User login
Two doses of HPV vaccine may be as effective as three
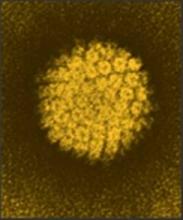
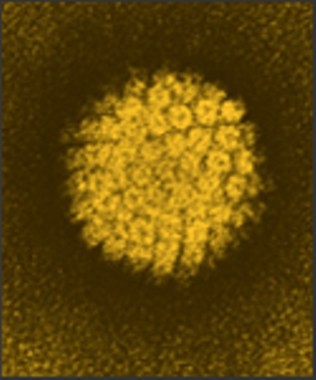
Two doses of the human papillomavirus vaccine may be as effective as three in young adolescent girls, a postmarketing study suggests.
The immunogenic response of girls who got the two vaccines 6 months apart was just as good – if not better – than it was in girls and young women who had the traditional three-dose schedule, reported Dr. Simon R.M. Dobson and colleagues. The study was published in the May 1 issue of JAMA.
Longer and larger studies will be necessary before considering any change to the recommended regime, but an effective two-dose schedule could impart an enormous benefit in both developed and underdeveloped countries, Dr. Dobson said during a press briefing.
In Canada, about 40% of girls are not fully vaccinated; in the United States, that number reaches 80%, said Dr. Dobson of the University of British Columbia, Vancouver. Cost is probably part of the issue. A two-dose schedule could increase compliance considerably, he said.
In less-developed countries – including those without routine cervical cancer screening, where the need for protection is arguably greater – the HPV vaccine is less accessible. "The three-dose schedule might simply be out of range, but a two-dose schedule might make it accessible to many more women," he said.
This trial involved 830 subjects: 520 girls aged 9-13 years, who were randomized to a two- or three-dose schedule of the HPV quadrivalent vaccine, and 310 women aged 16-26 years, all of whom were vaccinated according to the three-dose schedule. The main endpoint was immunogenicity at 7 and up to 36 months after the last vaccine dose (JAMA 2013;309:1793-1802).
The mean titer antibody levels in girls who got the two-dose schedule were noninferior to those of the young women who had three doses: 7,344 mMU/mL for HPV-16 and 1,169 mMU/mL for HPV-18 vs. 3,545 mMU/mL for HPV-16 and 664 mMU/mL for HPV-18.
The investigators determined that the geometric mean titer ratios also were noninferior in the younger group compared with the older group: 2.07 for HPV-16 and 1.76 for HPV-18.
The results were similar for HPV-6 and HPV-11, Dr. Dobson and the coauthors reported. The younger girls who received doses had mean titer levels of 2,117 mMU/mL for HPV-6 and 2,339 for HPV-11, compared with 943 mMU/mL and 1,268 mMU/mL, respectively, for the women. The titer ratios also were noninferior.
The girls who received two doses also had noninferior GMT titers and ratios compared with girls who received three doses for all four vaccine genotypes. Girls receiving three doses had GMT levels of 7,736 for HPV-16, 1,730 for HPV-18, 1,876 for HPV-6, and 2,117 for HPV-11. Likewise, girls who received two doses vs. three doses had a noninferior antibody response for all four vaccine genotypes, with GMT ratios of 0.95 for HPV-16; 0.68 for HPV-18; 1.13 for HPV-6; and 1.10 for HPV-11.
Nearly all of the subjects (about 99%) remained seropositive for HPV-16, -6 and -11 until the end of the 36-month follow-up period.
At 24 months, HPV-18 seropositivity was seen in 89% of the girls who got two doses, 94% of the girls who got three doses, and 83% of the women who got three doses. This remained nearly unchanged for all three groups at 36 months.
Between 7 and 18 months, antibody levels for all four HPV genotypes declined and plateaued through the end of follow-up. "Both girls’ groups continued to maintain higher plateau levels of antibody at 36 months than women," Dr. Dobson said. However, there was some evidence that the two-dose group lost noninferiority for HPV-18 by 24 months, and HPV-6 by 36 months.
This finding does bring into question the durability of immune response in a two-dose vaccine schedule, Dr. Dobson said Antibody levels peaked at 7 months, then began to wane and plateaued at about 18 months, staying level until the end of the study. It’s still unclear how that waning level might impact protection; the same pattern is seen in young women who have gone through the entire three dose schedule.
"These young women have been followed for up to 10 years now, and the vaccine has remained 100% protective. There’s no magic antibody level below which we know protection fails. This vaccine is so good that there haven’t been enough vaccine failures to determine this," he said.
There’s already good evidence that societal protection is as important as individual protection, Dr. Dobson added at the press conference. "Studies in Australia [which has the largest vaccinated population] are showing protection from genital warts not only in women who are immunized, but in unimmunized men and unimmunized women as well. There’s a herd effect occurring – but we don’t know if two doses will have the same effect here as three doses do."
Ministries of Health in British Columbia, Nova Scotia, and Quebec funded the study. Merck Laboratories conducted the antibody assays at no cost. Dr. Dobson and coauthors disclosed ties with various pharmaceutical companies; some had ties to Merck.
This study is the first to show preliminary evidence that a two-dose HPV vaccination regimen may provide protection as effective as that of the three-dose regimen. The findings are encouraging, but very preliminary, Dr. Jennifer Kahn and Dr. David L. Bernstein wrote in an editorial.
An effective two-dose regimen is a goal worthy of pursuing because many barriers prevent the fullest utilization of the HPV vaccine, wrote Dr. Kahn and Dr. Bernstein.
"These barriers include the high cost of vaccination and the many challenges associated with administration of a three-dose vaccine, especially given the lack of infrastructure for adolescent vaccination programs in many countries ... A two-dose series could lead to a substantial increase in the number of girls completing the vaccine series for the same cost, ensuring that greater numbers are protected."
The short-term equivalent immunogenicity demonstrated in the study indicates the potential for developing such a regimen and thus, reaching many more girls in their early adolescence – the time when they mount the most robust immune response. "However, and most important, antibody responses for HPV-18 at month 24 and HPV-6 at month 36 were significantly higher for girls who received three doses. This comparison of girls 9-13 years of age receiving two vs. three doses is important clinically, because girls in this age group are targeted for vaccination."
However, an amended regimen should also be tested in girls aged 13 and older, because, although younger girls are the primary vaccination target, many older girls are actually vaccinated.
"Depending on the results of these studies, clinical efficacy studies should be conducted to ensure that two doses of the ... vaccine are sufficient to prevent incident and persistent HPV infection, precancers (not only cervical but also anal, vaginal, and vulvar precancers), and anogenital warts, because the immunologic response that correlates with protection against HPV is unknown."
Males represent a big question mark in this entire equation, the authors noted.
"Studies of immunologic responses in men will also be important, because there is now a recommendation for men to receive the quadrivalent vaccine in the United States. If a two-dose series were immunogenic in men, cost-effectiveness estimates could become more favorable with respect to male vaccination, which in turn could alter vaccine recommendations for men. Also, if a two-dose schedule is recommended, consideration must be given to the possibility that a month 0 and 6 schedule may delay protection compared to a month 0, 1 to 2, and 6 schedule."
If these future studies do confirm a durable immune response from a two-dose schedule, the global impact could be significant, Dr. Kahn and Dr. Bernstein concluded.
"The potential to further reduce morbidity and mortality due to HPV-related cancers would be especially significant in less developed regions of the world, where the cost of vaccination and implementation of adolescent vaccination programs present significant barriers, but where primary prevention strategies are most urgently needed."
Dr. Kahn is an adolescent medicine expert in the department of pediatrics at Cincinnati Children’s Hospital Medical Center, and Dr. Bernstein is an infectious diseases expert in the department of pediatrics at the medical center. Dr. Kahn is on a grant review committee administered by the Society for Adolescent Health and Medicine (funded by Merck), which is funding projects designed to improve adolescent vaccination rates, and is cochair on clinical trials of the HPV vaccine in HIV-infected women and men funded by the National Institutes of Health, with Merck providing the vaccines. Dr. Bernstein receives speaking fees and royalties from GlaxoSmithKline for sales of a rotavirus vaccine.
This study is the first to show preliminary evidence that a two-dose HPV vaccination regimen may provide protection as effective as that of the three-dose regimen. The findings are encouraging, but very preliminary, Dr. Jennifer Kahn and Dr. David L. Bernstein wrote in an editorial.
An effective two-dose regimen is a goal worthy of pursuing because many barriers prevent the fullest utilization of the HPV vaccine, wrote Dr. Kahn and Dr. Bernstein.
"These barriers include the high cost of vaccination and the many challenges associated with administration of a three-dose vaccine, especially given the lack of infrastructure for adolescent vaccination programs in many countries ... A two-dose series could lead to a substantial increase in the number of girls completing the vaccine series for the same cost, ensuring that greater numbers are protected."
The short-term equivalent immunogenicity demonstrated in the study indicates the potential for developing such a regimen and thus, reaching many more girls in their early adolescence – the time when they mount the most robust immune response. "However, and most important, antibody responses for HPV-18 at month 24 and HPV-6 at month 36 were significantly higher for girls who received three doses. This comparison of girls 9-13 years of age receiving two vs. three doses is important clinically, because girls in this age group are targeted for vaccination."
However, an amended regimen should also be tested in girls aged 13 and older, because, although younger girls are the primary vaccination target, many older girls are actually vaccinated.
"Depending on the results of these studies, clinical efficacy studies should be conducted to ensure that two doses of the ... vaccine are sufficient to prevent incident and persistent HPV infection, precancers (not only cervical but also anal, vaginal, and vulvar precancers), and anogenital warts, because the immunologic response that correlates with protection against HPV is unknown."
Males represent a big question mark in this entire equation, the authors noted.
"Studies of immunologic responses in men will also be important, because there is now a recommendation for men to receive the quadrivalent vaccine in the United States. If a two-dose series were immunogenic in men, cost-effectiveness estimates could become more favorable with respect to male vaccination, which in turn could alter vaccine recommendations for men. Also, if a two-dose schedule is recommended, consideration must be given to the possibility that a month 0 and 6 schedule may delay protection compared to a month 0, 1 to 2, and 6 schedule."
If these future studies do confirm a durable immune response from a two-dose schedule, the global impact could be significant, Dr. Kahn and Dr. Bernstein concluded.
"The potential to further reduce morbidity and mortality due to HPV-related cancers would be especially significant in less developed regions of the world, where the cost of vaccination and implementation of adolescent vaccination programs present significant barriers, but where primary prevention strategies are most urgently needed."
Dr. Kahn is an adolescent medicine expert in the department of pediatrics at Cincinnati Children’s Hospital Medical Center, and Dr. Bernstein is an infectious diseases expert in the department of pediatrics at the medical center. Dr. Kahn is on a grant review committee administered by the Society for Adolescent Health and Medicine (funded by Merck), which is funding projects designed to improve adolescent vaccination rates, and is cochair on clinical trials of the HPV vaccine in HIV-infected women and men funded by the National Institutes of Health, with Merck providing the vaccines. Dr. Bernstein receives speaking fees and royalties from GlaxoSmithKline for sales of a rotavirus vaccine.
This study is the first to show preliminary evidence that a two-dose HPV vaccination regimen may provide protection as effective as that of the three-dose regimen. The findings are encouraging, but very preliminary, Dr. Jennifer Kahn and Dr. David L. Bernstein wrote in an editorial.
An effective two-dose regimen is a goal worthy of pursuing because many barriers prevent the fullest utilization of the HPV vaccine, wrote Dr. Kahn and Dr. Bernstein.
"These barriers include the high cost of vaccination and the many challenges associated with administration of a three-dose vaccine, especially given the lack of infrastructure for adolescent vaccination programs in many countries ... A two-dose series could lead to a substantial increase in the number of girls completing the vaccine series for the same cost, ensuring that greater numbers are protected."
The short-term equivalent immunogenicity demonstrated in the study indicates the potential for developing such a regimen and thus, reaching many more girls in their early adolescence – the time when they mount the most robust immune response. "However, and most important, antibody responses for HPV-18 at month 24 and HPV-6 at month 36 were significantly higher for girls who received three doses. This comparison of girls 9-13 years of age receiving two vs. three doses is important clinically, because girls in this age group are targeted for vaccination."
However, an amended regimen should also be tested in girls aged 13 and older, because, although younger girls are the primary vaccination target, many older girls are actually vaccinated.
"Depending on the results of these studies, clinical efficacy studies should be conducted to ensure that two doses of the ... vaccine are sufficient to prevent incident and persistent HPV infection, precancers (not only cervical but also anal, vaginal, and vulvar precancers), and anogenital warts, because the immunologic response that correlates with protection against HPV is unknown."
Males represent a big question mark in this entire equation, the authors noted.
"Studies of immunologic responses in men will also be important, because there is now a recommendation for men to receive the quadrivalent vaccine in the United States. If a two-dose series were immunogenic in men, cost-effectiveness estimates could become more favorable with respect to male vaccination, which in turn could alter vaccine recommendations for men. Also, if a two-dose schedule is recommended, consideration must be given to the possibility that a month 0 and 6 schedule may delay protection compared to a month 0, 1 to 2, and 6 schedule."
If these future studies do confirm a durable immune response from a two-dose schedule, the global impact could be significant, Dr. Kahn and Dr. Bernstein concluded.
"The potential to further reduce morbidity and mortality due to HPV-related cancers would be especially significant in less developed regions of the world, where the cost of vaccination and implementation of adolescent vaccination programs present significant barriers, but where primary prevention strategies are most urgently needed."
Dr. Kahn is an adolescent medicine expert in the department of pediatrics at Cincinnati Children’s Hospital Medical Center, and Dr. Bernstein is an infectious diseases expert in the department of pediatrics at the medical center. Dr. Kahn is on a grant review committee administered by the Society for Adolescent Health and Medicine (funded by Merck), which is funding projects designed to improve adolescent vaccination rates, and is cochair on clinical trials of the HPV vaccine in HIV-infected women and men funded by the National Institutes of Health, with Merck providing the vaccines. Dr. Bernstein receives speaking fees and royalties from GlaxoSmithKline for sales of a rotavirus vaccine.
Two doses of the human papillomavirus vaccine may be as effective as three in young adolescent girls, a postmarketing study suggests.
The immunogenic response of girls who got the two vaccines 6 months apart was just as good – if not better – than it was in girls and young women who had the traditional three-dose schedule, reported Dr. Simon R.M. Dobson and colleagues. The study was published in the May 1 issue of JAMA.
Longer and larger studies will be necessary before considering any change to the recommended regime, but an effective two-dose schedule could impart an enormous benefit in both developed and underdeveloped countries, Dr. Dobson said during a press briefing.
In Canada, about 40% of girls are not fully vaccinated; in the United States, that number reaches 80%, said Dr. Dobson of the University of British Columbia, Vancouver. Cost is probably part of the issue. A two-dose schedule could increase compliance considerably, he said.
In less-developed countries – including those without routine cervical cancer screening, where the need for protection is arguably greater – the HPV vaccine is less accessible. "The three-dose schedule might simply be out of range, but a two-dose schedule might make it accessible to many more women," he said.
This trial involved 830 subjects: 520 girls aged 9-13 years, who were randomized to a two- or three-dose schedule of the HPV quadrivalent vaccine, and 310 women aged 16-26 years, all of whom were vaccinated according to the three-dose schedule. The main endpoint was immunogenicity at 7 and up to 36 months after the last vaccine dose (JAMA 2013;309:1793-1802).
The mean titer antibody levels in girls who got the two-dose schedule were noninferior to those of the young women who had three doses: 7,344 mMU/mL for HPV-16 and 1,169 mMU/mL for HPV-18 vs. 3,545 mMU/mL for HPV-16 and 664 mMU/mL for HPV-18.
The investigators determined that the geometric mean titer ratios also were noninferior in the younger group compared with the older group: 2.07 for HPV-16 and 1.76 for HPV-18.
The results were similar for HPV-6 and HPV-11, Dr. Dobson and the coauthors reported. The younger girls who received doses had mean titer levels of 2,117 mMU/mL for HPV-6 and 2,339 for HPV-11, compared with 943 mMU/mL and 1,268 mMU/mL, respectively, for the women. The titer ratios also were noninferior.
The girls who received two doses also had noninferior GMT titers and ratios compared with girls who received three doses for all four vaccine genotypes. Girls receiving three doses had GMT levels of 7,736 for HPV-16, 1,730 for HPV-18, 1,876 for HPV-6, and 2,117 for HPV-11. Likewise, girls who received two doses vs. three doses had a noninferior antibody response for all four vaccine genotypes, with GMT ratios of 0.95 for HPV-16; 0.68 for HPV-18; 1.13 for HPV-6; and 1.10 for HPV-11.
