User login
Midwife-assisted community births post low adverse outcomes
Adverse event rates were similarly low overall for women delivering at home or at community birth centers, based on data from a retrospective study of more than 10,000 births.
Increasing numbers of women in the United States are choosing to give birth at home or in freestanding out-of-hospital birth centers, prompted by high patient satisfaction and low intervention, wrote Elizabeth Nethery, MSc, MSM, of the University of British Columbia, Vancouver, and colleagues. Although data from other countries with well-integrated midwifery show no significant difference in outcomes between home or community births and hospital births, data in the United States are limited, and some studies have shown an increase in perinatal mortality for home births, the researchers said.
“ACOG identified elements for safe planned home birth: high degree of integration of midwives, education meeting International Confederation of Midwives standards, ready access to consultation and transfer, and ‘appropriate selection of candidates,’ all of which are present in Washington State,” the researchers wrote.
In a study published in Obstetrics & Gynecology, the researchers reviewed outcomes for 10,609 births attended by members of a professional midwifery association in Washington State between Jan. 1, 2015, and June 30, 2020. Of these, 40.9% (4,344) were planned to take place at home and 59.1% (6,265) were planned to take place at birth centers. The births were attended by a total of 139 midwives. A majority (84%) of the women planning a home or community center birth were White non-Hispanic, and 64% were multiparous.
Overall, 86% of the women gave birth in the location of their choice. Intrapartum transfers to hospitals were significantly more likely for nulliparous women, compared with multiparous women (30.5% vs. 4.2%). However, the cesarean birth rates were not significantly different based on birth location (11% for nulliparous women vs. 1% for multiparous women overall), and maternal and neonatal outcomes were similar for home births and birth center births.
Approximately two-thirds (66%) of the women who transferred to a hospital had a vaginal birth, including 37% of nulliparous women and 20% of multiparous women.
Overall perinatal mortality after the onset of labor and within 7 days was 0.57 per 1,000 births, which was similar to rates seen in other high-income countries with established systems for community birth and midwifery, the researchers noted.
“This large study population of planned home and planned birth center births in a single state with well-integrated midwifery enabled our study to overcome previous limitations to studying planned community births in the United States,” they said.
The study findings were limited by several other factors, notably the inclusion only of members of the Midwives’ Association of Washington State, the researchers said. Although demographics of the women in the study were similar to those in other states, the results may not be generalizable to other states with different programs for training midwives or to a more diverse population; however, better integration of community midwives in the United States overall could lead to comparable outcomes in other states, the researchers concluded.
Birth location should be an informed decision
The current study takes on the controversial topic of safety differences between planned birth locations, wrote Julia C. Phillippi, PhD, CNM, of Vanderbilt University, Nashville, Tenn., in an accompanying editorial.
“Rates of community birth in the United States have increased by 85% since 2004, to more than 62,000 births in 2017, and thousands more individuals planned community births but needed transfer to hospital care,” she said. The interest in and use of home or community births may have increased in the wake of the COVID-19 pandemic as families considered the perceived risks of being in a hospital, she noted.
“There is broad consensus among U.S. perinatal and neonatal health care leadership that informed choice should be a cornerstone of maternity care,” Dr. Phillippi emphasized. Although outcomes were favorable for most patients using community or home birth options in the current study, the selection criteria encouraged only low-risk women to plan home or community births, and they were not compared directly to outcomes for low-risk patients in planned hospital birth settings, she noted.
“Evidence-based information about systems-level and individual characteristics associated with safe, physiologic birth can be helpful in assisting individuals planning location of birth – in terms of selecting hospital birth or opting for community birth if key safety provisions are met,” said Dr. Phillippi. However, “For community birth to have favorable outcomes, systems need open channels for transfer when laboring individuals are no longer low risk or require interventions,” she added.
Larger, prospective studies and ongoing risk assessment is needed to support informed decision-making, said Dr. Phillippi. Publicizing safety considerations and developing transfer pathways can not only improve patient satisfaction, but also reduce preventable perinatal morbidity and mortality, she concluded.
Patient selection is key to successful community birth
The current study is important at this time because of the relatively limited evidence on outcomes with planned community births in the United States, said Iris Krishna, MD, of Emory University, Atlanta, in an interview.
“Most information available is based on observational studies, as is the case with this study, and it is important to continue to add to growing literature,” she said.
Overall, Dr. Krishna said she was not surprised by the study findings. “In the well-selected, low-risk patient with a certified or licensed nurse-midwife, a low rate of adverse outcomes is to be expected,” she said.
“Strict criteria are necessary to guide selection of appropriate candidates for planned community birth to reduce the risk of adverse maternal and/or fetal outcomes,” Dr. Krishna added. “In the appropriately selected low-risk patient with a certified or licensed nurse-midwife, a favorable outcome is achievable. It is also important to have ready access to safe and timely transport to nearby hospitals,” she noted.
“Physicians should counsel patients contemplating a planned community birth that available data may not be generalizable to all birth settings in the United States or to all patients,” Dr. Krishna emphasized. “For example, this cohort is predominantly non-Hispanic White patients, which typically have lower rates of adverse perinatal events in comparison to other ethnicities,” she explained.
“More research is needed, and in particular research comparing planned community births with planned hospital births in the appropriately selected low-risk patient,” Dr. Krishna said.
The study received no outside funding. Lead author Ms. Nethery disclosed support from a Canadian Vanier Graduate Scholarship. The researchers had no financial conflicts to disclose. Dr. Phillippi had no financial conflicts to disclose. Dr. Krishna had no financial conflicts to disclose, but serves on the Editorial Advisory Board of Ob.Gyn News.
Adverse event rates were similarly low overall for women delivering at home or at community birth centers, based on data from a retrospective study of more than 10,000 births.
Increasing numbers of women in the United States are choosing to give birth at home or in freestanding out-of-hospital birth centers, prompted by high patient satisfaction and low intervention, wrote Elizabeth Nethery, MSc, MSM, of the University of British Columbia, Vancouver, and colleagues. Although data from other countries with well-integrated midwifery show no significant difference in outcomes between home or community births and hospital births, data in the United States are limited, and some studies have shown an increase in perinatal mortality for home births, the researchers said.
“ACOG identified elements for safe planned home birth: high degree of integration of midwives, education meeting International Confederation of Midwives standards, ready access to consultation and transfer, and ‘appropriate selection of candidates,’ all of which are present in Washington State,” the researchers wrote.
In a study published in Obstetrics & Gynecology, the researchers reviewed outcomes for 10,609 births attended by members of a professional midwifery association in Washington State between Jan. 1, 2015, and June 30, 2020. Of these, 40.9% (4,344) were planned to take place at home and 59.1% (6,265) were planned to take place at birth centers. The births were attended by a total of 139 midwives. A majority (84%) of the women planning a home or community center birth were White non-Hispanic, and 64% were multiparous.
Overall, 86% of the women gave birth in the location of their choice. Intrapartum transfers to hospitals were significantly more likely for nulliparous women, compared with multiparous women (30.5% vs. 4.2%). However, the cesarean birth rates were not significantly different based on birth location (11% for nulliparous women vs. 1% for multiparous women overall), and maternal and neonatal outcomes were similar for home births and birth center births.
Approximately two-thirds (66%) of the women who transferred to a hospital had a vaginal birth, including 37% of nulliparous women and 20% of multiparous women.
Overall perinatal mortality after the onset of labor and within 7 days was 0.57 per 1,000 births, which was similar to rates seen in other high-income countries with established systems for community birth and midwifery, the researchers noted.
“This large study population of planned home and planned birth center births in a single state with well-integrated midwifery enabled our study to overcome previous limitations to studying planned community births in the United States,” they said.
The study findings were limited by several other factors, notably the inclusion only of members of the Midwives’ Association of Washington State, the researchers said. Although demographics of the women in the study were similar to those in other states, the results may not be generalizable to other states with different programs for training midwives or to a more diverse population; however, better integration of community midwives in the United States overall could lead to comparable outcomes in other states, the researchers concluded.
Birth location should be an informed decision
The current study takes on the controversial topic of safety differences between planned birth locations, wrote Julia C. Phillippi, PhD, CNM, of Vanderbilt University, Nashville, Tenn., in an accompanying editorial.
“Rates of community birth in the United States have increased by 85% since 2004, to more than 62,000 births in 2017, and thousands more individuals planned community births but needed transfer to hospital care,” she said. The interest in and use of home or community births may have increased in the wake of the COVID-19 pandemic as families considered the perceived risks of being in a hospital, she noted.
“There is broad consensus among U.S. perinatal and neonatal health care leadership that informed choice should be a cornerstone of maternity care,” Dr. Phillippi emphasized. Although outcomes were favorable for most patients using community or home birth options in the current study, the selection criteria encouraged only low-risk women to plan home or community births, and they were not compared directly to outcomes for low-risk patients in planned hospital birth settings, she noted.
“Evidence-based information about systems-level and individual characteristics associated with safe, physiologic birth can be helpful in assisting individuals planning location of birth – in terms of selecting hospital birth or opting for community birth if key safety provisions are met,” said Dr. Phillippi. However, “For community birth to have favorable outcomes, systems need open channels for transfer when laboring individuals are no longer low risk or require interventions,” she added.
Larger, prospective studies and ongoing risk assessment is needed to support informed decision-making, said Dr. Phillippi. Publicizing safety considerations and developing transfer pathways can not only improve patient satisfaction, but also reduce preventable perinatal morbidity and mortality, she concluded.
Patient selection is key to successful community birth
The current study is important at this time because of the relatively limited evidence on outcomes with planned community births in the United States, said Iris Krishna, MD, of Emory University, Atlanta, in an interview.
“Most information available is based on observational studies, as is the case with this study, and it is important to continue to add to growing literature,” she said.
Overall, Dr. Krishna said she was not surprised by the study findings. “In the well-selected, low-risk patient with a certified or licensed nurse-midwife, a low rate of adverse outcomes is to be expected,” she said.
“Strict criteria are necessary to guide selection of appropriate candidates for planned community birth to reduce the risk of adverse maternal and/or fetal outcomes,” Dr. Krishna added. “In the appropriately selected low-risk patient with a certified or licensed nurse-midwife, a favorable outcome is achievable. It is also important to have ready access to safe and timely transport to nearby hospitals,” she noted.
“Physicians should counsel patients contemplating a planned community birth that available data may not be generalizable to all birth settings in the United States or to all patients,” Dr. Krishna emphasized. “For example, this cohort is predominantly non-Hispanic White patients, which typically have lower rates of adverse perinatal events in comparison to other ethnicities,” she explained.
“More research is needed, and in particular research comparing planned community births with planned hospital births in the appropriately selected low-risk patient,” Dr. Krishna said.
The study received no outside funding. Lead author Ms. Nethery disclosed support from a Canadian Vanier Graduate Scholarship. The researchers had no financial conflicts to disclose. Dr. Phillippi had no financial conflicts to disclose. Dr. Krishna had no financial conflicts to disclose, but serves on the Editorial Advisory Board of Ob.Gyn News.
Adverse event rates were similarly low overall for women delivering at home or at community birth centers, based on data from a retrospective study of more than 10,000 births.
Increasing numbers of women in the United States are choosing to give birth at home or in freestanding out-of-hospital birth centers, prompted by high patient satisfaction and low intervention, wrote Elizabeth Nethery, MSc, MSM, of the University of British Columbia, Vancouver, and colleagues. Although data from other countries with well-integrated midwifery show no significant difference in outcomes between home or community births and hospital births, data in the United States are limited, and some studies have shown an increase in perinatal mortality for home births, the researchers said.
“ACOG identified elements for safe planned home birth: high degree of integration of midwives, education meeting International Confederation of Midwives standards, ready access to consultation and transfer, and ‘appropriate selection of candidates,’ all of which are present in Washington State,” the researchers wrote.
In a study published in Obstetrics & Gynecology, the researchers reviewed outcomes for 10,609 births attended by members of a professional midwifery association in Washington State between Jan. 1, 2015, and June 30, 2020. Of these, 40.9% (4,344) were planned to take place at home and 59.1% (6,265) were planned to take place at birth centers. The births were attended by a total of 139 midwives. A majority (84%) of the women planning a home or community center birth were White non-Hispanic, and 64% were multiparous.
Overall, 86% of the women gave birth in the location of their choice. Intrapartum transfers to hospitals were significantly more likely for nulliparous women, compared with multiparous women (30.5% vs. 4.2%). However, the cesarean birth rates were not significantly different based on birth location (11% for nulliparous women vs. 1% for multiparous women overall), and maternal and neonatal outcomes were similar for home births and birth center births.
Approximately two-thirds (66%) of the women who transferred to a hospital had a vaginal birth, including 37% of nulliparous women and 20% of multiparous women.
Overall perinatal mortality after the onset of labor and within 7 days was 0.57 per 1,000 births, which was similar to rates seen in other high-income countries with established systems for community birth and midwifery, the researchers noted.
“This large study population of planned home and planned birth center births in a single state with well-integrated midwifery enabled our study to overcome previous limitations to studying planned community births in the United States,” they said.
The study findings were limited by several other factors, notably the inclusion only of members of the Midwives’ Association of Washington State, the researchers said. Although demographics of the women in the study were similar to those in other states, the results may not be generalizable to other states with different programs for training midwives or to a more diverse population; however, better integration of community midwives in the United States overall could lead to comparable outcomes in other states, the researchers concluded.
Birth location should be an informed decision
The current study takes on the controversial topic of safety differences between planned birth locations, wrote Julia C. Phillippi, PhD, CNM, of Vanderbilt University, Nashville, Tenn., in an accompanying editorial.
“Rates of community birth in the United States have increased by 85% since 2004, to more than 62,000 births in 2017, and thousands more individuals planned community births but needed transfer to hospital care,” she said. The interest in and use of home or community births may have increased in the wake of the COVID-19 pandemic as families considered the perceived risks of being in a hospital, she noted.
“There is broad consensus among U.S. perinatal and neonatal health care leadership that informed choice should be a cornerstone of maternity care,” Dr. Phillippi emphasized. Although outcomes were favorable for most patients using community or home birth options in the current study, the selection criteria encouraged only low-risk women to plan home or community births, and they were not compared directly to outcomes for low-risk patients in planned hospital birth settings, she noted.
“Evidence-based information about systems-level and individual characteristics associated with safe, physiologic birth can be helpful in assisting individuals planning location of birth – in terms of selecting hospital birth or opting for community birth if key safety provisions are met,” said Dr. Phillippi. However, “For community birth to have favorable outcomes, systems need open channels for transfer when laboring individuals are no longer low risk or require interventions,” she added.
Larger, prospective studies and ongoing risk assessment is needed to support informed decision-making, said Dr. Phillippi. Publicizing safety considerations and developing transfer pathways can not only improve patient satisfaction, but also reduce preventable perinatal morbidity and mortality, she concluded.
Patient selection is key to successful community birth
The current study is important at this time because of the relatively limited evidence on outcomes with planned community births in the United States, said Iris Krishna, MD, of Emory University, Atlanta, in an interview.
“Most information available is based on observational studies, as is the case with this study, and it is important to continue to add to growing literature,” she said.
Overall, Dr. Krishna said she was not surprised by the study findings. “In the well-selected, low-risk patient with a certified or licensed nurse-midwife, a low rate of adverse outcomes is to be expected,” she said.
“Strict criteria are necessary to guide selection of appropriate candidates for planned community birth to reduce the risk of adverse maternal and/or fetal outcomes,” Dr. Krishna added. “In the appropriately selected low-risk patient with a certified or licensed nurse-midwife, a favorable outcome is achievable. It is also important to have ready access to safe and timely transport to nearby hospitals,” she noted.
“Physicians should counsel patients contemplating a planned community birth that available data may not be generalizable to all birth settings in the United States or to all patients,” Dr. Krishna emphasized. “For example, this cohort is predominantly non-Hispanic White patients, which typically have lower rates of adverse perinatal events in comparison to other ethnicities,” she explained.
“More research is needed, and in particular research comparing planned community births with planned hospital births in the appropriately selected low-risk patient,” Dr. Krishna said.
The study received no outside funding. Lead author Ms. Nethery disclosed support from a Canadian Vanier Graduate Scholarship. The researchers had no financial conflicts to disclose. Dr. Phillippi had no financial conflicts to disclose. Dr. Krishna had no financial conflicts to disclose, but serves on the Editorial Advisory Board of Ob.Gyn News.
FROM OBSTETRICS & GYNECOLOGY
Obstetric care under threat in rural areas
Ready access to maternity services in rural areas is not a given, yet access to obstetric hospitals is associated with decreased rates of preterm birth and neonatal/perinatal mortality.
Little is known, however, about the availability of obstetric centers with respect to birth volume, geographic distribution among states, proximity of obstetric hospitals, and urban adjacency.
“This knowledge is fundamental to inform clinical and policy efforts to optimize perinatal regionalization, care delivery, and outcomes,” wrote Sara C. Handley, MD, MSCE, of the Roberts Center for Pediatric Research at the Children’s Hospital of Philadelphia, and colleagues, who undertook to fill that information gap in a study. It was published online Oct. 8 in JAMA Network Open.
Her group found birth volumes varied among obstetric hospitals, with many low-volume facilities located in rural, even isolated, areas, which suggests a need to ensure better access to perinatal care for women in these locations.
Using American Hospital Association data, the researchers examined the birth volumes and geographic distributions of 3,207 maternity hospitals from 2010 to 2018. In a cohort of 34,054,951 births, 56.8% occurred in high-volume obstetric facilities, and 37.4% in low-volume hospitals. Among the latter, 18.9% were isolated in location and not within 30 miles of any other obstetric hospital.
Most infants (19,327,487) were born in hospitals with more than 2,000 births per year, the study found, but a substantial 2,528,259, or 7.4%, were born in low-volume centers reporting 10 to 500 births annually.
“We were surprised by the number of low-volume hospitals and the number of births in low-volume hospitals,” Dr. Handley said in an interview. Many low-volume hospitals are in rural areas, which may require patients to drive long distances. These hospitals are at high risk of closure and such closures may further increase travel time.
Among low-volume hospitals, 23.9% were within the study proximity threshold of 30 miles of a hospital with more than 2,000 deliveries per year. “And when you’re in labor, even 30 miles is a long drive,” Dr. Handley said.
According to the authors, these findings highlight the need to balance care availability and sufficient patient volume by ensuring access and referral to high-quality perinatal care. They suggest perinatal care regionalization policies need review to improve maternal and infant outcomes.
But although the need is pressing, meeting it will not happen quickly, Dr. Handley said. “Change will require buy-in from multiple stakeholders and investment at many levels.”
The American College of Obstetrics and Gynecology has previously raised the alarm about general health disparities among women in underserved rural communities.
Anne L. Banfield, MD, director of women’s health services at Davis Medical Center in the mountain town of Elkins, W.V., is one obstetrician/gynecologist who is all too familiar with the problem of shrinking perinatal facilities. “Closures have impacted services,” she said in an interview, noting that one hospital in her region closed its birthing unit because of financial considerations. “The next closest facility to ours is 20 miles to the west and more than 60 miles in any other direction,” she said. “And geography can create challenges. Because we’re located in the mountains, it can take 2 hours to get to our facility.”
The hope is that these findings will inform discussions on regionalization policy for perinatal care to improve maternal and infant outcomes and address concerns about isolated obstetric hospitals, the authors said.
Dr. Banfield emphasized that obstetric facilities should be made a priority even if they’re less profitable than other services and not a major contributor to the bottom line. But that will require rethinking reimbursement models to align with community needs. “Everyone has a mother – no one springs from a pod – but the fact is, we’re not paying enough for maternal health care,” she said.
A top priority, she noted, is attracting sufficient staff; not only doctors, but also nurses and midwives with the skill sets required for perinatal care, which differ from those of general surgery and outpatient services. “We have to make financial changes to make this care feasible,” Dr. Banfield said.
In similar recent research, a study published online in the October issue of Health Affairs, showed that with rural hospitals facing increased financial distress, they may merge with other hospitals/systems, potentially reducing service lines that are less profitable or that duplicate services offered by the acquiring institution. Among those often on the chopping block is perinatal care.
“Our analysis of rural hospital discharge data found that merged hospitals were more likely than independent hospitals to eliminate maternal, neonatal, and also surgical care,” lead author Lan Liang, PhD, senior economist at the Agency for Healthcare Research and Quality (AHRQ) in Rockville, Md., said in an interview. This finding was consistent with previous AHRQ research using hospital self-reports, she added.
