User login
AVAHO
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]


Moderate drinking shows more benefit for older vs. younger adults
The health risks and benefits of moderate alcohol consumption are complex and remain a hot topic of debate. The data suggest that small amounts of alcohol may reduce the risk of certain health outcomes over time, but increase the risk of others, wrote Dana Bryazka, MS, a researcher at the Institute for Health Metrics and Evaluation (IHME) at the University of Washington, Seattle, and colleagues, in a paper published in the Lancet.
“The amount of alcohol that minimizes health loss is likely to depend on the distribution of underlying causes of disease burden in a given population. Since this distribution varies widely by geography, age, sex, and time, the level of alcohol consumption associated with the lowest risk to health would depend on the age structure and disease composition of that population,” the researchers wrote.
“We estimate that 1.78 million people worldwide died due to alcohol use in 2020,” Ms. Bryazka said in an interview. “It is important that alcohol consumption guidelines and policies are updated to minimize this harm, particularly in the populations at greatest risk,” she said.
“Existing alcohol consumption guidelines frequently vary by sex, with higher consumption thresholds set for males compared to females. Interestingly, with the currently available data we do not see evidence that risk of alcohol use varies by sex,” she noted.
Methods and results
In the study, the researchers conducted a systematic analysis of burden-weighted dose-response relative risk curves across 22 health outcomes. They used disease rates from the Global Burden of Diseases, Injuries, and Risk Factors Study (GBD) 2020 for the years 1990-2020 for 21 regions, including 204 countries and territories. The data were analyzed by 5-year age group, sex, and year for individuals aged 15-95 years and older. The researchers estimated the theoretical minimum risk exposure level (TMREL) and nondrinker equivalent (NDE), meaning the amount of alcohol at which the health risk equals that of a nondrinker.
One standard drink was defined as 10 g of pure alcohol, equivalent to a small glass of red wine (100 mL or 3.4 fluid ounces) at 13% alcohol by volume, a can or bottle of beer (375 mL or 12 fluid ounces) at 3.5% alcohol by volume, or a shot of whiskey or other spirits (30 mL or 1.0 fluid ounces) at 40% alcohol by volume.
Overall, the TMREL was low regardless of age, sex, time, or geography, and varied from 0 to 1.87 standard drinks per day. However, it was lowest for males aged 15-39 years (0.136 drinks per day) and only slightly higher for females aged 15-39 (0.273), representing 1-2 tenths of a standard drink.
For adults aged 40 and older without any underlying health conditions, drinking a small amount of alcohol may provide some benefits, such as reducing the risk of ischemic heart disease, stroke, and diabetes, the researchers noted. In general, for individuals aged 40-64 years, TMRELs ranged from about half a standard drink per day (0.527 drinks for males and 0.562 standard drinks per day for females) to almost two standard drinks (1.69 standard drinks per day for males and 1.82 for females). For those older than 65 years, the TMRELs represented just over 3 standard drinks per day (3.19 for males and 3.51 for females). For individuals aged 40 years and older, the distribution of disease burden varied by region, but was J-shaped across all regions, the researchers noted.
The researchers also found that those individuals consuming harmful amounts of alcohol were most likely to be aged 15-39 (59.1%) and male (76.9%).
The study findings were limited by several factors including the observational design and lack of data on drinking patterns, such as binge drinking, the researchers noted. Other limitations include the lack of data reflecting patterns of alcohol consumption during the COVID-19 pandemic, and exclusion of outcomes often associated with alcohol use, such as depression, anxiety, and dementia, that might reduce estimates of TMREL and NDE.
However, the results add to the ongoing discussion of the relationship between moderate alcohol consumption and health, the researchers said.
“The findings of this study support the development of tailored guidelines and recommendations on alcohol consumption by age and across regions and highlight that existing low consumption thresholds are too high for younger populations in all regions,” they concluded.
Consider individual factors when counseling patients
The takeaway message for primary care is that alcohol consumed in moderation can reduce the risk of ischemic heart disease, stroke, and diabetes, Ms. Bryazka noted. “However, it also increases the risk of many cancers, intentional and unintentional injuries, and infectious diseases like tuberculosis,” she said. “Of these health outcomes, young people are most likely to experience injuries, and as a result, we find that there are significant health risks associated with consuming alcohol for young people. Among older individuals, the relative proportions of these outcomes vary by geography, and so do the risks associated with consuming alcohol,” she explained.
“Importantly, our analysis was conducted at the population level; when evaluating risk at the individual level, it is also important to consider other factors such as the presence of comorbidities and interactions between alcohol and medications,” she emphasized.
Health and alcohol interaction is complicated
“These findings seemingly contradict a previous [Global Burden of Diseases, Injuries, and Risk Factors Study] estimate published in The Lancet, which emphasized that any alcohol use, regardless of amount, leads to health loss across populations,” wrote Robyn Burton, PhD, and Nick Sheron, MD, both of King’s College, London, in an accompanying comment.
However, the novel methods of weighting relative risk curves according to levels of underlying disease drive the difference in results, along with disaggregated estimates by age, sex, and region, they said.
“Across most geographical regions in this latest analysis, injuries accounted for most alcohol-related harm in younger age groups. This led to a minimum risk level of zero, or very close to zero, among individuals aged 15-39 years across all geographical regions,” which is lower than the level for older adults because of the shift in alcohol-related disease burden towards cardiovascular disease and cancers, they said. “This highlights the need to consider existing rates of disease in a population when trying to determine the total harm posed by alcohol,” the commentators wrote.
In an additional commentary, Tony Rao, MD, a visiting clinical research fellow in psychiatry at King’s College, London, noted that “the elephant in the room with this study is the interpretation of risk based on outcomes for cardiovascular disease – particularly in older people. We know that any purported health benefits from alcohol on the heart and circulation are balanced out by the increased risk from other conditions such as cancer, liver disease, and mental disorders such as depression and dementia,” Dr. Rao said. “If we are to simply draw the conclusion that older people should continue or start drinking small amounts because it protects against diseases affecting heart and circulation – which still remains controversial – other lifestyle changes or the use of drugs targeted at individual cardiovascular disorders seem like a less harmful way of improving health and wellbeing.”
Data can guide clinical practice
No previous study has examined the effect of the theoretical minimum risk of alcohol consumption by geography, age, sex, and time in the context of background disease rates, said Noel Deep, MD, in an interview.
“This study enabled the researchers to quantify the proportion of the population that consumed alcohol in amounts that exceeded the thresholds by location, age, sex, and year, and this can serve as a guide in our efforts to target the control of alcohol intake by individuals,” said Dr. Deep, a general internist in private practice in Antigo, Wisc. He also serves as chief medical officer and a staff physician at Aspirus Langlade Hospital in Antigo.
The first take-home message for clinicians is that even low levels of alcohol consumption can have deleterious effects on the health of patients, and patients should be advised accordingly based on the prevalence of diseases in that community and geographic area, Dr. Deep said. “Secondly, clinicians should also consider the risk of alcohol consumption on all forms of health impacts in a given population rather than just focusing on alcohol-related health conditions,” he added.
“This study provides us with the data to tailor our efforts in educating the clinicians and the public about the relationship between alcohol consumption and disease outcomes based on the observed disease rates in each population,” Dr. Deep explained. “The data should provide another reason for physicians to advise their younger patients, especially the younger males, to avoid or minimize alcohol use,” he said. The data also can help clinicians formulate public health messaging and community education to reduce harmful alcohol use, he added.
As for additional research, Dr. Deep said he would like to see data on the difference in the health-related effects of alcohol in binge-drinkers vs. those who regularly consume alcohol on a daily basis. “It would probably also be helpful to figure out what type of alcohol is being studied and the quality of the alcohol,” he said.
The study was supported by the Bill and Melinda Gates Foundation. Ms. Bryazka and colleagues had no financial conflicts to disclose. Dr. Burton disclosed serving as a consultant to the World Health Organization European Office for the Prevention and Control of Noncommunicable Diseases. Dr. Sheron had no financial conflicts to disclose. Dr. Deep had no financial conflicts to disclose, but serves on the Editorial Advisory Board of Internal Medicine News.
The study was supported by the Bill and Melinda Gates Foundation.
The health risks and benefits of moderate alcohol consumption are complex and remain a hot topic of debate. The data suggest that small amounts of alcohol may reduce the risk of certain health outcomes over time, but increase the risk of others, wrote Dana Bryazka, MS, a researcher at the Institute for Health Metrics and Evaluation (IHME) at the University of Washington, Seattle, and colleagues, in a paper published in the Lancet.
“The amount of alcohol that minimizes health loss is likely to depend on the distribution of underlying causes of disease burden in a given population. Since this distribution varies widely by geography, age, sex, and time, the level of alcohol consumption associated with the lowest risk to health would depend on the age structure and disease composition of that population,” the researchers wrote.
“We estimate that 1.78 million people worldwide died due to alcohol use in 2020,” Ms. Bryazka said in an interview. “It is important that alcohol consumption guidelines and policies are updated to minimize this harm, particularly in the populations at greatest risk,” she said.
“Existing alcohol consumption guidelines frequently vary by sex, with higher consumption thresholds set for males compared to females. Interestingly, with the currently available data we do not see evidence that risk of alcohol use varies by sex,” she noted.
Methods and results
In the study, the researchers conducted a systematic analysis of burden-weighted dose-response relative risk curves across 22 health outcomes. They used disease rates from the Global Burden of Diseases, Injuries, and Risk Factors Study (GBD) 2020 for the years 1990-2020 for 21 regions, including 204 countries and territories. The data were analyzed by 5-year age group, sex, and year for individuals aged 15-95 years and older. The researchers estimated the theoretical minimum risk exposure level (TMREL) and nondrinker equivalent (NDE), meaning the amount of alcohol at which the health risk equals that of a nondrinker.
One standard drink was defined as 10 g of pure alcohol, equivalent to a small glass of red wine (100 mL or 3.4 fluid ounces) at 13% alcohol by volume, a can or bottle of beer (375 mL or 12 fluid ounces) at 3.5% alcohol by volume, or a shot of whiskey or other spirits (30 mL or 1.0 fluid ounces) at 40% alcohol by volume.
Overall, the TMREL was low regardless of age, sex, time, or geography, and varied from 0 to 1.87 standard drinks per day. However, it was lowest for males aged 15-39 years (0.136 drinks per day) and only slightly higher for females aged 15-39 (0.273), representing 1-2 tenths of a standard drink.
For adults aged 40 and older without any underlying health conditions, drinking a small amount of alcohol may provide some benefits, such as reducing the risk of ischemic heart disease, stroke, and diabetes, the researchers noted. In general, for individuals aged 40-64 years, TMRELs ranged from about half a standard drink per day (0.527 drinks for males and 0.562 standard drinks per day for females) to almost two standard drinks (1.69 standard drinks per day for males and 1.82 for females). For those older than 65 years, the TMRELs represented just over 3 standard drinks per day (3.19 for males and 3.51 for females). For individuals aged 40 years and older, the distribution of disease burden varied by region, but was J-shaped across all regions, the researchers noted.
The researchers also found that those individuals consuming harmful amounts of alcohol were most likely to be aged 15-39 (59.1%) and male (76.9%).
The study findings were limited by several factors including the observational design and lack of data on drinking patterns, such as binge drinking, the researchers noted. Other limitations include the lack of data reflecting patterns of alcohol consumption during the COVID-19 pandemic, and exclusion of outcomes often associated with alcohol use, such as depression, anxiety, and dementia, that might reduce estimates of TMREL and NDE.
However, the results add to the ongoing discussion of the relationship between moderate alcohol consumption and health, the researchers said.
“The findings of this study support the development of tailored guidelines and recommendations on alcohol consumption by age and across regions and highlight that existing low consumption thresholds are too high for younger populations in all regions,” they concluded.
Consider individual factors when counseling patients
The takeaway message for primary care is that alcohol consumed in moderation can reduce the risk of ischemic heart disease, stroke, and diabetes, Ms. Bryazka noted. “However, it also increases the risk of many cancers, intentional and unintentional injuries, and infectious diseases like tuberculosis,” she said. “Of these health outcomes, young people are most likely to experience injuries, and as a result, we find that there are significant health risks associated with consuming alcohol for young people. Among older individuals, the relative proportions of these outcomes vary by geography, and so do the risks associated with consuming alcohol,” she explained.
“Importantly, our analysis was conducted at the population level; when evaluating risk at the individual level, it is also important to consider other factors such as the presence of comorbidities and interactions between alcohol and medications,” she emphasized.
Health and alcohol interaction is complicated
“These findings seemingly contradict a previous [Global Burden of Diseases, Injuries, and Risk Factors Study] estimate published in The Lancet, which emphasized that any alcohol use, regardless of amount, leads to health loss across populations,” wrote Robyn Burton, PhD, and Nick Sheron, MD, both of King’s College, London, in an accompanying comment.
However, the novel methods of weighting relative risk curves according to levels of underlying disease drive the difference in results, along with disaggregated estimates by age, sex, and region, they said.
“Across most geographical regions in this latest analysis, injuries accounted for most alcohol-related harm in younger age groups. This led to a minimum risk level of zero, or very close to zero, among individuals aged 15-39 years across all geographical regions,” which is lower than the level for older adults because of the shift in alcohol-related disease burden towards cardiovascular disease and cancers, they said. “This highlights the need to consider existing rates of disease in a population when trying to determine the total harm posed by alcohol,” the commentators wrote.
In an additional commentary, Tony Rao, MD, a visiting clinical research fellow in psychiatry at King’s College, London, noted that “the elephant in the room with this study is the interpretation of risk based on outcomes for cardiovascular disease – particularly in older people. We know that any purported health benefits from alcohol on the heart and circulation are balanced out by the increased risk from other conditions such as cancer, liver disease, and mental disorders such as depression and dementia,” Dr. Rao said. “If we are to simply draw the conclusion that older people should continue or start drinking small amounts because it protects against diseases affecting heart and circulation – which still remains controversial – other lifestyle changes or the use of drugs targeted at individual cardiovascular disorders seem like a less harmful way of improving health and wellbeing.”
Data can guide clinical practice
No previous study has examined the effect of the theoretical minimum risk of alcohol consumption by geography, age, sex, and time in the context of background disease rates, said Noel Deep, MD, in an interview.
“This study enabled the researchers to quantify the proportion of the population that consumed alcohol in amounts that exceeded the thresholds by location, age, sex, and year, and this can serve as a guide in our efforts to target the control of alcohol intake by individuals,” said Dr. Deep, a general internist in private practice in Antigo, Wisc. He also serves as chief medical officer and a staff physician at Aspirus Langlade Hospital in Antigo.
The first take-home message for clinicians is that even low levels of alcohol consumption can have deleterious effects on the health of patients, and patients should be advised accordingly based on the prevalence of diseases in that community and geographic area, Dr. Deep said. “Secondly, clinicians should also consider the risk of alcohol consumption on all forms of health impacts in a given population rather than just focusing on alcohol-related health conditions,” he added.
“This study provides us with the data to tailor our efforts in educating the clinicians and the public about the relationship between alcohol consumption and disease outcomes based on the observed disease rates in each population,” Dr. Deep explained. “The data should provide another reason for physicians to advise their younger patients, especially the younger males, to avoid or minimize alcohol use,” he said. The data also can help clinicians formulate public health messaging and community education to reduce harmful alcohol use, he added.
As for additional research, Dr. Deep said he would like to see data on the difference in the health-related effects of alcohol in binge-drinkers vs. those who regularly consume alcohol on a daily basis. “It would probably also be helpful to figure out what type of alcohol is being studied and the quality of the alcohol,” he said.
The study was supported by the Bill and Melinda Gates Foundation. Ms. Bryazka and colleagues had no financial conflicts to disclose. Dr. Burton disclosed serving as a consultant to the World Health Organization European Office for the Prevention and Control of Noncommunicable Diseases. Dr. Sheron had no financial conflicts to disclose. Dr. Deep had no financial conflicts to disclose, but serves on the Editorial Advisory Board of Internal Medicine News.
The study was supported by the Bill and Melinda Gates Foundation.
The health risks and benefits of moderate alcohol consumption are complex and remain a hot topic of debate. The data suggest that small amounts of alcohol may reduce the risk of certain health outcomes over time, but increase the risk of others, wrote Dana Bryazka, MS, a researcher at the Institute for Health Metrics and Evaluation (IHME) at the University of Washington, Seattle, and colleagues, in a paper published in the Lancet.
“The amount of alcohol that minimizes health loss is likely to depend on the distribution of underlying causes of disease burden in a given population. Since this distribution varies widely by geography, age, sex, and time, the level of alcohol consumption associated with the lowest risk to health would depend on the age structure and disease composition of that population,” the researchers wrote.
“We estimate that 1.78 million people worldwide died due to alcohol use in 2020,” Ms. Bryazka said in an interview. “It is important that alcohol consumption guidelines and policies are updated to minimize this harm, particularly in the populations at greatest risk,” she said.
“Existing alcohol consumption guidelines frequently vary by sex, with higher consumption thresholds set for males compared to females. Interestingly, with the currently available data we do not see evidence that risk of alcohol use varies by sex,” she noted.
Methods and results
In the study, the researchers conducted a systematic analysis of burden-weighted dose-response relative risk curves across 22 health outcomes. They used disease rates from the Global Burden of Diseases, Injuries, and Risk Factors Study (GBD) 2020 for the years 1990-2020 for 21 regions, including 204 countries and territories. The data were analyzed by 5-year age group, sex, and year for individuals aged 15-95 years and older. The researchers estimated the theoretical minimum risk exposure level (TMREL) and nondrinker equivalent (NDE), meaning the amount of alcohol at which the health risk equals that of a nondrinker.
One standard drink was defined as 10 g of pure alcohol, equivalent to a small glass of red wine (100 mL or 3.4 fluid ounces) at 13% alcohol by volume, a can or bottle of beer (375 mL or 12 fluid ounces) at 3.5% alcohol by volume, or a shot of whiskey or other spirits (30 mL or 1.0 fluid ounces) at 40% alcohol by volume.
Overall, the TMREL was low regardless of age, sex, time, or geography, and varied from 0 to 1.87 standard drinks per day. However, it was lowest for males aged 15-39 years (0.136 drinks per day) and only slightly higher for females aged 15-39 (0.273), representing 1-2 tenths of a standard drink.
For adults aged 40 and older without any underlying health conditions, drinking a small amount of alcohol may provide some benefits, such as reducing the risk of ischemic heart disease, stroke, and diabetes, the researchers noted. In general, for individuals aged 40-64 years, TMRELs ranged from about half a standard drink per day (0.527 drinks for males and 0.562 standard drinks per day for females) to almost two standard drinks (1.69 standard drinks per day for males and 1.82 for females). For those older than 65 years, the TMRELs represented just over 3 standard drinks per day (3.19 for males and 3.51 for females). For individuals aged 40 years and older, the distribution of disease burden varied by region, but was J-shaped across all regions, the researchers noted.
The researchers also found that those individuals consuming harmful amounts of alcohol were most likely to be aged 15-39 (59.1%) and male (76.9%).
The study findings were limited by several factors including the observational design and lack of data on drinking patterns, such as binge drinking, the researchers noted. Other limitations include the lack of data reflecting patterns of alcohol consumption during the COVID-19 pandemic, and exclusion of outcomes often associated with alcohol use, such as depression, anxiety, and dementia, that might reduce estimates of TMREL and NDE.
However, the results add to the ongoing discussion of the relationship between moderate alcohol consumption and health, the researchers said.
“The findings of this study support the development of tailored guidelines and recommendations on alcohol consumption by age and across regions and highlight that existing low consumption thresholds are too high for younger populations in all regions,” they concluded.
Consider individual factors when counseling patients
The takeaway message for primary care is that alcohol consumed in moderation can reduce the risk of ischemic heart disease, stroke, and diabetes, Ms. Bryazka noted. “However, it also increases the risk of many cancers, intentional and unintentional injuries, and infectious diseases like tuberculosis,” she said. “Of these health outcomes, young people are most likely to experience injuries, and as a result, we find that there are significant health risks associated with consuming alcohol for young people. Among older individuals, the relative proportions of these outcomes vary by geography, and so do the risks associated with consuming alcohol,” she explained.
“Importantly, our analysis was conducted at the population level; when evaluating risk at the individual level, it is also important to consider other factors such as the presence of comorbidities and interactions between alcohol and medications,” she emphasized.
Health and alcohol interaction is complicated
“These findings seemingly contradict a previous [Global Burden of Diseases, Injuries, and Risk Factors Study] estimate published in The Lancet, which emphasized that any alcohol use, regardless of amount, leads to health loss across populations,” wrote Robyn Burton, PhD, and Nick Sheron, MD, both of King’s College, London, in an accompanying comment.
However, the novel methods of weighting relative risk curves according to levels of underlying disease drive the difference in results, along with disaggregated estimates by age, sex, and region, they said.
