User login
The Role of Spirituality and Religious Coping in the Quality of Life of Patients With Advanced Cancer Receiving Palliative Radiation Therapy
The Role of Spirituality and Religious Coping in the Quality of Life of Patients With Advanced Cancer Receiving Palliative Radiation Therapy
Abstract
Objectives
National palliative care guidelines outline spiritual care as a domain of palliative care, yet patients' religiousness and/or spirituality (R/S) are underappreciated in the palliative oncology setting. Among patients with advanced cancer receiving palliative radiation therapy (RT), this study aims to characterize patient spirituality, religiousness, and religious coping; examine the relationships of these variables to quality of life (QOL); and assess patients' perceptions of spiritual care in the cancer care setting.
Methods
This is a multisite, cross-sectional survey of 69 patients with advanced cancer (response rate = 73%) receiving palliative RT. Scripted interviews assessed patient spirituality, religiousness, religious coping, QOL (McGill QOL Questionnaire), and perceptions of the importance of attention to spiritual needs by health providers. Multivariable models assessed the relationships of patient spirituality and R/S coping to patient QOL, controlling for other significant predictors of QOL.
Results
Most participants (84%) indicated reliance on R/S beliefs to cope with cancer. Patient spirituality and religious coping were associated with improved QOL in multivariable analyses (β = 10.57, P < .001 and β = 1.28, P = .01, respectively). Most patients considered attention to spiritual concerns an important part of cancer care by physicians (87%) and nurses (85%).
Limitations
Limitations include a small sample size, a cross-sectional study design, and a limited proportion of nonwhite participants (15%) from one US region.
Conclusion
Patients receiving palliative RT rely on R/S beliefs to cope with advanced cancer. Furthermore, spirituality and religious coping are contributors to better QOL. These findings highlight the importance of spiritual care in advanced cancer care.
*For a PDF of the full article click in the link to the left of this introduction.
The Role of Spirituality and Religious Coping in the Quality of Life of Patients With Advanced Cancer Receiving Palliative Radiation Therapy
Abstract
Objectives
National palliative care guidelines outline spiritual care as a domain of palliative care, yet patients' religiousness and/or spirituality (R/S) are underappreciated in the palliative oncology setting. Among patients with advanced cancer receiving palliative radiation therapy (RT), this study aims to characterize patient spirituality, religiousness, and religious coping; examine the relationships of these variables to quality of life (QOL); and assess patients' perceptions of spiritual care in the cancer care setting.
Methods
This is a multisite, cross-sectional survey of 69 patients with advanced cancer (response rate = 73%) receiving palliative RT. Scripted interviews assessed patient spirituality, religiousness, religious coping, QOL (McGill QOL Questionnaire), and perceptions of the importance of attention to spiritual needs by health providers. Multivariable models assessed the relationships of patient spirituality and R/S coping to patient QOL, controlling for other significant predictors of QOL.
Results
Most participants (84%) indicated reliance on R/S beliefs to cope with cancer. Patient spirituality and religious coping were associated with improved QOL in multivariable analyses (β = 10.57, P < .001 and β = 1.28, P = .01, respectively). Most patients considered attention to spiritual concerns an important part of cancer care by physicians (87%) and nurses (85%).
Limitations
Limitations include a small sample size, a cross-sectional study design, and a limited proportion of nonwhite participants (15%) from one US region.
Conclusion
Patients receiving palliative RT rely on R/S beliefs to cope with advanced cancer. Furthermore, spirituality and religious coping are contributors to better QOL. These findings highlight the importance of spiritual care in advanced cancer care.
*For a PDF of the full article click in the link to the left of this introduction.
The Role of Spirituality and Religious Coping in the Quality of Life of Patients With Advanced Cancer Receiving Palliative Radiation Therapy
Abstract
Objectives
National palliative care guidelines outline spiritual care as a domain of palliative care, yet patients' religiousness and/or spirituality (R/S) are underappreciated in the palliative oncology setting. Among patients with advanced cancer receiving palliative radiation therapy (RT), this study aims to characterize patient spirituality, religiousness, and religious coping; examine the relationships of these variables to quality of life (QOL); and assess patients' perceptions of spiritual care in the cancer care setting.
Methods
This is a multisite, cross-sectional survey of 69 patients with advanced cancer (response rate = 73%) receiving palliative RT. Scripted interviews assessed patient spirituality, religiousness, religious coping, QOL (McGill QOL Questionnaire), and perceptions of the importance of attention to spiritual needs by health providers. Multivariable models assessed the relationships of patient spirituality and R/S coping to patient QOL, controlling for other significant predictors of QOL.
Results
Most participants (84%) indicated reliance on R/S beliefs to cope with cancer. Patient spirituality and religious coping were associated with improved QOL in multivariable analyses (β = 10.57, P < .001 and β = 1.28, P = .01, respectively). Most patients considered attention to spiritual concerns an important part of cancer care by physicians (87%) and nurses (85%).
Limitations
Limitations include a small sample size, a cross-sectional study design, and a limited proportion of nonwhite participants (15%) from one US region.
Conclusion
Patients receiving palliative RT rely on R/S beliefs to cope with advanced cancer. Furthermore, spirituality and religious coping are contributors to better QOL. These findings highlight the importance of spiritual care in advanced cancer care.
*For a PDF of the full article click in the link to the left of this introduction.
Music Interventions Improve Anxiety, Pain in Cancer Patients
DENVER – Playing a musical instrument, singing, or simply listening to recorded music resulted in significant reduction in anxiety scores in patients with cancer, according to a Cochrane Database systematic review and meta-analysis.
The various music interventions offered by medical staff or trained music therapists also achieved significant improvements in the secondary end points of pain, mood, and quality of life scores, according to the review by investigators in the department of creative arts therapies at Drexel University in Philadelphia.
At the annual meeting of the American Academy of Hospice and Palliative Care Medicine, Dr. Wendy Anderson highlighted the Cochrane review findings as particularly on-point for her colleagues in oncology and palliative medicine.
"In palliative care we’re always trying to make the best of a difficult situation, and sometimes the best way to do this is through a nonpharmacologic intervention, even though it means doing something outside of our comfort zone," commented Dr. Anderson of the University of California, San Francisco.
Prior studies of music interventions in patients with cancer have been quite small. The Cochrane meta-analysis was designed to yield stronger, more definitive conclusions by incorporating those small studies which were sufficiently similar to combine.
The Cochrane report included 30 randomized clinical trials in seven countries with a total of 1,891 participating patients of all ages and with all types of cancer. In all, 13 trials involved the use of trained music therapists, while in the other 17 in the intervention consisted of listening to various genres of prerecorded music selected by the patients. Sessions were typically 30-45 minutes in length. The number of sessions varied widely from study to study. Subjects in the control arm received usual care or in some studies listened to white noise through headphones.
Sixteen randomized trials assessed anxiety. Collectively they showed that music interventions achieved statistically and clinically meaningful reductions in anxiety scores, with median to large effect sizes. For example, mean scores on the State Trait Anxiety Inventory fell by a mean of 11.2 units following the various music interventions while remaining unchanged in controls.
Similarly, the reduction in pain scores and improvements in structured measures of mood and quality of life in participants in the music interventions as compared to controls were also in the moderate to large range.
"Those are important outcomes in our patients with cancer," Dr. Anderson observed.
Music therapy is an established profession. Trained music therapists can perform a detailed patient evaluation and provide a tailored music experience. Although the Drexel researchers sought to learn whether the more elaborate, active participation music interventions provided by trained music therapists result in bigger improvements in psychologic and physical outcomes than the simpler music-listening interventions offered by medical staff, the investigators found that the research database wasn’t sufficiently large to draw any conclusions on that score (Cochrane Database Syst. Rev. 2011 [doi:10.1002/14651858.CD006911]).
The Cochrane group self-rated the quality of evidence on which the music intervention meta-analysis was based as low because these were nonblinded randomized trials. Patient awareness of whether they received a music intervention may have biased the results.
"Usually a low quality of evidence would, I think, make us reluctant to admit some innovations into our practices, but what’s interesting here is there’s a low potential for harm from music, and the patients feel the music improves their symptoms. That may be enough for us to implement music interventions in our practices," Dr. Anderson concluded.
The Cochrane review was funded by the State of Pennsylvania Formula Fund.
Dr. Anderson reported having no financial conflicts.
DENVER – Playing a musical instrument, singing, or simply listening to recorded music resulted in significant reduction in anxiety scores in patients with cancer, according to a Cochrane Database systematic review and meta-analysis.
The various music interventions offered by medical staff or trained music therapists also achieved significant improvements in the secondary end points of pain, mood, and quality of life scores, according to the review by investigators in the department of creative arts therapies at Drexel University in Philadelphia.
At the annual meeting of the American Academy of Hospice and Palliative Care Medicine, Dr. Wendy Anderson highlighted the Cochrane review findings as particularly on-point for her colleagues in oncology and palliative medicine.
"In palliative care we’re always trying to make the best of a difficult situation, and sometimes the best way to do this is through a nonpharmacologic intervention, even though it means doing something outside of our comfort zone," commented Dr. Anderson of the University of California, San Francisco.
Prior studies of music interventions in patients with cancer have been quite small. The Cochrane meta-analysis was designed to yield stronger, more definitive conclusions by incorporating those small studies which were sufficiently similar to combine.
The Cochrane report included 30 randomized clinical trials in seven countries with a total of 1,891 participating patients of all ages and with all types of cancer. In all, 13 trials involved the use of trained music therapists, while in the other 17 in the intervention consisted of listening to various genres of prerecorded music selected by the patients. Sessions were typically 30-45 minutes in length. The number of sessions varied widely from study to study. Subjects in the control arm received usual care or in some studies listened to white noise through headphones.
Sixteen randomized trials assessed anxiety. Collectively they showed that music interventions achieved statistically and clinically meaningful reductions in anxiety scores, with median to large effect sizes. For example, mean scores on the State Trait Anxiety Inventory fell by a mean of 11.2 units following the various music interventions while remaining unchanged in controls.
Similarly, the reduction in pain scores and improvements in structured measures of mood and quality of life in participants in the music interventions as compared to controls were also in the moderate to large range.
"Those are important outcomes in our patients with cancer," Dr. Anderson observed.
Music therapy is an established profession. Trained music therapists can perform a detailed patient evaluation and provide a tailored music experience. Although the Drexel researchers sought to learn whether the more elaborate, active participation music interventions provided by trained music therapists result in bigger improvements in psychologic and physical outcomes than the simpler music-listening interventions offered by medical staff, the investigators found that the research database wasn’t sufficiently large to draw any conclusions on that score (Cochrane Database Syst. Rev. 2011 [doi:10.1002/14651858.CD006911]).
The Cochrane group self-rated the quality of evidence on which the music intervention meta-analysis was based as low because these were nonblinded randomized trials. Patient awareness of whether they received a music intervention may have biased the results.
"Usually a low quality of evidence would, I think, make us reluctant to admit some innovations into our practices, but what’s interesting here is there’s a low potential for harm from music, and the patients feel the music improves their symptoms. That may be enough for us to implement music interventions in our practices," Dr. Anderson concluded.
The Cochrane review was funded by the State of Pennsylvania Formula Fund.
Dr. Anderson reported having no financial conflicts.
DENVER – Playing a musical instrument, singing, or simply listening to recorded music resulted in significant reduction in anxiety scores in patients with cancer, according to a Cochrane Database systematic review and meta-analysis.
The various music interventions offered by medical staff or trained music therapists also achieved significant improvements in the secondary end points of pain, mood, and quality of life scores, according to the review by investigators in the department of creative arts therapies at Drexel University in Philadelphia.
At the annual meeting of the American Academy of Hospice and Palliative Care Medicine, Dr. Wendy Anderson highlighted the Cochrane review findings as particularly on-point for her colleagues in oncology and palliative medicine.
"In palliative care we’re always trying to make the best of a difficult situation, and sometimes the best way to do this is through a nonpharmacologic intervention, even though it means doing something outside of our comfort zone," commented Dr. Anderson of the University of California, San Francisco.
Prior studies of music interventions in patients with cancer have been quite small. The Cochrane meta-analysis was designed to yield stronger, more definitive conclusions by incorporating those small studies which were sufficiently similar to combine.
The Cochrane report included 30 randomized clinical trials in seven countries with a total of 1,891 participating patients of all ages and with all types of cancer. In all, 13 trials involved the use of trained music therapists, while in the other 17 in the intervention consisted of listening to various genres of prerecorded music selected by the patients. Sessions were typically 30-45 minutes in length. The number of sessions varied widely from study to study. Subjects in the control arm received usual care or in some studies listened to white noise through headphones.
Sixteen randomized trials assessed anxiety. Collectively they showed that music interventions achieved statistically and clinically meaningful reductions in anxiety scores, with median to large effect sizes. For example, mean scores on the State Trait Anxiety Inventory fell by a mean of 11.2 units following the various music interventions while remaining unchanged in controls.
Similarly, the reduction in pain scores and improvements in structured measures of mood and quality of life in participants in the music interventions as compared to controls were also in the moderate to large range.
"Those are important outcomes in our patients with cancer," Dr. Anderson observed.
Music therapy is an established profession. Trained music therapists can perform a detailed patient evaluation and provide a tailored music experience. Although the Drexel researchers sought to learn whether the more elaborate, active participation music interventions provided by trained music therapists result in bigger improvements in psychologic and physical outcomes than the simpler music-listening interventions offered by medical staff, the investigators found that the research database wasn’t sufficiently large to draw any conclusions on that score (Cochrane Database Syst. Rev. 2011 [doi:10.1002/14651858.CD006911]).