The study sample comprised 172 rural hospitals that merged during the period 2009-2016 in 32 states and 549 nonmerged comparison hospitals. In the premerger period, 74.5% of hospitals that merged provided maternal/neonatal care. This percentage decreased to 61.1% in the postmerger period. In contrast, the percentage of comparison hospitals providing these services remained stable during both periods (64.3% and 65.1%, respectively).
After weighting and adjusting for variables, the researchers found that from the premerger period to 1 year post merger, the percentage of hospitals providing these services decreased by 6.7 percentage points more for merged than for comparison hospitals (P = .06).
In the second year post merger, the percentage of hospitals providing maternal/neonatal services decreased by 7.2 percentage points more for merged than for comparison hospitals (P = .09).
“We did not, however, see a reduction in the volume of maternity services in rural communities, which suggests that women are just traveling farther to give birth,” Dr. Liang said.
Although mergers might salvage hospitals’ sustainability, the authors wrote, they do not necessarily mean that service lines are retained or that hospitals are as responsive to community needs as they were before the merger.
The analysis concluded that continuing access to maternal/neonatal care in rural areas is not a given. “Stakeholders, including payers, policy makers, and community-based organizations, need to monitor the availability of maternity services to ensure women have options in childbirth providers,” Dr. Liang said.
She and her associates called for payer-supported, multi-stakeholder initiatives to transform rural health care to be both financially sustainable and responsive to population needs.
The study by Dr. Handley and colleagues was supported by the National Institutes of Health (NIH) and the Eunice Shriver National Institute of Child Health and Human Development (NICHD). Dr. Handley reported grants from the NIH outside of the submitted work. Several coauthors reported grants from, variously, the NIH, the NICHD, and the Agency for Healthcare Research and Quality (AHRQ). The study by Dr. Liang and associates was supported by the AHRQ’s Center for Financing, Access, and Cost Trends, and the Healthcare Cost and Utilization Project. The authors disclosed no competing interests.
Ready access to maternity services in rural areas is not a given, yet access to obstetric hospitals is associated with decreased rates of preterm birth and neonatal/perinatal mortality.
Little is known, however, about the availability of obstetric centers with respect to birth volume, geographic distribution among states, proximity of obstetric hospitals, and urban adjacency.
“This knowledge is fundamental to inform clinical and policy efforts to optimize perinatal regionalization, care delivery, and outcomes,” wrote Sara C. Handley, MD, MSCE, of the Roberts Center for Pediatric Research at the Children’s Hospital of Philadelphia, and colleagues, who undertook to fill that information gap in a study. It was published online Oct. 8 in JAMA Network Open.
Her group found birth volumes varied among obstetric hospitals, with many low-volume facilities located in rural, even isolated, areas, which suggests a need to ensure better access to perinatal care for women in these locations.
Using American Hospital Association data, the researchers examined the birth volumes and geographic distributions of 3,207 maternity hospitals from 2010 to 2018. In a cohort of 34,054,951 births, 56.8% occurred in high-volume obstetric facilities, and 37.4% in low-volume hospitals. Among the latter, 18.9% were isolated in location and not within 30 miles of any other obstetric hospital.
Most infants (19,327,487) were born in hospitals with more than 2,000 births per year, the study found, but a substantial 2,528,259, or 7.4%, were born in low-volume centers reporting 10 to 500 births annually.
“We were surprised by the number of low-volume hospitals and the number of births in low-volume hospitals,” Dr. Handley said in an interview. Many low-volume hospitals are in rural areas, which may require patients to drive long distances. These hospitals are at high risk of closure and such closures may further increase travel time.
Among low-volume hospitals, 23.9% were within the study proximity threshold of 30 miles of a hospital with more than 2,000 deliveries per year. “And when you’re in labor, even 30 miles is a long drive,” Dr. Handley said.
According to the authors, these findings highlight the need to balance care availability and sufficient patient volume by ensuring access and referral to high-quality perinatal care. They suggest perinatal care regionalization policies need review to improve maternal and infant outcomes.
But although the need is pressing, meeting it will not happen quickly, Dr. Handley said. “Change will require buy-in from multiple stakeholders and investment at many levels.”
The American College of Obstetrics and Gynecology has previously raised the alarm about general health disparities among women in underserved rural communities.
Anne L. Banfield, MD, director of women’s health services at Davis Medical Center in the mountain town of Elkins, W.V., is one obstetrician/gynecologist who is all too familiar with the problem of shrinking perinatal facilities. “Closures have impacted services,” she said in an interview, noting that one hospital in her region closed its birthing unit because of financial considerations. “The next closest facility to ours is 20 miles to the west and more than 60 miles in any other direction,” she said. “And geography can create challenges. Because we’re located in the mountains, it can take 2 hours to get to our facility.”
The hope is that these findings will inform discussions on regionalization policy for perinatal care to improve maternal and infant outcomes and address concerns about isolated obstetric hospitals, the authors said.
Dr. Banfield emphasized that obstetric facilities should be made a priority even if they’re less profitable than other services and not a major contributor to the bottom line. But that will require rethinking reimbursement models to align with community needs. “Everyone has a mother – no one springs from a pod – but the fact is, we’re not paying enough for maternal health care,” she said.
A top priority, she noted, is attracting sufficient staff; not only doctors, but also nurses and midwives with the skill sets required for perinatal care, which differ from those of general surgery and outpatient services. “We have to make financial changes to make this care feasible,” Dr. Banfield said.
In similar recent research, a study published online in the October issue of Health Affairs, showed that with rural hospitals facing increased financial distress, they may merge with other hospitals/systems, potentially reducing service lines that are less profitable or that duplicate services offered by the acquiring institution. Among those often on the chopping block is perinatal care.
“Our analysis of rural hospital discharge data found that merged hospitals were more likely than independent hospitals to eliminate maternal, neonatal, and also surgical care,” lead author Lan Liang, PhD, senior economist at the Agency for Healthcare Research and Quality (AHRQ) in Rockville, Md., said in an interview. This finding was consistent with previous AHRQ research using hospital self-reports, she added.
The study sample comprised 172 rural hospitals that merged during the period 2009-2016 in 32 states and 549 nonmerged comparison hospitals. In the premerger period, 74.5% of hospitals that merged provided maternal/neonatal care. This percentage decreased to 61.1% in the postmerger period. In contrast, the percentage of comparison hospitals providing these services remained stable during both periods (64.3% and 65.1%, respectively).
After weighting and adjusting for variables, the researchers found that from the premerger period to 1 year post merger, the percentage of hospitals providing these services decreased by 6.7 percentage points more for merged than for comparison hospitals (P = .06).
In the second year post merger, the percentage of hospitals providing maternal/neonatal services decreased by 7.2 percentage points more for merged than for comparison hospitals (P = .09).
“We did not, however, see a reduction in the volume of maternity services in rural communities, which suggests that women are just traveling farther to give birth,” Dr. Liang said.
Although mergers might salvage hospitals’ sustainability, the authors wrote, they do not necessarily mean that service lines are retained or that hospitals are as responsive to community needs as they were before the merger.
The analysis concluded that continuing access to maternal/neonatal care in rural areas is not a given. “Stakeholders, including payers, policy makers, and community-based organizations, need to monitor the availability of maternity services to ensure women have options in childbirth providers,” Dr. Liang said.
She and her associates called for payer-supported, multi-stakeholder initiatives to transform rural health care to be both financially sustainable and responsive to population needs.
The study by Dr. Handley and colleagues was supported by the National Institutes of Health (NIH) and the Eunice Shriver National Institute of Child Health and Human Development (NICHD). Dr. Handley reported grants from the NIH outside of the submitted work. Several coauthors reported grants from, variously, the NIH, the NICHD, and the Agency for Healthcare Research and Quality (AHRQ). The study by Dr. Liang and associates was supported by the AHRQ’s Center for Financing, Access, and Cost Trends, and the Healthcare Cost and Utilization Project. The authors disclosed no competing interests.
Ready access to maternity services in rural areas is not a given, yet access to obstetric hospitals is associated with decreased rates of preterm birth and neonatal/perinatal mortality.
Little is known, however, about the availability of obstetric centers with respect to birth volume, geographic distribution among states, proximity of obstetric hospitals, and urban adjacency.
“This knowledge is fundamental to inform clinical and policy efforts to optimize perinatal regionalization, care delivery, and outcomes,” wrote Sara C. Handley, MD, MSCE, of the Roberts Center for Pediatric Research at the Children’s Hospital of Philadelphia, and colleagues, who undertook to fill that information gap in a study. It was published online Oct. 8 in JAMA Network Open.
Her group found birth volumes varied among obstetric hospitals, with many low-volume facilities located in rural, even isolated, areas, which suggests a need to ensure better access to perinatal care for women in these locations.
Using American Hospital Association data, the researchers examined the birth volumes and geographic distributions of 3,207 maternity hospitals from 2010 to 2018. In a cohort of 34,054,951 births, 56.8% occurred in high-volume obstetric facilities, and 37.4% in low-volume hospitals. Among the latter, 18.9% were isolated in location and not within 30 miles of any other obstetric hospital.
Most infants (19,327,487) were born in hospitals with more than 2,000 births per year, the study found, but a substantial 2,528,259, or 7.4%, were born in low-volume centers reporting 10 to 500 births annually.
“We were surprised by the number of low-volume hospitals and the number of births in low-volume hospitals,” Dr. Handley said in an interview. Many low-volume hospitals are in rural areas, which may require patients to drive long distances. These hospitals are at high risk of closure and such closures may further increase travel time.
Among low-volume hospitals, 23.9% were within the study proximity threshold of 30 miles of a hospital with more than 2,000 deliveries per year. “And when you’re in labor, even 30 miles is a long drive,” Dr. Handley said.
According to the authors, these findings highlight the need to balance care availability and sufficient patient volume by ensuring access and referral to high-quality perinatal care. They suggest perinatal care regionalization policies need review to improve maternal and infant outcomes.
But although the need is pressing, meeting it will not happen quickly, Dr. Handley said. “Change will require buy-in from multiple stakeholders and investment at many levels.”
The American College of Obstetrics and Gynecology has previously raised the alarm about general health disparities among women in underserved rural communities.
Anne L. Banfield, MD, director of women’s health services at Davis Medical Center in the mountain town of Elkins, W.V., is one obstetrician/gynecologist who is all too familiar with the problem of shrinking perinatal facilities. “Closures have impacted services,” she said in an interview, noting that one hospital in her region closed its birthing unit because of financial considerations. “The next closest facility to ours is 20 miles to the west and more than 60 miles in any other direction,” she said. “And geography can create challenges. Because we’re located in the mountains, it can take 2 hours to get to our facility.”
The hope is that these findings will inform discussions on regionalization policy for perinatal care to improve maternal and infant outcomes and address concerns about isolated obstetric hospitals, the authors said.
Dr. Banfield emphasized that obstetric facilities should be made a priority even if they’re less profitable than other services and not a major contributor to the bottom line. But that will require rethinking reimbursement models to align with community needs. “Everyone has a mother – no one springs from a pod – but the fact is, we’re not paying enough for maternal health care,” she said.
A top priority, she noted, is attracting sufficient staff; not only doctors, but also nurses and midwives with the skill sets required for perinatal care, which differ from those of general surgery and outpatient services. “We have to make financial changes to make this care feasible,” Dr. Banfield said.
In similar recent research, a study published online in the October issue of Health Affairs, showed that with rural hospitals facing increased financial distress, they may merge with other hospitals/systems, potentially reducing service lines that are less profitable or that duplicate services offered by the acquiring institution. Among those often on the chopping block is perinatal care.
“Our analysis of rural hospital discharge data found that merged hospitals were more likely than independent hospitals to eliminate maternal, neonatal, and also surgical care,” lead author Lan Liang, PhD, senior economist at the Agency for Healthcare Research and Quality (AHRQ) in Rockville, Md., said in an interview. This finding was consistent with previous AHRQ research using hospital self-reports, she added.
The study sample comprised 172 rural hospitals that merged during the period 2009-2016 in 32 states and 549 nonmerged comparison hospitals. In the premerger period, 74.5% of hospitals that merged provided maternal/neonatal care. This percentage decreased to 61.1% in the postmerger period. In contrast, the percentage of comparison hospitals providing these services remained stable during both periods (64.3% and 65.1%, respectively).
After weighting and adjusting for variables, the researchers found that from the premerger period to 1 year post merger, the percentage of hospitals providing these services decreased by 6.7 percentage points more for merged than for comparison hospitals (P = .06).
In the second year post merger, the percentage of hospitals providing maternal/neonatal services decreased by 7.2 percentage points more for merged than for comparison hospitals (P = .09).
“We did not, however, see a reduction in the volume of maternity services in rural communities, which suggests that women are just traveling farther to give birth,” Dr. Liang said.
Although mergers might salvage hospitals’ sustainability, the authors wrote, they do not necessarily mean that service lines are retained or that hospitals are as responsive to community needs as they were before the merger.
The analysis concluded that continuing access to maternal/neonatal care in rural areas is not a given. “Stakeholders, including payers, policy makers, and community-based organizations, need to monitor the availability of maternity services to ensure women have options in childbirth providers,” Dr. Liang said.
She and her associates called for payer-supported, multi-stakeholder initiatives to transform rural health care to be both financially sustainable and responsive to population needs.
The study by Dr. Handley and colleagues was supported by the National Institutes of Health (NIH) and the Eunice Shriver National Institute of Child Health and Human Development (NICHD). Dr. Handley reported grants from the NIH outside of the submitted work. Several coauthors reported grants from, variously, the NIH, the NICHD, and the Agency for Healthcare Research and Quality (AHRQ). The study by Dr. Liang and associates was supported by the AHRQ’s Center for Financing, Access, and Cost Trends, and the Healthcare Cost and Utilization Project. The authors disclosed no competing interests.
Better bone builder: High-intensity exercise vs. Pilates
An 8-month high-intensity resistance and impact training program (HiRIT, Onero) led to greater gains in lumbar spine bone mineral density (BMD) and leg/back strength than a low-intensity Pilates-based program (Buff Bones).
These findings are from the Medication and Exercise for Osteoporosis (MEDEX-OP) trial, which included 115 postmenopausal women with low bone mass. Patients were randomly assigned to attend either the HiRIT or Pilates-based exercise program. The participants attended supervised 45-min sessions twice weekly.
HiRIT was better than the low-intensity Pilates-based exercise program for enhancing bone mass, muscle strength, functional performance, and stature, the researchers reported. The low-intensity program did improve function, but to a lesser extent
Of the 115 participants, most (86) were not taking osteoporosis medicine. For the 29 women who were receiving it, the medication appeared to enhance the effect of exercise.
Melanie Fischbacher, PhD candidate, Griffith University, Gold Coast, Australia, presented these findings in an oral session at the annual meeting of the American Society for Bone and Mineral Research; the study was also published in the Journal of Bone and Mineral Research.
The study’s senior author, Belinda R. Beck, PhD, director of the Bone Clinic in Brisbane, Australia, developed the Onero HiRIT program and has licensed it to others in Australia.
“It is a very effective program and we have shown it can be undertaken safely, but it must be supervised because of the heavy weights and high-risk clientele,” Beck stressed to this news organization.
“This is not a program you should just hand to a patient and tell them to do in a gym,” she said.
“Both forms of exercise in our study were beneficial for functional outcomes but Onero improved back extensor strength, mobility and stature considerably more than Buff Bones,” Ms. Fischbacher said in an interview.
Nevertheless, “the contribution of functional capacity to risk of falling and fracture cannot be overstated, and bone medications do not address function,” she noted.
“More trials combining bone medication and bone-targeted exercise are needed,” the researchers concluded.
Compliance stands out, study supports high-intensity exercise
Kristen M. Beavers, PhD, MPH, RD, who was not involved with this research, told this news organization that participant compliance in the study really stands out.
“Compliance to an 8-month, 2 day/week high-intensity resistance training program among older women with low bone mass was quite good in this study [>80%], with very few adverse events reported,” said Dr. Beavers, of the department of health and exercise science, Wake Forest University, Winston Salem, N.C.
“A lot of individuals wouldn’t even consider recommending this type/intensity of exercise to this population, because they are worried it is too risky and/or the uptake will be low,” she said.
Although the benefit in BMD and strength wasn’t seen universally across all bone/muscle outcomes assessed, the findings do reinforce the idea that high-intensity exercise is more efficacious for bone health than low-intensity exercise, she noted.
“The possible additive effect of high-intensity exercise when combined with medication is worth confirming in larger, adequately designed/powered studies,” according to Dr. Beavers.
“The general consensus in the field is that higher-intensity exercise is more osteogenic than low-intensity exercise, but improving muscle mass, quality, and function (including balance) are also important to reduce the risk of falls, which is a major contributor to incident fracture,” she noted.
Exercise, even low-intensity exercise, reduces the risk for falls, as shown in a recent meta-analysis, she added. This is something antiresorptive medications don’t do.
Building on the LIFTMOR and LIFTMOR-M Trials
Previously, the Australian group showed that HiRIT is efficacious and safe for bone formation in individuals with low to very low bone mass – in postmenopausal women in the LIFTMOR study (J Bone Miner Res. 2017 Oct 4 .doi: 10.1002/jbmr.3284), and in men in the LIFTMOR-M study.
The current study compared two exercise programs. The researchers randomly assigned 86 women who were not taking antiresorptive medication to the high-intensity (42) or low-intensity (44) exercise program. They also assigned 29 women who were receiving antiresorptive medication to the high-intensity (15) or low-intensity (14) exercise program.
In the high-intensity exercise plus medication subgroup, the women were taking denosumab (12), risedronate (2) or alendronate (1). In the low-intensity exercise plus medication subgroup, the women were taking denosumab (9), risedronate (1), alendronate (3), or zoledronic acid (1).
The mean age of the women was 64-68 years. The mean lumbar spine T score was –1.5 to –2.3, and the mean femoral neck T score was –1.7 to –2.0 (determined by dual-energy x-ray absorptiometry) .
The HiRIT training program consisted of three free-weight resistance training exercises (deadlift, back squat, overhead press), one high-impact exercise (jump drop), and two balance exercises. The exercises varied each session.
The low-intensity training consisted of bone-specific Pilates-based exercises performed on the mat; standing weight-bearing exercise with 1-kg dumbbells; and impact exercises, such as heel drops and stomping.
At 8 months, compared with women in the low-intensity exercise program, those in the HiRIT program demonstrated greater improvement in lumbar spine BMD (1.9% vs. 0.1%) and stature (0.2 cm vs. 0.0 cm), muscle strength, and functional performance.
Functional performance improved with both exercise programs, but the HiRIT program led to greater leg and back muscle strength and better results in the five times sit-to-stand test (P < .05).
HiRIT plus bone medication improved BMD at the femoral neck and total hip, whereas HiRIT alone did not. Low-intensity exercise plus bone medication improved BMD at the lumbar spine and total hip, whereas low-intensity exercise alone did not.
The retention rate was 90%. The rate of exercise compliance was 83% in the high-intensity group and 82% in the low-intensity group.
Thirty falls were reported by 24 participants (21%). One fracture occurred in each exercise group. Three adverse events occurred in the low-intensity group, and four occurred in the high-intensity group.
Dr. Beck owns the Bone Clinic and sells licenses to the Onero program. The other researchers disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
An 8-month high-intensity resistance and impact training program (HiRIT, Onero) led to greater gains in lumbar spine bone mineral density (BMD) and leg/back strength than a low-intensity Pilates-based program (Buff Bones).
These findings are from the Medication and Exercise for Osteoporosis (MEDEX-OP) trial, which included 115 postmenopausal women with low bone mass. Patients were randomly assigned to attend either the HiRIT or Pilates-based exercise program. The participants attended supervised 45-min sessions twice weekly.
HiRIT was better than the low-intensity Pilates-based exercise program for enhancing bone mass, muscle strength, functional performance, and stature, the researchers reported. The low-intensity program did improve function, but to a lesser extent
Of the 115 participants, most (86) were not taking osteoporosis medicine. For the 29 women who were receiving it, the medication appeared to enhance the effect of exercise.
