community hospital setting using a learning health system approach. This MS clinic uses feed-forward PROs to inform clinical care—PRO data are gathered before the clinic visit and analyzed ahead of or during the clinic visit by the clinician. Patients are asked to arrive early and complete a questionnaire comprised of PROMs measuring disability, functioning, quality of life, cognitive ability, pain, fatigue, sleep quality, anxiety, and depression. Clinicians score the PROMs and input scores into the electronic health record before the clinical encounter. During the clinic visit, PROM data is visually displayed so that the clinician and patient can discuss results and use the data to better inform decision-making. The visual data display contains longitudinal information, displaying trends in health status across multiple domains, and includes specified thresholds for clinically active symptom levels (Figure).16 Longitudinal monitoring of PROM data allows for real-time assessment of goal-related progress throughout treatment. As illustrated previously by Marion’s case study, the use of real-time feed-forward PROM data can strengthen the partnership between patient and clinician as well as improve empowerment, engagement, self-monitoring, and adherence.

PRO Dashboards
Performance dashboards are increasingly used in health care to visually display clinical and PRO data for individual patients, systems, and populations over time. Dashboards display a parsimonious group of critically important measures to give clinicians and patients a longitudinal view of PRO status. They can inform decision-making in clinical care, operations, health care improvement efforts, and population health initiatives.58 Effective dashboards allow for user customization with meaningful measures, knowledge discovery for analysis of health problems, accessibility of health information, clear visualization, alerts for unexpected data values, and system connectivity.59,60 Appropriate development of PRO dashboards requires meaningful patient and clinician involvement via focus groups and key informant interviews, Delphi process approaches to prioritize and finalize selection of priority measures, iterative building of the interface with design input from key informants and stakeholders (co-design), and pilot testing to assess feasibility and acceptability of use.61-63
Other Applications of PROs/PROMs in MS
Learning Health Systems
The National Quality Forum (NQF) and the Centers for Medicaid and Medicare Services have adopted PROs for use in quality measurement.64-66 This includes a movement towards the use of LHS, defined as a health system in which information from patients and clinicians is systematically collected and synthesized with external evidence to inform clinical care, improvement, and research.67-70 Often a LHS is undertaken as a collaborative effort between multiple health care centers to improve quality and outcomes of care.70 The MS Continuous Quality Improvement Collaborative (MS-CQI), the first multi-center systems-level health care improvement research collaborative for MS,71 as well as IBD Qorus and the Cystic Fibrosis Care Center Network utilize LHS approaches.72-77
IBD Qorus is a LHS developed by the Crohn’s and Colitis Foundation that uses performance dashboards to better inform clinical care for people with inflammatory bowel disease. It also employs system-level dashboards for performance benchmarking in quality improvement initiatives and aggregate-level dashboards to assess population health status.78,79 MS-CQI uses a LHS approach to inform the improvement of MS care across multiple centers using a comprehensive dashboard, including PROMs, for benchmarking and to monitor system and population health status. MS-CQI collects PROMs using a secure online platform that can be accessed by persons with MS and their clinicians and also includes a journaling feature for collecting qualitative information and for reference and self-monitoring.71
MS Research
PROMs are used in clinical and epidemiological research to evaluate many aspects of MS, including the FAMS, the PDSS, the Fatigue Impact Scale (FIS), and others.80-82 For example, the PROMIS FatigueMS and the Fatigue Performance Scale have been used to assess the impact of MS-related fatigue on social participation.83 Generic and MS-specific PROMs have been used to assess pain levels for people with MS,84-87 and multiple MS-specific PROMs, like PRIMUS and MSQoL-54,43 as well as the SF-3639 include pain assessment scales. PROMs have also been used to assess MS-related bladder, bowel, and sexual dysfunction. Urgency, frequency, and incontinence affect up to 75% of patients with MS,88 and many PROMs, such as the LMSQoL, MUSIQoL, and the MSQoL-54, are able to evaluate bladder control and sexual functioning.43,89
PROMs are employed in MS clinical trials to help assess the tolerability and effectiveness of DMTs.90,91 PROs have been used as secondary endpoints to understand the global experience of a DMT from the patient perspective.92-94 There are 15 FDA-approved DMTs for MS, and clinical trials for 6 of these have used PROMs as an effectiveness end point.54,91,95,96 However, most DMT clinical trials are powered for MRI, relapse rate, or disease progression primary outcomes rather than PROMs, often resulting in underpowered PROM analyses.97 In addition, many PROMs are not appropriate for use in DMT clinical trials.98,99
In order to bridge the gap between clinical research and practice, some industry entities are championing “patient-focused drug development” approaches. The Accelerated Cure Project for MS has launched iConquerMS, which collects PROMs from persons with MS to further PRO research in MS and follows 4700 individuals with MS worldwide.100 In 2018, the American College of Physicians announced a collaboration with an industry partner to share data to inform DMT clinical trials and develop and validate PROMs specifically designed for DMT clinical trials.101
Population Health
Registries following large cohorts of people with MS have the potential to develop knowledge about disease progression, treatment patterns, and outcomes.102 The Swedish EIMS study has identified associations between pre-disease body mass index and MS prognosis,102 alcohol and tobacco consumption affecting MS risk,103,104 and exposure to shift work at a young age and increased MS risk.105 The North American Research Committee on MS83,106,107 and iConquerMS registries are “PROM-driven” and have been useful in identifying reductions in disease progression in people using DMTs.107,108 The New York State MS Consortium has identified important demographic characteristics that influence MS progression.109,110 PROs can also be used to determine risk of MS-related mortality111 and decline in quality of life.112,113 Limitations of these approaches include use of different PROMs, inconsistencies in data collection processes, and different follow-up intervals used across registries.102
Patient-Centered Care
The Institute of Medicine defines patient-centeredness as “care that is respectful and responsive to individual patient preferences, needs, and values and ensures that patient values guide all clinical decisions.”114 PROs are useful for identifying a patient’s individual health concerns and preferences, something that is needed when treating a highly variable chronic health condition like MS. The use of PROs can help clinicicans visualize the lived experience of persons with MS and identify personal preferences,115 as well as improve self-monitoring, self-management, self-efficacy, adherence, wellness, and coping ability.116 At the system level, PROs can inform improvement initiatives and patient-centered care design efforts.117-120
Selecting PROMs
Initiatives from groups like the COnsensus-based Standards for the Selection of health Measurement INstruments (COSMIN)121 and the International Society for Quality of Life Research (ISOQOL)108 offer guidance on selecting PROs. The NINDS has promoted common data collection between clinical studies of the brain and nervous system.122 General guidance from these sources recommends first considering the outcome and target population, selecting PROMs to measure the outcome through a synthesis of the available evidence, assessing validity and reliability of selected PROMs, and using standard measures that can be compared across studies or populations.108,121 Other factors include feasibility, acceptability, and burden of use for patients, clinicians, and systems, as well as literacy, cultural, and linguistic factors.123
The NQF recommends that consideration be given to individual patient needs, insurance factors, clinical setting constraints, and available resources when selecting PROMs.124 To maximize response rate, PROMs that are sensitive, reliable, valid, and developed in a comparative demographic of patients are advised.125 ISOQOL has released a User’s Guide and several companion guides on implementing and utilizing PROMs.108,126,127 Finally, PRO-Performance Measures (PRO-PMs) are sometimes used to assess whether PROMs are appropriately contributing to performance improvement and accountability.124
The Cons of PROs
PROs can disrupt busy clinical care environments and overextend clinical staff.125 Online collection of PROs outside of clinical encounters can relieve PRO-related burden, but this requires finding and funding appropriate secure online networks to effectively collect PROs.128 In 2015, only 60% of people seen for primary care visits could access or view their records online, and of those, only 57% used messaging for medical questions or concerns.129 Ideally, online patient portal or mobile health apps could synchronize directly to electronic health records or virtual scribes to transfer patient communications into clinical documentation.130 There has been limited success with this approach in European countries131 and with some chronic illness conditions in the United States.74
Electronic health technologies, including mobile health (mHealth) solutions, have improved the self-monitoring and self-management capability of patients with MS via information sharing in patient networks, assistive technologies, smartphone applications, and wearable devices.132,133 A recent study found that communication modes included secure online patient portal use (29%) and email use (21%), and among those who owned tablets or smartphones, 46% used mHealth apps.134 Social media use has been associated with increased peer/social/emotional support and increased access to health information, as well as clinical monitoring and behavior change.134,135 Individuals using mHealth apps are younger, have comorbidities, and have higher socioeconomic and education levels,135,136 suggesting that inequities in mHealth access exist.
Questionnaires can be time-consuming and cause mental distress if not appropriately facilitated.137 Decreasing questionnaire length and providing the option for PROMs to be delivered and completed online or outside of the clinic context can reduce burden.138 Additionally, while some people are consistent in sharing their PROs, others struggle with using computers, especially while experiencing severe symptoms, forget to complete PROMs, or simply do not have internet access due to financial or geographic constraints.139 A group of disabled and elderly persons with MS reported barriers to internet use due to visual deficits, small website font sizes, and distracting color schemes.140
Interpretability
Interpreting PROMs and displays of longitudinal PROM data can be a challenge for persons with MS and their clinicians. There is little standardization in how PROMs are scored and presented, and there is often confusion about thresholds for clinical significance and how PROM scores can be compared to other PROMs.141,142 While guidelines exist for implementing PRO scores in clinical settings,126,143 there are few that aid PROM interpretation. As a result, clinicians often seek research evidence for PROMs used in other similar patient populations as a benchmark,142-144 or compare them to other patients seen in their clinical practice.
Longitudinal PRO data are usually displayed in simple line graphs.145,146 Overall, line graphs have been found to have the highest ease of understanding by both patients and clinicians, but sometimes can be confusing.147 For example, upward trending lines are usually viewed as improvement and downward trending lines as decline; however, upward trending scores on a PROM can indicate decline, such as increasing fatigue severity. Annotation of visual displays can help. Patients and clinicians find that employing thresholds and color coding is useful, and better than “stoplight” red-yellow-green shading schemes or red-circle formats to indicate data that warrant attention.142
PROs are not free of risk for error, especially if they are used independently of other information sources, such as clinical interview, examination, and diagnostic testing, or if they are utilized too frequently, too infrequently, or are duplicated in practice. If a PRO instrument is employed too frequently, score changes may reflect learning effects rather than actual clinical status. Conversely, if used too infrequently, PRO information will not be timely enough to inform real-time clinical practice. Duplication of PRO assessments (eg, multiple measures of the same PRO for the same patient on the same day) or use of multiple PRO measures to assess the same aspect (eg, 2 measures used to assess fatigue) could introduce unnecessary complexity and confusion to interpretation of PRO results.
PRO measures also can be biased or modified by clinical status and/or perceptions of people with MS at the time of assessment. For example, cognitive impairment, whether at baseline state or due to a cognitive MS relapse event, could impact patients’ ability to understand and respond to PRO assessments, producing erroneous results. However, when used appropriately, PROs targeting cognitive dysfunction may be able to detect onset of cognitive events or help to measure recovery from them. Finally, PROs measure perceived (self-reported) status, which may not be an accurate depiction of actual status.
All of these potential pitfalls support the argument that PROs should be utilized to augment the clinical interview, examination, and diagnostic (objective) testing aspects of comprehensive MS care. In this way, PROs can be correlated with other information sources to deepen the shared understanding of health status between a person with MS and her clinician, increasing the potential to make better treatment decisions and care plans together in partnership.
