User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Obesity dropping in kids aged 2-4 years in WIC program
During 2010-2016, the prevalence of obesity among children in the Special Supplemental Nutrition Program for Women, Infants, and Children (WIC) program significantly decreased in 73% of 56 states and territories, Liping Pan, MD, and colleagues reported in the Morbidity and Mortality Weekly Report.
The obesity prevalence decreases varied, but exceeded 3% in Guam, New Jersey, New Mexico, Northern Mariana Islands, Puerto Rico, Utah, and Virginia. Puerto Rico experienced the greatest benefit, with an 8% decrease in obesity among children aged 2-4 years enrolled in the WIC program, wrote Dr. Pan, an epidemiologist at the Centers for Disease Control and Prevention and coauthors.
Although the changes were small, the positive trend gains more import when viewed in light of the country’s long-term obesity trends, Dr. Pan and his team wrote. After a short-lived dip during 2007-2012, obesity has been on the rise among these children, jumping from 8% in 2012 to 14% in 2016. “Thus, even these small decreases indicate progress for this vulnerable WIC population,” the team said.
WIC extends nutritional assistance to families whose income is 185% or less of the federal poverty guideline or are eligible for other programs, as well as being deemed at nutritional risk.
The current study looked at obesity trends during 2010-2016 among 12,403,629 WIC recipients aged 2-4 years in all 50 U.S. states and five territories.
In 2010, obesity prevalence ranged from a low of 10% in Colorado to a high of 22% in Virginia. In Alaska, Puerto Rico, and Virginia, it was 20% or higher. Only in Colorado and Hawaii was obesity prevalence 10% or less among these children.
By 2016, obesity prevalence among children aged 2-4 years ranged from 8% in the Northern Mariana Islands to 19.8% in Alaska. It was less than 20% in all the states and territories, and less than 10% in Colorado, Guam, Hawaii, Northern Mariana Islands, Utah, and Wyoming.
It increased during 2010-2016, however, in Alabama (0.5%), North Carolina (0.6%), and West Virginia (2.2%).
The changes reflect the 2009 program revisions made to adhere to the 2005 Dietary Guidelines for Americans and the infant food and feeding practice guidelines of the American Academy of Pediatrics, Dr. Pan and associates wrote.
“The revised food packages include a broader range of healthy food options; promote fruit, vegetable, and whole wheat product purchases; support breastfeeding; and give WIC state and territory agencies more flexibility to accommodate cultural food preferences,” the authors noted.
In response to the changes, Dr. Pan and associates noted, authorized WIC stores began carrying healthier offerings. Tracking showed that children in the program consumed more fruits, vegetables, and whole grain products and less juice, white bread, and whole milk after the revisions than they did previously.
Despite the good news, childhood obesity rates still are too high and much remains to be done, they noted.
“Multiple approaches are needed to address and eliminate childhood obesity. The National Academy of Medicine and other groups have recommended a comprehensive and integrated approach that calls for positive changes in physical activity and food and beverage environments in multiple settings including home, early care and education [such as nutrition standards for food served], and community [such as neighborhood designs that encourage walking and biking] to promote healthy eating and physical activity for young children. Further implementation of these positive changes across the United States could further the decreases in child-hood obesity,” Dr. Pan and coauthors concluded.
Dr. Pan and coauthors had no financial disclosures.
SOURCE: Pan L et al. MMWR Morb Mortal Wkly Rep. 2019 Nov 22;68(46):1057-61.
During 2010-2016, the prevalence of obesity among children in the Special Supplemental Nutrition Program for Women, Infants, and Children (WIC) program significantly decreased in 73% of 56 states and territories, Liping Pan, MD, and colleagues reported in the Morbidity and Mortality Weekly Report.
The obesity prevalence decreases varied, but exceeded 3% in Guam, New Jersey, New Mexico, Northern Mariana Islands, Puerto Rico, Utah, and Virginia. Puerto Rico experienced the greatest benefit, with an 8% decrease in obesity among children aged 2-4 years enrolled in the WIC program, wrote Dr. Pan, an epidemiologist at the Centers for Disease Control and Prevention and coauthors.
Although the changes were small, the positive trend gains more import when viewed in light of the country’s long-term obesity trends, Dr. Pan and his team wrote. After a short-lived dip during 2007-2012, obesity has been on the rise among these children, jumping from 8% in 2012 to 14% in 2016. “Thus, even these small decreases indicate progress for this vulnerable WIC population,” the team said.
WIC extends nutritional assistance to families whose income is 185% or less of the federal poverty guideline or are eligible for other programs, as well as being deemed at nutritional risk.
The current study looked at obesity trends during 2010-2016 among 12,403,629 WIC recipients aged 2-4 years in all 50 U.S. states and five territories.
In 2010, obesity prevalence ranged from a low of 10% in Colorado to a high of 22% in Virginia. In Alaska, Puerto Rico, and Virginia, it was 20% or higher. Only in Colorado and Hawaii was obesity prevalence 10% or less among these children.
By 2016, obesity prevalence among children aged 2-4 years ranged from 8% in the Northern Mariana Islands to 19.8% in Alaska. It was less than 20% in all the states and territories, and less than 10% in Colorado, Guam, Hawaii, Northern Mariana Islands, Utah, and Wyoming.
It increased during 2010-2016, however, in Alabama (0.5%), North Carolina (0.6%), and West Virginia (2.2%).
The changes reflect the 2009 program revisions made to adhere to the 2005 Dietary Guidelines for Americans and the infant food and feeding practice guidelines of the American Academy of Pediatrics, Dr. Pan and associates wrote.
“The revised food packages include a broader range of healthy food options; promote fruit, vegetable, and whole wheat product purchases; support breastfeeding; and give WIC state and territory agencies more flexibility to accommodate cultural food preferences,” the authors noted.
In response to the changes, Dr. Pan and associates noted, authorized WIC stores began carrying healthier offerings. Tracking showed that children in the program consumed more fruits, vegetables, and whole grain products and less juice, white bread, and whole milk after the revisions than they did previously.
Despite the good news, childhood obesity rates still are too high and much remains to be done, they noted.
“Multiple approaches are needed to address and eliminate childhood obesity. The National Academy of Medicine and other groups have recommended a comprehensive and integrated approach that calls for positive changes in physical activity and food and beverage environments in multiple settings including home, early care and education [such as nutrition standards for food served], and community [such as neighborhood designs that encourage walking and biking] to promote healthy eating and physical activity for young children. Further implementation of these positive changes across the United States could further the decreases in child-hood obesity,” Dr. Pan and coauthors concluded.
Dr. Pan and coauthors had no financial disclosures.
SOURCE: Pan L et al. MMWR Morb Mortal Wkly Rep. 2019 Nov 22;68(46):1057-61.
During 2010-2016, the prevalence of obesity among children in the Special Supplemental Nutrition Program for Women, Infants, and Children (WIC) program significantly decreased in 73% of 56 states and territories, Liping Pan, MD, and colleagues reported in the Morbidity and Mortality Weekly Report.
The obesity prevalence decreases varied, but exceeded 3% in Guam, New Jersey, New Mexico, Northern Mariana Islands, Puerto Rico, Utah, and Virginia. Puerto Rico experienced the greatest benefit, with an 8% decrease in obesity among children aged 2-4 years enrolled in the WIC program, wrote Dr. Pan, an epidemiologist at the Centers for Disease Control and Prevention and coauthors.
Although the changes were small, the positive trend gains more import when viewed in light of the country’s long-term obesity trends, Dr. Pan and his team wrote. After a short-lived dip during 2007-2012, obesity has been on the rise among these children, jumping from 8% in 2012 to 14% in 2016. “Thus, even these small decreases indicate progress for this vulnerable WIC population,” the team said.
WIC extends nutritional assistance to families whose income is 185% or less of the federal poverty guideline or are eligible for other programs, as well as being deemed at nutritional risk.
The current study looked at obesity trends during 2010-2016 among 12,403,629 WIC recipients aged 2-4 years in all 50 U.S. states and five territories.
In 2010, obesity prevalence ranged from a low of 10% in Colorado to a high of 22% in Virginia. In Alaska, Puerto Rico, and Virginia, it was 20% or higher. Only in Colorado and Hawaii was obesity prevalence 10% or less among these children.
By 2016, obesity prevalence among children aged 2-4 years ranged from 8% in the Northern Mariana Islands to 19.8% in Alaska. It was less than 20% in all the states and territories, and less than 10% in Colorado, Guam, Hawaii, Northern Mariana Islands, Utah, and Wyoming.
It increased during 2010-2016, however, in Alabama (0.5%), North Carolina (0.6%), and West Virginia (2.2%).
The changes reflect the 2009 program revisions made to adhere to the 2005 Dietary Guidelines for Americans and the infant food and feeding practice guidelines of the American Academy of Pediatrics, Dr. Pan and associates wrote.
“The revised food packages include a broader range of healthy food options; promote fruit, vegetable, and whole wheat product purchases; support breastfeeding; and give WIC state and territory agencies more flexibility to accommodate cultural food preferences,” the authors noted.
In response to the changes, Dr. Pan and associates noted, authorized WIC stores began carrying healthier offerings. Tracking showed that children in the program consumed more fruits, vegetables, and whole grain products and less juice, white bread, and whole milk after the revisions than they did previously.
Despite the good news, childhood obesity rates still are too high and much remains to be done, they noted.
“Multiple approaches are needed to address and eliminate childhood obesity. The National Academy of Medicine and other groups have recommended a comprehensive and integrated approach that calls for positive changes in physical activity and food and beverage environments in multiple settings including home, early care and education [such as nutrition standards for food served], and community [such as neighborhood designs that encourage walking and biking] to promote healthy eating and physical activity for young children. Further implementation of these positive changes across the United States could further the decreases in child-hood obesity,” Dr. Pan and coauthors concluded.
Dr. Pan and coauthors had no financial disclosures.
SOURCE: Pan L et al. MMWR Morb Mortal Wkly Rep. 2019 Nov 22;68(46):1057-61.
FROM THE MORBIDITY AND MORTALITY WEEKLY REPORT
ASH preview: Key themes include tackling CAR T obstacles, sickle cell advances, VTE
Chimeric antigen receptor (CAR) T-cell therapies have garnered a great deal of attention given their “incredible efficacy” in treating B-cell malignancies, and new findings are taking aim at the drawbacks of therapy, such as the time, expense, and toxicity involved, according to Robert A. Brodsky, MD.
One example, from a study slated for presentation during a plenary session at the upcoming annual meeting of the American Society of Hematology involves the investigational T-cell bispecific antibody mosunetuzumab, which targets both CD20 on the surface of malignant B cells, and CD3 on cytotoxic T cells, engaging the T cells and directing their cytotoxicity against B cells.
In a study (Abstract 6) of 218 non-Hodgkin lymphoma patients, including 23 who had already received CAR T-cell therapy and had relapsed or were refractory to the treatment, 64% responded, 42% had a complete response, and the median duration of response is now out to 9 months, Dr. Brodsky, ASH secretary and director of the division of hematology at Johns Hopkins University, Baltimore, said during a premeeting press conference.
“It’s basically an antibody using the patient’s own T cell to do what a CAR-T cell would do – [a] very exciting study and large study,” he said. “It is an off-the-shelf product, it completely gets around the problem of the time to generate the CAR T-cell product, and because it’s going to be much simpler and faster to produce, it’s likely going to be much cheaper than CAR T cells.”
The preliminary results also suggest it is less toxic than CAR T-cell therapy, he added.
Two other CAR T-cell therapy–related studies highlighted during the press conference address its use for multiple myeloma. One, the phase 1b/2 CARTITUDE study (Abstract 577) uses CAR T cells against the B-cell maturation antigen (BCMA) in the relapsed/refractory setting.
Of 25 patients treated with chemotherapy followed by CAR T-cell infusion and followed for a median of 3 months, 91% responded, two achieved a complete remission, and “many other responses were very deep responses,” Dr. Brodsky said, noting that the second featured multiple myeloma trial (Abstract 930) looked at bispecific CAR T-cell therapy targeting BCMA and CD38 in an effort to reduce resistance to the therapy.
“Again, very interesting preliminary results,” he said, noting that of 16 patients followed for a median of 36 weeks, 87.5% responded, the treatment was well tolerated, and progression-free survival at 9 months was 75%.
In addition to the “key theme” of overcoming CAR T-cell therapy obstacles, three other themes have emerged from among the thousands of abstracts submitted for presentation at ASH. These, as presented during the press conference, include new venous thromboembolism (VTE) therapies and approaches to research; inclusive medicine, with abstracts focused on age- and race-related issues in clinical trials; and new advances in the treatment of sickle cell disease. All of these have potentially practice-changing implications, as do the six late-breaking abstracts selected from 93 abstracts submitted for consideration for oral presentation at ASH, Dr. Brodsky said.
One of the “truly practice-changing” late-breakers is a randomized phase 3 trial (Abstract LBA-1) comparing the bispecific antibody blinatumomab to chemotherapy for post-re-induction therapy in high- and intermediate-risk acute lymphoblastic leukemia (ALL) at first relapse in children, adolescents and young adults.
The study demonstrated the superiority of blinatumomab for efficacy and tolerability, which is particularly encouraging given the challenges in getting relapsed ALL patients back into remission so they can undergo bone marrow transplant, Dr. Brodsky said.
Of 208 patients randomized, 73% vs. 45% in the blinatumomab vs. chemotherapy arms were able to get to transplant – and therefore to potential cure, he said.
“Of note, the blinatumomab arm was less toxic and there was marked improvement in disease-free and overall survival, so this is clearly going to become a new standard of care for relapsed and refractory ALL,” he added.
Chimeric antigen receptor (CAR) T-cell therapies have garnered a great deal of attention given their “incredible efficacy” in treating B-cell malignancies, and new findings are taking aim at the drawbacks of therapy, such as the time, expense, and toxicity involved, according to Robert A. Brodsky, MD.
One example, from a study slated for presentation during a plenary session at the upcoming annual meeting of the American Society of Hematology involves the investigational T-cell bispecific antibody mosunetuzumab, which targets both CD20 on the surface of malignant B cells, and CD3 on cytotoxic T cells, engaging the T cells and directing their cytotoxicity against B cells.
In a study (Abstract 6) of 218 non-Hodgkin lymphoma patients, including 23 who had already received CAR T-cell therapy and had relapsed or were refractory to the treatment, 64% responded, 42% had a complete response, and the median duration of response is now out to 9 months, Dr. Brodsky, ASH secretary and director of the division of hematology at Johns Hopkins University, Baltimore, said during a premeeting press conference.
“It’s basically an antibody using the patient’s own T cell to do what a CAR-T cell would do – [a] very exciting study and large study,” he said. “It is an off-the-shelf product, it completely gets around the problem of the time to generate the CAR T-cell product, and because it’s going to be much simpler and faster to produce, it’s likely going to be much cheaper than CAR T cells.”
The preliminary results also suggest it is less toxic than CAR T-cell therapy, he added.
Two other CAR T-cell therapy–related studies highlighted during the press conference address its use for multiple myeloma. One, the phase 1b/2 CARTITUDE study (Abstract 577) uses CAR T cells against the B-cell maturation antigen (BCMA) in the relapsed/refractory setting.
Of 25 patients treated with chemotherapy followed by CAR T-cell infusion and followed for a median of 3 months, 91% responded, two achieved a complete remission, and “many other responses were very deep responses,” Dr. Brodsky said, noting that the second featured multiple myeloma trial (Abstract 930) looked at bispecific CAR T-cell therapy targeting BCMA and CD38 in an effort to reduce resistance to the therapy.
“Again, very interesting preliminary results,” he said, noting that of 16 patients followed for a median of 36 weeks, 87.5% responded, the treatment was well tolerated, and progression-free survival at 9 months was 75%.
In addition to the “key theme” of overcoming CAR T-cell therapy obstacles, three other themes have emerged from among the thousands of abstracts submitted for presentation at ASH. These, as presented during the press conference, include new venous thromboembolism (VTE) therapies and approaches to research; inclusive medicine, with abstracts focused on age- and race-related issues in clinical trials; and new advances in the treatment of sickle cell disease. All of these have potentially practice-changing implications, as do the six late-breaking abstracts selected from 93 abstracts submitted for consideration for oral presentation at ASH, Dr. Brodsky said.