The Cochrane group self-rated the quality of evidence on which the music intervention meta-analysis was based as low because these were nonblinded randomized trials. Patient awareness of whether they received a music intervention may have biased the results.
"Usually a low quality of evidence would, I think, make us reluctant to admit some innovations into our practices, but what’s interesting here is there’s a low potential for harm from music, and the patients feel the music improves their symptoms. That may be enough for us to implement music interventions in our practices," Dr. Anderson concluded.
The Cochrane review was funded by the State of Pennsylvania Formula Fund.
Dr. Anderson reported having no financial conflicts.
EXPERT ANALYSIS FROM THE ANNUAL ASSEMBLY OF THE AMERICAN ACADEMY OF HOSPICE AND PALLIATIVE MEDICINE
Antinausea Gel Not Absorbed at Therapeutic Levels
DENVER – Compounded ABH gel is widely prescribed for treatment of nausea in the hospice setting. It costs less than $1 per dose. Side effects are rare. And many clinicians swear by its effectiveness, noting that the constituent medications in ABH gel – Ativan (lorazepam), Benadryl (diphenhydramine), and Haldol (haloperidol) – target different mechanisms of nausea.
There is, however, a fly in the ointment ... er, gel.
It turns out that none of the three medications in topical ABH gel are absorbed systemically in anything remotely approaching therapeutic levels, Dr. Devon S. Fletcher reported at the annual meeting of the American Academy of Hospice and Palliative Care Medicine.
"Either ABH gel doesn’t work, or the effect is attributable to placebo, or possibly a nonpharmacologic mechanism is involved, such as stimulation of the P6 acupressure point on the wrist as patients rub the gel in," said Dr. Fletcher of Virginia Commonwealth University, Richmond. "If ABH doesn’t work, using it to treat a symptom leads to unacceptable suffering and unnecessary hospitalizations," she added.
Dr. Fletcher and coworkers measured levels of the three medications contained in ABH gel in six blood samples taken serially in each of 10 healthy volunteers up to 4 hours after they rubbed 1 mL of the gel between their volar wrists for 2 minutes. That’s the standard means of administering the medication, which is self-applied every 4-6 hours in clinical practice. A 1-mL dose contains 2 mg of lorazepam, 25 mg of diphenhydramine, and 2 mg of haloperidol compounded using a popular published formula.
Both lorazepam and haloperidol were undetectable in blood samples at all times in all 10 subjects; the medications never made it past the skin barrier. The highest concentration of diphenhydramine, reached in only a single patient, was 0.3 ng at 4 hours. That translates to 0.0003 mcg/mL, roughly 1/1000th of the therapeutic level, she noted.
The study was undertaken as a prelude to a planned single-center, randomized, placebo-controlled, crossover clinical trial of ABH gel in hospice patients with nausea. Dr. Fletcher and coinvestigators still plan to conduct that trial because ABH gel has never been subjected to a prospective controlled study.
"This is a wake-up call that we need further testing and evaluation before introducing treatments into widespread use," the physician declared.
Several audience members rose to applaud Dr. Fletcher for what they called a long-overdue rigorous look at an extremely popular yet unexamined therapy. One physician said he’d also like to see placebo-controlled studies of phenobarbital and dexamethasone for nausea, as these agents are widely prescribed but lacking in solid data supporting efficacy. He added that his own anecdotal experience with those two drugs as antiemetic agents in the hospice setting has been disappointing.
Dr. Fletcher reported having no financial conflicts.
DENVER – Compounded ABH gel is widely prescribed for treatment of nausea in the hospice setting. It costs less than $1 per dose. Side effects are rare. And many clinicians swear by its effectiveness, noting that the constituent medications in ABH gel – Ativan (lorazepam), Benadryl (diphenhydramine), and Haldol (haloperidol) – target different mechanisms of nausea.
There is, however, a fly in the ointment ... er, gel.
It turns out that none of the three medications in topical ABH gel are absorbed systemically in anything remotely approaching therapeutic levels, Dr. Devon S. Fletcher reported at the annual meeting of the American Academy of Hospice and Palliative Care Medicine.
"Either ABH gel doesn’t work, or the effect is attributable to placebo, or possibly a nonpharmacologic mechanism is involved, such as stimulation of the P6 acupressure point on the wrist as patients rub the gel in," said Dr. Fletcher of Virginia Commonwealth University, Richmond. "If ABH doesn’t work, using it to treat a symptom leads to unacceptable suffering and unnecessary hospitalizations," she added.
Dr. Fletcher and coworkers measured levels of the three medications contained in ABH gel in six blood samples taken serially in each of 10 healthy volunteers up to 4 hours after they rubbed 1 mL of the gel between their volar wrists for 2 minutes. That’s the standard means of administering the medication, which is self-applied every 4-6 hours in clinical practice. A 1-mL dose contains 2 mg of lorazepam, 25 mg of diphenhydramine, and 2 mg of haloperidol compounded using a popular published formula.
Both lorazepam and haloperidol were undetectable in blood samples at all times in all 10 subjects; the medications never made it past the skin barrier. The highest concentration of diphenhydramine, reached in only a single patient, was 0.3 ng at 4 hours. That translates to 0.0003 mcg/mL, roughly 1/1000th of the therapeutic level, she noted.
The study was undertaken as a prelude to a planned single-center, randomized, placebo-controlled, crossover clinical trial of ABH gel in hospice patients with nausea. Dr. Fletcher and coinvestigators still plan to conduct that trial because ABH gel has never been subjected to a prospective controlled study.
"This is a wake-up call that we need further testing and evaluation before introducing treatments into widespread use," the physician declared.
Several audience members rose to applaud Dr. Fletcher for what they called a long-overdue rigorous look at an extremely popular yet unexamined therapy. One physician said he’d also like to see placebo-controlled studies of phenobarbital and dexamethasone for nausea, as these agents are widely prescribed but lacking in solid data supporting efficacy. He added that his own anecdotal experience with those two drugs as antiemetic agents in the hospice setting has been disappointing.
Dr. Fletcher reported having no financial conflicts.
DENVER – Compounded ABH gel is widely prescribed for treatment of nausea in the hospice setting. It costs less than $1 per dose. Side effects are rare. And many clinicians swear by its effectiveness, noting that the constituent medications in ABH gel – Ativan (lorazepam), Benadryl (diphenhydramine), and Haldol (haloperidol) – target different mechanisms of nausea.
There is, however, a fly in the ointment ... er, gel.
It turns out that none of the three medications in topical ABH gel are absorbed systemically in anything remotely approaching therapeutic levels, Dr. Devon S. Fletcher reported at the annual meeting of the American Academy of Hospice and Palliative Care Medicine.
"Either ABH gel doesn’t work, or the effect is attributable to placebo, or possibly a nonpharmacologic mechanism is involved, such as stimulation of the P6 acupressure point on the wrist as patients rub the gel in," said Dr. Fletcher of Virginia Commonwealth University, Richmond. "If ABH doesn’t work, using it to treat a symptom leads to unacceptable suffering and unnecessary hospitalizations," she added.
Dr. Fletcher and coworkers measured levels of the three medications contained in ABH gel in six blood samples taken serially in each of 10 healthy volunteers up to 4 hours after they rubbed 1 mL of the gel between their volar wrists for 2 minutes. That’s the standard means of administering the medication, which is self-applied every 4-6 hours in clinical practice. A 1-mL dose contains 2 mg of lorazepam, 25 mg of diphenhydramine, and 2 mg of haloperidol compounded using a popular published formula.
Both lorazepam and haloperidol were undetectable in blood samples at all times in all 10 subjects; the medications never made it past the skin barrier. The highest concentration of diphenhydramine, reached in only a single patient, was 0.3 ng at 4 hours. That translates to 0.0003 mcg/mL, roughly 1/1000th of the therapeutic level, she noted.
The study was undertaken as a prelude to a planned single-center, randomized, placebo-controlled, crossover clinical trial of ABH gel in hospice patients with nausea. Dr. Fletcher and coinvestigators still plan to conduct that trial because ABH gel has never been subjected to a prospective controlled study.
"This is a wake-up call that we need further testing and evaluation before introducing treatments into widespread use," the physician declared.
Several audience members rose to applaud Dr. Fletcher for what they called a long-overdue rigorous look at an extremely popular yet unexamined therapy. One physician said he’d also like to see placebo-controlled studies of phenobarbital and dexamethasone for nausea, as these agents are widely prescribed but lacking in solid data supporting efficacy. He added that his own anecdotal experience with those two drugs as antiemetic agents in the hospice setting has been disappointing.
Dr. Fletcher reported having no financial conflicts.
EXPERT ANALYSIS FROM THE ANNUAL MEETING OF THE AMERICAN ACADEMY OF HOSPICE AND PALLIATIVE CARE MEDICINE
Late Oral Complications After Head/Neck Radiation 'Underreported'
PHOENIX – Late oral effects of head and neck cancer therapy are "multiple, underreported, and underappreciated."
That is the perspective of Joel Epstein, D.M.D., who has worked extensively with head and neck cancer patients experiencing severe dental and other oral problems following radiation therapy.
"The acute complications of head and neck cancer therapy are pretty well known, but the late complications are underappreciated," Dr. Epstein, director of oral medicine at City of Hope National Medical Center, Duarte, Calif., told attendees at the symposium.
As head and neck cancer treatments have advanced and patients are living longer, the spectrum of treatment complications has shifted, he explained. In a 5-year, prospective longitudinal study of 122 patients with oral carcinoma, dry mouth, sticky saliva, speech changes, dental problems, and sleep disturbance were reported by all patients except those treated only with surgery. These complications persisted at 1 and 5 years and affected quality of life (Head Neck 2008;30:461-70).
According to Dr. Epstein, the data illustrate the need for better collaboration between oncologists and dentists. "While people discuss the concept of multidisciplinary [and] interdisciplinary teams for the benefit of our patients, it is unfortunate that dentistry developed separately from physicians and surgeons. So while we need to interact, we’re not really well prepared to do so, particularly in the community," he said.
Clinically, it’s important to evaluate oral care, including brushing, flossing, fluoride, and tobacco abstinence, at all head and neck cancer treatment follow-up visits. Patients should be assessed for xerostomia, speech, swallowing, mucosal sensitivity, and taste. Head and neck and oral exams should include assessments for saliva (wet mucosa), exposed bone, infection, and new lesions or recurrent cancer, and a dental exam (for plaque, caries, and periodontal health), Dr. Epstein recommended.
Dry mouth, in particular, can lead to a host of other chronic problems related to swallowing, eating, sleeping, and dental health. When the 50-item Vanderbilt Head and Neck Symptom Survey was administered to a total of 70 patients, 67 reported having dry mouth at more than 6 months’ follow up (Head Neck 2011 Aug. 24 [doi:10.1002/hed.21816]).
The majority reported that dry mouth makes chewing/swallowing difficult (65) and that it affects their ability to sleep (67) and talk (64). With regard to eating and swallowing, similar majorities reported trouble eating solids (67) and drinking liquids (68), with food getting stuck in their mouth (66) and throat (67).
And, of concern, the same numbers of patients reported the sensation of choking or strangling on solids (66) and liquids (68). "The impact on function from the lack of saliva and the change in quality of saliva are issues we need to be more ready and willing to address," Dr. Epstein commented.
Taste and smell may also be profoundly altered. In the Vanderbilt survey, most patients reported altered taste (68), a decreased desire to eat (68), altered food choices (66), and a decrease in food eaten (66). A change in sense of smell was reported by 69 patients.
Such alterations often result in changes in diet, including decreased consumption of high-fiber food and of vitamins and other nutrients, along with increased consumption of fats, caffeine, and sugar. All of these factors increase the risk for dietary deficiencies, as well as dental caries.
Yet, altered taste sensation is not something patients might think to mention. "Half of patients experience altered taste sensation. But if they think you’re not interested or you don’t ask, you may not know," Dr. Epstein commented.
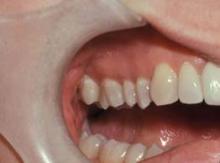
Periodontal health is often compromised by hyposalivation, which can lead to inflammation, bone/attachment loss, oral infection, and necrosis. Dental demineralization and cavitation may develop as early as 2-3 months after cancer treatment and progress rapidly, leading to fractures of the gum line, tooth loss, and necrosis.
Demineralization appears as a change to white, which may not be recognized as a problem because of the belief that white teeth are healthy. However, recognition at this stage is critical in order to prevent further dental damage, he said.
"The white change near the gum line and the tips of the teeth represent demineralization, and [in] time reversal can be accomplished prior to structural breakdown. Once cavitation has occurred, fillings are needed and prevention must be instituted or the cavities will recur and progress," Dr. Epstein said in an interview.
In the Vanderbilt survey, reported dental problems included difficulty chewing because of teeth/dentures (54 of the 70 patients); tooth sensitivity to hot, cold, or sweet foods (52); teeth feeling looser (51); teeth cracking/chipping (50); and trouble with dentures (24).
Oral candidiasis is another common problem, affecting approximately 39% of head and neck cancer patients during treatment and 33% afterward. One common clinical mistake is prescribing these patients antifungals that contain sugar, such as nystatin. "Nystatin is very high in sugar, and one of the [most commonly] used antifungals. The message is to avoid sugar-sweetened products in dry mouth patients and utilize alternatives," Dr. Epstein said in the interview.
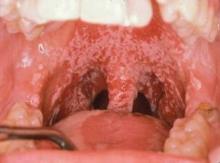
Mucosal sensitivity and pain is also frequent. In a meta-analysis of 22 studies published between 1990 and 2008, the prevalence of trismus was 25.4% in patients who received conventional radiotherapy and 5% for the few intensity-modulated radiation therapy studies that were included, suggesting that the newer radiation modality might diminish the problem (Support. Care Cancer 2010;18:1033-8).
