User login
Medical cannabis legalization outpacing evidence
For now, CBD is better option than whole-plant cannabis for psychiatric disorders
SAN FRANCISCO – Outside of prescription products for chemotherapy nausea/vomiting, AIDS anorexia, and rare pediatric epilepsies, medical cannabis has the strongest evidence for chronic pain, neuropathic pain, and multiple sclerosis spasticity, according to Kevin P. Hill, MD, director of the division of addiction psychiatry at Beth Israel Deaconess Medical Center, Boston.
“We are talking about multiple, positive RCTs [randomized, controlled trials]. I think you can’t ignore that. For people who are staunchly opposed to medical cannabis, [it’s becoming] harder and harder to take that tack. I do think we need to come to an understanding that there is some evidence,” Dr. Hill said at the American Psychiatric Association annual meeting.
However, “there’s incredible interest in using cannabinoids for a whole host of [other] conditions for which the level of evidence isn’t where we’d like it to be. In many ways,” legalization has “far outpaced the evidence.
Perhaps that’s most true for psychiatric disorders. Although many patients swear by cannabis, what little evidence there is comes from observational studies, and those have mostly been disappointing.
Whole-plant cannabis, for instance, has been associated with an increased risk of depression in a dose-dependent fashion, and increased depression and anxiety symptoms, including panic attacks, among inexperienced users. Cutting back has been associated with symptom relief.
No placebo-controlled trials have been conducted to address cause and effect, so it’s unknown whether people use because they have worse disease or have worse disease because they use.
For now, “whole-plant cannabis to treat anxiety is probably not a good idea. We may ultimately find that cannabidiol” – CBD, the nonpsychoactive component of cannabis – “may have utility. If patients are already using, CBD is a better bet; it has no abuse potential,” Dr. Hill said.
He sometimes recommends CBD when patients fail traditional options, but over-the-counter or online products rather than the “exorbitantly expensive” Food and Drug Administration–approved version Epidiolex, indicated for the rare pediatric epilepsies Lennox-Gastaut and Dravet syndromes.
He asks patients to “tell me what you’re going to buy and we’ll go from there, and try to dose it.” Dr. Hill has confidence in only a few CBD brands to be accurately labeled, one of which is Charlotte’s Web.
There’s been a lot of excitement over cannabis for PTSD, but at this point, positive findings are mostly anecdotal, and use was associated with worse symptoms, increased violence, more alcohol and drug use, and worse therapy outcomes in a longitudinal study of 2,276 veterans. At least one ongoing RCT is underway that should address cause-and-effect (J Clin Psychiatry. 2015 Sep;76[9]:1174-80).
Like anxiety, CBD “would probably have more promise than whole-plant cannabis” for PTSD, Dr. Hill said.
That also might be the case for bipolar disorder. Whole-plant use is particularly common among patients, and it, again, seems to make symptoms worse.
The data for insomnia are much like that seen with alcohol: quicker asleep but worse sleep quality. “One of the myths about CBD is that it improves sleep; I don’t think that’s really been shown to be the case. A lot of it has to do with other chemicals included in the CBD preparation,” he said.
Dr. Hill is the author of Marijuana: The Unbiased Truth about the World’s Most Popular Weed (Center City, Minn.: Hazelden Publishing, 2015). He had no industry disclosures.
For now, CBD is better option than whole-plant cannabis for psychiatric disorders
For now, CBD is better option than whole-plant cannabis for psychiatric disorders
SAN FRANCISCO – Outside of prescription products for chemotherapy nausea/vomiting, AIDS anorexia, and rare pediatric epilepsies, medical cannabis has the strongest evidence for chronic pain, neuropathic pain, and multiple sclerosis spasticity, according to Kevin P. Hill, MD, director of the division of addiction psychiatry at Beth Israel Deaconess Medical Center, Boston.
“We are talking about multiple, positive RCTs [randomized, controlled trials]. I think you can’t ignore that. For people who are staunchly opposed to medical cannabis, [it’s becoming] harder and harder to take that tack. I do think we need to come to an understanding that there is some evidence,” Dr. Hill said at the American Psychiatric Association annual meeting.
However, “there’s incredible interest in using cannabinoids for a whole host of [other] conditions for which the level of evidence isn’t where we’d like it to be. In many ways,” legalization has “far outpaced the evidence.
Perhaps that’s most true for psychiatric disorders. Although many patients swear by cannabis, what little evidence there is comes from observational studies, and those have mostly been disappointing.
