User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Geriatric IBD hospitalization carries steep inpatient mortality
SAN ANTONIO – Jeffrey Schwartz, MD, reported at the annual meeting of the American College of Gastroenterology.

The magnitude of the age-related increased risk highlighted in this large national study was strikingly larger than the differential inpatient mortality between geriatric and nongeriatric patients hospitalized for conditions other than inflammatory bowel disease (IBD). It’s a finding that reveals a major unmet need for improved systems of care for elderly hospitalized IBD patients, according to Dr. Schwartz, an internal medicine resident at Beth Israel Deaconess Medical Center, Boston.
“Given the high prevalence of IBD patients that require inpatient admission, as well as the rapidly aging nature of the U.S. population, it’s our hope that this study will provide some insight to drive efforts to improve standardized guideline-directed therapy and propose interventions to help close what I think is a very important gap in clinical care,” he said.
It’s well established that a second peak of IBD diagnoses occurs in 50- to 70-year-olds. At present, roughly 30% of all individuals carrying the diagnosis of IBD are over age 65, and with the graying of the baby-boomer population, this proportion is climbing.
Dr. Schwartz presented a study of the National Inpatient Sample for 2016, which is a representative sample comprising 20% of all U.S. hospital discharges for that year, the most recent year for which the data are available. The study population included all 71,040 patients hospitalized for acute management of Crohn’s disease or its immediate complications, of whom 10,095 were aged over age 75 years, as well as the 35,950 patients hospitalized for ulcerative colitis, 8,285 of whom were over 75.
Inpatient mortality occurred in 1.5% of the geriatric admissions, compared with 0.2% of nongeriatric admissions for Crohn’s disease. Similarly, the inpatient mortality rate in geriatric patients with ulcerative colitis was 1.0% versus 0.1% in patients under age 75 hospitalized for ulcerative colitis.
There are lots of reasons why the management of geriatric patients with IBD is particularly challenging, Dr. Schwartz noted. They have a higher burden of comorbid conditions, worse nutritional status, and increased risks of infection and cancer. In a regression analysis that attempted to control for such confounders using the Elixhauser mortality index, the nongeriatric Crohn’s disease patients were an adjusted 75% less likely to die in the hospital than those who were older. Nongeriatric ulcerative colitis patients were 81% less likely to die than geriatric patients with the disease. In contrast, nongeriatric patients admitted for reasons other than IBD had only an adjusted 50% lower risk of inpatient mortality than those who were older than 75.
Of note, in this analysis adjusted for confounders, there was no difference between geriatric and nongeriatric IBD patients in terms of resource utilization as reflected in average length of stay and hospital charges, Dr. Schwartz continued.
Asked if he could shed light on any specific complications that drove the age-related disparity in inpatient mortality in the IBD population, the physician replied that he and his coinvestigators were thwarted in their effort to do so because the inpatient mortality of 1.0%-1.5% was so low that further breakdown as to causes of death would have been statistically unreliable. It might be possible to do so successfully by combining several years of National Inpatient Sample data. That being said, it’s reasonable to hypothesize that cardiovascular complications are an important contributor, he added.
Dr. Schwartz reported having no financial conflicts regarding his study, conducted free of commercial support.
SAN ANTONIO – Jeffrey Schwartz, MD, reported at the annual meeting of the American College of Gastroenterology.

The magnitude of the age-related increased risk highlighted in this large national study was strikingly larger than the differential inpatient mortality between geriatric and nongeriatric patients hospitalized for conditions other than inflammatory bowel disease (IBD). It’s a finding that reveals a major unmet need for improved systems of care for elderly hospitalized IBD patients, according to Dr. Schwartz, an internal medicine resident at Beth Israel Deaconess Medical Center, Boston.
“Given the high prevalence of IBD patients that require inpatient admission, as well as the rapidly aging nature of the U.S. population, it’s our hope that this study will provide some insight to drive efforts to improve standardized guideline-directed therapy and propose interventions to help close what I think is a very important gap in clinical care,” he said.
It’s well established that a second peak of IBD diagnoses occurs in 50- to 70-year-olds. At present, roughly 30% of all individuals carrying the diagnosis of IBD are over age 65, and with the graying of the baby-boomer population, this proportion is climbing.
Dr. Schwartz presented a study of the National Inpatient Sample for 2016, which is a representative sample comprising 20% of all U.S. hospital discharges for that year, the most recent year for which the data are available. The study population included all 71,040 patients hospitalized for acute management of Crohn’s disease or its immediate complications, of whom 10,095 were aged over age 75 years, as well as the 35,950 patients hospitalized for ulcerative colitis, 8,285 of whom were over 75.
Inpatient mortality occurred in 1.5% of the geriatric admissions, compared with 0.2% of nongeriatric admissions for Crohn’s disease. Similarly, the inpatient mortality rate in geriatric patients with ulcerative colitis was 1.0% versus 0.1% in patients under age 75 hospitalized for ulcerative colitis.
There are lots of reasons why the management of geriatric patients with IBD is particularly challenging, Dr. Schwartz noted. They have a higher burden of comorbid conditions, worse nutritional status, and increased risks of infection and cancer. In a regression analysis that attempted to control for such confounders using the Elixhauser mortality index, the nongeriatric Crohn’s disease patients were an adjusted 75% less likely to die in the hospital than those who were older. Nongeriatric ulcerative colitis patients were 81% less likely to die than geriatric patients with the disease. In contrast, nongeriatric patients admitted for reasons other than IBD had only an adjusted 50% lower risk of inpatient mortality than those who were older than 75.
Of note, in this analysis adjusted for confounders, there was no difference between geriatric and nongeriatric IBD patients in terms of resource utilization as reflected in average length of stay and hospital charges, Dr. Schwartz continued.
Asked if he could shed light on any specific complications that drove the age-related disparity in inpatient mortality in the IBD population, the physician replied that he and his coinvestigators were thwarted in their effort to do so because the inpatient mortality of 1.0%-1.5% was so low that further breakdown as to causes of death would have been statistically unreliable. It might be possible to do so successfully by combining several years of National Inpatient Sample data. That being said, it’s reasonable to hypothesize that cardiovascular complications are an important contributor, he added.
Dr. Schwartz reported having no financial conflicts regarding his study, conducted free of commercial support.
SAN ANTONIO – Jeffrey Schwartz, MD, reported at the annual meeting of the American College of Gastroenterology.

The magnitude of the age-related increased risk highlighted in this large national study was strikingly larger than the differential inpatient mortality between geriatric and nongeriatric patients hospitalized for conditions other than inflammatory bowel disease (IBD). It’s a finding that reveals a major unmet need for improved systems of care for elderly hospitalized IBD patients, according to Dr. Schwartz, an internal medicine resident at Beth Israel Deaconess Medical Center, Boston.
“Given the high prevalence of IBD patients that require inpatient admission, as well as the rapidly aging nature of the U.S. population, it’s our hope that this study will provide some insight to drive efforts to improve standardized guideline-directed therapy and propose interventions to help close what I think is a very important gap in clinical care,” he said.
It’s well established that a second peak of IBD diagnoses occurs in 50- to 70-year-olds. At present, roughly 30% of all individuals carrying the diagnosis of IBD are over age 65, and with the graying of the baby-boomer population, this proportion is climbing.
Dr. Schwartz presented a study of the National Inpatient Sample for 2016, which is a representative sample comprising 20% of all U.S. hospital discharges for that year, the most recent year for which the data are available. The study population included all 71,040 patients hospitalized for acute management of Crohn’s disease or its immediate complications, of whom 10,095 were aged over age 75 years, as well as the 35,950 patients hospitalized for ulcerative colitis, 8,285 of whom were over 75.
Inpatient mortality occurred in 1.5% of the geriatric admissions, compared with 0.2% of nongeriatric admissions for Crohn’s disease. Similarly, the inpatient mortality rate in geriatric patients with ulcerative colitis was 1.0% versus 0.1% in patients under age 75 hospitalized for ulcerative colitis.
There are lots of reasons why the management of geriatric patients with IBD is particularly challenging, Dr. Schwartz noted. They have a higher burden of comorbid conditions, worse nutritional status, and increased risks of infection and cancer. In a regression analysis that attempted to control for such confounders using the Elixhauser mortality index, the nongeriatric Crohn’s disease patients were an adjusted 75% less likely to die in the hospital than those who were older. Nongeriatric ulcerative colitis patients were 81% less likely to die than geriatric patients with the disease. In contrast, nongeriatric patients admitted for reasons other than IBD had only an adjusted 50% lower risk of inpatient mortality than those who were older than 75.
Of note, in this analysis adjusted for confounders, there was no difference between geriatric and nongeriatric IBD patients in terms of resource utilization as reflected in average length of stay and hospital charges, Dr. Schwartz continued.
Asked if he could shed light on any specific complications that drove the age-related disparity in inpatient mortality in the IBD population, the physician replied that he and his coinvestigators were thwarted in their effort to do so because the inpatient mortality of 1.0%-1.5% was so low that further breakdown as to causes of death would have been statistically unreliable. It might be possible to do so successfully by combining several years of National Inpatient Sample data. That being said, it’s reasonable to hypothesize that cardiovascular complications are an important contributor, he added.
Dr. Schwartz reported having no financial conflicts regarding his study, conducted free of commercial support.
REPORTING FROM ACG 2019
Key clinical point: A major unmet need exists for better guideline-directed management of geriatric patients hospitalized for inflammatory bowel disease.
Major finding: The inpatient mortality rate among patients aged over age 75 years hospitalized for management of inflammatory bowel disease is four to five times higher than in those who are younger.
Study details: This was a retrospective analysis of all 106,990 hospital admissions for management of inflammatory bowel disease included in the 2016 National Inpatient Sample.
Disclosures: The presenter reported having no financial conflicts regarding his study, conducted free of commercial support.
Source: Schwartz J. ACG 2019, Abstract 42.
Treating LDL to below 70 reduces recurrent stroke
PHILADELPHIA – Treating patients to a lower LDL target after an ischemic stroke of atherosclerotic origin resulted in fewer recurrent strokes or major cardiovascular events, compared with a higher LDL goal, even though the international trial was stopped early because of lack of funding.
“In the Treat Stroke to Target [TST] trial we showed that the group of patients with an atherosclerotic stroke achieving an LDL cholesterol of less than 70 mg/dL had 22% less recurrent ischemic stroke or other major vascular events than the group achieving a LDL cholesterol between 90 and 110 mg/dL,” lead author Pierre Amarenco, MD, chairman of the department of neurology and the stroke center at Bichat Hospital in Paris, said in an interview.
“We avoided more than one in recurrence in five,” he added.
The findings of the investigator-initiated trial were reported during a late-breaking research session at the American Heart Association scientific sessions and simultaneously published online Nov. 18 in the New England Journal of Medicine (doi: 10.1056/NEJMoa1910355).
Discussant Mitchell S.V. Elkind, MD, president-elect of the American Heart Association, called the TST findings “practice confirming” of a strategy many cardiologists already follow for stroke patients.
“The TST study is only the second trial that was done in neurology for stroke prevention using statins and lipid-lowering therapy, and that’s what makes it a hopeful and real advance,” he said in an interview.
To achieve the LDL-lowering goal, two-thirds of patients received a high-dose statin therapy while the remainder received both high-dose statin and ezetimibe (Zetia, Merck). There were no significant increases in intracranial hemorrhage observed between lower- and higher-target groups.
“Now guidelines should move to recommending a target LDL cholesterol of less than 70 mg/dL in all patients with a proven ischemic stroke of atherosclerotic origin,” said Dr. Amarenco, who is also a professor of neurology at Denis Diderot Paris University.
Rare lipid study following stroke
American Heart Association/American Stroke Association guidelines recommend intense statin therapy after an atherothrombotic stroke “but no target level is given to the practitioners,” Dr. Amarenco said. “In reality, most patients receive a reduced dose of statin.”
For example, despite 70% of patients receiving a statin, the average LDL cholesterol level was 92 mg/dL in a real-world registry.
The TST trial is the first major study to evaluate treating to target LDL levels in the ischemic stroke population since the SPARCL trial in 2006. SPARCL was the first randomized, controlled clinical trial to evaluate whether daily statin therapy could reduce the risk of stroke in patients who had suffered a stroke or transient ischemic attack (TIA).
SPARCL demonstrated a 16% risk reduction with atorvastatin 80 mg daily versus placebo, and further risk reduction of 33% among those with carotid stenosis, over 5 years. There was some concern about safety for a time; post-hoc analysis showed what appeared to be an increased risk for intracranial hemorrhage with statin treatment. Subsequent analyses seemed to suggest the finding may have been a chance one, however.
For the TST study, Dr. Amarenco and colleagues enrolled participants between March 2010 and December 2018 at one of 61 centers in France. In 2015, the study expanded to include 16 sites in South Korea.
Investigators evaluated participants after an ischemic stroke or a TIA with evidence of atherosclerosis. Blood pressure, smoking cessation, and diabetes were well controlled, he said.
Dr. Amarenco and colleagues randomly assigned 1,430 participants to the low–LDL cholesterol target group, less than 70 mg/dL, and another 1,430 to a high-LDL group with a target of 100 mg/dL.
Assessments were every 6 months and up to 1 year after the last patient joined the study.
Treatment with any available statin on the market was allowed. Ezetimibe could be added on top of statin therapy as necessary. A total of 55% were statin naive at study entry.
Study stopped early
The trial was stopped in May 2019 after allocated funds ran out. At this point, researchers had 277 events to analyze, although their initial goal was to reach 385.
The primary endpoint of this event-driven trial was a composite of nonfatal stroke, nonfatal MI, and unstable angina followed by urgent coronary revascularization; TIA followed by urgent carotid revascularization; or cardiovascular death, including sudden deaths.
The endpoint was experienced by 8.5% of participants in the lower-target group versus 10.9% of those in the higher-target group. This translated to a 22% relative risk reduction (adjusted hazard ratio, 0.78; 95% confidence interval, 0.68-0.98; P =.04).
A total of 86% of participants had an ischemic stroke confirmed by brain MRI or CT scan. In this group, the relative risk reduction was 33% – “meaning that we could avoid one-third of recurrent major vascular events,” Dr. Amarenco said.
Furthermore, targeting the lower LDL levels was associated with a relative risk reduction of 40% among those with diabetes.
Secondary outcomes not significant
The investigators used hierarchical testing to compare two outcomes at a time in a prespecified order. They planned to continue this strategy until a comparison emerged as nonsignificant.
This occurred right away when their first composite secondary endpoint comparison between nonfatal MI and urgent revascularization was found to be not significantly different between groups (P = .12).
The early ending “weakened the results of the trial, and the results should be taken with caution because of that,” Dr. Amarenco said.
In addition, the number of hemorrhagic strokes did not differ significantly between groups. There were 18 of these events in the lower-target group and 13 in the higher-target cohort.
That numerical increase in intracranial hemorrhage was “driven by the Korean patients. … and that is something we will report soon,” Dr. Amarenco said.
Interestingly, the researchers also evaluated how much time participants spent within the target LDL cholesterol range, averaged by study site. They found that 53% of the lower–LDL target group, for example, was in the therapeutic range on average during the study.
When Dr. Amarenco and colleagues looked at participants who managed to spend 50%-100% in the target range, the relative risk reduction was 36%.
“So we can hypothesize that, if we had used a more potent drug like PCSK9 inhibitors to be closer to 100% in the therapeutic range, we may have had a greater effect size,” Dr. Amarenco said.
“Our results suggest that LDL cholesterol is causally related to atherosclerosis and confirm that the lower the LDL cholesterol the better,” Dr. Amarenco said.
“Future trials should explore the efficacy and safety of lowering LDL cholesterol to very low levels such as less than 55 mg/dL or even 30 mg/dL (as obtained in the FOURIER trial) by using PCSK9 inhibitors or equivalent in patients with an ischemic stroke due to atherosclerotic disease,” Dr. Amarenco said.
‘Practice-confirming’ findings
The findings are also in line with secondary analyses of the WASID (Neurology. 1995 Aug;45[8]:1488-93) and SAMMPRIS trials, which should dispel some concerns that persist about taking LDL to such low levels that it increases risk of intracerebral hemorrhage, Dr. Amarenco noted.
However, TST, he said, didn’t provide clear answers on what specific subgroups of patients with a stroke history would benefit from aggressive lipid lowering.
“What is stroke without atherosclerotic disease?” he said. “Some people say small-vessel disease is also a form of atherosclerosis, and most patients with atrial fibrillation, which is increasingly recognized as a cause of stroke, are also going to have atherosclerosis of the heart as well as the brain and blood vessels.
“Many, many stroke patients will fall into this category,” Dr. Elkind said, “and the question is, should they be treated more aggressively with lipid lowering?”
“The results of this study fit pretty nicely into the rubric of the AHA cholesterol guidelines,” said Donald M. Lloyd-Jones, MD, chairman, department of preventive medicine at Northwestern University Feinberg School of Medicine, Chicago, and chair of the AHA’s 2019 Council on Scientific Sessions Programming. Dr. Lloyd-Jones was also a member of the guideline committee.
Stroke patients are not “garden variety coronary patients,” he said. “The concern about intracerebral hemorrhage continues to be something that we wonder about: Should we be driving our stroke patients as low as our coronary patients? I think these data will certainly help us.”
Consideration for future guidelines
The study would have been more helpful if it provided more detail about the treatment regimens used, Jennifer Robinson, MD, director of the prevention intervention center, department of epidemiology, University of Iowa, said in an interview.
“What was the dose intensity of statins the patients were on?” Dr. Robinson said. “Part of our struggle has been to convince people to use high-intensity statins – get the maximum from statins that are generic now and cost saving in even very low-risk primary prevention patients.”
She said that a third of patients in TST also took ezetimibe with the statin “makes sense” because of its generic status.
Nonetheless, Dr. Robinson said, TST adds to the evidence that LDL of 100 mg/dL is not good enough, that high-intensity statin therapy is superior to a moderate regimen and that adding a nonstatin – ezetimibe in TST – can derive added benefit.
The TST findings may give guideline writers direction going forward, she said. “We really need to start thinking about the potential for net benefit from added therapy, whether it’s from intensifying LDL lowering, adding icosapent ethyl (Vascepa, Amarin), which seems to have remarkable benefits, or SGLT2 inhibitor,” she said.
“There are a lot of options,” Dr. Robinson said. “We need to have an outlook beyond just treating to target with what really is the best maximized accepted therapy.”
TST was funded primarily by French Government, but also with grants from Pfizer, Astra Zeneca and Merck. Dr. Amarenco disclosed that he is a consultant or advisor to Modest, Sanofi, Bristol-Myers Squibb, and Amgen; receives honoraria from Modest, Amgen, Kowa, Shing Poon, Kowa, Bayer, GSK, Fibrogen, and AstraZeneca. He also receives research grants from Pfizer, Astra Zeneca, Sanofi, BMS, Merck, Boston Scientific, and the French Government.
This article also appears on Medscape.com.
SOURCE: Amarenco P. ACC 2019, Late Breaking Science 6 session.
PHILADELPHIA – Treating patients to a lower LDL target after an ischemic stroke of atherosclerotic origin resulted in fewer recurrent strokes or major cardiovascular events, compared with a higher LDL goal, even though the international trial was stopped early because of lack of funding.
“In the Treat Stroke to Target [TST] trial we showed that the group of patients with an atherosclerotic stroke achieving an LDL cholesterol of less than 70 mg/dL had 22% less recurrent ischemic stroke or other major vascular events than the group achieving a LDL cholesterol between 90 and 110 mg/dL,” lead author Pierre Amarenco, MD, chairman of the department of neurology and the stroke center at Bichat Hospital in Paris, said in an interview.
“We avoided more than one in recurrence in five,” he added.
The findings of the investigator-initiated trial were reported during a late-breaking research session at the American Heart Association scientific sessions and simultaneously published online Nov. 18 in the New England Journal of Medicine (doi: 10.1056/NEJMoa1910355).
Discussant Mitchell S.V. Elkind, MD, president-elect of the American Heart Association, called the TST findings “practice confirming” of a strategy many cardiologists already follow for stroke patients.
“The TST study is only the second trial that was done in neurology for stroke prevention using statins and lipid-lowering therapy, and that’s what makes it a hopeful and real advance,” he said in an interview.
To achieve the LDL-lowering goal, two-thirds of patients received a high-dose statin therapy while the remainder received both high-dose statin and ezetimibe (Zetia, Merck). There were no significant increases in intracranial hemorrhage observed between lower- and higher-target groups.
“Now guidelines should move to recommending a target LDL cholesterol of less than 70 mg/dL in all patients with a proven ischemic stroke of atherosclerotic origin,” said Dr. Amarenco, who is also a professor of neurology at Denis Diderot Paris University.
Rare lipid study following stroke
American Heart Association/American Stroke Association guidelines recommend intense statin therapy after an atherothrombotic stroke “but no target level is given to the practitioners,” Dr. Amarenco said. “In reality, most patients receive a reduced dose of statin.”
