User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
FDA: Cybersecurity vulnerabilities identified in GE Healthcare monitoring devices
The Food and Drug Administration has issued a warning that certain GE Healthcare Clinical Information Central Stations and Telemetry Servers have cybersecurity vulnerabilities that may introduce risk to monitored patients.
silence alarms, generate false alarms, and interfere with alarms of patient monitors connected to these devices, according to an “Urgent Medical Device Correction” letter issued by GE Healthcare in November 2019.
The affected devices are the ApexPro Telemetry Server and CARESCAPE Telemetry Server, the CARESCAPE Central Station (CSCS) version 1, and the CIC Pro Clinical Information Center Central Station version 1. These devices are used in health care facilities for displaying information, such as the patient’s physiological parameters, and for monitoring patient status from a central location in a facility.
No adverse events related to the vulnerabilities have been reported to the FDA. Health care facility staff should update their devices when GE Healthcare issues a software patch that addresses the vulnerability, separate the network connecting patient monitors using affected devices from the rest of the hospital, and use firewalls and other means to minimize the risk of remote or local network attacks.
“The FDA takes reports of cybersecurity vulnerabilities in medical devices seriously and will continue to work with GE Healthcare as the firm develops software patches to correct these vulnerabilities as soon as possible. The FDA will continue to assess new information concerning the vulnerabilities and will keep the public informed if significant new information becomes available,” the FDA said in the Safety Communication.
The Food and Drug Administration has issued a warning that certain GE Healthcare Clinical Information Central Stations and Telemetry Servers have cybersecurity vulnerabilities that may introduce risk to monitored patients.
silence alarms, generate false alarms, and interfere with alarms of patient monitors connected to these devices, according to an “Urgent Medical Device Correction” letter issued by GE Healthcare in November 2019.
The affected devices are the ApexPro Telemetry Server and CARESCAPE Telemetry Server, the CARESCAPE Central Station (CSCS) version 1, and the CIC Pro Clinical Information Center Central Station version 1. These devices are used in health care facilities for displaying information, such as the patient’s physiological parameters, and for monitoring patient status from a central location in a facility.
No adverse events related to the vulnerabilities have been reported to the FDA. Health care facility staff should update their devices when GE Healthcare issues a software patch that addresses the vulnerability, separate the network connecting patient monitors using affected devices from the rest of the hospital, and use firewalls and other means to minimize the risk of remote or local network attacks.
“The FDA takes reports of cybersecurity vulnerabilities in medical devices seriously and will continue to work with GE Healthcare as the firm develops software patches to correct these vulnerabilities as soon as possible. The FDA will continue to assess new information concerning the vulnerabilities and will keep the public informed if significant new information becomes available,” the FDA said in the Safety Communication.
The Food and Drug Administration has issued a warning that certain GE Healthcare Clinical Information Central Stations and Telemetry Servers have cybersecurity vulnerabilities that may introduce risk to monitored patients.
silence alarms, generate false alarms, and interfere with alarms of patient monitors connected to these devices, according to an “Urgent Medical Device Correction” letter issued by GE Healthcare in November 2019.
The affected devices are the ApexPro Telemetry Server and CARESCAPE Telemetry Server, the CARESCAPE Central Station (CSCS) version 1, and the CIC Pro Clinical Information Center Central Station version 1. These devices are used in health care facilities for displaying information, such as the patient’s physiological parameters, and for monitoring patient status from a central location in a facility.
No adverse events related to the vulnerabilities have been reported to the FDA. Health care facility staff should update their devices when GE Healthcare issues a software patch that addresses the vulnerability, separate the network connecting patient monitors using affected devices from the rest of the hospital, and use firewalls and other means to minimize the risk of remote or local network attacks.
“The FDA takes reports of cybersecurity vulnerabilities in medical devices seriously and will continue to work with GE Healthcare as the firm develops software patches to correct these vulnerabilities as soon as possible. The FDA will continue to assess new information concerning the vulnerabilities and will keep the public informed if significant new information becomes available,” the FDA said in the Safety Communication.
New Vascepa indication opens up treatment to millions; “Most significant event since statins”
The newly approved U.S. indication for icosapent ethyl (Vascepa; Amarin) is broadly in line with the entry criteria for the REDUCE-IT trial and includes a large high-risk primary-prevention population, as well as those with established cardiovascular disease (CVD). The drug, thus, could well be used by millions of patients in the United States alone.
The high-dose, purified eicosapentaenoic acid product was approved last week by the Food and Drug Administration for cardiovascular risk reduction among adults already taking maximally tolerated statins with triglyceride levels of 150 mg/dL or higher who have either established CVD or diabetes and two or more additional risk factors for CVD.
The approval is based largely on the REDUCE-IT trial’s finding of a 25% reduction in risk for major adverse cardiovascular events versus placebo. The FDA stated that the approval is the first for an agent with this specific indication.
Noting that it recognizes the need for additional medical treatments for CVD, the FDA says the new approval “will give patients with elevated triglycerides and other important risk factors, including heart disease, stroke, and diabetes, an adjunctive treatment option that can help decrease their risk of cardiovascular events.”
The drug was unanimously recommended for approval by the FDA’s Endocrinologic and Metabolic Drugs Advisory Committee last month. But while the committee all agreed on its use in patients with established CVD, which made up 70% of the REDUCE-IT population, they were divided on whether the indication should be extended to the high-risk primary-prevention population, who made up just 30% of patients in the study.
Nonetheless, the FDA has gone for a broad indication based on the whole REDUCE-IT population.
In a conference call following the approval, Steven Ketchum, PhD, chief scientific officer at Amarin, pointed out that the primary-prevention population stipulated in the new approval differed very slightly from the REDUCE-IT enrollment criteria.
The trial specified that patients with diabetes should be older than 50 with one other cardiovascular risk factor, whereas the approved population is for diabetes and two cardiovascular risk factors. But as these two risk factors are not specified, they could include age, cigarette smoking, hypertension or use of an antihypertensive agent, low HDL cholesterol, high C-reactive protein, body mass index above 25 kg/m2, renal dysfunction, retinopathy, albuminuria, or an ankle branchial index below 0.9, Dr. Ketchum said.
“So while the label asks for two other risk factors, one of these could be age; so we believe the label is actually slightly broader than the REDUCE-IT inclusion criteria, and doctors have been left with significant leeway to decide which risk factors to consider on top of diabetes.”
Deepak Bhatt, MD, the lead investigator of REDUCE-IT, described the Vascepa approval as “the most significant event in the field of cardiovascular prevention since the introduction of statins nearly 3 decades ago.”
He commended the FDA on “a very evidence-based, prescriber-friendly, and most importantly, patient-friendly label,” which he said was in line with guidelines from multiple professional societies that have already incorporated the REDUCE-IT findings for secondary prevention and diabetic primary prevention.
Dr. Bhatt, who is a professor of medicine at Brigham and Women’s Hospital and Harvard Medical School, Boston, said the label essentially matches the REDUCE-IT population.
“The entry criteria for REDUCE-IT was fasting triglycerides greater than or equal to 150 mg/dL, with a 10% variance allowed (giving a minimum triglyceride value of 135 mg/dL). In actuality, we ended up with about 10% of the population with triglycerides between 100 and 150 mg/dL, and they had a similar degree of benefit as those with higher levels,” he reported.
“In the label, the 150 mg/dL does not specify fasting, and in fact many practices have moved away from fasting lipid measurements for the sake of patient comfort,” Dr. Bhatt added. “On average, nonfasting levels are about 50 mg/dL higher, so the label essentially mirrors those we studied, with the FDA applying good common sense and not being overly dogmatic about the exact wording of the trial inclusion criteria.”
No price change foreseen
Vascepa is already on the market for patients with very high triglyceride levels, and the company says it is not increasing the current price of about $300 a month, which is “relatively low, compared to other new breakthrough drugs.” However, it says it expects sales to grow from vastly increased volume based on the new indication.
Dr. Bhatt noted that REDUCE-IT cost-effectiveness data presented at the recent American Heart Association scientific sessions found the drug to be cost saving in the majority of cases. “That is something that is quite rare in cost-effectiveness research,” he said.
“Now, the key challenge is to identify and treat appropriate patients,” Dr. Bhatt noted. He says this task will largely fall on cardiologists, endocrinologists, and primary care physicians, though stroke neurologists, nephrologists, and vascular medicine specialists will also have patients for whom the data are relevant.
“I believe the drug will ultimately be widely prescribed, initially by subspecialists, but by primary care physicians also. It is overall very well tolerated, safe, and easy to use,” he said. “Much like statin prescription started in subspecialty practices but then became quite common in primary care, I envision the same happening with icosapent ethyl.”
Lipid expert Roger Blumenthal, MD, who was not involved in the REDUCE-IT trial, also welcomed the new approval for Vascepa.
“The indication is very appropriate; it is great to have another disease-modifying medication in our prevention toolkit,” Dr. Blumenthal, who is director of the Johns Hopkins Ciccarone Center for the Prevention of Cardiovascular Disease in Baltimore, said in an interview.
Some still unsure
But not everyone is in full agreement with the broad indication granted.
One expert who has reservations is James de Lemos, MD, professor of medicine at the University of Texas, Dallas, who sat on the FDA advisory committee that assessed the drug last month.
“I would have preferred a narrower label for now, limited to the secondary prevention indication, because I felt that REDUCE-IT did not include sufficient numbers of patients to justify the high-risk primary-prevention indication. We need an adequately powered, randomized, controlled trial to establish the risk/benefit and cost/benefit in primary prevention, and with this broad label, I worry there will be little incentive for the company to pursue this,” Dr. de Lemos commented in an interview.
“This is a slippery slope, and we should not allow broad indications that extend to primary prevention for drugs that were studied in mixed secondary- and primary-prevention patients, with the results driven by the secondary-prevention subset. These two subgroups are fundamentally different populations in whom the pathophysiology and the background treatments are very different,” he added.
However, Dr. de Lemos acknowledged that he would use Vascepa for some high-risk primary-prevention patients in his practice – those with diabetes, high triglycerides, and multiple risk factors. “I just wish we had more data coming so that I could be more certain of the benefit in this group,” he said.
Dr. Bhatt disclosed sitting on advisory boards for Cardax, Cereno Scientific, Medscape Cardiology, PhaseBio, and Regado Biosciences; conducting unfunded research in association with FlowCo, Fractyl, Merck, Novo Nordisk, PLx Pharma, and Takeda; and receiving research funding from Abbott, Afimmune, Amarin, Amgen, AstraZeneca, Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Chiesi, CSL Behring, Eisai, Ethicon, Ferring Pharmaceuticals, and Forest Laboratories. Dr. De Lemos reported receiving grant and consulting income from Roche Diagnostics and Abbott Diagnostics; and consulting for Ortho Clinical Diagnostics. Dr. Blumenthal has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
The newly approved U.S. indication for icosapent ethyl (Vascepa; Amarin) is broadly in line with the entry criteria for the REDUCE-IT trial and includes a large high-risk primary-prevention population, as well as those with established cardiovascular disease (CVD). The drug, thus, could well be used by millions of patients in the United States alone.
The high-dose, purified eicosapentaenoic acid product was approved last week by the Food and Drug Administration for cardiovascular risk reduction among adults already taking maximally tolerated statins with triglyceride levels of 150 mg/dL or higher who have either established CVD or diabetes and two or more additional risk factors for CVD.
The approval is based largely on the REDUCE-IT trial’s finding of a 25% reduction in risk for major adverse cardiovascular events versus placebo. The FDA stated that the approval is the first for an agent with this specific indication.
