User login
Clinical Psychiatry News is the online destination and multimedia properties of Clinica Psychiatry News, the independent news publication for psychiatrists. Since 1971, Clinical Psychiatry News has been the leading source of news and commentary about clinical developments in psychiatry as well as health care policy and regulations that affect the physician's practice.
Dear Drupal User: You're seeing this because you're logged in to Drupal, and not redirected to MDedge.com/psychiatry.
Depression
adolescent depression
adolescent major depressive disorder
adolescent schizophrenia
adolescent with major depressive disorder
animals
autism
baby
brexpiprazole
child
child bipolar
child depression
child schizophrenia
children with bipolar disorder
children with depression
children with major depressive disorder
compulsive behaviors
cure
elderly bipolar
elderly depression
elderly major depressive disorder
elderly schizophrenia
elderly with dementia
first break
first episode
gambling
gaming
geriatric depression
geriatric major depressive disorder
geriatric schizophrenia
infant
ketamine
kid
major depressive disorder
major depressive disorder in adolescents
major depressive disorder in children
parenting
pediatric
pediatric bipolar
pediatric depression
pediatric major depressive disorder
pediatric schizophrenia
pregnancy
pregnant
rexulti
skin care
suicide
teen
wine
section[contains(@class, 'nav-hidden')]
footer[@id='footer']
div[contains(@class, 'pane-pub-article-cpn')]
div[contains(@class, 'pane-pub-home-cpn')]
div[contains(@class, 'pane-pub-topic-cpn')]
div[contains(@class, 'panel-panel-inner')]
div[contains(@class, 'pane-node-field-article-topics')]
section[contains(@class, 'footer-nav-section-wrapper')]
‘Stunning’ report shows eating disorders are vastly underestimated
A “stunning” new analysis of global data on eating disorders show that they are far more prevalent and disabling than previously reported.
Investigators found the Global Burden of Diseases, Injuries, and Risk Factors Study (GBD) 2019 underestimated the prevalence of eating disorders by nearly 42 million cases, meaning these disorders are four times more common than previously reported.
“Our work highlights that eating disorders are far more prevalent and disabling than previously quantified,” lead author Damian F. Santomauro, PhD, University of Queensland and Center for Mental Health Research, Brisbane, Australia, said in an interview.
The study was published online March 3 in The Lancet Psychiatry.
Policy implications
The GBD Study 2019 reports the prevalence and burden of anorexia nervosa and bulimia nervosa under the umbrella of “eating disorders.”
However, binge-eating disorder (BED) and other specified feeding or eating disorder (OSFED) are more common, the investigators noted.
By excluding BED and OSFED, 41.9 million cases of eating disorders were not represented in the study.
The researchers calculate that the GBD 2019 overlooked 17.3 million people with BED and 24.6 million people with OSFED.
, bringing the total eating disorder DALYs to 6.6 million in 2019, they reported.
“When disorders are left out of the GBD, there is a risk that policymakers and service planners will interpret that these diseases are not prevalent or disabling and therefore not important to address,” said Dr. Santomauro.
“Our results show that the formal inclusion of binge-eating disorder and OSFED in GBD is both feasible and important and will lead to better representation of eating disorder burden globally.
“In turn, this will enhance recognition of the burden experienced by people living with these disorders and hopefully motivate increased investment in research, prevention, and treatment in future,” he added.
Landmark article, clarion call for action
In an accompanying commentary, Jennifer J. Thomas, PhD, and Kendra R. Becker, PhD, with the Eating Disorders Clinical and Research Program, Massachusetts General Hospital, Boston, said that this “stunning” analysis highlights that eating disorders are four times more common than previously thought.
This “landmark” analysis also demonstrates that BED and OSFED are especially common with increasing age. It highlights the burden of eating disorders in men, “shattering the inaccurate but entrenched stereotype that eating disorders affect only thin, young, White women,” Dr. Thomas and Dr. Becker pointed out.
This article, they wrote, is a “clarion call” for BED and OSFED to be included in future versions of the GBD Study.
Going a step further, Dr. Thomas and Dr. Becker said the GBD Study should also include estimates of the prevalence of avoidant/restrictive food intake disorder, rumination disorder, and pica and that the investigators should obtain direct measures of the disability associated with all feeding and eating disorders included in the DSM-5.
“If they do, the reported global burden will be even greater, underscoring the clear need for increased funding to study, prevent, and treat these debilitating illnesses,” they concluded.
The study was funded by Queensland Health, the Australian National Health and Medical Research Council, and the Bill & Melinda Gates Foundation. The authors have disclosed no relevant financial relationships. Disclosures for the editorialists are listed with the original article.
A version of this article first appeared on Medscape.com.
A “stunning” new analysis of global data on eating disorders show that they are far more prevalent and disabling than previously reported.
Investigators found the Global Burden of Diseases, Injuries, and Risk Factors Study (GBD) 2019 underestimated the prevalence of eating disorders by nearly 42 million cases, meaning these disorders are four times more common than previously reported.
“Our work highlights that eating disorders are far more prevalent and disabling than previously quantified,” lead author Damian F. Santomauro, PhD, University of Queensland and Center for Mental Health Research, Brisbane, Australia, said in an interview.
The study was published online March 3 in The Lancet Psychiatry.
Policy implications
The GBD Study 2019 reports the prevalence and burden of anorexia nervosa and bulimia nervosa under the umbrella of “eating disorders.”
However, binge-eating disorder (BED) and other specified feeding or eating disorder (OSFED) are more common, the investigators noted.
By excluding BED and OSFED, 41.9 million cases of eating disorders were not represented in the study.
The researchers calculate that the GBD 2019 overlooked 17.3 million people with BED and 24.6 million people with OSFED.
, bringing the total eating disorder DALYs to 6.6 million in 2019, they reported.
“When disorders are left out of the GBD, there is a risk that policymakers and service planners will interpret that these diseases are not prevalent or disabling and therefore not important to address,” said Dr. Santomauro.
“Our results show that the formal inclusion of binge-eating disorder and OSFED in GBD is both feasible and important and will lead to better representation of eating disorder burden globally.
“In turn, this will enhance recognition of the burden experienced by people living with these disorders and hopefully motivate increased investment in research, prevention, and treatment in future,” he added.
Landmark article, clarion call for action
In an accompanying commentary, Jennifer J. Thomas, PhD, and Kendra R. Becker, PhD, with the Eating Disorders Clinical and Research Program, Massachusetts General Hospital, Boston, said that this “stunning” analysis highlights that eating disorders are four times more common than previously thought.
This “landmark” analysis also demonstrates that BED and OSFED are especially common with increasing age. It highlights the burden of eating disorders in men, “shattering the inaccurate but entrenched stereotype that eating disorders affect only thin, young, White women,” Dr. Thomas and Dr. Becker pointed out.
This article, they wrote, is a “clarion call” for BED and OSFED to be included in future versions of the GBD Study.
Going a step further, Dr. Thomas and Dr. Becker said the GBD Study should also include estimates of the prevalence of avoidant/restrictive food intake disorder, rumination disorder, and pica and that the investigators should obtain direct measures of the disability associated with all feeding and eating disorders included in the DSM-5.
“If they do, the reported global burden will be even greater, underscoring the clear need for increased funding to study, prevent, and treat these debilitating illnesses,” they concluded.
The study was funded by Queensland Health, the Australian National Health and Medical Research Council, and the Bill & Melinda Gates Foundation. The authors have disclosed no relevant financial relationships. Disclosures for the editorialists are listed with the original article.
A version of this article first appeared on Medscape.com.
A “stunning” new analysis of global data on eating disorders show that they are far more prevalent and disabling than previously reported.
Investigators found the Global Burden of Diseases, Injuries, and Risk Factors Study (GBD) 2019 underestimated the prevalence of eating disorders by nearly 42 million cases, meaning these disorders are four times more common than previously reported.
“Our work highlights that eating disorders are far more prevalent and disabling than previously quantified,” lead author Damian F. Santomauro, PhD, University of Queensland and Center for Mental Health Research, Brisbane, Australia, said in an interview.
The study was published online March 3 in The Lancet Psychiatry.
Policy implications
The GBD Study 2019 reports the prevalence and burden of anorexia nervosa and bulimia nervosa under the umbrella of “eating disorders.”
However, binge-eating disorder (BED) and other specified feeding or eating disorder (OSFED) are more common, the investigators noted.
By excluding BED and OSFED, 41.9 million cases of eating disorders were not represented in the study.
The researchers calculate that the GBD 2019 overlooked 17.3 million people with BED and 24.6 million people with OSFED.
, bringing the total eating disorder DALYs to 6.6 million in 2019, they reported.
“When disorders are left out of the GBD, there is a risk that policymakers and service planners will interpret that these diseases are not prevalent or disabling and therefore not important to address,” said Dr. Santomauro.
“Our results show that the formal inclusion of binge-eating disorder and OSFED in GBD is both feasible and important and will lead to better representation of eating disorder burden globally.
“In turn, this will enhance recognition of the burden experienced by people living with these disorders and hopefully motivate increased investment in research, prevention, and treatment in future,” he added.
Landmark article, clarion call for action
In an accompanying commentary, Jennifer J. Thomas, PhD, and Kendra R. Becker, PhD, with the Eating Disorders Clinical and Research Program, Massachusetts General Hospital, Boston, said that this “stunning” analysis highlights that eating disorders are four times more common than previously thought.
This “landmark” analysis also demonstrates that BED and OSFED are especially common with increasing age. It highlights the burden of eating disorders in men, “shattering the inaccurate but entrenched stereotype that eating disorders affect only thin, young, White women,” Dr. Thomas and Dr. Becker pointed out.
This article, they wrote, is a “clarion call” for BED and OSFED to be included in future versions of the GBD Study.
Going a step further, Dr. Thomas and Dr. Becker said the GBD Study should also include estimates of the prevalence of avoidant/restrictive food intake disorder, rumination disorder, and pica and that the investigators should obtain direct measures of the disability associated with all feeding and eating disorders included in the DSM-5.
“If they do, the reported global burden will be even greater, underscoring the clear need for increased funding to study, prevent, and treat these debilitating illnesses,” they concluded.
The study was funded by Queensland Health, the Australian National Health and Medical Research Council, and the Bill & Melinda Gates Foundation. The authors have disclosed no relevant financial relationships. Disclosures for the editorialists are listed with the original article.
A version of this article first appeared on Medscape.com.
Delay surgery by 7 weeks after COVID-19 diagnosis, study shows
Seven weeks appears to be the ideal amount of time to delay surgery, when possible, after someone tests positive for COVID-19, researchers in the United Kingdom report.
Risk for death was about 3.5 to 4 times higher in the first 6 weeks after surgery among more than 3,000 people with a preoperative COVID-19 diagnosis compared with patients without COVID-19. After 7 weeks, the 30-day mortality rate dropped to a baseline level.
The study was published online March 9 in Anaesthesia.
Surgery should be further delayed for people who remain symptomatic at 7 weeks post diagnosis, lead author Dmitri Nepogodiev, MBChB, said in an interview.
“In this group we recommend waiting until COVID-19 symptoms resolve, if possible. However, our study did not capture specific data on long COVID … so we are unable to make specific recommendations for this group,” said Dr. Nepogodiev, research fellow at the NIHR Global Health Research Unit on Global Surgery at the University of Birmingham (England).
“This should be an area for future research,” he added.
The international, multicenter, prospective cohort study is notable for its sheer size – more than 15,000 investigators reported outcomes for 140,231 surgical patients from 1,674 hospitals across 116 countries. In total, 2.2% of these patients tested positive for SARS-CoV-2 prior to surgery.
Surgery of any type performed in October 2020 was assessed. A greater proportion of patients with a preoperative COVID-19 diagnosis had emergency surgery, 44%, compared with 30% of people who never had a COVID-19 diagnosis.
Most patients were asymptomatic at the time of surgery, either because they never experienced COVID-19 symptoms or their symptoms resolved. The 30-day mortality rate was the primary outcome.
Death rates among surgical patients with preoperative COVID-19 diagnosis
Comparing the timing of surgery after COVID-19 diagnosis vs. 30-day mortality yielded the following results:
- 0 to 2 weeks – 9.1% mortality.
- 3 to 4 weeks – 6.9%.
- 5 to 6 weeks – 5.5%.
- 7 weeks or longer – 2.0%..
For comparison, the 30-day mortality rate for surgical patients without a preoperative COVID-19 diagnosis was 1.4%. A COVID-19 diagnosis more than 7 weeks before surgery did not make a significant difference on outcomes.
The ‘why’ remains unknown
The reasons for the association between a COVID-19 diagnosis and higher postoperative death rates remain unknown. However, Dr. Nepogodiev speculated that it could be related to “some degree of lung injury, even if patients are initially asymptomatic.”
Intubation and mechanical ventilation during surgery could exacerbate the existing lung injury, he said, thereby leading to more severe COVID-19.
In fact, Dr. Nepogodiev and colleagues found that postoperative pulmonary complications followed a pattern similar to the findings on death. They reported higher rates of pneumonia, acute respiratory distress syndrome, and unexpected reventilation in the first 6 weeks following a COVID-19 diagnosis. Again, at 7 weeks and beyond, the rates returned to be relatively the same as those for people who never had COVID-19.
“Waiting for 7 or more weeks may allow time for the initial COVID-19 injury to resolve,” Dr. Nepogodiev said.
‘An important study’
“This is an important study of postoperative mortality among patients recovered from COVID-19,” Adrian Diaz, MD, MPH, said in an interview when asked to comment.
The large cohort and numerous practice settings are among the strengths of the research, said Dr. Diaz, of the University of Michigan Institute for Healthcare Policy and Innovation in Ann Arbor. He was lead author of a June 2020 review article on elective surgery in the time of COVID-19, published in The American Journal of Surgery.
“As with nearly all studies of this nature, results must be interpreted on a case-by-case basis for individual patients. However, this study does add important information for patients and providers in helping them have an informed discussion on the timing of surgery,” said Dr. Diaz, a fellow in the Center for Healthcare Outcomes and Policy and a resident in general surgery at the Ohio State University, Columbus.