Whole-plant cannabis, for instance, has been associated with an increased risk of depression in a dose-dependent fashion, and increased depression and anxiety symptoms, including panic attacks, among inexperienced users. Cutting back has been associated with symptom relief.
No placebo-controlled trials have been conducted to address cause and effect, so it’s unknown whether people use because they have worse disease or have worse disease because they use.
For now, “whole-plant cannabis to treat anxiety is probably not a good idea. We may ultimately find that cannabidiol” – CBD, the nonpsychoactive component of cannabis – “may have utility. If patients are already using, CBD is a better bet; it has no abuse potential,” Dr. Hill said.
He sometimes recommends CBD when patients fail traditional options, but over-the-counter or online products rather than the “exorbitantly expensive” Food and Drug Administration–approved version Epidiolex, indicated for the rare pediatric epilepsies Lennox-Gastaut and Dravet syndromes.
He asks patients to “tell me what you’re going to buy and we’ll go from there, and try to dose it.” Dr. Hill has confidence in only a few CBD brands to be accurately labeled, one of which is Charlotte’s Web.
There’s been a lot of excitement over cannabis for PTSD, but at this point, positive findings are mostly anecdotal, and use was associated with worse symptoms, increased violence, more alcohol and drug use, and worse therapy outcomes in a longitudinal study of 2,276 veterans. At least one ongoing RCT is underway that should address cause-and-effect (J Clin Psychiatry. 2015 Sep;76[9]:1174-80).
Like anxiety, CBD “would probably have more promise than whole-plant cannabis” for PTSD, Dr. Hill said.
That also might be the case for bipolar disorder. Whole-plant use is particularly common among patients, and it, again, seems to make symptoms worse.
The data for insomnia are much like that seen with alcohol: quicker asleep but worse sleep quality. “One of the myths about CBD is that it improves sleep; I don’t think that’s really been shown to be the case. A lot of it has to do with other chemicals included in the CBD preparation,” he said.
Dr. Hill is the author of Marijuana: The Unbiased Truth about the World’s Most Popular Weed (Center City, Minn.: Hazelden Publishing, 2015). He had no industry disclosures.
SAN FRANCISCO – Outside of prescription products for chemotherapy nausea/vomiting, AIDS anorexia, and rare pediatric epilepsies, medical cannabis has the strongest evidence for chronic pain, neuropathic pain, and multiple sclerosis spasticity, according to Kevin P. Hill, MD, director of the division of addiction psychiatry at Beth Israel Deaconess Medical Center, Boston.
“We are talking about multiple, positive RCTs [randomized, controlled trials]. I think you can’t ignore that. For people who are staunchly opposed to medical cannabis, [it’s becoming] harder and harder to take that tack. I do think we need to come to an understanding that there is some evidence,” Dr. Hill said at the American Psychiatric Association annual meeting.
However, “there’s incredible interest in using cannabinoids for a whole host of [other] conditions for which the level of evidence isn’t where we’d like it to be. In many ways,” legalization has “far outpaced the evidence.
Perhaps that’s most true for psychiatric disorders. Although many patients swear by cannabis, what little evidence there is comes from observational studies, and those have mostly been disappointing.
Whole-plant cannabis, for instance, has been associated with an increased risk of depression in a dose-dependent fashion, and increased depression and anxiety symptoms, including panic attacks, among inexperienced users. Cutting back has been associated with symptom relief.
No placebo-controlled trials have been conducted to address cause and effect, so it’s unknown whether people use because they have worse disease or have worse disease because they use.
For now, “whole-plant cannabis to treat anxiety is probably not a good idea. We may ultimately find that cannabidiol” – CBD, the nonpsychoactive component of cannabis – “may have utility. If patients are already using, CBD is a better bet; it has no abuse potential,” Dr. Hill said.
He sometimes recommends CBD when patients fail traditional options, but over-the-counter or online products rather than the “exorbitantly expensive” Food and Drug Administration–approved version Epidiolex, indicated for the rare pediatric epilepsies Lennox-Gastaut and Dravet syndromes.
He asks patients to “tell me what you’re going to buy and we’ll go from there, and try to dose it.” Dr. Hill has confidence in only a few CBD brands to be accurately labeled, one of which is Charlotte’s Web.
There’s been a lot of excitement over cannabis for PTSD, but at this point, positive findings are mostly anecdotal, and use was associated with worse symptoms, increased violence, more alcohol and drug use, and worse therapy outcomes in a longitudinal study of 2,276 veterans. At least one ongoing RCT is underway that should address cause-and-effect (J Clin Psychiatry. 2015 Sep;76[9]:1174-80).