Data suggest that the radiation effect on mandibular movement correlates with the radiation dose to the mastication muscles, with a steep dose-response curve. Onset is typically 2-6 months post treatment and is ongoing. Concurrent chemotherapy may increase the incidence and/or severity of mandibular immobility (Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 1999;88:365-73).
In the Vanderbilt survey, most patients reported burning in the throat or mouth (69); sensitivity to hot, spicy, or acid food (67); sensitivity to dryness (69); and changes in food intake because of mucosal sensitivity (67); most patients also reported that mucosal sensitivity prevents tooth brushing (63). "Mucosal sensitivity is a quality of life issue," Dr. Epstein said.
The Vanderbilt survey was particularly illuminating, Dr. Epstein commented. Studies that utilize claims data probably underrepresent the problem of long-term oral complications because dental and medical insurance are separate and the data are not easily combined, he added.
"Late oral effects are best diagnosed [and] managed in a multidisciplinary team with close communication between medical and dental providers. ... We really need to come together."
The Multinational Association of Supportive Care in Cancer is developing tools to improve communication between dentistry and medicine. These could be available for beta testing as early as this fall.
Dr. Epstein said he had no relevant financial disclosures.
PHOENIX – Late oral effects of head and neck cancer therapy are "multiple, underreported, and underappreciated."
That is the perspective of Joel Epstein, D.M.D., who has worked extensively with head and neck cancer patients experiencing severe dental and other oral problems following radiation therapy.
"The acute complications of head and neck cancer therapy are pretty well known, but the late complications are underappreciated," Dr. Epstein, director of oral medicine at City of Hope National Medical Center, Duarte, Calif., told attendees at the symposium.
As head and neck cancer treatments have advanced and patients are living longer, the spectrum of treatment complications has shifted, he explained. In a 5-year, prospective longitudinal study of 122 patients with oral carcinoma, dry mouth, sticky saliva, speech changes, dental problems, and sleep disturbance were reported by all patients except those treated only with surgery. These complications persisted at 1 and 5 years and affected quality of life (Head Neck 2008;30:461-70).
According to Dr. Epstein, the data illustrate the need for better collaboration between oncologists and dentists. "While people discuss the concept of multidisciplinary [and] interdisciplinary teams for the benefit of our patients, it is unfortunate that dentistry developed separately from physicians and surgeons. So while we need to interact, we’re not really well prepared to do so, particularly in the community," he said.
Clinically, it’s important to evaluate oral care, including brushing, flossing, fluoride, and tobacco abstinence, at all head and neck cancer treatment follow-up visits. Patients should be assessed for xerostomia, speech, swallowing, mucosal sensitivity, and taste. Head and neck and oral exams should include assessments for saliva (wet mucosa), exposed bone, infection, and new lesions or recurrent cancer, and a dental exam (for plaque, caries, and periodontal health), Dr. Epstein recommended.
Dry mouth, in particular, can lead to a host of other chronic problems related to swallowing, eating, sleeping, and dental health. When the 50-item Vanderbilt Head and Neck Symptom Survey was administered to a total of 70 patients, 67 reported having dry mouth at more than 6 months’ follow up (Head Neck 2011 Aug. 24 [doi:10.1002/hed.21816]).
The majority reported that dry mouth makes chewing/swallowing difficult (65) and that it affects their ability to sleep (67) and talk (64). With regard to eating and swallowing, similar majorities reported trouble eating solids (67) and drinking liquids (68), with food getting stuck in their mouth (66) and throat (67).
And, of concern, the same numbers of patients reported the sensation of choking or strangling on solids (66) and liquids (68). "The impact on function from the lack of saliva and the change in quality of saliva are issues we need to be more ready and willing to address," Dr. Epstein commented.
Taste and smell may also be profoundly altered. In the Vanderbilt survey, most patients reported altered taste (68), a decreased desire to eat (68), altered food choices (66), and a decrease in food eaten (66). A change in sense of smell was reported by 69 patients.
Such alterations often result in changes in diet, including decreased consumption of high-fiber food and of vitamins and other nutrients, along with increased consumption of fats, caffeine, and sugar. All of these factors increase the risk for dietary deficiencies, as well as dental caries.
Yet, altered taste sensation is not something patients might think to mention. "Half of patients experience altered taste sensation. But if they think you’re not interested or you don’t ask, you may not know," Dr. Epstein commented.
Periodontal health is often compromised by hyposalivation, which can lead to inflammation, bone/attachment loss, oral infection, and necrosis. Dental demineralization and cavitation may develop as early as 2-3 months after cancer treatment and progress rapidly, leading to fractures of the gum line, tooth loss, and necrosis.
Demineralization appears as a change to white, which may not be recognized as a problem because of the belief that white teeth are healthy. However, recognition at this stage is critical in order to prevent further dental damage, he said.
"The white change near the gum line and the tips of the teeth represent demineralization, and [in] time reversal can be accomplished prior to structural breakdown. Once cavitation has occurred, fillings are needed and prevention must be instituted or the cavities will recur and progress," Dr. Epstein said in an interview.
In the Vanderbilt survey, reported dental problems included difficulty chewing because of teeth/dentures (54 of the 70 patients); tooth sensitivity to hot, cold, or sweet foods (52); teeth feeling looser (51); teeth cracking/chipping (50); and trouble with dentures (24).
Oral candidiasis is another common problem, affecting approximately 39% of head and neck cancer patients during treatment and 33% afterward. One common clinical mistake is prescribing these patients antifungals that contain sugar, such as nystatin. "Nystatin is very high in sugar, and one of the [most commonly] used antifungals. The message is to avoid sugar-sweetened products in dry mouth patients and utilize alternatives," Dr. Epstein said in the interview.
Mucosal sensitivity and pain is also frequent. In a meta-analysis of 22 studies published between 1990 and 2008, the prevalence of trismus was 25.4% in patients who received conventional radiotherapy and 5% for the few intensity-modulated radiation therapy studies that were included, suggesting that the newer radiation modality might diminish the problem (Support. Care Cancer 2010;18:1033-8).
Data suggest that the radiation effect on mandibular movement correlates with the radiation dose to the mastication muscles, with a steep dose-response curve. Onset is typically 2-6 months post treatment and is ongoing. Concurrent chemotherapy may increase the incidence and/or severity of mandibular immobility (Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 1999;88:365-73).
In the Vanderbilt survey, most patients reported burning in the throat or mouth (69); sensitivity to hot, spicy, or acid food (67); sensitivity to dryness (69); and changes in food intake because of mucosal sensitivity (67); most patients also reported that mucosal sensitivity prevents tooth brushing (63). "Mucosal sensitivity is a quality of life issue," Dr. Epstein said.
The Vanderbilt survey was particularly illuminating, Dr. Epstein commented. Studies that utilize claims data probably underrepresent the problem of long-term oral complications because dental and medical insurance are separate and the data are not easily combined, he added.
"Late oral effects are best diagnosed [and] managed in a multidisciplinary team with close communication between medical and dental providers. ... We really need to come together."
The Multinational Association of Supportive Care in Cancer is developing tools to improve communication between dentistry and medicine. These could be available for beta testing as early as this fall.
Dr. Epstein said he had no relevant financial disclosures.
PHOENIX – Late oral effects of head and neck cancer therapy are "multiple, underreported, and underappreciated."
That is the perspective of Joel Epstein, D.M.D., who has worked extensively with head and neck cancer patients experiencing severe dental and other oral problems following radiation therapy.
"The acute complications of head and neck cancer therapy are pretty well known, but the late complications are underappreciated," Dr. Epstein, director of oral medicine at City of Hope National Medical Center, Duarte, Calif., told attendees at the symposium.
As head and neck cancer treatments have advanced and patients are living longer, the spectrum of treatment complications has shifted, he explained. In a 5-year, prospective longitudinal study of 122 patients with oral carcinoma, dry mouth, sticky saliva, speech changes, dental problems, and sleep disturbance were reported by all patients except those treated only with surgery. These complications persisted at 1 and 5 years and affected quality of life (Head Neck 2008;30:461-70).
According to Dr. Epstein, the data illustrate the need for better collaboration between oncologists and dentists. "While people discuss the concept of multidisciplinary [and] interdisciplinary teams for the benefit of our patients, it is unfortunate that dentistry developed separately from physicians and surgeons. So while we need to interact, we’re not really well prepared to do so, particularly in the community," he said.
Clinically, it’s important to evaluate oral care, including brushing, flossing, fluoride, and tobacco abstinence, at all head and neck cancer treatment follow-up visits. Patients should be assessed for xerostomia, speech, swallowing, mucosal sensitivity, and taste. Head and neck and oral exams should include assessments for saliva (wet mucosa), exposed bone, infection, and new lesions or recurrent cancer, and a dental exam (for plaque, caries, and periodontal health), Dr. Epstein recommended.
Dry mouth, in particular, can lead to a host of other chronic problems related to swallowing, eating, sleeping, and dental health. When the 50-item Vanderbilt Head and Neck Symptom Survey was administered to a total of 70 patients, 67 reported having dry mouth at more than 6 months’ follow up (Head Neck 2011 Aug. 24 [doi:10.1002/hed.21816]).
The majority reported that dry mouth makes chewing/swallowing difficult (65) and that it affects their ability to sleep (67) and talk (64). With regard to eating and swallowing, similar majorities reported trouble eating solids (67) and drinking liquids (68), with food getting stuck in their mouth (66) and throat (67).
And, of concern, the same numbers of patients reported the sensation of choking or strangling on solids (66) and liquids (68). "The impact on function from the lack of saliva and the change in quality of saliva are issues we need to be more ready and willing to address," Dr. Epstein commented.
Taste and smell may also be profoundly altered. In the Vanderbilt survey, most patients reported altered taste (68), a decreased desire to eat (68), altered food choices (66), and a decrease in food eaten (66). A change in sense of smell was reported by 69 patients.
Such alterations often result in changes in diet, including decreased consumption of high-fiber food and of vitamins and other nutrients, along with increased consumption of fats, caffeine, and sugar. All of these factors increase the risk for dietary deficiencies, as well as dental caries.
Yet, altered taste sensation is not something patients might think to mention. "Half of patients experience altered taste sensation. But if they think you’re not interested or you don’t ask, you may not know," Dr. Epstein commented.
Periodontal health is often compromised by hyposalivation, which can lead to inflammation, bone/attachment loss, oral infection, and necrosis. Dental demineralization and cavitation may develop as early as 2-3 months after cancer treatment and progress rapidly, leading to fractures of the gum line, tooth loss, and necrosis.
Demineralization appears as a change to white, which may not be recognized as a problem because of the belief that white teeth are healthy. However, recognition at this stage is critical in order to prevent further dental damage, he said.
"The white change near the gum line and the tips of the teeth represent demineralization, and [in] time reversal can be accomplished prior to structural breakdown. Once cavitation has occurred, fillings are needed and prevention must be instituted or the cavities will recur and progress," Dr. Epstein said in an interview.
In the Vanderbilt survey, reported dental problems included difficulty chewing because of teeth/dentures (54 of the 70 patients); tooth sensitivity to hot, cold, or sweet foods (52); teeth feeling looser (51); teeth cracking/chipping (50); and trouble with dentures (24).
Oral candidiasis is another common problem, affecting approximately 39% of head and neck cancer patients during treatment and 33% afterward. One common clinical mistake is prescribing these patients antifungals that contain sugar, such as nystatin. "Nystatin is very high in sugar, and one of the [most commonly] used antifungals. The message is to avoid sugar-sweetened products in dry mouth patients and utilize alternatives," Dr. Epstein said in the interview.
Mucosal sensitivity and pain is also frequent. In a meta-analysis of 22 studies published between 1990 and 2008, the prevalence of trismus was 25.4% in patients who received conventional radiotherapy and 5% for the few intensity-modulated radiation therapy studies that were included, suggesting that the newer radiation modality might diminish the problem (Support. Care Cancer 2010;18:1033-8).
Data suggest that the radiation effect on mandibular movement correlates with the radiation dose to the mastication muscles, with a steep dose-response curve. Onset is typically 2-6 months post treatment and is ongoing. Concurrent chemotherapy may increase the incidence and/or severity of mandibular immobility (Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 1999;88:365-73).
In the Vanderbilt survey, most patients reported burning in the throat or mouth (69); sensitivity to hot, spicy, or acid food (67); sensitivity to dryness (69); and changes in food intake because of mucosal sensitivity (67); most patients also reported that mucosal sensitivity prevents tooth brushing (63). "Mucosal sensitivity is a quality of life issue," Dr. Epstein said.
The Vanderbilt survey was particularly illuminating, Dr. Epstein commented. Studies that utilize claims data probably underrepresent the problem of long-term oral complications because dental and medical insurance are separate and the data are not easily combined, he added.
"Late oral effects are best diagnosed [and] managed in a multidisciplinary team with close communication between medical and dental providers. ... We really need to come together."
The Multinational Association of Supportive Care in Cancer is developing tools to improve communication between dentistry and medicine. These could be available for beta testing as early as this fall.
Dr. Epstein said he had no relevant financial disclosures.
EXPERT OPINION FROM A HEAD AND NECK CANCER SYMPOSIUM SPONSORED BY THE AMERICAN SOCIETY FOR RADIATION ONCOLOGY
Study: Myeloma Survival Gains, but QOL Slides
MIAMI – Patients who are diagnosed with multiple myeloma can expect to live longer than in the past, but their symptom burden remains considerable for at least 10 years after diagnosis, a prospective, population-based, Dutch study has concluded.