For example, despite 70% of patients receiving a statin, the average LDL cholesterol level was 92 mg/dL in a real-world registry.
The TST trial is the first major study to evaluate treating to target LDL levels in the ischemic stroke population since the SPARCL trial in 2006. SPARCL was the first randomized, controlled clinical trial to evaluate whether daily statin therapy could reduce the risk of stroke in patients who had suffered a stroke or transient ischemic attack (TIA).
SPARCL demonstrated a 16% risk reduction with atorvastatin 80 mg daily versus placebo, and further risk reduction of 33% among those with carotid stenosis, over 5 years. There was some concern about safety for a time; post-hoc analysis showed what appeared to be an increased risk for intracranial hemorrhage with statin treatment. Subsequent analyses seemed to suggest the finding may have been a chance one, however.
For the TST study, Dr. Amarenco and colleagues enrolled participants between March 2010 and December 2018 at one of 61 centers in France. In 2015, the study expanded to include 16 sites in South Korea.
Investigators evaluated participants after an ischemic stroke or a TIA with evidence of atherosclerosis. Blood pressure, smoking cessation, and diabetes were well controlled, he said.
Dr. Amarenco and colleagues randomly assigned 1,430 participants to the low–LDL cholesterol target group, less than 70 mg/dL, and another 1,430 to a high-LDL group with a target of 100 mg/dL.
Assessments were every 6 months and up to 1 year after the last patient joined the study.
Treatment with any available statin on the market was allowed. Ezetimibe could be added on top of statin therapy as necessary. A total of 55% were statin naive at study entry.
Study stopped early
The trial was stopped in May 2019 after allocated funds ran out. At this point, researchers had 277 events to analyze, although their initial goal was to reach 385.
The primary endpoint of this event-driven trial was a composite of nonfatal stroke, nonfatal MI, and unstable angina followed by urgent coronary revascularization; TIA followed by urgent carotid revascularization; or cardiovascular death, including sudden deaths.
The endpoint was experienced by 8.5% of participants in the lower-target group versus 10.9% of those in the higher-target group. This translated to a 22% relative risk reduction (adjusted hazard ratio, 0.78; 95% confidence interval, 0.68-0.98; P =.04).
A total of 86% of participants had an ischemic stroke confirmed by brain MRI or CT scan. In this group, the relative risk reduction was 33% – “meaning that we could avoid one-third of recurrent major vascular events,” Dr. Amarenco said.
Furthermore, targeting the lower LDL levels was associated with a relative risk reduction of 40% among those with diabetes.
Secondary outcomes not significant
The investigators used hierarchical testing to compare two outcomes at a time in a prespecified order. They planned to continue this strategy until a comparison emerged as nonsignificant.
This occurred right away when their first composite secondary endpoint comparison between nonfatal MI and urgent revascularization was found to be not significantly different between groups (P = .12).
The early ending “weakened the results of the trial, and the results should be taken with caution because of that,” Dr. Amarenco said.
In addition, the number of hemorrhagic strokes did not differ significantly between groups. There were 18 of these events in the lower-target group and 13 in the higher-target cohort.
That numerical increase in intracranial hemorrhage was “driven by the Korean patients. … and that is something we will report soon,” Dr. Amarenco said.
Interestingly, the researchers also evaluated how much time participants spent within the target LDL cholesterol range, averaged by study site. They found that 53% of the lower–LDL target group, for example, was in the therapeutic range on average during the study.
When Dr. Amarenco and colleagues looked at participants who managed to spend 50%-100% in the target range, the relative risk reduction was 36%.
“So we can hypothesize that, if we had used a more potent drug like PCSK9 inhibitors to be closer to 100% in the therapeutic range, we may have had a greater effect size,” Dr. Amarenco said.
“Our results suggest that LDL cholesterol is causally related to atherosclerosis and confirm that the lower the LDL cholesterol the better,” Dr. Amarenco said.
“Future trials should explore the efficacy and safety of lowering LDL cholesterol to very low levels such as less than 55 mg/dL or even 30 mg/dL (as obtained in the FOURIER trial) by using PCSK9 inhibitors or equivalent in patients with an ischemic stroke due to atherosclerotic disease,” Dr. Amarenco said.
‘Practice-confirming’ findings
The findings are also in line with secondary analyses of the WASID (Neurology. 1995 Aug;45[8]:1488-93) and SAMMPRIS trials, which should dispel some concerns that persist about taking LDL to such low levels that it increases risk of intracerebral hemorrhage, Dr. Amarenco noted.
However, TST, he said, didn’t provide clear answers on what specific subgroups of patients with a stroke history would benefit from aggressive lipid lowering.
“What is stroke without atherosclerotic disease?” he said. “Some people say small-vessel disease is also a form of atherosclerosis, and most patients with atrial fibrillation, which is increasingly recognized as a cause of stroke, are also going to have atherosclerosis of the heart as well as the brain and blood vessels.
“Many, many stroke patients will fall into this category,” Dr. Elkind said, “and the question is, should they be treated more aggressively with lipid lowering?”
“The results of this study fit pretty nicely into the rubric of the AHA cholesterol guidelines,” said Donald M. Lloyd-Jones, MD, chairman, department of preventive medicine at Northwestern University Feinberg School of Medicine, Chicago, and chair of the AHA’s 2019 Council on Scientific Sessions Programming. Dr. Lloyd-Jones was also a member of the guideline committee.
Stroke patients are not “garden variety coronary patients,” he said. “The concern about intracerebral hemorrhage continues to be something that we wonder about: Should we be driving our stroke patients as low as our coronary patients? I think these data will certainly help us.”
Consideration for future guidelines
The study would have been more helpful if it provided more detail about the treatment regimens used, Jennifer Robinson, MD, director of the prevention intervention center, department of epidemiology, University of Iowa, said in an interview.
“What was the dose intensity of statins the patients were on?” Dr. Robinson said. “Part of our struggle has been to convince people to use high-intensity statins – get the maximum from statins that are generic now and cost saving in even very low-risk primary prevention patients.”
She said that a third of patients in TST also took ezetimibe with the statin “makes sense” because of its generic status.
Nonetheless, Dr. Robinson said, TST adds to the evidence that LDL of 100 mg/dL is not good enough, that high-intensity statin therapy is superior to a moderate regimen and that adding a nonstatin – ezetimibe in TST – can derive added benefit.
The TST findings may give guideline writers direction going forward, she said. “We really need to start thinking about the potential for net benefit from added therapy, whether it’s from intensifying LDL lowering, adding icosapent ethyl (Vascepa, Amarin), which seems to have remarkable benefits, or SGLT2 inhibitor,” she said.
“There are a lot of options,” Dr. Robinson said. “We need to have an outlook beyond just treating to target with what really is the best maximized accepted therapy.”
TST was funded primarily by French Government, but also with grants from Pfizer, Astra Zeneca and Merck. Dr. Amarenco disclosed that he is a consultant or advisor to Modest, Sanofi, Bristol-Myers Squibb, and Amgen; receives honoraria from Modest, Amgen, Kowa, Shing Poon, Kowa, Bayer, GSK, Fibrogen, and AstraZeneca. He also receives research grants from Pfizer, Astra Zeneca, Sanofi, BMS, Merck, Boston Scientific, and the French Government.
This article also appears on Medscape.com.
SOURCE: Amarenco P. ACC 2019, Late Breaking Science 6 session.
PHILADELPHIA – Treating patients to a lower LDL target after an ischemic stroke of atherosclerotic origin resulted in fewer recurrent strokes or major cardiovascular events, compared with a higher LDL goal, even though the international trial was stopped early because of lack of funding.
“In the Treat Stroke to Target [TST] trial we showed that the group of patients with an atherosclerotic stroke achieving an LDL cholesterol of less than 70 mg/dL had 22% less recurrent ischemic stroke or other major vascular events than the group achieving a LDL cholesterol between 90 and 110 mg/dL,” lead author Pierre Amarenco, MD, chairman of the department of neurology and the stroke center at Bichat Hospital in Paris, said in an interview.
“We avoided more than one in recurrence in five,” he added.
The findings of the investigator-initiated trial were reported during a late-breaking research session at the American Heart Association scientific sessions and simultaneously published online Nov. 18 in the New England Journal of Medicine (doi: 10.1056/NEJMoa1910355).
Discussant Mitchell S.V. Elkind, MD, president-elect of the American Heart Association, called the TST findings “practice confirming” of a strategy many cardiologists already follow for stroke patients.
“The TST study is only the second trial that was done in neurology for stroke prevention using statins and lipid-lowering therapy, and that’s what makes it a hopeful and real advance,” he said in an interview.
To achieve the LDL-lowering goal, two-thirds of patients received a high-dose statin therapy while the remainder received both high-dose statin and ezetimibe (Zetia, Merck). There were no significant increases in intracranial hemorrhage observed between lower- and higher-target groups.
“Now guidelines should move to recommending a target LDL cholesterol of less than 70 mg/dL in all patients with a proven ischemic stroke of atherosclerotic origin,” said Dr. Amarenco, who is also a professor of neurology at Denis Diderot Paris University.
Rare lipid study following stroke
American Heart Association/American Stroke Association guidelines recommend intense statin therapy after an atherothrombotic stroke “but no target level is given to the practitioners,” Dr. Amarenco said. “In reality, most patients receive a reduced dose of statin.”
For example, despite 70% of patients receiving a statin, the average LDL cholesterol level was 92 mg/dL in a real-world registry.
The TST trial is the first major study to evaluate treating to target LDL levels in the ischemic stroke population since the SPARCL trial in 2006. SPARCL was the first randomized, controlled clinical trial to evaluate whether daily statin therapy could reduce the risk of stroke in patients who had suffered a stroke or transient ischemic attack (TIA).
SPARCL demonstrated a 16% risk reduction with atorvastatin 80 mg daily versus placebo, and further risk reduction of 33% among those with carotid stenosis, over 5 years. There was some concern about safety for a time; post-hoc analysis showed what appeared to be an increased risk for intracranial hemorrhage with statin treatment. Subsequent analyses seemed to suggest the finding may have been a chance one, however.
For the TST study, Dr. Amarenco and colleagues enrolled participants between March 2010 and December 2018 at one of 61 centers in France. In 2015, the study expanded to include 16 sites in South Korea.
Investigators evaluated participants after an ischemic stroke or a TIA with evidence of atherosclerosis. Blood pressure, smoking cessation, and diabetes were well controlled, he said.
Dr. Amarenco and colleagues randomly assigned 1,430 participants to the low–LDL cholesterol target group, less than 70 mg/dL, and another 1,430 to a high-LDL group with a target of 100 mg/dL.
Assessments were every 6 months and up to 1 year after the last patient joined the study.
Treatment with any available statin on the market was allowed. Ezetimibe could be added on top of statin therapy as necessary. A total of 55% were statin naive at study entry.
Study stopped early
The trial was stopped in May 2019 after allocated funds ran out. At this point, researchers had 277 events to analyze, although their initial goal was to reach 385.
The primary endpoint of this event-driven trial was a composite of nonfatal stroke, nonfatal MI, and unstable angina followed by urgent coronary revascularization; TIA followed by urgent carotid revascularization; or cardiovascular death, including sudden deaths.
The endpoint was experienced by 8.5% of participants in the lower-target group versus 10.9% of those in the higher-target group. This translated to a 22% relative risk reduction (adjusted hazard ratio, 0.78; 95% confidence interval, 0.68-0.98; P =.04).
A total of 86% of participants had an ischemic stroke confirmed by brain MRI or CT scan. In this group, the relative risk reduction was 33% – “meaning that we could avoid one-third of recurrent major vascular events,” Dr. Amarenco said.
Furthermore, targeting the lower LDL levels was associated with a relative risk reduction of 40% among those with diabetes.
Secondary outcomes not significant
The investigators used hierarchical testing to compare two outcomes at a time in a prespecified order. They planned to continue this strategy until a comparison emerged as nonsignificant.
This occurred right away when their first composite secondary endpoint comparison between nonfatal MI and urgent revascularization was found to be not significantly different between groups (P = .12).
The early ending “weakened the results of the trial, and the results should be taken with caution because of that,” Dr. Amarenco said.
In addition, the number of hemorrhagic strokes did not differ significantly between groups. There were 18 of these events in the lower-target group and 13 in the higher-target cohort.
That numerical increase in intracranial hemorrhage was “driven by the Korean patients. … and that is something we will report soon,” Dr. Amarenco said.
Interestingly, the researchers also evaluated how much time participants spent within the target LDL cholesterol range, averaged by study site. They found that 53% of the lower–LDL target group, for example, was in the therapeutic range on average during the study.
When Dr. Amarenco and colleagues looked at participants who managed to spend 50%-100% in the target range, the relative risk reduction was 36%.
“So we can hypothesize that, if we had used a more potent drug like PCSK9 inhibitors to be closer to 100% in the therapeutic range, we may have had a greater effect size,” Dr. Amarenco said.
“Our results suggest that LDL cholesterol is causally related to atherosclerosis and confirm that the lower the LDL cholesterol the better,” Dr. Amarenco said.
“Future trials should explore the efficacy and safety of lowering LDL cholesterol to very low levels such as less than 55 mg/dL or even 30 mg/dL (as obtained in the FOURIER trial) by using PCSK9 inhibitors or equivalent in patients with an ischemic stroke due to atherosclerotic disease,” Dr. Amarenco said.
‘Practice-confirming’ findings
The findings are also in line with secondary analyses of the WASID (Neurology. 1995 Aug;45[8]:1488-93) and SAMMPRIS trials, which should dispel some concerns that persist about taking LDL to such low levels that it increases risk of intracerebral hemorrhage, Dr. Amarenco noted.
However, TST, he said, didn’t provide clear answers on what specific subgroups of patients with a stroke history would benefit from aggressive lipid lowering.
“What is stroke without atherosclerotic disease?” he said. “Some people say small-vessel disease is also a form of atherosclerosis, and most patients with atrial fibrillation, which is increasingly recognized as a cause of stroke, are also going to have atherosclerosis of the heart as well as the brain and blood vessels.
“Many, many stroke patients will fall into this category,” Dr. Elkind said, “and the question is, should they be treated more aggressively with lipid lowering?”
“The results of this study fit pretty nicely into the rubric of the AHA cholesterol guidelines,” said Donald M. Lloyd-Jones, MD, chairman, department of preventive medicine at Northwestern University Feinberg School of Medicine, Chicago, and chair of the AHA’s 2019 Council on Scientific Sessions Programming. Dr. Lloyd-Jones was also a member of the guideline committee.
Stroke patients are not “garden variety coronary patients,” he said. “The concern about intracerebral hemorrhage continues to be something that we wonder about: Should we be driving our stroke patients as low as our coronary patients? I think these data will certainly help us.”
Consideration for future guidelines
The study would have been more helpful if it provided more detail about the treatment regimens used, Jennifer Robinson, MD, director of the prevention intervention center, department of epidemiology, University of Iowa, said in an interview.
“What was the dose intensity of statins the patients were on?” Dr. Robinson said. “Part of our struggle has been to convince people to use high-intensity statins – get the maximum from statins that are generic now and cost saving in even very low-risk primary prevention patients.”
She said that a third of patients in TST also took ezetimibe with the statin “makes sense” because of its generic status.
Nonetheless, Dr. Robinson said, TST adds to the evidence that LDL of 100 mg/dL is not good enough, that high-intensity statin therapy is superior to a moderate regimen and that adding a nonstatin – ezetimibe in TST – can derive added benefit.
The TST findings may give guideline writers direction going forward, she said. “We really need to start thinking about the potential for net benefit from added therapy, whether it’s from intensifying LDL lowering, adding icosapent ethyl (Vascepa, Amarin), which seems to have remarkable benefits, or SGLT2 inhibitor,” she said.
“There are a lot of options,” Dr. Robinson said. “We need to have an outlook beyond just treating to target with what really is the best maximized accepted therapy.”
TST was funded primarily by French Government, but also with grants from Pfizer, Astra Zeneca and Merck. Dr. Amarenco disclosed that he is a consultant or advisor to Modest, Sanofi, Bristol-Myers Squibb, and Amgen; receives honoraria from Modest, Amgen, Kowa, Shing Poon, Kowa, Bayer, GSK, Fibrogen, and AstraZeneca. He also receives research grants from Pfizer, Astra Zeneca, Sanofi, BMS, Merck, Boston Scientific, and the French Government.
This article also appears on Medscape.com.
SOURCE: Amarenco P. ACC 2019, Late Breaking Science 6 session.
REPORTING FROM AHA 2019
Evidence builds for AFib ablation’s efficacy in heart failure
Roughly a third of patients with heart failure also have atrial fibrillation, a comorbid combination notorious for working synergistically to worsen a patient’s quality of life and life expectancy.
During the past year, radiofrequency catheter ablation of atrial fibrillation in patients with both conditions has gathered steam as a way to intervene in at least selected patients, driven by study results that featured attention-grabbing reductions in death and cardiovascular hospitalizations.
The evidence favoring catheter ablation of atrial fibrillation (AFib) in patients with heart failure, particularly patients with heart failure with reduced ejection fraction (HFrEF), ramped up in 2019, spurred largely by a subgroup analysis from the CABANA trial, the largest randomized comparison by far of AFib ablation with antiarrhythmic drug treatment with 2,204 patients.
The past few months also featured release of two meta-analyses that took the CABANA results into account plus findings from about a dozen earlier randomized studies. Both meta-analyses, as well as the heart failure analysis from CABANA, all point in one direction, as stated in the conclusion of one of the meta-analyses: “In patients with AFib, catheter ablation is associated with all-cause mortality benefit, compared with medical therapy, that is driven by patients with AFib and HFrEF. Catheter ablation is safe and reduces cardiovascular hospitalizations and recurrences of atrial arrhythmias” both in patients with paroxysmal and persistent AFib,” wrote Stavros Stavrakis, MD, and his associates in their systematic review of 18 randomized, controlled trials of catheter ablation of AFib in a total of 4,464 patients with or without heart failure (Circ Arrhythm Electrophysiol. 2019 Sep;12[9]: e007414).
Despite these new data and analyses, clinicians seem to have very mixed reactions. Some call for an upgraded recommendation by professional societies that would support more aggressive use of AFib ablation in heart failure patients, and the anecdotal impressions of people who manage these patients are that ablation procedures have recently increased. But others advise caution, and note that in their opinion the efficacy data remain preliminary; the procedure has safety, logistical, and economic concerns; and questions remain about the ability of all active ablation programs to consistently deliver the results seen in published trials.
The meta-analysis led by Dr. Stavrakis showed that catheter ablation of AFib cut all-cause mortality during follow-up by a statistically significant 31%, compared with medical therapy, in all patients regardless of their heart failure status. But in patients with HFrEF, the reduction was 48%, along with a 38% cut in cardiovascular hospitalizations. In contrast, patients without heart failure who underwent AFib ablation showed no significant change in their all-cause mortality, compared with medical management of these patients.
“Based both on our meta-analysis and the CABANA data, patients with AFib most likely to benefit from ablation are patients younger than 65 and those with heart failure,” summed up Dr. Stavrakis, a cardiac electrophysiologist at the Heart Rhythm Institute of the University of Oklahoma in Oklahoma City.
The second meta-analysis, which initially appeared in July, analyzed data from 11 randomized trials of catheter ablations compared with anti-arrhythmic medical therapy for rate or rhythm control with in a total of 3,598 patients who all had heart failure, again including the patients enrolled in the CABANA study. The results showed a significant 49% relative drop in all cause mortality with ablation compared with medical treatment, and a statistically significant 56% cut in hospitalizations, as well as a significant, nearly 7% average, absolute improvement in left ventricular ejection fraction, plus benefits for preventing arrhythmia recurrence and improving quality of life (Eur Heart J. 2019 Jul 11. doi: 10.1093/eurheartj/ehz443).
“The magnitude of the effect seen in the meta-analysis, a 49% reduction in total mortality and a 56% reduction in hospitalizations, is rather staggering, and is larger than typically quoted for other medical interventions or device therapy in heart failure. The treatment effect was uniform among studies, and entirely compatible with the changes in left ventricular function, exercise capacity, and heart failure symptoms. Therefore, although more data are desirable, there are already arguably sufficient data to understand a great deal regarding the impact of a fib ablation,” commented Ross J. Hunter, MRCP, a cardiac electrophysiologist at Barts Heart Centre in London, and his associates in an editorial about this meta-analysis (Eur Heart J. 2019 Oct 22. doi: 10.1093/eurheartj/ehz704).