Like anxiety, CBD “would probably have more promise than whole-plant cannabis” for PTSD, Dr. Hill said.
That also might be the case for bipolar disorder. Whole-plant use is particularly common among patients, and it, again, seems to make symptoms worse.
The data for insomnia are much like that seen with alcohol: quicker asleep but worse sleep quality. “One of the myths about CBD is that it improves sleep; I don’t think that’s really been shown to be the case. A lot of it has to do with other chemicals included in the CBD preparation,” he said.
Dr. Hill is the author of Marijuana: The Unbiased Truth about the World’s Most Popular Weed (Center City, Minn.: Hazelden Publishing, 2015). He had no industry disclosures.
REPORTING FROM APA 2019
Opioid use curbed with patient education and lower prescription quantities
Patients given lower prescription quantities of opioid tablets with and without opioid education used significantly less of the medication compared with those given more tablets and no education, according to data from 264 adults and adolescents who underwent anterior cruciate ligament (ACL) surgery.
Although lower default prescription programs have been shown to reduce the number of tablets prescribed, “the effect of reduced prescription quantities on actual patient opioid consumption remains undetermined,” wrote Kevin X. Farley, BS, of Emory University, Atlanta, and colleagues.
In a study published in JAMA, the researchers examined whether patients took fewer tablets if given fewer, and whether patient education about opioids further reduced the number of tablets taken.
The study population included adults and adolescents who underwent ACL surgery at a single center. The patients were divided into three groups: 109 patients received 50 opioid tablets after surgery, 78 received 30 tablets plus education prior to surgery about appropriate opioid use and alternative pain management, and 77 received 30 tablets but no education on opioid use.
Patients given 50 tablets consumed an average of 25 tablets for an average of 5.8 days. By contrast, patients given 30 tablets but no opioid education consumed an average of 16 tablets for an average of 4.5 days, and those given 30 tablets and preoperative education consumed an average of 12 tablets for an average of 3.5 days.
In addition, patients given 30 tablets reported lower levels of constipation and fatigue compared with patients given 50 tablets. No differences were seen in medication refills among the groups.
The findings were limited by several factors including the use of data from a single center, the lack of randomization, and the potential for recall bias, the researchers noted. However, the results suggest that prescribing fewer tablets may further reduce use, as each group consumed approximately half of the tablets given, the researchers added.
“Further investigation should evaluate whether similar opioid stewardship and education protocols would be successful in other patient populations,” they said.
Corresponding author John Xerogeanes, MD, disclosed personal fees from Arthrex and stock options from Trice. The other researchers had no financial conflicts to disclose.
Patients given lower prescription quantities of opioid tablets with and without opioid education used significantly less of the medication compared with those given more tablets and no education, according to data from 264 adults and adolescents who underwent anterior cruciate ligament (ACL) surgery.
Although lower default prescription programs have been shown to reduce the number of tablets prescribed, “the effect of reduced prescription quantities on actual patient opioid consumption remains undetermined,” wrote Kevin X. Farley, BS, of Emory University, Atlanta, and colleagues.
In a study published in JAMA, the researchers examined whether patients took fewer tablets if given fewer, and whether patient education about opioids further reduced the number of tablets taken.
The study population included adults and adolescents who underwent ACL surgery at a single center. The patients were divided into three groups: 109 patients received 50 opioid tablets after surgery, 78 received 30 tablets plus education prior to surgery about appropriate opioid use and alternative pain management, and 77 received 30 tablets but no education on opioid use.
Patients given 50 tablets consumed an average of 25 tablets for an average of 5.8 days. By contrast, patients given 30 tablets but no opioid education consumed an average of 16 tablets for an average of 4.5 days, and those given 30 tablets and preoperative education consumed an average of 12 tablets for an average of 3.5 days.
In addition, patients given 30 tablets reported lower levels of constipation and fatigue compared with patients given 50 tablets. No differences were seen in medication refills among the groups.
The findings were limited by several factors including the use of data from a single center, the lack of randomization, and the potential for recall bias, the researchers noted. However, the results suggest that prescribing fewer tablets may further reduce use, as each group consumed approximately half of the tablets given, the researchers added.
“Further investigation should evaluate whether similar opioid stewardship and education protocols would be successful in other patient populations,” they said.
Corresponding author John Xerogeanes, MD, disclosed personal fees from Arthrex and stock options from Trice. The other researchers had no financial conflicts to disclose.
