User login
FDA Approves First Quadrivalent Seasonal Flu Vaccine
The first influenza vaccine that contains a second influenza B strain, in addition to two influenza A strains and one influenza B strain, has been approved for people aged 2 through 49 years, the Food and Drug Administration announced on Feb. 29.
The quadrivalent vaccine is an intranasal vaccine, similar to FluMist, the trivalent influenza vaccine licensed as a seasonal influenza vaccine for individuals aged 2 through 49 years. The quadrivalent vaccine contains two strains of type A influenza (A/H1N1 and A/H3N2) and two type B lineage strains, "which increases the likelihood of adequate protection against circulating influenza B strains," the FDA statement announcing the approval said.
"A vaccine containing the four virus strains most likely to spread and cause illness during the influenza season offers an additional option to aid in influenza prevention efforts," Dr. Karen Midthun, director of the FDA’s Center for Biologics Evaluation and Research, said in the statement. She pointed out that influenza B-related illness "affects children, particularly young and school-aged, more than any other population."
The new vaccine will be marketed as "FluMist Quadrivalent" by MedImmune LLC, which also markets FluMist, the trivalent intranasal influenza vaccine.
The safety and effectiveness of the Flu Mist quadrivalent vaccine is supported by studies of the trivalent formulation conducted in the past, as well as three new studies of the quadrivalent vaccine in about 4,000 children and adults in the United States, which "demonstrated that the immune responses were similar between FluMist Quadrivalent and FluMist," the FDA statement said. Adverse reactions associated with both vaccines were similar. A runny or stuffy nose in children and adults and headache and sore throat in adults were the most common adverse reactions reported, according to the FDA.
The other seasonal influenza vaccines that are licensed by the FDA contain the three strains only. At a meeting of the FDA’s vaccines advisory panel, held the day before the FDA announced the FluMist Quadrivalent approval, representatives of several manufacturers said that they had completed trials of quadrivalent formulations of their trivalent vaccines and had either filed or planned to file for approval with the FDA and anticipated approval in time for the 2012-2013 influenza season.
At that meeting, held annually to recommend the two influenza A and one influenza B strains to be included in the vaccine in the United States for the upcoming season, panelists pointed out how challenging it is every year to choose which B strain to recommend, based on the different B strains that are circulating worldwide, and described choosing the B component as a "gamble" and a "crapshoot."
"Flu B has been a dilemma, and I do look forward to having a quadrivalent so that our crapshoot is not so poor," one of the panel members, Dr. Pedro Piedra, professor in the department of molecular virology and microbiology, Baylor College of Medicine, Houston, said at the meeting.
"I’m really gratified to hear of all the progress that’s been made by the manufacturers in developing the quadrivalent product and I look forward to the end of this annual conversation about which B [strain] should go into the vaccine," said another panelist, Dr. Melinda Wharton, deputy director of the Centers for Disease Control and Prevention’s National Center for Immunization and Respiratory Diseases. "I always feel like we’re having to make a decision that has a pretty high probability of being wrong on these B strains," she added. (FDA panelists are cleared of potential conflicts related to the topic of the meeting.)
At that meeting, the panel recommended that the influenza B strain be replaced with a B/Yamagata lineage strain, a switch from a Victoria lineage B strain that has been in the vaccine for several years. But they were less confident about this recommendation than they were with their recommendations for the two influenza A strains.
During this flu season in the United States, which has started later than usual and with less activity than previous years, there is evidence that influenza B viruses from both lineages have been circulating. At the panel meeting, Dr. Lisa Grohskopf of the CDC’s influenza division, said that of the 397 influenza viruses tested at the CDC this season, 48 (12%) have been influenza B strains. About half of those have been a B/Victoria lineage strains and most of those match the strain in the current influenza vaccine, but the rest have been a Yamagata lineage strain, not included in the current vaccine. But Dr. Grohskopf, who was not a panelist, added that there have not been many B viruses available for testing in the United States.
Click here for prescribing information for FluMist Quadrivalent.
The first influenza vaccine that contains a second influenza B strain, in addition to two influenza A strains and one influenza B strain, has been approved for people aged 2 through 49 years, the Food and Drug Administration announced on Feb. 29.
The quadrivalent vaccine is an intranasal vaccine, similar to FluMist, the trivalent influenza vaccine licensed as a seasonal influenza vaccine for individuals aged 2 through 49 years. The quadrivalent vaccine contains two strains of type A influenza (A/H1N1 and A/H3N2) and two type B lineage strains, "which increases the likelihood of adequate protection against circulating influenza B strains," the FDA statement announcing the approval said.
"A vaccine containing the four virus strains most likely to spread and cause illness during the influenza season offers an additional option to aid in influenza prevention efforts," Dr. Karen Midthun, director of the FDA’s Center for Biologics Evaluation and Research, said in the statement. She pointed out that influenza B-related illness "affects children, particularly young and school-aged, more than any other population."
The new vaccine will be marketed as "FluMist Quadrivalent" by MedImmune LLC, which also markets FluMist, the trivalent intranasal influenza vaccine.
The safety and effectiveness of the Flu Mist quadrivalent vaccine is supported by studies of the trivalent formulation conducted in the past, as well as three new studies of the quadrivalent vaccine in about 4,000 children and adults in the United States, which "demonstrated that the immune responses were similar between FluMist Quadrivalent and FluMist," the FDA statement said. Adverse reactions associated with both vaccines were similar. A runny or stuffy nose in children and adults and headache and sore throat in adults were the most common adverse reactions reported, according to the FDA.
The other seasonal influenza vaccines that are licensed by the FDA contain the three strains only. At a meeting of the FDA’s vaccines advisory panel, held the day before the FDA announced the FluMist Quadrivalent approval, representatives of several manufacturers said that they had completed trials of quadrivalent formulations of their trivalent vaccines and had either filed or planned to file for approval with the FDA and anticipated approval in time for the 2012-2013 influenza season.
At that meeting, held annually to recommend the two influenza A and one influenza B strains to be included in the vaccine in the United States for the upcoming season, panelists pointed out how challenging it is every year to choose which B strain to recommend, based on the different B strains that are circulating worldwide, and described choosing the B component as a "gamble" and a "crapshoot."
"Flu B has been a dilemma, and I do look forward to having a quadrivalent so that our crapshoot is not so poor," one of the panel members, Dr. Pedro Piedra, professor in the department of molecular virology and microbiology, Baylor College of Medicine, Houston, said at the meeting.
"I’m really gratified to hear of all the progress that’s been made by the manufacturers in developing the quadrivalent product and I look forward to the end of this annual conversation about which B [strain] should go into the vaccine," said another panelist, Dr. Melinda Wharton, deputy director of the Centers for Disease Control and Prevention’s National Center for Immunization and Respiratory Diseases. "I always feel like we’re having to make a decision that has a pretty high probability of being wrong on these B strains," she added. (FDA panelists are cleared of potential conflicts related to the topic of the meeting.)
At that meeting, the panel recommended that the influenza B strain be replaced with a B/Yamagata lineage strain, a switch from a Victoria lineage B strain that has been in the vaccine for several years. But they were less confident about this recommendation than they were with their recommendations for the two influenza A strains.
During this flu season in the United States, which has started later than usual and with less activity than previous years, there is evidence that influenza B viruses from both lineages have been circulating. At the panel meeting, Dr. Lisa Grohskopf of the CDC’s influenza division, said that of the 397 influenza viruses tested at the CDC this season, 48 (12%) have been influenza B strains. About half of those have been a B/Victoria lineage strains and most of those match the strain in the current influenza vaccine, but the rest have been a Yamagata lineage strain, not included in the current vaccine. But Dr. Grohskopf, who was not a panelist, added that there have not been many B viruses available for testing in the United States.
Click here for prescribing information for FluMist Quadrivalent.
The first influenza vaccine that contains a second influenza B strain, in addition to two influenza A strains and one influenza B strain, has been approved for people aged 2 through 49 years, the Food and Drug Administration announced on Feb. 29.
The quadrivalent vaccine is an intranasal vaccine, similar to FluMist, the trivalent influenza vaccine licensed as a seasonal influenza vaccine for individuals aged 2 through 49 years. The quadrivalent vaccine contains two strains of type A influenza (A/H1N1 and A/H3N2) and two type B lineage strains, "which increases the likelihood of adequate protection against circulating influenza B strains," the FDA statement announcing the approval said.
"A vaccine containing the four virus strains most likely to spread and cause illness during the influenza season offers an additional option to aid in influenza prevention efforts," Dr. Karen Midthun, director of the FDA’s Center for Biologics Evaluation and Research, said in the statement. She pointed out that influenza B-related illness "affects children, particularly young and school-aged, more than any other population."
The new vaccine will be marketed as "FluMist Quadrivalent" by MedImmune LLC, which also markets FluMist, the trivalent intranasal influenza vaccine.
The safety and effectiveness of the Flu Mist quadrivalent vaccine is supported by studies of the trivalent formulation conducted in the past, as well as three new studies of the quadrivalent vaccine in about 4,000 children and adults in the United States, which "demonstrated that the immune responses were similar between FluMist Quadrivalent and FluMist," the FDA statement said. Adverse reactions associated with both vaccines were similar. A runny or stuffy nose in children and adults and headache and sore throat in adults were the most common adverse reactions reported, according to the FDA.
The other seasonal influenza vaccines that are licensed by the FDA contain the three strains only. At a meeting of the FDA’s vaccines advisory panel, held the day before the FDA announced the FluMist Quadrivalent approval, representatives of several manufacturers said that they had completed trials of quadrivalent formulations of their trivalent vaccines and had either filed or planned to file for approval with the FDA and anticipated approval in time for the 2012-2013 influenza season.
At that meeting, held annually to recommend the two influenza A and one influenza B strains to be included in the vaccine in the United States for the upcoming season, panelists pointed out how challenging it is every year to choose which B strain to recommend, based on the different B strains that are circulating worldwide, and described choosing the B component as a "gamble" and a "crapshoot."
"Flu B has been a dilemma, and I do look forward to having a quadrivalent so that our crapshoot is not so poor," one of the panel members, Dr. Pedro Piedra, professor in the department of molecular virology and microbiology, Baylor College of Medicine, Houston, said at the meeting.
"I’m really gratified to hear of all the progress that’s been made by the manufacturers in developing the quadrivalent product and I look forward to the end of this annual conversation about which B [strain] should go into the vaccine," said another panelist, Dr. Melinda Wharton, deputy director of the Centers for Disease Control and Prevention’s National Center for Immunization and Respiratory Diseases. "I always feel like we’re having to make a decision that has a pretty high probability of being wrong on these B strains," she added. (FDA panelists are cleared of potential conflicts related to the topic of the meeting.)
At that meeting, the panel recommended that the influenza B strain be replaced with a B/Yamagata lineage strain, a switch from a Victoria lineage B strain that has been in the vaccine for several years. But they were less confident about this recommendation than they were with their recommendations for the two influenza A strains.
During this flu season in the United States, which has started later than usual and with less activity than previous years, there is evidence that influenza B viruses from both lineages have been circulating. At the panel meeting, Dr. Lisa Grohskopf of the CDC’s influenza division, said that of the 397 influenza viruses tested at the CDC this season, 48 (12%) have been influenza B strains. About half of those have been a B/Victoria lineage strains and most of those match the strain in the current influenza vaccine, but the rest have been a Yamagata lineage strain, not included in the current vaccine. But Dr. Grohskopf, who was not a panelist, added that there have not been many B viruses available for testing in the United States.
Click here for prescribing information for FluMist Quadrivalent.
Survivorship: Creating partnerships for survival
The projected increase in the number of cancer survivors will present unprecedented challenges for community-based practices, and the Institute of Medicine1 and the American College of Surgeons’ Commission on Cancer2 have made recommendations to meet the needs of this growing patient population. However, most community-based practices do not have the resources to implement the recommendations and will have to work closely with resources outside of the practice to develop financially viable and sustainable programs...
*For a PDF of the full article, click on the link to the left of this introduction.
The projected increase in the number of cancer survivors will present unprecedented challenges for community-based practices, and the Institute of Medicine1 and the American College of Surgeons’ Commission on Cancer2 have made recommendations to meet the needs of this growing patient population. However, most community-based practices do not have the resources to implement the recommendations and will have to work closely with resources outside of the practice to develop financially viable and sustainable programs...
*For a PDF of the full article, click on the link to the left of this introduction.
The projected increase in the number of cancer survivors will present unprecedented challenges for community-based practices, and the Institute of Medicine1 and the American College of Surgeons’ Commission on Cancer2 have made recommendations to meet the needs of this growing patient population. However, most community-based practices do not have the resources to implement the recommendations and will have to work closely with resources outside of the practice to develop financially viable and sustainable programs...
*For a PDF of the full article, click on the link to the left of this introduction.
'Early, Proactive' Management of Chemo Toxicites Improves QOL
Skin conditions in cancer patients can lead to a decreased quality of life because of the psychosocial impact, financial burden, poor health, and disruption of anticancer treatments, said Dr. Mario E. Lacouture at the annual Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
Clinical trials are underway to address management, cost, and quality of life, but in the meantime, "an early and proactive approach toward toxicities is advisable," he said.
Dr. Lacouture, a dermatologist at Memorial Sloan-Kettering Cancer Center in New York, discussed common cancer-associated dermatology at the seminar.
• Rashes. A RAF inhibitor–induced rash can occur within 1-2 weeks of starting treatment, Dr. Lacouture noted. This rash is associated with sensations of burning or itching in photo-exposed areas. In most cases, the rash can be managed with topical steroids and oral GABA agonists. Vemurafenib use also has been associated with rashes, as well as with photosensitivity, itching, and alopecia, he said.
In addition, mTOR inhibitor–induced rashes involving erythematous pruritic papules and ulcerlike lesions have been observed in patients on everolimus and temsirolimus.
• Skin cancers. Data from several studies suggest that the incidence of squamous cell carcinomas in patients on RAF inhibitors is 7%, and the incidence of actinic keratoses in these patients is 4%, according to Dr. Lacouture. These conditions usually develop after approximately 6 months of treatment. They can be managed by surgery or destruction; and there have been no reports of metastases.
• Hand-foot syndrome. Hand-Foot Syndrome (HFSR) is a common side effect in cancer patients that usually occurs within 45 days of starting treatment, said Dr. Lacouture. HFSR is often associated with the use of sorafenib, sunitinib, and pazopanib, and the histology of these patients shows epidermal necrosis, he noted. Patients who develop HFSR may need to temporarily reduce their medications. Additional management of HFSR includes avoiding excess pressure on the affected areas and keeping them cool and moist to reduce patient discomfort.
• Hair and nail changes. Changes in hair can occur in patients being treated with epidermal growth factor receptor inhibitors (EGFRIs), including a slower hair growth rate and alopecia, Dr. Lacouture said. Changes in hair texture such as increased brittleness and hair curling also have been observed, he said. Other hair-related adverse events can include hirsutism on the face and eyelash trichomegaly. In addition, paronychia has been observed in patients on EGFRIs for more than 6 months.
• Radiation dermatitis. Approximately 50% of cancer patients develop radiation dermatitis, including 87% of breast cancer patients, reported Dr. Lacouture. However, the treatment of radiation dermatitis remains challenging, he said. In several studies, patients who used a nonsteroidal topical, including aloe vera and trolamine, showed no significant improvement in radiation dermatitis, compared with controls. However, studies are ongoing, and significant improvements in radiation dermatitis in breast cancer patients have been seen with topical corticosteroids including mometasone, beclomethasone, and betamethasone.
Dr. Lacouture disclosed financial relationships with multiple companies including Amgen, Bristol-Myers Squibb, Genentech, GlaxoSmithKline, Merck, and Roche. SDEF and this news organization are owned by Elsevier.
Skin conditions in cancer patients can lead to a decreased quality of life because of the psychosocial impact, financial burden, poor health, and disruption of anticancer treatments, said Dr. Mario E. Lacouture at the annual Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
Clinical trials are underway to address management, cost, and quality of life, but in the meantime, "an early and proactive approach toward toxicities is advisable," he said.
Dr. Lacouture, a dermatologist at Memorial Sloan-Kettering Cancer Center in New York, discussed common cancer-associated dermatology at the seminar.
• Rashes. A RAF inhibitor–induced rash can occur within 1-2 weeks of starting treatment, Dr. Lacouture noted. This rash is associated with sensations of burning or itching in photo-exposed areas. In most cases, the rash can be managed with topical steroids and oral GABA agonists. Vemurafenib use also has been associated with rashes, as well as with photosensitivity, itching, and alopecia, he said.
In addition, mTOR inhibitor–induced rashes involving erythematous pruritic papules and ulcerlike lesions have been observed in patients on everolimus and temsirolimus.
• Skin cancers. Data from several studies suggest that the incidence of squamous cell carcinomas in patients on RAF inhibitors is 7%, and the incidence of actinic keratoses in these patients is 4%, according to Dr. Lacouture. These conditions usually develop after approximately 6 months of treatment. They can be managed by surgery or destruction; and there have been no reports of metastases.
• Hand-foot syndrome. Hand-Foot Syndrome (HFSR) is a common side effect in cancer patients that usually occurs within 45 days of starting treatment, said Dr. Lacouture. HFSR is often associated with the use of sorafenib, sunitinib, and pazopanib, and the histology of these patients shows epidermal necrosis, he noted. Patients who develop HFSR may need to temporarily reduce their medications. Additional management of HFSR includes avoiding excess pressure on the affected areas and keeping them cool and moist to reduce patient discomfort.
• Hair and nail changes. Changes in hair can occur in patients being treated with epidermal growth factor receptor inhibitors (EGFRIs), including a slower hair growth rate and alopecia, Dr. Lacouture said. Changes in hair texture such as increased brittleness and hair curling also have been observed, he said. Other hair-related adverse events can include hirsutism on the face and eyelash trichomegaly. In addition, paronychia has been observed in patients on EGFRIs for more than 6 months.