One of the “truly practice-changing” late-breakers is a randomized phase 3 trial (Abstract LBA-1) comparing the bispecific antibody blinatumomab to chemotherapy for post-re-induction therapy in high- and intermediate-risk acute lymphoblastic leukemia (ALL) at first relapse in children, adolescents and young adults.
The study demonstrated the superiority of blinatumomab for efficacy and tolerability, which is particularly encouraging given the challenges in getting relapsed ALL patients back into remission so they can undergo bone marrow transplant, Dr. Brodsky said.
Of 208 patients randomized, 73% vs. 45% in the blinatumomab vs. chemotherapy arms were able to get to transplant – and therefore to potential cure, he said.
“Of note, the blinatumomab arm was less toxic and there was marked improvement in disease-free and overall survival, so this is clearly going to become a new standard of care for relapsed and refractory ALL,” he added.
Chimeric antigen receptor (CAR) T-cell therapies have garnered a great deal of attention given their “incredible efficacy” in treating B-cell malignancies, and new findings are taking aim at the drawbacks of therapy, such as the time, expense, and toxicity involved, according to Robert A. Brodsky, MD.
One example, from a study slated for presentation during a plenary session at the upcoming annual meeting of the American Society of Hematology involves the investigational T-cell bispecific antibody mosunetuzumab, which targets both CD20 on the surface of malignant B cells, and CD3 on cytotoxic T cells, engaging the T cells and directing their cytotoxicity against B cells.
In a study (Abstract 6) of 218 non-Hodgkin lymphoma patients, including 23 who had already received CAR T-cell therapy and had relapsed or were refractory to the treatment, 64% responded, 42% had a complete response, and the median duration of response is now out to 9 months, Dr. Brodsky, ASH secretary and director of the division of hematology at Johns Hopkins University, Baltimore, said during a premeeting press conference.
“It’s basically an antibody using the patient’s own T cell to do what a CAR-T cell would do – [a] very exciting study and large study,” he said. “It is an off-the-shelf product, it completely gets around the problem of the time to generate the CAR T-cell product, and because it’s going to be much simpler and faster to produce, it’s likely going to be much cheaper than CAR T cells.”
The preliminary results also suggest it is less toxic than CAR T-cell therapy, he added.
Two other CAR T-cell therapy–related studies highlighted during the press conference address its use for multiple myeloma. One, the phase 1b/2 CARTITUDE study (Abstract 577) uses CAR T cells against the B-cell maturation antigen (BCMA) in the relapsed/refractory setting.
Of 25 patients treated with chemotherapy followed by CAR T-cell infusion and followed for a median of 3 months, 91% responded, two achieved a complete remission, and “many other responses were very deep responses,” Dr. Brodsky said, noting that the second featured multiple myeloma trial (Abstract 930) looked at bispecific CAR T-cell therapy targeting BCMA and CD38 in an effort to reduce resistance to the therapy.
“Again, very interesting preliminary results,” he said, noting that of 16 patients followed for a median of 36 weeks, 87.5% responded, the treatment was well tolerated, and progression-free survival at 9 months was 75%.
In addition to the “key theme” of overcoming CAR T-cell therapy obstacles, three other themes have emerged from among the thousands of abstracts submitted for presentation at ASH. These, as presented during the press conference, include new venous thromboembolism (VTE) therapies and approaches to research; inclusive medicine, with abstracts focused on age- and race-related issues in clinical trials; and new advances in the treatment of sickle cell disease. All of these have potentially practice-changing implications, as do the six late-breaking abstracts selected from 93 abstracts submitted for consideration for oral presentation at ASH, Dr. Brodsky said.
One of the “truly practice-changing” late-breakers is a randomized phase 3 trial (Abstract LBA-1) comparing the bispecific antibody blinatumomab to chemotherapy for post-re-induction therapy in high- and intermediate-risk acute lymphoblastic leukemia (ALL) at first relapse in children, adolescents and young adults.
The study demonstrated the superiority of blinatumomab for efficacy and tolerability, which is particularly encouraging given the challenges in getting relapsed ALL patients back into remission so they can undergo bone marrow transplant, Dr. Brodsky said.
Of 208 patients randomized, 73% vs. 45% in the blinatumomab vs. chemotherapy arms were able to get to transplant – and therefore to potential cure, he said.
“Of note, the blinatumomab arm was less toxic and there was marked improvement in disease-free and overall survival, so this is clearly going to become a new standard of care for relapsed and refractory ALL,” he added.
DAPA-HF: Dapagliflozin benefits regardless of age, HF severity
PHILADELPHIA – The substantial benefits from adding dapagliflozin to guideline-directed medical therapy for patients with heart failure with reduced ejection fraction enrolled in the DAPA-HF trial applied to patients regardless of their age or baseline health status, a pair of new post hoc analyses suggest.
These findings emerged a day after a report that more fully delineated dapagliflozin’s consistent safety and efficacy in patients with heart failure with reduced ejection fraction (HFrEF) regardless of whether they also had type 2 diabetes. One of the new, post hoc analyses reported at the American Heart Association scientific sessions suggested that even the most elderly enrolled patients, 75 years and older, had a similar cut in mortality and acute heart failure exacerbations, compared with younger patients. A second post hoc analysis indicated that patients with severe heart failure symptoms at entry into the trial received about as much benefit from the addition of dapagliflozin as did patients with mild baseline symptoms, measured by the Kansas City Cardiomyopathy Questionnaire (KCCQ).
The primary results from the DAPA-HF (Dapagliflozin and Prevention of Adverse Outcomes in Heart Failure) trial, first reported in August 2019, showed that among more than 4,700 patients with HFrEF randomized to receive the sodium-glucose cotransporter 2 (SGLT2) inhibitor dapagliflozin (Farxiga) on top of standard HFrEF medications or placebo, those who received dapagliflozin had a statistically significant, 26% decrease in their incidence of the primary study endpoint over a median 18 months, regardless of diabetes status (N Engl J Med. 2019 Nov 21;381[21]:1995-2008).
“These benefits were entirely consistent across the range of ages studied,” extending from patients younger than 55 years to those older than 75 years, John McMurray, MD, said at the meeting. “In many parts of the world, particularly North America and Western Europe, we have an increasingly elderly population. Many patients with heart failure are much older than in clinical trials,” he said.
“The thing of concern is whether elderly patients get as much benefit and tolerate treatment as well as younger patients,” said Dr. McMurray, professor of medical cardiology at the University of Glasgow.
“Dapagliflozin worked across all ages, including some very elderly patients enrolled in the trial,” said Mary Norine Walsh, MD, medical director of the heart failure and transplant program at St. Vincent Heart Center of Indiana in Indianapolis. “Many trials have not looked at age like this. I hope this is a new way to analyze trials to produce more information that can help patients,” she said in an interview.
Quality-of-life outcomes
The other new, post hoc analysis showed that patients with severe HF symptoms at entry into the trial received about as much benefit from the addition of dapagliflozin as did patients with milder baseline symptoms and less impaired function, measured by the KCCQ. Dapagliflozin treatment “improved cardiovascular death and worsening heart failure to a similar extent across the entire range of KCCQ at baseline,” Mikhail N. Kosiborod, MD, said in a separate talk at the meeting. In addition, dapagliflozin treatment increased the rate of small, moderate, and large clinically meaningful improvements in patients’ KCCQ scores across all key domains of the metric, which scores symptom frequency and severity, physical and social limitations, and quality of life, said Dr. Kosiborod, a cardiologist and professor of medicine at the University of Missouri–Kansas City.
After the first 8 months of treatment in the DAPA-HF trial, 58% of the 2,373 patients who received dapagliflozin had a clinically meaningful improvement in their total KCCQ symptom score of at least 5 points, compared with a 51% rate in the 2,371 patients in the control arm, a statistically significant difference. This meant that the number needed to treat with dapagliflozin was 14 patients to produce one additional patient with at least a 5-point KCCQ improvement compared with controls, a “very small” number needed to treat, Dr. Kosiborod said in an interview.
Addition of the KCCQ to the panel of assessments that patients underwent during DAPA-HF reflected an evolved approach to measuring efficacy outcomes in clinical trials by including patient-reported outcomes. Earlier in 2019, the Food and Drug Administration released draft guidance for heart failure drug development that explicitly called for efficacy endpoints in pivotal studies that measure how patients feel and function, and stating that these endpoints can be the basis for new drug approvals.
“To many patients, how they feel matters as much if not more than how long they live,” Dr. Kosiborod noted. The goals of heart failure treatments are not only to extend survival and reduced hospitalizations, but also to improve symptoms, function, and quality of life, he said.
“There is a lot of interest now in having outcomes in heart failure trials that are more meaningful to patients, like feeling better and being able to do more,” noted Dr. Walsh.
The DAPA-HF results also showed that patients had similar rates of reduction in death, heart failure hospitalization, or urgent clinical visits, regardless of how severely they were affected by their heart failure when they began dapagliflozin treatment. The researchers ran an analysis that divided the entire trial population into tertiles based on their KCCQ score on entering the study. Patients in the most severely-affected tertile had a 30% cut in their rate of death or acute heart failure exacerbation on dapagliflozin compared with placebo, while patients in the tertile with the mildest symptoms at baseline had a 38% reduction in their primary outcome incidence compared with controls who received placebo. Concurrently with Dr. Kosiborod’s report, the results appeared in an article online (Circulation. 2019 Nov 17. doi: 10.1161/CIRCULATIONAHA.119.044138).
Outcomes by age
Not surprisingly in DAPA-HF, the older patients were, the sicker, Dr. McMurray observed. Of the study’s 1,149 patients (24% of the study cohort) who were at least 75 years old, 62% had chronic kidney disease, compared with a 14% prevalence among the 636 patients younger than age 55. The 75-and-older group showed a steeper, 32% decline in incidence of the primary endpoint – a composite of cardiovascular (CV) death, HF hospitalization, or urgent HF visit requiring intravenous therapy – than in the other studied age groups: a 24% decline in those 65-74 years old, a 29% cut in those 55-64 years old, and a 13% drop in patients younger than 55 years old.
In addition, patients aged 75 years or greater were just as likely as the overall group to show at least a 5-point improvement in their KCCQ Total Symptom Score on dapagliflozin, as well as about the same reduced rate of deterioration compared with placebo as tracked with the KCCQ.
Patients “got as much benefit in terms of symptoms as well as morbidity and mortality,” Dr. McMurray concluded. Concurrently with the meeting report the results appeared in an article online (Circulation. 2019 Nov 17. doi: 10.1161/CIRCULATIONAHA.119.044133).
“These data are of critical importance, as improving patient-reported outcomes in heart failure, especially in highly symptomatic patients, is an important goal in drug development,” G. Michael Felker, MD, wrote in an editorial accompanying the two published analyses (Circulation. 2019 Nov 17. doi: 10.1161/CIRCULATIONAHA.119.044578). These new analyses also highlight another attractive feature of dapagliflozin and, apparently, the entire class of SGLT2 inhibitors: They “ ‘play well with others’ when it comes to overlapping intolerances that often limit (either in reality or in perception) optimization of GDMT [guideline-directed medical therapy]. Although SGLT2 inhibitor therapy may lead to volume depletion and require adjustment of diuretics, the SGLT2 inhibitors generally lack some of the other dose-limiting adverse effects (such as renal dysfunction, hyperkalemia, and hypotension) that can make aggressive up-titration of GDMT problematic, particularly in older patients or those with more advanced disease,“ wrote Dr. Felker, professor of medicine at Duke University in Durham, N.C. “We stand at the beginning of a new era of ‘quadruple therapy’ for HFrEF with beta-blockers, an angiotensin receptor neprilysin inhibitor, mineralocorticoid receptor antagonists, and SGLT2 inhibitors,” he concluded.
A version of this article also appears on Medscape.com
In DAPA-HF, treatment with dapagliflozin met the three critical goals of heart failure management. When used on top of current guideline-directed medical therapy, the treatment reduced mortality, cut hospitalizations, and improved heart failure–related health status – all to a similar extent regardless of patients’ age or symptom severity at entry. These new, post hoc findings provide important, additional data supporting inhibition of sodium-glucose cotransporter (SGLT) 2 with dapagliflozin as the newest foundational pillar of treatment for heart failure with reduced ejection fraction (HFrEF).
The results of the analysis by baseline symptoms severity as measured by the Kansas City Cardiomyopathy Questionnaire (KCCQ) showed similar treatment effects from dapagliflozin regardless of a patient’s baseline KCCQ score, suggesting that the prior report of a blunted effect of dapagliflozin in patients classified at baseline as being in New York Heart Association functional class III or IV compared with class I and II patients was likely a chance finding.
Both the analyses by age and by KCCQ scores were limited by their post hoc status using data collected in a single study. No evidence addresses whether these are class effects for all drugs in the SGLT2-inhibitor class, whether these findings from DAPA-HF are generalizable to real world practice, or whether treatment with dapagliflozin would have similar effects on outcomes if it had been used more often in combination with sacubitril/valsartan. In DAPA-HF, 11% of patients also received sacubitril/valsartan even though existing management guidelines recommend sacubitril/valsartan as the preferred agent for inhibiting the renin-angiotensin system.
It’s also unclear whether patient-reported outcomes such as those measured by the KCCQ will help in sequencing the introduction of drugs for HFrEF patients, or drug selection by patients, providers, payers, and in guidelines.
Carolyn S.P. Lam, MD, is professor of medicine at Duke-National University of Singapore. She has been a consultant to and has received research funding from AstraZeneca and several other companies. She made these comments as designated discussant for the two reports.
In DAPA-HF, treatment with dapagliflozin met the three critical goals of heart failure management. When used on top of current guideline-directed medical therapy, the treatment reduced mortality, cut hospitalizations, and improved heart failure–related health status – all to a similar extent regardless of patients’ age or symptom severity at entry. These new, post hoc findings provide important, additional data supporting inhibition of sodium-glucose cotransporter (SGLT) 2 with dapagliflozin as the newest foundational pillar of treatment for heart failure with reduced ejection fraction (HFrEF).
The results of the analysis by baseline symptoms severity as measured by the Kansas City Cardiomyopathy Questionnaire (KCCQ) showed similar treatment effects from dapagliflozin regardless of a patient’s baseline KCCQ score, suggesting that the prior report of a blunted effect of dapagliflozin in patients classified at baseline as being in New York Heart Association functional class III or IV compared with class I and II patients was likely a chance finding.
Both the analyses by age and by KCCQ scores were limited by their post hoc status using data collected in a single study. No evidence addresses whether these are class effects for all drugs in the SGLT2-inhibitor class, whether these findings from DAPA-HF are generalizable to real world practice, or whether treatment with dapagliflozin would have similar effects on outcomes if it had been used more often in combination with sacubitril/valsartan. In DAPA-HF, 11% of patients also received sacubitril/valsartan even though existing management guidelines recommend sacubitril/valsartan as the preferred agent for inhibiting the renin-angiotensin system.
It’s also unclear whether patient-reported outcomes such as those measured by the KCCQ will help in sequencing the introduction of drugs for HFrEF patients, or drug selection by patients, providers, payers, and in guidelines.
Carolyn S.P. Lam, MD, is professor of medicine at Duke-National University of Singapore. She has been a consultant to and has received research funding from AstraZeneca and several other companies. She made these comments as designated discussant for the two reports.
In DAPA-HF, treatment with dapagliflozin met the three critical goals of heart failure management. When used on top of current guideline-directed medical therapy, the treatment reduced mortality, cut hospitalizations, and improved heart failure–related health status – all to a similar extent regardless of patients’ age or symptom severity at entry. These new, post hoc findings provide important, additional data supporting inhibition of sodium-glucose cotransporter (SGLT) 2 with dapagliflozin as the newest foundational pillar of treatment for heart failure with reduced ejection fraction (HFrEF).
The results of the analysis by baseline symptoms severity as measured by the Kansas City Cardiomyopathy Questionnaire (KCCQ) showed similar treatment effects from dapagliflozin regardless of a patient’s baseline KCCQ score, suggesting that the prior report of a blunted effect of dapagliflozin in patients classified at baseline as being in New York Heart Association functional class III or IV compared with class I and II patients was likely a chance finding.
Both the analyses by age and by KCCQ scores were limited by their post hoc status using data collected in a single study. No evidence addresses whether these are class effects for all drugs in the SGLT2-inhibitor class, whether these findings from DAPA-HF are generalizable to real world practice, or whether treatment with dapagliflozin would have similar effects on outcomes if it had been used more often in combination with sacubitril/valsartan. In DAPA-HF, 11% of patients also received sacubitril/valsartan even though existing management guidelines recommend sacubitril/valsartan as the preferred agent for inhibiting the renin-angiotensin system.
