User login
Androgen deprivation decreases lung tumors in men with prostate cancer
WASHINGTON – Men who received androgen blockade therapy as part of prostate cancer treatment were less likely to develop a secondary lung cancer than were those who didn’t receive the hormone regimen.
The significant finding suggests that estrogen may fuel non–small cell lung cancers in both men and women, Dr. Hyunseok Kang reported at the annual meeting of the American Association for Cancer Research.
"Recent literature suggests estrogen receptors and aromatase play a role in the development of non–small cell lung cancers in women with breast cancer," Dr. Kang said. "Aromatase can convert testosterone to estrogen – which could increase the same risk in men."
This recently observed association prompted Dr. Kang, a fellow at Emory University, Atlanta, and his colleagues to investigate whether a similar hormone-mediated link might exist for men with prostate cancer – and whether blocking it could also block lung cancer.
The retrospective observational study pulled data from the Veterans Affairs Central Cancer Registry. It comprised 62,589 men who were diagnosed with prostate cancer between 1999 and 2008, with follow-up through 2010.
There were some significant – and important – baseline differences between the 20,378 men who underwent androgen blockade and those who did not. Men who received the treatment were significantly older (mean age, 71 years vs. 66 years) and slightly more likely to have ever smoked tobacco (73% vs. 71%).
Prostate cancer stage at diagnosis was also significantly different, with fewer of the hormone therapy group presenting at stage II (77% vs. 92%) and more presenting at stage IV (17% vs. 2%).
Men from the VA database were significantly more likely than the general population to develop lung cancer, whether they underwent androgen blockade therapy or not (1.8/100,000 men who received treatment and 2.4/100,000 men who did not receive treatment vs. 0.80/100,000 in the general population).
Of men in the VA database who received androgen treatment, 258 developed secondary lung cancers, compared with 508 men who did not receive treatment.
In an unadjusted model, men who had treatment were 18% less likely to develop a secondary lung cancer (hazard ratio, 0.82). That finding held in a multivariate analysis that adjusted for smoking and age (HR, 0.83).
The study was sponsored by the Veterans Health Administration. Dr. Kang had no financial disclosures.
WASHINGTON – Men who received androgen blockade therapy as part of prostate cancer treatment were less likely to develop a secondary lung cancer than were those who didn’t receive the hormone regimen.
The significant finding suggests that estrogen may fuel non–small cell lung cancers in both men and women, Dr. Hyunseok Kang reported at the annual meeting of the American Association for Cancer Research.
"Recent literature suggests estrogen receptors and aromatase play a role in the development of non–small cell lung cancers in women with breast cancer," Dr. Kang said. "Aromatase can convert testosterone to estrogen – which could increase the same risk in men."
This recently observed association prompted Dr. Kang, a fellow at Emory University, Atlanta, and his colleagues to investigate whether a similar hormone-mediated link might exist for men with prostate cancer – and whether blocking it could also block lung cancer.
The retrospective observational study pulled data from the Veterans Affairs Central Cancer Registry. It comprised 62,589 men who were diagnosed with prostate cancer between 1999 and 2008, with follow-up through 2010.
There were some significant – and important – baseline differences between the 20,378 men who underwent androgen blockade and those who did not. Men who received the treatment were significantly older (mean age, 71 years vs. 66 years) and slightly more likely to have ever smoked tobacco (73% vs. 71%).
Prostate cancer stage at diagnosis was also significantly different, with fewer of the hormone therapy group presenting at stage II (77% vs. 92%) and more presenting at stage IV (17% vs. 2%).
Men from the VA database were significantly more likely than the general population to develop lung cancer, whether they underwent androgen blockade therapy or not (1.8/100,000 men who received treatment and 2.4/100,000 men who did not receive treatment vs. 0.80/100,000 in the general population).
Of men in the VA database who received androgen treatment, 258 developed secondary lung cancers, compared with 508 men who did not receive treatment.
In an unadjusted model, men who had treatment were 18% less likely to develop a secondary lung cancer (hazard ratio, 0.82). That finding held in a multivariate analysis that adjusted for smoking and age (HR, 0.83).
The study was sponsored by the Veterans Health Administration. Dr. Kang had no financial disclosures.
WASHINGTON – Men who received androgen blockade therapy as part of prostate cancer treatment were less likely to develop a secondary lung cancer than were those who didn’t receive the hormone regimen.
The significant finding suggests that estrogen may fuel non–small cell lung cancers in both men and women, Dr. Hyunseok Kang reported at the annual meeting of the American Association for Cancer Research.
"Recent literature suggests estrogen receptors and aromatase play a role in the development of non–small cell lung cancers in women with breast cancer," Dr. Kang said. "Aromatase can convert testosterone to estrogen – which could increase the same risk in men."
This recently observed association prompted Dr. Kang, a fellow at Emory University, Atlanta, and his colleagues to investigate whether a similar hormone-mediated link might exist for men with prostate cancer – and whether blocking it could also block lung cancer.
The retrospective observational study pulled data from the Veterans Affairs Central Cancer Registry. It comprised 62,589 men who were diagnosed with prostate cancer between 1999 and 2008, with follow-up through 2010.
There were some significant – and important – baseline differences between the 20,378 men who underwent androgen blockade and those who did not. Men who received the treatment were significantly older (mean age, 71 years vs. 66 years) and slightly more likely to have ever smoked tobacco (73% vs. 71%).
Prostate cancer stage at diagnosis was also significantly different, with fewer of the hormone therapy group presenting at stage II (77% vs. 92%) and more presenting at stage IV (17% vs. 2%).
Men from the VA database were significantly more likely than the general population to develop lung cancer, whether they underwent androgen blockade therapy or not (1.8/100,000 men who received treatment and 2.4/100,000 men who did not receive treatment vs. 0.80/100,000 in the general population).
Of men in the VA database who received androgen treatment, 258 developed secondary lung cancers, compared with 508 men who did not receive treatment.
In an unadjusted model, men who had treatment were 18% less likely to develop a secondary lung cancer (hazard ratio, 0.82). That finding held in a multivariate analysis that adjusted for smoking and age (HR, 0.83).
The study was sponsored by the Veterans Health Administration. Dr. Kang had no financial disclosures.
FROM THE AACR ANNUAL MEETING
Major finding: Men with prostate cancer who underwent androgen blockade were 17% less likely to develop secondary non–small cell lung cancer than were those who didn’t have the hormone treatment.
Data source: The observational study comprised 62,589 men who were diagnosed with prostate cancer during 1998-2008, with follow-up through 2010.
Disclosures: The Veterans Health Administration sponsored the study. Dr. Kang had no financial disclosures.
One in four melanoma patients shun sunscreen
WASHINGTON – While melanoma survivors appear to be more aware of sun safety than does the general public, more than a quarter do not regularly use sunscreen.
"We know that melanoma is a malignancy prevalent in our population, and we know that for many people with melanoma, sun exposure is a major risk factor for recurrence; and sun protection may reduce their chances of getting melanoma again," Dr. Anees B. Chagpar said at the annual meeting of the American Association for Cancer Research. "Although we found that melanoma survivors did better than the general public at protecting their skin from the sun, we also found that more than a quarter of melanoma survivors never wear sunscreen. That blew my mind."
A few survivors – about 2% – even frequent tanning salons, said Dr. Chagpar, an associate professor of surgery at Yale University, New Haven, Conn.
Dr. Chagpar and her colleagues based their findings on data extracted from the 2010 National Health Interview Survey. They focused on self-reported history of melanoma, sun protection practices, and indoor tanning.
The 2010 survey included information on 27,120 adults; 171 had a history of melanoma. Of the adults included in the survey, most (55%) were men; 10% were younger than 40 years.
Compared with the general population, melanoma survivors demonstrated an overall increased rate of sun awareness. Significantly more melanoma survivors reported that they always stay in the shade, compared with the general population (16% vs. 10%, respectively). They were significantly more likely to always wear a baseball cap or visor (31% vs. 18%), a wide-brimmed hat (20% vs. 6%), and a long-sleeved shirt (12% vs. 5%) when going outside on a warm, sunny day for more than 1 hour. They were significantly more likely to report always using sunscreen (32% vs. 17%).
However, Dr. Chagpar said, a good proportion of melanoma survivors are not adequately protecting themselves from sun exposure. About 15% reported rarely or never staying in the shade, and 27% reported never wearing sunscreen.
"The bright spot in this story is that melanoma survivors are more likely to use sunscreen than nonmelanoma survivors," she said in an interview. "But when over a quarter of melanoma survivors admit that they never use sunscreen, we have considerable work to do in educating people about the importance of sun protective behaviors."
And while melanoma survivors overall were significantly less likely to use indoor tanning devices, 2% still reported having done so in the past 12 moths.
"It is distressing to know that melanoma survivors continue to tan," she said. "We have to do a better job in educating our patients about the risks of UV irradiation and the risk of developing melanoma – particularly in those who have survived the disease once already."
Dr. Chagpar had no financial disclosures.
WASHINGTON – While melanoma survivors appear to be more aware of sun safety than does the general public, more than a quarter do not regularly use sunscreen.
"We know that melanoma is a malignancy prevalent in our population, and we know that for many people with melanoma, sun exposure is a major risk factor for recurrence; and sun protection may reduce their chances of getting melanoma again," Dr. Anees B. Chagpar said at the annual meeting of the American Association for Cancer Research. "Although we found that melanoma survivors did better than the general public at protecting their skin from the sun, we also found that more than a quarter of melanoma survivors never wear sunscreen. That blew my mind."
A few survivors – about 2% – even frequent tanning salons, said Dr. Chagpar, an associate professor of surgery at Yale University, New Haven, Conn.
Dr. Chagpar and her colleagues based their findings on data extracted from the 2010 National Health Interview Survey. They focused on self-reported history of melanoma, sun protection practices, and indoor tanning.
The 2010 survey included information on 27,120 adults; 171 had a history of melanoma. Of the adults included in the survey, most (55%) were men; 10% were younger than 40 years.
Compared with the general population, melanoma survivors demonstrated an overall increased rate of sun awareness. Significantly more melanoma survivors reported that they always stay in the shade, compared with the general population (16% vs. 10%, respectively). They were significantly more likely to always wear a baseball cap or visor (31% vs. 18%), a wide-brimmed hat (20% vs. 6%), and a long-sleeved shirt (12% vs. 5%) when going outside on a warm, sunny day for more than 1 hour. They were significantly more likely to report always using sunscreen (32% vs. 17%).
However, Dr. Chagpar said, a good proportion of melanoma survivors are not adequately protecting themselves from sun exposure. About 15% reported rarely or never staying in the shade, and 27% reported never wearing sunscreen.
"The bright spot in this story is that melanoma survivors are more likely to use sunscreen than nonmelanoma survivors," she said in an interview. "But when over a quarter of melanoma survivors admit that they never use sunscreen, we have considerable work to do in educating people about the importance of sun protective behaviors."
And while melanoma survivors overall were significantly less likely to use indoor tanning devices, 2% still reported having done so in the past 12 moths.
"It is distressing to know that melanoma survivors continue to tan," she said. "We have to do a better job in educating our patients about the risks of UV irradiation and the risk of developing melanoma – particularly in those who have survived the disease once already."
Dr. Chagpar had no financial disclosures.
WASHINGTON – While melanoma survivors appear to be more aware of sun safety than does the general public, more than a quarter do not regularly use sunscreen.
"We know that melanoma is a malignancy prevalent in our population, and we know that for many people with melanoma, sun exposure is a major risk factor for recurrence; and sun protection may reduce their chances of getting melanoma again," Dr. Anees B. Chagpar said at the annual meeting of the American Association for Cancer Research. "Although we found that melanoma survivors did better than the general public at protecting their skin from the sun, we also found that more than a quarter of melanoma survivors never wear sunscreen. That blew my mind."
A few survivors – about 2% – even frequent tanning salons, said Dr. Chagpar, an associate professor of surgery at Yale University, New Haven, Conn.
Dr. Chagpar and her colleagues based their findings on data extracted from the 2010 National Health Interview Survey. They focused on self-reported history of melanoma, sun protection practices, and indoor tanning.
The 2010 survey included information on 27,120 adults; 171 had a history of melanoma. Of the adults included in the survey, most (55%) were men; 10% were younger than 40 years.
Compared with the general population, melanoma survivors demonstrated an overall increased rate of sun awareness. Significantly more melanoma survivors reported that they always stay in the shade, compared with the general population (16% vs. 10%, respectively). They were significantly more likely to always wear a baseball cap or visor (31% vs. 18%), a wide-brimmed hat (20% vs. 6%), and a long-sleeved shirt (12% vs. 5%) when going outside on a warm, sunny day for more than 1 hour. They were significantly more likely to report always using sunscreen (32% vs. 17%).
However, Dr. Chagpar said, a good proportion of melanoma survivors are not adequately protecting themselves from sun exposure. About 15% reported rarely or never staying in the shade, and 27% reported never wearing sunscreen.
"The bright spot in this story is that melanoma survivors are more likely to use sunscreen than nonmelanoma survivors," she said in an interview. "But when over a quarter of melanoma survivors admit that they never use sunscreen, we have considerable work to do in educating people about the importance of sun protective behaviors."
And while melanoma survivors overall were significantly less likely to use indoor tanning devices, 2% still reported having done so in the past 12 moths.
"It is distressing to know that melanoma survivors continue to tan," she said. "We have to do a better job in educating our patients about the risks of UV irradiation and the risk of developing melanoma – particularly in those who have survived the disease once already."
