User login
Official Newspaper of the American College of Surgeons
Sodium fusidate noninferior to linezolid for acute skin infections
NEW ORLEANS – An oral antibiotic in development in the United States, fusidic acid (oral formulation, sodium fusidate) was noninferior to linezolid based on early clinical response in a randomized, double-blind, multicenter trial of 716 people with acute bacterial skin and skin structure infections (ABSSSI), including cellulitis, wound infection, and major cutaneous abscesses.
Early clinical response was defined as a 20% or greater reduction from baseline in the surface area of redness, edema, or induration at 48-72 hours after starting treatment with the study drugs. In an intent-to-treat analysis, 87.2% of patients randomized to fusidic acid and 86.6% of the linezolid group met this primary endpoint of the phase 3 study.
“Fusidic acid showed similar efficacy and comparable safety” that persisted through treatment, said Andy Strayer, PharmD, vice president of clinical programs at Cempra Pharmaceuticals, which is developing sodium fusidate as an oral agent to treat ABSSSI patients in the United States. Leo Pharmaceuticals has marketed sodium fusidate outside the United States in various formulations for decades.
Fusidic acid has potent activity against gram-positive aerobic organisms, including methicillin-resistant Staphylococcus aureus (MRSA). “Strikingly, fusidic acid showed 100% success in patients with MRSA in the microbiologically evaluable population at the end of treatment and posttherapy evaluation time points,” Dr. Strayer said at the annual meeting of the American Society for Microbiology. “Fusidic acid may offer an important oral therapy alternative for MRSA infection.”
“Fusidic acid, a drug long used in other parts of the world, has been demonstrated in this first phase 3 trial, to be a potential new option for the treatment of MRSA skin and skin structure infections in the U.S.,” said Carrie Cardenas, MD, lead study author and a principal investigator at eStudySite, San Diego, and an internist in private practice in La Mesa, California.
There was a microbiological diagnosis established in 75% of patients. S. aureus was the most commonly detected pathogen (422 patients; 59%), and the study included 235 patients diagnosed with MRSA infection.
About two-thirds, 65%, of participants were men. Mean age was 45 years. Infections were classified as wounds in 61%, cellulitis in 26%, and abscess in 13%. Notably, 68% of the recruited participants had ABSSSI associated with intravenous drug use, a “sometimes overlooked consequence of the ongoing epidemic of IV drug use in the U.S.,” Dr. Strayer said.
In terms of safety, treatment-emergent adverse event rates were comparable between the two groups (37.9% with fusidic acid versus 36.1% with linezolid). Gastrointestinal events were the most common adverse events, 22.8% versus 18.2%, respectively.
“Considering complicated skin infections are one of the most rapidly growing reasons for hospitalizations and emergency department visits each year, we anticipate that fusidic acid, if approved, may help clinicians decrease the length of inpatient stay or avoid hospitalization altogether,” Dr. Strayer said.
Cempra sponsored the study. Dr. Strayer is a Cempra employee and shareholder. Dr. Carrie Cardenas is a principal investigator at eStudySite, San Diego, and performs research for Cempra, Paratek, Debiopharm, Motif, Durata, MicuRx, Bristol-Myers Squibb, and Bayer.
NEW ORLEANS – An oral antibiotic in development in the United States, fusidic acid (oral formulation, sodium fusidate) was noninferior to linezolid based on early clinical response in a randomized, double-blind, multicenter trial of 716 people with acute bacterial skin and skin structure infections (ABSSSI), including cellulitis, wound infection, and major cutaneous abscesses.
Early clinical response was defined as a 20% or greater reduction from baseline in the surface area of redness, edema, or induration at 48-72 hours after starting treatment with the study drugs. In an intent-to-treat analysis, 87.2% of patients randomized to fusidic acid and 86.6% of the linezolid group met this primary endpoint of the phase 3 study.
“Fusidic acid showed similar efficacy and comparable safety” that persisted through treatment, said Andy Strayer, PharmD, vice president of clinical programs at Cempra Pharmaceuticals, which is developing sodium fusidate as an oral agent to treat ABSSSI patients in the United States. Leo Pharmaceuticals has marketed sodium fusidate outside the United States in various formulations for decades.
Fusidic acid has potent activity against gram-positive aerobic organisms, including methicillin-resistant Staphylococcus aureus (MRSA). “Strikingly, fusidic acid showed 100% success in patients with MRSA in the microbiologically evaluable population at the end of treatment and posttherapy evaluation time points,” Dr. Strayer said at the annual meeting of the American Society for Microbiology. “Fusidic acid may offer an important oral therapy alternative for MRSA infection.”
“Fusidic acid, a drug long used in other parts of the world, has been demonstrated in this first phase 3 trial, to be a potential new option for the treatment of MRSA skin and skin structure infections in the U.S.,” said Carrie Cardenas, MD, lead study author and a principal investigator at eStudySite, San Diego, and an internist in private practice in La Mesa, California.
There was a microbiological diagnosis established in 75% of patients. S. aureus was the most commonly detected pathogen (422 patients; 59%), and the study included 235 patients diagnosed with MRSA infection.
About two-thirds, 65%, of participants were men. Mean age was 45 years. Infections were classified as wounds in 61%, cellulitis in 26%, and abscess in 13%. Notably, 68% of the recruited participants had ABSSSI associated with intravenous drug use, a “sometimes overlooked consequence of the ongoing epidemic of IV drug use in the U.S.,” Dr. Strayer said.
In terms of safety, treatment-emergent adverse event rates were comparable between the two groups (37.9% with fusidic acid versus 36.1% with linezolid). Gastrointestinal events were the most common adverse events, 22.8% versus 18.2%, respectively.
“Considering complicated skin infections are one of the most rapidly growing reasons for hospitalizations and emergency department visits each year, we anticipate that fusidic acid, if approved, may help clinicians decrease the length of inpatient stay or avoid hospitalization altogether,” Dr. Strayer said.
Cempra sponsored the study. Dr. Strayer is a Cempra employee and shareholder. Dr. Carrie Cardenas is a principal investigator at eStudySite, San Diego, and performs research for Cempra, Paratek, Debiopharm, Motif, Durata, MicuRx, Bristol-Myers Squibb, and Bayer.
NEW ORLEANS – An oral antibiotic in development in the United States, fusidic acid (oral formulation, sodium fusidate) was noninferior to linezolid based on early clinical response in a randomized, double-blind, multicenter trial of 716 people with acute bacterial skin and skin structure infections (ABSSSI), including cellulitis, wound infection, and major cutaneous abscesses.
Early clinical response was defined as a 20% or greater reduction from baseline in the surface area of redness, edema, or induration at 48-72 hours after starting treatment with the study drugs. In an intent-to-treat analysis, 87.2% of patients randomized to fusidic acid and 86.6% of the linezolid group met this primary endpoint of the phase 3 study.
“Fusidic acid showed similar efficacy and comparable safety” that persisted through treatment, said Andy Strayer, PharmD, vice president of clinical programs at Cempra Pharmaceuticals, which is developing sodium fusidate as an oral agent to treat ABSSSI patients in the United States. Leo Pharmaceuticals has marketed sodium fusidate outside the United States in various formulations for decades.
Fusidic acid has potent activity against gram-positive aerobic organisms, including methicillin-resistant Staphylococcus aureus (MRSA). “Strikingly, fusidic acid showed 100% success in patients with MRSA in the microbiologically evaluable population at the end of treatment and posttherapy evaluation time points,” Dr. Strayer said at the annual meeting of the American Society for Microbiology. “Fusidic acid may offer an important oral therapy alternative for MRSA infection.”
“Fusidic acid, a drug long used in other parts of the world, has been demonstrated in this first phase 3 trial, to be a potential new option for the treatment of MRSA skin and skin structure infections in the U.S.,” said Carrie Cardenas, MD, lead study author and a principal investigator at eStudySite, San Diego, and an internist in private practice in La Mesa, California.
There was a microbiological diagnosis established in 75% of patients. S. aureus was the most commonly detected pathogen (422 patients; 59%), and the study included 235 patients diagnosed with MRSA infection.
About two-thirds, 65%, of participants were men. Mean age was 45 years. Infections were classified as wounds in 61%, cellulitis in 26%, and abscess in 13%. Notably, 68% of the recruited participants had ABSSSI associated with intravenous drug use, a “sometimes overlooked consequence of the ongoing epidemic of IV drug use in the U.S.,” Dr. Strayer said.
In terms of safety, treatment-emergent adverse event rates were comparable between the two groups (37.9% with fusidic acid versus 36.1% with linezolid). Gastrointestinal events were the most common adverse events, 22.8% versus 18.2%, respectively.
“Considering complicated skin infections are one of the most rapidly growing reasons for hospitalizations and emergency department visits each year, we anticipate that fusidic acid, if approved, may help clinicians decrease the length of inpatient stay or avoid hospitalization altogether,” Dr. Strayer said.
Cempra sponsored the study. Dr. Strayer is a Cempra employee and shareholder. Dr. Carrie Cardenas is a principal investigator at eStudySite, San Diego, and performs research for Cempra, Paratek, Debiopharm, Motif, Durata, MicuRx, Bristol-Myers Squibb, and Bayer.
AT ASM MICROBE 2017
Key clinical point: Sodium fusidate, active as fusidic acid, showed noninferiority to linezolid for early clinical response in ABSSI patients.
Major finding: 87.2% of patients given sodium fusidate and 86.6% of those receiving linezolid achieved an early clinical response.
Data source: Randomized, controlled, double-blind, phase 3 study with 716 participants.
Disclosures: Cempra sponsored the study. Dr. Carrier Cardenas is a researcher for Cempra, Paratek, Debiopharm, Motif, Durata, MicuRx, Bristol-Myers Squibb, and Bayer. Dr. Strayer is a Cempra employee and shareholder.
Hidden CABG costs will disrupt bundled payment systems
COLORADO SPRINGS – With bundled payment models for coronary artery bypass graft surgery looming ahead, it’s vital that cardiac surgeons take a hard look at the procedure’s hidden costs – namely, the steep price tag for postoperative complications, James H. Mehaffey, MD, said at the annual meeting of the Western Thoracic Surgical Association.
He presented a retrospective study of the 30-day hospital costs for all 36,588 patients who underwent isolated CABG during 2006-2015 at the 19 Virginia centers where the surgery is performed. This was a typical CABG population, with an average predicted risk of mortality of 1.9%. The actual 30-day mortality was 0.6%, so the surgical performance was better than expected.
“The population of patients experiencing one or more major comorbidities demonstrated a significant and dramatic increase in total hospital costs. It was an exponential increase with each additional major morbidity,” reported Dr. Mehaffey of the University of Virginia, Charlottesville.
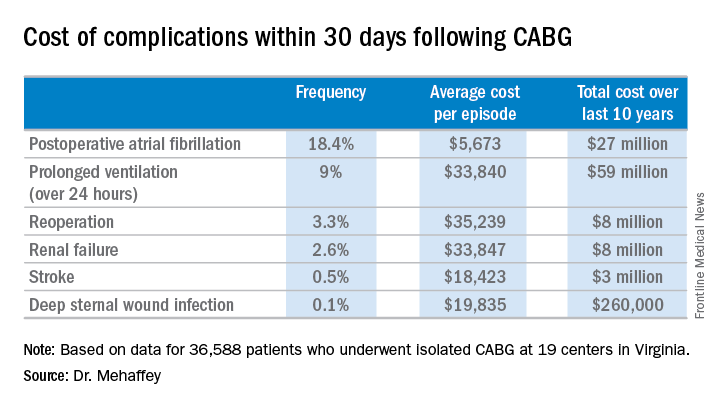
Indeed, the average cost jumped from $36,580 for uncomplicated surgery to $64,542 with one major complication, $111,239 with two, and $194,043 with three.
The two most frequent major complications were postoperative atrial fibrillation, which occurred in 18.4% of patients, and prolonged ventilation for longer than 24 hours, which occurred in 9%. Over the course of the decade-long study period, the 19 medical centers in the Virginia Cardiac Surgery Quality Initiative collectively spent roughly $59 million on prolonged ventilation and $27 million for postoperative atrial fibrillation.
The cost of CABG during the study years outpaced the CMS health care–specific inflation rate, and this escalating cost was driven primarily by postoperative complications.
For the Virginia cardiac surgery collaborative, these data on the cost of postoperative complications will be utilized to prioritize quality improvement projects.
For example, during the past decade, the Virginia collaborative made reduction in the rate of postoperative atrial fibrillation a priority. Toward that end, the collaborative developed a protocol for routine perioperative prophylactic amiodarone therapy.
“At the beginning of the study decade we had postoperative atrial fibrillation rates above 25%. The average for the entire decade was just over 18%, and in the last couple years we’ve been in the 15%-16% range. So I think we are moving the needle on this. We are making a meaningful impact,” Dr. Mehaffey said.
“We’ve already used the complication cost data to do a cost-effectiveness analysis of our prophylactic amiodarone innovation. We showed we saved an average of $250 per patient, even though we’re treating a bunch of patients who’d never get that complication,” he continued.
This sort of data on the cost of adverse events is also critical to accurately risk-adjust bundled payment models.
Discussant Richard J. Shemin, MD, asked if there was much variability in postoperative complication costs between the CABG centers in the Virginia collaborative.
The variability is enormous, Dr. Mehaffey replied. Investigators recently plugged the last 5 years worth of hospital cost and complication rate data into a proposed CABG bundled payment model and extrapolated what that would mean over the next 5 years.
“There were some institutions that would be positive by a couple million dollars from this payment system and some that were losing more than $20 million, just because of the cost variability,” said Dr. Mehaffey.
Dr. Shemin also noted that the Virginia collaborative was able to collect 30-day outcome data only through the STS database, yet the bundled payment programs are based on the 90-day postoperative experience.
“How do we capture the costs in that full 90 days that we’ll be responsible for?” asked Dr. Shemin, professor of surgery and codirector of the UCLA Cardiovascular Center.
Dr. Mehaffey said that’s indeed an important question, since a major complication such as stroke or deep sternal wound infection typically entails considerable long-term costs and repeated hospital admissions beyond the 30-day window. In Virginia, the cardiac surgery collaborative is working with payers to gain access to the 90 days worth of patient data.
