User login
Telemedicine models show some benefit in OA
Remote interventions using an Internet-based app and telephone outreach to engage patients with osteoarthritis to self-manage their disease have demonstrated the potential to improve some symptoms, at least in the short term, showing the potential for tools to interact with OA patients without having them come into an office or clinic.
Remote interaction using these two forms of telemedicine – one a sophisticated digital platform, the other using a device that’s been around for almost 150 years – may have greater utility for keeping physicians connected with their OA patients during the COVID-19 pandemic, OA experts said in an interview.
“This is certainly relevant during the pandemic, but this has been of high interest for years as well, as researchers and clinicians have been seeking the best ways to reach patients with these types of programs,” said Kelli Allen, PhD, a research health scientist at the University of North Carolina at Chapel Hill.
Two separate studies evaluated the telemedicine platforms. In JAMA Internal Medicine, researchers reported that telephone-based cognitive-behavioral therapy (CBT) for patients aged 60 and older with OA and insomnia led to improved sleep, fatigue and, to a lesser extent, pain, in a randomized, controlled trial with 327 patients.
A separate randomized, controlled trial of 105 OA patients at the University of Nottingham (England), published in JAMA Network Open, reported that users of a smartphone-based exercise intervention app had greater improvements in pain and function than did controls.
“I think these two studies represent a first step in terms of moving forward, and certainly the interventions could be refined and potentially combined together for patients in the future,” said C. Kent Kwoh, MD, director of the University of Arizona Arthritis Center in Tucson.
Phone-based CBT study
The telephone-based CBT study consisted of two groups: the CBT group (n = 163) who completed six 20- to 30-minute telephone calls over 8 weeks, kept daily diaries, and received tailored educational materials and an education-only group (n = 164). At 2 months after treatment, Insomnia Severity Index scores decreased 8.1 points on average in the CBT group versus 4.8 points in the education-only patients (P < .001).
That variation between the intervention group and controls was sustained out to a year: 7.7 points lower than baseline versus 4.7 points lower. At the same time point, 56.3% of the CBT group remained in remission with Insomnia Severity Index scores less than 7 versus 25.8% of controls. Fatigue outcomes were similarly disparate between the groups.
Pain outcomes were a different story, however. “Post treatment, significant differences were observed for pain, but these differences were not sustained at 12-month follow-up,” first author Susan M. McCurry, PhD, a clinical psychologist and faculty member at the University of Washington, Seattle, and colleagues wrote.
“I think their positive findings illustrate that remotely delivered interventions can be ‘low tech’ and still effective,” Dr. Allen said of the CBT phone study. She noted that complete case data were available for 282 of 327 patients. “The high rate of session attendance suggests that they chose a delivery modality appropriate for their target patient group.”
The scalability of the telephone model is noteworthy, Dr. Kwoh said. “Having a telemedicine intervention that could be scaled a little more easily rather than an in-person intervention, and having individualized treatment, that’s beneficial, as is targeting two symptoms that are very bothersome and burdensome to patients with OA: insomnia and fatigue.” Following patients out to 12 months is a strength of the study, he added.
Smartphone app–based exercise study
The U.K. study evaluated 6-week outcomes of 48 patients with knee OA who used a proprietary app-based exercise program (Joint Academy) and 57 controls who used traditional self-management. The app provided daily exercises and texts, along with email and smartphone reminders. The app was derived from the Better Management of Patients with OA program initiated in Sweden in 2008 that used OA treatment guidelines for education and exercise in person in primary care clinics.
App users showed a 1.5-point reduction in numeric rating scale (NRS) pain score at 6 weeks versus virtually no change in controls (P < .001). In terms of secondary outcomes, pain scores improved 2.2 points on average for app users versus 1.2 for controls (P = .02), with similar improvements recorded in both stiffness and physical function.
Average change in the 30-second sit-to-stand test measured 4.5 for the app users and 1.2 for the usual-care group (P < .001). The study found no difference between the two groups in changes in temporal summation, conditional pain modulation, or Arthritis Research UK Musculoskeletal Health Questionnaire scores.
First author Sameer Akram Gohir, MSc, PhD, and colleagues wrote that the reasons for differences in outcomes between app users and controls aren’t clear. “The superior outcome in the intervention group may depend on the content and context in the app, including a combination of standardized exercises and information, as well as using a digital delivery system.”
Data gathering was cut short because of COVID-19 restrictions in the United Kingdom, as 27 patients missed their in-person follow-up visits. That was one shortcoming of the study, Dr. Kwoh noted.
“Given the caveats certainly they were able to show robust changes in terms of decreased pain, and also improvement in a variety of performance measures. Certainly this may be beneficial – we don’t know – in terms of cost-effectiveness, but it may be beneficial for insurance companies to adapt such a program,” he said, adding that future studies into the cost effectiveness of the digital platform would be in order.
“Certainly, if this program were to decrease physician visits or postpone the need for joint replacement for individuals, then it could be certainly very cost effective,” Dr. Kwoh said.
The completion rate among patients in the study – almost 90% – was “impressive,” Dr. Allen said. “However, this is a relatively short-term study, and I think an important question for future research is whether patients continue with this level of engagement for a longer period of time.”
Dr. McCurry had no relevant financial relationships to disclose. The CBT phone study received funding from the Public Health Service and the National Institute on Aging. Coauthors disclosed relationships with Campbell Alliance Group, Mapi Research Trust, and Pfizer. Dr. Gohir reported no relevant financial relationships. The study received funding from the Versus Arthritis UK Plan Center, the National Institute for Health Research Nottingham Biomedical Research Center, and Pfizer Global. The Joint Academy provided software for the study. A coauthor reported a financial relationships with Pfizer. Dr. Kwoh said that in the past year he has consulted for Express Scripts, Kolon Tissue Gene, LG Chem, and Regeneron. In the past year, he also received institutional grants for clinical trials from AbbVie, Cumberland, Eicos, Eli Lilly, GlaxoSmithKline, Mitsubishi, and Pfizer. Dr. Allen had no relevant financial relationships to disclose.
Remote interventions using an Internet-based app and telephone outreach to engage patients with osteoarthritis to self-manage their disease have demonstrated the potential to improve some symptoms, at least in the short term, showing the potential for tools to interact with OA patients without having them come into an office or clinic.
Remote interaction using these two forms of telemedicine – one a sophisticated digital platform, the other using a device that’s been around for almost 150 years – may have greater utility for keeping physicians connected with their OA patients during the COVID-19 pandemic, OA experts said in an interview.
“This is certainly relevant during the pandemic, but this has been of high interest for years as well, as researchers and clinicians have been seeking the best ways to reach patients with these types of programs,” said Kelli Allen, PhD, a research health scientist at the University of North Carolina at Chapel Hill.
Two separate studies evaluated the telemedicine platforms. In JAMA Internal Medicine, researchers reported that telephone-based cognitive-behavioral therapy (CBT) for patients aged 60 and older with OA and insomnia led to improved sleep, fatigue and, to a lesser extent, pain, in a randomized, controlled trial with 327 patients.
A separate randomized, controlled trial of 105 OA patients at the University of Nottingham (England), published in JAMA Network Open, reported that users of a smartphone-based exercise intervention app had greater improvements in pain and function than did controls.
“I think these two studies represent a first step in terms of moving forward, and certainly the interventions could be refined and potentially combined together for patients in the future,” said C. Kent Kwoh, MD, director of the University of Arizona Arthritis Center in Tucson.
Phone-based CBT study
The telephone-based CBT study consisted of two groups: the CBT group (n = 163) who completed six 20- to 30-minute telephone calls over 8 weeks, kept daily diaries, and received tailored educational materials and an education-only group (n = 164). At 2 months after treatment, Insomnia Severity Index scores decreased 8.1 points on average in the CBT group versus 4.8 points in the education-only patients (P < .001).
That variation between the intervention group and controls was sustained out to a year: 7.7 points lower than baseline versus 4.7 points lower. At the same time point, 56.3% of the CBT group remained in remission with Insomnia Severity Index scores less than 7 versus 25.8% of controls. Fatigue outcomes were similarly disparate between the groups.
Pain outcomes were a different story, however. “Post treatment, significant differences were observed for pain, but these differences were not sustained at 12-month follow-up,” first author Susan M. McCurry, PhD, a clinical psychologist and faculty member at the University of Washington, Seattle, and colleagues wrote.
“I think their positive findings illustrate that remotely delivered interventions can be ‘low tech’ and still effective,” Dr. Allen said of the CBT phone study. She noted that complete case data were available for 282 of 327 patients. “The high rate of session attendance suggests that they chose a delivery modality appropriate for their target patient group.”
The scalability of the telephone model is noteworthy, Dr. Kwoh said. “Having a telemedicine intervention that could be scaled a little more easily rather than an in-person intervention, and having individualized treatment, that’s beneficial, as is targeting two symptoms that are very bothersome and burdensome to patients with OA: insomnia and fatigue.” Following patients out to 12 months is a strength of the study, he added.
Smartphone app–based exercise study
The U.K. study evaluated 6-week outcomes of 48 patients with knee OA who used a proprietary app-based exercise program (Joint Academy) and 57 controls who used traditional self-management. The app provided daily exercises and texts, along with email and smartphone reminders. The app was derived from the Better Management of Patients with OA program initiated in Sweden in 2008 that used OA treatment guidelines for education and exercise in person in primary care clinics.
App users showed a 1.5-point reduction in numeric rating scale (NRS) pain score at 6 weeks versus virtually no change in controls (P < .001). In terms of secondary outcomes, pain scores improved 2.2 points on average for app users versus 1.2 for controls (P = .02), with similar improvements recorded in both stiffness and physical function.
Average change in the 30-second sit-to-stand test measured 4.5 for the app users and 1.2 for the usual-care group (P < .001). The study found no difference between the two groups in changes in temporal summation, conditional pain modulation, or Arthritis Research UK Musculoskeletal Health Questionnaire scores.
First author Sameer Akram Gohir, MSc, PhD, and colleagues wrote that the reasons for differences in outcomes between app users and controls aren’t clear. “The superior outcome in the intervention group may depend on the content and context in the app, including a combination of standardized exercises and information, as well as using a digital delivery system.”
Data gathering was cut short because of COVID-19 restrictions in the United Kingdom, as 27 patients missed their in-person follow-up visits. That was one shortcoming of the study, Dr. Kwoh noted.
“Given the caveats certainly they were able to show robust changes in terms of decreased pain, and also improvement in a variety of performance measures. Certainly this may be beneficial – we don’t know – in terms of cost-effectiveness, but it may be beneficial for insurance companies to adapt such a program,” he said, adding that future studies into the cost effectiveness of the digital platform would be in order.
“Certainly, if this program were to decrease physician visits or postpone the need for joint replacement for individuals, then it could be certainly very cost effective,” Dr. Kwoh said.
The completion rate among patients in the study – almost 90% – was “impressive,” Dr. Allen said. “However, this is a relatively short-term study, and I think an important question for future research is whether patients continue with this level of engagement for a longer period of time.”
Dr. McCurry had no relevant financial relationships to disclose. The CBT phone study received funding from the Public Health Service and the National Institute on Aging. Coauthors disclosed relationships with Campbell Alliance Group, Mapi Research Trust, and Pfizer. Dr. Gohir reported no relevant financial relationships. The study received funding from the Versus Arthritis UK Plan Center, the National Institute for Health Research Nottingham Biomedical Research Center, and Pfizer Global. The Joint Academy provided software for the study. A coauthor reported a financial relationships with Pfizer. Dr. Kwoh said that in the past year he has consulted for Express Scripts, Kolon Tissue Gene, LG Chem, and Regeneron. In the past year, he also received institutional grants for clinical trials from AbbVie, Cumberland, Eicos, Eli Lilly, GlaxoSmithKline, Mitsubishi, and Pfizer. Dr. Allen had no relevant financial relationships to disclose.
Remote interventions using an Internet-based app and telephone outreach to engage patients with osteoarthritis to self-manage their disease have demonstrated the potential to improve some symptoms, at least in the short term, showing the potential for tools to interact with OA patients without having them come into an office or clinic.
Remote interaction using these two forms of telemedicine – one a sophisticated digital platform, the other using a device that’s been around for almost 150 years – may have greater utility for keeping physicians connected with their OA patients during the COVID-19 pandemic, OA experts said in an interview.
“This is certainly relevant during the pandemic, but this has been of high interest for years as well, as researchers and clinicians have been seeking the best ways to reach patients with these types of programs,” said Kelli Allen, PhD, a research health scientist at the University of North Carolina at Chapel Hill.
Two separate studies evaluated the telemedicine platforms. In JAMA Internal Medicine, researchers reported that telephone-based cognitive-behavioral therapy (CBT) for patients aged 60 and older with OA and insomnia led to improved sleep, fatigue and, to a lesser extent, pain, in a randomized, controlled trial with 327 patients.
A separate randomized, controlled trial of 105 OA patients at the University of Nottingham (England), published in JAMA Network Open, reported that users of a smartphone-based exercise intervention app had greater improvements in pain and function than did controls.
“I think these two studies represent a first step in terms of moving forward, and certainly the interventions could be refined and potentially combined together for patients in the future,” said C. Kent Kwoh, MD, director of the University of Arizona Arthritis Center in Tucson.
Phone-based CBT study
The telephone-based CBT study consisted of two groups: the CBT group (n = 163) who completed six 20- to 30-minute telephone calls over 8 weeks, kept daily diaries, and received tailored educational materials and an education-only group (n = 164). At 2 months after treatment, Insomnia Severity Index scores decreased 8.1 points on average in the CBT group versus 4.8 points in the education-only patients (P < .001).
That variation between the intervention group and controls was sustained out to a year: 7.7 points lower than baseline versus 4.7 points lower. At the same time point, 56.3% of the CBT group remained in remission with Insomnia Severity Index scores less than 7 versus 25.8% of controls. Fatigue outcomes were similarly disparate between the groups.
Pain outcomes were a different story, however. “Post treatment, significant differences were observed for pain, but these differences were not sustained at 12-month follow-up,” first author Susan M. McCurry, PhD, a clinical psychologist and faculty member at the University of Washington, Seattle, and colleagues wrote.
“I think their positive findings illustrate that remotely delivered interventions can be ‘low tech’ and still effective,” Dr. Allen said of the CBT phone study. She noted that complete case data were available for 282 of 327 patients. “The high rate of session attendance suggests that they chose a delivery modality appropriate for their target patient group.”
The scalability of the telephone model is noteworthy, Dr. Kwoh said. “Having a telemedicine intervention that could be scaled a little more easily rather than an in-person intervention, and having individualized treatment, that’s beneficial, as is targeting two symptoms that are very bothersome and burdensome to patients with OA: insomnia and fatigue.” Following patients out to 12 months is a strength of the study, he added.
Smartphone app–based exercise study
The U.K. study evaluated 6-week outcomes of 48 patients with knee OA who used a proprietary app-based exercise program (Joint Academy) and 57 controls who used traditional self-management. The app provided daily exercises and texts, along with email and smartphone reminders. The app was derived from the Better Management of Patients with OA program initiated in Sweden in 2008 that used OA treatment guidelines for education and exercise in person in primary care clinics.
App users showed a 1.5-point reduction in numeric rating scale (NRS) pain score at 6 weeks versus virtually no change in controls (P < .001). In terms of secondary outcomes, pain scores improved 2.2 points on average for app users versus 1.2 for controls (P = .02), with similar improvements recorded in both stiffness and physical function.
Average change in the 30-second sit-to-stand test measured 4.5 for the app users and 1.2 for the usual-care group (P < .001). The study found no difference between the two groups in changes in temporal summation, conditional pain modulation, or Arthritis Research UK Musculoskeletal Health Questionnaire scores.
First author Sameer Akram Gohir, MSc, PhD, and colleagues wrote that the reasons for differences in outcomes between app users and controls aren’t clear. “The superior outcome in the intervention group may depend on the content and context in the app, including a combination of standardized exercises and information, as well as using a digital delivery system.”
Data gathering was cut short because of COVID-19 restrictions in the United Kingdom, as 27 patients missed their in-person follow-up visits. That was one shortcoming of the study, Dr. Kwoh noted.
“Given the caveats certainly they were able to show robust changes in terms of decreased pain, and also improvement in a variety of performance measures. Certainly this may be beneficial – we don’t know – in terms of cost-effectiveness, but it may be beneficial for insurance companies to adapt such a program,” he said, adding that future studies into the cost effectiveness of the digital platform would be in order.
“Certainly, if this program were to decrease physician visits or postpone the need for joint replacement for individuals, then it could be certainly very cost effective,” Dr. Kwoh said.
The completion rate among patients in the study – almost 90% – was “impressive,” Dr. Allen said. “However, this is a relatively short-term study, and I think an important question for future research is whether patients continue with this level of engagement for a longer period of time.”
Dr. McCurry had no relevant financial relationships to disclose. The CBT phone study received funding from the Public Health Service and the National Institute on Aging. Coauthors disclosed relationships with Campbell Alliance Group, Mapi Research Trust, and Pfizer. Dr. Gohir reported no relevant financial relationships. The study received funding from the Versus Arthritis UK Plan Center, the National Institute for Health Research Nottingham Biomedical Research Center, and Pfizer Global. The Joint Academy provided software for the study. A coauthor reported a financial relationships with Pfizer. Dr. Kwoh said that in the past year he has consulted for Express Scripts, Kolon Tissue Gene, LG Chem, and Regeneron. In the past year, he also received institutional grants for clinical trials from AbbVie, Cumberland, Eicos, Eli Lilly, GlaxoSmithKline, Mitsubishi, and Pfizer. Dr. Allen had no relevant financial relationships to disclose.
FROM JAMA INTERNAL MEDICINE AND JAMA NETWORK OPEN
OA risk-reduction program targets injured knees
A novel educational and personalized physical therapy program is showing signs that it may help people to mitigate their risk of developing knee osteoarthritis after an injury.
Speaking at the Canadian Arthritis Research Conference: Research with Impact, Jackie Whittaker, PhD, observed that initial work from the Stop Osteoarthritis (SOAR) program showed that meaningful improvements in knee-related quality of life and improvement in participants’ perceived self-management could be achieved.
Further feasibility work is ongoing and a proof-of-concept and phase 3 study need to follow, but the research suggests the approach could potentially help to reduce the substantial burden of managing people who develop posttraumatic OA (PTOA) of the knee.
Understanding the post–knee injury period
“Despite the progress that we’ve made in preventing injuries, and reducing disability in people with osteoarthritis, we lack good evidence about what should be done in the period between joint injury and the onset of osteoarthritis to delay or halt that onset,” Dr. Whittaker said at the virtual meeting, which was sponsored by the Arthritis Society, the Canadian Rheumatology Association, and Canada’s Institute of Musculoskeletal Health and Arthritis.
That’s where the SOAR program comes in. For the past 8 years, Dr. Whittaker, an assistant professor in the department of physical therapy at the University of British Columbia in Vancouver and affiliated to Arthritis Research Canada, and collaborators have been looking into the post–knee injury period with the aim of developing an intervention that could potentially reduce the risk of OA further down the line.
Much work has gone into understanding the burden and risk factors for PTOA of the knee in order to know who exactly to target with the intervention and what the risk factors may be for the subsequent development of OA .
This research suggests that knee injuries are most commonly seen in people aged between 15 and 35 years who participated in sporting or other physical activities, so this is the target population for the SOAR intervention.
Broadly speaking, sustaining any knee injury is associated with a sixfold increased risk for subsequent PTOA, Dr. Whittaker observed.
“Despite the fact that ACL [anterior cruciate ligament] and meniscal tears get all the press, collateral ligament injury are still associated with about a fivefold increased risk of osteoarthritis, and therefore maybe shouldn’t be so easily dismissed as an important target,” Dr. Whittaker said.
Postinjury risk factors for OA
“Basically, what all prevention comes down to is our understanding of risk factors and our ability to be able to modify them,” she said.
Previous joint injury is one of the strongest and most established modifiable risk factors for developing knee OA, and Dr. Whittaker and associates have performed two small but “mighty” cohort studies comparing people who have and have not had a knee injury. These two studies have looked at different time periods following injury to see if they could first identify the risk factors for developing OA some 3-10 years later, and then to look more closely at some of those risk factors in first 2 years after injury with a view to targeting these with an intervention.
Data analysis of the latter study is still ongoing but have shown that, among injured subjects, there is a fear of movement and reinjury, knee strength is weaker in both injured and uninjured knees, and they are perhaps less physically active than those who have not been injured.
