User login
Official Newspaper of the American College of Surgeons
C-section raises hysterectomy complication risk later
Women who have at least one cesarean delivery have a more than 30% risk of a complication requiring reoperation after benign hysterectomy later in life, compared with women who have had vaginal deliveries only, according to a study of more than 7,600 women in a Danish patient registry.
Cesarean delivery is the most common major surgery performed in the world, and the rate is rapidly increasing, with the global average cesarean rate estimated at 18.6%, and rates as high as 52% in some European countries. However, the impact cesarean deliveries have on surgical complications later in life has not been thoroughly studied. The study authors said this might be the first population study of the association of cesarean delivery with hysterectomy complications.
Of the 388 women (5%) who had a hysterectomy and then a reoperation within 30 days, the risk increased with the number of previous cesarean deliveries. Those who had vaginal-only deliveries had reoperation rates of 4.4%, compared with 6.2% for those who had one cesarean delivery and 6.8% for those who had two or more. That represents increased risks of 31% and 35% for women who had one cesarean delivery and two or more cesarean deliveries, respectively, compared with women who had only vaginal deliveries.
Likewise, surgical complications were 16% more frequent in women who had one previous cesarean delivery and 30% more likely in women with two or more cesarean deliveries. Women who had two or more cesarean deliveries were almost twice as likely (odds ratio, 1.93) to receive a blood transfusion.
“Our results imply that information on long-term associations should be made more readily available to women, clinicians, and policymakers and suggest that decisions on cesarean delivery should take into account not only immediate maternal and neonatal influences, but also women’s health in the long term, including an increased risk of reoperation and complications associated with surgery later in life,” the researchers wrote. “The results support policies and clinical efforts to prevent cesarean deliveries that are not medically indicated.”
The study noted some limitations, including the observational design, which did not allow for elimination of all potential confounding factors.
The researchers reported having no relevant financial disclosures.
Women who have at least one cesarean delivery have a more than 30% risk of a complication requiring reoperation after benign hysterectomy later in life, compared with women who have had vaginal deliveries only, according to a study of more than 7,600 women in a Danish patient registry.
Cesarean delivery is the most common major surgery performed in the world, and the rate is rapidly increasing, with the global average cesarean rate estimated at 18.6%, and rates as high as 52% in some European countries. However, the impact cesarean deliveries have on surgical complications later in life has not been thoroughly studied. The study authors said this might be the first population study of the association of cesarean delivery with hysterectomy complications.
Of the 388 women (5%) who had a hysterectomy and then a reoperation within 30 days, the risk increased with the number of previous cesarean deliveries. Those who had vaginal-only deliveries had reoperation rates of 4.4%, compared with 6.2% for those who had one cesarean delivery and 6.8% for those who had two or more. That represents increased risks of 31% and 35% for women who had one cesarean delivery and two or more cesarean deliveries, respectively, compared with women who had only vaginal deliveries.
Likewise, surgical complications were 16% more frequent in women who had one previous cesarean delivery and 30% more likely in women with two or more cesarean deliveries. Women who had two or more cesarean deliveries were almost twice as likely (odds ratio, 1.93) to receive a blood transfusion.
“Our results imply that information on long-term associations should be made more readily available to women, clinicians, and policymakers and suggest that decisions on cesarean delivery should take into account not only immediate maternal and neonatal influences, but also women’s health in the long term, including an increased risk of reoperation and complications associated with surgery later in life,” the researchers wrote. “The results support policies and clinical efforts to prevent cesarean deliveries that are not medically indicated.”
The study noted some limitations, including the observational design, which did not allow for elimination of all potential confounding factors.
The researchers reported having no relevant financial disclosures.
Women who have at least one cesarean delivery have a more than 30% risk of a complication requiring reoperation after benign hysterectomy later in life, compared with women who have had vaginal deliveries only, according to a study of more than 7,600 women in a Danish patient registry.
Cesarean delivery is the most common major surgery performed in the world, and the rate is rapidly increasing, with the global average cesarean rate estimated at 18.6%, and rates as high as 52% in some European countries. However, the impact cesarean deliveries have on surgical complications later in life has not been thoroughly studied. The study authors said this might be the first population study of the association of cesarean delivery with hysterectomy complications.
Of the 388 women (5%) who had a hysterectomy and then a reoperation within 30 days, the risk increased with the number of previous cesarean deliveries. Those who had vaginal-only deliveries had reoperation rates of 4.4%, compared with 6.2% for those who had one cesarean delivery and 6.8% for those who had two or more. That represents increased risks of 31% and 35% for women who had one cesarean delivery and two or more cesarean deliveries, respectively, compared with women who had only vaginal deliveries.
Likewise, surgical complications were 16% more frequent in women who had one previous cesarean delivery and 30% more likely in women with two or more cesarean deliveries. Women who had two or more cesarean deliveries were almost twice as likely (odds ratio, 1.93) to receive a blood transfusion.
“Our results imply that information on long-term associations should be made more readily available to women, clinicians, and policymakers and suggest that decisions on cesarean delivery should take into account not only immediate maternal and neonatal influences, but also women’s health in the long term, including an increased risk of reoperation and complications associated with surgery later in life,” the researchers wrote. “The results support policies and clinical efforts to prevent cesarean deliveries that are not medically indicated.”
The study noted some limitations, including the observational design, which did not allow for elimination of all potential confounding factors.
The researchers reported having no relevant financial disclosures.
FROM JAMA SURGERY
Key clinical point:
Major finding: The rate of complications after hysterectomy was 4.4% for women who had vaginal birth only, 6.2% for those who had one cesarean delivery, and 6.8% for those who had two or more cesarean deliveries.
Data source: Danish National Patient Registry–based cohort study of 7,685 women who gave birth from 1993 to 2012.
Disclosures: The researchers reported having no financial disclosures.
Award for best hospital goes to … the Mayo Clinic
For the second consecutive year, the Mayo Clinic was named the top hospital in the country by U.S. News & World Report.
Also for the second consecutive year, the Cleveland Clinic is ranked second, while Johns Hopkins Hospital in Baltimore and Massachusetts General Hospital in Boston finished third and fourth – switching their places from last year’s ranking – and UCSF Medical Center in San Francisco is fifth after ranking seventh last year, according to the 2017-2018 Best Hospitals ranking.
The Mayo Clinic is nationally ranked in 15 of the 16 specialties included in the overall process, which started with 4,658 community inpatient hospitals and finished with 152 ranking nationally in at least one specialty and 20 earning Honor Roll status with high rankings in multiple specialties. The specialties used in the ranking process include 12 that are data driven – cancer; cardiology and heart surgery; diabetes and endocrinology; otolaryngology; gastroenterology and gastrointestinal surgery; geriatrics; gynecology; nephrology; neurology and neurosurgery; orthopedics; pulmonology; and urology – and four rated by reputation only – ophthalmology; psychiatry; rehabilitation; and rheumatology.
The research organization RTI International conducted the physician survey and produced the Best Hospitals methodology and national rankings under contract with U.S. News. The launch of this year’s edition of Best Hospitals is sponsored by Fidelity Investments.
For the second consecutive year, the Mayo Clinic was named the top hospital in the country by U.S. News & World Report.
Also for the second consecutive year, the Cleveland Clinic is ranked second, while Johns Hopkins Hospital in Baltimore and Massachusetts General Hospital in Boston finished third and fourth – switching their places from last year’s ranking – and UCSF Medical Center in San Francisco is fifth after ranking seventh last year, according to the 2017-2018 Best Hospitals ranking.
The Mayo Clinic is nationally ranked in 15 of the 16 specialties included in the overall process, which started with 4,658 community inpatient hospitals and finished with 152 ranking nationally in at least one specialty and 20 earning Honor Roll status with high rankings in multiple specialties. The specialties used in the ranking process include 12 that are data driven – cancer; cardiology and heart surgery; diabetes and endocrinology; otolaryngology; gastroenterology and gastrointestinal surgery; geriatrics; gynecology; nephrology; neurology and neurosurgery; orthopedics; pulmonology; and urology – and four rated by reputation only – ophthalmology; psychiatry; rehabilitation; and rheumatology.
The research organization RTI International conducted the physician survey and produced the Best Hospitals methodology and national rankings under contract with U.S. News. The launch of this year’s edition of Best Hospitals is sponsored by Fidelity Investments.
For the second consecutive year, the Mayo Clinic was named the top hospital in the country by U.S. News & World Report.
Also for the second consecutive year, the Cleveland Clinic is ranked second, while Johns Hopkins Hospital in Baltimore and Massachusetts General Hospital in Boston finished third and fourth – switching their places from last year’s ranking – and UCSF Medical Center in San Francisco is fifth after ranking seventh last year, according to the 2017-2018 Best Hospitals ranking.
The Mayo Clinic is nationally ranked in 15 of the 16 specialties included in the overall process, which started with 4,658 community inpatient hospitals and finished with 152 ranking nationally in at least one specialty and 20 earning Honor Roll status with high rankings in multiple specialties. The specialties used in the ranking process include 12 that are data driven – cancer; cardiology and heart surgery; diabetes and endocrinology; otolaryngology; gastroenterology and gastrointestinal surgery; geriatrics; gynecology; nephrology; neurology and neurosurgery; orthopedics; pulmonology; and urology – and four rated by reputation only – ophthalmology; psychiatry; rehabilitation; and rheumatology.
The research organization RTI International conducted the physician survey and produced the Best Hospitals methodology and national rankings under contract with U.S. News. The launch of this year’s edition of Best Hospitals is sponsored by Fidelity Investments.
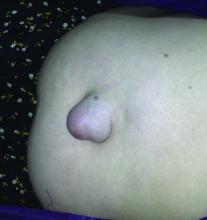
Botox smooths prep for hernia surgery
Injections of onabotulinumtoxinA prior to hernia surgery relaxed the abdominal muscles and increased abdominal wall length by an average of 8 cm, based on data from an observational study of 56 patients. The findings were published online in Surgical Endoscopy.
Although laparoscopic ventral hernia repair has a lower recurrence rate than open repair, expanding the abdominal wall remains a challenge, wrote Omar Rodriguez-Acevedo, MD, of the Hernia Institute Australia, Edgecliff, New South Wales, Australia, and colleagues (Surg Endosc. 2017 Jul 21. doi: 10.1007/s00464-017-5750-3).
Nearly three-fourths of the patients (73%) had at least one previous repair. The patients underwent injections of either 200 units or 300 units of BTA between 7 and 14 days before surgery. The average age of the patients was 60 years, and the average body mass index was 40 kg/m2. A subset of 18 patients with larger defects underwent preoperative progressive pneumoperitoneum (PPP) in addition to receiving BTA injections.
Overall, BTA injections significantly increased lateral abdominal length in all subgroups. On average, the length increase per side was 4.4 cm in the 300-unit group, 3.6 cm in the 200-unit group, 4.2 cm in the BtA-only group, and 3.7 cm in the BTA-plus-PPP group. In a pooled analysis, the average gain in length was 4.0 per side.
No significant difference in abdominal wall lengthening was observed between the 200-unit and 300-unit patients or between the BTA-plus-PPP and BTA-only patients.
Overall, the injections were well tolerated, and no complications required intervention, the researchers said. The most common side effects included superficial bruising at the injection site, bloating sensations, weak coughing, and back pain.
The findings were limited by the small study population and by the short follow-up period, and additional long-term follow-up is needed to identify delayed hernia recurrence, the researchers noted. However, the results suggest that “the flaccid paralysis delivered by BTA resulted in the relaxation, elongation, and thinning of the chronically contracted abdominal lateral wall musculature,” which “consequently facilitates laparoscopic repair and primary closure of large defects under minimal tension,” they said.
The researchers had no financial conflicts to disclose.
Injections of onabotulinumtoxinA prior to hernia surgery relaxed the abdominal muscles and increased abdominal wall length by an average of 8 cm, based on data from an observational study of 56 patients. The findings were published online in Surgical Endoscopy.