Value and Cost
National groups such as the Patient-Centered Outcomes Research Institute (PCORI) are working with regulatory bodies, funding agencies, insurance providers, patient advocacy groups, researchers, providers, and specialty groups to investigate how PROMs can be implemented into value-based health care reforms, including value-based reimbursement.148 However, practical PRO implementation requires considerable time and resources, and many methodological and operational questions must be addressed before widespread adoption and reimbursement for PROMs will be feasible.148,149
Summary
PROs can generate valuable information about perceived health status, function, quality of life, and experience of care using self-reported sources. Validated PRO assessment tools include PROMs and PREMs. PROs are currently utilized in research settings (especially PROMs) but are also being used in clinical practice, quality improvement initiatives, and population health applications using LHS approaches. PROs have the advantages of empowering and informing persons with MS and clinicians to optimize patient-centered care, improve systems of care, and study population health outcomes. Barriers include PROM validity, reliability, comparability, specificity, interpretability, equity, time, and cost. Generic PROMs and PREMs, and some MS-specific PROMs, can be used for persons with MS. Unfortunately, no PREMs have been developed specifically for persons with MS, and this is an area for future research. With appropriate development and utilization in LHS applications, PROs can inform patient-centered clinical care, system-level improvement initiatives, and population health research, and have the potential to help facilitate coproduction of health care services.
Acknowledgments: The authors thank Ann Cabot, DO, of the MS Specialty Care Program at Concord Hospital and (especially) peer mentors from a peer outreach wellness program for people with MS (who have asked to remain anonymous) for interviews conducted with their permission to inform the case study described in this article. The case study used in this manuscript has been de-identified, with some aspects modified from actual, and the person in the case study is fictitiously named.
Corresponding author: Brant Oliver, PhD, MS, MPH, APRN-BC, Department of Community & Family Medicine, Dartmouth-Hitchcock Medical Center, Lebanon, NH 03756; [email protected].
Financial disclosures: None.
From the Dartmouth Institute for Health Policy & Clinical Practice, Geisel School of Medicine, Hanover, NH (Ms. Manohar and Dr. Oliver), the Department of Community & Family Medicine, Dartmouth-Hitchcock Medical Center, Lebanon, NH (Ms. Perkins, Ms. Laurion, and Dr. Oliver), and the Multiple Sclerosis Specialty Care Program, Concord Hospital, Concord, NH (Dr. Oliver).
Abstract
- Background: Patient-reported outcomes (PROs), including patient-reported outcome measures (PROMs) and patient-reported experience measures (PREMs), can be used to assess perceived health status, functioning, quality of life, and experience of care. Complex chronic illnesses such as multiple sclerosis (MS) affect multiple aspects of health, and PROs can be applied in assessment and decision-making in MS care as well as in research pertaining to MS.
- Objective: To provide a general review of PROs, with a specific focus on implications for MS care.
- Methods: Evidence synthesis of available literature on PROs in MS care.
- Results: PROs (including PROMs and PREMs) have historically been utilized in research and are now being applied in clinical, improvement, and population health settings using learning health system approaches in many disease populations, including MS. Many challenges complicate the use of PROs in MS care, including reliability, validity, and interpretability of PROMs, as well as feasibility barriers due to time and financial constraints in clinical settings.
- Conclusion: PROs have the potential to better inform clinical care, empower patient-centered care, inform health care improvement efforts, and create the conditions for coproduction of health care services.
Keywords: PRO; PROM; patient-reported outcome measure; patient-reported experience measure; quality of life; patient-centered care.
Multiple sclerosis (MS) is a disabling, complex, chronic, immune-mediated disorder of the central nervous system (CNS). MS causes inflammatory and degenerative damage in the CNS, which disrupts signaling pathways.1 It is most commonly diagnosed in young adults and affects 2.3 million people worldwide.2 People with MS experience very different disease courses and a wide range of neurological symptoms, including visual, somatic, mental health, sensory, motor, and cognitive problems.1-3 Relapsing-remitting MS, the most common form, affects 85% of those with MS and is characterized by periods of relapse (exacerbation) and remission.1 Other forms of MS (primary progressive and secondary progressive MS) are characterized by progressive deterioration and worsening symptom severity without exacerbations. Disease-modifying therapies (DMTs) can reduce the frequency of exacerbations and disability progression, but unfortunately there is no cure for MS. Treatment is focused on increasing quality of life, minimizing disability, and maximizing wellness.
Patient-reported outcomes (PROs) describe the perceived health status, function, and/or experience of a person as obtained by direct self-report. Patient-reported outcome measures (PROMs) are validated PROs that can be used to inform clinical care,4 and have demonstrated effectiveness in improving patient-provider communication and decision-making.5-7 PROMs are currently used in some MS clinical trials to determine the impact of experimental interventions,8-10 and are also being used to inform and improve clinical care in some settings. Especially for persons with MS, they can provide individualized perspectives about health experience and outcomes.11 In more advanced applications, PROMs can be used to improve face-to-face collaborations between clinicians and patients and to inform patient-centered systems of care.12-14 PROMs can also be used to inform systems-level improvement for entire patient populations.15,16
In this article, we review current applications of PROs and PROMs in the care of persons with MS, as well as current limitations and barriers to their use.
At a recent visit to her neurologist, Marion reviews her health diary, in which she has been tracking her fatigue levels throughout the day and when she has to visit the bathroom. The PRO diary also helps her remember details that she might not otherwise be able to recall at the time of her clinic visit. They review the diary entrees together to develop a shared understanding of what Marion has been experiencing and identify trends in the PRO data. They discuss symptom management and use the PRO information from the diary to help guide adjustments to her physical therapy routine and medication regimen.
Part of Marion’s “PRO package” includes the Center for Epidemiologic Studies Depression Scale (CES-D), a validated depression screening and symptom severity questionnaire that she completes every 3 months. Although she denies being depressed, she has noticed that her CES-D scores in recent months have been consistently increasing. This prompts a discussion about mental health in MS and a referral to work on depression with the MS mental health specialist. Marion and the mental health specialist use CES-D measures at baseline and during treatment to set a remission target and to track progress during treatment. Marion finds this helpful because she says it is hard for her to “wrap my hands around depression… it’s not something that there is a blood test or a MRI for.” Marion is encouraged by being able to see her CES-D scores change as her depression severity decreases, and this helps motivate her to keep engaged in treatment.
PROs and PROMs: General Applications
PROs are measures obtained directly from an individual without a priori interpretation by a clinician.9,17 PROs capture individual perspectives on symptoms, capability, disability, and health-related quality of life.9 With increasing emphasis on patient-centered care,18 individual perspectives and preferences elicited using PROMs may be able to inform better quality of care and patient-centered disease treatment and management.19-21
PROMs are standardized, validated questionnaires used to assess PROs and can be generic or condition-specific. Generic PROMs can be used in any patient population. The SF-3622 is a set of quality of life measures that assess perceived ability to complete physical tasks and routine activities, general health status, fatigue, social functioning, pain, and emotional and mental health.23 Condition-specific PROMs can be used for particular patient populations and are helpful in identifying changes in health status for a specific disease, disability, or surgery. For example, the PDQ-39 assesses 8 dimensions of daily living, functioning, and well-being for people with Parkinson’s disease.24
PROMs have been used in some MS clinical trials and research studies to determine the effectiveness of experimental treatments from the viewpoint of study participants.9,25,26 PROs can also be utilized in clinical care to facilitate communication of needs and track health outcomes,27 and can inform improvement in outcomes for health systems and populations. They can also be used to assess experience of care,28 encouraging a focus on high-quality outcomes through PRO-connected reimbursement mechanisms,29 and provide aggregate data to evaluate clinical practice, population health outcomes, and the effectiveness of public policies.27
Patient-reported experience measures (PREMs) assess patient satisfaction and experience of health care.30,31 CollaboRATE32 is a PREM that assesses the degree of shared decision-making occurring between patients and clinicians during clinical care. PREMs are currently used for assessing self-efficacy and in shared decision-making and health care improvement applications. PREMs have yet to be developed specifically for persons with MS.
PROMs in MS Care
Generic PROMs have shown that persons with MS are disproportionately burdened by poor quality of life.33-35 Other generic PROMs, like the SF-36,36 the Sickness Impact Profile,37 and versions of the Health Utilities Index,38 can be used to gather information on dysfunction and to determine quality and duration of life modified by MS-related dysfunction and disability. MS-specific PROMs are used to assess MS impairments, including pain, fatigue, cognition, sexual dysfunction, and depression.12,39-42 PROMs have also been used in MS clinical trials, including the Multiple Sclerosis Impact Scale-29 (MSIS-29),43,44 the Leeds MS QoL (LMSQoL),45,46 the Functional Assessment of MS (FAMS),47 the Hamburg Quality of Life Questionnaire in MS (HAQUAMS),48 the MS Quality of Life-54 (MSQoL-54),49 the MS International QoL (MUSIQoL),50 and the Patient-Reported Indices for MS Activity Limitations Scale (PRIMUS).51
Condition-specific PROMs are more sensitive to changes in health status and functioning for persons with MS compared to generic PROMs. They are also more reliable during MS remission and relapse periods.44,52 For example, the SF-36 has floor and ceiling effects in MS populations—a high proportion of persons with MS are scored at the maximum or minimum levels of the scale, limiting discriminant capability.22 As a result, a “combined approach” using both generic and MS-specific measures is often recommended.53 Some MS PROMs (eg, MSQoL-54) include generic questions found in the SF-36 as well as additional MS-specific questions or scales.
The variety of PROMs available (see Table for a selected listing) introduces a significant challenge to using them—limited generalizability and difficulty comparing PROs across MS studies. Efforts to establish common PROMs have been undertaken to address this problem.54 The National Institute of Neurologicical Disorders and Stroke (NINDS) sponsored the development of a neurological quality of life battery, the Neuro-QOL.55 Neuro-QOL measures the physical, mental, and social effects of neurological conditions in adults and children with neurological disorders and has the capability to facilitate comparisons across different neurological conditions. Additionally, the Patient-Reported Outcomes Measure Information System (PROMIS) has been developed to assess physical, mental, and social effects of chronic disease. PROMIS has a hybrid design that includes generic and MS-specific measures (such as PROMIS FatigueMS).56 PROMIS can be used to assess persons with MS as well as to compare the MS population with other populations with chronic illness.

PROMs have varying levels of reliability and validity. The Evaluating the Measure of Patient-Reported Outcomes57 study evaluated the development process of MS PROMs,43 and found that the MSIS-29 and LMSQoL had the highest overall reliability among the most common MS PROMs. However, both scored poorly on validity due to lack of patient involvement during development. This questions the overall capability of existing MS PROMs to accurately and consistently assess PROs in persons with MS.
“Feed-Forward” PROMs
Oliver and colleagues16 have described “feed-forward” PROM applications in MS care in a community hospital setting using a learning health system approach. This MS clinic uses feed-forward PROs to inform clinical care—PRO data are gathered before the clinic visit and analyzed ahead of or during the clinic visit by the clinician. Patients are asked to arrive early and complete a questionnaire comprised of PROMs measuring disability, functioning, quality of life, cognitive ability, pain, fatigue, sleep quality, anxiety, and depression. Clinicians score the PROMs and input scores into the electronic health record before the clinical encounter. During the clinic visit, PROM data is visually displayed so that the clinician and patient can discuss results and use the data to better inform decision-making. The visual data display contains longitudinal information, displaying trends in health status across multiple domains, and includes specified thresholds for clinically active symptom levels (Figure).16 Longitudinal monitoring of PROM data allows for real-time assessment of goal-related progress throughout treatment. As illustrated previously by Marion’s case study, the use of real-time feed-forward PROM data can strengthen the partnership between patient and clinician as well as improve empowerment, engagement, self-monitoring, and adherence.