Nearly all of the subjects (about 99%) remained seropositive for HPV-16, -6 and -11 until the end of the 36-month follow-up period.
At 24 months, HPV-18 seropositivity was seen in 89% of the girls who got two doses, 94% of the girls who got three doses, and 83% of the women who got three doses. This remained nearly unchanged for all three groups at 36 months.
Between 7 and 18 months, antibody levels for all four HPV genotypes declined and plateaued through the end of follow-up. "Both girls’ groups continued to maintain higher plateau levels of antibody at 36 months than women," Dr. Dobson said. However, there was some evidence that the two-dose group lost noninferiority for HPV-18 by 24 months, and HPV-6 by 36 months.
This finding does bring into question the durability of immune response in a two-dose vaccine schedule, Dr. Dobson said Antibody levels peaked at 7 months, then began to wane and plateaued at about 18 months, staying level until the end of the study. It’s still unclear how that waning level might impact protection; the same pattern is seen in young women who have gone through the entire three dose schedule.
"These young women have been followed for up to 10 years now, and the vaccine has remained 100% protective. There’s no magic antibody level below which we know protection fails. This vaccine is so good that there haven’t been enough vaccine failures to determine this," he said.
There’s already good evidence that societal protection is as important as individual protection, Dr. Dobson added at the press conference. "Studies in Australia [which has the largest vaccinated population] are showing protection from genital warts not only in women who are immunized, but in unimmunized men and unimmunized women as well. There’s a herd effect occurring – but we don’t know if two doses will have the same effect here as three doses do."
Ministries of Health in British Columbia, Nova Scotia, and Quebec funded the study. Merck Laboratories conducted the antibody assays at no cost. Dr. Dobson and coauthors disclosed ties with various pharmaceutical companies; some had ties to Merck.
Two doses of the human papillomavirus vaccine may be as effective as three in young adolescent girls, a postmarketing study suggests.
The immunogenic response of girls who got the two vaccines 6 months apart was just as good – if not better – than it was in girls and young women who had the traditional three-dose schedule, reported Dr. Simon R.M. Dobson and colleagues. The study was published in the May 1 issue of JAMA.
Longer and larger studies will be necessary before considering any change to the recommended regime, but an effective two-dose schedule could impart an enormous benefit in both developed and underdeveloped countries, Dr. Dobson said during a press briefing.
In Canada, about 40% of girls are not fully vaccinated; in the United States, that number reaches 80%, said Dr. Dobson of the University of British Columbia, Vancouver. Cost is probably part of the issue. A two-dose schedule could increase compliance considerably, he said.
In less-developed countries – including those without routine cervical cancer screening, where the need for protection is arguably greater – the HPV vaccine is less accessible. "The three-dose schedule might simply be out of range, but a two-dose schedule might make it accessible to many more women," he said.
This trial involved 830 subjects: 520 girls aged 9-13 years, who were randomized to a two- or three-dose schedule of the HPV quadrivalent vaccine, and 310 women aged 16-26 years, all of whom were vaccinated according to the three-dose schedule. The main endpoint was immunogenicity at 7 and up to 36 months after the last vaccine dose (JAMA 2013;309:1793-1802).
The mean titer antibody levels in girls who got the two-dose schedule were noninferior to those of the young women who had three doses: 7,344 mMU/mL for HPV-16 and 1,169 mMU/mL for HPV-18 vs. 3,545 mMU/mL for HPV-16 and 664 mMU/mL for HPV-18.
The investigators determined that the geometric mean titer ratios also were noninferior in the younger group compared with the older group: 2.07 for HPV-16 and 1.76 for HPV-18.
The results were similar for HPV-6 and HPV-11, Dr. Dobson and the coauthors reported. The younger girls who received doses had mean titer levels of 2,117 mMU/mL for HPV-6 and 2,339 for HPV-11, compared with 943 mMU/mL and 1,268 mMU/mL, respectively, for the women. The titer ratios also were noninferior.
The girls who received two doses also had noninferior GMT titers and ratios compared with girls who received three doses for all four vaccine genotypes. Girls receiving three doses had GMT levels of 7,736 for HPV-16, 1,730 for HPV-18, 1,876 for HPV-6, and 2,117 for HPV-11. Likewise, girls who received two doses vs. three doses had a noninferior antibody response for all four vaccine genotypes, with GMT ratios of 0.95 for HPV-16; 0.68 for HPV-18; 1.13 for HPV-6; and 1.10 for HPV-11.
Nearly all of the subjects (about 99%) remained seropositive for HPV-16, -6 and -11 until the end of the 36-month follow-up period.
At 24 months, HPV-18 seropositivity was seen in 89% of the girls who got two doses, 94% of the girls who got three doses, and 83% of the women who got three doses. This remained nearly unchanged for all three groups at 36 months.
Between 7 and 18 months, antibody levels for all four HPV genotypes declined and plateaued through the end of follow-up. "Both girls’ groups continued to maintain higher plateau levels of antibody at 36 months than women," Dr. Dobson said. However, there was some evidence that the two-dose group lost noninferiority for HPV-18 by 24 months, and HPV-6 by 36 months.
This finding does bring into question the durability of immune response in a two-dose vaccine schedule, Dr. Dobson said Antibody levels peaked at 7 months, then began to wane and plateaued at about 18 months, staying level until the end of the study. It’s still unclear how that waning level might impact protection; the same pattern is seen in young women who have gone through the entire three dose schedule.
"These young women have been followed for up to 10 years now, and the vaccine has remained 100% protective. There’s no magic antibody level below which we know protection fails. This vaccine is so good that there haven’t been enough vaccine failures to determine this," he said.
There’s already good evidence that societal protection is as important as individual protection, Dr. Dobson added at the press conference. "Studies in Australia [which has the largest vaccinated population] are showing protection from genital warts not only in women who are immunized, but in unimmunized men and unimmunized women as well. There’s a herd effect occurring – but we don’t know if two doses will have the same effect here as three doses do."
Ministries of Health in British Columbia, Nova Scotia, and Quebec funded the study. Merck Laboratories conducted the antibody assays at no cost. Dr. Dobson and coauthors disclosed ties with various pharmaceutical companies; some had ties to Merck.
FROM JAMA
Major finding: The mean titer antibody levels in girls who got the two-dose schedule were noninferior to those of the young women who had three doses: 7,344 mMU/mL for HPV-16 and 1,169 mMU/mL for HPV-18 vs. 3,545 mMU/mL for HPV-16 and 664 mMU/mL for HPV-18.
Data source: A study of 520 girls aged 9-13 years randomized to a two- or three-dose vaccine schedule, and 310 women aged 16-26 years, all of whom were vaccinated according to the three-dose schedule.
Disclosures: Ministries of health in British Columbia, Nova Scotia, and Quebec funded the study. Merck Laboratories conducted the antibody assays at no cost. Dr. Dobson and coauthors disclosed ties with various pharmaceutical companies; some had ties to Merck.
Diabetes treatment algorithm sets goals, time limits
A new evidence-based diabetes management algorithm looks at the disease from a holistic view, tailoring a treatment plan to fit each patient’s comorbid risks and treatment goals.
The algorithm, a project of the American Association of Clinical Endocrinologists, highlights obesity as a crucial factor in both diabetes prevention and treatment. Weight management should be an integral part of an overall plan designed to reduce morbidity, mortality, and disability in the majority of patients with type 2 diabetes who are obese, according to a document published in the March/April issue of Endocrine Practice (2012;19:327-36).
"This is something that’s never been done before," said Dr. George Grunberger, a member of the task force that formulated the document and founder of the Grunberger Diabetes Institute, Bloomfield Hills, Mich. "It’s a totally different concept that looks at all aspects of diabetes. We now know that treating diabetes doesn’t just mean treating blood sugar. It means looking at nutrition, obesity, and cardiovascular risk factors as a whole."
The algorithm still uses 6.5% or lower as the cutoff point for hemoglobin A1c in patients who are otherwise healthy. But that goal can be individualized at higher numbers for those with concurrent illness and who are at risk for hypoglycemia.
Another new facet of the document is its prioritization of drug choices, Dr. Grunberger said in an interview. "What we did that is different, and maybe controversial, is to indicate to clinicians and patients some kind of order of preference," in drug therapy. "One problem with the previous algorithms has been that all the drugs are listed, but there’s no advice on which one to pick when. Good pharmacotherapy isn’t just an accident. There is an order of preference."
Organized in a colorful graphic, the algorithm indicates drug preference in the familiar stepwise manner, beginning with the lowest-risk drugs – metformin and acarbose – for prediabetes, moving on to combinations and drugs with a higher side effect profile as needed, until the patient reaches a predetermined blood sugar goal.
The algorithm is time bound as well, Dr. Grunberger noted. "Clinical inertia is a big problem in diabetes treatment. We tend to get to one treatment step and then not move, so that years later, a patient is still taking the same drug and not at goal. These steps have specific time limits of 2 or 3 months. If by that time you haven’t made progress, then it’s on to the next step."
The basic glycemic control algorithm, for example, begins with lifestyle modifications for everyone. The initial medication choices are stratified by HbA1c at entry – less than or greater than 7.5%, and greater than 9.0%. Each treatment path starts with drugs usually deemed safest. The algorithm recommends specific drug combinations as treatment intensifies. Each pathway has a 3-month window to explore the regimen’s effect. If that time expires without reaching the HbA1c goal, then the algorithm moves the patient to the next treatment level.
Unsuccessful triple therapy, or entry with an HbA1c of more than 9%, calls for basal insulin. Again, the algorithm specifies the order of treatment based on glucose levels. For a level of less than 8%, it calls for a total daily insulin dosage of 0.1-0.2 U/kg. For a blood sugar above 8%, the dosage is 0.2-0.3 U/kg. Both dosages should be titrated every 2-3 days to reach the glycemic goal. If the goal isn’t met, the algorithm moves on to prandial insulin at a total daily dosage of 0.3-0.5 U/kg.
Obesity management is a foundation treatment for all patients who are overweight. It starts with lifestyle modification, then moves through pharmacologic treatment and finally into surgical options.
The algorithm also tackles cardiovascular risk reduction, including lipid and blood pressure management, Dr. Grunberger said.
"An obese patient with dyslipidemia, high blood pressure, and diabetes is a ticking time bomb. All of these things need to be considered and treated together."
Dr. Grunberger is on the speakers board and scientific advisory panels for several pharmaceutical companies that manufacture diabetes treatment medications. He has also received research grants from a number of these companies.
A new evidence-based diabetes management algorithm looks at the disease from a holistic view, tailoring a treatment plan to fit each patient’s comorbid risks and treatment goals.
The algorithm, a project of the American Association of Clinical Endocrinologists, highlights obesity as a crucial factor in both diabetes prevention and treatment. Weight management should be an integral part of an overall plan designed to reduce morbidity, mortality, and disability in the majority of patients with type 2 diabetes who are obese, according to a document published in the March/April issue of Endocrine Practice (2012;19:327-36).
"This is something that’s never been done before," said Dr. George Grunberger, a member of the task force that formulated the document and founder of the Grunberger Diabetes Institute, Bloomfield Hills, Mich. "It’s a totally different concept that looks at all aspects of diabetes. We now know that treating diabetes doesn’t just mean treating blood sugar. It means looking at nutrition, obesity, and cardiovascular risk factors as a whole."
The algorithm still uses 6.5% or lower as the cutoff point for hemoglobin A1c in patients who are otherwise healthy. But that goal can be individualized at higher numbers for those with concurrent illness and who are at risk for hypoglycemia.
Another new facet of the document is its prioritization of drug choices, Dr. Grunberger said in an interview. "What we did that is different, and maybe controversial, is to indicate to clinicians and patients some kind of order of preference," in drug therapy. "One problem with the previous algorithms has been that all the drugs are listed, but there’s no advice on which one to pick when. Good pharmacotherapy isn’t just an accident. There is an order of preference."
Organized in a colorful graphic, the algorithm indicates drug preference in the familiar stepwise manner, beginning with the lowest-risk drugs – metformin and acarbose – for prediabetes, moving on to combinations and drugs with a higher side effect profile as needed, until the patient reaches a predetermined blood sugar goal.
The algorithm is time bound as well, Dr. Grunberger noted. "Clinical inertia is a big problem in diabetes treatment. We tend to get to one treatment step and then not move, so that years later, a patient is still taking the same drug and not at goal. These steps have specific time limits of 2 or 3 months. If by that time you haven’t made progress, then it’s on to the next step."
The basic glycemic control algorithm, for example, begins with lifestyle modifications for everyone. The initial medication choices are stratified by HbA1c at entry – less than or greater than 7.5%, and greater than 9.0%. Each treatment path starts with drugs usually deemed safest. The algorithm recommends specific drug combinations as treatment intensifies. Each pathway has a 3-month window to explore the regimen’s effect. If that time expires without reaching the HbA1c goal, then the algorithm moves the patient to the next treatment level.
Unsuccessful triple therapy, or entry with an HbA1c of more than 9%, calls for basal insulin. Again, the algorithm specifies the order of treatment based on glucose levels. For a level of less than 8%, it calls for a total daily insulin dosage of 0.1-0.2 U/kg. For a blood sugar above 8%, the dosage is 0.2-0.3 U/kg. Both dosages should be titrated every 2-3 days to reach the glycemic goal. If the goal isn’t met, the algorithm moves on to prandial insulin at a total daily dosage of 0.3-0.5 U/kg.
Obesity management is a foundation treatment for all patients who are overweight. It starts with lifestyle modification, then moves through pharmacologic treatment and finally into surgical options.
The algorithm also tackles cardiovascular risk reduction, including lipid and blood pressure management, Dr. Grunberger said.
"An obese patient with dyslipidemia, high blood pressure, and diabetes is a ticking time bomb. All of these things need to be considered and treated together."
Dr. Grunberger is on the speakers board and scientific advisory panels for several pharmaceutical companies that manufacture diabetes treatment medications. He has also received research grants from a number of these companies.
A new evidence-based diabetes management algorithm looks at the disease from a holistic view, tailoring a treatment plan to fit each patient’s comorbid risks and treatment goals.
The algorithm, a project of the American Association of Clinical Endocrinologists, highlights obesity as a crucial factor in both diabetes prevention and treatment. Weight management should be an integral part of an overall plan designed to reduce morbidity, mortality, and disability in the majority of patients with type 2 diabetes who are obese, according to a document published in the March/April issue of Endocrine Practice (2012;19:327-36).
"This is something that’s never been done before," said Dr. George Grunberger, a member of the task force that formulated the document and founder of the Grunberger Diabetes Institute, Bloomfield Hills, Mich. "It’s a totally different concept that looks at all aspects of diabetes. We now know that treating diabetes doesn’t just mean treating blood sugar. It means looking at nutrition, obesity, and cardiovascular risk factors as a whole."
The algorithm still uses 6.5% or lower as the cutoff point for hemoglobin A1c in patients who are otherwise healthy. But that goal can be individualized at higher numbers for those with concurrent illness and who are at risk for hypoglycemia.
Another new facet of the document is its prioritization of drug choices, Dr. Grunberger said in an interview. "What we did that is different, and maybe controversial, is to indicate to clinicians and patients some kind of order of preference," in drug therapy. "One problem with the previous algorithms has been that all the drugs are listed, but there’s no advice on which one to pick when. Good pharmacotherapy isn’t just an accident. There is an order of preference."
Organized in a colorful graphic, the algorithm indicates drug preference in the familiar stepwise manner, beginning with the lowest-risk drugs – metformin and acarbose – for prediabetes, moving on to combinations and drugs with a higher side effect profile as needed, until the patient reaches a predetermined blood sugar goal.
The algorithm is time bound as well, Dr. Grunberger noted. "Clinical inertia is a big problem in diabetes treatment. We tend to get to one treatment step and then not move, so that years later, a patient is still taking the same drug and not at goal. These steps have specific time limits of 2 or 3 months. If by that time you haven’t made progress, then it’s on to the next step."
The basic glycemic control algorithm, for example, begins with lifestyle modifications for everyone. The initial medication choices are stratified by HbA1c at entry – less than or greater than 7.5%, and greater than 9.0%. Each treatment path starts with drugs usually deemed safest. The algorithm recommends specific drug combinations as treatment intensifies. Each pathway has a 3-month window to explore the regimen’s effect. If that time expires without reaching the HbA1c goal, then the algorithm moves the patient to the next treatment level.
Unsuccessful triple therapy, or entry with an HbA1c of more than 9%, calls for basal insulin. Again, the algorithm specifies the order of treatment based on glucose levels. For a level of less than 8%, it calls for a total daily insulin dosage of 0.1-0.2 U/kg. For a blood sugar above 8%, the dosage is 0.2-0.3 U/kg. Both dosages should be titrated every 2-3 days to reach the glycemic goal. If the goal isn’t met, the algorithm moves on to prandial insulin at a total daily dosage of 0.3-0.5 U/kg.