Although laparoscopic ventral hernia repair has a lower recurrence rate than open repair, expanding the abdominal wall remains a challenge, wrote Omar Rodriguez-Acevedo, MD, of the Hernia Institute Australia, Edgecliff, New South Wales, Australia, and colleagues (Surg Endosc. 2017 Jul 21. doi: 10.1007/s00464-017-5750-3).
Nearly three-fourths of the patients (73%) had at least one previous repair. The patients underwent injections of either 200 units or 300 units of BTA between 7 and 14 days before surgery. The average age of the patients was 60 years, and the average body mass index was 40 kg/m2. A subset of 18 patients with larger defects underwent preoperative progressive pneumoperitoneum (PPP) in addition to receiving BTA injections.
Overall, BTA injections significantly increased lateral abdominal length in all subgroups. On average, the length increase per side was 4.4 cm in the 300-unit group, 3.6 cm in the 200-unit group, 4.2 cm in the BtA-only group, and 3.7 cm in the BTA-plus-PPP group. In a pooled analysis, the average gain in length was 4.0 per side.
No significant difference in abdominal wall lengthening was observed between the 200-unit and 300-unit patients or between the BTA-plus-PPP and BTA-only patients.
Overall, the injections were well tolerated, and no complications required intervention, the researchers said. The most common side effects included superficial bruising at the injection site, bloating sensations, weak coughing, and back pain.
The findings were limited by the small study population and by the short follow-up period, and additional long-term follow-up is needed to identify delayed hernia recurrence, the researchers noted. However, the results suggest that “the flaccid paralysis delivered by BTA resulted in the relaxation, elongation, and thinning of the chronically contracted abdominal lateral wall musculature,” which “consequently facilitates laparoscopic repair and primary closure of large defects under minimal tension,” they said.
The researchers had no financial conflicts to disclose.
Injections of onabotulinumtoxinA prior to hernia surgery relaxed the abdominal muscles and increased abdominal wall length by an average of 8 cm, based on data from an observational study of 56 patients. The findings were published online in Surgical Endoscopy.
Although laparoscopic ventral hernia repair has a lower recurrence rate than open repair, expanding the abdominal wall remains a challenge, wrote Omar Rodriguez-Acevedo, MD, of the Hernia Institute Australia, Edgecliff, New South Wales, Australia, and colleagues (Surg Endosc. 2017 Jul 21. doi: 10.1007/s00464-017-5750-3).
Nearly three-fourths of the patients (73%) had at least one previous repair. The patients underwent injections of either 200 units or 300 units of BTA between 7 and 14 days before surgery. The average age of the patients was 60 years, and the average body mass index was 40 kg/m2. A subset of 18 patients with larger defects underwent preoperative progressive pneumoperitoneum (PPP) in addition to receiving BTA injections.
Overall, BTA injections significantly increased lateral abdominal length in all subgroups. On average, the length increase per side was 4.4 cm in the 300-unit group, 3.6 cm in the 200-unit group, 4.2 cm in the BtA-only group, and 3.7 cm in the BTA-plus-PPP group. In a pooled analysis, the average gain in length was 4.0 per side.
No significant difference in abdominal wall lengthening was observed between the 200-unit and 300-unit patients or between the BTA-plus-PPP and BTA-only patients.
Overall, the injections were well tolerated, and no complications required intervention, the researchers said. The most common side effects included superficial bruising at the injection site, bloating sensations, weak coughing, and back pain.
The findings were limited by the small study population and by the short follow-up period, and additional long-term follow-up is needed to identify delayed hernia recurrence, the researchers noted. However, the results suggest that “the flaccid paralysis delivered by BTA resulted in the relaxation, elongation, and thinning of the chronically contracted abdominal lateral wall musculature,” which “consequently facilitates laparoscopic repair and primary closure of large defects under minimal tension,” they said.
The researchers had no financial conflicts to disclose.
FROM SURGICAL ENDOSCOPY
Key clinical point: Injection with botulinum toxin A prior to hernia repair serves as an effective surgical preparation by temporarily paralyzing the lateral abdominal wall muscles.
Major finding: A comparison of pre- and post-onabotulinumtoxinA images of the abdominal wall showed an unstretched average increase in length of 8.0 cm.
Data source: A prospective, observational study of 56 adults who underwent elective ventral hernia repairs at a single center.
Disclosures: The researchers had no financial conflicts to disclose.
Umbilical hernia repair during pregnancy safe, but often serious
Umbilical hernia repair during pregnancy is rare and safe, but more than half of surgeries required incarceration or strangulation repair, according to Dr. I.N. Haskins and associates.
A total of 126 pregnant women underwent umbilical hernia repair from 2005 to 2014, according to data collected from the American College of Surgeons National Surgical Quality Improvement Program. All but six women underwent open surgery, and of these 120 patients, 71 had umbilical hernia incarceration or strangulation at the time of surgery.
“Additional studies are needed to determine the long-term recurrence rate of umbilical hernia repairs performed in pregnant patients and the effects of surgical intervention and approach on the fetus,” the investigators concluded.
Find the study in Hernia (doi: 10.1007/s10029-017-1633-8).
Umbilical hernia repair during pregnancy is rare and safe, but more than half of surgeries required incarceration or strangulation repair, according to Dr. I.N. Haskins and associates.
A total of 126 pregnant women underwent umbilical hernia repair from 2005 to 2014, according to data collected from the American College of Surgeons National Surgical Quality Improvement Program. All but six women underwent open surgery, and of these 120 patients, 71 had umbilical hernia incarceration or strangulation at the time of surgery.
“Additional studies are needed to determine the long-term recurrence rate of umbilical hernia repairs performed in pregnant patients and the effects of surgical intervention and approach on the fetus,” the investigators concluded.
Find the study in Hernia (doi: 10.1007/s10029-017-1633-8).
Umbilical hernia repair during pregnancy is rare and safe, but more than half of surgeries required incarceration or strangulation repair, according to Dr. I.N. Haskins and associates.
A total of 126 pregnant women underwent umbilical hernia repair from 2005 to 2014, according to data collected from the American College of Surgeons National Surgical Quality Improvement Program. All but six women underwent open surgery, and of these 120 patients, 71 had umbilical hernia incarceration or strangulation at the time of surgery.
“Additional studies are needed to determine the long-term recurrence rate of umbilical hernia repairs performed in pregnant patients and the effects of surgical intervention and approach on the fetus,” the investigators concluded.
Find the study in Hernia (doi: 10.1007/s10029-017-1633-8).
FROM HERNIA
ACA: Five tactics could lead to implosion
With legislative efforts to repeal and/or replace the Affordable Care Act shelved for now, President Trump has tweeted that he wants to “let ObamaCare implode, then deal.” But just what can – and is – his administration doing to foster an implosion? Policy experts help us count the ways:
1. Lax enforcement of the individual mandate
Shortly after he took office, President Trump issued an executive order aimed at “minimizing the economic burden” of the ACA. The order directed all federal agencies to take legal steps to waive, defer, grant exemptions from, or delay the implementation of any ACA provision or requirement that would impose a fiscal burden on states, patients, providers, or health insurers.
As a result, the Internal Revenue Service announced that it would not reject tax returns that do not indicate whether the taxpayer has health insurance. That question is included to determine whether taxpayers will incur a financial penalty under the individual mandate.
“What the Trump administration has done has weakened it even further by effectively saying that they will not enforce the mandate if anyone challenges it,” said Dr. Forman, a practicing radiologist and operational chief for radiology at Yale New Haven Hospital. “So if an individual claims that they shouldn’t have to face the mandate for religious reasons or other objections, that they would be allowed out. By encouraging that, you’re basically weakening the mandate even more, [which] hurts the exchanges and ultimately drives up prices.”
2. Little advertising, outreach
The Trump administration canceled advertising and outreach efforts in the final week of the 2017 open enrollment period. As many as half a million people missed out on enrolling in a health insurance plan as a result, Joshua Peck, former chief marketing officer for healthcare.gov, estimated in a recent blog post.
In the past, the federal government has played a significant role in informing the public about marketplace coverage, their rights and responsibilities under the ACA, and the process of enrollment, said Sarah Lueck, a senior policy analyst for the Center on Budget and Policy Priorities, a nonpartisan research and policy institute. The last week of enrollment is known as a critical time to enroll patients, she said. In 2016 for example, about 700,000 people enrolled during the final week. It’s often the healthiest patients who wait until the last minute, Ms. Lueck added.
“One way you discourage healthy people from enrolling is by pulling back on advertising at the very moment they may be paying attention,” she said in an interview. “It sends a bad signal. Now as the next enrollment period is about to come up in November, it raises a concern about – what are the plans for outreach?”
Without sufficient promotion, the number of patients who learn about the ACA and enroll could drop off, and the percentage of sicker enrollees in the marketplace could rise, according to analysts. The Trump administration has not said whether it plans to advertise or promote enrollment during the upcoming November enrollment period. The Centers for Medicare & Medicaid Services recently shortened open enrollment from the previous 3 months to 45 days.
In a final rule issued in April, the CMS stated the change will “improve individual market risk pools by reducing opportunities for adverse selection ... and will encourage healthier individuals who might have previously enrolled in partial year coverage after December 15th to instead enroll in coverage for the full year.”
3. Highlight what’s “wrong” with the ACA
In addition to pulling positive advertisements about ACA, the Trump administration has also launched a campaign that criticizes the law.
Since January, the Department of Health & Human Services has published more than 20 videos featuring stories about how the ACA has harmed patients. The HHS has also used its Twitter account to advocate repeal and replacement of the ACA. Sen. Ron Wyden (D-Ore.), ranking member of the Finance Committee, and other legislators have raised concerns that the HHS is misusing federal resources to advance partisan legislation by funding the messages.
“It’s not just pulling advertisements and going dark and not telling people [information], but it’s also putting things out there that talk about people who don’t like the law,” Ms. Lueck said. “It’s counterproductive propaganda if you’re coming from the perspective of wanting people to sign up for coverage. The agencies that have been very engaged in trying to get people through the process and covered, are now working in cross purposes with that.”
Ms. Lueck said that the federal government is also putting a negative spin on the current participation of marketplace insurers and the future of the exchanges. On Aug. 2, the CMS released a map on projected insurer participation in the ACA’s 2018 health insurance exchanges. The map shows that 19 counties are projected to have no insurers in 2018, meaning that patients in those counties could be without marketplace options.
“For 2018, at least 13,008 Americans currently enrolled for health coverage on the exchanges live in the counties projected to be without coverage in 2018,” according to the CMS announcement. “In addition to overall issuer participation, increasing rates have also been a concern for the health insurance exchanges. ... A number of insurers in several states requested rate increases of 30% or more. Consumers in the 39 healthcare.gov states have already seen their premiums increase more than 100% since 2013.”
Unmentioned however, is that the number of potential “bare counties” has dropped in half from about a month ago. A similar map by the Kaiser Family Foundation shows that in June, 44 counties were at risk of having no marketplace insurer in 2018, a number that fell to 17 counties as of Aug. 4, according to Kaiser’s most recent map.
Kristine Grow, senior vice president of communications for America’s Health Insurance Plans (AHIP), noted that based on CMS’ projections thus far, the overall percentage of enrollees without an insurer for 2018 is 0.15%.
“We’re talking pretty small numbers, that’s about 15,000 people out of 10 million or so who get their coverage through an exchange,” Ms. Grow said in an interview. “It’s important for those people to have options, so the health plans have been working very hard to try to get into those counties.”
4. Work for Medicaid recipients
Potential work requirements for Medicaid beneficiaries may harm the Medicaid expansions that were part of the ACA.
