User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
PSA cancer screening: A case for shared decision-making
Prostate cancer is the most frequently diagnosed cancer in men and the third leading cause of cancer death in men worldwide.1 An estimated 174,650 new cases are diagnosed each year in the United States; 31,620 American men die annually from the disease.2 Although prostate cancer can be a serious disease, many men do not die from it. In fact, 2.9 million men who were diagnosed with prostate cancer at some point are alive today.3
Risk factors. Prostate cancer develops mainly in men ages ≥ 65 years and rarely occurs before age 40. In addition to age, family history and African American ethnicity are the major nonmodifiable risk factors for prostate cancer.4 From the 1970s to the most recent statistical analysis of the National Cancer Institute Surveillance, Epidemiology, and End Results (SEER) program, African American men have continued to have significantly higher incidence of, and mortality rates from, prostate cancer than their European American counterparts. African American men are also more likely than men of European ancestry to have aggressive prostate cancers.5 Other risk factors include geographic location (higher risk in Northern Europe, North America, and Australia; lower risk in Asia, Africa, and South and Central America), mutations in the BRCA2 gene, and hereditary non-polyposis colon cancer syndrome.4
Prostate-specific antigen (PSA) was first used as a screening tool for prostate cancer in 1991.6 Prostate cancer incidence, especially organ-confined disease, has dramatically increased since then.7 PSA testing has a low sensitivity and specificity for the detection of prostate cancer, and there is no clear threshold at which biopsy can or should be offered. The most commonly used cutoff value of 4 ng/mL has a false-positive rate of about 70%.8
Benign prostatic conditions such as hypertrophy and infection can elevate PSA levels. In addition, the PSA test does not distinguish between aggressive and slow-growing cancers, and about 15% of patients with prostate cancer have a normal PSA level.9
A word about the digital rectal exam. While PSA testing has been the mainstay of prostate cancer screening, a few studies have included digital rectal exam (DRE) in their protocols. Data from the Prostate, Lung, Colorectal, and Ovarian (PLCO) Cancer Screening Trial showed that DRE captured an additional 2% of men with prostate cancer in the setting of a normal PSA test result.10 In the Rotterdam arm of the European Randomized Study of Screening for Prostate Cancer (ERSPC) trial, the overall detection rate for prostate cancer was found to be better when DRE was combined with PSA and prostate biopsy than when DRE was used alone (4.5% vs 2.5%).11 Nevertheless, generally speaking, DRE can be omitted in the era of PSA screening.
Screening guidelines vary
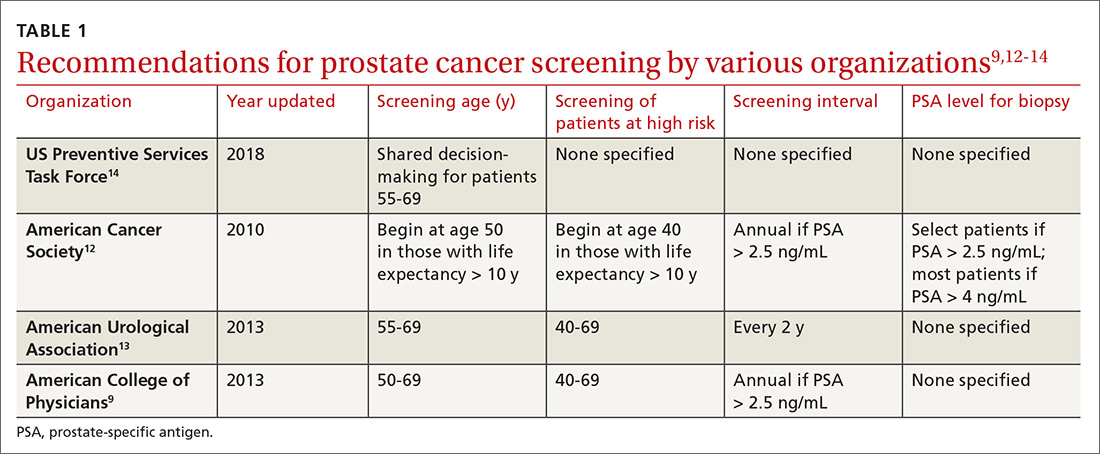
Recommendations for prostate cancer screening vary by organization and are summarized in TABLE 1.9,12-14 In 2012, the US Preventive Services Task Force (USPSTF) recommended against PSA-based screening for prostate cancer (Category D).15 In 2018, USPSTF provided an update with a new recommendation that clinicians inform men ages 55 to 69 years about the potential benefits and harms of PSA-based screening (Category C).14 The USPSTF continues to recommend against PSA-based screening for men ages ≥ 70 years (Category D).14
Does PSA-based screening improve patient-centered outcomes?
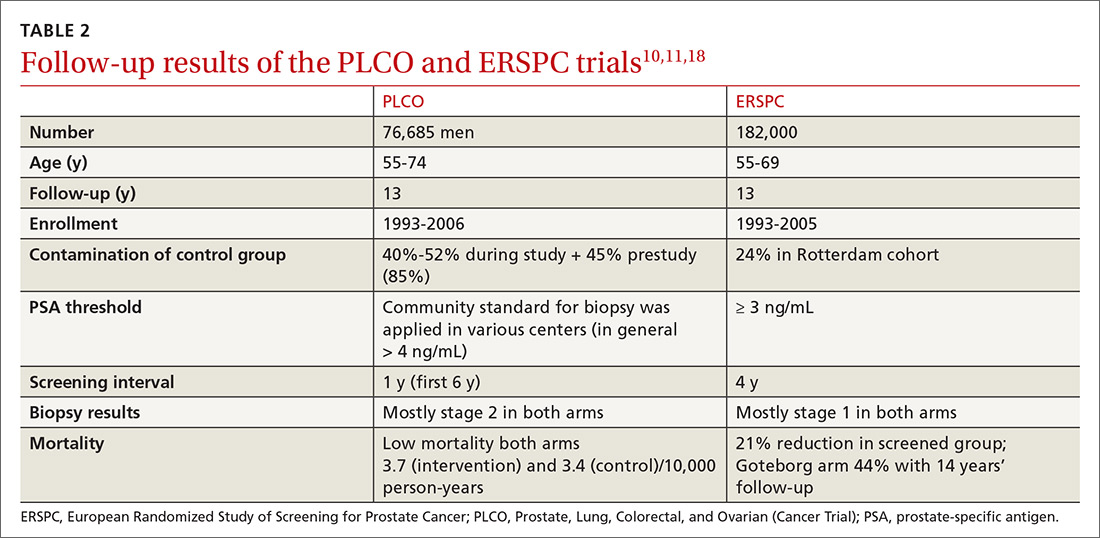
Several randomized controlled trials (RCTs) such as the Quebec Prospective Randomized Controlled Trial,16 the Norrköping Sweden Study,17 ERSPC,11 and PLCO10 have been conducted to assess the benefits of PSA testing. PLCO and ERSPC have contributed significantly to our understanding of prostate cancer screening even though their 13-year follow-up results are conflicting (TABLE 2).10,11,18
Continue to: In the ERSPC 13-year follow-up publication...
In the ERSPC 13-year follow-up publication, the authors concluded that a substantial reduction in prostate cancer mortality is attributable to testing with PSA.18 Despite limitations in the study design (eg, France entered after 2 years, screening intervals varied between 2 and 4 years, biopsy indications varied, and screening was discontinued at different times), PSA screening detected more prostate cancer than was detected in the control arm (10.2% vs 6.8%).
In the initial 11 years of follow-up, the study group experienced a 21% reduction in prostate cancer mortality, even though the absolute decrease ranged from only 0.6% (545 per 89,352) to 0.5% (355 per 72,891). The updated absolute risk reduction of death from prostate cancer at 13 years of follow-up showed a larger benefit: 0.11 per 1000 person-years or 1.28 per 1000 men randomized, which is equivalent to 1 prostate cancer death averted per 781 (95% confidence interval [CI], 490-1929) men invited for screening, or 1 per 27 (17-66) additional prostate cancers detected.
The PLCO trial did not show any significant difference in prostate cancer detection (11.1% screened vs 9.9% control), and there was no improvement in prostate cancer mortality (3.7 vs 3.4 death per 10,000 person-years).10 However, the PLCO trial suffered from issues of contamination, which may have influenced the overall results. About 52% of men in the control (usual care) group received a PSA test at some point during the study. And more than two-thirds of the men who had a prostate biopsy because of a positive PSA test did not have prostate cancer.
Community standards for the PSA threshold for biopsy were applied in various centers (> 4 ng/ml in general) in PLCO, whereas in ERSPC, a cut-off PSA value ≥ 3 ng/mL was used for biopsy. Because of the lower PSA threshold, ERSPC may have identified cancers that would have had good outcomes without any intervention.
The harms of PSA screening
While it is unclear whether PSA screening results in any improvement in patient-centered outcomes, it does lead to downstream intervention due to overdiagnosis, which precipitates unnecessary anxiety, biopsies, and overtreatment (eg, excess radiation, overuse of androgen deprivation therapy).19 Biopsies carry the risk of hematuria (22.6%), hematospermia (50.4%), and urinary tract infection.20 Data from SEER-Medicare showed that prostate biopsy was associated with a 2.65-fold increased risk of hospitalization within 30 days of the procedure compared to a control population.21
Continue to: Overdiagnosis leads to overtreatment...
Overdiagnosis leads to overtreatment of low-risk prostate cancer. Both traditional treatment options for prostate cancer—radical prostatectomy and radiotherapy—are associated with urinary incontinence, erectile dysfunction, and issues with bowel function.22,23
The Prostate Cancer Intervention vs Observation Trial (PIVOT),24 the Scandinavian Prostate Cancer Group Study Number 4 (SPCG-4),25 and the Prostate Testing for Cancer and Treatment (ProtecT) trial,22,23 are the major RCTs that looked at the outcomes of treatment modalities for localized prostate cancer in the modern era of PSA testing.
PIVOT compared passive observation with radical prostatectomy.24 After 20 years of follow-up on 731 patients, the researchers concluded that radical prostatectomy did not reduce all-cause or prostate cancer–related mortality (TABLE 3).24

SPCG-4 showed survival benefits for men who underwent radical prostatectomy compared with men in a watchful waiting group, but only 5% of the study cohort had cancer detected by PSA screening (TABLE 4).25 The rest had either palpable tumors or symptoms of a tumor.

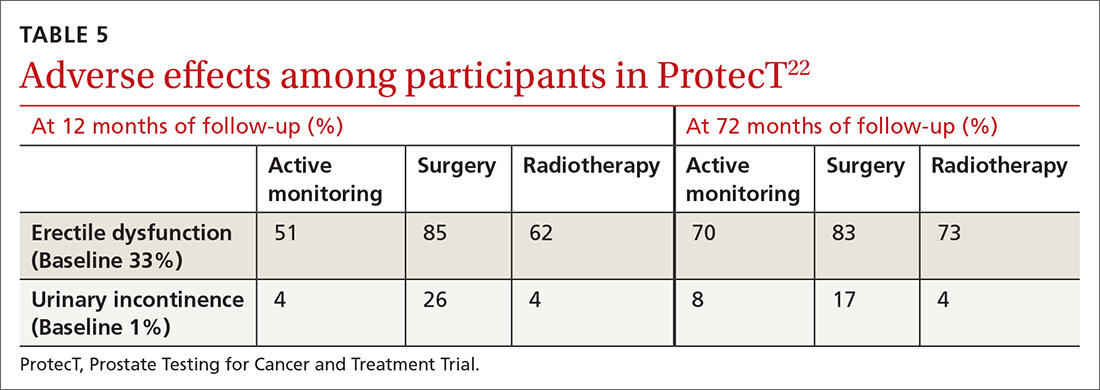
ProtecT, which followed patients with localized prostate cancer for more than 10 years,compared the outcomes and adverse effects of active surveillance, radical prostatectomy, and radiotherapy.23 Prostate cancer–specific mortality was low irrespective of the treatment,23 and there was no significant difference in all-cause mortality or prostate cancer–specific mortality between the 3 treatment groups.23 The active surveillance group had considerably fewer adverse events.22,23 The incidence rates of erectile dysfunction and urinary incontinence at the 1- and 6-year follow-up marks are outlined in TABLE 5.22
Continue to: The purpose of active monitoring...
The purpose of active monitoring is to minimize overtreatment by avoiding immediate radical intervention. Radical treatments with curative intent can be undertaken at any point while patients are being actively monitored. It is important to note that the active monitoring that took place in ProtecT23 was very different from the passive surveillance of PIVOT24 and SPCG-4.25 In ProtecT, once an elevated serum PSA level was noted, PSA levels were monitored every 3 months in the first year and every 6 to 12 months thereafter.23 Triggers to reassess patients and consider a change in clinical management were based largely on changes in PSA levels. Participants with an increase of at least 50% in PSA level during the previous 12 months were offered either continued monitoring or treatment after further testing.
Making individualized decisions about prostate cancer screening
Traditionally, the goal of cancer screening has been to maximize the number of people screened. Generally, the information provided to patients about cancer screening emphasizes the benefits and minimizes the harms. Recently, however, there has been a shift in communication about cancer screening with the emphasis now being placed on informed decision-making and encouraging patients to make individual decisions about screening participation.26
The treatment option of active surveillance, with its lower incidence of adverse outcomes, is an important reason for patients to make individualized decisions about prostate cancer screening.
Another reason relates to 5-alpha-reductase inhibitors. Although their role in the management of prostate cancer is currently not well defined, a reduction of almost 25% in the risk of prostate cancer and improvement in the performance of PSA has been reported.27
And yet another reason is that there are alternate strategies to manage the majority of patients who have been diagnosed with low-risk disease through transrectal ultrasound biopsy. The ERSPC study mentions multiparametric magnetic resonance imaging combined with targeted biopsy to identify high-grade disease.28,29 Genetic and epigenetic assays of the biopsied tissue can help grade disease based on aggressiveness.30 Transperineal mapping biopsy using a mapping software program can identify specific disease sites within the prostate gland, so that patients can be offered the option of targeted therapy.30
Continue to: Applying shared decision-making to prostate cancer screening
Applying shared decision-making to prostate cancer screening
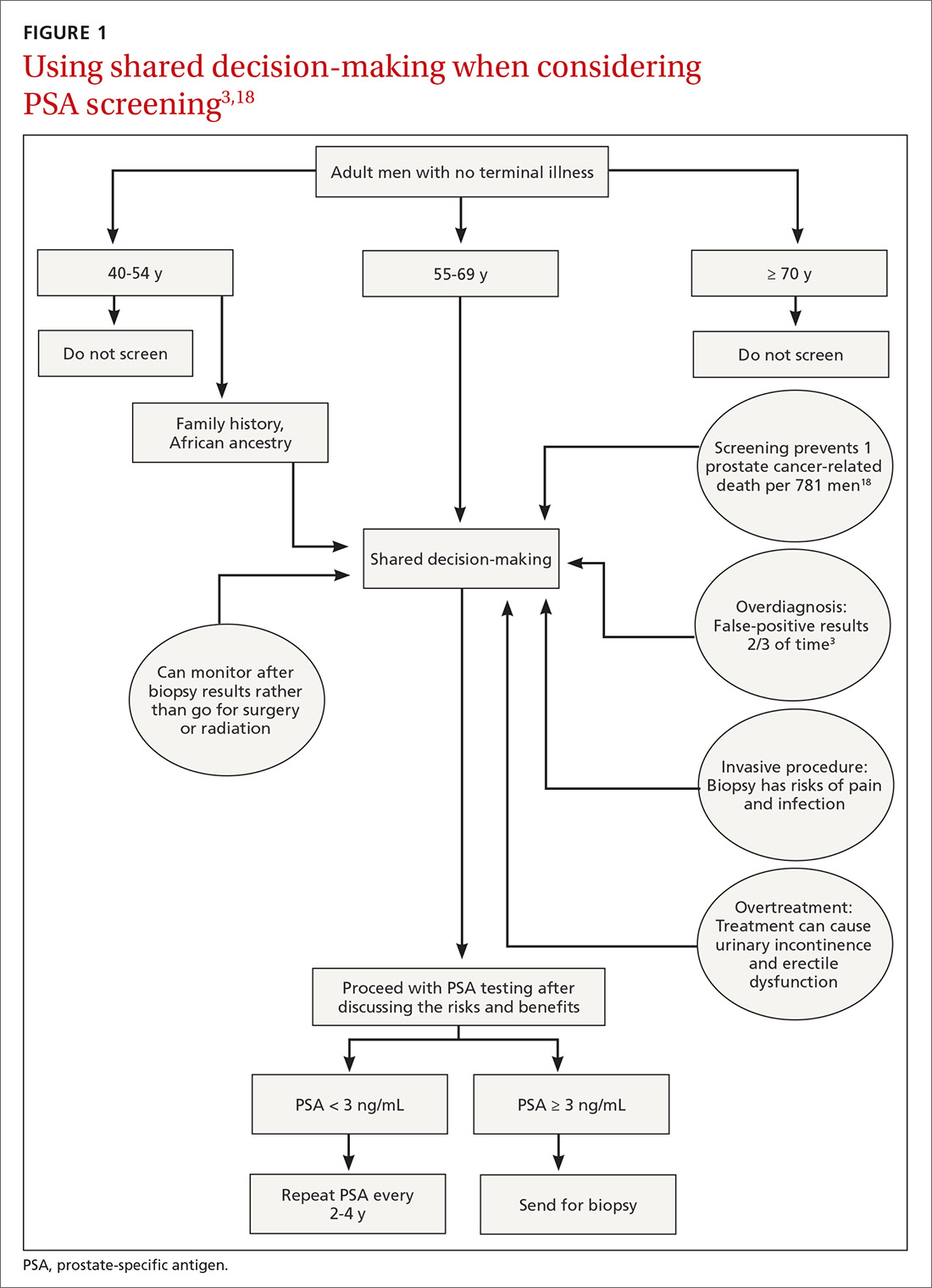
Balancing errors of omission with errors of commission is challenging. Shared decision-making (SDM) is an approach whereby clinicians and patients share the best available evidence when faced with the task of medical decision-making and in which patients are supported while they consider their options and achieve their preferences.31 SDM is well supported by evidence from a number of RCTs and results in increased knowledge, involvement, and confidence on the part of patients.32 An individualized approach using the schematic diagram (FIGURE 13,18) may be helpful.
Barriers to SDM success. Many factors can interfere with the success of SDM including limited or poor communication; lack of time during busy office visits; and patients’ cultural, informational, and/or emotional needs. To improve patient-centered communication, we can: (1) make information understandable and available to patients and families; (2) prioritize training in communication; (3) use decision aid tools to facilitate communication; and (4) work to improve the payment model to incentivize patient-centered communication. Tools that facilitate SDM include videotapes, patient group discussions, brief scripts read to patients, and informational pamphlets. One such tool is the American Society for Clinical Oncology’s decision aid tool for PSA testing.33
Limited knowledge among patients. Decisions regarding treatment among men diagnosed with localized prostate cancer can be difficult because there are several treatment options with similar prognoses, but there are differences in adverse effects. One population-based cohort study of men with newly diagnosed localized prostate cancer found that most men had significant knowledge deficits regarding the survival benefits of the 2 major treatment options—surgery and radiation.34 In a large population-based study, 38% of men with localized prostate cancer reported receiving help from their primary care providers in the decision-making process for treatment.35
Learning to employ SDM. Elwyn et al proposed a 3-step model to incorporate SDM into clinical practice.31 They described key steps that include: choice talk (making sure patients are informed about the reasonable options), option talk (providing more detailed information about the options), and decision talk (supporting the work of patients considering their preferences and deciding what is best). Properly employing these methods requires training using simulations.31
The bottom line
Although current guidelines regarding PSA screening differ by organization, generally speaking PSA screening should be offered only to men with a life expectancy > 10 years. The PSA test has low sensitivity and specificity and lacks a clear cut-off value that warrants prostate biopsy. Men who choose to have PSA testing increase their chances of detecting prostate cancer, but most prostate cancers are slow growing and do not cause death. The decision to undergo PSA screening should be made by both the provider and the patient, after a discussion of the limited benefits and associated harms. The interval of follow-up screening may vary from 2 to 4 years depending on patient age, level of PSA, and whether a patient is taking medications such as 5-alpha-reductase inhibitors.
CORRESPONDENCE
Jaividhya Dasarathy, MD, FAAFP, 2500 Metro Health Medical Drive, Cleveland, Ohio 44109; [email protected].
1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016;66:7-30.
2. National Cancer Institute Surveillance, Epidemiology, and End Results Program. Cancer Stat Facts: Prostate Cancer. https://seer.cancer.gov/statfacts/html/prost.html. Accessed January 16, 2020.
3. American Cancer Society. Key statistics for prostate cancer. Last revised August 1, 2019. www.cancer.org/cancer/prostate-cancer/about/key-statistics.html. Accessed January 16, 2020.
4. Brawley OW. Trends in prostate cancer in the United States. J Natl Cancer Inst Monogr. 2012;2012:152-156.
5. Powell IJ. Epidemiology and pathophysiology of prostate cancer in African-American men. J Urol. 2007;177:444-449.
6. Catalona WJ, Smith DS, Ratliff TL, et al. Measurement of prostate-specific antigen in serum as a screening test for prostate cancer. N Engl J Med. 1991;324:1156-1161.
7. Jacobsen SJ, Katusic SK, Bergstraih EJ. Incidence of prostate cancer diagnosis in the eras before and after serum prostate-specific antigen testing. JAMA. 1995;274:1445-1449.
8. Mistry K, Cable G. Meta-analysis of prostate-specific antigen and digital rectal examination as screening tests for prostate carcinoma. J Am Board Fam Pract. 2003;16:95-101.
9. Qaseem A, Barry MJ, Denberg TD, et al. Screening for prostate cancer: a guidance statement from the Clinical Guidelines Committee of the American College of Physicians. Ann Int Med. 2013;158:761-769.
10. Andriole GL, Crawford ED, Grubb RL 3rd, et al. Prostate cancer screening in the randomized Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial: mortality results after 13 years of follow-up. J Natl Cancer Inst. 2012;104:125-132.
11. Schröder FH, Hugosson J, Roobol MJ, et al. Screening and prostate-cancer mortality in a randomized European study. N Engl J Med. 2009;360:1320-1328.
12. American Cancer Society. American Cancer Society recommendations for prostate cancer early detection. Last revised August 1, 2019. www.cancer.org/cancer/prostate-cancer/detection-diagnosis-staging/acs-recommendations.html. Accessed January 16, 2020.
13. American Urologic Association. Early detection of prostate cancer (2018). Reviewed 2018. https://www.auanet.org/guidelines/prostate-cancer-early-detection-guideline. Accessed January 16, 2020.
14. US Preventive Services Task Force. Screening for Prostate Cancer: US Preventive Services Task Force Recommendation Statement. JAMA. 2018;319:1901-1913.
15 Moyer VA. Screening for prostate cancer: US Preventive Services Task Force recommendation statement. Ann Int Med. 2012;157:120-134.
16. Labrie F, Candas B, Dupont A, et al. Screening decreases prostate cancer death: first analysis of the 1988 Quebec prospective randomized controlled trial. Prostate. 1999;38:83-91.
17. Sandblom G, Varenhorst E, Rosell J, et al. Randomised prostate cancer screening trial: 20-year follow-up. BMJ. 2011;342:d1539.
18. Schröder FH, Hugosson J, Roobol MJ, et al. Screening and prostate cancer mortality: results of the European Randomized Study of Screening for Prostate Cancer (ERSPC) at 13 years of follow-up. Lancet. 2014;384:2027-2035.
19. McNaughton-Collins M, Fowler FJ Jr, Caubet JF, et al. Psychological effects of a suspicious prostate cancer screening test followed by a benign biopsy result. Am J Med. 2004;117:719-725.
20 Raaijmakers R, Kirkels WJ, Roobol MJ, et al. Complication rates and risk factors of 5802 transrectal ultrasound-guided sextant biopsies of the prostate within a population-based screening program. Urology. 2002;60:826-830.
21. Loeb S, Carter HB, Berndt SI, et al. Complications after prostate biopsy: data from SEER-Medicare. J Urol. 2011;186:1830-1834.
22. Donovan J, Hamdy F, Lane J, et al. Patient-reported outcomes after monitoring, surgery, or radiotherapy for prostate cancer. N Engl J Med. 2016;375:1425-1437.
23. Hamdy FC, Donovan JL, Lane JA, et al. 10-year outcomes after monitoring, surgery, or radiotherapy for localized prostate cancer. N Engl J Med. 2016;375:1415-1424.
24. Wilt TJ, Jones KM, Barry MJ, et al. Follow-up of prostatectomy versus observation for early prostate cancer. N Engl J Med. 2017;377:132-142.
25. Bill-Axelson A, Holmberg L, Garmo H, et al. Radical prostatectomy or watchful waiting in early prostate cancer. N Engl J Med. 2018;379:2319-2329.
26. Hersch JK, Nickel BL, Ghanouni A, et al. Improving communication about cancer screening: moving towards informed decision making. Public Health Res Pract. 2017;27(2).
27. Cuzick J, Thorat MA, Andriole G, et al. Prevention and early detection of prostate cancer. Lancet Oncol. 2014;15:e484-e492.
28. Pinto PA, Chung PH, Rastinehad AR, et al. Magnetic resonance imaging/ultrasound fusion guided prostate biopsy improves cancer detection following transrectal ultrasound biopsy and correlates with multiparametric magnetic resonance imaging. J Urol. 2011;186:1281-1285.
29. Kuru TH, Roethke MC, Seidenader J, et al. Critical evaluation of magnetic resonance imaging targeted, transrectal ultrasound guided transperineal fusion biopsy for detection of prostate cancer. J Urol. 2013;190:1380-1386.
30. Crawford ED, Rove KO, Barqawi AB, et al. Clinical-pathologic correlation between transperineal mapping biopsies of the prostate and three-dimensional reconstruction of prostatectomy specimens. Prostate. 2013;73:778-787.
31. Elwyn G, Frosch D, Thomson R, et al. Shared decision making: a model for clinical practice. J Gen Intern Med. 2012;27:1361-1367.
32. Stacey D, Légaré F, Lewis K, et al. Decision aids for people facing health treatment or screening decisions. Cochrane Database Syst Rev. 2017;4:CD001431.
33. ASCO. Decision aid tool: prostate cancer screening with PSA testing. https://www.asco.org/sites/new-www.asco.org/files/content-files/practice-and-guidelines/documents/2012-psa-pco-decision-aid.pdf. Accessed January 16, 2020.
34. Daum LM, Reamer EN, Ruterbusch JJ, et al. Patient knowledge and qualities of treatment decisions for localized prostate cancer. J Am Board Fam Med. 2017;30:288-297.
35. Radhakrishnan A, Grande D, Ross M, et al. When primary care providers (PCPs) help patients choose prostate cancer treatment. J Am Board Fam Med. 2017;30:298-307.
Prostate cancer is the most frequently diagnosed cancer in men and the third leading cause of cancer death in men worldwide.1 An estimated 174,650 new cases are diagnosed each year in the United States; 31,620 American men die annually from the disease.2 Although prostate cancer can be a serious disease, many men do not die from it. In fact, 2.9 million men who were diagnosed with prostate cancer at some point are alive today.3
Risk factors. Prostate cancer develops mainly in men ages ≥ 65 years and rarely occurs before age 40. In addition to age, family history and African American ethnicity are the major nonmodifiable risk factors for prostate cancer.4 From the 1970s to the most recent statistical analysis of the National Cancer Institute Surveillance, Epidemiology, and End Results (SEER) program, African American men have continued to have significantly higher incidence of, and mortality rates from, prostate cancer than their European American counterparts. African American men are also more likely than men of European ancestry to have aggressive prostate cancers.5 Other risk factors include geographic location (higher risk in Northern Europe, North America, and Australia; lower risk in Asia, Africa, and South and Central America), mutations in the BRCA2 gene, and hereditary non-polyposis colon cancer syndrome.4
Prostate-specific antigen (PSA) was first used as a screening tool for prostate cancer in 1991.6 Prostate cancer incidence, especially organ-confined disease, has dramatically increased since then.7 PSA testing has a low sensitivity and specificity for the detection of prostate cancer, and there is no clear threshold at which biopsy can or should be offered. The most commonly used cutoff value of 4 ng/mL has a false-positive rate of about 70%.8
Benign prostatic conditions such as hypertrophy and infection can elevate PSA levels. In addition, the PSA test does not distinguish between aggressive and slow-growing cancers, and about 15% of patients with prostate cancer have a normal PSA level.9
A word about the digital rectal exam. While PSA testing has been the mainstay of prostate cancer screening, a few studies have included digital rectal exam (DRE) in their protocols. Data from the Prostate, Lung, Colorectal, and Ovarian (PLCO) Cancer Screening Trial showed that DRE captured an additional 2% of men with prostate cancer in the setting of a normal PSA test result.10 In the Rotterdam arm of the European Randomized Study of Screening for Prostate Cancer (ERSPC) trial, the overall detection rate for prostate cancer was found to be better when DRE was combined with PSA and prostate biopsy than when DRE was used alone (4.5% vs 2.5%).11 Nevertheless, generally speaking, DRE can be omitted in the era of PSA screening.
Screening guidelines vary
Recommendations for prostate cancer screening vary by organization and are summarized in TABLE 1.9,12-14 In 2012, the US Preventive Services Task Force (USPSTF) recommended against PSA-based screening for prostate cancer (Category D).15 In 2018, USPSTF provided an update with a new recommendation that clinicians inform men ages 55 to 69 years about the potential benefits and harms of PSA-based screening (Category C).14 The USPSTF continues to recommend against PSA-based screening for men ages ≥ 70 years (Category D).14