Obesity management is a foundation treatment for all patients who are overweight. It starts with lifestyle modification, then moves through pharmacologic treatment and finally into surgical options.
The algorithm also tackles cardiovascular risk reduction, including lipid and blood pressure management, Dr. Grunberger said.
"An obese patient with dyslipidemia, high blood pressure, and diabetes is a ticking time bomb. All of these things need to be considered and treated together."
Dr. Grunberger is on the speakers board and scientific advisory panels for several pharmaceutical companies that manufacture diabetes treatment medications. He has also received research grants from a number of these companies.
FROM ENDOCRINE PRACTICE
Risk of HCV transmission very low in monogamous heterosexual couples
The risk of sexually transmitting a chronic hepatitis C infection to a long-term monogamous heterosexual partner is very low, averaging just about 1% per year.
That risk level works out to a transmission rate of about one in every 190,000 sexual contacts, Dr. Norah Terrault and her colleagues reported in the April issue of Hepatology (2013;57:881-9).
The cross-sectional study also found that no one sexual practice – including anal intercourse or intercourse during menses – significantly increased the risk of transmission, wrote Dr. Terrault of the University of California, San Francisco. The findings can be used to provide "unambiguous and reassuring counseling messages," she and her coinvestigators noted.
The study included 500 subjects with chronic HCV infections, and their sexual partners. All couples reported longtime, monogamous relationships (median duration, 15 years); however, the relationship duration varied widely, spanning 2-52 years.
Each of the partners was interviewed separately about their sexual contacts and practices. At the time of interview, the index subjects were a median of 49 years old and the partners, a median of 48 years.
The HCV-positive subjects reported the highest incidence of past risk factors, including blood transfusions before 1992 (32%), injected illegal drugs (54%), and being stuck by a bloody sharp item in a hospital (4%). Nearly half (46%) reported having had at least 20 lifetime sexual partners, with 21% having had 50 or more.
However, partners also reported some risk factors: 11% had an early transfusion, 2% used illegal drugs, and 2% had a hospital sharps incident. Many (27%) also reported having had at least 20 sexual partners.
Among the 500 couples, 20 partners (4%) were coinfected with HCV. Of these, nine were concordantly infected, eight discordantly, and three were indeterminate.
Six of the concordant couples underwent phylogenetic typing. Three were infected with the same HCV isolate and three with different strains. The investigators estimated the time of transmission and any additional risk factor among the three couples with concordant strains.
For the first couple, with an 18-year relationship, transmission probably occurred after about 6.5 years. The female partner had a history of injected drug use, while the male had no identifiable risk factors.
The second couple had a 28-year relationship; transmission probably occurred at around 15 years, the investigators said. "The female partner had a history of injectable drug use and both partners reported more than 20 prior sexual partners, a history of sexual transmitted diseases, and a history of snorting of drugs."
For the third couple, who had been together for 10 years, transmission probably occurred at around year 6. "The male partner had a history of injectable drug use, of being stuck by a sharp bloody object while working in a hospital, and more than 20 prior sexual partners; both partners reported snorting drugs and sharing snorting equipment."
The investigators determined that these infections were probably sexually transmitted between the partners – a prevalence of about 1%. "The estimated risk per sexual contact ranged from 1/380,000 to 1/190, 000," they said.
However, they were unable to identify any behaviors that significantly increased the risk of transmission. Compared with couples without coinfection, coinfected couples were more likely to have vaginal intercourse during menses (100% vs. 66%), more likely to have anal intercourse (67% vs. 30%), and less likely to use condoms (0% vs. 30%), but none of these differences was statistically significant.
"HCV transmission by sex from chronically infected persons to their heterosexual partners in a long-term monogamous relationship likely occurs, but is a rare event," the authors concluded. "Our results provide a basis for specific counseling messages that clinicians can use with their patients... [that] support the current national recommendations that couples not change their sexual practices if they are in a monogamous heterosexual relationship."
None of the study authors reported any financial conflicts.
The risk of sexually transmitting a chronic hepatitis C infection to a long-term monogamous heterosexual partner is very low, averaging just about 1% per year.
That risk level works out to a transmission rate of about one in every 190,000 sexual contacts, Dr. Norah Terrault and her colleagues reported in the April issue of Hepatology (2013;57:881-9).
The cross-sectional study also found that no one sexual practice – including anal intercourse or intercourse during menses – significantly increased the risk of transmission, wrote Dr. Terrault of the University of California, San Francisco. The findings can be used to provide "unambiguous and reassuring counseling messages," she and her coinvestigators noted.
The study included 500 subjects with chronic HCV infections, and their sexual partners. All couples reported longtime, monogamous relationships (median duration, 15 years); however, the relationship duration varied widely, spanning 2-52 years.
Each of the partners was interviewed separately about their sexual contacts and practices. At the time of interview, the index subjects were a median of 49 years old and the partners, a median of 48 years.
The HCV-positive subjects reported the highest incidence of past risk factors, including blood transfusions before 1992 (32%), injected illegal drugs (54%), and being stuck by a bloody sharp item in a hospital (4%). Nearly half (46%) reported having had at least 20 lifetime sexual partners, with 21% having had 50 or more.
However, partners also reported some risk factors: 11% had an early transfusion, 2% used illegal drugs, and 2% had a hospital sharps incident. Many (27%) also reported having had at least 20 sexual partners.
Among the 500 couples, 20 partners (4%) were coinfected with HCV. Of these, nine were concordantly infected, eight discordantly, and three were indeterminate.
Six of the concordant couples underwent phylogenetic typing. Three were infected with the same HCV isolate and three with different strains. The investigators estimated the time of transmission and any additional risk factor among the three couples with concordant strains.
For the first couple, with an 18-year relationship, transmission probably occurred after about 6.5 years. The female partner had a history of injected drug use, while the male had no identifiable risk factors.
The second couple had a 28-year relationship; transmission probably occurred at around 15 years, the investigators said. "The female partner had a history of injectable drug use and both partners reported more than 20 prior sexual partners, a history of sexual transmitted diseases, and a history of snorting of drugs."
For the third couple, who had been together for 10 years, transmission probably occurred at around year 6. "The male partner had a history of injectable drug use, of being stuck by a sharp bloody object while working in a hospital, and more than 20 prior sexual partners; both partners reported snorting drugs and sharing snorting equipment."
The investigators determined that these infections were probably sexually transmitted between the partners – a prevalence of about 1%. "The estimated risk per sexual contact ranged from 1/380,000 to 1/190, 000," they said.
However, they were unable to identify any behaviors that significantly increased the risk of transmission. Compared with couples without coinfection, coinfected couples were more likely to have vaginal intercourse during menses (100% vs. 66%), more likely to have anal intercourse (67% vs. 30%), and less likely to use condoms (0% vs. 30%), but none of these differences was statistically significant.
"HCV transmission by sex from chronically infected persons to their heterosexual partners in a long-term monogamous relationship likely occurs, but is a rare event," the authors concluded. "Our results provide a basis for specific counseling messages that clinicians can use with their patients... [that] support the current national recommendations that couples not change their sexual practices if they are in a monogamous heterosexual relationship."
None of the study authors reported any financial conflicts.
The risk of sexually transmitting a chronic hepatitis C infection to a long-term monogamous heterosexual partner is very low, averaging just about 1% per year.
That risk level works out to a transmission rate of about one in every 190,000 sexual contacts, Dr. Norah Terrault and her colleagues reported in the April issue of Hepatology (2013;57:881-9).
The cross-sectional study also found that no one sexual practice – including anal intercourse or intercourse during menses – significantly increased the risk of transmission, wrote Dr. Terrault of the University of California, San Francisco. The findings can be used to provide "unambiguous and reassuring counseling messages," she and her coinvestigators noted.
The study included 500 subjects with chronic HCV infections, and their sexual partners. All couples reported longtime, monogamous relationships (median duration, 15 years); however, the relationship duration varied widely, spanning 2-52 years.
Each of the partners was interviewed separately about their sexual contacts and practices. At the time of interview, the index subjects were a median of 49 years old and the partners, a median of 48 years.
The HCV-positive subjects reported the highest incidence of past risk factors, including blood transfusions before 1992 (32%), injected illegal drugs (54%), and being stuck by a bloody sharp item in a hospital (4%). Nearly half (46%) reported having had at least 20 lifetime sexual partners, with 21% having had 50 or more.
However, partners also reported some risk factors: 11% had an early transfusion, 2% used illegal drugs, and 2% had a hospital sharps incident. Many (27%) also reported having had at least 20 sexual partners.
Among the 500 couples, 20 partners (4%) were coinfected with HCV. Of these, nine were concordantly infected, eight discordantly, and three were indeterminate.
Six of the concordant couples underwent phylogenetic typing. Three were infected with the same HCV isolate and three with different strains. The investigators estimated the time of transmission and any additional risk factor among the three couples with concordant strains.
For the first couple, with an 18-year relationship, transmission probably occurred after about 6.5 years. The female partner had a history of injected drug use, while the male had no identifiable risk factors.
The second couple had a 28-year relationship; transmission probably occurred at around 15 years, the investigators said. "The female partner had a history of injectable drug use and both partners reported more than 20 prior sexual partners, a history of sexual transmitted diseases, and a history of snorting of drugs."
For the third couple, who had been together for 10 years, transmission probably occurred at around year 6. "The male partner had a history of injectable drug use, of being stuck by a sharp bloody object while working in a hospital, and more than 20 prior sexual partners; both partners reported snorting drugs and sharing snorting equipment."
The investigators determined that these infections were probably sexually transmitted between the partners – a prevalence of about 1%. "The estimated risk per sexual contact ranged from 1/380,000 to 1/190, 000," they said.
However, they were unable to identify any behaviors that significantly increased the risk of transmission. Compared with couples without coinfection, coinfected couples were more likely to have vaginal intercourse during menses (100% vs. 66%), more likely to have anal intercourse (67% vs. 30%), and less likely to use condoms (0% vs. 30%), but none of these differences was statistically significant.
"HCV transmission by sex from chronically infected persons to their heterosexual partners in a long-term monogamous relationship likely occurs, but is a rare event," the authors concluded. "Our results provide a basis for specific counseling messages that clinicians can use with their patients... [that] support the current national recommendations that couples not change their sexual practices if they are in a monogamous heterosexual relationship."
None of the study authors reported any financial conflicts.
FROM HEPATOLOGY
Major finding: Sexual transmission of chronic hepatitis C infection averaged about 1% per year in monogamous heterosexual couples.
Data source: A cross-sectional study of 500 couples in long-term monogamous relationships.
Disclosures: None of the study authors reported any financial disclosures.
New blood test could identify early pancreatic cancer
An investigational serum biomarker panel accurately identified potentially resectable cases of pancreatic cancer, opening up the possibility that a blood test could identify the cancer early and improve its dismal prognosis.
The panel measured four metabolites and had an overall sensitivity of 71% and a specificity of 78% for pancreatic cancer; the specificity for resectable tumors also approached 78%, reported Dr. Masaru Yoshida and his colleagues (Cancer Epidemiol. Biomarkers Prev. 2013 March 29 [doi: 10.1158/1055-9965.EPI-12-1033]).
The panel was more accurate than were any of the currently used biomarkers, including CA19-9 and CEA, said Dr. Yoshida, professor and chief of metabolomics research at Kobe University, Japan, and his colleagues.
"This novel diagnostic approach, which is safe and easy to apply as a screening method, is expected to improve the prognosis of patients with pancreatic cancer by detecting their cancers early, when still in a resectable and curable state," Dr. Yoshida said in a press statement.
The researchers created the panel based on a study of a cohort of 85 subjects: 43 with pancreatic cancer and 42 healthy controls. First, they pared down 113 potentially useful metabolites into a workable testing panel. Of the 45 candidate metabolites, four of them – xylitol, 1,5-anhydro-D-glucitol, histidine, and inositol – showed the highest variation between the healthy controls and the patients. In the training cohort, the panel of these four markers had a sensitivity of 86% and specificity of 88% for pancreatic cancer.
Next, the researchers evaluated a validation cohort of 42 cancer patients, 41 healthy controls, and 23 patients with chronic pancreatitis. In this cohort, with an area under the curve of 0.76, the panel’s sensitivity for pancreatic cancer was 71% and specificity, 78%. In contrast, the sensitivity of CA19-9 was 69% and specificity 86%; for CEA, those numbers were 36% and 80%, respectively.
In the subset of patients with resectable pancreatic cancer, the panel’s sensitivity was 78%, compared with 56% for CA19-9 and 44% for CEA.
The metabolic panel was also able to identify chronic pancreatitis, with a 17% rate of false positives, compared with a false-positive rate of 30% for CA19-9 and 44% for CEA.
A blood panel could address three key problems in the field of pancreatic cancer, Dr. Yoshida and his colleagues said. "The first is the difficulty of detecting pancreatic cancer early in resectable stages. A sensitivity and specificity of about 80% for resectable disease ... should be acceptable for clinical use, because most patients with resectable cancer have no symptoms, and blood examinations are useful tools for initial screening examinations."
The second problem is the difficulty in differentiating pancreatic cancer from chronic pancreatitis. "Many gastroenterologists follow up chronic pancreatitis with scheduled CT, magnetic resonance imaging (MRI), and endoscopic ultrasound scans (EUS); tumor marker tests; and endoscopic retrograde cholangiopancreatography (ERCP), but the initial malignant changes are frequently overlooked, and pancreatic tumors can rapidly become unresectable. In addition, unnecessary resections for benign inflammatory lesions are sometimes done because of false-positive results from CA19-9 and/or imaging examinations."
The third clinical problem is the risk of complications that result from pancreatic examinations. Serum samples are easy and safe to obtain, with a low risk of adverse events and, with this panel, a potentially good return of information. A blood test could also be a useful population screening tool, the researchers noted.
The metabolites probably reflect metabolic derangement that arises not only from focal tumorigenesis, but also from systemic reactions to pancreatic disease, Dr. Yoshida and his coinvestigators wrote. Pancreatic cancer causes significant decreases in some amino and fatty acids, probably because the tumor recruits these substances to aid its rapid cellular proliferation.
"Patients with pancreatic disease are also troubled by malnutrition because of pancreatic endocrine and exocrine insufficiency. Therefore, there is a possibility that the decreases in serum metabolite levels also reflect malnutrition."
Finally, they said, the decrease in 1,5-anhydro-D-glucitol indicates the presence of hyperglycemia and recent glycosuria. "These results suggest that glucose tolerance was impaired in these patients because of pancreatic insufficiency."
The authors reported no financial conflicts. The work was supported by grants administered by agencies of the Japanese government.
The results of Kobayashi et al. are encouraging in many respects, but need to be viewed with caution. The first question is whether the findings are repeatable in other populations. If so, their test may prove to be more sensitive and specific than CA19-9 and CEA for smaller pancreatic cancers, especially in the setting of chronic pancreatitis. Replication in larger studies is important because this study was relatively small, with only nine cases of cancer stage 0-IIB. Second, is the assay more rapid, reliable, robust, and/or significantly less expensive than CA19-9 and CEA? These factors are important for physician and health care system acceptance. Third, this approach has not been adequately tested in other disease states, with the important exception of chronic pancreatitis. Fourth, the test was done in the serum of patients who were already diagnosed with pancreatic cancer by other methods. Even in this setting, the sensitivity and specificity were inferior to clinical suspicion and imaging techniques.
The study was not designed to evaluate this test for early detection of pancreatic cancer, and with the current sensitivity and specificity, this test will have little if any value in screening of otherwise healthy populations. Finally, this approach did identify specific metabolites that were different between pancreatic cancer, chronic pancreatitis, and controls, which may provide further insights into the metabolic derangements of pancreatic cancer. Thus, it is too early to embrace or reject this new approach in the average clinic, but it does provide new insights and perspectives – and is moving us in a positive direction.
David C. Whitcomb, M.D., Ph.D., is the Giant Eagle Professor of Cancer Genetics; professor of medicine, cell biology, and physiology and human genetics; and chief of the division of gastroenterology, hepatology, and nutrition at the University of Pittsburgh and University of Pittsburgh Medical Center. He reports no significant financial conflicts.