Patients given lower prescription quantities of opioid tablets with and without opioid education used significantly less of the medication compared with those given more tablets and no education, according to data from 264 adults and adolescents who underwent anterior cruciate ligament (ACL) surgery.
Although lower default prescription programs have been shown to reduce the number of tablets prescribed, “the effect of reduced prescription quantities on actual patient opioid consumption remains undetermined,” wrote Kevin X. Farley, BS, of Emory University, Atlanta, and colleagues.
In a study published in JAMA, the researchers examined whether patients took fewer tablets if given fewer, and whether patient education about opioids further reduced the number of tablets taken.
The study population included adults and adolescents who underwent ACL surgery at a single center. The patients were divided into three groups: 109 patients received 50 opioid tablets after surgery, 78 received 30 tablets plus education prior to surgery about appropriate opioid use and alternative pain management, and 77 received 30 tablets but no education on opioid use.
Patients given 50 tablets consumed an average of 25 tablets for an average of 5.8 days. By contrast, patients given 30 tablets but no opioid education consumed an average of 16 tablets for an average of 4.5 days, and those given 30 tablets and preoperative education consumed an average of 12 tablets for an average of 3.5 days.
In addition, patients given 30 tablets reported lower levels of constipation and fatigue compared with patients given 50 tablets. No differences were seen in medication refills among the groups.
The findings were limited by several factors including the use of data from a single center, the lack of randomization, and the potential for recall bias, the researchers noted. However, the results suggest that prescribing fewer tablets may further reduce use, as each group consumed approximately half of the tablets given, the researchers added.
“Further investigation should evaluate whether similar opioid stewardship and education protocols would be successful in other patient populations,” they said.
Corresponding author John Xerogeanes, MD, disclosed personal fees from Arthrex and stock options from Trice. The other researchers had no financial conflicts to disclose.
FROM JAMA
Key clinical point: Patient education and fewer tablets prescribed significantly reduced the amount of opioid tablets taken compared with no education and more tablets prescribed.
Major finding: Patients given 50 tablets and no patient education, 30 tablets and no patient education, and 30 tablets plus education consumed an average of 25, 16, and 12 tablets, respectively.
Study details: The data come from 264 adolescents and adults who underwent ACL surgery at a single center.
Disclosures: Corresponding author John Xerogeanes, MD, disclosed personal fees from Arthrex and stock options from Trice. The other researchers had no financial conflicts to disclose.
Source: Farley KX et al. JAMA. 2019 June 25.321(24):2465-7.
Sexual assault in military linked to sexual pain
according to an observational study involving interviews with more than 1,000 military women.
Female veterans with histories of both childhood sexual abuse and sexual assault in the military were 4.33 times more likely to report sexual pain than female veterans with no history of sexual assault; women whose history of sexual assault occurred in the military only were 2.37 times more likely to report sexual pain. Those with histories of childhood sexual abuse but no military assaults were 1.75 times more likely to report sexual pain than those who had no history of sexual assault.
The findings suggest that sexual assault in the military is more detrimental to sexual function than childhood sexual abuse alone, which “is distinct from the pattern long observed in civilian women that childhood sexual abuse confers a greater risk for sexual pain than adulthood sexual assault,” Carey S. Pulverman, PhD, then of the Department of Veterans Affairs Center of Excellence for Research on Returning War Veterans in Waco, Tex., and coinvestigators wrote in Obstetrics & Gynecology.
The findings come from a secondary analysis of data collected for a larger project titled Sexual Violence and Women Veterans’ Gynecologic Health . The research team conducted telephone interviews with 1,004 female veterans younger than 52 years of age (mean, 38 years) who were enrolled at two large Midwestern VA medical centers and associated clinics. Sexual pain was assessed by one question: “Does it hurt you to have sexual intercourse or penetration?”
The study also identified high comorbidity between sexual pain and mental health concerns. As with sexual pain, rates of depression and PTSD were highest among female veterans with histories of both sexual abuse in childhood and sexual assault in the military, followed by women with histories of sexual assaults in the military alone, and then women with histories of childhood sexual abuse alone. Women with both histories were 6.35 times more likely to report PTSD, and 3.91 times more likely to report depression, compared with female veterans with no history of sexual assault.
Women who experienced sexual assault during their childhood and/or while serving in the military also may have been exposed to sexual assault during their pre- or postmilitary adulthood as well, but this was a small number and its effects were not evaluated, the authors noted.