He reported having no financial conflicts regarding his study.
COLORADO SPRINGS – With bundled payment models for coronary artery bypass graft surgery looming ahead, it’s vital that cardiac surgeons take a hard look at the procedure’s hidden costs – namely, the steep price tag for postoperative complications, James H. Mehaffey, MD, said at the annual meeting of the Western Thoracic Surgical Association.
He presented a retrospective study of the 30-day hospital costs for all 36,588 patients who underwent isolated CABG during 2006-2015 at the 19 Virginia centers where the surgery is performed. This was a typical CABG population, with an average predicted risk of mortality of 1.9%. The actual 30-day mortality was 0.6%, so the surgical performance was better than expected.
“The population of patients experiencing one or more major comorbidities demonstrated a significant and dramatic increase in total hospital costs. It was an exponential increase with each additional major morbidity,” reported Dr. Mehaffey of the University of Virginia, Charlottesville.
Indeed, the average cost jumped from $36,580 for uncomplicated surgery to $64,542 with one major complication, $111,239 with two, and $194,043 with three.
The two most frequent major complications were postoperative atrial fibrillation, which occurred in 18.4% of patients, and prolonged ventilation for longer than 24 hours, which occurred in 9%. Over the course of the decade-long study period, the 19 medical centers in the Virginia Cardiac Surgery Quality Initiative collectively spent roughly $59 million on prolonged ventilation and $27 million for postoperative atrial fibrillation.
The cost of CABG during the study years outpaced the CMS health care–specific inflation rate, and this escalating cost was driven primarily by postoperative complications.
For the Virginia cardiac surgery collaborative, these data on the cost of postoperative complications will be utilized to prioritize quality improvement projects.

For example, during the past decade, the Virginia collaborative made reduction in the rate of postoperative atrial fibrillation a priority. Toward that end, the collaborative developed a protocol for routine perioperative prophylactic amiodarone therapy.
“At the beginning of the study decade we had postoperative atrial fibrillation rates above 25%. The average for the entire decade was just over 18%, and in the last couple years we’ve been in the 15%-16% range. So I think we are moving the needle on this. We are making a meaningful impact,” Dr. Mehaffey said.
“We’ve already used the complication cost data to do a cost-effectiveness analysis of our prophylactic amiodarone innovation. We showed we saved an average of $250 per patient, even though we’re treating a bunch of patients who’d never get that complication,” he continued.
This sort of data on the cost of adverse events is also critical to accurately risk-adjust bundled payment models.
Discussant Richard J. Shemin, MD, asked if there was much variability in postoperative complication costs between the CABG centers in the Virginia collaborative.
The variability is enormous, Dr. Mehaffey replied. Investigators recently plugged the last 5 years worth of hospital cost and complication rate data into a proposed CABG bundled payment model and extrapolated what that would mean over the next 5 years.
“There were some institutions that would be positive by a couple million dollars from this payment system and some that were losing more than $20 million, just because of the cost variability,” said Dr. Mehaffey.
Dr. Shemin also noted that the Virginia collaborative was able to collect 30-day outcome data only through the STS database, yet the bundled payment programs are based on the 90-day postoperative experience.
“How do we capture the costs in that full 90 days that we’ll be responsible for?” asked Dr. Shemin, professor of surgery and codirector of the UCLA Cardiovascular Center.
Dr. Mehaffey said that’s indeed an important question, since a major complication such as stroke or deep sternal wound infection typically entails considerable long-term costs and repeated hospital admissions beyond the 30-day window. In Virginia, the cardiac surgery collaborative is working with payers to gain access to the 90 days worth of patient data.
He reported having no financial conflicts regarding his study.
COLORADO SPRINGS – With bundled payment models for coronary artery bypass graft surgery looming ahead, it’s vital that cardiac surgeons take a hard look at the procedure’s hidden costs – namely, the steep price tag for postoperative complications, James H. Mehaffey, MD, said at the annual meeting of the Western Thoracic Surgical Association.
He presented a retrospective study of the 30-day hospital costs for all 36,588 patients who underwent isolated CABG during 2006-2015 at the 19 Virginia centers where the surgery is performed. This was a typical CABG population, with an average predicted risk of mortality of 1.9%. The actual 30-day mortality was 0.6%, so the surgical performance was better than expected.
“The population of patients experiencing one or more major comorbidities demonstrated a significant and dramatic increase in total hospital costs. It was an exponential increase with each additional major morbidity,” reported Dr. Mehaffey of the University of Virginia, Charlottesville.
Indeed, the average cost jumped from $36,580 for uncomplicated surgery to $64,542 with one major complication, $111,239 with two, and $194,043 with three.
The two most frequent major complications were postoperative atrial fibrillation, which occurred in 18.4% of patients, and prolonged ventilation for longer than 24 hours, which occurred in 9%. Over the course of the decade-long study period, the 19 medical centers in the Virginia Cardiac Surgery Quality Initiative collectively spent roughly $59 million on prolonged ventilation and $27 million for postoperative atrial fibrillation.
The cost of CABG during the study years outpaced the CMS health care–specific inflation rate, and this escalating cost was driven primarily by postoperative complications.
For the Virginia cardiac surgery collaborative, these data on the cost of postoperative complications will be utilized to prioritize quality improvement projects.

For example, during the past decade, the Virginia collaborative made reduction in the rate of postoperative atrial fibrillation a priority. Toward that end, the collaborative developed a protocol for routine perioperative prophylactic amiodarone therapy.
“At the beginning of the study decade we had postoperative atrial fibrillation rates above 25%. The average for the entire decade was just over 18%, and in the last couple years we’ve been in the 15%-16% range. So I think we are moving the needle on this. We are making a meaningful impact,” Dr. Mehaffey said.
“We’ve already used the complication cost data to do a cost-effectiveness analysis of our prophylactic amiodarone innovation. We showed we saved an average of $250 per patient, even though we’re treating a bunch of patients who’d never get that complication,” he continued.
This sort of data on the cost of adverse events is also critical to accurately risk-adjust bundled payment models.
Discussant Richard J. Shemin, MD, asked if there was much variability in postoperative complication costs between the CABG centers in the Virginia collaborative.
The variability is enormous, Dr. Mehaffey replied. Investigators recently plugged the last 5 years worth of hospital cost and complication rate data into a proposed CABG bundled payment model and extrapolated what that would mean over the next 5 years.
“There were some institutions that would be positive by a couple million dollars from this payment system and some that were losing more than $20 million, just because of the cost variability,” said Dr. Mehaffey.
Dr. Shemin also noted that the Virginia collaborative was able to collect 30-day outcome data only through the STS database, yet the bundled payment programs are based on the 90-day postoperative experience.
“How do we capture the costs in that full 90 days that we’ll be responsible for?” asked Dr. Shemin, professor of surgery and codirector of the UCLA Cardiovascular Center.
Dr. Mehaffey said that’s indeed an important question, since a major complication such as stroke or deep sternal wound infection typically entails considerable long-term costs and repeated hospital admissions beyond the 30-day window. In Virginia, the cardiac surgery collaborative is working with payers to gain access to the 90 days worth of patient data.
He reported having no financial conflicts regarding his study.
AT THE WTSA ANNUAL MEETING
Key clinical point:
Major finding: The average 30-day total hospital cost of an isolated CABG procedure during 2006-2015 in Virginia was $36,580 if there were no postoperative complications, jumping to $64,542 with one major complication and $111,239 with two.
Data source: A retrospective study of the 30-day total hospital costs for all isolated CABG procedures performed in Virginia during 2006-2015.
Disclosures: The study presenter reported having no financial conflicts.
Medicare Part D premiums dip as drug costs continue to rise
As insurance premiums in other sectors of the health care industry continue to soar, seniors enrolled in the Medicare Part D prescription drug plan will see a slight decrease in the average premium for their drug coverage.
The Centers for Medicare & Medicaid Services announced Aug. 2 that the average basic premium for drug coverage is projected to drop to $33.50 per month in 2018, down $1.20 from 2017’s average monthly premium of $34.70.
Average premiums are falling despite the rise in drug spending, according to the 2017 Medicare Trustees report, which details the solvency of the Medicare program.
The report notes, however, that drug costs are lower than previous reports due to higher manufacturer rebates and decreased use of direct acting antiviral therapies for hepatitis C virus. Overall, Medicare Part D expenditures per enrollee are estimated to increase by an average of 4.7% annually from 2017 through 2026.
As insurance premiums in other sectors of the health care industry continue to soar, seniors enrolled in the Medicare Part D prescription drug plan will see a slight decrease in the average premium for their drug coverage.
The Centers for Medicare & Medicaid Services announced Aug. 2 that the average basic premium for drug coverage is projected to drop to $33.50 per month in 2018, down $1.20 from 2017’s average monthly premium of $34.70.
Average premiums are falling despite the rise in drug spending, according to the 2017 Medicare Trustees report, which details the solvency of the Medicare program.
The report notes, however, that drug costs are lower than previous reports due to higher manufacturer rebates and decreased use of direct acting antiviral therapies for hepatitis C virus. Overall, Medicare Part D expenditures per enrollee are estimated to increase by an average of 4.7% annually from 2017 through 2026.
As insurance premiums in other sectors of the health care industry continue to soar, seniors enrolled in the Medicare Part D prescription drug plan will see a slight decrease in the average premium for their drug coverage.
The Centers for Medicare & Medicaid Services announced Aug. 2 that the average basic premium for drug coverage is projected to drop to $33.50 per month in 2018, down $1.20 from 2017’s average monthly premium of $34.70.
Average premiums are falling despite the rise in drug spending, according to the 2017 Medicare Trustees report, which details the solvency of the Medicare program.
The report notes, however, that drug costs are lower than previous reports due to higher manufacturer rebates and decreased use of direct acting antiviral therapies for hepatitis C virus. Overall, Medicare Part D expenditures per enrollee are estimated to increase by an average of 4.7% annually from 2017 through 2026.
VIDEO: Study highlights risks of postponing cholecystectomy
Almost half of patients who underwent endoscopic retrograde cholangiopancreatography (ERCP) did not undergo cholecystectomy (CCY) within the next 60 days according to the results of a large, retrospective cohort study reported in the September issue of Gastroenterology (doi: 10.1053/j.gastro.2017.05.048).
“Although early and delayed CCY equally reduce the risk of subsequent recurrent biliary events, patients are at 10-fold higher risk of a recurrent biliary event while waiting for a delayed CCY, compared with patients who underwent early CCY,” wrote Robert J. Huang, MD, and his associates of Stanford (Calif.) University Medical Center. Delayed CCY is cost effective, but that benefit must be weighed against the risk of loss to follow-up, especially if patients have “little or no health insurance,” they said.
Source: American Gastroenterological Association
Gallstone disease affects up to 15% of adults in developed societies, including about 20-25 million Americans. Yearly costs of treatment tally at more than $6.2 billion and have risen by more than 20% in 3 decades, according to multiple studies. Approximately 20% of patients with gallstone disease have choledocholithiasis, mainly because gallstones can pass from the gallbladder into the common bile duct. After undergoing ERCP, such patients are typically referred for CCY, but there are no “societal guidelines” on timing the referral, the researchers said. Practice patterns remain “largely institution based and may be subject to the vagaries of surgeon availability and other institutional resource constraints.” One prior study linked a median 7-week wait time for CCY with a 20% rate of recurrent biliary events. To evaluate large-scale practice patterns, the researchers studied 4,516 patients who had undergone ERCP for choledocholithiasis in California (during 2009-2011), New York (during 2011-2013), and Florida (during 2012-2014) and calculated timing and rates of subsequent CCY, recurrent biliary events, and deaths. Patients were followed for up to 365 days after ERCP.
Of the 4,516 patients studied, 1,859 (41.2%) patients underwent CCY during their index hospital admission (early CCY). Of the 2,657 (58.8%) patients who were discharged without CCY, only 491 (18%) had a planned CCY within 60 days (delayed CCY), 350 (71.3%) of which were done in an outpatient setting. Of the patients in the study, 2,168 (48.0%) did not have a CCY (no CCY) during their index visit or within 60 days. Over 365 days of follow-up, 10% of patients who did not have a CCY had recurrent biliary events, compared with 1.3% of patients who underwent early or delayed CCY. The risk of recurrent biliary events for patients who underwent early or delayed CCY was about 88% lower than if they had had no CCY within 60 days of ERCP (P less than .001 for each comparison). Performing CCY during index admission cut the risk of recurrent biliary events occurring within 60 days by 92%, compared with delayed or no CCY (P less than .001).
In all, 15 (0.7%) patients who did not undergo CCY died after subsequent hospitalization for a recurrent biliary event, compared with 1 patient who underwent early CCY (0.1%; P less than .001). There were no deaths associated with recurrent biliary events in the delayed-CCY group. Rates of all-cause mortality over 365 days were 3.1% in the no-CCY group, 0.6% in the early-CCY group, and 0% in the delayed-CCY group. Thus, cumulative death rates were about seven times higher among patients who did not undergo CCY compared with those who did (P less than .001).
Patients who did not undergo CCY tended to be older than delayed- and early-CCY patients (mean ages 66 years, 58 years, and 52 years, respectively). No-CCY patients also tended to have more comorbidities. Nonetheless, having an early CCY retained a “robust” protective effect against recurrent biliary events after accounting for age, sex, comorbidities, stent placement, facility volume, and state of residence. Even after researchers adjusted for those factors, the protective effect of early CCY dropped by less than 5% (from 92% to about 87%), the investigators said.
They also noted that the overall cohort averaged 60 years of age and that 64% were female, which is consistent with the epidemiology of biliary stone disease. Just over half were non-Hispanic whites. Medicare was the single largest primary payer (46%), followed by private insurance (28%) and Medicaid (16%).
“A strategy of delayed CCY performed on an outpatient basis was least costly,” the researchers said. “Performance of early CCY was inversely associated with low facility volume. Hispanic race, Asian race, Medicaid insurance, and no insurance associated inversely with performance of delayed CCY.”
Funders included a seed grant from the Stanford division of gastroenterology and hepatology and the National Institutes of Health. The investigators had no conflicts of interest.