Melanie Fischbacher, PhD candidate, Griffith University, Gold Coast, Australia, presented these findings in an oral session at the annual meeting of the American Society for Bone and Mineral Research; the study was also published in the Journal of Bone and Mineral Research.
The study’s senior author, Belinda R. Beck, PhD, director of the Bone Clinic in Brisbane, Australia, developed the Onero HiRIT program and has licensed it to others in Australia.
“It is a very effective program and we have shown it can be undertaken safely, but it must be supervised because of the heavy weights and high-risk clientele,” Beck stressed to this news organization.
“This is not a program you should just hand to a patient and tell them to do in a gym,” she said.
“Both forms of exercise in our study were beneficial for functional outcomes but Onero improved back extensor strength, mobility and stature considerably more than Buff Bones,” Ms. Fischbacher said in an interview.
Nevertheless, “the contribution of functional capacity to risk of falling and fracture cannot be overstated, and bone medications do not address function,” she noted.
“More trials combining bone medication and bone-targeted exercise are needed,” the researchers concluded.
Compliance stands out, study supports high-intensity exercise
Kristen M. Beavers, PhD, MPH, RD, who was not involved with this research, told this news organization that participant compliance in the study really stands out.
“Compliance to an 8-month, 2 day/week high-intensity resistance training program among older women with low bone mass was quite good in this study [>80%], with very few adverse events reported,” said Dr. Beavers, of the department of health and exercise science, Wake Forest University, Winston Salem, N.C.
“A lot of individuals wouldn’t even consider recommending this type/intensity of exercise to this population, because they are worried it is too risky and/or the uptake will be low,” she said.
Although the benefit in BMD and strength wasn’t seen universally across all bone/muscle outcomes assessed, the findings do reinforce the idea that high-intensity exercise is more efficacious for bone health than low-intensity exercise, she noted.
“The possible additive effect of high-intensity exercise when combined with medication is worth confirming in larger, adequately designed/powered studies,” according to Dr. Beavers.
“The general consensus in the field is that higher-intensity exercise is more osteogenic than low-intensity exercise, but improving muscle mass, quality, and function (including balance) are also important to reduce the risk of falls, which is a major contributor to incident fracture,” she noted.
Exercise, even low-intensity exercise, reduces the risk for falls, as shown in a recent meta-analysis, she added. This is something antiresorptive medications don’t do.
Building on the LIFTMOR and LIFTMOR-M Trials
Previously, the Australian group showed that HiRIT is efficacious and safe for bone formation in individuals with low to very low bone mass – in postmenopausal women in the LIFTMOR study (J Bone Miner Res. 2017 Oct 4 .doi: 10.1002/jbmr.3284), and in men in the LIFTMOR-M study.
The current study compared two exercise programs. The researchers randomly assigned 86 women who were not taking antiresorptive medication to the high-intensity (42) or low-intensity (44) exercise program. They also assigned 29 women who were receiving antiresorptive medication to the high-intensity (15) or low-intensity (14) exercise program.
In the high-intensity exercise plus medication subgroup, the women were taking denosumab (12), risedronate (2) or alendronate (1). In the low-intensity exercise plus medication subgroup, the women were taking denosumab (9), risedronate (1), alendronate (3), or zoledronic acid (1).
The mean age of the women was 64-68 years. The mean lumbar spine T score was –1.5 to –2.3, and the mean femoral neck T score was –1.7 to –2.0 (determined by dual-energy x-ray absorptiometry) .
The HiRIT training program consisted of three free-weight resistance training exercises (deadlift, back squat, overhead press), one high-impact exercise (jump drop), and two balance exercises. The exercises varied each session.
The low-intensity training consisted of bone-specific Pilates-based exercises performed on the mat; standing weight-bearing exercise with 1-kg dumbbells; and impact exercises, such as heel drops and stomping.
At 8 months, compared with women in the low-intensity exercise program, those in the HiRIT program demonstrated greater improvement in lumbar spine BMD (1.9% vs. 0.1%) and stature (0.2 cm vs. 0.0 cm), muscle strength, and functional performance.
Functional performance improved with both exercise programs, but the HiRIT program led to greater leg and back muscle strength and better results in the five times sit-to-stand test (P < .05).
HiRIT plus bone medication improved BMD at the femoral neck and total hip, whereas HiRIT alone did not. Low-intensity exercise plus bone medication improved BMD at the lumbar spine and total hip, whereas low-intensity exercise alone did not.
The retention rate was 90%. The rate of exercise compliance was 83% in the high-intensity group and 82% in the low-intensity group.
Thirty falls were reported by 24 participants (21%). One fracture occurred in each exercise group. Three adverse events occurred in the low-intensity group, and four occurred in the high-intensity group.
Dr. Beck owns the Bone Clinic and sells licenses to the Onero program. The other researchers disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
An 8-month high-intensity resistance and impact training program (HiRIT, Onero) led to greater gains in lumbar spine bone mineral density (BMD) and leg/back strength than a low-intensity Pilates-based program (Buff Bones).
These findings are from the Medication and Exercise for Osteoporosis (MEDEX-OP) trial, which included 115 postmenopausal women with low bone mass. Patients were randomly assigned to attend either the HiRIT or Pilates-based exercise program. The participants attended supervised 45-min sessions twice weekly.
HiRIT was better than the low-intensity Pilates-based exercise program for enhancing bone mass, muscle strength, functional performance, and stature, the researchers reported. The low-intensity program did improve function, but to a lesser extent
Of the 115 participants, most (86) were not taking osteoporosis medicine. For the 29 women who were receiving it, the medication appeared to enhance the effect of exercise.
Melanie Fischbacher, PhD candidate, Griffith University, Gold Coast, Australia, presented these findings in an oral session at the annual meeting of the American Society for Bone and Mineral Research; the study was also published in the Journal of Bone and Mineral Research.
The study’s senior author, Belinda R. Beck, PhD, director of the Bone Clinic in Brisbane, Australia, developed the Onero HiRIT program and has licensed it to others in Australia.
“It is a very effective program and we have shown it can be undertaken safely, but it must be supervised because of the heavy weights and high-risk clientele,” Beck stressed to this news organization.
“This is not a program you should just hand to a patient and tell them to do in a gym,” she said.
“Both forms of exercise in our study were beneficial for functional outcomes but Onero improved back extensor strength, mobility and stature considerably more than Buff Bones,” Ms. Fischbacher said in an interview.
Nevertheless, “the contribution of functional capacity to risk of falling and fracture cannot be overstated, and bone medications do not address function,” she noted.
“More trials combining bone medication and bone-targeted exercise are needed,” the researchers concluded.
Compliance stands out, study supports high-intensity exercise
Kristen M. Beavers, PhD, MPH, RD, who was not involved with this research, told this news organization that participant compliance in the study really stands out.
“Compliance to an 8-month, 2 day/week high-intensity resistance training program among older women with low bone mass was quite good in this study [>80%], with very few adverse events reported,” said Dr. Beavers, of the department of health and exercise science, Wake Forest University, Winston Salem, N.C.
“A lot of individuals wouldn’t even consider recommending this type/intensity of exercise to this population, because they are worried it is too risky and/or the uptake will be low,” she said.
Although the benefit in BMD and strength wasn’t seen universally across all bone/muscle outcomes assessed, the findings do reinforce the idea that high-intensity exercise is more efficacious for bone health than low-intensity exercise, she noted.
“The possible additive effect of high-intensity exercise when combined with medication is worth confirming in larger, adequately designed/powered studies,” according to Dr. Beavers.
“The general consensus in the field is that higher-intensity exercise is more osteogenic than low-intensity exercise, but improving muscle mass, quality, and function (including balance) are also important to reduce the risk of falls, which is a major contributor to incident fracture,” she noted.
Exercise, even low-intensity exercise, reduces the risk for falls, as shown in a recent meta-analysis, she added. This is something antiresorptive medications don’t do.
Building on the LIFTMOR and LIFTMOR-M Trials
Previously, the Australian group showed that HiRIT is efficacious and safe for bone formation in individuals with low to very low bone mass – in postmenopausal women in the LIFTMOR study (J Bone Miner Res. 2017 Oct 4 .doi: 10.1002/jbmr.3284), and in men in the LIFTMOR-M study.
The current study compared two exercise programs. The researchers randomly assigned 86 women who were not taking antiresorptive medication to the high-intensity (42) or low-intensity (44) exercise program. They also assigned 29 women who were receiving antiresorptive medication to the high-intensity (15) or low-intensity (14) exercise program.
In the high-intensity exercise plus medication subgroup, the women were taking denosumab (12), risedronate (2) or alendronate (1). In the low-intensity exercise plus medication subgroup, the women were taking denosumab (9), risedronate (1), alendronate (3), or zoledronic acid (1).
The mean age of the women was 64-68 years. The mean lumbar spine T score was –1.5 to –2.3, and the mean femoral neck T score was –1.7 to –2.0 (determined by dual-energy x-ray absorptiometry) .
The HiRIT training program consisted of three free-weight resistance training exercises (deadlift, back squat, overhead press), one high-impact exercise (jump drop), and two balance exercises. The exercises varied each session.
The low-intensity training consisted of bone-specific Pilates-based exercises performed on the mat; standing weight-bearing exercise with 1-kg dumbbells; and impact exercises, such as heel drops and stomping.
At 8 months, compared with women in the low-intensity exercise program, those in the HiRIT program demonstrated greater improvement in lumbar spine BMD (1.9% vs. 0.1%) and stature (0.2 cm vs. 0.0 cm), muscle strength, and functional performance.
Functional performance improved with both exercise programs, but the HiRIT program led to greater leg and back muscle strength and better results in the five times sit-to-stand test (P < .05).
HiRIT plus bone medication improved BMD at the femoral neck and total hip, whereas HiRIT alone did not. Low-intensity exercise plus bone medication improved BMD at the lumbar spine and total hip, whereas low-intensity exercise alone did not.
The retention rate was 90%. The rate of exercise compliance was 83% in the high-intensity group and 82% in the low-intensity group.
Thirty falls were reported by 24 participants (21%). One fracture occurred in each exercise group. Three adverse events occurred in the low-intensity group, and four occurred in the high-intensity group.
Dr. Beck owns the Bone Clinic and sells licenses to the Onero program. The other researchers disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New approval in early breast cancer: First advance in 20 years
Abemaciclib had already been approved for use in the treatment of HR+, HER2– advanced or metastatic breast cancer.
Now it is also approved for use in HR+, HER2– early breast cancer for patients who have high-risk, node-positive disease and whose tumors have a Ki-67 score of 20% or higher, as determined by a U.S. Food and Drug Administration–approved test.
The FDA also approved the Ki-67 IHC MIB-1 pharmDx (Dako Omnis) assay for use as a companion diagnostic test.
This is the first CDK4/6 inhibitor to be approved for use in this patient population.
Approximately 70% of all breast cancers are of the HR+, HER2– subtype.
The approval is based on some of the results from the monarchE study, which was presented last year at the annual meeting of the European Society of Medical Oncology and was simultaneously published in the Journal of Clinical Oncology.
The results showed that the addition of abemaciclib to endocrine therapy (tamoxifen or aromatase inhibitors) significantly improved invasive disease-free survival (IDFS), which was defined on the basis of the length of time before breast cancer comes back, any new cancer develops, or death.
The 2-year IDFS rates were 92.2% with the combination vs. 88.7% for endocrine therapy alone for the overall patient population.
“This is the first time in more than 20 years that we have seen an advance in the adjuvant treatment of this form of breast cancer,” lead investigator Stephen Johnston, MD, PhD, from the Royal Marsden Hospital NHS Foundation Trust, London, said at the meeting, as reported at the time by this news organization.
Reacting to the findings, Giuseppe Curigliano, MD, PhD, head of the division of early drug development at the European Institute of Oncology, Milan, said, “This is a very important trial and the findings will change practice.”
He predicted that once the drug is approved for use in high-risk HR+, HER2– early breast cancer, “the new standard of care for these patients will be to add 2 years of abemaciclib to endocrine therapy.”
In a press release about the new approval from the manufacturer (Lilly), another investigator on the monarchE study, Sara M. Tolaney, MD, MPH, Harvard Medical School and the Dana-Farber Cancer Institute, Boston, agreed that the results are practice changing. She said that the combination of abemaciclib and endocrine therapy is a potential new standard of care for this patient population. “We are encouraged by the marked reduction in the risk of recurrence even beyond the 2-year treatment period in these patients, and I’m grateful to be able to offer this as a treatment option to my patients,” she said.
On Twitter, she commented that restricting the indication to patients who show Ki67 ≥20% is “interesting,” inasmuch as benefits were seen in patients with both low and high Ki67.
Hal Burstein, MD, from Dana-Farber, also found this detail “interesting, as Ki67 testing remains a very controversial topic and difficult to standardize.”
Replying, Pedro Exman, MD, from the Hospital Alemão Oswaldo Cruz, in São Paulo, said: “Does it make sense to approve only in a subset of patients based in a positive subgroup analysis of a positive ITT study that was not even described in the JCO publication?”
Other experts said they were eagerly awaiting further results, particularly on overall survival, from the monarchE trial. New data are due to be presented on Oct. 14 at an ESMO virtual plenary session.
Commenting late last year about these results, George W. Sledge Jr, MD, professor of medicine at Stanford University Medical Center, Palo Alto, Calif., said that the median follow-up time “is still quite short for a study of ER+ adjuvant therapy, where the majority of recurrences and deaths occur after 5 years in many studies.”
Consequently, “we still have a long way to go to understand the ultimate effects of CDK4/6 inhibition on early-stage ER+ breast cancer, particularly on late recurrences,” he told this news organization at the time.
Agreed, said C. Kent Osborne, MD, codirector of the San Antonio Breast Cancer Symposium and founding director of the Duncan Cancer Center at Baylor College of Medicine, Houston, Tex. The results are “very encouraging, especially in the subgroup of tumors with high proliferation” (identified by the K1-67 score).
However, Dr. Osborne also urged caution in the interpretation of the results, “given the still rather short follow-up, given that that ER+ disease is known for its persistent recurrence rate, even past 10 years.”
He also noted that “this class of inhibitors is likely cytostatic, rather than cytocidal, meaning that it blocks cell proliferation rather than killing the cells.” Questions therefore remain over whether the survival curves for combination therapy will come together with those for endocrine therapy alone once patients stop taking the drug.
Study details
The monarchE trial involved patients with HR+, HER2–, high-risk early breast cancer who had undergone surgery and, as indicated, radiotherapy and/or adjuvant/neoadjuvant chemotherapy. Patients with four or more positive nodes or one to three nodes and either tumors of size ≥5 cm, histologic grade 3, or central Ki-67 ≥20% were eligible; 5,637 patients were randomly assigned in a 1:1 ratio to receive standard-of-care adjuvant endocrine therapy (ET) with or without abemaciclib (150 mg twice daily for 2 years).
A preplanned interim analysis was carried out after 323 IDFS events were observed in the intent-to-treat population. The results, as published last year in the Journal of Clinical Oncology, show that abemaciclib plus ET yielded superior IDFS in comparison with ET alone (P = .01; hazard ratio, 0.75; 95% confidence interval, 0.60-0.93), with 2-year IDFS rates of 92.2% vs. 88.7%.
In the press release announcing the approval of the new indication, the manufacturer notes that the approval was based on the results from a subgroup of 2,003 patients whose tumors had a Ki-67 score of ≥20% and who were also at high risk for recurrence (≥four positive axillary lymph nodes [ALN], or one-three positive ALN with grade 3 disease and/or tumor size ≥5 cm).
There was a statistically significant improvement in IDFS for this prespecified subgroup of patients (HR, 0.643; 95% CI, 0.475-0.872; P = .0042).
With additional follow-up, conducted post hoc, the results showed a 37% decrease in the risk for breast cancer recurrence or death, compared with ET alone (HR, 0.626; 95% CI, 0.49-0.80) and an absolute benefit in IDFS event rate of 7.1% at 3 years. IDFS was 86.1% for abemaciclib plus ET vs. 79.0% for ET alone.
Adverse reactions from monarchE were consistent with the known safety profile for abemaciclib, the company noted. Safety and tolerability were evaluated in 5,591 patients. The most common adverse reactions reported (≥10%) with abemaciclib plus ET vs. ET alone were diarrhea (84% vs. 9%), infections (51% vs. 39%), neutropenia (46% vs. 6%), fatigue (41% vs. 18%), leukopenia (38% vs. 7%), nausea (30% vs. 9%), anemia (24% vs. 4%), headache (20% vs. 15%), vomiting (18% vs. 4.6%), stomatitis (14% vs. 5%), lymphopenia (14% vs. 3%), thrombocytopenia (13% vs. 2%), decreased appetite (12% vs. 2.4%), increased ALT (12% vs. 6%), increased AST (12% vs. 5%), dizziness (11% vs. 7%), rash (11% vs. 4.5%), and alopecia (11% vs. 2.7 %).
A version of this article first appeared on Medscape.com.
Abemaciclib had already been approved for use in the treatment of HR+, HER2– advanced or metastatic breast cancer.
Now it is also approved for use in HR+, HER2– early breast cancer for patients who have high-risk, node-positive disease and whose tumors have a Ki-67 score of 20% or higher, as determined by a U.S. Food and Drug Administration–approved test.
The FDA also approved the Ki-67 IHC MIB-1 pharmDx (Dako Omnis) assay for use as a companion diagnostic test.
This is the first CDK4/6 inhibitor to be approved for use in this patient population.
Approximately 70% of all breast cancers are of the HR+, HER2– subtype.
The approval is based on some of the results from the monarchE study, which was presented last year at the annual meeting of the European Society of Medical Oncology and was simultaneously published in the Journal of Clinical Oncology.
The results showed that the addition of abemaciclib to endocrine therapy (tamoxifen or aromatase inhibitors) significantly improved invasive disease-free survival (IDFS), which was defined on the basis of the length of time before breast cancer comes back, any new cancer develops, or death.
The 2-year IDFS rates were 92.2% with the combination vs. 88.7% for endocrine therapy alone for the overall patient population.
“This is the first time in more than 20 years that we have seen an advance in the adjuvant treatment of this form of breast cancer,” lead investigator Stephen Johnston, MD, PhD, from the Royal Marsden Hospital NHS Foundation Trust, London, said at the meeting, as reported at the time by this news organization.
Reacting to the findings, Giuseppe Curigliano, MD, PhD, head of the division of early drug development at the European Institute of Oncology, Milan, said, “This is a very important trial and the findings will change practice.”
He predicted that once the drug is approved for use in high-risk HR+, HER2– early breast cancer, “the new standard of care for these patients will be to add 2 years of abemaciclib to endocrine therapy.”
In a press release about the new approval from the manufacturer (Lilly), another investigator on the monarchE study, Sara M. Tolaney, MD, MPH, Harvard Medical School and the Dana-Farber Cancer Institute, Boston, agreed that the results are practice changing. She said that the combination of abemaciclib and endocrine therapy is a potential new standard of care for this patient population. “We are encouraged by the marked reduction in the risk of recurrence even beyond the 2-year treatment period in these patients, and I’m grateful to be able to offer this as a treatment option to my patients,” she said.
On Twitter, she commented that restricting the indication to patients who show Ki67 ≥20% is “interesting,” inasmuch as benefits were seen in patients with both low and high Ki67.
Hal Burstein, MD, from Dana-Farber, also found this detail “interesting, as Ki67 testing remains a very controversial topic and difficult to standardize.”
Replying, Pedro Exman, MD, from the Hospital Alemão Oswaldo Cruz, in São Paulo, said: “Does it make sense to approve only in a subset of patients based in a positive subgroup analysis of a positive ITT study that was not even described in the JCO publication?”
Other experts said they were eagerly awaiting further results, particularly on overall survival, from the monarchE trial. New data are due to be presented on Oct. 14 at an ESMO virtual plenary session.
Commenting late last year about these results, George W. Sledge Jr, MD, professor of medicine at Stanford University Medical Center, Palo Alto, Calif., said that the median follow-up time “is still quite short for a study of ER+ adjuvant therapy, where the majority of recurrences and deaths occur after 5 years in many studies.”
Consequently, “we still have a long way to go to understand the ultimate effects of CDK4/6 inhibition on early-stage ER+ breast cancer, particularly on late recurrences,” he told this news organization at the time.