“Going into those two studies, we knew that this group of people already [had an] increased risk for osteoarthritis because they had an injury. However, what we found is that it looks like this risk may be compounded through adiposity [and] deficits in muscle strength and physical inactivity, which are associated with pain, stiffness, lack of confidence, and at times, unrealistic expectations and poor pacing,” Dr. Whittaker said.
She added: “It also looks like some of these additional factors and particular adiposity or fat gain may develop after injury, which would then give us a concrete target for delaying or halting the onset of osteoarthritis in the segment of the population.”
SOAR program components
The SOAR program intervention is an 8-week, physiotherapist-led program that targets people aged 15-35 years who have had a sport-related knee injury and received formal care. All of this is conducted via videoconferencing software and starts off with a 2-hour group education session or “knee camp.” This is followed by a one-on-one assessment with a physiotherapist and setting exercise and physical activity goals for the week.
Participants then undertake their personalized exercise and physical activity programs at home and track their progress using an activity monitor. They can participate in an optional weekly group exercise class and receive weekly one-on-one physiotherapy counseling where goals can be modified and any issues participants might be experiencing solved.
According to Dr. Whittaker, “this program really aims to increase participants capacity to manage their elevated risk for osteoarthritis, and we’re doing this by also optimizing their knee muscle function and their physical activity participation.”
While the knee camp enables a therapeutic alliance to be formed between participants and their physiotherapists, the weekly group classes provide social support and an opportunity to interact with others.
“Brief action planning builds self-efficacy [and] promotes autonomous health behaviors, while goal setting and tracking provide accountability, feedback about progress, and facilitated adherence,” she said.
And finally, regular communication with a physiotherapist in the program ensures timely support to learn how to navigate obstacles and helps participants to learn how to deal with their own knee health.
Testing the feasibility of the SOAR program intervention
“Currently we are smack in the middle of our feasibility study,” Dr. Whittaker said. So far, four physiotherapists have been trained to deliver an abridged, 4-week version of the program, and 25 of a planned 30 participants have been enrolled.
Results seem promising so far. No participants have dropped out of the program to date and attendance is at 100%.
“Based on data from the first 12 participants who completed the program, we are meeting all of our ‘a priori’ program benchmarks,” Dr. Whittaker said.
“It is very early days,” she emphasized, but “we are excited to see clinically important improvements in both knee-related quality of life and perceived self-management.
“This gives us some confidence that maybe all this time that we’ve put into developing our intervention is paying off, but obviously time will tell if we’re headed in the right direction,” she said. “Perhaps in time, we may be able to look at whether or not the individuals that participated in that program have fewer symptoms of OA disease. But that will obviously take us a few years before we’ll be able to get to that point.”
Dr. Whittaker acknowledged receiving funding for the SOAR program from the Arthritis Society, the Michael Smith Foundation for Health Research, BC SUPPORT Unit, and the Canadian Musculoskeletal Rehab Network.
A novel educational and personalized physical therapy program is showing signs that it may help people to mitigate their risk of developing knee osteoarthritis after an injury.
Speaking at the Canadian Arthritis Research Conference: Research with Impact, Jackie Whittaker, PhD, observed that initial work from the Stop Osteoarthritis (SOAR) program showed that meaningful improvements in knee-related quality of life and improvement in participants’ perceived self-management could be achieved.
Further feasibility work is ongoing and a proof-of-concept and phase 3 study need to follow, but the research suggests the approach could potentially help to reduce the substantial burden of managing people who develop posttraumatic OA (PTOA) of the knee.
Understanding the post–knee injury period
“Despite the progress that we’ve made in preventing injuries, and reducing disability in people with osteoarthritis, we lack good evidence about what should be done in the period between joint injury and the onset of osteoarthritis to delay or halt that onset,” Dr. Whittaker said at the virtual meeting, which was sponsored by the Arthritis Society, the Canadian Rheumatology Association, and Canada’s Institute of Musculoskeletal Health and Arthritis.
That’s where the SOAR program comes in. For the past 8 years, Dr. Whittaker, an assistant professor in the department of physical therapy at the University of British Columbia in Vancouver and affiliated to Arthritis Research Canada, and collaborators have been looking into the post–knee injury period with the aim of developing an intervention that could potentially reduce the risk of OA further down the line.
Much work has gone into understanding the burden and risk factors for PTOA of the knee in order to know who exactly to target with the intervention and what the risk factors may be for the subsequent development of OA .
This research suggests that knee injuries are most commonly seen in people aged between 15 and 35 years who participated in sporting or other physical activities, so this is the target population for the SOAR intervention.
Broadly speaking, sustaining any knee injury is associated with a sixfold increased risk for subsequent PTOA, Dr. Whittaker observed.
“Despite the fact that ACL [anterior cruciate ligament] and meniscal tears get all the press, collateral ligament injury are still associated with about a fivefold increased risk of osteoarthritis, and therefore maybe shouldn’t be so easily dismissed as an important target,” Dr. Whittaker said.
Postinjury risk factors for OA
“Basically, what all prevention comes down to is our understanding of risk factors and our ability to be able to modify them,” she said.
Previous joint injury is one of the strongest and most established modifiable risk factors for developing knee OA, and Dr. Whittaker and associates have performed two small but “mighty” cohort studies comparing people who have and have not had a knee injury. These two studies have looked at different time periods following injury to see if they could first identify the risk factors for developing OA some 3-10 years later, and then to look more closely at some of those risk factors in first 2 years after injury with a view to targeting these with an intervention.
Data analysis of the latter study is still ongoing but have shown that, among injured subjects, there is a fear of movement and reinjury, knee strength is weaker in both injured and uninjured knees, and they are perhaps less physically active than those who have not been injured.
“Going into those two studies, we knew that this group of people already [had an] increased risk for osteoarthritis because they had an injury. However, what we found is that it looks like this risk may be compounded through adiposity [and] deficits in muscle strength and physical inactivity, which are associated with pain, stiffness, lack of confidence, and at times, unrealistic expectations and poor pacing,” Dr. Whittaker said.
She added: “It also looks like some of these additional factors and particular adiposity or fat gain may develop after injury, which would then give us a concrete target for delaying or halting the onset of osteoarthritis in the segment of the population.”
SOAR program components
The SOAR program intervention is an 8-week, physiotherapist-led program that targets people aged 15-35 years who have had a sport-related knee injury and received formal care. All of this is conducted via videoconferencing software and starts off with a 2-hour group education session or “knee camp.” This is followed by a one-on-one assessment with a physiotherapist and setting exercise and physical activity goals for the week.
Participants then undertake their personalized exercise and physical activity programs at home and track their progress using an activity monitor. They can participate in an optional weekly group exercise class and receive weekly one-on-one physiotherapy counseling where goals can be modified and any issues participants might be experiencing solved.
According to Dr. Whittaker, “this program really aims to increase participants capacity to manage their elevated risk for osteoarthritis, and we’re doing this by also optimizing their knee muscle function and their physical activity participation.”
While the knee camp enables a therapeutic alliance to be formed between participants and their physiotherapists, the weekly group classes provide social support and an opportunity to interact with others.
“Brief action planning builds self-efficacy [and] promotes autonomous health behaviors, while goal setting and tracking provide accountability, feedback about progress, and facilitated adherence,” she said.
And finally, regular communication with a physiotherapist in the program ensures timely support to learn how to navigate obstacles and helps participants to learn how to deal with their own knee health.
Testing the feasibility of the SOAR program intervention
“Currently we are smack in the middle of our feasibility study,” Dr. Whittaker said. So far, four physiotherapists have been trained to deliver an abridged, 4-week version of the program, and 25 of a planned 30 participants have been enrolled.
Results seem promising so far. No participants have dropped out of the program to date and attendance is at 100%.
“Based on data from the first 12 participants who completed the program, we are meeting all of our ‘a priori’ program benchmarks,” Dr. Whittaker said.
“It is very early days,” she emphasized, but “we are excited to see clinically important improvements in both knee-related quality of life and perceived self-management.
“This gives us some confidence that maybe all this time that we’ve put into developing our intervention is paying off, but obviously time will tell if we’re headed in the right direction,” she said. “Perhaps in time, we may be able to look at whether or not the individuals that participated in that program have fewer symptoms of OA disease. But that will obviously take us a few years before we’ll be able to get to that point.”
Dr. Whittaker acknowledged receiving funding for the SOAR program from the Arthritis Society, the Michael Smith Foundation for Health Research, BC SUPPORT Unit, and the Canadian Musculoskeletal Rehab Network.
A novel educational and personalized physical therapy program is showing signs that it may help people to mitigate their risk of developing knee osteoarthritis after an injury.
Speaking at the Canadian Arthritis Research Conference: Research with Impact, Jackie Whittaker, PhD, observed that initial work from the Stop Osteoarthritis (SOAR) program showed that meaningful improvements in knee-related quality of life and improvement in participants’ perceived self-management could be achieved.
Further feasibility work is ongoing and a proof-of-concept and phase 3 study need to follow, but the research suggests the approach could potentially help to reduce the substantial burden of managing people who develop posttraumatic OA (PTOA) of the knee.
Understanding the post–knee injury period
“Despite the progress that we’ve made in preventing injuries, and reducing disability in people with osteoarthritis, we lack good evidence about what should be done in the period between joint injury and the onset of osteoarthritis to delay or halt that onset,” Dr. Whittaker said at the virtual meeting, which was sponsored by the Arthritis Society, the Canadian Rheumatology Association, and Canada’s Institute of Musculoskeletal Health and Arthritis.
That’s where the SOAR program comes in. For the past 8 years, Dr. Whittaker, an assistant professor in the department of physical therapy at the University of British Columbia in Vancouver and affiliated to Arthritis Research Canada, and collaborators have been looking into the post–knee injury period with the aim of developing an intervention that could potentially reduce the risk of OA further down the line.
Much work has gone into understanding the burden and risk factors for PTOA of the knee in order to know who exactly to target with the intervention and what the risk factors may be for the subsequent development of OA .
This research suggests that knee injuries are most commonly seen in people aged between 15 and 35 years who participated in sporting or other physical activities, so this is the target population for the SOAR intervention.
Broadly speaking, sustaining any knee injury is associated with a sixfold increased risk for subsequent PTOA, Dr. Whittaker observed.
“Despite the fact that ACL [anterior cruciate ligament] and meniscal tears get all the press, collateral ligament injury are still associated with about a fivefold increased risk of osteoarthritis, and therefore maybe shouldn’t be so easily dismissed as an important target,” Dr. Whittaker said.
Postinjury risk factors for OA
“Basically, what all prevention comes down to is our understanding of risk factors and our ability to be able to modify them,” she said.
Previous joint injury is one of the strongest and most established modifiable risk factors for developing knee OA, and Dr. Whittaker and associates have performed two small but “mighty” cohort studies comparing people who have and have not had a knee injury. These two studies have looked at different time periods following injury to see if they could first identify the risk factors for developing OA some 3-10 years later, and then to look more closely at some of those risk factors in first 2 years after injury with a view to targeting these with an intervention.
Data analysis of the latter study is still ongoing but have shown that, among injured subjects, there is a fear of movement and reinjury, knee strength is weaker in both injured and uninjured knees, and they are perhaps less physically active than those who have not been injured.
“Going into those two studies, we knew that this group of people already [had an] increased risk for osteoarthritis because they had an injury. However, what we found is that it looks like this risk may be compounded through adiposity [and] deficits in muscle strength and physical inactivity, which are associated with pain, stiffness, lack of confidence, and at times, unrealistic expectations and poor pacing,” Dr. Whittaker said.
She added: “It also looks like some of these additional factors and particular adiposity or fat gain may develop after injury, which would then give us a concrete target for delaying or halting the onset of osteoarthritis in the segment of the population.”
SOAR program components
The SOAR program intervention is an 8-week, physiotherapist-led program that targets people aged 15-35 years who have had a sport-related knee injury and received formal care. All of this is conducted via videoconferencing software and starts off with a 2-hour group education session or “knee camp.” This is followed by a one-on-one assessment with a physiotherapist and setting exercise and physical activity goals for the week.
Participants then undertake their personalized exercise and physical activity programs at home and track their progress using an activity monitor. They can participate in an optional weekly group exercise class and receive weekly one-on-one physiotherapy counseling where goals can be modified and any issues participants might be experiencing solved.
According to Dr. Whittaker, “this program really aims to increase participants capacity to manage their elevated risk for osteoarthritis, and we’re doing this by also optimizing their knee muscle function and their physical activity participation.”
While the knee camp enables a therapeutic alliance to be formed between participants and their physiotherapists, the weekly group classes provide social support and an opportunity to interact with others.
“Brief action planning builds self-efficacy [and] promotes autonomous health behaviors, while goal setting and tracking provide accountability, feedback about progress, and facilitated adherence,” she said.
And finally, regular communication with a physiotherapist in the program ensures timely support to learn how to navigate obstacles and helps participants to learn how to deal with their own knee health.
Testing the feasibility of the SOAR program intervention
“Currently we are smack in the middle of our feasibility study,” Dr. Whittaker said. So far, four physiotherapists have been trained to deliver an abridged, 4-week version of the program, and 25 of a planned 30 participants have been enrolled.
Results seem promising so far. No participants have dropped out of the program to date and attendance is at 100%.
“Based on data from the first 12 participants who completed the program, we are meeting all of our ‘a priori’ program benchmarks,” Dr. Whittaker said.
“It is very early days,” she emphasized, but “we are excited to see clinically important improvements in both knee-related quality of life and perceived self-management.
“This gives us some confidence that maybe all this time that we’ve put into developing our intervention is paying off, but obviously time will tell if we’re headed in the right direction,” she said. “Perhaps in time, we may be able to look at whether or not the individuals that participated in that program have fewer symptoms of OA disease. But that will obviously take us a few years before we’ll be able to get to that point.”
Dr. Whittaker acknowledged receiving funding for the SOAR program from the Arthritis Society, the Michael Smith Foundation for Health Research, BC SUPPORT Unit, and the Canadian Musculoskeletal Rehab Network.
FROM CARC 2021
Rise of JAK inhibitors highlights axial spondyloarthritis year in review
Rheumatologists can look forward to the likely regulatory approval of the oral Janus kinase inhibitors tofacitinib and upadacitinib for the treatment of axial spondyloarthritis in the first half of 2021, speakers predicted at the 2021 Rheumatology Winter Clinical Symposium.
This will be a major advance in the treatment of axial spondyloarthritis (axSpA) and promises to be one of the overall highlights of the coming year in rheumatology, according to the speakers. Both medications are now under Food and Drug Administration review for the proposed new indication.
“My sense is within the next 6 months we’re going to have two different oral JAK inhibitors that offer a new option for our ankylosing spondylitis and axial spondyloarthritis patients,” predicted Eric M. Ruderman, MD, professor of medicine (rheumatology) at Northwestern University, Chicago.
Alexis R. Ogdie, MD, MSCE, noted that, at present, only two classes of potent medications are available for treatment of axial spondyloarthritis: tumor necrosis factor (TNF) inhibitors and anti–interleukin-17 biologics.
“I think it would be so exciting to have more treatment options. To have only two classes of drugs you can use for this disease is not enough,” said Dr. Ogdie, a rheumatologist and epidemiologist at the University of Pennsylvania, Philadelphia.
She and her fellow panelists also highlighted other recent key developments in axSpA, including epidemiologic evidence that case numbers are climbing sharply, identification of two previously unrecognized common comorbidities, a successful biologic remission induction and maintenance dose–reduction strategy, data on the best and worst biologics for patients with anterior uveitis, and evidence regarding next-step therapy in axSpA patients who’ve had an inadequate response to a TNF inhibitor.
The JAK inhibitors are coming
Oral tofacitinib (Xeljanz) at 5 mg twice daily was the focus of a pivotal phase 3, double-blind, 16-week, placebo-controlled clinical trial including 269 patients with axSpA. The results were presented at the 2020 annual meeting of the American College of Rheumatology. The primary endpoint was at least a 20% improvement in Assessment of Spondyloarthritis International Society response criteria (ASAS 20). This was accomplished in 56.4% of patients on tofacitinib and 29.4% of placebo-treated controls. The ASAS 40 response rate was even more impressive: 40.6% with the JAK inhibitor, compared with 12.5% with placebo. There was one serious infection in the tofacitinib group, but no cases of venous thromboembolism, interstitial lung disease, opportunistic infection, major adverse cardiovascular events, or malignancy in this brief 4-month study.
Also now under FDA review are data from SELECT-AXIS 1, a phase 2/3, double-blind trial in which 187 biologic-naive patients with ankylosing spondylitis were randomized to 14 weeks of upadacitinib (Rinvoq) at 15 mg once daily or placebo. The primary endpoint, an ASAS 40 response, occurred in 51.6% of patients on the JAK inhibitor and half as many controls.
“They saw improvement in MRI scores with upadacitinib, so there’s biologic plausibility to this,” Dr. Ruderman noted.
He predicted the JAK inhibitors are going to have a big impact in clinical practice, especially in men.
“I have a lot of ankylosing spondylitis and axial spondyloarthritis patients on NSAIDs who I’m not convinced are doing as well as they could, but they push back every time I raise the possibility of going on a biologic,” the rheumatologist said. “I suspect that, given the rapid response with JAK inhibitors here, as in rheumatoid arthritis, it might be a little bit easier to persuade these people to give this a try for 4-6 weeks and then see how much better they are. It’s a pill. You don’t have to give yourself a shot.”
Dr. Ogdie predicted the new oral agents will bring more axSpA patients into rheumatologists’ offices.
“I think it’ll be kind of like the apremilast effect in psoriasis, where the drug got a lot more people into the market,” she said.
U.S. ankylosing spondylitis prevalence rising
The diagnostic prevalence of axSpA in the United States increased by 86% between 2006 and 2014 in a retrospective analysis based on Medicare fee-for-service claims data. A separate analysis using IBM MarketScan data for the same years was confirmatory, showing a 56% increase, Jeffrey R. Curtis, MD, MS, MPH, of the University of Alabama at Birmingham reported at the 2020 annual meeting of the European League Against Rheumatism (EULAR), now known as the European Alliance of Associations for Rheumatology.
“I think the take home is we’re seeing more of this. Some of this is likely due to increased awareness and inclusion of nonradiographic disease,” according to Dr. Ruderman.
Two previously overlooked comorbidities
It’s well recognized that 5%-10% of patients with axSpA have concurrent inflammatory bowel disease. But how about irritable bowel syndrome (IBS)?
A study of 186 Swedish patients with axSpA in the population-based SPARTAKUS registry, none with inflammatory bowel disease, concluded that 30% of them met ROME III diagnostic criteria for IBS, compared with 16% of healthy controls. Of note, the axSpA patients with comorbid IBS had significantly worse axSpA disease outcomes, compared with those without IBS as measured by pain, fatigue, and quality-of-life scores, as well as significantly greater disease activity on the Bath Ankylosing Spondylitis Disease Activity Index.
New-onset inflammatory back pain occurred in a hefty 24% of 513 Saudi patients placed on isotretinoin for acne. About 42% of those with inflammatory back pain displayed evidence of sacroiliitis on MRI. Moreover, 52% of patients with MRI-proven sacroiliitis fulfilled ASAS criteria for axSpA. In this longitudinal study, the MRI abnormalities and back pain symptoms completely resolved after isotretinoin discontinuation, but it took a long time: up to 9 months.
“When you see these people with inflammatory back pain on isotretinoin, I think it’s important that before you saddle them with a diagnosis that they have axSpA – a diagnosis that will go with them forever – give them time off drug, because this can look like the real thing. It’s something to think about as these pretty young kids come in to see you with back pain: Always ask about their medication history because it could be important,” Dr. Ruderman said.
A successful biologic remission induction-and-maintenance strategy
The phase 3b, multicenter C-OPTIMISE study sought to determine the best strategy for avoiding axSpA flares once sustained clinical remission has been achieved with a TNF inhibitor, in this case certolizumab pegol (Cimzia). The first part of the trial involved 736 patients with early axSpA, including 329 with nonradiographic disease. During the 48-week open-label induction period, 43.9% of patients achieved sustained clinical remission at the approved dose of 200 mg every 2 weeks, with similar success rates in radiographic and nonradiographic axSpA.