It’s also unclear whether patient-reported outcomes such as those measured by the KCCQ will help in sequencing the introduction of drugs for HFrEF patients, or drug selection by patients, providers, payers, and in guidelines.
Carolyn S.P. Lam, MD, is professor of medicine at Duke-National University of Singapore. She has been a consultant to and has received research funding from AstraZeneca and several other companies. She made these comments as designated discussant for the two reports.
PHILADELPHIA – The substantial benefits from adding dapagliflozin to guideline-directed medical therapy for patients with heart failure with reduced ejection fraction enrolled in the DAPA-HF trial applied to patients regardless of their age or baseline health status, a pair of new post hoc analyses suggest.
These findings emerged a day after a report that more fully delineated dapagliflozin’s consistent safety and efficacy in patients with heart failure with reduced ejection fraction (HFrEF) regardless of whether they also had type 2 diabetes. One of the new, post hoc analyses reported at the American Heart Association scientific sessions suggested that even the most elderly enrolled patients, 75 years and older, had a similar cut in mortality and acute heart failure exacerbations, compared with younger patients. A second post hoc analysis indicated that patients with severe heart failure symptoms at entry into the trial received about as much benefit from the addition of dapagliflozin as did patients with mild baseline symptoms, measured by the Kansas City Cardiomyopathy Questionnaire (KCCQ).
The primary results from the DAPA-HF (Dapagliflozin and Prevention of Adverse Outcomes in Heart Failure) trial, first reported in August 2019, showed that among more than 4,700 patients with HFrEF randomized to receive the sodium-glucose cotransporter 2 (SGLT2) inhibitor dapagliflozin (Farxiga) on top of standard HFrEF medications or placebo, those who received dapagliflozin had a statistically significant, 26% decrease in their incidence of the primary study endpoint over a median 18 months, regardless of diabetes status (N Engl J Med. 2019 Nov 21;381[21]:1995-2008).
“These benefits were entirely consistent across the range of ages studied,” extending from patients younger than 55 years to those older than 75 years, John McMurray, MD, said at the meeting. “In many parts of the world, particularly North America and Western Europe, we have an increasingly elderly population. Many patients with heart failure are much older than in clinical trials,” he said.
“The thing of concern is whether elderly patients get as much benefit and tolerate treatment as well as younger patients,” said Dr. McMurray, professor of medical cardiology at the University of Glasgow.
“Dapagliflozin worked across all ages, including some very elderly patients enrolled in the trial,” said Mary Norine Walsh, MD, medical director of the heart failure and transplant program at St. Vincent Heart Center of Indiana in Indianapolis. “Many trials have not looked at age like this. I hope this is a new way to analyze trials to produce more information that can help patients,” she said in an interview.
Quality-of-life outcomes
The other new, post hoc analysis showed that patients with severe HF symptoms at entry into the trial received about as much benefit from the addition of dapagliflozin as did patients with milder baseline symptoms and less impaired function, measured by the KCCQ. Dapagliflozin treatment “improved cardiovascular death and worsening heart failure to a similar extent across the entire range of KCCQ at baseline,” Mikhail N. Kosiborod, MD, said in a separate talk at the meeting. In addition, dapagliflozin treatment increased the rate of small, moderate, and large clinically meaningful improvements in patients’ KCCQ scores across all key domains of the metric, which scores symptom frequency and severity, physical and social limitations, and quality of life, said Dr. Kosiborod, a cardiologist and professor of medicine at the University of Missouri–Kansas City.
After the first 8 months of treatment in the DAPA-HF trial, 58% of the 2,373 patients who received dapagliflozin had a clinically meaningful improvement in their total KCCQ symptom score of at least 5 points, compared with a 51% rate in the 2,371 patients in the control arm, a statistically significant difference. This meant that the number needed to treat with dapagliflozin was 14 patients to produce one additional patient with at least a 5-point KCCQ improvement compared with controls, a “very small” number needed to treat, Dr. Kosiborod said in an interview.
Addition of the KCCQ to the panel of assessments that patients underwent during DAPA-HF reflected an evolved approach to measuring efficacy outcomes in clinical trials by including patient-reported outcomes. Earlier in 2019, the Food and Drug Administration released draft guidance for heart failure drug development that explicitly called for efficacy endpoints in pivotal studies that measure how patients feel and function, and stating that these endpoints can be the basis for new drug approvals.
“To many patients, how they feel matters as much if not more than how long they live,” Dr. Kosiborod noted. The goals of heart failure treatments are not only to extend survival and reduced hospitalizations, but also to improve symptoms, function, and quality of life, he said.
“There is a lot of interest now in having outcomes in heart failure trials that are more meaningful to patients, like feeling better and being able to do more,” noted Dr. Walsh.
The DAPA-HF results also showed that patients had similar rates of reduction in death, heart failure hospitalization, or urgent clinical visits, regardless of how severely they were affected by their heart failure when they began dapagliflozin treatment. The researchers ran an analysis that divided the entire trial population into tertiles based on their KCCQ score on entering the study. Patients in the most severely-affected tertile had a 30% cut in their rate of death or acute heart failure exacerbation on dapagliflozin compared with placebo, while patients in the tertile with the mildest symptoms at baseline had a 38% reduction in their primary outcome incidence compared with controls who received placebo. Concurrently with Dr. Kosiborod’s report, the results appeared in an article online (Circulation. 2019 Nov 17. doi: 10.1161/CIRCULATIONAHA.119.044138).
Outcomes by age
Not surprisingly in DAPA-HF, the older patients were, the sicker, Dr. McMurray observed. Of the study’s 1,149 patients (24% of the study cohort) who were at least 75 years old, 62% had chronic kidney disease, compared with a 14% prevalence among the 636 patients younger than age 55. The 75-and-older group showed a steeper, 32% decline in incidence of the primary endpoint – a composite of cardiovascular (CV) death, HF hospitalization, or urgent HF visit requiring intravenous therapy – than in the other studied age groups: a 24% decline in those 65-74 years old, a 29% cut in those 55-64 years old, and a 13% drop in patients younger than 55 years old.
In addition, patients aged 75 years or greater were just as likely as the overall group to show at least a 5-point improvement in their KCCQ Total Symptom Score on dapagliflozin, as well as about the same reduced rate of deterioration compared with placebo as tracked with the KCCQ.
Patients “got as much benefit in terms of symptoms as well as morbidity and mortality,” Dr. McMurray concluded. Concurrently with the meeting report the results appeared in an article online (Circulation. 2019 Nov 17. doi: 10.1161/CIRCULATIONAHA.119.044133).
“These data are of critical importance, as improving patient-reported outcomes in heart failure, especially in highly symptomatic patients, is an important goal in drug development,” G. Michael Felker, MD, wrote in an editorial accompanying the two published analyses (Circulation. 2019 Nov 17. doi: 10.1161/CIRCULATIONAHA.119.044578). These new analyses also highlight another attractive feature of dapagliflozin and, apparently, the entire class of SGLT2 inhibitors: They “ ‘play well with others’ when it comes to overlapping intolerances that often limit (either in reality or in perception) optimization of GDMT [guideline-directed medical therapy]. Although SGLT2 inhibitor therapy may lead to volume depletion and require adjustment of diuretics, the SGLT2 inhibitors generally lack some of the other dose-limiting adverse effects (such as renal dysfunction, hyperkalemia, and hypotension) that can make aggressive up-titration of GDMT problematic, particularly in older patients or those with more advanced disease,“ wrote Dr. Felker, professor of medicine at Duke University in Durham, N.C. “We stand at the beginning of a new era of ‘quadruple therapy’ for HFrEF with beta-blockers, an angiotensin receptor neprilysin inhibitor, mineralocorticoid receptor antagonists, and SGLT2 inhibitors,” he concluded.
A version of this article also appears on Medscape.com
PHILADELPHIA – The substantial benefits from adding dapagliflozin to guideline-directed medical therapy for patients with heart failure with reduced ejection fraction enrolled in the DAPA-HF trial applied to patients regardless of their age or baseline health status, a pair of new post hoc analyses suggest.
These findings emerged a day after a report that more fully delineated dapagliflozin’s consistent safety and efficacy in patients with heart failure with reduced ejection fraction (HFrEF) regardless of whether they also had type 2 diabetes. One of the new, post hoc analyses reported at the American Heart Association scientific sessions suggested that even the most elderly enrolled patients, 75 years and older, had a similar cut in mortality and acute heart failure exacerbations, compared with younger patients. A second post hoc analysis indicated that patients with severe heart failure symptoms at entry into the trial received about as much benefit from the addition of dapagliflozin as did patients with mild baseline symptoms, measured by the Kansas City Cardiomyopathy Questionnaire (KCCQ).
The primary results from the DAPA-HF (Dapagliflozin and Prevention of Adverse Outcomes in Heart Failure) trial, first reported in August 2019, showed that among more than 4,700 patients with HFrEF randomized to receive the sodium-glucose cotransporter 2 (SGLT2) inhibitor dapagliflozin (Farxiga) on top of standard HFrEF medications or placebo, those who received dapagliflozin had a statistically significant, 26% decrease in their incidence of the primary study endpoint over a median 18 months, regardless of diabetes status (N Engl J Med. 2019 Nov 21;381[21]:1995-2008).
“These benefits were entirely consistent across the range of ages studied,” extending from patients younger than 55 years to those older than 75 years, John McMurray, MD, said at the meeting. “In many parts of the world, particularly North America and Western Europe, we have an increasingly elderly population. Many patients with heart failure are much older than in clinical trials,” he said.
“The thing of concern is whether elderly patients get as much benefit and tolerate treatment as well as younger patients,” said Dr. McMurray, professor of medical cardiology at the University of Glasgow.
“Dapagliflozin worked across all ages, including some very elderly patients enrolled in the trial,” said Mary Norine Walsh, MD, medical director of the heart failure and transplant program at St. Vincent Heart Center of Indiana in Indianapolis. “Many trials have not looked at age like this. I hope this is a new way to analyze trials to produce more information that can help patients,” she said in an interview.
Quality-of-life outcomes
The other new, post hoc analysis showed that patients with severe HF symptoms at entry into the trial received about as much benefit from the addition of dapagliflozin as did patients with milder baseline symptoms and less impaired function, measured by the KCCQ. Dapagliflozin treatment “improved cardiovascular death and worsening heart failure to a similar extent across the entire range of KCCQ at baseline,” Mikhail N. Kosiborod, MD, said in a separate talk at the meeting. In addition, dapagliflozin treatment increased the rate of small, moderate, and large clinically meaningful improvements in patients’ KCCQ scores across all key domains of the metric, which scores symptom frequency and severity, physical and social limitations, and quality of life, said Dr. Kosiborod, a cardiologist and professor of medicine at the University of Missouri–Kansas City.
After the first 8 months of treatment in the DAPA-HF trial, 58% of the 2,373 patients who received dapagliflozin had a clinically meaningful improvement in their total KCCQ symptom score of at least 5 points, compared with a 51% rate in the 2,371 patients in the control arm, a statistically significant difference. This meant that the number needed to treat with dapagliflozin was 14 patients to produce one additional patient with at least a 5-point KCCQ improvement compared with controls, a “very small” number needed to treat, Dr. Kosiborod said in an interview.
Addition of the KCCQ to the panel of assessments that patients underwent during DAPA-HF reflected an evolved approach to measuring efficacy outcomes in clinical trials by including patient-reported outcomes. Earlier in 2019, the Food and Drug Administration released draft guidance for heart failure drug development that explicitly called for efficacy endpoints in pivotal studies that measure how patients feel and function, and stating that these endpoints can be the basis for new drug approvals.
“To many patients, how they feel matters as much if not more than how long they live,” Dr. Kosiborod noted. The goals of heart failure treatments are not only to extend survival and reduced hospitalizations, but also to improve symptoms, function, and quality of life, he said.
“There is a lot of interest now in having outcomes in heart failure trials that are more meaningful to patients, like feeling better and being able to do more,” noted Dr. Walsh.
The DAPA-HF results also showed that patients had similar rates of reduction in death, heart failure hospitalization, or urgent clinical visits, regardless of how severely they were affected by their heart failure when they began dapagliflozin treatment. The researchers ran an analysis that divided the entire trial population into tertiles based on their KCCQ score on entering the study. Patients in the most severely-affected tertile had a 30% cut in their rate of death or acute heart failure exacerbation on dapagliflozin compared with placebo, while patients in the tertile with the mildest symptoms at baseline had a 38% reduction in their primary outcome incidence compared with controls who received placebo. Concurrently with Dr. Kosiborod’s report, the results appeared in an article online (Circulation. 2019 Nov 17. doi: 10.1161/CIRCULATIONAHA.119.044138).
Outcomes by age
Not surprisingly in DAPA-HF, the older patients were, the sicker, Dr. McMurray observed. Of the study’s 1,149 patients (24% of the study cohort) who were at least 75 years old, 62% had chronic kidney disease, compared with a 14% prevalence among the 636 patients younger than age 55. The 75-and-older group showed a steeper, 32% decline in incidence of the primary endpoint – a composite of cardiovascular (CV) death, HF hospitalization, or urgent HF visit requiring intravenous therapy – than in the other studied age groups: a 24% decline in those 65-74 years old, a 29% cut in those 55-64 years old, and a 13% drop in patients younger than 55 years old.
In addition, patients aged 75 years or greater were just as likely as the overall group to show at least a 5-point improvement in their KCCQ Total Symptom Score on dapagliflozin, as well as about the same reduced rate of deterioration compared with placebo as tracked with the KCCQ.
Patients “got as much benefit in terms of symptoms as well as morbidity and mortality,” Dr. McMurray concluded. Concurrently with the meeting report the results appeared in an article online (Circulation. 2019 Nov 17. doi: 10.1161/CIRCULATIONAHA.119.044133).
“These data are of critical importance, as improving patient-reported outcomes in heart failure, especially in highly symptomatic patients, is an important goal in drug development,” G. Michael Felker, MD, wrote in an editorial accompanying the two published analyses (Circulation. 2019 Nov 17. doi: 10.1161/CIRCULATIONAHA.119.044578). These new analyses also highlight another attractive feature of dapagliflozin and, apparently, the entire class of SGLT2 inhibitors: They “ ‘play well with others’ when it comes to overlapping intolerances that often limit (either in reality or in perception) optimization of GDMT [guideline-directed medical therapy]. Although SGLT2 inhibitor therapy may lead to volume depletion and require adjustment of diuretics, the SGLT2 inhibitors generally lack some of the other dose-limiting adverse effects (such as renal dysfunction, hyperkalemia, and hypotension) that can make aggressive up-titration of GDMT problematic, particularly in older patients or those with more advanced disease,“ wrote Dr. Felker, professor of medicine at Duke University in Durham, N.C. “We stand at the beginning of a new era of ‘quadruple therapy’ for HFrEF with beta-blockers, an angiotensin receptor neprilysin inhibitor, mineralocorticoid receptor antagonists, and SGLT2 inhibitors,” he concluded.
A version of this article also appears on Medscape.com
REPORTING FROM THE AHA SCIENTIFIC SESSIONS
Combo elicits lasting responses in metastatic melanoma
NATIONAL HARBOR, MD. – The combination of bempegaldesleukin and nivolumab produced durable responses in a phase 1/2 trial of patients with previously untreated metastatic melanoma.
The overall response rate was 53%, and most responders were still in response at a median follow-up of about 19 months. The median progression-free survival was not reached, and the combination was considered well tolerated.
Adi Diab, MD, of the University of Texas MD Anderson Cancer Center, Houston, presented these results from the PIVOT-02 study at the annual meeting of the Society for Immunotherapy of Cancer.
Dr. Diab explained that bempegaldesleukin (bempeg) is a CD122-preferential interleukin-2 pathway agonist, and earlier results from the PIVOT-02 trial showed that adding bempeg to nivolumab can convert baseline tumors from programmed death–ligand 1 (PD-L1) negative to PD-L1 positive (SITC 2018, Abstract O4).
Dr. Diab presented updated results from PIVOT-02 (NCT02983045) in 41 patients with metastatic melanoma who received bempeg plus nivolumab as first-line treatment. The patients had a median age of 63 years (range, 22-80 years) at baseline, and 58.5% were male. Most patients (58.5%) were PD-L1 positive, although PD-L1 status was unknown in 7.3% of patients.