Dr. Chagpar had no financial disclosures.
FROM THE AACR ANNUAL MEETING
Major finding: About 27% of melanoma survivors reported never wearing sunscreen, and 2% reported using a tanning bed.
Data source: A database review comparing sun safety behaviors between 171 melanoma survivors and almost 27,000 people who never had the cancer.
Disclosures: Dr. Chagpar had no financial disclosures.
Mammography screening at 75 may have value
WASHINGTON – For women aged 75 years and older, a span of 5 or more years between mammograms translated into a threefold increased risk of death from breast cancer, compared with women in the same age group who had yearly exams.
While the exact reasons for the finding can’t be pinpointed, it should at least reawaken the discussion about whether older women should get a regular mammogram, Dr. Michael Simon noted at the annual meeting of the American Association for Cancer Research.
In 2009, the U.S. Preventive Services Task Force changed its recommendation on screening exams for women aged 75 and older, noting that there was insufficient evidence to recommend for or against breast cancer screening in this population (Ann. Intern. Med. 2009;151:716-26).
However, the absence of evidence does not automatically translate into absence of benefit, Dr. Simon said in an interview. "You can’t assume that at 75, it’s not worth screening anymore. Yes, there’s no evidence from randomized trials of women older than 75, and the recommendations are based on this kind of evidence. But just because there are no trials doesn’t mean there is no benefit."
Dr. Simon and his coinvestigators extracted data from the 15-year Women’s Health Initiative study. The cohort comprised 9,057 women who had been diagnosed with breast cancer over a 12-year period.
The multivariate analysis examined the relationship between death from breast cancer and mammography screening intervals. The screening intervals studied were from 6 months to 1 year, 1 to 2 years, 2 to 5 years, and more than 5 years or never. The study cohort was divided into women by age: 50-74 years (6,497) and 75 years or older (2,560).
Baseline characteristics showed that the older group of women presented with dangerous breast cancers almost as often as did the younger group, including moderately differentiated (22% vs. 21%, respectively) and poorly differentiated tumors (28% vs. 31%). Older women were more likely to have hormone receptor–positive tumors (75% vs. 67%), and just as likely to have hormone receptor–negative tumors (12% of each group).
Stage was also similar between the older and younger groups: in situ (18% vs. 20%, respectively); localized (62% vs. 59%); regional (17% vs. 19%); and distant (1% in both groups).
Significant differences occurred when the investigators broke the group of more than 9,000 women out by mammogram intervals.
Overall, an interval of 5 or more years between a woman’s last mammogram and breast cancer diagnosis was associated with advanced-stage disease in 23%, compared with 20% in women with an interval of 1 year or less – a statistically significant difference, which could affect large numbers of women, said Dr. Simon, head of the breast multidisciplinary team at Barbara Ann Karmanos Cancer Institute in Detroit.
"We also saw significantly more aggressive cancers that were hormone receptor negative in the group with the longest interval between mammograms," he said (22% vs. 16%).
The multivariate analysis adjusted for age at enrollment in the WHI study and the trial component in which subjects enrolled, age at diagnosis, race and ethnicity, insurance and marital status, comorbidities, and body mass index at baseline.
In this analysis, women aged 75 years and older who went at least 5 years between mammograms were 67% more likely to die from a breast cancer than were those who had yearly exams. Older women who went 5 or more years between mammograms were three times more likely to die than were those who had yearly screening, Dr. Simon said.
The results show a need for stronger emphasis on regular mammograms for older women, he said. "Women are living longer and living vitally longer. If we have a test that can identify an early cancer ... we should take advantage of it."
For women of any age, mammograms have their downsides, including anxiety while waiting for results, overdiagnosis, and treatment of indolent, in situ tumors that may never progress to serious cancers – especially in women with a shorter lifespan ahead of them. Dr. Simon said those risks are small compared with the benefit of finding and treating a potentially lethal tumor.
Cost, however, is the sticking point, admitted Dr. Simon, who is also a public health expert.
"Resource allocation is something we can’t ignore. We know that it takes 1,700 mammograms to benefit one woman. That sounds like a lot, but when you think about it in a global perspective – how many women there are in this country – you see it in a different light. What we’re paying for mammograms is small potatoes."
Dr. Simon had no financial disclosures.
WASHINGTON – For women aged 75 years and older, a span of 5 or more years between mammograms translated into a threefold increased risk of death from breast cancer, compared with women in the same age group who had yearly exams.
While the exact reasons for the finding can’t be pinpointed, it should at least reawaken the discussion about whether older women should get a regular mammogram, Dr. Michael Simon noted at the annual meeting of the American Association for Cancer Research.
In 2009, the U.S. Preventive Services Task Force changed its recommendation on screening exams for women aged 75 and older, noting that there was insufficient evidence to recommend for or against breast cancer screening in this population (Ann. Intern. Med. 2009;151:716-26).
However, the absence of evidence does not automatically translate into absence of benefit, Dr. Simon said in an interview. "You can’t assume that at 75, it’s not worth screening anymore. Yes, there’s no evidence from randomized trials of women older than 75, and the recommendations are based on this kind of evidence. But just because there are no trials doesn’t mean there is no benefit."
Dr. Simon and his coinvestigators extracted data from the 15-year Women’s Health Initiative study. The cohort comprised 9,057 women who had been diagnosed with breast cancer over a 12-year period.
The multivariate analysis examined the relationship between death from breast cancer and mammography screening intervals. The screening intervals studied were from 6 months to 1 year, 1 to 2 years, 2 to 5 years, and more than 5 years or never. The study cohort was divided into women by age: 50-74 years (6,497) and 75 years or older (2,560).
Baseline characteristics showed that the older group of women presented with dangerous breast cancers almost as often as did the younger group, including moderately differentiated (22% vs. 21%, respectively) and poorly differentiated tumors (28% vs. 31%). Older women were more likely to have hormone receptor–positive tumors (75% vs. 67%), and just as likely to have hormone receptor–negative tumors (12% of each group).
Stage was also similar between the older and younger groups: in situ (18% vs. 20%, respectively); localized (62% vs. 59%); regional (17% vs. 19%); and distant (1% in both groups).
Significant differences occurred when the investigators broke the group of more than 9,000 women out by mammogram intervals.
Overall, an interval of 5 or more years between a woman’s last mammogram and breast cancer diagnosis was associated with advanced-stage disease in 23%, compared with 20% in women with an interval of 1 year or less – a statistically significant difference, which could affect large numbers of women, said Dr. Simon, head of the breast multidisciplinary team at Barbara Ann Karmanos Cancer Institute in Detroit.
"We also saw significantly more aggressive cancers that were hormone receptor negative in the group with the longest interval between mammograms," he said (22% vs. 16%).
The multivariate analysis adjusted for age at enrollment in the WHI study and the trial component in which subjects enrolled, age at diagnosis, race and ethnicity, insurance and marital status, comorbidities, and body mass index at baseline.
In this analysis, women aged 75 years and older who went at least 5 years between mammograms were 67% more likely to die from a breast cancer than were those who had yearly exams. Older women who went 5 or more years between mammograms were three times more likely to die than were those who had yearly screening, Dr. Simon said.
The results show a need for stronger emphasis on regular mammograms for older women, he said. "Women are living longer and living vitally longer. If we have a test that can identify an early cancer ... we should take advantage of it."
For women of any age, mammograms have their downsides, including anxiety while waiting for results, overdiagnosis, and treatment of indolent, in situ tumors that may never progress to serious cancers – especially in women with a shorter lifespan ahead of them. Dr. Simon said those risks are small compared with the benefit of finding and treating a potentially lethal tumor.
Cost, however, is the sticking point, admitted Dr. Simon, who is also a public health expert.
"Resource allocation is something we can’t ignore. We know that it takes 1,700 mammograms to benefit one woman. That sounds like a lot, but when you think about it in a global perspective – how many women there are in this country – you see it in a different light. What we’re paying for mammograms is small potatoes."
Dr. Simon had no financial disclosures.
WASHINGTON – For women aged 75 years and older, a span of 5 or more years between mammograms translated into a threefold increased risk of death from breast cancer, compared with women in the same age group who had yearly exams.
While the exact reasons for the finding can’t be pinpointed, it should at least reawaken the discussion about whether older women should get a regular mammogram, Dr. Michael Simon noted at the annual meeting of the American Association for Cancer Research.
In 2009, the U.S. Preventive Services Task Force changed its recommendation on screening exams for women aged 75 and older, noting that there was insufficient evidence to recommend for or against breast cancer screening in this population (Ann. Intern. Med. 2009;151:716-26).
However, the absence of evidence does not automatically translate into absence of benefit, Dr. Simon said in an interview. "You can’t assume that at 75, it’s not worth screening anymore. Yes, there’s no evidence from randomized trials of women older than 75, and the recommendations are based on this kind of evidence. But just because there are no trials doesn’t mean there is no benefit."
Dr. Simon and his coinvestigators extracted data from the 15-year Women’s Health Initiative study. The cohort comprised 9,057 women who had been diagnosed with breast cancer over a 12-year period.
The multivariate analysis examined the relationship between death from breast cancer and mammography screening intervals. The screening intervals studied were from 6 months to 1 year, 1 to 2 years, 2 to 5 years, and more than 5 years or never. The study cohort was divided into women by age: 50-74 years (6,497) and 75 years or older (2,560).
Baseline characteristics showed that the older group of women presented with dangerous breast cancers almost as often as did the younger group, including moderately differentiated (22% vs. 21%, respectively) and poorly differentiated tumors (28% vs. 31%). Older women were more likely to have hormone receptor–positive tumors (75% vs. 67%), and just as likely to have hormone receptor–negative tumors (12% of each group).
Stage was also similar between the older and younger groups: in situ (18% vs. 20%, respectively); localized (62% vs. 59%); regional (17% vs. 19%); and distant (1% in both groups).
Significant differences occurred when the investigators broke the group of more than 9,000 women out by mammogram intervals.
Overall, an interval of 5 or more years between a woman’s last mammogram and breast cancer diagnosis was associated with advanced-stage disease in 23%, compared with 20% in women with an interval of 1 year or less – a statistically significant difference, which could affect large numbers of women, said Dr. Simon, head of the breast multidisciplinary team at Barbara Ann Karmanos Cancer Institute in Detroit.
"We also saw significantly more aggressive cancers that were hormone receptor negative in the group with the longest interval between mammograms," he said (22% vs. 16%).
The multivariate analysis adjusted for age at enrollment in the WHI study and the trial component in which subjects enrolled, age at diagnosis, race and ethnicity, insurance and marital status, comorbidities, and body mass index at baseline.
In this analysis, women aged 75 years and older who went at least 5 years between mammograms were 67% more likely to die from a breast cancer than were those who had yearly exams. Older women who went 5 or more years between mammograms were three times more likely to die than were those who had yearly screening, Dr. Simon said.
The results show a need for stronger emphasis on regular mammograms for older women, he said. "Women are living longer and living vitally longer. If we have a test that can identify an early cancer ... we should take advantage of it."
For women of any age, mammograms have their downsides, including anxiety while waiting for results, overdiagnosis, and treatment of indolent, in situ tumors that may never progress to serious cancers – especially in women with a shorter lifespan ahead of them. Dr. Simon said those risks are small compared with the benefit of finding and treating a potentially lethal tumor.
Cost, however, is the sticking point, admitted Dr. Simon, who is also a public health expert.
"Resource allocation is something we can’t ignore. We know that it takes 1,700 mammograms to benefit one woman. That sounds like a lot, but when you think about it in a global perspective – how many women there are in this country – you see it in a different light. What we’re paying for mammograms is small potatoes."
Dr. Simon had no financial disclosures.
AT THE AACR ANNUAL MEETING
Major finding: Women aged 75 years and older were 3 times more likely to die from breast cancer if they went 5 or more years between mammograms, compared with those who had a yearly screening.
Data source: Women’s Health Initiative data on more than 9,000 women diagnosed with breast cancer.
Disclosures: Dr. Simon had no financial disclosures.
Immunotherapy boosts response in recurrent ovarian cancer
WASHINGTON – A novel ovarian cancer treatment that uses the patient’s own tumor to stimulate her immune system provoked a positive response in 66% of women with advanced stage disease – including 20 patients who had no obvious disease at the end of treatment, and one who is still in complete remission nearly 4 years later.
When researchers added a second step of T-cell reprogramming for patients with residual disease, clinical benefit was seen in about 75% of patients, Lana Kandalaft, Ph.D., said during a press briefing at the annual meeting of the American Association of Cancer Research.
"This is the first time such a combination immunotherapy approach has been used for patients with ovarian cancer," said Dr. Kandalaft of the University of Pennsylvania. "Most patients with ovarian cancer are diagnosed at an advanced stage and many of those relapse within 2 years; most die within 5 years. Given these grim outcomes, there is definitely a vast unmet need for the development of novel, alternate therapies."
The team reported on its first 6 patients in January (Oncoimmunology. 2013;2: e22664). Four patients achieved clinical benefits, defined as tumor shrinkage of at least 30%. Two had a partial response; one with a partial response had previously progressed on chemotherapy alone but improved after the vaccine was added to her treatment, suggesting that immunomodulation conferred an additional therapeutic benefit.
Two other subjects exhibited stable disease. One of these still in remission. The last two patients continued to progress.
At the press briefing, Dr. Kandalaft reported on 31 patients – the original cohort plus 25 more women. All had recurrent progressive stage III or IV ovarian cancer and had undergone surgical reduction with tumor cryopreservation. The immunotherapy regimen began after each patient completed chemotherapy.