"The symptom burden is not only caused by the disease itself but also by the new aggressive treatments," said Floortje Mols, Ph.D., of Tilburg (the Netherlands) University at the annual conference of the American Psychosocial Oncology Society.
Dr. Mols and her coauthors identified multiple myeloma patients in the Eindhoven (the Netherlands) Cancer Registry who had been diagnosed during 1999-2010 in that country. They surveyed the survivors twice – at baseline and a year later – along with an age- and sex-matched population-based cohort.
At baseline, 156 survivors reported significantly lower scores than did the control group on every subscale of the EORTC QLQ-C30 (European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire), a validated instrument that has been used in more than 3,000 published studies. No significant differences were seen in symptom and quality of life scores between short-term (defined as up to 5 years) and long-term (more than 5 years) survivors, she reported.
Physical functioning, fatigue, and dyspnea scores diverged from those in the controls most strikingly, but the difference was statistically significant for every subscale with P scores of less than 0.01 and beyond, said Dr. Mols, a medical psychologist at the university’s center of research on psychology in somatic diseases and the Comprehensive Cancer Centre South, Eindhoven.
In addition, multiple myeloma survivors reported a high rate of disease-specific symptoms, including tingling and numbness, pain, and drowsiness – even years beyond diagnosis. Peripheral neuropathy is a common side effect of therapy for multiple myeloma.
At baseline, 37% of short- and long-term survivors reported worrying about their health during the previous week, 34% reported worrying about their disease, and 21% reported worrying "very much or quite a bit" about dying.
In the second round, at a 1-year follow-up, 80 survivors (including some diagnosed more recently) responded to the same questionnaire. Over that time, quality of life had diminished further for 74% of respondents (mean score, 55 vs. 68; P less than .001).
Half of the patients reported more fatigue, 71% more nausea and vomiting, 59% more pain, and 66% more dyspnea than at baseline. The most bothersome symptoms cited in the follow-up survey included tingling of the hands and feet (32%), back pain (28%), bone aches and pain (26%), pain in the arms and shoulders (19%), and drowsiness (18%).
"Maximizing disease control while minimizing these symptoms with supportive care for the entire survivorship trajectory is, I think, one of the major challenges of multiple myeloma treatment," said Dr. Mols.
She recommended referral of survivors to specific cancer survivorship care programs for management of symptoms that extend beyond the active phase of their treatment.
Improvement – and Side Effects
Dr. William I. Bensinger, who was not affiliated with the study, said following the meeting that the results were "not surprising."
"We have made great strides in the management of multiple myeloma, and patients are clearly living longer. Some of these improved treatments, however, come with the risk of significant side effects," said Dr. Bensinger, professor of medicine at the University of Washington and Fred Hutchinson Cancer Research Center in Seattle.
Dr. Bensinger called the results "a wake-up call to my fellow oncologists that we need to do a better job of proactively managing symptoms of treatment in order to improve our patients’ quality of life."
Ebb and Flow in QOL
Another multiple myeloma specialist, Dr. Jayesh Mehta, a professor of medicine at Northwestern University in Chicago, said that quality of life may ebb and flow over the course of many years in survivors.
"What we see in a typical patient is somewhat impaired quality of life at baseline that may worsen or remain stable in the short term, but improves by a year after diagnosis, remains good for a few years – say, 3 to 5 – worsens somewhat when there is relapse due to disease and therapy, stabilizes as the disease responds, and then worsens again when the disease returns."
Funding for the study was provided by the Netherlands Organization for Scientific Research, the Dutch Cancer Society, the Jonker-Driessen Foundation, ZonMW (the Netherlands organization for health research and development), and PHAROS (Population-Based Haematological Registry for Observational Studies).
MIAMI – Patients who are diagnosed with multiple myeloma can expect to live longer than in the past, but their symptom burden remains considerable for at least 10 years after diagnosis, a prospective, population-based, Dutch study has concluded.
"The symptom burden is not only caused by the disease itself but also by the new aggressive treatments," said Floortje Mols, Ph.D., of Tilburg (the Netherlands) University at the annual conference of the American Psychosocial Oncology Society.
Dr. Mols and her coauthors identified multiple myeloma patients in the Eindhoven (the Netherlands) Cancer Registry who had been diagnosed during 1999-2010 in that country. They surveyed the survivors twice – at baseline and a year later – along with an age- and sex-matched population-based cohort.
At baseline, 156 survivors reported significantly lower scores than did the control group on every subscale of the EORTC QLQ-C30 (European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire), a validated instrument that has been used in more than 3,000 published studies. No significant differences were seen in symptom and quality of life scores between short-term (defined as up to 5 years) and long-term (more than 5 years) survivors, she reported.
Physical functioning, fatigue, and dyspnea scores diverged from those in the controls most strikingly, but the difference was statistically significant for every subscale with P scores of less than 0.01 and beyond, said Dr. Mols, a medical psychologist at the university’s center of research on psychology in somatic diseases and the Comprehensive Cancer Centre South, Eindhoven.
In addition, multiple myeloma survivors reported a high rate of disease-specific symptoms, including tingling and numbness, pain, and drowsiness – even years beyond diagnosis. Peripheral neuropathy is a common side effect of therapy for multiple myeloma.
At baseline, 37% of short- and long-term survivors reported worrying about their health during the previous week, 34% reported worrying about their disease, and 21% reported worrying "very much or quite a bit" about dying.
In the second round, at a 1-year follow-up, 80 survivors (including some diagnosed more recently) responded to the same questionnaire. Over that time, quality of life had diminished further for 74% of respondents (mean score, 55 vs. 68; P less than .001).
Half of the patients reported more fatigue, 71% more nausea and vomiting, 59% more pain, and 66% more dyspnea than at baseline. The most bothersome symptoms cited in the follow-up survey included tingling of the hands and feet (32%), back pain (28%), bone aches and pain (26%), pain in the arms and shoulders (19%), and drowsiness (18%).
"Maximizing disease control while minimizing these symptoms with supportive care for the entire survivorship trajectory is, I think, one of the major challenges of multiple myeloma treatment," said Dr. Mols.
She recommended referral of survivors to specific cancer survivorship care programs for management of symptoms that extend beyond the active phase of their treatment.
Improvement – and Side Effects
Dr. William I. Bensinger, who was not affiliated with the study, said following the meeting that the results were "not surprising."
"We have made great strides in the management of multiple myeloma, and patients are clearly living longer. Some of these improved treatments, however, come with the risk of significant side effects," said Dr. Bensinger, professor of medicine at the University of Washington and Fred Hutchinson Cancer Research Center in Seattle.
Dr. Bensinger called the results "a wake-up call to my fellow oncologists that we need to do a better job of proactively managing symptoms of treatment in order to improve our patients’ quality of life."
Ebb and Flow in QOL
Another multiple myeloma specialist, Dr. Jayesh Mehta, a professor of medicine at Northwestern University in Chicago, said that quality of life may ebb and flow over the course of many years in survivors.
"What we see in a typical patient is somewhat impaired quality of life at baseline that may worsen or remain stable in the short term, but improves by a year after diagnosis, remains good for a few years – say, 3 to 5 – worsens somewhat when there is relapse due to disease and therapy, stabilizes as the disease responds, and then worsens again when the disease returns."
Funding for the study was provided by the Netherlands Organization for Scientific Research, the Dutch Cancer Society, the Jonker-Driessen Foundation, ZonMW (the Netherlands organization for health research and development), and PHAROS (Population-Based Haematological Registry for Observational Studies).
MIAMI – Patients who are diagnosed with multiple myeloma can expect to live longer than in the past, but their symptom burden remains considerable for at least 10 years after diagnosis, a prospective, population-based, Dutch study has concluded.
"The symptom burden is not only caused by the disease itself but also by the new aggressive treatments," said Floortje Mols, Ph.D., of Tilburg (the Netherlands) University at the annual conference of the American Psychosocial Oncology Society.
Dr. Mols and her coauthors identified multiple myeloma patients in the Eindhoven (the Netherlands) Cancer Registry who had been diagnosed during 1999-2010 in that country. They surveyed the survivors twice – at baseline and a year later – along with an age- and sex-matched population-based cohort.
At baseline, 156 survivors reported significantly lower scores than did the control group on every subscale of the EORTC QLQ-C30 (European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire), a validated instrument that has been used in more than 3,000 published studies. No significant differences were seen in symptom and quality of life scores between short-term (defined as up to 5 years) and long-term (more than 5 years) survivors, she reported.
Physical functioning, fatigue, and dyspnea scores diverged from those in the controls most strikingly, but the difference was statistically significant for every subscale with P scores of less than 0.01 and beyond, said Dr. Mols, a medical psychologist at the university’s center of research on psychology in somatic diseases and the Comprehensive Cancer Centre South, Eindhoven.
In addition, multiple myeloma survivors reported a high rate of disease-specific symptoms, including tingling and numbness, pain, and drowsiness – even years beyond diagnosis. Peripheral neuropathy is a common side effect of therapy for multiple myeloma.
At baseline, 37% of short- and long-term survivors reported worrying about their health during the previous week, 34% reported worrying about their disease, and 21% reported worrying "very much or quite a bit" about dying.
In the second round, at a 1-year follow-up, 80 survivors (including some diagnosed more recently) responded to the same questionnaire. Over that time, quality of life had diminished further for 74% of respondents (mean score, 55 vs. 68; P less than .001).
Half of the patients reported more fatigue, 71% more nausea and vomiting, 59% more pain, and 66% more dyspnea than at baseline. The most bothersome symptoms cited in the follow-up survey included tingling of the hands and feet (32%), back pain (28%), bone aches and pain (26%), pain in the arms and shoulders (19%), and drowsiness (18%).
"Maximizing disease control while minimizing these symptoms with supportive care for the entire survivorship trajectory is, I think, one of the major challenges of multiple myeloma treatment," said Dr. Mols.
She recommended referral of survivors to specific cancer survivorship care programs for management of symptoms that extend beyond the active phase of their treatment.
Improvement – and Side Effects
Dr. William I. Bensinger, who was not affiliated with the study, said following the meeting that the results were "not surprising."
"We have made great strides in the management of multiple myeloma, and patients are clearly living longer. Some of these improved treatments, however, come with the risk of significant side effects," said Dr. Bensinger, professor of medicine at the University of Washington and Fred Hutchinson Cancer Research Center in Seattle.
Dr. Bensinger called the results "a wake-up call to my fellow oncologists that we need to do a better job of proactively managing symptoms of treatment in order to improve our patients’ quality of life."
Ebb and Flow in QOL
Another multiple myeloma specialist, Dr. Jayesh Mehta, a professor of medicine at Northwestern University in Chicago, said that quality of life may ebb and flow over the course of many years in survivors.
"What we see in a typical patient is somewhat impaired quality of life at baseline that may worsen or remain stable in the short term, but improves by a year after diagnosis, remains good for a few years – say, 3 to 5 – worsens somewhat when there is relapse due to disease and therapy, stabilizes as the disease responds, and then worsens again when the disease returns."
Funding for the study was provided by the Netherlands Organization for Scientific Research, the Dutch Cancer Society, the Jonker-Driessen Foundation, ZonMW (the Netherlands organization for health research and development), and PHAROS (Population-Based Haematological Registry for Observational Studies).
FROM THE ANNUAL CONFERENCE OF THE AMERICAN PSYCHOSOCIAL ONCOLOGY SOCIETY
Major Finding: In 1 year, quality of life diminished for 74% of respondents (mean score, 55 vs. 68; P less than .001).
Data Source: The data were self-reported on the EORTC QLQ-C30 questionnaire by multiple myeloma survivors in the Eindhoven (the Netherlands) Cancer Registry.
Disclosures: Funding for the study was provided by the Netherlands Organization for Scientific Research, the Dutch Cancer Society, the Jonker-Driessen Foundation, ZonMW (the Netherlands organization for health research and development), and the PHAROS (Population-Based Haematological Registry for Observational Studies).
Identify Depressive Symptoms Early in Breast Cancer Patients
More than one in four women who were later diagnosed with breast cancer had a combined state anxiety and depressive symptoms, and this helped predict quality of life, state anxiety, depressive symptoms, and fatigue at 12 and 24 months after surgery, according to a multicenter prospective study reported in the February 2012 issue of the Journal of Affective Disorders.
Incidence of breast cancer in Europe is 88/100,000 individuals, with survival rates of 24.3/100,000. A recent study estimated that 16.3% of cancer patients have clinical depression and 20.7% have all types of depression, respectively. However, few studies address the relationship between baseline anxiety and/or depression and quality of life.
So, Lotje van Esch, a doctoral candidate at the Center of Research on Psychology and Somatic Diseases at Tilburg University in the Netherlands, and her coauthors began to investigate state anxiety (namely, the level of momentary anxiety), depressive symptoms, and combined state anxiety and depressive symptoms (CADS) the relationship to quality of life, fatigue, and mood at 12 and 24 months after surgery in women with breast cancer (J. Affect. Disord. 2012;136:895-901).
They recruited 1,501 women referred by the national screening program or their general practitioners to six hospitals. These women, who had not yet received a diagnosis, completed a set of questionnaires that measured state anxiety, the presence and degree of depressive symptoms over the previous week, fatigue, quality of life, personality trait neuroticism, and trait anxiety.