Agreed, said C. Kent Osborne, MD, codirector of the San Antonio Breast Cancer Symposium and founding director of the Duncan Cancer Center at Baylor College of Medicine, Houston, Tex. The results are “very encouraging, especially in the subgroup of tumors with high proliferation” (identified by the K1-67 score).
However, Dr. Osborne also urged caution in the interpretation of the results, “given the still rather short follow-up, given that that ER+ disease is known for its persistent recurrence rate, even past 10 years.”
He also noted that “this class of inhibitors is likely cytostatic, rather than cytocidal, meaning that it blocks cell proliferation rather than killing the cells.” Questions therefore remain over whether the survival curves for combination therapy will come together with those for endocrine therapy alone once patients stop taking the drug.
Study details
The monarchE trial involved patients with HR+, HER2–, high-risk early breast cancer who had undergone surgery and, as indicated, radiotherapy and/or adjuvant/neoadjuvant chemotherapy. Patients with four or more positive nodes or one to three nodes and either tumors of size ≥5 cm, histologic grade 3, or central Ki-67 ≥20% were eligible; 5,637 patients were randomly assigned in a 1:1 ratio to receive standard-of-care adjuvant endocrine therapy (ET) with or without abemaciclib (150 mg twice daily for 2 years).
A preplanned interim analysis was carried out after 323 IDFS events were observed in the intent-to-treat population. The results, as published last year in the Journal of Clinical Oncology, show that abemaciclib plus ET yielded superior IDFS in comparison with ET alone (P = .01; hazard ratio, 0.75; 95% confidence interval, 0.60-0.93), with 2-year IDFS rates of 92.2% vs. 88.7%.
In the press release announcing the approval of the new indication, the manufacturer notes that the approval was based on the results from a subgroup of 2,003 patients whose tumors had a Ki-67 score of ≥20% and who were also at high risk for recurrence (≥four positive axillary lymph nodes [ALN], or one-three positive ALN with grade 3 disease and/or tumor size ≥5 cm).
There was a statistically significant improvement in IDFS for this prespecified subgroup of patients (HR, 0.643; 95% CI, 0.475-0.872; P = .0042).
With additional follow-up, conducted post hoc, the results showed a 37% decrease in the risk for breast cancer recurrence or death, compared with ET alone (HR, 0.626; 95% CI, 0.49-0.80) and an absolute benefit in IDFS event rate of 7.1% at 3 years. IDFS was 86.1% for abemaciclib plus ET vs. 79.0% for ET alone.
Adverse reactions from monarchE were consistent with the known safety profile for abemaciclib, the company noted. Safety and tolerability were evaluated in 5,591 patients. The most common adverse reactions reported (≥10%) with abemaciclib plus ET vs. ET alone were diarrhea (84% vs. 9%), infections (51% vs. 39%), neutropenia (46% vs. 6%), fatigue (41% vs. 18%), leukopenia (38% vs. 7%), nausea (30% vs. 9%), anemia (24% vs. 4%), headache (20% vs. 15%), vomiting (18% vs. 4.6%), stomatitis (14% vs. 5%), lymphopenia (14% vs. 3%), thrombocytopenia (13% vs. 2%), decreased appetite (12% vs. 2.4%), increased ALT (12% vs. 6%), increased AST (12% vs. 5%), dizziness (11% vs. 7%), rash (11% vs. 4.5%), and alopecia (11% vs. 2.7 %).
A version of this article first appeared on Medscape.com.
Abemaciclib had already been approved for use in the treatment of HR+, HER2– advanced or metastatic breast cancer.
Now it is also approved for use in HR+, HER2– early breast cancer for patients who have high-risk, node-positive disease and whose tumors have a Ki-67 score of 20% or higher, as determined by a U.S. Food and Drug Administration–approved test.
The FDA also approved the Ki-67 IHC MIB-1 pharmDx (Dako Omnis) assay for use as a companion diagnostic test.
This is the first CDK4/6 inhibitor to be approved for use in this patient population.
Approximately 70% of all breast cancers are of the HR+, HER2– subtype.
The approval is based on some of the results from the monarchE study, which was presented last year at the annual meeting of the European Society of Medical Oncology and was simultaneously published in the Journal of Clinical Oncology.
The results showed that the addition of abemaciclib to endocrine therapy (tamoxifen or aromatase inhibitors) significantly improved invasive disease-free survival (IDFS), which was defined on the basis of the length of time before breast cancer comes back, any new cancer develops, or death.
The 2-year IDFS rates were 92.2% with the combination vs. 88.7% for endocrine therapy alone for the overall patient population.
“This is the first time in more than 20 years that we have seen an advance in the adjuvant treatment of this form of breast cancer,” lead investigator Stephen Johnston, MD, PhD, from the Royal Marsden Hospital NHS Foundation Trust, London, said at the meeting, as reported at the time by this news organization.
Reacting to the findings, Giuseppe Curigliano, MD, PhD, head of the division of early drug development at the European Institute of Oncology, Milan, said, “This is a very important trial and the findings will change practice.”
He predicted that once the drug is approved for use in high-risk HR+, HER2– early breast cancer, “the new standard of care for these patients will be to add 2 years of abemaciclib to endocrine therapy.”
In a press release about the new approval from the manufacturer (Lilly), another investigator on the monarchE study, Sara M. Tolaney, MD, MPH, Harvard Medical School and the Dana-Farber Cancer Institute, Boston, agreed that the results are practice changing. She said that the combination of abemaciclib and endocrine therapy is a potential new standard of care for this patient population. “We are encouraged by the marked reduction in the risk of recurrence even beyond the 2-year treatment period in these patients, and I’m grateful to be able to offer this as a treatment option to my patients,” she said.
On Twitter, she commented that restricting the indication to patients who show Ki67 ≥20% is “interesting,” inasmuch as benefits were seen in patients with both low and high Ki67.
Hal Burstein, MD, from Dana-Farber, also found this detail “interesting, as Ki67 testing remains a very controversial topic and difficult to standardize.”
Replying, Pedro Exman, MD, from the Hospital Alemão Oswaldo Cruz, in São Paulo, said: “Does it make sense to approve only in a subset of patients based in a positive subgroup analysis of a positive ITT study that was not even described in the JCO publication?”
Other experts said they were eagerly awaiting further results, particularly on overall survival, from the monarchE trial. New data are due to be presented on Oct. 14 at an ESMO virtual plenary session.
Commenting late last year about these results, George W. Sledge Jr, MD, professor of medicine at Stanford University Medical Center, Palo Alto, Calif., said that the median follow-up time “is still quite short for a study of ER+ adjuvant therapy, where the majority of recurrences and deaths occur after 5 years in many studies.”
Consequently, “we still have a long way to go to understand the ultimate effects of CDK4/6 inhibition on early-stage ER+ breast cancer, particularly on late recurrences,” he told this news organization at the time.
Agreed, said C. Kent Osborne, MD, codirector of the San Antonio Breast Cancer Symposium and founding director of the Duncan Cancer Center at Baylor College of Medicine, Houston, Tex. The results are “very encouraging, especially in the subgroup of tumors with high proliferation” (identified by the K1-67 score).
However, Dr. Osborne also urged caution in the interpretation of the results, “given the still rather short follow-up, given that that ER+ disease is known for its persistent recurrence rate, even past 10 years.”
He also noted that “this class of inhibitors is likely cytostatic, rather than cytocidal, meaning that it blocks cell proliferation rather than killing the cells.” Questions therefore remain over whether the survival curves for combination therapy will come together with those for endocrine therapy alone once patients stop taking the drug.
Study details
The monarchE trial involved patients with HR+, HER2–, high-risk early breast cancer who had undergone surgery and, as indicated, radiotherapy and/or adjuvant/neoadjuvant chemotherapy. Patients with four or more positive nodes or one to three nodes and either tumors of size ≥5 cm, histologic grade 3, or central Ki-67 ≥20% were eligible; 5,637 patients were randomly assigned in a 1:1 ratio to receive standard-of-care adjuvant endocrine therapy (ET) with or without abemaciclib (150 mg twice daily for 2 years).
A preplanned interim analysis was carried out after 323 IDFS events were observed in the intent-to-treat population. The results, as published last year in the Journal of Clinical Oncology, show that abemaciclib plus ET yielded superior IDFS in comparison with ET alone (P = .01; hazard ratio, 0.75; 95% confidence interval, 0.60-0.93), with 2-year IDFS rates of 92.2% vs. 88.7%.
In the press release announcing the approval of the new indication, the manufacturer notes that the approval was based on the results from a subgroup of 2,003 patients whose tumors had a Ki-67 score of ≥20% and who were also at high risk for recurrence (≥four positive axillary lymph nodes [ALN], or one-three positive ALN with grade 3 disease and/or tumor size ≥5 cm).
There was a statistically significant improvement in IDFS for this prespecified subgroup of patients (HR, 0.643; 95% CI, 0.475-0.872; P = .0042).
With additional follow-up, conducted post hoc, the results showed a 37% decrease in the risk for breast cancer recurrence or death, compared with ET alone (HR, 0.626; 95% CI, 0.49-0.80) and an absolute benefit in IDFS event rate of 7.1% at 3 years. IDFS was 86.1% for abemaciclib plus ET vs. 79.0% for ET alone.
Adverse reactions from monarchE were consistent with the known safety profile for abemaciclib, the company noted. Safety and tolerability were evaluated in 5,591 patients. The most common adverse reactions reported (≥10%) with abemaciclib plus ET vs. ET alone were diarrhea (84% vs. 9%), infections (51% vs. 39%), neutropenia (46% vs. 6%), fatigue (41% vs. 18%), leukopenia (38% vs. 7%), nausea (30% vs. 9%), anemia (24% vs. 4%), headache (20% vs. 15%), vomiting (18% vs. 4.6%), stomatitis (14% vs. 5%), lymphopenia (14% vs. 3%), thrombocytopenia (13% vs. 2%), decreased appetite (12% vs. 2.4%), increased ALT (12% vs. 6%), increased AST (12% vs. 5%), dizziness (11% vs. 7%), rash (11% vs. 4.5%), and alopecia (11% vs. 2.7 %).
A version of this article first appeared on Medscape.com.
Oral PTH shows promise for osteoporosis in early phase 2 study
An investigational oral form of parathyroid hormone (PTH 1-34), EB 613 (Entera Bio) met its primary efficacy outcome in a phase 2 dosing study involving postmenopausal women with low bone mineral density (BMD).
The adverse effect profile of the drug was similar to that of the injectable PTH 1-34 teriparatide (Forteo), which is approved for osteoporosis.
Arthur C. Santora, MD, chief medical officer, Entera Bio, presented 6-month findings from the study during an oral session at the annual meeting of the American Society of Bone and Mineral Research. The 3-month findings from the study were reported as a poster.
If the drug demonstrates efficacy and safety in larger phase 3 trials, it could be the first oral bone-building (anabolic) therapy for osteoporosis.
Clifford J. Rosen, MD, PhD, who was not involved with the research, told this news organization: “I think this is an intriguing study.” The most likely patients for oral PTH, he added, “are those that have osteoporosis, previous fracture, or very low BMD, particularly those unlikely or unwilling to take bisphosphonates.”
However, “this is very early in the process before this drug could come to market,” cautioned Dr. Rosen, who is director of the Center for Clinical and Translational Research, Maine Medical Research Institute, Scarborough.
“Much more data on efficacy are required at 12 and 24 months for phase 2, and then a full phase 3 [clinical trial] with high-risk fracture patients,” he said.
The company is seeking input from the Food and Drug Administration to develop the protocol for a phase 3 trial. They expect to start this trial in 2022 at sites in the United States, Europe, and Israel, Dr. Santora said.
Primary outcome met
The study randomly assigned 161 postmenopausal women with osteoporosis or low BMD to receive placebo or the investigational oral PTH for 6 months.
Compared with women who received placebo, those who received the study drug experienced a significantly greater increase in the bone formation marker procollagen type I N-terminal propeptide (P1NP) from baseline to 3 months, thereby meeting the study’s primary outcome.
In secondary outcomes, women who received the 2.5-mg/d dose experienced a similar 6-month increase in BMD at the spine and greater increases in BMD at the total hip and femoral neck than those who received injectable teriparatide, Dr. Santora reported.
“The study’s key takeaway is that a once-daily oral PTH [tablet] has the potential to produce the same BMD effects as subcutaneous injections of PTH,” he said in an interview.
Additionally, “the drug was well tolerated when the dose was titrated by adding additional tablets, which suggests that the dose can be tailored to each patient,” he said.
Other study findings
Injectable teriparatide reduces the risk for vertebral fractures by up to 80%, Dr. Santora noted, but the fact that the drug must be administered by injection may deter some older patients from using it.
The company developed an oral form of biosynthetic human PTH with a proprietary drug delivery.
The researchers conducted the phase 2 study at four sites in Israel between June 2019 and May 2021. They enrolled women aged 50 years and older who had entered menopause at least 3 years earlier and who had osteoporosis or low BMD.
Forty-three women received placebo, and the others received oral PTH at doses of 0.5 mg/d (n = 25), 1.0 mg/d (n = 29), 1.5 mg/d (n = 28), 2.5 mg/d (n = 19), or at a dose that was titrated up to 2.5 mg/d starting at 1.5 mg/d for month 1, then 2 mg/d for month 2, and then 2.5 mg/d for months 3 to 6 (n = 17).
The mean age of the patients was 61 years, the mean body mass index was 25-27 kg/m2, and the mean T score at the spine of –2.2 to –2.45.
Among the women who received 2.5 mg/d of oral PTH for the full 6 months, serum levels of the bone resorption marker C-terminal telopeptide of type I collagen (CTX) decreased 21% from baseline to 6 months, and serum levels of P1NP increased at month 1 and then decreased to baseline by month 6.
The women who received 2.5 mg/d of oral PTH for the full 6 months also demonstrated significantly greater increases in BMD at the lumbar spine (3.8%), total hip (1.4%), and femoral neck (2.4%), compared with women who received placebo.
The safety profile of oral PTH was consistent with that of subcutaneous PTH. Patients experienced headache, nausea, presyncope, and dizziness; there were no treatment-emergent hypercalcemia adverse events.
A few ‘unexpected findings’
Suzanne M. Jan De Beur, MD, outgoing ASBMR president, said, “Oral PTH appeared to increase BMD by [dual-energy x-ray absorptiometry] at the lumbar spine effectively and to a similar degree as teriparatide in previous studies.”
She identified two unexpected findings.
“There were increases in BMD by DXA at the femoral neck and total hip at 6 months that were [greater than those] seen in previous trials of teriparatide. Second, markers of bone resorption (CTX) decreased at 6 months, and this is in stark contrast to the increases observed with teriparatide treatment,” she noted in an interview.
Dr. Rosen also noted that “the decrease in CTX is very unusual for PTH and difficult to explain.” He added: “P1NP, a marker of bone formation, was not increased.”
Dr. Jan de Beur continued: “Teriparatide (PTH1-34) and abaloparatide are effective anabolic agents that we use to treat patients with high risk of osteoporotic fracture. Although effective, the burden of daily subcutaneous injection can be a barrier for older individuals, those with poor dexterity, and those that are averse to self-injection.
“Taken together, these results appear promising, that oral PTH may prove to be an effective anabolic agent for osteoporosis treatment,” she summarized.
She stressed that a larger phase 3 study is needed to demonstrate safety and efficacy.
The study was funded by Entera Bio. Dr. Santora is chief medical officer of Entera Bio.
A version of this article first appeared on Medscape.com .
An investigational oral form of parathyroid hormone (PTH 1-34), EB 613 (Entera Bio) met its primary efficacy outcome in a phase 2 dosing study involving postmenopausal women with low bone mineral density (BMD).
The adverse effect profile of the drug was similar to that of the injectable PTH 1-34 teriparatide (Forteo), which is approved for osteoporosis.
Arthur C. Santora, MD, chief medical officer, Entera Bio, presented 6-month findings from the study during an oral session at the annual meeting of the American Society of Bone and Mineral Research. The 3-month findings from the study were reported as a poster.
If the drug demonstrates efficacy and safety in larger phase 3 trials, it could be the first oral bone-building (anabolic) therapy for osteoporosis.
Clifford J. Rosen, MD, PhD, who was not involved with the research, told this news organization: “I think this is an intriguing study.” The most likely patients for oral PTH, he added, “are those that have osteoporosis, previous fracture, or very low BMD, particularly those unlikely or unwilling to take bisphosphonates.”
However, “this is very early in the process before this drug could come to market,” cautioned Dr. Rosen, who is director of the Center for Clinical and Translational Research, Maine Medical Research Institute, Scarborough.
“Much more data on efficacy are required at 12 and 24 months for phase 2, and then a full phase 3 [clinical trial] with high-risk fracture patients,” he said.
The company is seeking input from the Food and Drug Administration to develop the protocol for a phase 3 trial. They expect to start this trial in 2022 at sites in the United States, Europe, and Israel, Dr. Santora said.
Primary outcome met
The study randomly assigned 161 postmenopausal women with osteoporosis or low BMD to receive placebo or the investigational oral PTH for 6 months.
Compared with women who received placebo, those who received the study drug experienced a significantly greater increase in the bone formation marker procollagen type I N-terminal propeptide (P1NP) from baseline to 3 months, thereby meeting the study’s primary outcome.
In secondary outcomes, women who received the 2.5-mg/d dose experienced a similar 6-month increase in BMD at the spine and greater increases in BMD at the total hip and femoral neck than those who received injectable teriparatide, Dr. Santora reported.
“The study’s key takeaway is that a once-daily oral PTH [tablet] has the potential to produce the same BMD effects as subcutaneous injections of PTH,” he said in an interview.
Additionally, “the drug was well tolerated when the dose was titrated by adding additional tablets, which suggests that the dose can be tailored to each patient,” he said.
Other study findings
Injectable teriparatide reduces the risk for vertebral fractures by up to 80%, Dr. Santora noted, but the fact that the drug must be administered by injection may deter some older patients from using it.
The company developed an oral form of biosynthetic human PTH with a proprietary drug delivery.
The researchers conducted the phase 2 study at four sites in Israel between June 2019 and May 2021. They enrolled women aged 50 years and older who had entered menopause at least 3 years earlier and who had osteoporosis or low BMD.
Forty-three women received placebo, and the others received oral PTH at doses of 0.5 mg/d (n = 25), 1.0 mg/d (n = 29), 1.5 mg/d (n = 28), 2.5 mg/d (n = 19), or at a dose that was titrated up to 2.5 mg/d starting at 1.5 mg/d for month 1, then 2 mg/d for month 2, and then 2.5 mg/d for months 3 to 6 (n = 17).
The mean age of the patients was 61 years, the mean body mass index was 25-27 kg/m2, and the mean T score at the spine of –2.2 to –2.45.
Among the women who received 2.5 mg/d of oral PTH for the full 6 months, serum levels of the bone resorption marker C-terminal telopeptide of type I collagen (CTX) decreased 21% from baseline to 6 months, and serum levels of P1NP increased at month 1 and then decreased to baseline by month 6.
The women who received 2.5 mg/d of oral PTH for the full 6 months also demonstrated significantly greater increases in BMD at the lumbar spine (3.8%), total hip (1.4%), and femoral neck (2.4%), compared with women who received placebo.
The safety profile of oral PTH was consistent with that of subcutaneous PTH. Patients experienced headache, nausea, presyncope, and dizziness; there were no treatment-emergent hypercalcemia adverse events.
A few ‘unexpected findings’
Suzanne M. Jan De Beur, MD, outgoing ASBMR president, said, “Oral PTH appeared to increase BMD by [dual-energy x-ray absorptiometry] at the lumbar spine effectively and to a similar degree as teriparatide in previous studies.”
She identified two unexpected findings.
“There were increases in BMD by DXA at the femoral neck and total hip at 6 months that were [greater than those] seen in previous trials of teriparatide. Second, markers of bone resorption (CTX) decreased at 6 months, and this is in stark contrast to the increases observed with teriparatide treatment,” she noted in an interview.
Dr. Rosen also noted that “the decrease in CTX is very unusual for PTH and difficult to explain.” He added: “P1NP, a marker of bone formation, was not increased.”
Dr. Jan de Beur continued: “Teriparatide (PTH1-34) and abaloparatide are effective anabolic agents that we use to treat patients with high risk of osteoporotic fracture. Although effective, the burden of daily subcutaneous injection can be a barrier for older individuals, those with poor dexterity, and those that are averse to self-injection.