The heart failure analysis of CABANA (Catheter Ablation vs. Anti-Arrhythmic Drug Therapy for Atrial Fibrillation Trial) itself also showed striking findings when first reported at the annual scientific sessions of the Heart Rhythm Society last May. In presentations he made at this meeting, Douglas L. Packer, MD, CABANA’s lead investigator, reported details of a prespecified subgroup analysis of the 778 patients enrolled in CABANA who had heart failure at baseline, slightly more than a third of the total study enrollment. This was more than double the number of patients identified as specifically having heart failure at entry in the initial publication of CABANA’s findings (JAMA. 2019 Mar 15;321[134]:1261-74). Comparison of the 378 patients with heart failure and randomized to undergo ablation with the 400 with heart failure randomized to medical treatment showed a 36% reduction in the study’s primary, composite endpoint relative to the control group in an intention-to-treat analysis, and a 43% relative cut in all-cause mortality during follow-up, Dr. Packer reported at the May meeting. (As of early November 2019, these results had not yet appeared in a published article.) In contrast, in the 1,422 CABANA patients randomized who did not have heart failure, ablation produced results for these endpoints that were similar to and not statistically different from the outcomes in patients treated medically, said Dr. Packer, a cardiac electrophysiologist and professor of medicine at the Mayo Clinic in Rochester, Minn.
The CABANA results added to what had been previously reported from two other landmark studies that documented incremental efficacy of AFib ablation compared with medical treatment in patients with heart failure: The AATAC (Ablation vs Amiodarone for Treatment of AFib in Patients With Congestive HF and an Implanted Device) study, which randomized 203 patients (Circulation. 2016 Apr 26;133[17]:1637-44), and CASTLE-AF (Catheter Ablation vs. Standard Conventional Therapy in Patients with Left Ventricular Dysfunction and Atrial Fibrillation) trial, which randomized 363 patients (N Engl J Med. 2018 Feb 1;378[5]:417-27). These three studies contributed the most patients and outcomes to the two recent meta-analyses.
“The CASTLE-AF and AATAC trials both showed improved cardiovascular outcomes with ablation in patients with heart failure and AFib. The meta-analysis [by Dr. Stavrakis and his associates] and CABANA subgroup analysis further support use of catheter ablation to improve the outcomes in these patients,” noted Jonathan P. Piccini, MD, a cardiac electrophysiologist at Duke University, Durham, N.C., and a CABANA coinvestigator.
“The CABANA trial was very important because it confirmed the safety of catheter ablation, and more importantly suggested that patients with heart failure may benefit the most [from AFib ablation]. The evidence is very strong to advocate ablation as first-line therapy for selected patients with heart failure. Perhaps the optimal patients are those with [New York Heart Association] class I-III or ambulatory class IV heart failure who are on optimized, guideline-directed medical therapy. We have enough data to make this a class I recommendation. The question that remains is whether this is a cost effective strategy. Because it lowers rehospitalization and death, I suspect it is,” said Luigi Di Biase, MD, lead investigator of AATAC, and director of arrhythmia services at Montefiore Medical Center and professor of medicine at Albert Einstein College of Medicine, both in New York.
Opinions differ on AFib ablation’s role
Despite this expansive assessment of the current status of AFib ablation for patients with heart failure from Dr. Di Biase and shared by others, another camp of cardiologists currently sees ablation as having more limited current utility, as recommended earlier this year by a guideline-update panel representing the American Heart Association, the American College of Cardiology, and the Heart Rhythm Society. The guideline update included this new recommendation for how to use AFib ablation in heart failure patients: “AF catheter ablation may be reasonable in selected patients with symptomatic AFib and heart failure with reduced left ventricular ejection fraction to potentially lower mortality rate and reduce hospitalization for heart failure,” a class IIb recommendation. (J Am Coll Cardiol. 2019 Jul 9;74[1]:104-32). The guideline’s text cited the findings from AATAC and CASTLE-AF, but qualified both studies as “relatively small” and with “highly selected patient populations.” The guideline also incorporated the CABANA results into its considerations (although they may not have had the full analysis in heart failure patients available during their deliberations), but cited the study’s main limitation: CABANA failed to show a statistically significant difference in the primary endpoint in its primary, intention-to-treat analysis, which meant that by the strict statistical criteria that trialists apply to study findings, all other endpoints analyzed using CABANA’s are merely “hypothesis generating” and not definitive.
Questions about the extent of patient selection required to see a clear clinical-endpoint benefit from AFib ablation in heart failure patients, as well as the flawed validity of the CABANA results for making unqualified practice recommendation are the main arguments advanced by experts who caution against broader and more routine ablations.
“The findings from the heart failure subgroup of CABANA are hypothesis generating rather than definitive. Even with the recent meta-analysis, uncertainty remains regarding the ability of catheter ablation to improve outcomes beyond reducing AFib-related symptoms,” commented Gregg Fonarow, MD, a heart failure physician and professor of medicine at the University of California, Los Angeles.
“CASTLE-HF had fewer than 100 deaths combined in both arms, which means very unstable results. We don’t know a lot of detail about the heart failure patients in CABANA, and overall we do not have much data from patients with heart failure with preserved ejection fraction [HFpEF],” said Javed Butler, MD, a heart failure physician and professor and chairman of medicine at the University of Mississippi in Jackson. Dr. Butler also voiced his concerns (shared by other heart failure specialists) about the safety of ablation in heart failure patients, noting that “many patients require multiple ablations; many burns result in scarring and can worsen atrial function. In short, ablation of AFib is probably good for selected patients, but to have a class 1 recommendation, we need much larger trials with well-phenotyped heart failure patients,” Dr. Butler said in an interview.
“The totality of data still captures a relatively small number of patients. CASTLE-HF took 8 years to enroll fewer than 400 patients, and the results showed some heterogeneity. Study patients were a decade younger than average HFrEF patients in the community, and thus the effectiveness and safety of catheter ablation in people with more comorbidity and frailty remains in question. Certain HFrEF patients may be less likely to benefit, such as those with amyloid cardiomyopathy. And with the increasing availability of other treatments for HFrEF such as sacubitril/valsartan, dapagliflozin, and MitraClip, it is less clear how catheter ablation would [benefit patients] on top of what is now current best therapy,” said Larry Allen, MD, a heart failure physician and professor of medicine at the University of Colorado in Aurora.
“With these limitations and the fact that catheter ablation is not a simple procedure, a large randomized, controlled trial of ablation, compared with no ablation, in a wide range of HFrEF patients on contemporary therapy would be welcome,” Dr. Allen said. “Given the prevalence of heart failure and AFib and the potential positive and negative implications of catheter ablation running such a trial seems critical for patients and for society.”
“For ablation of AFib in heart failure to become a class I recommendation there will need to be results from larger randomized studies,” summed up Dr. Stavrakis. The meta-analysis that he coauthored noted that “the benefits of catheter ablation for AFib in HFrEF patients have been consistently shown for over a decade now; however, the uptake of this procedure by clinicians in practice has been slow.”
Despite this history of reticence and ongoing caution about ablation, some cardiology experts see the indications for AFib ablation in heart failure steadily creeping forward, buoyed by a safety record that has more benign than ablation’s reputation suggests.
The CABANA results showed that “ablation is remarkably safe in the hands of experienced clinicians, with risks comparable to anti-arrhythmic drugs,” said Peter R. Kowey, MD, a specialist in treating AFib and professor of medicine at Thomas Jefferson University in Philadelphia, who made this assessment during a talk at an AFib meeting in early 2019. Dr. Kowey’s take on what the CABANA safety data showed contrasts with the impression of other cardiologists who are wary of perceived dangers from ablation.
“Ablation comes with a lot of morbidity and mortality. It’s not that the idea of ablation is wrong, but the ability to do it without a lot of adverse effects. ... We’re not quite there yet,” said Douglas L. Mann, MD, a heart failure physician and professor of medicine and chief of the cardiovascular division at Washington University in St. Louis.
“If I had a patient with HFrEF and AFib who was really sick, I’m not so sure I’d send them for ablation, which is not a simple procedure. The patients we tend to send for ablation are selected. Ablation is a big undertaking in patients who are already sick, and it’s expensive. I don’t think the data we have now will change the consensus view, but every heart failure physician is sending some patients for AFib ablation. People are turning to AFib ablation earlier than before. I think the consensus is that ablation is for symptoms or poor rate control, not for better outcomes,” said Mariell Jessup, MD, a heart failure specialist and chief science and medical officer of the American Heart Association in Dallas.
However, this caution about safety and skepticism over efficacy may be dissipating as experience with ablation accumulates.
“CASTLE-AF and other data, including evidence for the apparent isolation of beta-blocker benefit to patients in sinus rhythm, have made me much more proactive about considering catheter ablation in my HFrEF patients. I think many other cardiologists have a similar view,” said Dr. Allen in an interview.
“A lot [of heart failure] patients are [being] referred for ablation, depending on the practice, setting, the local availability of electrophysiologists, and patient interest in ablation,” said Dr. Butler.
“We have no absolutely compelling data, but the data we have all point in the same direction. Like most, I am becoming convinced that AFib ablation in heart failure patients is a very valuable method for managing patients, but I can’t point to one study that was conclusive. Results from lots of studies show that it is likely, and when you add them all together it looks indisputable,” commented A. John Camm, MD, an atrial fibrillation specialist and professor of clinical cardiology at St. George’s University in London. “The findings put a responsibility on cardiologists to assess patients with heart failure for AFib. But there are nothing like enough resources to deal with all the patients who have heart failure who also have AFib.”
A rough estimate of just the U.S. volume of patients with heart failure and AFib is likely in the ball park of 2 million people (a third of the estimated 6 million American currently living with heart failure), and with the prevalence of each of these disorders rising precipitously (more than 5 million Americans have AFib) the confluence of the two should also show a steady increase. “It will take a major change in our concept of heart failure management to really address this. Potentially it would mean a large increase in the number of RF ablations of AFib, but the resources for that are not now present,” Dr. Camm said in an interview.
The attractions of catheter ablation also stand in contrast to the limitations of alternative treatments. Ablation is effective in a majority of patients for reducing AFib burden, both the frequency and duration of AFib episodes, and safety issues are mostly limited to the procedural and immediate postprocedural periods. The drugs available for trying to control AFib are beta-blockers, which provide rate control and can help prevent AFib onset, and rhythm-controlling anti-arrhythmic drugs like amiodarone, which have substantial limitations in both their ability to prevent arrhythmia recurrences as well as for safety.
“Most of the conventional antiarrhythmic drugs are contraindicated, frequently ineffective, or not well tolerated in patients with HFrEF. Catheter ablation of AFib provides an increasingly important option for rhythm control in these patients without using antiarrhythmic drugs,” Dr. Di Biase and his associates wrote in a recent review of AFib ablation in heart failure patients (Eur Heart J. 2019 Feb 21;40[8]:663-71).
“The guidelines that are controversial still make amiodarone a class I drug even though it’s been associated with serious side effects and has been shown in several heart failure trials to increase mortality. I can’t believe that ablation is a class IIb recommendation while a drug like amiodarone is a class I recommendation,” Dr. Di Biase said.
And although beta-blockers are a mainstay of heart failure treatment, once AFib becomes established they are less useful for maintaining sinus rhythm. “Beta-blockers provide effective rate control, but they can’t convert patients to sinus rhythm [once AFib begins], and there is no convincing evidence that patients on beta-blockers stay in sinus rhythm longer. You can’t just say: the patient is on a beta-blocker so I’ve done my best,” noted Dr. Jessup.
CABANA received funding from Biosense Webster, Boston Scientific, Medtronic, and St. Jude. Dr. Stavrakis, Dr, Jessup, and Dr. Di Biase. Dr. Hunter has received research funding, educational grants, and speakers fees from Biosense Webster and Medtronic. Dr. Packer had received honoraria from Biotronik and MediaSphere Medical and research support from several companies. Dr. Piccini has been a consultant to Allergan, Biotronik, Medtronic, Phillips, and Sanofi Aventis, he has received research funding from Abbott, ARCA biopharma, Boston Scientific, Gilead, and Johnson & Johnson, and he had a financial relationship with GlaxoSmithKline. Dr. Fonarow has been a consultant to Abbott, Amgen, Bayer, Janssen, and Novartis. Dr. Butler has been a consultant to several companies. Dr. Allen has been a consultant to Boston Scientific, Janssen, and Novartis. Dr. Kowey has been a consultant to several companies. Dr. Mann has been a consultant to Bristol-Myers Squibb, Corvia, and Novartis, and an adviser to miRagen. Dr. Camm has been a consultant to several companies.
This is part one of a two-part article.
Roughly a third of patients with heart failure also have atrial fibrillation, a comorbid combination notorious for working synergistically to worsen a patient’s quality of life and life expectancy.
During the past year, radiofrequency catheter ablation of atrial fibrillation in patients with both conditions has gathered steam as a way to intervene in at least selected patients, driven by study results that featured attention-grabbing reductions in death and cardiovascular hospitalizations.
The evidence favoring catheter ablation of atrial fibrillation (AFib) in patients with heart failure, particularly patients with heart failure with reduced ejection fraction (HFrEF), ramped up in 2019, spurred largely by a subgroup analysis from the CABANA trial, the largest randomized comparison by far of AFib ablation with antiarrhythmic drug treatment with 2,204 patients.
The past few months also featured release of two meta-analyses that took the CABANA results into account plus findings from about a dozen earlier randomized studies. Both meta-analyses, as well as the heart failure analysis from CABANA, all point in one direction, as stated in the conclusion of one of the meta-analyses: “In patients with AFib, catheter ablation is associated with all-cause mortality benefit, compared with medical therapy, that is driven by patients with AFib and HFrEF. Catheter ablation is safe and reduces cardiovascular hospitalizations and recurrences of atrial arrhythmias” both in patients with paroxysmal and persistent AFib,” wrote Stavros Stavrakis, MD, and his associates in their systematic review of 18 randomized, controlled trials of catheter ablation of AFib in a total of 4,464 patients with or without heart failure (Circ Arrhythm Electrophysiol. 2019 Sep;12[9]: e007414).
Despite these new data and analyses, clinicians seem to have very mixed reactions. Some call for an upgraded recommendation by professional societies that would support more aggressive use of AFib ablation in heart failure patients, and the anecdotal impressions of people who manage these patients are that ablation procedures have recently increased. But others advise caution, and note that in their opinion the efficacy data remain preliminary; the procedure has safety, logistical, and economic concerns; and questions remain about the ability of all active ablation programs to consistently deliver the results seen in published trials.
The meta-analysis led by Dr. Stavrakis showed that catheter ablation of AFib cut all-cause mortality during follow-up by a statistically significant 31%, compared with medical therapy, in all patients regardless of their heart failure status. But in patients with HFrEF, the reduction was 48%, along with a 38% cut in cardiovascular hospitalizations. In contrast, patients without heart failure who underwent AFib ablation showed no significant change in their all-cause mortality, compared with medical management of these patients.
“Based both on our meta-analysis and the CABANA data, patients with AFib most likely to benefit from ablation are patients younger than 65 and those with heart failure,” summed up Dr. Stavrakis, a cardiac electrophysiologist at the Heart Rhythm Institute of the University of Oklahoma in Oklahoma City.
The second meta-analysis, which initially appeared in July, analyzed data from 11 randomized trials of catheter ablations compared with anti-arrhythmic medical therapy for rate or rhythm control with in a total of 3,598 patients who all had heart failure, again including the patients enrolled in the CABANA study. The results showed a significant 49% relative drop in all cause mortality with ablation compared with medical treatment, and a statistically significant 56% cut in hospitalizations, as well as a significant, nearly 7% average, absolute improvement in left ventricular ejection fraction, plus benefits for preventing arrhythmia recurrence and improving quality of life (Eur Heart J. 2019 Jul 11. doi: 10.1093/eurheartj/ehz443).
“The magnitude of the effect seen in the meta-analysis, a 49% reduction in total mortality and a 56% reduction in hospitalizations, is rather staggering, and is larger than typically quoted for other medical interventions or device therapy in heart failure. The treatment effect was uniform among studies, and entirely compatible with the changes in left ventricular function, exercise capacity, and heart failure symptoms. Therefore, although more data are desirable, there are already arguably sufficient data to understand a great deal regarding the impact of a fib ablation,” commented Ross J. Hunter, MRCP, a cardiac electrophysiologist at Barts Heart Centre in London, and his associates in an editorial about this meta-analysis (Eur Heart J. 2019 Oct 22. doi: 10.1093/eurheartj/ehz704).
The heart failure analysis of CABANA (Catheter Ablation vs. Anti-Arrhythmic Drug Therapy for Atrial Fibrillation Trial) itself also showed striking findings when first reported at the annual scientific sessions of the Heart Rhythm Society last May. In presentations he made at this meeting, Douglas L. Packer, MD, CABANA’s lead investigator, reported details of a prespecified subgroup analysis of the 778 patients enrolled in CABANA who had heart failure at baseline, slightly more than a third of the total study enrollment. This was more than double the number of patients identified as specifically having heart failure at entry in the initial publication of CABANA’s findings (JAMA. 2019 Mar 15;321[134]:1261-74). Comparison of the 378 patients with heart failure and randomized to undergo ablation with the 400 with heart failure randomized to medical treatment showed a 36% reduction in the study’s primary, composite endpoint relative to the control group in an intention-to-treat analysis, and a 43% relative cut in all-cause mortality during follow-up, Dr. Packer reported at the May meeting. (As of early November 2019, these results had not yet appeared in a published article.) In contrast, in the 1,422 CABANA patients randomized who did not have heart failure, ablation produced results for these endpoints that were similar to and not statistically different from the outcomes in patients treated medically, said Dr. Packer, a cardiac electrophysiologist and professor of medicine at the Mayo Clinic in Rochester, Minn.
The CABANA results added to what had been previously reported from two other landmark studies that documented incremental efficacy of AFib ablation compared with medical treatment in patients with heart failure: The AATAC (Ablation vs Amiodarone for Treatment of AFib in Patients With Congestive HF and an Implanted Device) study, which randomized 203 patients (Circulation. 2016 Apr 26;133[17]:1637-44), and CASTLE-AF (Catheter Ablation vs. Standard Conventional Therapy in Patients with Left Ventricular Dysfunction and Atrial Fibrillation) trial, which randomized 363 patients (N Engl J Med. 2018 Feb 1;378[5]:417-27). These three studies contributed the most patients and outcomes to the two recent meta-analyses.
“The CASTLE-AF and AATAC trials both showed improved cardiovascular outcomes with ablation in patients with heart failure and AFib. The meta-analysis [by Dr. Stavrakis and his associates] and CABANA subgroup analysis further support use of catheter ablation to improve the outcomes in these patients,” noted Jonathan P. Piccini, MD, a cardiac electrophysiologist at Duke University, Durham, N.C., and a CABANA coinvestigator.
“The CABANA trial was very important because it confirmed the safety of catheter ablation, and more importantly suggested that patients with heart failure may benefit the most [from AFib ablation]. The evidence is very strong to advocate ablation as first-line therapy for selected patients with heart failure. Perhaps the optimal patients are those with [New York Heart Association] class I-III or ambulatory class IV heart failure who are on optimized, guideline-directed medical therapy. We have enough data to make this a class I recommendation. The question that remains is whether this is a cost effective strategy. Because it lowers rehospitalization and death, I suspect it is,” said Luigi Di Biase, MD, lead investigator of AATAC, and director of arrhythmia services at Montefiore Medical Center and professor of medicine at Albert Einstein College of Medicine, both in New York.
Opinions differ on AFib ablation’s role
Despite this expansive assessment of the current status of AFib ablation for patients with heart failure from Dr. Di Biase and shared by others, another camp of cardiologists currently sees ablation as having more limited current utility, as recommended earlier this year by a guideline-update panel representing the American Heart Association, the American College of Cardiology, and the Heart Rhythm Society. The guideline update included this new recommendation for how to use AFib ablation in heart failure patients: “AF catheter ablation may be reasonable in selected patients with symptomatic AFib and heart failure with reduced left ventricular ejection fraction to potentially lower mortality rate and reduce hospitalization for heart failure,” a class IIb recommendation. (J Am Coll Cardiol. 2019 Jul 9;74[1]:104-32). The guideline’s text cited the findings from AATAC and CASTLE-AF, but qualified both studies as “relatively small” and with “highly selected patient populations.” The guideline also incorporated the CABANA results into its considerations (although they may not have had the full analysis in heart failure patients available during their deliberations), but cited the study’s main limitation: CABANA failed to show a statistically significant difference in the primary endpoint in its primary, intention-to-treat analysis, which meant that by the strict statistical criteria that trialists apply to study findings, all other endpoints analyzed using CABANA’s are merely “hypothesis generating” and not definitive.
Questions about the extent of patient selection required to see a clear clinical-endpoint benefit from AFib ablation in heart failure patients, as well as the flawed validity of the CABANA results for making unqualified practice recommendation are the main arguments advanced by experts who caution against broader and more routine ablations.
“The findings from the heart failure subgroup of CABANA are hypothesis generating rather than definitive. Even with the recent meta-analysis, uncertainty remains regarding the ability of catheter ablation to improve outcomes beyond reducing AFib-related symptoms,” commented Gregg Fonarow, MD, a heart failure physician and professor of medicine at the University of California, Los Angeles.