Among these subjects, 407 women (27% of all subjects) were diagnosed with breast cancer, and 111 (28% of those diagnosed with cancer) had CADS at baseline. These patients completed questionnaires again at 12 and 24 months after surgery, although 75 dropped out between baseline and 12 months, and 24 dropped out between 12 and 14 months. The researchers also gathered demographic data and medical information on disease stage at diagnosis, type of operation and adjuvant treatment, such as chemotherapy, radiotherapy, or hormone treatment.
Of the remaining 332 patients who were in the study at 12 months, 14% had CADS, the results show. And, 10% of the remaining 246 patients had CADS at 24 months, and 10% had CADS at 12 and 24 months, respectively.
And, 10% of the remaining 246 patients at 24 months, 10% had CADS at 12 and 24 months, respectively.
At 12 and 24 months, 21% of the group had elevated depressive symptoms, and 20% had state anxiety. Of the group that had CADS at baseline, a greater percentage had elevated levels of anxiety, depressive symptoms, and CADS at 12 and 24 months than did the group that did not have CADS at baseline.
"[The] CADS group had elevated levels of state anxiety, depressive symptoms, and CADS at all follow-up measure moments in the 2 years after baseline, compared with the non-CADS group," the researchers said. "In both groups, these scores decreased over time but, after 24 months, the CADS, state anxiety, and depressive symptoms scores of the CADS group were still about three times as high as the scores in the non-CADS group. This implies that the CADS group experienced considerably more anxiety and/or depressive symptoms even 24 months after surgery."
On further analysis, the researchers found that CADS and neuroticism were both significant predictors of patients’ quality of life, fatigue, depressive symptoms, and state anxiety at 12 and 24 months after surgery, the researchers found.
The findings in this study suggest that clinicians use questionnaires or screening instruments to identify those women with a higher score on state anxiety and depressive symptom, the researchers say, and that they should do so as soon as possible when diagnosing and treating breast cancer. "Only by identifying this group of patients, tailored psychological care can be accomplished," they add.
The study’s strength was its prospective multicenter and longitudinal design. A limitation, however, was that some data were not available because some patients dropped out, and because some women were included less than 2 years earlier. Also, those who dropped out often cited a high stress level, meaning that anxiety levels might have been higher in the patients who did not participate.
The authors had no conflicts of interest to declare, and there were no study sponsors.
More than one in four women who were later diagnosed with breast cancer had a combined state anxiety and depressive symptoms, and this helped predict quality of life, state anxiety, depressive symptoms, and fatigue at 12 and 24 months after surgery, according to a multicenter prospective study reported in the February 2012 issue of the Journal of Affective Disorders.
Incidence of breast cancer in Europe is 88/100,000 individuals, with survival rates of 24.3/100,000. A recent study estimated that 16.3% of cancer patients have clinical depression and 20.7% have all types of depression, respectively. However, few studies address the relationship between baseline anxiety and/or depression and quality of life.
So, Lotje van Esch, a doctoral candidate at the Center of Research on Psychology and Somatic Diseases at Tilburg University in the Netherlands, and her coauthors began to investigate state anxiety (namely, the level of momentary anxiety), depressive symptoms, and combined state anxiety and depressive symptoms (CADS) the relationship to quality of life, fatigue, and mood at 12 and 24 months after surgery in women with breast cancer (J. Affect. Disord. 2012;136:895-901).
They recruited 1,501 women referred by the national screening program or their general practitioners to six hospitals. These women, who had not yet received a diagnosis, completed a set of questionnaires that measured state anxiety, the presence and degree of depressive symptoms over the previous week, fatigue, quality of life, personality trait neuroticism, and trait anxiety.
Among these subjects, 407 women (27% of all subjects) were diagnosed with breast cancer, and 111 (28% of those diagnosed with cancer) had CADS at baseline. These patients completed questionnaires again at 12 and 24 months after surgery, although 75 dropped out between baseline and 12 months, and 24 dropped out between 12 and 14 months. The researchers also gathered demographic data and medical information on disease stage at diagnosis, type of operation and adjuvant treatment, such as chemotherapy, radiotherapy, or hormone treatment.
Of the remaining 332 patients who were in the study at 12 months, 14% had CADS, the results show. And, 10% of the remaining 246 patients had CADS at 24 months, and 10% had CADS at 12 and 24 months, respectively.
And, 10% of the remaining 246 patients at 24 months, 10% had CADS at 12 and 24 months, respectively.
At 12 and 24 months, 21% of the group had elevated depressive symptoms, and 20% had state anxiety. Of the group that had CADS at baseline, a greater percentage had elevated levels of anxiety, depressive symptoms, and CADS at 12 and 24 months than did the group that did not have CADS at baseline.
"[The] CADS group had elevated levels of state anxiety, depressive symptoms, and CADS at all follow-up measure moments in the 2 years after baseline, compared with the non-CADS group," the researchers said. "In both groups, these scores decreased over time but, after 24 months, the CADS, state anxiety, and depressive symptoms scores of the CADS group were still about three times as high as the scores in the non-CADS group. This implies that the CADS group experienced considerably more anxiety and/or depressive symptoms even 24 months after surgery."
On further analysis, the researchers found that CADS and neuroticism were both significant predictors of patients’ quality of life, fatigue, depressive symptoms, and state anxiety at 12 and 24 months after surgery, the researchers found.
The findings in this study suggest that clinicians use questionnaires or screening instruments to identify those women with a higher score on state anxiety and depressive symptom, the researchers say, and that they should do so as soon as possible when diagnosing and treating breast cancer. "Only by identifying this group of patients, tailored psychological care can be accomplished," they add.
The study’s strength was its prospective multicenter and longitudinal design. A limitation, however, was that some data were not available because some patients dropped out, and because some women were included less than 2 years earlier. Also, those who dropped out often cited a high stress level, meaning that anxiety levels might have been higher in the patients who did not participate.
The authors had no conflicts of interest to declare, and there were no study sponsors.
More than one in four women who were later diagnosed with breast cancer had a combined state anxiety and depressive symptoms, and this helped predict quality of life, state anxiety, depressive symptoms, and fatigue at 12 and 24 months after surgery, according to a multicenter prospective study reported in the February 2012 issue of the Journal of Affective Disorders.
Incidence of breast cancer in Europe is 88/100,000 individuals, with survival rates of 24.3/100,000. A recent study estimated that 16.3% of cancer patients have clinical depression and 20.7% have all types of depression, respectively. However, few studies address the relationship between baseline anxiety and/or depression and quality of life.
So, Lotje van Esch, a doctoral candidate at the Center of Research on Psychology and Somatic Diseases at Tilburg University in the Netherlands, and her coauthors began to investigate state anxiety (namely, the level of momentary anxiety), depressive symptoms, and combined state anxiety and depressive symptoms (CADS) the relationship to quality of life, fatigue, and mood at 12 and 24 months after surgery in women with breast cancer (J. Affect. Disord. 2012;136:895-901).
They recruited 1,501 women referred by the national screening program or their general practitioners to six hospitals. These women, who had not yet received a diagnosis, completed a set of questionnaires that measured state anxiety, the presence and degree of depressive symptoms over the previous week, fatigue, quality of life, personality trait neuroticism, and trait anxiety.
Among these subjects, 407 women (27% of all subjects) were diagnosed with breast cancer, and 111 (28% of those diagnosed with cancer) had CADS at baseline. These patients completed questionnaires again at 12 and 24 months after surgery, although 75 dropped out between baseline and 12 months, and 24 dropped out between 12 and 14 months. The researchers also gathered demographic data and medical information on disease stage at diagnosis, type of operation and adjuvant treatment, such as chemotherapy, radiotherapy, or hormone treatment.
Of the remaining 332 patients who were in the study at 12 months, 14% had CADS, the results show. And, 10% of the remaining 246 patients had CADS at 24 months, and 10% had CADS at 12 and 24 months, respectively.
And, 10% of the remaining 246 patients at 24 months, 10% had CADS at 12 and 24 months, respectively.
At 12 and 24 months, 21% of the group had elevated depressive symptoms, and 20% had state anxiety. Of the group that had CADS at baseline, a greater percentage had elevated levels of anxiety, depressive symptoms, and CADS at 12 and 24 months than did the group that did not have CADS at baseline.
"[The] CADS group had elevated levels of state anxiety, depressive symptoms, and CADS at all follow-up measure moments in the 2 years after baseline, compared with the non-CADS group," the researchers said. "In both groups, these scores decreased over time but, after 24 months, the CADS, state anxiety, and depressive symptoms scores of the CADS group were still about three times as high as the scores in the non-CADS group. This implies that the CADS group experienced considerably more anxiety and/or depressive symptoms even 24 months after surgery."
On further analysis, the researchers found that CADS and neuroticism were both significant predictors of patients’ quality of life, fatigue, depressive symptoms, and state anxiety at 12 and 24 months after surgery, the researchers found.
The findings in this study suggest that clinicians use questionnaires or screening instruments to identify those women with a higher score on state anxiety and depressive symptom, the researchers say, and that they should do so as soon as possible when diagnosing and treating breast cancer. "Only by identifying this group of patients, tailored psychological care can be accomplished," they add.
The study’s strength was its prospective multicenter and longitudinal design. A limitation, however, was that some data were not available because some patients dropped out, and because some women were included less than 2 years earlier. Also, those who dropped out often cited a high stress level, meaning that anxiety levels might have been higher in the patients who did not participate.
The authors had no conflicts of interest to declare, and there were no study sponsors.
FROM THE JOURNAL OF AFFECTIVE DISORDERS
Major Finding: Almost one in four women had a combined state anxiety and depressive symptoms before their diagnosis of breast cancer, and a higher percentage had elevated levels of anxiety, depressive symptoms, and CADS at 12- and 24-month follow-up.
Data Source: A multicenter prospective study of 1,501 women referred to six hospitals.
Disclosures: The authors had no conflicts of interest to declare, and there were no study sponsors.
FDA: Continue Study of Antinerve Growth Factors for Pain
SILVER SPRING, MD. – The clinical development of antinerve growth factors as a treatment for pain should continue, despite reports of serious joint-related adverse effects associated with these agents in clinical trials, a Food and Drug Administration panel unanimously agreed on March 12.
At the meeting, the FDA’s Arthritis Advisory Committee voted 21-0 that there was a role for the ongoing development of antinerve growth factors (anti-NGF), which panelists agreed appeared to show promise as analgesics in studies and may eventually have a niche in the treatment of chronic pain conditions. But they strongly recommended that future studies should closely monitor patients for rapidly progressing osteoarthritis (OA) and other joint-related adverse events that have been observed in trials, and include a rigorous informed-consent process so that patients are well informed about the potential risks. The panel also recommended that the manufacturers of these products conduct studies to investigate the mechanism behind the joint-related adverse events, and that in clinical trials, the use of concomitant NSAIDs should be contraindicated or limited.
The FDA convened the panel to discuss reports of joint destruction in patients treated with two of the three agents in clinical development, which resulted in a hold on clinical trials of all three agents, tanezumab (Pfizer), fulranumab (Janssen), and REGN475 (Regeneron). Anti-NGF agents, monoclonal antibodies directed against nerve growth factor, are being studied for chronic pain caused by various conditions, primarily osteoarthritis. The greatest amounts of data are available on tanezumab, which was being studied in phase III trials of patients with OA and chronic low back pain.
The FDA put a hold on clinical trials of the three anti-NGF factors in 2010, after cases of osteonecrosis and avascular necrosis were reported in osteoarthritis patients treated with tanezumab and in those treated with fulranumab, because of concerns that these cases might be a class effect. There were no such cases among patients on placebo or comparator drugs. The risk increased further among those on an NSAID and an anti-NGF agent, compared with the anti-NGF agent alone.
Panelists agreed with the FDA reviewers that that these cases were unusual and severe.
In addition to OA, the companies are developing these agents for other chronic pain conditions, including low back pain, postherpetic neuralgia, chronic pancreatitis, endometriosis, and cancer pain.
The FDA usually follows the recommendations of its advisory panels. Panelists have been cleared of potential conflicts of interest related to the topic of the meeting. Occasionally, a panelist may be given a waiver, but not at this meeting.
SILVER SPRING, MD. – The clinical development of antinerve growth factors as a treatment for pain should continue, despite reports of serious joint-related adverse effects associated with these agents in clinical trials, a Food and Drug Administration panel unanimously agreed on March 12.
At the meeting, the FDA’s Arthritis Advisory Committee voted 21-0 that there was a role for the ongoing development of antinerve growth factors (anti-NGF), which panelists agreed appeared to show promise as analgesics in studies and may eventually have a niche in the treatment of chronic pain conditions. But they strongly recommended that future studies should closely monitor patients for rapidly progressing osteoarthritis (OA) and other joint-related adverse events that have been observed in trials, and include a rigorous informed-consent process so that patients are well informed about the potential risks. The panel also recommended that the manufacturers of these products conduct studies to investigate the mechanism behind the joint-related adverse events, and that in clinical trials, the use of concomitant NSAIDs should be contraindicated or limited.
The FDA convened the panel to discuss reports of joint destruction in patients treated with two of the three agents in clinical development, which resulted in a hold on clinical trials of all three agents, tanezumab (Pfizer), fulranumab (Janssen), and REGN475 (Regeneron). Anti-NGF agents, monoclonal antibodies directed against nerve growth factor, are being studied for chronic pain caused by various conditions, primarily osteoarthritis. The greatest amounts of data are available on tanezumab, which was being studied in phase III trials of patients with OA and chronic low back pain.
The FDA put a hold on clinical trials of the three anti-NGF factors in 2010, after cases of osteonecrosis and avascular necrosis were reported in osteoarthritis patients treated with tanezumab and in those treated with fulranumab, because of concerns that these cases might be a class effect. There were no such cases among patients on placebo or comparator drugs. The risk increased further among those on an NSAID and an anti-NGF agent, compared with the anti-NGF agent alone.