• Radiation dermatitis. Approximately 50% of cancer patients develop radiation dermatitis, including 87% of breast cancer patients, reported Dr. Lacouture. However, the treatment of radiation dermatitis remains challenging, he said. In several studies, patients who used a nonsteroidal topical, including aloe vera and trolamine, showed no significant improvement in radiation dermatitis, compared with controls. However, studies are ongoing, and significant improvements in radiation dermatitis in breast cancer patients have been seen with topical corticosteroids including mometasone, beclomethasone, and betamethasone.
Dr. Lacouture disclosed financial relationships with multiple companies including Amgen, Bristol-Myers Squibb, Genentech, GlaxoSmithKline, Merck, and Roche. SDEF and this news organization are owned by Elsevier.
Skin conditions in cancer patients can lead to a decreased quality of life because of the psychosocial impact, financial burden, poor health, and disruption of anticancer treatments, said Dr. Mario E. Lacouture at the annual Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
Clinical trials are underway to address management, cost, and quality of life, but in the meantime, "an early and proactive approach toward toxicities is advisable," he said.
Dr. Lacouture, a dermatologist at Memorial Sloan-Kettering Cancer Center in New York, discussed common cancer-associated dermatology at the seminar.
• Rashes. A RAF inhibitor–induced rash can occur within 1-2 weeks of starting treatment, Dr. Lacouture noted. This rash is associated with sensations of burning or itching in photo-exposed areas. In most cases, the rash can be managed with topical steroids and oral GABA agonists. Vemurafenib use also has been associated with rashes, as well as with photosensitivity, itching, and alopecia, he said.
In addition, mTOR inhibitor–induced rashes involving erythematous pruritic papules and ulcerlike lesions have been observed in patients on everolimus and temsirolimus.
• Skin cancers. Data from several studies suggest that the incidence of squamous cell carcinomas in patients on RAF inhibitors is 7%, and the incidence of actinic keratoses in these patients is 4%, according to Dr. Lacouture. These conditions usually develop after approximately 6 months of treatment. They can be managed by surgery or destruction; and there have been no reports of metastases.
• Hand-foot syndrome. Hand-Foot Syndrome (HFSR) is a common side effect in cancer patients that usually occurs within 45 days of starting treatment, said Dr. Lacouture. HFSR is often associated with the use of sorafenib, sunitinib, and pazopanib, and the histology of these patients shows epidermal necrosis, he noted. Patients who develop HFSR may need to temporarily reduce their medications. Additional management of HFSR includes avoiding excess pressure on the affected areas and keeping them cool and moist to reduce patient discomfort.
• Hair and nail changes. Changes in hair can occur in patients being treated with epidermal growth factor receptor inhibitors (EGFRIs), including a slower hair growth rate and alopecia, Dr. Lacouture said. Changes in hair texture such as increased brittleness and hair curling also have been observed, he said. Other hair-related adverse events can include hirsutism on the face and eyelash trichomegaly. In addition, paronychia has been observed in patients on EGFRIs for more than 6 months.
• Radiation dermatitis. Approximately 50% of cancer patients develop radiation dermatitis, including 87% of breast cancer patients, reported Dr. Lacouture. However, the treatment of radiation dermatitis remains challenging, he said. In several studies, patients who used a nonsteroidal topical, including aloe vera and trolamine, showed no significant improvement in radiation dermatitis, compared with controls. However, studies are ongoing, and significant improvements in radiation dermatitis in breast cancer patients have been seen with topical corticosteroids including mometasone, beclomethasone, and betamethasone.
Dr. Lacouture disclosed financial relationships with multiple companies including Amgen, Bristol-Myers Squibb, Genentech, GlaxoSmithKline, Merck, and Roche. SDEF and this news organization are owned by Elsevier.
EXPERT ANALYSIS FROM THE SDEF HAWAII DERMATOLOGY SEMINAR
Infrared Imaging Eyes Mucositis After Head & Neck Radiation
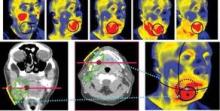
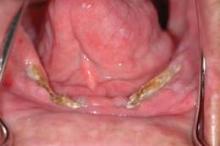
PHOENIX – Thermal imaging was able to detect small and early changes in the temperature of mucosal surfaces – a possible predictor of the development of mucositis – among 34 patients who were treated with chemoradiotherapy for locally advanced squamous cell carcinoma of the head and neck.
"Detection of these early changes using sensitive thermal imaging technology would allow identification of patients who will require more intensive supportive care," said Dr. Ezra Cohen, who presented the pilot study at the head and neck cancer symposium sponsored by the American Society for Radiation Oncology.
Radiotherapy-associated mucositis manifests initially as erythematous areas in the treatment field, which are accompanied by an intense inflammatory response histologically. Thus, Dr. Cohen and his associates had hypothesized that patients destined to display severe mucocutaneous toxicity would demonstrate greater alterations in thermal intensity early in therapy, compared with identically treated counterparts who do not subsequently develop the toxicity.
The researchers further hypothesized that they could measure those changes with infrared thermal imaging, a noninvasive technique that allows visualization and quantification of changes in skin or mucosal surface temperature.
The 34 patients (28 male, mean age 58 years) in the pilot study were treated with identical chemoradiotherapy regimens of 5-fluorouracil and hydroxyurea with a median radiation dose of 74 Gy for cancers of the oral cavity or oropharynx.
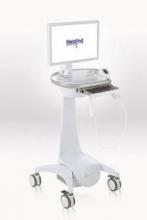
Using a portable device developed at Argonne National Laboratory in Illinois, the investigators conducted noninvasive baseline and weekly thermal imaging. The device detects infrared light naturally emitted from the skin or mucosal surface and generates an electrical signal, which is amplified and converted into digital data flow that is visualized in color on a monitor.
Grade 3 mucositis based on the National Cancer Institute’s Common Terminology Criteria for Adverse Events v3.0 was observed in 53% of the patients, and dermatitis in 21%. All patients displayed an increase in temperature within the radiation field.
Investigators charted a statistically significant positive association between an early rise in temperature in oral mucous membranes when compared with a reference area and mucositis grade (P = .03). For every 1 C increase in temperature, compared with the reference (temperature near the medial angle of one eye), there was a 0.157 increase in average subsequent mucositis grade, reported Dr. Cohen, codirector of the head and neck cancer program at the University of Chicago.
Mucositis and its clinical sequelae are consistently reported as the most clinically significant acute toxicity in the treatment of locally advanced squamous cell carcinoma of the head and neck with chemoradiotherapy, according to Dr. Cohen, who also chaired the meeting. Patient to patient variability in mucositis is related to radiotherapy dosing, fractionation, and volumes, but there also appear to be individual differences in "normal tissue tolerance," even among patients on the same treatment regimen, he said.
"Larger studies with greater dynamic ranges in mucositis scoring are warranted to evaluate whether this tool can help predict which patients would be in need of early intervention to prevent acute complications," he said.
The study was funded by the National Institutes of Health and the University of Chicago Comprehensive Cancer Center. Dr. Cohen and his associates said they had no relevant financial disclosures.
PHOENIX – Thermal imaging was able to detect small and early changes in the temperature of mucosal surfaces – a possible predictor of the development of mucositis – among 34 patients who were treated with chemoradiotherapy for locally advanced squamous cell carcinoma of the head and neck.
"Detection of these early changes using sensitive thermal imaging technology would allow identification of patients who will require more intensive supportive care," said Dr. Ezra Cohen, who presented the pilot study at the head and neck cancer symposium sponsored by the American Society for Radiation Oncology.
Radiotherapy-associated mucositis manifests initially as erythematous areas in the treatment field, which are accompanied by an intense inflammatory response histologically. Thus, Dr. Cohen and his associates had hypothesized that patients destined to display severe mucocutaneous toxicity would demonstrate greater alterations in thermal intensity early in therapy, compared with identically treated counterparts who do not subsequently develop the toxicity.
The researchers further hypothesized that they could measure those changes with infrared thermal imaging, a noninvasive technique that allows visualization and quantification of changes in skin or mucosal surface temperature.
The 34 patients (28 male, mean age 58 years) in the pilot study were treated with identical chemoradiotherapy regimens of 5-fluorouracil and hydroxyurea with a median radiation dose of 74 Gy for cancers of the oral cavity or oropharynx.
Using a portable device developed at Argonne National Laboratory in Illinois, the investigators conducted noninvasive baseline and weekly thermal imaging. The device detects infrared light naturally emitted from the skin or mucosal surface and generates an electrical signal, which is amplified and converted into digital data flow that is visualized in color on a monitor.
Grade 3 mucositis based on the National Cancer Institute’s Common Terminology Criteria for Adverse Events v3.0 was observed in 53% of the patients, and dermatitis in 21%. All patients displayed an increase in temperature within the radiation field.
Investigators charted a statistically significant positive association between an early rise in temperature in oral mucous membranes when compared with a reference area and mucositis grade (P = .03). For every 1 C increase in temperature, compared with the reference (temperature near the medial angle of one eye), there was a 0.157 increase in average subsequent mucositis grade, reported Dr. Cohen, codirector of the head and neck cancer program at the University of Chicago.
Mucositis and its clinical sequelae are consistently reported as the most clinically significant acute toxicity in the treatment of locally advanced squamous cell carcinoma of the head and neck with chemoradiotherapy, according to Dr. Cohen, who also chaired the meeting. Patient to patient variability in mucositis is related to radiotherapy dosing, fractionation, and volumes, but there also appear to be individual differences in "normal tissue tolerance," even among patients on the same treatment regimen, he said.
"Larger studies with greater dynamic ranges in mucositis scoring are warranted to evaluate whether this tool can help predict which patients would be in need of early intervention to prevent acute complications," he said.
The study was funded by the National Institutes of Health and the University of Chicago Comprehensive Cancer Center. Dr. Cohen and his associates said they had no relevant financial disclosures.
PHOENIX – Thermal imaging was able to detect small and early changes in the temperature of mucosal surfaces – a possible predictor of the development of mucositis – among 34 patients who were treated with chemoradiotherapy for locally advanced squamous cell carcinoma of the head and neck.
"Detection of these early changes using sensitive thermal imaging technology would allow identification of patients who will require more intensive supportive care," said Dr. Ezra Cohen, who presented the pilot study at the head and neck cancer symposium sponsored by the American Society for Radiation Oncology.
Radiotherapy-associated mucositis manifests initially as erythematous areas in the treatment field, which are accompanied by an intense inflammatory response histologically. Thus, Dr. Cohen and his associates had hypothesized that patients destined to display severe mucocutaneous toxicity would demonstrate greater alterations in thermal intensity early in therapy, compared with identically treated counterparts who do not subsequently develop the toxicity.
The researchers further hypothesized that they could measure those changes with infrared thermal imaging, a noninvasive technique that allows visualization and quantification of changes in skin or mucosal surface temperature.
The 34 patients (28 male, mean age 58 years) in the pilot study were treated with identical chemoradiotherapy regimens of 5-fluorouracil and hydroxyurea with a median radiation dose of 74 Gy for cancers of the oral cavity or oropharynx.
Using a portable device developed at Argonne National Laboratory in Illinois, the investigators conducted noninvasive baseline and weekly thermal imaging. The device detects infrared light naturally emitted from the skin or mucosal surface and generates an electrical signal, which is amplified and converted into digital data flow that is visualized in color on a monitor.
Grade 3 mucositis based on the National Cancer Institute’s Common Terminology Criteria for Adverse Events v3.0 was observed in 53% of the patients, and dermatitis in 21%. All patients displayed an increase in temperature within the radiation field.
Investigators charted a statistically significant positive association between an early rise in temperature in oral mucous membranes when compared with a reference area and mucositis grade (P = .03). For every 1 C increase in temperature, compared with the reference (temperature near the medial angle of one eye), there was a 0.157 increase in average subsequent mucositis grade, reported Dr. Cohen, codirector of the head and neck cancer program at the University of Chicago.
Mucositis and its clinical sequelae are consistently reported as the most clinically significant acute toxicity in the treatment of locally advanced squamous cell carcinoma of the head and neck with chemoradiotherapy, according to Dr. Cohen, who also chaired the meeting. Patient to patient variability in mucositis is related to radiotherapy dosing, fractionation, and volumes, but there also appear to be individual differences in "normal tissue tolerance," even among patients on the same treatment regimen, he said.
"Larger studies with greater dynamic ranges in mucositis scoring are warranted to evaluate whether this tool can help predict which patients would be in need of early intervention to prevent acute complications," he said.
The study was funded by the National Institutes of Health and the University of Chicago Comprehensive Cancer Center. Dr. Cohen and his associates said they had no relevant financial disclosures.
FROM A HEAD AND NECK CANCER SYMPOSIUM SPONSORED BY THE AMERICAN SOCIETY FOR RADIATION ONCOLOGY
Major Finding: For every 1 C increase in temperature, compared with the reference (temperature near the medial angle of one eye), there was a 0.157 increase in average subsequent mucositis.
Data Source: Data come from a pilot study of functional infrared imaging in 34 patients with locally advanced head and neck cancer who were treated with chemoradiotherapy.
Disclosures: The study was funded by the National Institutes of Health and the University of Chicago Comprehensive Cancer Center. Dr. Cohen and his associates said they had no relevant financial disclosures.
New Vemurafenib Data Highlight Long-Term Melanoma Survival
A phase-II clinical trial with the longest follow-up to date confirmed that vemurafenib induces a clinical response in more than half of patients with advanced melanoma expressing BRAFV600 mutations, according to a report in the Feb. 23 issue of the New England Journal of Medicine.
Median overall survival was 16 months in these patients, who had highly unfavorable characteristics at baseline. With follow-up as long as 20 months, this study "provides critical information on long-term survival" that was not available from previous studies of vemurafenib (Zelboraf) in metastatic melanoma, said Dr. Jeffrey A. Sosman of the Vanderbilt-Ingram Cancer Center, Nashville, Tenn., and his associates.
The Food and Drug Administration approved vemurafenib on Aug. 17, 2011 for patients with unresectable or metastatic melanoma with the BRAFV600E mutation as detected by an FDA-approved test. The FDA acted after a pivotal phase-III trial showed vemurafenib improved overall survival compared with dacarbazine. When the phase-III data were analyzed, median follow-up was just 6.2 months in the experimental arm, and median overall survival had not been reached for patients treated with vemurafenib.
The FDA also considered the currently reported phase-II trial, which included 132 patients with stage-IV melanoma that had progressed despite at least one systemic treatment. Tumor tissue from 56% of patients screened for the trial had a BRAFV600 mutation, which was required for study entry. Vemurafenib is a potent kinase inhibitor that was developed to target this mutation within cancer cells specifically.
Patients in the study were given oral vemurafenib (960 mg twice daily) and followed every 6 weeks for disease progression at 10 medical centers in the United States and 3 in Australia. Median follow-up for efficacy was 13 months (range, 0.6-20 months). Treatment response was assessed by both the investigative team and by a blinded independent review committee.
The committee found that 8 patients (6%) showed a complete response and 62 (47%) showed a partial response to vemurafenib, for an overall response rate of 53%. A total of 38 patients (29%) showed stable disease. Only 18 (14%) showed progressive disease; 6 patients could not be assessed.
Dr. Sosman and his colleagues found a 5% complete response rate and a 52% partial response rate, for an overall response rate of 57%, which generally accorded with the independent review committee’s findings.
Molecular sequencing showed 122 patients in the trial had BRAFV600E mutations and 10 patients had BRAFV600K mutations. Four of the 10 patients with the BRAFV600K mutation had a partial response and 3 had stable disease.
These response rates are higher than those associated with any previous treatments for advanced melanoma, the researchers noted. The lowest response occurred in patients with an LDL level more than 1.5 times the upper limit of normal.
The median duration of response was 6.7 months, and one-fourth of the subjects remained progression free for more than a year. Median overall survival was 16 months. At the time of the data cut-off (July 1, 2011), 62 patients (47% of the study population) were alive.
"Most objective responses were evident at the time of the first set of scans (week 6), but in some patients, responses did not appear until the patient had been receiving the drug for more than 6 months," the researchers said (N. Engl. J. Med. 2012;366:707-14).
The longer follow-up in this study thus permitted the investigators to observe these late responses.
Overall survival was 77% at 6 months and 58% at 12 months, and it was estimated to be 43% at 18 months, they noted.
Until now, median overall of metastatic melanoma has been 6-10 months, Dr. Sosman and his colleagues noted. In previously untreated patients who responded to ipilimumab (Yervoy) – an immunotherapy also approved last year – it reached 11 months.
Most patients had at least one adverse event related to vemurafenib, but most were not severe. Adverse effects chiefly were arthralgia, rash, photosensitivity, fatigue, and alopecia. Although many patients required dose interruptions (64%) or reductions (45%), "patients were able to receive most of their intended daily dose," the investigators wrote.
Four patients discontinued the study drug altogether, including one who developed a retinal-vein occlusion. Another patient died from rapidly progressive melanoma and renal failure that may have been related to the study drug.
Vemurafenib is known to have the paradoxical effect of promoting cutaneous squamous cell carcinomas or keratoacanthomas, so the study subjects were closely monitored for these. Thirty-four patients (26%) developed such neoplasms, usually at the start of therapy. All were managed with resection and vemurafenib was not discontinued. Another eight patients were found to have basal cell carcinomas.
"As with most targeted therapies that block a driver oncogene," vemurafenib will likely encounter resistance with long-term use, Dr. Sosman and his colleagues said. The mechanisms of resistance to vemurafenib are currently being investigated, so that strategies to prevent or overcome it can be devised.
This study was funded by Hoffman-LaRoche. Dr. Sosman and his associates reported ties to various companies, including Abraxis, Altor, Amgen, BMS, Celldex, Genentech, Genesis Biopharma, GlaxoSmithKline, Hoffman-La Roche, Merck, Novartis, Pfizer, Plexxikon, Prometheus, Roche, and Serametrix.