At that point, Dr. Kandalaft and her colleagues prepared a tumor lysate and used it to prime harvested dendritic cells. Patients received this individualized vaccine through intranodal injections in the groin, along with bevacizumab and cyclophosphamide, every 2 weeks for 3 months.
Eleven patients who responded to the vaccine treatment but still had residual disease moved to a second step: 3 months of adoptive T-cell therapy. Dr. Kandalaft and her colleagues removed T-cells, which had been educated in the body by the tumor-stimulated dendritic cells, and expanded these in culture. They then transferred the T-cells back to the patient, creating a population completely primed to attack the tumor.
Of these 11 patients, seven showed stable disease and one had a complete response.
Dr. Kandalaft noted that there is set length for this treatment, because each patient’s vaccine amount is limited by her tumor’s size and characteristics. During the follow-up period, patients receive the treatment until their supply is exhausted. The woman who remains in remission has been without vaccine for about a year, Dr. Kandalaft noted.
The team continues to refine the protocol. Some patients are now taking aspirin in addition to bevacizumab and cyclophosphamide. "We have some evidence from the lab that aspirin opens the endothelial barrier on the tumor and makes it easier for the T cells to attack."
Side effects are common, but so far have been mild or moderate; most were flu-like symptoms as well as the adverse events usually associated with bevacizumab and cyclophosphamide.
The study continues to accrue patients, Dr. Kandalaft said. Although other facilities are not approved to administer the vaccine, women with recurrent stage III or IV ovarian cancer who meet the eligibility criteria can still participate.
"These women would have their surgery at their own facility, and have the tumor cryopreserved according to our specifications, and then sent to us," Dr. Kandalaft said. "Then we would prepare the lysate and the woman would come to us for the remainder of her treatment.
She hopes to include a new set of patients as well – those who are in early remission after their primary treatment. "The immune system is healthier in this setting," she said. "For these women we will be trying to boost that system to prevent a recurrence in the first place. We think that is population who would get the biggest benefit from this treatment."
The study was funded by a National Cancer Institute Ovarian Specialized Program of Research Excellence grant, the National Institutes of Health and the Ovarian Cancer Immunotherapy Initiative.
WASHINGTON – A novel ovarian cancer treatment that uses the patient’s own tumor to stimulate her immune system provoked a positive response in 66% of women with advanced stage disease – including 20 patients who had no obvious disease at the end of treatment, and one who is still in complete remission nearly 4 years later.
When researchers added a second step of T-cell reprogramming for patients with residual disease, clinical benefit was seen in about 75% of patients, Lana Kandalaft, Ph.D., said during a press briefing at the annual meeting of the American Association of Cancer Research.
"This is the first time such a combination immunotherapy approach has been used for patients with ovarian cancer," said Dr. Kandalaft of the University of Pennsylvania. "Most patients with ovarian cancer are diagnosed at an advanced stage and many of those relapse within 2 years; most die within 5 years. Given these grim outcomes, there is definitely a vast unmet need for the development of novel, alternate therapies."
The team reported on its first 6 patients in January (Oncoimmunology. 2013;2: e22664). Four patients achieved clinical benefits, defined as tumor shrinkage of at least 30%. Two had a partial response; one with a partial response had previously progressed on chemotherapy alone but improved after the vaccine was added to her treatment, suggesting that immunomodulation conferred an additional therapeutic benefit.
Two other subjects exhibited stable disease. One of these still in remission. The last two patients continued to progress.
At the press briefing, Dr. Kandalaft reported on 31 patients – the original cohort plus 25 more women. All had recurrent progressive stage III or IV ovarian cancer and had undergone surgical reduction with tumor cryopreservation. The immunotherapy regimen began after each patient completed chemotherapy.
At that point, Dr. Kandalaft and her colleagues prepared a tumor lysate and used it to prime harvested dendritic cells. Patients received this individualized vaccine through intranodal injections in the groin, along with bevacizumab and cyclophosphamide, every 2 weeks for 3 months.
Eleven patients who responded to the vaccine treatment but still had residual disease moved to a second step: 3 months of adoptive T-cell therapy. Dr. Kandalaft and her colleagues removed T-cells, which had been educated in the body by the tumor-stimulated dendritic cells, and expanded these in culture. They then transferred the T-cells back to the patient, creating a population completely primed to attack the tumor.
Of these 11 patients, seven showed stable disease and one had a complete response.
Dr. Kandalaft noted that there is set length for this treatment, because each patient’s vaccine amount is limited by her tumor’s size and characteristics. During the follow-up period, patients receive the treatment until their supply is exhausted. The woman who remains in remission has been without vaccine for about a year, Dr. Kandalaft noted.
The team continues to refine the protocol. Some patients are now taking aspirin in addition to bevacizumab and cyclophosphamide. "We have some evidence from the lab that aspirin opens the endothelial barrier on the tumor and makes it easier for the T cells to attack."
Side effects are common, but so far have been mild or moderate; most were flu-like symptoms as well as the adverse events usually associated with bevacizumab and cyclophosphamide.
The study continues to accrue patients, Dr. Kandalaft said. Although other facilities are not approved to administer the vaccine, women with recurrent stage III or IV ovarian cancer who meet the eligibility criteria can still participate.
"These women would have their surgery at their own facility, and have the tumor cryopreserved according to our specifications, and then sent to us," Dr. Kandalaft said. "Then we would prepare the lysate and the woman would come to us for the remainder of her treatment.
She hopes to include a new set of patients as well – those who are in early remission after their primary treatment. "The immune system is healthier in this setting," she said. "For these women we will be trying to boost that system to prevent a recurrence in the first place. We think that is population who would get the biggest benefit from this treatment."
The study was funded by a National Cancer Institute Ovarian Specialized Program of Research Excellence grant, the National Institutes of Health and the Ovarian Cancer Immunotherapy Initiative.
WASHINGTON – A novel ovarian cancer treatment that uses the patient’s own tumor to stimulate her immune system provoked a positive response in 66% of women with advanced stage disease – including 20 patients who had no obvious disease at the end of treatment, and one who is still in complete remission nearly 4 years later.
When researchers added a second step of T-cell reprogramming for patients with residual disease, clinical benefit was seen in about 75% of patients, Lana Kandalaft, Ph.D., said during a press briefing at the annual meeting of the American Association of Cancer Research.
"This is the first time such a combination immunotherapy approach has been used for patients with ovarian cancer," said Dr. Kandalaft of the University of Pennsylvania. "Most patients with ovarian cancer are diagnosed at an advanced stage and many of those relapse within 2 years; most die within 5 years. Given these grim outcomes, there is definitely a vast unmet need for the development of novel, alternate therapies."
The team reported on its first 6 patients in January (Oncoimmunology. 2013;2: e22664). Four patients achieved clinical benefits, defined as tumor shrinkage of at least 30%. Two had a partial response; one with a partial response had previously progressed on chemotherapy alone but improved after the vaccine was added to her treatment, suggesting that immunomodulation conferred an additional therapeutic benefit.
Two other subjects exhibited stable disease. One of these still in remission. The last two patients continued to progress.
At the press briefing, Dr. Kandalaft reported on 31 patients – the original cohort plus 25 more women. All had recurrent progressive stage III or IV ovarian cancer and had undergone surgical reduction with tumor cryopreservation. The immunotherapy regimen began after each patient completed chemotherapy.
At that point, Dr. Kandalaft and her colleagues prepared a tumor lysate and used it to prime harvested dendritic cells. Patients received this individualized vaccine through intranodal injections in the groin, along with bevacizumab and cyclophosphamide, every 2 weeks for 3 months.
Eleven patients who responded to the vaccine treatment but still had residual disease moved to a second step: 3 months of adoptive T-cell therapy. Dr. Kandalaft and her colleagues removed T-cells, which had been educated in the body by the tumor-stimulated dendritic cells, and expanded these in culture. They then transferred the T-cells back to the patient, creating a population completely primed to attack the tumor.
Of these 11 patients, seven showed stable disease and one had a complete response.
Dr. Kandalaft noted that there is set length for this treatment, because each patient’s vaccine amount is limited by her tumor’s size and characteristics. During the follow-up period, patients receive the treatment until their supply is exhausted. The woman who remains in remission has been without vaccine for about a year, Dr. Kandalaft noted.
The team continues to refine the protocol. Some patients are now taking aspirin in addition to bevacizumab and cyclophosphamide. "We have some evidence from the lab that aspirin opens the endothelial barrier on the tumor and makes it easier for the T cells to attack."
Side effects are common, but so far have been mild or moderate; most were flu-like symptoms as well as the adverse events usually associated with bevacizumab and cyclophosphamide.
The study continues to accrue patients, Dr. Kandalaft said. Although other facilities are not approved to administer the vaccine, women with recurrent stage III or IV ovarian cancer who meet the eligibility criteria can still participate.
"These women would have their surgery at their own facility, and have the tumor cryopreserved according to our specifications, and then sent to us," Dr. Kandalaft said. "Then we would prepare the lysate and the woman would come to us for the remainder of her treatment.
She hopes to include a new set of patients as well – those who are in early remission after their primary treatment. "The immune system is healthier in this setting," she said. "For these women we will be trying to boost that system to prevent a recurrence in the first place. We think that is population who would get the biggest benefit from this treatment."
The study was funded by a National Cancer Institute Ovarian Specialized Program of Research Excellence grant, the National Institutes of Health and the Ovarian Cancer Immunotherapy Initiative.
AT THE AACR ANNUAL MEETING
Major finding: A two-step immunotherapy regimen combined with chemotherapy had up to a 75% response rate for women with recurrent stage III or IV ovarian cancer.
Data source: The study comprised 31 women who underwent dendritic cell priming with tumor lysate; 11 of these also underwent T-cell reprogramming.
Disclosures: The study was funded by a National Cancer Institute Ovarian Specialized Program of Research Excellence grant, the National Institutes of Health, and the Ovarian Cancer Immunotherapy Initiative.
New blood test could identify early pancreatic cancer
An investigational serum biomarker panel accurately identified potentially resectable cases of pancreatic cancer, opening the possibility that a blood test could identify the cancer early and improve its dismal prognosis.
The panel measured four metabolites and had an overall sensitivity of 71% and a specificity of 78% for pancreatic cancer; the specificity for resectable tumors also approached 78%, reported Dr. Masaru Yoshida (Cancer Epidemiol. Biomarkers Prev. 2013 March 29 [doi: 10.1158/1055-9965.EPI-12-1033]).
The panel was more accurate than were any of the currently used biomarkers, including CA19-9 and CEA, said Dr. Yoshida, professor and chief of metabolomics research at Kobe University, Japan.
"This novel diagnostic approach, which is safe and easy to apply as a screening method, is expected to improve the prognosis of patients with pancreatic cancer by detecting their cancers early, when still in a resectable and curable state," Dr. Yoshida said in a press statement.
The researchers created the panel based on study of a cohort of 85 subjects: 43 with pancreatic cancer and 42 healthy controls. First, they pared down 113 potentially useful metabolites into a workable testing panel. Of the 45 candidate metabolites, four of them – xylitol, 1,5-anhydro-D-glucitol, histidine, and inositol – showed the highest variation between the healthy controls and the patients. In the training cohort, the panel of these four markers had a sensitivity of 86% and specificity of 88% for pancreatic cancer.
Next, the researchers evaluated a validation cohort of 42 cancer patients, 41 healthy controls, and 23 patients with chronic pancreatitis. In this cohort, with an area under the curve of 0.76, the panel’s sensitivity for pancreatic cancer was 71% and specificity, 78%. In contrast, the sensitivity of CA19-9 was 69% and specificity 86%; for CEA, those numbers were 36% and 80%, respectively.
In the subset of patients with resectable pancreatic cancer, the panel’s sensitivity was 78%, compared with 56% for CA19-9 and 44% for CEA.
The metabolic panel was also able to identify chronic pancreatitis, with a 17% rate of false positives, compared with a false positive rate of 30% for CA19-9 and 44% for CEA.
A blood panel could address three key problems in the field of pancreatic cancer, Dr. Yoshida and his colleagues said. "The first is the difficulty of detecting pancreatic cancer early in resectable stages. A sensitivity and specificity of about 80% for resectable disease ... should be acceptable for clinical use because most patients with resectable cancer have no symptoms, and blood examinations are useful tools for initial screening examinations."
The second problem is the difficulty in differentiating pancreatic cancer from chronic pancreatitis. "Many gastroenterologists follow up chronic pancreatitis with scheduled CT, magnetic resonance imaging (MRI), and endoscopic ultrasound scans (EUS); tumor marker tests; and endoscopic retrograde cholangiopancreatography (ERCP), but the initial malignant changes are frequently overlooked, and pancreatic tumors can rapidly become unresectable. In addition, unnecessary resections for benign inflammatory lesions are sometimes done because of false-positive results from CA19-9 and/or imaging examinations."
The third clinical problem is the risk of complications that result from pancreatic examinations. Serum samples are easy and safe to obtain, with a low risk of adverse events and, with this panel, a potentially good return of information. A blood test could also be a useful population screening tool, the researchers noted.
The metabolites probably reflect metabolic derangement that arises not only from focal tumorigenesis, but also from systemic reactions to pancreatic disease, Dr. Yoshida and his coinvestigators wrote. Pancreatic cancer causes significant decreases in some amino and fatty acids, probably because the tumor recruits these substances to aid its rapid cellular proliferation.