The results of Kobayashi et al. are encouraging in many respects, but need to be viewed with caution. The first question is whether the findings are repeatable in other populations. If so, their test may prove to be more sensitive and specific than CA19-9 and CEA for smaller pancreatic cancers, especially in the setting of chronic pancreatitis. Replication in larger studies is important because this study was relatively small, with only nine cases of cancer stage 0-IIB. Second, is the assay more rapid, reliable, robust, and/or significantly less expensive than CA19-9 and CEA? These factors are important for physician and health care system acceptance. Third, this approach has not been adequately tested in other disease states, with the important exception of chronic pancreatitis. Fourth, the test was done in the serum of patients who were already diagnosed with pancreatic cancer by other methods. Even in this setting, the sensitivity and specificity were inferior to clinical suspicion and imaging techniques.
The study was not designed to evaluate this test for early detection of pancreatic cancer, and with the current sensitivity and specificity, this test will have little if any value in screening of otherwise healthy populations. Finally, this approach did identify specific metabolites that were different between pancreatic cancer, chronic pancreatitis, and controls, which may provide further insights into the metabolic derangements of pancreatic cancer. Thus, it is too early to embrace or reject this new approach in the average clinic, but it does provide new insights and perspectives – and is moving us in a positive direction.
David C. Whitcomb, M.D., Ph.D., is the Giant Eagle Professor of Cancer Genetics; professor of medicine, cell biology, and physiology and human genetics; and chief of the division of gastroenterology, hepatology, and nutrition at the University of Pittsburgh and University of Pittsburgh Medical Center. He reports no significant financial conflicts.
The results of Kobayashi et al. are encouraging in many respects, but need to be viewed with caution. The first question is whether the findings are repeatable in other populations. If so, their test may prove to be more sensitive and specific than CA19-9 and CEA for smaller pancreatic cancers, especially in the setting of chronic pancreatitis. Replication in larger studies is important because this study was relatively small, with only nine cases of cancer stage 0-IIB. Second, is the assay more rapid, reliable, robust, and/or significantly less expensive than CA19-9 and CEA? These factors are important for physician and health care system acceptance. Third, this approach has not been adequately tested in other disease states, with the important exception of chronic pancreatitis. Fourth, the test was done in the serum of patients who were already diagnosed with pancreatic cancer by other methods. Even in this setting, the sensitivity and specificity were inferior to clinical suspicion and imaging techniques.
The study was not designed to evaluate this test for early detection of pancreatic cancer, and with the current sensitivity and specificity, this test will have little if any value in screening of otherwise healthy populations. Finally, this approach did identify specific metabolites that were different between pancreatic cancer, chronic pancreatitis, and controls, which may provide further insights into the metabolic derangements of pancreatic cancer. Thus, it is too early to embrace or reject this new approach in the average clinic, but it does provide new insights and perspectives – and is moving us in a positive direction.
David C. Whitcomb, M.D., Ph.D., is the Giant Eagle Professor of Cancer Genetics; professor of medicine, cell biology, and physiology and human genetics; and chief of the division of gastroenterology, hepatology, and nutrition at the University of Pittsburgh and University of Pittsburgh Medical Center. He reports no significant financial conflicts.
An investigational serum biomarker panel accurately identified potentially resectable cases of pancreatic cancer, opening up the possibility that a blood test could identify the cancer early and improve its dismal prognosis.
The panel measured four metabolites and had an overall sensitivity of 71% and a specificity of 78% for pancreatic cancer; the specificity for resectable tumors also approached 78%, reported Dr. Masaru Yoshida and his colleagues (Cancer Epidemiol. Biomarkers Prev. 2013 March 29 [doi: 10.1158/1055-9965.EPI-12-1033]).
The panel was more accurate than were any of the currently used biomarkers, including CA19-9 and CEA, said Dr. Yoshida, professor and chief of metabolomics research at Kobe University, Japan, and his colleagues.
"This novel diagnostic approach, which is safe and easy to apply as a screening method, is expected to improve the prognosis of patients with pancreatic cancer by detecting their cancers early, when still in a resectable and curable state," Dr. Yoshida said in a press statement.
The researchers created the panel based on a study of a cohort of 85 subjects: 43 with pancreatic cancer and 42 healthy controls. First, they pared down 113 potentially useful metabolites into a workable testing panel. Of the 45 candidate metabolites, four of them – xylitol, 1,5-anhydro-D-glucitol, histidine, and inositol – showed the highest variation between the healthy controls and the patients. In the training cohort, the panel of these four markers had a sensitivity of 86% and specificity of 88% for pancreatic cancer.
Next, the researchers evaluated a validation cohort of 42 cancer patients, 41 healthy controls, and 23 patients with chronic pancreatitis. In this cohort, with an area under the curve of 0.76, the panel’s sensitivity for pancreatic cancer was 71% and specificity, 78%. In contrast, the sensitivity of CA19-9 was 69% and specificity 86%; for CEA, those numbers were 36% and 80%, respectively.
In the subset of patients with resectable pancreatic cancer, the panel’s sensitivity was 78%, compared with 56% for CA19-9 and 44% for CEA.
The metabolic panel was also able to identify chronic pancreatitis, with a 17% rate of false positives, compared with a false-positive rate of 30% for CA19-9 and 44% for CEA.
A blood panel could address three key problems in the field of pancreatic cancer, Dr. Yoshida and his colleagues said. "The first is the difficulty of detecting pancreatic cancer early in resectable stages. A sensitivity and specificity of about 80% for resectable disease ... should be acceptable for clinical use, because most patients with resectable cancer have no symptoms, and blood examinations are useful tools for initial screening examinations."
The second problem is the difficulty in differentiating pancreatic cancer from chronic pancreatitis. "Many gastroenterologists follow up chronic pancreatitis with scheduled CT, magnetic resonance imaging (MRI), and endoscopic ultrasound scans (EUS); tumor marker tests; and endoscopic retrograde cholangiopancreatography (ERCP), but the initial malignant changes are frequently overlooked, and pancreatic tumors can rapidly become unresectable. In addition, unnecessary resections for benign inflammatory lesions are sometimes done because of false-positive results from CA19-9 and/or imaging examinations."
The third clinical problem is the risk of complications that result from pancreatic examinations. Serum samples are easy and safe to obtain, with a low risk of adverse events and, with this panel, a potentially good return of information. A blood test could also be a useful population screening tool, the researchers noted.
The metabolites probably reflect metabolic derangement that arises not only from focal tumorigenesis, but also from systemic reactions to pancreatic disease, Dr. Yoshida and his coinvestigators wrote. Pancreatic cancer causes significant decreases in some amino and fatty acids, probably because the tumor recruits these substances to aid its rapid cellular proliferation.
"Patients with pancreatic disease are also troubled by malnutrition because of pancreatic endocrine and exocrine insufficiency. Therefore, there is a possibility that the decreases in serum metabolite levels also reflect malnutrition."
Finally, they said, the decrease in 1,5-anhydro-D-glucitol indicates the presence of hyperglycemia and recent glycosuria. "These results suggest that glucose tolerance was impaired in these patients because of pancreatic insufficiency."
The authors reported no financial conflicts. The work was supported by grants administered by agencies of the Japanese government.
An investigational serum biomarker panel accurately identified potentially resectable cases of pancreatic cancer, opening up the possibility that a blood test could identify the cancer early and improve its dismal prognosis.
The panel measured four metabolites and had an overall sensitivity of 71% and a specificity of 78% for pancreatic cancer; the specificity for resectable tumors also approached 78%, reported Dr. Masaru Yoshida and his colleagues (Cancer Epidemiol. Biomarkers Prev. 2013 March 29 [doi: 10.1158/1055-9965.EPI-12-1033]).
The panel was more accurate than were any of the currently used biomarkers, including CA19-9 and CEA, said Dr. Yoshida, professor and chief of metabolomics research at Kobe University, Japan, and his colleagues.
"This novel diagnostic approach, which is safe and easy to apply as a screening method, is expected to improve the prognosis of patients with pancreatic cancer by detecting their cancers early, when still in a resectable and curable state," Dr. Yoshida said in a press statement.
The researchers created the panel based on a study of a cohort of 85 subjects: 43 with pancreatic cancer and 42 healthy controls. First, they pared down 113 potentially useful metabolites into a workable testing panel. Of the 45 candidate metabolites, four of them – xylitol, 1,5-anhydro-D-glucitol, histidine, and inositol – showed the highest variation between the healthy controls and the patients. In the training cohort, the panel of these four markers had a sensitivity of 86% and specificity of 88% for pancreatic cancer.
Next, the researchers evaluated a validation cohort of 42 cancer patients, 41 healthy controls, and 23 patients with chronic pancreatitis. In this cohort, with an area under the curve of 0.76, the panel’s sensitivity for pancreatic cancer was 71% and specificity, 78%. In contrast, the sensitivity of CA19-9 was 69% and specificity 86%; for CEA, those numbers were 36% and 80%, respectively.
In the subset of patients with resectable pancreatic cancer, the panel’s sensitivity was 78%, compared with 56% for CA19-9 and 44% for CEA.
The metabolic panel was also able to identify chronic pancreatitis, with a 17% rate of false positives, compared with a false-positive rate of 30% for CA19-9 and 44% for CEA.
A blood panel could address three key problems in the field of pancreatic cancer, Dr. Yoshida and his colleagues said. "The first is the difficulty of detecting pancreatic cancer early in resectable stages. A sensitivity and specificity of about 80% for resectable disease ... should be acceptable for clinical use, because most patients with resectable cancer have no symptoms, and blood examinations are useful tools for initial screening examinations."
The second problem is the difficulty in differentiating pancreatic cancer from chronic pancreatitis. "Many gastroenterologists follow up chronic pancreatitis with scheduled CT, magnetic resonance imaging (MRI), and endoscopic ultrasound scans (EUS); tumor marker tests; and endoscopic retrograde cholangiopancreatography (ERCP), but the initial malignant changes are frequently overlooked, and pancreatic tumors can rapidly become unresectable. In addition, unnecessary resections for benign inflammatory lesions are sometimes done because of false-positive results from CA19-9 and/or imaging examinations."
The third clinical problem is the risk of complications that result from pancreatic examinations. Serum samples are easy and safe to obtain, with a low risk of adverse events and, with this panel, a potentially good return of information. A blood test could also be a useful population screening tool, the researchers noted.
The metabolites probably reflect metabolic derangement that arises not only from focal tumorigenesis, but also from systemic reactions to pancreatic disease, Dr. Yoshida and his coinvestigators wrote. Pancreatic cancer causes significant decreases in some amino and fatty acids, probably because the tumor recruits these substances to aid its rapid cellular proliferation.
"Patients with pancreatic disease are also troubled by malnutrition because of pancreatic endocrine and exocrine insufficiency. Therefore, there is a possibility that the decreases in serum metabolite levels also reflect malnutrition."
Finally, they said, the decrease in 1,5-anhydro-D-glucitol indicates the presence of hyperglycemia and recent glycosuria. "These results suggest that glucose tolerance was impaired in these patients because of pancreatic insufficiency."
The authors reported no financial conflicts. The work was supported by grants administered by agencies of the Japanese government.
Major finding: A serum panel of four metabolites had a 78% sensitivity for detecting resectable pancreatic cancers.
Data source: The panel was derived from a test cohort of 85, and a validation cohort of 42 cancer patients, 41 healthy controls, and 23 patients with chronic pancreatitis.
Disclosures: The authors had no financial disclosures. Japanese government agencies funded the study.
Study suggests statin use decreases breast cancer mortality
WASHINGTON – Statin use – both before and after a diagnosis of breast cancer – was associated with up to a 66% reduction in the risk of dying from that cancer, a large database study has concluded.
Investigators reported that the risk reduction was consistent even after controlling for age, tumor stage and morphology, and primary treatment choice, Dr. Teemu Murtola said during an interview at the annual meeting of the American Association for Cancer Research.
"We saw that there was clearly a dose-dependent reduction in the risk of breast cancer mortality among statin users," who comprised 13% of the more than 31,000 women in the study, said Dr. Murtola, an epidemiologist at Johns Hopkins University in Baltimore. "The association held for both localized and metastatic tumors. There really appears to be something going on here, and it’s certainly a reason for further study."
During the follow-up, 6,011 women died; 3,169 deaths were due to breast cancer. The death rate among users was significantly lower than it was among nonusers (7.5% vs. 21%). In the adjusted analysis, statin users were 67% less likely to die than were nonusers with localized disease (hazard ratio, 0.33). Among those with metastatic disease, statins conferred a 48% decreased risk of death (HR, 0.52).
All of the statins investigated in the study cohort were associated with a significantly decreased risk of breast cancer death, including simvastatin (HR, 0.47), atorvastatin (HR, 0.27), fluvastatin (HR, 0.35), and pravastatin (HR, 0.50).
The retrospective study looked at statin use and breast cancer mortality among 31,114 women with breast cancer who were diagnosed in Finland during 1995-2003. All of the patients were included in the Finnish Cancer Registry. The country’s national health database also contained detailed information on statin use and other health indicators. The data on statins included the date of each prescription purchase, the numbers of refills, the dosage, and the number of pills in each refill.
Breast cancer data included the date of diagnosis, stage (local, lymph-node positive or metastatic), morphology, primary treatment choice (surgery, radiation, chemotherapy, hormonal therapy, or other), and the date and cause of death.
Follow-up started at the time of diagnosis and continued until death or the common closing date of Dec. 31, 2003, whichever came first. Median follow-up was about 3 years, but ranged from less than 1 year to 9 years.
Statin users accounted for 13% of the cohort (4,169); the remainder never used the drugs. Women who took the medications were significantly older than nonusers (64 years vs. 58 years), and more likely to have had surgery as a treatment (96% vs. 92%). They were also more likely to have undergone radiation (55% vs. 54%), but significantly less likely to have had chemotherapy (15% vs. 24%) or hormonal therapy (20% vs. 25%). Metastatic cancer was significantly less common among statin users – 4% vs. 7%. These differences may reflect an early identification of breast cancer among users, perhaps because they were visiting a physician more frequently for lipid monitoring, Dr. Murtola said. However, the multivariate analysis controlled for all these differences, including metastatic and local disease.
Dr. Murtola then investigated the mortality effects of both pre- and postdiagnostic statin use.
Compliance was high among women who started the drugs before their breast cancer diagnosis, with 85% continuing until the end of follow-up. Compared with nonusers, women who used statins for up to 490 days were 46% less likely to die of local disease and 30% less likely to die from metastatic disease.
Those who took the drugs for 491 days or more before diagnosis were 66% less likely to die from local disease and 40% less likely to die from metastatic disease.
There was no such clear, dose-dependent relationship between mortality risk and statins taken after diagnosis, Dr. Murtola said. But, he added, low compliance could have masked any benefit. "Only 14% of those who started statins after diagnosis continued to take them until the end of the follow-up period," he said. "When we limited the analysis to those who were compliant until the end of their follow-up, we saw a very similar dose-dependent risk reduction in breast cancer mortality."
In vitro studies suggest that statins could have a direct effect on breast cancer cells, he said. They seem to block the mevalonate pathway – a process involved in the manufacture of cholesterol, and also key to cell metabolism. Cells with abnormally high metabolic rates – like breast cancer cells – could be particularly vulnerable to this action, he said.
"I think that because the drugs target this pathway their impact extends beyond cholesterol. It’s certainly worth further study."
Dr. Murtola had no financial disclosures.
WASHINGTON – Statin use – both before and after a diagnosis of breast cancer – was associated with up to a 66% reduction in the risk of dying from that cancer, a large database study has concluded.
Investigators reported that the risk reduction was consistent even after controlling for age, tumor stage and morphology, and primary treatment choice, Dr. Teemu Murtola said during an interview at the annual meeting of the American Association for Cancer Research.
"We saw that there was clearly a dose-dependent reduction in the risk of breast cancer mortality among statin users," who comprised 13% of the more than 31,000 women in the study, said Dr. Murtola, an epidemiologist at Johns Hopkins University in Baltimore. "The association held for both localized and metastatic tumors. There really appears to be something going on here, and it’s certainly a reason for further study."
During the follow-up, 6,011 women died; 3,169 deaths were due to breast cancer. The death rate among users was significantly lower than it was among nonusers (7.5% vs. 21%). In the adjusted analysis, statin users were 67% less likely to die than were nonusers with localized disease (hazard ratio, 0.33). Among those with metastatic disease, statins conferred a 48% decreased risk of death (HR, 0.52).
All of the statins investigated in the study cohort were associated with a significantly decreased risk of breast cancer death, including simvastatin (HR, 0.47), atorvastatin (HR, 0.27), fluvastatin (HR, 0.35), and pravastatin (HR, 0.50).
The retrospective study looked at statin use and breast cancer mortality among 31,114 women with breast cancer who were diagnosed in Finland during 1995-2003. All of the patients were included in the Finnish Cancer Registry. The country’s national health database also contained detailed information on statin use and other health indicators. The data on statins included the date of each prescription purchase, the numbers of refills, the dosage, and the number of pills in each refill.