Panelists agreed with the FDA reviewers that that these cases were unusual and severe.
In addition to OA, the companies are developing these agents for other chronic pain conditions, including low back pain, postherpetic neuralgia, chronic pancreatitis, endometriosis, and cancer pain.
The FDA usually follows the recommendations of its advisory panels. Panelists have been cleared of potential conflicts of interest related to the topic of the meeting. Occasionally, a panelist may be given a waiver, but not at this meeting.
SILVER SPRING, MD. – The clinical development of antinerve growth factors as a treatment for pain should continue, despite reports of serious joint-related adverse effects associated with these agents in clinical trials, a Food and Drug Administration panel unanimously agreed on March 12.
At the meeting, the FDA’s Arthritis Advisory Committee voted 21-0 that there was a role for the ongoing development of antinerve growth factors (anti-NGF), which panelists agreed appeared to show promise as analgesics in studies and may eventually have a niche in the treatment of chronic pain conditions. But they strongly recommended that future studies should closely monitor patients for rapidly progressing osteoarthritis (OA) and other joint-related adverse events that have been observed in trials, and include a rigorous informed-consent process so that patients are well informed about the potential risks. The panel also recommended that the manufacturers of these products conduct studies to investigate the mechanism behind the joint-related adverse events, and that in clinical trials, the use of concomitant NSAIDs should be contraindicated or limited.
The FDA convened the panel to discuss reports of joint destruction in patients treated with two of the three agents in clinical development, which resulted in a hold on clinical trials of all three agents, tanezumab (Pfizer), fulranumab (Janssen), and REGN475 (Regeneron). Anti-NGF agents, monoclonal antibodies directed against nerve growth factor, are being studied for chronic pain caused by various conditions, primarily osteoarthritis. The greatest amounts of data are available on tanezumab, which was being studied in phase III trials of patients with OA and chronic low back pain.
The FDA put a hold on clinical trials of the three anti-NGF factors in 2010, after cases of osteonecrosis and avascular necrosis were reported in osteoarthritis patients treated with tanezumab and in those treated with fulranumab, because of concerns that these cases might be a class effect. There were no such cases among patients on placebo or comparator drugs. The risk increased further among those on an NSAID and an anti-NGF agent, compared with the anti-NGF agent alone.
Panelists agreed with the FDA reviewers that that these cases were unusual and severe.
In addition to OA, the companies are developing these agents for other chronic pain conditions, including low back pain, postherpetic neuralgia, chronic pancreatitis, endometriosis, and cancer pain.
The FDA usually follows the recommendations of its advisory panels. Panelists have been cleared of potential conflicts of interest related to the topic of the meeting. Occasionally, a panelist may be given a waiver, but not at this meeting.
FROM A MEETING OF THE FDA'S ARTHRITIS ADVISORY COMMITTEE
Long-Acting Opioids May Trigger Hypogonadism
PALM SPRINGS, CALIF. – Low testosterone levels were nearly five times more likely in men on long-acting or sustained-release opioids, compared with equipotent short-acting or immediate-release formulations, judging from the findings of a study of 81 men on daily opioids for chronic pain.
Neither the age of the patient nor the total daily dose of opioid significantly affected the risk of hypogonadism, Dr. Andrea Rubenstein reported in her award-winning poster and a plenary presentation at the annual meeting of the American Academy of Pain Medicine.
With her associates, Dr. Rubenstein reviewed records on the morning measurements of total testosterone levels in men who were on a stable dose of an opioid for at least 3 months and who had no history of hypogonadism. In all, 46 of the 81 men had total testosterone levels below 25 ng/dl (57%), consistent with published reports of a high rate of hypogonadism in men on chronic opioid therapy ranging from 54% to 86%.
Previous studies, however, evaluated only men on sustained-release or intrathecal formulations and could not identify what aspect of opioid use may be contributing to hypogonadism.
The current study found that 34 of 46 men on long-acting or sustained-release opioids had low total testosterone levels (74%), compared with 12 of 35 men on short-acting or immediate-release opioids (34%), a significant difference, said Dr. Rubenstein, an anesthesiologist and pain specialist at Kaiser Permanente Medical Group, Santa Rosa, Calif.
After adjusting for the effects of dosage and body mass index (BMI), men on long-acting or sustained-release opioids were 4.8 times more likely to have low testosterone levels, compared with men on short-acting or immediate-release formulations.
Higher BMI had a small but significant association, increasing the risk for hypogonadism by 13%.
Opioid-related androgen deficiency has been documented in one fashion or another since the 1970s, and appears to come on quickly, within days or weeks of starting chronic opioid therapy, she said.
"This phenomenon is not new, even though it’s kind of a hot topic this year," Dr. Rubenstein said.
For the past 20 years, the trend has been to put more and more patients on long-acting opioids because these were believed to be safer than short-acting formulations. If the association between hypogonadism and long-acting opioids holds up in further studies, "it will be the first evidence of a difference in safety, though not in the direction we had thought," she said.
Dr. Steven Linder of the Veterans Affairs Healthcare System Hospital, Palo Alto, Calif., commented on the study in an interview at the poster presentation. The V.A. is seeing a large number of young veterans with spinal and other injuries who are on long-term opioids, and often these patients are not screened for hypogonadism. Dr. Rubenstein’s study is an important reminder to check testosterone levels in men on long-acting or sustained-release opioids, he said.
If unrecognized and untreated, hypogonadism can lead to osteoporosis, low libido, lower function and mood, insulin resistance, increased pain, and obesity. Often, these are managed with other medications that contribute side effects, Dr. Rubenstein said.
"The last thing we need in a guy on 40 mg of methadone for back pain is to get osteoporosis of the spine," she said.
Patients in the study included 25 on hydrocodone, 8 on continuous-release oxycodone, 10 on immediate-release oxycodone, 12 on continuous-release morphine, 4 on the fentanyl patch, 14 on methadone, and 8 on off-label sublingual buprenorphine. (Only the patch form of buprenorphine is approved to treat pain.)
The study was limited by its small size, "but in the opioid literature, 81 is a nice number," she said. The study also is limited by its retrospective design, potential bias if the symptoms of hypogonadism played a role in the initial referral, and questionable generalizability if the men at this tertiary-care pain clinic are not representative of men with chronic pain in the general population.
The investigators next will repeat the study on a much larger sample of patients, and if the association is replicated, will design a prospective study to see if changing opioid therapy modifies the risk for hypogonadism. The findings of the current study are too preliminary to generate recommendations, she said.
The current study excluded patients with a history of cancer, HIV, or endocrine disease other than hypothyroidism.
Dr. Rubenstein and her associates reported having no financial disclosures.
PALM SPRINGS, CALIF. – Low testosterone levels were nearly five times more likely in men on long-acting or sustained-release opioids, compared with equipotent short-acting or immediate-release formulations, judging from the findings of a study of 81 men on daily opioids for chronic pain.
Neither the age of the patient nor the total daily dose of opioid significantly affected the risk of hypogonadism, Dr. Andrea Rubenstein reported in her award-winning poster and a plenary presentation at the annual meeting of the American Academy of Pain Medicine.
With her associates, Dr. Rubenstein reviewed records on the morning measurements of total testosterone levels in men who were on a stable dose of an opioid for at least 3 months and who had no history of hypogonadism. In all, 46 of the 81 men had total testosterone levels below 25 ng/dl (57%), consistent with published reports of a high rate of hypogonadism in men on chronic opioid therapy ranging from 54% to 86%.
Previous studies, however, evaluated only men on sustained-release or intrathecal formulations and could not identify what aspect of opioid use may be contributing to hypogonadism.
The current study found that 34 of 46 men on long-acting or sustained-release opioids had low total testosterone levels (74%), compared with 12 of 35 men on short-acting or immediate-release opioids (34%), a significant difference, said Dr. Rubenstein, an anesthesiologist and pain specialist at Kaiser Permanente Medical Group, Santa Rosa, Calif.
After adjusting for the effects of dosage and body mass index (BMI), men on long-acting or sustained-release opioids were 4.8 times more likely to have low testosterone levels, compared with men on short-acting or immediate-release formulations.
Higher BMI had a small but significant association, increasing the risk for hypogonadism by 13%.
Opioid-related androgen deficiency has been documented in one fashion or another since the 1970s, and appears to come on quickly, within days or weeks of starting chronic opioid therapy, she said.
"This phenomenon is not new, even though it’s kind of a hot topic this year," Dr. Rubenstein said.
For the past 20 years, the trend has been to put more and more patients on long-acting opioids because these were believed to be safer than short-acting formulations. If the association between hypogonadism and long-acting opioids holds up in further studies, "it will be the first evidence of a difference in safety, though not in the direction we had thought," she said.
Dr. Steven Linder of the Veterans Affairs Healthcare System Hospital, Palo Alto, Calif., commented on the study in an interview at the poster presentation. The V.A. is seeing a large number of young veterans with spinal and other injuries who are on long-term opioids, and often these patients are not screened for hypogonadism. Dr. Rubenstein’s study is an important reminder to check testosterone levels in men on long-acting or sustained-release opioids, he said.
If unrecognized and untreated, hypogonadism can lead to osteoporosis, low libido, lower function and mood, insulin resistance, increased pain, and obesity. Often, these are managed with other medications that contribute side effects, Dr. Rubenstein said.
"The last thing we need in a guy on 40 mg of methadone for back pain is to get osteoporosis of the spine," she said.
Patients in the study included 25 on hydrocodone, 8 on continuous-release oxycodone, 10 on immediate-release oxycodone, 12 on continuous-release morphine, 4 on the fentanyl patch, 14 on methadone, and 8 on off-label sublingual buprenorphine. (Only the patch form of buprenorphine is approved to treat pain.)
The study was limited by its small size, "but in the opioid literature, 81 is a nice number," she said. The study also is limited by its retrospective design, potential bias if the symptoms of hypogonadism played a role in the initial referral, and questionable generalizability if the men at this tertiary-care pain clinic are not representative of men with chronic pain in the general population.
The investigators next will repeat the study on a much larger sample of patients, and if the association is replicated, will design a prospective study to see if changing opioid therapy modifies the risk for hypogonadism. The findings of the current study are too preliminary to generate recommendations, she said.
The current study excluded patients with a history of cancer, HIV, or endocrine disease other than hypothyroidism.
Dr. Rubenstein and her associates reported having no financial disclosures.
PALM SPRINGS, CALIF. – Low testosterone levels were nearly five times more likely in men on long-acting or sustained-release opioids, compared with equipotent short-acting or immediate-release formulations, judging from the findings of a study of 81 men on daily opioids for chronic pain.
Neither the age of the patient nor the total daily dose of opioid significantly affected the risk of hypogonadism, Dr. Andrea Rubenstein reported in her award-winning poster and a plenary presentation at the annual meeting of the American Academy of Pain Medicine.
With her associates, Dr. Rubenstein reviewed records on the morning measurements of total testosterone levels in men who were on a stable dose of an opioid for at least 3 months and who had no history of hypogonadism. In all, 46 of the 81 men had total testosterone levels below 25 ng/dl (57%), consistent with published reports of a high rate of hypogonadism in men on chronic opioid therapy ranging from 54% to 86%.
Previous studies, however, evaluated only men on sustained-release or intrathecal formulations and could not identify what aspect of opioid use may be contributing to hypogonadism.
The current study found that 34 of 46 men on long-acting or sustained-release opioids had low total testosterone levels (74%), compared with 12 of 35 men on short-acting or immediate-release opioids (34%), a significant difference, said Dr. Rubenstein, an anesthesiologist and pain specialist at Kaiser Permanente Medical Group, Santa Rosa, Calif.
After adjusting for the effects of dosage and body mass index (BMI), men on long-acting or sustained-release opioids were 4.8 times more likely to have low testosterone levels, compared with men on short-acting or immediate-release formulations.
Higher BMI had a small but significant association, increasing the risk for hypogonadism by 13%.
Opioid-related androgen deficiency has been documented in one fashion or another since the 1970s, and appears to come on quickly, within days or weeks of starting chronic opioid therapy, she said.
"This phenomenon is not new, even though it’s kind of a hot topic this year," Dr. Rubenstein said.
For the past 20 years, the trend has been to put more and more patients on long-acting opioids because these were believed to be safer than short-acting formulations. If the association between hypogonadism and long-acting opioids holds up in further studies, "it will be the first evidence of a difference in safety, though not in the direction we had thought," she said.
Dr. Steven Linder of the Veterans Affairs Healthcare System Hospital, Palo Alto, Calif., commented on the study in an interview at the poster presentation. The V.A. is seeing a large number of young veterans with spinal and other injuries who are on long-term opioids, and often these patients are not screened for hypogonadism. Dr. Rubenstein’s study is an important reminder to check testosterone levels in men on long-acting or sustained-release opioids, he said.
If unrecognized and untreated, hypogonadism can lead to osteoporosis, low libido, lower function and mood, insulin resistance, increased pain, and obesity. Often, these are managed with other medications that contribute side effects, Dr. Rubenstein said.
"The last thing we need in a guy on 40 mg of methadone for back pain is to get osteoporosis of the spine," she said.
Patients in the study included 25 on hydrocodone, 8 on continuous-release oxycodone, 10 on immediate-release oxycodone, 12 on continuous-release morphine, 4 on the fentanyl patch, 14 on methadone, and 8 on off-label sublingual buprenorphine. (Only the patch form of buprenorphine is approved to treat pain.)
