User login
Trump could clean house at health agencies
Others may soon depart voluntarily. Politico reported in late October that more than two dozen political appointees had already left the U.S. Department Health and Human Services (HHS) since the start of the COVID-19 pandemic in February and that potentially dozens of the more than 100 in the department would leave if Trump was not reelected.
Trump hasn’t conceded, he is challenging the election results, and he has already fired his Defense Secretary, Mark Esper.
Among those possibly in Trump’s sights: HHS Secretary Alex Azar, US Food and Drug Administration (FDA) Commissioner Stephen Hahn, MD, Centers for Disease Control and Prevention (CDC) Director Robert Redfield, MD, and White House Coronavirus Task Force member Anthony Fauci, MD, who is also the director of the National Institutes of Allergy and Infectious Diseases.
Seema Verma, the administrator of the Centers for Medicare & Medicaid Services (CMS), is likely safe. According to Politico, Verma is expected to leave on her own terms.
Azar has had a long run as a Trump appointee. He took office in January 2018 and has been a staunch loyalist. But he’s frequently been the butt of grousing by Trump for not doing enough to help lower drug prices and for his handling of the coronavirus pandemic. Azar was initially in charge of the Trump virus effort but was quickly replaced by Vice President Mike Pence.
It was widely reported in late April that Trump was considering firing Azar, but the president called that “fake news” in a tweet.
Azar has complained about Hahn, who was confirmed in December 2019. According to Politico, Azar was looking into how to remove Hahn as commissioner because of the FDA’s battle with the White House over standards for emergency use authorization of a coronavirus vaccine.
In addition, Trump was infuriated by the agency’s insistence that it stick to the highest bar for an emergency approval. “The deep state, or whoever, over at the FDA is making it very difficult for drug companies to get people in order to test the vaccines and therapeutics. Obviously, they are hoping to delay the answer until after November 3rd,” Trump tweeted at Hahn.
Fauci on the firing line?
Most of the president’s ire has been directed at Fauci. As far back as April, Trump retweeted a call for Fauci’s firing. Twitter removed the original tweet but kept Trump’s comments on the original tweet.
The president has frequently questioned Fauci’s advice, sidelined him from task force meetings, and infrequently met with him. Trump called Fauci a “disaster” during a call with supporters in October, and then, at a campaign rally in November, intimated that he would fire the scientist after the election, according to The Washington Post.
But such a firing cannot be easily done. Some have speculated that Trump could pressure Fauci’s boss, Francis Collins, MD, PhD — the director of the National Institutes of Health (NIH), who is a political appointee — to get rid of him. But Collins would have to come up with a reason to fire Fauci. Because he is not a political appointee, Fauci is afforded a raft of protections given to civil service employees of the federal government.
To demote or fire Fauci, Collins would have to give him 30 days’ notice unless there’s a belief that he committed a crime. Fauci would have at least a week to offer evidence and affidavits in support of his service.
He’d also be entitled to legal representation, a written decision, and the specific reasons for the action being taken quickly. He could also request a hearing, and he’d be able to appeal any action to the Merit Systems Protection Board. The process could take months, if not years.
In late October, Trump issued an executive order that would reclassify certain federal employees so that they wouldn’t have such protections. But agencies have until mid-January to come up with lists of such workers, according to Government Executive.
Collins has been with NIH since 1993, when he headed the Human Genome Project and the National Human Genome Research Institute. Politico has speculated that Collins, 70, might retire if Trump was reelected. It’s unclear what he’ll do now.
Redfield, who has taken heat for his leadership from many in public health — and was asked in October to stand up to Trump by former CDC Director William H. Foege, MD — has been openly contradicted by the president on more than one occasion, according to The New York Times.
In September, The Hill reported that Trump told reporters that he’d chastised Redfield by phone soon after Redfield had told a Senate committee that a coronavirus vaccine would not be available until mid-2021.
This article first appeared on Medscape.com.
Others may soon depart voluntarily. Politico reported in late October that more than two dozen political appointees had already left the U.S. Department Health and Human Services (HHS) since the start of the COVID-19 pandemic in February and that potentially dozens of the more than 100 in the department would leave if Trump was not reelected.
Trump hasn’t conceded, he is challenging the election results, and he has already fired his Defense Secretary, Mark Esper.
Among those possibly in Trump’s sights: HHS Secretary Alex Azar, US Food and Drug Administration (FDA) Commissioner Stephen Hahn, MD, Centers for Disease Control and Prevention (CDC) Director Robert Redfield, MD, and White House Coronavirus Task Force member Anthony Fauci, MD, who is also the director of the National Institutes of Allergy and Infectious Diseases.
Seema Verma, the administrator of the Centers for Medicare & Medicaid Services (CMS), is likely safe. According to Politico, Verma is expected to leave on her own terms.
Azar has had a long run as a Trump appointee. He took office in January 2018 and has been a staunch loyalist. But he’s frequently been the butt of grousing by Trump for not doing enough to help lower drug prices and for his handling of the coronavirus pandemic. Azar was initially in charge of the Trump virus effort but was quickly replaced by Vice President Mike Pence.
It was widely reported in late April that Trump was considering firing Azar, but the president called that “fake news” in a tweet.
Azar has complained about Hahn, who was confirmed in December 2019. According to Politico, Azar was looking into how to remove Hahn as commissioner because of the FDA’s battle with the White House over standards for emergency use authorization of a coronavirus vaccine.
In addition, Trump was infuriated by the agency’s insistence that it stick to the highest bar for an emergency approval. “The deep state, or whoever, over at the FDA is making it very difficult for drug companies to get people in order to test the vaccines and therapeutics. Obviously, they are hoping to delay the answer until after November 3rd,” Trump tweeted at Hahn.
Fauci on the firing line?
Most of the president’s ire has been directed at Fauci. As far back as April, Trump retweeted a call for Fauci’s firing. Twitter removed the original tweet but kept Trump’s comments on the original tweet.
The president has frequently questioned Fauci’s advice, sidelined him from task force meetings, and infrequently met with him. Trump called Fauci a “disaster” during a call with supporters in October, and then, at a campaign rally in November, intimated that he would fire the scientist after the election, according to The Washington Post.
But such a firing cannot be easily done. Some have speculated that Trump could pressure Fauci’s boss, Francis Collins, MD, PhD — the director of the National Institutes of Health (NIH), who is a political appointee — to get rid of him. But Collins would have to come up with a reason to fire Fauci. Because he is not a political appointee, Fauci is afforded a raft of protections given to civil service employees of the federal government.
To demote or fire Fauci, Collins would have to give him 30 days’ notice unless there’s a belief that he committed a crime. Fauci would have at least a week to offer evidence and affidavits in support of his service.
He’d also be entitled to legal representation, a written decision, and the specific reasons for the action being taken quickly. He could also request a hearing, and he’d be able to appeal any action to the Merit Systems Protection Board. The process could take months, if not years.
In late October, Trump issued an executive order that would reclassify certain federal employees so that they wouldn’t have such protections. But agencies have until mid-January to come up with lists of such workers, according to Government Executive.
Collins has been with NIH since 1993, when he headed the Human Genome Project and the National Human Genome Research Institute. Politico has speculated that Collins, 70, might retire if Trump was reelected. It’s unclear what he’ll do now.
Redfield, who has taken heat for his leadership from many in public health — and was asked in October to stand up to Trump by former CDC Director William H. Foege, MD — has been openly contradicted by the president on more than one occasion, according to The New York Times.
In September, The Hill reported that Trump told reporters that he’d chastised Redfield by phone soon after Redfield had told a Senate committee that a coronavirus vaccine would not be available until mid-2021.
This article first appeared on Medscape.com.
Others may soon depart voluntarily. Politico reported in late October that more than two dozen political appointees had already left the U.S. Department Health and Human Services (HHS) since the start of the COVID-19 pandemic in February and that potentially dozens of the more than 100 in the department would leave if Trump was not reelected.
Trump hasn’t conceded, he is challenging the election results, and he has already fired his Defense Secretary, Mark Esper.
Among those possibly in Trump’s sights: HHS Secretary Alex Azar, US Food and Drug Administration (FDA) Commissioner Stephen Hahn, MD, Centers for Disease Control and Prevention (CDC) Director Robert Redfield, MD, and White House Coronavirus Task Force member Anthony Fauci, MD, who is also the director of the National Institutes of Allergy and Infectious Diseases.
Seema Verma, the administrator of the Centers for Medicare & Medicaid Services (CMS), is likely safe. According to Politico, Verma is expected to leave on her own terms.
Azar has had a long run as a Trump appointee. He took office in January 2018 and has been a staunch loyalist. But he’s frequently been the butt of grousing by Trump for not doing enough to help lower drug prices and for his handling of the coronavirus pandemic. Azar was initially in charge of the Trump virus effort but was quickly replaced by Vice President Mike Pence.
It was widely reported in late April that Trump was considering firing Azar, but the president called that “fake news” in a tweet.
Azar has complained about Hahn, who was confirmed in December 2019. According to Politico, Azar was looking into how to remove Hahn as commissioner because of the FDA’s battle with the White House over standards for emergency use authorization of a coronavirus vaccine.
In addition, Trump was infuriated by the agency’s insistence that it stick to the highest bar for an emergency approval. “The deep state, or whoever, over at the FDA is making it very difficult for drug companies to get people in order to test the vaccines and therapeutics. Obviously, they are hoping to delay the answer until after November 3rd,” Trump tweeted at Hahn.
Fauci on the firing line?
Most of the president’s ire has been directed at Fauci. As far back as April, Trump retweeted a call for Fauci’s firing. Twitter removed the original tweet but kept Trump’s comments on the original tweet.
The president has frequently questioned Fauci’s advice, sidelined him from task force meetings, and infrequently met with him. Trump called Fauci a “disaster” during a call with supporters in October, and then, at a campaign rally in November, intimated that he would fire the scientist after the election, according to The Washington Post.
But such a firing cannot be easily done. Some have speculated that Trump could pressure Fauci’s boss, Francis Collins, MD, PhD — the director of the National Institutes of Health (NIH), who is a political appointee — to get rid of him. But Collins would have to come up with a reason to fire Fauci. Because he is not a political appointee, Fauci is afforded a raft of protections given to civil service employees of the federal government.
To demote or fire Fauci, Collins would have to give him 30 days’ notice unless there’s a belief that he committed a crime. Fauci would have at least a week to offer evidence and affidavits in support of his service.
He’d also be entitled to legal representation, a written decision, and the specific reasons for the action being taken quickly. He could also request a hearing, and he’d be able to appeal any action to the Merit Systems Protection Board. The process could take months, if not years.
In late October, Trump issued an executive order that would reclassify certain federal employees so that they wouldn’t have such protections. But agencies have until mid-January to come up with lists of such workers, according to Government Executive.
Collins has been with NIH since 1993, when he headed the Human Genome Project and the National Human Genome Research Institute. Politico has speculated that Collins, 70, might retire if Trump was reelected. It’s unclear what he’ll do now.
Redfield, who has taken heat for his leadership from many in public health — and was asked in October to stand up to Trump by former CDC Director William H. Foege, MD — has been openly contradicted by the president on more than one occasion, according to The New York Times.
In September, The Hill reported that Trump told reporters that he’d chastised Redfield by phone soon after Redfield had told a Senate committee that a coronavirus vaccine would not be available until mid-2021.
This article first appeared on Medscape.com.
What to know as ACA heads to Supreme Court – again
The case, California v. Texas, is the result of a change to the health law made by Congress in 2017. As part of a major tax bill, Congress reduced to zero the penalty for not having health insurance. But it was that penalty – a tax – that the high court ruled made the law constitutional in a 2012 decision, argues a group of Republican state attorneys general. Without the tax, they say in their suit, the rest of the law must fall, too.
After originally contending that the entire law should not be struck down when the suit was filed in 2018, the Trump administration changed course in 2019 and joined the GOP officials who brought the case.
Here are some key questions and answers about the case.
What are the possibilities for how the court could rule?
There is a long list of ways this could play out.
The justices could declare the entire law unconstitutional – which is what a federal district judge in Texas ruled in December 2018. But legal experts say that’s not the most likely outcome of this case.
First, the court may avoid deciding the case on its merits entirely by ruling that the plaintiffs do not have “standing” to sue. The central issue in the case is whether the requirement in the law to have insurance – which remains even though Congress eliminated the penalty or tax – is constitutional. But states are not subject to the so-called individual mandate, so some analysts suggest the Republican officials have no standing. In addition, questions have been raised about the individual plaintiffs in the case, two consultants from Texas who argue that they felt compelled to buy insurance even without a possible penalty.
The court could also rule that, by eliminating the penalty but not the rest of the mandate (which Congress could not do in that 2017 tax bill for procedural reasons), lawmakers “didn’t mean to coerce anyone to do anything, and so there’s no constitutional problem,” University of Michigan law professor Nicholas Bagley said in a recent webinar for the NIHCM Foundation, the Commonwealth Fund, and the University of Southern California’s Center for Health Journalism.
Or, said Bagley, the court could rule that, without the tax, the requirement to have health insurance is unconstitutional, but the rest of the law is not. In that case, the justices might strike the mandate only, which would have basically no impact.
It gets more complicated if the court decides that, as the plaintiffs argue, the individual mandate language without the penalty is unconstitutional and so closely tied to other parts of the law that some of them must fall as well.
Even there the court has choices. One option would be, as the Trump administration originally argued, to strike down the mandate and just the pieces of the law most closely related to it – which happen to include the insurance protections for people with preexisting conditions, an extremely popular provision of the law. The two parts are connected because the original purpose of the mandate was to make sure enough healthy people sign up for insurance to offset the added costs to insurers of sicker people.
Another option, of course, would be for the court to follow the lead of the Texas judge and strike down the entire law.
While that’s not the most likely outcome, said Bagley, if it happens it could be “a hot mess” for the nation’s entire health care system. As just one example, he said, “every hospital is getting paid pursuant to changes made by the ACA. How do you even go about making payments if the thing that you are looking to guide what those payments ought to be is itself invalid?”
What impact will new Justice Amy Coney Barrett have?
Perhaps a lot. Before the death of Justice Ruth Bader Ginsburg, most court observers thought the case was highly unlikely to result in the entire law being struck down. That’s because Chief Justice John Roberts voted to uphold the law in 2012, and again when it was challenged in a less sweeping way in 2015.
But with Barrett replacing Ginsburg, even if Roberts joined the court’s remaining three liberals they could still be outvoted by the other five conservatives. Barrett was coy about her views on the Affordable Care Act during her confirmation hearings in October, but she has written that she thinks Roberts was wrong to uphold the law in 2012.
Could a new president and Congress make the case go away?
Many have suggested that, if Joe Biden assumes the presidency, his Justice Department could simply drop the case. But the administration did not bring the case; the GOP state officials did. And while normally the Justice Department’s job is to defend existing laws in court, in this case the ACA is being defended by a group of Democratic state attorneys general. A new administration could change that position, but that’s not the same as dropping the case.
Congress, on the other hand, could easily make the case moot. It could add back even a nominal financial penalty for not having insurance. It could eliminate the mandate altogether, although that would require 60 votes in the Senate under current rules. Congress could also pass a “severability” provision saying that, if any portion of the law is struck down, the rest should remain.
“The problem is not technical,” said Bagley. “It’s political.”
What is the timeline for a decision? Could the court delay implementation of its ruling?
The court usually hears oral arguments in a case months before it issues a decision. Unless the decision is unanimous or turns out to be very simple, Bagley said, he would expect to see an opinion “sometime in the spring.”
As to whether the court could find some or all of the law unconstitutional but delay when its decision takes effect, Bagley said that happened from time to time as recently as the 1970s. “That practice has been more or less abandoned,” he said, but in the case of a law so large, “you could imagine the Supreme Court using its discretion to say the decision wouldn’t take effect immediately.”
If the court does invalidate the entire ACA, Congress could act to fix things, but it’s unclear if it will be able to, especially if Republicans still control the Senate. If the justices strike the law, Bagley said, “I honestly think the likeliest outcome is that Congress runs around like a chicken with its head cut off, doesn’t come to a deal, and we’re back to where we were before 2010” when the ACA passed.
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of KFF (Kaiser Family Foundation), which is not affiliated with Kaiser Permanente.
The case, California v. Texas, is the result of a change to the health law made by Congress in 2017. As part of a major tax bill, Congress reduced to zero the penalty for not having health insurance. But it was that penalty – a tax – that the high court ruled made the law constitutional in a 2012 decision, argues a group of Republican state attorneys general. Without the tax, they say in their suit, the rest of the law must fall, too.
After originally contending that the entire law should not be struck down when the suit was filed in 2018, the Trump administration changed course in 2019 and joined the GOP officials who brought the case.
Here are some key questions and answers about the case.
What are the possibilities for how the court could rule?
There is a long list of ways this could play out.
The justices could declare the entire law unconstitutional – which is what a federal district judge in Texas ruled in December 2018. But legal experts say that’s not the most likely outcome of this case.
First, the court may avoid deciding the case on its merits entirely by ruling that the plaintiffs do not have “standing” to sue. The central issue in the case is whether the requirement in the law to have insurance – which remains even though Congress eliminated the penalty or tax – is constitutional. But states are not subject to the so-called individual mandate, so some analysts suggest the Republican officials have no standing. In addition, questions have been raised about the individual plaintiffs in the case, two consultants from Texas who argue that they felt compelled to buy insurance even without a possible penalty.
The court could also rule that, by eliminating the penalty but not the rest of the mandate (which Congress could not do in that 2017 tax bill for procedural reasons), lawmakers “didn’t mean to coerce anyone to do anything, and so there’s no constitutional problem,” University of Michigan law professor Nicholas Bagley said in a recent webinar for the NIHCM Foundation, the Commonwealth Fund, and the University of Southern California’s Center for Health Journalism.
Or, said Bagley, the court could rule that, without the tax, the requirement to have health insurance is unconstitutional, but the rest of the law is not. In that case, the justices might strike the mandate only, which would have basically no impact.
It gets more complicated if the court decides that, as the plaintiffs argue, the individual mandate language without the penalty is unconstitutional and so closely tied to other parts of the law that some of them must fall as well.
Even there the court has choices. One option would be, as the Trump administration originally argued, to strike down the mandate and just the pieces of the law most closely related to it – which happen to include the insurance protections for people with preexisting conditions, an extremely popular provision of the law. The two parts are connected because the original purpose of the mandate was to make sure enough healthy people sign up for insurance to offset the added costs to insurers of sicker people.
Another option, of course, would be for the court to follow the lead of the Texas judge and strike down the entire law.
While that’s not the most likely outcome, said Bagley, if it happens it could be “a hot mess” for the nation’s entire health care system. As just one example, he said, “every hospital is getting paid pursuant to changes made by the ACA. How do you even go about making payments if the thing that you are looking to guide what those payments ought to be is itself invalid?”
What impact will new Justice Amy Coney Barrett have?
Perhaps a lot. Before the death of Justice Ruth Bader Ginsburg, most court observers thought the case was highly unlikely to result in the entire law being struck down. That’s because Chief Justice John Roberts voted to uphold the law in 2012, and again when it was challenged in a less sweeping way in 2015.
But with Barrett replacing Ginsburg, even if Roberts joined the court’s remaining three liberals they could still be outvoted by the other five conservatives. Barrett was coy about her views on the Affordable Care Act during her confirmation hearings in October, but she has written that she thinks Roberts was wrong to uphold the law in 2012.
Could a new president and Congress make the case go away?
Many have suggested that, if Joe Biden assumes the presidency, his Justice Department could simply drop the case. But the administration did not bring the case; the GOP state officials did. And while normally the Justice Department’s job is to defend existing laws in court, in this case the ACA is being defended by a group of Democratic state attorneys general. A new administration could change that position, but that’s not the same as dropping the case.
Congress, on the other hand, could easily make the case moot. It could add back even a nominal financial penalty for not having insurance. It could eliminate the mandate altogether, although that would require 60 votes in the Senate under current rules. Congress could also pass a “severability” provision saying that, if any portion of the law is struck down, the rest should remain.
“The problem is not technical,” said Bagley. “It’s political.”
What is the timeline for a decision? Could the court delay implementation of its ruling?
The court usually hears oral arguments in a case months before it issues a decision. Unless the decision is unanimous or turns out to be very simple, Bagley said, he would expect to see an opinion “sometime in the spring.”
As to whether the court could find some or all of the law unconstitutional but delay when its decision takes effect, Bagley said that happened from time to time as recently as the 1970s. “That practice has been more or less abandoned,” he said, but in the case of a law so large, “you could imagine the Supreme Court using its discretion to say the decision wouldn’t take effect immediately.”
If the court does invalidate the entire ACA, Congress could act to fix things, but it’s unclear if it will be able to, especially if Republicans still control the Senate. If the justices strike the law, Bagley said, “I honestly think the likeliest outcome is that Congress runs around like a chicken with its head cut off, doesn’t come to a deal, and we’re back to where we were before 2010” when the ACA passed.
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of KFF (Kaiser Family Foundation), which is not affiliated with Kaiser Permanente.
The case, California v. Texas, is the result of a change to the health law made by Congress in 2017. As part of a major tax bill, Congress reduced to zero the penalty for not having health insurance. But it was that penalty – a tax – that the high court ruled made the law constitutional in a 2012 decision, argues a group of Republican state attorneys general. Without the tax, they say in their suit, the rest of the law must fall, too.
After originally contending that the entire law should not be struck down when the suit was filed in 2018, the Trump administration changed course in 2019 and joined the GOP officials who brought the case.
Here are some key questions and answers about the case.
What are the possibilities for how the court could rule?
There is a long list of ways this could play out.
The justices could declare the entire law unconstitutional – which is what a federal district judge in Texas ruled in December 2018. But legal experts say that’s not the most likely outcome of this case.