Breast cancer data included the date of diagnosis, stage (local, lymph-node positive or metastatic), morphology, primary treatment choice (surgery, radiation, chemotherapy, hormonal therapy, or other), and the date and cause of death.
Follow-up started at the time of diagnosis and continued until death or the common closing date of Dec. 31, 2003, whichever came first. Median follow-up was about 3 years, but ranged from less than 1 year to 9 years.
Statin users accounted for 13% of the cohort (4,169); the remainder never used the drugs. Women who took the medications were significantly older than nonusers (64 years vs. 58 years), and more likely to have had surgery as a treatment (96% vs. 92%). They were also more likely to have undergone radiation (55% vs. 54%), but significantly less likely to have had chemotherapy (15% vs. 24%) or hormonal therapy (20% vs. 25%). Metastatic cancer was significantly less common among statin users – 4% vs. 7%. These differences may reflect an early identification of breast cancer among users, perhaps because they were visiting a physician more frequently for lipid monitoring, Dr. Murtola said. However, the multivariate analysis controlled for all these differences, including metastatic and local disease.
Dr. Murtola then investigated the mortality effects of both pre- and postdiagnostic statin use.
Compliance was high among women who started the drugs before their breast cancer diagnosis, with 85% continuing until the end of follow-up. Compared with nonusers, women who used statins for up to 490 days were 46% less likely to die of local disease and 30% less likely to die from metastatic disease.
Those who took the drugs for 491 days or more before diagnosis were 66% less likely to die from local disease and 40% less likely to die from metastatic disease.
There was no such clear, dose-dependent relationship between mortality risk and statins taken after diagnosis, Dr. Murtola said. But, he added, low compliance could have masked any benefit. "Only 14% of those who started statins after diagnosis continued to take them until the end of the follow-up period," he said. "When we limited the analysis to those who were compliant until the end of their follow-up, we saw a very similar dose-dependent risk reduction in breast cancer mortality."
In vitro studies suggest that statins could have a direct effect on breast cancer cells, he said. They seem to block the mevalonate pathway – a process involved in the manufacture of cholesterol, and also key to cell metabolism. Cells with abnormally high metabolic rates – like breast cancer cells – could be particularly vulnerable to this action, he said.
"I think that because the drugs target this pathway their impact extends beyond cholesterol. It’s certainly worth further study."
Dr. Murtola had no financial disclosures.
WASHINGTON – Statin use – both before and after a diagnosis of breast cancer – was associated with up to a 66% reduction in the risk of dying from that cancer, a large database study has concluded.
Investigators reported that the risk reduction was consistent even after controlling for age, tumor stage and morphology, and primary treatment choice, Dr. Teemu Murtola said during an interview at the annual meeting of the American Association for Cancer Research.
"We saw that there was clearly a dose-dependent reduction in the risk of breast cancer mortality among statin users," who comprised 13% of the more than 31,000 women in the study, said Dr. Murtola, an epidemiologist at Johns Hopkins University in Baltimore. "The association held for both localized and metastatic tumors. There really appears to be something going on here, and it’s certainly a reason for further study."
During the follow-up, 6,011 women died; 3,169 deaths were due to breast cancer. The death rate among users was significantly lower than it was among nonusers (7.5% vs. 21%). In the adjusted analysis, statin users were 67% less likely to die than were nonusers with localized disease (hazard ratio, 0.33). Among those with metastatic disease, statins conferred a 48% decreased risk of death (HR, 0.52).
All of the statins investigated in the study cohort were associated with a significantly decreased risk of breast cancer death, including simvastatin (HR, 0.47), atorvastatin (HR, 0.27), fluvastatin (HR, 0.35), and pravastatin (HR, 0.50).
The retrospective study looked at statin use and breast cancer mortality among 31,114 women with breast cancer who were diagnosed in Finland during 1995-2003. All of the patients were included in the Finnish Cancer Registry. The country’s national health database also contained detailed information on statin use and other health indicators. The data on statins included the date of each prescription purchase, the numbers of refills, the dosage, and the number of pills in each refill.
Breast cancer data included the date of diagnosis, stage (local, lymph-node positive or metastatic), morphology, primary treatment choice (surgery, radiation, chemotherapy, hormonal therapy, or other), and the date and cause of death.
Follow-up started at the time of diagnosis and continued until death or the common closing date of Dec. 31, 2003, whichever came first. Median follow-up was about 3 years, but ranged from less than 1 year to 9 years.
Statin users accounted for 13% of the cohort (4,169); the remainder never used the drugs. Women who took the medications were significantly older than nonusers (64 years vs. 58 years), and more likely to have had surgery as a treatment (96% vs. 92%). They were also more likely to have undergone radiation (55% vs. 54%), but significantly less likely to have had chemotherapy (15% vs. 24%) or hormonal therapy (20% vs. 25%). Metastatic cancer was significantly less common among statin users – 4% vs. 7%. These differences may reflect an early identification of breast cancer among users, perhaps because they were visiting a physician more frequently for lipid monitoring, Dr. Murtola said. However, the multivariate analysis controlled for all these differences, including metastatic and local disease.
Dr. Murtola then investigated the mortality effects of both pre- and postdiagnostic statin use.
Compliance was high among women who started the drugs before their breast cancer diagnosis, with 85% continuing until the end of follow-up. Compared with nonusers, women who used statins for up to 490 days were 46% less likely to die of local disease and 30% less likely to die from metastatic disease.
Those who took the drugs for 491 days or more before diagnosis were 66% less likely to die from local disease and 40% less likely to die from metastatic disease.
There was no such clear, dose-dependent relationship between mortality risk and statins taken after diagnosis, Dr. Murtola said. But, he added, low compliance could have masked any benefit. "Only 14% of those who started statins after diagnosis continued to take them until the end of the follow-up period," he said. "When we limited the analysis to those who were compliant until the end of their follow-up, we saw a very similar dose-dependent risk reduction in breast cancer mortality."
In vitro studies suggest that statins could have a direct effect on breast cancer cells, he said. They seem to block the mevalonate pathway – a process involved in the manufacture of cholesterol, and also key to cell metabolism. Cells with abnormally high metabolic rates – like breast cancer cells – could be particularly vulnerable to this action, he said.
"I think that because the drugs target this pathway their impact extends beyond cholesterol. It’s certainly worth further study."
Dr. Murtola had no financial disclosures.
AT THE AACR ANNUAL MEETING
Major finding: Women who took statins before a diagnosis of breast cancer were up to 67% less likely to die from their disease than were those who never took the medications.
Data source: The database study comprised 31,114 women with breast cancer included in the Finnish Cancer Registry.
Disclosures: Dr. Murtola had no financial disclosures.
Reflux after surgery increases risk of esophageal cancer
Patients who experience recurrent reflux despite surgical treatment are three times more likely to develop esophageal adenocarcinoma than are those who have a successful surgery.
The findings of a national database study suggest that careful observation may be key to prevention of cancer when antireflux surgery doesn’t deliver, reported Dr. Hedvig E. Lofdahl and colleagues. The study was published in the April issue of Annals of Surgery.
"From a clinical point of view, this study suggests that it might be valuable to carefully evaluate the result of the antireflux surgery, and consider the patients with recurrent GERD, particularly those with Barrett’s esophagus, for endoscopic surveillance," wrote Dr. Lofdahl of Karolinska Institutet, Stockholm, and his coauthors (Ann. Surg. 2013;257:579-82).
The case-control study drew its data from the Swedish Cancer Register. It comprised 295 patients who underwent antireflux surgery from 1996 to 2006. Fifty-five of the patients developed an adenocarcinoma of the esophagus sometime during the 7-year follow-up period.
Most of the patients in the study were male (87%). Smoking status did not differ significantly between the cases and controls (47% vs.42%, respectively). Recurrent reflux was significantly more common among the cases than among the controls (35% vs. 18%).
The multivariate analysis controlled for body mass index, smoking, and the type of antireflux surgery. In the final adjusted model, recurrent reflux conferred a threefold increase in the risk of a later esophageal adenocarcinoma. A BMI of more than 25 kg/m2 also increased the risk, but not significantly (odds ratio [OR] 1.6; confidence interval [CI]: 0.8-3.5). There were not enough patients with a BMI of greater than 30 kg/m2 to further tease out the effect of weight.
Having ever smoked tobacco also increased the risk of esophageal cancer, but again, the increase was not statistically significant (OR 1.4; CI: 0.7-2.8).
Compared with a partial fundoplication, a total 360-degree fundoplication was associated with a lower risk of cancer, but the difference was not statistically significant (OR 0.6; CI: 0.3-1.3).
"This finding might at least partly explain the lack of cancer-preventive effect of antireflux surgery," the investigators wrote.
The study was supported by the Swedish Research Council, the Swedish Cancer Society, and the Stockholm Cancer Society. None of the authors had financial disclosures.
Patients who experience recurrent reflux despite surgical treatment are three times more likely to develop esophageal adenocarcinoma than are those who have a successful surgery.
The findings of a national database study suggest that careful observation may be key to prevention of cancer when antireflux surgery doesn’t deliver, reported Dr. Hedvig E. Lofdahl and colleagues. The study was published in the April issue of Annals of Surgery.
"From a clinical point of view, this study suggests that it might be valuable to carefully evaluate the result of the antireflux surgery, and consider the patients with recurrent GERD, particularly those with Barrett’s esophagus, for endoscopic surveillance," wrote Dr. Lofdahl of Karolinska Institutet, Stockholm, and his coauthors (Ann. Surg. 2013;257:579-82).
The case-control study drew its data from the Swedish Cancer Register. It comprised 295 patients who underwent antireflux surgery from 1996 to 2006. Fifty-five of the patients developed an adenocarcinoma of the esophagus sometime during the 7-year follow-up period.
Most of the patients in the study were male (87%). Smoking status did not differ significantly between the cases and controls (47% vs.42%, respectively). Recurrent reflux was significantly more common among the cases than among the controls (35% vs. 18%).
The multivariate analysis controlled for body mass index, smoking, and the type of antireflux surgery. In the final adjusted model, recurrent reflux conferred a threefold increase in the risk of a later esophageal adenocarcinoma. A BMI of more than 25 kg/m2 also increased the risk, but not significantly (odds ratio [OR] 1.6; confidence interval [CI]: 0.8-3.5). There were not enough patients with a BMI of greater than 30 kg/m2 to further tease out the effect of weight.
Having ever smoked tobacco also increased the risk of esophageal cancer, but again, the increase was not statistically significant (OR 1.4; CI: 0.7-2.8).
Compared with a partial fundoplication, a total 360-degree fundoplication was associated with a lower risk of cancer, but the difference was not statistically significant (OR 0.6; CI: 0.3-1.3).
"This finding might at least partly explain the lack of cancer-preventive effect of antireflux surgery," the investigators wrote.
The study was supported by the Swedish Research Council, the Swedish Cancer Society, and the Stockholm Cancer Society. None of the authors had financial disclosures.
Patients who experience recurrent reflux despite surgical treatment are three times more likely to develop esophageal adenocarcinoma than are those who have a successful surgery.
The findings of a national database study suggest that careful observation may be key to prevention of cancer when antireflux surgery doesn’t deliver, reported Dr. Hedvig E. Lofdahl and colleagues. The study was published in the April issue of Annals of Surgery.
"From a clinical point of view, this study suggests that it might be valuable to carefully evaluate the result of the antireflux surgery, and consider the patients with recurrent GERD, particularly those with Barrett’s esophagus, for endoscopic surveillance," wrote Dr. Lofdahl of Karolinska Institutet, Stockholm, and his coauthors (Ann. Surg. 2013;257:579-82).
The case-control study drew its data from the Swedish Cancer Register. It comprised 295 patients who underwent antireflux surgery from 1996 to 2006. Fifty-five of the patients developed an adenocarcinoma of the esophagus sometime during the 7-year follow-up period.
Most of the patients in the study were male (87%). Smoking status did not differ significantly between the cases and controls (47% vs.42%, respectively). Recurrent reflux was significantly more common among the cases than among the controls (35% vs. 18%).
The multivariate analysis controlled for body mass index, smoking, and the type of antireflux surgery. In the final adjusted model, recurrent reflux conferred a threefold increase in the risk of a later esophageal adenocarcinoma. A BMI of more than 25 kg/m2 also increased the risk, but not significantly (odds ratio [OR] 1.6; confidence interval [CI]: 0.8-3.5). There were not enough patients with a BMI of greater than 30 kg/m2 to further tease out the effect of weight.
Having ever smoked tobacco also increased the risk of esophageal cancer, but again, the increase was not statistically significant (OR 1.4; CI: 0.7-2.8).
Compared with a partial fundoplication, a total 360-degree fundoplication was associated with a lower risk of cancer, but the difference was not statistically significant (OR 0.6; CI: 0.3-1.3).
"This finding might at least partly explain the lack of cancer-preventive effect of antireflux surgery," the investigators wrote.
The study was supported by the Swedish Research Council, the Swedish Cancer Society, and the Stockholm Cancer Society. None of the authors had financial disclosures.
FROM ANNALS OF SURGERY
Major finding: Patients with recurrent reflux after surgical treatment are three times more likely to develop an esophageal adenocarcinoma than are those whose symptoms resolve after surgery,
Data source: The case-control study comprised 295 patients included in a national cancer registry.
Disclosures: The study was supported by the Swedish Research Council, the Swedish Cancer Society, and the Stockholm Cancer Society. None of the authors had financial disclosures.
DVT project finds budget balance at the intersection of cost, quality
Two rather uncomplicated clinical practice changes could save one hospital system about $4 million over 1 year, proof that adopting a uniform best practice can improve the bottom line without sacrificing patient care.
The changes – switching from low-molecular-weight to unfractionated heparin and limiting postsurgical sequential compression devices to short-stay and outpatients only – consolidated practice and lowered expenses in eight hospitals in the Washington, D.C., area. If those same changes could be applied to larger systems, the associated health care savings could be enormous, according to Dr. John. R. Kirkpatrick, a former surgeon in the MedStar Health system and one of the forces behind the project.
"These savings were based on 120,000 surgeries in 1 year," he said in an interview. "There are 30 or 40 million surgeries done in the United States every year. So do the math – the savings could be almost $1 billion every year."
A report published in the April issue of the Journal of the American College of Surgeons describes the project’s genesis, implementation, and results (J. Am. Coll. Surg. 2013;216:800-13). The seed was planted in 2007, when Dr. Kirkpatrick stepped down from his post as the chair of surgery at MedStar Washington Hospital Center. He founded the Surgical Advisory Group, a company he described as a "surgical think tank" offering strategic planning aimed at increasing quality, operational efficiencies, and enhancing patient care.
Value, defined
The new team wanted to examine value in terms of health care – defining it as the intersection of quality and cost. They chose to investigate five perioperative variables: preoperative testing, prophylactic antibiotics, deep venous thrombophlebitis prophylaxis, operating room fluid resuscitation, and invasive monitoring in the OR. For each analysis, they would perform an extensive literature search to describe the currently accepted best clinical practice.
"The reason we chose DVT prophylaxis for the first case was that the data supporting the practices were so clear and compelling that they made our case," Dr. Kirkpatrick said. Studies clearly indicated that unfractionated heparin was just as effective as low-molecular-weight heparin, suitable for all but a select few patient types, and about 10 times cheaper. Low-molecular-weight heparin should be reserved for those cases and for patients who had experienced a DVT while on unfractionated heparin, the group advised.
Data also confirmed that sequential compression devices (SCDs) were most clinically and cost effective when used in the OR, at a price of about $22/stocking. Once the patient returned to the floor, however, the nursing time required to care for the device and patient noncompliance after discharge chipped away at the value without adding to quality. In-hospital daily cost could rise to about $150, and there was no way to even gauge the at-home cost, since many patients simply eventually abandoned the stockings.
Therefore, the group advised, SCDs should be employed only for outpatient or short-stay surgeries.
Assessing each hospital’s current practice was next. A survey sent to top hospital staffers and showed a wide variation in the use of both heparin and SCDs, Dr. Kirkpatrick said. "Four of the hospitals were already following the best practices," but the others were not.
It wasn’t immediately clear why four of the hospitals were almost exclusively using low-molecular-weight heparin and others, unfractionated heparin. Dr. Kirkpatrick said some use seemed related to institutional memory and whether the hospital served as a large teaching facility. Nevertheless, prophylaxis failure rates were similar (1%-1.8%) at all the facilities, confirming that both types of heparin effectively reduced DVT risk.
Stocking up on savings
While the average systemwide cost per dose was around $4, it varied widely depending on the type used: about $2.50 at the four centers using unfractionated heparin and $8.14 at those using low-molecular-weight heparin. The highest cost was $16.60 for low-molecular-weight heparin and the lowest, about $2, for unfractionated. Switching everyone to unfractionated heparin, except patients with failed prophylaxis or those special cases, was projected to save about $3 million/year.