The study was limited by its small size, "but in the opioid literature, 81 is a nice number," she said. The study also is limited by its retrospective design, potential bias if the symptoms of hypogonadism played a role in the initial referral, and questionable generalizability if the men at this tertiary-care pain clinic are not representative of men with chronic pain in the general population.
The investigators next will repeat the study on a much larger sample of patients, and if the association is replicated, will design a prospective study to see if changing opioid therapy modifies the risk for hypogonadism. The findings of the current study are too preliminary to generate recommendations, she said.
The current study excluded patients with a history of cancer, HIV, or endocrine disease other than hypothyroidism.
Dr. Rubenstein and her associates reported having no financial disclosures.
FROM THE ANNUAL MEETING OF THE AMERICAN ACADEMY OF PAIN MEDICINE
Major Finding: Tests found low testosterone levels in 74% of men on long-acting or sustained-release opioids, compared with 34% of men on equipotent short-acting or immediate-release opioids.
Data Source: The review included records on 81 men treated with daily opioids for chronic pain at one institution.
Disclosures: Dr. Rubenstein and her associates reported having no financial disclosures.
Surrogates' Emotions Influence Decisions Made for Critically Ill
Surrogate decision makers for incapacitated, critically ill patients systematically interpreted prognostic information as being more optimistic than it actually was, in a study published in the March 6 issue of the Annals of Internal Medicine.
These surrogate decision makers accurately interpreted prognostic information that was positive, but not prognostic information expressing a high risk of death. Instead, they showed "a systematic bias," consistently interpreting "grim" prognostic statements in an overly optimistic way, said Dr. Lucas S. Zier of the University of California, San Francisco, and his associates.
"These findings challenge the prevailing assumption in the medical literature that discordance between physicians and surrogates about prognosis is due largely to unclear disclosure by physicians or simple misunderstandings by surrogates," the investigators noted.
The results also indicate that any efforts to improve this aspect of decision making must address not just the clarity of prognostic statements but also the "emotional and psychological factors that affect how individuals process such information," they added.
"Clinicians who communicate with surrogate decision makers in the care of incapacitated patients should be aware of the diverse causes for discordance about prognosis."
Dr. Zier and his colleagues examined how surrogate decision makers interpret physicians’ statements about their loved ones’ prognosis by administering a questionnaire to 80 such surrogates who were accompanying an incapacitated patient at medical-surgical ICUs in a Veterans Affairs hospital, a tertiary academic hospital, and a public county hospital.
The questionnaire presented 16 possible prognostic statements in the language used by physicians and made it clear that these represented hypothetical clinical situations unrelated to their loved one’s cases. Study subjects were asked what exactly each prognostic statement meant to them, and used a numerical scale to demarcate the patient’s chance of survival corresponding to each prognostic statement.
Examples of general and somewhat equivocal prognostic statements included "It is very likely that he will survive," "It is very unlikely that he will die," "I am concerned that he will not survive," "It is possible he will not survive," and "He probably will not survive." Unequivocal prognostic statements were "He will definitely survive" and "He will definitely not survive."
There also were three unequivocal numerical prognostic statements: "He has a 90% chance of surviving," "He has a 50% chance of surviving," and "He has a 5% chance of surviving."
Only twelve surrogate decision makers (15% of the entire group) interpreted the three numerical prognostic statements accurately. Almost as many – 13% –interpreted all three numerical prognostic statements more optimistically than they actually were.
Most surrogates interpreted the statement of a low risk of death ("90% chance of surviving") accurately, but severely misinterpreted the statements of a median or high risk of death. Forty percent thought that "a 50% chance of surviving" meant that the patient was likely to survive, and 65% thought that "a 5% chance of surviving" meant that the patient was likely to survive.
No surrogates interpreted any of the numerical prognostic statements more pessimistically than they actually were.
Some experts have advocated using straightforward, numerical language when communicating medical risk to surrogate decision makers, but these findings clearly show that numbers are not straightforward to everyone and are frequently misinterpreted, Dr. Zier and his associates said (Ann. Intern. Med. 2012;156:360-6).
In the second phase of this study, 15 subjects whose interpretations of prognostic statements had been particularly off-base were interviewed about why there was such a discrepancy between what the physician said and what the subject thought was meant.
Seven of the 15 said they were unaware that almost all their interpretations were overly optimistic. These subjects were surprised when it was pointed out to them and couldn’t offer of an explanation.
The other eight surrogate decision makers gave four reasons for their "optimism bias."
Some said they intentionally expressed optimism as long as there was any hope whatsoever. This may represent a coping strategy to help surrogates deal with having a critically ill loved one. Or it may represent "magical thinking" in which people believe their positive thoughts and expectations can actually improve the patient’s outcome, the researchers said.
Other surrogate decision makers said they believed their loved one was exceptional and wouldn’t die because of an unusual will to live and ability to survive. This "may represent a cognitive bias known as illusory superiority, unrealistic optimism, or the ‘Lake Wobegon effect,’ a cognitive bias that leads people to overestimate, in relation to others, their likelihood of experiencing positive outcomes and avoiding negative outcomes," the investigators said.
Some study subjects said they intentionally ignored numerical probabilities and precise language, preferring to judge "the overall feeling that the doctor is conveying."
And some said they didn’t believe physicians could ever accurately predict survival. However, such skepticism about physicians’ accuracy would result in a random distribution of interpretations rather than in the systematic optimism observed here, Dr. Zier and his colleagues noted.
The findings of this study show that "clinicians who communicate with surrogate decision makers in the care of incapacitated patients should be aware of the diverse causes for discordance about prognosis." Unrealistic optimism may not be benign; it can lead to treatment decisions that do not reflect the true values of the patient but that instead serve the surrogate decision maker’s impulse toward self-protection, they added.
This study was supported by the National Heart, Lung, and Blood Institute, the Greenwall Foundation, and the University of California, Berkeley–University of California, San Francisco, Joint Medical Program.
Surrogate decision makers for incapacitated, critically ill patients systematically interpreted prognostic information as being more optimistic than it actually was, in a study published in the March 6 issue of the Annals of Internal Medicine.
These surrogate decision makers accurately interpreted prognostic information that was positive, but not prognostic information expressing a high risk of death. Instead, they showed "a systematic bias," consistently interpreting "grim" prognostic statements in an overly optimistic way, said Dr. Lucas S. Zier of the University of California, San Francisco, and his associates.
"These findings challenge the prevailing assumption in the medical literature that discordance between physicians and surrogates about prognosis is due largely to unclear disclosure by physicians or simple misunderstandings by surrogates," the investigators noted.
The results also indicate that any efforts to improve this aspect of decision making must address not just the clarity of prognostic statements but also the "emotional and psychological factors that affect how individuals process such information," they added.
"Clinicians who communicate with surrogate decision makers in the care of incapacitated patients should be aware of the diverse causes for discordance about prognosis."
Dr. Zier and his colleagues examined how surrogate decision makers interpret physicians’ statements about their loved ones’ prognosis by administering a questionnaire to 80 such surrogates who were accompanying an incapacitated patient at medical-surgical ICUs in a Veterans Affairs hospital, a tertiary academic hospital, and a public county hospital.
The questionnaire presented 16 possible prognostic statements in the language used by physicians and made it clear that these represented hypothetical clinical situations unrelated to their loved one’s cases. Study subjects were asked what exactly each prognostic statement meant to them, and used a numerical scale to demarcate the patient’s chance of survival corresponding to each prognostic statement.
Examples of general and somewhat equivocal prognostic statements included "It is very likely that he will survive," "It is very unlikely that he will die," "I am concerned that he will not survive," "It is possible he will not survive," and "He probably will not survive." Unequivocal prognostic statements were "He will definitely survive" and "He will definitely not survive."
There also were three unequivocal numerical prognostic statements: "He has a 90% chance of surviving," "He has a 50% chance of surviving," and "He has a 5% chance of surviving."
Only twelve surrogate decision makers (15% of the entire group) interpreted the three numerical prognostic statements accurately. Almost as many – 13% –interpreted all three numerical prognostic statements more optimistically than they actually were.
Most surrogates interpreted the statement of a low risk of death ("90% chance of surviving") accurately, but severely misinterpreted the statements of a median or high risk of death. Forty percent thought that "a 50% chance of surviving" meant that the patient was likely to survive, and 65% thought that "a 5% chance of surviving" meant that the patient was likely to survive.
No surrogates interpreted any of the numerical prognostic statements more pessimistically than they actually were.
Some experts have advocated using straightforward, numerical language when communicating medical risk to surrogate decision makers, but these findings clearly show that numbers are not straightforward to everyone and are frequently misinterpreted, Dr. Zier and his associates said (Ann. Intern. Med. 2012;156:360-6).
In the second phase of this study, 15 subjects whose interpretations of prognostic statements had been particularly off-base were interviewed about why there was such a discrepancy between what the physician said and what the subject thought was meant.
Seven of the 15 said they were unaware that almost all their interpretations were overly optimistic. These subjects were surprised when it was pointed out to them and couldn’t offer of an explanation.
The other eight surrogate decision makers gave four reasons for their "optimism bias."
Some said they intentionally expressed optimism as long as there was any hope whatsoever. This may represent a coping strategy to help surrogates deal with having a critically ill loved one. Or it may represent "magical thinking" in which people believe their positive thoughts and expectations can actually improve the patient’s outcome, the researchers said.
Other surrogate decision makers said they believed their loved one was exceptional and wouldn’t die because of an unusual will to live and ability to survive. This "may represent a cognitive bias known as illusory superiority, unrealistic optimism, or the ‘Lake Wobegon effect,’ a cognitive bias that leads people to overestimate, in relation to others, their likelihood of experiencing positive outcomes and avoiding negative outcomes," the investigators said.
Some study subjects said they intentionally ignored numerical probabilities and precise language, preferring to judge "the overall feeling that the doctor is conveying."
And some said they didn’t believe physicians could ever accurately predict survival. However, such skepticism about physicians’ accuracy would result in a random distribution of interpretations rather than in the systematic optimism observed here, Dr. Zier and his colleagues noted.
The findings of this study show that "clinicians who communicate with surrogate decision makers in the care of incapacitated patients should be aware of the diverse causes for discordance about prognosis." Unrealistic optimism may not be benign; it can lead to treatment decisions that do not reflect the true values of the patient but that instead serve the surrogate decision maker’s impulse toward self-protection, they added.
This study was supported by the National Heart, Lung, and Blood Institute, the Greenwall Foundation, and the University of California, Berkeley–University of California, San Francisco, Joint Medical Program.
Surrogate decision makers for incapacitated, critically ill patients systematically interpreted prognostic information as being more optimistic than it actually was, in a study published in the March 6 issue of the Annals of Internal Medicine.
These surrogate decision makers accurately interpreted prognostic information that was positive, but not prognostic information expressing a high risk of death. Instead, they showed "a systematic bias," consistently interpreting "grim" prognostic statements in an overly optimistic way, said Dr. Lucas S. Zier of the University of California, San Francisco, and his associates.
"These findings challenge the prevailing assumption in the medical literature that discordance between physicians and surrogates about prognosis is due largely to unclear disclosure by physicians or simple misunderstandings by surrogates," the investigators noted.
The results also indicate that any efforts to improve this aspect of decision making must address not just the clarity of prognostic statements but also the "emotional and psychological factors that affect how individuals process such information," they added.
"Clinicians who communicate with surrogate decision makers in the care of incapacitated patients should be aware of the diverse causes for discordance about prognosis."
Dr. Zier and his colleagues examined how surrogate decision makers interpret physicians’ statements about their loved ones’ prognosis by administering a questionnaire to 80 such surrogates who were accompanying an incapacitated patient at medical-surgical ICUs in a Veterans Affairs hospital, a tertiary academic hospital, and a public county hospital.
The questionnaire presented 16 possible prognostic statements in the language used by physicians and made it clear that these represented hypothetical clinical situations unrelated to their loved one’s cases. Study subjects were asked what exactly each prognostic statement meant to them, and used a numerical scale to demarcate the patient’s chance of survival corresponding to each prognostic statement.
Examples of general and somewhat equivocal prognostic statements included "It is very likely that he will survive," "It is very unlikely that he will die," "I am concerned that he will not survive," "It is possible he will not survive," and "He probably will not survive." Unequivocal prognostic statements were "He will definitely survive" and "He will definitely not survive."
There also were three unequivocal numerical prognostic statements: "He has a 90% chance of surviving," "He has a 50% chance of surviving," and "He has a 5% chance of surviving."
Only twelve surrogate decision makers (15% of the entire group) interpreted the three numerical prognostic statements accurately. Almost as many – 13% –interpreted all three numerical prognostic statements more optimistically than they actually were.
Most surrogates interpreted the statement of a low risk of death ("90% chance of surviving") accurately, but severely misinterpreted the statements of a median or high risk of death. Forty percent thought that "a 50% chance of surviving" meant that the patient was likely to survive, and 65% thought that "a 5% chance of surviving" meant that the patient was likely to survive.
No surrogates interpreted any of the numerical prognostic statements more pessimistically than they actually were.
Some experts have advocated using straightforward, numerical language when communicating medical risk to surrogate decision makers, but these findings clearly show that numbers are not straightforward to everyone and are frequently misinterpreted, Dr. Zier and his associates said (Ann. Intern. Med. 2012;156:360-6).
In the second phase of this study, 15 subjects whose interpretations of prognostic statements had been particularly off-base were interviewed about why there was such a discrepancy between what the physician said and what the subject thought was meant.
Seven of the 15 said they were unaware that almost all their interpretations were overly optimistic. These subjects were surprised when it was pointed out to them and couldn’t offer of an explanation.
The other eight surrogate decision makers gave four reasons for their "optimism bias."