First, the court may avoid deciding the case on its merits entirely by ruling that the plaintiffs do not have “standing” to sue. The central issue in the case is whether the requirement in the law to have insurance – which remains even though Congress eliminated the penalty or tax – is constitutional. But states are not subject to the so-called individual mandate, so some analysts suggest the Republican officials have no standing. In addition, questions have been raised about the individual plaintiffs in the case, two consultants from Texas who argue that they felt compelled to buy insurance even without a possible penalty.
The court could also rule that, by eliminating the penalty but not the rest of the mandate (which Congress could not do in that 2017 tax bill for procedural reasons), lawmakers “didn’t mean to coerce anyone to do anything, and so there’s no constitutional problem,” University of Michigan law professor Nicholas Bagley said in a recent webinar for the NIHCM Foundation, the Commonwealth Fund, and the University of Southern California’s Center for Health Journalism.
Or, said Bagley, the court could rule that, without the tax, the requirement to have health insurance is unconstitutional, but the rest of the law is not. In that case, the justices might strike the mandate only, which would have basically no impact.
It gets more complicated if the court decides that, as the plaintiffs argue, the individual mandate language without the penalty is unconstitutional and so closely tied to other parts of the law that some of them must fall as well.
Even there the court has choices. One option would be, as the Trump administration originally argued, to strike down the mandate and just the pieces of the law most closely related to it – which happen to include the insurance protections for people with preexisting conditions, an extremely popular provision of the law. The two parts are connected because the original purpose of the mandate was to make sure enough healthy people sign up for insurance to offset the added costs to insurers of sicker people.
Another option, of course, would be for the court to follow the lead of the Texas judge and strike down the entire law.
While that’s not the most likely outcome, said Bagley, if it happens it could be “a hot mess” for the nation’s entire health care system. As just one example, he said, “every hospital is getting paid pursuant to changes made by the ACA. How do you even go about making payments if the thing that you are looking to guide what those payments ought to be is itself invalid?”
What impact will new Justice Amy Coney Barrett have?
Perhaps a lot. Before the death of Justice Ruth Bader Ginsburg, most court observers thought the case was highly unlikely to result in the entire law being struck down. That’s because Chief Justice John Roberts voted to uphold the law in 2012, and again when it was challenged in a less sweeping way in 2015.
But with Barrett replacing Ginsburg, even if Roberts joined the court’s remaining three liberals they could still be outvoted by the other five conservatives. Barrett was coy about her views on the Affordable Care Act during her confirmation hearings in October, but she has written that she thinks Roberts was wrong to uphold the law in 2012.
Could a new president and Congress make the case go away?
Many have suggested that, if Joe Biden assumes the presidency, his Justice Department could simply drop the case. But the administration did not bring the case; the GOP state officials did. And while normally the Justice Department’s job is to defend existing laws in court, in this case the ACA is being defended by a group of Democratic state attorneys general. A new administration could change that position, but that’s not the same as dropping the case.
Congress, on the other hand, could easily make the case moot. It could add back even a nominal financial penalty for not having insurance. It could eliminate the mandate altogether, although that would require 60 votes in the Senate under current rules. Congress could also pass a “severability” provision saying that, if any portion of the law is struck down, the rest should remain.
“The problem is not technical,” said Bagley. “It’s political.”
What is the timeline for a decision? Could the court delay implementation of its ruling?
The court usually hears oral arguments in a case months before it issues a decision. Unless the decision is unanimous or turns out to be very simple, Bagley said, he would expect to see an opinion “sometime in the spring.”
As to whether the court could find some or all of the law unconstitutional but delay when its decision takes effect, Bagley said that happened from time to time as recently as the 1970s. “That practice has been more or less abandoned,” he said, but in the case of a law so large, “you could imagine the Supreme Court using its discretion to say the decision wouldn’t take effect immediately.”
If the court does invalidate the entire ACA, Congress could act to fix things, but it’s unclear if it will be able to, especially if Republicans still control the Senate. If the justices strike the law, Bagley said, “I honestly think the likeliest outcome is that Congress runs around like a chicken with its head cut off, doesn’t come to a deal, and we’re back to where we were before 2010” when the ACA passed.
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of KFF (Kaiser Family Foundation), which is not affiliated with Kaiser Permanente.
Biden victory: What it means for COVID, health care
The former vice president has sketched out a big health agenda: ramping up the federal response to COVID-19, boosting the Affordable Care Act, creating a new “public option” to cover uninsured Americans, and expanding Medicare and Medicaid.
But the president-elect’s long to-do list on health is likely to face significant roadblocks in Congress and the courts, experts say.
For instance, Biden’s ambitious proposals on COVID-19 -- including his recent call for a national mask mandate -- could be waylaid by legal challenges and run into political hurdles on Capitol Hill, where he may face a divided Congress.
Joseph Antos, PhD, a health policy expert with the conservative American Enterprise Institute, predicts Biden will encounter the same type of congressional “gridlock situation” that President Barack Obama ran into during his second term.
“We have a situation that has been like this for a very, very long time -- lack of cooperation, lack of recognition that either party is capable of rising above their own electoral views to deal with problems that the country actually has.”
Antos also suggests that Biden may also face enormous political pressure to address the economic fallout from the coronavirus, including record unemployment and business closures, before anything else.
“I think it’s really going to be efforts that are intended to promote economic development and promote the economy,” he says.
In addition, Biden’s plans to expand Obamacare might face a new challenge from the Supreme Court in the year ahead. This month, the high court will take up a new case seeking to overturn the law.
Even so, experts say Biden’s plans on COVID-19 and expanding health care are likely to define his tenure in the White House as a central focus of his presidency.
“Health care will be at the very top of the list of the president’s priorities,” says Sabrina Corlette, JD, co-director of the Center on Health Insurance Reforms at Georgetown University’s McCourt School of Public Policy. “I do think, however, that the administration is going to be very preoccupied with the response to COVID-19 and the economic fallout … particularly in the first year.”
Here’s a closer look at what we can expect from a Biden presidency.
COVID-19: Federalizing response efforts
Biden will move to federalize the response to COVID-19. He has said he will take back major responsibilities from the states -- such as setting national policies on mask wearing, social distancing, and the reopening of schools and businesses, based on CDC guidance. In the days leading up to the election, Biden called for a national mask mandate, after waffling on the issue throughout the summer.
He has said he will let public health science drive political policy. Biden is also planning to create his own task force to advise officials during the transition on managing the new surge in COVID-19 cases, vaccine safety and protecting at-risk populations, Politico reported this week. He received a virtual briefing on the pandemic from a panel of experts as he awaited the election’s outcome.
“I think we will no longer have this confused and contradictory public messaging,” Corlette says, “but I also think there will be humility and the recognition that the evidence is evolving -- that we don’t have all the answers, but we’re learning as we go.”
But national mandates on masks and social distancing will be challenging to enforce, experts say. They are also likely to face pushback from business interests, opposition from public officials in GOP-led states, and even legal challenges.
Biden’s ability to work with Congress -- or not -- may determine whether he is able to implement some of the key components of his coronavirus action plan, which includes:
- Providing free COVID-19 testing for all Americans
- Hiring 100,000 contact tracers
- Eliminating out-of-pocket expenses for coronavirus treatment
- Delivering “sufficient” PPE for essential workers
- Supporting science-backed vaccines and medical treatments being developed
- Requiring the reopening of businesses, workplaces, and schools only after “sufficient” reductions in community transmission -- under evidence-based protocols put forward by the CDC
- Giving emergency paid leave for workers dislocated by the pandemic and more financial aid for workers, families, and small businesses
- Shoring up safeguards to protect at-risk Americans, including older people
- Boosting pay for health care workers on the front lines
Biden has not detailed how he would pay for many of these, beyond promising to force wealthy Americans to “pay their fair share” of taxes to help. He has proposed a tax increase on Americans making more than $400,000 a year, which would require congressional approval.
Antos says he expects Biden’s proposed COVID-19 action plan to be virtually the same as Trump’s in two areas: efforts to develop a vaccine and antiviral treatments.
The administration has spent some $225 million on COVID-19 testing efforts, with a particular focus on rural areas.
Trump launched Operation Warp Speed to fast-track a vaccine. As part of that, the federal government has contracted with six drug companies, spending nearly $11 billion. The operation aims to provide at least 300 million doses of a coronavirus vaccine by January 2021.
Antos would like to see “a more sophisticated approach to social distancing” from the president-elect that takes into account the different challenges facing Americans depending on their income, work situation, and other factors during the pandemic.
“There are a lot of people in this country where working from home is fine and their jobs are secure,” he notes. “It’s the person who used to work at a restaurant that closed, it’s the line worker at a factory that has severely cut back its hours. It’s basically lower-middle-class people, low-income people, middle-class people, and it’s not the elite.
“And the policies have not given enough consideration to the fact that their circumstances and their tradeoffs would differ from the tradeoffs of somebody who doesn’t have anything to worry about economically.
“So, what we need is a more supple policy [that] will give people the information they need and give them the financial support that they also need … so they can make good decisions for themselves and their families. And we basically haven’t done that.”
Obamacare on the blocks?
The Supreme Court’s decision to take up another case seeking to overturn the Affordable Care Act could hand Biden’s health agenda a major setback -- and put the medical care for millions of Americans in jeopardy.
On Nov. 10, the high court will hear oral arguments on a lawsuit that would strike down all of Obamacare. A decision is not expected until next year.
The court has previously upheld the 2010 law, which Biden helped usher through Congress as vice president. But the addition of right-leaning Supreme Court Justice Amy Coney Barrett to the bench last month gives the court a clear conservative majority that could mean the end of Obamacare, legal experts say.
Republicans have opposed the law since its passage, but they have been unable to muster the votes to repeal it, or to pass an alternative
Antos, from the American Enterprise Institute, notes conservatives believe the law has increased costs for health care and insurance over the past decade, in part because of its protections for Americans with preexisting conditions and requiring insurers to provide comprehensive “gold-plated” policies.
“It’s driven up costs, offers plans that are not very strong, put high-risk folks into the same [insurance pool], which has increased costs for everyone, the employer mandate … these are all the reasons,” he says.
The Supreme Court isn’t expected to deliver a decision on the Affordable Care Act before the middle of next year. But the uncertainty will likely push back Biden’s proposals to expand on the law.
Overturning Obamacare would have huge impacts on millions of Americans:
- As many as 133 million Americans -- roughly half the U.S. population -- with preexisting conditions could find it harder, if not impossible, to find affordable health insurance. That figure does not include Americans infected with COVID-19.
- About 165 million who require expensive treatments -- for cancer and other conditions -- would no longer be protected from huge costs for care by federal caps on out-of-pocket expenditures the Affordable Care Act requires.
- An estimated 21 million who now buy insurance through the Obamacare Marketplaces could lose their coverage.
- Another 12 million on Medicaid could find themselves without insurance.
- At least 2 million young adults ages 26 and under, now on their parents’ health policies, could be kicked off.
- Millions of people who use Medicare could face higher costs.
- Federal subsidies for lower-income Americans to buy policies would disappear.
Throughout the campaign, Biden repeatedly stressed the need to preserve the law’s provision barring insurance companies from refusing coverage for Americans with preexisting conditions, such as diabetes, cancer, and heart disease. It also outlaws charging higher premiums on the basis of health status, age, or gender.
Biden has also pledged to bolster the law as president.
He has proposed a variety of add-ons to the Affordable Care Act he says will “insure more than an estimated 97% of Americans,” according to the Biden campaign site.
Biden’s proposals include offering larger federal subsidies to help low- and middle-income Americans pay for policies purchased through Obamacare insurance Marketplaces.
The boldest of Biden’s proposals is the creation of a “public option” for insurance -- a Medicare-like program that small businesses and individuals could choose if they do not have coverage, cannot afford it, or don’t like their employer-based coverage.
It would also automatically enroll millions of uninsured Americans living in the 14 states that have not expanded Medicaid, which covers low-income people.
But such a plan would require congressional approval -- including a “super majority” of 60 Senate votes to block a likely GOP filibuster. That will be a significant challenge Biden will have to overcome, with Congress so evenly divided.
The White House would also have to defeat heavy lobbying from some of the most influential industry interest groups in Washington, Corlette says.
“I’m not even confident they would get all the Democrat votes,” she says.
“So, it’s a going to be an uphill battle to get a public option passed.”
Taken together, Biden’s plans for expanding Obamacare are projected to cost $750 billion over 10 years. He has said much of that financing would come from increasing taxes on the wealthy.
That means it would likely require congressional approval, which Antos suggests is unlikely given the polarization on Capitol Hill.
Medicare, Medicaid, and drug costs
Biden has called for a host of reforms targeting Medicare, Medicaid, and rising drug costs.
On Medicare, which primarily covers seniors 65 and older, Biden has proposed lowering the eligibility age from 65 to 60. That could extend Medicare to up to 20 million more Americans.
On Medicaid, the health care safety net for low-income and disabled Americans, the president-elect supports increased federal funding to states during the current economic crisis, and potentially beyond.
Medicare is likely to become a key focus of the new administration, in light of the pressures the pandemic is placing on Medicare funding.
In April, Medicare’s trustees said that the Part A trust fund for the program, which pays for hospital and inpatient care, could start to run dry in 2026.
But those projections did not include the impact of COVID-19. Some economists have since projected that Medicare Part A could become insolvent as early as 2022.
Medicare Part B, which pays for doctor and outpatient costs, is funded by general tax funding and beneficiary insurance premiums, so it is not in danger of drying up.
Adding to those pressures is an executive order Trump signed in August temporarily deferring payroll taxes, a primary funding vehicle for Medicare and Social Security.
Under these taxes, employees pay 6.2% of their earnings (on annual income up to $137,700) toward Social Security and 1.45% for Medicare taxes each pay period. Employers pay the same rate per paycheck, adding up to a combined 12.4% Social Security tax and 2.9% Medicare tax.
Biden has said he would reverse the tax cut when he takes office.
But to get a handle on Medicare and Medicaid funding issues, he is likely to need congressional support. Corlette and other experts say that could be a challenge while the nation remains in the grip of the coronavirus pandemic.
In addition to his Medicare and Medicaid reforms, Biden has proposed several plans to lower drug prices, a subset of rising health care and insurance costs.
U.S. spending on prescription drugs has increased nearly 42% over the past decade -- from $253.1 billion in 2010 to $358.7 billion in 2020 (projected) -- according to the Centers for Medicare & Medicaid Services.
In 2020, retail prices for 460 commonly prescribed drugs have spiked an average of 5.2%, according to new analysis by 3 Axis Advisors, a health research firm.
That’s more than double the projected rate of inflation.
To control drug costs, Biden supports legislation approved by the Democratic-led House of Representatives last year that would empower Medicare to negotiate drug prices with drug companies, as private insurers do.
Federal law now bars Medicare from negotiating prices on behalf of the 67.7 million Americans who use it. Drug companies and many GOP leaders argue that the current law is necessary to allow them to spend more on research and development of new medications.
In addition, Biden supports the idea of lifting bans on importing drugs from foreign countries with lower costs.
He also backs creating an independent review board to set price caps for new medications with no competitors; making high-quality generics more available; ending tax breaks for drug company advertising; and limiting their leeway in raising prices.
All of these proposals would likely require congressional approval and could face legal challenges in the courts.
This article first appeared on WebMD.com.
The former vice president has sketched out a big health agenda: ramping up the federal response to COVID-19, boosting the Affordable Care Act, creating a new “public option” to cover uninsured Americans, and expanding Medicare and Medicaid.
But the president-elect’s long to-do list on health is likely to face significant roadblocks in Congress and the courts, experts say.
For instance, Biden’s ambitious proposals on COVID-19 -- including his recent call for a national mask mandate -- could be waylaid by legal challenges and run into political hurdles on Capitol Hill, where he may face a divided Congress.
Joseph Antos, PhD, a health policy expert with the conservative American Enterprise Institute, predicts Biden will encounter the same type of congressional “gridlock situation” that President Barack Obama ran into during his second term.
“We have a situation that has been like this for a very, very long time -- lack of cooperation, lack of recognition that either party is capable of rising above their own electoral views to deal with problems that the country actually has.”
Antos also suggests that Biden may also face enormous political pressure to address the economic fallout from the coronavirus, including record unemployment and business closures, before anything else.
“I think it’s really going to be efforts that are intended to promote economic development and promote the economy,” he says.
In addition, Biden’s plans to expand Obamacare might face a new challenge from the Supreme Court in the year ahead. This month, the high court will take up a new case seeking to overturn the law.
Even so, experts say Biden’s plans on COVID-19 and expanding health care are likely to define his tenure in the White House as a central focus of his presidency.
“Health care will be at the very top of the list of the president’s priorities,” says Sabrina Corlette, JD, co-director of the Center on Health Insurance Reforms at Georgetown University’s McCourt School of Public Policy. “I do think, however, that the administration is going to be very preoccupied with the response to COVID-19 and the economic fallout … particularly in the first year.”
Here’s a closer look at what we can expect from a Biden presidency.
COVID-19: Federalizing response efforts
Biden will move to federalize the response to COVID-19. He has said he will take back major responsibilities from the states -- such as setting national policies on mask wearing, social distancing, and the reopening of schools and businesses, based on CDC guidance. In the days leading up to the election, Biden called for a national mask mandate, after waffling on the issue throughout the summer.
He has said he will let public health science drive political policy. Biden is also planning to create his own task force to advise officials during the transition on managing the new surge in COVID-19 cases, vaccine safety and protecting at-risk populations, Politico reported this week. He received a virtual briefing on the pandemic from a panel of experts as he awaited the election’s outcome.
“I think we will no longer have this confused and contradictory public messaging,” Corlette says, “but I also think there will be humility and the recognition that the evidence is evolving -- that we don’t have all the answers, but we’re learning as we go.”
But national mandates on masks and social distancing will be challenging to enforce, experts say. They are also likely to face pushback from business interests, opposition from public officials in GOP-led states, and even legal challenges.
Biden’s ability to work with Congress -- or not -- may determine whether he is able to implement some of the key components of his coronavirus action plan, which includes:
- Providing free COVID-19 testing for all Americans
- Hiring 100,000 contact tracers
- Eliminating out-of-pocket expenses for coronavirus treatment
- Delivering “sufficient” PPE for essential workers
- Supporting science-backed vaccines and medical treatments being developed
- Requiring the reopening of businesses, workplaces, and schools only after “sufficient” reductions in community transmission -- under evidence-based protocols put forward by the CDC
- Giving emergency paid leave for workers dislocated by the pandemic and more financial aid for workers, families, and small businesses
- Shoring up safeguards to protect at-risk Americans, including older people
- Boosting pay for health care workers on the front lines
Biden has not detailed how he would pay for many of these, beyond promising to force wealthy Americans to “pay their fair share” of taxes to help. He has proposed a tax increase on Americans making more than $400,000 a year, which would require congressional approval.
Antos says he expects Biden’s proposed COVID-19 action plan to be virtually the same as Trump’s in two areas: efforts to develop a vaccine and antiviral treatments.
The administration has spent some $225 million on COVID-19 testing efforts, with a particular focus on rural areas.
Trump launched Operation Warp Speed to fast-track a vaccine. As part of that, the federal government has contracted with six drug companies, spending nearly $11 billion. The operation aims to provide at least 300 million doses of a coronavirus vaccine by January 2021.
Antos would like to see “a more sophisticated approach to social distancing” from the president-elect that takes into account the different challenges facing Americans depending on their income, work situation, and other factors during the pandemic.
“There are a lot of people in this country where working from home is fine and their jobs are secure,” he notes. “It’s the person who used to work at a restaurant that closed, it’s the line worker at a factory that has severely cut back its hours. It’s basically lower-middle-class people, low-income people, middle-class people, and it’s not the elite.
“And the policies have not given enough consideration to the fact that their circumstances and their tradeoffs would differ from the tradeoffs of somebody who doesn’t have anything to worry about economically.
“So, what we need is a more supple policy [that] will give people the information they need and give them the financial support that they also need … so they can make good decisions for themselves and their families. And we basically haven’t done that.”
Obamacare on the blocks?
The Supreme Court’s decision to take up another case seeking to overturn the Affordable Care Act could hand Biden’s health agenda a major setback -- and put the medical care for millions of Americans in jeopardy.
On Nov. 10, the high court will hear oral arguments on a lawsuit that would strike down all of Obamacare. A decision is not expected until next year.
The court has previously upheld the 2010 law, which Biden helped usher through Congress as vice president. But the addition of right-leaning Supreme Court Justice Amy Coney Barrett to the bench last month gives the court a clear conservative majority that could mean the end of Obamacare, legal experts say.
Republicans have opposed the law since its passage, but they have been unable to muster the votes to repeal it, or to pass an alternative
Antos, from the American Enterprise Institute, notes conservatives believe the law has increased costs for health care and insurance over the past decade, in part because of its protections for Americans with preexisting conditions and requiring insurers to provide comprehensive “gold-plated” policies.
“It’s driven up costs, offers plans that are not very strong, put high-risk folks into the same [insurance pool], which has increased costs for everyone, the employer mandate … these are all the reasons,” he says.
The Supreme Court isn’t expected to deliver a decision on the Affordable Care Act before the middle of next year. But the uncertainty will likely push back Biden’s proposals to expand on the law.
Overturning Obamacare would have huge impacts on millions of Americans:
- As many as 133 million Americans -- roughly half the U.S. population -- with preexisting conditions could find it harder, if not impossible, to find affordable health insurance. That figure does not include Americans infected with COVID-19.
- About 165 million who require expensive treatments -- for cancer and other conditions -- would no longer be protected from huge costs for care by federal caps on out-of-pocket expenditures the Affordable Care Act requires.
- An estimated 21 million who now buy insurance through the Obamacare Marketplaces could lose their coverage.
- Another 12 million on Medicaid could find themselves without insurance.
- At least 2 million young adults ages 26 and under, now on their parents’ health policies, could be kicked off.
