User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
New child COVID-19 cases decline as total passes 3 million
New COVID-19 cases in children continue to drop each week, but the total number of cases has now surpassed 3 million since the start of the pandemic, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
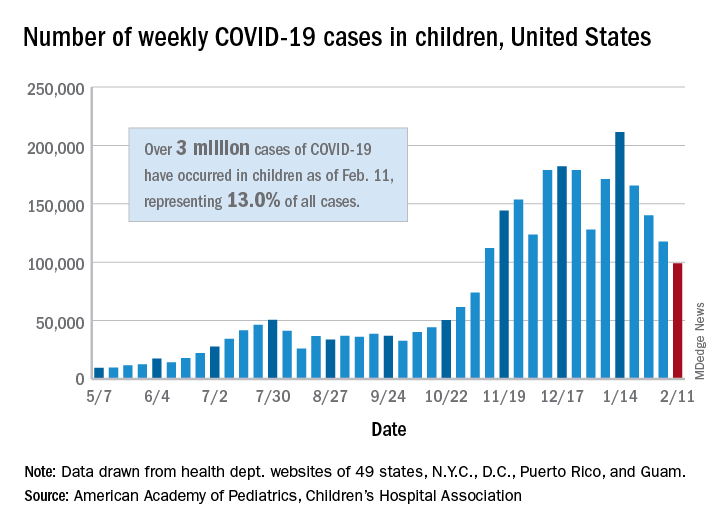
It was still enough, though, to bring the total to 3.03 million children infected with SARS-CoV-19 in the United States, the AAP and the CHA said in their weekly report.
The nation also hit a couple of other ignominious milestones. The cumulative rate of COVID-19 infection now stands at 4,030 per 100,000, so 4% of all children have been infected. Also, children represented 16.9% of all new cases for the week, which equals the highest proportion seen throughout the pandemic, based on data from health departments in 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
There have been 241 COVID-19–related deaths in children so far, with 14 reported during the week of Feb. 5-11. Kansas just recorded its first pediatric death, which leaves 10 states that have had no fatalities. Texas, with 39 deaths, has had more than any other state, among the 43 that are reporting mortality by age, the AAP/CHA report showed.
New COVID-19 cases in children continue to drop each week, but the total number of cases has now surpassed 3 million since the start of the pandemic, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

It was still enough, though, to bring the total to 3.03 million children infected with SARS-CoV-19 in the United States, the AAP and the CHA said in their weekly report.
The nation also hit a couple of other ignominious milestones. The cumulative rate of COVID-19 infection now stands at 4,030 per 100,000, so 4% of all children have been infected. Also, children represented 16.9% of all new cases for the week, which equals the highest proportion seen throughout the pandemic, based on data from health departments in 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
There have been 241 COVID-19–related deaths in children so far, with 14 reported during the week of Feb. 5-11. Kansas just recorded its first pediatric death, which leaves 10 states that have had no fatalities. Texas, with 39 deaths, has had more than any other state, among the 43 that are reporting mortality by age, the AAP/CHA report showed.
New COVID-19 cases in children continue to drop each week, but the total number of cases has now surpassed 3 million since the start of the pandemic, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

It was still enough, though, to bring the total to 3.03 million children infected with SARS-CoV-19 in the United States, the AAP and the CHA said in their weekly report.
The nation also hit a couple of other ignominious milestones. The cumulative rate of COVID-19 infection now stands at 4,030 per 100,000, so 4% of all children have been infected. Also, children represented 16.9% of all new cases for the week, which equals the highest proportion seen throughout the pandemic, based on data from health departments in 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
There have been 241 COVID-19–related deaths in children so far, with 14 reported during the week of Feb. 5-11. Kansas just recorded its first pediatric death, which leaves 10 states that have had no fatalities. Texas, with 39 deaths, has had more than any other state, among the 43 that are reporting mortality by age, the AAP/CHA report showed.
One-third of health care workers leery of getting COVID-19 vaccine, survey shows
Moreover, 54% of direct care providers indicated that they would take the vaccine if offered, compared with 60% of noncare providers.
The findings come from what is believed to be the largest survey of health care provider attitudes toward COVID-19 vaccination, published online Jan. 25 in Clinical Infectious Diseases.
“We have shown that self-reported willingness to receive vaccination against COVID-19 differs by age, gender, race and hospital role, with physicians and research scientists showing the highest acceptance,” Jana Shaw, MD, MPH, State University of New York, Syracuse, N.Y, the study’s corresponding author, told this news organization. “Building trust in authorities and confidence in vaccines is a complex and time-consuming process that requires commitment and resources. We have to make those investments as hesitancy can severely undermine vaccination coverage. Because health care providers are members of our communities, it is possible that their views are shared by the public at large. Our findings can assist public health professionals as a starting point of discussion and engagement with communities to ensure that we vaccinate at least 80% of the public to end the pandemic.”
For the study, Dr. Shaw and her colleagues emailed an anonymous survey to 9,565 employees of State University of New York Upstate Medical University, Syracuse, an academic medical center that cares for an estimated 1.8 million people. The survey, which contained questions intended to evaluate attitudes, belief, and willingness to get vaccinated, took place between Nov. 23 and Dec. 5, about a week before the U.S. Food and Drug Administration granted the first emergency use authorization for the Pfizer-BioNTech BNT162b2 mRNA vaccine.
Survey recipients included physicians, nurse practitioners, physician assistants, nurses, pharmacists, medical and nursing students, allied health professionals, and nonclinical ancillary staff.
Of the 9,565 surveys sent, 5,287 responses were collected and used in the final analysis, for a response rate of 55%. The mean age of respondents was 43, 73% were female, 85% were White, 6% were Asian, 5% were Black/African American, and the rest were Native American, Native Hawaiian/Pacific Islander, or from other races. More than half of respondents (59%) reported that they provided direct patient care, and 32% said they provided care for patients with COVID-19.
Of all survey respondents, 58% expressed their intent to receive a COVID-19 vaccine, but this varied by their role in the health care system. For example, in response to the statement, “If a vaccine were offered free of charge, I would take it,” 80% of scientists and physicians agreed that they would, while colleagues in other roles were unsure whether they would take the vaccine, including 34% of registered nurses, 32% of allied health professionals, and 32% of master’s-level clinicians. These differences across roles were significant (P less than .001).
The researchers also found that direct patient care or care for COVID-19 patients was associated with lower vaccination intent. For example, 54% of direct care providers and 62% of non-care providers indicated they would take the vaccine if offered, compared with 52% of those who had provided care for COVID-19 patients vs. 61% of those who had not (P less than .001).
“This was a really surprising finding,” said Dr. Shaw, who is a pediatric infectious diseases physician at SUNY Upstate. “In general, one would expect that perceived severity of disease would lead to a greater desire to get vaccinated. Because our question did not address severity of disease, it is possible that we oversampled respondents who took care of patients with mild disease (i.e., in an outpatient setting). This could have led to an underestimation of disease severity and resulted in lower vaccination intent.”
A focus on rebuilding trust
Survey respondents who agreed or strongly agreed that they would accept a vaccine were older (a mean age of 44 years), compared with those who were not sure or who disagreed (a mean age of 42 vs. 38 years, respectively; P less than .001). In addition, fewer females agreed or strongly agreed that they would accept a vaccine (54% vs. 73% of males), whereas those who self-identified as Black/African American were least likely to want to get vaccinated, compared with those from other ethnic groups (31%, compared with 74% of Asians, 58% of Whites, and 39% of American Indians or Alaska Natives).
“We are deeply aware of the poor decisions scientists made in the past, which led to a prevailing skepticism and ‘feeling like guinea pigs’ among people of color, especially Black adults,” Dr. Shaw said. “Black adults are less likely, compared [with] White adults, to have confidence that scientists act in the public interest. Rebuilding trust will take time and has to start with addressing health care disparities. In addition, we need to acknowledge contributions of Black researchers to science. For example, until recently very few knew that the Moderna vaccine was developed [with the help of] Dr. Kizzmekia Corbett, who is Black.”
The top five main areas of unease that all respondents expressed about a COVID-19 vaccine were concern about adverse events/side effects (47%), efficacy (15%), rushed release (11%), safety (11%), and the research and authorization process (3%).
“I think it is important that fellow clinicians recognize that, in order to boost vaccine confidence we will need careful, individually tailored communication strategies,” Dr. Shaw said. “A consideration should be given to those [strategies] that utilize interpersonal channels that deliver leadership by example and leverage influencers in the institution to encourage wider adoption of vaccination.”
Aaron M. Milstone, MD, MHS, asked to comment on the research, recommended that health care workers advocate for the vaccine and encourage their patients, friends, and loved ones to get vaccinated. “Soon, COVID-19 will have taken more than half a million lives in the U.S.,” said Dr. Milstone, a pediatric epidemiologist at Johns Hopkins University, Baltimore. “Although vaccines can have side effects like fever and muscle aches, and very, very rare more serious side effects, the risks of dying from COVID are much greater than the risk of a serious vaccine reaction. The study’s authors shed light on the ongoing need for leaders of all communities to support the COVID vaccines, not just the scientific community, but religious leaders, political leaders, and community leaders.”
Addressing vaccine hesitancy
Informed by their own survey, Dr. Shaw and her colleagues have developed a plan to address vaccine hesitancy to ensure high vaccine uptake at SUNY Upstate. Those strategies include, but aren’t limited to, institution-wide forums for all employees on COVID-19 vaccine safety, risks, and benefits followed by Q&A sessions, grand rounds for providers summarizing clinical trial data on mRNA vaccines, development of an Ask COVID email line for staff to ask vaccine-related questions, and a detailed vaccine-specific FAQ document.
In addition, SUNY Upstate experts have engaged in numerous media interviews to provide education and updates on the benefits of vaccination to public and staff, stationary vaccine locations, and mobile COVID-19 vaccine carts. “To date, the COVID-19 vaccination process has been well received, and we anticipate strong vaccine uptake,” she said.
Dr. Shaw acknowledged certain limitations of the survey, including its cross-sectional design and the fact that it was conducted in a single health care system in the northeastern United States. “Thus, generalizability to other regions of the U.S. and other countries may be limited,” Dr. Shaw said. “The study was also conducted before EUA [emergency use authorization] was granted to either the Moderna or Pfizer-BioNTech vaccines. It is therefore likely that vaccine acceptance will change over time as more people get vaccinated.”
The authors have disclosed no relevant financial relationships. Dr. Milstone disclosed that he has received a research grant from Merck, but it is not related to vaccines.
A version of this article first appeared on Medscape.com.
Moreover, 54% of direct care providers indicated that they would take the vaccine if offered, compared with 60% of noncare providers.
The findings come from what is believed to be the largest survey of health care provider attitudes toward COVID-19 vaccination, published online Jan. 25 in Clinical Infectious Diseases.
“We have shown that self-reported willingness to receive vaccination against COVID-19 differs by age, gender, race and hospital role, with physicians and research scientists showing the highest acceptance,” Jana Shaw, MD, MPH, State University of New York, Syracuse, N.Y, the study’s corresponding author, told this news organization. “Building trust in authorities and confidence in vaccines is a complex and time-consuming process that requires commitment and resources. We have to make those investments as hesitancy can severely undermine vaccination coverage. Because health care providers are members of our communities, it is possible that their views are shared by the public at large. Our findings can assist public health professionals as a starting point of discussion and engagement with communities to ensure that we vaccinate at least 80% of the public to end the pandemic.”
For the study, Dr. Shaw and her colleagues emailed an anonymous survey to 9,565 employees of State University of New York Upstate Medical University, Syracuse, an academic medical center that cares for an estimated 1.8 million people. The survey, which contained questions intended to evaluate attitudes, belief, and willingness to get vaccinated, took place between Nov. 23 and Dec. 5, about a week before the U.S. Food and Drug Administration granted the first emergency use authorization for the Pfizer-BioNTech BNT162b2 mRNA vaccine.
Survey recipients included physicians, nurse practitioners, physician assistants, nurses, pharmacists, medical and nursing students, allied health professionals, and nonclinical ancillary staff.
Of the 9,565 surveys sent, 5,287 responses were collected and used in the final analysis, for a response rate of 55%. The mean age of respondents was 43, 73% were female, 85% were White, 6% were Asian, 5% were Black/African American, and the rest were Native American, Native Hawaiian/Pacific Islander, or from other races. More than half of respondents (59%) reported that they provided direct patient care, and 32% said they provided care for patients with COVID-19.
Of all survey respondents, 58% expressed their intent to receive a COVID-19 vaccine, but this varied by their role in the health care system. For example, in response to the statement, “If a vaccine were offered free of charge, I would take it,” 80% of scientists and physicians agreed that they would, while colleagues in other roles were unsure whether they would take the vaccine, including 34% of registered nurses, 32% of allied health professionals, and 32% of master’s-level clinicians. These differences across roles were significant (P less than .001).
The researchers also found that direct patient care or care for COVID-19 patients was associated with lower vaccination intent. For example, 54% of direct care providers and 62% of non-care providers indicated they would take the vaccine if offered, compared with 52% of those who had provided care for COVID-19 patients vs. 61% of those who had not (P less than .001).
“This was a really surprising finding,” said Dr. Shaw, who is a pediatric infectious diseases physician at SUNY Upstate. “In general, one would expect that perceived severity of disease would lead to a greater desire to get vaccinated. Because our question did not address severity of disease, it is possible that we oversampled respondents who took care of patients with mild disease (i.e., in an outpatient setting). This could have led to an underestimation of disease severity and resulted in lower vaccination intent.”
A focus on rebuilding trust
Survey respondents who agreed or strongly agreed that they would accept a vaccine were older (a mean age of 44 years), compared with those who were not sure or who disagreed (a mean age of 42 vs. 38 years, respectively; P less than .001). In addition, fewer females agreed or strongly agreed that they would accept a vaccine (54% vs. 73% of males), whereas those who self-identified as Black/African American were least likely to want to get vaccinated, compared with those from other ethnic groups (31%, compared with 74% of Asians, 58% of Whites, and 39% of American Indians or Alaska Natives).
“We are deeply aware of the poor decisions scientists made in the past, which led to a prevailing skepticism and ‘feeling like guinea pigs’ among people of color, especially Black adults,” Dr. Shaw said. “Black adults are less likely, compared [with] White adults, to have confidence that scientists act in the public interest. Rebuilding trust will take time and has to start with addressing health care disparities. In addition, we need to acknowledge contributions of Black researchers to science. For example, until recently very few knew that the Moderna vaccine was developed [with the help of] Dr. Kizzmekia Corbett, who is Black.”
The top five main areas of unease that all respondents expressed about a COVID-19 vaccine were concern about adverse events/side effects (47%), efficacy (15%), rushed release (11%), safety (11%), and the research and authorization process (3%).
“I think it is important that fellow clinicians recognize that, in order to boost vaccine confidence we will need careful, individually tailored communication strategies,” Dr. Shaw said. “A consideration should be given to those [strategies] that utilize interpersonal channels that deliver leadership by example and leverage influencers in the institution to encourage wider adoption of vaccination.”
Aaron M. Milstone, MD, MHS, asked to comment on the research, recommended that health care workers advocate for the vaccine and encourage their patients, friends, and loved ones to get vaccinated. “Soon, COVID-19 will have taken more than half a million lives in the U.S.,” said Dr. Milstone, a pediatric epidemiologist at Johns Hopkins University, Baltimore. “Although vaccines can have side effects like fever and muscle aches, and very, very rare more serious side effects, the risks of dying from COVID are much greater than the risk of a serious vaccine reaction. The study’s authors shed light on the ongoing need for leaders of all communities to support the COVID vaccines, not just the scientific community, but religious leaders, political leaders, and community leaders.”
Addressing vaccine hesitancy
Informed by their own survey, Dr. Shaw and her colleagues have developed a plan to address vaccine hesitancy to ensure high vaccine uptake at SUNY Upstate. Those strategies include, but aren’t limited to, institution-wide forums for all employees on COVID-19 vaccine safety, risks, and benefits followed by Q&A sessions, grand rounds for providers summarizing clinical trial data on mRNA vaccines, development of an Ask COVID email line for staff to ask vaccine-related questions, and a detailed vaccine-specific FAQ document.
In addition, SUNY Upstate experts have engaged in numerous media interviews to provide education and updates on the benefits of vaccination to public and staff, stationary vaccine locations, and mobile COVID-19 vaccine carts. “To date, the COVID-19 vaccination process has been well received, and we anticipate strong vaccine uptake,” she said.
Dr. Shaw acknowledged certain limitations of the survey, including its cross-sectional design and the fact that it was conducted in a single health care system in the northeastern United States. “Thus, generalizability to other regions of the U.S. and other countries may be limited,” Dr. Shaw said. “The study was also conducted before EUA [emergency use authorization] was granted to either the Moderna or Pfizer-BioNTech vaccines. It is therefore likely that vaccine acceptance will change over time as more people get vaccinated.”
The authors have disclosed no relevant financial relationships. Dr. Milstone disclosed that he has received a research grant from Merck, but it is not related to vaccines.
A version of this article first appeared on Medscape.com.
Moreover, 54% of direct care providers indicated that they would take the vaccine if offered, compared with 60% of noncare providers.
The findings come from what is believed to be the largest survey of health care provider attitudes toward COVID-19 vaccination, published online Jan. 25 in Clinical Infectious Diseases.
“We have shown that self-reported willingness to receive vaccination against COVID-19 differs by age, gender, race and hospital role, with physicians and research scientists showing the highest acceptance,” Jana Shaw, MD, MPH, State University of New York, Syracuse, N.Y, the study’s corresponding author, told this news organization. “Building trust in authorities and confidence in vaccines is a complex and time-consuming process that requires commitment and resources. We have to make those investments as hesitancy can severely undermine vaccination coverage. Because health care providers are members of our communities, it is possible that their views are shared by the public at large. Our findings can assist public health professionals as a starting point of discussion and engagement with communities to ensure that we vaccinate at least 80% of the public to end the pandemic.”
For the study, Dr. Shaw and her colleagues emailed an anonymous survey to 9,565 employees of State University of New York Upstate Medical University, Syracuse, an academic medical center that cares for an estimated 1.8 million people. The survey, which contained questions intended to evaluate attitudes, belief, and willingness to get vaccinated, took place between Nov. 23 and Dec. 5, about a week before the U.S. Food and Drug Administration granted the first emergency use authorization for the Pfizer-BioNTech BNT162b2 mRNA vaccine.
Survey recipients included physicians, nurse practitioners, physician assistants, nurses, pharmacists, medical and nursing students, allied health professionals, and nonclinical ancillary staff.
Of the 9,565 surveys sent, 5,287 responses were collected and used in the final analysis, for a response rate of 55%. The mean age of respondents was 43, 73% were female, 85% were White, 6% were Asian, 5% were Black/African American, and the rest were Native American, Native Hawaiian/Pacific Islander, or from other races. More than half of respondents (59%) reported that they provided direct patient care, and 32% said they provided care for patients with COVID-19.
Of all survey respondents, 58% expressed their intent to receive a COVID-19 vaccine, but this varied by their role in the health care system. For example, in response to the statement, “If a vaccine were offered free of charge, I would take it,” 80% of scientists and physicians agreed that they would, while colleagues in other roles were unsure whether they would take the vaccine, including 34% of registered nurses, 32% of allied health professionals, and 32% of master’s-level clinicians. These differences across roles were significant (P less than .001).
The researchers also found that direct patient care or care for COVID-19 patients was associated with lower vaccination intent. For example, 54% of direct care providers and 62% of non-care providers indicated they would take the vaccine if offered, compared with 52% of those who had provided care for COVID-19 patients vs. 61% of those who had not (P less than .001).
“This was a really surprising finding,” said Dr. Shaw, who is a pediatric infectious diseases physician at SUNY Upstate. “In general, one would expect that perceived severity of disease would lead to a greater desire to get vaccinated. Because our question did not address severity of disease, it is possible that we oversampled respondents who took care of patients with mild disease (i.e., in an outpatient setting). This could have led to an underestimation of disease severity and resulted in lower vaccination intent.”
A focus on rebuilding trust
Survey respondents who agreed or strongly agreed that they would accept a vaccine were older (a mean age of 44 years), compared with those who were not sure or who disagreed (a mean age of 42 vs. 38 years, respectively; P less than .001). In addition, fewer females agreed or strongly agreed that they would accept a vaccine (54% vs. 73% of males), whereas those who self-identified as Black/African American were least likely to want to get vaccinated, compared with those from other ethnic groups (31%, compared with 74% of Asians, 58% of Whites, and 39% of American Indians or Alaska Natives).
“We are deeply aware of the poor decisions scientists made in the past, which led to a prevailing skepticism and ‘feeling like guinea pigs’ among people of color, especially Black adults,” Dr. Shaw said. “Black adults are less likely, compared [with] White adults, to have confidence that scientists act in the public interest. Rebuilding trust will take time and has to start with addressing health care disparities. In addition, we need to acknowledge contributions of Black researchers to science. For example, until recently very few knew that the Moderna vaccine was developed [with the help of] Dr. Kizzmekia Corbett, who is Black.”
The top five main areas of unease that all respondents expressed about a COVID-19 vaccine were concern about adverse events/side effects (47%), efficacy (15%), rushed release (11%), safety (11%), and the research and authorization process (3%).
“I think it is important that fellow clinicians recognize that, in order to boost vaccine confidence we will need careful, individually tailored communication strategies,” Dr. Shaw said. “A consideration should be given to those [strategies] that utilize interpersonal channels that deliver leadership by example and leverage influencers in the institution to encourage wider adoption of vaccination.”
Aaron M. Milstone, MD, MHS, asked to comment on the research, recommended that health care workers advocate for the vaccine and encourage their patients, friends, and loved ones to get vaccinated. “Soon, COVID-19 will have taken more than half a million lives in the U.S.,” said Dr. Milstone, a pediatric epidemiologist at Johns Hopkins University, Baltimore. “Although vaccines can have side effects like fever and muscle aches, and very, very rare more serious side effects, the risks of dying from COVID are much greater than the risk of a serious vaccine reaction. The study’s authors shed light on the ongoing need for leaders of all communities to support the COVID vaccines, not just the scientific community, but religious leaders, political leaders, and community leaders.”
Addressing vaccine hesitancy
Informed by their own survey, Dr. Shaw and her colleagues have developed a plan to address vaccine hesitancy to ensure high vaccine uptake at SUNY Upstate. Those strategies include, but aren’t limited to, institution-wide forums for all employees on COVID-19 vaccine safety, risks, and benefits followed by Q&A sessions, grand rounds for providers summarizing clinical trial data on mRNA vaccines, development of an Ask COVID email line for staff to ask vaccine-related questions, and a detailed vaccine-specific FAQ document.
In addition, SUNY Upstate experts have engaged in numerous media interviews to provide education and updates on the benefits of vaccination to public and staff, stationary vaccine locations, and mobile COVID-19 vaccine carts. “To date, the COVID-19 vaccination process has been well received, and we anticipate strong vaccine uptake,” she said.
Dr. Shaw acknowledged certain limitations of the survey, including its cross-sectional design and the fact that it was conducted in a single health care system in the northeastern United States. “Thus, generalizability to other regions of the U.S. and other countries may be limited,” Dr. Shaw said. “The study was also conducted before EUA [emergency use authorization] was granted to either the Moderna or Pfizer-BioNTech vaccines. It is therefore likely that vaccine acceptance will change over time as more people get vaccinated.”
The authors have disclosed no relevant financial relationships. Dr. Milstone disclosed that he has received a research grant from Merck, but it is not related to vaccines.
A version of this article first appeared on Medscape.com.
FDA approves first drug that protects against chemo-induced myelosuppression
A novel drug that offers multilineage protection from chemotherapy-induced myelosuppression has been approved by the Food and Drug Administration.
The drug, trilaciclib (Cosela, G1 Therapeutics) is administered intravenously as a 30-minute infusion within 4 hours prior to the start of chemotherapy. It is indicated specifically for use in adults with extensive-stage small-cell lung cancer (ES-SCLC) who are receiving chemotherapy.
Trilaciclib is a CDK4/6 inhibitor, and this action appears to protect normal bone marrow cells from the harmful effects of chemotherapy.
“For patients with extensive-stage small-cell lung cancer, protecting bone marrow function may help make their chemotherapy safer and allow them to complete their course of treatment on time and according to plan,” Albert Deisseroth, MD, PhD, of the FDA’s Center for Drug Evaluation and Research, said in an FDA press release.
First drug of its type
Trilaciclib “is the first and only therapy designed to help protect bone marrow (myeloprotection) when administered prior to treatment with chemotherapy,” according to the drug’s manufacturer.
Myelosuppression is one of the most severe adverse effects of chemotherapy, and it can be life-threatening. It can increase the risk of infection and lead to severe anemia and/or bleeding.
“These complications impact patients’ quality of life and may also result in chemotherapy dose reductions and delays,” Jeffrey Crawford, MD, of Duke Cancer Institute, Durham, N.C., said in a company press release.
“To date, approaches have included the use of growth factor agents to accelerate blood cell recovery after the bone marrow injury has occurred, along with antibiotics and transfusions as needed. By contrast, trilaciclib provides the first proactive approach to myelosuppression through a unique mechanism of action that helps protect the bone marrow from damage by chemotherapy.”
Approval based on randomized, placebo-controlled trials
The approval of trilaciclib is based on data from three randomized, double-blind, placebo-controlled studies, involving a total of 245 patients with ES-SCLC.
These patients were being treated with chemotherapy regimens that were based on the combination of carboplatin and etoposide (with or without the immunotherapy atezolizumab) or regimens that were based on topotecan.
Before receiving the chemotherapy, patients were randomly assigned to receive trilaciclib or placebo.
Results showed that patients who had received an infusion of trilaciclib before receiving chemotherapy had a lower chance of developing severe neutropenia compared with patients who received a placebo, the FDA noted. In addition, among the patients who did develop severe neutropenia, this had a shorter duration among patients who received trilaciclib than among those who received placebo.
The most common side effects of trilaciclib were fatigue; low levels of calcium, potassium, and phosphate in the blood; increased levels of aspartate aminotransferase; headache; and pneumonia.
The FDA noted that patients should also be advised about injection site reactions, acute drug hypersensitivity, interstitial lung disease/pneumonitis, and embryo-fetal toxicity.
The approval received a priority review, based on the drug’s breakthrough therapy designation. As is common for such products, the company plans postmarketing activities that will assess the effects of trilaciclib on disease progression or survival with at least a 2-year follow up. This clinical trial is scheduled to start in 2022.
A version of this article first appeared on Medscape.com.
A novel drug that offers multilineage protection from chemotherapy-induced myelosuppression has been approved by the Food and Drug Administration.
The drug, trilaciclib (Cosela, G1 Therapeutics) is administered intravenously as a 30-minute infusion within 4 hours prior to the start of chemotherapy. It is indicated specifically for use in adults with extensive-stage small-cell lung cancer (ES-SCLC) who are receiving chemotherapy.
Trilaciclib is a CDK4/6 inhibitor, and this action appears to protect normal bone marrow cells from the harmful effects of chemotherapy.
“For patients with extensive-stage small-cell lung cancer, protecting bone marrow function may help make their chemotherapy safer and allow them to complete their course of treatment on time and according to plan,” Albert Deisseroth, MD, PhD, of the FDA’s Center for Drug Evaluation and Research, said in an FDA press release.
First drug of its type
Trilaciclib “is the first and only therapy designed to help protect bone marrow (myeloprotection) when administered prior to treatment with chemotherapy,” according to the drug’s manufacturer.
Myelosuppression is one of the most severe adverse effects of chemotherapy, and it can be life-threatening. It can increase the risk of infection and lead to severe anemia and/or bleeding.
“These complications impact patients’ quality of life and may also result in chemotherapy dose reductions and delays,” Jeffrey Crawford, MD, of Duke Cancer Institute, Durham, N.C., said in a company press release.
“To date, approaches have included the use of growth factor agents to accelerate blood cell recovery after the bone marrow injury has occurred, along with antibiotics and transfusions as needed. By contrast, trilaciclib provides the first proactive approach to myelosuppression through a unique mechanism of action that helps protect the bone marrow from damage by chemotherapy.”
Approval based on randomized, placebo-controlled trials
The approval of trilaciclib is based on data from three randomized, double-blind, placebo-controlled studies, involving a total of 245 patients with ES-SCLC.
These patients were being treated with chemotherapy regimens that were based on the combination of carboplatin and etoposide (with or without the immunotherapy atezolizumab) or regimens that were based on topotecan.
Before receiving the chemotherapy, patients were randomly assigned to receive trilaciclib or placebo.
Results showed that patients who had received an infusion of trilaciclib before receiving chemotherapy had a lower chance of developing severe neutropenia compared with patients who received a placebo, the FDA noted. In addition, among the patients who did develop severe neutropenia, this had a shorter duration among patients who received trilaciclib than among those who received placebo.
The most common side effects of trilaciclib were fatigue; low levels of calcium, potassium, and phosphate in the blood; increased levels of aspartate aminotransferase; headache; and pneumonia.
The FDA noted that patients should also be advised about injection site reactions, acute drug hypersensitivity, interstitial lung disease/pneumonitis, and embryo-fetal toxicity.
The approval received a priority review, based on the drug’s breakthrough therapy designation. As is common for such products, the company plans postmarketing activities that will assess the effects of trilaciclib on disease progression or survival with at least a 2-year follow up. This clinical trial is scheduled to start in 2022.
A version of this article first appeared on Medscape.com.
A novel drug that offers multilineage protection from chemotherapy-induced myelosuppression has been approved by the Food and Drug Administration.
The drug, trilaciclib (Cosela, G1 Therapeutics) is administered intravenously as a 30-minute infusion within 4 hours prior to the start of chemotherapy. It is indicated specifically for use in adults with extensive-stage small-cell lung cancer (ES-SCLC) who are receiving chemotherapy.
Trilaciclib is a CDK4/6 inhibitor, and this action appears to protect normal bone marrow cells from the harmful effects of chemotherapy.
“For patients with extensive-stage small-cell lung cancer, protecting bone marrow function may help make their chemotherapy safer and allow them to complete their course of treatment on time and according to plan,” Albert Deisseroth, MD, PhD, of the FDA’s Center for Drug Evaluation and Research, said in an FDA press release.
First drug of its type
Trilaciclib “is the first and only therapy designed to help protect bone marrow (myeloprotection) when administered prior to treatment with chemotherapy,” according to the drug’s manufacturer.
Myelosuppression is one of the most severe adverse effects of chemotherapy, and it can be life-threatening. It can increase the risk of infection and lead to severe anemia and/or bleeding.
“These complications impact patients’ quality of life and may also result in chemotherapy dose reductions and delays,” Jeffrey Crawford, MD, of Duke Cancer Institute, Durham, N.C., said in a company press release.
“To date, approaches have included the use of growth factor agents to accelerate blood cell recovery after the bone marrow injury has occurred, along with antibiotics and transfusions as needed. By contrast, trilaciclib provides the first proactive approach to myelosuppression through a unique mechanism of action that helps protect the bone marrow from damage by chemotherapy.”
Approval based on randomized, placebo-controlled trials
The approval of trilaciclib is based on data from three randomized, double-blind, placebo-controlled studies, involving a total of 245 patients with ES-SCLC.
These patients were being treated with chemotherapy regimens that were based on the combination of carboplatin and etoposide (with or without the immunotherapy atezolizumab) or regimens that were based on topotecan.
Before receiving the chemotherapy, patients were randomly assigned to receive trilaciclib or placebo.
Results showed that patients who had received an infusion of trilaciclib before receiving chemotherapy had a lower chance of developing severe neutropenia compared with patients who received a placebo, the FDA noted. In addition, among the patients who did develop severe neutropenia, this had a shorter duration among patients who received trilaciclib than among those who received placebo.
The most common side effects of trilaciclib were fatigue; low levels of calcium, potassium, and phosphate in the blood; increased levels of aspartate aminotransferase; headache; and pneumonia.
The FDA noted that patients should also be advised about injection site reactions, acute drug hypersensitivity, interstitial lung disease/pneumonitis, and embryo-fetal toxicity.
The approval received a priority review, based on the drug’s breakthrough therapy designation. As is common for such products, the company plans postmarketing activities that will assess the effects of trilaciclib on disease progression or survival with at least a 2-year follow up. This clinical trial is scheduled to start in 2022.
A version of this article first appeared on Medscape.com.
Super Bowl ad for diabetes device prompts debate
A commercial for the continuous glucose monitor (CGM) Dexcom G6 shown during the Super Bowl has provoked strong reactions in the diabetes community, both positive and negative.
The 30-second ad, which aired between the first two quarters of the American football game yesterday, features singer-songwriter-actor Nick Jonas, who has type 1 diabetes. During the ad, Mr. Jonas asks – with so much technology available today, including drones that deliver packages and self-driving cars – why are people with diabetes still pricking their fingers to test their blood sugar?
Mr. Jonas goes on to demonstrate the Dexcom G6 smartphone glucose app as it displays three different glucose levels including two trending upward, explaining: “It shows your glucose right in your phone, and where it’s heading, without fingersticks. Finally, technology that makes it easier to manage our diabetes.”
Diabetes type or insulin treatment are not mentioned in the ad, despite the fact that most insurance plans typically only cover CGMs for people with type 1 diabetes and sometimes for those with type 2 diabetes who take multiple daily insulin doses (given the risk for hypoglycemia).
Ad prompts mixed reaction on social media
Reactions rolled in on Twitter after the ad debuted Feb. 2, and then again after it aired during the game.
Some people who have type 1 diabetes themselves or have children with the disease who use the product were thrilled.
“Thanks to @NickJonas for his advocacy on T1. My 11-year old has been on the Dexcom for 3 weeks. For a newly diagnosed kid, it removes a lot of anxiety (and for his parents, too!) Plus, he is thrilled his meter has a Super Bowl commercial!” tweeted @KatisJewell.
Another positive tweet, from @rturnerroy, read: “@nickjonas Thank you for bringing representation to #type1diabetes. And hey #Dexcom, you’re the best.”
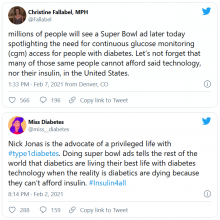
But many others were critical, both of Jonas and Dexcom. @hb_herrick tweeted: “Diabetes awareness is fantastic. Dexcom being able to afford Nick Jonas for a #SuperBowl commercial is not. This is a health care product. Make it more affordable for those who need it.”
Another Twitter user, @universeofdust, tweeted: “Feeling ambivalent about the #Dexcom ad tbh. I love the awareness & representation. But also not a big fan of dexcom spending $5.5 mill+ to make the CGM seem like this ~cool & trendy~ thing when many type 1s can’t afford their insulin, let alone a CGM.”
And @andricheli wrote: “Only people lucky enough to have excellent insurance and be able to afford the out-of-pocket costs have access. Many others do not.”
And in another tweet the same user said, “The #Dexcom is an amazing device. It’s literally lifesaving and life extending. But it’s also very expensive and not available to everyone. Maybe instead of spending $5 mil on a Super Bowl ad, @dexcom should spend that on getting Dex into the handle of people who need it.”
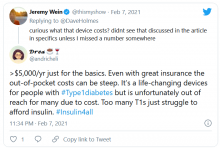
Others, including @1hitwonderdate, criticized Mr. Jonas directly, asking him: “As someone who has struggled with diabetes and is trying to support themselves along with millions of others, why not use this platform to help those who can’t afford their supplies or are rationing them?!”
Dexcom and Jonas’ organization respond
This news organization reached out to both Dexcom and to Beyond Type 1, a nonprofit organization cofounded by Mr. Jonas, for comment. Both emailed responses.
Regarding the intended audience for the ad, Dexcom acknowledged that it hoped to reach a much wider group than just people with type 1 diabetes or even just insulin users.
“We believe our CGM technology has the ability to empower any person with diabetes and significantly improve their treatment and quality of life, whether they are using insulin or not,” the company said, adding that the ad was also aimed at “loved ones, caregivers, and even health care professionals who need to know about this technology.”
According to Dexcom, the G6 is covered by 99% of commercial insurance in the United States, in addition to Medicare, and by Medicaid in more than 40 states. Over 70% of Dexcom patients with pharmacy coverage in the United States pay under $60 per month for CGM, and a third pay $0 out-of-pocket.
“That said, we know there’s more to be done to improve access, and we are working with several partners to broaden access to Dexcom CGM, especially for people with type 2 diabetes not on mealtime insulin,” the company noted.
Beyond Type 1 responded to the criticisms about Mr. Jonas personally, noting that the celebrity is, in fact, heavily involved in advocacy.
“Nick was involved in the launch of GetInsulin.org this past October,” they said. “GetInsulin.org is a tool created by Beyond Type 1 to connect people with diabetes in the United States to the insulin access and affordability options that match their unique circumstances. ... Beyond Type 1 will continue driving awareness of short-term solutions related to insulin access and affordability while fighting for systemic change.”
The organization “is also advocating for systemic payment policies that will make devices less expensive and avoid the same pitfalls (and rising prices) as the drug pricing system in the U.S.”
Mr. Jonas himself appears aware of the concerns.
Is 2021’s most expensive Super Bowl ad justified?
Meanwhile, in a piece in Esquire, Dave Holmes, who has type 1 diabetes, weighs up the pros and cons of the ad.
He writes: “While Jonas makes it look fun and easy to use a Dexcom G6 – a program to just get with like you would a drone or LED eyelashes – the process of acquiring one is complicated and often very expensive, even for people with good insurance. Which makes the year’s most expensive ad buy, for a product that only a small percentage of the U.S. population needs, confusing to me and others.”
Mr. Holmes also spoke with Craig Stubing, founder of the Beta Cell Foundation, a nonprofit that aims to educate and empower those with type 1 diabetes.
“Spending all this money on an ad, when people’s lives are at stake. I don’t know if offensive is the right word, but it seems out of touch with the reality that their patients are facing,” Mr. Stubing told Mr. Holmes.
A version of this article first appeared on Medscape.com.
A commercial for the continuous glucose monitor (CGM) Dexcom G6 shown during the Super Bowl has provoked strong reactions in the diabetes community, both positive and negative.
The 30-second ad, which aired between the first two quarters of the American football game yesterday, features singer-songwriter-actor Nick Jonas, who has type 1 diabetes. During the ad, Mr. Jonas asks – with so much technology available today, including drones that deliver packages and self-driving cars – why are people with diabetes still pricking their fingers to test their blood sugar?
Mr. Jonas goes on to demonstrate the Dexcom G6 smartphone glucose app as it displays three different glucose levels including two trending upward, explaining: “It shows your glucose right in your phone, and where it’s heading, without fingersticks. Finally, technology that makes it easier to manage our diabetes.”
Diabetes type or insulin treatment are not mentioned in the ad, despite the fact that most insurance plans typically only cover CGMs for people with type 1 diabetes and sometimes for those with type 2 diabetes who take multiple daily insulin doses (given the risk for hypoglycemia).
Ad prompts mixed reaction on social media
Reactions rolled in on Twitter after the ad debuted Feb. 2, and then again after it aired during the game.
Some people who have type 1 diabetes themselves or have children with the disease who use the product were thrilled.
“Thanks to @NickJonas for his advocacy on T1. My 11-year old has been on the Dexcom for 3 weeks. For a newly diagnosed kid, it removes a lot of anxiety (and for his parents, too!) Plus, he is thrilled his meter has a Super Bowl commercial!” tweeted @KatisJewell.
Another positive tweet, from @rturnerroy, read: “@nickjonas Thank you for bringing representation to #type1diabetes. And hey #Dexcom, you’re the best.”
But many others were critical, both of Jonas and Dexcom. @hb_herrick tweeted: “Diabetes awareness is fantastic. Dexcom being able to afford Nick Jonas for a #SuperBowl commercial is not. This is a health care product. Make it more affordable for those who need it.”
Another Twitter user, @universeofdust, tweeted: “Feeling ambivalent about the #Dexcom ad tbh. I love the awareness & representation. But also not a big fan of dexcom spending $5.5 mill+ to make the CGM seem like this ~cool & trendy~ thing when many type 1s can’t afford their insulin, let alone a CGM.”
And @andricheli wrote: “Only people lucky enough to have excellent insurance and be able to afford the out-of-pocket costs have access. Many others do not.”
And in another tweet the same user said, “The #Dexcom is an amazing device. It’s literally lifesaving and life extending. But it’s also very expensive and not available to everyone. Maybe instead of spending $5 mil on a Super Bowl ad, @dexcom should spend that on getting Dex into the handle of people who need it.”
Others, including @1hitwonderdate, criticized Mr. Jonas directly, asking him: “As someone who has struggled with diabetes and is trying to support themselves along with millions of others, why not use this platform to help those who can’t afford their supplies or are rationing them?!”
Dexcom and Jonas’ organization respond
This news organization reached out to both Dexcom and to Beyond Type 1, a nonprofit organization cofounded by Mr. Jonas, for comment. Both emailed responses.
Regarding the intended audience for the ad, Dexcom acknowledged that it hoped to reach a much wider group than just people with type 1 diabetes or even just insulin users.
“We believe our CGM technology has the ability to empower any person with diabetes and significantly improve their treatment and quality of life, whether they are using insulin or not,” the company said, adding that the ad was also aimed at “loved ones, caregivers, and even health care professionals who need to know about this technology.”
According to Dexcom, the G6 is covered by 99% of commercial insurance in the United States, in addition to Medicare, and by Medicaid in more than 40 states. Over 70% of Dexcom patients with pharmacy coverage in the United States pay under $60 per month for CGM, and a third pay $0 out-of-pocket.
“That said, we know there’s more to be done to improve access, and we are working with several partners to broaden access to Dexcom CGM, especially for people with type 2 diabetes not on mealtime insulin,” the company noted.
Beyond Type 1 responded to the criticisms about Mr. Jonas personally, noting that the celebrity is, in fact, heavily involved in advocacy.
“Nick was involved in the launch of GetInsulin.org this past October,” they said. “GetInsulin.org is a tool created by Beyond Type 1 to connect people with diabetes in the United States to the insulin access and affordability options that match their unique circumstances. ... Beyond Type 1 will continue driving awareness of short-term solutions related to insulin access and affordability while fighting for systemic change.”
The organization “is also advocating for systemic payment policies that will make devices less expensive and avoid the same pitfalls (and rising prices) as the drug pricing system in the U.S.”
Mr. Jonas himself appears aware of the concerns.
Is 2021’s most expensive Super Bowl ad justified?
Meanwhile, in a piece in Esquire, Dave Holmes, who has type 1 diabetes, weighs up the pros and cons of the ad.
He writes: “While Jonas makes it look fun and easy to use a Dexcom G6 – a program to just get with like you would a drone or LED eyelashes – the process of acquiring one is complicated and often very expensive, even for people with good insurance. Which makes the year’s most expensive ad buy, for a product that only a small percentage of the U.S. population needs, confusing to me and others.”
Mr. Holmes also spoke with Craig Stubing, founder of the Beta Cell Foundation, a nonprofit that aims to educate and empower those with type 1 diabetes.
“Spending all this money on an ad, when people’s lives are at stake. I don’t know if offensive is the right word, but it seems out of touch with the reality that their patients are facing,” Mr. Stubing told Mr. Holmes.
A version of this article first appeared on Medscape.com.
A commercial for the continuous glucose monitor (CGM) Dexcom G6 shown during the Super Bowl has provoked strong reactions in the diabetes community, both positive and negative.
The 30-second ad, which aired between the first two quarters of the American football game yesterday, features singer-songwriter-actor Nick Jonas, who has type 1 diabetes. During the ad, Mr. Jonas asks – with so much technology available today, including drones that deliver packages and self-driving cars – why are people with diabetes still pricking their fingers to test their blood sugar?
Mr. Jonas goes on to demonstrate the Dexcom G6 smartphone glucose app as it displays three different glucose levels including two trending upward, explaining: “It shows your glucose right in your phone, and where it’s heading, without fingersticks. Finally, technology that makes it easier to manage our diabetes.”
Diabetes type or insulin treatment are not mentioned in the ad, despite the fact that most insurance plans typically only cover CGMs for people with type 1 diabetes and sometimes for those with type 2 diabetes who take multiple daily insulin doses (given the risk for hypoglycemia).
Ad prompts mixed reaction on social media
Reactions rolled in on Twitter after the ad debuted Feb. 2, and then again after it aired during the game.
Some people who have type 1 diabetes themselves or have children with the disease who use the product were thrilled.
“Thanks to @NickJonas for his advocacy on T1. My 11-year old has been on the Dexcom for 3 weeks. For a newly diagnosed kid, it removes a lot of anxiety (and for his parents, too!) Plus, he is thrilled his meter has a Super Bowl commercial!” tweeted @KatisJewell.
Another positive tweet, from @rturnerroy, read: “@nickjonas Thank you for bringing representation to #type1diabetes. And hey #Dexcom, you’re the best.”
But many others were critical, both of Jonas and Dexcom. @hb_herrick tweeted: “Diabetes awareness is fantastic. Dexcom being able to afford Nick Jonas for a #SuperBowl commercial is not. This is a health care product. Make it more affordable for those who need it.”
Another Twitter user, @universeofdust, tweeted: “Feeling ambivalent about the #Dexcom ad tbh. I love the awareness & representation. But also not a big fan of dexcom spending $5.5 mill+ to make the CGM seem like this ~cool & trendy~ thing when many type 1s can’t afford their insulin, let alone a CGM.”
And @andricheli wrote: “Only people lucky enough to have excellent insurance and be able to afford the out-of-pocket costs have access. Many others do not.”
And in another tweet the same user said, “The #Dexcom is an amazing device. It’s literally lifesaving and life extending. But it’s also very expensive and not available to everyone. Maybe instead of spending $5 mil on a Super Bowl ad, @dexcom should spend that on getting Dex into the handle of people who need it.”
Others, including @1hitwonderdate, criticized Mr. Jonas directly, asking him: “As someone who has struggled with diabetes and is trying to support themselves along with millions of others, why not use this platform to help those who can’t afford their supplies or are rationing them?!”
Dexcom and Jonas’ organization respond
This news organization reached out to both Dexcom and to Beyond Type 1, a nonprofit organization cofounded by Mr. Jonas, for comment. Both emailed responses.
Regarding the intended audience for the ad, Dexcom acknowledged that it hoped to reach a much wider group than just people with type 1 diabetes or even just insulin users.
“We believe our CGM technology has the ability to empower any person with diabetes and significantly improve their treatment and quality of life, whether they are using insulin or not,” the company said, adding that the ad was also aimed at “loved ones, caregivers, and even health care professionals who need to know about this technology.”
According to Dexcom, the G6 is covered by 99% of commercial insurance in the United States, in addition to Medicare, and by Medicaid in more than 40 states. Over 70% of Dexcom patients with pharmacy coverage in the United States pay under $60 per month for CGM, and a third pay $0 out-of-pocket.
“That said, we know there’s more to be done to improve access, and we are working with several partners to broaden access to Dexcom CGM, especially for people with type 2 diabetes not on mealtime insulin,” the company noted.
Beyond Type 1 responded to the criticisms about Mr. Jonas personally, noting that the celebrity is, in fact, heavily involved in advocacy.
“Nick was involved in the launch of GetInsulin.org this past October,” they said. “GetInsulin.org is a tool created by Beyond Type 1 to connect people with diabetes in the United States to the insulin access and affordability options that match their unique circumstances. ... Beyond Type 1 will continue driving awareness of short-term solutions related to insulin access and affordability while fighting for systemic change.”
The organization “is also advocating for systemic payment policies that will make devices less expensive and avoid the same pitfalls (and rising prices) as the drug pricing system in the U.S.”
Mr. Jonas himself appears aware of the concerns.
Is 2021’s most expensive Super Bowl ad justified?
Meanwhile, in a piece in Esquire, Dave Holmes, who has type 1 diabetes, weighs up the pros and cons of the ad.
He writes: “While Jonas makes it look fun and easy to use a Dexcom G6 – a program to just get with like you would a drone or LED eyelashes – the process of acquiring one is complicated and often very expensive, even for people with good insurance. Which makes the year’s most expensive ad buy, for a product that only a small percentage of the U.S. population needs, confusing to me and others.”
Mr. Holmes also spoke with Craig Stubing, founder of the Beta Cell Foundation, a nonprofit that aims to educate and empower those with type 1 diabetes.
“Spending all this money on an ad, when people’s lives are at stake. I don’t know if offensive is the right word, but it seems out of touch with the reality that their patients are facing,” Mr. Stubing told Mr. Holmes.
A version of this article first appeared on Medscape.com.
Antidepressants may scupper efficacy of MDMA for PTSD
Pooled data from four phase 2 trials reveal that patients with recent SSRI exposure were significantly more likely to continue to meet PTSD diagnostic criteria after methylenedioxymethamphetamine (MDMA)-assisted psychotherapy than their peers who had not recently taken SSRIs.
Although preliminary, the findings have implications for clinical practice if MDMA-assisted psychotherapy is approved by the Food and Drug Administration, Allison Feduccia, PhD, study coauthor and founder of the education platform Psychedelic.Support, said in an interview.
“As psychedelic medicines become available, it’s going to be important that we try to understand what factors impact the response rate and if there are ways that we can improve the treatment outcomes. Allowing for a longer period for tapering completely off SSRIs before initiating MDMA sessions might increase the effectiveness of MDMA,” Dr. Feduccia said.
The study was published online Nov. 20, 2020, in Psychopharmacology (doi: 10.1007/s00213-020-05710-w).
Reduced response
The primary mechanism of action of MDMA involves the same reuptake transporters that are targeted by antidepressant medications commonly prescribed for PTSD. These medications include SSRIs, serotonin-norepinephrine reuptake inhibitors (SNRIs), NRIs, and norepinephrine-dopamine reuptake inhibitors (NDRIs).
Prior research shows that, when MDMA is coadministered with a reuptake inhibitor, subjective and psychological effects of the therapy are attenuated.
The researchers sought to determine whether or not recent tapering off of an antidepressant that targets the same primary binding sites as MDMA would affect treatment response. They analyzed data on 50 adults who underwent two sessions of MDMA-assisted psychotherapy in phase 2 clinical trials.
For 16 of these patients, SSRI therapy was tapered off prior to the MDMA sessions. For 34 patients, SSRI therapy was not tapered off, because the patients had not been taking the medication at the time of initial study screening (nontaper group).
The taper protocols specified that medications be tapered gradually over a period of weeks to minimize withdrawal symptoms and for them to be discontinued at least five half-lives of each drug prior to MDMA administration.
Demographics, baseline PTSD, and depression severity were similar between the taper and the nontaper groups. Participants in the studies had chronic PTSD (symptoms lasting >6 months). Severity scores on the Clinician-Administered PTSD Scale for DSM IV (CAPS-IV) were at least 50.
After MDMA-assisted psychotherapy, the nontaper group had significantly lower (better) CAPS-IV total scores, compared with the taper group (mean, 45.7 vs. 70.3; P = .009).
About two-thirds (63.6%) of the nontaper group no longer met PTSD criteria after MDMA-assisted therapy, compared with only 25% of those in the taper group.
The nontaper group also had lower depression symptom severity scores on the Beck Depression Inventory–II, compared with the taper group (mean, 12.7 vs. 22.6; P = .010).
“Another really interesting” observation, said Dr. Feduccia, is that the expected increases in systolic and diastolic blood pressure following MDMA administration were reduced in the taper group, compared with the nontaper group.
“This suggests that MDMA didn’t have the same physiological response in individuals who tapered SSRIs. This should be followed up,” she said.
The investigators offerred several potential mechanisms for the negative effect of recent SSRI use on MDMA-assisted psychotherapy for PTSD.
These include the down-regulation of binding sites (serotonin, dopamine, and/or norepinephrine) related to SSRI use, reduced MDMA treatment-relevant increases in blood pressure in patients with recent SSRI use, and the possibility that withdrawal symptoms from SSRIs may reduce the effectiveness of MDMA psychotherapy.
Important clinical implications
In a comment, Steven R. Thorp, PhD, professor at Alliant International University, San Diego, said the findings are “very interesting” and likely “not well known.”
“There has been great interest in MDMA-assisted psychotherapy in recent years, and if this finding is replicated, it will have important implications for that research,” Dr. Thorp said.
“Although psychotherapy is often preferred by clients with PTSD, compared to medications, and typically shows efficacy that is as strong or stronger (and longer lasting) than medications, many individuals with PTSD are provided with medication only,” Dr. Thorp noted.
“This study suggests that, in addition to the other potential disadvantages of medications (e.g., cost, side effects, potential for addiction), those who take SSRIs, SNRIs, NRIs, and NDRIs for PTSD may also benefit less from MDMA-assisted psychotherapy,” Dr. Thorp added.
The four phase 2 studies used in the analysis were sponsored by the Multidisciplinary Association for Psychedelic Studies, a nonprofit organization. Dr. Feduccia received salary support for full-time employment with MAPS Public Benefit Corporation. Dr. Thorp disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Pooled data from four phase 2 trials reveal that patients with recent SSRI exposure were significantly more likely to continue to meet PTSD diagnostic criteria after methylenedioxymethamphetamine (MDMA)-assisted psychotherapy than their peers who had not recently taken SSRIs.
Although preliminary, the findings have implications for clinical practice if MDMA-assisted psychotherapy is approved by the Food and Drug Administration, Allison Feduccia, PhD, study coauthor and founder of the education platform Psychedelic.Support, said in an interview.
“As psychedelic medicines become available, it’s going to be important that we try to understand what factors impact the response rate and if there are ways that we can improve the treatment outcomes. Allowing for a longer period for tapering completely off SSRIs before initiating MDMA sessions might increase the effectiveness of MDMA,” Dr. Feduccia said.
The study was published online Nov. 20, 2020, in Psychopharmacology (doi: 10.1007/s00213-020-05710-w).
Reduced response
The primary mechanism of action of MDMA involves the same reuptake transporters that are targeted by antidepressant medications commonly prescribed for PTSD. These medications include SSRIs, serotonin-norepinephrine reuptake inhibitors (SNRIs), NRIs, and norepinephrine-dopamine reuptake inhibitors (NDRIs).
Prior research shows that, when MDMA is coadministered with a reuptake inhibitor, subjective and psychological effects of the therapy are attenuated.
The researchers sought to determine whether or not recent tapering off of an antidepressant that targets the same primary binding sites as MDMA would affect treatment response. They analyzed data on 50 adults who underwent two sessions of MDMA-assisted psychotherapy in phase 2 clinical trials.
For 16 of these patients, SSRI therapy was tapered off prior to the MDMA sessions. For 34 patients, SSRI therapy was not tapered off, because the patients had not been taking the medication at the time of initial study screening (nontaper group).
The taper protocols specified that medications be tapered gradually over a period of weeks to minimize withdrawal symptoms and for them to be discontinued at least five half-lives of each drug prior to MDMA administration.
Demographics, baseline PTSD, and depression severity were similar between the taper and the nontaper groups. Participants in the studies had chronic PTSD (symptoms lasting >6 months). Severity scores on the Clinician-Administered PTSD Scale for DSM IV (CAPS-IV) were at least 50.
After MDMA-assisted psychotherapy, the nontaper group had significantly lower (better) CAPS-IV total scores, compared with the taper group (mean, 45.7 vs. 70.3; P = .009).
About two-thirds (63.6%) of the nontaper group no longer met PTSD criteria after MDMA-assisted therapy, compared with only 25% of those in the taper group.
The nontaper group also had lower depression symptom severity scores on the Beck Depression Inventory–II, compared with the taper group (mean, 12.7 vs. 22.6; P = .010).
“Another really interesting” observation, said Dr. Feduccia, is that the expected increases in systolic and diastolic blood pressure following MDMA administration were reduced in the taper group, compared with the nontaper group.
“This suggests that MDMA didn’t have the same physiological response in individuals who tapered SSRIs. This should be followed up,” she said.
The investigators offerred several potential mechanisms for the negative effect of recent SSRI use on MDMA-assisted psychotherapy for PTSD.
These include the down-regulation of binding sites (serotonin, dopamine, and/or norepinephrine) related to SSRI use, reduced MDMA treatment-relevant increases in blood pressure in patients with recent SSRI use, and the possibility that withdrawal symptoms from SSRIs may reduce the effectiveness of MDMA psychotherapy.
Important clinical implications
In a comment, Steven R. Thorp, PhD, professor at Alliant International University, San Diego, said the findings are “very interesting” and likely “not well known.”
“There has been great interest in MDMA-assisted psychotherapy in recent years, and if this finding is replicated, it will have important implications for that research,” Dr. Thorp said.
“Although psychotherapy is often preferred by clients with PTSD, compared to medications, and typically shows efficacy that is as strong or stronger (and longer lasting) than medications, many individuals with PTSD are provided with medication only,” Dr. Thorp noted.
“This study suggests that, in addition to the other potential disadvantages of medications (e.g., cost, side effects, potential for addiction), those who take SSRIs, SNRIs, NRIs, and NDRIs for PTSD may also benefit less from MDMA-assisted psychotherapy,” Dr. Thorp added.
The four phase 2 studies used in the analysis were sponsored by the Multidisciplinary Association for Psychedelic Studies, a nonprofit organization. Dr. Feduccia received salary support for full-time employment with MAPS Public Benefit Corporation. Dr. Thorp disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Pooled data from four phase 2 trials reveal that patients with recent SSRI exposure were significantly more likely to continue to meet PTSD diagnostic criteria after methylenedioxymethamphetamine (MDMA)-assisted psychotherapy than their peers who had not recently taken SSRIs.
Although preliminary, the findings have implications for clinical practice if MDMA-assisted psychotherapy is approved by the Food and Drug Administration, Allison Feduccia, PhD, study coauthor and founder of the education platform Psychedelic.Support, said in an interview.
“As psychedelic medicines become available, it’s going to be important that we try to understand what factors impact the response rate and if there are ways that we can improve the treatment outcomes. Allowing for a longer period for tapering completely off SSRIs before initiating MDMA sessions might increase the effectiveness of MDMA,” Dr. Feduccia said.
The study was published online Nov. 20, 2020, in Psychopharmacology (doi: 10.1007/s00213-020-05710-w).
Reduced response
The primary mechanism of action of MDMA involves the same reuptake transporters that are targeted by antidepressant medications commonly prescribed for PTSD. These medications include SSRIs, serotonin-norepinephrine reuptake inhibitors (SNRIs), NRIs, and norepinephrine-dopamine reuptake inhibitors (NDRIs).
Prior research shows that, when MDMA is coadministered with a reuptake inhibitor, subjective and psychological effects of the therapy are attenuated.
The researchers sought to determine whether or not recent tapering off of an antidepressant that targets the same primary binding sites as MDMA would affect treatment response. They analyzed data on 50 adults who underwent two sessions of MDMA-assisted psychotherapy in phase 2 clinical trials.
For 16 of these patients, SSRI therapy was tapered off prior to the MDMA sessions. For 34 patients, SSRI therapy was not tapered off, because the patients had not been taking the medication at the time of initial study screening (nontaper group).
The taper protocols specified that medications be tapered gradually over a period of weeks to minimize withdrawal symptoms and for them to be discontinued at least five half-lives of each drug prior to MDMA administration.
Demographics, baseline PTSD, and depression severity were similar between the taper and the nontaper groups. Participants in the studies had chronic PTSD (symptoms lasting >6 months). Severity scores on the Clinician-Administered PTSD Scale for DSM IV (CAPS-IV) were at least 50.
After MDMA-assisted psychotherapy, the nontaper group had significantly lower (better) CAPS-IV total scores, compared with the taper group (mean, 45.7 vs. 70.3; P = .009).
About two-thirds (63.6%) of the nontaper group no longer met PTSD criteria after MDMA-assisted therapy, compared with only 25% of those in the taper group.
The nontaper group also had lower depression symptom severity scores on the Beck Depression Inventory–II, compared with the taper group (mean, 12.7 vs. 22.6; P = .010).
“Another really interesting” observation, said Dr. Feduccia, is that the expected increases in systolic and diastolic blood pressure following MDMA administration were reduced in the taper group, compared with the nontaper group.
“This suggests that MDMA didn’t have the same physiological response in individuals who tapered SSRIs. This should be followed up,” she said.
The investigators offerred several potential mechanisms for the negative effect of recent SSRI use on MDMA-assisted psychotherapy for PTSD.
These include the down-regulation of binding sites (serotonin, dopamine, and/or norepinephrine) related to SSRI use, reduced MDMA treatment-relevant increases in blood pressure in patients with recent SSRI use, and the possibility that withdrawal symptoms from SSRIs may reduce the effectiveness of MDMA psychotherapy.
Important clinical implications
In a comment, Steven R. Thorp, PhD, professor at Alliant International University, San Diego, said the findings are “very interesting” and likely “not well known.”
“There has been great interest in MDMA-assisted psychotherapy in recent years, and if this finding is replicated, it will have important implications for that research,” Dr. Thorp said.
“Although psychotherapy is often preferred by clients with PTSD, compared to medications, and typically shows efficacy that is as strong or stronger (and longer lasting) than medications, many individuals with PTSD are provided with medication only,” Dr. Thorp noted.
“This study suggests that, in addition to the other potential disadvantages of medications (e.g., cost, side effects, potential for addiction), those who take SSRIs, SNRIs, NRIs, and NDRIs for PTSD may also benefit less from MDMA-assisted psychotherapy,” Dr. Thorp added.
The four phase 2 studies used in the analysis were sponsored by the Multidisciplinary Association for Psychedelic Studies, a nonprofit organization. Dr. Feduccia received salary support for full-time employment with MAPS Public Benefit Corporation. Dr. Thorp disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FDA approves orphan drug evinacumab-dgnb for homozygous FH
The Food and Drug Administration has approved the fully human monoclonal antibody evinacumab-dgnb (Evkeeza, Regeneron Pharmaceuticals) for use on top of other cholesterol-modifying medication in patients aged 12 years and older with homozygous familial hypercholesterolemia (HoFH), the agency and Regeneron have announced.
Evinacumab had received orphan drug designation and underwent priority regulatory review based primarily on the phase 3 ELIPSE trial, presented at a meeting in March 2020 and published in August 2020 in the New England Journal of Medicine (doi: 10.1056/NEJMoa2004215).
In the trial with 65 patients with HoFH on guideline-based lipid-modifying therapy, those who also received evinacumab 15 mg/kg intravenously every 4 weeks showed a nearly 50% drop in LDL cholesterol levels after 24 weeks, compared with patients given a placebo. Only 2% of patients in both groups discontinued therapy because of adverse reactions.
The drug blocks angiopoietin-like 3, itself an inhibitor of lipoprotein lipase and endothelial lipase. It therefore lowers LDL cholesterol levels by mechanisms that don’t directly involve the LDL receptor.
Regeneron estimates that about 1300 people in the United States have the homozygous genetic disorder, which can lead to LDL cholesterol levels of a 1,000 mg/dL or higher, advanced premature atherosclerosis, and extreme risk for cardiovascular events.
The drug’s average wholesale acquisition cost per patient in the United States is expected to be about $450,000 per year, the company said, adding that it has a financial support program to help qualified patients with out-of-pocket costs.
Regeneron’s announcement included a comment from dyslipidemia-therapy expert Daniel J. Rader, MD, University of Pennsylvania, Philadelphia, who called evinacumab “a potentially transformational new treatment for people with HoFH.”
The drug is currently under regulatory review for the same indication in Europe, the company said.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved the fully human monoclonal antibody evinacumab-dgnb (Evkeeza, Regeneron Pharmaceuticals) for use on top of other cholesterol-modifying medication in patients aged 12 years and older with homozygous familial hypercholesterolemia (HoFH), the agency and Regeneron have announced.
Evinacumab had received orphan drug designation and underwent priority regulatory review based primarily on the phase 3 ELIPSE trial, presented at a meeting in March 2020 and published in August 2020 in the New England Journal of Medicine (doi: 10.1056/NEJMoa2004215).
In the trial with 65 patients with HoFH on guideline-based lipid-modifying therapy, those who also received evinacumab 15 mg/kg intravenously every 4 weeks showed a nearly 50% drop in LDL cholesterol levels after 24 weeks, compared with patients given a placebo. Only 2% of patients in both groups discontinued therapy because of adverse reactions.
The drug blocks angiopoietin-like 3, itself an inhibitor of lipoprotein lipase and endothelial lipase. It therefore lowers LDL cholesterol levels by mechanisms that don’t directly involve the LDL receptor.
Regeneron estimates that about 1300 people in the United States have the homozygous genetic disorder, which can lead to LDL cholesterol levels of a 1,000 mg/dL or higher, advanced premature atherosclerosis, and extreme risk for cardiovascular events.
The drug’s average wholesale acquisition cost per patient in the United States is expected to be about $450,000 per year, the company said, adding that it has a financial support program to help qualified patients with out-of-pocket costs.
Regeneron’s announcement included a comment from dyslipidemia-therapy expert Daniel J. Rader, MD, University of Pennsylvania, Philadelphia, who called evinacumab “a potentially transformational new treatment for people with HoFH.”
The drug is currently under regulatory review for the same indication in Europe, the company said.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved the fully human monoclonal antibody evinacumab-dgnb (Evkeeza, Regeneron Pharmaceuticals) for use on top of other cholesterol-modifying medication in patients aged 12 years and older with homozygous familial hypercholesterolemia (HoFH), the agency and Regeneron have announced.
Evinacumab had received orphan drug designation and underwent priority regulatory review based primarily on the phase 3 ELIPSE trial, presented at a meeting in March 2020 and published in August 2020 in the New England Journal of Medicine (doi: 10.1056/NEJMoa2004215).
In the trial with 65 patients with HoFH on guideline-based lipid-modifying therapy, those who also received evinacumab 15 mg/kg intravenously every 4 weeks showed a nearly 50% drop in LDL cholesterol levels after 24 weeks, compared with patients given a placebo. Only 2% of patients in both groups discontinued therapy because of adverse reactions.
The drug blocks angiopoietin-like 3, itself an inhibitor of lipoprotein lipase and endothelial lipase. It therefore lowers LDL cholesterol levels by mechanisms that don’t directly involve the LDL receptor.
Regeneron estimates that about 1300 people in the United States have the homozygous genetic disorder, which can lead to LDL cholesterol levels of a 1,000 mg/dL or higher, advanced premature atherosclerosis, and extreme risk for cardiovascular events.
The drug’s average wholesale acquisition cost per patient in the United States is expected to be about $450,000 per year, the company said, adding that it has a financial support program to help qualified patients with out-of-pocket costs.
Regeneron’s announcement included a comment from dyslipidemia-therapy expert Daniel J. Rader, MD, University of Pennsylvania, Philadelphia, who called evinacumab “a potentially transformational new treatment for people with HoFH.”
The drug is currently under regulatory review for the same indication in Europe, the company said.
A version of this article first appeared on Medscape.com.
2021 ACIP adult schedule released
The Advisory Committee on Immunization Practices of the Centers for Disease Control and Prevention has updated its recommended immunization schedule for adults for 2021.
A summary of the annual update was published online Feb. 11 in the CDC’s Morbidity and Mortality Weekly Report and is available in Annals of Internal Medicine and on the CDC website.
It features a special section on vaccination during the pandemic as well as interim recommendations on administering the Pfizer-BioNtech and Moderna COVID-19 vaccines.
The authors, led by Mark S. Freedman, DVM, MPH, DACVPM, of the CDC’s National Center for Immunization and Respiratory Diseases, in Atlanta, note that this year’s recommendations for adults – persons aged 19 years and older – are largely the same as last year’s. “There have been very few changes,” Dr. Freedman said in an interview. “Changes to the schedule tables and notes were made to harmonize to the greatest extent possible the adult and child/adolescent schedules.”
Changes in the schedule include new or updated ACIP recommendations for influenza, hepatitis A, hepatitis B (Hep B), and human papillomavirus (HPV) as well as for meningococcal serogroups A, C, W, and Y (MenACYW) vaccines, meningococcal B (MenB) vaccines, and the zoster vaccine.
Vaccine-specific changes
Influenza
The schedule highlights updates to the composition of several influenza vaccines, which apply to components in both trivalent and quadrivalent formulations.
The cover page abbreviation for live attenuated influenza vaccine (LAIV) was changed to LAIV4. The abbreviation for live recombinant influenza vaccine (RIV) was changed to RIV4.
For individuals with a history of egg allergy who experience reactions other than hives, the following procedural warning has been added: “If using an influenza vaccine other than RIV4 or ccIIV4, administer in medical setting under supervision of health care provider who can recognize and manage severe allergic reactions.”
Zoster
The zoster vaccine live (Zostavax) has been removed from the schedule because it is no longer available in the United States. The recombinant zoster vaccine Shingrix remains available as a 2-dose regimen for adults aged 50 years or older.
HPV
As in previous years, HPV vaccination is routinely recommended for persons aged 11-12 years, with catch-up vaccination for those aged 26 or younger. Catch-up vaccination can be considered with shared decision making for those aged 27 through 45. In this year’s schedule, in the pregnancy column, the color pink, which formerly indicated “delay until after pregnancy,” has been replaced with red and an asterisk, indicating “vaccinate after pregnancy.”
HepB
ACIP continues to recommend vaccination of adults at risk for HepB; however, the text overlay has been changed to read, “2, 3, or 4 doses, depending on vaccine or condition.” Additionally, HepB vaccination is now routinely recommended for adults younger than 60 years with diabetes. For those with diabetes who are older than 60, shared decision making is recommended.
Meningococcal vaccine
ACIP continues to recommend routine vaccination with a quadrivalent meningococcal conjugate vaccine (MenACWY) for persons at increased risk for meningococcal disease caused by serogroups A, C, W, or Y. The MenQuadfi (MenACWY-TT) vaccine, which was first licensed in 2020, has been added to all relevant sections of MenACWY vaccines. For MenACWY booster doses, new text addresses special situations, including outbreaks.
Improvements have been made to text and layout, Dr. Freedman said. An example is the minimizing of specialized text. Other changes were made to ensure more consistent text structure and language. Various fine-tunings of color and positioning were made to the cover page and tables, and the wording of the notes sections was improved.
Vaccination in the pandemic
The updated schedule outlines guidance on the use of COVID-19 vaccines approved by the Food and Drug Administration under emergency use authorization, with interim recommendations for the Pfizer-BioNTech COVID-19 vaccine for people aged 16 and older and the Moderna COVID-19 vaccine for people aged 18 and older.
The authors stress the importance of receiving the recommended routine and catch-up immunizations notwithstanding widespread anxiety about visiting medical offices. Last spring, the CDC reported a dramatic drop in child vaccinations after the declaration of the national emergency in mid-March, a drop attributed to fear of COVID-19 exposure.
“ACIP continued to meet and make recommendations during the pandemic,” Dr. Freedman said. “Our recommendation remains that despite challenges caused by the COVID-19 pandemic, adults and their healthcare providers should follow the recommended vaccine schedule to protect against serious and sometimes deadly diseases.”
Regular vaccines can be safely administered even as COVID-19 retains its grasp on the United States. “Healthcare providers should follow the CDC’s interim guidance for the safe delivery of vaccines during the pandemic, which includes the use of personal protective equipment and physical distancing,” Dr. Freedman said.
Dr. Freedman has disclosed no relevant financial relationships. Coauthor Henry Bernstein, DO, is the editor of the Current Opinion in Pediatrics Office Pediatrics Series, is a Harvard School of Public Health faculty member, and is a member of the data safety and monitoring board for a Takeda study on intrathecal enzymes for Hunter and San Filippo syndromes. Coauthor Kevin Ault, MD, has served on the data safety and monitoring committee for ACI Clinical.
A version of this article first appeared on Medscape.com .
The Advisory Committee on Immunization Practices of the Centers for Disease Control and Prevention has updated its recommended immunization schedule for adults for 2021.
A summary of the annual update was published online Feb. 11 in the CDC’s Morbidity and Mortality Weekly Report and is available in Annals of Internal Medicine and on the CDC website.
It features a special section on vaccination during the pandemic as well as interim recommendations on administering the Pfizer-BioNtech and Moderna COVID-19 vaccines.
The authors, led by Mark S. Freedman, DVM, MPH, DACVPM, of the CDC’s National Center for Immunization and Respiratory Diseases, in Atlanta, note that this year’s recommendations for adults – persons aged 19 years and older – are largely the same as last year’s. “There have been very few changes,” Dr. Freedman said in an interview. “Changes to the schedule tables and notes were made to harmonize to the greatest extent possible the adult and child/adolescent schedules.”
Changes in the schedule include new or updated ACIP recommendations for influenza, hepatitis A, hepatitis B (Hep B), and human papillomavirus (HPV) as well as for meningococcal serogroups A, C, W, and Y (MenACYW) vaccines, meningococcal B (MenB) vaccines, and the zoster vaccine.
Vaccine-specific changes
Influenza
The schedule highlights updates to the composition of several influenza vaccines, which apply to components in both trivalent and quadrivalent formulations.
The cover page abbreviation for live attenuated influenza vaccine (LAIV) was changed to LAIV4. The abbreviation for live recombinant influenza vaccine (RIV) was changed to RIV4.
For individuals with a history of egg allergy who experience reactions other than hives, the following procedural warning has been added: “If using an influenza vaccine other than RIV4 or ccIIV4, administer in medical setting under supervision of health care provider who can recognize and manage severe allergic reactions.”
Zoster
The zoster vaccine live (Zostavax) has been removed from the schedule because it is no longer available in the United States. The recombinant zoster vaccine Shingrix remains available as a 2-dose regimen for adults aged 50 years or older.
HPV
As in previous years, HPV vaccination is routinely recommended for persons aged 11-12 years, with catch-up vaccination for those aged 26 or younger. Catch-up vaccination can be considered with shared decision making for those aged 27 through 45. In this year’s schedule, in the pregnancy column, the color pink, which formerly indicated “delay until after pregnancy,” has been replaced with red and an asterisk, indicating “vaccinate after pregnancy.”
HepB
ACIP continues to recommend vaccination of adults at risk for HepB; however, the text overlay has been changed to read, “2, 3, or 4 doses, depending on vaccine or condition.” Additionally, HepB vaccination is now routinely recommended for adults younger than 60 years with diabetes. For those with diabetes who are older than 60, shared decision making is recommended.
Meningococcal vaccine
ACIP continues to recommend routine vaccination with a quadrivalent meningococcal conjugate vaccine (MenACWY) for persons at increased risk for meningococcal disease caused by serogroups A, C, W, or Y. The MenQuadfi (MenACWY-TT) vaccine, which was first licensed in 2020, has been added to all relevant sections of MenACWY vaccines. For MenACWY booster doses, new text addresses special situations, including outbreaks.
Improvements have been made to text and layout, Dr. Freedman said. An example is the minimizing of specialized text. Other changes were made to ensure more consistent text structure and language. Various fine-tunings of color and positioning were made to the cover page and tables, and the wording of the notes sections was improved.
Vaccination in the pandemic
The updated schedule outlines guidance on the use of COVID-19 vaccines approved by the Food and Drug Administration under emergency use authorization, with interim recommendations for the Pfizer-BioNTech COVID-19 vaccine for people aged 16 and older and the Moderna COVID-19 vaccine for people aged 18 and older.
The authors stress the importance of receiving the recommended routine and catch-up immunizations notwithstanding widespread anxiety about visiting medical offices. Last spring, the CDC reported a dramatic drop in child vaccinations after the declaration of the national emergency in mid-March, a drop attributed to fear of COVID-19 exposure.
“ACIP continued to meet and make recommendations during the pandemic,” Dr. Freedman said. “Our recommendation remains that despite challenges caused by the COVID-19 pandemic, adults and their healthcare providers should follow the recommended vaccine schedule to protect against serious and sometimes deadly diseases.”
Regular vaccines can be safely administered even as COVID-19 retains its grasp on the United States. “Healthcare providers should follow the CDC’s interim guidance for the safe delivery of vaccines during the pandemic, which includes the use of personal protective equipment and physical distancing,” Dr. Freedman said.
Dr. Freedman has disclosed no relevant financial relationships. Coauthor Henry Bernstein, DO, is the editor of the Current Opinion in Pediatrics Office Pediatrics Series, is a Harvard School of Public Health faculty member, and is a member of the data safety and monitoring board for a Takeda study on intrathecal enzymes for Hunter and San Filippo syndromes. Coauthor Kevin Ault, MD, has served on the data safety and monitoring committee for ACI Clinical.
A version of this article first appeared on Medscape.com .
The Advisory Committee on Immunization Practices of the Centers for Disease Control and Prevention has updated its recommended immunization schedule for adults for 2021.
A summary of the annual update was published online Feb. 11 in the CDC’s Morbidity and Mortality Weekly Report and is available in Annals of Internal Medicine and on the CDC website.
It features a special section on vaccination during the pandemic as well as interim recommendations on administering the Pfizer-BioNtech and Moderna COVID-19 vaccines.
The authors, led by Mark S. Freedman, DVM, MPH, DACVPM, of the CDC’s National Center for Immunization and Respiratory Diseases, in Atlanta, note that this year’s recommendations for adults – persons aged 19 years and older – are largely the same as last year’s. “There have been very few changes,” Dr. Freedman said in an interview. “Changes to the schedule tables and notes were made to harmonize to the greatest extent possible the adult and child/adolescent schedules.”
Changes in the schedule include new or updated ACIP recommendations for influenza, hepatitis A, hepatitis B (Hep B), and human papillomavirus (HPV) as well as for meningococcal serogroups A, C, W, and Y (MenACYW) vaccines, meningococcal B (MenB) vaccines, and the zoster vaccine.
Vaccine-specific changes
Influenza
The schedule highlights updates to the composition of several influenza vaccines, which apply to components in both trivalent and quadrivalent formulations.
The cover page abbreviation for live attenuated influenza vaccine (LAIV) was changed to LAIV4. The abbreviation for live recombinant influenza vaccine (RIV) was changed to RIV4.
For individuals with a history of egg allergy who experience reactions other than hives, the following procedural warning has been added: “If using an influenza vaccine other than RIV4 or ccIIV4, administer in medical setting under supervision of health care provider who can recognize and manage severe allergic reactions.”
Zoster
The zoster vaccine live (Zostavax) has been removed from the schedule because it is no longer available in the United States. The recombinant zoster vaccine Shingrix remains available as a 2-dose regimen for adults aged 50 years or older.
HPV
As in previous years, HPV vaccination is routinely recommended for persons aged 11-12 years, with catch-up vaccination for those aged 26 or younger. Catch-up vaccination can be considered with shared decision making for those aged 27 through 45. In this year’s schedule, in the pregnancy column, the color pink, which formerly indicated “delay until after pregnancy,” has been replaced with red and an asterisk, indicating “vaccinate after pregnancy.”
HepB
ACIP continues to recommend vaccination of adults at risk for HepB; however, the text overlay has been changed to read, “2, 3, or 4 doses, depending on vaccine or condition.” Additionally, HepB vaccination is now routinely recommended for adults younger than 60 years with diabetes. For those with diabetes who are older than 60, shared decision making is recommended.
Meningococcal vaccine
ACIP continues to recommend routine vaccination with a quadrivalent meningococcal conjugate vaccine (MenACWY) for persons at increased risk for meningococcal disease caused by serogroups A, C, W, or Y. The MenQuadfi (MenACWY-TT) vaccine, which was first licensed in 2020, has been added to all relevant sections of MenACWY vaccines. For MenACWY booster doses, new text addresses special situations, including outbreaks.
Improvements have been made to text and layout, Dr. Freedman said. An example is the minimizing of specialized text. Other changes were made to ensure more consistent text structure and language. Various fine-tunings of color and positioning were made to the cover page and tables, and the wording of the notes sections was improved.
Vaccination in the pandemic
The updated schedule outlines guidance on the use of COVID-19 vaccines approved by the Food and Drug Administration under emergency use authorization, with interim recommendations for the Pfizer-BioNTech COVID-19 vaccine for people aged 16 and older and the Moderna COVID-19 vaccine for people aged 18 and older.
The authors stress the importance of receiving the recommended routine and catch-up immunizations notwithstanding widespread anxiety about visiting medical offices. Last spring, the CDC reported a dramatic drop in child vaccinations after the declaration of the national emergency in mid-March, a drop attributed to fear of COVID-19 exposure.
“ACIP continued to meet and make recommendations during the pandemic,” Dr. Freedman said. “Our recommendation remains that despite challenges caused by the COVID-19 pandemic, adults and their healthcare providers should follow the recommended vaccine schedule to protect against serious and sometimes deadly diseases.”
Regular vaccines can be safely administered even as COVID-19 retains its grasp on the United States. “Healthcare providers should follow the CDC’s interim guidance for the safe delivery of vaccines during the pandemic, which includes the use of personal protective equipment and physical distancing,” Dr. Freedman said.
Dr. Freedman has disclosed no relevant financial relationships. Coauthor Henry Bernstein, DO, is the editor of the Current Opinion in Pediatrics Office Pediatrics Series, is a Harvard School of Public Health faculty member, and is a member of the data safety and monitoring board for a Takeda study on intrathecal enzymes for Hunter and San Filippo syndromes. Coauthor Kevin Ault, MD, has served on the data safety and monitoring committee for ACI Clinical.
A version of this article first appeared on Medscape.com .
Prostate drugs tied to lower risk for Parkinson’s disease
new research suggests. Treatment of BPH with terazosin (Hytrin), doxazosin (Cardura), or alfuzosin (Uroxatral), all of which enhance glycolysis, was associated with a lower risk of developing Parkinson’s disease than patients taking a drug used for the same indication, tamsulosin (Flomax), which does not affect glycolysis.
“If giving someone terazosin or similar medications truly reduces their risk of disease, these results could have significant clinical implications for neurologists,” said lead author Jacob E. Simmering, PhD, assistant professor of internal medicine at the University of Iowa, Iowa City.
There are few reliable neuroprotective treatments for Parkinson’s disease, he said. “We can manage some of the symptoms, but we can’t stop it from progressing. If a randomized trial finds the same result, this will provide a new option to slow progression of Parkinson’s disease.”
The pathogenesis of Parkinson’s disease is heterogeneous, however, and not all patients may benefit from glycolysis-enhancing drugs, the investigators noted. Future research will be needed to identify potential candidates for this treatment, and clarify the effects of these drugs, they wrote.
The findings were published online Feb. 1, 2021, in JAMA Neurology.
Time-dependent effects
The major risk factor for Parkinson’s disease is age, which is associated with impaired energy metabolism. Glycolysis is decreased among patients with Parkinson’s disease, yet impaired energy metabolism has not been investigated widely as a pathogenic factor in the disease, the authors wrote.
Studies have indicated that terazosin increases the activity of an enzyme important in glycolysis. Doxazosin and alfuzosin have a similar mechanism of action and enhance energy metabolism. Tamsulosin, a structurally unrelated drug, has the same mechanism of action as the other three drugs, but does not enhance energy metabolism.
In this report, the researchers investigated the hypothesis that patients who received therapy with terazosin, doxazosin, or alfuzosin would have a lower risk of developing Parkinson’s disease than patients receiving tamsulosin. To do that, they used health care utilization data from Denmark and the United States, including the Danish National Prescription Registry, the Danish National Patient Registry, the Danish Civil Registration System, and the Truven Health Analytics MarketScan database.
The investigators searched the records for patients who filled prescriptions for any of the four drugs of interest. They excluded any patients who developed Parkinson’s disease within 1 year of starting medication. Because use of these drugs is rare among women, they included only men in their analysis.
They looked at patient outcomes beginning at 1 year after the initiation of treatment. They also required patients to fill at least two prescriptions before the beginning of follow-up. Patients who switched from tamsulosin to any of the other drugs, or vice versa, were excluded from analysis.
The investigators used propensity-score matching to ensure that patients in the tamsulosin and terazosin/doxazosin/alfuzosin groups were similar in terms of their other potential risk factors. The primary outcome was the development of Parkinson’s disease.
They identified 52,365 propensity score–matched pairs in the Danish registries and 94,883 pairs in the Truven database. The mean age was 67.9 years in the Danish registries and 63.8 years in the Truven database, and follow-up was approximately 5 years and 3 years respectively. Baseline covariates were well balanced between cohorts.
Among Danish patients, those who took terazosin, doxazosin, or alfuzosin had a lower risk of developing Parkinson’s disease versus those who took tamsulosin (hazard ratio, 0.88). Similarly, patients in the Truven database who took terazosin, doxazosin, or alfuzosin had a lower risk of developing Parkinson’s disease than those who took tamsulosin (HR, 0.63).
In both cohorts, the risk for Parkinson’s disease among patients receiving terazosin, doxazosin, or alfuzosin, compared with those receiving tamsulosin, decreased with increasing numbers of prescriptions filled. Long-term treatment with any of the three glycolysis-enhancing drugs was associated with greater risk reduction in the Danish (HR, 0.79) and Truven (HR, 0.46) cohorts versus tamsulosin.
Differences in case definitions, which may reflect how Parkinson’s disease was managed, complicate comparisons between the Danish and Truven cohorts, said Dr. Simmering. Another challenge is the source of the data. “The Truven data set was derived from insurance claims from people with private insurance or Medicare supplemental plans,” he said. “This group is quite large but may not be representative of everyone in the United States. We would also only be able to follow people while they were on one insurance plan. If they switched coverage to a company that doesn’t contribute data, we would lose them.”
The Danish database, however, includes all residents of Denmark. Only people who left the country were lost to follow-up.
The results support the hypothesis that increasing energy in cells slows disease progression, Dr. Simmering added. “There are a few conditions, mostly REM sleep disorders, that are associated with future diagnosis of Parkinson’s disease. Right now, we don’t have anything to offer people at elevated risk of Parkinson’s disease that might prevent the disease. If a controlled trial finds that terazosin slows or prevents Parkinson’s disease, we would have something truly protective to offer these patients.”
Biomarker needed
Commenting on the results, Alberto J. Espay, MD, MSc, professor of neurology at the University of Cincinnati Academic Health Center, was cautious. “These findings are of unclear applicability to any particular patient without a biomarker for a deficit of glycolysis that these drugs are presumed to affect,” Dr. Espay said. “Hence, there is no feasible or warranted change in practice as a result of this study.”
Pathogenic mechanisms are heterogeneous among patients with Parkinson’s disease, Dr. Espay added. “We will need to understand who among the large biological universe of Parkinson’s patients may have impaired energy metabolism as a pathogenic mechanism to be selected for a future clinical trial evaluating terazosin, doxazosin, or alfuzosin as a potential disease-modifying intervention.”
Parkinson’s disease is not one disease, but a group of disorders with unique biological abnormalities, said Dr. Espay. “We know so much about ‘Parkinson’s disease’ and next to nothing about the biology of individuals with Parkinson’s disease.”
This situation has enabled the development of symptomatic treatments, such as dopaminergic therapies, but failed to yield disease-modifying treatments, he said.
The University of Iowa contributed funds for this study. Dr. Simmering has received pilot funding from the University of Iowa Institute for Clinical and Translational Science. He had no conflicts of interest to disclose. Dr. Espay disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests. Treatment of BPH with terazosin (Hytrin), doxazosin (Cardura), or alfuzosin (Uroxatral), all of which enhance glycolysis, was associated with a lower risk of developing Parkinson’s disease than patients taking a drug used for the same indication, tamsulosin (Flomax), which does not affect glycolysis.
“If giving someone terazosin or similar medications truly reduces their risk of disease, these results could have significant clinical implications for neurologists,” said lead author Jacob E. Simmering, PhD, assistant professor of internal medicine at the University of Iowa, Iowa City.
There are few reliable neuroprotective treatments for Parkinson’s disease, he said. “We can manage some of the symptoms, but we can’t stop it from progressing. If a randomized trial finds the same result, this will provide a new option to slow progression of Parkinson’s disease.”
The pathogenesis of Parkinson’s disease is heterogeneous, however, and not all patients may benefit from glycolysis-enhancing drugs, the investigators noted. Future research will be needed to identify potential candidates for this treatment, and clarify the effects of these drugs, they wrote.
The findings were published online Feb. 1, 2021, in JAMA Neurology.
Time-dependent effects
The major risk factor for Parkinson’s disease is age, which is associated with impaired energy metabolism. Glycolysis is decreased among patients with Parkinson’s disease, yet impaired energy metabolism has not been investigated widely as a pathogenic factor in the disease, the authors wrote.
Studies have indicated that terazosin increases the activity of an enzyme important in glycolysis. Doxazosin and alfuzosin have a similar mechanism of action and enhance energy metabolism. Tamsulosin, a structurally unrelated drug, has the same mechanism of action as the other three drugs, but does not enhance energy metabolism.
In this report, the researchers investigated the hypothesis that patients who received therapy with terazosin, doxazosin, or alfuzosin would have a lower risk of developing Parkinson’s disease than patients receiving tamsulosin. To do that, they used health care utilization data from Denmark and the United States, including the Danish National Prescription Registry, the Danish National Patient Registry, the Danish Civil Registration System, and the Truven Health Analytics MarketScan database.
The investigators searched the records for patients who filled prescriptions for any of the four drugs of interest. They excluded any patients who developed Parkinson’s disease within 1 year of starting medication. Because use of these drugs is rare among women, they included only men in their analysis.
They looked at patient outcomes beginning at 1 year after the initiation of treatment. They also required patients to fill at least two prescriptions before the beginning of follow-up. Patients who switched from tamsulosin to any of the other drugs, or vice versa, were excluded from analysis.
The investigators used propensity-score matching to ensure that patients in the tamsulosin and terazosin/doxazosin/alfuzosin groups were similar in terms of their other potential risk factors. The primary outcome was the development of Parkinson’s disease.
They identified 52,365 propensity score–matched pairs in the Danish registries and 94,883 pairs in the Truven database. The mean age was 67.9 years in the Danish registries and 63.8 years in the Truven database, and follow-up was approximately 5 years and 3 years respectively. Baseline covariates were well balanced between cohorts.
Among Danish patients, those who took terazosin, doxazosin, or alfuzosin had a lower risk of developing Parkinson’s disease versus those who took tamsulosin (hazard ratio, 0.88). Similarly, patients in the Truven database who took terazosin, doxazosin, or alfuzosin had a lower risk of developing Parkinson’s disease than those who took tamsulosin (HR, 0.63).
In both cohorts, the risk for Parkinson’s disease among patients receiving terazosin, doxazosin, or alfuzosin, compared with those receiving tamsulosin, decreased with increasing numbers of prescriptions filled. Long-term treatment with any of the three glycolysis-enhancing drugs was associated with greater risk reduction in the Danish (HR, 0.79) and Truven (HR, 0.46) cohorts versus tamsulosin.
Differences in case definitions, which may reflect how Parkinson’s disease was managed, complicate comparisons between the Danish and Truven cohorts, said Dr. Simmering. Another challenge is the source of the data. “The Truven data set was derived from insurance claims from people with private insurance or Medicare supplemental plans,” he said. “This group is quite large but may not be representative of everyone in the United States. We would also only be able to follow people while they were on one insurance plan. If they switched coverage to a company that doesn’t contribute data, we would lose them.”
The Danish database, however, includes all residents of Denmark. Only people who left the country were lost to follow-up.
The results support the hypothesis that increasing energy in cells slows disease progression, Dr. Simmering added. “There are a few conditions, mostly REM sleep disorders, that are associated with future diagnosis of Parkinson’s disease. Right now, we don’t have anything to offer people at elevated risk of Parkinson’s disease that might prevent the disease. If a controlled trial finds that terazosin slows or prevents Parkinson’s disease, we would have something truly protective to offer these patients.”
Biomarker needed
Commenting on the results, Alberto J. Espay, MD, MSc, professor of neurology at the University of Cincinnati Academic Health Center, was cautious. “These findings are of unclear applicability to any particular patient without a biomarker for a deficit of glycolysis that these drugs are presumed to affect,” Dr. Espay said. “Hence, there is no feasible or warranted change in practice as a result of this study.”
Pathogenic mechanisms are heterogeneous among patients with Parkinson’s disease, Dr. Espay added. “We will need to understand who among the large biological universe of Parkinson’s patients may have impaired energy metabolism as a pathogenic mechanism to be selected for a future clinical trial evaluating terazosin, doxazosin, or alfuzosin as a potential disease-modifying intervention.”
Parkinson’s disease is not one disease, but a group of disorders with unique biological abnormalities, said Dr. Espay. “We know so much about ‘Parkinson’s disease’ and next to nothing about the biology of individuals with Parkinson’s disease.”
This situation has enabled the development of symptomatic treatments, such as dopaminergic therapies, but failed to yield disease-modifying treatments, he said.
The University of Iowa contributed funds for this study. Dr. Simmering has received pilot funding from the University of Iowa Institute for Clinical and Translational Science. He had no conflicts of interest to disclose. Dr. Espay disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests. Treatment of BPH with terazosin (Hytrin), doxazosin (Cardura), or alfuzosin (Uroxatral), all of which enhance glycolysis, was associated with a lower risk of developing Parkinson’s disease than patients taking a drug used for the same indication, tamsulosin (Flomax), which does not affect glycolysis.
“If giving someone terazosin or similar medications truly reduces their risk of disease, these results could have significant clinical implications for neurologists,” said lead author Jacob E. Simmering, PhD, assistant professor of internal medicine at the University of Iowa, Iowa City.
There are few reliable neuroprotective treatments for Parkinson’s disease, he said. “We can manage some of the symptoms, but we can’t stop it from progressing. If a randomized trial finds the same result, this will provide a new option to slow progression of Parkinson’s disease.”
The pathogenesis of Parkinson’s disease is heterogeneous, however, and not all patients may benefit from glycolysis-enhancing drugs, the investigators noted. Future research will be needed to identify potential candidates for this treatment, and clarify the effects of these drugs, they wrote.
The findings were published online Feb. 1, 2021, in JAMA Neurology.
Time-dependent effects
The major risk factor for Parkinson’s disease is age, which is associated with impaired energy metabolism. Glycolysis is decreased among patients with Parkinson’s disease, yet impaired energy metabolism has not been investigated widely as a pathogenic factor in the disease, the authors wrote.
Studies have indicated that terazosin increases the activity of an enzyme important in glycolysis. Doxazosin and alfuzosin have a similar mechanism of action and enhance energy metabolism. Tamsulosin, a structurally unrelated drug, has the same mechanism of action as the other three drugs, but does not enhance energy metabolism.
In this report, the researchers investigated the hypothesis that patients who received therapy with terazosin, doxazosin, or alfuzosin would have a lower risk of developing Parkinson’s disease than patients receiving tamsulosin. To do that, they used health care utilization data from Denmark and the United States, including the Danish National Prescription Registry, the Danish National Patient Registry, the Danish Civil Registration System, and the Truven Health Analytics MarketScan database.
The investigators searched the records for patients who filled prescriptions for any of the four drugs of interest. They excluded any patients who developed Parkinson’s disease within 1 year of starting medication. Because use of these drugs is rare among women, they included only men in their analysis.
They looked at patient outcomes beginning at 1 year after the initiation of treatment. They also required patients to fill at least two prescriptions before the beginning of follow-up. Patients who switched from tamsulosin to any of the other drugs, or vice versa, were excluded from analysis.
The investigators used propensity-score matching to ensure that patients in the tamsulosin and terazosin/doxazosin/alfuzosin groups were similar in terms of their other potential risk factors. The primary outcome was the development of Parkinson’s disease.
They identified 52,365 propensity score–matched pairs in the Danish registries and 94,883 pairs in the Truven database. The mean age was 67.9 years in the Danish registries and 63.8 years in the Truven database, and follow-up was approximately 5 years and 3 years respectively. Baseline covariates were well balanced between cohorts.
Among Danish patients, those who took terazosin, doxazosin, or alfuzosin had a lower risk of developing Parkinson’s disease versus those who took tamsulosin (hazard ratio, 0.88). Similarly, patients in the Truven database who took terazosin, doxazosin, or alfuzosin had a lower risk of developing Parkinson’s disease than those who took tamsulosin (HR, 0.63).
In both cohorts, the risk for Parkinson’s disease among patients receiving terazosin, doxazosin, or alfuzosin, compared with those receiving tamsulosin, decreased with increasing numbers of prescriptions filled. Long-term treatment with any of the three glycolysis-enhancing drugs was associated with greater risk reduction in the Danish (HR, 0.79) and Truven (HR, 0.46) cohorts versus tamsulosin.
Differences in case definitions, which may reflect how Parkinson’s disease was managed, complicate comparisons between the Danish and Truven cohorts, said Dr. Simmering. Another challenge is the source of the data. “The Truven data set was derived from insurance claims from people with private insurance or Medicare supplemental plans,” he said. “This group is quite large but may not be representative of everyone in the United States. We would also only be able to follow people while they were on one insurance plan. If they switched coverage to a company that doesn’t contribute data, we would lose them.”
The Danish database, however, includes all residents of Denmark. Only people who left the country were lost to follow-up.
The results support the hypothesis that increasing energy in cells slows disease progression, Dr. Simmering added. “There are a few conditions, mostly REM sleep disorders, that are associated with future diagnosis of Parkinson’s disease. Right now, we don’t have anything to offer people at elevated risk of Parkinson’s disease that might prevent the disease. If a controlled trial finds that terazosin slows or prevents Parkinson’s disease, we would have something truly protective to offer these patients.”
Biomarker needed
Commenting on the results, Alberto J. Espay, MD, MSc, professor of neurology at the University of Cincinnati Academic Health Center, was cautious. “These findings are of unclear applicability to any particular patient without a biomarker for a deficit of glycolysis that these drugs are presumed to affect,” Dr. Espay said. “Hence, there is no feasible or warranted change in practice as a result of this study.”
Pathogenic mechanisms are heterogeneous among patients with Parkinson’s disease, Dr. Espay added. “We will need to understand who among the large biological universe of Parkinson’s patients may have impaired energy metabolism as a pathogenic mechanism to be selected for a future clinical trial evaluating terazosin, doxazosin, or alfuzosin as a potential disease-modifying intervention.”
Parkinson’s disease is not one disease, but a group of disorders with unique biological abnormalities, said Dr. Espay. “We know so much about ‘Parkinson’s disease’ and next to nothing about the biology of individuals with Parkinson’s disease.”
This situation has enabled the development of symptomatic treatments, such as dopaminergic therapies, but failed to yield disease-modifying treatments, he said.
The University of Iowa contributed funds for this study. Dr. Simmering has received pilot funding from the University of Iowa Institute for Clinical and Translational Science. He had no conflicts of interest to disclose. Dr. Espay disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM NEUROLOGY
Lifestyle coaching for obesity associated with improved cardiometabolic numbers in study
Patients who received intensive lifestyle training by coaches in the primary care setting experienced improvement in several indicators of cardiometabolic health in a 2-year trial.
The 803 trial participants comprised a racially diverse, low-income population with obesity. In this study, primary care clinics were randomly assigned to provide weight-loss coaching or usual care. Patients at the intensive training clinics lost significantly more weight than the other patients, as reported in a paper published in September in the New England Journal of Medicine on the PROmoting Successful Weight Loss in Primary CarE in Louisiana (PROPEL) trial. The patients who received weight loss coaching also had significantly more improvement in HDL cholesterol levels, total to HDL cholesterol ratios, and metabolic syndrome severity score, said researchers in the new paper on the PROPEL trial, which was published in Circulation on February 8 .
“We believe that one reason for success of the program was the use of a health coach [who] was embedded in the primary care office,” said lead author Peter Katzmarzyk, PhD, associate executive director for population and public health sciences at the Pennington Biomedical Research Center, Baton Rouge, La. “This way, the patients could get their counseling in a familiar environment and did not have to go to a different setting. The coaches developed close relationships with the patients over the 2 years, and this helped develop a sense of responsibility in the patients as the coaches were helping the patients to set goals and kept them accountable.”
In the PROPEL study, 67% of patients were Black and had low health literacy scores that corresponded with less than a ninth-grade education level. The intensive lifestyle intervention program included weekly sessions with the trained health coaches over the first 6 months — 16 face-to-face and 6 over the phone — and then at least monthly for the last 18 months. The coaches had higher education degrees in nutrition, physical activity, or behavioral medicine. Before the program started, the coaches also received training in the management of obesity and related health issues, health literacy, and patient communication and education. The goal of the program was 10% weight loss, using personalized action plans on eating, dieting, and physical activity.
Those in the usual-care clinics continued receiving normal care and received newsletters on health topics, such as the importance of sleep and tips for limiting time spent sitting. The primary care physicians at those clinics also were given a presentation with Centers for Medicare & Medicaid Services (CMS) information on intensive lifestyle interventions for obesity.
Cholesterol changes in intervention vs. control group
HDL cholesterol improved significantly among the coached patients, compared with the other patients, with a mean difference of 4.1 mg/dL at 1 year and 4.6 mg/dL at 2 years (P less than .01 for both). The total cholesterol to HDL cholesterol ratio showed a similarly significant difference in decline, with a between-group difference of –0.29 at 1 year and –0.31 at 2 years (P less than .01 for both). Also, the difference in the change in metabolic severity scores were –0.40 at 1 year and –0.21 at 2 years (P less than .01 for both).