"Patients with pancreatic disease are also troubled by malnutrition because of pancreatic endocrine and exocrine insufficiency. Therefore, there is a possibility that the decreases in serum metabolite levels also reflect malnutrition."
Finally, they said, the decrease in 1,5-anhydro-D-glucitol indicates the presence of hyperglycemia and recent glycosuria. "These results suggest that glucose tolerance was impaired in these patients because of pancreatic insufficiency."
The authors reported no financial conflicts. The work was supported by grants administered by agencies of the Japanese government.
An investigational serum biomarker panel accurately identified potentially resectable cases of pancreatic cancer, opening the possibility that a blood test could identify the cancer early and improve its dismal prognosis.
The panel measured four metabolites and had an overall sensitivity of 71% and a specificity of 78% for pancreatic cancer; the specificity for resectable tumors also approached 78%, reported Dr. Masaru Yoshida (Cancer Epidemiol. Biomarkers Prev. 2013 March 29 [doi: 10.1158/1055-9965.EPI-12-1033]).
The panel was more accurate than were any of the currently used biomarkers, including CA19-9 and CEA, said Dr. Yoshida, professor and chief of metabolomics research at Kobe University, Japan.
"This novel diagnostic approach, which is safe and easy to apply as a screening method, is expected to improve the prognosis of patients with pancreatic cancer by detecting their cancers early, when still in a resectable and curable state," Dr. Yoshida said in a press statement.
The researchers created the panel based on study of a cohort of 85 subjects: 43 with pancreatic cancer and 42 healthy controls. First, they pared down 113 potentially useful metabolites into a workable testing panel. Of the 45 candidate metabolites, four of them – xylitol, 1,5-anhydro-D-glucitol, histidine, and inositol – showed the highest variation between the healthy controls and the patients. In the training cohort, the panel of these four markers had a sensitivity of 86% and specificity of 88% for pancreatic cancer.
Next, the researchers evaluated a validation cohort of 42 cancer patients, 41 healthy controls, and 23 patients with chronic pancreatitis. In this cohort, with an area under the curve of 0.76, the panel’s sensitivity for pancreatic cancer was 71% and specificity, 78%. In contrast, the sensitivity of CA19-9 was 69% and specificity 86%; for CEA, those numbers were 36% and 80%, respectively.
In the subset of patients with resectable pancreatic cancer, the panel’s sensitivity was 78%, compared with 56% for CA19-9 and 44% for CEA.
The metabolic panel was also able to identify chronic pancreatitis, with a 17% rate of false positives, compared with a false positive rate of 30% for CA19-9 and 44% for CEA.
A blood panel could address three key problems in the field of pancreatic cancer, Dr. Yoshida and his colleagues said. "The first is the difficulty of detecting pancreatic cancer early in resectable stages. A sensitivity and specificity of about 80% for resectable disease ... should be acceptable for clinical use because most patients with resectable cancer have no symptoms, and blood examinations are useful tools for initial screening examinations."
The second problem is the difficulty in differentiating pancreatic cancer from chronic pancreatitis. "Many gastroenterologists follow up chronic pancreatitis with scheduled CT, magnetic resonance imaging (MRI), and endoscopic ultrasound scans (EUS); tumor marker tests; and endoscopic retrograde cholangiopancreatography (ERCP), but the initial malignant changes are frequently overlooked, and pancreatic tumors can rapidly become unresectable. In addition, unnecessary resections for benign inflammatory lesions are sometimes done because of false-positive results from CA19-9 and/or imaging examinations."
The third clinical problem is the risk of complications that result from pancreatic examinations. Serum samples are easy and safe to obtain, with a low risk of adverse events and, with this panel, a potentially good return of information. A blood test could also be a useful population screening tool, the researchers noted.
The metabolites probably reflect metabolic derangement that arises not only from focal tumorigenesis, but also from systemic reactions to pancreatic disease, Dr. Yoshida and his coinvestigators wrote. Pancreatic cancer causes significant decreases in some amino and fatty acids, probably because the tumor recruits these substances to aid its rapid cellular proliferation.
"Patients with pancreatic disease are also troubled by malnutrition because of pancreatic endocrine and exocrine insufficiency. Therefore, there is a possibility that the decreases in serum metabolite levels also reflect malnutrition."
Finally, they said, the decrease in 1,5-anhydro-D-glucitol indicates the presence of hyperglycemia and recent glycosuria. "These results suggest that glucose tolerance was impaired in these patients because of pancreatic insufficiency."
The authors reported no financial conflicts. The work was supported by grants administered by agencies of the Japanese government.
An investigational serum biomarker panel accurately identified potentially resectable cases of pancreatic cancer, opening the possibility that a blood test could identify the cancer early and improve its dismal prognosis.
The panel measured four metabolites and had an overall sensitivity of 71% and a specificity of 78% for pancreatic cancer; the specificity for resectable tumors also approached 78%, reported Dr. Masaru Yoshida (Cancer Epidemiol. Biomarkers Prev. 2013 March 29 [doi: 10.1158/1055-9965.EPI-12-1033]).
The panel was more accurate than were any of the currently used biomarkers, including CA19-9 and CEA, said Dr. Yoshida, professor and chief of metabolomics research at Kobe University, Japan.
"This novel diagnostic approach, which is safe and easy to apply as a screening method, is expected to improve the prognosis of patients with pancreatic cancer by detecting their cancers early, when still in a resectable and curable state," Dr. Yoshida said in a press statement.
The researchers created the panel based on study of a cohort of 85 subjects: 43 with pancreatic cancer and 42 healthy controls. First, they pared down 113 potentially useful metabolites into a workable testing panel. Of the 45 candidate metabolites, four of them – xylitol, 1,5-anhydro-D-glucitol, histidine, and inositol – showed the highest variation between the healthy controls and the patients. In the training cohort, the panel of these four markers had a sensitivity of 86% and specificity of 88% for pancreatic cancer.
Next, the researchers evaluated a validation cohort of 42 cancer patients, 41 healthy controls, and 23 patients with chronic pancreatitis. In this cohort, with an area under the curve of 0.76, the panel’s sensitivity for pancreatic cancer was 71% and specificity, 78%. In contrast, the sensitivity of CA19-9 was 69% and specificity 86%; for CEA, those numbers were 36% and 80%, respectively.
In the subset of patients with resectable pancreatic cancer, the panel’s sensitivity was 78%, compared with 56% for CA19-9 and 44% for CEA.
The metabolic panel was also able to identify chronic pancreatitis, with a 17% rate of false positives, compared with a false positive rate of 30% for CA19-9 and 44% for CEA.
A blood panel could address three key problems in the field of pancreatic cancer, Dr. Yoshida and his colleagues said. "The first is the difficulty of detecting pancreatic cancer early in resectable stages. A sensitivity and specificity of about 80% for resectable disease ... should be acceptable for clinical use because most patients with resectable cancer have no symptoms, and blood examinations are useful tools for initial screening examinations."
The second problem is the difficulty in differentiating pancreatic cancer from chronic pancreatitis. "Many gastroenterologists follow up chronic pancreatitis with scheduled CT, magnetic resonance imaging (MRI), and endoscopic ultrasound scans (EUS); tumor marker tests; and endoscopic retrograde cholangiopancreatography (ERCP), but the initial malignant changes are frequently overlooked, and pancreatic tumors can rapidly become unresectable. In addition, unnecessary resections for benign inflammatory lesions are sometimes done because of false-positive results from CA19-9 and/or imaging examinations."
The third clinical problem is the risk of complications that result from pancreatic examinations. Serum samples are easy and safe to obtain, with a low risk of adverse events and, with this panel, a potentially good return of information. A blood test could also be a useful population screening tool, the researchers noted.
The metabolites probably reflect metabolic derangement that arises not only from focal tumorigenesis, but also from systemic reactions to pancreatic disease, Dr. Yoshida and his coinvestigators wrote. Pancreatic cancer causes significant decreases in some amino and fatty acids, probably because the tumor recruits these substances to aid its rapid cellular proliferation.
"Patients with pancreatic disease are also troubled by malnutrition because of pancreatic endocrine and exocrine insufficiency. Therefore, there is a possibility that the decreases in serum metabolite levels also reflect malnutrition."
Finally, they said, the decrease in 1,5-anhydro-D-glucitol indicates the presence of hyperglycemia and recent glycosuria. "These results suggest that glucose tolerance was impaired in these patients because of pancreatic insufficiency."
The authors reported no financial conflicts. The work was supported by grants administered by agencies of the Japanese government.
FROM CANCER EPIDEMIOLOGY, BIOMARKERS, AND PREVENTION
Major finding: A serum panel of four metabolites had a 78% sensitivity for detecting resectable pancreatic cancers.
Data source: The panel was derived from a test cohort of 85, and a validation cohort of 42 cancer patients, 41 healthy controls, and 23 patients with chronic pancreatitis.
Disclosures: The authors had no financial disclosures. Japanese government agencies funded the study.
Baldness, prostate cancer linked among black men
Baldness seems to confer a significantly increased risk of prostate cancer upon black men – particularly if they lose their hair before age 60 years.
Different patterns of baldness were also related to different grades of cancer, Charnita Zeigler-Johnson, Ph.D., and her colleagues reported in the March 26 online issue of Cancer, Epidemiology, Biomarkers, and Prevention (Canc. Ep. Biomark. Prev. 2013;22: 589-96).Those with frontal baldness were more than twice as likely to have high-grade and high-stage disease at diagnosis than were those with other hair loss patterns, wrote Dr. Zeigler-Johnson of the University of Pennsylvania, Philadelphia, and her coauthors.
The researchers’ case-control study comprised 318 black patients with prostate cancer and 219 black controls. The subjects were matched for age and other baseline characteristics. However, patients were significantly older than controls (60 vs. 57 years), and more likely to report a family history of prostate cancer (36% vs. 27%). Any form of baldness occurred in significantly more patients than in controls (20% vs.13%).
When the investigators conducted a multivariate analysis, they found a number of significant associations between the cancer and hair loss. Compared with those without hair loss, men with any form of baldness were 69% more likely to have prostate cancer. Frontal baldness was associated with more than a doubling in the risk of both high-stage and high-grade disease (odds ratio, 2.61 and 2.20, respectively).
Men with vertex balding who developed prostate cancer were significantly more likely to present with a low-grade tumor (OR, 1.45).
When the authors broke the groups down by age, they found no significant associations with disease severity among men older than 60 years. Instead, these risks were concentrated in men younger than 60 years. Among these, baldness increased the risk of high-stage cancer by more than three times (OR, 3.43) and more than doubled the risk high-grade disease (OR, 2.33). Frontal baldness was a particularly ominous risk factor for younger men, being associated with more than six times the risk of high-stage disease and more than four times the risk of high-grade disease (OR, 6.51 and 4.23, respectively).
There were also significant relationships observed between baldness and prostate specific antigen levels at diagnosis among younger men. Any baldness was associated with a tripling in the risk of a high PSA (10 ng/mL or more) at diagnosis. The association was stronger for men with frontal-only baldness (OR, 5.29).
The authors speculated that the elevated risks are related to genetically determined androgen metabolism. "There are differences in the prevalence of genotypes that metabolize testosterone and influence dihydrotestosterone (DHT) levels," they wrote. "High DHT levels have been associated with both early pattern baldness and prostate cancer processes, including increases in PSA levels."
In particular, they noted, four genes known to be associated with early-onset baldness are also involved in pathways of androgen metabolism, hair development, and age-related neurodegenerative disease.
"Given the high prevalence of prostate cancer in African Americans, early-onset baldness may be a particularly relevant indicator of risk that deserves attention in future studies as we seek to advance our knowledge about high-risk populations."
None of the authors had any financial disclosures. The work was funded by the Department of Defense and the Public Health Service.
Baldness seems to confer a significantly increased risk of prostate cancer upon black men – particularly if they lose their hair before age 60 years.
Different patterns of baldness were also related to different grades of cancer, Charnita Zeigler-Johnson, Ph.D., and her colleagues reported in the March 26 online issue of Cancer, Epidemiology, Biomarkers, and Prevention (Canc. Ep. Biomark. Prev. 2013;22: 589-96).Those with frontal baldness were more than twice as likely to have high-grade and high-stage disease at diagnosis than were those with other hair loss patterns, wrote Dr. Zeigler-Johnson of the University of Pennsylvania, Philadelphia, and her coauthors.
The researchers’ case-control study comprised 318 black patients with prostate cancer and 219 black controls. The subjects were matched for age and other baseline characteristics. However, patients were significantly older than controls (60 vs. 57 years), and more likely to report a family history of prostate cancer (36% vs. 27%). Any form of baldness occurred in significantly more patients than in controls (20% vs.13%).
When the investigators conducted a multivariate analysis, they found a number of significant associations between the cancer and hair loss. Compared with those without hair loss, men with any form of baldness were 69% more likely to have prostate cancer. Frontal baldness was associated with more than a doubling in the risk of both high-stage and high-grade disease (odds ratio, 2.61 and 2.20, respectively).
Men with vertex balding who developed prostate cancer were significantly more likely to present with a low-grade tumor (OR, 1.45).
When the authors broke the groups down by age, they found no significant associations with disease severity among men older than 60 years. Instead, these risks were concentrated in men younger than 60 years. Among these, baldness increased the risk of high-stage cancer by more than three times (OR, 3.43) and more than doubled the risk high-grade disease (OR, 2.33). Frontal baldness was a particularly ominous risk factor for younger men, being associated with more than six times the risk of high-stage disease and more than four times the risk of high-grade disease (OR, 6.51 and 4.23, respectively).