Dr. Nepogodiev and colleagues included both urgent and elective surgeries in the study. Dr. Diaz said this was a potential limitation because emergency operations “should never be delayed, by definition.” Lack of indications for the surgeries and information on cause of death were additional limitations.
Future research should evaluate any benefit in delaying surgery longer than 7 or more weeks, Dr. Diaz added, perhaps looking specifically at 10, 12, or 14 weeks, or considering outcomes as a continuous variable. This would help health care providers “garner more insight into risk and benefits of delaying surgery beyond 7 weeks.”
Dr. Nepogodiev and Dr. Diaz disclosed no relevant financial relationships. The study had multiple funding sources, including the National Institute for Health Research Global Health Research Unit, the Association of Upper Gastrointestinal Surgeons, the British Association of Surgical Oncology, and Medtronic.
A version of this article first appeared on Medscape.com.
Seven weeks appears to be the ideal amount of time to delay surgery, when possible, after someone tests positive for COVID-19, researchers in the United Kingdom report.
Risk for death was about 3.5 to 4 times higher in the first 6 weeks after surgery among more than 3,000 people with a preoperative COVID-19 diagnosis compared with patients without COVID-19. After 7 weeks, the 30-day mortality rate dropped to a baseline level.
The study was published online March 9 in Anaesthesia.
Surgery should be further delayed for people who remain symptomatic at 7 weeks post diagnosis, lead author Dmitri Nepogodiev, MBChB, said in an interview.
“In this group we recommend waiting until COVID-19 symptoms resolve, if possible. However, our study did not capture specific data on long COVID … so we are unable to make specific recommendations for this group,” said Dr. Nepogodiev, research fellow at the NIHR Global Health Research Unit on Global Surgery at the University of Birmingham (England).
“This should be an area for future research,” he added.
The international, multicenter, prospective cohort study is notable for its sheer size – more than 15,000 investigators reported outcomes for 140,231 surgical patients from 1,674 hospitals across 116 countries. In total, 2.2% of these patients tested positive for SARS-CoV-2 prior to surgery.
Surgery of any type performed in October 2020 was assessed. A greater proportion of patients with a preoperative COVID-19 diagnosis had emergency surgery, 44%, compared with 30% of people who never had a COVID-19 diagnosis.
Most patients were asymptomatic at the time of surgery, either because they never experienced COVID-19 symptoms or their symptoms resolved. The 30-day mortality rate was the primary outcome.
Death rates among surgical patients with preoperative COVID-19 diagnosis
Comparing the timing of surgery after COVID-19 diagnosis vs. 30-day mortality yielded the following results:
- 0 to 2 weeks – 9.1% mortality.
- 3 to 4 weeks – 6.9%.
- 5 to 6 weeks – 5.5%.
- 7 weeks or longer – 2.0%..
For comparison, the 30-day mortality rate for surgical patients without a preoperative COVID-19 diagnosis was 1.4%. A COVID-19 diagnosis more than 7 weeks before surgery did not make a significant difference on outcomes.
The ‘why’ remains unknown
The reasons for the association between a COVID-19 diagnosis and higher postoperative death rates remain unknown. However, Dr. Nepogodiev speculated that it could be related to “some degree of lung injury, even if patients are initially asymptomatic.”
Intubation and mechanical ventilation during surgery could exacerbate the existing lung injury, he said, thereby leading to more severe COVID-19.
In fact, Dr. Nepogodiev and colleagues found that postoperative pulmonary complications followed a pattern similar to the findings on death. They reported higher rates of pneumonia, acute respiratory distress syndrome, and unexpected reventilation in the first 6 weeks following a COVID-19 diagnosis. Again, at 7 weeks and beyond, the rates returned to be relatively the same as those for people who never had COVID-19.
“Waiting for 7 or more weeks may allow time for the initial COVID-19 injury to resolve,” Dr. Nepogodiev said.
‘An important study’
“This is an important study of postoperative mortality among patients recovered from COVID-19,” Adrian Diaz, MD, MPH, said in an interview when asked to comment.
The large cohort and numerous practice settings are among the strengths of the research, said Dr. Diaz, of the University of Michigan Institute for Healthcare Policy and Innovation in Ann Arbor. He was lead author of a June 2020 review article on elective surgery in the time of COVID-19, published in The American Journal of Surgery.
“As with nearly all studies of this nature, results must be interpreted on a case-by-case basis for individual patients. However, this study does add important information for patients and providers in helping them have an informed discussion on the timing of surgery,” said Dr. Diaz, a fellow in the Center for Healthcare Outcomes and Policy and a resident in general surgery at the Ohio State University, Columbus.
Dr. Nepogodiev and colleagues included both urgent and elective surgeries in the study. Dr. Diaz said this was a potential limitation because emergency operations “should never be delayed, by definition.” Lack of indications for the surgeries and information on cause of death were additional limitations.
Future research should evaluate any benefit in delaying surgery longer than 7 or more weeks, Dr. Diaz added, perhaps looking specifically at 10, 12, or 14 weeks, or considering outcomes as a continuous variable. This would help health care providers “garner more insight into risk and benefits of delaying surgery beyond 7 weeks.”
Dr. Nepogodiev and Dr. Diaz disclosed no relevant financial relationships. The study had multiple funding sources, including the National Institute for Health Research Global Health Research Unit, the Association of Upper Gastrointestinal Surgeons, the British Association of Surgical Oncology, and Medtronic.
A version of this article first appeared on Medscape.com.
Seven weeks appears to be the ideal amount of time to delay surgery, when possible, after someone tests positive for COVID-19, researchers in the United Kingdom report.
Risk for death was about 3.5 to 4 times higher in the first 6 weeks after surgery among more than 3,000 people with a preoperative COVID-19 diagnosis compared with patients without COVID-19. After 7 weeks, the 30-day mortality rate dropped to a baseline level.
The study was published online March 9 in Anaesthesia.
Surgery should be further delayed for people who remain symptomatic at 7 weeks post diagnosis, lead author Dmitri Nepogodiev, MBChB, said in an interview.
“In this group we recommend waiting until COVID-19 symptoms resolve, if possible. However, our study did not capture specific data on long COVID … so we are unable to make specific recommendations for this group,” said Dr. Nepogodiev, research fellow at the NIHR Global Health Research Unit on Global Surgery at the University of Birmingham (England).
“This should be an area for future research,” he added.
The international, multicenter, prospective cohort study is notable for its sheer size – more than 15,000 investigators reported outcomes for 140,231 surgical patients from 1,674 hospitals across 116 countries. In total, 2.2% of these patients tested positive for SARS-CoV-2 prior to surgery.
Surgery of any type performed in October 2020 was assessed. A greater proportion of patients with a preoperative COVID-19 diagnosis had emergency surgery, 44%, compared with 30% of people who never had a COVID-19 diagnosis.
Most patients were asymptomatic at the time of surgery, either because they never experienced COVID-19 symptoms or their symptoms resolved. The 30-day mortality rate was the primary outcome.
Death rates among surgical patients with preoperative COVID-19 diagnosis
Comparing the timing of surgery after COVID-19 diagnosis vs. 30-day mortality yielded the following results:
- 0 to 2 weeks – 9.1% mortality.
- 3 to 4 weeks – 6.9%.
- 5 to 6 weeks – 5.5%.
- 7 weeks or longer – 2.0%..
For comparison, the 30-day mortality rate for surgical patients without a preoperative COVID-19 diagnosis was 1.4%. A COVID-19 diagnosis more than 7 weeks before surgery did not make a significant difference on outcomes.
The ‘why’ remains unknown
The reasons for the association between a COVID-19 diagnosis and higher postoperative death rates remain unknown. However, Dr. Nepogodiev speculated that it could be related to “some degree of lung injury, even if patients are initially asymptomatic.”
Intubation and mechanical ventilation during surgery could exacerbate the existing lung injury, he said, thereby leading to more severe COVID-19.
In fact, Dr. Nepogodiev and colleagues found that postoperative pulmonary complications followed a pattern similar to the findings on death. They reported higher rates of pneumonia, acute respiratory distress syndrome, and unexpected reventilation in the first 6 weeks following a COVID-19 diagnosis. Again, at 7 weeks and beyond, the rates returned to be relatively the same as those for people who never had COVID-19.
“Waiting for 7 or more weeks may allow time for the initial COVID-19 injury to resolve,” Dr. Nepogodiev said.
‘An important study’
“This is an important study of postoperative mortality among patients recovered from COVID-19,” Adrian Diaz, MD, MPH, said in an interview when asked to comment.
The large cohort and numerous practice settings are among the strengths of the research, said Dr. Diaz, of the University of Michigan Institute for Healthcare Policy and Innovation in Ann Arbor. He was lead author of a June 2020 review article on elective surgery in the time of COVID-19, published in The American Journal of Surgery.
“As with nearly all studies of this nature, results must be interpreted on a case-by-case basis for individual patients. However, this study does add important information for patients and providers in helping them have an informed discussion on the timing of surgery,” said Dr. Diaz, a fellow in the Center for Healthcare Outcomes and Policy and a resident in general surgery at the Ohio State University, Columbus.
Dr. Nepogodiev and colleagues included both urgent and elective surgeries in the study. Dr. Diaz said this was a potential limitation because emergency operations “should never be delayed, by definition.” Lack of indications for the surgeries and information on cause of death were additional limitations.
Future research should evaluate any benefit in delaying surgery longer than 7 or more weeks, Dr. Diaz added, perhaps looking specifically at 10, 12, or 14 weeks, or considering outcomes as a continuous variable. This would help health care providers “garner more insight into risk and benefits of delaying surgery beyond 7 weeks.”
Dr. Nepogodiev and Dr. Diaz disclosed no relevant financial relationships. The study had multiple funding sources, including the National Institute for Health Research Global Health Research Unit, the Association of Upper Gastrointestinal Surgeons, the British Association of Surgical Oncology, and Medtronic.
A version of this article first appeared on Medscape.com.
Benefits of bremelanotide to women with HSDD questioned in analysis paper
Dr. Spielmans, professor of psychology at Metropolitan State University in Saint Paul, Minn., examined data from the FDA application for bremelanotide, clinicaltrials.gov entries for two phase 3 trials of the drug, and a 2019 article published in Obstetrics & Gynecology that described results from the 24-week trials.
In Dr. Speilman’s analysis, which was published online March 7 in the Journal of Sex Research, he notes that 42.1% of trial participants who received bremelanotide did not complete the trial, compared with 20.48% of participants who received placebo.
Of those who completed the study, 87.22% who received placebo wanted to continue treatment in an open-label extension, compared with 69.97% who received bremelanotide, he wrote.
Women “should be aware of the small degree of bremelanotide’s efficacy, that the protocol-specified outcomes of bremelanotide are mostly unknown, and that participants would rather take a placebo than bremelanotide,” Dr. Spielmans said.
Anita H. Clayton, MD, an author of the Obstetrics & Gynecology paper addressed in Dr. Spielmans’ analysis, says the Journal of Sex Research article does not provide new information and is a disservice to women because it questions accurate scientific data.
Measuring outcomes in HSDD is an evolving field, Dr. Clayton, a psychiatrist at the University of Virginia in Charlottesville, said in an interview. Initial FDA guidance relied on satisfying sexual events as an outcome measure, but this measure was derived from erectile dysfunction studies and is not necessarily adequate for assessing HSDD, she said. The FDA and drug developers agreed to use the desire subscale of the Female Sexual Function Index (FSFI-D) as a coprimary outcome measure instead, she noted.
Dr. Spielmans’ critique of Obstetrics & Gynecology paper
The article published in Obstetrics & Gynecology reporting bremelanotide trial results was noteworthy, although the various issues involved can be seen in reports about other drug trials, Dr. Spielmans said in an interview.
“It is well-established that journal articles reporting clinical trial data overstate benefits and understate harms,” he continued. In this case, “the very incomplete data reporting, reliance on many post-hoc measures of questionable validity, hiding the concerning number of dropouts due to adverse events, and putting a positive spin on efficacy and tolerability is both remarkable and highly problematic,” Dr. Spielmans said.
Dr. Clayton’s reaction
Data about dropout rates due to adverse events have been reported and presented at national meetings, she said in an interview. In addition, a questionnaire found that bremelanotide was superior to placebo in terms of patients feeling that the treatment had provided clinically meaningful benefit, Dr. Clayton said.
The available information enables patients to make informed treatment decisions, Dr. Clayton continued. “There is really this sexist attitude of women needing protection from their own decisions,” she said.
Diagnosing and treating HSDD
Eight of 11 efficacy outcomes in the clinicaltrials.gov study protocols for bremelanotide were not reported in the Obstetrics & Gynecology article in a way that was consistent with the protocols, Dr. Spielmans said. Changing a coprimary outcome to the key secondary outcome “occurred over a year after the trials had begun,” and the authors of the journal article “did not mention that this change occurred,” Dr. Spielmans wrote.
For the coprimary outcome measures of mean change on FSFI-D and Female Sexual Distress Scale–Desire/Arousal/Orgasm #13, “bremelanotide offers modest benefits over placebo,” Dr. Spielmans reported.
In addition to outlining his concerns about transparency in the reporting of trial data and raising questions about the outcome measures used in the Obstetrics & Gynecology article, Dr. Spielmans wrote that the diagnosis of HSDD is problematic.
“The lack of specifying symptom duration, questionable validity for the lack of sexual fantasies as a diagnostic criterion, difficulty in disentangling individual sexual problems from relational problems, and the failure to consider cultural influence (including the pressure on women to satisfy the sexual desires of their male partners) in the experience of sexuality all render HSDD as a problematic entity,” Dr. Spielmans wrote.
The fifth edition of the Diagnostic and Statistical Manual of Mental Disorders replaced HSDD and female sexual arousal disorder with the combined condition female sexual interest/arousal disorder. HSDD is in the 11th edition of the International Classification of Diseases and can be applied to men or women, Dr. Spielmans said.
FDA acknowledged HSDD as an unmet medical need
Dr. Clayton pointed out that HSDD was described decades ago and the FDA acknowledged it as an unmet medical need, and she expressed dissatisfaction with the fact the hypoactive sexual desire disorder appears with quotation marks around it in the title of Dr. Spielmans’ article. This way of presenting HSDD indicates that “the author has no concept of sexual health or sexual dysfunction,” Dr. Clayton said. “Basically this is sort of a dramatic tool, I think, to act like this is not a real disorder,” she added.