“Across most geographical regions in this latest analysis, injuries accounted for most alcohol-related harm in younger age groups. This led to a minimum risk level of zero, or very close to zero, among individuals aged 15-39 years across all geographical regions,” which is lower than the level for older adults because of the shift in alcohol-related disease burden towards cardiovascular disease and cancers, they said. “This highlights the need to consider existing rates of disease in a population when trying to determine the total harm posed by alcohol,” the commentators wrote.
In an additional commentary, Tony Rao, MD, a visiting clinical research fellow in psychiatry at King’s College, London, noted that “the elephant in the room with this study is the interpretation of risk based on outcomes for cardiovascular disease – particularly in older people. We know that any purported health benefits from alcohol on the heart and circulation are balanced out by the increased risk from other conditions such as cancer, liver disease, and mental disorders such as depression and dementia,” Dr. Rao said. “If we are to simply draw the conclusion that older people should continue or start drinking small amounts because it protects against diseases affecting heart and circulation – which still remains controversial – other lifestyle changes or the use of drugs targeted at individual cardiovascular disorders seem like a less harmful way of improving health and wellbeing.”
Data can guide clinical practice
No previous study has examined the effect of the theoretical minimum risk of alcohol consumption by geography, age, sex, and time in the context of background disease rates, said Noel Deep, MD, in an interview.
“This study enabled the researchers to quantify the proportion of the population that consumed alcohol in amounts that exceeded the thresholds by location, age, sex, and year, and this can serve as a guide in our efforts to target the control of alcohol intake by individuals,” said Dr. Deep, a general internist in private practice in Antigo, Wisc. He also serves as chief medical officer and a staff physician at Aspirus Langlade Hospital in Antigo.
The first take-home message for clinicians is that even low levels of alcohol consumption can have deleterious effects on the health of patients, and patients should be advised accordingly based on the prevalence of diseases in that community and geographic area, Dr. Deep said. “Secondly, clinicians should also consider the risk of alcohol consumption on all forms of health impacts in a given population rather than just focusing on alcohol-related health conditions,” he added.
“This study provides us with the data to tailor our efforts in educating the clinicians and the public about the relationship between alcohol consumption and disease outcomes based on the observed disease rates in each population,” Dr. Deep explained. “The data should provide another reason for physicians to advise their younger patients, especially the younger males, to avoid or minimize alcohol use,” he said. The data also can help clinicians formulate public health messaging and community education to reduce harmful alcohol use, he added.
As for additional research, Dr. Deep said he would like to see data on the difference in the health-related effects of alcohol in binge-drinkers vs. those who regularly consume alcohol on a daily basis. “It would probably also be helpful to figure out what type of alcohol is being studied and the quality of the alcohol,” he said.
The study was supported by the Bill and Melinda Gates Foundation. Ms. Bryazka and colleagues had no financial conflicts to disclose. Dr. Burton disclosed serving as a consultant to the World Health Organization European Office for the Prevention and Control of Noncommunicable Diseases. Dr. Sheron had no financial conflicts to disclose. Dr. Deep had no financial conflicts to disclose, but serves on the Editorial Advisory Board of Internal Medicine News.
The study was supported by the Bill and Melinda Gates Foundation.
FROM THE LANCET
Risky business: Most cancer drugs don’t reach the market
, a new analysis suggests.
The researchers also found that about 8% of approved agents were subsequently taken off the market.
“The 6% is not a big surprise to us, since a few other studies using different methodologies and foci have estimated similar percentages,” Alyson Haslam, PhD, University of California, San Francisco, told this news organization. “When you look at drug development, it makes sense that you have to test a lot of drugs to get one that works, but sometimes it is nice to quantify the actual percentage in order to fully appreciate the process.”
The fact that 8% were withdrawn, however, “elicits the question of how the approval process can be improved to avoid ineffective or harmful drugs from coming onto the market,” Dr. Haslam added.
The study was published online in the International Journal of Cancer.
More desirable features?
Monitoring trends over time helps oncologists assess whether more drugs are making it to market and if certain factors make some drugs more likely to get approved.
Prior published estimates put the likelihood of approval between 6.7% and 13.4%, but these estimates were for drugs tested more than a decade ago.
To provide updated estimates, the researchers searched the literature for all oncology drugs tested in phase 1 studies during 2015 and evaluated their fate in subsequent phase 2/3 studies through FDA clearance.
Overall, the team found 803 phase 1 studies that met initial inclusion criteria; 48 trials that included only Japanese participants were excluded because these studies often evaluated drugs already approved in the United States, leaving 755 studies for the analysis.
The most common tumor types were solid/multiple tumors (24.2%), leukemias (12.8%), and lung cancer (8.5%). Just under half (47%) of the trials tested a drug as monotherapy; 43% were combination trials with one dose-escalated drug; and about 10% were combination trials with both drugs dose-escalated.
The FDA approved 51 drugs during the study period. Four (7.8%) were subsequently withdrawn: nivolumab (Opdivo) and pembrolizumab (Keytruda) for small cell lung cancer, olaratumab (Lartruvo) for soft tissue sarcoma, and melflufen (Pepaxto) for multiple myeloma. These four were not counted in the overall number of approvals.
“We really wanted to look at the end fate of drugs (within a reasonable time frame), which is why we did not include the four drugs that were initially approved but later withdrawn, although this had little impact on the main finding,” Dr. Haslam explained.
The estimated probability of any drug or drug combination tested in a phase 1 trial published in 2015 and approved that year was 1.7% and reached 6.2% by the end of 2021, the researchers found.
Monoclonal antibodies had a higher probability of being approved (15.3%), compared with inhibitors (5.1%) and chemotherapy drugs (4.2%).
The FDA was also more apt to green-light drugs tested as monotherapy, compared with drug combinations (odds ratio, 0.22). Drugs tested in monotherapy had a 9.4% probability of approval versus those tested in combination, which had a 5.6% probability of being approved when pairing a novel drug with one or more established agents, as well as when combining two novel drugs. The probability of approval was less than 1% for trials testing two established drug combinations.
Other factors that boosted the odds of FDA approval include having a response rate over 40% in phase 1 testing, demonstrating an overall survival benefit in phase 3 testing, and having the trial sponsored by a top-20 drug company, compared with a non–top-20 drug company.
Dr. Haslam found the last finding rather surprising, given the recent trend for bigger companies to invest in smaller companies who are developing promising drugs, rather than doing all of the development themselves. “In fact, a recent analysis found that only 25% of new drugs are sponsored by larger companies,” she noted.
Reached for comment, Jeff Allen, PhD, who wasn’t involved in the study, noted that “these types of landscape analyses are quite helpful in understanding the current state of oncology science and drug development.”
When looking at a 6.2% success rate for phase 1–tested oncology drugs, “it can be difficult holistically to determine all factors for which development didn’t continue,” said Dr. Allen, president and CEO of the nonprofit Friends of Cancer Research.
For instance, lack of approval may not signal the drug was a failure “but rather an artifact of circumstances such as resource limitations or reprioritization,” Dr. Allen said.
Plus, he commented, “I don’t think that we should expect all these early studies to lead to eventual approvals, but it’s clear from the authors’ findings that continued efforts to improve the overall success rate in developing new cancer medicines are greatly needed.”
The study was funded by Arnold Ventures. Dr. Haslam and Dr. Allen have no relevant disclosures. Study author Vinay Prasad, MD, MPH, receives royalties from Arnold Ventures.
A version of this article first appeared on Medscape.com.
, a new analysis suggests.
The researchers also found that about 8% of approved agents were subsequently taken off the market.
“The 6% is not a big surprise to us, since a few other studies using different methodologies and foci have estimated similar percentages,” Alyson Haslam, PhD, University of California, San Francisco, told this news organization. “When you look at drug development, it makes sense that you have to test a lot of drugs to get one that works, but sometimes it is nice to quantify the actual percentage in order to fully appreciate the process.”
The fact that 8% were withdrawn, however, “elicits the question of how the approval process can be improved to avoid ineffective or harmful drugs from coming onto the market,” Dr. Haslam added.
The study was published online in the International Journal of Cancer.
More desirable features?
Monitoring trends over time helps oncologists assess whether more drugs are making it to market and if certain factors make some drugs more likely to get approved.
Prior published estimates put the likelihood of approval between 6.7% and 13.4%, but these estimates were for drugs tested more than a decade ago.
To provide updated estimates, the researchers searched the literature for all oncology drugs tested in phase 1 studies during 2015 and evaluated their fate in subsequent phase 2/3 studies through FDA clearance.
Overall, the team found 803 phase 1 studies that met initial inclusion criteria; 48 trials that included only Japanese participants were excluded because these studies often evaluated drugs already approved in the United States, leaving 755 studies for the analysis.
The most common tumor types were solid/multiple tumors (24.2%), leukemias (12.8%), and lung cancer (8.5%). Just under half (47%) of the trials tested a drug as monotherapy; 43% were combination trials with one dose-escalated drug; and about 10% were combination trials with both drugs dose-escalated.
The FDA approved 51 drugs during the study period. Four (7.8%) were subsequently withdrawn: nivolumab (Opdivo) and pembrolizumab (Keytruda) for small cell lung cancer, olaratumab (Lartruvo) for soft tissue sarcoma, and melflufen (Pepaxto) for multiple myeloma. These four were not counted in the overall number of approvals.
“We really wanted to look at the end fate of drugs (within a reasonable time frame), which is why we did not include the four drugs that were initially approved but later withdrawn, although this had little impact on the main finding,” Dr. Haslam explained.
The estimated probability of any drug or drug combination tested in a phase 1 trial published in 2015 and approved that year was 1.7% and reached 6.2% by the end of 2021, the researchers found.
Monoclonal antibodies had a higher probability of being approved (15.3%), compared with inhibitors (5.1%) and chemotherapy drugs (4.2%).
The FDA was also more apt to green-light drugs tested as monotherapy, compared with drug combinations (odds ratio, 0.22). Drugs tested in monotherapy had a 9.4% probability of approval versus those tested in combination, which had a 5.6% probability of being approved when pairing a novel drug with one or more established agents, as well as when combining two novel drugs. The probability of approval was less than 1% for trials testing two established drug combinations.
Other factors that boosted the odds of FDA approval include having a response rate over 40% in phase 1 testing, demonstrating an overall survival benefit in phase 3 testing, and having the trial sponsored by a top-20 drug company, compared with a non–top-20 drug company.
Dr. Haslam found the last finding rather surprising, given the recent trend for bigger companies to invest in smaller companies who are developing promising drugs, rather than doing all of the development themselves. “In fact, a recent analysis found that only 25% of new drugs are sponsored by larger companies,” she noted.
Reached for comment, Jeff Allen, PhD, who wasn’t involved in the study, noted that “these types of landscape analyses are quite helpful in understanding the current state of oncology science and drug development.”
When looking at a 6.2% success rate for phase 1–tested oncology drugs, “it can be difficult holistically to determine all factors for which development didn’t continue,” said Dr. Allen, president and CEO of the nonprofit Friends of Cancer Research.
For instance, lack of approval may not signal the drug was a failure “but rather an artifact of circumstances such as resource limitations or reprioritization,” Dr. Allen said.
Plus, he commented, “I don’t think that we should expect all these early studies to lead to eventual approvals, but it’s clear from the authors’ findings that continued efforts to improve the overall success rate in developing new cancer medicines are greatly needed.”
The study was funded by Arnold Ventures. Dr. Haslam and Dr. Allen have no relevant disclosures. Study author Vinay Prasad, MD, MPH, receives royalties from Arnold Ventures.
A version of this article first appeared on Medscape.com.
, a new analysis suggests.
The researchers also found that about 8% of approved agents were subsequently taken off the market.
“The 6% is not a big surprise to us, since a few other studies using different methodologies and foci have estimated similar percentages,” Alyson Haslam, PhD, University of California, San Francisco, told this news organization. “When you look at drug development, it makes sense that you have to test a lot of drugs to get one that works, but sometimes it is nice to quantify the actual percentage in order to fully appreciate the process.”
The fact that 8% were withdrawn, however, “elicits the question of how the approval process can be improved to avoid ineffective or harmful drugs from coming onto the market,” Dr. Haslam added.
The study was published online in the International Journal of Cancer.
More desirable features?
Monitoring trends over time helps oncologists assess whether more drugs are making it to market and if certain factors make some drugs more likely to get approved.
Prior published estimates put the likelihood of approval between 6.7% and 13.4%, but these estimates were for drugs tested more than a decade ago.
To provide updated estimates, the researchers searched the literature for all oncology drugs tested in phase 1 studies during 2015 and evaluated their fate in subsequent phase 2/3 studies through FDA clearance.
Overall, the team found 803 phase 1 studies that met initial inclusion criteria; 48 trials that included only Japanese participants were excluded because these studies often evaluated drugs already approved in the United States, leaving 755 studies for the analysis.
The most common tumor types were solid/multiple tumors (24.2%), leukemias (12.8%), and lung cancer (8.5%). Just under half (47%) of the trials tested a drug as monotherapy; 43% were combination trials with one dose-escalated drug; and about 10% were combination trials with both drugs dose-escalated.
The FDA approved 51 drugs during the study period. Four (7.8%) were subsequently withdrawn: nivolumab (Opdivo) and pembrolizumab (Keytruda) for small cell lung cancer, olaratumab (Lartruvo) for soft tissue sarcoma, and melflufen (Pepaxto) for multiple myeloma. These four were not counted in the overall number of approvals.
“We really wanted to look at the end fate of drugs (within a reasonable time frame), which is why we did not include the four drugs that were initially approved but later withdrawn, although this had little impact on the main finding,” Dr. Haslam explained.
The estimated probability of any drug or drug combination tested in a phase 1 trial published in 2015 and approved that year was 1.7% and reached 6.2% by the end of 2021, the researchers found.
Monoclonal antibodies had a higher probability of being approved (15.3%), compared with inhibitors (5.1%) and chemotherapy drugs (4.2%).
The FDA was also more apt to green-light drugs tested as monotherapy, compared with drug combinations (odds ratio, 0.22). Drugs tested in monotherapy had a 9.4% probability of approval versus those tested in combination, which had a 5.6% probability of being approved when pairing a novel drug with one or more established agents, as well as when combining two novel drugs. The probability of approval was less than 1% for trials testing two established drug combinations.
Other factors that boosted the odds of FDA approval include having a response rate over 40% in phase 1 testing, demonstrating an overall survival benefit in phase 3 testing, and having the trial sponsored by a top-20 drug company, compared with a non–top-20 drug company.
Dr. Haslam found the last finding rather surprising, given the recent trend for bigger companies to invest in smaller companies who are developing promising drugs, rather than doing all of the development themselves. “In fact, a recent analysis found that only 25% of new drugs are sponsored by larger companies,” she noted.
Reached for comment, Jeff Allen, PhD, who wasn’t involved in the study, noted that “these types of landscape analyses are quite helpful in understanding the current state of oncology science and drug development.”
When looking at a 6.2% success rate for phase 1–tested oncology drugs, “it can be difficult holistically to determine all factors for which development didn’t continue,” said Dr. Allen, president and CEO of the nonprofit Friends of Cancer Research.
For instance, lack of approval may not signal the drug was a failure “but rather an artifact of circumstances such as resource limitations or reprioritization,” Dr. Allen said.
Plus, he commented, “I don’t think that we should expect all these early studies to lead to eventual approvals, but it’s clear from the authors’ findings that continued efforts to improve the overall success rate in developing new cancer medicines are greatly needed.”
The study was funded by Arnold Ventures. Dr. Haslam and Dr. Allen have no relevant disclosures. Study author Vinay Prasad, MD, MPH, receives royalties from Arnold Ventures.
A version of this article first appeared on Medscape.com.
FROM INTERNATIONAL JOURNAL OF CANCER
U.S. hot, cold spots of young-onset CRC may help target interventions
The so-called hot and cold spots of mortality from young-onset CRC differed slightly for people younger than 50 and those younger than 35, report the researchers, who say such studies may lead to better understanding of the underlying factors as well as to targeted interventions.
The authors suggest that deaths in the youngest young-onset CRC individuals “may be driven by a distinct set of factors, compared with deaths among older young-onset CRC and average-onset CRC patients.”
They add that “unmeasured factors ... may drive anomalous young-onset CRC mortality rates, either independently or in conjunction with demographic [and] modifiable variables accounted for here.”
The research was published online in Gastroenterology.
Incidence, mortality rates on the rise
The incidence and mortality rates of young-onset CRC have been increasing for decades, the authors write, but it has only recently begun to attract public health attention.
Risk factors and prognostic indicators, such as smoking, obesity, alcohol consumption, diabetes, sex, race, and socioeconomic factors, have been implicated in the development of the condition.
Geospatial distribution of young-onset CRC adds an “important [layer] for understanding the underlying drivers of mortality and allocating public health resources,” the authors write.
It is “too soon” to draw conclusions about the cause of the hot and cold spots, cautioned senior author Stephanie L. Schmit, PhD, vice chair of the Genomic Medicine Institute at the Lerner Research Institute, Cleveland Clinic.
Speaking to this news organization, she said, “Additional factors like proximity to primary care, gastroenterology, and cancer care facilities or novel environmental exposures may contribute to hot spots.”
On the other hand, “lifestyle factors like diet and exercise might contribute to some extent to cold spots,” she added.
While Dr. Schmit said it would be “challenging” to replicate the findings nationally, “further analyses at more granular geographic levels would be incredibly helpful.”
Exploring the geographical distribution
To explore the geographical distribution of young-onset CRC mortality, the researchers gathered 20 years of data on more than 1 million CRC deaths from 3,036 U.S. counties. With aggregated county-level information from 1999 to 2019, they derived mortality rates from CDC WONDER underlying cause of death data.
Over the study period, there were 69,976 deaths from CRC among individuals diagnosed before age 50, including 7,325 persons diagnosed younger than 35. Most CRC deaths (1,033,541) occurred in people diagnosed at age 50 and older.
The researchers calculated an average county-level young-onset CRC mortality rate of 1.78 deaths per 100,000 population, compared with a CRC mortality rate of 56.82 per 100,000 population among individuals 50 and older.
Overall, for individuals younger than 50 at diagnosis, the researchers found two hot spots – in the Southeast (relative risk, 1.24) and in the Great Lakes region (RR, 1.10). They identified cold spots in lower Wisconsin (RR, 0.87), the Northeast (RR, 0.92), southwest Texas (RR, 0.90), and Western counties more broadly, including Alaska (RR, 0.82).
Further analysis of those diagnosed when younger than 35 revealed two significant young-onset CRC mortality hot spots – in the Northeast (RR, 1.25) and the upper Midwest (RR, 1.11). In this youngest group, the team also found three significant cold spots – in the Southwest (RR, 0.74), in California (RR, 0.78), and in the Mountain West (RR, 0.82).
Among those aged 35-49 years at diagnosis, researchers found three hot spots – two in the Southeast (RR,1.20 and 1.16) and in the Great Lakes region (RR, 1.12). Several cold spots emerged from the mortality data on young-onset CRC in this age group – in the Pacific/Mountain West (RR, 0.90), in California (RR, 0.82), southern Texas (RR, 0.89), and the Southwest more broadly (RR, 0.86).
“Though cold spots were similar across strata, young-onset CRC hot spots shifted southward in the 35-49 age stratum in comparison to the less than 35 group,” the team notes.
They acknowledge several limitations to the study, including its “ecological nature” and the lack of adjustment for stage at diagnosis.
In comments to this news organization, Andrew T. Chan, MD, MPH, of Massachusetts General Hospital and Harvard Medical School, Boston, said the approach used by the researchers was “very interesting.”
Dr. Chan said that this is “one of the first studies that has given us insight into whether there is potential geographic variation in the incidence of young-onset colorectal cancer.”
This, he continued, is “very helpful in terms of thinking about potential risk factors for early-onset cancer and giving us more information about where we might want to focus our efforts in terms of prevention.”
Dr. Chan added that another interesting aspect of the study was that “the patterns might be different, depending on how you define early-onset cancer,” whether as “very-early onset,” defined as onset in those younger than 35, or the “less stringent definition” of 35-49 years.
He said that, “within the group that we’re calling very-early onset, there may be enriched factors,” compared with people who are “a little bit older.”
The research was supported by a National Cancer Institute of the National Institutes of Health grant to Case Comprehensive Cancer Center. Dr. Schmit reports no relevant financial relationships. Other authors have relationships with Exelixis, Tempus, Olympus, Anthos, Bayer, BMS, Janssen, Nektar Therapeutics, Pfizer, Sanofi, and WebMD/Medscape. Dr. Chan reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The so-called hot and cold spots of mortality from young-onset CRC differed slightly for people younger than 50 and those younger than 35, report the researchers, who say such studies may lead to better understanding of the underlying factors as well as to targeted interventions.
The authors suggest that deaths in the youngest young-onset CRC individuals “may be driven by a distinct set of factors, compared with deaths among older young-onset CRC and average-onset CRC patients.”
They add that “unmeasured factors ... may drive anomalous young-onset CRC mortality rates, either independently or in conjunction with demographic [and] modifiable variables accounted for here.”