PRO Dashboards
Performance dashboards are increasingly used in health care to visually display clinical and PRO data for individual patients, systems, and populations over time. Dashboards display a parsimonious group of critically important measures to give clinicians and patients a longitudinal view of PRO status. They can inform decision-making in clinical care, operations, health care improvement efforts, and population health initiatives.58 Effective dashboards allow for user customization with meaningful measures, knowledge discovery for analysis of health problems, accessibility of health information, clear visualization, alerts for unexpected data values, and system connectivity.59,60 Appropriate development of PRO dashboards requires meaningful patient and clinician involvement via focus groups and key informant interviews, Delphi process approaches to prioritize and finalize selection of priority measures, iterative building of the interface with design input from key informants and stakeholders (co-design), and pilot testing to assess feasibility and acceptability of use.61-63
Other Applications of PROs/PROMs in MS
Learning Health Systems
The National Quality Forum (NQF) and the Centers for Medicaid and Medicare Services have adopted PROs for use in quality measurement.64-66 This includes a movement towards the use of LHS, defined as a health system in which information from patients and clinicians is systematically collected and synthesized with external evidence to inform clinical care, improvement, and research.67-70 Often a LHS is undertaken as a collaborative effort between multiple health care centers to improve quality and outcomes of care.70 The MS Continuous Quality Improvement Collaborative (MS-CQI), the first multi-center systems-level health care improvement research collaborative for MS,71 as well as IBD Qorus and the Cystic Fibrosis Care Center Network utilize LHS approaches.72-77
IBD Qorus is a LHS developed by the Crohn’s and Colitis Foundation that uses performance dashboards to better inform clinical care for people with inflammatory bowel disease. It also employs system-level dashboards for performance benchmarking in quality improvement initiatives and aggregate-level dashboards to assess population health status.78,79 MS-CQI uses a LHS approach to inform the improvement of MS care across multiple centers using a comprehensive dashboard, including PROMs, for benchmarking and to monitor system and population health status. MS-CQI collects PROMs using a secure online platform that can be accessed by persons with MS and their clinicians and also includes a journaling feature for collecting qualitative information and for reference and self-monitoring.71
MS Research
PROMs are used in clinical and epidemiological research to evaluate many aspects of MS, including the FAMS, the PDSS, the Fatigue Impact Scale (FIS), and others.80-82 For example, the PROMIS FatigueMS and the Fatigue Performance Scale have been used to assess the impact of MS-related fatigue on social participation.83 Generic and MS-specific PROMs have been used to assess pain levels for people with MS,84-87 and multiple MS-specific PROMs, like PRIMUS and MSQoL-54,43 as well as the SF-3639 include pain assessment scales. PROMs have also been used to assess MS-related bladder, bowel, and sexual dysfunction. Urgency, frequency, and incontinence affect up to 75% of patients with MS,88 and many PROMs, such as the LMSQoL, MUSIQoL, and the MSQoL-54, are able to evaluate bladder control and sexual functioning.43,89
PROMs are employed in MS clinical trials to help assess the tolerability and effectiveness of DMTs.90,91 PROs have been used as secondary endpoints to understand the global experience of a DMT from the patient perspective.92-94 There are 15 FDA-approved DMTs for MS, and clinical trials for 6 of these have used PROMs as an effectiveness end point.54,91,95,96 However, most DMT clinical trials are powered for MRI, relapse rate, or disease progression primary outcomes rather than PROMs, often resulting in underpowered PROM analyses.97 In addition, many PROMs are not appropriate for use in DMT clinical trials.98,99
In order to bridge the gap between clinical research and practice, some industry entities are championing “patient-focused drug development” approaches. The Accelerated Cure Project for MS has launched iConquerMS, which collects PROMs from persons with MS to further PRO research in MS and follows 4700 individuals with MS worldwide.100 In 2018, the American College of Physicians announced a collaboration with an industry partner to share data to inform DMT clinical trials and develop and validate PROMs specifically designed for DMT clinical trials.101
Population Health
Registries following large cohorts of people with MS have the potential to develop knowledge about disease progression, treatment patterns, and outcomes.102 The Swedish EIMS study has identified associations between pre-disease body mass index and MS prognosis,102 alcohol and tobacco consumption affecting MS risk,103,104 and exposure to shift work at a young age and increased MS risk.105 The North American Research Committee on MS83,106,107 and iConquerMS registries are “PROM-driven” and have been useful in identifying reductions in disease progression in people using DMTs.107,108 The New York State MS Consortium has identified important demographic characteristics that influence MS progression.109,110 PROs can also be used to determine risk of MS-related mortality111 and decline in quality of life.112,113 Limitations of these approaches include use of different PROMs, inconsistencies in data collection processes, and different follow-up intervals used across registries.102
Patient-Centered Care
The Institute of Medicine defines patient-centeredness as “care that is respectful and responsive to individual patient preferences, needs, and values and ensures that patient values guide all clinical decisions.”114 PROs are useful for identifying a patient’s individual health concerns and preferences, something that is needed when treating a highly variable chronic health condition like MS. The use of PROs can help clinicicans visualize the lived experience of persons with MS and identify personal preferences,115 as well as improve self-monitoring, self-management, self-efficacy, adherence, wellness, and coping ability.116 At the system level, PROs can inform improvement initiatives and patient-centered care design efforts.117-120
Selecting PROMs
Initiatives from groups like the COnsensus-based Standards for the Selection of health Measurement INstruments (COSMIN)121 and the International Society for Quality of Life Research (ISOQOL)108 offer guidance on selecting PROs. The NINDS has promoted common data collection between clinical studies of the brain and nervous system.122 General guidance from these sources recommends first considering the outcome and target population, selecting PROMs to measure the outcome through a synthesis of the available evidence, assessing validity and reliability of selected PROMs, and using standard measures that can be compared across studies or populations.108,121 Other factors include feasibility, acceptability, and burden of use for patients, clinicians, and systems, as well as literacy, cultural, and linguistic factors.123
The NQF recommends that consideration be given to individual patient needs, insurance factors, clinical setting constraints, and available resources when selecting PROMs.124 To maximize response rate, PROMs that are sensitive, reliable, valid, and developed in a comparative demographic of patients are advised.125 ISOQOL has released a User’s Guide and several companion guides on implementing and utilizing PROMs.108,126,127 Finally, PRO-Performance Measures (PRO-PMs) are sometimes used to assess whether PROMs are appropriately contributing to performance improvement and accountability.124
The Cons of PROs
PROs can disrupt busy clinical care environments and overextend clinical staff.125 Online collection of PROs outside of clinical encounters can relieve PRO-related burden, but this requires finding and funding appropriate secure online networks to effectively collect PROs.128 In 2015, only 60% of people seen for primary care visits could access or view their records online, and of those, only 57% used messaging for medical questions or concerns.129 Ideally, online patient portal or mobile health apps could synchronize directly to electronic health records or virtual scribes to transfer patient communications into clinical documentation.130 There has been limited success with this approach in European countries131 and with some chronic illness conditions in the United States.74
Electronic health technologies, including mobile health (mHealth) solutions, have improved the self-monitoring and self-management capability of patients with MS via information sharing in patient networks, assistive technologies, smartphone applications, and wearable devices.132,133 A recent study found that communication modes included secure online patient portal use (29%) and email use (21%), and among those who owned tablets or smartphones, 46% used mHealth apps.134 Social media use has been associated with increased peer/social/emotional support and increased access to health information, as well as clinical monitoring and behavior change.134,135 Individuals using mHealth apps are younger, have comorbidities, and have higher socioeconomic and education levels,135,136 suggesting that inequities in mHealth access exist.
Questionnaires can be time-consuming and cause mental distress if not appropriately facilitated.137 Decreasing questionnaire length and providing the option for PROMs to be delivered and completed online or outside of the clinic context can reduce burden.138 Additionally, while some people are consistent in sharing their PROs, others struggle with using computers, especially while experiencing severe symptoms, forget to complete PROMs, or simply do not have internet access due to financial or geographic constraints.139 A group of disabled and elderly persons with MS reported barriers to internet use due to visual deficits, small website font sizes, and distracting color schemes.140
Interpretability
Interpreting PROMs and displays of longitudinal PROM data can be a challenge for persons with MS and their clinicians. There is little standardization in how PROMs are scored and presented, and there is often confusion about thresholds for clinical significance and how PROM scores can be compared to other PROMs.141,142 While guidelines exist for implementing PRO scores in clinical settings,126,143 there are few that aid PROM interpretation. As a result, clinicians often seek research evidence for PROMs used in other similar patient populations as a benchmark,142-144 or compare them to other patients seen in their clinical practice.
Longitudinal PRO data are usually displayed in simple line graphs.145,146 Overall, line graphs have been found to have the highest ease of understanding by both patients and clinicians, but sometimes can be confusing.147 For example, upward trending lines are usually viewed as improvement and downward trending lines as decline; however, upward trending scores on a PROM can indicate decline, such as increasing fatigue severity. Annotation of visual displays can help. Patients and clinicians find that employing thresholds and color coding is useful, and better than “stoplight” red-yellow-green shading schemes or red-circle formats to indicate data that warrant attention.142
PROs are not free of risk for error, especially if they are used independently of other information sources, such as clinical interview, examination, and diagnostic testing, or if they are utilized too frequently, too infrequently, or are duplicated in practice. If a PRO instrument is employed too frequently, score changes may reflect learning effects rather than actual clinical status. Conversely, if used too infrequently, PRO information will not be timely enough to inform real-time clinical practice. Duplication of PRO assessments (eg, multiple measures of the same PRO for the same patient on the same day) or use of multiple PRO measures to assess the same aspect (eg, 2 measures used to assess fatigue) could introduce unnecessary complexity and confusion to interpretation of PRO results.
PRO measures also can be biased or modified by clinical status and/or perceptions of people with MS at the time of assessment. For example, cognitive impairment, whether at baseline state or due to a cognitive MS relapse event, could impact patients’ ability to understand and respond to PRO assessments, producing erroneous results. However, when used appropriately, PROs targeting cognitive dysfunction may be able to detect onset of cognitive events or help to measure recovery from them. Finally, PROs measure perceived (self-reported) status, which may not be an accurate depiction of actual status.
All of these potential pitfalls support the argument that PROs should be utilized to augment the clinical interview, examination, and diagnostic (objective) testing aspects of comprehensive MS care. In this way, PROs can be correlated with other information sources to deepen the shared understanding of health status between a person with MS and her clinician, increasing the potential to make better treatment decisions and care plans together in partnership.
Value and Cost
National groups such as the Patient-Centered Outcomes Research Institute (PCORI) are working with regulatory bodies, funding agencies, insurance providers, patient advocacy groups, researchers, providers, and specialty groups to investigate how PROMs can be implemented into value-based health care reforms, including value-based reimbursement.148 However, practical PRO implementation requires considerable time and resources, and many methodological and operational questions must be addressed before widespread adoption and reimbursement for PROMs will be feasible.148,149
Summary
PROs can generate valuable information about perceived health status, function, quality of life, and experience of care using self-reported sources. Validated PRO assessment tools include PROMs and PREMs. PROs are currently utilized in research settings (especially PROMs) but are also being used in clinical practice, quality improvement initiatives, and population health applications using LHS approaches. PROs have the advantages of empowering and informing persons with MS and clinicians to optimize patient-centered care, improve systems of care, and study population health outcomes. Barriers include PROM validity, reliability, comparability, specificity, interpretability, equity, time, and cost. Generic PROMs and PREMs, and some MS-specific PROMs, can be used for persons with MS. Unfortunately, no PREMs have been developed specifically for persons with MS, and this is an area for future research. With appropriate development and utilization in LHS applications, PROs can inform patient-centered clinical care, system-level improvement initiatives, and population health research, and have the potential to help facilitate coproduction of health care services.