On March 14, the HHS sent a letter to 50 U.S. governors encouraging states to come up with innovative ideas for their Medicaid programs, including the possibility of work requirements. The letter included specific suggestions, such as introducing plans that include health savings account–like features, encouraging Medicaid patients to secure employer-provided insurance, and requiring small premiums or other contributions from patients to encourage personal responsibility. The letter noted that the HHS would be open to states proposing work requirements for some Medicaid recipients, an approach that has “produced proven results for Americans enrolled in other federal, state, and local programs.”
Four states – Arizona, Indiana, Kentucky, and Pennsylvania – have formally submitted waiver requests to the HHS that would require work as an eligibility condition. To date, none has been approved. Arkansas also recently announced that it would seek changes to its waiver, including a work requirement.
Imposing work requirements would hurt access to Medicaid for patients who need health assistance, but who cannot work, Dr. Forman said. Under the ACA, 31 states and the District of Columbia have expanded their Medicaid coverage to people previously uncovered. Dr. Forman stresses that the bulk of Medicaid funding is spent on elderly, disabled, and mentally ill patients.
5. Withhold cost sharing reduction payments
For months, President Trump has threatened to stop making cost saving reduction (CSR) payments to insurers in the marketplace, a move that analysts say would raise premiums and cause insurers to exit the marketplaces. Most recently, the President on July 29 tweeted, “If a new HealthCare Bill is not approved quickly, BAILOUTS for Insurance Companies and BAILOUTS for Members of Congress will end very soon!”
Under the ACA, the federal government provides CSR payments to insurers to offset the costs for providing discount plans to patients who earn up to 200% of the federal poverty level. Plans on the individual exchanges are required to cover a package of essential benefits with pricing limitations to ensure that out-of-pocket costs are low enough for poorer patients. Because insurers lose money on these plans, the ACA provides about $7 billion to insurers through CSR payments.
Republican members of the House of Representatives sued the HHS over the CSR payments under the Obama administration, claiming the funding was illegal because it was never appropriated by Congress. A court ruled in favor of the House in 2016, but an appeal filed by the Obama administration allowed the CSR payments to continue.
President Trump has not indicated whether he plans to drop the appeal or carry on the case. But if he fails to continue the suit, the move would immediately end the CSR payments.
“If the funding for the CSR benefits goes away, premiums will go up, taxpayer dollars will go up, and choices will go down,” Ms. Grow of AHIP said in an interview. “The benefits as we understand them are still required to be offered on the exchanges. In order to cover those benefits, the premiums for everybody in the individual market will have to go up, and they will go up by about 20%.”
While the federal government would save money by ending the CSR payments, it would face increased costs for tax credits that subsidize premiums for marketplace enrollees with incomes that are 100% to 400% of the poverty level, according to an analysis by the Kaiser Family Foundation.
Following President Trump’s most recent threat to stop the CSR payments, AHIP issued a joint statement with the American Academy of Family Physicians, the American Medical Association, and several others underscoring the importance of the payments.
“Cost-sharing reductions are used to help those who need it most – low- and moderate-income consumers,” the associations said in the Aug. 2 statement. “Without these funds, consumers’ access to care is jeopardized, their premiums will increase dramatically, and they will be left with even fewer coverage options ... As medical professionals, insurers providing health care services and coverage to hundreds of millions of Americans, and business leaders concerned with maintaining a stable health insurance marketplace for consumers, we believe it is imperative that the administration fund the cost-sharing reduction program.”
[email protected]
On Twitter @legal_med
With legislative efforts to repeal and/or replace the Affordable Care Act shelved for now, President Trump has tweeted that he wants to “let ObamaCare implode, then deal.” But just what can – and is – his administration doing to foster an implosion? Policy experts help us count the ways:
1. Lax enforcement of the individual mandate
Shortly after he took office, President Trump issued an executive order aimed at “minimizing the economic burden” of the ACA. The order directed all federal agencies to take legal steps to waive, defer, grant exemptions from, or delay the implementation of any ACA provision or requirement that would impose a fiscal burden on states, patients, providers, or health insurers.
As a result, the Internal Revenue Service announced that it would not reject tax returns that do not indicate whether the taxpayer has health insurance. That question is included to determine whether taxpayers will incur a financial penalty under the individual mandate.
“What the Trump administration has done has weakened it even further by effectively saying that they will not enforce the mandate if anyone challenges it,” said Dr. Forman, a practicing radiologist and operational chief for radiology at Yale New Haven Hospital. “So if an individual claims that they shouldn’t have to face the mandate for religious reasons or other objections, that they would be allowed out. By encouraging that, you’re basically weakening the mandate even more, [which] hurts the exchanges and ultimately drives up prices.”
2. Little advertising, outreach
The Trump administration canceled advertising and outreach efforts in the final week of the 2017 open enrollment period. As many as half a million people missed out on enrolling in a health insurance plan as a result, Joshua Peck, former chief marketing officer for healthcare.gov, estimated in a recent blog post.
In the past, the federal government has played a significant role in informing the public about marketplace coverage, their rights and responsibilities under the ACA, and the process of enrollment, said Sarah Lueck, a senior policy analyst for the Center on Budget and Policy Priorities, a nonpartisan research and policy institute. The last week of enrollment is known as a critical time to enroll patients, she said. In 2016 for example, about 700,000 people enrolled during the final week. It’s often the healthiest patients who wait until the last minute, Ms. Lueck added.
“One way you discourage healthy people from enrolling is by pulling back on advertising at the very moment they may be paying attention,” she said in an interview. “It sends a bad signal. Now as the next enrollment period is about to come up in November, it raises a concern about – what are the plans for outreach?”
Without sufficient promotion, the number of patients who learn about the ACA and enroll could drop off, and the percentage of sicker enrollees in the marketplace could rise, according to analysts. The Trump administration has not said whether it plans to advertise or promote enrollment during the upcoming November enrollment period. The Centers for Medicare & Medicaid Services recently shortened open enrollment from the previous 3 months to 45 days.
In a final rule issued in April, the CMS stated the change will “improve individual market risk pools by reducing opportunities for adverse selection ... and will encourage healthier individuals who might have previously enrolled in partial year coverage after December 15th to instead enroll in coverage for the full year.”
3. Highlight what’s “wrong” with the ACA
In addition to pulling positive advertisements about ACA, the Trump administration has also launched a campaign that criticizes the law.
Since January, the Department of Health & Human Services has published more than 20 videos featuring stories about how the ACA has harmed patients. The HHS has also used its Twitter account to advocate repeal and replacement of the ACA. Sen. Ron Wyden (D-Ore.), ranking member of the Finance Committee, and other legislators have raised concerns that the HHS is misusing federal resources to advance partisan legislation by funding the messages.
“It’s not just pulling advertisements and going dark and not telling people [information], but it’s also putting things out there that talk about people who don’t like the law,” Ms. Lueck said. “It’s counterproductive propaganda if you’re coming from the perspective of wanting people to sign up for coverage. The agencies that have been very engaged in trying to get people through the process and covered, are now working in cross purposes with that.”
Ms. Lueck said that the federal government is also putting a negative spin on the current participation of marketplace insurers and the future of the exchanges. On Aug. 2, the CMS released a map on projected insurer participation in the ACA’s 2018 health insurance exchanges. The map shows that 19 counties are projected to have no insurers in 2018, meaning that patients in those counties could be without marketplace options.
“For 2018, at least 13,008 Americans currently enrolled for health coverage on the exchanges live in the counties projected to be without coverage in 2018,” according to the CMS announcement. “In addition to overall issuer participation, increasing rates have also been a concern for the health insurance exchanges. ... A number of insurers in several states requested rate increases of 30% or more. Consumers in the 39 healthcare.gov states have already seen their premiums increase more than 100% since 2013.”
Unmentioned however, is that the number of potential “bare counties” has dropped in half from about a month ago. A similar map by the Kaiser Family Foundation shows that in June, 44 counties were at risk of having no marketplace insurer in 2018, a number that fell to 17 counties as of Aug. 4, according to Kaiser’s most recent map.
Kristine Grow, senior vice president of communications for America’s Health Insurance Plans (AHIP), noted that based on CMS’ projections thus far, the overall percentage of enrollees without an insurer for 2018 is 0.15%.
“We’re talking pretty small numbers, that’s about 15,000 people out of 10 million or so who get their coverage through an exchange,” Ms. Grow said in an interview. “It’s important for those people to have options, so the health plans have been working very hard to try to get into those counties.”
4. Work for Medicaid recipients
Potential work requirements for Medicaid beneficiaries may harm the Medicaid expansions that were part of the ACA.
On March 14, the HHS sent a letter to 50 U.S. governors encouraging states to come up with innovative ideas for their Medicaid programs, including the possibility of work requirements. The letter included specific suggestions, such as introducing plans that include health savings account–like features, encouraging Medicaid patients to secure employer-provided insurance, and requiring small premiums or other contributions from patients to encourage personal responsibility. The letter noted that the HHS would be open to states proposing work requirements for some Medicaid recipients, an approach that has “produced proven results for Americans enrolled in other federal, state, and local programs.”
Four states – Arizona, Indiana, Kentucky, and Pennsylvania – have formally submitted waiver requests to the HHS that would require work as an eligibility condition. To date, none has been approved. Arkansas also recently announced that it would seek changes to its waiver, including a work requirement.
Imposing work requirements would hurt access to Medicaid for patients who need health assistance, but who cannot work, Dr. Forman said. Under the ACA, 31 states and the District of Columbia have expanded their Medicaid coverage to people previously uncovered. Dr. Forman stresses that the bulk of Medicaid funding is spent on elderly, disabled, and mentally ill patients.
5. Withhold cost sharing reduction payments
For months, President Trump has threatened to stop making cost saving reduction (CSR) payments to insurers in the marketplace, a move that analysts say would raise premiums and cause insurers to exit the marketplaces. Most recently, the President on July 29 tweeted, “If a new HealthCare Bill is not approved quickly, BAILOUTS for Insurance Companies and BAILOUTS for Members of Congress will end very soon!”
Under the ACA, the federal government provides CSR payments to insurers to offset the costs for providing discount plans to patients who earn up to 200% of the federal poverty level. Plans on the individual exchanges are required to cover a package of essential benefits with pricing limitations to ensure that out-of-pocket costs are low enough for poorer patients. Because insurers lose money on these plans, the ACA provides about $7 billion to insurers through CSR payments.
Republican members of the House of Representatives sued the HHS over the CSR payments under the Obama administration, claiming the funding was illegal because it was never appropriated by Congress. A court ruled in favor of the House in 2016, but an appeal filed by the Obama administration allowed the CSR payments to continue.
President Trump has not indicated whether he plans to drop the appeal or carry on the case. But if he fails to continue the suit, the move would immediately end the CSR payments.
“If the funding for the CSR benefits goes away, premiums will go up, taxpayer dollars will go up, and choices will go down,” Ms. Grow of AHIP said in an interview. “The benefits as we understand them are still required to be offered on the exchanges. In order to cover those benefits, the premiums for everybody in the individual market will have to go up, and they will go up by about 20%.”
While the federal government would save money by ending the CSR payments, it would face increased costs for tax credits that subsidize premiums for marketplace enrollees with incomes that are 100% to 400% of the poverty level, according to an analysis by the Kaiser Family Foundation.
Following President Trump’s most recent threat to stop the CSR payments, AHIP issued a joint statement with the American Academy of Family Physicians, the American Medical Association, and several others underscoring the importance of the payments.
“Cost-sharing reductions are used to help those who need it most – low- and moderate-income consumers,” the associations said in the Aug. 2 statement. “Without these funds, consumers’ access to care is jeopardized, their premiums will increase dramatically, and they will be left with even fewer coverage options ... As medical professionals, insurers providing health care services and coverage to hundreds of millions of Americans, and business leaders concerned with maintaining a stable health insurance marketplace for consumers, we believe it is imperative that the administration fund the cost-sharing reduction program.”