Almost half of patients who underwent endoscopic retrograde cholangiopancreatography (ERCP) did not undergo cholecystectomy (CCY) within the next 60 days according to the results of a large, retrospective cohort study reported in the September issue of Gastroenterology (doi: 10.1053/j.gastro.2017.05.048).
“Although early and delayed CCY equally reduce the risk of subsequent recurrent biliary events, patients are at 10-fold higher risk of a recurrent biliary event while waiting for a delayed CCY, compared with patients who underwent early CCY,” wrote Robert J. Huang, MD, and his associates of Stanford (Calif.) University Medical Center. Delayed CCY is cost effective, but that benefit must be weighed against the risk of loss to follow-up, especially if patients have “little or no health insurance,” they said.
Source: American Gastroenterological Association
Gallstone disease affects up to 15% of adults in developed societies, including about 20-25 million Americans. Yearly costs of treatment tally at more than $6.2 billion and have risen by more than 20% in 3 decades, according to multiple studies. Approximately 20% of patients with gallstone disease have choledocholithiasis, mainly because gallstones can pass from the gallbladder into the common bile duct. After undergoing ERCP, such patients are typically referred for CCY, but there are no “societal guidelines” on timing the referral, the researchers said. Practice patterns remain “largely institution based and may be subject to the vagaries of surgeon availability and other institutional resource constraints.” One prior study linked a median 7-week wait time for CCY with a 20% rate of recurrent biliary events. To evaluate large-scale practice patterns, the researchers studied 4,516 patients who had undergone ERCP for choledocholithiasis in California (during 2009-2011), New York (during 2011-2013), and Florida (during 2012-2014) and calculated timing and rates of subsequent CCY, recurrent biliary events, and deaths. Patients were followed for up to 365 days after ERCP.
Of the 4,516 patients studied, 1,859 (41.2%) patients underwent CCY during their index hospital admission (early CCY). Of the 2,657 (58.8%) patients who were discharged without CCY, only 491 (18%) had a planned CCY within 60 days (delayed CCY), 350 (71.3%) of which were done in an outpatient setting. Of the patients in the study, 2,168 (48.0%) did not have a CCY (no CCY) during their index visit or within 60 days. Over 365 days of follow-up, 10% of patients who did not have a CCY had recurrent biliary events, compared with 1.3% of patients who underwent early or delayed CCY. The risk of recurrent biliary events for patients who underwent early or delayed CCY was about 88% lower than if they had had no CCY within 60 days of ERCP (P less than .001 for each comparison). Performing CCY during index admission cut the risk of recurrent biliary events occurring within 60 days by 92%, compared with delayed or no CCY (P less than .001).
In all, 15 (0.7%) patients who did not undergo CCY died after subsequent hospitalization for a recurrent biliary event, compared with 1 patient who underwent early CCY (0.1%; P less than .001). There were no deaths associated with recurrent biliary events in the delayed-CCY group. Rates of all-cause mortality over 365 days were 3.1% in the no-CCY group, 0.6% in the early-CCY group, and 0% in the delayed-CCY group. Thus, cumulative death rates were about seven times higher among patients who did not undergo CCY compared with those who did (P less than .001).
Patients who did not undergo CCY tended to be older than delayed- and early-CCY patients (mean ages 66 years, 58 years, and 52 years, respectively). No-CCY patients also tended to have more comorbidities. Nonetheless, having an early CCY retained a “robust” protective effect against recurrent biliary events after accounting for age, sex, comorbidities, stent placement, facility volume, and state of residence. Even after researchers adjusted for those factors, the protective effect of early CCY dropped by less than 5% (from 92% to about 87%), the investigators said.
They also noted that the overall cohort averaged 60 years of age and that 64% were female, which is consistent with the epidemiology of biliary stone disease. Just over half were non-Hispanic whites. Medicare was the single largest primary payer (46%), followed by private insurance (28%) and Medicaid (16%).
“A strategy of delayed CCY performed on an outpatient basis was least costly,” the researchers said. “Performance of early CCY was inversely associated with low facility volume. Hispanic race, Asian race, Medicaid insurance, and no insurance associated inversely with performance of delayed CCY.”
Funders included a seed grant from the Stanford division of gastroenterology and hepatology and the National Institutes of Health. The investigators had no conflicts of interest.
Almost half of patients who underwent endoscopic retrograde cholangiopancreatography (ERCP) did not undergo cholecystectomy (CCY) within the next 60 days according to the results of a large, retrospective cohort study reported in the September issue of Gastroenterology (doi: 10.1053/j.gastro.2017.05.048).
“Although early and delayed CCY equally reduce the risk of subsequent recurrent biliary events, patients are at 10-fold higher risk of a recurrent biliary event while waiting for a delayed CCY, compared with patients who underwent early CCY,” wrote Robert J. Huang, MD, and his associates of Stanford (Calif.) University Medical Center. Delayed CCY is cost effective, but that benefit must be weighed against the risk of loss to follow-up, especially if patients have “little or no health insurance,” they said.
Source: American Gastroenterological Association
Gallstone disease affects up to 15% of adults in developed societies, including about 20-25 million Americans. Yearly costs of treatment tally at more than $6.2 billion and have risen by more than 20% in 3 decades, according to multiple studies. Approximately 20% of patients with gallstone disease have choledocholithiasis, mainly because gallstones can pass from the gallbladder into the common bile duct. After undergoing ERCP, such patients are typically referred for CCY, but there are no “societal guidelines” on timing the referral, the researchers said. Practice patterns remain “largely institution based and may be subject to the vagaries of surgeon availability and other institutional resource constraints.” One prior study linked a median 7-week wait time for CCY with a 20% rate of recurrent biliary events. To evaluate large-scale practice patterns, the researchers studied 4,516 patients who had undergone ERCP for choledocholithiasis in California (during 2009-2011), New York (during 2011-2013), and Florida (during 2012-2014) and calculated timing and rates of subsequent CCY, recurrent biliary events, and deaths. Patients were followed for up to 365 days after ERCP.
Of the 4,516 patients studied, 1,859 (41.2%) patients underwent CCY during their index hospital admission (early CCY). Of the 2,657 (58.8%) patients who were discharged without CCY, only 491 (18%) had a planned CCY within 60 days (delayed CCY), 350 (71.3%) of which were done in an outpatient setting. Of the patients in the study, 2,168 (48.0%) did not have a CCY (no CCY) during their index visit or within 60 days. Over 365 days of follow-up, 10% of patients who did not have a CCY had recurrent biliary events, compared with 1.3% of patients who underwent early or delayed CCY. The risk of recurrent biliary events for patients who underwent early or delayed CCY was about 88% lower than if they had had no CCY within 60 days of ERCP (P less than .001 for each comparison). Performing CCY during index admission cut the risk of recurrent biliary events occurring within 60 days by 92%, compared with delayed or no CCY (P less than .001).
In all, 15 (0.7%) patients who did not undergo CCY died after subsequent hospitalization for a recurrent biliary event, compared with 1 patient who underwent early CCY (0.1%; P less than .001). There were no deaths associated with recurrent biliary events in the delayed-CCY group. Rates of all-cause mortality over 365 days were 3.1% in the no-CCY group, 0.6% in the early-CCY group, and 0% in the delayed-CCY group. Thus, cumulative death rates were about seven times higher among patients who did not undergo CCY compared with those who did (P less than .001).
Patients who did not undergo CCY tended to be older than delayed- and early-CCY patients (mean ages 66 years, 58 years, and 52 years, respectively). No-CCY patients also tended to have more comorbidities. Nonetheless, having an early CCY retained a “robust” protective effect against recurrent biliary events after accounting for age, sex, comorbidities, stent placement, facility volume, and state of residence. Even after researchers adjusted for those factors, the protective effect of early CCY dropped by less than 5% (from 92% to about 87%), the investigators said.
They also noted that the overall cohort averaged 60 years of age and that 64% were female, which is consistent with the epidemiology of biliary stone disease. Just over half were non-Hispanic whites. Medicare was the single largest primary payer (46%), followed by private insurance (28%) and Medicaid (16%).
“A strategy of delayed CCY performed on an outpatient basis was least costly,” the researchers said. “Performance of early CCY was inversely associated with low facility volume. Hispanic race, Asian race, Medicaid insurance, and no insurance associated inversely with performance of delayed CCY.”
Funders included a seed grant from the Stanford division of gastroenterology and hepatology and the National Institutes of Health. The investigators had no conflicts of interest.
FROM GASTROENTEROLOGY
Key clinical point: Almost half of patients who underwent endoscopic retrograde cholangiopancreatography (ERCP) did not undergo cholecystectomy within 60 days.
Major finding: A total of 48% had no cholecystectomy within 60 days. Performing cholecystectomy during index admission cut the risk of recurrent biliary events within 60 days by 92%, compared with delayed or no cholecystectomy (P less than .001).
Data source: A multistate, retrospective study of 4,516 patients hospitalized with choledocholithiasis.
Disclosures: Funders included a Stanford division of gastroenterology and hepatology divisional seed grant and the National Institutes of Health. The investigators had no conflicts of interest.
Solid organ transplantation contributes significantly to incidence of NHL among children and adolescents
Solid organ transplant recipients contribute a disproportionate fraction of pediatric non-Hodgkin lymphoma (NHL) cases, especially diffuse large B-cell lymphoma cases (DLBCL), according to an analysis of data drawn from transplant and cancer registries.
Investigators calculated that the incidence of NHL for the pediatric transplant population was 257 times higher than the general population, after analysis from the U.S. transplant registry and 16 cancer registries from around the country between 1990 and 2012. The incidence of NHL for the pediatric transplant population was 306 cases per 100,000 person-years (95% CI, 271-344), compared to 1.19 cases per 100,000 person-years (95% CI, 1.12-1.27) in the general population. Furthermore, transplant recipients made up a much larger proportion of general population DLBCL cases (7.62%; 95% CI, 6.35%-8.88%).
In the general population, the most common subtypes were DLBCL (25% of cases), Burkitt lymphoma (24%), and precursor cell lymphoblastic lymphoma (20%). Among NHLs diagnosed in transplant recipients, 65% were DLBCL, and 9% were Burkitt lymphoma, whereas there were no cases of precursor cell lymphoblastic lymphoma, reported Elizabeth L. Yanik, PhD, of Washington University, St. Louis, and her associates (Cancer. 2017 Jul 31. doi: 10.1002/cncr.30923).
The increased risk of NHL and other cancers for organ transplant recipients is primarily the result of the immunosuppressant medications administered after transplantation, leading to an increased risk of infection-related cancers, the authors said.
“NHL cases are largely attributable to EBV [Epstein-Barr virus] infections that occur while recipients are immunosuppressed, as evidenced by the high prevalence of EBV detectable in NHL tumors and particularly in cases diagnosed during the heavily immunosuppressed period early after transplantation,” wrote Dr. Yanik and her colleagues. “Patients who experience a primary EBV infection after transplantation are particularly susceptible because transplant recipients have a higher NHL risk if they are seronegative for EBV before transplantation.”
Among those at risk, Dr. Yanik and fellow investigators found transplant patients younger than 5 years were the most susceptible to NHL, making up 19.79% of those diagnosed with DLBCL.
The proportion of NHL in solid organ transplant recipients within the entire pediatric NHL population has risen over time, from 1.66% of the NHL population during 1990-1994 to 3.73% during 2010-2012.
“This trend is driven by the rising prevalence of transplant recipients in the general population and not by increases in NHL risk among transplant recipients,” noted Dr. Yanik and her coauthors. “The proportion of NHL diagnoses attributable to transplant recipients has grown over time, and it is likely that this population will be an important source of pediatric NHL cases in the future.”
This study was partially funded by the National Cancer Institute. One coauthor reported being an employee at GRAIL Inc. No other relevant financial disclosures were reported.
[email protected]
On Twitter @eaztweets
Solid organ transplant recipients contribute a disproportionate fraction of pediatric non-Hodgkin lymphoma (NHL) cases, especially diffuse large B-cell lymphoma cases (DLBCL), according to an analysis of data drawn from transplant and cancer registries.
Investigators calculated that the incidence of NHL for the pediatric transplant population was 257 times higher than the general population, after analysis from the U.S. transplant registry and 16 cancer registries from around the country between 1990 and 2012. The incidence of NHL for the pediatric transplant population was 306 cases per 100,000 person-years (95% CI, 271-344), compared to 1.19 cases per 100,000 person-years (95% CI, 1.12-1.27) in the general population. Furthermore, transplant recipients made up a much larger proportion of general population DLBCL cases (7.62%; 95% CI, 6.35%-8.88%).
In the general population, the most common subtypes were DLBCL (25% of cases), Burkitt lymphoma (24%), and precursor cell lymphoblastic lymphoma (20%). Among NHLs diagnosed in transplant recipients, 65% were DLBCL, and 9% were Burkitt lymphoma, whereas there were no cases of precursor cell lymphoblastic lymphoma, reported Elizabeth L. Yanik, PhD, of Washington University, St. Louis, and her associates (Cancer. 2017 Jul 31. doi: 10.1002/cncr.30923).
The increased risk of NHL and other cancers for organ transplant recipients is primarily the result of the immunosuppressant medications administered after transplantation, leading to an increased risk of infection-related cancers, the authors said.
“NHL cases are largely attributable to EBV [Epstein-Barr virus] infections that occur while recipients are immunosuppressed, as evidenced by the high prevalence of EBV detectable in NHL tumors and particularly in cases diagnosed during the heavily immunosuppressed period early after transplantation,” wrote Dr. Yanik and her colleagues. “Patients who experience a primary EBV infection after transplantation are particularly susceptible because transplant recipients have a higher NHL risk if they are seronegative for EBV before transplantation.”
Among those at risk, Dr. Yanik and fellow investigators found transplant patients younger than 5 years were the most susceptible to NHL, making up 19.79% of those diagnosed with DLBCL.
The proportion of NHL in solid organ transplant recipients within the entire pediatric NHL population has risen over time, from 1.66% of the NHL population during 1990-1994 to 3.73% during 2010-2012.