Acknowledgments: The authors thank Ann Cabot, DO, of the MS Specialty Care Program at Concord Hospital and (especially) peer mentors from a peer outreach wellness program for people with MS (who have asked to remain anonymous) for interviews conducted with their permission to inform the case study described in this article. The case study used in this manuscript has been de-identified, with some aspects modified from actual, and the person in the case study is fictitiously named.
Corresponding author: Brant Oliver, PhD, MS, MPH, APRN-BC, Department of Community & Family Medicine, Dartmouth-Hitchcock Medical Center, Lebanon, NH 03756; [email protected].
Financial disclosures: None.
1. Goldenberg MM. Multiple sclerosis review. PT. 2012;37:175-184.
2. Doshi A, Chataway J. Multiple sclerosis, a treatable disease. Clin Med. 2016;16(Suppl 6):s53-s9.
3. Perrin Ross A. Management of multiple sclerosis. Am J Managed Care. 2013;19(16 Suppl):s301-s306.
4. Batalden M, Batalden P, Margolis P, et al. Coproduction of healthcare service. BMJ Qual Saf. 2016;25:509-517.
5. Chen J, Ou L, Hollis SJ. A systematic review of the impact of routine collection of patient reported outcome measures on patients, providers and health organisations in an oncologic setting. BMC Health Serv Res. 2013;13:211.
6. Knaup C, Koesters M, Schoefer D, et al. Effect of feedback of treatment outcome in specialist mental healthcare: meta-analysis. Br J Psychiatr. 2009;195:15-22.
7. Dudgeon D. The impact of measuring patient-reported outcome measures on quality of and access to palliative care. J Palliat Med. 2018;21(S1):S76-s80.
8. Snyder CF, Herman JM, White SM, et al. When using patient-reported outcomes in clinical practice, the measure matters: a randomized controlled trial. J Oncol Pract. 2014;10:e299-e306.
9. Guidance for industry: patient-reported outcome measures: use in medical product development to support labeling claims: draft guidance. Health Qual Life Outcomes. 2006;4:79.
10. de Jong MJ, Huibregtse R, Masclee AAM, et al. Patient-reported outcome measures for use in clinical trials and clinical practice in inflammatory bowel diseases: a systematic review. Clin Gastroenterol Hepatol. 2018;16:648-663.
11. Roman D, Osborne-Stafsnes J, Amy CH, et al. Early lessons from four ‘aligning forces for quality’ communities bolster the case for patient-centered care. Health Affairs. 2013;32:232-241.
12. Ehde DM, Alschuler KN, Sullivan MD, et al. Improving the quality of depression and pain care in multiple sclerosis using collaborative care: The MS-care trial protocol. Contemporary Clinical Trials. 2018;64:219-229.
13. Lavallee DC, Chenok KE, Love RM, et al. Incorporating patient-reported outcomes into health care to engage patients and enhance care. Health Affairs. 2016;35:575-582.
14. Schick-Makaroff K, Thummapol O, Thompson S, et al. Strategies for incorporating patient-reported outcomes in the care of people with chronic kidney disease (PRO kidney): a protocol for a realist synthesis. Clin Gastroenterol Hepatol. 2018;16:648-663.
15. Nelson EC, Dixon-Woods M, Batalden PB, et al. Patient focused registries can improve health, care, and science. BMJ. 2016;354:i3319.
16. Oliver BJ, Nelson EC, Kerrigan CL. Turning feed-forward and feedback processes on patient-reported data into intelligent action and informed decision-making: case studies and principles. Med Care. 2019;57 Suppl 1:S31-s37.
17. Deshpande PR, Rajan S, Sudeepthi BL, Abdul Nazir CP. Patient-reported outcomes: A new era in clinical research. Perspect Clin Res. 2011;2:137-144.
18. Epstein RM, Street RL, Jr. The values and value of patient-centered care. Ann Fam Med. 2011;9:100-103.
19. Payne SA. A study of quality of life in cancer patients receiving palliative chemotherapy. Soc Sci Med. 1992;35:1505-1509.
20. Borgaonkar MR, Irvine EJ. Quality of life measurement in gastrointestinal and liver disorders. Gut. 2000;47:444-454.
21. Macdonell R, Nagels G, Laplaud DA, et al. Improved patient-reported health impact of multiple sclerosis: The ENABLE study of PR-fampridine. Multiple Sclerosis. 2016;22:944-954.
22. Hobart J, Freeman J, Lamping D, et al. The SF-36 in multiple sclerosis: why basic assumptions must be tested. J Neurol Neurosurg Psychiatry. 2001;71:363-370.
23. Lins L, Carvalho FM. SF-36 total score as a single measure of health-related quality of life: Scoping review. SAGE Open Med. 2016;4:2050312116671725.
24. Parkinson’s Disease Society of the United Kingdom. The Parkinson’s Disease Questionnaire (PDQ-39). https://www.parkinsons.org.uk/professionals/resources/parkinsons-disease-questionnaire-pdq-39. Accessed October 20, 2019.
25. Basch E. The missing voice of patients in drug-safety reporting. N Engl J Med. 2010;362:865-869.
26. Calvert M, Brundage M, Jacobsen PB, et al. The CONSORT Patient-Reported Outcome (PRO) extension: implications for clinical trials and practice. Health Qual Life Outcomes. 2013;11:184.
27. Black N. Patient reported outcome measures could help transform healthcare. BMJ. 2013;346:f167.
28. Franks P, Fiscella K, Shields CG, et al. Are patients’ ratings of their physicians related to health outcomes? Ann Fam Med. 2005;3:229-234.
29. Hostetter M, Klein S. Using patient-reported outcomes to improve health care quality. The Commonwealth Fund 2019. www.commonwealthfund.org/publications/newsletter-article/using-patient-reported-outcomes-improve-health-care-quality. Accessed October 24, 2019.
30. Charlotte Kingsley SP. Patient-reported outcome measures and patient-reported experience measures. BJA Education. 2017;17:137-144.
31. Weldring T, Smith SM. Patient-Reported outcomes (PROs) and patient-reported outcome measures (PROMs). Health Services Insights. 2013;6:61-68.
32. Elwyn G, Barr PJ, Grande SW, et al. Developing CollaboRATE: A fast and frugal patient-reported measure of shared decision making in clinical encounters. Patient Educ Couns. 2013;93:102-107.
33. Kurtzke JF. Rating neurologic impairment in multiple sclerosis: an expanded disability status scale (EDSS). Neurology. 1983;33:1444-1452.
34. Rudick RA, Miller D, Clough JD, et al. Quality of life in multiple sclerosis. Comparison with inflammatory bowel disease and rheumatoid arthritis. Arch Neurol. 1992;49:1237-1242.
35. Benito-Leon J, Morales JM, Rivera-Navarro J, Mitchell A. A review about the impact of multiple sclerosis on health-related quality of life. Disabil Rehabil. 2003;25:1291-1303.
36. RAND Corporation. 36-Item Short Form Survey (SF-36). [https://www.rand.org/health-care/surveys_tools/mos/36-item-short-form.html. Accessed October 20, 2019.
37. Bergner M, Bobbitt RA, Carter WB, Gilson BS. The sickness impact profile: development and final revision of a health status measure. Medical Care. 1981;19:787-805.
38. Horsman J, Furlong W, Feeny D, Torrance G. The Health Utilities Index (HUI): concepts, measurement properties and applications. Health Qual Life Outcomes. 2003;1:54.
39. Kinkel RP, Laforet G, You X. Disease-related determinants of quality of life 10 years after clinically isolated syndrome. Int J MS Care. 2015;17:26-34.
40. Baumstarck-Barrau K, Simeoni MC, Reuter F, et al. Cognitive function and quality of life in multiple sclerosis patients: a cross-sectional study. BMC Neurology. 2011;11:17.
41. Samartzis L, Gavala E, Zoukos Y, et al. Perceived cognitive decline in multiple sclerosis impacts quality of life independently of depression. Rehabil Res Pract. 2014;2014:128751.
42. Vitkova M, Rosenberger J, Krokavcova M, et al. Health-related quality of life in multiple sclerosis patients with bladder, bowel and sexual dysfunction. Disabil Rehabil. 2014;36:987-992.
43. Khurana V, Sharma H, Afroz N, et al. Patient-reported outcomes in multiple sclerosis: a systematic comparison of available measures. Eur J Neurol. 2017;24:1099-1107.
44. Hobart J, Lamping D, Fitzpatrick R, et al. The Multiple Sclerosis Impact Scale (MSIS-29): a new patient-based outcome measure. Brain. 2001;124(Pt 5):962-973.
45. Lily O, McFadden E, Hensor E, et al. Disease-specific quality of life in multiple sclerosis: the effect of disease modifying treatment. Multiple Sclerosis. 2006;12:808-813.
46. Ford HL, Gerry E, Tennant A, et al. Developing a disease-specific quality of life measure for people with multiple sclerosis. Clin Rehabil. 2001;15:247-258.
47. Cella DF, Dineen K, Arnason B, et al. Validation of the functional assessment of multiple sclerosis quality of life instrument. Neurology. 1996;47:129-139.
48. Gold SM, Heesen C, Schulz H, et al. Disease specific quality of life instruments in multiple sclerosis: validation of the Hamburg Quality of Life Questionnaire in Multiple Sclerosis (HAQUAMS). Multiple Sclerosis. 2001;7:119-130.
49. Vickrey BG, Hays RD, Harooni R, et al. A health-related quality of life measure for multiple sclerosis. Qual Life Res. 1995;4:187-206.
50. Simeoni M, Auquier P, Fernandez O, et al. Validation of the Multiple Sclerosis International Quality of Life questionnaire. Multiple Sclerosis. 2008;14:219-230.
51. Doward LC, McKenna SP, Meads DM, et al. The development of patient-reported outcome indices for multiple sclerosis (PRIMUS). Multiple Sclerosis. 2009;15:1092-1102.
52. Ozakbas S, Akdede BB, Kosehasanogullari G, et al. Difference between generic and multiple sclerosis-specific quality of life instruments regarding the assessment of treatment efficacy. J Neurol Sci. 2007;256:30-34.
53. Cella DF, Wiklund I, Shumaker SA, Aaronson NK. Integrating health-related quality of life into cross-national clinical trials. Qual Life Res. 1993;2:433-440.
54. Nowinski CJ, Miller DM, Cella D. Evolution of patient-reported outcomes and their role in multiple sclerosis clinical trials. Neurotherapeutics. 2017;14:934-944.
55. Cella D, Nowinski C, Peterman A, et al. The neurology quality-of-life measurement initiative. Arch Phys Med Rehabil. 2011;92(10 Suppl):S28-S36.
56. HealthMeasures: PROMIS. http://www.healthmeasures.net/explore-measurement-systems/promis. Accessed October 20, 2019.
57. Valderas JM, Ferrer M, Mendivil J, et al. Development of EMPRO: a tool for the standardized assessment of patient-reported outcome measures. Value Health. 2008;11:700-708.
58. Bach K, Martling C, Mork PJ, et al. Design of a clinician dashboard to facilitate co-decision making in the management of non-specific low back pain. J Intelligent Helath Syst. 2019;52:269-284.