A phase-II clinical trial with the longest follow-up to date confirmed that vemurafenib induces a clinical response in more than half of patients with advanced melanoma expressing BRAFV600 mutations, according to a report in the Feb. 23 issue of the New England Journal of Medicine.
Median overall survival was 16 months in these patients, who had highly unfavorable characteristics at baseline. With follow-up as long as 20 months, this study "provides critical information on long-term survival" that was not available from previous studies of vemurafenib (Zelboraf) in metastatic melanoma, said Dr. Jeffrey A. Sosman of the Vanderbilt-Ingram Cancer Center, Nashville, Tenn., and his associates.
The Food and Drug Administration approved vemurafenib on Aug. 17, 2011 for patients with unresectable or metastatic melanoma with the BRAFV600E mutation as detected by an FDA-approved test. The FDA acted after a pivotal phase-III trial showed vemurafenib improved overall survival compared with dacarbazine. When the phase-III data were analyzed, median follow-up was just 6.2 months in the experimental arm, and median overall survival had not been reached for patients treated with vemurafenib.
The FDA also considered the currently reported phase-II trial, which included 132 patients with stage-IV melanoma that had progressed despite at least one systemic treatment. Tumor tissue from 56% of patients screened for the trial had a BRAFV600 mutation, which was required for study entry. Vemurafenib is a potent kinase inhibitor that was developed to target this mutation within cancer cells specifically.
Patients in the study were given oral vemurafenib (960 mg twice daily) and followed every 6 weeks for disease progression at 10 medical centers in the United States and 3 in Australia. Median follow-up for efficacy was 13 months (range, 0.6-20 months). Treatment response was assessed by both the investigative team and by a blinded independent review committee.
The committee found that 8 patients (6%) showed a complete response and 62 (47%) showed a partial response to vemurafenib, for an overall response rate of 53%. A total of 38 patients (29%) showed stable disease. Only 18 (14%) showed progressive disease; 6 patients could not be assessed.
Dr. Sosman and his colleagues found a 5% complete response rate and a 52% partial response rate, for an overall response rate of 57%, which generally accorded with the independent review committee’s findings.
Molecular sequencing showed 122 patients in the trial had BRAFV600E mutations and 10 patients had BRAFV600K mutations. Four of the 10 patients with the BRAFV600K mutation had a partial response and 3 had stable disease.
These response rates are higher than those associated with any previous treatments for advanced melanoma, the researchers noted. The lowest response occurred in patients with an LDL level more than 1.5 times the upper limit of normal.
The median duration of response was 6.7 months, and one-fourth of the subjects remained progression free for more than a year. Median overall survival was 16 months. At the time of the data cut-off (July 1, 2011), 62 patients (47% of the study population) were alive.
"Most objective responses were evident at the time of the first set of scans (week 6), but in some patients, responses did not appear until the patient had been receiving the drug for more than 6 months," the researchers said (N. Engl. J. Med. 2012;366:707-14).
The longer follow-up in this study thus permitted the investigators to observe these late responses.
Overall survival was 77% at 6 months and 58% at 12 months, and it was estimated to be 43% at 18 months, they noted.
Until now, median overall of metastatic melanoma has been 6-10 months, Dr. Sosman and his colleagues noted. In previously untreated patients who responded to ipilimumab (Yervoy) – an immunotherapy also approved last year – it reached 11 months.
Most patients had at least one adverse event related to vemurafenib, but most were not severe. Adverse effects chiefly were arthralgia, rash, photosensitivity, fatigue, and alopecia. Although many patients required dose interruptions (64%) or reductions (45%), "patients were able to receive most of their intended daily dose," the investigators wrote.
Four patients discontinued the study drug altogether, including one who developed a retinal-vein occlusion. Another patient died from rapidly progressive melanoma and renal failure that may have been related to the study drug.
Vemurafenib is known to have the paradoxical effect of promoting cutaneous squamous cell carcinomas or keratoacanthomas, so the study subjects were closely monitored for these. Thirty-four patients (26%) developed such neoplasms, usually at the start of therapy. All were managed with resection and vemurafenib was not discontinued. Another eight patients were found to have basal cell carcinomas.
"As with most targeted therapies that block a driver oncogene," vemurafenib will likely encounter resistance with long-term use, Dr. Sosman and his colleagues said. The mechanisms of resistance to vemurafenib are currently being investigated, so that strategies to prevent or overcome it can be devised.
This study was funded by Hoffman-LaRoche. Dr. Sosman and his associates reported ties to various companies, including Abraxis, Altor, Amgen, BMS, Celldex, Genentech, Genesis Biopharma, GlaxoSmithKline, Hoffman-La Roche, Merck, Novartis, Pfizer, Plexxikon, Prometheus, Roche, and Serametrix.
A phase-II clinical trial with the longest follow-up to date confirmed that vemurafenib induces a clinical response in more than half of patients with advanced melanoma expressing BRAFV600 mutations, according to a report in the Feb. 23 issue of the New England Journal of Medicine.
Median overall survival was 16 months in these patients, who had highly unfavorable characteristics at baseline. With follow-up as long as 20 months, this study "provides critical information on long-term survival" that was not available from previous studies of vemurafenib (Zelboraf) in metastatic melanoma, said Dr. Jeffrey A. Sosman of the Vanderbilt-Ingram Cancer Center, Nashville, Tenn., and his associates.
The Food and Drug Administration approved vemurafenib on Aug. 17, 2011 for patients with unresectable or metastatic melanoma with the BRAFV600E mutation as detected by an FDA-approved test. The FDA acted after a pivotal phase-III trial showed vemurafenib improved overall survival compared with dacarbazine. When the phase-III data were analyzed, median follow-up was just 6.2 months in the experimental arm, and median overall survival had not been reached for patients treated with vemurafenib.
The FDA also considered the currently reported phase-II trial, which included 132 patients with stage-IV melanoma that had progressed despite at least one systemic treatment. Tumor tissue from 56% of patients screened for the trial had a BRAFV600 mutation, which was required for study entry. Vemurafenib is a potent kinase inhibitor that was developed to target this mutation within cancer cells specifically.
Patients in the study were given oral vemurafenib (960 mg twice daily) and followed every 6 weeks for disease progression at 10 medical centers in the United States and 3 in Australia. Median follow-up for efficacy was 13 months (range, 0.6-20 months). Treatment response was assessed by both the investigative team and by a blinded independent review committee.
The committee found that 8 patients (6%) showed a complete response and 62 (47%) showed a partial response to vemurafenib, for an overall response rate of 53%. A total of 38 patients (29%) showed stable disease. Only 18 (14%) showed progressive disease; 6 patients could not be assessed.
Dr. Sosman and his colleagues found a 5% complete response rate and a 52% partial response rate, for an overall response rate of 57%, which generally accorded with the independent review committee’s findings.
Molecular sequencing showed 122 patients in the trial had BRAFV600E mutations and 10 patients had BRAFV600K mutations. Four of the 10 patients with the BRAFV600K mutation had a partial response and 3 had stable disease.
These response rates are higher than those associated with any previous treatments for advanced melanoma, the researchers noted. The lowest response occurred in patients with an LDL level more than 1.5 times the upper limit of normal.
The median duration of response was 6.7 months, and one-fourth of the subjects remained progression free for more than a year. Median overall survival was 16 months. At the time of the data cut-off (July 1, 2011), 62 patients (47% of the study population) were alive.
"Most objective responses were evident at the time of the first set of scans (week 6), but in some patients, responses did not appear until the patient had been receiving the drug for more than 6 months," the researchers said (N. Engl. J. Med. 2012;366:707-14).
The longer follow-up in this study thus permitted the investigators to observe these late responses.
Overall survival was 77% at 6 months and 58% at 12 months, and it was estimated to be 43% at 18 months, they noted.
Until now, median overall of metastatic melanoma has been 6-10 months, Dr. Sosman and his colleagues noted. In previously untreated patients who responded to ipilimumab (Yervoy) – an immunotherapy also approved last year – it reached 11 months.
Most patients had at least one adverse event related to vemurafenib, but most were not severe. Adverse effects chiefly were arthralgia, rash, photosensitivity, fatigue, and alopecia. Although many patients required dose interruptions (64%) or reductions (45%), "patients were able to receive most of their intended daily dose," the investigators wrote.
Four patients discontinued the study drug altogether, including one who developed a retinal-vein occlusion. Another patient died from rapidly progressive melanoma and renal failure that may have been related to the study drug.
Vemurafenib is known to have the paradoxical effect of promoting cutaneous squamous cell carcinomas or keratoacanthomas, so the study subjects were closely monitored for these. Thirty-four patients (26%) developed such neoplasms, usually at the start of therapy. All were managed with resection and vemurafenib was not discontinued. Another eight patients were found to have basal cell carcinomas.
"As with most targeted therapies that block a driver oncogene," vemurafenib will likely encounter resistance with long-term use, Dr. Sosman and his colleagues said. The mechanisms of resistance to vemurafenib are currently being investigated, so that strategies to prevent or overcome it can be devised.
This study was funded by Hoffman-LaRoche. Dr. Sosman and his associates reported ties to various companies, including Abraxis, Altor, Amgen, BMS, Celldex, Genentech, Genesis Biopharma, GlaxoSmithKline, Hoffman-La Roche, Merck, Novartis, Pfizer, Plexxikon, Prometheus, Roche, and Serametrix.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: The overall response rate to vemurafenib was 53%, and median overall survival was 16 months.
Data Source: The multicenter phase II clinical trial involved 132 patients who had advanced melanoma that expressed BRAFV600 mutations and were followed for as long as 20 months.
Disclosures: This study was funded by Hoffman-La Roche. Dr. Sosman and his associates reported ties to various companies, including Abraxis, Altor, Amgen, BMS, Celldex, Genentech, Genesis Biopharma, GlaxoSmithKline, Hoffman-La Roche, Merck, Novartis, Pfizer, Plexxikon, Prometheus, Roche, and Serametrix.
ACOG Releases Guideline for Breast Cancer Management
The American College of Obstetricians and Gynecologists advises that selective serotonin reuptake inhibitors, or SSRIs, can safely be used to treat hot flashes in some women with breast cancer.
And women who are able to become pregnant following breast cancer treatment can do so without increasing the risk of disease recurrence, according to the college.
The advice, published online Feb. 21 in Obstetrics and Gynecology as Practice Bulletin No. 126, is part of the college’s comprehensive new clinical guideline on the gynecological management of women who are being treated for – or who have been treated for – breast cancer (Obstet. Gynecol. 2012;119:666-82).
The guideline, which cites evidence from 166 published sources, covers such diverse issues as vasomotor symptoms; vaginal atrophy; contraception and fertility; uterine evaluation; and the treatment and prevention of bone loss in the context of current treatment regimens, which may include chemotherapy, hormonal treatments, radiation, and surgery.
"More and more women are living with breast cancer, and breast cancer treatments – particularly the hormonal therapies – have gynecological side effects," Dr. Mindy E. Goldman, lead author of the guideline, said in an interview.
"Ob.gyns. need to be aware of how these drugs work and know about any of the gynecological side effects," said Dr. Goldman, who is director of women’s cancer care for the department of obstetrics, gynecology, and reproductive sciences at the University of California, San Francisco.
For managing vasomotor symptoms such as hot flashes, the guideline recommends that – because hormonal therapy is generally contraindicated in women with hormone-positive breast cancer – an SSRI, an SNRI (serotonin norepinephrine reuptake inhibitor), or gabapentin be used instead. For women on tamoxifen, an SNRI is a better choice than an SSRI, because it avoids a potential interaction.
One SNRI, venlafaxine, was shown in randomized clinical trials to provide significant relief at low doses (75 mg) among women who had been treated for breast cancer. Gabapentin, an anticonvulsant used to control neuropathic pain in breast cancer patients, also helps relieve vasomotor symptoms at low doses, and may improve sleep quality. Pregabalin, a drug in the same class as gabapentin, also was seen as helpful, as was clonidine.
None of these medicines is licensed in the United States for the treatment of hot flashes.
Because chemotherapy, ovarian suppression, and aromatase inhibitors contribute to bone loss and increase fracture risk, the guideline recommends that pharmacologic therapy with bisphosphonates be considered for women who have T scores between –1.5 and –2.0, and be strongly considered for women with T scores less than –2.0, or who have a 10-year risk greater than 20% for a major fracture, or a 10-year hip fracture risk greater than 3%. Zoledronic acid was seen as a strong option among the bisphosphonates, and although raloxifene was generally well tolerated, vasomotor symptoms are among its reported adverse effects.
The guideline also recommends annual monitoring of women whose risks of bone loss significantly change as a result of treatment (for example, premenopausal women being treated with aromatase inhibitors). And vitamin D levels should be checked in women with breast cancer.
Up to 40% of women with breast cancer have severe vaginal dryness, and the topical hormonal creams, suppositories, and vaginal rings commonly used to treat vaginal dryness and atrophy have not been shown to be safe in women with breast cancer. Preference should be given to nonhormonal vaginal moisturizers, with hormonal treatments used on a short-term basis when nonhormonal options have failed. Testosterone supplementation, in patches or creams, remains without enough breast safety data to support it.
Contraceptives that are appropriate for women with breast cancer include barrier methods, the copper intrauterine device, and sterilization. Hormonal methods are contraindicated in women with breast cancer and are considered a risk even for women who have been cancer free for 5 or more years.
One exception may be the levonorgestrel-releasing intrauterine system, in which systemic absorption of levonorgestrel is minimal. However, the system has not been thoroughly studied with regard to long-term breast risk and should be considered only on a case-by-case basis, the guideline states.
Pregnancy following breast cancer treatment has not been shown to increase the risk of recurrence or mortality, according to a recent meta-analysis cited in the guideline (Eur. J. Cancer 2011;47:74-83).
But chemotherapy can compromise fertility, and 5-year use of tamoxifen may diminish a woman’s ovarian reserve before she may safely conceive. Therefore, many women of childbearing age will have difficulty becoming pregnant after treatment, and a fertility consultation at diagnosis is recommended so that advance planning can take place.
In vitro fertilization (IVF) with embryo cryopreservation is seen as a strong option for preserving the potential to have a child; however, there is concern that ovarian stimulation in IVF could cause proliferation of breast cancer cells, leading some practitioners to recommend natural cycle (nonstimulated) IVF.
It is not clear whether ovarian suppression during cancer treatment preserves fertility, according to the guideline. Tamoxifen as an ovarian stimulant has been investigated and has shown promise in treating women whose fertility has been compromised as a result of breast cancer treatment. Although the aromatase inhibitor letrozole cannot be used in premenopausal women as a breast cancer treatment, it may be used in combination with gonadotropins as a fertility agent following treatment.
Finally, routine endometrial biopsy and uterine ultrasonography are not recommended for postmenopausal women taking tamoxifen without evidence of vaginal bleeding, as ultrasound has been associated with a significant false-positive rate leading to unnecessary invasive diagnostic procedures.
However, for women on tamoxifen who experience vaginal bleeding, an endometrial evaluation – including biopsy and follow-up of possible uterine structural anomalies – is essential, the guideline states.
The members of the Committee on Practice Bulletins–Gynecology who wrote the guideline reported no relevant conflicts of interest.
The American College of Obstetricians and Gynecologists advises that selective serotonin reuptake inhibitors, or SSRIs, can safely be used to treat hot flashes in some women with breast cancer.
And women who are able to become pregnant following breast cancer treatment can do so without increasing the risk of disease recurrence, according to the college.
The advice, published online Feb. 21 in Obstetrics and Gynecology as Practice Bulletin No. 126, is part of the college’s comprehensive new clinical guideline on the gynecological management of women who are being treated for – or who have been treated for – breast cancer (Obstet. Gynecol. 2012;119:666-82).
The guideline, which cites evidence from 166 published sources, covers such diverse issues as vasomotor symptoms; vaginal atrophy; contraception and fertility; uterine evaluation; and the treatment and prevention of bone loss in the context of current treatment regimens, which may include chemotherapy, hormonal treatments, radiation, and surgery.
"More and more women are living with breast cancer, and breast cancer treatments – particularly the hormonal therapies – have gynecological side effects," Dr. Mindy E. Goldman, lead author of the guideline, said in an interview.
"Ob.gyns. need to be aware of how these drugs work and know about any of the gynecological side effects," said Dr. Goldman, who is director of women’s cancer care for the department of obstetrics, gynecology, and reproductive sciences at the University of California, San Francisco.
For managing vasomotor symptoms such as hot flashes, the guideline recommends that – because hormonal therapy is generally contraindicated in women with hormone-positive breast cancer – an SSRI, an SNRI (serotonin norepinephrine reuptake inhibitor), or gabapentin be used instead. For women on tamoxifen, an SNRI is a better choice than an SSRI, because it avoids a potential interaction.
One SNRI, venlafaxine, was shown in randomized clinical trials to provide significant relief at low doses (75 mg) among women who had been treated for breast cancer. Gabapentin, an anticonvulsant used to control neuropathic pain in breast cancer patients, also helps relieve vasomotor symptoms at low doses, and may improve sleep quality. Pregabalin, a drug in the same class as gabapentin, also was seen as helpful, as was clonidine.
None of these medicines is licensed in the United States for the treatment of hot flashes.
Because chemotherapy, ovarian suppression, and aromatase inhibitors contribute to bone loss and increase fracture risk, the guideline recommends that pharmacologic therapy with bisphosphonates be considered for women who have T scores between –1.5 and –2.0, and be strongly considered for women with T scores less than –2.0, or who have a 10-year risk greater than 20% for a major fracture, or a 10-year hip fracture risk greater than 3%. Zoledronic acid was seen as a strong option among the bisphosphonates, and although raloxifene was generally well tolerated, vasomotor symptoms are among its reported adverse effects.
The guideline also recommends annual monitoring of women whose risks of bone loss significantly change as a result of treatment (for example, premenopausal women being treated with aromatase inhibitors). And vitamin D levels should be checked in women with breast cancer.
