User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Endoscopy-related occupational injuries run rampant in gastroenterology
SAN ANTONIO – Swati Pawa, MD, reported at the annual meeting of the American College of Gastroenterology.
Moreover, most respondents said they received zero training in ergonomic strategies for endoscopy-related injury (ERI) prevention during their fellowship training. And there’s been none since. Eighty-one percent of respondents indicated they would welcome such training, added Dr. Pawa, a gastroenterologist at Wake Forest University, Winston-Salem, N.C.
The survey results expose a glaring unmet need in clinical practice, she said: “There have been no published guidelines from any of the major professional GI societies to date addressing how to prevent endoscopy-related injuries.”
The 38-item survey was created by the ACG Women in GI Committee and sponsored by the ACG governing board.
Among the key findings was the identification of sex differences in the types of ERIs reported, which suggests different contributory mechanisms. For example, female gastroenterologists were more likely than were their male colleagues to have experienced ERIs involving the upper back, by a margin of 49% to 36%. Upper extremity pain was more common among the women, too, with 63% reporting hand or finger pain, compared with 53% of men. Twenty-four percent of women reported carpal tunnel syndrome and an equal percentage developed tendonitis, compared with 18% and 17% of men, respectively.
Seventy-one percent of women attributed their ERI to torquing with their right hand, as did 63% of men. Women also more frequently cited having to deal with a nonadjustable bed or monitor as contributing to injury. In contrast, roughly twice as many men as women attributed their ERI to wearing a lead apron or use of the elevator on the duodenoscope.
Equally common causes of ERIs in men and women included standing in awkward positions while supporting an endoscope, standing for a long time, and adjusting tip angulation with the left hand.
Male and female gastroenterologists differed in their practice patterns. The men had been performing endoscopy for a mean of 23 years, compared with 13 years for the women. Fifty-six percent of the men were in private practice, compared with 35% of the women. In contrast, 43% of the women worked in academic settings versus 28% of the men. Thirty percent of the male gastroenterologists characterized themselves as interventional specialists, a rate more than twice that in women, who more commonly specialized in inflammatory bowel disease.
The survey was sent to nearly 16,000 ACG members. It generated a 14% response rate. Roughly two-thirds of responses were provided by male gastroenterologists.
Dr. Pawa and her coinvestigators are now drilling down through the survey data in an effort to identify an appropriate endoscopy workload limit that’s associated with reduced ERI risk.
One audience member commented, “The incidence of ERI in your survey is much higher than most of us would expect.” He speculated that response bias might be at work, with gastroenterologists who have personally experienced an ERI being perhaps more highly motivated to be among the 14% who completed the 38-question survey. Dr. Pawa replied that the survey figures are in line with other, smaller studies.
She reported having no financial conflicts regarding her study.
SAN ANTONIO – Swati Pawa, MD, reported at the annual meeting of the American College of Gastroenterology.
Moreover, most respondents said they received zero training in ergonomic strategies for endoscopy-related injury (ERI) prevention during their fellowship training. And there’s been none since. Eighty-one percent of respondents indicated they would welcome such training, added Dr. Pawa, a gastroenterologist at Wake Forest University, Winston-Salem, N.C.
The survey results expose a glaring unmet need in clinical practice, she said: “There have been no published guidelines from any of the major professional GI societies to date addressing how to prevent endoscopy-related injuries.”
The 38-item survey was created by the ACG Women in GI Committee and sponsored by the ACG governing board.
Among the key findings was the identification of sex differences in the types of ERIs reported, which suggests different contributory mechanisms. For example, female gastroenterologists were more likely than were their male colleagues to have experienced ERIs involving the upper back, by a margin of 49% to 36%. Upper extremity pain was more common among the women, too, with 63% reporting hand or finger pain, compared with 53% of men. Twenty-four percent of women reported carpal tunnel syndrome and an equal percentage developed tendonitis, compared with 18% and 17% of men, respectively.
Seventy-one percent of women attributed their ERI to torquing with their right hand, as did 63% of men. Women also more frequently cited having to deal with a nonadjustable bed or monitor as contributing to injury. In contrast, roughly twice as many men as women attributed their ERI to wearing a lead apron or use of the elevator on the duodenoscope.
Equally common causes of ERIs in men and women included standing in awkward positions while supporting an endoscope, standing for a long time, and adjusting tip angulation with the left hand.
Male and female gastroenterologists differed in their practice patterns. The men had been performing endoscopy for a mean of 23 years, compared with 13 years for the women. Fifty-six percent of the men were in private practice, compared with 35% of the women. In contrast, 43% of the women worked in academic settings versus 28% of the men. Thirty percent of the male gastroenterologists characterized themselves as interventional specialists, a rate more than twice that in women, who more commonly specialized in inflammatory bowel disease.
The survey was sent to nearly 16,000 ACG members. It generated a 14% response rate. Roughly two-thirds of responses were provided by male gastroenterologists.
Dr. Pawa and her coinvestigators are now drilling down through the survey data in an effort to identify an appropriate endoscopy workload limit that’s associated with reduced ERI risk.
One audience member commented, “The incidence of ERI in your survey is much higher than most of us would expect.” He speculated that response bias might be at work, with gastroenterologists who have personally experienced an ERI being perhaps more highly motivated to be among the 14% who completed the 38-question survey. Dr. Pawa replied that the survey figures are in line with other, smaller studies.
She reported having no financial conflicts regarding her study.
SAN ANTONIO – Swati Pawa, MD, reported at the annual meeting of the American College of Gastroenterology.
Moreover, most respondents said they received zero training in ergonomic strategies for endoscopy-related injury (ERI) prevention during their fellowship training. And there’s been none since. Eighty-one percent of respondents indicated they would welcome such training, added Dr. Pawa, a gastroenterologist at Wake Forest University, Winston-Salem, N.C.
The survey results expose a glaring unmet need in clinical practice, she said: “There have been no published guidelines from any of the major professional GI societies to date addressing how to prevent endoscopy-related injuries.”
The 38-item survey was created by the ACG Women in GI Committee and sponsored by the ACG governing board.
Among the key findings was the identification of sex differences in the types of ERIs reported, which suggests different contributory mechanisms. For example, female gastroenterologists were more likely than were their male colleagues to have experienced ERIs involving the upper back, by a margin of 49% to 36%. Upper extremity pain was more common among the women, too, with 63% reporting hand or finger pain, compared with 53% of men. Twenty-four percent of women reported carpal tunnel syndrome and an equal percentage developed tendonitis, compared with 18% and 17% of men, respectively.
Seventy-one percent of women attributed their ERI to torquing with their right hand, as did 63% of men. Women also more frequently cited having to deal with a nonadjustable bed or monitor as contributing to injury. In contrast, roughly twice as many men as women attributed their ERI to wearing a lead apron or use of the elevator on the duodenoscope.
Equally common causes of ERIs in men and women included standing in awkward positions while supporting an endoscope, standing for a long time, and adjusting tip angulation with the left hand.
Male and female gastroenterologists differed in their practice patterns. The men had been performing endoscopy for a mean of 23 years, compared with 13 years for the women. Fifty-six percent of the men were in private practice, compared with 35% of the women. In contrast, 43% of the women worked in academic settings versus 28% of the men. Thirty percent of the male gastroenterologists characterized themselves as interventional specialists, a rate more than twice that in women, who more commonly specialized in inflammatory bowel disease.
The survey was sent to nearly 16,000 ACG members. It generated a 14% response rate. Roughly two-thirds of responses were provided by male gastroenterologists.
Dr. Pawa and her coinvestigators are now drilling down through the survey data in an effort to identify an appropriate endoscopy workload limit that’s associated with reduced ERI risk.
One audience member commented, “The incidence of ERI in your survey is much higher than most of us would expect.” He speculated that response bias might be at work, with gastroenterologists who have personally experienced an ERI being perhaps more highly motivated to be among the 14% who completed the 38-question survey. Dr. Pawa replied that the survey figures are in line with other, smaller studies.
She reported having no financial conflicts regarding her study.
REPORTING FROM ACG 2019
Higher risk of bipolar disorder, depression, anxiety found with autism
Individuals with autism spectrum disorder might be at significantly higher risk of bipolar disorder, anxiety, and depression, a new study suggests.
and associates. The report was published in JAMA Pediatrics.
Dr. Kirsch and associates reported the outcomes of a population-based cohort study involving 1,014 individuals with autism spectrum disorder and 2,028 age-and sex-matched controls without autism spectrum disorder. They found that individuals with autism spectrum disorder were more than nine times more likely to be diagnosed with bipolar disorder, 2.81 times more likely to be diagnosed with depression, and 3.45 times more likely to be diagnosed with anxiety, compared with controls.
“Significant psychosocial sequelae associated with having ASD, including difficulties developing and maintaining relationships, challenges succeeding academically and vocationally, and behaviors that can be problematic to manage, particularly increase risk for mood and anxiety symptoms in individuals with ASD,” wrote Dr. Kirsch of the department of psychiatry and psychology at the Mayo Clinic, Rochester, Minn., and associates. “Individuals with ASD also experience greater rates of other mental health challenges, including attention-deficit/hyperactivity disorder and substance abuse.”
Individuals with autism spectrum disorder who received a diagnosis of depression, anxiety, or bipolar disorder also were more likely to be diagnosed at a younger age than were those without autism. In the case of depression, the median age of diagnosis was 15.7 years, compared with 18.1 years among controls. For anxiety, the median age of diagnosis among individuals with autism spectrum disorder was 15.2 years, compared with 20.3 years for controls. For bipolar disorder, it was 20.3 years, compared with 27 years although the small number of individuals meant this was not statistically significant.
The authors suggested that the earlier age at diagnosis might reflect that individuals with autism spectrum disorder generally are monitored more closely, and are more likely to be connected to screening and diagnostic resources because of their original diagnosis.
The researchers also found that the increased risk of depression and anxiety was even higher among men with autism spectrum disorder, even though the cumulative incidence of these conditions was greater in women both with and without autism. In addition, the researchers noted that individuals with autism spectrum disorder were more likely to be diagnosed with multiple psychiatric conditions than were those without autism.
Dr. Kirsch and associates cited several limitations. One is that the population studied came from Olmsted County, Minn., which is wealthier and less diverse than the general population. Nevertheless, the results could help guide treatments for patients with ASD.
“Given the high rates of comorbidity, researchers and practitioners should develop tools that are specific to the unique needs of this population and effective medications and treatments for mood and anxiety concerns, which remain limited in this population,” they wrote.
The study was funded by grants from the National Institutes of Health and the U.S. Public Health Service. No conflicts of interest were disclosed.
SOURCE: Kirsch A et al. JAMA Pediatr. 2019 Dec 2. doi: 10.1001/jamapediatrics.2019.4368.
Individuals with autism spectrum disorder might be at significantly higher risk of bipolar disorder, anxiety, and depression, a new study suggests.
and associates. The report was published in JAMA Pediatrics.
Dr. Kirsch and associates reported the outcomes of a population-based cohort study involving 1,014 individuals with autism spectrum disorder and 2,028 age-and sex-matched controls without autism spectrum disorder. They found that individuals with autism spectrum disorder were more than nine times more likely to be diagnosed with bipolar disorder, 2.81 times more likely to be diagnosed with depression, and 3.45 times more likely to be diagnosed with anxiety, compared with controls.
“Significant psychosocial sequelae associated with having ASD, including difficulties developing and maintaining relationships, challenges succeeding academically and vocationally, and behaviors that can be problematic to manage, particularly increase risk for mood and anxiety symptoms in individuals with ASD,” wrote Dr. Kirsch of the department of psychiatry and psychology at the Mayo Clinic, Rochester, Minn., and associates. “Individuals with ASD also experience greater rates of other mental health challenges, including attention-deficit/hyperactivity disorder and substance abuse.”
Individuals with autism spectrum disorder who received a diagnosis of depression, anxiety, or bipolar disorder also were more likely to be diagnosed at a younger age than were those without autism. In the case of depression, the median age of diagnosis was 15.7 years, compared with 18.1 years among controls. For anxiety, the median age of diagnosis among individuals with autism spectrum disorder was 15.2 years, compared with 20.3 years for controls. For bipolar disorder, it was 20.3 years, compared with 27 years although the small number of individuals meant this was not statistically significant.
The authors suggested that the earlier age at diagnosis might reflect that individuals with autism spectrum disorder generally are monitored more closely, and are more likely to be connected to screening and diagnostic resources because of their original diagnosis.
The researchers also found that the increased risk of depression and anxiety was even higher among men with autism spectrum disorder, even though the cumulative incidence of these conditions was greater in women both with and without autism. In addition, the researchers noted that individuals with autism spectrum disorder were more likely to be diagnosed with multiple psychiatric conditions than were those without autism.
Dr. Kirsch and associates cited several limitations. One is that the population studied came from Olmsted County, Minn., which is wealthier and less diverse than the general population. Nevertheless, the results could help guide treatments for patients with ASD.
“Given the high rates of comorbidity, researchers and practitioners should develop tools that are specific to the unique needs of this population and effective medications and treatments for mood and anxiety concerns, which remain limited in this population,” they wrote.
The study was funded by grants from the National Institutes of Health and the U.S. Public Health Service. No conflicts of interest were disclosed.
SOURCE: Kirsch A et al. JAMA Pediatr. 2019 Dec 2. doi: 10.1001/jamapediatrics.2019.4368.
Individuals with autism spectrum disorder might be at significantly higher risk of bipolar disorder, anxiety, and depression, a new study suggests.
and associates. The report was published in JAMA Pediatrics.
Dr. Kirsch and associates reported the outcomes of a population-based cohort study involving 1,014 individuals with autism spectrum disorder and 2,028 age-and sex-matched controls without autism spectrum disorder. They found that individuals with autism spectrum disorder were more than nine times more likely to be diagnosed with bipolar disorder, 2.81 times more likely to be diagnosed with depression, and 3.45 times more likely to be diagnosed with anxiety, compared with controls.
“Significant psychosocial sequelae associated with having ASD, including difficulties developing and maintaining relationships, challenges succeeding academically and vocationally, and behaviors that can be problematic to manage, particularly increase risk for mood and anxiety symptoms in individuals with ASD,” wrote Dr. Kirsch of the department of psychiatry and psychology at the Mayo Clinic, Rochester, Minn., and associates. “Individuals with ASD also experience greater rates of other mental health challenges, including attention-deficit/hyperactivity disorder and substance abuse.”
Individuals with autism spectrum disorder who received a diagnosis of depression, anxiety, or bipolar disorder also were more likely to be diagnosed at a younger age than were those without autism. In the case of depression, the median age of diagnosis was 15.7 years, compared with 18.1 years among controls. For anxiety, the median age of diagnosis among individuals with autism spectrum disorder was 15.2 years, compared with 20.3 years for controls. For bipolar disorder, it was 20.3 years, compared with 27 years although the small number of individuals meant this was not statistically significant.
The authors suggested that the earlier age at diagnosis might reflect that individuals with autism spectrum disorder generally are monitored more closely, and are more likely to be connected to screening and diagnostic resources because of their original diagnosis.
The researchers also found that the increased risk of depression and anxiety was even higher among men with autism spectrum disorder, even though the cumulative incidence of these conditions was greater in women both with and without autism. In addition, the researchers noted that individuals with autism spectrum disorder were more likely to be diagnosed with multiple psychiatric conditions than were those without autism.
Dr. Kirsch and associates cited several limitations. One is that the population studied came from Olmsted County, Minn., which is wealthier and less diverse than the general population. Nevertheless, the results could help guide treatments for patients with ASD.
“Given the high rates of comorbidity, researchers and practitioners should develop tools that are specific to the unique needs of this population and effective medications and treatments for mood and anxiety concerns, which remain limited in this population,” they wrote.
The study was funded by grants from the National Institutes of Health and the U.S. Public Health Service. No conflicts of interest were disclosed.
SOURCE: Kirsch A et al. JAMA Pediatr. 2019 Dec 2. doi: 10.1001/jamapediatrics.2019.4368.
FROM JAMA PEDIATRICS
Black-box warnings: How they can improve your clinical practice
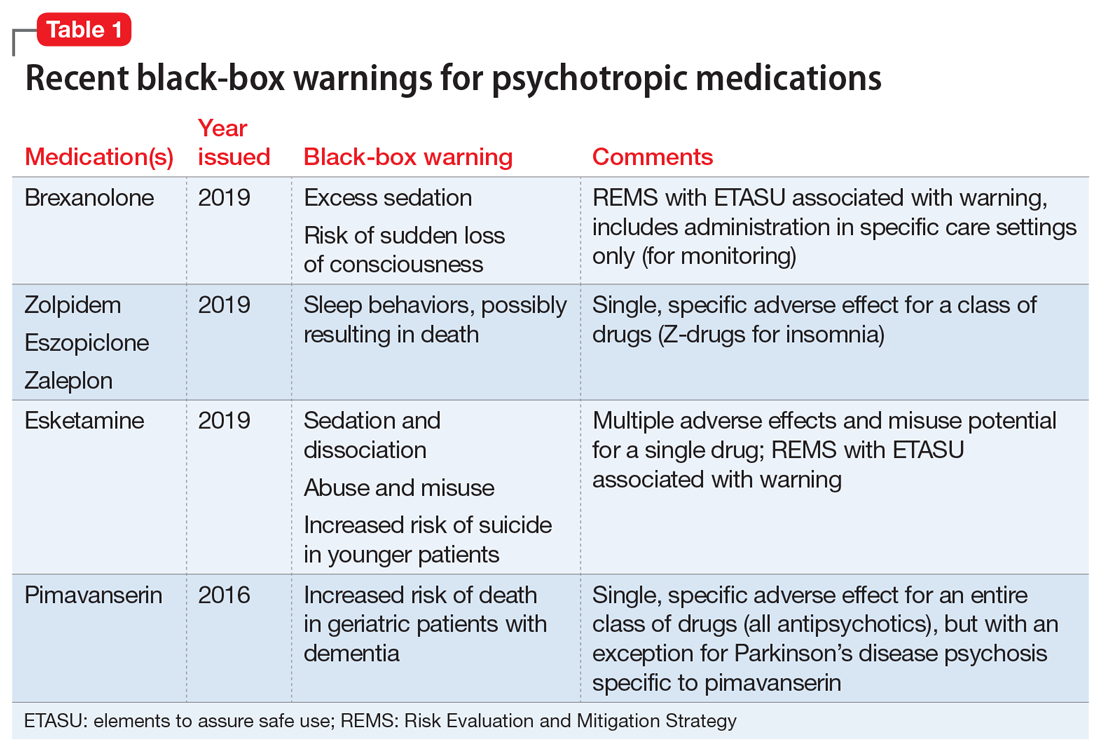
Recently, the FDA issued “black-box” warnings, its most prominent drug safety statements, for esketamine,1 which is indicated for treatment-resistant depression, and the Z-drugs, which are indicated for insomnia2 (Table 1). A black-box warning also comes with brexanolone, which was recently approved for postpartum depression.3 While these newly issued warnings serve as a timely reminder of the importance of black-box warnings, older black-box warnings also cover large areas of psychiatric prescribing, including all medications indicated for treating psychosis or schizophrenia (increased mortality in patients with dementia), and all psychotropic medications with a depression indication (suicidality in younger people).
In this article, we help busy prescribers navigate the landscape of black-box warnings by providing a concise review of how to use them in clinical practice, and where to find information to keep up-to-date.
What are black-box warnings?
A black-box warning is a summary of the potential serious or life-threatening risks of a specific prescription medication. The black-box warning is formatted within a black border found at the top of the manufacturer’s prescribing information document (also known as the package insert or product label). Below the black-box warning, potential risks appear in descending order in sections titled “Contraindications,” “Warnings and Precautions,” and “Adverse Reactions.”4 The FDA issues black-box warnings either during drug development, to take effect upon approval of a new agent, or (more commonly) based on post-marketing safety information,5 which the FDA continuously gathers from reports by patients, clinicians, and industry.6 Federal law mandates the existence of black-box warnings, stating in part that, “special problems, particularly those that may lead to death or serious injury, may be required by the [FDA] to be placed in a prominently displayed box” (21 CFR 201.57(e)).
When is a black-box warning necessary?