[email protected]
On Twitter @legal_med
With legislative efforts to repeal and/or replace the Affordable Care Act shelved for now, President Trump has tweeted that he wants to “let ObamaCare implode, then deal.” But just what can – and is – his administration doing to foster an implosion? Policy experts help us count the ways:
1. Lax enforcement of the individual mandate
Shortly after he took office, President Trump issued an executive order aimed at “minimizing the economic burden” of the ACA. The order directed all federal agencies to take legal steps to waive, defer, grant exemptions from, or delay the implementation of any ACA provision or requirement that would impose a fiscal burden on states, patients, providers, or health insurers.
As a result, the Internal Revenue Service announced that it would not reject tax returns that do not indicate whether the taxpayer has health insurance. That question is included to determine whether taxpayers will incur a financial penalty under the individual mandate.
“What the Trump administration has done has weakened it even further by effectively saying that they will not enforce the mandate if anyone challenges it,” said Dr. Forman, a practicing radiologist and operational chief for radiology at Yale New Haven Hospital. “So if an individual claims that they shouldn’t have to face the mandate for religious reasons or other objections, that they would be allowed out. By encouraging that, you’re basically weakening the mandate even more, [which] hurts the exchanges and ultimately drives up prices.”
2. Little advertising, outreach
The Trump administration canceled advertising and outreach efforts in the final week of the 2017 open enrollment period. As many as half a million people missed out on enrolling in a health insurance plan as a result, Joshua Peck, former chief marketing officer for healthcare.gov, estimated in a recent blog post.
In the past, the federal government has played a significant role in informing the public about marketplace coverage, their rights and responsibilities under the ACA, and the process of enrollment, said Sarah Lueck, a senior policy analyst for the Center on Budget and Policy Priorities, a nonpartisan research and policy institute. The last week of enrollment is known as a critical time to enroll patients, she said. In 2016 for example, about 700,000 people enrolled during the final week. It’s often the healthiest patients who wait until the last minute, Ms. Lueck added.
“One way you discourage healthy people from enrolling is by pulling back on advertising at the very moment they may be paying attention,” she said in an interview. “It sends a bad signal. Now as the next enrollment period is about to come up in November, it raises a concern about – what are the plans for outreach?”
Without sufficient promotion, the number of patients who learn about the ACA and enroll could drop off, and the percentage of sicker enrollees in the marketplace could rise, according to analysts. The Trump administration has not said whether it plans to advertise or promote enrollment during the upcoming November enrollment period. The Centers for Medicare & Medicaid Services recently shortened open enrollment from the previous 3 months to 45 days.
In a final rule issued in April, the CMS stated the change will “improve individual market risk pools by reducing opportunities for adverse selection ... and will encourage healthier individuals who might have previously enrolled in partial year coverage after December 15th to instead enroll in coverage for the full year.”
3. Highlight what’s “wrong” with the ACA
In addition to pulling positive advertisements about ACA, the Trump administration has also launched a campaign that criticizes the law.
Since January, the Department of Health & Human Services has published more than 20 videos featuring stories about how the ACA has harmed patients. The HHS has also used its Twitter account to advocate repeal and replacement of the ACA. Sen. Ron Wyden (D-Ore.), ranking member of the Finance Committee, and other legislators have raised concerns that the HHS is misusing federal resources to advance partisan legislation by funding the messages.
“It’s not just pulling advertisements and going dark and not telling people [information], but it’s also putting things out there that talk about people who don’t like the law,” Ms. Lueck said. “It’s counterproductive propaganda if you’re coming from the perspective of wanting people to sign up for coverage. The agencies that have been very engaged in trying to get people through the process and covered, are now working in cross purposes with that.”
Ms. Lueck said that the federal government is also putting a negative spin on the current participation of marketplace insurers and the future of the exchanges. On Aug. 2, the CMS released a map on projected insurer participation in the ACA’s 2018 health insurance exchanges. The map shows that 19 counties are projected to have no insurers in 2018, meaning that patients in those counties could be without marketplace options.
“For 2018, at least 13,008 Americans currently enrolled for health coverage on the exchanges live in the counties projected to be without coverage in 2018,” according to the CMS announcement. “In addition to overall issuer participation, increasing rates have also been a concern for the health insurance exchanges. ... A number of insurers in several states requested rate increases of 30% or more. Consumers in the 39 healthcare.gov states have already seen their premiums increase more than 100% since 2013.”
Unmentioned however, is that the number of potential “bare counties” has dropped in half from about a month ago. A similar map by the Kaiser Family Foundation shows that in June, 44 counties were at risk of having no marketplace insurer in 2018, a number that fell to 17 counties as of Aug. 4, according to Kaiser’s most recent map.
Kristine Grow, senior vice president of communications for America’s Health Insurance Plans (AHIP), noted that based on CMS’ projections thus far, the overall percentage of enrollees without an insurer for 2018 is 0.15%.
“We’re talking pretty small numbers, that’s about 15,000 people out of 10 million or so who get their coverage through an exchange,” Ms. Grow said in an interview. “It’s important for those people to have options, so the health plans have been working very hard to try to get into those counties.”
4. Work for Medicaid recipients
Potential work requirements for Medicaid beneficiaries may harm the Medicaid expansions that were part of the ACA.
On March 14, the HHS sent a letter to 50 U.S. governors encouraging states to come up with innovative ideas for their Medicaid programs, including the possibility of work requirements. The letter included specific suggestions, such as introducing plans that include health savings account–like features, encouraging Medicaid patients to secure employer-provided insurance, and requiring small premiums or other contributions from patients to encourage personal responsibility. The letter noted that the HHS would be open to states proposing work requirements for some Medicaid recipients, an approach that has “produced proven results for Americans enrolled in other federal, state, and local programs.”
Four states – Arizona, Indiana, Kentucky, and Pennsylvania – have formally submitted waiver requests to the HHS that would require work as an eligibility condition. To date, none has been approved. Arkansas also recently announced that it would seek changes to its waiver, including a work requirement.
Imposing work requirements would hurt access to Medicaid for patients who need health assistance, but who cannot work, Dr. Forman said. Under the ACA, 31 states and the District of Columbia have expanded their Medicaid coverage to people previously uncovered. Dr. Forman stresses that the bulk of Medicaid funding is spent on elderly, disabled, and mentally ill patients.
5. Withhold cost sharing reduction payments
For months, President Trump has threatened to stop making cost saving reduction (CSR) payments to insurers in the marketplace, a move that analysts say would raise premiums and cause insurers to exit the marketplaces. Most recently, the President on July 29 tweeted, “If a new HealthCare Bill is not approved quickly, BAILOUTS for Insurance Companies and BAILOUTS for Members of Congress will end very soon!”
Under the ACA, the federal government provides CSR payments to insurers to offset the costs for providing discount plans to patients who earn up to 200% of the federal poverty level. Plans on the individual exchanges are required to cover a package of essential benefits with pricing limitations to ensure that out-of-pocket costs are low enough for poorer patients. Because insurers lose money on these plans, the ACA provides about $7 billion to insurers through CSR payments.
Republican members of the House of Representatives sued the HHS over the CSR payments under the Obama administration, claiming the funding was illegal because it was never appropriated by Congress. A court ruled in favor of the House in 2016, but an appeal filed by the Obama administration allowed the CSR payments to continue.
President Trump has not indicated whether he plans to drop the appeal or carry on the case. But if he fails to continue the suit, the move would immediately end the CSR payments.
“If the funding for the CSR benefits goes away, premiums will go up, taxpayer dollars will go up, and choices will go down,” Ms. Grow of AHIP said in an interview. “The benefits as we understand them are still required to be offered on the exchanges. In order to cover those benefits, the premiums for everybody in the individual market will have to go up, and they will go up by about 20%.”
While the federal government would save money by ending the CSR payments, it would face increased costs for tax credits that subsidize premiums for marketplace enrollees with incomes that are 100% to 400% of the poverty level, according to an analysis by the Kaiser Family Foundation.
Following President Trump’s most recent threat to stop the CSR payments, AHIP issued a joint statement with the American Academy of Family Physicians, the American Medical Association, and several others underscoring the importance of the payments.
“Cost-sharing reductions are used to help those who need it most – low- and moderate-income consumers,” the associations said in the Aug. 2 statement. “Without these funds, consumers’ access to care is jeopardized, their premiums will increase dramatically, and they will be left with even fewer coverage options ... As medical professionals, insurers providing health care services and coverage to hundreds of millions of Americans, and business leaders concerned with maintaining a stable health insurance marketplace for consumers, we believe it is imperative that the administration fund the cost-sharing reduction program.”
[email protected]
On Twitter @legal_med
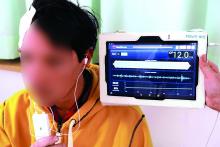
Small study advances noninvasive ICP monitoring
A device that noninvasively measures intracranial pressure (ICP) had a sensitivity of 75% and a specificity of 89%, compared with standard invasive monitoring, based on the results of an industry-sponsored study of 14 patients with traumatic brain injury or subarachnoid hemorrhage.
“This study provides the first clinical data on the accuracy of the HS-1000 noninvasive ICP monitor, which uses advanced signal analysis algorithms to evaluate properties of acoustic signals traveling through the brain,” wrote Oliver Ganslandt, MD, of Klinikum Stuttgart (Germany) and his associates. The findings were published online Aug. 8 in the Journal of Neurosurgery.
The noninvasive and invasive measurements produced more than 2,500 parallel ICP data points. Notably, each of the two methods produced the same number of data points. Readings averaged 10 (standard deviation, 6.1) mm Hg with invasive monitoring and 9.5 (SD, 4.7) mm Hg for noninvasive monitoring with the HS-1000. Compared with invasive ICP monitoring, the HS-1000 had a sensitivity of 75% and a specificity of 89% at an arbitrary cutoff of at least 17 mm Hg. Linear regression showed a “strong positive relationship between the [noninvasive and invasive] measurements,” the investigators said. In all, 63% of paired data points fell within 3 mm Hg of each other, and 85% fell within 5 mm Hg of each other. A receiver operating characteristic area under the curve analysis of the two methods generated an area under the curve of almost 90%.
The study did not include children or patients who were pregnant or had ear disease or ear injuries, rhinorrhea or otorrhea, or skull defects, the researchers said. If the HS-1000 holds up in other ongoing studies (NCT02284217, NCT02773901), physicians might be able to use it to decide if patients needs invasive ICP monitoring, they added. Use of the noninvasive method could also help prevent infections and other morbidity associated with invasive ICP monitoring in both neurocritical intensive care units and low-resource settings, they said.
HeadSense Medical sponsored the study. The researchers had no relevant disclosures.
A device that noninvasively measures intracranial pressure (ICP) had a sensitivity of 75% and a specificity of 89%, compared with standard invasive monitoring, based on the results of an industry-sponsored study of 14 patients with traumatic brain injury or subarachnoid hemorrhage.
“This study provides the first clinical data on the accuracy of the HS-1000 noninvasive ICP monitor, which uses advanced signal analysis algorithms to evaluate properties of acoustic signals traveling through the brain,” wrote Oliver Ganslandt, MD, of Klinikum Stuttgart (Germany) and his associates. The findings were published online Aug. 8 in the Journal of Neurosurgery.
The noninvasive and invasive measurements produced more than 2,500 parallel ICP data points. Notably, each of the two methods produced the same number of data points. Readings averaged 10 (standard deviation, 6.1) mm Hg with invasive monitoring and 9.5 (SD, 4.7) mm Hg for noninvasive monitoring with the HS-1000. Compared with invasive ICP monitoring, the HS-1000 had a sensitivity of 75% and a specificity of 89% at an arbitrary cutoff of at least 17 mm Hg. Linear regression showed a “strong positive relationship between the [noninvasive and invasive] measurements,” the investigators said. In all, 63% of paired data points fell within 3 mm Hg of each other, and 85% fell within 5 mm Hg of each other. A receiver operating characteristic area under the curve analysis of the two methods generated an area under the curve of almost 90%.