“This trend is driven by the rising prevalence of transplant recipients in the general population and not by increases in NHL risk among transplant recipients,” noted Dr. Yanik and her coauthors. “The proportion of NHL diagnoses attributable to transplant recipients has grown over time, and it is likely that this population will be an important source of pediatric NHL cases in the future.”
This study was partially funded by the National Cancer Institute. One coauthor reported being an employee at GRAIL Inc. No other relevant financial disclosures were reported.
[email protected]
On Twitter @eaztweets
Solid organ transplant recipients contribute a disproportionate fraction of pediatric non-Hodgkin lymphoma (NHL) cases, especially diffuse large B-cell lymphoma cases (DLBCL), according to an analysis of data drawn from transplant and cancer registries.
Investigators calculated that the incidence of NHL for the pediatric transplant population was 257 times higher than the general population, after analysis from the U.S. transplant registry and 16 cancer registries from around the country between 1990 and 2012. The incidence of NHL for the pediatric transplant population was 306 cases per 100,000 person-years (95% CI, 271-344), compared to 1.19 cases per 100,000 person-years (95% CI, 1.12-1.27) in the general population. Furthermore, transplant recipients made up a much larger proportion of general population DLBCL cases (7.62%; 95% CI, 6.35%-8.88%).
In the general population, the most common subtypes were DLBCL (25% of cases), Burkitt lymphoma (24%), and precursor cell lymphoblastic lymphoma (20%). Among NHLs diagnosed in transplant recipients, 65% were DLBCL, and 9% were Burkitt lymphoma, whereas there were no cases of precursor cell lymphoblastic lymphoma, reported Elizabeth L. Yanik, PhD, of Washington University, St. Louis, and her associates (Cancer. 2017 Jul 31. doi: 10.1002/cncr.30923).
The increased risk of NHL and other cancers for organ transplant recipients is primarily the result of the immunosuppressant medications administered after transplantation, leading to an increased risk of infection-related cancers, the authors said.
“NHL cases are largely attributable to EBV [Epstein-Barr virus] infections that occur while recipients are immunosuppressed, as evidenced by the high prevalence of EBV detectable in NHL tumors and particularly in cases diagnosed during the heavily immunosuppressed period early after transplantation,” wrote Dr. Yanik and her colleagues. “Patients who experience a primary EBV infection after transplantation are particularly susceptible because transplant recipients have a higher NHL risk if they are seronegative for EBV before transplantation.”
Among those at risk, Dr. Yanik and fellow investigators found transplant patients younger than 5 years were the most susceptible to NHL, making up 19.79% of those diagnosed with DLBCL.
The proportion of NHL in solid organ transplant recipients within the entire pediatric NHL population has risen over time, from 1.66% of the NHL population during 1990-1994 to 3.73% during 2010-2012.
“This trend is driven by the rising prevalence of transplant recipients in the general population and not by increases in NHL risk among transplant recipients,” noted Dr. Yanik and her coauthors. “The proportion of NHL diagnoses attributable to transplant recipients has grown over time, and it is likely that this population will be an important source of pediatric NHL cases in the future.”
This study was partially funded by the National Cancer Institute. One coauthor reported being an employee at GRAIL Inc. No other relevant financial disclosures were reported.
[email protected]
On Twitter @eaztweets
FROM CANCER
Key clinical point: Solid organ transplant recipients contribute a disproportionate fraction of pediatric NHL cases, especially DLBCL cases.
Major finding: Incidence of NHL among the pediatric transplant population was 257 times higher at 306 cases per 100,000 person-years (95% CI, 271-344), compared to 1.19 cases per 100,000 person-years (95% CI, 1.12-1.27) in the general pediatric population.
Data source: Retrospective cohort study of children and adolescents in the U.S. transplant registry and 16 cancer registries from around the country between 1990 and 2012.
Disclosures: This study was partially funded by the National Cancer Institute. One coauthor reported being an employee at GRAIL Inc. No other relevant financial disclosures were reported.
Malperfusion key in aortic dissection repair outcomes
Early repair is the standard of care for patients with type A aortic dissection, but the presence of malperfusion rather than the timing of surgery may be a major determinant in patient survival both in the hospital and in the long term, according to an analysis of patients with acute type A aortic dissection over a 17-year period at the University of Bristol (England).
“Malperfusion at presentation rather than the timing of intervention is the major risk factor for death in both the short term and long term in patients undergoing surgical repair of type A aortic dissection,” Pradeep Narayan, FRCS, and his colleagues said in reporting their findings in the July issue of the Journal of Thoracic and Cardiovascular Surgery (154:81-6). Nonetheless, Dr. Narayan and his colleagues acknowledged that early operation prevents the development of malperfusion and is the best option for restoring normal perfusion for patients who already have malperfusion.
Their study analyzed results from two different groups of patients who had surgery for repair of acute type A aortic dissection over a 17-year period: 72 in the early surgery group that had operative repair within 12 hours of symptom onset; and 80 in the late-surgery group that had the operation 12 hours or more after symptoms first appeared. A total of 205 patients underwent surgical repair for acute type A aortic dissection in that period, but only 152 cases had recorded the timing of surgery from onset of symptoms. The median time between arrival at the center and surgery was 3 hours.
Dr. Narayan and his coauthors reported that 39% (60) of the 152 patients had malperfusion. Organ malperfusion was actually more common in the early surgery group, although the difference was not significant: 48.6% vs. 31.3% in the late-surgery group (P = .29). Early mortality was also similar between the two groups: 19.4% in the early surgery group and 13.8% in the late surgery group (P = .8). In terms of late survival, the study found no difference between the two groups.
Dr. Narayan and his coauthors reported that malperfusion and concomitant coronary artery bypass grafting were independent predictors of survival, with hazard ratios of 2.65 (P = .01) and 3.03 (P = .03), respectively. As a nonlinear variable, time to surgery showed an inverse relationship with late mortality (HR, 0.51; P = .26), but as a linear variable when adjusted for other covariates, including malperfusion, it did not affect survival (HR, 1.01; P = .09).
“The main finding of the present study is that almost 40% of patients undergoing repair of type A aortic dissection had evidence of malperfusion,” Dr. Narayan and his coauthors said. “The second important finding is that the presence of malperfusion was associated with significantly increased risk of death in both the short-term and long-term follow-up.” While a delayed operation was associated with a reduced risk of death, it was not significant when accounting for malperfusion.
Dr. Narayan and his coauthors acknowledged limitations of their study, the most important of which was the including of different types of malperfusion as a single variable. Also, the small sample size may explain the lack of statistically significant differences between the two groups.
Dr. Narayan and his coauthors had no financial relationships to disclose.
Malperfusion has the potential to serve as a marker for the need for surgery in type A aortic dissection, but the inability to identify the true risk of developing malperfusion in the first 12-24 hours after acute type A dissection means that the indication for early surgery will remain unchanged, James I. Fann, MD, of Stanford (Calif.) University says in his invited commentary (J Thorac Cardiovasc Surg. 2017;154:87-8).
“The findings of Narayan and colleagues impel us to review the history of the development of the classification and treatment (or in fact vice versa) of acute type A dissection and to acknowledge that early timing of surgery in these high-risk patients was originally proposed to prevent malperfusion and to respond to the most catastrophic complications,” Dr. Fann said.
But Dr. Fann cautioned against “being dismissive” of their findings, because such questioning and re-evaluation are essential in developing appropriate treatments. “Now, the question is whether we can identify the cohort of patients who are at lower risk for the development of malperfusion and tailor their treatment,” he said.
Dr. Fann had no financial relationships to disclose.
Malperfusion has the potential to serve as a marker for the need for surgery in type A aortic dissection, but the inability to identify the true risk of developing malperfusion in the first 12-24 hours after acute type A dissection means that the indication for early surgery will remain unchanged, James I. Fann, MD, of Stanford (Calif.) University says in his invited commentary (J Thorac Cardiovasc Surg. 2017;154:87-8).
“The findings of Narayan and colleagues impel us to review the history of the development of the classification and treatment (or in fact vice versa) of acute type A dissection and to acknowledge that early timing of surgery in these high-risk patients was originally proposed to prevent malperfusion and to respond to the most catastrophic complications,” Dr. Fann said.
But Dr. Fann cautioned against “being dismissive” of their findings, because such questioning and re-evaluation are essential in developing appropriate treatments. “Now, the question is whether we can identify the cohort of patients who are at lower risk for the development of malperfusion and tailor their treatment,” he said.
Dr. Fann had no financial relationships to disclose.
Malperfusion has the potential to serve as a marker for the need for surgery in type A aortic dissection, but the inability to identify the true risk of developing malperfusion in the first 12-24 hours after acute type A dissection means that the indication for early surgery will remain unchanged, James I. Fann, MD, of Stanford (Calif.) University says in his invited commentary (J Thorac Cardiovasc Surg. 2017;154:87-8).
“The findings of Narayan and colleagues impel us to review the history of the development of the classification and treatment (or in fact vice versa) of acute type A dissection and to acknowledge that early timing of surgery in these high-risk patients was originally proposed to prevent malperfusion and to respond to the most catastrophic complications,” Dr. Fann said.
But Dr. Fann cautioned against “being dismissive” of their findings, because such questioning and re-evaluation are essential in developing appropriate treatments. “Now, the question is whether we can identify the cohort of patients who are at lower risk for the development of malperfusion and tailor their treatment,” he said.
Dr. Fann had no financial relationships to disclose.
Early repair is the standard of care for patients with type A aortic dissection, but the presence of malperfusion rather than the timing of surgery may be a major determinant in patient survival both in the hospital and in the long term, according to an analysis of patients with acute type A aortic dissection over a 17-year period at the University of Bristol (England).
“Malperfusion at presentation rather than the timing of intervention is the major risk factor for death in both the short term and long term in patients undergoing surgical repair of type A aortic dissection,” Pradeep Narayan, FRCS, and his colleagues said in reporting their findings in the July issue of the Journal of Thoracic and Cardiovascular Surgery (154:81-6). Nonetheless, Dr. Narayan and his colleagues acknowledged that early operation prevents the development of malperfusion and is the best option for restoring normal perfusion for patients who already have malperfusion.
Their study analyzed results from two different groups of patients who had surgery for repair of acute type A aortic dissection over a 17-year period: 72 in the early surgery group that had operative repair within 12 hours of symptom onset; and 80 in the late-surgery group that had the operation 12 hours or more after symptoms first appeared. A total of 205 patients underwent surgical repair for acute type A aortic dissection in that period, but only 152 cases had recorded the timing of surgery from onset of symptoms. The median time between arrival at the center and surgery was 3 hours.
Dr. Narayan and his coauthors reported that 39% (60) of the 152 patients had malperfusion. Organ malperfusion was actually more common in the early surgery group, although the difference was not significant: 48.6% vs. 31.3% in the late-surgery group (P = .29). Early mortality was also similar between the two groups: 19.4% in the early surgery group and 13.8% in the late surgery group (P = .8). In terms of late survival, the study found no difference between the two groups.
Dr. Narayan and his coauthors reported that malperfusion and concomitant coronary artery bypass grafting were independent predictors of survival, with hazard ratios of 2.65 (P = .01) and 3.03 (P = .03), respectively. As a nonlinear variable, time to surgery showed an inverse relationship with late mortality (HR, 0.51; P = .26), but as a linear variable when adjusted for other covariates, including malperfusion, it did not affect survival (HR, 1.01; P = .09).
“The main finding of the present study is that almost 40% of patients undergoing repair of type A aortic dissection had evidence of malperfusion,” Dr. Narayan and his coauthors said. “The second important finding is that the presence of malperfusion was associated with significantly increased risk of death in both the short-term and long-term follow-up.” While a delayed operation was associated with a reduced risk of death, it was not significant when accounting for malperfusion.
Dr. Narayan and his coauthors acknowledged limitations of their study, the most important of which was the including of different types of malperfusion as a single variable. Also, the small sample size may explain the lack of statistically significant differences between the two groups.
Dr. Narayan and his coauthors had no financial relationships to disclose.
Early repair is the standard of care for patients with type A aortic dissection, but the presence of malperfusion rather than the timing of surgery may be a major determinant in patient survival both in the hospital and in the long term, according to an analysis of patients with acute type A aortic dissection over a 17-year period at the University of Bristol (England).
“Malperfusion at presentation rather than the timing of intervention is the major risk factor for death in both the short term and long term in patients undergoing surgical repair of type A aortic dissection,” Pradeep Narayan, FRCS, and his colleagues said in reporting their findings in the July issue of the Journal of Thoracic and Cardiovascular Surgery (154:81-6). Nonetheless, Dr. Narayan and his colleagues acknowledged that early operation prevents the development of malperfusion and is the best option for restoring normal perfusion for patients who already have malperfusion.
Their study analyzed results from two different groups of patients who had surgery for repair of acute type A aortic dissection over a 17-year period: 72 in the early surgery group that had operative repair within 12 hours of symptom onset; and 80 in the late-surgery group that had the operation 12 hours or more after symptoms first appeared. A total of 205 patients underwent surgical repair for acute type A aortic dissection in that period, but only 152 cases had recorded the timing of surgery from onset of symptoms. The median time between arrival at the center and surgery was 3 hours.
Dr. Narayan and his coauthors reported that 39% (60) of the 152 patients had malperfusion. Organ malperfusion was actually more common in the early surgery group, although the difference was not significant: 48.6% vs. 31.3% in the late-surgery group (P = .29). Early mortality was also similar between the two groups: 19.4% in the early surgery group and 13.8% in the late surgery group (P = .8). In terms of late survival, the study found no difference between the two groups.
Dr. Narayan and his coauthors reported that malperfusion and concomitant coronary artery bypass grafting were independent predictors of survival, with hazard ratios of 2.65 (P = .01) and 3.03 (P = .03), respectively. As a nonlinear variable, time to surgery showed an inverse relationship with late mortality (HR, 0.51; P = .26), but as a linear variable when adjusted for other covariates, including malperfusion, it did not affect survival (HR, 1.01; P = .09).
“The main finding of the present study is that almost 40% of patients undergoing repair of type A aortic dissection had evidence of malperfusion,” Dr. Narayan and his coauthors said. “The second important finding is that the presence of malperfusion was associated with significantly increased risk of death in both the short-term and long-term follow-up.” While a delayed operation was associated with a reduced risk of death, it was not significant when accounting for malperfusion.