Does PSA-based screening improve patient-centered outcomes?
Several randomized controlled trials (RCTs) such as the Quebec Prospective Randomized Controlled Trial,16 the Norrköping Sweden Study,17 ERSPC,11 and PLCO10 have been conducted to assess the benefits of PSA testing. PLCO and ERSPC have contributed significantly to our understanding of prostate cancer screening even though their 13-year follow-up results are conflicting (TABLE 2).10,11,18

Continue to: In the ERSPC 13-year follow-up publication...
In the ERSPC 13-year follow-up publication, the authors concluded that a substantial reduction in prostate cancer mortality is attributable to testing with PSA.18 Despite limitations in the study design (eg, France entered after 2 years, screening intervals varied between 2 and 4 years, biopsy indications varied, and screening was discontinued at different times), PSA screening detected more prostate cancer than was detected in the control arm (10.2% vs 6.8%).
In the initial 11 years of follow-up, the study group experienced a 21% reduction in prostate cancer mortality, even though the absolute decrease ranged from only 0.6% (545 per 89,352) to 0.5% (355 per 72,891). The updated absolute risk reduction of death from prostate cancer at 13 years of follow-up showed a larger benefit: 0.11 per 1000 person-years or 1.28 per 1000 men randomized, which is equivalent to 1 prostate cancer death averted per 781 (95% confidence interval [CI], 490-1929) men invited for screening, or 1 per 27 (17-66) additional prostate cancers detected.
The PLCO trial did not show any significant difference in prostate cancer detection (11.1% screened vs 9.9% control), and there was no improvement in prostate cancer mortality (3.7 vs 3.4 death per 10,000 person-years).10 However, the PLCO trial suffered from issues of contamination, which may have influenced the overall results. About 52% of men in the control (usual care) group received a PSA test at some point during the study. And more than two-thirds of the men who had a prostate biopsy because of a positive PSA test did not have prostate cancer.
Community standards for the PSA threshold for biopsy were applied in various centers (> 4 ng/ml in general) in PLCO, whereas in ERSPC, a cut-off PSA value ≥ 3 ng/mL was used for biopsy. Because of the lower PSA threshold, ERSPC may have identified cancers that would have had good outcomes without any intervention.
The harms of PSA screening
While it is unclear whether PSA screening results in any improvement in patient-centered outcomes, it does lead to downstream intervention due to overdiagnosis, which precipitates unnecessary anxiety, biopsies, and overtreatment (eg, excess radiation, overuse of androgen deprivation therapy).19 Biopsies carry the risk of hematuria (22.6%), hematospermia (50.4%), and urinary tract infection.20 Data from SEER-Medicare showed that prostate biopsy was associated with a 2.65-fold increased risk of hospitalization within 30 days of the procedure compared to a control population.21
Continue to: Overdiagnosis leads to overtreatment...
Overdiagnosis leads to overtreatment of low-risk prostate cancer. Both traditional treatment options for prostate cancer—radical prostatectomy and radiotherapy—are associated with urinary incontinence, erectile dysfunction, and issues with bowel function.22,23
The Prostate Cancer Intervention vs Observation Trial (PIVOT),24 the Scandinavian Prostate Cancer Group Study Number 4 (SPCG-4),25 and the Prostate Testing for Cancer and Treatment (ProtecT) trial,22,23 are the major RCTs that looked at the outcomes of treatment modalities for localized prostate cancer in the modern era of PSA testing.
PIVOT compared passive observation with radical prostatectomy.24 After 20 years of follow-up on 731 patients, the researchers concluded that radical prostatectomy did not reduce all-cause or prostate cancer–related mortality (TABLE 3).24

SPCG-4 showed survival benefits for men who underwent radical prostatectomy compared with men in a watchful waiting group, but only 5% of the study cohort had cancer detected by PSA screening (TABLE 4).25 The rest had either palpable tumors or symptoms of a tumor.

ProtecT, which followed patients with localized prostate cancer for more than 10 years,compared the outcomes and adverse effects of active surveillance, radical prostatectomy, and radiotherapy.23 Prostate cancer–specific mortality was low irrespective of the treatment,23 and there was no significant difference in all-cause mortality or prostate cancer–specific mortality between the 3 treatment groups.23 The active surveillance group had considerably fewer adverse events.22,23 The incidence rates of erectile dysfunction and urinary incontinence at the 1- and 6-year follow-up marks are outlined in TABLE 5.22

Continue to: The purpose of active monitoring...
The purpose of active monitoring is to minimize overtreatment by avoiding immediate radical intervention. Radical treatments with curative intent can be undertaken at any point while patients are being actively monitored. It is important to note that the active monitoring that took place in ProtecT23 was very different from the passive surveillance of PIVOT24 and SPCG-4.25 In ProtecT, once an elevated serum PSA level was noted, PSA levels were monitored every 3 months in the first year and every 6 to 12 months thereafter.23 Triggers to reassess patients and consider a change in clinical management were based largely on changes in PSA levels. Participants with an increase of at least 50% in PSA level during the previous 12 months were offered either continued monitoring or treatment after further testing.
Making individualized decisions about prostate cancer screening
Traditionally, the goal of cancer screening has been to maximize the number of people screened. Generally, the information provided to patients about cancer screening emphasizes the benefits and minimizes the harms. Recently, however, there has been a shift in communication about cancer screening with the emphasis now being placed on informed decision-making and encouraging patients to make individual decisions about screening participation.26
The treatment option of active surveillance, with its lower incidence of adverse outcomes, is an important reason for patients to make individualized decisions about prostate cancer screening.
Another reason relates to 5-alpha-reductase inhibitors. Although their role in the management of prostate cancer is currently not well defined, a reduction of almost 25% in the risk of prostate cancer and improvement in the performance of PSA has been reported.27
And yet another reason is that there are alternate strategies to manage the majority of patients who have been diagnosed with low-risk disease through transrectal ultrasound biopsy. The ERSPC study mentions multiparametric magnetic resonance imaging combined with targeted biopsy to identify high-grade disease.28,29 Genetic and epigenetic assays of the biopsied tissue can help grade disease based on aggressiveness.30 Transperineal mapping biopsy using a mapping software program can identify specific disease sites within the prostate gland, so that patients can be offered the option of targeted therapy.30
Continue to: Applying shared decision-making to prostate cancer screening
Applying shared decision-making to prostate cancer screening
Balancing errors of omission with errors of commission is challenging. Shared decision-making (SDM) is an approach whereby clinicians and patients share the best available evidence when faced with the task of medical decision-making and in which patients are supported while they consider their options and achieve their preferences.31 SDM is well supported by evidence from a number of RCTs and results in increased knowledge, involvement, and confidence on the part of patients.32 An individualized approach using the schematic diagram (FIGURE 13,18) may be helpful.

Barriers to SDM success. Many factors can interfere with the success of SDM including limited or poor communication; lack of time during busy office visits; and patients’ cultural, informational, and/or emotional needs. To improve patient-centered communication, we can: (1) make information understandable and available to patients and families; (2) prioritize training in communication; (3) use decision aid tools to facilitate communication; and (4) work to improve the payment model to incentivize patient-centered communication. Tools that facilitate SDM include videotapes, patient group discussions, brief scripts read to patients, and informational pamphlets. One such tool is the American Society for Clinical Oncology’s decision aid tool for PSA testing.33
Limited knowledge among patients. Decisions regarding treatment among men diagnosed with localized prostate cancer can be difficult because there are several treatment options with similar prognoses, but there are differences in adverse effects. One population-based cohort study of men with newly diagnosed localized prostate cancer found that most men had significant knowledge deficits regarding the survival benefits of the 2 major treatment options—surgery and radiation.34 In a large population-based study, 38% of men with localized prostate cancer reported receiving help from their primary care providers in the decision-making process for treatment.35
Learning to employ SDM. Elwyn et al proposed a 3-step model to incorporate SDM into clinical practice.31 They described key steps that include: choice talk (making sure patients are informed about the reasonable options), option talk (providing more detailed information about the options), and decision talk (supporting the work of patients considering their preferences and deciding what is best). Properly employing these methods requires training using simulations.31
The bottom line
Although current guidelines regarding PSA screening differ by organization, generally speaking PSA screening should be offered only to men with a life expectancy > 10 years. The PSA test has low sensitivity and specificity and lacks a clear cut-off value that warrants prostate biopsy. Men who choose to have PSA testing increase their chances of detecting prostate cancer, but most prostate cancers are slow growing and do not cause death. The decision to undergo PSA screening should be made by both the provider and the patient, after a discussion of the limited benefits and associated harms. The interval of follow-up screening may vary from 2 to 4 years depending on patient age, level of PSA, and whether a patient is taking medications such as 5-alpha-reductase inhibitors.
CORRESPONDENCE
Jaividhya Dasarathy, MD, FAAFP, 2500 Metro Health Medical Drive, Cleveland, Ohio 44109; [email protected].
Prostate cancer is the most frequently diagnosed cancer in men and the third leading cause of cancer death in men worldwide.1 An estimated 174,650 new cases are diagnosed each year in the United States; 31,620 American men die annually from the disease.2 Although prostate cancer can be a serious disease, many men do not die from it. In fact, 2.9 million men who were diagnosed with prostate cancer at some point are alive today.3
Risk factors. Prostate cancer develops mainly in men ages ≥ 65 years and rarely occurs before age 40. In addition to age, family history and African American ethnicity are the major nonmodifiable risk factors for prostate cancer.4 From the 1970s to the most recent statistical analysis of the National Cancer Institute Surveillance, Epidemiology, and End Results (SEER) program, African American men have continued to have significantly higher incidence of, and mortality rates from, prostate cancer than their European American counterparts. African American men are also more likely than men of European ancestry to have aggressive prostate cancers.5 Other risk factors include geographic location (higher risk in Northern Europe, North America, and Australia; lower risk in Asia, Africa, and South and Central America), mutations in the BRCA2 gene, and hereditary non-polyposis colon cancer syndrome.4
Prostate-specific antigen (PSA) was first used as a screening tool for prostate cancer in 1991.6 Prostate cancer incidence, especially organ-confined disease, has dramatically increased since then.7 PSA testing has a low sensitivity and specificity for the detection of prostate cancer, and there is no clear threshold at which biopsy can or should be offered. The most commonly used cutoff value of 4 ng/mL has a false-positive rate of about 70%.8
Benign prostatic conditions such as hypertrophy and infection can elevate PSA levels. In addition, the PSA test does not distinguish between aggressive and slow-growing cancers, and about 15% of patients with prostate cancer have a normal PSA level.9
A word about the digital rectal exam. While PSA testing has been the mainstay of prostate cancer screening, a few studies have included digital rectal exam (DRE) in their protocols. Data from the Prostate, Lung, Colorectal, and Ovarian (PLCO) Cancer Screening Trial showed that DRE captured an additional 2% of men with prostate cancer in the setting of a normal PSA test result.10 In the Rotterdam arm of the European Randomized Study of Screening for Prostate Cancer (ERSPC) trial, the overall detection rate for prostate cancer was found to be better when DRE was combined with PSA and prostate biopsy than when DRE was used alone (4.5% vs 2.5%).11 Nevertheless, generally speaking, DRE can be omitted in the era of PSA screening.
Screening guidelines vary
Recommendations for prostate cancer screening vary by organization and are summarized in TABLE 1.9,12-14 In 2012, the US Preventive Services Task Force (USPSTF) recommended against PSA-based screening for prostate cancer (Category D).15 In 2018, USPSTF provided an update with a new recommendation that clinicians inform men ages 55 to 69 years about the potential benefits and harms of PSA-based screening (Category C).14 The USPSTF continues to recommend against PSA-based screening for men ages ≥ 70 years (Category D).14

Does PSA-based screening improve patient-centered outcomes?
Several randomized controlled trials (RCTs) such as the Quebec Prospective Randomized Controlled Trial,16 the Norrköping Sweden Study,17 ERSPC,11 and PLCO10 have been conducted to assess the benefits of PSA testing. PLCO and ERSPC have contributed significantly to our understanding of prostate cancer screening even though their 13-year follow-up results are conflicting (TABLE 2).10,11,18

Continue to: In the ERSPC 13-year follow-up publication...
In the ERSPC 13-year follow-up publication, the authors concluded that a substantial reduction in prostate cancer mortality is attributable to testing with PSA.18 Despite limitations in the study design (eg, France entered after 2 years, screening intervals varied between 2 and 4 years, biopsy indications varied, and screening was discontinued at different times), PSA screening detected more prostate cancer than was detected in the control arm (10.2% vs 6.8%).
In the initial 11 years of follow-up, the study group experienced a 21% reduction in prostate cancer mortality, even though the absolute decrease ranged from only 0.6% (545 per 89,352) to 0.5% (355 per 72,891). The updated absolute risk reduction of death from prostate cancer at 13 years of follow-up showed a larger benefit: 0.11 per 1000 person-years or 1.28 per 1000 men randomized, which is equivalent to 1 prostate cancer death averted per 781 (95% confidence interval [CI], 490-1929) men invited for screening, or 1 per 27 (17-66) additional prostate cancers detected.
The PLCO trial did not show any significant difference in prostate cancer detection (11.1% screened vs 9.9% control), and there was no improvement in prostate cancer mortality (3.7 vs 3.4 death per 10,000 person-years).10 However, the PLCO trial suffered from issues of contamination, which may have influenced the overall results. About 52% of men in the control (usual care) group received a PSA test at some point during the study. And more than two-thirds of the men who had a prostate biopsy because of a positive PSA test did not have prostate cancer.
Community standards for the PSA threshold for biopsy were applied in various centers (> 4 ng/ml in general) in PLCO, whereas in ERSPC, a cut-off PSA value ≥ 3 ng/mL was used for biopsy. Because of the lower PSA threshold, ERSPC may have identified cancers that would have had good outcomes without any intervention.
The harms of PSA screening
While it is unclear whether PSA screening results in any improvement in patient-centered outcomes, it does lead to downstream intervention due to overdiagnosis, which precipitates unnecessary anxiety, biopsies, and overtreatment (eg, excess radiation, overuse of androgen deprivation therapy).19 Biopsies carry the risk of hematuria (22.6%), hematospermia (50.4%), and urinary tract infection.20 Data from SEER-Medicare showed that prostate biopsy was associated with a 2.65-fold increased risk of hospitalization within 30 days of the procedure compared to a control population.21
Continue to: Overdiagnosis leads to overtreatment...
Overdiagnosis leads to overtreatment of low-risk prostate cancer. Both traditional treatment options for prostate cancer—radical prostatectomy and radiotherapy—are associated with urinary incontinence, erectile dysfunction, and issues with bowel function.22,23
The Prostate Cancer Intervention vs Observation Trial (PIVOT),24 the Scandinavian Prostate Cancer Group Study Number 4 (SPCG-4),25 and the Prostate Testing for Cancer and Treatment (ProtecT) trial,22,23 are the major RCTs that looked at the outcomes of treatment modalities for localized prostate cancer in the modern era of PSA testing.
PIVOT compared passive observation with radical prostatectomy.24 After 20 years of follow-up on 731 patients, the researchers concluded that radical prostatectomy did not reduce all-cause or prostate cancer–related mortality (TABLE 3).24

SPCG-4 showed survival benefits for men who underwent radical prostatectomy compared with men in a watchful waiting group, but only 5% of the study cohort had cancer detected by PSA screening (TABLE 4).25 The rest had either palpable tumors or symptoms of a tumor.