Up to 40% of women with breast cancer have severe vaginal dryness, and the topical hormonal creams, suppositories, and vaginal rings commonly used to treat vaginal dryness and atrophy have not been shown to be safe in women with breast cancer. Preference should be given to nonhormonal vaginal moisturizers, with hormonal treatments used on a short-term basis when nonhormonal options have failed. Testosterone supplementation, in patches or creams, remains without enough breast safety data to support it.
Contraceptives that are appropriate for women with breast cancer include barrier methods, the copper intrauterine device, and sterilization. Hormonal methods are contraindicated in women with breast cancer and are considered a risk even for women who have been cancer free for 5 or more years.
One exception may be the levonorgestrel-releasing intrauterine system, in which systemic absorption of levonorgestrel is minimal. However, the system has not been thoroughly studied with regard to long-term breast risk and should be considered only on a case-by-case basis, the guideline states.
Pregnancy following breast cancer treatment has not been shown to increase the risk of recurrence or mortality, according to a recent meta-analysis cited in the guideline (Eur. J. Cancer 2011;47:74-83).
But chemotherapy can compromise fertility, and 5-year use of tamoxifen may diminish a woman’s ovarian reserve before she may safely conceive. Therefore, many women of childbearing age will have difficulty becoming pregnant after treatment, and a fertility consultation at diagnosis is recommended so that advance planning can take place.
In vitro fertilization (IVF) with embryo cryopreservation is seen as a strong option for preserving the potential to have a child; however, there is concern that ovarian stimulation in IVF could cause proliferation of breast cancer cells, leading some practitioners to recommend natural cycle (nonstimulated) IVF.
It is not clear whether ovarian suppression during cancer treatment preserves fertility, according to the guideline. Tamoxifen as an ovarian stimulant has been investigated and has shown promise in treating women whose fertility has been compromised as a result of breast cancer treatment. Although the aromatase inhibitor letrozole cannot be used in premenopausal women as a breast cancer treatment, it may be used in combination with gonadotropins as a fertility agent following treatment.
Finally, routine endometrial biopsy and uterine ultrasonography are not recommended for postmenopausal women taking tamoxifen without evidence of vaginal bleeding, as ultrasound has been associated with a significant false-positive rate leading to unnecessary invasive diagnostic procedures.
However, for women on tamoxifen who experience vaginal bleeding, an endometrial evaluation – including biopsy and follow-up of possible uterine structural anomalies – is essential, the guideline states.
The members of the Committee on Practice Bulletins–Gynecology who wrote the guideline reported no relevant conflicts of interest.
The American College of Obstetricians and Gynecologists advises that selective serotonin reuptake inhibitors, or SSRIs, can safely be used to treat hot flashes in some women with breast cancer.
And women who are able to become pregnant following breast cancer treatment can do so without increasing the risk of disease recurrence, according to the college.
The advice, published online Feb. 21 in Obstetrics and Gynecology as Practice Bulletin No. 126, is part of the college’s comprehensive new clinical guideline on the gynecological management of women who are being treated for – or who have been treated for – breast cancer (Obstet. Gynecol. 2012;119:666-82).
The guideline, which cites evidence from 166 published sources, covers such diverse issues as vasomotor symptoms; vaginal atrophy; contraception and fertility; uterine evaluation; and the treatment and prevention of bone loss in the context of current treatment regimens, which may include chemotherapy, hormonal treatments, radiation, and surgery.
"More and more women are living with breast cancer, and breast cancer treatments – particularly the hormonal therapies – have gynecological side effects," Dr. Mindy E. Goldman, lead author of the guideline, said in an interview.
"Ob.gyns. need to be aware of how these drugs work and know about any of the gynecological side effects," said Dr. Goldman, who is director of women’s cancer care for the department of obstetrics, gynecology, and reproductive sciences at the University of California, San Francisco.
For managing vasomotor symptoms such as hot flashes, the guideline recommends that – because hormonal therapy is generally contraindicated in women with hormone-positive breast cancer – an SSRI, an SNRI (serotonin norepinephrine reuptake inhibitor), or gabapentin be used instead. For women on tamoxifen, an SNRI is a better choice than an SSRI, because it avoids a potential interaction.
One SNRI, venlafaxine, was shown in randomized clinical trials to provide significant relief at low doses (75 mg) among women who had been treated for breast cancer. Gabapentin, an anticonvulsant used to control neuropathic pain in breast cancer patients, also helps relieve vasomotor symptoms at low doses, and may improve sleep quality. Pregabalin, a drug in the same class as gabapentin, also was seen as helpful, as was clonidine.
None of these medicines is licensed in the United States for the treatment of hot flashes.
Because chemotherapy, ovarian suppression, and aromatase inhibitors contribute to bone loss and increase fracture risk, the guideline recommends that pharmacologic therapy with bisphosphonates be considered for women who have T scores between –1.5 and –2.0, and be strongly considered for women with T scores less than –2.0, or who have a 10-year risk greater than 20% for a major fracture, or a 10-year hip fracture risk greater than 3%. Zoledronic acid was seen as a strong option among the bisphosphonates, and although raloxifene was generally well tolerated, vasomotor symptoms are among its reported adverse effects.
The guideline also recommends annual monitoring of women whose risks of bone loss significantly change as a result of treatment (for example, premenopausal women being treated with aromatase inhibitors). And vitamin D levels should be checked in women with breast cancer.
Up to 40% of women with breast cancer have severe vaginal dryness, and the topical hormonal creams, suppositories, and vaginal rings commonly used to treat vaginal dryness and atrophy have not been shown to be safe in women with breast cancer. Preference should be given to nonhormonal vaginal moisturizers, with hormonal treatments used on a short-term basis when nonhormonal options have failed. Testosterone supplementation, in patches or creams, remains without enough breast safety data to support it.
Contraceptives that are appropriate for women with breast cancer include barrier methods, the copper intrauterine device, and sterilization. Hormonal methods are contraindicated in women with breast cancer and are considered a risk even for women who have been cancer free for 5 or more years.
One exception may be the levonorgestrel-releasing intrauterine system, in which systemic absorption of levonorgestrel is minimal. However, the system has not been thoroughly studied with regard to long-term breast risk and should be considered only on a case-by-case basis, the guideline states.
Pregnancy following breast cancer treatment has not been shown to increase the risk of recurrence or mortality, according to a recent meta-analysis cited in the guideline (Eur. J. Cancer 2011;47:74-83).
But chemotherapy can compromise fertility, and 5-year use of tamoxifen may diminish a woman’s ovarian reserve before she may safely conceive. Therefore, many women of childbearing age will have difficulty becoming pregnant after treatment, and a fertility consultation at diagnosis is recommended so that advance planning can take place.
In vitro fertilization (IVF) with embryo cryopreservation is seen as a strong option for preserving the potential to have a child; however, there is concern that ovarian stimulation in IVF could cause proliferation of breast cancer cells, leading some practitioners to recommend natural cycle (nonstimulated) IVF.
It is not clear whether ovarian suppression during cancer treatment preserves fertility, according to the guideline. Tamoxifen as an ovarian stimulant has been investigated and has shown promise in treating women whose fertility has been compromised as a result of breast cancer treatment. Although the aromatase inhibitor letrozole cannot be used in premenopausal women as a breast cancer treatment, it may be used in combination with gonadotropins as a fertility agent following treatment.
Finally, routine endometrial biopsy and uterine ultrasonography are not recommended for postmenopausal women taking tamoxifen without evidence of vaginal bleeding, as ultrasound has been associated with a significant false-positive rate leading to unnecessary invasive diagnostic procedures.
However, for women on tamoxifen who experience vaginal bleeding, an endometrial evaluation – including biopsy and follow-up of possible uterine structural anomalies – is essential, the guideline states.
The members of the Committee on Practice Bulletins–Gynecology who wrote the guideline reported no relevant conflicts of interest.
FROM OBSTETRICS AND GYNECOLOGY
The Oncology Report Guide to Cancer Drugs and Devices in 2011
From an immunotherapy that improves melanoma survival to a device that zaps brain tumors, 2011 was a year of firsts from the FDA. This is our second annual guide to approvals of new cancer drugs, indications, and devices – along with a review of safety warnings and other actions affecting oncology practices.
(Click here for a PDF version of The Oncology Report Guide to Cancer Drugs and Devices in 2011.)
NEW DRUG APPROVALS
• Abiraterone acetate (Zytiga Tablets, Centocor Ortho Biotech, Inc.). An androgen suppressant that decreases testosterone production for use in combination with prednisone for the treatment of patients with metastatic castration-resistant prostate cancer (mCRPC) who have received prior chemotherapy containing docetaxel. The fourth approval in late-stage prostate cancer since April 2010.
Basis: An international study of 1,195 men who had previously received chemotherapy containing docetaxel. Median overall survival was 14.8 months with abiraterone plus prednisone, vs. 10.9 months with prednisone alone.
• Asparaginase Erwinia chrysanthemi [Erwinaze for injection, EUSA Pharma (USA), Inc.] An asparagine-specific enzyme approved as a component of a multiagent chemotherapeutic regimen for the treatment of patients with acute lymphoblastic leukemia (ALL) who developed hypersensitivity to E. coli-derived asparaginase. An orphan drug, it works by the same mechanism as the two previously approved treatments that block asparagine, a protein necessary for the proliferation of neoplastic cells.
Basis: In a single-arm, multicenter, open-label study of 58 patients, all 48 patients with available samples achieved threshold trough asparaginase levels shown to correlate with asparagine depletion and with serum levels that predict clinical efficacy.
• Brentuximab vedotin (Adcetris for injection, Seattle Genetics, Inc.). A CD30-directed antibody-drug conjugate for the treatment of Hodgkin’s lymphoma after failure of autologous stem cell transplant (ASCT) or at least two prior multiagent chemotherapy regimens in patients who are not ASCT candidates, and for the treatment of systemic anaplastic large cell lymphoma (ALCL) after failure of at least one prior multiagent chemotherapy regimen. The first new treatment approved for Hodgkin’s since 1977 and the first treatment approved for ALCL.
Basis: In a single-arm study of 102 patients treated with brentuximab, 73% had either a complete or partial response. In a similarly designed study of 58 patients with ALCL, 86% achieved a partial or complete response.
Addendum: An accelerated approval, based on clinical data suggesting that treatment resulted in significant responses; conversion to full approval is contingent on confirmation of benefits in follow-up studies. (In January 2012, the FDA added a warning about the risk of a rare brain infection to the label.)
• Crizotinib (Xalkori Capsules, Pfizer, Inc.). A kinase inhibitor for the treatment of locally advanced or metastatic non–small cell lung cancer (NSCLC) that express an abnormal anaplastic lymphoma kinase (ALK) gene, as detected by the Vysis ALK Break-Apart FISH Probe Kit (Abbott Molecular, Inc.), a companion diagnostic test approved at the same time.
Basis: In two single-arm studies of 255 patients with locally advanced or metastatic, ALK-positive NSCLC, most of whom had received prior chemotherapy, the overall response rate was 50% and 61%, with a median response duration of 42 and 48 weeks, respectively.
Addendum: An accelerated approval, so conversion to full approval is contingent on follow-up studies confirming clinical benefit.
• Deferiprone (Ferriprox Tablets, ApoPharma, Inc.). An oral iron chelator for the treatment of patients with transfusional iron overload due to thalassemia syndromes when current chelation therapy is inadequate.
Basis: A prospective pooled analysis of previously conducted studies of 236 patients (most had thalassemia syndromes) with transfusion-dependent iron overload, for which previous iron chelation therapy had failed or was considered inadequate because of poor tolerance. Within 1 year of starting treatment, 50% of the patients had at least a 20% decrease in serum ferritin levels.
Addendum: An accelerated approval. Safety and effectiveness not established for the treatment of transfusional iron overload due to other chronic anemias, including sickle cell disease. As a condition of approval, ApoPharma will conduct a study of safety and efficacy in patients with sickle cell disease and transfusional iron overload not adequately treated with available chelating agents.
• Hemacord (New York Blood Center). A hematopoietic progenitor cells–cord (HPC-C) preparation for allogenic hematopoietic stem cell transplantation. The first approval of a license for an umbilical cord blood therapy.
Basis: Safety and effectiveness data submitted to a public docket and data submitted in the license application demonstrating compliance with other regulatory requirements.
• Ipilimumab injection (Yervoy, Bristol-Myers Squibb). A human cytotoxic T-lymphocyte antigen (CTLA-4)-blocking antibody for the treatment of unresectable or metastatic melanoma. The first treatment proven to prolong survival in patients with metastatic melanoma.
Basis: An international study of 676 patients with previously treated unresectable stage III or IV melanoma whose disease had progressed, comparing ipilimumab plus an experimental tumor vaccine (gp100), gp100 alone, and ipilimumab alone. Median overall survival was 10 months among those who received ipilimumab with or without the vaccine, vs. 6 months among those who received the vaccine alone.
Addendum: Approved with a Risk Evaluation and Mitigation Strategy (REMS) program addressing life-threatening toxicity and deaths associated with ipilimumab in the study. Acts slowly, and some patients may have tumor progression before therapy becomes effective.
• Ruxolitinib (Jakafi oral tablets, Incyte Corp.). A Janus-associated kinase (JAK) inhibitor for the treatment of intermediate- or high-risk myelofibrosis, including primary myelofibrosis, post–polycythemia vera myelofibrosis, and post–essential thrombocythemia myelofibrosis. An orphan drug that is the first treatment approved for the rare blood disease – and the first approved JAK inhibitor.
Basis: Two randomized controlled trials of 528 patients with intermediate- or high-risk myelofibrosis. In one study, 42% of those on ruxolitinib had more than at least a 35% reduction in spleen volume vs. 1% of those on placebo at 24 weeks, and nearly half of those on treatment had achieved at least a 50% reduction in myelofibrosis symptoms, vs. 5% of those on placebo. In the second study, 29% of those on ruxolitinib had at least a 35% reduction in spleen size, vs. none of those on best available therapy after 48 weeks.
• Vandetanib (Caprelsa, AstraZeneca Pharmaceuticals LP). A kinase inhibitor for the treatment of symptomatic or progressive medullary thyroid cancer in adults with unresectable, locally advanced, or metastatic disease. The first drug approved for this rare cancer.
Basis: An international study of 331 patients with unresectable locally advanced or metastatic medullary thyroid cancer. Median progression-free survival was at least 22.6 months with vandetanib vs. 16.4 months among with placebo, a highly significant difference.
Addendum: Approved with a REMS addressing the risk of QT interval prolongation and a note that use in patients with indolent, asymptomatic, or slowly progressing disease "should be carefully considered" because of treatment-associated risks, as well as a boxed warning and a restricted distribution program.
• Vemurafenib (Zelboraf tablets, Hoffmann-LaRoche, Inc.). A BRAF inhibitor for the treatment of patients with unresectable or metastatic melanoma with the BRAFV600E mutation, as detected by the cobas 4800 BRAF V600 Mutation Test (Roche Molecular Systems), a companion diagnostic test approved at the same time. The second therapy found to prolong survival in patients with melanoma.
Basis: A study of 675 treatment-naive patients with unresectable or metastatic melanoma, positive for the BRAFV600E mutation. At the time of review, median survival was 7.9 months in a control group treated with dacarbazine, but had not been reached among those on vemurafenib. Median progression-free survival was 1.6 and 5.3 months, respectively.
Addendum: About half of melanoma patients have the BRAF mutation, and about half of them respond to vemurafenib. Approved with a medication guide to inform patients about the need for the diagnostic test and potential risks associated with treatment, including cutaneous squamous cell carcinoma. Roche and ipilimumab manufacturer Bristol-Meyers Squibb announced collaboration on a phase I/II study to determine whether combining the two agents is safe and effective in patients with BRAF mutations.
NEW DEVICES
• MelaFind (Mela Sciences). A noninvasive melanoma detection device for use "on clinically atypical cutaneous pigmented lesions with one or more clinical or historical characteristics of melanoma, excluding those with a clinical diagnosis of melanoma or likely melanoma."
Basis: The road to approval was controversial, with a split vote from an advisory panel, a citizens petition submitted by the manufacturer, and an agreement between the agency and manufacturer on how the device would be used and by whom.
• NovoTTF-100A System (NovoTTF, Novocure). A portable, battery-powered device that delivers electrical fields to the brain as a treatment for adults with glioblastoma multiforme that has recurred after chemotherapy.
Basis: In a randomized study of 237 patients whose GBM had recurred after surgery, radiation, and chemotherapy, median overall survival was 6.3 months with the approximately 6-pound device and 6.4 months with best available chemotherapy. The agency cited evidence suggesting better quality of life without chemotherapy side effects.
NEW INDICATIONS
• Cetuximab (Erbitux, ImClone LLC, a wholly owned subsidiary of Eli Lilly & Co. and Bristol-Myers Squibb). The epidermal growth factor receptor (EGFR) antagonist in combination with platinum-based therapy plus 5-fluorouracil for the first-line treatment of recurrent locoregional and/or metastatic squamous cell carcinoma of the head and neck.
Basis: A multicenter non-U.S. study of 442 patients not suitable for potentially curative treatment with surgery or radiation. Mean overall survival was 10.1 months with cetuximab and chemotherapy (cisplatin or carboplatin and 5-fluorouracil), vs. 7.4 months with chemotherapy alone.
Addendum: Cetuximab available in the United States provides about 22% greater exposure than the European Union–approved cetuximab used in this study, but the study results, pharmacokinetic data, and other clinical trial data "establish the efficacy" of cetuximab at the dose recommended, according to the prescribing information.
• Denosumab (Prolia, Amgen, Inc.). The RANK ligand (RANKL) inhibitor to increase bone mass in patients at high risk for fracture when receiving androgen deprivation therapy (ADT) for nonmetastatic prostate cancer or adjuvant aromatase inhibitor (AI) therapy for breast cancer.