Dr. Narayan and his coauthors acknowledged limitations of their study, the most important of which was the including of different types of malperfusion as a single variable. Also, the small sample size may explain the lack of statistically significant differences between the two groups.
Dr. Narayan and his coauthors had no financial relationships to disclose.
FROM THE JOURNAL OF THORACIC AND CARDIOVASCULAR SURGERY
Key clinical point: Malperfusion is a main determinant of outcomes for patients having surgical repair for acute type A aortic dissection.
Major finding: Patients in the early surgery group (surgery within 12 hours of onset) were more likely to have malperfusion than those who had surgery later, 47% vs. 31%.
Data source: Single-center analysis of 152 operations for repair of acute type A aortic dissections over a 17-year period.
Disclosures: Dr. Narayan and his coauthors had no financial relationships to disclose.
Fluid protocol takes aim at bariatric surgery readmissions
NEW YORK – With dehydration considered a contributor to hospital readmission after bariatric weight loss surgery, a multidisciplinary team of clinicians searched for a way to get patients to drink up. Specifically,
The project results were presented at a poster session at the American College of Surgeons Quality and Safety Conference.
“Our QI project was to streamline that process to ultimately eliminate or decrease those readmits,” Ms. Melei said.
They used 8-ounce water bottles. To track water consumption, they also numbered the bottles one through six for each patient. One-ounce cups were used during mealtimes. RNs, certified nursing assistants, and food service staff were educated about the fluid intake initiative. Only fluids provided by nursing were permitted and patients also received a clear message about fluid goals.
Another focus was standardizing communication with patients. They were not getting a consistent message from staff on what to expect before, during, and after surgery. “So we had to make sure [we] were all saying the same thing.”
The nurses and the other staff now ask the patients “Did you finish the bottle?” or “Where are you with the bottles?” Ms. Melei said. “At the end of a shift, the nurses document fluid consumption.”
During a 2-month baseline period, 12 patients drank an average 381.5 mL over 24 hours. Since the close of the project, the average daily fluid intake for patients undergoing bariatric surgery is 1,007 mL. In 12 months post implementation, average fluid intake for 39 patients jumped to 1,109.5 mL over 24 hours. “It was just such a hugely successful program. We had buy-in from every department, it worked really well, and we had great results,” said Cheryl Williams, the current bariatric program coordinator at Greenwich Hospital.
Sometimes dehydrated patients present to an emergency department complaining of vomiting, headache, and dizziness if they are not properly hydrated after surgery, Ms. Williams said. “Preventing dehydration helps to improve the patient’s recovery, as well as decrease emergency room visits and hospital admissions.”
Ashutosh Kaul, MD, FACS, medical director of the bariatric surgery program at Greenwich Hospital, said this study shows the importance of team building to improve patient care. “We built a core team that implemented a simple, low-cost, structured, and well-defined water distribution and documentation process, which resulted in better compliance of water intake. This reduced variability and improved postoperative intake progression, and we are presently studying its effect on reduction of readmissions and facilitating early discharge.”
Greenwich Hospital’s bariatric surgery center is accredited by the Metabolic and Bariatric Surgery Accreditation and Quality Improvement Program (MBSAQIP), a collaboration between the American College of Surgeons and the American Society for Metabolic and Bariatric Surgery. Ms. Williams and Ms. Melei had no relevant financial disclosures.
NEW YORK – With dehydration considered a contributor to hospital readmission after bariatric weight loss surgery, a multidisciplinary team of clinicians searched for a way to get patients to drink up. Specifically,
The project results were presented at a poster session at the American College of Surgeons Quality and Safety Conference.
“Our QI project was to streamline that process to ultimately eliminate or decrease those readmits,” Ms. Melei said.
They used 8-ounce water bottles. To track water consumption, they also numbered the bottles one through six for each patient. One-ounce cups were used during mealtimes. RNs, certified nursing assistants, and food service staff were educated about the fluid intake initiative. Only fluids provided by nursing were permitted and patients also received a clear message about fluid goals.
Another focus was standardizing communication with patients. They were not getting a consistent message from staff on what to expect before, during, and after surgery. “So we had to make sure [we] were all saying the same thing.”
The nurses and the other staff now ask the patients “Did you finish the bottle?” or “Where are you with the bottles?” Ms. Melei said. “At the end of a shift, the nurses document fluid consumption.”
During a 2-month baseline period, 12 patients drank an average 381.5 mL over 24 hours. Since the close of the project, the average daily fluid intake for patients undergoing bariatric surgery is 1,007 mL. In 12 months post implementation, average fluid intake for 39 patients jumped to 1,109.5 mL over 24 hours. “It was just such a hugely successful program. We had buy-in from every department, it worked really well, and we had great results,” said Cheryl Williams, the current bariatric program coordinator at Greenwich Hospital.
Sometimes dehydrated patients present to an emergency department complaining of vomiting, headache, and dizziness if they are not properly hydrated after surgery, Ms. Williams said. “Preventing dehydration helps to improve the patient’s recovery, as well as decrease emergency room visits and hospital admissions.”
Ashutosh Kaul, MD, FACS, medical director of the bariatric surgery program at Greenwich Hospital, said this study shows the importance of team building to improve patient care. “We built a core team that implemented a simple, low-cost, structured, and well-defined water distribution and documentation process, which resulted in better compliance of water intake. This reduced variability and improved postoperative intake progression, and we are presently studying its effect on reduction of readmissions and facilitating early discharge.”
Greenwich Hospital’s bariatric surgery center is accredited by the Metabolic and Bariatric Surgery Accreditation and Quality Improvement Program (MBSAQIP), a collaboration between the American College of Surgeons and the American Society for Metabolic and Bariatric Surgery. Ms. Williams and Ms. Melei had no relevant financial disclosures.
NEW YORK – With dehydration considered a contributor to hospital readmission after bariatric weight loss surgery, a multidisciplinary team of clinicians searched for a way to get patients to drink up. Specifically,
The project results were presented at a poster session at the American College of Surgeons Quality and Safety Conference.
“Our QI project was to streamline that process to ultimately eliminate or decrease those readmits,” Ms. Melei said.
They used 8-ounce water bottles. To track water consumption, they also numbered the bottles one through six for each patient. One-ounce cups were used during mealtimes. RNs, certified nursing assistants, and food service staff were educated about the fluid intake initiative. Only fluids provided by nursing were permitted and patients also received a clear message about fluid goals.
Another focus was standardizing communication with patients. They were not getting a consistent message from staff on what to expect before, during, and after surgery. “So we had to make sure [we] were all saying the same thing.”
The nurses and the other staff now ask the patients “Did you finish the bottle?” or “Where are you with the bottles?” Ms. Melei said. “At the end of a shift, the nurses document fluid consumption.”
During a 2-month baseline period, 12 patients drank an average 381.5 mL over 24 hours. Since the close of the project, the average daily fluid intake for patients undergoing bariatric surgery is 1,007 mL. In 12 months post implementation, average fluid intake for 39 patients jumped to 1,109.5 mL over 24 hours. “It was just such a hugely successful program. We had buy-in from every department, it worked really well, and we had great results,” said Cheryl Williams, the current bariatric program coordinator at Greenwich Hospital.
Sometimes dehydrated patients present to an emergency department complaining of vomiting, headache, and dizziness if they are not properly hydrated after surgery, Ms. Williams said. “Preventing dehydration helps to improve the patient’s recovery, as well as decrease emergency room visits and hospital admissions.”
Ashutosh Kaul, MD, FACS, medical director of the bariatric surgery program at Greenwich Hospital, said this study shows the importance of team building to improve patient care. “We built a core team that implemented a simple, low-cost, structured, and well-defined water distribution and documentation process, which resulted in better compliance of water intake. This reduced variability and improved postoperative intake progression, and we are presently studying its effect on reduction of readmissions and facilitating early discharge.”
Greenwich Hospital’s bariatric surgery center is accredited by the Metabolic and Bariatric Surgery Accreditation and Quality Improvement Program (MBSAQIP), a collaboration between the American College of Surgeons and the American Society for Metabolic and Bariatric Surgery. Ms. Williams and Ms. Melei had no relevant financial disclosures.
AT THE ACS QUALITY & SAFETY CONFERENCE
Key clinical point: QI project to increase water intake after bariatric surgery could decrease hospital readmission rates.
Major finding: Multidisciplinary protocol increases 24-hour fluid intake from 382 mL to 1,110 mL on average.
Data source: Comparison of water consumed by 12 patients before versus 39 patients after implementation.
Disclosures: Ms. Williams and Ms. Melei had no relevant financial disclosures.
Senate sinks GOP health care reform effort
After casting the deciding vote to begin debate in the Senate on the Republican effort to repeal and replace the Affordable Care Act, Sen. John McCain (R-Ariz.) brought the effort to an end when he voted against the so-called “skinny repeal” bill introduced by Senate Majority Leader Mitch McConnell (R-Ky.).
Sen. McCain crossed the aisle with Sen. Susan Collins (R-Maine) and Sen. Lisa Murkowski (R-Alaska) and voted with the 48 members of the chamber’s Democratic caucus to kill the bill. Republicans would have needed 50 votes to pass the measure, with Vice President Mike Pence on hand to cast a tie-breaking vote if necessary. With three senators voting against the measure, however, the 49 votes were not enough.
In the early evening on July 27, however, Sen. McCain made it clear during a press conference that he would not vote for a skinny repeal. Other senators expressed concern over the skinny repeal but said they would vote for it if there were assurances that the House would actually go to conference committee (a procedure used when the House and Senate pass different versions of the same bill to allow a bipartisan group to find consensus and create a unified bill) and work out a final bill and not simply move the skinny repeal straight to the president’s desk.
Speaker of the House Paul Ryan (R-Wisc.) had issued a statement to that effect, but the House was laying a path to simply move any bill passed by the Senate, leaving it up in the air as to whether a conference committee would actually be created.
Despite a reported phone call from Speaker Ryan and conversations directly with Vice President Pence on the Senate floor, Sen. McCain ultimately could not be convinced to vote with the majority of Republicans.
“From the beginning, I have believed that Obamacare should be repealed and replaced with a solution that increases competition, lowers costs, and improves care for the American people,” Sen. McCain said in a statement issued July 28, following the early-morning vote. “The so-called ‘skinny repeal’ amendment the Senate voted on today would not accomplish those goals. While the amendment would have repealed some of Obamacare’s most burdensome regulations, it offered no replacement to actually reform our health care system and deliver affordable, quality health care to our citizens. The Speaker’s statement that the House would be ‘willing’ to go to conference does not ease my concern that this shell of a bill could be taken up and passed at any time.”
The vote brought to an end a few days of minimal activity. Every attempt by the GOP to pass some form of amendment to move the process forward – from the tweaked version of the Better Care Replacement Act to a straight repeal of the ACA – failed. Sen. Steve Daines (R-Mont.) even introduced a single-payer amendment, but that failed to get a single yes vote, with all 52 GOP members and five Democrats voting against it and the remaining 43 Democrats voting “present.”
Very few amendments were proposed, though one did get passed in the process that would have permanently repealed the so-called “Cadillac tax,” which levies a 40% tax on plans that exceed a certain value. That amendment passed along party lines by a 52-48 vote.
The future of the ACA and future reform efforts remain unclear at the moment. Following the vote, a defeated Majority Leader McConnell asked the minority party what plan they have to offer. Senate Minority Leader Charles E. Schumer (D-N.Y.) echoed Sen. McCain’s speech earlier in the week after the vote was cast to start the debate to return to regular and hammer out a bipartisan solution though the committee process.
Meanwhile, President Donald Trump advocated via Twitter to simply let the ACA continue to crumble and when it failed, then to work on a deal.
The American Medical Association called on Congress to continue its work on fixing the health care system. “While we are relieved that the Senate did not adopt legislation that would have harmed patients and critical safety net programs, the status quo is not acceptable,” AMA President David Barbe said in a statement. “We urge Congress to initiate a bipartisan effort to address shortcomings in the Affordable Care Act. The first priority should be to stabilize the individual marketplace to achieve the goal of providing access to quality, affordable health coverage for more Americans.”
After casting the deciding vote to begin debate in the Senate on the Republican effort to repeal and replace the Affordable Care Act, Sen. John McCain (R-Ariz.) brought the effort to an end when he voted against the so-called “skinny repeal” bill introduced by Senate Majority Leader Mitch McConnell (R-Ky.).
Sen. McCain crossed the aisle with Sen. Susan Collins (R-Maine) and Sen. Lisa Murkowski (R-Alaska) and voted with the 48 members of the chamber’s Democratic caucus to kill the bill. Republicans would have needed 50 votes to pass the measure, with Vice President Mike Pence on hand to cast a tie-breaking vote if necessary. With three senators voting against the measure, however, the 49 votes were not enough.
In the early evening on July 27, however, Sen. McCain made it clear during a press conference that he would not vote for a skinny repeal. Other senators expressed concern over the skinny repeal but said they would vote for it if there were assurances that the House would actually go to conference committee (a procedure used when the House and Senate pass different versions of the same bill to allow a bipartisan group to find consensus and create a unified bill) and work out a final bill and not simply move the skinny repeal straight to the president’s desk.
Speaker of the House Paul Ryan (R-Wisc.) had issued a statement to that effect, but the House was laying a path to simply move any bill passed by the Senate, leaving it up in the air as to whether a conference committee would actually be created.
Despite a reported phone call from Speaker Ryan and conversations directly with Vice President Pence on the Senate floor, Sen. McCain ultimately could not be convinced to vote with the majority of Republicans.
“From the beginning, I have believed that Obamacare should be repealed and replaced with a solution that increases competition, lowers costs, and improves care for the American people,” Sen. McCain said in a statement issued July 28, following the early-morning vote. “The so-called ‘skinny repeal’ amendment the Senate voted on today would not accomplish those goals. While the amendment would have repealed some of Obamacare’s most burdensome regulations, it offered no replacement to actually reform our health care system and deliver affordable, quality health care to our citizens. The Speaker’s statement that the House would be ‘willing’ to go to conference does not ease my concern that this shell of a bill could be taken up and passed at any time.”