The FDA issues a black-box warning based upon its judgment of the seriousness of the adverse effect. However, by definition, these risks do not inherently outweigh the benefits a medication may offer to certain patients. According to the FDA,7 black-box warnings are placed when:
- an adverse reaction so significant exists that this potential negative effect must be considered in risks and benefits when prescribing the medication
- a serious adverse reaction exists that can be prevented, or the risk reduced, by appropriate use of the medication
- the FDA has approved the medication with restrictions to ensure safe use.
Table 2 shows examples of scenarios where black-box warnings have been issued.8 Black-box warnings may be placed on an individual agent or on an entire class of medications. For example, both antipsychotics and antidepressants have class-wide warnings. Finally, black-box warnings are not static, and their content may change; in a study of black-box warnings issued from 2007 to 2015, 29% were entirely new, 32% were considered major updates to existing black-box warnings, and 40% were minor updates.5

Critiques of black-box warnings focus on the absence of published, formal criteria for instituting such warnings, the lack of a consistent approach in their content, and the infrequent inclusion of any information on the relative size of the risk.9 Suggestions for improvement include offering guidance on how to implement the black-box warnings in a patient-centered, shared decision-making model by adding evidence profiles and implementation guides.10 Less frequently considered, black-box warnings may be discontinued if new evidence demonstrates that the risk is lower than previously appreciated; however, similarly to their placement, no explicit criteria for the removal of black-box warnings have been made public.11
When a medication poses an especially high safety risk, the FDA may require the manufacturer to implement a Risk Evaluation and Mitigation Strategy (REMS) program. These programs can describe specific steps to improve medication safety, known as elements to assure safe use (ETASU).4 A familiar example is the clozapine REMS. In order to reduce the risk of severe neutropenia, the clozapine REMS requires prescribers (and pharmacists) to complete specialized training (making up the ETASU). Surprisingly, not every medication with a REMS has a corresponding black-box warning12; more understandably, many medications with black-box warnings do not have an associated REMS, because their risks are evaluated to be manageable by an individual prescriber’s clinical judgment. Most recently, esketamine carries both a black-box warning and a REMS. The black-box warning focuses on adverse effects (Table 1), while the REMS focuses on specific steps used to lessen these risks, including requiring use of a patient enrollment and monitoring form, a fact sheet for patients, and health care setting and pharmacy enrollment forms.13
Continue to: Psychotropic medications and black-box warnings
Psychotropic medications and black-box warnings
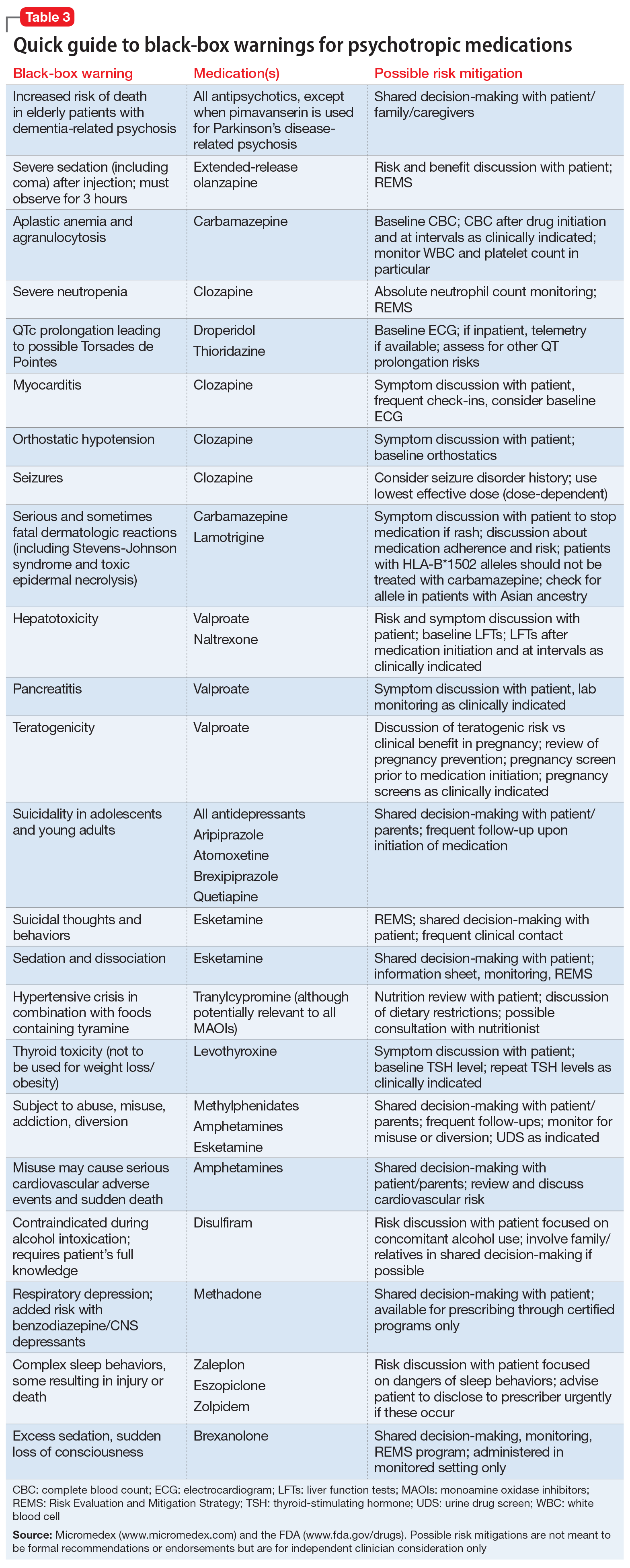
Psychotropic medications have a large number of black-box warnings.14 Because it is difficult to find black-box warnings for multiple medications in one place, we have provided 2 convenient resources to address this gap: a concise summary guide (Table 3) and a more detailed database (Table 4, Table 5, Table 6, Table 7, and Table 8). In these Tables, the possible risk mitigations, off-label uses, and monitoring are not meant to be formal recommendations or endorsements but are for independent clinician consideration only.
The information in these Tables was drawn from publicly available data, primarily the Micromedex and FDA web sites (see Related Resources). Because this information changes over time, at the end of this article we suggest ways for clinicians to stay updated with black-box warnings and build on the information provided in this article. These tools can be useful for day-to-day clinical practice in addition to studying for professional examinations. The following are selected high-profile black-box warnings.
Antidepressants and suicide risk. As a class, antidepressants carry a black-box warning on suicide risk in patients age ≤24. Initially issued in 2005, this warning was extended in 2007 to indicate that depression itself is associated with an increased risk of suicide. This black-box warning is used for an entire class of medications as well as for a specific patient population (age ≤24). Moreover, it indicates that suicide rates in patients age >65 were lower among patients using antidepressants.
Among psychotropic medication black-box warnings, this warning has perhaps been the most controversial. For example, it has been suggested that this black-box warning may have inadvertently increased suicide rates by discouraging clinicians from prescribing antidepressants,15 although this also has been called into question.16 This black-box warning illustrates that the consequences of issuing black-box warnings can be very difficult to assess, which makes their clinical effects highly complex and challenging to evaluate.14
Antipsychotics and dementia-related psychosis. This warning was initially issued in 2005 for second-generation antipsychotics and extended to first-generation antipsychotics in 2008. Antipsychotics as a class carry a black-box warning for increased risk of death in patients with dementia (major neurocognitive disorder). This warning extends to the recently approved antipsychotic pimavanserin, even though this agent’s proposed mechanism of action differs from that of other antipsychotics.17 However, it specifically allows for use in Parkinson’s disease psychosis, which is pimavanserin’s indication.18 In light of recent research suggesting pimavanserin is effective in dementia-related psychosis,19 it bears watching whether this agent becomes the first antipsychotic to have this warning removed.
Continue to: This class warning has...
This class warning has had widespread effects. For example, it has prompted less use of antipsychotics in nursing home facilities, as a result of stricter Centers for Medicare and Medicaid Services regulations20; overall, there is some evidence that there has been reduced prescribing of antipsychotics in general.21 Additionally, this black-box warning is unusual in that it warns about a specific off-label indication, which is itself poorly supported by evidence.21 Concomitantly, few other treatment options are available for this clinical situation. These medications are often seen as the only option for patients with dementia complicated by severe behavioral disturbance, and thus this black-box warning reflects real-world practices.14
Varenicline and neuropsychiatric complications. The withdrawal of the black-box warning on potential neuropsychiatric complications of using varenicline for smoking cessation shows that black-box warnings are not static and can, though infrequently, be removed as more safety data accumulates.11 As additional post-marketing information emerged on this risk, this black-box warning was reconsidered and withdrawn in 2016.22 Its withdrawal could potentially make clinicians more comfortable prescribing varenicline and in turn, help to reduce smoking rates.
How to use black-box warnings
To enhance their clinical practice, prescribers can use black-box warnings to inform safe prescribing practices, to guide shared decision-making, and to improve documentation of their treatment decisions.
Informing safe prescribing practices. A prescriber should be aware of the main safety concerns contained in a medication’s black-box warning; at the same time, these warnings are not meant to unduly limit use when crucial treatment is needed.14 In issuing a black-box warning, the FDA has clearly stated the priority and seriousness of its concern. These safety issues must be balanced against the medication’s utility for a given patient, at the prescriber’s clinical judgment.
Guiding shared decision-making. Clinicians are not required to disclose black-box warnings to patients, and there are no criteria that clearly define the role of these warnings in patient care. As is often noted, the FDA does not regulate the practice of medicine.6 However, given the seriousness of the potential adverse effects delineated by black-box warnings, it is reasonable for clinicians to have a solid grasp of black-box warnings for all medications they prescribe, and to be able to relate these warnings to patients, in appropriate language. This patient-centered discussion should include weighing the risks and benefits with the patient and educating the patient about the risks and strategies to mitigate those risks. This discussion can be augmented by patient handouts, which are often offered by pharmaceutical manufacturers, and by shared decision-making tools. A proactive discussion with patients and families about black-box warnings and other risks discussed in product labels can help reduce fears associated with taking medications and may improve adherence.
Continue to: Improving documentation of treatment decisions
Improving documentation of treatment decisions. Fluent knowledge of black-box warnings may help clinicians improve documentation of their treatment decisions, particularly the risks and benefits of their medication choices. Fluency with black-box warnings will help clinicians accurately document both their awareness of these risks, and how these risks informed their risk-benefit analysis in specific clinical situations.
Despite the clear importance the FDA places on black-box warnings, they are not often a topic of study in training or in postgraduate continuing education, and as a result, not all clinicians may be equally conversant with black-box warnings. While black-box warnings do change over time, many psychotropic medication black-box warnings are long-standing and well-established, and they evolve slowly enough to make mastering these warnings worthwhile in order to make the most informed clinical decisions for patient care.
Keeping up-to-date
There are practical and useful ways for busy clinicians to stay up-to-date with black-box warnings. Although these resources exist in multiple locations, together they provide convenient ways to keep current.
The FDA provides access to black-box warnings via its comprehensive database, DRUGS@FDA (https://www.accessdata.fda.gov/scripts/cder/daf/). Detailed information about REMS (and corresponding ETASU and other information related to REMS programs) is available at REMS@FDA (https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm). Clinicians can make safety reports that may contribute to FDA decision-making on black-box warnings by contacting MedWatch (https://www.fda.gov/safety/medwatch-fda-safety-information-and-adverse-event-reporting-program), the FDA’s adverse events reporting system. MedWatch releases safety information reports, which can be followed on Twitter @FDAMedWatch. Note that FDA information generally is organized by specific drug, and not into categories, such as psychotropic medications.
BlackBoxRx (www.blackboxrx.com) is a subscription-based web service that some clinicians may have access to via facility or academic resources as part of a larger FormWeb software package. Individuals also can subscribe (currently, $89/year).
Continue to: Micromedex
Micromedex (www.micromedex.com), which is widely available through medical libraries, is a subscription-based web service that provides black-box warning information from a separate tab that is easily accessed in each drug’s information front page. There is also an alphabetical list of black-box warnings under a separate tab on the Micromedex landing page.
ePocrates (www.epocrates.com) is a subscription-based service that provides extensive drug information, including black-box warnings, in a convenient mobile app.
Bottom Line
Black-box warnings are the most prominent drug safety warnings issued by the FDA. Many psychotropic medications carry black-box warnings that are crucial to everyday psychiatric prescribing. A better understanding of blackbox warnings can enhance your clinical practice by informing safe prescribing practices, guiding shared decision-making, and improving documentation of your treatment decisions.
Related Resources
- US Food and Drug Administration. DRUGS@FDA: FDAapproved drug products. www.accessdata.fda.gov/scripts/cder/daf/.
- US Food and Drug Administration. Drug safety and availability. www.fda.gov/drugs/drug-safety-and-availability. Updated October 10, 2019.
- BlackBoxRx. www.blackboxrx.com. (Subscription required.)
- Mircromedex. www.micromedex.com. (Subscription required.)
- ePocrates. www.epocrates.com. (Subscription required.)
Drug Brand Names
Amitriptyline • Elavil, Vanatrip
Amoxatine • Strattera
Amoxapine • Asendin
Aripiprazole • Abilify
Asenapine • Saphris
Brexanolone • Zulresso
Brexpiprazole • Rexulti
Bupropion • Wellbutrin
Carbamazepine • Tegretol
Cariprazine • Vraylar
Chlorpromazine • Thorazine
Citalopram • Celexa
Clomipramine • Anafranil
Clozapine • Clozaril
Desipramine • Norpramin
Desvenlafaxine • Pristiq
Dexmethylphenidate • Focalin
Dextroamphetamine/amphetamine • Adderall
Disulfiram • Antabuse
Doxepin • Prudoxin, Silenor
Droperidol • Inapsine
Duloxetine • Cymbalta
Escitalopram • Lexapro
Esketamine • Spravato
Eszopiclone • Lunesta
Fluoxetine • Prozac
Fluphenazine • Prolixin
Fluvoxamine • Luvox
Haloperidol • Haldol
Iloperidone • Fanapt
Imipramine • Tofranil
Isocarboxazid • Marplan
Lamotrigine • Lamictal
Levomilnacipran • Fetzima
Levothyroxine • Synthroid
Linezolid • Zyvox
Lisdexamfetamine • Vyvanse
Lithium • Eskalith, Lithobid
Loxapine • Loxitane
Lurasidone • Latuda
Maprotiline • Ludiomil
Methadone • Dolophine, Methadose
Methylphenidate • Ritalin, Concerta
Midazolam • Versed
Milnacipran • Savella
Mirtazapine • Remeron
Naltrexone • Revia, Vivitrol
Nefazodone • Serzone
Nortriptyline • Aventyl, Pamelor
Olanzapine • Zyprexa
Paliperidone • Invega
Paroxetine • Paxil
Perphenazine • Trilafon
Phenelzine • Nardil
Pimavanserin • Nuplazid
Prochlorperazine • Compro
Protriptyline • Vivactil
Quetiapine • Seroquel
Risperidone • Risperdal
Selegiline • Emsam
Sertraline • Zoloft
Thioridazine • Mellaril
Thiothixene • Navane
Tranylcypromine • Parnate
Trazodone • Desyrel, Oleptro
Trifluoperazine • Stelazine
Trimipramine • Surmontil
Valproate • Depakote
Varenicline • Chantix, Wellbutrin
Vilazodone • Viibryd
Venlafaxine • Effexor
Vortioxetine • Trintellix
Zaleplon • Sonata
Ziprasidone • Geodon
Zolpidem • Ambien
1. Spravato [package insert]. Titusville, NJ: Janssen Pharmaceutical Companies; 2019.
2. U.S. Food and Drug Administration. FDA drug safety announcement: FDA adds boxed warning for risk of serious injuries caused by sleepwalking with certain prescription insomnia medicines. https://www.fda.gov/drugs/drug-safety-and-availability/fda-adds-boxed-warning-risk-serious-injuries-caused-sleepwalking-certain-prescription-insomnia. Published April 30, 2019. Accessed October 28, 2019.
3. Zulresso [package insert]. Cambridge, Mass.: Sage Therapeutics Inc.; 2019.
4. Gassman AL, Nguyen CP, Joffe HV. FDA regulation of prescription drugs. N Engl J Med. 2017;376(7):674-682.
5. Solotke MT, Dhruva SS, Downing NS, et al. New and incremental FDA black box warnings from 2008 to 2015. Expert Opin Drug Saf. 2018;17(2):117-123.
6. Murphy S, Roberts R. “Black box” 101: how the Food and Drug Administration evaluates, communicates, and manages drug benefit/risk. J Allergy Clin Immunol. 2006;117(1):34-39.
7. U.S. Food and Drug Administration. Guidance document: Warnings and precautions, contraindications, and boxed warning sections of labeling for human prescription drug and biological products – content and format. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/warnings-and-precautions-contraindications-and-boxed-warning-sections-labeling-human-prescription. Published October 2011. Accessed October 28, 2019.
8. Beach JE, Faich GA, Bormel FG, et al. Black box warnings in prescription drug labeling: results of a survey of 206 drugs. Food Drug Law J. 1998;53(3):403-411.
9. Matlock A, Allan N, Wills B, et al. A continuing black hole? The FDA boxed warning: an appeal to improve its clinical utility. Clinical Toxicol (Phila). 2011;49(6):443-447.
10. Elraiyah T, Gionfriddo MR, Montori VM, et al. Content, consistency, and quality of black box warnings: time for a change. Ann Intern Med. 2015;163(11):875-876.
11. Yeh JS, Sarpatwari A, Kesselheim AS. Ethical and practical considerations in removing black box warnings from drug labels. Drug Saf. 2016;39(8):709-714.
12. Boudes PF. Risk Evaluation and Mitigation Strategies (REMSs): are they improving drug safety? A critical review of REMSs requiring Elements to Assure Safe Use (ETASU). Drugs R D. 2017;17(2):245-254.
13. U.S. Food and Drug Administration. Approved risk evaluation mitigation strategies (REMS): Spravato (esketamine) REMS program. https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm?event=IndvRemsDetails.page&REMS=386. Updated June 25, 2019. Accessed October 28, 2018.
14. Stevens JR, Jarrahzadeh T, Brendel RW, et al. Strategies for the prescription of psychotropic drugs with black box warnings. Psychosomatics. 2014;55(2):123-133.
15. Friedman RA. Antidepressants’ black-box warning--10 years later. N Engl J Med. 2014;371(18):1666-1668.
16. Stone MB. The FDA warning on antidepressants and suicidality--why the controversy? N Engl J Med. 2014;371(18):1668-1671.
17. Mathis MV, Muoio BM, Andreason P, et al. The US Food and Drug Administration’s perspective on the new antipsychotic pimavanserin. J Clin Psychiatry. 2017;78(6):e668-e673. doi: 10.4088/JCP.16r11119.
18. Nuplazid [package insert]. San Diego, CA: Acadia Pharmaceuticals Inc.; May 2019.
19. Ballard C, Banister C, Khan Z, et al. Evaluation of the safety, tolerability, and efficacy of pimavanserin versus placebo in patients with Alzheimer’s disease psychosis: a phase 2, randomised, placebo-controlled, double-blind study. Lancet Neurol. 2018;17(3):213-222.
20. Maust DT, Kim HM, Chiang C, et al. Association of the Centers for Medicare & Medicaid Services’ National Partnership to Improve Dementia Care with the use of antipsychotics and other psychotropics in long-term care in the United States from 2009 to 2014. JAMA Intern Med. 2018;178(5):640-647.
21. Dorsey ER, Rabbani A, Gallagher SA, et al. Impact of FDA black box advisory on antipsychotic medication use. Arch Intern Med. 2010;170(1):96-103.
22. U.S. Food and Drug Administration. FDA drug safety communication: FDA revises description of mental health side effects of the stop-smoking medicines Chantix (varenicline) and Zyban (bupropion) to reflect clinical trial findings. https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-revises-description-mental-health-side-effects-stop-smoking. Published December 16, 2016. Accessed October 28, 2019.