Basis: Two international, randomized, double-blind placebo-controlled studies. In one study of 1,468 men with nonmetastatic prostate cancer receiving ADT, lumbar spine bone mineral density (BMD) at 2 years was significantly higher with denosumab vs. placebo (6.7% difference). At 3 years of treatment, the incidence of new vertebral fractures was 1.5% with denosumab vs. 3.9% with placebo, a risk reduction of 62%. In a study of 252 postmenopausal women with breast cancer on AI treatment, BMD at the lumbar spine was significantly higher with denosumab vs. placebo after 12 months of treatment (5.5% difference).
Addendum: Denosumab was initially approved in 2010. It is marketed as Prolia for treating postmenopausal women with osteoporosis at high risk of fracture, and as Xgeva for preventing skeletal-related events in patients with bone metastases from solid tumors; Xgeva is administered more frequently and at a higher dose for the latter indication.
• Everolimus (Afinitor Tablets, Novartis Pharmaceuticals Corp.). The mammalian target of rapamycin (mTOR) inhibitor for progressive pancreatic neuroendocrine tumors (pNETs) that are unresectable, locally advanced, or metastatic. (Safety and effectiveness of everolimus for treating carcinoid tumors have not been established). The first new drug in about 30 years for the treatment of advanced pNET.
Basis: A randomized controlled study of 410 patients, all of whom also received best supportive care. Median progression-free survival was 11 months with everolimus, vs. 4.6 months with placebo. In an interim analysis, overall survival was not different between the two groups.
• Peg-interferon alfa-2b (Sylatron, Schering Corp.). An alpha interferon for the adjuvant treatment of melanoma with microscopic or gross nodal involvement within 84 days of definitive surgical resection, including complete lymphadenectomy.
Basis: An open-label multicenter study of 1,256 patients. Median relapse-free survival was 34.8 months among those treated with peg-interferon alfa-2b, vs. 25.5 months with observation.
• Rituximab (Rituxan, Genentech, Inc.). The CD20-directed cytolytic antibody for maintenance therapy in people with previously untreated follicular, CD-20 positive, B-cell non-Hodgkin lymphoma who achieve a response to rituximab in combination with chemotherapy.
Basis: An international study of 1,217 previously untreated patients with advanced follicular lymphoma; it randomized 1,018 patients who had a complete or partial response with rituximab and chemotherapy. Maintenance treatment with rituximab reduced the risk of progression by 46% compared with observation only.
• Sunitinib (Sutent Capsules, Pfizer, Inc.). A kinase inhibitor for the treatment of progressive, well-differentiated pancreatic neuroendocrine tumors (pNETs) in patients with unresectable, locally advanced, or metastatic disease; it was approved soon after everolimus for this indication.
Basis: A randomized controlled study of 171 patients with unresectable, locally advanced or well-differentiated pNET; treatment with somatostatin analogues was allowed. Median progression-free survival was 10.2 months with sunitinib, vs. 5.4 months with placebo.
WARNINGS AND LABEL CHANGES
• Bevacizumab (Avastin, Genentech, Inc.). Revised prescribing information includes the risk of ovarian failure in premenopausal women treated with bevacizumab and chemotherapy; postmarketing reports of osteonecrosis of the jaw in patients treated with bevacizumab but not bisphosphonates; and information from a clinical trial about the risk of venous thromboembolic events and bleeding in patients receiving anticoagulation therapy after a first VTE event while receiving bevacizumab.
• Dasatinib (Sprycel, Bristol-Myers Squibb). Information on increased risk for pulmonary arterial hypertension added to prescribing information.
• Fentanyl. A classwide risk management program approved for all transmucosal immediate-release fentanyl formulations.
• IV methotrexate and proton pump inhibitors (PPIs). A warning added to the methotrexate label advises caution when high-dose methotrexate is administered to patients taking a PPI, because concomitant administration may result in elevated and prolonged serum levels of methotrexate and/or its metabolite hydroxymethotrexate, with possible toxic effects.
• Lenalidomide (Revlimid, Celgene). Advisory informed the public that the FDA is monitoring the risk of secondary cancers in patients treated for myelodysplastic syndromes or multiple myeloma.
• Ondansetron (Zofran, GlaxoSmithKline, and generic formulations). Following a review of information on the risk of QT prolongation, label now recommends ECG monitoring in patients who are treated with ondansetron and have an electrolyte abnormality, congestive heart failure, or bradyarrhythmias; and in patients taking other medications that prolong the QT interval – who are at an increased risk of developing torsades de pointes. Label recommends against use in patients with congenital long QT syndrome. Safety review ongoing.
• Opioids. Draft issued for opioid prescriber education program, as part of the Risk Evaluation and Mitigation Strategy (REMS) now in place for brand-name and generic long-acting and extended-release opioids.
• Pioglitazone. Bladder cancer warning risk added to the label for type 2 diabetes drug marketed as Actos (or in combination with metformin as Actoplus Met, and with glimepiride as Duetact).
• Romiplostim (Nplate for subcutaneous injection, Amgen, Inc.) and eltrombopag (Promacta tablets, GlaxoSmithKline LLC). Elements of the REMS for the two thrombopoietin receptor agonists were dropped, including the restricted distribution program that required health care professionals, hospitals, and patients to be enrolled in order to prescribe, dispense, or receive these drugs. Clinicians no longer need to file periodic safety forms for their patients on these treatments.
• Tumor necrosis factor (TNF) blockers. Manufacturers asked to report cases of malignancies in children, adolescents, and adults aged 30 years and younger treated with TNF blockers and to conduct in-depth follow-up of them.
OTHER ACTIONS
• Bevacizumab (Avastin, Genentech, Inc.). FDA Commissioner Margaret Hamburg withdrew bevacizumab’s indication in metastatic breast cancer. She cited a lack of data establishing benefit and serious risks associated with treatment. The decision followed a July hearing, where the Oncologic Drugs Advisory Committee unanimously voted that the indication be withdrawn despite emotional testimony from patients who said they benefited from the monoclonal antibody.
• Drug shortages. A new rule requires some manufacturers to give early warning of shortages. The agency also held a workshop on the crisis and is working to avert and/or resolve shortages.
• Office of Oncology Products. Many New Drug Applications, Biologic License Applications, and Investigational New Drug Applications for cancer drugs are being reassigned to new review divisions as part of a reorganization.
From an immunotherapy that improves melanoma survival to a device that zaps brain tumors, 2011 was a year of firsts from the FDA. This is our second annual guide to approvals of new cancer drugs, indications, and devices – along with a review of safety warnings and other actions affecting oncology practices.
(Click here for a PDF version of The Oncology Report Guide to Cancer Drugs and Devices in 2011.)
NEW DRUG APPROVALS
• Abiraterone acetate (Zytiga Tablets, Centocor Ortho Biotech, Inc.). An androgen suppressant that decreases testosterone production for use in combination with prednisone for the treatment of patients with metastatic castration-resistant prostate cancer (mCRPC) who have received prior chemotherapy containing docetaxel. The fourth approval in late-stage prostate cancer since April 2010.
Basis: An international study of 1,195 men who had previously received chemotherapy containing docetaxel. Median overall survival was 14.8 months with abiraterone plus prednisone, vs. 10.9 months with prednisone alone.
• Asparaginase Erwinia chrysanthemi [Erwinaze for injection, EUSA Pharma (USA), Inc.] An asparagine-specific enzyme approved as a component of a multiagent chemotherapeutic regimen for the treatment of patients with acute lymphoblastic leukemia (ALL) who developed hypersensitivity to E. coli-derived asparaginase. An orphan drug, it works by the same mechanism as the two previously approved treatments that block asparagine, a protein necessary for the proliferation of neoplastic cells.
Basis: In a single-arm, multicenter, open-label study of 58 patients, all 48 patients with available samples achieved threshold trough asparaginase levels shown to correlate with asparagine depletion and with serum levels that predict clinical efficacy.
• Brentuximab vedotin (Adcetris for injection, Seattle Genetics, Inc.). A CD30-directed antibody-drug conjugate for the treatment of Hodgkin’s lymphoma after failure of autologous stem cell transplant (ASCT) or at least two prior multiagent chemotherapy regimens in patients who are not ASCT candidates, and for the treatment of systemic anaplastic large cell lymphoma (ALCL) after failure of at least one prior multiagent chemotherapy regimen. The first new treatment approved for Hodgkin’s since 1977 and the first treatment approved for ALCL.
Basis: In a single-arm study of 102 patients treated with brentuximab, 73% had either a complete or partial response. In a similarly designed study of 58 patients with ALCL, 86% achieved a partial or complete response.
Addendum: An accelerated approval, based on clinical data suggesting that treatment resulted in significant responses; conversion to full approval is contingent on confirmation of benefits in follow-up studies. (In January 2012, the FDA added a warning about the risk of a rare brain infection to the label.)
• Crizotinib (Xalkori Capsules, Pfizer, Inc.). A kinase inhibitor for the treatment of locally advanced or metastatic non–small cell lung cancer (NSCLC) that express an abnormal anaplastic lymphoma kinase (ALK) gene, as detected by the Vysis ALK Break-Apart FISH Probe Kit (Abbott Molecular, Inc.), a companion diagnostic test approved at the same time.
Basis: In two single-arm studies of 255 patients with locally advanced or metastatic, ALK-positive NSCLC, most of whom had received prior chemotherapy, the overall response rate was 50% and 61%, with a median response duration of 42 and 48 weeks, respectively.
Addendum: An accelerated approval, so conversion to full approval is contingent on follow-up studies confirming clinical benefit.
• Deferiprone (Ferriprox Tablets, ApoPharma, Inc.). An oral iron chelator for the treatment of patients with transfusional iron overload due to thalassemia syndromes when current chelation therapy is inadequate.
Basis: A prospective pooled analysis of previously conducted studies of 236 patients (most had thalassemia syndromes) with transfusion-dependent iron overload, for which previous iron chelation therapy had failed or was considered inadequate because of poor tolerance. Within 1 year of starting treatment, 50% of the patients had at least a 20% decrease in serum ferritin levels.
Addendum: An accelerated approval. Safety and effectiveness not established for the treatment of transfusional iron overload due to other chronic anemias, including sickle cell disease. As a condition of approval, ApoPharma will conduct a study of safety and efficacy in patients with sickle cell disease and transfusional iron overload not adequately treated with available chelating agents.
• Hemacord (New York Blood Center). A hematopoietic progenitor cells–cord (HPC-C) preparation for allogenic hematopoietic stem cell transplantation. The first approval of a license for an umbilical cord blood therapy.
Basis: Safety and effectiveness data submitted to a public docket and data submitted in the license application demonstrating compliance with other regulatory requirements.
• Ipilimumab injection (Yervoy, Bristol-Myers Squibb). A human cytotoxic T-lymphocyte antigen (CTLA-4)-blocking antibody for the treatment of unresectable or metastatic melanoma. The first treatment proven to prolong survival in patients with metastatic melanoma.
Basis: An international study of 676 patients with previously treated unresectable stage III or IV melanoma whose disease had progressed, comparing ipilimumab plus an experimental tumor vaccine (gp100), gp100 alone, and ipilimumab alone. Median overall survival was 10 months among those who received ipilimumab with or without the vaccine, vs. 6 months among those who received the vaccine alone.
Addendum: Approved with a Risk Evaluation and Mitigation Strategy (REMS) program addressing life-threatening toxicity and deaths associated with ipilimumab in the study. Acts slowly, and some patients may have tumor progression before therapy becomes effective.
• Ruxolitinib (Jakafi oral tablets, Incyte Corp.). A Janus-associated kinase (JAK) inhibitor for the treatment of intermediate- or high-risk myelofibrosis, including primary myelofibrosis, post–polycythemia vera myelofibrosis, and post–essential thrombocythemia myelofibrosis. An orphan drug that is the first treatment approved for the rare blood disease – and the first approved JAK inhibitor.
Basis: Two randomized controlled trials of 528 patients with intermediate- or high-risk myelofibrosis. In one study, 42% of those on ruxolitinib had more than at least a 35% reduction in spleen volume vs. 1% of those on placebo at 24 weeks, and nearly half of those on treatment had achieved at least a 50% reduction in myelofibrosis symptoms, vs. 5% of those on placebo. In the second study, 29% of those on ruxolitinib had at least a 35% reduction in spleen size, vs. none of those on best available therapy after 48 weeks.
• Vandetanib (Caprelsa, AstraZeneca Pharmaceuticals LP). A kinase inhibitor for the treatment of symptomatic or progressive medullary thyroid cancer in adults with unresectable, locally advanced, or metastatic disease. The first drug approved for this rare cancer.
Basis: An international study of 331 patients with unresectable locally advanced or metastatic medullary thyroid cancer. Median progression-free survival was at least 22.6 months with vandetanib vs. 16.4 months among with placebo, a highly significant difference.
Addendum: Approved with a REMS addressing the risk of QT interval prolongation and a note that use in patients with indolent, asymptomatic, or slowly progressing disease "should be carefully considered" because of treatment-associated risks, as well as a boxed warning and a restricted distribution program.
• Vemurafenib (Zelboraf tablets, Hoffmann-LaRoche, Inc.). A BRAF inhibitor for the treatment of patients with unresectable or metastatic melanoma with the BRAFV600E mutation, as detected by the cobas 4800 BRAF V600 Mutation Test (Roche Molecular Systems), a companion diagnostic test approved at the same time. The second therapy found to prolong survival in patients with melanoma.
Basis: A study of 675 treatment-naive patients with unresectable or metastatic melanoma, positive for the BRAFV600E mutation. At the time of review, median survival was 7.9 months in a control group treated with dacarbazine, but had not been reached among those on vemurafenib. Median progression-free survival was 1.6 and 5.3 months, respectively.
Addendum: About half of melanoma patients have the BRAF mutation, and about half of them respond to vemurafenib. Approved with a medication guide to inform patients about the need for the diagnostic test and potential risks associated with treatment, including cutaneous squamous cell carcinoma. Roche and ipilimumab manufacturer Bristol-Meyers Squibb announced collaboration on a phase I/II study to determine whether combining the two agents is safe and effective in patients with BRAF mutations.
NEW DEVICES
• MelaFind (Mela Sciences). A noninvasive melanoma detection device for use "on clinically atypical cutaneous pigmented lesions with one or more clinical or historical characteristics of melanoma, excluding those with a clinical diagnosis of melanoma or likely melanoma."
Basis: The road to approval was controversial, with a split vote from an advisory panel, a citizens petition submitted by the manufacturer, and an agreement between the agency and manufacturer on how the device would be used and by whom.
• NovoTTF-100A System (NovoTTF, Novocure). A portable, battery-powered device that delivers electrical fields to the brain as a treatment for adults with glioblastoma multiforme that has recurred after chemotherapy.
Basis: In a randomized study of 237 patients whose GBM had recurred after surgery, radiation, and chemotherapy, median overall survival was 6.3 months with the approximately 6-pound device and 6.4 months with best available chemotherapy. The agency cited evidence suggesting better quality of life without chemotherapy side effects.
NEW INDICATIONS
• Cetuximab (Erbitux, ImClone LLC, a wholly owned subsidiary of Eli Lilly & Co. and Bristol-Myers Squibb). The epidermal growth factor receptor (EGFR) antagonist in combination with platinum-based therapy plus 5-fluorouracil for the first-line treatment of recurrent locoregional and/or metastatic squamous cell carcinoma of the head and neck.
Basis: A multicenter non-U.S. study of 442 patients not suitable for potentially curative treatment with surgery or radiation. Mean overall survival was 10.1 months with cetuximab and chemotherapy (cisplatin or carboplatin and 5-fluorouracil), vs. 7.4 months with chemotherapy alone.
Addendum: Cetuximab available in the United States provides about 22% greater exposure than the European Union–approved cetuximab used in this study, but the study results, pharmacokinetic data, and other clinical trial data "establish the efficacy" of cetuximab at the dose recommended, according to the prescribing information.
• Denosumab (Prolia, Amgen, Inc.). The RANK ligand (RANKL) inhibitor to increase bone mass in patients at high risk for fracture when receiving androgen deprivation therapy (ADT) for nonmetastatic prostate cancer or adjuvant aromatase inhibitor (AI) therapy for breast cancer.
Basis: Two international, randomized, double-blind placebo-controlled studies. In one study of 1,468 men with nonmetastatic prostate cancer receiving ADT, lumbar spine bone mineral density (BMD) at 2 years was significantly higher with denosumab vs. placebo (6.7% difference). At 3 years of treatment, the incidence of new vertebral fractures was 1.5% with denosumab vs. 3.9% with placebo, a risk reduction of 62%. In a study of 252 postmenopausal women with breast cancer on AI treatment, BMD at the lumbar spine was significantly higher with denosumab vs. placebo after 12 months of treatment (5.5% difference).
Addendum: Denosumab was initially approved in 2010. It is marketed as Prolia for treating postmenopausal women with osteoporosis at high risk of fracture, and as Xgeva for preventing skeletal-related events in patients with bone metastases from solid tumors; Xgeva is administered more frequently and at a higher dose for the latter indication.
• Everolimus (Afinitor Tablets, Novartis Pharmaceuticals Corp.). The mammalian target of rapamycin (mTOR) inhibitor for progressive pancreatic neuroendocrine tumors (pNETs) that are unresectable, locally advanced, or metastatic. (Safety and effectiveness of everolimus for treating carcinoid tumors have not been established). The first new drug in about 30 years for the treatment of advanced pNET.
Basis: A randomized controlled study of 410 patients, all of whom also received best supportive care. Median progression-free survival was 11 months with everolimus, vs. 4.6 months with placebo. In an interim analysis, overall survival was not different between the two groups.
• Peg-interferon alfa-2b (Sylatron, Schering Corp.). An alpha interferon for the adjuvant treatment of melanoma with microscopic or gross nodal involvement within 84 days of definitive surgical resection, including complete lymphadenectomy.
Basis: An open-label multicenter study of 1,256 patients. Median relapse-free survival was 34.8 months among those treated with peg-interferon alfa-2b, vs. 25.5 months with observation.