“Taken together, these results appear promising, that oral PTH may prove to be an effective anabolic agent for osteoporosis treatment,” she summarized.
She stressed that a larger phase 3 study is needed to demonstrate safety and efficacy.
The study was funded by Entera Bio. Dr. Santora is chief medical officer of Entera Bio.
A version of this article first appeared on Medscape.com .
An investigational oral form of parathyroid hormone (PTH 1-34), EB 613 (Entera Bio) met its primary efficacy outcome in a phase 2 dosing study involving postmenopausal women with low bone mineral density (BMD).
The adverse effect profile of the drug was similar to that of the injectable PTH 1-34 teriparatide (Forteo), which is approved for osteoporosis.
Arthur C. Santora, MD, chief medical officer, Entera Bio, presented 6-month findings from the study during an oral session at the annual meeting of the American Society of Bone and Mineral Research. The 3-month findings from the study were reported as a poster.
If the drug demonstrates efficacy and safety in larger phase 3 trials, it could be the first oral bone-building (anabolic) therapy for osteoporosis.
Clifford J. Rosen, MD, PhD, who was not involved with the research, told this news organization: “I think this is an intriguing study.” The most likely patients for oral PTH, he added, “are those that have osteoporosis, previous fracture, or very low BMD, particularly those unlikely or unwilling to take bisphosphonates.”
However, “this is very early in the process before this drug could come to market,” cautioned Dr. Rosen, who is director of the Center for Clinical and Translational Research, Maine Medical Research Institute, Scarborough.
“Much more data on efficacy are required at 12 and 24 months for phase 2, and then a full phase 3 [clinical trial] with high-risk fracture patients,” he said.
The company is seeking input from the Food and Drug Administration to develop the protocol for a phase 3 trial. They expect to start this trial in 2022 at sites in the United States, Europe, and Israel, Dr. Santora said.
Primary outcome met
The study randomly assigned 161 postmenopausal women with osteoporosis or low BMD to receive placebo or the investigational oral PTH for 6 months.
Compared with women who received placebo, those who received the study drug experienced a significantly greater increase in the bone formation marker procollagen type I N-terminal propeptide (P1NP) from baseline to 3 months, thereby meeting the study’s primary outcome.
In secondary outcomes, women who received the 2.5-mg/d dose experienced a similar 6-month increase in BMD at the spine and greater increases in BMD at the total hip and femoral neck than those who received injectable teriparatide, Dr. Santora reported.
“The study’s key takeaway is that a once-daily oral PTH [tablet] has the potential to produce the same BMD effects as subcutaneous injections of PTH,” he said in an interview.
Additionally, “the drug was well tolerated when the dose was titrated by adding additional tablets, which suggests that the dose can be tailored to each patient,” he said.
Other study findings
Injectable teriparatide reduces the risk for vertebral fractures by up to 80%, Dr. Santora noted, but the fact that the drug must be administered by injection may deter some older patients from using it.
The company developed an oral form of biosynthetic human PTH with a proprietary drug delivery.
The researchers conducted the phase 2 study at four sites in Israel between June 2019 and May 2021. They enrolled women aged 50 years and older who had entered menopause at least 3 years earlier and who had osteoporosis or low BMD.
Forty-three women received placebo, and the others received oral PTH at doses of 0.5 mg/d (n = 25), 1.0 mg/d (n = 29), 1.5 mg/d (n = 28), 2.5 mg/d (n = 19), or at a dose that was titrated up to 2.5 mg/d starting at 1.5 mg/d for month 1, then 2 mg/d for month 2, and then 2.5 mg/d for months 3 to 6 (n = 17).
The mean age of the patients was 61 years, the mean body mass index was 25-27 kg/m2, and the mean T score at the spine of –2.2 to –2.45.
Among the women who received 2.5 mg/d of oral PTH for the full 6 months, serum levels of the bone resorption marker C-terminal telopeptide of type I collagen (CTX) decreased 21% from baseline to 6 months, and serum levels of P1NP increased at month 1 and then decreased to baseline by month 6.
The women who received 2.5 mg/d of oral PTH for the full 6 months also demonstrated significantly greater increases in BMD at the lumbar spine (3.8%), total hip (1.4%), and femoral neck (2.4%), compared with women who received placebo.
The safety profile of oral PTH was consistent with that of subcutaneous PTH. Patients experienced headache, nausea, presyncope, and dizziness; there were no treatment-emergent hypercalcemia adverse events.
A few ‘unexpected findings’
Suzanne M. Jan De Beur, MD, outgoing ASBMR president, said, “Oral PTH appeared to increase BMD by [dual-energy x-ray absorptiometry] at the lumbar spine effectively and to a similar degree as teriparatide in previous studies.”
She identified two unexpected findings.
“There were increases in BMD by DXA at the femoral neck and total hip at 6 months that were [greater than those] seen in previous trials of teriparatide. Second, markers of bone resorption (CTX) decreased at 6 months, and this is in stark contrast to the increases observed with teriparatide treatment,” she noted in an interview.
Dr. Rosen also noted that “the decrease in CTX is very unusual for PTH and difficult to explain.” He added: “P1NP, a marker of bone formation, was not increased.”
Dr. Jan de Beur continued: “Teriparatide (PTH1-34) and abaloparatide are effective anabolic agents that we use to treat patients with high risk of osteoporotic fracture. Although effective, the burden of daily subcutaneous injection can be a barrier for older individuals, those with poor dexterity, and those that are averse to self-injection.
“Taken together, these results appear promising, that oral PTH may prove to be an effective anabolic agent for osteoporosis treatment,” she summarized.
She stressed that a larger phase 3 study is needed to demonstrate safety and efficacy.
The study was funded by Entera Bio. Dr. Santora is chief medical officer of Entera Bio.
A version of this article first appeared on Medscape.com .
Homicide remains a top cause of maternal mortality
The prevalence of homicide was 16% higher in pregnant women or postpartum women than nonpregnant or nonpostpartum women in the United States, according to 2018 and 2019 mortality data from the National Center for Health Statistics.
Homicide has long been identified as a leading cause of death during pregnancy, but homicide is not counted in estimates of maternal mortality, nor is it emphasized as a target for prevention and intervention, wrote Maeve Wallace, PhD, of Tulane University, New Orleans, and colleagues.
Data on maternal mortality (defined as “death while pregnant or within 42 days of the end of pregnancy from causes related to or aggravated by pregnancy”) were limited until the addition of pregnancy to the U.S. Standard Certificate of Death in 2003; all 50 states had adopted it by 2018, the researchers noted.
In a study published in Obstetrics & Gynecology, the researchers analyzed the first 2 years of nationally available data to identify pregnancy-associated mortality and characterize other risk factors such as age and race.
The researchers identified 4,705 female homicides in 2018 and 2019. Of these, 273 (5.8%) occurred in women who were pregnant or within a year of the end of pregnancy. Approximately half (50.2%) of the pregnant or postpartum victims were non-Hispanic Black, 30% were non-Hispanic white, 9.5% were Hispanic, and 10.3% were other races; approximately one-third (35.5%) were in the 20- to 24-year age group.
Overall, the ratio was 3.62 homicides per 100,000 live births among females who were either pregnant or within 1 year post partum, compared to 3.12 homicides per 100,000 live births in nonpregnant, nonpostpartum females aged 10-44 years (P = .05).
“Patterns were similar in further stratification by both race and age such that pregnancy was associated with more than a doubled risk of homicide among girls and women aged 10–24 in both the non-Hispanic White and non-Hispanic Black populations,” the researchers wrote.
The findings are consistent with previous studies, which “implicates health and social system failures. Although we are unable to directly evaluate the involvement of intimate partner violence (IPV) in this report, we did find that a majority of pregnancy-associated homicides occurred in the home, implicating the likelihood of involvement by persons known to the victim,” they noted. In addition, the data showed that approximately 70% of the incidents of homicide in pregnant and postpartum women involved a firearm, an increase over previous estimates.
The study findings were limited by several factors including the lack of circumstantial information and incomplete data on victim characteristics, the researchers noted. Other key limitations included the potential for false-positives and false-negatives when recording pregnancy status, which could lead to underestimates of pregnancy-associated homicides, and the lack of data on pregnancy outcomes for women who experienced live birth, abortion, or miscarriage within a year of death.
However, the results highlight the need for increased awareness and training of physicians in completing the pregnancy checkbox on death certificates, and the need for action on recommendations and interventions to prevent maternal deaths from homicide, they emphasized.
“Although encouraging, a commitment to the actual implementation of policies and investments known to be effective at protecting and the promoting the health and safety of girls and women must follow,” they concluded.
Data highlight disparities
“This study could not be done effectively prior to now, as the adoption of the pregnancy checkbox on the U.S. Standard Certificate of Death was only available in all 50 states as of 2018,” Sarah W. Prager, MD, of the University of Washington, Seattle, said in an interview.
“This study also demonstrates what was already known, which is that pregnancy is a high-risk time period for intimate partner violence, including homicide. The differences in homicide rates based on race and ethnicity also highlight the clear disparities in maternal mortality in the U.S. that are attributable to racism. There is more attention being paid to maternal mortality and the differential experience based on race, and this demonstrates that simply addressing medical management during pregnancy is not enough – we need to address root causes of racism if we truly want to reduce maternal mortality,” Dr. Prager said.
“The primary take-home message for clinicians is to ascertain safety from every patient, and to try to reduce the impacts of racism on health care for patients, especially during pregnancy,” she said.
Although more detailed records would help with elucidating causes versus associations, “more research is not the answer,” Dr. Prager stated. “The real solution here is to have better gun safety laws, and to put significant resources toward reducing the impacts of racism on health care and our society.”
The study was supported by the Eunice Kennedy Shriver National Institute of Child Health and Human Development. The researchers had no financial conflicts to disclose. Dr. Prager had no financial conflicts to disclose, but serves on the editorial advisory board of Ob.Gyn News.
The prevalence of homicide was 16% higher in pregnant women or postpartum women than nonpregnant or nonpostpartum women in the United States, according to 2018 and 2019 mortality data from the National Center for Health Statistics.
Homicide has long been identified as a leading cause of death during pregnancy, but homicide is not counted in estimates of maternal mortality, nor is it emphasized as a target for prevention and intervention, wrote Maeve Wallace, PhD, of Tulane University, New Orleans, and colleagues.
Data on maternal mortality (defined as “death while pregnant or within 42 days of the end of pregnancy from causes related to or aggravated by pregnancy”) were limited until the addition of pregnancy to the U.S. Standard Certificate of Death in 2003; all 50 states had adopted it by 2018, the researchers noted.
In a study published in Obstetrics & Gynecology, the researchers analyzed the first 2 years of nationally available data to identify pregnancy-associated mortality and characterize other risk factors such as age and race.
The researchers identified 4,705 female homicides in 2018 and 2019. Of these, 273 (5.8%) occurred in women who were pregnant or within a year of the end of pregnancy. Approximately half (50.2%) of the pregnant or postpartum victims were non-Hispanic Black, 30% were non-Hispanic white, 9.5% were Hispanic, and 10.3% were other races; approximately one-third (35.5%) were in the 20- to 24-year age group.
Overall, the ratio was 3.62 homicides per 100,000 live births among females who were either pregnant or within 1 year post partum, compared to 3.12 homicides per 100,000 live births in nonpregnant, nonpostpartum females aged 10-44 years (P = .05).
“Patterns were similar in further stratification by both race and age such that pregnancy was associated with more than a doubled risk of homicide among girls and women aged 10–24 in both the non-Hispanic White and non-Hispanic Black populations,” the researchers wrote.
The findings are consistent with previous studies, which “implicates health and social system failures. Although we are unable to directly evaluate the involvement of intimate partner violence (IPV) in this report, we did find that a majority of pregnancy-associated homicides occurred in the home, implicating the likelihood of involvement by persons known to the victim,” they noted. In addition, the data showed that approximately 70% of the incidents of homicide in pregnant and postpartum women involved a firearm, an increase over previous estimates.
The study findings were limited by several factors including the lack of circumstantial information and incomplete data on victim characteristics, the researchers noted. Other key limitations included the potential for false-positives and false-negatives when recording pregnancy status, which could lead to underestimates of pregnancy-associated homicides, and the lack of data on pregnancy outcomes for women who experienced live birth, abortion, or miscarriage within a year of death.
However, the results highlight the need for increased awareness and training of physicians in completing the pregnancy checkbox on death certificates, and the need for action on recommendations and interventions to prevent maternal deaths from homicide, they emphasized.
“Although encouraging, a commitment to the actual implementation of policies and investments known to be effective at protecting and the promoting the health and safety of girls and women must follow,” they concluded.
Data highlight disparities
“This study could not be done effectively prior to now, as the adoption of the pregnancy checkbox on the U.S. Standard Certificate of Death was only available in all 50 states as of 2018,” Sarah W. Prager, MD, of the University of Washington, Seattle, said in an interview.
“This study also demonstrates what was already known, which is that pregnancy is a high-risk time period for intimate partner violence, including homicide. The differences in homicide rates based on race and ethnicity also highlight the clear disparities in maternal mortality in the U.S. that are attributable to racism. There is more attention being paid to maternal mortality and the differential experience based on race, and this demonstrates that simply addressing medical management during pregnancy is not enough – we need to address root causes of racism if we truly want to reduce maternal mortality,” Dr. Prager said.
“The primary take-home message for clinicians is to ascertain safety from every patient, and to try to reduce the impacts of racism on health care for patients, especially during pregnancy,” she said.
Although more detailed records would help with elucidating causes versus associations, “more research is not the answer,” Dr. Prager stated. “The real solution here is to have better gun safety laws, and to put significant resources toward reducing the impacts of racism on health care and our society.”
The study was supported by the Eunice Kennedy Shriver National Institute of Child Health and Human Development. The researchers had no financial conflicts to disclose. Dr. Prager had no financial conflicts to disclose, but serves on the editorial advisory board of Ob.Gyn News.
The prevalence of homicide was 16% higher in pregnant women or postpartum women than nonpregnant or nonpostpartum women in the United States, according to 2018 and 2019 mortality data from the National Center for Health Statistics.
Homicide has long been identified as a leading cause of death during pregnancy, but homicide is not counted in estimates of maternal mortality, nor is it emphasized as a target for prevention and intervention, wrote Maeve Wallace, PhD, of Tulane University, New Orleans, and colleagues.
Data on maternal mortality (defined as “death while pregnant or within 42 days of the end of pregnancy from causes related to or aggravated by pregnancy”) were limited until the addition of pregnancy to the U.S. Standard Certificate of Death in 2003; all 50 states had adopted it by 2018, the researchers noted.
In a study published in Obstetrics & Gynecology, the researchers analyzed the first 2 years of nationally available data to identify pregnancy-associated mortality and characterize other risk factors such as age and race.
The researchers identified 4,705 female homicides in 2018 and 2019. Of these, 273 (5.8%) occurred in women who were pregnant or within a year of the end of pregnancy. Approximately half (50.2%) of the pregnant or postpartum victims were non-Hispanic Black, 30% were non-Hispanic white, 9.5% were Hispanic, and 10.3% were other races; approximately one-third (35.5%) were in the 20- to 24-year age group.
Overall, the ratio was 3.62 homicides per 100,000 live births among females who were either pregnant or within 1 year post partum, compared to 3.12 homicides per 100,000 live births in nonpregnant, nonpostpartum females aged 10-44 years (P = .05).
“Patterns were similar in further stratification by both race and age such that pregnancy was associated with more than a doubled risk of homicide among girls and women aged 10–24 in both the non-Hispanic White and non-Hispanic Black populations,” the researchers wrote.
The findings are consistent with previous studies, which “implicates health and social system failures. Although we are unable to directly evaluate the involvement of intimate partner violence (IPV) in this report, we did find that a majority of pregnancy-associated homicides occurred in the home, implicating the likelihood of involvement by persons known to the victim,” they noted. In addition, the data showed that approximately 70% of the incidents of homicide in pregnant and postpartum women involved a firearm, an increase over previous estimates.
The study findings were limited by several factors including the lack of circumstantial information and incomplete data on victim characteristics, the researchers noted. Other key limitations included the potential for false-positives and false-negatives when recording pregnancy status, which could lead to underestimates of pregnancy-associated homicides, and the lack of data on pregnancy outcomes for women who experienced live birth, abortion, or miscarriage within a year of death.
However, the results highlight the need for increased awareness and training of physicians in completing the pregnancy checkbox on death certificates, and the need for action on recommendations and interventions to prevent maternal deaths from homicide, they emphasized.
“Although encouraging, a commitment to the actual implementation of policies and investments known to be effective at protecting and the promoting the health and safety of girls and women must follow,” they concluded.
Data highlight disparities
“This study could not be done effectively prior to now, as the adoption of the pregnancy checkbox on the U.S. Standard Certificate of Death was only available in all 50 states as of 2018,” Sarah W. Prager, MD, of the University of Washington, Seattle, said in an interview.
“This study also demonstrates what was already known, which is that pregnancy is a high-risk time period for intimate partner violence, including homicide. The differences in homicide rates based on race and ethnicity also highlight the clear disparities in maternal mortality in the U.S. that are attributable to racism. There is more attention being paid to maternal mortality and the differential experience based on race, and this demonstrates that simply addressing medical management during pregnancy is not enough – we need to address root causes of racism if we truly want to reduce maternal mortality,” Dr. Prager said.
“The primary take-home message for clinicians is to ascertain safety from every patient, and to try to reduce the impacts of racism on health care for patients, especially during pregnancy,” she said.
Although more detailed records would help with elucidating causes versus associations, “more research is not the answer,” Dr. Prager stated. “The real solution here is to have better gun safety laws, and to put significant resources toward reducing the impacts of racism on health care and our society.”
The study was supported by the Eunice Kennedy Shriver National Institute of Child Health and Human Development. The researchers had no financial conflicts to disclose. Dr. Prager had no financial conflicts to disclose, but serves on the editorial advisory board of Ob.Gyn News.
FROM OBSTETRICS & GYNECOLOGY
Women with recurrent UTIs express fear, frustration
Fear of antibiotic overuse and frustration with physicians who prescribe them too freely are key sentiments expressed by women with recurrent urinary tract infections (rUTIs), according to findings from a study involving six focus groups.
“Here in our female pelvic medicine reconstructive urology clinic at Cedars-Sinai and at UCLA, we see many women who are referred for evaluation of rUTIs who are very frustrated with their care,” Victoria Scott, MD, Cedars-Sinai Medical Center, Los Angeles, said in an interview.
“So with these focus groups, we saw an opportunity to explore why women are so frustrated and to try and improve the care delivered,” she added.
Findings from the study were published online Sept. 1 in The Journal of Urology.
“There is a need for physicians to modify management strategies ... and to devote more research efforts to improving nonantibiotic options for the prevention and treatment of recurrent urinary tract infections, as well as management strategies that better empower patients,” the authors wrote.
Six focus groups
Four or five participants were included in each of the six focus groups – a total of 29 women. All participants reported a history of symptomatic, culture-proven UTI episodes. They had experienced two or more infections in 6 months or three or more infections within 1 year. Women were predominantly White. Most were employed part- or full-time and held a college degree.
From a qualitative analysis of all focus group transcripts, two main themes emerged:
- The negative impact of taking antibiotics for the prevention and treatment of rUTIs.
- Resentment of the medical profession for the way it managed rUTIs.
The researchers found that participants had a good understanding of the deleterious effects from inappropriate antibiotic use, largely gleaned from media sources and the Internet. “Numerous women stated that they had reached such a level of concern about antibiotics that they would resist taking them for prevention or treatment of infections,” Dr. Scott and colleagues pointed out.
These concerns centered around the risk of developing resistance to antibiotics and the ill effects that antibiotics can have on the gastrointestinal and genitourinary microbiomes. Several women reported that they had developed Clostridium difficile infections after taking antibiotics; one of the patients required hospitalization for the infection.
Women also reported concerns that they had been given an antibiotic needlessly for symptoms that might have been caused by a genitourinary condition other than a UTI. They also reported feeling resentful toward practitioners, particularly if they felt the practitioner was overprescribing antibiotics. Some had resorted to consultations with alternative practitioners, such as herbalists. “A second concern discussed by participants was the feeling of being ignored by physicians,” the authors observed.