Some said they intentionally expressed optimism as long as there was any hope whatsoever. This may represent a coping strategy to help surrogates deal with having a critically ill loved one. Or it may represent "magical thinking" in which people believe their positive thoughts and expectations can actually improve the patient’s outcome, the researchers said.
Other surrogate decision makers said they believed their loved one was exceptional and wouldn’t die because of an unusual will to live and ability to survive. This "may represent a cognitive bias known as illusory superiority, unrealistic optimism, or the ‘Lake Wobegon effect,’ a cognitive bias that leads people to overestimate, in relation to others, their likelihood of experiencing positive outcomes and avoiding negative outcomes," the investigators said.
Some study subjects said they intentionally ignored numerical probabilities and precise language, preferring to judge "the overall feeling that the doctor is conveying."
And some said they didn’t believe physicians could ever accurately predict survival. However, such skepticism about physicians’ accuracy would result in a random distribution of interpretations rather than in the systematic optimism observed here, Dr. Zier and his colleagues noted.
The findings of this study show that "clinicians who communicate with surrogate decision makers in the care of incapacitated patients should be aware of the diverse causes for discordance about prognosis." Unrealistic optimism may not be benign; it can lead to treatment decisions that do not reflect the true values of the patient but that instead serve the surrogate decision maker’s impulse toward self-protection, they added.
This study was supported by the National Heart, Lung, and Blood Institute, the Greenwall Foundation, and the University of California, Berkeley–University of California, San Francisco, Joint Medical Program.
FROM THE ANNALS OF INTERNAL MEDICINE
Major Finding: Only 12 of 80 surrogate decision makers for incapacitated, critically ill patients interpreted direct numerical predictions of survival accurately; the rest consistently put an overly optimistic spin on "grim" prognostic statements.
Data Source: A questionnaire survey was done with 80 surrogate decision-makers plus in-person interviews of a subgroup of 15 of these surrogates.
Disclosures: This study was supported by the National Heart, Lung, and Blood Institute, the Greenwall Foundation, and the University of California, Berkeley–University of California, San Francisco Joint Medical Program.
Off-Label Use of Aprepitant Quells Refractory Pruritus
WAIKOLOA, HAWAII – Oral aprepitant has shown considerable promise as a novel therapy in patients with refractory pruritus, including those with severe itching induced by biologic therapies for cancer.
"I think it’s a tool we should consider using in our patients with pruritus who do not respond to the usual measures," Dr. Mario E. Lacouture said at the annual Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
Aprepitant (Emend) is approved as an antiemetic agent for cancer patients undergoing chemotherapy. But a growing body of data, including two single-arm proof-of-concept studies, has shown a high response rate in patients with itching resistant to standard treatment, including corticosteroids, UV therapy, and antihistamines, noted Dr. Lacouture, a dermatologist at Memorial Sloan-Kettering Cancer Center, New York.
Severe itching is increasingly recognized as an important side effect of targeted biologic agents for cancer, including erlotinib (Tarceva) and cetuximab (Erbitux). At last year’s meeting of the American Society of Clinical Oncology, Dr. Daniele Santini and coworkers at the Università "Campus Bio-Medico" di Roma presented a study involving 22 patients with severe pruritus treated with aprepitant using a regimen of 125 mg on day 1 and 80 mg on days 3 and 5.
After the single 3-dose cycle of aprepitant, the patients’ median pruritus intensity plummeted from a baseline of 8 down to 1 on a 0-10 visual analog scale (VAS). Twenty of 22 patients experienced a greater than 50% reduction in pruritus. The median duration of benefit following a 3-dose cycle given over 5 days was 25 days.
In the second proof-of-concept study, dermatologists at the Westfälische Wilhelms-Universität Münster (Germany) reported on 20 patients with refractory chronic pruritus from nonmalignant underlying disorders placed on aprepitant at 80 mg/day for 1 week. Sixteen of the 20 patients experienced a marked reduction in itch intensity. From a baseline mean of 8.4 points on the VAS, the group as a whole averaged a 42% improvement to a post-treatment score of 4.3.
A significant reduction in itching was observed as early as 2 days after initiating aprepitant in the study population (PLoS One. 2010;5:e10968).
Patients with itching related to prurigo nodularis or atopic disease responded better than those without the conditions, and patients under age 60 obtained greater improvement than older patients. However, patients with chronic kidney disease as the underlying cause of their itching achieved only minimal benefit.
Side effects were confined to mild, non-treatment-limiting nausea, dizziness, and drowsiness in three patients.
Between the two proof-of-concept studies and smaller published case series, Dr. Lacouture said that he is aware of 63 reported patients with severe refractory pruritus, including 6 with Sézary syndrome and 2 with mycosis fungoides, who were treated with aprepitant on various dosing regimens. The result was an improvement from a mean baseline VAS of 8.4 to 2.3.
Aprepitant’s mechanism of benefit in cases of severe pruritus is believed to lie in the drug’s high affinity as an antagonist of neurokinin receptor 1, which is expressed in the skin and CNS, he explained. Substance P, an important mediator in the initiation and maintenance of pruritus, binds to this receptor.
Dry skin due to radiation therapy, chemotherapy, or cachexia is the most common cause of pruritus in oncology. But he added that pruritus is intrinsically often a prominent feature of a variety of paraneoplastic diseases and malignancies, and aprepitant is worthy of study in refractory cases.
Pruritic paraneoplastic dermatoses in which the skin findings are an indication of potential concurrent underlying malignancy include dermatomyositis, erythroderma, generalized granuloma annulare, and acrokeratosis paraneoplastica.
Hematologic malignancies where itch often figures prominently include Hodgkin’s disease, polycythemia vera, and Waldenström macroglobulinemia. The solid cancers most frequently accompanied by severe itching include malignancies of the brain, prostate, rectum, and vulva.
Dr. Lacouture reported serving as a consultant to more than a dozen pharmaceutical companies, although Merck, which markets aprepitant, is not among them.
SDEF and this news organization are owned by Elsevier.
WAIKOLOA, HAWAII – Oral aprepitant has shown considerable promise as a novel therapy in patients with refractory pruritus, including those with severe itching induced by biologic therapies for cancer.
"I think it’s a tool we should consider using in our patients with pruritus who do not respond to the usual measures," Dr. Mario E. Lacouture said at the annual Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
Aprepitant (Emend) is approved as an antiemetic agent for cancer patients undergoing chemotherapy. But a growing body of data, including two single-arm proof-of-concept studies, has shown a high response rate in patients with itching resistant to standard treatment, including corticosteroids, UV therapy, and antihistamines, noted Dr. Lacouture, a dermatologist at Memorial Sloan-Kettering Cancer Center, New York.
Severe itching is increasingly recognized as an important side effect of targeted biologic agents for cancer, including erlotinib (Tarceva) and cetuximab (Erbitux). At last year’s meeting of the American Society of Clinical Oncology, Dr. Daniele Santini and coworkers at the Università "Campus Bio-Medico" di Roma presented a study involving 22 patients with severe pruritus treated with aprepitant using a regimen of 125 mg on day 1 and 80 mg on days 3 and 5.
After the single 3-dose cycle of aprepitant, the patients’ median pruritus intensity plummeted from a baseline of 8 down to 1 on a 0-10 visual analog scale (VAS). Twenty of 22 patients experienced a greater than 50% reduction in pruritus. The median duration of benefit following a 3-dose cycle given over 5 days was 25 days.
In the second proof-of-concept study, dermatologists at the Westfälische Wilhelms-Universität Münster (Germany) reported on 20 patients with refractory chronic pruritus from nonmalignant underlying disorders placed on aprepitant at 80 mg/day for 1 week. Sixteen of the 20 patients experienced a marked reduction in itch intensity. From a baseline mean of 8.4 points on the VAS, the group as a whole averaged a 42% improvement to a post-treatment score of 4.3.
A significant reduction in itching was observed as early as 2 days after initiating aprepitant in the study population (PLoS One. 2010;5:e10968).
Patients with itching related to prurigo nodularis or atopic disease responded better than those without the conditions, and patients under age 60 obtained greater improvement than older patients. However, patients with chronic kidney disease as the underlying cause of their itching achieved only minimal benefit.
Side effects were confined to mild, non-treatment-limiting nausea, dizziness, and drowsiness in three patients.
Between the two proof-of-concept studies and smaller published case series, Dr. Lacouture said that he is aware of 63 reported patients with severe refractory pruritus, including 6 with Sézary syndrome and 2 with mycosis fungoides, who were treated with aprepitant on various dosing regimens. The result was an improvement from a mean baseline VAS of 8.4 to 2.3.
Aprepitant’s mechanism of benefit in cases of severe pruritus is believed to lie in the drug’s high affinity as an antagonist of neurokinin receptor 1, which is expressed in the skin and CNS, he explained. Substance P, an important mediator in the initiation and maintenance of pruritus, binds to this receptor.
Dry skin due to radiation therapy, chemotherapy, or cachexia is the most common cause of pruritus in oncology. But he added that pruritus is intrinsically often a prominent feature of a variety of paraneoplastic diseases and malignancies, and aprepitant is worthy of study in refractory cases.
Pruritic paraneoplastic dermatoses in which the skin findings are an indication of potential concurrent underlying malignancy include dermatomyositis, erythroderma, generalized granuloma annulare, and acrokeratosis paraneoplastica.
Hematologic malignancies where itch often figures prominently include Hodgkin’s disease, polycythemia vera, and Waldenström macroglobulinemia. The solid cancers most frequently accompanied by severe itching include malignancies of the brain, prostate, rectum, and vulva.
Dr. Lacouture reported serving as a consultant to more than a dozen pharmaceutical companies, although Merck, which markets aprepitant, is not among them.
SDEF and this news organization are owned by Elsevier.
WAIKOLOA, HAWAII – Oral aprepitant has shown considerable promise as a novel therapy in patients with refractory pruritus, including those with severe itching induced by biologic therapies for cancer.
"I think it’s a tool we should consider using in our patients with pruritus who do not respond to the usual measures," Dr. Mario E. Lacouture said at the annual Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
Aprepitant (Emend) is approved as an antiemetic agent for cancer patients undergoing chemotherapy. But a growing body of data, including two single-arm proof-of-concept studies, has shown a high response rate in patients with itching resistant to standard treatment, including corticosteroids, UV therapy, and antihistamines, noted Dr. Lacouture, a dermatologist at Memorial Sloan-Kettering Cancer Center, New York.
Severe itching is increasingly recognized as an important side effect of targeted biologic agents for cancer, including erlotinib (Tarceva) and cetuximab (Erbitux). At last year’s meeting of the American Society of Clinical Oncology, Dr. Daniele Santini and coworkers at the Università "Campus Bio-Medico" di Roma presented a study involving 22 patients with severe pruritus treated with aprepitant using a regimen of 125 mg on day 1 and 80 mg on days 3 and 5.
After the single 3-dose cycle of aprepitant, the patients’ median pruritus intensity plummeted from a baseline of 8 down to 1 on a 0-10 visual analog scale (VAS). Twenty of 22 patients experienced a greater than 50% reduction in pruritus. The median duration of benefit following a 3-dose cycle given over 5 days was 25 days.
In the second proof-of-concept study, dermatologists at the Westfälische Wilhelms-Universität Münster (Germany) reported on 20 patients with refractory chronic pruritus from nonmalignant underlying disorders placed on aprepitant at 80 mg/day for 1 week. Sixteen of the 20 patients experienced a marked reduction in itch intensity. From a baseline mean of 8.4 points on the VAS, the group as a whole averaged a 42% improvement to a post-treatment score of 4.3.
A significant reduction in itching was observed as early as 2 days after initiating aprepitant in the study population (PLoS One. 2010;5:e10968).
Patients with itching related to prurigo nodularis or atopic disease responded better than those without the conditions, and patients under age 60 obtained greater improvement than older patients. However, patients with chronic kidney disease as the underlying cause of their itching achieved only minimal benefit.
Side effects were confined to mild, non-treatment-limiting nausea, dizziness, and drowsiness in three patients.
Between the two proof-of-concept studies and smaller published case series, Dr. Lacouture said that he is aware of 63 reported patients with severe refractory pruritus, including 6 with Sézary syndrome and 2 with mycosis fungoides, who were treated with aprepitant on various dosing regimens. The result was an improvement from a mean baseline VAS of 8.4 to 2.3.
Aprepitant’s mechanism of benefit in cases of severe pruritus is believed to lie in the drug’s high affinity as an antagonist of neurokinin receptor 1, which is expressed in the skin and CNS, he explained. Substance P, an important mediator in the initiation and maintenance of pruritus, binds to this receptor.
Dry skin due to radiation therapy, chemotherapy, or cachexia is the most common cause of pruritus in oncology. But he added that pruritus is intrinsically often a prominent feature of a variety of paraneoplastic diseases and malignancies, and aprepitant is worthy of study in refractory cases.
Pruritic paraneoplastic dermatoses in which the skin findings are an indication of potential concurrent underlying malignancy include dermatomyositis, erythroderma, generalized granuloma annulare, and acrokeratosis paraneoplastica.
Hematologic malignancies where itch often figures prominently include Hodgkin’s disease, polycythemia vera, and Waldenström macroglobulinemia. The solid cancers most frequently accompanied by severe itching include malignancies of the brain, prostate, rectum, and vulva.
Dr. Lacouture reported serving as a consultant to more than a dozen pharmaceutical companies, although Merck, which markets aprepitant, is not among them.
SDEF and this news organization are owned by Elsevier.
EXPERT ANALYSIS FROM THE SDEF HAWAII DERMATOLOGY SEMINAR