- Millions of people who use Medicare could face higher costs.
- Federal subsidies for lower-income Americans to buy policies would disappear.
Throughout the campaign, Biden repeatedly stressed the need to preserve the law’s provision barring insurance companies from refusing coverage for Americans with preexisting conditions, such as diabetes, cancer, and heart disease. It also outlaws charging higher premiums on the basis of health status, age, or gender.
Biden has also pledged to bolster the law as president.
He has proposed a variety of add-ons to the Affordable Care Act he says will “insure more than an estimated 97% of Americans,” according to the Biden campaign site.
Biden’s proposals include offering larger federal subsidies to help low- and middle-income Americans pay for policies purchased through Obamacare insurance Marketplaces.
The boldest of Biden’s proposals is the creation of a “public option” for insurance -- a Medicare-like program that small businesses and individuals could choose if they do not have coverage, cannot afford it, or don’t like their employer-based coverage.
It would also automatically enroll millions of uninsured Americans living in the 14 states that have not expanded Medicaid, which covers low-income people.
But such a plan would require congressional approval -- including a “super majority” of 60 Senate votes to block a likely GOP filibuster. That will be a significant challenge Biden will have to overcome, with Congress so evenly divided.
The White House would also have to defeat heavy lobbying from some of the most influential industry interest groups in Washington, Corlette says.
“I’m not even confident they would get all the Democrat votes,” she says.
“So, it’s a going to be an uphill battle to get a public option passed.”
Taken together, Biden’s plans for expanding Obamacare are projected to cost $750 billion over 10 years. He has said much of that financing would come from increasing taxes on the wealthy.
That means it would likely require congressional approval, which Antos suggests is unlikely given the polarization on Capitol Hill.
Medicare, Medicaid, and drug costs
Biden has called for a host of reforms targeting Medicare, Medicaid, and rising drug costs.
On Medicare, which primarily covers seniors 65 and older, Biden has proposed lowering the eligibility age from 65 to 60. That could extend Medicare to up to 20 million more Americans.
On Medicaid, the health care safety net for low-income and disabled Americans, the president-elect supports increased federal funding to states during the current economic crisis, and potentially beyond.
Medicare is likely to become a key focus of the new administration, in light of the pressures the pandemic is placing on Medicare funding.
In April, Medicare’s trustees said that the Part A trust fund for the program, which pays for hospital and inpatient care, could start to run dry in 2026.
But those projections did not include the impact of COVID-19. Some economists have since projected that Medicare Part A could become insolvent as early as 2022.
Medicare Part B, which pays for doctor and outpatient costs, is funded by general tax funding and beneficiary insurance premiums, so it is not in danger of drying up.
Adding to those pressures is an executive order Trump signed in August temporarily deferring payroll taxes, a primary funding vehicle for Medicare and Social Security.
Under these taxes, employees pay 6.2% of their earnings (on annual income up to $137,700) toward Social Security and 1.45% for Medicare taxes each pay period. Employers pay the same rate per paycheck, adding up to a combined 12.4% Social Security tax and 2.9% Medicare tax.
Biden has said he would reverse the tax cut when he takes office.
But to get a handle on Medicare and Medicaid funding issues, he is likely to need congressional support. Corlette and other experts say that could be a challenge while the nation remains in the grip of the coronavirus pandemic.
In addition to his Medicare and Medicaid reforms, Biden has proposed several plans to lower drug prices, a subset of rising health care and insurance costs.
U.S. spending on prescription drugs has increased nearly 42% over the past decade -- from $253.1 billion in 2010 to $358.7 billion in 2020 (projected) -- according to the Centers for Medicare & Medicaid Services.
In 2020, retail prices for 460 commonly prescribed drugs have spiked an average of 5.2%, according to new analysis by 3 Axis Advisors, a health research firm.
That’s more than double the projected rate of inflation.
To control drug costs, Biden supports legislation approved by the Democratic-led House of Representatives last year that would empower Medicare to negotiate drug prices with drug companies, as private insurers do.
Federal law now bars Medicare from negotiating prices on behalf of the 67.7 million Americans who use it. Drug companies and many GOP leaders argue that the current law is necessary to allow them to spend more on research and development of new medications.
In addition, Biden supports the idea of lifting bans on importing drugs from foreign countries with lower costs.
He also backs creating an independent review board to set price caps for new medications with no competitors; making high-quality generics more available; ending tax breaks for drug company advertising; and limiting their leeway in raising prices.
All of these proposals would likely require congressional approval and could face legal challenges in the courts.
This article first appeared on WebMD.com.
The former vice president has sketched out a big health agenda: ramping up the federal response to COVID-19, boosting the Affordable Care Act, creating a new “public option” to cover uninsured Americans, and expanding Medicare and Medicaid.
But the president-elect’s long to-do list on health is likely to face significant roadblocks in Congress and the courts, experts say.
For instance, Biden’s ambitious proposals on COVID-19 -- including his recent call for a national mask mandate -- could be waylaid by legal challenges and run into political hurdles on Capitol Hill, where he may face a divided Congress.
Joseph Antos, PhD, a health policy expert with the conservative American Enterprise Institute, predicts Biden will encounter the same type of congressional “gridlock situation” that President Barack Obama ran into during his second term.
“We have a situation that has been like this for a very, very long time -- lack of cooperation, lack of recognition that either party is capable of rising above their own electoral views to deal with problems that the country actually has.”
Antos also suggests that Biden may also face enormous political pressure to address the economic fallout from the coronavirus, including record unemployment and business closures, before anything else.
“I think it’s really going to be efforts that are intended to promote economic development and promote the economy,” he says.
In addition, Biden’s plans to expand Obamacare might face a new challenge from the Supreme Court in the year ahead. This month, the high court will take up a new case seeking to overturn the law.
Even so, experts say Biden’s plans on COVID-19 and expanding health care are likely to define his tenure in the White House as a central focus of his presidency.
“Health care will be at the very top of the list of the president’s priorities,” says Sabrina Corlette, JD, co-director of the Center on Health Insurance Reforms at Georgetown University’s McCourt School of Public Policy. “I do think, however, that the administration is going to be very preoccupied with the response to COVID-19 and the economic fallout … particularly in the first year.”
Here’s a closer look at what we can expect from a Biden presidency.
COVID-19: Federalizing response efforts
Biden will move to federalize the response to COVID-19. He has said he will take back major responsibilities from the states -- such as setting national policies on mask wearing, social distancing, and the reopening of schools and businesses, based on CDC guidance. In the days leading up to the election, Biden called for a national mask mandate, after waffling on the issue throughout the summer.
He has said he will let public health science drive political policy. Biden is also planning to create his own task force to advise officials during the transition on managing the new surge in COVID-19 cases, vaccine safety and protecting at-risk populations, Politico reported this week. He received a virtual briefing on the pandemic from a panel of experts as he awaited the election’s outcome.
“I think we will no longer have this confused and contradictory public messaging,” Corlette says, “but I also think there will be humility and the recognition that the evidence is evolving -- that we don’t have all the answers, but we’re learning as we go.”
But national mandates on masks and social distancing will be challenging to enforce, experts say. They are also likely to face pushback from business interests, opposition from public officials in GOP-led states, and even legal challenges.
Biden’s ability to work with Congress -- or not -- may determine whether he is able to implement some of the key components of his coronavirus action plan, which includes:
- Providing free COVID-19 testing for all Americans
- Hiring 100,000 contact tracers
- Eliminating out-of-pocket expenses for coronavirus treatment
- Delivering “sufficient” PPE for essential workers
- Supporting science-backed vaccines and medical treatments being developed
- Requiring the reopening of businesses, workplaces, and schools only after “sufficient” reductions in community transmission -- under evidence-based protocols put forward by the CDC
- Giving emergency paid leave for workers dislocated by the pandemic and more financial aid for workers, families, and small businesses
- Shoring up safeguards to protect at-risk Americans, including older people
- Boosting pay for health care workers on the front lines
Biden has not detailed how he would pay for many of these, beyond promising to force wealthy Americans to “pay their fair share” of taxes to help. He has proposed a tax increase on Americans making more than $400,000 a year, which would require congressional approval.
Antos says he expects Biden’s proposed COVID-19 action plan to be virtually the same as Trump’s in two areas: efforts to develop a vaccine and antiviral treatments.
The administration has spent some $225 million on COVID-19 testing efforts, with a particular focus on rural areas.
Trump launched Operation Warp Speed to fast-track a vaccine. As part of that, the federal government has contracted with six drug companies, spending nearly $11 billion. The operation aims to provide at least 300 million doses of a coronavirus vaccine by January 2021.
Antos would like to see “a more sophisticated approach to social distancing” from the president-elect that takes into account the different challenges facing Americans depending on their income, work situation, and other factors during the pandemic.
“There are a lot of people in this country where working from home is fine and their jobs are secure,” he notes. “It’s the person who used to work at a restaurant that closed, it’s the line worker at a factory that has severely cut back its hours. It’s basically lower-middle-class people, low-income people, middle-class people, and it’s not the elite.
“And the policies have not given enough consideration to the fact that their circumstances and their tradeoffs would differ from the tradeoffs of somebody who doesn’t have anything to worry about economically.
“So, what we need is a more supple policy [that] will give people the information they need and give them the financial support that they also need … so they can make good decisions for themselves and their families. And we basically haven’t done that.”
Obamacare on the blocks?
The Supreme Court’s decision to take up another case seeking to overturn the Affordable Care Act could hand Biden’s health agenda a major setback -- and put the medical care for millions of Americans in jeopardy.
On Nov. 10, the high court will hear oral arguments on a lawsuit that would strike down all of Obamacare. A decision is not expected until next year.
The court has previously upheld the 2010 law, which Biden helped usher through Congress as vice president. But the addition of right-leaning Supreme Court Justice Amy Coney Barrett to the bench last month gives the court a clear conservative majority that could mean the end of Obamacare, legal experts say.
Republicans have opposed the law since its passage, but they have been unable to muster the votes to repeal it, or to pass an alternative
Antos, from the American Enterprise Institute, notes conservatives believe the law has increased costs for health care and insurance over the past decade, in part because of its protections for Americans with preexisting conditions and requiring insurers to provide comprehensive “gold-plated” policies.
“It’s driven up costs, offers plans that are not very strong, put high-risk folks into the same [insurance pool], which has increased costs for everyone, the employer mandate … these are all the reasons,” he says.
The Supreme Court isn’t expected to deliver a decision on the Affordable Care Act before the middle of next year. But the uncertainty will likely push back Biden’s proposals to expand on the law.
Overturning Obamacare would have huge impacts on millions of Americans:
- As many as 133 million Americans -- roughly half the U.S. population -- with preexisting conditions could find it harder, if not impossible, to find affordable health insurance. That figure does not include Americans infected with COVID-19.
- About 165 million who require expensive treatments -- for cancer and other conditions -- would no longer be protected from huge costs for care by federal caps on out-of-pocket expenditures the Affordable Care Act requires.
- An estimated 21 million who now buy insurance through the Obamacare Marketplaces could lose their coverage.
- Another 12 million on Medicaid could find themselves without insurance.
- At least 2 million young adults ages 26 and under, now on their parents’ health policies, could be kicked off.
- Millions of people who use Medicare could face higher costs.
- Federal subsidies for lower-income Americans to buy policies would disappear.
Throughout the campaign, Biden repeatedly stressed the need to preserve the law’s provision barring insurance companies from refusing coverage for Americans with preexisting conditions, such as diabetes, cancer, and heart disease. It also outlaws charging higher premiums on the basis of health status, age, or gender.
Biden has also pledged to bolster the law as president.
He has proposed a variety of add-ons to the Affordable Care Act he says will “insure more than an estimated 97% of Americans,” according to the Biden campaign site.
Biden’s proposals include offering larger federal subsidies to help low- and middle-income Americans pay for policies purchased through Obamacare insurance Marketplaces.
The boldest of Biden’s proposals is the creation of a “public option” for insurance -- a Medicare-like program that small businesses and individuals could choose if they do not have coverage, cannot afford it, or don’t like their employer-based coverage.
It would also automatically enroll millions of uninsured Americans living in the 14 states that have not expanded Medicaid, which covers low-income people.
But such a plan would require congressional approval -- including a “super majority” of 60 Senate votes to block a likely GOP filibuster. That will be a significant challenge Biden will have to overcome, with Congress so evenly divided.
The White House would also have to defeat heavy lobbying from some of the most influential industry interest groups in Washington, Corlette says.
“I’m not even confident they would get all the Democrat votes,” she says.
“So, it’s a going to be an uphill battle to get a public option passed.”
Taken together, Biden’s plans for expanding Obamacare are projected to cost $750 billion over 10 years. He has said much of that financing would come from increasing taxes on the wealthy.
That means it would likely require congressional approval, which Antos suggests is unlikely given the polarization on Capitol Hill.
Medicare, Medicaid, and drug costs
Biden has called for a host of reforms targeting Medicare, Medicaid, and rising drug costs.
On Medicare, which primarily covers seniors 65 and older, Biden has proposed lowering the eligibility age from 65 to 60. That could extend Medicare to up to 20 million more Americans.
On Medicaid, the health care safety net for low-income and disabled Americans, the president-elect supports increased federal funding to states during the current economic crisis, and potentially beyond.
Medicare is likely to become a key focus of the new administration, in light of the pressures the pandemic is placing on Medicare funding.
In April, Medicare’s trustees said that the Part A trust fund for the program, which pays for hospital and inpatient care, could start to run dry in 2026.
But those projections did not include the impact of COVID-19. Some economists have since projected that Medicare Part A could become insolvent as early as 2022.
Medicare Part B, which pays for doctor and outpatient costs, is funded by general tax funding and beneficiary insurance premiums, so it is not in danger of drying up.
Adding to those pressures is an executive order Trump signed in August temporarily deferring payroll taxes, a primary funding vehicle for Medicare and Social Security.
Under these taxes, employees pay 6.2% of their earnings (on annual income up to $137,700) toward Social Security and 1.45% for Medicare taxes each pay period. Employers pay the same rate per paycheck, adding up to a combined 12.4% Social Security tax and 2.9% Medicare tax.
Biden has said he would reverse the tax cut when he takes office.
But to get a handle on Medicare and Medicaid funding issues, he is likely to need congressional support. Corlette and other experts say that could be a challenge while the nation remains in the grip of the coronavirus pandemic.
In addition to his Medicare and Medicaid reforms, Biden has proposed several plans to lower drug prices, a subset of rising health care and insurance costs.
U.S. spending on prescription drugs has increased nearly 42% over the past decade -- from $253.1 billion in 2010 to $358.7 billion in 2020 (projected) -- according to the Centers for Medicare & Medicaid Services.
In 2020, retail prices for 460 commonly prescribed drugs have spiked an average of 5.2%, according to new analysis by 3 Axis Advisors, a health research firm.
That’s more than double the projected rate of inflation.
To control drug costs, Biden supports legislation approved by the Democratic-led House of Representatives last year that would empower Medicare to negotiate drug prices with drug companies, as private insurers do.
Federal law now bars Medicare from negotiating prices on behalf of the 67.7 million Americans who use it. Drug companies and many GOP leaders argue that the current law is necessary to allow them to spend more on research and development of new medications.
In addition, Biden supports the idea of lifting bans on importing drugs from foreign countries with lower costs.
He also backs creating an independent review board to set price caps for new medications with no competitors; making high-quality generics more available; ending tax breaks for drug company advertising; and limiting their leeway in raising prices.
All of these proposals would likely require congressional approval and could face legal challenges in the courts.
This article first appeared on WebMD.com.
Bonds and Bridges: The Role of Social Capital in Building a More Diverse Dermatology Workforce
As our specialty seeks to address its lack of racial diversity, many dermatologists have answered recent calls to action.1,2 As we work toward dismantling systemic issues that have created pervasive inequality in our residency application review and interview processes, consideration also should be given to psychosocial issues that underrepresented-in-medicine (UIM) students face before their applications come to our attention. In this article, we explore how potential differences in the social capital of UIM and other disadvantaged dermatology residency applicants contribute to persistent homogeneity among dermatology training programs and the workforce.
The Theory of Capital
The concepts of economic, social, and cultural capital originate from the writings of social theorist Pierre Bourdieu.3 All 3 forms of capital are interconnected, and they relate to each other in ways that often facilitate social division and inequality. Economic capital denotes an individual’s economic resources or wealth, while cultural capital refers to the knowledge, behaviors, and skills that demonstrate his/her economic class (eg, communication style, table manners).3 Social capital refers to an individual’s interpersonal connections in personal and professional settings and can be subdivided into 3 categories: bonds, bridges, and linkages.4,5 Herein, we will focus on bonds and bridges.
It has been suggested that bonds are important for “getting by,” while bridges are critical for “getting ahead.”5 Bonds refer to close relationships within a community of people with shared characteristics, such as racial/ethnic identity and culture, access to information, and resources (eg, family, friends). These bonds provide trust, safety, and financial and emotional support; however, they are considered to be inward-looking and can promote exclusion and homogeneity.5
On the other hand, bridges refer to social relationships that extend outward beyond one’s close circle of family and friends to other people with shared interests and goals who may have different social or cultural identities (eg, professional colleagues). These bridges are considered to be outward-looking and provide many benefits to individuals and society. They link diverse individuals, which tends to increase tolerance and disrupt stereotypes, and they facilitate the sharing of ideas, information, and innovation. Additionally, bridges between individuals from different networks facilitate access to increased resources and opportunities for all parties.5
The 3 forms of capital are inextricably linked. For example, with economic capital, a child’s family can purchase access to a prestigious private high school, where he/she will gain valuable social capital through bridges with other students and their families. At this school, the child also will accumulate cultural capital that increases his/her sense of belonging in these circles. Subsequently, both the social and cultural capital accumulated at this private high school can be exchanged for economic capital via social networks, skills, values, and behaviors that facilitate entry into higher education and professional training. As such, these 3 forms of capital work together to continue social/class divisions, hierarchies, and ultimately inequality.
Impact of Social Capital in Pursuing a Medical Career
For medical students whose bonds (ie, close family, friends) include physicians or other health care professionals, the journey to studying medicine and entering their chosen specialty will be facilitated by financial security, valuable “inside information” about the application process, study skills, and even clinical guidance. Additionally, these students will have access to professional networks for mentorship, shadowing experiences, and other potential advantages. Furthermore, social capital is associated with higher self-esteem,6 which likely improves academic performance and wards off imposter syndrome in these students.
For medical students from lower socioeconomic status backgrounds or those whose inner circles do not include physicians or other health care professionals, accumulating the social and cultural capital needed to successfully navigate a medical career is more difficult. Although they may receive support and encouragement from family and friends, they will not have access to the same valuable information and connections that facilitate success; rather, they will have a further distance to travel, and this distance should be acknowledged in the residency application review process.
Acquiring Social Capital as a UIM Student
Despite the benefits of social and cultural capital, acquiring them takes a toll. For those UIM students who start life from a disadvantaged place, the accumulation of social capital does not come easily; rather, it demands effort and time that has the potential to detract from a student’s focus on the academic demands of medical education.7 Programs that attempt to improve disadvantaged students’ access to credible information, role models, and mentors can help lift some of the burden from the individual student’s shoulders. For example, studies have demonstrated the benefits of harnessing technology to enhance mentorship programs that increase social capital of disadvantaged populations.8-11 This approach already is in progress, bolstered by advances made in digital communications during the coronavirus disease 2019 pandemic.12 Student-led networking groups that connect remotely have been shown to build social capital bonds and bridges that facilitate collaborative learning, relationship building, and information sharing.8-11 There are existing online UIM student networks that individual dermatologists, institutions, and national organizations can partner with to facilitate the construction of bridges between these UIM student groups and dermatologists who can provide accurate, high-yield information and professional networking; however, one limitation of this suggestion is the disparate access to technology in the UIM community.
Final Thoughts
It is important to note that assumptions should not be made about the level of economic, social, or cultural capital an individual possesses based on his/her race or ethnicity. Instead, mentors should attempt to be available to a diverse pool of students; take the time to get to know these students; and then provide the types of mentorship, information, exposure, and networking that each individual student needs. Another approach is to make a concerted effort to ensure that all students receive the same amount and quality of information about medical education and our specialty regardless of their level of economic, cultural, or social capital. Moreover, beyond the promotion of diversity through increasing numbers of UIM applicants, we should seek to reshape our specialty into a space that does not require students to subdue their existing diverse forms of capital but rather to bring these different perspectives and lived experiences to the table.13
- Bray JK, McMichael AJ, Huang WW, et al. Publication rates on the topic of racial and ethnic diversity in dermatology versus other specialties. Dermatol Online J. 2020;26:7.
- Pritchett EN, Pandya AG, Ferguson NN, et al. Diversity in dermatology: roadmap for improvement. J Am Acad Dermatol. 2018;79:337-341.
- Bourdieu P. The forms of capital. In: Richardson J, ed. Handbook of Theory and Research for the Sociology of Education. Westport, CT: Greenwood; 1986:241-258.
- Granovetter MS. The strength of weak ties. Am J Sociol. 1973;78:1360-1380.
- Putnam RD. Bowling alone: America’s declining social capital. J Democracy. 1995;6:65-78.
- Han S. Longitudinal association between social capital and self-esteem: a matter of context. Psychiatry Research. 2015;226:340-346.
- Kirschling JM. Building social capital: leading and leveraging constituencies outside the college. J Nurs Educ. 2004;43:517-519.
- Radlick RL, Svedberg P, Nygren JM, et al. Digitally enhanced mentoring for immigrant youth social capital: protocol for a mixed methods pilot study and a randomized controlled trial [published online March 17, 2020]. JMIR Research Protocols. doi:10.2196/16472.
- Koh LC, Walker R, Wollersheim D, et al. I think someone is walking with me: the use of mobile phone for social capital development among women in four refugee communities. Int J Migration Health Social Care. 2018;14:411-424.