Those in sustained remission were then randomized double blind to one of three groups: an additional 48 weeks of certolizumab pegol at the full maintenance dose of 200 mg every 2 weeks, reduced maintenance dosing at 200 mg every 4 weeks, or placebo. During this period, 83.7% of the group who continued on full-dose certolizumab remained flare free, as did 79% of those on the reduced maintenance dose. In contrast, only 20.2% of patients in whom the biologic was completely withdrawn remained flare free. The investigators concluded that certolizumab dose reduction is a winning strategy for maintenance of clinical remission, as it reduces costs and limits long-term exposure to immunosuppressive therapy while maintaining clinical benefits.
All biologics aren’t equal when it comes to anterior uveitis risk
An analysis of Swedish Rheumatology Quality Register data presented at the 2020 EULAR meeting concluded that, among 3,568 patients started on one of four biologics for treatment of spondyloarthritis, the incidence of anterior uveitis was 2.9 per 100 patient-years in those on infliximab (Remicade), 4.0 per 100 patient-years with adalimumab (Humira), 6.8 per 100 patient-years with secukinumab (Cosentyx), and 7.5 per 100 patient-years with etanercept (Enbrel).
“This is important information for us in the clinic. The big question has been, do we see a reduced risk of anterior uveitis with secukinumab, an interleukin-17 inhibitor,” observed RWCS director Arthur Kavanaugh, MD, professor of medicine and director of the Center for Innovative Therapy in the division of rheumatology, allergy, and immunology at the University of California, San Diego.
“When I’ve switched people with uveitis to secukinumab or ixekizumab [Taltz], I do see it come back. So I think it’s important to have these data out there,” Dr. Ogdie said.
Certolizumab pegol markedly reduced the incidence of anterior uveitis flares in patients with radiographic and nonradiographic axSpA and a history of recurrent uveitis flares in the ongoing phase 4 C-VIEW study. In the 48 weeks prior to going on certolizumab pegol, the 89 participants included in this analysis had an acute anterior uveitis incidence of 1.5 episodes per patient; during their first 48 weeks on the TNF inhibitor, the rate plunged to 0.2 episodes, representing an 87% reduction.
Secukinumab ‘not the obvious choice’ after inadequate response to a TNF inhibitor
While it might seem logical to turn to an IL-17 inhibitor in patients with an inadequate response to one or more TNF inhibitors, two recently published studies suggest that starting secukinumab is not more effective than trying yet another TNF inhibitor.
A retrospective analysis of Swiss registry data on next-step therapy in 390 axSpA patients who had withdrawn from one or more TNF inhibitors concluded that efficacy at 1 year in the 106 who switched to secukinumab wasn’t significantly different than in the 284 who moved on to another TNF inhibitor.
Similarly, an analysis of 10,583 courses of biologic therapy in 8,050 axSpA patients in five Nordic registries concluded that secukinumab and adalimumab as second-line therapy in patients with inadequate response to an initial TNF inhibitor performed similarly through 1 year of follow-up. However, in patients who’d previously failed to respond to two or three different biologic agents, adalimumab proved superior to the interleukin-17 inhibitor.
“These are two studies that don’t support the intuitive notion of trying a drug with a different mechanism of action when a patient has an inadequate response to a TNF inhibitor. It’s not clear that’s going to make a difference. It doesn’t mean secukinumab can’t work, but it means secukinumab is not the obvious choice,” Dr. Ruderman commented.
All three speakers reported financial relationships with numerous pharmaceutical companies.
Rheumatologists can look forward to the likely regulatory approval of the oral Janus kinase inhibitors tofacitinib and upadacitinib for the treatment of axial spondyloarthritis in the first half of 2021, speakers predicted at the 2021 Rheumatology Winter Clinical Symposium.
This will be a major advance in the treatment of axial spondyloarthritis (axSpA) and promises to be one of the overall highlights of the coming year in rheumatology, according to the speakers. Both medications are now under Food and Drug Administration review for the proposed new indication.
“My sense is within the next 6 months we’re going to have two different oral JAK inhibitors that offer a new option for our ankylosing spondylitis and axial spondyloarthritis patients,” predicted Eric M. Ruderman, MD, professor of medicine (rheumatology) at Northwestern University, Chicago.
Alexis R. Ogdie, MD, MSCE, noted that, at present, only two classes of potent medications are available for treatment of axial spondyloarthritis: tumor necrosis factor (TNF) inhibitors and anti–interleukin-17 biologics.
“I think it would be so exciting to have more treatment options. To have only two classes of drugs you can use for this disease is not enough,” said Dr. Ogdie, a rheumatologist and epidemiologist at the University of Pennsylvania, Philadelphia.
She and her fellow panelists also highlighted other recent key developments in axSpA, including epidemiologic evidence that case numbers are climbing sharply, identification of two previously unrecognized common comorbidities, a successful biologic remission induction and maintenance dose–reduction strategy, data on the best and worst biologics for patients with anterior uveitis, and evidence regarding next-step therapy in axSpA patients who’ve had an inadequate response to a TNF inhibitor.
The JAK inhibitors are coming
Oral tofacitinib (Xeljanz) at 5 mg twice daily was the focus of a pivotal phase 3, double-blind, 16-week, placebo-controlled clinical trial including 269 patients with axSpA. The results were presented at the 2020 annual meeting of the American College of Rheumatology. The primary endpoint was at least a 20% improvement in Assessment of Spondyloarthritis International Society response criteria (ASAS 20). This was accomplished in 56.4% of patients on tofacitinib and 29.4% of placebo-treated controls. The ASAS 40 response rate was even more impressive: 40.6% with the JAK inhibitor, compared with 12.5% with placebo. There was one serious infection in the tofacitinib group, but no cases of venous thromboembolism, interstitial lung disease, opportunistic infection, major adverse cardiovascular events, or malignancy in this brief 4-month study.
Also now under FDA review are data from SELECT-AXIS 1, a phase 2/3, double-blind trial in which 187 biologic-naive patients with ankylosing spondylitis were randomized to 14 weeks of upadacitinib (Rinvoq) at 15 mg once daily or placebo. The primary endpoint, an ASAS 40 response, occurred in 51.6% of patients on the JAK inhibitor and half as many controls.
“They saw improvement in MRI scores with upadacitinib, so there’s biologic plausibility to this,” Dr. Ruderman noted.
He predicted the JAK inhibitors are going to have a big impact in clinical practice, especially in men.
“I have a lot of ankylosing spondylitis and axial spondyloarthritis patients on NSAIDs who I’m not convinced are doing as well as they could, but they push back every time I raise the possibility of going on a biologic,” the rheumatologist said. “I suspect that, given the rapid response with JAK inhibitors here, as in rheumatoid arthritis, it might be a little bit easier to persuade these people to give this a try for 4-6 weeks and then see how much better they are. It’s a pill. You don’t have to give yourself a shot.”
Dr. Ogdie predicted the new oral agents will bring more axSpA patients into rheumatologists’ offices.
“I think it’ll be kind of like the apremilast effect in psoriasis, where the drug got a lot more people into the market,” she said.
U.S. ankylosing spondylitis prevalence rising
The diagnostic prevalence of axSpA in the United States increased by 86% between 2006 and 2014 in a retrospective analysis based on Medicare fee-for-service claims data. A separate analysis using IBM MarketScan data for the same years was confirmatory, showing a 56% increase, Jeffrey R. Curtis, MD, MS, MPH, of the University of Alabama at Birmingham reported at the 2020 annual meeting of the European League Against Rheumatism (EULAR), now known as the European Alliance of Associations for Rheumatology.
“I think the take home is we’re seeing more of this. Some of this is likely due to increased awareness and inclusion of nonradiographic disease,” according to Dr. Ruderman.
Two previously overlooked comorbidities
It’s well recognized that 5%-10% of patients with axSpA have concurrent inflammatory bowel disease. But how about irritable bowel syndrome (IBS)?
A study of 186 Swedish patients with axSpA in the population-based SPARTAKUS registry, none with inflammatory bowel disease, concluded that 30% of them met ROME III diagnostic criteria for IBS, compared with 16% of healthy controls. Of note, the axSpA patients with comorbid IBS had significantly worse axSpA disease outcomes, compared with those without IBS as measured by pain, fatigue, and quality-of-life scores, as well as significantly greater disease activity on the Bath Ankylosing Spondylitis Disease Activity Index.
New-onset inflammatory back pain occurred in a hefty 24% of 513 Saudi patients placed on isotretinoin for acne. About 42% of those with inflammatory back pain displayed evidence of sacroiliitis on MRI. Moreover, 52% of patients with MRI-proven sacroiliitis fulfilled ASAS criteria for axSpA. In this longitudinal study, the MRI abnormalities and back pain symptoms completely resolved after isotretinoin discontinuation, but it took a long time: up to 9 months.
“When you see these people with inflammatory back pain on isotretinoin, I think it’s important that before you saddle them with a diagnosis that they have axSpA – a diagnosis that will go with them forever – give them time off drug, because this can look like the real thing. It’s something to think about as these pretty young kids come in to see you with back pain: Always ask about their medication history because it could be important,” Dr. Ruderman said.
A successful biologic remission induction-and-maintenance strategy
The phase 3b, multicenter C-OPTIMISE study sought to determine the best strategy for avoiding axSpA flares once sustained clinical remission has been achieved with a TNF inhibitor, in this case certolizumab pegol (Cimzia). The first part of the trial involved 736 patients with early axSpA, including 329 with nonradiographic disease. During the 48-week open-label induction period, 43.9% of patients achieved sustained clinical remission at the approved dose of 200 mg every 2 weeks, with similar success rates in radiographic and nonradiographic axSpA.
Those in sustained remission were then randomized double blind to one of three groups: an additional 48 weeks of certolizumab pegol at the full maintenance dose of 200 mg every 2 weeks, reduced maintenance dosing at 200 mg every 4 weeks, or placebo. During this period, 83.7% of the group who continued on full-dose certolizumab remained flare free, as did 79% of those on the reduced maintenance dose. In contrast, only 20.2% of patients in whom the biologic was completely withdrawn remained flare free. The investigators concluded that certolizumab dose reduction is a winning strategy for maintenance of clinical remission, as it reduces costs and limits long-term exposure to immunosuppressive therapy while maintaining clinical benefits.
All biologics aren’t equal when it comes to anterior uveitis risk
An analysis of Swedish Rheumatology Quality Register data presented at the 2020 EULAR meeting concluded that, among 3,568 patients started on one of four biologics for treatment of spondyloarthritis, the incidence of anterior uveitis was 2.9 per 100 patient-years in those on infliximab (Remicade), 4.0 per 100 patient-years with adalimumab (Humira), 6.8 per 100 patient-years with secukinumab (Cosentyx), and 7.5 per 100 patient-years with etanercept (Enbrel).
“This is important information for us in the clinic. The big question has been, do we see a reduced risk of anterior uveitis with secukinumab, an interleukin-17 inhibitor,” observed RWCS director Arthur Kavanaugh, MD, professor of medicine and director of the Center for Innovative Therapy in the division of rheumatology, allergy, and immunology at the University of California, San Diego.
“When I’ve switched people with uveitis to secukinumab or ixekizumab [Taltz], I do see it come back. So I think it’s important to have these data out there,” Dr. Ogdie said.
Certolizumab pegol markedly reduced the incidence of anterior uveitis flares in patients with radiographic and nonradiographic axSpA and a history of recurrent uveitis flares in the ongoing phase 4 C-VIEW study. In the 48 weeks prior to going on certolizumab pegol, the 89 participants included in this analysis had an acute anterior uveitis incidence of 1.5 episodes per patient; during their first 48 weeks on the TNF inhibitor, the rate plunged to 0.2 episodes, representing an 87% reduction.
Secukinumab ‘not the obvious choice’ after inadequate response to a TNF inhibitor
While it might seem logical to turn to an IL-17 inhibitor in patients with an inadequate response to one or more TNF inhibitors, two recently published studies suggest that starting secukinumab is not more effective than trying yet another TNF inhibitor.
A retrospective analysis of Swiss registry data on next-step therapy in 390 axSpA patients who had withdrawn from one or more TNF inhibitors concluded that efficacy at 1 year in the 106 who switched to secukinumab wasn’t significantly different than in the 284 who moved on to another TNF inhibitor.
Similarly, an analysis of 10,583 courses of biologic therapy in 8,050 axSpA patients in five Nordic registries concluded that secukinumab and adalimumab as second-line therapy in patients with inadequate response to an initial TNF inhibitor performed similarly through 1 year of follow-up. However, in patients who’d previously failed to respond to two or three different biologic agents, adalimumab proved superior to the interleukin-17 inhibitor.
“These are two studies that don’t support the intuitive notion of trying a drug with a different mechanism of action when a patient has an inadequate response to a TNF inhibitor. It’s not clear that’s going to make a difference. It doesn’t mean secukinumab can’t work, but it means secukinumab is not the obvious choice,” Dr. Ruderman commented.
All three speakers reported financial relationships with numerous pharmaceutical companies.
Rheumatologists can look forward to the likely regulatory approval of the oral Janus kinase inhibitors tofacitinib and upadacitinib for the treatment of axial spondyloarthritis in the first half of 2021, speakers predicted at the 2021 Rheumatology Winter Clinical Symposium.
This will be a major advance in the treatment of axial spondyloarthritis (axSpA) and promises to be one of the overall highlights of the coming year in rheumatology, according to the speakers. Both medications are now under Food and Drug Administration review for the proposed new indication.
“My sense is within the next 6 months we’re going to have two different oral JAK inhibitors that offer a new option for our ankylosing spondylitis and axial spondyloarthritis patients,” predicted Eric M. Ruderman, MD, professor of medicine (rheumatology) at Northwestern University, Chicago.
Alexis R. Ogdie, MD, MSCE, noted that, at present, only two classes of potent medications are available for treatment of axial spondyloarthritis: tumor necrosis factor (TNF) inhibitors and anti–interleukin-17 biologics.
“I think it would be so exciting to have more treatment options. To have only two classes of drugs you can use for this disease is not enough,” said Dr. Ogdie, a rheumatologist and epidemiologist at the University of Pennsylvania, Philadelphia.
She and her fellow panelists also highlighted other recent key developments in axSpA, including epidemiologic evidence that case numbers are climbing sharply, identification of two previously unrecognized common comorbidities, a successful biologic remission induction and maintenance dose–reduction strategy, data on the best and worst biologics for patients with anterior uveitis, and evidence regarding next-step therapy in axSpA patients who’ve had an inadequate response to a TNF inhibitor.
The JAK inhibitors are coming
Oral tofacitinib (Xeljanz) at 5 mg twice daily was the focus of a pivotal phase 3, double-blind, 16-week, placebo-controlled clinical trial including 269 patients with axSpA. The results were presented at the 2020 annual meeting of the American College of Rheumatology. The primary endpoint was at least a 20% improvement in Assessment of Spondyloarthritis International Society response criteria (ASAS 20). This was accomplished in 56.4% of patients on tofacitinib and 29.4% of placebo-treated controls. The ASAS 40 response rate was even more impressive: 40.6% with the JAK inhibitor, compared with 12.5% with placebo. There was one serious infection in the tofacitinib group, but no cases of venous thromboembolism, interstitial lung disease, opportunistic infection, major adverse cardiovascular events, or malignancy in this brief 4-month study.
Also now under FDA review are data from SELECT-AXIS 1, a phase 2/3, double-blind trial in which 187 biologic-naive patients with ankylosing spondylitis were randomized to 14 weeks of upadacitinib (Rinvoq) at 15 mg once daily or placebo. The primary endpoint, an ASAS 40 response, occurred in 51.6% of patients on the JAK inhibitor and half as many controls.
“They saw improvement in MRI scores with upadacitinib, so there’s biologic plausibility to this,” Dr. Ruderman noted.
He predicted the JAK inhibitors are going to have a big impact in clinical practice, especially in men.
“I have a lot of ankylosing spondylitis and axial spondyloarthritis patients on NSAIDs who I’m not convinced are doing as well as they could, but they push back every time I raise the possibility of going on a biologic,” the rheumatologist said. “I suspect that, given the rapid response with JAK inhibitors here, as in rheumatoid arthritis, it might be a little bit easier to persuade these people to give this a try for 4-6 weeks and then see how much better they are. It’s a pill. You don’t have to give yourself a shot.”
Dr. Ogdie predicted the new oral agents will bring more axSpA patients into rheumatologists’ offices.
“I think it’ll be kind of like the apremilast effect in psoriasis, where the drug got a lot more people into the market,” she said.
U.S. ankylosing spondylitis prevalence rising
The diagnostic prevalence of axSpA in the United States increased by 86% between 2006 and 2014 in a retrospective analysis based on Medicare fee-for-service claims data. A separate analysis using IBM MarketScan data for the same years was confirmatory, showing a 56% increase, Jeffrey R. Curtis, MD, MS, MPH, of the University of Alabama at Birmingham reported at the 2020 annual meeting of the European League Against Rheumatism (EULAR), now known as the European Alliance of Associations for Rheumatology.
“I think the take home is we’re seeing more of this. Some of this is likely due to increased awareness and inclusion of nonradiographic disease,” according to Dr. Ruderman.
Two previously overlooked comorbidities
It’s well recognized that 5%-10% of patients with axSpA have concurrent inflammatory bowel disease. But how about irritable bowel syndrome (IBS)?
A study of 186 Swedish patients with axSpA in the population-based SPARTAKUS registry, none with inflammatory bowel disease, concluded that 30% of them met ROME III diagnostic criteria for IBS, compared with 16% of healthy controls. Of note, the axSpA patients with comorbid IBS had significantly worse axSpA disease outcomes, compared with those without IBS as measured by pain, fatigue, and quality-of-life scores, as well as significantly greater disease activity on the Bath Ankylosing Spondylitis Disease Activity Index.
New-onset inflammatory back pain occurred in a hefty 24% of 513 Saudi patients placed on isotretinoin for acne. About 42% of those with inflammatory back pain displayed evidence of sacroiliitis on MRI. Moreover, 52% of patients with MRI-proven sacroiliitis fulfilled ASAS criteria for axSpA. In this longitudinal study, the MRI abnormalities and back pain symptoms completely resolved after isotretinoin discontinuation, but it took a long time: up to 9 months.
“When you see these people with inflammatory back pain on isotretinoin, I think it’s important that before you saddle them with a diagnosis that they have axSpA – a diagnosis that will go with them forever – give them time off drug, because this can look like the real thing. It’s something to think about as these pretty young kids come in to see you with back pain: Always ask about their medication history because it could be important,” Dr. Ruderman said.
A successful biologic remission induction-and-maintenance strategy
The phase 3b, multicenter C-OPTIMISE study sought to determine the best strategy for avoiding axSpA flares once sustained clinical remission has been achieved with a TNF inhibitor, in this case certolizumab pegol (Cimzia). The first part of the trial involved 736 patients with early axSpA, including 329 with nonradiographic disease. During the 48-week open-label induction period, 43.9% of patients achieved sustained clinical remission at the approved dose of 200 mg every 2 weeks, with similar success rates in radiographic and nonradiographic axSpA.
Those in sustained remission were then randomized double blind to one of three groups: an additional 48 weeks of certolizumab pegol at the full maintenance dose of 200 mg every 2 weeks, reduced maintenance dosing at 200 mg every 4 weeks, or placebo. During this period, 83.7% of the group who continued on full-dose certolizumab remained flare free, as did 79% of those on the reduced maintenance dose. In contrast, only 20.2% of patients in whom the biologic was completely withdrawn remained flare free. The investigators concluded that certolizumab dose reduction is a winning strategy for maintenance of clinical remission, as it reduces costs and limits long-term exposure to immunosuppressive therapy while maintaining clinical benefits.
All biologics aren’t equal when it comes to anterior uveitis risk
An analysis of Swedish Rheumatology Quality Register data presented at the 2020 EULAR meeting concluded that, among 3,568 patients started on one of four biologics for treatment of spondyloarthritis, the incidence of anterior uveitis was 2.9 per 100 patient-years in those on infliximab (Remicade), 4.0 per 100 patient-years with adalimumab (Humira), 6.8 per 100 patient-years with secukinumab (Cosentyx), and 7.5 per 100 patient-years with etanercept (Enbrel).
“This is important information for us in the clinic. The big question has been, do we see a reduced risk of anterior uveitis with secukinumab, an interleukin-17 inhibitor,” observed RWCS director Arthur Kavanaugh, MD, professor of medicine and director of the Center for Innovative Therapy in the division of rheumatology, allergy, and immunology at the University of California, San Diego.
“When I’ve switched people with uveitis to secukinumab or ixekizumab [Taltz], I do see it come back. So I think it’s important to have these data out there,” Dr. Ogdie said.
Certolizumab pegol markedly reduced the incidence of anterior uveitis flares in patients with radiographic and nonradiographic axSpA and a history of recurrent uveitis flares in the ongoing phase 4 C-VIEW study. In the 48 weeks prior to going on certolizumab pegol, the 89 participants included in this analysis had an acute anterior uveitis incidence of 1.5 episodes per patient; during their first 48 weeks on the TNF inhibitor, the rate plunged to 0.2 episodes, representing an 87% reduction.
Secukinumab ‘not the obvious choice’ after inadequate response to a TNF inhibitor
While it might seem logical to turn to an IL-17 inhibitor in patients with an inadequate response to one or more TNF inhibitors, two recently published studies suggest that starting secukinumab is not more effective than trying yet another TNF inhibitor.
A retrospective analysis of Swiss registry data on next-step therapy in 390 axSpA patients who had withdrawn from one or more TNF inhibitors concluded that efficacy at 1 year in the 106 who switched to secukinumab wasn’t significantly different than in the 284 who moved on to another TNF inhibitor.
Similarly, an analysis of 10,583 courses of biologic therapy in 8,050 axSpA patients in five Nordic registries concluded that secukinumab and adalimumab as second-line therapy in patients with inadequate response to an initial TNF inhibitor performed similarly through 1 year of follow-up. However, in patients who’d previously failed to respond to two or three different biologic agents, adalimumab proved superior to the interleukin-17 inhibitor.
“These are two studies that don’t support the intuitive notion of trying a drug with a different mechanism of action when a patient has an inadequate response to a TNF inhibitor. It’s not clear that’s going to make a difference. It doesn’t mean secukinumab can’t work, but it means secukinumab is not the obvious choice,” Dr. Ruderman commented.
All three speakers reported financial relationships with numerous pharmaceutical companies.
FROM RWCS 2021
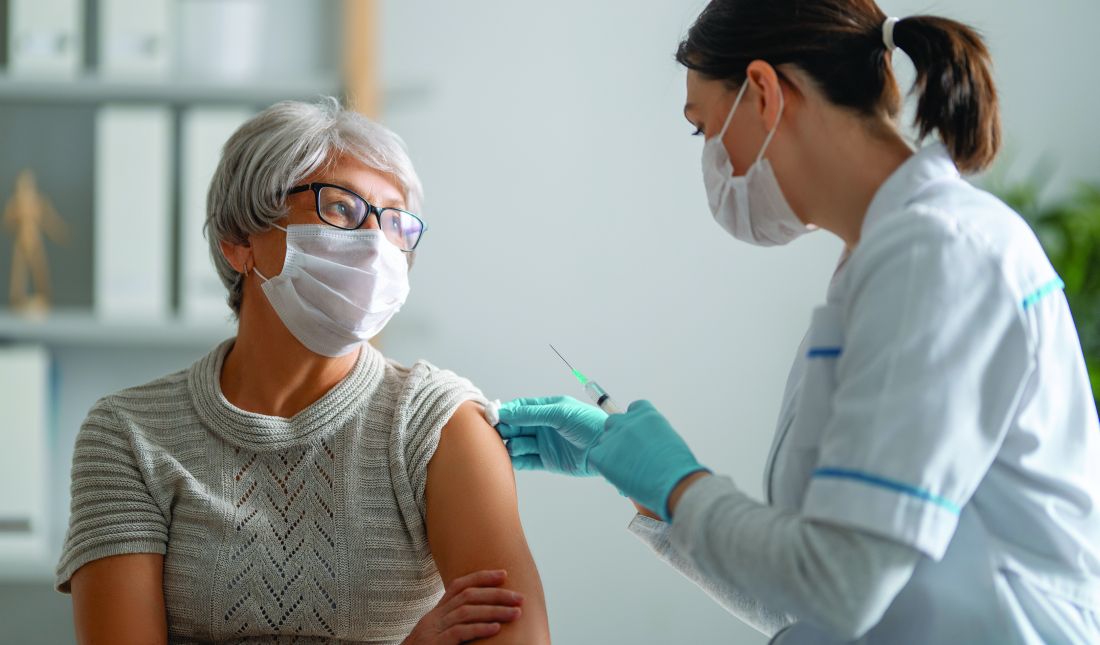
COVID-19 vaccination recommended for rheumatology patients
People with rheumatic diseases should get vaccinated against SARS-CoV-2 as soon as possible, the American College of Rheumatology (ACR) recommends.
“It may be that people with rheumatic diseases are at increased risk of developing COVID or serious COVID-related complications,” Jonathan Hausmann, MD, assistant professor of medicine at Harvard Medical School, Boston, said in an ACR podcast. “So the need to prevent COVID-19 is incredibly important in this group of patients.”
The guidelines recommend a delay in vaccination only in rare circumstances, such as for patients with very severe illness or who have recently been administered rituximab, Jeffrey R. Curtis, MD, MPH, lead author of the guidelines, said in the podcast.
“Our members have been inundated with questions and concerns from their patients on whether they should receive the vaccine,” ACR President David Karp, MD, PhD, said in a press release.
So the ACR convened a panel of nine rheumatologists, two infectious disease specialists, and two public health experts. Over the course of 8 weeks, the task force reviewed the literature and agreed on recommendations. The organization posted a summary of the guidelines on its website after its board of directors approved it Feb. 8. The paper is pending journal peer review.
Some risks are real
The task force confined its research to the COVID-19 vaccines being offered by Pfizer and Moderna because they are currently the only ones approved by the Food and Drug Administration. It found no reason to distinguish between the two vaccines in its recommendations.
Because little research has directly addressed the question concerning COVID-19 vaccination for patients with rheumatic diseases, the task force extrapolated from data on other vaccinations in people with rheumatic disease and on the COVID-19 vaccinations in other populations.
It analyzed reports that other types of vaccination, such as for influenza, triggered flares of rheumatic conditions. “It is really individual case reports or small cohorts where there may be a somewhat higher incidence of flare, but it’s usually not very large in its magnitude nor duration,” said Dr. Curtis of the University of Alabama at Birmingham.
The task force also considered the possibility that vaccinations could lead to a new autoimmune disorder, such as Guillain-Barré syndrome or Bell palsy. The risk is real, the task force decided, but not significant enough to influence their recommendations.
Likewise, in immunocompromised people, vaccinations with live virus, such as those for shingles, might trigger the infection the vaccination is meant to prevent. But this can’t happen with the Pfizer and Moderna COVID-19 vaccines because they contain messenger RNA instead of live viruses, Dr. Curtis said.
Although it might be optimal to administer the vaccines when rheumatic diseases are quiescent, the urgency of getting vaccinated overrides that consideration, Dr. Curtis said. “By and large, there was a general consensus to not want to delay vaccination until somebody was stable and doing great, because you don’t know how long that’s going to be,” he said.
How well does it work?
One unanswered question is whether the COVID-19 vaccines work as well for patients with rheumatic diseases. The task force was reassured by data showing efficacy across a range of subgroups, including some with immunosenescence, Dr. Curtis said. “But until we have data in rheumatology patients, we’re just not going to know,” he said.
The guidelines specify that some drug regimens be modified when patients are vaccinated.
For patients taking rituximab, vaccination should be delayed, but only for those who are able to maintain safe social distancing to reduce the risk for COVID-19 exposure, Dr. Curtis said. “If somebody has just gotten rituximab recently, it might be more ideal to complete the vaccine series about 2-4 weeks before the next rituximab dose,” he said. “So if you are giving that therapy, say, at 6-month intervals, if you could vaccinate them at around month 5 from the most recent rituximab cycle, that might be more ideal.”
The guidance calls for withholding JAK inhibitors for a week after each vaccine dose is administered.
It calls for holding SQ abatacept 1 week prior and 1 week after the first COVID-19 vaccine dose, with no interruption after the second dose.
For abatacept IV, clinicians should “time vaccine administration so that the first vaccination will occur 4 weeks after abatacept infusion (i.e., the entire dosing interval), and postpone the subsequent abatacept infusion by 1 week (i.e., a 5-week gap in total).” It recommends no medication adjustment for the second vaccine dose.
For cyclophosphamide, the guidance recommends timing administration to occur about a week after each vaccine dose, when feasible.
None of this advice should supersede clinical judgment, Dr. Curtis said.
A version of this article first appeared on Medscape.com.
People with rheumatic diseases should get vaccinated against SARS-CoV-2 as soon as possible, the American College of Rheumatology (ACR) recommends.
“It may be that people with rheumatic diseases are at increased risk of developing COVID or serious COVID-related complications,” Jonathan Hausmann, MD, assistant professor of medicine at Harvard Medical School, Boston, said in an ACR podcast. “So the need to prevent COVID-19 is incredibly important in this group of patients.”
The guidelines recommend a delay in vaccination only in rare circumstances, such as for patients with very severe illness or who have recently been administered rituximab, Jeffrey R. Curtis, MD, MPH, lead author of the guidelines, said in the podcast.
“Our members have been inundated with questions and concerns from their patients on whether they should receive the vaccine,” ACR President David Karp, MD, PhD, said in a press release.
So the ACR convened a panel of nine rheumatologists, two infectious disease specialists, and two public health experts. Over the course of 8 weeks, the task force reviewed the literature and agreed on recommendations. The organization posted a summary of the guidelines on its website after its board of directors approved it Feb. 8. The paper is pending journal peer review.
Some risks are real
The task force confined its research to the COVID-19 vaccines being offered by Pfizer and Moderna because they are currently the only ones approved by the Food and Drug Administration. It found no reason to distinguish between the two vaccines in its recommendations.
Because little research has directly addressed the question concerning COVID-19 vaccination for patients with rheumatic diseases, the task force extrapolated from data on other vaccinations in people with rheumatic disease and on the COVID-19 vaccinations in other populations.
It analyzed reports that other types of vaccination, such as for influenza, triggered flares of rheumatic conditions. “It is really individual case reports or small cohorts where there may be a somewhat higher incidence of flare, but it’s usually not very large in its magnitude nor duration,” said Dr. Curtis of the University of Alabama at Birmingham.
The task force also considered the possibility that vaccinations could lead to a new autoimmune disorder, such as Guillain-Barré syndrome or Bell palsy. The risk is real, the task force decided, but not significant enough to influence their recommendations.
Likewise, in immunocompromised people, vaccinations with live virus, such as those for shingles, might trigger the infection the vaccination is meant to prevent. But this can’t happen with the Pfizer and Moderna COVID-19 vaccines because they contain messenger RNA instead of live viruses, Dr. Curtis said.
Although it might be optimal to administer the vaccines when rheumatic diseases are quiescent, the urgency of getting vaccinated overrides that consideration, Dr. Curtis said. “By and large, there was a general consensus to not want to delay vaccination until somebody was stable and doing great, because you don’t know how long that’s going to be,” he said.
How well does it work?
One unanswered question is whether the COVID-19 vaccines work as well for patients with rheumatic diseases. The task force was reassured by data showing efficacy across a range of subgroups, including some with immunosenescence, Dr. Curtis said. “But until we have data in rheumatology patients, we’re just not going to know,” he said.
The guidelines specify that some drug regimens be modified when patients are vaccinated.
For patients taking rituximab, vaccination should be delayed, but only for those who are able to maintain safe social distancing to reduce the risk for COVID-19 exposure, Dr. Curtis said. “If somebody has just gotten rituximab recently, it might be more ideal to complete the vaccine series about 2-4 weeks before the next rituximab dose,” he said. “So if you are giving that therapy, say, at 6-month intervals, if you could vaccinate them at around month 5 from the most recent rituximab cycle, that might be more ideal.”
The guidance calls for withholding JAK inhibitors for a week after each vaccine dose is administered.
It calls for holding SQ abatacept 1 week prior and 1 week after the first COVID-19 vaccine dose, with no interruption after the second dose.
For abatacept IV, clinicians should “time vaccine administration so that the first vaccination will occur 4 weeks after abatacept infusion (i.e., the entire dosing interval), and postpone the subsequent abatacept infusion by 1 week (i.e., a 5-week gap in total).” It recommends no medication adjustment for the second vaccine dose.
For cyclophosphamide, the guidance recommends timing administration to occur about a week after each vaccine dose, when feasible.
None of this advice should supersede clinical judgment, Dr. Curtis said.
A version of this article first appeared on Medscape.com.
People with rheumatic diseases should get vaccinated against SARS-CoV-2 as soon as possible, the American College of Rheumatology (ACR) recommends.
“It may be that people with rheumatic diseases are at increased risk of developing COVID or serious COVID-related complications,” Jonathan Hausmann, MD, assistant professor of medicine at Harvard Medical School, Boston, said in an ACR podcast. “So the need to prevent COVID-19 is incredibly important in this group of patients.”
The guidelines recommend a delay in vaccination only in rare circumstances, such as for patients with very severe illness or who have recently been administered rituximab, Jeffrey R. Curtis, MD, MPH, lead author of the guidelines, said in the podcast.
“Our members have been inundated with questions and concerns from their patients on whether they should receive the vaccine,” ACR President David Karp, MD, PhD, said in a press release.
So the ACR convened a panel of nine rheumatologists, two infectious disease specialists, and two public health experts. Over the course of 8 weeks, the task force reviewed the literature and agreed on recommendations. The organization posted a summary of the guidelines on its website after its board of directors approved it Feb. 8. The paper is pending journal peer review.
Some risks are real
The task force confined its research to the COVID-19 vaccines being offered by Pfizer and Moderna because they are currently the only ones approved by the Food and Drug Administration. It found no reason to distinguish between the two vaccines in its recommendations.
Because little research has directly addressed the question concerning COVID-19 vaccination for patients with rheumatic diseases, the task force extrapolated from data on other vaccinations in people with rheumatic disease and on the COVID-19 vaccinations in other populations.
It analyzed reports that other types of vaccination, such as for influenza, triggered flares of rheumatic conditions. “It is really individual case reports or small cohorts where there may be a somewhat higher incidence of flare, but it’s usually not very large in its magnitude nor duration,” said Dr. Curtis of the University of Alabama at Birmingham.
The task force also considered the possibility that vaccinations could lead to a new autoimmune disorder, such as Guillain-Barré syndrome or Bell palsy. The risk is real, the task force decided, but not significant enough to influence their recommendations.
Likewise, in immunocompromised people, vaccinations with live virus, such as those for shingles, might trigger the infection the vaccination is meant to prevent. But this can’t happen with the Pfizer and Moderna COVID-19 vaccines because they contain messenger RNA instead of live viruses, Dr. Curtis said.
Although it might be optimal to administer the vaccines when rheumatic diseases are quiescent, the urgency of getting vaccinated overrides that consideration, Dr. Curtis said. “By and large, there was a general consensus to not want to delay vaccination until somebody was stable and doing great, because you don’t know how long that’s going to be,” he said.
How well does it work?
One unanswered question is whether the COVID-19 vaccines work as well for patients with rheumatic diseases. The task force was reassured by data showing efficacy across a range of subgroups, including some with immunosenescence, Dr. Curtis said. “But until we have data in rheumatology patients, we’re just not going to know,” he said.
The guidelines specify that some drug regimens be modified when patients are vaccinated.
For patients taking rituximab, vaccination should be delayed, but only for those who are able to maintain safe social distancing to reduce the risk for COVID-19 exposure, Dr. Curtis said. “If somebody has just gotten rituximab recently, it might be more ideal to complete the vaccine series about 2-4 weeks before the next rituximab dose,” he said. “So if you are giving that therapy, say, at 6-month intervals, if you could vaccinate them at around month 5 from the most recent rituximab cycle, that might be more ideal.”
The guidance calls for withholding JAK inhibitors for a week after each vaccine dose is administered.
It calls for holding SQ abatacept 1 week prior and 1 week after the first COVID-19 vaccine dose, with no interruption after the second dose.
For abatacept IV, clinicians should “time vaccine administration so that the first vaccination will occur 4 weeks after abatacept infusion (i.e., the entire dosing interval), and postpone the subsequent abatacept infusion by 1 week (i.e., a 5-week gap in total).” It recommends no medication adjustment for the second vaccine dose.
For cyclophosphamide, the guidance recommends timing administration to occur about a week after each vaccine dose, when feasible.
None of this advice should supersede clinical judgment, Dr. Curtis said.
A version of this article first appeared on Medscape.com.
Pandemic puts patients with psoriatic disease off seeking medical help
More than half of respondents to a recent survey looking at how the COVID-19 pandemic has affected people with psoriasis or psoriatic arthritis (PsA) said that they had avoided seeking medical care in person with a doctor or at a hospital.
Moreover, around a quarter had their appointment with a rheumatologist canceled, rescheduled, or conducted virtually. Another 1 in 10 had their treatment plan disrupted, and 6% had to change or stop treatment entirely.
The mental health impact of living with these conditions during the pandemic was also notable, said Rachael Manion, the executive director of the Canadian Association of Psoriasis Patients (CAPP), which conducted the survey in collaboration with the Canadian Psoriasis Network (CPN) and Unmasking Psoriasis.
“It’s important to know that there have been a lot of different impacts of the pandemic on people living with psoriatic arthritis and psoriasis. Mental health in particular has had a really big hit as a result,” she said at the Canadian Arthritis Research Conference: Research with Impact.
“About half of the people who responded to our survey noted that their mental health was ‘worse’ or ‘much worse’ during the pandemic,” she said at the meeting, which was sponsored by the Arthritis Society, the Canadian Rheumatology Association, and Canada’s Institute of Musculoskeletal Health and Arthritis. Anxiety and feelings of isolation were reported by a respective 57% and 58% of respondents, and 40% reported depression.
“We can compare that to our earlier information around depression,” Ms. Manion said, which showed that, prior to the pandemic, 24% of people with psoriasis and 23% of those with PsA had said they experienced depression.
“What I found alarming looking at these results was that about a third of people were experiencing despair. Now that’s a really big, scary, overwhelming emotion that has a lot of burden on your mental health,” Ms. Manion said.
Despite the substantial effects on mental health, only 29% of respondents said they had been able to access mental health services during the pandemic.
To look at the impact of the COVID-19 pandemic on the psoriasis and PsA community in Canada, three patient advocacy groups – CAPP, CPN, and Unmasking Psoriasis – codeveloped a survey to look at the disease experience before and after the start of the COVID-19 pandemic. The survey was performed once, with 830 respondents providing information on their lives with psoriasis or PsA in the months before the start of the pandemic and at the time they were surveyed in September and October 2020.
Most of the survey respondents lived in Ontario, Quebec, British Columbia, or Alberta, although other provinces or territories were represented. Almost all respondents (96%) had psoriasis, and 60% also had PsA.
Pre-COVID, nearly half (49%) of patients said that they had not been seen by a rheumatologist, and 39% had not seen a dermatologist for treatment. Asked why, 56% and 27%, respectively, had not been referred, 9% and 15% said they had no specialist located nearby, and 7% and 10% stated that the wait list was too long.
“This tells us that there’s a lot more work that can be done and a lot more education of general practitioners and family medicine professionals about the benefits and the value of specialized care for psoriatic arthritis,” Ms. Manion suggested.
Before the pandemic, joint pain was occurring in 88% of patients, stiffness in 71%, and joint swelling in 67%. Disease flares or sudden periods of worsening occurred on a daily basis for 17%, and around one in five (21%) experienced multiple flares every month.
Prepandemic data also highlighted the negative impact that living with psoriasis or PsA has on people’s ability to sleep, interactions and intimacy with others, and on their school or work lives.
During the pandemic, around a quarter (26%) of respondents said they had worse or much worse access to employment, as well as its benefits such as a stable income (24%). A minority of respondent also described worse access to prescription medication (15%) and over-the-counter medication (13%).
“There are all kinds of things going on for patients in our community: changes to their work, changes to their drug coverage, their ability to sleep and sleep well, their mental health, and their ability to access care and treatments as part of their disease management,” Ms. Manion said.
Her final message to health care professionals was: “I just want to encourage you to continue to check in with your patients about what their experiences have been during the pandemic, and to really consider those impacts as you’re working with them to manage their disease.”
The survey received funding support from AbbVie, Bausch Health, Boehringer Ingelheim, Janssen, LEO Pharma, and Novartis.
More than half of respondents to a recent survey looking at how the COVID-19 pandemic has affected people with psoriasis or psoriatic arthritis (PsA) said that they had avoided seeking medical care in person with a doctor or at a hospital.
Moreover, around a quarter had their appointment with a rheumatologist canceled, rescheduled, or conducted virtually. Another 1 in 10 had their treatment plan disrupted, and 6% had to change or stop treatment entirely.
The mental health impact of living with these conditions during the pandemic was also notable, said Rachael Manion, the executive director of the Canadian Association of Psoriasis Patients (CAPP), which conducted the survey in collaboration with the Canadian Psoriasis Network (CPN) and Unmasking Psoriasis.
“It’s important to know that there have been a lot of different impacts of the pandemic on people living with psoriatic arthritis and psoriasis. Mental health in particular has had a really big hit as a result,” she said at the Canadian Arthritis Research Conference: Research with Impact.
“About half of the people who responded to our survey noted that their mental health was ‘worse’ or ‘much worse’ during the pandemic,” she said at the meeting, which was sponsored by the Arthritis Society, the Canadian Rheumatology Association, and Canada’s Institute of Musculoskeletal Health and Arthritis. Anxiety and feelings of isolation were reported by a respective 57% and 58% of respondents, and 40% reported depression.
“We can compare that to our earlier information around depression,” Ms. Manion said, which showed that, prior to the pandemic, 24% of people with psoriasis and 23% of those with PsA had said they experienced depression.
“What I found alarming looking at these results was that about a third of people were experiencing despair. Now that’s a really big, scary, overwhelming emotion that has a lot of burden on your mental health,” Ms. Manion said.
Despite the substantial effects on mental health, only 29% of respondents said they had been able to access mental health services during the pandemic.
To look at the impact of the COVID-19 pandemic on the psoriasis and PsA community in Canada, three patient advocacy groups – CAPP, CPN, and Unmasking Psoriasis – codeveloped a survey to look at the disease experience before and after the start of the COVID-19 pandemic. The survey was performed once, with 830 respondents providing information on their lives with psoriasis or PsA in the months before the start of the pandemic and at the time they were surveyed in September and October 2020.
Most of the survey respondents lived in Ontario, Quebec, British Columbia, or Alberta, although other provinces or territories were represented. Almost all respondents (96%) had psoriasis, and 60% also had PsA.
Pre-COVID, nearly half (49%) of patients said that they had not been seen by a rheumatologist, and 39% had not seen a dermatologist for treatment. Asked why, 56% and 27%, respectively, had not been referred, 9% and 15% said they had no specialist located nearby, and 7% and 10% stated that the wait list was too long.
“This tells us that there’s a lot more work that can be done and a lot more education of general practitioners and family medicine professionals about the benefits and the value of specialized care for psoriatic arthritis,” Ms. Manion suggested.
Before the pandemic, joint pain was occurring in 88% of patients, stiffness in 71%, and joint swelling in 67%. Disease flares or sudden periods of worsening occurred on a daily basis for 17%, and around one in five (21%) experienced multiple flares every month.
Prepandemic data also highlighted the negative impact that living with psoriasis or PsA has on people’s ability to sleep, interactions and intimacy with others, and on their school or work lives.
During the pandemic, around a quarter (26%) of respondents said they had worse or much worse access to employment, as well as its benefits such as a stable income (24%). A minority of respondent also described worse access to prescription medication (15%) and over-the-counter medication (13%).
“There are all kinds of things going on for patients in our community: changes to their work, changes to their drug coverage, their ability to sleep and sleep well, their mental health, and their ability to access care and treatments as part of their disease management,” Ms. Manion said.
Her final message to health care professionals was: “I just want to encourage you to continue to check in with your patients about what their experiences have been during the pandemic, and to really consider those impacts as you’re working with them to manage their disease.”
The survey received funding support from AbbVie, Bausch Health, Boehringer Ingelheim, Janssen, LEO Pharma, and Novartis.
More than half of respondents to a recent survey looking at how the COVID-19 pandemic has affected people with psoriasis or psoriatic arthritis (PsA) said that they had avoided seeking medical care in person with a doctor or at a hospital.
Moreover, around a quarter had their appointment with a rheumatologist canceled, rescheduled, or conducted virtually. Another 1 in 10 had their treatment plan disrupted, and 6% had to change or stop treatment entirely.
The mental health impact of living with these conditions during the pandemic was also notable, said Rachael Manion, the executive director of the Canadian Association of Psoriasis Patients (CAPP), which conducted the survey in collaboration with the Canadian Psoriasis Network (CPN) and Unmasking Psoriasis.
“It’s important to know that there have been a lot of different impacts of the pandemic on people living with psoriatic arthritis and psoriasis. Mental health in particular has had a really big hit as a result,” she said at the Canadian Arthritis Research Conference: Research with Impact.
“About half of the people who responded to our survey noted that their mental health was ‘worse’ or ‘much worse’ during the pandemic,” she said at the meeting, which was sponsored by the Arthritis Society, the Canadian Rheumatology Association, and Canada’s Institute of Musculoskeletal Health and Arthritis. Anxiety and feelings of isolation were reported by a respective 57% and 58% of respondents, and 40% reported depression.
“We can compare that to our earlier information around depression,” Ms. Manion said, which showed that, prior to the pandemic, 24% of people with psoriasis and 23% of those with PsA had said they experienced depression.
“What I found alarming looking at these results was that about a third of people were experiencing despair. Now that’s a really big, scary, overwhelming emotion that has a lot of burden on your mental health,” Ms. Manion said.
Despite the substantial effects on mental health, only 29% of respondents said they had been able to access mental health services during the pandemic.
To look at the impact of the COVID-19 pandemic on the psoriasis and PsA community in Canada, three patient advocacy groups – CAPP, CPN, and Unmasking Psoriasis – codeveloped a survey to look at the disease experience before and after the start of the COVID-19 pandemic. The survey was performed once, with 830 respondents providing information on their lives with psoriasis or PsA in the months before the start of the pandemic and at the time they were surveyed in September and October 2020.
Most of the survey respondents lived in Ontario, Quebec, British Columbia, or Alberta, although other provinces or territories were represented. Almost all respondents (96%) had psoriasis, and 60% also had PsA.
Pre-COVID, nearly half (49%) of patients said that they had not been seen by a rheumatologist, and 39% had not seen a dermatologist for treatment. Asked why, 56% and 27%, respectively, had not been referred, 9% and 15% said they had no specialist located nearby, and 7% and 10% stated that the wait list was too long.
“This tells us that there’s a lot more work that can be done and a lot more education of general practitioners and family medicine professionals about the benefits and the value of specialized care for psoriatic arthritis,” Ms. Manion suggested.
Before the pandemic, joint pain was occurring in 88% of patients, stiffness in 71%, and joint swelling in 67%. Disease flares or sudden periods of worsening occurred on a daily basis for 17%, and around one in five (21%) experienced multiple flares every month.
Prepandemic data also highlighted the negative impact that living with psoriasis or PsA has on people’s ability to sleep, interactions and intimacy with others, and on their school or work lives.
During the pandemic, around a quarter (26%) of respondents said they had worse or much worse access to employment, as well as its benefits such as a stable income (24%). A minority of respondent also described worse access to prescription medication (15%) and over-the-counter medication (13%).
“There are all kinds of things going on for patients in our community: changes to their work, changes to their drug coverage, their ability to sleep and sleep well, their mental health, and their ability to access care and treatments as part of their disease management,” Ms. Manion said.
Her final message to health care professionals was: “I just want to encourage you to continue to check in with your patients about what their experiences have been during the pandemic, and to really consider those impacts as you’re working with them to manage their disease.”
The survey received funding support from AbbVie, Bausch Health, Boehringer Ingelheim, Janssen, LEO Pharma, and Novartis.
FROM CARC 2021
Frozen sections can guide biopsies for giant cell arteritis, but are they feasible?
Positive findings from frozen sections of a first temporal artery biopsy can effectively identify giant cell arteritis, ruling out in those cases the need to perform a second biopsy on the contralateral side and arguing against the use of simultaneous bilateral biopsies, according to results from a retrospective study of nearly 800 patients who underwent the procedure at the Mayo Clinic during 2010-2018.
Although temporal artery biopsy (TAB) remains the standard diagnostic test for giant cell arteritis (GCA), second TAB procedures are often performed in patients with a high level of suspicion for GCA, which may result in unnecessary treatments and complications, Devon A. Cohen, MD, of the Mayo Clinic, Rochester, Minn., and colleagues wrote. (Dr. Cohen is now a clinical fellow in ophthalmology at the Massachusetts Eye and Ear Infirmary.)
At the Mayo Clinic, TAB specimens are first examined with frozen sections at the time of the biopsy; this process, followed within days by formalin-fixed tissue permanent sections, is unique to Mayo. “A frozen section–guided sequential TAB is commonly performed, with the results of the first biopsy obtained within minutes, which determines the need for evaluation of the contralateral side,” the researchers said. However, the use of frozen sections to evaluate patients with GCA has not been well studied.
In a retrospective cohort study published in JAMA Ophthalmology, the researchers identified TAB patients aged 40 years and older who underwent TAB procedures between Jan. 1, 2010, and Dec. 1, 2018, at the Mayo Clinic. The average age of the patients was 72 years, and 41% were men.
Strong positive predictions from frozen sections
The researchers analyzed 1,162 TABs from 795 patients using frozen and permanent histologic sections.
Overall, 119 patients (15.0%) and 138 TABs had positive permanent section findings, and 103 (86.6%) of these patients also had positive frozen section findings, including 4 false positives and 20 false negatives. The frozen section specificity and sensitivity was 99.4% and 83.2%, respectively, for detecting inflammation suggestive of GCA, and the positive and negative predictive values were 96.1% and 96.6%, respectively. Positive and negative likelihood ratios for frozen section were 140.6 and 0.17, respectively.
In a multivariate analysis, the odds of a positive permanent section TAB significantly increased with age (odds ratio, 1.04), vision loss (OR, 2.72), diplopia (OR, 3.33), headache (OR, 2.32), weight loss (OR, 2.37), and anorexia (OR, 5.65).
A total of 60 patients underwent bilateral TABs, and 307 patients underwent bilateral frozen section–guided sequential TABs; the discordance rates based on permanent sections were 5.0% and 5.5%, respectively.
Those discordance rates are “an important result applying to everyone working with patients suspected for GCA,” Patricia Chévez-Barrios, MD, of Houston Methodist Hospital, wrote in an accompanying editorial. “This is on the low end of what was previously published (3%-40%) and supports the relative low need for bilateral synchronous TAB for the diagnosis of GCA.”
A key issue in GCA diagnosis is the need to confirm inflammation, Dr. Chévez-Barrios said. “The surgeon must obtain a significant portion of the artery, and the pathologist should review several sections and levels of the tissue to confidently say whether there is inflammation or no.”
Frozen sections can spare patients from second procedures
The findings suggest a role for frozen section to help to determine whether a unilateral or bilateral simultaneous TAB should be performed, the study authors noted.
“If the frozen section is positive on the first TAB, a contralateral TAB is deferred, given the very low false-positive rate (0.6%). However, if the frozen section does not align with the permanent section result, in particular if the frozen section is positive but permanent section is negative, the patient returns for a TAB on the contralateral side if the GCA suspicion remains high,” they said.
The use of frozen sections requires ideal conditions in order to be effective, Dr. Chévez-Barrios said. The Mayo Clinic approach “is only possible because of their appropriate hospital setting, the training of the histotechnologists, and the experience of the pathologists interpreting the stains and sections. For most pathology laboratories outside of the Mayo Clinic, frozen sections on arteries are the exception and are used only in specific scenarios.”
In addition, the American College of Rheumatology recommends that patients with a high suspicion of GCA should begin corticosteroids as soon as laboratory studies are obtained; “As a result, if a TAB is performed after treatment begins, the typical active pattern of inflammation in the artery changes,” Dr. Chévez-Barrios said. “This further challenges the diagnosis in a frozen section setting because of the need for immunohistochemistry.” Although frozen sections are feasible in specialized settings such as the Mayo Clinic, most patients receive adequate diagnosis and treatment based on permanent sections.
The study findings were limited by several factors including the use of data from patients at a single center and the unique setup of the Mayo Clinic to perform rapid processing of frozen sections, the researchers noted.
“Additionally, we acknowledge that there is controversy regarding the clinical interpretation of healed arteritis. At our institution, healed arteritis is interpreted in the context of patient clinical characteristics and radiographic findings, which may differ from other institutions and may impact the results of this study,” they said.
Overall, the results support the potential of frozen sections in guiding TAB, although “more studies with a comparative analysis of laboratory results, clinical symptoms, and patient demographic characteristics between positive and negative frozen and permanent TAB results are needed to confirm our findings,” they concluded.
The study received no outside funding. One author reported receiving grants from Eli Lilly and Kiniksa Pharmaceuticals as well as personal fees from Genentech-Roche and Sanofi. Dr. Chévez-Barrios had no financial conflicts to disclose.
Positive findings from frozen sections of a first temporal artery biopsy can effectively identify giant cell arteritis, ruling out in those cases the need to perform a second biopsy on the contralateral side and arguing against the use of simultaneous bilateral biopsies, according to results from a retrospective study of nearly 800 patients who underwent the procedure at the Mayo Clinic during 2010-2018.
Although temporal artery biopsy (TAB) remains the standard diagnostic test for giant cell arteritis (GCA), second TAB procedures are often performed in patients with a high level of suspicion for GCA, which may result in unnecessary treatments and complications, Devon A. Cohen, MD, of the Mayo Clinic, Rochester, Minn., and colleagues wrote. (Dr. Cohen is now a clinical fellow in ophthalmology at the Massachusetts Eye and Ear Infirmary.)
At the Mayo Clinic, TAB specimens are first examined with frozen sections at the time of the biopsy; this process, followed within days by formalin-fixed tissue permanent sections, is unique to Mayo. “A frozen section–guided sequential TAB is commonly performed, with the results of the first biopsy obtained within minutes, which determines the need for evaluation of the contralateral side,” the researchers said. However, the use of frozen sections to evaluate patients with GCA has not been well studied.
In a retrospective cohort study published in JAMA Ophthalmology, the researchers identified TAB patients aged 40 years and older who underwent TAB procedures between Jan. 1, 2010, and Dec. 1, 2018, at the Mayo Clinic. The average age of the patients was 72 years, and 41% were men.
Strong positive predictions from frozen sections
The researchers analyzed 1,162 TABs from 795 patients using frozen and permanent histologic sections.
Overall, 119 patients (15.0%) and 138 TABs had positive permanent section findings, and 103 (86.6%) of these patients also had positive frozen section findings, including 4 false positives and 20 false negatives. The frozen section specificity and sensitivity was 99.4% and 83.2%, respectively, for detecting inflammation suggestive of GCA, and the positive and negative predictive values were 96.1% and 96.6%, respectively. Positive and negative likelihood ratios for frozen section were 140.6 and 0.17, respectively.
In a multivariate analysis, the odds of a positive permanent section TAB significantly increased with age (odds ratio, 1.04), vision loss (OR, 2.72), diplopia (OR, 3.33), headache (OR, 2.32), weight loss (OR, 2.37), and anorexia (OR, 5.65).
A total of 60 patients underwent bilateral TABs, and 307 patients underwent bilateral frozen section–guided sequential TABs; the discordance rates based on permanent sections were 5.0% and 5.5%, respectively.
Those discordance rates are “an important result applying to everyone working with patients suspected for GCA,” Patricia Chévez-Barrios, MD, of Houston Methodist Hospital, wrote in an accompanying editorial. “This is on the low end of what was previously published (3%-40%) and supports the relative low need for bilateral synchronous TAB for the diagnosis of GCA.”
A key issue in GCA diagnosis is the need to confirm inflammation, Dr. Chévez-Barrios said. “The surgeon must obtain a significant portion of the artery, and the pathologist should review several sections and levels of the tissue to confidently say whether there is inflammation or no.”
Frozen sections can spare patients from second procedures
The findings suggest a role for frozen section to help to determine whether a unilateral or bilateral simultaneous TAB should be performed, the study authors noted.
“If the frozen section is positive on the first TAB, a contralateral TAB is deferred, given the very low false-positive rate (0.6%). However, if the frozen section does not align with the permanent section result, in particular if the frozen section is positive but permanent section is negative, the patient returns for a TAB on the contralateral side if the GCA suspicion remains high,” they said.
The use of frozen sections requires ideal conditions in order to be effective, Dr. Chévez-Barrios said. The Mayo Clinic approach “is only possible because of their appropriate hospital setting, the training of the histotechnologists, and the experience of the pathologists interpreting the stains and sections. For most pathology laboratories outside of the Mayo Clinic, frozen sections on arteries are the exception and are used only in specific scenarios.”
In addition, the American College of Rheumatology recommends that patients with a high suspicion of GCA should begin corticosteroids as soon as laboratory studies are obtained; “As a result, if a TAB is performed after treatment begins, the typical active pattern of inflammation in the artery changes,” Dr. Chévez-Barrios said. “This further challenges the diagnosis in a frozen section setting because of the need for immunohistochemistry.” Although frozen sections are feasible in specialized settings such as the Mayo Clinic, most patients receive adequate diagnosis and treatment based on permanent sections.
The study findings were limited by several factors including the use of data from patients at a single center and the unique setup of the Mayo Clinic to perform rapid processing of frozen sections, the researchers noted.
“Additionally, we acknowledge that there is controversy regarding the clinical interpretation of healed arteritis. At our institution, healed arteritis is interpreted in the context of patient clinical characteristics and radiographic findings, which may differ from other institutions and may impact the results of this study,” they said.
Overall, the results support the potential of frozen sections in guiding TAB, although “more studies with a comparative analysis of laboratory results, clinical symptoms, and patient demographic characteristics between positive and negative frozen and permanent TAB results are needed to confirm our findings,” they concluded.
The study received no outside funding. One author reported receiving grants from Eli Lilly and Kiniksa Pharmaceuticals as well as personal fees from Genentech-Roche and Sanofi. Dr. Chévez-Barrios had no financial conflicts to disclose.
Positive findings from frozen sections of a first temporal artery biopsy can effectively identify giant cell arteritis, ruling out in those cases the need to perform a second biopsy on the contralateral side and arguing against the use of simultaneous bilateral biopsies, according to results from a retrospective study of nearly 800 patients who underwent the procedure at the Mayo Clinic during 2010-2018.
Although temporal artery biopsy (TAB) remains the standard diagnostic test for giant cell arteritis (GCA), second TAB procedures are often performed in patients with a high level of suspicion for GCA, which may result in unnecessary treatments and complications, Devon A. Cohen, MD, of the Mayo Clinic, Rochester, Minn., and colleagues wrote. (Dr. Cohen is now a clinical fellow in ophthalmology at the Massachusetts Eye and Ear Infirmary.)
At the Mayo Clinic, TAB specimens are first examined with frozen sections at the time of the biopsy; this process, followed within days by formalin-fixed tissue permanent sections, is unique to Mayo. “A frozen section–guided sequential TAB is commonly performed, with the results of the first biopsy obtained within minutes, which determines the need for evaluation of the contralateral side,” the researchers said. However, the use of frozen sections to evaluate patients with GCA has not been well studied.
In a retrospective cohort study published in JAMA Ophthalmology, the researchers identified TAB patients aged 40 years and older who underwent TAB procedures between Jan. 1, 2010, and Dec. 1, 2018, at the Mayo Clinic. The average age of the patients was 72 years, and 41% were men.
Strong positive predictions from frozen sections
The researchers analyzed 1,162 TABs from 795 patients using frozen and permanent histologic sections.
Overall, 119 patients (15.0%) and 138 TABs had positive permanent section findings, and 103 (86.6%) of these patients also had positive frozen section findings, including 4 false positives and 20 false negatives. The frozen section specificity and sensitivity was 99.4% and 83.2%, respectively, for detecting inflammation suggestive of GCA, and the positive and negative predictive values were 96.1% and 96.6%, respectively. Positive and negative likelihood ratios for frozen section were 140.6 and 0.17, respectively.
In a multivariate analysis, the odds of a positive permanent section TAB significantly increased with age (odds ratio, 1.04), vision loss (OR, 2.72), diplopia (OR, 3.33), headache (OR, 2.32), weight loss (OR, 2.37), and anorexia (OR, 5.65).
A total of 60 patients underwent bilateral TABs, and 307 patients underwent bilateral frozen section–guided sequential TABs; the discordance rates based on permanent sections were 5.0% and 5.5%, respectively.
Those discordance rates are “an important result applying to everyone working with patients suspected for GCA,” Patricia Chévez-Barrios, MD, of Houston Methodist Hospital, wrote in an accompanying editorial. “This is on the low end of what was previously published (3%-40%) and supports the relative low need for bilateral synchronous TAB for the diagnosis of GCA.”
A key issue in GCA diagnosis is the need to confirm inflammation, Dr. Chévez-Barrios said. “The surgeon must obtain a significant portion of the artery, and the pathologist should review several sections and levels of the tissue to confidently say whether there is inflammation or no.”
Frozen sections can spare patients from second procedures
The findings suggest a role for frozen section to help to determine whether a unilateral or bilateral simultaneous TAB should be performed, the study authors noted.
“If the frozen section is positive on the first TAB, a contralateral TAB is deferred, given the very low false-positive rate (0.6%). However, if the frozen section does not align with the permanent section result, in particular if the frozen section is positive but permanent section is negative, the patient returns for a TAB on the contralateral side if the GCA suspicion remains high,” they said.
The use of frozen sections requires ideal conditions in order to be effective, Dr. Chévez-Barrios said. The Mayo Clinic approach “is only possible because of their appropriate hospital setting, the training of the histotechnologists, and the experience of the pathologists interpreting the stains and sections. For most pathology laboratories outside of the Mayo Clinic, frozen sections on arteries are the exception and are used only in specific scenarios.”
In addition, the American College of Rheumatology recommends that patients with a high suspicion of GCA should begin corticosteroids as soon as laboratory studies are obtained; “As a result, if a TAB is performed after treatment begins, the typical active pattern of inflammation in the artery changes,” Dr. Chévez-Barrios said. “This further challenges the diagnosis in a frozen section setting because of the need for immunohistochemistry.” Although frozen sections are feasible in specialized settings such as the Mayo Clinic, most patients receive adequate diagnosis and treatment based on permanent sections.
The study findings were limited by several factors including the use of data from patients at a single center and the unique setup of the Mayo Clinic to perform rapid processing of frozen sections, the researchers noted.
“Additionally, we acknowledge that there is controversy regarding the clinical interpretation of healed arteritis. At our institution, healed arteritis is interpreted in the context of patient clinical characteristics and radiographic findings, which may differ from other institutions and may impact the results of this study,” they said.
Overall, the results support the potential of frozen sections in guiding TAB, although “more studies with a comparative analysis of laboratory results, clinical symptoms, and patient demographic characteristics between positive and negative frozen and permanent TAB results are needed to confirm our findings,” they concluded.
The study received no outside funding. One author reported receiving grants from Eli Lilly and Kiniksa Pharmaceuticals as well as personal fees from Genentech-Roche and Sanofi. Dr. Chévez-Barrios had no financial conflicts to disclose.
FROM JAMA OPHTHALMOLOGY
Romosozumab may not increase cardiovascular risk after all
The potent anabolic, antiosteoporosis agent romosozumab has been saddled with an Food and Drug Administration–mandated black-box warning for increased cardiovascular risk that may not be warranted, Glenn Haugeberg, MD, PhD, asserted at the 2021 Rheumatology Winter Clinical Symposium.
The black-box warning states that romosozumab (Evenity), a monoclonal antibody approved in 2019 for fracture prevention in patients with osteoporosis, may increase the risk of MI, stroke, and cardiovascular death. The warning arose from FDA concerns raised by the results of the phase 3 ARCH trial in which 4,093 postmenopausal women at high fracture risk were randomized to monthly subcutaneous injections of romosozumab or weekly dosing of the oral bisphosphonate alendronate (Fosamax) for 1 year, followed by 12 months of open-label alendronate for all. Alarm bells went off at the FDA because during year 1, the incidence of adjudicated major adverse cardiovascular events was 2.5% in the romosozumab arm, compared with 1.9% with alendronate.
Could a cardioprotective effect of bisphosphonates explain cardiovascular concerns?
However, evidence from multiple animal and human studies suggests that bisphosphonates actually have a cardioprotective effect. For example, a Taiwanese population-based cohort study of 1,548 patients on bisphosphonate therapy for osteoporotic fractures and 4,644 individuals with hip or vertebral fractures who were not on a bisphosphonate showed a 65% reduction in the risk of acute MI during 2 years of follow-up in those who received a bisphosphonate.
“That may explain the ARCH finding. It may – I say may – be that this concern in the ARCH study can be explained by the positive effect of the bisphosphonates on cardiovascular events,” according to Dr. Haugeberg, head of the division of rheumatology at the Southern Norway Hospital Trust, Kristiansand, and professor of medicine at the Norwegian University of Science and Technology, Trondheim.
He noted that, in the FRAME trial, another pivotal phase 3 trial of romosozumab, there was no signal of increased cardiovascular risk, compared with placebo. In FRAME, which included 7,180 osteoporotic postmenopausal women, rates of major adverse cardiovascular events and other adverse events were balanced between the two study arms at 12 months. Indeed, the incidence of adjudicated serious cardiovascular events was 0.5% with romosozumab and 0.4% with placebo injections. After 12 months, all participants were transitioned to denosumab (Prolia) for another 12 months. At 24 months, there remained no significant between-group difference in cardiovascular events, cancer, osteoarthritis, hyperostosis, or other major adverse events.
Potency of romosozumab
Romosozumab’s efficacy for fracture prevention in these two pivotal trials was striking. The risk of new vertebral fractures was reduced by 73% with romosozumab, compared with placebo at 12 months in FRAME, and by 75% at 24 months in the romosozumab-to-denosumab group.
“FRAME was a 12-month study for the primary endpoint. The bisphosphonate studies typically had a 3-year design in order to show benefit, but here you see only 12-month follow-up. This illustrates the potency of this drug. We saw rapid increase in bone density and a huge decrease in new vertebral fractures versus placebo in the first 12 months, then during follow-up with denosumab the reduction in fractures was maintained,” the rheumatologist commented.
In the ARCH trial, where romosozumab went head to head with a very effective oral bisphosphonate, the risk of new vertebral fractures was 48% lower at 24 months in the romosozumab-to-alendronate group than in women on alendronate for the full 24 months, while the risk of hip fractures was reduced by 38%.
Romosozumab is a humanized monoclonal antibody with a novel mechanism of anabolic action: This agent binds to sclerostin, which is produced in osteocytes. When sclerostin binds to receptors on osteoblasts it reduces their activity, thereby inhibiting bone formation. Romosozumab takes away this inhibition of osteoblasts, boosting their activity. The result is increased bone formation accompanied by decreased bone resorption. This allows for a logical treatment approach: first using an anabolic agent – in this instance, subcutaneously injected romosozumab at 210 mg once monthly for 12 months – then switching to an antiresorptive agent in order to maintain the gain in bone mineral density and decrease fracture risk. This is the same treatment strategy recommended when using the anabolic agents teriparatide (Forteo) and abaloparatide (Tymlos); however, those parathyroid hormone and parathyroid hormone–related protein analogs are seldom used in Norway because their cost is substantially greater than for romosozumab, he explained.
Updated Endocrine Society guidelines
Dr. Haugeberg called romosozumab “a new and wonderful drug.” The Endocrine Society also considers romosozumab an important new drug, as evidenced by the release of an 8-page update of the group’s clinical practice guideline on the pharmacologic management of osteoporosis in postmenopausal women; the update was devoted specifically to the use of romosozumab. The update, published in response to the biologic’s recent approval by U.S., Canadian, and European regulatory agencies, came just 10 months after release of the Endocrine Society’s comprehensive 28-page clinical practice guideline.
Dr. Haugeberg is a fan of the Endocrine Society guideline, which recommends romosozumab as a first-line therapy in postmenopausal women at very high risk of osteoporotic fracture, defined as those with a history of multiple vertebral fractures or severe osteoporosis with a T score of –2.5 or less at the hip or spine plus fractures. The updated guideline also recommends consideration of the antisclerostin biologic in high-risk patients who have failed on antiresorptive treatments.
The practice guideline states that the issue of a possible cardioprotective effect of alendronate in the ARCH trial “remains uncertain at this time.”
“Women at high risk of cardiovascular disease and stroke should not be considered for romosozumab pending further studies on cardiovascular risk associated with this treatment,” according to the Endocrine Society.
Dr. Haugeberg reported receiving research grants from Pfizer and Biogen and serving as a consultant to and/or on speakers’ bureaus for Amgen, which markets romosozumab, and more than a dozen other pharmaceutical companies.
The potent anabolic, antiosteoporosis agent romosozumab has been saddled with an Food and Drug Administration–mandated black-box warning for increased cardiovascular risk that may not be warranted, Glenn Haugeberg, MD, PhD, asserted at the 2021 Rheumatology Winter Clinical Symposium.
The black-box warning states that romosozumab (Evenity), a monoclonal antibody approved in 2019 for fracture prevention in patients with osteoporosis, may increase the risk of MI, stroke, and cardiovascular death. The warning arose from FDA concerns raised by the results of the phase 3 ARCH trial in which 4,093 postmenopausal women at high fracture risk were randomized to monthly subcutaneous injections of romosozumab or weekly dosing of the oral bisphosphonate alendronate (Fosamax) for 1 year, followed by 12 months of open-label alendronate for all. Alarm bells went off at the FDA because during year 1, the incidence of adjudicated major adverse cardiovascular events was 2.5% in the romosozumab arm, compared with 1.9% with alendronate.
Could a cardioprotective effect of bisphosphonates explain cardiovascular concerns?
However, evidence from multiple animal and human studies suggests that bisphosphonates actually have a cardioprotective effect. For example, a Taiwanese population-based cohort study of 1,548 patients on bisphosphonate therapy for osteoporotic fractures and 4,644 individuals with hip or vertebral fractures who were not on a bisphosphonate showed a 65% reduction in the risk of acute MI during 2 years of follow-up in those who received a bisphosphonate.
“That may explain the ARCH finding. It may – I say may – be that this concern in the ARCH study can be explained by the positive effect of the bisphosphonates on cardiovascular events,” according to Dr. Haugeberg, head of the division of rheumatology at the Southern Norway Hospital Trust, Kristiansand, and professor of medicine at the Norwegian University of Science and Technology, Trondheim.
He noted that, in the FRAME trial, another pivotal phase 3 trial of romosozumab, there was no signal of increased cardiovascular risk, compared with placebo. In FRAME, which included 7,180 osteoporotic postmenopausal women, rates of major adverse cardiovascular events and other adverse events were balanced between the two study arms at 12 months. Indeed, the incidence of adjudicated serious cardiovascular events was 0.5% with romosozumab and 0.4% with placebo injections. After 12 months, all participants were transitioned to denosumab (Prolia) for another 12 months. At 24 months, there remained no significant between-group difference in cardiovascular events, cancer, osteoarthritis, hyperostosis, or other major adverse events.
Potency of romosozumab
Romosozumab’s efficacy for fracture prevention in these two pivotal trials was striking. The risk of new vertebral fractures was reduced by 73% with romosozumab, compared with placebo at 12 months in FRAME, and by 75% at 24 months in the romosozumab-to-denosumab group.
“FRAME was a 12-month study for the primary endpoint. The bisphosphonate studies typically had a 3-year design in order to show benefit, but here you see only 12-month follow-up. This illustrates the potency of this drug. We saw rapid increase in bone density and a huge decrease in new vertebral fractures versus placebo in the first 12 months, then during follow-up with denosumab the reduction in fractures was maintained,” the rheumatologist commented.
In the ARCH trial, where romosozumab went head to head with a very effective oral bisphosphonate, the risk of new vertebral fractures was 48% lower at 24 months in the romosozumab-to-alendronate group than in women on alendronate for the full 24 months, while the risk of hip fractures was reduced by 38%.
Romosozumab is a humanized monoclonal antibody with a novel mechanism of anabolic action: This agent binds to sclerostin, which is produced in osteocytes. When sclerostin binds to receptors on osteoblasts it reduces their activity, thereby inhibiting bone formation. Romosozumab takes away this inhibition of osteoblasts, boosting their activity. The result is increased bone formation accompanied by decreased bone resorption. This allows for a logical treatment approach: first using an anabolic agent – in this instance, subcutaneously injected romosozumab at 210 mg once monthly for 12 months – then switching to an antiresorptive agent in order to maintain the gain in bone mineral density and decrease fracture risk. This is the same treatment strategy recommended when using the anabolic agents teriparatide (Forteo) and abaloparatide (Tymlos); however, those parathyroid hormone and parathyroid hormone–related protein analogs are seldom used in Norway because their cost is substantially greater than for romosozumab, he explained.
Updated Endocrine Society guidelines
Dr. Haugeberg called romosozumab “a new and wonderful drug.” The Endocrine Society also considers romosozumab an important new drug, as evidenced by the release of an 8-page update of the group’s clinical practice guideline on the pharmacologic management of osteoporosis in postmenopausal women; the update was devoted specifically to the use of romosozumab. The update, published in response to the biologic’s recent approval by U.S., Canadian, and European regulatory agencies, came just 10 months after release of the Endocrine Society’s comprehensive 28-page clinical practice guideline.
Dr. Haugeberg is a fan of the Endocrine Society guideline, which recommends romosozumab as a first-line therapy in postmenopausal women at very high risk of osteoporotic fracture, defined as those with a history of multiple vertebral fractures or severe osteoporosis with a T score of –2.5 or less at the hip or spine plus fractures. The updated guideline also recommends consideration of the antisclerostin biologic in high-risk patients who have failed on antiresorptive treatments.
The practice guideline states that the issue of a possible cardioprotective effect of alendronate in the ARCH trial “remains uncertain at this time.”
“Women at high risk of cardiovascular disease and stroke should not be considered for romosozumab pending further studies on cardiovascular risk associated with this treatment,” according to the Endocrine Society.
Dr. Haugeberg reported receiving research grants from Pfizer and Biogen and serving as a consultant to and/or on speakers’ bureaus for Amgen, which markets romosozumab, and more than a dozen other pharmaceutical companies.
The potent anabolic, antiosteoporosis agent romosozumab has been saddled with an Food and Drug Administration–mandated black-box warning for increased cardiovascular risk that may not be warranted, Glenn Haugeberg, MD, PhD, asserted at the 2021 Rheumatology Winter Clinical Symposium.
The black-box warning states that romosozumab (Evenity), a monoclonal antibody approved in 2019 for fracture prevention in patients with osteoporosis, may increase the risk of MI, stroke, and cardiovascular death. The warning arose from FDA concerns raised by the results of the phase 3 ARCH trial in which 4,093 postmenopausal women at high fracture risk were randomized to monthly subcutaneous injections of romosozumab or weekly dosing of the oral bisphosphonate alendronate (Fosamax) for 1 year, followed by 12 months of open-label alendronate for all. Alarm bells went off at the FDA because during year 1, the incidence of adjudicated major adverse cardiovascular events was 2.5% in the romosozumab arm, compared with 1.9% with alendronate.
Could a cardioprotective effect of bisphosphonates explain cardiovascular concerns?
However, evidence from multiple animal and human studies suggests that bisphosphonates actually have a cardioprotective effect. For example, a Taiwanese population-based cohort study of 1,548 patients on bisphosphonate therapy for osteoporotic fractures and 4,644 individuals with hip or vertebral fractures who were not on a bisphosphonate showed a 65% reduction in the risk of acute MI during 2 years of follow-up in those who received a bisphosphonate.
“That may explain the ARCH finding. It may – I say may – be that this concern in the ARCH study can be explained by the positive effect of the bisphosphonates on cardiovascular events,” according to Dr. Haugeberg, head of the division of rheumatology at the Southern Norway Hospital Trust, Kristiansand, and professor of medicine at the Norwegian University of Science and Technology, Trondheim.
He noted that, in the FRAME trial, another pivotal phase 3 trial of romosozumab, there was no signal of increased cardiovascular risk, compared with placebo. In FRAME, which included 7,180 osteoporotic postmenopausal women, rates of major adverse cardiovascular events and other adverse events were balanced between the two study arms at 12 months. Indeed, the incidence of adjudicated serious cardiovascular events was 0.5% with romosozumab and 0.4% with placebo injections. After 12 months, all participants were transitioned to denosumab (Prolia) for another 12 months. At 24 months, there remained no significant between-group difference in cardiovascular events, cancer, osteoarthritis, hyperostosis, or other major adverse events.
Potency of romosozumab
Romosozumab’s efficacy for fracture prevention in these two pivotal trials was striking. The risk of new vertebral fractures was reduced by 73% with romosozumab, compared with placebo at 12 months in FRAME, and by 75% at 24 months in the romosozumab-to-denosumab group.
“FRAME was a 12-month study for the primary endpoint. The bisphosphonate studies typically had a 3-year design in order to show benefit, but here you see only 12-month follow-up. This illustrates the potency of this drug. We saw rapid increase in bone density and a huge decrease in new vertebral fractures versus placebo in the first 12 months, then during follow-up with denosumab the reduction in fractures was maintained,” the rheumatologist commented.
In the ARCH trial, where romosozumab went head to head with a very effective oral bisphosphonate, the risk of new vertebral fractures was 48% lower at 24 months in the romosozumab-to-alendronate group than in women on alendronate for the full 24 months, while the risk of hip fractures was reduced by 38%.
Romosozumab is a humanized monoclonal antibody with a novel mechanism of anabolic action: This agent binds to sclerostin, which is produced in osteocytes. When sclerostin binds to receptors on osteoblasts it reduces their activity, thereby inhibiting bone formation. Romosozumab takes away this inhibition of osteoblasts, boosting their activity. The result is increased bone formation accompanied by decreased bone resorption. This allows for a logical treatment approach: first using an anabolic agent – in this instance, subcutaneously injected romosozumab at 210 mg once monthly for 12 months – then switching to an antiresorptive agent in order to maintain the gain in bone mineral density and decrease fracture risk. This is the same treatment strategy recommended when using the anabolic agents teriparatide (Forteo) and abaloparatide (Tymlos); however, those parathyroid hormone and parathyroid hormone–related protein analogs are seldom used in Norway because their cost is substantially greater than for romosozumab, he explained.
Updated Endocrine Society guidelines
Dr. Haugeberg called romosozumab “a new and wonderful drug.” The Endocrine Society also considers romosozumab an important new drug, as evidenced by the release of an 8-page update of the group’s clinical practice guideline on the pharmacologic management of osteoporosis in postmenopausal women; the update was devoted specifically to the use of romosozumab. The update, published in response to the biologic’s recent approval by U.S., Canadian, and European regulatory agencies, came just 10 months after release of the Endocrine Society’s comprehensive 28-page clinical practice guideline.
Dr. Haugeberg is a fan of the Endocrine Society guideline, which recommends romosozumab as a first-line therapy in postmenopausal women at very high risk of osteoporotic fracture, defined as those with a history of multiple vertebral fractures or severe osteoporosis with a T score of –2.5 or less at the hip or spine plus fractures. The updated guideline also recommends consideration of the antisclerostin biologic in high-risk patients who have failed on antiresorptive treatments.
The practice guideline states that the issue of a possible cardioprotective effect of alendronate in the ARCH trial “remains uncertain at this time.”
“Women at high risk of cardiovascular disease and stroke should not be considered for romosozumab pending further studies on cardiovascular risk associated with this treatment,” according to the Endocrine Society.
Dr. Haugeberg reported receiving research grants from Pfizer and Biogen and serving as a consultant to and/or on speakers’ bureaus for Amgen, which markets romosozumab, and more than a dozen other pharmaceutical companies.
FROM RWCS 2021
Outcomes have improved for PAH in connective tissue disease
Survival rates for patients with pulmonary arterial hypertension associated with connective tissue diseases have improved significantly in recent years, and there is growing evidence that treatments for idiopathic pulmonary arterial hypertension can also benefit this group.
In an article published online Feb. 3, 2021, in Arthritis & Rheumatology, researchers report the outcomes of a meta-analysis to explore the effect of more modern pulmonary arterial hypertension treatments on patients with conditions such as systemic sclerosis.
First author Dinesh Khanna, MBBS, MSc, of the division of rheumatology at the University of Michigan, Ann Arbor, said in an interview that connective tissue disease–associated pulmonary arterial hypertension (CTD-PAH) was a leading cause of death, but earlier clinical trials had found poor outcomes in patients with CTD, compared with those with idiopathic PAH.
“Recent clinical trial data show that aggressive, up-front PAH treatments have better outcomes in those with CTD-PAH, and we wanted to explore these observations carefully in a systematic review and meta-analysis,” Dr. Khanna said.
The analysis included 11 randomized, controlled trials, involving 4,329 patients with PAH (1,267 with CTD), and 19 registries with a total of 9,739 patients with PAH, including 4,008 with CTD. Trials were required to report long-term clinical outcomes with a median enrollment time of greater than 6 months, and outcomes measured between 3-6 months after the patients started treatment.
Patients with CTDs had an older mean age and a lower 6-minute walk distance than did those with idiopathic PAH.
Five randomized, controlled trials – involving 3,172 patients, 941 of whom had a CTD – found that additional PAH treatment was associated with a 36% reduction in the risk of morbidity or mortality events, compared with controls both in the overall PAH group and in those with CTD.
Additional therapy was also associated with a 34.6-meter increase in 6-minute walk distance in the general PAH population, and a 20.4-meter increase in those with CTD.
The authors commented that the smaller improvement in 6-minute walk distance among patients with CTD may be influenced by comorbidities such as musculoskeletal involvement that would be independent of their cardiopulmonary function.
Differential patient survival among PAH etiologies
“Our meta-analysis of RCTs demonstrated that patients with CTD-PAH derive a clinically significant benefit from currently available PAH therapies which, in many patients, comprised the addition of a drug targeting a second or third pathway involved in the pathophysiology of PAH,” the authors wrote.
When researchers analyzed data from nine registries that included a wide range of PAH etiologies, they found the overall survival rates were lower among patients with CTD, compared with the overall population. The analysis also suggested that patients with systemic sclerosis and PAH had lower survival rates than did those with systemic lupus erythematosus.
Dr. Khanna said this may relate to different pathophysiology of PAH in patients with CTDs, but could also be a reflection of other differences, such as older age and the involvement of other comorbidities, including lung fibrosis and heart involvement.
Data across all 19 registries also showed that survival rates among those with CTD were higher in registries where more than 50% of the registry study period was during or after 2010, compared with registries where 50% or more of the study period was before 2010.
The authors suggested the differences in survival rates may relate to increased screening for PAH, particularly among people with CTDs. They noted that increased screening leads to earlier diagnosis, which could introduce a lead-time bias such that later registries would have younger participants with less severe disease. However, their analysis found that the later registries had older patients but also with less severe disease, and they suggested that it wasn’t possible to determine if lead-time bias was playing a role in their results.
Improvements in treatment options could also account for differences in survival over time, although the authors commented that only six registries in the study included patients from 2015 or later, when currently available treatments came into use and early combination therapy was used more.
“These data also support the 2018 World Symposium on Pulmonary Hypertension recommendations to initiate up-front combination pulmonary arterial hypertension therapy in majority of cases with CTD-PAH,” Dr. Khanna said.
‘Still have to be aggressive at identifying the high-risk patients’
Commenting on the findings, Virginia Steen, MD, of the division of rheumatology at Georgetown University, Washington, said clinicians were finally seeing some significant changes over time in scleroderma-associated PAH.
“Although some of it may be just early diagnosis, I think that the combination of early diagnosis and more aggressive treatment with combination medication is definitely making a difference,” Dr. Steen said in an interview. “The bottom line is that we as rheumatologists still have to be aggressive at identifying the high-risk patients, making an early diagnosis, and working with our pulmonary hypertension colleagues and aggressively treating these patients so we can make a long-term difference.”
The authors of an accompanying editorial said the meta-analysis’ findings showed the positive impact of early combination therapy and early diagnosis through proactive screening.
“It is notable because the present analysis again confirms that outcomes are worse in CTD-PAH than in idiopathic or familial forms of PAH, the impact of treatments should no longer be regarded as insignificant,” the editorial’s authors wrote. “This is a practice changing observation, especially now that many of the drugs are available in generic formulations and so the cost of modern PAH treatment has fallen at the same time as its true value is convincingly demonstrated.”
They also argued there was strong evidence for the value of combination therapies, both for PAH-targeted drugs used in combination and concurrent use of immunosuppression and drugs specifically for PAH in some patients with CTD-PAH.
However, they pointed out that not all treatments for idiopathic PAH were suitable for patients with CTDs, highlighting the example of anticoagulation that can improve survival in the first but worsen it in the second.
The study was funded by Actelion. Six authors declared funding and grants from the pharmaceutical sector, including the study sponsor, and three authors were employees of Actelion.
Survival rates for patients with pulmonary arterial hypertension associated with connective tissue diseases have improved significantly in recent years, and there is growing evidence that treatments for idiopathic pulmonary arterial hypertension can also benefit this group.
In an article published online Feb. 3, 2021, in Arthritis & Rheumatology, researchers report the outcomes of a meta-analysis to explore the effect of more modern pulmonary arterial hypertension treatments on patients with conditions such as systemic sclerosis.
First author Dinesh Khanna, MBBS, MSc, of the division of rheumatology at the University of Michigan, Ann Arbor, said in an interview that connective tissue disease–associated pulmonary arterial hypertension (CTD-PAH) was a leading cause of death, but earlier clinical trials had found poor outcomes in patients with CTD, compared with those with idiopathic PAH.
“Recent clinical trial data show that aggressive, up-front PAH treatments have better outcomes in those with CTD-PAH, and we wanted to explore these observations carefully in a systematic review and meta-analysis,” Dr. Khanna said.
The analysis included 11 randomized, controlled trials, involving 4,329 patients with PAH (1,267 with CTD), and 19 registries with a total of 9,739 patients with PAH, including 4,008 with CTD. Trials were required to report long-term clinical outcomes with a median enrollment time of greater than 6 months, and outcomes measured between 3-6 months after the patients started treatment.
Patients with CTDs had an older mean age and a lower 6-minute walk distance than did those with idiopathic PAH.
Five randomized, controlled trials – involving 3,172 patients, 941 of whom had a CTD – found that additional PAH treatment was associated with a 36% reduction in the risk of morbidity or mortality events, compared with controls both in the overall PAH group and in those with CTD.
Additional therapy was also associated with a 34.6-meter increase in 6-minute walk distance in the general PAH population, and a 20.4-meter increase in those with CTD.
The authors commented that the smaller improvement in 6-minute walk distance among patients with CTD may be influenced by comorbidities such as musculoskeletal involvement that would be independent of their cardiopulmonary function.
Differential patient survival among PAH etiologies
“Our meta-analysis of RCTs demonstrated that patients with CTD-PAH derive a clinically significant benefit from currently available PAH therapies which, in many patients, comprised the addition of a drug targeting a second or third pathway involved in the pathophysiology of PAH,” the authors wrote.
When researchers analyzed data from nine registries that included a wide range of PAH etiologies, they found the overall survival rates were lower among patients with CTD, compared with the overall population. The analysis also suggested that patients with systemic sclerosis and PAH had lower survival rates than did those with systemic lupus erythematosus.
Dr. Khanna said this may relate to different pathophysiology of PAH in patients with CTDs, but could also be a reflection of other differences, such as older age and the involvement of other comorbidities, including lung fibrosis and heart involvement.
Data across all 19 registries also showed that survival rates among those with CTD were higher in registries where more than 50% of the registry study period was during or after 2010, compared with registries where 50% or more of the study period was before 2010.
The authors suggested the differences in survival rates may relate to increased screening for PAH, particularly among people with CTDs. They noted that increased screening leads to earlier diagnosis, which could introduce a lead-time bias such that later registries would have younger participants with less severe disease. However, their analysis found that the later registries had older patients but also with less severe disease, and they suggested that it wasn’t possible to determine if lead-time bias was playing a role in their results.
Improvements in treatment options could also account for differences in survival over time, although the authors commented that only six registries in the study included patients from 2015 or later, when currently available treatments came into use and early combination therapy was used more.
“These data also support the 2018 World Symposium on Pulmonary Hypertension recommendations to initiate up-front combination pulmonary arterial hypertension therapy in majority of cases with CTD-PAH,” Dr. Khanna said.
‘Still have to be aggressive at identifying the high-risk patients’
Commenting on the findings, Virginia Steen, MD, of the division of rheumatology at Georgetown University, Washington, said clinicians were finally seeing some significant changes over time in scleroderma-associated PAH.
“Although some of it may be just early diagnosis, I think that the combination of early diagnosis and more aggressive treatment with combination medication is definitely making a difference,” Dr. Steen said in an interview. “The bottom line is that we as rheumatologists still have to be aggressive at identifying the high-risk patients, making an early diagnosis, and working with our pulmonary hypertension colleagues and aggressively treating these patients so we can make a long-term difference.”
The authors of an accompanying editorial said the meta-analysis’ findings showed the positive impact of early combination therapy and early diagnosis through proactive screening.
“It is notable because the present analysis again confirms that outcomes are worse in CTD-PAH than in idiopathic or familial forms of PAH, the impact of treatments should no longer be regarded as insignificant,” the editorial’s authors wrote. “This is a practice changing observation, especially now that many of the drugs are available in generic formulations and so the cost of modern PAH treatment has fallen at the same time as its true value is convincingly demonstrated.”
They also argued there was strong evidence for the value of combination therapies, both for PAH-targeted drugs used in combination and concurrent use of immunosuppression and drugs specifically for PAH in some patients with CTD-PAH.
However, they pointed out that not all treatments for idiopathic PAH were suitable for patients with CTDs, highlighting the example of anticoagulation that can improve survival in the first but worsen it in the second.
The study was funded by Actelion. Six authors declared funding and grants from the pharmaceutical sector, including the study sponsor, and three authors were employees of Actelion.
Survival rates for patients with pulmonary arterial hypertension associated with connective tissue diseases have improved significantly in recent years, and there is growing evidence that treatments for idiopathic pulmonary arterial hypertension can also benefit this group.
In an article published online Feb. 3, 2021, in Arthritis & Rheumatology, researchers report the outcomes of a meta-analysis to explore the effect of more modern pulmonary arterial hypertension treatments on patients with conditions such as systemic sclerosis.
First author Dinesh Khanna, MBBS, MSc, of the division of rheumatology at the University of Michigan, Ann Arbor, said in an interview that connective tissue disease–associated pulmonary arterial hypertension (CTD-PAH) was a leading cause of death, but earlier clinical trials had found poor outcomes in patients with CTD, compared with those with idiopathic PAH.
“Recent clinical trial data show that aggressive, up-front PAH treatments have better outcomes in those with CTD-PAH, and we wanted to explore these observations carefully in a systematic review and meta-analysis,” Dr. Khanna said.
The analysis included 11 randomized, controlled trials, involving 4,329 patients with PAH (1,267 with CTD), and 19 registries with a total of 9,739 patients with PAH, including 4,008 with CTD. Trials were required to report long-term clinical outcomes with a median enrollment time of greater than 6 months, and outcomes measured between 3-6 months after the patients started treatment.
Patients with CTDs had an older mean age and a lower 6-minute walk distance than did those with idiopathic PAH.
Five randomized, controlled trials – involving 3,172 patients, 941 of whom had a CTD – found that additional PAH treatment was associated with a 36% reduction in the risk of morbidity or mortality events, compared with controls both in the overall PAH group and in those with CTD.
Additional therapy was also associated with a 34.6-meter increase in 6-minute walk distance in the general PAH population, and a 20.4-meter increase in those with CTD.
The authors commented that the smaller improvement in 6-minute walk distance among patients with CTD may be influenced by comorbidities such as musculoskeletal involvement that would be independent of their cardiopulmonary function.
Differential patient survival among PAH etiologies
“Our meta-analysis of RCTs demonstrated that patients with CTD-PAH derive a clinically significant benefit from currently available PAH therapies which, in many patients, comprised the addition of a drug targeting a second or third pathway involved in the pathophysiology of PAH,” the authors wrote.
When researchers analyzed data from nine registries that included a wide range of PAH etiologies, they found the overall survival rates were lower among patients with CTD, compared with the overall population. The analysis also suggested that patients with systemic sclerosis and PAH had lower survival rates than did those with systemic lupus erythematosus.
Dr. Khanna said this may relate to different pathophysiology of PAH in patients with CTDs, but could also be a reflection of other differences, such as older age and the involvement of other comorbidities, including lung fibrosis and heart involvement.
Data across all 19 registries also showed that survival rates among those with CTD were higher in registries where more than 50% of the registry study period was during or after 2010, compared with registries where 50% or more of the study period was before 2010.
The authors suggested the differences in survival rates may relate to increased screening for PAH, particularly among people with CTDs. They noted that increased screening leads to earlier diagnosis, which could introduce a lead-time bias such that later registries would have younger participants with less severe disease. However, their analysis found that the later registries had older patients but also with less severe disease, and they suggested that it wasn’t possible to determine if lead-time bias was playing a role in their results.
Improvements in treatment options could also account for differences in survival over time, although the authors commented that only six registries in the study included patients from 2015 or later, when currently available treatments came into use and early combination therapy was used more.
“These data also support the 2018 World Symposium on Pulmonary Hypertension recommendations to initiate up-front combination pulmonary arterial hypertension therapy in majority of cases with CTD-PAH,” Dr. Khanna said.
‘Still have to be aggressive at identifying the high-risk patients’
Commenting on the findings, Virginia Steen, MD, of the division of rheumatology at Georgetown University, Washington, said clinicians were finally seeing some significant changes over time in scleroderma-associated PAH.
“Although some of it may be just early diagnosis, I think that the combination of early diagnosis and more aggressive treatment with combination medication is definitely making a difference,” Dr. Steen said in an interview. “The bottom line is that we as rheumatologists still have to be aggressive at identifying the high-risk patients, making an early diagnosis, and working with our pulmonary hypertension colleagues and aggressively treating these patients so we can make a long-term difference.”
The authors of an accompanying editorial said the meta-analysis’ findings showed the positive impact of early combination therapy and early diagnosis through proactive screening.
“It is notable because the present analysis again confirms that outcomes are worse in CTD-PAH than in idiopathic or familial forms of PAH, the impact of treatments should no longer be regarded as insignificant,” the editorial’s authors wrote. “This is a practice changing observation, especially now that many of the drugs are available in generic formulations and so the cost of modern PAH treatment has fallen at the same time as its true value is convincingly demonstrated.”
They also argued there was strong evidence for the value of combination therapies, both for PAH-targeted drugs used in combination and concurrent use of immunosuppression and drugs specifically for PAH in some patients with CTD-PAH.
However, they pointed out that not all treatments for idiopathic PAH were suitable for patients with CTDs, highlighting the example of anticoagulation that can improve survival in the first but worsen it in the second.
The study was funded by Actelion. Six authors declared funding and grants from the pharmaceutical sector, including the study sponsor, and three authors were employees of Actelion.
FROM ARTHRITIS & RHEUMATOLOGY
Consider home subcutaneous immune globulin for refractory dermatomyositis
Home-based subcutaneous immune globulin therapy is a promising alternative to intravenous immune globulin therapy for patients with refractory dermatomyositis or polymyositis, Anna Postolova, MD, MPH, declared at the 2021 Rheumatology Winter Clinical Symposium.
“This is really exciting. I think in the years to come we may see a change to having our patients be able to do immune globulin therapy at home,” said Dr. Postolova, a rheumatologist and allergist/immunologist at Stanford (Calif.) Health Care.
“The technology is there. I think our patients might feel more comfortable getting immune globulin at home,” she said. “I would love to switch more patients from IVIg to SCIg [subcutaneous immune globulin] in my practice.”
A few caveats: SCIg remains off label for treatment of dermatomyositis (DM) or polymyositis (PM). Its approved indication is as replacement therapy in patients with primary or secondary immunodeficiency diseases. IVIg is approved for this indication, but is also approved for DM/PM refractory to high-dose corticosteroids and immunosuppressants. Yet SCIg is clearly effective for these autoimmune inflammatory diseases, albeit to date the supporting evidence comes chiefly from observational studies and anecdotal experience.
“I don’t know if insurers will cover it, but they should because it’s obviously a lot cheaper to do it at home,” she noted.
SCIg advantages
SCIg offers compelling advantages over IVIg in addition to its substantially lower cost. These include far fewer systemic side effects, shorter infusion time, greater bioavailability, and better quality of life. Patients self-administer SCIg at home, avoiding the inconvenience of IVIg therapy, which entails travel time for once-monthly hospitalization or long hours spent in an infusion center, she explained.
French investigators recently documented a previously unappreciated further advantage of home-based SCIg. They convened a focus group of patients with DM or PM experienced with both IVIg and home SCIg and determined that participants uniformly preferred home SCIg. The patients cited a new and welcome feeling of autonomy and control.
“All patients with experience of IVIg and SCIg expressed a clear preference for SCIg, which was described to be easy, less disruptive for daily life, well tolerated, and less time-consuming. Preference was mainly related to a restoration of autonomy. Home-based self-administration reinforced the feeling of independence,” according to the investigators.
Available products
Six preparations of SCIg are commercially available. Most are in 10% concentration, as are all IVIg products. However, a 20% formulation of SCIg known as Hizentra allows for a smaller infusion volume and quicker completion of a treatment session. And one SCIg product, HyQvia, uses recombinant human hyaluronidase-facilitated 10% immune globulin, allowing home infusion of large volumes of sustained-release immune globulin on a once-monthly basis.
The relatively recent introduction of home SCIg for treatment of autoimmune inflammatory diseases, including DM, PM, and chronic inflammatory demyelinating polyneuropathy, has been pioneered mainly by European investigators. The treatment is often given by programmable mechanical pump once weekly. Italian investigators have reported efficacy in DM using 0.2 g/kg per week, which is about half the monthly total dose of IVIg employed. The infusion rate is 10-40 mL/hour, with a volume of around 35 mL per injection site.
Alternatively, SCIg can be delivered by rapid push infusions of smaller volumes with a syringe two or three times per week; that’s the regimen that was used at 2 g/kg over the course of a month by patients in the French focus group study, who didn’t mind the more frequent dosing.
“As they have had severe long-lasting symptoms, SCIg was perceived as a curative rather than a preventive therapy,” according to the French investigators.
More than 40% of patients experience adverse reactions to IVIg. These often involve headaches, nausea, back or abdominal pain, arthralgias, and/or difficulty breathing. Thromboembolic events and acute renal failure occur occasionally. For this reason, many physicians give a prophylactic dose of corticosteroids an hour before a patient’s first dose of IVIg. These systemic side effects are so rare with SCIg that Dr. Postolova has never pretreated with steroids, even though the main reason she resorts to the home therapy is a patient’s track record of poor tolerance of IVIg. The lower abdomen and thigh are the most commonly used subcutaneous infusion sites. Mild local infusion site reactions are fairly common.
Formulating IVIg and SCIg is a complex process that entails plasma procurement and pooling, fractionation, and purification. It takes 10,000-60,000 plasma donations to make one lot of IVIg. Donations are accepted only from repeated donors. Samples are held for 6 months and tested for infectious agents. However, efforts are underway to develop bioengineered recombinant immune globulin products that don’t require donated plasma. These products are being designed to capture and enhance the most important mechanisms of benefit of plasma-derived immunoglobulins using Fc fragments that target key receptors, rather than relying on full-length immune globulin. The goal is enhanced efficacy at much lower doses than with IVIg or SCIg.
Dr. Postolova reported having no financial conflicts regarding her presentation.
Home-based subcutaneous immune globulin therapy is a promising alternative to intravenous immune globulin therapy for patients with refractory dermatomyositis or polymyositis, Anna Postolova, MD, MPH, declared at the 2021 Rheumatology Winter Clinical Symposium.
“This is really exciting. I think in the years to come we may see a change to having our patients be able to do immune globulin therapy at home,” said Dr. Postolova, a rheumatologist and allergist/immunologist at Stanford (Calif.) Health Care.
“The technology is there. I think our patients might feel more comfortable getting immune globulin at home,” she said. “I would love to switch more patients from IVIg to SCIg [subcutaneous immune globulin] in my practice.”
A few caveats: SCIg remains off label for treatment of dermatomyositis (DM) or polymyositis (PM). Its approved indication is as replacement therapy in patients with primary or secondary immunodeficiency diseases. IVIg is approved for this indication, but is also approved for DM/PM refractory to high-dose corticosteroids and immunosuppressants. Yet SCIg is clearly effective for these autoimmune inflammatory diseases, albeit to date the supporting evidence comes chiefly from observational studies and anecdotal experience.
“I don’t know if insurers will cover it, but they should because it’s obviously a lot cheaper to do it at home,” she noted.
SCIg advantages
SCIg offers compelling advantages over IVIg in addition to its substantially lower cost. These include far fewer systemic side effects, shorter infusion time, greater bioavailability, and better quality of life. Patients self-administer SCIg at home, avoiding the inconvenience of IVIg therapy, which entails travel time for once-monthly hospitalization or long hours spent in an infusion center, she explained.
French investigators recently documented a previously unappreciated further advantage of home-based SCIg. They convened a focus group of patients with DM or PM experienced with both IVIg and home SCIg and determined that participants uniformly preferred home SCIg. The patients cited a new and welcome feeling of autonomy and control.
“All patients with experience of IVIg and SCIg expressed a clear preference for SCIg, which was described to be easy, less disruptive for daily life, well tolerated, and less time-consuming. Preference was mainly related to a restoration of autonomy. Home-based self-administration reinforced the feeling of independence,” according to the investigators.
Available products
Six preparations of SCIg are commercially available. Most are in 10% concentration, as are all IVIg products. However, a 20% formulation of SCIg known as Hizentra allows for a smaller infusion volume and quicker completion of a treatment session. And one SCIg product, HyQvia, uses recombinant human hyaluronidase-facilitated 10% immune globulin, allowing home infusion of large volumes of sustained-release immune globulin on a once-monthly basis.
The relatively recent introduction of home SCIg for treatment of autoimmune inflammatory diseases, including DM, PM, and chronic inflammatory demyelinating polyneuropathy, has been pioneered mainly by European investigators. The treatment is often given by programmable mechanical pump once weekly. Italian investigators have reported efficacy in DM using 0.2 g/kg per week, which is about half the monthly total dose of IVIg employed. The infusion rate is 10-40 mL/hour, with a volume of around 35 mL per injection site.
Alternatively, SCIg can be delivered by rapid push infusions of smaller volumes with a syringe two or three times per week; that’s the regimen that was used at 2 g/kg over the course of a month by patients in the French focus group study, who didn’t mind the more frequent dosing.
“As they have had severe long-lasting symptoms, SCIg was perceived as a curative rather than a preventive therapy,” according to the French investigators.
More than 40% of patients experience adverse reactions to IVIg. These often involve headaches, nausea, back or abdominal pain, arthralgias, and/or difficulty breathing. Thromboembolic events and acute renal failure occur occasionally. For this reason, many physicians give a prophylactic dose of corticosteroids an hour before a patient’s first dose of IVIg. These systemic side effects are so rare with SCIg that Dr. Postolova has never pretreated with steroids, even though the main reason she resorts to the home therapy is a patient’s track record of poor tolerance of IVIg. The lower abdomen and thigh are the most commonly used subcutaneous infusion sites. Mild local infusion site reactions are fairly common.
Formulating IVIg and SCIg is a complex process that entails plasma procurement and pooling, fractionation, and purification. It takes 10,000-60,000 plasma donations to make one lot of IVIg. Donations are accepted only from repeated donors. Samples are held for 6 months and tested for infectious agents. However, efforts are underway to develop bioengineered recombinant immune globulin products that don’t require donated plasma. These products are being designed to capture and enhance the most important mechanisms of benefit of plasma-derived immunoglobulins using Fc fragments that target key receptors, rather than relying on full-length immune globulin. The goal is enhanced efficacy at much lower doses than with IVIg or SCIg.
Dr. Postolova reported having no financial conflicts regarding her presentation.
Home-based subcutaneous immune globulin therapy is a promising alternative to intravenous immune globulin therapy for patients with refractory dermatomyositis or polymyositis, Anna Postolova, MD, MPH, declared at the 2021 Rheumatology Winter Clinical Symposium.
“This is really exciting. I think in the years to come we may see a change to having our patients be able to do immune globulin therapy at home,” said Dr. Postolova, a rheumatologist and allergist/immunologist at Stanford (Calif.) Health Care.
“The technology is there. I think our patients might feel more comfortable getting immune globulin at home,” she said. “I would love to switch more patients from IVIg to SCIg [subcutaneous immune globulin] in my practice.”
A few caveats: SCIg remains off label for treatment of dermatomyositis (DM) or polymyositis (PM). Its approved indication is as replacement therapy in patients with primary or secondary immunodeficiency diseases. IVIg is approved for this indication, but is also approved for DM/PM refractory to high-dose corticosteroids and immunosuppressants. Yet SCIg is clearly effective for these autoimmune inflammatory diseases, albeit to date the supporting evidence comes chiefly from observational studies and anecdotal experience.
“I don’t know if insurers will cover it, but they should because it’s obviously a lot cheaper to do it at home,” she noted.
SCIg advantages
SCIg offers compelling advantages over IVIg in addition to its substantially lower cost. These include far fewer systemic side effects, shorter infusion time, greater bioavailability, and better quality of life. Patients self-administer SCIg at home, avoiding the inconvenience of IVIg therapy, which entails travel time for once-monthly hospitalization or long hours spent in an infusion center, she explained.
French investigators recently documented a previously unappreciated further advantage of home-based SCIg. They convened a focus group of patients with DM or PM experienced with both IVIg and home SCIg and determined that participants uniformly preferred home SCIg. The patients cited a new and welcome feeling of autonomy and control.
“All patients with experience of IVIg and SCIg expressed a clear preference for SCIg, which was described to be easy, less disruptive for daily life, well tolerated, and less time-consuming. Preference was mainly related to a restoration of autonomy. Home-based self-administration reinforced the feeling of independence,” according to the investigators.
Available products
Six preparations of SCIg are commercially available. Most are in 10% concentration, as are all IVIg products. However, a 20% formulation of SCIg known as Hizentra allows for a smaller infusion volume and quicker completion of a treatment session. And one SCIg product, HyQvia, uses recombinant human hyaluronidase-facilitated 10% immune globulin, allowing home infusion of large volumes of sustained-release immune globulin on a once-monthly basis.
The relatively recent introduction of home SCIg for treatment of autoimmune inflammatory diseases, including DM, PM, and chronic inflammatory demyelinating polyneuropathy, has been pioneered mainly by European investigators. The treatment is often given by programmable mechanical pump once weekly. Italian investigators have reported efficacy in DM using 0.2 g/kg per week, which is about half the monthly total dose of IVIg employed. The infusion rate is 10-40 mL/hour, with a volume of around 35 mL per injection site.
Alternatively, SCIg can be delivered by rapid push infusions of smaller volumes with a syringe two or three times per week; that’s the regimen that was used at 2 g/kg over the course of a month by patients in the French focus group study, who didn’t mind the more frequent dosing.
“As they have had severe long-lasting symptoms, SCIg was perceived as a curative rather than a preventive therapy,” according to the French investigators.
More than 40% of patients experience adverse reactions to IVIg. These often involve headaches, nausea, back or abdominal pain, arthralgias, and/or difficulty breathing. Thromboembolic events and acute renal failure occur occasionally. For this reason, many physicians give a prophylactic dose of corticosteroids an hour before a patient’s first dose of IVIg. These systemic side effects are so rare with SCIg that Dr. Postolova has never pretreated with steroids, even though the main reason she resorts to the home therapy is a patient’s track record of poor tolerance of IVIg. The lower abdomen and thigh are the most commonly used subcutaneous infusion sites. Mild local infusion site reactions are fairly common.
Formulating IVIg and SCIg is a complex process that entails plasma procurement and pooling, fractionation, and purification. It takes 10,000-60,000 plasma donations to make one lot of IVIg. Donations are accepted only from repeated donors. Samples are held for 6 months and tested for infectious agents. However, efforts are underway to develop bioengineered recombinant immune globulin products that don’t require donated plasma. These products are being designed to capture and enhance the most important mechanisms of benefit of plasma-derived immunoglobulins using Fc fragments that target key receptors, rather than relying on full-length immune globulin. The goal is enhanced efficacy at much lower doses than with IVIg or SCIg.
Dr. Postolova reported having no financial conflicts regarding her presentation.
FROM RWCS 2021
Tocilizumab may improve lung function in early systemic sclerosis
Treatment with tocilizumab (Actemra) could stabilize or improve lung function in people with early interstitial lung disease associated with systemic sclerosis (SSc-ILD), a new study has found.
A paper published online Feb. 3 in Arthritis & Rheumatology presents the results of a post hoc analysis of data from a phase 3, placebo-controlled, double-blind trial of subcutaneous tocilizumab in patients with SSc and progressive skin disease, which included high-resolution chest CT to assess lung involvement and fibrosis.
Tocilizumab is a monoclonal antibody that targets interleukin-6 and is currently approved for the treatment of immune-mediated diseases such as rheumatoid arthritis, giant cell arteritis, cytokine release syndrome, and systemic and polyarticular course juvenile idiopathic arthritis.
Two previous studies of tocilizumab in patients with early, diffuse cutaneous SSc had also found that the treatment was associated with preservation of lung function but did not characterize that effect using radiography.
Of the 210 participants in the trial, called focuSSced, 136 were found to have interstitial lung disease at baseline and were randomized to 162 mg tocilizumab weekly or placebo for 48 weeks.
At baseline, around three-quarters of those with interstitial lung disease had moderate to severe lung involvement, defined as ground glass opacities, honeycombing, and fibrotic reticulation across at least 20% of the whole lung.
Those in the tocilizumab group showed a 0.1% mean decline in forced vital capacity (FVC) over the 48-week study, while those in the placebo group had a mean decline of 6.3%.
When stratified by severity of lung involvement, those with mild lung disease group treated with tocilizumab had a 4.1% decline in FVC, compared with a 10% decline in the placebo group; those with moderate disease in the treatment group had an 0.7% mean increase in FVC, compared with a 5.7% decrease in the placebo group, and those with severe lung involvement in the treatment arm had a 2.1% increase in FVC, compared with a 6.7% decrease in the placebo arm.
Those treated with tocilizumab also showed a statistically significant 1.8% improvement in the amount of lung involvement, which was largely seen in those with more extensive lung involvement at baseline. Those with more than 20% of the lung affected had a significant 4.9% reduction in lung area affected, while those in the placebo arm showed a significant increase in fibrosis.
First author David Roofeh, MD, of the University of Michigan Scleroderma Program, and colleagues wrote that most patients with SSc will develop interstitial lung disease – particularly those with early, diffuse cutaneous SSc and elevated markers such as C-reactive protein.
“Patients with these high-risk features, especially those with disease in the initial phase of development, represent an important target for early intervention as ILD is largely irreversible in SSc,” the authors wrote.
Findings from a specific patient population may not be generalizable
Commenting on the findings, Lorinda Chung, MD, of Stanford (Calif.) University, said in an interview that the study demonstrated that tocilizumab could prevent radiographic progression of ILD in early diffuse SSc patients with mild to severe lung disease and evidence of active skin disease, as well as elevated inflammatory markers.
“This was a very specific patient population who was studied in the focuSSced clinical trial, and this paper only evaluated a subset of these patients,” Dr. Chung said. “The results may not be generalizable to all SSc-ILD patients and further studies are needed.”
The authors suggested that the patients with progressive skin disease and elevated acute phase reactants may represent a group in the immunoinflammatory phase of the disease rather than the advanced fibrotic stage, and that this might be a “window of therapeutic opportunity to preserve lung function.”
Dr. Chung noted that the radiographic improvement induced by tocilizumab treatment was greatest in those with the most radiographic disease at baseline.
“This may reflect tocilizumab’s impact on decreasing inflammation, but we are not provided the data on the effects of tocilizumab on the individual components of the QILD [quantitative ILD: summation of ground glass opacities, honeycombing, and fibrotic reticulation],” she said.
The study’s authors also made a point about the utility of screening patients with high-resolution chest CT to detect early signs of ILD.
“Our data demonstrate the value of obtaining HRCT at the time of diagnosis: PFTs [pulmonary function tests] are not sensitive enough to accurately assess the presence of ILD and delays in treatment initiation may lead to irreversible disease,” they wrote.
Describing the results as ‘hypothesis-generating’ owing to the post hoc nature of the analysis, the authors said that FVC was an indirect measure of the flow-resistive properties of the lung, and that other aspects of SSc – such as hide-bound chest thickness – could cause thoracic restriction.
Two authors were funded by the National Institutes of Health. Six authors declared grants, funding, and other support from the pharmaceutical sector, including Roche, which sponsored the original focuSSced trial.
Treatment with tocilizumab (Actemra) could stabilize or improve lung function in people with early interstitial lung disease associated with systemic sclerosis (SSc-ILD), a new study has found.
A paper published online Feb. 3 in Arthritis & Rheumatology presents the results of a post hoc analysis of data from a phase 3, placebo-controlled, double-blind trial of subcutaneous tocilizumab in patients with SSc and progressive skin disease, which included high-resolution chest CT to assess lung involvement and fibrosis.
Tocilizumab is a monoclonal antibody that targets interleukin-6 and is currently approved for the treatment of immune-mediated diseases such as rheumatoid arthritis, giant cell arteritis, cytokine release syndrome, and systemic and polyarticular course juvenile idiopathic arthritis.
Two previous studies of tocilizumab in patients with early, diffuse cutaneous SSc had also found that the treatment was associated with preservation of lung function but did not characterize that effect using radiography.
Of the 210 participants in the trial, called focuSSced, 136 were found to have interstitial lung disease at baseline and were randomized to 162 mg tocilizumab weekly or placebo for 48 weeks.
At baseline, around three-quarters of those with interstitial lung disease had moderate to severe lung involvement, defined as ground glass opacities, honeycombing, and fibrotic reticulation across at least 20% of the whole lung.
Those in the tocilizumab group showed a 0.1% mean decline in forced vital capacity (FVC) over the 48-week study, while those in the placebo group had a mean decline of 6.3%.
When stratified by severity of lung involvement, those with mild lung disease group treated with tocilizumab had a 4.1% decline in FVC, compared with a 10% decline in the placebo group; those with moderate disease in the treatment group had an 0.7% mean increase in FVC, compared with a 5.7% decrease in the placebo group, and those with severe lung involvement in the treatment arm had a 2.1% increase in FVC, compared with a 6.7% decrease in the placebo arm.
Those treated with tocilizumab also showed a statistically significant 1.8% improvement in the amount of lung involvement, which was largely seen in those with more extensive lung involvement at baseline. Those with more than 20% of the lung affected had a significant 4.9% reduction in lung area affected, while those in the placebo arm showed a significant increase in fibrosis.
First author David Roofeh, MD, of the University of Michigan Scleroderma Program, and colleagues wrote that most patients with SSc will develop interstitial lung disease – particularly those with early, diffuse cutaneous SSc and elevated markers such as C-reactive protein.
“Patients with these high-risk features, especially those with disease in the initial phase of development, represent an important target for early intervention as ILD is largely irreversible in SSc,” the authors wrote.
Findings from a specific patient population may not be generalizable
Commenting on the findings, Lorinda Chung, MD, of Stanford (Calif.) University, said in an interview that the study demonstrated that tocilizumab could prevent radiographic progression of ILD in early diffuse SSc patients with mild to severe lung disease and evidence of active skin disease, as well as elevated inflammatory markers.
“This was a very specific patient population who was studied in the focuSSced clinical trial, and this paper only evaluated a subset of these patients,” Dr. Chung said. “The results may not be generalizable to all SSc-ILD patients and further studies are needed.”
The authors suggested that the patients with progressive skin disease and elevated acute phase reactants may represent a group in the immunoinflammatory phase of the disease rather than the advanced fibrotic stage, and that this might be a “window of therapeutic opportunity to preserve lung function.”
Dr. Chung noted that the radiographic improvement induced by tocilizumab treatment was greatest in those with the most radiographic disease at baseline.
“This may reflect tocilizumab’s impact on decreasing inflammation, but we are not provided the data on the effects of tocilizumab on the individual components of the QILD [quantitative ILD: summation of ground glass opacities, honeycombing, and fibrotic reticulation],” she said.
The study’s authors also made a point about the utility of screening patients with high-resolution chest CT to detect early signs of ILD.
“Our data demonstrate the value of obtaining HRCT at the time of diagnosis: PFTs [pulmonary function tests] are not sensitive enough to accurately assess the presence of ILD and delays in treatment initiation may lead to irreversible disease,” they wrote.
Describing the results as ‘hypothesis-generating’ owing to the post hoc nature of the analysis, the authors said that FVC was an indirect measure of the flow-resistive properties of the lung, and that other aspects of SSc – such as hide-bound chest thickness – could cause thoracic restriction.
Two authors were funded by the National Institutes of Health. Six authors declared grants, funding, and other support from the pharmaceutical sector, including Roche, which sponsored the original focuSSced trial.
Treatment with tocilizumab (Actemra) could stabilize or improve lung function in people with early interstitial lung disease associated with systemic sclerosis (SSc-ILD), a new study has found.
A paper published online Feb. 3 in Arthritis & Rheumatology presents the results of a post hoc analysis of data from a phase 3, placebo-controlled, double-blind trial of subcutaneous tocilizumab in patients with SSc and progressive skin disease, which included high-resolution chest CT to assess lung involvement and fibrosis.
Tocilizumab is a monoclonal antibody that targets interleukin-6 and is currently approved for the treatment of immune-mediated diseases such as rheumatoid arthritis, giant cell arteritis, cytokine release syndrome, and systemic and polyarticular course juvenile idiopathic arthritis.
Two previous studies of tocilizumab in patients with early, diffuse cutaneous SSc had also found that the treatment was associated with preservation of lung function but did not characterize that effect using radiography.
Of the 210 participants in the trial, called focuSSced, 136 were found to have interstitial lung disease at baseline and were randomized to 162 mg tocilizumab weekly or placebo for 48 weeks.
At baseline, around three-quarters of those with interstitial lung disease had moderate to severe lung involvement, defined as ground glass opacities, honeycombing, and fibrotic reticulation across at least 20% of the whole lung.
Those in the tocilizumab group showed a 0.1% mean decline in forced vital capacity (FVC) over the 48-week study, while those in the placebo group had a mean decline of 6.3%.
When stratified by severity of lung involvement, those with mild lung disease group treated with tocilizumab had a 4.1% decline in FVC, compared with a 10% decline in the placebo group; those with moderate disease in the treatment group had an 0.7% mean increase in FVC, compared with a 5.7% decrease in the placebo group, and those with severe lung involvement in the treatment arm had a 2.1% increase in FVC, compared with a 6.7% decrease in the placebo arm.
Those treated with tocilizumab also showed a statistically significant 1.8% improvement in the amount of lung involvement, which was largely seen in those with more extensive lung involvement at baseline. Those with more than 20% of the lung affected had a significant 4.9% reduction in lung area affected, while those in the placebo arm showed a significant increase in fibrosis.
First author David Roofeh, MD, of the University of Michigan Scleroderma Program, and colleagues wrote that most patients with SSc will develop interstitial lung disease – particularly those with early, diffuse cutaneous SSc and elevated markers such as C-reactive protein.
“Patients with these high-risk features, especially those with disease in the initial phase of development, represent an important target for early intervention as ILD is largely irreversible in SSc,” the authors wrote.
Findings from a specific patient population may not be generalizable
Commenting on the findings, Lorinda Chung, MD, of Stanford (Calif.) University, said in an interview that the study demonstrated that tocilizumab could prevent radiographic progression of ILD in early diffuse SSc patients with mild to severe lung disease and evidence of active skin disease, as well as elevated inflammatory markers.
“This was a very specific patient population who was studied in the focuSSced clinical trial, and this paper only evaluated a subset of these patients,” Dr. Chung said. “The results may not be generalizable to all SSc-ILD patients and further studies are needed.”
The authors suggested that the patients with progressive skin disease and elevated acute phase reactants may represent a group in the immunoinflammatory phase of the disease rather than the advanced fibrotic stage, and that this might be a “window of therapeutic opportunity to preserve lung function.”
Dr. Chung noted that the radiographic improvement induced by tocilizumab treatment was greatest in those with the most radiographic disease at baseline.
“This may reflect tocilizumab’s impact on decreasing inflammation, but we are not provided the data on the effects of tocilizumab on the individual components of the QILD [quantitative ILD: summation of ground glass opacities, honeycombing, and fibrotic reticulation],” she said.
The study’s authors also made a point about the utility of screening patients with high-resolution chest CT to detect early signs of ILD.
“Our data demonstrate the value of obtaining HRCT at the time of diagnosis: PFTs [pulmonary function tests] are not sensitive enough to accurately assess the presence of ILD and delays in treatment initiation may lead to irreversible disease,” they wrote.
Describing the results as ‘hypothesis-generating’ owing to the post hoc nature of the analysis, the authors said that FVC was an indirect measure of the flow-resistive properties of the lung, and that other aspects of SSc – such as hide-bound chest thickness – could cause thoracic restriction.
Two authors were funded by the National Institutes of Health. Six authors declared grants, funding, and other support from the pharmaceutical sector, including Roche, which sponsored the original focuSSced trial.
FROM ARTHRITIS & RHEUMATOLOGY