The study did not include children or patients who were pregnant or had ear disease or ear injuries, rhinorrhea or otorrhea, or skull defects, the researchers said. If the HS-1000 holds up in other ongoing studies (NCT02284217, NCT02773901), physicians might be able to use it to decide if patients needs invasive ICP monitoring, they added. Use of the noninvasive method could also help prevent infections and other morbidity associated with invasive ICP monitoring in both neurocritical intensive care units and low-resource settings, they said.
HeadSense Medical sponsored the study. The researchers had no relevant disclosures.
A device that noninvasively measures intracranial pressure (ICP) had a sensitivity of 75% and a specificity of 89%, compared with standard invasive monitoring, based on the results of an industry-sponsored study of 14 patients with traumatic brain injury or subarachnoid hemorrhage.
“This study provides the first clinical data on the accuracy of the HS-1000 noninvasive ICP monitor, which uses advanced signal analysis algorithms to evaluate properties of acoustic signals traveling through the brain,” wrote Oliver Ganslandt, MD, of Klinikum Stuttgart (Germany) and his associates. The findings were published online Aug. 8 in the Journal of Neurosurgery.
The noninvasive and invasive measurements produced more than 2,500 parallel ICP data points. Notably, each of the two methods produced the same number of data points. Readings averaged 10 (standard deviation, 6.1) mm Hg with invasive monitoring and 9.5 (SD, 4.7) mm Hg for noninvasive monitoring with the HS-1000. Compared with invasive ICP monitoring, the HS-1000 had a sensitivity of 75% and a specificity of 89% at an arbitrary cutoff of at least 17 mm Hg. Linear regression showed a “strong positive relationship between the [noninvasive and invasive] measurements,” the investigators said. In all, 63% of paired data points fell within 3 mm Hg of each other, and 85% fell within 5 mm Hg of each other. A receiver operating characteristic area under the curve analysis of the two methods generated an area under the curve of almost 90%.
The study did not include children or patients who were pregnant or had ear disease or ear injuries, rhinorrhea or otorrhea, or skull defects, the researchers said. If the HS-1000 holds up in other ongoing studies (NCT02284217, NCT02773901), physicians might be able to use it to decide if patients needs invasive ICP monitoring, they added. Use of the noninvasive method could also help prevent infections and other morbidity associated with invasive ICP monitoring in both neurocritical intensive care units and low-resource settings, they said.
HeadSense Medical sponsored the study. The researchers had no relevant disclosures.
FROM THE JOURNAL OF NEUROSURGERY
Key clinical point: A noninvasive device that measures intracranial pressure generated data that was comparable with standard invasive methods.
Major finding: Sensitivity was 75%, and specificity was 89%, compared with standard invasive monitoring at an arbitrary cutoff of at least 17 mm Hg.
Data source: Noninvasive and invasive intracranial pressure monitoring of 14 patients with traumatic brain injury or subarachnoid hemorrhage.
Disclosures: HeadSense Medical sponsored the study. The researchers had no relevant disclosures.
Federal medical tort reform: Has its time come?
Question: Congressional proposals on medical tort reform can be expected to include the following, except:
A. A no-fault system akin to automobile no-fault insurance.
B. A cap on noneconomic damages.
C. “Safe-harbor” immunity against medical negligence.
D. Health courts in place of the judge/jury system to adjudicate claims.
E. Promotion of laws that encourage apologies and error disclosures.
Answer: A. Under the current Republican administration, one can expect legislative efforts at federal tort reform, especially given that Thomas E. Price, MD, the new secretary of the Department of Health & Human Services, is an orthopedic surgeon who has spoken passionately about defensive medicine, damage caps, health tribunals, and practice guidelines. As a former House representative for Georgia, Dr. Price has introduced several tort reform bills, so it is likely that any omnibus federal law will incorporate some of his proposals.1
Over the decades, many states have gone ahead in enacting their own statutes while awaiting federal action. Iowa is the latest example. It recently passed legislation that included a noneconomic damages cap of $250,000, stronger expert witness standards, a certificate of merit in all medical liability lawsuits, and an expansion of its “candor” protections.2 Additional reforms in other states include pretrial screening panels; arbitration; structured periodic payments in lieu of lump sum payments; penalties for frivolous suits; shortened statutes of limitations; making the loser bear all litigation costs; abolishing the collateral source rule, as well as joint and several liability; and limiting attorney contingency fees.
The best-known reform is a cap on noneconomic losses, such as pain and suffering, that doesn’t abridge compensation for economic losses, i.e., medical expenses and lost wages. This provides some predictability because noneconomic damages are difficult to quantify, and jury sympathy may result in unrealistically high payments.
Interestingly, Dr. Price himself has not pushed for a federal cap on noneconomic damages, but other Republican bills have proposed a cap of $250,000. Many states, such as California, Kansas, and Texas, have seen their cap statutes withstand constitutional challenge. However, other jurisdictions, notably Georgia, Illinois, and Missouri, have ruled them unconstitutional.
California’s law, popularly known as MICRA (Medical Injury Compensation Reform Act), came under renewed attack in 2015 with a wrongful death suit from hemorrhagic complications related to Coumadin (warfarin) use following heart surgery.3 The plaintiff’s constitutional challenges included violation of equal protection, due process, and the right to a jury trial, but these were essentially all grounded on an entitlement to recover additional noneconomic damages sufficient to cover attorney fees. The trial court had reduced her $1 million noneconomic damages to $250,000, as required under MICRA. A California court of appeal rejected her claim as being “contrary to many well-established legal principles.”
On the other hand, Florida’s Supreme Court recently held in a closely divided decision of 4-3 that the state’s caps were unconstitutional.4 The law limited noneconomic damages in malpractice cases to either $500,000 or $1 million if the injuries were catastrophic. The court ruled that the caps were arbitrary and unfairly hurt the most severely injured. It was unconvinced that they would reduce malpractice insurance rates; at any rate, there was no present crisis to justify the caps. The decision came 3 years after the court had struck down caps in a case of wrongful death.5
Three relative newcomers to the legal landscape – health courts, apology laws, and safe harbors – appear to be taking center stage in any forthcoming federal reform measures.
Health courts
Under this proposal, so-called health panels and tribunals would now adjudicate malpractice claims. Such health courts would dispense with the jury; further, regular judges would be replaced with specialized judges who would make binding determinations. In one version, a panel of medical experts would initially screen the complaint, followed by an administrative health care tribunal that would feature judges with medical expertise. These tribunals would issue binding rulings, but either party could appeal to a state court for a reversal.
In countries such as Scandinavia and New Zealand, these administrative compensation approaches are coupled to a no-fault system and appear to work well. However, unlike auto no-fault and workers’ compensation, the notion of medical no-fault has never caught on in the United States.
As currently construed, health courts evince dramatic departures from traditional rules of civil procedure. For one, the panels may render decisions before discovery has occurred, which would substantially limit a patient’s ability to learn the facts of what had happened to cause the injury. The panel may rely on a standard of “gross negligence” instead of “ordinary negligence,” requiring evidence not merely of substandard care but of recklessness. This would be a heavier burden on the victim, and could be expected to generate stiff opposition from the plaintiff’s bar. In addition, evidentiary rules may be modified, requiring that an appeal show with clear and convincing proof that the tribunal had erred.
Apology law
Disclosure of medical errors to the injured patient is believed to serve as an ethical and effective way of thwarting potential malpractice claims. Many states have enacted so-called apology laws that disallow statements of sympathy from being admitted into evidence. In some cases, these laws may assist the physician.
For example, the Ohio Supreme Court ruled that a surgeon’s comments and alleged admission of guilt (“I take full responsibility for this” regarding accidentally sectioning the common bile duct) were properly shielded from discovery by the state’s apology statute.6 Apology laws vary from state to state, and some do not shield admissions regarding causation of error or fault.
However, it is unclear if apology laws work. A recent study from Vanderbilt University reported that, for physicians who do not regularly perform surgery, apology laws actually increased the probability of facing a lawsuit.7 And for surgeons, apology laws do not have a substantial effect on the probability of facing a claim or the average payment made to resolve a claim.
Safe harbors
A proposal released by U.S. House Speaker Paul Ryan (R-Wis.) in June 2016 made reference to “safe harbors” from liability for those adhering to clinical practice guidelines. The Institute of Medicine defines practice guidelines as “systematically developed statements to assist practitioner and patient decisions about appropriate health care for specific clinical circumstances.”
There are thousands of guidelines that have been developed by medical organizations and governmental agencies, as well as by insurance carriers, managed care organizations, and others. They purport to define the best evidence-based medicine, and if they were arrived at by the consensus of an authoritative body of experts, courts will tend to view them as reflective, though not necessarily dispositive, of customary medical standards.
Theoretically, adherence to guidelines could reduce the practice of defensive medicine and improve the quality of care. However, the available evidence does not indicate that guideline-based safe harbors will prove very effective in reducing malpractice claims: They are inapplicable in 85% of cases, and they have been estimated to eliminate defendants’ payments in less than 1% of claims.
Whether any form of tort reform emerges from the current Congress is as much about politics as it is about justice. It comes at an inopportune time, given the impasse over the health care debate. Still, on June 29, 2017, the U.S. House passed a medical liability reform bill with a vote of 218-210 along party lines that would cap noneconomic damages at $250,000, shorten the statute of limitations to 3 years after the date of injury, and abolish joint and several liability.8 The outlook in the U.S. Senate, however, is anything but certain.
References
1. N Engl J Med. 2017 May 11;376(19):1806-8.
2. “Sweeping new tort reforms will protect Iowa physicians” AMA Wire. June 1, 2017.
3. Chan v. Curran, 237 CA 4th 601 (2015).
4. N. Broward Hospital District v. Kalitan, (Florida Supreme Court, decided June 8, 2017).
5. Estate of Michelle Evette McCall v. U.S., 2014 Fla LEXIS 933 (No. SC 11-1148; March 13, 2014).
6. Estate of Johnson v. Randall Smith, Inc., 135 Ohio St.3d 440 (2013).
7. “Sorry is Never Enough: The Effect of State Apology Laws on Medical Malpractice Liability Risk” SSRN. 2016 Dec 10.
8. Protecting Access to Care Act of 2017 (H.R. 1215).
Question: Congressional proposals on medical tort reform can be expected to include the following, except:
A. A no-fault system akin to automobile no-fault insurance.
B. A cap on noneconomic damages.
C. “Safe-harbor” immunity against medical negligence.
D. Health courts in place of the judge/jury system to adjudicate claims.
E. Promotion of laws that encourage apologies and error disclosures.
Answer: A. Under the current Republican administration, one can expect legislative efforts at federal tort reform, especially given that Thomas E. Price, MD, the new secretary of the Department of Health & Human Services, is an orthopedic surgeon who has spoken passionately about defensive medicine, damage caps, health tribunals, and practice guidelines. As a former House representative for Georgia, Dr. Price has introduced several tort reform bills, so it is likely that any omnibus federal law will incorporate some of his proposals.1
Over the decades, many states have gone ahead in enacting their own statutes while awaiting federal action. Iowa is the latest example. It recently passed legislation that included a noneconomic damages cap of $250,000, stronger expert witness standards, a certificate of merit in all medical liability lawsuits, and an expansion of its “candor” protections.2 Additional reforms in other states include pretrial screening panels; arbitration; structured periodic payments in lieu of lump sum payments; penalties for frivolous suits; shortened statutes of limitations; making the loser bear all litigation costs; abolishing the collateral source rule, as well as joint and several liability; and limiting attorney contingency fees.
The best-known reform is a cap on noneconomic losses, such as pain and suffering, that doesn’t abridge compensation for economic losses, i.e., medical expenses and lost wages. This provides some predictability because noneconomic damages are difficult to quantify, and jury sympathy may result in unrealistically high payments.