The research was published online in Gastroenterology.
Incidence, mortality rates on the rise
The incidence and mortality rates of young-onset CRC have been increasing for decades, the authors write, but it has only recently begun to attract public health attention.
Risk factors and prognostic indicators, such as smoking, obesity, alcohol consumption, diabetes, sex, race, and socioeconomic factors, have been implicated in the development of the condition.
Geospatial distribution of young-onset CRC adds an “important [layer] for understanding the underlying drivers of mortality and allocating public health resources,” the authors write.
It is “too soon” to draw conclusions about the cause of the hot and cold spots, cautioned senior author Stephanie L. Schmit, PhD, vice chair of the Genomic Medicine Institute at the Lerner Research Institute, Cleveland Clinic.
Speaking to this news organization, she said, “Additional factors like proximity to primary care, gastroenterology, and cancer care facilities or novel environmental exposures may contribute to hot spots.”
On the other hand, “lifestyle factors like diet and exercise might contribute to some extent to cold spots,” she added.
While Dr. Schmit said it would be “challenging” to replicate the findings nationally, “further analyses at more granular geographic levels would be incredibly helpful.”
Exploring the geographical distribution
To explore the geographical distribution of young-onset CRC mortality, the researchers gathered 20 years of data on more than 1 million CRC deaths from 3,036 U.S. counties. With aggregated county-level information from 1999 to 2019, they derived mortality rates from CDC WONDER underlying cause of death data.
Over the study period, there were 69,976 deaths from CRC among individuals diagnosed before age 50, including 7,325 persons diagnosed younger than 35. Most CRC deaths (1,033,541) occurred in people diagnosed at age 50 and older.
The researchers calculated an average county-level young-onset CRC mortality rate of 1.78 deaths per 100,000 population, compared with a CRC mortality rate of 56.82 per 100,000 population among individuals 50 and older.
Overall, for individuals younger than 50 at diagnosis, the researchers found two hot spots – in the Southeast (relative risk, 1.24) and in the Great Lakes region (RR, 1.10). They identified cold spots in lower Wisconsin (RR, 0.87), the Northeast (RR, 0.92), southwest Texas (RR, 0.90), and Western counties more broadly, including Alaska (RR, 0.82).
Further analysis of those diagnosed when younger than 35 revealed two significant young-onset CRC mortality hot spots – in the Northeast (RR, 1.25) and the upper Midwest (RR, 1.11). In this youngest group, the team also found three significant cold spots – in the Southwest (RR, 0.74), in California (RR, 0.78), and in the Mountain West (RR, 0.82).
Among those aged 35-49 years at diagnosis, researchers found three hot spots – two in the Southeast (RR,1.20 and 1.16) and in the Great Lakes region (RR, 1.12). Several cold spots emerged from the mortality data on young-onset CRC in this age group – in the Pacific/Mountain West (RR, 0.90), in California (RR, 0.82), southern Texas (RR, 0.89), and the Southwest more broadly (RR, 0.86).
“Though cold spots were similar across strata, young-onset CRC hot spots shifted southward in the 35-49 age stratum in comparison to the less than 35 group,” the team notes.
They acknowledge several limitations to the study, including its “ecological nature” and the lack of adjustment for stage at diagnosis.
In comments to this news organization, Andrew T. Chan, MD, MPH, of Massachusetts General Hospital and Harvard Medical School, Boston, said the approach used by the researchers was “very interesting.”
Dr. Chan said that this is “one of the first studies that has given us insight into whether there is potential geographic variation in the incidence of young-onset colorectal cancer.”
This, he continued, is “very helpful in terms of thinking about potential risk factors for early-onset cancer and giving us more information about where we might want to focus our efforts in terms of prevention.”
Dr. Chan added that another interesting aspect of the study was that “the patterns might be different, depending on how you define early-onset cancer,” whether as “very-early onset,” defined as onset in those younger than 35, or the “less stringent definition” of 35-49 years.
He said that, “within the group that we’re calling very-early onset, there may be enriched factors,” compared with people who are “a little bit older.”
The research was supported by a National Cancer Institute of the National Institutes of Health grant to Case Comprehensive Cancer Center. Dr. Schmit reports no relevant financial relationships. Other authors have relationships with Exelixis, Tempus, Olympus, Anthos, Bayer, BMS, Janssen, Nektar Therapeutics, Pfizer, Sanofi, and WebMD/Medscape. Dr. Chan reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The so-called hot and cold spots of mortality from young-onset CRC differed slightly for people younger than 50 and those younger than 35, report the researchers, who say such studies may lead to better understanding of the underlying factors as well as to targeted interventions.
The authors suggest that deaths in the youngest young-onset CRC individuals “may be driven by a distinct set of factors, compared with deaths among older young-onset CRC and average-onset CRC patients.”
They add that “unmeasured factors ... may drive anomalous young-onset CRC mortality rates, either independently or in conjunction with demographic [and] modifiable variables accounted for here.”
The research was published online in Gastroenterology.
Incidence, mortality rates on the rise
The incidence and mortality rates of young-onset CRC have been increasing for decades, the authors write, but it has only recently begun to attract public health attention.
Risk factors and prognostic indicators, such as smoking, obesity, alcohol consumption, diabetes, sex, race, and socioeconomic factors, have been implicated in the development of the condition.
Geospatial distribution of young-onset CRC adds an “important [layer] for understanding the underlying drivers of mortality and allocating public health resources,” the authors write.
It is “too soon” to draw conclusions about the cause of the hot and cold spots, cautioned senior author Stephanie L. Schmit, PhD, vice chair of the Genomic Medicine Institute at the Lerner Research Institute, Cleveland Clinic.
Speaking to this news organization, she said, “Additional factors like proximity to primary care, gastroenterology, and cancer care facilities or novel environmental exposures may contribute to hot spots.”
On the other hand, “lifestyle factors like diet and exercise might contribute to some extent to cold spots,” she added.
While Dr. Schmit said it would be “challenging” to replicate the findings nationally, “further analyses at more granular geographic levels would be incredibly helpful.”
Exploring the geographical distribution
To explore the geographical distribution of young-onset CRC mortality, the researchers gathered 20 years of data on more than 1 million CRC deaths from 3,036 U.S. counties. With aggregated county-level information from 1999 to 2019, they derived mortality rates from CDC WONDER underlying cause of death data.
Over the study period, there were 69,976 deaths from CRC among individuals diagnosed before age 50, including 7,325 persons diagnosed younger than 35. Most CRC deaths (1,033,541) occurred in people diagnosed at age 50 and older.
The researchers calculated an average county-level young-onset CRC mortality rate of 1.78 deaths per 100,000 population, compared with a CRC mortality rate of 56.82 per 100,000 population among individuals 50 and older.
Overall, for individuals younger than 50 at diagnosis, the researchers found two hot spots – in the Southeast (relative risk, 1.24) and in the Great Lakes region (RR, 1.10). They identified cold spots in lower Wisconsin (RR, 0.87), the Northeast (RR, 0.92), southwest Texas (RR, 0.90), and Western counties more broadly, including Alaska (RR, 0.82).
Further analysis of those diagnosed when younger than 35 revealed two significant young-onset CRC mortality hot spots – in the Northeast (RR, 1.25) and the upper Midwest (RR, 1.11). In this youngest group, the team also found three significant cold spots – in the Southwest (RR, 0.74), in California (RR, 0.78), and in the Mountain West (RR, 0.82).
Among those aged 35-49 years at diagnosis, researchers found three hot spots – two in the Southeast (RR,1.20 and 1.16) and in the Great Lakes region (RR, 1.12). Several cold spots emerged from the mortality data on young-onset CRC in this age group – in the Pacific/Mountain West (RR, 0.90), in California (RR, 0.82), southern Texas (RR, 0.89), and the Southwest more broadly (RR, 0.86).
“Though cold spots were similar across strata, young-onset CRC hot spots shifted southward in the 35-49 age stratum in comparison to the less than 35 group,” the team notes.
They acknowledge several limitations to the study, including its “ecological nature” and the lack of adjustment for stage at diagnosis.
In comments to this news organization, Andrew T. Chan, MD, MPH, of Massachusetts General Hospital and Harvard Medical School, Boston, said the approach used by the researchers was “very interesting.”
Dr. Chan said that this is “one of the first studies that has given us insight into whether there is potential geographic variation in the incidence of young-onset colorectal cancer.”
This, he continued, is “very helpful in terms of thinking about potential risk factors for early-onset cancer and giving us more information about where we might want to focus our efforts in terms of prevention.”
Dr. Chan added that another interesting aspect of the study was that “the patterns might be different, depending on how you define early-onset cancer,” whether as “very-early onset,” defined as onset in those younger than 35, or the “less stringent definition” of 35-49 years.
He said that, “within the group that we’re calling very-early onset, there may be enriched factors,” compared with people who are “a little bit older.”
The research was supported by a National Cancer Institute of the National Institutes of Health grant to Case Comprehensive Cancer Center. Dr. Schmit reports no relevant financial relationships. Other authors have relationships with Exelixis, Tempus, Olympus, Anthos, Bayer, BMS, Janssen, Nektar Therapeutics, Pfizer, Sanofi, and WebMD/Medscape. Dr. Chan reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM GASTROENTEROLOGY
Amazon involved with new cancer vaccine clinical trial
The trial is aimed at finding “personalized vaccines” to treat breast cancer and melanoma. The phase 1 trial is recruiting 20 people over the age of 18 to study the safety of the vaccines, according to CNBC.
The Fred Hutchinson Cancer Research Center and University of Washington Cancer Consortium are listed as the researchers of the clinical trial, and Amazon is listed as a collaborator, according to a filing on the ClinicalTrials.gov database.
“Amazon is contributing scientific and machine learning expertise to a partnership with Fred Hutch to explore the development of a personalized treatment for certain forms of cancer,” an Amazon spokesperson told CNBC.
“It’s very early, but Fred Hutch recently received permission from the U.S. Food and Drug Administration to proceed with a phase 1 clinical trial, and it’s unclear whether it will be successful,” the spokesperson said. “This will be a long, multiyear process – should it progress, we would be open to working with other organizations in health care and life sciences that might also be interested in similar efforts.”
In recent years, Amazon has grown its presence in the health care industry, CNBC reported. The company launched an online pharmacy in 2020, developed a telehealth service called Amazon Care, and released its own COVID-19 test during the pandemic.
A research and development group inside Amazon, known as Grand Challenge, oversaw the company’s early cancer vaccine effort, according to Business Insider. It’s now under the purview of a cancer research team that reports to Robert Williams, the company’s vice president of devices.
The study was first posted on ClinicalTrials.gov in October 2021 and began recruiting patients on June 9, according to the filing. The phase 1 trial is expected to run through November 2023.
The phase 1 trial will study the safety of personalized vaccines to treat patients with late-stage melanoma or hormone receptor-positive HER2-negative breast cancer which has either spread to other parts of the body or doesn’t respond to treatment.
More information about the study can be found on ClinicalTrials.gov under the identifier NCT05098210.
A version of this article first appeared on WebMD.com.
The trial is aimed at finding “personalized vaccines” to treat breast cancer and melanoma. The phase 1 trial is recruiting 20 people over the age of 18 to study the safety of the vaccines, according to CNBC.
The Fred Hutchinson Cancer Research Center and University of Washington Cancer Consortium are listed as the researchers of the clinical trial, and Amazon is listed as a collaborator, according to a filing on the ClinicalTrials.gov database.
“Amazon is contributing scientific and machine learning expertise to a partnership with Fred Hutch to explore the development of a personalized treatment for certain forms of cancer,” an Amazon spokesperson told CNBC.
“It’s very early, but Fred Hutch recently received permission from the U.S. Food and Drug Administration to proceed with a phase 1 clinical trial, and it’s unclear whether it will be successful,” the spokesperson said. “This will be a long, multiyear process – should it progress, we would be open to working with other organizations in health care and life sciences that might also be interested in similar efforts.”
In recent years, Amazon has grown its presence in the health care industry, CNBC reported. The company launched an online pharmacy in 2020, developed a telehealth service called Amazon Care, and released its own COVID-19 test during the pandemic.
A research and development group inside Amazon, known as Grand Challenge, oversaw the company’s early cancer vaccine effort, according to Business Insider. It’s now under the purview of a cancer research team that reports to Robert Williams, the company’s vice president of devices.
The study was first posted on ClinicalTrials.gov in October 2021 and began recruiting patients on June 9, according to the filing. The phase 1 trial is expected to run through November 2023.
The phase 1 trial will study the safety of personalized vaccines to treat patients with late-stage melanoma or hormone receptor-positive HER2-negative breast cancer which has either spread to other parts of the body or doesn’t respond to treatment.
More information about the study can be found on ClinicalTrials.gov under the identifier NCT05098210.
A version of this article first appeared on WebMD.com.
The trial is aimed at finding “personalized vaccines” to treat breast cancer and melanoma. The phase 1 trial is recruiting 20 people over the age of 18 to study the safety of the vaccines, according to CNBC.
The Fred Hutchinson Cancer Research Center and University of Washington Cancer Consortium are listed as the researchers of the clinical trial, and Amazon is listed as a collaborator, according to a filing on the ClinicalTrials.gov database.
“Amazon is contributing scientific and machine learning expertise to a partnership with Fred Hutch to explore the development of a personalized treatment for certain forms of cancer,” an Amazon spokesperson told CNBC.
“It’s very early, but Fred Hutch recently received permission from the U.S. Food and Drug Administration to proceed with a phase 1 clinical trial, and it’s unclear whether it will be successful,” the spokesperson said. “This will be a long, multiyear process – should it progress, we would be open to working with other organizations in health care and life sciences that might also be interested in similar efforts.”
In recent years, Amazon has grown its presence in the health care industry, CNBC reported. The company launched an online pharmacy in 2020, developed a telehealth service called Amazon Care, and released its own COVID-19 test during the pandemic.
A research and development group inside Amazon, known as Grand Challenge, oversaw the company’s early cancer vaccine effort, according to Business Insider. It’s now under the purview of a cancer research team that reports to Robert Williams, the company’s vice president of devices.
The study was first posted on ClinicalTrials.gov in October 2021 and began recruiting patients on June 9, according to the filing. The phase 1 trial is expected to run through November 2023.
The phase 1 trial will study the safety of personalized vaccines to treat patients with late-stage melanoma or hormone receptor-positive HER2-negative breast cancer which has either spread to other parts of the body or doesn’t respond to treatment.
More information about the study can be found on ClinicalTrials.gov under the identifier NCT05098210.
A version of this article first appeared on WebMD.com.
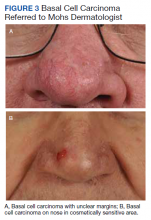
Surgical Treatment of Nonmelanoma Skin Cancer in Older Adult Veterans
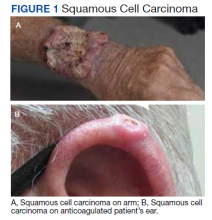
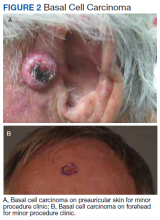
Skin cancer is the most diagnosed cancer in the United States. Nonmelanoma skin cancers (NMSC), which include basal cell carcinoma and squamous cell carcinoma, are usually cured with removal.1 The incidence of NMSC increases with age and is commonly found in nursing homes and geriatric units. These cancers are not usually metastatic or fatal but can cause local destruction and disfigurement if neglected.2 The current standard of care is to treat diagnosed NMSC; however, the dermatology and geriatric care literature have questioned the logic of treating asymptomatic skin cancers that will not affect a patient’s life expectancy.2-4
Forty-seven percent of the current living veteran population is aged ≥ 65 years.5 Older adult patients are frequently referred to the US Department of Veterans Affairs (VA) surgical service for the treatment of NMSC. The veteran population includes a higher percentage of individuals at an elevated risk of skin cancers (older, White, and male) compared with the general population.6 World War II veterans deployed in regions closer to the equator have been found to have an elevated risk of melanoma and nonmelanoma skin carcinomas.7 A retrospective study of Vietnam veterans exposed to Agent Orange (2,3,7,8-tetrachlorodibenzodioxin) found a significantly higher risk of invasive NMSC in Fitzpatrick skin types I-IV compared with an age-matched subset of the general population.8 Younger veterans who were deployed in Afghanistan and Iraq for Operation Enduring Freedom/Operation Iraqi Freedom worked at more equatorial latitudes than the rest of the US population and may be at increased risk of NMSC. Inadequate sunscreen access, immediate safety concerns, outdoor recreational activities, harsh weather, and insufficient emphasis on sun protection have created a multifactorial challenge for the military population. Riemenschneider and colleagues recommended targeted screening for at-risk veteran patients and prioritizing annual skin cancer screenings during medical mission physical examinations for active military.7
The plastic surgery service regularly receives consults from dermatology, general surgery, and primary care to remove skin cancers on the face, scalp, hands, and forearms. Skin cancer treatment can create serious hardships for older adult patients and their families with multiple appointments for the consult, procedure, and follow-up. Patients are often told to hold their anticoagulant medications when the surgery will be performed on a highly vascular region, such as the scalp or face. This can create wide swings in their laboratory test values and result in life-threatening complications from either bleeding or clotting. The appropriateness of offering surgery to patients with serious comorbidities and a limited life expectancy has been questioned.2-4 The purpose of this study was to measure the morbidity and unrelated 5-year mortality for patients with skin cancer referred to the plastic surgery service to help patients and families make a more informed treatment decision, particularly when the patients are aged > 80 years and have significant life-threatening comorbidities.
Methods
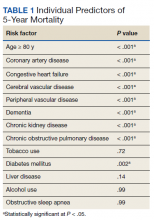
The University of Florida and Malcom Randall VA Medical Center Institutional review board in Gainesville, approved a retrospective review of all consults completed by the plastic surgery service for the treatment of NMSC performed from July 1, 2011 to June 30, 2015. Data collected included age and common life-limiting comorbidities at the time of referral. Morbidities were found on the electronic health record, including coronary artery disease (CAD), congestive heart failure (CHF), cerebral vascular disease (CVD), peripheral vascular disease, dementia, chronic kidney disease (CKD), chronic obstructive pulmonary disease (COPD), tobacco use, diabetes mellitus (DM), liver disease, alcohol use, and obstructive sleep apnea.
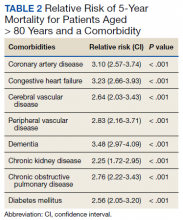
Treatment, complications, and 5-year mortality were recorded. A χ2 analysis with P value < .05 was used to determine statistical significance between individual risk factors and 5-year mortality. The relative risk of 5-year mortality was calculated by combining advanced age (aged > 80 years) with the individual comorbidities.
Results
Over 4 years, 800 consults for NMSC were completed by the plastic surgery service. Treatment decisions included 210 excisions (with or without reconstruction) in the operating room, 402 excisions (with or without reconstruction) under local anesthesia in clinic, 55 Mohs surgical dermatology referrals, 21 other service or hospital referrals, and 112 patient who were observed, declined intervention, or died prior to intervention. Five-year mortality was 28.6%. No patients died of NMSC. The median age at consult submission for patients deceased 5 years later was 78 years. Complication rate was 5% and included wound infection, dehiscence, bleeding, or graft loss. Two patients, both deceased within 5 years, had unplanned admissions due to bleeding from either a skin graft donor site or recipient bleeding. Aged ≥ 80 years, CAD, CHF, CVD, peripheral vascular disease, dementia, CKD, COPD, and DM were all found individually to be statistically significant predictors of 5-year mortality (Table 1). Combining aged ≥ 80 years plus CAD, CHF, or dementia all increased the 5-year mortality by a relative risk of > 3 (Table 2).