There were also significant relationships observed between baldness and prostate specific antigen levels at diagnosis among younger men. Any baldness was associated with a tripling in the risk of a high PSA (10 ng/mL or more) at diagnosis. The association was stronger for men with frontal-only baldness (OR, 5.29).
The authors speculated that the elevated risks are related to genetically determined androgen metabolism. "There are differences in the prevalence of genotypes that metabolize testosterone and influence dihydrotestosterone (DHT) levels," they wrote. "High DHT levels have been associated with both early pattern baldness and prostate cancer processes, including increases in PSA levels."
In particular, they noted, four genes known to be associated with early-onset baldness are also involved in pathways of androgen metabolism, hair development, and age-related neurodegenerative disease.
"Given the high prevalence of prostate cancer in African Americans, early-onset baldness may be a particularly relevant indicator of risk that deserves attention in future studies as we seek to advance our knowledge about high-risk populations."
None of the authors had any financial disclosures. The work was funded by the Department of Defense and the Public Health Service.
Baldness seems to confer a significantly increased risk of prostate cancer upon black men – particularly if they lose their hair before age 60 years.
Different patterns of baldness were also related to different grades of cancer, Charnita Zeigler-Johnson, Ph.D., and her colleagues reported in the March 26 online issue of Cancer, Epidemiology, Biomarkers, and Prevention (Canc. Ep. Biomark. Prev. 2013;22: 589-96).Those with frontal baldness were more than twice as likely to have high-grade and high-stage disease at diagnosis than were those with other hair loss patterns, wrote Dr. Zeigler-Johnson of the University of Pennsylvania, Philadelphia, and her coauthors.
The researchers’ case-control study comprised 318 black patients with prostate cancer and 219 black controls. The subjects were matched for age and other baseline characteristics. However, patients were significantly older than controls (60 vs. 57 years), and more likely to report a family history of prostate cancer (36% vs. 27%). Any form of baldness occurred in significantly more patients than in controls (20% vs.13%).
When the investigators conducted a multivariate analysis, they found a number of significant associations between the cancer and hair loss. Compared with those without hair loss, men with any form of baldness were 69% more likely to have prostate cancer. Frontal baldness was associated with more than a doubling in the risk of both high-stage and high-grade disease (odds ratio, 2.61 and 2.20, respectively).
Men with vertex balding who developed prostate cancer were significantly more likely to present with a low-grade tumor (OR, 1.45).
When the authors broke the groups down by age, they found no significant associations with disease severity among men older than 60 years. Instead, these risks were concentrated in men younger than 60 years. Among these, baldness increased the risk of high-stage cancer by more than three times (OR, 3.43) and more than doubled the risk high-grade disease (OR, 2.33). Frontal baldness was a particularly ominous risk factor for younger men, being associated with more than six times the risk of high-stage disease and more than four times the risk of high-grade disease (OR, 6.51 and 4.23, respectively).
There were also significant relationships observed between baldness and prostate specific antigen levels at diagnosis among younger men. Any baldness was associated with a tripling in the risk of a high PSA (10 ng/mL or more) at diagnosis. The association was stronger for men with frontal-only baldness (OR, 5.29).
The authors speculated that the elevated risks are related to genetically determined androgen metabolism. "There are differences in the prevalence of genotypes that metabolize testosterone and influence dihydrotestosterone (DHT) levels," they wrote. "High DHT levels have been associated with both early pattern baldness and prostate cancer processes, including increases in PSA levels."
In particular, they noted, four genes known to be associated with early-onset baldness are also involved in pathways of androgen metabolism, hair development, and age-related neurodegenerative disease.
"Given the high prevalence of prostate cancer in African Americans, early-onset baldness may be a particularly relevant indicator of risk that deserves attention in future studies as we seek to advance our knowledge about high-risk populations."
None of the authors had any financial disclosures. The work was funded by the Department of Defense and the Public Health Service.
FROM CANCER, EPIDEMIOLOGY, BIOMARKERS & PREVENTION
Major finding: Black men aged 60 years and younger who have frontal baldness had more than double in the risk of both high-stage and high-grade disease (OR 2.61 and 2.20, respectively).
Data source: The study included 318 cases and 219 controls.
Disclosures: None of the authors had any financial disclosures. The work was funded by the Department of Defense and the Public Health Service.
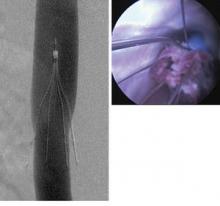
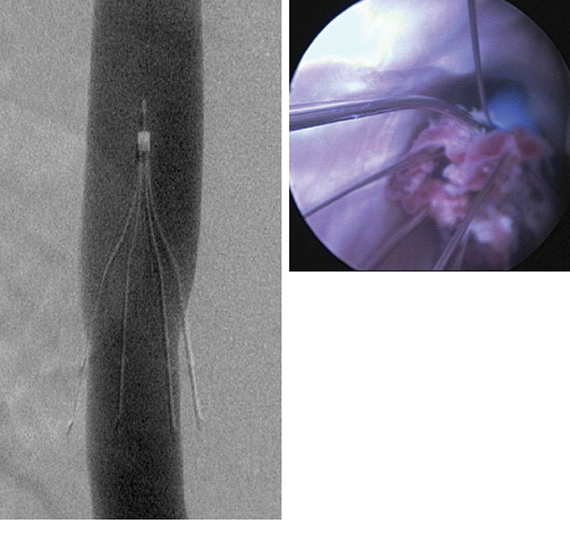
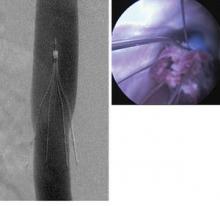
PRESERVE gets to the heart of IVC filters
The new PRESERVE study (Predicting the Safety and Effectiveness of Inferior Vena Cava Filters) seeks to examine the best use of IVC filters by focusing on safety endpoints. The 5-year, multispecialty, prospective study will enroll approximately 1,800 patients who have inferior vena cava (IVC) filters placed at 50 U.S. facilities. The goal of investigators is to keep in contact with patients every 6 months with phone calls, clinical exams, and repeat imaging. Anyone who has a filter removed will be followed for 3 months.
The primary safety endpoints will be freedom from perforation, migration, embolization, deep vein thrombosis (DVT), or fatal pulmon- ary embolism (PE). Secondary endpoints include procedural complications, insertion site, DVT, or any other major complication including death or PE. Investigators hope to begin PRESERVE this year.
"This study is going to bring the care of people with venous thromboembolic disease under a magnifying glass and actually figure out the safety and efficacy of these filters," said Dr. Matthew Johnson, one of the PRESERVE study directors.
After the Food and Drug Administration (FDA) issued its 2010 safety communication on retrievable IVC filters, the Society for Vascular Surgery (SVS) and the Society of Interventional Radiology (SIR) convened a task force to study the issue. SVS and SIR formed the IVC Filter Study Group Foundation, a collaborative body to oversee the study – underscoring this paradigm-shifting, collaborative undertaking.
Citing 921 device adverse events, FDA urged follow-up with patients who had the filters and for physicians to consider removing the devices based on the individual risk/benefit ratio. The problem, FDA said, was not inherent in the filters themselves, but more "related to a retrievable filter remaining in the body for long periods of time, beyond the time when the risk of pulmonary embolism has subsided."
When Dr. Lazar Greenfield invented the filter in the early 1970s, it was intended to be used in patients at risk of a pulmonary embolism, but who had contraindications to anticoagulation therapy. Since that time, however, off-label use has expanded and filters proliferated. Now there are more than a dozen on the market, all approved by the FDA’s 510(k) process."In 2010, about 210,000 of these filters were deployed worldwide, but 85% of those were done in the U.S.," said Dr. Johnson, a professor of radiology and surgery at the Indiana University School of Medicine, Indianapolis, and a member of SIR. "In the best interest of our patients, this study will provide a systematic, functional view, repre- sentative of most filters placed in the United States. It will assist with quality improvement in filter placement, management, and retrieval."
There are no concrete answers because very little research has been done on the topic. A 2010 Cochrane review found just two IVC trials comprising 529 patients. One of those examined the filters’ use in hip fracture patients, but had just a 1-month follow-up period.
The other study – which experts call the only substantial look at the issue – was the 1998 PREPIC, which randomized 400 patients with DVT or PE to filter or no filter, with a total of 8 years’ follow-up.
"No reduction in mortality was seen," the Cochrane report noted, "but this reflected an older study population; the majority of deaths were due to cancer or cardiovascular causes." The filter group had an increased, though not significantly so, incidence of DVTs, and the trial didn’t report filter-associated adverse events. Because all patients in both arms of the trial received anticoagulation therapy, it’s possible that the benefits of the filters were diminished.
"PREPIC did IVC filters a huge disservice, because it said that filters didn’t provide any benefit but were associated with a trend toward increased risk," of clotting. Nor did the trial include most of the filters in use today, Dr. Johnson added, furthering limiting its usefulness.
Because of the paucity of data, the Cochrane review could not make a recommendation on filter use. PRESERVE is important because it will not only provide long-term safety and efficacy data for a broad range of filters, but also paint a picture of how they are used, and how patients who have them are cared for.
One of FDA’s biggest safety concerns was that filters designed to be retrievable are, essentially, becoming permanent, Dr. David Gillespie said in an interview. Dr. Gillespie, an SVS member and PRESERVE study director, believes the devices are over-used in the U.S., where "practically every trauma patient" receives one early in the course of treatment, when the risk of developing a clot is highest.
Prophylactic use has caused an exponential growth in the way IVC filters are employed, said. Dr. Gillespie, a professor of surgery and chief of vascular surgery at the University of Rochester, N.Y. "Patients have them put in and then they aren’t being followed closely enough. The indication for use is and always will be for preventing PE death in patients who can’t have standard anticoagulation treatment, but this has been overextended to the point where people now think of them as a bona fide treatment for PE. We know that filters are good at preventing PE death, but they don’t seem to improve overall survival, and there is some physical cost to putting them in."
Even before its launch, the study may be changing practice, Dr. Johnson said. "Part of the change is acknowledgment of the problem. The discussion certainly is changing how we follow our patients."
Dr. Gillespie had received research money from BSN-Jobst, and served on advisory committees for Covidian and Angiodynmanics. Dr. Johnson is a paid consultant for Boston Scientific, Cook Medical, CeloNow BioSciences, and Nordion.
The new PRESERVE study (Predicting the Safety and Effectiveness of Inferior Vena Cava Filters) seeks to examine the best use of IVC filters by focusing on safety endpoints. The 5-year, multispecialty, prospective study will enroll approximately 1,800 patients who have inferior vena cava (IVC) filters placed at 50 U.S. facilities. The goal of investigators is to keep in contact with patients every 6 months with phone calls, clinical exams, and repeat imaging. Anyone who has a filter removed will be followed for 3 months.
The primary safety endpoints will be freedom from perforation, migration, embolization, deep vein thrombosis (DVT), or fatal pulmon- ary embolism (PE). Secondary endpoints include procedural complications, insertion site, DVT, or any other major complication including death or PE. Investigators hope to begin PRESERVE this year.
"This study is going to bring the care of people with venous thromboembolic disease under a magnifying glass and actually figure out the safety and efficacy of these filters," said Dr. Matthew Johnson, one of the PRESERVE study directors.
After the Food and Drug Administration (FDA) issued its 2010 safety communication on retrievable IVC filters, the Society for Vascular Surgery (SVS) and the Society of Interventional Radiology (SIR) convened a task force to study the issue. SVS and SIR formed the IVC Filter Study Group Foundation, a collaborative body to oversee the study – underscoring this paradigm-shifting, collaborative undertaking.
Citing 921 device adverse events, FDA urged follow-up with patients who had the filters and for physicians to consider removing the devices based on the individual risk/benefit ratio. The problem, FDA said, was not inherent in the filters themselves, but more "related to a retrievable filter remaining in the body for long periods of time, beyond the time when the risk of pulmonary embolism has subsided."
When Dr. Lazar Greenfield invented the filter in the early 1970s, it was intended to be used in patients at risk of a pulmonary embolism, but who had contraindications to anticoagulation therapy. Since that time, however, off-label use has expanded and filters proliferated. Now there are more than a dozen on the market, all approved by the FDA’s 510(k) process."In 2010, about 210,000 of these filters were deployed worldwide, but 85% of those were done in the U.S.," said Dr. Johnson, a professor of radiology and surgery at the Indiana University School of Medicine, Indianapolis, and a member of SIR. "In the best interest of our patients, this study will provide a systematic, functional view, repre- sentative of most filters placed in the United States. It will assist with quality improvement in filter placement, management, and retrieval."
There are no concrete answers because very little research has been done on the topic. A 2010 Cochrane review found just two IVC trials comprising 529 patients. One of those examined the filters’ use in hip fracture patients, but had just a 1-month follow-up period.
The other study – which experts call the only substantial look at the issue – was the 1998 PREPIC, which randomized 400 patients with DVT or PE to filter or no filter, with a total of 8 years’ follow-up.
"No reduction in mortality was seen," the Cochrane report noted, "but this reflected an older study population; the majority of deaths were due to cancer or cardiovascular causes." The filter group had an increased, though not significantly so, incidence of DVTs, and the trial didn’t report filter-associated adverse events. Because all patients in both arms of the trial received anticoagulation therapy, it’s possible that the benefits of the filters were diminished.
"PREPIC did IVC filters a huge disservice, because it said that filters didn’t provide any benefit but were associated with a trend toward increased risk," of clotting. Nor did the trial include most of the filters in use today, Dr. Johnson added, furthering limiting its usefulness.