The cost of SCDs was not as clear cut, since it included the difficult-to-measure "hidden costs" of extra nursing time to care for the stockings and loss of nursing time spent on other duties. But all of the facilities employed the devices in all surgical patients, regardless of the postoperative length of stay, at a materials cost of $22. Since 52% of the cases were outpatient or short-stay procedures, limiting the stockings to those patients alone was projected to save about $1 million each year.
The changes have been in place about 3-8 months now, depending on the facility, Dr. Kirkpatrick said. "The thing that changed immediately was the heparin usage. We saw immediately that we were going to get about $1.5 million in savings there. That put us almost to the halfway point of our projected $4 million. The compression devices will take a little longer to measure."
Getting the changes worked into the system wasn’t overly difficult, said Dr. Frederick Finelli, vice president of medical affairs at MedStar Montgomery Medical Center in Olney, Md.
"The physicians were mixed about how they felt about it," he said in an interview. "Some liked one heparin over another, for instance. We had to spend some time on getting a consensus, but we did work it through."
Implementing the medication change wasn’t hard either. The hospital system reorganized its computerized physician order entry system to offer unfractionated heparin as the default choice. "We made it easy to do the right thing and hard to do the wrong thing," Dr. Finelli said. "The algorithm for determining the right dosage is right on the order screen. The heparin is prioritized with unfractionated at the top of the list."
But a physician’s clinical judgment can still override the system, he added. "If you order the low-molecular-weight heparin, the instructions do come up saying that it’s recommended only for patients with cardiac surgery, trauma, or spinal cord injury. So you can still order it. If a physician understands what MedStar wants to do, but disagrees in a particular case, the low molecular weight is available. We think it’s a system that works."
Changes in the use of SCDs went into effect at the beginning of 2013, Dr. Finelli said. Physicians had to work to come to consensus on this point as well – and it was not as easily or thoroughly accomplished.
"There were some surgeons who really liked using them and not using an anticoagulant, so getting buy-in from them was not possible," said Dr. Finelli. This was especially true for those who dealt with cases in which bleeding would cause a great deal of harm, such as spine surgeries.
"These surgeons do continue to use the SCDs vs. heparin. So they do maintain control. We leave some leeway for it."
The next ‘best’ thing
The whole strategy gets a continual tune-up, he added. "We have an established committee of doctors, nurse practitioners, and information technology people who meet every 2 weeks to keep the process up to date."
In fact, flexibility is one of the best things about a value-based systems analysis, Dr. Kirkpatrick said. "We’re always looking for the new ‘best practice.’ What’s a best practice now could be obsolete in 2 years. We will keep updating what we do."
After just 8 months, some of that change may already be upon the program, Dr. Finelli said. In 2010 – 3 years after the Surgical Advisory Group began looking at the issue – low-molecular-weight heparin became available in a generic formula, narrowing the 10-fold cost differential.
"If the price drops low enough, we may want to switch that entire practice around. It’s much more attractive because it’s given once a day, there’s less nursing administration time, and greater patient satisfaction."
For now, though, it’s tough to ignore the total cost savings both practice changes will generate, said Dr. Kirkpatrick. "The reason it’s so significant is that those savings go right to the bottom line. A system like MedStar is very efficient, but the profit margins are still small – 1.5%-2%. There are not many $4 billion/year operations that can survive on a margin like that."
There’s always a cost – even to saving – and that, in the end, determines a project’s worth, said Dr. Stephen Evans, executive vice president of medical affairs and chief medical officer of MedStar Health.
"There are thresholds of cost usefulness. If you have a $40,000 savings, but it’s a laborious process to get there, that’s one thing. But a $4 million savings that’s relatively simple is obvious."
In this case, with the money going directly into the operational budget, the entire system benefits. "This is anew CAT scanner, or 10 new nurses. Part of the incentive for people to get costs under control is so they can look to reinvestment in the business. If everyone understands the goal of reinvestment, it’s a much greater incentive for everyone to participate."
Dr. Kirkpatrick is executive director of the Surgical Advisory Group. Dr. Evans and Dr. Finelli had no financial disclosures, other than their MedStar employment.
*Correction, 4/22/13: The photo caption in an earlier version of this story incorrectly identified Dr. Frederick Finelli.
Two rather uncomplicated clinical practice changes could save one hospital system about $4 million over 1 year, proof that adopting a uniform best practice can improve the bottom line without sacrificing patient care.
The changes – switching from low-molecular-weight to unfractionated heparin and limiting postsurgical sequential compression devices to short-stay and outpatients only – consolidated practice and lowered expenses in eight hospitals in the Washington, D.C., area. If those same changes could be applied to larger systems, the associated health care savings could be enormous, according to Dr. John. R. Kirkpatrick, a former surgeon in the MedStar Health system and one of the forces behind the project.
"These savings were based on 120,000 surgeries in 1 year," he said in an interview. "There are 30 or 40 million surgeries done in the United States every year. So do the math – the savings could be almost $1 billion every year."
A report published in the April issue of the Journal of the American College of Surgeons describes the project’s genesis, implementation, and results (J. Am. Coll. Surg. 2013;216:800-13). The seed was planted in 2007, when Dr. Kirkpatrick stepped down from his post as the chair of surgery at MedStar Washington Hospital Center. He founded the Surgical Advisory Group, a company he described as a "surgical think tank" offering strategic planning aimed at increasing quality, operational efficiencies, and enhancing patient care.
Value, defined
The new team wanted to examine value in terms of health care – defining it as the intersection of quality and cost. They chose to investigate five perioperative variables: preoperative testing, prophylactic antibiotics, deep venous thrombophlebitis prophylaxis, operating room fluid resuscitation, and invasive monitoring in the OR. For each analysis, they would perform an extensive literature search to describe the currently accepted best clinical practice.
"The reason we chose DVT prophylaxis for the first case was that the data supporting the practices were so clear and compelling that they made our case," Dr. Kirkpatrick said. Studies clearly indicated that unfractionated heparin was just as effective as low-molecular-weight heparin, suitable for all but a select few patient types, and about 10 times cheaper. Low-molecular-weight heparin should be reserved for those cases and for patients who had experienced a DVT while on unfractionated heparin, the group advised.
Data also confirmed that sequential compression devices (SCDs) were most clinically and cost effective when used in the OR, at a price of about $22/stocking. Once the patient returned to the floor, however, the nursing time required to care for the device and patient noncompliance after discharge chipped away at the value without adding to quality. In-hospital daily cost could rise to about $150, and there was no way to even gauge the at-home cost, since many patients simply eventually abandoned the stockings.
Therefore, the group advised, SCDs should be employed only for outpatient or short-stay surgeries.
Assessing each hospital’s current practice was next. A survey sent to top hospital staffers and showed a wide variation in the use of both heparin and SCDs, Dr. Kirkpatrick said. "Four of the hospitals were already following the best practices," but the others were not.
It wasn’t immediately clear why four of the hospitals were almost exclusively using low-molecular-weight heparin and others, unfractionated heparin. Dr. Kirkpatrick said some use seemed related to institutional memory and whether the hospital served as a large teaching facility. Nevertheless, prophylaxis failure rates were similar (1%-1.8%) at all the facilities, confirming that both types of heparin effectively reduced DVT risk.
Stocking up on savings
While the average systemwide cost per dose was around $4, it varied widely depending on the type used: about $2.50 at the four centers using unfractionated heparin and $8.14 at those using low-molecular-weight heparin. The highest cost was $16.60 for low-molecular-weight heparin and the lowest, about $2, for unfractionated. Switching everyone to unfractionated heparin, except patients with failed prophylaxis or those special cases, was projected to save about $3 million/year.
The cost of SCDs was not as clear cut, since it included the difficult-to-measure "hidden costs" of extra nursing time to care for the stockings and loss of nursing time spent on other duties. But all of the facilities employed the devices in all surgical patients, regardless of the postoperative length of stay, at a materials cost of $22. Since 52% of the cases were outpatient or short-stay procedures, limiting the stockings to those patients alone was projected to save about $1 million each year.
The changes have been in place about 3-8 months now, depending on the facility, Dr. Kirkpatrick said. "The thing that changed immediately was the heparin usage. We saw immediately that we were going to get about $1.5 million in savings there. That put us almost to the halfway point of our projected $4 million. The compression devices will take a little longer to measure."
Getting the changes worked into the system wasn’t overly difficult, said Dr. Frederick Finelli, vice president of medical affairs at MedStar Montgomery Medical Center in Olney, Md.
"The physicians were mixed about how they felt about it," he said in an interview. "Some liked one heparin over another, for instance. We had to spend some time on getting a consensus, but we did work it through."
Implementing the medication change wasn’t hard either. The hospital system reorganized its computerized physician order entry system to offer unfractionated heparin as the default choice. "We made it easy to do the right thing and hard to do the wrong thing," Dr. Finelli said. "The algorithm for determining the right dosage is right on the order screen. The heparin is prioritized with unfractionated at the top of the list."
But a physician’s clinical judgment can still override the system, he added. "If you order the low-molecular-weight heparin, the instructions do come up saying that it’s recommended only for patients with cardiac surgery, trauma, or spinal cord injury. So you can still order it. If a physician understands what MedStar wants to do, but disagrees in a particular case, the low molecular weight is available. We think it’s a system that works."
Changes in the use of SCDs went into effect at the beginning of 2013, Dr. Finelli said. Physicians had to work to come to consensus on this point as well – and it was not as easily or thoroughly accomplished.
"There were some surgeons who really liked using them and not using an anticoagulant, so getting buy-in from them was not possible," said Dr. Finelli. This was especially true for those who dealt with cases in which bleeding would cause a great deal of harm, such as spine surgeries.
"These surgeons do continue to use the SCDs vs. heparin. So they do maintain control. We leave some leeway for it."
The next ‘best’ thing
The whole strategy gets a continual tune-up, he added. "We have an established committee of doctors, nurse practitioners, and information technology people who meet every 2 weeks to keep the process up to date."
In fact, flexibility is one of the best things about a value-based systems analysis, Dr. Kirkpatrick said. "We’re always looking for the new ‘best practice.’ What’s a best practice now could be obsolete in 2 years. We will keep updating what we do."
After just 8 months, some of that change may already be upon the program, Dr. Finelli said. In 2010 – 3 years after the Surgical Advisory Group began looking at the issue – low-molecular-weight heparin became available in a generic formula, narrowing the 10-fold cost differential.
"If the price drops low enough, we may want to switch that entire practice around. It’s much more attractive because it’s given once a day, there’s less nursing administration time, and greater patient satisfaction."
For now, though, it’s tough to ignore the total cost savings both practice changes will generate, said Dr. Kirkpatrick. "The reason it’s so significant is that those savings go right to the bottom line. A system like MedStar is very efficient, but the profit margins are still small – 1.5%-2%. There are not many $4 billion/year operations that can survive on a margin like that."
There’s always a cost – even to saving – and that, in the end, determines a project’s worth, said Dr. Stephen Evans, executive vice president of medical affairs and chief medical officer of MedStar Health.
"There are thresholds of cost usefulness. If you have a $40,000 savings, but it’s a laborious process to get there, that’s one thing. But a $4 million savings that’s relatively simple is obvious."
In this case, with the money going directly into the operational budget, the entire system benefits. "This is anew CAT scanner, or 10 new nurses. Part of the incentive for people to get costs under control is so they can look to reinvestment in the business. If everyone understands the goal of reinvestment, it’s a much greater incentive for everyone to participate."
Dr. Kirkpatrick is executive director of the Surgical Advisory Group. Dr. Evans and Dr. Finelli had no financial disclosures, other than their MedStar employment.
*Correction, 4/22/13: The photo caption in an earlier version of this story incorrectly identified Dr. Frederick Finelli.
Two rather uncomplicated clinical practice changes could save one hospital system about $4 million over 1 year, proof that adopting a uniform best practice can improve the bottom line without sacrificing patient care.
The changes – switching from low-molecular-weight to unfractionated heparin and limiting postsurgical sequential compression devices to short-stay and outpatients only – consolidated practice and lowered expenses in eight hospitals in the Washington, D.C., area. If those same changes could be applied to larger systems, the associated health care savings could be enormous, according to Dr. John. R. Kirkpatrick, a former surgeon in the MedStar Health system and one of the forces behind the project.
"These savings were based on 120,000 surgeries in 1 year," he said in an interview. "There are 30 or 40 million surgeries done in the United States every year. So do the math – the savings could be almost $1 billion every year."
A report published in the April issue of the Journal of the American College of Surgeons describes the project’s genesis, implementation, and results (J. Am. Coll. Surg. 2013;216:800-13). The seed was planted in 2007, when Dr. Kirkpatrick stepped down from his post as the chair of surgery at MedStar Washington Hospital Center. He founded the Surgical Advisory Group, a company he described as a "surgical think tank" offering strategic planning aimed at increasing quality, operational efficiencies, and enhancing patient care.
Value, defined
The new team wanted to examine value in terms of health care – defining it as the intersection of quality and cost. They chose to investigate five perioperative variables: preoperative testing, prophylactic antibiotics, deep venous thrombophlebitis prophylaxis, operating room fluid resuscitation, and invasive monitoring in the OR. For each analysis, they would perform an extensive literature search to describe the currently accepted best clinical practice.
"The reason we chose DVT prophylaxis for the first case was that the data supporting the practices were so clear and compelling that they made our case," Dr. Kirkpatrick said. Studies clearly indicated that unfractionated heparin was just as effective as low-molecular-weight heparin, suitable for all but a select few patient types, and about 10 times cheaper. Low-molecular-weight heparin should be reserved for those cases and for patients who had experienced a DVT while on unfractionated heparin, the group advised.
Data also confirmed that sequential compression devices (SCDs) were most clinically and cost effective when used in the OR, at a price of about $22/stocking. Once the patient returned to the floor, however, the nursing time required to care for the device and patient noncompliance after discharge chipped away at the value without adding to quality. In-hospital daily cost could rise to about $150, and there was no way to even gauge the at-home cost, since many patients simply eventually abandoned the stockings.
Therefore, the group advised, SCDs should be employed only for outpatient or short-stay surgeries.
Assessing each hospital’s current practice was next. A survey sent to top hospital staffers and showed a wide variation in the use of both heparin and SCDs, Dr. Kirkpatrick said. "Four of the hospitals were already following the best practices," but the others were not.
It wasn’t immediately clear why four of the hospitals were almost exclusively using low-molecular-weight heparin and others, unfractionated heparin. Dr. Kirkpatrick said some use seemed related to institutional memory and whether the hospital served as a large teaching facility. Nevertheless, prophylaxis failure rates were similar (1%-1.8%) at all the facilities, confirming that both types of heparin effectively reduced DVT risk.
Stocking up on savings
While the average systemwide cost per dose was around $4, it varied widely depending on the type used: about $2.50 at the four centers using unfractionated heparin and $8.14 at those using low-molecular-weight heparin. The highest cost was $16.60 for low-molecular-weight heparin and the lowest, about $2, for unfractionated. Switching everyone to unfractionated heparin, except patients with failed prophylaxis or those special cases, was projected to save about $3 million/year.
The cost of SCDs was not as clear cut, since it included the difficult-to-measure "hidden costs" of extra nursing time to care for the stockings and loss of nursing time spent on other duties. But all of the facilities employed the devices in all surgical patients, regardless of the postoperative length of stay, at a materials cost of $22. Since 52% of the cases were outpatient or short-stay procedures, limiting the stockings to those patients alone was projected to save about $1 million each year.
The changes have been in place about 3-8 months now, depending on the facility, Dr. Kirkpatrick said. "The thing that changed immediately was the heparin usage. We saw immediately that we were going to get about $1.5 million in savings there. That put us almost to the halfway point of our projected $4 million. The compression devices will take a little longer to measure."
Getting the changes worked into the system wasn’t overly difficult, said Dr. Frederick Finelli, vice president of medical affairs at MedStar Montgomery Medical Center in Olney, Md.
"The physicians were mixed about how they felt about it," he said in an interview. "Some liked one heparin over another, for instance. We had to spend some time on getting a consensus, but we did work it through."
Implementing the medication change wasn’t hard either. The hospital system reorganized its computerized physician order entry system to offer unfractionated heparin as the default choice. "We made it easy to do the right thing and hard to do the wrong thing," Dr. Finelli said. "The algorithm for determining the right dosage is right on the order screen. The heparin is prioritized with unfractionated at the top of the list."