Especially given the “growing numbers of women serving in the military and prevalence of sexual assault in this population,” there’s a need for more research on the sexual function of female veterans and development of “targeted treatments,” the investigators wrote.
For now, providers should be “more comprehensive in their assessment of sexual assault history” and should consider developing relationships with community providers who specialize in sexual health, they added.
The study was funded by the VA. The authors did not report any relevant financial disclosures.
SOURCE: Pulverman CS et al. Obstet Gynecol. 2019;134:63-71.
according to an observational study involving interviews with more than 1,000 military women.
Female veterans with histories of both childhood sexual abuse and sexual assault in the military were 4.33 times more likely to report sexual pain than female veterans with no history of sexual assault; women whose history of sexual assault occurred in the military only were 2.37 times more likely to report sexual pain. Those with histories of childhood sexual abuse but no military assaults were 1.75 times more likely to report sexual pain than those who had no history of sexual assault.
The findings suggest that sexual assault in the military is more detrimental to sexual function than childhood sexual abuse alone, which “is distinct from the pattern long observed in civilian women that childhood sexual abuse confers a greater risk for sexual pain than adulthood sexual assault,” Carey S. Pulverman, PhD, then of the Department of Veterans Affairs Center of Excellence for Research on Returning War Veterans in Waco, Tex., and coinvestigators wrote in Obstetrics & Gynecology.
The findings come from a secondary analysis of data collected for a larger project titled Sexual Violence and Women Veterans’ Gynecologic Health . The research team conducted telephone interviews with 1,004 female veterans younger than 52 years of age (mean, 38 years) who were enrolled at two large Midwestern VA medical centers and associated clinics. Sexual pain was assessed by one question: “Does it hurt you to have sexual intercourse or penetration?”
The study also identified high comorbidity between sexual pain and mental health concerns. As with sexual pain, rates of depression and PTSD were highest among female veterans with histories of both sexual abuse in childhood and sexual assault in the military, followed by women with histories of sexual assaults in the military alone, and then women with histories of childhood sexual abuse alone. Women with both histories were 6.35 times more likely to report PTSD, and 3.91 times more likely to report depression, compared with female veterans with no history of sexual assault.
Women who experienced sexual assault during their childhood and/or while serving in the military also may have been exposed to sexual assault during their pre- or postmilitary adulthood as well, but this was a small number and its effects were not evaluated, the authors noted.
Especially given the “growing numbers of women serving in the military and prevalence of sexual assault in this population,” there’s a need for more research on the sexual function of female veterans and development of “targeted treatments,” the investigators wrote.
For now, providers should be “more comprehensive in their assessment of sexual assault history” and should consider developing relationships with community providers who specialize in sexual health, they added.
The study was funded by the VA. The authors did not report any relevant financial disclosures.
SOURCE: Pulverman CS et al. Obstet Gynecol. 2019;134:63-71.
according to an observational study involving interviews with more than 1,000 military women.
Female veterans with histories of both childhood sexual abuse and sexual assault in the military were 4.33 times more likely to report sexual pain than female veterans with no history of sexual assault; women whose history of sexual assault occurred in the military only were 2.37 times more likely to report sexual pain. Those with histories of childhood sexual abuse but no military assaults were 1.75 times more likely to report sexual pain than those who had no history of sexual assault.
The findings suggest that sexual assault in the military is more detrimental to sexual function than childhood sexual abuse alone, which “is distinct from the pattern long observed in civilian women that childhood sexual abuse confers a greater risk for sexual pain than adulthood sexual assault,” Carey S. Pulverman, PhD, then of the Department of Veterans Affairs Center of Excellence for Research on Returning War Veterans in Waco, Tex., and coinvestigators wrote in Obstetrics & Gynecology.
The findings come from a secondary analysis of data collected for a larger project titled Sexual Violence and Women Veterans’ Gynecologic Health . The research team conducted telephone interviews with 1,004 female veterans younger than 52 years of age (mean, 38 years) who were enrolled at two large Midwestern VA medical centers and associated clinics. Sexual pain was assessed by one question: “Does it hurt you to have sexual intercourse or penetration?”
The study also identified high comorbidity between sexual pain and mental health concerns. As with sexual pain, rates of depression and PTSD were highest among female veterans with histories of both sexual abuse in childhood and sexual assault in the military, followed by women with histories of sexual assaults in the military alone, and then women with histories of childhood sexual abuse alone. Women with both histories were 6.35 times more likely to report PTSD, and 3.91 times more likely to report depression, compared with female veterans with no history of sexual assault.