Because of the paucity of data, the Cochrane review could not make a recommendation on filter use. PRESERVE is important because it will not only provide long-term safety and efficacy data for a broad range of filters, but also paint a picture of how they are used, and how patients who have them are cared for.
One of FDA’s biggest safety concerns was that filters designed to be retrievable are, essentially, becoming permanent, Dr. David Gillespie said in an interview. Dr. Gillespie, an SVS member and PRESERVE study director, believes the devices are over-used in the U.S., where "practically every trauma patient" receives one early in the course of treatment, when the risk of developing a clot is highest.
Prophylactic use has caused an exponential growth in the way IVC filters are employed, said. Dr. Gillespie, a professor of surgery and chief of vascular surgery at the University of Rochester, N.Y. "Patients have them put in and then they aren’t being followed closely enough. The indication for use is and always will be for preventing PE death in patients who can’t have standard anticoagulation treatment, but this has been overextended to the point where people now think of them as a bona fide treatment for PE. We know that filters are good at preventing PE death, but they don’t seem to improve overall survival, and there is some physical cost to putting them in."
Even before its launch, the study may be changing practice, Dr. Johnson said. "Part of the change is acknowledgment of the problem. The discussion certainly is changing how we follow our patients."
Dr. Gillespie had received research money from BSN-Jobst, and served on advisory committees for Covidian and Angiodynmanics. Dr. Johnson is a paid consultant for Boston Scientific, Cook Medical, CeloNow BioSciences, and Nordion.
The new PRESERVE study (Predicting the Safety and Effectiveness of Inferior Vena Cava Filters) seeks to examine the best use of IVC filters by focusing on safety endpoints. The 5-year, multispecialty, prospective study will enroll approximately 1,800 patients who have inferior vena cava (IVC) filters placed at 50 U.S. facilities. The goal of investigators is to keep in contact with patients every 6 months with phone calls, clinical exams, and repeat imaging. Anyone who has a filter removed will be followed for 3 months.
The primary safety endpoints will be freedom from perforation, migration, embolization, deep vein thrombosis (DVT), or fatal pulmon- ary embolism (PE). Secondary endpoints include procedural complications, insertion site, DVT, or any other major complication including death or PE. Investigators hope to begin PRESERVE this year.
"This study is going to bring the care of people with venous thromboembolic disease under a magnifying glass and actually figure out the safety and efficacy of these filters," said Dr. Matthew Johnson, one of the PRESERVE study directors.
After the Food and Drug Administration (FDA) issued its 2010 safety communication on retrievable IVC filters, the Society for Vascular Surgery (SVS) and the Society of Interventional Radiology (SIR) convened a task force to study the issue. SVS and SIR formed the IVC Filter Study Group Foundation, a collaborative body to oversee the study – underscoring this paradigm-shifting, collaborative undertaking.
Citing 921 device adverse events, FDA urged follow-up with patients who had the filters and for physicians to consider removing the devices based on the individual risk/benefit ratio. The problem, FDA said, was not inherent in the filters themselves, but more "related to a retrievable filter remaining in the body for long periods of time, beyond the time when the risk of pulmonary embolism has subsided."
When Dr. Lazar Greenfield invented the filter in the early 1970s, it was intended to be used in patients at risk of a pulmonary embolism, but who had contraindications to anticoagulation therapy. Since that time, however, off-label use has expanded and filters proliferated. Now there are more than a dozen on the market, all approved by the FDA’s 510(k) process."In 2010, about 210,000 of these filters were deployed worldwide, but 85% of those were done in the U.S.," said Dr. Johnson, a professor of radiology and surgery at the Indiana University School of Medicine, Indianapolis, and a member of SIR. "In the best interest of our patients, this study will provide a systematic, functional view, repre- sentative of most filters placed in the United States. It will assist with quality improvement in filter placement, management, and retrieval."
There are no concrete answers because very little research has been done on the topic. A 2010 Cochrane review found just two IVC trials comprising 529 patients. One of those examined the filters’ use in hip fracture patients, but had just a 1-month follow-up period.
The other study – which experts call the only substantial look at the issue – was the 1998 PREPIC, which randomized 400 patients with DVT or PE to filter or no filter, with a total of 8 years’ follow-up.
"No reduction in mortality was seen," the Cochrane report noted, "but this reflected an older study population; the majority of deaths were due to cancer or cardiovascular causes." The filter group had an increased, though not significantly so, incidence of DVTs, and the trial didn’t report filter-associated adverse events. Because all patients in both arms of the trial received anticoagulation therapy, it’s possible that the benefits of the filters were diminished.
"PREPIC did IVC filters a huge disservice, because it said that filters didn’t provide any benefit but were associated with a trend toward increased risk," of clotting. Nor did the trial include most of the filters in use today, Dr. Johnson added, furthering limiting its usefulness.
Because of the paucity of data, the Cochrane review could not make a recommendation on filter use. PRESERVE is important because it will not only provide long-term safety and efficacy data for a broad range of filters, but also paint a picture of how they are used, and how patients who have them are cared for.
One of FDA’s biggest safety concerns was that filters designed to be retrievable are, essentially, becoming permanent, Dr. David Gillespie said in an interview. Dr. Gillespie, an SVS member and PRESERVE study director, believes the devices are over-used in the U.S., where "practically every trauma patient" receives one early in the course of treatment, when the risk of developing a clot is highest.
Prophylactic use has caused an exponential growth in the way IVC filters are employed, said. Dr. Gillespie, a professor of surgery and chief of vascular surgery at the University of Rochester, N.Y. "Patients have them put in and then they aren’t being followed closely enough. The indication for use is and always will be for preventing PE death in patients who can’t have standard anticoagulation treatment, but this has been overextended to the point where people now think of them as a bona fide treatment for PE. We know that filters are good at preventing PE death, but they don’t seem to improve overall survival, and there is some physical cost to putting them in."
Even before its launch, the study may be changing practice, Dr. Johnson said. "Part of the change is acknowledgment of the problem. The discussion certainly is changing how we follow our patients."
Dr. Gillespie had received research money from BSN-Jobst, and served on advisory committees for Covidian and Angiodynmanics. Dr. Johnson is a paid consultant for Boston Scientific, Cook Medical, CeloNow BioSciences, and Nordion.
PRESERVE gets to the heart of IVC filters
The new PRESERVE study (Predicting the Safety and Effectiveness of Inferior Vena Cava Filters) seeks to examine the best use of IVC filters by focusing on safety endpoints. The 5-year, multispecialty, prospective study will enroll approximately 1,800 patients who have inferior vena cava (IVC) filters placed at 50 U.S. facilities. The goal of investigators is to keep in contact with patients every 6 months with phone calls, clinical exams, and repeat imaging. Anyone who has a filter removed will be followed for 3 months.
The primary safety endpoints will be freedom from perforation, migration, embolization, deep vein thrombosis (DVT), or fatal pulmon- ary embolism (PE). Secondary endpoints include procedural complications, insertion site, DVT, or any other major complication including death or PE. Investigators hope to begin PRESERVE this year.
"This study is going to bring the care of people with venous thromboembolic disease under a magnifying glass and actually figure out the safety and efficacy of these filters," said Dr. Matthew Johnson, one of the PRESERVE study directors.
After the Food and Drug Administration (FDA) issued its 2010 safety communication on retrievable IVC filters, the Society for Vascular Surgery (SVS) and the Society of Interventional Radiology (SIR) convened a task force to study the issue. SVS and SIR formed the IVC Filter Study Group Foundation, a collaborative body to oversee the study – underscoring this paradigm-shifting, collaborative undertaking.
Citing 921 device adverse events, FDA urged follow-up with patients who had the filters and for physicians to consider removing the devices based on the individual risk/benefit ratio. The problem, FDA said, was not inherent in the filters themselves, but more "related to a retrievable filter remaining in the body for long periods of time, beyond the time when the risk of pulmonary embolism has subsided."
When Dr. Lazar Greenfield invented the filter in the early 1970s, it was intended to be used in patients at risk of a pulmonary embolism, but who had contraindications to anticoagulation therapy. Since that time, however, off-label use has expanded and filters proliferated. Now there are more than a dozen on the market, all approved by the FDA’s 510(k) process."In 2010, about 210,000 of these filters were deployed worldwide, but 85% of those were done in the U.S.," said Dr. Johnson, a professor of radiology and surgery at the Indiana University School of Medicine, Indianapolis, and a member of SIR. "In the best interest of our patients, this study will provide a systematic, functional view, repre- sentative of most filters placed in the United States. It will assist with quality improvement in filter placement, management, and retrieval."
There are no concrete answers because very little research has been done on the topic. A 2010 Cochrane review found just two IVC trials comprising 529 patients. One of those examined the filters’ use in hip fracture patients, but had just a 1-month follow-up period.
The other study – which experts call the only substantial look at the issue – was the 1998 PREPIC, which randomized 400 patients with DVT or PE to filter or no filter, with a total of 8 years’ follow-up.
"No reduction in mortality was seen," the Cochrane report noted, "but this reflected an older study population; the majority of deaths were due to cancer or cardiovascular causes." The filter group had an increased, though not significantly so, incidence of DVTs, and the trial didn’t report filter-associated adverse events. Because all patients in both arms of the trial received anticoagulation therapy, it’s possible that the benefits of the filters were diminished.
"PREPIC did IVC filters a huge disservice, because it said that filters didn’t provide any benefit but were associated with a trend toward increased risk," of clotting. Nor did the trial include most of the filters in use today, Dr. Johnson added, furthering limiting its usefulness.
Because of the paucity of data, the Cochrane review could not make a recommendation on filter use. PRESERVE is important because it will not only provide long-term safety and efficacy data for a broad range of filters, but also paint a picture of how they are used, and how patients who have them are cared for.
One of FDA’s biggest safety concerns was that filters designed to be retrievable are, essentially, becoming permanent, Dr. David Gillespie said in an interview. Dr. Gillespie, an SVS member and PRESERVE study director, believes the devices are over-used in the U.S., where "practically every trauma patient" receives one early in the course of treatment, when the risk of developing a clot is highest.
Prophylactic use has caused an exponential growth in the way IVC filters are employed, said. Dr. Gillespie, a professor of surgery and chief of vascular surgery at the University of Rochester, N.Y. "Patients have them put in and then they aren’t being followed closely enough. The indication for use is and always will be for preventing PE death in patients who can’t have standard anticoagulation treatment, but this has been overextended to the point where people now think of them as a bona fide treatment for PE. We know that filters are good at preventing PE death, but they don’t seem to improve overall survival, and there is some physical cost to putting them in."
Even before its launch, the study may be changing practice, Dr. Johnson said. "Part of the change is acknowledgment of the problem. The discussion certainly is changing how we follow our patients."
Dr. Gillespie had received research money from BSN-Jobst, and served on advisory committees for Covidian and Angiodynmanics. Dr. Johnson is a paid consultant for Boston Scientific, Cook Medical, CeloNow BioSciences, and Nordion.
The new PRESERVE study (Predicting the Safety and Effectiveness of Inferior Vena Cava Filters) seeks to examine the best use of IVC filters by focusing on safety endpoints. The 5-year, multispecialty, prospective study will enroll approximately 1,800 patients who have inferior vena cava (IVC) filters placed at 50 U.S. facilities. The goal of investigators is to keep in contact with patients every 6 months with phone calls, clinical exams, and repeat imaging. Anyone who has a filter removed will be followed for 3 months.
The primary safety endpoints will be freedom from perforation, migration, embolization, deep vein thrombosis (DVT), or fatal pulmon- ary embolism (PE). Secondary endpoints include procedural complications, insertion site, DVT, or any other major complication including death or PE. Investigators hope to begin PRESERVE this year.
"This study is going to bring the care of people with venous thromboembolic disease under a magnifying glass and actually figure out the safety and efficacy of these filters," said Dr. Matthew Johnson, one of the PRESERVE study directors.
After the Food and Drug Administration (FDA) issued its 2010 safety communication on retrievable IVC filters, the Society for Vascular Surgery (SVS) and the Society of Interventional Radiology (SIR) convened a task force to study the issue. SVS and SIR formed the IVC Filter Study Group Foundation, a collaborative body to oversee the study – underscoring this paradigm-shifting, collaborative undertaking.
Citing 921 device adverse events, FDA urged follow-up with patients who had the filters and for physicians to consider removing the devices based on the individual risk/benefit ratio. The problem, FDA said, was not inherent in the filters themselves, but more "related to a retrievable filter remaining in the body for long periods of time, beyond the time when the risk of pulmonary embolism has subsided."
When Dr. Lazar Greenfield invented the filter in the early 1970s, it was intended to be used in patients at risk of a pulmonary embolism, but who had contraindications to anticoagulation therapy. Since that time, however, off-label use has expanded and filters proliferated. Now there are more than a dozen on the market, all approved by the FDA’s 510(k) process."In 2010, about 210,000 of these filters were deployed worldwide, but 85% of those were done in the U.S.," said Dr. Johnson, a professor of radiology and surgery at the Indiana University School of Medicine, Indianapolis, and a member of SIR. "In the best interest of our patients, this study will provide a systematic, functional view, repre- sentative of most filters placed in the United States. It will assist with quality improvement in filter placement, management, and retrieval."
There are no concrete answers because very little research has been done on the topic. A 2010 Cochrane review found just two IVC trials comprising 529 patients. One of those examined the filters’ use in hip fracture patients, but had just a 1-month follow-up period.