ProtecT, which followed patients with localized prostate cancer for more than 10 years,compared the outcomes and adverse effects of active surveillance, radical prostatectomy, and radiotherapy.23 Prostate cancer–specific mortality was low irrespective of the treatment,23 and there was no significant difference in all-cause mortality or prostate cancer–specific mortality between the 3 treatment groups.23 The active surveillance group had considerably fewer adverse events.22,23 The incidence rates of erectile dysfunction and urinary incontinence at the 1- and 6-year follow-up marks are outlined in TABLE 5.22

Continue to: The purpose of active monitoring...
The purpose of active monitoring is to minimize overtreatment by avoiding immediate radical intervention. Radical treatments with curative intent can be undertaken at any point while patients are being actively monitored. It is important to note that the active monitoring that took place in ProtecT23 was very different from the passive surveillance of PIVOT24 and SPCG-4.25 In ProtecT, once an elevated serum PSA level was noted, PSA levels were monitored every 3 months in the first year and every 6 to 12 months thereafter.23 Triggers to reassess patients and consider a change in clinical management were based largely on changes in PSA levels. Participants with an increase of at least 50% in PSA level during the previous 12 months were offered either continued monitoring or treatment after further testing.
Making individualized decisions about prostate cancer screening
Traditionally, the goal of cancer screening has been to maximize the number of people screened. Generally, the information provided to patients about cancer screening emphasizes the benefits and minimizes the harms. Recently, however, there has been a shift in communication about cancer screening with the emphasis now being placed on informed decision-making and encouraging patients to make individual decisions about screening participation.26
The treatment option of active surveillance, with its lower incidence of adverse outcomes, is an important reason for patients to make individualized decisions about prostate cancer screening.
Another reason relates to 5-alpha-reductase inhibitors. Although their role in the management of prostate cancer is currently not well defined, a reduction of almost 25% in the risk of prostate cancer and improvement in the performance of PSA has been reported.27
And yet another reason is that there are alternate strategies to manage the majority of patients who have been diagnosed with low-risk disease through transrectal ultrasound biopsy. The ERSPC study mentions multiparametric magnetic resonance imaging combined with targeted biopsy to identify high-grade disease.28,29 Genetic and epigenetic assays of the biopsied tissue can help grade disease based on aggressiveness.30 Transperineal mapping biopsy using a mapping software program can identify specific disease sites within the prostate gland, so that patients can be offered the option of targeted therapy.30
Continue to: Applying shared decision-making to prostate cancer screening
Applying shared decision-making to prostate cancer screening
Balancing errors of omission with errors of commission is challenging. Shared decision-making (SDM) is an approach whereby clinicians and patients share the best available evidence when faced with the task of medical decision-making and in which patients are supported while they consider their options and achieve their preferences.31 SDM is well supported by evidence from a number of RCTs and results in increased knowledge, involvement, and confidence on the part of patients.32 An individualized approach using the schematic diagram (FIGURE 13,18) may be helpful.