Patients received bempeg at 0.006 mg/kg and nivolumab at 360 mg every 3 weeks. They received a median of nine cycles (range, 1-34), and the median follow-up was 18.6 months.
Efficacy
In the 38 patients who were evaluable for efficacy, the overall response rate was 53% (n = 20), and the complete response rate was 34% (n = 13). The median time to response was 2.0 months, and the median time to complete response was 7.9 months.
Dr. Diab noted that responses were seen regardless of PD-L1 expression at baseline. The response rate was 39% among PD-L1-negative patients, 64% among PD-L1-positive patients, and 33% among patients whose PD-L1 status was unknown.
Dr. Diab also pointed out that responses were durable and deepened over time. The median duration of response was not reached, and 17 of the 20 responders had ongoing responses at last follow-up. The median progression-free survival has not been reached.
Safety
“This combination is safe and tolerable, there’s no overlapping immune-related adverse events, and the most common side effects are grade 1/2 flu-like symptoms,” Dr. Diab said.
The most common grade 1/2 treatment-related adverse events (AEs) were flu-like symptoms (80.5%), rash (70.7%), fatigue (65.9%), pruritus (48.8%), nausea (46.3%), arthralgia (43.9%), decreased appetite (36.6%), and myalgia (36.6%).
Dr. Diab noted that cytokine-related AEs (flu-like symptoms, rash, and pruritus) were easily managed with NSAIDs; decreased with subsequent cycles of treatment; and did not necessitate dose delays, reductions, or discontinuations.
Grade 3/4 treatment-related AEs included two cases of acute kidney injury, two cases of atrial fibrillation, one case of dizziness, one case of dyspnea, one case of hypoxia, one case of hyperglycemia, and one case of hypernatremia.
Five patients discontinued treatment because of related AEs, including cerebrovascular accident, peripheral edema, blood creatinine increase, malaise, and pharyngitis. There were no treatment-related deaths.
Dr. Diab said these results were used to support the recent breakthrough therapy designation granted to bempeg in combination with nivolumab. The results have also prompted a phase 3 trial in which researchers are comparing the combination with nivolumab alone (NCT03635983).
The phase 1/2 trial is sponsored by Nektar Therapeutics in collaboration with Bristol-Myers Squibb. Dr. Diab reported relationships with Nektar, Celgene, CureVac, Idera, and Pfizer.
SOURCE: Diab A et al. SITC 2019, Abstract O35.
NATIONAL HARBOR, MD. – The combination of bempegaldesleukin and nivolumab produced durable responses in a phase 1/2 trial of patients with previously untreated metastatic melanoma.
The overall response rate was 53%, and most responders were still in response at a median follow-up of about 19 months. The median progression-free survival was not reached, and the combination was considered well tolerated.
Adi Diab, MD, of the University of Texas MD Anderson Cancer Center, Houston, presented these results from the PIVOT-02 study at the annual meeting of the Society for Immunotherapy of Cancer.
Dr. Diab explained that bempegaldesleukin (bempeg) is a CD122-preferential interleukin-2 pathway agonist, and earlier results from the PIVOT-02 trial showed that adding bempeg to nivolumab can convert baseline tumors from programmed death–ligand 1 (PD-L1) negative to PD-L1 positive (SITC 2018, Abstract O4).
Dr. Diab presented updated results from PIVOT-02 (NCT02983045) in 41 patients with metastatic melanoma who received bempeg plus nivolumab as first-line treatment. The patients had a median age of 63 years (range, 22-80 years) at baseline, and 58.5% were male. Most patients (58.5%) were PD-L1 positive, although PD-L1 status was unknown in 7.3% of patients.
Patients received bempeg at 0.006 mg/kg and nivolumab at 360 mg every 3 weeks. They received a median of nine cycles (range, 1-34), and the median follow-up was 18.6 months.
Efficacy
In the 38 patients who were evaluable for efficacy, the overall response rate was 53% (n = 20), and the complete response rate was 34% (n = 13). The median time to response was 2.0 months, and the median time to complete response was 7.9 months.
Dr. Diab noted that responses were seen regardless of PD-L1 expression at baseline. The response rate was 39% among PD-L1-negative patients, 64% among PD-L1-positive patients, and 33% among patients whose PD-L1 status was unknown.
Dr. Diab also pointed out that responses were durable and deepened over time. The median duration of response was not reached, and 17 of the 20 responders had ongoing responses at last follow-up. The median progression-free survival has not been reached.
Safety
“This combination is safe and tolerable, there’s no overlapping immune-related adverse events, and the most common side effects are grade 1/2 flu-like symptoms,” Dr. Diab said.
The most common grade 1/2 treatment-related adverse events (AEs) were flu-like symptoms (80.5%), rash (70.7%), fatigue (65.9%), pruritus (48.8%), nausea (46.3%), arthralgia (43.9%), decreased appetite (36.6%), and myalgia (36.6%).
Dr. Diab noted that cytokine-related AEs (flu-like symptoms, rash, and pruritus) were easily managed with NSAIDs; decreased with subsequent cycles of treatment; and did not necessitate dose delays, reductions, or discontinuations.
Grade 3/4 treatment-related AEs included two cases of acute kidney injury, two cases of atrial fibrillation, one case of dizziness, one case of dyspnea, one case of hypoxia, one case of hyperglycemia, and one case of hypernatremia.
Five patients discontinued treatment because of related AEs, including cerebrovascular accident, peripheral edema, blood creatinine increase, malaise, and pharyngitis. There were no treatment-related deaths.
Dr. Diab said these results were used to support the recent breakthrough therapy designation granted to bempeg in combination with nivolumab. The results have also prompted a phase 3 trial in which researchers are comparing the combination with nivolumab alone (NCT03635983).
The phase 1/2 trial is sponsored by Nektar Therapeutics in collaboration with Bristol-Myers Squibb. Dr. Diab reported relationships with Nektar, Celgene, CureVac, Idera, and Pfizer.
SOURCE: Diab A et al. SITC 2019, Abstract O35.
NATIONAL HARBOR, MD. – The combination of bempegaldesleukin and nivolumab produced durable responses in a phase 1/2 trial of patients with previously untreated metastatic melanoma.
The overall response rate was 53%, and most responders were still in response at a median follow-up of about 19 months. The median progression-free survival was not reached, and the combination was considered well tolerated.
Adi Diab, MD, of the University of Texas MD Anderson Cancer Center, Houston, presented these results from the PIVOT-02 study at the annual meeting of the Society for Immunotherapy of Cancer.
Dr. Diab explained that bempegaldesleukin (bempeg) is a CD122-preferential interleukin-2 pathway agonist, and earlier results from the PIVOT-02 trial showed that adding bempeg to nivolumab can convert baseline tumors from programmed death–ligand 1 (PD-L1) negative to PD-L1 positive (SITC 2018, Abstract O4).
Dr. Diab presented updated results from PIVOT-02 (NCT02983045) in 41 patients with metastatic melanoma who received bempeg plus nivolumab as first-line treatment. The patients had a median age of 63 years (range, 22-80 years) at baseline, and 58.5% were male. Most patients (58.5%) were PD-L1 positive, although PD-L1 status was unknown in 7.3% of patients.
Patients received bempeg at 0.006 mg/kg and nivolumab at 360 mg every 3 weeks. They received a median of nine cycles (range, 1-34), and the median follow-up was 18.6 months.
Efficacy
In the 38 patients who were evaluable for efficacy, the overall response rate was 53% (n = 20), and the complete response rate was 34% (n = 13). The median time to response was 2.0 months, and the median time to complete response was 7.9 months.
Dr. Diab noted that responses were seen regardless of PD-L1 expression at baseline. The response rate was 39% among PD-L1-negative patients, 64% among PD-L1-positive patients, and 33% among patients whose PD-L1 status was unknown.
Dr. Diab also pointed out that responses were durable and deepened over time. The median duration of response was not reached, and 17 of the 20 responders had ongoing responses at last follow-up. The median progression-free survival has not been reached.
Safety
“This combination is safe and tolerable, there’s no overlapping immune-related adverse events, and the most common side effects are grade 1/2 flu-like symptoms,” Dr. Diab said.
The most common grade 1/2 treatment-related adverse events (AEs) were flu-like symptoms (80.5%), rash (70.7%), fatigue (65.9%), pruritus (48.8%), nausea (46.3%), arthralgia (43.9%), decreased appetite (36.6%), and myalgia (36.6%).
Dr. Diab noted that cytokine-related AEs (flu-like symptoms, rash, and pruritus) were easily managed with NSAIDs; decreased with subsequent cycles of treatment; and did not necessitate dose delays, reductions, or discontinuations.
Grade 3/4 treatment-related AEs included two cases of acute kidney injury, two cases of atrial fibrillation, one case of dizziness, one case of dyspnea, one case of hypoxia, one case of hyperglycemia, and one case of hypernatremia.
Five patients discontinued treatment because of related AEs, including cerebrovascular accident, peripheral edema, blood creatinine increase, malaise, and pharyngitis. There were no treatment-related deaths.
Dr. Diab said these results were used to support the recent breakthrough therapy designation granted to bempeg in combination with nivolumab. The results have also prompted a phase 3 trial in which researchers are comparing the combination with nivolumab alone (NCT03635983).
The phase 1/2 trial is sponsored by Nektar Therapeutics in collaboration with Bristol-Myers Squibb. Dr. Diab reported relationships with Nektar, Celgene, CureVac, Idera, and Pfizer.
SOURCE: Diab A et al. SITC 2019, Abstract O35.
REPORTING FROM SITC 2019
Survey: Cancer-related pain, opioid use up since 2018
Cancer-related pain was more common among patients in 2019 than in 2018, as was the use of prescription opioids, according to the American Society of Clinical Oncology.
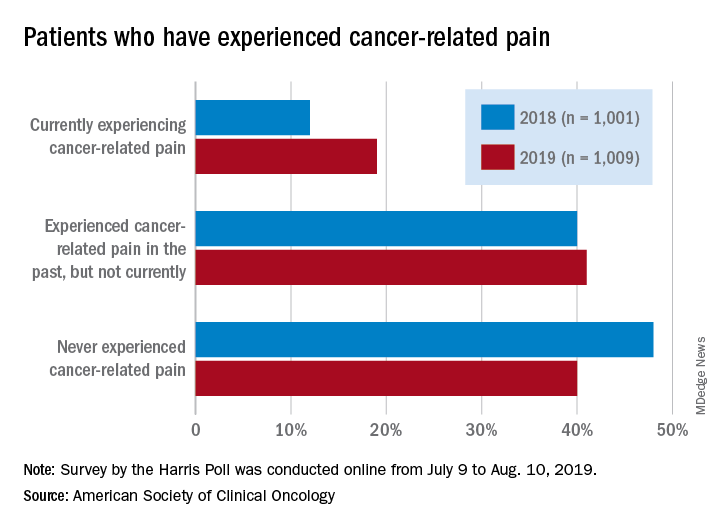
Patients who have/had cancer were significantly more likely to report that they were currently experiencing cancer-related pain in 2019 (19%) than in 2018 (12%), but there was only a slight increase in patients who said that they had experienced cancer-related pain in the past, the society reported in its National Cancer Opinion Survey.
When asked about methods used to manage pain, nausea, and other symptoms, patients diagnosed with cancer most often said that they had not used anything in the last 12 months, although this response was significantly less common in 2019 (48%) than in 2018 (55%). Over-the-counter pain relievers were the most common method used (24% in 2019 and 22% in 2018), followed by vitamins/minerals/herbs (18% in 2019 and 17% in 2018), ASCO said.
Prescription opioids were the third most popular choice for symptom management both years, but use was significantly higher in 2019 (17%) than in 2018 (12%). Also showing a significant increase from 2018 to 2019 was use of medical marijuana, which went from 5% to 10%, ASCO said.
The survey was conducted online for ASCO by the Harris Poll from July 9 to Aug. 10, 2019. The total sample consisted of 4,815 U.S. adults, of whom 1,009 had been diagnosed with cancer.
Cancer-related pain was more common among patients in 2019 than in 2018, as was the use of prescription opioids, according to the American Society of Clinical Oncology.

Patients who have/had cancer were significantly more likely to report that they were currently experiencing cancer-related pain in 2019 (19%) than in 2018 (12%), but there was only a slight increase in patients who said that they had experienced cancer-related pain in the past, the society reported in its National Cancer Opinion Survey.
When asked about methods used to manage pain, nausea, and other symptoms, patients diagnosed with cancer most often said that they had not used anything in the last 12 months, although this response was significantly less common in 2019 (48%) than in 2018 (55%). Over-the-counter pain relievers were the most common method used (24% in 2019 and 22% in 2018), followed by vitamins/minerals/herbs (18% in 2019 and 17% in 2018), ASCO said.
Prescription opioids were the third most popular choice for symptom management both years, but use was significantly higher in 2019 (17%) than in 2018 (12%). Also showing a significant increase from 2018 to 2019 was use of medical marijuana, which went from 5% to 10%, ASCO said.
The survey was conducted online for ASCO by the Harris Poll from July 9 to Aug. 10, 2019. The total sample consisted of 4,815 U.S. adults, of whom 1,009 had been diagnosed with cancer.
Cancer-related pain was more common among patients in 2019 than in 2018, as was the use of prescription opioids, according to the American Society of Clinical Oncology.

Patients who have/had cancer were significantly more likely to report that they were currently experiencing cancer-related pain in 2019 (19%) than in 2018 (12%), but there was only a slight increase in patients who said that they had experienced cancer-related pain in the past, the society reported in its National Cancer Opinion Survey.
When asked about methods used to manage pain, nausea, and other symptoms, patients diagnosed with cancer most often said that they had not used anything in the last 12 months, although this response was significantly less common in 2019 (48%) than in 2018 (55%). Over-the-counter pain relievers were the most common method used (24% in 2019 and 22% in 2018), followed by vitamins/minerals/herbs (18% in 2019 and 17% in 2018), ASCO said.
Prescription opioids were the third most popular choice for symptom management both years, but use was significantly higher in 2019 (17%) than in 2018 (12%). Also showing a significant increase from 2018 to 2019 was use of medical marijuana, which went from 5% to 10%, ASCO said.
The survey was conducted online for ASCO by the Harris Poll from July 9 to Aug. 10, 2019. The total sample consisted of 4,815 U.S. adults, of whom 1,009 had been diagnosed with cancer.
OA management guidelines forgo treatment hierarchy or order but emphasize severity, patient risk factors
ATLANTA – New guidelines for management of osteoarthritis of the hand, knee, and hip from the American College of Rheumatology and the Arthritis Foundation lay out a wide range of treatment options without an algorithm or hierarchy, making strong recommendations for nondrug interventions and for tailoring plans to individual patient-level factors.
Since the ACR last released OA management guidelines in 2012, a number of recommendations have been added, changed, and removed, and the structure of the guidelines has also changed. For instance, the new OA guidelines include a broad list of management options, Sharon L. Kolasinski, MD, chair of the ACR guidelines panel and professor of clinical medicine in the division of rheumatology at the University of Pennsylvania, Philadelphia, said in a presentation at the annual meeting of the American College of Rheumatology.
“The new guideline emphasizes comprehensive management of patients with OA, rather than a stepwise algorithm in a linear manner,” she said.
There is also no hierarchy to the recommendations, apart from the strength of the recommendation. “For any individual patient, a single option may be chosen at a particular time point, perhaps with or without other options, and may be reused in the future. For a given intervention, there might be a period of time over which it’s useful, and then the option might be changed,” Dr. Kolasinski noted.
Dr. Kolasinski advised making treatment decisions based on a patient’s disease severity, whether the patient uses medical devices, and in consideration of patient risk factors. “A history of injuries, surgical history, access to care, personal beliefs and preferences should all be brought to bear on decision making for osteoarthritis management,” she said.
The guidelines also advise considering a patient’s overall well-being and factors related to a patient’s perception of pain and function, such as mood disorders, altered sleep, chronic pain, impaired coping measures, and stress level. “Comprehensive management requires a broad assessment of how pain and function are affecting the patient with OA as a whole and recognizing that multiple options are available. They might be used in combination or change over time,” Dr. Kolasinski said.