Women who experienced sexual assault during their childhood and/or while serving in the military also may have been exposed to sexual assault during their pre- or postmilitary adulthood as well, but this was a small number and its effects were not evaluated, the authors noted.
Especially given the “growing numbers of women serving in the military and prevalence of sexual assault in this population,” there’s a need for more research on the sexual function of female veterans and development of “targeted treatments,” the investigators wrote.
For now, providers should be “more comprehensive in their assessment of sexual assault history” and should consider developing relationships with community providers who specialize in sexual health, they added.
The study was funded by the VA. The authors did not report any relevant financial disclosures.
SOURCE: Pulverman CS et al. Obstet Gynecol. 2019;134:63-71.
FROM OBSTETRICS & GYNECOLOGY
Medical cannabis laws appear no longer tied to drop in opioid overdose mortality
Correlations do not hold when analysis is expanded to 2017
Contrary to previous research indicating that medical cannabis laws reduced opioid overdose mortality, the association between these two has reversed, with opioid overdose mortality increased in states with comprehensive medical cannabis laws, according to Chelsea L. Shover, PhD, and associates.
The original research by Marcus A. Bachhuber, MD, and associates showed that the introduction of state medical cannabis laws was associated with a 24.8% reduction in opioid overdose deaths per 100,000 population between 1999 and 2010. In contrast, the new research – which looked at a longer time period than the original research did – found that the association between state medical cannabis laws and opioid overdose mortality reversed direction, from –21% to +23%.
“We find it unlikely that medical cannabis – used by about 2.5% of the U.S. population – has exerted large conflicting effects on opioid overdose mortality,” wrote Dr. Shover, of the department of psychiatry and behavioral sciences at Stanford (Calif.) University, and associates. “A more plausible interpretation is that this association is spurious.” Their study was published in the Proceedings of the National Academy of Sciences.
To conduct their analysis, Dr. Shover and associates extended the timeline reviewed by Dr. Bachhuber and associates to 2017. During 2010-2017, 32 states enacted medical cannabis laws, including 17 allowing only medical cannabis with low levels of tetrahydrocannabinol (THC), and 8 legalized recreational marijuana. In the expanded timeline during 1999-2017, states possessing a comprehensive medical marijuana law saw an increase in opioid overdose mortality of 28.2%. Meanwhile, states with recreational marijuana laws saw a decrease of 14.7% in opioid overdose mortality, and states with low-THC medical cannabis laws saw a decrease of 7.1%. However, the investigators noted that those values had wide confidence intervals, which indicates “compatibility with large range of true associations.”
Corporate actors with deep pockets have substantial ability to promote congenial results, and suffering people are desperate for effective solutions. Cannabinoids have demonstrated therapeutic benefits, but reducing population-level opioid overdose mortality does not appear to be among them,” Dr. Shover and associates noted.
Dr. Shover reported receiving support from National Institute on Drug Abuse and the Wu Tsai Neurosciences Institute. Another coauthor received support from the Veterans Health Administration, Wu Tsai Neurosciences Institute, and the Esther Ting Memorial Professorship at Stanford.
SOURCE: Shover CL et al. Proc Natl Acad Sci U S A. 2019 Jun 10. doi: 10.1073/pnas.1903434116.
Correlations do not hold when analysis is expanded to 2017
Correlations do not hold when analysis is expanded to 2017
Contrary to previous research indicating that medical cannabis laws reduced opioid overdose mortality, the association between these two has reversed, with opioid overdose mortality increased in states with comprehensive medical cannabis laws, according to Chelsea L. Shover, PhD, and associates.
The original research by Marcus A. Bachhuber, MD, and associates showed that the introduction of state medical cannabis laws was associated with a 24.8% reduction in opioid overdose deaths per 100,000 population between 1999 and 2010. In contrast, the new research – which looked at a longer time period than the original research did – found that the association between state medical cannabis laws and opioid overdose mortality reversed direction, from –21% to +23%.
“We find it unlikely that medical cannabis – used by about 2.5% of the U.S. population – has exerted large conflicting effects on opioid overdose mortality,” wrote Dr. Shover, of the department of psychiatry and behavioral sciences at Stanford (Calif.) University, and associates. “A more plausible interpretation is that this association is spurious.” Their study was published in the Proceedings of the National Academy of Sciences.