Noting that it recognizes the need for additional medical treatments for CVD, the FDA says the new approval “will give patients with elevated triglycerides and other important risk factors, including heart disease, stroke, and diabetes, an adjunctive treatment option that can help decrease their risk of cardiovascular events.”
The drug was unanimously recommended for approval by the FDA’s Endocrinologic and Metabolic Drugs Advisory Committee last month. But while the committee all agreed on its use in patients with established CVD, which made up 70% of the REDUCE-IT population, they were divided on whether the indication should be extended to the high-risk primary-prevention population, who made up just 30% of patients in the study.
Nonetheless, the FDA has gone for a broad indication based on the whole REDUCE-IT population.
In a conference call following the approval, Steven Ketchum, PhD, chief scientific officer at Amarin, pointed out that the primary-prevention population stipulated in the new approval differed very slightly from the REDUCE-IT enrollment criteria.
The trial specified that patients with diabetes should be older than 50 with one other cardiovascular risk factor, whereas the approved population is for diabetes and two cardiovascular risk factors. But as these two risk factors are not specified, they could include age, cigarette smoking, hypertension or use of an antihypertensive agent, low HDL cholesterol, high C-reactive protein, body mass index above 25 kg/m2, renal dysfunction, retinopathy, albuminuria, or an ankle branchial index below 0.9, Dr. Ketchum said.
“So while the label asks for two other risk factors, one of these could be age; so we believe the label is actually slightly broader than the REDUCE-IT inclusion criteria, and doctors have been left with significant leeway to decide which risk factors to consider on top of diabetes.”
Deepak Bhatt, MD, the lead investigator of REDUCE-IT, described the Vascepa approval as “the most significant event in the field of cardiovascular prevention since the introduction of statins nearly 3 decades ago.”
He commended the FDA on “a very evidence-based, prescriber-friendly, and most importantly, patient-friendly label,” which he said was in line with guidelines from multiple professional societies that have already incorporated the REDUCE-IT findings for secondary prevention and diabetic primary prevention.
Dr. Bhatt, who is a professor of medicine at Brigham and Women’s Hospital and Harvard Medical School, Boston, said the label essentially matches the REDUCE-IT population.
“The entry criteria for REDUCE-IT was fasting triglycerides greater than or equal to 150 mg/dL, with a 10% variance allowed (giving a minimum triglyceride value of 135 mg/dL). In actuality, we ended up with about 10% of the population with triglycerides between 100 and 150 mg/dL, and they had a similar degree of benefit as those with higher levels,” he reported.
“In the label, the 150 mg/dL does not specify fasting, and in fact many practices have moved away from fasting lipid measurements for the sake of patient comfort,” Dr. Bhatt added. “On average, nonfasting levels are about 50 mg/dL higher, so the label essentially mirrors those we studied, with the FDA applying good common sense and not being overly dogmatic about the exact wording of the trial inclusion criteria.”
No price change foreseen
Vascepa is already on the market for patients with very high triglyceride levels, and the company says it is not increasing the current price of about $300 a month, which is “relatively low, compared to other new breakthrough drugs.” However, it says it expects sales to grow from vastly increased volume based on the new indication.
Dr. Bhatt noted that REDUCE-IT cost-effectiveness data presented at the recent American Heart Association scientific sessions found the drug to be cost saving in the majority of cases. “That is something that is quite rare in cost-effectiveness research,” he said.
“Now, the key challenge is to identify and treat appropriate patients,” Dr. Bhatt noted. He says this task will largely fall on cardiologists, endocrinologists, and primary care physicians, though stroke neurologists, nephrologists, and vascular medicine specialists will also have patients for whom the data are relevant.
“I believe the drug will ultimately be widely prescribed, initially by subspecialists, but by primary care physicians also. It is overall very well tolerated, safe, and easy to use,” he said. “Much like statin prescription started in subspecialty practices but then became quite common in primary care, I envision the same happening with icosapent ethyl.”
Lipid expert Roger Blumenthal, MD, who was not involved in the REDUCE-IT trial, also welcomed the new approval for Vascepa.
“The indication is very appropriate; it is great to have another disease-modifying medication in our prevention toolkit,” Dr. Blumenthal, who is director of the Johns Hopkins Ciccarone Center for the Prevention of Cardiovascular Disease in Baltimore, said in an interview.
Some still unsure
But not everyone is in full agreement with the broad indication granted.
One expert who has reservations is James de Lemos, MD, professor of medicine at the University of Texas, Dallas, who sat on the FDA advisory committee that assessed the drug last month.
“I would have preferred a narrower label for now, limited to the secondary prevention indication, because I felt that REDUCE-IT did not include sufficient numbers of patients to justify the high-risk primary-prevention indication. We need an adequately powered, randomized, controlled trial to establish the risk/benefit and cost/benefit in primary prevention, and with this broad label, I worry there will be little incentive for the company to pursue this,” Dr. de Lemos commented in an interview.
“This is a slippery slope, and we should not allow broad indications that extend to primary prevention for drugs that were studied in mixed secondary- and primary-prevention patients, with the results driven by the secondary-prevention subset. These two subgroups are fundamentally different populations in whom the pathophysiology and the background treatments are very different,” he added.
However, Dr. de Lemos acknowledged that he would use Vascepa for some high-risk primary-prevention patients in his practice – those with diabetes, high triglycerides, and multiple risk factors. “I just wish we had more data coming so that I could be more certain of the benefit in this group,” he said.
Dr. Bhatt disclosed sitting on advisory boards for Cardax, Cereno Scientific, Medscape Cardiology, PhaseBio, and Regado Biosciences; conducting unfunded research in association with FlowCo, Fractyl, Merck, Novo Nordisk, PLx Pharma, and Takeda; and receiving research funding from Abbott, Afimmune, Amarin, Amgen, AstraZeneca, Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Chiesi, CSL Behring, Eisai, Ethicon, Ferring Pharmaceuticals, and Forest Laboratories. Dr. De Lemos reported receiving grant and consulting income from Roche Diagnostics and Abbott Diagnostics; and consulting for Ortho Clinical Diagnostics. Dr. Blumenthal has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
The newly approved U.S. indication for icosapent ethyl (Vascepa; Amarin) is broadly in line with the entry criteria for the REDUCE-IT trial and includes a large high-risk primary-prevention population, as well as those with established cardiovascular disease (CVD). The drug, thus, could well be used by millions of patients in the United States alone.
The high-dose, purified eicosapentaenoic acid product was approved last week by the Food and Drug Administration for cardiovascular risk reduction among adults already taking maximally tolerated statins with triglyceride levels of 150 mg/dL or higher who have either established CVD or diabetes and two or more additional risk factors for CVD.
The approval is based largely on the REDUCE-IT trial’s finding of a 25% reduction in risk for major adverse cardiovascular events versus placebo. The FDA stated that the approval is the first for an agent with this specific indication.
Noting that it recognizes the need for additional medical treatments for CVD, the FDA says the new approval “will give patients with elevated triglycerides and other important risk factors, including heart disease, stroke, and diabetes, an adjunctive treatment option that can help decrease their risk of cardiovascular events.”
The drug was unanimously recommended for approval by the FDA’s Endocrinologic and Metabolic Drugs Advisory Committee last month. But while the committee all agreed on its use in patients with established CVD, which made up 70% of the REDUCE-IT population, they were divided on whether the indication should be extended to the high-risk primary-prevention population, who made up just 30% of patients in the study.
Nonetheless, the FDA has gone for a broad indication based on the whole REDUCE-IT population.
In a conference call following the approval, Steven Ketchum, PhD, chief scientific officer at Amarin, pointed out that the primary-prevention population stipulated in the new approval differed very slightly from the REDUCE-IT enrollment criteria.
The trial specified that patients with diabetes should be older than 50 with one other cardiovascular risk factor, whereas the approved population is for diabetes and two cardiovascular risk factors. But as these two risk factors are not specified, they could include age, cigarette smoking, hypertension or use of an antihypertensive agent, low HDL cholesterol, high C-reactive protein, body mass index above 25 kg/m2, renal dysfunction, retinopathy, albuminuria, or an ankle branchial index below 0.9, Dr. Ketchum said.
“So while the label asks for two other risk factors, one of these could be age; so we believe the label is actually slightly broader than the REDUCE-IT inclusion criteria, and doctors have been left with significant leeway to decide which risk factors to consider on top of diabetes.”
Deepak Bhatt, MD, the lead investigator of REDUCE-IT, described the Vascepa approval as “the most significant event in the field of cardiovascular prevention since the introduction of statins nearly 3 decades ago.”
He commended the FDA on “a very evidence-based, prescriber-friendly, and most importantly, patient-friendly label,” which he said was in line with guidelines from multiple professional societies that have already incorporated the REDUCE-IT findings for secondary prevention and diabetic primary prevention.
Dr. Bhatt, who is a professor of medicine at Brigham and Women’s Hospital and Harvard Medical School, Boston, said the label essentially matches the REDUCE-IT population.
“The entry criteria for REDUCE-IT was fasting triglycerides greater than or equal to 150 mg/dL, with a 10% variance allowed (giving a minimum triglyceride value of 135 mg/dL). In actuality, we ended up with about 10% of the population with triglycerides between 100 and 150 mg/dL, and they had a similar degree of benefit as those with higher levels,” he reported.
“In the label, the 150 mg/dL does not specify fasting, and in fact many practices have moved away from fasting lipid measurements for the sake of patient comfort,” Dr. Bhatt added. “On average, nonfasting levels are about 50 mg/dL higher, so the label essentially mirrors those we studied, with the FDA applying good common sense and not being overly dogmatic about the exact wording of the trial inclusion criteria.”
No price change foreseen
Vascepa is already on the market for patients with very high triglyceride levels, and the company says it is not increasing the current price of about $300 a month, which is “relatively low, compared to other new breakthrough drugs.” However, it says it expects sales to grow from vastly increased volume based on the new indication.
Dr. Bhatt noted that REDUCE-IT cost-effectiveness data presented at the recent American Heart Association scientific sessions found the drug to be cost saving in the majority of cases. “That is something that is quite rare in cost-effectiveness research,” he said.
“Now, the key challenge is to identify and treat appropriate patients,” Dr. Bhatt noted. He says this task will largely fall on cardiologists, endocrinologists, and primary care physicians, though stroke neurologists, nephrologists, and vascular medicine specialists will also have patients for whom the data are relevant.
“I believe the drug will ultimately be widely prescribed, initially by subspecialists, but by primary care physicians also. It is overall very well tolerated, safe, and easy to use,” he said. “Much like statin prescription started in subspecialty practices but then became quite common in primary care, I envision the same happening with icosapent ethyl.”
Lipid expert Roger Blumenthal, MD, who was not involved in the REDUCE-IT trial, also welcomed the new approval for Vascepa.
“The indication is very appropriate; it is great to have another disease-modifying medication in our prevention toolkit,” Dr. Blumenthal, who is director of the Johns Hopkins Ciccarone Center for the Prevention of Cardiovascular Disease in Baltimore, said in an interview.
Some still unsure
But not everyone is in full agreement with the broad indication granted.
One expert who has reservations is James de Lemos, MD, professor of medicine at the University of Texas, Dallas, who sat on the FDA advisory committee that assessed the drug last month.
“I would have preferred a narrower label for now, limited to the secondary prevention indication, because I felt that REDUCE-IT did not include sufficient numbers of patients to justify the high-risk primary-prevention indication. We need an adequately powered, randomized, controlled trial to establish the risk/benefit and cost/benefit in primary prevention, and with this broad label, I worry there will be little incentive for the company to pursue this,” Dr. de Lemos commented in an interview.