Interestingly, Dr. Price himself has not pushed for a federal cap on noneconomic damages, but other Republican bills have proposed a cap of $250,000. Many states, such as California, Kansas, and Texas, have seen their cap statutes withstand constitutional challenge. However, other jurisdictions, notably Georgia, Illinois, and Missouri, have ruled them unconstitutional.
California’s law, popularly known as MICRA (Medical Injury Compensation Reform Act), came under renewed attack in 2015 with a wrongful death suit from hemorrhagic complications related to Coumadin (warfarin) use following heart surgery.3 The plaintiff’s constitutional challenges included violation of equal protection, due process, and the right to a jury trial, but these were essentially all grounded on an entitlement to recover additional noneconomic damages sufficient to cover attorney fees. The trial court had reduced her $1 million noneconomic damages to $250,000, as required under MICRA. A California court of appeal rejected her claim as being “contrary to many well-established legal principles.”
On the other hand, Florida’s Supreme Court recently held in a closely divided decision of 4-3 that the state’s caps were unconstitutional.4 The law limited noneconomic damages in malpractice cases to either $500,000 or $1 million if the injuries were catastrophic. The court ruled that the caps were arbitrary and unfairly hurt the most severely injured. It was unconvinced that they would reduce malpractice insurance rates; at any rate, there was no present crisis to justify the caps. The decision came 3 years after the court had struck down caps in a case of wrongful death.5
Three relative newcomers to the legal landscape – health courts, apology laws, and safe harbors – appear to be taking center stage in any forthcoming federal reform measures.
Health courts
Under this proposal, so-called health panels and tribunals would now adjudicate malpractice claims. Such health courts would dispense with the jury; further, regular judges would be replaced with specialized judges who would make binding determinations. In one version, a panel of medical experts would initially screen the complaint, followed by an administrative health care tribunal that would feature judges with medical expertise. These tribunals would issue binding rulings, but either party could appeal to a state court for a reversal.
In countries such as Scandinavia and New Zealand, these administrative compensation approaches are coupled to a no-fault system and appear to work well. However, unlike auto no-fault and workers’ compensation, the notion of medical no-fault has never caught on in the United States.
As currently construed, health courts evince dramatic departures from traditional rules of civil procedure. For one, the panels may render decisions before discovery has occurred, which would substantially limit a patient’s ability to learn the facts of what had happened to cause the injury. The panel may rely on a standard of “gross negligence” instead of “ordinary negligence,” requiring evidence not merely of substandard care but of recklessness. This would be a heavier burden on the victim, and could be expected to generate stiff opposition from the plaintiff’s bar. In addition, evidentiary rules may be modified, requiring that an appeal show with clear and convincing proof that the tribunal had erred.
Apology law
Disclosure of medical errors to the injured patient is believed to serve as an ethical and effective way of thwarting potential malpractice claims. Many states have enacted so-called apology laws that disallow statements of sympathy from being admitted into evidence. In some cases, these laws may assist the physician.
For example, the Ohio Supreme Court ruled that a surgeon’s comments and alleged admission of guilt (“I take full responsibility for this” regarding accidentally sectioning the common bile duct) were properly shielded from discovery by the state’s apology statute.6 Apology laws vary from state to state, and some do not shield admissions regarding causation of error or fault.
However, it is unclear if apology laws work. A recent study from Vanderbilt University reported that, for physicians who do not regularly perform surgery, apology laws actually increased the probability of facing a lawsuit.7 And for surgeons, apology laws do not have a substantial effect on the probability of facing a claim or the average payment made to resolve a claim.
Safe harbors
A proposal released by U.S. House Speaker Paul Ryan (R-Wis.) in June 2016 made reference to “safe harbors” from liability for those adhering to clinical practice guidelines. The Institute of Medicine defines practice guidelines as “systematically developed statements to assist practitioner and patient decisions about appropriate health care for specific clinical circumstances.”
There are thousands of guidelines that have been developed by medical organizations and governmental agencies, as well as by insurance carriers, managed care organizations, and others. They purport to define the best evidence-based medicine, and if they were arrived at by the consensus of an authoritative body of experts, courts will tend to view them as reflective, though not necessarily dispositive, of customary medical standards.
Theoretically, adherence to guidelines could reduce the practice of defensive medicine and improve the quality of care. However, the available evidence does not indicate that guideline-based safe harbors will prove very effective in reducing malpractice claims: They are inapplicable in 85% of cases, and they have been estimated to eliminate defendants’ payments in less than 1% of claims.
Whether any form of tort reform emerges from the current Congress is as much about politics as it is about justice. It comes at an inopportune time, given the impasse over the health care debate. Still, on June 29, 2017, the U.S. House passed a medical liability reform bill with a vote of 218-210 along party lines that would cap noneconomic damages at $250,000, shorten the statute of limitations to 3 years after the date of injury, and abolish joint and several liability.8 The outlook in the U.S. Senate, however, is anything but certain.
References
1. N Engl J Med. 2017 May 11;376(19):1806-8.
2. “Sweeping new tort reforms will protect Iowa physicians” AMA Wire. June 1, 2017.
3. Chan v. Curran, 237 CA 4th 601 (2015).
4. N. Broward Hospital District v. Kalitan, (Florida Supreme Court, decided June 8, 2017).
5. Estate of Michelle Evette McCall v. U.S., 2014 Fla LEXIS 933 (No. SC 11-1148; March 13, 2014).
6. Estate of Johnson v. Randall Smith, Inc., 135 Ohio St.3d 440 (2013).
7. “Sorry is Never Enough: The Effect of State Apology Laws on Medical Malpractice Liability Risk” SSRN. 2016 Dec 10.
8. Protecting Access to Care Act of 2017 (H.R. 1215).
Question: Congressional proposals on medical tort reform can be expected to include the following, except:
A. A no-fault system akin to automobile no-fault insurance.
B. A cap on noneconomic damages.
C. “Safe-harbor” immunity against medical negligence.
D. Health courts in place of the judge/jury system to adjudicate claims.
E. Promotion of laws that encourage apologies and error disclosures.
Answer: A. Under the current Republican administration, one can expect legislative efforts at federal tort reform, especially given that Thomas E. Price, MD, the new secretary of the Department of Health & Human Services, is an orthopedic surgeon who has spoken passionately about defensive medicine, damage caps, health tribunals, and practice guidelines. As a former House representative for Georgia, Dr. Price has introduced several tort reform bills, so it is likely that any omnibus federal law will incorporate some of his proposals.1
Over the decades, many states have gone ahead in enacting their own statutes while awaiting federal action. Iowa is the latest example. It recently passed legislation that included a noneconomic damages cap of $250,000, stronger expert witness standards, a certificate of merit in all medical liability lawsuits, and an expansion of its “candor” protections.2 Additional reforms in other states include pretrial screening panels; arbitration; structured periodic payments in lieu of lump sum payments; penalties for frivolous suits; shortened statutes of limitations; making the loser bear all litigation costs; abolishing the collateral source rule, as well as joint and several liability; and limiting attorney contingency fees.
The best-known reform is a cap on noneconomic losses, such as pain and suffering, that doesn’t abridge compensation for economic losses, i.e., medical expenses and lost wages. This provides some predictability because noneconomic damages are difficult to quantify, and jury sympathy may result in unrealistically high payments.
Interestingly, Dr. Price himself has not pushed for a federal cap on noneconomic damages, but other Republican bills have proposed a cap of $250,000. Many states, such as California, Kansas, and Texas, have seen their cap statutes withstand constitutional challenge. However, other jurisdictions, notably Georgia, Illinois, and Missouri, have ruled them unconstitutional.
California’s law, popularly known as MICRA (Medical Injury Compensation Reform Act), came under renewed attack in 2015 with a wrongful death suit from hemorrhagic complications related to Coumadin (warfarin) use following heart surgery.3 The plaintiff’s constitutional challenges included violation of equal protection, due process, and the right to a jury trial, but these were essentially all grounded on an entitlement to recover additional noneconomic damages sufficient to cover attorney fees. The trial court had reduced her $1 million noneconomic damages to $250,000, as required under MICRA. A California court of appeal rejected her claim as being “contrary to many well-established legal principles.”
On the other hand, Florida’s Supreme Court recently held in a closely divided decision of 4-3 that the state’s caps were unconstitutional.4 The law limited noneconomic damages in malpractice cases to either $500,000 or $1 million if the injuries were catastrophic. The court ruled that the caps were arbitrary and unfairly hurt the most severely injured. It was unconvinced that they would reduce malpractice insurance rates; at any rate, there was no present crisis to justify the caps. The decision came 3 years after the court had struck down caps in a case of wrongful death.5
Three relative newcomers to the legal landscape – health courts, apology laws, and safe harbors – appear to be taking center stage in any forthcoming federal reform measures.
Health courts
Under this proposal, so-called health panels and tribunals would now adjudicate malpractice claims. Such health courts would dispense with the jury; further, regular judges would be replaced with specialized judges who would make binding determinations. In one version, a panel of medical experts would initially screen the complaint, followed by an administrative health care tribunal that would feature judges with medical expertise. These tribunals would issue binding rulings, but either party could appeal to a state court for a reversal.
In countries such as Scandinavia and New Zealand, these administrative compensation approaches are coupled to a no-fault system and appear to work well. However, unlike auto no-fault and workers’ compensation, the notion of medical no-fault has never caught on in the United States.
As currently construed, health courts evince dramatic departures from traditional rules of civil procedure. For one, the panels may render decisions before discovery has occurred, which would substantially limit a patient’s ability to learn the facts of what had happened to cause the injury. The panel may rely on a standard of “gross negligence” instead of “ordinary negligence,” requiring evidence not merely of substandard care but of recklessness. This would be a heavier burden on the victim, and could be expected to generate stiff opposition from the plaintiff’s bar. In addition, evidentiary rules may be modified, requiring that an appeal show with clear and convincing proof that the tribunal had erred.
Apology law
Disclosure of medical errors to the injured patient is believed to serve as an ethical and effective way of thwarting potential malpractice claims. Many states have enacted so-called apology laws that disallow statements of sympathy from being admitted into evidence. In some cases, these laws may assist the physician.
For example, the Ohio Supreme Court ruled that a surgeon’s comments and alleged admission of guilt (“I take full responsibility for this” regarding accidentally sectioning the common bile duct) were properly shielded from discovery by the state’s apology statute.6 Apology laws vary from state to state, and some do not shield admissions regarding causation of error or fault.
However, it is unclear if apology laws work. A recent study from Vanderbilt University reported that, for physicians who do not regularly perform surgery, apology laws actually increased the probability of facing a lawsuit.7 And for surgeons, apology laws do not have a substantial effect on the probability of facing a claim or the average payment made to resolve a claim.
Safe harbors
A proposal released by U.S. House Speaker Paul Ryan (R-Wis.) in June 2016 made reference to “safe harbors” from liability for those adhering to clinical practice guidelines. The Institute of Medicine defines practice guidelines as “systematically developed statements to assist practitioner and patient decisions about appropriate health care for specific clinical circumstances.”
There are thousands of guidelines that have been developed by medical organizations and governmental agencies, as well as by insurance carriers, managed care organizations, and others. They purport to define the best evidence-based medicine, and if they were arrived at by the consensus of an authoritative body of experts, courts will tend to view them as reflective, though not necessarily dispositive, of customary medical standards.
Theoretically, adherence to guidelines could reduce the practice of defensive medicine and improve the quality of care. However, the available evidence does not indicate that guideline-based safe harbors will prove very effective in reducing malpractice claims: They are inapplicable in 85% of cases, and they have been estimated to eliminate defendants’ payments in less than 1% of claims.
Whether any form of tort reform emerges from the current Congress is as much about politics as it is about justice. It comes at an inopportune time, given the impasse over the health care debate. Still, on June 29, 2017, the U.S. House passed a medical liability reform bill with a vote of 218-210 along party lines that would cap noneconomic damages at $250,000, shorten the statute of limitations to 3 years after the date of injury, and abolish joint and several liability.8 The outlook in the U.S. Senate, however, is anything but certain.