To conduct their analysis, Dr. Shover and associates extended the timeline reviewed by Dr. Bachhuber and associates to 2017. During 2010-2017, 32 states enacted medical cannabis laws, including 17 allowing only medical cannabis with low levels of tetrahydrocannabinol (THC), and 8 legalized recreational marijuana. In the expanded timeline during 1999-2017, states possessing a comprehensive medical marijuana law saw an increase in opioid overdose mortality of 28.2%. Meanwhile, states with recreational marijuana laws saw a decrease of 14.7% in opioid overdose mortality, and states with low-THC medical cannabis laws saw a decrease of 7.1%. However, the investigators noted that those values had wide confidence intervals, which indicates “compatibility with large range of true associations.”
Corporate actors with deep pockets have substantial ability to promote congenial results, and suffering people are desperate for effective solutions. Cannabinoids have demonstrated therapeutic benefits, but reducing population-level opioid overdose mortality does not appear to be among them,” Dr. Shover and associates noted.
Dr. Shover reported receiving support from National Institute on Drug Abuse and the Wu Tsai Neurosciences Institute. Another coauthor received support from the Veterans Health Administration, Wu Tsai Neurosciences Institute, and the Esther Ting Memorial Professorship at Stanford.
SOURCE: Shover CL et al. Proc Natl Acad Sci U S A. 2019 Jun 10. doi: 10.1073/pnas.1903434116.
Contrary to previous research indicating that medical cannabis laws reduced opioid overdose mortality, the association between these two has reversed, with opioid overdose mortality increased in states with comprehensive medical cannabis laws, according to Chelsea L. Shover, PhD, and associates.
The original research by Marcus A. Bachhuber, MD, and associates showed that the introduction of state medical cannabis laws was associated with a 24.8% reduction in opioid overdose deaths per 100,000 population between 1999 and 2010. In contrast, the new research – which looked at a longer time period than the original research did – found that the association between state medical cannabis laws and opioid overdose mortality reversed direction, from –21% to +23%.
“We find it unlikely that medical cannabis – used by about 2.5% of the U.S. population – has exerted large conflicting effects on opioid overdose mortality,” wrote Dr. Shover, of the department of psychiatry and behavioral sciences at Stanford (Calif.) University, and associates. “A more plausible interpretation is that this association is spurious.” Their study was published in the Proceedings of the National Academy of Sciences.
To conduct their analysis, Dr. Shover and associates extended the timeline reviewed by Dr. Bachhuber and associates to 2017. During 2010-2017, 32 states enacted medical cannabis laws, including 17 allowing only medical cannabis with low levels of tetrahydrocannabinol (THC), and 8 legalized recreational marijuana. In the expanded timeline during 1999-2017, states possessing a comprehensive medical marijuana law saw an increase in opioid overdose mortality of 28.2%. Meanwhile, states with recreational marijuana laws saw a decrease of 14.7% in opioid overdose mortality, and states with low-THC medical cannabis laws saw a decrease of 7.1%. However, the investigators noted that those values had wide confidence intervals, which indicates “compatibility with large range of true associations.”
Corporate actors with deep pockets have substantial ability to promote congenial results, and suffering people are desperate for effective solutions. Cannabinoids have demonstrated therapeutic benefits, but reducing population-level opioid overdose mortality does not appear to be among them,” Dr. Shover and associates noted.
Dr. Shover reported receiving support from National Institute on Drug Abuse and the Wu Tsai Neurosciences Institute. Another coauthor received support from the Veterans Health Administration, Wu Tsai Neurosciences Institute, and the Esther Ting Memorial Professorship at Stanford.
SOURCE: Shover CL et al. Proc Natl Acad Sci U S A. 2019 Jun 10. doi: 10.1073/pnas.1903434116.
FROM PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES
Mindfulness-based stress reduction reduces migraine frequency
SAN ANTONIO – Episodic migraine patients benefit from mindfulness-based stress reduction training, according to new research. The intervention reduced headache frequency, slightly increased whole-brain gray matter volume, and reduced symptoms of anxiety, depression, and stress.
The gray matter findings may indicate opportunities for therapeutic targets, while the psychosocial findings are important in understanding migraine burden, treatment response, and personalized medicine opportunities, Shana Burrowes, PhD, a postdoctoral associate at Boston University, said at the annual meeting of the College on Problems of Drug Dependence.
In a session focused on exploring alternatives to opioids for pain treatment, Dr. Burrowes described interim results of a randomized, controlled trial testing the effectiveness of mindfulness-based stress reduction (MBSR) training for managing migraine.