“This is a slippery slope, and we should not allow broad indications that extend to primary prevention for drugs that were studied in mixed secondary- and primary-prevention patients, with the results driven by the secondary-prevention subset. These two subgroups are fundamentally different populations in whom the pathophysiology and the background treatments are very different,” he added.
However, Dr. de Lemos acknowledged that he would use Vascepa for some high-risk primary-prevention patients in his practice – those with diabetes, high triglycerides, and multiple risk factors. “I just wish we had more data coming so that I could be more certain of the benefit in this group,” he said.
Dr. Bhatt disclosed sitting on advisory boards for Cardax, Cereno Scientific, Medscape Cardiology, PhaseBio, and Regado Biosciences; conducting unfunded research in association with FlowCo, Fractyl, Merck, Novo Nordisk, PLx Pharma, and Takeda; and receiving research funding from Abbott, Afimmune, Amarin, Amgen, AstraZeneca, Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Chiesi, CSL Behring, Eisai, Ethicon, Ferring Pharmaceuticals, and Forest Laboratories. Dr. De Lemos reported receiving grant and consulting income from Roche Diagnostics and Abbott Diagnostics; and consulting for Ortho Clinical Diagnostics. Dr. Blumenthal has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Blood test may advance diagnosis of GI cancers
SAN FRANCISCO – A new assay based on methylation patterns in plasma cell-free DNA holds promise for improving detection of diverse gastrointestinal (GI) cancers when they are still curable, findings of the Circulating Cell-free Genome Atlas (CCGA) study suggest.
Among 447 patients with GI cancers of varied types and stages, the multicancer assay had sensitivity exceeding 80% for all stages (exceeding 70% for stages I to III) and specificity exceeding 99%, investigators reported at the 2020 GI Cancers Symposium. In addition, the assay correctly determined the tissue of origin about 90% of the time.
“To pursue early detection at a population scale while minimizing harm and cost, several features are important, including a low false-positive rate, a high detection rate, the ability to localize the site of origin of the malignancy, and limiting overdiagnosis,” noted lead investigator Brian M. Wolpin, MD, MPH, director of the Gastrointestinal Cancer Center and director of the Hale Family Center for Pancreatic Cancer Research at Dana-Farber Cancer Institute in Boston. “This multicancer early detection test evaluating cell-free DNA methylation may be a useful test to detect GI cancers and may guide further evaluation and workup.”
About three-quarters of all cancer patients in the study had symptoms that ultimately led to their diagnosis, Dr. Wolpin acknowledged. But the investigators are conducting two large additional studies among asymptomatic populations to better assess the assay’s screening potential: the STRIVE study, which has enrolled nearly 100,000 women undergoing screening mammography, and the SUMMIT study, which is enrolling 50,000 men and women without a known cancer diagnosis, with enrichment of the sample for smokers.
Clinical potential
“The holy grail, of course, is to try to find any test – a blood test, a urine test, a breath test, a stool test – that will allow us to detect cancer at an earlier and more curable stage, and we have hints now that we are headed in the right direction,” session cochair George A. Fisher Jr., MD, PhD, said in an interview. “I don’t know that this is the technology that will do it, but I think it’s superior to the technology just looking at DNA mutations.”
It is a further merit that the assay also has good accuracy in ascertaining the cancer’s tissue of origin, as that should help streamline the diagnostic workup, he agreed.
The assay is already attractive for patients with suspicious lesions or symptoms, but further research will be needed to assess its performance in detecting asymptomatic cancer, and logistic issues would have to be addressed, according to Dr. Fisher of Stanford (Calif.) University.
“As an outright screening test, I don’t think we are there yet. We would have to identify the populations in whom to use the test and come up with some frequency of how often we use it,” he said. Consideration would need to be given to informed consent, especially as some patients may have a positive assay result but a cancer that can’t be found, generating psychological distress.
And cost will come into play. “The problem is, for most stage I cancers you can screen for, they are still very low in likelihood, so you have to screen a lot of people, which means a lot of expense,” he elaborated.
“It will be interesting in the research world to decide when such a test would become a reasonable standard of care, and how regulators will view that and how insurers will view that in terms of cost-efficacy analysis,” Dr. Fisher concluded.
Study details
The CCGA study has enrolled 15,254 participants with and without cancer at 142 U.S and Canadian sites. Participants provide blood samples (at diagnosis for those with cancer) and are followed up for 5 years.
To develop the assay, the investigators performed targeted methylation sequencing of cell-free DNA from plasma and trained an algorithm to use the methylation patterns to detect more than 20 cancer types and classify them based on the organ of origin.
Dr. Wolpin reported results for a CCGA substudy conducted among 2,185 patients with cancer and 2,131 individuals without cancer. About a fifth of the former group had cancers of the GI tract (colon or rectum in 174 patients, pancreas in 123, esophagus in 71, liver or bile duct in 40, stomach in 25, gallbladder in 14). The population was split into training and validation sets.
The assay had a specificity of 99.8% in the entire training set and 99.3% in the entire validation set, corresponding to false-positives in just 0.2% and 0.7% of the individuals without cancer, according to data reported at the symposium, which is sponsored by the American Gastroenterological Association, American Society of Clinical Oncology, the American Society for Radiation Oncology, and the Society of Surgical Oncology.
For GI cancers, assay sensitivity for stage I-IV disease was 82% in the training set and 81% in the validation set, and sensitivity for stage I-III disease was 73% and 71%, respectively. By specific nonmetastatic stage, sensitivity was about 50% for stage I disease, 75% for stage II disease, and 85% for stage III disease.
Fully 97% of the GI cancers having detectable cell-free DNA were assigned a tissue of origin by the assay. The predicted tissue of origin had an accuracy of 91% in the training set and 89% in the validation set, with similar values across GI cancer types.
Dr. Wolpin disclosed having a relationship with various pharmaceutical companies, including GRAIL, which funded the study. Dr. Fisher disclosed relationships with numerous pharmaceutical companies and that an immediate family member has stock in Seattle Genetics
SOURCE: Wolpin BM et al. 2020 GI Symposium, Abstract 283.
SAN FRANCISCO – A new assay based on methylation patterns in plasma cell-free DNA holds promise for improving detection of diverse gastrointestinal (GI) cancers when they are still curable, findings of the Circulating Cell-free Genome Atlas (CCGA) study suggest.
Among 447 patients with GI cancers of varied types and stages, the multicancer assay had sensitivity exceeding 80% for all stages (exceeding 70% for stages I to III) and specificity exceeding 99%, investigators reported at the 2020 GI Cancers Symposium. In addition, the assay correctly determined the tissue of origin about 90% of the time.
“To pursue early detection at a population scale while minimizing harm and cost, several features are important, including a low false-positive rate, a high detection rate, the ability to localize the site of origin of the malignancy, and limiting overdiagnosis,” noted lead investigator Brian M. Wolpin, MD, MPH, director of the Gastrointestinal Cancer Center and director of the Hale Family Center for Pancreatic Cancer Research at Dana-Farber Cancer Institute in Boston. “This multicancer early detection test evaluating cell-free DNA methylation may be a useful test to detect GI cancers and may guide further evaluation and workup.”
About three-quarters of all cancer patients in the study had symptoms that ultimately led to their diagnosis, Dr. Wolpin acknowledged. But the investigators are conducting two large additional studies among asymptomatic populations to better assess the assay’s screening potential: the STRIVE study, which has enrolled nearly 100,000 women undergoing screening mammography, and the SUMMIT study, which is enrolling 50,000 men and women without a known cancer diagnosis, with enrichment of the sample for smokers.
Clinical potential
“The holy grail, of course, is to try to find any test – a blood test, a urine test, a breath test, a stool test – that will allow us to detect cancer at an earlier and more curable stage, and we have hints now that we are headed in the right direction,” session cochair George A. Fisher Jr., MD, PhD, said in an interview. “I don’t know that this is the technology that will do it, but I think it’s superior to the technology just looking at DNA mutations.”
It is a further merit that the assay also has good accuracy in ascertaining the cancer’s tissue of origin, as that should help streamline the diagnostic workup, he agreed.
The assay is already attractive for patients with suspicious lesions or symptoms, but further research will be needed to assess its performance in detecting asymptomatic cancer, and logistic issues would have to be addressed, according to Dr. Fisher of Stanford (Calif.) University.
“As an outright screening test, I don’t think we are there yet. We would have to identify the populations in whom to use the test and come up with some frequency of how often we use it,” he said. Consideration would need to be given to informed consent, especially as some patients may have a positive assay result but a cancer that can’t be found, generating psychological distress.
And cost will come into play. “The problem is, for most stage I cancers you can screen for, they are still very low in likelihood, so you have to screen a lot of people, which means a lot of expense,” he elaborated.
“It will be interesting in the research world to decide when such a test would become a reasonable standard of care, and how regulators will view that and how insurers will view that in terms of cost-efficacy analysis,” Dr. Fisher concluded.
Study details
The CCGA study has enrolled 15,254 participants with and without cancer at 142 U.S and Canadian sites. Participants provide blood samples (at diagnosis for those with cancer) and are followed up for 5 years.
To develop the assay, the investigators performed targeted methylation sequencing of cell-free DNA from plasma and trained an algorithm to use the methylation patterns to detect more than 20 cancer types and classify them based on the organ of origin.
Dr. Wolpin reported results for a CCGA substudy conducted among 2,185 patients with cancer and 2,131 individuals without cancer. About a fifth of the former group had cancers of the GI tract (colon or rectum in 174 patients, pancreas in 123, esophagus in 71, liver or bile duct in 40, stomach in 25, gallbladder in 14). The population was split into training and validation sets.
The assay had a specificity of 99.8% in the entire training set and 99.3% in the entire validation set, corresponding to false-positives in just 0.2% and 0.7% of the individuals without cancer, according to data reported at the symposium, which is sponsored by the American Gastroenterological Association, American Society of Clinical Oncology, the American Society for Radiation Oncology, and the Society of Surgical Oncology.
For GI cancers, assay sensitivity for stage I-IV disease was 82% in the training set and 81% in the validation set, and sensitivity for stage I-III disease was 73% and 71%, respectively. By specific nonmetastatic stage, sensitivity was about 50% for stage I disease, 75% for stage II disease, and 85% for stage III disease.
Fully 97% of the GI cancers having detectable cell-free DNA were assigned a tissue of origin by the assay. The predicted tissue of origin had an accuracy of 91% in the training set and 89% in the validation set, with similar values across GI cancer types.
Dr. Wolpin disclosed having a relationship with various pharmaceutical companies, including GRAIL, which funded the study. Dr. Fisher disclosed relationships with numerous pharmaceutical companies and that an immediate family member has stock in Seattle Genetics
SOURCE: Wolpin BM et al. 2020 GI Symposium, Abstract 283.
SAN FRANCISCO – A new assay based on methylation patterns in plasma cell-free DNA holds promise for improving detection of diverse gastrointestinal (GI) cancers when they are still curable, findings of the Circulating Cell-free Genome Atlas (CCGA) study suggest.
Among 447 patients with GI cancers of varied types and stages, the multicancer assay had sensitivity exceeding 80% for all stages (exceeding 70% for stages I to III) and specificity exceeding 99%, investigators reported at the 2020 GI Cancers Symposium. In addition, the assay correctly determined the tissue of origin about 90% of the time.
“To pursue early detection at a population scale while minimizing harm and cost, several features are important, including a low false-positive rate, a high detection rate, the ability to localize the site of origin of the malignancy, and limiting overdiagnosis,” noted lead investigator Brian M. Wolpin, MD, MPH, director of the Gastrointestinal Cancer Center and director of the Hale Family Center for Pancreatic Cancer Research at Dana-Farber Cancer Institute in Boston. “This multicancer early detection test evaluating cell-free DNA methylation may be a useful test to detect GI cancers and may guide further evaluation and workup.”