Discussion
The standard of care is to treat NMSC. Most NMSCs are treated surgically without consideration of patient age or life expectancy.2,4,9,10 A prospective cohort study involving a university-based private practice and a VA medical center in San Francisco found a 22.6% overall 5-year mortality and a 43.3% mortality in the group defined as limited life expectancy (LLE) based on age (≥ 85 years) and medical comorbidities. None died due to the NMSC. Leading cause of death was cardiac, cerebrovascular, and respiratory disease, lung and prostate cancer, and Alzheimer disease. The authors suggested the LLE group may be exposed to wound complications without benefiting from the treatment.4
Another study of 440 patients receiving excision for biopsy-proven facial NMSC at the Roudebush VA Medical Center in Indianapolis, Indiana, found no residual carcinoma in 35.3% of excisions, and in patients aged > 90 years, more than half of the excisions had no residual carcinoma. More than half of the patients aged > 90 years died within 1 year, not as a result of the NMSC. The authors argued for watchful waiting in select patients to maximize comfort and outcomes.10
NMSCs are often asymptomatic and not immediately life threatening. Although NMSCs tend to have a favorable prognosis, studies have found that NMSC may be a marker for other poor health outcomes. A significant increased risk for all-cause mortality was found for patients with a history of SCC, which may be attributed to immune status.11 The aging veteran population has more complex health care needs to be considered when developing surgical treatment plans. These medical problems may limit their life expectancy much sooner than the skin cancer will become symptomatic. We found that individuals aged ≥ 80 years who had CAD, CHF, or dementia had a relative risk of 3 or higher for 5-year mortality. The leading cause of death in the United States in years 2011 to 2015 was heart disease. Alzheimer disease was the sixth leading cause of death in those same years.12-14
Skin cancer excisions do not typically require general anesthesia, deep sedation, or large fluid shifts; however, studies have found that when frail patients undergo low-risk procedures, they tend to have a higher mortality rate than their healthier counterparts.15 Frailty is a concept that identifies patients who are at increased risk of dying in 6 to 60 months due to a decline in their physical reserve. Frail patients have increased rates of perioperative mortality and complications. Various tools have been used to assess the components of physical performance, speed, mobility, nutrition status, mental health, and cognition.16 Frailty screening has been initiated in several VA hospitals, including our own in Gainesville, Florida, with the goal of decreasing postoperative morbidity and mortality in older adult patients.17 The patients are given a 1-page screening assessment that asks about their living situation, medical conditions, nutrition status, cognition, and activities of daily living. The results can trigger the clinician to rethink the surgical plan and mobilize more resources to optimize the patient’s health. This study period precedes the initiative at our institution.
The plastic surgery service’s routine practice is to excise skin cancers in the operating room if sedation or general anesthesia will be needed (Figure 1A), for optimal control of bleeding (Figure 1B) in a patient who cannot safely stop blood thinners, or for excision of a highly vascularized area such as the scalp. Surgery is offered in an office-based setting if the area can be closed primarily, left open to close secondarily, or closed with a small skin graft under local anesthesia only (Figure 2). We prefer treating frail patients in the minor procedure clinic, when possible, to avoid the risks of sedation and the additional preoperative visits and transportation requirements. NMSC with unclear margins (Figure 3A) or in cosmetically sensitive areas where tissue needs to be preserved (Figure 3B) are referred to the Mohs dermatologist. The skin cancers in this study were most frequently found on the face, scalp, hands, and forearms based on referral patterns.
Other treatment options for NMSC include curettage and electrodessication, cryotherapy, and radiation; however, ours is a surgical service and patients are typically referred to us by primary care or dermatology when those are not reasonable or desirable options.18 Published complication rates of patients having skin cancer surgery without age restriction have a rate of 3% to 6%, which is consistent with our study of 5%.19-21 Two bleeding complications that needed to be admitted did not require more than a bedside procedure and neither required transfusions. One patient had been instructed to continue taking coumadin during the perioperative office-based procedure due to a recent carotid stent placement in the setting of a rapidly growing basal cell on an easily accessible location.
The most noted comorbidity in patients with wound complications was found to be DM; however, this was not found to be a statistically significant risk factor for wound complications (P = .10). We do not have a set rule for advising for or against NMSC surgery. We do counsel frail patients and their families that not all cancer is immediately life threatening and will work with them to do whatever makes the most sense to achieve their goals, occasionally accepting positive margins in order to debulk a symptomatic growth. The objective of this paper is to contribute to the discussion of performing invasive procedures on older adult veterans with life-limiting comorbidities. Patients and their families will have different thresholds for what they feel needs intervention, especially if other medical problems are consuming much of their time. We also have the community care referral option for patients whose treatment decisions are being dictated by travel hardships.
Strengths and Limitations
A strength of this study is that the data were obtained from a closed system. Patients tend to stay long-term within the VA and their health record is accessible throughout the country as long as they are seen at a VA facility. Complications, therefore, return to the treating service or primary care, who would route the patient back to the surgeon.
One limitation of the study is that this is a retrospective review from 2011. The authors are limited to data that are recorded in the patient record. Multiple health care professionals saw the patients and notes lack consistency in detail. Size of the lesions were not consistently recorded and did not get logged into our database for that reason.
Conclusions
Treatment of NMSC in older adult patients has a low morbidity but needs to be balanced against a patient and family’s goals when the patient presents with life-limiting comorbidities. An elevated 5-year mortality in patients aged > 80 years with serious unrelated medical conditions is intuitive, but this study may help put treatment plans into perspective for families and health care professionals who want to provide an indicated service while maximizing patient quality of life.
Acknowledgments
This manuscript is the result of work supported with resources and the use of facilities at the North Florida/South Georgia Veterans Health System, Gainesville, Florida.
1. American Cancer Society. Cancer Facts & Figures 2021. Accessed May 26, 2022. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2021/cancer-facts-and-figures-2021.pdf
2. Albert A, Knoll MA, Conti JA, Zbar RIS. Non-melanoma skin cancers in the older patient. Curr Oncol Rep. 2019;21(9):79. Published 2019 Jul 29. doi:10.1007/s11912-019-0828-9
3. Linos E, Chren MM, Stijacic Cenzer I, Covinsky KE. Skin cancer in U.S. elderly adults: does life expectancy play a role in treatment decisions? J Am Geriatr Soc. 2016;64(8):1610-1615. doi:10.1111/jgs.14202
4. Linos E, Parvataneni R, Stuart SE, Boscardin WJ, Landefeld CS, Chren MM. Treatment of nonfatal conditions at the end of life: nonmelanoma skin cancer. JAMA Intern Med. 2013;173(11):1006-1012. doi:10.1001/jamainternmed.2013.639
5. O’Malley KA, Vinson L, Kaiser AP, Sager Z, Hinrichs K. Mental health and aging veterans: how the Veterans Health Administration meets the needs of aging veterans. Public Policy Aging Rep. 2020;30(1):19-23. doi:10.1093/ppar/prz027
6. US Department of Veterans Affairs, National Center for Veterans Analysis and Statistics. Profile of veterans: 2017. Accessed May 26, 2022. https://www.va.gov/vetdata/docs/SpecialReports/Profile_of_Veterans_2017.pdf 7. Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78(6):1185-1192. doi:10.1016/j.jaad.2017.11.062
8. Clemens MW, Kochuba AL, Carter ME, Han K, Liu J, Evans K. Association between Agent Orange exposure and nonmelanotic invasive skin cancer: a pilot study. Plast Reconstr Surg. 2014;133(2):432-437. doi:10.1097/01.prs.0000436859.40151.cf
9. Cameron MC, Lee E, Hibler BP, et al. Basal cell carcinoma: epidemiology; pathophysiology; clinical and histological subtypes; and disease associations. J Am Acad Dermatol. 2019;80(2):303-317. doi:10.1016/j.jaad.2018.03.060
10. Chauhan R, Munger BN, Chu MW, et al. Age at diagnosis as a relative contraindication for intervention in facial nonmelanoma skin cancer. JAMA Surg. 2018;153(4):390-392. doi:10.1001/jamasurg.2017.5073
11. Barton V, Armeson K, Hampras S, et al. Nonmelanoma skin cancer and risk of all-cause and cancer-related mortality: a systematic review. Arch Dermatol Res. 2017;309(4):243-251. doi:10.1007/s00403-017-1724-5
12. Kochanek KD, Murphy SL, Xu JQ, Arias E. Mortality in the United States, 2013. NCHS Data Brief 178. Accessed May 26, 2022. https://www.cdc.gov/nchs/products/databriefs/db178.htm
13. Xu JQ, Kochanek KD, Murphy SL, Arias E. Mortality in the United States, 2012. NCHS Data Brief 168. Accessed May 26, 2022. https://www.cdc.gov/nchs/products/databriefs/db168.htm
14. Xu JQ, Murphy SL, Kochanek KD, Arias E. Mortality in the United States, 2015. NCHS Data Brief 267. Accessed May 26, 2022. https://www.cdc.gov/nchs/products/databriefs/db267.htm
15. Varley PR , Borrebach JD, Arya S, et al. Clinical utility of the risk analysis index as a prospective frailty screening tool within a multi-practice, multi-hospital integrated healthcare system. Ann Surg. 2021;274(6):e1230-e1237. doi:10.1097/SLA.0000000000003808
16. Hall DE, Arya S , Schmid KK, et al. Development and initial validation of the risk analysis index for measuring frailty in surgical populations. JAMA Surg. 2017;152(2):175-182. doi:10.1001/jamasurg.2016.4202
17. US Department of Veterans Affairs, Health Services Research & Development. Improving healthcare for aging veterans. Updated August 30, 2017. Accessed May 26, 2022. https://www.hsrd.research.va.gov/news/feature/aging0917.cfm
18. Leus AJG, Frie M, Haisma MS, et al. Treatment of keratinocyte carcinoma in elderly patients – a review of the current literature. J Eur Acad Dermatol Venereol. 2020;34(9):1932-1943. doi:10.1111/jdv.16268
19. Amici JM, Rogues AM, Lasheras A, et al. A prospective study of the incidence of complications associated with dermatological surgery. Br J Dermatol. 2005;153(5):967-971. doi:10.1111/j.1365-2133.2005.06861.x
20. Arguello-Guerra L, Vargas-Chandomid E, Díaz-González JM, Méndez-Flores S, Ruelas-Villavicencio A, Domínguez-Cherit J. Incidence of complications in dermatological surgery of melanoma and non-melanoma skin cancer in patients with multiple comorbidity and/or antiplatelet-anticoagulants. Five-year experience in our hospital. Cir Cir. 2019;86(1):15-23. doi:10.24875/CIRUE.M18000003
21. Keith DJ, de Berker DA, Bray AP, Cheung ST, Brain A, Mohd Mustapa MF. British Association of Dermatologists’ national audit on nonmelanoma skin cancer excision, 2014. Clin Exp Dermatol. 2017;42(1):46-53. doi:10.1111/ced.12990
Skin cancer is the most diagnosed cancer in the United States. Nonmelanoma skin cancers (NMSC), which include basal cell carcinoma and squamous cell carcinoma, are usually cured with removal.1 The incidence of NMSC increases with age and is commonly found in nursing homes and geriatric units. These cancers are not usually metastatic or fatal but can cause local destruction and disfigurement if neglected.2 The current standard of care is to treat diagnosed NMSC; however, the dermatology and geriatric care literature have questioned the logic of treating asymptomatic skin cancers that will not affect a patient’s life expectancy.2-4
Forty-seven percent of the current living veteran population is aged ≥ 65 years.5 Older adult patients are frequently referred to the US Department of Veterans Affairs (VA) surgical service for the treatment of NMSC. The veteran population includes a higher percentage of individuals at an elevated risk of skin cancers (older, White, and male) compared with the general population.6 World War II veterans deployed in regions closer to the equator have been found to have an elevated risk of melanoma and nonmelanoma skin carcinomas.7 A retrospective study of Vietnam veterans exposed to Agent Orange (2,3,7,8-tetrachlorodibenzodioxin) found a significantly higher risk of invasive NMSC in Fitzpatrick skin types I-IV compared with an age-matched subset of the general population.8 Younger veterans who were deployed in Afghanistan and Iraq for Operation Enduring Freedom/Operation Iraqi Freedom worked at more equatorial latitudes than the rest of the US population and may be at increased risk of NMSC. Inadequate sunscreen access, immediate safety concerns, outdoor recreational activities, harsh weather, and insufficient emphasis on sun protection have created a multifactorial challenge for the military population. Riemenschneider and colleagues recommended targeted screening for at-risk veteran patients and prioritizing annual skin cancer screenings during medical mission physical examinations for active military.7
The plastic surgery service regularly receives consults from dermatology, general surgery, and primary care to remove skin cancers on the face, scalp, hands, and forearms. Skin cancer treatment can create serious hardships for older adult patients and their families with multiple appointments for the consult, procedure, and follow-up. Patients are often told to hold their anticoagulant medications when the surgery will be performed on a highly vascular region, such as the scalp or face. This can create wide swings in their laboratory test values and result in life-threatening complications from either bleeding or clotting. The appropriateness of offering surgery to patients with serious comorbidities and a limited life expectancy has been questioned.2-4 The purpose of this study was to measure the morbidity and unrelated 5-year mortality for patients with skin cancer referred to the plastic surgery service to help patients and families make a more informed treatment decision, particularly when the patients are aged > 80 years and have significant life-threatening comorbidities.
Methods
The University of Florida and Malcom Randall VA Medical Center Institutional review board in Gainesville, approved a retrospective review of all consults completed by the plastic surgery service for the treatment of NMSC performed from July 1, 2011 to June 30, 2015. Data collected included age and common life-limiting comorbidities at the time of referral. Morbidities were found on the electronic health record, including coronary artery disease (CAD), congestive heart failure (CHF), cerebral vascular disease (CVD), peripheral vascular disease, dementia, chronic kidney disease (CKD), chronic obstructive pulmonary disease (COPD), tobacco use, diabetes mellitus (DM), liver disease, alcohol use, and obstructive sleep apnea.
Treatment, complications, and 5-year mortality were recorded. A χ2 analysis with P value < .05 was used to determine statistical significance between individual risk factors and 5-year mortality. The relative risk of 5-year mortality was calculated by combining advanced age (aged > 80 years) with the individual comorbidities.
Results
Over 4 years, 800 consults for NMSC were completed by the plastic surgery service. Treatment decisions included 210 excisions (with or without reconstruction) in the operating room, 402 excisions (with or without reconstruction) under local anesthesia in clinic, 55 Mohs surgical dermatology referrals, 21 other service or hospital referrals, and 112 patient who were observed, declined intervention, or died prior to intervention. Five-year mortality was 28.6%. No patients died of NMSC. The median age at consult submission for patients deceased 5 years later was 78 years. Complication rate was 5% and included wound infection, dehiscence, bleeding, or graft loss. Two patients, both deceased within 5 years, had unplanned admissions due to bleeding from either a skin graft donor site or recipient bleeding. Aged ≥ 80 years, CAD, CHF, CVD, peripheral vascular disease, dementia, CKD, COPD, and DM were all found individually to be statistically significant predictors of 5-year mortality (Table 1). Combining aged ≥ 80 years plus CAD, CHF, or dementia all increased the 5-year mortality by a relative risk of > 3 (Table 2).
Discussion
The standard of care is to treat NMSC. Most NMSCs are treated surgically without consideration of patient age or life expectancy.2,4,9,10 A prospective cohort study involving a university-based private practice and a VA medical center in San Francisco found a 22.6% overall 5-year mortality and a 43.3% mortality in the group defined as limited life expectancy (LLE) based on age (≥ 85 years) and medical comorbidities. None died due to the NMSC. Leading cause of death was cardiac, cerebrovascular, and respiratory disease, lung and prostate cancer, and Alzheimer disease. The authors suggested the LLE group may be exposed to wound complications without benefiting from the treatment.4
Another study of 440 patients receiving excision for biopsy-proven facial NMSC at the Roudebush VA Medical Center in Indianapolis, Indiana, found no residual carcinoma in 35.3% of excisions, and in patients aged > 90 years, more than half of the excisions had no residual carcinoma. More than half of the patients aged > 90 years died within 1 year, not as a result of the NMSC. The authors argued for watchful waiting in select patients to maximize comfort and outcomes.10
NMSCs are often asymptomatic and not immediately life threatening. Although NMSCs tend to have a favorable prognosis, studies have found that NMSC may be a marker for other poor health outcomes. A significant increased risk for all-cause mortality was found for patients with a history of SCC, which may be attributed to immune status.11 The aging veteran population has more complex health care needs to be considered when developing surgical treatment plans. These medical problems may limit their life expectancy much sooner than the skin cancer will become symptomatic. We found that individuals aged ≥ 80 years who had CAD, CHF, or dementia had a relative risk of 3 or higher for 5-year mortality. The leading cause of death in the United States in years 2011 to 2015 was heart disease. Alzheimer disease was the sixth leading cause of death in those same years.12-14
Skin cancer excisions do not typically require general anesthesia, deep sedation, or large fluid shifts; however, studies have found that when frail patients undergo low-risk procedures, they tend to have a higher mortality rate than their healthier counterparts.15 Frailty is a concept that identifies patients who are at increased risk of dying in 6 to 60 months due to a decline in their physical reserve. Frail patients have increased rates of perioperative mortality and complications. Various tools have been used to assess the components of physical performance, speed, mobility, nutrition status, mental health, and cognition.16 Frailty screening has been initiated in several VA hospitals, including our own in Gainesville, Florida, with the goal of decreasing postoperative morbidity and mortality in older adult patients.17 The patients are given a 1-page screening assessment that asks about their living situation, medical conditions, nutrition status, cognition, and activities of daily living. The results can trigger the clinician to rethink the surgical plan and mobilize more resources to optimize the patient’s health. This study period precedes the initiative at our institution.
The plastic surgery service’s routine practice is to excise skin cancers in the operating room if sedation or general anesthesia will be needed (Figure 1A), for optimal control of bleeding (Figure 1B) in a patient who cannot safely stop blood thinners, or for excision of a highly vascularized area such as the scalp. Surgery is offered in an office-based setting if the area can be closed primarily, left open to close secondarily, or closed with a small skin graft under local anesthesia only (Figure 2). We prefer treating frail patients in the minor procedure clinic, when possible, to avoid the risks of sedation and the additional preoperative visits and transportation requirements. NMSC with unclear margins (Figure 3A) or in cosmetically sensitive areas where tissue needs to be preserved (Figure 3B) are referred to the Mohs dermatologist. The skin cancers in this study were most frequently found on the face, scalp, hands, and forearms based on referral patterns.
Other treatment options for NMSC include curettage and electrodessication, cryotherapy, and radiation; however, ours is a surgical service and patients are typically referred to us by primary care or dermatology when those are not reasonable or desirable options.18 Published complication rates of patients having skin cancer surgery without age restriction have a rate of 3% to 6%, which is consistent with our study of 5%.19-21 Two bleeding complications that needed to be admitted did not require more than a bedside procedure and neither required transfusions. One patient had been instructed to continue taking coumadin during the perioperative office-based procedure due to a recent carotid stent placement in the setting of a rapidly growing basal cell on an easily accessible location.
The most noted comorbidity in patients with wound complications was found to be DM; however, this was not found to be a statistically significant risk factor for wound complications (P = .10). We do not have a set rule for advising for or against NMSC surgery. We do counsel frail patients and their families that not all cancer is immediately life threatening and will work with them to do whatever makes the most sense to achieve their goals, occasionally accepting positive margins in order to debulk a symptomatic growth. The objective of this paper is to contribute to the discussion of performing invasive procedures on older adult veterans with life-limiting comorbidities. Patients and their families will have different thresholds for what they feel needs intervention, especially if other medical problems are consuming much of their time. We also have the community care referral option for patients whose treatment decisions are being dictated by travel hardships.
Strengths and Limitations
A strength of this study is that the data were obtained from a closed system. Patients tend to stay long-term within the VA and their health record is accessible throughout the country as long as they are seen at a VA facility. Complications, therefore, return to the treating service or primary care, who would route the patient back to the surgeon.
One limitation of the study is that this is a retrospective review from 2011. The authors are limited to data that are recorded in the patient record. Multiple health care professionals saw the patients and notes lack consistency in detail. Size of the lesions were not consistently recorded and did not get logged into our database for that reason.
Conclusions
Treatment of NMSC in older adult patients has a low morbidity but needs to be balanced against a patient and family’s goals when the patient presents with life-limiting comorbidities. An elevated 5-year mortality in patients aged > 80 years with serious unrelated medical conditions is intuitive, but this study may help put treatment plans into perspective for families and health care professionals who want to provide an indicated service while maximizing patient quality of life.
Acknowledgments
This manuscript is the result of work supported with resources and the use of facilities at the North Florida/South Georgia Veterans Health System, Gainesville, Florida.
Skin cancer is the most diagnosed cancer in the United States. Nonmelanoma skin cancers (NMSC), which include basal cell carcinoma and squamous cell carcinoma, are usually cured with removal.1 The incidence of NMSC increases with age and is commonly found in nursing homes and geriatric units. These cancers are not usually metastatic or fatal but can cause local destruction and disfigurement if neglected.2 The current standard of care is to treat diagnosed NMSC; however, the dermatology and geriatric care literature have questioned the logic of treating asymptomatic skin cancers that will not affect a patient’s life expectancy.2-4
Forty-seven percent of the current living veteran population is aged ≥ 65 years.5 Older adult patients are frequently referred to the US Department of Veterans Affairs (VA) surgical service for the treatment of NMSC. The veteran population includes a higher percentage of individuals at an elevated risk of skin cancers (older, White, and male) compared with the general population.6 World War II veterans deployed in regions closer to the equator have been found to have an elevated risk of melanoma and nonmelanoma skin carcinomas.7 A retrospective study of Vietnam veterans exposed to Agent Orange (2,3,7,8-tetrachlorodibenzodioxin) found a significantly higher risk of invasive NMSC in Fitzpatrick skin types I-IV compared with an age-matched subset of the general population.8 Younger veterans who were deployed in Afghanistan and Iraq for Operation Enduring Freedom/Operation Iraqi Freedom worked at more equatorial latitudes than the rest of the US population and may be at increased risk of NMSC. Inadequate sunscreen access, immediate safety concerns, outdoor recreational activities, harsh weather, and insufficient emphasis on sun protection have created a multifactorial challenge for the military population. Riemenschneider and colleagues recommended targeted screening for at-risk veteran patients and prioritizing annual skin cancer screenings during medical mission physical examinations for active military.7
The plastic surgery service regularly receives consults from dermatology, general surgery, and primary care to remove skin cancers on the face, scalp, hands, and forearms. Skin cancer treatment can create serious hardships for older adult patients and their families with multiple appointments for the consult, procedure, and follow-up. Patients are often told to hold their anticoagulant medications when the surgery will be performed on a highly vascular region, such as the scalp or face. This can create wide swings in their laboratory test values and result in life-threatening complications from either bleeding or clotting. The appropriateness of offering surgery to patients with serious comorbidities and a limited life expectancy has been questioned.2-4 The purpose of this study was to measure the morbidity and unrelated 5-year mortality for patients with skin cancer referred to the plastic surgery service to help patients and families make a more informed treatment decision, particularly when the patients are aged > 80 years and have significant life-threatening comorbidities.