Carl Spana, PhD, CEO and president of Palatin Technologies, the developer of bremelanotide, defined the article in the Journal of Sex Research as a “retrospective meta-analysis, and not a re-analysis of the data.
“As a meta-analysis, it is open to various interpretations and reflects the author’s interpretations, which appear to have clear biases,” Dr. Spana said in an interview. “We believe several of this author’s interpretations are contrary to the FDA’s positive assessment that led to Vyleesi’s approval as a safe and effective treatment for women suffering from hypoactive sexual desire disorder.”
The author is unaware of the validation that was conducted at the direction of the FDA to establish clinically meaningful cutoffs for patient-reported outcomes and to establish metrics that define clinical benefit, Dr. Spana said
“Vyleesi was approved by the FDA after a thorough analysis of data from two well-controlled phase 3 clinical studies and multiple clinical and preclinical safety studies,” he said. “The analyses in the New Drug Application were prespecified and conducted according to a statistical analysis plan that the sponsor and FDA agreed to prior to database lock.”
Dr. Spielmans disclosed holdings in Vanguard Healthcare, a mutual fund that invests in pharmaceutical firms. Dr. Clayton has received financial support from Palatin and AMAG Pharmaceuticals, the companies that developed bremelanotide, in previous years.
Dr. Spielmans, professor of psychology at Metropolitan State University in Saint Paul, Minn., examined data from the FDA application for bremelanotide, clinicaltrials.gov entries for two phase 3 trials of the drug, and a 2019 article published in Obstetrics & Gynecology that described results from the 24-week trials.
In Dr. Speilman’s analysis, which was published online March 7 in the Journal of Sex Research, he notes that 42.1% of trial participants who received bremelanotide did not complete the trial, compared with 20.48% of participants who received placebo.
Of those who completed the study, 87.22% who received placebo wanted to continue treatment in an open-label extension, compared with 69.97% who received bremelanotide, he wrote.
Women “should be aware of the small degree of bremelanotide’s efficacy, that the protocol-specified outcomes of bremelanotide are mostly unknown, and that participants would rather take a placebo than bremelanotide,” Dr. Spielmans said.
Anita H. Clayton, MD, an author of the Obstetrics & Gynecology paper addressed in Dr. Spielmans’ analysis, says the Journal of Sex Research article does not provide new information and is a disservice to women because it questions accurate scientific data.
Measuring outcomes in HSDD is an evolving field, Dr. Clayton, a psychiatrist at the University of Virginia in Charlottesville, said in an interview. Initial FDA guidance relied on satisfying sexual events as an outcome measure, but this measure was derived from erectile dysfunction studies and is not necessarily adequate for assessing HSDD, she said. The FDA and drug developers agreed to use the desire subscale of the Female Sexual Function Index (FSFI-D) as a coprimary outcome measure instead, she noted.
Dr. Spielmans’ critique of Obstetrics & Gynecology paper
The article published in Obstetrics & Gynecology reporting bremelanotide trial results was noteworthy, although the various issues involved can be seen in reports about other drug trials, Dr. Spielmans said in an interview.
“It is well-established that journal articles reporting clinical trial data overstate benefits and understate harms,” he continued. In this case, “the very incomplete data reporting, reliance on many post-hoc measures of questionable validity, hiding the concerning number of dropouts due to adverse events, and putting a positive spin on efficacy and tolerability is both remarkable and highly problematic,” Dr. Spielmans said.
Dr. Clayton’s reaction
Data about dropout rates due to adverse events have been reported and presented at national meetings, she said in an interview. In addition, a questionnaire found that bremelanotide was superior to placebo in terms of patients feeling that the treatment had provided clinically meaningful benefit, Dr. Clayton said.
The available information enables patients to make informed treatment decisions, Dr. Clayton continued. “There is really this sexist attitude of women needing protection from their own decisions,” she said.
Diagnosing and treating HSDD
Eight of 11 efficacy outcomes in the clinicaltrials.gov study protocols for bremelanotide were not reported in the Obstetrics & Gynecology article in a way that was consistent with the protocols, Dr. Spielmans said. Changing a coprimary outcome to the key secondary outcome “occurred over a year after the trials had begun,” and the authors of the journal article “did not mention that this change occurred,” Dr. Spielmans wrote.
For the coprimary outcome measures of mean change on FSFI-D and Female Sexual Distress Scale–Desire/Arousal/Orgasm #13, “bremelanotide offers modest benefits over placebo,” Dr. Spielmans reported.
In addition to outlining his concerns about transparency in the reporting of trial data and raising questions about the outcome measures used in the Obstetrics & Gynecology article, Dr. Spielmans wrote that the diagnosis of HSDD is problematic.
“The lack of specifying symptom duration, questionable validity for the lack of sexual fantasies as a diagnostic criterion, difficulty in disentangling individual sexual problems from relational problems, and the failure to consider cultural influence (including the pressure on women to satisfy the sexual desires of their male partners) in the experience of sexuality all render HSDD as a problematic entity,” Dr. Spielmans wrote.
The fifth edition of the Diagnostic and Statistical Manual of Mental Disorders replaced HSDD and female sexual arousal disorder with the combined condition female sexual interest/arousal disorder. HSDD is in the 11th edition of the International Classification of Diseases and can be applied to men or women, Dr. Spielmans said.
FDA acknowledged HSDD as an unmet medical need
Dr. Clayton pointed out that HSDD was described decades ago and the FDA acknowledged it as an unmet medical need, and she expressed dissatisfaction with the fact the hypoactive sexual desire disorder appears with quotation marks around it in the title of Dr. Spielmans’ article. This way of presenting HSDD indicates that “the author has no concept of sexual health or sexual dysfunction,” Dr. Clayton said. “Basically this is sort of a dramatic tool, I think, to act like this is not a real disorder,” she added.
Carl Spana, PhD, CEO and president of Palatin Technologies, the developer of bremelanotide, defined the article in the Journal of Sex Research as a “retrospective meta-analysis, and not a re-analysis of the data.
“As a meta-analysis, it is open to various interpretations and reflects the author’s interpretations, which appear to have clear biases,” Dr. Spana said in an interview. “We believe several of this author’s interpretations are contrary to the FDA’s positive assessment that led to Vyleesi’s approval as a safe and effective treatment for women suffering from hypoactive sexual desire disorder.”
The author is unaware of the validation that was conducted at the direction of the FDA to establish clinically meaningful cutoffs for patient-reported outcomes and to establish metrics that define clinical benefit, Dr. Spana said
“Vyleesi was approved by the FDA after a thorough analysis of data from two well-controlled phase 3 clinical studies and multiple clinical and preclinical safety studies,” he said. “The analyses in the New Drug Application were prespecified and conducted according to a statistical analysis plan that the sponsor and FDA agreed to prior to database lock.”
Dr. Spielmans disclosed holdings in Vanguard Healthcare, a mutual fund that invests in pharmaceutical firms. Dr. Clayton has received financial support from Palatin and AMAG Pharmaceuticals, the companies that developed bremelanotide, in previous years.
Dr. Spielmans, professor of psychology at Metropolitan State University in Saint Paul, Minn., examined data from the FDA application for bremelanotide, clinicaltrials.gov entries for two phase 3 trials of the drug, and a 2019 article published in Obstetrics & Gynecology that described results from the 24-week trials.
In Dr. Speilman’s analysis, which was published online March 7 in the Journal of Sex Research, he notes that 42.1% of trial participants who received bremelanotide did not complete the trial, compared with 20.48% of participants who received placebo.
Of those who completed the study, 87.22% who received placebo wanted to continue treatment in an open-label extension, compared with 69.97% who received bremelanotide, he wrote.
Women “should be aware of the small degree of bremelanotide’s efficacy, that the protocol-specified outcomes of bremelanotide are mostly unknown, and that participants would rather take a placebo than bremelanotide,” Dr. Spielmans said.
Anita H. Clayton, MD, an author of the Obstetrics & Gynecology paper addressed in Dr. Spielmans’ analysis, says the Journal of Sex Research article does not provide new information and is a disservice to women because it questions accurate scientific data.
Measuring outcomes in HSDD is an evolving field, Dr. Clayton, a psychiatrist at the University of Virginia in Charlottesville, said in an interview. Initial FDA guidance relied on satisfying sexual events as an outcome measure, but this measure was derived from erectile dysfunction studies and is not necessarily adequate for assessing HSDD, she said. The FDA and drug developers agreed to use the desire subscale of the Female Sexual Function Index (FSFI-D) as a coprimary outcome measure instead, she noted.
Dr. Spielmans’ critique of Obstetrics & Gynecology paper
The article published in Obstetrics & Gynecology reporting bremelanotide trial results was noteworthy, although the various issues involved can be seen in reports about other drug trials, Dr. Spielmans said in an interview.
“It is well-established that journal articles reporting clinical trial data overstate benefits and understate harms,” he continued. In this case, “the very incomplete data reporting, reliance on many post-hoc measures of questionable validity, hiding the concerning number of dropouts due to adverse events, and putting a positive spin on efficacy and tolerability is both remarkable and highly problematic,” Dr. Spielmans said.
Dr. Clayton’s reaction
Data about dropout rates due to adverse events have been reported and presented at national meetings, she said in an interview. In addition, a questionnaire found that bremelanotide was superior to placebo in terms of patients feeling that the treatment had provided clinically meaningful benefit, Dr. Clayton said.
The available information enables patients to make informed treatment decisions, Dr. Clayton continued. “There is really this sexist attitude of women needing protection from their own decisions,” she said.
Diagnosing and treating HSDD
Eight of 11 efficacy outcomes in the clinicaltrials.gov study protocols for bremelanotide were not reported in the Obstetrics & Gynecology article in a way that was consistent with the protocols, Dr. Spielmans said. Changing a coprimary outcome to the key secondary outcome “occurred over a year after the trials had begun,” and the authors of the journal article “did not mention that this change occurred,” Dr. Spielmans wrote.
For the coprimary outcome measures of mean change on FSFI-D and Female Sexual Distress Scale–Desire/Arousal/Orgasm #13, “bremelanotide offers modest benefits over placebo,” Dr. Spielmans reported.
In addition to outlining his concerns about transparency in the reporting of trial data and raising questions about the outcome measures used in the Obstetrics & Gynecology article, Dr. Spielmans wrote that the diagnosis of HSDD is problematic.
“The lack of specifying symptom duration, questionable validity for the lack of sexual fantasies as a diagnostic criterion, difficulty in disentangling individual sexual problems from relational problems, and the failure to consider cultural influence (including the pressure on women to satisfy the sexual desires of their male partners) in the experience of sexuality all render HSDD as a problematic entity,” Dr. Spielmans wrote.
The fifth edition of the Diagnostic and Statistical Manual of Mental Disorders replaced HSDD and female sexual arousal disorder with the combined condition female sexual interest/arousal disorder. HSDD is in the 11th edition of the International Classification of Diseases and can be applied to men or women, Dr. Spielmans said.
FDA acknowledged HSDD as an unmet medical need
Dr. Clayton pointed out that HSDD was described decades ago and the FDA acknowledged it as an unmet medical need, and she expressed dissatisfaction with the fact the hypoactive sexual desire disorder appears with quotation marks around it in the title of Dr. Spielmans’ article. This way of presenting HSDD indicates that “the author has no concept of sexual health or sexual dysfunction,” Dr. Clayton said. “Basically this is sort of a dramatic tool, I think, to act like this is not a real disorder,” she added.
Carl Spana, PhD, CEO and president of Palatin Technologies, the developer of bremelanotide, defined the article in the Journal of Sex Research as a “retrospective meta-analysis, and not a re-analysis of the data.
“As a meta-analysis, it is open to various interpretations and reflects the author’s interpretations, which appear to have clear biases,” Dr. Spana said in an interview. “We believe several of this author’s interpretations are contrary to the FDA’s positive assessment that led to Vyleesi’s approval as a safe and effective treatment for women suffering from hypoactive sexual desire disorder.”
The author is unaware of the validation that was conducted at the direction of the FDA to establish clinically meaningful cutoffs for patient-reported outcomes and to establish metrics that define clinical benefit, Dr. Spana said
“Vyleesi was approved by the FDA after a thorough analysis of data from two well-controlled phase 3 clinical studies and multiple clinical and preclinical safety studies,” he said. “The analyses in the New Drug Application were prespecified and conducted according to a statistical analysis plan that the sponsor and FDA agreed to prior to database lock.”
Dr. Spielmans disclosed holdings in Vanguard Healthcare, a mutual fund that invests in pharmaceutical firms. Dr. Clayton has received financial support from Palatin and AMAG Pharmaceuticals, the companies that developed bremelanotide, in previous years.
FROM THE JOURNAL OF SEX RESEARCH
Novel therapeutic target for depression identified
An antiseizure medication appears to reduce anhedonia in patients with depression via a novel mechanism that may offer a new therapeutic target for the disorder, new research suggests.
Results of a small, randomized trial show those who received ezogabine (Potiga) experienced a significant reduction in key measures of depression and anhedonia versus placebo.
Participants in the treatment group also showed a trend toward increased response to reward anticipation on functional MRI (fMRI), compared with those treated with placebo, although the effect did not reach statistical significance.
“Our study was the first randomized, placebo-controlled trial to show that a drug affecting this kind of ion channel in the brain can improve depression and anhedonia in patients,” senior investigator James Murrough, MD, PhD, associate professor of psychiatry and neuroscience at the Icahn School of Medicine at Mount Sinai, New York, said in a press release.
“Targeting this channel represents a completely different mechanism of action than any currently available antidepressant treatment,” said Dr. Murrough, who is also director of the Depression and Anxiety Center for Discovery and Treatment at Mount Sinai.
The study was published online March 3 in the American Journal of Psychiatry.
Need for a novel target
“One of the main issues in treating depression is that many of our current antidepressants have similar mechanisms of action,” Dr. Murrough said in an interview. “Once a patient hasn’t responded to currently available agents, it’s hard think of new medications to fill that need.”