About three-quarters of all cancer patients in the study had symptoms that ultimately led to their diagnosis, Dr. Wolpin acknowledged. But the investigators are conducting two large additional studies among asymptomatic populations to better assess the assay’s screening potential: the STRIVE study, which has enrolled nearly 100,000 women undergoing screening mammography, and the SUMMIT study, which is enrolling 50,000 men and women without a known cancer diagnosis, with enrichment of the sample for smokers.
Clinical potential
“The holy grail, of course, is to try to find any test – a blood test, a urine test, a breath test, a stool test – that will allow us to detect cancer at an earlier and more curable stage, and we have hints now that we are headed in the right direction,” session cochair George A. Fisher Jr., MD, PhD, said in an interview. “I don’t know that this is the technology that will do it, but I think it’s superior to the technology just looking at DNA mutations.”
It is a further merit that the assay also has good accuracy in ascertaining the cancer’s tissue of origin, as that should help streamline the diagnostic workup, he agreed.
The assay is already attractive for patients with suspicious lesions or symptoms, but further research will be needed to assess its performance in detecting asymptomatic cancer, and logistic issues would have to be addressed, according to Dr. Fisher of Stanford (Calif.) University.
“As an outright screening test, I don’t think we are there yet. We would have to identify the populations in whom to use the test and come up with some frequency of how often we use it,” he said. Consideration would need to be given to informed consent, especially as some patients may have a positive assay result but a cancer that can’t be found, generating psychological distress.
And cost will come into play. “The problem is, for most stage I cancers you can screen for, they are still very low in likelihood, so you have to screen a lot of people, which means a lot of expense,” he elaborated.
“It will be interesting in the research world to decide when such a test would become a reasonable standard of care, and how regulators will view that and how insurers will view that in terms of cost-efficacy analysis,” Dr. Fisher concluded.
Study details
The CCGA study has enrolled 15,254 participants with and without cancer at 142 U.S and Canadian sites. Participants provide blood samples (at diagnosis for those with cancer) and are followed up for 5 years.
To develop the assay, the investigators performed targeted methylation sequencing of cell-free DNA from plasma and trained an algorithm to use the methylation patterns to detect more than 20 cancer types and classify them based on the organ of origin.
Dr. Wolpin reported results for a CCGA substudy conducted among 2,185 patients with cancer and 2,131 individuals without cancer. About a fifth of the former group had cancers of the GI tract (colon or rectum in 174 patients, pancreas in 123, esophagus in 71, liver or bile duct in 40, stomach in 25, gallbladder in 14). The population was split into training and validation sets.
The assay had a specificity of 99.8% in the entire training set and 99.3% in the entire validation set, corresponding to false-positives in just 0.2% and 0.7% of the individuals without cancer, according to data reported at the symposium, which is sponsored by the American Gastroenterological Association, American Society of Clinical Oncology, the American Society for Radiation Oncology, and the Society of Surgical Oncology.
For GI cancers, assay sensitivity for stage I-IV disease was 82% in the training set and 81% in the validation set, and sensitivity for stage I-III disease was 73% and 71%, respectively. By specific nonmetastatic stage, sensitivity was about 50% for stage I disease, 75% for stage II disease, and 85% for stage III disease.
Fully 97% of the GI cancers having detectable cell-free DNA were assigned a tissue of origin by the assay. The predicted tissue of origin had an accuracy of 91% in the training set and 89% in the validation set, with similar values across GI cancer types.
Dr. Wolpin disclosed having a relationship with various pharmaceutical companies, including GRAIL, which funded the study. Dr. Fisher disclosed relationships with numerous pharmaceutical companies and that an immediate family member has stock in Seattle Genetics
SOURCE: Wolpin BM et al. 2020 GI Symposium, Abstract 283.
REPORTING FROM THE 2020 GI CANCERS SYMPOSIUM
FDA approves first treatment for advanced epithelioid sarcoma
The Food and Drug Administration has granted accelerated approval to tazemetostat (Tazverik) for the treatment of adults and pediatric patients aged 16 years and older with metastatic or locally advanced epithelioid sarcoma not eligible for complete resection.
Approval was based on overall response rate in a trial enrolling 62 patients with metastatic or locally advanced epithelioid sarcoma. The overall response rate was 15%, with 1.6% of patients having a complete response and 13% having a partial response. Of the nine patients that had a response, six (67%) had a response lasting 6 months or longer, the FDA said in a press statement.
The most common side effects for patients taking tazemetostat were pain, fatigue, nausea, decreased appetite, vomiting, and constipation. Patients treated with tazemetostat are at increased risk of developing secondary malignancies, including T-cell lymphoblastic lymphoma, myelodysplastic syndrome, and acute myeloid leukemia.
“Epithelioid sarcoma accounts for less than 1% of all soft-tissue sarcomas,” said Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence and acting director of the Office of Oncologic Diseases in the Center for Drug Evaluation and Research. “Until today, there were no treatment options specifically for patients with epithelioid sarcoma. The approval of Tazverik provides a treatment option that specifically targets this disease.”
Tazemetostat must be dispensed with a patient medication guide that describes important information about the drug’s uses and risks, the FDA said.
The Food and Drug Administration has granted accelerated approval to tazemetostat (Tazverik) for the treatment of adults and pediatric patients aged 16 years and older with metastatic or locally advanced epithelioid sarcoma not eligible for complete resection.
Approval was based on overall response rate in a trial enrolling 62 patients with metastatic or locally advanced epithelioid sarcoma. The overall response rate was 15%, with 1.6% of patients having a complete response and 13% having a partial response. Of the nine patients that had a response, six (67%) had a response lasting 6 months or longer, the FDA said in a press statement.
The most common side effects for patients taking tazemetostat were pain, fatigue, nausea, decreased appetite, vomiting, and constipation. Patients treated with tazemetostat are at increased risk of developing secondary malignancies, including T-cell lymphoblastic lymphoma, myelodysplastic syndrome, and acute myeloid leukemia.
“Epithelioid sarcoma accounts for less than 1% of all soft-tissue sarcomas,” said Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence and acting director of the Office of Oncologic Diseases in the Center for Drug Evaluation and Research. “Until today, there were no treatment options specifically for patients with epithelioid sarcoma. The approval of Tazverik provides a treatment option that specifically targets this disease.”
Tazemetostat must be dispensed with a patient medication guide that describes important information about the drug’s uses and risks, the FDA said.
The Food and Drug Administration has granted accelerated approval to tazemetostat (Tazverik) for the treatment of adults and pediatric patients aged 16 years and older with metastatic or locally advanced epithelioid sarcoma not eligible for complete resection.
Approval was based on overall response rate in a trial enrolling 62 patients with metastatic or locally advanced epithelioid sarcoma. The overall response rate was 15%, with 1.6% of patients having a complete response and 13% having a partial response. Of the nine patients that had a response, six (67%) had a response lasting 6 months or longer, the FDA said in a press statement.
The most common side effects for patients taking tazemetostat were pain, fatigue, nausea, decreased appetite, vomiting, and constipation. Patients treated with tazemetostat are at increased risk of developing secondary malignancies, including T-cell lymphoblastic lymphoma, myelodysplastic syndrome, and acute myeloid leukemia.
“Epithelioid sarcoma accounts for less than 1% of all soft-tissue sarcomas,” said Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence and acting director of the Office of Oncologic Diseases in the Center for Drug Evaluation and Research. “Until today, there were no treatment options specifically for patients with epithelioid sarcoma. The approval of Tazverik provides a treatment option that specifically targets this disease.”
Tazemetostat must be dispensed with a patient medication guide that describes important information about the drug’s uses and risks, the FDA said.
Family-focused therapy linked to longer remissions in youth at risk for bipolar disorder
A 4-month intensive program of family-focused therapy worked better than a less-intensive program in delaying new mood episodes among young people at risk of developing bipolar disorder, new research shows.
“This study extends the results of other randomized clinical trials indicating effects of family psychoeducation and skill training on the long-term trajectory of depressive symptoms in pediatric mood disorders,” wrote David J. Miklowitz, PhD, of the department of psychiatry and biobehavioral sciences at the University of California, Los Angeles, and colleagues. The study was published in JAMA Psychiatry.
For their research, the investigators recruited 127 subjects aged 9-17 years (mean age, 13 years) deemed at high risk for later bipolar I or II disorder for having depression or subthreshold mania along with active mood symptoms and a family history of bipolar disorder. Some 85% of subjects had depression symptoms at enrollment.
Subjects were randomized to 12 sessions over 4 months of family-focused therapy – a psychoeducation, communication, and problem-solving training program incorporating caretakers and also siblings if possible (n = 61) – or to 3 sessions of family-focused therapy and an additional 3 of individual therapy in the same 4-month time frame (n = 66). Medication was allowed for all subjects, and patients were followed for a median 2 years after the intervention. Baseline characteristics, medication use, and dropout rates were similar between the groups.
Both groups saw similarly high rates of new episodes of major depression, mania, or hypomania during follow-up; however, those in the intensive family-focused therapy group saw longer intervals of wellness, with a median 81 weeks (95% confidence interval, 56-123 weeks) from randomization until the first observed mood episode, compared with 63 weeks (95% CI, 44-78 weeks) to an episode for the less-intensive group (P = .03). Dr. Miklowitz and colleagues did not find differences in the severity of mood episodes following either treatment mode or in later conversion to bipolar I or II.
The researchers described as limitations of their study its inability to measure the “temporal relationship between changes in family communication and symptom changes in patients,” which would help answer whether improvements in communication patterns aid symptom regulation, or whether more stable patients are better able to manage difficult family interactions.
Family-focused therapy “may have uniquely enduring effects that extend into the maintenance phase of treatment,” Dr. Miklowitz and colleagues wrote.
The study was funded by the National Institute of Mental Health. Several coauthors, including the lead author, reported receiving research grants from NIMH and other foundations. Two additional coauthors reported receiving pharmaceutical industry funding, including advisory board and consulting fees.
SOURCE: Miklowitz DJ et al. JAMA Psychiatry. 2020 Jan 15. doi: 10.1001/jamapsychiatry.2019.4520.
A 4-month intensive program of family-focused therapy worked better than a less-intensive program in delaying new mood episodes among young people at risk of developing bipolar disorder, new research shows.
“This study extends the results of other randomized clinical trials indicating effects of family psychoeducation and skill training on the long-term trajectory of depressive symptoms in pediatric mood disorders,” wrote David J. Miklowitz, PhD, of the department of psychiatry and biobehavioral sciences at the University of California, Los Angeles, and colleagues. The study was published in JAMA Psychiatry.
For their research, the investigators recruited 127 subjects aged 9-17 years (mean age, 13 years) deemed at high risk for later bipolar I or II disorder for having depression or subthreshold mania along with active mood symptoms and a family history of bipolar disorder. Some 85% of subjects had depression symptoms at enrollment.
Subjects were randomized to 12 sessions over 4 months of family-focused therapy – a psychoeducation, communication, and problem-solving training program incorporating caretakers and also siblings if possible (n = 61) – or to 3 sessions of family-focused therapy and an additional 3 of individual therapy in the same 4-month time frame (n = 66). Medication was allowed for all subjects, and patients were followed for a median 2 years after the intervention. Baseline characteristics, medication use, and dropout rates were similar between the groups.
Both groups saw similarly high rates of new episodes of major depression, mania, or hypomania during follow-up; however, those in the intensive family-focused therapy group saw longer intervals of wellness, with a median 81 weeks (95% confidence interval, 56-123 weeks) from randomization until the first observed mood episode, compared with 63 weeks (95% CI, 44-78 weeks) to an episode for the less-intensive group (P = .03). Dr. Miklowitz and colleagues did not find differences in the severity of mood episodes following either treatment mode or in later conversion to bipolar I or II.
The researchers described as limitations of their study its inability to measure the “temporal relationship between changes in family communication and symptom changes in patients,” which would help answer whether improvements in communication patterns aid symptom regulation, or whether more stable patients are better able to manage difficult family interactions.
Family-focused therapy “may have uniquely enduring effects that extend into the maintenance phase of treatment,” Dr. Miklowitz and colleagues wrote.
The study was funded by the National Institute of Mental Health. Several coauthors, including the lead author, reported receiving research grants from NIMH and other foundations. Two additional coauthors reported receiving pharmaceutical industry funding, including advisory board and consulting fees.
SOURCE: Miklowitz DJ et al. JAMA Psychiatry. 2020 Jan 15. doi: 10.1001/jamapsychiatry.2019.4520.
A 4-month intensive program of family-focused therapy worked better than a less-intensive program in delaying new mood episodes among young people at risk of developing bipolar disorder, new research shows.
“This study extends the results of other randomized clinical trials indicating effects of family psychoeducation and skill training on the long-term trajectory of depressive symptoms in pediatric mood disorders,” wrote David J. Miklowitz, PhD, of the department of psychiatry and biobehavioral sciences at the University of California, Los Angeles, and colleagues. The study was published in JAMA Psychiatry.
For their research, the investigators recruited 127 subjects aged 9-17 years (mean age, 13 years) deemed at high risk for later bipolar I or II disorder for having depression or subthreshold mania along with active mood symptoms and a family history of bipolar disorder. Some 85% of subjects had depression symptoms at enrollment.
Subjects were randomized to 12 sessions over 4 months of family-focused therapy – a psychoeducation, communication, and problem-solving training program incorporating caretakers and also siblings if possible (n = 61) – or to 3 sessions of family-focused therapy and an additional 3 of individual therapy in the same 4-month time frame (n = 66). Medication was allowed for all subjects, and patients were followed for a median 2 years after the intervention. Baseline characteristics, medication use, and dropout rates were similar between the groups.
Both groups saw similarly high rates of new episodes of major depression, mania, or hypomania during follow-up; however, those in the intensive family-focused therapy group saw longer intervals of wellness, with a median 81 weeks (95% confidence interval, 56-123 weeks) from randomization until the first observed mood episode, compared with 63 weeks (95% CI, 44-78 weeks) to an episode for the less-intensive group (P = .03). Dr. Miklowitz and colleagues did not find differences in the severity of mood episodes following either treatment mode or in later conversion to bipolar I or II.
The researchers described as limitations of their study its inability to measure the “temporal relationship between changes in family communication and symptom changes in patients,” which would help answer whether improvements in communication patterns aid symptom regulation, or whether more stable patients are better able to manage difficult family interactions.
Family-focused therapy “may have uniquely enduring effects that extend into the maintenance phase of treatment,” Dr. Miklowitz and colleagues wrote.
The study was funded by the National Institute of Mental Health. Several coauthors, including the lead author, reported receiving research grants from NIMH and other foundations. Two additional coauthors reported receiving pharmaceutical industry funding, including advisory board and consulting fees.
SOURCE: Miklowitz DJ et al. JAMA Psychiatry. 2020 Jan 15. doi: 10.1001/jamapsychiatry.2019.4520.
FROM JAMA PSYCHIATRY
Suicide rate higher than average for female clinicians
The suicide rate for women who provide health care is higher than that of all women of working age, while male health care practitioners are less likely to end their lives than working-age men as a whole, according to the Centers for Disease Control and Prevention.
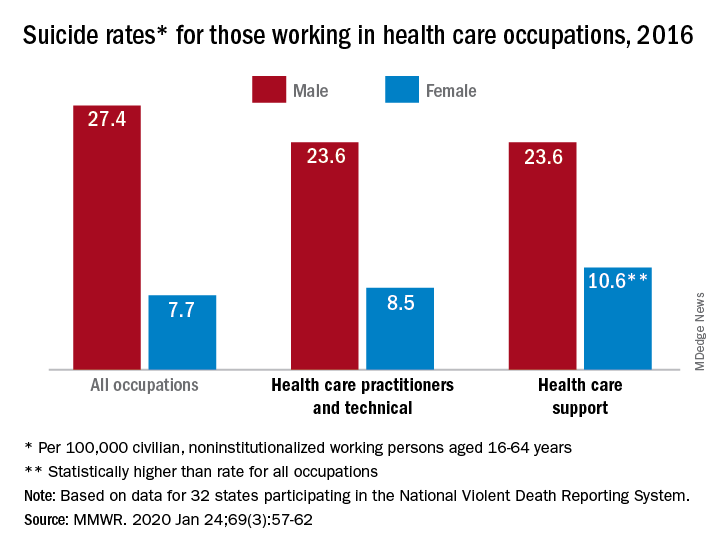
In 2016, the suicide rate for women classified as “healthcare practitioners and technical” – a category that includes physicians and surgeons, as well as chiropractors, physician assistants, and nurse practitioners – was 8.5 per 100,000 population, compared with 7.7 per 100,000 for all working women aged 16-64 years. That difference, however, was not statistically significant, Cora Peterson, PhD, and associates at the CDC said in the Morbidity and Mortality Weekly Report.
For females classified as “healthcare support” – medical assistants and transcriptionists, phlebotomists, and pharmacy aides – the suicide rate of 10.6 per 100,000 was significantly higher than that of all working women, the investigators noted.
The suicide rate for males in each of the two occupation categories was 23.6 per 100,000 population in 2016, lower than the rate of 27.4 per 100,000 for males of all occupations, they said, based on data from 32 states that participated in the 2016 National Violent Death Reporting System.
For males, the highest suicide rates in occupations meeting criteria for sample size were “construction and extraction” (49.4 per 100,000); “installation, maintenance, and repair” (36.9); and “arts, design, entertainment, sports, and media” (32.0). Among females, the highest rates were seen in “construction and extraction” (25.5 per 100,000), “protective service” (14.0), and “transportation and material moving” (12.5), with healthcare support next, Dr. Peterson and associates reported.
“Although relative comparisons of suicide rates in this manner are useful for prevention purposes, Therefore, all industry sectors and occupational groups can contribute to reducing suicide incidence,” they wrote.
SOURCE: Peterson C et al. MMWR. 2020 Jan 24;69(3):57-62.
The suicide rate for women who provide health care is higher than that of all women of working age, while male health care practitioners are less likely to end their lives than working-age men as a whole, according to the Centers for Disease Control and Prevention.

In 2016, the suicide rate for women classified as “healthcare practitioners and technical” – a category that includes physicians and surgeons, as well as chiropractors, physician assistants, and nurse practitioners – was 8.5 per 100,000 population, compared with 7.7 per 100,000 for all working women aged 16-64 years. That difference, however, was not statistically significant, Cora Peterson, PhD, and associates at the CDC said in the Morbidity and Mortality Weekly Report.
For females classified as “healthcare support” – medical assistants and transcriptionists, phlebotomists, and pharmacy aides – the suicide rate of 10.6 per 100,000 was significantly higher than that of all working women, the investigators noted.
The suicide rate for males in each of the two occupation categories was 23.6 per 100,000 population in 2016, lower than the rate of 27.4 per 100,000 for males of all occupations, they said, based on data from 32 states that participated in the 2016 National Violent Death Reporting System.
For males, the highest suicide rates in occupations meeting criteria for sample size were “construction and extraction” (49.4 per 100,000); “installation, maintenance, and repair” (36.9); and “arts, design, entertainment, sports, and media” (32.0). Among females, the highest rates were seen in “construction and extraction” (25.5 per 100,000), “protective service” (14.0), and “transportation and material moving” (12.5), with healthcare support next, Dr. Peterson and associates reported.
“Although relative comparisons of suicide rates in this manner are useful for prevention purposes, Therefore, all industry sectors and occupational groups can contribute to reducing suicide incidence,” they wrote.
SOURCE: Peterson C et al. MMWR. 2020 Jan 24;69(3):57-62.
The suicide rate for women who provide health care is higher than that of all women of working age, while male health care practitioners are less likely to end their lives than working-age men as a whole, according to the Centers for Disease Control and Prevention.

In 2016, the suicide rate for women classified as “healthcare practitioners and technical” – a category that includes physicians and surgeons, as well as chiropractors, physician assistants, and nurse practitioners – was 8.5 per 100,000 population, compared with 7.7 per 100,000 for all working women aged 16-64 years. That difference, however, was not statistically significant, Cora Peterson, PhD, and associates at the CDC said in the Morbidity and Mortality Weekly Report.
For females classified as “healthcare support” – medical assistants and transcriptionists, phlebotomists, and pharmacy aides – the suicide rate of 10.6 per 100,000 was significantly higher than that of all working women, the investigators noted.
The suicide rate for males in each of the two occupation categories was 23.6 per 100,000 population in 2016, lower than the rate of 27.4 per 100,000 for males of all occupations, they said, based on data from 32 states that participated in the 2016 National Violent Death Reporting System.
For males, the highest suicide rates in occupations meeting criteria for sample size were “construction and extraction” (49.4 per 100,000); “installation, maintenance, and repair” (36.9); and “arts, design, entertainment, sports, and media” (32.0). Among females, the highest rates were seen in “construction and extraction” (25.5 per 100,000), “protective service” (14.0), and “transportation and material moving” (12.5), with healthcare support next, Dr. Peterson and associates reported.
“Although relative comparisons of suicide rates in this manner are useful for prevention purposes, Therefore, all industry sectors and occupational groups can contribute to reducing suicide incidence,” they wrote.
SOURCE: Peterson C et al. MMWR. 2020 Jan 24;69(3):57-62.
FROM MMWR
New nomogram better predicts bladder cancer risk
A new and simple nomogram for predicting the risk of bladder cancer in patients with microscopic hematuria could optimize the diagnostic work up process, according to a recent study.
The tool may help improve patient understanding about their risk of bladder cancer, as well as alleviate unnecessary diagnostic evaluations for some patients.
“The goal of this study was to identify objective clinical factors associated with a bladder cancer diagnosis and to use these factors to create a nomogram that accurately predicts risk of bladder cancer,” wrote Richard S. Matulewicz, MD, MS, of Northwestern University, Chicago, and colleagues in Urologic Oncology.
Researchers identified 4,178 patients with a new diagnosis of microscopic hematuria from 2007 to 2015. Data was collected from an enterprise data repository of the Northwestern Medicine healthcare system. Study participants who underwent a full microhematuria evaluation were randomized to either a training or validation subgroup. In the training cohort, logistic regression analysis was used to detect factors linked to the diagnosis of bladder cancer. In the model, receiver operating curves were built to predict a diagnosis of bladder cancer among participants. In addition, calibration plots were computed for both subgroups to evaluate the discriminative ability of the model. After analysis, the researchers found significant differences in urinalysis results and demographics among patients with and without a diagnosis of bladder cancer. Patients with bladder cancer had a higher amount of microhematuria (RBC/hpf) on urinalysis (P less than .0001), were more likely previous or current smokers (P = .001), were more often male (68.2% vs. 49.7%; P = .0002), and were older (69.1 vs. 58.2 years; P less than .0001).
With respect to the predictive ability of the model, the area under the curve (AUC) in the training and validation set was 0.79 (95% confidence interval, 0.75-0.83) and 0.74 (95% CI, 0.67-0.80), respectively.
In addition, calibration plots demonstrated that the tool was able to predict the risk of bladder cancer diagnosis for patients with a probability of 0.3 or below.
“These results indicate that the model works best for a range of probabilities of (0-0.30), which is the vast majority of patients clinically and in our data,” the researchers explained.
The team acknowledged that characterizing risk beyond these levels should be done with caution given poor calibration beyond this threshold.
“External validation [of the model] and continued evolution of risk stratification models are needed,” they concluded.
The study was funded by the National Institutes of Health and the American Association of Medical Colleges. The authors reported having no conflicts of interest.
SOURCE: Matulewicz RS et al. Urol Oncol. 2020 Jan 14. doi: 10.1016/j.urolonc.2019.12.010.
A new and simple nomogram for predicting the risk of bladder cancer in patients with microscopic hematuria could optimize the diagnostic work up process, according to a recent study.
The tool may help improve patient understanding about their risk of bladder cancer, as well as alleviate unnecessary diagnostic evaluations for some patients.
“The goal of this study was to identify objective clinical factors associated with a bladder cancer diagnosis and to use these factors to create a nomogram that accurately predicts risk of bladder cancer,” wrote Richard S. Matulewicz, MD, MS, of Northwestern University, Chicago, and colleagues in Urologic Oncology.
Researchers identified 4,178 patients with a new diagnosis of microscopic hematuria from 2007 to 2015. Data was collected from an enterprise data repository of the Northwestern Medicine healthcare system. Study participants who underwent a full microhematuria evaluation were randomized to either a training or validation subgroup. In the training cohort, logistic regression analysis was used to detect factors linked to the diagnosis of bladder cancer. In the model, receiver operating curves were built to predict a diagnosis of bladder cancer among participants. In addition, calibration plots were computed for both subgroups to evaluate the discriminative ability of the model. After analysis, the researchers found significant differences in urinalysis results and demographics among patients with and without a diagnosis of bladder cancer. Patients with bladder cancer had a higher amount of microhematuria (RBC/hpf) on urinalysis (P less than .0001), were more likely previous or current smokers (P = .001), were more often male (68.2% vs. 49.7%; P = .0002), and were older (69.1 vs. 58.2 years; P less than .0001).
With respect to the predictive ability of the model, the area under the curve (AUC) in the training and validation set was 0.79 (95% confidence interval, 0.75-0.83) and 0.74 (95% CI, 0.67-0.80), respectively.
In addition, calibration plots demonstrated that the tool was able to predict the risk of bladder cancer diagnosis for patients with a probability of 0.3 or below.
“These results indicate that the model works best for a range of probabilities of (0-0.30), which is the vast majority of patients clinically and in our data,” the researchers explained.
The team acknowledged that characterizing risk beyond these levels should be done with caution given poor calibration beyond this threshold.
“External validation [of the model] and continued evolution of risk stratification models are needed,” they concluded.
The study was funded by the National Institutes of Health and the American Association of Medical Colleges. The authors reported having no conflicts of interest.
SOURCE: Matulewicz RS et al. Urol Oncol. 2020 Jan 14. doi: 10.1016/j.urolonc.2019.12.010.
A new and simple nomogram for predicting the risk of bladder cancer in patients with microscopic hematuria could optimize the diagnostic work up process, according to a recent study.
The tool may help improve patient understanding about their risk of bladder cancer, as well as alleviate unnecessary diagnostic evaluations for some patients.
“The goal of this study was to identify objective clinical factors associated with a bladder cancer diagnosis and to use these factors to create a nomogram that accurately predicts risk of bladder cancer,” wrote Richard S. Matulewicz, MD, MS, of Northwestern University, Chicago, and colleagues in Urologic Oncology.
Researchers identified 4,178 patients with a new diagnosis of microscopic hematuria from 2007 to 2015. Data was collected from an enterprise data repository of the Northwestern Medicine healthcare system. Study participants who underwent a full microhematuria evaluation were randomized to either a training or validation subgroup. In the training cohort, logistic regression analysis was used to detect factors linked to the diagnosis of bladder cancer. In the model, receiver operating curves were built to predict a diagnosis of bladder cancer among participants. In addition, calibration plots were computed for both subgroups to evaluate the discriminative ability of the model. After analysis, the researchers found significant differences in urinalysis results and demographics among patients with and without a diagnosis of bladder cancer. Patients with bladder cancer had a higher amount of microhematuria (RBC/hpf) on urinalysis (P less than .0001), were more likely previous or current smokers (P = .001), were more often male (68.2% vs. 49.7%; P = .0002), and were older (69.1 vs. 58.2 years; P less than .0001).
With respect to the predictive ability of the model, the area under the curve (AUC) in the training and validation set was 0.79 (95% confidence interval, 0.75-0.83) and 0.74 (95% CI, 0.67-0.80), respectively.
In addition, calibration plots demonstrated that the tool was able to predict the risk of bladder cancer diagnosis for patients with a probability of 0.3 or below.
“These results indicate that the model works best for a range of probabilities of (0-0.30), which is the vast majority of patients clinically and in our data,” the researchers explained.
The team acknowledged that characterizing risk beyond these levels should be done with caution given poor calibration beyond this threshold.
“External validation [of the model] and continued evolution of risk stratification models are needed,” they concluded.
The study was funded by the National Institutes of Health and the American Association of Medical Colleges. The authors reported having no conflicts of interest.
SOURCE: Matulewicz RS et al. Urol Oncol. 2020 Jan 14. doi: 10.1016/j.urolonc.2019.12.010.
FROM UROLOGIC ONCOLOGY
Actor Alan Alda discusses using empathy as an antidote to burnout
LA JOLLA, CALIF. – Physicians and other medical professionals who routinely foster empathic connections with patients may be helping themselves steer clear of burnout.
That’s what iconic actor Alan Alda suggested during a media briefing at Scripps Research on Jan. 16, 2020.

“There’s a tremendous pressure on doctors now to have shorter and shorter visits with their patients,” said the 83-year-old Mr. Alda, who received the Public Welfare Medal from the National Academy of Sciences in 2016 for his work as a champion of science. “A lot of that time is taken up with recording on a computer, which can only put pressure on the doctor.”
Practicing empathy, he continued, “kind of opens people up to one another, which inspirits them.”
Mr. Alda appeared on the research campus to announce that Scripps Research will serve as the new West Coast home of Alda Communication Training, which will work in tandem with the Alan Alda Center for Communicating Science at Stony Brook (N.Y.) University, a nonprofit organization that Mr. Alda helped found in 2009.
“This will be a center where people can come to get training in effective communication,” Mr. Alda, who is the winner of six Emmy Awards and six Golden Globe awards, told an audience of scientists and medical professionals prior to the media briefing.
“It’s an experiential kind of training,” he explained. “We don’t give tips. We don’t give lectures. We put you through exercises that are fun and actually make you laugh, but turn you into a better communicator, so you’re better able to connect to the people you’re talking to.”
During a question-and-answer session, Mr. Alda opened up about his Parkinson’s disease, which he said was diagnosed about 5 years ago. In 2018, he decided to speak publicly about his diagnosis for the first time.
“The reason was that I wanted to communicate to people who had recently been diagnosed not to believe or give into the stereotype that, when you get a diagnosis, your life is over,” said Mr. Alda, who played army surgeon “Hawkeye” Pierce on the TV series “M*A*S*H.”
“Under the burden of that belief, some people won’t tell their family or workplace colleagues,” he said. “There are exercises you can do and medications you can take to prolong the time it takes before Parkinson’s gets much more serious. It’s not to diminish the fact that it can get really bad; but to think that your life is over as soon as you get a diagnosis is wrong.”
The first 2-day training session at Scripps Research will be held in June 2020. Additional sessions are scheduled to take place in October and December. Registration is available at aldacommunicationtraining.com/workshops.
LA JOLLA, CALIF. – Physicians and other medical professionals who routinely foster empathic connections with patients may be helping themselves steer clear of burnout.
That’s what iconic actor Alan Alda suggested during a media briefing at Scripps Research on Jan. 16, 2020.

“There’s a tremendous pressure on doctors now to have shorter and shorter visits with their patients,” said the 83-year-old Mr. Alda, who received the Public Welfare Medal from the National Academy of Sciences in 2016 for his work as a champion of science. “A lot of that time is taken up with recording on a computer, which can only put pressure on the doctor.”
Practicing empathy, he continued, “kind of opens people up to one another, which inspirits them.”
Mr. Alda appeared on the research campus to announce that Scripps Research will serve as the new West Coast home of Alda Communication Training, which will work in tandem with the Alan Alda Center for Communicating Science at Stony Brook (N.Y.) University, a nonprofit organization that Mr. Alda helped found in 2009.
“This will be a center where people can come to get training in effective communication,” Mr. Alda, who is the winner of six Emmy Awards and six Golden Globe awards, told an audience of scientists and medical professionals prior to the media briefing.
“It’s an experiential kind of training,” he explained. “We don’t give tips. We don’t give lectures. We put you through exercises that are fun and actually make you laugh, but turn you into a better communicator, so you’re better able to connect to the people you’re talking to.”
During a question-and-answer session, Mr. Alda opened up about his Parkinson’s disease, which he said was diagnosed about 5 years ago. In 2018, he decided to speak publicly about his diagnosis for the first time.
“The reason was that I wanted to communicate to people who had recently been diagnosed not to believe or give into the stereotype that, when you get a diagnosis, your life is over,” said Mr. Alda, who played army surgeon “Hawkeye” Pierce on the TV series “M*A*S*H.”
“Under the burden of that belief, some people won’t tell their family or workplace colleagues,” he said. “There are exercises you can do and medications you can take to prolong the time it takes before Parkinson’s gets much more serious. It’s not to diminish the fact that it can get really bad; but to think that your life is over as soon as you get a diagnosis is wrong.”
The first 2-day training session at Scripps Research will be held in June 2020. Additional sessions are scheduled to take place in October and December. Registration is available at aldacommunicationtraining.com/workshops.
LA JOLLA, CALIF. – Physicians and other medical professionals who routinely foster empathic connections with patients may be helping themselves steer clear of burnout.
That’s what iconic actor Alan Alda suggested during a media briefing at Scripps Research on Jan. 16, 2020.

“There’s a tremendous pressure on doctors now to have shorter and shorter visits with their patients,” said the 83-year-old Mr. Alda, who received the Public Welfare Medal from the National Academy of Sciences in 2016 for his work as a champion of science. “A lot of that time is taken up with recording on a computer, which can only put pressure on the doctor.”
Practicing empathy, he continued, “kind of opens people up to one another, which inspirits them.”
Mr. Alda appeared on the research campus to announce that Scripps Research will serve as the new West Coast home of Alda Communication Training, which will work in tandem with the Alan Alda Center for Communicating Science at Stony Brook (N.Y.) University, a nonprofit organization that Mr. Alda helped found in 2009.
“This will be a center where people can come to get training in effective communication,” Mr. Alda, who is the winner of six Emmy Awards and six Golden Globe awards, told an audience of scientists and medical professionals prior to the media briefing.
“It’s an experiential kind of training,” he explained. “We don’t give tips. We don’t give lectures. We put you through exercises that are fun and actually make you laugh, but turn you into a better communicator, so you’re better able to connect to the people you’re talking to.”
During a question-and-answer session, Mr. Alda opened up about his Parkinson’s disease, which he said was diagnosed about 5 years ago. In 2018, he decided to speak publicly about his diagnosis for the first time.