Methods
The University of Florida and Malcom Randall VA Medical Center Institutional review board in Gainesville, approved a retrospective review of all consults completed by the plastic surgery service for the treatment of NMSC performed from July 1, 2011 to June 30, 2015. Data collected included age and common life-limiting comorbidities at the time of referral. Morbidities were found on the electronic health record, including coronary artery disease (CAD), congestive heart failure (CHF), cerebral vascular disease (CVD), peripheral vascular disease, dementia, chronic kidney disease (CKD), chronic obstructive pulmonary disease (COPD), tobacco use, diabetes mellitus (DM), liver disease, alcohol use, and obstructive sleep apnea.
Treatment, complications, and 5-year mortality were recorded. A χ2 analysis with P value < .05 was used to determine statistical significance between individual risk factors and 5-year mortality. The relative risk of 5-year mortality was calculated by combining advanced age (aged > 80 years) with the individual comorbidities.
Results
Over 4 years, 800 consults for NMSC were completed by the plastic surgery service. Treatment decisions included 210 excisions (with or without reconstruction) in the operating room, 402 excisions (with or without reconstruction) under local anesthesia in clinic, 55 Mohs surgical dermatology referrals, 21 other service or hospital referrals, and 112 patient who were observed, declined intervention, or died prior to intervention. Five-year mortality was 28.6%. No patients died of NMSC. The median age at consult submission for patients deceased 5 years later was 78 years. Complication rate was 5% and included wound infection, dehiscence, bleeding, or graft loss. Two patients, both deceased within 5 years, had unplanned admissions due to bleeding from either a skin graft donor site or recipient bleeding. Aged ≥ 80 years, CAD, CHF, CVD, peripheral vascular disease, dementia, CKD, COPD, and DM were all found individually to be statistically significant predictors of 5-year mortality (Table 1). Combining aged ≥ 80 years plus CAD, CHF, or dementia all increased the 5-year mortality by a relative risk of > 3 (Table 2).
Discussion
The standard of care is to treat NMSC. Most NMSCs are treated surgically without consideration of patient age or life expectancy.2,4,9,10 A prospective cohort study involving a university-based private practice and a VA medical center in San Francisco found a 22.6% overall 5-year mortality and a 43.3% mortality in the group defined as limited life expectancy (LLE) based on age (≥ 85 years) and medical comorbidities. None died due to the NMSC. Leading cause of death was cardiac, cerebrovascular, and respiratory disease, lung and prostate cancer, and Alzheimer disease. The authors suggested the LLE group may be exposed to wound complications without benefiting from the treatment.4
Another study of 440 patients receiving excision for biopsy-proven facial NMSC at the Roudebush VA Medical Center in Indianapolis, Indiana, found no residual carcinoma in 35.3% of excisions, and in patients aged > 90 years, more than half of the excisions had no residual carcinoma. More than half of the patients aged > 90 years died within 1 year, not as a result of the NMSC. The authors argued for watchful waiting in select patients to maximize comfort and outcomes.10
NMSCs are often asymptomatic and not immediately life threatening. Although NMSCs tend to have a favorable prognosis, studies have found that NMSC may be a marker for other poor health outcomes. A significant increased risk for all-cause mortality was found for patients with a history of SCC, which may be attributed to immune status.11 The aging veteran population has more complex health care needs to be considered when developing surgical treatment plans. These medical problems may limit their life expectancy much sooner than the skin cancer will become symptomatic. We found that individuals aged ≥ 80 years who had CAD, CHF, or dementia had a relative risk of 3 or higher for 5-year mortality. The leading cause of death in the United States in years 2011 to 2015 was heart disease. Alzheimer disease was the sixth leading cause of death in those same years.12-14
Skin cancer excisions do not typically require general anesthesia, deep sedation, or large fluid shifts; however, studies have found that when frail patients undergo low-risk procedures, they tend to have a higher mortality rate than their healthier counterparts.15 Frailty is a concept that identifies patients who are at increased risk of dying in 6 to 60 months due to a decline in their physical reserve. Frail patients have increased rates of perioperative mortality and complications. Various tools have been used to assess the components of physical performance, speed, mobility, nutrition status, mental health, and cognition.16 Frailty screening has been initiated in several VA hospitals, including our own in Gainesville, Florida, with the goal of decreasing postoperative morbidity and mortality in older adult patients.17 The patients are given a 1-page screening assessment that asks about their living situation, medical conditions, nutrition status, cognition, and activities of daily living. The results can trigger the clinician to rethink the surgical plan and mobilize more resources to optimize the patient’s health. This study period precedes the initiative at our institution.
The plastic surgery service’s routine practice is to excise skin cancers in the operating room if sedation or general anesthesia will be needed (Figure 1A), for optimal control of bleeding (Figure 1B) in a patient who cannot safely stop blood thinners, or for excision of a highly vascularized area such as the scalp. Surgery is offered in an office-based setting if the area can be closed primarily, left open to close secondarily, or closed with a small skin graft under local anesthesia only (Figure 2). We prefer treating frail patients in the minor procedure clinic, when possible, to avoid the risks of sedation and the additional preoperative visits and transportation requirements. NMSC with unclear margins (Figure 3A) or in cosmetically sensitive areas where tissue needs to be preserved (Figure 3B) are referred to the Mohs dermatologist. The skin cancers in this study were most frequently found on the face, scalp, hands, and forearms based on referral patterns.
Other treatment options for NMSC include curettage and electrodessication, cryotherapy, and radiation; however, ours is a surgical service and patients are typically referred to us by primary care or dermatology when those are not reasonable or desirable options.18 Published complication rates of patients having skin cancer surgery without age restriction have a rate of 3% to 6%, which is consistent with our study of 5%.19-21 Two bleeding complications that needed to be admitted did not require more than a bedside procedure and neither required transfusions. One patient had been instructed to continue taking coumadin during the perioperative office-based procedure due to a recent carotid stent placement in the setting of a rapidly growing basal cell on an easily accessible location.
The most noted comorbidity in patients with wound complications was found to be DM; however, this was not found to be a statistically significant risk factor for wound complications (P = .10). We do not have a set rule for advising for or against NMSC surgery. We do counsel frail patients and their families that not all cancer is immediately life threatening and will work with them to do whatever makes the most sense to achieve their goals, occasionally accepting positive margins in order to debulk a symptomatic growth. The objective of this paper is to contribute to the discussion of performing invasive procedures on older adult veterans with life-limiting comorbidities. Patients and their families will have different thresholds for what they feel needs intervention, especially if other medical problems are consuming much of their time. We also have the community care referral option for patients whose treatment decisions are being dictated by travel hardships.
Strengths and Limitations
A strength of this study is that the data were obtained from a closed system. Patients tend to stay long-term within the VA and their health record is accessible throughout the country as long as they are seen at a VA facility. Complications, therefore, return to the treating service or primary care, who would route the patient back to the surgeon.
One limitation of the study is that this is a retrospective review from 2011. The authors are limited to data that are recorded in the patient record. Multiple health care professionals saw the patients and notes lack consistency in detail. Size of the lesions were not consistently recorded and did not get logged into our database for that reason.
Conclusions
Treatment of NMSC in older adult patients has a low morbidity but needs to be balanced against a patient and family’s goals when the patient presents with life-limiting comorbidities. An elevated 5-year mortality in patients aged > 80 years with serious unrelated medical conditions is intuitive, but this study may help put treatment plans into perspective for families and health care professionals who want to provide an indicated service while maximizing patient quality of life.
Acknowledgments
This manuscript is the result of work supported with resources and the use of facilities at the North Florida/South Georgia Veterans Health System, Gainesville, Florida.
1. American Cancer Society. Cancer Facts & Figures 2021. Accessed May 26, 2022. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2021/cancer-facts-and-figures-2021.pdf
2. Albert A, Knoll MA, Conti JA, Zbar RIS. Non-melanoma skin cancers in the older patient. Curr Oncol Rep. 2019;21(9):79. Published 2019 Jul 29. doi:10.1007/s11912-019-0828-9
3. Linos E, Chren MM, Stijacic Cenzer I, Covinsky KE. Skin cancer in U.S. elderly adults: does life expectancy play a role in treatment decisions? J Am Geriatr Soc. 2016;64(8):1610-1615. doi:10.1111/jgs.14202
4. Linos E, Parvataneni R, Stuart SE, Boscardin WJ, Landefeld CS, Chren MM. Treatment of nonfatal conditions at the end of life: nonmelanoma skin cancer. JAMA Intern Med. 2013;173(11):1006-1012. doi:10.1001/jamainternmed.2013.639
5. O’Malley KA, Vinson L, Kaiser AP, Sager Z, Hinrichs K. Mental health and aging veterans: how the Veterans Health Administration meets the needs of aging veterans. Public Policy Aging Rep. 2020;30(1):19-23. doi:10.1093/ppar/prz027
6. US Department of Veterans Affairs, National Center for Veterans Analysis and Statistics. Profile of veterans: 2017. Accessed May 26, 2022. https://www.va.gov/vetdata/docs/SpecialReports/Profile_of_Veterans_2017.pdf 7. Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78(6):1185-1192. doi:10.1016/j.jaad.2017.11.062
8. Clemens MW, Kochuba AL, Carter ME, Han K, Liu J, Evans K. Association between Agent Orange exposure and nonmelanotic invasive skin cancer: a pilot study. Plast Reconstr Surg. 2014;133(2):432-437. doi:10.1097/01.prs.0000436859.40151.cf
9. Cameron MC, Lee E, Hibler BP, et al. Basal cell carcinoma: epidemiology; pathophysiology; clinical and histological subtypes; and disease associations. J Am Acad Dermatol. 2019;80(2):303-317. doi:10.1016/j.jaad.2018.03.060
10. Chauhan R, Munger BN, Chu MW, et al. Age at diagnosis as a relative contraindication for intervention in facial nonmelanoma skin cancer. JAMA Surg. 2018;153(4):390-392. doi:10.1001/jamasurg.2017.5073
11. Barton V, Armeson K, Hampras S, et al. Nonmelanoma skin cancer and risk of all-cause and cancer-related mortality: a systematic review. Arch Dermatol Res. 2017;309(4):243-251. doi:10.1007/s00403-017-1724-5
12. Kochanek KD, Murphy SL, Xu JQ, Arias E. Mortality in the United States, 2013. NCHS Data Brief 178. Accessed May 26, 2022. https://www.cdc.gov/nchs/products/databriefs/db178.htm
13. Xu JQ, Kochanek KD, Murphy SL, Arias E. Mortality in the United States, 2012. NCHS Data Brief 168. Accessed May 26, 2022. https://www.cdc.gov/nchs/products/databriefs/db168.htm
14. Xu JQ, Murphy SL, Kochanek KD, Arias E. Mortality in the United States, 2015. NCHS Data Brief 267. Accessed May 26, 2022. https://www.cdc.gov/nchs/products/databriefs/db267.htm
15. Varley PR , Borrebach JD, Arya S, et al. Clinical utility of the risk analysis index as a prospective frailty screening tool within a multi-practice, multi-hospital integrated healthcare system. Ann Surg. 2021;274(6):e1230-e1237. doi:10.1097/SLA.0000000000003808
16. Hall DE, Arya S , Schmid KK, et al. Development and initial validation of the risk analysis index for measuring frailty in surgical populations. JAMA Surg. 2017;152(2):175-182. doi:10.1001/jamasurg.2016.4202
17. US Department of Veterans Affairs, Health Services Research & Development. Improving healthcare for aging veterans. Updated August 30, 2017. Accessed May 26, 2022. https://www.hsrd.research.va.gov/news/feature/aging0917.cfm
18. Leus AJG, Frie M, Haisma MS, et al. Treatment of keratinocyte carcinoma in elderly patients – a review of the current literature. J Eur Acad Dermatol Venereol. 2020;34(9):1932-1943. doi:10.1111/jdv.16268
19. Amici JM, Rogues AM, Lasheras A, et al. A prospective study of the incidence of complications associated with dermatological surgery. Br J Dermatol. 2005;153(5):967-971. doi:10.1111/j.1365-2133.2005.06861.x
20. Arguello-Guerra L, Vargas-Chandomid E, Díaz-González JM, Méndez-Flores S, Ruelas-Villavicencio A, Domínguez-Cherit J. Incidence of complications in dermatological surgery of melanoma and non-melanoma skin cancer in patients with multiple comorbidity and/or antiplatelet-anticoagulants. Five-year experience in our hospital. Cir Cir. 2019;86(1):15-23. doi:10.24875/CIRUE.M18000003
21. Keith DJ, de Berker DA, Bray AP, Cheung ST, Brain A, Mohd Mustapa MF. British Association of Dermatologists’ national audit on nonmelanoma skin cancer excision, 2014. Clin Exp Dermatol. 2017;42(1):46-53. doi:10.1111/ced.12990
1. American Cancer Society. Cancer Facts & Figures 2021. Accessed May 26, 2022. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2021/cancer-facts-and-figures-2021.pdf
2. Albert A, Knoll MA, Conti JA, Zbar RIS. Non-melanoma skin cancers in the older patient. Curr Oncol Rep. 2019;21(9):79. Published 2019 Jul 29. doi:10.1007/s11912-019-0828-9
3. Linos E, Chren MM, Stijacic Cenzer I, Covinsky KE. Skin cancer in U.S. elderly adults: does life expectancy play a role in treatment decisions? J Am Geriatr Soc. 2016;64(8):1610-1615. doi:10.1111/jgs.14202
4. Linos E, Parvataneni R, Stuart SE, Boscardin WJ, Landefeld CS, Chren MM. Treatment of nonfatal conditions at the end of life: nonmelanoma skin cancer. JAMA Intern Med. 2013;173(11):1006-1012. doi:10.1001/jamainternmed.2013.639
5. O’Malley KA, Vinson L, Kaiser AP, Sager Z, Hinrichs K. Mental health and aging veterans: how the Veterans Health Administration meets the needs of aging veterans. Public Policy Aging Rep. 2020;30(1):19-23. doi:10.1093/ppar/prz027
6. US Department of Veterans Affairs, National Center for Veterans Analysis and Statistics. Profile of veterans: 2017. Accessed May 26, 2022. https://www.va.gov/vetdata/docs/SpecialReports/Profile_of_Veterans_2017.pdf 7. Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78(6):1185-1192. doi:10.1016/j.jaad.2017.11.062
8. Clemens MW, Kochuba AL, Carter ME, Han K, Liu J, Evans K. Association between Agent Orange exposure and nonmelanotic invasive skin cancer: a pilot study. Plast Reconstr Surg. 2014;133(2):432-437. doi:10.1097/01.prs.0000436859.40151.cf
9. Cameron MC, Lee E, Hibler BP, et al. Basal cell carcinoma: epidemiology; pathophysiology; clinical and histological subtypes; and disease associations. J Am Acad Dermatol. 2019;80(2):303-317. doi:10.1016/j.jaad.2018.03.060
10. Chauhan R, Munger BN, Chu MW, et al. Age at diagnosis as a relative contraindication for intervention in facial nonmelanoma skin cancer. JAMA Surg. 2018;153(4):390-392. doi:10.1001/jamasurg.2017.5073
11. Barton V, Armeson K, Hampras S, et al. Nonmelanoma skin cancer and risk of all-cause and cancer-related mortality: a systematic review. Arch Dermatol Res. 2017;309(4):243-251. doi:10.1007/s00403-017-1724-5
12. Kochanek KD, Murphy SL, Xu JQ, Arias E. Mortality in the United States, 2013. NCHS Data Brief 178. Accessed May 26, 2022. https://www.cdc.gov/nchs/products/databriefs/db178.htm
13. Xu JQ, Kochanek KD, Murphy SL, Arias E. Mortality in the United States, 2012. NCHS Data Brief 168. Accessed May 26, 2022. https://www.cdc.gov/nchs/products/databriefs/db168.htm
14. Xu JQ, Murphy SL, Kochanek KD, Arias E. Mortality in the United States, 2015. NCHS Data Brief 267. Accessed May 26, 2022. https://www.cdc.gov/nchs/products/databriefs/db267.htm
15. Varley PR , Borrebach JD, Arya S, et al. Clinical utility of the risk analysis index as a prospective frailty screening tool within a multi-practice, multi-hospital integrated healthcare system. Ann Surg. 2021;274(6):e1230-e1237. doi:10.1097/SLA.0000000000003808
16. Hall DE, Arya S , Schmid KK, et al. Development and initial validation of the risk analysis index for measuring frailty in surgical populations. JAMA Surg. 2017;152(2):175-182. doi:10.1001/jamasurg.2016.4202
17. US Department of Veterans Affairs, Health Services Research & Development. Improving healthcare for aging veterans. Updated August 30, 2017. Accessed May 26, 2022. https://www.hsrd.research.va.gov/news/feature/aging0917.cfm
18. Leus AJG, Frie M, Haisma MS, et al. Treatment of keratinocyte carcinoma in elderly patients – a review of the current literature. J Eur Acad Dermatol Venereol. 2020;34(9):1932-1943. doi:10.1111/jdv.16268
19. Amici JM, Rogues AM, Lasheras A, et al. A prospective study of the incidence of complications associated with dermatological surgery. Br J Dermatol. 2005;153(5):967-971. doi:10.1111/j.1365-2133.2005.06861.x
20. Arguello-Guerra L, Vargas-Chandomid E, Díaz-González JM, Méndez-Flores S, Ruelas-Villavicencio A, Domínguez-Cherit J. Incidence of complications in dermatological surgery of melanoma and non-melanoma skin cancer in patients with multiple comorbidity and/or antiplatelet-anticoagulants. Five-year experience in our hospital. Cir Cir. 2019;86(1):15-23. doi:10.24875/CIRUE.M18000003
21. Keith DJ, de Berker DA, Bray AP, Cheung ST, Brain A, Mohd Mustapa MF. British Association of Dermatologists’ national audit on nonmelanoma skin cancer excision, 2014. Clin Exp Dermatol. 2017;42(1):46-53. doi:10.1111/ced.12990
Nodular Sclerosing Hodgkin Lymphoma With Paraneoplastic Cerebellar Degeneration
Paraneoplastic syndrome is a rare disorder involving manifestations of immune dysregulation triggered by malignancy. The immune system develops antibodies to the malignancy, which can cause cross reactivation with various tissues in the body, resulting in an autoimmune response. Paraneoplastic cerebellar degeneration (PCD) is a rare condition caused by immune-mediated damage to the Purkinje cells of the cerebellar tract. Symptoms may include gait instability, double vision, decreased fine motor skills, and ataxia, with progression to brainstem-associated symptoms, such as nystagmus, dysarthria, and dysphagia. Early detection and treatment of the underlying malignancy is critical to halt the progression of autoimmune-mediated destruction. We present a case of a young adult female patient with PCD caused by Purkinje cell cytoplasmic–Tr (PCA-Tr) antibody with Hodgkin lymphoma.
Case Presentation
A 20-year-old previously healthy active-duty female patient presented to the emergency department with acute worsening of chronic intermittent, recurrent episodes of lightheadedness and vertigo. Symptoms persisted for 9 months until acutely worsening over the 2 weeks prior to presentation. She reported left eye double vision but did not report seeing spots, photophobia, tinnitus, or headache. She felt off-balance, leaning on nearby objects to remain standing. Symptoms primarily occurred during ambulation; however, occasionally they happened at rest. Episodes lasted up to several minutes and occurred up to 15 times a day. The patient reported no fever, night sweats, unexplained weight loss, muscle aches, weakness, numbness or tingling, loss of bowel or bladder function, or rash. She had no recent illnesses, changes to medications, or recent travel. Oral intake to include food and water was adequate and unchanged. The patient had a remote history of mild concussions without loss of consciousness while playing sports 4 years previously. She reported no recent trauma. Nine months before, she received treatment for benign paroxysmal positional vertigo (BPPV) with the Epley maneuver with full resolution of symptoms lasting several days. She reported no prescription or over-the-counter medications, herbal remedies, or supplements. She reported no other medical or surgical history and no pertinent social or family history.