Fasting blood glucose had declined after the 1st year by a significantly greater degree in the clinics with coaching, compared with the others, but not after the second year, researchers found.
There were no significant differences seen in total cholesterol, LDL cholesterol, non-HDL cholesterol, or blood pressure. Dr. Katzmarzyk said the likely reason for no change in blood pressure was that it was already relatively well-controlled at baseline for all the patients.
Funding barriers to obesity treatment
The CMS currently cover intensive training for obesity if delivered directly by a primary care physician, according to the authors of the new paper. Dr. Katzmarzyk said he hopes that will change.
“We are hoping that the evidence provided in this study may change the way that CMS funds obesity treatment in the future by allowing an expansion of the care team,” he said.
John Flack, MD, chair of internal medicine at Southern Illinois University, Springfield, said that the main achievement of the study was that it showed that intensive weight-loss training in the primary-care setting could be accomplished in a racially diverse population with low health literacy.
“You can’t just automatically assume just because you’ve seen it in some other populations that you can replicate this in every population, so they’ve done a really good job,” he said.
That programs are eligible for reimbursement only if they’re run by primary-care physicians is an ongoing problem, he said.
“You don’t necessarily need to be a physician to do this,” Dr. Flack said.
For best results, payment for coaching should not be tied to office visits, Dr. Flack noted.
“If they’re de-tethered from the office visits and you’re paid for quality ... you’re going to build out your infrastructure differently to care for people,” he said.
Andrew Freeman, MD, associate professor of medicine at the University of Colorado, Denver, and cochair of the American College of Cardiology’s nutrition and lifestyle work group, said the findings dovetail with his experience.
“I’m a huge believer that when people need to make lifestyle changes, having someone hold their hand and guide them through the effort is incredibly rewarding and incredibly powerful,” said Dr. Freeman, who also oversees the intensive cardiac rehab program at National Jewish Health in Denver.
A program like this needs proper funding in order to work, Dr, Freeman noted. He added that, even with coaches being paid well, “if you are able to prevent just one readmission for, say, heart failure a month . . . you could be saving millions of dollars over just a couple of years.”
Dr. Katzmarzyk, Dr. Flack, and Dr. Freeman reported no relevant disclosures. Louisiana State University, Pennington Biomedical Research Center, and Montclair State University have interest in the intellectual property surrounding a weight graph used in the study. The other researchers reported grants and/or fees from Bayer, Boehringer Ingelheim, Gilead, Takeda, Novo Nordisk, and other companies.
Patients who received intensive lifestyle training by coaches in the primary care setting experienced improvement in several indicators of cardiometabolic health in a 2-year trial.
The 803 trial participants comprised a racially diverse, low-income population with obesity. In this study, primary care clinics were randomly assigned to provide weight-loss coaching or usual care. Patients at the intensive training clinics lost significantly more weight than the other patients, as reported in a paper published in September in the New England Journal of Medicine on the PROmoting Successful Weight Loss in Primary CarE in Louisiana (PROPEL) trial. The patients who received weight loss coaching also had significantly more improvement in HDL cholesterol levels, total to HDL cholesterol ratios, and metabolic syndrome severity score, said researchers in the new paper on the PROPEL trial, which was published in Circulation on February 8 .
“We believe that one reason for success of the program was the use of a health coach [who] was embedded in the primary care office,” said lead author Peter Katzmarzyk, PhD, associate executive director for population and public health sciences at the Pennington Biomedical Research Center, Baton Rouge, La. “This way, the patients could get their counseling in a familiar environment and did not have to go to a different setting. The coaches developed close relationships with the patients over the 2 years, and this helped develop a sense of responsibility in the patients as the coaches were helping the patients to set goals and kept them accountable.”
In the PROPEL study, 67% of patients were Black and had low health literacy scores that corresponded with less than a ninth-grade education level. The intensive lifestyle intervention program included weekly sessions with the trained health coaches over the first 6 months — 16 face-to-face and 6 over the phone — and then at least monthly for the last 18 months. The coaches had higher education degrees in nutrition, physical activity, or behavioral medicine. Before the program started, the coaches also received training in the management of obesity and related health issues, health literacy, and patient communication and education. The goal of the program was 10% weight loss, using personalized action plans on eating, dieting, and physical activity.
Those in the usual-care clinics continued receiving normal care and received newsletters on health topics, such as the importance of sleep and tips for limiting time spent sitting. The primary care physicians at those clinics also were given a presentation with Centers for Medicare & Medicaid Services (CMS) information on intensive lifestyle interventions for obesity.
Cholesterol changes in intervention vs. control group
HDL cholesterol improved significantly among the coached patients, compared with the other patients, with a mean difference of 4.1 mg/dL at 1 year and 4.6 mg/dL at 2 years (P less than .01 for both). The total cholesterol to HDL cholesterol ratio showed a similarly significant difference in decline, with a between-group difference of –0.29 at 1 year and –0.31 at 2 years (P less than .01 for both). Also, the difference in the change in metabolic severity scores were –0.40 at 1 year and –0.21 at 2 years (P less than .01 for both).
Fasting blood glucose had declined after the 1st year by a significantly greater degree in the clinics with coaching, compared with the others, but not after the second year, researchers found.
There were no significant differences seen in total cholesterol, LDL cholesterol, non-HDL cholesterol, or blood pressure. Dr. Katzmarzyk said the likely reason for no change in blood pressure was that it was already relatively well-controlled at baseline for all the patients.
Funding barriers to obesity treatment
The CMS currently cover intensive training for obesity if delivered directly by a primary care physician, according to the authors of the new paper. Dr. Katzmarzyk said he hopes that will change.
“We are hoping that the evidence provided in this study may change the way that CMS funds obesity treatment in the future by allowing an expansion of the care team,” he said.
John Flack, MD, chair of internal medicine at Southern Illinois University, Springfield, said that the main achievement of the study was that it showed that intensive weight-loss training in the primary-care setting could be accomplished in a racially diverse population with low health literacy.
“You can’t just automatically assume just because you’ve seen it in some other populations that you can replicate this in every population, so they’ve done a really good job,” he said.
That programs are eligible for reimbursement only if they’re run by primary-care physicians is an ongoing problem, he said.
“You don’t necessarily need to be a physician to do this,” Dr. Flack said.
For best results, payment for coaching should not be tied to office visits, Dr. Flack noted.
“If they’re de-tethered from the office visits and you’re paid for quality ... you’re going to build out your infrastructure differently to care for people,” he said.
Andrew Freeman, MD, associate professor of medicine at the University of Colorado, Denver, and cochair of the American College of Cardiology’s nutrition and lifestyle work group, said the findings dovetail with his experience.
“I’m a huge believer that when people need to make lifestyle changes, having someone hold their hand and guide them through the effort is incredibly rewarding and incredibly powerful,” said Dr. Freeman, who also oversees the intensive cardiac rehab program at National Jewish Health in Denver.
A program like this needs proper funding in order to work, Dr, Freeman noted. He added that, even with coaches being paid well, “if you are able to prevent just one readmission for, say, heart failure a month . . . you could be saving millions of dollars over just a couple of years.”
Dr. Katzmarzyk, Dr. Flack, and Dr. Freeman reported no relevant disclosures. Louisiana State University, Pennington Biomedical Research Center, and Montclair State University have interest in the intellectual property surrounding a weight graph used in the study. The other researchers reported grants and/or fees from Bayer, Boehringer Ingelheim, Gilead, Takeda, Novo Nordisk, and other companies.
Patients who received intensive lifestyle training by coaches in the primary care setting experienced improvement in several indicators of cardiometabolic health in a 2-year trial.
The 803 trial participants comprised a racially diverse, low-income population with obesity. In this study, primary care clinics were randomly assigned to provide weight-loss coaching or usual care. Patients at the intensive training clinics lost significantly more weight than the other patients, as reported in a paper published in September in the New England Journal of Medicine on the PROmoting Successful Weight Loss in Primary CarE in Louisiana (PROPEL) trial. The patients who received weight loss coaching also had significantly more improvement in HDL cholesterol levels, total to HDL cholesterol ratios, and metabolic syndrome severity score, said researchers in the new paper on the PROPEL trial, which was published in Circulation on February 8 .
“We believe that one reason for success of the program was the use of a health coach [who] was embedded in the primary care office,” said lead author Peter Katzmarzyk, PhD, associate executive director for population and public health sciences at the Pennington Biomedical Research Center, Baton Rouge, La. “This way, the patients could get their counseling in a familiar environment and did not have to go to a different setting. The coaches developed close relationships with the patients over the 2 years, and this helped develop a sense of responsibility in the patients as the coaches were helping the patients to set goals and kept them accountable.”
In the PROPEL study, 67% of patients were Black and had low health literacy scores that corresponded with less than a ninth-grade education level. The intensive lifestyle intervention program included weekly sessions with the trained health coaches over the first 6 months — 16 face-to-face and 6 over the phone — and then at least monthly for the last 18 months. The coaches had higher education degrees in nutrition, physical activity, or behavioral medicine. Before the program started, the coaches also received training in the management of obesity and related health issues, health literacy, and patient communication and education. The goal of the program was 10% weight loss, using personalized action plans on eating, dieting, and physical activity.
Those in the usual-care clinics continued receiving normal care and received newsletters on health topics, such as the importance of sleep and tips for limiting time spent sitting. The primary care physicians at those clinics also were given a presentation with Centers for Medicare & Medicaid Services (CMS) information on intensive lifestyle interventions for obesity.
Cholesterol changes in intervention vs. control group
HDL cholesterol improved significantly among the coached patients, compared with the other patients, with a mean difference of 4.1 mg/dL at 1 year and 4.6 mg/dL at 2 years (P less than .01 for both). The total cholesterol to HDL cholesterol ratio showed a similarly significant difference in decline, with a between-group difference of –0.29 at 1 year and –0.31 at 2 years (P less than .01 for both). Also, the difference in the change in metabolic severity scores were –0.40 at 1 year and –0.21 at 2 years (P less than .01 for both).
Fasting blood glucose had declined after the 1st year by a significantly greater degree in the clinics with coaching, compared with the others, but not after the second year, researchers found.
There were no significant differences seen in total cholesterol, LDL cholesterol, non-HDL cholesterol, or blood pressure. Dr. Katzmarzyk said the likely reason for no change in blood pressure was that it was already relatively well-controlled at baseline for all the patients.
Funding barriers to obesity treatment
The CMS currently cover intensive training for obesity if delivered directly by a primary care physician, according to the authors of the new paper. Dr. Katzmarzyk said he hopes that will change.
“We are hoping that the evidence provided in this study may change the way that CMS funds obesity treatment in the future by allowing an expansion of the care team,” he said.
John Flack, MD, chair of internal medicine at Southern Illinois University, Springfield, said that the main achievement of the study was that it showed that intensive weight-loss training in the primary-care setting could be accomplished in a racially diverse population with low health literacy.
“You can’t just automatically assume just because you’ve seen it in some other populations that you can replicate this in every population, so they’ve done a really good job,” he said.
That programs are eligible for reimbursement only if they’re run by primary-care physicians is an ongoing problem, he said.
“You don’t necessarily need to be a physician to do this,” Dr. Flack said.
For best results, payment for coaching should not be tied to office visits, Dr. Flack noted.
“If they’re de-tethered from the office visits and you’re paid for quality ... you’re going to build out your infrastructure differently to care for people,” he said.
Andrew Freeman, MD, associate professor of medicine at the University of Colorado, Denver, and cochair of the American College of Cardiology’s nutrition and lifestyle work group, said the findings dovetail with his experience.
“I’m a huge believer that when people need to make lifestyle changes, having someone hold their hand and guide them through the effort is incredibly rewarding and incredibly powerful,” said Dr. Freeman, who also oversees the intensive cardiac rehab program at National Jewish Health in Denver.
A program like this needs proper funding in order to work, Dr, Freeman noted. He added that, even with coaches being paid well, “if you are able to prevent just one readmission for, say, heart failure a month . . . you could be saving millions of dollars over just a couple of years.”
Dr. Katzmarzyk, Dr. Flack, and Dr. Freeman reported no relevant disclosures. Louisiana State University, Pennington Biomedical Research Center, and Montclair State University have interest in the intellectual property surrounding a weight graph used in the study. The other researchers reported grants and/or fees from Bayer, Boehringer Ingelheim, Gilead, Takeda, Novo Nordisk, and other companies.
ColCORONA: More questions than answers for colchicine in COVID-19
Science by press release and preprint has cooled clinician enthusiasm for the use of colchicine in nonhospitalized patients with COVID-19, despite a pressing need for early treatments.
As previously reported by this news organization, a Jan. 22 press release announced that the massive ColCORONA study missed its primary endpoint of hospitalization or death among 4,488 newly diagnosed patients at increased risk for hospitalization.
But it also touted that use of the anti-inflammatory drug significantly reduced the primary endpoint in 4,159 of those patients with polymerase chain reaction–confirmed COVID and led to reductions of 25%, 50%, and 44%, respectively, for hospitalizations, ventilations, and death.
Lead investigator Jean-Claude Tardif, MD, director of the Montreal Heart Institute Research Centre, deemed the findings a “medical breakthrough.”
When the preprint released a few days later, however, newly revealed confidence intervals showed colchicine did not meaningfully reduce the need for mechanical ventilation (odds ratio, 0.50; 95% confidence interval, 0.23-1.07) or death alone (OR, 0.56; 95% CI, 0.19-1.66).
Further, the significant benefit on the primary outcome came at the cost of a fivefold increase in pulmonary embolism (11 vs. 2; P = .01), which was not mentioned in the press release.
“Whether this represents a real phenomenon or simply the play of chance is not known,” Dr. Tardif and colleagues noted later in the preprint.
“I read the preprint on colchicine and I have so many questions,” Aaron E. Glatt, MD, spokesperson for the Infectious Diseases Society of America and chief of infectious diseases, Mount Sinai South Nassau, Hewlett, N.Y., said in an interview. “I’ve been burned too many times with COVID and prefer to see better data.
“People sometimes say if you wait for perfect data, people are going to die,” he said. “Yeah, but we have no idea if people are going to die from getting this drug more than not getting it. That’s what concerns me. How many pulmonary emboli are going to be fatal versus the slight benefit that the study showed?”
The pushback to the non–peer-reviewed data on social media and via emails was so strong that Dr. Tardif posted a nearly 2,000-word letter responding to the many questions at play.
Chief among them was why the trial, originally planned for 6,000 patients, was stopped early by the investigators without consultation with the data safety monitoring board (DSMB).
The explanation in the letter that logistical issues like running the study call center, budget constraints, and a perceived need to quickly communicate the results left some calling foul that the study wasn’t allowed to finish and come to a more definitive conclusion.
“I can be a little bit sympathetic to their cause but at the same time the DSMB should have said no,” said David Boulware, MD, MPH, who led a recent hydroxychloroquine trial in COVID-19. “The problem is we’re sort of left in limbo, where some people kind of believe it and some say it’s not really a thing. So it’s not really moving the needle, as far as guidelines go.”
Indeed, a Twitter poll by cardiologist James Januzzi Jr., MD, captured the uncertainty, with 28% of respondents saying the trial was “neutral,” 58% saying “maybe but meh,” and 14% saying “colchicine for all.”
Another poll cheekily asked whether ColCORONA was the Gamestop/Reddit equivalent of COVID.
“The press release really didn’t help things because it very much oversold the effect. That, I think, poisoned the well,” said Dr. Boulware, professor of medicine in infectious diseases at the University of Minnesota, Minneapolis.
“The question I’m left with is not whether colchicine works, but who does it work in,” he said. “That’s really the fundamental question because it does seem that there are probably high-risk groups in their trial and others where they benefit, whereas other groups don’t benefit. In the subgroup analysis, there was absolutely no beneficial effect in women.”
According to the authors, the number needed to treat to prevent one death or hospitalization was 71 overall, but 29 for patients with diabetes, 31 for those aged 70 years and older, 53 for patients with respiratory disease, and 25 for those with coronary disease or heart failure.
Men are at higher risk overall for poor outcomes. But “the authors didn’t present a multivariable analysis, so it is unclear if another factor, such as a differential prevalence of smoking or cardiovascular risk factors, contributed to the differential benefit,” Rachel Bender Ignacio, MD, MPH, infectious disease specialist, University of Washington, Seattle, said in an interview.
Importantly, in this pragmatic study, duration and severity of symptoms were not reported, observed Dr. Bender Ignacio, who is also a STOP-COVID-2 investigator. “We don’t yet have data as to whether colchicine shortens duration or severity of symptoms or prevents long COVID, so we need more data on that.”
The overall risk for serious adverse events was lower in the colchicine group, but the difference in pulmonary embolism (PE) was striking, she said. This could be caused by a real biologic effect, or it’s possible that persons with shortness of breath and hypoxia, without evident viral pneumonia on chest x-ray after a positive COVID-19 test, were more likely to receive a CT-PE study.
The press release also failed to include information, later noted in the preprint, that the MHI has submitted two patents related to colchicine: “Methods of treating a coronavirus infection using colchicine” and “Early administration of low-dose colchicine after myocardial infarction.”