Barriers to SDM success. Many factors can interfere with the success of SDM including limited or poor communication; lack of time during busy office visits; and patients’ cultural, informational, and/or emotional needs. To improve patient-centered communication, we can: (1) make information understandable and available to patients and families; (2) prioritize training in communication; (3) use decision aid tools to facilitate communication; and (4) work to improve the payment model to incentivize patient-centered communication. Tools that facilitate SDM include videotapes, patient group discussions, brief scripts read to patients, and informational pamphlets. One such tool is the American Society for Clinical Oncology’s decision aid tool for PSA testing.33
Limited knowledge among patients. Decisions regarding treatment among men diagnosed with localized prostate cancer can be difficult because there are several treatment options with similar prognoses, but there are differences in adverse effects. One population-based cohort study of men with newly diagnosed localized prostate cancer found that most men had significant knowledge deficits regarding the survival benefits of the 2 major treatment options—surgery and radiation.34 In a large population-based study, 38% of men with localized prostate cancer reported receiving help from their primary care providers in the decision-making process for treatment.35
Learning to employ SDM. Elwyn et al proposed a 3-step model to incorporate SDM into clinical practice.31 They described key steps that include: choice talk (making sure patients are informed about the reasonable options), option talk (providing more detailed information about the options), and decision talk (supporting the work of patients considering their preferences and deciding what is best). Properly employing these methods requires training using simulations.31
The bottom line
Although current guidelines regarding PSA screening differ by organization, generally speaking PSA screening should be offered only to men with a life expectancy > 10 years. The PSA test has low sensitivity and specificity and lacks a clear cut-off value that warrants prostate biopsy. Men who choose to have PSA testing increase their chances of detecting prostate cancer, but most prostate cancers are slow growing and do not cause death. The decision to undergo PSA screening should be made by both the provider and the patient, after a discussion of the limited benefits and associated harms. The interval of follow-up screening may vary from 2 to 4 years depending on patient age, level of PSA, and whether a patient is taking medications such as 5-alpha-reductase inhibitors.
CORRESPONDENCE
Jaividhya Dasarathy, MD, FAAFP, 2500 Metro Health Medical Drive, Cleveland, Ohio 44109; [email protected].
1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016;66:7-30.
2. National Cancer Institute Surveillance, Epidemiology, and End Results Program. Cancer Stat Facts: Prostate Cancer. https://seer.cancer.gov/statfacts/html/prost.html. Accessed January 16, 2020.
3. American Cancer Society. Key statistics for prostate cancer. Last revised August 1, 2019. www.cancer.org/cancer/prostate-cancer/about/key-statistics.html. Accessed January 16, 2020.
4. Brawley OW. Trends in prostate cancer in the United States. J Natl Cancer Inst Monogr. 2012;2012:152-156.
5. Powell IJ. Epidemiology and pathophysiology of prostate cancer in African-American men. J Urol. 2007;177:444-449.
6. Catalona WJ, Smith DS, Ratliff TL, et al. Measurement of prostate-specific antigen in serum as a screening test for prostate cancer. N Engl J Med. 1991;324:1156-1161.
7. Jacobsen SJ, Katusic SK, Bergstraih EJ. Incidence of prostate cancer diagnosis in the eras before and after serum prostate-specific antigen testing. JAMA. 1995;274:1445-1449.
8. Mistry K, Cable G. Meta-analysis of prostate-specific antigen and digital rectal examination as screening tests for prostate carcinoma. J Am Board Fam Pract. 2003;16:95-101.
9. Qaseem A, Barry MJ, Denberg TD, et al. Screening for prostate cancer: a guidance statement from the Clinical Guidelines Committee of the American College of Physicians. Ann Int Med. 2013;158:761-769.
10. Andriole GL, Crawford ED, Grubb RL 3rd, et al. Prostate cancer screening in the randomized Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial: mortality results after 13 years of follow-up. J Natl Cancer Inst. 2012;104:125-132.
11. Schröder FH, Hugosson J, Roobol MJ, et al. Screening and prostate-cancer mortality in a randomized European study. N Engl J Med. 2009;360:1320-1328.
12. American Cancer Society. American Cancer Society recommendations for prostate cancer early detection. Last revised August 1, 2019. www.cancer.org/cancer/prostate-cancer/detection-diagnosis-staging/acs-recommendations.html. Accessed January 16, 2020.
13. American Urologic Association. Early detection of prostate cancer (2018). Reviewed 2018. https://www.auanet.org/guidelines/prostate-cancer-early-detection-guideline. Accessed January 16, 2020.
14. US Preventive Services Task Force. Screening for Prostate Cancer: US Preventive Services Task Force Recommendation Statement. JAMA. 2018;319:1901-1913.
15 Moyer VA. Screening for prostate cancer: US Preventive Services Task Force recommendation statement. Ann Int Med. 2012;157:120-134.
16. Labrie F, Candas B, Dupont A, et al. Screening decreases prostate cancer death: first analysis of the 1988 Quebec prospective randomized controlled trial. Prostate. 1999;38:83-91.
17. Sandblom G, Varenhorst E, Rosell J, et al. Randomised prostate cancer screening trial: 20-year follow-up. BMJ. 2011;342:d1539.
18. Schröder FH, Hugosson J, Roobol MJ, et al. Screening and prostate cancer mortality: results of the European Randomized Study of Screening for Prostate Cancer (ERSPC) at 13 years of follow-up. Lancet. 2014;384:2027-2035.
19. McNaughton-Collins M, Fowler FJ Jr, Caubet JF, et al. Psychological effects of a suspicious prostate cancer screening test followed by a benign biopsy result. Am J Med. 2004;117:719-725.
20 Raaijmakers R, Kirkels WJ, Roobol MJ, et al. Complication rates and risk factors of 5802 transrectal ultrasound-guided sextant biopsies of the prostate within a population-based screening program. Urology. 2002;60:826-830.
21. Loeb S, Carter HB, Berndt SI, et al. Complications after prostate biopsy: data from SEER-Medicare. J Urol. 2011;186:1830-1834.
22. Donovan J, Hamdy F, Lane J, et al. Patient-reported outcomes after monitoring, surgery, or radiotherapy for prostate cancer. N Engl J Med. 2016;375:1425-1437.
23. Hamdy FC, Donovan JL, Lane JA, et al. 10-year outcomes after monitoring, surgery, or radiotherapy for localized prostate cancer. N Engl J Med. 2016;375:1415-1424.
24. Wilt TJ, Jones KM, Barry MJ, et al. Follow-up of prostatectomy versus observation for early prostate cancer. N Engl J Med. 2017;377:132-142.
25. Bill-Axelson A, Holmberg L, Garmo H, et al. Radical prostatectomy or watchful waiting in early prostate cancer. N Engl J Med. 2018;379:2319-2329.
26. Hersch JK, Nickel BL, Ghanouni A, et al. Improving communication about cancer screening: moving towards informed decision making. Public Health Res Pract. 2017;27(2).
27. Cuzick J, Thorat MA, Andriole G, et al. Prevention and early detection of prostate cancer. Lancet Oncol. 2014;15:e484-e492.
28. Pinto PA, Chung PH, Rastinehad AR, et al. Magnetic resonance imaging/ultrasound fusion guided prostate biopsy improves cancer detection following transrectal ultrasound biopsy and correlates with multiparametric magnetic resonance imaging. J Urol. 2011;186:1281-1285.
29. Kuru TH, Roethke MC, Seidenader J, et al. Critical evaluation of magnetic resonance imaging targeted, transrectal ultrasound guided transperineal fusion biopsy for detection of prostate cancer. J Urol. 2013;190:1380-1386.
30. Crawford ED, Rove KO, Barqawi AB, et al. Clinical-pathologic correlation between transperineal mapping biopsies of the prostate and three-dimensional reconstruction of prostatectomy specimens. Prostate. 2013;73:778-787.
31. Elwyn G, Frosch D, Thomson R, et al. Shared decision making: a model for clinical practice. J Gen Intern Med. 2012;27:1361-1367.
32. Stacey D, Légaré F, Lewis K, et al. Decision aids for people facing health treatment or screening decisions. Cochrane Database Syst Rev. 2017;4:CD001431.
33. ASCO. Decision aid tool: prostate cancer screening with PSA testing. https://www.asco.org/sites/new-www.asco.org/files/content-files/practice-and-guidelines/documents/2012-psa-pco-decision-aid.pdf. Accessed January 16, 2020.
34. Daum LM, Reamer EN, Ruterbusch JJ, et al. Patient knowledge and qualities of treatment decisions for localized prostate cancer. J Am Board Fam Med. 2017;30:288-297.
35. Radhakrishnan A, Grande D, Ross M, et al. When primary care providers (PCPs) help patients choose prostate cancer treatment. J Am Board Fam Med. 2017;30:298-307.
1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016;66:7-30.
2. National Cancer Institute Surveillance, Epidemiology, and End Results Program. Cancer Stat Facts: Prostate Cancer. https://seer.cancer.gov/statfacts/html/prost.html. Accessed January 16, 2020.
3. American Cancer Society. Key statistics for prostate cancer. Last revised August 1, 2019. www.cancer.org/cancer/prostate-cancer/about/key-statistics.html. Accessed January 16, 2020.
4. Brawley OW. Trends in prostate cancer in the United States. J Natl Cancer Inst Monogr. 2012;2012:152-156.
5. Powell IJ. Epidemiology and pathophysiology of prostate cancer in African-American men. J Urol. 2007;177:444-449.
6. Catalona WJ, Smith DS, Ratliff TL, et al. Measurement of prostate-specific antigen in serum as a screening test for prostate cancer. N Engl J Med. 1991;324:1156-1161.
7. Jacobsen SJ, Katusic SK, Bergstraih EJ. Incidence of prostate cancer diagnosis in the eras before and after serum prostate-specific antigen testing. JAMA. 1995;274:1445-1449.
8. Mistry K, Cable G. Meta-analysis of prostate-specific antigen and digital rectal examination as screening tests for prostate carcinoma. J Am Board Fam Pract. 2003;16:95-101.
9. Qaseem A, Barry MJ, Denberg TD, et al. Screening for prostate cancer: a guidance statement from the Clinical Guidelines Committee of the American College of Physicians. Ann Int Med. 2013;158:761-769.
10. Andriole GL, Crawford ED, Grubb RL 3rd, et al. Prostate cancer screening in the randomized Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial: mortality results after 13 years of follow-up. J Natl Cancer Inst. 2012;104:125-132.
11. Schröder FH, Hugosson J, Roobol MJ, et al. Screening and prostate-cancer mortality in a randomized European study. N Engl J Med. 2009;360:1320-1328.
12. American Cancer Society. American Cancer Society recommendations for prostate cancer early detection. Last revised August 1, 2019. www.cancer.org/cancer/prostate-cancer/detection-diagnosis-staging/acs-recommendations.html. Accessed January 16, 2020.
13. American Urologic Association. Early detection of prostate cancer (2018). Reviewed 2018. https://www.auanet.org/guidelines/prostate-cancer-early-detection-guideline. Accessed January 16, 2020.
14. US Preventive Services Task Force. Screening for Prostate Cancer: US Preventive Services Task Force Recommendation Statement. JAMA. 2018;319:1901-1913.
15 Moyer VA. Screening for prostate cancer: US Preventive Services Task Force recommendation statement. Ann Int Med. 2012;157:120-134.
16. Labrie F, Candas B, Dupont A, et al. Screening decreases prostate cancer death: first analysis of the 1988 Quebec prospective randomized controlled trial. Prostate. 1999;38:83-91.
17. Sandblom G, Varenhorst E, Rosell J, et al. Randomised prostate cancer screening trial: 20-year follow-up. BMJ. 2011;342:d1539.
18. Schröder FH, Hugosson J, Roobol MJ, et al. Screening and prostate cancer mortality: results of the European Randomized Study of Screening for Prostate Cancer (ERSPC) at 13 years of follow-up. Lancet. 2014;384:2027-2035.
19. McNaughton-Collins M, Fowler FJ Jr, Caubet JF, et al. Psychological effects of a suspicious prostate cancer screening test followed by a benign biopsy result. Am J Med. 2004;117:719-725.
20 Raaijmakers R, Kirkels WJ, Roobol MJ, et al. Complication rates and risk factors of 5802 transrectal ultrasound-guided sextant biopsies of the prostate within a population-based screening program. Urology. 2002;60:826-830.
21. Loeb S, Carter HB, Berndt SI, et al. Complications after prostate biopsy: data from SEER-Medicare. J Urol. 2011;186:1830-1834.
22. Donovan J, Hamdy F, Lane J, et al. Patient-reported outcomes after monitoring, surgery, or radiotherapy for prostate cancer. N Engl J Med. 2016;375:1425-1437.
23. Hamdy FC, Donovan JL, Lane JA, et al. 10-year outcomes after monitoring, surgery, or radiotherapy for localized prostate cancer. N Engl J Med. 2016;375:1415-1424.
24. Wilt TJ, Jones KM, Barry MJ, et al. Follow-up of prostatectomy versus observation for early prostate cancer. N Engl J Med. 2017;377:132-142.
25. Bill-Axelson A, Holmberg L, Garmo H, et al. Radical prostatectomy or watchful waiting in early prostate cancer. N Engl J Med. 2018;379:2319-2329.
26. Hersch JK, Nickel BL, Ghanouni A, et al. Improving communication about cancer screening: moving towards informed decision making. Public Health Res Pract. 2017;27(2).
27. Cuzick J, Thorat MA, Andriole G, et al. Prevention and early detection of prostate cancer. Lancet Oncol. 2014;15:e484-e492.
28. Pinto PA, Chung PH, Rastinehad AR, et al. Magnetic resonance imaging/ultrasound fusion guided prostate biopsy improves cancer detection following transrectal ultrasound biopsy and correlates with multiparametric magnetic resonance imaging. J Urol. 2011;186:1281-1285.
29. Kuru TH, Roethke MC, Seidenader J, et al. Critical evaluation of magnetic resonance imaging targeted, transrectal ultrasound guided transperineal fusion biopsy for detection of prostate cancer. J Urol. 2013;190:1380-1386.
30. Crawford ED, Rove KO, Barqawi AB, et al. Clinical-pathologic correlation between transperineal mapping biopsies of the prostate and three-dimensional reconstruction of prostatectomy specimens. Prostate. 2013;73:778-787.
31. Elwyn G, Frosch D, Thomson R, et al. Shared decision making: a model for clinical practice. J Gen Intern Med. 2012;27:1361-1367.
32. Stacey D, Légaré F, Lewis K, et al. Decision aids for people facing health treatment or screening decisions. Cochrane Database Syst Rev. 2017;4:CD001431.
33. ASCO. Decision aid tool: prostate cancer screening with PSA testing. https://www.asco.org/sites/new-www.asco.org/files/content-files/practice-and-guidelines/documents/2012-psa-pco-decision-aid.pdf. Accessed January 16, 2020.
34. Daum LM, Reamer EN, Ruterbusch JJ, et al. Patient knowledge and qualities of treatment decisions for localized prostate cancer. J Am Board Fam Med. 2017;30:288-297.
35. Radhakrishnan A, Grande D, Ross M, et al. When primary care providers (PCPs) help patients choose prostate cancer treatment. J Am Board Fam Med. 2017;30:298-307.
PRACTICE RECOMMENDATIONS
› Recommend individualized decision-making to men ages 55 to 69 years after discussing the potential benefits and risks of prostate-specific antigen (PSA)-based screening. B
› Do not use a PSA-based screening method for prostate cancer in men ages < 50 years or > 70 years or men with a life expectancy < 10 years. C
› Do not routinely recommend PSA-based screening to men with a family history of prostate cancer or to men who are African American. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Opioid deaths boost donor heart supply
SNOWMASS, COLO. – The tragic opioid epidemic has “one small bright spot”: an expanding pool of eligible donor hearts for transplantation, Akshay S. Desai, MD, said at the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
For decades, the annual volume of heart transplantations performed in the U.S. was static because of the huge mismatch between donor organ supply and demand. But heart transplant volume has increased steadily in the last few years – a result of the opioid epidemic.
Data from the U.S. Organ Procurement and Transplantation Network show that the proportion of donor hearts obtained from individuals who died from drug intoxication climbed from a mere 1.5% in 1999 to 17.6% in 2017, the most recent year for which data are available. Meanwhile, the size of the heart transplant waiting list, which rose year after year in 2009-2015, has since declined (N Engl J Med. 2019 Feb 7;380[6]:597-9).
“What’s amazing is that, even though these patients might have historically been considered high risk in general, the organs recovered from these patients – and particularly the hearts – don’t seem to be any worse in terms of allograft survival than the organs recovered from patients who died from other causes, which are the traditional sources, like blunt head trauma, gunshot wounds, or stroke, that lead to brain death. In general, these organs are useful and do quite well,” according to Dr. Desai, medical director of the cardiomyopathy and heart failure program at Brigham and Women’s Hospital, Boston.
He highlighted several other recent developments in the field of cardiac transplantation that promise to further expand the donor heart pool, including acceptance of hepatitis C–infected donors and organ donation after circulatory rather than brain death. Dr. Desai also drew attention to the unintended perverse consequences of a recent redesign of the U.S. donor heart allocation system and discussed the impressive improvement in clinical outcomes with mechanical circulatory support. He noted that, while relatively few cardiologists practice in the highly specialized centers where heart transplants take place, virtually all cardiologists are affected by advances in heart transplantation since hundreds of thousands of the estimated 7 million Americans with heart failure have advanced disease.
Heart transplantation, he emphasized, is becoming increasingly complex. Recipients are on average older, sicker, and have more comorbidities than in times past. As a result, there is greater need for dual organ transplants: heart/lung, heart/liver, or heart/kidney. Plus, more patients come to transplantation after prior cardiac surgery for implantation of a ventricular assist device, so sensitization to blood products is a growing issue. And, of course, the pool of transplant candidates has expanded.
“We’re now forced to take patients previously considered to have contraindications to transplant; for example, diabetes was a contraindication to transplant in the early years, but now it’s the rule in 35%-40% of our patients who present with advanced heart failure,” the cardiologist noted.
Transplants from HCV-infected donors to uninfected recipients
Hearts and lungs from donors with hepatitis C viremia were traditionally deemed unsuitable for transplant. That’s all changed in the current era of highly effective direct-acting antiviral agents for the treatment of HCV infection.
In the DONATE HCV trial, Dr. Desai’s colleagues at Brigham and Women’s Hospital showed that giving HCV-uninfected recipients of hearts or lungs from HCV-viremic donors a shortened 4-week course of treatment with sofosbuvir-velpatasvir (Epclusa) beginning within a few hours after transplantation uniformly blocked viral replication. Six months after transplantation, none of the study participants had a detectable HCV viral load, and all had excellent graft function (N Engl J Med. 2019 Apr 25;380[17]:1606-17).
“This is effective prevention of HCV infection by aggressive upfront therapy,” Dr. Desai explained. “We can now take organs from HCV-viremic patients and use them in solid organ transplantation. This has led to a skyrocketing increase in donors with HCV infection, and those donations have helped us clear the waiting list.”
Donation after circulatory death
Australian transplant physicians have pioneered the use of donor hearts obtained after circulatory death in individuals with devastating neurologic injury who didn’t quite meet the criteria for brain death, which is the traditional prerequisite. In the new scenario, withdrawal of life-supporting therapy is followed by circulatory death, then the donor heart is procured and preserved via extracorporeal perfusion until transplantation.
The Australians report excellent outcomes, with rates of overall survival and rejection episodes similar to outcomes from brain-dead donors (J Am Coll Cardiol. 2019 Apr 2;73[12]:1447-59). The first U.S. heart transplant involving donation after circulatory death took place at Duke University in Durham, North Carolina. A multicenter U.S. clinical trial of this practice is underway.
If the results are positive and the practice of donation after circulatory death becomes widely implemented, the U.S. heart donor pool could increase by 30%.
Recent overhaul of donor heart allocation system may have backfired
The U.S. donor heart allocation system was redesigned in the fall of 2018 in an effort to reduce waiting times. One of the biggest changes involved breaking down the category with the highest urgency status into three new subcategories based upon sickness. Now, the highest-urgency category is for patients in cardiogenic shock who are supported by extracorporeal membrane oxygenation (ECMO) or other temporary mechanical circulatory support devices.
But an analysis of United Network for Organ Sharing (UNOS) data suggests this change has unintended adverse consequences for clinical outcomes.
Indeed, the investigators reported that the use of ECMO support is fourfold greater in the new system, the use of durable left ventricular assist devices (LVADs) as a bridge to transplant is down, and outcomes are worse. The 180-day rate of freedom from death or retransplantation was 77.9%, down significantly from 93.4% in the former system. In a multivariate analysis, patients transplanted in the new system had an adjusted 2.1-fold increased risk of death or retransplantation (J Heart Lung Transplant. 2020 Jan;39[1]:1-4).
“When you create a new listing system, you create new incentives, and people start to manage patients differently,” Dr. Desai observed. “Increasingly now, the path direct to transplant is through temporary mechanical circulatory support rather than durable mechanical circulatory support. Is that a good idea? We don’t know, but if you look at the best data, those on ECMO or percutaneous VADs have the worst outcomes. So the question of whether we should take the sickest of sick patients directly to transplant as a standard strategy has come under scrutiny.”
Improved durable LVAD technology brings impressive clinical outcomes
Results of the landmark MOMENTUM 3 randomized trial showed that 2-year clinical outcomes with the magnetically levitated centrifugal-flow HeartMate 3 LVAD now rival those of percutaneous mitral valve repair using the MitraClip device. Two-year all-cause mortality in the LVAD recipients was 22% versus 29.1% with the MitraClip in the COAPT trial and 34.9% in the MITRA-FR trial. The HeartMate 3 reduces the hemocompatibility issues that plagued earlier-generation durable LVADs, with resultant lower rates of pump thrombosis, stroke, and GI bleeding. Indeed, the outcomes in MOMENTUM 3 were so good – and so similar – with the HeartMate 3, regardless of whether the intended treatment goal was as a bridge to transplant or as lifelong destination therapy, that the investigators have recently proposed doing away with those distinctions.
“It is possible that use of arbitrary categorizations based on current or future transplant eligibility should be clinically abandoned in favor of a single preimplant strategy: to extend the survival and improve the quality of life of patients with medically refractory heart failure,” according to the investigators (JAMA Cardiol. 2020 Jan 15. doi: 10.1001/jamacardio.2019.5323).
The next step forward in LVAD technology is already on the horizon: a fully implantable device that eliminates the transcutaneous drive-line for the power supply, which is prone to infection and diminishes overall quality of life. This investigational device utilizes wireless coplanar energy transfer, with a coil ring placed around the lung and fixed to the chest wall. The implanted battery provides more than 6 hours of power without a recharge (J Heart Lung Transplant. 2019 Apr;38[4]:339-43).
“The first LVAD patient has gone swimming in Kazakhstan,” according to Dr. Desai.
Myocardial recovery in LVAD recipients remains elusive
The initial hope for LVADs was that they would not only be able to serve as a bridge to transplantation or as lifetime therapy, but that the prolonged unloading of the ventricle would enable potent medical therapy to rescue myocardial function so that the device could eventually be explanted. That does happen, but only rarely. In a large registry study, myocardial recovery occurred in only about 1% of patients on mechanical circulatory support. Attempts to enhance the process by add-on stem cell therapy have thus far been ineffective.
“For the moment, recovery is still a hope, not a reality,” the cardiologist said.
He reported serving as a consultant to more than a dozen pharmaceutical or medical device companies and receiving research grants from Alnylam, AstraZeneca, Bayer Healthcare, MyoKardia, and Novartis.
SNOWMASS, COLO. – The tragic opioid epidemic has “one small bright spot”: an expanding pool of eligible donor hearts for transplantation, Akshay S. Desai, MD, said at the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
For decades, the annual volume of heart transplantations performed in the U.S. was static because of the huge mismatch between donor organ supply and demand. But heart transplant volume has increased steadily in the last few years – a result of the opioid epidemic.
Data from the U.S. Organ Procurement and Transplantation Network show that the proportion of donor hearts obtained from individuals who died from drug intoxication climbed from a mere 1.5% in 1999 to 17.6% in 2017, the most recent year for which data are available. Meanwhile, the size of the heart transplant waiting list, which rose year after year in 2009-2015, has since declined (N Engl J Med. 2019 Feb 7;380[6]:597-9).
“What’s amazing is that, even though these patients might have historically been considered high risk in general, the organs recovered from these patients – and particularly the hearts – don’t seem to be any worse in terms of allograft survival than the organs recovered from patients who died from other causes, which are the traditional sources, like blunt head trauma, gunshot wounds, or stroke, that lead to brain death. In general, these organs are useful and do quite well,” according to Dr. Desai, medical director of the cardiomyopathy and heart failure program at Brigham and Women’s Hospital, Boston.
He highlighted several other recent developments in the field of cardiac transplantation that promise to further expand the donor heart pool, including acceptance of hepatitis C–infected donors and organ donation after circulatory rather than brain death. Dr. Desai also drew attention to the unintended perverse consequences of a recent redesign of the U.S. donor heart allocation system and discussed the impressive improvement in clinical outcomes with mechanical circulatory support. He noted that, while relatively few cardiologists practice in the highly specialized centers where heart transplants take place, virtually all cardiologists are affected by advances in heart transplantation since hundreds of thousands of the estimated 7 million Americans with heart failure have advanced disease.
Heart transplantation, he emphasized, is becoming increasingly complex. Recipients are on average older, sicker, and have more comorbidities than in times past. As a result, there is greater need for dual organ transplants: heart/lung, heart/liver, or heart/kidney. Plus, more patients come to transplantation after prior cardiac surgery for implantation of a ventricular assist device, so sensitization to blood products is a growing issue. And, of course, the pool of transplant candidates has expanded.
“We’re now forced to take patients previously considered to have contraindications to transplant; for example, diabetes was a contraindication to transplant in the early years, but now it’s the rule in 35%-40% of our patients who present with advanced heart failure,” the cardiologist noted.
Transplants from HCV-infected donors to uninfected recipients
Hearts and lungs from donors with hepatitis C viremia were traditionally deemed unsuitable for transplant. That’s all changed in the current era of highly effective direct-acting antiviral agents for the treatment of HCV infection.
In the DONATE HCV trial, Dr. Desai’s colleagues at Brigham and Women’s Hospital showed that giving HCV-uninfected recipients of hearts or lungs from HCV-viremic donors a shortened 4-week course of treatment with sofosbuvir-velpatasvir (Epclusa) beginning within a few hours after transplantation uniformly blocked viral replication. Six months after transplantation, none of the study participants had a detectable HCV viral load, and all had excellent graft function (N Engl J Med. 2019 Apr 25;380[17]:1606-17).
“This is effective prevention of HCV infection by aggressive upfront therapy,” Dr. Desai explained. “We can now take organs from HCV-viremic patients and use them in solid organ transplantation. This has led to a skyrocketing increase in donors with HCV infection, and those donations have helped us clear the waiting list.”
Donation after circulatory death
Australian transplant physicians have pioneered the use of donor hearts obtained after circulatory death in individuals with devastating neurologic injury who didn’t quite meet the criteria for brain death, which is the traditional prerequisite. In the new scenario, withdrawal of life-supporting therapy is followed by circulatory death, then the donor heart is procured and preserved via extracorporeal perfusion until transplantation.
The Australians report excellent outcomes, with rates of overall survival and rejection episodes similar to outcomes from brain-dead donors (J Am Coll Cardiol. 2019 Apr 2;73[12]:1447-59). The first U.S. heart transplant involving donation after circulatory death took place at Duke University in Durham, North Carolina. A multicenter U.S. clinical trial of this practice is underway.
If the results are positive and the practice of donation after circulatory death becomes widely implemented, the U.S. heart donor pool could increase by 30%.
Recent overhaul of donor heart allocation system may have backfired
The U.S. donor heart allocation system was redesigned in the fall of 2018 in an effort to reduce waiting times. One of the biggest changes involved breaking down the category with the highest urgency status into three new subcategories based upon sickness. Now, the highest-urgency category is for patients in cardiogenic shock who are supported by extracorporeal membrane oxygenation (ECMO) or other temporary mechanical circulatory support devices.
But an analysis of United Network for Organ Sharing (UNOS) data suggests this change has unintended adverse consequences for clinical outcomes.
Indeed, the investigators reported that the use of ECMO support is fourfold greater in the new system, the use of durable left ventricular assist devices (LVADs) as a bridge to transplant is down, and outcomes are worse. The 180-day rate of freedom from death or retransplantation was 77.9%, down significantly from 93.4% in the former system. In a multivariate analysis, patients transplanted in the new system had an adjusted 2.1-fold increased risk of death or retransplantation (J Heart Lung Transplant. 2020 Jan;39[1]:1-4).
“When you create a new listing system, you create new incentives, and people start to manage patients differently,” Dr. Desai observed. “Increasingly now, the path direct to transplant is through temporary mechanical circulatory support rather than durable mechanical circulatory support. Is that a good idea? We don’t know, but if you look at the best data, those on ECMO or percutaneous VADs have the worst outcomes. So the question of whether we should take the sickest of sick patients directly to transplant as a standard strategy has come under scrutiny.”
Improved durable LVAD technology brings impressive clinical outcomes
Results of the landmark MOMENTUM 3 randomized trial showed that 2-year clinical outcomes with the magnetically levitated centrifugal-flow HeartMate 3 LVAD now rival those of percutaneous mitral valve repair using the MitraClip device. Two-year all-cause mortality in the LVAD recipients was 22% versus 29.1% with the MitraClip in the COAPT trial and 34.9% in the MITRA-FR trial. The HeartMate 3 reduces the hemocompatibility issues that plagued earlier-generation durable LVADs, with resultant lower rates of pump thrombosis, stroke, and GI bleeding. Indeed, the outcomes in MOMENTUM 3 were so good – and so similar – with the HeartMate 3, regardless of whether the intended treatment goal was as a bridge to transplant or as lifelong destination therapy, that the investigators have recently proposed doing away with those distinctions.
“It is possible that use of arbitrary categorizations based on current or future transplant eligibility should be clinically abandoned in favor of a single preimplant strategy: to extend the survival and improve the quality of life of patients with medically refractory heart failure,” according to the investigators (JAMA Cardiol. 2020 Jan 15. doi: 10.1001/jamacardio.2019.5323).
The next step forward in LVAD technology is already on the horizon: a fully implantable device that eliminates the transcutaneous drive-line for the power supply, which is prone to infection and diminishes overall quality of life. This investigational device utilizes wireless coplanar energy transfer, with a coil ring placed around the lung and fixed to the chest wall. The implanted battery provides more than 6 hours of power without a recharge (J Heart Lung Transplant. 2019 Apr;38[4]:339-43).
“The first LVAD patient has gone swimming in Kazakhstan,” according to Dr. Desai.
Myocardial recovery in LVAD recipients remains elusive
The initial hope for LVADs was that they would not only be able to serve as a bridge to transplantation or as lifetime therapy, but that the prolonged unloading of the ventricle would enable potent medical therapy to rescue myocardial function so that the device could eventually be explanted. That does happen, but only rarely. In a large registry study, myocardial recovery occurred in only about 1% of patients on mechanical circulatory support. Attempts to enhance the process by add-on stem cell therapy have thus far been ineffective.
“For the moment, recovery is still a hope, not a reality,” the cardiologist said.
He reported serving as a consultant to more than a dozen pharmaceutical or medical device companies and receiving research grants from Alnylam, AstraZeneca, Bayer Healthcare, MyoKardia, and Novartis.
SNOWMASS, COLO. – The tragic opioid epidemic has “one small bright spot”: an expanding pool of eligible donor hearts for transplantation, Akshay S. Desai, MD, said at the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
For decades, the annual volume of heart transplantations performed in the U.S. was static because of the huge mismatch between donor organ supply and demand. But heart transplant volume has increased steadily in the last few years – a result of the opioid epidemic.
Data from the U.S. Organ Procurement and Transplantation Network show that the proportion of donor hearts obtained from individuals who died from drug intoxication climbed from a mere 1.5% in 1999 to 17.6% in 2017, the most recent year for which data are available. Meanwhile, the size of the heart transplant waiting list, which rose year after year in 2009-2015, has since declined (N Engl J Med. 2019 Feb 7;380[6]:597-9).
“What’s amazing is that, even though these patients might have historically been considered high risk in general, the organs recovered from these patients – and particularly the hearts – don’t seem to be any worse in terms of allograft survival than the organs recovered from patients who died from other causes, which are the traditional sources, like blunt head trauma, gunshot wounds, or stroke, that lead to brain death. In general, these organs are useful and do quite well,” according to Dr. Desai, medical director of the cardiomyopathy and heart failure program at Brigham and Women’s Hospital, Boston.
He highlighted several other recent developments in the field of cardiac transplantation that promise to further expand the donor heart pool, including acceptance of hepatitis C–infected donors and organ donation after circulatory rather than brain death. Dr. Desai also drew attention to the unintended perverse consequences of a recent redesign of the U.S. donor heart allocation system and discussed the impressive improvement in clinical outcomes with mechanical circulatory support. He noted that, while relatively few cardiologists practice in the highly specialized centers where heart transplants take place, virtually all cardiologists are affected by advances in heart transplantation since hundreds of thousands of the estimated 7 million Americans with heart failure have advanced disease.
Heart transplantation, he emphasized, is becoming increasingly complex. Recipients are on average older, sicker, and have more comorbidities than in times past. As a result, there is greater need for dual organ transplants: heart/lung, heart/liver, or heart/kidney. Plus, more patients come to transplantation after prior cardiac surgery for implantation of a ventricular assist device, so sensitization to blood products is a growing issue. And, of course, the pool of transplant candidates has expanded.
“We’re now forced to take patients previously considered to have contraindications to transplant; for example, diabetes was a contraindication to transplant in the early years, but now it’s the rule in 35%-40% of our patients who present with advanced heart failure,” the cardiologist noted.
Transplants from HCV-infected donors to uninfected recipients
Hearts and lungs from donors with hepatitis C viremia were traditionally deemed unsuitable for transplant. That’s all changed in the current era of highly effective direct-acting antiviral agents for the treatment of HCV infection.
In the DONATE HCV trial, Dr. Desai’s colleagues at Brigham and Women’s Hospital showed that giving HCV-uninfected recipients of hearts or lungs from HCV-viremic donors a shortened 4-week course of treatment with sofosbuvir-velpatasvir (Epclusa) beginning within a few hours after transplantation uniformly blocked viral replication. Six months after transplantation, none of the study participants had a detectable HCV viral load, and all had excellent graft function (N Engl J Med. 2019 Apr 25;380[17]:1606-17).
“This is effective prevention of HCV infection by aggressive upfront therapy,” Dr. Desai explained. “We can now take organs from HCV-viremic patients and use them in solid organ transplantation. This has led to a skyrocketing increase in donors with HCV infection, and those donations have helped us clear the waiting list.”
Donation after circulatory death
Australian transplant physicians have pioneered the use of donor hearts obtained after circulatory death in individuals with devastating neurologic injury who didn’t quite meet the criteria for brain death, which is the traditional prerequisite. In the new scenario, withdrawal of life-supporting therapy is followed by circulatory death, then the donor heart is procured and preserved via extracorporeal perfusion until transplantation.
The Australians report excellent outcomes, with rates of overall survival and rejection episodes similar to outcomes from brain-dead donors (J Am Coll Cardiol. 2019 Apr 2;73[12]:1447-59). The first U.S. heart transplant involving donation after circulatory death took place at Duke University in Durham, North Carolina. A multicenter U.S. clinical trial of this practice is underway.
If the results are positive and the practice of donation after circulatory death becomes widely implemented, the U.S. heart donor pool could increase by 30%.
Recent overhaul of donor heart allocation system may have backfired
The U.S. donor heart allocation system was redesigned in the fall of 2018 in an effort to reduce waiting times. One of the biggest changes involved breaking down the category with the highest urgency status into three new subcategories based upon sickness. Now, the highest-urgency category is for patients in cardiogenic shock who are supported by extracorporeal membrane oxygenation (ECMO) or other temporary mechanical circulatory support devices.
But an analysis of United Network for Organ Sharing (UNOS) data suggests this change has unintended adverse consequences for clinical outcomes.
Indeed, the investigators reported that the use of ECMO support is fourfold greater in the new system, the use of durable left ventricular assist devices (LVADs) as a bridge to transplant is down, and outcomes are worse. The 180-day rate of freedom from death or retransplantation was 77.9%, down significantly from 93.4% in the former system. In a multivariate analysis, patients transplanted in the new system had an adjusted 2.1-fold increased risk of death or retransplantation (J Heart Lung Transplant. 2020 Jan;39[1]:1-4).
“When you create a new listing system, you create new incentives, and people start to manage patients differently,” Dr. Desai observed. “Increasingly now, the path direct to transplant is through temporary mechanical circulatory support rather than durable mechanical circulatory support. Is that a good idea? We don’t know, but if you look at the best data, those on ECMO or percutaneous VADs have the worst outcomes. So the question of whether we should take the sickest of sick patients directly to transplant as a standard strategy has come under scrutiny.”
Improved durable LVAD technology brings impressive clinical outcomes
Results of the landmark MOMENTUM 3 randomized trial showed that 2-year clinical outcomes with the magnetically levitated centrifugal-flow HeartMate 3 LVAD now rival those of percutaneous mitral valve repair using the MitraClip device. Two-year all-cause mortality in the LVAD recipients was 22% versus 29.1% with the MitraClip in the COAPT trial and 34.9% in the MITRA-FR trial. The HeartMate 3 reduces the hemocompatibility issues that plagued earlier-generation durable LVADs, with resultant lower rates of pump thrombosis, stroke, and GI bleeding. Indeed, the outcomes in MOMENTUM 3 were so good – and so similar – with the HeartMate 3, regardless of whether the intended treatment goal was as a bridge to transplant or as lifelong destination therapy, that the investigators have recently proposed doing away with those distinctions.
“It is possible that use of arbitrary categorizations based on current or future transplant eligibility should be clinically abandoned in favor of a single preimplant strategy: to extend the survival and improve the quality of life of patients with medically refractory heart failure,” according to the investigators (JAMA Cardiol. 2020 Jan 15. doi: 10.1001/jamacardio.2019.5323).
The next step forward in LVAD technology is already on the horizon: a fully implantable device that eliminates the transcutaneous drive-line for the power supply, which is prone to infection and diminishes overall quality of life. This investigational device utilizes wireless coplanar energy transfer, with a coil ring placed around the lung and fixed to the chest wall. The implanted battery provides more than 6 hours of power without a recharge (J Heart Lung Transplant. 2019 Apr;38[4]:339-43).
“The first LVAD patient has gone swimming in Kazakhstan,” according to Dr. Desai.
Myocardial recovery in LVAD recipients remains elusive
The initial hope for LVADs was that they would not only be able to serve as a bridge to transplantation or as lifetime therapy, but that the prolonged unloading of the ventricle would enable potent medical therapy to rescue myocardial function so that the device could eventually be explanted. That does happen, but only rarely. In a large registry study, myocardial recovery occurred in only about 1% of patients on mechanical circulatory support. Attempts to enhance the process by add-on stem cell therapy have thus far been ineffective.
“For the moment, recovery is still a hope, not a reality,” the cardiologist said.
He reported serving as a consultant to more than a dozen pharmaceutical or medical device companies and receiving research grants from Alnylam, AstraZeneca, Bayer Healthcare, MyoKardia, and Novartis.
EXPERT ANALYSIS FROM ACC SNOWMASS 2020
Vigilance safely keeps AFib patients off anticoagulants post ablation
NATIONAL HARBOR, MD. – A pilot program of daily arrhythmia self-vigilance has allowed selected patients with no atrial fibrillation following a catheter ablation procedure to safely come off a regimen of daily oral anticoagulation despite having residual risk factors for ischemic stroke.
This program, which started several years ago at the University of Pennsylvania in Philadelphia, has now managed 190 patients and followed them for a median of just over 3 years, and during 576 patient-years of follow-up, just a single patient had an ischemic cerebrovascular event that occurred with no atrial fibrillation (AFib) recurrence and appeared to be caused by an atherosclerotic embolism, Francis E. Marchlinski, MD, said at the annual International AF Symposium.
Although this strategy has not yet been tested in a prospective, randomized trial, this anecdotal, single-center experience suggests that the approach is “safe and effective” for selected patients who are eager to come off of their anticoagulation regimen when they remain arrhythmia free following catheter ablation of their AFib, said Dr. Marchlinski, professor of medicine and director of electrophysiology at the University of Pennsylvania. He and his associates developed this strategy as a way to more safely allow these patients to stop taking a daily oral anticoagulant because he found that many patients were stopping on their own, with no safety strategy in place.
“Patients tell me they don’t want to be on an oral anticoagulant because a parent had a hemorrhagic stroke, and they say they’re willing to accept the risk” of having an ischemic stroke by coming off anticoagulation. “This is a way for them to do it safely,” Dr. Marchlinski said in an interview. He stressed that he only allows his patients to go this route if they understand the risk and accept their shared responsibility for vigilant, twice-daily pulse monitoring to detect resumption of an irregular heart beat.
Since 2011, Dr. Marchlinski’s program ablated 1,216 patients with AFib who then remained arrhythmia free during 3 weeks of continuous ECG monitoring following their procedure. Among these patients, 443 had a CHA2DS2-VAScscore of either 0 (men) or 1 (women) that indicated no ongoing need for oral anticoagulation according to current guidelines. Of the remaining 773 patients with a CHA2DS2-VASc score of at least 1 in men and 2 in women, the clinicians determined 583 to be ineligible for the program because of their unwillingness to accept the risk, unwillingness to comply with daily pulse checks, a history of asymptomatic AFib, a CHA2DS2-VASc score greater than 4, or a resting pulse above 90 beats per minute, leaving 190 patients eligible to participate. Among these patients, 105 (55%) had a CHA2DS2-VASc score of 2-4, which should prompt anticoagulation according to current guidelines.Participating patients committed to check their resting pulse by palpation at least twice daily and to contacting the program immediately if their resting rate spiked by more than 20 beats per minutes or in another way seemed irregular. Patients were also instructed to restart their oral anticoagulation immediately if they experienced AFib symptoms that persisted for more than 5 minutes. Many patients in the program also use a wearable device (usually a watch) to monitor their resting pulse and to generate a 30-second ECG recording that they can send as an electronic file to the University of Pennsylvania staff. “We embrace wearables,” Dr. Marchlinski said. Those without a wearable can undergo transtelephonic EEG monitoring to document a suspected arrhythmia recurrence, and all patients undergo annual monitoring by continuous ECG for at least 2 weeks.During follow-up, in addition to the 1 patient free from recurrent AFib who had an atherosclerotic embolism, 34 patients resumed anticoagulant treatment because of AFib recurrence; 12 withdrew from the program because of noncompliance or preference, or because an exclusion appeared; 29 resumed oral anticoagulation transiently but then discontinued the drug a second time when their AFib recurrence resolved; and 114 patients (60% of the starting cohort of 190) remained completely off anticoagulation during a median of 37 months. These data updated a published report from Dr. Marchlinski and his associates on their first 99 patients followed for a median of 30 months (J Cardiovasc Electrophysiol. 2019 May;30[5]:631-8).
This experience underscored the need for ongoing rhythm monitoring even in the absence of AFib symptoms, as six patients developed asymptomatic AFib detected by monitoring, including one patient whose recurrence occurred 30 months after the ablation procedure.
Dr. Marchlinski stressed the stringent selection process he applies to limit this approach to patients who are willing to faithfully monitor their pulse and symptoms daily, and who accept the risk that this approach may pose and their responsibility to stay in contact with the clinical team. The program calls patients at the 6-month mark between annual monitoring to remind them of their need for daily attention.
“Being off anticoagulants is very important to these patients,” he explained, and he highlighted the added workload this strategy places on his staff. “I think this has legs” for adoption by other cardiac arrhythmia programs, “but it depends on the time the staff is willing to spend” monitoring and following these patients, some of whom regularly send in ECG traces from their wearable devices for assessment. “It takes a village” to make this program work, he said.
Dr. Marchlinski has been a consultant to or has received honoraria from Abbott EP/St. Jude, Biosense Webster, Biotronik, Boston Scientific, and Medtronic.
NATIONAL HARBOR, MD. – A pilot program of daily arrhythmia self-vigilance has allowed selected patients with no atrial fibrillation following a catheter ablation procedure to safely come off a regimen of daily oral anticoagulation despite having residual risk factors for ischemic stroke.
This program, which started several years ago at the University of Pennsylvania in Philadelphia, has now managed 190 patients and followed them for a median of just over 3 years, and during 576 patient-years of follow-up, just a single patient had an ischemic cerebrovascular event that occurred with no atrial fibrillation (AFib) recurrence and appeared to be caused by an atherosclerotic embolism, Francis E. Marchlinski, MD, said at the annual International AF Symposium.
Although this strategy has not yet been tested in a prospective, randomized trial, this anecdotal, single-center experience suggests that the approach is “safe and effective” for selected patients who are eager to come off of their anticoagulation regimen when they remain arrhythmia free following catheter ablation of their AFib, said Dr. Marchlinski, professor of medicine and director of electrophysiology at the University of Pennsylvania. He and his associates developed this strategy as a way to more safely allow these patients to stop taking a daily oral anticoagulant because he found that many patients were stopping on their own, with no safety strategy in place.