• Rituximab (Rituxan, Genentech, Inc.). The CD20-directed cytolytic antibody for maintenance therapy in people with previously untreated follicular, CD-20 positive, B-cell non-Hodgkin lymphoma who achieve a response to rituximab in combination with chemotherapy.
Basis: An international study of 1,217 previously untreated patients with advanced follicular lymphoma; it randomized 1,018 patients who had a complete or partial response with rituximab and chemotherapy. Maintenance treatment with rituximab reduced the risk of progression by 46% compared with observation only.
• Sunitinib (Sutent Capsules, Pfizer, Inc.). A kinase inhibitor for the treatment of progressive, well-differentiated pancreatic neuroendocrine tumors (pNETs) in patients with unresectable, locally advanced, or metastatic disease; it was approved soon after everolimus for this indication.
Basis: A randomized controlled study of 171 patients with unresectable, locally advanced or well-differentiated pNET; treatment with somatostatin analogues was allowed. Median progression-free survival was 10.2 months with sunitinib, vs. 5.4 months with placebo.
WARNINGS AND LABEL CHANGES
• Bevacizumab (Avastin, Genentech, Inc.). Revised prescribing information includes the risk of ovarian failure in premenopausal women treated with bevacizumab and chemotherapy; postmarketing reports of osteonecrosis of the jaw in patients treated with bevacizumab but not bisphosphonates; and information from a clinical trial about the risk of venous thromboembolic events and bleeding in patients receiving anticoagulation therapy after a first VTE event while receiving bevacizumab.
• Dasatinib (Sprycel, Bristol-Myers Squibb). Information on increased risk for pulmonary arterial hypertension added to prescribing information.
• Fentanyl. A classwide risk management program approved for all transmucosal immediate-release fentanyl formulations.
• IV methotrexate and proton pump inhibitors (PPIs). A warning added to the methotrexate label advises caution when high-dose methotrexate is administered to patients taking a PPI, because concomitant administration may result in elevated and prolonged serum levels of methotrexate and/or its metabolite hydroxymethotrexate, with possible toxic effects.
• Lenalidomide (Revlimid, Celgene). Advisory informed the public that the FDA is monitoring the risk of secondary cancers in patients treated for myelodysplastic syndromes or multiple myeloma.
• Ondansetron (Zofran, GlaxoSmithKline, and generic formulations). Following a review of information on the risk of QT prolongation, label now recommends ECG monitoring in patients who are treated with ondansetron and have an electrolyte abnormality, congestive heart failure, or bradyarrhythmias; and in patients taking other medications that prolong the QT interval – who are at an increased risk of developing torsades de pointes. Label recommends against use in patients with congenital long QT syndrome. Safety review ongoing.
• Opioids. Draft issued for opioid prescriber education program, as part of the Risk Evaluation and Mitigation Strategy (REMS) now in place for brand-name and generic long-acting and extended-release opioids.
• Pioglitazone. Bladder cancer warning risk added to the label for type 2 diabetes drug marketed as Actos (or in combination with metformin as Actoplus Met, and with glimepiride as Duetact).
• Romiplostim (Nplate for subcutaneous injection, Amgen, Inc.) and eltrombopag (Promacta tablets, GlaxoSmithKline LLC). Elements of the REMS for the two thrombopoietin receptor agonists were dropped, including the restricted distribution program that required health care professionals, hospitals, and patients to be enrolled in order to prescribe, dispense, or receive these drugs. Clinicians no longer need to file periodic safety forms for their patients on these treatments.
• Tumor necrosis factor (TNF) blockers. Manufacturers asked to report cases of malignancies in children, adolescents, and adults aged 30 years and younger treated with TNF blockers and to conduct in-depth follow-up of them.
OTHER ACTIONS
• Bevacizumab (Avastin, Genentech, Inc.). FDA Commissioner Margaret Hamburg withdrew bevacizumab’s indication in metastatic breast cancer. She cited a lack of data establishing benefit and serious risks associated with treatment. The decision followed a July hearing, where the Oncologic Drugs Advisory Committee unanimously voted that the indication be withdrawn despite emotional testimony from patients who said they benefited from the monoclonal antibody.
• Drug shortages. A new rule requires some manufacturers to give early warning of shortages. The agency also held a workshop on the crisis and is working to avert and/or resolve shortages.
• Office of Oncology Products. Many New Drug Applications, Biologic License Applications, and Investigational New Drug Applications for cancer drugs are being reassigned to new review divisions as part of a reorganization.
From an immunotherapy that improves melanoma survival to a device that zaps brain tumors, 2011 was a year of firsts from the FDA. This is our second annual guide to approvals of new cancer drugs, indications, and devices – along with a review of safety warnings and other actions affecting oncology practices.
(Click here for a PDF version of The Oncology Report Guide to Cancer Drugs and Devices in 2011.)
NEW DRUG APPROVALS
• Abiraterone acetate (Zytiga Tablets, Centocor Ortho Biotech, Inc.). An androgen suppressant that decreases testosterone production for use in combination with prednisone for the treatment of patients with metastatic castration-resistant prostate cancer (mCRPC) who have received prior chemotherapy containing docetaxel. The fourth approval in late-stage prostate cancer since April 2010.
Basis: An international study of 1,195 men who had previously received chemotherapy containing docetaxel. Median overall survival was 14.8 months with abiraterone plus prednisone, vs. 10.9 months with prednisone alone.
• Asparaginase Erwinia chrysanthemi [Erwinaze for injection, EUSA Pharma (USA), Inc.] An asparagine-specific enzyme approved as a component of a multiagent chemotherapeutic regimen for the treatment of patients with acute lymphoblastic leukemia (ALL) who developed hypersensitivity to E. coli-derived asparaginase. An orphan drug, it works by the same mechanism as the two previously approved treatments that block asparagine, a protein necessary for the proliferation of neoplastic cells.
Basis: In a single-arm, multicenter, open-label study of 58 patients, all 48 patients with available samples achieved threshold trough asparaginase levels shown to correlate with asparagine depletion and with serum levels that predict clinical efficacy.
• Brentuximab vedotin (Adcetris for injection, Seattle Genetics, Inc.). A CD30-directed antibody-drug conjugate for the treatment of Hodgkin’s lymphoma after failure of autologous stem cell transplant (ASCT) or at least two prior multiagent chemotherapy regimens in patients who are not ASCT candidates, and for the treatment of systemic anaplastic large cell lymphoma (ALCL) after failure of at least one prior multiagent chemotherapy regimen. The first new treatment approved for Hodgkin’s since 1977 and the first treatment approved for ALCL.
Basis: In a single-arm study of 102 patients treated with brentuximab, 73% had either a complete or partial response. In a similarly designed study of 58 patients with ALCL, 86% achieved a partial or complete response.
Addendum: An accelerated approval, based on clinical data suggesting that treatment resulted in significant responses; conversion to full approval is contingent on confirmation of benefits in follow-up studies. (In January 2012, the FDA added a warning about the risk of a rare brain infection to the label.)
• Crizotinib (Xalkori Capsules, Pfizer, Inc.). A kinase inhibitor for the treatment of locally advanced or metastatic non–small cell lung cancer (NSCLC) that express an abnormal anaplastic lymphoma kinase (ALK) gene, as detected by the Vysis ALK Break-Apart FISH Probe Kit (Abbott Molecular, Inc.), a companion diagnostic test approved at the same time.
Basis: In two single-arm studies of 255 patients with locally advanced or metastatic, ALK-positive NSCLC, most of whom had received prior chemotherapy, the overall response rate was 50% and 61%, with a median response duration of 42 and 48 weeks, respectively.
Addendum: An accelerated approval, so conversion to full approval is contingent on follow-up studies confirming clinical benefit.
• Deferiprone (Ferriprox Tablets, ApoPharma, Inc.). An oral iron chelator for the treatment of patients with transfusional iron overload due to thalassemia syndromes when current chelation therapy is inadequate.
Basis: A prospective pooled analysis of previously conducted studies of 236 patients (most had thalassemia syndromes) with transfusion-dependent iron overload, for which previous iron chelation therapy had failed or was considered inadequate because of poor tolerance. Within 1 year of starting treatment, 50% of the patients had at least a 20% decrease in serum ferritin levels.
Addendum: An accelerated approval. Safety and effectiveness not established for the treatment of transfusional iron overload due to other chronic anemias, including sickle cell disease. As a condition of approval, ApoPharma will conduct a study of safety and efficacy in patients with sickle cell disease and transfusional iron overload not adequately treated with available chelating agents.
• Hemacord (New York Blood Center). A hematopoietic progenitor cells–cord (HPC-C) preparation for allogenic hematopoietic stem cell transplantation. The first approval of a license for an umbilical cord blood therapy.
Basis: Safety and effectiveness data submitted to a public docket and data submitted in the license application demonstrating compliance with other regulatory requirements.
• Ipilimumab injection (Yervoy, Bristol-Myers Squibb). A human cytotoxic T-lymphocyte antigen (CTLA-4)-blocking antibody for the treatment of unresectable or metastatic melanoma. The first treatment proven to prolong survival in patients with metastatic melanoma.
Basis: An international study of 676 patients with previously treated unresectable stage III or IV melanoma whose disease had progressed, comparing ipilimumab plus an experimental tumor vaccine (gp100), gp100 alone, and ipilimumab alone. Median overall survival was 10 months among those who received ipilimumab with or without the vaccine, vs. 6 months among those who received the vaccine alone.
Addendum: Approved with a Risk Evaluation and Mitigation Strategy (REMS) program addressing life-threatening toxicity and deaths associated with ipilimumab in the study. Acts slowly, and some patients may have tumor progression before therapy becomes effective.
• Ruxolitinib (Jakafi oral tablets, Incyte Corp.). A Janus-associated kinase (JAK) inhibitor for the treatment of intermediate- or high-risk myelofibrosis, including primary myelofibrosis, post–polycythemia vera myelofibrosis, and post–essential thrombocythemia myelofibrosis. An orphan drug that is the first treatment approved for the rare blood disease – and the first approved JAK inhibitor.
Basis: Two randomized controlled trials of 528 patients with intermediate- or high-risk myelofibrosis. In one study, 42% of those on ruxolitinib had more than at least a 35% reduction in spleen volume vs. 1% of those on placebo at 24 weeks, and nearly half of those on treatment had achieved at least a 50% reduction in myelofibrosis symptoms, vs. 5% of those on placebo. In the second study, 29% of those on ruxolitinib had at least a 35% reduction in spleen size, vs. none of those on best available therapy after 48 weeks.
• Vandetanib (Caprelsa, AstraZeneca Pharmaceuticals LP). A kinase inhibitor for the treatment of symptomatic or progressive medullary thyroid cancer in adults with unresectable, locally advanced, or metastatic disease. The first drug approved for this rare cancer.
Basis: An international study of 331 patients with unresectable locally advanced or metastatic medullary thyroid cancer. Median progression-free survival was at least 22.6 months with vandetanib vs. 16.4 months among with placebo, a highly significant difference.
Addendum: Approved with a REMS addressing the risk of QT interval prolongation and a note that use in patients with indolent, asymptomatic, or slowly progressing disease "should be carefully considered" because of treatment-associated risks, as well as a boxed warning and a restricted distribution program.
• Vemurafenib (Zelboraf tablets, Hoffmann-LaRoche, Inc.). A BRAF inhibitor for the treatment of patients with unresectable or metastatic melanoma with the BRAFV600E mutation, as detected by the cobas 4800 BRAF V600 Mutation Test (Roche Molecular Systems), a companion diagnostic test approved at the same time. The second therapy found to prolong survival in patients with melanoma.
Basis: A study of 675 treatment-naive patients with unresectable or metastatic melanoma, positive for the BRAFV600E mutation. At the time of review, median survival was 7.9 months in a control group treated with dacarbazine, but had not been reached among those on vemurafenib. Median progression-free survival was 1.6 and 5.3 months, respectively.
Addendum: About half of melanoma patients have the BRAF mutation, and about half of them respond to vemurafenib. Approved with a medication guide to inform patients about the need for the diagnostic test and potential risks associated with treatment, including cutaneous squamous cell carcinoma. Roche and ipilimumab manufacturer Bristol-Meyers Squibb announced collaboration on a phase I/II study to determine whether combining the two agents is safe and effective in patients with BRAF mutations.
NEW DEVICES
• MelaFind (Mela Sciences). A noninvasive melanoma detection device for use "on clinically atypical cutaneous pigmented lesions with one or more clinical or historical characteristics of melanoma, excluding those with a clinical diagnosis of melanoma or likely melanoma."
Basis: The road to approval was controversial, with a split vote from an advisory panel, a citizens petition submitted by the manufacturer, and an agreement between the agency and manufacturer on how the device would be used and by whom.
• NovoTTF-100A System (NovoTTF, Novocure). A portable, battery-powered device that delivers electrical fields to the brain as a treatment for adults with glioblastoma multiforme that has recurred after chemotherapy.
Basis: In a randomized study of 237 patients whose GBM had recurred after surgery, radiation, and chemotherapy, median overall survival was 6.3 months with the approximately 6-pound device and 6.4 months with best available chemotherapy. The agency cited evidence suggesting better quality of life without chemotherapy side effects.
NEW INDICATIONS
• Cetuximab (Erbitux, ImClone LLC, a wholly owned subsidiary of Eli Lilly & Co. and Bristol-Myers Squibb). The epidermal growth factor receptor (EGFR) antagonist in combination with platinum-based therapy plus 5-fluorouracil for the first-line treatment of recurrent locoregional and/or metastatic squamous cell carcinoma of the head and neck.
Basis: A multicenter non-U.S. study of 442 patients not suitable for potentially curative treatment with surgery or radiation. Mean overall survival was 10.1 months with cetuximab and chemotherapy (cisplatin or carboplatin and 5-fluorouracil), vs. 7.4 months with chemotherapy alone.
Addendum: Cetuximab available in the United States provides about 22% greater exposure than the European Union–approved cetuximab used in this study, but the study results, pharmacokinetic data, and other clinical trial data "establish the efficacy" of cetuximab at the dose recommended, according to the prescribing information.
• Denosumab (Prolia, Amgen, Inc.). The RANK ligand (RANKL) inhibitor to increase bone mass in patients at high risk for fracture when receiving androgen deprivation therapy (ADT) for nonmetastatic prostate cancer or adjuvant aromatase inhibitor (AI) therapy for breast cancer.
Basis: Two international, randomized, double-blind placebo-controlled studies. In one study of 1,468 men with nonmetastatic prostate cancer receiving ADT, lumbar spine bone mineral density (BMD) at 2 years was significantly higher with denosumab vs. placebo (6.7% difference). At 3 years of treatment, the incidence of new vertebral fractures was 1.5% with denosumab vs. 3.9% with placebo, a risk reduction of 62%. In a study of 252 postmenopausal women with breast cancer on AI treatment, BMD at the lumbar spine was significantly higher with denosumab vs. placebo after 12 months of treatment (5.5% difference).
Addendum: Denosumab was initially approved in 2010. It is marketed as Prolia for treating postmenopausal women with osteoporosis at high risk of fracture, and as Xgeva for preventing skeletal-related events in patients with bone metastases from solid tumors; Xgeva is administered more frequently and at a higher dose for the latter indication.
• Everolimus (Afinitor Tablets, Novartis Pharmaceuticals Corp.). The mammalian target of rapamycin (mTOR) inhibitor for progressive pancreatic neuroendocrine tumors (pNETs) that are unresectable, locally advanced, or metastatic. (Safety and effectiveness of everolimus for treating carcinoid tumors have not been established). The first new drug in about 30 years for the treatment of advanced pNET.
Basis: A randomized controlled study of 410 patients, all of whom also received best supportive care. Median progression-free survival was 11 months with everolimus, vs. 4.6 months with placebo. In an interim analysis, overall survival was not different between the two groups.
• Peg-interferon alfa-2b (Sylatron, Schering Corp.). An alpha interferon for the adjuvant treatment of melanoma with microscopic or gross nodal involvement within 84 days of definitive surgical resection, including complete lymphadenectomy.
Basis: An open-label multicenter study of 1,256 patients. Median relapse-free survival was 34.8 months among those treated with peg-interferon alfa-2b, vs. 25.5 months with observation.
• Rituximab (Rituxan, Genentech, Inc.). The CD20-directed cytolytic antibody for maintenance therapy in people with previously untreated follicular, CD-20 positive, B-cell non-Hodgkin lymphoma who achieve a response to rituximab in combination with chemotherapy.
Basis: An international study of 1,217 previously untreated patients with advanced follicular lymphoma; it randomized 1,018 patients who had a complete or partial response with rituximab and chemotherapy. Maintenance treatment with rituximab reduced the risk of progression by 46% compared with observation only.
• Sunitinib (Sutent Capsules, Pfizer, Inc.). A kinase inhibitor for the treatment of progressive, well-differentiated pancreatic neuroendocrine tumors (pNETs) in patients with unresectable, locally advanced, or metastatic disease; it was approved soon after everolimus for this indication.
Basis: A randomized controlled study of 171 patients with unresectable, locally advanced or well-differentiated pNET; treatment with somatostatin analogues was allowed. Median progression-free survival was 10.2 months with sunitinib, vs. 5.4 months with placebo.
WARNINGS AND LABEL CHANGES
• Bevacizumab (Avastin, Genentech, Inc.). Revised prescribing information includes the risk of ovarian failure in premenopausal women treated with bevacizumab and chemotherapy; postmarketing reports of osteonecrosis of the jaw in patients treated with bevacizumab but not bisphosphonates; and information from a clinical trial about the risk of venous thromboembolic events and bleeding in patients receiving anticoagulation therapy after a first VTE event while receiving bevacizumab.
• Dasatinib (Sprycel, Bristol-Myers Squibb). Information on increased risk for pulmonary arterial hypertension added to prescribing information.
• Fentanyl. A classwide risk management program approved for all transmucosal immediate-release fentanyl formulations.