Reached for clarification, MHI communications adviser Camille Turbide said in an interview that the first patent “simply refers to the novel concept of preventing complications of COVID-19, such as admission to the hospital, with colchicine as tested in the ColCORONA study.”
The second patent, she said, refers to the “novel concept that administering colchicine early after a major adverse cardiovascular event is better than waiting several days,” as supported by the COLCOT study, which Dr. Tardif also led.
The patents are being reviewed by authorities and “Dr. Tardif has waived his rights in these patents and does not stand to benefit financially at all if colchicine becomes used as a treatment for COVID-19,” Ms. Turbide said.
Dr. Tardif did not respond to interview requests for this story. Dr. Glatt said conflicts of interest must be assessed and are “something that is of great concern in any scientific study.”
Cardiologist Steve Nissen, MD, of the Cleveland Clinic said in an interview that, “despite the negative results, the study does suggest that colchicine might have a benefit and should be studied in future trials. These findings are not sufficient evidence to suggest use of the drug in patients infected with COVID-19.”
He noted that adverse effects like diarrhea were expected but that the excess PE was unexpected and needs greater clarification.
“Stopping the trial for administrative reasons is puzzling and undermined the ability of the trial to give a reliable answer,” Dr. Nissen said. “This is a reasonable pilot study that should be viewed as hypothesis generating but inconclusive.”
Several sources said a new trial is unlikely, particularly given the cost and 28 trials already evaluating colchicine. Among these are RECOVERY and COLCOVID, testing whether colchicine can reduce the duration of hospitalization or death in hospitalized patients with COVID-19.
Because there are so many trials ongoing right now, including for antivirals and other immunomodulators, it’s important that, if colchicine comes to routine clinical use, it provides access to treatment for those not able or willing to access clinical trials, rather than impeding clinical trial enrollment, Dr. Bender Ignacio suggested.
“We have already learned the lesson in the pandemic that early adoption of potentially promising therapies can negatively impact our ability to study and develop other promising treatments,” she said.
The trial was coordinated by the Montreal Heart Institute and funded by the government of Quebec; the National Heart, Lung, and Blood Institute of the National Institutes of Health; Montreal philanthropist Sophie Desmarais, and the COVID-19 Therapeutics Accelerator launched by the Bill & Melinda Gates Foundation, Wellcome, and Mastercard. CGI, Dacima, and Pharmascience of Montreal were also collaborators. Dr. Glatt reported no conflicts of interest. Dr. Boulware reported receiving $18 in food and beverages from Gilead Sciences in 2018.
A version of this article first appeared on Medscape.com.
Science by press release and preprint has cooled clinician enthusiasm for the use of colchicine in nonhospitalized patients with COVID-19, despite a pressing need for early treatments.
As previously reported by this news organization, a Jan. 22 press release announced that the massive ColCORONA study missed its primary endpoint of hospitalization or death among 4,488 newly diagnosed patients at increased risk for hospitalization.
But it also touted that use of the anti-inflammatory drug significantly reduced the primary endpoint in 4,159 of those patients with polymerase chain reaction–confirmed COVID and led to reductions of 25%, 50%, and 44%, respectively, for hospitalizations, ventilations, and death.
Lead investigator Jean-Claude Tardif, MD, director of the Montreal Heart Institute Research Centre, deemed the findings a “medical breakthrough.”
When the preprint released a few days later, however, newly revealed confidence intervals showed colchicine did not meaningfully reduce the need for mechanical ventilation (odds ratio, 0.50; 95% confidence interval, 0.23-1.07) or death alone (OR, 0.56; 95% CI, 0.19-1.66).
Further, the significant benefit on the primary outcome came at the cost of a fivefold increase in pulmonary embolism (11 vs. 2; P = .01), which was not mentioned in the press release.
“Whether this represents a real phenomenon or simply the play of chance is not known,” Dr. Tardif and colleagues noted later in the preprint.
“I read the preprint on colchicine and I have so many questions,” Aaron E. Glatt, MD, spokesperson for the Infectious Diseases Society of America and chief of infectious diseases, Mount Sinai South Nassau, Hewlett, N.Y., said in an interview. “I’ve been burned too many times with COVID and prefer to see better data.
“People sometimes say if you wait for perfect data, people are going to die,” he said. “Yeah, but we have no idea if people are going to die from getting this drug more than not getting it. That’s what concerns me. How many pulmonary emboli are going to be fatal versus the slight benefit that the study showed?”
The pushback to the non–peer-reviewed data on social media and via emails was so strong that Dr. Tardif posted a nearly 2,000-word letter responding to the many questions at play.
Chief among them was why the trial, originally planned for 6,000 patients, was stopped early by the investigators without consultation with the data safety monitoring board (DSMB).
The explanation in the letter that logistical issues like running the study call center, budget constraints, and a perceived need to quickly communicate the results left some calling foul that the study wasn’t allowed to finish and come to a more definitive conclusion.
“I can be a little bit sympathetic to their cause but at the same time the DSMB should have said no,” said David Boulware, MD, MPH, who led a recent hydroxychloroquine trial in COVID-19. “The problem is we’re sort of left in limbo, where some people kind of believe it and some say it’s not really a thing. So it’s not really moving the needle, as far as guidelines go.”
Indeed, a Twitter poll by cardiologist James Januzzi Jr., MD, captured the uncertainty, with 28% of respondents saying the trial was “neutral,” 58% saying “maybe but meh,” and 14% saying “colchicine for all.”
Another poll cheekily asked whether ColCORONA was the Gamestop/Reddit equivalent of COVID.
“The press release really didn’t help things because it very much oversold the effect. That, I think, poisoned the well,” said Dr. Boulware, professor of medicine in infectious diseases at the University of Minnesota, Minneapolis.
“The question I’m left with is not whether colchicine works, but who does it work in,” he said. “That’s really the fundamental question because it does seem that there are probably high-risk groups in their trial and others where they benefit, whereas other groups don’t benefit. In the subgroup analysis, there was absolutely no beneficial effect in women.”
According to the authors, the number needed to treat to prevent one death or hospitalization was 71 overall, but 29 for patients with diabetes, 31 for those aged 70 years and older, 53 for patients with respiratory disease, and 25 for those with coronary disease or heart failure.
Men are at higher risk overall for poor outcomes. But “the authors didn’t present a multivariable analysis, so it is unclear if another factor, such as a differential prevalence of smoking or cardiovascular risk factors, contributed to the differential benefit,” Rachel Bender Ignacio, MD, MPH, infectious disease specialist, University of Washington, Seattle, said in an interview.
Importantly, in this pragmatic study, duration and severity of symptoms were not reported, observed Dr. Bender Ignacio, who is also a STOP-COVID-2 investigator. “We don’t yet have data as to whether colchicine shortens duration or severity of symptoms or prevents long COVID, so we need more data on that.”
The overall risk for serious adverse events was lower in the colchicine group, but the difference in pulmonary embolism (PE) was striking, she said. This could be caused by a real biologic effect, or it’s possible that persons with shortness of breath and hypoxia, without evident viral pneumonia on chest x-ray after a positive COVID-19 test, were more likely to receive a CT-PE study.
The press release also failed to include information, later noted in the preprint, that the MHI has submitted two patents related to colchicine: “Methods of treating a coronavirus infection using colchicine” and “Early administration of low-dose colchicine after myocardial infarction.”
Reached for clarification, MHI communications adviser Camille Turbide said in an interview that the first patent “simply refers to the novel concept of preventing complications of COVID-19, such as admission to the hospital, with colchicine as tested in the ColCORONA study.”
The second patent, she said, refers to the “novel concept that administering colchicine early after a major adverse cardiovascular event is better than waiting several days,” as supported by the COLCOT study, which Dr. Tardif also led.
The patents are being reviewed by authorities and “Dr. Tardif has waived his rights in these patents and does not stand to benefit financially at all if colchicine becomes used as a treatment for COVID-19,” Ms. Turbide said.
Dr. Tardif did not respond to interview requests for this story. Dr. Glatt said conflicts of interest must be assessed and are “something that is of great concern in any scientific study.”
Cardiologist Steve Nissen, MD, of the Cleveland Clinic said in an interview that, “despite the negative results, the study does suggest that colchicine might have a benefit and should be studied in future trials. These findings are not sufficient evidence to suggest use of the drug in patients infected with COVID-19.”
He noted that adverse effects like diarrhea were expected but that the excess PE was unexpected and needs greater clarification.
“Stopping the trial for administrative reasons is puzzling and undermined the ability of the trial to give a reliable answer,” Dr. Nissen said. “This is a reasonable pilot study that should be viewed as hypothesis generating but inconclusive.”
Several sources said a new trial is unlikely, particularly given the cost and 28 trials already evaluating colchicine. Among these are RECOVERY and COLCOVID, testing whether colchicine can reduce the duration of hospitalization or death in hospitalized patients with COVID-19.
Because there are so many trials ongoing right now, including for antivirals and other immunomodulators, it’s important that, if colchicine comes to routine clinical use, it provides access to treatment for those not able or willing to access clinical trials, rather than impeding clinical trial enrollment, Dr. Bender Ignacio suggested.
“We have already learned the lesson in the pandemic that early adoption of potentially promising therapies can negatively impact our ability to study and develop other promising treatments,” she said.
The trial was coordinated by the Montreal Heart Institute and funded by the government of Quebec; the National Heart, Lung, and Blood Institute of the National Institutes of Health; Montreal philanthropist Sophie Desmarais, and the COVID-19 Therapeutics Accelerator launched by the Bill & Melinda Gates Foundation, Wellcome, and Mastercard. CGI, Dacima, and Pharmascience of Montreal were also collaborators. Dr. Glatt reported no conflicts of interest. Dr. Boulware reported receiving $18 in food and beverages from Gilead Sciences in 2018.
A version of this article first appeared on Medscape.com.
Science by press release and preprint has cooled clinician enthusiasm for the use of colchicine in nonhospitalized patients with COVID-19, despite a pressing need for early treatments.
As previously reported by this news organization, a Jan. 22 press release announced that the massive ColCORONA study missed its primary endpoint of hospitalization or death among 4,488 newly diagnosed patients at increased risk for hospitalization.
But it also touted that use of the anti-inflammatory drug significantly reduced the primary endpoint in 4,159 of those patients with polymerase chain reaction–confirmed COVID and led to reductions of 25%, 50%, and 44%, respectively, for hospitalizations, ventilations, and death.
Lead investigator Jean-Claude Tardif, MD, director of the Montreal Heart Institute Research Centre, deemed the findings a “medical breakthrough.”
When the preprint released a few days later, however, newly revealed confidence intervals showed colchicine did not meaningfully reduce the need for mechanical ventilation (odds ratio, 0.50; 95% confidence interval, 0.23-1.07) or death alone (OR, 0.56; 95% CI, 0.19-1.66).
Further, the significant benefit on the primary outcome came at the cost of a fivefold increase in pulmonary embolism (11 vs. 2; P = .01), which was not mentioned in the press release.
“Whether this represents a real phenomenon or simply the play of chance is not known,” Dr. Tardif and colleagues noted later in the preprint.
“I read the preprint on colchicine and I have so many questions,” Aaron E. Glatt, MD, spokesperson for the Infectious Diseases Society of America and chief of infectious diseases, Mount Sinai South Nassau, Hewlett, N.Y., said in an interview. “I’ve been burned too many times with COVID and prefer to see better data.
“People sometimes say if you wait for perfect data, people are going to die,” he said. “Yeah, but we have no idea if people are going to die from getting this drug more than not getting it. That’s what concerns me. How many pulmonary emboli are going to be fatal versus the slight benefit that the study showed?”
The pushback to the non–peer-reviewed data on social media and via emails was so strong that Dr. Tardif posted a nearly 2,000-word letter responding to the many questions at play.
Chief among them was why the trial, originally planned for 6,000 patients, was stopped early by the investigators without consultation with the data safety monitoring board (DSMB).
The explanation in the letter that logistical issues like running the study call center, budget constraints, and a perceived need to quickly communicate the results left some calling foul that the study wasn’t allowed to finish and come to a more definitive conclusion.
“I can be a little bit sympathetic to their cause but at the same time the DSMB should have said no,” said David Boulware, MD, MPH, who led a recent hydroxychloroquine trial in COVID-19. “The problem is we’re sort of left in limbo, where some people kind of believe it and some say it’s not really a thing. So it’s not really moving the needle, as far as guidelines go.”
Indeed, a Twitter poll by cardiologist James Januzzi Jr., MD, captured the uncertainty, with 28% of respondents saying the trial was “neutral,” 58% saying “maybe but meh,” and 14% saying “colchicine for all.”
Another poll cheekily asked whether ColCORONA was the Gamestop/Reddit equivalent of COVID.
“The press release really didn’t help things because it very much oversold the effect. That, I think, poisoned the well,” said Dr. Boulware, professor of medicine in infectious diseases at the University of Minnesota, Minneapolis.
“The question I’m left with is not whether colchicine works, but who does it work in,” he said. “That’s really the fundamental question because it does seem that there are probably high-risk groups in their trial and others where they benefit, whereas other groups don’t benefit. In the subgroup analysis, there was absolutely no beneficial effect in women.”
According to the authors, the number needed to treat to prevent one death or hospitalization was 71 overall, but 29 for patients with diabetes, 31 for those aged 70 years and older, 53 for patients with respiratory disease, and 25 for those with coronary disease or heart failure.
Men are at higher risk overall for poor outcomes. But “the authors didn’t present a multivariable analysis, so it is unclear if another factor, such as a differential prevalence of smoking or cardiovascular risk factors, contributed to the differential benefit,” Rachel Bender Ignacio, MD, MPH, infectious disease specialist, University of Washington, Seattle, said in an interview.
Importantly, in this pragmatic study, duration and severity of symptoms were not reported, observed Dr. Bender Ignacio, who is also a STOP-COVID-2 investigator. “We don’t yet have data as to whether colchicine shortens duration or severity of symptoms or prevents long COVID, so we need more data on that.”
The overall risk for serious adverse events was lower in the colchicine group, but the difference in pulmonary embolism (PE) was striking, she said. This could be caused by a real biologic effect, or it’s possible that persons with shortness of breath and hypoxia, without evident viral pneumonia on chest x-ray after a positive COVID-19 test, were more likely to receive a CT-PE study.
The press release also failed to include information, later noted in the preprint, that the MHI has submitted two patents related to colchicine: “Methods of treating a coronavirus infection using colchicine” and “Early administration of low-dose colchicine after myocardial infarction.”
Reached for clarification, MHI communications adviser Camille Turbide said in an interview that the first patent “simply refers to the novel concept of preventing complications of COVID-19, such as admission to the hospital, with colchicine as tested in the ColCORONA study.”
The second patent, she said, refers to the “novel concept that administering colchicine early after a major adverse cardiovascular event is better than waiting several days,” as supported by the COLCOT study, which Dr. Tardif also led.
The patents are being reviewed by authorities and “Dr. Tardif has waived his rights in these patents and does not stand to benefit financially at all if colchicine becomes used as a treatment for COVID-19,” Ms. Turbide said.
Dr. Tardif did not respond to interview requests for this story. Dr. Glatt said conflicts of interest must be assessed and are “something that is of great concern in any scientific study.”
Cardiologist Steve Nissen, MD, of the Cleveland Clinic said in an interview that, “despite the negative results, the study does suggest that colchicine might have a benefit and should be studied in future trials. These findings are not sufficient evidence to suggest use of the drug in patients infected with COVID-19.”
He noted that adverse effects like diarrhea were expected but that the excess PE was unexpected and needs greater clarification.
“Stopping the trial for administrative reasons is puzzling and undermined the ability of the trial to give a reliable answer,” Dr. Nissen said. “This is a reasonable pilot study that should be viewed as hypothesis generating but inconclusive.”
Several sources said a new trial is unlikely, particularly given the cost and 28 trials already evaluating colchicine. Among these are RECOVERY and COLCOVID, testing whether colchicine can reduce the duration of hospitalization or death in hospitalized patients with COVID-19.
Because there are so many trials ongoing right now, including for antivirals and other immunomodulators, it’s important that, if colchicine comes to routine clinical use, it provides access to treatment for those not able or willing to access clinical trials, rather than impeding clinical trial enrollment, Dr. Bender Ignacio suggested.
“We have already learned the lesson in the pandemic that early adoption of potentially promising therapies can negatively impact our ability to study and develop other promising treatments,” she said.
The trial was coordinated by the Montreal Heart Institute and funded by the government of Quebec; the National Heart, Lung, and Blood Institute of the National Institutes of Health; Montreal philanthropist Sophie Desmarais, and the COVID-19 Therapeutics Accelerator launched by the Bill & Melinda Gates Foundation, Wellcome, and Mastercard. CGI, Dacima, and Pharmascience of Montreal were also collaborators. Dr. Glatt reported no conflicts of interest. Dr. Boulware reported receiving $18 in food and beverages from Gilead Sciences in 2018.
A version of this article first appeared on Medscape.com.