“The reason was that I wanted to communicate to people who had recently been diagnosed not to believe or give into the stereotype that, when you get a diagnosis, your life is over,” said Mr. Alda, who played army surgeon “Hawkeye” Pierce on the TV series “M*A*S*H.”
“Under the burden of that belief, some people won’t tell their family or workplace colleagues,” he said. “There are exercises you can do and medications you can take to prolong the time it takes before Parkinson’s gets much more serious. It’s not to diminish the fact that it can get really bad; but to think that your life is over as soon as you get a diagnosis is wrong.”
The first 2-day training session at Scripps Research will be held in June 2020. Additional sessions are scheduled to take place in October and December. Registration is available at aldacommunicationtraining.com/workshops.
Exogenous boosting against shingles not as robust as thought
Exposure to children with chickenpox reduces the incidence of shingles in adults 33% over 2 years, and 27% out to 20 years, according to British investigators.
Being exposed to children with illness due to varicella infection acts as an “exogenous booster” in adults who had chickenpox themselves as children, making shingles less likely, they explained in a BMJ article.
Although that’s good news, it’s been reported previously that exposure to children with chickenpox confers complete protection against shingles in adults for years afterward.
The finding matters in the United Kingdom because varicella vaccine is not part of the pediatric immunization schedule. The United States is the only country that mandates two shots as a requirement for children to attend school.
The United Kingdom, however, is reconsidering its policy. In the past, the exogenous booster idea has been one of the arguments used against mandating the vaccine for children; the concern is that preventing chickenpox in children – and subsequent reexposure to herpes zoster in adults – would kick off a costly wave of shingles in adults.
The study results “are themselves unable to justify for or against specific vaccination schedules, but they do suggest that revised mathematical models are required to estimate the impact of varicella vaccination, with the updated assumption that exogenous boosting is incomplete and only reduces the risk of zoster by about 30%,” noted the investigators, led by Harriet Forbes of the London School of Hygiene and Tropical Medicine.
The researchers identified 9,604 adults with a shingles diagnosis during 1997-2018 who at some point lived with a child who had chickenpox. Data came from the U.K. Clinical Practice Research Datalink, a general practice database.
They then looked at the incidence of shingles within 20 years of exposure to the sick child and compared it with the incidence before exposure and after 20 years, by which time the exogenous booster is thought to wear off. It was a self-controlled case series analysis, “a relatively novel epidemiological study design where individuals act as their own controls. Comparisons are made within individuals rather than between individuals as in a cohort or case control study,” Ms. Forbes and colleagues explained.
After adjustment for age, calendar time, and season, they found that in the 2 years after household exposure to a child with varicella, adults were 33% less likely to develop zoster (incidence ratio 0.67, 95% confidence interval 0.62-0.73), and 27% less likely from 10 to 20 years (IR 0.73, CI 0.62-0.87). The boosting effect appeared to be stronger in men.
“Exogenous boosting provides some protection from the risk of herpes zoster, but not complete immunity, as assumed by previous cost effectiveness estimates of varicella immunization,” the researchers said.
More than two-thirds of the adults with shingles were women, which fits with previous reports. Median age of exposure to a child with varicella was 38 years.
Ms. Forbes and colleagues noted that “the study design required patients with zoster to be living with a child with varicella, therefore the study cohort is younger than a general population with zoster. ... However, when we restricted our analysis to adults aged 50 and older at exposure to varicella, a similar pattern of association was observed, with no evidence of effect modification by age. This suggests that although the median age of our study cohort ... was low, the findings can be generalized to older people.”
There was no external funding for the work, and the lead investigator had no relevant financial disclosures. One investigator reported research grants from GSK and Merck, both makers of chickenpox and shingles vaccines.
SOURCE: Forbes H et al. BMJ. 2020 Jan 22;368:l6987.
Exposure to children with chickenpox reduces the incidence of shingles in adults 33% over 2 years, and 27% out to 20 years, according to British investigators.
Being exposed to children with illness due to varicella infection acts as an “exogenous booster” in adults who had chickenpox themselves as children, making shingles less likely, they explained in a BMJ article.
Although that’s good news, it’s been reported previously that exposure to children with chickenpox confers complete protection against shingles in adults for years afterward.
The finding matters in the United Kingdom because varicella vaccine is not part of the pediatric immunization schedule. The United States is the only country that mandates two shots as a requirement for children to attend school.
The United Kingdom, however, is reconsidering its policy. In the past, the exogenous booster idea has been one of the arguments used against mandating the vaccine for children; the concern is that preventing chickenpox in children – and subsequent reexposure to herpes zoster in adults – would kick off a costly wave of shingles in adults.
The study results “are themselves unable to justify for or against specific vaccination schedules, but they do suggest that revised mathematical models are required to estimate the impact of varicella vaccination, with the updated assumption that exogenous boosting is incomplete and only reduces the risk of zoster by about 30%,” noted the investigators, led by Harriet Forbes of the London School of Hygiene and Tropical Medicine.
The researchers identified 9,604 adults with a shingles diagnosis during 1997-2018 who at some point lived with a child who had chickenpox. Data came from the U.K. Clinical Practice Research Datalink, a general practice database.
They then looked at the incidence of shingles within 20 years of exposure to the sick child and compared it with the incidence before exposure and after 20 years, by which time the exogenous booster is thought to wear off. It was a self-controlled case series analysis, “a relatively novel epidemiological study design where individuals act as their own controls. Comparisons are made within individuals rather than between individuals as in a cohort or case control study,” Ms. Forbes and colleagues explained.
After adjustment for age, calendar time, and season, they found that in the 2 years after household exposure to a child with varicella, adults were 33% less likely to develop zoster (incidence ratio 0.67, 95% confidence interval 0.62-0.73), and 27% less likely from 10 to 20 years (IR 0.73, CI 0.62-0.87). The boosting effect appeared to be stronger in men.
“Exogenous boosting provides some protection from the risk of herpes zoster, but not complete immunity, as assumed by previous cost effectiveness estimates of varicella immunization,” the researchers said.
More than two-thirds of the adults with shingles were women, which fits with previous reports. Median age of exposure to a child with varicella was 38 years.
Ms. Forbes and colleagues noted that “the study design required patients with zoster to be living with a child with varicella, therefore the study cohort is younger than a general population with zoster. ... However, when we restricted our analysis to adults aged 50 and older at exposure to varicella, a similar pattern of association was observed, with no evidence of effect modification by age. This suggests that although the median age of our study cohort ... was low, the findings can be generalized to older people.”
There was no external funding for the work, and the lead investigator had no relevant financial disclosures. One investigator reported research grants from GSK and Merck, both makers of chickenpox and shingles vaccines.
SOURCE: Forbes H et al. BMJ. 2020 Jan 22;368:l6987.
Exposure to children with chickenpox reduces the incidence of shingles in adults 33% over 2 years, and 27% out to 20 years, according to British investigators.
Being exposed to children with illness due to varicella infection acts as an “exogenous booster” in adults who had chickenpox themselves as children, making shingles less likely, they explained in a BMJ article.
Although that’s good news, it’s been reported previously that exposure to children with chickenpox confers complete protection against shingles in adults for years afterward.
The finding matters in the United Kingdom because varicella vaccine is not part of the pediatric immunization schedule. The United States is the only country that mandates two shots as a requirement for children to attend school.
The United Kingdom, however, is reconsidering its policy. In the past, the exogenous booster idea has been one of the arguments used against mandating the vaccine for children; the concern is that preventing chickenpox in children – and subsequent reexposure to herpes zoster in adults – would kick off a costly wave of shingles in adults.
The study results “are themselves unable to justify for or against specific vaccination schedules, but they do suggest that revised mathematical models are required to estimate the impact of varicella vaccination, with the updated assumption that exogenous boosting is incomplete and only reduces the risk of zoster by about 30%,” noted the investigators, led by Harriet Forbes of the London School of Hygiene and Tropical Medicine.
The researchers identified 9,604 adults with a shingles diagnosis during 1997-2018 who at some point lived with a child who had chickenpox. Data came from the U.K. Clinical Practice Research Datalink, a general practice database.
They then looked at the incidence of shingles within 20 years of exposure to the sick child and compared it with the incidence before exposure and after 20 years, by which time the exogenous booster is thought to wear off. It was a self-controlled case series analysis, “a relatively novel epidemiological study design where individuals act as their own controls. Comparisons are made within individuals rather than between individuals as in a cohort or case control study,” Ms. Forbes and colleagues explained.
After adjustment for age, calendar time, and season, they found that in the 2 years after household exposure to a child with varicella, adults were 33% less likely to develop zoster (incidence ratio 0.67, 95% confidence interval 0.62-0.73), and 27% less likely from 10 to 20 years (IR 0.73, CI 0.62-0.87). The boosting effect appeared to be stronger in men.
“Exogenous boosting provides some protection from the risk of herpes zoster, but not complete immunity, as assumed by previous cost effectiveness estimates of varicella immunization,” the researchers said.
More than two-thirds of the adults with shingles were women, which fits with previous reports. Median age of exposure to a child with varicella was 38 years.
Ms. Forbes and colleagues noted that “the study design required patients with zoster to be living with a child with varicella, therefore the study cohort is younger than a general population with zoster. ... However, when we restricted our analysis to adults aged 50 and older at exposure to varicella, a similar pattern of association was observed, with no evidence of effect modification by age. This suggests that although the median age of our study cohort ... was low, the findings can be generalized to older people.”
There was no external funding for the work, and the lead investigator had no relevant financial disclosures. One investigator reported research grants from GSK and Merck, both makers of chickenpox and shingles vaccines.
SOURCE: Forbes H et al. BMJ. 2020 Jan 22;368:l6987.
FROM BMJ
Race, ethnicity may influence outcomes after supratentorial intracerebral hemorrhage
researchers reported Jan. 22 in Neurology.
“There has been considerable research on stroke in older people, but there is still much to be learned about stroke in younger people and how it affects people of different races and ethnicities,” study author Daniel Woo, MD, professor of neurology at the University of Cincinnati, said in a news release. “Our study found that, even when you account for factors that affect outcomes, such as how big the stroke is, race and ethnicity were still independent predictors of how well people would recover.”
A subset of ERICH participants
To examine predictors of functional outcome in young patients with ICH, researchers analyzed data from a subset of patients in the Ethnic/Racial Variations in Intracerebral Hemorrhage (ERICH) study. ERICH enrolled patients with nontraumatic ICHs at 42 sites in the United States. It included 1,000 non-Hispanic black patients, 1,000 non-Hispanic white patients, and 1,000 Hispanic patients. Participants self-reported race and ethnicity.
Lead author Laura C. Miyares from Yale School of Medicine in New Haven, Conn., and colleagues analyzed data from 418 patients in ERICH who were aged 18-49 years and had supratentorial ICH. The cohort had an average age of 43 years, and 69% were male. In this subset, 41% were black, 12% were white, and 47% were Hispanic.
The primary outcome was modified Rankin Scale (mRS) score 3 months after the ICH, and the investigators defined a poor outcome as a score of 4 or greater. At 3 months, 35% had a poor functional outcome. Approximately 18% were unable to walk without assistance and attend to their bodily needs (mRS 4); 8% were bedridden, incontinent, and required nursing care (mRS 5); and 10% had died (mRS 6).
The percentage of patients with a poor functional outcome was 52% among white patients, 35% among black patients, and 31% among Hispanic patients. In a univariable analysis, black patients had a 51% reduction in odds of a poor outcome, compared with white patients, and Hispanic patients had a 59% reduction.