Physical examination revealed a nontoxic-appearing female patient with intermittent conversational dysarthria, saccadic pursuits, horizontal nystagmus with lateral gaze, and vertical nystagmus with vertical gaze. The patient exhibited dysdiadochokinesia, or impaired ability to perform rapid alternating hand movements with repetition. Finger-to-nose testing was impaired and heel-to-shin motion remained intact. A Romberg test was positive, and the patient had tandem gait instability. Strength testing, sensation, reflexes, and cranial nerves were otherwise intact. Initial laboratory testing was unremarkable except for mild normocytic anemia. Her infectious workup, including testing for venereal disease, HIV, COVID-19, and Coccidioidies was negative. Heavy metals analysis and urine drug screen were negative. Ophthalmology was consulted and workup revealed small amplitude downbeat nystagmus in primary gaze, sustained gaze evoked lateral beating jerk nystagmus with rebound nystagmus R>L gaze, but there was no evidence of afferent package defect and optic nerve function remained intact. Magnetic resonance imaging of the brain demonstrated cerebellar vermis hypoplasia with prominence of the superior cerebellar folia. Due to concerns for autoimmune encephalitis, a lumbar puncture was performed. Antibody testing revealed PCA-Tr antibodies, which is commonly associated with Hodgkin lymphoma, prompting further evaluation for malignancy.
Computed tomography (CT) of the chest with contrast demonstrated multiple mediastinal masses with a conglomeration of lymph nodes along the right paratracheal region. Further evaluation was performed with a positron emission tomography (PET)–CT, revealing a large conglomeration of hypermetabolic pretracheal, mediastinal, and right supraclavicular lymph that were suggestive of lymphoma. Mediastinoscopy with excisional lymph node biopsy was performed with immunohistochemical staining confirming diagnosis of a nodular sclerosing variant of Hodgkin lymphoma. The patient was treated with IV immunoglobulin at 0.4g/kg daily for 5 days. A central venous catheter was placed into the patient’s right internal jugular vein and a chemotherapy regimen of doxorubicin 46 mg, vinblastine 11 mg, bleomycin 19 units, and dacarbazine 700 mg was initiated. The patient’s symptoms improved with resolution of dysarthria; however, her visual impairment and gait instability persisted. Repeat PET-CT imaging 2 months later revealed interval improvement with decreased intensity and extent of the hypermetabolic lymph nodes and no new hypermetabolic foci.
Discussion
PCA-Tr antibodies affect the delta/notchlike epidermal growth factor–related receptor, expressed on the dendrites of cerebellar Purkinje cells.1 These fibers are the only output neurons of the cerebellar cortex and are critical to the coordination of motor movements, accounting for the ataxia experienced by patients with this subtype of PCD.2 The link between Hodgkin lymphoma and PCA-Tr antibodies has been established; however, most reports involve men with a median age of 61 years with lymphoma-associated symptoms (such as lymphadenopathy) or systemic symptoms (fever, night sweats, or weight loss) preceding neurologic manifestations in 80% of cases.3
Our patient was a young, previously healthy adult female who initially presented with vertigo, a common concern with frequently benign origins. Although there was temporary resolution of symptoms after Epley maneuvers, symptoms recurred and progressed over several months to include brainstem manifestations of nystagmus, diplopia, and dysarthria. Previous reports indicate that after remission of the Hodgkin lymphoma, PCA-Tr antibodies disappear and symptoms can improve or resolve.4,5 Treatment has just begun for our patient and although there has been initial clinical improvement, given the chronicity of symptoms, it is unclear if complete resolution will be achieved.
Conclusions
PCD can result in debilitating neurologic dysfunction and may be associated with malignancy such as Hodgkin lymphoma. This case offers unique insight due to the patient’s demographics and presentation, which involved brainstem pathology typically associated with late-onset disease and preceded by constitutional symptoms. Clinical suspicion of this rare disorder should be considered in all ages, especially if symptoms are progressive or neurologic manifestations arise, as early detection and treatment of the underlying malignancy are paramount to the prevention of significant disability.
1. de Graaff E, Maat P, Hulsenboom E, et al. Identification of delta/notch-like epidermal growth factor-related receptor as the Tr antigen in paraneoplastic cerebellar degeneration. Ann Neurol. 2012;71(6):815-824. doi:10.1002/ana.23550
2. MacKenzie-Graham A, Tiwari-Woodruff SK, Sharma G, et al. Purkinje cell loss in experimental autoimmune encephalomyelitis. Neuroimage. 2009;48(4):637-651. doi:10.1016/j.neuroimage.2009.06.073
3. Bernal F, Shams’ili S, Rojas I, et al. Anti-Tr antibodies as markers of paraneoplastic cerebellar degeneration and Hodgkin’s disease. Neurology. 2003;60(2):230-234. doi:10.1212/01.wnl.0000041495.87539.98
4. Graus F, Ariño H, Dalmau J. Paraneoplastic neurological syndromes in Hodgkin and non-Hodgkin lymphomas. Blood. 2014;123(21):3230-3238. doi:10.1182/blood-2014-03-537506
5. Aly R, Emmady PD. Paraneoplastic cerebellar degeneration. Updated May 8, 2022. Accessed March 30, 2022. https://www.ncbi.nlm.nih.gov/books/NBK560638
Paraneoplastic syndrome is a rare disorder involving manifestations of immune dysregulation triggered by malignancy. The immune system develops antibodies to the malignancy, which can cause cross reactivation with various tissues in the body, resulting in an autoimmune response. Paraneoplastic cerebellar degeneration (PCD) is a rare condition caused by immune-mediated damage to the Purkinje cells of the cerebellar tract. Symptoms may include gait instability, double vision, decreased fine motor skills, and ataxia, with progression to brainstem-associated symptoms, such as nystagmus, dysarthria, and dysphagia. Early detection and treatment of the underlying malignancy is critical to halt the progression of autoimmune-mediated destruction. We present a case of a young adult female patient with PCD caused by Purkinje cell cytoplasmic–Tr (PCA-Tr) antibody with Hodgkin lymphoma.
Case Presentation
A 20-year-old previously healthy active-duty female patient presented to the emergency department with acute worsening of chronic intermittent, recurrent episodes of lightheadedness and vertigo. Symptoms persisted for 9 months until acutely worsening over the 2 weeks prior to presentation. She reported left eye double vision but did not report seeing spots, photophobia, tinnitus, or headache. She felt off-balance, leaning on nearby objects to remain standing. Symptoms primarily occurred during ambulation; however, occasionally they happened at rest. Episodes lasted up to several minutes and occurred up to 15 times a day. The patient reported no fever, night sweats, unexplained weight loss, muscle aches, weakness, numbness or tingling, loss of bowel or bladder function, or rash. She had no recent illnesses, changes to medications, or recent travel. Oral intake to include food and water was adequate and unchanged. The patient had a remote history of mild concussions without loss of consciousness while playing sports 4 years previously. She reported no recent trauma. Nine months before, she received treatment for benign paroxysmal positional vertigo (BPPV) with the Epley maneuver with full resolution of symptoms lasting several days. She reported no prescription or over-the-counter medications, herbal remedies, or supplements. She reported no other medical or surgical history and no pertinent social or family history.
Physical examination revealed a nontoxic-appearing female patient with intermittent conversational dysarthria, saccadic pursuits, horizontal nystagmus with lateral gaze, and vertical nystagmus with vertical gaze. The patient exhibited dysdiadochokinesia, or impaired ability to perform rapid alternating hand movements with repetition. Finger-to-nose testing was impaired and heel-to-shin motion remained intact. A Romberg test was positive, and the patient had tandem gait instability. Strength testing, sensation, reflexes, and cranial nerves were otherwise intact. Initial laboratory testing was unremarkable except for mild normocytic anemia. Her infectious workup, including testing for venereal disease, HIV, COVID-19, and Coccidioidies was negative. Heavy metals analysis and urine drug screen were negative. Ophthalmology was consulted and workup revealed small amplitude downbeat nystagmus in primary gaze, sustained gaze evoked lateral beating jerk nystagmus with rebound nystagmus R>L gaze, but there was no evidence of afferent package defect and optic nerve function remained intact. Magnetic resonance imaging of the brain demonstrated cerebellar vermis hypoplasia with prominence of the superior cerebellar folia. Due to concerns for autoimmune encephalitis, a lumbar puncture was performed. Antibody testing revealed PCA-Tr antibodies, which is commonly associated with Hodgkin lymphoma, prompting further evaluation for malignancy.
Computed tomography (CT) of the chest with contrast demonstrated multiple mediastinal masses with a conglomeration of lymph nodes along the right paratracheal region. Further evaluation was performed with a positron emission tomography (PET)–CT, revealing a large conglomeration of hypermetabolic pretracheal, mediastinal, and right supraclavicular lymph that were suggestive of lymphoma. Mediastinoscopy with excisional lymph node biopsy was performed with immunohistochemical staining confirming diagnosis of a nodular sclerosing variant of Hodgkin lymphoma. The patient was treated with IV immunoglobulin at 0.4g/kg daily for 5 days. A central venous catheter was placed into the patient’s right internal jugular vein and a chemotherapy regimen of doxorubicin 46 mg, vinblastine 11 mg, bleomycin 19 units, and dacarbazine 700 mg was initiated. The patient’s symptoms improved with resolution of dysarthria; however, her visual impairment and gait instability persisted. Repeat PET-CT imaging 2 months later revealed interval improvement with decreased intensity and extent of the hypermetabolic lymph nodes and no new hypermetabolic foci.
Discussion
PCA-Tr antibodies affect the delta/notchlike epidermal growth factor–related receptor, expressed on the dendrites of cerebellar Purkinje cells.1 These fibers are the only output neurons of the cerebellar cortex and are critical to the coordination of motor movements, accounting for the ataxia experienced by patients with this subtype of PCD.2 The link between Hodgkin lymphoma and PCA-Tr antibodies has been established; however, most reports involve men with a median age of 61 years with lymphoma-associated symptoms (such as lymphadenopathy) or systemic symptoms (fever, night sweats, or weight loss) preceding neurologic manifestations in 80% of cases.3
Our patient was a young, previously healthy adult female who initially presented with vertigo, a common concern with frequently benign origins. Although there was temporary resolution of symptoms after Epley maneuvers, symptoms recurred and progressed over several months to include brainstem manifestations of nystagmus, diplopia, and dysarthria. Previous reports indicate that after remission of the Hodgkin lymphoma, PCA-Tr antibodies disappear and symptoms can improve or resolve.4,5 Treatment has just begun for our patient and although there has been initial clinical improvement, given the chronicity of symptoms, it is unclear if complete resolution will be achieved.
Conclusions
PCD can result in debilitating neurologic dysfunction and may be associated with malignancy such as Hodgkin lymphoma. This case offers unique insight due to the patient’s demographics and presentation, which involved brainstem pathology typically associated with late-onset disease and preceded by constitutional symptoms. Clinical suspicion of this rare disorder should be considered in all ages, especially if symptoms are progressive or neurologic manifestations arise, as early detection and treatment of the underlying malignancy are paramount to the prevention of significant disability.
Paraneoplastic syndrome is a rare disorder involving manifestations of immune dysregulation triggered by malignancy. The immune system develops antibodies to the malignancy, which can cause cross reactivation with various tissues in the body, resulting in an autoimmune response. Paraneoplastic cerebellar degeneration (PCD) is a rare condition caused by immune-mediated damage to the Purkinje cells of the cerebellar tract. Symptoms may include gait instability, double vision, decreased fine motor skills, and ataxia, with progression to brainstem-associated symptoms, such as nystagmus, dysarthria, and dysphagia. Early detection and treatment of the underlying malignancy is critical to halt the progression of autoimmune-mediated destruction. We present a case of a young adult female patient with PCD caused by Purkinje cell cytoplasmic–Tr (PCA-Tr) antibody with Hodgkin lymphoma.
Case Presentation
A 20-year-old previously healthy active-duty female patient presented to the emergency department with acute worsening of chronic intermittent, recurrent episodes of lightheadedness and vertigo. Symptoms persisted for 9 months until acutely worsening over the 2 weeks prior to presentation. She reported left eye double vision but did not report seeing spots, photophobia, tinnitus, or headache. She felt off-balance, leaning on nearby objects to remain standing. Symptoms primarily occurred during ambulation; however, occasionally they happened at rest. Episodes lasted up to several minutes and occurred up to 15 times a day. The patient reported no fever, night sweats, unexplained weight loss, muscle aches, weakness, numbness or tingling, loss of bowel or bladder function, or rash. She had no recent illnesses, changes to medications, or recent travel. Oral intake to include food and water was adequate and unchanged. The patient had a remote history of mild concussions without loss of consciousness while playing sports 4 years previously. She reported no recent trauma. Nine months before, she received treatment for benign paroxysmal positional vertigo (BPPV) with the Epley maneuver with full resolution of symptoms lasting several days. She reported no prescription or over-the-counter medications, herbal remedies, or supplements. She reported no other medical or surgical history and no pertinent social or family history.
Physical examination revealed a nontoxic-appearing female patient with intermittent conversational dysarthria, saccadic pursuits, horizontal nystagmus with lateral gaze, and vertical nystagmus with vertical gaze. The patient exhibited dysdiadochokinesia, or impaired ability to perform rapid alternating hand movements with repetition. Finger-to-nose testing was impaired and heel-to-shin motion remained intact. A Romberg test was positive, and the patient had tandem gait instability. Strength testing, sensation, reflexes, and cranial nerves were otherwise intact. Initial laboratory testing was unremarkable except for mild normocytic anemia. Her infectious workup, including testing for venereal disease, HIV, COVID-19, and Coccidioidies was negative. Heavy metals analysis and urine drug screen were negative. Ophthalmology was consulted and workup revealed small amplitude downbeat nystagmus in primary gaze, sustained gaze evoked lateral beating jerk nystagmus with rebound nystagmus R>L gaze, but there was no evidence of afferent package defect and optic nerve function remained intact. Magnetic resonance imaging of the brain demonstrated cerebellar vermis hypoplasia with prominence of the superior cerebellar folia. Due to concerns for autoimmune encephalitis, a lumbar puncture was performed. Antibody testing revealed PCA-Tr antibodies, which is commonly associated with Hodgkin lymphoma, prompting further evaluation for malignancy.
Computed tomography (CT) of the chest with contrast demonstrated multiple mediastinal masses with a conglomeration of lymph nodes along the right paratracheal region. Further evaluation was performed with a positron emission tomography (PET)–CT, revealing a large conglomeration of hypermetabolic pretracheal, mediastinal, and right supraclavicular lymph that were suggestive of lymphoma. Mediastinoscopy with excisional lymph node biopsy was performed with immunohistochemical staining confirming diagnosis of a nodular sclerosing variant of Hodgkin lymphoma. The patient was treated with IV immunoglobulin at 0.4g/kg daily for 5 days. A central venous catheter was placed into the patient’s right internal jugular vein and a chemotherapy regimen of doxorubicin 46 mg, vinblastine 11 mg, bleomycin 19 units, and dacarbazine 700 mg was initiated. The patient’s symptoms improved with resolution of dysarthria; however, her visual impairment and gait instability persisted. Repeat PET-CT imaging 2 months later revealed interval improvement with decreased intensity and extent of the hypermetabolic lymph nodes and no new hypermetabolic foci.
Discussion
PCA-Tr antibodies affect the delta/notchlike epidermal growth factor–related receptor, expressed on the dendrites of cerebellar Purkinje cells.1 These fibers are the only output neurons of the cerebellar cortex and are critical to the coordination of motor movements, accounting for the ataxia experienced by patients with this subtype of PCD.2 The link between Hodgkin lymphoma and PCA-Tr antibodies has been established; however, most reports involve men with a median age of 61 years with lymphoma-associated symptoms (such as lymphadenopathy) or systemic symptoms (fever, night sweats, or weight loss) preceding neurologic manifestations in 80% of cases.3
Our patient was a young, previously healthy adult female who initially presented with vertigo, a common concern with frequently benign origins. Although there was temporary resolution of symptoms after Epley maneuvers, symptoms recurred and progressed over several months to include brainstem manifestations of nystagmus, diplopia, and dysarthria. Previous reports indicate that after remission of the Hodgkin lymphoma, PCA-Tr antibodies disappear and symptoms can improve or resolve.4,5 Treatment has just begun for our patient and although there has been initial clinical improvement, given the chronicity of symptoms, it is unclear if complete resolution will be achieved.
Conclusions
PCD can result in debilitating neurologic dysfunction and may be associated with malignancy such as Hodgkin lymphoma. This case offers unique insight due to the patient’s demographics and presentation, which involved brainstem pathology typically associated with late-onset disease and preceded by constitutional symptoms. Clinical suspicion of this rare disorder should be considered in all ages, especially if symptoms are progressive or neurologic manifestations arise, as early detection and treatment of the underlying malignancy are paramount to the prevention of significant disability.
1. de Graaff E, Maat P, Hulsenboom E, et al. Identification of delta/notch-like epidermal growth factor-related receptor as the Tr antigen in paraneoplastic cerebellar degeneration. Ann Neurol. 2012;71(6):815-824. doi:10.1002/ana.23550
2. MacKenzie-Graham A, Tiwari-Woodruff SK, Sharma G, et al. Purkinje cell loss in experimental autoimmune encephalomyelitis. Neuroimage. 2009;48(4):637-651. doi:10.1016/j.neuroimage.2009.06.073
3. Bernal F, Shams’ili S, Rojas I, et al. Anti-Tr antibodies as markers of paraneoplastic cerebellar degeneration and Hodgkin’s disease. Neurology. 2003;60(2):230-234. doi:10.1212/01.wnl.0000041495.87539.98
4. Graus F, Ariño H, Dalmau J. Paraneoplastic neurological syndromes in Hodgkin and non-Hodgkin lymphomas. Blood. 2014;123(21):3230-3238. doi:10.1182/blood-2014-03-537506
5. Aly R, Emmady PD. Paraneoplastic cerebellar degeneration. Updated May 8, 2022. Accessed March 30, 2022. https://www.ncbi.nlm.nih.gov/books/NBK560638
1. de Graaff E, Maat P, Hulsenboom E, et al. Identification of delta/notch-like epidermal growth factor-related receptor as the Tr antigen in paraneoplastic cerebellar degeneration. Ann Neurol. 2012;71(6):815-824. doi:10.1002/ana.23550
2. MacKenzie-Graham A, Tiwari-Woodruff SK, Sharma G, et al. Purkinje cell loss in experimental autoimmune encephalomyelitis. Neuroimage. 2009;48(4):637-651. doi:10.1016/j.neuroimage.2009.06.073
3. Bernal F, Shams’ili S, Rojas I, et al. Anti-Tr antibodies as markers of paraneoplastic cerebellar degeneration and Hodgkin’s disease. Neurology. 2003;60(2):230-234. doi:10.1212/01.wnl.0000041495.87539.98
4. Graus F, Ariño H, Dalmau J. Paraneoplastic neurological syndromes in Hodgkin and non-Hodgkin lymphomas. Blood. 2014;123(21):3230-3238. doi:10.1182/blood-2014-03-537506
5. Aly R, Emmady PD. Paraneoplastic cerebellar degeneration. Updated May 8, 2022. Accessed March 30, 2022. https://www.ncbi.nlm.nih.gov/books/NBK560638
Cancer drug significantly cuts risk for COVID-19 death
, an interim analysis of a phase 3 placebo-controlled trial found.
Sabizabulin treatment consistently and significantly reduced deaths across patient subgroups “regardless of standard of care treatment received, baseline World Health Organization scores, age, comorbidities, vaccination status, COVID-19 variant, or geography,” study investigator Mitchell Steiner, MD, chairman, president, and CEO of Veru, said in a news release.
The company has submitted an emergency use authorization request to the U.S. Food and Drug Administration to use sabizabulin to treat COVID-19.
The analysis was published online in NEJM Evidence.
Sabizabulin, originally developed to treat metastatic castration-resistant prostate cancer, is a novel, investigational, oral microtubule disruptor with dual antiviral and anti-inflammatory activities. Given the drug’s mechanism, researchers at Veru thought that sabizabulin could help treat lung inflammation in patients with COVID-19 as well.
Findings of the interim analysis are based on 150 adults hospitalized with moderate to severe COVID-19 at high risk for acute respiratory distress syndrome and death. The patients were randomly allocated to receive 9 mg oral sabizabulin (n = 98) or placebo (n = 52) once daily for up to 21 days.
Overall, the mortality rate was 20.2% in the sabizabulin group vs. 45.1% in the placebo group. Compared with placebo, treatment with sabizabulin led to a 24.9–percentage point absolute reduction and a 55.2% relative reduction in death (odds ratio, 3.23; P = .0042).