This need for a novel target motivated Dr. Murrough and associates to research the KCNQ2/3 potassium channel, which has not been previously studied as a therapeutic target for depression.
The KCNQ2/3 channel controls brain cell excitability and function by controlling the flow of the electrical charge across the cell membrane in the form of potassium ions, Dr. Murrough explained.
Previous research using a chronic social defeat model of depression in mice showed changes in the KCNQ2/3 channel. “This was key to determining whether a mouse showed depressive behavior in the context of stress, or whether the mice were resistant or resilient to stress,” he said.
Mice resistant to stress showed increased markers in brain regions associated with reward, while the less resilient mice showed excessive excitability and dysfunction. Dysfunction in the brain’s reward system leads to anhedonia, a “core feature” of depressive disorders.
This inspired Dr. Murrough’s group to identify ezogabine, a drug that acts on this channel.
“ for addressing depressive symptoms,” Dr. Murrough said
Nonsignificant trend
The researchers studied 45 adults diagnosed with depression who exhibited significant anhedonia and at least moderate illness severity.
Participants were randomly assigned to receive either ezogabine (n = 21, mean age 44 years at enrollment, 28.3% male) or placebo (n = 24, mean age 39 years at enrollment, 50% male). At baseline and following treatment, participants completed the incentive flanker test under fMRI conditions to model brain activity during anticipation of a reward. In addition, clinical measures of depression and anhedonia were assessed at weekly visits.
The study groups did not differ significantly in performance accuracy during the fMRI task at baseline or following treatment. The table below summarizes the percentage of errors in each group, with standard deviation.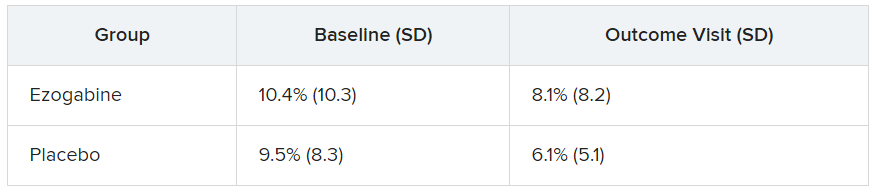
Participants in the ezogabine group showed a numerical increase in ventral striatum activation in response to reward anticipation, compared with participants in the placebo group, but this trend was not considered significant.
Heterogeneous condition
In contrast, there were notable improvements from baseline to the final outcome visit in clinical measures of depression and anhedonia in the ezogabine group, compared with the placebo group. Mean (SD) differences in depression scores, based on the MADRS (Montgomery-Åsberg Depression Rating Scale) from baseline to endpoint as follows: mean difference, –7.9 (3.0); effect size, 0.76; response rate, 61.9% (ezogabine) and 37.5% (placebo); remission rate: 38.1% (ezogabine) and 20.8% (placebo)
Compared with placebo, there were also large improvements in hedonic capacity, as measured by the Snaith-Hamilton Pleasure Scale and the anticipatory subscale of the Temporal Experience of Pleasure Scale (t, –4.1; df, 212; P < .001 and t, 3.4; df, 213; P < .001, respectively).
Compared with placebo, ezogabine was associated with “significant benefit” in global illness severity and improvement (Clinical Global Impression–Severity: t, –2.2; df, 214; P = .026 and CGI-Improvement: t, –2.9; d, 214; P = .004, respectively).
Ezogabine was well tolerated. Dizziness and headache were the most common adverse events.
Depression is a “heterogeneous condition” with a single diagnosis encompassing a “large, multifaceted” array of symptoms, Dr. Murrough noted. A growing body of research is focusing on specific components as potential treatment targets. “Our study looked specifically at patients with a diagnosis of depression but high scores on the anhedonia scale and we found that boosting the function of the KCNQ2/3 channel may have a beneficial antidepressant effect by improving anhedonia.”
Potential gain
In a comment, Alan Schatzberg, MD, professor in the department of psychiatry and behavioral sciences at Stanford (Calif.) University, said that “anytime there’s a new treatment with a new mechanism of action for a given condition, there’s a potential gain for the field.”
Dr. Schatzberg, who was not involved with the study, said that ezogabine, with its “potentially new mechanism of action, seems to have an effect and reasonable safety and could be important for patients who may not respond to traditional medications. It might also be important for all sorts of patients, depending on findings of later trials.”
Dr. Murrough said that ezogabine in still in “early stages” of research. “We hope that future studies will look at other agents that would also affect this channel,” he added.
This research was supported by the National Institute of Mental Health. Additional funding was provided by the Friedman Brain Institute at the Icahn School of Medicine at Mount Sinai and the Ehrenkranz Laboratory for Human Resilience, a component of the Depression and Anxiety Center for Discovery and Treatment at the Icahn School of Medicine at Mount Sinai. Dr. Murrough is an inventor of a pending patent application for the use of ezogabine and other KCNQ channel openers to treat depression and related disorders. Dr. Schatzberg disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
An antiseizure medication appears to reduce anhedonia in patients with depression via a novel mechanism that may offer a new therapeutic target for the disorder, new research suggests.
Results of a small, randomized trial show those who received ezogabine (Potiga) experienced a significant reduction in key measures of depression and anhedonia versus placebo.
Participants in the treatment group also showed a trend toward increased response to reward anticipation on functional MRI (fMRI), compared with those treated with placebo, although the effect did not reach statistical significance.
“Our study was the first randomized, placebo-controlled trial to show that a drug affecting this kind of ion channel in the brain can improve depression and anhedonia in patients,” senior investigator James Murrough, MD, PhD, associate professor of psychiatry and neuroscience at the Icahn School of Medicine at Mount Sinai, New York, said in a press release.
“Targeting this channel represents a completely different mechanism of action than any currently available antidepressant treatment,” said Dr. Murrough, who is also director of the Depression and Anxiety Center for Discovery and Treatment at Mount Sinai.
The study was published online March 3 in the American Journal of Psychiatry.
Need for a novel target
“One of the main issues in treating depression is that many of our current antidepressants have similar mechanisms of action,” Dr. Murrough said in an interview. “Once a patient hasn’t responded to currently available agents, it’s hard think of new medications to fill that need.”
This need for a novel target motivated Dr. Murrough and associates to research the KCNQ2/3 potassium channel, which has not been previously studied as a therapeutic target for depression.
The KCNQ2/3 channel controls brain cell excitability and function by controlling the flow of the electrical charge across the cell membrane in the form of potassium ions, Dr. Murrough explained.
Previous research using a chronic social defeat model of depression in mice showed changes in the KCNQ2/3 channel. “This was key to determining whether a mouse showed depressive behavior in the context of stress, or whether the mice were resistant or resilient to stress,” he said.
Mice resistant to stress showed increased markers in brain regions associated with reward, while the less resilient mice showed excessive excitability and dysfunction. Dysfunction in the brain’s reward system leads to anhedonia, a “core feature” of depressive disorders.
This inspired Dr. Murrough’s group to identify ezogabine, a drug that acts on this channel.
“ for addressing depressive symptoms,” Dr. Murrough said
Nonsignificant trend
The researchers studied 45 adults diagnosed with depression who exhibited significant anhedonia and at least moderate illness severity.
Participants were randomly assigned to receive either ezogabine (n = 21, mean age 44 years at enrollment, 28.3% male) or placebo (n = 24, mean age 39 years at enrollment, 50% male). At baseline and following treatment, participants completed the incentive flanker test under fMRI conditions to model brain activity during anticipation of a reward. In addition, clinical measures of depression and anhedonia were assessed at weekly visits.
The study groups did not differ significantly in performance accuracy during the fMRI task at baseline or following treatment. The table below summarizes the percentage of errors in each group, with standard deviation.
Participants in the ezogabine group showed a numerical increase in ventral striatum activation in response to reward anticipation, compared with participants in the placebo group, but this trend was not considered significant.
Heterogeneous condition
In contrast, there were notable improvements from baseline to the final outcome visit in clinical measures of depression and anhedonia in the ezogabine group, compared with the placebo group. Mean (SD) differences in depression scores, based on the MADRS (Montgomery-Åsberg Depression Rating Scale) from baseline to endpoint as follows: mean difference, –7.9 (3.0); effect size, 0.76; response rate, 61.9% (ezogabine) and 37.5% (placebo); remission rate: 38.1% (ezogabine) and 20.8% (placebo)
Compared with placebo, there were also large improvements in hedonic capacity, as measured by the Snaith-Hamilton Pleasure Scale and the anticipatory subscale of the Temporal Experience of Pleasure Scale (t, –4.1; df, 212; P < .001 and t, 3.4; df, 213; P < .001, respectively).
Compared with placebo, ezogabine was associated with “significant benefit” in global illness severity and improvement (Clinical Global Impression–Severity: t, –2.2; df, 214; P = .026 and CGI-Improvement: t, –2.9; d, 214; P = .004, respectively).
Ezogabine was well tolerated. Dizziness and headache were the most common adverse events.
Depression is a “heterogeneous condition” with a single diagnosis encompassing a “large, multifaceted” array of symptoms, Dr. Murrough noted. A growing body of research is focusing on specific components as potential treatment targets. “Our study looked specifically at patients with a diagnosis of depression but high scores on the anhedonia scale and we found that boosting the function of the KCNQ2/3 channel may have a beneficial antidepressant effect by improving anhedonia.”
Potential gain
In a comment, Alan Schatzberg, MD, professor in the department of psychiatry and behavioral sciences at Stanford (Calif.) University, said that “anytime there’s a new treatment with a new mechanism of action for a given condition, there’s a potential gain for the field.”
Dr. Schatzberg, who was not involved with the study, said that ezogabine, with its “potentially new mechanism of action, seems to have an effect and reasonable safety and could be important for patients who may not respond to traditional medications. It might also be important for all sorts of patients, depending on findings of later trials.”
Dr. Murrough said that ezogabine in still in “early stages” of research. “We hope that future studies will look at other agents that would also affect this channel,” he added.
This research was supported by the National Institute of Mental Health. Additional funding was provided by the Friedman Brain Institute at the Icahn School of Medicine at Mount Sinai and the Ehrenkranz Laboratory for Human Resilience, a component of the Depression and Anxiety Center for Discovery and Treatment at the Icahn School of Medicine at Mount Sinai. Dr. Murrough is an inventor of a pending patent application for the use of ezogabine and other KCNQ channel openers to treat depression and related disorders. Dr. Schatzberg disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
An antiseizure medication appears to reduce anhedonia in patients with depression via a novel mechanism that may offer a new therapeutic target for the disorder, new research suggests.
Results of a small, randomized trial show those who received ezogabine (Potiga) experienced a significant reduction in key measures of depression and anhedonia versus placebo.
Participants in the treatment group also showed a trend toward increased response to reward anticipation on functional MRI (fMRI), compared with those treated with placebo, although the effect did not reach statistical significance.
“Our study was the first randomized, placebo-controlled trial to show that a drug affecting this kind of ion channel in the brain can improve depression and anhedonia in patients,” senior investigator James Murrough, MD, PhD, associate professor of psychiatry and neuroscience at the Icahn School of Medicine at Mount Sinai, New York, said in a press release.
“Targeting this channel represents a completely different mechanism of action than any currently available antidepressant treatment,” said Dr. Murrough, who is also director of the Depression and Anxiety Center for Discovery and Treatment at Mount Sinai.
The study was published online March 3 in the American Journal of Psychiatry.
Need for a novel target
“One of the main issues in treating depression is that many of our current antidepressants have similar mechanisms of action,” Dr. Murrough said in an interview. “Once a patient hasn’t responded to currently available agents, it’s hard think of new medications to fill that need.”
This need for a novel target motivated Dr. Murrough and associates to research the KCNQ2/3 potassium channel, which has not been previously studied as a therapeutic target for depression.
The KCNQ2/3 channel controls brain cell excitability and function by controlling the flow of the electrical charge across the cell membrane in the form of potassium ions, Dr. Murrough explained.
Previous research using a chronic social defeat model of depression in mice showed changes in the KCNQ2/3 channel. “This was key to determining whether a mouse showed depressive behavior in the context of stress, or whether the mice were resistant or resilient to stress,” he said.
Mice resistant to stress showed increased markers in brain regions associated with reward, while the less resilient mice showed excessive excitability and dysfunction. Dysfunction in the brain’s reward system leads to anhedonia, a “core feature” of depressive disorders.
This inspired Dr. Murrough’s group to identify ezogabine, a drug that acts on this channel.
“ for addressing depressive symptoms,” Dr. Murrough said
Nonsignificant trend
The researchers studied 45 adults diagnosed with depression who exhibited significant anhedonia and at least moderate illness severity.
Participants were randomly assigned to receive either ezogabine (n = 21, mean age 44 years at enrollment, 28.3% male) or placebo (n = 24, mean age 39 years at enrollment, 50% male). At baseline and following treatment, participants completed the incentive flanker test under fMRI conditions to model brain activity during anticipation of a reward. In addition, clinical measures of depression and anhedonia were assessed at weekly visits.
The study groups did not differ significantly in performance accuracy during the fMRI task at baseline or following treatment. The table below summarizes the percentage of errors in each group, with standard deviation.
Participants in the ezogabine group showed a numerical increase in ventral striatum activation in response to reward anticipation, compared with participants in the placebo group, but this trend was not considered significant.
Heterogeneous condition
In contrast, there were notable improvements from baseline to the final outcome visit in clinical measures of depression and anhedonia in the ezogabine group, compared with the placebo group. Mean (SD) differences in depression scores, based on the MADRS (Montgomery-Åsberg Depression Rating Scale) from baseline to endpoint as follows: mean difference, –7.9 (3.0); effect size, 0.76; response rate, 61.9% (ezogabine) and 37.5% (placebo); remission rate: 38.1% (ezogabine) and 20.8% (placebo)
Compared with placebo, there were also large improvements in hedonic capacity, as measured by the Snaith-Hamilton Pleasure Scale and the anticipatory subscale of the Temporal Experience of Pleasure Scale (t, –4.1; df, 212; P < .001 and t, 3.4; df, 213; P < .001, respectively).