“The association between race/ethnicity and 3-month post-ICH functional outcome remained significant after adjusting for age, sex, premorbid disability, ICH location, ICH volume, [intraventricular hemorrhage] extension, systolic blood pressure, and [Glasgow Coma Scale] score on admission,” the researchers said. “In multivariable analysis, using white patients as the reference category, black patients had a 58% reduction in the odds of poor functional outcome at 3 months and Hispanic patients had a 66% reduction in the same odds.”
Their analysis identified the importance of other risk factors as well. “The volume of the hematoma, the most powerful predictor of outcome in older patients with ICH, was also found to be the most significant predictor of poor outcome in young patients,” they said.
Vascular risks and oral anticoagulants
About 80% of the young adults with ICH had a history of diagnosed hypertension. In nearly half, the condition was untreated. “After hypertension, the most common stroke risk factors in the young were diabetes, high cholesterol, smoking, and alcohol abuse,” the authors said. “In combination, these results indicate that vascular risk factors, especially untreated, could explain a large proportion of cases of ICH in the young.”
“Our results also point to treatment with oral anticoagulants before hospitalization as a potential mediator of the effect of race/ethnicity on short-term functional outcomes,” they said. About 8% of the white patients used oral anticoagulants, compared with 4% of the black patients and 1% of the Hispanic patients. Oral anticoagulant treatment “is a known risk factor for ICH and an established predictor of poor outcome in this condition. However, because only a small proportion of enrolled young patients with ICH were on [oral anticoagulants] prior to presentation, these results should be further validated by future studies.”
The study’s limitations include the broad categorization of racial and ethnic groups, the fact that younger patients with supratentorial ICH were more likely to be black or Hispanic and less likely to be white, and the exclusion of a significant proportion of cases of young white patients with smaller ICH volumes because of missing data, the researchers noted. Although the cohort was large, researchers may need to study more patients to capture differences among racial and ethnic groups, the investigators said.
The association between race/ethnicity and functional outcome could relate to “distinct pathophysiologies of the initial bleed or unique mechanisms of secondary injury,” the researchers suggested. “Future studies are necessary to probe the potential biological and social mediators of these findings to elucidate the role of race/ethnicity in ICH severity and functional recovery, and to develop improved prognostication for a racially varied population.”
ERICH is supported by the National Institute of Neurological Disorders and Stroke. Authors disclosed grants from the government, professional societies, and a university.
SOURCE: Miyares LC et al. Neurology. 2020 Jan 22. doi: 10.1212/WNL.0000000000008930.
researchers reported Jan. 22 in Neurology.
“There has been considerable research on stroke in older people, but there is still much to be learned about stroke in younger people and how it affects people of different races and ethnicities,” study author Daniel Woo, MD, professor of neurology at the University of Cincinnati, said in a news release. “Our study found that, even when you account for factors that affect outcomes, such as how big the stroke is, race and ethnicity were still independent predictors of how well people would recover.”
A subset of ERICH participants
To examine predictors of functional outcome in young patients with ICH, researchers analyzed data from a subset of patients in the Ethnic/Racial Variations in Intracerebral Hemorrhage (ERICH) study. ERICH enrolled patients with nontraumatic ICHs at 42 sites in the United States. It included 1,000 non-Hispanic black patients, 1,000 non-Hispanic white patients, and 1,000 Hispanic patients. Participants self-reported race and ethnicity.
Lead author Laura C. Miyares from Yale School of Medicine in New Haven, Conn., and colleagues analyzed data from 418 patients in ERICH who were aged 18-49 years and had supratentorial ICH. The cohort had an average age of 43 years, and 69% were male. In this subset, 41% were black, 12% were white, and 47% were Hispanic.
The primary outcome was modified Rankin Scale (mRS) score 3 months after the ICH, and the investigators defined a poor outcome as a score of 4 or greater. At 3 months, 35% had a poor functional outcome. Approximately 18% were unable to walk without assistance and attend to their bodily needs (mRS 4); 8% were bedridden, incontinent, and required nursing care (mRS 5); and 10% had died (mRS 6).
The percentage of patients with a poor functional outcome was 52% among white patients, 35% among black patients, and 31% among Hispanic patients. In a univariable analysis, black patients had a 51% reduction in odds of a poor outcome, compared with white patients, and Hispanic patients had a 59% reduction.
“The association between race/ethnicity and 3-month post-ICH functional outcome remained significant after adjusting for age, sex, premorbid disability, ICH location, ICH volume, [intraventricular hemorrhage] extension, systolic blood pressure, and [Glasgow Coma Scale] score on admission,” the researchers said. “In multivariable analysis, using white patients as the reference category, black patients had a 58% reduction in the odds of poor functional outcome at 3 months and Hispanic patients had a 66% reduction in the same odds.”
Their analysis identified the importance of other risk factors as well. “The volume of the hematoma, the most powerful predictor of outcome in older patients with ICH, was also found to be the most significant predictor of poor outcome in young patients,” they said.
Vascular risks and oral anticoagulants
About 80% of the young adults with ICH had a history of diagnosed hypertension. In nearly half, the condition was untreated. “After hypertension, the most common stroke risk factors in the young were diabetes, high cholesterol, smoking, and alcohol abuse,” the authors said. “In combination, these results indicate that vascular risk factors, especially untreated, could explain a large proportion of cases of ICH in the young.”
“Our results also point to treatment with oral anticoagulants before hospitalization as a potential mediator of the effect of race/ethnicity on short-term functional outcomes,” they said. About 8% of the white patients used oral anticoagulants, compared with 4% of the black patients and 1% of the Hispanic patients. Oral anticoagulant treatment “is a known risk factor for ICH and an established predictor of poor outcome in this condition. However, because only a small proportion of enrolled young patients with ICH were on [oral anticoagulants] prior to presentation, these results should be further validated by future studies.”
The study’s limitations include the broad categorization of racial and ethnic groups, the fact that younger patients with supratentorial ICH were more likely to be black or Hispanic and less likely to be white, and the exclusion of a significant proportion of cases of young white patients with smaller ICH volumes because of missing data, the researchers noted. Although the cohort was large, researchers may need to study more patients to capture differences among racial and ethnic groups, the investigators said.
The association between race/ethnicity and functional outcome could relate to “distinct pathophysiologies of the initial bleed or unique mechanisms of secondary injury,” the researchers suggested. “Future studies are necessary to probe the potential biological and social mediators of these findings to elucidate the role of race/ethnicity in ICH severity and functional recovery, and to develop improved prognostication for a racially varied population.”
ERICH is supported by the National Institute of Neurological Disorders and Stroke. Authors disclosed grants from the government, professional societies, and a university.
SOURCE: Miyares LC et al. Neurology. 2020 Jan 22. doi: 10.1212/WNL.0000000000008930.
researchers reported Jan. 22 in Neurology.
“There has been considerable research on stroke in older people, but there is still much to be learned about stroke in younger people and how it affects people of different races and ethnicities,” study author Daniel Woo, MD, professor of neurology at the University of Cincinnati, said in a news release. “Our study found that, even when you account for factors that affect outcomes, such as how big the stroke is, race and ethnicity were still independent predictors of how well people would recover.”
A subset of ERICH participants
To examine predictors of functional outcome in young patients with ICH, researchers analyzed data from a subset of patients in the Ethnic/Racial Variations in Intracerebral Hemorrhage (ERICH) study. ERICH enrolled patients with nontraumatic ICHs at 42 sites in the United States. It included 1,000 non-Hispanic black patients, 1,000 non-Hispanic white patients, and 1,000 Hispanic patients. Participants self-reported race and ethnicity.
Lead author Laura C. Miyares from Yale School of Medicine in New Haven, Conn., and colleagues analyzed data from 418 patients in ERICH who were aged 18-49 years and had supratentorial ICH. The cohort had an average age of 43 years, and 69% were male. In this subset, 41% were black, 12% were white, and 47% were Hispanic.
The primary outcome was modified Rankin Scale (mRS) score 3 months after the ICH, and the investigators defined a poor outcome as a score of 4 or greater. At 3 months, 35% had a poor functional outcome. Approximately 18% were unable to walk without assistance and attend to their bodily needs (mRS 4); 8% were bedridden, incontinent, and required nursing care (mRS 5); and 10% had died (mRS 6).
The percentage of patients with a poor functional outcome was 52% among white patients, 35% among black patients, and 31% among Hispanic patients. In a univariable analysis, black patients had a 51% reduction in odds of a poor outcome, compared with white patients, and Hispanic patients had a 59% reduction.
“The association between race/ethnicity and 3-month post-ICH functional outcome remained significant after adjusting for age, sex, premorbid disability, ICH location, ICH volume, [intraventricular hemorrhage] extension, systolic blood pressure, and [Glasgow Coma Scale] score on admission,” the researchers said. “In multivariable analysis, using white patients as the reference category, black patients had a 58% reduction in the odds of poor functional outcome at 3 months and Hispanic patients had a 66% reduction in the same odds.”
Their analysis identified the importance of other risk factors as well. “The volume of the hematoma, the most powerful predictor of outcome in older patients with ICH, was also found to be the most significant predictor of poor outcome in young patients,” they said.
Vascular risks and oral anticoagulants
About 80% of the young adults with ICH had a history of diagnosed hypertension. In nearly half, the condition was untreated. “After hypertension, the most common stroke risk factors in the young were diabetes, high cholesterol, smoking, and alcohol abuse,” the authors said. “In combination, these results indicate that vascular risk factors, especially untreated, could explain a large proportion of cases of ICH in the young.”
“Our results also point to treatment with oral anticoagulants before hospitalization as a potential mediator of the effect of race/ethnicity on short-term functional outcomes,” they said. About 8% of the white patients used oral anticoagulants, compared with 4% of the black patients and 1% of the Hispanic patients. Oral anticoagulant treatment “is a known risk factor for ICH and an established predictor of poor outcome in this condition. However, because only a small proportion of enrolled young patients with ICH were on [oral anticoagulants] prior to presentation, these results should be further validated by future studies.”
The study’s limitations include the broad categorization of racial and ethnic groups, the fact that younger patients with supratentorial ICH were more likely to be black or Hispanic and less likely to be white, and the exclusion of a significant proportion of cases of young white patients with smaller ICH volumes because of missing data, the researchers noted. Although the cohort was large, researchers may need to study more patients to capture differences among racial and ethnic groups, the investigators said.
The association between race/ethnicity and functional outcome could relate to “distinct pathophysiologies of the initial bleed or unique mechanisms of secondary injury,” the researchers suggested. “Future studies are necessary to probe the potential biological and social mediators of these findings to elucidate the role of race/ethnicity in ICH severity and functional recovery, and to develop improved prognostication for a racially varied population.”
ERICH is supported by the National Institute of Neurological Disorders and Stroke. Authors disclosed grants from the government, professional societies, and a university.
SOURCE: Miyares LC et al. Neurology. 2020 Jan 22. doi: 10.1212/WNL.0000000000008930.
FROM NEUROLOGY
Key clinical point: Among young adults with supratentorial intracerebral hemorrhage (ICH), black race and Hispanic ethnicity are associated with better functional outcomes, compared with white race.
Major finding: In multivariable analysis, black patients had a 58% reduction in the odds of poor functional outcome at 3 months, compared with white patients, and Hispanic patients had a 66% reduction.
Study details: An analysis of data from a subset of 418 patients in the Ethnic/Racial Variations in Intracerebral Hemorrhage (ERICH) study.
Disclosures: ERICH is supported by the National Institute of Neurological Disorders and Stroke. Authors disclosed grants from the government, professional societies, and a university.
Source: Miyares LC et al. Neurology. 2020 Jan 22. doi: 10.1212/WNL.0000000000008930.