59. Karami M, Safdari R, Rahimi A. Effective radiology dashboards: key research findings. Radiology Management. 2013;35:42-45.
60. Dowding D, Randell R, Gardner P, et al. Dashboards for improving patient care: review of the literature. Int J Med Informatic. 2015;84:87-100.
61. Mlaver E, Schnipper JL, Boxer RB, et al. User-centered collaborative design and development of an inpatient safety dashboard. Jt Comm J Qual Patient Saf. 2017;43:676-685.
62. Dowding D, Merrill J, Russell D. Using feedback intervention theory to guide clinical dashboard design. AMIA Annu Symp Proc. 2018;2018:395-403.
63. Hartzler AL, Izard JP, Dalkin BL, et al. Design and feasibility of integrating personalized PRO dashboards into prostate cancer care. J Am Med Inform Assoc. 2016;23:38-47.
64. Lavallee DC, Chenok KE, Love RM, et al. Incorporating patient-reported outcomes into health care to engage patients and enhance care. Health Affairs. 2016;35:575-582.
65. U.S. Centers for Medicare & Medicaid Services. Comprehensive Care for Joint Replacement Model. https://innovation.cms.gov/initiatives/CJR. Accessed August 3, 2019.
66. National Quality Forum. Patient-reported outcomes: Accessed August 3, 2019. http://www.qualityforum.org/Patient-Reported_Outcomes.aspx. Accessed October 20, 2019.
67. Institute of Medicine Roundtable on Evidence-Based Medicine. The National Academies Collection: Reports funded by National Institutes of Health. In: Olsen LA, Aisner D, McGinnis JM, eds. The Learning Healthcare System: Workshop Summary. Washington (DC): National Academies Press (US).National Academy of Sciences; 2007.
68. About Learning Health Systems: Agency for Healthcare Research and Quality. https://www.ahrq.gov/learning-health-systems/about.html. Accessed August 3, 2019.
69. Best Care at Lower Cost: The Path to Continuously Learning Health Care in America. Washington (DC): National Academies Press; 2013.
70. Committee on Quality of Health Care in America. Crossing the Quality Chasm: A New Health System for the 21st Century. Washington, DC: National Academies Press; 2001.
71. Oliver BJ, for the MS-CQI Investigators. The Multiple Sclerosis Continuous improvement collaborative: the first coproduction learning health system improvement science research collaborative for multiple sclerosis. The International Society for Quality in Health Care (ISQua) Annual International Conference; October 2019; Cape Town, South Africa.
72. Britto MT, Fuller SC, Kaplan HC, et al. Using a network organisational architecture to support the development of Learning Healthcare Systems. BMJ Qual Saf. 2018;27:937-946.
73. Care Centers: Cystic Fibrosis Foundation. https://www.cff.org/Care/Care-Centers/. Accessed August 3, 2019.
74. Schechter MS, Fink AK, Homa K, Goss CH. The Cystic Fibrosis Foundation patient registry as a tool for use in quality improvement. BMJ Qual Saf. 2014;23(Suppl 1):i9-i14.
75. Mogayzel PJ, Dunitz J, Marrow LC, Hazle LA. Improving chronic care delivery and outcomes: the impact of the cystic fibrosis Care Center Network. BMJ Qual Saf. 2014;23(Suppl 1):i3-i8.
76. Godfrey MM, Oliver BJ. Accelerating the rate of improvement in cystic fibrosis care: contributions and insights of the learning and leadership collaborative. BMJ Qual Saf. 2014;23(Suppl 1):i23-i32.
77. Nelson EC, Godfrey MM. Quality by Design: A Clinical Microsystems Approach. San Francisco, California: Jossey-Bass; 2007.
78. Crohn’s & Colitis Foundation.Quality of Care: IBD Qorus: https://www.crohnscolitisfoundation.org/research/ibd-qorus. Accessed October 23, 2019.
79. Johnson LC, Melmed GY, Nelson EC, et al. Fostering collaboration through creation of an ibd learning health system. Am J Gastroenterol. 2017;112:406-468.
80. Fernandez-Munoz JJ, Moron-Verdasco A, Cigaran-Mendez M, et al. Disability, quality of life, personality, cognitive and psychological variables associated with fatigue in patients with multiple sclerosis. Acta Neurologica Scandinavica. 2015;132:118-124.
81. Flachenecker P, Kumpfel T, Kallmann B, et al. Fatigue in multiple sclerosis: a comparison of different rating scales and correlation to clinical parameters. Multiple Sclerosis. 2002;8:523-526.
82. Tellez N, Rio J, Tintore M, et al. Does the Modified Fatigue Impact Scale offer a more comprehensive assessment of fatigue in MS? Multiple Sclerosis. 2005;11:198-202.
83. Salter A, Fox RJ, Tyry T, et al. The association of fatigue and social participation in multiple sclerosis as assessed using two different instruments. Mult Scler Relat Disord. 2019;31:165-72.
84. Solaro C, Trabucco E, Messmer Uccelli M. Pain and multiple sclerosis: pathophysiology and treatment. Curr Neurol Neurosci Rep. 2013;13:320.
85. Jawahar R, Oh U, Yang S, Lapane KL. A systematic review of pharmacological pain management in multiple sclerosis. Drugs. 2013;73:1711-1722.
86. Rintala A, Hakkinen A, Paltamaa J. Ten-year follow-up of health-related quality of life among ambulatory persons with multiple sclerosis at baseline. Qual Life Res. 2016;25:3119-3127.
87. Tepavcevic DK, Pekmezovic T, Stojsavljevic N, et al. Change in quality of life and predictors of change among patients with multiple sclerosis: a prospective cohort study. Qual Life Res. 2014;23:1027-1037.
88. DasGupta R, Fowler CJ. Bladder, bowel and sexual dysfunction in multiple sclerosis: management strategies. Drugs. 2003;63:153-166.
89. Marck CH, Jelinek PL, Weiland TJ, et al. Sexual function in multiple sclerosis and associations with demographic, disease and lifestyle characteristics: an international cross-sectional study. BMC Neurology. 2016;16:210.
90. Gajofatto A, Benedetti MD. Treatment strategies for multiple sclerosis: When to start, when to change, when to stop? World J Clinical Cases. 2015;3:545-555.
91. National Multiple Sclerosis Society. Disease-modifying therapies for MS. 2018.
92. Rudick RA, Panzara MA. Natalizumab for the treatment of relapsing multiple sclerosis. Biologics. 2008;2:189-199.
93. Cohen JA, Cutter GR, Fischer JS, et al. Benefit of interferon beta-1a on MSFC progression in secondary progressive MS. Neurology. 2002;59:679-687.
94. Patti F, Pappalardo A, Montanari E, et al. Interferon-beta-1a treatment has a positive effect on quality of life of relapsing-remitting multiple sclerosis: results from a longitudinal study. J Neurol Sci. 2014;337:180-185.
95. Novartis. Exploring the efficacy and safety of Siponimod in patients with secondary progressive multiple sclerosis (EXPAND). Available from clinicaltrials.gov/ct2/show/NCT01665144. NLM identifier: NCT01665144.
96. Singal AG, Higgins PD, Waljee AK. A primer on effectiveness and efficacy trials. Clin Transl Gastroenterol. 2014;5:e45.
97. Jongen PJ. Health-related quality of life in patients with multiple sclerosis: impact of disease-modifying drugs. CNS Drugs. 2017;31:585-602.
98. Coretti S, Ruggeri M, McNamee P. The minimum clinically important difference for EQ-5D index: a critical review. Expert Rev Pharmacoecon Outcomes Res. 2014;14:221-233.
99. Clinical Review Report: Ocrelizumab (Ocrevus). CADTH Common Drug Review. 2018:Appendix 4, Validity of Outcome Measures.
100. iCounquer MS; Empowering patients: how individuals with ms are contributing to the fight to find a cure: 2018. https://www.iconquerms.org/empowering-patients-how-individuals-ms-are-contributing-fight-find-cure. Accessed October 23, 2019.
101. K M. Accelerated Cure Project Announces Collaboration with EMD Serono to Advance Patient-Focused Drug Development in Multiple Sclerosis2018. https://www.iconquerms.org/accelerated-cure-project-announces-collaboration-emd-serono-advance-patient-focused-drug-development. Accessed October 24, 2019.
102. Bebo BF Jr, Fox RJ, Lee K, et al. Landscape of MS patient cohorts and registries: Recommendations for maximizing impact. Multiple Sclerosis. 2018;24:579-586.
103. Hedstrom AK, Hillert J, Olsson T, Alfredsson L. Alcohol as a modifiable lifestyle factor affecting multiple sclerosis risk. JAMA Neurology. 2014;71:300-305.
104. Hedstrom AK, Baarnhielm M, Olsson T, Alfredsson L. Tobacco smoking, but not Swedish snuff use, increases the risk of multiple sclerosis. Neurology. 2009;73:696-701.
105. Hedstrom AK, Akerstedt T, Hillert J, et al. Shift work at young age is associated with increased risk for multiple sclerosis. Ann Neurol. 2011;70:733-741.
106. Hadjimichael O, Vollmer T, Oleen-Burkey M. Fatigue characteristics in multiple sclerosis: the North American Research Committee on Multiple Sclerosis (NARCOMS) survey. Health Qual Life Outcomes. 2008;6:100.
107. Fox RJ, Salter AR, Tyry T, et al. Treatment discontinuation and disease progression with injectable disease-modifying therapies: findings from the North American research committee on multiple sclerosis database. Int J MS Care. 2013;15:194-201.
108. Reeve BB, Wyrwich KW, Wu AW, et al. ISOQOL recommends minimum standards for patient-reported outcome measures used in patient-centered outcomes and comparative effectiveness research. Qual Life Res. 2013;22:1889-1905.
109. Jacobs LD, Wende KE, Brownscheidle CM, et al. A profile of multiple sclerosis: the New York State Multiple Sclerosis Consortium. Multiple Sclerosis. 1999;5:369-376.
110. Weinstock-Guttman B, Jacobs LD, Brownscheidle CM, et al. Multiple sclerosis characteristics in African American patients in the New York State Multiple Sclerosis Consortium. Multiple Sclerosis. 2003;9:293-298.
111. Raffel J, Wallace A, Gveric D, et al. Patient-reported outcomes and survival in multiple sclerosis: A 10-year retrospective cohort study using the Multiple Sclerosis Impact Scale-29. PLoS Med. 2017;14:e1002346.
112. Vaughn CB, Kavak KS, Dwyer MG, et al. Fatigue at enrollment predicts EDSS worsening in the New York State Multiple Sclerosis Consortium. Multiple Sclerosis. 2018:1352458518816619.
113. Alroughani RA, Akhtar S, Ahmed SF, Al-Hashel JY. Clinical predictors of disease progression in multiple sclerosis patients with relapsing onset in a nation-wide cohort. Int J Neuroscience. 2015;125:831-837.
114. Dolan JG, Veazie PJ, Russ AJ. Development and initial evaluation of a treatment decision dashboard. BMC Med Inform Decision Mak. 2013;13:51.
115. Stuifbergen AK, Becker H, Blozis S, et al. A randomized clinical trial of a wellness intervention for women with multiple sclerosis. Arch Phys Med Rehabil. 2003;84:467-476.
116. Finkelstein J, Martin C, Bhushan A, et al. Feasibility of computer-assisted education in patients with multiple sclerosis. In: Proceedings of the IEEE Symposium on Computer-Based Medical Systems. Bethesda, MD; 2004.