Compared with placebo, ezogabine was associated with “significant benefit” in global illness severity and improvement (Clinical Global Impression–Severity: t, –2.2; df, 214; P = .026 and CGI-Improvement: t, –2.9; d, 214; P = .004, respectively).
Ezogabine was well tolerated. Dizziness and headache were the most common adverse events.
Depression is a “heterogeneous condition” with a single diagnosis encompassing a “large, multifaceted” array of symptoms, Dr. Murrough noted. A growing body of research is focusing on specific components as potential treatment targets. “Our study looked specifically at patients with a diagnosis of depression but high scores on the anhedonia scale and we found that boosting the function of the KCNQ2/3 channel may have a beneficial antidepressant effect by improving anhedonia.”
Potential gain
In a comment, Alan Schatzberg, MD, professor in the department of psychiatry and behavioral sciences at Stanford (Calif.) University, said that “anytime there’s a new treatment with a new mechanism of action for a given condition, there’s a potential gain for the field.”
Dr. Schatzberg, who was not involved with the study, said that ezogabine, with its “potentially new mechanism of action, seems to have an effect and reasonable safety and could be important for patients who may not respond to traditional medications. It might also be important for all sorts of patients, depending on findings of later trials.”
Dr. Murrough said that ezogabine in still in “early stages” of research. “We hope that future studies will look at other agents that would also affect this channel,” he added.
This research was supported by the National Institute of Mental Health. Additional funding was provided by the Friedman Brain Institute at the Icahn School of Medicine at Mount Sinai and the Ehrenkranz Laboratory for Human Resilience, a component of the Depression and Anxiety Center for Discovery and Treatment at the Icahn School of Medicine at Mount Sinai. Dr. Murrough is an inventor of a pending patent application for the use of ezogabine and other KCNQ channel openers to treat depression and related disorders. Dr. Schatzberg disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Palliative care for patients with dementia: When to refer?
Palliative care for people with dementia is increasingly recognized as a way to improve quality of life and provide relief from the myriad physical and psychological symptoms of advancing neurodegenerative disease. But unlike in cancer,
A new literature review has found these referrals to be all over the map among patients with dementia – with many occurring very late in the disease process – and do not reflect any consistent criteria based on patient needs.
For their research, published March 2 in the Journal of the American Geriatrics Society, Li Mo, MD, of the University of Texas MD Anderson Cancer Center in Houston, and colleagues looked at nearly 60 studies dating back to the early 1990s that contained information on referrals to palliative care for patients with dementia. While a palliative care approach can be provided by nonspecialists, all the included studies dealt at least in part with specialist care.
Standardized criteria is lacking
The investigators found advanced or late-stage dementia to be the most common reason cited for referral, with three quarters of the studies recommending palliative care for late-stage or advanced dementia, generally without qualifying what symptoms or needs were present. Patients received palliative care across a range of settings, including nursing homes, hospitals, and their own homes, though many articles did not include information on where patients received care.
A fifth of the articles suggested that medical complications of dementia including falls, pneumonia, and ulcers should trigger referrals to palliative care, while another fifth cited poor prognosis, defined varyingly as having between 2 years and 6 months likely left to live. Poor nutrition status was identified in 10% of studies as meriting referral.
Only 20% of the studies identified patient needs – evidence of psychological distress or functional decline, for example – as criteria for referral, despite these being ubiquitous in dementia. The authors said they were surprised by this finding, which could possibly be explained, they wrote, by “the interest among geriatrician, neurologist, and primary care teams to provide good symptom management,” reflecting a de facto palliative care approach. “There is also significant stigma associated with a specialist palliative care referral,” the authors noted.
Curiously, the researchers noted, a new diagnosis of dementia in more than a quarter of the studies triggered referral, a finding that possibly reflected delayed diagnoses.
The findings revealed “heterogeneity in the literature in reasons for involving specialist palliative care, which may partly explain the variation in patterns of palliative care referral,” Dr. Mo and colleagues wrote, stressing that more standardized criteria are urgently needed to bring dementia in line with cancer in terms of providing timely palliative care.
Patients with advancing dementia have little chance to self-report symptoms, meaning that more attention to patient complaints earlier in the disease course, and greater sensitivity to patient distress, are required. By routinely screening symptoms, clinicians could use specific cutoffs “as triggers to initiate automatic timely palliative care referral,” the authors concluded, noting that more research was needed before these cutoffs, whether based on symptom intensity or other measures, could be calculated.
Dr. Mo and colleagues acknowledged as weaknesses of their study the fact that a third of the articles in the review were based on expert consensus, while others did not distinguish clearly between primary and specialist palliative care.
A starting point for further discussion
Asked to comment on the findings, Elizabeth Sampson, MD, a palliative care researcher at University College London, praised Dr. Mo and colleagues’ study as “starting to pull together the strands” of a systematic approach to referrals and access to palliative care in dementia.
“Sometimes you need a paper like this to kick off the discussion to say look, this is where we are,” Dr. Sampson said, noting that the focus on need-based criteria dovetailed with a “general feeling in the field that we need to really think about needs, and what palliative care needs might be. What the threshold for referral should be we don’t know yet. Should it be three unmet needs? Or five? We’re still a long way from knowing.”
Dr. Sampson’s group is leading a UK-government funded research effort that aims to develop cost-effective palliative care interventions in dementia, in part through a tool that uses caregiver reports to assess symptom burden and patient needs. The research program “is founded on a needs-based approach, which aims to look at people’s individual needs and responding to them in a proactive way,” she said.
One of the obstacles to timely palliative care in dementia, Dr. Sampson said, is weighing resource allocation against what can be wildly varying prognoses. “Hospices understand when someone has terminal cancer and [is] likely to die within a few weeks, but it’s not unheard of for someone in very advanced stages of dementia to live another year,” she said. “There are concerns that a rapid increase in people with dementia being moved to palliative care could overwhelm already limited hospice capacity. We would argue that the best approach is to get palliative care out to where people with dementia live, which is usually the care home.”
Dr. Mo and colleagues’ study received funding from the National Institutes of Health, and its authors disclosed no financial conflicts of interest. Dr. Sampson’s work is supported by the UK’s Economic and Social Research Council and National Institute for Health Research. She disclosed no conflicts of interest.
Palliative care for people with dementia is increasingly recognized as a way to improve quality of life and provide relief from the myriad physical and psychological symptoms of advancing neurodegenerative disease. But unlike in cancer,
A new literature review has found these referrals to be all over the map among patients with dementia – with many occurring very late in the disease process – and do not reflect any consistent criteria based on patient needs.
For their research, published March 2 in the Journal of the American Geriatrics Society, Li Mo, MD, of the University of Texas MD Anderson Cancer Center in Houston, and colleagues looked at nearly 60 studies dating back to the early 1990s that contained information on referrals to palliative care for patients with dementia. While a palliative care approach can be provided by nonspecialists, all the included studies dealt at least in part with specialist care.
Standardized criteria is lacking
The investigators found advanced or late-stage dementia to be the most common reason cited for referral, with three quarters of the studies recommending palliative care for late-stage or advanced dementia, generally without qualifying what symptoms or needs were present. Patients received palliative care across a range of settings, including nursing homes, hospitals, and their own homes, though many articles did not include information on where patients received care.
A fifth of the articles suggested that medical complications of dementia including falls, pneumonia, and ulcers should trigger referrals to palliative care, while another fifth cited poor prognosis, defined varyingly as having between 2 years and 6 months likely left to live. Poor nutrition status was identified in 10% of studies as meriting referral.
Only 20% of the studies identified patient needs – evidence of psychological distress or functional decline, for example – as criteria for referral, despite these being ubiquitous in dementia. The authors said they were surprised by this finding, which could possibly be explained, they wrote, by “the interest among geriatrician, neurologist, and primary care teams to provide good symptom management,” reflecting a de facto palliative care approach. “There is also significant stigma associated with a specialist palliative care referral,” the authors noted.
Curiously, the researchers noted, a new diagnosis of dementia in more than a quarter of the studies triggered referral, a finding that possibly reflected delayed diagnoses.
The findings revealed “heterogeneity in the literature in reasons for involving specialist palliative care, which may partly explain the variation in patterns of palliative care referral,” Dr. Mo and colleagues wrote, stressing that more standardized criteria are urgently needed to bring dementia in line with cancer in terms of providing timely palliative care.
Patients with advancing dementia have little chance to self-report symptoms, meaning that more attention to patient complaints earlier in the disease course, and greater sensitivity to patient distress, are required. By routinely screening symptoms, clinicians could use specific cutoffs “as triggers to initiate automatic timely palliative care referral,” the authors concluded, noting that more research was needed before these cutoffs, whether based on symptom intensity or other measures, could be calculated.
Dr. Mo and colleagues acknowledged as weaknesses of their study the fact that a third of the articles in the review were based on expert consensus, while others did not distinguish clearly between primary and specialist palliative care.
A starting point for further discussion
Asked to comment on the findings, Elizabeth Sampson, MD, a palliative care researcher at University College London, praised Dr. Mo and colleagues’ study as “starting to pull together the strands” of a systematic approach to referrals and access to palliative care in dementia.
“Sometimes you need a paper like this to kick off the discussion to say look, this is where we are,” Dr. Sampson said, noting that the focus on need-based criteria dovetailed with a “general feeling in the field that we need to really think about needs, and what palliative care needs might be. What the threshold for referral should be we don’t know yet. Should it be three unmet needs? Or five? We’re still a long way from knowing.”
Dr. Sampson’s group is leading a UK-government funded research effort that aims to develop cost-effective palliative care interventions in dementia, in part through a tool that uses caregiver reports to assess symptom burden and patient needs. The research program “is founded on a needs-based approach, which aims to look at people’s individual needs and responding to them in a proactive way,” she said.
One of the obstacles to timely palliative care in dementia, Dr. Sampson said, is weighing resource allocation against what can be wildly varying prognoses. “Hospices understand when someone has terminal cancer and [is] likely to die within a few weeks, but it’s not unheard of for someone in very advanced stages of dementia to live another year,” she said. “There are concerns that a rapid increase in people with dementia being moved to palliative care could overwhelm already limited hospice capacity. We would argue that the best approach is to get palliative care out to where people with dementia live, which is usually the care home.”
Dr. Mo and colleagues’ study received funding from the National Institutes of Health, and its authors disclosed no financial conflicts of interest. Dr. Sampson’s work is supported by the UK’s Economic and Social Research Council and National Institute for Health Research. She disclosed no conflicts of interest.
Palliative care for people with dementia is increasingly recognized as a way to improve quality of life and provide relief from the myriad physical and psychological symptoms of advancing neurodegenerative disease. But unlike in cancer,
A new literature review has found these referrals to be all over the map among patients with dementia – with many occurring very late in the disease process – and do not reflect any consistent criteria based on patient needs.
For their research, published March 2 in the Journal of the American Geriatrics Society, Li Mo, MD, of the University of Texas MD Anderson Cancer Center in Houston, and colleagues looked at nearly 60 studies dating back to the early 1990s that contained information on referrals to palliative care for patients with dementia. While a palliative care approach can be provided by nonspecialists, all the included studies dealt at least in part with specialist care.
Standardized criteria is lacking
The investigators found advanced or late-stage dementia to be the most common reason cited for referral, with three quarters of the studies recommending palliative care for late-stage or advanced dementia, generally without qualifying what symptoms or needs were present. Patients received palliative care across a range of settings, including nursing homes, hospitals, and their own homes, though many articles did not include information on where patients received care.
A fifth of the articles suggested that medical complications of dementia including falls, pneumonia, and ulcers should trigger referrals to palliative care, while another fifth cited poor prognosis, defined varyingly as having between 2 years and 6 months likely left to live. Poor nutrition status was identified in 10% of studies as meriting referral.
Only 20% of the studies identified patient needs – evidence of psychological distress or functional decline, for example – as criteria for referral, despite these being ubiquitous in dementia. The authors said they were surprised by this finding, which could possibly be explained, they wrote, by “the interest among geriatrician, neurologist, and primary care teams to provide good symptom management,” reflecting a de facto palliative care approach. “There is also significant stigma associated with a specialist palliative care referral,” the authors noted.
Curiously, the researchers noted, a new diagnosis of dementia in more than a quarter of the studies triggered referral, a finding that possibly reflected delayed diagnoses.
The findings revealed “heterogeneity in the literature in reasons for involving specialist palliative care, which may partly explain the variation in patterns of palliative care referral,” Dr. Mo and colleagues wrote, stressing that more standardized criteria are urgently needed to bring dementia in line with cancer in terms of providing timely palliative care.
Patients with advancing dementia have little chance to self-report symptoms, meaning that more attention to patient complaints earlier in the disease course, and greater sensitivity to patient distress, are required. By routinely screening symptoms, clinicians could use specific cutoffs “as triggers to initiate automatic timely palliative care referral,” the authors concluded, noting that more research was needed before these cutoffs, whether based on symptom intensity or other measures, could be calculated.
Dr. Mo and colleagues acknowledged as weaknesses of their study the fact that a third of the articles in the review were based on expert consensus, while others did not distinguish clearly between primary and specialist palliative care.
A starting point for further discussion
Asked to comment on the findings, Elizabeth Sampson, MD, a palliative care researcher at University College London, praised Dr. Mo and colleagues’ study as “starting to pull together the strands” of a systematic approach to referrals and access to palliative care in dementia.
“Sometimes you need a paper like this to kick off the discussion to say look, this is where we are,” Dr. Sampson said, noting that the focus on need-based criteria dovetailed with a “general feeling in the field that we need to really think about needs, and what palliative care needs might be. What the threshold for referral should be we don’t know yet. Should it be three unmet needs? Or five? We’re still a long way from knowing.”
Dr. Sampson’s group is leading a UK-government funded research effort that aims to develop cost-effective palliative care interventions in dementia, in part through a tool that uses caregiver reports to assess symptom burden and patient needs. The research program “is founded on a needs-based approach, which aims to look at people’s individual needs and responding to them in a proactive way,” she said.