References
1. N Engl J Med. 2017 May 11;376(19):1806-8.
2. “Sweeping new tort reforms will protect Iowa physicians” AMA Wire. June 1, 2017.
3. Chan v. Curran, 237 CA 4th 601 (2015).
4. N. Broward Hospital District v. Kalitan, (Florida Supreme Court, decided June 8, 2017).
5. Estate of Michelle Evette McCall v. U.S., 2014 Fla LEXIS 933 (No. SC 11-1148; March 13, 2014).
6. Estate of Johnson v. Randall Smith, Inc., 135 Ohio St.3d 440 (2013).
7. “Sorry is Never Enough: The Effect of State Apology Laws on Medical Malpractice Liability Risk” SSRN. 2016 Dec 10.
8. Protecting Access to Care Act of 2017 (H.R. 1215).
Wide variability found in invasive mediastinal staging rates for lung cancer
COLORADO SPRINGS – Significant variability exists between hospitals in Washington state in their rates of invasive mediastinal staging for lung cancer, Farhood Farjah, MD, reported at the annual meeting of the Western Thoracic Surgical Association.
“We found evidence of a fivefold variation in hospital-level rates of invasive mediastinal staging not explained by chance or case mix,” according to Dr. Farjah of the University of Washington, Seattle.
“This has led to substantial concerns about quality of thoracic surgical care in the community at large,” he noted.
The Washington study is the first to show hospital-by-hospital variation in rates of invasive mediastinal staging.
Invasive mediastinal staging for lung cancer is considered important because imaging is known to have a substantial false-negative rate, and staging results have a profound impact on treatment recommendations, which can range from surgery alone to additional chemoradiation therapy.
Yet the meaning of the hospital-level huge variability in practice observed in the Washington study remains unclear.
“Our understanding of the underutilization of invasive mediastinal staging is further complicated by the fact that patterns of invasive mediastinal staging are highly variable across hospitals staffed by at least one board-certified thoracic surgeon with a noncardiac practice,” Dr. Farjah explained. “This variability could be a marker of poor-quality care. However, because the guidelines are not supported by level 1 evidence, it’s equally plausible that this variability might represent uncertainty or even disagreement with the practice guidelines – and specifically about the appropriate indication for invasive staging.”
He presented a retrospective cohort study of 406 patients whose non–small cell lung cancer was resected during July 2011–December 2013 at one of five Washington hospitals, each with at least one board-certified thoracic surgeon with a noncardiac practice on staff. The four participating community hospitals and one academic medical center were involved in a National Cancer Institute–funded, physician-led quality improvement initiative.
Overall, 66% of the 406 patients underwent any form of invasive mediastinal staging: 85% by mediastinoscopy only; 12% by mediastinoscopy plus endobronchial ultrasound-guided nodal aspiration (EBUS); 3% by EBUS only; and the remaining handful by mediastinoscopy, EBUS, and esophageal ultrasound-guided nodal aspiration. The invasive staging was performed at the time of resection in 64% of cases. A median of three nodal stations were sampled.
After statistical adjustment for random variation and between-hospital differences in clinical stage, rates of invasive staging were all over the map. While an overall mean of 66% of the lung cancer patients underwent invasive mediastinal staging, the rates at the five hospitals were 94%, 84%, 31%, 80%, and 17%.
Dr. Farjah and his coinvestigators are now conducting provider interviews and focus groups in an effort to understand what drove the participating surgeons’ wide variability in performing invasive mediastinal staging.
Discussant Jane Yanagawa, MD, of the University of California, Los Angeles, commented, “I think this is a really interesting study because, historically, lower rates of mediastinoscopy are assumed to be a reflection of low-quality care – and you suggest that might not be the case, that it might be more complicated than that.”
Dr. Yanagawa sketched one fairly common scenario that might represent a surgeon’s reasonable avoidance of guideline-recommended invasive mediastinal staging: a patient who by all preoperative imaging appears to have stage IA lung cancer and wishes to avoid the morbidity, time, and cost of needle biopsy, instead choosing to go straight to the operating room for a diagnosis by wedge resection, followed by a completion lobectomy based upon the frozen section results. Could such a pathway account for the variability seen in the Washington study?
“I think it could have,” Dr. Farjah replied. “I would say that’s probably one driver of variability.”
As for the generalizability of the findings of a five-hospital study carried out in a single state, Dr. Farjah said he thinks the results are applicable to any academic or community hospital with at least one board-certified thoracic surgeon with a noncardiac practice.
He reported having no financial conflicts of interest regarding the study.
M. Patricia Rivera, MD, FCCP, comments: Staging of lung cancer is essential to select the best treatment strategy for a given patient. However, despite multiple guideline recommendations
M. Patricia Rivera, MD, FCCP, comments: Staging of lung cancer is essential to select the best treatment strategy for a given patient. However, despite multiple guideline recommendations
M. Patricia Rivera, MD, FCCP, comments: Staging of lung cancer is essential to select the best treatment strategy for a given patient. However, despite multiple guideline recommendations
COLORADO SPRINGS – Significant variability exists between hospitals in Washington state in their rates of invasive mediastinal staging for lung cancer, Farhood Farjah, MD, reported at the annual meeting of the Western Thoracic Surgical Association.
“We found evidence of a fivefold variation in hospital-level rates of invasive mediastinal staging not explained by chance or case mix,” according to Dr. Farjah of the University of Washington, Seattle.
“This has led to substantial concerns about quality of thoracic surgical care in the community at large,” he noted.
The Washington study is the first to show hospital-by-hospital variation in rates of invasive mediastinal staging.
Invasive mediastinal staging for lung cancer is considered important because imaging is known to have a substantial false-negative rate, and staging results have a profound impact on treatment recommendations, which can range from surgery alone to additional chemoradiation therapy.
Yet the meaning of the hospital-level huge variability in practice observed in the Washington study remains unclear.
“Our understanding of the underutilization of invasive mediastinal staging is further complicated by the fact that patterns of invasive mediastinal staging are highly variable across hospitals staffed by at least one board-certified thoracic surgeon with a noncardiac practice,” Dr. Farjah explained. “This variability could be a marker of poor-quality care. However, because the guidelines are not supported by level 1 evidence, it’s equally plausible that this variability might represent uncertainty or even disagreement with the practice guidelines – and specifically about the appropriate indication for invasive staging.”
He presented a retrospective cohort study of 406 patients whose non–small cell lung cancer was resected during July 2011–December 2013 at one of five Washington hospitals, each with at least one board-certified thoracic surgeon with a noncardiac practice on staff. The four participating community hospitals and one academic medical center were involved in a National Cancer Institute–funded, physician-led quality improvement initiative.
Overall, 66% of the 406 patients underwent any form of invasive mediastinal staging: 85% by mediastinoscopy only; 12% by mediastinoscopy plus endobronchial ultrasound-guided nodal aspiration (EBUS); 3% by EBUS only; and the remaining handful by mediastinoscopy, EBUS, and esophageal ultrasound-guided nodal aspiration. The invasive staging was performed at the time of resection in 64% of cases. A median of three nodal stations were sampled.
After statistical adjustment for random variation and between-hospital differences in clinical stage, rates of invasive staging were all over the map. While an overall mean of 66% of the lung cancer patients underwent invasive mediastinal staging, the rates at the five hospitals were 94%, 84%, 31%, 80%, and 17%.
Dr. Farjah and his coinvestigators are now conducting provider interviews and focus groups in an effort to understand what drove the participating surgeons’ wide variability in performing invasive mediastinal staging.
Discussant Jane Yanagawa, MD, of the University of California, Los Angeles, commented, “I think this is a really interesting study because, historically, lower rates of mediastinoscopy are assumed to be a reflection of low-quality care – and you suggest that might not be the case, that it might be more complicated than that.”
Dr. Yanagawa sketched one fairly common scenario that might represent a surgeon’s reasonable avoidance of guideline-recommended invasive mediastinal staging: a patient who by all preoperative imaging appears to have stage IA lung cancer and wishes to avoid the morbidity, time, and cost of needle biopsy, instead choosing to go straight to the operating room for a diagnosis by wedge resection, followed by a completion lobectomy based upon the frozen section results. Could such a pathway account for the variability seen in the Washington study?
“I think it could have,” Dr. Farjah replied. “I would say that’s probably one driver of variability.”
As for the generalizability of the findings of a five-hospital study carried out in a single state, Dr. Farjah said he thinks the results are applicable to any academic or community hospital with at least one board-certified thoracic surgeon with a noncardiac practice.
He reported having no financial conflicts of interest regarding the study.
COLORADO SPRINGS – Significant variability exists between hospitals in Washington state in their rates of invasive mediastinal staging for lung cancer, Farhood Farjah, MD, reported at the annual meeting of the Western Thoracic Surgical Association.
“We found evidence of a fivefold variation in hospital-level rates of invasive mediastinal staging not explained by chance or case mix,” according to Dr. Farjah of the University of Washington, Seattle.
“This has led to substantial concerns about quality of thoracic surgical care in the community at large,” he noted.
The Washington study is the first to show hospital-by-hospital variation in rates of invasive mediastinal staging.
Invasive mediastinal staging for lung cancer is considered important because imaging is known to have a substantial false-negative rate, and staging results have a profound impact on treatment recommendations, which can range from surgery alone to additional chemoradiation therapy.
Yet the meaning of the hospital-level huge variability in practice observed in the Washington study remains unclear.
“Our understanding of the underutilization of invasive mediastinal staging is further complicated by the fact that patterns of invasive mediastinal staging are highly variable across hospitals staffed by at least one board-certified thoracic surgeon with a noncardiac practice,” Dr. Farjah explained. “This variability could be a marker of poor-quality care. However, because the guidelines are not supported by level 1 evidence, it’s equally plausible that this variability might represent uncertainty or even disagreement with the practice guidelines – and specifically about the appropriate indication for invasive staging.”
He presented a retrospective cohort study of 406 patients whose non–small cell lung cancer was resected during July 2011–December 2013 at one of five Washington hospitals, each with at least one board-certified thoracic surgeon with a noncardiac practice on staff. The four participating community hospitals and one academic medical center were involved in a National Cancer Institute–funded, physician-led quality improvement initiative.
Overall, 66% of the 406 patients underwent any form of invasive mediastinal staging: 85% by mediastinoscopy only; 12% by mediastinoscopy plus endobronchial ultrasound-guided nodal aspiration (EBUS); 3% by EBUS only; and the remaining handful by mediastinoscopy, EBUS, and esophageal ultrasound-guided nodal aspiration. The invasive staging was performed at the time of resection in 64% of cases. A median of three nodal stations were sampled.
After statistical adjustment for random variation and between-hospital differences in clinical stage, rates of invasive staging were all over the map. While an overall mean of 66% of the lung cancer patients underwent invasive mediastinal staging, the rates at the five hospitals were 94%, 84%, 31%, 80%, and 17%.
Dr. Farjah and his coinvestigators are now conducting provider interviews and focus groups in an effort to understand what drove the participating surgeons’ wide variability in performing invasive mediastinal staging.
Discussant Jane Yanagawa, MD, of the University of California, Los Angeles, commented, “I think this is a really interesting study because, historically, lower rates of mediastinoscopy are assumed to be a reflection of low-quality care – and you suggest that might not be the case, that it might be more complicated than that.”
Dr. Yanagawa sketched one fairly common scenario that might represent a surgeon’s reasonable avoidance of guideline-recommended invasive mediastinal staging: a patient who by all preoperative imaging appears to have stage IA lung cancer and wishes to avoid the morbidity, time, and cost of needle biopsy, instead choosing to go straight to the operating room for a diagnosis by wedge resection, followed by a completion lobectomy based upon the frozen section results. Could such a pathway account for the variability seen in the Washington study?
“I think it could have,” Dr. Farjah replied. “I would say that’s probably one driver of variability.”