In this regard, the women felt that their physicians underestimated the burden that rUTIs had on their lives and the detrimental effect that repeated infections had on their relationships, work, and overall quality of life. “These perceptions led to a prevalent mistrust of physicians,” the investigators wrote. This prompted many women to insist that the medical community devote more effort to the development of nonantibiotic options for the prevention and treatment of UTIs.
Improved management strategies
Asked how physicians might improve their management of rUTIs, Dr. Scott shared a number of suggestions. Cardinal rule No. 1: Have the patient undergo a urinalysis to make sure she does have a UTI. “There is a subset of patients among women with rUTIs who come in with a diagnosis of an rUTI but who really have not had documentation of more than one positive urine culture,” Dr. Scott noted. Such a history suggests that they do not have an rUTI.
It’s imperative that physicians rule out commonly misdiagnosed disorders, such as overactive bladder, as a cause of the patient’s symptoms. Symptoms of overactive bladder and rUTIs often overlap. While waiting for results from the urinalysis to confirm or rule out a UTI, young and healthy women may be prescribed a nonsteroidal anti-inflammatory drug (NSAID), such as naproxen, which can help ameliorate symptoms.
Because UTIs are frequently self-limiting, Dr. Scott and others have found that for young, otherwise healthy women, NSAIDs alone can often resolve symptoms of the UTI without use of an antibiotic. For relatively severe symptoms, a urinary analgesic, such as phenazopyridine (Pyridium), may soothe the lining of the urinary tract and relieve pain. Cystex is an over-the-counter urinary analgesic that women can procure themselves, Dr. Scott added.
If an antibiotic is indicated, those most commonly prescribed for a single episode of acute cystitis are nitrofurantoin and sulfamethoxazole plus trimethoprim (Bactrim). For recurrent UTIs, “patients are a bit more complicated,” Dr. Scott admitted. “I think the best practice is to look back at a woman’s prior urine culture and select an antibiotic that showed good sensitivity in the last positive urine test,” she said.
Prevention starts with behavioral strategies, such as voiding after sexual intercourse and wiping from front to back following urination to avoid introducing fecal bacteria into the urethra. Evidence suggests that premenopausal women who drink at least 1.5 L of water a day have significantly fewer UTI episodes, Dr. Scott noted. There is also “pretty good” evidence that cranberry supplements (not juice) can prevent rUTIs. Use of cranberry supplements is supported by the American Urological Association (conditional recommendation; evidence level of grade C).
For peri- and postmenopausal women, vaginal estrogen may be effective. It’s use for UTI prevention is well supported by the literature. Although not as well supported by evidence, some women find that a supplement such as D-mannose may prevent or treat UTIs by causing bacteria to bind to it rather than to the bladder wall. Probiotics are another possibility, she noted. Empathy can’t hurt, she added.
“A common theme among satisfied women was the sentiment that their physicians understood their problems and had a system in place to allow rapid diagnosis and treatment for UTI episodes,” the authors emphasized.
“[Such attitudes] highlight the need to investigate each patient’s experience and perceptions to allow for shared decision making regarding the management of rUTIs,” they wrote.
Further commentary
Asked to comment on the findings, editorialist Michelle Van Kuiken, MD, assistant professor of urology, University of California, San Francisco, acknowledged that there is not a lot of good evidence to support many of the strategies recommended by the American Urological Association to prevent and treat rUTIs, but she often follows these recommendations anyway. “The one statement in the guidelines that is the most supported by evidence is the use of cranberry supplements, and I do routinely recommended daily use of some form of concentrated cranberry supplements for all of my patients with rUTIs,” she said in an interview.
Dr. Van Kuiken said that vaginal estrogen is a very good option for all postmenopausal women who suffer from rUTIs and that there is growing acceptance of its use for this and other indications. There is some evidence to support D-mannose as well, although it’s not that robust, she acknowledged.
She said the evidence supporting the use of probiotics for this indication is very thin. She does not routinely recommend them for rUTIs, although they are not inherently harmful. “I think for a lot of women who have rUTIs, it can be pretty debilitating and upsetting for them – it can impact travel plans, work, and social events,” Dr. Van Kuiken said.
“Until we develop better diagnostic and therapeutic strategies, validating women’s experiences and concerns with rUTI while limiting unnecessary antibiotics remains our best option,” she wrote.
Dr. Scott and Dr. Van Kuiken have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Fear of antibiotic overuse and frustration with physicians who prescribe them too freely are key sentiments expressed by women with recurrent urinary tract infections (rUTIs), according to findings from a study involving six focus groups.
“Here in our female pelvic medicine reconstructive urology clinic at Cedars-Sinai and at UCLA, we see many women who are referred for evaluation of rUTIs who are very frustrated with their care,” Victoria Scott, MD, Cedars-Sinai Medical Center, Los Angeles, said in an interview.
“So with these focus groups, we saw an opportunity to explore why women are so frustrated and to try and improve the care delivered,” she added.
Findings from the study were published online Sept. 1 in The Journal of Urology.
“There is a need for physicians to modify management strategies ... and to devote more research efforts to improving nonantibiotic options for the prevention and treatment of recurrent urinary tract infections, as well as management strategies that better empower patients,” the authors wrote.
Six focus groups
Four or five participants were included in each of the six focus groups – a total of 29 women. All participants reported a history of symptomatic, culture-proven UTI episodes. They had experienced two or more infections in 6 months or three or more infections within 1 year. Women were predominantly White. Most were employed part- or full-time and held a college degree.
From a qualitative analysis of all focus group transcripts, two main themes emerged:
- The negative impact of taking antibiotics for the prevention and treatment of rUTIs.
- Resentment of the medical profession for the way it managed rUTIs.
The researchers found that participants had a good understanding of the deleterious effects from inappropriate antibiotic use, largely gleaned from media sources and the Internet. “Numerous women stated that they had reached such a level of concern about antibiotics that they would resist taking them for prevention or treatment of infections,” Dr. Scott and colleagues pointed out.
These concerns centered around the risk of developing resistance to antibiotics and the ill effects that antibiotics can have on the gastrointestinal and genitourinary microbiomes. Several women reported that they had developed Clostridium difficile infections after taking antibiotics; one of the patients required hospitalization for the infection.
Women also reported concerns that they had been given an antibiotic needlessly for symptoms that might have been caused by a genitourinary condition other than a UTI. They also reported feeling resentful toward practitioners, particularly if they felt the practitioner was overprescribing antibiotics. Some had resorted to consultations with alternative practitioners, such as herbalists. “A second concern discussed by participants was the feeling of being ignored by physicians,” the authors observed.
In this regard, the women felt that their physicians underestimated the burden that rUTIs had on their lives and the detrimental effect that repeated infections had on their relationships, work, and overall quality of life. “These perceptions led to a prevalent mistrust of physicians,” the investigators wrote. This prompted many women to insist that the medical community devote more effort to the development of nonantibiotic options for the prevention and treatment of UTIs.
Improved management strategies
Asked how physicians might improve their management of rUTIs, Dr. Scott shared a number of suggestions. Cardinal rule No. 1: Have the patient undergo a urinalysis to make sure she does have a UTI. “There is a subset of patients among women with rUTIs who come in with a diagnosis of an rUTI but who really have not had documentation of more than one positive urine culture,” Dr. Scott noted. Such a history suggests that they do not have an rUTI.
It’s imperative that physicians rule out commonly misdiagnosed disorders, such as overactive bladder, as a cause of the patient’s symptoms. Symptoms of overactive bladder and rUTIs often overlap. While waiting for results from the urinalysis to confirm or rule out a UTI, young and healthy women may be prescribed a nonsteroidal anti-inflammatory drug (NSAID), such as naproxen, which can help ameliorate symptoms.
Because UTIs are frequently self-limiting, Dr. Scott and others have found that for young, otherwise healthy women, NSAIDs alone can often resolve symptoms of the UTI without use of an antibiotic. For relatively severe symptoms, a urinary analgesic, such as phenazopyridine (Pyridium), may soothe the lining of the urinary tract and relieve pain. Cystex is an over-the-counter urinary analgesic that women can procure themselves, Dr. Scott added.
If an antibiotic is indicated, those most commonly prescribed for a single episode of acute cystitis are nitrofurantoin and sulfamethoxazole plus trimethoprim (Bactrim). For recurrent UTIs, “patients are a bit more complicated,” Dr. Scott admitted. “I think the best practice is to look back at a woman’s prior urine culture and select an antibiotic that showed good sensitivity in the last positive urine test,” she said.
Prevention starts with behavioral strategies, such as voiding after sexual intercourse and wiping from front to back following urination to avoid introducing fecal bacteria into the urethra. Evidence suggests that premenopausal women who drink at least 1.5 L of water a day have significantly fewer UTI episodes, Dr. Scott noted. There is also “pretty good” evidence that cranberry supplements (not juice) can prevent rUTIs. Use of cranberry supplements is supported by the American Urological Association (conditional recommendation; evidence level of grade C).
For peri- and postmenopausal women, vaginal estrogen may be effective. It’s use for UTI prevention is well supported by the literature. Although not as well supported by evidence, some women find that a supplement such as D-mannose may prevent or treat UTIs by causing bacteria to bind to it rather than to the bladder wall. Probiotics are another possibility, she noted. Empathy can’t hurt, she added.
“A common theme among satisfied women was the sentiment that their physicians understood their problems and had a system in place to allow rapid diagnosis and treatment for UTI episodes,” the authors emphasized.
“[Such attitudes] highlight the need to investigate each patient’s experience and perceptions to allow for shared decision making regarding the management of rUTIs,” they wrote.
Further commentary
Asked to comment on the findings, editorialist Michelle Van Kuiken, MD, assistant professor of urology, University of California, San Francisco, acknowledged that there is not a lot of good evidence to support many of the strategies recommended by the American Urological Association to prevent and treat rUTIs, but she often follows these recommendations anyway. “The one statement in the guidelines that is the most supported by evidence is the use of cranberry supplements, and I do routinely recommended daily use of some form of concentrated cranberry supplements for all of my patients with rUTIs,” she said in an interview.
Dr. Van Kuiken said that vaginal estrogen is a very good option for all postmenopausal women who suffer from rUTIs and that there is growing acceptance of its use for this and other indications. There is some evidence to support D-mannose as well, although it’s not that robust, she acknowledged.
She said the evidence supporting the use of probiotics for this indication is very thin. She does not routinely recommend them for rUTIs, although they are not inherently harmful. “I think for a lot of women who have rUTIs, it can be pretty debilitating and upsetting for them – it can impact travel plans, work, and social events,” Dr. Van Kuiken said.
“Until we develop better diagnostic and therapeutic strategies, validating women’s experiences and concerns with rUTI while limiting unnecessary antibiotics remains our best option,” she wrote.
Dr. Scott and Dr. Van Kuiken have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Fear of antibiotic overuse and frustration with physicians who prescribe them too freely are key sentiments expressed by women with recurrent urinary tract infections (rUTIs), according to findings from a study involving six focus groups.
“Here in our female pelvic medicine reconstructive urology clinic at Cedars-Sinai and at UCLA, we see many women who are referred for evaluation of rUTIs who are very frustrated with their care,” Victoria Scott, MD, Cedars-Sinai Medical Center, Los Angeles, said in an interview.
“So with these focus groups, we saw an opportunity to explore why women are so frustrated and to try and improve the care delivered,” she added.
Findings from the study were published online Sept. 1 in The Journal of Urology.
“There is a need for physicians to modify management strategies ... and to devote more research efforts to improving nonantibiotic options for the prevention and treatment of recurrent urinary tract infections, as well as management strategies that better empower patients,” the authors wrote.
Six focus groups
Four or five participants were included in each of the six focus groups – a total of 29 women. All participants reported a history of symptomatic, culture-proven UTI episodes. They had experienced two or more infections in 6 months or three or more infections within 1 year. Women were predominantly White. Most were employed part- or full-time and held a college degree.
From a qualitative analysis of all focus group transcripts, two main themes emerged:
- The negative impact of taking antibiotics for the prevention and treatment of rUTIs.
- Resentment of the medical profession for the way it managed rUTIs.
The researchers found that participants had a good understanding of the deleterious effects from inappropriate antibiotic use, largely gleaned from media sources and the Internet. “Numerous women stated that they had reached such a level of concern about antibiotics that they would resist taking them for prevention or treatment of infections,” Dr. Scott and colleagues pointed out.
These concerns centered around the risk of developing resistance to antibiotics and the ill effects that antibiotics can have on the gastrointestinal and genitourinary microbiomes. Several women reported that they had developed Clostridium difficile infections after taking antibiotics; one of the patients required hospitalization for the infection.
Women also reported concerns that they had been given an antibiotic needlessly for symptoms that might have been caused by a genitourinary condition other than a UTI. They also reported feeling resentful toward practitioners, particularly if they felt the practitioner was overprescribing antibiotics. Some had resorted to consultations with alternative practitioners, such as herbalists. “A second concern discussed by participants was the feeling of being ignored by physicians,” the authors observed.
In this regard, the women felt that their physicians underestimated the burden that rUTIs had on their lives and the detrimental effect that repeated infections had on their relationships, work, and overall quality of life. “These perceptions led to a prevalent mistrust of physicians,” the investigators wrote. This prompted many women to insist that the medical community devote more effort to the development of nonantibiotic options for the prevention and treatment of UTIs.
Improved management strategies
Asked how physicians might improve their management of rUTIs, Dr. Scott shared a number of suggestions. Cardinal rule No. 1: Have the patient undergo a urinalysis to make sure she does have a UTI. “There is a subset of patients among women with rUTIs who come in with a diagnosis of an rUTI but who really have not had documentation of more than one positive urine culture,” Dr. Scott noted. Such a history suggests that they do not have an rUTI.
It’s imperative that physicians rule out commonly misdiagnosed disorders, such as overactive bladder, as a cause of the patient’s symptoms. Symptoms of overactive bladder and rUTIs often overlap. While waiting for results from the urinalysis to confirm or rule out a UTI, young and healthy women may be prescribed a nonsteroidal anti-inflammatory drug (NSAID), such as naproxen, which can help ameliorate symptoms.
Because UTIs are frequently self-limiting, Dr. Scott and others have found that for young, otherwise healthy women, NSAIDs alone can often resolve symptoms of the UTI without use of an antibiotic. For relatively severe symptoms, a urinary analgesic, such as phenazopyridine (Pyridium), may soothe the lining of the urinary tract and relieve pain. Cystex is an over-the-counter urinary analgesic that women can procure themselves, Dr. Scott added.
If an antibiotic is indicated, those most commonly prescribed for a single episode of acute cystitis are nitrofurantoin and sulfamethoxazole plus trimethoprim (Bactrim). For recurrent UTIs, “patients are a bit more complicated,” Dr. Scott admitted. “I think the best practice is to look back at a woman’s prior urine culture and select an antibiotic that showed good sensitivity in the last positive urine test,” she said.
Prevention starts with behavioral strategies, such as voiding after sexual intercourse and wiping from front to back following urination to avoid introducing fecal bacteria into the urethra. Evidence suggests that premenopausal women who drink at least 1.5 L of water a day have significantly fewer UTI episodes, Dr. Scott noted. There is also “pretty good” evidence that cranberry supplements (not juice) can prevent rUTIs. Use of cranberry supplements is supported by the American Urological Association (conditional recommendation; evidence level of grade C).
For peri- and postmenopausal women, vaginal estrogen may be effective. It’s use for UTI prevention is well supported by the literature. Although not as well supported by evidence, some women find that a supplement such as D-mannose may prevent or treat UTIs by causing bacteria to bind to it rather than to the bladder wall. Probiotics are another possibility, she noted. Empathy can’t hurt, she added.
“A common theme among satisfied women was the sentiment that their physicians understood their problems and had a system in place to allow rapid diagnosis and treatment for UTI episodes,” the authors emphasized.
“[Such attitudes] highlight the need to investigate each patient’s experience and perceptions to allow for shared decision making regarding the management of rUTIs,” they wrote.
Further commentary
Asked to comment on the findings, editorialist Michelle Van Kuiken, MD, assistant professor of urology, University of California, San Francisco, acknowledged that there is not a lot of good evidence to support many of the strategies recommended by the American Urological Association to prevent and treat rUTIs, but she often follows these recommendations anyway. “The one statement in the guidelines that is the most supported by evidence is the use of cranberry supplements, and I do routinely recommended daily use of some form of concentrated cranberry supplements for all of my patients with rUTIs,” she said in an interview.
Dr. Van Kuiken said that vaginal estrogen is a very good option for all postmenopausal women who suffer from rUTIs and that there is growing acceptance of its use for this and other indications. There is some evidence to support D-mannose as well, although it’s not that robust, she acknowledged.
She said the evidence supporting the use of probiotics for this indication is very thin. She does not routinely recommend them for rUTIs, although they are not inherently harmful. “I think for a lot of women who have rUTIs, it can be pretty debilitating and upsetting for them – it can impact travel plans, work, and social events,” Dr. Van Kuiken said.
“Until we develop better diagnostic and therapeutic strategies, validating women’s experiences and concerns with rUTI while limiting unnecessary antibiotics remains our best option,” she wrote.
Dr. Scott and Dr. Van Kuiken have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Low preconception complement levels linked to adverse pregnancy outcomes in antiphospholipid syndrome
Low serum levels of two complement proteins are linked to worse pregnancy outcomes in women with antiphospholipid syndrome (APS), the results of a multicenter study appear to confirm.
The study evaluated preconception complement levels in 260 pregnancies in 197 women who had APS or carried antiphospholipid antibodies (aPL), and found that low levels of C3 and C4 in the 6 months prior to pregnancy were associated with several gestational complications and resulted in pregnancy losses.
“This study has validated, on large scale, the possible utility of preconception measurement of C3 and C4 levels to predict pregnancy loss in patients with aPL, even at a high-risk profile,” said study investigator Daniele Lini, MD, of ASST Spedali Civili and the University of Brescia (Italy).
“The tests are easy and cheap to be routinely performed, and they could therefore represent a valid aid to identify women that need particular monitoring and management,” he said at the 14th International Congress on Systemic Lupus Erythematosus held together with the 6th International Congress on Controversies in Rheumatology and Autoimmunity.
aPL and adverse obstetric outcomes
aPL, which include lupus anticoagulant, anti–beta2-glycoprotein 1, and anticardiolipin antibodies, have been shown to induce fetal loss in animal models. Their influence on the outcome of human pregnancies, however, has been less clear, with several studies failing to prove a link between their presence and obstetric complications.
Dr. Lini and coinvestigators conducted a multicenter study involving 11 Italian centers and one Russian center, retrospectively looking for women with primary APS or women who had persistently high levels of aPL but no symptoms who had become pregnant. Of 503 pregnancies, information on complement levels before conception was available for 260, of which 184 had occurred in women with APS and 76 in women with persistently high aPL.
The pregnancies were grouped according to whether there were low (n = 93) or normal (n = 167) levels of C3 and C4 in the last 6 months.
“Women with adverse pregnancy outcomes showed significantly lower preconception complement levels than those with successful pregnancies, without any difference between APS and aPL carriers,” Dr. Lini reported.
Comparing those with low to those with high complement levels, the preterm live birth rate (before 37 weeks’ gestation) was 37% versus 18% (P < .0001).
The full-term live birth rates were a respective 42% and 72% (P < .0001).
The rate of pregnancy loss, which included both abortion and miscarriage, was a respective 21% and 10% (P = .008).
A subgroup analysis focusing on where there was triple aPL positivity found that preconception low C3 and/or C4 levels was associated with an increased rate of pregnancy loss (P = .05). This association disappeared if there was just one or two aPL present.
The researchers found no correlation between complement levels and rates of venous thromboembolism or thrombocytopenia.
Study highlights ‘impact and importance’ of complement in APS
The study indicates “the impact and the importance of complement” in APS, said Yehuda Shoenfeld, MD, the founder and head of the Zabludowicz Center for Autoimmune Diseases at the Sheba Medical Center in Tel Hashomer, Israel.
In the early days of understanding APS, said Dr. Shoenfeld, it was thought that complement was not as important as it was in systemic lupus erythematosus (SLE). The importance of raised complement seen in studies of APS would often be discounted or neglected in comparison to SLE.
However, “slowly, slowly” it has been found that “complement [in APS] is activated very similarly to SLE,” Dr. Shoenfeld noted.