The new guidelines place a strong emphasis on educational, behavioral, psychosocial, mind-body, and physical approaches. There are strong recommendations for the use of exercise, including aerobic, strengthening, neuromuscular, and aquatic exercise. Weight loss also carries a strong recommendation for patients with hip and knee OA, with a focus on group-based exercise, education, fitness and exercise goals, and a multidisciplinary approach using self-efficacy and self-management programs. The panels made a strong recommendation for tai chi to improve hip and knee OA. There are also strong recommendations for orthoses; aids and assistive devices such as canes, first carpometacarpal (CMC) orthoses, and tibiofemoral knee braces. Other interventions, such as Kinesio tape for first CMC joint and knee OA, hand orthoses, and patellofemoral knee braces, carried a conditional recommendation. Other conditional recommendations made by the panel were for acupuncture, thermal interventions, and radiofrequency ablation for patients with knee OA. Balance training for hip and knee OA, yoga for knee OA, and cognitive-behavioral therapy all were conditionally recommended by the panel.
The panel strongly recommended against the use of transcutaneous nerve stimulation for hip and knee OA, Dr. Kolasinski noted. The panel also conditionally recommended against use of modified shoes and pulsed vibration therapy in knee OA; lateral or medial wedged insoles, massage, and manual therapy with exercise in hip or knee OA; and iontophoresis in first CMC OA.
Tuhina Neogi, MD, PhD, chief of rheumatology at Boston University and member of the core team that developed the guidelines, said in her presentation the panel chose not to use the term “nonpharmacologic” in the guidelines because it may give patients a false impression that they are not receiving a treatment. “We really need to change our language and change the way in which we approach these conversations with our patients so that they don’t feel that they are not getting a treatment when we’re giving these recommendations,” she said.
Recommendations for, against pharmacologic approaches
The ACR has changed conditional recommendations for topical NSAIDs for knee and hand OA, oral NSAIDs, and intra-articular steroids for knee and hip OA into strong recommendations for the 2019 guidelines, Dr. Kolasinski said. While the 2012 guidelines conditionally recommended against topical capsaicin for knee OA, the new guidelines conditionally recommend it.
Other pharmacologic conditional recommendations included topical NSAIDs, chondroitin sulfate, and intra-articular corticosteroid injections for hand OA, acetaminophen, and duloxetine for knee OA.
With the new recommendations come changes that some rheumatologists and health care providers may find controversial. “I think that the practicing rheumatologist may be surprised that we have a recommendation against the use of hyaluronic acid in the knee as a conditional recommendation,” Dr. Kolasinski said. “The assessment of the literature at this point really reveals that there is equivalence between intra-articular hyaluronic acid injection and intra-articular saline injection, and so it was the feeling of the panel that, really, this was worth changing the recommendation from the 2012 guideline.”
The panel made strong recommendations against use of the following pharmacologic interventions:
- Bisphosphonates.
- Glucosamine sulfate.
- Combination glucosamine sulfate-chondroitin sulfate products.
- Hydroxychloroquine.
- Methotrexate.
- Intra-articular hyaluronic acid injections in hip OA.
- Chondroitin sulfate, platelet-rich plasma injections, and stem cell injections in hip and knee OA.
- Tumor necrosis factor (TNF) inhibitors.
- Interleukin-1–receptor antagonists.
Additionally, the panel made a conditional recommendation against topical capsaicin on the hand, colchicine, fish oil, vitamin D, intra-articular hyaluronic acid injections in the first CMC, and intra-articular botulinum toxin and prolotherapy in hip and knee OA.
The panel did not recommend for or against use of yoga for hip and hand OA, topical lidocaine, pregabalin, gabapentin, selective serotonin reuptake inhibitors, serotonin norepinephrine reuptake inhibitors apart from duloxetine, tricyclic antidepressants, and anti-nerve growth factor agents.
While the panel conditionally recommended against use of opioids, they made a conditional recommendation for use of tramadol opioids, and there was “a heated discussion about that distinction,” Dr. Neogi noted in a discussion session at the meeting. “There was a recent observational study that indicated that tramadol may have an increased risk of [all-cause] mortality, but there are lots of issues of confounding by indication in that study.”
The patient panel also raised strong concerns about the ACR and the Arthritis Foundation coming out against opioids for OA management in their guidelines. “They don’t want to damn opioids, but they’re also concerned about a specialty society coming out strongly against opioids in the concern that their physicians may limit their access to opioids if they’re in a situation where nothing else is helping them,” Dr. Neogi said.
Dr. Kolasinski noted the guidelines will be published online in Arthritis & Rheumatology in December, and will appear in print in February of next year.
Dr. Kolasinski reported no relevant financial disclosures. Dr. Neogi reported relationships with EMD Serono, Merck, and Pfizer.
ATLANTA – New guidelines for management of osteoarthritis of the hand, knee, and hip from the American College of Rheumatology and the Arthritis Foundation lay out a wide range of treatment options without an algorithm or hierarchy, making strong recommendations for nondrug interventions and for tailoring plans to individual patient-level factors.
Since the ACR last released OA management guidelines in 2012, a number of recommendations have been added, changed, and removed, and the structure of the guidelines has also changed. For instance, the new OA guidelines include a broad list of management options, Sharon L. Kolasinski, MD, chair of the ACR guidelines panel and professor of clinical medicine in the division of rheumatology at the University of Pennsylvania, Philadelphia, said in a presentation at the annual meeting of the American College of Rheumatology.
“The new guideline emphasizes comprehensive management of patients with OA, rather than a stepwise algorithm in a linear manner,” she said.
There is also no hierarchy to the recommendations, apart from the strength of the recommendation. “For any individual patient, a single option may be chosen at a particular time point, perhaps with or without other options, and may be reused in the future. For a given intervention, there might be a period of time over which it’s useful, and then the option might be changed,” Dr. Kolasinski noted.
Dr. Kolasinski advised making treatment decisions based on a patient’s disease severity, whether the patient uses medical devices, and in consideration of patient risk factors. “A history of injuries, surgical history, access to care, personal beliefs and preferences should all be brought to bear on decision making for osteoarthritis management,” she said.
The guidelines also advise considering a patient’s overall well-being and factors related to a patient’s perception of pain and function, such as mood disorders, altered sleep, chronic pain, impaired coping measures, and stress level. “Comprehensive management requires a broad assessment of how pain and function are affecting the patient with OA as a whole and recognizing that multiple options are available. They might be used in combination or change over time,” Dr. Kolasinski said.
The new guidelines place a strong emphasis on educational, behavioral, psychosocial, mind-body, and physical approaches. There are strong recommendations for the use of exercise, including aerobic, strengthening, neuromuscular, and aquatic exercise. Weight loss also carries a strong recommendation for patients with hip and knee OA, with a focus on group-based exercise, education, fitness and exercise goals, and a multidisciplinary approach using self-efficacy and self-management programs. The panels made a strong recommendation for tai chi to improve hip and knee OA. There are also strong recommendations for orthoses; aids and assistive devices such as canes, first carpometacarpal (CMC) orthoses, and tibiofemoral knee braces. Other interventions, such as Kinesio tape for first CMC joint and knee OA, hand orthoses, and patellofemoral knee braces, carried a conditional recommendation. Other conditional recommendations made by the panel were for acupuncture, thermal interventions, and radiofrequency ablation for patients with knee OA. Balance training for hip and knee OA, yoga for knee OA, and cognitive-behavioral therapy all were conditionally recommended by the panel.
The panel strongly recommended against the use of transcutaneous nerve stimulation for hip and knee OA, Dr. Kolasinski noted. The panel also conditionally recommended against use of modified shoes and pulsed vibration therapy in knee OA; lateral or medial wedged insoles, massage, and manual therapy with exercise in hip or knee OA; and iontophoresis in first CMC OA.
Tuhina Neogi, MD, PhD, chief of rheumatology at Boston University and member of the core team that developed the guidelines, said in her presentation the panel chose not to use the term “nonpharmacologic” in the guidelines because it may give patients a false impression that they are not receiving a treatment. “We really need to change our language and change the way in which we approach these conversations with our patients so that they don’t feel that they are not getting a treatment when we’re giving these recommendations,” she said.
Recommendations for, against pharmacologic approaches
The ACR has changed conditional recommendations for topical NSAIDs for knee and hand OA, oral NSAIDs, and intra-articular steroids for knee and hip OA into strong recommendations for the 2019 guidelines, Dr. Kolasinski said. While the 2012 guidelines conditionally recommended against topical capsaicin for knee OA, the new guidelines conditionally recommend it.
Other pharmacologic conditional recommendations included topical NSAIDs, chondroitin sulfate, and intra-articular corticosteroid injections for hand OA, acetaminophen, and duloxetine for knee OA.
With the new recommendations come changes that some rheumatologists and health care providers may find controversial. “I think that the practicing rheumatologist may be surprised that we have a recommendation against the use of hyaluronic acid in the knee as a conditional recommendation,” Dr. Kolasinski said. “The assessment of the literature at this point really reveals that there is equivalence between intra-articular hyaluronic acid injection and intra-articular saline injection, and so it was the feeling of the panel that, really, this was worth changing the recommendation from the 2012 guideline.”
The panel made strong recommendations against use of the following pharmacologic interventions:
- Bisphosphonates.
- Glucosamine sulfate.
- Combination glucosamine sulfate-chondroitin sulfate products.
- Hydroxychloroquine.
- Methotrexate.
- Intra-articular hyaluronic acid injections in hip OA.
- Chondroitin sulfate, platelet-rich plasma injections, and stem cell injections in hip and knee OA.
- Tumor necrosis factor (TNF) inhibitors.
- Interleukin-1–receptor antagonists.
Additionally, the panel made a conditional recommendation against topical capsaicin on the hand, colchicine, fish oil, vitamin D, intra-articular hyaluronic acid injections in the first CMC, and intra-articular botulinum toxin and prolotherapy in hip and knee OA.
The panel did not recommend for or against use of yoga for hip and hand OA, topical lidocaine, pregabalin, gabapentin, selective serotonin reuptake inhibitors, serotonin norepinephrine reuptake inhibitors apart from duloxetine, tricyclic antidepressants, and anti-nerve growth factor agents.
While the panel conditionally recommended against use of opioids, they made a conditional recommendation for use of tramadol opioids, and there was “a heated discussion about that distinction,” Dr. Neogi noted in a discussion session at the meeting. “There was a recent observational study that indicated that tramadol may have an increased risk of [all-cause] mortality, but there are lots of issues of confounding by indication in that study.”
The patient panel also raised strong concerns about the ACR and the Arthritis Foundation coming out against opioids for OA management in their guidelines. “They don’t want to damn opioids, but they’re also concerned about a specialty society coming out strongly against opioids in the concern that their physicians may limit their access to opioids if they’re in a situation where nothing else is helping them,” Dr. Neogi said.
Dr. Kolasinski noted the guidelines will be published online in Arthritis & Rheumatology in December, and will appear in print in February of next year.
Dr. Kolasinski reported no relevant financial disclosures. Dr. Neogi reported relationships with EMD Serono, Merck, and Pfizer.
ATLANTA – New guidelines for management of osteoarthritis of the hand, knee, and hip from the American College of Rheumatology and the Arthritis Foundation lay out a wide range of treatment options without an algorithm or hierarchy, making strong recommendations for nondrug interventions and for tailoring plans to individual patient-level factors.
Since the ACR last released OA management guidelines in 2012, a number of recommendations have been added, changed, and removed, and the structure of the guidelines has also changed. For instance, the new OA guidelines include a broad list of management options, Sharon L. Kolasinski, MD, chair of the ACR guidelines panel and professor of clinical medicine in the division of rheumatology at the University of Pennsylvania, Philadelphia, said in a presentation at the annual meeting of the American College of Rheumatology.
“The new guideline emphasizes comprehensive management of patients with OA, rather than a stepwise algorithm in a linear manner,” she said.
There is also no hierarchy to the recommendations, apart from the strength of the recommendation. “For any individual patient, a single option may be chosen at a particular time point, perhaps with or without other options, and may be reused in the future. For a given intervention, there might be a period of time over which it’s useful, and then the option might be changed,” Dr. Kolasinski noted.
Dr. Kolasinski advised making treatment decisions based on a patient’s disease severity, whether the patient uses medical devices, and in consideration of patient risk factors. “A history of injuries, surgical history, access to care, personal beliefs and preferences should all be brought to bear on decision making for osteoarthritis management,” she said.
The guidelines also advise considering a patient’s overall well-being and factors related to a patient’s perception of pain and function, such as mood disorders, altered sleep, chronic pain, impaired coping measures, and stress level. “Comprehensive management requires a broad assessment of how pain and function are affecting the patient with OA as a whole and recognizing that multiple options are available. They might be used in combination or change over time,” Dr. Kolasinski said.
The new guidelines place a strong emphasis on educational, behavioral, psychosocial, mind-body, and physical approaches. There are strong recommendations for the use of exercise, including aerobic, strengthening, neuromuscular, and aquatic exercise. Weight loss also carries a strong recommendation for patients with hip and knee OA, with a focus on group-based exercise, education, fitness and exercise goals, and a multidisciplinary approach using self-efficacy and self-management programs. The panels made a strong recommendation for tai chi to improve hip and knee OA. There are also strong recommendations for orthoses; aids and assistive devices such as canes, first carpometacarpal (CMC) orthoses, and tibiofemoral knee braces. Other interventions, such as Kinesio tape for first CMC joint and knee OA, hand orthoses, and patellofemoral knee braces, carried a conditional recommendation. Other conditional recommendations made by the panel were for acupuncture, thermal interventions, and radiofrequency ablation for patients with knee OA. Balance training for hip and knee OA, yoga for knee OA, and cognitive-behavioral therapy all were conditionally recommended by the panel.
The panel strongly recommended against the use of transcutaneous nerve stimulation for hip and knee OA, Dr. Kolasinski noted. The panel also conditionally recommended against use of modified shoes and pulsed vibration therapy in knee OA; lateral or medial wedged insoles, massage, and manual therapy with exercise in hip or knee OA; and iontophoresis in first CMC OA.
Tuhina Neogi, MD, PhD, chief of rheumatology at Boston University and member of the core team that developed the guidelines, said in her presentation the panel chose not to use the term “nonpharmacologic” in the guidelines because it may give patients a false impression that they are not receiving a treatment. “We really need to change our language and change the way in which we approach these conversations with our patients so that they don’t feel that they are not getting a treatment when we’re giving these recommendations,” she said.
Recommendations for, against pharmacologic approaches
The ACR has changed conditional recommendations for topical NSAIDs for knee and hand OA, oral NSAIDs, and intra-articular steroids for knee and hip OA into strong recommendations for the 2019 guidelines, Dr. Kolasinski said. While the 2012 guidelines conditionally recommended against topical capsaicin for knee OA, the new guidelines conditionally recommend it.
Other pharmacologic conditional recommendations included topical NSAIDs, chondroitin sulfate, and intra-articular corticosteroid injections for hand OA, acetaminophen, and duloxetine for knee OA.
With the new recommendations come changes that some rheumatologists and health care providers may find controversial. “I think that the practicing rheumatologist may be surprised that we have a recommendation against the use of hyaluronic acid in the knee as a conditional recommendation,” Dr. Kolasinski said. “The assessment of the literature at this point really reveals that there is equivalence between intra-articular hyaluronic acid injection and intra-articular saline injection, and so it was the feeling of the panel that, really, this was worth changing the recommendation from the 2012 guideline.”
The panel made strong recommendations against use of the following pharmacologic interventions:
- Bisphosphonates.
- Glucosamine sulfate.
- Combination glucosamine sulfate-chondroitin sulfate products.
- Hydroxychloroquine.
- Methotrexate.
- Intra-articular hyaluronic acid injections in hip OA.
- Chondroitin sulfate, platelet-rich plasma injections, and stem cell injections in hip and knee OA.
- Tumor necrosis factor (TNF) inhibitors.
- Interleukin-1–receptor antagonists.
Additionally, the panel made a conditional recommendation against topical capsaicin on the hand, colchicine, fish oil, vitamin D, intra-articular hyaluronic acid injections in the first CMC, and intra-articular botulinum toxin and prolotherapy in hip and knee OA.
The panel did not recommend for or against use of yoga for hip and hand OA, topical lidocaine, pregabalin, gabapentin, selective serotonin reuptake inhibitors, serotonin norepinephrine reuptake inhibitors apart from duloxetine, tricyclic antidepressants, and anti-nerve growth factor agents.