But a physician’s clinical judgment can still override the system, he added. "If you order the low-molecular-weight heparin, the instructions do come up saying that it’s recommended only for patients with cardiac surgery, trauma, or spinal cord injury. So you can still order it. If a physician understands what MedStar wants to do, but disagrees in a particular case, the low molecular weight is available. We think it’s a system that works."
Changes in the use of SCDs went into effect at the beginning of 2013, Dr. Finelli said. Physicians had to work to come to consensus on this point as well – and it was not as easily or thoroughly accomplished.
"There were some surgeons who really liked using them and not using an anticoagulant, so getting buy-in from them was not possible," said Dr. Finelli. This was especially true for those who dealt with cases in which bleeding would cause a great deal of harm, such as spine surgeries.
"These surgeons do continue to use the SCDs vs. heparin. So they do maintain control. We leave some leeway for it."
The next ‘best’ thing
The whole strategy gets a continual tune-up, he added. "We have an established committee of doctors, nurse practitioners, and information technology people who meet every 2 weeks to keep the process up to date."
In fact, flexibility is one of the best things about a value-based systems analysis, Dr. Kirkpatrick said. "We’re always looking for the new ‘best practice.’ What’s a best practice now could be obsolete in 2 years. We will keep updating what we do."
After just 8 months, some of that change may already be upon the program, Dr. Finelli said. In 2010 – 3 years after the Surgical Advisory Group began looking at the issue – low-molecular-weight heparin became available in a generic formula, narrowing the 10-fold cost differential.
"If the price drops low enough, we may want to switch that entire practice around. It’s much more attractive because it’s given once a day, there’s less nursing administration time, and greater patient satisfaction."
For now, though, it’s tough to ignore the total cost savings both practice changes will generate, said Dr. Kirkpatrick. "The reason it’s so significant is that those savings go right to the bottom line. A system like MedStar is very efficient, but the profit margins are still small – 1.5%-2%. There are not many $4 billion/year operations that can survive on a margin like that."
There’s always a cost – even to saving – and that, in the end, determines a project’s worth, said Dr. Stephen Evans, executive vice president of medical affairs and chief medical officer of MedStar Health.
"There are thresholds of cost usefulness. If you have a $40,000 savings, but it’s a laborious process to get there, that’s one thing. But a $4 million savings that’s relatively simple is obvious."
In this case, with the money going directly into the operational budget, the entire system benefits. "This is anew CAT scanner, or 10 new nurses. Part of the incentive for people to get costs under control is so they can look to reinvestment in the business. If everyone understands the goal of reinvestment, it’s a much greater incentive for everyone to participate."
Dr. Kirkpatrick is executive director of the Surgical Advisory Group. Dr. Evans and Dr. Finelli had no financial disclosures, other than their MedStar employment.
*Correction, 4/22/13: The photo caption in an earlier version of this story incorrectly identified Dr. Frederick Finelli.
States differ in screening for congenital heart defects
Routine newborn screening for congenital heart defects in New Jersey hospitals was an easy-to-implement procedure that didn’t substantially impact the cost of either labor or materials.
Yet in Georgia, hospitals that hadn’t implemented this nationally recommended protocol cited concerns about staffing and equipment as barriers to carrying out such screening, according to two reports published in the April 19 issue of the Morbidity and Mortality Weekly Report (2013;62:288-91, 292-4).
In 2011, the U.S. Secretary of Health and Human Services recommended that the Recommended Uniform Screening Panel (RUSP) for newborns include screening for critical congenital heart defects, which is generally accomplished by pulse oximetry. However, it’s up to each state to decide whether to make screening mandatory, and each hospital to decide how to implement it.
The new reports documented the results of surveys conducted by the Centers for Disease Control and Prevention, the Georgia Department of Public Health, and the New Jersey Department of Health. Surveys were sent to a sampling of birthing hospitals in each state. Georgia does not mandate screening; New Jersey does.
The Georgia survey asked the facilities to document whether or not screening was performed; how those screens were implemented, recorded, and followed up; and any barriers to screening.
Also, 11 pulse oximetry screenings were observed at five hospitals in Georgia. They found that screening took little time – an average of 10 minutes – and was easily conducted.
In that state, 71 facilities responded to the survey request. Of these, 22 (31%) were currently screening, 20 (28%) planned to start screening in 2012; 14 (20%) planned to start screening at some other time; and 15 (21%) had no plans to start.
The investigators, led by Pamela Clark of the Oak Ridge Institute for Science and Education, Tenn., found a number of barriers that hospitals said affected their decision to screen. Some (35%) said they did not have on-site echocardiography. Almost half (45%) said they had to transfer patients for screening, with a median driving distance of 54 miles.
Of the 29 hospitals that were not screening, 13 said they had no clear plan of how to follow up on positive results and 13 said they were unsure of how to report them.
Cost was an issue for everyone, the survey found. Seven of the 22 (32%) screening hospitals expressed concern about reimbursement, as did 8 of 20 (40%) of those who planned to start in 2012, 4 of 14 (29%) of those that were going to start at some other time, and 2 of 15 (13%) of those who had no plans to do it.
Equipment was a barrier for all of the nonscreening facilities: 35% of those who were going to start screening in 2012 said they needed to buy equipment, as did 50% of those who said they’d start some other time, and 33% of those who had no plans to start.
However, almost all of the hospitals said they already did record pulse oximetry or planned to start doing it, including 80% of the hospitals that weren’t going to screen, all of which recorded pulse oximetry in the electronic medical record.
All that did or would start screening have access to a pediatric cardiologist. Only one hospital that had no plans to screen and one that might start at some point said they had no access to such a specialist.
The lack of a state mandate also had an impact – almost half of the hospitals with no plans to start screening (47%) said that was a factor in their decision.
There were some concerns, however, even among the hospitals already screening: 12 didn’t know how often to send screening data to the Department of Public Health and 11 didn’t know what kind of data to send.
The picture in New Jersey, the first state to mandate screening, was quite different. Five months after the requirement went into effect on Aug. 31, 2011, a similar survey was carried out among a sample of 11 birthing hospitals. All of these were screening according to the recommendations using pulse oximetry.
Three hospitals submitted the data electronically, five manually, and three with both methods. Staff saw the handling and submission of records as a "moderate burden." Nurses said pulse oximetry was easily incorporated into their duties and was not burdensome.
During the first 3 months of screening, 98% of 25,214 newborns received the test. Twelve babies tested positive for a congenital heart defect, reported Dr. Lorraine F. Garg of the New Jersey Department of Health and her associates.
In addition to identifying heart problems, pulse oximetry screening could have other effects. "Pulse oximetry screening also might identify other medical conditions, such as pulmonary conditions or sepsis, potentially improving newborn care and subsequent outcomes," according to an editorial note.
Routine newborn screening for congenital heart defects in New Jersey hospitals was an easy-to-implement procedure that didn’t substantially impact the cost of either labor or materials.
Yet in Georgia, hospitals that hadn’t implemented this nationally recommended protocol cited concerns about staffing and equipment as barriers to carrying out such screening, according to two reports published in the April 19 issue of the Morbidity and Mortality Weekly Report (2013;62:288-91, 292-4).
In 2011, the U.S. Secretary of Health and Human Services recommended that the Recommended Uniform Screening Panel (RUSP) for newborns include screening for critical congenital heart defects, which is generally accomplished by pulse oximetry. However, it’s up to each state to decide whether to make screening mandatory, and each hospital to decide how to implement it.
The new reports documented the results of surveys conducted by the Centers for Disease Control and Prevention, the Georgia Department of Public Health, and the New Jersey Department of Health. Surveys were sent to a sampling of birthing hospitals in each state. Georgia does not mandate screening; New Jersey does.
The Georgia survey asked the facilities to document whether or not screening was performed; how those screens were implemented, recorded, and followed up; and any barriers to screening.
Also, 11 pulse oximetry screenings were observed at five hospitals in Georgia. They found that screening took little time – an average of 10 minutes – and was easily conducted.
In that state, 71 facilities responded to the survey request. Of these, 22 (31%) were currently screening, 20 (28%) planned to start screening in 2012; 14 (20%) planned to start screening at some other time; and 15 (21%) had no plans to start.
The investigators, led by Pamela Clark of the Oak Ridge Institute for Science and Education, Tenn., found a number of barriers that hospitals said affected their decision to screen. Some (35%) said they did not have on-site echocardiography. Almost half (45%) said they had to transfer patients for screening, with a median driving distance of 54 miles.
Of the 29 hospitals that were not screening, 13 said they had no clear plan of how to follow up on positive results and 13 said they were unsure of how to report them.
Cost was an issue for everyone, the survey found. Seven of the 22 (32%) screening hospitals expressed concern about reimbursement, as did 8 of 20 (40%) of those who planned to start in 2012, 4 of 14 (29%) of those that were going to start at some other time, and 2 of 15 (13%) of those who had no plans to do it.
Equipment was a barrier for all of the nonscreening facilities: 35% of those who were going to start screening in 2012 said they needed to buy equipment, as did 50% of those who said they’d start some other time, and 33% of those who had no plans to start.
However, almost all of the hospitals said they already did record pulse oximetry or planned to start doing it, including 80% of the hospitals that weren’t going to screen, all of which recorded pulse oximetry in the electronic medical record.
All that did or would start screening have access to a pediatric cardiologist. Only one hospital that had no plans to screen and one that might start at some point said they had no access to such a specialist.
The lack of a state mandate also had an impact – almost half of the hospitals with no plans to start screening (47%) said that was a factor in their decision.
There were some concerns, however, even among the hospitals already screening: 12 didn’t know how often to send screening data to the Department of Public Health and 11 didn’t know what kind of data to send.
The picture in New Jersey, the first state to mandate screening, was quite different. Five months after the requirement went into effect on Aug. 31, 2011, a similar survey was carried out among a sample of 11 birthing hospitals. All of these were screening according to the recommendations using pulse oximetry.
Three hospitals submitted the data electronically, five manually, and three with both methods. Staff saw the handling and submission of records as a "moderate burden." Nurses said pulse oximetry was easily incorporated into their duties and was not burdensome.
During the first 3 months of screening, 98% of 25,214 newborns received the test. Twelve babies tested positive for a congenital heart defect, reported Dr. Lorraine F. Garg of the New Jersey Department of Health and her associates.
In addition to identifying heart problems, pulse oximetry screening could have other effects. "Pulse oximetry screening also might identify other medical conditions, such as pulmonary conditions or sepsis, potentially improving newborn care and subsequent outcomes," according to an editorial note.
Routine newborn screening for congenital heart defects in New Jersey hospitals was an easy-to-implement procedure that didn’t substantially impact the cost of either labor or materials.
Yet in Georgia, hospitals that hadn’t implemented this nationally recommended protocol cited concerns about staffing and equipment as barriers to carrying out such screening, according to two reports published in the April 19 issue of the Morbidity and Mortality Weekly Report (2013;62:288-91, 292-4).
In 2011, the U.S. Secretary of Health and Human Services recommended that the Recommended Uniform Screening Panel (RUSP) for newborns include screening for critical congenital heart defects, which is generally accomplished by pulse oximetry. However, it’s up to each state to decide whether to make screening mandatory, and each hospital to decide how to implement it.
The new reports documented the results of surveys conducted by the Centers for Disease Control and Prevention, the Georgia Department of Public Health, and the New Jersey Department of Health. Surveys were sent to a sampling of birthing hospitals in each state. Georgia does not mandate screening; New Jersey does.
The Georgia survey asked the facilities to document whether or not screening was performed; how those screens were implemented, recorded, and followed up; and any barriers to screening.
Also, 11 pulse oximetry screenings were observed at five hospitals in Georgia. They found that screening took little time – an average of 10 minutes – and was easily conducted.
In that state, 71 facilities responded to the survey request. Of these, 22 (31%) were currently screening, 20 (28%) planned to start screening in 2012; 14 (20%) planned to start screening at some other time; and 15 (21%) had no plans to start.
The investigators, led by Pamela Clark of the Oak Ridge Institute for Science and Education, Tenn., found a number of barriers that hospitals said affected their decision to screen. Some (35%) said they did not have on-site echocardiography. Almost half (45%) said they had to transfer patients for screening, with a median driving distance of 54 miles.
Of the 29 hospitals that were not screening, 13 said they had no clear plan of how to follow up on positive results and 13 said they were unsure of how to report them.
Cost was an issue for everyone, the survey found. Seven of the 22 (32%) screening hospitals expressed concern about reimbursement, as did 8 of 20 (40%) of those who planned to start in 2012, 4 of 14 (29%) of those that were going to start at some other time, and 2 of 15 (13%) of those who had no plans to do it.
Equipment was a barrier for all of the nonscreening facilities: 35% of those who were going to start screening in 2012 said they needed to buy equipment, as did 50% of those who said they’d start some other time, and 33% of those who had no plans to start.
However, almost all of the hospitals said they already did record pulse oximetry or planned to start doing it, including 80% of the hospitals that weren’t going to screen, all of which recorded pulse oximetry in the electronic medical record.
All that did or would start screening have access to a pediatric cardiologist. Only one hospital that had no plans to screen and one that might start at some point said they had no access to such a specialist.
The lack of a state mandate also had an impact – almost half of the hospitals with no plans to start screening (47%) said that was a factor in their decision.
There were some concerns, however, even among the hospitals already screening: 12 didn’t know how often to send screening data to the Department of Public Health and 11 didn’t know what kind of data to send.
The picture in New Jersey, the first state to mandate screening, was quite different. Five months after the requirement went into effect on Aug. 31, 2011, a similar survey was carried out among a sample of 11 birthing hospitals. All of these were screening according to the recommendations using pulse oximetry.
Three hospitals submitted the data electronically, five manually, and three with both methods. Staff saw the handling and submission of records as a "moderate burden." Nurses said pulse oximetry was easily incorporated into their duties and was not burdensome.
During the first 3 months of screening, 98% of 25,214 newborns received the test. Twelve babies tested positive for a congenital heart defect, reported Dr. Lorraine F. Garg of the New Jersey Department of Health and her associates.
In addition to identifying heart problems, pulse oximetry screening could have other effects. "Pulse oximetry screening also might identify other medical conditions, such as pulmonary conditions or sepsis, potentially improving newborn care and subsequent outcomes," according to an editorial note.
FROM MMWR
Major finding: Of 71 Georgia hospitals surveyed, 22 (31%) were screening newborns for congenital heart defects, compared with 100% of the 8 hospitals surveyed in New Jersey.
Data source: Reports of surveys in Georgia, which does not mandate screening, and in New Jersey, which does.
Disclosures: As employees of the public health system, the authors of each study had no financial disclosures.
Prenatal exposure to air pollution boosts childhood cancer risk
WASHINGTON – Children whose mothers were exposed to air pollution during pregnancy may have a small, but significant, increase in pediatric cancer risk.
A 9-year case-control study found that children who were exposed to the highest levels of traffic-related pollution prenatally and during the first year of life were 19% more likely to develop bilateral retinoblastoma and 17% more likely to develop germ cell tumors than were children who were exposed to the lowest level.
"This is the first study to report upon traffic pollution in relation to retinoblastoma or germ cell tumors, and since both of these are rare, the findings need to be replicated in other studies," Julia E. Heck, Ph.D., said in an interview at the annual meeting of the American Association for Cancer Research.
Dr. Heck, an epidemiologist at the University of California’s Fielding School of Public Health, Los Angeles, used a state air pollution modeling system to detect the patterns in her study. The California Line Source Dispersion Modeling system, version 4 (CALINE4) uses a test fleet of motor vehicles to predict traffic-related carbon monoxide emissions from vehicles in idle, cruise, acceleration, and deceleration.
Her study cohort comprised 3,590 children with cancer who were included in a California cancer registry. All were born between 1998 and 2007 and were aged younger than 6 years. She compared these children to a random selection of 80,224 children drawn form California birth records. The CALINE4 system generated estimates of local traffic exposure based on where the mothers were living during each trimester of pregnancy.
The model imputed emissions from gasoline and diesel vehicles within a 1,500-meter radius of the address. It included data on traffic volume, roadway geometry, vehicle emission rates, and weather patterns, and divided the cohort into quartiles according to exposure. Dr. Heck looked specifically at carbon monoxide measurements in increments of 53 parts per billion.
Each increase in CO emission raised the risk of acute lymphoblastic leukemia by 4%. Each quartile was associated with an overall 14% increased risk of a retinoblastoma, with the risk significantly elevated for bilateral tumors rather than unilateral tumors (a 19% increase). Dr. Heck also saw a 17% increased risk for germ cell tumors for every quartile increase in CO.
Associations with other cancers, including acute myeloid leukemia, non-Hodgkin’s lymphoma, ependymoma, astrocytoma, neuroblastoma, Wilms tumor, hepatoblastoma, and rhabdomyosarcoma, were nonsignificant.