The key secondary endpoint of mortality through day 29 also favored sabizabulin over placebo, with a mortality rate of 17% vs. 35.3%. In this scenario, treatment with sabizabulin resulted in an absolute reduction in deaths of 18.3 percentage points and a relative reduction of 51.8%.
Sabizabulin led to a significant 43% relative reduction in ICU days, a 49% relative reduction in days on mechanical ventilation, and a 26% relative reduction in days in the hospital, compared with placebo.
Adverse and serious adverse events were also lower in the sabizabulin group (61.5%) than the placebo group (78.3%).
The data are “pretty impressive and in a group of patients that we really have limited things to offer,” Aaron Glatt, MD, a spokesperson for the Infectious Diseases Society of America and chief of infectious diseases and hospital epidemiologist at Mount Sinai South Nassau in Oceanside, N.Y., said in an interview. “This is an interim analysis and obviously we’d like to see more data, but it certainly is something that is novel and quite interesting.”
David Boulware, MD, MPH, an infectious disease expert at the University of Minnesota, Minneapolis, told the New York Times that the large number of deaths in the placebo group seemed “rather high” and that the final analysis might reveal a more modest benefit for sabizabulin.
“I would be skeptical” that the reduced risk for death remains 55%, he noted.
The study was funded by Veru Pharmaceuticals. Several authors are employed by the company or have financial relationships with the company.
A version of this article first appeared on Medscape.com.
, an interim analysis of a phase 3 placebo-controlled trial found.
Sabizabulin treatment consistently and significantly reduced deaths across patient subgroups “regardless of standard of care treatment received, baseline World Health Organization scores, age, comorbidities, vaccination status, COVID-19 variant, or geography,” study investigator Mitchell Steiner, MD, chairman, president, and CEO of Veru, said in a news release.
The company has submitted an emergency use authorization request to the U.S. Food and Drug Administration to use sabizabulin to treat COVID-19.
The analysis was published online in NEJM Evidence.
Sabizabulin, originally developed to treat metastatic castration-resistant prostate cancer, is a novel, investigational, oral microtubule disruptor with dual antiviral and anti-inflammatory activities. Given the drug’s mechanism, researchers at Veru thought that sabizabulin could help treat lung inflammation in patients with COVID-19 as well.
Findings of the interim analysis are based on 150 adults hospitalized with moderate to severe COVID-19 at high risk for acute respiratory distress syndrome and death. The patients were randomly allocated to receive 9 mg oral sabizabulin (n = 98) or placebo (n = 52) once daily for up to 21 days.
Overall, the mortality rate was 20.2% in the sabizabulin group vs. 45.1% in the placebo group. Compared with placebo, treatment with sabizabulin led to a 24.9–percentage point absolute reduction and a 55.2% relative reduction in death (odds ratio, 3.23; P = .0042).
The key secondary endpoint of mortality through day 29 also favored sabizabulin over placebo, with a mortality rate of 17% vs. 35.3%. In this scenario, treatment with sabizabulin resulted in an absolute reduction in deaths of 18.3 percentage points and a relative reduction of 51.8%.
Sabizabulin led to a significant 43% relative reduction in ICU days, a 49% relative reduction in days on mechanical ventilation, and a 26% relative reduction in days in the hospital, compared with placebo.
Adverse and serious adverse events were also lower in the sabizabulin group (61.5%) than the placebo group (78.3%).
The data are “pretty impressive and in a group of patients that we really have limited things to offer,” Aaron Glatt, MD, a spokesperson for the Infectious Diseases Society of America and chief of infectious diseases and hospital epidemiologist at Mount Sinai South Nassau in Oceanside, N.Y., said in an interview. “This is an interim analysis and obviously we’d like to see more data, but it certainly is something that is novel and quite interesting.”
David Boulware, MD, MPH, an infectious disease expert at the University of Minnesota, Minneapolis, told the New York Times that the large number of deaths in the placebo group seemed “rather high” and that the final analysis might reveal a more modest benefit for sabizabulin.
“I would be skeptical” that the reduced risk for death remains 55%, he noted.
The study was funded by Veru Pharmaceuticals. Several authors are employed by the company or have financial relationships with the company.
A version of this article first appeared on Medscape.com.
, an interim analysis of a phase 3 placebo-controlled trial found.
Sabizabulin treatment consistently and significantly reduced deaths across patient subgroups “regardless of standard of care treatment received, baseline World Health Organization scores, age, comorbidities, vaccination status, COVID-19 variant, or geography,” study investigator Mitchell Steiner, MD, chairman, president, and CEO of Veru, said in a news release.
The company has submitted an emergency use authorization request to the U.S. Food and Drug Administration to use sabizabulin to treat COVID-19.
The analysis was published online in NEJM Evidence.
Sabizabulin, originally developed to treat metastatic castration-resistant prostate cancer, is a novel, investigational, oral microtubule disruptor with dual antiviral and anti-inflammatory activities. Given the drug’s mechanism, researchers at Veru thought that sabizabulin could help treat lung inflammation in patients with COVID-19 as well.
Findings of the interim analysis are based on 150 adults hospitalized with moderate to severe COVID-19 at high risk for acute respiratory distress syndrome and death. The patients were randomly allocated to receive 9 mg oral sabizabulin (n = 98) or placebo (n = 52) once daily for up to 21 days.
Overall, the mortality rate was 20.2% in the sabizabulin group vs. 45.1% in the placebo group. Compared with placebo, treatment with sabizabulin led to a 24.9–percentage point absolute reduction and a 55.2% relative reduction in death (odds ratio, 3.23; P = .0042).
The key secondary endpoint of mortality through day 29 also favored sabizabulin over placebo, with a mortality rate of 17% vs. 35.3%. In this scenario, treatment with sabizabulin resulted in an absolute reduction in deaths of 18.3 percentage points and a relative reduction of 51.8%.
Sabizabulin led to a significant 43% relative reduction in ICU days, a 49% relative reduction in days on mechanical ventilation, and a 26% relative reduction in days in the hospital, compared with placebo.
Adverse and serious adverse events were also lower in the sabizabulin group (61.5%) than the placebo group (78.3%).
The data are “pretty impressive and in a group of patients that we really have limited things to offer,” Aaron Glatt, MD, a spokesperson for the Infectious Diseases Society of America and chief of infectious diseases and hospital epidemiologist at Mount Sinai South Nassau in Oceanside, N.Y., said in an interview. “This is an interim analysis and obviously we’d like to see more data, but it certainly is something that is novel and quite interesting.”
David Boulware, MD, MPH, an infectious disease expert at the University of Minnesota, Minneapolis, told the New York Times that the large number of deaths in the placebo group seemed “rather high” and that the final analysis might reveal a more modest benefit for sabizabulin.
“I would be skeptical” that the reduced risk for death remains 55%, he noted.
The study was funded by Veru Pharmaceuticals. Several authors are employed by the company or have financial relationships with the company.
A version of this article first appeared on Medscape.com.
FROM NEJM EVIDENCE
Pembrolizumab for melanoma bittersweet, doctor says
CHICAGO – Pembrolizumab has shown promise as adjuvant therapy for stage IIB and IIC melanoma, shows the first interim analysis of the phase 3 KEYNOTE-716 study recently published in The Lancet.
The findings meet an unmet need as the recurrence risk in stage IIB and IIC melanoma is “underrecognized,” said author Georgina Long, MD, comedical director of the Melanoma Institute Australia, University of Sydney.
In fact, their risk of recurrence is similar to patients with stage IIIB disease, wrote David Killock, PhD, in a related commentary published in Nature Reviews.
The adjuvant treatment resulted in an 89% recurrence-free survival in patients who received pembrolizumab, compared with 83% of patients in the placebo group (hazard ratio, 0.65; P = .0066). These findings were used as the basis for Food and Drug Administration approval of pembrolizumab (Keytruda, Merck) for this patient population in December 2021.
Despite the positive findings, Dr. Killock called for more research on distant metastasis-free survival, overall survival, and quality of life data to “establish the true clinical benefit of adjuvant pembrolizumab.”
At the annual meeting of the American Society of Clinical Oncology, Dr. Long presented the third interim analysis which showed pembrolizumab reduced recurrence and distant metastases at 24 months, although the clinical benefit was relatively small at an approximately 8% improvement in recurrence-free survival and about a 6% improvement in distant metastasis-free survival. About 83% in the pembrolizumab group had treatment-related toxicities versus 64% in the placebo group. There were no deaths caused by treatment. About 90% of pembrolizumab-related endocrinopathies led to long-term hormone replacement.
In a discussion that followed the presentation at ASCO, Charlotte Eielson Ariyan, MD, PhD, said the results are bittersweet. Higher-risk stage IIC patients have a risk of recurrence of about 40%. “It’s high, but the absolute risk reduction is about 8%. This is a very personalized discussion with the patient and the physician in understanding their risk of toxicity is about 17% and higher than their absolute risk reduction with the treatment. For me, this is a bitter pill to swallow because you’re treating people longer and you’re not sure if you’re really helping them. Until we can further define who the highest-risk patients are, I think it’s hard to give it to everyone,” said Dr. Ariyan, who is a surgeon with Memorial Sloan Kettering Cancer Center, New York.
In addition to weighing short-term benefits and toxicity, there are longer-term concerns. Toxicity experienced from PD-1 inhibitors in the adjuvant setting could impact future treatment decisions. “We’re very lucky here in melanoma to know that systemic therapies are effective and we can cure people who recur. I would argue this is why we probably will never really see a difference in the survival benefit in this group because people who cross over will probably do well,” Dr. Ariyan said.
During the Q&A session, Vernon Sondek, MD, Moffitt Cancer Center, Tampa, encouraged physician colleagues to have an open mind about treatments. “Beware of dogma. We thought that adjuvant immunotherapy works much better in patients with ulcerated primary tumors. That’s a dogma in some parts of the world. Yet the T4a patients in KEYNOTE-716 dramatically outperformed the ulcerated T3b and T4b [patients]. We still don’t know what we don’t know.”
The study details
KEYNOTE-716 included 976 patients 12 years or older with newly diagnosed completely resected stage IIB or IIC melanoma with a negative sentinel lymph node. Patients were randomized to placebo or 200 mg pembrolizumab every 3 weeks, or 2 mg/kg in pediatric patients, over 17 cycles. Almost 40% of patients were age 65 or older. T3b and T4b were the most common melanoma subcategories at 41% and 35%, respectively.
The planned third interim analysis occurred after the occurrence of 146 distant metastases. After a median follow-up of 27.4 months, distant metastasis-free survival favored the pembrolizumab group (HR, 0.64; P = .0029). At 24 months, the pembrolizumab group had a higher distant metastasis-free survival at 88.1% versus 82.2% and a lower recurrence rate at 81.2% versus 72.8% (HR, 0.64; 95% confidence interval, 0.50-0.84).
At 24 months, only the T4a patients had a statistically significant reduction in distant metastases at 58% (HR, 0.42; 95% CI, 0.19-0.96), although there were numerical reductions in T3a (HR, 0.71; 95% CI, 0.41-1.22) and T4b (HR, 0.70; 95% CI, 0.44-1.33) patients. Of patients experiencing a distant metastasis, 73% of the placebo group had a first distant metastasis to the lung compared with 49% of the pembrolizumab group.
Dr. Long has held consulting or advisory roles for Merck Sharpe & Dohme, which funded this study.
CHICAGO – Pembrolizumab has shown promise as adjuvant therapy for stage IIB and IIC melanoma, shows the first interim analysis of the phase 3 KEYNOTE-716 study recently published in The Lancet.
The findings meet an unmet need as the recurrence risk in stage IIB and IIC melanoma is “underrecognized,” said author Georgina Long, MD, comedical director of the Melanoma Institute Australia, University of Sydney.
In fact, their risk of recurrence is similar to patients with stage IIIB disease, wrote David Killock, PhD, in a related commentary published in Nature Reviews.
The adjuvant treatment resulted in an 89% recurrence-free survival in patients who received pembrolizumab, compared with 83% of patients in the placebo group (hazard ratio, 0.65; P = .0066). These findings were used as the basis for Food and Drug Administration approval of pembrolizumab (Keytruda, Merck) for this patient population in December 2021.
Despite the positive findings, Dr. Killock called for more research on distant metastasis-free survival, overall survival, and quality of life data to “establish the true clinical benefit of adjuvant pembrolizumab.”
At the annual meeting of the American Society of Clinical Oncology, Dr. Long presented the third interim analysis which showed pembrolizumab reduced recurrence and distant metastases at 24 months, although the clinical benefit was relatively small at an approximately 8% improvement in recurrence-free survival and about a 6% improvement in distant metastasis-free survival. About 83% in the pembrolizumab group had treatment-related toxicities versus 64% in the placebo group. There were no deaths caused by treatment. About 90% of pembrolizumab-related endocrinopathies led to long-term hormone replacement.
In a discussion that followed the presentation at ASCO, Charlotte Eielson Ariyan, MD, PhD, said the results are bittersweet. Higher-risk stage IIC patients have a risk of recurrence of about 40%. “It’s high, but the absolute risk reduction is about 8%. This is a very personalized discussion with the patient and the physician in understanding their risk of toxicity is about 17% and higher than their absolute risk reduction with the treatment. For me, this is a bitter pill to swallow because you’re treating people longer and you’re not sure if you’re really helping them. Until we can further define who the highest-risk patients are, I think it’s hard to give it to everyone,” said Dr. Ariyan, who is a surgeon with Memorial Sloan Kettering Cancer Center, New York.
In addition to weighing short-term benefits and toxicity, there are longer-term concerns. Toxicity experienced from PD-1 inhibitors in the adjuvant setting could impact future treatment decisions. “We’re very lucky here in melanoma to know that systemic therapies are effective and we can cure people who recur. I would argue this is why we probably will never really see a difference in the survival benefit in this group because people who cross over will probably do well,” Dr. Ariyan said.
During the Q&A session, Vernon Sondek, MD, Moffitt Cancer Center, Tampa, encouraged physician colleagues to have an open mind about treatments. “Beware of dogma. We thought that adjuvant immunotherapy works much better in patients with ulcerated primary tumors. That’s a dogma in some parts of the world. Yet the T4a patients in KEYNOTE-716 dramatically outperformed the ulcerated T3b and T4b [patients]. We still don’t know what we don’t know.”
The study details
KEYNOTE-716 included 976 patients 12 years or older with newly diagnosed completely resected stage IIB or IIC melanoma with a negative sentinel lymph node. Patients were randomized to placebo or 200 mg pembrolizumab every 3 weeks, or 2 mg/kg in pediatric patients, over 17 cycles. Almost 40% of patients were age 65 or older. T3b and T4b were the most common melanoma subcategories at 41% and 35%, respectively.
The planned third interim analysis occurred after the occurrence of 146 distant metastases. After a median follow-up of 27.4 months, distant metastasis-free survival favored the pembrolizumab group (HR, 0.64; P = .0029). At 24 months, the pembrolizumab group had a higher distant metastasis-free survival at 88.1% versus 82.2% and a lower recurrence rate at 81.2% versus 72.8% (HR, 0.64; 95% confidence interval, 0.50-0.84).
At 24 months, only the T4a patients had a statistically significant reduction in distant metastases at 58% (HR, 0.42; 95% CI, 0.19-0.96), although there were numerical reductions in T3a (HR, 0.71; 95% CI, 0.41-1.22) and T4b (HR, 0.70; 95% CI, 0.44-1.33) patients. Of patients experiencing a distant metastasis, 73% of the placebo group had a first distant metastasis to the lung compared with 49% of the pembrolizumab group.
Dr. Long has held consulting or advisory roles for Merck Sharpe & Dohme, which funded this study.
CHICAGO – Pembrolizumab has shown promise as adjuvant therapy for stage IIB and IIC melanoma, shows the first interim analysis of the phase 3 KEYNOTE-716 study recently published in The Lancet.
The findings meet an unmet need as the recurrence risk in stage IIB and IIC melanoma is “underrecognized,” said author Georgina Long, MD, comedical director of the Melanoma Institute Australia, University of Sydney.
In fact, their risk of recurrence is similar to patients with stage IIIB disease, wrote David Killock, PhD, in a related commentary published in Nature Reviews.
The adjuvant treatment resulted in an 89% recurrence-free survival in patients who received pembrolizumab, compared with 83% of patients in the placebo group (hazard ratio, 0.65; P = .0066). These findings were used as the basis for Food and Drug Administration approval of pembrolizumab (Keytruda, Merck) for this patient population in December 2021.
Despite the positive findings, Dr. Killock called for more research on distant metastasis-free survival, overall survival, and quality of life data to “establish the true clinical benefit of adjuvant pembrolizumab.”
At the annual meeting of the American Society of Clinical Oncology, Dr. Long presented the third interim analysis which showed pembrolizumab reduced recurrence and distant metastases at 24 months, although the clinical benefit was relatively small at an approximately 8% improvement in recurrence-free survival and about a 6% improvement in distant metastasis-free survival. About 83% in the pembrolizumab group had treatment-related toxicities versus 64% in the placebo group. There were no deaths caused by treatment. About 90% of pembrolizumab-related endocrinopathies led to long-term hormone replacement.
In a discussion that followed the presentation at ASCO, Charlotte Eielson Ariyan, MD, PhD, said the results are bittersweet. Higher-risk stage IIC patients have a risk of recurrence of about 40%. “It’s high, but the absolute risk reduction is about 8%. This is a very personalized discussion with the patient and the physician in understanding their risk of toxicity is about 17% and higher than their absolute risk reduction with the treatment. For me, this is a bitter pill to swallow because you’re treating people longer and you’re not sure if you’re really helping them. Until we can further define who the highest-risk patients are, I think it’s hard to give it to everyone,” said Dr. Ariyan, who is a surgeon with Memorial Sloan Kettering Cancer Center, New York.
In addition to weighing short-term benefits and toxicity, there are longer-term concerns. Toxicity experienced from PD-1 inhibitors in the adjuvant setting could impact future treatment decisions. “We’re very lucky here in melanoma to know that systemic therapies are effective and we can cure people who recur. I would argue this is why we probably will never really see a difference in the survival benefit in this group because people who cross over will probably do well,” Dr. Ariyan said.
During the Q&A session, Vernon Sondek, MD, Moffitt Cancer Center, Tampa, encouraged physician colleagues to have an open mind about treatments. “Beware of dogma. We thought that adjuvant immunotherapy works much better in patients with ulcerated primary tumors. That’s a dogma in some parts of the world. Yet the T4a patients in KEYNOTE-716 dramatically outperformed the ulcerated T3b and T4b [patients]. We still don’t know what we don’t know.”
The study details
KEYNOTE-716 included 976 patients 12 years or older with newly diagnosed completely resected stage IIB or IIC melanoma with a negative sentinel lymph node. Patients were randomized to placebo or 200 mg pembrolizumab every 3 weeks, or 2 mg/kg in pediatric patients, over 17 cycles. Almost 40% of patients were age 65 or older. T3b and T4b were the most common melanoma subcategories at 41% and 35%, respectively.
The planned third interim analysis occurred after the occurrence of 146 distant metastases. After a median follow-up of 27.4 months, distant metastasis-free survival favored the pembrolizumab group (HR, 0.64; P = .0029). At 24 months, the pembrolizumab group had a higher distant metastasis-free survival at 88.1% versus 82.2% and a lower recurrence rate at 81.2% versus 72.8% (HR, 0.64; 95% confidence interval, 0.50-0.84).
At 24 months, only the T4a patients had a statistically significant reduction in distant metastases at 58% (HR, 0.42; 95% CI, 0.19-0.96), although there were numerical reductions in T3a (HR, 0.71; 95% CI, 0.41-1.22) and T4b (HR, 0.70; 95% CI, 0.44-1.33) patients. Of patients experiencing a distant metastasis, 73% of the placebo group had a first distant metastasis to the lung compared with 49% of the pembrolizumab group.
Dr. Long has held consulting or advisory roles for Merck Sharpe & Dohme, which funded this study.
AT ASCO 2022
Pregnancy, breastfeeding, and more linked to lower CRC risk
Estrogen exposure helps protect against colorectal cancer (CRC), and in some instances, the protection is site specific, a new analysis finds.
In a 17-year study involving almost 5,000 women, researchers from Germany found that hormone replacement therapy, oral contraceptive use, pregnancy, breastfeeding, and menopause at age 50 or older were all significantly associated with reductions in CRC risk.
Interestingly, the reduced risk of CRC observed for pregnancy and breastfeeding only applied to proximal colon cancer, while the association with oral contraceptive use was confined to the distal colon and rectum.
The results were published online in JNCI Cancer Spectrum.
CRC is the second most common cause of cancer death. It is responsible for more than one million deaths globally, according to the latest figures from the Global Burden of Disease 2019 Cancer Collaboration.
And sex seems to make a difference. The Global Burden analysis, echoing previous data, found that CRC is less common among women and that fewer women die from the disease.
Little, however, is known about the mechanisms of estrogen signaling in CRC or the impact of reproductive factors on CRC, despite a large amount of literature linking CRC risk to exogenous estrogens, such as hormone replacement therapy and oral contraceptives.