The vote brought to an end a few days of minimal activity. Every attempt by the GOP to pass some form of amendment to move the process forward – from the tweaked version of the Better Care Replacement Act to a straight repeal of the ACA – failed. Sen. Steve Daines (R-Mont.) even introduced a single-payer amendment, but that failed to get a single yes vote, with all 52 GOP members and five Democrats voting against it and the remaining 43 Democrats voting “present.”
Very few amendments were proposed, though one did get passed in the process that would have permanently repealed the so-called “Cadillac tax,” which levies a 40% tax on plans that exceed a certain value. That amendment passed along party lines by a 52-48 vote.
The future of the ACA and future reform efforts remain unclear at the moment. Following the vote, a defeated Majority Leader McConnell asked the minority party what plan they have to offer. Senate Minority Leader Charles E. Schumer (D-N.Y.) echoed Sen. McCain’s speech earlier in the week after the vote was cast to start the debate to return to regular and hammer out a bipartisan solution though the committee process.
Meanwhile, President Donald Trump advocated via Twitter to simply let the ACA continue to crumble and when it failed, then to work on a deal.
The American Medical Association called on Congress to continue its work on fixing the health care system. “While we are relieved that the Senate did not adopt legislation that would have harmed patients and critical safety net programs, the status quo is not acceptable,” AMA President David Barbe said in a statement. “We urge Congress to initiate a bipartisan effort to address shortcomings in the Affordable Care Act. The first priority should be to stabilize the individual marketplace to achieve the goal of providing access to quality, affordable health coverage for more Americans.”
After casting the deciding vote to begin debate in the Senate on the Republican effort to repeal and replace the Affordable Care Act, Sen. John McCain (R-Ariz.) brought the effort to an end when he voted against the so-called “skinny repeal” bill introduced by Senate Majority Leader Mitch McConnell (R-Ky.).
Sen. McCain crossed the aisle with Sen. Susan Collins (R-Maine) and Sen. Lisa Murkowski (R-Alaska) and voted with the 48 members of the chamber’s Democratic caucus to kill the bill. Republicans would have needed 50 votes to pass the measure, with Vice President Mike Pence on hand to cast a tie-breaking vote if necessary. With three senators voting against the measure, however, the 49 votes were not enough.
In the early evening on July 27, however, Sen. McCain made it clear during a press conference that he would not vote for a skinny repeal. Other senators expressed concern over the skinny repeal but said they would vote for it if there were assurances that the House would actually go to conference committee (a procedure used when the House and Senate pass different versions of the same bill to allow a bipartisan group to find consensus and create a unified bill) and work out a final bill and not simply move the skinny repeal straight to the president’s desk.
Speaker of the House Paul Ryan (R-Wisc.) had issued a statement to that effect, but the House was laying a path to simply move any bill passed by the Senate, leaving it up in the air as to whether a conference committee would actually be created.
Despite a reported phone call from Speaker Ryan and conversations directly with Vice President Pence on the Senate floor, Sen. McCain ultimately could not be convinced to vote with the majority of Republicans.
“From the beginning, I have believed that Obamacare should be repealed and replaced with a solution that increases competition, lowers costs, and improves care for the American people,” Sen. McCain said in a statement issued July 28, following the early-morning vote. “The so-called ‘skinny repeal’ amendment the Senate voted on today would not accomplish those goals. While the amendment would have repealed some of Obamacare’s most burdensome regulations, it offered no replacement to actually reform our health care system and deliver affordable, quality health care to our citizens. The Speaker’s statement that the House would be ‘willing’ to go to conference does not ease my concern that this shell of a bill could be taken up and passed at any time.”
The vote brought to an end a few days of minimal activity. Every attempt by the GOP to pass some form of amendment to move the process forward – from the tweaked version of the Better Care Replacement Act to a straight repeal of the ACA – failed. Sen. Steve Daines (R-Mont.) even introduced a single-payer amendment, but that failed to get a single yes vote, with all 52 GOP members and five Democrats voting against it and the remaining 43 Democrats voting “present.”
Very few amendments were proposed, though one did get passed in the process that would have permanently repealed the so-called “Cadillac tax,” which levies a 40% tax on plans that exceed a certain value. That amendment passed along party lines by a 52-48 vote.
The future of the ACA and future reform efforts remain unclear at the moment. Following the vote, a defeated Majority Leader McConnell asked the minority party what plan they have to offer. Senate Minority Leader Charles E. Schumer (D-N.Y.) echoed Sen. McCain’s speech earlier in the week after the vote was cast to start the debate to return to regular and hammer out a bipartisan solution though the committee process.
Meanwhile, President Donald Trump advocated via Twitter to simply let the ACA continue to crumble and when it failed, then to work on a deal.
The American Medical Association called on Congress to continue its work on fixing the health care system. “While we are relieved that the Senate did not adopt legislation that would have harmed patients and critical safety net programs, the status quo is not acceptable,” AMA President David Barbe said in a statement. “We urge Congress to initiate a bipartisan effort to address shortcomings in the Affordable Care Act. The first priority should be to stabilize the individual marketplace to achieve the goal of providing access to quality, affordable health coverage for more Americans.”
Integrated health system builds collegial network of rural surgeons
Recruiting and retaining general surgeons is a longstanding problem for rural hospitals. A regional medical system in the Upper Midwest tackles this challenge by integrating small-town general surgeons into the network and emphasizing professional development, fair compensation, and a sustainable call and leave schedule.
The Gundersen Health System (GHS) is a physician-led, nonprofit health care network that operates in 19 rural counties in parts of Wisconsin, Minnesota, and Iowa, with its main campus in La Crosse, Wis. The network, established over a period of almost 40 years, is a mix of larger GHS medical centers, community hospitals, medical clinics, and GHS-managed and independent critical access hospitals (CAHs).
A sustainable model
“It is clear that the older paradigm of a single rural surgeon providing care 24/7 to an isolated community is vanishing. Design of a more sustainable model involves small groups of rural surgeons working together to provide general surgery and some subspecialty care locally, but who are also part of a larger network for administrative and clinical support,” said Dr. Cogbill in an interview.
Lessons learned
Dr. Cogbill said, “Our 38-year experience with rural surgery in our region has taught us many lessons. The strategy of trying to place a solo general surgeon in every small town with a CAH within our service area was not sustainable nor practical. The development of several rural centers of care within our region has allowed us to be more successful in the recruitment and retention of rural general surgeons who are hired to be part of a small group (optimally three) who provide care to their home community as well as outreach surgical care to several outlying CAHs near their home CAH. This has made it possible to offer a reasonable call schedule, mutual assistance, and the chance to build adequate case volumes. Connectivity to the health system should not mean ‘send all the great cases to the main campus,’ but instead should support the rural surgeons in performing appropriate cases locally.”
The survey respondents were aged 36-55 years, five were male, and all were graduates of U.S. medical schools. Eight are board certified and seven are either fellows or associate members of the American College of Surgeons. Their tenure in the GHS system averaged at least 7 years, ranging from 2 years to more than 20. Their surgical logs for a recent 1-year period show a case mix of endoscopy (63.8%), general surgery (26.7%), and obstetrics (6.1%). Mean annual relative value units for the group were 3,627 (range 2,456-5,846).
One goal of the confidential survey was to explore the reasons behind these surgeons’ choice of a rural practice. Their primary motivations were a preference for a rural lifestyle and a desire for a broad scope of practice. Loan forgiveness motivated some (37.5%), and the influence of a mentor was important for others (25%). The opportunity to join an integrated health system such as GHS was deemed extremely important to seven of the respondents.
Retention of rural surgeons
The most important factors mentioned by survey respondents for remaining in their positions were lifestyle (87.5%), family (75.0%), relationship with patients and colleagues, and scope of practice (75.0%), and compensation (62.6%).
Reasons to consider leaving were call burden (37.5%), relationship with the local hospital (25.0%), and compensation (25.0%).
The survey also looked at potential retention of these general surgeons in the coming 5 years: 37.5% said they were somewhat likely to remain, 25% said they were very likely to remain, and 37.5% said they were extremely likely to stay.
Two successful strategies have been promoting a satisfactory case mix and comanagement of patients who are referred to the main campus. The surgeons from the small towns are encouraged to come to La Crosse to assist in procedures on referred patients, to teach in the surgical residency and the Transition to Practice General Surgery fellowship programs at Gundersen, to participate in clinical research activities, and to engage in a variety of professional activities that strengthen the bonds between GHS and rural surgeons. These interactions help minimize professional isolation, a serious problem for surgeons working on their own in small communities.
Communication is maintained electronically. “Our system includes the use of a common EMR across the entire system allowing mutual access to both inpatient and outpatient records, including full access to digitized diagnostic imaging. GHS has established a number of distance-learning telemedicine links between the main campus and the rural communities that permit real-time patient consultations as well as participation in teaching conferences including Morbidity and Mortality Conferences.”
Reducing burnout in rural surgeons
The GHS model may have some impact on burnout among the rural surgeons in the system, said Dr. Cogbill. “Rural surgeon employment as part of a fully integrated regional network has the potential to reduce the magnitude of burnout by providing administrative assistance to help navigate bureaucratic complexities, easy access for subspecialty consults with colleagues who are known entities, and a model of rural surgery involving pods of three colleagues who can share call, mutual assistance, and case volumes.” Fair and competitive compensation and some degree of loan forgiveness have been in the mix of factors that have helped with recruitment. Administrative assistance from the main campus eases the clerical burden the surgeons face. Guaranteed free time for vacations and educational meetings, as well as a reasonable call schedule, are all built into contracts; this has had a big impact on recruitment. GHS has concluded that three general surgeons in a community is the optimal number to maintain call coverage and mutual assistance. Dr. Cogbill said, “The call schedule is managed by each “pod” of rural general surgeons themselves. With a full complement of three rural surgeons in a pod, they maintain an every third night call schedule. In towns in which there are fewer than three surgeons, the GHS surgeons often share call with surgeons who are not part of GHS to maintain a reasonable/sustainable call schedule.”
The retention track record at GHS is impressive. Since 1978, 19 rural general surgeons have been employed by GHS. Four (21%) rural general surgeons have retired 10 (53%) continue to practice in the network; only 5 (26%) left prior to retirement. Six rural general surgeons practiced in one location for over 20 years.
[email protected]
On Twitter @ThereseBorden
The rural surgeon needs some financial assurance, a reasonable call schedule that allows him/her time away from the job without compromising the care of patients in his/her town, regulatory relief, and a relationship with a “mother ship” referral center that is a true two-way street. The days of the independent private solo practitioner are numbered, and the statistics bear this out. The convergence of declining reimbursement, increasing burden of scrutiny and documentation placed by payers and the government, and an emerging workforce that values work-life balance all contribute to the need to develop programs like this one at Gunderson to maintain the surgical workforce in our small towns. Rural surgery comes with a great deal of intrinsic reward, which makes it an excellent career, if these obstacles can be overcome.
Mark Savarise, MD, FACS, is a general surgeon practicing in South Jordan, Utah, and is clinical associate professor of surgery at the University of Utah, Salt Lake City. He has no disclosures.
The rural surgeon needs some financial assurance, a reasonable call schedule that allows him/her time away from the job without compromising the care of patients in his/her town, regulatory relief, and a relationship with a “mother ship” referral center that is a true two-way street. The days of the independent private solo practitioner are numbered, and the statistics bear this out. The convergence of declining reimbursement, increasing burden of scrutiny and documentation placed by payers and the government, and an emerging workforce that values work-life balance all contribute to the need to develop programs like this one at Gunderson to maintain the surgical workforce in our small towns. Rural surgery comes with a great deal of intrinsic reward, which makes it an excellent career, if these obstacles can be overcome.
Mark Savarise, MD, FACS, is a general surgeon practicing in South Jordan, Utah, and is clinical associate professor of surgery at the University of Utah, Salt Lake City. He has no disclosures.
The rural surgeon needs some financial assurance, a reasonable call schedule that allows him/her time away from the job without compromising the care of patients in his/her town, regulatory relief, and a relationship with a “mother ship” referral center that is a true two-way street. The days of the independent private solo practitioner are numbered, and the statistics bear this out. The convergence of declining reimbursement, increasing burden of scrutiny and documentation placed by payers and the government, and an emerging workforce that values work-life balance all contribute to the need to develop programs like this one at Gunderson to maintain the surgical workforce in our small towns. Rural surgery comes with a great deal of intrinsic reward, which makes it an excellent career, if these obstacles can be overcome.
Mark Savarise, MD, FACS, is a general surgeon practicing in South Jordan, Utah, and is clinical associate professor of surgery at the University of Utah, Salt Lake City. He has no disclosures.
Recruiting and retaining general surgeons is a longstanding problem for rural hospitals. A regional medical system in the Upper Midwest tackles this challenge by integrating small-town general surgeons into the network and emphasizing professional development, fair compensation, and a sustainable call and leave schedule.
The Gundersen Health System (GHS) is a physician-led, nonprofit health care network that operates in 19 rural counties in parts of Wisconsin, Minnesota, and Iowa, with its main campus in La Crosse, Wis. The network, established over a period of almost 40 years, is a mix of larger GHS medical centers, community hospitals, medical clinics, and GHS-managed and independent critical access hospitals (CAHs).
A sustainable model
“It is clear that the older paradigm of a single rural surgeon providing care 24/7 to an isolated community is vanishing. Design of a more sustainable model involves small groups of rural surgeons working together to provide general surgery and some subspecialty care locally, but who are also part of a larger network for administrative and clinical support,” said Dr. Cogbill in an interview.
Lessons learned
Dr. Cogbill said, “Our 38-year experience with rural surgery in our region has taught us many lessons. The strategy of trying to place a solo general surgeon in every small town with a CAH within our service area was not sustainable nor practical. The development of several rural centers of care within our region has allowed us to be more successful in the recruitment and retention of rural general surgeons who are hired to be part of a small group (optimally three) who provide care to their home community as well as outreach surgical care to several outlying CAHs near their home CAH. This has made it possible to offer a reasonable call schedule, mutual assistance, and the chance to build adequate case volumes. Connectivity to the health system should not mean ‘send all the great cases to the main campus,’ but instead should support the rural surgeons in performing appropriate cases locally.”