One of the obstacles to timely palliative care in dementia, Dr. Sampson said, is weighing resource allocation against what can be wildly varying prognoses. “Hospices understand when someone has terminal cancer and [is] likely to die within a few weeks, but it’s not unheard of for someone in very advanced stages of dementia to live another year,” she said. “There are concerns that a rapid increase in people with dementia being moved to palliative care could overwhelm already limited hospice capacity. We would argue that the best approach is to get palliative care out to where people with dementia live, which is usually the care home.”
Dr. Mo and colleagues’ study received funding from the National Institutes of Health, and its authors disclosed no financial conflicts of interest. Dr. Sampson’s work is supported by the UK’s Economic and Social Research Council and National Institute for Health Research. She disclosed no conflicts of interest.
FROM THE JOURNAL OF THE AMERICAN GERIATRICS SOCIETY
CDC data strengthen link between obesity and severe COVID
Officials have previously linked being overweight or obese to a greater risk for more severe COVID-19. A report today from the U.S. Centers for Disease Control and Prevention adds numbers and some nuance to the association.
Data from nearly 150,000 U.S. adults hospitalized with COVID-19 nationwide indicate that risk for more severe disease outcomes increases along with body mass index (BMI). The risk of COVID-19–related hospitalization and death associated with obesity was particularly high among people younger than 65.
“As clinicians develop care plans for COVID-19 patients, they should consider the risk for severe outcomes in patients with higher BMIs, especially for those with severe obesity,” the researchers note. They add that their findings suggest “progressively intensive management of COVID-19 might be needed for patients with more severe obesity.”
People with COVID-19 close to the border between a healthy and overweight BMI – from 23.7 kg/m2 to 25.9 kg/m2 – had the lowest risks for adverse outcomes.
The study was published online today in Morbidity and Mortality Weekly Report.
Greater need for critical care
The risk of ICU admission was particularly associated with severe obesity. For example, those with a BMI in the 40-44.9 kg/m2 category had a 6% increased risk, which jumped to 16% higher among those with a BMI of 45 or greater.
Compared to people with a healthy BMI, the need for invasive mechanical ventilation was 12% more likely among overweight adults with a BMI of 25-29.2. The risked jumped to 108% greater among the most obese people, those with a BMI of 45 or greater, lead CDC researcher Lyudmyla Kompaniyets, PhD, and colleagues reported.
Moreover, the risks for hospitalization and death increased in a dose-response relationship with obesity.
For example, risks of being hospitalized were 7% greater for adults with a BMI between 30 and 34.9 and climbed to 33% greater for those with a BMI of 45. Risks were calculated as adjusted relative risks compared with people with a healthy BMI between 18.5 and 24.9.
Interestingly, being underweight was associated with elevated risk for COVID-19 hospitalization as well. For example, people with a BMI of less than 18.5 had a 20% greater chance of admission vs. people in the healthy BMI range. Unknown underlying medical conditions or issues related to nutrition or immune function could be contributing factors, the researchers note.
Elevated risk of dying
The risk of death in adults with obesity ranged from 8% higher in the 30-34.9 range up to 61% greater for those with a BMI of 45.
Chronic inflammation or impaired lung function from excess weight are possible reasons that higher BMI imparts greater risk, the researchers note.
The CDC researchers evaluated 148,494 adults from 238 hospitals participating in PHD-SR database. Because the study was limited to people hospitalized with COVID-19, the findings may not apply to all adults with COVID-19.
Another potential limitation is that investigators were unable to calculate BMI for all patients in the database because about 28% of participating hospitals did not report height and weight.
The study authors had no relevant financial relationships to disclose.
A version of this article first appeared on Medscape.com.
Officials have previously linked being overweight or obese to a greater risk for more severe COVID-19. A report today from the U.S. Centers for Disease Control and Prevention adds numbers and some nuance to the association.
Data from nearly 150,000 U.S. adults hospitalized with COVID-19 nationwide indicate that risk for more severe disease outcomes increases along with body mass index (BMI). The risk of COVID-19–related hospitalization and death associated with obesity was particularly high among people younger than 65.
“As clinicians develop care plans for COVID-19 patients, they should consider the risk for severe outcomes in patients with higher BMIs, especially for those with severe obesity,” the researchers note. They add that their findings suggest “progressively intensive management of COVID-19 might be needed for patients with more severe obesity.”
People with COVID-19 close to the border between a healthy and overweight BMI – from 23.7 kg/m2 to 25.9 kg/m2 – had the lowest risks for adverse outcomes.
The study was published online today in Morbidity and Mortality Weekly Report.
Greater need for critical care
The risk of ICU admission was particularly associated with severe obesity. For example, those with a BMI in the 40-44.9 kg/m2 category had a 6% increased risk, which jumped to 16% higher among those with a BMI of 45 or greater.
Compared to people with a healthy BMI, the need for invasive mechanical ventilation was 12% more likely among overweight adults with a BMI of 25-29.2. The risked jumped to 108% greater among the most obese people, those with a BMI of 45 or greater, lead CDC researcher Lyudmyla Kompaniyets, PhD, and colleagues reported.
Moreover, the risks for hospitalization and death increased in a dose-response relationship with obesity.
For example, risks of being hospitalized were 7% greater for adults with a BMI between 30 and 34.9 and climbed to 33% greater for those with a BMI of 45. Risks were calculated as adjusted relative risks compared with people with a healthy BMI between 18.5 and 24.9.
Interestingly, being underweight was associated with elevated risk for COVID-19 hospitalization as well. For example, people with a BMI of less than 18.5 had a 20% greater chance of admission vs. people in the healthy BMI range. Unknown underlying medical conditions or issues related to nutrition or immune function could be contributing factors, the researchers note.
Elevated risk of dying
The risk of death in adults with obesity ranged from 8% higher in the 30-34.9 range up to 61% greater for those with a BMI of 45.
Chronic inflammation or impaired lung function from excess weight are possible reasons that higher BMI imparts greater risk, the researchers note.
The CDC researchers evaluated 148,494 adults from 238 hospitals participating in PHD-SR database. Because the study was limited to people hospitalized with COVID-19, the findings may not apply to all adults with COVID-19.
Another potential limitation is that investigators were unable to calculate BMI for all patients in the database because about 28% of participating hospitals did not report height and weight.
The study authors had no relevant financial relationships to disclose.
A version of this article first appeared on Medscape.com.
Officials have previously linked being overweight or obese to a greater risk for more severe COVID-19. A report today from the U.S. Centers for Disease Control and Prevention adds numbers and some nuance to the association.
Data from nearly 150,000 U.S. adults hospitalized with COVID-19 nationwide indicate that risk for more severe disease outcomes increases along with body mass index (BMI). The risk of COVID-19–related hospitalization and death associated with obesity was particularly high among people younger than 65.
“As clinicians develop care plans for COVID-19 patients, they should consider the risk for severe outcomes in patients with higher BMIs, especially for those with severe obesity,” the researchers note. They add that their findings suggest “progressively intensive management of COVID-19 might be needed for patients with more severe obesity.”
People with COVID-19 close to the border between a healthy and overweight BMI – from 23.7 kg/m2 to 25.9 kg/m2 – had the lowest risks for adverse outcomes.
The study was published online today in Morbidity and Mortality Weekly Report.
Greater need for critical care
The risk of ICU admission was particularly associated with severe obesity. For example, those with a BMI in the 40-44.9 kg/m2 category had a 6% increased risk, which jumped to 16% higher among those with a BMI of 45 or greater.
Compared to people with a healthy BMI, the need for invasive mechanical ventilation was 12% more likely among overweight adults with a BMI of 25-29.2. The risked jumped to 108% greater among the most obese people, those with a BMI of 45 or greater, lead CDC researcher Lyudmyla Kompaniyets, PhD, and colleagues reported.
Moreover, the risks for hospitalization and death increased in a dose-response relationship with obesity.
For example, risks of being hospitalized were 7% greater for adults with a BMI between 30 and 34.9 and climbed to 33% greater for those with a BMI of 45. Risks were calculated as adjusted relative risks compared with people with a healthy BMI between 18.5 and 24.9.
Interestingly, being underweight was associated with elevated risk for COVID-19 hospitalization as well. For example, people with a BMI of less than 18.5 had a 20% greater chance of admission vs. people in the healthy BMI range. Unknown underlying medical conditions or issues related to nutrition or immune function could be contributing factors, the researchers note.
Elevated risk of dying
The risk of death in adults with obesity ranged from 8% higher in the 30-34.9 range up to 61% greater for those with a BMI of 45.
Chronic inflammation or impaired lung function from excess weight are possible reasons that higher BMI imparts greater risk, the researchers note.
The CDC researchers evaluated 148,494 adults from 238 hospitals participating in PHD-SR database. Because the study was limited to people hospitalized with COVID-19, the findings may not apply to all adults with COVID-19.
Another potential limitation is that investigators were unable to calculate BMI for all patients in the database because about 28% of participating hospitals did not report height and weight.
The study authors had no relevant financial relationships to disclose.
A version of this article first appeared on Medscape.com.
FDA authorizes first molecular at-home, OTC COVID-19 test
The U.S. Food and Drug Administration has granted emergency use authorization (EUA) for the Cue COVID-19 Test for Home and Over The Counter Use (Cue OTC Test, Cue Health).
The Cue OTC Test is the first molecular diagnostic test available to consumers without a prescription.
The test detects genetic material from SARS-CoV-2 present in the nostrils and delivers results in about 20 minutes to the user’s mobile smart device via the Cue Health app.
In testing, the Cue OTC Test correctly identified 96% of positive nasal swab samples from individuals known to have symptoms and correctly identified 100% of positive samples from individuals without symptoms.
The test is intended for use in people aged 2 years and older with and without symptoms.
“With this authorization, consumers can purchase and self-administer one of the easiest, fastest, and most accurate tests without a prescription,” Clint Sever, cofounder and chief product officer of Cue Health, said in a news release.
“This FDA authorization will help us improve patient outcomes with a solution that provides the accuracy of central lab tests, with the speed and accessibility required to address emergent global health issues,” he said.
Cue Health expects to produce more than 100,000 single-use test kits per day by this summer. Dena Cook, the company’s chief communications officer, told this news organization that the company hasn’t announced pricing information yet, but the price will be “comparable” to other price points and other products on the market.
“The FDA continues to prioritize the availability of more at-home testing options in response to the pandemic,” Jeff Shuren, MD, JD, director of the FDA’s Center for Devices and Radiological Health, said in a statement.
“Cue COVID-19 Test for Home and Over-the-Counter Use provides access to accurate and reliable testing at home, without a prescription. The FDA will continue to work collaboratively with test developers to advance effective testing options for doctors, clinicians, and the public,” he said.
In June, the FDA granted an EUA to Cue Health’s COVID-19 test for use in clinical and point-of-care settings.
The test is currently being used in hospitals, physicians’ offices, and dental clinics, as well as schools, essential businesses, nursing homes, and other congregate-care facilities. The test is also being distributed through a program led by the U.S. Department of Defense and the U.S. Department of Health & Human Services across several states.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has granted emergency use authorization (EUA) for the Cue COVID-19 Test for Home and Over The Counter Use (Cue OTC Test, Cue Health).
The Cue OTC Test is the first molecular diagnostic test available to consumers without a prescription.
The test detects genetic material from SARS-CoV-2 present in the nostrils and delivers results in about 20 minutes to the user’s mobile smart device via the Cue Health app.
In testing, the Cue OTC Test correctly identified 96% of positive nasal swab samples from individuals known to have symptoms and correctly identified 100% of positive samples from individuals without symptoms.
The test is intended for use in people aged 2 years and older with and without symptoms.
“With this authorization, consumers can purchase and self-administer one of the easiest, fastest, and most accurate tests without a prescription,” Clint Sever, cofounder and chief product officer of Cue Health, said in a news release.
“This FDA authorization will help us improve patient outcomes with a solution that provides the accuracy of central lab tests, with the speed and accessibility required to address emergent global health issues,” he said.
Cue Health expects to produce more than 100,000 single-use test kits per day by this summer. Dena Cook, the company’s chief communications officer, told this news organization that the company hasn’t announced pricing information yet, but the price will be “comparable” to other price points and other products on the market.
“The FDA continues to prioritize the availability of more at-home testing options in response to the pandemic,” Jeff Shuren, MD, JD, director of the FDA’s Center for Devices and Radiological Health, said in a statement.
“Cue COVID-19 Test for Home and Over-the-Counter Use provides access to accurate and reliable testing at home, without a prescription. The FDA will continue to work collaboratively with test developers to advance effective testing options for doctors, clinicians, and the public,” he said.
In June, the FDA granted an EUA to Cue Health’s COVID-19 test for use in clinical and point-of-care settings.
The test is currently being used in hospitals, physicians’ offices, and dental clinics, as well as schools, essential businesses, nursing homes, and other congregate-care facilities. The test is also being distributed through a program led by the U.S. Department of Defense and the U.S. Department of Health & Human Services across several states.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has granted emergency use authorization (EUA) for the Cue COVID-19 Test for Home and Over The Counter Use (Cue OTC Test, Cue Health).
The Cue OTC Test is the first molecular diagnostic test available to consumers without a prescription.
The test detects genetic material from SARS-CoV-2 present in the nostrils and delivers results in about 20 minutes to the user’s mobile smart device via the Cue Health app.
In testing, the Cue OTC Test correctly identified 96% of positive nasal swab samples from individuals known to have symptoms and correctly identified 100% of positive samples from individuals without symptoms.
The test is intended for use in people aged 2 years and older with and without symptoms.
“With this authorization, consumers can purchase and self-administer one of the easiest, fastest, and most accurate tests without a prescription,” Clint Sever, cofounder and chief product officer of Cue Health, said in a news release.
“This FDA authorization will help us improve patient outcomes with a solution that provides the accuracy of central lab tests, with the speed and accessibility required to address emergent global health issues,” he said.
Cue Health expects to produce more than 100,000 single-use test kits per day by this summer. Dena Cook, the company’s chief communications officer, told this news organization that the company hasn’t announced pricing information yet, but the price will be “comparable” to other price points and other products on the market.
“The FDA continues to prioritize the availability of more at-home testing options in response to the pandemic,” Jeff Shuren, MD, JD, director of the FDA’s Center for Devices and Radiological Health, said in a statement.