While the panel conditionally recommended against use of opioids, they made a conditional recommendation for use of tramadol opioids, and there was “a heated discussion about that distinction,” Dr. Neogi noted in a discussion session at the meeting. “There was a recent observational study that indicated that tramadol may have an increased risk of [all-cause] mortality, but there are lots of issues of confounding by indication in that study.”
The patient panel also raised strong concerns about the ACR and the Arthritis Foundation coming out against opioids for OA management in their guidelines. “They don’t want to damn opioids, but they’re also concerned about a specialty society coming out strongly against opioids in the concern that their physicians may limit their access to opioids if they’re in a situation where nothing else is helping them,” Dr. Neogi said.
Dr. Kolasinski noted the guidelines will be published online in Arthritis & Rheumatology in December, and will appear in print in February of next year.
Dr. Kolasinski reported no relevant financial disclosures. Dr. Neogi reported relationships with EMD Serono, Merck, and Pfizer.
REPORTING FROM ACR 2019
Large population-based study underscores link between gout, CVD event risk
ATLANTA – Gout is associated with an increased risk of both fatal and nonfatal cardiovascular disease events, according to a large population-based health data linkage study in New Zealand.
“Overall, the survival was quite good within both cohorts, but ... there is a clear and statistically significant difference in the survival between the people with gout and those without gout,” Ken Cai, MBBS, reported at the annual meeting of the American College of Rheumatology, noting that a similarly “significant and clear” difference was seen in nonfatal CVD events between the groups.
Of 968,387 individuals included in the analysis, 34,056 had gout, said Dr. Cai, a rheumatology clinical fellow at the University of Auckland (New Zealand). After adjusting for population-level estimated 5-year CVD risk for cardiovascular death, nonfatal myocardial infarction, stroke, or other vascular event, the adjusted hazard ratios were 1.20 for fatal and 1.32 for nonfatal first CVD events in patients with gout. The CVD risk score used in the analysis accounted for age, gender, ethnicity, level of social deprivation, diabetes status, previous hospitalization for atrial fibrillation, and baseline dispensing of blood pressure–lowering, lipid-lowering, and antiplatelet/anticoagulant medications.
“To allow for any other differences between the gout and nongout cohorts with respect to gender, age, ethnicity, and social deprivation, we further adjusted for these factors again, even though they had been accounted for within our CVD risk score,” he said, noting that “gout continued to demonstrate an increased adjusted hazard ratio” for fatal and nonfatal events after that adjustment (HRs, 1.40 and 1.35, respectively)
Additional analysis in the gout patients showed that CVD risk was similarly increased both in those who had been dispensed allopurinol at least once in the prior 5 years and those who had not (adjusted HRs for fatal events, 1.41 and 1.33; and for nonfatal, first CVD events, 1.34 and 1.38, respectively), and “there was no significant difference between these two groups, compared to people without gout,” he said.
Adjustment for serum urate levels in gout patients also showed similarly increased risk for fatal and nonfatal events for those with levels less than 6 mg/dL and those with levels of 6 mg/dL or greater (adjusted HRs of 1.32 and 1.42 for fatal events, and 1.27 and 1.43 for nonfatal first CVD events, respectively).
Again, no significant difference was seen in the risk of events between these two groups and those without gout, Dr. Cai said, noting that patients with no serum urate monitoring also had an increased risk of events (adjusted HR of 1.41 for fatal events and 1.29 for nonfatal, first CVD events).
Gout and hyperuricemia have previously been reported to be independent risk factors for CVD and CVD events, and urate-lowering therapy such as allopurinol have been thought to potentially be associated with reduced risk of CVD, he said, noting that the relationships are of particular concern in New Zealand, where gout affects more than 4% of the adult population.
“Maori, who are the indigenous people of New Zealand, and Pasifika people are disproportionately affected by gout; 8.5% of Maori, and 13.9% of Pasifika adults have gout,” he said, adding that an estimated one-third of Maori and Pasifika adults over age 65 years have gout.
To further assess the relationships between gout and CVD risk, he and his colleagues used validated population-level risk-prediction equations and linked National Health Identifier (NHI) data, he said.
National registries of medicines dispensing data, hospitalization, and death were linked to the Auckland/Northland regional repository of laboratory results from Jan. 1, 2012 to Dec. 31, 2016.
“We included all New Zealand residents aged 20 years or older who were in contact with publicly funded services in 2011 and were alive at the end of December, 2011,” he said, adding that those with a previous hospitalization for CVD or heart failure prior to the end of December 2011 were excluded, as were those with primary residence outside of the region for the prior 3 years and those missing predictor variable data.
Although the findings are limited by an inability to adjust for smoking status, body mass index, and blood pressure – as such data are not collected at the national level, and by the population-based nature of the study, which does not allow determination about causation, they nevertheless reinforce the association between gout and an increased estimated risk of CVD events, Dr. Cai said.
“Even after adjustment for estimated 5-year CVD risk and the additional weighting of risk factors within it, gout independently increased the hazard ratio for fatal and nonfatal events,” he said. “In our study, this effect was not ameliorated by allopurinol use or serum urate lowering to treatment target.”
Similar studies are needed in other populations, he said.
Dr. Cai reported grant support from Arthritis Australia.
SOURCE: Cai K et al. Arthritis Rheumatol. 2019;71(suppl 10), Abstract 2732.
ATLANTA – Gout is associated with an increased risk of both fatal and nonfatal cardiovascular disease events, according to a large population-based health data linkage study in New Zealand.
“Overall, the survival was quite good within both cohorts, but ... there is a clear and statistically significant difference in the survival between the people with gout and those without gout,” Ken Cai, MBBS, reported at the annual meeting of the American College of Rheumatology, noting that a similarly “significant and clear” difference was seen in nonfatal CVD events between the groups.
Of 968,387 individuals included in the analysis, 34,056 had gout, said Dr. Cai, a rheumatology clinical fellow at the University of Auckland (New Zealand). After adjusting for population-level estimated 5-year CVD risk for cardiovascular death, nonfatal myocardial infarction, stroke, or other vascular event, the adjusted hazard ratios were 1.20 for fatal and 1.32 for nonfatal first CVD events in patients with gout. The CVD risk score used in the analysis accounted for age, gender, ethnicity, level of social deprivation, diabetes status, previous hospitalization for atrial fibrillation, and baseline dispensing of blood pressure–lowering, lipid-lowering, and antiplatelet/anticoagulant medications.
“To allow for any other differences between the gout and nongout cohorts with respect to gender, age, ethnicity, and social deprivation, we further adjusted for these factors again, even though they had been accounted for within our CVD risk score,” he said, noting that “gout continued to demonstrate an increased adjusted hazard ratio” for fatal and nonfatal events after that adjustment (HRs, 1.40 and 1.35, respectively)
Additional analysis in the gout patients showed that CVD risk was similarly increased both in those who had been dispensed allopurinol at least once in the prior 5 years and those who had not (adjusted HRs for fatal events, 1.41 and 1.33; and for nonfatal, first CVD events, 1.34 and 1.38, respectively), and “there was no significant difference between these two groups, compared to people without gout,” he said.
Adjustment for serum urate levels in gout patients also showed similarly increased risk for fatal and nonfatal events for those with levels less than 6 mg/dL and those with levels of 6 mg/dL or greater (adjusted HRs of 1.32 and 1.42 for fatal events, and 1.27 and 1.43 for nonfatal first CVD events, respectively).
Again, no significant difference was seen in the risk of events between these two groups and those without gout, Dr. Cai said, noting that patients with no serum urate monitoring also had an increased risk of events (adjusted HR of 1.41 for fatal events and 1.29 for nonfatal, first CVD events).
Gout and hyperuricemia have previously been reported to be independent risk factors for CVD and CVD events, and urate-lowering therapy such as allopurinol have been thought to potentially be associated with reduced risk of CVD, he said, noting that the relationships are of particular concern in New Zealand, where gout affects more than 4% of the adult population.
“Maori, who are the indigenous people of New Zealand, and Pasifika people are disproportionately affected by gout; 8.5% of Maori, and 13.9% of Pasifika adults have gout,” he said, adding that an estimated one-third of Maori and Pasifika adults over age 65 years have gout.
To further assess the relationships between gout and CVD risk, he and his colleagues used validated population-level risk-prediction equations and linked National Health Identifier (NHI) data, he said.
National registries of medicines dispensing data, hospitalization, and death were linked to the Auckland/Northland regional repository of laboratory results from Jan. 1, 2012 to Dec. 31, 2016.
“We included all New Zealand residents aged 20 years or older who were in contact with publicly funded services in 2011 and were alive at the end of December, 2011,” he said, adding that those with a previous hospitalization for CVD or heart failure prior to the end of December 2011 were excluded, as were those with primary residence outside of the region for the prior 3 years and those missing predictor variable data.
Although the findings are limited by an inability to adjust for smoking status, body mass index, and blood pressure – as such data are not collected at the national level, and by the population-based nature of the study, which does not allow determination about causation, they nevertheless reinforce the association between gout and an increased estimated risk of CVD events, Dr. Cai said.
“Even after adjustment for estimated 5-year CVD risk and the additional weighting of risk factors within it, gout independently increased the hazard ratio for fatal and nonfatal events,” he said. “In our study, this effect was not ameliorated by allopurinol use or serum urate lowering to treatment target.”
Similar studies are needed in other populations, he said.
Dr. Cai reported grant support from Arthritis Australia.
SOURCE: Cai K et al. Arthritis Rheumatol. 2019;71(suppl 10), Abstract 2732.
ATLANTA – Gout is associated with an increased risk of both fatal and nonfatal cardiovascular disease events, according to a large population-based health data linkage study in New Zealand.
“Overall, the survival was quite good within both cohorts, but ... there is a clear and statistically significant difference in the survival between the people with gout and those without gout,” Ken Cai, MBBS, reported at the annual meeting of the American College of Rheumatology, noting that a similarly “significant and clear” difference was seen in nonfatal CVD events between the groups.
Of 968,387 individuals included in the analysis, 34,056 had gout, said Dr. Cai, a rheumatology clinical fellow at the University of Auckland (New Zealand). After adjusting for population-level estimated 5-year CVD risk for cardiovascular death, nonfatal myocardial infarction, stroke, or other vascular event, the adjusted hazard ratios were 1.20 for fatal and 1.32 for nonfatal first CVD events in patients with gout. The CVD risk score used in the analysis accounted for age, gender, ethnicity, level of social deprivation, diabetes status, previous hospitalization for atrial fibrillation, and baseline dispensing of blood pressure–lowering, lipid-lowering, and antiplatelet/anticoagulant medications.
“To allow for any other differences between the gout and nongout cohorts with respect to gender, age, ethnicity, and social deprivation, we further adjusted for these factors again, even though they had been accounted for within our CVD risk score,” he said, noting that “gout continued to demonstrate an increased adjusted hazard ratio” for fatal and nonfatal events after that adjustment (HRs, 1.40 and 1.35, respectively)
Additional analysis in the gout patients showed that CVD risk was similarly increased both in those who had been dispensed allopurinol at least once in the prior 5 years and those who had not (adjusted HRs for fatal events, 1.41 and 1.33; and for nonfatal, first CVD events, 1.34 and 1.38, respectively), and “there was no significant difference between these two groups, compared to people without gout,” he said.
Adjustment for serum urate levels in gout patients also showed similarly increased risk for fatal and nonfatal events for those with levels less than 6 mg/dL and those with levels of 6 mg/dL or greater (adjusted HRs of 1.32 and 1.42 for fatal events, and 1.27 and 1.43 for nonfatal first CVD events, respectively).
Again, no significant difference was seen in the risk of events between these two groups and those without gout, Dr. Cai said, noting that patients with no serum urate monitoring also had an increased risk of events (adjusted HR of 1.41 for fatal events and 1.29 for nonfatal, first CVD events).
Gout and hyperuricemia have previously been reported to be independent risk factors for CVD and CVD events, and urate-lowering therapy such as allopurinol have been thought to potentially be associated with reduced risk of CVD, he said, noting that the relationships are of particular concern in New Zealand, where gout affects more than 4% of the adult population.
“Maori, who are the indigenous people of New Zealand, and Pasifika people are disproportionately affected by gout; 8.5% of Maori, and 13.9% of Pasifika adults have gout,” he said, adding that an estimated one-third of Maori and Pasifika adults over age 65 years have gout.
To further assess the relationships between gout and CVD risk, he and his colleagues used validated population-level risk-prediction equations and linked National Health Identifier (NHI) data, he said.
National registries of medicines dispensing data, hospitalization, and death were linked to the Auckland/Northland regional repository of laboratory results from Jan. 1, 2012 to Dec. 31, 2016.
“We included all New Zealand residents aged 20 years or older who were in contact with publicly funded services in 2011 and were alive at the end of December, 2011,” he said, adding that those with a previous hospitalization for CVD or heart failure prior to the end of December 2011 were excluded, as were those with primary residence outside of the region for the prior 3 years and those missing predictor variable data.
Although the findings are limited by an inability to adjust for smoking status, body mass index, and blood pressure – as such data are not collected at the national level, and by the population-based nature of the study, which does not allow determination about causation, they nevertheless reinforce the association between gout and an increased estimated risk of CVD events, Dr. Cai said.
“Even after adjustment for estimated 5-year CVD risk and the additional weighting of risk factors within it, gout independently increased the hazard ratio for fatal and nonfatal events,” he said. “In our study, this effect was not ameliorated by allopurinol use or serum urate lowering to treatment target.”
Similar studies are needed in other populations, he said.
Dr. Cai reported grant support from Arthritis Australia.
SOURCE: Cai K et al. Arthritis Rheumatol. 2019;71(suppl 10), Abstract 2732.
REPORTING FROM ACR 2019
Baseline subtypes predict dementia and death in patients with Parkinson’s disease
ST. LOUIS – , according to the results of a longitudinal study of 162 patients at Washington University, St. Louis.
Parkinson’s disease is often only staged as mild, moderate, or severe, and sometimes cognitive and psychiatric symptoms are not assessed. The St. Louis team wanted to see if there are actual subtypes, and if they have clinical relevance, said lead investigator Peter Myers, PhD, a postdoctoral research associate at Washington University.
The magnitude of the findings was “surprising. ... We don’t think we are seeing stages” because the different symptom patterns were apparent at baseline. Instead, “we are seeing really distinct clinical subtypes that could inform patient prognosis and help prepare families and caregivers. It is important that you assess more than just motor symptoms,” he said at the annual meeting of the American Neurological Association.
Some of the subjects were newly diagnosed and others diagnosed years earlier. At baseline, they had symptoms for an average of 6 years, and none had dementia.
After a battery of cognitive, psychiatric, and movement tests, Dr. Myers and his colleagues found that their subjects fell into three patterns: motor symptoms only, with normal cognitive and psychiatric performance (63 subjects); motor symptoms plus prominent anxiety or depression (17); and motor symptoms plus cognitive deficits (82).
A total of 42 patients developed dementia – a score of at least 1 on the Clinical Dementia Rating evaluation – including three in the motor-only group (5%), two in the psychiatric/motor group (12%), and 37 in the cognitive/motor group (45%).
After controlling for age, sex, and symptom duration, the risk of dementia conversion was far higher in the cognitive/motor group (relative risk, 4.16, 95% confidence interval, 1.18-14.65) and psychiatric/motor group (RR, 3.08; 95% CI, 0.72-13.28), compared with motor-only subjects.
Thirty-eight patients died, the majority because of Parkinson’s disease-related causes, including five in the motor-only group (8%), two in the psychiatric/motor group (12%), and 31 in the cognitive/motor group (38%).
The risk of death, relative to motor-only patients, was higher with both the cognitive/motor (RR, 4.06; 95% CI, 1.37-12.03) and psychiatric/motor subtypes (RR, 4.17; 95% CI, 0.70-24.91).
It is unclear what leads to the progression differences, but the researchers’ hypothesis is that the broader scope of symptoms indicates a greater extent of brain pathology. The St. Louis team is looking at brain-imaging data to see if that is true, and if the subtypes can be distinguished on imaging. They are also interested in seeing if genetics plays a role in susceptibility to the different subtypes, and if the baseline subtypes are stable over time or if patients convert between them.
Subjects were, on average, 66 years old, and 62% were men. The mean levodopa-equivalent daily dose was 613 mg in the motor-only group, 1,004 mg in the psychiatric/motor group, and 783 mg in the cognitive/motor group (P = .03). Mean symptom duration was shorter in motor-only patients (5.1 years) versus the psychiatric/motor group (7.2 years) and cognitive/motor group (6.9 years, P less than .01).