The survey respondents were aged 36-55 years, five were male, and all were graduates of U.S. medical schools. Eight are board certified and seven are either fellows or associate members of the American College of Surgeons. Their tenure in the GHS system averaged at least 7 years, ranging from 2 years to more than 20. Their surgical logs for a recent 1-year period show a case mix of endoscopy (63.8%), general surgery (26.7%), and obstetrics (6.1%). Mean annual relative value units for the group were 3,627 (range 2,456-5,846).
One goal of the confidential survey was to explore the reasons behind these surgeons’ choice of a rural practice. Their primary motivations were a preference for a rural lifestyle and a desire for a broad scope of practice. Loan forgiveness motivated some (37.5%), and the influence of a mentor was important for others (25%). The opportunity to join an integrated health system such as GHS was deemed extremely important to seven of the respondents.
Retention of rural surgeons
The most important factors mentioned by survey respondents for remaining in their positions were lifestyle (87.5%), family (75.0%), relationship with patients and colleagues, and scope of practice (75.0%), and compensation (62.6%).
Reasons to consider leaving were call burden (37.5%), relationship with the local hospital (25.0%), and compensation (25.0%).
The survey also looked at potential retention of these general surgeons in the coming 5 years: 37.5% said they were somewhat likely to remain, 25% said they were very likely to remain, and 37.5% said they were extremely likely to stay.
Two successful strategies have been promoting a satisfactory case mix and comanagement of patients who are referred to the main campus. The surgeons from the small towns are encouraged to come to La Crosse to assist in procedures on referred patients, to teach in the surgical residency and the Transition to Practice General Surgery fellowship programs at Gundersen, to participate in clinical research activities, and to engage in a variety of professional activities that strengthen the bonds between GHS and rural surgeons. These interactions help minimize professional isolation, a serious problem for surgeons working on their own in small communities.
Communication is maintained electronically. “Our system includes the use of a common EMR across the entire system allowing mutual access to both inpatient and outpatient records, including full access to digitized diagnostic imaging. GHS has established a number of distance-learning telemedicine links between the main campus and the rural communities that permit real-time patient consultations as well as participation in teaching conferences including Morbidity and Mortality Conferences.”
Reducing burnout in rural surgeons
The GHS model may have some impact on burnout among the rural surgeons in the system, said Dr. Cogbill. “Rural surgeon employment as part of a fully integrated regional network has the potential to reduce the magnitude of burnout by providing administrative assistance to help navigate bureaucratic complexities, easy access for subspecialty consults with colleagues who are known entities, and a model of rural surgery involving pods of three colleagues who can share call, mutual assistance, and case volumes.” Fair and competitive compensation and some degree of loan forgiveness have been in the mix of factors that have helped with recruitment. Administrative assistance from the main campus eases the clerical burden the surgeons face. Guaranteed free time for vacations and educational meetings, as well as a reasonable call schedule, are all built into contracts; this has had a big impact on recruitment. GHS has concluded that three general surgeons in a community is the optimal number to maintain call coverage and mutual assistance. Dr. Cogbill said, “The call schedule is managed by each “pod” of rural general surgeons themselves. With a full complement of three rural surgeons in a pod, they maintain an every third night call schedule. In towns in which there are fewer than three surgeons, the GHS surgeons often share call with surgeons who are not part of GHS to maintain a reasonable/sustainable call schedule.”
The retention track record at GHS is impressive. Since 1978, 19 rural general surgeons have been employed by GHS. Four (21%) rural general surgeons have retired 10 (53%) continue to practice in the network; only 5 (26%) left prior to retirement. Six rural general surgeons practiced in one location for over 20 years.
[email protected]
On Twitter @ThereseBorden
Recruiting and retaining general surgeons is a longstanding problem for rural hospitals. A regional medical system in the Upper Midwest tackles this challenge by integrating small-town general surgeons into the network and emphasizing professional development, fair compensation, and a sustainable call and leave schedule.
The Gundersen Health System (GHS) is a physician-led, nonprofit health care network that operates in 19 rural counties in parts of Wisconsin, Minnesota, and Iowa, with its main campus in La Crosse, Wis. The network, established over a period of almost 40 years, is a mix of larger GHS medical centers, community hospitals, medical clinics, and GHS-managed and independent critical access hospitals (CAHs).
A sustainable model
“It is clear that the older paradigm of a single rural surgeon providing care 24/7 to an isolated community is vanishing. Design of a more sustainable model involves small groups of rural surgeons working together to provide general surgery and some subspecialty care locally, but who are also part of a larger network for administrative and clinical support,” said Dr. Cogbill in an interview.
Lessons learned
Dr. Cogbill said, “Our 38-year experience with rural surgery in our region has taught us many lessons. The strategy of trying to place a solo general surgeon in every small town with a CAH within our service area was not sustainable nor practical. The development of several rural centers of care within our region has allowed us to be more successful in the recruitment and retention of rural general surgeons who are hired to be part of a small group (optimally three) who provide care to their home community as well as outreach surgical care to several outlying CAHs near their home CAH. This has made it possible to offer a reasonable call schedule, mutual assistance, and the chance to build adequate case volumes. Connectivity to the health system should not mean ‘send all the great cases to the main campus,’ but instead should support the rural surgeons in performing appropriate cases locally.”
The survey respondents were aged 36-55 years, five were male, and all were graduates of U.S. medical schools. Eight are board certified and seven are either fellows or associate members of the American College of Surgeons. Their tenure in the GHS system averaged at least 7 years, ranging from 2 years to more than 20. Their surgical logs for a recent 1-year period show a case mix of endoscopy (63.8%), general surgery (26.7%), and obstetrics (6.1%). Mean annual relative value units for the group were 3,627 (range 2,456-5,846).
One goal of the confidential survey was to explore the reasons behind these surgeons’ choice of a rural practice. Their primary motivations were a preference for a rural lifestyle and a desire for a broad scope of practice. Loan forgiveness motivated some (37.5%), and the influence of a mentor was important for others (25%). The opportunity to join an integrated health system such as GHS was deemed extremely important to seven of the respondents.
Retention of rural surgeons
The most important factors mentioned by survey respondents for remaining in their positions were lifestyle (87.5%), family (75.0%), relationship with patients and colleagues, and scope of practice (75.0%), and compensation (62.6%).
Reasons to consider leaving were call burden (37.5%), relationship with the local hospital (25.0%), and compensation (25.0%).
The survey also looked at potential retention of these general surgeons in the coming 5 years: 37.5% said they were somewhat likely to remain, 25% said they were very likely to remain, and 37.5% said they were extremely likely to stay.
Two successful strategies have been promoting a satisfactory case mix and comanagement of patients who are referred to the main campus. The surgeons from the small towns are encouraged to come to La Crosse to assist in procedures on referred patients, to teach in the surgical residency and the Transition to Practice General Surgery fellowship programs at Gundersen, to participate in clinical research activities, and to engage in a variety of professional activities that strengthen the bonds between GHS and rural surgeons. These interactions help minimize professional isolation, a serious problem for surgeons working on their own in small communities.
Communication is maintained electronically. “Our system includes the use of a common EMR across the entire system allowing mutual access to both inpatient and outpatient records, including full access to digitized diagnostic imaging. GHS has established a number of distance-learning telemedicine links between the main campus and the rural communities that permit real-time patient consultations as well as participation in teaching conferences including Morbidity and Mortality Conferences.”
Reducing burnout in rural surgeons
The GHS model may have some impact on burnout among the rural surgeons in the system, said Dr. Cogbill. “Rural surgeon employment as part of a fully integrated regional network has the potential to reduce the magnitude of burnout by providing administrative assistance to help navigate bureaucratic complexities, easy access for subspecialty consults with colleagues who are known entities, and a model of rural surgery involving pods of three colleagues who can share call, mutual assistance, and case volumes.” Fair and competitive compensation and some degree of loan forgiveness have been in the mix of factors that have helped with recruitment. Administrative assistance from the main campus eases the clerical burden the surgeons face. Guaranteed free time for vacations and educational meetings, as well as a reasonable call schedule, are all built into contracts; this has had a big impact on recruitment. GHS has concluded that three general surgeons in a community is the optimal number to maintain call coverage and mutual assistance. Dr. Cogbill said, “The call schedule is managed by each “pod” of rural general surgeons themselves. With a full complement of three rural surgeons in a pod, they maintain an every third night call schedule. In towns in which there are fewer than three surgeons, the GHS surgeons often share call with surgeons who are not part of GHS to maintain a reasonable/sustainable call schedule.”
The retention track record at GHS is impressive. Since 1978, 19 rural general surgeons have been employed by GHS. Four (21%) rural general surgeons have retired 10 (53%) continue to practice in the network; only 5 (26%) left prior to retirement. Six rural general surgeons practiced in one location for over 20 years.
[email protected]
On Twitter @ThereseBorden
Comprehensive guidelines released for enhanced colorectal surgery recovery
New guidelines for enhanced recovery from colon and rectal surgery highlight the small steps that can add up to big improvements in patient outcomes.
“I think one of the most surprising aspects” of the guidelines – a joint effort from the American Society of Colon and Rectal Surgeons (ASCRS) and the Society of American Gastrointestinal and Endoscopic Surgeons (SAGES) – “is how enhanced recovery in many ways involves all the little things,” said senior author Scott Steele, MD, FACS, chairman of the department of colorectal surgery at the Cleveland Clinic (Dis Colon Rectum. 2017 Aug;60[8]:761-84. doi: 10.1097/DCR.0000000000000883). The guideline includes 24 literature-based recommendations covering everything from preoperative stoma counseling to postop chewing gum, all rated by quality of evidence.
“Many are easy to incorporate into day-to-day practice: getting [patients] out of bed, avoiding nasogastric tubes, not giving as much IV fluid as we used to, having patients take oral food and drink right after surgery, and having nursing/anesthesia/surgeons all on the same page and understanding that ... multidisciplinary, multisetting care leads to the best outcomes,” he said.
ASCRS and SAGES joined forces after noting that previous guidelines for enhanced recovery – perhaps better known as enhanced recovery after surgery, or ERAS, protocols – are dated, including studies only up to 2012; much has been published since then.
Preop measures
Some of the new recommendations encourage closer patient involvement with care. For instance, the groups strongly recommend discussing goals and discharge criteria with patients before surgery. Recent work has found that compliance and success go up when patients understand what’s going on, and length of stay and complications go down. For similar reasons, stoma education, stoma marking, and counseling on avoiding dehydration should happen preoperatively.
Meanwhile, “although there appear to be no meaningful benefits of [mechanical bowel prep (MBP)] alone in terms of complications,” the groups made a weak recommendation for MBP plus oral antibiotics before surgery. “A meta-analysis of seven RCTs comparing MBP with [antibiotics] versus MBP alone showed a reduction in total surgical site infection and incisional site infection,” they noted.
ASCRS and SAGES strongly recommended that patients drink clear fluids in the 2 hours before surgery, and also recommended carbohydrate loading – specifically drinks high in complex carbohydrates – in nondiabetic patients to attenuate insulin resistance induced by surgery and starvation.
The groups also recommended preset orders to standardize care, and care bundles to reduce surgical site infections. Measures could include preop chlorhexidine showers, ertapenem (Invanz) within an hour of incision, gown and glove changes before fascial closure, and washing incisions with chlorhexidine during recovery.
Pain control
“A multimodal, opioid-sparing, pain management plan should be ... implemented before the induction of anesthesia” for earlier return of bowel function and shorter lengths of stay, they said in a strong recommendation. “One of the simplest techniques to limit opioid intake is to schedule narcotic alternatives, such as oral acetaminophen, nonsteroidal anti-inflammatory drugs (NSAIDs), and gabapentin, rather than giving them on an as-needed basis.” The risk of anastomotic leaks with NSAIDs appears to be most pronounced when patients are on them for more than 3 days.
Wound infiltration and abdominal trunk blocks with liposomal bupivacaine have shown promising results, as well. “Limited data demonstrate that the (TAP) block with a local anesthetic [is] associated with decreased length of stay ... TAP blocks performed before surgery appear to provide better analgesia than TAP blocks performed at the end,” the groups said.
ASCRS and SAGES strongly recommended thoracic epidural analgesia for open colorectal cases, but not for routine use in laparoscopic cases. “The modest analgesic benefits provided by TEA do not support a faster recovery in laparoscopic surgery,” they said, noting that at least in open cases, infusion of a local anesthetic and a lipophilic opioid seems to work better than either option alone.
They also strongly recommended that surgery teams preempt postop nausea and vomiting. Dexamethasone at anesthesia induction and ondansetron at emergence is a common option for patients at risk. Others include total intravenous anesthesia, intravenous acetaminophen, and gabapentin.
Fluid management
Intraoperative crystalloids have to be managed to avoid volume overload and its bad effects. “A maintenance infusion of 1.5-2 mL/kg/h of balanced crystalloid solution is sufficient to cover the needs derived from salt water homeostasis during major abdominal surgery,” ASCRS and SAGES said in a strong recommendation.
“The neuroendocrine response induced by surgical trauma leads to a physiologic reduction of urine output that, in the absence of other signs of hypovolemia, should not trigger additional fluid administration.” Also, “crystalloid or colloid preloading does not prevent hypotension induced by neuraxial blockade ... hypotension induced by epidural analgesia should be managed by reducing the epidural infusion rate and with small doses of vasopressors” – not IV fluids – “so long at the patient is normovolemic,” they noted.
Intravenous fluids should be stopped after recovery room discharge, and clear fluids encouraged as soon as patients can tolerate them.
Postop care
ASCRS and SAGES made strong recommendations for minimally invasive surgery when possible, and for avoiding intra-abdominal drains and nasogastric tubes, both recommendations that support current practice in many places. NG tubes can push oral intake back 2 days, and there’s no evidence that abdominal drains prevent anastomotic leaks, plus there can be complications with both.
The groups also strongly recommended early and progressive patient mobilization to shorten length of stay, and a regular diet immediately after surgery.