The other study – which experts call the only substantial look at the issue – was the 1998 PREPIC, which randomized 400 patients with DVT or PE to filter or no filter, with a total of 8 years’ follow-up.
"No reduction in mortality was seen," the Cochrane report noted, "but this reflected an older study population; the majority of deaths were due to cancer or cardiovascular causes." The filter group had an increased, though not significantly so, incidence of DVTs, and the trial didn’t report filter-associated adverse events. Because all patients in both arms of the trial received anticoagulation therapy, it’s possible that the benefits of the filters were diminished.
"PREPIC did IVC filters a huge disservice, because it said that filters didn’t provide any benefit but were associated with a trend toward increased risk," of clotting. Nor did the trial include most of the filters in use today, Dr. Johnson added, furthering limiting its usefulness.
Because of the paucity of data, the Cochrane review could not make a recommendation on filter use. PRESERVE is important because it will not only provide long-term safety and efficacy data for a broad range of filters, but also paint a picture of how they are used, and how patients who have them are cared for.
One of FDA’s biggest safety concerns was that filters designed to be retrievable are, essentially, becoming permanent, Dr. David Gillespie said in an interview. Dr. Gillespie, an SVS member and PRESERVE study director, believes the devices are over-used in the U.S., where "practically every trauma patient" receives one early in the course of treatment, when the risk of developing a clot is highest.
Prophylactic use has caused an exponential growth in the way IVC filters are employed, said. Dr. Gillespie, a professor of surgery and chief of vascular surgery at the University of Rochester, N.Y. "Patients have them put in and then they aren’t being followed closely enough. The indication for use is and always will be for preventing PE death in patients who can’t have standard anticoagulation treatment, but this has been overextended to the point where people now think of them as a bona fide treatment for PE. We know that filters are good at preventing PE death, but they don’t seem to improve overall survival, and there is some physical cost to putting them in."
Even before its launch, the study may be changing practice, Dr. Johnson said. "Part of the change is acknowledgment of the problem. The discussion certainly is changing how we follow our patients."
Dr. Gillespie had received research money from BSN-Jobst, and served on advisory committees for Covidian and Angiodynmanics. Dr. Johnson is a paid consultant for Boston Scientific, Cook Medical, CeloNow BioSciences, and Nordion.
The new PRESERVE study (Predicting the Safety and Effectiveness of Inferior Vena Cava Filters) seeks to examine the best use of IVC filters by focusing on safety endpoints. The 5-year, multispecialty, prospective study will enroll approximately 1,800 patients who have inferior vena cava (IVC) filters placed at 50 U.S. facilities. The goal of investigators is to keep in contact with patients every 6 months with phone calls, clinical exams, and repeat imaging. Anyone who has a filter removed will be followed for 3 months.
The primary safety endpoints will be freedom from perforation, migration, embolization, deep vein thrombosis (DVT), or fatal pulmon- ary embolism (PE). Secondary endpoints include procedural complications, insertion site, DVT, or any other major complication including death or PE. Investigators hope to begin PRESERVE this year.
"This study is going to bring the care of people with venous thromboembolic disease under a magnifying glass and actually figure out the safety and efficacy of these filters," said Dr. Matthew Johnson, one of the PRESERVE study directors.
After the Food and Drug Administration (FDA) issued its 2010 safety communication on retrievable IVC filters, the Society for Vascular Surgery (SVS) and the Society of Interventional Radiology (SIR) convened a task force to study the issue. SVS and SIR formed the IVC Filter Study Group Foundation, a collaborative body to oversee the study – underscoring this paradigm-shifting, collaborative undertaking.
Citing 921 device adverse events, FDA urged follow-up with patients who had the filters and for physicians to consider removing the devices based on the individual risk/benefit ratio. The problem, FDA said, was not inherent in the filters themselves, but more "related to a retrievable filter remaining in the body for long periods of time, beyond the time when the risk of pulmonary embolism has subsided."
When Dr. Lazar Greenfield invented the filter in the early 1970s, it was intended to be used in patients at risk of a pulmonary embolism, but who had contraindications to anticoagulation therapy. Since that time, however, off-label use has expanded and filters proliferated. Now there are more than a dozen on the market, all approved by the FDA’s 510(k) process."In 2010, about 210,000 of these filters were deployed worldwide, but 85% of those were done in the U.S.," said Dr. Johnson, a professor of radiology and surgery at the Indiana University School of Medicine, Indianapolis, and a member of SIR. "In the best interest of our patients, this study will provide a systematic, functional view, repre- sentative of most filters placed in the United States. It will assist with quality improvement in filter placement, management, and retrieval."
There are no concrete answers because very little research has been done on the topic. A 2010 Cochrane review found just two IVC trials comprising 529 patients. One of those examined the filters’ use in hip fracture patients, but had just a 1-month follow-up period.
The other study – which experts call the only substantial look at the issue – was the 1998 PREPIC, which randomized 400 patients with DVT or PE to filter or no filter, with a total of 8 years’ follow-up.
"No reduction in mortality was seen," the Cochrane report noted, "but this reflected an older study population; the majority of deaths were due to cancer or cardiovascular causes." The filter group had an increased, though not significantly so, incidence of DVTs, and the trial didn’t report filter-associated adverse events. Because all patients in both arms of the trial received anticoagulation therapy, it’s possible that the benefits of the filters were diminished.
"PREPIC did IVC filters a huge disservice, because it said that filters didn’t provide any benefit but were associated with a trend toward increased risk," of clotting. Nor did the trial include most of the filters in use today, Dr. Johnson added, furthering limiting its usefulness.
Because of the paucity of data, the Cochrane review could not make a recommendation on filter use. PRESERVE is important because it will not only provide long-term safety and efficacy data for a broad range of filters, but also paint a picture of how they are used, and how patients who have them are cared for.
One of FDA’s biggest safety concerns was that filters designed to be retrievable are, essentially, becoming permanent, Dr. David Gillespie said in an interview. Dr. Gillespie, an SVS member and PRESERVE study director, believes the devices are over-used in the U.S., where "practically every trauma patient" receives one early in the course of treatment, when the risk of developing a clot is highest.
Prophylactic use has caused an exponential growth in the way IVC filters are employed, said. Dr. Gillespie, a professor of surgery and chief of vascular surgery at the University of Rochester, N.Y. "Patients have them put in and then they aren’t being followed closely enough. The indication for use is and always will be for preventing PE death in patients who can’t have standard anticoagulation treatment, but this has been overextended to the point where people now think of them as a bona fide treatment for PE. We know that filters are good at preventing PE death, but they don’t seem to improve overall survival, and there is some physical cost to putting them in."
Even before its launch, the study may be changing practice, Dr. Johnson said. "Part of the change is acknowledgment of the problem. The discussion certainly is changing how we follow our patients."
Dr. Gillespie had received research money from BSN-Jobst, and served on advisory committees for Covidian and Angiodynmanics. Dr. Johnson is a paid consultant for Boston Scientific, Cook Medical, CeloNow BioSciences, and Nordion.
Steroids may decrease length of hospital stay for pneumonia
Steroid treatment may not improve mortality in community-acquired pneumonia, but it was associated with significantly shorter hospital stays and an increase in the chance of a clean chest radiograph after treatment.
A meta-analysis of eight studies on the topic showed that steroid treatment reduced the overall length of stay by a little over 1 day. There also was an 87% reduction in the risk of an abnormal chest x-ray at 1 week and an 88% reduction in the risk of delayed shock. These last findings, however, were based on just a few of the analysis’ studies, which were considered only of moderate quality, Dr. Majid Shafiq and colleagues wrote in the March issue of the Journal of Hospital Medicine.
"The data are not strong enough to recommend routine use of steroids among all adults hospitalized with" community-acquired pneumonia (CAP), wrote Dr. Shafiq and his coauthors at the Mayo Clinic in Rochester, Minn. "However, considering that there was no increase in mortality or hospital length of stay with steroid use, it is reasonable to continue steroids if warranted for treatment of underlying comorbid conditions," they noted (J. Hosp. Med. 2013;8:68-75).
The analysis included a total of 1,119 patients, and there were four randomized controlled trials among the studies. In seven studies, the mean patient age ranged from 60 to 80 years. In one study, patients in the experimental arm were a mean of 32 years and those in the control arm were a mean of 41 years. Only one study used a chest x-ray score.
The mean length of stay in the intensive care unit was 13 days for patients taking steroids and 12 for the control patients. The mean hospital length of stay was 10 days for those taking steroids and 14 days for those who did not, said Dr. Shafiq, who is now with Johns Hopkins University, Baltimore, and his associates.
Steroid use did not significantly impact the length of ICU stay or mortality. Four studies showed significantly lower clinical cure rates and a higher number of late failures in patients taking steroids. Two showed no between-group differences in the occurrence of superinfections. Three studies reported that the drugs did not affect gylcemic levels, while four found more frequent hyperglycemia in the steroid group.
"Our study is the first to demonstrate decreased length of hospital stay in this patient population," the investigators wrote. "Importantly, each of the five studies that reported this outcome (including three relatively recent randomized controlled trials) showed the same trend. However, it is not inconceivable that steroid use led to a quicker decline in cytokine levels resulting in an earlier resolution of fever and hence earlier discharge without a faster cure per se."
Dr. Shafiq and his associates reported no financial conflicts.
Steroid treatment may not improve mortality in community-acquired pneumonia, but it was associated with significantly shorter hospital stays and an increase in the chance of a clean chest radiograph after treatment.
A meta-analysis of eight studies on the topic showed that steroid treatment reduced the overall length of stay by a little over 1 day. There also was an 87% reduction in the risk of an abnormal chest x-ray at 1 week and an 88% reduction in the risk of delayed shock. These last findings, however, were based on just a few of the analysis’ studies, which were considered only of moderate quality, Dr. Majid Shafiq and colleagues wrote in the March issue of the Journal of Hospital Medicine.
"The data are not strong enough to recommend routine use of steroids among all adults hospitalized with" community-acquired pneumonia (CAP), wrote Dr. Shafiq and his coauthors at the Mayo Clinic in Rochester, Minn. "However, considering that there was no increase in mortality or hospital length of stay with steroid use, it is reasonable to continue steroids if warranted for treatment of underlying comorbid conditions," they noted (J. Hosp. Med. 2013;8:68-75).
The analysis included a total of 1,119 patients, and there were four randomized controlled trials among the studies. In seven studies, the mean patient age ranged from 60 to 80 years. In one study, patients in the experimental arm were a mean of 32 years and those in the control arm were a mean of 41 years. Only one study used a chest x-ray score.
The mean length of stay in the intensive care unit was 13 days for patients taking steroids and 12 for the control patients. The mean hospital length of stay was 10 days for those taking steroids and 14 days for those who did not, said Dr. Shafiq, who is now with Johns Hopkins University, Baltimore, and his associates.
Steroid use did not significantly impact the length of ICU stay or mortality. Four studies showed significantly lower clinical cure rates and a higher number of late failures in patients taking steroids. Two showed no between-group differences in the occurrence of superinfections. Three studies reported that the drugs did not affect gylcemic levels, while four found more frequent hyperglycemia in the steroid group.
"Our study is the first to demonstrate decreased length of hospital stay in this patient population," the investigators wrote. "Importantly, each of the five studies that reported this outcome (including three relatively recent randomized controlled trials) showed the same trend. However, it is not inconceivable that steroid use led to a quicker decline in cytokine levels resulting in an earlier resolution of fever and hence earlier discharge without a faster cure per se."
Dr. Shafiq and his associates reported no financial conflicts.
Steroid treatment may not improve mortality in community-acquired pneumonia, but it was associated with significantly shorter hospital stays and an increase in the chance of a clean chest radiograph after treatment.
A meta-analysis of eight studies on the topic showed that steroid treatment reduced the overall length of stay by a little over 1 day. There also was an 87% reduction in the risk of an abnormal chest x-ray at 1 week and an 88% reduction in the risk of delayed shock. These last findings, however, were based on just a few of the analysis’ studies, which were considered only of moderate quality, Dr. Majid Shafiq and colleagues wrote in the March issue of the Journal of Hospital Medicine.
"The data are not strong enough to recommend routine use of steroids among all adults hospitalized with" community-acquired pneumonia (CAP), wrote Dr. Shafiq and his coauthors at the Mayo Clinic in Rochester, Minn. "However, considering that there was no increase in mortality or hospital length of stay with steroid use, it is reasonable to continue steroids if warranted for treatment of underlying comorbid conditions," they noted (J. Hosp. Med. 2013;8:68-75).
The analysis included a total of 1,119 patients, and there were four randomized controlled trials among the studies. In seven studies, the mean patient age ranged from 60 to 80 years. In one study, patients in the experimental arm were a mean of 32 years and those in the control arm were a mean of 41 years. Only one study used a chest x-ray score.
The mean length of stay in the intensive care unit was 13 days for patients taking steroids and 12 for the control patients. The mean hospital length of stay was 10 days for those taking steroids and 14 days for those who did not, said Dr. Shafiq, who is now with Johns Hopkins University, Baltimore, and his associates.
Steroid use did not significantly impact the length of ICU stay or mortality. Four studies showed significantly lower clinical cure rates and a higher number of late failures in patients taking steroids. Two showed no between-group differences in the occurrence of superinfections. Three studies reported that the drugs did not affect gylcemic levels, while four found more frequent hyperglycemia in the steroid group.