“I think that it’s important to assess the component levels,” Dr. Lini said in discussion. “This is needed to be done in the preconception counseling for APS and aPL carrier patients.”
Determining whether there is single, double, or even triple aPL positivity could be useful in guiding clinical decisions.
“If we have triple positivity, that could mean that there may be a more immunologic activation of the system and that it could be useful to administrate hydroxychloroquine [to] those patients who would like to have a pregnancy,” Dr. Lini suggested.
Plus, in those with decreased complement levels, “this could be a very useful tool” to identify where something could go wrong during their pregnancy.
The study had no outside funding. Dr. Lini and Dr. Shoenfeld disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Low serum levels of two complement proteins are linked to worse pregnancy outcomes in women with antiphospholipid syndrome (APS), the results of a multicenter study appear to confirm.
The study evaluated preconception complement levels in 260 pregnancies in 197 women who had APS or carried antiphospholipid antibodies (aPL), and found that low levels of C3 and C4 in the 6 months prior to pregnancy were associated with several gestational complications and resulted in pregnancy losses.
“This study has validated, on large scale, the possible utility of preconception measurement of C3 and C4 levels to predict pregnancy loss in patients with aPL, even at a high-risk profile,” said study investigator Daniele Lini, MD, of ASST Spedali Civili and the University of Brescia (Italy).
“The tests are easy and cheap to be routinely performed, and they could therefore represent a valid aid to identify women that need particular monitoring and management,” he said at the 14th International Congress on Systemic Lupus Erythematosus held together with the 6th International Congress on Controversies in Rheumatology and Autoimmunity.
aPL and adverse obstetric outcomes
aPL, which include lupus anticoagulant, anti–beta2-glycoprotein 1, and anticardiolipin antibodies, have been shown to induce fetal loss in animal models. Their influence on the outcome of human pregnancies, however, has been less clear, with several studies failing to prove a link between their presence and obstetric complications.
Dr. Lini and coinvestigators conducted a multicenter study involving 11 Italian centers and one Russian center, retrospectively looking for women with primary APS or women who had persistently high levels of aPL but no symptoms who had become pregnant. Of 503 pregnancies, information on complement levels before conception was available for 260, of which 184 had occurred in women with APS and 76 in women with persistently high aPL.
The pregnancies were grouped according to whether there were low (n = 93) or normal (n = 167) levels of C3 and C4 in the last 6 months.
“Women with adverse pregnancy outcomes showed significantly lower preconception complement levels than those with successful pregnancies, without any difference between APS and aPL carriers,” Dr. Lini reported.
Comparing those with low to those with high complement levels, the preterm live birth rate (before 37 weeks’ gestation) was 37% versus 18% (P < .0001).
The full-term live birth rates were a respective 42% and 72% (P < .0001).
The rate of pregnancy loss, which included both abortion and miscarriage, was a respective 21% and 10% (P = .008).
A subgroup analysis focusing on where there was triple aPL positivity found that preconception low C3 and/or C4 levels was associated with an increased rate of pregnancy loss (P = .05). This association disappeared if there was just one or two aPL present.
The researchers found no correlation between complement levels and rates of venous thromboembolism or thrombocytopenia.
Study highlights ‘impact and importance’ of complement in APS
The study indicates “the impact and the importance of complement” in APS, said Yehuda Shoenfeld, MD, the founder and head of the Zabludowicz Center for Autoimmune Diseases at the Sheba Medical Center in Tel Hashomer, Israel.
In the early days of understanding APS, said Dr. Shoenfeld, it was thought that complement was not as important as it was in systemic lupus erythematosus (SLE). The importance of raised complement seen in studies of APS would often be discounted or neglected in comparison to SLE.
However, “slowly, slowly” it has been found that “complement [in APS] is activated very similarly to SLE,” Dr. Shoenfeld noted.
“I think that it’s important to assess the component levels,” Dr. Lini said in discussion. “This is needed to be done in the preconception counseling for APS and aPL carrier patients.”
Determining whether there is single, double, or even triple aPL positivity could be useful in guiding clinical decisions.
“If we have triple positivity, that could mean that there may be a more immunologic activation of the system and that it could be useful to administrate hydroxychloroquine [to] those patients who would like to have a pregnancy,” Dr. Lini suggested.
Plus, in those with decreased complement levels, “this could be a very useful tool” to identify where something could go wrong during their pregnancy.
The study had no outside funding. Dr. Lini and Dr. Shoenfeld disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Low serum levels of two complement proteins are linked to worse pregnancy outcomes in women with antiphospholipid syndrome (APS), the results of a multicenter study appear to confirm.
The study evaluated preconception complement levels in 260 pregnancies in 197 women who had APS or carried antiphospholipid antibodies (aPL), and found that low levels of C3 and C4 in the 6 months prior to pregnancy were associated with several gestational complications and resulted in pregnancy losses.
“This study has validated, on large scale, the possible utility of preconception measurement of C3 and C4 levels to predict pregnancy loss in patients with aPL, even at a high-risk profile,” said study investigator Daniele Lini, MD, of ASST Spedali Civili and the University of Brescia (Italy).
“The tests are easy and cheap to be routinely performed, and they could therefore represent a valid aid to identify women that need particular monitoring and management,” he said at the 14th International Congress on Systemic Lupus Erythematosus held together with the 6th International Congress on Controversies in Rheumatology and Autoimmunity.
aPL and adverse obstetric outcomes
aPL, which include lupus anticoagulant, anti–beta2-glycoprotein 1, and anticardiolipin antibodies, have been shown to induce fetal loss in animal models. Their influence on the outcome of human pregnancies, however, has been less clear, with several studies failing to prove a link between their presence and obstetric complications.
Dr. Lini and coinvestigators conducted a multicenter study involving 11 Italian centers and one Russian center, retrospectively looking for women with primary APS or women who had persistently high levels of aPL but no symptoms who had become pregnant. Of 503 pregnancies, information on complement levels before conception was available for 260, of which 184 had occurred in women with APS and 76 in women with persistently high aPL.
The pregnancies were grouped according to whether there were low (n = 93) or normal (n = 167) levels of C3 and C4 in the last 6 months.
“Women with adverse pregnancy outcomes showed significantly lower preconception complement levels than those with successful pregnancies, without any difference between APS and aPL carriers,” Dr. Lini reported.
Comparing those with low to those with high complement levels, the preterm live birth rate (before 37 weeks’ gestation) was 37% versus 18% (P < .0001).
The full-term live birth rates were a respective 42% and 72% (P < .0001).
The rate of pregnancy loss, which included both abortion and miscarriage, was a respective 21% and 10% (P = .008).
A subgroup analysis focusing on where there was triple aPL positivity found that preconception low C3 and/or C4 levels was associated with an increased rate of pregnancy loss (P = .05). This association disappeared if there was just one or two aPL present.
The researchers found no correlation between complement levels and rates of venous thromboembolism or thrombocytopenia.
Study highlights ‘impact and importance’ of complement in APS
The study indicates “the impact and the importance of complement” in APS, said Yehuda Shoenfeld, MD, the founder and head of the Zabludowicz Center for Autoimmune Diseases at the Sheba Medical Center in Tel Hashomer, Israel.
In the early days of understanding APS, said Dr. Shoenfeld, it was thought that complement was not as important as it was in systemic lupus erythematosus (SLE). The importance of raised complement seen in studies of APS would often be discounted or neglected in comparison to SLE.
However, “slowly, slowly” it has been found that “complement [in APS] is activated very similarly to SLE,” Dr. Shoenfeld noted.
“I think that it’s important to assess the component levels,” Dr. Lini said in discussion. “This is needed to be done in the preconception counseling for APS and aPL carrier patients.”
Determining whether there is single, double, or even triple aPL positivity could be useful in guiding clinical decisions.
“If we have triple positivity, that could mean that there may be a more immunologic activation of the system and that it could be useful to administrate hydroxychloroquine [to] those patients who would like to have a pregnancy,” Dr. Lini suggested.
Plus, in those with decreased complement levels, “this could be a very useful tool” to identify where something could go wrong during their pregnancy.
The study had no outside funding. Dr. Lini and Dr. Shoenfeld disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Abaloparatide significantly reduced fractures, increased BMD in women at high fracture risk
Postmenopausal women at high or very high risk of fracture gained significantly more bone mineral density and were significantly less likely to experience a fracture when taking abaloparatide for 18 months, according to new research presented at the hybrid annual meeting of the North American Menopause Society.
“The findings showed that abaloparatide was better than teriparatide in a number of parameters important in osteoporosis treatment, and similar in others, in high-risk and very-high-risk postmenopausal women with osteoporosis,” Bart Clarke, MD, a professor of medicine at Mayo Clinic in Rochester, Minn., said in an interview. “Abaloparatide is safe and effective for use in high-risk or very-high-risk postmenopausal women,” as defined by the new American Association of Clinical Endocrinology/American College of Endocrinology osteoporosis guidelines.
Ricardo R. Correa, MD, of the department of endocrinology and director of diversity for graduate medical education at the University of Arizona, Phoenix, said that the study demonstrates that abaloparatide and teriparatide have a very similar effect with abaloparatide providing a slightly better absolute risk reduction in fracture. Dr. Correa was not involved in the research.
“What will drive my decision in what to prescribe will be the cost and insurance coverage,” Dr. Correa said. “At the Veterans Administration hospital, the option that we have is abaloparatide, so this is the option that we use.”
Among women at least 65 years old who have already had one fracture, 1 in 10 will experience another fracture within the next year, and 30% will have another fracture within the next 5 years, the authors noted in their background material. Since phase 3 ACTIVE study data in 2016 showed that abaloparatide reduces fracture risk while increasing bone mineral density, compared with placebo, the researchers reanalyzed that data to assess the drug’s efficacy in patients at high or very high risk for fracture.
The study involved 2,463 postmenopausal women with osteoporosis who received one of three interventions: 80 mcg abaloparatide daily, placebo, or 20 mcg subcutaneous teriparatide daily. Only the abaloparatide and placebo groups were double blinded.
“Teriparatide was used as the comparator drug because teriparatide was previously approved as the first anabolic drug for osteoporosis,” Dr. Clarke said in an interview. “The hope was to show that abaloparatide was a better anabolic drug.”
Women were considered at high or very high risk of fracture if they met at least one of the following four criteria from the 2020 American Association of Clinical Endocrinology guidelines:
- Fracture within the past 12 months or prevalent vertebral fracture.
- Very low T-score (less than –3.0) at baseline at any site.
- Multiple fractures at baseline since age 45.
- Very high fracture risk based on the Fracture Risk Assessment Tool (FRAX) (at least 30% for major osteoporotic fracture or at least 4.5% for hip fracture).
Among the 2,026 patients who met at least one of these criteria, 664 received abaloparatide, 685 received teriparatide, and 677 received placebo. Both the abaloparatide and teriparatide significantly reduced new vertebral fracture risk, compared with placebo. In the abaloparatide group, 0.72% of women had a new vertebral fracture, compared with 0.99% in the teriparatide group and 4.77% in the placebo group (P < .0001).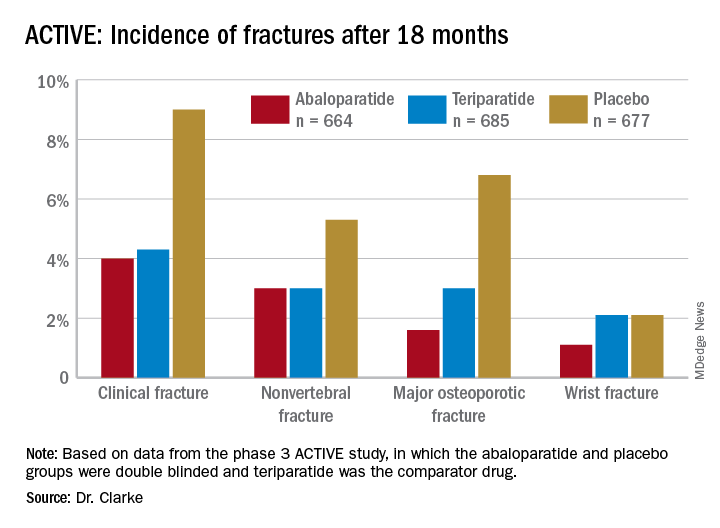
Abaloparatide and teriparatide also led to significant increases in lumbar spine, total hip, and femoral neck bone mineral density, compared with placebo (P < .0001).
The study was limited by its duration of 18 months and the Food and Drug Administration’s restriction on using abaloparatide for more than 2 years because of the theoretical risk of increasing osteosarcoma, although that risk has never been demonstrated in humans, Dr. Correa said. ”We need more data with abaloparitide in more than 2 years,” he added.
In determining which medication clinicians should first prescribe to manage osteoporosis, Dr. Correa said practitioners should consider the type of osteoporosis women have, their preferences, and their labs on kidney function.
With mild to moderate osteoporosis, bisphosphonates will be the first option while denosumab will be preferred for moderate to severe osteoporosis. Teriparatide and abaloparitide are the first-line options for severe osteoporosis, he said.
“If the glomerular filtration rate is low, we cannot use bisphosphonate and we will have to limit our use to denosumab,” he said. Route and frequency of delivery plays a role in patient preferences.
“If the patient prefers an infusion once a year or a pill, then bisphosphonate,” he said, but “if the patient is fine with an injection every 6 months, then denosumab.” Patients who need and can do an injection every day can take abaloparitide or teriparatide.
Failure of previous treatments also guide clinical decisions, he added. ”If the patient has been on one medication and has a fracture or the bone mineral density decreases, then we need to switch to another medication, usually teriparatide or abaloparitide, to build new bone.”
Contraindications for abaloparatide include a high serum calcium before therapy or prior allergic reactions to components in abaloparatide, Dr. Clarke said. No new safety signals showed up in the data analysis.
The research was funded by Radius Health. Dr. Clarke is an advisory board member of Amgen, and another author consults and speaks for Amgen and is a Radius Health Advisory Board member. Two other authors are Radius Health employees who own stock in the company. Dr Correa has no disclosures.
Postmenopausal women at high or very high risk of fracture gained significantly more bone mineral density and were significantly less likely to experience a fracture when taking abaloparatide for 18 months, according to new research presented at the hybrid annual meeting of the North American Menopause Society.
“The findings showed that abaloparatide was better than teriparatide in a number of parameters important in osteoporosis treatment, and similar in others, in high-risk and very-high-risk postmenopausal women with osteoporosis,” Bart Clarke, MD, a professor of medicine at Mayo Clinic in Rochester, Minn., said in an interview. “Abaloparatide is safe and effective for use in high-risk or very-high-risk postmenopausal women,” as defined by the new American Association of Clinical Endocrinology/American College of Endocrinology osteoporosis guidelines.
Ricardo R. Correa, MD, of the department of endocrinology and director of diversity for graduate medical education at the University of Arizona, Phoenix, said that the study demonstrates that abaloparatide and teriparatide have a very similar effect with abaloparatide providing a slightly better absolute risk reduction in fracture. Dr. Correa was not involved in the research.
“What will drive my decision in what to prescribe will be the cost and insurance coverage,” Dr. Correa said. “At the Veterans Administration hospital, the option that we have is abaloparatide, so this is the option that we use.”
Among women at least 65 years old who have already had one fracture, 1 in 10 will experience another fracture within the next year, and 30% will have another fracture within the next 5 years, the authors noted in their background material. Since phase 3 ACTIVE study data in 2016 showed that abaloparatide reduces fracture risk while increasing bone mineral density, compared with placebo, the researchers reanalyzed that data to assess the drug’s efficacy in patients at high or very high risk for fracture.
The study involved 2,463 postmenopausal women with osteoporosis who received one of three interventions: 80 mcg abaloparatide daily, placebo, or 20 mcg subcutaneous teriparatide daily. Only the abaloparatide and placebo groups were double blinded.
“Teriparatide was used as the comparator drug because teriparatide was previously approved as the first anabolic drug for osteoporosis,” Dr. Clarke said in an interview. “The hope was to show that abaloparatide was a better anabolic drug.”
Women were considered at high or very high risk of fracture if they met at least one of the following four criteria from the 2020 American Association of Clinical Endocrinology guidelines:
- Fracture within the past 12 months or prevalent vertebral fracture.
- Very low T-score (less than –3.0) at baseline at any site.
- Multiple fractures at baseline since age 45.
- Very high fracture risk based on the Fracture Risk Assessment Tool (FRAX) (at least 30% for major osteoporotic fracture or at least 4.5% for hip fracture).
Among the 2,026 patients who met at least one of these criteria, 664 received abaloparatide, 685 received teriparatide, and 677 received placebo. Both the abaloparatide and teriparatide significantly reduced new vertebral fracture risk, compared with placebo. In the abaloparatide group, 0.72% of women had a new vertebral fracture, compared with 0.99% in the teriparatide group and 4.77% in the placebo group (P < .0001).
Abaloparatide and teriparatide also led to significant increases in lumbar spine, total hip, and femoral neck bone mineral density, compared with placebo (P < .0001).
The study was limited by its duration of 18 months and the Food and Drug Administration’s restriction on using abaloparatide for more than 2 years because of the theoretical risk of increasing osteosarcoma, although that risk has never been demonstrated in humans, Dr. Correa said. ”We need more data with abaloparitide in more than 2 years,” he added.
In determining which medication clinicians should first prescribe to manage osteoporosis, Dr. Correa said practitioners should consider the type of osteoporosis women have, their preferences, and their labs on kidney function.
With mild to moderate osteoporosis, bisphosphonates will be the first option while denosumab will be preferred for moderate to severe osteoporosis. Teriparatide and abaloparitide are the first-line options for severe osteoporosis, he said.
“If the glomerular filtration rate is low, we cannot use bisphosphonate and we will have to limit our use to denosumab,” he said. Route and frequency of delivery plays a role in patient preferences.
“If the patient prefers an infusion once a year or a pill, then bisphosphonate,” he said, but “if the patient is fine with an injection every 6 months, then denosumab.” Patients who need and can do an injection every day can take abaloparitide or teriparatide.
Failure of previous treatments also guide clinical decisions, he added. ”If the patient has been on one medication and has a fracture or the bone mineral density decreases, then we need to switch to another medication, usually teriparatide or abaloparitide, to build new bone.”
Contraindications for abaloparatide include a high serum calcium before therapy or prior allergic reactions to components in abaloparatide, Dr. Clarke said. No new safety signals showed up in the data analysis.
The research was funded by Radius Health. Dr. Clarke is an advisory board member of Amgen, and another author consults and speaks for Amgen and is a Radius Health Advisory Board member. Two other authors are Radius Health employees who own stock in the company. Dr Correa has no disclosures.
Postmenopausal women at high or very high risk of fracture gained significantly more bone mineral density and were significantly less likely to experience a fracture when taking abaloparatide for 18 months, according to new research presented at the hybrid annual meeting of the North American Menopause Society.
“The findings showed that abaloparatide was better than teriparatide in a number of parameters important in osteoporosis treatment, and similar in others, in high-risk and very-high-risk postmenopausal women with osteoporosis,” Bart Clarke, MD, a professor of medicine at Mayo Clinic in Rochester, Minn., said in an interview. “Abaloparatide is safe and effective for use in high-risk or very-high-risk postmenopausal women,” as defined by the new American Association of Clinical Endocrinology/American College of Endocrinology osteoporosis guidelines.
Ricardo R. Correa, MD, of the department of endocrinology and director of diversity for graduate medical education at the University of Arizona, Phoenix, said that the study demonstrates that abaloparatide and teriparatide have a very similar effect with abaloparatide providing a slightly better absolute risk reduction in fracture. Dr. Correa was not involved in the research.
“What will drive my decision in what to prescribe will be the cost and insurance coverage,” Dr. Correa said. “At the Veterans Administration hospital, the option that we have is abaloparatide, so this is the option that we use.”
Among women at least 65 years old who have already had one fracture, 1 in 10 will experience another fracture within the next year, and 30% will have another fracture within the next 5 years, the authors noted in their background material. Since phase 3 ACTIVE study data in 2016 showed that abaloparatide reduces fracture risk while increasing bone mineral density, compared with placebo, the researchers reanalyzed that data to assess the drug’s efficacy in patients at high or very high risk for fracture.