As for the chewing gum, “sham feeding (i.e., chewing sugar-free gum for [at least] 10 minutes 3-4 times per day) after colorectal surgery is safe, results in small improvements in GI recovery” – flatus and bowel moments happen sooner – “and may be associated with a reduction in the length of hospital stay.” The groups strongly recommended it based on high-quality evidence
Alvimopan was also a strong recommendation to reverse increased GI transit time and constipation from opioids after open cases. “Several RCTs and pooled post hoc analyses showed accelerated time to recovery of GI function with 6- and 12-mg doses compared with placebo and a significantly shorter hospital length of stay in the alvimopan 12-mg group.” It’s unclear at this point, however, if alvimopan has a role in laparoscopic cases, the groups said.
To reduce the risk of urinary tract infections, they said urinary catheters should be pulled within 24 hours of elective colonic or upper rectal resection not involving a vesicular fistula, and within 48 hours of midrectal/lower rectal resections, which carry a greater risk of urinary retention.
ASCRS and SAGES funded the work. Seven of the 10 authors, including Dr. Steele, had no financial disclosures. One author is a speaker for Pacira Pharmaceuticals, and her institution has received unrestricted educational grants from the company. Another author reported grant support from Medtronic and Merck, maker of alvimopan and ertapenem, and a third reported collaborations with Medtronic and Johnson & Johnson.
New guidelines for enhanced recovery from colon and rectal surgery highlight the small steps that can add up to big improvements in patient outcomes.
“I think one of the most surprising aspects” of the guidelines – a joint effort from the American Society of Colon and Rectal Surgeons (ASCRS) and the Society of American Gastrointestinal and Endoscopic Surgeons (SAGES) – “is how enhanced recovery in many ways involves all the little things,” said senior author Scott Steele, MD, FACS, chairman of the department of colorectal surgery at the Cleveland Clinic (Dis Colon Rectum. 2017 Aug;60[8]:761-84. doi: 10.1097/DCR.0000000000000883). The guideline includes 24 literature-based recommendations covering everything from preoperative stoma counseling to postop chewing gum, all rated by quality of evidence.
“Many are easy to incorporate into day-to-day practice: getting [patients] out of bed, avoiding nasogastric tubes, not giving as much IV fluid as we used to, having patients take oral food and drink right after surgery, and having nursing/anesthesia/surgeons all on the same page and understanding that ... multidisciplinary, multisetting care leads to the best outcomes,” he said.
ASCRS and SAGES joined forces after noting that previous guidelines for enhanced recovery – perhaps better known as enhanced recovery after surgery, or ERAS, protocols – are dated, including studies only up to 2012; much has been published since then.
Preop measures
Some of the new recommendations encourage closer patient involvement with care. For instance, the groups strongly recommend discussing goals and discharge criteria with patients before surgery. Recent work has found that compliance and success go up when patients understand what’s going on, and length of stay and complications go down. For similar reasons, stoma education, stoma marking, and counseling on avoiding dehydration should happen preoperatively.
Meanwhile, “although there appear to be no meaningful benefits of [mechanical bowel prep (MBP)] alone in terms of complications,” the groups made a weak recommendation for MBP plus oral antibiotics before surgery. “A meta-analysis of seven RCTs comparing MBP with [antibiotics] versus MBP alone showed a reduction in total surgical site infection and incisional site infection,” they noted.
ASCRS and SAGES strongly recommended that patients drink clear fluids in the 2 hours before surgery, and also recommended carbohydrate loading – specifically drinks high in complex carbohydrates – in nondiabetic patients to attenuate insulin resistance induced by surgery and starvation.
The groups also recommended preset orders to standardize care, and care bundles to reduce surgical site infections. Measures could include preop chlorhexidine showers, ertapenem (Invanz) within an hour of incision, gown and glove changes before fascial closure, and washing incisions with chlorhexidine during recovery.
Pain control
“A multimodal, opioid-sparing, pain management plan should be ... implemented before the induction of anesthesia” for earlier return of bowel function and shorter lengths of stay, they said in a strong recommendation. “One of the simplest techniques to limit opioid intake is to schedule narcotic alternatives, such as oral acetaminophen, nonsteroidal anti-inflammatory drugs (NSAIDs), and gabapentin, rather than giving them on an as-needed basis.” The risk of anastomotic leaks with NSAIDs appears to be most pronounced when patients are on them for more than 3 days.
Wound infiltration and abdominal trunk blocks with liposomal bupivacaine have shown promising results, as well. “Limited data demonstrate that the (TAP) block with a local anesthetic [is] associated with decreased length of stay ... TAP blocks performed before surgery appear to provide better analgesia than TAP blocks performed at the end,” the groups said.
ASCRS and SAGES strongly recommended thoracic epidural analgesia for open colorectal cases, but not for routine use in laparoscopic cases. “The modest analgesic benefits provided by TEA do not support a faster recovery in laparoscopic surgery,” they said, noting that at least in open cases, infusion of a local anesthetic and a lipophilic opioid seems to work better than either option alone.
They also strongly recommended that surgery teams preempt postop nausea and vomiting. Dexamethasone at anesthesia induction and ondansetron at emergence is a common option for patients at risk. Others include total intravenous anesthesia, intravenous acetaminophen, and gabapentin.
Fluid management
Intraoperative crystalloids have to be managed to avoid volume overload and its bad effects. “A maintenance infusion of 1.5-2 mL/kg/h of balanced crystalloid solution is sufficient to cover the needs derived from salt water homeostasis during major abdominal surgery,” ASCRS and SAGES said in a strong recommendation.
“The neuroendocrine response induced by surgical trauma leads to a physiologic reduction of urine output that, in the absence of other signs of hypovolemia, should not trigger additional fluid administration.” Also, “crystalloid or colloid preloading does not prevent hypotension induced by neuraxial blockade ... hypotension induced by epidural analgesia should be managed by reducing the epidural infusion rate and with small doses of vasopressors” – not IV fluids – “so long at the patient is normovolemic,” they noted.
Intravenous fluids should be stopped after recovery room discharge, and clear fluids encouraged as soon as patients can tolerate them.
Postop care
ASCRS and SAGES made strong recommendations for minimally invasive surgery when possible, and for avoiding intra-abdominal drains and nasogastric tubes, both recommendations that support current practice in many places. NG tubes can push oral intake back 2 days, and there’s no evidence that abdominal drains prevent anastomotic leaks, plus there can be complications with both.
The groups also strongly recommended early and progressive patient mobilization to shorten length of stay, and a regular diet immediately after surgery.
As for the chewing gum, “sham feeding (i.e., chewing sugar-free gum for [at least] 10 minutes 3-4 times per day) after colorectal surgery is safe, results in small improvements in GI recovery” – flatus and bowel moments happen sooner – “and may be associated with a reduction in the length of hospital stay.” The groups strongly recommended it based on high-quality evidence
Alvimopan was also a strong recommendation to reverse increased GI transit time and constipation from opioids after open cases. “Several RCTs and pooled post hoc analyses showed accelerated time to recovery of GI function with 6- and 12-mg doses compared with placebo and a significantly shorter hospital length of stay in the alvimopan 12-mg group.” It’s unclear at this point, however, if alvimopan has a role in laparoscopic cases, the groups said.
To reduce the risk of urinary tract infections, they said urinary catheters should be pulled within 24 hours of elective colonic or upper rectal resection not involving a vesicular fistula, and within 48 hours of midrectal/lower rectal resections, which carry a greater risk of urinary retention.
ASCRS and SAGES funded the work. Seven of the 10 authors, including Dr. Steele, had no financial disclosures. One author is a speaker for Pacira Pharmaceuticals, and her institution has received unrestricted educational grants from the company. Another author reported grant support from Medtronic and Merck, maker of alvimopan and ertapenem, and a third reported collaborations with Medtronic and Johnson & Johnson.
New guidelines for enhanced recovery from colon and rectal surgery highlight the small steps that can add up to big improvements in patient outcomes.
“I think one of the most surprising aspects” of the guidelines – a joint effort from the American Society of Colon and Rectal Surgeons (ASCRS) and the Society of American Gastrointestinal and Endoscopic Surgeons (SAGES) – “is how enhanced recovery in many ways involves all the little things,” said senior author Scott Steele, MD, FACS, chairman of the department of colorectal surgery at the Cleveland Clinic (Dis Colon Rectum. 2017 Aug;60[8]:761-84. doi: 10.1097/DCR.0000000000000883). The guideline includes 24 literature-based recommendations covering everything from preoperative stoma counseling to postop chewing gum, all rated by quality of evidence.
“Many are easy to incorporate into day-to-day practice: getting [patients] out of bed, avoiding nasogastric tubes, not giving as much IV fluid as we used to, having patients take oral food and drink right after surgery, and having nursing/anesthesia/surgeons all on the same page and understanding that ... multidisciplinary, multisetting care leads to the best outcomes,” he said.
ASCRS and SAGES joined forces after noting that previous guidelines for enhanced recovery – perhaps better known as enhanced recovery after surgery, or ERAS, protocols – are dated, including studies only up to 2012; much has been published since then.
Preop measures
Some of the new recommendations encourage closer patient involvement with care. For instance, the groups strongly recommend discussing goals and discharge criteria with patients before surgery. Recent work has found that compliance and success go up when patients understand what’s going on, and length of stay and complications go down. For similar reasons, stoma education, stoma marking, and counseling on avoiding dehydration should happen preoperatively.
Meanwhile, “although there appear to be no meaningful benefits of [mechanical bowel prep (MBP)] alone in terms of complications,” the groups made a weak recommendation for MBP plus oral antibiotics before surgery. “A meta-analysis of seven RCTs comparing MBP with [antibiotics] versus MBP alone showed a reduction in total surgical site infection and incisional site infection,” they noted.
ASCRS and SAGES strongly recommended that patients drink clear fluids in the 2 hours before surgery, and also recommended carbohydrate loading – specifically drinks high in complex carbohydrates – in nondiabetic patients to attenuate insulin resistance induced by surgery and starvation.
The groups also recommended preset orders to standardize care, and care bundles to reduce surgical site infections. Measures could include preop chlorhexidine showers, ertapenem (Invanz) within an hour of incision, gown and glove changes before fascial closure, and washing incisions with chlorhexidine during recovery.
Pain control
“A multimodal, opioid-sparing, pain management plan should be ... implemented before the induction of anesthesia” for earlier return of bowel function and shorter lengths of stay, they said in a strong recommendation. “One of the simplest techniques to limit opioid intake is to schedule narcotic alternatives, such as oral acetaminophen, nonsteroidal anti-inflammatory drugs (NSAIDs), and gabapentin, rather than giving them on an as-needed basis.” The risk of anastomotic leaks with NSAIDs appears to be most pronounced when patients are on them for more than 3 days.
Wound infiltration and abdominal trunk blocks with liposomal bupivacaine have shown promising results, as well. “Limited data demonstrate that the (TAP) block with a local anesthetic [is] associated with decreased length of stay ... TAP blocks performed before surgery appear to provide better analgesia than TAP blocks performed at the end,” the groups said.
ASCRS and SAGES strongly recommended thoracic epidural analgesia for open colorectal cases, but not for routine use in laparoscopic cases. “The modest analgesic benefits provided by TEA do not support a faster recovery in laparoscopic surgery,” they said, noting that at least in open cases, infusion of a local anesthetic and a lipophilic opioid seems to work better than either option alone.
They also strongly recommended that surgery teams preempt postop nausea and vomiting. Dexamethasone at anesthesia induction and ondansetron at emergence is a common option for patients at risk. Others include total intravenous anesthesia, intravenous acetaminophen, and gabapentin.
Fluid management
Intraoperative crystalloids have to be managed to avoid volume overload and its bad effects. “A maintenance infusion of 1.5-2 mL/kg/h of balanced crystalloid solution is sufficient to cover the needs derived from salt water homeostasis during major abdominal surgery,” ASCRS and SAGES said in a strong recommendation.
“The neuroendocrine response induced by surgical trauma leads to a physiologic reduction of urine output that, in the absence of other signs of hypovolemia, should not trigger additional fluid administration.” Also, “crystalloid or colloid preloading does not prevent hypotension induced by neuraxial blockade ... hypotension induced by epidural analgesia should be managed by reducing the epidural infusion rate and with small doses of vasopressors” – not IV fluids – “so long at the patient is normovolemic,” they noted.
Intravenous fluids should be stopped after recovery room discharge, and clear fluids encouraged as soon as patients can tolerate them.
Postop care
ASCRS and SAGES made strong recommendations for minimally invasive surgery when possible, and for avoiding intra-abdominal drains and nasogastric tubes, both recommendations that support current practice in many places. NG tubes can push oral intake back 2 days, and there’s no evidence that abdominal drains prevent anastomotic leaks, plus there can be complications with both.
The groups also strongly recommended early and progressive patient mobilization to shorten length of stay, and a regular diet immediately after surgery.
As for the chewing gum, “sham feeding (i.e., chewing sugar-free gum for [at least] 10 minutes 3-4 times per day) after colorectal surgery is safe, results in small improvements in GI recovery” – flatus and bowel moments happen sooner – “and may be associated with a reduction in the length of hospital stay.” The groups strongly recommended it based on high-quality evidence
Alvimopan was also a strong recommendation to reverse increased GI transit time and constipation from opioids after open cases. “Several RCTs and pooled post hoc analyses showed accelerated time to recovery of GI function with 6- and 12-mg doses compared with placebo and a significantly shorter hospital length of stay in the alvimopan 12-mg group.” It’s unclear at this point, however, if alvimopan has a role in laparoscopic cases, the groups said.
To reduce the risk of urinary tract infections, they said urinary catheters should be pulled within 24 hours of elective colonic or upper rectal resection not involving a vesicular fistula, and within 48 hours of midrectal/lower rectal resections, which carry a greater risk of urinary retention.
ASCRS and SAGES funded the work. Seven of the 10 authors, including Dr. Steele, had no financial disclosures. One author is a speaker for Pacira Pharmaceuticals, and her institution has received unrestricted educational grants from the company. Another author reported grant support from Medtronic and Merck, maker of alvimopan and ertapenem, and a third reported collaborations with Medtronic and Johnson & Johnson.
FROM DISEASES OF THE COLON AND RECTUM