“Cue COVID-19 Test for Home and Over-the-Counter Use provides access to accurate and reliable testing at home, without a prescription. The FDA will continue to work collaboratively with test developers to advance effective testing options for doctors, clinicians, and the public,” he said.
In June, the FDA granted an EUA to Cue Health’s COVID-19 test for use in clinical and point-of-care settings.
The test is currently being used in hospitals, physicians’ offices, and dental clinics, as well as schools, essential businesses, nursing homes, and other congregate-care facilities. The test is also being distributed through a program led by the U.S. Department of Defense and the U.S. Department of Health & Human Services across several states.
A version of this article first appeared on Medscape.com.
Missed visits during pandemic cause ‘detrimental ripple effects’
according to a new report from the Urban Institute.
Among the adults who postponed or missed care, 32.6% said the gap worsened one or more health conditions or limited their ability to work or perform daily activities. The findings highlight “the detrimental ripple effects of delaying or forgoing care on overall health, functioning, and well-being,” researchers write.
The survey, conducted among 4,007 U.S. adults aged 18-64 in September 2020, found that adults with one or more chronic conditions were more likely than adults without chronic conditions to have delayed or missed care (40.7% vs. 26.4%). Adults with a mental health condition were particularly likely to have delayed or gone without care, write Dulce Gonzalez, MPP, a research associate in the Health Policy Center at the Urban Institute, and colleagues.
Doctors are already seeing the consequences of the missed visits, says Jacqueline W. Fincher, MD, president of the American College of Physicians.
Two of her patients with chronic conditions missed appointments last year. By the time they resumed care in 2021, their previsit lab tests showed significant kidney deterioration.
“Lo and behold, their kidneys were in failure. … One was in the hospital for 3 days and the other one was in for 5 days,” said Dr. Fincher, who practices general internal medicine in Georgia.
Dr. Fincher’s office has been proactive about calling patients with chronic diseases who missed follow-up visits or laboratory testing or who may have run out of medication, she said.
In her experience, delays mainly have been because of patients postponing visits. “We have stayed open the whole time now,” Dr. Fincher said. Her office offers telemedicine visits and in-person visits with safety precautions.
Still, some patients have decided to postpone care during the pandemic instead of asking their primary care doctor what they should do.
“We do know that chronic problems left without appropriate follow-up can create worse problems for them in terms of stroke, heart attack, and end organ damage,” Dr. Fincher said.
Lost lives
Future studies may help researchers understand the effects of delayed and missed care during the pandemic, said Russell S. Phillips, MD, director of the Center for Primary Care at Harvard Medical School, Boston.
“Although it is still early, and more data on patient outcomes will need to be collected, I anticipate that the ... delays in diagnosis, in cancer screening, and in management of chronic illness will result in lost lives and will emphasize the important role that primary care plays in saving lives,” Dr. Phillips said.
During the first several months of the pandemic, there were fewer diagnoses of hypertension, diabetes, and depression, Dr. Phillips said.
“In addition, and most importantly, the mortality rate for non-COVID conditions increased, suggesting that patients were not seeking care for symptoms of stroke or heart attack, which can be fatal if untreated,” he said. “We have also seen substantial decreases in cancer screening tests such as colonoscopy, and modeling studies suggest this will cost more lives based on delayed diagnoses of cancer.”
Vaccinating patients against COVID-19 may help primary care practices and patients get back on track, Dr. Phillips suggested.
In the meantime, some patients remain reluctant to come in. “Volumes are still lower than prepandemic, so it is challenging to overcome what is likely to be pent-up demand,” he told this news organization in an email. “Additionally, the continued burden of evaluating, testing, and monitoring patients with COVID or COVID-like symptoms makes it difficult to focus on chronic illness.”
Care most often skipped
The Urban Institute survey asked respondents about delays in prescription drugs, general doctor and specialist visits, going to a hospital, preventive health screenings or medical tests, treatment or follow-up care, dental care, mental health care or counseling, treatment or counseling for alcohol or drug use, and other types of medical care.
Dental care was the most common type of care that adults delayed or did not receive because of the pandemic (25.3%), followed by general doctor or specialist visits (20.6%) and preventive health screenings or medical tests (15.5%).
Black adults were more likely than White or Hispanic/Latinx adults to have delayed or forgone care (39.7% vs. 34.3% and 35.5%), the researchers found. Compared with adults with higher incomes, adults with lower incomes were more likely to have missed multiple types of care (26.6% vs. 20.3%).
The report by the Urban Institute researchers was supported by the Robert Wood Johnson Foundation. Dr. Phillips is an adviser to two telemedicine companies, Bicycle Health and Grow Health. Dr. Fincher has disclosed no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
according to a new report from the Urban Institute.
Among the adults who postponed or missed care, 32.6% said the gap worsened one or more health conditions or limited their ability to work or perform daily activities. The findings highlight “the detrimental ripple effects of delaying or forgoing care on overall health, functioning, and well-being,” researchers write.
The survey, conducted among 4,007 U.S. adults aged 18-64 in September 2020, found that adults with one or more chronic conditions were more likely than adults without chronic conditions to have delayed or missed care (40.7% vs. 26.4%). Adults with a mental health condition were particularly likely to have delayed or gone without care, write Dulce Gonzalez, MPP, a research associate in the Health Policy Center at the Urban Institute, and colleagues.
Doctors are already seeing the consequences of the missed visits, says Jacqueline W. Fincher, MD, president of the American College of Physicians.
Two of her patients with chronic conditions missed appointments last year. By the time they resumed care in 2021, their previsit lab tests showed significant kidney deterioration.
“Lo and behold, their kidneys were in failure. … One was in the hospital for 3 days and the other one was in for 5 days,” said Dr. Fincher, who practices general internal medicine in Georgia.
Dr. Fincher’s office has been proactive about calling patients with chronic diseases who missed follow-up visits or laboratory testing or who may have run out of medication, she said.
In her experience, delays mainly have been because of patients postponing visits. “We have stayed open the whole time now,” Dr. Fincher said. Her office offers telemedicine visits and in-person visits with safety precautions.
Still, some patients have decided to postpone care during the pandemic instead of asking their primary care doctor what they should do.
“We do know that chronic problems left without appropriate follow-up can create worse problems for them in terms of stroke, heart attack, and end organ damage,” Dr. Fincher said.
Lost lives
Future studies may help researchers understand the effects of delayed and missed care during the pandemic, said Russell S. Phillips, MD, director of the Center for Primary Care at Harvard Medical School, Boston.
“Although it is still early, and more data on patient outcomes will need to be collected, I anticipate that the ... delays in diagnosis, in cancer screening, and in management of chronic illness will result in lost lives and will emphasize the important role that primary care plays in saving lives,” Dr. Phillips said.
During the first several months of the pandemic, there were fewer diagnoses of hypertension, diabetes, and depression, Dr. Phillips said.
“In addition, and most importantly, the mortality rate for non-COVID conditions increased, suggesting that patients were not seeking care for symptoms of stroke or heart attack, which can be fatal if untreated,” he said. “We have also seen substantial decreases in cancer screening tests such as colonoscopy, and modeling studies suggest this will cost more lives based on delayed diagnoses of cancer.”
Vaccinating patients against COVID-19 may help primary care practices and patients get back on track, Dr. Phillips suggested.
In the meantime, some patients remain reluctant to come in. “Volumes are still lower than prepandemic, so it is challenging to overcome what is likely to be pent-up demand,” he told this news organization in an email. “Additionally, the continued burden of evaluating, testing, and monitoring patients with COVID or COVID-like symptoms makes it difficult to focus on chronic illness.”
Care most often skipped
The Urban Institute survey asked respondents about delays in prescription drugs, general doctor and specialist visits, going to a hospital, preventive health screenings or medical tests, treatment or follow-up care, dental care, mental health care or counseling, treatment or counseling for alcohol or drug use, and other types of medical care.
Dental care was the most common type of care that adults delayed or did not receive because of the pandemic (25.3%), followed by general doctor or specialist visits (20.6%) and preventive health screenings or medical tests (15.5%).
Black adults were more likely than White or Hispanic/Latinx adults to have delayed or forgone care (39.7% vs. 34.3% and 35.5%), the researchers found. Compared with adults with higher incomes, adults with lower incomes were more likely to have missed multiple types of care (26.6% vs. 20.3%).
The report by the Urban Institute researchers was supported by the Robert Wood Johnson Foundation. Dr. Phillips is an adviser to two telemedicine companies, Bicycle Health and Grow Health. Dr. Fincher has disclosed no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
according to a new report from the Urban Institute.
Among the adults who postponed or missed care, 32.6% said the gap worsened one or more health conditions or limited their ability to work or perform daily activities. The findings highlight “the detrimental ripple effects of delaying or forgoing care on overall health, functioning, and well-being,” researchers write.
The survey, conducted among 4,007 U.S. adults aged 18-64 in September 2020, found that adults with one or more chronic conditions were more likely than adults without chronic conditions to have delayed or missed care (40.7% vs. 26.4%). Adults with a mental health condition were particularly likely to have delayed or gone without care, write Dulce Gonzalez, MPP, a research associate in the Health Policy Center at the Urban Institute, and colleagues.
Doctors are already seeing the consequences of the missed visits, says Jacqueline W. Fincher, MD, president of the American College of Physicians.
Two of her patients with chronic conditions missed appointments last year. By the time they resumed care in 2021, their previsit lab tests showed significant kidney deterioration.
“Lo and behold, their kidneys were in failure. … One was in the hospital for 3 days and the other one was in for 5 days,” said Dr. Fincher, who practices general internal medicine in Georgia.
Dr. Fincher’s office has been proactive about calling patients with chronic diseases who missed follow-up visits or laboratory testing or who may have run out of medication, she said.
In her experience, delays mainly have been because of patients postponing visits. “We have stayed open the whole time now,” Dr. Fincher said. Her office offers telemedicine visits and in-person visits with safety precautions.
Still, some patients have decided to postpone care during the pandemic instead of asking their primary care doctor what they should do.
“We do know that chronic problems left without appropriate follow-up can create worse problems for them in terms of stroke, heart attack, and end organ damage,” Dr. Fincher said.
Lost lives
Future studies may help researchers understand the effects of delayed and missed care during the pandemic, said Russell S. Phillips, MD, director of the Center for Primary Care at Harvard Medical School, Boston.
“Although it is still early, and more data on patient outcomes will need to be collected, I anticipate that the ... delays in diagnosis, in cancer screening, and in management of chronic illness will result in lost lives and will emphasize the important role that primary care plays in saving lives,” Dr. Phillips said.
During the first several months of the pandemic, there were fewer diagnoses of hypertension, diabetes, and depression, Dr. Phillips said.
“In addition, and most importantly, the mortality rate for non-COVID conditions increased, suggesting that patients were not seeking care for symptoms of stroke or heart attack, which can be fatal if untreated,” he said. “We have also seen substantial decreases in cancer screening tests such as colonoscopy, and modeling studies suggest this will cost more lives based on delayed diagnoses of cancer.”
Vaccinating patients against COVID-19 may help primary care practices and patients get back on track, Dr. Phillips suggested.
In the meantime, some patients remain reluctant to come in. “Volumes are still lower than prepandemic, so it is challenging to overcome what is likely to be pent-up demand,” he told this news organization in an email. “Additionally, the continued burden of evaluating, testing, and monitoring patients with COVID or COVID-like symptoms makes it difficult to focus on chronic illness.”
Care most often skipped
The Urban Institute survey asked respondents about delays in prescription drugs, general doctor and specialist visits, going to a hospital, preventive health screenings or medical tests, treatment or follow-up care, dental care, mental health care or counseling, treatment or counseling for alcohol or drug use, and other types of medical care.
Dental care was the most common type of care that adults delayed or did not receive because of the pandemic (25.3%), followed by general doctor or specialist visits (20.6%) and preventive health screenings or medical tests (15.5%).
Black adults were more likely than White or Hispanic/Latinx adults to have delayed or forgone care (39.7% vs. 34.3% and 35.5%), the researchers found. Compared with adults with higher incomes, adults with lower incomes were more likely to have missed multiple types of care (26.6% vs. 20.3%).
The report by the Urban Institute researchers was supported by the Robert Wood Johnson Foundation. Dr. Phillips is an adviser to two telemedicine companies, Bicycle Health and Grow Health. Dr. Fincher has disclosed no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
FDA okays novel dual-action stimulant med for ADHD
The Food and Drug Administration has approved a new, once-daily oral stimulant medication for treatment of ADHD in people aged 6 years and older.
Azstarys (KemPharm) combines extended-release serdexmethylphenidate (SDX), KemPharm’s prodrug of dexmethylphenidate (d-MPH), coformulated with immediate-release d-MPH.
Following absorption in the gastrointestinal tract, SDX is converted to d-MPH, which is gradually released throughout the day, providing symptom control both rapidly with the d-MPH and for an extended duration with SDX.
The dual action of Azstarys addresses an unmet need for a medication that has early onset of action and long duration of therapy, with steady ADHD symptom control in one capsule, Corium, the company that will lead U.S. commercialization of the drug, stated in a news release.
“The data documenting the efficacy and safety of this new dual-action medicine, the first ever to use the novel prodrug serdexmethylphenidate together with dexmethylphenidate, is welcome news for clinicians and families to consider when choosing an appropriate ADHD therapy for children,” Ann Childress, MD, president of the Center for Psychiatry and Behavioral Medicine in Las Vegas, who led the phase 3 trial of the drug, said in the release.
The study included 150 children aged 6-12 years with ADHD. Compared with placebo, treatment with Azstarys led to significant improvement in ADHD symptoms, as measured by the primary endpoint, the change from baseline in Swanson, Kotkin, Agler, M-Flynn, and Pelham Rating Scale–Combined scores averaged over 13 hours.
Adverse events seen more often with Azstarys than placebo were headache (5.4% vs. 1.3%), upper abdominal pain (4.1% vs. 1.3%), insomnia (2.7% vs. 1.3%) and pharyngitis (2.7% vs. 0%). No serious adverse events were reported.
The FDA has recommended a schedule II controlled substance classification for Azstarys and the Drug Enforcement Administration will decide on scheduling within 90 days.