117. Batalden P. Getting more health from healthcare: quality improvement must acknowledge patient coproduction—an essay by Paul Batalden. BMJ. 2018;362:k3617.
118. Batalden PB, Davidoff F. What is “quality improvement” and how can it transform healthcare? Qual Saf Health Care. 2007;16:2-3.
119. Ovretveit J, Zubkoff L, Nelson EC, et al. Using patient-reported outcome measurement to improve patient care. Int J Qual Health Care. 2017;29:874-879.
120. Prodinger B, Taylor P. Improving quality of care through patient-reported outcome measures (PROMs): expert interviews using the NHS PROMs Programme and the Swedish quality registers for knee and hip arthroplasty as examples. BMC Health Services Res. 2018;18:87.
121. Prinsen CA, Vohra S, Rose MR, et al. How to select outcome measurement instruments for outcomes included in a “Core Outcome Set” – a practical guideline. Trials. 2016;17:449.
122. NINDS. NINDS Common Data Elements: Streamline Your Neuroscience Clinical Research National Institute of Health. https://www.commondataelements.ninds.nih.gov/#page=Default. Accessed October 23, 2019.
123. Francis DO, McPheeters ML, Noud M, et al. Checklist to operationalize measurement characteristics of patient-reported outcome measures. Systematic Rev. 2016;5:129.
124. National Quality Forum. Patient-reported outcomes (PROs) in performance measurement. 2013. https://www.qualityforum.org/Publications/2012/12/Patient-Reported_Outcomes_in_Performance_Measurement.aspx. Accessed October 15, 2019.
125. Dawson J, Doll H, Fitzpatrick R, et al. The routine use of patient reported outcome measures in healthcare settings. BMJ. 2010;340:c186.
126. Aaronson N ET, Greenlhalgh J, Halyard M, et al. User’s guide to implementation of patient reported outcomes assessment in clinical practice. International Society for Quality of Life. 2015.
127. Chan EKH, Edwards TC, Haywood K, et al. Implementing patient-reported outcomes mesaures in clinical practice: a companion guide to the ISOQOL user’s guide. Qual Life Res. 2018;28:621-627.
128. Snyder CF, Wu AW, Miller RS, et al. The role of informatics in promoting patient-centered care. Cancer J. 2011;17:211-218
129. Osborn R, Moulds D, Schneider EC, et al. Primary care physicians in ten countries report challenges caring for patients with complex health needs. Health Affairs. 2015;34:2104-2112.
130. Wu AW, Kharrazi H, Boulware LE, Snyder CF. Measure once, cut 0wice--adding patient-reported outcome measures to the electronic health record for comparative effectiveness research. J Clin Epidemiol. 2013;66(8 Suppl):S12-S20.
131. Trojano M, Bergamaschi R, Amato MP, et al. The Italian multiple sclerosis register. Neurol Sci. 2019;40:155-165.
132. Moorhead SA, Hazlett DE, Harrison L, et al. A new dimension of health care: systematic review of the uses, benefits, and limitations of social media for health communication. J Med Internet Res. 2013;15:e85.
133. Luigi L, Francesco B, Marcello M, et al. e-Health and multiple sclerosis: An update. Multiple Sclerosis J. 2018;24:1657-1664.
134. Marrie RA, Leung S, Tyry T, et al. Use of eHealth and mHealth technology by persons with multiple sclerosis. Mult Scler Relat Dis. 2019;27:13-19.
135. Giunti G, Kool J, Rivera Romero O, Dorronzoro Zubiete E. Exploring the specific needs of persons with multiple sclerosis for mhealth solutions for physical activity: mixed-methods study. JMIR Mhealth Uhealth. 2018;6:e37.
136. Spooner KK, Salemi JL, Salihu HM, Zoorob RJ. eHealth patient-provider communication in the United States: interest, inequalities, and predictors. J Am Med Inform Assoc. 2017;24(e1):e18-e27.
137. Safdar N, Abbo LM, Knobloch MJ, Seo SK. Research methods in healthcare epidemiology: survey and qualitative research. Infect Control Hosp Epidemiol. 2016;37:1272-1277.
138. Rolstad S, Adler J, Rydén A. Response burden and questionnaire length: is shorter better? A review and meta-analysis. Value Health. 2011;14:1101-1108.
139. Chiu C, Bishop M, Pionke JJ, et al. Barriers to the accessibility and continuity of health-care services in people with multiple sclerosis: a literature review. Int J MS Care. 2017;19:313-321.
140. Atreja A, Mehta N, Miller D, et al. One size does not fit all: using qualitative methods to inform the development of an Internet portal for multiple sclerosis patients. AMIA Annu Symp Proc Symp. 2005:16-20.
141. Snyder CF, Blackford AL, Brahmer JR, et al. Needs assessments can identify scores on HRQOL questionnaires that represent problems for patients: an illustration with the Supportive Care Needs Survey and the QLQ-C30. Qual Life Res. 2010;19:837-845.
142. de Vet HC, Terwee CB. The minimal detectable change should not replace the minimal important difference. J Clin Epidemiol. 2010;63:804-805.
143. Snyder CF, Aaronson NK, Choucair AK, et al. Implementing patient-reported outcomes assessment in clinical practice: a review of the options and considerations. Qual Life. 2012;21:1305-1314.
144. Myla DG, Melanie DW, Robert WM, et al. Identification and validation of clinically meaningful benchmarks in the 12-item Multiple Sclerosis Walking Scale. Multiple Sclerosis J. 2017;23:1405-1414.
145. van Munster CEP, Uitdehaag BMJ. Outcome measures in clinical trials for multiple sclerosis. CNS Drugs. 2017;31:217-236.
146. Snyder CF, Jensen R, Courtin SO, Wu AW. PatientViewpoint: a website for patient-reported outcomes assessment. Quality Life Res. 2009;18:793-800.
147. Snyder CF, Smith KC, Bantug ET, et al. What do these scores mean? Presenting patient-reported outcomes data to patients and clinicians to improve interpretability. Cancer. 2017;123:1848-1859.
148. Squitieri L, Bozic KJ, Pusic AL. The role of patient-reported outcome measures in value-based payment reform. Value Health. 2017;20:834-836.
149. Boyce MB, Browne JP. The effectiveness of providing peer benchmarked feedback to hip replacement surgeons based on patient-reported outcome measures—results from the PROFILE trial: a cluster randomised controlled study. BMJ Open. 2015;5:e008325.
1. Goldenberg MM. Multiple sclerosis review. PT. 2012;37:175-184.
2. Doshi A, Chataway J. Multiple sclerosis, a treatable disease. Clin Med. 2016;16(Suppl 6):s53-s9.
3. Perrin Ross A. Management of multiple sclerosis. Am J Managed Care. 2013;19(16 Suppl):s301-s306.
4. Batalden M, Batalden P, Margolis P, et al. Coproduction of healthcare service. BMJ Qual Saf. 2016;25:509-517.
5. Chen J, Ou L, Hollis SJ. A systematic review of the impact of routine collection of patient reported outcome measures on patients, providers and health organisations in an oncologic setting. BMC Health Serv Res. 2013;13:211.
6. Knaup C, Koesters M, Schoefer D, et al. Effect of feedback of treatment outcome in specialist mental healthcare: meta-analysis. Br J Psychiatr. 2009;195:15-22.
7. Dudgeon D. The impact of measuring patient-reported outcome measures on quality of and access to palliative care. J Palliat Med. 2018;21(S1):S76-s80.
8. Snyder CF, Herman JM, White SM, et al. When using patient-reported outcomes in clinical practice, the measure matters: a randomized controlled trial. J Oncol Pract. 2014;10:e299-e306.
9. Guidance for industry: patient-reported outcome measures: use in medical product development to support labeling claims: draft guidance. Health Qual Life Outcomes. 2006;4:79.
10. de Jong MJ, Huibregtse R, Masclee AAM, et al. Patient-reported outcome measures for use in clinical trials and clinical practice in inflammatory bowel diseases: a systematic review. Clin Gastroenterol Hepatol. 2018;16:648-663.
11. Roman D, Osborne-Stafsnes J, Amy CH, et al. Early lessons from four ‘aligning forces for quality’ communities bolster the case for patient-centered care. Health Affairs. 2013;32:232-241.
12. Ehde DM, Alschuler KN, Sullivan MD, et al. Improving the quality of depression and pain care in multiple sclerosis using collaborative care: The MS-care trial protocol. Contemporary Clinical Trials. 2018;64:219-229.
13. Lavallee DC, Chenok KE, Love RM, et al. Incorporating patient-reported outcomes into health care to engage patients and enhance care. Health Affairs. 2016;35:575-582.
14. Schick-Makaroff K, Thummapol O, Thompson S, et al. Strategies for incorporating patient-reported outcomes in the care of people with chronic kidney disease (PRO kidney): a protocol for a realist synthesis. Clin Gastroenterol Hepatol. 2018;16:648-663.
15. Nelson EC, Dixon-Woods M, Batalden PB, et al. Patient focused registries can improve health, care, and science. BMJ. 2016;354:i3319.
16. Oliver BJ, Nelson EC, Kerrigan CL. Turning feed-forward and feedback processes on patient-reported data into intelligent action and informed decision-making: case studies and principles. Med Care. 2019;57 Suppl 1:S31-s37.
17. Deshpande PR, Rajan S, Sudeepthi BL, Abdul Nazir CP. Patient-reported outcomes: A new era in clinical research. Perspect Clin Res. 2011;2:137-144.
18. Epstein RM, Street RL, Jr. The values and value of patient-centered care. Ann Fam Med. 2011;9:100-103.
19. Payne SA. A study of quality of life in cancer patients receiving palliative chemotherapy. Soc Sci Med. 1992;35:1505-1509.
20. Borgaonkar MR, Irvine EJ. Quality of life measurement in gastrointestinal and liver disorders. Gut. 2000;47:444-454.
21. Macdonell R, Nagels G, Laplaud DA, et al. Improved patient-reported health impact of multiple sclerosis: The ENABLE study of PR-fampridine. Multiple Sclerosis. 2016;22:944-954.
22. Hobart J, Freeman J, Lamping D, et al. The SF-36 in multiple sclerosis: why basic assumptions must be tested. J Neurol Neurosurg Psychiatry. 2001;71:363-370.
23. Lins L, Carvalho FM. SF-36 total score as a single measure of health-related quality of life: Scoping review. SAGE Open Med. 2016;4:2050312116671725.
24. Parkinson’s Disease Society of the United Kingdom. The Parkinson’s Disease Questionnaire (PDQ-39). https://www.parkinsons.org.uk/professionals/resources/parkinsons-disease-questionnaire-pdq-39. Accessed October 20, 2019.
25. Basch E. The missing voice of patients in drug-safety reporting. N Engl J Med. 2010;362:865-869.
26. Calvert M, Brundage M, Jacobsen PB, et al. The CONSORT Patient-Reported Outcome (PRO) extension: implications for clinical trials and practice. Health Qual Life Outcomes. 2013;11:184.
27. Black N. Patient reported outcome measures could help transform healthcare. BMJ. 2013;346:f167.
28. Franks P, Fiscella K, Shields CG, et al. Are patients’ ratings of their physicians related to health outcomes? Ann Fam Med. 2005;3:229-234.
29. Hostetter M, Klein S. Using patient-reported outcomes to improve health care quality. The Commonwealth Fund 2019. www.commonwealthfund.org/publications/newsletter-article/using-patient-reported-outcomes-improve-health-care-quality. Accessed October 24, 2019.
30. Charlotte Kingsley SP. Patient-reported outcome measures and patient-reported experience measures. BJA Education. 2017;17:137-144.