Recently, the FDA issued “black-box” warnings, its most prominent drug safety statements, for esketamine,1 which is indicated for treatment-resistant depression, and the Z-drugs, which are indicated for insomnia2 (Table 1). A black-box warning also comes with brexanolone, which was recently approved for postpartum depression.3 While these newly issued warnings serve as a timely reminder of the importance of black-box warnings, older black-box warnings also cover large areas of psychiatric prescribing, including all medications indicated for treating psychosis or schizophrenia (increased mortality in patients with dementia), and all psychotropic medications with a depression indication (suicidality in younger people).

In this article, we help busy prescribers navigate the landscape of black-box warnings by providing a concise review of how to use them in clinical practice, and where to find information to keep up-to-date.
What are black-box warnings?
A black-box warning is a summary of the potential serious or life-threatening risks of a specific prescription medication. The black-box warning is formatted within a black border found at the top of the manufacturer’s prescribing information document (also known as the package insert or product label). Below the black-box warning, potential risks appear in descending order in sections titled “Contraindications,” “Warnings and Precautions,” and “Adverse Reactions.”4 The FDA issues black-box warnings either during drug development, to take effect upon approval of a new agent, or (more commonly) based on post-marketing safety information,5 which the FDA continuously gathers from reports by patients, clinicians, and industry.6 Federal law mandates the existence of black-box warnings, stating in part that, “special problems, particularly those that may lead to death or serious injury, may be required by the [FDA] to be placed in a prominently displayed box” (21 CFR 201.57(e)).
When is a black-box warning necessary?
The FDA issues a black-box warning based upon its judgment of the seriousness of the adverse effect. However, by definition, these risks do not inherently outweigh the benefits a medication may offer to certain patients. According to the FDA,7 black-box warnings are placed when:
- an adverse reaction so significant exists that this potential negative effect must be considered in risks and benefits when prescribing the medication
- a serious adverse reaction exists that can be prevented, or the risk reduced, by appropriate use of the medication
- the FDA has approved the medication with restrictions to ensure safe use.
Table 2 shows examples of scenarios where black-box warnings have been issued.8 Black-box warnings may be placed on an individual agent or on an entire class of medications. For example, both antipsychotics and antidepressants have class-wide warnings. Finally, black-box warnings are not static, and their content may change; in a study of black-box warnings issued from 2007 to 2015, 29% were entirely new, 32% were considered major updates to existing black-box warnings, and 40% were minor updates.5

Critiques of black-box warnings focus on the absence of published, formal criteria for instituting such warnings, the lack of a consistent approach in their content, and the infrequent inclusion of any information on the relative size of the risk.9 Suggestions for improvement include offering guidance on how to implement the black-box warnings in a patient-centered, shared decision-making model by adding evidence profiles and implementation guides.10 Less frequently considered, black-box warnings may be discontinued if new evidence demonstrates that the risk is lower than previously appreciated; however, similarly to their placement, no explicit criteria for the removal of black-box warnings have been made public.11
When a medication poses an especially high safety risk, the FDA may require the manufacturer to implement a Risk Evaluation and Mitigation Strategy (REMS) program. These programs can describe specific steps to improve medication safety, known as elements to assure safe use (ETASU).4 A familiar example is the clozapine REMS. In order to reduce the risk of severe neutropenia, the clozapine REMS requires prescribers (and pharmacists) to complete specialized training (making up the ETASU). Surprisingly, not every medication with a REMS has a corresponding black-box warning12; more understandably, many medications with black-box warnings do not have an associated REMS, because their risks are evaluated to be manageable by an individual prescriber’s clinical judgment. Most recently, esketamine carries both a black-box warning and a REMS. The black-box warning focuses on adverse effects (Table 1), while the REMS focuses on specific steps used to lessen these risks, including requiring use of a patient enrollment and monitoring form, a fact sheet for patients, and health care setting and pharmacy enrollment forms.13
Continue to: Psychotropic medications and black-box warnings
Psychotropic medications and black-box warnings
Psychotropic medications have a large number of black-box warnings.14 Because it is difficult to find black-box warnings for multiple medications in one place, we have provided 2 convenient resources to address this gap: a concise summary guide (Table 3) and a more detailed database (Table 4, Table 5, Table 6, Table 7, and Table 8). In these Tables, the possible risk mitigations, off-label uses, and monitoring are not meant to be formal recommendations or endorsements but are for independent clinician consideration only.

The information in these Tables was drawn from publicly available data, primarily the Micromedex and FDA web sites (see Related Resources). Because this information changes over time, at the end of this article we suggest ways for clinicians to stay updated with black-box warnings and build on the information provided in this article. These tools can be useful for day-to-day clinical practice in addition to studying for professional examinations. The following are selected high-profile black-box warnings.
Antidepressants and suicide risk. As a class, antidepressants carry a black-box warning on suicide risk in patients age ≤24. Initially issued in 2005, this warning was extended in 2007 to indicate that depression itself is associated with an increased risk of suicide. This black-box warning is used for an entire class of medications as well as for a specific patient population (age ≤24). Moreover, it indicates that suicide rates in patients age >65 were lower among patients using antidepressants.
Among psychotropic medication black-box warnings, this warning has perhaps been the most controversial. For example, it has been suggested that this black-box warning may have inadvertently increased suicide rates by discouraging clinicians from prescribing antidepressants,15 although this also has been called into question.16 This black-box warning illustrates that the consequences of issuing black-box warnings can be very difficult to assess, which makes their clinical effects highly complex and challenging to evaluate.14
Antipsychotics and dementia-related psychosis. This warning was initially issued in 2005 for second-generation antipsychotics and extended to first-generation antipsychotics in 2008. Antipsychotics as a class carry a black-box warning for increased risk of death in patients with dementia (major neurocognitive disorder). This warning extends to the recently approved antipsychotic pimavanserin, even though this agent’s proposed mechanism of action differs from that of other antipsychotics.17 However, it specifically allows for use in Parkinson’s disease psychosis, which is pimavanserin’s indication.18 In light of recent research suggesting pimavanserin is effective in dementia-related psychosis,19 it bears watching whether this agent becomes the first antipsychotic to have this warning removed.
Continue to: This class warning has...
This class warning has had widespread effects. For example, it has prompted less use of antipsychotics in nursing home facilities, as a result of stricter Centers for Medicare and Medicaid Services regulations20; overall, there is some evidence that there has been reduced prescribing of antipsychotics in general.21 Additionally, this black-box warning is unusual in that it warns about a specific off-label indication, which is itself poorly supported by evidence.21 Concomitantly, few other treatment options are available for this clinical situation. These medications are often seen as the only option for patients with dementia complicated by severe behavioral disturbance, and thus this black-box warning reflects real-world practices.14
Varenicline and neuropsychiatric complications. The withdrawal of the black-box warning on potential neuropsychiatric complications of using varenicline for smoking cessation shows that black-box warnings are not static and can, though infrequently, be removed as more safety data accumulates.11 As additional post-marketing information emerged on this risk, this black-box warning was reconsidered and withdrawn in 2016.22 Its withdrawal could potentially make clinicians more comfortable prescribing varenicline and in turn, help to reduce smoking rates.
How to use black-box warnings
To enhance their clinical practice, prescribers can use black-box warnings to inform safe prescribing practices, to guide shared decision-making, and to improve documentation of their treatment decisions.
Informing safe prescribing practices. A prescriber should be aware of the main safety concerns contained in a medication’s black-box warning; at the same time, these warnings are not meant to unduly limit use when crucial treatment is needed.14 In issuing a black-box warning, the FDA has clearly stated the priority and seriousness of its concern. These safety issues must be balanced against the medication’s utility for a given patient, at the prescriber’s clinical judgment.
Guiding shared decision-making. Clinicians are not required to disclose black-box warnings to patients, and there are no criteria that clearly define the role of these warnings in patient care. As is often noted, the FDA does not regulate the practice of medicine.6 However, given the seriousness of the potential adverse effects delineated by black-box warnings, it is reasonable for clinicians to have a solid grasp of black-box warnings for all medications they prescribe, and to be able to relate these warnings to patients, in appropriate language. This patient-centered discussion should include weighing the risks and benefits with the patient and educating the patient about the risks and strategies to mitigate those risks. This discussion can be augmented by patient handouts, which are often offered by pharmaceutical manufacturers, and by shared decision-making tools. A proactive discussion with patients and families about black-box warnings and other risks discussed in product labels can help reduce fears associated with taking medications and may improve adherence.
Continue to: Improving documentation of treatment decisions
Improving documentation of treatment decisions. Fluent knowledge of black-box warnings may help clinicians improve documentation of their treatment decisions, particularly the risks and benefits of their medication choices. Fluency with black-box warnings will help clinicians accurately document both their awareness of these risks, and how these risks informed their risk-benefit analysis in specific clinical situations.
Despite the clear importance the FDA places on black-box warnings, they are not often a topic of study in training or in postgraduate continuing education, and as a result, not all clinicians may be equally conversant with black-box warnings. While black-box warnings do change over time, many psychotropic medication black-box warnings are long-standing and well-established, and they evolve slowly enough to make mastering these warnings worthwhile in order to make the most informed clinical decisions for patient care.
Keeping up-to-date
There are practical and useful ways for busy clinicians to stay up-to-date with black-box warnings. Although these resources exist in multiple locations, together they provide convenient ways to keep current.
The FDA provides access to black-box warnings via its comprehensive database, DRUGS@FDA (https://www.accessdata.fda.gov/scripts/cder/daf/). Detailed information about REMS (and corresponding ETASU and other information related to REMS programs) is available at REMS@FDA (https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm). Clinicians can make safety reports that may contribute to FDA decision-making on black-box warnings by contacting MedWatch (https://www.fda.gov/safety/medwatch-fda-safety-information-and-adverse-event-reporting-program), the FDA’s adverse events reporting system. MedWatch releases safety information reports, which can be followed on Twitter @FDAMedWatch. Note that FDA information generally is organized by specific drug, and not into categories, such as psychotropic medications.
BlackBoxRx (www.blackboxrx.com) is a subscription-based web service that some clinicians may have access to via facility or academic resources as part of a larger FormWeb software package. Individuals also can subscribe (currently, $89/year).
Continue to: Micromedex
Micromedex (www.micromedex.com), which is widely available through medical libraries, is a subscription-based web service that provides black-box warning information from a separate tab that is easily accessed in each drug’s information front page. There is also an alphabetical list of black-box warnings under a separate tab on the Micromedex landing page.
ePocrates (www.epocrates.com) is a subscription-based service that provides extensive drug information, including black-box warnings, in a convenient mobile app.
Bottom Line
Black-box warnings are the most prominent drug safety warnings issued by the FDA. Many psychotropic medications carry black-box warnings that are crucial to everyday psychiatric prescribing. A better understanding of blackbox warnings can enhance your clinical practice by informing safe prescribing practices, guiding shared decision-making, and improving documentation of your treatment decisions.
Related Resources
- US Food and Drug Administration. DRUGS@FDA: FDAapproved drug products. www.accessdata.fda.gov/scripts/cder/daf/.
- US Food and Drug Administration. Drug safety and availability. www.fda.gov/drugs/drug-safety-and-availability. Updated October 10, 2019.
- BlackBoxRx. www.blackboxrx.com. (Subscription required.)
- Mircromedex. www.micromedex.com. (Subscription required.)
- ePocrates. www.epocrates.com. (Subscription required.)
Drug Brand Names
Amitriptyline • Elavil, Vanatrip
Amoxatine • Strattera
Amoxapine • Asendin
Aripiprazole • Abilify
Asenapine • Saphris
Brexanolone • Zulresso
Brexpiprazole • Rexulti
Bupropion • Wellbutrin
Carbamazepine • Tegretol
Cariprazine • Vraylar
Chlorpromazine • Thorazine
Citalopram • Celexa
Clomipramine • Anafranil
Clozapine • Clozaril
Desipramine • Norpramin
Desvenlafaxine • Pristiq
Dexmethylphenidate • Focalin
Dextroamphetamine/amphetamine • Adderall
Disulfiram • Antabuse
Doxepin • Prudoxin, Silenor
Droperidol • Inapsine
Duloxetine • Cymbalta
Escitalopram • Lexapro
Esketamine • Spravato
Eszopiclone • Lunesta
Fluoxetine • Prozac
Fluphenazine • Prolixin
Fluvoxamine • Luvox
Haloperidol • Haldol
Iloperidone • Fanapt
Imipramine • Tofranil
Isocarboxazid • Marplan
Lamotrigine • Lamictal
Levomilnacipran • Fetzima
Levothyroxine • Synthroid
Linezolid • Zyvox
Lisdexamfetamine • Vyvanse
Lithium • Eskalith, Lithobid
Loxapine • Loxitane
Lurasidone • Latuda
Maprotiline • Ludiomil
Methadone • Dolophine, Methadose
Methylphenidate • Ritalin, Concerta
Midazolam • Versed
Milnacipran • Savella
Mirtazapine • Remeron
Naltrexone • Revia, Vivitrol
Nefazodone • Serzone
Nortriptyline • Aventyl, Pamelor
Olanzapine • Zyprexa
Paliperidone • Invega
Paroxetine • Paxil
Perphenazine • Trilafon
Phenelzine • Nardil
Pimavanserin • Nuplazid
Prochlorperazine • Compro
Protriptyline • Vivactil
Quetiapine • Seroquel
Risperidone • Risperdal
Selegiline • Emsam
Sertraline • Zoloft
Thioridazine • Mellaril
Thiothixene • Navane
Tranylcypromine • Parnate
Trazodone • Desyrel, Oleptro
Trifluoperazine • Stelazine
Trimipramine • Surmontil
Valproate • Depakote
Varenicline • Chantix, Wellbutrin
Vilazodone • Viibryd
Venlafaxine • Effexor
Vortioxetine • Trintellix
Zaleplon • Sonata
Ziprasidone • Geodon
Zolpidem • Ambien
Recently, the FDA issued “black-box” warnings, its most prominent drug safety statements, for esketamine,1 which is indicated for treatment-resistant depression, and the Z-drugs, which are indicated for insomnia2 (Table 1). A black-box warning also comes with brexanolone, which was recently approved for postpartum depression.3 While these newly issued warnings serve as a timely reminder of the importance of black-box warnings, older black-box warnings also cover large areas of psychiatric prescribing, including all medications indicated for treating psychosis or schizophrenia (increased mortality in patients with dementia), and all psychotropic medications with a depression indication (suicidality in younger people).

In this article, we help busy prescribers navigate the landscape of black-box warnings by providing a concise review of how to use them in clinical practice, and where to find information to keep up-to-date.
What are black-box warnings?
A black-box warning is a summary of the potential serious or life-threatening risks of a specific prescription medication. The black-box warning is formatted within a black border found at the top of the manufacturer’s prescribing information document (also known as the package insert or product label). Below the black-box warning, potential risks appear in descending order in sections titled “Contraindications,” “Warnings and Precautions,” and “Adverse Reactions.”4 The FDA issues black-box warnings either during drug development, to take effect upon approval of a new agent, or (more commonly) based on post-marketing safety information,5 which the FDA continuously gathers from reports by patients, clinicians, and industry.6 Federal law mandates the existence of black-box warnings, stating in part that, “special problems, particularly those that may lead to death or serious injury, may be required by the [FDA] to be placed in a prominently displayed box” (21 CFR 201.57(e)).
When is a black-box warning necessary?
The FDA issues a black-box warning based upon its judgment of the seriousness of the adverse effect. However, by definition, these risks do not inherently outweigh the benefits a medication may offer to certain patients. According to the FDA,7 black-box warnings are placed when:
- an adverse reaction so significant exists that this potential negative effect must be considered in risks and benefits when prescribing the medication
- a serious adverse reaction exists that can be prevented, or the risk reduced, by appropriate use of the medication
- the FDA has approved the medication with restrictions to ensure safe use.
Table 2 shows examples of scenarios where black-box warnings have been issued.8 Black-box warnings may be placed on an individual agent or on an entire class of medications. For example, both antipsychotics and antidepressants have class-wide warnings. Finally, black-box warnings are not static, and their content may change; in a study of black-box warnings issued from 2007 to 2015, 29% were entirely new, 32% were considered major updates to existing black-box warnings, and 40% were minor updates.5

Critiques of black-box warnings focus on the absence of published, formal criteria for instituting such warnings, the lack of a consistent approach in their content, and the infrequent inclusion of any information on the relative size of the risk.9 Suggestions for improvement include offering guidance on how to implement the black-box warnings in a patient-centered, shared decision-making model by adding evidence profiles and implementation guides.10 Less frequently considered, black-box warnings may be discontinued if new evidence demonstrates that the risk is lower than previously appreciated; however, similarly to their placement, no explicit criteria for the removal of black-box warnings have been made public.11
When a medication poses an especially high safety risk, the FDA may require the manufacturer to implement a Risk Evaluation and Mitigation Strategy (REMS) program. These programs can describe specific steps to improve medication safety, known as elements to assure safe use (ETASU).4 A familiar example is the clozapine REMS. In order to reduce the risk of severe neutropenia, the clozapine REMS requires prescribers (and pharmacists) to complete specialized training (making up the ETASU). Surprisingly, not every medication with a REMS has a corresponding black-box warning12; more understandably, many medications with black-box warnings do not have an associated REMS, because their risks are evaluated to be manageable by an individual prescriber’s clinical judgment. Most recently, esketamine carries both a black-box warning and a REMS. The black-box warning focuses on adverse effects (Table 1), while the REMS focuses on specific steps used to lessen these risks, including requiring use of a patient enrollment and monitoring form, a fact sheet for patients, and health care setting and pharmacy enrollment forms.13
Continue to: Psychotropic medications and black-box warnings
Psychotropic medications and black-box warnings
Psychotropic medications have a large number of black-box warnings.14 Because it is difficult to find black-box warnings for multiple medications in one place, we have provided 2 convenient resources to address this gap: a concise summary guide (Table 3) and a more detailed database (Table 4, Table 5, Table 6, Table 7, and Table 8). In these Tables, the possible risk mitigations, off-label uses, and monitoring are not meant to be formal recommendations or endorsements but are for independent clinician consideration only.

The information in these Tables was drawn from publicly available data, primarily the Micromedex and FDA web sites (see Related Resources). Because this information changes over time, at the end of this article we suggest ways for clinicians to stay updated with black-box warnings and build on the information provided in this article. These tools can be useful for day-to-day clinical practice in addition to studying for professional examinations. The following are selected high-profile black-box warnings.
Antidepressants and suicide risk. As a class, antidepressants carry a black-box warning on suicide risk in patients age ≤24. Initially issued in 2005, this warning was extended in 2007 to indicate that depression itself is associated with an increased risk of suicide. This black-box warning is used for an entire class of medications as well as for a specific patient population (age ≤24). Moreover, it indicates that suicide rates in patients age >65 were lower among patients using antidepressants.
Among psychotropic medication black-box warnings, this warning has perhaps been the most controversial. For example, it has been suggested that this black-box warning may have inadvertently increased suicide rates by discouraging clinicians from prescribing antidepressants,15 although this also has been called into question.16 This black-box warning illustrates that the consequences of issuing black-box warnings can be very difficult to assess, which makes their clinical effects highly complex and challenging to evaluate.14
Antipsychotics and dementia-related psychosis. This warning was initially issued in 2005 for second-generation antipsychotics and extended to first-generation antipsychotics in 2008. Antipsychotics as a class carry a black-box warning for increased risk of death in patients with dementia (major neurocognitive disorder). This warning extends to the recently approved antipsychotic pimavanserin, even though this agent’s proposed mechanism of action differs from that of other antipsychotics.17 However, it specifically allows for use in Parkinson’s disease psychosis, which is pimavanserin’s indication.18 In light of recent research suggesting pimavanserin is effective in dementia-related psychosis,19 it bears watching whether this agent becomes the first antipsychotic to have this warning removed.
Continue to: This class warning has...