As for the generalizability of the findings of a five-hospital study carried out in a single state, Dr. Farjah said he thinks the results are applicable to any academic or community hospital with at least one board-certified thoracic surgeon with a noncardiac practice.
He reported having no financial conflicts of interest regarding the study.
AT THE WTSA ANNUAL MEETING
Key clinical point:
Major finding: Rates of invasive mediastinal staging after adjustment for clinical stage ranged from a low of 17% at one hospital to as high as 94% at another.
Data source: This retrospective cohort study included 406 patients.
Disclosures: Dr. Farjah reported having no financial conflicts of interest.
Bailout stenting for coronary bifurcations brings ‘unacceptable’ hazards
PARIS – Bailout stenting during percutaneous coronary intervention for coronary bifurcations doubled the risk of major adverse cardiovascular events in the world’s largest registry of patients with these often-challenging lesions treated using bioactive stents, Marco Zimarino, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
Indeed, resort to bailout stenting stood out as the major potentially modifiable risk factor for adverse outcomes among the 4,306 participants in the P2BiTO registry, an international collaboration supported by members of the EuroBifurcation Club. Most of the other independent risk factors identified in a multivariate regression analysis of the P2BiTO database were beyond operator control, including diabetes, advanced age, and presentation with an acute coronary syndrome, according to Dr. Zimarino of the University of Chieti (Italy).
Bailout stenting is largely avoidable through meticulous procedural planning, the interventional cardiologist added.
“Careful planning is always mandatory because bailout stenting is associated with an unacceptably higher risk of both in-hospital and 1-year adverse outcomes,” Dr. Zimarino emphasized. “It’s much better to leave a degraded side branch instead of using bailout stenting to get an excellent angiographic outcome that’s a predictor of a worse clinical outcome.”
Conventional wisdom holds that single stenting of either the main artery or a side branch in a patient with coronary bifurcation is safer than double stenting of both. However, that wasn’t really borne out in the P2BiTO registry provided the operator’s plan was for double stenting. The difference in 1-year major adverse cardiovascular events (MACE) between patients treated using a single- or double-stenting strategy wasn’t statistically significant, provided bailout stenting wasn’t utilized. If bailout stenting was employed, though, the risk of MACE was 2.2-fold greater than if the cardiologist stuck with the plan.
Ninety-eight percent of patients in the P2BiTO registry received drug-eluting stents. The other 2% got the Absorb bioabsorbable vascular scaffold. The percutaneous coronary intervention access site, treatment strategy, choice of stent, and duration of dual-antiplatelet therapy were left up to the operator’s discretion.
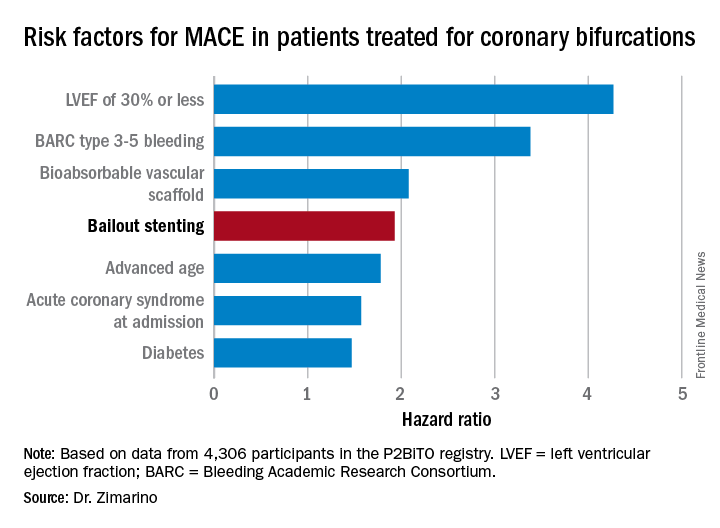
The risk of MACE was reduced by 39% in patients on dual-antiplatelet therapy for 6-12 months, compared with less than 6 months.
Discussant Graham Cassel, MD, director of the heart transplant unit at Milpark Hospital in Johannesburg, commented, “The message comes through very clearly that, if you plan your procedure well, the chance of bailout is far less – and if you do have to bail out, the results are uniformly bad. If you can avoid putting in two or three stents, that’s beneficial.”
Dr. Zimarino reported having no financial conflicts of interest regarding his presentation.
PARIS – Bailout stenting during percutaneous coronary intervention for coronary bifurcations doubled the risk of major adverse cardiovascular events in the world’s largest registry of patients with these often-challenging lesions treated using bioactive stents, Marco Zimarino, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
Indeed, resort to bailout stenting stood out as the major potentially modifiable risk factor for adverse outcomes among the 4,306 participants in the P2BiTO registry, an international collaboration supported by members of the EuroBifurcation Club. Most of the other independent risk factors identified in a multivariate regression analysis of the P2BiTO database were beyond operator control, including diabetes, advanced age, and presentation with an acute coronary syndrome, according to Dr. Zimarino of the University of Chieti (Italy).
Bailout stenting is largely avoidable through meticulous procedural planning, the interventional cardiologist added.
“Careful planning is always mandatory because bailout stenting is associated with an unacceptably higher risk of both in-hospital and 1-year adverse outcomes,” Dr. Zimarino emphasized. “It’s much better to leave a degraded side branch instead of using bailout stenting to get an excellent angiographic outcome that’s a predictor of a worse clinical outcome.”
Conventional wisdom holds that single stenting of either the main artery or a side branch in a patient with coronary bifurcation is safer than double stenting of both. However, that wasn’t really borne out in the P2BiTO registry provided the operator’s plan was for double stenting. The difference in 1-year major adverse cardiovascular events (MACE) between patients treated using a single- or double-stenting strategy wasn’t statistically significant, provided bailout stenting wasn’t utilized. If bailout stenting was employed, though, the risk of MACE was 2.2-fold greater than if the cardiologist stuck with the plan.
Ninety-eight percent of patients in the P2BiTO registry received drug-eluting stents. The other 2% got the Absorb bioabsorbable vascular scaffold. The percutaneous coronary intervention access site, treatment strategy, choice of stent, and duration of dual-antiplatelet therapy were left up to the operator’s discretion.

The risk of MACE was reduced by 39% in patients on dual-antiplatelet therapy for 6-12 months, compared with less than 6 months.
Discussant Graham Cassel, MD, director of the heart transplant unit at Milpark Hospital in Johannesburg, commented, “The message comes through very clearly that, if you plan your procedure well, the chance of bailout is far less – and if you do have to bail out, the results are uniformly bad. If you can avoid putting in two or three stents, that’s beneficial.”
Dr. Zimarino reported having no financial conflicts of interest regarding his presentation.
PARIS – Bailout stenting during percutaneous coronary intervention for coronary bifurcations doubled the risk of major adverse cardiovascular events in the world’s largest registry of patients with these often-challenging lesions treated using bioactive stents, Marco Zimarino, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
Indeed, resort to bailout stenting stood out as the major potentially modifiable risk factor for adverse outcomes among the 4,306 participants in the P2BiTO registry, an international collaboration supported by members of the EuroBifurcation Club. Most of the other independent risk factors identified in a multivariate regression analysis of the P2BiTO database were beyond operator control, including diabetes, advanced age, and presentation with an acute coronary syndrome, according to Dr. Zimarino of the University of Chieti (Italy).
Bailout stenting is largely avoidable through meticulous procedural planning, the interventional cardiologist added.
“Careful planning is always mandatory because bailout stenting is associated with an unacceptably higher risk of both in-hospital and 1-year adverse outcomes,” Dr. Zimarino emphasized. “It’s much better to leave a degraded side branch instead of using bailout stenting to get an excellent angiographic outcome that’s a predictor of a worse clinical outcome.”
Conventional wisdom holds that single stenting of either the main artery or a side branch in a patient with coronary bifurcation is safer than double stenting of both. However, that wasn’t really borne out in the P2BiTO registry provided the operator’s plan was for double stenting. The difference in 1-year major adverse cardiovascular events (MACE) between patients treated using a single- or double-stenting strategy wasn’t statistically significant, provided bailout stenting wasn’t utilized. If bailout stenting was employed, though, the risk of MACE was 2.2-fold greater than if the cardiologist stuck with the plan.
Ninety-eight percent of patients in the P2BiTO registry received drug-eluting stents. The other 2% got the Absorb bioabsorbable vascular scaffold. The percutaneous coronary intervention access site, treatment strategy, choice of stent, and duration of dual-antiplatelet therapy were left up to the operator’s discretion.

The risk of MACE was reduced by 39% in patients on dual-antiplatelet therapy for 6-12 months, compared with less than 6 months.
Discussant Graham Cassel, MD, director of the heart transplant unit at Milpark Hospital in Johannesburg, commented, “The message comes through very clearly that, if you plan your procedure well, the chance of bailout is far less – and if you do have to bail out, the results are uniformly bad. If you can avoid putting in two or three stents, that’s beneficial.”
Dr. Zimarino reported having no financial conflicts of interest regarding his presentation.
AT EUROPCR
Key clinical point:
Major finding: Bailout stenting during PCI for coronary bifurcations doubles the risk of major adverse cardiovascular events.
Data source: The P2BiTO registry includes 4,306 patients who received one or more drug-eluting stents or bioabsorbable vascular scaffolds for treatment of coronary bifurcations.
Disclosures: The study presenter reported having no financial conflicts of interest.
Less than half of office visits involve primary care
, according to the National Center for Health Statistics.
Primary care physicians’ share of office visits fell from 66.2% in 1980 to 49.1% in 2013, the NCHS reported in “Health, United States, 2016.” The corresponding increase among specialty care physicians gave them a total of 50.9% of all office visits in 2013, up from 33.8% in 1980.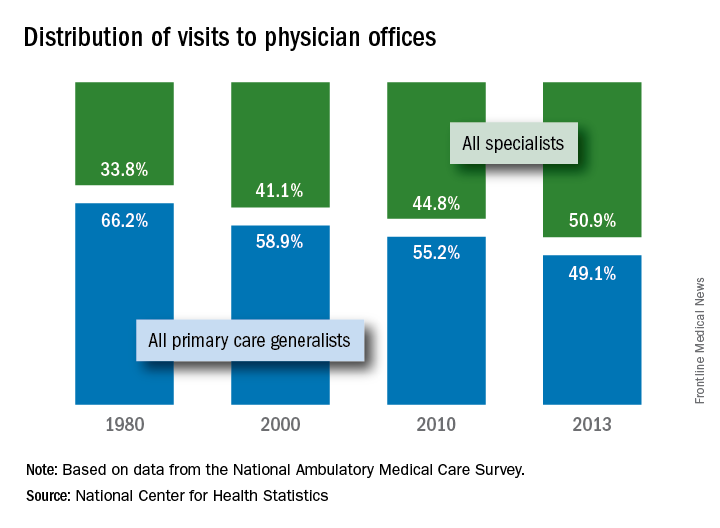
The NCHS estimates are based on data collected by the National Ambulatory Medical Care Survey, which excluded Alaska and Hawaii in 1980.
, according to the National Center for Health Statistics.
Primary care physicians’ share of office visits fell from 66.2% in 1980 to 49.1% in 2013, the NCHS reported in “Health, United States, 2016.” The corresponding increase among specialty care physicians gave them a total of 50.9% of all office visits in 2013, up from 33.8% in 1980.
The NCHS estimates are based on data collected by the National Ambulatory Medical Care Survey, which excluded Alaska and Hawaii in 1980.
, according to the National Center for Health Statistics.
Primary care physicians’ share of office visits fell from 66.2% in 1980 to 49.1% in 2013, the NCHS reported in “Health, United States, 2016.” The corresponding increase among specialty care physicians gave them a total of 50.9% of all office visits in 2013, up from 33.8% in 1980.
The NCHS estimates are based on data collected by the National Ambulatory Medical Care Survey, which excluded Alaska and Hawaii in 1980.