Pending the DEA’s action, the launch of Azstarys is anticipated this summer. Azstarys will be available in three once-daily dosage strengths of SDX/d-MPH: 26.1/5.2 mg, 39.2/7.8 mg, and 52.3/10.4 mg.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved a new, once-daily oral stimulant medication for treatment of ADHD in people aged 6 years and older.
Azstarys (KemPharm) combines extended-release serdexmethylphenidate (SDX), KemPharm’s prodrug of dexmethylphenidate (d-MPH), coformulated with immediate-release d-MPH.
Following absorption in the gastrointestinal tract, SDX is converted to d-MPH, which is gradually released throughout the day, providing symptom control both rapidly with the d-MPH and for an extended duration with SDX.
The dual action of Azstarys addresses an unmet need for a medication that has early onset of action and long duration of therapy, with steady ADHD symptom control in one capsule, Corium, the company that will lead U.S. commercialization of the drug, stated in a news release.
“The data documenting the efficacy and safety of this new dual-action medicine, the first ever to use the novel prodrug serdexmethylphenidate together with dexmethylphenidate, is welcome news for clinicians and families to consider when choosing an appropriate ADHD therapy for children,” Ann Childress, MD, president of the Center for Psychiatry and Behavioral Medicine in Las Vegas, who led the phase 3 trial of the drug, said in the release.
The study included 150 children aged 6-12 years with ADHD. Compared with placebo, treatment with Azstarys led to significant improvement in ADHD symptoms, as measured by the primary endpoint, the change from baseline in Swanson, Kotkin, Agler, M-Flynn, and Pelham Rating Scale–Combined scores averaged over 13 hours.
Adverse events seen more often with Azstarys than placebo were headache (5.4% vs. 1.3%), upper abdominal pain (4.1% vs. 1.3%), insomnia (2.7% vs. 1.3%) and pharyngitis (2.7% vs. 0%). No serious adverse events were reported.
The FDA has recommended a schedule II controlled substance classification for Azstarys and the Drug Enforcement Administration will decide on scheduling within 90 days.
Pending the DEA’s action, the launch of Azstarys is anticipated this summer. Azstarys will be available in three once-daily dosage strengths of SDX/d-MPH: 26.1/5.2 mg, 39.2/7.8 mg, and 52.3/10.4 mg.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved a new, once-daily oral stimulant medication for treatment of ADHD in people aged 6 years and older.
Azstarys (KemPharm) combines extended-release serdexmethylphenidate (SDX), KemPharm’s prodrug of dexmethylphenidate (d-MPH), coformulated with immediate-release d-MPH.
Following absorption in the gastrointestinal tract, SDX is converted to d-MPH, which is gradually released throughout the day, providing symptom control both rapidly with the d-MPH and for an extended duration with SDX.
The dual action of Azstarys addresses an unmet need for a medication that has early onset of action and long duration of therapy, with steady ADHD symptom control in one capsule, Corium, the company that will lead U.S. commercialization of the drug, stated in a news release.
“The data documenting the efficacy and safety of this new dual-action medicine, the first ever to use the novel prodrug serdexmethylphenidate together with dexmethylphenidate, is welcome news for clinicians and families to consider when choosing an appropriate ADHD therapy for children,” Ann Childress, MD, president of the Center for Psychiatry and Behavioral Medicine in Las Vegas, who led the phase 3 trial of the drug, said in the release.
The study included 150 children aged 6-12 years with ADHD. Compared with placebo, treatment with Azstarys led to significant improvement in ADHD symptoms, as measured by the primary endpoint, the change from baseline in Swanson, Kotkin, Agler, M-Flynn, and Pelham Rating Scale–Combined scores averaged over 13 hours.
Adverse events seen more often with Azstarys than placebo were headache (5.4% vs. 1.3%), upper abdominal pain (4.1% vs. 1.3%), insomnia (2.7% vs. 1.3%) and pharyngitis (2.7% vs. 0%). No serious adverse events were reported.
The FDA has recommended a schedule II controlled substance classification for Azstarys and the Drug Enforcement Administration will decide on scheduling within 90 days.
Pending the DEA’s action, the launch of Azstarys is anticipated this summer. Azstarys will be available in three once-daily dosage strengths of SDX/d-MPH: 26.1/5.2 mg, 39.2/7.8 mg, and 52.3/10.4 mg.
A version of this article first appeared on Medscape.com.
Call to action on obesity amid COVID-19 pandemic
Hundreds of thousands of deaths worldwide from COVID-19 could have been avoided if obesity rates were lower, a new report says.
An analysis by the World Obesity Federation found that of the 2.5 million COVID-19 deaths reported by the end of February 2021, almost 90% (2.2 million) were in countries where more than half the population is classified as overweight.
The report, released to coincide with World Obesity Day, calls for obesity to be recognized as a disease in its own right around the world, and for people with obesity to be included in priority lists for COVID-19 testing and vaccination.
“Overweight is a highly significant predictor of developing complications from COVID-19, including the need for hospitalization, for intensive care and for mechanical ventilation,” the WOF notes in the report.
It adds that in countries where less than half the adult population is classified as overweight (body mass index > 25 mg/kg2), for example, Vietnam, the likelihood of death from COVID-19 is a small fraction – around one-tenth – of the level seen in countries where more than half the population is classified as overweight.
And while it acknowledges that figures for COVID-19 deaths are affected by the age structure of national populations and a country’s relative wealth and reporting capacity, “our findings appear to be independent of these contributory factors. Furthermore, other studies have found that overweight remains a highly significant predictor of the need for COVID-19 health care after accounting for these other influences.”
As an example, based on the U.K. experience, where an estimated 36% of COVID-19 hospitalizations have been attributed to lack of physical activity and excess body weight, it can be suggested that up to a third of the costs – between $6 trillion and $7 trillion over the longer period – might be attributable to these predisposing risks.
The report said the prevalence of obesity in the United Kingdom is expected to rise from 27.8% in 2016 to more than 35% by 2025.
Rachel Batterham, lead adviser on obesity at the Royal College of Physicians, commented: “The link between high levels of obesity and deaths from COVID-19 in the U.K. is indisputable, as is the urgent need to address the factors that lead so many people to be living with obesity.
“With 30% of COVID-19 hospitalizations in the U.K. directly attributed to overweight and obesity, and three-quarters of all critically ill patients having overweight or obesity, the human and financial costs are high.”
Window of opportunity to prioritize obesity as a disease
WOF says that evolving evidence on the close association between COVID-19 and underlying obesity “provides a new urgency … for political and collective action.”
“Obesity is a disease that does not receive prioritization commensurate with its prevalence and impact, which is rising fastest in emerging economies. It is a gateway to many other noncommunicable diseases and mental-health illness and is now a major factor in COVID-19 complications and mortality.”
The WOF also shows that COVID-19 is not a special case, noting that several other respiratory viruses lead to more severe consequences in people living with excess bodyweight, giving good reasons to expect the next pandemic to have similar effects. “For these reasons we need to recognize overweight as a major risk factor for infectious diseases including respiratory viruses.”
“To prevent pandemic health crises in future requires action now: we call on all readers to support the World Obesity Federation’s call for stronger, more resilient economies that prioritize investment in people’s health.”
There is, it stresses, “a window of opportunity to advocate for, fund and implement these actions in all countries to ensure better, more resilient and sustainable health for all, “now and in our postCOVID-19 future.”
It proposes a ROOTS approach:
- Recognize that obesity is a disease in its own right.
- Obesity monitoring and surveillance must be enhanced.
- Obesity prevention strategies must be developed.
- Treatment of obesity.
- Systems-based approaches should be applied.
A version of this article first appeared on Medscape.com.
Hundreds of thousands of deaths worldwide from COVID-19 could have been avoided if obesity rates were lower, a new report says.
An analysis by the World Obesity Federation found that of the 2.5 million COVID-19 deaths reported by the end of February 2021, almost 90% (2.2 million) were in countries where more than half the population is classified as overweight.
The report, released to coincide with World Obesity Day, calls for obesity to be recognized as a disease in its own right around the world, and for people with obesity to be included in priority lists for COVID-19 testing and vaccination.
“Overweight is a highly significant predictor of developing complications from COVID-19, including the need for hospitalization, for intensive care and for mechanical ventilation,” the WOF notes in the report.
It adds that in countries where less than half the adult population is classified as overweight (body mass index > 25 mg/kg2), for example, Vietnam, the likelihood of death from COVID-19 is a small fraction – around one-tenth – of the level seen in countries where more than half the population is classified as overweight.
And while it acknowledges that figures for COVID-19 deaths are affected by the age structure of national populations and a country’s relative wealth and reporting capacity, “our findings appear to be independent of these contributory factors. Furthermore, other studies have found that overweight remains a highly significant predictor of the need for COVID-19 health care after accounting for these other influences.”
As an example, based on the U.K. experience, where an estimated 36% of COVID-19 hospitalizations have been attributed to lack of physical activity and excess body weight, it can be suggested that up to a third of the costs – between $6 trillion and $7 trillion over the longer period – might be attributable to these predisposing risks.
The report said the prevalence of obesity in the United Kingdom is expected to rise from 27.8% in 2016 to more than 35% by 2025.
Rachel Batterham, lead adviser on obesity at the Royal College of Physicians, commented: “The link between high levels of obesity and deaths from COVID-19 in the U.K. is indisputable, as is the urgent need to address the factors that lead so many people to be living with obesity.
“With 30% of COVID-19 hospitalizations in the U.K. directly attributed to overweight and obesity, and three-quarters of all critically ill patients having overweight or obesity, the human and financial costs are high.”
Window of opportunity to prioritize obesity as a disease
WOF says that evolving evidence on the close association between COVID-19 and underlying obesity “provides a new urgency … for political and collective action.”
“Obesity is a disease that does not receive prioritization commensurate with its prevalence and impact, which is rising fastest in emerging economies. It is a gateway to many other noncommunicable diseases and mental-health illness and is now a major factor in COVID-19 complications and mortality.”
The WOF also shows that COVID-19 is not a special case, noting that several other respiratory viruses lead to more severe consequences in people living with excess bodyweight, giving good reasons to expect the next pandemic to have similar effects. “For these reasons we need to recognize overweight as a major risk factor for infectious diseases including respiratory viruses.”
“To prevent pandemic health crises in future requires action now: we call on all readers to support the World Obesity Federation’s call for stronger, more resilient economies that prioritize investment in people’s health.”
There is, it stresses, “a window of opportunity to advocate for, fund and implement these actions in all countries to ensure better, more resilient and sustainable health for all, “now and in our postCOVID-19 future.”
It proposes a ROOTS approach:
- Recognize that obesity is a disease in its own right.
- Obesity monitoring and surveillance must be enhanced.
- Obesity prevention strategies must be developed.
- Treatment of obesity.
- Systems-based approaches should be applied.
A version of this article first appeared on Medscape.com.
Hundreds of thousands of deaths worldwide from COVID-19 could have been avoided if obesity rates were lower, a new report says.
An analysis by the World Obesity Federation found that of the 2.5 million COVID-19 deaths reported by the end of February 2021, almost 90% (2.2 million) were in countries where more than half the population is classified as overweight.
The report, released to coincide with World Obesity Day, calls for obesity to be recognized as a disease in its own right around the world, and for people with obesity to be included in priority lists for COVID-19 testing and vaccination.
“Overweight is a highly significant predictor of developing complications from COVID-19, including the need for hospitalization, for intensive care and for mechanical ventilation,” the WOF notes in the report.
It adds that in countries where less than half the adult population is classified as overweight (body mass index > 25 mg/kg2), for example, Vietnam, the likelihood of death from COVID-19 is a small fraction – around one-tenth – of the level seen in countries where more than half the population is classified as overweight.
And while it acknowledges that figures for COVID-19 deaths are affected by the age structure of national populations and a country’s relative wealth and reporting capacity, “our findings appear to be independent of these contributory factors. Furthermore, other studies have found that overweight remains a highly significant predictor of the need for COVID-19 health care after accounting for these other influences.”
As an example, based on the U.K. experience, where an estimated 36% of COVID-19 hospitalizations have been attributed to lack of physical activity and excess body weight, it can be suggested that up to a third of the costs – between $6 trillion and $7 trillion over the longer period – might be attributable to these predisposing risks.
The report said the prevalence of obesity in the United Kingdom is expected to rise from 27.8% in 2016 to more than 35% by 2025.
Rachel Batterham, lead adviser on obesity at the Royal College of Physicians, commented: “The link between high levels of obesity and deaths from COVID-19 in the U.K. is indisputable, as is the urgent need to address the factors that lead so many people to be living with obesity.
“With 30% of COVID-19 hospitalizations in the U.K. directly attributed to overweight and obesity, and three-quarters of all critically ill patients having overweight or obesity, the human and financial costs are high.”
Window of opportunity to prioritize obesity as a disease
WOF says that evolving evidence on the close association between COVID-19 and underlying obesity “provides a new urgency … for political and collective action.”
“Obesity is a disease that does not receive prioritization commensurate with its prevalence and impact, which is rising fastest in emerging economies. It is a gateway to many other noncommunicable diseases and mental-health illness and is now a major factor in COVID-19 complications and mortality.”
The WOF also shows that COVID-19 is not a special case, noting that several other respiratory viruses lead to more severe consequences in people living with excess bodyweight, giving good reasons to expect the next pandemic to have similar effects. “For these reasons we need to recognize overweight as a major risk factor for infectious diseases including respiratory viruses.”
“To prevent pandemic health crises in future requires action now: we call on all readers to support the World Obesity Federation’s call for stronger, more resilient economies that prioritize investment in people’s health.”
There is, it stresses, “a window of opportunity to advocate for, fund and implement these actions in all countries to ensure better, more resilient and sustainable health for all, “now and in our postCOVID-19 future.”
It proposes a ROOTS approach:
- Recognize that obesity is a disease in its own right.
- Obesity monitoring and surveillance must be enhanced.
- Obesity prevention strategies must be developed.
- Treatment of obesity.
- Systems-based approaches should be applied.
A version of this article first appeared on Medscape.com.