“Patients tell me they don’t want to be on an oral anticoagulant because a parent had a hemorrhagic stroke, and they say they’re willing to accept the risk” of having an ischemic stroke by coming off anticoagulation. “This is a way for them to do it safely,” Dr. Marchlinski said in an interview. He stressed that he only allows his patients to go this route if they understand the risk and accept their shared responsibility for vigilant, twice-daily pulse monitoring to detect resumption of an irregular heart beat.
Since 2011, Dr. Marchlinski’s program ablated 1,216 patients with AFib who then remained arrhythmia free during 3 weeks of continuous ECG monitoring following their procedure. Among these patients, 443 had a CHA2DS2-VAScscore of either 0 (men) or 1 (women) that indicated no ongoing need for oral anticoagulation according to current guidelines. Of the remaining 773 patients with a CHA2DS2-VASc score of at least 1 in men and 2 in women, the clinicians determined 583 to be ineligible for the program because of their unwillingness to accept the risk, unwillingness to comply with daily pulse checks, a history of asymptomatic AFib, a CHA2DS2-VASc score greater than 4, or a resting pulse above 90 beats per minute, leaving 190 patients eligible to participate. Among these patients, 105 (55%) had a CHA2DS2-VASc score of 2-4, which should prompt anticoagulation according to current guidelines.Participating patients committed to check their resting pulse by palpation at least twice daily and to contacting the program immediately if their resting rate spiked by more than 20 beats per minutes or in another way seemed irregular. Patients were also instructed to restart their oral anticoagulation immediately if they experienced AFib symptoms that persisted for more than 5 minutes. Many patients in the program also use a wearable device (usually a watch) to monitor their resting pulse and to generate a 30-second ECG recording that they can send as an electronic file to the University of Pennsylvania staff. “We embrace wearables,” Dr. Marchlinski said. Those without a wearable can undergo transtelephonic EEG monitoring to document a suspected arrhythmia recurrence, and all patients undergo annual monitoring by continuous ECG for at least 2 weeks.During follow-up, in addition to the 1 patient free from recurrent AFib who had an atherosclerotic embolism, 34 patients resumed anticoagulant treatment because of AFib recurrence; 12 withdrew from the program because of noncompliance or preference, or because an exclusion appeared; 29 resumed oral anticoagulation transiently but then discontinued the drug a second time when their AFib recurrence resolved; and 114 patients (60% of the starting cohort of 190) remained completely off anticoagulation during a median of 37 months. These data updated a published report from Dr. Marchlinski and his associates on their first 99 patients followed for a median of 30 months (J Cardiovasc Electrophysiol. 2019 May;30[5]:631-8).
This experience underscored the need for ongoing rhythm monitoring even in the absence of AFib symptoms, as six patients developed asymptomatic AFib detected by monitoring, including one patient whose recurrence occurred 30 months after the ablation procedure.
Dr. Marchlinski stressed the stringent selection process he applies to limit this approach to patients who are willing to faithfully monitor their pulse and symptoms daily, and who accept the risk that this approach may pose and their responsibility to stay in contact with the clinical team. The program calls patients at the 6-month mark between annual monitoring to remind them of their need for daily attention.
“Being off anticoagulants is very important to these patients,” he explained, and he highlighted the added workload this strategy places on his staff. “I think this has legs” for adoption by other cardiac arrhythmia programs, “but it depends on the time the staff is willing to spend” monitoring and following these patients, some of whom regularly send in ECG traces from their wearable devices for assessment. “It takes a village” to make this program work, he said.
Dr. Marchlinski has been a consultant to or has received honoraria from Abbott EP/St. Jude, Biosense Webster, Biotronik, Boston Scientific, and Medtronic.
NATIONAL HARBOR, MD. – A pilot program of daily arrhythmia self-vigilance has allowed selected patients with no atrial fibrillation following a catheter ablation procedure to safely come off a regimen of daily oral anticoagulation despite having residual risk factors for ischemic stroke.
This program, which started several years ago at the University of Pennsylvania in Philadelphia, has now managed 190 patients and followed them for a median of just over 3 years, and during 576 patient-years of follow-up, just a single patient had an ischemic cerebrovascular event that occurred with no atrial fibrillation (AFib) recurrence and appeared to be caused by an atherosclerotic embolism, Francis E. Marchlinski, MD, said at the annual International AF Symposium.
Although this strategy has not yet been tested in a prospective, randomized trial, this anecdotal, single-center experience suggests that the approach is “safe and effective” for selected patients who are eager to come off of their anticoagulation regimen when they remain arrhythmia free following catheter ablation of their AFib, said Dr. Marchlinski, professor of medicine and director of electrophysiology at the University of Pennsylvania. He and his associates developed this strategy as a way to more safely allow these patients to stop taking a daily oral anticoagulant because he found that many patients were stopping on their own, with no safety strategy in place.
“Patients tell me they don’t want to be on an oral anticoagulant because a parent had a hemorrhagic stroke, and they say they’re willing to accept the risk” of having an ischemic stroke by coming off anticoagulation. “This is a way for them to do it safely,” Dr. Marchlinski said in an interview. He stressed that he only allows his patients to go this route if they understand the risk and accept their shared responsibility for vigilant, twice-daily pulse monitoring to detect resumption of an irregular heart beat.
Since 2011, Dr. Marchlinski’s program ablated 1,216 patients with AFib who then remained arrhythmia free during 3 weeks of continuous ECG monitoring following their procedure. Among these patients, 443 had a CHA2DS2-VAScscore of either 0 (men) or 1 (women) that indicated no ongoing need for oral anticoagulation according to current guidelines. Of the remaining 773 patients with a CHA2DS2-VASc score of at least 1 in men and 2 in women, the clinicians determined 583 to be ineligible for the program because of their unwillingness to accept the risk, unwillingness to comply with daily pulse checks, a history of asymptomatic AFib, a CHA2DS2-VASc score greater than 4, or a resting pulse above 90 beats per minute, leaving 190 patients eligible to participate. Among these patients, 105 (55%) had a CHA2DS2-VASc score of 2-4, which should prompt anticoagulation according to current guidelines.Participating patients committed to check their resting pulse by palpation at least twice daily and to contacting the program immediately if their resting rate spiked by more than 20 beats per minutes or in another way seemed irregular. Patients were also instructed to restart their oral anticoagulation immediately if they experienced AFib symptoms that persisted for more than 5 minutes. Many patients in the program also use a wearable device (usually a watch) to monitor their resting pulse and to generate a 30-second ECG recording that they can send as an electronic file to the University of Pennsylvania staff. “We embrace wearables,” Dr. Marchlinski said. Those without a wearable can undergo transtelephonic EEG monitoring to document a suspected arrhythmia recurrence, and all patients undergo annual monitoring by continuous ECG for at least 2 weeks.During follow-up, in addition to the 1 patient free from recurrent AFib who had an atherosclerotic embolism, 34 patients resumed anticoagulant treatment because of AFib recurrence; 12 withdrew from the program because of noncompliance or preference, or because an exclusion appeared; 29 resumed oral anticoagulation transiently but then discontinued the drug a second time when their AFib recurrence resolved; and 114 patients (60% of the starting cohort of 190) remained completely off anticoagulation during a median of 37 months. These data updated a published report from Dr. Marchlinski and his associates on their first 99 patients followed for a median of 30 months (J Cardiovasc Electrophysiol. 2019 May;30[5]:631-8).
This experience underscored the need for ongoing rhythm monitoring even in the absence of AFib symptoms, as six patients developed asymptomatic AFib detected by monitoring, including one patient whose recurrence occurred 30 months after the ablation procedure.
Dr. Marchlinski stressed the stringent selection process he applies to limit this approach to patients who are willing to faithfully monitor their pulse and symptoms daily, and who accept the risk that this approach may pose and their responsibility to stay in contact with the clinical team. The program calls patients at the 6-month mark between annual monitoring to remind them of their need for daily attention.
“Being off anticoagulants is very important to these patients,” he explained, and he highlighted the added workload this strategy places on his staff. “I think this has legs” for adoption by other cardiac arrhythmia programs, “but it depends on the time the staff is willing to spend” monitoring and following these patients, some of whom regularly send in ECG traces from their wearable devices for assessment. “It takes a village” to make this program work, he said.
Dr. Marchlinski has been a consultant to or has received honoraria from Abbott EP/St. Jude, Biosense Webster, Biotronik, Boston Scientific, and Medtronic.
REPORTING FROM THE AF SYMPOSIUM 2020
FDA approves fidaxomicin for treatment of C. difficile–associated diarrhea
The Food and Drug Administration has approved fidaxomicin (Dificid) for the treatment of Clostridioides difficile–associated diarrhea in children aged 6 months and older.
Approval was based on results from SUNSHINE, a phase 3, multicenter, investigator-blind, randomized, parallel-group study in 142 pediatric patients aged between 6 months and 18 years with confirmed C. difficile infection who received either fidaxomicin or vancomycin for 10 days. Clinical response 2 days after the conclusion of treatment was similar in both groups (77.6% for fidaxomicin vs. 70.5% for vancomycin), and fidaxomicin had a superior sustained response 30 days after the conclusion of treatment (68.4% vs. 50.0%).
The safety of fidaxomicin was assessed in a pair of clinical trials involving 136 patients; the most common adverse events were pyrexia, abdominal pain, vomiting, diarrhea, constipation, increased aminotransferases, and rash. Four patients discontinued fidaxomicin treatment because of adverse events, and four patients died during the trials, though all deaths were in patients aged younger than 2 years and seemed to be related to other comorbidities.
“C. difficile is an important cause of health care– and community-associated diarrheal illness in children, and sustained cure is difficult to achieve in some patients. The fidaxomicin pediatric trial was the first randomized, controlled trial of C. difficile infection treatment in children,” Larry K. Kociolek, MD, associate medical director of infection prevention and control at Ann & Robert H. Lurie Children’s Hospital of Chicago, said in the press release from Merck, manufacturer of fidaxomicin.
*This story was updated on 1/27/2020.
The Food and Drug Administration has approved fidaxomicin (Dificid) for the treatment of Clostridioides difficile–associated diarrhea in children aged 6 months and older.
Approval was based on results from SUNSHINE, a phase 3, multicenter, investigator-blind, randomized, parallel-group study in 142 pediatric patients aged between 6 months and 18 years with confirmed C. difficile infection who received either fidaxomicin or vancomycin for 10 days. Clinical response 2 days after the conclusion of treatment was similar in both groups (77.6% for fidaxomicin vs. 70.5% for vancomycin), and fidaxomicin had a superior sustained response 30 days after the conclusion of treatment (68.4% vs. 50.0%).
The safety of fidaxomicin was assessed in a pair of clinical trials involving 136 patients; the most common adverse events were pyrexia, abdominal pain, vomiting, diarrhea, constipation, increased aminotransferases, and rash. Four patients discontinued fidaxomicin treatment because of adverse events, and four patients died during the trials, though all deaths were in patients aged younger than 2 years and seemed to be related to other comorbidities.
“C. difficile is an important cause of health care– and community-associated diarrheal illness in children, and sustained cure is difficult to achieve in some patients. The fidaxomicin pediatric trial was the first randomized, controlled trial of C. difficile infection treatment in children,” Larry K. Kociolek, MD, associate medical director of infection prevention and control at Ann & Robert H. Lurie Children’s Hospital of Chicago, said in the press release from Merck, manufacturer of fidaxomicin.
*This story was updated on 1/27/2020.
The Food and Drug Administration has approved fidaxomicin (Dificid) for the treatment of Clostridioides difficile–associated diarrhea in children aged 6 months and older.
Approval was based on results from SUNSHINE, a phase 3, multicenter, investigator-blind, randomized, parallel-group study in 142 pediatric patients aged between 6 months and 18 years with confirmed C. difficile infection who received either fidaxomicin or vancomycin for 10 days. Clinical response 2 days after the conclusion of treatment was similar in both groups (77.6% for fidaxomicin vs. 70.5% for vancomycin), and fidaxomicin had a superior sustained response 30 days after the conclusion of treatment (68.4% vs. 50.0%).
The safety of fidaxomicin was assessed in a pair of clinical trials involving 136 patients; the most common adverse events were pyrexia, abdominal pain, vomiting, diarrhea, constipation, increased aminotransferases, and rash. Four patients discontinued fidaxomicin treatment because of adverse events, and four patients died during the trials, though all deaths were in patients aged younger than 2 years and seemed to be related to other comorbidities.
“C. difficile is an important cause of health care– and community-associated diarrheal illness in children, and sustained cure is difficult to achieve in some patients. The fidaxomicin pediatric trial was the first randomized, controlled trial of C. difficile infection treatment in children,” Larry K. Kociolek, MD, associate medical director of infection prevention and control at Ann & Robert H. Lurie Children’s Hospital of Chicago, said in the press release from Merck, manufacturer of fidaxomicin.
*This story was updated on 1/27/2020.
Sociodemographic disadvantage confers poorer survival in young adults with CRC
SAN FRANCISCO – Young adults with colorectal cancer who live in neighborhoods with higher levels of disadvantage differ on health measures, present with more advanced disease, and have poorer survival. These were among key findings of a retrospective cohort study reported at the 2020 GI Cancers Symposium.
The incidence of colorectal cancer has risen sharply – 51% – since 1994 among individuals aged younger than age 50 years, with the greatest uptick seen among those aged 20-29 years (J Natl Cancer Inst. 2017;109[8]. doi: 10.1093/jnci/djw322).
“Sociodemographic disparities have been linked to inferior survival. However, their impact and association with outcome in young adults is not well described,” said lead investigator Ashley Matusz-Fisher, MD, of the Levine Cancer Institute in Charlotte, N.C.
The investigators analyzed data from the National Cancer Database for the years 2004-2016, identifying 26,768 patients who received a colorectal cancer diagnosis when aged 18-40 years.
Results showed that those living in areas with low income (less than $38,000 annually) and low educational attainment (high school graduation rate less than 79%), and those living in urban or rural areas (versus metropolitan areas) had 24% and 10% higher risks of death, respectively.
Patients in the low-income, low-education group were more than six times as likely to be black and to lack private health insurance, had greater comorbidity, had larger tumors and more nodal involvement at diagnosis, and were less likely to undergo surgery.
Several factors may be at play for the low-income, low-education group, Dr. Matusz-Fisher speculated: limited access to care, lack of awareness of important symptoms, and inability to afford treatment when it is needed. “That could very well be contributing to them presenting at later stages and then maybe not getting the treatment that other people who have insurance would be getting.
“To try to eliminate these disparities, the first step is recognition, which is what we are doing – recognizing there are disparities – and then making people aware of these disparities,” she commented. “More efforts are needed to increase access and remove barriers to care, with the hope of eliminating disparities and achieving health equity.”
Mitigating disparities
Several studies have looked at mitigating sociodemographic-related disparities in colorectal cancer outcomes, according to session cochair John M. Carethers, MD, AGAF, professor and chair of the department of internal medicine at the University of Michigan, Ann Arbor.
A large Delaware initiative tackled the problem via screening (J Clin Oncol. 2013;31:1928-30). “Now this was over 50 – we don’t typically screen under 50 – but over 50, you can essentially eliminate this disparity with navigation services and screening. How do you do that under 50? I’m not quite sure,” he said in an interview, adding that some organizations are recommending lowering the screening age to 45 or even 40 years in light of rising incidence among young adults.
However, accumulating evidence suggests that there may be inherent biological differences that are harder to overcome. “There is a lot of data … showing that polyps happen earlier and they are bigger in certain racial groups, particularly African Americans and American Indians,” Dr. Carethers elaborated. What is driving the biology is unknown, but the microbiome has come under scrutiny.
“So you are a victim of your circumstances,” he summarized. “You are living in a low-income area, you are eating more proinflammatory-type foods, you are getting your polyps earlier, and then you are getting your cancers earlier.”
Study details
Rural, urban, or metropolitan status was ascertained for 25,861 patients in the study, and area income and education were ascertained for 7,743 patients, according to data reported at the symposium, sponsored by the American Gastroenterological Association, the American Society of Clinical Oncology, the American Society for Radiation Oncology, and the Society of Surgical Oncology.
Compared with counterparts living in areas with both high annual income (greater than $68,000) and education (greater than 93% high school graduation rate), patients living in areas with both low annual income (less than $38,000) and education ( less than 79% high school graduation rate) were significantly more likely to be black (odds ratio, 6.4), not have private insurance (odds ratio, 6.3), have pathologic T3/T4 stage (OR, 1.4), have positive nodes (OR, 1.2), and have a Charlson-Deyo comorbidity score of 1 or greater (OR, 1.6). They also were less likely to undergo surgery (OR, 0.63) and more likely to be rehospitalized within 30 days (OR, 1.3).
After adjusting for race, insurance status, T/N stage, and comorbidity score, relative to counterparts in the high-income, high-education group, patients in the low-income, low-education group had an increased risk of death (hazard ratio, 1.24; P = .004). And relative to counterparts living in metropolitan areas, patients living in urban or rural areas had an increased risk of death (HR, 1.10; P = .02).
Among patients with stage IV disease, median overall survival was 26.1 months for those from high-income, high-education areas, but 20.7 months for those from low-income, low-education areas (P less than .001).
Dr. Matusz-Fisher did not report any conflicts of interest. The study did not receive any funding.
SOURCE: Matusz-Fisher A et al. 2020 GI Cancers Symposium, Abstract 13.
SAN FRANCISCO – Young adults with colorectal cancer who live in neighborhoods with higher levels of disadvantage differ on health measures, present with more advanced disease, and have poorer survival. These were among key findings of a retrospective cohort study reported at the 2020 GI Cancers Symposium.
The incidence of colorectal cancer has risen sharply – 51% – since 1994 among individuals aged younger than age 50 years, with the greatest uptick seen among those aged 20-29 years (J Natl Cancer Inst. 2017;109[8]. doi: 10.1093/jnci/djw322).
“Sociodemographic disparities have been linked to inferior survival. However, their impact and association with outcome in young adults is not well described,” said lead investigator Ashley Matusz-Fisher, MD, of the Levine Cancer Institute in Charlotte, N.C.
The investigators analyzed data from the National Cancer Database for the years 2004-2016, identifying 26,768 patients who received a colorectal cancer diagnosis when aged 18-40 years.
Results showed that those living in areas with low income (less than $38,000 annually) and low educational attainment (high school graduation rate less than 79%), and those living in urban or rural areas (versus metropolitan areas) had 24% and 10% higher risks of death, respectively.
Patients in the low-income, low-education group were more than six times as likely to be black and to lack private health insurance, had greater comorbidity, had larger tumors and more nodal involvement at diagnosis, and were less likely to undergo surgery.
Several factors may be at play for the low-income, low-education group, Dr. Matusz-Fisher speculated: limited access to care, lack of awareness of important symptoms, and inability to afford treatment when it is needed. “That could very well be contributing to them presenting at later stages and then maybe not getting the treatment that other people who have insurance would be getting.
“To try to eliminate these disparities, the first step is recognition, which is what we are doing – recognizing there are disparities – and then making people aware of these disparities,” she commented. “More efforts are needed to increase access and remove barriers to care, with the hope of eliminating disparities and achieving health equity.”
Mitigating disparities
Several studies have looked at mitigating sociodemographic-related disparities in colorectal cancer outcomes, according to session cochair John M. Carethers, MD, AGAF, professor and chair of the department of internal medicine at the University of Michigan, Ann Arbor.
A large Delaware initiative tackled the problem via screening (J Clin Oncol. 2013;31:1928-30). “Now this was over 50 – we don’t typically screen under 50 – but over 50, you can essentially eliminate this disparity with navigation services and screening. How do you do that under 50? I’m not quite sure,” he said in an interview, adding that some organizations are recommending lowering the screening age to 45 or even 40 years in light of rising incidence among young adults.
However, accumulating evidence suggests that there may be inherent biological differences that are harder to overcome. “There is a lot of data … showing that polyps happen earlier and they are bigger in certain racial groups, particularly African Americans and American Indians,” Dr. Carethers elaborated. What is driving the biology is unknown, but the microbiome has come under scrutiny.
“So you are a victim of your circumstances,” he summarized. “You are living in a low-income area, you are eating more proinflammatory-type foods, you are getting your polyps earlier, and then you are getting your cancers earlier.”
Study details
Rural, urban, or metropolitan status was ascertained for 25,861 patients in the study, and area income and education were ascertained for 7,743 patients, according to data reported at the symposium, sponsored by the American Gastroenterological Association, the American Society of Clinical Oncology, the American Society for Radiation Oncology, and the Society of Surgical Oncology.
Compared with counterparts living in areas with both high annual income (greater than $68,000) and education (greater than 93% high school graduation rate), patients living in areas with both low annual income (less than $38,000) and education ( less than 79% high school graduation rate) were significantly more likely to be black (odds ratio, 6.4), not have private insurance (odds ratio, 6.3), have pathologic T3/T4 stage (OR, 1.4), have positive nodes (OR, 1.2), and have a Charlson-Deyo comorbidity score of 1 or greater (OR, 1.6). They also were less likely to undergo surgery (OR, 0.63) and more likely to be rehospitalized within 30 days (OR, 1.3).
After adjusting for race, insurance status, T/N stage, and comorbidity score, relative to counterparts in the high-income, high-education group, patients in the low-income, low-education group had an increased risk of death (hazard ratio, 1.24; P = .004). And relative to counterparts living in metropolitan areas, patients living in urban or rural areas had an increased risk of death (HR, 1.10; P = .02).
Among patients with stage IV disease, median overall survival was 26.1 months for those from high-income, high-education areas, but 20.7 months for those from low-income, low-education areas (P less than .001).
Dr. Matusz-Fisher did not report any conflicts of interest. The study did not receive any funding.
SOURCE: Matusz-Fisher A et al. 2020 GI Cancers Symposium, Abstract 13.
SAN FRANCISCO – Young adults with colorectal cancer who live in neighborhoods with higher levels of disadvantage differ on health measures, present with more advanced disease, and have poorer survival. These were among key findings of a retrospective cohort study reported at the 2020 GI Cancers Symposium.
The incidence of colorectal cancer has risen sharply – 51% – since 1994 among individuals aged younger than age 50 years, with the greatest uptick seen among those aged 20-29 years (J Natl Cancer Inst. 2017;109[8]. doi: 10.1093/jnci/djw322).
“Sociodemographic disparities have been linked to inferior survival. However, their impact and association with outcome in young adults is not well described,” said lead investigator Ashley Matusz-Fisher, MD, of the Levine Cancer Institute in Charlotte, N.C.
The investigators analyzed data from the National Cancer Database for the years 2004-2016, identifying 26,768 patients who received a colorectal cancer diagnosis when aged 18-40 years.
Results showed that those living in areas with low income (less than $38,000 annually) and low educational attainment (high school graduation rate less than 79%), and those living in urban or rural areas (versus metropolitan areas) had 24% and 10% higher risks of death, respectively.
Patients in the low-income, low-education group were more than six times as likely to be black and to lack private health insurance, had greater comorbidity, had larger tumors and more nodal involvement at diagnosis, and were less likely to undergo surgery.
Several factors may be at play for the low-income, low-education group, Dr. Matusz-Fisher speculated: limited access to care, lack of awareness of important symptoms, and inability to afford treatment when it is needed. “That could very well be contributing to them presenting at later stages and then maybe not getting the treatment that other people who have insurance would be getting.
“To try to eliminate these disparities, the first step is recognition, which is what we are doing – recognizing there are disparities – and then making people aware of these disparities,” she commented. “More efforts are needed to increase access and remove barriers to care, with the hope of eliminating disparities and achieving health equity.”
Mitigating disparities
Several studies have looked at mitigating sociodemographic-related disparities in colorectal cancer outcomes, according to session cochair John M. Carethers, MD, AGAF, professor and chair of the department of internal medicine at the University of Michigan, Ann Arbor.
A large Delaware initiative tackled the problem via screening (J Clin Oncol. 2013;31:1928-30). “Now this was over 50 – we don’t typically screen under 50 – but over 50, you can essentially eliminate this disparity with navigation services and screening. How do you do that under 50? I’m not quite sure,” he said in an interview, adding that some organizations are recommending lowering the screening age to 45 or even 40 years in light of rising incidence among young adults.
However, accumulating evidence suggests that there may be inherent biological differences that are harder to overcome. “There is a lot of data … showing that polyps happen earlier and they are bigger in certain racial groups, particularly African Americans and American Indians,” Dr. Carethers elaborated. What is driving the biology is unknown, but the microbiome has come under scrutiny.
“So you are a victim of your circumstances,” he summarized. “You are living in a low-income area, you are eating more proinflammatory-type foods, you are getting your polyps earlier, and then you are getting your cancers earlier.”
Study details
Rural, urban, or metropolitan status was ascertained for 25,861 patients in the study, and area income and education were ascertained for 7,743 patients, according to data reported at the symposium, sponsored by the American Gastroenterological Association, the American Society of Clinical Oncology, the American Society for Radiation Oncology, and the Society of Surgical Oncology.
Compared with counterparts living in areas with both high annual income (greater than $68,000) and education (greater than 93% high school graduation rate), patients living in areas with both low annual income (less than $38,000) and education ( less than 79% high school graduation rate) were significantly more likely to be black (odds ratio, 6.4), not have private insurance (odds ratio, 6.3), have pathologic T3/T4 stage (OR, 1.4), have positive nodes (OR, 1.2), and have a Charlson-Deyo comorbidity score of 1 or greater (OR, 1.6). They also were less likely to undergo surgery (OR, 0.63) and more likely to be rehospitalized within 30 days (OR, 1.3).
After adjusting for race, insurance status, T/N stage, and comorbidity score, relative to counterparts in the high-income, high-education group, patients in the low-income, low-education group had an increased risk of death (hazard ratio, 1.24; P = .004). And relative to counterparts living in metropolitan areas, patients living in urban or rural areas had an increased risk of death (HR, 1.10; P = .02).
Among patients with stage IV disease, median overall survival was 26.1 months for those from high-income, high-education areas, but 20.7 months for those from low-income, low-education areas (P less than .001).
Dr. Matusz-Fisher did not report any conflicts of interest. The study did not receive any funding.
SOURCE: Matusz-Fisher A et al. 2020 GI Cancers Symposium, Abstract 13.
REPORTING FROM THE 2020 GI CANCERS SYMPOSIUM
Cannabis for sleep: Short-term benefit, long-term disruption?
, new research shows.
Investigators found whole-plant medical cannabis use was associated with fewer problems with respect to waking up at night, but they also found that frequent medical cannabis use was associated with more problems initiating and maintaining sleep.
“Cannabis may improve overall sleep in the short term,” study investigator Sharon Sznitman, PhD, University of Haifa (Israel) Faculty of Social Welfare and Health Sciences, said in an interview. “But it’s also very interesting that when we looked at frequency of use in the group that used medical cannabis, individuals who had more frequent use also had poorer sleep in the long term.
“This suggests that while cannabis may improve overall sleep, it’s also possible that there is a tolerance that develops with either very frequent or long-term use,” she added.
The study was published online Jan. 20 in BMJ Supportive and Palliative Care.
A common problem
Estimates suggest chronic pain affects up to 37% of adults in the developed world. Individuals who suffer chronic pain often experience comorbid insomnia, which includes difficulty initiating sleep, sleep disruption, and early morning wakening.
For its part, medical cannabis to treat chronic pain symptoms and manage sleep problems has been widely reported as a prime motivation for medical cannabis use. Indeed, previous studies have concluded that the endocannabinoid system plays a role in sleep regulation, including sleep promotion and maintenance.
In recent years, investigators have reported the beneficial effects of medical cannabis for sleep. Nevertheless, some preclinical research has also concluded that chronic administration of tetrahydrocannabinol may result in tolerance to the sleep-enhancing effects of cannabis.
With that in mind, the researchers set out to examine the potential impact of whole-plant medicinal cannabis on sleep problems experienced by middle-aged patients suffering from chronic pain.
“People are self-reporting that they’re using cannabis for sleep and that it helps, but as we know, just because people are reporting that it works doesn’t mean that it will hold up in research,” Dr. Sznitman said.
The study included 128 individuals (mean age, 61±6 years; 51% females) with chronic neuropathic pain: 66 were medical cannabis users and 62 were not.
Three indicators of insomnia were measured using the 7-point Likert scale to assess issues with sleep initiation and maintenance.
In addition, investigators collected sociodemographic information, as well as data on daily consumption of tobacco, frequency of alcohol use, and pain severity. Finally, they collected patient data on the use of sleep-aid medications during the past month as well as tricyclic antidepressant use.
Frequent use, more sleep problems?
On average, medical cannabis users were 3 years younger than their nonusing counterparts (mean age, 60±6 vs. 63±6 years, respectively, P = .003) and more likely to be male (58% vs 40%, respectively, P = .038). Otherwise, the two groups were comparable.
Medical cannabis users reported taking the drug for an average of 4 years, at an average quantity of 31 g per month. The primary mode of administration was smoking (68.6%), followed by oil extracts (21.4%) and vaporization (20%).
Results showed that, of the total sample, 24.1% reported always waking up early and not falling back to sleep, 20.2% reported always having difficulty falling asleep, and 27.2% reported always waking up during the night.
After adjusting for patient age, sex, pain level, and use of sleep medications and antidepressants, medical cannabis use was associated with fewer problems with waking up at night, compared with nonmedical cannabis use. No differences were found between groups with respect to problems falling asleep or waking up early without being able to fall back to sleep, Dr. Sznitman and associates reported.
The final analysis of a subsample of patients that only included medical cannabis users showed frequency of medical cannabis use was associated with sleep problems, they said.
Specifically, more frequent cannabis use was associated with more problems related to waking up at night, as well as problems falling asleep.
Sleep problems associated with frequent medical cannabis use may signal the development of tolerance to the agent. However, frequent users of medical cannabis also maybsuffer pain or other comorbidities, which, in turn, may be linked to more sleep problems.
Either way, Dr. Sznitman said the study might open the door to another treatment option for patients suffering from chronic pain who struggle with sleep.
“If future research shows that the effect of medical cannabis on sleep is a consistent one, then we may be adding a new therapy for sleep problems, which are huge in society and especially in chronic pain patients,” she said.
Early days
Commenting on the findings in an interview, Ryan G. Vandrey, PhD, who was not involved in the study, said the findings are in line with previous research.
“I think the results make sense with respect to the data I’ve collected and from what I’ve seen,” said Dr. Vandrey, associate professor of psychiatry and behavioral sciences at Johns Hopkins Medicine in Baltimore.
“We typically only want to use sleep medications for short periods of time,” he continued. “When you think about recommended prescribing practices for any hypnotic medication, it’s usually short term, 2 weeks or less. Longer-term use often leads to tolerance, dependence, and withdrawal symptoms when the medication is stopped, which leads to an exacerbation of disordered sleep,” Dr. Vandrey said.
Nevertheless, he urged caution when interpreting the results.
“I think the study warrants caution about long-term daily use of cannabinoids with respect to sleep,” he said. “But we need more detailed evaluations, as the trial wasn’t testing a defined product, specific dose, or dose regimen.
“In addition, this was all done in the context of people with chronic pain and not treating disordered sleep or insomnia, but the study highlights the importance of recognizing that long-term chronic use of cannabis is not likely to fully resolve sleep problems.”
Dr. Sznitman agreed that the research is still in its very early stages.
“We’re still far from saying we have the evidence to support the use of medical cannabis for sleep,” she said. “For in the end it was just a cross-sectional, observational study, so we cannot say anything about cause and effect. But if these results pan out, they could be far-reaching and exciting.”
The study was funded by the University of Haifa and Rambam Hospital in Israel, and by the Evelyn Lipper Foundation. Dr. Sznitman and Dr. Vandrey have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
, new research shows.
Investigators found whole-plant medical cannabis use was associated with fewer problems with respect to waking up at night, but they also found that frequent medical cannabis use was associated with more problems initiating and maintaining sleep.
“Cannabis may improve overall sleep in the short term,” study investigator Sharon Sznitman, PhD, University of Haifa (Israel) Faculty of Social Welfare and Health Sciences, said in an interview. “But it’s also very interesting that when we looked at frequency of use in the group that used medical cannabis, individuals who had more frequent use also had poorer sleep in the long term.
“This suggests that while cannabis may improve overall sleep, it’s also possible that there is a tolerance that develops with either very frequent or long-term use,” she added.
The study was published online Jan. 20 in BMJ Supportive and Palliative Care.
A common problem
Estimates suggest chronic pain affects up to 37% of adults in the developed world. Individuals who suffer chronic pain often experience comorbid insomnia, which includes difficulty initiating sleep, sleep disruption, and early morning wakening.
For its part, medical cannabis to treat chronic pain symptoms and manage sleep problems has been widely reported as a prime motivation for medical cannabis use. Indeed, previous studies have concluded that the endocannabinoid system plays a role in sleep regulation, including sleep promotion and maintenance.
In recent years, investigators have reported the beneficial effects of medical cannabis for sleep. Nevertheless, some preclinical research has also concluded that chronic administration of tetrahydrocannabinol may result in tolerance to the sleep-enhancing effects of cannabis.
With that in mind, the researchers set out to examine the potential impact of whole-plant medicinal cannabis on sleep problems experienced by middle-aged patients suffering from chronic pain.
“People are self-reporting that they’re using cannabis for sleep and that it helps, but as we know, just because people are reporting that it works doesn’t mean that it will hold up in research,” Dr. Sznitman said.
The study included 128 individuals (mean age, 61±6 years; 51% females) with chronic neuropathic pain: 66 were medical cannabis users and 62 were not.
Three indicators of insomnia were measured using the 7-point Likert scale to assess issues with sleep initiation and maintenance.
In addition, investigators collected sociodemographic information, as well as data on daily consumption of tobacco, frequency of alcohol use, and pain severity. Finally, they collected patient data on the use of sleep-aid medications during the past month as well as tricyclic antidepressant use.
Frequent use, more sleep problems?
On average, medical cannabis users were 3 years younger than their nonusing counterparts (mean age, 60±6 vs. 63±6 years, respectively, P = .003) and more likely to be male (58% vs 40%, respectively, P = .038). Otherwise, the two groups were comparable.
Medical cannabis users reported taking the drug for an average of 4 years, at an average quantity of 31 g per month. The primary mode of administration was smoking (68.6%), followed by oil extracts (21.4%) and vaporization (20%).
Results showed that, of the total sample, 24.1% reported always waking up early and not falling back to sleep, 20.2% reported always having difficulty falling asleep, and 27.2% reported always waking up during the night.
After adjusting for patient age, sex, pain level, and use of sleep medications and antidepressants, medical cannabis use was associated with fewer problems with waking up at night, compared with nonmedical cannabis use. No differences were found between groups with respect to problems falling asleep or waking up early without being able to fall back to sleep, Dr. Sznitman and associates reported.
The final analysis of a subsample of patients that only included medical cannabis users showed frequency of medical cannabis use was associated with sleep problems, they said.
Specifically, more frequent cannabis use was associated with more problems related to waking up at night, as well as problems falling asleep.
Sleep problems associated with frequent medical cannabis use may signal the development of tolerance to the agent. However, frequent users of medical cannabis also maybsuffer pain or other comorbidities, which, in turn, may be linked to more sleep problems.
Either way, Dr. Sznitman said the study might open the door to another treatment option for patients suffering from chronic pain who struggle with sleep.
“If future research shows that the effect of medical cannabis on sleep is a consistent one, then we may be adding a new therapy for sleep problems, which are huge in society and especially in chronic pain patients,” she said.
Early days
Commenting on the findings in an interview, Ryan G. Vandrey, PhD, who was not involved in the study, said the findings are in line with previous research.
“I think the results make sense with respect to the data I’ve collected and from what I’ve seen,” said Dr. Vandrey, associate professor of psychiatry and behavioral sciences at Johns Hopkins Medicine in Baltimore.
“We typically only want to use sleep medications for short periods of time,” he continued. “When you think about recommended prescribing practices for any hypnotic medication, it’s usually short term, 2 weeks or less. Longer-term use often leads to tolerance, dependence, and withdrawal symptoms when the medication is stopped, which leads to an exacerbation of disordered sleep,” Dr. Vandrey said.
Nevertheless, he urged caution when interpreting the results.
“I think the study warrants caution about long-term daily use of cannabinoids with respect to sleep,” he said. “But we need more detailed evaluations, as the trial wasn’t testing a defined product, specific dose, or dose regimen.
“In addition, this was all done in the context of people with chronic pain and not treating disordered sleep or insomnia, but the study highlights the importance of recognizing that long-term chronic use of cannabis is not likely to fully resolve sleep problems.”
Dr. Sznitman agreed that the research is still in its very early stages.
“We’re still far from saying we have the evidence to support the use of medical cannabis for sleep,” she said. “For in the end it was just a cross-sectional, observational study, so we cannot say anything about cause and effect. But if these results pan out, they could be far-reaching and exciting.”
The study was funded by the University of Haifa and Rambam Hospital in Israel, and by the Evelyn Lipper Foundation. Dr. Sznitman and Dr. Vandrey have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
, new research shows.
Investigators found whole-plant medical cannabis use was associated with fewer problems with respect to waking up at night, but they also found that frequent medical cannabis use was associated with more problems initiating and maintaining sleep.
“Cannabis may improve overall sleep in the short term,” study investigator Sharon Sznitman, PhD, University of Haifa (Israel) Faculty of Social Welfare and Health Sciences, said in an interview. “But it’s also very interesting that when we looked at frequency of use in the group that used medical cannabis, individuals who had more frequent use also had poorer sleep in the long term.
“This suggests that while cannabis may improve overall sleep, it’s also possible that there is a tolerance that develops with either very frequent or long-term use,” she added.
The study was published online Jan. 20 in BMJ Supportive and Palliative Care.
A common problem
Estimates suggest chronic pain affects up to 37% of adults in the developed world. Individuals who suffer chronic pain often experience comorbid insomnia, which includes difficulty initiating sleep, sleep disruption, and early morning wakening.
For its part, medical cannabis to treat chronic pain symptoms and manage sleep problems has been widely reported as a prime motivation for medical cannabis use. Indeed, previous studies have concluded that the endocannabinoid system plays a role in sleep regulation, including sleep promotion and maintenance.
In recent years, investigators have reported the beneficial effects of medical cannabis for sleep. Nevertheless, some preclinical research has also concluded that chronic administration of tetrahydrocannabinol may result in tolerance to the sleep-enhancing effects of cannabis.
With that in mind, the researchers set out to examine the potential impact of whole-plant medicinal cannabis on sleep problems experienced by middle-aged patients suffering from chronic pain.
“People are self-reporting that they’re using cannabis for sleep and that it helps, but as we know, just because people are reporting that it works doesn’t mean that it will hold up in research,” Dr. Sznitman said.
The study included 128 individuals (mean age, 61±6 years; 51% females) with chronic neuropathic pain: 66 were medical cannabis users and 62 were not.
Three indicators of insomnia were measured using the 7-point Likert scale to assess issues with sleep initiation and maintenance.
In addition, investigators collected sociodemographic information, as well as data on daily consumption of tobacco, frequency of alcohol use, and pain severity. Finally, they collected patient data on the use of sleep-aid medications during the past month as well as tricyclic antidepressant use.
Frequent use, more sleep problems?
On average, medical cannabis users were 3 years younger than their nonusing counterparts (mean age, 60±6 vs. 63±6 years, respectively, P = .003) and more likely to be male (58% vs 40%, respectively, P = .038). Otherwise, the two groups were comparable.
Medical cannabis users reported taking the drug for an average of 4 years, at an average quantity of 31 g per month. The primary mode of administration was smoking (68.6%), followed by oil extracts (21.4%) and vaporization (20%).
Results showed that, of the total sample, 24.1% reported always waking up early and not falling back to sleep, 20.2% reported always having difficulty falling asleep, and 27.2% reported always waking up during the night.
After adjusting for patient age, sex, pain level, and use of sleep medications and antidepressants, medical cannabis use was associated with fewer problems with waking up at night, compared with nonmedical cannabis use. No differences were found between groups with respect to problems falling asleep or waking up early without being able to fall back to sleep, Dr. Sznitman and associates reported.
The final analysis of a subsample of patients that only included medical cannabis users showed frequency of medical cannabis use was associated with sleep problems, they said.
Specifically, more frequent cannabis use was associated with more problems related to waking up at night, as well as problems falling asleep.
Sleep problems associated with frequent medical cannabis use may signal the development of tolerance to the agent. However, frequent users of medical cannabis also maybsuffer pain or other comorbidities, which, in turn, may be linked to more sleep problems.
Either way, Dr. Sznitman said the study might open the door to another treatment option for patients suffering from chronic pain who struggle with sleep.
“If future research shows that the effect of medical cannabis on sleep is a consistent one, then we may be adding a new therapy for sleep problems, which are huge in society and especially in chronic pain patients,” she said.
Early days
Commenting on the findings in an interview, Ryan G. Vandrey, PhD, who was not involved in the study, said the findings are in line with previous research.
“I think the results make sense with respect to the data I’ve collected and from what I’ve seen,” said Dr. Vandrey, associate professor of psychiatry and behavioral sciences at Johns Hopkins Medicine in Baltimore.
“We typically only want to use sleep medications for short periods of time,” he continued. “When you think about recommended prescribing practices for any hypnotic medication, it’s usually short term, 2 weeks or less. Longer-term use often leads to tolerance, dependence, and withdrawal symptoms when the medication is stopped, which leads to an exacerbation of disordered sleep,” Dr. Vandrey said.
Nevertheless, he urged caution when interpreting the results.
“I think the study warrants caution about long-term daily use of cannabinoids with respect to sleep,” he said. “But we need more detailed evaluations, as the trial wasn’t testing a defined product, specific dose, or dose regimen.
“In addition, this was all done in the context of people with chronic pain and not treating disordered sleep or insomnia, but the study highlights the importance of recognizing that long-term chronic use of cannabis is not likely to fully resolve sleep problems.”
Dr. Sznitman agreed that the research is still in its very early stages.
“We’re still far from saying we have the evidence to support the use of medical cannabis for sleep,” she said. “For in the end it was just a cross-sectional, observational study, so we cannot say anything about cause and effect. But if these results pan out, they could be far-reaching and exciting.”
The study was funded by the University of Haifa and Rambam Hospital in Israel, and by the Evelyn Lipper Foundation. Dr. Sznitman and Dr. Vandrey have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
FROM BMJ SUPPORTIVE AND PALLIATIVE CARE
Wuhan virus: What clinicians need to know
As the Wuhan coronavirus story unfolds, , according to infectious disease experts.