• IV methotrexate and proton pump inhibitors (PPIs). A warning added to the methotrexate label advises caution when high-dose methotrexate is administered to patients taking a PPI, because concomitant administration may result in elevated and prolonged serum levels of methotrexate and/or its metabolite hydroxymethotrexate, with possible toxic effects.
• Lenalidomide (Revlimid, Celgene). Advisory informed the public that the FDA is monitoring the risk of secondary cancers in patients treated for myelodysplastic syndromes or multiple myeloma.
• Ondansetron (Zofran, GlaxoSmithKline, and generic formulations). Following a review of information on the risk of QT prolongation, label now recommends ECG monitoring in patients who are treated with ondansetron and have an electrolyte abnormality, congestive heart failure, or bradyarrhythmias; and in patients taking other medications that prolong the QT interval – who are at an increased risk of developing torsades de pointes. Label recommends against use in patients with congenital long QT syndrome. Safety review ongoing.
• Opioids. Draft issued for opioid prescriber education program, as part of the Risk Evaluation and Mitigation Strategy (REMS) now in place for brand-name and generic long-acting and extended-release opioids.
• Pioglitazone. Bladder cancer warning risk added to the label for type 2 diabetes drug marketed as Actos (or in combination with metformin as Actoplus Met, and with glimepiride as Duetact).
• Romiplostim (Nplate for subcutaneous injection, Amgen, Inc.) and eltrombopag (Promacta tablets, GlaxoSmithKline LLC). Elements of the REMS for the two thrombopoietin receptor agonists were dropped, including the restricted distribution program that required health care professionals, hospitals, and patients to be enrolled in order to prescribe, dispense, or receive these drugs. Clinicians no longer need to file periodic safety forms for their patients on these treatments.
• Tumor necrosis factor (TNF) blockers. Manufacturers asked to report cases of malignancies in children, adolescents, and adults aged 30 years and younger treated with TNF blockers and to conduct in-depth follow-up of them.
OTHER ACTIONS
• Bevacizumab (Avastin, Genentech, Inc.). FDA Commissioner Margaret Hamburg withdrew bevacizumab’s indication in metastatic breast cancer. She cited a lack of data establishing benefit and serious risks associated with treatment. The decision followed a July hearing, where the Oncologic Drugs Advisory Committee unanimously voted that the indication be withdrawn despite emotional testimony from patients who said they benefited from the monoclonal antibody.
• Drug shortages. A new rule requires some manufacturers to give early warning of shortages. The agency also held a workshop on the crisis and is working to avert and/or resolve shortages.
• Office of Oncology Products. Many New Drug Applications, Biologic License Applications, and Investigational New Drug Applications for cancer drugs are being reassigned to new review divisions as part of a reorganization.
Jaw Complications Persist With Head & Neck Cancer Treatment
PHOENIX – An analysis of Medicare data for more than 1,800 head and neck cancer patients suggests that jaw complications following radiation treatment may be more common than recognized.
Moreover, intensity-modulated radiation therapy (IMRT) was not associated with significantly lower rates of jaw complications, compared with older radiation techniques, although there was a slight trend in that direction and the interval to developing jaw complications was longer following IMRT, reported Dr. Beth M. Beadle at a head and neck cancer symposium sponsored by the American Society for Radiation Oncology.
In the literature, osteoradionecrosis (ORN), the most severe jaw manifestation resulting from head/neck radiation, was reported on average in 11.8% of patients in 10 studies (total, 3,312 irradiated patients) from the 1930s through the 1960s, said Dr. Beadle, a radiation oncologist at the University of Texas M.D. Anderson Cancer Center, Houston.
With improved technologies, the rate dropped to 5.4%, as documented in 21 studies involving a total of 11,077 patients conducted from the 1970s through the early 1990s. Since 1997, 25 retrospective and prospective studies involving 9,632 patients overall have reported an overall average ORN rate of 3.0%, she said.
For the current study, data were taken from the SEER (Surveillance, Epidemiology, and End Results) database for Medicare beneficiaries. Patients who were diagnosed with oral cancers in 1999-2007 were identified using SEER, ICD-9, and CPT codes. Primary tumor sites included those of the lip, tongue, floor of mouth, gum, tonsil, oropharynx, and other oral cavity and pharynx.
Of 1,848 patients overall, 16.1% (297) had at least one osteoradionecrosis code within 90 days of radiation therapy completion. Of those, 256 patients had ICD-9 diagnostic codes that included those for osteonecrosis of jaw (733.45), osteomyelitis of jaw (526.4), and other diseases of the jaws, including inflammatory conditions, alveolitis, and periradicular pathology (all 526.x).
Some 41 patients (3.8% of the total) had CPT procedure codes, including drainage of abscess (41800), alveolectomy (41830), operations on facial bones or joints with concurrent ORN diagnosis (76.3), or hyperbaric oxygen therapy (99183), and 30 patients (1.6% of the total) had both a diagnostic and a procedural ORN code, Dr. Beadle said.
The percentage of patients who required intervention for ORN (3.8%) is more consistent with the overall ORN rates from the literature, suggesting that most previous studies have captured only the more severe osteoradionecrosis, which is usually defined as that requiring a procedure, she explained.
In univariate analysis, female sex, not receiving chemotherapy, and a lower number of comorbidities on the Charlson Comorbidity Index were the only factors significantly associated with all jaw complications.
Receipt of IMRT was not a significant predictor, although there was a trend. Patients who received IMRT differed significantly from non-IMRT patients in several ways, however, including their younger age and their greater likelihood to be male, to have advanced-stage disease, to have received definitive vs. adjuvant treatment, to have received chemotherapy, and to have fewer comorbidities.
Although IMRT per se did not predict jaw complications, there was a longer time interval between treatment and the development of jaw complications among the patients who received IMRT (462 days vs. 386 days for the non-IMRT patient).
In response to questions from the audience, Dr. Beadle said that it wasn’t clear why jaw complications would be more common among patients who did not receive chemotherapy, but it might be that those patients received higher doses of radiation. As for the sex difference, she suggested it may be that women have better follow-up care, or it might relate to either osteoporosis or use of bisphosphonates. Her group is investigating those possibilities.
Dr. Beadle stated that she has no disclosures.
PHOENIX – An analysis of Medicare data for more than 1,800 head and neck cancer patients suggests that jaw complications following radiation treatment may be more common than recognized.
Moreover, intensity-modulated radiation therapy (IMRT) was not associated with significantly lower rates of jaw complications, compared with older radiation techniques, although there was a slight trend in that direction and the interval to developing jaw complications was longer following IMRT, reported Dr. Beth M. Beadle at a head and neck cancer symposium sponsored by the American Society for Radiation Oncology.
In the literature, osteoradionecrosis (ORN), the most severe jaw manifestation resulting from head/neck radiation, was reported on average in 11.8% of patients in 10 studies (total, 3,312 irradiated patients) from the 1930s through the 1960s, said Dr. Beadle, a radiation oncologist at the University of Texas M.D. Anderson Cancer Center, Houston.
With improved technologies, the rate dropped to 5.4%, as documented in 21 studies involving a total of 11,077 patients conducted from the 1970s through the early 1990s. Since 1997, 25 retrospective and prospective studies involving 9,632 patients overall have reported an overall average ORN rate of 3.0%, she said.
For the current study, data were taken from the SEER (Surveillance, Epidemiology, and End Results) database for Medicare beneficiaries. Patients who were diagnosed with oral cancers in 1999-2007 were identified using SEER, ICD-9, and CPT codes. Primary tumor sites included those of the lip, tongue, floor of mouth, gum, tonsil, oropharynx, and other oral cavity and pharynx.
Of 1,848 patients overall, 16.1% (297) had at least one osteoradionecrosis code within 90 days of radiation therapy completion. Of those, 256 patients had ICD-9 diagnostic codes that included those for osteonecrosis of jaw (733.45), osteomyelitis of jaw (526.4), and other diseases of the jaws, including inflammatory conditions, alveolitis, and periradicular pathology (all 526.x).
Some 41 patients (3.8% of the total) had CPT procedure codes, including drainage of abscess (41800), alveolectomy (41830), operations on facial bones or joints with concurrent ORN diagnosis (76.3), or hyperbaric oxygen therapy (99183), and 30 patients (1.6% of the total) had both a diagnostic and a procedural ORN code, Dr. Beadle said.
The percentage of patients who required intervention for ORN (3.8%) is more consistent with the overall ORN rates from the literature, suggesting that most previous studies have captured only the more severe osteoradionecrosis, which is usually defined as that requiring a procedure, she explained.
In univariate analysis, female sex, not receiving chemotherapy, and a lower number of comorbidities on the Charlson Comorbidity Index were the only factors significantly associated with all jaw complications.
Receipt of IMRT was not a significant predictor, although there was a trend. Patients who received IMRT differed significantly from non-IMRT patients in several ways, however, including their younger age and their greater likelihood to be male, to have advanced-stage disease, to have received definitive vs. adjuvant treatment, to have received chemotherapy, and to have fewer comorbidities.
Although IMRT per se did not predict jaw complications, there was a longer time interval between treatment and the development of jaw complications among the patients who received IMRT (462 days vs. 386 days for the non-IMRT patient).
In response to questions from the audience, Dr. Beadle said that it wasn’t clear why jaw complications would be more common among patients who did not receive chemotherapy, but it might be that those patients received higher doses of radiation. As for the sex difference, she suggested it may be that women have better follow-up care, or it might relate to either osteoporosis or use of bisphosphonates. Her group is investigating those possibilities.
Dr. Beadle stated that she has no disclosures.
PHOENIX – An analysis of Medicare data for more than 1,800 head and neck cancer patients suggests that jaw complications following radiation treatment may be more common than recognized.
Moreover, intensity-modulated radiation therapy (IMRT) was not associated with significantly lower rates of jaw complications, compared with older radiation techniques, although there was a slight trend in that direction and the interval to developing jaw complications was longer following IMRT, reported Dr. Beth M. Beadle at a head and neck cancer symposium sponsored by the American Society for Radiation Oncology.
In the literature, osteoradionecrosis (ORN), the most severe jaw manifestation resulting from head/neck radiation, was reported on average in 11.8% of patients in 10 studies (total, 3,312 irradiated patients) from the 1930s through the 1960s, said Dr. Beadle, a radiation oncologist at the University of Texas M.D. Anderson Cancer Center, Houston.
With improved technologies, the rate dropped to 5.4%, as documented in 21 studies involving a total of 11,077 patients conducted from the 1970s through the early 1990s. Since 1997, 25 retrospective and prospective studies involving 9,632 patients overall have reported an overall average ORN rate of 3.0%, she said.
For the current study, data were taken from the SEER (Surveillance, Epidemiology, and End Results) database for Medicare beneficiaries. Patients who were diagnosed with oral cancers in 1999-2007 were identified using SEER, ICD-9, and CPT codes. Primary tumor sites included those of the lip, tongue, floor of mouth, gum, tonsil, oropharynx, and other oral cavity and pharynx.
Of 1,848 patients overall, 16.1% (297) had at least one osteoradionecrosis code within 90 days of radiation therapy completion. Of those, 256 patients had ICD-9 diagnostic codes that included those for osteonecrosis of jaw (733.45), osteomyelitis of jaw (526.4), and other diseases of the jaws, including inflammatory conditions, alveolitis, and periradicular pathology (all 526.x).
Some 41 patients (3.8% of the total) had CPT procedure codes, including drainage of abscess (41800), alveolectomy (41830), operations on facial bones or joints with concurrent ORN diagnosis (76.3), or hyperbaric oxygen therapy (99183), and 30 patients (1.6% of the total) had both a diagnostic and a procedural ORN code, Dr. Beadle said.
The percentage of patients who required intervention for ORN (3.8%) is more consistent with the overall ORN rates from the literature, suggesting that most previous studies have captured only the more severe osteoradionecrosis, which is usually defined as that requiring a procedure, she explained.
In univariate analysis, female sex, not receiving chemotherapy, and a lower number of comorbidities on the Charlson Comorbidity Index were the only factors significantly associated with all jaw complications.
Receipt of IMRT was not a significant predictor, although there was a trend. Patients who received IMRT differed significantly from non-IMRT patients in several ways, however, including their younger age and their greater likelihood to be male, to have advanced-stage disease, to have received definitive vs. adjuvant treatment, to have received chemotherapy, and to have fewer comorbidities.
Although IMRT per se did not predict jaw complications, there was a longer time interval between treatment and the development of jaw complications among the patients who received IMRT (462 days vs. 386 days for the non-IMRT patient).
In response to questions from the audience, Dr. Beadle said that it wasn’t clear why jaw complications would be more common among patients who did not receive chemotherapy, but it might be that those patients received higher doses of radiation. As for the sex difference, she suggested it may be that women have better follow-up care, or it might relate to either osteoporosis or use of bisphosphonates. Her group is investigating those possibilities.
Dr. Beadle stated that she has no disclosures.
FROM A HEAD AND NECK CANCER SYMPOSIUM SPONSORED BY THE AMERICAN SOCIETY FOR RADIATION ONCOLOGY
Major Finding: Of a total 1,848 patients with oral cancers, 16% (297) had at least one osteoradionecrosis code within 90 days of radiation therapy completion.
Data source: Data were taken from the SEER database for Medicare beneficiaries.
Disclosures: Dr. Beadle reported having no financial disclosures.
ASCO Urges Early Palliative Care in Metastatic Cancers
Compelling evidence from a recent randomized trial has prompted the American Society for Clinical Oncology to recommend that palliative care be integrated early on into standard cancer therapies for patients with metastatic cancers or a high burden of cancer symptoms.
Potentially practice changing, the opinion is based on the best currently available clinical evidence. Palliative care is typically relegated to the final days of life of patients with advanced metastatic cancers, as it is provided only after all other options have failed.
Authors of the opinion, intended to offer guidance to oncologists on this issue, cite a study published in 2010 (N. Engl. J. Med. 2010;363:733-42). The study showed that patients who were randomized to palliative care plus standard therapy for metastatic non–small cell lung cancer (NSCLC) had significantly longer overall survival than did patients randomized to standard care alone, (11.6 vs. 8.9 months, P = .02), even though the palliative care group had less aggressive end-of-life care.
Patients receiving palliative care also had significantly better quality of life scores on a standardized assessment scale, and significantly fewer had depressive symptoms, compared with patients on standard care.
"While a survival benefit from early involvement of palliative care has not yet been demonstrated in other oncology settings, substantial evidence demonstrates that palliative care – when combined with standard cancer care or as the main focus of care – leads to better patient and caregiver outcomes. These include improvement in symptoms, quality of life, and patient satisfaction, with reduced caregiver burden," wrote Dr. Thomas J. Smith and his colleagues in an American Society of Clinical Oncology (ASCO) provisional clinical opinion published online Feb. 6 in the Journal of Clinical Oncology (doi:10.1200/JCO.2011.38.5161).
Palliative care also eases patients and families through the anguish of dashed hopes and has the potential to reduce costs by limiting expensive but often futile intensive hospital-based services, the authors wrote.
"All the data suggest that there’s absolutely no harm from earlier integration of hospice and palliative medicine into patient care. A couple of trials have shown improved survival, and there are very good data from observational studies that people who use hospice actually live longer," Dr. Smith said in an interview. He is director of palliative care for Johns Hopkins University and Hopkins’ Sidney Kimmel Comprehensive Cancer Center in Baltimore.
"I think that ASCO is sending a really strong message to oncologists that we need to do more than we’re currently doing and that comprehensive cancer care needs to included supportive care on top of cancer-directed therapy," said Dr. Jennifer S. Temel, clinical director of thoracic oncology at Massachusetts General Hospital in Boston, and lead author of the randomized trial mentioned earlier.
She noted, however, that the study was not powered to detect an overall survival benefit. "All we were hoping for was that early palliative care didn’t lead to a survival detriment ... people could have been concerned that because of the involvement of palliative care, patients would receive less-intensive therapy and have shorter survival," Dr. Temel said. "I’m just very happy that’s not what we saw, but whether the survival benefit we saw was real and will be replicated, we'll have to wait and see."
The primary value of the study, she added, is that it demonstrated distinct benefits of palliative care on patient mood and quality of life.
The Will but Not the Way?
But many oncology practices, particularly those in community settings, may not have the resources to provide a full complement of palliative care services, said an oncologist in community-based practice.
"Those types of palliative care options are not widely available, and they certainly aren’t available in smaller communities," said Dr. Patrick Cobb, managing partner at the Frontier Cancer Center in Billings, Mont.
Services required for effective palliative care, such as patient and family counseling, are not typically reimbursed under current payments systems. In addition, palliative care reimbursement is often an "either/or" proposition: insurers pay for either therapeutic services or hospice care, but not both, said Dr. Cobb, former president of the Community Oncology Alliance.
He added that the so-called Stark law – actually a set of provisions in federal law governing the ability of clinicians to refer patients to clinical or diagnostic facilities in which the clinician has a financial interest – is another barrier to palliative care in the community, particularly in rural areas where the population may not be large enough to support separate palliative care facilities or programs.
Dr. Amy P. Abernethy, medical director of oncology quality, outcomes and patient-centered care in the Duke University Health System, Durham, N.C., and a coauthor of the ASCO provisional clinical opinion, agrees that there are multiple impediments to reimbursement of palliative care.
"The Stark law is one impediment; a second is that the reimbursement mechanisms that are clear in hospice aren’t necessarily as clear in community-based care, and then there are workforce issues. Right now, we have only a finite number of palliative care practitioners, and we only have a finite number of blocks in our graduate training programs, and we're not going to be able, using those slots, to train enough palliative care docs to fill the need that's highlighted in this provisional clinical opinion," she said.
Insurers, Younger Clinicians May Be Open to Change
Insurers seem to be coming around to the idea that palliative care can mean better patient care, however, said Dr Smith.