- Hartley A, Kassam AA. Social networking for learning in higher education: capitalising on social capital. ResearchGate website.https://www.researchgate.net/publication/311097860_Social_Networking_for_Learning_in_Higher_Education_Capitalising_on_Social_Capital. Published November 2016. Accessed October 19, 2020.
- Zalon ML. Using technology to build community in professional associations. J Contin Educ Nurs. 2008;39:235-240.
- Stewart CR, Chernoff KA, Wildman HF, et al. Recommendations for medical student preparedness and equity for dermatology residency applications during the COVID-19 pandemic. J Am Acad Dermatol. 2020;83:E225-E226.
- Brosnan C, Southgate E, Outram S, et al. Experiences of medical students who are first in family to attend university. Med Educ. 2016;50:842-851.
As our specialty seeks to address its lack of racial diversity, many dermatologists have answered recent calls to action.1,2 As we work toward dismantling systemic issues that have created pervasive inequality in our residency application review and interview processes, consideration also should be given to psychosocial issues that underrepresented-in-medicine (UIM) students face before their applications come to our attention. In this article, we explore how potential differences in the social capital of UIM and other disadvantaged dermatology residency applicants contribute to persistent homogeneity among dermatology training programs and the workforce.
The Theory of Capital
The concepts of economic, social, and cultural capital originate from the writings of social theorist Pierre Bourdieu.3 All 3 forms of capital are interconnected, and they relate to each other in ways that often facilitate social division and inequality. Economic capital denotes an individual’s economic resources or wealth, while cultural capital refers to the knowledge, behaviors, and skills that demonstrate his/her economic class (eg, communication style, table manners).3 Social capital refers to an individual’s interpersonal connections in personal and professional settings and can be subdivided into 3 categories: bonds, bridges, and linkages.4,5 Herein, we will focus on bonds and bridges.
It has been suggested that bonds are important for “getting by,” while bridges are critical for “getting ahead.”5 Bonds refer to close relationships within a community of people with shared characteristics, such as racial/ethnic identity and culture, access to information, and resources (eg, family, friends). These bonds provide trust, safety, and financial and emotional support; however, they are considered to be inward-looking and can promote exclusion and homogeneity.5
On the other hand, bridges refer to social relationships that extend outward beyond one’s close circle of family and friends to other people with shared interests and goals who may have different social or cultural identities (eg, professional colleagues). These bridges are considered to be outward-looking and provide many benefits to individuals and society. They link diverse individuals, which tends to increase tolerance and disrupt stereotypes, and they facilitate the sharing of ideas, information, and innovation. Additionally, bridges between individuals from different networks facilitate access to increased resources and opportunities for all parties.5
The 3 forms of capital are inextricably linked. For example, with economic capital, a child’s family can purchase access to a prestigious private high school, where he/she will gain valuable social capital through bridges with other students and their families. At this school, the child also will accumulate cultural capital that increases his/her sense of belonging in these circles. Subsequently, both the social and cultural capital accumulated at this private high school can be exchanged for economic capital via social networks, skills, values, and behaviors that facilitate entry into higher education and professional training. As such, these 3 forms of capital work together to continue social/class divisions, hierarchies, and ultimately inequality.
Impact of Social Capital in Pursuing a Medical Career
For medical students whose bonds (ie, close family, friends) include physicians or other health care professionals, the journey to studying medicine and entering their chosen specialty will be facilitated by financial security, valuable “inside information” about the application process, study skills, and even clinical guidance. Additionally, these students will have access to professional networks for mentorship, shadowing experiences, and other potential advantages. Furthermore, social capital is associated with higher self-esteem,6 which likely improves academic performance and wards off imposter syndrome in these students.
For medical students from lower socioeconomic status backgrounds or those whose inner circles do not include physicians or other health care professionals, accumulating the social and cultural capital needed to successfully navigate a medical career is more difficult. Although they may receive support and encouragement from family and friends, they will not have access to the same valuable information and connections that facilitate success; rather, they will have a further distance to travel, and this distance should be acknowledged in the residency application review process.
Acquiring Social Capital as a UIM Student
Despite the benefits of social and cultural capital, acquiring them takes a toll. For those UIM students who start life from a disadvantaged place, the accumulation of social capital does not come easily; rather, it demands effort and time that has the potential to detract from a student’s focus on the academic demands of medical education.7 Programs that attempt to improve disadvantaged students’ access to credible information, role models, and mentors can help lift some of the burden from the individual student’s shoulders. For example, studies have demonstrated the benefits of harnessing technology to enhance mentorship programs that increase social capital of disadvantaged populations.8-11 This approach already is in progress, bolstered by advances made in digital communications during the coronavirus disease 2019 pandemic.12 Student-led networking groups that connect remotely have been shown to build social capital bonds and bridges that facilitate collaborative learning, relationship building, and information sharing.8-11 There are existing online UIM student networks that individual dermatologists, institutions, and national organizations can partner with to facilitate the construction of bridges between these UIM student groups and dermatologists who can provide accurate, high-yield information and professional networking; however, one limitation of this suggestion is the disparate access to technology in the UIM community.
Final Thoughts
It is important to note that assumptions should not be made about the level of economic, social, or cultural capital an individual possesses based on his/her race or ethnicity. Instead, mentors should attempt to be available to a diverse pool of students; take the time to get to know these students; and then provide the types of mentorship, information, exposure, and networking that each individual student needs. Another approach is to make a concerted effort to ensure that all students receive the same amount and quality of information about medical education and our specialty regardless of their level of economic, cultural, or social capital. Moreover, beyond the promotion of diversity through increasing numbers of UIM applicants, we should seek to reshape our specialty into a space that does not require students to subdue their existing diverse forms of capital but rather to bring these different perspectives and lived experiences to the table.13
As our specialty seeks to address its lack of racial diversity, many dermatologists have answered recent calls to action.1,2 As we work toward dismantling systemic issues that have created pervasive inequality in our residency application review and interview processes, consideration also should be given to psychosocial issues that underrepresented-in-medicine (UIM) students face before their applications come to our attention. In this article, we explore how potential differences in the social capital of UIM and other disadvantaged dermatology residency applicants contribute to persistent homogeneity among dermatology training programs and the workforce.
The Theory of Capital
The concepts of economic, social, and cultural capital originate from the writings of social theorist Pierre Bourdieu.3 All 3 forms of capital are interconnected, and they relate to each other in ways that often facilitate social division and inequality. Economic capital denotes an individual’s economic resources or wealth, while cultural capital refers to the knowledge, behaviors, and skills that demonstrate his/her economic class (eg, communication style, table manners).3 Social capital refers to an individual’s interpersonal connections in personal and professional settings and can be subdivided into 3 categories: bonds, bridges, and linkages.4,5 Herein, we will focus on bonds and bridges.
It has been suggested that bonds are important for “getting by,” while bridges are critical for “getting ahead.”5 Bonds refer to close relationships within a community of people with shared characteristics, such as racial/ethnic identity and culture, access to information, and resources (eg, family, friends). These bonds provide trust, safety, and financial and emotional support; however, they are considered to be inward-looking and can promote exclusion and homogeneity.5
On the other hand, bridges refer to social relationships that extend outward beyond one’s close circle of family and friends to other people with shared interests and goals who may have different social or cultural identities (eg, professional colleagues). These bridges are considered to be outward-looking and provide many benefits to individuals and society. They link diverse individuals, which tends to increase tolerance and disrupt stereotypes, and they facilitate the sharing of ideas, information, and innovation. Additionally, bridges between individuals from different networks facilitate access to increased resources and opportunities for all parties.5
The 3 forms of capital are inextricably linked. For example, with economic capital, a child’s family can purchase access to a prestigious private high school, where he/she will gain valuable social capital through bridges with other students and their families. At this school, the child also will accumulate cultural capital that increases his/her sense of belonging in these circles. Subsequently, both the social and cultural capital accumulated at this private high school can be exchanged for economic capital via social networks, skills, values, and behaviors that facilitate entry into higher education and professional training. As such, these 3 forms of capital work together to continue social/class divisions, hierarchies, and ultimately inequality.
Impact of Social Capital in Pursuing a Medical Career
For medical students whose bonds (ie, close family, friends) include physicians or other health care professionals, the journey to studying medicine and entering their chosen specialty will be facilitated by financial security, valuable “inside information” about the application process, study skills, and even clinical guidance. Additionally, these students will have access to professional networks for mentorship, shadowing experiences, and other potential advantages. Furthermore, social capital is associated with higher self-esteem,6 which likely improves academic performance and wards off imposter syndrome in these students.
For medical students from lower socioeconomic status backgrounds or those whose inner circles do not include physicians or other health care professionals, accumulating the social and cultural capital needed to successfully navigate a medical career is more difficult. Although they may receive support and encouragement from family and friends, they will not have access to the same valuable information and connections that facilitate success; rather, they will have a further distance to travel, and this distance should be acknowledged in the residency application review process.
Acquiring Social Capital as a UIM Student
Despite the benefits of social and cultural capital, acquiring them takes a toll. For those UIM students who start life from a disadvantaged place, the accumulation of social capital does not come easily; rather, it demands effort and time that has the potential to detract from a student’s focus on the academic demands of medical education.7 Programs that attempt to improve disadvantaged students’ access to credible information, role models, and mentors can help lift some of the burden from the individual student’s shoulders. For example, studies have demonstrated the benefits of harnessing technology to enhance mentorship programs that increase social capital of disadvantaged populations.8-11 This approach already is in progress, bolstered by advances made in digital communications during the coronavirus disease 2019 pandemic.12 Student-led networking groups that connect remotely have been shown to build social capital bonds and bridges that facilitate collaborative learning, relationship building, and information sharing.8-11 There are existing online UIM student networks that individual dermatologists, institutions, and national organizations can partner with to facilitate the construction of bridges between these UIM student groups and dermatologists who can provide accurate, high-yield information and professional networking; however, one limitation of this suggestion is the disparate access to technology in the UIM community.
Final Thoughts
It is important to note that assumptions should not be made about the level of economic, social, or cultural capital an individual possesses based on his/her race or ethnicity. Instead, mentors should attempt to be available to a diverse pool of students; take the time to get to know these students; and then provide the types of mentorship, information, exposure, and networking that each individual student needs. Another approach is to make a concerted effort to ensure that all students receive the same amount and quality of information about medical education and our specialty regardless of their level of economic, cultural, or social capital. Moreover, beyond the promotion of diversity through increasing numbers of UIM applicants, we should seek to reshape our specialty into a space that does not require students to subdue their existing diverse forms of capital but rather to bring these different perspectives and lived experiences to the table.13
- Bray JK, McMichael AJ, Huang WW, et al. Publication rates on the topic of racial and ethnic diversity in dermatology versus other specialties. Dermatol Online J. 2020;26:7.
- Pritchett EN, Pandya AG, Ferguson NN, et al. Diversity in dermatology: roadmap for improvement. J Am Acad Dermatol. 2018;79:337-341.
- Bourdieu P. The forms of capital. In: Richardson J, ed. Handbook of Theory and Research for the Sociology of Education. Westport, CT: Greenwood; 1986:241-258.
- Granovetter MS. The strength of weak ties. Am J Sociol. 1973;78:1360-1380.
- Putnam RD. Bowling alone: America’s declining social capital. J Democracy. 1995;6:65-78.
- Han S. Longitudinal association between social capital and self-esteem: a matter of context. Psychiatry Research. 2015;226:340-346.
- Kirschling JM. Building social capital: leading and leveraging constituencies outside the college. J Nurs Educ. 2004;43:517-519.
- Radlick RL, Svedberg P, Nygren JM, et al. Digitally enhanced mentoring for immigrant youth social capital: protocol for a mixed methods pilot study and a randomized controlled trial [published online March 17, 2020]. JMIR Research Protocols. doi:10.2196/16472.
- Koh LC, Walker R, Wollersheim D, et al. I think someone is walking with me: the use of mobile phone for social capital development among women in four refugee communities. Int J Migration Health Social Care. 2018;14:411-424.
- Hartley A, Kassam AA. Social networking for learning in higher education: capitalising on social capital. ResearchGate website.https://www.researchgate.net/publication/311097860_Social_Networking_for_Learning_in_Higher_Education_Capitalising_on_Social_Capital. Published November 2016. Accessed October 19, 2020.
- Zalon ML. Using technology to build community in professional associations. J Contin Educ Nurs. 2008;39:235-240.
- Stewart CR, Chernoff KA, Wildman HF, et al. Recommendations for medical student preparedness and equity for dermatology residency applications during the COVID-19 pandemic. J Am Acad Dermatol. 2020;83:E225-E226.
- Brosnan C, Southgate E, Outram S, et al. Experiences of medical students who are first in family to attend university. Med Educ. 2016;50:842-851.
- Bray JK, McMichael AJ, Huang WW, et al. Publication rates on the topic of racial and ethnic diversity in dermatology versus other specialties. Dermatol Online J. 2020;26:7.
- Pritchett EN, Pandya AG, Ferguson NN, et al. Diversity in dermatology: roadmap for improvement. J Am Acad Dermatol. 2018;79:337-341.
- Bourdieu P. The forms of capital. In: Richardson J, ed. Handbook of Theory and Research for the Sociology of Education. Westport, CT: Greenwood; 1986:241-258.
- Granovetter MS. The strength of weak ties. Am J Sociol. 1973;78:1360-1380.
- Putnam RD. Bowling alone: America’s declining social capital. J Democracy. 1995;6:65-78.
- Han S. Longitudinal association between social capital and self-esteem: a matter of context. Psychiatry Research. 2015;226:340-346.
- Kirschling JM. Building social capital: leading and leveraging constituencies outside the college. J Nurs Educ. 2004;43:517-519.
- Radlick RL, Svedberg P, Nygren JM, et al. Digitally enhanced mentoring for immigrant youth social capital: protocol for a mixed methods pilot study and a randomized controlled trial [published online March 17, 2020]. JMIR Research Protocols. doi:10.2196/16472.
- Koh LC, Walker R, Wollersheim D, et al. I think someone is walking with me: the use of mobile phone for social capital development among women in four refugee communities. Int J Migration Health Social Care. 2018;14:411-424.
- Hartley A, Kassam AA. Social networking for learning in higher education: capitalising on social capital. ResearchGate website.https://www.researchgate.net/publication/311097860_Social_Networking_for_Learning_in_Higher_Education_Capitalising_on_Social_Capital. Published November 2016. Accessed October 19, 2020.
- Zalon ML. Using technology to build community in professional associations. J Contin Educ Nurs. 2008;39:235-240.
- Stewart CR, Chernoff KA, Wildman HF, et al. Recommendations for medical student preparedness and equity for dermatology residency applications during the COVID-19 pandemic. J Am Acad Dermatol. 2020;83:E225-E226.
- Brosnan C, Southgate E, Outram S, et al. Experiences of medical students who are first in family to attend university. Med Educ. 2016;50:842-851.
Practice Points
- Achieving diversity in the field of dermatology will require a concerted effort to equalize access to mentorship, information, exposure, and networking for students of all backgrounds.
- Valuing diverse forms of capital in applicants ultimately will strengthen the dermatology workforce through inclusion of various lived experiences and perspectives.
Primary care journals address systemic racism in medicine
Sumi Sexton, MD, editor in chief of American Family Physician (AFP), said in an interview she had been working on changes at her journal that would answer the need for action that was made clear by this summer’s Black Lives Matter protests and realized the issue was much bigger than one journal. She proposed the collaboration with the other editors.
The editors wrote a joint statement explaining what they plan to do collectively. It was published online Oct. 15 ahead of print and will be published in all 10 journals at the beginning of the year.
Following the action by family medicine editors, the American College of Physicians issued a statement expressing commitment to being an antiracist organization. It calls on all doctors to speak out against hate and discrimination and to act against institutional and systemic racism. The statement also apologizes for the organization’s own past actions: “ACP acknowledges and regrets its own historical organizational injustices and inequities, and past racism, discrimination and exclusionary practices throughout its history, whether intentional or unintentional, by act or omission.”
Family medicine journals plan changes
Changes will differ at each family medicine publication, according to Sexton and other interviewees. Some specific changes at AFP, for example, include creating a medical editor role dedicated to diversity, equity, and inclusion to ensure that content is not only accurate but also that more content addresses racism, Dr. Sexton said.
AFP is creating a Web page dedicated to diversity and will now capitalize the word “Black” in racial and cultural references. Recent calls for papers have included emphasis on finding authors from underrepresented groups and on mentoring new authors.
“We really need to enable our colleagues,” Dr. Sexton said.
The journals are also pooling their published research on topics of racism and inclusion and have established a joint bibliography.
The steps are important, Dr. Sexton said, because reform in research will start a “cascade of action” that will result in better patient care.
“Our mission is to care for the individual as a whole person,” Dr. Sexton said. “This is part of that mission.”
Increasing diversity on editorial boards
Family physician Kameron Leigh Matthews, MD, chief medical officer for the Veterans Health Administration, praised the journals’ plan.
She noted that the groups are addressing diversity on their editorial boards, as well as evaluating content. Effective change must also happen regarding the people reviewing the content, she said in an interview. “It has to be both.
“I’m very proud as a family physician that our editors came together and are giving the right response. It’s not enough to say we stand against racism. They’re actually offering concrete actions that they will take as editors, and that will influence health care,” she said.
Dr. Matthews pointed to an example of what can happen when the editorial process fails and racism is introduced in research.
She cited the retraction of an article in the Journal of the American Heart Association entitled, “Evolution of Race and Ethnicity Considerations for the Cardiology Workforce.” The article advocated for ending racial and ethnic preferences in undergraduate and medical school admissions.
The American Heart Association said the article concluded “incorrectly that Black and Hispanic trainees in medicine are less qualified than White and Asian trainees.” The article had “rightfully drawn criticism for its misrepresentations and conclusions,” the AHA said, adding that it would launch an investigation into how the article came to be published.
Dr. Matthews says that’s why it’s so important that, in their statement, the family medicine editors vow to address not only the content but also the editing process to avoid similar systemic lapses.
Dr. Matthews added that, because the proportion of physicians from underrepresented groups is small – only 5% of physicians are Black and 6% are Hispanic – it is vital, as recommended in the editors’ statement, to mentor researchers from underrepresented groups and to reach out to students and residents to be coauthors.
“To sit back and say there’s not enough to recruit from is not sufficient,” Dr. Matthews said. “You need to recognize that you need to assist with expanding the pool.”
She also said she would like to see the journals focus more heavily on solutions to racial disparities in health care rather than on pointing them out.
At the Journal of Family Practice (JFP), Editor in Chief John Hickner, MD, said adding diversity to the editorial board is a top priority. He also reiterated that diversity in top leadership is a concern across all the journals, inasmuch as only 1 of the 10 editors in chief is a person of color.
As an editor, he said, he will personally, as well as through family medicine department chairs, be seeking authors who are members of underrepresented groups and that he will be assisting those who need help.
“I’m committed to giving them special attention in the editorial process,” he said.
Dr. Hickner said the 10 journals have also committed to periodically evaluate whether their approaches are making substantial changes. He said the editors have vowed to meet at least once a year to review progress “and hold each other accountable.”
Statement authors, in addition to Dr. Sexton and Dr. Hickner, include these editors in chief: Caroline R. Richardson, MD, Annals of Family Medicine; Sarina B. Schrager, MD, FPM; Marjorie A. Bowman, MD, The Journal of the American Board of Family Medicine; Christopher P. Morley, PhD, PRiMER; Nicholas Pimlott, MD, PhD, Canadian Family Physician; John W. Saultz, MD, Family Medicine; and Barry D. Weiss, MD, FP Essentials.
The authors have disclosed no relevant financial relationships. The Journal of Family Practice is owned by the same news organization as this publication.
A version of this article originally appeared on Medscape.com.
Sumi Sexton, MD, editor in chief of American Family Physician (AFP), said in an interview she had been working on changes at her journal that would answer the need for action that was made clear by this summer’s Black Lives Matter protests and realized the issue was much bigger than one journal. She proposed the collaboration with the other editors.
The editors wrote a joint statement explaining what they plan to do collectively. It was published online Oct. 15 ahead of print and will be published in all 10 journals at the beginning of the year.
Following the action by family medicine editors, the American College of Physicians issued a statement expressing commitment to being an antiracist organization. It calls on all doctors to speak out against hate and discrimination and to act against institutional and systemic racism. The statement also apologizes for the organization’s own past actions: “ACP acknowledges and regrets its own historical organizational injustices and inequities, and past racism, discrimination and exclusionary practices throughout its history, whether intentional or unintentional, by act or omission.”
Family medicine journals plan changes
Changes will differ at each family medicine publication, according to Sexton and other interviewees. Some specific changes at AFP, for example, include creating a medical editor role dedicated to diversity, equity, and inclusion to ensure that content is not only accurate but also that more content addresses racism, Dr. Sexton said.
AFP is creating a Web page dedicated to diversity and will now capitalize the word “Black” in racial and cultural references. Recent calls for papers have included emphasis on finding authors from underrepresented groups and on mentoring new authors.
“We really need to enable our colleagues,” Dr. Sexton said.
The journals are also pooling their published research on topics of racism and inclusion and have established a joint bibliography.
The steps are important, Dr. Sexton said, because reform in research will start a “cascade of action” that will result in better patient care.
“Our mission is to care for the individual as a whole person,” Dr. Sexton said. “This is part of that mission.”
Increasing diversity on editorial boards
Family physician Kameron Leigh Matthews, MD, chief medical officer for the Veterans Health Administration, praised the journals’ plan.
She noted that the groups are addressing diversity on their editorial boards, as well as evaluating content. Effective change must also happen regarding the people reviewing the content, she said in an interview. “It has to be both.
“I’m very proud as a family physician that our editors came together and are giving the right response. It’s not enough to say we stand against racism. They’re actually offering concrete actions that they will take as editors, and that will influence health care,” she said.