31. Weldring T, Smith SM. Patient-Reported outcomes (PROs) and patient-reported outcome measures (PROMs). Health Services Insights. 2013;6:61-68.
32. Elwyn G, Barr PJ, Grande SW, et al. Developing CollaboRATE: A fast and frugal patient-reported measure of shared decision making in clinical encounters. Patient Educ Couns. 2013;93:102-107.
33. Kurtzke JF. Rating neurologic impairment in multiple sclerosis: an expanded disability status scale (EDSS). Neurology. 1983;33:1444-1452.
34. Rudick RA, Miller D, Clough JD, et al. Quality of life in multiple sclerosis. Comparison with inflammatory bowel disease and rheumatoid arthritis. Arch Neurol. 1992;49:1237-1242.
35. Benito-Leon J, Morales JM, Rivera-Navarro J, Mitchell A. A review about the impact of multiple sclerosis on health-related quality of life. Disabil Rehabil. 2003;25:1291-1303.
36. RAND Corporation. 36-Item Short Form Survey (SF-36). [https://www.rand.org/health-care/surveys_tools/mos/36-item-short-form.html. Accessed October 20, 2019.
37. Bergner M, Bobbitt RA, Carter WB, Gilson BS. The sickness impact profile: development and final revision of a health status measure. Medical Care. 1981;19:787-805.
38. Horsman J, Furlong W, Feeny D, Torrance G. The Health Utilities Index (HUI): concepts, measurement properties and applications. Health Qual Life Outcomes. 2003;1:54.
39. Kinkel RP, Laforet G, You X. Disease-related determinants of quality of life 10 years after clinically isolated syndrome. Int J MS Care. 2015;17:26-34.
40. Baumstarck-Barrau K, Simeoni MC, Reuter F, et al. Cognitive function and quality of life in multiple sclerosis patients: a cross-sectional study. BMC Neurology. 2011;11:17.
41. Samartzis L, Gavala E, Zoukos Y, et al. Perceived cognitive decline in multiple sclerosis impacts quality of life independently of depression. Rehabil Res Pract. 2014;2014:128751.
42. Vitkova M, Rosenberger J, Krokavcova M, et al. Health-related quality of life in multiple sclerosis patients with bladder, bowel and sexual dysfunction. Disabil Rehabil. 2014;36:987-992.
43. Khurana V, Sharma H, Afroz N, et al. Patient-reported outcomes in multiple sclerosis: a systematic comparison of available measures. Eur J Neurol. 2017;24:1099-1107.
44. Hobart J, Lamping D, Fitzpatrick R, et al. The Multiple Sclerosis Impact Scale (MSIS-29): a new patient-based outcome measure. Brain. 2001;124(Pt 5):962-973.
45. Lily O, McFadden E, Hensor E, et al. Disease-specific quality of life in multiple sclerosis: the effect of disease modifying treatment. Multiple Sclerosis. 2006;12:808-813.
46. Ford HL, Gerry E, Tennant A, et al. Developing a disease-specific quality of life measure for people with multiple sclerosis. Clin Rehabil. 2001;15:247-258.
47. Cella DF, Dineen K, Arnason B, et al. Validation of the functional assessment of multiple sclerosis quality of life instrument. Neurology. 1996;47:129-139.
48. Gold SM, Heesen C, Schulz H, et al. Disease specific quality of life instruments in multiple sclerosis: validation of the Hamburg Quality of Life Questionnaire in Multiple Sclerosis (HAQUAMS). Multiple Sclerosis. 2001;7:119-130.
49. Vickrey BG, Hays RD, Harooni R, et al. A health-related quality of life measure for multiple sclerosis. Qual Life Res. 1995;4:187-206.
50. Simeoni M, Auquier P, Fernandez O, et al. Validation of the Multiple Sclerosis International Quality of Life questionnaire. Multiple Sclerosis. 2008;14:219-230.
51. Doward LC, McKenna SP, Meads DM, et al. The development of patient-reported outcome indices for multiple sclerosis (PRIMUS). Multiple Sclerosis. 2009;15:1092-1102.
52. Ozakbas S, Akdede BB, Kosehasanogullari G, et al. Difference between generic and multiple sclerosis-specific quality of life instruments regarding the assessment of treatment efficacy. J Neurol Sci. 2007;256:30-34.
53. Cella DF, Wiklund I, Shumaker SA, Aaronson NK. Integrating health-related quality of life into cross-national clinical trials. Qual Life Res. 1993;2:433-440.
54. Nowinski CJ, Miller DM, Cella D. Evolution of patient-reported outcomes and their role in multiple sclerosis clinical trials. Neurotherapeutics. 2017;14:934-944.
55. Cella D, Nowinski C, Peterman A, et al. The neurology quality-of-life measurement initiative. Arch Phys Med Rehabil. 2011;92(10 Suppl):S28-S36.
56. HealthMeasures: PROMIS. http://www.healthmeasures.net/explore-measurement-systems/promis. Accessed October 20, 2019.
57. Valderas JM, Ferrer M, Mendivil J, et al. Development of EMPRO: a tool for the standardized assessment of patient-reported outcome measures. Value Health. 2008;11:700-708.
58. Bach K, Martling C, Mork PJ, et al. Design of a clinician dashboard to facilitate co-decision making in the management of non-specific low back pain. J Intelligent Helath Syst. 2019;52:269-284.
59. Karami M, Safdari R, Rahimi A. Effective radiology dashboards: key research findings. Radiology Management. 2013;35:42-45.
60. Dowding D, Randell R, Gardner P, et al. Dashboards for improving patient care: review of the literature. Int J Med Informatic. 2015;84:87-100.
61. Mlaver E, Schnipper JL, Boxer RB, et al. User-centered collaborative design and development of an inpatient safety dashboard. Jt Comm J Qual Patient Saf. 2017;43:676-685.
62. Dowding D, Merrill J, Russell D. Using feedback intervention theory to guide clinical dashboard design. AMIA Annu Symp Proc. 2018;2018:395-403.
63. Hartzler AL, Izard JP, Dalkin BL, et al. Design and feasibility of integrating personalized PRO dashboards into prostate cancer care. J Am Med Inform Assoc. 2016;23:38-47.
64. Lavallee DC, Chenok KE, Love RM, et al. Incorporating patient-reported outcomes into health care to engage patients and enhance care. Health Affairs. 2016;35:575-582.
65. U.S. Centers for Medicare & Medicaid Services. Comprehensive Care for Joint Replacement Model. https://innovation.cms.gov/initiatives/CJR. Accessed August 3, 2019.
66. National Quality Forum. Patient-reported outcomes: Accessed August 3, 2019. http://www.qualityforum.org/Patient-Reported_Outcomes.aspx. Accessed October 20, 2019.
67. Institute of Medicine Roundtable on Evidence-Based Medicine. The National Academies Collection: Reports funded by National Institutes of Health. In: Olsen LA, Aisner D, McGinnis JM, eds. The Learning Healthcare System: Workshop Summary. Washington (DC): National Academies Press (US).National Academy of Sciences; 2007.
68. About Learning Health Systems: Agency for Healthcare Research and Quality. https://www.ahrq.gov/learning-health-systems/about.html. Accessed August 3, 2019.
69. Best Care at Lower Cost: The Path to Continuously Learning Health Care in America. Washington (DC): National Academies Press; 2013.
70. Committee on Quality of Health Care in America. Crossing the Quality Chasm: A New Health System for the 21st Century. Washington, DC: National Academies Press; 2001.
71. Oliver BJ, for the MS-CQI Investigators. The Multiple Sclerosis Continuous improvement collaborative: the first coproduction learning health system improvement science research collaborative for multiple sclerosis. The International Society for Quality in Health Care (ISQua) Annual International Conference; October 2019; Cape Town, South Africa.
72. Britto MT, Fuller SC, Kaplan HC, et al. Using a network organisational architecture to support the development of Learning Healthcare Systems. BMJ Qual Saf. 2018;27:937-946.
73. Care Centers: Cystic Fibrosis Foundation. https://www.cff.org/Care/Care-Centers/. Accessed August 3, 2019.
74. Schechter MS, Fink AK, Homa K, Goss CH. The Cystic Fibrosis Foundation patient registry as a tool for use in quality improvement. BMJ Qual Saf. 2014;23(Suppl 1):i9-i14.
75. Mogayzel PJ, Dunitz J, Marrow LC, Hazle LA. Improving chronic care delivery and outcomes: the impact of the cystic fibrosis Care Center Network. BMJ Qual Saf. 2014;23(Suppl 1):i3-i8.
76. Godfrey MM, Oliver BJ. Accelerating the rate of improvement in cystic fibrosis care: contributions and insights of the learning and leadership collaborative. BMJ Qual Saf. 2014;23(Suppl 1):i23-i32.
77. Nelson EC, Godfrey MM. Quality by Design: A Clinical Microsystems Approach. San Francisco, California: Jossey-Bass; 2007.
78. Crohn’s & Colitis Foundation.Quality of Care: IBD Qorus: https://www.crohnscolitisfoundation.org/research/ibd-qorus. Accessed October 23, 2019.
79. Johnson LC, Melmed GY, Nelson EC, et al. Fostering collaboration through creation of an ibd learning health system. Am J Gastroenterol. 2017;112:406-468.
80. Fernandez-Munoz JJ, Moron-Verdasco A, Cigaran-Mendez M, et al. Disability, quality of life, personality, cognitive and psychological variables associated with fatigue in patients with multiple sclerosis. Acta Neurologica Scandinavica. 2015;132:118-124.
81. Flachenecker P, Kumpfel T, Kallmann B, et al. Fatigue in multiple sclerosis: a comparison of different rating scales and correlation to clinical parameters. Multiple Sclerosis. 2002;8:523-526.
82. Tellez N, Rio J, Tintore M, et al. Does the Modified Fatigue Impact Scale offer a more comprehensive assessment of fatigue in MS? Multiple Sclerosis. 2005;11:198-202.
83. Salter A, Fox RJ, Tyry T, et al. The association of fatigue and social participation in multiple sclerosis as assessed using two different instruments. Mult Scler Relat Disord. 2019;31:165-72.
84. Solaro C, Trabucco E, Messmer Uccelli M. Pain and multiple sclerosis: pathophysiology and treatment. Curr Neurol Neurosci Rep. 2013;13:320.
85. Jawahar R, Oh U, Yang S, Lapane KL. A systematic review of pharmacological pain management in multiple sclerosis. Drugs. 2013;73:1711-1722.
86. Rintala A, Hakkinen A, Paltamaa J. Ten-year follow-up of health-related quality of life among ambulatory persons with multiple sclerosis at baseline. Qual Life Res. 2016;25:3119-3127.
87. Tepavcevic DK, Pekmezovic T, Stojsavljevic N, et al. Change in quality of life and predictors of change among patients with multiple sclerosis: a prospective cohort study. Qual Life Res. 2014;23:1027-1037.
88. DasGupta R, Fowler CJ. Bladder, bowel and sexual dysfunction in multiple sclerosis: management strategies. Drugs. 2003;63:153-166.
89. Marck CH, Jelinek PL, Weiland TJ, et al. Sexual function in multiple sclerosis and associations with demographic, disease and lifestyle characteristics: an international cross-sectional study. BMC Neurology. 2016;16:210.
90. Gajofatto A, Benedetti MD. Treatment strategies for multiple sclerosis: When to start, when to change, when to stop? World J Clinical Cases. 2015;3:545-555.