“CASTLE-HF had fewer than 100 deaths combined in both arms, which means very unstable results. We don’t know a lot of detail about the heart failure patients in CABANA, and overall we do not have much data from patients with heart failure with preserved ejection fraction [HFpEF],” said Javed Butler, MD, a heart failure physician and professor and chairman of medicine at the University of Mississippi in Jackson. Dr. Butler also voiced his concerns (shared by other heart failure specialists) about the safety of ablation in heart failure patients, noting that “many patients require multiple ablations; many burns result in scarring and can worsen atrial function. In short, ablation of AFib is probably good for selected patients, but to have a class 1 recommendation, we need much larger trials with well-phenotyped heart failure patients,” Dr. Butler said in an interview.
“The totality of data still captures a relatively small number of patients. CASTLE-HF took 8 years to enroll fewer than 400 patients, and the results showed some heterogeneity. Study patients were a decade younger than average HFrEF patients in the community, and thus the effectiveness and safety of catheter ablation in people with more comorbidity and frailty remains in question. Certain HFrEF patients may be less likely to benefit, such as those with amyloid cardiomyopathy. And with the increasing availability of other treatments for HFrEF such as sacubitril/valsartan, dapagliflozin, and MitraClip, it is less clear how catheter ablation would [benefit patients] on top of what is now current best therapy,” said Larry Allen, MD, a heart failure physician and professor of medicine at the University of Colorado in Aurora.
“With these limitations and the fact that catheter ablation is not a simple procedure, a large randomized, controlled trial of ablation, compared with no ablation, in a wide range of HFrEF patients on contemporary therapy would be welcome,” Dr. Allen said. “Given the prevalence of heart failure and AFib and the potential positive and negative implications of catheter ablation running such a trial seems critical for patients and for society.”
“For ablation of AFib in heart failure to become a class I recommendation there will need to be results from larger randomized studies,” summed up Dr. Stavrakis. The meta-analysis that he coauthored noted that “the benefits of catheter ablation for AFib in HFrEF patients have been consistently shown for over a decade now; however, the uptake of this procedure by clinicians in practice has been slow.”
Despite this history of reticence and ongoing caution about ablation, some cardiology experts see the indications for AFib ablation in heart failure steadily creeping forward, buoyed by a safety record that has more benign than ablation’s reputation suggests.
The CABANA results showed that “ablation is remarkably safe in the hands of experienced clinicians, with risks comparable to anti-arrhythmic drugs,” said Peter R. Kowey, MD, a specialist in treating AFib and professor of medicine at Thomas Jefferson University in Philadelphia, who made this assessment during a talk at an AFib meeting in early 2019. Dr. Kowey’s take on what the CABANA safety data showed contrasts with the impression of other cardiologists who are wary of perceived dangers from ablation.
“Ablation comes with a lot of morbidity and mortality. It’s not that the idea of ablation is wrong, but the ability to do it without a lot of adverse effects. ... We’re not quite there yet,” said Douglas L. Mann, MD, a heart failure physician and professor of medicine and chief of the cardiovascular division at Washington University in St. Louis.
“If I had a patient with HFrEF and AFib who was really sick, I’m not so sure I’d send them for ablation, which is not a simple procedure. The patients we tend to send for ablation are selected. Ablation is a big undertaking in patients who are already sick, and it’s expensive. I don’t think the data we have now will change the consensus view, but every heart failure physician is sending some patients for AFib ablation. People are turning to AFib ablation earlier than before. I think the consensus is that ablation is for symptoms or poor rate control, not for better outcomes,” said Mariell Jessup, MD, a heart failure specialist and chief science and medical officer of the American Heart Association in Dallas.
However, this caution about safety and skepticism over efficacy may be dissipating as experience with ablation accumulates.
“CASTLE-AF and other data, including evidence for the apparent isolation of beta-blocker benefit to patients in sinus rhythm, have made me much more proactive about considering catheter ablation in my HFrEF patients. I think many other cardiologists have a similar view,” said Dr. Allen in an interview.
“A lot [of heart failure] patients are [being] referred for ablation, depending on the practice, setting, the local availability of electrophysiologists, and patient interest in ablation,” said Dr. Butler.
“We have no absolutely compelling data, but the data we have all point in the same direction. Like most, I am becoming convinced that AFib ablation in heart failure patients is a very valuable method for managing patients, but I can’t point to one study that was conclusive. Results from lots of studies show that it is likely, and when you add them all together it looks indisputable,” commented A. John Camm, MD, an atrial fibrillation specialist and professor of clinical cardiology at St. George’s University in London. “The findings put a responsibility on cardiologists to assess patients with heart failure for AFib. But there are nothing like enough resources to deal with all the patients who have heart failure who also have AFib.”
A rough estimate of just the U.S. volume of patients with heart failure and AFib is likely in the ball park of 2 million people (a third of the estimated 6 million American currently living with heart failure), and with the prevalence of each of these disorders rising precipitously (more than 5 million Americans have AFib) the confluence of the two should also show a steady increase. “It will take a major change in our concept of heart failure management to really address this. Potentially it would mean a large increase in the number of RF ablations of AFib, but the resources for that are not now present,” Dr. Camm said in an interview.
The attractions of catheter ablation also stand in contrast to the limitations of alternative treatments. Ablation is effective in a majority of patients for reducing AFib burden, both the frequency and duration of AFib episodes, and safety issues are mostly limited to the procedural and immediate postprocedural periods. The drugs available for trying to control AFib are beta-blockers, which provide rate control and can help prevent AFib onset, and rhythm-controlling anti-arrhythmic drugs like amiodarone, which have substantial limitations in both their ability to prevent arrhythmia recurrences as well as for safety.
“Most of the conventional antiarrhythmic drugs are contraindicated, frequently ineffective, or not well tolerated in patients with HFrEF. Catheter ablation of AFib provides an increasingly important option for rhythm control in these patients without using antiarrhythmic drugs,” Dr. Di Biase and his associates wrote in a recent review of AFib ablation in heart failure patients (Eur Heart J. 2019 Feb 21;40[8]:663-71).
“The guidelines that are controversial still make amiodarone a class I drug even though it’s been associated with serious side effects and has been shown in several heart failure trials to increase mortality. I can’t believe that ablation is a class IIb recommendation while a drug like amiodarone is a class I recommendation,” Dr. Di Biase said.
And although beta-blockers are a mainstay of heart failure treatment, once AFib becomes established they are less useful for maintaining sinus rhythm. “Beta-blockers provide effective rate control, but they can’t convert patients to sinus rhythm [once AFib begins], and there is no convincing evidence that patients on beta-blockers stay in sinus rhythm longer. You can’t just say: the patient is on a beta-blocker so I’ve done my best,” noted Dr. Jessup.
CABANA received funding from Biosense Webster, Boston Scientific, Medtronic, and St. Jude. Dr. Stavrakis, Dr, Jessup, and Dr. Di Biase. Dr. Hunter has received research funding, educational grants, and speakers fees from Biosense Webster and Medtronic. Dr. Packer had received honoraria from Biotronik and MediaSphere Medical and research support from several companies. Dr. Piccini has been a consultant to Allergan, Biotronik, Medtronic, Phillips, and Sanofi Aventis, he has received research funding from Abbott, ARCA biopharma, Boston Scientific, Gilead, and Johnson & Johnson, and he had a financial relationship with GlaxoSmithKline. Dr. Fonarow has been a consultant to Abbott, Amgen, Bayer, Janssen, and Novartis. Dr. Butler has been a consultant to several companies. Dr. Allen has been a consultant to Boston Scientific, Janssen, and Novartis. Dr. Kowey has been a consultant to several companies. Dr. Mann has been a consultant to Bristol-Myers Squibb, Corvia, and Novartis, and an adviser to miRagen. Dr. Camm has been a consultant to several companies.
This is part one of a two-part article.
Roughly a third of patients with heart failure also have atrial fibrillation, a comorbid combination notorious for working synergistically to worsen a patient’s quality of life and life expectancy.
During the past year, radiofrequency catheter ablation of atrial fibrillation in patients with both conditions has gathered steam as a way to intervene in at least selected patients, driven by study results that featured attention-grabbing reductions in death and cardiovascular hospitalizations.
The evidence favoring catheter ablation of atrial fibrillation (AFib) in patients with heart failure, particularly patients with heart failure with reduced ejection fraction (HFrEF), ramped up in 2019, spurred largely by a subgroup analysis from the CABANA trial, the largest randomized comparison by far of AFib ablation with antiarrhythmic drug treatment with 2,204 patients.
The past few months also featured release of two meta-analyses that took the CABANA results into account plus findings from about a dozen earlier randomized studies. Both meta-analyses, as well as the heart failure analysis from CABANA, all point in one direction, as stated in the conclusion of one of the meta-analyses: “In patients with AFib, catheter ablation is associated with all-cause mortality benefit, compared with medical therapy, that is driven by patients with AFib and HFrEF. Catheter ablation is safe and reduces cardiovascular hospitalizations and recurrences of atrial arrhythmias” both in patients with paroxysmal and persistent AFib,” wrote Stavros Stavrakis, MD, and his associates in their systematic review of 18 randomized, controlled trials of catheter ablation of AFib in a total of 4,464 patients with or without heart failure (Circ Arrhythm Electrophysiol. 2019 Sep;12[9]: e007414).
Despite these new data and analyses, clinicians seem to have very mixed reactions. Some call for an upgraded recommendation by professional societies that would support more aggressive use of AFib ablation in heart failure patients, and the anecdotal impressions of people who manage these patients are that ablation procedures have recently increased. But others advise caution, and note that in their opinion the efficacy data remain preliminary; the procedure has safety, logistical, and economic concerns; and questions remain about the ability of all active ablation programs to consistently deliver the results seen in published trials.
The meta-analysis led by Dr. Stavrakis showed that catheter ablation of AFib cut all-cause mortality during follow-up by a statistically significant 31%, compared with medical therapy, in all patients regardless of their heart failure status. But in patients with HFrEF, the reduction was 48%, along with a 38% cut in cardiovascular hospitalizations. In contrast, patients without heart failure who underwent AFib ablation showed no significant change in their all-cause mortality, compared with medical management of these patients.
“Based both on our meta-analysis and the CABANA data, patients with AFib most likely to benefit from ablation are patients younger than 65 and those with heart failure,” summed up Dr. Stavrakis, a cardiac electrophysiologist at the Heart Rhythm Institute of the University of Oklahoma in Oklahoma City.
The second meta-analysis, which initially appeared in July, analyzed data from 11 randomized trials of catheter ablations compared with anti-arrhythmic medical therapy for rate or rhythm control with in a total of 3,598 patients who all had heart failure, again including the patients enrolled in the CABANA study. The results showed a significant 49% relative drop in all cause mortality with ablation compared with medical treatment, and a statistically significant 56% cut in hospitalizations, as well as a significant, nearly 7% average, absolute improvement in left ventricular ejection fraction, plus benefits for preventing arrhythmia recurrence and improving quality of life (Eur Heart J. 2019 Jul 11. doi: 10.1093/eurheartj/ehz443).
“The magnitude of the effect seen in the meta-analysis, a 49% reduction in total mortality and a 56% reduction in hospitalizations, is rather staggering, and is larger than typically quoted for other medical interventions or device therapy in heart failure. The treatment effect was uniform among studies, and entirely compatible with the changes in left ventricular function, exercise capacity, and heart failure symptoms. Therefore, although more data are desirable, there are already arguably sufficient data to understand a great deal regarding the impact of a fib ablation,” commented Ross J. Hunter, MRCP, a cardiac electrophysiologist at Barts Heart Centre in London, and his associates in an editorial about this meta-analysis (Eur Heart J. 2019 Oct 22. doi: 10.1093/eurheartj/ehz704).
The heart failure analysis of CABANA (Catheter Ablation vs. Anti-Arrhythmic Drug Therapy for Atrial Fibrillation Trial) itself also showed striking findings when first reported at the annual scientific sessions of the Heart Rhythm Society last May. In presentations he made at this meeting, Douglas L. Packer, MD, CABANA’s lead investigator, reported details of a prespecified subgroup analysis of the 778 patients enrolled in CABANA who had heart failure at baseline, slightly more than a third of the total study enrollment. This was more than double the number of patients identified as specifically having heart failure at entry in the initial publication of CABANA’s findings (JAMA. 2019 Mar 15;321[134]:1261-74). Comparison of the 378 patients with heart failure and randomized to undergo ablation with the 400 with heart failure randomized to medical treatment showed a 36% reduction in the study’s primary, composite endpoint relative to the control group in an intention-to-treat analysis, and a 43% relative cut in all-cause mortality during follow-up, Dr. Packer reported at the May meeting. (As of early November 2019, these results had not yet appeared in a published article.) In contrast, in the 1,422 CABANA patients randomized who did not have heart failure, ablation produced results for these endpoints that were similar to and not statistically different from the outcomes in patients treated medically, said Dr. Packer, a cardiac electrophysiologist and professor of medicine at the Mayo Clinic in Rochester, Minn.
The CABANA results added to what had been previously reported from two other landmark studies that documented incremental efficacy of AFib ablation compared with medical treatment in patients with heart failure: The AATAC (Ablation vs Amiodarone for Treatment of AFib in Patients With Congestive HF and an Implanted Device) study, which randomized 203 patients (Circulation. 2016 Apr 26;133[17]:1637-44), and CASTLE-AF (Catheter Ablation vs. Standard Conventional Therapy in Patients with Left Ventricular Dysfunction and Atrial Fibrillation) trial, which randomized 363 patients (N Engl J Med. 2018 Feb 1;378[5]:417-27). These three studies contributed the most patients and outcomes to the two recent meta-analyses.
“The CASTLE-AF and AATAC trials both showed improved cardiovascular outcomes with ablation in patients with heart failure and AFib. The meta-analysis [by Dr. Stavrakis and his associates] and CABANA subgroup analysis further support use of catheter ablation to improve the outcomes in these patients,” noted Jonathan P. Piccini, MD, a cardiac electrophysiologist at Duke University, Durham, N.C., and a CABANA coinvestigator.
“The CABANA trial was very important because it confirmed the safety of catheter ablation, and more importantly suggested that patients with heart failure may benefit the most [from AFib ablation]. The evidence is very strong to advocate ablation as first-line therapy for selected patients with heart failure. Perhaps the optimal patients are those with [New York Heart Association] class I-III or ambulatory class IV heart failure who are on optimized, guideline-directed medical therapy. We have enough data to make this a class I recommendation. The question that remains is whether this is a cost effective strategy. Because it lowers rehospitalization and death, I suspect it is,” said Luigi Di Biase, MD, lead investigator of AATAC, and director of arrhythmia services at Montefiore Medical Center and professor of medicine at Albert Einstein College of Medicine, both in New York.
Opinions differ on AFib ablation’s role
Despite this expansive assessment of the current status of AFib ablation for patients with heart failure from Dr. Di Biase and shared by others, another camp of cardiologists currently sees ablation as having more limited current utility, as recommended earlier this year by a guideline-update panel representing the American Heart Association, the American College of Cardiology, and the Heart Rhythm Society. The guideline update included this new recommendation for how to use AFib ablation in heart failure patients: “AF catheter ablation may be reasonable in selected patients with symptomatic AFib and heart failure with reduced left ventricular ejection fraction to potentially lower mortality rate and reduce hospitalization for heart failure,” a class IIb recommendation. (J Am Coll Cardiol. 2019 Jul 9;74[1]:104-32). The guideline’s text cited the findings from AATAC and CASTLE-AF, but qualified both studies as “relatively small” and with “highly selected patient populations.” The guideline also incorporated the CABANA results into its considerations (although they may not have had the full analysis in heart failure patients available during their deliberations), but cited the study’s main limitation: CABANA failed to show a statistically significant difference in the primary endpoint in its primary, intention-to-treat analysis, which meant that by the strict statistical criteria that trialists apply to study findings, all other endpoints analyzed using CABANA’s are merely “hypothesis generating” and not definitive.
Questions about the extent of patient selection required to see a clear clinical-endpoint benefit from AFib ablation in heart failure patients, as well as the flawed validity of the CABANA results for making unqualified practice recommendation are the main arguments advanced by experts who caution against broader and more routine ablations.
“The findings from the heart failure subgroup of CABANA are hypothesis generating rather than definitive. Even with the recent meta-analysis, uncertainty remains regarding the ability of catheter ablation to improve outcomes beyond reducing AFib-related symptoms,” commented Gregg Fonarow, MD, a heart failure physician and professor of medicine at the University of California, Los Angeles.
“CASTLE-HF had fewer than 100 deaths combined in both arms, which means very unstable results. We don’t know a lot of detail about the heart failure patients in CABANA, and overall we do not have much data from patients with heart failure with preserved ejection fraction [HFpEF],” said Javed Butler, MD, a heart failure physician and professor and chairman of medicine at the University of Mississippi in Jackson. Dr. Butler also voiced his concerns (shared by other heart failure specialists) about the safety of ablation in heart failure patients, noting that “many patients require multiple ablations; many burns result in scarring and can worsen atrial function. In short, ablation of AFib is probably good for selected patients, but to have a class 1 recommendation, we need much larger trials with well-phenotyped heart failure patients,” Dr. Butler said in an interview.
“The totality of data still captures a relatively small number of patients. CASTLE-HF took 8 years to enroll fewer than 400 patients, and the results showed some heterogeneity. Study patients were a decade younger than average HFrEF patients in the community, and thus the effectiveness and safety of catheter ablation in people with more comorbidity and frailty remains in question. Certain HFrEF patients may be less likely to benefit, such as those with amyloid cardiomyopathy. And with the increasing availability of other treatments for HFrEF such as sacubitril/valsartan, dapagliflozin, and MitraClip, it is less clear how catheter ablation would [benefit patients] on top of what is now current best therapy,” said Larry Allen, MD, a heart failure physician and professor of medicine at the University of Colorado in Aurora.
“With these limitations and the fact that catheter ablation is not a simple procedure, a large randomized, controlled trial of ablation, compared with no ablation, in a wide range of HFrEF patients on contemporary therapy would be welcome,” Dr. Allen said. “Given the prevalence of heart failure and AFib and the potential positive and negative implications of catheter ablation running such a trial seems critical for patients and for society.”
“For ablation of AFib in heart failure to become a class I recommendation there will need to be results from larger randomized studies,” summed up Dr. Stavrakis. The meta-analysis that he coauthored noted that “the benefits of catheter ablation for AFib in HFrEF patients have been consistently shown for over a decade now; however, the uptake of this procedure by clinicians in practice has been slow.”
Despite this history of reticence and ongoing caution about ablation, some cardiology experts see the indications for AFib ablation in heart failure steadily creeping forward, buoyed by a safety record that has more benign than ablation’s reputation suggests.
The CABANA results showed that “ablation is remarkably safe in the hands of experienced clinicians, with risks comparable to anti-arrhythmic drugs,” said Peter R. Kowey, MD, a specialist in treating AFib and professor of medicine at Thomas Jefferson University in Philadelphia, who made this assessment during a talk at an AFib meeting in early 2019. Dr. Kowey’s take on what the CABANA safety data showed contrasts with the impression of other cardiologists who are wary of perceived dangers from ablation.
“Ablation comes with a lot of morbidity and mortality. It’s not that the idea of ablation is wrong, but the ability to do it without a lot of adverse effects. ... We’re not quite there yet,” said Douglas L. Mann, MD, a heart failure physician and professor of medicine and chief of the cardiovascular division at Washington University in St. Louis.
“If I had a patient with HFrEF and AFib who was really sick, I’m not so sure I’d send them for ablation, which is not a simple procedure. The patients we tend to send for ablation are selected. Ablation is a big undertaking in patients who are already sick, and it’s expensive. I don’t think the data we have now will change the consensus view, but every heart failure physician is sending some patients for AFib ablation. People are turning to AFib ablation earlier than before. I think the consensus is that ablation is for symptoms or poor rate control, not for better outcomes,” said Mariell Jessup, MD, a heart failure specialist and chief science and medical officer of the American Heart Association in Dallas.
However, this caution about safety and skepticism over efficacy may be dissipating as experience with ablation accumulates.
“CASTLE-AF and other data, including evidence for the apparent isolation of beta-blocker benefit to patients in sinus rhythm, have made me much more proactive about considering catheter ablation in my HFrEF patients. I think many other cardiologists have a similar view,” said Dr. Allen in an interview.
“A lot [of heart failure] patients are [being] referred for ablation, depending on the practice, setting, the local availability of electrophysiologists, and patient interest in ablation,” said Dr. Butler.
“We have no absolutely compelling data, but the data we have all point in the same direction. Like most, I am becoming convinced that AFib ablation in heart failure patients is a very valuable method for managing patients, but I can’t point to one study that was conclusive. Results from lots of studies show that it is likely, and when you add them all together it looks indisputable,” commented A. John Camm, MD, an atrial fibrillation specialist and professor of clinical cardiology at St. George’s University in London. “The findings put a responsibility on cardiologists to assess patients with heart failure for AFib. But there are nothing like enough resources to deal with all the patients who have heart failure who also have AFib.”