Dr. Matthews pointed to an example of what can happen when the editorial process fails and racism is introduced in research.
She cited the retraction of an article in the Journal of the American Heart Association entitled, “Evolution of Race and Ethnicity Considerations for the Cardiology Workforce.” The article advocated for ending racial and ethnic preferences in undergraduate and medical school admissions.
The American Heart Association said the article concluded “incorrectly that Black and Hispanic trainees in medicine are less qualified than White and Asian trainees.” The article had “rightfully drawn criticism for its misrepresentations and conclusions,” the AHA said, adding that it would launch an investigation into how the article came to be published.
Dr. Matthews says that’s why it’s so important that, in their statement, the family medicine editors vow to address not only the content but also the editing process to avoid similar systemic lapses.
Dr. Matthews added that, because the proportion of physicians from underrepresented groups is small – only 5% of physicians are Black and 6% are Hispanic – it is vital, as recommended in the editors’ statement, to mentor researchers from underrepresented groups and to reach out to students and residents to be coauthors.
“To sit back and say there’s not enough to recruit from is not sufficient,” Dr. Matthews said. “You need to recognize that you need to assist with expanding the pool.”
She also said she would like to see the journals focus more heavily on solutions to racial disparities in health care rather than on pointing them out.
At the Journal of Family Practice (JFP), Editor in Chief John Hickner, MD, said adding diversity to the editorial board is a top priority. He also reiterated that diversity in top leadership is a concern across all the journals, inasmuch as only 1 of the 10 editors in chief is a person of color.
As an editor, he said, he will personally, as well as through family medicine department chairs, be seeking authors who are members of underrepresented groups and that he will be assisting those who need help.
“I’m committed to giving them special attention in the editorial process,” he said.
Dr. Hickner said the 10 journals have also committed to periodically evaluate whether their approaches are making substantial changes. He said the editors have vowed to meet at least once a year to review progress “and hold each other accountable.”
Statement authors, in addition to Dr. Sexton and Dr. Hickner, include these editors in chief: Caroline R. Richardson, MD, Annals of Family Medicine; Sarina B. Schrager, MD, FPM; Marjorie A. Bowman, MD, The Journal of the American Board of Family Medicine; Christopher P. Morley, PhD, PRiMER; Nicholas Pimlott, MD, PhD, Canadian Family Physician; John W. Saultz, MD, Family Medicine; and Barry D. Weiss, MD, FP Essentials.
The authors have disclosed no relevant financial relationships. The Journal of Family Practice is owned by the same news organization as this publication.
A version of this article originally appeared on Medscape.com.
Sumi Sexton, MD, editor in chief of American Family Physician (AFP), said in an interview she had been working on changes at her journal that would answer the need for action that was made clear by this summer’s Black Lives Matter protests and realized the issue was much bigger than one journal. She proposed the collaboration with the other editors.
The editors wrote a joint statement explaining what they plan to do collectively. It was published online Oct. 15 ahead of print and will be published in all 10 journals at the beginning of the year.
Following the action by family medicine editors, the American College of Physicians issued a statement expressing commitment to being an antiracist organization. It calls on all doctors to speak out against hate and discrimination and to act against institutional and systemic racism. The statement also apologizes for the organization’s own past actions: “ACP acknowledges and regrets its own historical organizational injustices and inequities, and past racism, discrimination and exclusionary practices throughout its history, whether intentional or unintentional, by act or omission.”
Family medicine journals plan changes
Changes will differ at each family medicine publication, according to Sexton and other interviewees. Some specific changes at AFP, for example, include creating a medical editor role dedicated to diversity, equity, and inclusion to ensure that content is not only accurate but also that more content addresses racism, Dr. Sexton said.
AFP is creating a Web page dedicated to diversity and will now capitalize the word “Black” in racial and cultural references. Recent calls for papers have included emphasis on finding authors from underrepresented groups and on mentoring new authors.
“We really need to enable our colleagues,” Dr. Sexton said.
The journals are also pooling their published research on topics of racism and inclusion and have established a joint bibliography.
The steps are important, Dr. Sexton said, because reform in research will start a “cascade of action” that will result in better patient care.
“Our mission is to care for the individual as a whole person,” Dr. Sexton said. “This is part of that mission.”
Increasing diversity on editorial boards
Family physician Kameron Leigh Matthews, MD, chief medical officer for the Veterans Health Administration, praised the journals’ plan.
She noted that the groups are addressing diversity on their editorial boards, as well as evaluating content. Effective change must also happen regarding the people reviewing the content, she said in an interview. “It has to be both.
“I’m very proud as a family physician that our editors came together and are giving the right response. It’s not enough to say we stand against racism. They’re actually offering concrete actions that they will take as editors, and that will influence health care,” she said.
Dr. Matthews pointed to an example of what can happen when the editorial process fails and racism is introduced in research.
She cited the retraction of an article in the Journal of the American Heart Association entitled, “Evolution of Race and Ethnicity Considerations for the Cardiology Workforce.” The article advocated for ending racial and ethnic preferences in undergraduate and medical school admissions.
The American Heart Association said the article concluded “incorrectly that Black and Hispanic trainees in medicine are less qualified than White and Asian trainees.” The article had “rightfully drawn criticism for its misrepresentations and conclusions,” the AHA said, adding that it would launch an investigation into how the article came to be published.
Dr. Matthews says that’s why it’s so important that, in their statement, the family medicine editors vow to address not only the content but also the editing process to avoid similar systemic lapses.
Dr. Matthews added that, because the proportion of physicians from underrepresented groups is small – only 5% of physicians are Black and 6% are Hispanic – it is vital, as recommended in the editors’ statement, to mentor researchers from underrepresented groups and to reach out to students and residents to be coauthors.
“To sit back and say there’s not enough to recruit from is not sufficient,” Dr. Matthews said. “You need to recognize that you need to assist with expanding the pool.”
She also said she would like to see the journals focus more heavily on solutions to racial disparities in health care rather than on pointing them out.
At the Journal of Family Practice (JFP), Editor in Chief John Hickner, MD, said adding diversity to the editorial board is a top priority. He also reiterated that diversity in top leadership is a concern across all the journals, inasmuch as only 1 of the 10 editors in chief is a person of color.
As an editor, he said, he will personally, as well as through family medicine department chairs, be seeking authors who are members of underrepresented groups and that he will be assisting those who need help.
“I’m committed to giving them special attention in the editorial process,” he said.
Dr. Hickner said the 10 journals have also committed to periodically evaluate whether their approaches are making substantial changes. He said the editors have vowed to meet at least once a year to review progress “and hold each other accountable.”
Statement authors, in addition to Dr. Sexton and Dr. Hickner, include these editors in chief: Caroline R. Richardson, MD, Annals of Family Medicine; Sarina B. Schrager, MD, FPM; Marjorie A. Bowman, MD, The Journal of the American Board of Family Medicine; Christopher P. Morley, PhD, PRiMER; Nicholas Pimlott, MD, PhD, Canadian Family Physician; John W. Saultz, MD, Family Medicine; and Barry D. Weiss, MD, FP Essentials.
The authors have disclosed no relevant financial relationships. The Journal of Family Practice is owned by the same news organization as this publication.
A version of this article originally appeared on Medscape.com.
Dermatologists as Social Media Contributors During the COVID-19 Pandemic
On December 31, 2019, cases of a severe pneumonia in patients in Wuhan, Hubei Province, China, were reported to the World Health Organization.1,2 The novel coronavirus—severe acute respiratory syndrome coronavirus 2—was identified, and the coronavirus disease 2019 (COVID-19) became a public health emergency of international concern.1 In March 2020, the World Health Organization officially characterized COVID-19 as a pandemic.3 As of October 2020, more than 42.3 million cases and 1.1 million deaths from COVID-19 have been confirmed worldwide.4
As more understanding of severe acute respiratory syndrome coronavirus 2 develops, various cutaneous manifestations of COVID-19 are being uncovered.5 The most common cutaneous manifestations of COVID-19 reported in the literature are maculopapular or morbilliform exanthem (36.1% of cutaneous manifestations), papulovesicular rash (34.7%), painful acral red purple papules (15.3%), urticaria (9.7%), livedo reticularis lesions (2.8%), and petechiae (1.4%).5
Interestingly, a series of unique cases was identified in April 2020 by a group of dermatologists in Spain. Most patients were children (median age, 13 years) or young adults (median age, 31 years; average age, 36 years; adult age range, 18–91 years).1 Reporting on a representative sample of 6 patients in that series, the group noted that lesions, initially reddish, papular, and resembling chilblains (pernio), progressively became purpuric and flattened in the course of 1 week. Although the lesions presented with some referred discomfort or pain with palpation, they were not highly symptomatic, and no signs of ischemia or Raynaud syndrome were identified. Over time, lesions self-resolved without intervention. Most patients also did not present with what are considered classic COVID-19 signs or symptoms. Only the oldest patient (aged 91 years) presented with a notable respiratory condition; the remaining patients generally were in good health.1 Dermatologists in Italy, France, and the United States also have witnessed these COVID-19–associated cutaneous manifestations.
Scientific understanding of COVID-19 and its associated dermatologic symptoms is evolving. Attention has turned to social media to inform and provide possible health solutions during this unprecedented medical crisis.6 Strict physical distancing measures have made patients and providers alike reliant on global digital social networks, such as Instagram, Twitter, and Facebook, to facilitate information sharing about COVID-19.7 The abundance of nonexpert advice and misinformation on social media makes communication of unbiased expert information difficult.8,9 Furthermore, there is a need for dermatologists to provide medical information to patients regarding COVID-19, such as dermatologic manifestations, and clear guidance on immunobiologic or systemic medications during this unprecedented time.9
In recent years, dermatologists have established a growing presence on social media, with many recognized as social media influencers with the ability to affect patients’ health-related behavior.10 Social media frequently has been used by patients to solicit advice regarding skin concerns.9,10 Many individuals, in fact, never see a physician after consulting social media for medical concerns or professional advice.9
In addition, as of March 2020, more than 61% of health care workers were found to use social media as a source of COVID-19–related information.11 Therefore, dermatologists should utilize social media as a platform to share evidence-based information with the public and other health care workers.
Through social media, dermatologists can post high-quality images with clear descriptions to fully characterize skin manifestations in patients with COVID-19. The process of capturing and posting images to the virtual world using a smartphone allows practitioners to gain advice from peers and consultants, share findings with colleagues, and inform the public.12 Social media posts of many deidentified clinical images of rashes in COVID-19–infected patients already have enabled rapid recognition of skin signs by dermatologists.13
Social media sites also are resources where organizations can post updated, evidence-based findings from academic journals. For example, the American Academy of Dermatology and its official journal, the Journal of the American Academy of Dermatology, had more than 22,000 and 27,000 Instagram followers, respectively, as of a March 2020 analysis.14 Recent online forums and social media posts contain accessible, graphical, patient-friendly images and information on evidence-based treatments for skin disease during the COVID-19 pandemic.13
We should consider initiatives that empower dermatologists to use social media to post unbiased, evidence-based information regarding manifestations of COVID-19 and guidelines for treatment of skin disease during this medical crisis. We hope that dermatologists will help lead the global response to the COVID-19 pandemic and contribute to the evolving knowledge base by characterizing COVID-19–related rashes, understanding their implications, and determining the best evidence for treatment.
- Landa N, Mendieta-Eckert M, Fonda-Pascual P, et al. Chilblain-like lesions on feet and hands during the COVID-19 pandemic. Int J Dermatol. 2020;59:739-743.
- Phelan AL, Katz R, Gostin LO. The novel coronavirus originating in Wuhan, China: challenges for global health governance. JAMA. 2020;323:709-710.
- World Health Organization. Coronavirus disease (COVID-19) Situation Report – 133. WHO Website. June 1, 2020. www.who.int/docs/default-source/coronaviruse/situation-reports/20200601-covid-19-sitrep-133.pdf?sfvrsn=9a56f2ac_4. Accessed October 14, 2020.
- COVID-19 dashboard by the Center for Systems Science and Engineering (CSSE) at John Hopkins University. John Hopkins Coronavirus Resource Center website. https://coronavirus.jhu.edu/map.html. Accessed October 24, 2020.
- Sachdeva M, Gianotti R, Shah M, et al. Cutaneous manifestations of COVID-19: report of three cases and a review of literature. J Dermatolog Sci. 2020;98:75-81.
- Kapoor A, Guha S, Kanti Das M, et al. Digital healthcare: the only solution for better healthcare during COVID-19 pandemic? Indian Heart J. 2020;72:61-64.
- Limaye RJ, Sauer M, Ali J, et al. Building trust while influencing online COVID-19 content in the social media world. Lancet Digit Health. 2020;2:E277-E278.
- Chawla S. COVID-19: challenges and opportunities for dermatology response. J Dermatolog Treat. 2020;31:326-326.
- Schoenberg E, Shalabi D, Wang JV, et al. Public social media consultations for dermatologic conditions: an online survey. Dermatol Online J. 2020;26:6.
- DeBord LC, Patel V, Braun TL, et al. Social media in dermatology: clinical relevance, academic value, and trends across platforms. J Dermatolog Treat. 2019;30:511-518.
- Bhagavathula AS, Aldhaleei WA, Rahmani J, et al. Knowledge and perceptions of COVID-19 among health care workers: cross-sectional study. JMIR Public Health Surveill. 2020;6:E19160.
- Ashique KT, Kaliyadan F, Aurangabadkar SJ. Clinical photography in dermatology using smartphones: an overview. Indian Dermatol Online J. 2015;6:158-163.
- Madigan LM, Micheletti RG, Shinkai K. How dermatologists can learn and contribute at the leading edge of the COVID-19 global pandemic. JAMA Dermatol. 2020;156:733-734.
- Guzman AK, Barbieri JS. Response to: “Dermatologists in social media: a study on top influencers, posts, and user engagement” [published online April 20, 2020]. J Am Acad Dermatol. doi:10.1016/j.jaad.2020.03.118.
On December 31, 2019, cases of a severe pneumonia in patients in Wuhan, Hubei Province, China, were reported to the World Health Organization.1,2 The novel coronavirus—severe acute respiratory syndrome coronavirus 2—was identified, and the coronavirus disease 2019 (COVID-19) became a public health emergency of international concern.1 In March 2020, the World Health Organization officially characterized COVID-19 as a pandemic.3 As of October 2020, more than 42.3 million cases and 1.1 million deaths from COVID-19 have been confirmed worldwide.4
As more understanding of severe acute respiratory syndrome coronavirus 2 develops, various cutaneous manifestations of COVID-19 are being uncovered.5 The most common cutaneous manifestations of COVID-19 reported in the literature are maculopapular or morbilliform exanthem (36.1% of cutaneous manifestations), papulovesicular rash (34.7%), painful acral red purple papules (15.3%), urticaria (9.7%), livedo reticularis lesions (2.8%), and petechiae (1.4%).5
Interestingly, a series of unique cases was identified in April 2020 by a group of dermatologists in Spain. Most patients were children (median age, 13 years) or young adults (median age, 31 years; average age, 36 years; adult age range, 18–91 years).1 Reporting on a representative sample of 6 patients in that series, the group noted that lesions, initially reddish, papular, and resembling chilblains (pernio), progressively became purpuric and flattened in the course of 1 week. Although the lesions presented with some referred discomfort or pain with palpation, they were not highly symptomatic, and no signs of ischemia or Raynaud syndrome were identified. Over time, lesions self-resolved without intervention. Most patients also did not present with what are considered classic COVID-19 signs or symptoms. Only the oldest patient (aged 91 years) presented with a notable respiratory condition; the remaining patients generally were in good health.1 Dermatologists in Italy, France, and the United States also have witnessed these COVID-19–associated cutaneous manifestations.
Scientific understanding of COVID-19 and its associated dermatologic symptoms is evolving. Attention has turned to social media to inform and provide possible health solutions during this unprecedented medical crisis.6 Strict physical distancing measures have made patients and providers alike reliant on global digital social networks, such as Instagram, Twitter, and Facebook, to facilitate information sharing about COVID-19.7 The abundance of nonexpert advice and misinformation on social media makes communication of unbiased expert information difficult.8,9 Furthermore, there is a need for dermatologists to provide medical information to patients regarding COVID-19, such as dermatologic manifestations, and clear guidance on immunobiologic or systemic medications during this unprecedented time.9
In recent years, dermatologists have established a growing presence on social media, with many recognized as social media influencers with the ability to affect patients’ health-related behavior.10 Social media frequently has been used by patients to solicit advice regarding skin concerns.9,10 Many individuals, in fact, never see a physician after consulting social media for medical concerns or professional advice.9
In addition, as of March 2020, more than 61% of health care workers were found to use social media as a source of COVID-19–related information.11 Therefore, dermatologists should utilize social media as a platform to share evidence-based information with the public and other health care workers.
Through social media, dermatologists can post high-quality images with clear descriptions to fully characterize skin manifestations in patients with COVID-19. The process of capturing and posting images to the virtual world using a smartphone allows practitioners to gain advice from peers and consultants, share findings with colleagues, and inform the public.12 Social media posts of many deidentified clinical images of rashes in COVID-19–infected patients already have enabled rapid recognition of skin signs by dermatologists.13
Social media sites also are resources where organizations can post updated, evidence-based findings from academic journals. For example, the American Academy of Dermatology and its official journal, the Journal of the American Academy of Dermatology, had more than 22,000 and 27,000 Instagram followers, respectively, as of a March 2020 analysis.14 Recent online forums and social media posts contain accessible, graphical, patient-friendly images and information on evidence-based treatments for skin disease during the COVID-19 pandemic.13
We should consider initiatives that empower dermatologists to use social media to post unbiased, evidence-based information regarding manifestations of COVID-19 and guidelines for treatment of skin disease during this medical crisis. We hope that dermatologists will help lead the global response to the COVID-19 pandemic and contribute to the evolving knowledge base by characterizing COVID-19–related rashes, understanding their implications, and determining the best evidence for treatment.
On December 31, 2019, cases of a severe pneumonia in patients in Wuhan, Hubei Province, China, were reported to the World Health Organization.1,2 The novel coronavirus—severe acute respiratory syndrome coronavirus 2—was identified, and the coronavirus disease 2019 (COVID-19) became a public health emergency of international concern.1 In March 2020, the World Health Organization officially characterized COVID-19 as a pandemic.3 As of October 2020, more than 42.3 million cases and 1.1 million deaths from COVID-19 have been confirmed worldwide.4
As more understanding of severe acute respiratory syndrome coronavirus 2 develops, various cutaneous manifestations of COVID-19 are being uncovered.5 The most common cutaneous manifestations of COVID-19 reported in the literature are maculopapular or morbilliform exanthem (36.1% of cutaneous manifestations), papulovesicular rash (34.7%), painful acral red purple papules (15.3%), urticaria (9.7%), livedo reticularis lesions (2.8%), and petechiae (1.4%).5
Interestingly, a series of unique cases was identified in April 2020 by a group of dermatologists in Spain. Most patients were children (median age, 13 years) or young adults (median age, 31 years; average age, 36 years; adult age range, 18–91 years).1 Reporting on a representative sample of 6 patients in that series, the group noted that lesions, initially reddish, papular, and resembling chilblains (pernio), progressively became purpuric and flattened in the course of 1 week. Although the lesions presented with some referred discomfort or pain with palpation, they were not highly symptomatic, and no signs of ischemia or Raynaud syndrome were identified. Over time, lesions self-resolved without intervention. Most patients also did not present with what are considered classic COVID-19 signs or symptoms. Only the oldest patient (aged 91 years) presented with a notable respiratory condition; the remaining patients generally were in good health.1 Dermatologists in Italy, France, and the United States also have witnessed these COVID-19–associated cutaneous manifestations.
Scientific understanding of COVID-19 and its associated dermatologic symptoms is evolving. Attention has turned to social media to inform and provide possible health solutions during this unprecedented medical crisis.6 Strict physical distancing measures have made patients and providers alike reliant on global digital social networks, such as Instagram, Twitter, and Facebook, to facilitate information sharing about COVID-19.7 The abundance of nonexpert advice and misinformation on social media makes communication of unbiased expert information difficult.8,9 Furthermore, there is a need for dermatologists to provide medical information to patients regarding COVID-19, such as dermatologic manifestations, and clear guidance on immunobiologic or systemic medications during this unprecedented time.9
In recent years, dermatologists have established a growing presence on social media, with many recognized as social media influencers with the ability to affect patients’ health-related behavior.10 Social media frequently has been used by patients to solicit advice regarding skin concerns.9,10 Many individuals, in fact, never see a physician after consulting social media for medical concerns or professional advice.9
In addition, as of March 2020, more than 61% of health care workers were found to use social media as a source of COVID-19–related information.11 Therefore, dermatologists should utilize social media as a platform to share evidence-based information with the public and other health care workers.
Through social media, dermatologists can post high-quality images with clear descriptions to fully characterize skin manifestations in patients with COVID-19. The process of capturing and posting images to the virtual world using a smartphone allows practitioners to gain advice from peers and consultants, share findings with colleagues, and inform the public.12 Social media posts of many deidentified clinical images of rashes in COVID-19–infected patients already have enabled rapid recognition of skin signs by dermatologists.13
Social media sites also are resources where organizations can post updated, evidence-based findings from academic journals. For example, the American Academy of Dermatology and its official journal, the Journal of the American Academy of Dermatology, had more than 22,000 and 27,000 Instagram followers, respectively, as of a March 2020 analysis.14 Recent online forums and social media posts contain accessible, graphical, patient-friendly images and information on evidence-based treatments for skin disease during the COVID-19 pandemic.13
We should consider initiatives that empower dermatologists to use social media to post unbiased, evidence-based information regarding manifestations of COVID-19 and guidelines for treatment of skin disease during this medical crisis. We hope that dermatologists will help lead the global response to the COVID-19 pandemic and contribute to the evolving knowledge base by characterizing COVID-19–related rashes, understanding their implications, and determining the best evidence for treatment.