This class warning has had widespread effects. For example, it has prompted less use of antipsychotics in nursing home facilities, as a result of stricter Centers for Medicare and Medicaid Services regulations20; overall, there is some evidence that there has been reduced prescribing of antipsychotics in general.21 Additionally, this black-box warning is unusual in that it warns about a specific off-label indication, which is itself poorly supported by evidence.21 Concomitantly, few other treatment options are available for this clinical situation. These medications are often seen as the only option for patients with dementia complicated by severe behavioral disturbance, and thus this black-box warning reflects real-world practices.14
Varenicline and neuropsychiatric complications. The withdrawal of the black-box warning on potential neuropsychiatric complications of using varenicline for smoking cessation shows that black-box warnings are not static and can, though infrequently, be removed as more safety data accumulates.11 As additional post-marketing information emerged on this risk, this black-box warning was reconsidered and withdrawn in 2016.22 Its withdrawal could potentially make clinicians more comfortable prescribing varenicline and in turn, help to reduce smoking rates.
How to use black-box warnings
To enhance their clinical practice, prescribers can use black-box warnings to inform safe prescribing practices, to guide shared decision-making, and to improve documentation of their treatment decisions.
Informing safe prescribing practices. A prescriber should be aware of the main safety concerns contained in a medication’s black-box warning; at the same time, these warnings are not meant to unduly limit use when crucial treatment is needed.14 In issuing a black-box warning, the FDA has clearly stated the priority and seriousness of its concern. These safety issues must be balanced against the medication’s utility for a given patient, at the prescriber’s clinical judgment.
Guiding shared decision-making. Clinicians are not required to disclose black-box warnings to patients, and there are no criteria that clearly define the role of these warnings in patient care. As is often noted, the FDA does not regulate the practice of medicine.6 However, given the seriousness of the potential adverse effects delineated by black-box warnings, it is reasonable for clinicians to have a solid grasp of black-box warnings for all medications they prescribe, and to be able to relate these warnings to patients, in appropriate language. This patient-centered discussion should include weighing the risks and benefits with the patient and educating the patient about the risks and strategies to mitigate those risks. This discussion can be augmented by patient handouts, which are often offered by pharmaceutical manufacturers, and by shared decision-making tools. A proactive discussion with patients and families about black-box warnings and other risks discussed in product labels can help reduce fears associated with taking medications and may improve adherence.
Continue to: Improving documentation of treatment decisions
Improving documentation of treatment decisions. Fluent knowledge of black-box warnings may help clinicians improve documentation of their treatment decisions, particularly the risks and benefits of their medication choices. Fluency with black-box warnings will help clinicians accurately document both their awareness of these risks, and how these risks informed their risk-benefit analysis in specific clinical situations.
Despite the clear importance the FDA places on black-box warnings, they are not often a topic of study in training or in postgraduate continuing education, and as a result, not all clinicians may be equally conversant with black-box warnings. While black-box warnings do change over time, many psychotropic medication black-box warnings are long-standing and well-established, and they evolve slowly enough to make mastering these warnings worthwhile in order to make the most informed clinical decisions for patient care.
Keeping up-to-date
There are practical and useful ways for busy clinicians to stay up-to-date with black-box warnings. Although these resources exist in multiple locations, together they provide convenient ways to keep current.
The FDA provides access to black-box warnings via its comprehensive database, DRUGS@FDA (https://www.accessdata.fda.gov/scripts/cder/daf/). Detailed information about REMS (and corresponding ETASU and other information related to REMS programs) is available at REMS@FDA (https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm). Clinicians can make safety reports that may contribute to FDA decision-making on black-box warnings by contacting MedWatch (https://www.fda.gov/safety/medwatch-fda-safety-information-and-adverse-event-reporting-program), the FDA’s adverse events reporting system. MedWatch releases safety information reports, which can be followed on Twitter @FDAMedWatch. Note that FDA information generally is organized by specific drug, and not into categories, such as psychotropic medications.
BlackBoxRx (www.blackboxrx.com) is a subscription-based web service that some clinicians may have access to via facility or academic resources as part of a larger FormWeb software package. Individuals also can subscribe (currently, $89/year).
Continue to: Micromedex
Micromedex (www.micromedex.com), which is widely available through medical libraries, is a subscription-based web service that provides black-box warning information from a separate tab that is easily accessed in each drug’s information front page. There is also an alphabetical list of black-box warnings under a separate tab on the Micromedex landing page.
ePocrates (www.epocrates.com) is a subscription-based service that provides extensive drug information, including black-box warnings, in a convenient mobile app.
Bottom Line
Black-box warnings are the most prominent drug safety warnings issued by the FDA. Many psychotropic medications carry black-box warnings that are crucial to everyday psychiatric prescribing. A better understanding of blackbox warnings can enhance your clinical practice by informing safe prescribing practices, guiding shared decision-making, and improving documentation of your treatment decisions.
Related Resources
- US Food and Drug Administration. DRUGS@FDA: FDAapproved drug products. www.accessdata.fda.gov/scripts/cder/daf/.
- US Food and Drug Administration. Drug safety and availability. www.fda.gov/drugs/drug-safety-and-availability. Updated October 10, 2019.
- BlackBoxRx. www.blackboxrx.com. (Subscription required.)
- Mircromedex. www.micromedex.com. (Subscription required.)
- ePocrates. www.epocrates.com. (Subscription required.)
Drug Brand Names
Amitriptyline • Elavil, Vanatrip
Amoxatine • Strattera
Amoxapine • Asendin
Aripiprazole • Abilify
Asenapine • Saphris
Brexanolone • Zulresso
Brexpiprazole • Rexulti
Bupropion • Wellbutrin
Carbamazepine • Tegretol
Cariprazine • Vraylar
Chlorpromazine • Thorazine
Citalopram • Celexa
Clomipramine • Anafranil
Clozapine • Clozaril
Desipramine • Norpramin
Desvenlafaxine • Pristiq
Dexmethylphenidate • Focalin
Dextroamphetamine/amphetamine • Adderall
Disulfiram • Antabuse
Doxepin • Prudoxin, Silenor
Droperidol • Inapsine
Duloxetine • Cymbalta
Escitalopram • Lexapro
Esketamine • Spravato
Eszopiclone • Lunesta
Fluoxetine • Prozac
Fluphenazine • Prolixin
Fluvoxamine • Luvox
Haloperidol • Haldol
Iloperidone • Fanapt
Imipramine • Tofranil
Isocarboxazid • Marplan
Lamotrigine • Lamictal
Levomilnacipran • Fetzima
Levothyroxine • Synthroid
Linezolid • Zyvox
Lisdexamfetamine • Vyvanse
Lithium • Eskalith, Lithobid
Loxapine • Loxitane
Lurasidone • Latuda
Maprotiline • Ludiomil
Methadone • Dolophine, Methadose
Methylphenidate • Ritalin, Concerta
Midazolam • Versed
Milnacipran • Savella
Mirtazapine • Remeron
Naltrexone • Revia, Vivitrol
Nefazodone • Serzone
Nortriptyline • Aventyl, Pamelor
Olanzapine • Zyprexa
Paliperidone • Invega
Paroxetine • Paxil
Perphenazine • Trilafon
Phenelzine • Nardil
Pimavanserin • Nuplazid
Prochlorperazine • Compro
Protriptyline • Vivactil
Quetiapine • Seroquel
Risperidone • Risperdal
Selegiline • Emsam
Sertraline • Zoloft
Thioridazine • Mellaril
Thiothixene • Navane
Tranylcypromine • Parnate
Trazodone • Desyrel, Oleptro
Trifluoperazine • Stelazine
Trimipramine • Surmontil
Valproate • Depakote
Varenicline • Chantix, Wellbutrin
Vilazodone • Viibryd
Venlafaxine • Effexor
Vortioxetine • Trintellix
Zaleplon • Sonata
Ziprasidone • Geodon
Zolpidem • Ambien
1. Spravato [package insert]. Titusville, NJ: Janssen Pharmaceutical Companies; 2019.
2. U.S. Food and Drug Administration. FDA drug safety announcement: FDA adds boxed warning for risk of serious injuries caused by sleepwalking with certain prescription insomnia medicines. https://www.fda.gov/drugs/drug-safety-and-availability/fda-adds-boxed-warning-risk-serious-injuries-caused-sleepwalking-certain-prescription-insomnia. Published April 30, 2019. Accessed October 28, 2019.
3. Zulresso [package insert]. Cambridge, Mass.: Sage Therapeutics Inc.; 2019.
4. Gassman AL, Nguyen CP, Joffe HV. FDA regulation of prescription drugs. N Engl J Med. 2017;376(7):674-682.
5. Solotke MT, Dhruva SS, Downing NS, et al. New and incremental FDA black box warnings from 2008 to 2015. Expert Opin Drug Saf. 2018;17(2):117-123.
6. Murphy S, Roberts R. “Black box” 101: how the Food and Drug Administration evaluates, communicates, and manages drug benefit/risk. J Allergy Clin Immunol. 2006;117(1):34-39.
7. U.S. Food and Drug Administration. Guidance document: Warnings and precautions, contraindications, and boxed warning sections of labeling for human prescription drug and biological products – content and format. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/warnings-and-precautions-contraindications-and-boxed-warning-sections-labeling-human-prescription. Published October 2011. Accessed October 28, 2019.
8. Beach JE, Faich GA, Bormel FG, et al. Black box warnings in prescription drug labeling: results of a survey of 206 drugs. Food Drug Law J. 1998;53(3):403-411.
9. Matlock A, Allan N, Wills B, et al. A continuing black hole? The FDA boxed warning: an appeal to improve its clinical utility. Clinical Toxicol (Phila). 2011;49(6):443-447.
10. Elraiyah T, Gionfriddo MR, Montori VM, et al. Content, consistency, and quality of black box warnings: time for a change. Ann Intern Med. 2015;163(11):875-876.
11. Yeh JS, Sarpatwari A, Kesselheim AS. Ethical and practical considerations in removing black box warnings from drug labels. Drug Saf. 2016;39(8):709-714.
12. Boudes PF. Risk Evaluation and Mitigation Strategies (REMSs): are they improving drug safety? A critical review of REMSs requiring Elements to Assure Safe Use (ETASU). Drugs R D. 2017;17(2):245-254.
13. U.S. Food and Drug Administration. Approved risk evaluation mitigation strategies (REMS): Spravato (esketamine) REMS program. https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm?event=IndvRemsDetails.page&REMS=386. Updated June 25, 2019. Accessed October 28, 2018.
14. Stevens JR, Jarrahzadeh T, Brendel RW, et al. Strategies for the prescription of psychotropic drugs with black box warnings. Psychosomatics. 2014;55(2):123-133.
15. Friedman RA. Antidepressants’ black-box warning--10 years later. N Engl J Med. 2014;371(18):1666-1668.
16. Stone MB. The FDA warning on antidepressants and suicidality--why the controversy? N Engl J Med. 2014;371(18):1668-1671.
17. Mathis MV, Muoio BM, Andreason P, et al. The US Food and Drug Administration’s perspective on the new antipsychotic pimavanserin. J Clin Psychiatry. 2017;78(6):e668-e673. doi: 10.4088/JCP.16r11119.
18. Nuplazid [package insert]. San Diego, CA: Acadia Pharmaceuticals Inc.; May 2019.
19. Ballard C, Banister C, Khan Z, et al. Evaluation of the safety, tolerability, and efficacy of pimavanserin versus placebo in patients with Alzheimer’s disease psychosis: a phase 2, randomised, placebo-controlled, double-blind study. Lancet Neurol. 2018;17(3):213-222.
20. Maust DT, Kim HM, Chiang C, et al. Association of the Centers for Medicare & Medicaid Services’ National Partnership to Improve Dementia Care with the use of antipsychotics and other psychotropics in long-term care in the United States from 2009 to 2014. JAMA Intern Med. 2018;178(5):640-647.
21. Dorsey ER, Rabbani A, Gallagher SA, et al. Impact of FDA black box advisory on antipsychotic medication use. Arch Intern Med. 2010;170(1):96-103.
22. U.S. Food and Drug Administration. FDA drug safety communication: FDA revises description of mental health side effects of the stop-smoking medicines Chantix (varenicline) and Zyban (bupropion) to reflect clinical trial findings. https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-revises-description-mental-health-side-effects-stop-smoking. Published December 16, 2016. Accessed October 28, 2019.
1. Spravato [package insert]. Titusville, NJ: Janssen Pharmaceutical Companies; 2019.
2. U.S. Food and Drug Administration. FDA drug safety announcement: FDA adds boxed warning for risk of serious injuries caused by sleepwalking with certain prescription insomnia medicines. https://www.fda.gov/drugs/drug-safety-and-availability/fda-adds-boxed-warning-risk-serious-injuries-caused-sleepwalking-certain-prescription-insomnia. Published April 30, 2019. Accessed October 28, 2019.
3. Zulresso [package insert]. Cambridge, Mass.: Sage Therapeutics Inc.; 2019.
4. Gassman AL, Nguyen CP, Joffe HV. FDA regulation of prescription drugs. N Engl J Med. 2017;376(7):674-682.
5. Solotke MT, Dhruva SS, Downing NS, et al. New and incremental FDA black box warnings from 2008 to 2015. Expert Opin Drug Saf. 2018;17(2):117-123.
6. Murphy S, Roberts R. “Black box” 101: how the Food and Drug Administration evaluates, communicates, and manages drug benefit/risk. J Allergy Clin Immunol. 2006;117(1):34-39.
7. U.S. Food and Drug Administration. Guidance document: Warnings and precautions, contraindications, and boxed warning sections of labeling for human prescription drug and biological products – content and format. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/warnings-and-precautions-contraindications-and-boxed-warning-sections-labeling-human-prescription. Published October 2011. Accessed October 28, 2019.
8. Beach JE, Faich GA, Bormel FG, et al. Black box warnings in prescription drug labeling: results of a survey of 206 drugs. Food Drug Law J. 1998;53(3):403-411.
9. Matlock A, Allan N, Wills B, et al. A continuing black hole? The FDA boxed warning: an appeal to improve its clinical utility. Clinical Toxicol (Phila). 2011;49(6):443-447.
10. Elraiyah T, Gionfriddo MR, Montori VM, et al. Content, consistency, and quality of black box warnings: time for a change. Ann Intern Med. 2015;163(11):875-876.
11. Yeh JS, Sarpatwari A, Kesselheim AS. Ethical and practical considerations in removing black box warnings from drug labels. Drug Saf. 2016;39(8):709-714.
12. Boudes PF. Risk Evaluation and Mitigation Strategies (REMSs): are they improving drug safety? A critical review of REMSs requiring Elements to Assure Safe Use (ETASU). Drugs R D. 2017;17(2):245-254.
13. U.S. Food and Drug Administration. Approved risk evaluation mitigation strategies (REMS): Spravato (esketamine) REMS program. https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm?event=IndvRemsDetails.page&REMS=386. Updated June 25, 2019. Accessed October 28, 2018.
14. Stevens JR, Jarrahzadeh T, Brendel RW, et al. Strategies for the prescription of psychotropic drugs with black box warnings. Psychosomatics. 2014;55(2):123-133.
15. Friedman RA. Antidepressants’ black-box warning--10 years later. N Engl J Med. 2014;371(18):1666-1668.
16. Stone MB. The FDA warning on antidepressants and suicidality--why the controversy? N Engl J Med. 2014;371(18):1668-1671.
17. Mathis MV, Muoio BM, Andreason P, et al. The US Food and Drug Administration’s perspective on the new antipsychotic pimavanserin. J Clin Psychiatry. 2017;78(6):e668-e673. doi: 10.4088/JCP.16r11119.
18. Nuplazid [package insert]. San Diego, CA: Acadia Pharmaceuticals Inc.; May 2019.
19. Ballard C, Banister C, Khan Z, et al. Evaluation of the safety, tolerability, and efficacy of pimavanserin versus placebo in patients with Alzheimer’s disease psychosis: a phase 2, randomised, placebo-controlled, double-blind study. Lancet Neurol. 2018;17(3):213-222.
20. Maust DT, Kim HM, Chiang C, et al. Association of the Centers for Medicare & Medicaid Services’ National Partnership to Improve Dementia Care with the use of antipsychotics and other psychotropics in long-term care in the United States from 2009 to 2014. JAMA Intern Med. 2018;178(5):640-647.
21. Dorsey ER, Rabbani A, Gallagher SA, et al. Impact of FDA black box advisory on antipsychotic medication use. Arch Intern Med. 2010;170(1):96-103.
22. U.S. Food and Drug Administration. FDA drug safety communication: FDA revises description of mental health side effects of the stop-smoking medicines Chantix (varenicline) and Zyban (bupropion) to reflect clinical trial findings. https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-revises-description-mental-health-side-effects-stop-smoking. Published December 16, 2016. Accessed October 28, 2019.
Bile acid diarrhea guideline highlights data shortage
The Canadian Association of Gastroenterology (CAG) recently co-published a clinical practice guideline for the management of bile acid diarrhea (BAD) in Clinical Gastroenterology and Hepatology and the Journal of the Canadian Association of Gastroenterology.
Given a minimal evidence base, 16 out of the 17 guideline recommendations are conditional, according to lead author Daniel C. Sadowski, MD, of Royal Alexandra Hospital, Edmonton, Alta., and colleagues. Considering the shortage of high-quality evidence, the panel called for more randomized clinical trials to address current knowledge gaps.
“BAD is an understudied, often underappreciated condition, and questions remain regarding its diagnosis and treatment,” the panelists wrote in Clinical Gastroenterology and Hepatology. “There have been guidelines on the management of chronic diarrhea from the American Gastroenterological Association, and the British Society of Gastroenterology, but diagnosis and management of BAD was not assessed extensively in these publications. The British Society of Gastroenterology updated guidelines on the investigation of chronic diarrhea in adults, published after the consensus meeting, addressed some issues related to BAD.”
For the current guideline, using available evidence and clinical experience, expert panelists from Canada, the United States, and the United Kingdom aimed to “provide a reasonable and practical approach to care for specialists.” The guideline was further reviewed by the CAG Practice Affairs and Clinical Affairs Committees and the CAG Board of Directors.
The guideline first puts BAD in clinical context, noting a chronic diarrhea prevalence rate of approximately 5%. According to the guideline, approximately 1 out of 4 of these patients with chronic diarrhea may have BAD and prevalence of BAD is likely higher among those with other conditions, such as terminal ileal disease.
While BAD may be relatively common, it isn’t necessarily easy to diagnose, the panelists noted.
“The diagnosis of BAD continues to be a challenge, although this may be improved in the future with the general availability of screening serologic tests and other diagnostic tests,” the panelists wrote. “Although a treatment trial with bile acid sequestrants therapy (BAST) often is used, this approach has not been studied adequately, and likely is imprecise, and may lead to both undertreatment and overtreatment.”
Instead, the panelists recommended testing for BAD with 75-selenium homocholic acid taurine (SeHCAT) or 7-alpha-hydroxy-4-cholesten-3-one.
After addressing treatable causes of BAD, the guideline recommends initial therapy with cholestyramine or, if this is poorly tolerated, switching to BAST. However, the panelists advised against BAST for patients with resection or ileal Crohn’s disease, for whom other antidiarrheal agents are more suitable. When appropriate, BAST should be given at the lowest effective dose, with periodic trials of on-demand, intermittent administration, the panelists recommended. When BAST is ineffective, the guideline recommends that clinicians review concurrent medications as a possible cause of BAD or reinvestigate.
Concluding the guideline, the panelists emphasized the need for more high-quality research.
“The group recognized that specific, high-certainty evidence was lacking in many areas and recommended further studies that would improve the data available in future methodologic evaluations,” the panelists wrote.
While improving diagnostic accuracy of BAD should be a major goal of such research, progress is currently limited by an integral shortcoming of diagnostic test accuracy (DTA) studies, the panelists wrote.
“The main challenge in conducting DTA studies for BAD is the lack of a widely accepted or universally agreed-upon reference standard because the condition is defined and classified based on pathophysiologic mechanisms and its response to treatment (BAST),” the panelists wrote. “In addition, the index tests (SeHCAT, C4, FGF19, fecal bile acid assay) provide a continuous measure of metabolic function. Hence, DTA studies are not the most appropriate study design.”
“Therefore, one of the research priorities in BAD is for the scientific and clinical communities to agree on a reference standard that best represents BAD (e.g., response to BAST), with full understanding that the reference standard is and likely will be imperfect.”
The guideline was funded by unrestricted grants from Pendopharm and GE Healthcare Canada. The panelists disclosed relationships with AstraZeneca, AbbVie, Merck, Pfizer, and others.