Although relative risks crossed 1 in the psychiatric/motor group, it was probably because there were only 17 patients. “We do think there is a very strong likelihood that” the psychiatric/motor findings, like the cognitive/motor findings, “are valid,” Dr. Myer said.
“I think [the findings] could be real,” said Clair Henchcliffe, MD, DPhil, vice chair for clinical research in the department of neurology at Weill Cornell Medical College in New York.
“Other publications have shown that people who have cognitive symptoms at onset are more likely to go on to develop dementia down the line. This is much in line with that,” she said when asked for comment.
The field has “always been interested in finding data that help us personalize treatment, and a lot of people are looking to subtype Parkinson’s disease to help us plan with our patients.” It could also help with frequency of follow-up, trial participation, and maybe someday treatment selection. “We are always thinking a few years ahead” with Parkinson’s disease, she said.
There was no industry funding, and the investigators did not have any relevant disclosures.
ST. LOUIS – , according to the results of a longitudinal study of 162 patients at Washington University, St. Louis.
Parkinson’s disease is often only staged as mild, moderate, or severe, and sometimes cognitive and psychiatric symptoms are not assessed. The St. Louis team wanted to see if there are actual subtypes, and if they have clinical relevance, said lead investigator Peter Myers, PhD, a postdoctoral research associate at Washington University.
The magnitude of the findings was “surprising. ... We don’t think we are seeing stages” because the different symptom patterns were apparent at baseline. Instead, “we are seeing really distinct clinical subtypes that could inform patient prognosis and help prepare families and caregivers. It is important that you assess more than just motor symptoms,” he said at the annual meeting of the American Neurological Association.
Some of the subjects were newly diagnosed and others diagnosed years earlier. At baseline, they had symptoms for an average of 6 years, and none had dementia.
After a battery of cognitive, psychiatric, and movement tests, Dr. Myers and his colleagues found that their subjects fell into three patterns: motor symptoms only, with normal cognitive and psychiatric performance (63 subjects); motor symptoms plus prominent anxiety or depression (17); and motor symptoms plus cognitive deficits (82).
A total of 42 patients developed dementia – a score of at least 1 on the Clinical Dementia Rating evaluation – including three in the motor-only group (5%), two in the psychiatric/motor group (12%), and 37 in the cognitive/motor group (45%).
After controlling for age, sex, and symptom duration, the risk of dementia conversion was far higher in the cognitive/motor group (relative risk, 4.16, 95% confidence interval, 1.18-14.65) and psychiatric/motor group (RR, 3.08; 95% CI, 0.72-13.28), compared with motor-only subjects.
Thirty-eight patients died, the majority because of Parkinson’s disease-related causes, including five in the motor-only group (8%), two in the psychiatric/motor group (12%), and 31 in the cognitive/motor group (38%).
The risk of death, relative to motor-only patients, was higher with both the cognitive/motor (RR, 4.06; 95% CI, 1.37-12.03) and psychiatric/motor subtypes (RR, 4.17; 95% CI, 0.70-24.91).
It is unclear what leads to the progression differences, but the researchers’ hypothesis is that the broader scope of symptoms indicates a greater extent of brain pathology. The St. Louis team is looking at brain-imaging data to see if that is true, and if the subtypes can be distinguished on imaging. They are also interested in seeing if genetics plays a role in susceptibility to the different subtypes, and if the baseline subtypes are stable over time or if patients convert between them.
Subjects were, on average, 66 years old, and 62% were men. The mean levodopa-equivalent daily dose was 613 mg in the motor-only group, 1,004 mg in the psychiatric/motor group, and 783 mg in the cognitive/motor group (P = .03). Mean symptom duration was shorter in motor-only patients (5.1 years) versus the psychiatric/motor group (7.2 years) and cognitive/motor group (6.9 years, P less than .01).
Although relative risks crossed 1 in the psychiatric/motor group, it was probably because there were only 17 patients. “We do think there is a very strong likelihood that” the psychiatric/motor findings, like the cognitive/motor findings, “are valid,” Dr. Myer said.
“I think [the findings] could be real,” said Clair Henchcliffe, MD, DPhil, vice chair for clinical research in the department of neurology at Weill Cornell Medical College in New York.
“Other publications have shown that people who have cognitive symptoms at onset are more likely to go on to develop dementia down the line. This is much in line with that,” she said when asked for comment.
The field has “always been interested in finding data that help us personalize treatment, and a lot of people are looking to subtype Parkinson’s disease to help us plan with our patients.” It could also help with frequency of follow-up, trial participation, and maybe someday treatment selection. “We are always thinking a few years ahead” with Parkinson’s disease, she said.
There was no industry funding, and the investigators did not have any relevant disclosures.
ST. LOUIS – , according to the results of a longitudinal study of 162 patients at Washington University, St. Louis.
Parkinson’s disease is often only staged as mild, moderate, or severe, and sometimes cognitive and psychiatric symptoms are not assessed. The St. Louis team wanted to see if there are actual subtypes, and if they have clinical relevance, said lead investigator Peter Myers, PhD, a postdoctoral research associate at Washington University.
The magnitude of the findings was “surprising. ... We don’t think we are seeing stages” because the different symptom patterns were apparent at baseline. Instead, “we are seeing really distinct clinical subtypes that could inform patient prognosis and help prepare families and caregivers. It is important that you assess more than just motor symptoms,” he said at the annual meeting of the American Neurological Association.
Some of the subjects were newly diagnosed and others diagnosed years earlier. At baseline, they had symptoms for an average of 6 years, and none had dementia.
After a battery of cognitive, psychiatric, and movement tests, Dr. Myers and his colleagues found that their subjects fell into three patterns: motor symptoms only, with normal cognitive and psychiatric performance (63 subjects); motor symptoms plus prominent anxiety or depression (17); and motor symptoms plus cognitive deficits (82).
A total of 42 patients developed dementia – a score of at least 1 on the Clinical Dementia Rating evaluation – including three in the motor-only group (5%), two in the psychiatric/motor group (12%), and 37 in the cognitive/motor group (45%).
After controlling for age, sex, and symptom duration, the risk of dementia conversion was far higher in the cognitive/motor group (relative risk, 4.16, 95% confidence interval, 1.18-14.65) and psychiatric/motor group (RR, 3.08; 95% CI, 0.72-13.28), compared with motor-only subjects.
Thirty-eight patients died, the majority because of Parkinson’s disease-related causes, including five in the motor-only group (8%), two in the psychiatric/motor group (12%), and 31 in the cognitive/motor group (38%).
The risk of death, relative to motor-only patients, was higher with both the cognitive/motor (RR, 4.06; 95% CI, 1.37-12.03) and psychiatric/motor subtypes (RR, 4.17; 95% CI, 0.70-24.91).
It is unclear what leads to the progression differences, but the researchers’ hypothesis is that the broader scope of symptoms indicates a greater extent of brain pathology. The St. Louis team is looking at brain-imaging data to see if that is true, and if the subtypes can be distinguished on imaging. They are also interested in seeing if genetics plays a role in susceptibility to the different subtypes, and if the baseline subtypes are stable over time or if patients convert between them.
Subjects were, on average, 66 years old, and 62% were men. The mean levodopa-equivalent daily dose was 613 mg in the motor-only group, 1,004 mg in the psychiatric/motor group, and 783 mg in the cognitive/motor group (P = .03). Mean symptom duration was shorter in motor-only patients (5.1 years) versus the psychiatric/motor group (7.2 years) and cognitive/motor group (6.9 years, P less than .01).
Although relative risks crossed 1 in the psychiatric/motor group, it was probably because there were only 17 patients. “We do think there is a very strong likelihood that” the psychiatric/motor findings, like the cognitive/motor findings, “are valid,” Dr. Myer said.
“I think [the findings] could be real,” said Clair Henchcliffe, MD, DPhil, vice chair for clinical research in the department of neurology at Weill Cornell Medical College in New York.
“Other publications have shown that people who have cognitive symptoms at onset are more likely to go on to develop dementia down the line. This is much in line with that,” she said when asked for comment.
The field has “always been interested in finding data that help us personalize treatment, and a lot of people are looking to subtype Parkinson’s disease to help us plan with our patients.” It could also help with frequency of follow-up, trial participation, and maybe someday treatment selection. “We are always thinking a few years ahead” with Parkinson’s disease, she said.
There was no industry funding, and the investigators did not have any relevant disclosures.
REPORTING FROM ANA 2019
Differences in U.S. and European aneurysm guidelines called unavoidable
NEW YORK – Published 12 months apart, guidelines on management of abdominal aortic aneurysm (AAA) from the European Society for Vascular Surgery are similar but diverged in instructive ways from those of the Society for Vascular Surgery, according to a critical review at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation. “Some of the differences were almost unavoidable in the sense that the ESVS guidelines represent multiple idiosyncratic health care systems across Europe,” reported Ronald L. Dalman, MD, chief of vascular surgery, Stanford (Calif.) University.
As a result, the ESVS guidelines provide very little specificity about pharmacologic options because of the differences in availability of these treatments within specific health systems. In addition, both open and endovascular aneurysm repair (EVAR) are given similar emphasis because of the limited availability of EVAR in some parts of Europe.
“The ESVS guidelines specifically recommend repair of an aneurysm within 8 weeks when repair is indicated, but there are not many aneurysms that go 8 weeks in the U.S. without being fixed by a fee-for-service surgeon,” Dr. Dalman observed.
The SVS AAA guidelines were published in January 2018 (J Vasc Surg 2018;67:2-77) and the ESVS guidelines followed 1 year later (Eur J Vasc Surg 2019;57:8-93).
The differences in the guidelines, although modest, are interesting because each set of guidelines was based largely on the same set of trials and published studies, according to Dr. Dalman, who was a coauthor of the SVS guidelines and an external reviewer for the ESVS guidelines.
In the lag between completion of the two guidelines, new information led to three ESVS additions not found in the SVS guidelines, according to Dr. Dalman. They involved the importance of considering aneurysm diameter as a prognostic factor, new understanding of the limitations on endovascular aneurysm sealing (EVAS), and new information about how aneurysm size should affect frequency of surveillance.
Overall, the U.S. guidelines contain 111 recommendations based on 177 references, while the ESVS guidelines contain 125 guidelines based on 189 references. In retrospect, Dr. Dalman believes both sets of guidelines omitted some clinically meaningful information, such as the risk of large-diameter devices for causing endoleaks.
The authors of the ESVS guidelines did have an opportunity to review of a draft of the SVS guidelines, so differences can be interpreted as intentional. For example, the SVS guidelines recommend risk calculators, but Dr. Dalman suggested that the authors of the ESVS guidelines were less convinced that their utility was established.
The decision not to recommend a door-to-treatment time for ruptured aneurysms, as in the SVS recommendations, might have been in deference to disparate practice across European countries, Dr. Dalman suggested.
Ultimately, the guidelines are “substantially similar,” according to Dr. Dalman, but he expressed concerned that neither guideline is accompanied by a specific mechanism or recommended strategy to ensure implementation.
Many of the SVS recommendations are likely to be translated into quality metrics at U.S. institutions, but “there are implementation issues” for ensuring that each guideline is applied, Dr. Dalman said.
Given the agreement on the vast majority of the recommendations, Dr. Dalman suggested that “it might be time to consider global guidelines” for management of AAA and other vascular diseases. Some type of language might be required to accommodate divergent resources or practices across borders, but Dr. Dalman questioned the need to review the same literature to arrive at mostly the same conclusions.
NEW YORK – Published 12 months apart, guidelines on management of abdominal aortic aneurysm (AAA) from the European Society for Vascular Surgery are similar but diverged in instructive ways from those of the Society for Vascular Surgery, according to a critical review at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation. “Some of the differences were almost unavoidable in the sense that the ESVS guidelines represent multiple idiosyncratic health care systems across Europe,” reported Ronald L. Dalman, MD, chief of vascular surgery, Stanford (Calif.) University.
As a result, the ESVS guidelines provide very little specificity about pharmacologic options because of the differences in availability of these treatments within specific health systems. In addition, both open and endovascular aneurysm repair (EVAR) are given similar emphasis because of the limited availability of EVAR in some parts of Europe.
“The ESVS guidelines specifically recommend repair of an aneurysm within 8 weeks when repair is indicated, but there are not many aneurysms that go 8 weeks in the U.S. without being fixed by a fee-for-service surgeon,” Dr. Dalman observed.
The SVS AAA guidelines were published in January 2018 (J Vasc Surg 2018;67:2-77) and the ESVS guidelines followed 1 year later (Eur J Vasc Surg 2019;57:8-93).
The differences in the guidelines, although modest, are interesting because each set of guidelines was based largely on the same set of trials and published studies, according to Dr. Dalman, who was a coauthor of the SVS guidelines and an external reviewer for the ESVS guidelines.
In the lag between completion of the two guidelines, new information led to three ESVS additions not found in the SVS guidelines, according to Dr. Dalman. They involved the importance of considering aneurysm diameter as a prognostic factor, new understanding of the limitations on endovascular aneurysm sealing (EVAS), and new information about how aneurysm size should affect frequency of surveillance.
Overall, the U.S. guidelines contain 111 recommendations based on 177 references, while the ESVS guidelines contain 125 guidelines based on 189 references. In retrospect, Dr. Dalman believes both sets of guidelines omitted some clinically meaningful information, such as the risk of large-diameter devices for causing endoleaks.
The authors of the ESVS guidelines did have an opportunity to review of a draft of the SVS guidelines, so differences can be interpreted as intentional. For example, the SVS guidelines recommend risk calculators, but Dr. Dalman suggested that the authors of the ESVS guidelines were less convinced that their utility was established.
The decision not to recommend a door-to-treatment time for ruptured aneurysms, as in the SVS recommendations, might have been in deference to disparate practice across European countries, Dr. Dalman suggested.
Ultimately, the guidelines are “substantially similar,” according to Dr. Dalman, but he expressed concerned that neither guideline is accompanied by a specific mechanism or recommended strategy to ensure implementation.
Many of the SVS recommendations are likely to be translated into quality metrics at U.S. institutions, but “there are implementation issues” for ensuring that each guideline is applied, Dr. Dalman said.
Given the agreement on the vast majority of the recommendations, Dr. Dalman suggested that “it might be time to consider global guidelines” for management of AAA and other vascular diseases. Some type of language might be required to accommodate divergent resources or practices across borders, but Dr. Dalman questioned the need to review the same literature to arrive at mostly the same conclusions.
NEW YORK – Published 12 months apart, guidelines on management of abdominal aortic aneurysm (AAA) from the European Society for Vascular Surgery are similar but diverged in instructive ways from those of the Society for Vascular Surgery, according to a critical review at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation. “Some of the differences were almost unavoidable in the sense that the ESVS guidelines represent multiple idiosyncratic health care systems across Europe,” reported Ronald L. Dalman, MD, chief of vascular surgery, Stanford (Calif.) University.
As a result, the ESVS guidelines provide very little specificity about pharmacologic options because of the differences in availability of these treatments within specific health systems. In addition, both open and endovascular aneurysm repair (EVAR) are given similar emphasis because of the limited availability of EVAR in some parts of Europe.
“The ESVS guidelines specifically recommend repair of an aneurysm within 8 weeks when repair is indicated, but there are not many aneurysms that go 8 weeks in the U.S. without being fixed by a fee-for-service surgeon,” Dr. Dalman observed.
The SVS AAA guidelines were published in January 2018 (J Vasc Surg 2018;67:2-77) and the ESVS guidelines followed 1 year later (Eur J Vasc Surg 2019;57:8-93).
The differences in the guidelines, although modest, are interesting because each set of guidelines was based largely on the same set of trials and published studies, according to Dr. Dalman, who was a coauthor of the SVS guidelines and an external reviewer for the ESVS guidelines.
In the lag between completion of the two guidelines, new information led to three ESVS additions not found in the SVS guidelines, according to Dr. Dalman. They involved the importance of considering aneurysm diameter as a prognostic factor, new understanding of the limitations on endovascular aneurysm sealing (EVAS), and new information about how aneurysm size should affect frequency of surveillance.
Overall, the U.S. guidelines contain 111 recommendations based on 177 references, while the ESVS guidelines contain 125 guidelines based on 189 references. In retrospect, Dr. Dalman believes both sets of guidelines omitted some clinically meaningful information, such as the risk of large-diameter devices for causing endoleaks.