- Landa N, Mendieta-Eckert M, Fonda-Pascual P, et al. Chilblain-like lesions on feet and hands during the COVID-19 pandemic. Int J Dermatol. 2020;59:739-743.
- Phelan AL, Katz R, Gostin LO. The novel coronavirus originating in Wuhan, China: challenges for global health governance. JAMA. 2020;323:709-710.
- World Health Organization. Coronavirus disease (COVID-19) Situation Report – 133. WHO Website. June 1, 2020. www.who.int/docs/default-source/coronaviruse/situation-reports/20200601-covid-19-sitrep-133.pdf?sfvrsn=9a56f2ac_4. Accessed October 14, 2020.
- COVID-19 dashboard by the Center for Systems Science and Engineering (CSSE) at John Hopkins University. John Hopkins Coronavirus Resource Center website. https://coronavirus.jhu.edu/map.html. Accessed October 24, 2020.
- Sachdeva M, Gianotti R, Shah M, et al. Cutaneous manifestations of COVID-19: report of three cases and a review of literature. J Dermatolog Sci. 2020;98:75-81.
- Kapoor A, Guha S, Kanti Das M, et al. Digital healthcare: the only solution for better healthcare during COVID-19 pandemic? Indian Heart J. 2020;72:61-64.
- Limaye RJ, Sauer M, Ali J, et al. Building trust while influencing online COVID-19 content in the social media world. Lancet Digit Health. 2020;2:E277-E278.
- Chawla S. COVID-19: challenges and opportunities for dermatology response. J Dermatolog Treat. 2020;31:326-326.
- Schoenberg E, Shalabi D, Wang JV, et al. Public social media consultations for dermatologic conditions: an online survey. Dermatol Online J. 2020;26:6.
- DeBord LC, Patel V, Braun TL, et al. Social media in dermatology: clinical relevance, academic value, and trends across platforms. J Dermatolog Treat. 2019;30:511-518.
- Bhagavathula AS, Aldhaleei WA, Rahmani J, et al. Knowledge and perceptions of COVID-19 among health care workers: cross-sectional study. JMIR Public Health Surveill. 2020;6:E19160.
- Ashique KT, Kaliyadan F, Aurangabadkar SJ. Clinical photography in dermatology using smartphones: an overview. Indian Dermatol Online J. 2015;6:158-163.
- Madigan LM, Micheletti RG, Shinkai K. How dermatologists can learn and contribute at the leading edge of the COVID-19 global pandemic. JAMA Dermatol. 2020;156:733-734.
- Guzman AK, Barbieri JS. Response to: “Dermatologists in social media: a study on top influencers, posts, and user engagement” [published online April 20, 2020]. J Am Acad Dermatol. doi:10.1016/j.jaad.2020.03.118.
- Landa N, Mendieta-Eckert M, Fonda-Pascual P, et al. Chilblain-like lesions on feet and hands during the COVID-19 pandemic. Int J Dermatol. 2020;59:739-743.
- Phelan AL, Katz R, Gostin LO. The novel coronavirus originating in Wuhan, China: challenges for global health governance. JAMA. 2020;323:709-710.
- World Health Organization. Coronavirus disease (COVID-19) Situation Report – 133. WHO Website. June 1, 2020. www.who.int/docs/default-source/coronaviruse/situation-reports/20200601-covid-19-sitrep-133.pdf?sfvrsn=9a56f2ac_4. Accessed October 14, 2020.
- COVID-19 dashboard by the Center for Systems Science and Engineering (CSSE) at John Hopkins University. John Hopkins Coronavirus Resource Center website. https://coronavirus.jhu.edu/map.html. Accessed October 24, 2020.
- Sachdeva M, Gianotti R, Shah M, et al. Cutaneous manifestations of COVID-19: report of three cases and a review of literature. J Dermatolog Sci. 2020;98:75-81.
- Kapoor A, Guha S, Kanti Das M, et al. Digital healthcare: the only solution for better healthcare during COVID-19 pandemic? Indian Heart J. 2020;72:61-64.
- Limaye RJ, Sauer M, Ali J, et al. Building trust while influencing online COVID-19 content in the social media world. Lancet Digit Health. 2020;2:E277-E278.
- Chawla S. COVID-19: challenges and opportunities for dermatology response. J Dermatolog Treat. 2020;31:326-326.
- Schoenberg E, Shalabi D, Wang JV, et al. Public social media consultations for dermatologic conditions: an online survey. Dermatol Online J. 2020;26:6.
- DeBord LC, Patel V, Braun TL, et al. Social media in dermatology: clinical relevance, academic value, and trends across platforms. J Dermatolog Treat. 2019;30:511-518.
- Bhagavathula AS, Aldhaleei WA, Rahmani J, et al. Knowledge and perceptions of COVID-19 among health care workers: cross-sectional study. JMIR Public Health Surveill. 2020;6:E19160.
- Ashique KT, Kaliyadan F, Aurangabadkar SJ. Clinical photography in dermatology using smartphones: an overview. Indian Dermatol Online J. 2015;6:158-163.
- Madigan LM, Micheletti RG, Shinkai K. How dermatologists can learn and contribute at the leading edge of the COVID-19 global pandemic. JAMA Dermatol. 2020;156:733-734.
- Guzman AK, Barbieri JS. Response to: “Dermatologists in social media: a study on top influencers, posts, and user engagement” [published online April 20, 2020]. J Am Acad Dermatol. doi:10.1016/j.jaad.2020.03.118.
Practice Points
- With the coronavirus disease 2019 (COVID-19) pandemic, strict physical distancing measures have made patients and providers alike reliant on global digital social networks such as Instagram, Twitter, and Facebook to facilitate information sharing about COVID-19.
- Dermatologists should utilize social media as a platform to post unbiased, evidence-based information regarding manifestations of COVID-19 and guidelines for treatment of skin disease during the global pandemic.
Twelve medical groups pen letter opposing UHC copay accumulator program
ACR leads outcry against the insurer’s proposed move
Last month, the American College of Rheumatology joined with 11 other medical associations and disease societies asking health insurance giant UnitedHealthcare (UHC) to not proceed with its proposed copay accumulator medical benefit program.
Copay accumulators are policies adopted by insurance companies or their pharmacy benefit managers to exclude patient copayment assistance programs for high-cost drugs, which are promulgated by the drug manufacturers, from being applied to a patient’s annual deductibles or out-of-pocket maximums. The manufacturer’s copay assistance, such as in the form of coupons, is designed to minimize the patient’s out-of-pocket costs. But insurers believe manufacturers will have no pressure to lower the prices of expensive specialty drugs unless patients are unable to afford them. Copay accumulators thus are aimed at giving insurers more leverage in negotiating prices for high-cost drugs.
UHC issued its new copay accumulator protocol for commercial individual and fully insured group plans in early October, effective Jan. 1, 2021, “in order to align employer costs for specialty medications with actual member out of pocket and deductibles,” according to the company’s announcement. In other words, patients will need to pay a higher share of the costs of these medications, said rheumatologist Christopher Phillips, MD, who chairs the Insurance Subcommittee of ACR’s Rheumatologic Care Committee. The annual price of biologic therapies for rheumatologic conditions ranges from $22,000 to $44,000, according to a recent press release from ACR.
The copay accumulator will negate the benefits of manufacturers’ copayment assistance programs for the patient, shifting more of the cost to the patient. With patients being forced to pay a higher share of drug costs for expensive biologic treatments for rheumatoid arthritis, lupus, and other rheumatologic conditions, they’ll stop taking the treatments, Dr. Phillips said.
“In my solo rheumatology practice in Paducah, Kentucky, when I’ve seen this kind of program applied on the pharmacy benefit side, rather than the medical benefit side, almost uniformly patients stop taking the high-cost treatments.” That can lead to disease flares, complications, and permanent disability. The newer rheumatologic drugs can cost $500 to $1,000 per treatment, and in many cases, there’s no generic or lower-cost alternative, he says. “We see policies like this as sacrificing patients to the battle over high drug prices. It’s bad practice, bad for patient outcomes, and nobody – apart from the payer – benefits.”
In ACR’s 2020 Rheumatic Disease Patient Survey, nearly half of 1,109 online survey respondents who had rheumatic diseases reported out-of-pocket costs greater than $1,000 per year for treatment. An IQVIA report from 2016 found that one in four specialty brand prescriptions are abandoned during the deductible phase, three times the rate seen when there is no deductible.
In an Oct. 7 letter to UHC, the 12 groups acknowledged that the drugs targeted by the accumulator policy are expensive. “However, they are also vitally important for our patients.” In addition to the ACR, the organizations involved include the AIDS Institute, American Academy of Dermatology Association, American Academy of Neurology, American College of Gastroenterology, American Gastroenterological Association, American Kidney Fund, Arthritis Foundation, Association for Clinical Oncology, Cancer Support Community, Coalition of State Rheumatology Organizations, and National Multiple Sclerosis Society.
UHC did not reply to questions in time for publication.
First large-scale payer to try copay accumulator program
Under UHC’s proposed policy, providers will be required to use UHC’s portal to report payment information received from drug manufacturer copay assistance programs that are applied to patients’ cost share of these drugs through a complex, 14-step “coupon submission process” involving multiple technology interfaces. “My first oath as a physician is to do no harm to my patient. Many of us are concerned about making these reports, which could harm our patients and undermine the doctor-patient relationship,” Dr. Phillips said.
“If I don’t report, what happens? I don’t think we know the answer to that. Some of us may decide we need to part ways with UHC.” Others may decline to participate in the drug manufacturers’ coupon programs beyond simply informing patients that manufacturer assistance is available.
“We’ve watched these copay accumulator policies for several years,” he said. “Some of them are rather opaque, with names like ‘copay savings programs’ or ‘copay value programs.’ But we had not seen a large-scale payer try to do this until now. Let’s face it: If UHC’s policy goes through, you can count the days until we see it from others.”
The Department of Health & Human Services, in its May 2020 final federal “Notice of Benefit and Payment Parameters for 2021,” indicated that individual states have the responsibility to regulate copay accumulator programs. Five states have banned them or restricted their use for individual and small group health plans. Arizona, Illinois, Virginia, and West Virginia passed such laws in 2019, and Georgia did so earlier this year.
“In next year’s state legislative sessions, we’ll make it a priority to pursue similar laws in other states,” Dr. Phillips said. “I’d encourage rheumatologists to educate their patients on the issues and be active in advocating for them.”
ACR leads outcry against the insurer’s proposed move
ACR leads outcry against the insurer’s proposed move
Last month, the American College of Rheumatology joined with 11 other medical associations and disease societies asking health insurance giant UnitedHealthcare (UHC) to not proceed with its proposed copay accumulator medical benefit program.
Copay accumulators are policies adopted by insurance companies or their pharmacy benefit managers to exclude patient copayment assistance programs for high-cost drugs, which are promulgated by the drug manufacturers, from being applied to a patient’s annual deductibles or out-of-pocket maximums. The manufacturer’s copay assistance, such as in the form of coupons, is designed to minimize the patient’s out-of-pocket costs. But insurers believe manufacturers will have no pressure to lower the prices of expensive specialty drugs unless patients are unable to afford them. Copay accumulators thus are aimed at giving insurers more leverage in negotiating prices for high-cost drugs.
UHC issued its new copay accumulator protocol for commercial individual and fully insured group plans in early October, effective Jan. 1, 2021, “in order to align employer costs for specialty medications with actual member out of pocket and deductibles,” according to the company’s announcement. In other words, patients will need to pay a higher share of the costs of these medications, said rheumatologist Christopher Phillips, MD, who chairs the Insurance Subcommittee of ACR’s Rheumatologic Care Committee. The annual price of biologic therapies for rheumatologic conditions ranges from $22,000 to $44,000, according to a recent press release from ACR.
The copay accumulator will negate the benefits of manufacturers’ copayment assistance programs for the patient, shifting more of the cost to the patient. With patients being forced to pay a higher share of drug costs for expensive biologic treatments for rheumatoid arthritis, lupus, and other rheumatologic conditions, they’ll stop taking the treatments, Dr. Phillips said.
“In my solo rheumatology practice in Paducah, Kentucky, when I’ve seen this kind of program applied on the pharmacy benefit side, rather than the medical benefit side, almost uniformly patients stop taking the high-cost treatments.” That can lead to disease flares, complications, and permanent disability. The newer rheumatologic drugs can cost $500 to $1,000 per treatment, and in many cases, there’s no generic or lower-cost alternative, he says. “We see policies like this as sacrificing patients to the battle over high drug prices. It’s bad practice, bad for patient outcomes, and nobody – apart from the payer – benefits.”
In ACR’s 2020 Rheumatic Disease Patient Survey, nearly half of 1,109 online survey respondents who had rheumatic diseases reported out-of-pocket costs greater than $1,000 per year for treatment. An IQVIA report from 2016 found that one in four specialty brand prescriptions are abandoned during the deductible phase, three times the rate seen when there is no deductible.
In an Oct. 7 letter to UHC, the 12 groups acknowledged that the drugs targeted by the accumulator policy are expensive. “However, they are also vitally important for our patients.” In addition to the ACR, the organizations involved include the AIDS Institute, American Academy of Dermatology Association, American Academy of Neurology, American College of Gastroenterology, American Gastroenterological Association, American Kidney Fund, Arthritis Foundation, Association for Clinical Oncology, Cancer Support Community, Coalition of State Rheumatology Organizations, and National Multiple Sclerosis Society.
UHC did not reply to questions in time for publication.
First large-scale payer to try copay accumulator program
Under UHC’s proposed policy, providers will be required to use UHC’s portal to report payment information received from drug manufacturer copay assistance programs that are applied to patients’ cost share of these drugs through a complex, 14-step “coupon submission process” involving multiple technology interfaces. “My first oath as a physician is to do no harm to my patient. Many of us are concerned about making these reports, which could harm our patients and undermine the doctor-patient relationship,” Dr. Phillips said.
“If I don’t report, what happens? I don’t think we know the answer to that. Some of us may decide we need to part ways with UHC.” Others may decline to participate in the drug manufacturers’ coupon programs beyond simply informing patients that manufacturer assistance is available.
“We’ve watched these copay accumulator policies for several years,” he said. “Some of them are rather opaque, with names like ‘copay savings programs’ or ‘copay value programs.’ But we had not seen a large-scale payer try to do this until now. Let’s face it: If UHC’s policy goes through, you can count the days until we see it from others.”
The Department of Health & Human Services, in its May 2020 final federal “Notice of Benefit and Payment Parameters for 2021,” indicated that individual states have the responsibility to regulate copay accumulator programs. Five states have banned them or restricted their use for individual and small group health plans. Arizona, Illinois, Virginia, and West Virginia passed such laws in 2019, and Georgia did so earlier this year.
“In next year’s state legislative sessions, we’ll make it a priority to pursue similar laws in other states,” Dr. Phillips said. “I’d encourage rheumatologists to educate their patients on the issues and be active in advocating for them.”
Last month, the American College of Rheumatology joined with 11 other medical associations and disease societies asking health insurance giant UnitedHealthcare (UHC) to not proceed with its proposed copay accumulator medical benefit program.
Copay accumulators are policies adopted by insurance companies or their pharmacy benefit managers to exclude patient copayment assistance programs for high-cost drugs, which are promulgated by the drug manufacturers, from being applied to a patient’s annual deductibles or out-of-pocket maximums. The manufacturer’s copay assistance, such as in the form of coupons, is designed to minimize the patient’s out-of-pocket costs. But insurers believe manufacturers will have no pressure to lower the prices of expensive specialty drugs unless patients are unable to afford them. Copay accumulators thus are aimed at giving insurers more leverage in negotiating prices for high-cost drugs.
UHC issued its new copay accumulator protocol for commercial individual and fully insured group plans in early October, effective Jan. 1, 2021, “in order to align employer costs for specialty medications with actual member out of pocket and deductibles,” according to the company’s announcement. In other words, patients will need to pay a higher share of the costs of these medications, said rheumatologist Christopher Phillips, MD, who chairs the Insurance Subcommittee of ACR’s Rheumatologic Care Committee. The annual price of biologic therapies for rheumatologic conditions ranges from $22,000 to $44,000, according to a recent press release from ACR.
The copay accumulator will negate the benefits of manufacturers’ copayment assistance programs for the patient, shifting more of the cost to the patient. With patients being forced to pay a higher share of drug costs for expensive biologic treatments for rheumatoid arthritis, lupus, and other rheumatologic conditions, they’ll stop taking the treatments, Dr. Phillips said.
“In my solo rheumatology practice in Paducah, Kentucky, when I’ve seen this kind of program applied on the pharmacy benefit side, rather than the medical benefit side, almost uniformly patients stop taking the high-cost treatments.” That can lead to disease flares, complications, and permanent disability. The newer rheumatologic drugs can cost $500 to $1,000 per treatment, and in many cases, there’s no generic or lower-cost alternative, he says. “We see policies like this as sacrificing patients to the battle over high drug prices. It’s bad practice, bad for patient outcomes, and nobody – apart from the payer – benefits.”
In ACR’s 2020 Rheumatic Disease Patient Survey, nearly half of 1,109 online survey respondents who had rheumatic diseases reported out-of-pocket costs greater than $1,000 per year for treatment. An IQVIA report from 2016 found that one in four specialty brand prescriptions are abandoned during the deductible phase, three times the rate seen when there is no deductible.
In an Oct. 7 letter to UHC, the 12 groups acknowledged that the drugs targeted by the accumulator policy are expensive. “However, they are also vitally important for our patients.” In addition to the ACR, the organizations involved include the AIDS Institute, American Academy of Dermatology Association, American Academy of Neurology, American College of Gastroenterology, American Gastroenterological Association, American Kidney Fund, Arthritis Foundation, Association for Clinical Oncology, Cancer Support Community, Coalition of State Rheumatology Organizations, and National Multiple Sclerosis Society.
UHC did not reply to questions in time for publication.
First large-scale payer to try copay accumulator program
Under UHC’s proposed policy, providers will be required to use UHC’s portal to report payment information received from drug manufacturer copay assistance programs that are applied to patients’ cost share of these drugs through a complex, 14-step “coupon submission process” involving multiple technology interfaces. “My first oath as a physician is to do no harm to my patient. Many of us are concerned about making these reports, which could harm our patients and undermine the doctor-patient relationship,” Dr. Phillips said.
“If I don’t report, what happens? I don’t think we know the answer to that. Some of us may decide we need to part ways with UHC.” Others may decline to participate in the drug manufacturers’ coupon programs beyond simply informing patients that manufacturer assistance is available.
“We’ve watched these copay accumulator policies for several years,” he said. “Some of them are rather opaque, with names like ‘copay savings programs’ or ‘copay value programs.’ But we had not seen a large-scale payer try to do this until now. Let’s face it: If UHC’s policy goes through, you can count the days until we see it from others.”
The Department of Health & Human Services, in its May 2020 final federal “Notice of Benefit and Payment Parameters for 2021,” indicated that individual states have the responsibility to regulate copay accumulator programs. Five states have banned them or restricted their use for individual and small group health plans. Arizona, Illinois, Virginia, and West Virginia passed such laws in 2019, and Georgia did so earlier this year.
“In next year’s state legislative sessions, we’ll make it a priority to pursue similar laws in other states,” Dr. Phillips said. “I’d encourage rheumatologists to educate their patients on the issues and be active in advocating for them.”
Family planning issues loom large for female radiation oncologists
Results from the survey were reported at the American Society for Radiation Oncology Annual Meeting 2020.
“Female radiation oncologists often spend their childbearing years in training and establishing careers,” commented lead investigator Anna Lee, MD, of the University of Texas MD Anderson Cancer Center in Houston.
“Currently, physician fertility and family planning are rarely discussed or taught in medical school or postgraduate training,” Dr. Lee said.
Dr. Lee and colleagues conducted a national anonymous cross-sectional online survey of female oncologists of all types and all career levels (including trainees). The team circulated a 39-item questionnaire exploring attitudes toward and experiences related to family planning and assisted reproductive technology (ART) by email and social media channels.
A total of 351 radiation oncologists participated, representing one-fifth of the specialty’s entire female workforce nationally and making this study the largest to date on family planning among these physicians.
Most respondents were aged 31-40 years (60%) and married (79%), had children (68%), and were in training (26%) or academic practice (48%).
Survey results
Fully 74% of respondents reported that their career plans strongly influenced the timing of when to start a family, and 29% said family planning considerations influenced their decision regarding their choice of academia versus private practice, Dr. Lee reported.
Overall, 24% of respondents indicated that they had difficulty with infertility or required fertility counseling/treatment, 66% said they wished fertility preservation was discussed at some point during their training, and 22% said either that ART would have benefited them if it had been available or that they were planning to or had already used fertility preservation.
On the topic of maternity leave, some respondents reported that their institution either had no formal leave policy during training or provided less than 1 month of leave (23%) and that they felt pressure to take less time off than was policy (15%).
“Of note, 32 women in our survey were not offered non–radiation-exposing assignments during pregnancy, and an additional 57 had to specifically ask for them,” Dr. Lee remarked.
About one-third of respondents each reported that they did not feel supported during training for issues related to fertility and/or pregnancy (33%) and that they experienced discrimination for being pregnant (32%) and taking maternity leave (30%).
“Systemic changes are necessary early in medical education and training to ensure women are supported and able to advance equitably in the field. As less than a third of the current radiation oncology workforce are women, improvement upon these issues will be necessary to draw more women into the field,” Dr. Lee commented. “Education on ART risks, benefits, and success rates can help physicians and those in training in their family planning, while the lack of education and structured policy can exacerbate the emotional, physical, and financial impact of infertility.
“Until recently, there has been a dearth of policy at the programmatic, institutional, and national level allowing time and protection for pregnancy and maternity leave,” she added. “Thankfully, this summer, the American Board of Medical Specialties announced a progressive leave policy for residents and fellows.”