A rough estimate of just the U.S. volume of patients with heart failure and AFib is likely in the ball park of 2 million people (a third of the estimated 6 million American currently living with heart failure), and with the prevalence of each of these disorders rising precipitously (more than 5 million Americans have AFib) the confluence of the two should also show a steady increase. “It will take a major change in our concept of heart failure management to really address this. Potentially it would mean a large increase in the number of RF ablations of AFib, but the resources for that are not now present,” Dr. Camm said in an interview.
The attractions of catheter ablation also stand in contrast to the limitations of alternative treatments. Ablation is effective in a majority of patients for reducing AFib burden, both the frequency and duration of AFib episodes, and safety issues are mostly limited to the procedural and immediate postprocedural periods. The drugs available for trying to control AFib are beta-blockers, which provide rate control and can help prevent AFib onset, and rhythm-controlling anti-arrhythmic drugs like amiodarone, which have substantial limitations in both their ability to prevent arrhythmia recurrences as well as for safety.
“Most of the conventional antiarrhythmic drugs are contraindicated, frequently ineffective, or not well tolerated in patients with HFrEF. Catheter ablation of AFib provides an increasingly important option for rhythm control in these patients without using antiarrhythmic drugs,” Dr. Di Biase and his associates wrote in a recent review of AFib ablation in heart failure patients (Eur Heart J. 2019 Feb 21;40[8]:663-71).
“The guidelines that are controversial still make amiodarone a class I drug even though it’s been associated with serious side effects and has been shown in several heart failure trials to increase mortality. I can’t believe that ablation is a class IIb recommendation while a drug like amiodarone is a class I recommendation,” Dr. Di Biase said.
And although beta-blockers are a mainstay of heart failure treatment, once AFib becomes established they are less useful for maintaining sinus rhythm. “Beta-blockers provide effective rate control, but they can’t convert patients to sinus rhythm [once AFib begins], and there is no convincing evidence that patients on beta-blockers stay in sinus rhythm longer. You can’t just say: the patient is on a beta-blocker so I’ve done my best,” noted Dr. Jessup.
CABANA received funding from Biosense Webster, Boston Scientific, Medtronic, and St. Jude. Dr. Stavrakis, Dr, Jessup, and Dr. Di Biase. Dr. Hunter has received research funding, educational grants, and speakers fees from Biosense Webster and Medtronic. Dr. Packer had received honoraria from Biotronik and MediaSphere Medical and research support from several companies. Dr. Piccini has been a consultant to Allergan, Biotronik, Medtronic, Phillips, and Sanofi Aventis, he has received research funding from Abbott, ARCA biopharma, Boston Scientific, Gilead, and Johnson & Johnson, and he had a financial relationship with GlaxoSmithKline. Dr. Fonarow has been a consultant to Abbott, Amgen, Bayer, Janssen, and Novartis. Dr. Butler has been a consultant to several companies. Dr. Allen has been a consultant to Boston Scientific, Janssen, and Novartis. Dr. Kowey has been a consultant to several companies. Dr. Mann has been a consultant to Bristol-Myers Squibb, Corvia, and Novartis, and an adviser to miRagen. Dr. Camm has been a consultant to several companies.
This is part one of a two-part article.
Data build on cardiovascular disease risk after GDM, HDP
WASHINGTON – Cardiovascular risk factors may be elevated “as soon as the first postpartum year” in women who have gestational diabetes or hypertensive disorders of pregnancy, recent findings have affirmed, Deborah B. Ehrenthal, MD, MPH, said at the biennial meeting of the Diabetes in Pregnancy Study Group of North America.
Dr. Ehrenthal was one of several researchers who urged innovative strategies and improved care coordination to boost women’s follow-up after gestational diabetes mellitus (GDM) and other adverse pregnancy outcomes and complications. “The metabolic stress of pregnancy can uncover underlying susceptibilities,” she said. “And ”
Evidence that adverse pregnancy outcomes – including GDM and hypertensive disorders of pregnancy (HDP) – can elevate cardiovascular risk comes most recently from the Nulliparous Pregnancy Outcomes Study – Monitoring Mothers to be Heart Health Study (nuMoM2b–HHS study), a prospective observational cohort that followed 4,484 women 2-7 years after their first pregnancy. Women had a follow-up exam, with blood pressure and anthropometric measurements and clinical/biological testing, an average of 3 years post partum.
An analysis published in October 2019 in the Journal of the American Heart Association shows that women with HDP (including preeclampsia and gestational hypertension) had a relative risk of hypertension of 2.5 at follow-up, compared with women without HDP. Women who had preeclampsia specifically were 2.3 times as likely as were women who did not have preeclampsia to have incident hypertension at follow-up, said Dr. Ehrenthal, a coinvestigator of the study.
The analysis focused on incident hypertension as the primary outcome, and adjusted for age, body mass index, and other important cardiovascular disease risk factors, she noted. Researchers utilized the diagnostic threshold for hypertension extant at the time of study design: A systolic blood pressure of 140 mm Hg or greater, or a diastolic BP of 90 mm Hg or greater (J Am Heart Assoc. 2019;8:e013092).
HDP was the most common adverse pregnancy outcome in the nuMoM2b–HHS study (14%). Among all participants, 4% had GDM. Approximately 82% had neither HDP nor GDM. Other adverse pregnancy outcomes included in the analysis were preterm birth, small-for-gestational-age birth, and stillbirth.
Additional preliminary estimates presented by Dr. Ehrenthal show that, based on the new (2017) lower threshold for hypertension – 130 mg Hg systolic or 80 mm Hg diastolic – the disorder afflicted 37% of women who had experienced HDP (relative risk 2.1), and 32% of women who had GDM (RR 1.8). Prediabetes/diabetes (using a fasting blood glucose threshold of 100 mg/dL) at follow-up affected an estimated 21% of women who had HDP (RR 1.4) and 38% of women who had GDM (RR 2.5).
Notably, across the entire study cohort, 20% had hypertension at follow-up, “which is extraordinary” considering the short time frame from pregnancy and the young age of the study population – a mean maternal age of 27 years, said Dr. Ehrenthal, associate professor of population health sciences and obstetrics & gynecology at the University of Wisconsin, Madison.
Also across the cohort, 15% had prediabetes/diabetes at follow-up. “We need to think about women more generally,” she cautioned. “While we recognize the significant elevated risk of HDP and GDM [for the development of subsequent hypertension and cardiovascular risk], we will miss a lot of women [if we focus only on the history of HDP and GDM.]”
The majority of women found to have hypertension or prediabetes/diabetes at follow-up had experienced neither HDP nor GDM, but a good many of them (47% of those who had hypertension and 47% of those found to have prediabetes/diabetes) had a BMI of 30 or above, Dr. Ehrenthal said at the DPSG-NA meeting.
Nurses Health Study, hyperglycemia and adverse pregnancy outcome follow-up data
The new findings from the nuMoM2b–HHS study add to a robust and growing body of evidence that pregnancy is an important window to future health, and that follow up and screening after GDM and HDP are crucial.
Regarding GDM specifically, “there’s quite a bit of literature by now demonstrating that GDM history is a risk factor for hypertension, even 1-2 years post partum, and that the risk is elevated as well for dyslipidemia and vascular dysfunction,” Deirdre K. Tobias, D.Sc., an epidemiologist at Brigham and Women’s Hospital and assistant professor of nutrition at Harvard TH Chan School of Public Health, Boston, said at the DPSG meeting.
An analysis of the Nurses Health Study II (NHS II) cohort published in 2017 found a 40% higher relative risk of cardiovascular disease events (largely myocardial infarction) in women who had GDM, compared with women who did not have GDM over a median follow-up of 26 years. This was after adjustments were made for age, time since pregnancy, menopausal status, family history of MI or stroke, hypertension in pregnancy, white race/ethnicity, prepregnancy BMI, and other factors (JAMA Intern Med. 2017;177[12]:1735-42).
The NHS data also have shown, however, that the elevated risk for cardiovascular disease after a GDM pregnancy “can be mitigated by adopting a healthy lifestyle,” said Dr. Tobias, lead author of the 2017 NHS II analysis. Adjustments for postpregnancy weight gain and lifestyle factors attenuated the relative risk of cardiovascular disease events after a GDM pregnancy to a 30% increased risk.
Dr. Tobias and colleagues currently are looking within the NHS cohort for “metabolomic signatures” or signals – various amino acid and lipid metabolites – to identify the progression of GDM to type 2 diabetes. Metabolomics “may help further refine our understanding of the long-term links between GDM and prevention of type 2 diabetes and of cardiovascular disease in mothers,” she said.
The Hyperglycemia and Adverse Pregnancy Outcome (HAPO) Follow-Up Study, in the meantime, is documenting associations of maternal glucose levels during pregnancy not only with prediabetes or type 2 diabetes 10-14 years later, but also with measures of cardiovascular risk in mothers 10-14 years later.
Just as perinatal outcomes were strongly associated with glucose as a continuous variable in the original HAPO study, “it’s clear there’s a progressive increase in the risk of [later] disorders of glucose metabolism as [fasting blood glucose levels and 1- and-2-hour glucose values] in pregnancy are higher,” said Boyd E. Metzger, MD, the Tom D. Spies emeritus professor of metabolism and nutrition at Northwestern University, Chicago, and principal investigator of the original HAPO study and its follow up.
“Another message is that the more normal you are in pregnancy, the more normal you will be many years later. Good values [during pregnancy] produce good outcomes.”
Currently unpublished data from the HAPO Follow-Up Study are being analyzed, but it appears thus far that GDM is not associated with hypertension (per the old diagnostic threshold) in this cohort after adjustment for maternal age, BMI, smoking, and family history of hypertension. GDM appears to be a significant risk factor for dyslipidemia, however. HDL cholesterol at follow-up was significantly lower for mothers who had GDM compared with those without, whereas LDL cholesterol and triglycerides at follow-up were significantly higher for mothers with GDM, Dr. Metzger said.
Racial/ethnic disparities, postpartum care
Neither long-term study – the NHS II or the HAPO Follow-Up Study – has looked at racial and ethnic differences. The HAPO cohort is racially-ethnically diverse but the NHS II cohort is predominantly white women.
Research suggests that GDM is a heterogeneous condition with some unique phenotypes in subgroups that vary by race and ethnicity. And just as there appear to be racial-ethnic differences in the pathophysiology of GDM, there appear to be racial-ethnic differences in the progression to type 2 diabetes – a known risk factor for cardiovascular disease, said Monique Henderson, PhD, a research scientist at Kaiser Permanente Northern California (KPNC).
On the broadest level, while Asian Americans have the highest prevalence of GDM, African Americans have the highest rates of progressing to type 2 diabetes, Dr. Henderson said. Disparities “may [stem from] metabolic differences in terms of insulin resistance and secretion that are different between pregnancy and the postpartum period, and that might vary [across racial-ethnic subgroups],” she said. Lifestyle differences and variation in postpartum screening rates also may play a role.
At KPNC, where women with GDM receive calls and letters reminding them of the need for postpartum screening, only 48% overall completed an oral glucose tolerance test at 4-12 weeks post partum, as recommended by both the American Diabetes Association and the American College of Obstetricians and Gynecologists. Both before and after adjustment for education, attendance at a postpartum visit, and other variables, Chinese women were most likely to have screening, and black women were least likely, said Dr. Henderson, referring to ongoing research.
A study Dr. Ehrenthal led of women with GDM or HDP recruited from the postpartum service of a large community-based, academic obstetrical hospital in Delaware showed that while nearly all women attended a 6-week postpartum visit with their ob.gyns., 59% of women with GDM had not yet completed diabetes screening when they were interviewed 3 months post partum. Most women with HDP indicated they had follow-up blood pressure testing, and just over half of women with either diagnosis recalled having ever had lipid testing (J Women’s Health 2014;23[9]:760-4).
Women least likely to complete screening tests were those who had no college education, those who had less than a high school level of health literacy, and those who were not privately insured, Dr. Ehrenthal said.
A large national study of privately insured women also found low rates of follow-up testing, however. While the majority of women with GDM had a postpartum visit with an obstetrician or primary care physician within a year after delivery, only a minority of women had a glycemic screening test completed (Obstet Gynecol. 2016;128[1]:159-67).
“We can’t place the blame on women,” Dr. Ehrenthal said. “We need increased attention to screening,” including screening for cardiovascular disease risk factors, and a “deliberate hand-off to primary care.”
For follow-up cardiovascular disease risk factor assessment after HDP, ACOG recommends periodic (perhaps annually) assessment and referral for treatment as needed, and the cardiology professional organizations recommend that pregnancy history be considered when assessing risk in order to decide on lipid treatment, she noted.
Each of the speakers reported that they have no financial or other interests that pose a conflict of interest. The HAPO Follow-Up Study is funded by the National Institute of Diabetes and Digestive and Kidney Diseases, and the nuMoM2b–HHS study has been funded by several National Institutes of Health institutes and other programs and initiatives.
WASHINGTON – Cardiovascular risk factors may be elevated “as soon as the first postpartum year” in women who have gestational diabetes or hypertensive disorders of pregnancy, recent findings have affirmed, Deborah B. Ehrenthal, MD, MPH, said at the biennial meeting of the Diabetes in Pregnancy Study Group of North America.
Dr. Ehrenthal was one of several researchers who urged innovative strategies and improved care coordination to boost women’s follow-up after gestational diabetes mellitus (GDM) and other adverse pregnancy outcomes and complications. “The metabolic stress of pregnancy can uncover underlying susceptibilities,” she said. “And ”
Evidence that adverse pregnancy outcomes – including GDM and hypertensive disorders of pregnancy (HDP) – can elevate cardiovascular risk comes most recently from the Nulliparous Pregnancy Outcomes Study – Monitoring Mothers to be Heart Health Study (nuMoM2b–HHS study), a prospective observational cohort that followed 4,484 women 2-7 years after their first pregnancy. Women had a follow-up exam, with blood pressure and anthropometric measurements and clinical/biological testing, an average of 3 years post partum.
An analysis published in October 2019 in the Journal of the American Heart Association shows that women with HDP (including preeclampsia and gestational hypertension) had a relative risk of hypertension of 2.5 at follow-up, compared with women without HDP. Women who had preeclampsia specifically were 2.3 times as likely as were women who did not have preeclampsia to have incident hypertension at follow-up, said Dr. Ehrenthal, a coinvestigator of the study.
The analysis focused on incident hypertension as the primary outcome, and adjusted for age, body mass index, and other important cardiovascular disease risk factors, she noted. Researchers utilized the diagnostic threshold for hypertension extant at the time of study design: A systolic blood pressure of 140 mm Hg or greater, or a diastolic BP of 90 mm Hg or greater (J Am Heart Assoc. 2019;8:e013092).
HDP was the most common adverse pregnancy outcome in the nuMoM2b–HHS study (14%). Among all participants, 4% had GDM. Approximately 82% had neither HDP nor GDM. Other adverse pregnancy outcomes included in the analysis were preterm birth, small-for-gestational-age birth, and stillbirth.
Additional preliminary estimates presented by Dr. Ehrenthal show that, based on the new (2017) lower threshold for hypertension – 130 mg Hg systolic or 80 mm Hg diastolic – the disorder afflicted 37% of women who had experienced HDP (relative risk 2.1), and 32% of women who had GDM (RR 1.8). Prediabetes/diabetes (using a fasting blood glucose threshold of 100 mg/dL) at follow-up affected an estimated 21% of women who had HDP (RR 1.4) and 38% of women who had GDM (RR 2.5).
Notably, across the entire study cohort, 20% had hypertension at follow-up, “which is extraordinary” considering the short time frame from pregnancy and the young age of the study population – a mean maternal age of 27 years, said Dr. Ehrenthal, associate professor of population health sciences and obstetrics & gynecology at the University of Wisconsin, Madison.
Also across the cohort, 15% had prediabetes/diabetes at follow-up. “We need to think about women more generally,” she cautioned. “While we recognize the significant elevated risk of HDP and GDM [for the development of subsequent hypertension and cardiovascular risk], we will miss a lot of women [if we focus only on the history of HDP and GDM.]”
The majority of women found to have hypertension or prediabetes/diabetes at follow-up had experienced neither HDP nor GDM, but a good many of them (47% of those who had hypertension and 47% of those found to have prediabetes/diabetes) had a BMI of 30 or above, Dr. Ehrenthal said at the DPSG-NA meeting.
Nurses Health Study, hyperglycemia and adverse pregnancy outcome follow-up data
The new findings from the nuMoM2b–HHS study add to a robust and growing body of evidence that pregnancy is an important window to future health, and that follow up and screening after GDM and HDP are crucial.
Regarding GDM specifically, “there’s quite a bit of literature by now demonstrating that GDM history is a risk factor for hypertension, even 1-2 years post partum, and that the risk is elevated as well for dyslipidemia and vascular dysfunction,” Deirdre K. Tobias, D.Sc., an epidemiologist at Brigham and Women’s Hospital and assistant professor of nutrition at Harvard TH Chan School of Public Health, Boston, said at the DPSG meeting.
An analysis of the Nurses Health Study II (NHS II) cohort published in 2017 found a 40% higher relative risk of cardiovascular disease events (largely myocardial infarction) in women who had GDM, compared with women who did not have GDM over a median follow-up of 26 years. This was after adjustments were made for age, time since pregnancy, menopausal status, family history of MI or stroke, hypertension in pregnancy, white race/ethnicity, prepregnancy BMI, and other factors (JAMA Intern Med. 2017;177[12]:1735-42).
The NHS data also have shown, however, that the elevated risk for cardiovascular disease after a GDM pregnancy “can be mitigated by adopting a healthy lifestyle,” said Dr. Tobias, lead author of the 2017 NHS II analysis. Adjustments for postpregnancy weight gain and lifestyle factors attenuated the relative risk of cardiovascular disease events after a GDM pregnancy to a 30% increased risk.
Dr. Tobias and colleagues currently are looking within the NHS cohort for “metabolomic signatures” or signals – various amino acid and lipid metabolites – to identify the progression of GDM to type 2 diabetes. Metabolomics “may help further refine our understanding of the long-term links between GDM and prevention of type 2 diabetes and of cardiovascular disease in mothers,” she said.
The Hyperglycemia and Adverse Pregnancy Outcome (HAPO) Follow-Up Study, in the meantime, is documenting associations of maternal glucose levels during pregnancy not only with prediabetes or type 2 diabetes 10-14 years later, but also with measures of cardiovascular risk in mothers 10-14 years later.
Just as perinatal outcomes were strongly associated with glucose as a continuous variable in the original HAPO study, “it’s clear there’s a progressive increase in the risk of [later] disorders of glucose metabolism as [fasting blood glucose levels and 1- and-2-hour glucose values] in pregnancy are higher,” said Boyd E. Metzger, MD, the Tom D. Spies emeritus professor of metabolism and nutrition at Northwestern University, Chicago, and principal investigator of the original HAPO study and its follow up.
“Another message is that the more normal you are in pregnancy, the more normal you will be many years later. Good values [during pregnancy] produce good outcomes.”
Currently unpublished data from the HAPO Follow-Up Study are being analyzed, but it appears thus far that GDM is not associated with hypertension (per the old diagnostic threshold) in this cohort after adjustment for maternal age, BMI, smoking, and family history of hypertension. GDM appears to be a significant risk factor for dyslipidemia, however. HDL cholesterol at follow-up was significantly lower for mothers who had GDM compared with those without, whereas LDL cholesterol and triglycerides at follow-up were significantly higher for mothers with GDM, Dr. Metzger said.
Racial/ethnic disparities, postpartum care
Neither long-term study – the NHS II or the HAPO Follow-Up Study – has looked at racial and ethnic differences. The HAPO cohort is racially-ethnically diverse but the NHS II cohort is predominantly white women.
Research suggests that GDM is a heterogeneous condition with some unique phenotypes in subgroups that vary by race and ethnicity. And just as there appear to be racial-ethnic differences in the pathophysiology of GDM, there appear to be racial-ethnic differences in the progression to type 2 diabetes – a known risk factor for cardiovascular disease, said Monique Henderson, PhD, a research scientist at Kaiser Permanente Northern California (KPNC).
On the broadest level, while Asian Americans have the highest prevalence of GDM, African Americans have the highest rates of progressing to type 2 diabetes, Dr. Henderson said. Disparities “may [stem from] metabolic differences in terms of insulin resistance and secretion that are different between pregnancy and the postpartum period, and that might vary [across racial-ethnic subgroups],” she said. Lifestyle differences and variation in postpartum screening rates also may play a role.