Review the AGA clinical practice guideline on the laboratory evaluation of functional diarrhea and diarrhea-predominan irritable bowel syndrome in adults at https:/www.gastrojournal.org/article/S0016-5085(19)41083-4/fulltext.
SOURCE: Sadowski DC et al. Clin Gastroenterol Hepatol. 2019 Sep 14. doi: 10.1016/j.cgh.2019.08.062.
The Canadian Association of Gastroenterology (CAG) recently co-published a clinical practice guideline for the management of bile acid diarrhea (BAD) in Clinical Gastroenterology and Hepatology and the Journal of the Canadian Association of Gastroenterology.
Given a minimal evidence base, 16 out of the 17 guideline recommendations are conditional, according to lead author Daniel C. Sadowski, MD, of Royal Alexandra Hospital, Edmonton, Alta., and colleagues. Considering the shortage of high-quality evidence, the panel called for more randomized clinical trials to address current knowledge gaps.
“BAD is an understudied, often underappreciated condition, and questions remain regarding its diagnosis and treatment,” the panelists wrote in Clinical Gastroenterology and Hepatology. “There have been guidelines on the management of chronic diarrhea from the American Gastroenterological Association, and the British Society of Gastroenterology, but diagnosis and management of BAD was not assessed extensively in these publications. The British Society of Gastroenterology updated guidelines on the investigation of chronic diarrhea in adults, published after the consensus meeting, addressed some issues related to BAD.”
For the current guideline, using available evidence and clinical experience, expert panelists from Canada, the United States, and the United Kingdom aimed to “provide a reasonable and practical approach to care for specialists.” The guideline was further reviewed by the CAG Practice Affairs and Clinical Affairs Committees and the CAG Board of Directors.
The guideline first puts BAD in clinical context, noting a chronic diarrhea prevalence rate of approximately 5%. According to the guideline, approximately 1 out of 4 of these patients with chronic diarrhea may have BAD and prevalence of BAD is likely higher among those with other conditions, such as terminal ileal disease.
While BAD may be relatively common, it isn’t necessarily easy to diagnose, the panelists noted.
“The diagnosis of BAD continues to be a challenge, although this may be improved in the future with the general availability of screening serologic tests and other diagnostic tests,” the panelists wrote. “Although a treatment trial with bile acid sequestrants therapy (BAST) often is used, this approach has not been studied adequately, and likely is imprecise, and may lead to both undertreatment and overtreatment.”
Instead, the panelists recommended testing for BAD with 75-selenium homocholic acid taurine (SeHCAT) or 7-alpha-hydroxy-4-cholesten-3-one.
After addressing treatable causes of BAD, the guideline recommends initial therapy with cholestyramine or, if this is poorly tolerated, switching to BAST. However, the panelists advised against BAST for patients with resection or ileal Crohn’s disease, for whom other antidiarrheal agents are more suitable. When appropriate, BAST should be given at the lowest effective dose, with periodic trials of on-demand, intermittent administration, the panelists recommended. When BAST is ineffective, the guideline recommends that clinicians review concurrent medications as a possible cause of BAD or reinvestigate.
Concluding the guideline, the panelists emphasized the need for more high-quality research.
“The group recognized that specific, high-certainty evidence was lacking in many areas and recommended further studies that would improve the data available in future methodologic evaluations,” the panelists wrote.
While improving diagnostic accuracy of BAD should be a major goal of such research, progress is currently limited by an integral shortcoming of diagnostic test accuracy (DTA) studies, the panelists wrote.
“The main challenge in conducting DTA studies for BAD is the lack of a widely accepted or universally agreed-upon reference standard because the condition is defined and classified based on pathophysiologic mechanisms and its response to treatment (BAST),” the panelists wrote. “In addition, the index tests (SeHCAT, C4, FGF19, fecal bile acid assay) provide a continuous measure of metabolic function. Hence, DTA studies are not the most appropriate study design.”
“Therefore, one of the research priorities in BAD is for the scientific and clinical communities to agree on a reference standard that best represents BAD (e.g., response to BAST), with full understanding that the reference standard is and likely will be imperfect.”
The guideline was funded by unrestricted grants from Pendopharm and GE Healthcare Canada. The panelists disclosed relationships with AstraZeneca, AbbVie, Merck, Pfizer, and others.
Review the AGA clinical practice guideline on the laboratory evaluation of functional diarrhea and diarrhea-predominan irritable bowel syndrome in adults at https:/www.gastrojournal.org/article/S0016-5085(19)41083-4/fulltext.
SOURCE: Sadowski DC et al. Clin Gastroenterol Hepatol. 2019 Sep 14. doi: 10.1016/j.cgh.2019.08.062.
The Canadian Association of Gastroenterology (CAG) recently co-published a clinical practice guideline for the management of bile acid diarrhea (BAD) in Clinical Gastroenterology and Hepatology and the Journal of the Canadian Association of Gastroenterology.
Given a minimal evidence base, 16 out of the 17 guideline recommendations are conditional, according to lead author Daniel C. Sadowski, MD, of Royal Alexandra Hospital, Edmonton, Alta., and colleagues. Considering the shortage of high-quality evidence, the panel called for more randomized clinical trials to address current knowledge gaps.
“BAD is an understudied, often underappreciated condition, and questions remain regarding its diagnosis and treatment,” the panelists wrote in Clinical Gastroenterology and Hepatology. “There have been guidelines on the management of chronic diarrhea from the American Gastroenterological Association, and the British Society of Gastroenterology, but diagnosis and management of BAD was not assessed extensively in these publications. The British Society of Gastroenterology updated guidelines on the investigation of chronic diarrhea in adults, published after the consensus meeting, addressed some issues related to BAD.”
For the current guideline, using available evidence and clinical experience, expert panelists from Canada, the United States, and the United Kingdom aimed to “provide a reasonable and practical approach to care for specialists.” The guideline was further reviewed by the CAG Practice Affairs and Clinical Affairs Committees and the CAG Board of Directors.
The guideline first puts BAD in clinical context, noting a chronic diarrhea prevalence rate of approximately 5%. According to the guideline, approximately 1 out of 4 of these patients with chronic diarrhea may have BAD and prevalence of BAD is likely higher among those with other conditions, such as terminal ileal disease.
While BAD may be relatively common, it isn’t necessarily easy to diagnose, the panelists noted.
“The diagnosis of BAD continues to be a challenge, although this may be improved in the future with the general availability of screening serologic tests and other diagnostic tests,” the panelists wrote. “Although a treatment trial with bile acid sequestrants therapy (BAST) often is used, this approach has not been studied adequately, and likely is imprecise, and may lead to both undertreatment and overtreatment.”
Instead, the panelists recommended testing for BAD with 75-selenium homocholic acid taurine (SeHCAT) or 7-alpha-hydroxy-4-cholesten-3-one.
After addressing treatable causes of BAD, the guideline recommends initial therapy with cholestyramine or, if this is poorly tolerated, switching to BAST. However, the panelists advised against BAST for patients with resection or ileal Crohn’s disease, for whom other antidiarrheal agents are more suitable. When appropriate, BAST should be given at the lowest effective dose, with periodic trials of on-demand, intermittent administration, the panelists recommended. When BAST is ineffective, the guideline recommends that clinicians review concurrent medications as a possible cause of BAD or reinvestigate.
Concluding the guideline, the panelists emphasized the need for more high-quality research.
“The group recognized that specific, high-certainty evidence was lacking in many areas and recommended further studies that would improve the data available in future methodologic evaluations,” the panelists wrote.
While improving diagnostic accuracy of BAD should be a major goal of such research, progress is currently limited by an integral shortcoming of diagnostic test accuracy (DTA) studies, the panelists wrote.
“The main challenge in conducting DTA studies for BAD is the lack of a widely accepted or universally agreed-upon reference standard because the condition is defined and classified based on pathophysiologic mechanisms and its response to treatment (BAST),” the panelists wrote. “In addition, the index tests (SeHCAT, C4, FGF19, fecal bile acid assay) provide a continuous measure of metabolic function. Hence, DTA studies are not the most appropriate study design.”
“Therefore, one of the research priorities in BAD is for the scientific and clinical communities to agree on a reference standard that best represents BAD (e.g., response to BAST), with full understanding that the reference standard is and likely will be imperfect.”
The guideline was funded by unrestricted grants from Pendopharm and GE Healthcare Canada. The panelists disclosed relationships with AstraZeneca, AbbVie, Merck, Pfizer, and others.
Review the AGA clinical practice guideline on the laboratory evaluation of functional diarrhea and diarrhea-predominan irritable bowel syndrome in adults at https:/www.gastrojournal.org/article/S0016-5085(19)41083-4/fulltext.
SOURCE: Sadowski DC et al. Clin Gastroenterol Hepatol. 2019 Sep 14. doi: 10.1016/j.cgh.2019.08.062.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Key clinical point: The Canadian Association of Gastroenterology recently published a clinical practice guideline for the management of bile acid diarrhea (BAD).
Major finding: BAD occurs in up to 35% of patients with chronic diarrhea or diarrhea-predominant irritable bowel syndrome.
Study details: A clinical practice guideline for the management of BAD.
Disclosures: The guideline was funded by unrestricted grants from Pendopharm and GE Healthcare Canada. The panelists disclosed relationships with AstraZeneca, AbbVie, Merck, Pfizer, and others.
Source: Sadowski DC et al. Clin Gastroenterol Hepatol. 2019 Sep 14. doi: 10.1016/j.cgh.2019.08.062.
AGA releases clinical practice update for pancreatic necrosis
The American Gastroenterological Association recently issued a clinical practice update for the management of pancreatic necrosis, including 15 recommendations based on a comprehensive literature review and the experiences of leading experts.
Recommendations range from the general, such as the need for a multidisciplinary approach, to the specific, such as the superiority of metal over plastic stents for endoscopic transmural drainage.
The expert review, which was conducted by lead author Todd H. Baron, MD, of the University of North Carolina in Chapel Hill and three other colleagues, was vetted by the AGA Institute Clinical Practice Updates Committee (CPUC) and the AGA Governing Board. In addition, the update underwent external peer review prior to publication in Gastroenterology.
In the update, the authors outlined the clinical landscape for pancreatic necrosis, including challenges posed by complex cases and a mortality rate as high as 30%.
“Successful management of these patients requires expert multidisciplinary care by gastroenterologists, surgeons, interventional radiologists, and specialists in critical care medicine, infectious disease, and nutrition,” the investigators wrote.
They went on to explain how management has evolved over the past 10 years.
“Whereby major surgical intervention and debridement was once the mainstay of therapy for patients with symptomatic necrotic collections, a minimally invasive approach focusing on percutaneous drainage and/or endoscopic drainage or debridement is now favored,” they wrote. They added that debridement is still generally agreed to be the best choice for cases of infected necrosis or patients with sterile necrosis “marked by abdominal pain, nausea, vomiting, and nutritional failure or with associated complications including gastrointestinal luminal obstruction, biliary obstruction, recurrent acute pancreatitis, fistulas, or persistent systemic inflammatory response syndrome (SIRS).”
Other elements of care, however, remain debated, the investigators noted, which has led to variations in multiple aspects of care, such as interventional timing, intravenous fluids, antibiotics, and nutrition. Within this framework, the present practice update is aimed at offering “concise best practice advice for the optimal management of patients with this highly morbid condition.”
Among these pieces of advice, the authors emphasized that routine prophylactic antibiotics and/or antifungals to prevent infected necrosis are unsupported by multiple clinical trials. When infection is suspected, the update recommends broad spectrum intravenous antibiotics, noting that, in most cases, it is unnecessary to perform CT-guided fine-needle aspiration for cultures and gram stain.
Regarding nutrition, the update recommends against “pancreatic rest”; instead, it calls for early oral intake and, if this is not possible, then initiation of total enteral nutrition. Although the authors deemed multiple routes of enteral feeding acceptable, they favored nasogastric or nasoduodenal tubes, when appropriate, because of ease of placement and maintenance. For prolonged total enteral nutrition or patients unable to tolerate nasoenteric feeding, the authors recommended endoscopic feeding tube placement with a percutaneous endoscopic gastrostomy tube for those who can tolerate gastric feeding or a percutaneous endoscopic jejunostomy tube for those who cannot or have a high risk of aspiration.
As described above, the update recommends debridement for cases of infected pancreatic necrosis. Ideally, this should be performed at least 4 weeks after onset, and avoided altogether within the first 2 weeks, because of associated risks of morbidity and mortality; instead, during this acute phase, percutaneous drainage may be considered.
For walled-off pancreatic necrosis, the authors recommended transmural drainage via endoscopic therapy because this mitigates risk of pancreatocutaneous fistula. Percutaneous drainage may be considered in addition to, or in absence of, endoscopic drainage, depending on clinical status.
The remainder of the update covers decisions related to stents, other minimally invasive techniques, open operative debridement, and disconnected left pancreatic remnants, along with discussions of key supporting clinical trials.
The investigators disclosed relationships with Cook Endoscopy, Boston Scientific, Olympus, and others.
SOURCE: Baron TH et al. Gastroenterology. 2019 Aug 31. doi: 10.1053/j.gastro.2019.07.064.
The American Gastroenterological Association recently issued a clinical practice update for the management of pancreatic necrosis, including 15 recommendations based on a comprehensive literature review and the experiences of leading experts.
Recommendations range from the general, such as the need for a multidisciplinary approach, to the specific, such as the superiority of metal over plastic stents for endoscopic transmural drainage.
The expert review, which was conducted by lead author Todd H. Baron, MD, of the University of North Carolina in Chapel Hill and three other colleagues, was vetted by the AGA Institute Clinical Practice Updates Committee (CPUC) and the AGA Governing Board. In addition, the update underwent external peer review prior to publication in Gastroenterology.
In the update, the authors outlined the clinical landscape for pancreatic necrosis, including challenges posed by complex cases and a mortality rate as high as 30%.
“Successful management of these patients requires expert multidisciplinary care by gastroenterologists, surgeons, interventional radiologists, and specialists in critical care medicine, infectious disease, and nutrition,” the investigators wrote.
They went on to explain how management has evolved over the past 10 years.
“Whereby major surgical intervention and debridement was once the mainstay of therapy for patients with symptomatic necrotic collections, a minimally invasive approach focusing on percutaneous drainage and/or endoscopic drainage or debridement is now favored,” they wrote. They added that debridement is still generally agreed to be the best choice for cases of infected necrosis or patients with sterile necrosis “marked by abdominal pain, nausea, vomiting, and nutritional failure or with associated complications including gastrointestinal luminal obstruction, biliary obstruction, recurrent acute pancreatitis, fistulas, or persistent systemic inflammatory response syndrome (SIRS).”
Other elements of care, however, remain debated, the investigators noted, which has led to variations in multiple aspects of care, such as interventional timing, intravenous fluids, antibiotics, and nutrition. Within this framework, the present practice update is aimed at offering “concise best practice advice for the optimal management of patients with this highly morbid condition.”
Among these pieces of advice, the authors emphasized that routine prophylactic antibiotics and/or antifungals to prevent infected necrosis are unsupported by multiple clinical trials. When infection is suspected, the update recommends broad spectrum intravenous antibiotics, noting that, in most cases, it is unnecessary to perform CT-guided fine-needle aspiration for cultures and gram stain.
Regarding nutrition, the update recommends against “pancreatic rest”; instead, it calls for early oral intake and, if this is not possible, then initiation of total enteral nutrition. Although the authors deemed multiple routes of enteral feeding acceptable, they favored nasogastric or nasoduodenal tubes, when appropriate, because of ease of placement and maintenance. For prolonged total enteral nutrition or patients unable to tolerate nasoenteric feeding, the authors recommended endoscopic feeding tube placement with a percutaneous endoscopic gastrostomy tube for those who can tolerate gastric feeding or a percutaneous endoscopic jejunostomy tube for those who cannot or have a high risk of aspiration.
As described above, the update recommends debridement for cases of infected pancreatic necrosis. Ideally, this should be performed at least 4 weeks after onset, and avoided altogether within the first 2 weeks, because of associated risks of morbidity and mortality; instead, during this acute phase, percutaneous drainage may be considered.
For walled-off pancreatic necrosis, the authors recommended transmural drainage via endoscopic therapy because this mitigates risk of pancreatocutaneous fistula. Percutaneous drainage may be considered in addition to, or in absence of, endoscopic drainage, depending on clinical status.
The remainder of the update covers decisions related to stents, other minimally invasive techniques, open operative debridement, and disconnected left pancreatic remnants, along with discussions of key supporting clinical trials.
The investigators disclosed relationships with Cook Endoscopy, Boston Scientific, Olympus, and others.
SOURCE: Baron TH et al. Gastroenterology. 2019 Aug 31. doi: 10.1053/j.gastro.2019.07.064.
The American Gastroenterological Association recently issued a clinical practice update for the management of pancreatic necrosis, including 15 recommendations based on a comprehensive literature review and the experiences of leading experts.
Recommendations range from the general, such as the need for a multidisciplinary approach, to the specific, such as the superiority of metal over plastic stents for endoscopic transmural drainage.
The expert review, which was conducted by lead author Todd H. Baron, MD, of the University of North Carolina in Chapel Hill and three other colleagues, was vetted by the AGA Institute Clinical Practice Updates Committee (CPUC) and the AGA Governing Board. In addition, the update underwent external peer review prior to publication in Gastroenterology.
In the update, the authors outlined the clinical landscape for pancreatic necrosis, including challenges posed by complex cases and a mortality rate as high as 30%.
“Successful management of these patients requires expert multidisciplinary care by gastroenterologists, surgeons, interventional radiologists, and specialists in critical care medicine, infectious disease, and nutrition,” the investigators wrote.
They went on to explain how management has evolved over the past 10 years.
“Whereby major surgical intervention and debridement was once the mainstay of therapy for patients with symptomatic necrotic collections, a minimally invasive approach focusing on percutaneous drainage and/or endoscopic drainage or debridement is now favored,” they wrote. They added that debridement is still generally agreed to be the best choice for cases of infected necrosis or patients with sterile necrosis “marked by abdominal pain, nausea, vomiting, and nutritional failure or with associated complications including gastrointestinal luminal obstruction, biliary obstruction, recurrent acute pancreatitis, fistulas, or persistent systemic inflammatory response syndrome (SIRS).”
Other elements of care, however, remain debated, the investigators noted, which has led to variations in multiple aspects of care, such as interventional timing, intravenous fluids, antibiotics, and nutrition. Within this framework, the present practice update is aimed at offering “concise best practice advice for the optimal management of patients with this highly morbid condition.”
Among these pieces of advice, the authors emphasized that routine prophylactic antibiotics and/or antifungals to prevent infected necrosis are unsupported by multiple clinical trials. When infection is suspected, the update recommends broad spectrum intravenous antibiotics, noting that, in most cases, it is unnecessary to perform CT-guided fine-needle aspiration for cultures and gram stain.
Regarding nutrition, the update recommends against “pancreatic rest”; instead, it calls for early oral intake and, if this is not possible, then initiation of total enteral nutrition. Although the authors deemed multiple routes of enteral feeding acceptable, they favored nasogastric or nasoduodenal tubes, when appropriate, because of ease of placement and maintenance. For prolonged total enteral nutrition or patients unable to tolerate nasoenteric feeding, the authors recommended endoscopic feeding tube placement with a percutaneous endoscopic gastrostomy tube for those who can tolerate gastric feeding or a percutaneous endoscopic jejunostomy tube for those who cannot or have a high risk of aspiration.
As described above, the update recommends debridement for cases of infected pancreatic necrosis. Ideally, this should be performed at least 4 weeks after onset, and avoided altogether within the first 2 weeks, because of associated risks of morbidity and mortality; instead, during this acute phase, percutaneous drainage may be considered.
For walled-off pancreatic necrosis, the authors recommended transmural drainage via endoscopic therapy because this mitigates risk of pancreatocutaneous fistula. Percutaneous drainage may be considered in addition to, or in absence of, endoscopic drainage, depending on clinical status.
The remainder of the update covers decisions related to stents, other minimally invasive techniques, open operative debridement, and disconnected left pancreatic remnants, along with discussions of key supporting clinical trials.