The new policy, which goes into effect July 2021, allows a minimum of 6 weeks away without exhausting time allowed for vacation or sick leave and without requiring an extension in training.
When career and biology collide
“The collision of professional and biological clocks for women in medicine is an important issue highlighted by this study,” Reshma Jagsi, MD, DPhil, of the University of Michigan in Ann Arbor, said in an interview.
“Prior work focused on women in medicine more generally. A deeper dive into the experiences of women in a specific field may be even more compelling to drive change on the part of professional societies and organizations,” Dr. Jagsi added.
The infertility rate observed in the study could have potentially been skewed by the preponderance of younger respondents (resulting in underestimation) or by greater participation of those interested in the subject (resulting in overestimation), she noted. However, it aligns well with the rate in a study Dr. Jagsi and colleagues conducted among female physicians generally using somewhat different methods. That study was published in the Journal of Women’s Health.
Concern about radiation exposure and its potential reproductive health effects should not deter women from choosing radiation oncology as a specialty, according to Dr. Jagsi.
“Radiation exposure is actually very low in radiation oncology, much lower than in specialties like interventional cardiology, where physicians are in the room where fluoroscopy is being used. It is actually an important misconception about this field that merits correction,” she stressed. “Rather, the fertility concerns are related to the expectations of training and demands of work during the prime childbearing years more generally that can lead women to delay pregnancy, which is an issue common to all medical specialties.”
“The investigators’ conclusions are very reasonable,” Dr. Jagsi said. “Although one might quibble whether the exact proportions reflect the experiences of all women in the field perfectly due to the possibility of selection bias, one cannot question whether a substantial number of women are experiencing these challenges and that they merit intervention.”
The study did not receive specific funding. Dr. Lee and Dr. Jagsi disclosed no relevant conflicts of interest.
SOURCE: Lee A et al. ASTRO 2020, Abstract LBA 6.
Results from the survey were reported at the American Society for Radiation Oncology Annual Meeting 2020.
“Female radiation oncologists often spend their childbearing years in training and establishing careers,” commented lead investigator Anna Lee, MD, of the University of Texas MD Anderson Cancer Center in Houston.
“Currently, physician fertility and family planning are rarely discussed or taught in medical school or postgraduate training,” Dr. Lee said.
Dr. Lee and colleagues conducted a national anonymous cross-sectional online survey of female oncologists of all types and all career levels (including trainees). The team circulated a 39-item questionnaire exploring attitudes toward and experiences related to family planning and assisted reproductive technology (ART) by email and social media channels.
A total of 351 radiation oncologists participated, representing one-fifth of the specialty’s entire female workforce nationally and making this study the largest to date on family planning among these physicians.
Most respondents were aged 31-40 years (60%) and married (79%), had children (68%), and were in training (26%) or academic practice (48%).
Survey results
Fully 74% of respondents reported that their career plans strongly influenced the timing of when to start a family, and 29% said family planning considerations influenced their decision regarding their choice of academia versus private practice, Dr. Lee reported.
Overall, 24% of respondents indicated that they had difficulty with infertility or required fertility counseling/treatment, 66% said they wished fertility preservation was discussed at some point during their training, and 22% said either that ART would have benefited them if it had been available or that they were planning to or had already used fertility preservation.
On the topic of maternity leave, some respondents reported that their institution either had no formal leave policy during training or provided less than 1 month of leave (23%) and that they felt pressure to take less time off than was policy (15%).
“Of note, 32 women in our survey were not offered non–radiation-exposing assignments during pregnancy, and an additional 57 had to specifically ask for them,” Dr. Lee remarked.
About one-third of respondents each reported that they did not feel supported during training for issues related to fertility and/or pregnancy (33%) and that they experienced discrimination for being pregnant (32%) and taking maternity leave (30%).
“Systemic changes are necessary early in medical education and training to ensure women are supported and able to advance equitably in the field. As less than a third of the current radiation oncology workforce are women, improvement upon these issues will be necessary to draw more women into the field,” Dr. Lee commented. “Education on ART risks, benefits, and success rates can help physicians and those in training in their family planning, while the lack of education and structured policy can exacerbate the emotional, physical, and financial impact of infertility.
“Until recently, there has been a dearth of policy at the programmatic, institutional, and national level allowing time and protection for pregnancy and maternity leave,” she added. “Thankfully, this summer, the American Board of Medical Specialties announced a progressive leave policy for residents and fellows.”
The new policy, which goes into effect July 2021, allows a minimum of 6 weeks away without exhausting time allowed for vacation or sick leave and without requiring an extension in training.
When career and biology collide
“The collision of professional and biological clocks for women in medicine is an important issue highlighted by this study,” Reshma Jagsi, MD, DPhil, of the University of Michigan in Ann Arbor, said in an interview.
“Prior work focused on women in medicine more generally. A deeper dive into the experiences of women in a specific field may be even more compelling to drive change on the part of professional societies and organizations,” Dr. Jagsi added.
The infertility rate observed in the study could have potentially been skewed by the preponderance of younger respondents (resulting in underestimation) or by greater participation of those interested in the subject (resulting in overestimation), she noted. However, it aligns well with the rate in a study Dr. Jagsi and colleagues conducted among female physicians generally using somewhat different methods. That study was published in the Journal of Women’s Health.
Concern about radiation exposure and its potential reproductive health effects should not deter women from choosing radiation oncology as a specialty, according to Dr. Jagsi.
“Radiation exposure is actually very low in radiation oncology, much lower than in specialties like interventional cardiology, where physicians are in the room where fluoroscopy is being used. It is actually an important misconception about this field that merits correction,” she stressed. “Rather, the fertility concerns are related to the expectations of training and demands of work during the prime childbearing years more generally that can lead women to delay pregnancy, which is an issue common to all medical specialties.”
“The investigators’ conclusions are very reasonable,” Dr. Jagsi said. “Although one might quibble whether the exact proportions reflect the experiences of all women in the field perfectly due to the possibility of selection bias, one cannot question whether a substantial number of women are experiencing these challenges and that they merit intervention.”
The study did not receive specific funding. Dr. Lee and Dr. Jagsi disclosed no relevant conflicts of interest.
SOURCE: Lee A et al. ASTRO 2020, Abstract LBA 6.
Results from the survey were reported at the American Society for Radiation Oncology Annual Meeting 2020.
“Female radiation oncologists often spend their childbearing years in training and establishing careers,” commented lead investigator Anna Lee, MD, of the University of Texas MD Anderson Cancer Center in Houston.
“Currently, physician fertility and family planning are rarely discussed or taught in medical school or postgraduate training,” Dr. Lee said.
Dr. Lee and colleagues conducted a national anonymous cross-sectional online survey of female oncologists of all types and all career levels (including trainees). The team circulated a 39-item questionnaire exploring attitudes toward and experiences related to family planning and assisted reproductive technology (ART) by email and social media channels.
A total of 351 radiation oncologists participated, representing one-fifth of the specialty’s entire female workforce nationally and making this study the largest to date on family planning among these physicians.
Most respondents were aged 31-40 years (60%) and married (79%), had children (68%), and were in training (26%) or academic practice (48%).
Survey results
Fully 74% of respondents reported that their career plans strongly influenced the timing of when to start a family, and 29% said family planning considerations influenced their decision regarding their choice of academia versus private practice, Dr. Lee reported.
Overall, 24% of respondents indicated that they had difficulty with infertility or required fertility counseling/treatment, 66% said they wished fertility preservation was discussed at some point during their training, and 22% said either that ART would have benefited them if it had been available or that they were planning to or had already used fertility preservation.
On the topic of maternity leave, some respondents reported that their institution either had no formal leave policy during training or provided less than 1 month of leave (23%) and that they felt pressure to take less time off than was policy (15%).
“Of note, 32 women in our survey were not offered non–radiation-exposing assignments during pregnancy, and an additional 57 had to specifically ask for them,” Dr. Lee remarked.
About one-third of respondents each reported that they did not feel supported during training for issues related to fertility and/or pregnancy (33%) and that they experienced discrimination for being pregnant (32%) and taking maternity leave (30%).
“Systemic changes are necessary early in medical education and training to ensure women are supported and able to advance equitably in the field. As less than a third of the current radiation oncology workforce are women, improvement upon these issues will be necessary to draw more women into the field,” Dr. Lee commented. “Education on ART risks, benefits, and success rates can help physicians and those in training in their family planning, while the lack of education and structured policy can exacerbate the emotional, physical, and financial impact of infertility.
“Until recently, there has been a dearth of policy at the programmatic, institutional, and national level allowing time and protection for pregnancy and maternity leave,” she added. “Thankfully, this summer, the American Board of Medical Specialties announced a progressive leave policy for residents and fellows.”
The new policy, which goes into effect July 2021, allows a minimum of 6 weeks away without exhausting time allowed for vacation or sick leave and without requiring an extension in training.
When career and biology collide
“The collision of professional and biological clocks for women in medicine is an important issue highlighted by this study,” Reshma Jagsi, MD, DPhil, of the University of Michigan in Ann Arbor, said in an interview.
“Prior work focused on women in medicine more generally. A deeper dive into the experiences of women in a specific field may be even more compelling to drive change on the part of professional societies and organizations,” Dr. Jagsi added.
The infertility rate observed in the study could have potentially been skewed by the preponderance of younger respondents (resulting in underestimation) or by greater participation of those interested in the subject (resulting in overestimation), she noted. However, it aligns well with the rate in a study Dr. Jagsi and colleagues conducted among female physicians generally using somewhat different methods. That study was published in the Journal of Women’s Health.
Concern about radiation exposure and its potential reproductive health effects should not deter women from choosing radiation oncology as a specialty, according to Dr. Jagsi.
“Radiation exposure is actually very low in radiation oncology, much lower than in specialties like interventional cardiology, where physicians are in the room where fluoroscopy is being used. It is actually an important misconception about this field that merits correction,” she stressed. “Rather, the fertility concerns are related to the expectations of training and demands of work during the prime childbearing years more generally that can lead women to delay pregnancy, which is an issue common to all medical specialties.”
“The investigators’ conclusions are very reasonable,” Dr. Jagsi said. “Although one might quibble whether the exact proportions reflect the experiences of all women in the field perfectly due to the possibility of selection bias, one cannot question whether a substantial number of women are experiencing these challenges and that they merit intervention.”
The study did not receive specific funding. Dr. Lee and Dr. Jagsi disclosed no relevant conflicts of interest.
SOURCE: Lee A et al. ASTRO 2020, Abstract LBA 6.
FROM ASTRO 2020
Apps for applying to ObGyn residency programs in the era of virtual interviews
The coronavirus disease 2019 (COVID-19) pandemic has upended the traditional 2020–2021 application season for ObGyn residency programs. In May 2020, the 2 national ObGyn education organizations, the Association of Professors of Gynecology and Obstetrics (APGO) and Council on Resident Education in ObGyn (CREOG), issued guidelines to ensure a fair and equitable application process.1 These guidelines are consistent with recommendations from the Association of American Medical Colleges (AAMC) and the Coalition for Physician Accountability. Important recommendations include:
- limiting away rotations
- being flexible in the number of specialty-specific letters of recommendation required
- encouraging residency programs to develop alternate means of conveying information about their curriculum.
In addition, these statements provide timing on when programs should release interview offers and when to begin interviews. Finally, programs are required to commit to online interviews and virtual visits for all applicants, including local students, rather than in-person interviews.
Here, we focus on identifying apps that students can use to help them with the application process—apps for the nuts and bolts of applying and interviewing and apps to learn more about individual programs.
Students must use the Electronic Residency Application Service (ERAS) platform from AAMC to enter their information and register with the National Resident Matching Program (NRMP). Students also must use the ERAS to submit their applications to their selected residency programs. The ERAS platform does not include an app to aid in the completion or submission of an application. The NRMP has developed the MATCH PRISM app, but this does not allow students to register for the match or submit their rank list. To learn about how to schedule interviews, residency programs may use one of the following sources: ERAS, Interview Broker, or Thalamus. Moreover, APGO/CREOG has partnered with Thalamus for the upcoming application cycle, which provides residency programs and applicants tools for application management, interview scheduling, and itinerary building. Thalamus offers a free app.
This year offers some unique challenges. The application process for ObGyn residencies is likely to be more competitive, and students face the added stress of having to navigate the interview season:
- without away rotations (audition interviews)
- without in-person visits of the city/hospital/program or social events before or after interview day
- with an all-virtual interview day.
Continue to: To find information on individual residency programs...
To find information on individual residency programs, the APGO website lists the FREIDA and APGO Residency Directories, which are not apps. Students are also aware of the Doximity Residency Navigator, which does include an app. The NRMP MATCH PRISM app is another resource, as it provides students with a directory of residency programs and information about each program.
The American College of Obstetricians and Gynecologists (ACOG) recognizes that residency program websites and social media will be crucial in helping applicants learn about individual programs, faculty, and residents. As such, ACOG hosted a Virtual Residency Showcase in September 2020 in which programs posted content on Instagram and Twitter using the hashtag #ACOG-ResWeek20.2 Similarly, APGO and CREOG produced a report containing a social media directory, which lists individual residency programs and whether or not they have a social media handle/account.3 In a recent webinar,4 Drs. Sarah Santiago and Elizabeth Southworth noted that the number of residency programs that have an Instagram account more than doubled (from 60 to 128) between May and September 2020.

We present 2 tables describing the important features and caveats of apps available to students to assist them with residency applications this year—TABLE 1 summarizes apps to aid with applications and interviews; TABLE 2 lists apps designed for students to learn more about individual residency programs. We wish all of this year’s students every success in their search for the right program. ●
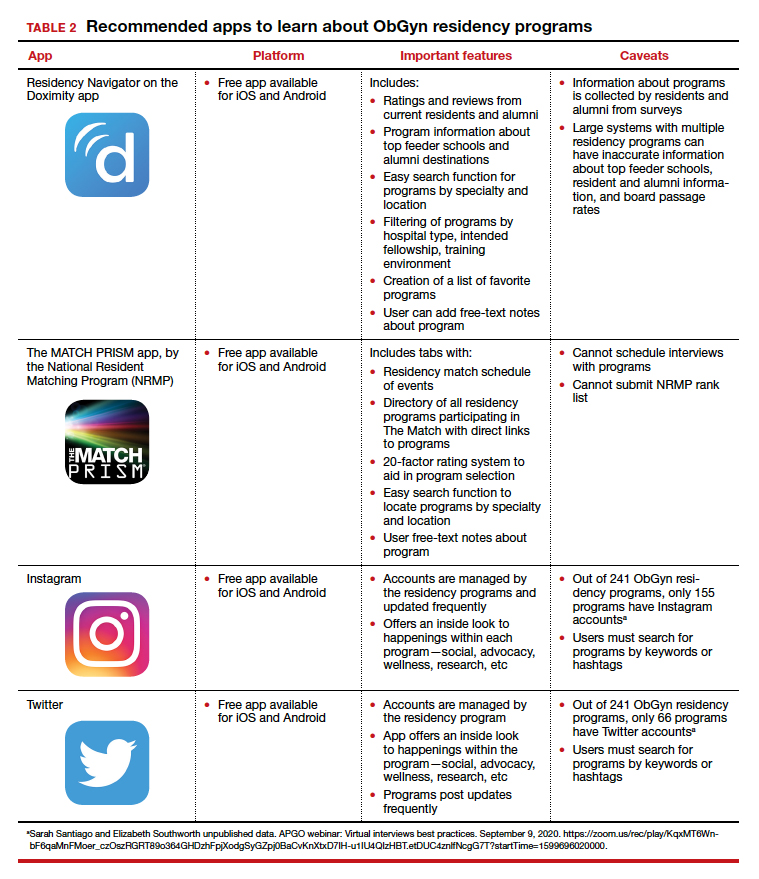
- Association of Professors of Gynecology and Obstetrics, Council on Resident Education in ObGyn. Updated APGO and CREOG Residency Application Response to COVID-19. https://www.apgo.org/wp-content/uploads/2020/05 /Updated-APGO-CREOG-Residency-Response-to -COVID-19-.pdf. Accessed October 27, 2020.
- https://www.acog.org/education-and-events/webinars /virtual-residency-showcase. Accessed October 4, 2020.
- Social media directory-ObGyn. https://docs.google.com /spreadsheets/d/e/2PACX-1vQ6boyn7FWV9tEhfQp1o3 XJgNIPNBQ3qCYf4IpV-rOPcd212J-HNR84p0r85nXrAz MvOmcNlgjywDP/pubhtml?gid=1472916499&single =true. Accessed October 27, 2020.
- APGO webinar: Virtual interviews best practices. September 9, 2020. https://zoom.us/rec/play/KqxMT6Wnb F6qaMnFMoer_czOszRGRT89o364GHDzhFpjXodgSyGZpj 0BaCvKnXtxD7IH-u1IU4QIzHBT.etDUC4znlfNcgG7T?start Time=1599696020000. Accessed October 4, 2020.
The coronavirus disease 2019 (COVID-19) pandemic has upended the traditional 2020–2021 application season for ObGyn residency programs. In May 2020, the 2 national ObGyn education organizations, the Association of Professors of Gynecology and Obstetrics (APGO) and Council on Resident Education in ObGyn (CREOG), issued guidelines to ensure a fair and equitable application process.1 These guidelines are consistent with recommendations from the Association of American Medical Colleges (AAMC) and the Coalition for Physician Accountability. Important recommendations include:
- limiting away rotations
- being flexible in the number of specialty-specific letters of recommendation required
- encouraging residency programs to develop alternate means of conveying information about their curriculum.
In addition, these statements provide timing on when programs should release interview offers and when to begin interviews. Finally, programs are required to commit to online interviews and virtual visits for all applicants, including local students, rather than in-person interviews.
Here, we focus on identifying apps that students can use to help them with the application process—apps for the nuts and bolts of applying and interviewing and apps to learn more about individual programs.
Students must use the Electronic Residency Application Service (ERAS) platform from AAMC to enter their information and register with the National Resident Matching Program (NRMP). Students also must use the ERAS to submit their applications to their selected residency programs. The ERAS platform does not include an app to aid in the completion or submission of an application. The NRMP has developed the MATCH PRISM app, but this does not allow students to register for the match or submit their rank list. To learn about how to schedule interviews, residency programs may use one of the following sources: ERAS, Interview Broker, or Thalamus. Moreover, APGO/CREOG has partnered with Thalamus for the upcoming application cycle, which provides residency programs and applicants tools for application management, interview scheduling, and itinerary building. Thalamus offers a free app.
This year offers some unique challenges. The application process for ObGyn residencies is likely to be more competitive, and students face the added stress of having to navigate the interview season:
- without away rotations (audition interviews)
- without in-person visits of the city/hospital/program or social events before or after interview day
- with an all-virtual interview day.
Continue to: To find information on individual residency programs...
To find information on individual residency programs, the APGO website lists the FREIDA and APGO Residency Directories, which are not apps. Students are also aware of the Doximity Residency Navigator, which does include an app. The NRMP MATCH PRISM app is another resource, as it provides students with a directory of residency programs and information about each program.
The American College of Obstetricians and Gynecologists (ACOG) recognizes that residency program websites and social media will be crucial in helping applicants learn about individual programs, faculty, and residents. As such, ACOG hosted a Virtual Residency Showcase in September 2020 in which programs posted content on Instagram and Twitter using the hashtag #ACOG-ResWeek20.2 Similarly, APGO and CREOG produced a report containing a social media directory, which lists individual residency programs and whether or not they have a social media handle/account.3 In a recent webinar,4 Drs. Sarah Santiago and Elizabeth Southworth noted that the number of residency programs that have an Instagram account more than doubled (from 60 to 128) between May and September 2020.

We present 2 tables describing the important features and caveats of apps available to students to assist them with residency applications this year—TABLE 1 summarizes apps to aid with applications and interviews; TABLE 2 lists apps designed for students to learn more about individual residency programs. We wish all of this year’s students every success in their search for the right program. ●

The coronavirus disease 2019 (COVID-19) pandemic has upended the traditional 2020–2021 application season for ObGyn residency programs. In May 2020, the 2 national ObGyn education organizations, the Association of Professors of Gynecology and Obstetrics (APGO) and Council on Resident Education in ObGyn (CREOG), issued guidelines to ensure a fair and equitable application process.1 These guidelines are consistent with recommendations from the Association of American Medical Colleges (AAMC) and the Coalition for Physician Accountability. Important recommendations include:
- limiting away rotations
- being flexible in the number of specialty-specific letters of recommendation required
- encouraging residency programs to develop alternate means of conveying information about their curriculum.
In addition, these statements provide timing on when programs should release interview offers and when to begin interviews. Finally, programs are required to commit to online interviews and virtual visits for all applicants, including local students, rather than in-person interviews.
Here, we focus on identifying apps that students can use to help them with the application process—apps for the nuts and bolts of applying and interviewing and apps to learn more about individual programs.
Students must use the Electronic Residency Application Service (ERAS) platform from AAMC to enter their information and register with the National Resident Matching Program (NRMP). Students also must use the ERAS to submit their applications to their selected residency programs. The ERAS platform does not include an app to aid in the completion or submission of an application. The NRMP has developed the MATCH PRISM app, but this does not allow students to register for the match or submit their rank list. To learn about how to schedule interviews, residency programs may use one of the following sources: ERAS, Interview Broker, or Thalamus. Moreover, APGO/CREOG has partnered with Thalamus for the upcoming application cycle, which provides residency programs and applicants tools for application management, interview scheduling, and itinerary building. Thalamus offers a free app.
This year offers some unique challenges. The application process for ObGyn residencies is likely to be more competitive, and students face the added stress of having to navigate the interview season:
- without away rotations (audition interviews)
- without in-person visits of the city/hospital/program or social events before or after interview day
- with an all-virtual interview day.