At KPNC, where women with GDM receive calls and letters reminding them of the need for postpartum screening, only 48% overall completed an oral glucose tolerance test at 4-12 weeks post partum, as recommended by both the American Diabetes Association and the American College of Obstetricians and Gynecologists. Both before and after adjustment for education, attendance at a postpartum visit, and other variables, Chinese women were most likely to have screening, and black women were least likely, said Dr. Henderson, referring to ongoing research.
A study Dr. Ehrenthal led of women with GDM or HDP recruited from the postpartum service of a large community-based, academic obstetrical hospital in Delaware showed that while nearly all women attended a 6-week postpartum visit with their ob.gyns., 59% of women with GDM had not yet completed diabetes screening when they were interviewed 3 months post partum. Most women with HDP indicated they had follow-up blood pressure testing, and just over half of women with either diagnosis recalled having ever had lipid testing (J Women’s Health 2014;23[9]:760-4).
Women least likely to complete screening tests were those who had no college education, those who had less than a high school level of health literacy, and those who were not privately insured, Dr. Ehrenthal said.
A large national study of privately insured women also found low rates of follow-up testing, however. While the majority of women with GDM had a postpartum visit with an obstetrician or primary care physician within a year after delivery, only a minority of women had a glycemic screening test completed (Obstet Gynecol. 2016;128[1]:159-67).
“We can’t place the blame on women,” Dr. Ehrenthal said. “We need increased attention to screening,” including screening for cardiovascular disease risk factors, and a “deliberate hand-off to primary care.”
For follow-up cardiovascular disease risk factor assessment after HDP, ACOG recommends periodic (perhaps annually) assessment and referral for treatment as needed, and the cardiology professional organizations recommend that pregnancy history be considered when assessing risk in order to decide on lipid treatment, she noted.
Each of the speakers reported that they have no financial or other interests that pose a conflict of interest. The HAPO Follow-Up Study is funded by the National Institute of Diabetes and Digestive and Kidney Diseases, and the nuMoM2b–HHS study has been funded by several National Institutes of Health institutes and other programs and initiatives.
WASHINGTON – Cardiovascular risk factors may be elevated “as soon as the first postpartum year” in women who have gestational diabetes or hypertensive disorders of pregnancy, recent findings have affirmed, Deborah B. Ehrenthal, MD, MPH, said at the biennial meeting of the Diabetes in Pregnancy Study Group of North America.
Dr. Ehrenthal was one of several researchers who urged innovative strategies and improved care coordination to boost women’s follow-up after gestational diabetes mellitus (GDM) and other adverse pregnancy outcomes and complications. “The metabolic stress of pregnancy can uncover underlying susceptibilities,” she said. “And ”
Evidence that adverse pregnancy outcomes – including GDM and hypertensive disorders of pregnancy (HDP) – can elevate cardiovascular risk comes most recently from the Nulliparous Pregnancy Outcomes Study – Monitoring Mothers to be Heart Health Study (nuMoM2b–HHS study), a prospective observational cohort that followed 4,484 women 2-7 years after their first pregnancy. Women had a follow-up exam, with blood pressure and anthropometric measurements and clinical/biological testing, an average of 3 years post partum.
An analysis published in October 2019 in the Journal of the American Heart Association shows that women with HDP (including preeclampsia and gestational hypertension) had a relative risk of hypertension of 2.5 at follow-up, compared with women without HDP. Women who had preeclampsia specifically were 2.3 times as likely as were women who did not have preeclampsia to have incident hypertension at follow-up, said Dr. Ehrenthal, a coinvestigator of the study.
The analysis focused on incident hypertension as the primary outcome, and adjusted for age, body mass index, and other important cardiovascular disease risk factors, she noted. Researchers utilized the diagnostic threshold for hypertension extant at the time of study design: A systolic blood pressure of 140 mm Hg or greater, or a diastolic BP of 90 mm Hg or greater (J Am Heart Assoc. 2019;8:e013092).
HDP was the most common adverse pregnancy outcome in the nuMoM2b–HHS study (14%). Among all participants, 4% had GDM. Approximately 82% had neither HDP nor GDM. Other adverse pregnancy outcomes included in the analysis were preterm birth, small-for-gestational-age birth, and stillbirth.
Additional preliminary estimates presented by Dr. Ehrenthal show that, based on the new (2017) lower threshold for hypertension – 130 mg Hg systolic or 80 mm Hg diastolic – the disorder afflicted 37% of women who had experienced HDP (relative risk 2.1), and 32% of women who had GDM (RR 1.8). Prediabetes/diabetes (using a fasting blood glucose threshold of 100 mg/dL) at follow-up affected an estimated 21% of women who had HDP (RR 1.4) and 38% of women who had GDM (RR 2.5).
Notably, across the entire study cohort, 20% had hypertension at follow-up, “which is extraordinary” considering the short time frame from pregnancy and the young age of the study population – a mean maternal age of 27 years, said Dr. Ehrenthal, associate professor of population health sciences and obstetrics & gynecology at the University of Wisconsin, Madison.
Also across the cohort, 15% had prediabetes/diabetes at follow-up. “We need to think about women more generally,” she cautioned. “While we recognize the significant elevated risk of HDP and GDM [for the development of subsequent hypertension and cardiovascular risk], we will miss a lot of women [if we focus only on the history of HDP and GDM.]”
The majority of women found to have hypertension or prediabetes/diabetes at follow-up had experienced neither HDP nor GDM, but a good many of them (47% of those who had hypertension and 47% of those found to have prediabetes/diabetes) had a BMI of 30 or above, Dr. Ehrenthal said at the DPSG-NA meeting.
Nurses Health Study, hyperglycemia and adverse pregnancy outcome follow-up data
The new findings from the nuMoM2b–HHS study add to a robust and growing body of evidence that pregnancy is an important window to future health, and that follow up and screening after GDM and HDP are crucial.
Regarding GDM specifically, “there’s quite a bit of literature by now demonstrating that GDM history is a risk factor for hypertension, even 1-2 years post partum, and that the risk is elevated as well for dyslipidemia and vascular dysfunction,” Deirdre K. Tobias, D.Sc., an epidemiologist at Brigham and Women’s Hospital and assistant professor of nutrition at Harvard TH Chan School of Public Health, Boston, said at the DPSG meeting.
An analysis of the Nurses Health Study II (NHS II) cohort published in 2017 found a 40% higher relative risk of cardiovascular disease events (largely myocardial infarction) in women who had GDM, compared with women who did not have GDM over a median follow-up of 26 years. This was after adjustments were made for age, time since pregnancy, menopausal status, family history of MI or stroke, hypertension in pregnancy, white race/ethnicity, prepregnancy BMI, and other factors (JAMA Intern Med. 2017;177[12]:1735-42).
The NHS data also have shown, however, that the elevated risk for cardiovascular disease after a GDM pregnancy “can be mitigated by adopting a healthy lifestyle,” said Dr. Tobias, lead author of the 2017 NHS II analysis. Adjustments for postpregnancy weight gain and lifestyle factors attenuated the relative risk of cardiovascular disease events after a GDM pregnancy to a 30% increased risk.
Dr. Tobias and colleagues currently are looking within the NHS cohort for “metabolomic signatures” or signals – various amino acid and lipid metabolites – to identify the progression of GDM to type 2 diabetes. Metabolomics “may help further refine our understanding of the long-term links between GDM and prevention of type 2 diabetes and of cardiovascular disease in mothers,” she said.
The Hyperglycemia and Adverse Pregnancy Outcome (HAPO) Follow-Up Study, in the meantime, is documenting associations of maternal glucose levels during pregnancy not only with prediabetes or type 2 diabetes 10-14 years later, but also with measures of cardiovascular risk in mothers 10-14 years later.
Just as perinatal outcomes were strongly associated with glucose as a continuous variable in the original HAPO study, “it’s clear there’s a progressive increase in the risk of [later] disorders of glucose metabolism as [fasting blood glucose levels and 1- and-2-hour glucose values] in pregnancy are higher,” said Boyd E. Metzger, MD, the Tom D. Spies emeritus professor of metabolism and nutrition at Northwestern University, Chicago, and principal investigator of the original HAPO study and its follow up.
“Another message is that the more normal you are in pregnancy, the more normal you will be many years later. Good values [during pregnancy] produce good outcomes.”
Currently unpublished data from the HAPO Follow-Up Study are being analyzed, but it appears thus far that GDM is not associated with hypertension (per the old diagnostic threshold) in this cohort after adjustment for maternal age, BMI, smoking, and family history of hypertension. GDM appears to be a significant risk factor for dyslipidemia, however. HDL cholesterol at follow-up was significantly lower for mothers who had GDM compared with those without, whereas LDL cholesterol and triglycerides at follow-up were significantly higher for mothers with GDM, Dr. Metzger said.
Racial/ethnic disparities, postpartum care
Neither long-term study – the NHS II or the HAPO Follow-Up Study – has looked at racial and ethnic differences. The HAPO cohort is racially-ethnically diverse but the NHS II cohort is predominantly white women.
Research suggests that GDM is a heterogeneous condition with some unique phenotypes in subgroups that vary by race and ethnicity. And just as there appear to be racial-ethnic differences in the pathophysiology of GDM, there appear to be racial-ethnic differences in the progression to type 2 diabetes – a known risk factor for cardiovascular disease, said Monique Henderson, PhD, a research scientist at Kaiser Permanente Northern California (KPNC).
On the broadest level, while Asian Americans have the highest prevalence of GDM, African Americans have the highest rates of progressing to type 2 diabetes, Dr. Henderson said. Disparities “may [stem from] metabolic differences in terms of insulin resistance and secretion that are different between pregnancy and the postpartum period, and that might vary [across racial-ethnic subgroups],” she said. Lifestyle differences and variation in postpartum screening rates also may play a role.
At KPNC, where women with GDM receive calls and letters reminding them of the need for postpartum screening, only 48% overall completed an oral glucose tolerance test at 4-12 weeks post partum, as recommended by both the American Diabetes Association and the American College of Obstetricians and Gynecologists. Both before and after adjustment for education, attendance at a postpartum visit, and other variables, Chinese women were most likely to have screening, and black women were least likely, said Dr. Henderson, referring to ongoing research.
A study Dr. Ehrenthal led of women with GDM or HDP recruited from the postpartum service of a large community-based, academic obstetrical hospital in Delaware showed that while nearly all women attended a 6-week postpartum visit with their ob.gyns., 59% of women with GDM had not yet completed diabetes screening when they were interviewed 3 months post partum. Most women with HDP indicated they had follow-up blood pressure testing, and just over half of women with either diagnosis recalled having ever had lipid testing (J Women’s Health 2014;23[9]:760-4).
Women least likely to complete screening tests were those who had no college education, those who had less than a high school level of health literacy, and those who were not privately insured, Dr. Ehrenthal said.
A large national study of privately insured women also found low rates of follow-up testing, however. While the majority of women with GDM had a postpartum visit with an obstetrician or primary care physician within a year after delivery, only a minority of women had a glycemic screening test completed (Obstet Gynecol. 2016;128[1]:159-67).
“We can’t place the blame on women,” Dr. Ehrenthal said. “We need increased attention to screening,” including screening for cardiovascular disease risk factors, and a “deliberate hand-off to primary care.”
For follow-up cardiovascular disease risk factor assessment after HDP, ACOG recommends periodic (perhaps annually) assessment and referral for treatment as needed, and the cardiology professional organizations recommend that pregnancy history be considered when assessing risk in order to decide on lipid treatment, she noted.
Each of the speakers reported that they have no financial or other interests that pose a conflict of interest. The HAPO Follow-Up Study is funded by the National Institute of Diabetes and Digestive and Kidney Diseases, and the nuMoM2b–HHS study has been funded by several National Institutes of Health institutes and other programs and initiatives.
REPORTING FROM THE DPSG-NA 2019
Depression linked to persistent opioid use after hysterectomy
VANCOUVER –
Women with depression had an 8% increased risk of perioperative opioid use but a 43% increased risk of persistent use, defined as at least one perioperative prescription followed by at least one prescription 90 days or longer after surgery.
Opioid prescriptions after surgery have been on the rise in recent years, and this has led to a focus on how chronic pain disorders are managed. But studies have shown that patients undergoing general surgery, both minor and major, are at increased risk of persistent opioid use, even after a single surgery, according to Erin Carey, MD, director of the division of minimally invasive gynecologic surgery at the University of North Carolina at Chapel Hill, who presented the research at the meeting sponsored by AAGL.
“We also know that preoperative depression has been linked to adverse outcomes after hysterectomy, both acute postoperative pain in the first 2 days after surgery, and increasing the risk of chronic postoperative pain,” Dr. Carey said.
That prompted her and her team to look at whether preoperative depression might influence the risk of new persistent opioid use after hysterectomy. They analyzed data from the IBM Watson/Truven Health Analytics MarketScan database of claims-based data, which collects information from a variety of sources, including electronic medical records and workplace records such as absences, disability, and long-term disability.
“So it does allow for long-term tracking, which makes it optimal for this type of study,” said Dr. Carey.
The study included 382,078 hysterectomies performed between 2001 and 2015 on women who had continuous prescription plans 180 days before to 180 days after the procedure, excluding anyone who had an opioid prescription in the previous 180 days; 60% of the procedures were minimally invasive. About 20% of women were considered to have depression before the procedure, based on a diagnosis (55%), an antidepressant prescription (22%), or both (23%).
There were some differences at baseline between the two populations: Women with preoperative depression were more likely to have a comorbid pain disorder, compared with patients without depression (20% vs. 14%), another psychiatric disorder (2% vs. less than 1%), and a Charlson comorbidity (12% vs. 9%). They also were less likely to undergo a minimally invasive procedure than women without depression (66% vs. 79%). There was an increase in the prevalence of depression over time, from 16% to 23%.
Overall, 74% of women were prescribed an opioid during the perioperative period; 17% were filled before the hysterectomy was performed. Preoperative fills also increased over time, from 4% in 2001 to 21% in 2015.
Women with preoperative depression were at a slightly greater risk for perioperative opioid use (risk ratio, 1.08), but a greater risk for persistent postoperative opioid use (11% vs. 8%; RR, 1.43). The heightened risk for opioid use was similar whether the surgery was performed on an outpatient or inpatient basis.
The presence of other comorbidities in women with diagnosed depression or prescribed antidepressants complicates the findings, according to Dr. Carey. “There may be additional chronic pain factors that are confounding this data, but it is consistent with other data that de novo postoperative opioid dependence may be a higher risk for these patients, so it’s important for us to look at that critically.”
Dr. Carey has been a consultant for Teleflex Medical and a speaker for Med-IQ.
VANCOUVER –
Women with depression had an 8% increased risk of perioperative opioid use but a 43% increased risk of persistent use, defined as at least one perioperative prescription followed by at least one prescription 90 days or longer after surgery.
Opioid prescriptions after surgery have been on the rise in recent years, and this has led to a focus on how chronic pain disorders are managed. But studies have shown that patients undergoing general surgery, both minor and major, are at increased risk of persistent opioid use, even after a single surgery, according to Erin Carey, MD, director of the division of minimally invasive gynecologic surgery at the University of North Carolina at Chapel Hill, who presented the research at the meeting sponsored by AAGL.
“We also know that preoperative depression has been linked to adverse outcomes after hysterectomy, both acute postoperative pain in the first 2 days after surgery, and increasing the risk of chronic postoperative pain,” Dr. Carey said.
That prompted her and her team to look at whether preoperative depression might influence the risk of new persistent opioid use after hysterectomy. They analyzed data from the IBM Watson/Truven Health Analytics MarketScan database of claims-based data, which collects information from a variety of sources, including electronic medical records and workplace records such as absences, disability, and long-term disability.
“So it does allow for long-term tracking, which makes it optimal for this type of study,” said Dr. Carey.
The study included 382,078 hysterectomies performed between 2001 and 2015 on women who had continuous prescription plans 180 days before to 180 days after the procedure, excluding anyone who had an opioid prescription in the previous 180 days; 60% of the procedures were minimally invasive. About 20% of women were considered to have depression before the procedure, based on a diagnosis (55%), an antidepressant prescription (22%), or both (23%).
There were some differences at baseline between the two populations: Women with preoperative depression were more likely to have a comorbid pain disorder, compared with patients without depression (20% vs. 14%), another psychiatric disorder (2% vs. less than 1%), and a Charlson comorbidity (12% vs. 9%). They also were less likely to undergo a minimally invasive procedure than women without depression (66% vs. 79%). There was an increase in the prevalence of depression over time, from 16% to 23%.
Overall, 74% of women were prescribed an opioid during the perioperative period; 17% were filled before the hysterectomy was performed. Preoperative fills also increased over time, from 4% in 2001 to 21% in 2015.
Women with preoperative depression were at a slightly greater risk for perioperative opioid use (risk ratio, 1.08), but a greater risk for persistent postoperative opioid use (11% vs. 8%; RR, 1.43). The heightened risk for opioid use was similar whether the surgery was performed on an outpatient or inpatient basis.
The presence of other comorbidities in women with diagnosed depression or prescribed antidepressants complicates the findings, according to Dr. Carey. “There may be additional chronic pain factors that are confounding this data, but it is consistent with other data that de novo postoperative opioid dependence may be a higher risk for these patients, so it’s important for us to look at that critically.”
Dr. Carey has been a consultant for Teleflex Medical and a speaker for Med-IQ.
VANCOUVER –
Women with depression had an 8% increased risk of perioperative opioid use but a 43% increased risk of persistent use, defined as at least one perioperative prescription followed by at least one prescription 90 days or longer after surgery.
Opioid prescriptions after surgery have been on the rise in recent years, and this has led to a focus on how chronic pain disorders are managed. But studies have shown that patients undergoing general surgery, both minor and major, are at increased risk of persistent opioid use, even after a single surgery, according to Erin Carey, MD, director of the division of minimally invasive gynecologic surgery at the University of North Carolina at Chapel Hill, who presented the research at the meeting sponsored by AAGL.
“We also know that preoperative depression has been linked to adverse outcomes after hysterectomy, both acute postoperative pain in the first 2 days after surgery, and increasing the risk of chronic postoperative pain,” Dr. Carey said.
That prompted her and her team to look at whether preoperative depression might influence the risk of new persistent opioid use after hysterectomy. They analyzed data from the IBM Watson/Truven Health Analytics MarketScan database of claims-based data, which collects information from a variety of sources, including electronic medical records and workplace records such as absences, disability, and long-term disability.
“So it does allow for long-term tracking, which makes it optimal for this type of study,” said Dr. Carey.
The study included 382,078 hysterectomies performed between 2001 and 2015 on women who had continuous prescription plans 180 days before to 180 days after the procedure, excluding anyone who had an opioid prescription in the previous 180 days; 60% of the procedures were minimally invasive. About 20% of women were considered to have depression before the procedure, based on a diagnosis (55%), an antidepressant prescription (22%), or both (23%).
There were some differences at baseline between the two populations: Women with preoperative depression were more likely to have a comorbid pain disorder, compared with patients without depression (20% vs. 14%), another psychiatric disorder (2% vs. less than 1%), and a Charlson comorbidity (12% vs. 9%). They also were less likely to undergo a minimally invasive procedure than women without depression (66% vs. 79%). There was an increase in the prevalence of depression over time, from 16% to 23%.
Overall, 74% of women were prescribed an opioid during the perioperative period; 17% were filled before the hysterectomy was performed. Preoperative fills also increased over time, from 4% in 2001 to 21% in 2015.
Women with preoperative depression were at a slightly greater risk for perioperative opioid use (risk ratio, 1.08), but a greater risk for persistent postoperative opioid use (11% vs. 8%; RR, 1.43). The heightened risk for opioid use was similar whether the surgery was performed on an outpatient or inpatient basis.
The presence of other comorbidities in women with diagnosed depression or prescribed antidepressants complicates the findings, according to Dr. Carey. “There may be additional chronic pain factors that are confounding this data, but it is consistent with other data that de novo postoperative opioid dependence may be a higher risk for these patients, so it’s important for us to look at that critically.”
Dr. Carey has been a consultant for Teleflex Medical and a speaker for Med-IQ.
REPORTING FROM THE AAGL GLOBAL CONGRESS
Pulmonary embolism treatment teams adopted widely for complex disease
NEW YORK – Seven years after the formation of the first pulmonary embolism response team (PERT), more than 100 institutions have joined the PERT Consortium, which was created to guide care and research for this thrombotic complication, according to a status report at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation.
“Why are PERTs needed? Pulmonary embolism patients are like snowflakes. No two are the same,” explained Richard Channick, MD, director of the pulmonary vascular disease program, University of California, Los Angeles.
Patient variability is an issue because algorithms for pulmonary embolism (PE) often differ at the point of diagnosis, such as the emergency department or intensive are unit, according to Dr. Channick, who was present when the first PERT was created in 2012 at Massachusetts General Hospital (MGH) in Boston. In addition, treatment algorithms can seem complex at a time when patients are deteriorating quickly.
“The treatment algorithms always say consider this or consider that, and then you get a recommendation with a 2B grade of evidence. So what do you do?” Dr. Channick asked, “This has really been crying for an organized approach.”