The study involved 2,463 postmenopausal women with osteoporosis who received one of three interventions: 80 mcg abaloparatide daily, placebo, or 20 mcg subcutaneous teriparatide daily. Only the abaloparatide and placebo groups were double blinded.
“Teriparatide was used as the comparator drug because teriparatide was previously approved as the first anabolic drug for osteoporosis,” Dr. Clarke said in an interview. “The hope was to show that abaloparatide was a better anabolic drug.”
Women were considered at high or very high risk of fracture if they met at least one of the following four criteria from the 2020 American Association of Clinical Endocrinology guidelines:
- Fracture within the past 12 months or prevalent vertebral fracture.
- Very low T-score (less than –3.0) at baseline at any site.
- Multiple fractures at baseline since age 45.
- Very high fracture risk based on the Fracture Risk Assessment Tool (FRAX) (at least 30% for major osteoporotic fracture or at least 4.5% for hip fracture).
Among the 2,026 patients who met at least one of these criteria, 664 received abaloparatide, 685 received teriparatide, and 677 received placebo. Both the abaloparatide and teriparatide significantly reduced new vertebral fracture risk, compared with placebo. In the abaloparatide group, 0.72% of women had a new vertebral fracture, compared with 0.99% in the teriparatide group and 4.77% in the placebo group (P < .0001).
Abaloparatide and teriparatide also led to significant increases in lumbar spine, total hip, and femoral neck bone mineral density, compared with placebo (P < .0001).
The study was limited by its duration of 18 months and the Food and Drug Administration’s restriction on using abaloparatide for more than 2 years because of the theoretical risk of increasing osteosarcoma, although that risk has never been demonstrated in humans, Dr. Correa said. ”We need more data with abaloparitide in more than 2 years,” he added.
In determining which medication clinicians should first prescribe to manage osteoporosis, Dr. Correa said practitioners should consider the type of osteoporosis women have, their preferences, and their labs on kidney function.
With mild to moderate osteoporosis, bisphosphonates will be the first option while denosumab will be preferred for moderate to severe osteoporosis. Teriparatide and abaloparitide are the first-line options for severe osteoporosis, he said.
“If the glomerular filtration rate is low, we cannot use bisphosphonate and we will have to limit our use to denosumab,” he said. Route and frequency of delivery plays a role in patient preferences.
“If the patient prefers an infusion once a year or a pill, then bisphosphonate,” he said, but “if the patient is fine with an injection every 6 months, then denosumab.” Patients who need and can do an injection every day can take abaloparitide or teriparatide.
Failure of previous treatments also guide clinical decisions, he added. ”If the patient has been on one medication and has a fracture or the bone mineral density decreases, then we need to switch to another medication, usually teriparatide or abaloparitide, to build new bone.”
Contraindications for abaloparatide include a high serum calcium before therapy or prior allergic reactions to components in abaloparatide, Dr. Clarke said. No new safety signals showed up in the data analysis.
The research was funded by Radius Health. Dr. Clarke is an advisory board member of Amgen, and another author consults and speaks for Amgen and is a Radius Health Advisory Board member. Two other authors are Radius Health employees who own stock in the company. Dr Correa has no disclosures.
FROM NAMS 2021
Steroid a promising short-term treatment option for major depression?
Study results of an experimental agent that improves symptoms of major depression and boosts quality of life in as little as 3 days suggest it may be an effective short-term treatment option.
Phase 3 results of a randomized, placebo-controlled trial compared zuranolone, an neuroactive steroid that binds to both synaptic and extra-synaptic GABA-A receptors, to placebo in patients with major depressive disorder (MDD). Overall, 30% of participants were already taking antidepressants.
Investigators found the drug was associated with a significant improvement in depression scores versus placebo, with benefit observed as early as day 3. This was accompanied by improved function and well-being.
, said study presenter Colville Brown, MD, Sage Therapeutics, Cambridge, Mass.
“These data continue to support the development of zuranolone as a potential 14-day short course treatment for major depressive disorder episodes.”
The findings were presented at the virtual congress of the European College of Neuropsychopharmacology.
High placebo response
However, despite being significant, the drug’s benefit was only slightly higher than that of placebo, raising questions about the study design and the true performance of the drug.
Dr. Brown explained that patients with MDD were randomized to oral zuranolone 50 mg or placebo once daily for 14 days, with dose reductions to 40 mg or matching placebo permitted in case of perceived intolerance.
Patients were assessed at baseline and day 15 via the 17-item Hamilton Rating Scale for Depression (HAMD-17) before entering a 28-day follow-up period off the study drug.
Among the 268 participants who received zuranolone, 90.3% completed the study, compared with 87.4% of 269 patients in the placebo group.
The mean age of participants was 40 years. Women made up 69.4% of those who received zuranolone and 61.7% assigned to placebo.
The mean HAMD-17 score at baseline was 26.8 and 26.9 in the zuranolone and placebo groups, respectively. Dr. Brown noted that 29.5% of patients in the zuranolone group and 30.1% of those assigned to placebo were taking antidepressants at baseline.
The study’s primary endpoint was met, with patients taking the study drug experiencing a significantly greater reduction in HAMD-17 scores from baseline to day 15 versus those given placebo, at 14.1 versus 12.3 points (P = .0141).
Dr. Brown highlighted that the difference in reduction in HAMD-17 scores between the zuranolone and placebo groups was already significant at day 3 (P < .0001), and again at day 8 (P < .0001) and day 12 (P < .001).
At day 3, response rates on the HAMD-17 were significantly higher among zuranolone-treated patients than among those given placebo, at 29.3% versus 16.3% (P < .001). However, the differences on day 15 and on day 42 were no longer significant.
A similar effect was seen for HAMD-17 remissions, which were seen in 7.6% of zuranolone-treated patients and 2.3% of those given placebo at day 3 (P < .01), rising to 29.8% versus 27.1% at day 15, and 30.8% versus 29.6% at day 42, and neither difference was significant.
Dr. Brown also showed that, at all time points during the treatment and follow-up periods, improvements in response rates in Global Improvement on the Clinical Global Impression scale favored zuranolone.
On the SF-36v2 quality of life questionnaire, improvements again favored zuranolone on all domains, although the difference between active treatment and placebo was significant only for vitality on day 15, at 12.8 versus 9.7 points (P < .05).
Treatment-emergent adverse events were more common with zuranolone, with 60.1% of patients experiencing at least one event of any grade versus 44.6% with placebo. However, severe events were seen in only 3.0% versus 1.1% of patients, and serious adverse events were recorded in only two patients (0.7%) in both groups.
The most common adverse events were somnolence, dizziness, headache, sedation, and diarrhea, with no increase in suicidal ideation or withdrawal. Dr. Brown noted that there was “no change in the safety signal” between patients with or without prior antidepressant therapy.
From the audience, Marie-Josée Filteau, MD, department of psychiatry, Laval University, Quebec, drew attention to the similarity in the improvement in HAMD-17 scores between the zuranolone and placebo groups, asking: “How is that compelling?”
Dr. Brown replied that “what they are excited about is that change from baseline with zuranolone,” adding: “You do see it in the placebo group as well, and ... this isn’t new to psychiatry.
“This is a heterogeneous disease, and remember this [study] was conducted during COVID, so patients were being seen with clinic visits during COVID.
“What impact did that have? The placebo is not really placebo” in this case.
More effective than results suggest?
Approached for comment by this news organization, Maurizio Fava, MD, executive vice chair, department of psychiatry, and executive director, Clinical Trials Network and Institute, Massachusetts General Hospital, Boston, noted there are several issues with the trial.
Because of those, the drug “is likely to be much more efficacious than it looks because it achieved statistical significance despite an extremely high placebo response,” he said
“Whenever your change on placebo is greater than 10 points on the HAMD, you have an excessive response ... and a very, very low chance of detecting a signal,” he said.
Dr. Fava said that another issue was including patients who were either on or off antidepressants, which meant the population was not sufficiently homogenous.
Another “flaw” was to assume that the placebo effect would be “transient” and deteriorate over time, whereas the results showed the opposite.
Nevertheless, “it’s a positive study because of the sample size ... that provides further evidence for the antidepressant activity of zuranolone” and the drug was “well tolerated.”
Dr. Fava expects zuranolone “will make it to the market,” as an indication from the Food and Drug Administration is likely, “but if you’re asking me: Is the drug as effective as shown in their studies? It’s probably much more effective.”
The study was funded by Sage Therapeutics and Biogen. Dr. Brown is an employee of Sage Therapeutics. Lead investigator Anita Clayton, MD, University of Virginia, Charlottesville, has reported relationships with Dario Bioscience, Janssen, Praxis Precision Medicines, Relmada Therapeutics, Sage Therapeutics, Fabre-Kramer, MindCure, Ovoca Bio, PureTech Health, S1 Biopharma, Vella Bioscience, WCG MedAvante-ProPhase, Ballantine Books/Random House, Guilford Publications, Euthymics, and Mediflix.
A version of this article first appeared on Medscape.com.
Study results of an experimental agent that improves symptoms of major depression and boosts quality of life in as little as 3 days suggest it may be an effective short-term treatment option.
Phase 3 results of a randomized, placebo-controlled trial compared zuranolone, an neuroactive steroid that binds to both synaptic and extra-synaptic GABA-A receptors, to placebo in patients with major depressive disorder (MDD). Overall, 30% of participants were already taking antidepressants.
Investigators found the drug was associated with a significant improvement in depression scores versus placebo, with benefit observed as early as day 3. This was accompanied by improved function and well-being.
, said study presenter Colville Brown, MD, Sage Therapeutics, Cambridge, Mass.
“These data continue to support the development of zuranolone as a potential 14-day short course treatment for major depressive disorder episodes.”
The findings were presented at the virtual congress of the European College of Neuropsychopharmacology.
High placebo response
However, despite being significant, the drug’s benefit was only slightly higher than that of placebo, raising questions about the study design and the true performance of the drug.
Dr. Brown explained that patients with MDD were randomized to oral zuranolone 50 mg or placebo once daily for 14 days, with dose reductions to 40 mg or matching placebo permitted in case of perceived intolerance.
Patients were assessed at baseline and day 15 via the 17-item Hamilton Rating Scale for Depression (HAMD-17) before entering a 28-day follow-up period off the study drug.
Among the 268 participants who received zuranolone, 90.3% completed the study, compared with 87.4% of 269 patients in the placebo group.
The mean age of participants was 40 years. Women made up 69.4% of those who received zuranolone and 61.7% assigned to placebo.
The mean HAMD-17 score at baseline was 26.8 and 26.9 in the zuranolone and placebo groups, respectively. Dr. Brown noted that 29.5% of patients in the zuranolone group and 30.1% of those assigned to placebo were taking antidepressants at baseline.
The study’s primary endpoint was met, with patients taking the study drug experiencing a significantly greater reduction in HAMD-17 scores from baseline to day 15 versus those given placebo, at 14.1 versus 12.3 points (P = .0141).
Dr. Brown highlighted that the difference in reduction in HAMD-17 scores between the zuranolone and placebo groups was already significant at day 3 (P < .0001), and again at day 8 (P < .0001) and day 12 (P < .001).
At day 3, response rates on the HAMD-17 were significantly higher among zuranolone-treated patients than among those given placebo, at 29.3% versus 16.3% (P < .001). However, the differences on day 15 and on day 42 were no longer significant.
A similar effect was seen for HAMD-17 remissions, which were seen in 7.6% of zuranolone-treated patients and 2.3% of those given placebo at day 3 (P < .01), rising to 29.8% versus 27.1% at day 15, and 30.8% versus 29.6% at day 42, and neither difference was significant.
Dr. Brown also showed that, at all time points during the treatment and follow-up periods, improvements in response rates in Global Improvement on the Clinical Global Impression scale favored zuranolone.
On the SF-36v2 quality of life questionnaire, improvements again favored zuranolone on all domains, although the difference between active treatment and placebo was significant only for vitality on day 15, at 12.8 versus 9.7 points (P < .05).
Treatment-emergent adverse events were more common with zuranolone, with 60.1% of patients experiencing at least one event of any grade versus 44.6% with placebo. However, severe events were seen in only 3.0% versus 1.1% of patients, and serious adverse events were recorded in only two patients (0.7%) in both groups.
The most common adverse events were somnolence, dizziness, headache, sedation, and diarrhea, with no increase in suicidal ideation or withdrawal. Dr. Brown noted that there was “no change in the safety signal” between patients with or without prior antidepressant therapy.
From the audience, Marie-Josée Filteau, MD, department of psychiatry, Laval University, Quebec, drew attention to the similarity in the improvement in HAMD-17 scores between the zuranolone and placebo groups, asking: “How is that compelling?”
Dr. Brown replied that “what they are excited about is that change from baseline with zuranolone,” adding: “You do see it in the placebo group as well, and ... this isn’t new to psychiatry.
“This is a heterogeneous disease, and remember this [study] was conducted during COVID, so patients were being seen with clinic visits during COVID.
“What impact did that have? The placebo is not really placebo” in this case.
More effective than results suggest?
Approached for comment by this news organization, Maurizio Fava, MD, executive vice chair, department of psychiatry, and executive director, Clinical Trials Network and Institute, Massachusetts General Hospital, Boston, noted there are several issues with the trial.
Because of those, the drug “is likely to be much more efficacious than it looks because it achieved statistical significance despite an extremely high placebo response,” he said
“Whenever your change on placebo is greater than 10 points on the HAMD, you have an excessive response ... and a very, very low chance of detecting a signal,” he said.
Dr. Fava said that another issue was including patients who were either on or off antidepressants, which meant the population was not sufficiently homogenous.
Another “flaw” was to assume that the placebo effect would be “transient” and deteriorate over time, whereas the results showed the opposite.
Nevertheless, “it’s a positive study because of the sample size ... that provides further evidence for the antidepressant activity of zuranolone” and the drug was “well tolerated.”
Dr. Fava expects zuranolone “will make it to the market,” as an indication from the Food and Drug Administration is likely, “but if you’re asking me: Is the drug as effective as shown in their studies? It’s probably much more effective.”
The study was funded by Sage Therapeutics and Biogen. Dr. Brown is an employee of Sage Therapeutics. Lead investigator Anita Clayton, MD, University of Virginia, Charlottesville, has reported relationships with Dario Bioscience, Janssen, Praxis Precision Medicines, Relmada Therapeutics, Sage Therapeutics, Fabre-Kramer, MindCure, Ovoca Bio, PureTech Health, S1 Biopharma, Vella Bioscience, WCG MedAvante-ProPhase, Ballantine Books/Random House, Guilford Publications, Euthymics, and Mediflix.
A version of this article first appeared on Medscape.com.
Study results of an experimental agent that improves symptoms of major depression and boosts quality of life in as little as 3 days suggest it may be an effective short-term treatment option.
Phase 3 results of a randomized, placebo-controlled trial compared zuranolone, an neuroactive steroid that binds to both synaptic and extra-synaptic GABA-A receptors, to placebo in patients with major depressive disorder (MDD). Overall, 30% of participants were already taking antidepressants.
Investigators found the drug was associated with a significant improvement in depression scores versus placebo, with benefit observed as early as day 3. This was accompanied by improved function and well-being.
, said study presenter Colville Brown, MD, Sage Therapeutics, Cambridge, Mass.
“These data continue to support the development of zuranolone as a potential 14-day short course treatment for major depressive disorder episodes.”
The findings were presented at the virtual congress of the European College of Neuropsychopharmacology.
High placebo response
However, despite being significant, the drug’s benefit was only slightly higher than that of placebo, raising questions about the study design and the true performance of the drug.
Dr. Brown explained that patients with MDD were randomized to oral zuranolone 50 mg or placebo once daily for 14 days, with dose reductions to 40 mg or matching placebo permitted in case of perceived intolerance.
Patients were assessed at baseline and day 15 via the 17-item Hamilton Rating Scale for Depression (HAMD-17) before entering a 28-day follow-up period off the study drug.
Among the 268 participants who received zuranolone, 90.3% completed the study, compared with 87.4% of 269 patients in the placebo group.
The mean age of participants was 40 years. Women made up 69.4% of those who received zuranolone and 61.7% assigned to placebo.
The mean HAMD-17 score at baseline was 26.8 and 26.9 in the zuranolone and placebo groups, respectively. Dr. Brown noted that 29.5% of patients in the zuranolone group and 30.1% of those assigned to placebo were taking antidepressants at baseline.
The study’s primary endpoint was met, with patients taking the study drug experiencing a significantly greater reduction in HAMD-17 scores from baseline to day 15 versus those given placebo, at 14.1 versus 12.3 points (P = .0141).
Dr. Brown highlighted that the difference in reduction in HAMD-17 scores between the zuranolone and placebo groups was already significant at day 3 (P < .0001), and again at day 8 (P < .0001) and day 12 (P < .001).
At day 3, response rates on the HAMD-17 were significantly higher among zuranolone-treated patients than among those given placebo, at 29.3% versus 16.3% (P < .001). However, the differences on day 15 and on day 42 were no longer significant.
A similar effect was seen for HAMD-17 remissions, which were seen in 7.6% of zuranolone-treated patients and 2.3% of those given placebo at day 3 (P < .01), rising to 29.8% versus 27.1% at day 15, and 30.8% versus 29.6% at day 42, and neither difference was significant.
Dr. Brown also showed that, at all time points during the treatment and follow-up periods, improvements in response rates in Global Improvement on the Clinical Global Impression scale favored zuranolone.
On the SF-36v2 quality of life questionnaire, improvements again favored zuranolone on all domains, although the difference between active treatment and placebo was significant only for vitality on day 15, at 12.8 versus 9.7 points (P < .05).
Treatment-emergent adverse events were more common with zuranolone, with 60.1% of patients experiencing at least one event of any grade versus 44.6% with placebo. However, severe events were seen in only 3.0% versus 1.1% of patients, and serious adverse events were recorded in only two patients (0.7%) in both groups.
The most common adverse events were somnolence, dizziness, headache, sedation, and diarrhea, with no increase in suicidal ideation or withdrawal. Dr. Brown noted that there was “no change in the safety signal” between patients with or without prior antidepressant therapy.
From the audience, Marie-Josée Filteau, MD, department of psychiatry, Laval University, Quebec, drew attention to the similarity in the improvement in HAMD-17 scores between the zuranolone and placebo groups, asking: “How is that compelling?”
Dr. Brown replied that “what they are excited about is that change from baseline with zuranolone,” adding: “You do see it in the placebo group as well, and ... this isn’t new to psychiatry.
“This is a heterogeneous disease, and remember this [study] was conducted during COVID, so patients were being seen with clinic visits during COVID.
“What impact did that have? The placebo is not really placebo” in this case.
More effective than results suggest?
Approached for comment by this news organization, Maurizio Fava, MD, executive vice chair, department of psychiatry, and executive director, Clinical Trials Network and Institute, Massachusetts General Hospital, Boston, noted there are several issues with the trial.
Because of those, the drug “is likely to be much more efficacious than it looks because it achieved statistical significance despite an extremely high placebo response,” he said
“Whenever your change on placebo is greater than 10 points on the HAMD, you have an excessive response ... and a very, very low chance of detecting a signal,” he said.
Dr. Fava said that another issue was including patients who were either on or off antidepressants, which meant the population was not sufficiently homogenous.
Another “flaw” was to assume that the placebo effect would be “transient” and deteriorate over time, whereas the results showed the opposite.
Nevertheless, “it’s a positive study because of the sample size ... that provides further evidence for the antidepressant activity of zuranolone” and the drug was “well tolerated.”
Dr. Fava expects zuranolone “will make it to the market,” as an indication from the Food and Drug Administration is likely, “but if you’re asking me: Is the drug as effective as shown in their studies? It’s probably much more effective.”
The study was funded by Sage Therapeutics and Biogen. Dr. Brown is an employee of Sage Therapeutics. Lead investigator Anita Clayton, MD, University of Virginia, Charlottesville, has reported relationships with Dario Bioscience, Janssen, Praxis Precision Medicines, Relmada Therapeutics, Sage Therapeutics, Fabre-Kramer, MindCure, Ovoca Bio, PureTech Health, S1 Biopharma, Vella Bioscience, WCG MedAvante-ProPhase, Ballantine Books/Random House, Guilford Publications, Euthymics, and Mediflix.
A version of this article first appeared on Medscape.com.
FROM ECNP 2021