Continue to: To find information on individual residency programs...
To find information on individual residency programs, the APGO website lists the FREIDA and APGO Residency Directories, which are not apps. Students are also aware of the Doximity Residency Navigator, which does include an app. The NRMP MATCH PRISM app is another resource, as it provides students with a directory of residency programs and information about each program.
The American College of Obstetricians and Gynecologists (ACOG) recognizes that residency program websites and social media will be crucial in helping applicants learn about individual programs, faculty, and residents. As such, ACOG hosted a Virtual Residency Showcase in September 2020 in which programs posted content on Instagram and Twitter using the hashtag #ACOG-ResWeek20.2 Similarly, APGO and CREOG produced a report containing a social media directory, which lists individual residency programs and whether or not they have a social media handle/account.3 In a recent webinar,4 Drs. Sarah Santiago and Elizabeth Southworth noted that the number of residency programs that have an Instagram account more than doubled (from 60 to 128) between May and September 2020.

We present 2 tables describing the important features and caveats of apps available to students to assist them with residency applications this year—TABLE 1 summarizes apps to aid with applications and interviews; TABLE 2 lists apps designed for students to learn more about individual residency programs. We wish all of this year’s students every success in their search for the right program. ●

- Association of Professors of Gynecology and Obstetrics, Council on Resident Education in ObGyn. Updated APGO and CREOG Residency Application Response to COVID-19. https://www.apgo.org/wp-content/uploads/2020/05 /Updated-APGO-CREOG-Residency-Response-to -COVID-19-.pdf. Accessed October 27, 2020.
- https://www.acog.org/education-and-events/webinars /virtual-residency-showcase. Accessed October 4, 2020.
- Social media directory-ObGyn. https://docs.google.com /spreadsheets/d/e/2PACX-1vQ6boyn7FWV9tEhfQp1o3 XJgNIPNBQ3qCYf4IpV-rOPcd212J-HNR84p0r85nXrAz MvOmcNlgjywDP/pubhtml?gid=1472916499&single =true. Accessed October 27, 2020.
- APGO webinar: Virtual interviews best practices. September 9, 2020. https://zoom.us/rec/play/KqxMT6Wnb F6qaMnFMoer_czOszRGRT89o364GHDzhFpjXodgSyGZpj 0BaCvKnXtxD7IH-u1IU4QIzHBT.etDUC4znlfNcgG7T?start Time=1599696020000. Accessed October 4, 2020.
- Association of Professors of Gynecology and Obstetrics, Council on Resident Education in ObGyn. Updated APGO and CREOG Residency Application Response to COVID-19. https://www.apgo.org/wp-content/uploads/2020/05 /Updated-APGO-CREOG-Residency-Response-to -COVID-19-.pdf. Accessed October 27, 2020.
- https://www.acog.org/education-and-events/webinars /virtual-residency-showcase. Accessed October 4, 2020.
- Social media directory-ObGyn. https://docs.google.com /spreadsheets/d/e/2PACX-1vQ6boyn7FWV9tEhfQp1o3 XJgNIPNBQ3qCYf4IpV-rOPcd212J-HNR84p0r85nXrAz MvOmcNlgjywDP/pubhtml?gid=1472916499&single =true. Accessed October 27, 2020.
- APGO webinar: Virtual interviews best practices. September 9, 2020. https://zoom.us/rec/play/KqxMT6Wnb F6qaMnFMoer_czOszRGRT89o364GHDzhFpjXodgSyGZpj 0BaCvKnXtxD7IH-u1IU4QIzHBT.etDUC4znlfNcgG7T?start Time=1599696020000. Accessed October 4, 2020.
Understand the legal implications of telehealth medicine
Telehealth has been steadily gaining mainstream use throughout the last decade, but the practice was recently shoved, almost overnight, into the forefront of the health care profession. Telehealth is now used more frequently by medical groups and physicians than ever before. General reports before the COVID-19 pandemic approximated 90% of health care organizations used or planned to use telehealth in the future. This future may already be a reality, with a McKinsey & Company report estimating that physicians saw 50-175 times more patients over telehealth platforms since the pandemic’s start.1
In general, telehealth includes use of electronic communication and information technologies to deliver long-distance or remote health care. A physician’s use of telemedicine (clinical services) is one of the most common uses, but the industry also includes other professionals, such as pharmacists and nurses.
Telehealth platforms can be used to monitor, diagnose, treat, and counsel patients successfully. It works best for reading images, follow-up care, outpatient care, and long-term care. However, telemedicine is inappropriate for urgent issues, diagnosing underlying health conditions, or any practice where the standard of care would require a physical exam. There is potential liability for decision making without a proper physical exam.
There are many advantages to telehealth over more traditional health care options. Some of these advantages include:
- Increased access to health care.
- Increased access to medical specialists in small and rural communities.
- Improved long-term care from the comfort of patients’ homes.
- Improved platforms to document patient care outside regular business hours.
But along with these benefits, telehealth carries the disadvantage of potential increased liability. This increased liability could stem from:
- Breached standards of care.
- Inadequate or improper licensing.
- Limited care options.
- Decision making without a proper physical exam.
- Increased informed consent requirements.
- Restricted prescription access.
Before expanding any practice into telemedicine, awareness of potential legal issues is crucial.
Standard of care
Currently, telehealth laws and regulations vary significantly from state to state. But one rule is consistent across the board – that the standard of care for practicing medicine through telemedicine is identical to the standard of care required for practicing medicine during physical practice. It still requires the appropriate examination, testing, labs, imaging, and consultations that any in-person diagnosis needs. For physicians, it also includes supervising nonphysician clinicians, where state law requires supervision.
The American Telemedicine Association currently determines the primary governing standards and guidelines for telemedicine. These can help physicians understand best practices in meeting the standard of care through telemedicine. The American Gastroenterological Association provides coding guidelines and other resources to help physicians with telehealth and e-visits. Other professional societies, such as the American College of Radiology and the American Academy of Dermatology, offer guidelines specific to their medical specialties’ standards of care. These standards still vary from state to state, so medical professionals must be aware of any differences before treating patients in multiple states.
Licensing
Licensing is one of telemedicine’s most confusing legal issues. All states require a license to practice medicine (traditional or telehealth) within their borders. Without that license, practicing medicine in the state is a crime. On top of being criminal, unlicensed practice can affect insurance, liability, billing, and malpractice coverage. When in a brick-and-mortar clinic, a physician’s confidence in practicing within the licensed jurisdiction is easy. Now, the distinction is not so clear. Patients and physicians no longer have to be in the same room, city, or even state, meaning there could be unknown conflicting laws between the two locations. With rare exceptions, standards of care are based on the patient’s location, not the physician’s location. This increases the risk of practicing without being correctly licensed to higher than ever.
Because licensing is a significant roadblock in providing telemedicine, efforts are underway to make the process simpler and more streamlined. The Federation of State Medical Boards developed the Interstate Medical Licensure Compact (IMLC).2 This can qualify physicians to practice medicine across state lines within the compact so long as they meet specific eligibility requirements. The IMLC creates a fast-track option for physicians to fill out one application and receive licenses from multiple states at once. Currently, the compact includes 32 states, the District of Columbia, and Guam.3
Informed consent
Telemedicine health care still requires informed consent from patients. In fact, in some states, the requirements for care provided through telehealth are actually stricter than requirements for informed consent obtained in person.
Most informed consent laws require physicians to cover the risks and benefits of a recommended course of treatment and all feasible and reasonable material alternatives. On top of this traditional informed consent, physicians must get additional consent to receive care over a telehealth platform. This unique requirement explains what telehealth is, possible risks and expected benefits, and security measures used to protect patient information. States vary regarding when verbal consent is sufficient, and when written consent is required.
Prescriptions
Telemedicine is still a relatively new industry, and few legal opinions specifically address telemedicine malpractice. However, prescribing medication based on telemedicine information is among the few issues the courts have addressed. A 2008 decision found that a physician review of patient questionnaires submitted over the Internet was insufficient to prescribe medication without a physical examination determining patient health.4 This cautious approach stemmed from telehealth’s early concern about the absence of patient-physician relationships and potential online pharmacy abuse. Since this decision, many states require an “in-person” visit with a patient before prescribing medication. The definition of what qualifies as an in-person visit varies from state to state – some still consider the use of real-time, audiovisual conferencing sufficient.
The law is still evolving for prescriptions. Some states don’t allow any prescriptions, while others allow physicians to prescribe their patients’ medications as part of an appropriate treatment plan according to their professional discretion. Almost every state prohibits the prescription of controlled substances based on telemedicine.
Conclusion
Telemedicine is becoming an increasingly significant part of both physician-patient relationships and the broader health care industry. Used appropriately, it can be an incredibly effective method of care for physicians and patients. Physicians should learn the laws governing telemedicine in every state they want to practice and continue to stay current on any changes. The Center for Connected Health Policy offers a report, updated semiannually, to help physicians stay up to date on their state laws. These efforts will help prevent physicians from exposure to liability and medical malpractice claims.
Mr. Hyde is a partner at Younker Hyde Macfarlane, a law firm that focuses on prosecuting medical malpractice claims on behalf of injured patients. Ms. Johnson is an associate attorney with the firm. You can find them at YHMLaw.com.
References
1. Bestsennyy O, Harris A, Rost J. Telehealth: A quarter-trillion-dollar post-COVID-19 reality? Mckinsey & Company, May 29, 2020.
2. FSMB: Draft Interstate Compact for Physician Licensure Nears Completion, 2014.
3. Interstate Medical Licensure Compact: U.S. State Participation in the Compact.
4. See, Low Cost Pharm., Inc. v. Ariz. State Bd. Of Pharm, 2008 Ariz. App. Unpub. LEXIS 790, referencing conclusion of Arizona Medical Board.
Telehealth has been steadily gaining mainstream use throughout the last decade, but the practice was recently shoved, almost overnight, into the forefront of the health care profession. Telehealth is now used more frequently by medical groups and physicians than ever before. General reports before the COVID-19 pandemic approximated 90% of health care organizations used or planned to use telehealth in the future. This future may already be a reality, with a McKinsey & Company report estimating that physicians saw 50-175 times more patients over telehealth platforms since the pandemic’s start.1
In general, telehealth includes use of electronic communication and information technologies to deliver long-distance or remote health care. A physician’s use of telemedicine (clinical services) is one of the most common uses, but the industry also includes other professionals, such as pharmacists and nurses.
Telehealth platforms can be used to monitor, diagnose, treat, and counsel patients successfully. It works best for reading images, follow-up care, outpatient care, and long-term care. However, telemedicine is inappropriate for urgent issues, diagnosing underlying health conditions, or any practice where the standard of care would require a physical exam. There is potential liability for decision making without a proper physical exam.
There are many advantages to telehealth over more traditional health care options. Some of these advantages include:
- Increased access to health care.
- Increased access to medical specialists in small and rural communities.
- Improved long-term care from the comfort of patients’ homes.
- Improved platforms to document patient care outside regular business hours.
But along with these benefits, telehealth carries the disadvantage of potential increased liability. This increased liability could stem from:
- Breached standards of care.
- Inadequate or improper licensing.
- Limited care options.
- Decision making without a proper physical exam.
- Increased informed consent requirements.
- Restricted prescription access.
Before expanding any practice into telemedicine, awareness of potential legal issues is crucial.
Standard of care
Currently, telehealth laws and regulations vary significantly from state to state. But one rule is consistent across the board – that the standard of care for practicing medicine through telemedicine is identical to the standard of care required for practicing medicine during physical practice. It still requires the appropriate examination, testing, labs, imaging, and consultations that any in-person diagnosis needs. For physicians, it also includes supervising nonphysician clinicians, where state law requires supervision.
The American Telemedicine Association currently determines the primary governing standards and guidelines for telemedicine. These can help physicians understand best practices in meeting the standard of care through telemedicine. The American Gastroenterological Association provides coding guidelines and other resources to help physicians with telehealth and e-visits. Other professional societies, such as the American College of Radiology and the American Academy of Dermatology, offer guidelines specific to their medical specialties’ standards of care. These standards still vary from state to state, so medical professionals must be aware of any differences before treating patients in multiple states.
Licensing
Licensing is one of telemedicine’s most confusing legal issues. All states require a license to practice medicine (traditional or telehealth) within their borders. Without that license, practicing medicine in the state is a crime. On top of being criminal, unlicensed practice can affect insurance, liability, billing, and malpractice coverage. When in a brick-and-mortar clinic, a physician’s confidence in practicing within the licensed jurisdiction is easy. Now, the distinction is not so clear. Patients and physicians no longer have to be in the same room, city, or even state, meaning there could be unknown conflicting laws between the two locations. With rare exceptions, standards of care are based on the patient’s location, not the physician’s location. This increases the risk of practicing without being correctly licensed to higher than ever.
Because licensing is a significant roadblock in providing telemedicine, efforts are underway to make the process simpler and more streamlined. The Federation of State Medical Boards developed the Interstate Medical Licensure Compact (IMLC).2 This can qualify physicians to practice medicine across state lines within the compact so long as they meet specific eligibility requirements. The IMLC creates a fast-track option for physicians to fill out one application and receive licenses from multiple states at once. Currently, the compact includes 32 states, the District of Columbia, and Guam.3
Informed consent
Telemedicine health care still requires informed consent from patients. In fact, in some states, the requirements for care provided through telehealth are actually stricter than requirements for informed consent obtained in person.
Most informed consent laws require physicians to cover the risks and benefits of a recommended course of treatment and all feasible and reasonable material alternatives. On top of this traditional informed consent, physicians must get additional consent to receive care over a telehealth platform. This unique requirement explains what telehealth is, possible risks and expected benefits, and security measures used to protect patient information. States vary regarding when verbal consent is sufficient, and when written consent is required.
Prescriptions
Telemedicine is still a relatively new industry, and few legal opinions specifically address telemedicine malpractice. However, prescribing medication based on telemedicine information is among the few issues the courts have addressed. A 2008 decision found that a physician review of patient questionnaires submitted over the Internet was insufficient to prescribe medication without a physical examination determining patient health.4 This cautious approach stemmed from telehealth’s early concern about the absence of patient-physician relationships and potential online pharmacy abuse. Since this decision, many states require an “in-person” visit with a patient before prescribing medication. The definition of what qualifies as an in-person visit varies from state to state – some still consider the use of real-time, audiovisual conferencing sufficient.
The law is still evolving for prescriptions. Some states don’t allow any prescriptions, while others allow physicians to prescribe their patients’ medications as part of an appropriate treatment plan according to their professional discretion. Almost every state prohibits the prescription of controlled substances based on telemedicine.
Conclusion
Telemedicine is becoming an increasingly significant part of both physician-patient relationships and the broader health care industry. Used appropriately, it can be an incredibly effective method of care for physicians and patients. Physicians should learn the laws governing telemedicine in every state they want to practice and continue to stay current on any changes. The Center for Connected Health Policy offers a report, updated semiannually, to help physicians stay up to date on their state laws. These efforts will help prevent physicians from exposure to liability and medical malpractice claims.
Mr. Hyde is a partner at Younker Hyde Macfarlane, a law firm that focuses on prosecuting medical malpractice claims on behalf of injured patients. Ms. Johnson is an associate attorney with the firm. You can find them at YHMLaw.com.
References
1. Bestsennyy O, Harris A, Rost J. Telehealth: A quarter-trillion-dollar post-COVID-19 reality? Mckinsey & Company, May 29, 2020.
2. FSMB: Draft Interstate Compact for Physician Licensure Nears Completion, 2014.
3. Interstate Medical Licensure Compact: U.S. State Participation in the Compact.
4. See, Low Cost Pharm., Inc. v. Ariz. State Bd. Of Pharm, 2008 Ariz. App. Unpub. LEXIS 790, referencing conclusion of Arizona Medical Board.
Telehealth has been steadily gaining mainstream use throughout the last decade, but the practice was recently shoved, almost overnight, into the forefront of the health care profession. Telehealth is now used more frequently by medical groups and physicians than ever before. General reports before the COVID-19 pandemic approximated 90% of health care organizations used or planned to use telehealth in the future. This future may already be a reality, with a McKinsey & Company report estimating that physicians saw 50-175 times more patients over telehealth platforms since the pandemic’s start.1
In general, telehealth includes use of electronic communication and information technologies to deliver long-distance or remote health care. A physician’s use of telemedicine (clinical services) is one of the most common uses, but the industry also includes other professionals, such as pharmacists and nurses.
Telehealth platforms can be used to monitor, diagnose, treat, and counsel patients successfully. It works best for reading images, follow-up care, outpatient care, and long-term care. However, telemedicine is inappropriate for urgent issues, diagnosing underlying health conditions, or any practice where the standard of care would require a physical exam. There is potential liability for decision making without a proper physical exam.
There are many advantages to telehealth over more traditional health care options. Some of these advantages include:
- Increased access to health care.
- Increased access to medical specialists in small and rural communities.
- Improved long-term care from the comfort of patients’ homes.
- Improved platforms to document patient care outside regular business hours.
But along with these benefits, telehealth carries the disadvantage of potential increased liability. This increased liability could stem from:
- Breached standards of care.
- Inadequate or improper licensing.
- Limited care options.
- Decision making without a proper physical exam.
- Increased informed consent requirements.
- Restricted prescription access.
Before expanding any practice into telemedicine, awareness of potential legal issues is crucial.
Standard of care
Currently, telehealth laws and regulations vary significantly from state to state. But one rule is consistent across the board – that the standard of care for practicing medicine through telemedicine is identical to the standard of care required for practicing medicine during physical practice. It still requires the appropriate examination, testing, labs, imaging, and consultations that any in-person diagnosis needs. For physicians, it also includes supervising nonphysician clinicians, where state law requires supervision.
The American Telemedicine Association currently determines the primary governing standards and guidelines for telemedicine. These can help physicians understand best practices in meeting the standard of care through telemedicine. The American Gastroenterological Association provides coding guidelines and other resources to help physicians with telehealth and e-visits. Other professional societies, such as the American College of Radiology and the American Academy of Dermatology, offer guidelines specific to their medical specialties’ standards of care. These standards still vary from state to state, so medical professionals must be aware of any differences before treating patients in multiple states.
Licensing
Licensing is one of telemedicine’s most confusing legal issues. All states require a license to practice medicine (traditional or telehealth) within their borders. Without that license, practicing medicine in the state is a crime. On top of being criminal, unlicensed practice can affect insurance, liability, billing, and malpractice coverage. When in a brick-and-mortar clinic, a physician’s confidence in practicing within the licensed jurisdiction is easy. Now, the distinction is not so clear. Patients and physicians no longer have to be in the same room, city, or even state, meaning there could be unknown conflicting laws between the two locations. With rare exceptions, standards of care are based on the patient’s location, not the physician’s location. This increases the risk of practicing without being correctly licensed to higher than ever.
Because licensing is a significant roadblock in providing telemedicine, efforts are underway to make the process simpler and more streamlined. The Federation of State Medical Boards developed the Interstate Medical Licensure Compact (IMLC).2 This can qualify physicians to practice medicine across state lines within the compact so long as they meet specific eligibility requirements. The IMLC creates a fast-track option for physicians to fill out one application and receive licenses from multiple states at once. Currently, the compact includes 32 states, the District of Columbia, and Guam.3
Informed consent
Telemedicine health care still requires informed consent from patients. In fact, in some states, the requirements for care provided through telehealth are actually stricter than requirements for informed consent obtained in person.
Most informed consent laws require physicians to cover the risks and benefits of a recommended course of treatment and all feasible and reasonable material alternatives. On top of this traditional informed consent, physicians must get additional consent to receive care over a telehealth platform. This unique requirement explains what telehealth is, possible risks and expected benefits, and security measures used to protect patient information. States vary regarding when verbal consent is sufficient, and when written consent is required.
Prescriptions
Telemedicine is still a relatively new industry, and few legal opinions specifically address telemedicine malpractice. However, prescribing medication based on telemedicine information is among the few issues the courts have addressed. A 2008 decision found that a physician review of patient questionnaires submitted over the Internet was insufficient to prescribe medication without a physical examination determining patient health.4 This cautious approach stemmed from telehealth’s early concern about the absence of patient-physician relationships and potential online pharmacy abuse. Since this decision, many states require an “in-person” visit with a patient before prescribing medication. The definition of what qualifies as an in-person visit varies from state to state – some still consider the use of real-time, audiovisual conferencing sufficient.
The law is still evolving for prescriptions. Some states don’t allow any prescriptions, while others allow physicians to prescribe their patients’ medications as part of an appropriate treatment plan according to their professional discretion. Almost every state prohibits the prescription of controlled substances based on telemedicine.
Conclusion
Telemedicine is becoming an increasingly significant part of both physician-patient relationships and the broader health care industry. Used appropriately, it can be an incredibly effective method of care for physicians and patients. Physicians should learn the laws governing telemedicine in every state they want to practice and continue to stay current on any changes. The Center for Connected Health Policy offers a report, updated semiannually, to help physicians stay up to date on their state laws. These efforts will help prevent physicians from exposure to liability and medical malpractice claims.
Mr. Hyde is a partner at Younker Hyde Macfarlane, a law firm that focuses on prosecuting medical malpractice claims on behalf of injured patients. Ms. Johnson is an associate attorney with the firm. You can find them at YHMLaw.com.
References
1. Bestsennyy O, Harris A, Rost J. Telehealth: A quarter-trillion-dollar post-COVID-19 reality? Mckinsey & Company, May 29, 2020.
2. FSMB: Draft Interstate Compact for Physician Licensure Nears Completion, 2014.
3. Interstate Medical Licensure Compact: U.S. State Participation in the Compact.
4. See, Low Cost Pharm., Inc. v. Ariz. State Bd. Of Pharm, 2008 Ariz. App. Unpub. LEXIS 790, referencing conclusion of Arizona Medical Board.