PERTs were created to fill this need. In most centers, PERTs are organized to respond to a diagnosis of PE wherever it occurs in the hospital. The goal is rapid activation of a team of experts who can reach a single consensus recommendation.
At MGH and UCLA, a similar relatively simple scheme has been created to guide physicians on how to activate the PERT and which situations make this appropriate.
“A big part of the PERT value has been our ability to conduct a real-time virtual consultation where we leverage online technology to look at images together in order to agree on a strategy,” Dr. Channick explained.
Although frequently asked what specialists are needed for an effective PERT, Dr. Channick said it depends on institutional structures, the types of specialists available, and, in some cases, the specific characteristics of the patient. In many situations, a pulmonary vascular specialist and an interventional radiologist might be sufficient. In others, team members might include some combination of an interventional cardiologist, a cardiac surgeon, and a hematologist.
It is also appropriate to include clinicians likely to participate in care following acute treatment of the PE. “One of the most critical values to PERT is the ability to systematically follow patients” after the PE is treated, Dr. Channick said.
So far, there are no data to confirm patients managed with PERT achieve better outcomes than those who are not. Reductions in mortality, length of stay, and costs are reasonably anticipated and might eventually be demonstrated, but Dr. Channick said that PERTs already have value.
“I think the efficiency of care is important,”he said. He called PERT a “one-stop shopping” approach to ensuring that multiple strategies are considered systematically.
There are many anecdotal examples of the benefits of shared decision-making for PE treatment. In one, a pulmonary specialist in a PERT team narrowly averted a planned thrombolysis in a patient diagnosed with PE who was actually found to have severe pulmonary fibrosis, according to Dr. Channick.
Not least important, the shared decision-making of a PERT could relieve the burden of difficult choices in complex situations. Bad outcomes in PE can be unavoidable even with optimal therapy.
“To me personally, a very important benefit of being part of a PERT is the feeling that we are all in it together,” Dr. Channick said. “Patients can go from being pretty stable to being dead very quickly.”
The PERT Consortium has sponsored an annual meeting on PE since 2015. It also maintains an ongoing registry for PE data from member institutions. These data are expected to have increasing value for comparing the impact of patient characteristics, treatment strategies, and other variables on outcomes.
For clinicians who are uncertain whether the PE incidence at their institution justifies a PERT, Dr. Channick had some advice. “If you build it, they will clot,” he said, meaning that due to the frequency of PE, a PERT will generally have plenty of work once created.
SOURCE: VEITHSYMPOSIUM
NEW YORK – Seven years after the formation of the first pulmonary embolism response team (PERT), more than 100 institutions have joined the PERT Consortium, which was created to guide care and research for this thrombotic complication, according to a status report at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation.
“Why are PERTs needed? Pulmonary embolism patients are like snowflakes. No two are the same,” explained Richard Channick, MD, director of the pulmonary vascular disease program, University of California, Los Angeles.
Patient variability is an issue because algorithms for pulmonary embolism (PE) often differ at the point of diagnosis, such as the emergency department or intensive are unit, according to Dr. Channick, who was present when the first PERT was created in 2012 at Massachusetts General Hospital (MGH) in Boston. In addition, treatment algorithms can seem complex at a time when patients are deteriorating quickly.
“The treatment algorithms always say consider this or consider that, and then you get a recommendation with a 2B grade of evidence. So what do you do?” Dr. Channick asked, “This has really been crying for an organized approach.”
PERTs were created to fill this need. In most centers, PERTs are organized to respond to a diagnosis of PE wherever it occurs in the hospital. The goal is rapid activation of a team of experts who can reach a single consensus recommendation.
At MGH and UCLA, a similar relatively simple scheme has been created to guide physicians on how to activate the PERT and which situations make this appropriate.
“A big part of the PERT value has been our ability to conduct a real-time virtual consultation where we leverage online technology to look at images together in order to agree on a strategy,” Dr. Channick explained.
Although frequently asked what specialists are needed for an effective PERT, Dr. Channick said it depends on institutional structures, the types of specialists available, and, in some cases, the specific characteristics of the patient. In many situations, a pulmonary vascular specialist and an interventional radiologist might be sufficient. In others, team members might include some combination of an interventional cardiologist, a cardiac surgeon, and a hematologist.
It is also appropriate to include clinicians likely to participate in care following acute treatment of the PE. “One of the most critical values to PERT is the ability to systematically follow patients” after the PE is treated, Dr. Channick said.
So far, there are no data to confirm patients managed with PERT achieve better outcomes than those who are not. Reductions in mortality, length of stay, and costs are reasonably anticipated and might eventually be demonstrated, but Dr. Channick said that PERTs already have value.
“I think the efficiency of care is important,”he said. He called PERT a “one-stop shopping” approach to ensuring that multiple strategies are considered systematically.
There are many anecdotal examples of the benefits of shared decision-making for PE treatment. In one, a pulmonary specialist in a PERT team narrowly averted a planned thrombolysis in a patient diagnosed with PE who was actually found to have severe pulmonary fibrosis, according to Dr. Channick.
Not least important, the shared decision-making of a PERT could relieve the burden of difficult choices in complex situations. Bad outcomes in PE can be unavoidable even with optimal therapy.
“To me personally, a very important benefit of being part of a PERT is the feeling that we are all in it together,” Dr. Channick said. “Patients can go from being pretty stable to being dead very quickly.”
The PERT Consortium has sponsored an annual meeting on PE since 2015. It also maintains an ongoing registry for PE data from member institutions. These data are expected to have increasing value for comparing the impact of patient characteristics, treatment strategies, and other variables on outcomes.
For clinicians who are uncertain whether the PE incidence at their institution justifies a PERT, Dr. Channick had some advice. “If you build it, they will clot,” he said, meaning that due to the frequency of PE, a PERT will generally have plenty of work once created.
SOURCE: VEITHSYMPOSIUM
NEW YORK – Seven years after the formation of the first pulmonary embolism response team (PERT), more than 100 institutions have joined the PERT Consortium, which was created to guide care and research for this thrombotic complication, according to a status report at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation.
“Why are PERTs needed? Pulmonary embolism patients are like snowflakes. No two are the same,” explained Richard Channick, MD, director of the pulmonary vascular disease program, University of California, Los Angeles.
Patient variability is an issue because algorithms for pulmonary embolism (PE) often differ at the point of diagnosis, such as the emergency department or intensive are unit, according to Dr. Channick, who was present when the first PERT was created in 2012 at Massachusetts General Hospital (MGH) in Boston. In addition, treatment algorithms can seem complex at a time when patients are deteriorating quickly.
“The treatment algorithms always say consider this or consider that, and then you get a recommendation with a 2B grade of evidence. So what do you do?” Dr. Channick asked, “This has really been crying for an organized approach.”
PERTs were created to fill this need. In most centers, PERTs are organized to respond to a diagnosis of PE wherever it occurs in the hospital. The goal is rapid activation of a team of experts who can reach a single consensus recommendation.
At MGH and UCLA, a similar relatively simple scheme has been created to guide physicians on how to activate the PERT and which situations make this appropriate.
“A big part of the PERT value has been our ability to conduct a real-time virtual consultation where we leverage online technology to look at images together in order to agree on a strategy,” Dr. Channick explained.
Although frequently asked what specialists are needed for an effective PERT, Dr. Channick said it depends on institutional structures, the types of specialists available, and, in some cases, the specific characteristics of the patient. In many situations, a pulmonary vascular specialist and an interventional radiologist might be sufficient. In others, team members might include some combination of an interventional cardiologist, a cardiac surgeon, and a hematologist.
It is also appropriate to include clinicians likely to participate in care following acute treatment of the PE. “One of the most critical values to PERT is the ability to systematically follow patients” after the PE is treated, Dr. Channick said.
So far, there are no data to confirm patients managed with PERT achieve better outcomes than those who are not. Reductions in mortality, length of stay, and costs are reasonably anticipated and might eventually be demonstrated, but Dr. Channick said that PERTs already have value.
“I think the efficiency of care is important,”he said. He called PERT a “one-stop shopping” approach to ensuring that multiple strategies are considered systematically.
There are many anecdotal examples of the benefits of shared decision-making for PE treatment. In one, a pulmonary specialist in a PERT team narrowly averted a planned thrombolysis in a patient diagnosed with PE who was actually found to have severe pulmonary fibrosis, according to Dr. Channick.
Not least important, the shared decision-making of a PERT could relieve the burden of difficult choices in complex situations. Bad outcomes in PE can be unavoidable even with optimal therapy.
“To me personally, a very important benefit of being part of a PERT is the feeling that we are all in it together,” Dr. Channick said. “Patients can go from being pretty stable to being dead very quickly.”
The PERT Consortium has sponsored an annual meeting on PE since 2015. It also maintains an ongoing registry for PE data from member institutions. These data are expected to have increasing value for comparing the impact of patient characteristics, treatment strategies, and other variables on outcomes.
For clinicians who are uncertain whether the PE incidence at their institution justifies a PERT, Dr. Channick had some advice. “If you build it, they will clot,” he said, meaning that due to the frequency of PE, a PERT will generally have plenty of work once created.
SOURCE: VEITHSYMPOSIUM
REPORTING FROM THE VEITHSYMPOSIUM
Not all lung cancer patients receive treatment
In the United States, just over 15% of patients with lung cancer receive no treatment, according to the American Lung Association.
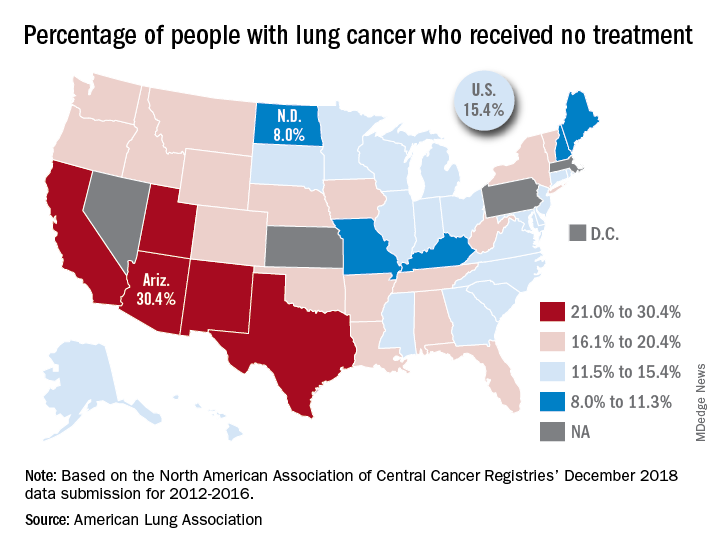
“This can happen for multiple reasons, such as the tumor having spread too far, poor health, or refusal of treatment,” the ALA said in its 2019 State of Lung Cancer report.
On the state level, the disparities were considerable. Arizona had the highest rate of nontreatment at 30.4%, followed by the neighboring states of New Mexico (24.2%) and California (24.0%). The lowest rate in the country, 8.0%, came from North Dakota, with Missouri next at 9.4% and Maine third at 9.6%, based on data from the North American Association of Central Cancer Registries’ December 2018 data submission, which covered the years from 2012 to 2016.
Although some cases of lung cancer may be unavoidable, “no one should go untreated because of lack of provider or patient knowledge, stigma associated with lung cancer, fatalism after diagnosis, or cost of treatment. Dismantling these and other barriers is important to reducing the percent of untreated patients,” the ALA said.
In the United States, just over 15% of patients with lung cancer receive no treatment, according to the American Lung Association.

“This can happen for multiple reasons, such as the tumor having spread too far, poor health, or refusal of treatment,” the ALA said in its 2019 State of Lung Cancer report.
On the state level, the disparities were considerable. Arizona had the highest rate of nontreatment at 30.4%, followed by the neighboring states of New Mexico (24.2%) and California (24.0%). The lowest rate in the country, 8.0%, came from North Dakota, with Missouri next at 9.4% and Maine third at 9.6%, based on data from the North American Association of Central Cancer Registries’ December 2018 data submission, which covered the years from 2012 to 2016.
Although some cases of lung cancer may be unavoidable, “no one should go untreated because of lack of provider or patient knowledge, stigma associated with lung cancer, fatalism after diagnosis, or cost of treatment. Dismantling these and other barriers is important to reducing the percent of untreated patients,” the ALA said.
In the United States, just over 15% of patients with lung cancer receive no treatment, according to the American Lung Association.

“This can happen for multiple reasons, such as the tumor having spread too far, poor health, or refusal of treatment,” the ALA said in its 2019 State of Lung Cancer report.
On the state level, the disparities were considerable. Arizona had the highest rate of nontreatment at 30.4%, followed by the neighboring states of New Mexico (24.2%) and California (24.0%). The lowest rate in the country, 8.0%, came from North Dakota, with Missouri next at 9.4% and Maine third at 9.6%, based on data from the North American Association of Central Cancer Registries’ December 2018 data submission, which covered the years from 2012 to 2016.
Although some cases of lung cancer may be unavoidable, “no one should go untreated because of lack of provider or patient knowledge, stigma associated with lung cancer, fatalism after diagnosis, or cost of treatment. Dismantling these and other barriers is important to reducing the percent of untreated patients,” the ALA said.
Hepatitis B debrief: Key themes that emerged at AASLD
BOSTON – Some of the most notable abstracts presented here at the annual meeting of the American Association for the Study of Liver Diseases dealt with key topics including the natural history of hepatitis B virus, novel treatment approaches, and prevention, according to Marc Ghany, MD.
During a special hepatitis debriefing session held on the last day of the conference, Dr. Ghany reviewed his key selections in hepatitis B virus (HBV) research, including the following:
Natural history
Steatohepatitis may worsen HBV-related liver injury, according to results of an analysis of liver biopsies from adult patients enrolled in a North American cohort study (Abstract 162). Investigators found that steatohepatitis was associated with a 1.6-fold increased risk of advanced fibrosis.
“For all patients with hepatitis B, I think it’s important to screen and manage metabolic abnormalities to prevent liver disease progression,” said Dr. Ghany, who is with the Liver Diseases Branch of the National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, Bethesda, Md.
Risk of hepatocellular carcinoma (HCC) in patients with chronic hepatitis B was evaluated in two notable studies, Dr. Ghany said, including one that found no difference in risk of HCC development among white patients who received long-term entecavir versus those who received long-term tenofovir (Abstract 454). This stands in contrast to a previous and controversial finding, according to investigators, that HCC incidence was lower in Asian patients treated with tenofovir versus those treated with entecavir.
In the other study, investigators found that dual-antiplatelet therapy (DAPT) was associated with a lower risk of HCC versus aspirin monotherapy in patients with chronic hepatitis B (Abstract 934). Antiplatelet therapy was associated with a near 20% reduction in HCC risk, while DAPT as compared with aspirin monotherapy was linked to a near 30% reduction. “These are very provocative findings,” Dr. Ghany said. “Of course they need to be confirmed, but if so, may open new avenues of HCC chemoprevention.”
Novel therapies
Several new and promising drugs are undergoing clinical trials, including JNJ-64530440 (JNJ-0440), a novel class N capsid assembly modulator. In phase 1a data presented here at The Liver Meeting, the treatment was safe, well tolerated, and resulted in potent inhibition of viral replication (Abstract 0089). “We await further studies on its effect on functional cure,” Dr. Ghany told attendees.
Another treatment to watch is GSK3389404 (GSK404), a liver-targeted antisense oligonucleotide; in a phase 2 placebo-controlled study in patients with chronic hepatitis B on stable nucleos(t)ide therapy, this treatment had acceptable safety and produced dose-dependent declines in hepatitis B surface antigen (HBsAg), according to investigators (Abstract 0695). Dr. Ghany said this constitutes “proof of principle” that antisense oligonucleotides can decrease HBsAg levels.
In a phase 2 randomized, placebo-controlled study in virally suppressed adults with chronic hepatitis B, the toll-like receptor 8 (TLR8) agonist GS-9688 was safe and well tolerated, and resulted in dose-dependent pharmacodynamic changes, with 5% of patients experiencing a 1 log10 IU/mL or greater decline in HBsAg levels or an HBsAg loss by week 24 (Abstract 0697). This is a “promising approach” that merits further study, according to Dr. Ghany.
Prevention: Vaccination and screening
A trivalent HBV vaccine is superior to monovalent vaccine, according to results from the double-blind, randomized, controlled, phase 3 PROTECT study presented here at the meeting (Abstract LP13). Known as Sci-B-Vac, the mammalian cell-derived trivalent vaccine had higher response rates versus the recombinant monovalent vaccine Engerix-B in difficult-to-vaccinate populations, according to Dr. Ghany.
A separate report based on a national health insurance cohort study in Korea demonstrated that regular follow-up, that is to say, every 3-6 months, significantly reduced liver cancer–related mortality (Abstract 0159). Patients compliant with screening in the study not only had a 44% reduction in risk of death from HCC, but also were more likely to receive curative treatments (23.1% versus 15.1%). “Notwithstanding the limitations or cohort studies, I think these data reinforce the need to screen patients with chronic hepatitis B,” Dr. Ghany said.
He provided no disclosures in his presentation.
BOSTON – Some of the most notable abstracts presented here at the annual meeting of the American Association for the Study of Liver Diseases dealt with key topics including the natural history of hepatitis B virus, novel treatment approaches, and prevention, according to Marc Ghany, MD.
During a special hepatitis debriefing session held on the last day of the conference, Dr. Ghany reviewed his key selections in hepatitis B virus (HBV) research, including the following:
Natural history
Steatohepatitis may worsen HBV-related liver injury, according to results of an analysis of liver biopsies from adult patients enrolled in a North American cohort study (Abstract 162). Investigators found that steatohepatitis was associated with a 1.6-fold increased risk of advanced fibrosis.
“For all patients with hepatitis B, I think it’s important to screen and manage metabolic abnormalities to prevent liver disease progression,” said Dr. Ghany, who is with the Liver Diseases Branch of the National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, Bethesda, Md.
Risk of hepatocellular carcinoma (HCC) in patients with chronic hepatitis B was evaluated in two notable studies, Dr. Ghany said, including one that found no difference in risk of HCC development among white patients who received long-term entecavir versus those who received long-term tenofovir (Abstract 454). This stands in contrast to a previous and controversial finding, according to investigators, that HCC incidence was lower in Asian patients treated with tenofovir versus those treated with entecavir.
In the other study, investigators found that dual-antiplatelet therapy (DAPT) was associated with a lower risk of HCC versus aspirin monotherapy in patients with chronic hepatitis B (Abstract 934). Antiplatelet therapy was associated with a near 20% reduction in HCC risk, while DAPT as compared with aspirin monotherapy was linked to a near 30% reduction. “These are very provocative findings,” Dr. Ghany said. “Of course they need to be confirmed, but if so, may open new avenues of HCC chemoprevention.”
Novel therapies
Several new and promising drugs are undergoing clinical trials, including JNJ-64530440 (JNJ-0440), a novel class N capsid assembly modulator. In phase 1a data presented here at The Liver Meeting, the treatment was safe, well tolerated, and resulted in potent inhibition of viral replication (Abstract 0089). “We await further studies on its effect on functional cure,” Dr. Ghany told attendees.
Another treatment to watch is GSK3389404 (GSK404), a liver-targeted antisense oligonucleotide; in a phase 2 placebo-controlled study in patients with chronic hepatitis B on stable nucleos(t)ide therapy, this treatment had acceptable safety and produced dose-dependent declines in hepatitis B surface antigen (HBsAg), according to investigators (Abstract 0695). Dr. Ghany said this constitutes “proof of principle” that antisense oligonucleotides can decrease HBsAg levels.
In a phase 2 randomized, placebo-controlled study in virally suppressed adults with chronic hepatitis B, the toll-like receptor 8 (TLR8) agonist GS-9688 was safe and well tolerated, and resulted in dose-dependent pharmacodynamic changes, with 5% of patients experiencing a 1 log10 IU/mL or greater decline in HBsAg levels or an HBsAg loss by week 24 (Abstract 0697). This is a “promising approach” that merits further study, according to Dr. Ghany.
Prevention: Vaccination and screening
A trivalent HBV vaccine is superior to monovalent vaccine, according to results from the double-blind, randomized, controlled, phase 3 PROTECT study presented here at the meeting (Abstract LP13). Known as Sci-B-Vac, the mammalian cell-derived trivalent vaccine had higher response rates versus the recombinant monovalent vaccine Engerix-B in difficult-to-vaccinate populations, according to Dr. Ghany.
A separate report based on a national health insurance cohort study in Korea demonstrated that regular follow-up, that is to say, every 3-6 months, significantly reduced liver cancer–related mortality (Abstract 0159). Patients compliant with screening in the study not only had a 44% reduction in risk of death from HCC, but also were more likely to receive curative treatments (23.1% versus 15.1%). “Notwithstanding the limitations or cohort studies, I think these data reinforce the need to screen patients with chronic hepatitis B,” Dr. Ghany said.
He provided no disclosures in his presentation.
BOSTON – Some of the most notable abstracts presented here at the annual meeting of the American Association for the Study of Liver Diseases dealt with key topics including the natural history of hepatitis B virus, novel treatment approaches, and prevention, according to Marc Ghany, MD.
During a special hepatitis debriefing session held on the last day of the conference, Dr. Ghany reviewed his key selections in hepatitis B virus (HBV) research, including the following:
Natural history
Steatohepatitis may worsen HBV-related liver injury, according to results of an analysis of liver biopsies from adult patients enrolled in a North American cohort study (Abstract 162). Investigators found that steatohepatitis was associated with a 1.6-fold increased risk of advanced fibrosis.
“For all patients with hepatitis B, I think it’s important to screen and manage metabolic abnormalities to prevent liver disease progression,” said Dr. Ghany, who is with the Liver Diseases Branch of the National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, Bethesda, Md.
Risk of hepatocellular carcinoma (HCC) in patients with chronic hepatitis B was evaluated in two notable studies, Dr. Ghany said, including one that found no difference in risk of HCC development among white patients who received long-term entecavir versus those who received long-term tenofovir (Abstract 454). This stands in contrast to a previous and controversial finding, according to investigators, that HCC incidence was lower in Asian patients treated with tenofovir versus those treated with entecavir.
In the other study, investigators found that dual-antiplatelet therapy (DAPT) was associated with a lower risk of HCC versus aspirin monotherapy in patients with chronic hepatitis B (Abstract 934). Antiplatelet therapy was associated with a near 20% reduction in HCC risk, while DAPT as compared with aspirin monotherapy was linked to a near 30% reduction. “These are very provocative findings,” Dr. Ghany said. “Of course they need to be confirmed, but if so, may open new avenues of HCC chemoprevention.”
Novel therapies
Several new and promising drugs are undergoing clinical trials, including JNJ-64530440 (JNJ-0440), a novel class N capsid assembly modulator. In phase 1a data presented here at The Liver Meeting, the treatment was safe, well tolerated, and resulted in potent inhibition of viral replication (Abstract 0089). “We await further studies on its effect on functional cure,” Dr. Ghany told attendees.
Another treatment to watch is GSK3389404 (GSK404), a liver-targeted antisense oligonucleotide; in a phase 2 placebo-controlled study in patients with chronic hepatitis B on stable nucleos(t)ide therapy, this treatment had acceptable safety and produced dose-dependent declines in hepatitis B surface antigen (HBsAg), according to investigators (Abstract 0695). Dr. Ghany said this constitutes “proof of principle” that antisense oligonucleotides can decrease HBsAg levels.
In a phase 2 randomized, placebo-controlled study in virally suppressed adults with chronic hepatitis B, the toll-like receptor 8 (TLR8) agonist GS-9688 was safe and well tolerated, and resulted in dose-dependent pharmacodynamic changes, with 5% of patients experiencing a 1 log10 IU/mL or greater decline in HBsAg levels or an HBsAg loss by week 24 (Abstract 0697). This is a “promising approach” that merits further study, according to Dr. Ghany.
Prevention: Vaccination and screening
A trivalent HBV vaccine is superior to monovalent vaccine, according to results from the double-blind, randomized, controlled, phase 3 PROTECT study presented here at the meeting (Abstract LP13). Known as Sci-B-Vac, the mammalian cell-derived trivalent vaccine had higher response rates versus the recombinant monovalent vaccine Engerix-B in difficult-to-vaccinate populations, according to Dr. Ghany.
A separate report based on a national health insurance cohort study in Korea demonstrated that regular follow-up, that is to say, every 3-6 months, significantly reduced liver cancer–related mortality (Abstract 0159). Patients compliant with screening in the study not only had a 44% reduction in risk of death from HCC, but also were more likely to receive curative treatments (23.1% versus 15.1%). “Notwithstanding the limitations or cohort studies, I think these data reinforce the need to screen patients with chronic hepatitis B,” Dr. Ghany said.
He provided no disclosures in his presentation.
REPORTING FROM THE LIVER MEETING 2019
Health policy Q&A: Oncology Care Model
The Oncology Care Model is a value-based payment approach aimed at encouraging coordinated cancer care through targeted bonus payments to practices. The payment experiment was launched by the Centers for Medicare & Medicaid Services in 2016 and now includes 175 practices and 10 payers. It is set to end in 2021. As agency officials consider whether to continue the program, Stephen S. Grubbs, MD, vice president for clinical affairs at the American Society of Clinical Oncology, weighs in on the model’s track record and its future.
Question: How would you rate the Oncology Care Model in helping to drive practice transformation?
Dr. Grubbs: Participants in the Oncology Care Model (OCM) have demonstrated improved care coordination, psychosocial support, use of risk assessment tools, and other strategies to lower costs and adverse events. Over the past 2 years, ASCO has accepted numerous posters, articles, and abstracts from OCM participants on their outstanding work to advance cancer care delivery.
Question: Should the model be extended beyond 2021?
Dr. Grubbs: Changes to the model are necessary prior to a significant extension or expansion. Some have suggested that CMS extend OCM for an additional year with current participants. This would give CMS time to consider input from all stakeholders on its eventual replacement.
Question: What additional resources or payments do oncology practices need to be more successful in meeting the goals of the Oncology Care Model?
Dr. Grubbs: OCM has shown that by providing oncologists with payment for care management – OCM participants receive $160 per patient, per month – the results are better care coordination and reduced hospital and emergency department visits. If CMS chooses to expand payments to all oncology providers, we could expect to see improved care for cancer patients.
Question: ASCO has advanced its own Patient-Centered Oncology Payment model. What are the main elements of this strategy and how does it differ from the Oncology Care Model?
Dr. Grubbs: The Patient-Centered Oncology Payment (PCOP) model is the result of input from a wide group of stakeholders, including providers, employers, and managed care organizations. In the coming months, ASCO will publish an updated copy of the PCOP model.
Our review of OCM is that the included prediction model and two-sided risk options place small, rural, and certain other practices at considerable peril because of imprecise and inconsistent cost predictions. PCOP takes a different approach. Rather than requiring that practices take on actuarial risk for total cost of care, PCOP includes a three-part performance methodology. Practices are measured on adherence to clinical treatment pathways; electronically capturable quality measures; and select, targeted cost-of-care measures. Practices that perform well in PCOP’s performance methodology receive increased incentive payments to fund further advancements in care.
Question: The PCOP model includes payments to oncology practices for participation in clinical trials. How might that drive a change in behavior in a typical practice?
Dr. Grubbs: Practices that enroll patients in clinical trials have the same or greater storage and handling requirements as those treated with standard treatments, yet forgo revenue associated with the Medicare Part B average sales price methodology. PCOP ensures that such practices are not disadvantaged for supporting clinical research.
Question: Are there other areas – such as tumor biomarker tests – in which a tailored payment approach would improve the quality of care?
Dr. Grubbs: Recent studies have shown that not all patients receive the appropriate genomic profiling and other tests necessary to ensure that they benefit from personalized therapies. Clinical treatment pathways have the ability to inform and measure diagnostic completeness to improve the quality of care.
Question: What are the barriers that are keeping oncology practices from participating in alternative payment models designed to improve care?
Dr. Grubbs: Some alternative payment models, such as OCM, place a high administrative burden on their participants. Manual reporting of measures and clinical data, complicated billing requirements, and lack of support from electronic health record vendors create barriers for expanded participation. Practices are also concerned about the financial risks placed upon participants; it is impractical to expect that physicians hire actuaries in order to participate in a Medicare program.
ASCO has offered support for OCM practices through its PracticeNET benchmarking program, but we have also proposed PCOP as an appropriate alternative, applicable to practices of all types and sizes.
Dr. Grubbs joined ASCO in 2015 as the vice president of the newly launched clinical affairs department. Before joining ASCO, Dr. Grubbs worked as a community oncologist and managing partner at Medical Oncology Hematology Consultants in Newark, Del. Dr. Grubbs is a volunteer and the principal investigator of the Delaware Christiana Care National Cancer Institute Community Oncology Research Program. Dr. Grubbs reported having no financial disclosures.
The Oncology Care Model is a value-based payment approach aimed at encouraging coordinated cancer care through targeted bonus payments to practices. The payment experiment was launched by the Centers for Medicare & Medicaid Services in 2016 and now includes 175 practices and 10 payers. It is set to end in 2021. As agency officials consider whether to continue the program, Stephen S. Grubbs, MD, vice president for clinical affairs at the American Society of Clinical Oncology, weighs in on the model’s track record and its future.
Question: How would you rate the Oncology Care Model in helping to drive practice transformation?
Dr. Grubbs: Participants in the Oncology Care Model (OCM) have demonstrated improved care coordination, psychosocial support, use of risk assessment tools, and other strategies to lower costs and adverse events. Over the past 2 years, ASCO has accepted numerous posters, articles, and abstracts from OCM participants on their outstanding work to advance cancer care delivery.
Question: Should the model be extended beyond 2021?
Dr. Grubbs: Changes to the model are necessary prior to a significant extension or expansion. Some have suggested that CMS extend OCM for an additional year with current participants. This would give CMS time to consider input from all stakeholders on its eventual replacement.
Question: What additional resources or payments do oncology practices need to be more successful in meeting the goals of the Oncology Care Model?
Dr. Grubbs: OCM has shown that by providing oncologists with payment for care management – OCM participants receive $160 per patient, per month – the results are better care coordination and reduced hospital and emergency department visits. If CMS chooses to expand payments to all oncology providers, we could expect to see improved care for cancer patients.
Question: ASCO has advanced its own Patient-Centered Oncology Payment model. What are the main elements of this strategy and how does it differ from the Oncology Care Model?
Dr. Grubbs: The Patient-Centered Oncology Payment (PCOP) model is the result of input from a wide group of stakeholders, including providers, employers, and managed care organizations. In the coming months, ASCO will publish an updated copy of the PCOP model.
Our review of OCM is that the included prediction model and two-sided risk options place small, rural, and certain other practices at considerable peril because of imprecise and inconsistent cost predictions. PCOP takes a different approach. Rather than requiring that practices take on actuarial risk for total cost of care, PCOP includes a three-part performance methodology. Practices are measured on adherence to clinical treatment pathways; electronically capturable quality measures; and select, targeted cost-of-care measures. Practices that perform well in PCOP’s performance methodology receive increased incentive payments to fund further advancements in care.
Question: The PCOP model includes payments to oncology practices for participation in clinical trials. How might that drive a change in behavior in a typical practice?
Dr. Grubbs: Practices that enroll patients in clinical trials have the same or greater storage and handling requirements as those treated with standard treatments, yet forgo revenue associated with the Medicare Part B average sales price methodology. PCOP ensures that such practices are not disadvantaged for supporting clinical research.
Question: Are there other areas – such as tumor biomarker tests – in which a tailored payment approach would improve the quality of care?
Dr. Grubbs: Recent studies have shown that not all patients receive the appropriate genomic profiling and other tests necessary to ensure that they benefit from personalized therapies. Clinical treatment pathways have the ability to inform and measure diagnostic completeness to improve the quality of care.
Question: What are the barriers that are keeping oncology practices from participating in alternative payment models designed to improve care?
Dr. Grubbs: Some alternative payment models, such as OCM, place a high administrative burden on their participants. Manual reporting of measures and clinical data, complicated billing requirements, and lack of support from electronic health record vendors create barriers for expanded participation. Practices are also concerned about the financial risks placed upon participants; it is impractical to expect that physicians hire actuaries in order to participate in a Medicare program.
ASCO has offered support for OCM practices through its PracticeNET benchmarking program, but we have also proposed PCOP as an appropriate alternative, applicable to practices of all types and sizes.
Dr. Grubbs joined ASCO in 2015 as the vice president of the newly launched clinical affairs department. Before joining ASCO, Dr. Grubbs worked as a community oncologist and managing partner at Medical Oncology Hematology Consultants in Newark, Del. Dr. Grubbs is a volunteer and the principal investigator of the Delaware Christiana Care National Cancer Institute Community Oncology Research Program. Dr. Grubbs reported having no financial disclosures.
The Oncology Care Model is a value-based payment approach aimed at encouraging coordinated cancer care through targeted bonus payments to practices. The payment experiment was launched by the Centers for Medicare & Medicaid Services in 2016 and now includes 175 practices and 10 payers. It is set to end in 2021. As agency officials consider whether to continue the program, Stephen S. Grubbs, MD, vice president for clinical affairs at the American Society of Clinical Oncology, weighs in on the model’s track record and its future.
Question: How would you rate the Oncology Care Model in helping to drive practice transformation?
Dr. Grubbs: Participants in the Oncology Care Model (OCM) have demonstrated improved care coordination, psychosocial support, use of risk assessment tools, and other strategies to lower costs and adverse events. Over the past 2 years, ASCO has accepted numerous posters, articles, and abstracts from OCM participants on their outstanding work to advance cancer care delivery.
Question: Should the model be extended beyond 2021?
Dr. Grubbs: Changes to the model are necessary prior to a significant extension or expansion. Some have suggested that CMS extend OCM for an additional year with current participants. This would give CMS time to consider input from all stakeholders on its eventual replacement.
Question: What additional resources or payments do oncology practices need to be more successful in meeting the goals of the Oncology Care Model?
Dr. Grubbs: OCM has shown that by providing oncologists with payment for care management – OCM participants receive $160 per patient, per month – the results are better care coordination and reduced hospital and emergency department visits. If CMS chooses to expand payments to all oncology providers, we could expect to see improved care for cancer patients.
Question: ASCO has advanced its own Patient-Centered Oncology Payment model. What are the main elements of this strategy and how does it differ from the Oncology Care Model?
Dr. Grubbs: The Patient-Centered Oncology Payment (PCOP) model is the result of input from a wide group of stakeholders, including providers, employers, and managed care organizations. In the coming months, ASCO will publish an updated copy of the PCOP model.
Our review of OCM is that the included prediction model and two-sided risk options place small, rural, and certain other practices at considerable peril because of imprecise and inconsistent cost predictions. PCOP takes a different approach. Rather than requiring that practices take on actuarial risk for total cost of care, PCOP includes a three-part performance methodology. Practices are measured on adherence to clinical treatment pathways; electronically capturable quality measures; and select, targeted cost-of-care measures. Practices that perform well in PCOP’s performance methodology receive increased incentive payments to fund further advancements in care.
Question: The PCOP model includes payments to oncology practices for participation in clinical trials. How might that drive a change in behavior in a typical practice?
Dr. Grubbs: Practices that enroll patients in clinical trials have the same or greater storage and handling requirements as those treated with standard treatments, yet forgo revenue associated with the Medicare Part B average sales price methodology. PCOP ensures that such practices are not disadvantaged for supporting clinical research.
Question: Are there other areas – such as tumor biomarker tests – in which a tailored payment approach would improve the quality of care?
Dr. Grubbs: Recent studies have shown that not all patients receive the appropriate genomic profiling and other tests necessary to ensure that they benefit from personalized therapies. Clinical treatment pathways have the ability to inform and measure diagnostic completeness to improve the quality of care.
Question: What are the barriers that are keeping oncology practices from participating in alternative payment models designed to improve care?
Dr. Grubbs: Some alternative payment models, such as OCM, place a high administrative burden on their participants. Manual reporting of measures and clinical data, complicated billing requirements, and lack of support from electronic health record vendors create barriers for expanded participation. Practices are also concerned about the financial risks placed upon participants; it is impractical to expect that physicians hire actuaries in order to participate in a Medicare program.
ASCO has offered support for OCM practices through its PracticeNET benchmarking program, but we have also proposed PCOP as an appropriate alternative, applicable to practices of all types and sizes.
Dr. Grubbs joined ASCO in 2015 as the vice president of the newly launched clinical affairs department. Before joining ASCO, Dr. Grubbs worked as a community oncologist and managing partner at Medical Oncology Hematology Consultants in Newark, Del. Dr. Grubbs is a volunteer and the principal investigator of the Delaware Christiana Care National Cancer Institute Community Oncology Research Program. Dr. Grubbs reported having no financial disclosures.
Inpatient care declining among family physicians
and by 2017, only one of four FPs was practicing hospital medicine, according to the American Academy of Family Physicians.
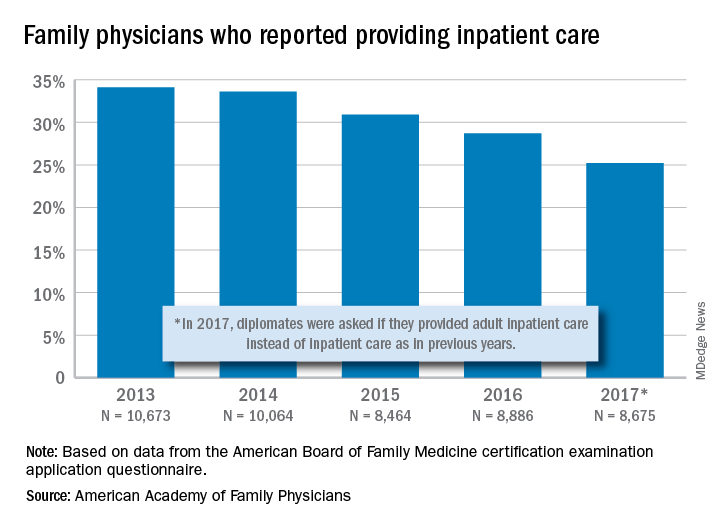
The share of family physicians who provided hospital care went from 34.1% in 2013 to 25.2% in 2017, for a relative decrease of 26% that left only a quarter of FPs seeing inpatients, based on data from the annual American Board of Family Medicine certification exam application questionnaire. For the 5-year period, 46,762 individuals were included in the study sample of FPs in direct patient care.
“As observed in other domains (prenatal care, home visits, nursing home care, and obstetric care), this study adds to the evidence demonstrating contracting scope of practice among FPs,” Anuradha Jetty, MPH, of the AAFP’s Robert Graham Center in Washington, D.C., and associates said in a recent Policy Brief published in the Journal of the American Board of Family Medicine.
Much of that contraction is occurring among new family physicians who can’t “find positions that allow them to use all their expertise,” the investigators said in a separate statement. The AAFP had previously reported that about 40% of family physicians had full hospital privileges in 2018, compared with 56% in 2012.
Many new FPs now work in large multispecialty practices or hospital systems, and “[some] of these employers dictate scope of practice, limiting family physicians to coordinating outpatient care and relying on subspecialists or hospitalists to provide inpatient care,” they noted.
and by 2017, only one of four FPs was practicing hospital medicine, according to the American Academy of Family Physicians.

The share of family physicians who provided hospital care went from 34.1% in 2013 to 25.2% in 2017, for a relative decrease of 26% that left only a quarter of FPs seeing inpatients, based on data from the annual American Board of Family Medicine certification exam application questionnaire. For the 5-year period, 46,762 individuals were included in the study sample of FPs in direct patient care.
“As observed in other domains (prenatal care, home visits, nursing home care, and obstetric care), this study adds to the evidence demonstrating contracting scope of practice among FPs,” Anuradha Jetty, MPH, of the AAFP’s Robert Graham Center in Washington, D.C., and associates said in a recent Policy Brief published in the Journal of the American Board of Family Medicine.
Much of that contraction is occurring among new family physicians who can’t “find positions that allow them to use all their expertise,” the investigators said in a separate statement. The AAFP had previously reported that about 40% of family physicians had full hospital privileges in 2018, compared with 56% in 2012.
Many new FPs now work in large multispecialty practices or hospital systems, and “[some] of these employers dictate scope of practice, limiting family physicians to coordinating outpatient care and relying on subspecialists or hospitalists to provide inpatient care,” they noted.
and by 2017, only one of four FPs was practicing hospital medicine, according to the American Academy of Family Physicians.

The share of family physicians who provided hospital care went from 34.1% in 2013 to 25.2% in 2017, for a relative decrease of 26% that left only a quarter of FPs seeing inpatients, based on data from the annual American Board of Family Medicine certification exam application questionnaire. For the 5-year period, 46,762 individuals were included in the study sample of FPs in direct patient care.
“As observed in other domains (prenatal care, home visits, nursing home care, and obstetric care), this study adds to the evidence demonstrating contracting scope of practice among FPs,” Anuradha Jetty, MPH, of the AAFP’s Robert Graham Center in Washington, D.C., and associates said in a recent Policy Brief published in the Journal of the American Board of Family Medicine.
Much of that contraction is occurring among new family physicians who can’t “find positions that allow them to use all their expertise,” the investigators said in a separate statement. The AAFP had previously reported that about 40% of family physicians had full hospital privileges in 2018, compared with 56% in 2012.
Many new FPs now work in large multispecialty practices or hospital systems, and “[some] of these employers dictate scope of practice, limiting family physicians to coordinating outpatient care and relying on subspecialists or hospitalists to provide inpatient care,” they noted.