“We are asking that of everyone with fever and respiratory symptoms who comes to our clinics, hospital, or emergency room. It’s a powerful screening tool,” said William Schaffner, MD, professor of preventive medicine and infectious diseases at Vanderbilt University Medical Center, Nashville, Tenn.
In addition to fever, common signs of infection include cough, shortness of breath, and breathing difficulties. Some patients have had diarrhea, vomiting, and other gastrointestinal symptoms. In more severe cases, infection can cause pneumonia, severe acute respiratory syndrome, kidney failure, and death. The incubation period appears to be up to 2 weeks, according to the World Health Organization (WHO).
If patients exhibit symptoms and either they or a close contact has returned from China recently, take standard airborne precautions and send specimens – a serum sample, oral and nasal pharyngeal swabs, and lower respiratory tract specimens if available – to the local health department, which will forward them to the Centers for Disease Control and Prevention (CDC) for testing. Turnaround time is 24-48 hours.
The 2019 Novel Coronavirus (2019-nCoV), identified as the cause of an outbreak of respiratory illness first detected in December in association with a live animal market in Wuhan, China, has been implicated in almost 2,000 cases and 56 deaths in that country. Cases have been reported in 13 countries besides China. Five cases of 2019-nCoV infection have been confirmed in the United States, all in people recently returned from Wuhan. As the virus spreads in China, however, it’s almost certain more cases will show up in the United States. Travel history is key, Dr. Schaffner and others said.
Plan and rehearse
The first step to prepare is to use the CDC’s Interim Guidance for Healthcare Professionals to make a written plan specific to your practice to respond to a potential case. The plan must include notifying the local health department, the CDC liaison for testing, and tracking down patient contacts.
“It’s not good enough to just download CDC’s guidance; use it to make your own local plan and know what to do 24/7,” said Daniel Lucey, MD, an infectious disease expert at Georgetown University Medical Center, Washington, D.C.
“Know who is on call at the health department on weekends and nights,” he said. Know where the patient is going to be isolated; figure out what to do if there’s more than one, and tests come back positive. Have masks on hand, and rehearse the response. “Make a coronavirus team, and absolutely have the nurses involved,” as well as other providers who may come into contact with a case, he added.
“You want to be able to do as well as your counterparts in Washington state and Chicago,” where the first two U.S. cases emerged. “They were prepared. They knew what to do,” Dr. Lucey said.
Those first two U.S. patients – a man in Everett, Wash., and a Chicago woman – developed symptoms after returning from Wuhan, a city of 11 million just over 400 miles inland from the port city of Shanghai. On Jan. 26 three more cases were confirmed by the CDC, two in California and one in Arizona, and each had recently traveled to Wuhan. All five patients remain hospitalized, and there’s no evidence they spread the infection further. There is also no evidence of human-to-human transmission of other cases exported from China to any other countries, according to the WHO.
WHO declined to declare a global health emergency – a Public Health Emergency of International Concern, in its parlance – on Jan. 23. The step would have triggered travel and trade restrictions in member states, including the United States. For now, at least, the group said it wasn’t warranted at this point.
Fatality rates
The focus right now is China. The outbreak has spread beyond Wuhan to other parts of the country, and there’s evidence of fourth-generation spread.
Transportation into and out of Wuhan and other cities has been curtailed, Lunar New Year festivals have been canceled, and the Shanghai Disneyland has been closed, among other measures taken by Chinese officials.
The government could be taking drastic measures in part to prevent the public criticism it took in the early 2000’s for the delayed response and lack of transparency during the global outbreak of another wildlife market coronavirus epidemic, severe acute respiratory syndrome (SARS). In a press conference Jan. 22, WHO officials commended the government’s containment efforts but did not say they recommended them.
According to WHO, serious cases in China have mostly been in people over 40 years old with significant comorbidities and have skewed towards men. Spread seems to be limited to family members, health care providers, and other close contacts, probably by respiratory droplets. If that pattern holds, WHO officials said, the outbreak is containable.
The fatality rate appears to be around 3%, a good deal lower than the 10% reported for SARS and much lower than the nearly 40% reported for Middle East respiratory syndrome (MERS), another recent coronavirus mutation from the animal trade.
The Wuhan virus fatality rate might drop as milder cases are detected and added to the denominator. “It definitely appears to be less severe than SARS and MERS,” said Amesh Adalja, MD, an infectious disease physician in Pittsburgh and emerging infectious disease researcher at Johns Hopkins University, Baltimore.
SARS: Lessons learned
In general, the world is much better equipped for coronavirus outbreaks than when SARS, in particular, emerged in 2003.
WHO officials in their press conference lauded China for it openness with the current outbreak, and for isolating and sequencing the virus immediately, which gave the world a diagnostic test in the first days of the outbreak, something that wasn’t available for SARS. China and other countries also are cooperating and working closely to contain the Wuhan virus.
“What we know today might change tomorrow, so we have to keep tuned in to new information, but we learned a lot from SARS,” Dr. Shaffner said. Overall, it’s likely “the impact on the United States of this new coronavirus is going to be trivial,” he predicted.
Dr. Lucey, however, recalled that the SARS outbreak in Toronto in 2003 started with one missed case. A woman returned asymptomatic from Hong Kong and spread the infection to her family members before she died. Her cause of death wasn’t immediately recognized, nor was the reason her family members were sick, since they hadn’t been to Hong Kong recently.
The infection ultimately spread to more than 200 people, about half of them health care workers. A few people died.
If a virus is sufficiently contagious, “it just takes one. You don’t want to be the one who misses that first patient,” Dr. Lucey said.
Currently, there are no antivirals or vaccines for coronaviruses; researchers are working on both, but for now, care is supportive.
This article was updated with new case numbers on 1/26/20.
As the Wuhan coronavirus story unfolds, , according to infectious disease experts.

“We are asking that of everyone with fever and respiratory symptoms who comes to our clinics, hospital, or emergency room. It’s a powerful screening tool,” said William Schaffner, MD, professor of preventive medicine and infectious diseases at Vanderbilt University Medical Center, Nashville, Tenn.
In addition to fever, common signs of infection include cough, shortness of breath, and breathing difficulties. Some patients have had diarrhea, vomiting, and other gastrointestinal symptoms. In more severe cases, infection can cause pneumonia, severe acute respiratory syndrome, kidney failure, and death. The incubation period appears to be up to 2 weeks, according to the World Health Organization (WHO).
If patients exhibit symptoms and either they or a close contact has returned from China recently, take standard airborne precautions and send specimens – a serum sample, oral and nasal pharyngeal swabs, and lower respiratory tract specimens if available – to the local health department, which will forward them to the Centers for Disease Control and Prevention (CDC) for testing. Turnaround time is 24-48 hours.
The 2019 Novel Coronavirus (2019-nCoV), identified as the cause of an outbreak of respiratory illness first detected in December in association with a live animal market in Wuhan, China, has been implicated in almost 2,000 cases and 56 deaths in that country. Cases have been reported in 13 countries besides China. Five cases of 2019-nCoV infection have been confirmed in the United States, all in people recently returned from Wuhan. As the virus spreads in China, however, it’s almost certain more cases will show up in the United States. Travel history is key, Dr. Schaffner and others said.
Plan and rehearse
The first step to prepare is to use the CDC’s Interim Guidance for Healthcare Professionals to make a written plan specific to your practice to respond to a potential case. The plan must include notifying the local health department, the CDC liaison for testing, and tracking down patient contacts.
“It’s not good enough to just download CDC’s guidance; use it to make your own local plan and know what to do 24/7,” said Daniel Lucey, MD, an infectious disease expert at Georgetown University Medical Center, Washington, D.C.
“Know who is on call at the health department on weekends and nights,” he said. Know where the patient is going to be isolated; figure out what to do if there’s more than one, and tests come back positive. Have masks on hand, and rehearse the response. “Make a coronavirus team, and absolutely have the nurses involved,” as well as other providers who may come into contact with a case, he added.
“You want to be able to do as well as your counterparts in Washington state and Chicago,” where the first two U.S. cases emerged. “They were prepared. They knew what to do,” Dr. Lucey said.
Those first two U.S. patients – a man in Everett, Wash., and a Chicago woman – developed symptoms after returning from Wuhan, a city of 11 million just over 400 miles inland from the port city of Shanghai. On Jan. 26 three more cases were confirmed by the CDC, two in California and one in Arizona, and each had recently traveled to Wuhan. All five patients remain hospitalized, and there’s no evidence they spread the infection further. There is also no evidence of human-to-human transmission of other cases exported from China to any other countries, according to the WHO.
WHO declined to declare a global health emergency – a Public Health Emergency of International Concern, in its parlance – on Jan. 23. The step would have triggered travel and trade restrictions in member states, including the United States. For now, at least, the group said it wasn’t warranted at this point.
Fatality rates
The focus right now is China. The outbreak has spread beyond Wuhan to other parts of the country, and there’s evidence of fourth-generation spread.
Transportation into and out of Wuhan and other cities has been curtailed, Lunar New Year festivals have been canceled, and the Shanghai Disneyland has been closed, among other measures taken by Chinese officials.
The government could be taking drastic measures in part to prevent the public criticism it took in the early 2000’s for the delayed response and lack of transparency during the global outbreak of another wildlife market coronavirus epidemic, severe acute respiratory syndrome (SARS). In a press conference Jan. 22, WHO officials commended the government’s containment efforts but did not say they recommended them.
According to WHO, serious cases in China have mostly been in people over 40 years old with significant comorbidities and have skewed towards men. Spread seems to be limited to family members, health care providers, and other close contacts, probably by respiratory droplets. If that pattern holds, WHO officials said, the outbreak is containable.
The fatality rate appears to be around 3%, a good deal lower than the 10% reported for SARS and much lower than the nearly 40% reported for Middle East respiratory syndrome (MERS), another recent coronavirus mutation from the animal trade.
The Wuhan virus fatality rate might drop as milder cases are detected and added to the denominator. “It definitely appears to be less severe than SARS and MERS,” said Amesh Adalja, MD, an infectious disease physician in Pittsburgh and emerging infectious disease researcher at Johns Hopkins University, Baltimore.
SARS: Lessons learned
In general, the world is much better equipped for coronavirus outbreaks than when SARS, in particular, emerged in 2003.
WHO officials in their press conference lauded China for it openness with the current outbreak, and for isolating and sequencing the virus immediately, which gave the world a diagnostic test in the first days of the outbreak, something that wasn’t available for SARS. China and other countries also are cooperating and working closely to contain the Wuhan virus.
“What we know today might change tomorrow, so we have to keep tuned in to new information, but we learned a lot from SARS,” Dr. Shaffner said. Overall, it’s likely “the impact on the United States of this new coronavirus is going to be trivial,” he predicted.
Dr. Lucey, however, recalled that the SARS outbreak in Toronto in 2003 started with one missed case. A woman returned asymptomatic from Hong Kong and spread the infection to her family members before she died. Her cause of death wasn’t immediately recognized, nor was the reason her family members were sick, since they hadn’t been to Hong Kong recently.
The infection ultimately spread to more than 200 people, about half of them health care workers. A few people died.
If a virus is sufficiently contagious, “it just takes one. You don’t want to be the one who misses that first patient,” Dr. Lucey said.
Currently, there are no antivirals or vaccines for coronaviruses; researchers are working on both, but for now, care is supportive.
This article was updated with new case numbers on 1/26/20.
As the Wuhan coronavirus story unfolds, , according to infectious disease experts.