91. National Multiple Sclerosis Society. Disease-modifying therapies for MS. 2018.
92. Rudick RA, Panzara MA. Natalizumab for the treatment of relapsing multiple sclerosis. Biologics. 2008;2:189-199.
93. Cohen JA, Cutter GR, Fischer JS, et al. Benefit of interferon beta-1a on MSFC progression in secondary progressive MS. Neurology. 2002;59:679-687.
94. Patti F, Pappalardo A, Montanari E, et al. Interferon-beta-1a treatment has a positive effect on quality of life of relapsing-remitting multiple sclerosis: results from a longitudinal study. J Neurol Sci. 2014;337:180-185.
95. Novartis. Exploring the efficacy and safety of Siponimod in patients with secondary progressive multiple sclerosis (EXPAND). Available from clinicaltrials.gov/ct2/show/NCT01665144. NLM identifier: NCT01665144.
96. Singal AG, Higgins PD, Waljee AK. A primer on effectiveness and efficacy trials. Clin Transl Gastroenterol. 2014;5:e45.
97. Jongen PJ. Health-related quality of life in patients with multiple sclerosis: impact of disease-modifying drugs. CNS Drugs. 2017;31:585-602.
98. Coretti S, Ruggeri M, McNamee P. The minimum clinically important difference for EQ-5D index: a critical review. Expert Rev Pharmacoecon Outcomes Res. 2014;14:221-233.
99. Clinical Review Report: Ocrelizumab (Ocrevus). CADTH Common Drug Review. 2018:Appendix 4, Validity of Outcome Measures.
100. iCounquer MS; Empowering patients: how individuals with ms are contributing to the fight to find a cure: 2018. https://www.iconquerms.org/empowering-patients-how-individuals-ms-are-contributing-fight-find-cure. Accessed October 23, 2019.
101. K M. Accelerated Cure Project Announces Collaboration with EMD Serono to Advance Patient-Focused Drug Development in Multiple Sclerosis2018. https://www.iconquerms.org/accelerated-cure-project-announces-collaboration-emd-serono-advance-patient-focused-drug-development. Accessed October 24, 2019.
102. Bebo BF Jr, Fox RJ, Lee K, et al. Landscape of MS patient cohorts and registries: Recommendations for maximizing impact. Multiple Sclerosis. 2018;24:579-586.
103. Hedstrom AK, Hillert J, Olsson T, Alfredsson L. Alcohol as a modifiable lifestyle factor affecting multiple sclerosis risk. JAMA Neurology. 2014;71:300-305.
104. Hedstrom AK, Baarnhielm M, Olsson T, Alfredsson L. Tobacco smoking, but not Swedish snuff use, increases the risk of multiple sclerosis. Neurology. 2009;73:696-701.
105. Hedstrom AK, Akerstedt T, Hillert J, et al. Shift work at young age is associated with increased risk for multiple sclerosis. Ann Neurol. 2011;70:733-741.
106. Hadjimichael O, Vollmer T, Oleen-Burkey M. Fatigue characteristics in multiple sclerosis: the North American Research Committee on Multiple Sclerosis (NARCOMS) survey. Health Qual Life Outcomes. 2008;6:100.
107. Fox RJ, Salter AR, Tyry T, et al. Treatment discontinuation and disease progression with injectable disease-modifying therapies: findings from the North American research committee on multiple sclerosis database. Int J MS Care. 2013;15:194-201.
108. Reeve BB, Wyrwich KW, Wu AW, et al. ISOQOL recommends minimum standards for patient-reported outcome measures used in patient-centered outcomes and comparative effectiveness research. Qual Life Res. 2013;22:1889-1905.
109. Jacobs LD, Wende KE, Brownscheidle CM, et al. A profile of multiple sclerosis: the New York State Multiple Sclerosis Consortium. Multiple Sclerosis. 1999;5:369-376.
110. Weinstock-Guttman B, Jacobs LD, Brownscheidle CM, et al. Multiple sclerosis characteristics in African American patients in the New York State Multiple Sclerosis Consortium. Multiple Sclerosis. 2003;9:293-298.
111. Raffel J, Wallace A, Gveric D, et al. Patient-reported outcomes and survival in multiple sclerosis: A 10-year retrospective cohort study using the Multiple Sclerosis Impact Scale-29. PLoS Med. 2017;14:e1002346.
112. Vaughn CB, Kavak KS, Dwyer MG, et al. Fatigue at enrollment predicts EDSS worsening in the New York State Multiple Sclerosis Consortium. Multiple Sclerosis. 2018:1352458518816619.
113. Alroughani RA, Akhtar S, Ahmed SF, Al-Hashel JY. Clinical predictors of disease progression in multiple sclerosis patients with relapsing onset in a nation-wide cohort. Int J Neuroscience. 2015;125:831-837.
114. Dolan JG, Veazie PJ, Russ AJ. Development and initial evaluation of a treatment decision dashboard. BMC Med Inform Decision Mak. 2013;13:51.
115. Stuifbergen AK, Becker H, Blozis S, et al. A randomized clinical trial of a wellness intervention for women with multiple sclerosis. Arch Phys Med Rehabil. 2003;84:467-476.
116. Finkelstein J, Martin C, Bhushan A, et al. Feasibility of computer-assisted education in patients with multiple sclerosis. In: Proceedings of the IEEE Symposium on Computer-Based Medical Systems. Bethesda, MD; 2004.
117. Batalden P. Getting more health from healthcare: quality improvement must acknowledge patient coproduction—an essay by Paul Batalden. BMJ. 2018;362:k3617.
118. Batalden PB, Davidoff F. What is “quality improvement” and how can it transform healthcare? Qual Saf Health Care. 2007;16:2-3.
119. Ovretveit J, Zubkoff L, Nelson EC, et al. Using patient-reported outcome measurement to improve patient care. Int J Qual Health Care. 2017;29:874-879.
120. Prodinger B, Taylor P. Improving quality of care through patient-reported outcome measures (PROMs): expert interviews using the NHS PROMs Programme and the Swedish quality registers for knee and hip arthroplasty as examples. BMC Health Services Res. 2018;18:87.
121. Prinsen CA, Vohra S, Rose MR, et al. How to select outcome measurement instruments for outcomes included in a “Core Outcome Set” – a practical guideline. Trials. 2016;17:449.
122. NINDS. NINDS Common Data Elements: Streamline Your Neuroscience Clinical Research National Institute of Health. https://www.commondataelements.ninds.nih.gov/#page=Default. Accessed October 23, 2019.
123. Francis DO, McPheeters ML, Noud M, et al. Checklist to operationalize measurement characteristics of patient-reported outcome measures. Systematic Rev. 2016;5:129.
124. National Quality Forum. Patient-reported outcomes (PROs) in performance measurement. 2013. https://www.qualityforum.org/Publications/2012/12/Patient-Reported_Outcomes_in_Performance_Measurement.aspx. Accessed October 15, 2019.
125. Dawson J, Doll H, Fitzpatrick R, et al. The routine use of patient reported outcome measures in healthcare settings. BMJ. 2010;340:c186.
126. Aaronson N ET, Greenlhalgh J, Halyard M, et al. User’s guide to implementation of patient reported outcomes assessment in clinical practice. International Society for Quality of Life. 2015.
127. Chan EKH, Edwards TC, Haywood K, et al. Implementing patient-reported outcomes mesaures in clinical practice: a companion guide to the ISOQOL user’s guide. Qual Life Res. 2018;28:621-627.
128. Snyder CF, Wu AW, Miller RS, et al. The role of informatics in promoting patient-centered care. Cancer J. 2011;17:211-218
129. Osborn R, Moulds D, Schneider EC, et al. Primary care physicians in ten countries report challenges caring for patients with complex health needs. Health Affairs. 2015;34:2104-2112.
130. Wu AW, Kharrazi H, Boulware LE, Snyder CF. Measure once, cut 0wice--adding patient-reported outcome measures to the electronic health record for comparative effectiveness research. J Clin Epidemiol. 2013;66(8 Suppl):S12-S20.
131. Trojano M, Bergamaschi R, Amato MP, et al. The Italian multiple sclerosis register. Neurol Sci. 2019;40:155-165.
132. Moorhead SA, Hazlett DE, Harrison L, et al. A new dimension of health care: systematic review of the uses, benefits, and limitations of social media for health communication. J Med Internet Res. 2013;15:e85.
133. Luigi L, Francesco B, Marcello M, et al. e-Health and multiple sclerosis: An update. Multiple Sclerosis J. 2018;24:1657-1664.
134. Marrie RA, Leung S, Tyry T, et al. Use of eHealth and mHealth technology by persons with multiple sclerosis. Mult Scler Relat Dis. 2019;27:13-19.
135. Giunti G, Kool J, Rivera Romero O, Dorronzoro Zubiete E. Exploring the specific needs of persons with multiple sclerosis for mhealth solutions for physical activity: mixed-methods study. JMIR Mhealth Uhealth. 2018;6:e37.
136. Spooner KK, Salemi JL, Salihu HM, Zoorob RJ. eHealth patient-provider communication in the United States: interest, inequalities, and predictors. J Am Med Inform Assoc. 2017;24(e1):e18-e27.
137. Safdar N, Abbo LM, Knobloch MJ, Seo SK. Research methods in healthcare epidemiology: survey and qualitative research. Infect Control Hosp Epidemiol. 2016;37:1272-1277.
138. Rolstad S, Adler J, Rydén A. Response burden and questionnaire length: is shorter better? A review and meta-analysis. Value Health. 2011;14:1101-1108.
139. Chiu C, Bishop M, Pionke JJ, et al. Barriers to the accessibility and continuity of health-care services in people with multiple sclerosis: a literature review. Int J MS Care. 2017;19:313-321.
140. Atreja A, Mehta N, Miller D, et al. One size does not fit all: using qualitative methods to inform the development of an Internet portal for multiple sclerosis patients. AMIA Annu Symp Proc Symp. 2005:16-20.
141. Snyder CF, Blackford AL, Brahmer JR, et al. Needs assessments can identify scores on HRQOL questionnaires that represent problems for patients: an illustration with the Supportive Care Needs Survey and the QLQ-C30. Qual Life Res. 2010;19:837-845.
142. de Vet HC, Terwee CB. The minimal detectable change should not replace the minimal important difference. J Clin Epidemiol. 2010;63:804-805.
143. Snyder CF, Aaronson NK, Choucair AK, et al. Implementing patient-reported outcomes assessment in clinical practice: a review of the options and considerations. Qual Life. 2012;21:1305-1314.
144. Myla DG, Melanie DW, Robert WM, et al. Identification and validation of clinically meaningful benchmarks in the 12-item Multiple Sclerosis Walking Scale. Multiple Sclerosis J. 2017;23:1405-1414.
145. van Munster CEP, Uitdehaag BMJ. Outcome measures in clinical trials for multiple sclerosis. CNS Drugs. 2017;31:217-236.
146. Snyder CF, Jensen R, Courtin SO, Wu AW. PatientViewpoint: a website for patient-reported outcomes assessment. Quality Life Res. 2009;18:793-800.
147. Snyder CF, Smith KC, Bantug ET, et al. What do these scores mean? Presenting patient-reported outcomes data to patients and clinicians to improve interpretability. Cancer. 2017;123:1848-1859.
148. Squitieri L, Bozic KJ, Pusic AL. The role of patient-reported outcome measures in value-based payment reform. Value Health. 2017;20:834-836.
149. Boyce MB, Browne JP. The effectiveness of providing peer benchmarked feedback to hip replacement surgeons based on patient-reported outcome measures—results from the PROFILE trial: a cluster randomised controlled study. BMJ Open. 2015;5:e008325.