He points to Aetna, which has a "Compassionate Care" program in which specially trained triage nurses coordinate care, identify resources, and help manage palliative care and hospice benefits for patients with terminal illnesses and their families.
Clinicians in training or new to practice are also more comfortable with the idea of advance directives, palliative care, and hospice than are their more seasoned colleagues who were trained to never give up, Dr. Smith added.
Dr. Abernethy agreed: "What we’re seeing is that young physicians totally get this. Probably because they haven’t grown up in a world where the only thing you focus on is survival, they’ve understood the language of focusing on quality of life from the time they were first exposed to what medicine is," she said.
Randomized Trials Show Benefits, No Harm
In their provisional opinion, the researchers reviewed the study by Dr. Temel and her colleagues, as well as six other randomized controlled trials looking at palliative care in patients with various terminal illnesses; two of the seven total studies evaluated palliative care in cancer patients exclusively, whereas others included diagnoses such as heart failure and advanced chronic obstructive pulmonary disease.
They found that "overall, the addition of palliative care interventions to standard oncology care delivered via different models to patients with cancer provided evidence of benefit. No harm to any patient was observed in any trial, even with discussions of end-of-life planning, such as hospice and advance directives."
There were statistically significant improvements in symptoms with palliative care in 2 of 5 clinical trials that measured such changes, and improvements in quality-of-life measures in 2 of 5 trials. Additionally, in 2 of 3 trials palliative care was associated with improved satisfaction of patients and caregivers, the consensus panel found.
The studies also showed, to varying degrees, improvements in patient mood and a reduction in costs, in one study (J. Am. Geriatr. Soc. 2007;55:993-1000) from $20,222 for usual care to $12,670 for palliative care (P = .03), and in a second study (J. Palliat. Med. 2008;11:180-90) from a total mean of $21,252 for usual care to $14,486 for interdisciplinary palliative care (P less than .001). The latter study also found savings of nearly $5,000 per patient in staffing costs with palliative care.
"Therefore, most trials showed benefits ranging from equal to improved overall survival, reduced depression, improved caregiver and/or patient quality of life, and overall lower resource use and cost because end-of-life hospitalizations were avoided," the opinion authors wrote.
All physicians interviewed for this article reported that they did not have financial conflicts of interest.
Click here to see earlier coverage of this subject and a video of Dr. Temel discussing results of the randomized trial.
Compelling evidence from a recent randomized trial has prompted the American Society for Clinical Oncology to recommend that palliative care be integrated early on into standard cancer therapies for patients with metastatic cancers or a high burden of cancer symptoms.
Potentially practice changing, the opinion is based on the best currently available clinical evidence. Palliative care is typically relegated to the final days of life of patients with advanced metastatic cancers, as it is provided only after all other options have failed.
Authors of the opinion, intended to offer guidance to oncologists on this issue, cite a study published in 2010 (N. Engl. J. Med. 2010;363:733-42). The study showed that patients who were randomized to palliative care plus standard therapy for metastatic non–small cell lung cancer (NSCLC) had significantly longer overall survival than did patients randomized to standard care alone, (11.6 vs. 8.9 months, P = .02), even though the palliative care group had less aggressive end-of-life care.
Patients receiving palliative care also had significantly better quality of life scores on a standardized assessment scale, and significantly fewer had depressive symptoms, compared with patients on standard care.
"While a survival benefit from early involvement of palliative care has not yet been demonstrated in other oncology settings, substantial evidence demonstrates that palliative care – when combined with standard cancer care or as the main focus of care – leads to better patient and caregiver outcomes. These include improvement in symptoms, quality of life, and patient satisfaction, with reduced caregiver burden," wrote Dr. Thomas J. Smith and his colleagues in an American Society of Clinical Oncology (ASCO) provisional clinical opinion published online Feb. 6 in the Journal of Clinical Oncology (doi:10.1200/JCO.2011.38.5161).
Palliative care also eases patients and families through the anguish of dashed hopes and has the potential to reduce costs by limiting expensive but often futile intensive hospital-based services, the authors wrote.
"All the data suggest that there’s absolutely no harm from earlier integration of hospice and palliative medicine into patient care. A couple of trials have shown improved survival, and there are very good data from observational studies that people who use hospice actually live longer," Dr. Smith said in an interview. He is director of palliative care for Johns Hopkins University and Hopkins’ Sidney Kimmel Comprehensive Cancer Center in Baltimore.
"I think that ASCO is sending a really strong message to oncologists that we need to do more than we’re currently doing and that comprehensive cancer care needs to included supportive care on top of cancer-directed therapy," said Dr. Jennifer S. Temel, clinical director of thoracic oncology at Massachusetts General Hospital in Boston, and lead author of the randomized trial mentioned earlier.
She noted, however, that the study was not powered to detect an overall survival benefit. "All we were hoping for was that early palliative care didn’t lead to a survival detriment ... people could have been concerned that because of the involvement of palliative care, patients would receive less-intensive therapy and have shorter survival," Dr. Temel said. "I’m just very happy that’s not what we saw, but whether the survival benefit we saw was real and will be replicated, we'll have to wait and see."
The primary value of the study, she added, is that it demonstrated distinct benefits of palliative care on patient mood and quality of life.
The Will but Not the Way?
But many oncology practices, particularly those in community settings, may not have the resources to provide a full complement of palliative care services, said an oncologist in community-based practice.
"Those types of palliative care options are not widely available, and they certainly aren’t available in smaller communities," said Dr. Patrick Cobb, managing partner at the Frontier Cancer Center in Billings, Mont.
Services required for effective palliative care, such as patient and family counseling, are not typically reimbursed under current payments systems. In addition, palliative care reimbursement is often an "either/or" proposition: insurers pay for either therapeutic services or hospice care, but not both, said Dr. Cobb, former president of the Community Oncology Alliance.
He added that the so-called Stark law – actually a set of provisions in federal law governing the ability of clinicians to refer patients to clinical or diagnostic facilities in which the clinician has a financial interest – is another barrier to palliative care in the community, particularly in rural areas where the population may not be large enough to support separate palliative care facilities or programs.
Dr. Amy P. Abernethy, medical director of oncology quality, outcomes and patient-centered care in the Duke University Health System, Durham, N.C., and a coauthor of the ASCO provisional clinical opinion, agrees that there are multiple impediments to reimbursement of palliative care.
"The Stark law is one impediment; a second is that the reimbursement mechanisms that are clear in hospice aren’t necessarily as clear in community-based care, and then there are workforce issues. Right now, we have only a finite number of palliative care practitioners, and we only have a finite number of blocks in our graduate training programs, and we're not going to be able, using those slots, to train enough palliative care docs to fill the need that's highlighted in this provisional clinical opinion," she said.
Insurers, Younger Clinicians May Be Open to Change
Insurers seem to be coming around to the idea that palliative care can mean better patient care, however, said Dr Smith.
He points to Aetna, which has a "Compassionate Care" program in which specially trained triage nurses coordinate care, identify resources, and help manage palliative care and hospice benefits for patients with terminal illnesses and their families.
Clinicians in training or new to practice are also more comfortable with the idea of advance directives, palliative care, and hospice than are their more seasoned colleagues who were trained to never give up, Dr. Smith added.
Dr. Abernethy agreed: "What we’re seeing is that young physicians totally get this. Probably because they haven’t grown up in a world where the only thing you focus on is survival, they’ve understood the language of focusing on quality of life from the time they were first exposed to what medicine is," she said.
Randomized Trials Show Benefits, No Harm
In their provisional opinion, the researchers reviewed the study by Dr. Temel and her colleagues, as well as six other randomized controlled trials looking at palliative care in patients with various terminal illnesses; two of the seven total studies evaluated palliative care in cancer patients exclusively, whereas others included diagnoses such as heart failure and advanced chronic obstructive pulmonary disease.
They found that "overall, the addition of palliative care interventions to standard oncology care delivered via different models to patients with cancer provided evidence of benefit. No harm to any patient was observed in any trial, even with discussions of end-of-life planning, such as hospice and advance directives."
There were statistically significant improvements in symptoms with palliative care in 2 of 5 clinical trials that measured such changes, and improvements in quality-of-life measures in 2 of 5 trials. Additionally, in 2 of 3 trials palliative care was associated with improved satisfaction of patients and caregivers, the consensus panel found.
The studies also showed, to varying degrees, improvements in patient mood and a reduction in costs, in one study (J. Am. Geriatr. Soc. 2007;55:993-1000) from $20,222 for usual care to $12,670 for palliative care (P = .03), and in a second study (J. Palliat. Med. 2008;11:180-90) from a total mean of $21,252 for usual care to $14,486 for interdisciplinary palliative care (P less than .001). The latter study also found savings of nearly $5,000 per patient in staffing costs with palliative care.
"Therefore, most trials showed benefits ranging from equal to improved overall survival, reduced depression, improved caregiver and/or patient quality of life, and overall lower resource use and cost because end-of-life hospitalizations were avoided," the opinion authors wrote.
All physicians interviewed for this article reported that they did not have financial conflicts of interest.
Click here to see earlier coverage of this subject and a video of Dr. Temel discussing results of the randomized trial.
Compelling evidence from a recent randomized trial has prompted the American Society for Clinical Oncology to recommend that palliative care be integrated early on into standard cancer therapies for patients with metastatic cancers or a high burden of cancer symptoms.
Potentially practice changing, the opinion is based on the best currently available clinical evidence. Palliative care is typically relegated to the final days of life of patients with advanced metastatic cancers, as it is provided only after all other options have failed.
Authors of the opinion, intended to offer guidance to oncologists on this issue, cite a study published in 2010 (N. Engl. J. Med. 2010;363:733-42). The study showed that patients who were randomized to palliative care plus standard therapy for metastatic non–small cell lung cancer (NSCLC) had significantly longer overall survival than did patients randomized to standard care alone, (11.6 vs. 8.9 months, P = .02), even though the palliative care group had less aggressive end-of-life care.
Patients receiving palliative care also had significantly better quality of life scores on a standardized assessment scale, and significantly fewer had depressive symptoms, compared with patients on standard care.
"While a survival benefit from early involvement of palliative care has not yet been demonstrated in other oncology settings, substantial evidence demonstrates that palliative care – when combined with standard cancer care or as the main focus of care – leads to better patient and caregiver outcomes. These include improvement in symptoms, quality of life, and patient satisfaction, with reduced caregiver burden," wrote Dr. Thomas J. Smith and his colleagues in an American Society of Clinical Oncology (ASCO) provisional clinical opinion published online Feb. 6 in the Journal of Clinical Oncology (doi:10.1200/JCO.2011.38.5161).
Palliative care also eases patients and families through the anguish of dashed hopes and has the potential to reduce costs by limiting expensive but often futile intensive hospital-based services, the authors wrote.
"All the data suggest that there’s absolutely no harm from earlier integration of hospice and palliative medicine into patient care. A couple of trials have shown improved survival, and there are very good data from observational studies that people who use hospice actually live longer," Dr. Smith said in an interview. He is director of palliative care for Johns Hopkins University and Hopkins’ Sidney Kimmel Comprehensive Cancer Center in Baltimore.
"I think that ASCO is sending a really strong message to oncologists that we need to do more than we’re currently doing and that comprehensive cancer care needs to included supportive care on top of cancer-directed therapy," said Dr. Jennifer S. Temel, clinical director of thoracic oncology at Massachusetts General Hospital in Boston, and lead author of the randomized trial mentioned earlier.
She noted, however, that the study was not powered to detect an overall survival benefit. "All we were hoping for was that early palliative care didn’t lead to a survival detriment ... people could have been concerned that because of the involvement of palliative care, patients would receive less-intensive therapy and have shorter survival," Dr. Temel said. "I’m just very happy that’s not what we saw, but whether the survival benefit we saw was real and will be replicated, we'll have to wait and see."
The primary value of the study, she added, is that it demonstrated distinct benefits of palliative care on patient mood and quality of life.
The Will but Not the Way?
But many oncology practices, particularly those in community settings, may not have the resources to provide a full complement of palliative care services, said an oncologist in community-based practice.
"Those types of palliative care options are not widely available, and they certainly aren’t available in smaller communities," said Dr. Patrick Cobb, managing partner at the Frontier Cancer Center in Billings, Mont.
Services required for effective palliative care, such as patient and family counseling, are not typically reimbursed under current payments systems. In addition, palliative care reimbursement is often an "either/or" proposition: insurers pay for either therapeutic services or hospice care, but not both, said Dr. Cobb, former president of the Community Oncology Alliance.
He added that the so-called Stark law – actually a set of provisions in federal law governing the ability of clinicians to refer patients to clinical or diagnostic facilities in which the clinician has a financial interest – is another barrier to palliative care in the community, particularly in rural areas where the population may not be large enough to support separate palliative care facilities or programs.
Dr. Amy P. Abernethy, medical director of oncology quality, outcomes and patient-centered care in the Duke University Health System, Durham, N.C., and a coauthor of the ASCO provisional clinical opinion, agrees that there are multiple impediments to reimbursement of palliative care.
"The Stark law is one impediment; a second is that the reimbursement mechanisms that are clear in hospice aren’t necessarily as clear in community-based care, and then there are workforce issues. Right now, we have only a finite number of palliative care practitioners, and we only have a finite number of blocks in our graduate training programs, and we're not going to be able, using those slots, to train enough palliative care docs to fill the need that's highlighted in this provisional clinical opinion," she said.
Insurers, Younger Clinicians May Be Open to Change
Insurers seem to be coming around to the idea that palliative care can mean better patient care, however, said Dr Smith.
He points to Aetna, which has a "Compassionate Care" program in which specially trained triage nurses coordinate care, identify resources, and help manage palliative care and hospice benefits for patients with terminal illnesses and their families.
Clinicians in training or new to practice are also more comfortable with the idea of advance directives, palliative care, and hospice than are their more seasoned colleagues who were trained to never give up, Dr. Smith added.
Dr. Abernethy agreed: "What we’re seeing is that young physicians totally get this. Probably because they haven’t grown up in a world where the only thing you focus on is survival, they’ve understood the language of focusing on quality of life from the time they were first exposed to what medicine is," she said.
Randomized Trials Show Benefits, No Harm
In their provisional opinion, the researchers reviewed the study by Dr. Temel and her colleagues, as well as six other randomized controlled trials looking at palliative care in patients with various terminal illnesses; two of the seven total studies evaluated palliative care in cancer patients exclusively, whereas others included diagnoses such as heart failure and advanced chronic obstructive pulmonary disease.
They found that "overall, the addition of palliative care interventions to standard oncology care delivered via different models to patients with cancer provided evidence of benefit. No harm to any patient was observed in any trial, even with discussions of end-of-life planning, such as hospice and advance directives."
There were statistically significant improvements in symptoms with palliative care in 2 of 5 clinical trials that measured such changes, and improvements in quality-of-life measures in 2 of 5 trials. Additionally, in 2 of 3 trials palliative care was associated with improved satisfaction of patients and caregivers, the consensus panel found.
The studies also showed, to varying degrees, improvements in patient mood and a reduction in costs, in one study (J. Am. Geriatr. Soc. 2007;55:993-1000) from $20,222 for usual care to $12,670 for palliative care (P = .03), and in a second study (J. Palliat. Med. 2008;11:180-90) from a total mean of $21,252 for usual care to $14,486 for interdisciplinary palliative care (P less than .001). The latter study also found savings of nearly $5,000 per patient in staffing costs with palliative care.
"Therefore, most trials showed benefits ranging from equal to improved overall survival, reduced depression, improved caregiver and/or patient quality of life, and overall lower resource use and cost because end-of-life hospitalizations were avoided," the opinion authors wrote.
All physicians interviewed for this article reported that they did not have financial conflicts of interest.
Click here to see earlier coverage of this subject and a video of Dr. Temel discussing results of the randomized trial.
Survivorship: Evaluating needs and the integrated model
Who is a survivor? Am I a survivor? Are you a survivor? What does a survivor need? How can community oncologists help? These are among the many questions a community oncologist can expect to hear during the course of treating a cancer patient, from the diagnosis, through the decisions about therapy, at all stages of treatment, and well into aftercare and follow-up. A “patient” used to be someone in active treatment, and a “survivor” was someone who had been free of disease for 5 years. More recently, Ellen Stoval of the National Coalition for Cancer Survivorship noted that the term “cancer survivor” can be used “to describe anyone who has been diagnosed with cancer as well as caregivers and loved ones of those diagnosed with the disease.”1 Her broader perspective more accurately captures the multifaceted nature of survivorship...
*For a PDF of the full article, click on the link to the left of this introduction.
Who is a survivor? Am I a survivor? Are you a survivor? What does a survivor need? How can community oncologists help? These are among the many questions a community oncologist can expect to hear during the course of treating a cancer patient, from the diagnosis, through the decisions about therapy, at all stages of treatment, and well into aftercare and follow-up. A “patient” used to be someone in active treatment, and a “survivor” was someone who had been free of disease for 5 years. More recently, Ellen Stoval of the National Coalition for Cancer Survivorship noted that the term “cancer survivor” can be used “to describe anyone who has been diagnosed with cancer as well as caregivers and loved ones of those diagnosed with the disease.”1 Her broader perspective more accurately captures the multifaceted nature of survivorship...
*For a PDF of the full article, click on the link to the left of this introduction.
Who is a survivor? Am I a survivor? Are you a survivor? What does a survivor need? How can community oncologists help? These are among the many questions a community oncologist can expect to hear during the course of treating a cancer patient, from the diagnosis, through the decisions about therapy, at all stages of treatment, and well into aftercare and follow-up. A “patient” used to be someone in active treatment, and a “survivor” was someone who had been free of disease for 5 years. More recently, Ellen Stoval of the National Coalition for Cancer Survivorship noted that the term “cancer survivor” can be used “to describe anyone who has been diagnosed with cancer as well as caregivers and loved ones of those diagnosed with the disease.”1 Her broader perspective more accurately captures the multifaceted nature of survivorship...
*For a PDF of the full article, click on the link to the left of this introduction.