The investigators disclosed relationships with Cook Endoscopy, Boston Scientific, Olympus, and others.
SOURCE: Baron TH et al. Gastroenterology. 2019 Aug 31. doi: 10.1053/j.gastro.2019.07.064.
FROM GASTROENTEROLOGY
Key clinical point: The American Gastroenterological Association has issued a clinical practice update for the management of pancreatic necrosis.
Major finding: N/A
Study details: A clinical practice update for the management of pancreatic necrosis.
Disclosures: The investigators disclosed relationships with Cook Endoscopy, Boston Scientific, Olympus, and others.
Source: Baron TH et al. Gastroenterology. 2019 Aug 31. doi: 10.1053/j.gastro.2019.07.064.
Trial finds three drugs equally effective for established status epilepticus
according to a study published Nov. 27 in the New England Journal of Medicine. The effectiveness and safety of the intravenous medications do not differ significantly, the researchers wrote.
“Having three equally effective second-line intravenous medications means that the clinician may choose a drug that takes into account individual situations,” wrote Phil E.M. Smith, MD, in an accompanying editorial (doi: 10.1056/NEJMe1913775). Clinicians may consider “factors such as the presumed underlying cause of status epilepticus; coexisting conditions, including allergy, liver and renal disease, hypotension, propensity to cardiac arrhythmia, and alcohol and drug dependence; the currently prescribed antiepileptic treatment; the cost of the medication; and governmental agency drug approval,” said Dr. Smith, who is affiliated with University Hospital of Wales in Cardiff.
A gap in guidance
Evidence supports benzodiazepines as the initial treatment for status epilepticus, but these drugs do not work in up to a third of patients, said first study author Jaideep Kapur, MBBS, PhD, and colleagues. “Clinical guidelines emphasize the need for rapid control of benzodiazepine-refractory status epilepticus but do not provide guidance regarding the choice of medication on the basis of either efficacy or safety,” they wrote. Dr. Kapur is a professor of neurology and the director of UVA Brain Institute at University of Virginia in Charlottesville.
Levetiracetam, fosphenytoin, and valproate are the three most commonly used medications for benzodiazepine-refractory status epilepticus. The Food and Drug Administration has labeled fosphenytoin for this indication in adults, and none of the drugs is approved for children. To determine the superiority or inferiority of these medications, the researchers conducted the Established Status Epilepticus Treatment Trial (ESETT). The blinded, comparative-effectiveness trial enrolled 384 patients at 57 hospital EDs in the United States. Patients were aged 2 years or older, had received a generally accepted cumulative dose of benzodiazepines for generalized convulsive seizures lasting more than 5 minutes and continued to have persistent or recurrent convulsions between 5-30 minutes after the last dose of benzodiazepine.
Patients randomly received one of the three trial drugs, which “were identical in appearance, formulation, packaging, and administration,” the authors said. The primary outcome was absence of clinically apparent seizures and improving responsiveness at 60 minutes after the start of the infusion without administration of additional anticonvulsant medication. ED physicians determined the presence of seizure and improvement in responsiveness.
Trial was stopped for futility
The trial included 400 enrollments of 384 unique patients during 2015-2017. Sixteen patients were enrolled twice, and their second enrollments were not included in the intention-to-treat analysis. A planned interim analysis after 400 enrollments to assess the likelihood of success or futility found that the trial had met the futility criterion. “There was a 1% chance of showing a most effective or least effective treatment if the trial were to continue to the maximum sample size” of 795 patients, Dr. Kapur and coauthors wrote. The researchers continued enrollment in a pediatric subcohort for a planned subgroup analysis by age.
In all, 55% of the patients were male, 43% were black, and 16% were Hispanic. The population was 39% children and adolescents, 48% adults aged 18-65 years, and 13% older than 65 years. Most patients had a final diagnosis of status epilepticus (87%). Other final diagnoses included psychogenic nonepileptic seizures (10%).
At 60 minutes after treatment administration, absence of seizures and improved responsiveness occurred in 47% of patients who received levetiracetam, 45% who received fosphenytoin, and 46% who received valproate.
In 39 patients for whom the researchers had reliable information about time to seizure cessation, median time to seizure cessation numerically favored valproate (7 minutes for valproate vs. 10.5 minutes for levetiracetam vs. 11.7 minutes for fosphenytoin), but the number of patients was limited, the authors noted.
“Hypotension and endotracheal intubation were more frequent with fosphenytoin than with the other two drugs, and deaths were more frequent with levetiracetam, but these differences were not significant,” wrote Dr. Kapur and colleagues. Seven patients who received levetiracetam died, compared with three who received fosphenytoin and two who received valproate. Life-threatening hypotension occurred in 3.2% of patients who received fosphenytoin, compared with 1.6% who received valproate and 0.7% who received levetiracetam. Endotracheal intubation occurred in 26.4% or patients who received fosphenytoin, compared with 20% of patients in the levetiracetam group and 16.8% in the valproate group.
The trial’s limitations include the enrollment of patients with psychogenic nonepileptic seizures and the use of clinical instead of electroencephalographic criteria for the primary outcome measure, the investigators wrote.
Dr. Smith noted that third- and fourth-line management of status epilepticus is not supported by high-quality evidence, and further studies are needed. Given the evidence from ESETT, “the practical challenge for the management of status epilepticus remains the same as in the past: ensuring that clinicians are familiar with, and follow, a treatment protocol,” he said.
The trial was funded by the National Institute of Neurological Disorders and Stroke. Dr. Kapur had no financial disclosures. A coauthor holds a patent on intravenous carbamazepine and intellectual property on intravenous topiramate. Other coauthors have ties to pharmaceutical and medical device companies.
Dr. Smith is coeditor of Practical Neurology and a member of the U.K. National Institute for Health and Clinical Excellence (NICE) guidelines committee for epilepsy.
SOURCE: Kapur J et al. N Engl J Med. 2019 Nov 27. doi: 10.1056/NEJMoa1905795.
according to a study published Nov. 27 in the New England Journal of Medicine. The effectiveness and safety of the intravenous medications do not differ significantly, the researchers wrote.
“Having three equally effective second-line intravenous medications means that the clinician may choose a drug that takes into account individual situations,” wrote Phil E.M. Smith, MD, in an accompanying editorial (doi: 10.1056/NEJMe1913775). Clinicians may consider “factors such as the presumed underlying cause of status epilepticus; coexisting conditions, including allergy, liver and renal disease, hypotension, propensity to cardiac arrhythmia, and alcohol and drug dependence; the currently prescribed antiepileptic treatment; the cost of the medication; and governmental agency drug approval,” said Dr. Smith, who is affiliated with University Hospital of Wales in Cardiff.
A gap in guidance
Evidence supports benzodiazepines as the initial treatment for status epilepticus, but these drugs do not work in up to a third of patients, said first study author Jaideep Kapur, MBBS, PhD, and colleagues. “Clinical guidelines emphasize the need for rapid control of benzodiazepine-refractory status epilepticus but do not provide guidance regarding the choice of medication on the basis of either efficacy or safety,” they wrote. Dr. Kapur is a professor of neurology and the director of UVA Brain Institute at University of Virginia in Charlottesville.
Levetiracetam, fosphenytoin, and valproate are the three most commonly used medications for benzodiazepine-refractory status epilepticus. The Food and Drug Administration has labeled fosphenytoin for this indication in adults, and none of the drugs is approved for children. To determine the superiority or inferiority of these medications, the researchers conducted the Established Status Epilepticus Treatment Trial (ESETT). The blinded, comparative-effectiveness trial enrolled 384 patients at 57 hospital EDs in the United States. Patients were aged 2 years or older, had received a generally accepted cumulative dose of benzodiazepines for generalized convulsive seizures lasting more than 5 minutes and continued to have persistent or recurrent convulsions between 5-30 minutes after the last dose of benzodiazepine.
Patients randomly received one of the three trial drugs, which “were identical in appearance, formulation, packaging, and administration,” the authors said. The primary outcome was absence of clinically apparent seizures and improving responsiveness at 60 minutes after the start of the infusion without administration of additional anticonvulsant medication. ED physicians determined the presence of seizure and improvement in responsiveness.
Trial was stopped for futility
The trial included 400 enrollments of 384 unique patients during 2015-2017. Sixteen patients were enrolled twice, and their second enrollments were not included in the intention-to-treat analysis. A planned interim analysis after 400 enrollments to assess the likelihood of success or futility found that the trial had met the futility criterion. “There was a 1% chance of showing a most effective or least effective treatment if the trial were to continue to the maximum sample size” of 795 patients, Dr. Kapur and coauthors wrote. The researchers continued enrollment in a pediatric subcohort for a planned subgroup analysis by age.
In all, 55% of the patients were male, 43% were black, and 16% were Hispanic. The population was 39% children and adolescents, 48% adults aged 18-65 years, and 13% older than 65 years. Most patients had a final diagnosis of status epilepticus (87%). Other final diagnoses included psychogenic nonepileptic seizures (10%).
At 60 minutes after treatment administration, absence of seizures and improved responsiveness occurred in 47% of patients who received levetiracetam, 45% who received fosphenytoin, and 46% who received valproate.
In 39 patients for whom the researchers had reliable information about time to seizure cessation, median time to seizure cessation numerically favored valproate (7 minutes for valproate vs. 10.5 minutes for levetiracetam vs. 11.7 minutes for fosphenytoin), but the number of patients was limited, the authors noted.
“Hypotension and endotracheal intubation were more frequent with fosphenytoin than with the other two drugs, and deaths were more frequent with levetiracetam, but these differences were not significant,” wrote Dr. Kapur and colleagues. Seven patients who received levetiracetam died, compared with three who received fosphenytoin and two who received valproate. Life-threatening hypotension occurred in 3.2% of patients who received fosphenytoin, compared with 1.6% who received valproate and 0.7% who received levetiracetam. Endotracheal intubation occurred in 26.4% or patients who received fosphenytoin, compared with 20% of patients in the levetiracetam group and 16.8% in the valproate group.
The trial’s limitations include the enrollment of patients with psychogenic nonepileptic seizures and the use of clinical instead of electroencephalographic criteria for the primary outcome measure, the investigators wrote.
Dr. Smith noted that third- and fourth-line management of status epilepticus is not supported by high-quality evidence, and further studies are needed. Given the evidence from ESETT, “the practical challenge for the management of status epilepticus remains the same as in the past: ensuring that clinicians are familiar with, and follow, a treatment protocol,” he said.
The trial was funded by the National Institute of Neurological Disorders and Stroke. Dr. Kapur had no financial disclosures. A coauthor holds a patent on intravenous carbamazepine and intellectual property on intravenous topiramate. Other coauthors have ties to pharmaceutical and medical device companies.
Dr. Smith is coeditor of Practical Neurology and a member of the U.K. National Institute for Health and Clinical Excellence (NICE) guidelines committee for epilepsy.
SOURCE: Kapur J et al. N Engl J Med. 2019 Nov 27. doi: 10.1056/NEJMoa1905795.
according to a study published Nov. 27 in the New England Journal of Medicine. The effectiveness and safety of the intravenous medications do not differ significantly, the researchers wrote.
“Having three equally effective second-line intravenous medications means that the clinician may choose a drug that takes into account individual situations,” wrote Phil E.M. Smith, MD, in an accompanying editorial (doi: 10.1056/NEJMe1913775). Clinicians may consider “factors such as the presumed underlying cause of status epilepticus; coexisting conditions, including allergy, liver and renal disease, hypotension, propensity to cardiac arrhythmia, and alcohol and drug dependence; the currently prescribed antiepileptic treatment; the cost of the medication; and governmental agency drug approval,” said Dr. Smith, who is affiliated with University Hospital of Wales in Cardiff.
A gap in guidance
Evidence supports benzodiazepines as the initial treatment for status epilepticus, but these drugs do not work in up to a third of patients, said first study author Jaideep Kapur, MBBS, PhD, and colleagues. “Clinical guidelines emphasize the need for rapid control of benzodiazepine-refractory status epilepticus but do not provide guidance regarding the choice of medication on the basis of either efficacy or safety,” they wrote. Dr. Kapur is a professor of neurology and the director of UVA Brain Institute at University of Virginia in Charlottesville.
Levetiracetam, fosphenytoin, and valproate are the three most commonly used medications for benzodiazepine-refractory status epilepticus. The Food and Drug Administration has labeled fosphenytoin for this indication in adults, and none of the drugs is approved for children. To determine the superiority or inferiority of these medications, the researchers conducted the Established Status Epilepticus Treatment Trial (ESETT). The blinded, comparative-effectiveness trial enrolled 384 patients at 57 hospital EDs in the United States. Patients were aged 2 years or older, had received a generally accepted cumulative dose of benzodiazepines for generalized convulsive seizures lasting more than 5 minutes and continued to have persistent or recurrent convulsions between 5-30 minutes after the last dose of benzodiazepine.
Patients randomly received one of the three trial drugs, which “were identical in appearance, formulation, packaging, and administration,” the authors said. The primary outcome was absence of clinically apparent seizures and improving responsiveness at 60 minutes after the start of the infusion without administration of additional anticonvulsant medication. ED physicians determined the presence of seizure and improvement in responsiveness.
Trial was stopped for futility
The trial included 400 enrollments of 384 unique patients during 2015-2017. Sixteen patients were enrolled twice, and their second enrollments were not included in the intention-to-treat analysis. A planned interim analysis after 400 enrollments to assess the likelihood of success or futility found that the trial had met the futility criterion. “There was a 1% chance of showing a most effective or least effective treatment if the trial were to continue to the maximum sample size” of 795 patients, Dr. Kapur and coauthors wrote. The researchers continued enrollment in a pediatric subcohort for a planned subgroup analysis by age.
In all, 55% of the patients were male, 43% were black, and 16% were Hispanic. The population was 39% children and adolescents, 48% adults aged 18-65 years, and 13% older than 65 years. Most patients had a final diagnosis of status epilepticus (87%). Other final diagnoses included psychogenic nonepileptic seizures (10%).
At 60 minutes after treatment administration, absence of seizures and improved responsiveness occurred in 47% of patients who received levetiracetam, 45% who received fosphenytoin, and 46% who received valproate.
In 39 patients for whom the researchers had reliable information about time to seizure cessation, median time to seizure cessation numerically favored valproate (7 minutes for valproate vs. 10.5 minutes for levetiracetam vs. 11.7 minutes for fosphenytoin), but the number of patients was limited, the authors noted.
“Hypotension and endotracheal intubation were more frequent with fosphenytoin than with the other two drugs, and deaths were more frequent with levetiracetam, but these differences were not significant,” wrote Dr. Kapur and colleagues. Seven patients who received levetiracetam died, compared with three who received fosphenytoin and two who received valproate. Life-threatening hypotension occurred in 3.2% of patients who received fosphenytoin, compared with 1.6% who received valproate and 0.7% who received levetiracetam. Endotracheal intubation occurred in 26.4% or patients who received fosphenytoin, compared with 20% of patients in the levetiracetam group and 16.8% in the valproate group.
The trial’s limitations include the enrollment of patients with psychogenic nonepileptic seizures and the use of clinical instead of electroencephalographic criteria for the primary outcome measure, the investigators wrote.
Dr. Smith noted that third- and fourth-line management of status epilepticus is not supported by high-quality evidence, and further studies are needed. Given the evidence from ESETT, “the practical challenge for the management of status epilepticus remains the same as in the past: ensuring that clinicians are familiar with, and follow, a treatment protocol,” he said.
The trial was funded by the National Institute of Neurological Disorders and Stroke. Dr. Kapur had no financial disclosures. A coauthor holds a patent on intravenous carbamazepine and intellectual property on intravenous topiramate. Other coauthors have ties to pharmaceutical and medical device companies.
Dr. Smith is coeditor of Practical Neurology and a member of the U.K. National Institute for Health and Clinical Excellence (NICE) guidelines committee for epilepsy.
SOURCE: Kapur J et al. N Engl J Med. 2019 Nov 27. doi: 10.1056/NEJMoa1905795.
FROM NEJM
Key clinical point: Among children and adults with benzodiazepine-refractory status epilepticus, fosphenytoin, valproate, and levetiracetam each stop seizures by 60 minutes in approximately half of patients.
Major finding: Absence of seizures and improved responsiveness occurred in 47% of patients who received levetiracetam, 45% who received fosphenytoin, and 46% who received valproate.
Study details: The Established Status Epilepticus Treatment Trial (ESETT) was a blinded, comparative-effectiveness trial that enrolled 384 patients at 57 hospital EDs in the United States.
Disclosures: The trial was funded by the National Institute of Neurological Disorders and Stroke. Dr. Kapur had no financial disclosures. A coauthor holds a patent on intravenous carbamazepine and intellectual property on intravenous topiramate. Other coauthors have ties to pharmaceutical and medical device companies.
Source: Kapur J et al. N Engl J Med. 2019 Nov 27. doi: 10.1056/NEJMoa1905795.
ART treatment at birth found to benefit neonates with HIV
Initiating antiretroviral therapy within an hour after birth, rather than waiting a few weeks, lowers the reservoir of HIV virus and improves immune response, early results from an ongoing study in Botswana, Africa, showed.
Despite advances in treatment programs during pregnancy that prevent mother to child HIV transmission, 300-500 pediatric HIV infections occur each day in sub-Saharan Africa, Roger Shapiro, MD, MPH, said during a media teleconference organized by the American Association for the Advancement of Science. “Most pediatric HIV diagnosis programs currently test children at 4-6 weeks of age to identify infections that occur either in pregnancy or during delivery,” said Dr. Shapiro, associate professor of immunology and infectious diseases at the Harvard T.H. Chan School of Public Health, Boston. “However, these programs miss the opportunity to begin immediate antiretroviral treatment for children who can be identified earlier. There are benefits to starting treatment and arresting HIV replication in the first week of life. These include limiting the viral reservoir or the population of infected cells, limiting potentially harmful immune responses to the virus, and preventing the rapid decline in health that can occur in the early weeks of HIV infection in infants. Without treatment, 50% of HIV-infected children regress to death by 2 years. Starting treatment in the first weeks or months of life has been shown to improve survival.”
With these benefits in mind, he and his associates initiated the Early Infant Treatment (EIT) study in 2015 to diagnose and treat HIV infected infants in Botswana in the first week of life or as early as possible after infection. They screened more than 10,000 children and identified 40 that were HIV infected. “This low transmission rate is a testament to the fact that most HIV-positive women in Botswana receive three-drug treatment in pregnancy, which is highly successful in blocking transmission,” Dr. Shapiro said. “When we identified an HIV-infected infant, we consented mothers to allow us to start treatment right away. We used a series of regimens because there are limited options. The available options include older drugs, some of which are no longer used for adults but which were the only options for children.”
The researchers initiated three initial drugs approved for newborns: nevirapine, zidovudine, and lamivudine, and then changed the regimen slightly after a few weeks, when they used ritonavir-boosted lopinavir, plus the lamivudine and zidovudine. “We followed the children weekly at first, then at monthly refill visits, and kept close track of how they were taking the medicines and the level of virus in each child’s blood,” Dr. Shapiro said.
In a manuscript published online in Science Translational Medicine on Nov. 27, 2019, he and his associates reported results of the first 10 children enrolled in the EIT study who reached about 96 weeks on treatment. For comparison, they also enrolled a group of children as controls, who started treatment later in the first year of life, after being identified at a more standard time of 4-6 weeks. Tests performed included droplet digital polymerase chain reaction, HIV near-full-genome sequencing, whole-genome amplification, and flow cytometry.
“What we wanted to focus on are the HIV reservoir cells that are persisting in the setting of antiretroviral treatment,” study coauthor Mathias Lichterfeld, MD, PhD, explained during the teleconference. “Those are the cells that would cause viral rebound if treatment were to be interrupted. We used complex technology to look at these cells, using next-generation sequencing, which allows us to identify those cells that harbor HIV that has the ability to initiate new viral replication.”
He and his colleagues observed that the number of reservoir cells was significantly smaller than in adults who were on ART for a median of 16 years. It also was smaller than in infected infants who started ART treatment weeks after birth.
In addition, immune activation was reduced in the cohort of infants who were treated immediately after birth.
“We are seeing a distinct advantage of early treatment initiation,” said Dr. Lichterfeld of the infectious disease division at Brigham and Women’s Hospital, Boston. “By doing these assays we see both virological benefits in terms of a very-low reservoir size, and we see immune system characteristics that are also associated with better abilities for antimicrobial immune defense and a lower level of immune activation.”
Another study coauthor, Daniel R. Kuritzkes, MD, chief of the infectious disease division at Brigham and Women’s Hospital, said the findings show “how critically important” it is to extend studies of HIV cure or long-term remission to infants and children. “Very-early intervention in neonates limits the size of the reservoir and offers us the best opportunity for future interventions aimed at cure and long-term drug-free remission of HIV infection,” he said. “We don’t think the current intervention is itself curative, but it sets the stage for the capacity to offer additional innovative interventions in the future. Beyond the importance of this work for cure research per se, this very early intervention in neonates also has the potential of conferring important clinical benefits to the children who participated in this study. Finally, our study demonstrates the feasibility and importance of doing this type of research in neonates in resource-limited settings, given the appropriate infrastructure.”
EIT is supported by the National Institutes of Health. Dr. Lichterfeld disclosed having received speaking and consulting honoraria from Merck and Gilead. Dr. Kuritzkes disclosed having received consulting honoraria and/or research support from Gilead, Merck, and ViiV.
SOURCE: Garcia-Broncano P et al. Sci Transl Med. 2019 Nov 27. eaax7350.
Initiating antiretroviral therapy within an hour after birth, rather than waiting a few weeks, lowers the reservoir of HIV virus and improves immune response, early results from an ongoing study in Botswana, Africa, showed.
Despite advances in treatment programs during pregnancy that prevent mother to child HIV transmission, 300-500 pediatric HIV infections occur each day in sub-Saharan Africa, Roger Shapiro, MD, MPH, said during a media teleconference organized by the American Association for the Advancement of Science. “Most pediatric HIV diagnosis programs currently test children at 4-6 weeks of age to identify infections that occur either in pregnancy or during delivery,” said Dr. Shapiro, associate professor of immunology and infectious diseases at the Harvard T.H. Chan School of Public Health, Boston. “However, these programs miss the opportunity to begin immediate antiretroviral treatment for children who can be identified earlier. There are benefits to starting treatment and arresting HIV replication in the first week of life. These include limiting the viral reservoir or the population of infected cells, limiting potentially harmful immune responses to the virus, and preventing the rapid decline in health that can occur in the early weeks of HIV infection in infants. Without treatment, 50% of HIV-infected children regress to death by 2 years. Starting treatment in the first weeks or months of life has been shown to improve survival.”
With these benefits in mind, he and his associates initiated the Early Infant Treatment (EIT) study in 2015 to diagnose and treat HIV infected infants in Botswana in the first week of life or as early as possible after infection. They screened more than 10,000 children and identified 40 that were HIV infected. “This low transmission rate is a testament to the fact that most HIV-positive women in Botswana receive three-drug treatment in pregnancy, which is highly successful in blocking transmission,” Dr. Shapiro said. “When we identified an HIV-infected infant, we consented mothers to allow us to start treatment right away. We used a series of regimens because there are limited options. The available options include older drugs, some of which are no longer used for adults but which were the only options for children.”
The researchers initiated three initial drugs approved for newborns: nevirapine, zidovudine, and lamivudine, and then changed the regimen slightly after a few weeks, when they used ritonavir-boosted lopinavir, plus the lamivudine and zidovudine. “We followed the children weekly at first, then at monthly refill visits, and kept close track of how they were taking the medicines and the level of virus in each child’s blood,” Dr. Shapiro said.
In a manuscript published online in Science Translational Medicine on Nov. 27, 2019, he and his associates reported results of the first 10 children enrolled in the EIT study who reached about 96 weeks on treatment. For comparison, they also enrolled a group of children as controls, who started treatment later in the first year of life, after being identified at a more standard time of 4-6 weeks. Tests performed included droplet digital polymerase chain reaction, HIV near-full-genome sequencing, whole-genome amplification, and flow cytometry.
“What we wanted to focus on are the HIV reservoir cells that are persisting in the setting of antiretroviral treatment,” study coauthor Mathias Lichterfeld, MD, PhD, explained during the teleconference. “Those are the cells that would cause viral rebound if treatment were to be interrupted. We used complex technology to look at these cells, using next-generation sequencing, which allows us to identify those cells that harbor HIV that has the ability to initiate new viral replication.”
He and his colleagues observed that the number of reservoir cells was significantly smaller than in adults who were on ART for a median of 16 years. It also was smaller than in infected infants who started ART treatment weeks after birth.
In addition, immune activation was reduced in the cohort of infants who were treated immediately after birth.
“We are seeing a distinct advantage of early treatment initiation,” said Dr. Lichterfeld of the infectious disease division at Brigham and Women’s Hospital, Boston. “By doing these assays we see both virological benefits in terms of a very-low reservoir size, and we see immune system characteristics that are also associated with better abilities for antimicrobial immune defense and a lower level of immune activation.”
Another study coauthor, Daniel R. Kuritzkes, MD, chief of the infectious disease division at Brigham and Women’s Hospital, said the findings show “how critically important” it is to extend studies of HIV cure or long-term remission to infants and children. “Very-early intervention in neonates limits the size of the reservoir and offers us the best opportunity for future interventions aimed at cure and long-term drug-free remission of HIV infection,” he said. “We don’t think the current intervention is itself curative, but it sets the stage for the capacity to offer additional innovative interventions in the future. Beyond the importance of this work for cure research per se, this very early intervention in neonates also has the potential of conferring important clinical benefits to the children who participated in this study. Finally, our study demonstrates the feasibility and importance of doing this type of research in neonates in resource-limited settings, given the appropriate infrastructure.”
EIT is supported by the National Institutes of Health. Dr. Lichterfeld disclosed having received speaking and consulting honoraria from Merck and Gilead. Dr. Kuritzkes disclosed having received consulting honoraria and/or research support from Gilead, Merck, and ViiV.
SOURCE: Garcia-Broncano P et al. Sci Transl Med. 2019 Nov 27. eaax7350.
Initiating antiretroviral therapy within an hour after birth, rather than waiting a few weeks, lowers the reservoir of HIV virus and improves immune response, early results from an ongoing study in Botswana, Africa, showed.
Despite advances in treatment programs during pregnancy that prevent mother to child HIV transmission, 300-500 pediatric HIV infections occur each day in sub-Saharan Africa, Roger Shapiro, MD, MPH, said during a media teleconference organized by the American Association for the Advancement of Science. “Most pediatric HIV diagnosis programs currently test children at 4-6 weeks of age to identify infections that occur either in pregnancy or during delivery,” said Dr. Shapiro, associate professor of immunology and infectious diseases at the Harvard T.H. Chan School of Public Health, Boston. “However, these programs miss the opportunity to begin immediate antiretroviral treatment for children who can be identified earlier. There are benefits to starting treatment and arresting HIV replication in the first week of life. These include limiting the viral reservoir or the population of infected cells, limiting potentially harmful immune responses to the virus, and preventing the rapid decline in health that can occur in the early weeks of HIV infection in infants. Without treatment, 50% of HIV-infected children regress to death by 2 years. Starting treatment in the first weeks or months of life has been shown to improve survival.”
With these benefits in mind, he and his associates initiated the Early Infant Treatment (EIT) study in 2015 to diagnose and treat HIV infected infants in Botswana in the first week of life or as early as possible after infection. They screened more than 10,000 children and identified 40 that were HIV infected. “This low transmission rate is a testament to the fact that most HIV-positive women in Botswana receive three-drug treatment in pregnancy, which is highly successful in blocking transmission,” Dr. Shapiro said. “When we identified an HIV-infected infant, we consented mothers to allow us to start treatment right away. We used a series of regimens because there are limited options. The available options include older drugs, some of which are no longer used for adults but which were the only options for children.”
The researchers initiated three initial drugs approved for newborns: nevirapine, zidovudine, and lamivudine, and then changed the regimen slightly after a few weeks, when they used ritonavir-boosted lopinavir, plus the lamivudine and zidovudine. “We followed the children weekly at first, then at monthly refill visits, and kept close track of how they were taking the medicines and the level of virus in each child’s blood,” Dr. Shapiro said.
In a manuscript published online in Science Translational Medicine on Nov. 27, 2019, he and his associates reported results of the first 10 children enrolled in the EIT study who reached about 96 weeks on treatment. For comparison, they also enrolled a group of children as controls, who started treatment later in the first year of life, after being identified at a more standard time of 4-6 weeks. Tests performed included droplet digital polymerase chain reaction, HIV near-full-genome sequencing, whole-genome amplification, and flow cytometry.
“What we wanted to focus on are the HIV reservoir cells that are persisting in the setting of antiretroviral treatment,” study coauthor Mathias Lichterfeld, MD, PhD, explained during the teleconference. “Those are the cells that would cause viral rebound if treatment were to be interrupted. We used complex technology to look at these cells, using next-generation sequencing, which allows us to identify those cells that harbor HIV that has the ability to initiate new viral replication.”
He and his colleagues observed that the number of reservoir cells was significantly smaller than in adults who were on ART for a median of 16 years. It also was smaller than in infected infants who started ART treatment weeks after birth.
In addition, immune activation was reduced in the cohort of infants who were treated immediately after birth.
“We are seeing a distinct advantage of early treatment initiation,” said Dr. Lichterfeld of the infectious disease division at Brigham and Women’s Hospital, Boston. “By doing these assays we see both virological benefits in terms of a very-low reservoir size, and we see immune system characteristics that are also associated with better abilities for antimicrobial immune defense and a lower level of immune activation.”
Another study coauthor, Daniel R. Kuritzkes, MD, chief of the infectious disease division at Brigham and Women’s Hospital, said the findings show “how critically important” it is to extend studies of HIV cure or long-term remission to infants and children. “Very-early intervention in neonates limits the size of the reservoir and offers us the best opportunity for future interventions aimed at cure and long-term drug-free remission of HIV infection,” he said. “We don’t think the current intervention is itself curative, but it sets the stage for the capacity to offer additional innovative interventions in the future. Beyond the importance of this work for cure research per se, this very early intervention in neonates also has the potential of conferring important clinical benefits to the children who participated in this study. Finally, our study demonstrates the feasibility and importance of doing this type of research in neonates in resource-limited settings, given the appropriate infrastructure.”
EIT is supported by the National Institutes of Health. Dr. Lichterfeld disclosed having received speaking and consulting honoraria from Merck and Gilead. Dr. Kuritzkes disclosed having received consulting honoraria and/or research support from Gilead, Merck, and ViiV.
SOURCE: Garcia-Broncano P et al. Sci Transl Med. 2019 Nov 27. eaax7350.
FROM SCIENCE TRANSLATIONAL MEDICINE
Key clinical point: Antiretroviral treatment initiation immediately after birth reduced HIV-1 viral reservoir size and alters innate immune responses in neonates.
Major finding: Very-early ART intervention in neonates infected with HIV limited the number of virally infected cells and improves immune response.
Study details: A cohort study of 10 infants infected with HIV who were born in Botswana, Africa.
Disclosures: The Early Infant Treatment study is supported by the National Institutes of Health. Dr. Lichterfeld disclosed having received speaking and consulting honoraria from Merck and Gilead. Dr. Kuritzkes disclosed having received consulting honoraria and/or research support from Gilead, Merck, and ViiV.
Source: Garcia-Broncano P et al. Sci Transl Med. 2019 Nov 27. eaax7350.
ACGME deepening its commitment to physician well-being, leader says
NEW ORLEANS – When Timothy P. Brigham, MDiv, PhD, thinks about the impact of burnout and stress on the ability of physicians to practice medicine, Lewin’s equation comes to mind.
Developed by psychologist Kurt Lewin in 1936, the equation holds that behavior stems from a person’s personality and the environment that person inhabits.
Dr. Brigham, chief of staff and chief education and organizational development officer at the Chicago-based Accreditation Council for Graduate Medical Education (ACGME), said at the annual meeting of the American Academy of Pediatrics.
“It’s a toxic mine, in some ways. What we tend to do is when we detect that physicians in general are, or a particular residency program is, too stressed out or burned out, we give them resilience training. Not that that’s unimportant, but it’s like putting a canary in a toxic mine full of poison and saying, ‘We’re going to teach you to hold your breath a little bit longer.’ Our job is to detoxify the mine.”
Troubled by the rise of suicides among physicians in recent years as well as mounting evidence about the adverse impact of burnout and stress on the practice of medicine, Dr. Brigham said that the ACGME is deepening its commitment to the well-being of faculty, residents, patients, and all members of the health care team. Since launching a “call to arms” on the topic at its annual educational conference in 2015, the ACGME has added courses on well-being to its annual meeting and remolded its Clinical Learning Environmental Review program to include all clinicians, “because everybody is affected by this: nurses, coordinators, et cetera,” he said. The ACGME also has revised Common Program Requirements, disseminated tools and resources to promote well-being and new knowledge on the topic, and partnered with the National Academy of Medicine Action Collaborative on Clinician Well-Being and Resilience – all in an effort to bring about culture change.
“But we’re well aware that the ACGME can’t do this alone,” Dr. Brigham said. “We can’t ‘requirement’ our way out of this problem. It’s going to take a culture shift. Only you physicians, in collaboration with everyone in your community of learning, can create the systemic change required to improve our culture. We have a good handle on the problem at this point, but the solutions are a little bit more difficult to get a hold of. As Martin Luther King Jr. once said, ‘You don’t have to see the whole staircase, just take the first step.’ ”
The ACGME wants to work with physicians “to collect data and do joint research, to share insights, and to share tools and resources to create a better world for practicing physicians, for other members of the health care team, and for patients. After all, clinicians who care for themselves provide better care for others. They’re less likely to make errors or leave the profession,” Dr. Brigham told attendees.
He added that clinicians can gauge their risk for burnout by asking themselves three simple questions about their work environment: Does it support self-care? Does it increase and support connection with colleagues? Does it connect people to purpose and meaningful work?
“One of the problems with our resident clinical work hours is not terrible program directors saying, ‘work longer.’ It’s residents who want to take care of families for 1 more hour,” Dr. Brigham continued. “It’s residents who want to take care of patients who are going through a difficult time. You represent the top 2% in the world in terms of your intelligence and achievement, yet that’s not what makes you special. What makes you special is that the level of self-doubt in this room exceeds that of the general population by about 10 times. You also tend to run toward what everyone else runs away from: disease, despair, people who are injured and suffering. That takes a toll.”
He emphasized that positive social relationships with others are crucial to joy and well-being in the practice of medicine. “Burnout isn’t just about exhaustion; it’s about loneliness,” Dr. Brigham said. “There’s a surprising power in just asking people how they’re doing, and really wanting to know the answer.”
Negative social connections are highly correlated with burnout and depression, such as harassment, bullying, mistreatment, discrimination, “and using the power gradient to squash somebody who’s trying their best to be a physician,” he said.
Dr. Brigham acknowledged the tall task of bringing a spotlight to well-being as physicians continue to engage in tasks such as the burden and lack of standardization of prior authorization requirements, the burden of clinical documentation requirements, electronic health records and related work flow, and quality payment programs. “This is what we need to shift; this is what we need to take away so you can get back in touch with why you became a physician in the first place.”
Dr. Brigham reported having no financial disclosures.
NEW ORLEANS – When Timothy P. Brigham, MDiv, PhD, thinks about the impact of burnout and stress on the ability of physicians to practice medicine, Lewin’s equation comes to mind.
Developed by psychologist Kurt Lewin in 1936, the equation holds that behavior stems from a person’s personality and the environment that person inhabits.
Dr. Brigham, chief of staff and chief education and organizational development officer at the Chicago-based Accreditation Council for Graduate Medical Education (ACGME), said at the annual meeting of the American Academy of Pediatrics.
“It’s a toxic mine, in some ways. What we tend to do is when we detect that physicians in general are, or a particular residency program is, too stressed out or burned out, we give them resilience training. Not that that’s unimportant, but it’s like putting a canary in a toxic mine full of poison and saying, ‘We’re going to teach you to hold your breath a little bit longer.’ Our job is to detoxify the mine.”
Troubled by the rise of suicides among physicians in recent years as well as mounting evidence about the adverse impact of burnout and stress on the practice of medicine, Dr. Brigham said that the ACGME is deepening its commitment to the well-being of faculty, residents, patients, and all members of the health care team. Since launching a “call to arms” on the topic at its annual educational conference in 2015, the ACGME has added courses on well-being to its annual meeting and remolded its Clinical Learning Environmental Review program to include all clinicians, “because everybody is affected by this: nurses, coordinators, et cetera,” he said. The ACGME also has revised Common Program Requirements, disseminated tools and resources to promote well-being and new knowledge on the topic, and partnered with the National Academy of Medicine Action Collaborative on Clinician Well-Being and Resilience – all in an effort to bring about culture change.
“But we’re well aware that the ACGME can’t do this alone,” Dr. Brigham said. “We can’t ‘requirement’ our way out of this problem. It’s going to take a culture shift. Only you physicians, in collaboration with everyone in your community of learning, can create the systemic change required to improve our culture. We have a good handle on the problem at this point, but the solutions are a little bit more difficult to get a hold of. As Martin Luther King Jr. once said, ‘You don’t have to see the whole staircase, just take the first step.’ ”
The ACGME wants to work with physicians “to collect data and do joint research, to share insights, and to share tools and resources to create a better world for practicing physicians, for other members of the health care team, and for patients. After all, clinicians who care for themselves provide better care for others. They’re less likely to make errors or leave the profession,” Dr. Brigham told attendees.
He added that clinicians can gauge their risk for burnout by asking themselves three simple questions about their work environment: Does it support self-care? Does it increase and support connection with colleagues? Does it connect people to purpose and meaningful work?
“One of the problems with our resident clinical work hours is not terrible program directors saying, ‘work longer.’ It’s residents who want to take care of families for 1 more hour,” Dr. Brigham continued. “It’s residents who want to take care of patients who are going through a difficult time. You represent the top 2% in the world in terms of your intelligence and achievement, yet that’s not what makes you special. What makes you special is that the level of self-doubt in this room exceeds that of the general population by about 10 times. You also tend to run toward what everyone else runs away from: disease, despair, people who are injured and suffering. That takes a toll.”
He emphasized that positive social relationships with others are crucial to joy and well-being in the practice of medicine. “Burnout isn’t just about exhaustion; it’s about loneliness,” Dr. Brigham said. “There’s a surprising power in just asking people how they’re doing, and really wanting to know the answer.”
Negative social connections are highly correlated with burnout and depression, such as harassment, bullying, mistreatment, discrimination, “and using the power gradient to squash somebody who’s trying their best to be a physician,” he said.
Dr. Brigham acknowledged the tall task of bringing a spotlight to well-being as physicians continue to engage in tasks such as the burden and lack of standardization of prior authorization requirements, the burden of clinical documentation requirements, electronic health records and related work flow, and quality payment programs. “This is what we need to shift; this is what we need to take away so you can get back in touch with why you became a physician in the first place.”
Dr. Brigham reported having no financial disclosures.
NEW ORLEANS – When Timothy P. Brigham, MDiv, PhD, thinks about the impact of burnout and stress on the ability of physicians to practice medicine, Lewin’s equation comes to mind.
Developed by psychologist Kurt Lewin in 1936, the equation holds that behavior stems from a person’s personality and the environment that person inhabits.
Dr. Brigham, chief of staff and chief education and organizational development officer at the Chicago-based Accreditation Council for Graduate Medical Education (ACGME), said at the annual meeting of the American Academy of Pediatrics.
“It’s a toxic mine, in some ways. What we tend to do is when we detect that physicians in general are, or a particular residency program is, too stressed out or burned out, we give them resilience training. Not that that’s unimportant, but it’s like putting a canary in a toxic mine full of poison and saying, ‘We’re going to teach you to hold your breath a little bit longer.’ Our job is to detoxify the mine.”