“We are asking that of everyone with fever and respiratory symptoms who comes to our clinics, hospital, or emergency room. It’s a powerful screening tool,” said William Schaffner, MD, professor of preventive medicine and infectious diseases at Vanderbilt University Medical Center, Nashville, Tenn.
In addition to fever, common signs of infection include cough, shortness of breath, and breathing difficulties. Some patients have had diarrhea, vomiting, and other gastrointestinal symptoms. In more severe cases, infection can cause pneumonia, severe acute respiratory syndrome, kidney failure, and death. The incubation period appears to be up to 2 weeks, according to the World Health Organization (WHO).
If patients exhibit symptoms and either they or a close contact has returned from China recently, take standard airborne precautions and send specimens – a serum sample, oral and nasal pharyngeal swabs, and lower respiratory tract specimens if available – to the local health department, which will forward them to the Centers for Disease Control and Prevention (CDC) for testing. Turnaround time is 24-48 hours.
The 2019 Novel Coronavirus (2019-nCoV), identified as the cause of an outbreak of respiratory illness first detected in December in association with a live animal market in Wuhan, China, has been implicated in almost 2,000 cases and 56 deaths in that country. Cases have been reported in 13 countries besides China. Five cases of 2019-nCoV infection have been confirmed in the United States, all in people recently returned from Wuhan. As the virus spreads in China, however, it’s almost certain more cases will show up in the United States. Travel history is key, Dr. Schaffner and others said.
Plan and rehearse
The first step to prepare is to use the CDC’s Interim Guidance for Healthcare Professionals to make a written plan specific to your practice to respond to a potential case. The plan must include notifying the local health department, the CDC liaison for testing, and tracking down patient contacts.
“It’s not good enough to just download CDC’s guidance; use it to make your own local plan and know what to do 24/7,” said Daniel Lucey, MD, an infectious disease expert at Georgetown University Medical Center, Washington, D.C.
“Know who is on call at the health department on weekends and nights,” he said. Know where the patient is going to be isolated; figure out what to do if there’s more than one, and tests come back positive. Have masks on hand, and rehearse the response. “Make a coronavirus team, and absolutely have the nurses involved,” as well as other providers who may come into contact with a case, he added.
“You want to be able to do as well as your counterparts in Washington state and Chicago,” where the first two U.S. cases emerged. “They were prepared. They knew what to do,” Dr. Lucey said.
Those first two U.S. patients – a man in Everett, Wash., and a Chicago woman – developed symptoms after returning from Wuhan, a city of 11 million just over 400 miles inland from the port city of Shanghai. On Jan. 26 three more cases were confirmed by the CDC, two in California and one in Arizona, and each had recently traveled to Wuhan. All five patients remain hospitalized, and there’s no evidence they spread the infection further. There is also no evidence of human-to-human transmission of other cases exported from China to any other countries, according to the WHO.
WHO declined to declare a global health emergency – a Public Health Emergency of International Concern, in its parlance – on Jan. 23. The step would have triggered travel and trade restrictions in member states, including the United States. For now, at least, the group said it wasn’t warranted at this point.
Fatality rates
The focus right now is China. The outbreak has spread beyond Wuhan to other parts of the country, and there’s evidence of fourth-generation spread.
Transportation into and out of Wuhan and other cities has been curtailed, Lunar New Year festivals have been canceled, and the Shanghai Disneyland has been closed, among other measures taken by Chinese officials.
The government could be taking drastic measures in part to prevent the public criticism it took in the early 2000’s for the delayed response and lack of transparency during the global outbreak of another wildlife market coronavirus epidemic, severe acute respiratory syndrome (SARS). In a press conference Jan. 22, WHO officials commended the government’s containment efforts but did not say they recommended them.
According to WHO, serious cases in China have mostly been in people over 40 years old with significant comorbidities and have skewed towards men. Spread seems to be limited to family members, health care providers, and other close contacts, probably by respiratory droplets. If that pattern holds, WHO officials said, the outbreak is containable.
The fatality rate appears to be around 3%, a good deal lower than the 10% reported for SARS and much lower than the nearly 40% reported for Middle East respiratory syndrome (MERS), another recent coronavirus mutation from the animal trade.
The Wuhan virus fatality rate might drop as milder cases are detected and added to the denominator. “It definitely appears to be less severe than SARS and MERS,” said Amesh Adalja, MD, an infectious disease physician in Pittsburgh and emerging infectious disease researcher at Johns Hopkins University, Baltimore.
SARS: Lessons learned
In general, the world is much better equipped for coronavirus outbreaks than when SARS, in particular, emerged in 2003.
WHO officials in their press conference lauded China for it openness with the current outbreak, and for isolating and sequencing the virus immediately, which gave the world a diagnostic test in the first days of the outbreak, something that wasn’t available for SARS. China and other countries also are cooperating and working closely to contain the Wuhan virus.
“What we know today might change tomorrow, so we have to keep tuned in to new information, but we learned a lot from SARS,” Dr. Shaffner said. Overall, it’s likely “the impact on the United States of this new coronavirus is going to be trivial,” he predicted.
Dr. Lucey, however, recalled that the SARS outbreak in Toronto in 2003 started with one missed case. A woman returned asymptomatic from Hong Kong and spread the infection to her family members before she died. Her cause of death wasn’t immediately recognized, nor was the reason her family members were sick, since they hadn’t been to Hong Kong recently.
The infection ultimately spread to more than 200 people, about half of them health care workers. A few people died.
If a virus is sufficiently contagious, “it just takes one. You don’t want to be the one who misses that first patient,” Dr. Lucey said.
Currently, there are no antivirals or vaccines for coronaviruses; researchers are working on both, but for now, care is supportive.
This article was updated with new case numbers on 1/26/20.
Catheter cryoablation effective for persistent AFib in pivotal trial
NATIONAL HARBOR, MD. – , setting the stage for the device to become the first to receive U.S. labeling for catheter ablation in this atrial fibrillation population.
The Arctic Front Advance cryoballoon, used on 165 patients with persistent atrial fibrillation (AFib) enrolled in the trial, produced a 55% rate of treatment success, including freedom from recurrent AFib during 12 months of follow-up, and produced one prespecified serious adverse event in the primary safety endpoint.
Both results easily surpassed the prespecified performance goals set by negotiation with the FDA, Hugh Calkins, MD, said at the annual International AF Symposium. The trial design included no control group and instead assessed safety and efficacy against prespecified standards set by the regulatory agency.
The cryoballoon “showed excellent performance. I don’t see how this could possibly be turned down by the FDA,” said Dr. Calkins, professor of medicine and director of the cardiac arrhythmia service at Johns Hopkins Medicine in Baltimore.
Cardiac electrophysiologists have for years routinely performed catheter ablation procedures on patients with persistent AFib even though the devices, based on ablation by radiofrequency or by chilling, have been labeled for use only in treating patients with paroxysmal AFib. Although this off-label use has not resulted in any problems with health insurance coverage, Dr. Calkins said, it has kept manufacturers from marketing their ablation devices for use in persistent AFib patients.
If the reported data result in labeling for the tested cryoballoon for persistant AFib patients, “it will have a big impact,” he predicted. “People have used cryoballoons for ablating persistent AFib for years, but this would put more fuel in the fire, both the [very positive] safety and efficacy data, and getting an FDA label, which is worth a lot,” he said in an interview.
But Dr. Calkins stopped short of anticipating that the results would convince operators experienced and focused on performing radiofrequency ablation to switch to cryo devices for treating persistent AFib patients. “People are pretty stuck in their ways,” he noted, and reports are expected soon from pivotal trials that are now testing various radiofrequency devices, as well as other types of cryo devices, in persistent AFib patients, so the range of device options labeled for this population may soon grow even more.
The STOP Persistent AF trial ran at 25 sites in the United States, Canada, and Japan during March 2017-August 2019, and included 165 adults with symptomatic, persistent AFib who had not responded to at least one antiarrhythmic drug and had a history of AFib episodes lasting at least 7 days but with no episodes persisting for 6 months or longer. The study excluded patients with prior ablation or left atrial surgery, a recent cerebrovascular event, substantially reduced left ventricular function, or substantial left atrial enlargement.
The enrolled patients were an average 65 years of age and 70% were men. Patients had been diagnosed with paroxysmal AFib an average of 5 years before study entry and with persistent AFib a little over 6 months before entry. The most recent AFib episode of enrolled patients averaged about 60 days, on average they had been unsuccessfully treated with just over one antiarrhythmic drug, and on average they had previously undergone about two cardioversions, after which their arrhythmia recurred.
The primary efficacy endpoint – a 55% rate of acute procedural success plus freedom from AFib recurrence during the 9 months following a 90-day blanking period immediately after ablation plus no added or increased antiarrhythmic drugs – significantly exceeded the prespecified performance goal of a 40% rate, Dr. Calkins reported. The study used the standard measure of recurrence as any 30-second or longer AFib episode detected during a weekly ECG telemonitoring session or during 48-hour ambulatory ECG monitoring at 6- and 12-month follow-ups or during in-office 12-lead ECG assessment at 3-, 6-, and 12-month follow-up. Twelve-month follow-up occurred for 145 of the enrolled patients.
The only prespecified primary safety event was one episode of cardiac perforation, which occurred during a repeat procedure. This rate of one safety event among 165 patients (0.6%) fell well within the prespecified safety performance goal of no more than 13%. In addition to this perforation, five additional serious adverse events (3%) occurred that were attributable to the cryoballoon treatment, including two cases of vascular pseudoaneurysm, one puncture-site hematoma, one case of pericarditis, and one episode of atrial tachycardia. Four additional serious adverse events occurred that were attributable to the ablation procedure (one acute cardiac failure, one postprocedure ileus, one respiratory failure, and one urinary tract infection), for an overall serious event rate of 5%.
During follow-up, 13% of patients had a repeat ablation procedure, following an initial ablation limited to pulmonary-vein isolation (PVI). The overall rate of 1-year efficacy, including the relatively low rate of need for redo ablation, “are an impressive endorsement of a PVI-only strategy” for initial ablation, Dr. Calkins said. “I’m a strong believer in PVI only for the first ablation for both paroxysmal and persistent AFib.”
He also noted that the 30-second threshold for scoring recurrent arrhythmia episodes following the 90-day blanking period after ablation was a very conservative measure of treatment failure, but it continues to define recurrence in this and many other current AFib ablation studies because it is the historical criteria for measuring ablation success or failure. It is especially important to maintain this criteria in a study that relied on prespecified performance criteria rather than a control arm for judging efficacy, Dr. Calkins said.
The study also included three quality-of-life measures. Patient scores on the Atrial Fibrillation Effect on Quality-of-Life (AFEQT) questionnaire rose by an average of nearly 26 points from baseline to 12 months, a statistically significant and clinically meaningful increase. Scores on both the physical and mental domains of the Short Form-12 (SF-12) improved from baseline by an average of about five points on each subscale, also statistically significant and clinically meaningful changes. Patients also reported statistically significant and in some cases substantial reductions in the prevalence rates of each of five different AFib symptoms: dizziness, dyspnea, fatigue, palpitations, and rapid heartbeat.
The study was funded by Medtronic, the company that markets the tested cryoballoon (Arctic Front Advance). Dr. Calkins has been a consultant to and has received honoraria from Medtronic, as well as from Abbott, AtriCure, Boehringer Ingelheim, Boston Scientific, and Johnson & Johnson.
NATIONAL HARBOR, MD. – , setting the stage for the device to become the first to receive U.S. labeling for catheter ablation in this atrial fibrillation population.
The Arctic Front Advance cryoballoon, used on 165 patients with persistent atrial fibrillation (AFib) enrolled in the trial, produced a 55% rate of treatment success, including freedom from recurrent AFib during 12 months of follow-up, and produced one prespecified serious adverse event in the primary safety endpoint.
Both results easily surpassed the prespecified performance goals set by negotiation with the FDA, Hugh Calkins, MD, said at the annual International AF Symposium. The trial design included no control group and instead assessed safety and efficacy against prespecified standards set by the regulatory agency.
The cryoballoon “showed excellent performance. I don’t see how this could possibly be turned down by the FDA,” said Dr. Calkins, professor of medicine and director of the cardiac arrhythmia service at Johns Hopkins Medicine in Baltimore.
Cardiac electrophysiologists have for years routinely performed catheter ablation procedures on patients with persistent AFib even though the devices, based on ablation by radiofrequency or by chilling, have been labeled for use only in treating patients with paroxysmal AFib. Although this off-label use has not resulted in any problems with health insurance coverage, Dr. Calkins said, it has kept manufacturers from marketing their ablation devices for use in persistent AFib patients.
If the reported data result in labeling for the tested cryoballoon for persistant AFib patients, “it will have a big impact,” he predicted. “People have used cryoballoons for ablating persistent AFib for years, but this would put more fuel in the fire, both the [very positive] safety and efficacy data, and getting an FDA label, which is worth a lot,” he said in an interview.
But Dr. Calkins stopped short of anticipating that the results would convince operators experienced and focused on performing radiofrequency ablation to switch to cryo devices for treating persistent AFib patients. “People are pretty stuck in their ways,” he noted, and reports are expected soon from pivotal trials that are now testing various radiofrequency devices, as well as other types of cryo devices, in persistent AFib patients, so the range of device options labeled for this population may soon grow even more.
The STOP Persistent AF trial ran at 25 sites in the United States, Canada, and Japan during March 2017-August 2019, and included 165 adults with symptomatic, persistent AFib who had not responded to at least one antiarrhythmic drug and had a history of AFib episodes lasting at least 7 days but with no episodes persisting for 6 months or longer. The study excluded patients with prior ablation or left atrial surgery, a recent cerebrovascular event, substantially reduced left ventricular function, or substantial left atrial enlargement.
The enrolled patients were an average 65 years of age and 70% were men. Patients had been diagnosed with paroxysmal AFib an average of 5 years before study entry and with persistent AFib a little over 6 months before entry. The most recent AFib episode of enrolled patients averaged about 60 days, on average they had been unsuccessfully treated with just over one antiarrhythmic drug, and on average they had previously undergone about two cardioversions, after which their arrhythmia recurred.
The primary efficacy endpoint – a 55% rate of acute procedural success plus freedom from AFib recurrence during the 9 months following a 90-day blanking period immediately after ablation plus no added or increased antiarrhythmic drugs – significantly exceeded the prespecified performance goal of a 40% rate, Dr. Calkins reported. The study used the standard measure of recurrence as any 30-second or longer AFib episode detected during a weekly ECG telemonitoring session or during 48-hour ambulatory ECG monitoring at 6- and 12-month follow-ups or during in-office 12-lead ECG assessment at 3-, 6-, and 12-month follow-up. Twelve-month follow-up occurred for 145 of the enrolled patients.
The only prespecified primary safety event was one episode of cardiac perforation, which occurred during a repeat procedure. This rate of one safety event among 165 patients (0.6%) fell well within the prespecified safety performance goal of no more than 13%. In addition to this perforation, five additional serious adverse events (3%) occurred that were attributable to the cryoballoon treatment, including two cases of vascular pseudoaneurysm, one puncture-site hematoma, one case of pericarditis, and one episode of atrial tachycardia. Four additional serious adverse events occurred that were attributable to the ablation procedure (one acute cardiac failure, one postprocedure ileus, one respiratory failure, and one urinary tract infection), for an overall serious event rate of 5%.
During follow-up, 13% of patients had a repeat ablation procedure, following an initial ablation limited to pulmonary-vein isolation (PVI). The overall rate of 1-year efficacy, including the relatively low rate of need for redo ablation, “are an impressive endorsement of a PVI-only strategy” for initial ablation, Dr. Calkins said. “I’m a strong believer in PVI only for the first ablation for both paroxysmal and persistent AFib.”
He also noted that the 30-second threshold for scoring recurrent arrhythmia episodes following the 90-day blanking period after ablation was a very conservative measure of treatment failure, but it continues to define recurrence in this and many other current AFib ablation studies because it is the historical criteria for measuring ablation success or failure. It is especially important to maintain this criteria in a study that relied on prespecified performance criteria rather than a control arm for judging efficacy, Dr. Calkins said.
The study also included three quality-of-life measures. Patient scores on the Atrial Fibrillation Effect on Quality-of-Life (AFEQT) questionnaire rose by an average of nearly 26 points from baseline to 12 months, a statistically significant and clinically meaningful increase. Scores on both the physical and mental domains of the Short Form-12 (SF-12) improved from baseline by an average of about five points on each subscale, also statistically significant and clinically meaningful changes. Patients also reported statistically significant and in some cases substantial reductions in the prevalence rates of each of five different AFib symptoms: dizziness, dyspnea, fatigue, palpitations, and rapid heartbeat.
The study was funded by Medtronic, the company that markets the tested cryoballoon (Arctic Front Advance). Dr. Calkins has been a consultant to and has received honoraria from Medtronic, as well as from Abbott, AtriCure, Boehringer Ingelheim, Boston Scientific, and Johnson & Johnson.
NATIONAL HARBOR, MD. – , setting the stage for the device to become the first to receive U.S. labeling for catheter ablation in this atrial fibrillation population.
The Arctic Front Advance cryoballoon, used on 165 patients with persistent atrial fibrillation (AFib) enrolled in the trial, produced a 55% rate of treatment success, including freedom from recurrent AFib during 12 months of follow-up, and produced one prespecified serious adverse event in the primary safety endpoint.
Both results easily surpassed the prespecified performance goals set by negotiation with the FDA, Hugh Calkins, MD, said at the annual International AF Symposium. The trial design included no control group and instead assessed safety and efficacy against prespecified standards set by the regulatory agency.
The cryoballoon “showed excellent performance. I don’t see how this could possibly be turned down by the FDA,” said Dr. Calkins, professor of medicine and director of the cardiac arrhythmia service at Johns Hopkins Medicine in Baltimore.
Cardiac electrophysiologists have for years routinely performed catheter ablation procedures on patients with persistent AFib even though the devices, based on ablation by radiofrequency or by chilling, have been labeled for use only in treating patients with paroxysmal AFib. Although this off-label use has not resulted in any problems with health insurance coverage, Dr. Calkins said, it has kept manufacturers from marketing their ablation devices for use in persistent AFib patients.
If the reported data result in labeling for the tested cryoballoon for persistant AFib patients, “it will have a big impact,” he predicted. “People have used cryoballoons for ablating persistent AFib for years, but this would put more fuel in the fire, both the [very positive] safety and efficacy data, and getting an FDA label, which is worth a lot,” he said in an interview.
But Dr. Calkins stopped short of anticipating that the results would convince operators experienced and focused on performing radiofrequency ablation to switch to cryo devices for treating persistent AFib patients. “People are pretty stuck in their ways,” he noted, and reports are expected soon from pivotal trials that are now testing various radiofrequency devices, as well as other types of cryo devices, in persistent AFib patients, so the range of device options labeled for this population may soon grow even more.
The STOP Persistent AF trial ran at 25 sites in the United States, Canada, and Japan during March 2017-August 2019, and included 165 adults with symptomatic, persistent AFib who had not responded to at least one antiarrhythmic drug and had a history of AFib episodes lasting at least 7 days but with no episodes persisting for 6 months or longer. The study excluded patients with prior ablation or left atrial surgery, a recent cerebrovascular event, substantially reduced left ventricular function, or substantial left atrial enlargement.
The enrolled patients were an average 65 years of age and 70% were men. Patients had been diagnosed with paroxysmal AFib an average of 5 years before study entry and with persistent AFib a little over 6 months before entry. The most recent AFib episode of enrolled patients averaged about 60 days, on average they had been unsuccessfully treated with just over one antiarrhythmic drug, and on average they had previously undergone about two cardioversions, after which their arrhythmia recurred.
The primary efficacy endpoint – a 55% rate of acute procedural success plus freedom from AFib recurrence during the 9 months following a 90-day blanking period immediately after ablation plus no added or increased antiarrhythmic drugs – significantly exceeded the prespecified performance goal of a 40% rate, Dr. Calkins reported. The study used the standard measure of recurrence as any 30-second or longer AFib episode detected during a weekly ECG telemonitoring session or during 48-hour ambulatory ECG monitoring at 6- and 12-month follow-ups or during in-office 12-lead ECG assessment at 3-, 6-, and 12-month follow-up. Twelve-month follow-up occurred for 145 of the enrolled patients.
The only prespecified primary safety event was one episode of cardiac perforation, which occurred during a repeat procedure. This rate of one safety event among 165 patients (0.6%) fell well within the prespecified safety performance goal of no more than 13%. In addition to this perforation, five additional serious adverse events (3%) occurred that were attributable to the cryoballoon treatment, including two cases of vascular pseudoaneurysm, one puncture-site hematoma, one case of pericarditis, and one episode of atrial tachycardia. Four additional serious adverse events occurred that were attributable to the ablation procedure (one acute cardiac failure, one postprocedure ileus, one respiratory failure, and one urinary tract infection), for an overall serious event rate of 5%.
During follow-up, 13% of patients had a repeat ablation procedure, following an initial ablation limited to pulmonary-vein isolation (PVI). The overall rate of 1-year efficacy, including the relatively low rate of need for redo ablation, “are an impressive endorsement of a PVI-only strategy” for initial ablation, Dr. Calkins said. “I’m a strong believer in PVI only for the first ablation for both paroxysmal and persistent AFib.”
He also noted that the 30-second threshold for scoring recurrent arrhythmia episodes following the 90-day blanking period after ablation was a very conservative measure of treatment failure, but it continues to define recurrence in this and many other current AFib ablation studies because it is the historical criteria for measuring ablation success or failure. It is especially important to maintain this criteria in a study that relied on prespecified performance criteria rather than a control arm for judging efficacy, Dr. Calkins said.
The study also included three quality-of-life measures. Patient scores on the Atrial Fibrillation Effect on Quality-of-Life (AFEQT) questionnaire rose by an average of nearly 26 points from baseline to 12 months, a statistically significant and clinically meaningful increase. Scores on both the physical and mental domains of the Short Form-12 (SF-12) improved from baseline by an average of about five points on each subscale, also statistically significant and clinically meaningful changes. Patients also reported statistically significant and in some cases substantial reductions in the prevalence rates of each of five different AFib symptoms: dizziness, dyspnea, fatigue, palpitations, and rapid heartbeat.
The study was funded by Medtronic, the company that markets the tested cryoballoon (Arctic Front Advance). Dr. Calkins has been a consultant to and has received honoraria from Medtronic, as well as from Abbott, AtriCure, Boehringer Ingelheim, Boston Scientific, and Johnson & Johnson.
REPORTING FROM THE AF SYMPOSIUM 2020
Key clinical point: Catheter ablation using a cryoballoon was safe and effective for patients with persistent atrial fibrillation in a pivotal trial.
Major finding: The rate of freedom from treatment failure after 12 months was 55%, significantly exceeding the prespecified performance goal of 40%.
Study details: A multicenter, international study with 165 enrolled patients.
Disclosures: The study was funded by Medtronic, the company that markets the tested cryoballoon (Arctic Front Advance). Dr. Calkins has been a consultant to and has received honoraria from Medtronic, as well as from Abbott, AtriCure, Boehringer Ingelheim, Boston Scientific, and Johnson & Johnson.
EVALI update warns of chemicals in vaping products
A report issued by the Centers for Disease Control and Prevention confirms that 82% of patients presenting with e-cigarette– or vaping product use–associated lung injury (EVALI) used products containing tetrahydrocannabinol (THC).
Another report published in the CDC’s Morbidity and Mortality Weekly Report assessed cases in which the patients reported using only nicotine-containing vaping products.
“As of Jan. 14, 2020, a total of 2,668 hospitalized EVALI cases had been reported to CDC,” based on data from the National Syndromic Surveillance Program (NSSP), wrote Vikram P. Krishnasamy, MD, of the National Center for Injury Prevention and Control at the CDC, Atlanta, and colleagues. Cases have occurred in all 50 states, the District of Columbia, the U.S. Virgin Islands, and Puerto Rico. The age of the patients ranged from 13 to 85 years, with an average age of 24 years; 66% were male, and 73% were non-Hispanic white.
In addition, 57% of the patients reported using any nicotine-containing product, and 14% of these reported use of nicotine products exclusively.
Previous studies have shown that vitamin E acetate is associated with the EVALI outbreak, which peaked during the week of Sept. 15, 2019, with 215 reported hospital admissions, Dr. Krishnasamy and associates noted. “However, evidence is not sufficient to rule out the contribution of other chemicals of concern, including chemicals in either THC- or non-THC–containing products, in some reported EVALI cases,” they said.
The study findings were limited by several factors, including incomplete data on product use, increased reporting of vaping product use at emergency department visits after increased public awareness of risk, and inconsistency in the health care facilities contributing data via the NSSP, the researchers wrote.
The decline in EVALI cases since September 2019 may be related to factors including the rapid public health response to increase awareness of the risks of vaping, and the possible removal of vitamin E acetate as a diluent in THC-containing products, but clinicians and public health professionals should remain on alert for new EVALI cases and continue to discourage the use of THC-containing e-cigarette or vaping products, Dr. Krishnasamy and associates concluded.
Nicotine-only vaping products
In a second report published in MMWR, Isaac Ghinai, MBBS, of the Illinois Department of Public Health and CDC researchers examined characteristics of EVALI patients in Illinois who reported using only nicotine-containing vaping products.
A total of 9 of 121 (7%) EVALI patients surveyed in Illinois reported no indication of THC use. These patients were more likely than those who reported any use of THC-containing products to be female (78% vs. 25%) and aged 45 years and older (33% vs. 2%); P less than .01 in both cases.
In addition, EVALI patients with no indication of THC-containing product use were less likely than THC product users to present with constitutional symptoms (56% vs. 96%) or initial leukocytosis (38% vs. 91%), or to have previously visited an outpatient provider or ED before being hospitalized (25% vs. 80%).
Other presenting characteristics including initial vital signs and lab results, as well as the frequency of severe outcomes such as death or respiratory failure, were not significantly different between users and nonusers of THC-containing vaping products.
The study findings were limited by factors including the use of self-reports, the small sample size, and lack of initial and follow-up interviews for all EVALI patients, the researchers noted. However, the results support the CDC’s recommendation that “persons should not use THC-containing e-cigarette, or vaping, products, particularly those obtained from informal sources such as friends, family members, or from in-person or online dealers,” and should not add vitamin E acetate or other substances to these products, they said.
In addition, users of nicotine-containing e-cigarette or vaping products as an alternative to cigarettes should not return to cigarettes, but should explore other options to help them quit, Dr. Ghinai, and associates said.
The studies were supported by the CDC. The researchers in both studies had no financial conflicts to disclose.
SOURCES: Krishnasamy VP et al. MMWR Morb Mortal Wkly Rep. 17 Jan 2020. doi: 10.15585/mmwr.mm6903e2; Ghinai I et al. MMWR Morb Mortal Wkly Rep. 17 Jan 2020. doi: 10.15585/mmwr.mm6903e1.
A report issued by the Centers for Disease Control and Prevention confirms that 82% of patients presenting with e-cigarette– or vaping product use–associated lung injury (EVALI) used products containing tetrahydrocannabinol (THC).
Another report published in the CDC’s Morbidity and Mortality Weekly Report assessed cases in which the patients reported using only nicotine-containing vaping products.
“As of Jan. 14, 2020, a total of 2,668 hospitalized EVALI cases had been reported to CDC,” based on data from the National Syndromic Surveillance Program (NSSP), wrote Vikram P. Krishnasamy, MD, of the National Center for Injury Prevention and Control at the CDC, Atlanta, and colleagues. Cases have occurred in all 50 states, the District of Columbia, the U.S. Virgin Islands, and Puerto Rico. The age of the patients ranged from 13 to 85 years, with an average age of 24 years; 66% were male, and 73% were non-Hispanic white.
In addition, 57% of the patients reported using any nicotine-containing product, and 14% of these reported use of nicotine products exclusively.
Previous studies have shown that vitamin E acetate is associated with the EVALI outbreak, which peaked during the week of Sept. 15, 2019, with 215 reported hospital admissions, Dr. Krishnasamy and associates noted. “However, evidence is not sufficient to rule out the contribution of other chemicals of concern, including chemicals in either THC- or non-THC–containing products, in some reported EVALI cases,” they said.
The study findings were limited by several factors, including incomplete data on product use, increased reporting of vaping product use at emergency department visits after increased public awareness of risk, and inconsistency in the health care facilities contributing data via the NSSP, the researchers wrote.
The decline in EVALI cases since September 2019 may be related to factors including the rapid public health response to increase awareness of the risks of vaping, and the possible removal of vitamin E acetate as a diluent in THC-containing products, but clinicians and public health professionals should remain on alert for new EVALI cases and continue to discourage the use of THC-containing e-cigarette or vaping products, Dr. Krishnasamy and associates concluded.
Nicotine-only vaping products
In a second report published in MMWR, Isaac Ghinai, MBBS, of the Illinois Department of Public Health and CDC researchers examined characteristics of EVALI patients in Illinois who reported using only nicotine-containing vaping products.
A total of 9 of 121 (7%) EVALI patients surveyed in Illinois reported no indication of THC use. These patients were more likely than those who reported any use of THC-containing products to be female (78% vs. 25%) and aged 45 years and older (33% vs. 2%); P less than .01 in both cases.
In addition, EVALI patients with no indication of THC-containing product use were less likely than THC product users to present with constitutional symptoms (56% vs. 96%) or initial leukocytosis (38% vs. 91%), or to have previously visited an outpatient provider or ED before being hospitalized (25% vs. 80%).
Other presenting characteristics including initial vital signs and lab results, as well as the frequency of severe outcomes such as death or respiratory failure, were not significantly different between users and nonusers of THC-containing vaping products.
The study findings were limited by factors including the use of self-reports, the small sample size, and lack of initial and follow-up interviews for all EVALI patients, the researchers noted. However, the results support the CDC’s recommendation that “persons should not use THC-containing e-cigarette, or vaping, products, particularly those obtained from informal sources such as friends, family members, or from in-person or online dealers,” and should not add vitamin E acetate or other substances to these products, they said.
In addition, users of nicotine-containing e-cigarette or vaping products as an alternative to cigarettes should not return to cigarettes, but should explore other options to help them quit, Dr. Ghinai, and associates said.
The studies were supported by the CDC. The researchers in both studies had no financial conflicts to disclose.
SOURCES: Krishnasamy VP et al. MMWR Morb Mortal Wkly Rep. 17 Jan 2020. doi: 10.15585/mmwr.mm6903e2; Ghinai I et al. MMWR Morb Mortal Wkly Rep. 17 Jan 2020. doi: 10.15585/mmwr.mm6903e1.
A report issued by the Centers for Disease Control and Prevention confirms that 82% of patients presenting with e-cigarette– or vaping product use–associated lung injury (EVALI) used products containing tetrahydrocannabinol (THC).
Another report published in the CDC’s Morbidity and Mortality Weekly Report assessed cases in which the patients reported using only nicotine-containing vaping products.
“As of Jan. 14, 2020, a total of 2,668 hospitalized EVALI cases had been reported to CDC,” based on data from the National Syndromic Surveillance Program (NSSP), wrote Vikram P. Krishnasamy, MD, of the National Center for Injury Prevention and Control at the CDC, Atlanta, and colleagues. Cases have occurred in all 50 states, the District of Columbia, the U.S. Virgin Islands, and Puerto Rico. The age of the patients ranged from 13 to 85 years, with an average age of 24 years; 66% were male, and 73% were non-Hispanic white.
In addition, 57% of the patients reported using any nicotine-containing product, and 14% of these reported use of nicotine products exclusively.
Previous studies have shown that vitamin E acetate is associated with the EVALI outbreak, which peaked during the week of Sept. 15, 2019, with 215 reported hospital admissions, Dr. Krishnasamy and associates noted. “However, evidence is not sufficient to rule out the contribution of other chemicals of concern, including chemicals in either THC- or non-THC–containing products, in some reported EVALI cases,” they said.
The study findings were limited by several factors, including incomplete data on product use, increased reporting of vaping product use at emergency department visits after increased public awareness of risk, and inconsistency in the health care facilities contributing data via the NSSP, the researchers wrote.
The decline in EVALI cases since September 2019 may be related to factors including the rapid public health response to increase awareness of the risks of vaping, and the possible removal of vitamin E acetate as a diluent in THC-containing products, but clinicians and public health professionals should remain on alert for new EVALI cases and continue to discourage the use of THC-containing e-cigarette or vaping products, Dr. Krishnasamy and associates concluded.
Nicotine-only vaping products
In a second report published in MMWR, Isaac Ghinai, MBBS, of the Illinois Department of Public Health and CDC researchers examined characteristics of EVALI patients in Illinois who reported using only nicotine-containing vaping products.
A total of 9 of 121 (7%) EVALI patients surveyed in Illinois reported no indication of THC use. These patients were more likely than those who reported any use of THC-containing products to be female (78% vs. 25%) and aged 45 years and older (33% vs. 2%); P less than .01 in both cases.
In addition, EVALI patients with no indication of THC-containing product use were less likely than THC product users to present with constitutional symptoms (56% vs. 96%) or initial leukocytosis (38% vs. 91%), or to have previously visited an outpatient provider or ED before being hospitalized (25% vs. 80%).
Other presenting characteristics including initial vital signs and lab results, as well as the frequency of severe outcomes such as death or respiratory failure, were not significantly different between users and nonusers of THC-containing vaping products.
The study findings were limited by factors including the use of self-reports, the small sample size, and lack of initial and follow-up interviews for all EVALI patients, the researchers noted. However, the results support the CDC’s recommendation that “persons should not use THC-containing e-cigarette, or vaping, products, particularly those obtained from informal sources such as friends, family members, or from in-person or online dealers,” and should not add vitamin E acetate or other substances to these products, they said.
In addition, users of nicotine-containing e-cigarette or vaping products as an alternative to cigarettes should not return to cigarettes, but should explore other options to help them quit, Dr. Ghinai, and associates said.
The studies were supported by the CDC. The researchers in both studies had no financial conflicts to disclose.
SOURCES: Krishnasamy VP et al. MMWR Morb Mortal Wkly Rep. 17 Jan 2020. doi: 10.15585/mmwr.mm6903e2; Ghinai I et al. MMWR Morb Mortal Wkly Rep. 17 Jan 2020. doi: 10.15585/mmwr.mm6903e1.
FROM MMWR

