The authors of the ESVS guidelines did have an opportunity to review of a draft of the SVS guidelines, so differences can be interpreted as intentional. For example, the SVS guidelines recommend risk calculators, but Dr. Dalman suggested that the authors of the ESVS guidelines were less convinced that their utility was established.
The decision not to recommend a door-to-treatment time for ruptured aneurysms, as in the SVS recommendations, might have been in deference to disparate practice across European countries, Dr. Dalman suggested.
Ultimately, the guidelines are “substantially similar,” according to Dr. Dalman, but he expressed concerned that neither guideline is accompanied by a specific mechanism or recommended strategy to ensure implementation.
Many of the SVS recommendations are likely to be translated into quality metrics at U.S. institutions, but “there are implementation issues” for ensuring that each guideline is applied, Dr. Dalman said.
Given the agreement on the vast majority of the recommendations, Dr. Dalman suggested that “it might be time to consider global guidelines” for management of AAA and other vascular diseases. Some type of language might be required to accommodate divergent resources or practices across borders, but Dr. Dalman questioned the need to review the same literature to arrive at mostly the same conclusions.
REPORTING FROM VEITHsymposium
Key clinical point:
Major finding: Less emphasis on endovascular repair and specific drugs in Europe reflects accommodation of nationalized health systems.
Study details: Expert review.
Disclosures: Dr. Dalman reports no potential financial conflicts of interest relevant to this topic.
Source: Dalman RL et al. 46th VEITHsymposium.
What’s new in hepatitis C: Four themes that dominated at the Liver Meeting
BOSTON – Treatment of persons who inject drugs, updates in pangenotypic direct-acting antiviral therapy, the benefits of sustained virologic response, and preemptive therapy in donor-positive organ transplantation topped the list of notable hepatitis C–related abstracts this year at the annual meeting of the American Association for the Study of Liver Diseases.
That’s according to Marc Ghany, MD, of the liver diseases branch of the National Institute of Diabetes and Digestive and Kidney Diseases at the National Institutes of Health, who gave a hepatitis C debrief to attendees on the final day of the meeting. Here are some of the meeting highlights as summarized by Dr. Ghany in this well-attended last-day session.
Treatment of HCV in people who inject drugs
Emerging data suggest it is feasible to treat hepatitis C virus (HCV) infection in persons who inject drugs (PWIDs); however, overcoming adherence issues remains a challenge, Dr. Ghany told attendees.
According to one study presented at AASLD by Dhiman and coauthors (Abstract 0165), decentralized care of PWIDs using direct-acting antiviral (DAA) therapy was safe and effective, even in those with cirrhosis. Authors demonstrated an “impressive” rate of sustained virologic response at 12 weeks (SVR12) of 91% by a modified intention-to-treat analysis, Dr. Ghany said; however, treatment interruptions were frequent and reduced the overall SVR rate in the study to 78%.
Other studies at the meeting looked at strategies to improve DAA efficacy in this population of patients at high risk of nonadherence, including use of a digital medicine program (Abstract 1554) and a model of care in which an internist-addiction medicine specialist evaluated opiate-dependent patients for HCV infection in a hepatology clinic (Abstract 1589).
Reinfection remains a focus of research in PWIDs. At this meeting, Janjua and coauthors reported that DAA-treated PWIDs in British Columbia had a threefold higher rate of reinfection versus non-PWIDs; however, there were no detected reinfections among PWIDs who had received uninterrupted opioid agonist therapy. “These data suggested that opioid agonist therapy should be given before and after HCV treatment in persons who inject drugs to prevent the infection,” Dr. Ghany said in his presentation.
Updates on pangenotypic DAA therapy
Jonas and coauthors (Abstract 1551) reported on the safety and efficacy of glecaprevir/pibrentasvir for 8 weeks in children with chronic HCV infection enrolled in the ongoing phase 2/3 DORA study. The SVR12 was high, according to Dr. Ghany, at 96% overall, and consistent across age cohorts from 3 to less than 12 years of age.
“In the near future, we should have a safe and effective regimen (approved) for children 3 years or older,” Dr. Ghany said. “I think this will serve us well, as we try to eliminate HCV in children, who number up to 5 million cases worldwide.”
A short course of glecaprevir/pibrentasvir is approved for patients with HCV and compensated cirrhosis, and data to support that was presented last year at The Liver Meeting; however, data were not presented on patients with genotype 3, the most difficult-to-treat genotype, Dr. Ghany said. That gap was filled at this year’s meeting with a report (Abstract LP9) showing SVR12 rates of 98.4% per protocol and 95.2% in intention-to-treat analysis.
Relationship of SVR to clinical outcomes
While the impact of sustained virologic response (SVR) on all-cause mortality is clear in patients with HCV, less is known about the effect of SVR on liver-related mortality and other outcomes, Dr. Ghany said. In one study presented here (Abstract 0039), based on analysis of a Veterans Affairs database of patients with chronic HCV infection, SVR was linked to a significant reduction in liver-related mortality, while in another report (Abstract 0037), SVR was associated with significant reductions in acute coronary syndromes, end-stage renal disease, and ischemic stroke.
Similarly, a multinational, propensity score–matched analysis (Abstract 0040) demonstrated that SVR had an impact on 5-year overall survival and liver-related survival in patients with HCV-related hepatocellular carcinoma (HCC). “For HCC patients who are candidates for HCC therapy, consideration should also be given to treating these individuals (with DAA therapy) because of the impact on overall survival,” Dr. Ghany said.
Preemptive DAA therapy in organ transplantation
Exciting new data show that preemptive therapy, given for short durations, appears to either prevent or cure HCV infection after organ transplant, said Dr. Ghany.
A retrospective analysis by Wijarnpreecha and colleagues (Abstract 0003) showed that 12 or 24 weeks of direct-acting antiviral (DAA) therapy resulted in an SVR12 for 24 out of 24 HCV-seropositive to HCV-seronegative liver transplants, while Durand and colleagues (Abstract 0042) showed that just 4 weeks of pre- and postexposure DAA prophylaxis resulted in SVR12s for 9 out of 9 HCV donor-positive, recipient-negative kidney transplants. Finally, Feld and coauthors (Abstract 0038) showed that preemptive ezetimibe with DAA therapy for 7 days prevented or rapidly cured infection in an experience that included 16 HCV-positive organ donors and 25 HCV-negative recipients.
“While these data are very encouraging, I think we do need to have long-term follow-up of these patients for graft survival, as well as the effect on wait times,” Dr. Ghany said.
Dr. Ghany reported no disclosures related to his presentation.
BOSTON – Treatment of persons who inject drugs, updates in pangenotypic direct-acting antiviral therapy, the benefits of sustained virologic response, and preemptive therapy in donor-positive organ transplantation topped the list of notable hepatitis C–related abstracts this year at the annual meeting of the American Association for the Study of Liver Diseases.
That’s according to Marc Ghany, MD, of the liver diseases branch of the National Institute of Diabetes and Digestive and Kidney Diseases at the National Institutes of Health, who gave a hepatitis C debrief to attendees on the final day of the meeting. Here are some of the meeting highlights as summarized by Dr. Ghany in this well-attended last-day session.
Treatment of HCV in people who inject drugs
Emerging data suggest it is feasible to treat hepatitis C virus (HCV) infection in persons who inject drugs (PWIDs); however, overcoming adherence issues remains a challenge, Dr. Ghany told attendees.
According to one study presented at AASLD by Dhiman and coauthors (Abstract 0165), decentralized care of PWIDs using direct-acting antiviral (DAA) therapy was safe and effective, even in those with cirrhosis. Authors demonstrated an “impressive” rate of sustained virologic response at 12 weeks (SVR12) of 91% by a modified intention-to-treat analysis, Dr. Ghany said; however, treatment interruptions were frequent and reduced the overall SVR rate in the study to 78%.
Other studies at the meeting looked at strategies to improve DAA efficacy in this population of patients at high risk of nonadherence, including use of a digital medicine program (Abstract 1554) and a model of care in which an internist-addiction medicine specialist evaluated opiate-dependent patients for HCV infection in a hepatology clinic (Abstract 1589).
Reinfection remains a focus of research in PWIDs. At this meeting, Janjua and coauthors reported that DAA-treated PWIDs in British Columbia had a threefold higher rate of reinfection versus non-PWIDs; however, there were no detected reinfections among PWIDs who had received uninterrupted opioid agonist therapy. “These data suggested that opioid agonist therapy should be given before and after HCV treatment in persons who inject drugs to prevent the infection,” Dr. Ghany said in his presentation.
Updates on pangenotypic DAA therapy
Jonas and coauthors (Abstract 1551) reported on the safety and efficacy of glecaprevir/pibrentasvir for 8 weeks in children with chronic HCV infection enrolled in the ongoing phase 2/3 DORA study. The SVR12 was high, according to Dr. Ghany, at 96% overall, and consistent across age cohorts from 3 to less than 12 years of age.
“In the near future, we should have a safe and effective regimen (approved) for children 3 years or older,” Dr. Ghany said. “I think this will serve us well, as we try to eliminate HCV in children, who number up to 5 million cases worldwide.”
A short course of glecaprevir/pibrentasvir is approved for patients with HCV and compensated cirrhosis, and data to support that was presented last year at The Liver Meeting; however, data were not presented on patients with genotype 3, the most difficult-to-treat genotype, Dr. Ghany said. That gap was filled at this year’s meeting with a report (Abstract LP9) showing SVR12 rates of 98.4% per protocol and 95.2% in intention-to-treat analysis.
Relationship of SVR to clinical outcomes
While the impact of sustained virologic response (SVR) on all-cause mortality is clear in patients with HCV, less is known about the effect of SVR on liver-related mortality and other outcomes, Dr. Ghany said. In one study presented here (Abstract 0039), based on analysis of a Veterans Affairs database of patients with chronic HCV infection, SVR was linked to a significant reduction in liver-related mortality, while in another report (Abstract 0037), SVR was associated with significant reductions in acute coronary syndromes, end-stage renal disease, and ischemic stroke.
Similarly, a multinational, propensity score–matched analysis (Abstract 0040) demonstrated that SVR had an impact on 5-year overall survival and liver-related survival in patients with HCV-related hepatocellular carcinoma (HCC). “For HCC patients who are candidates for HCC therapy, consideration should also be given to treating these individuals (with DAA therapy) because of the impact on overall survival,” Dr. Ghany said.
Preemptive DAA therapy in organ transplantation
Exciting new data show that preemptive therapy, given for short durations, appears to either prevent or cure HCV infection after organ transplant, said Dr. Ghany.
A retrospective analysis by Wijarnpreecha and colleagues (Abstract 0003) showed that 12 or 24 weeks of direct-acting antiviral (DAA) therapy resulted in an SVR12 for 24 out of 24 HCV-seropositive to HCV-seronegative liver transplants, while Durand and colleagues (Abstract 0042) showed that just 4 weeks of pre- and postexposure DAA prophylaxis resulted in SVR12s for 9 out of 9 HCV donor-positive, recipient-negative kidney transplants. Finally, Feld and coauthors (Abstract 0038) showed that preemptive ezetimibe with DAA therapy for 7 days prevented or rapidly cured infection in an experience that included 16 HCV-positive organ donors and 25 HCV-negative recipients.
“While these data are very encouraging, I think we do need to have long-term follow-up of these patients for graft survival, as well as the effect on wait times,” Dr. Ghany said.
Dr. Ghany reported no disclosures related to his presentation.
BOSTON – Treatment of persons who inject drugs, updates in pangenotypic direct-acting antiviral therapy, the benefits of sustained virologic response, and preemptive therapy in donor-positive organ transplantation topped the list of notable hepatitis C–related abstracts this year at the annual meeting of the American Association for the Study of Liver Diseases.
That’s according to Marc Ghany, MD, of the liver diseases branch of the National Institute of Diabetes and Digestive and Kidney Diseases at the National Institutes of Health, who gave a hepatitis C debrief to attendees on the final day of the meeting. Here are some of the meeting highlights as summarized by Dr. Ghany in this well-attended last-day session.
Treatment of HCV in people who inject drugs
Emerging data suggest it is feasible to treat hepatitis C virus (HCV) infection in persons who inject drugs (PWIDs); however, overcoming adherence issues remains a challenge, Dr. Ghany told attendees.
According to one study presented at AASLD by Dhiman and coauthors (Abstract 0165), decentralized care of PWIDs using direct-acting antiviral (DAA) therapy was safe and effective, even in those with cirrhosis. Authors demonstrated an “impressive” rate of sustained virologic response at 12 weeks (SVR12) of 91% by a modified intention-to-treat analysis, Dr. Ghany said; however, treatment interruptions were frequent and reduced the overall SVR rate in the study to 78%.
Other studies at the meeting looked at strategies to improve DAA efficacy in this population of patients at high risk of nonadherence, including use of a digital medicine program (Abstract 1554) and a model of care in which an internist-addiction medicine specialist evaluated opiate-dependent patients for HCV infection in a hepatology clinic (Abstract 1589).
Reinfection remains a focus of research in PWIDs. At this meeting, Janjua and coauthors reported that DAA-treated PWIDs in British Columbia had a threefold higher rate of reinfection versus non-PWIDs; however, there were no detected reinfections among PWIDs who had received uninterrupted opioid agonist therapy. “These data suggested that opioid agonist therapy should be given before and after HCV treatment in persons who inject drugs to prevent the infection,” Dr. Ghany said in his presentation.
Updates on pangenotypic DAA therapy
Jonas and coauthors (Abstract 1551) reported on the safety and efficacy of glecaprevir/pibrentasvir for 8 weeks in children with chronic HCV infection enrolled in the ongoing phase 2/3 DORA study. The SVR12 was high, according to Dr. Ghany, at 96% overall, and consistent across age cohorts from 3 to less than 12 years of age.
“In the near future, we should have a safe and effective regimen (approved) for children 3 years or older,” Dr. Ghany said. “I think this will serve us well, as we try to eliminate HCV in children, who number up to 5 million cases worldwide.”
A short course of glecaprevir/pibrentasvir is approved for patients with HCV and compensated cirrhosis, and data to support that was presented last year at The Liver Meeting; however, data were not presented on patients with genotype 3, the most difficult-to-treat genotype, Dr. Ghany said. That gap was filled at this year’s meeting with a report (Abstract LP9) showing SVR12 rates of 98.4% per protocol and 95.2% in intention-to-treat analysis.
Relationship of SVR to clinical outcomes
While the impact of sustained virologic response (SVR) on all-cause mortality is clear in patients with HCV, less is known about the effect of SVR on liver-related mortality and other outcomes, Dr. Ghany said. In one study presented here (Abstract 0039), based on analysis of a Veterans Affairs database of patients with chronic HCV infection, SVR was linked to a significant reduction in liver-related mortality, while in another report (Abstract 0037), SVR was associated with significant reductions in acute coronary syndromes, end-stage renal disease, and ischemic stroke.
Similarly, a multinational, propensity score–matched analysis (Abstract 0040) demonstrated that SVR had an impact on 5-year overall survival and liver-related survival in patients with HCV-related hepatocellular carcinoma (HCC). “For HCC patients who are candidates for HCC therapy, consideration should also be given to treating these individuals (with DAA therapy) because of the impact on overall survival,” Dr. Ghany said.
Preemptive DAA therapy in organ transplantation
Exciting new data show that preemptive therapy, given for short durations, appears to either prevent or cure HCV infection after organ transplant, said Dr. Ghany.
A retrospective analysis by Wijarnpreecha and colleagues (Abstract 0003) showed that 12 or 24 weeks of direct-acting antiviral (DAA) therapy resulted in an SVR12 for 24 out of 24 HCV-seropositive to HCV-seronegative liver transplants, while Durand and colleagues (Abstract 0042) showed that just 4 weeks of pre- and postexposure DAA prophylaxis resulted in SVR12s for 9 out of 9 HCV donor-positive, recipient-negative kidney transplants. Finally, Feld and coauthors (Abstract 0038) showed that preemptive ezetimibe with DAA therapy for 7 days prevented or rapidly cured infection in an experience that included 16 HCV-positive organ donors and 25 HCV-negative recipients.
“While these data are very encouraging, I think we do need to have long-term follow-up of these patients for graft survival, as well as the effect on wait times,” Dr. Ghany said.
Dr. Ghany reported no disclosures related to his presentation.
REPORTING FROM THE LIVER MEETING 2019




