In the current analysis, the team recruited 2,650 patients with CRC from 20 German cancer centers between 2003 and 2020. Researchers used standardized questionnaires to garner the women’s reproductive histories.
A matched control group of 2,175 participants who did not have a history of CRC was randomly selected from population registries. All analyses were adjusted for known CRC risk factors, such as age; body mass index; education level; family history; having previously undergone large-bowel endoscopy; diabetes; and smoking status.
The researchers found that each pregnancy was associated with a small but significant 9% reduction in CRC risk (odds ratio, 0.91), specifically in the proximal colon (OR, 0.86).
Overall, breastfeeding for a year or longer was associated with a significantly lower CRC risk, compared with never breastfeeding (OR, 0.74), but the results were only significant for the proximal colon (OR, 0.58).
Oral contraceptive use for 9 years or longer was associated with a lower CRC risk (OR, 0.75) but was only significant for the distal colon (OR, 0.63). Hormone replacement therapy was associated with a lower risk of CRC irrespective of tumor location (OR, 0.76). And using both was linked to a 42% CRC risk reduction (OR, 0.58).
Although age at menarche was not associated with CRC risk, menopause at age 50 or older was associated with a significant 17% lower risk of CRC.
In an email interview, lead author Tobias Niedermaier, PhD, expressed surprise at two of the findings. The first was the small association between pregnancies and CRC risk, “despite the strong increase in estrogen levels during pregnancy,” he said. He speculated that pregnancy-related increases in insulin levels may have “largely offset the protection effects of estrogen exposure during pregnancy.”
The second surprise was that the age at menarche did not have a bearing on CRC risk, which could be because “exposure to estrogen levels in younger ages [is] less relevant with respect to CRC risk, because CRC typically develops at comparably old age.”
John Marshall, MD, who was not involved in the research, commented that such studies “put a lot of pressure on people to perform in a certain way to modify their personal risk of something.” However, “we would not recommend people alter their life choices for reproduction for this,” said Dr. Marshall, chief of the Division of Hematology/Oncology at Georgetown University, Washington, D.C.
Dr. Niedermaier agreed that “while this knowledge will certainly not change a woman’s decision on family planning,” he noted that the findings “could influence current CRC screening strategies, for example, by risk-adapted screening intervals [and] start and stop ages of screening.”
Dr. Niedermaier and colleagues’ work was funded by the German Research Council, the German Federal Ministry of Education and Research, and the Interdisciplinary Research Program of the National Center for Tumor Diseases. Dr. Niedermaier has disclosed no relevant financial relationships. Dr. Marshall writes a column that appears regularly on Medscape: Marshall on Oncology. He has served as speaker or member of a speakers’ bureau for Genentech, Amgen, Bayer, Celgene Corporation, and Caris Life Sciences.
A version of this article first appeared on Medscape.com.
Estrogen exposure helps protect against colorectal cancer (CRC), and in some instances, the protection is site specific, a new analysis finds.
In a 17-year study involving almost 5,000 women, researchers from Germany found that hormone replacement therapy, oral contraceptive use, pregnancy, breastfeeding, and menopause at age 50 or older were all significantly associated with reductions in CRC risk.
Interestingly, the reduced risk of CRC observed for pregnancy and breastfeeding only applied to proximal colon cancer, while the association with oral contraceptive use was confined to the distal colon and rectum.
The results were published online in JNCI Cancer Spectrum.
CRC is the second most common cause of cancer death. It is responsible for more than one million deaths globally, according to the latest figures from the Global Burden of Disease 2019 Cancer Collaboration.
And sex seems to make a difference. The Global Burden analysis, echoing previous data, found that CRC is less common among women and that fewer women die from the disease.
Little, however, is known about the mechanisms of estrogen signaling in CRC or the impact of reproductive factors on CRC, despite a large amount of literature linking CRC risk to exogenous estrogens, such as hormone replacement therapy and oral contraceptives.
In the current analysis, the team recruited 2,650 patients with CRC from 20 German cancer centers between 2003 and 2020. Researchers used standardized questionnaires to garner the women’s reproductive histories.
A matched control group of 2,175 participants who did not have a history of CRC was randomly selected from population registries. All analyses were adjusted for known CRC risk factors, such as age; body mass index; education level; family history; having previously undergone large-bowel endoscopy; diabetes; and smoking status.
The researchers found that each pregnancy was associated with a small but significant 9% reduction in CRC risk (odds ratio, 0.91), specifically in the proximal colon (OR, 0.86).
Overall, breastfeeding for a year or longer was associated with a significantly lower CRC risk, compared with never breastfeeding (OR, 0.74), but the results were only significant for the proximal colon (OR, 0.58).
Oral contraceptive use for 9 years or longer was associated with a lower CRC risk (OR, 0.75) but was only significant for the distal colon (OR, 0.63). Hormone replacement therapy was associated with a lower risk of CRC irrespective of tumor location (OR, 0.76). And using both was linked to a 42% CRC risk reduction (OR, 0.58).
Although age at menarche was not associated with CRC risk, menopause at age 50 or older was associated with a significant 17% lower risk of CRC.
In an email interview, lead author Tobias Niedermaier, PhD, expressed surprise at two of the findings. The first was the small association between pregnancies and CRC risk, “despite the strong increase in estrogen levels during pregnancy,” he said. He speculated that pregnancy-related increases in insulin levels may have “largely offset the protection effects of estrogen exposure during pregnancy.”
The second surprise was that the age at menarche did not have a bearing on CRC risk, which could be because “exposure to estrogen levels in younger ages [is] less relevant with respect to CRC risk, because CRC typically develops at comparably old age.”
John Marshall, MD, who was not involved in the research, commented that such studies “put a lot of pressure on people to perform in a certain way to modify their personal risk of something.” However, “we would not recommend people alter their life choices for reproduction for this,” said Dr. Marshall, chief of the Division of Hematology/Oncology at Georgetown University, Washington, D.C.
Dr. Niedermaier agreed that “while this knowledge will certainly not change a woman’s decision on family planning,” he noted that the findings “could influence current CRC screening strategies, for example, by risk-adapted screening intervals [and] start and stop ages of screening.”
Dr. Niedermaier and colleagues’ work was funded by the German Research Council, the German Federal Ministry of Education and Research, and the Interdisciplinary Research Program of the National Center for Tumor Diseases. Dr. Niedermaier has disclosed no relevant financial relationships. Dr. Marshall writes a column that appears regularly on Medscape: Marshall on Oncology. He has served as speaker or member of a speakers’ bureau for Genentech, Amgen, Bayer, Celgene Corporation, and Caris Life Sciences.
A version of this article first appeared on Medscape.com.
Estrogen exposure helps protect against colorectal cancer (CRC), and in some instances, the protection is site specific, a new analysis finds.
In a 17-year study involving almost 5,000 women, researchers from Germany found that hormone replacement therapy, oral contraceptive use, pregnancy, breastfeeding, and menopause at age 50 or older were all significantly associated with reductions in CRC risk.
Interestingly, the reduced risk of CRC observed for pregnancy and breastfeeding only applied to proximal colon cancer, while the association with oral contraceptive use was confined to the distal colon and rectum.
The results were published online in JNCI Cancer Spectrum.
CRC is the second most common cause of cancer death. It is responsible for more than one million deaths globally, according to the latest figures from the Global Burden of Disease 2019 Cancer Collaboration.
And sex seems to make a difference. The Global Burden analysis, echoing previous data, found that CRC is less common among women and that fewer women die from the disease.
Little, however, is known about the mechanisms of estrogen signaling in CRC or the impact of reproductive factors on CRC, despite a large amount of literature linking CRC risk to exogenous estrogens, such as hormone replacement therapy and oral contraceptives.
In the current analysis, the team recruited 2,650 patients with CRC from 20 German cancer centers between 2003 and 2020. Researchers used standardized questionnaires to garner the women’s reproductive histories.
A matched control group of 2,175 participants who did not have a history of CRC was randomly selected from population registries. All analyses were adjusted for known CRC risk factors, such as age; body mass index; education level; family history; having previously undergone large-bowel endoscopy; diabetes; and smoking status.
The researchers found that each pregnancy was associated with a small but significant 9% reduction in CRC risk (odds ratio, 0.91), specifically in the proximal colon (OR, 0.86).
Overall, breastfeeding for a year or longer was associated with a significantly lower CRC risk, compared with never breastfeeding (OR, 0.74), but the results were only significant for the proximal colon (OR, 0.58).
Oral contraceptive use for 9 years or longer was associated with a lower CRC risk (OR, 0.75) but was only significant for the distal colon (OR, 0.63). Hormone replacement therapy was associated with a lower risk of CRC irrespective of tumor location (OR, 0.76). And using both was linked to a 42% CRC risk reduction (OR, 0.58).
Although age at menarche was not associated with CRC risk, menopause at age 50 or older was associated with a significant 17% lower risk of CRC.
In an email interview, lead author Tobias Niedermaier, PhD, expressed surprise at two of the findings. The first was the small association between pregnancies and CRC risk, “despite the strong increase in estrogen levels during pregnancy,” he said. He speculated that pregnancy-related increases in insulin levels may have “largely offset the protection effects of estrogen exposure during pregnancy.”
The second surprise was that the age at menarche did not have a bearing on CRC risk, which could be because “exposure to estrogen levels in younger ages [is] less relevant with respect to CRC risk, because CRC typically develops at comparably old age.”
John Marshall, MD, who was not involved in the research, commented that such studies “put a lot of pressure on people to perform in a certain way to modify their personal risk of something.” However, “we would not recommend people alter their life choices for reproduction for this,” said Dr. Marshall, chief of the Division of Hematology/Oncology at Georgetown University, Washington, D.C.
Dr. Niedermaier agreed that “while this knowledge will certainly not change a woman’s decision on family planning,” he noted that the findings “could influence current CRC screening strategies, for example, by risk-adapted screening intervals [and] start and stop ages of screening.”
Dr. Niedermaier and colleagues’ work was funded by the German Research Council, the German Federal Ministry of Education and Research, and the Interdisciplinary Research Program of the National Center for Tumor Diseases. Dr. Niedermaier has disclosed no relevant financial relationships. Dr. Marshall writes a column that appears regularly on Medscape: Marshall on Oncology. He has served as speaker or member of a speakers’ bureau for Genentech, Amgen, Bayer, Celgene Corporation, and Caris Life Sciences.
A version of this article first appeared on Medscape.com.
New KRAS inhibitor shows promise in NSCLC
In a phase 2 cohort study, who had previously been treated with platinum-based chemotherapy and immune therapy.
Adagrasib targets KRAS (G12C), which had long been thought undruggable until research published in 2013 revealed a new binding pocket that did not compete directly against the protein’s natural binding partner. The new trial further validates the approach. “It supports that clinically effective targeted therapies can be developed for patients with KRAS (G12C)–mutant NSCLC,” said Pasi Jänne, MD, PhD, who is the lead author of the study describing the new results published online in the New England Journal of Medicine.
KRAS is the most frequently mutated oncogene in human cancers. A mutated form is found in about 25% of NSCLCs. KRAS plays a key role in cell signaling governing growth, maturation, and cell death. The mutated form is linked to cancer growth and spread. Patients with mutated KRAS have few effective treatment options.
Adagrasib is currently under study and not yet approved by the Food and Drug Administration. However, sotorasib (Lumakras, Amgen), which also inhibits KRAS (G12C), was approved in May 2021 by the FDA for KRAS (G12C)–mutated NSCLC. There are some key differences between the drugs. Adagrasib has a half-life of 23 hours versus 5 hours for sotorasib, and the newer drug has the potential to penetrate the central nervous system. That could be an important consideration in NSCLC since it often metastasizes to the brain. “Having pharmacological approaches to treat brain metastases is a wonderful new therapeutic option for lung cancer patients,” said Dr. Jänne, who is director of the Lowe Center for Thoracic Oncology at Dana Farber Cancer Institute, Boston.
Adagrasib is being investigated as part of the KRYSTAL-1 study, alone and as part of combinations in various solid tumors. Previously treated NSCLC KRAS (G12C) patients are also being enrolled in a phase 3 study of adagrasib combined with docetaxel, as well as another phase 2 study of adagrasib combined with pembrolizumab as first-line therapy for NSCLC KRAS (G12C).
Adagrasib is likely to remain a second-line therapy following chemotherapy and immunotherapy. “The activity by itself at the moment is not sufficient to be a first-line treatment. That may change in the future in combination with a standard of care agent or in a subset of patients with KRAS (G12C)–mutant NSCLC, although no subset with higher efficacy has been identified to date. Identification of predictive biomarkers for patients likely to benefit from single agent or an adagrasib combination treatment remains a high priority,” Dr. Jänne said.
The study included 116 patients who had previously been treated with platinum-based chemotherapy and anti–programmed death 1 or programmed death–ligand 1 therapy. They received 600 mg oral adagrasib twice per day over a median follow-up period of 12.9 months. About 42.9% (95% confidence interval, 33.5%-52.6%) experienced a confirmed objective response with a median duration of 8.5 months (95% CI, 6.2-13.8 months). The median progression-free survival was 6.5 months (95% CI, 4.7-8.4 months). After a median follow-up of 15.6 months, the median overall survival was 12.6 months (95% CI, 9.2-19.2 months). The estimated overall survival at 1 year was 50.8% (95% CI, 40.9%-60.0%).
33 patients had stable central nervous system metastases that had been previously treated. About 33.3% had an intracranial confirmed objective response (95% CI, 18.0-51.8%) with a median duration of response of 11.2 months (95% CI, 2.99 months to not available).
Adverse events are similar to what is seen with other targeted therapies, according to Dr. Jänne. 97.4% of patient reported a treatment-related adverse event; 52.6% had grade 1-2 adverse events, and 44.8% had grade 3 adverse events. 6.9% discontinued the drug as a result.
Dr. Jänne has consulted for Mirati Therapeutics and is a member of its scientific advisory board. The study was funded by Mirati Therapeutics.
In a phase 2 cohort study, who had previously been treated with platinum-based chemotherapy and immune therapy.
Adagrasib targets KRAS (G12C), which had long been thought undruggable until research published in 2013 revealed a new binding pocket that did not compete directly against the protein’s natural binding partner. The new trial further validates the approach. “It supports that clinically effective targeted therapies can be developed for patients with KRAS (G12C)–mutant NSCLC,” said Pasi Jänne, MD, PhD, who is the lead author of the study describing the new results published online in the New England Journal of Medicine.
KRAS is the most frequently mutated oncogene in human cancers. A mutated form is found in about 25% of NSCLCs. KRAS plays a key role in cell signaling governing growth, maturation, and cell death. The mutated form is linked to cancer growth and spread. Patients with mutated KRAS have few effective treatment options.
Adagrasib is currently under study and not yet approved by the Food and Drug Administration. However, sotorasib (Lumakras, Amgen), which also inhibits KRAS (G12C), was approved in May 2021 by the FDA for KRAS (G12C)–mutated NSCLC. There are some key differences between the drugs. Adagrasib has a half-life of 23 hours versus 5 hours for sotorasib, and the newer drug has the potential to penetrate the central nervous system. That could be an important consideration in NSCLC since it often metastasizes to the brain. “Having pharmacological approaches to treat brain metastases is a wonderful new therapeutic option for lung cancer patients,” said Dr. Jänne, who is director of the Lowe Center for Thoracic Oncology at Dana Farber Cancer Institute, Boston.
Adagrasib is being investigated as part of the KRYSTAL-1 study, alone and as part of combinations in various solid tumors. Previously treated NSCLC KRAS (G12C) patients are also being enrolled in a phase 3 study of adagrasib combined with docetaxel, as well as another phase 2 study of adagrasib combined with pembrolizumab as first-line therapy for NSCLC KRAS (G12C).
Adagrasib is likely to remain a second-line therapy following chemotherapy and immunotherapy. “The activity by itself at the moment is not sufficient to be a first-line treatment. That may change in the future in combination with a standard of care agent or in a subset of patients with KRAS (G12C)–mutant NSCLC, although no subset with higher efficacy has been identified to date. Identification of predictive biomarkers for patients likely to benefit from single agent or an adagrasib combination treatment remains a high priority,” Dr. Jänne said.
The study included 116 patients who had previously been treated with platinum-based chemotherapy and anti–programmed death 1 or programmed death–ligand 1 therapy. They received 600 mg oral adagrasib twice per day over a median follow-up period of 12.9 months. About 42.9% (95% confidence interval, 33.5%-52.6%) experienced a confirmed objective response with a median duration of 8.5 months (95% CI, 6.2-13.8 months). The median progression-free survival was 6.5 months (95% CI, 4.7-8.4 months). After a median follow-up of 15.6 months, the median overall survival was 12.6 months (95% CI, 9.2-19.2 months). The estimated overall survival at 1 year was 50.8% (95% CI, 40.9%-60.0%).
33 patients had stable central nervous system metastases that had been previously treated. About 33.3% had an intracranial confirmed objective response (95% CI, 18.0-51.8%) with a median duration of response of 11.2 months (95% CI, 2.99 months to not available).
Adverse events are similar to what is seen with other targeted therapies, according to Dr. Jänne. 97.4% of patient reported a treatment-related adverse event; 52.6% had grade 1-2 adverse events, and 44.8% had grade 3 adverse events. 6.9% discontinued the drug as a result.
Dr. Jänne has consulted for Mirati Therapeutics and is a member of its scientific advisory board. The study was funded by Mirati Therapeutics.
In a phase 2 cohort study, who had previously been treated with platinum-based chemotherapy and immune therapy.
Adagrasib targets KRAS (G12C), which had long been thought undruggable until research published in 2013 revealed a new binding pocket that did not compete directly against the protein’s natural binding partner. The new trial further validates the approach. “It supports that clinically effective targeted therapies can be developed for patients with KRAS (G12C)–mutant NSCLC,” said Pasi Jänne, MD, PhD, who is the lead author of the study describing the new results published online in the New England Journal of Medicine.
KRAS is the most frequently mutated oncogene in human cancers. A mutated form is found in about 25% of NSCLCs. KRAS plays a key role in cell signaling governing growth, maturation, and cell death. The mutated form is linked to cancer growth and spread. Patients with mutated KRAS have few effective treatment options.
Adagrasib is currently under study and not yet approved by the Food and Drug Administration. However, sotorasib (Lumakras, Amgen), which also inhibits KRAS (G12C), was approved in May 2021 by the FDA for KRAS (G12C)–mutated NSCLC. There are some key differences between the drugs. Adagrasib has a half-life of 23 hours versus 5 hours for sotorasib, and the newer drug has the potential to penetrate the central nervous system. That could be an important consideration in NSCLC since it often metastasizes to the brain. “Having pharmacological approaches to treat brain metastases is a wonderful new therapeutic option for lung cancer patients,” said Dr. Jänne, who is director of the Lowe Center for Thoracic Oncology at Dana Farber Cancer Institute, Boston.
Adagrasib is being investigated as part of the KRYSTAL-1 study, alone and as part of combinations in various solid tumors. Previously treated NSCLC KRAS (G12C) patients are also being enrolled in a phase 3 study of adagrasib combined with docetaxel, as well as another phase 2 study of adagrasib combined with pembrolizumab as first-line therapy for NSCLC KRAS (G12C).
Adagrasib is likely to remain a second-line therapy following chemotherapy and immunotherapy. “The activity by itself at the moment is not sufficient to be a first-line treatment. That may change in the future in combination with a standard of care agent or in a subset of patients with KRAS (G12C)–mutant NSCLC, although no subset with higher efficacy has been identified to date. Identification of predictive biomarkers for patients likely to benefit from single agent or an adagrasib combination treatment remains a high priority,” Dr. Jänne said.
The study included 116 patients who had previously been treated with platinum-based chemotherapy and anti–programmed death 1 or programmed death–ligand 1 therapy. They received 600 mg oral adagrasib twice per day over a median follow-up period of 12.9 months. About 42.9% (95% confidence interval, 33.5%-52.6%) experienced a confirmed objective response with a median duration of 8.5 months (95% CI, 6.2-13.8 months). The median progression-free survival was 6.5 months (95% CI, 4.7-8.4 months). After a median follow-up of 15.6 months, the median overall survival was 12.6 months (95% CI, 9.2-19.2 months). The estimated overall survival at 1 year was 50.8% (95% CI, 40.9%-60.0%).
33 patients had stable central nervous system metastases that had been previously treated. About 33.3% had an intracranial confirmed objective response (95% CI, 18.0-51.8%) with a median duration of response of 11.2 months (95% CI, 2.99 months to not available).
Adverse events are similar to what is seen with other targeted therapies, according to Dr. Jänne. 97.4% of patient reported a treatment-related adverse event; 52.6% had grade 1-2 adverse events, and 44.8% had grade 3 adverse events. 6.9% discontinued the drug as a result.
Dr. Jänne has consulted for Mirati Therapeutics and is a member of its scientific advisory board. The study was funded by Mirati Therapeutics.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE