User login
What docs don’t know about the Disabilities Act can hurt them and patients
Lisa Iezzoni, MD, a professor of medicine at Harvard Medical School and a disability researcher at Massachusetts General Hospital, both in Boston, has used a wheelchair for more than 30 years because of multiple sclerosis. When she visits her primary care doctor, she doesn’t get weighed because the scales are not wheelchair accessible.
This failure to weigh her and other patients in wheelchairs could lead to serious medical problems. Weight is used to monitor a person’s overall health and prenatal health and to determine accurate doses for medications such as some chemotherapies, said Dr. Iezzoni.
In another situation, a man who used a wheelchair said that his primary care doctor never got him out of it for a complete physical exam. The patient later developed lymphoma, which first appeared in his groin. The doctor should have accommodated his disability and used a height-adjustable exam table or a portable lift to transfer him onto the table.
When physicians don’t provide access to medical care that patients with disabilities need, they put themselves at greater risk of lawsuits, fines, and settlements.
Yet, a new study in Health Affairs suggests that a large percentage of doctors are not fully aware of what they are legally required to do.
Under federal nondiscrimination laws (Americans With Disabilities Act, American Rehabilitation Act, and ADA Amendments Act), medical practices must provide equal access to people with disabilities, accommodate their disability-related needs, and not refuse them medical services because of their disabilities, say disability experts.
Where doctors go wrong with disability laws
What doctors don’t know about providing reasonable accommodations makes them vulnerable to lawsuits, which worries more than two-thirds of the 714 outpatient doctors surveyed.
Not only are they required to provide reasonable accommodations, but they also have to pay for them, the researchers said. One-fifth of the surveyed doctors said they didn’t know that practice owners have to pay.
More than one practice has made patients pay for services needed for their disability, such as sign language interpreters – the patients later complained this violated the ADA to enforcement agencies.
Doctors also don’t know that they have to collaborate with patients to determine what reasonable accommodations they need – over two-thirds of those surveyed said they didn’t know it was a joint responsibility, the study found.
When doctors fail to accommodate patients’ disability needs, they engage in discrimination and violate the ADA, says Elizabeth Pendo, JD, a coauthor of the study and the Joseph J. Simeone Professor of Law at Saint Louis University.
The Department of Justice has investigated several patient complaints of alleged disability discrimination recently and resolved the disputes with agreements and small fines in some cases. “The goal is not to get large financial settlements but to work with practices to get the correct procedures in place to be compliant,” said Ms. Pendo.
Physicians would be wise to check out whether their practices are as accessible as they think. Even if there’s a ramp to the office building, the parking lot may not have a van-accessible space or enough handicapped parking signs, or the exam room may be too narrow for a wheelchair to navigate.
These practices violated the ADA and agreed to make changes:
- Hamden, Conn., has two buildings that patients with physical disabilities couldn’t easily enter. The physician owners agreed to change the buildings’ entrances and access routes and add features to make it easier to use examination rooms and restrooms and the check-in and check-out areas.
- Seven medical offices in Riverside, Calif., failed to communicate effectively with deaf and hard-of-hearing patients. They should have had a qualified sign language interpreter, an assistive listening device, or another appropriate aid or service available to a deaf patient and her family. Instead, the office relied on a video remote interpretation system that often failed to work. The agreement requires the clinic to provide those aids and services to patients and their companions who are deaf or hard of hearing, advertise their availability, assess each patient who is deaf or hard of hearing to determine the best aids and services for their needs, and pay $5,000 in compensation to the complainant and a $1,000 civil penalty to the United States.
- Springfield, Mass., refused to provide full joint replacements to two patients being treated with buprenorphine, a medication used to treat opioid use disorder. Rather than accommodate the patients, the surgeons referred them elsewhere because they were uncomfortable with the postoperative pain management protocol for patients prescribed buprenorphine. “The Americans With Disabilities Act protects health care access for people under medical treatment for opioid use disorder,” said Acting U.S. Attorney Nathaniel R. Mendell. “Health care providers must comply with the ADA, even when doing so is inconvenient or makes them uncomfortable.” The agreement requires the practice to adopt a nondiscrimination policy, provide training on the ADA and opioid use disorder, and pay two complainants $15,000 each for pain and suffering.
The DOJ has filed civil lawsuits against medical practices when they failed to resolve the allegations. Recent cases include an ophthalmology practice with 24 facilities in Arizona that refused to help transfer patients in wheelchairs to surgery tables for eye surgery and required them to pay for transfer support services and two obstetricians-gynecologists in Bakersfield, Calif., who refused to provide routine medical care to a patient because of her HIV status.
What doctors should know
Many people tend to think of a person with a disability as being in a wheelchair. But the ADA has a very broad definition of disability, which includes any physical or mental impairment that substantially limits any major life activity, said Ms. Pendo.
“It was amended in 2008 to clarify that the definition includes people with chronic diseases such as diabetes and cancer, cognitive and neurological disorders, substance abuse disorders, vision and hearing loss, and learning and other disabilities,” she said.
That means that doctors have to accommodate many types of disabilities, which can be challenging. The ADA only specifies that fixed structures need to be accessible, such as parking lots, driveways, and buildings, said Dr. Iezzoni.
When it comes to “reasonable accommodations,” doctors should decide that on a case-by-case basis, she said.
“We can say based on our study that 71% of doctors don’t know the right way to think about the accommodations – they don’t know they need to talk to patients so they can explain to them exactly what they need to accommodate their disability,” said Dr. Iezzoni.
Doctors are also required to provide effective communication for patients with sensory or cognitive disabilities, which can depend on the severity, said Ms. Pendo. Is the person deaf or hard of hearing, blind or partially sighted – is the dementia mild or severe?
“The requirement is there, but what that looks like will vary by patient. That’s what’s challenging,” said Ms. Pendo.
Dr. Iezzoni recommends that doctor’s offices ask patients whether they need special help or individual assistance when they make appointments and enter their responses in their records. She also suggests that patients be asked at follow-up appointments whether they still need the same help or not.
“Disabilities can change over time – a person with bad arthritis may need help getting onto an exam table, but later get a knee or hip replacement that is effective and no longer need that help,” said Dr. Iezzoni.
Benefits outweigh costs
Physicians have made progress in meeting the ADA’s physical accessibility requirements, said Dr. Iezzoni. “The literature suggests that doctors have done a good job at fixing the structural barriers people with mobility issues face, such as ramps and bathrooms.”
However, there are exceptions in rural older buildings which can be harder to retrofit for wheelchair accessibility, she said. “I recall interviewing a rural doctor several years ago who said that he knew his patients well and when a patient visits with mobility problems, he goes down and carries the patient up the steps to his office. My response was that is not respectful of the patient or safe for the patient or you. That doctor has since changed the location of his practice,” said Dr. Iezzoni.
Some doctors may resist paying for accessible medical equipment because of cost, but she said the benefits are worth it. These include preventing staff injuries when they transfer patients and being used by patients with temporary disabilities and aging people with bad knees, backs, hearing and sight. In addition, businesses may be eligible for federal and state tax credits.
Dr. Iezzoni recently visited her doctor where they finally got height-adjustable exam tables. “I asked the assistant, who really likes these tables? She said it’s the elderly ladies of short stature – the table is lowered and they sit down and get on it.”
But, Dr. Iezonni’s main message to doctors is that patients with disabilities deserve equal quality of care. “Just because we have a disability doesn’t mean we should get worse care than other people. It’s a matter of professionalism that doctors should want to give the same quality care to all their patients.”
A version of this article first appeared on Medscape.com.
Lisa Iezzoni, MD, a professor of medicine at Harvard Medical School and a disability researcher at Massachusetts General Hospital, both in Boston, has used a wheelchair for more than 30 years because of multiple sclerosis. When she visits her primary care doctor, she doesn’t get weighed because the scales are not wheelchair accessible.
This failure to weigh her and other patients in wheelchairs could lead to serious medical problems. Weight is used to monitor a person’s overall health and prenatal health and to determine accurate doses for medications such as some chemotherapies, said Dr. Iezzoni.
In another situation, a man who used a wheelchair said that his primary care doctor never got him out of it for a complete physical exam. The patient later developed lymphoma, which first appeared in his groin. The doctor should have accommodated his disability and used a height-adjustable exam table or a portable lift to transfer him onto the table.
When physicians don’t provide access to medical care that patients with disabilities need, they put themselves at greater risk of lawsuits, fines, and settlements.
Yet, a new study in Health Affairs suggests that a large percentage of doctors are not fully aware of what they are legally required to do.
Under federal nondiscrimination laws (Americans With Disabilities Act, American Rehabilitation Act, and ADA Amendments Act), medical practices must provide equal access to people with disabilities, accommodate their disability-related needs, and not refuse them medical services because of their disabilities, say disability experts.
Where doctors go wrong with disability laws
What doctors don’t know about providing reasonable accommodations makes them vulnerable to lawsuits, which worries more than two-thirds of the 714 outpatient doctors surveyed.
Not only are they required to provide reasonable accommodations, but they also have to pay for them, the researchers said. One-fifth of the surveyed doctors said they didn’t know that practice owners have to pay.
More than one practice has made patients pay for services needed for their disability, such as sign language interpreters – the patients later complained this violated the ADA to enforcement agencies.
Doctors also don’t know that they have to collaborate with patients to determine what reasonable accommodations they need – over two-thirds of those surveyed said they didn’t know it was a joint responsibility, the study found.
When doctors fail to accommodate patients’ disability needs, they engage in discrimination and violate the ADA, says Elizabeth Pendo, JD, a coauthor of the study and the Joseph J. Simeone Professor of Law at Saint Louis University.
The Department of Justice has investigated several patient complaints of alleged disability discrimination recently and resolved the disputes with agreements and small fines in some cases. “The goal is not to get large financial settlements but to work with practices to get the correct procedures in place to be compliant,” said Ms. Pendo.
Physicians would be wise to check out whether their practices are as accessible as they think. Even if there’s a ramp to the office building, the parking lot may not have a van-accessible space or enough handicapped parking signs, or the exam room may be too narrow for a wheelchair to navigate.
These practices violated the ADA and agreed to make changes:
- Hamden, Conn., has two buildings that patients with physical disabilities couldn’t easily enter. The physician owners agreed to change the buildings’ entrances and access routes and add features to make it easier to use examination rooms and restrooms and the check-in and check-out areas.
- Seven medical offices in Riverside, Calif., failed to communicate effectively with deaf and hard-of-hearing patients. They should have had a qualified sign language interpreter, an assistive listening device, or another appropriate aid or service available to a deaf patient and her family. Instead, the office relied on a video remote interpretation system that often failed to work. The agreement requires the clinic to provide those aids and services to patients and their companions who are deaf or hard of hearing, advertise their availability, assess each patient who is deaf or hard of hearing to determine the best aids and services for their needs, and pay $5,000 in compensation to the complainant and a $1,000 civil penalty to the United States.
- Springfield, Mass., refused to provide full joint replacements to two patients being treated with buprenorphine, a medication used to treat opioid use disorder. Rather than accommodate the patients, the surgeons referred them elsewhere because they were uncomfortable with the postoperative pain management protocol for patients prescribed buprenorphine. “The Americans With Disabilities Act protects health care access for people under medical treatment for opioid use disorder,” said Acting U.S. Attorney Nathaniel R. Mendell. “Health care providers must comply with the ADA, even when doing so is inconvenient or makes them uncomfortable.” The agreement requires the practice to adopt a nondiscrimination policy, provide training on the ADA and opioid use disorder, and pay two complainants $15,000 each for pain and suffering.
The DOJ has filed civil lawsuits against medical practices when they failed to resolve the allegations. Recent cases include an ophthalmology practice with 24 facilities in Arizona that refused to help transfer patients in wheelchairs to surgery tables for eye surgery and required them to pay for transfer support services and two obstetricians-gynecologists in Bakersfield, Calif., who refused to provide routine medical care to a patient because of her HIV status.
What doctors should know
Many people tend to think of a person with a disability as being in a wheelchair. But the ADA has a very broad definition of disability, which includes any physical or mental impairment that substantially limits any major life activity, said Ms. Pendo.
“It was amended in 2008 to clarify that the definition includes people with chronic diseases such as diabetes and cancer, cognitive and neurological disorders, substance abuse disorders, vision and hearing loss, and learning and other disabilities,” she said.
That means that doctors have to accommodate many types of disabilities, which can be challenging. The ADA only specifies that fixed structures need to be accessible, such as parking lots, driveways, and buildings, said Dr. Iezzoni.
When it comes to “reasonable accommodations,” doctors should decide that on a case-by-case basis, she said.
“We can say based on our study that 71% of doctors don’t know the right way to think about the accommodations – they don’t know they need to talk to patients so they can explain to them exactly what they need to accommodate their disability,” said Dr. Iezzoni.
Doctors are also required to provide effective communication for patients with sensory or cognitive disabilities, which can depend on the severity, said Ms. Pendo. Is the person deaf or hard of hearing, blind or partially sighted – is the dementia mild or severe?
“The requirement is there, but what that looks like will vary by patient. That’s what’s challenging,” said Ms. Pendo.
Dr. Iezzoni recommends that doctor’s offices ask patients whether they need special help or individual assistance when they make appointments and enter their responses in their records. She also suggests that patients be asked at follow-up appointments whether they still need the same help or not.
“Disabilities can change over time – a person with bad arthritis may need help getting onto an exam table, but later get a knee or hip replacement that is effective and no longer need that help,” said Dr. Iezzoni.
Benefits outweigh costs
Physicians have made progress in meeting the ADA’s physical accessibility requirements, said Dr. Iezzoni. “The literature suggests that doctors have done a good job at fixing the structural barriers people with mobility issues face, such as ramps and bathrooms.”
However, there are exceptions in rural older buildings which can be harder to retrofit for wheelchair accessibility, she said. “I recall interviewing a rural doctor several years ago who said that he knew his patients well and when a patient visits with mobility problems, he goes down and carries the patient up the steps to his office. My response was that is not respectful of the patient or safe for the patient or you. That doctor has since changed the location of his practice,” said Dr. Iezzoni.
Some doctors may resist paying for accessible medical equipment because of cost, but she said the benefits are worth it. These include preventing staff injuries when they transfer patients and being used by patients with temporary disabilities and aging people with bad knees, backs, hearing and sight. In addition, businesses may be eligible for federal and state tax credits.
Dr. Iezzoni recently visited her doctor where they finally got height-adjustable exam tables. “I asked the assistant, who really likes these tables? She said it’s the elderly ladies of short stature – the table is lowered and they sit down and get on it.”
But, Dr. Iezonni’s main message to doctors is that patients with disabilities deserve equal quality of care. “Just because we have a disability doesn’t mean we should get worse care than other people. It’s a matter of professionalism that doctors should want to give the same quality care to all their patients.”
A version of this article first appeared on Medscape.com.
Lisa Iezzoni, MD, a professor of medicine at Harvard Medical School and a disability researcher at Massachusetts General Hospital, both in Boston, has used a wheelchair for more than 30 years because of multiple sclerosis. When she visits her primary care doctor, she doesn’t get weighed because the scales are not wheelchair accessible.
This failure to weigh her and other patients in wheelchairs could lead to serious medical problems. Weight is used to monitor a person’s overall health and prenatal health and to determine accurate doses for medications such as some chemotherapies, said Dr. Iezzoni.
In another situation, a man who used a wheelchair said that his primary care doctor never got him out of it for a complete physical exam. The patient later developed lymphoma, which first appeared in his groin. The doctor should have accommodated his disability and used a height-adjustable exam table or a portable lift to transfer him onto the table.
When physicians don’t provide access to medical care that patients with disabilities need, they put themselves at greater risk of lawsuits, fines, and settlements.
Yet, a new study in Health Affairs suggests that a large percentage of doctors are not fully aware of what they are legally required to do.
Under federal nondiscrimination laws (Americans With Disabilities Act, American Rehabilitation Act, and ADA Amendments Act), medical practices must provide equal access to people with disabilities, accommodate their disability-related needs, and not refuse them medical services because of their disabilities, say disability experts.
Where doctors go wrong with disability laws
What doctors don’t know about providing reasonable accommodations makes them vulnerable to lawsuits, which worries more than two-thirds of the 714 outpatient doctors surveyed.
Not only are they required to provide reasonable accommodations, but they also have to pay for them, the researchers said. One-fifth of the surveyed doctors said they didn’t know that practice owners have to pay.
More than one practice has made patients pay for services needed for their disability, such as sign language interpreters – the patients later complained this violated the ADA to enforcement agencies.
Doctors also don’t know that they have to collaborate with patients to determine what reasonable accommodations they need – over two-thirds of those surveyed said they didn’t know it was a joint responsibility, the study found.
When doctors fail to accommodate patients’ disability needs, they engage in discrimination and violate the ADA, says Elizabeth Pendo, JD, a coauthor of the study and the Joseph J. Simeone Professor of Law at Saint Louis University.
The Department of Justice has investigated several patient complaints of alleged disability discrimination recently and resolved the disputes with agreements and small fines in some cases. “The goal is not to get large financial settlements but to work with practices to get the correct procedures in place to be compliant,” said Ms. Pendo.
Physicians would be wise to check out whether their practices are as accessible as they think. Even if there’s a ramp to the office building, the parking lot may not have a van-accessible space or enough handicapped parking signs, or the exam room may be too narrow for a wheelchair to navigate.
These practices violated the ADA and agreed to make changes:
- Hamden, Conn., has two buildings that patients with physical disabilities couldn’t easily enter. The physician owners agreed to change the buildings’ entrances and access routes and add features to make it easier to use examination rooms and restrooms and the check-in and check-out areas.
- Seven medical offices in Riverside, Calif., failed to communicate effectively with deaf and hard-of-hearing patients. They should have had a qualified sign language interpreter, an assistive listening device, or another appropriate aid or service available to a deaf patient and her family. Instead, the office relied on a video remote interpretation system that often failed to work. The agreement requires the clinic to provide those aids and services to patients and their companions who are deaf or hard of hearing, advertise their availability, assess each patient who is deaf or hard of hearing to determine the best aids and services for their needs, and pay $5,000 in compensation to the complainant and a $1,000 civil penalty to the United States.
- Springfield, Mass., refused to provide full joint replacements to two patients being treated with buprenorphine, a medication used to treat opioid use disorder. Rather than accommodate the patients, the surgeons referred them elsewhere because they were uncomfortable with the postoperative pain management protocol for patients prescribed buprenorphine. “The Americans With Disabilities Act protects health care access for people under medical treatment for opioid use disorder,” said Acting U.S. Attorney Nathaniel R. Mendell. “Health care providers must comply with the ADA, even when doing so is inconvenient or makes them uncomfortable.” The agreement requires the practice to adopt a nondiscrimination policy, provide training on the ADA and opioid use disorder, and pay two complainants $15,000 each for pain and suffering.
The DOJ has filed civil lawsuits against medical practices when they failed to resolve the allegations. Recent cases include an ophthalmology practice with 24 facilities in Arizona that refused to help transfer patients in wheelchairs to surgery tables for eye surgery and required them to pay for transfer support services and two obstetricians-gynecologists in Bakersfield, Calif., who refused to provide routine medical care to a patient because of her HIV status.
What doctors should know
Many people tend to think of a person with a disability as being in a wheelchair. But the ADA has a very broad definition of disability, which includes any physical or mental impairment that substantially limits any major life activity, said Ms. Pendo.
“It was amended in 2008 to clarify that the definition includes people with chronic diseases such as diabetes and cancer, cognitive and neurological disorders, substance abuse disorders, vision and hearing loss, and learning and other disabilities,” she said.
That means that doctors have to accommodate many types of disabilities, which can be challenging. The ADA only specifies that fixed structures need to be accessible, such as parking lots, driveways, and buildings, said Dr. Iezzoni.
When it comes to “reasonable accommodations,” doctors should decide that on a case-by-case basis, she said.
“We can say based on our study that 71% of doctors don’t know the right way to think about the accommodations – they don’t know they need to talk to patients so they can explain to them exactly what they need to accommodate their disability,” said Dr. Iezzoni.
Doctors are also required to provide effective communication for patients with sensory or cognitive disabilities, which can depend on the severity, said Ms. Pendo. Is the person deaf or hard of hearing, blind or partially sighted – is the dementia mild or severe?
“The requirement is there, but what that looks like will vary by patient. That’s what’s challenging,” said Ms. Pendo.
Dr. Iezzoni recommends that doctor’s offices ask patients whether they need special help or individual assistance when they make appointments and enter their responses in their records. She also suggests that patients be asked at follow-up appointments whether they still need the same help or not.
“Disabilities can change over time – a person with bad arthritis may need help getting onto an exam table, but later get a knee or hip replacement that is effective and no longer need that help,” said Dr. Iezzoni.
Benefits outweigh costs
Physicians have made progress in meeting the ADA’s physical accessibility requirements, said Dr. Iezzoni. “The literature suggests that doctors have done a good job at fixing the structural barriers people with mobility issues face, such as ramps and bathrooms.”
However, there are exceptions in rural older buildings which can be harder to retrofit for wheelchair accessibility, she said. “I recall interviewing a rural doctor several years ago who said that he knew his patients well and when a patient visits with mobility problems, he goes down and carries the patient up the steps to his office. My response was that is not respectful of the patient or safe for the patient or you. That doctor has since changed the location of his practice,” said Dr. Iezzoni.
Some doctors may resist paying for accessible medical equipment because of cost, but she said the benefits are worth it. These include preventing staff injuries when they transfer patients and being used by patients with temporary disabilities and aging people with bad knees, backs, hearing and sight. In addition, businesses may be eligible for federal and state tax credits.
Dr. Iezzoni recently visited her doctor where they finally got height-adjustable exam tables. “I asked the assistant, who really likes these tables? She said it’s the elderly ladies of short stature – the table is lowered and they sit down and get on it.”
But, Dr. Iezonni’s main message to doctors is that patients with disabilities deserve equal quality of care. “Just because we have a disability doesn’t mean we should get worse care than other people. It’s a matter of professionalism that doctors should want to give the same quality care to all their patients.”
A version of this article first appeared on Medscape.com.
Ways to make sure 2022 doesn’t stink for docs
Depending on the data you’re looking at, 40%-60% of physicians are burned out.
Research studies and the eye test reveal the painfully obvious: Colleagues are tired, winded, spent, and at times way past burned out. People aren’t asking me if they’re burned out. They know they’re burned out; heck, they can even recite the Maslach burnout inventory, forward and backward, in a mask, or while completing a COVID quarantine. A fair share of people know the key steps to prevent burnout and promote recovery.
What I’m starting to see more of is, “Why should I even bother to recover from this? Why pick myself up again just to get another occupational stress injury (burnout, demoralization, moral injury, etc.)?” In other words, it’s not just simply about negating burnout; it’s about supporting and facilitating the motivation to work.
We’ve been through so much with COVID that it might be challenging to remember when you saw a truly engaged work environment. No doubt, we have outstanding professionals across medicine who answer the bell every day. However, if you’ve been looking closely, many teams/units have lost a bit of the zip and pep. The synergy and trust aren’t as smooth, and at noon, everyone counts the hours to the end of the shift.
You may be thinking, Well, of course, they are; we’re still amid a pandemic, and people have been through hell. Your observation would be correct, except I’ve personally seen some teams weather the pandemic storm and still remain engaged (some even more involved).
The No. 1 consult result for the GW Resiliency and Well-Being Center, where I work, has been on lectures for burnout. The R&WC has given so many of these lectures that my dreams take the form of a PowerPoint presentation. Overall the talks have gone very well. We’ve added skills sections on practices of whole-person care. We’ve blitzed the daylights out of restorative sleep, yet I know we are still searching for the correct narrative.
Motivated staff, faculty, and students will genuinely take in the information and follow the recommendations; however, they still struggle to find that drive and zest for work. Yes, moving from burnout to neutral is reasonable but likely won’t move the needle of your professional or personal life. We need to have the emotional energy and the clear desire to utilize that energy for a meaningful purpose.
Talking about burnout in specific ways is straightforward and, in my opinion, much easier than talking about engagement. Part of the challenge when trying to discuss engagement is that people can feel invalidated or that you’re telling them to be stoic. Or worse yet, that the problem of burnout primarily lies with them. It’s essential to recognize the role of an organizational factor in burnout (approximately 80%, depending on the study); still, even if you address burnout, people may not be miserable, but it doesn’t mean they will stay at their current job (please cue intro music for the Great Resignation).
Engagement models have existed for some time and certainly have gained much more attention in health care settings over the past 2 decades. Engagement can be described as having three components: dedication, vigor, and absorption. When a person is filling all three of these components over time, presto – you get the much-sought-after state of the supremely engaged professional.
These models definitely give us excellent starting points to approach engagement from a pre-COVID era. In COVID and beyond, I’m not sure how these models will stand up in a hybrid work environment, where autonomy and flexibility could be more valued than ever. Personally, COVID revealed some things I was missing in my work pre-COVID:
- Time to think and process. This was one of the great things about being a consultation-liaison psychiatrist; it was literally feast or famine.
- Doing what I’m talented at and really enjoy.
- Time is short, and I want to be more present in the life of my family.
The list above isn’t exhaustive, but I’ve found them to be my own personal recipe for being engaged. Over the next series of articles, I’m going to focus on engagement and factors related to key resilience. These articles will be informed by a front-line view from my colleagues, and hopefully start to separate the myth from reality on the subject of health professional engagement and resilience.
Everyone be safe and well!
A version of this article first appeared on Medscape.com.
Depending on the data you’re looking at, 40%-60% of physicians are burned out.
Research studies and the eye test reveal the painfully obvious: Colleagues are tired, winded, spent, and at times way past burned out. People aren’t asking me if they’re burned out. They know they’re burned out; heck, they can even recite the Maslach burnout inventory, forward and backward, in a mask, or while completing a COVID quarantine. A fair share of people know the key steps to prevent burnout and promote recovery.
What I’m starting to see more of is, “Why should I even bother to recover from this? Why pick myself up again just to get another occupational stress injury (burnout, demoralization, moral injury, etc.)?” In other words, it’s not just simply about negating burnout; it’s about supporting and facilitating the motivation to work.
We’ve been through so much with COVID that it might be challenging to remember when you saw a truly engaged work environment. No doubt, we have outstanding professionals across medicine who answer the bell every day. However, if you’ve been looking closely, many teams/units have lost a bit of the zip and pep. The synergy and trust aren’t as smooth, and at noon, everyone counts the hours to the end of the shift.
You may be thinking, Well, of course, they are; we’re still amid a pandemic, and people have been through hell. Your observation would be correct, except I’ve personally seen some teams weather the pandemic storm and still remain engaged (some even more involved).
The No. 1 consult result for the GW Resiliency and Well-Being Center, where I work, has been on lectures for burnout. The R&WC has given so many of these lectures that my dreams take the form of a PowerPoint presentation. Overall the talks have gone very well. We’ve added skills sections on practices of whole-person care. We’ve blitzed the daylights out of restorative sleep, yet I know we are still searching for the correct narrative.
Motivated staff, faculty, and students will genuinely take in the information and follow the recommendations; however, they still struggle to find that drive and zest for work. Yes, moving from burnout to neutral is reasonable but likely won’t move the needle of your professional or personal life. We need to have the emotional energy and the clear desire to utilize that energy for a meaningful purpose.
Talking about burnout in specific ways is straightforward and, in my opinion, much easier than talking about engagement. Part of the challenge when trying to discuss engagement is that people can feel invalidated or that you’re telling them to be stoic. Or worse yet, that the problem of burnout primarily lies with them. It’s essential to recognize the role of an organizational factor in burnout (approximately 80%, depending on the study); still, even if you address burnout, people may not be miserable, but it doesn’t mean they will stay at their current job (please cue intro music for the Great Resignation).
Engagement models have existed for some time and certainly have gained much more attention in health care settings over the past 2 decades. Engagement can be described as having three components: dedication, vigor, and absorption. When a person is filling all three of these components over time, presto – you get the much-sought-after state of the supremely engaged professional.
These models definitely give us excellent starting points to approach engagement from a pre-COVID era. In COVID and beyond, I’m not sure how these models will stand up in a hybrid work environment, where autonomy and flexibility could be more valued than ever. Personally, COVID revealed some things I was missing in my work pre-COVID:
- Time to think and process. This was one of the great things about being a consultation-liaison psychiatrist; it was literally feast or famine.
- Doing what I’m talented at and really enjoy.
- Time is short, and I want to be more present in the life of my family.
The list above isn’t exhaustive, but I’ve found them to be my own personal recipe for being engaged. Over the next series of articles, I’m going to focus on engagement and factors related to key resilience. These articles will be informed by a front-line view from my colleagues, and hopefully start to separate the myth from reality on the subject of health professional engagement and resilience.
Everyone be safe and well!
A version of this article first appeared on Medscape.com.
Depending on the data you’re looking at, 40%-60% of physicians are burned out.
Research studies and the eye test reveal the painfully obvious: Colleagues are tired, winded, spent, and at times way past burned out. People aren’t asking me if they’re burned out. They know they’re burned out; heck, they can even recite the Maslach burnout inventory, forward and backward, in a mask, or while completing a COVID quarantine. A fair share of people know the key steps to prevent burnout and promote recovery.
What I’m starting to see more of is, “Why should I even bother to recover from this? Why pick myself up again just to get another occupational stress injury (burnout, demoralization, moral injury, etc.)?” In other words, it’s not just simply about negating burnout; it’s about supporting and facilitating the motivation to work.
We’ve been through so much with COVID that it might be challenging to remember when you saw a truly engaged work environment. No doubt, we have outstanding professionals across medicine who answer the bell every day. However, if you’ve been looking closely, many teams/units have lost a bit of the zip and pep. The synergy and trust aren’t as smooth, and at noon, everyone counts the hours to the end of the shift.
You may be thinking, Well, of course, they are; we’re still amid a pandemic, and people have been through hell. Your observation would be correct, except I’ve personally seen some teams weather the pandemic storm and still remain engaged (some even more involved).
The No. 1 consult result for the GW Resiliency and Well-Being Center, where I work, has been on lectures for burnout. The R&WC has given so many of these lectures that my dreams take the form of a PowerPoint presentation. Overall the talks have gone very well. We’ve added skills sections on practices of whole-person care. We’ve blitzed the daylights out of restorative sleep, yet I know we are still searching for the correct narrative.
Motivated staff, faculty, and students will genuinely take in the information and follow the recommendations; however, they still struggle to find that drive and zest for work. Yes, moving from burnout to neutral is reasonable but likely won’t move the needle of your professional or personal life. We need to have the emotional energy and the clear desire to utilize that energy for a meaningful purpose.
Talking about burnout in specific ways is straightforward and, in my opinion, much easier than talking about engagement. Part of the challenge when trying to discuss engagement is that people can feel invalidated or that you’re telling them to be stoic. Or worse yet, that the problem of burnout primarily lies with them. It’s essential to recognize the role of an organizational factor in burnout (approximately 80%, depending on the study); still, even if you address burnout, people may not be miserable, but it doesn’t mean they will stay at their current job (please cue intro music for the Great Resignation).
Engagement models have existed for some time and certainly have gained much more attention in health care settings over the past 2 decades. Engagement can be described as having three components: dedication, vigor, and absorption. When a person is filling all three of these components over time, presto – you get the much-sought-after state of the supremely engaged professional.
These models definitely give us excellent starting points to approach engagement from a pre-COVID era. In COVID and beyond, I’m not sure how these models will stand up in a hybrid work environment, where autonomy and flexibility could be more valued than ever. Personally, COVID revealed some things I was missing in my work pre-COVID:
- Time to think and process. This was one of the great things about being a consultation-liaison psychiatrist; it was literally feast or famine.
- Doing what I’m talented at and really enjoy.
- Time is short, and I want to be more present in the life of my family.
The list above isn’t exhaustive, but I’ve found them to be my own personal recipe for being engaged. Over the next series of articles, I’m going to focus on engagement and factors related to key resilience. These articles will be informed by a front-line view from my colleagues, and hopefully start to separate the myth from reality on the subject of health professional engagement and resilience.
Everyone be safe and well!
A version of this article first appeared on Medscape.com.
Keeping an open mind about functional medicine
Considering the controversy surrounding functional medicine, you may be wondering why JFP published an article about it last month.1 David Gorski, MD, PhD, FACS, a vocal critic of functional medicine, commented: “Functional medicine. It sounds so … scientific and reasonable. It’s anything but. In fact, functional medicine combines the worst features of conventional medicine with a heapin’ helpin’ of quackery.”2 On its website, however, The Institute for Functional Medicine claims that “functional medicine determines how and why illness occurs and restores health by addressing the root causes of disease for each individual.”3
I suspect the truth lies somewhere in between.
Because functional medicine has gained a certain degree of popularity, I felt it was important for family physicians and other primary care clinicians to know enough about this alternative healing method to discuss it with patients who express interest.
In their review article in JFP, Orlando and colleagues tell us there are 7 defining characteristics of functional medicine.1 It is patient centered rather than disease centered, uses a “systems biology” approach, considers the dynamic balance of gene-environment interactions, is personalized based on biochemical individuality, promotes organ reserve and sustained health span, sees health as a positive vitality (not merely the absence of disease), and focuses on function rather than pathology.
Most of these statements about functional medicine apply to traditional family medicine. The clinical approach stressing lifestyle changes is mainstream, not unique. The focus on digestion and the microbiome as an important determinant of health is based on interesting basic science studies and associations noted between certain microbiome profiles and diseases.
But association is not causation. So far there is scant evidence that changing the microbiome results in better health, although some preliminary case series have generated intriguing hypotheses. And there is evidence that probiotics improve some symptoms. Ongoing research into the microbiome and health will, no doubt, be illuminating. We have much to learn.
What does seem unique, but suspect, about functional medicine is its focus on biochemical testing of unproven value and the prescribing of diets and supplements based on the test results. There are no sound scientific studies showing the benefit of this approach.
I suggest you read Orlando et al’s article. Functional medicine is an interesting, mostly unproven, approach to patient care. But I will keep an open mind until we see better research that either does—or doesn’t—support the validity of its practices.
1. Orlando FA, Chang KL, Estores IM. Functional medicine: focusing on imbalances in core metabolic processes. J Fam Pract. 2021;70:482-488,498.
2. Gorski D. Functional medicine: the ultimate misnomer in the world of integrative medicine. Science-Based Medicine. April 11, 2016. Accessed January 4, 2022. https://sciencebasedmedicine.org/functional-medicine-the-ultimate-misnomer-in-the-world-of-integrative-medicine/
3. The Institute for Functional Medicine. Accessed January 4, 2022. www.ifm.org
Considering the controversy surrounding functional medicine, you may be wondering why JFP published an article about it last month.1 David Gorski, MD, PhD, FACS, a vocal critic of functional medicine, commented: “Functional medicine. It sounds so … scientific and reasonable. It’s anything but. In fact, functional medicine combines the worst features of conventional medicine with a heapin’ helpin’ of quackery.”2 On its website, however, The Institute for Functional Medicine claims that “functional medicine determines how and why illness occurs and restores health by addressing the root causes of disease for each individual.”3
I suspect the truth lies somewhere in between.
Because functional medicine has gained a certain degree of popularity, I felt it was important for family physicians and other primary care clinicians to know enough about this alternative healing method to discuss it with patients who express interest.
In their review article in JFP, Orlando and colleagues tell us there are 7 defining characteristics of functional medicine.1 It is patient centered rather than disease centered, uses a “systems biology” approach, considers the dynamic balance of gene-environment interactions, is personalized based on biochemical individuality, promotes organ reserve and sustained health span, sees health as a positive vitality (not merely the absence of disease), and focuses on function rather than pathology.
Most of these statements about functional medicine apply to traditional family medicine. The clinical approach stressing lifestyle changes is mainstream, not unique. The focus on digestion and the microbiome as an important determinant of health is based on interesting basic science studies and associations noted between certain microbiome profiles and diseases.
But association is not causation. So far there is scant evidence that changing the microbiome results in better health, although some preliminary case series have generated intriguing hypotheses. And there is evidence that probiotics improve some symptoms. Ongoing research into the microbiome and health will, no doubt, be illuminating. We have much to learn.
What does seem unique, but suspect, about functional medicine is its focus on biochemical testing of unproven value and the prescribing of diets and supplements based on the test results. There are no sound scientific studies showing the benefit of this approach.
I suggest you read Orlando et al’s article. Functional medicine is an interesting, mostly unproven, approach to patient care. But I will keep an open mind until we see better research that either does—or doesn’t—support the validity of its practices.
Considering the controversy surrounding functional medicine, you may be wondering why JFP published an article about it last month.1 David Gorski, MD, PhD, FACS, a vocal critic of functional medicine, commented: “Functional medicine. It sounds so … scientific and reasonable. It’s anything but. In fact, functional medicine combines the worst features of conventional medicine with a heapin’ helpin’ of quackery.”2 On its website, however, The Institute for Functional Medicine claims that “functional medicine determines how and why illness occurs and restores health by addressing the root causes of disease for each individual.”3
I suspect the truth lies somewhere in between.
Because functional medicine has gained a certain degree of popularity, I felt it was important for family physicians and other primary care clinicians to know enough about this alternative healing method to discuss it with patients who express interest.
In their review article in JFP, Orlando and colleagues tell us there are 7 defining characteristics of functional medicine.1 It is patient centered rather than disease centered, uses a “systems biology” approach, considers the dynamic balance of gene-environment interactions, is personalized based on biochemical individuality, promotes organ reserve and sustained health span, sees health as a positive vitality (not merely the absence of disease), and focuses on function rather than pathology.
Most of these statements about functional medicine apply to traditional family medicine. The clinical approach stressing lifestyle changes is mainstream, not unique. The focus on digestion and the microbiome as an important determinant of health is based on interesting basic science studies and associations noted between certain microbiome profiles and diseases.
But association is not causation. So far there is scant evidence that changing the microbiome results in better health, although some preliminary case series have generated intriguing hypotheses. And there is evidence that probiotics improve some symptoms. Ongoing research into the microbiome and health will, no doubt, be illuminating. We have much to learn.
What does seem unique, but suspect, about functional medicine is its focus on biochemical testing of unproven value and the prescribing of diets and supplements based on the test results. There are no sound scientific studies showing the benefit of this approach.
I suggest you read Orlando et al’s article. Functional medicine is an interesting, mostly unproven, approach to patient care. But I will keep an open mind until we see better research that either does—or doesn’t—support the validity of its practices.
1. Orlando FA, Chang KL, Estores IM. Functional medicine: focusing on imbalances in core metabolic processes. J Fam Pract. 2021;70:482-488,498.
2. Gorski D. Functional medicine: the ultimate misnomer in the world of integrative medicine. Science-Based Medicine. April 11, 2016. Accessed January 4, 2022. https://sciencebasedmedicine.org/functional-medicine-the-ultimate-misnomer-in-the-world-of-integrative-medicine/
3. The Institute for Functional Medicine. Accessed January 4, 2022. www.ifm.org
1. Orlando FA, Chang KL, Estores IM. Functional medicine: focusing on imbalances in core metabolic processes. J Fam Pract. 2021;70:482-488,498.
2. Gorski D. Functional medicine: the ultimate misnomer in the world of integrative medicine. Science-Based Medicine. April 11, 2016. Accessed January 4, 2022. https://sciencebasedmedicine.org/functional-medicine-the-ultimate-misnomer-in-the-world-of-integrative-medicine/
3. The Institute for Functional Medicine. Accessed January 4, 2022. www.ifm.org
How to screen for and treat teen alcohol use
THE CASE
Paul F* is a 16-year-old White boy who lives with his mother and spends some weekends with his father who has shared custody. He recently presented to the clinic for treatment due to an arrest for disorderly conduct at school. He and a friend were found drinking liquor outside the school building when they were scheduled to be in class. Paul reported that he and his friends often drink at school and at extracurricular functions. He has been using alcohol for the past 2 years, with escalating consumption (5 or more drinks per episode) in the past year. Paul has been drinking most days of the week and has even driven under the influence at times. He said, “I just feel happier when I am drinking.” An accomplished soccer player recruited by colleges, Paul recently was suspended from the team due to his poor grades. His response was, “It’s stupid anyway. What’s the point of playing?”
●
* The patient’s name and some personal details have been changed to protect his identity.
Alcohol is the number 1 substance of abuse for adolescents, used more than tobacco or drugs.1-3 In 2007 and again in 2016, the Surgeon General of the United States issued reports to highlight this important topic,1,2 noting that early and repeated exposure to alcohol during this crucial time of brain development increases the risk for future problems, including addiction.2
Adolescent alcohol use is often underestimated by parents and physicians, including misjudging how much, how often, and how young children are when they begin to drink.1 Boys and girls tend to start drinking at similar ages (13.9 and 14.4 years, respectively),3 but as girls age, they tend to drink more and binge more.4 In 2019, 1 in 4 adolescents reported drinking and more than 4 million reported at least 1 episode of binge drinking in the prior month.4 These numbers have further ramifications: early drinking is associated with alcohol dependence, relapse, use of other substances, risky sexual behaviors, injurious behaviors, suicide, motor vehicle accidents, and dating violence.4-6
Diagnosing alcohol use disorder
The range of alcohol use includes consumption, bingeing, abuse, and dependence.7,8 Consumption is defined as the drinking of alcoholic beverages. Bingeing is the consumption of more than 5 drinks for men or 4 drinks for women in 2 hours, according to the National Institute on Alcohol Abuse and Alcoholism.7 However, the criterion is slightly different for the Substance Abuse and Mental Health Services Administration, which broadens the timeframe to “on the same occasion.”9 While previously known as separate disorders, alcohol abuse (or misuse) and alcohol dependence are now diagnostically classified together as alcohol use disorders (AUDs), per the Diagnostic and Statistical Manual of Mental Disorders-5 (DSM-5).8 AUD is further stratified as mild, moderate, or severe, depending on the number of criteria that are met by the patient (TABLE).8,10

Alcohol screening
Currently, the US Preventive Services Task Force (USPSTF) does not recommend screening adolescents ages 12 to 17 for AUD, and has instead issued an “I” statement (insufficient evidence).11 While the USPSTF recognizes the potential burdens of adolescent alcohol use, the potential harms of screening include “stigma, anxiety, labeling, discrimination, privacy concerns, and interference with the patient–clinician relationship.”11 The USPSTF also notes that it “did not find any evidence that specifically examined the harms of screening for alcohol use in adolescents.”11
This is at odds with recommendations from the American Academy of Pediatrics (AAP), which in 2011 released a policy statement advocating screening, brief intervention, and referral to treatment for adolescent substance use.12 In the United States, even though 83% of adolescents see a physician at least once each year,12,13 alcohol misuse screening still varies, occurring in the range of 50% to 86% of office visits.12 When screening does occur, it is often based on clinical impression only.12 Studies have shown that when a screening tool is not used, up to two-thirds of substance use disorders may be missed.12-15
Continue to: A full and complete biopsychosocial interview
A full and complete biopsychosocial interview with adolescents is a necessity, and should include queries about alcohol, drugs, and other substances. Acknowledgment of use should trigger further investigation into the substance use areas. Interviews may start with open-ended questions about alcohol use at home or at school before moving to more personalized and detailed questioning and use of screening tools.16
While various screening instruments exist, for the sake of brevity we provide as an example the Screening to Brief Intervention (S2BI) tool. It is an efficient, single-page tool that can help clinicians in their routine care of adolescents to quickly stratify the patient risk of substance use disorder as none/low, moderate, or severe.12 It can be found here: www.mcpap.com/pdf/S2Bi%20Toolkit.pdf (see page 10).
For all patients, but particularly for adolescents, confidentiality is important, and many specialty societies have created language to address this issue.12 Discuss confidentiality with both the adolescent patient and the patient’s caregiver simultaneously, with dialogue that includes: (a) the need to speak with adolescents alone during the office visit, (b) the benefits of confidentiality in the physician–patient relationship, and (c) the need to disclose selected information to keep patients safe.12 Describing the process for required disclosures is essential. Benefits of disclosure include further support for the adolescent patient as well as appropriate parental participation and support for possible referrals.12
Treating AUD
Treatment for AUD should be multifaceted. Screen for comorbid mood disorders, such as generalized anxiety,17,18 social anxiety,18 and depression,19 as well as for insomnia.18 Studies have demonstrated a strong link between insomnia and anxiety, and again between anxiety and AUD.17-19 Finally, screen for adverse childhood events such as trauma, victimization, and abuse.20 Addressing issues discovered in screening allows for more targeted and personalized treatment of AUD.
The National Institute on Drug Abuse categorizes evidence-based treatment into 3 areas: behavioral therapies, family therapies, and medications.21
Continue to: Behavioral therapies
Behavioral therapies can include group therapy, cognitive behavioral therapy (CBT), motivational enhancement therapy, 12-Step facilitation, and contingency management, in which small rewards or incentives are given for participation in treatment to reinforce positive behaviors.21
Family-based therapies, such as brief strategic family therapy, functional family therapy, and multisystem therapy recognize that adolescents exist in systems of families in communities, and that the patient’s success in treatment may be supported by these relationships.21
Some medications may achieve modest benefit for treatment of adolescents with AUD. Naltrexone, acamprosate, and disulfiram have all been used successfully to treat AUD in adults21; some physicians may choose to use these medications “off label” in adolescents. Bupropion has been used successfully in the treatment of nicotine use disorder,21 and a small study in 2005 showed some success with bupropion in treating adolescents with attention-deficit/hyperactivity disorder, comorbid depression, and substance use disorder.22 Naltrexone has also been studied in adolescents with opioid use disorder, although these were not large studies.23
Adolescents with serious, sustained issues with AUD may require more in-depth treatments such as an intensive outpatient program, a partial hospitalization program, or a residential treatment program.15 The least-restrictive environment is preferable.15 Families are generally included as part of the treatment and recovery process in those settings.21 Some patients may require detoxification prior to referral to residential treatment settings; the American Society of Addiction Medicine has published a comprehensive guideline on alcohol withdrawal.24
Paul’s family physician diagnosed his condition as AUD and referred him for CBT with a psychologist, who treated him for both the AUD and an underlying depressive disorder that was later identified. CBT focused on cognitive restructuring of depressive thoughts as well as support for continued abstinence from alcohol. The patient, with family support, declined antidepressant medication.
After 6 months of treatment, Paul and his parents were pleased with his progress. His grades improved to the point that he was permitted to play soccer again, and he was seriously looking at his future college options.
CORRESPONDENCE
Scott A. Fields, PhD, 3200 MacCorkle Avenue Southeast, 5th Floor, Robert C. Byrd Clinical Teaching Center, Department of Family Medicine, Charleston, WV 25304; [email protected]
1. US Department of Health and Human Services. The Surgeon General’s Call to Action to Prevent and Reduce Underage Drinking. Washington, DC; US Department of Health and Human Services, Office of the Surgeon General. 2007.
2. US Department of Health and Human Services. Facing Addiction in America: The Surgeon General’s Report on Alcohol, Drugs, and Health. Washington, DC; US Department of Health and Human Services, Office of the Surgeon General. 2016.
3. Hingson R, White A. New research findings since the 2007 Surgeon General’s Call to Action to Prevent and Reduce Underage Drinking: A review. J Stud Alcohol Drugs Suppl. 2014; 75:158-169.
4. National Institute on Alcohol Abuse and Alcoholism. Underage drinking. National Institute of Health. Accessed December 22, 2021. www.niaaa.nih.gov/publications/brochures-and-fact-sheets/underage-drinking.
5. Hingson R, Zha W, Iannotti R, et al. Physician advice to adolescents about drinking and other health behaviors. Pediatrics. 2013;131:249-257.
6. Schaus JF, Sole ML, McCoy TP, et al. Screening for high-risk drinking in a college student health center: characterizing students based on quantity, frequency, and harms. J Stud Alcohol Drugs Suppl. 2009;16:34-44.
7. National Institute on Alcohol Abuse and Alcoholism. Drinking levels defined. Accessed December 27, 2021. www.niaaa.nih.gov/alcohol-health/overview-alcohol-consumption/moderate-binge-drinking
8. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-5). Arlington, VA; American Psychiatric Association. 2013.
9. Substance Abuse and Mental Health Services Administration. Bringing down binge drinking. Accessed December 27, 2021. www.samhsa.gov/sites/default/files/programs_campaigns/nation_prevention_week/data-binge-drinking.pdf
10. Grant BF, Goldstein RB, Saha TD, et al. Epidemiology of DSM-5 Alcohol Use Disorder: results from the National Epidemiologic Survey on Alcohol and Related Conditions III. JAMA Psychiatry. 2015;72:757-766.
11. USPSTF. Screening and behavioral counseling interventions to reduce unhealthy alcohol use in adolescents and adults: US Preventive Services Task Force Recommendation Statement. JAMA. 2018;320:1899-1909.
12. Levy SJ, Williams JF, Committee on Substance Use and Prevention. Substance use screening, brief intervention, and referral to treatment. Pediatrics. 2016;138:e20161211.
13. MacKay AP, Duran CP. Adolescent Health in the United States. National Center for Health Statistics, Centers for Disease Control and Prevention. 2007.
14. Haller DM, Meynard A, Lefebvre D, et al. Effectiveness of training family physicians to deliver a brief intervention to address excessive substance use among young patients: a cluster randomized controlled trial. CMAJ. 2014;186:E263-E272.
15. Borus J, Parhami I, Levy S. Screening, brief intervention, and referral to treatment. Child Adolesc Psychiatric Clin N Am. 2016;25:579-601.
16. Knight J, Roberts T, Gabrielli J, et al. Adolescent alcohol and substance use and abuse. Performing preventive services: A bright futures handbook. Accessed December 22, 2021. American Academy of Pediatrics. https://ocfcpacourts.us/wp-content/uploads/2020/06/Adolescent_Alcohol_and_Substance_Abuse_001005.pdf
17. Dyer ML, Heron J, Hickman M, et al. Alcohol use in late adolescence and early adulthood: the role of generalized anxiety disorder and drinking to cope motives. Drug Alcohol Depend. 2019;204:107480.
18. Blumenthal H, Taylor DJ, Cloutier RM, et al. The links between social anxiety disorder, insomnia symptoms, and alcohol use disorders: findings from a large sample of adolescents in the United States. Behav Ther. 2019;50:50-59.
19. Pedrelli P, Shapero B, Archibald A, et al. Alcohol use and depression during adolescence and young adulthood: a summary and interpretation of mixed findings. Curr Addict Rep. 2016;3:91-97.
20. Davis JP, Dworkin ER, Helton J, et al. Extending poly-victimization theory: differential effects of adolescents’ experiences of victimization on substance use disorder diagnoses upon treatment entry. Child Abuse Negl. 2019; 89:165-177.
21. NIDA. Principles of adolescent substance use disorder treatment: a research-based guide. Accessed December 22, 2021. www.drugabuse.gov/publications/principles-adolescent-substance-use-disorder-treatment-research-based-guide
22. Solhkhah R, Wilens TE, Daly J, et al. Bupropion SR for the treatment of substance-abusing outpatient adolescents with attention-deficit/hyperactivity disorder and mood disorders. J Child Adolesc Psychopharmacol. 2005:15:777-786.
23. Camenga DR, Colon-Rivera HA, Muvvala SB. Medications for maintenance treatment of opioid use disorder in adolescents. J Stud Alcohol Drugs. 2019;80:393-402.
24. American Society of Addiction Medicine. The ASAM clinical practice guideline on alcohol withdrawal management. Accessed December 22, 2021. www.asam.org/quality-care/clinical-guidelines/alcohol-withdrawal-management-guideline
THE CASE
Paul F* is a 16-year-old White boy who lives with his mother and spends some weekends with his father who has shared custody. He recently presented to the clinic for treatment due to an arrest for disorderly conduct at school. He and a friend were found drinking liquor outside the school building when they were scheduled to be in class. Paul reported that he and his friends often drink at school and at extracurricular functions. He has been using alcohol for the past 2 years, with escalating consumption (5 or more drinks per episode) in the past year. Paul has been drinking most days of the week and has even driven under the influence at times. He said, “I just feel happier when I am drinking.” An accomplished soccer player recruited by colleges, Paul recently was suspended from the team due to his poor grades. His response was, “It’s stupid anyway. What’s the point of playing?”
●
* The patient’s name and some personal details have been changed to protect his identity.
Alcohol is the number 1 substance of abuse for adolescents, used more than tobacco or drugs.1-3 In 2007 and again in 2016, the Surgeon General of the United States issued reports to highlight this important topic,1,2 noting that early and repeated exposure to alcohol during this crucial time of brain development increases the risk for future problems, including addiction.2
Adolescent alcohol use is often underestimated by parents and physicians, including misjudging how much, how often, and how young children are when they begin to drink.1 Boys and girls tend to start drinking at similar ages (13.9 and 14.4 years, respectively),3 but as girls age, they tend to drink more and binge more.4 In 2019, 1 in 4 adolescents reported drinking and more than 4 million reported at least 1 episode of binge drinking in the prior month.4 These numbers have further ramifications: early drinking is associated with alcohol dependence, relapse, use of other substances, risky sexual behaviors, injurious behaviors, suicide, motor vehicle accidents, and dating violence.4-6
Diagnosing alcohol use disorder
The range of alcohol use includes consumption, bingeing, abuse, and dependence.7,8 Consumption is defined as the drinking of alcoholic beverages. Bingeing is the consumption of more than 5 drinks for men or 4 drinks for women in 2 hours, according to the National Institute on Alcohol Abuse and Alcoholism.7 However, the criterion is slightly different for the Substance Abuse and Mental Health Services Administration, which broadens the timeframe to “on the same occasion.”9 While previously known as separate disorders, alcohol abuse (or misuse) and alcohol dependence are now diagnostically classified together as alcohol use disorders (AUDs), per the Diagnostic and Statistical Manual of Mental Disorders-5 (DSM-5).8 AUD is further stratified as mild, moderate, or severe, depending on the number of criteria that are met by the patient (TABLE).8,10

Alcohol screening
Currently, the US Preventive Services Task Force (USPSTF) does not recommend screening adolescents ages 12 to 17 for AUD, and has instead issued an “I” statement (insufficient evidence).11 While the USPSTF recognizes the potential burdens of adolescent alcohol use, the potential harms of screening include “stigma, anxiety, labeling, discrimination, privacy concerns, and interference with the patient–clinician relationship.”11 The USPSTF also notes that it “did not find any evidence that specifically examined the harms of screening for alcohol use in adolescents.”11
This is at odds with recommendations from the American Academy of Pediatrics (AAP), which in 2011 released a policy statement advocating screening, brief intervention, and referral to treatment for adolescent substance use.12 In the United States, even though 83% of adolescents see a physician at least once each year,12,13 alcohol misuse screening still varies, occurring in the range of 50% to 86% of office visits.12 When screening does occur, it is often based on clinical impression only.12 Studies have shown that when a screening tool is not used, up to two-thirds of substance use disorders may be missed.12-15
Continue to: A full and complete biopsychosocial interview
A full and complete biopsychosocial interview with adolescents is a necessity, and should include queries about alcohol, drugs, and other substances. Acknowledgment of use should trigger further investigation into the substance use areas. Interviews may start with open-ended questions about alcohol use at home or at school before moving to more personalized and detailed questioning and use of screening tools.16
While various screening instruments exist, for the sake of brevity we provide as an example the Screening to Brief Intervention (S2BI) tool. It is an efficient, single-page tool that can help clinicians in their routine care of adolescents to quickly stratify the patient risk of substance use disorder as none/low, moderate, or severe.12 It can be found here: www.mcpap.com/pdf/S2Bi%20Toolkit.pdf (see page 10).
For all patients, but particularly for adolescents, confidentiality is important, and many specialty societies have created language to address this issue.12 Discuss confidentiality with both the adolescent patient and the patient’s caregiver simultaneously, with dialogue that includes: (a) the need to speak with adolescents alone during the office visit, (b) the benefits of confidentiality in the physician–patient relationship, and (c) the need to disclose selected information to keep patients safe.12 Describing the process for required disclosures is essential. Benefits of disclosure include further support for the adolescent patient as well as appropriate parental participation and support for possible referrals.12
Treating AUD
Treatment for AUD should be multifaceted. Screen for comorbid mood disorders, such as generalized anxiety,17,18 social anxiety,18 and depression,19 as well as for insomnia.18 Studies have demonstrated a strong link between insomnia and anxiety, and again between anxiety and AUD.17-19 Finally, screen for adverse childhood events such as trauma, victimization, and abuse.20 Addressing issues discovered in screening allows for more targeted and personalized treatment of AUD.
The National Institute on Drug Abuse categorizes evidence-based treatment into 3 areas: behavioral therapies, family therapies, and medications.21
Continue to: Behavioral therapies
Behavioral therapies can include group therapy, cognitive behavioral therapy (CBT), motivational enhancement therapy, 12-Step facilitation, and contingency management, in which small rewards or incentives are given for participation in treatment to reinforce positive behaviors.21
Family-based therapies, such as brief strategic family therapy, functional family therapy, and multisystem therapy recognize that adolescents exist in systems of families in communities, and that the patient’s success in treatment may be supported by these relationships.21
Some medications may achieve modest benefit for treatment of adolescents with AUD. Naltrexone, acamprosate, and disulfiram have all been used successfully to treat AUD in adults21; some physicians may choose to use these medications “off label” in adolescents. Bupropion has been used successfully in the treatment of nicotine use disorder,21 and a small study in 2005 showed some success with bupropion in treating adolescents with attention-deficit/hyperactivity disorder, comorbid depression, and substance use disorder.22 Naltrexone has also been studied in adolescents with opioid use disorder, although these were not large studies.23
Adolescents with serious, sustained issues with AUD may require more in-depth treatments such as an intensive outpatient program, a partial hospitalization program, or a residential treatment program.15 The least-restrictive environment is preferable.15 Families are generally included as part of the treatment and recovery process in those settings.21 Some patients may require detoxification prior to referral to residential treatment settings; the American Society of Addiction Medicine has published a comprehensive guideline on alcohol withdrawal.24
Paul’s family physician diagnosed his condition as AUD and referred him for CBT with a psychologist, who treated him for both the AUD and an underlying depressive disorder that was later identified. CBT focused on cognitive restructuring of depressive thoughts as well as support for continued abstinence from alcohol. The patient, with family support, declined antidepressant medication.
After 6 months of treatment, Paul and his parents were pleased with his progress. His grades improved to the point that he was permitted to play soccer again, and he was seriously looking at his future college options.
CORRESPONDENCE
Scott A. Fields, PhD, 3200 MacCorkle Avenue Southeast, 5th Floor, Robert C. Byrd Clinical Teaching Center, Department of Family Medicine, Charleston, WV 25304; [email protected]
THE CASE
Paul F* is a 16-year-old White boy who lives with his mother and spends some weekends with his father who has shared custody. He recently presented to the clinic for treatment due to an arrest for disorderly conduct at school. He and a friend were found drinking liquor outside the school building when they were scheduled to be in class. Paul reported that he and his friends often drink at school and at extracurricular functions. He has been using alcohol for the past 2 years, with escalating consumption (5 or more drinks per episode) in the past year. Paul has been drinking most days of the week and has even driven under the influence at times. He said, “I just feel happier when I am drinking.” An accomplished soccer player recruited by colleges, Paul recently was suspended from the team due to his poor grades. His response was, “It’s stupid anyway. What’s the point of playing?”
●
* The patient’s name and some personal details have been changed to protect his identity.
Alcohol is the number 1 substance of abuse for adolescents, used more than tobacco or drugs.1-3 In 2007 and again in 2016, the Surgeon General of the United States issued reports to highlight this important topic,1,2 noting that early and repeated exposure to alcohol during this crucial time of brain development increases the risk for future problems, including addiction.2
Adolescent alcohol use is often underestimated by parents and physicians, including misjudging how much, how often, and how young children are when they begin to drink.1 Boys and girls tend to start drinking at similar ages (13.9 and 14.4 years, respectively),3 but as girls age, they tend to drink more and binge more.4 In 2019, 1 in 4 adolescents reported drinking and more than 4 million reported at least 1 episode of binge drinking in the prior month.4 These numbers have further ramifications: early drinking is associated with alcohol dependence, relapse, use of other substances, risky sexual behaviors, injurious behaviors, suicide, motor vehicle accidents, and dating violence.4-6
Diagnosing alcohol use disorder
The range of alcohol use includes consumption, bingeing, abuse, and dependence.7,8 Consumption is defined as the drinking of alcoholic beverages. Bingeing is the consumption of more than 5 drinks for men or 4 drinks for women in 2 hours, according to the National Institute on Alcohol Abuse and Alcoholism.7 However, the criterion is slightly different for the Substance Abuse and Mental Health Services Administration, which broadens the timeframe to “on the same occasion.”9 While previously known as separate disorders, alcohol abuse (or misuse) and alcohol dependence are now diagnostically classified together as alcohol use disorders (AUDs), per the Diagnostic and Statistical Manual of Mental Disorders-5 (DSM-5).8 AUD is further stratified as mild, moderate, or severe, depending on the number of criteria that are met by the patient (TABLE).8,10

Alcohol screening
Currently, the US Preventive Services Task Force (USPSTF) does not recommend screening adolescents ages 12 to 17 for AUD, and has instead issued an “I” statement (insufficient evidence).11 While the USPSTF recognizes the potential burdens of adolescent alcohol use, the potential harms of screening include “stigma, anxiety, labeling, discrimination, privacy concerns, and interference with the patient–clinician relationship.”11 The USPSTF also notes that it “did not find any evidence that specifically examined the harms of screening for alcohol use in adolescents.”11
This is at odds with recommendations from the American Academy of Pediatrics (AAP), which in 2011 released a policy statement advocating screening, brief intervention, and referral to treatment for adolescent substance use.12 In the United States, even though 83% of adolescents see a physician at least once each year,12,13 alcohol misuse screening still varies, occurring in the range of 50% to 86% of office visits.12 When screening does occur, it is often based on clinical impression only.12 Studies have shown that when a screening tool is not used, up to two-thirds of substance use disorders may be missed.12-15
Continue to: A full and complete biopsychosocial interview
A full and complete biopsychosocial interview with adolescents is a necessity, and should include queries about alcohol, drugs, and other substances. Acknowledgment of use should trigger further investigation into the substance use areas. Interviews may start with open-ended questions about alcohol use at home or at school before moving to more personalized and detailed questioning and use of screening tools.16
While various screening instruments exist, for the sake of brevity we provide as an example the Screening to Brief Intervention (S2BI) tool. It is an efficient, single-page tool that can help clinicians in their routine care of adolescents to quickly stratify the patient risk of substance use disorder as none/low, moderate, or severe.12 It can be found here: www.mcpap.com/pdf/S2Bi%20Toolkit.pdf (see page 10).
For all patients, but particularly for adolescents, confidentiality is important, and many specialty societies have created language to address this issue.12 Discuss confidentiality with both the adolescent patient and the patient’s caregiver simultaneously, with dialogue that includes: (a) the need to speak with adolescents alone during the office visit, (b) the benefits of confidentiality in the physician–patient relationship, and (c) the need to disclose selected information to keep patients safe.12 Describing the process for required disclosures is essential. Benefits of disclosure include further support for the adolescent patient as well as appropriate parental participation and support for possible referrals.12
Treating AUD
Treatment for AUD should be multifaceted. Screen for comorbid mood disorders, such as generalized anxiety,17,18 social anxiety,18 and depression,19 as well as for insomnia.18 Studies have demonstrated a strong link between insomnia and anxiety, and again between anxiety and AUD.17-19 Finally, screen for adverse childhood events such as trauma, victimization, and abuse.20 Addressing issues discovered in screening allows for more targeted and personalized treatment of AUD.
The National Institute on Drug Abuse categorizes evidence-based treatment into 3 areas: behavioral therapies, family therapies, and medications.21
Continue to: Behavioral therapies
Behavioral therapies can include group therapy, cognitive behavioral therapy (CBT), motivational enhancement therapy, 12-Step facilitation, and contingency management, in which small rewards or incentives are given for participation in treatment to reinforce positive behaviors.21
Family-based therapies, such as brief strategic family therapy, functional family therapy, and multisystem therapy recognize that adolescents exist in systems of families in communities, and that the patient’s success in treatment may be supported by these relationships.21
Some medications may achieve modest benefit for treatment of adolescents with AUD. Naltrexone, acamprosate, and disulfiram have all been used successfully to treat AUD in adults21; some physicians may choose to use these medications “off label” in adolescents. Bupropion has been used successfully in the treatment of nicotine use disorder,21 and a small study in 2005 showed some success with bupropion in treating adolescents with attention-deficit/hyperactivity disorder, comorbid depression, and substance use disorder.22 Naltrexone has also been studied in adolescents with opioid use disorder, although these were not large studies.23
Adolescents with serious, sustained issues with AUD may require more in-depth treatments such as an intensive outpatient program, a partial hospitalization program, or a residential treatment program.15 The least-restrictive environment is preferable.15 Families are generally included as part of the treatment and recovery process in those settings.21 Some patients may require detoxification prior to referral to residential treatment settings; the American Society of Addiction Medicine has published a comprehensive guideline on alcohol withdrawal.24
Paul’s family physician diagnosed his condition as AUD and referred him for CBT with a psychologist, who treated him for both the AUD and an underlying depressive disorder that was later identified. CBT focused on cognitive restructuring of depressive thoughts as well as support for continued abstinence from alcohol. The patient, with family support, declined antidepressant medication.
After 6 months of treatment, Paul and his parents were pleased with his progress. His grades improved to the point that he was permitted to play soccer again, and he was seriously looking at his future college options.
CORRESPONDENCE
Scott A. Fields, PhD, 3200 MacCorkle Avenue Southeast, 5th Floor, Robert C. Byrd Clinical Teaching Center, Department of Family Medicine, Charleston, WV 25304; [email protected]
1. US Department of Health and Human Services. The Surgeon General’s Call to Action to Prevent and Reduce Underage Drinking. Washington, DC; US Department of Health and Human Services, Office of the Surgeon General. 2007.
2. US Department of Health and Human Services. Facing Addiction in America: The Surgeon General’s Report on Alcohol, Drugs, and Health. Washington, DC; US Department of Health and Human Services, Office of the Surgeon General. 2016.
3. Hingson R, White A. New research findings since the 2007 Surgeon General’s Call to Action to Prevent and Reduce Underage Drinking: A review. J Stud Alcohol Drugs Suppl. 2014; 75:158-169.
4. National Institute on Alcohol Abuse and Alcoholism. Underage drinking. National Institute of Health. Accessed December 22, 2021. www.niaaa.nih.gov/publications/brochures-and-fact-sheets/underage-drinking.
5. Hingson R, Zha W, Iannotti R, et al. Physician advice to adolescents about drinking and other health behaviors. Pediatrics. 2013;131:249-257.
6. Schaus JF, Sole ML, McCoy TP, et al. Screening for high-risk drinking in a college student health center: characterizing students based on quantity, frequency, and harms. J Stud Alcohol Drugs Suppl. 2009;16:34-44.
7. National Institute on Alcohol Abuse and Alcoholism. Drinking levels defined. Accessed December 27, 2021. www.niaaa.nih.gov/alcohol-health/overview-alcohol-consumption/moderate-binge-drinking
8. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-5). Arlington, VA; American Psychiatric Association. 2013.
9. Substance Abuse and Mental Health Services Administration. Bringing down binge drinking. Accessed December 27, 2021. www.samhsa.gov/sites/default/files/programs_campaigns/nation_prevention_week/data-binge-drinking.pdf
10. Grant BF, Goldstein RB, Saha TD, et al. Epidemiology of DSM-5 Alcohol Use Disorder: results from the National Epidemiologic Survey on Alcohol and Related Conditions III. JAMA Psychiatry. 2015;72:757-766.
11. USPSTF. Screening and behavioral counseling interventions to reduce unhealthy alcohol use in adolescents and adults: US Preventive Services Task Force Recommendation Statement. JAMA. 2018;320:1899-1909.
12. Levy SJ, Williams JF, Committee on Substance Use and Prevention. Substance use screening, brief intervention, and referral to treatment. Pediatrics. 2016;138:e20161211.
13. MacKay AP, Duran CP. Adolescent Health in the United States. National Center for Health Statistics, Centers for Disease Control and Prevention. 2007.
14. Haller DM, Meynard A, Lefebvre D, et al. Effectiveness of training family physicians to deliver a brief intervention to address excessive substance use among young patients: a cluster randomized controlled trial. CMAJ. 2014;186:E263-E272.
15. Borus J, Parhami I, Levy S. Screening, brief intervention, and referral to treatment. Child Adolesc Psychiatric Clin N Am. 2016;25:579-601.
16. Knight J, Roberts T, Gabrielli J, et al. Adolescent alcohol and substance use and abuse. Performing preventive services: A bright futures handbook. Accessed December 22, 2021. American Academy of Pediatrics. https://ocfcpacourts.us/wp-content/uploads/2020/06/Adolescent_Alcohol_and_Substance_Abuse_001005.pdf
17. Dyer ML, Heron J, Hickman M, et al. Alcohol use in late adolescence and early adulthood: the role of generalized anxiety disorder and drinking to cope motives. Drug Alcohol Depend. 2019;204:107480.
18. Blumenthal H, Taylor DJ, Cloutier RM, et al. The links between social anxiety disorder, insomnia symptoms, and alcohol use disorders: findings from a large sample of adolescents in the United States. Behav Ther. 2019;50:50-59.
19. Pedrelli P, Shapero B, Archibald A, et al. Alcohol use and depression during adolescence and young adulthood: a summary and interpretation of mixed findings. Curr Addict Rep. 2016;3:91-97.
20. Davis JP, Dworkin ER, Helton J, et al. Extending poly-victimization theory: differential effects of adolescents’ experiences of victimization on substance use disorder diagnoses upon treatment entry. Child Abuse Negl. 2019; 89:165-177.
21. NIDA. Principles of adolescent substance use disorder treatment: a research-based guide. Accessed December 22, 2021. www.drugabuse.gov/publications/principles-adolescent-substance-use-disorder-treatment-research-based-guide
22. Solhkhah R, Wilens TE, Daly J, et al. Bupropion SR for the treatment of substance-abusing outpatient adolescents with attention-deficit/hyperactivity disorder and mood disorders. J Child Adolesc Psychopharmacol. 2005:15:777-786.
23. Camenga DR, Colon-Rivera HA, Muvvala SB. Medications for maintenance treatment of opioid use disorder in adolescents. J Stud Alcohol Drugs. 2019;80:393-402.
24. American Society of Addiction Medicine. The ASAM clinical practice guideline on alcohol withdrawal management. Accessed December 22, 2021. www.asam.org/quality-care/clinical-guidelines/alcohol-withdrawal-management-guideline
1. US Department of Health and Human Services. The Surgeon General’s Call to Action to Prevent and Reduce Underage Drinking. Washington, DC; US Department of Health and Human Services, Office of the Surgeon General. 2007.
2. US Department of Health and Human Services. Facing Addiction in America: The Surgeon General’s Report on Alcohol, Drugs, and Health. Washington, DC; US Department of Health and Human Services, Office of the Surgeon General. 2016.
3. Hingson R, White A. New research findings since the 2007 Surgeon General’s Call to Action to Prevent and Reduce Underage Drinking: A review. J Stud Alcohol Drugs Suppl. 2014; 75:158-169.
4. National Institute on Alcohol Abuse and Alcoholism. Underage drinking. National Institute of Health. Accessed December 22, 2021. www.niaaa.nih.gov/publications/brochures-and-fact-sheets/underage-drinking.
5. Hingson R, Zha W, Iannotti R, et al. Physician advice to adolescents about drinking and other health behaviors. Pediatrics. 2013;131:249-257.
6. Schaus JF, Sole ML, McCoy TP, et al. Screening for high-risk drinking in a college student health center: characterizing students based on quantity, frequency, and harms. J Stud Alcohol Drugs Suppl. 2009;16:34-44.
7. National Institute on Alcohol Abuse and Alcoholism. Drinking levels defined. Accessed December 27, 2021. www.niaaa.nih.gov/alcohol-health/overview-alcohol-consumption/moderate-binge-drinking
8. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-5). Arlington, VA; American Psychiatric Association. 2013.
9. Substance Abuse and Mental Health Services Administration. Bringing down binge drinking. Accessed December 27, 2021. www.samhsa.gov/sites/default/files/programs_campaigns/nation_prevention_week/data-binge-drinking.pdf
10. Grant BF, Goldstein RB, Saha TD, et al. Epidemiology of DSM-5 Alcohol Use Disorder: results from the National Epidemiologic Survey on Alcohol and Related Conditions III. JAMA Psychiatry. 2015;72:757-766.
11. USPSTF. Screening and behavioral counseling interventions to reduce unhealthy alcohol use in adolescents and adults: US Preventive Services Task Force Recommendation Statement. JAMA. 2018;320:1899-1909.
12. Levy SJ, Williams JF, Committee on Substance Use and Prevention. Substance use screening, brief intervention, and referral to treatment. Pediatrics. 2016;138:e20161211.
13. MacKay AP, Duran CP. Adolescent Health in the United States. National Center for Health Statistics, Centers for Disease Control and Prevention. 2007.
14. Haller DM, Meynard A, Lefebvre D, et al. Effectiveness of training family physicians to deliver a brief intervention to address excessive substance use among young patients: a cluster randomized controlled trial. CMAJ. 2014;186:E263-E272.
15. Borus J, Parhami I, Levy S. Screening, brief intervention, and referral to treatment. Child Adolesc Psychiatric Clin N Am. 2016;25:579-601.
16. Knight J, Roberts T, Gabrielli J, et al. Adolescent alcohol and substance use and abuse. Performing preventive services: A bright futures handbook. Accessed December 22, 2021. American Academy of Pediatrics. https://ocfcpacourts.us/wp-content/uploads/2020/06/Adolescent_Alcohol_and_Substance_Abuse_001005.pdf
17. Dyer ML, Heron J, Hickman M, et al. Alcohol use in late adolescence and early adulthood: the role of generalized anxiety disorder and drinking to cope motives. Drug Alcohol Depend. 2019;204:107480.
18. Blumenthal H, Taylor DJ, Cloutier RM, et al. The links between social anxiety disorder, insomnia symptoms, and alcohol use disorders: findings from a large sample of adolescents in the United States. Behav Ther. 2019;50:50-59.
19. Pedrelli P, Shapero B, Archibald A, et al. Alcohol use and depression during adolescence and young adulthood: a summary and interpretation of mixed findings. Curr Addict Rep. 2016;3:91-97.
20. Davis JP, Dworkin ER, Helton J, et al. Extending poly-victimization theory: differential effects of adolescents’ experiences of victimization on substance use disorder diagnoses upon treatment entry. Child Abuse Negl. 2019; 89:165-177.
21. NIDA. Principles of adolescent substance use disorder treatment: a research-based guide. Accessed December 22, 2021. www.drugabuse.gov/publications/principles-adolescent-substance-use-disorder-treatment-research-based-guide
22. Solhkhah R, Wilens TE, Daly J, et al. Bupropion SR for the treatment of substance-abusing outpatient adolescents with attention-deficit/hyperactivity disorder and mood disorders. J Child Adolesc Psychopharmacol. 2005:15:777-786.
23. Camenga DR, Colon-Rivera HA, Muvvala SB. Medications for maintenance treatment of opioid use disorder in adolescents. J Stud Alcohol Drugs. 2019;80:393-402.
24. American Society of Addiction Medicine. The ASAM clinical practice guideline on alcohol withdrawal management. Accessed December 22, 2021. www.asam.org/quality-care/clinical-guidelines/alcohol-withdrawal-management-guideline
What’s in a White Coat? The Changing Trends in Physician Attire and What it Means for Dermatology
The White Coat Ceremony is an enduring memory from my medical school years. Amidst the tumult of memories of seemingly endless sleepless nights spent in libraries and cramming for clerkship examinations between surgical cases, I recall a sunny spring day in 2016 where I gathered with my classmates, family, and friends in the medical school campus courtyard. There were several short, mostly forgotten speeches after which proud fathers and mothers, partners, or siblings slipped the all-important white coat onto the shoulders of the physicians-to-be. At that moment, I felt the weight of tradition centuries in the making resting on my shoulders. Of course, the pomp of the ceremony might have felt a tad overblown had I known that the whole thing had fewer years under its belt than the movie Die Hard.
That’s right, the first White Coat Ceremony was held 5 years after the release of that Bruce Willis classic. Dr. Arnold Gold, a pediatric neurologist on faculty at Columbia University, conceived the ceremony in 1993, and it spread rapidly to medical schools—and later nursing schools—across the United States.1 Although the values highlighted by the White Coat Ceremony—humanism and compassion in medicine—are timeless, the ceremony itself is a more modern undertaking. What, then, of the white coat itself? Is it the timeless symbol of doctoring—of medicine—that we all presume it to be? Or is it a symbol of modern marketing, just a trend that caught on? And is it encountering its twilight—as trends often do—in the face of changing fashion and, more fundamentally, in changes to who our physicians are and to their roles in our society?
The Cleanliness of the White Coat
Until the end of the 19th century, physicians in the Western world most frequently dressed in black formal wear. The rationale behind this attire seems to have been twofold. First, society as a whole perceived the physician’s work as a serious and formal matter, and any medical encounter had to reflect the gravity of the occasion. Additionally, physicians’ visits often were a portent of impending demise, as physicians in the era prior to antibiotics and antisepsis frequently had little to offer their patients outside of—at best—anecdotal treatments and—at worst—sheer quackery.2 Black may have seemed a respectful choice for patients who likely faced dire outcomes regardless of the treatment afforded.3
With the turn of the century came a new understanding of the concepts of antisepsis and disease transmission. While Joseph Lister first published on the use of antisepsis in 1867, his practices did not become commonplace until the early 1900s.4 Around the same time came the Flexner report,5 the publication of William Osler’s Principles and Practice of Medicine,6 and the establishment of the modern medical residency, all of which contributed to the shift from the patient’s own bedside and to the hospital as the house of medicine, with cleanliness and antisepsis as part of its core principles.7 The white coat arose as a symbol of purity and freedom from disease. Throughout the 20th century and into the 21st, it has remained the predominant symbol of cleanliness and professionalism for the medical practitioner.
Patient Preference of Physician Attire
Although the white coat may serve as a professional symbol and is well respected medicine, it also plays an important role in the layperson’s perception of their health care providers.8 There is little denying that patients prefer their physicians, almost uniformly, to wear a white coat. A systematic review of physician attire that included 30 studies mainly from North America, Europe, and the United Kingdom found that patient preference for formal attire and white coats is near universal.9 Patients routinely rate physicians wearing a white coat as more intelligent and trustworthy and feel more confident in the care they will receive.10-13 They also freely admit that a physician’s appearance influences their satisfaction with their care.14 The recent adoption of the fleece, or softshell, jacket has not yet pervaded patients’ perceptions of what is considered appropriate physician attire. A 500-respondent survey found that patients were more likely to rate a model wearing a white coat as more professional and experienced compared to the same model wearing a fleece or softshell jacket or other formal attire sans white coat.15
Closer examination of the same data, however, reveals results reproduced with startling consistency across several studies, which suggest those of us adopting other attire need not dig those white coats out of the closet just yet. First, while many studies point to patient preference for white coats, this preference is uniformly strongest in older patients, beginning around age 40 years and becoming an entrenched preference in those older than 65 years.9,14,16-18 On the other hand, younger patient populations display little to no such preference, and some studies indicate that younger patients actually prefer scrubs over formal attire in specific settings such as surgical offices, procedural spaces, or the emergency department.12,14,19 This suggests that bias in favor of traditional physician garb may be more linked to age demographics and may continue to shift as the overall population ages. Additionally, although patients might profess a strong preference for physician attire in theory, it often does not translate into any impact on the patient’s perception of the physician following a clinic visit. The large systematic review on the topic noted that only 25% of studies that surveyed patients about a clinical visit following the encounter reported that physician attire influenced their satisfaction with that visit, suggesting that attire may be less likely to influence patients in the real-world context of receiving care.9 In fact, a prospective study of patient perception of medical staff and interactions found that staff style of dress not only had no bearing on the perception of staff or visit satisfaction but that patients often failed to even accurately recall physician attire when surveyed.20 Another survey study echoed these conclusions, finding that physician attire had no effect on the perception of a proposed treatment plan.21
What do we know about patient perception of physician attire in the dermatology setting specifically, where visits can be unique in their tendency to transition from medical to procedural in the span of a 15-minute encounter depending on the patient’s chief concern? A survey study of dermatology patients at the general, surgical, and wound care dermatology clinics of an academic medical center (Miami, Florida) found that professional attire with a white coat was strongly preferred across a litany of scenarios assessing many aspects of dermatologic care.21 Similarly, a study of patients visiting a single institution’s dermatology and pediatric dermatology clinics surveyed patients and parents regarding attire prior to an appointment and specifically asked if a white coat should be worn.13 Fifty-four percent of the adult patients (n=176) surveyed professed a preference for physicians in white coats, with a stronger preference for white coats reported by those 50 years and older (55%; n=113). Parents or guardians presenting to the pediatric dermatology clinic, on the other hand, favored less formal attire.13 A recent, real-world study performed at an outpatient dermatology clinic examined the influence of changing physician attire on a patient’s perceptions of care received during clinic encounters. They found no substantial difference in patient satisfaction scores before and following the adoption of a new clinic uniform that transitioned from formal attire to fitted scrubs.22
Racial and Gender Bias Affecting Attire Preference
With any study of preference, there is the underlying concern over respondent bias. Many of the studies discussed here have found secondarily that a patient’s implicit bias does not end at the clothes their physician is wearing. The survey study of dermatology patients from the academic medical center in Miami, Florida, found that patients preferred that Black physicians of either sex be garbed in professional attire at all times but generally were more accepting of White physicians in less formal attire.21 Adamson et al23 published a response to the study’s findings urging dermatologists to recognize that a physician’s race and gender influence patients’ perceptions in much the same way that physician attire seems to and encouraged the development of a more diverse dermatologic workforce to help combat this prejudice. The issue of bias is not limited to the specialty of dermatology; the recent survey study by Xun et al15 found that respondents consistently rated female models garbed in physician attire as less professional than male model counterparts. Additionally, female models wearing white coats were mistakenly identified as medical technicians, physician assistants, or nurses with substantially more frequency than males, despite being clothed in the traditional physician garb. Several other publications on the subject have uncovered implicit bias, though it is rarely, if ever, the principle focus of the study.10,24,25 As is unfortunately true in many professions, female physicians and physicians from ethnic minorities face barriers to being perceived as fully competent physicians.
Impact of the COVID-19 Pandemic
Finally, of course, there is the ever-present question of the effect of the pandemic. Although the exact role of the white coat as a fomite for infection—and especially for the spread of viral illness—remains controversial, the perception nonetheless has helped catalyze the movement to alternatives such as short-sleeved white coats, technical jackets, and more recently, fitted scrubs.26-29 As with much in this realm, facts seem less important than perceptions; Zahrina et al30 found that when patients were presented with information regarding the risk for microbial contamination associated with white coats, preference for physicians in professional garb plummeted from 72% to only 22%. To date no articles have examined patient perceptions of the white coat in the context of microbial transmission in the age of COVID-19, but future articles on this topic are likely and may serve to further the demise of the white coat.
Final Thoughts
From my vantage point, it seems the white coat will be claimed by the outgoing tide. During this most recent residency interview season, I do not recall a single medical student wearing a short white coat. The closest I came was a quick glimpse of a crumpled white jacket slung over an arm or stuffed in a shoulder bag. Rotating interns and residents from other services on rotation in our department present in softshell or fleece jackets. Fitted scrubs in the newest trendy colors speckle a previously all-white canvas. I, for one, have not donned my own white coat in at least a year, and perhaps it is all for the best. Physician attire is one small aspect of the practice of medicine and likely bears little, if any, relation to the wearer’s qualifications. Our focus should be on building rapport with our patients, providing high-quality care, reducing the risk for nosocomial infection, and developing a health care system that is fair and equitable for patients and health care workers alike, not on who is wearing what. Perhaps the introduction of new physician attire is a small part of the disruption we need to help address persistent gender and racial biases in our field and help shepherd our patients and colleagues to a worldview that is more open and accepting of physicians of diverse backgrounds.
- White Coat Ceremony. Gold Foundation website. Accessed December 26, 2021. https://www.gold-foundation.org/programs/white-coat-ceremony/
- Shryock RH. The Development of Modern Medicine. University of Pennsylvania Press; 2017.
- Hochberg MS. The doctor’s white coat—an historical perspective. Virtual Mentor. 2007;9:310-314.
- Lister J. On the antiseptic principle in the practice of surgery. Lancet. 1867;90:353-356.
- Flexner A. Medical Education in the United States and Canada: A Report to the Carnegie Foundation for the Advancement of Teaching. Carnegie Foundation for the Advancement of Teaching; 1910.
- Osler W. Principles and Practice of Medicine: Designed for the Use of Practitioners and Students of Medicine. D. Appleton & Company; 1892.
- Blumhagen DW. The doctor’s white coat: the image of the physician in modern America. Ann Intern Med. 1979;91:111-116.
- Verghese BG, Kashinath SK, Jadhav N, et al. Physician attire: physicians’ perspectives on attire in a community hospital setting among non-surgical specialties. J Community Hosp Intern Med Perspect. 2020;10:1-5.
- Petrilli CM, Mack M, Petrilli JJ, et al. Understanding the role of physician attire on patient perceptions: a systematic review of the literature—targeting attire to improve likelihood of rapport (TAILOR) investigators. BMJ Open. 2015;5:E006678.
- Rehman SU, Nietert PJ, Cope DW, et al. What to wear today? effect of doctor’s attire on the trust and confidence of patients. Am J Med. 2005;118:1279-1286.
- Jennings JD, Ciaravino SG, Ramsey FV, et al. Physicians’ attire influences patients’ perceptions in the urban outpatient orthopaedic surgery setting. Clin Orthop Relat Res. 2016;474:1908-1918.
- Gherardi G, Cameron J, West A, et al. Are we dressed to impress? a descriptive survey assessing patients preference of doctors’ attire in the hospital setting. Clin Med (Lond). 2009;9:519-524.
- Thomas MW, Burkhart CN, Lugo-Somolinos A, et al. Patients’ perceptions of physician attire in dermatology clinics. Arch Dermatol. 2011;147:505-506.
- Petrilli CM, Saint S, Jennings JJ, et al. Understanding patient preference for physician attire: a cross-sectional observational study of 10 academic medical centres in the USA. BMJ Open. 2018;8:E021239.
- Xun H, Chen J, Sun AH, et al. Public perceptions of physician attire and professionalism in the US. JAMA Network Open. 2021;4:E2117779.
- Kamata K, Kuriyama A, Chopra V, et al. Patient preferences for physician attire: a multicenter study in Japan [published online February 11, 2020]. J Hosp Med. 2020;15:204-210.
- Budny AM, Rogers LC, Mandracchia VJ, et al. The physician’s attire and its influence on patient confidence. J Am Podiatr Assoc. 2006;96:132-138.
- Lill MM, Wilkinson TJ. Judging a book by its cover: descriptive survey of patients’ preferences for doctors’ appearance and mode of address. Br Med J. 2005;331:1524-1527.
- Hossler EW, Shipp D, Palmer M, et al. Impact of provider attire on patient satisfaction in an outpatient dermatology clinic. Cutis. 2018;102:127-129.
- Boon D, Wardrope J. What should doctors wear in the accident and emergency department? patients’ perception. J Accid Emerg Med. 1994;11:175-177.
- Fox JD, Prado G, Baquerizo Nole KL, et al. Patient preference in dermatologist attire in the medical, surgical, and wound care settings. JAMA Dermatol. 2016;152:913-919.
- Bray JK, Porter C, Feldman SR. The effect of physician appearance on patient perceptions of treatment plans. Dermatol Online J. 2021;27. doi:10.5070/D327553611
- Adamson AS, Wright SW, Pandya AG. A missed opportunity to discuss racial and gender bias in dermatology. JAMA Dermatol. 2017;153:110-111.
- Hartmans C, Heremans S, Lagrain M, et al. The doctor’s new clothes: professional or fashionable? Primary Health Care. 2013;3:135.
- Kurihara H, Maeno T, Maeno T. Importance of physicians’ attire: factors influencing the impression it makes on patients, a cross-sectional study. Asia Pac Fam Med. 2014;13:2.
- Treakle AM, Thom KA, Furuno JP, et al. Bacterial contamination of health care workers’ white coats. Am J Infect Control. 2009;37:101-105.
- Banu A, Anand M, Nagi N, et al. White coats as a vehicle for bacterial dissemination. J Clin Diagn Res. 2012;6:1381-1384.
- Haun N, Hooper-Lane C, Safdar N. Healthcare personnel attire and devices as fomites: a systematic review. Infect Control Hosp Epidemiol. 2016;37:1367-1373.
- Tse G, Withey S, Yeo JM, et al. Bare below the elbows: was the target the white coat? J Hosp Infect. 2015;91:299-301.
- Zahrina AZ, Haymond P, Rosanna P, et al. Does the attire of a primary care physician affect patients’ perceptions and their levels of trust in the doctor? Malays Fam Physician. 2018;13:3-11.
The White Coat Ceremony is an enduring memory from my medical school years. Amidst the tumult of memories of seemingly endless sleepless nights spent in libraries and cramming for clerkship examinations between surgical cases, I recall a sunny spring day in 2016 where I gathered with my classmates, family, and friends in the medical school campus courtyard. There were several short, mostly forgotten speeches after which proud fathers and mothers, partners, or siblings slipped the all-important white coat onto the shoulders of the physicians-to-be. At that moment, I felt the weight of tradition centuries in the making resting on my shoulders. Of course, the pomp of the ceremony might have felt a tad overblown had I known that the whole thing had fewer years under its belt than the movie Die Hard.
That’s right, the first White Coat Ceremony was held 5 years after the release of that Bruce Willis classic. Dr. Arnold Gold, a pediatric neurologist on faculty at Columbia University, conceived the ceremony in 1993, and it spread rapidly to medical schools—and later nursing schools—across the United States.1 Although the values highlighted by the White Coat Ceremony—humanism and compassion in medicine—are timeless, the ceremony itself is a more modern undertaking. What, then, of the white coat itself? Is it the timeless symbol of doctoring—of medicine—that we all presume it to be? Or is it a symbol of modern marketing, just a trend that caught on? And is it encountering its twilight—as trends often do—in the face of changing fashion and, more fundamentally, in changes to who our physicians are and to their roles in our society?
The Cleanliness of the White Coat
Until the end of the 19th century, physicians in the Western world most frequently dressed in black formal wear. The rationale behind this attire seems to have been twofold. First, society as a whole perceived the physician’s work as a serious and formal matter, and any medical encounter had to reflect the gravity of the occasion. Additionally, physicians’ visits often were a portent of impending demise, as physicians in the era prior to antibiotics and antisepsis frequently had little to offer their patients outside of—at best—anecdotal treatments and—at worst—sheer quackery.2 Black may have seemed a respectful choice for patients who likely faced dire outcomes regardless of the treatment afforded.3
With the turn of the century came a new understanding of the concepts of antisepsis and disease transmission. While Joseph Lister first published on the use of antisepsis in 1867, his practices did not become commonplace until the early 1900s.4 Around the same time came the Flexner report,5 the publication of William Osler’s Principles and Practice of Medicine,6 and the establishment of the modern medical residency, all of which contributed to the shift from the patient’s own bedside and to the hospital as the house of medicine, with cleanliness and antisepsis as part of its core principles.7 The white coat arose as a symbol of purity and freedom from disease. Throughout the 20th century and into the 21st, it has remained the predominant symbol of cleanliness and professionalism for the medical practitioner.
Patient Preference of Physician Attire
Although the white coat may serve as a professional symbol and is well respected medicine, it also plays an important role in the layperson’s perception of their health care providers.8 There is little denying that patients prefer their physicians, almost uniformly, to wear a white coat. A systematic review of physician attire that included 30 studies mainly from North America, Europe, and the United Kingdom found that patient preference for formal attire and white coats is near universal.9 Patients routinely rate physicians wearing a white coat as more intelligent and trustworthy and feel more confident in the care they will receive.10-13 They also freely admit that a physician’s appearance influences their satisfaction with their care.14 The recent adoption of the fleece, or softshell, jacket has not yet pervaded patients’ perceptions of what is considered appropriate physician attire. A 500-respondent survey found that patients were more likely to rate a model wearing a white coat as more professional and experienced compared to the same model wearing a fleece or softshell jacket or other formal attire sans white coat.15
Closer examination of the same data, however, reveals results reproduced with startling consistency across several studies, which suggest those of us adopting other attire need not dig those white coats out of the closet just yet. First, while many studies point to patient preference for white coats, this preference is uniformly strongest in older patients, beginning around age 40 years and becoming an entrenched preference in those older than 65 years.9,14,16-18 On the other hand, younger patient populations display little to no such preference, and some studies indicate that younger patients actually prefer scrubs over formal attire in specific settings such as surgical offices, procedural spaces, or the emergency department.12,14,19 This suggests that bias in favor of traditional physician garb may be more linked to age demographics and may continue to shift as the overall population ages. Additionally, although patients might profess a strong preference for physician attire in theory, it often does not translate into any impact on the patient’s perception of the physician following a clinic visit. The large systematic review on the topic noted that only 25% of studies that surveyed patients about a clinical visit following the encounter reported that physician attire influenced their satisfaction with that visit, suggesting that attire may be less likely to influence patients in the real-world context of receiving care.9 In fact, a prospective study of patient perception of medical staff and interactions found that staff style of dress not only had no bearing on the perception of staff or visit satisfaction but that patients often failed to even accurately recall physician attire when surveyed.20 Another survey study echoed these conclusions, finding that physician attire had no effect on the perception of a proposed treatment plan.21
What do we know about patient perception of physician attire in the dermatology setting specifically, where visits can be unique in their tendency to transition from medical to procedural in the span of a 15-minute encounter depending on the patient’s chief concern? A survey study of dermatology patients at the general, surgical, and wound care dermatology clinics of an academic medical center (Miami, Florida) found that professional attire with a white coat was strongly preferred across a litany of scenarios assessing many aspects of dermatologic care.21 Similarly, a study of patients visiting a single institution’s dermatology and pediatric dermatology clinics surveyed patients and parents regarding attire prior to an appointment and specifically asked if a white coat should be worn.13 Fifty-four percent of the adult patients (n=176) surveyed professed a preference for physicians in white coats, with a stronger preference for white coats reported by those 50 years and older (55%; n=113). Parents or guardians presenting to the pediatric dermatology clinic, on the other hand, favored less formal attire.13 A recent, real-world study performed at an outpatient dermatology clinic examined the influence of changing physician attire on a patient’s perceptions of care received during clinic encounters. They found no substantial difference in patient satisfaction scores before and following the adoption of a new clinic uniform that transitioned from formal attire to fitted scrubs.22
Racial and Gender Bias Affecting Attire Preference
With any study of preference, there is the underlying concern over respondent bias. Many of the studies discussed here have found secondarily that a patient’s implicit bias does not end at the clothes their physician is wearing. The survey study of dermatology patients from the academic medical center in Miami, Florida, found that patients preferred that Black physicians of either sex be garbed in professional attire at all times but generally were more accepting of White physicians in less formal attire.21 Adamson et al23 published a response to the study’s findings urging dermatologists to recognize that a physician’s race and gender influence patients’ perceptions in much the same way that physician attire seems to and encouraged the development of a more diverse dermatologic workforce to help combat this prejudice. The issue of bias is not limited to the specialty of dermatology; the recent survey study by Xun et al15 found that respondents consistently rated female models garbed in physician attire as less professional than male model counterparts. Additionally, female models wearing white coats were mistakenly identified as medical technicians, physician assistants, or nurses with substantially more frequency than males, despite being clothed in the traditional physician garb. Several other publications on the subject have uncovered implicit bias, though it is rarely, if ever, the principle focus of the study.10,24,25 As is unfortunately true in many professions, female physicians and physicians from ethnic minorities face barriers to being perceived as fully competent physicians.
Impact of the COVID-19 Pandemic
Finally, of course, there is the ever-present question of the effect of the pandemic. Although the exact role of the white coat as a fomite for infection—and especially for the spread of viral illness—remains controversial, the perception nonetheless has helped catalyze the movement to alternatives such as short-sleeved white coats, technical jackets, and more recently, fitted scrubs.26-29 As with much in this realm, facts seem less important than perceptions; Zahrina et al30 found that when patients were presented with information regarding the risk for microbial contamination associated with white coats, preference for physicians in professional garb plummeted from 72% to only 22%. To date no articles have examined patient perceptions of the white coat in the context of microbial transmission in the age of COVID-19, but future articles on this topic are likely and may serve to further the demise of the white coat.
Final Thoughts
From my vantage point, it seems the white coat will be claimed by the outgoing tide. During this most recent residency interview season, I do not recall a single medical student wearing a short white coat. The closest I came was a quick glimpse of a crumpled white jacket slung over an arm or stuffed in a shoulder bag. Rotating interns and residents from other services on rotation in our department present in softshell or fleece jackets. Fitted scrubs in the newest trendy colors speckle a previously all-white canvas. I, for one, have not donned my own white coat in at least a year, and perhaps it is all for the best. Physician attire is one small aspect of the practice of medicine and likely bears little, if any, relation to the wearer’s qualifications. Our focus should be on building rapport with our patients, providing high-quality care, reducing the risk for nosocomial infection, and developing a health care system that is fair and equitable for patients and health care workers alike, not on who is wearing what. Perhaps the introduction of new physician attire is a small part of the disruption we need to help address persistent gender and racial biases in our field and help shepherd our patients and colleagues to a worldview that is more open and accepting of physicians of diverse backgrounds.
The White Coat Ceremony is an enduring memory from my medical school years. Amidst the tumult of memories of seemingly endless sleepless nights spent in libraries and cramming for clerkship examinations between surgical cases, I recall a sunny spring day in 2016 where I gathered with my classmates, family, and friends in the medical school campus courtyard. There were several short, mostly forgotten speeches after which proud fathers and mothers, partners, or siblings slipped the all-important white coat onto the shoulders of the physicians-to-be. At that moment, I felt the weight of tradition centuries in the making resting on my shoulders. Of course, the pomp of the ceremony might have felt a tad overblown had I known that the whole thing had fewer years under its belt than the movie Die Hard.
That’s right, the first White Coat Ceremony was held 5 years after the release of that Bruce Willis classic. Dr. Arnold Gold, a pediatric neurologist on faculty at Columbia University, conceived the ceremony in 1993, and it spread rapidly to medical schools—and later nursing schools—across the United States.1 Although the values highlighted by the White Coat Ceremony—humanism and compassion in medicine—are timeless, the ceremony itself is a more modern undertaking. What, then, of the white coat itself? Is it the timeless symbol of doctoring—of medicine—that we all presume it to be? Or is it a symbol of modern marketing, just a trend that caught on? And is it encountering its twilight—as trends often do—in the face of changing fashion and, more fundamentally, in changes to who our physicians are and to their roles in our society?
The Cleanliness of the White Coat
Until the end of the 19th century, physicians in the Western world most frequently dressed in black formal wear. The rationale behind this attire seems to have been twofold. First, society as a whole perceived the physician’s work as a serious and formal matter, and any medical encounter had to reflect the gravity of the occasion. Additionally, physicians’ visits often were a portent of impending demise, as physicians in the era prior to antibiotics and antisepsis frequently had little to offer their patients outside of—at best—anecdotal treatments and—at worst—sheer quackery.2 Black may have seemed a respectful choice for patients who likely faced dire outcomes regardless of the treatment afforded.3
With the turn of the century came a new understanding of the concepts of antisepsis and disease transmission. While Joseph Lister first published on the use of antisepsis in 1867, his practices did not become commonplace until the early 1900s.4 Around the same time came the Flexner report,5 the publication of William Osler’s Principles and Practice of Medicine,6 and the establishment of the modern medical residency, all of which contributed to the shift from the patient’s own bedside and to the hospital as the house of medicine, with cleanliness and antisepsis as part of its core principles.7 The white coat arose as a symbol of purity and freedom from disease. Throughout the 20th century and into the 21st, it has remained the predominant symbol of cleanliness and professionalism for the medical practitioner.
Patient Preference of Physician Attire
Although the white coat may serve as a professional symbol and is well respected medicine, it also plays an important role in the layperson’s perception of their health care providers.8 There is little denying that patients prefer their physicians, almost uniformly, to wear a white coat. A systematic review of physician attire that included 30 studies mainly from North America, Europe, and the United Kingdom found that patient preference for formal attire and white coats is near universal.9 Patients routinely rate physicians wearing a white coat as more intelligent and trustworthy and feel more confident in the care they will receive.10-13 They also freely admit that a physician’s appearance influences their satisfaction with their care.14 The recent adoption of the fleece, or softshell, jacket has not yet pervaded patients’ perceptions of what is considered appropriate physician attire. A 500-respondent survey found that patients were more likely to rate a model wearing a white coat as more professional and experienced compared to the same model wearing a fleece or softshell jacket or other formal attire sans white coat.15
Closer examination of the same data, however, reveals results reproduced with startling consistency across several studies, which suggest those of us adopting other attire need not dig those white coats out of the closet just yet. First, while many studies point to patient preference for white coats, this preference is uniformly strongest in older patients, beginning around age 40 years and becoming an entrenched preference in those older than 65 years.9,14,16-18 On the other hand, younger patient populations display little to no such preference, and some studies indicate that younger patients actually prefer scrubs over formal attire in specific settings such as surgical offices, procedural spaces, or the emergency department.12,14,19 This suggests that bias in favor of traditional physician garb may be more linked to age demographics and may continue to shift as the overall population ages. Additionally, although patients might profess a strong preference for physician attire in theory, it often does not translate into any impact on the patient’s perception of the physician following a clinic visit. The large systematic review on the topic noted that only 25% of studies that surveyed patients about a clinical visit following the encounter reported that physician attire influenced their satisfaction with that visit, suggesting that attire may be less likely to influence patients in the real-world context of receiving care.9 In fact, a prospective study of patient perception of medical staff and interactions found that staff style of dress not only had no bearing on the perception of staff or visit satisfaction but that patients often failed to even accurately recall physician attire when surveyed.20 Another survey study echoed these conclusions, finding that physician attire had no effect on the perception of a proposed treatment plan.21
What do we know about patient perception of physician attire in the dermatology setting specifically, where visits can be unique in their tendency to transition from medical to procedural in the span of a 15-minute encounter depending on the patient’s chief concern? A survey study of dermatology patients at the general, surgical, and wound care dermatology clinics of an academic medical center (Miami, Florida) found that professional attire with a white coat was strongly preferred across a litany of scenarios assessing many aspects of dermatologic care.21 Similarly, a study of patients visiting a single institution’s dermatology and pediatric dermatology clinics surveyed patients and parents regarding attire prior to an appointment and specifically asked if a white coat should be worn.13 Fifty-four percent of the adult patients (n=176) surveyed professed a preference for physicians in white coats, with a stronger preference for white coats reported by those 50 years and older (55%; n=113). Parents or guardians presenting to the pediatric dermatology clinic, on the other hand, favored less formal attire.13 A recent, real-world study performed at an outpatient dermatology clinic examined the influence of changing physician attire on a patient’s perceptions of care received during clinic encounters. They found no substantial difference in patient satisfaction scores before and following the adoption of a new clinic uniform that transitioned from formal attire to fitted scrubs.22
Racial and Gender Bias Affecting Attire Preference
With any study of preference, there is the underlying concern over respondent bias. Many of the studies discussed here have found secondarily that a patient’s implicit bias does not end at the clothes their physician is wearing. The survey study of dermatology patients from the academic medical center in Miami, Florida, found that patients preferred that Black physicians of either sex be garbed in professional attire at all times but generally were more accepting of White physicians in less formal attire.21 Adamson et al23 published a response to the study’s findings urging dermatologists to recognize that a physician’s race and gender influence patients’ perceptions in much the same way that physician attire seems to and encouraged the development of a more diverse dermatologic workforce to help combat this prejudice. The issue of bias is not limited to the specialty of dermatology; the recent survey study by Xun et al15 found that respondents consistently rated female models garbed in physician attire as less professional than male model counterparts. Additionally, female models wearing white coats were mistakenly identified as medical technicians, physician assistants, or nurses with substantially more frequency than males, despite being clothed in the traditional physician garb. Several other publications on the subject have uncovered implicit bias, though it is rarely, if ever, the principle focus of the study.10,24,25 As is unfortunately true in many professions, female physicians and physicians from ethnic minorities face barriers to being perceived as fully competent physicians.
Impact of the COVID-19 Pandemic
Finally, of course, there is the ever-present question of the effect of the pandemic. Although the exact role of the white coat as a fomite for infection—and especially for the spread of viral illness—remains controversial, the perception nonetheless has helped catalyze the movement to alternatives such as short-sleeved white coats, technical jackets, and more recently, fitted scrubs.26-29 As with much in this realm, facts seem less important than perceptions; Zahrina et al30 found that when patients were presented with information regarding the risk for microbial contamination associated with white coats, preference for physicians in professional garb plummeted from 72% to only 22%. To date no articles have examined patient perceptions of the white coat in the context of microbial transmission in the age of COVID-19, but future articles on this topic are likely and may serve to further the demise of the white coat.
Final Thoughts
From my vantage point, it seems the white coat will be claimed by the outgoing tide. During this most recent residency interview season, I do not recall a single medical student wearing a short white coat. The closest I came was a quick glimpse of a crumpled white jacket slung over an arm or stuffed in a shoulder bag. Rotating interns and residents from other services on rotation in our department present in softshell or fleece jackets. Fitted scrubs in the newest trendy colors speckle a previously all-white canvas. I, for one, have not donned my own white coat in at least a year, and perhaps it is all for the best. Physician attire is one small aspect of the practice of medicine and likely bears little, if any, relation to the wearer’s qualifications. Our focus should be on building rapport with our patients, providing high-quality care, reducing the risk for nosocomial infection, and developing a health care system that is fair and equitable for patients and health care workers alike, not on who is wearing what. Perhaps the introduction of new physician attire is a small part of the disruption we need to help address persistent gender and racial biases in our field and help shepherd our patients and colleagues to a worldview that is more open and accepting of physicians of diverse backgrounds.
- White Coat Ceremony. Gold Foundation website. Accessed December 26, 2021. https://www.gold-foundation.org/programs/white-coat-ceremony/
- Shryock RH. The Development of Modern Medicine. University of Pennsylvania Press; 2017.
- Hochberg MS. The doctor’s white coat—an historical perspective. Virtual Mentor. 2007;9:310-314.
- Lister J. On the antiseptic principle in the practice of surgery. Lancet. 1867;90:353-356.
- Flexner A. Medical Education in the United States and Canada: A Report to the Carnegie Foundation for the Advancement of Teaching. Carnegie Foundation for the Advancement of Teaching; 1910.
- Osler W. Principles and Practice of Medicine: Designed for the Use of Practitioners and Students of Medicine. D. Appleton & Company; 1892.
- Blumhagen DW. The doctor’s white coat: the image of the physician in modern America. Ann Intern Med. 1979;91:111-116.
- Verghese BG, Kashinath SK, Jadhav N, et al. Physician attire: physicians’ perspectives on attire in a community hospital setting among non-surgical specialties. J Community Hosp Intern Med Perspect. 2020;10:1-5.
- Petrilli CM, Mack M, Petrilli JJ, et al. Understanding the role of physician attire on patient perceptions: a systematic review of the literature—targeting attire to improve likelihood of rapport (TAILOR) investigators. BMJ Open. 2015;5:E006678.
- Rehman SU, Nietert PJ, Cope DW, et al. What to wear today? effect of doctor’s attire on the trust and confidence of patients. Am J Med. 2005;118:1279-1286.
- Jennings JD, Ciaravino SG, Ramsey FV, et al. Physicians’ attire influences patients’ perceptions in the urban outpatient orthopaedic surgery setting. Clin Orthop Relat Res. 2016;474:1908-1918.
- Gherardi G, Cameron J, West A, et al. Are we dressed to impress? a descriptive survey assessing patients preference of doctors’ attire in the hospital setting. Clin Med (Lond). 2009;9:519-524.
- Thomas MW, Burkhart CN, Lugo-Somolinos A, et al. Patients’ perceptions of physician attire in dermatology clinics. Arch Dermatol. 2011;147:505-506.
- Petrilli CM, Saint S, Jennings JJ, et al. Understanding patient preference for physician attire: a cross-sectional observational study of 10 academic medical centres in the USA. BMJ Open. 2018;8:E021239.
- Xun H, Chen J, Sun AH, et al. Public perceptions of physician attire and professionalism in the US. JAMA Network Open. 2021;4:E2117779.
- Kamata K, Kuriyama A, Chopra V, et al. Patient preferences for physician attire: a multicenter study in Japan [published online February 11, 2020]. J Hosp Med. 2020;15:204-210.
- Budny AM, Rogers LC, Mandracchia VJ, et al. The physician’s attire and its influence on patient confidence. J Am Podiatr Assoc. 2006;96:132-138.
- Lill MM, Wilkinson TJ. Judging a book by its cover: descriptive survey of patients’ preferences for doctors’ appearance and mode of address. Br Med J. 2005;331:1524-1527.
- Hossler EW, Shipp D, Palmer M, et al. Impact of provider attire on patient satisfaction in an outpatient dermatology clinic. Cutis. 2018;102:127-129.
- Boon D, Wardrope J. What should doctors wear in the accident and emergency department? patients’ perception. J Accid Emerg Med. 1994;11:175-177.
- Fox JD, Prado G, Baquerizo Nole KL, et al. Patient preference in dermatologist attire in the medical, surgical, and wound care settings. JAMA Dermatol. 2016;152:913-919.
- Bray JK, Porter C, Feldman SR. The effect of physician appearance on patient perceptions of treatment plans. Dermatol Online J. 2021;27. doi:10.5070/D327553611
- Adamson AS, Wright SW, Pandya AG. A missed opportunity to discuss racial and gender bias in dermatology. JAMA Dermatol. 2017;153:110-111.
- Hartmans C, Heremans S, Lagrain M, et al. The doctor’s new clothes: professional or fashionable? Primary Health Care. 2013;3:135.
- Kurihara H, Maeno T, Maeno T. Importance of physicians’ attire: factors influencing the impression it makes on patients, a cross-sectional study. Asia Pac Fam Med. 2014;13:2.
- Treakle AM, Thom KA, Furuno JP, et al. Bacterial contamination of health care workers’ white coats. Am J Infect Control. 2009;37:101-105.
- Banu A, Anand M, Nagi N, et al. White coats as a vehicle for bacterial dissemination. J Clin Diagn Res. 2012;6:1381-1384.
- Haun N, Hooper-Lane C, Safdar N. Healthcare personnel attire and devices as fomites: a systematic review. Infect Control Hosp Epidemiol. 2016;37:1367-1373.
- Tse G, Withey S, Yeo JM, et al. Bare below the elbows: was the target the white coat? J Hosp Infect. 2015;91:299-301.
- Zahrina AZ, Haymond P, Rosanna P, et al. Does the attire of a primary care physician affect patients’ perceptions and their levels of trust in the doctor? Malays Fam Physician. 2018;13:3-11.
- White Coat Ceremony. Gold Foundation website. Accessed December 26, 2021. https://www.gold-foundation.org/programs/white-coat-ceremony/
- Shryock RH. The Development of Modern Medicine. University of Pennsylvania Press; 2017.
- Hochberg MS. The doctor’s white coat—an historical perspective. Virtual Mentor. 2007;9:310-314.
- Lister J. On the antiseptic principle in the practice of surgery. Lancet. 1867;90:353-356.
- Flexner A. Medical Education in the United States and Canada: A Report to the Carnegie Foundation for the Advancement of Teaching. Carnegie Foundation for the Advancement of Teaching; 1910.
- Osler W. Principles and Practice of Medicine: Designed for the Use of Practitioners and Students of Medicine. D. Appleton & Company; 1892.
- Blumhagen DW. The doctor’s white coat: the image of the physician in modern America. Ann Intern Med. 1979;91:111-116.
- Verghese BG, Kashinath SK, Jadhav N, et al. Physician attire: physicians’ perspectives on attire in a community hospital setting among non-surgical specialties. J Community Hosp Intern Med Perspect. 2020;10:1-5.
- Petrilli CM, Mack M, Petrilli JJ, et al. Understanding the role of physician attire on patient perceptions: a systematic review of the literature—targeting attire to improve likelihood of rapport (TAILOR) investigators. BMJ Open. 2015;5:E006678.
- Rehman SU, Nietert PJ, Cope DW, et al. What to wear today? effect of doctor’s attire on the trust and confidence of patients. Am J Med. 2005;118:1279-1286.
- Jennings JD, Ciaravino SG, Ramsey FV, et al. Physicians’ attire influences patients’ perceptions in the urban outpatient orthopaedic surgery setting. Clin Orthop Relat Res. 2016;474:1908-1918.
- Gherardi G, Cameron J, West A, et al. Are we dressed to impress? a descriptive survey assessing patients preference of doctors’ attire in the hospital setting. Clin Med (Lond). 2009;9:519-524.
- Thomas MW, Burkhart CN, Lugo-Somolinos A, et al. Patients’ perceptions of physician attire in dermatology clinics. Arch Dermatol. 2011;147:505-506.
- Petrilli CM, Saint S, Jennings JJ, et al. Understanding patient preference for physician attire: a cross-sectional observational study of 10 academic medical centres in the USA. BMJ Open. 2018;8:E021239.
- Xun H, Chen J, Sun AH, et al. Public perceptions of physician attire and professionalism in the US. JAMA Network Open. 2021;4:E2117779.
- Kamata K, Kuriyama A, Chopra V, et al. Patient preferences for physician attire: a multicenter study in Japan [published online February 11, 2020]. J Hosp Med. 2020;15:204-210.
- Budny AM, Rogers LC, Mandracchia VJ, et al. The physician’s attire and its influence on patient confidence. J Am Podiatr Assoc. 2006;96:132-138.
- Lill MM, Wilkinson TJ. Judging a book by its cover: descriptive survey of patients’ preferences for doctors’ appearance and mode of address. Br Med J. 2005;331:1524-1527.
- Hossler EW, Shipp D, Palmer M, et al. Impact of provider attire on patient satisfaction in an outpatient dermatology clinic. Cutis. 2018;102:127-129.
- Boon D, Wardrope J. What should doctors wear in the accident and emergency department? patients’ perception. J Accid Emerg Med. 1994;11:175-177.
- Fox JD, Prado G, Baquerizo Nole KL, et al. Patient preference in dermatologist attire in the medical, surgical, and wound care settings. JAMA Dermatol. 2016;152:913-919.
- Bray JK, Porter C, Feldman SR. The effect of physician appearance on patient perceptions of treatment plans. Dermatol Online J. 2021;27. doi:10.5070/D327553611
- Adamson AS, Wright SW, Pandya AG. A missed opportunity to discuss racial and gender bias in dermatology. JAMA Dermatol. 2017;153:110-111.
- Hartmans C, Heremans S, Lagrain M, et al. The doctor’s new clothes: professional or fashionable? Primary Health Care. 2013;3:135.
- Kurihara H, Maeno T, Maeno T. Importance of physicians’ attire: factors influencing the impression it makes on patients, a cross-sectional study. Asia Pac Fam Med. 2014;13:2.
- Treakle AM, Thom KA, Furuno JP, et al. Bacterial contamination of health care workers’ white coats. Am J Infect Control. 2009;37:101-105.
- Banu A, Anand M, Nagi N, et al. White coats as a vehicle for bacterial dissemination. J Clin Diagn Res. 2012;6:1381-1384.
- Haun N, Hooper-Lane C, Safdar N. Healthcare personnel attire and devices as fomites: a systematic review. Infect Control Hosp Epidemiol. 2016;37:1367-1373.
- Tse G, Withey S, Yeo JM, et al. Bare below the elbows: was the target the white coat? J Hosp Infect. 2015;91:299-301.
- Zahrina AZ, Haymond P, Rosanna P, et al. Does the attire of a primary care physician affect patients’ perceptions and their levels of trust in the doctor? Malays Fam Physician. 2018;13:3-11.
Resident Pearls
- Until the end of the 19th century, Western physicians most commonly wore black formal wear. The rise of the physician’s white coat occurred in conjunction with the shift to hospital medicine.
- Patient surveys repeatedly have demonstrated a preference for physicians to wear white coats; whether or not this has any bearing on patient satisfaction in real-world scenarios is less clear.
- The impact of the COVID-19 pandemic on trends in white coat wear has not yet been elucidated.
Prior authorization abuse: It’s time for health insurance CEOs and their proxies to cease and desist the practice once and for all!
Before reading this editorial and concluding that the author (me) has lost his grip on reality, I would ask that you consider the facts I provide below and the ramifications incurred by your patients and practices, due to the misbehaviors adopted by the health insurance industry.
- Two of the most common issues discussed in today’s health care environment are revenue generation and provider/staff burnout.
While these issues are impacted by several factors, one of the most common denominators is increasing administrative workloads driven by non–revenue-generating activities. Consider this:
- A recent American Medical Association survey pointed out that during the course of the average workweek, a physician completes an average of 37 prior authorization requests. Physicians and their staff spend an average of 16.4 hours per week completing prior authorization requirements for patient medicines, procedures, and medical services that they may need.1
- While physicians report that about 65% of prior authorizations take only 1 day, they report that 26% take 3 or more days.2
The potential significance of the generated delays
While this may not seem like a long time (other than the impact it has on staff workload), consider the impact this can have on the patient if the medication being requested is: PrEP, the morning after pill, or other contraceptives? The consequences of the delay or denial could be a lifetime living with HIV, or an unintended pregnancy. This is to say nothing on the larger impact to family, partners, and the potential social stigma faced by all.
Beyond the personal costs and costs within your practice associated with the additional workload, consider the financial costs. The average cost to complete a prior authorization remains the single highest cost for the health care industry at $13.40 per manual transaction, and $7.19 per partially electronic web portal transaction,3 meaning that if I did only one prescription per week, I probably would not mind, but at $13.40 per prior authorization, this burden amounts to millions, actually $767 million by recent estimates.3 Additionally, if you factor in the number of denials and potential follow-ups, this creates a significant amount of waste and spending.
Ultimately, in my experience, I have found that most prior authorizations are simply unnecessary. Here, I’ve picked key examples from just my own recent experiences:
- My patient was denied access to a particular birth control pill she had been on successfully before, and my office was told she needed to try and fail on 5 different generic pills before she could be approved. However, the Affordable Care Act’s (ACA; aka Obamacare) Contraceptive Mandate requires coverage of all contraceptives determined to be most appropriate between a patient and their provider (see below).
- A menopausal patient was denied coverage twice (electronically) for generic micronized progesterone, and I was asked to write a letter of appeal because the insurance company wanted me to use medroxyprogesterone acetate instead. Polling my nearby retail independent pharmacy, the total cost difference per year was $19.96 savings/year ($47.01 ‒ $27.05 = $19.96). My pharmacist did note it could have been a different amount at a large chain pharmacy. Really? I had to write a letter, following two denials, to save less than $20, for a full year!
- A 78-year-old patient using Prolia for severe osteoporosis and preexisting fractures was delayed in getting her next Prolia injection due to a prior authorization snafu. She ended up with multiple additional fractures, a well-described effect of the increase in bone turnover when stopping or delaying this medication. She is now disabled.
- A 94-year-old patient was sent an email reminder to get the medical practice to authorize a refill of ileostomy bags. The email went to spam, and the patient ran out of bags prior to a holiday weekend. I got them in 2 days on Amazon Prime. But who emails a 94-year-old? And ileostomy bags! When does anyone stop needing ileostomy bags?
- I requested a prior authorization for Orilissa (clearly off label) because a severely progestogen-sensitive patient (augmented depression) with severe premenstrual dysphoric disorder requiring hospitalization was thought by her psychiatrist to be better off without menstrual periods. I completed the proper paperwork, two electronic appeals, and a letter of explanation including available references on the use of gonadotropin-releasing hormone analogues for such patients. I was then told I would need to have a peer-to-peer discussion, so I filled out that paperwork, which clearly noted that I am a board-certified reproductive endocrinologist. I got a phone call a few days later by a pleasant, young-sounding pediatric rheumatologist. Our interaction did not go well for him. This was not peer-to-peer!
Let us be clear, prior authorizations have nothing to do with patient care. In fact, they are solely about the money. We in ObGyn have mostly inexpensive and generic products, but even that fact has not lowered the excessive burden of the prior authorization process. In the case of contraception, whether you like the ACA or not it is the law, and it contains specific provisions regarding contraception. With the goals of providing broad access to patients and incentives to developers for new and novel contraceptive methods, these provisions require insurers to cover, without cost-sharing, women’s preventive services including the full range of FDA-approved contraceptives (currently 18 different method categories), and additional methods identified by the FDA as they become available. Further, providers must have an easily accessible, transparent, and sufficiently expedient exceptions process that is not unduly burdensome on the individual or a provider (or other individuals acting as a patient’s authorized representative).
And while I can regale you with chapter and verse and citations of the legal precedent and language, it boils down to this:
- The AMA reported that medical practices spend an average of 2 business days a week per physician to comply with health plans’ inefficient and overused prior-authorization protocols.4 To keep up with the administrative burden, 2 out of 5 physicians (40%) employ staff members who work exclusively on tasks associated with prior authorization.4
- About 86% of practices reported an increased burden of prior authorizations in the last 5 years.5
Continue to: What is to be done?
What is to be done?
I do have suggested solutions. Given the insurance industry’s complete lack of progress in voluntarily reducing the burdens of prior authorizations agreed to in their consensus statement with the AMA, American Hospital Association, America’s Health Insurance Plans, American Pharmacists Association, Blue Cross Blue Shield Association, and the Medical Group Management Association, I say, why not fine them? The AMA is calling on Congress to pass legislation that would codify much of the agreement, in which the above parties had already agreed that reforms were needed to reduce prior authorization burdens and enhance patient-centered care.6
A good model for enforcement via fines could be based on the old “incident to” rules of Medicare. These state that a physician needs to be “in the space” when advanced practice nurses or physician assistants see Medicare recipients. If they are not actually “in the space” they are subject to a fine. As a completely theoretical example, let’s say the claim was for $100. The practitioner would have to pay it back plus triple that amount in damages, or $400. They can also be fined up to $11,000 per claim and kick you out of Medicare and Medicaid. Take my example of Prolia from above…a single shot of Prolia is about $1,000. The insurer would theoretically have to pay $14,000/claim (the claim + triple damages + $11,000) if it was determined that the prior authorization was unnecessary. Seems about right to me. Or we could just sit the health insurance CEOs and their proxies in the corner on 2-foot-tall plastic Little Tikes® chairs for a “timeout” (dunce cap optional), like the outset of the article says.
Until the detrimental prior authorization process is challenged at all levels, we will continue to see and feel the effects of the harm it causes. Being able to drive change through advocacy and education is the best way we as clinicians can impact not just the future of health care but provide for the daily care of our patients who depend on and trust us to provide for their medical needs. We must be the impactors of change for ourselves, colleagues, staff, and profession if we are to really make advancements into the future.
Oh…and health insurance CEOs and their proxies, to get out of their “time-out” would still be entitled to one phone call to beg forgiveness from their mommies/daddies, priest/ rabbi/pastor, psychologist/psychiatrist/mystic healer, etc., but alas, the average wait time is an hour, and if anyone answers the phone, they have a grade school education used in following an irrelevant algorithm. ●
- Corder JC. Streamlining the insurance prior authorization debacle. Mo Med. 2018;115:312-314.
- Prior authorization hurdles have led to serious adverse events. American Medical Association website. February 5, 2019. https://www.ama-assn .org/press-center/press-releases/prior-author ization-hurdles-have-led-serious-adverse -events. Accessed November 29, 2021.
- Council for Affordable Quality Healthcare. 2020 CAQH INDEX. https://www.caqh.org/sites /default/files/explorations/index/2020-caqh -index.pdf. Accessed November 22, 2021.
- Most physicians had little relief from prior authorization as COVID cases soared. American Medical Association website. April 7, 2021. https:// www.ama-assn.org/press-center/press-releases /most-physicians-had-little-relief-prior-author ization-covid-cases. Accessed November 29, 2021.
- Robeznieks A. 1 in 4 doctors say prior authorization has led to a serious adverse event. American Medical Association website. February 5, 2019. https://www .ama-assn.org/practice-management/sustainability /1-4-doctors-say-prior-authorization-has-led-serious -adverse. Accessed November 29, 2021.
- Physicians call on Congress to address prior authorization reform. American Medical Association website. May 14, 2021. https://www .ama-assn.org/press-center/press-releases /physicians-call-congress-address-prior-author ization-reform. Accessed November 29, 2021.
Before reading this editorial and concluding that the author (me) has lost his grip on reality, I would ask that you consider the facts I provide below and the ramifications incurred by your patients and practices, due to the misbehaviors adopted by the health insurance industry.
- Two of the most common issues discussed in today’s health care environment are revenue generation and provider/staff burnout.
While these issues are impacted by several factors, one of the most common denominators is increasing administrative workloads driven by non–revenue-generating activities. Consider this:
- A recent American Medical Association survey pointed out that during the course of the average workweek, a physician completes an average of 37 prior authorization requests. Physicians and their staff spend an average of 16.4 hours per week completing prior authorization requirements for patient medicines, procedures, and medical services that they may need.1
- While physicians report that about 65% of prior authorizations take only 1 day, they report that 26% take 3 or more days.2
The potential significance of the generated delays
While this may not seem like a long time (other than the impact it has on staff workload), consider the impact this can have on the patient if the medication being requested is: PrEP, the morning after pill, or other contraceptives? The consequences of the delay or denial could be a lifetime living with HIV, or an unintended pregnancy. This is to say nothing on the larger impact to family, partners, and the potential social stigma faced by all.
Beyond the personal costs and costs within your practice associated with the additional workload, consider the financial costs. The average cost to complete a prior authorization remains the single highest cost for the health care industry at $13.40 per manual transaction, and $7.19 per partially electronic web portal transaction,3 meaning that if I did only one prescription per week, I probably would not mind, but at $13.40 per prior authorization, this burden amounts to millions, actually $767 million by recent estimates.3 Additionally, if you factor in the number of denials and potential follow-ups, this creates a significant amount of waste and spending.
Ultimately, in my experience, I have found that most prior authorizations are simply unnecessary. Here, I’ve picked key examples from just my own recent experiences:
- My patient was denied access to a particular birth control pill she had been on successfully before, and my office was told she needed to try and fail on 5 different generic pills before she could be approved. However, the Affordable Care Act’s (ACA; aka Obamacare) Contraceptive Mandate requires coverage of all contraceptives determined to be most appropriate between a patient and their provider (see below).
- A menopausal patient was denied coverage twice (electronically) for generic micronized progesterone, and I was asked to write a letter of appeal because the insurance company wanted me to use medroxyprogesterone acetate instead. Polling my nearby retail independent pharmacy, the total cost difference per year was $19.96 savings/year ($47.01 ‒ $27.05 = $19.96). My pharmacist did note it could have been a different amount at a large chain pharmacy. Really? I had to write a letter, following two denials, to save less than $20, for a full year!
- A 78-year-old patient using Prolia for severe osteoporosis and preexisting fractures was delayed in getting her next Prolia injection due to a prior authorization snafu. She ended up with multiple additional fractures, a well-described effect of the increase in bone turnover when stopping or delaying this medication. She is now disabled.
- A 94-year-old patient was sent an email reminder to get the medical practice to authorize a refill of ileostomy bags. The email went to spam, and the patient ran out of bags prior to a holiday weekend. I got them in 2 days on Amazon Prime. But who emails a 94-year-old? And ileostomy bags! When does anyone stop needing ileostomy bags?
- I requested a prior authorization for Orilissa (clearly off label) because a severely progestogen-sensitive patient (augmented depression) with severe premenstrual dysphoric disorder requiring hospitalization was thought by her psychiatrist to be better off without menstrual periods. I completed the proper paperwork, two electronic appeals, and a letter of explanation including available references on the use of gonadotropin-releasing hormone analogues for such patients. I was then told I would need to have a peer-to-peer discussion, so I filled out that paperwork, which clearly noted that I am a board-certified reproductive endocrinologist. I got a phone call a few days later by a pleasant, young-sounding pediatric rheumatologist. Our interaction did not go well for him. This was not peer-to-peer!
Let us be clear, prior authorizations have nothing to do with patient care. In fact, they are solely about the money. We in ObGyn have mostly inexpensive and generic products, but even that fact has not lowered the excessive burden of the prior authorization process. In the case of contraception, whether you like the ACA or not it is the law, and it contains specific provisions regarding contraception. With the goals of providing broad access to patients and incentives to developers for new and novel contraceptive methods, these provisions require insurers to cover, without cost-sharing, women’s preventive services including the full range of FDA-approved contraceptives (currently 18 different method categories), and additional methods identified by the FDA as they become available. Further, providers must have an easily accessible, transparent, and sufficiently expedient exceptions process that is not unduly burdensome on the individual or a provider (or other individuals acting as a patient’s authorized representative).
And while I can regale you with chapter and verse and citations of the legal precedent and language, it boils down to this:
- The AMA reported that medical practices spend an average of 2 business days a week per physician to comply with health plans’ inefficient and overused prior-authorization protocols.4 To keep up with the administrative burden, 2 out of 5 physicians (40%) employ staff members who work exclusively on tasks associated with prior authorization.4
- About 86% of practices reported an increased burden of prior authorizations in the last 5 years.5
Continue to: What is to be done?
What is to be done?
I do have suggested solutions. Given the insurance industry’s complete lack of progress in voluntarily reducing the burdens of prior authorizations agreed to in their consensus statement with the AMA, American Hospital Association, America’s Health Insurance Plans, American Pharmacists Association, Blue Cross Blue Shield Association, and the Medical Group Management Association, I say, why not fine them? The AMA is calling on Congress to pass legislation that would codify much of the agreement, in which the above parties had already agreed that reforms were needed to reduce prior authorization burdens and enhance patient-centered care.6
A good model for enforcement via fines could be based on the old “incident to” rules of Medicare. These state that a physician needs to be “in the space” when advanced practice nurses or physician assistants see Medicare recipients. If they are not actually “in the space” they are subject to a fine. As a completely theoretical example, let’s say the claim was for $100. The practitioner would have to pay it back plus triple that amount in damages, or $400. They can also be fined up to $11,000 per claim and kick you out of Medicare and Medicaid. Take my example of Prolia from above…a single shot of Prolia is about $1,000. The insurer would theoretically have to pay $14,000/claim (the claim + triple damages + $11,000) if it was determined that the prior authorization was unnecessary. Seems about right to me. Or we could just sit the health insurance CEOs and their proxies in the corner on 2-foot-tall plastic Little Tikes® chairs for a “timeout” (dunce cap optional), like the outset of the article says.
Until the detrimental prior authorization process is challenged at all levels, we will continue to see and feel the effects of the harm it causes. Being able to drive change through advocacy and education is the best way we as clinicians can impact not just the future of health care but provide for the daily care of our patients who depend on and trust us to provide for their medical needs. We must be the impactors of change for ourselves, colleagues, staff, and profession if we are to really make advancements into the future.
Oh…and health insurance CEOs and their proxies, to get out of their “time-out” would still be entitled to one phone call to beg forgiveness from their mommies/daddies, priest/ rabbi/pastor, psychologist/psychiatrist/mystic healer, etc., but alas, the average wait time is an hour, and if anyone answers the phone, they have a grade school education used in following an irrelevant algorithm. ●
Before reading this editorial and concluding that the author (me) has lost his grip on reality, I would ask that you consider the facts I provide below and the ramifications incurred by your patients and practices, due to the misbehaviors adopted by the health insurance industry.
- Two of the most common issues discussed in today’s health care environment are revenue generation and provider/staff burnout.
While these issues are impacted by several factors, one of the most common denominators is increasing administrative workloads driven by non–revenue-generating activities. Consider this:
- A recent American Medical Association survey pointed out that during the course of the average workweek, a physician completes an average of 37 prior authorization requests. Physicians and their staff spend an average of 16.4 hours per week completing prior authorization requirements for patient medicines, procedures, and medical services that they may need.1
- While physicians report that about 65% of prior authorizations take only 1 day, they report that 26% take 3 or more days.2
The potential significance of the generated delays
While this may not seem like a long time (other than the impact it has on staff workload), consider the impact this can have on the patient if the medication being requested is: PrEP, the morning after pill, or other contraceptives? The consequences of the delay or denial could be a lifetime living with HIV, or an unintended pregnancy. This is to say nothing on the larger impact to family, partners, and the potential social stigma faced by all.
Beyond the personal costs and costs within your practice associated with the additional workload, consider the financial costs. The average cost to complete a prior authorization remains the single highest cost for the health care industry at $13.40 per manual transaction, and $7.19 per partially electronic web portal transaction,3 meaning that if I did only one prescription per week, I probably would not mind, but at $13.40 per prior authorization, this burden amounts to millions, actually $767 million by recent estimates.3 Additionally, if you factor in the number of denials and potential follow-ups, this creates a significant amount of waste and spending.
Ultimately, in my experience, I have found that most prior authorizations are simply unnecessary. Here, I’ve picked key examples from just my own recent experiences:
- My patient was denied access to a particular birth control pill she had been on successfully before, and my office was told she needed to try and fail on 5 different generic pills before she could be approved. However, the Affordable Care Act’s (ACA; aka Obamacare) Contraceptive Mandate requires coverage of all contraceptives determined to be most appropriate between a patient and their provider (see below).
- A menopausal patient was denied coverage twice (electronically) for generic micronized progesterone, and I was asked to write a letter of appeal because the insurance company wanted me to use medroxyprogesterone acetate instead. Polling my nearby retail independent pharmacy, the total cost difference per year was $19.96 savings/year ($47.01 ‒ $27.05 = $19.96). My pharmacist did note it could have been a different amount at a large chain pharmacy. Really? I had to write a letter, following two denials, to save less than $20, for a full year!
- A 78-year-old patient using Prolia for severe osteoporosis and preexisting fractures was delayed in getting her next Prolia injection due to a prior authorization snafu. She ended up with multiple additional fractures, a well-described effect of the increase in bone turnover when stopping or delaying this medication. She is now disabled.
- A 94-year-old patient was sent an email reminder to get the medical practice to authorize a refill of ileostomy bags. The email went to spam, and the patient ran out of bags prior to a holiday weekend. I got them in 2 days on Amazon Prime. But who emails a 94-year-old? And ileostomy bags! When does anyone stop needing ileostomy bags?
- I requested a prior authorization for Orilissa (clearly off label) because a severely progestogen-sensitive patient (augmented depression) with severe premenstrual dysphoric disorder requiring hospitalization was thought by her psychiatrist to be better off without menstrual periods. I completed the proper paperwork, two electronic appeals, and a letter of explanation including available references on the use of gonadotropin-releasing hormone analogues for such patients. I was then told I would need to have a peer-to-peer discussion, so I filled out that paperwork, which clearly noted that I am a board-certified reproductive endocrinologist. I got a phone call a few days later by a pleasant, young-sounding pediatric rheumatologist. Our interaction did not go well for him. This was not peer-to-peer!
Let us be clear, prior authorizations have nothing to do with patient care. In fact, they are solely about the money. We in ObGyn have mostly inexpensive and generic products, but even that fact has not lowered the excessive burden of the prior authorization process. In the case of contraception, whether you like the ACA or not it is the law, and it contains specific provisions regarding contraception. With the goals of providing broad access to patients and incentives to developers for new and novel contraceptive methods, these provisions require insurers to cover, without cost-sharing, women’s preventive services including the full range of FDA-approved contraceptives (currently 18 different method categories), and additional methods identified by the FDA as they become available. Further, providers must have an easily accessible, transparent, and sufficiently expedient exceptions process that is not unduly burdensome on the individual or a provider (or other individuals acting as a patient’s authorized representative).
And while I can regale you with chapter and verse and citations of the legal precedent and language, it boils down to this:
- The AMA reported that medical practices spend an average of 2 business days a week per physician to comply with health plans’ inefficient and overused prior-authorization protocols.4 To keep up with the administrative burden, 2 out of 5 physicians (40%) employ staff members who work exclusively on tasks associated with prior authorization.4
- About 86% of practices reported an increased burden of prior authorizations in the last 5 years.5
Continue to: What is to be done?
What is to be done?
I do have suggested solutions. Given the insurance industry’s complete lack of progress in voluntarily reducing the burdens of prior authorizations agreed to in their consensus statement with the AMA, American Hospital Association, America’s Health Insurance Plans, American Pharmacists Association, Blue Cross Blue Shield Association, and the Medical Group Management Association, I say, why not fine them? The AMA is calling on Congress to pass legislation that would codify much of the agreement, in which the above parties had already agreed that reforms were needed to reduce prior authorization burdens and enhance patient-centered care.6
A good model for enforcement via fines could be based on the old “incident to” rules of Medicare. These state that a physician needs to be “in the space” when advanced practice nurses or physician assistants see Medicare recipients. If they are not actually “in the space” they are subject to a fine. As a completely theoretical example, let’s say the claim was for $100. The practitioner would have to pay it back plus triple that amount in damages, or $400. They can also be fined up to $11,000 per claim and kick you out of Medicare and Medicaid. Take my example of Prolia from above…a single shot of Prolia is about $1,000. The insurer would theoretically have to pay $14,000/claim (the claim + triple damages + $11,000) if it was determined that the prior authorization was unnecessary. Seems about right to me. Or we could just sit the health insurance CEOs and their proxies in the corner on 2-foot-tall plastic Little Tikes® chairs for a “timeout” (dunce cap optional), like the outset of the article says.
Until the detrimental prior authorization process is challenged at all levels, we will continue to see and feel the effects of the harm it causes. Being able to drive change through advocacy and education is the best way we as clinicians can impact not just the future of health care but provide for the daily care of our patients who depend on and trust us to provide for their medical needs. We must be the impactors of change for ourselves, colleagues, staff, and profession if we are to really make advancements into the future.
Oh…and health insurance CEOs and their proxies, to get out of their “time-out” would still be entitled to one phone call to beg forgiveness from their mommies/daddies, priest/ rabbi/pastor, psychologist/psychiatrist/mystic healer, etc., but alas, the average wait time is an hour, and if anyone answers the phone, they have a grade school education used in following an irrelevant algorithm. ●
- Corder JC. Streamlining the insurance prior authorization debacle. Mo Med. 2018;115:312-314.
- Prior authorization hurdles have led to serious adverse events. American Medical Association website. February 5, 2019. https://www.ama-assn .org/press-center/press-releases/prior-author ization-hurdles-have-led-serious-adverse -events. Accessed November 29, 2021.
- Council for Affordable Quality Healthcare. 2020 CAQH INDEX. https://www.caqh.org/sites /default/files/explorations/index/2020-caqh -index.pdf. Accessed November 22, 2021.
- Most physicians had little relief from prior authorization as COVID cases soared. American Medical Association website. April 7, 2021. https:// www.ama-assn.org/press-center/press-releases /most-physicians-had-little-relief-prior-author ization-covid-cases. Accessed November 29, 2021.
- Robeznieks A. 1 in 4 doctors say prior authorization has led to a serious adverse event. American Medical Association website. February 5, 2019. https://www .ama-assn.org/practice-management/sustainability /1-4-doctors-say-prior-authorization-has-led-serious -adverse. Accessed November 29, 2021.
- Physicians call on Congress to address prior authorization reform. American Medical Association website. May 14, 2021. https://www .ama-assn.org/press-center/press-releases /physicians-call-congress-address-prior-author ization-reform. Accessed November 29, 2021.
- Corder JC. Streamlining the insurance prior authorization debacle. Mo Med. 2018;115:312-314.
- Prior authorization hurdles have led to serious adverse events. American Medical Association website. February 5, 2019. https://www.ama-assn .org/press-center/press-releases/prior-author ization-hurdles-have-led-serious-adverse -events. Accessed November 29, 2021.
- Council for Affordable Quality Healthcare. 2020 CAQH INDEX. https://www.caqh.org/sites /default/files/explorations/index/2020-caqh -index.pdf. Accessed November 22, 2021.
- Most physicians had little relief from prior authorization as COVID cases soared. American Medical Association website. April 7, 2021. https:// www.ama-assn.org/press-center/press-releases /most-physicians-had-little-relief-prior-author ization-covid-cases. Accessed November 29, 2021.
- Robeznieks A. 1 in 4 doctors say prior authorization has led to a serious adverse event. American Medical Association website. February 5, 2019. https://www .ama-assn.org/practice-management/sustainability /1-4-doctors-say-prior-authorization-has-led-serious -adverse. Accessed November 29, 2021.
- Physicians call on Congress to address prior authorization reform. American Medical Association website. May 14, 2021. https://www .ama-assn.org/press-center/press-releases /physicians-call-congress-address-prior-author ization-reform. Accessed November 29, 2021.
Physicians react: Should docs lose their licenses for spreading false COVID information?
Doctors providing “fraudulent” COVID-19 information became a hot-button issue for physicians responding to Medscape’s recent article, "Shouldn’t Doctors Who Spread False COVID-19 Information Lose Their Licenses?”
COVID-19 safety recommendations are set by mainstream medical organizations as new information becomes available, but some doctors consistently oppose advice from the Centers for Disease Control and Prevention and other medical authorities. These physicians often promote off-label, unapproved use of medications for COVID-19 and/or contradict mainstream safety guidelines such as vaccines, masks, and social distancing.
Some medical organizations are concerned that these doctors are hampering efforts to control the highly contagious coronavirus and are, at worst, placing lives in danger with their contrarian views that can spread like wildfire on social media sites. Their words are often used by those who refuse to be vaccinated or wear masks.
State licensing boards have mostly refused to discipline these doctors for making false and/or misleading claims, but as the virus spreads, there are calls to take action against them. However, others worry that such actions would violate free speech and critical thought.
Yes, those doctors are doing wrong
Several physicians took a strong stand against their fellow doctors who are spreading misinformation about COVID-19.
One doctor endorsed the idea of removing licenses for spreading misinformation and called for criminal prosecution: “It should certainly be grounds for cancellation of all licensing (after appropriate examination to rule out acute psychotic episodes, dementia, tumor, etc.) and very likely [include] a charge of manslaughter.”
Another health care provider said, “A person who does not accept science should not, of course, be allowed to practice medicine. One who argues publicly that vaccines and masks don’t work should be prosecuted for crimes ranging from reckless endangerment to attempted murder.”
One reader framed COVID-19 misinformers in stark terms: “These men and women are medical prostitutes. Their medical and surgical colleges [should] have a panel to track in-court testimony and the disinformation they spread ...”
“This is malpractice of the worst kind,” said a clinician. “Public health officials and science are quite clear on [the] best practices for safety during a pandemic, which is killing millions. This is a standard of care.”
“Medical Boards should suspend licenses and give the physician a chance to testify [about] the scientific basis for his comments,” added a health care provider. “Boards involve themselves in all kinds of perceived disciplinary infractions. We are in the midst of a lethal pandemic. I would think that would take precedence over many other issues?”
“I do believe that physicians have the responsibility to speak the truth and have scientifically displayed minds,” said a reader. “Not [to] promulgate misleading, false, and/or unverified information.”
“Any physician, who holds a license, should abide [by] government and state regulation,” asserted a doctor. “He should be disciplined by the board for spreading medical/public misinformation since he is creating potential harm to the population.”
One specialist insisted that “state boards do not do enough to restrict/limit the practice of physicians touting questionable therapies.”
“Any doctor who spreads false information about Covid is hurting our country, our individuals, and our economy and leading to needless deaths,” asserted a physician. “However, there are uncertainties, and where those exist, physicians [should] simply say ‘it is unknown.’”
No, those physicians have a right to speak their beliefs
However, many physicians worried that science and controversial thought were being muzzled.
“Absolutely no,” a doctor stated. “Who judges what is misinformation in this age where debate is canceled? Science advances with challenge, and it’s not about an authority dictating the allowable opinion.”
Another clinician claimed the “truth is very difficult to discern from less-than-truth in a country running on a profit-oriented economic ideology.”
One specialist warned that if disinformation doctors are held responsible, then “that means a lot of doctors” will be “gone” because “almost anything that is written or said about COVID can be contested.”
Another physician warned his colleagues about suppressing new ideas: “To condemn what we didn’t try, or purposefully ignore a different approach because [it] doesn’t agree with our opinion is suppression of information.”
Some doctors insisted the issue extended beyond medicine and into Constitutional freedoms. They also expressed their mistrust in the government to regulate physicians.
“There is a First Amendment in this country,” said one reader. “What you think is false may not be so. The people can listen to whoever they want to and make their own medical decisions. We do not need one iota more of politicizing medicine. Having an MD or DO does not mean you relinquish your First Amendment rights.”
“One of the fundamental problems with a system that allows government to ‘license’ physicians, or any other profession, is that politics inevitably turn to cronyism, and big businesses and wealthy people start controlling the government,” argued a doctor.
One clinician suggested enforcement against health food, drug company commercials, and talk shows: “What about all the [misinformation] at the health food stores and the like. Doctors of natural-whatever? Those info-commercials on tv. How many faxes do I get to ‘approve’ because ‘patients request’ braces and pain-treating expensive compounds advertised on TV? We tolerate those ... What about Dr. Oz and the docs on talk shows claiming BS?”
And the debate goes even further
Some physicians questioned the very notion of claiming “truth.”
“Nobody should be certain that they have the ‘absolute truth,’” said one reader. “In fact, the best clinical insights exceed so-called knowledge by at least one step.”
“Who can determine exactly what is truth?” asked another clinician. “For sure, the ‘Federal Government,’ who ‘is here to help you,’ is not qualified to make such determinations, and who are you to make such a suggestion as to remove someone’s license because they disagree with you? Give me a break!”
Another physician echoed that sentiment: “What’s true and false is often and certainly currently debatable. There are well-qualified physicians (with credentials such as the development of mRNA technology), virologists, and biostatisticians that have valid thoughts on this but do not necessarily agree with the drug company-sponsored journals and news channels (most of them). Their voices should be heard, and they should not lose their licenses. They are doing their work in good conscience.”
One reader commented that he wanted his “freedom of speech,” and offered this defiant advice: “You can take this license and shove it.”
Finally, a physician noted that the political climate has influenced medical directives: “If someone in a leadership role knowingly, and with intent, spread false information, that is wrong. However, during this global pandemic the active and the politics have combined. Red state no mandate, blue state mandate – what does that tell you about American leadership?”
A version of this article first appeared on Medscape.com.
Doctors providing “fraudulent” COVID-19 information became a hot-button issue for physicians responding to Medscape’s recent article, "Shouldn’t Doctors Who Spread False COVID-19 Information Lose Their Licenses?”
COVID-19 safety recommendations are set by mainstream medical organizations as new information becomes available, but some doctors consistently oppose advice from the Centers for Disease Control and Prevention and other medical authorities. These physicians often promote off-label, unapproved use of medications for COVID-19 and/or contradict mainstream safety guidelines such as vaccines, masks, and social distancing.
Some medical organizations are concerned that these doctors are hampering efforts to control the highly contagious coronavirus and are, at worst, placing lives in danger with their contrarian views that can spread like wildfire on social media sites. Their words are often used by those who refuse to be vaccinated or wear masks.
State licensing boards have mostly refused to discipline these doctors for making false and/or misleading claims, but as the virus spreads, there are calls to take action against them. However, others worry that such actions would violate free speech and critical thought.
Yes, those doctors are doing wrong
Several physicians took a strong stand against their fellow doctors who are spreading misinformation about COVID-19.
One doctor endorsed the idea of removing licenses for spreading misinformation and called for criminal prosecution: “It should certainly be grounds for cancellation of all licensing (after appropriate examination to rule out acute psychotic episodes, dementia, tumor, etc.) and very likely [include] a charge of manslaughter.”
Another health care provider said, “A person who does not accept science should not, of course, be allowed to practice medicine. One who argues publicly that vaccines and masks don’t work should be prosecuted for crimes ranging from reckless endangerment to attempted murder.”
One reader framed COVID-19 misinformers in stark terms: “These men and women are medical prostitutes. Their medical and surgical colleges [should] have a panel to track in-court testimony and the disinformation they spread ...”
“This is malpractice of the worst kind,” said a clinician. “Public health officials and science are quite clear on [the] best practices for safety during a pandemic, which is killing millions. This is a standard of care.”
“Medical Boards should suspend licenses and give the physician a chance to testify [about] the scientific basis for his comments,” added a health care provider. “Boards involve themselves in all kinds of perceived disciplinary infractions. We are in the midst of a lethal pandemic. I would think that would take precedence over many other issues?”
“I do believe that physicians have the responsibility to speak the truth and have scientifically displayed minds,” said a reader. “Not [to] promulgate misleading, false, and/or unverified information.”
“Any physician, who holds a license, should abide [by] government and state regulation,” asserted a doctor. “He should be disciplined by the board for spreading medical/public misinformation since he is creating potential harm to the population.”
One specialist insisted that “state boards do not do enough to restrict/limit the practice of physicians touting questionable therapies.”
“Any doctor who spreads false information about Covid is hurting our country, our individuals, and our economy and leading to needless deaths,” asserted a physician. “However, there are uncertainties, and where those exist, physicians [should] simply say ‘it is unknown.’”
No, those physicians have a right to speak their beliefs
However, many physicians worried that science and controversial thought were being muzzled.
“Absolutely no,” a doctor stated. “Who judges what is misinformation in this age where debate is canceled? Science advances with challenge, and it’s not about an authority dictating the allowable opinion.”
Another clinician claimed the “truth is very difficult to discern from less-than-truth in a country running on a profit-oriented economic ideology.”
One specialist warned that if disinformation doctors are held responsible, then “that means a lot of doctors” will be “gone” because “almost anything that is written or said about COVID can be contested.”
Another physician warned his colleagues about suppressing new ideas: “To condemn what we didn’t try, or purposefully ignore a different approach because [it] doesn’t agree with our opinion is suppression of information.”
Some doctors insisted the issue extended beyond medicine and into Constitutional freedoms. They also expressed their mistrust in the government to regulate physicians.
“There is a First Amendment in this country,” said one reader. “What you think is false may not be so. The people can listen to whoever they want to and make their own medical decisions. We do not need one iota more of politicizing medicine. Having an MD or DO does not mean you relinquish your First Amendment rights.”
“One of the fundamental problems with a system that allows government to ‘license’ physicians, or any other profession, is that politics inevitably turn to cronyism, and big businesses and wealthy people start controlling the government,” argued a doctor.
One clinician suggested enforcement against health food, drug company commercials, and talk shows: “What about all the [misinformation] at the health food stores and the like. Doctors of natural-whatever? Those info-commercials on tv. How many faxes do I get to ‘approve’ because ‘patients request’ braces and pain-treating expensive compounds advertised on TV? We tolerate those ... What about Dr. Oz and the docs on talk shows claiming BS?”
And the debate goes even further
Some physicians questioned the very notion of claiming “truth.”
“Nobody should be certain that they have the ‘absolute truth,’” said one reader. “In fact, the best clinical insights exceed so-called knowledge by at least one step.”
“Who can determine exactly what is truth?” asked another clinician. “For sure, the ‘Federal Government,’ who ‘is here to help you,’ is not qualified to make such determinations, and who are you to make such a suggestion as to remove someone’s license because they disagree with you? Give me a break!”
Another physician echoed that sentiment: “What’s true and false is often and certainly currently debatable. There are well-qualified physicians (with credentials such as the development of mRNA technology), virologists, and biostatisticians that have valid thoughts on this but do not necessarily agree with the drug company-sponsored journals and news channels (most of them). Their voices should be heard, and they should not lose their licenses. They are doing their work in good conscience.”
One reader commented that he wanted his “freedom of speech,” and offered this defiant advice: “You can take this license and shove it.”
Finally, a physician noted that the political climate has influenced medical directives: “If someone in a leadership role knowingly, and with intent, spread false information, that is wrong. However, during this global pandemic the active and the politics have combined. Red state no mandate, blue state mandate – what does that tell you about American leadership?”
A version of this article first appeared on Medscape.com.
Doctors providing “fraudulent” COVID-19 information became a hot-button issue for physicians responding to Medscape’s recent article, "Shouldn’t Doctors Who Spread False COVID-19 Information Lose Their Licenses?”
COVID-19 safety recommendations are set by mainstream medical organizations as new information becomes available, but some doctors consistently oppose advice from the Centers for Disease Control and Prevention and other medical authorities. These physicians often promote off-label, unapproved use of medications for COVID-19 and/or contradict mainstream safety guidelines such as vaccines, masks, and social distancing.
Some medical organizations are concerned that these doctors are hampering efforts to control the highly contagious coronavirus and are, at worst, placing lives in danger with their contrarian views that can spread like wildfire on social media sites. Their words are often used by those who refuse to be vaccinated or wear masks.
State licensing boards have mostly refused to discipline these doctors for making false and/or misleading claims, but as the virus spreads, there are calls to take action against them. However, others worry that such actions would violate free speech and critical thought.
Yes, those doctors are doing wrong
Several physicians took a strong stand against their fellow doctors who are spreading misinformation about COVID-19.
One doctor endorsed the idea of removing licenses for spreading misinformation and called for criminal prosecution: “It should certainly be grounds for cancellation of all licensing (after appropriate examination to rule out acute psychotic episodes, dementia, tumor, etc.) and very likely [include] a charge of manslaughter.”
Another health care provider said, “A person who does not accept science should not, of course, be allowed to practice medicine. One who argues publicly that vaccines and masks don’t work should be prosecuted for crimes ranging from reckless endangerment to attempted murder.”
One reader framed COVID-19 misinformers in stark terms: “These men and women are medical prostitutes. Their medical and surgical colleges [should] have a panel to track in-court testimony and the disinformation they spread ...”
“This is malpractice of the worst kind,” said a clinician. “Public health officials and science are quite clear on [the] best practices for safety during a pandemic, which is killing millions. This is a standard of care.”
“Medical Boards should suspend licenses and give the physician a chance to testify [about] the scientific basis for his comments,” added a health care provider. “Boards involve themselves in all kinds of perceived disciplinary infractions. We are in the midst of a lethal pandemic. I would think that would take precedence over many other issues?”
“I do believe that physicians have the responsibility to speak the truth and have scientifically displayed minds,” said a reader. “Not [to] promulgate misleading, false, and/or unverified information.”
“Any physician, who holds a license, should abide [by] government and state regulation,” asserted a doctor. “He should be disciplined by the board for spreading medical/public misinformation since he is creating potential harm to the population.”
One specialist insisted that “state boards do not do enough to restrict/limit the practice of physicians touting questionable therapies.”
“Any doctor who spreads false information about Covid is hurting our country, our individuals, and our economy and leading to needless deaths,” asserted a physician. “However, there are uncertainties, and where those exist, physicians [should] simply say ‘it is unknown.’”
No, those physicians have a right to speak their beliefs
However, many physicians worried that science and controversial thought were being muzzled.
“Absolutely no,” a doctor stated. “Who judges what is misinformation in this age where debate is canceled? Science advances with challenge, and it’s not about an authority dictating the allowable opinion.”
Another clinician claimed the “truth is very difficult to discern from less-than-truth in a country running on a profit-oriented economic ideology.”
One specialist warned that if disinformation doctors are held responsible, then “that means a lot of doctors” will be “gone” because “almost anything that is written or said about COVID can be contested.”
Another physician warned his colleagues about suppressing new ideas: “To condemn what we didn’t try, or purposefully ignore a different approach because [it] doesn’t agree with our opinion is suppression of information.”
Some doctors insisted the issue extended beyond medicine and into Constitutional freedoms. They also expressed their mistrust in the government to regulate physicians.
“There is a First Amendment in this country,” said one reader. “What you think is false may not be so. The people can listen to whoever they want to and make their own medical decisions. We do not need one iota more of politicizing medicine. Having an MD or DO does not mean you relinquish your First Amendment rights.”
“One of the fundamental problems with a system that allows government to ‘license’ physicians, or any other profession, is that politics inevitably turn to cronyism, and big businesses and wealthy people start controlling the government,” argued a doctor.
One clinician suggested enforcement against health food, drug company commercials, and talk shows: “What about all the [misinformation] at the health food stores and the like. Doctors of natural-whatever? Those info-commercials on tv. How many faxes do I get to ‘approve’ because ‘patients request’ braces and pain-treating expensive compounds advertised on TV? We tolerate those ... What about Dr. Oz and the docs on talk shows claiming BS?”
And the debate goes even further
Some physicians questioned the very notion of claiming “truth.”
“Nobody should be certain that they have the ‘absolute truth,’” said one reader. “In fact, the best clinical insights exceed so-called knowledge by at least one step.”
“Who can determine exactly what is truth?” asked another clinician. “For sure, the ‘Federal Government,’ who ‘is here to help you,’ is not qualified to make such determinations, and who are you to make such a suggestion as to remove someone’s license because they disagree with you? Give me a break!”
Another physician echoed that sentiment: “What’s true and false is often and certainly currently debatable. There are well-qualified physicians (with credentials such as the development of mRNA technology), virologists, and biostatisticians that have valid thoughts on this but do not necessarily agree with the drug company-sponsored journals and news channels (most of them). Their voices should be heard, and they should not lose their licenses. They are doing their work in good conscience.”
One reader commented that he wanted his “freedom of speech,” and offered this defiant advice: “You can take this license and shove it.”
Finally, a physician noted that the political climate has influenced medical directives: “If someone in a leadership role knowingly, and with intent, spread false information, that is wrong. However, during this global pandemic the active and the politics have combined. Red state no mandate, blue state mandate – what does that tell you about American leadership?”
A version of this article first appeared on Medscape.com.
A COVID-19 Clinical Management Committee to Standardize Care in a 2-Hospital System
From the Department of Medicine (Drs. Meisenberg, Muganlinskaya, Sharma, Amjadi, Arnold, Barnes, Clance, Khalil, Miller, Mooradian, O’Connell, Patel, Press, Samaras, Shanmugam, Tavadze, and Thompson), Department of Pharmacy (Drs. Jiang, Jarawan, Sheth, and Trinh), Department of Nursing (Dr. Ohnmacht), and Department of Women and Children’s Services (Dr. Raji), Luminis Health, Annapolis, MD, and Lanham, MD.
Objective: The COVID-19 pandemic has been a challenge for hospital medical staffs worldwide due to high volumes of patients acutely ill with novel syndromes and prevailing uncertainty regarding optimum supportive and therapeutic interventions. Additionally, the response to this crisis was driven by a plethora of nontraditional information sources, such as email chains, websites, non–peer-reviewed preprints, and press releases. Care patterns became idiosyncratic and often incorporated unproven interventions driven by these nontraditional information sources. This report evaluates the efforts of a health system to create and empower a multidisciplinary committee to develop, implement, and monitor evidence-based, standardized protocols for patients with COVID-19.
Methods: This report describes the composition of the committee, its scope, and its important interactions with the health system pharmacy and therapeutics committee, research teams, and other work groups planning other aspects of COVID-19 management. It illustrates how the committee was used to demonstrate for trainees the process and value of critically examining evidence, even in a chaotic environment.
Results: Data show successful interventions in reducing excessive ordering of certain laboratory tests, reduction of nonrecommended therapies, and rapid uptake of evidence-based or guidelines-supported interventions.
Conclusions: A multidisciplinary committee dedicated solely to planning, implementing, and monitoring standard approaches that eventually became evidence-based decision-making led to an improved focus on treatment options and outcomes for COVID-19 patients. Data presented illustrate the attainable success that is both adaptable and suitable for similar emergencies in the future.
Keywords: COVID-19; clinical management; pharmacy and therapeutics; treatment; therapy.
The COVID-19 pandemic has spread to nearly all countries, carrying with it high morbidity, mortality, and severe impacts on both well-developed and less-well-developed health systems. Media reports of chaos within overwhelmed hospitals have been prominent.1,2 As of January 5, 2022, SARS-CoV-2 has infected more than 295 million people globally and directly caused the death of more than 5.4 million,3 though this number is likely an undercount even in countries with well-developed mortality tracking.4
Throughout the COVID-19 pandemic, hospital-based medical teams have been confronted with a flood of severely ill patients with novel syndromes. Initially, there were no standards for therapy or supportive care except for treatments borrowed from similar syndromes. In the setting of high volumes, high acuity, and public dismay, it is unsurprising that the usual deliberative methods for weighing evidence and initiating interventions were often pushed aside in favor of the solace of active intervention.5 In this milieu of limited evidence, there was a lamentable, if understandable, tendency to seek guidance from “nontraditional” sources,6 including email chains from colleagues, hospital websites, non–peer-reviewed manuscripts, advanced publication by medical journals,7 and nonscientific media presentations. In many localities, practitioners responded in idiosyncratic ways. For example, findings of high cytokine levels in COVID-19,8 along with reports of in-vitro antiviral activity with drugs like hydroxychloroquine against both SARS9 and SARS-CoV-2,10 drove laboratory test ordering and therapeutic interventions, respectively, carving shortcuts into the traditional clinical trial–dependent standards. Clinical trial results eventually emerged.11COVID-19 created a clinical dilemma for hospital medical staffs in terms of how to organize, standardize, and rapidly adapt to a flood of new information. In this report, we describe how 1 health system responded to these challenges by forming a COVID-19 Clinical Management Committee (CCMC) and empowering this interdisciplinary team to review evidence, create and adjust order sets, educate practitioners, oversee care, and collaborate across teams addressing other aspects of the COVID-19 response.
Program Overview
Health System Description
Luminis Health is a health system with 2 acute care hospitals that was formed in 2019 just before the start of the pandemic. Anne Arundel Medical Center (hospital A) is a 385-bed teaching hospital in Annapolis, MD. It has more than 23 000 discharges annually. Patients with COVID-19 were cared for by either an internal medicine teaching service or nonteaching hospitalist services on cohorted nursing units. Doctor’s Community Medical Center, in Lanham, MD (hospital B), is a 206-bed acute care hospital with more than 10 350 annual discharges. COVID-19 patients were cared for by hospitalist groups, initially in noncohorted units with transition to cohorted nursing units after a few months. The medical staffs are generally distinct, with different leadership structures, though the Luminis Health Department of Medicine has oversight responsibilities at both hospitals. More than 47 physicians attended COVID-19 patients at hospital A (with medical residents) and 30 individual physicians at hospital B, respectively, including intensivists. The nursing and pharmacy staffs are distinct, but there is a shared oversight Pharmacy and Therapeutics (P&T) Committee.
The 2 hospitals had distinct electronic medical records (EMR) until January 2021, when hospital B adopted the same EMR as hospital A (Epic).
Mission and Formation of CCMC
In order to coordinate the therapeutic approach across the health system, it was important for the CCMC to be designated by the health system P&T committee as an official subcommittee so that decisions on restrictions of medications and/or new or revised order sets could be rapidly initiated across the system without waiting for the subsequent P&T meetings. The full committee retained oversight of the CCMC. Some P&T members were also on the CCMC.
The committee reviewed new reports in medical journals and prepublication servers and consulted recommendations of professional societies, such as the National Institutes of Health (NIH) COVID-19 guidelines, Infectious Diseases Society of America, Society of Critical Care Medicine, and US Food and Drug Administration (FDA) Emergency Use Authorizations (EUA), among other sources.
Composition of the CCMC
Physician leaders from both hospitals in the following specialties were solicited for participation: critical care, epidemiology, hospital medicine (internal medicine), emergency medicine, infectious diseases, nephrology, women and children’s services, and medical informatics. Specialists in other areas, such as hematology, were invited for topic-specific discussions. Hospital pharmacists with different specialties and nursing leadership were essential contributors. The committee members were expected to use various communication channels to inform frontline clinicians of new care standards and the existence of new order sets, which were embedded in the EMR.
Clinical Research
An important connection for the CCMC was with theCOVID-19 clinical research team. Three members of the research team were also members of the CCMC. All new study proposals for therapeutics were discussed with the CCMC as they were being considered by the research team. In this way, feedback on the feasibility and acceptance of new study opportunities could be discussed with the CCMC. Occasionally, CCMC decisions impacted clinical research accrual strategies. For example, new data from randomized trials about tocilizumab1,2 demonstrated benefits in some subsets of patients and resulted in a recommendation for use by the NIH guideline committee in these populations.1 The CCMC quickly adopted this recommendation, which required a reprioritization of clinical research enrollment for studies testing other immune-modulating agents. This important dialogue was mediated within the CCMC.
Guideline Distribution, Reinforcement, and Platform for Feedback
New guidelines were disseminated to clinicians via daily brief patient huddles held on COVID units, with participation by nursing and pharmacy, and by weekly meetings with hospitalist leaders and frontline hospital physicians. Order sets and guidelines were maintained on the intranet. Adherence was reinforced by unit-based and central pharmacists. Order sets, including admission order sets, could be created only by designated informatics personnel, thus enforcing standardization. Feedback on the utility of the order sets was obtained during the weekly meetings or huddles, as described above. To ensure a sense of transparency, physicians who had interest in commenting on a particular therapy, or who wished to discuss a particular manuscript, news article, or website, were invited to attend CCMC meetings.
Scope of CCMC
In order to be effective and timely, we limited the scope of our work to the report to the inpatient therapeutic environment, allowing other committees to work on other aspects of the pandemic response. In addition to issuing guidance and creating order sets to direct clinical practice, the CCMC also monitored COVID-19 therapeutic shortages15,16 and advised on prioritization of such treatments as convalescent plasma, remdesivir (prioritization and duration of therapy, 5 vs 10 days), baricitinib, and tocilizumab, depending upon the location of the patient (critical care or not). The CCMC was not involved in the management of non–COVID-19 shortages brought about by supply chain deficiencies.
Table 1 shows some aspects of the health system pandemic-response planning and the committee workforce that undertook that work. Though many items were out of scope for the CCMC, members of the CCMC did participate in the planning work of these other committees and therefore stayed connected to this complementary work.
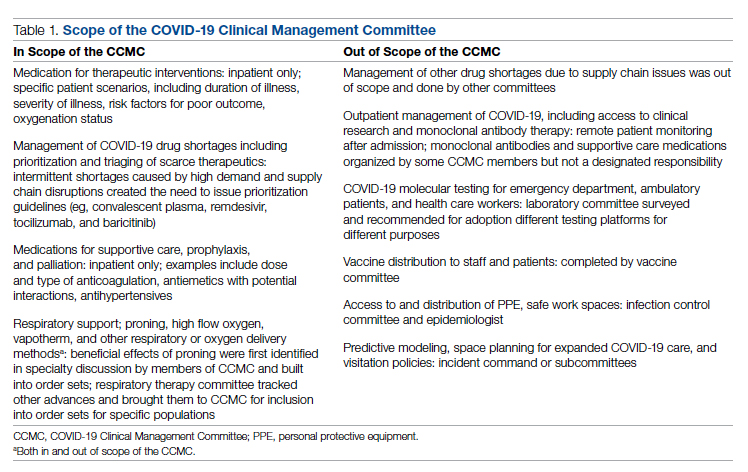
A Teaching Opportunity About Making Thoughtful Choices
Another important feature of the CCMC was the contributions of residents from both pharmacy and internal medicine. The purpose and operations of the committee were recognized as an opportunity to involve learners in a curriculum based on Kern’s 6-step approach.17 Though the problem identification and general needs assessment were easily defined, the targeted needs assessment, extracted from individual and group interviews with learners and the committee members, pointed at the need to learn how to assess and analyze a rapidly growing body of literature on several relevant clinical aspects of SARS-CoV-2 and COVID-19. To achieve goals and objectives, residents were assigned to present current literature on a particular intervention during a committee meeting, specifically commenting on the merit or deficiencies of the study design, the strength of the data, and applicability to the local context with a recommendation. Prior to the presentations, the residents worked with faculty to identify the best studies or systematic analyses with potential to alter current practices. We thus used the CCMC process as a teaching tool about evidence-based medicine and the dilemma of clinical equipoise. This was imperative, since trainees thrust into the COVID-19 response have often keenly observed a movement away from deliberative decision-making.18 Indeed, including residents in the process of deliberative responses to COVID-19 addresses a recent call to adjust medical education during COVID-19 to “adapt curriculum to current issues in real time.”19
Interventions and Therapies Considered
Table 2 shows the topics reviewed by the CCMC. By the time of the first meeting, nonstandardization of care was already a source of concern for clinicians. Dialogue often continued outside of the formal meetings. Many topics were considered more than once as new guidance developed, changes to EUAs occurred, and new data or new publicity arose.
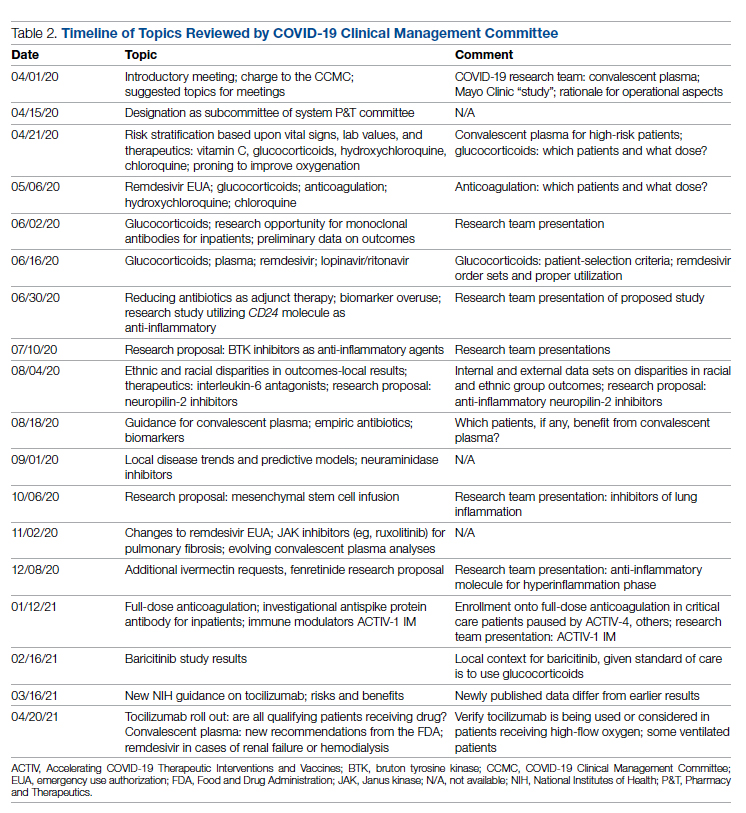
Methods
The Human Protections Administrator determined that this work constituted “quality improvement, and not research” and was therefore exempt from institutional review board review.
Quantitative Analysis
All admitted patients from March 10, 2020, through April 20, 2021, were considered in the quantitative aspects of this report except as noted. Patients diagnosed with COVID-19 were identified by searching our internal data base using diagnostic codes. Patient admissions with the following diagnostic codes were included (prior to April 1, 2020): J12.89, J20.8, J40, J22, J98.8, J80, each with the additional code of B97.29. After April 1, 2020, the guideline for coding COVID-19 was U07.1.
Descriptive statistics were used to measure utilization rates of certain medications and laboratory tests of interest over time. These data were adjusted for number of unique admissions. In a few cases, not all data elements were available from both hospitals due to differences in the EMR.
Case fatality rate was calculated based upon whether the patient died or was admitted to inpatient hospice as a result of COVID-19. Four patients transferred out of hospital A and 18 transferred out of hospital B were censored from case-fatality-rate determination.
Figure 1 shows the number of admissions for each acute care hospital in the health system and the combined COVID-19 case-fatality rate over time.
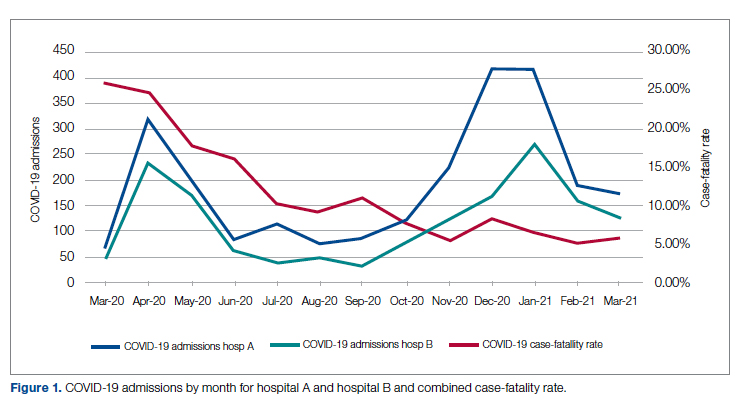
Results
A total of 5955 consecutive COVID-19 patients admitted from March 10, 2020, through April 30, 2021, were analyzed. Patients with International Statistical Classification of Diseases, Tenth Revision codes J12.89. J20.8, J40, J22, J98.8, J80, each with the additional code of B97.29 (or the code UO7.1 after April 1, 2020), were included in the analysis. The median age of admitted patients was 65 years (range 19-91 years). Using the NIH classification system for severity,20 the distribution of severity during the first 24 hours after the time of hospital admission was as follows: asymptomatic/presymptomatic, 0.5%; mild illness, 5.3%; moderate illness, 37.1%; severe illness, 55.5%; and critical illness, 1.1%.
The impact of the CCMC can be estimated by looking at care patterns over time. Since the work of the CCMC was aimed at influencing and standardizing physician ordering and therapy choices through order set creation and other forms of oversight, we measured the use of the CCMC-approved order sets at both hospitals and the use of certain laboratory tests and therapies that the CCMC sought either to limit or increase. These counts were adjusted for number of unique COVID-19 admissions. But the limits of the case collection tool meant it also collected cases that were not eligible for some of the interventions. For example, COVID-19 admissions without hypoxemia would not have been eligible for remdesivir or glucocorticoids. When admitted, some patients were already on steroids for other medical indications and did not receive the prescribed dexamethasone dose that we measured in pharmacy databases. Similarly, a few patients were hospitalized for indications unrelated to COVID-19, such as surgery or childbirth, and were found to be SARS-CoV-2-positive on routine screening.
Figure 2 shows the utilization of CCMC-approved standard COVID-19 admission order sets as a proportion of all COVID-19 admissions over time. The trend reveals a modest increase in usage (R2 = 0.34), but these data do not reflect the progressive build of content into order sets over time. One of the goals of the order sets was to standardize and reduce the ordering of certain biomarkers: C-reactive protein, serum ferritin, and D-dimer, which were ordered frequently in many early patients. Orders for these 3 laboratory tests are combined and expressed as an average number of labs per COVID-19 admission in Figure 2. A downward trend, with an R2 value of 0.65, is suggestive of impact from the order sets, though other explanations are possible.
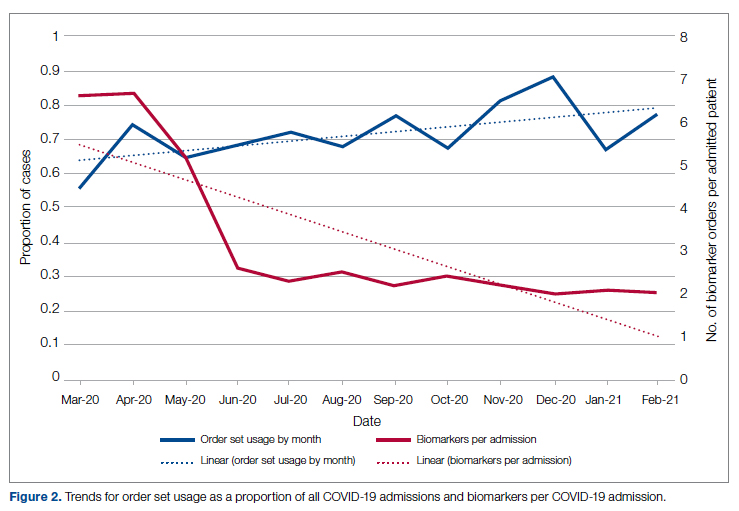
Medication guidance was also a goal of the CCMC, simultaneously discouraging poorly supported interventions and driving uptake of the recommended evidence-based interventions in appropriate patients. Figure 3 shows the utilization pattern for some drugs of interest over the course of the pandemic, specifically the proportion of patients receiving at least 1 dose of medication among all COVID-19 admissions by month. (Data for hospital B was excluded from this analysis because it did not include all admitted patients.)
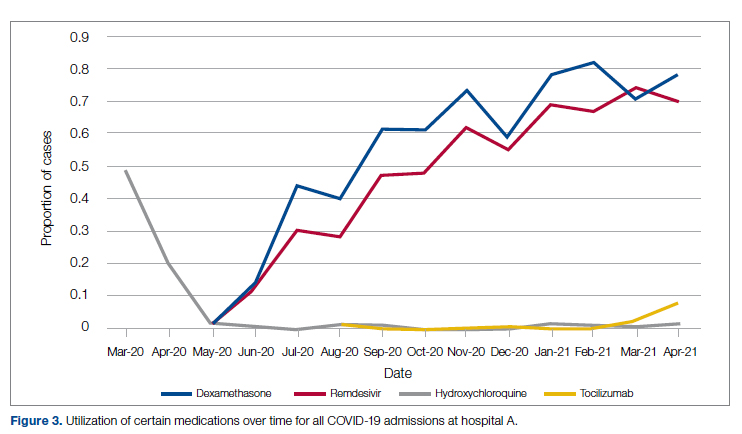
Hydroxychloroquine, which enjoyed a wave of popularity early on during the pandemic, was a target of successful order stewardship through the CCMC. Use of hydroxychloroquine as a COVID-19 therapeutic option after the first 2 months of the pandemic stopped, and subsequent use at low levels likely represented continuation therapy for outpatients who took hydroxychloroquine for rheumatologic indications.
Dexamethasone, as used in the RECOVERY trial,21 had a swift uptake among physicians after it was incorporated into order sets and its use encouraged. Similarly, uptake was immediate for remdesivir when, in May 2020, preliminary reports showed at least some benefits, confirmed by later analysis,22 and it received an FDA EUA.
Our data also show successful stewardship of the interleukin-6 antagonist toclilizumab, which was discouraged early on by the CCMC due to lack of data or negative results. But in March 2021, with new studies releasing data12,13 and new recommendations14 for its use in some subsets of patients with COVID-19, this drug was encouraged in appropriate subsets. A new order set with qualifying indications was prepared by the CCMC and new educational efforts made to encourage its use in appropriate patients.
Ivermectin was nonformulary at the start of the pandemic. This drug enjoyed much publicity from media sources and was promoted by certain physicians and on websites,23 based on in-vitro activity against coronaviruses. Eventually, the World Health Organization24 and the FDA25 found it necessary to issue advisory statements to the public against its use outside of clinical trials. The CCMC had requests from physicians to incorporate ivermectin but declined to add it to the formulary and recommended not approving nonformulary requests due to lack of data. As a result, ivermectin was not used at either hospital.
Discussion
COVID-19 represents many challenges to health systems all over the world. For Luminis Health, the high volume of acutely ill patients with novel syndromes was a particular challenge for the hospital-based care teams. A flood of information from preprints, press releases, preliminary reports, and many other nontraditional sources made deliberative management decisions difficult for individual physicians. Much commentary has appeared around the phenomenon but with less practical advice about how to make day-to-day care decisions at a time of scientific uncertainty and intense pressure to intervene.26,27 The CCMC was designed to overcome the information management dilemma. The need to coordinate, standardize, and oversee care was necessary given the large number of physicians who cared for COVID-19 patients on inpatient services.
It should be noted that creating order sets and issuing guidance is necessary, but not sufficient, to achieve our goals of being updated and consistent. This is especially true with large cadres of health care workers attending COVID-19 patients. Guidelines and recommendations were reinforced by unit-based oversight and stewardship from pharmacy and other leaders who constituted the CCMC.
The reduction in COVID-19 mortality over time experienced in this health care system was not unique and cannot necessarily be attributed to standardization of care. Similar improvements in mortality have been reported at many US hospitals in aggregate.28 Many other factors, including changes in patient characteristics, may be responsible for reduction in mortality over time.
Throughout this report we have relied upon an implicit assumption that standardization of medical therapeutics is desirable and leads to better outcomes as compared with allowing unlimited empiricism by individual physicians, either consultants or hospitalists. Our program represents a single health system with 2 acute care hospitals located 25 miles apart and which thus were similarly impacted by the different phases of the pandemic. Generalizability to health systems either smaller or larger, or in different geographical areas, has not been established. Data limitations have already been discussed.
We did not measure user satisfaction with the program either from physicians or nurses. However, the high rate of compliance suggests general agreement with the content and process.
We cannot definitively ascribe reduction in utilization of some nonrecommended treatments and increased utilization of the recommended therapies to the work of the CCMC. Individual physicians may have made these adjustments on their own or under the influence of other sources.
Finally, it should be noted that the mission to rapidly respond to data from well-conducted trials might be thwarted by too rigid a process or a committee’s lack of a sense of urgency. Organizing a committee and then empowering it to act is no guarantee of success; commitment to the mission is.
Conclusion
COVID-19 represented a challenge to medical staffs everywhere, inundating them with high volumes of acutely ill patients presenting with unfamiliar syndromes. Initial responses were characterized by idiosyncratic management approaches based on nontraditional sources of opinion and influences.
This report describes how a complex medical system brought order and standardization through a deliberative, but urgent, multidisciplinary committee with responsibility for planning, implementing, and monitoring standard approaches that eventually became evidence based. The composition of the committee and its scope of influence, limited to inpatient management, were important elements of success, allowing for better focus on the many treatment decisions. The important connection between the management committee and the system P&T committee, the clinical research effort, and teaching programs in both medicine and pharmacy are offered as exemplars of coordination. The data presented show success in achieving standardized, guideline-directed care. The approach is adoptable and suitable for similar emergencies in the future.
Acknowledgments: The authors thank Gary Scabis, Kip Waite, John Moxley, Angela Clubb, and David Woodley for their assistance in gathering data. We express appreciation and admiration for all our colleagues at the bedside.
Corresponding author: Barry R. Meisenberg, MD, Department of Medicine, Luminis Health, 2001 Medical Pkwy, Annapolis, MD 21401; [email protected].
Financial disclosures: None.
1. Gettleman J, Raj S, Kumar H. India’s health system cracks under the strain as coronavirus cases surge. The New York Times. April 22, 2021. https://www.nytimes.com/2021/04/21/world/asia/india-coronavirus-oxygen.html
2. Rappleye H, Lehren AW, Strickler L, Fitzpatrick S. ‘This system is doomed’: doctors, nurses sound off in NBC News coronavirus survey. NBC News. March 20, 2020. https://www.nbcnews.com/news/us-news/system-doomed-doctors-nurses-sound-nbc-news-coronavirus-survey-n1164841
3. Johns Hopkins Coronavirus Resource Center. Accessed January 5, 2022. https://coronavirus.jhu.edu/map.html
4. Fineberg HV. The toll of COVID-19. JAMA. 2020;324(15):1502-1503. doi:10.1001/jama.2020.20019
5. Meisenberg BR. Medical staffs response to COVID-19 ‘data’: have we misplaced our skeptic’s eye? Am J Med. 2021;134(2):151-152. doi:10.1016/j.amjmed.2020.09.013
6. McMahon JH, Lydeamore MH, Stewardson AJ. Bringing evidence from press release to the clinic in the era of COVID-19. J Antimicrob Chemother. 2021;76(3):547-549. doi:10.1093/jac/dkaa506
7. Rubin EJ, Baden LR, Morrissey S, Campion EW. Medical journals and the 2019-nCoV outbreak. N Engl J Med. 2020;382(9):866. doi:10.1056/NEJMe2001329
8. Liu F, Li L, Xu M, et al. Prognostic value of interleukin-6, C-reactive protein, and procalcitonin in patients with COVID-19. J Clin Virol. 2020;127:104370. doi:10.1016/j.jcv.2020.104370
9. Vincent MJ, Bergeron E, Benjannet S, et al. Chloroquine is a potent inhibitor of SARS coronavirus infection and spread. Virol J. 2005;2:69. doi:10.1186/1743-422X-2-69
10. Wang M, Cao R, Zhang L, et al. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res. 2020;30:269-271. doi:10.1038/s41422-020-0282-0
11. RECOVERY Collaborative Group. Effect of hydroxychloroquine in hospitalized patients with Covid-19. N Engl J Med. 2020;383:2030-2040. doi:10.1056/NEJMoa2022926
12. RECOVERY Collaborative Group. Tocilizumab in patients admitted to hospital with COVID-19 (RECOVERY): preliminary results of a randomised, controlled, open-label, platform trial [preprint]. February 11, 2021. doi:10.1101/2021.02.11.21249258 https://www.medrxiv.org/content/10.1101/2021.02.11.21249258v1
13. REMAP-CAP Investigators. Interleukin-6 receptor antagonists in critically ill patients with COVID-19. N Engl J Med. 2021;384(16):1491-1502. doi:10.1056/NEJMoa2100433
14. National Institutes of Health. COVID-19 treatment guidelines: interleukin-6 inhibitors. https://www.covid19treatmentguidelines.nih.gov/immunomodulators/interleukin-6-inhibitors/
15. Deana C, Vetrugno L, Tonizzo A, et al. Drug supply during COVID-19 pandemic: remember not to run with your tank empty. Hosp Pharm. 2021;56(5):405-407. doi:10.1177/0018578720931749
16. Choe J, Crane M, Greene J, et al. The Pandemic and the Supply Chain: Addressing Gaps in Pharmaceutical Production and Distribution. Johns Hopkins University, November 2020. https://www.jhsph.edu/research/affiliated-programs/johns-hopkins-drug-access-and-affordability-initiative/publications/Pandemic_Supply_Chain.pdf
17. Kern DE. Overview: a six-step approach to curriculum development. In: Kern DE, Thornton PA, Hughes MT, eds. Curriculum Development for Medical Education: A Six-Step Approach. 3rd ed. Johns Hopkins University Press; 2016.
18. Rice TW, Janz DR. In defense of evidence-based medicine for the treatment of COVID-19 acute respiratory distress syndrome. Ann Am Thorac Soc. 2020;17(7):787-789. doi:10.1513/AnnalsATS.202004-325IP
19. Lucey CR, Johnston SC. The transformational effects of COVID-19 on medical education. JAMA. 2020;324(11):1033-1034. doi:10.1001/jama.2020.14136
20. National Institutes of Health. COVID-19 treatment guidelines: clinical spectrum of SARS-CoV-2 infection. https://www.covid19treatmentguidelines.nih.gov/overview/clinical-spectrum/
21. RECOVERY Collaborative Group. Dexamethasone in hospitalized patients with Covid-19. N Engl J Med. 2021;384:693-704. doi:10.1056/NEJMoa2021436
22. Beigel JH, Tomashek KM, Dodd LE, et al. Remdesivir for the treatment of Covid-19—final report. N Engl J Med. 2020;383:1813-1826. doi:10.1056/NEJMoa2007764
23. Jiminez D. Ivermectin and Covid-19: how a cheap antiparasitic became political. April 19, 2021. https://www.pharmaceutical-technology.com/features/ivermectin-covid-19-antiparasitic-political/
24. World Health Organization. WHO advises that ivermectin only be used to treat COVID-19 within clinical trials. March 31, 2021. https://www.who.int/news-room/feature-stories/detail/who-advises-that-ivermectin-only-be-used-to-treat-covid-19-within-clinical-trials
25. U.S. Food and Drug Administration. Why you should not use ivermectin to treat or prevent COVID-19. March 5, 2021. https://www.fda.gov/consumers/consumer-updates/why-you-should-not-use-ivermectin-treat-or-prevent-covid-19
26. Seymour CW, McCreary EK, Stegenga J. Sensible medicine-balancing intervention and inaction during the COVID-19 pandemic. JAMA. 2020;324(18):1827-1828. doi:10.1001/jama.2020.20271
27. Flanagin A, Fontanarosa PB, Bauchner H. Preprints involving medical research—do the benefits outweigh the challenges? JAMA. 2020;324(18):1840-1843. doi:10.1001/jama.2020.20674
28. Asch DA, Shells NE, Islam N, et al. Variation in US hospital mortality rates for patients admitted with COVID-19 during the first 6 months of the pandemic. JAMA Intern Med. 2021;181(4):471-478. doi:10.1001/jamainternmed.2020.8193
From the Department of Medicine (Drs. Meisenberg, Muganlinskaya, Sharma, Amjadi, Arnold, Barnes, Clance, Khalil, Miller, Mooradian, O’Connell, Patel, Press, Samaras, Shanmugam, Tavadze, and Thompson), Department of Pharmacy (Drs. Jiang, Jarawan, Sheth, and Trinh), Department of Nursing (Dr. Ohnmacht), and Department of Women and Children’s Services (Dr. Raji), Luminis Health, Annapolis, MD, and Lanham, MD.
Objective: The COVID-19 pandemic has been a challenge for hospital medical staffs worldwide due to high volumes of patients acutely ill with novel syndromes and prevailing uncertainty regarding optimum supportive and therapeutic interventions. Additionally, the response to this crisis was driven by a plethora of nontraditional information sources, such as email chains, websites, non–peer-reviewed preprints, and press releases. Care patterns became idiosyncratic and often incorporated unproven interventions driven by these nontraditional information sources. This report evaluates the efforts of a health system to create and empower a multidisciplinary committee to develop, implement, and monitor evidence-based, standardized protocols for patients with COVID-19.
Methods: This report describes the composition of the committee, its scope, and its important interactions with the health system pharmacy and therapeutics committee, research teams, and other work groups planning other aspects of COVID-19 management. It illustrates how the committee was used to demonstrate for trainees the process and value of critically examining evidence, even in a chaotic environment.
Results: Data show successful interventions in reducing excessive ordering of certain laboratory tests, reduction of nonrecommended therapies, and rapid uptake of evidence-based or guidelines-supported interventions.
Conclusions: A multidisciplinary committee dedicated solely to planning, implementing, and monitoring standard approaches that eventually became evidence-based decision-making led to an improved focus on treatment options and outcomes for COVID-19 patients. Data presented illustrate the attainable success that is both adaptable and suitable for similar emergencies in the future.
Keywords: COVID-19; clinical management; pharmacy and therapeutics; treatment; therapy.
The COVID-19 pandemic has spread to nearly all countries, carrying with it high morbidity, mortality, and severe impacts on both well-developed and less-well-developed health systems. Media reports of chaos within overwhelmed hospitals have been prominent.1,2 As of January 5, 2022, SARS-CoV-2 has infected more than 295 million people globally and directly caused the death of more than 5.4 million,3 though this number is likely an undercount even in countries with well-developed mortality tracking.4
Throughout the COVID-19 pandemic, hospital-based medical teams have been confronted with a flood of severely ill patients with novel syndromes. Initially, there were no standards for therapy or supportive care except for treatments borrowed from similar syndromes. In the setting of high volumes, high acuity, and public dismay, it is unsurprising that the usual deliberative methods for weighing evidence and initiating interventions were often pushed aside in favor of the solace of active intervention.5 In this milieu of limited evidence, there was a lamentable, if understandable, tendency to seek guidance from “nontraditional” sources,6 including email chains from colleagues, hospital websites, non–peer-reviewed manuscripts, advanced publication by medical journals,7 and nonscientific media presentations. In many localities, practitioners responded in idiosyncratic ways. For example, findings of high cytokine levels in COVID-19,8 along with reports of in-vitro antiviral activity with drugs like hydroxychloroquine against both SARS9 and SARS-CoV-2,10 drove laboratory test ordering and therapeutic interventions, respectively, carving shortcuts into the traditional clinical trial–dependent standards. Clinical trial results eventually emerged.11COVID-19 created a clinical dilemma for hospital medical staffs in terms of how to organize, standardize, and rapidly adapt to a flood of new information. In this report, we describe how 1 health system responded to these challenges by forming a COVID-19 Clinical Management Committee (CCMC) and empowering this interdisciplinary team to review evidence, create and adjust order sets, educate practitioners, oversee care, and collaborate across teams addressing other aspects of the COVID-19 response.
Program Overview
Health System Description
Luminis Health is a health system with 2 acute care hospitals that was formed in 2019 just before the start of the pandemic. Anne Arundel Medical Center (hospital A) is a 385-bed teaching hospital in Annapolis, MD. It has more than 23 000 discharges annually. Patients with COVID-19 were cared for by either an internal medicine teaching service or nonteaching hospitalist services on cohorted nursing units. Doctor’s Community Medical Center, in Lanham, MD (hospital B), is a 206-bed acute care hospital with more than 10 350 annual discharges. COVID-19 patients were cared for by hospitalist groups, initially in noncohorted units with transition to cohorted nursing units after a few months. The medical staffs are generally distinct, with different leadership structures, though the Luminis Health Department of Medicine has oversight responsibilities at both hospitals. More than 47 physicians attended COVID-19 patients at hospital A (with medical residents) and 30 individual physicians at hospital B, respectively, including intensivists. The nursing and pharmacy staffs are distinct, but there is a shared oversight Pharmacy and Therapeutics (P&T) Committee.
The 2 hospitals had distinct electronic medical records (EMR) until January 2021, when hospital B adopted the same EMR as hospital A (Epic).
Mission and Formation of CCMC
In order to coordinate the therapeutic approach across the health system, it was important for the CCMC to be designated by the health system P&T committee as an official subcommittee so that decisions on restrictions of medications and/or new or revised order sets could be rapidly initiated across the system without waiting for the subsequent P&T meetings. The full committee retained oversight of the CCMC. Some P&T members were also on the CCMC.
The committee reviewed new reports in medical journals and prepublication servers and consulted recommendations of professional societies, such as the National Institutes of Health (NIH) COVID-19 guidelines, Infectious Diseases Society of America, Society of Critical Care Medicine, and US Food and Drug Administration (FDA) Emergency Use Authorizations (EUA), among other sources.
Composition of the CCMC
Physician leaders from both hospitals in the following specialties were solicited for participation: critical care, epidemiology, hospital medicine (internal medicine), emergency medicine, infectious diseases, nephrology, women and children’s services, and medical informatics. Specialists in other areas, such as hematology, were invited for topic-specific discussions. Hospital pharmacists with different specialties and nursing leadership were essential contributors. The committee members were expected to use various communication channels to inform frontline clinicians of new care standards and the existence of new order sets, which were embedded in the EMR.
Clinical Research
An important connection for the CCMC was with theCOVID-19 clinical research team. Three members of the research team were also members of the CCMC. All new study proposals for therapeutics were discussed with the CCMC as they were being considered by the research team. In this way, feedback on the feasibility and acceptance of new study opportunities could be discussed with the CCMC. Occasionally, CCMC decisions impacted clinical research accrual strategies. For example, new data from randomized trials about tocilizumab1,2 demonstrated benefits in some subsets of patients and resulted in a recommendation for use by the NIH guideline committee in these populations.1 The CCMC quickly adopted this recommendation, which required a reprioritization of clinical research enrollment for studies testing other immune-modulating agents. This important dialogue was mediated within the CCMC.
Guideline Distribution, Reinforcement, and Platform for Feedback
New guidelines were disseminated to clinicians via daily brief patient huddles held on COVID units, with participation by nursing and pharmacy, and by weekly meetings with hospitalist leaders and frontline hospital physicians. Order sets and guidelines were maintained on the intranet. Adherence was reinforced by unit-based and central pharmacists. Order sets, including admission order sets, could be created only by designated informatics personnel, thus enforcing standardization. Feedback on the utility of the order sets was obtained during the weekly meetings or huddles, as described above. To ensure a sense of transparency, physicians who had interest in commenting on a particular therapy, or who wished to discuss a particular manuscript, news article, or website, were invited to attend CCMC meetings.
Scope of CCMC
In order to be effective and timely, we limited the scope of our work to the report to the inpatient therapeutic environment, allowing other committees to work on other aspects of the pandemic response. In addition to issuing guidance and creating order sets to direct clinical practice, the CCMC also monitored COVID-19 therapeutic shortages15,16 and advised on prioritization of such treatments as convalescent plasma, remdesivir (prioritization and duration of therapy, 5 vs 10 days), baricitinib, and tocilizumab, depending upon the location of the patient (critical care or not). The CCMC was not involved in the management of non–COVID-19 shortages brought about by supply chain deficiencies.
Table 1 shows some aspects of the health system pandemic-response planning and the committee workforce that undertook that work. Though many items were out of scope for the CCMC, members of the CCMC did participate in the planning work of these other committees and therefore stayed connected to this complementary work.

A Teaching Opportunity About Making Thoughtful Choices
Another important feature of the CCMC was the contributions of residents from both pharmacy and internal medicine. The purpose and operations of the committee were recognized as an opportunity to involve learners in a curriculum based on Kern’s 6-step approach.17 Though the problem identification and general needs assessment were easily defined, the targeted needs assessment, extracted from individual and group interviews with learners and the committee members, pointed at the need to learn how to assess and analyze a rapidly growing body of literature on several relevant clinical aspects of SARS-CoV-2 and COVID-19. To achieve goals and objectives, residents were assigned to present current literature on a particular intervention during a committee meeting, specifically commenting on the merit or deficiencies of the study design, the strength of the data, and applicability to the local context with a recommendation. Prior to the presentations, the residents worked with faculty to identify the best studies or systematic analyses with potential to alter current practices. We thus used the CCMC process as a teaching tool about evidence-based medicine and the dilemma of clinical equipoise. This was imperative, since trainees thrust into the COVID-19 response have often keenly observed a movement away from deliberative decision-making.18 Indeed, including residents in the process of deliberative responses to COVID-19 addresses a recent call to adjust medical education during COVID-19 to “adapt curriculum to current issues in real time.”19
Interventions and Therapies Considered
Table 2 shows the topics reviewed by the CCMC. By the time of the first meeting, nonstandardization of care was already a source of concern for clinicians. Dialogue often continued outside of the formal meetings. Many topics were considered more than once as new guidance developed, changes to EUAs occurred, and new data or new publicity arose.

Methods
The Human Protections Administrator determined that this work constituted “quality improvement, and not research” and was therefore exempt from institutional review board review.
Quantitative Analysis
All admitted patients from March 10, 2020, through April 20, 2021, were considered in the quantitative aspects of this report except as noted. Patients diagnosed with COVID-19 were identified by searching our internal data base using diagnostic codes. Patient admissions with the following diagnostic codes were included (prior to April 1, 2020): J12.89, J20.8, J40, J22, J98.8, J80, each with the additional code of B97.29. After April 1, 2020, the guideline for coding COVID-19 was U07.1.
Descriptive statistics were used to measure utilization rates of certain medications and laboratory tests of interest over time. These data were adjusted for number of unique admissions. In a few cases, not all data elements were available from both hospitals due to differences in the EMR.
Case fatality rate was calculated based upon whether the patient died or was admitted to inpatient hospice as a result of COVID-19. Four patients transferred out of hospital A and 18 transferred out of hospital B were censored from case-fatality-rate determination.
Figure 1 shows the number of admissions for each acute care hospital in the health system and the combined COVID-19 case-fatality rate over time.

Results
A total of 5955 consecutive COVID-19 patients admitted from March 10, 2020, through April 30, 2021, were analyzed. Patients with International Statistical Classification of Diseases, Tenth Revision codes J12.89. J20.8, J40, J22, J98.8, J80, each with the additional code of B97.29 (or the code UO7.1 after April 1, 2020), were included in the analysis. The median age of admitted patients was 65 years (range 19-91 years). Using the NIH classification system for severity,20 the distribution of severity during the first 24 hours after the time of hospital admission was as follows: asymptomatic/presymptomatic, 0.5%; mild illness, 5.3%; moderate illness, 37.1%; severe illness, 55.5%; and critical illness, 1.1%.
The impact of the CCMC can be estimated by looking at care patterns over time. Since the work of the CCMC was aimed at influencing and standardizing physician ordering and therapy choices through order set creation and other forms of oversight, we measured the use of the CCMC-approved order sets at both hospitals and the use of certain laboratory tests and therapies that the CCMC sought either to limit or increase. These counts were adjusted for number of unique COVID-19 admissions. But the limits of the case collection tool meant it also collected cases that were not eligible for some of the interventions. For example, COVID-19 admissions without hypoxemia would not have been eligible for remdesivir or glucocorticoids. When admitted, some patients were already on steroids for other medical indications and did not receive the prescribed dexamethasone dose that we measured in pharmacy databases. Similarly, a few patients were hospitalized for indications unrelated to COVID-19, such as surgery or childbirth, and were found to be SARS-CoV-2-positive on routine screening.
Figure 2 shows the utilization of CCMC-approved standard COVID-19 admission order sets as a proportion of all COVID-19 admissions over time. The trend reveals a modest increase in usage (R2 = 0.34), but these data do not reflect the progressive build of content into order sets over time. One of the goals of the order sets was to standardize and reduce the ordering of certain biomarkers: C-reactive protein, serum ferritin, and D-dimer, which were ordered frequently in many early patients. Orders for these 3 laboratory tests are combined and expressed as an average number of labs per COVID-19 admission in Figure 2. A downward trend, with an R2 value of 0.65, is suggestive of impact from the order sets, though other explanations are possible.

Medication guidance was also a goal of the CCMC, simultaneously discouraging poorly supported interventions and driving uptake of the recommended evidence-based interventions in appropriate patients. Figure 3 shows the utilization pattern for some drugs of interest over the course of the pandemic, specifically the proportion of patients receiving at least 1 dose of medication among all COVID-19 admissions by month. (Data for hospital B was excluded from this analysis because it did not include all admitted patients.)

Hydroxychloroquine, which enjoyed a wave of popularity early on during the pandemic, was a target of successful order stewardship through the CCMC. Use of hydroxychloroquine as a COVID-19 therapeutic option after the first 2 months of the pandemic stopped, and subsequent use at low levels likely represented continuation therapy for outpatients who took hydroxychloroquine for rheumatologic indications.
Dexamethasone, as used in the RECOVERY trial,21 had a swift uptake among physicians after it was incorporated into order sets and its use encouraged. Similarly, uptake was immediate for remdesivir when, in May 2020, preliminary reports showed at least some benefits, confirmed by later analysis,22 and it received an FDA EUA.
Our data also show successful stewardship of the interleukin-6 antagonist toclilizumab, which was discouraged early on by the CCMC due to lack of data or negative results. But in March 2021, with new studies releasing data12,13 and new recommendations14 for its use in some subsets of patients with COVID-19, this drug was encouraged in appropriate subsets. A new order set with qualifying indications was prepared by the CCMC and new educational efforts made to encourage its use in appropriate patients.
Ivermectin was nonformulary at the start of the pandemic. This drug enjoyed much publicity from media sources and was promoted by certain physicians and on websites,23 based on in-vitro activity against coronaviruses. Eventually, the World Health Organization24 and the FDA25 found it necessary to issue advisory statements to the public against its use outside of clinical trials. The CCMC had requests from physicians to incorporate ivermectin but declined to add it to the formulary and recommended not approving nonformulary requests due to lack of data. As a result, ivermectin was not used at either hospital.
Discussion
COVID-19 represents many challenges to health systems all over the world. For Luminis Health, the high volume of acutely ill patients with novel syndromes was a particular challenge for the hospital-based care teams. A flood of information from preprints, press releases, preliminary reports, and many other nontraditional sources made deliberative management decisions difficult for individual physicians. Much commentary has appeared around the phenomenon but with less practical advice about how to make day-to-day care decisions at a time of scientific uncertainty and intense pressure to intervene.26,27 The CCMC was designed to overcome the information management dilemma. The need to coordinate, standardize, and oversee care was necessary given the large number of physicians who cared for COVID-19 patients on inpatient services.
It should be noted that creating order sets and issuing guidance is necessary, but not sufficient, to achieve our goals of being updated and consistent. This is especially true with large cadres of health care workers attending COVID-19 patients. Guidelines and recommendations were reinforced by unit-based oversight and stewardship from pharmacy and other leaders who constituted the CCMC.
The reduction in COVID-19 mortality over time experienced in this health care system was not unique and cannot necessarily be attributed to standardization of care. Similar improvements in mortality have been reported at many US hospitals in aggregate.28 Many other factors, including changes in patient characteristics, may be responsible for reduction in mortality over time.
Throughout this report we have relied upon an implicit assumption that standardization of medical therapeutics is desirable and leads to better outcomes as compared with allowing unlimited empiricism by individual physicians, either consultants or hospitalists. Our program represents a single health system with 2 acute care hospitals located 25 miles apart and which thus were similarly impacted by the different phases of the pandemic. Generalizability to health systems either smaller or larger, or in different geographical areas, has not been established. Data limitations have already been discussed.
We did not measure user satisfaction with the program either from physicians or nurses. However, the high rate of compliance suggests general agreement with the content and process.
We cannot definitively ascribe reduction in utilization of some nonrecommended treatments and increased utilization of the recommended therapies to the work of the CCMC. Individual physicians may have made these adjustments on their own or under the influence of other sources.
Finally, it should be noted that the mission to rapidly respond to data from well-conducted trials might be thwarted by too rigid a process or a committee’s lack of a sense of urgency. Organizing a committee and then empowering it to act is no guarantee of success; commitment to the mission is.
Conclusion
COVID-19 represented a challenge to medical staffs everywhere, inundating them with high volumes of acutely ill patients presenting with unfamiliar syndromes. Initial responses were characterized by idiosyncratic management approaches based on nontraditional sources of opinion and influences.
This report describes how a complex medical system brought order and standardization through a deliberative, but urgent, multidisciplinary committee with responsibility for planning, implementing, and monitoring standard approaches that eventually became evidence based. The composition of the committee and its scope of influence, limited to inpatient management, were important elements of success, allowing for better focus on the many treatment decisions. The important connection between the management committee and the system P&T committee, the clinical research effort, and teaching programs in both medicine and pharmacy are offered as exemplars of coordination. The data presented show success in achieving standardized, guideline-directed care. The approach is adoptable and suitable for similar emergencies in the future.
Acknowledgments: The authors thank Gary Scabis, Kip Waite, John Moxley, Angela Clubb, and David Woodley for their assistance in gathering data. We express appreciation and admiration for all our colleagues at the bedside.
Corresponding author: Barry R. Meisenberg, MD, Department of Medicine, Luminis Health, 2001 Medical Pkwy, Annapolis, MD 21401; [email protected].
Financial disclosures: None.
From the Department of Medicine (Drs. Meisenberg, Muganlinskaya, Sharma, Amjadi, Arnold, Barnes, Clance, Khalil, Miller, Mooradian, O’Connell, Patel, Press, Samaras, Shanmugam, Tavadze, and Thompson), Department of Pharmacy (Drs. Jiang, Jarawan, Sheth, and Trinh), Department of Nursing (Dr. Ohnmacht), and Department of Women and Children’s Services (Dr. Raji), Luminis Health, Annapolis, MD, and Lanham, MD.
Objective: The COVID-19 pandemic has been a challenge for hospital medical staffs worldwide due to high volumes of patients acutely ill with novel syndromes and prevailing uncertainty regarding optimum supportive and therapeutic interventions. Additionally, the response to this crisis was driven by a plethora of nontraditional information sources, such as email chains, websites, non–peer-reviewed preprints, and press releases. Care patterns became idiosyncratic and often incorporated unproven interventions driven by these nontraditional information sources. This report evaluates the efforts of a health system to create and empower a multidisciplinary committee to develop, implement, and monitor evidence-based, standardized protocols for patients with COVID-19.
Methods: This report describes the composition of the committee, its scope, and its important interactions with the health system pharmacy and therapeutics committee, research teams, and other work groups planning other aspects of COVID-19 management. It illustrates how the committee was used to demonstrate for trainees the process and value of critically examining evidence, even in a chaotic environment.
Results: Data show successful interventions in reducing excessive ordering of certain laboratory tests, reduction of nonrecommended therapies, and rapid uptake of evidence-based or guidelines-supported interventions.
Conclusions: A multidisciplinary committee dedicated solely to planning, implementing, and monitoring standard approaches that eventually became evidence-based decision-making led to an improved focus on treatment options and outcomes for COVID-19 patients. Data presented illustrate the attainable success that is both adaptable and suitable for similar emergencies in the future.
Keywords: COVID-19; clinical management; pharmacy and therapeutics; treatment; therapy.
The COVID-19 pandemic has spread to nearly all countries, carrying with it high morbidity, mortality, and severe impacts on both well-developed and less-well-developed health systems. Media reports of chaos within overwhelmed hospitals have been prominent.1,2 As of January 5, 2022, SARS-CoV-2 has infected more than 295 million people globally and directly caused the death of more than 5.4 million,3 though this number is likely an undercount even in countries with well-developed mortality tracking.4
Throughout the COVID-19 pandemic, hospital-based medical teams have been confronted with a flood of severely ill patients with novel syndromes. Initially, there were no standards for therapy or supportive care except for treatments borrowed from similar syndromes. In the setting of high volumes, high acuity, and public dismay, it is unsurprising that the usual deliberative methods for weighing evidence and initiating interventions were often pushed aside in favor of the solace of active intervention.5 In this milieu of limited evidence, there was a lamentable, if understandable, tendency to seek guidance from “nontraditional” sources,6 including email chains from colleagues, hospital websites, non–peer-reviewed manuscripts, advanced publication by medical journals,7 and nonscientific media presentations. In many localities, practitioners responded in idiosyncratic ways. For example, findings of high cytokine levels in COVID-19,8 along with reports of in-vitro antiviral activity with drugs like hydroxychloroquine against both SARS9 and SARS-CoV-2,10 drove laboratory test ordering and therapeutic interventions, respectively, carving shortcuts into the traditional clinical trial–dependent standards. Clinical trial results eventually emerged.11COVID-19 created a clinical dilemma for hospital medical staffs in terms of how to organize, standardize, and rapidly adapt to a flood of new information. In this report, we describe how 1 health system responded to these challenges by forming a COVID-19 Clinical Management Committee (CCMC) and empowering this interdisciplinary team to review evidence, create and adjust order sets, educate practitioners, oversee care, and collaborate across teams addressing other aspects of the COVID-19 response.
Program Overview
Health System Description
Luminis Health is a health system with 2 acute care hospitals that was formed in 2019 just before the start of the pandemic. Anne Arundel Medical Center (hospital A) is a 385-bed teaching hospital in Annapolis, MD. It has more than 23 000 discharges annually. Patients with COVID-19 were cared for by either an internal medicine teaching service or nonteaching hospitalist services on cohorted nursing units. Doctor’s Community Medical Center, in Lanham, MD (hospital B), is a 206-bed acute care hospital with more than 10 350 annual discharges. COVID-19 patients were cared for by hospitalist groups, initially in noncohorted units with transition to cohorted nursing units after a few months. The medical staffs are generally distinct, with different leadership structures, though the Luminis Health Department of Medicine has oversight responsibilities at both hospitals. More than 47 physicians attended COVID-19 patients at hospital A (with medical residents) and 30 individual physicians at hospital B, respectively, including intensivists. The nursing and pharmacy staffs are distinct, but there is a shared oversight Pharmacy and Therapeutics (P&T) Committee.
The 2 hospitals had distinct electronic medical records (EMR) until January 2021, when hospital B adopted the same EMR as hospital A (Epic).
Mission and Formation of CCMC
In order to coordinate the therapeutic approach across the health system, it was important for the CCMC to be designated by the health system P&T committee as an official subcommittee so that decisions on restrictions of medications and/or new or revised order sets could be rapidly initiated across the system without waiting for the subsequent P&T meetings. The full committee retained oversight of the CCMC. Some P&T members were also on the CCMC.
The committee reviewed new reports in medical journals and prepublication servers and consulted recommendations of professional societies, such as the National Institutes of Health (NIH) COVID-19 guidelines, Infectious Diseases Society of America, Society of Critical Care Medicine, and US Food and Drug Administration (FDA) Emergency Use Authorizations (EUA), among other sources.
Composition of the CCMC
Physician leaders from both hospitals in the following specialties were solicited for participation: critical care, epidemiology, hospital medicine (internal medicine), emergency medicine, infectious diseases, nephrology, women and children’s services, and medical informatics. Specialists in other areas, such as hematology, were invited for topic-specific discussions. Hospital pharmacists with different specialties and nursing leadership were essential contributors. The committee members were expected to use various communication channels to inform frontline clinicians of new care standards and the existence of new order sets, which were embedded in the EMR.
Clinical Research
An important connection for the CCMC was with theCOVID-19 clinical research team. Three members of the research team were also members of the CCMC. All new study proposals for therapeutics were discussed with the CCMC as they were being considered by the research team. In this way, feedback on the feasibility and acceptance of new study opportunities could be discussed with the CCMC. Occasionally, CCMC decisions impacted clinical research accrual strategies. For example, new data from randomized trials about tocilizumab1,2 demonstrated benefits in some subsets of patients and resulted in a recommendation for use by the NIH guideline committee in these populations.1 The CCMC quickly adopted this recommendation, which required a reprioritization of clinical research enrollment for studies testing other immune-modulating agents. This important dialogue was mediated within the CCMC.
Guideline Distribution, Reinforcement, and Platform for Feedback
New guidelines were disseminated to clinicians via daily brief patient huddles held on COVID units, with participation by nursing and pharmacy, and by weekly meetings with hospitalist leaders and frontline hospital physicians. Order sets and guidelines were maintained on the intranet. Adherence was reinforced by unit-based and central pharmacists. Order sets, including admission order sets, could be created only by designated informatics personnel, thus enforcing standardization. Feedback on the utility of the order sets was obtained during the weekly meetings or huddles, as described above. To ensure a sense of transparency, physicians who had interest in commenting on a particular therapy, or who wished to discuss a particular manuscript, news article, or website, were invited to attend CCMC meetings.
Scope of CCMC
In order to be effective and timely, we limited the scope of our work to the report to the inpatient therapeutic environment, allowing other committees to work on other aspects of the pandemic response. In addition to issuing guidance and creating order sets to direct clinical practice, the CCMC also monitored COVID-19 therapeutic shortages15,16 and advised on prioritization of such treatments as convalescent plasma, remdesivir (prioritization and duration of therapy, 5 vs 10 days), baricitinib, and tocilizumab, depending upon the location of the patient (critical care or not). The CCMC was not involved in the management of non–COVID-19 shortages brought about by supply chain deficiencies.
Table 1 shows some aspects of the health system pandemic-response planning and the committee workforce that undertook that work. Though many items were out of scope for the CCMC, members of the CCMC did participate in the planning work of these other committees and therefore stayed connected to this complementary work.

A Teaching Opportunity About Making Thoughtful Choices
Another important feature of the CCMC was the contributions of residents from both pharmacy and internal medicine. The purpose and operations of the committee were recognized as an opportunity to involve learners in a curriculum based on Kern’s 6-step approach.17 Though the problem identification and general needs assessment were easily defined, the targeted needs assessment, extracted from individual and group interviews with learners and the committee members, pointed at the need to learn how to assess and analyze a rapidly growing body of literature on several relevant clinical aspects of SARS-CoV-2 and COVID-19. To achieve goals and objectives, residents were assigned to present current literature on a particular intervention during a committee meeting, specifically commenting on the merit or deficiencies of the study design, the strength of the data, and applicability to the local context with a recommendation. Prior to the presentations, the residents worked with faculty to identify the best studies or systematic analyses with potential to alter current practices. We thus used the CCMC process as a teaching tool about evidence-based medicine and the dilemma of clinical equipoise. This was imperative, since trainees thrust into the COVID-19 response have often keenly observed a movement away from deliberative decision-making.18 Indeed, including residents in the process of deliberative responses to COVID-19 addresses a recent call to adjust medical education during COVID-19 to “adapt curriculum to current issues in real time.”19
Interventions and Therapies Considered
Table 2 shows the topics reviewed by the CCMC. By the time of the first meeting, nonstandardization of care was already a source of concern for clinicians. Dialogue often continued outside of the formal meetings. Many topics were considered more than once as new guidance developed, changes to EUAs occurred, and new data or new publicity arose.

Methods
The Human Protections Administrator determined that this work constituted “quality improvement, and not research” and was therefore exempt from institutional review board review.
Quantitative Analysis
All admitted patients from March 10, 2020, through April 20, 2021, were considered in the quantitative aspects of this report except as noted. Patients diagnosed with COVID-19 were identified by searching our internal data base using diagnostic codes. Patient admissions with the following diagnostic codes were included (prior to April 1, 2020): J12.89, J20.8, J40, J22, J98.8, J80, each with the additional code of B97.29. After April 1, 2020, the guideline for coding COVID-19 was U07.1.
Descriptive statistics were used to measure utilization rates of certain medications and laboratory tests of interest over time. These data were adjusted for number of unique admissions. In a few cases, not all data elements were available from both hospitals due to differences in the EMR.
Case fatality rate was calculated based upon whether the patient died or was admitted to inpatient hospice as a result of COVID-19. Four patients transferred out of hospital A and 18 transferred out of hospital B were censored from case-fatality-rate determination.
Figure 1 shows the number of admissions for each acute care hospital in the health system and the combined COVID-19 case-fatality rate over time.

Results
A total of 5955 consecutive COVID-19 patients admitted from March 10, 2020, through April 30, 2021, were analyzed. Patients with International Statistical Classification of Diseases, Tenth Revision codes J12.89. J20.8, J40, J22, J98.8, J80, each with the additional code of B97.29 (or the code UO7.1 after April 1, 2020), were included in the analysis. The median age of admitted patients was 65 years (range 19-91 years). Using the NIH classification system for severity,20 the distribution of severity during the first 24 hours after the time of hospital admission was as follows: asymptomatic/presymptomatic, 0.5%; mild illness, 5.3%; moderate illness, 37.1%; severe illness, 55.5%; and critical illness, 1.1%.
The impact of the CCMC can be estimated by looking at care patterns over time. Since the work of the CCMC was aimed at influencing and standardizing physician ordering and therapy choices through order set creation and other forms of oversight, we measured the use of the CCMC-approved order sets at both hospitals and the use of certain laboratory tests and therapies that the CCMC sought either to limit or increase. These counts were adjusted for number of unique COVID-19 admissions. But the limits of the case collection tool meant it also collected cases that were not eligible for some of the interventions. For example, COVID-19 admissions without hypoxemia would not have been eligible for remdesivir or glucocorticoids. When admitted, some patients were already on steroids for other medical indications and did not receive the prescribed dexamethasone dose that we measured in pharmacy databases. Similarly, a few patients were hospitalized for indications unrelated to COVID-19, such as surgery or childbirth, and were found to be SARS-CoV-2-positive on routine screening.
Figure 2 shows the utilization of CCMC-approved standard COVID-19 admission order sets as a proportion of all COVID-19 admissions over time. The trend reveals a modest increase in usage (R2 = 0.34), but these data do not reflect the progressive build of content into order sets over time. One of the goals of the order sets was to standardize and reduce the ordering of certain biomarkers: C-reactive protein, serum ferritin, and D-dimer, which were ordered frequently in many early patients. Orders for these 3 laboratory tests are combined and expressed as an average number of labs per COVID-19 admission in Figure 2. A downward trend, with an R2 value of 0.65, is suggestive of impact from the order sets, though other explanations are possible.

Medication guidance was also a goal of the CCMC, simultaneously discouraging poorly supported interventions and driving uptake of the recommended evidence-based interventions in appropriate patients. Figure 3 shows the utilization pattern for some drugs of interest over the course of the pandemic, specifically the proportion of patients receiving at least 1 dose of medication among all COVID-19 admissions by month. (Data for hospital B was excluded from this analysis because it did not include all admitted patients.)

Hydroxychloroquine, which enjoyed a wave of popularity early on during the pandemic, was a target of successful order stewardship through the CCMC. Use of hydroxychloroquine as a COVID-19 therapeutic option after the first 2 months of the pandemic stopped, and subsequent use at low levels likely represented continuation therapy for outpatients who took hydroxychloroquine for rheumatologic indications.
Dexamethasone, as used in the RECOVERY trial,21 had a swift uptake among physicians after it was incorporated into order sets and its use encouraged. Similarly, uptake was immediate for remdesivir when, in May 2020, preliminary reports showed at least some benefits, confirmed by later analysis,22 and it received an FDA EUA.
Our data also show successful stewardship of the interleukin-6 antagonist toclilizumab, which was discouraged early on by the CCMC due to lack of data or negative results. But in March 2021, with new studies releasing data12,13 and new recommendations14 for its use in some subsets of patients with COVID-19, this drug was encouraged in appropriate subsets. A new order set with qualifying indications was prepared by the CCMC and new educational efforts made to encourage its use in appropriate patients.
Ivermectin was nonformulary at the start of the pandemic. This drug enjoyed much publicity from media sources and was promoted by certain physicians and on websites,23 based on in-vitro activity against coronaviruses. Eventually, the World Health Organization24 and the FDA25 found it necessary to issue advisory statements to the public against its use outside of clinical trials. The CCMC had requests from physicians to incorporate ivermectin but declined to add it to the formulary and recommended not approving nonformulary requests due to lack of data. As a result, ivermectin was not used at either hospital.
Discussion
COVID-19 represents many challenges to health systems all over the world. For Luminis Health, the high volume of acutely ill patients with novel syndromes was a particular challenge for the hospital-based care teams. A flood of information from preprints, press releases, preliminary reports, and many other nontraditional sources made deliberative management decisions difficult for individual physicians. Much commentary has appeared around the phenomenon but with less practical advice about how to make day-to-day care decisions at a time of scientific uncertainty and intense pressure to intervene.26,27 The CCMC was designed to overcome the information management dilemma. The need to coordinate, standardize, and oversee care was necessary given the large number of physicians who cared for COVID-19 patients on inpatient services.
It should be noted that creating order sets and issuing guidance is necessary, but not sufficient, to achieve our goals of being updated and consistent. This is especially true with large cadres of health care workers attending COVID-19 patients. Guidelines and recommendations were reinforced by unit-based oversight and stewardship from pharmacy and other leaders who constituted the CCMC.
The reduction in COVID-19 mortality over time experienced in this health care system was not unique and cannot necessarily be attributed to standardization of care. Similar improvements in mortality have been reported at many US hospitals in aggregate.28 Many other factors, including changes in patient characteristics, may be responsible for reduction in mortality over time.
Throughout this report we have relied upon an implicit assumption that standardization of medical therapeutics is desirable and leads to better outcomes as compared with allowing unlimited empiricism by individual physicians, either consultants or hospitalists. Our program represents a single health system with 2 acute care hospitals located 25 miles apart and which thus were similarly impacted by the different phases of the pandemic. Generalizability to health systems either smaller or larger, or in different geographical areas, has not been established. Data limitations have already been discussed.
We did not measure user satisfaction with the program either from physicians or nurses. However, the high rate of compliance suggests general agreement with the content and process.
We cannot definitively ascribe reduction in utilization of some nonrecommended treatments and increased utilization of the recommended therapies to the work of the CCMC. Individual physicians may have made these adjustments on their own or under the influence of other sources.
Finally, it should be noted that the mission to rapidly respond to data from well-conducted trials might be thwarted by too rigid a process or a committee’s lack of a sense of urgency. Organizing a committee and then empowering it to act is no guarantee of success; commitment to the mission is.
Conclusion
COVID-19 represented a challenge to medical staffs everywhere, inundating them with high volumes of acutely ill patients presenting with unfamiliar syndromes. Initial responses were characterized by idiosyncratic management approaches based on nontraditional sources of opinion and influences.
This report describes how a complex medical system brought order and standardization through a deliberative, but urgent, multidisciplinary committee with responsibility for planning, implementing, and monitoring standard approaches that eventually became evidence based. The composition of the committee and its scope of influence, limited to inpatient management, were important elements of success, allowing for better focus on the many treatment decisions. The important connection between the management committee and the system P&T committee, the clinical research effort, and teaching programs in both medicine and pharmacy are offered as exemplars of coordination. The data presented show success in achieving standardized, guideline-directed care. The approach is adoptable and suitable for similar emergencies in the future.
Acknowledgments: The authors thank Gary Scabis, Kip Waite, John Moxley, Angela Clubb, and David Woodley for their assistance in gathering data. We express appreciation and admiration for all our colleagues at the bedside.
Corresponding author: Barry R. Meisenberg, MD, Department of Medicine, Luminis Health, 2001 Medical Pkwy, Annapolis, MD 21401; [email protected].
Financial disclosures: None.
1. Gettleman J, Raj S, Kumar H. India’s health system cracks under the strain as coronavirus cases surge. The New York Times. April 22, 2021. https://www.nytimes.com/2021/04/21/world/asia/india-coronavirus-oxygen.html
2. Rappleye H, Lehren AW, Strickler L, Fitzpatrick S. ‘This system is doomed’: doctors, nurses sound off in NBC News coronavirus survey. NBC News. March 20, 2020. https://www.nbcnews.com/news/us-news/system-doomed-doctors-nurses-sound-nbc-news-coronavirus-survey-n1164841
3. Johns Hopkins Coronavirus Resource Center. Accessed January 5, 2022. https://coronavirus.jhu.edu/map.html
4. Fineberg HV. The toll of COVID-19. JAMA. 2020;324(15):1502-1503. doi:10.1001/jama.2020.20019
5. Meisenberg BR. Medical staffs response to COVID-19 ‘data’: have we misplaced our skeptic’s eye? Am J Med. 2021;134(2):151-152. doi:10.1016/j.amjmed.2020.09.013
6. McMahon JH, Lydeamore MH, Stewardson AJ. Bringing evidence from press release to the clinic in the era of COVID-19. J Antimicrob Chemother. 2021;76(3):547-549. doi:10.1093/jac/dkaa506
7. Rubin EJ, Baden LR, Morrissey S, Campion EW. Medical journals and the 2019-nCoV outbreak. N Engl J Med. 2020;382(9):866. doi:10.1056/NEJMe2001329
8. Liu F, Li L, Xu M, et al. Prognostic value of interleukin-6, C-reactive protein, and procalcitonin in patients with COVID-19. J Clin Virol. 2020;127:104370. doi:10.1016/j.jcv.2020.104370
9. Vincent MJ, Bergeron E, Benjannet S, et al. Chloroquine is a potent inhibitor of SARS coronavirus infection and spread. Virol J. 2005;2:69. doi:10.1186/1743-422X-2-69
10. Wang M, Cao R, Zhang L, et al. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res. 2020;30:269-271. doi:10.1038/s41422-020-0282-0
11. RECOVERY Collaborative Group. Effect of hydroxychloroquine in hospitalized patients with Covid-19. N Engl J Med. 2020;383:2030-2040. doi:10.1056/NEJMoa2022926
12. RECOVERY Collaborative Group. Tocilizumab in patients admitted to hospital with COVID-19 (RECOVERY): preliminary results of a randomised, controlled, open-label, platform trial [preprint]. February 11, 2021. doi:10.1101/2021.02.11.21249258 https://www.medrxiv.org/content/10.1101/2021.02.11.21249258v1
13. REMAP-CAP Investigators. Interleukin-6 receptor antagonists in critically ill patients with COVID-19. N Engl J Med. 2021;384(16):1491-1502. doi:10.1056/NEJMoa2100433
14. National Institutes of Health. COVID-19 treatment guidelines: interleukin-6 inhibitors. https://www.covid19treatmentguidelines.nih.gov/immunomodulators/interleukin-6-inhibitors/
15. Deana C, Vetrugno L, Tonizzo A, et al. Drug supply during COVID-19 pandemic: remember not to run with your tank empty. Hosp Pharm. 2021;56(5):405-407. doi:10.1177/0018578720931749
16. Choe J, Crane M, Greene J, et al. The Pandemic and the Supply Chain: Addressing Gaps in Pharmaceutical Production and Distribution. Johns Hopkins University, November 2020. https://www.jhsph.edu/research/affiliated-programs/johns-hopkins-drug-access-and-affordability-initiative/publications/Pandemic_Supply_Chain.pdf
17. Kern DE. Overview: a six-step approach to curriculum development. In: Kern DE, Thornton PA, Hughes MT, eds. Curriculum Development for Medical Education: A Six-Step Approach. 3rd ed. Johns Hopkins University Press; 2016.
18. Rice TW, Janz DR. In defense of evidence-based medicine for the treatment of COVID-19 acute respiratory distress syndrome. Ann Am Thorac Soc. 2020;17(7):787-789. doi:10.1513/AnnalsATS.202004-325IP
19. Lucey CR, Johnston SC. The transformational effects of COVID-19 on medical education. JAMA. 2020;324(11):1033-1034. doi:10.1001/jama.2020.14136
20. National Institutes of Health. COVID-19 treatment guidelines: clinical spectrum of SARS-CoV-2 infection. https://www.covid19treatmentguidelines.nih.gov/overview/clinical-spectrum/
21. RECOVERY Collaborative Group. Dexamethasone in hospitalized patients with Covid-19. N Engl J Med. 2021;384:693-704. doi:10.1056/NEJMoa2021436
22. Beigel JH, Tomashek KM, Dodd LE, et al. Remdesivir for the treatment of Covid-19—final report. N Engl J Med. 2020;383:1813-1826. doi:10.1056/NEJMoa2007764
23. Jiminez D. Ivermectin and Covid-19: how a cheap antiparasitic became political. April 19, 2021. https://www.pharmaceutical-technology.com/features/ivermectin-covid-19-antiparasitic-political/
24. World Health Organization. WHO advises that ivermectin only be used to treat COVID-19 within clinical trials. March 31, 2021. https://www.who.int/news-room/feature-stories/detail/who-advises-that-ivermectin-only-be-used-to-treat-covid-19-within-clinical-trials
25. U.S. Food and Drug Administration. Why you should not use ivermectin to treat or prevent COVID-19. March 5, 2021. https://www.fda.gov/consumers/consumer-updates/why-you-should-not-use-ivermectin-treat-or-prevent-covid-19
26. Seymour CW, McCreary EK, Stegenga J. Sensible medicine-balancing intervention and inaction during the COVID-19 pandemic. JAMA. 2020;324(18):1827-1828. doi:10.1001/jama.2020.20271
27. Flanagin A, Fontanarosa PB, Bauchner H. Preprints involving medical research—do the benefits outweigh the challenges? JAMA. 2020;324(18):1840-1843. doi:10.1001/jama.2020.20674
28. Asch DA, Shells NE, Islam N, et al. Variation in US hospital mortality rates for patients admitted with COVID-19 during the first 6 months of the pandemic. JAMA Intern Med. 2021;181(4):471-478. doi:10.1001/jamainternmed.2020.8193
1. Gettleman J, Raj S, Kumar H. India’s health system cracks under the strain as coronavirus cases surge. The New York Times. April 22, 2021. https://www.nytimes.com/2021/04/21/world/asia/india-coronavirus-oxygen.html
2. Rappleye H, Lehren AW, Strickler L, Fitzpatrick S. ‘This system is doomed’: doctors, nurses sound off in NBC News coronavirus survey. NBC News. March 20, 2020. https://www.nbcnews.com/news/us-news/system-doomed-doctors-nurses-sound-nbc-news-coronavirus-survey-n1164841
3. Johns Hopkins Coronavirus Resource Center. Accessed January 5, 2022. https://coronavirus.jhu.edu/map.html
4. Fineberg HV. The toll of COVID-19. JAMA. 2020;324(15):1502-1503. doi:10.1001/jama.2020.20019
5. Meisenberg BR. Medical staffs response to COVID-19 ‘data’: have we misplaced our skeptic’s eye? Am J Med. 2021;134(2):151-152. doi:10.1016/j.amjmed.2020.09.013
6. McMahon JH, Lydeamore MH, Stewardson AJ. Bringing evidence from press release to the clinic in the era of COVID-19. J Antimicrob Chemother. 2021;76(3):547-549. doi:10.1093/jac/dkaa506
7. Rubin EJ, Baden LR, Morrissey S, Campion EW. Medical journals and the 2019-nCoV outbreak. N Engl J Med. 2020;382(9):866. doi:10.1056/NEJMe2001329
8. Liu F, Li L, Xu M, et al. Prognostic value of interleukin-6, C-reactive protein, and procalcitonin in patients with COVID-19. J Clin Virol. 2020;127:104370. doi:10.1016/j.jcv.2020.104370
9. Vincent MJ, Bergeron E, Benjannet S, et al. Chloroquine is a potent inhibitor of SARS coronavirus infection and spread. Virol J. 2005;2:69. doi:10.1186/1743-422X-2-69
10. Wang M, Cao R, Zhang L, et al. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res. 2020;30:269-271. doi:10.1038/s41422-020-0282-0
11. RECOVERY Collaborative Group. Effect of hydroxychloroquine in hospitalized patients with Covid-19. N Engl J Med. 2020;383:2030-2040. doi:10.1056/NEJMoa2022926
12. RECOVERY Collaborative Group. Tocilizumab in patients admitted to hospital with COVID-19 (RECOVERY): preliminary results of a randomised, controlled, open-label, platform trial [preprint]. February 11, 2021. doi:10.1101/2021.02.11.21249258 https://www.medrxiv.org/content/10.1101/2021.02.11.21249258v1
13. REMAP-CAP Investigators. Interleukin-6 receptor antagonists in critically ill patients with COVID-19. N Engl J Med. 2021;384(16):1491-1502. doi:10.1056/NEJMoa2100433
14. National Institutes of Health. COVID-19 treatment guidelines: interleukin-6 inhibitors. https://www.covid19treatmentguidelines.nih.gov/immunomodulators/interleukin-6-inhibitors/
15. Deana C, Vetrugno L, Tonizzo A, et al. Drug supply during COVID-19 pandemic: remember not to run with your tank empty. Hosp Pharm. 2021;56(5):405-407. doi:10.1177/0018578720931749
16. Choe J, Crane M, Greene J, et al. The Pandemic and the Supply Chain: Addressing Gaps in Pharmaceutical Production and Distribution. Johns Hopkins University, November 2020. https://www.jhsph.edu/research/affiliated-programs/johns-hopkins-drug-access-and-affordability-initiative/publications/Pandemic_Supply_Chain.pdf
17. Kern DE. Overview: a six-step approach to curriculum development. In: Kern DE, Thornton PA, Hughes MT, eds. Curriculum Development for Medical Education: A Six-Step Approach. 3rd ed. Johns Hopkins University Press; 2016.
18. Rice TW, Janz DR. In defense of evidence-based medicine for the treatment of COVID-19 acute respiratory distress syndrome. Ann Am Thorac Soc. 2020;17(7):787-789. doi:10.1513/AnnalsATS.202004-325IP
19. Lucey CR, Johnston SC. The transformational effects of COVID-19 on medical education. JAMA. 2020;324(11):1033-1034. doi:10.1001/jama.2020.14136
20. National Institutes of Health. COVID-19 treatment guidelines: clinical spectrum of SARS-CoV-2 infection. https://www.covid19treatmentguidelines.nih.gov/overview/clinical-spectrum/
21. RECOVERY Collaborative Group. Dexamethasone in hospitalized patients with Covid-19. N Engl J Med. 2021;384:693-704. doi:10.1056/NEJMoa2021436
22. Beigel JH, Tomashek KM, Dodd LE, et al. Remdesivir for the treatment of Covid-19—final report. N Engl J Med. 2020;383:1813-1826. doi:10.1056/NEJMoa2007764
23. Jiminez D. Ivermectin and Covid-19: how a cheap antiparasitic became political. April 19, 2021. https://www.pharmaceutical-technology.com/features/ivermectin-covid-19-antiparasitic-political/
24. World Health Organization. WHO advises that ivermectin only be used to treat COVID-19 within clinical trials. March 31, 2021. https://www.who.int/news-room/feature-stories/detail/who-advises-that-ivermectin-only-be-used-to-treat-covid-19-within-clinical-trials
25. U.S. Food and Drug Administration. Why you should not use ivermectin to treat or prevent COVID-19. March 5, 2021. https://www.fda.gov/consumers/consumer-updates/why-you-should-not-use-ivermectin-treat-or-prevent-covid-19
26. Seymour CW, McCreary EK, Stegenga J. Sensible medicine-balancing intervention and inaction during the COVID-19 pandemic. JAMA. 2020;324(18):1827-1828. doi:10.1001/jama.2020.20271
27. Flanagin A, Fontanarosa PB, Bauchner H. Preprints involving medical research—do the benefits outweigh the challenges? JAMA. 2020;324(18):1840-1843. doi:10.1001/jama.2020.20674
28. Asch DA, Shells NE, Islam N, et al. Variation in US hospital mortality rates for patients admitted with COVID-19 during the first 6 months of the pandemic. JAMA Intern Med. 2021;181(4):471-478. doi:10.1001/jamainternmed.2020.8193
Teledermatology During the COVID-19 Pandemic: Lessons Learned and Future Directions
Although teledermatology utilization in the United States traditionally has lagged behind other countries,1,2 the COVID-19 pandemic upended this trend by creating the need for a massive teledermatology experiment. Recently reported survey results from a large representative sample of US dermatologists (5000 participants) on perceptions of teledermatology during COVID-19 indicated that only 14.1% of participants used teledermatology prior to the COVID-19 pandemic vs 54.1% of dermatologists in Europe.2,3 Since the pandemic started, 97% of US dermatologists reported teledermatology use,3 demonstrating a huge shift in utilization. This trend is notable, as teledermatology has been shown to increase access to dermatology in underserved areas, reduce patient travel times, improve patient triage, and even reduce carbon footprints.1,4 Thus, to sustain the momentum, insights from the recent teledermatology experience during the pandemic should inform future development.
Notably, the COVID-19 pandemic led to a rapid shift in focus from store-and-forward teledermatology to live video–based models.1,2 Logistically, live video visits are challenging, require more time and resources, and often are diagnostically limited, with concerns regarding technology, connectivity, reimbursement, and appropriate use.3 Prior to COVID-19, formal Health Insurance Portability and Accountability Act–compliant teledermatology platforms often were costly to establish and maintain, largely relegating use to academic centers and Veterans Affairs hospitals. Thus, many fewer private practice dermatologists had used teledermatology compared to academic dermatologists in the United States (11.4% vs 27.6%).3 Government regulations—a key barrier to the adoption of teledermatology in private practice before COVID-19—were greatly relaxed during the pandemic. The Centers for Medicare and Medicaid Services removed restrictions on where patients could be seen, improved reimbursement for video visits, and allowed the use of platforms that are not Health Insurance Portability and Accountability Act compliant. Many states also relaxed medical licensing rules.
Overall, the general outlook on telehealth seems positive. Reimbursement has been found to be a primary factor in dermatologists’ willingness to use teledermatology.3 Thus, sustainable use of teledermatology likely will depend on continued reimbursement parity for live video as well as store-and-forward consultations, which have several advantages but currently are de-incentivized by low reimbursement. The survey also found that 70% of respondents felt that teledermatology use will continue after COVID-19, while 58% intended to continue use—nearly 5-fold more than before the pandemic.3 We suspect the discrepancy between participants’ predictions regarding future use of teledermatology and their personal intent to use it highlights perceived barriers and limitations of the long-term success of teledermatology. Aside from reimbursement, connectivity and functionality were common concerns, emphasizing the need for innovative technological solutions.3 Moving forward, we anticipate that dermatologists will need to establish consistent workflows to establish consistent triage for the most appropriate visit—in-person visits vs teledermatology, which may include augmented, intelligence-enhanced solutions. Similar to prior clinician perspectives about which types of visits are conducive to teledermatology,2 most survey participants believed virtual visits were effective for acne, routine follow-ups, medication monitoring, and some inflammatory conditions.3
Importantly, we must be mindful of patients who may be left behind by the digital divide, such as those with lack of access to a smartphone or the internet, language barriers, or limited telehealth experience.5 Systems should be designed to provide these patients with technologic and health literacy aid or alternate modalities to access care. For example, structured methods could be introduced to provide training and instructions on how to access phone applications, computer-based programs, and more. Likewise, for those with hearing or vision deficits, it will be important to improve sound amplification and accessibility for headphones or hearing aid connectivity, as well as appropriate font size, button size, and application navigation. In remote areas, existing clinics may be used to help field specialty consultation teleconferences. Certainly, applications and platforms devised for teledermatology must be designed to serve diverse patient groups, with special consideration for the elderly, those who speak languages other than English, and those with disabilities that may make telehealth use more challenging.
Large-scale regulatory changes and reimbursement parity can have a substantial impact on future teledermatology use. Advocacy efforts continue to push for fair valuation of telemedicine, coverage of store-and-forward teledermatology codes, and coverage for all models of care. It is imperative for the dermatology community to continue discussions on implementation and methodology to best leverage this technology for the most patient benefit.
- Tensen E, van der Heijden JP, Jaspers MWM, et al. Two decades of teledermatology: current status and integration in national healthcare systems. Curr Dermatol Rep. 2016;5:96-104.
- Moscarella E, Pasquali P, Cinotti E, et al. A survey on teledermatology use and doctors’ perception in times of COVID-19 [published online August 17, 2020]. J Eur Acad Dermatol Venereol. 2020;34:E772-E773.
- Kennedy J, Arey S, Hopkins Z, et al. Dermatologist perceptions of teledermatology implementation and future use after COVID-19: demographics, barriers, and insights. JAMA Dermatol. 2021;157:595-597.
- Bonsall A. Unleashing carbon emissions savings with regular teledermatology clinics. Clin Exp Dermatol. 2021;46:574-575.
- Bakhtiar M, Elbuluk N, Lipoff JB. The digital divide: how COVID-19’s telemedicine expansion could exacerbate disparities. J Am Acad Dermatol. 2020;83:E345-E346.
Although teledermatology utilization in the United States traditionally has lagged behind other countries,1,2 the COVID-19 pandemic upended this trend by creating the need for a massive teledermatology experiment. Recently reported survey results from a large representative sample of US dermatologists (5000 participants) on perceptions of teledermatology during COVID-19 indicated that only 14.1% of participants used teledermatology prior to the COVID-19 pandemic vs 54.1% of dermatologists in Europe.2,3 Since the pandemic started, 97% of US dermatologists reported teledermatology use,3 demonstrating a huge shift in utilization. This trend is notable, as teledermatology has been shown to increase access to dermatology in underserved areas, reduce patient travel times, improve patient triage, and even reduce carbon footprints.1,4 Thus, to sustain the momentum, insights from the recent teledermatology experience during the pandemic should inform future development.
Notably, the COVID-19 pandemic led to a rapid shift in focus from store-and-forward teledermatology to live video–based models.1,2 Logistically, live video visits are challenging, require more time and resources, and often are diagnostically limited, with concerns regarding technology, connectivity, reimbursement, and appropriate use.3 Prior to COVID-19, formal Health Insurance Portability and Accountability Act–compliant teledermatology platforms often were costly to establish and maintain, largely relegating use to academic centers and Veterans Affairs hospitals. Thus, many fewer private practice dermatologists had used teledermatology compared to academic dermatologists in the United States (11.4% vs 27.6%).3 Government regulations—a key barrier to the adoption of teledermatology in private practice before COVID-19—were greatly relaxed during the pandemic. The Centers for Medicare and Medicaid Services removed restrictions on where patients could be seen, improved reimbursement for video visits, and allowed the use of platforms that are not Health Insurance Portability and Accountability Act compliant. Many states also relaxed medical licensing rules.
Overall, the general outlook on telehealth seems positive. Reimbursement has been found to be a primary factor in dermatologists’ willingness to use teledermatology.3 Thus, sustainable use of teledermatology likely will depend on continued reimbursement parity for live video as well as store-and-forward consultations, which have several advantages but currently are de-incentivized by low reimbursement. The survey also found that 70% of respondents felt that teledermatology use will continue after COVID-19, while 58% intended to continue use—nearly 5-fold more than before the pandemic.3 We suspect the discrepancy between participants’ predictions regarding future use of teledermatology and their personal intent to use it highlights perceived barriers and limitations of the long-term success of teledermatology. Aside from reimbursement, connectivity and functionality were common concerns, emphasizing the need for innovative technological solutions.3 Moving forward, we anticipate that dermatologists will need to establish consistent workflows to establish consistent triage for the most appropriate visit—in-person visits vs teledermatology, which may include augmented, intelligence-enhanced solutions. Similar to prior clinician perspectives about which types of visits are conducive to teledermatology,2 most survey participants believed virtual visits were effective for acne, routine follow-ups, medication monitoring, and some inflammatory conditions.3
Importantly, we must be mindful of patients who may be left behind by the digital divide, such as those with lack of access to a smartphone or the internet, language barriers, or limited telehealth experience.5 Systems should be designed to provide these patients with technologic and health literacy aid or alternate modalities to access care. For example, structured methods could be introduced to provide training and instructions on how to access phone applications, computer-based programs, and more. Likewise, for those with hearing or vision deficits, it will be important to improve sound amplification and accessibility for headphones or hearing aid connectivity, as well as appropriate font size, button size, and application navigation. In remote areas, existing clinics may be used to help field specialty consultation teleconferences. Certainly, applications and platforms devised for teledermatology must be designed to serve diverse patient groups, with special consideration for the elderly, those who speak languages other than English, and those with disabilities that may make telehealth use more challenging.
Large-scale regulatory changes and reimbursement parity can have a substantial impact on future teledermatology use. Advocacy efforts continue to push for fair valuation of telemedicine, coverage of store-and-forward teledermatology codes, and coverage for all models of care. It is imperative for the dermatology community to continue discussions on implementation and methodology to best leverage this technology for the most patient benefit.
Although teledermatology utilization in the United States traditionally has lagged behind other countries,1,2 the COVID-19 pandemic upended this trend by creating the need for a massive teledermatology experiment. Recently reported survey results from a large representative sample of US dermatologists (5000 participants) on perceptions of teledermatology during COVID-19 indicated that only 14.1% of participants used teledermatology prior to the COVID-19 pandemic vs 54.1% of dermatologists in Europe.2,3 Since the pandemic started, 97% of US dermatologists reported teledermatology use,3 demonstrating a huge shift in utilization. This trend is notable, as teledermatology has been shown to increase access to dermatology in underserved areas, reduce patient travel times, improve patient triage, and even reduce carbon footprints.1,4 Thus, to sustain the momentum, insights from the recent teledermatology experience during the pandemic should inform future development.
Notably, the COVID-19 pandemic led to a rapid shift in focus from store-and-forward teledermatology to live video–based models.1,2 Logistically, live video visits are challenging, require more time and resources, and often are diagnostically limited, with concerns regarding technology, connectivity, reimbursement, and appropriate use.3 Prior to COVID-19, formal Health Insurance Portability and Accountability Act–compliant teledermatology platforms often were costly to establish and maintain, largely relegating use to academic centers and Veterans Affairs hospitals. Thus, many fewer private practice dermatologists had used teledermatology compared to academic dermatologists in the United States (11.4% vs 27.6%).3 Government regulations—a key barrier to the adoption of teledermatology in private practice before COVID-19—were greatly relaxed during the pandemic. The Centers for Medicare and Medicaid Services removed restrictions on where patients could be seen, improved reimbursement for video visits, and allowed the use of platforms that are not Health Insurance Portability and Accountability Act compliant. Many states also relaxed medical licensing rules.
Overall, the general outlook on telehealth seems positive. Reimbursement has been found to be a primary factor in dermatologists’ willingness to use teledermatology.3 Thus, sustainable use of teledermatology likely will depend on continued reimbursement parity for live video as well as store-and-forward consultations, which have several advantages but currently are de-incentivized by low reimbursement. The survey also found that 70% of respondents felt that teledermatology use will continue after COVID-19, while 58% intended to continue use—nearly 5-fold more than before the pandemic.3 We suspect the discrepancy between participants’ predictions regarding future use of teledermatology and their personal intent to use it highlights perceived barriers and limitations of the long-term success of teledermatology. Aside from reimbursement, connectivity and functionality were common concerns, emphasizing the need for innovative technological solutions.3 Moving forward, we anticipate that dermatologists will need to establish consistent workflows to establish consistent triage for the most appropriate visit—in-person visits vs teledermatology, which may include augmented, intelligence-enhanced solutions. Similar to prior clinician perspectives about which types of visits are conducive to teledermatology,2 most survey participants believed virtual visits were effective for acne, routine follow-ups, medication monitoring, and some inflammatory conditions.3
Importantly, we must be mindful of patients who may be left behind by the digital divide, such as those with lack of access to a smartphone or the internet, language barriers, or limited telehealth experience.5 Systems should be designed to provide these patients with technologic and health literacy aid or alternate modalities to access care. For example, structured methods could be introduced to provide training and instructions on how to access phone applications, computer-based programs, and more. Likewise, for those with hearing or vision deficits, it will be important to improve sound amplification and accessibility for headphones or hearing aid connectivity, as well as appropriate font size, button size, and application navigation. In remote areas, existing clinics may be used to help field specialty consultation teleconferences. Certainly, applications and platforms devised for teledermatology must be designed to serve diverse patient groups, with special consideration for the elderly, those who speak languages other than English, and those with disabilities that may make telehealth use more challenging.
Large-scale regulatory changes and reimbursement parity can have a substantial impact on future teledermatology use. Advocacy efforts continue to push for fair valuation of telemedicine, coverage of store-and-forward teledermatology codes, and coverage for all models of care. It is imperative for the dermatology community to continue discussions on implementation and methodology to best leverage this technology for the most patient benefit.
- Tensen E, van der Heijden JP, Jaspers MWM, et al. Two decades of teledermatology: current status and integration in national healthcare systems. Curr Dermatol Rep. 2016;5:96-104.
- Moscarella E, Pasquali P, Cinotti E, et al. A survey on teledermatology use and doctors’ perception in times of COVID-19 [published online August 17, 2020]. J Eur Acad Dermatol Venereol. 2020;34:E772-E773.
- Kennedy J, Arey S, Hopkins Z, et al. Dermatologist perceptions of teledermatology implementation and future use after COVID-19: demographics, barriers, and insights. JAMA Dermatol. 2021;157:595-597.
- Bonsall A. Unleashing carbon emissions savings with regular teledermatology clinics. Clin Exp Dermatol. 2021;46:574-575.
- Bakhtiar M, Elbuluk N, Lipoff JB. The digital divide: how COVID-19’s telemedicine expansion could exacerbate disparities. J Am Acad Dermatol. 2020;83:E345-E346.
- Tensen E, van der Heijden JP, Jaspers MWM, et al. Two decades of teledermatology: current status and integration in national healthcare systems. Curr Dermatol Rep. 2016;5:96-104.
- Moscarella E, Pasquali P, Cinotti E, et al. A survey on teledermatology use and doctors’ perception in times of COVID-19 [published online August 17, 2020]. J Eur Acad Dermatol Venereol. 2020;34:E772-E773.
- Kennedy J, Arey S, Hopkins Z, et al. Dermatologist perceptions of teledermatology implementation and future use after COVID-19: demographics, barriers, and insights. JAMA Dermatol. 2021;157:595-597.
- Bonsall A. Unleashing carbon emissions savings with regular teledermatology clinics. Clin Exp Dermatol. 2021;46:574-575.
- Bakhtiar M, Elbuluk N, Lipoff JB. The digital divide: how COVID-19’s telemedicine expansion could exacerbate disparities. J Am Acad Dermatol. 2020;83:E345-E346.
Telemedicine Alopecia Assessment: Highlighting Patients With Skin of Color
Practice Gap
In accordance with World Health Organization guidelines on social distancing to limit transmission of SARS-CoV-2, dermatologists have relied on teledermatology (TD) to develop novel adaptations of traditional workflows, optimize patient care, and limit in-person appointments during the COVID-19 pandemic. Pandemic-induced physical and emotional stress were anticipated to increase the incidence of dermatologic diseases with psychologic triggers.
The connection between hair loss and emotional stress is well documented for telogen effluvium and alopecia areata.1,2 As anticipated, dermatology visits increased during the COVID-19 pandemic for the diagnosis of alopecia1-4; a survey performed during the pandemic found that alopecia was one of the most common diagnoses dermatologists made through telehealth platforms.5
This article provides a practical guide for dermatology practitioners to efficiently and accurately assess alopecia by TD in all patients, with added considerations for skin of color patients.
Diagnostic Tools
The intersection of TD, as an effective mechanism for the diagnosis and treatment of dermatologic disorders, and the increase in alopecia observed during the COVID-19 pandemic prompted us to develop a workflow for conducting virtual scalp examinations. Seven dermatologists (A.M., A.A., O.A., N.E., V.C., C.M.B., S.C.T.) who are experts in hair disorders contributed to developing workflows to optimize the assessment of alopecia through a virtual scalp examination, with an emphasis on patients of color. These experts completed a 7-question survey (Table) detailing their approach to the virtual scalp examination. One author (B.N.W.) served as an independent reviewer and collated responses into the following workflows.

Telemedicine Previsit Workflow
Components of the previsit workflow include:
• Instruct patients to provide all laboratory values and biopsy reports before the appointment.
• Test for a stable Wi-Fi connection using a speed test (available at https://www.speedtest.net/). A speed of 10 megabits/second or more is required for high-quality video via TD.6
• Provide a handout illustrating the required photographs of the anterior hairline; the mid scalp, including vertex, bilateral parietal, and occipital scalp; and posterior hairline. Photographs should be uploaded 2 hours before the visit. Figures 1 and 2 are examples of photographs that should be requested.
• Request images with 2 or 3 different angles of the area of the scalp with the greatest involvement to help appreciate primary and secondary characteristics.
• Encourage patients to present with clean, recently shampooed, dried, and detangled natural hair, unless they have an itchy or flaky scalp.
• For concerns of scalp, hairline, eyebrow, or facial flaking and scaling, instruct the patient to avoid applying a moisturizer before the visit.
• Instruct the patient to remove false eyelashes, eyelash extensions, eyebrow pencil, hair camouflage, hair accessories, braids, extensions, weaves, twists, and other hairstyles so that the hair can be maneuvered to expose the scalp surface.
• Instruct the patient to have a comb, pic, or brush, or more than one of these implements, available during the visit.
Telemedicine Visit Workflow
Components of the visit workflow include:
• If a stable Wi-Fi connection cannot be established, switch to an audio-only visit to collect a pertinent history. Advise the patient that in-person follow-up must be scheduled.
• Confirm that (1) the patient is in a private setting where the scalp can be viewed and (2) lighting is positioned in front of the patient.
• Ensure that the patient’s hairline, full face, eyebrows, and eyelashes and, upon request, the vertex and posterior scalp, are completely visible.
• Initiate the virtual scalp examination by instructing the patient how to perform a hair pull test. Then, examine the pattern and distribution of hair loss alongside supplemental photographs.
• Instruct the patient to apply pressure with the fingertips throughout the scalp to help localize tenderness, which, in combination with the pattern of hair loss observed, might inform the diagnosis.
• Instruct the patient to scan the scalp with the fingertips for “bumps” to locate papules, pustules, and keloidal scars.
Diagnostic Pearls
Distribution of Alopecia—The experts noted that the pattern, distribution, and location of hair loss determined from the telemedicine alopecia assessment provided important clues to distinguish the type of alopecia.
Diagnostic clues for diffuse or generalized alopecia include:
• Either of these findings might be indicative of telogen effluvium or acquired trichorrhexis nodosa. Results of the hair pull test can help distinguish between these diagnoses.
• Recent stressful life events along with the presence of telogen hairs extracted during a hair pull test support the diagnosis of telogen effluvium.
• A history of external stress on the hair—thermal, traction, or chemical—along with broken hair shafts following the hair pull test support the diagnosis of acquired trichorrhexis nodosa.
Diagnostic clues for focal or patchy alopecia include:
• Alopecia areata generally presents as focal hair loss in an annular distribution; pruritus, erythema, and scale are absent.
• Seborrheic dermatitis can present as pruritic erythematous patches with scale distributed on the scalp and, in some cases, in the eyebrows, nasolabial folds, or paranasal skin.7 Some skin of color patients present with petaloid seborrheic dermatitis—pink or hypopigmented polycyclic coalescing rings with minimal scale.7,8
• Discoid lupus erythematosus, similar to seborrheic dermatitis, might present as pruritic, scaly, hypopigmented patches. However, in the experience of the experts, a more common presentation is tender erythematous patches of hair loss with central hypopigmentation and surrounding hyperpigmentation.
Diagnostic clues for vertex and mid scalp alopecia include:
• Androgenetic alopecia typically presents as a reduction of terminal hair density in the vertex and mid scalp regions (with widening through the midline part) and fine hair along the anterior hairline.9 Signs of concomitant hyperandrogenism, including facial hirsutism, acne, and obesity, might be observed.10
• Central centrifugal cicatricial alopecia typically affects the vertex and mid scalp with a shiny scalp appearance and follicular dropout.
Diagnostic clues for frontotemporal alopecia include:
• Frontal fibrosing alopecia (FFA) often presents with spared single terminal hairs (lonely hair sign).
• Traction alopecia commonly presents with the fringe hair sign.
Scalp Symptoms—The experts noted that the presence of symptoms (eg, pain, tenderness, pruritus) in conjunction with the pattern of hair loss might support the diagnosis of an inflammatory scarring alopecia.
When do symptoms raise suspicion of central centrifugal cicatricial alopecia?
• Suspected in the setting of vertex alopecia associated with tenderness, pain, or itching.
When do symptoms raise suspicion of FFA?
• Suspected when patients experience frontotemporal tenderness, pain, or burning associated with alopecia.
• The skin hue of the affected area might be lighter in color than, and contrast with, the darker hue of the photoaged upper forehead.11
• The lonely hair sign can aid in diagnosing FFA and distinguish it from the fringe sign of traction alopecia.
• Concurrent madarosis, flesh-colored papules on the cheeks, or lichen planus pigmentosus identified by visual inspection of the face confirms the diagnosis.9,12 Madarosis of the eyebrow was frequently cited by the experts as an associated symptom of FFA.
When do symptoms raise suspicion of lichen planopilaris?
• Suspected in the presence of pruritus, burning, tenderness, or pain associated with perifollicular erythema and scale in the setting of vertex and parietal alopecia.13
• Anagen hair release is observed during the hair pull test.11,14• The experts cited flesh-colored papules and lichen planus pigmentosus as frequently associated symptoms of lichen planopilaris.
Practice Implications
There are limitations to a virtual scalp examination—the inability to perform a scalp biopsy or administer certain treatments—but the consensus of the expert panel is that an initial alopecia assessment can be completed successfully utilizing TD. Although TD is not a replacement for an in-person dermatology visit, this technology has allowed for the diagnosis, treatment, and continuing care of many common dermatologic conditions without the patient needing to travel to the office.5
With the increased frequency of hair loss concerns documented over the last year and more patients seeking TD, it is imperative that dermatologists feel confident performing a virtual hair and scalp examination on all patients.1,3,4
- Kutlu Ö, Aktas¸ H, I·mren IG, et al. Short-term stress-related increasing cases of alopecia areata during the COVID-19 pandemic. J Dermatolog Treat. 2020;1. doi:10.1080/09546634.2020.1782820
- Cline A, Kazemi A, Moy J, et al. A surge in the incidence of telogen effluvium in minority predominant communities heavily impacted by COVID-19. J Am Acad Dermatol. 2021;84:773-775. doi:10.1016/j.jaad.2020.11.032
- Kutlu Ö, Metin A. Relative changes in the pattern of diseases presenting in dermatology outpatient clinic in the era of the COVID-19 pandemic. Dermatol Ther. 2020;33:e14096. doi:10.1111/dth.14096
- Tanacan E, Aksoy Sarac G, Emeksiz MAC, et al. Changing trends in dermatology practice during COVID-19 pandemic: a single tertiary center experience. Dermatol Ther. 2020;33:e14136. doi:10.1111/dth.14136
- Sharma A, Jindal V, Singla P, et al. Will teledermatology be the silver lining during and after COVID-19? Dermatol Ther. 2020;33:e13643. doi:10.1111/dth.13643
- Iscrupe L. How to receive virtual medical treatment while under quarantine. Allconnect website. Published March 26, 2020. Accessed December 9, 2021. https://www.allconnect.com/blog/online-doctor-visit-faq
- Elgash M, Dlova N, Ogunleye T, et al. Seborrheic dermatitis in skin of color: clinical considerations. J Drugs Dermatol. 2019;18:24-27.
- McLaurin CI. Annular facial dermatoses in blacks. Cutis. 1983;32:369-370, 384.
- Suchonwanit P, Hector CE, Bin Saif GA, McMichael AJ. Factors affecting the severity of central centrifugal cicatricial alopecia. Int J Dermatol. 2016;55:e338-343. doi:10.1111/ijd.13061
- Gabros S, Masood S. Central centrifugal cicatricial alopecia. StatPearls [Internet]. StatPearls Publishing; 2021. Updated July 20, 2021. Accessed December 9, 2021. https://www.ncbi.nlm.nih.gov/books/NBK559187/
- Ross EK, Tan E, Shapiro J. Update on primary cicatricial alopecias. J Am Acad Dermatol. 2005;53:1-37. doi:10.1016/j.jaad.2004.06.015
- Cobos G, Kim RH, Meehan S, et al. Lichen planus pigmentosus and lichen planopilaris. Dermatol Online J. 2016;22:13030/qt7hp8n6dn.
- Lyakhovitsky A, Amichai B, Sizopoulou C, et al. A case series of 46 patients with lichen planopilaris: demographics, clinical evaluation, and treatment experience. J Dermatolog Treat. 2015;26:275-279. doi:10.3109/09546634.2014.933165
- Tan E, Martinka M, Ball N, et al. Primary cicatricial alopecias: clinicopathology of 112 cases. J Am Acad Dermatol. 2004;50:25-32. doi:10.1016/j.jaad.2003.04.001
Practice Gap
In accordance with World Health Organization guidelines on social distancing to limit transmission of SARS-CoV-2, dermatologists have relied on teledermatology (TD) to develop novel adaptations of traditional workflows, optimize patient care, and limit in-person appointments during the COVID-19 pandemic. Pandemic-induced physical and emotional stress were anticipated to increase the incidence of dermatologic diseases with psychologic triggers.
The connection between hair loss and emotional stress is well documented for telogen effluvium and alopecia areata.1,2 As anticipated, dermatology visits increased during the COVID-19 pandemic for the diagnosis of alopecia1-4; a survey performed during the pandemic found that alopecia was one of the most common diagnoses dermatologists made through telehealth platforms.5
This article provides a practical guide for dermatology practitioners to efficiently and accurately assess alopecia by TD in all patients, with added considerations for skin of color patients.
Diagnostic Tools
The intersection of TD, as an effective mechanism for the diagnosis and treatment of dermatologic disorders, and the increase in alopecia observed during the COVID-19 pandemic prompted us to develop a workflow for conducting virtual scalp examinations. Seven dermatologists (A.M., A.A., O.A., N.E., V.C., C.M.B., S.C.T.) who are experts in hair disorders contributed to developing workflows to optimize the assessment of alopecia through a virtual scalp examination, with an emphasis on patients of color. These experts completed a 7-question survey (Table) detailing their approach to the virtual scalp examination. One author (B.N.W.) served as an independent reviewer and collated responses into the following workflows.

Telemedicine Previsit Workflow
Components of the previsit workflow include:
• Instruct patients to provide all laboratory values and biopsy reports before the appointment.
• Test for a stable Wi-Fi connection using a speed test (available at https://www.speedtest.net/). A speed of 10 megabits/second or more is required for high-quality video via TD.6

• Provide a handout illustrating the required photographs of the anterior hairline; the mid scalp, including vertex, bilateral parietal, and occipital scalp; and posterior hairline. Photographs should be uploaded 2 hours before the visit. Figures 1 and 2 are examples of photographs that should be requested.

• Request images with 2 or 3 different angles of the area of the scalp with the greatest involvement to help appreciate primary and secondary characteristics.
• Encourage patients to present with clean, recently shampooed, dried, and detangled natural hair, unless they have an itchy or flaky scalp.
• For concerns of scalp, hairline, eyebrow, or facial flaking and scaling, instruct the patient to avoid applying a moisturizer before the visit.
• Instruct the patient to remove false eyelashes, eyelash extensions, eyebrow pencil, hair camouflage, hair accessories, braids, extensions, weaves, twists, and other hairstyles so that the hair can be maneuvered to expose the scalp surface.
• Instruct the patient to have a comb, pic, or brush, or more than one of these implements, available during the visit.
Telemedicine Visit Workflow
Components of the visit workflow include:
• If a stable Wi-Fi connection cannot be established, switch to an audio-only visit to collect a pertinent history. Advise the patient that in-person follow-up must be scheduled.
• Confirm that (1) the patient is in a private setting where the scalp can be viewed and (2) lighting is positioned in front of the patient.
• Ensure that the patient’s hairline, full face, eyebrows, and eyelashes and, upon request, the vertex and posterior scalp, are completely visible.
• Initiate the virtual scalp examination by instructing the patient how to perform a hair pull test. Then, examine the pattern and distribution of hair loss alongside supplemental photographs.
• Instruct the patient to apply pressure with the fingertips throughout the scalp to help localize tenderness, which, in combination with the pattern of hair loss observed, might inform the diagnosis.
• Instruct the patient to scan the scalp with the fingertips for “bumps” to locate papules, pustules, and keloidal scars.
Diagnostic Pearls
Distribution of Alopecia—The experts noted that the pattern, distribution, and location of hair loss determined from the telemedicine alopecia assessment provided important clues to distinguish the type of alopecia.
Diagnostic clues for diffuse or generalized alopecia include:
• Either of these findings might be indicative of telogen effluvium or acquired trichorrhexis nodosa. Results of the hair pull test can help distinguish between these diagnoses.
• Recent stressful life events along with the presence of telogen hairs extracted during a hair pull test support the diagnosis of telogen effluvium.
• A history of external stress on the hair—thermal, traction, or chemical—along with broken hair shafts following the hair pull test support the diagnosis of acquired trichorrhexis nodosa.
Diagnostic clues for focal or patchy alopecia include:
• Alopecia areata generally presents as focal hair loss in an annular distribution; pruritus, erythema, and scale are absent.
• Seborrheic dermatitis can present as pruritic erythematous patches with scale distributed on the scalp and, in some cases, in the eyebrows, nasolabial folds, or paranasal skin.7 Some skin of color patients present with petaloid seborrheic dermatitis—pink or hypopigmented polycyclic coalescing rings with minimal scale.7,8
• Discoid lupus erythematosus, similar to seborrheic dermatitis, might present as pruritic, scaly, hypopigmented patches. However, in the experience of the experts, a more common presentation is tender erythematous patches of hair loss with central hypopigmentation and surrounding hyperpigmentation.
Diagnostic clues for vertex and mid scalp alopecia include:
• Androgenetic alopecia typically presents as a reduction of terminal hair density in the vertex and mid scalp regions (with widening through the midline part) and fine hair along the anterior hairline.9 Signs of concomitant hyperandrogenism, including facial hirsutism, acne, and obesity, might be observed.10
• Central centrifugal cicatricial alopecia typically affects the vertex and mid scalp with a shiny scalp appearance and follicular dropout.
Diagnostic clues for frontotemporal alopecia include:
• Frontal fibrosing alopecia (FFA) often presents with spared single terminal hairs (lonely hair sign).
• Traction alopecia commonly presents with the fringe hair sign.
Scalp Symptoms—The experts noted that the presence of symptoms (eg, pain, tenderness, pruritus) in conjunction with the pattern of hair loss might support the diagnosis of an inflammatory scarring alopecia.
When do symptoms raise suspicion of central centrifugal cicatricial alopecia?
• Suspected in the setting of vertex alopecia associated with tenderness, pain, or itching.
When do symptoms raise suspicion of FFA?
• Suspected when patients experience frontotemporal tenderness, pain, or burning associated with alopecia.
• The skin hue of the affected area might be lighter in color than, and contrast with, the darker hue of the photoaged upper forehead.11
• The lonely hair sign can aid in diagnosing FFA and distinguish it from the fringe sign of traction alopecia.
• Concurrent madarosis, flesh-colored papules on the cheeks, or lichen planus pigmentosus identified by visual inspection of the face confirms the diagnosis.9,12 Madarosis of the eyebrow was frequently cited by the experts as an associated symptom of FFA.
When do symptoms raise suspicion of lichen planopilaris?
• Suspected in the presence of pruritus, burning, tenderness, or pain associated with perifollicular erythema and scale in the setting of vertex and parietal alopecia.13
• Anagen hair release is observed during the hair pull test.11,14• The experts cited flesh-colored papules and lichen planus pigmentosus as frequently associated symptoms of lichen planopilaris.
Practice Implications
There are limitations to a virtual scalp examination—the inability to perform a scalp biopsy or administer certain treatments—but the consensus of the expert panel is that an initial alopecia assessment can be completed successfully utilizing TD. Although TD is not a replacement for an in-person dermatology visit, this technology has allowed for the diagnosis, treatment, and continuing care of many common dermatologic conditions without the patient needing to travel to the office.5
With the increased frequency of hair loss concerns documented over the last year and more patients seeking TD, it is imperative that dermatologists feel confident performing a virtual hair and scalp examination on all patients.1,3,4
Practice Gap
In accordance with World Health Organization guidelines on social distancing to limit transmission of SARS-CoV-2, dermatologists have relied on teledermatology (TD) to develop novel adaptations of traditional workflows, optimize patient care, and limit in-person appointments during the COVID-19 pandemic. Pandemic-induced physical and emotional stress were anticipated to increase the incidence of dermatologic diseases with psychologic triggers.
The connection between hair loss and emotional stress is well documented for telogen effluvium and alopecia areata.1,2 As anticipated, dermatology visits increased during the COVID-19 pandemic for the diagnosis of alopecia1-4; a survey performed during the pandemic found that alopecia was one of the most common diagnoses dermatologists made through telehealth platforms.5
This article provides a practical guide for dermatology practitioners to efficiently and accurately assess alopecia by TD in all patients, with added considerations for skin of color patients.
Diagnostic Tools
The intersection of TD, as an effective mechanism for the diagnosis and treatment of dermatologic disorders, and the increase in alopecia observed during the COVID-19 pandemic prompted us to develop a workflow for conducting virtual scalp examinations. Seven dermatologists (A.M., A.A., O.A., N.E., V.C., C.M.B., S.C.T.) who are experts in hair disorders contributed to developing workflows to optimize the assessment of alopecia through a virtual scalp examination, with an emphasis on patients of color. These experts completed a 7-question survey (Table) detailing their approach to the virtual scalp examination. One author (B.N.W.) served as an independent reviewer and collated responses into the following workflows.

Telemedicine Previsit Workflow
Components of the previsit workflow include:
• Instruct patients to provide all laboratory values and biopsy reports before the appointment.
• Test for a stable Wi-Fi connection using a speed test (available at https://www.speedtest.net/). A speed of 10 megabits/second or more is required for high-quality video via TD.6

• Provide a handout illustrating the required photographs of the anterior hairline; the mid scalp, including vertex, bilateral parietal, and occipital scalp; and posterior hairline. Photographs should be uploaded 2 hours before the visit. Figures 1 and 2 are examples of photographs that should be requested.

• Request images with 2 or 3 different angles of the area of the scalp with the greatest involvement to help appreciate primary and secondary characteristics.
• Encourage patients to present with clean, recently shampooed, dried, and detangled natural hair, unless they have an itchy or flaky scalp.
• For concerns of scalp, hairline, eyebrow, or facial flaking and scaling, instruct the patient to avoid applying a moisturizer before the visit.
• Instruct the patient to remove false eyelashes, eyelash extensions, eyebrow pencil, hair camouflage, hair accessories, braids, extensions, weaves, twists, and other hairstyles so that the hair can be maneuvered to expose the scalp surface.
• Instruct the patient to have a comb, pic, or brush, or more than one of these implements, available during the visit.
Telemedicine Visit Workflow
Components of the visit workflow include:
• If a stable Wi-Fi connection cannot be established, switch to an audio-only visit to collect a pertinent history. Advise the patient that in-person follow-up must be scheduled.
• Confirm that (1) the patient is in a private setting where the scalp can be viewed and (2) lighting is positioned in front of the patient.
• Ensure that the patient’s hairline, full face, eyebrows, and eyelashes and, upon request, the vertex and posterior scalp, are completely visible.
• Initiate the virtual scalp examination by instructing the patient how to perform a hair pull test. Then, examine the pattern and distribution of hair loss alongside supplemental photographs.
• Instruct the patient to apply pressure with the fingertips throughout the scalp to help localize tenderness, which, in combination with the pattern of hair loss observed, might inform the diagnosis.
• Instruct the patient to scan the scalp with the fingertips for “bumps” to locate papules, pustules, and keloidal scars.
Diagnostic Pearls
Distribution of Alopecia—The experts noted that the pattern, distribution, and location of hair loss determined from the telemedicine alopecia assessment provided important clues to distinguish the type of alopecia.
Diagnostic clues for diffuse or generalized alopecia include:
• Either of these findings might be indicative of telogen effluvium or acquired trichorrhexis nodosa. Results of the hair pull test can help distinguish between these diagnoses.
• Recent stressful life events along with the presence of telogen hairs extracted during a hair pull test support the diagnosis of telogen effluvium.
• A history of external stress on the hair—thermal, traction, or chemical—along with broken hair shafts following the hair pull test support the diagnosis of acquired trichorrhexis nodosa.
Diagnostic clues for focal or patchy alopecia include:
• Alopecia areata generally presents as focal hair loss in an annular distribution; pruritus, erythema, and scale are absent.
• Seborrheic dermatitis can present as pruritic erythematous patches with scale distributed on the scalp and, in some cases, in the eyebrows, nasolabial folds, or paranasal skin.7 Some skin of color patients present with petaloid seborrheic dermatitis—pink or hypopigmented polycyclic coalescing rings with minimal scale.7,8
• Discoid lupus erythematosus, similar to seborrheic dermatitis, might present as pruritic, scaly, hypopigmented patches. However, in the experience of the experts, a more common presentation is tender erythematous patches of hair loss with central hypopigmentation and surrounding hyperpigmentation.
Diagnostic clues for vertex and mid scalp alopecia include:
• Androgenetic alopecia typically presents as a reduction of terminal hair density in the vertex and mid scalp regions (with widening through the midline part) and fine hair along the anterior hairline.9 Signs of concomitant hyperandrogenism, including facial hirsutism, acne, and obesity, might be observed.10
• Central centrifugal cicatricial alopecia typically affects the vertex and mid scalp with a shiny scalp appearance and follicular dropout.
Diagnostic clues for frontotemporal alopecia include:
• Frontal fibrosing alopecia (FFA) often presents with spared single terminal hairs (lonely hair sign).
• Traction alopecia commonly presents with the fringe hair sign.
Scalp Symptoms—The experts noted that the presence of symptoms (eg, pain, tenderness, pruritus) in conjunction with the pattern of hair loss might support the diagnosis of an inflammatory scarring alopecia.
When do symptoms raise suspicion of central centrifugal cicatricial alopecia?
• Suspected in the setting of vertex alopecia associated with tenderness, pain, or itching.
When do symptoms raise suspicion of FFA?
• Suspected when patients experience frontotemporal tenderness, pain, or burning associated with alopecia.
• The skin hue of the affected area might be lighter in color than, and contrast with, the darker hue of the photoaged upper forehead.11
• The lonely hair sign can aid in diagnosing FFA and distinguish it from the fringe sign of traction alopecia.
• Concurrent madarosis, flesh-colored papules on the cheeks, or lichen planus pigmentosus identified by visual inspection of the face confirms the diagnosis.9,12 Madarosis of the eyebrow was frequently cited by the experts as an associated symptom of FFA.
When do symptoms raise suspicion of lichen planopilaris?
• Suspected in the presence of pruritus, burning, tenderness, or pain associated with perifollicular erythema and scale in the setting of vertex and parietal alopecia.13
• Anagen hair release is observed during the hair pull test.11,14• The experts cited flesh-colored papules and lichen planus pigmentosus as frequently associated symptoms of lichen planopilaris.
Practice Implications
There are limitations to a virtual scalp examination—the inability to perform a scalp biopsy or administer certain treatments—but the consensus of the expert panel is that an initial alopecia assessment can be completed successfully utilizing TD. Although TD is not a replacement for an in-person dermatology visit, this technology has allowed for the diagnosis, treatment, and continuing care of many common dermatologic conditions without the patient needing to travel to the office.5
With the increased frequency of hair loss concerns documented over the last year and more patients seeking TD, it is imperative that dermatologists feel confident performing a virtual hair and scalp examination on all patients.1,3,4
- Kutlu Ö, Aktas¸ H, I·mren IG, et al. Short-term stress-related increasing cases of alopecia areata during the COVID-19 pandemic. J Dermatolog Treat. 2020;1. doi:10.1080/09546634.2020.1782820
- Cline A, Kazemi A, Moy J, et al. A surge in the incidence of telogen effluvium in minority predominant communities heavily impacted by COVID-19. J Am Acad Dermatol. 2021;84:773-775. doi:10.1016/j.jaad.2020.11.032
- Kutlu Ö, Metin A. Relative changes in the pattern of diseases presenting in dermatology outpatient clinic in the era of the COVID-19 pandemic. Dermatol Ther. 2020;33:e14096. doi:10.1111/dth.14096
- Tanacan E, Aksoy Sarac G, Emeksiz MAC, et al. Changing trends in dermatology practice during COVID-19 pandemic: a single tertiary center experience. Dermatol Ther. 2020;33:e14136. doi:10.1111/dth.14136
- Sharma A, Jindal V, Singla P, et al. Will teledermatology be the silver lining during and after COVID-19? Dermatol Ther. 2020;33:e13643. doi:10.1111/dth.13643
- Iscrupe L. How to receive virtual medical treatment while under quarantine. Allconnect website. Published March 26, 2020. Accessed December 9, 2021. https://www.allconnect.com/blog/online-doctor-visit-faq
- Elgash M, Dlova N, Ogunleye T, et al. Seborrheic dermatitis in skin of color: clinical considerations. J Drugs Dermatol. 2019;18:24-27.
- McLaurin CI. Annular facial dermatoses in blacks. Cutis. 1983;32:369-370, 384.
- Suchonwanit P, Hector CE, Bin Saif GA, McMichael AJ. Factors affecting the severity of central centrifugal cicatricial alopecia. Int J Dermatol. 2016;55:e338-343. doi:10.1111/ijd.13061
- Gabros S, Masood S. Central centrifugal cicatricial alopecia. StatPearls [Internet]. StatPearls Publishing; 2021. Updated July 20, 2021. Accessed December 9, 2021. https://www.ncbi.nlm.nih.gov/books/NBK559187/
- Ross EK, Tan E, Shapiro J. Update on primary cicatricial alopecias. J Am Acad Dermatol. 2005;53:1-37. doi:10.1016/j.jaad.2004.06.015
- Cobos G, Kim RH, Meehan S, et al. Lichen planus pigmentosus and lichen planopilaris. Dermatol Online J. 2016;22:13030/qt7hp8n6dn.
- Lyakhovitsky A, Amichai B, Sizopoulou C, et al. A case series of 46 patients with lichen planopilaris: demographics, clinical evaluation, and treatment experience. J Dermatolog Treat. 2015;26:275-279. doi:10.3109/09546634.2014.933165
- Tan E, Martinka M, Ball N, et al. Primary cicatricial alopecias: clinicopathology of 112 cases. J Am Acad Dermatol. 2004;50:25-32. doi:10.1016/j.jaad.2003.04.001
- Kutlu Ö, Aktas¸ H, I·mren IG, et al. Short-term stress-related increasing cases of alopecia areata during the COVID-19 pandemic. J Dermatolog Treat. 2020;1. doi:10.1080/09546634.2020.1782820
- Cline A, Kazemi A, Moy J, et al. A surge in the incidence of telogen effluvium in minority predominant communities heavily impacted by COVID-19. J Am Acad Dermatol. 2021;84:773-775. doi:10.1016/j.jaad.2020.11.032
- Kutlu Ö, Metin A. Relative changes in the pattern of diseases presenting in dermatology outpatient clinic in the era of the COVID-19 pandemic. Dermatol Ther. 2020;33:e14096. doi:10.1111/dth.14096
- Tanacan E, Aksoy Sarac G, Emeksiz MAC, et al. Changing trends in dermatology practice during COVID-19 pandemic: a single tertiary center experience. Dermatol Ther. 2020;33:e14136. doi:10.1111/dth.14136
- Sharma A, Jindal V, Singla P, et al. Will teledermatology be the silver lining during and after COVID-19? Dermatol Ther. 2020;33:e13643. doi:10.1111/dth.13643
- Iscrupe L. How to receive virtual medical treatment while under quarantine. Allconnect website. Published March 26, 2020. Accessed December 9, 2021. https://www.allconnect.com/blog/online-doctor-visit-faq
- Elgash M, Dlova N, Ogunleye T, et al. Seborrheic dermatitis in skin of color: clinical considerations. J Drugs Dermatol. 2019;18:24-27.
- McLaurin CI. Annular facial dermatoses in blacks. Cutis. 1983;32:369-370, 384.
- Suchonwanit P, Hector CE, Bin Saif GA, McMichael AJ. Factors affecting the severity of central centrifugal cicatricial alopecia. Int J Dermatol. 2016;55:e338-343. doi:10.1111/ijd.13061
- Gabros S, Masood S. Central centrifugal cicatricial alopecia. StatPearls [Internet]. StatPearls Publishing; 2021. Updated July 20, 2021. Accessed December 9, 2021. https://www.ncbi.nlm.nih.gov/books/NBK559187/
- Ross EK, Tan E, Shapiro J. Update on primary cicatricial alopecias. J Am Acad Dermatol. 2005;53:1-37. doi:10.1016/j.jaad.2004.06.015
- Cobos G, Kim RH, Meehan S, et al. Lichen planus pigmentosus and lichen planopilaris. Dermatol Online J. 2016;22:13030/qt7hp8n6dn.
- Lyakhovitsky A, Amichai B, Sizopoulou C, et al. A case series of 46 patients with lichen planopilaris: demographics, clinical evaluation, and treatment experience. J Dermatolog Treat. 2015;26:275-279. doi:10.3109/09546634.2014.933165
- Tan E, Martinka M, Ball N, et al. Primary cicatricial alopecias: clinicopathology of 112 cases. J Am Acad Dermatol. 2004;50:25-32. doi:10.1016/j.jaad.2003.04.001