Troubled by the rise of suicides among physicians in recent years as well as mounting evidence about the adverse impact of burnout and stress on the practice of medicine, Dr. Brigham said that the ACGME is deepening its commitment to the well-being of faculty, residents, patients, and all members of the health care team. Since launching a “call to arms” on the topic at its annual educational conference in 2015, the ACGME has added courses on well-being to its annual meeting and remolded its Clinical Learning Environmental Review program to include all clinicians, “because everybody is affected by this: nurses, coordinators, et cetera,” he said. The ACGME also has revised Common Program Requirements, disseminated tools and resources to promote well-being and new knowledge on the topic, and partnered with the National Academy of Medicine Action Collaborative on Clinician Well-Being and Resilience – all in an effort to bring about culture change.
“But we’re well aware that the ACGME can’t do this alone,” Dr. Brigham said. “We can’t ‘requirement’ our way out of this problem. It’s going to take a culture shift. Only you physicians, in collaboration with everyone in your community of learning, can create the systemic change required to improve our culture. We have a good handle on the problem at this point, but the solutions are a little bit more difficult to get a hold of. As Martin Luther King Jr. once said, ‘You don’t have to see the whole staircase, just take the first step.’ ”
The ACGME wants to work with physicians “to collect data and do joint research, to share insights, and to share tools and resources to create a better world for practicing physicians, for other members of the health care team, and for patients. After all, clinicians who care for themselves provide better care for others. They’re less likely to make errors or leave the profession,” Dr. Brigham told attendees.
He added that clinicians can gauge their risk for burnout by asking themselves three simple questions about their work environment: Does it support self-care? Does it increase and support connection with colleagues? Does it connect people to purpose and meaningful work?
“One of the problems with our resident clinical work hours is not terrible program directors saying, ‘work longer.’ It’s residents who want to take care of families for 1 more hour,” Dr. Brigham continued. “It’s residents who want to take care of patients who are going through a difficult time. You represent the top 2% in the world in terms of your intelligence and achievement, yet that’s not what makes you special. What makes you special is that the level of self-doubt in this room exceeds that of the general population by about 10 times. You also tend to run toward what everyone else runs away from: disease, despair, people who are injured and suffering. That takes a toll.”
He emphasized that positive social relationships with others are crucial to joy and well-being in the practice of medicine. “Burnout isn’t just about exhaustion; it’s about loneliness,” Dr. Brigham said. “There’s a surprising power in just asking people how they’re doing, and really wanting to know the answer.”
Negative social connections are highly correlated with burnout and depression, such as harassment, bullying, mistreatment, discrimination, “and using the power gradient to squash somebody who’s trying their best to be a physician,” he said.
Dr. Brigham acknowledged the tall task of bringing a spotlight to well-being as physicians continue to engage in tasks such as the burden and lack of standardization of prior authorization requirements, the burden of clinical documentation requirements, electronic health records and related work flow, and quality payment programs. “This is what we need to shift; this is what we need to take away so you can get back in touch with why you became a physician in the first place.”
Dr. Brigham reported having no financial disclosures.
EXPERT ANALYSIS AT AAP 2019
Late-onset MS is often more severe than earlier-onset MS
ST. LOUIS –
Whether the pathophysiology is different, or if the disease is simply compounded by the effects of aging, is uncertain. It is likely, however, that patients with late onset are at greater risk of misdiagnosis and delayed treatment, said Mattia Wruble, a 4th-year medical student at the University of Virginia.
The reason is that MS usually presents in young women as relapsing-remitting disease, but late onset is more typically primary progressive, with a higher proportion of men, which is something that has been demonstrated in previous work and also found in the new study.
“What we didn’t expect to find,” however, was greater “disability and higher disability accrual rates” across all subtypes. Late-onset MS “is a worse disease, a more severe disease” that might even need different treatment options, Ms. Wruble said at the American Neurological Association annual meeting.
Ms. Wruble and colleagues compared the charts of 1,381 patients with MS onset before age 50 years, at a median age of 33 years, with 143 patients whose symptoms began at age 50 years or later, at a median of 55 years.
It was one of the largest late-onset MS samples to date. Incidence of the condition is on the rise, but research has been limited. The investigators hoped to address a need for “a better and more thorough characterization of” the disease to aid treatment and improve outcomes, Ms. Wruble said.
There was a higher proportion of men in the late-onset group, 38% vs. 26%. About 75% of patients with early onset had relapsing remitting disease, and 10% had primary progressive disease. The late-onset group was about evenly split between relapsing-remitting and primary progressive MS (P less than .001).
Patients with late onset MS also had a higher degree of disability at their most recent visit across all subtypes (median Expanded Disability Status Scale score of 5 versus 3), despite having a shorter duration of disease (mean 12 vs. 17 years from symptom onset).
Overall disease accrual rate in the late-onset group was twice that of early-onset group, a decline of 0.647 points per year on the EDSS versus 0.357 points (P less than .001). The finding held when limited to early and late relapsing-remitting cases. The accrual rate for late-onset primary and secondary progressive MS was 1.5 times that of early-onset cases.
Transition to secondary progressive disease also was faster in the late-onset group, a mean of about 7 years versus 15-19 years. Patients with late onset also were more likely to have a history of smoking.
The cerebrospinal fluid in late-onset MS generally has fewer unique oligoclonal bands, which are part of the McDonald diagnostic criteria for MS. “The McDonald criteria were made for patients between 20 and 50 years old; maybe they are not the best criteria to diagnose this late-onset group,” Ms. Wruble said.
There was no external funding, and Ms. Wruble had no disclosures.
SOURCE: Wruble M et al. ANA 2019. Abstract S234.
ST. LOUIS –
Whether the pathophysiology is different, or if the disease is simply compounded by the effects of aging, is uncertain. It is likely, however, that patients with late onset are at greater risk of misdiagnosis and delayed treatment, said Mattia Wruble, a 4th-year medical student at the University of Virginia.
The reason is that MS usually presents in young women as relapsing-remitting disease, but late onset is more typically primary progressive, with a higher proportion of men, which is something that has been demonstrated in previous work and also found in the new study.
“What we didn’t expect to find,” however, was greater “disability and higher disability accrual rates” across all subtypes. Late-onset MS “is a worse disease, a more severe disease” that might even need different treatment options, Ms. Wruble said at the American Neurological Association annual meeting.
Ms. Wruble and colleagues compared the charts of 1,381 patients with MS onset before age 50 years, at a median age of 33 years, with 143 patients whose symptoms began at age 50 years or later, at a median of 55 years.
It was one of the largest late-onset MS samples to date. Incidence of the condition is on the rise, but research has been limited. The investigators hoped to address a need for “a better and more thorough characterization of” the disease to aid treatment and improve outcomes, Ms. Wruble said.
There was a higher proportion of men in the late-onset group, 38% vs. 26%. About 75% of patients with early onset had relapsing remitting disease, and 10% had primary progressive disease. The late-onset group was about evenly split between relapsing-remitting and primary progressive MS (P less than .001).
Patients with late onset MS also had a higher degree of disability at their most recent visit across all subtypes (median Expanded Disability Status Scale score of 5 versus 3), despite having a shorter duration of disease (mean 12 vs. 17 years from symptom onset).
Overall disease accrual rate in the late-onset group was twice that of early-onset group, a decline of 0.647 points per year on the EDSS versus 0.357 points (P less than .001). The finding held when limited to early and late relapsing-remitting cases. The accrual rate for late-onset primary and secondary progressive MS was 1.5 times that of early-onset cases.
Transition to secondary progressive disease also was faster in the late-onset group, a mean of about 7 years versus 15-19 years. Patients with late onset also were more likely to have a history of smoking.
The cerebrospinal fluid in late-onset MS generally has fewer unique oligoclonal bands, which are part of the McDonald diagnostic criteria for MS. “The McDonald criteria were made for patients between 20 and 50 years old; maybe they are not the best criteria to diagnose this late-onset group,” Ms. Wruble said.
There was no external funding, and Ms. Wruble had no disclosures.
SOURCE: Wruble M et al. ANA 2019. Abstract S234.
ST. LOUIS –
Whether the pathophysiology is different, or if the disease is simply compounded by the effects of aging, is uncertain. It is likely, however, that patients with late onset are at greater risk of misdiagnosis and delayed treatment, said Mattia Wruble, a 4th-year medical student at the University of Virginia.
The reason is that MS usually presents in young women as relapsing-remitting disease, but late onset is more typically primary progressive, with a higher proportion of men, which is something that has been demonstrated in previous work and also found in the new study.
“What we didn’t expect to find,” however, was greater “disability and higher disability accrual rates” across all subtypes. Late-onset MS “is a worse disease, a more severe disease” that might even need different treatment options, Ms. Wruble said at the American Neurological Association annual meeting.
Ms. Wruble and colleagues compared the charts of 1,381 patients with MS onset before age 50 years, at a median age of 33 years, with 143 patients whose symptoms began at age 50 years or later, at a median of 55 years.
It was one of the largest late-onset MS samples to date. Incidence of the condition is on the rise, but research has been limited. The investigators hoped to address a need for “a better and more thorough characterization of” the disease to aid treatment and improve outcomes, Ms. Wruble said.
There was a higher proportion of men in the late-onset group, 38% vs. 26%. About 75% of patients with early onset had relapsing remitting disease, and 10% had primary progressive disease. The late-onset group was about evenly split between relapsing-remitting and primary progressive MS (P less than .001).
Patients with late onset MS also had a higher degree of disability at their most recent visit across all subtypes (median Expanded Disability Status Scale score of 5 versus 3), despite having a shorter duration of disease (mean 12 vs. 17 years from symptom onset).
Overall disease accrual rate in the late-onset group was twice that of early-onset group, a decline of 0.647 points per year on the EDSS versus 0.357 points (P less than .001). The finding held when limited to early and late relapsing-remitting cases. The accrual rate for late-onset primary and secondary progressive MS was 1.5 times that of early-onset cases.
Transition to secondary progressive disease also was faster in the late-onset group, a mean of about 7 years versus 15-19 years. Patients with late onset also were more likely to have a history of smoking.
The cerebrospinal fluid in late-onset MS generally has fewer unique oligoclonal bands, which are part of the McDonald diagnostic criteria for MS. “The McDonald criteria were made for patients between 20 and 50 years old; maybe they are not the best criteria to diagnose this late-onset group,” Ms. Wruble said.
There was no external funding, and Ms. Wruble had no disclosures.
SOURCE: Wruble M et al. ANA 2019. Abstract S234.
REPORTING FROM ANA 2019
ASCO releases revised version of its Patient-Centered Oncology Payment model
The American Society of Clinical Oncology has made some adjustments to its Patient-Centered Oncology Payment (PCOP) advanced alternative payment model and will now be looking to gain approval from the Physician-Focused Payment Model Technical Advisory Committee (PTAC).
PTAC reviews physician-developed advanced alternative payment models and sends those APMs that are approved to the Centers for Medicare & Medicaid Services’ Center for Medicare & Medicaid Innovation (CMMI) to determine if there will be further testing and possible implementation for use by physicians as part of the APM track of the Quality Payment Program.
Even if approved by PTAC, the ASCO model faces an uphill battle. CMS already has its own Oncology Care Model (OCM), and given the agency’s track record of not testing APMs that have been approved by PTAC, the deck may be stacked against ASCO in terms of getting its model into Medicare.
But officials at ASCO are hoping a PTAC vetting and approval will open the door to its implementation by commercial payers.
“We are now ready to take this to PTAC,” Jeffery Ward, MD, past chair of ASCO’s government relations committee, said in an interview. “I don’t expect any trouble getting through PTAC. The other question is whether CMMI would decide to actually test a model like this, and so far, they have seemed pretty much a one-trick pony, as in they’ve got their Oncology Care Model and they are going to run with it.”
However, Dr. Ward, a medical oncologist at Swedish Cancer Institute of Edmonds, Wash., said that PCOP has been designed with the commercial market in mind and the PTAC recommendation will help validate the model and make it more appealing to commercial payers.
A key feature of the adjusted payment model is that it has taken a lesson learned from CMS’s Oncology Care Model in terms of how to account for the price of drugs. Including the price of drugs in a value-based payment model – the approach taken by the OCM – creates too much variability, Dr. Ward noted. He used lung cancer as an example, noting that the type of lung cancer could have more of an impact on physician spending than any specific treatment decisions a physician can make because the treatment choices may not be there.
“When that happens, whether you are successful or not depends on the luck of the draw instead of the choices you make,” he said.
In an effort not to penalize practices because of their patient mix, PCOP takes the cost of drugs out of the value mix.
“What this model does is it doesn’t hold practices responsible for the cost of the drugs. It holds them responsible for how they utilize the drugs,” he said. “It really begins to make me responsible for making value-based decisions as opposed to just getting lucky and having a cheaper panel of patients.”
Overall, Dr. Ward described the PCOP model as accomplishing the goals of CMS’s APMs by encouraging physicians to treat patients through high-value treatments.
“This model does that by including quality measures that play a role in how much you get paid,” he said. “There are pathways that are value-based pathways and your ability to follow those pathways plays a role in how much you get paid. The amount of care that you send to the ER or the hospital plays a role in how much you get paid. Those things are then used in adjusting what is our fee-for-service model of medicine. So you are still getting paid a base rate based on the care that you give on a fee-for-service model, but that gets adjusted up or down depending on those other characteristics.”
But for the model to have the biggest impact, it needs to be adopted by all payers in a given state or region and used by all physicians, who by the design of the payment model, will have a role in shaping the implementation details.
Savings could come from lower administrative costs at the physician office because there would be no need for separate contracts between payers, something that could generate savings of up to 8%, compared with the current system, Dr. Ward noted.
“This is designed to be a multipayer and multipractice initiative,” he said. “In its grandest form, a state or states would implement this and it would be participated in by all the oncology practices and all of the payers.”
The American Society of Clinical Oncology has made some adjustments to its Patient-Centered Oncology Payment (PCOP) advanced alternative payment model and will now be looking to gain approval from the Physician-Focused Payment Model Technical Advisory Committee (PTAC).
PTAC reviews physician-developed advanced alternative payment models and sends those APMs that are approved to the Centers for Medicare & Medicaid Services’ Center for Medicare & Medicaid Innovation (CMMI) to determine if there will be further testing and possible implementation for use by physicians as part of the APM track of the Quality Payment Program.
Even if approved by PTAC, the ASCO model faces an uphill battle. CMS already has its own Oncology Care Model (OCM), and given the agency’s track record of not testing APMs that have been approved by PTAC, the deck may be stacked against ASCO in terms of getting its model into Medicare.
But officials at ASCO are hoping a PTAC vetting and approval will open the door to its implementation by commercial payers.
“We are now ready to take this to PTAC,” Jeffery Ward, MD, past chair of ASCO’s government relations committee, said in an interview. “I don’t expect any trouble getting through PTAC. The other question is whether CMMI would decide to actually test a model like this, and so far, they have seemed pretty much a one-trick pony, as in they’ve got their Oncology Care Model and they are going to run with it.”
However, Dr. Ward, a medical oncologist at Swedish Cancer Institute of Edmonds, Wash., said that PCOP has been designed with the commercial market in mind and the PTAC recommendation will help validate the model and make it more appealing to commercial payers.
A key feature of the adjusted payment model is that it has taken a lesson learned from CMS’s Oncology Care Model in terms of how to account for the price of drugs. Including the price of drugs in a value-based payment model – the approach taken by the OCM – creates too much variability, Dr. Ward noted. He used lung cancer as an example, noting that the type of lung cancer could have more of an impact on physician spending than any specific treatment decisions a physician can make because the treatment choices may not be there.
“When that happens, whether you are successful or not depends on the luck of the draw instead of the choices you make,” he said.
In an effort not to penalize practices because of their patient mix, PCOP takes the cost of drugs out of the value mix.
“What this model does is it doesn’t hold practices responsible for the cost of the drugs. It holds them responsible for how they utilize the drugs,” he said. “It really begins to make me responsible for making value-based decisions as opposed to just getting lucky and having a cheaper panel of patients.”
Overall, Dr. Ward described the PCOP model as accomplishing the goals of CMS’s APMs by encouraging physicians to treat patients through high-value treatments.
“This model does that by including quality measures that play a role in how much you get paid,” he said. “There are pathways that are value-based pathways and your ability to follow those pathways plays a role in how much you get paid. The amount of care that you send to the ER or the hospital plays a role in how much you get paid. Those things are then used in adjusting what is our fee-for-service model of medicine. So you are still getting paid a base rate based on the care that you give on a fee-for-service model, but that gets adjusted up or down depending on those other characteristics.”
But for the model to have the biggest impact, it needs to be adopted by all payers in a given state or region and used by all physicians, who by the design of the payment model, will have a role in shaping the implementation details.
Savings could come from lower administrative costs at the physician office because there would be no need for separate contracts between payers, something that could generate savings of up to 8%, compared with the current system, Dr. Ward noted.
“This is designed to be a multipayer and multipractice initiative,” he said. “In its grandest form, a state or states would implement this and it would be participated in by all the oncology practices and all of the payers.”
The American Society of Clinical Oncology has made some adjustments to its Patient-Centered Oncology Payment (PCOP) advanced alternative payment model and will now be looking to gain approval from the Physician-Focused Payment Model Technical Advisory Committee (PTAC).
PTAC reviews physician-developed advanced alternative payment models and sends those APMs that are approved to the Centers for Medicare & Medicaid Services’ Center for Medicare & Medicaid Innovation (CMMI) to determine if there will be further testing and possible implementation for use by physicians as part of the APM track of the Quality Payment Program.
Even if approved by PTAC, the ASCO model faces an uphill battle. CMS already has its own Oncology Care Model (OCM), and given the agency’s track record of not testing APMs that have been approved by PTAC, the deck may be stacked against ASCO in terms of getting its model into Medicare.
But officials at ASCO are hoping a PTAC vetting and approval will open the door to its implementation by commercial payers.
“We are now ready to take this to PTAC,” Jeffery Ward, MD, past chair of ASCO’s government relations committee, said in an interview. “I don’t expect any trouble getting through PTAC. The other question is whether CMMI would decide to actually test a model like this, and so far, they have seemed pretty much a one-trick pony, as in they’ve got their Oncology Care Model and they are going to run with it.”
However, Dr. Ward, a medical oncologist at Swedish Cancer Institute of Edmonds, Wash., said that PCOP has been designed with the commercial market in mind and the PTAC recommendation will help validate the model and make it more appealing to commercial payers.
A key feature of the adjusted payment model is that it has taken a lesson learned from CMS’s Oncology Care Model in terms of how to account for the price of drugs. Including the price of drugs in a value-based payment model – the approach taken by the OCM – creates too much variability, Dr. Ward noted. He used lung cancer as an example, noting that the type of lung cancer could have more of an impact on physician spending than any specific treatment decisions a physician can make because the treatment choices may not be there.
“When that happens, whether you are successful or not depends on the luck of the draw instead of the choices you make,” he said.
In an effort not to penalize practices because of their patient mix, PCOP takes the cost of drugs out of the value mix.
“What this model does is it doesn’t hold practices responsible for the cost of the drugs. It holds them responsible for how they utilize the drugs,” he said. “It really begins to make me responsible for making value-based decisions as opposed to just getting lucky and having a cheaper panel of patients.”
Overall, Dr. Ward described the PCOP model as accomplishing the goals of CMS’s APMs by encouraging physicians to treat patients through high-value treatments.
“This model does that by including quality measures that play a role in how much you get paid,” he said. “There are pathways that are value-based pathways and your ability to follow those pathways plays a role in how much you get paid. The amount of care that you send to the ER or the hospital plays a role in how much you get paid. Those things are then used in adjusting what is our fee-for-service model of medicine. So you are still getting paid a base rate based on the care that you give on a fee-for-service model, but that gets adjusted up or down depending on those other characteristics.”
But for the model to have the biggest impact, it needs to be adopted by all payers in a given state or region and used by all physicians, who by the design of the payment model, will have a role in shaping the implementation details.
Savings could come from lower administrative costs at the physician office because there would be no need for separate contracts between payers, something that could generate savings of up to 8%, compared with the current system, Dr. Ward noted.
“This is designed to be a multipayer and multipractice initiative,” he said. “In its grandest form, a state or states would implement this and it would be participated in by all the oncology practices and all of the payers.”