Because Dr. Heck’s study was a retrospective analysis, it cannot prove causation. "The results need to be confirmed in other studies," she said in an interview. "But our findings do support a link between traffic pollution and some childhood cancers."
The field of childhood cancers and air pollution has not been well studied in recent years, Dr. Heck said, although there were a number of such studies in the late 1990s and early 2000s.
One was a British case-control analysis of birth and death records of 12,018 children in the United Kingdom who had been born and died from leukemia and other cancers from 1955 to 1980. The cancer-related deaths were linked to locations of rail and bus stations, ferry terminals, railways, roads, canals, and rivers (J. Epidemiol. Community Health 2005;59:755-60).
"The most striking result is the extraordinary concentration of cancer births within 0.3 km of bus/coach stations (OR = 12.5)," wrote Dr. E.G. Knox, professor emeritus at the University of Birmingham, England. "This was followed by hospitals (OR = 2.6) and heavy transport centers (OR = 1.6)."
Of the individual pollutants examined, both carbon monoxide and 1,3-butadiene more than doubled the risk of cancer.
Because the study encompassed the child’s location at birth, it demonstrated a link to prenatal exposure. "Childhood cancers are strongly determined by prenatal or early postnatal exposures to oil-based combustion gases, especially from engine exhausts," he noted. "The chief carcinogenic agent is probably 1,3-butadiene."
The National Institute for Occupational Safety and Health has recommended that 1,3-butadiene be treated as a potential occupational carcinogen, teratogen, and as a reproduction hazard.
In 2006, Dr. Knox published another study on the same group, in which he attributed about 24% of the cancers to prenatal or early life proximity to areas of high airborne pollutants. "Child cancer initiations are strongly determined by prenatal or early postnatal exposures to engine exhaust gases, probably through maternal inhalation and accumulation of carcinogens over many months," he noted. (J. Epidemiol. Community Health 2006;60:136-41).
Dr. Heck’s study was funded by the National Institutes of Health. She had no financial disclosures.
WASHINGTON – Children whose mothers were exposed to air pollution during pregnancy may have a small, but significant, increase in pediatric cancer risk.
A 9-year case-control study found that children who were exposed to the highest levels of traffic-related pollution prenatally and during the first year of life were 19% more likely to develop bilateral retinoblastoma and 17% more likely to develop germ cell tumors than were children who were exposed to the lowest level.
"This is the first study to report upon traffic pollution in relation to retinoblastoma or germ cell tumors, and since both of these are rare, the findings need to be replicated in other studies," Julia E. Heck, Ph.D., said in an interview at the annual meeting of the American Association for Cancer Research.
Dr. Heck, an epidemiologist at the University of California’s Fielding School of Public Health, Los Angeles, used a state air pollution modeling system to detect the patterns in her study. The California Line Source Dispersion Modeling system, version 4 (CALINE4) uses a test fleet of motor vehicles to predict traffic-related carbon monoxide emissions from vehicles in idle, cruise, acceleration, and deceleration.
Her study cohort comprised 3,590 children with cancer who were included in a California cancer registry. All were born between 1998 and 2007 and were aged younger than 6 years. She compared these children to a random selection of 80,224 children drawn form California birth records. The CALINE4 system generated estimates of local traffic exposure based on where the mothers were living during each trimester of pregnancy.
The model imputed emissions from gasoline and diesel vehicles within a 1,500-meter radius of the address. It included data on traffic volume, roadway geometry, vehicle emission rates, and weather patterns, and divided the cohort into quartiles according to exposure. Dr. Heck looked specifically at carbon monoxide measurements in increments of 53 parts per billion.
Each increase in CO emission raised the risk of acute lymphoblastic leukemia by 4%. Each quartile was associated with an overall 14% increased risk of a retinoblastoma, with the risk significantly elevated for bilateral tumors rather than unilateral tumors (a 19% increase). Dr. Heck also saw a 17% increased risk for germ cell tumors for every quartile increase in CO.
Associations with other cancers, including acute myeloid leukemia, non-Hodgkin’s lymphoma, ependymoma, astrocytoma, neuroblastoma, Wilms tumor, hepatoblastoma, and rhabdomyosarcoma, were nonsignificant.
Because Dr. Heck’s study was a retrospective analysis, it cannot prove causation. "The results need to be confirmed in other studies," she said in an interview. "But our findings do support a link between traffic pollution and some childhood cancers."
The field of childhood cancers and air pollution has not been well studied in recent years, Dr. Heck said, although there were a number of such studies in the late 1990s and early 2000s.
One was a British case-control analysis of birth and death records of 12,018 children in the United Kingdom who had been born and died from leukemia and other cancers from 1955 to 1980. The cancer-related deaths were linked to locations of rail and bus stations, ferry terminals, railways, roads, canals, and rivers (J. Epidemiol. Community Health 2005;59:755-60).
"The most striking result is the extraordinary concentration of cancer births within 0.3 km of bus/coach stations (OR = 12.5)," wrote Dr. E.G. Knox, professor emeritus at the University of Birmingham, England. "This was followed by hospitals (OR = 2.6) and heavy transport centers (OR = 1.6)."
Of the individual pollutants examined, both carbon monoxide and 1,3-butadiene more than doubled the risk of cancer.
Because the study encompassed the child’s location at birth, it demonstrated a link to prenatal exposure. "Childhood cancers are strongly determined by prenatal or early postnatal exposures to oil-based combustion gases, especially from engine exhausts," he noted. "The chief carcinogenic agent is probably 1,3-butadiene."
The National Institute for Occupational Safety and Health has recommended that 1,3-butadiene be treated as a potential occupational carcinogen, teratogen, and as a reproduction hazard.
In 2006, Dr. Knox published another study on the same group, in which he attributed about 24% of the cancers to prenatal or early life proximity to areas of high airborne pollutants. "Child cancer initiations are strongly determined by prenatal or early postnatal exposures to engine exhaust gases, probably through maternal inhalation and accumulation of carcinogens over many months," he noted. (J. Epidemiol. Community Health 2006;60:136-41).
Dr. Heck’s study was funded by the National Institutes of Health. She had no financial disclosures.
WASHINGTON – Children whose mothers were exposed to air pollution during pregnancy may have a small, but significant, increase in pediatric cancer risk.
A 9-year case-control study found that children who were exposed to the highest levels of traffic-related pollution prenatally and during the first year of life were 19% more likely to develop bilateral retinoblastoma and 17% more likely to develop germ cell tumors than were children who were exposed to the lowest level.
"This is the first study to report upon traffic pollution in relation to retinoblastoma or germ cell tumors, and since both of these are rare, the findings need to be replicated in other studies," Julia E. Heck, Ph.D., said in an interview at the annual meeting of the American Association for Cancer Research.
Dr. Heck, an epidemiologist at the University of California’s Fielding School of Public Health, Los Angeles, used a state air pollution modeling system to detect the patterns in her study. The California Line Source Dispersion Modeling system, version 4 (CALINE4) uses a test fleet of motor vehicles to predict traffic-related carbon monoxide emissions from vehicles in idle, cruise, acceleration, and deceleration.
Her study cohort comprised 3,590 children with cancer who were included in a California cancer registry. All were born between 1998 and 2007 and were aged younger than 6 years. She compared these children to a random selection of 80,224 children drawn form California birth records. The CALINE4 system generated estimates of local traffic exposure based on where the mothers were living during each trimester of pregnancy.
The model imputed emissions from gasoline and diesel vehicles within a 1,500-meter radius of the address. It included data on traffic volume, roadway geometry, vehicle emission rates, and weather patterns, and divided the cohort into quartiles according to exposure. Dr. Heck looked specifically at carbon monoxide measurements in increments of 53 parts per billion.
Each increase in CO emission raised the risk of acute lymphoblastic leukemia by 4%. Each quartile was associated with an overall 14% increased risk of a retinoblastoma, with the risk significantly elevated for bilateral tumors rather than unilateral tumors (a 19% increase). Dr. Heck also saw a 17% increased risk for germ cell tumors for every quartile increase in CO.
Associations with other cancers, including acute myeloid leukemia, non-Hodgkin’s lymphoma, ependymoma, astrocytoma, neuroblastoma, Wilms tumor, hepatoblastoma, and rhabdomyosarcoma, were nonsignificant.
Because Dr. Heck’s study was a retrospective analysis, it cannot prove causation. "The results need to be confirmed in other studies," she said in an interview. "But our findings do support a link between traffic pollution and some childhood cancers."
The field of childhood cancers and air pollution has not been well studied in recent years, Dr. Heck said, although there were a number of such studies in the late 1990s and early 2000s.
One was a British case-control analysis of birth and death records of 12,018 children in the United Kingdom who had been born and died from leukemia and other cancers from 1955 to 1980. The cancer-related deaths were linked to locations of rail and bus stations, ferry terminals, railways, roads, canals, and rivers (J. Epidemiol. Community Health 2005;59:755-60).
"The most striking result is the extraordinary concentration of cancer births within 0.3 km of bus/coach stations (OR = 12.5)," wrote Dr. E.G. Knox, professor emeritus at the University of Birmingham, England. "This was followed by hospitals (OR = 2.6) and heavy transport centers (OR = 1.6)."
Of the individual pollutants examined, both carbon monoxide and 1,3-butadiene more than doubled the risk of cancer.
Because the study encompassed the child’s location at birth, it demonstrated a link to prenatal exposure. "Childhood cancers are strongly determined by prenatal or early postnatal exposures to oil-based combustion gases, especially from engine exhausts," he noted. "The chief carcinogenic agent is probably 1,3-butadiene."
The National Institute for Occupational Safety and Health has recommended that 1,3-butadiene be treated as a potential occupational carcinogen, teratogen, and as a reproduction hazard.
In 2006, Dr. Knox published another study on the same group, in which he attributed about 24% of the cancers to prenatal or early life proximity to areas of high airborne pollutants. "Child cancer initiations are strongly determined by prenatal or early postnatal exposures to engine exhaust gases, probably through maternal inhalation and accumulation of carcinogens over many months," he noted. (J. Epidemiol. Community Health 2006;60:136-41).
Dr. Heck’s study was funded by the National Institutes of Health. She had no financial disclosures.
FROM THE AACR ANNUAL MEETING
Major finding: Children exposed prenatally to high levels of motor vehicle exhaust were 19% more likely to develop bilateral retinoblastoma and 17% more likely to develop germ cell tumors than were children with less exposure.
Data source: Case-control analysis of 3,590 children who developed cancer matched to 80,224 controls.
Disclosures: The study was funded by the National Institutes of Health. Dr. Heck had no financial disclosures.
Androgen deprivation decreases lung tumors in men with prostate cancer
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
WASHINGTON – Men who received androgen blockade therapy as part of prostate cancer treatment were less likely to develop a secondary lung cancer than were those who didn’t receive the hormone regimen.
The significant finding suggests that estrogen may fuel non–small cell lung cancers in both men and women, Dr. Hyunseok Kang reported at the annual meeting of the American Association for Cancer Research.
"Recent literature suggests estrogen receptors and aromatase play a role in the development of non–small cell lung cancers in women with breast cancer," Dr. Kang said. "Aromatase can convert testosterone to estrogen – which could increase the same risk in men."
This recently observed association prompted Dr. Kang, a fellow at Emory University, Atlanta, and his colleagues to investigate whether a similar hormone-mediated link might exist for men with prostate cancer – and whether blocking it could also block lung cancer.
The retrospective observational study pulled data from the Veterans Affairs Central Cancer Registry. It comprised 62,589 men who were diagnosed with prostate cancer between 1999 and 2008, with follow-up through 2010.
There were some significant – and important – baseline differences between the 20,378 men who underwent androgen blockade and those who did not. Men who received the treatment were significantly older (mean age, 71 years vs. 66 years) and slightly more likely to have ever smoked tobacco (73% vs. 71%).
Prostate cancer stage at diagnosis was also significantly different, with fewer of the hormone therapy group presenting at stage II (77% vs. 92%) and more presenting at stage IV (17% vs. 2%).
Men from the VA database were significantly more likely than the general population to develop lung cancer, whether they underwent androgen blockade therapy or not (1.8/100,000 men who received treatment and 2.4/100,000 men who did not receive treatment vs. 0.80/100,000 in the general population).
Of men in the VA database who received androgen treatment, 258 developed secondary lung cancers, compared with 508 men who did not receive treatment.
In an unadjusted model, men who had treatment were 18% less likely to develop a secondary lung cancer (hazard ratio, 0.82). That finding held in a multivariate analysis that adjusted for smoking and age (HR, 0.83).
The study was sponsored by the Veterans Health Administration. Dr. Kang had no financial disclosures.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
WASHINGTON – Men who received androgen blockade therapy as part of prostate cancer treatment were less likely to develop a secondary lung cancer than were those who didn’t receive the hormone regimen.
The significant finding suggests that estrogen may fuel non–small cell lung cancers in both men and women, Dr. Hyunseok Kang reported at the annual meeting of the American Association for Cancer Research.
"Recent literature suggests estrogen receptors and aromatase play a role in the development of non–small cell lung cancers in women with breast cancer," Dr. Kang said. "Aromatase can convert testosterone to estrogen – which could increase the same risk in men."
This recently observed association prompted Dr. Kang, a fellow at Emory University, Atlanta, and his colleagues to investigate whether a similar hormone-mediated link might exist for men with prostate cancer – and whether blocking it could also block lung cancer.
The retrospective observational study pulled data from the Veterans Affairs Central Cancer Registry. It comprised 62,589 men who were diagnosed with prostate cancer between 1999 and 2008, with follow-up through 2010.
There were some significant – and important – baseline differences between the 20,378 men who underwent androgen blockade and those who did not. Men who received the treatment were significantly older (mean age, 71 years vs. 66 years) and slightly more likely to have ever smoked tobacco (73% vs. 71%).
Prostate cancer stage at diagnosis was also significantly different, with fewer of the hormone therapy group presenting at stage II (77% vs. 92%) and more presenting at stage IV (17% vs. 2%).
Men from the VA database were significantly more likely than the general population to develop lung cancer, whether they underwent androgen blockade therapy or not (1.8/100,000 men who received treatment and 2.4/100,000 men who did not receive treatment vs. 0.80/100,000 in the general population).
Of men in the VA database who received androgen treatment, 258 developed secondary lung cancers, compared with 508 men who did not receive treatment.
In an unadjusted model, men who had treatment were 18% less likely to develop a secondary lung cancer (hazard ratio, 0.82). That finding held in a multivariate analysis that adjusted for smoking and age (HR, 0.83).
The study was sponsored by the Veterans Health Administration. Dr. Kang had no financial disclosures.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
WASHINGTON – Men who received androgen blockade therapy as part of prostate cancer treatment were less likely to develop a secondary lung cancer than were those who didn’t receive the hormone regimen.
The significant finding suggests that estrogen may fuel non–small cell lung cancers in both men and women, Dr. Hyunseok Kang reported at the annual meeting of the American Association for Cancer Research.
"Recent literature suggests estrogen receptors and aromatase play a role in the development of non–small cell lung cancers in women with breast cancer," Dr. Kang said. "Aromatase can convert testosterone to estrogen – which could increase the same risk in men."
This recently observed association prompted Dr. Kang, a fellow at Emory University, Atlanta, and his colleagues to investigate whether a similar hormone-mediated link might exist for men with prostate cancer – and whether blocking it could also block lung cancer.
The retrospective observational study pulled data from the Veterans Affairs Central Cancer Registry. It comprised 62,589 men who were diagnosed with prostate cancer between 1999 and 2008, with follow-up through 2010.
There were some significant – and important – baseline differences between the 20,378 men who underwent androgen blockade and those who did not. Men who received the treatment were significantly older (mean age, 71 years vs. 66 years) and slightly more likely to have ever smoked tobacco (73% vs. 71%).
Prostate cancer stage at diagnosis was also significantly different, with fewer of the hormone therapy group presenting at stage II (77% vs. 92%) and more presenting at stage IV (17% vs. 2%).
Men from the VA database were significantly more likely than the general population to develop lung cancer, whether they underwent androgen blockade therapy or not (1.8/100,000 men who received treatment and 2.4/100,000 men who did not receive treatment vs. 0.80/100,000 in the general population).
Of men in the VA database who received androgen treatment, 258 developed secondary lung cancers, compared with 508 men who did not receive treatment.
In an unadjusted model, men who had treatment were 18% less likely to develop a secondary lung cancer (hazard ratio, 0.82). That finding held in a multivariate analysis that adjusted for smoking and age (HR, 0.83).
The study was sponsored by the Veterans Health Administration. Dr. Kang had no financial disclosures.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
FROM THE AACR ANNUAL MEETING