"Our study is the first to demonstrate decreased length of hospital stay in this patient population," the investigators wrote. "Importantly, each of the five studies that reported this outcome (including three relatively recent randomized controlled trials) showed the same trend. However, it is not inconceivable that steroid use led to a quicker decline in cytokine levels resulting in an earlier resolution of fever and hence earlier discharge without a faster cure per se."
Dr. Shafiq and his associates reported no financial conflicts.
FROM THE JOURNAL OF HOSPITAL MEDICINE
Major Finding: Steroid treatment reduced the overall length of stay by a little more than 1 day in patients with community-acquire pneumonia. The study also showed an 87% reduction in the risk of an abnormal chest x-ray at 1 week and an 88% reduction in the risk of delayed shock, compared with patients who did not take the drugs.
Data Source: Data were drawn from a meta-analysis of eight studies comprising 1,119 patients.
Disclosures: Neither Dr. Shafiq nor his coauthors reported any financial conflicts.
Aliskiren doesn't help heart failure patients, may harm diabetics
For patients hospitalized with heart failure, aliskiren added no significant benefit to standard therapy in preventing cardiovascular death or heart failure readmission.
The drug did, however, have some problematic effects on patients with diabetes, including significant increases in the risk of cardiovascular death and heart failure readmission at 6 months and all-cause death at 1 year.
The negative interaction between diabetes and aliskiren – an inhibitor of the angiotensin-renin-aldosterone system (RAAS) – is unclear, according to Dr. Mihai Gheorghiade and colleagues.
"[The issue] may be related to the increased risk of hypotension, hyperkalemia, and worsening renal function with the study drug," wrote Dr. Gheorghiade of Northwestern University, St. Louis, Mo., and his colleagues. The conclusions were published in the March 10 issue of JAMA and simultaneously presented at the annual meeting of the American College of Cardiology in San Francisco (JAMA 2013;309:doi10.1001/jama.2013.1954).
The randomized, placebo-controlled ASTRONAUT trial compared 6-and 12-month outcomes of aliskiren plus standard therapy to standard therapy alone in patients hospitalized with heart failure.
The study randomized 1,639 patients; 1,615 were included in the final efficacy analysis. Patients in the investigational arm received 150 mg aliskiren daily in addition to standard treatment. Standard treatment was at the treating physician’s discretion, and could include, among others, diuretics, digoxin, ACE inhibitors, angiotensin receptor blockers, and beta-blockers. The study enrolled patients from 2009-2011; the median follow-up was 11 months
The patients were a mean of 65 years old. Their mean left ventricular ejection fraction was 28%, and mean estimated glomerular filtration rate was 67 mL/min/1.73 m3. Nearly half (41%) had diabetes.
At admission, the mean N-terminal pro–brain natriuretic peptide (NT-proBNP) level was 4,239 pg/mL. Other drugs taken at baseline included diuretics (96%), beta-blockers (82%), ACE inhibitors or ARBs (84%), and mineralocorticoid antagonists (57%).
The primary efficacy endpoint was a combination of cardiovascular death or heart failure readmission within 6 months. This occurred in 201 (25%) of the aliskiren group and 214 (26%) of the placebo group – a nonsignificant difference. Nor were there significant differences when CV death and readmission were examined separately.
The main secondary endpoint was the composite of cardiovascular death or heart failure readmission at 12 months; again, there was no significant between-group difference (35% vs. 37%), although the events were numerically less in the aliskiren group than the control group (283 vs. 301). Parsing down the number of first cardiovascular events after treatment, the investigators did find that myocardial infarction was significantly less common in the aliskiren group (2% vs. 5%; HR 0.93; P = 0.009).
A subgroup analysis found that the drug was especially problematic for patients with diabetes. Compared to patients without the disorder who had standard treatment, diabetic patients who took aliskiren were 16% more likely to experience cardiovascular death or heart failure readmission by 6 months, and 64% more likely to experience all-cause death by 12 months.
During the treatment period, death occurred in 24% of patients with diabetes who took aliskiren, compared with 17% who took placebo – also a significant difference.
"In contrast, the rates of death among patients without a history [of diabetes] were 15% and 20% in the aliskiren and placebo groups, respectively," the authors wrote.
A number of adverse events were significantly higher in the aliskiren group, including hyperkalemia (14% vs. 13%) and severe hyperkalemia (8% vs. 5%). Hyperkalemia-related events occurred in 21% of the aliskiren group and 17% of the control group. Hypotension was also more common in the aliskiren group (17% vs. 13%). Decreased eGFR was significantly more common among those taking the study drug (11% vs. 9%), as were events potentially related to renal dysfunction (17% vs. 12%).
"The results of the ASTRONAUT study do not support the routine administration of aliskiren in addition to standard therapy, to patients hospitalized for worsening chronic heart failure," the investigators concluded, adding that the subgroup analysis was "consistent with previous reports of poor outcomes with the use of aliskiren in patients with diabetes already taking RAAS inhibitors."
Further studies will be necessary to evaluate renin inhibiting drugs in patients with diabetes, they added.
Dr. Gheorghiade reported financial relationships with 32 pharmaceutical companies, including Novartis Pharma AG, the study sponsor.
For patients hospitalized with heart failure, aliskiren added no significant benefit to standard therapy in preventing cardiovascular death or heart failure readmission.
The drug did, however, have some problematic effects on patients with diabetes, including significant increases in the risk of cardiovascular death and heart failure readmission at 6 months and all-cause death at 1 year.
The negative interaction between diabetes and aliskiren – an inhibitor of the angiotensin-renin-aldosterone system (RAAS) – is unclear, according to Dr. Mihai Gheorghiade and colleagues.
"[The issue] may be related to the increased risk of hypotension, hyperkalemia, and worsening renal function with the study drug," wrote Dr. Gheorghiade of Northwestern University, St. Louis, Mo., and his colleagues. The conclusions were published in the March 10 issue of JAMA and simultaneously presented at the annual meeting of the American College of Cardiology in San Francisco (JAMA 2013;309:doi10.1001/jama.2013.1954).
The randomized, placebo-controlled ASTRONAUT trial compared 6-and 12-month outcomes of aliskiren plus standard therapy to standard therapy alone in patients hospitalized with heart failure.
The study randomized 1,639 patients; 1,615 were included in the final efficacy analysis. Patients in the investigational arm received 150 mg aliskiren daily in addition to standard treatment. Standard treatment was at the treating physician’s discretion, and could include, among others, diuretics, digoxin, ACE inhibitors, angiotensin receptor blockers, and beta-blockers. The study enrolled patients from 2009-2011; the median follow-up was 11 months
The patients were a mean of 65 years old. Their mean left ventricular ejection fraction was 28%, and mean estimated glomerular filtration rate was 67 mL/min/1.73 m3. Nearly half (41%) had diabetes.
At admission, the mean N-terminal pro–brain natriuretic peptide (NT-proBNP) level was 4,239 pg/mL. Other drugs taken at baseline included diuretics (96%), beta-blockers (82%), ACE inhibitors or ARBs (84%), and mineralocorticoid antagonists (57%).
The primary efficacy endpoint was a combination of cardiovascular death or heart failure readmission within 6 months. This occurred in 201 (25%) of the aliskiren group and 214 (26%) of the placebo group – a nonsignificant difference. Nor were there significant differences when CV death and readmission were examined separately.
The main secondary endpoint was the composite of cardiovascular death or heart failure readmission at 12 months; again, there was no significant between-group difference (35% vs. 37%), although the events were numerically less in the aliskiren group than the control group (283 vs. 301). Parsing down the number of first cardiovascular events after treatment, the investigators did find that myocardial infarction was significantly less common in the aliskiren group (2% vs. 5%; HR 0.93; P = 0.009).
A subgroup analysis found that the drug was especially problematic for patients with diabetes. Compared to patients without the disorder who had standard treatment, diabetic patients who took aliskiren were 16% more likely to experience cardiovascular death or heart failure readmission by 6 months, and 64% more likely to experience all-cause death by 12 months.
During the treatment period, death occurred in 24% of patients with diabetes who took aliskiren, compared with 17% who took placebo – also a significant difference.
"In contrast, the rates of death among patients without a history [of diabetes] were 15% and 20% in the aliskiren and placebo groups, respectively," the authors wrote.
A number of adverse events were significantly higher in the aliskiren group, including hyperkalemia (14% vs. 13%) and severe hyperkalemia (8% vs. 5%). Hyperkalemia-related events occurred in 21% of the aliskiren group and 17% of the control group. Hypotension was also more common in the aliskiren group (17% vs. 13%). Decreased eGFR was significantly more common among those taking the study drug (11% vs. 9%), as were events potentially related to renal dysfunction (17% vs. 12%).
"The results of the ASTRONAUT study do not support the routine administration of aliskiren in addition to standard therapy, to patients hospitalized for worsening chronic heart failure," the investigators concluded, adding that the subgroup analysis was "consistent with previous reports of poor outcomes with the use of aliskiren in patients with diabetes already taking RAAS inhibitors."
Further studies will be necessary to evaluate renin inhibiting drugs in patients with diabetes, they added.
Dr. Gheorghiade reported financial relationships with 32 pharmaceutical companies, including Novartis Pharma AG, the study sponsor.
For patients hospitalized with heart failure, aliskiren added no significant benefit to standard therapy in preventing cardiovascular death or heart failure readmission.
The drug did, however, have some problematic effects on patients with diabetes, including significant increases in the risk of cardiovascular death and heart failure readmission at 6 months and all-cause death at 1 year.
The negative interaction between diabetes and aliskiren – an inhibitor of the angiotensin-renin-aldosterone system (RAAS) – is unclear, according to Dr. Mihai Gheorghiade and colleagues.
"[The issue] may be related to the increased risk of hypotension, hyperkalemia, and worsening renal function with the study drug," wrote Dr. Gheorghiade of Northwestern University, St. Louis, Mo., and his colleagues. The conclusions were published in the March 10 issue of JAMA and simultaneously presented at the annual meeting of the American College of Cardiology in San Francisco (JAMA 2013;309:doi10.1001/jama.2013.1954).
The randomized, placebo-controlled ASTRONAUT trial compared 6-and 12-month outcomes of aliskiren plus standard therapy to standard therapy alone in patients hospitalized with heart failure.
The study randomized 1,639 patients; 1,615 were included in the final efficacy analysis. Patients in the investigational arm received 150 mg aliskiren daily in addition to standard treatment. Standard treatment was at the treating physician’s discretion, and could include, among others, diuretics, digoxin, ACE inhibitors, angiotensin receptor blockers, and beta-blockers. The study enrolled patients from 2009-2011; the median follow-up was 11 months
The patients were a mean of 65 years old. Their mean left ventricular ejection fraction was 28%, and mean estimated glomerular filtration rate was 67 mL/min/1.73 m3. Nearly half (41%) had diabetes.
At admission, the mean N-terminal pro–brain natriuretic peptide (NT-proBNP) level was 4,239 pg/mL. Other drugs taken at baseline included diuretics (96%), beta-blockers (82%), ACE inhibitors or ARBs (84%), and mineralocorticoid antagonists (57%).
The primary efficacy endpoint was a combination of cardiovascular death or heart failure readmission within 6 months. This occurred in 201 (25%) of the aliskiren group and 214 (26%) of the placebo group – a nonsignificant difference. Nor were there significant differences when CV death and readmission were examined separately.
The main secondary endpoint was the composite of cardiovascular death or heart failure readmission at 12 months; again, there was no significant between-group difference (35% vs. 37%), although the events were numerically less in the aliskiren group than the control group (283 vs. 301). Parsing down the number of first cardiovascular events after treatment, the investigators did find that myocardial infarction was significantly less common in the aliskiren group (2% vs. 5%; HR 0.93; P = 0.009).
A subgroup analysis found that the drug was especially problematic for patients with diabetes. Compared to patients without the disorder who had standard treatment, diabetic patients who took aliskiren were 16% more likely to experience cardiovascular death or heart failure readmission by 6 months, and 64% more likely to experience all-cause death by 12 months.
During the treatment period, death occurred in 24% of patients with diabetes who took aliskiren, compared with 17% who took placebo – also a significant difference.
"In contrast, the rates of death among patients without a history [of diabetes] were 15% and 20% in the aliskiren and placebo groups, respectively," the authors wrote.
A number of adverse events were significantly higher in the aliskiren group, including hyperkalemia (14% vs. 13%) and severe hyperkalemia (8% vs. 5%). Hyperkalemia-related events occurred in 21% of the aliskiren group and 17% of the control group. Hypotension was also more common in the aliskiren group (17% vs. 13%). Decreased eGFR was significantly more common among those taking the study drug (11% vs. 9%), as were events potentially related to renal dysfunction (17% vs. 12%).
"The results of the ASTRONAUT study do not support the routine administration of aliskiren in addition to standard therapy, to patients hospitalized for worsening chronic heart failure," the investigators concluded, adding that the subgroup analysis was "consistent with previous reports of poor outcomes with the use of aliskiren in patients with diabetes already taking RAAS inhibitors."
Further studies will be necessary to evaluate renin inhibiting drugs in patients with diabetes, they added.
Dr. Gheorghiade reported financial relationships with 32 pharmaceutical companies, including Novartis Pharma AG, the study sponsor.
Major Finding: Aliskiren
added no significant benefit for patients hospitalized with heart failure, and,
in fact, was associated with increased mortality among patients with diabetes,
compared with standard therapy.
Data Source: ASTRONAUT, a randomized, placebo-controlled trial comprising 1,639 hospitalized heart failure patients.
Disclosures: Dr. Gheorghiade
reported financial relationships with 32 pharmaceutical companies, including
Novartis Pharma AG, the study sponsor.