In discussing the rationale for study endpoints, she explained a three-pronged model for understanding migraine. Those elements include the symptoms themselves – unilateral throbbing pain, nausea, and photophobia – and the psychosocial symptoms and comorbidities, including anxiety, depression, stress, and catastrophizing. Up to 30%* of migraine patients have comorbid depression.
Those two prongs have a bidirectional relationship, since each increases the risk of the other. For example, frequent migraine can leave people feeling anxious about when their next migraine will occur, and that anxiety can increase the risk of it occurring.
Both elements lead to the third prong, which is change in gray matter volume. “If you’re a patient with migraine, an MRI on your brain is going to look different from somebody who does not have migraine,” Dr. Burrowes said. “With all these things going on in a patient, a migraine patient is actually pretty difficult to treat.”
Therefore, the researchers focused on outcomes from each of these three domains: gray matter volume in MRI; headache frequency as a clinical outcome; and the psychosocial comorbidities of anxiety, stress, and depression.
Study participants included 98 patients with episodic migraine, defined as fewer than 15 headache days a month, and 27 controls* matched by demographics to the patients and without any chronic pain conditions. The groups were 92% female and had similar ratios of whites (75% and 77%) and college graduates (95% and 96%).
Only the patients were randomized to the two interventions, one a training on MBSR and the other focusing on stress management for headache (SMH).
The MBSR training involved group sessions, eight 2.5-hour meditation sessions, at-home practice, a half-day retreat, and then an additional four biweekly sessions. The mindfulness training specifically focused on intentionally paying attention in the moment without judgment. The SMH arm focused on education for managing headache symptoms, stress, sleep hygiene, and diet, but it did not involve any specific skills training, such as relaxation training.
All participants, including healthy controls, underwent clinical assessment and baseline MRI and psychosocial questionnaires, followed by MRI and psychosocial questionnaire follow-ups at 3 and 6 months. MRI imaging focused on the whole brain and on the bilateral insula, dorsolateral prefrontal cortex, anterior cingulate cortex, and superior frontal gyrus. Patients also kept headache diaries throughout the trial.
Both intervention groups showed an increase in gray matter volume over 6 months, compared with healthy controls: 1.3% in the whole brain for SMH participants and 1.01% in the MBSR patients, compared with –1.37% in healthy participants. In the right superior frontal gyrus, gray matter volume also increased 2.62% in SMH participants and 1.25% in MBSR patients but decreased 0.19% in healthy participants.
Dr. Burrowes said she could not share specific findings on headache frequency and psychosocial outcomes because her team’s research is currently under review. Overall, however, headache frequency declined more than 50% post intervention, and 39% of migraine patients responded to the therapy.
In addition, anxiety, stress, and depression symptoms all saw improvements from MBSR and slightly but significantly mediated the effect of MBSR on migraine reduction.
Dr. Burrowes reported having no disclosures.
*The story was updated 6/20/2019.
SAN ANTONIO – Episodic migraine patients benefit from mindfulness-based stress reduction training, according to new research. The intervention reduced headache frequency, slightly increased whole-brain gray matter volume, and reduced symptoms of anxiety, depression, and stress.
The gray matter findings may indicate opportunities for therapeutic targets, while the psychosocial findings are important in understanding migraine burden, treatment response, and personalized medicine opportunities, Shana Burrowes, PhD, a postdoctoral associate at Boston University, said at the annual meeting of the College on Problems of Drug Dependence.
In a session focused on exploring alternatives to opioids for pain treatment, Dr. Burrowes described interim results of a randomized, controlled trial testing the effectiveness of mindfulness-based stress reduction (MBSR) training for managing migraine.
In discussing the rationale for study endpoints, she explained a three-pronged model for understanding migraine. Those elements include the symptoms themselves – unilateral throbbing pain, nausea, and photophobia – and the psychosocial symptoms and comorbidities, including anxiety, depression, stress, and catastrophizing. Up to 30%* of migraine patients have comorbid depression.
Those two prongs have a bidirectional relationship, since each increases the risk of the other. For example, frequent migraine can leave people feeling anxious about when their next migraine will occur, and that anxiety can increase the risk of it occurring.
Both elements lead to the third prong, which is change in gray matter volume. “If you’re a patient with migraine, an MRI on your brain is going to look different from somebody who does not have migraine,” Dr. Burrowes said. “With all these things going on in a patient, a migraine patient is actually pretty difficult to treat.”
Therefore, the researchers focused on outcomes from each of these three domains: gray matter volume in MRI; headache frequency as a clinical outcome; and the psychosocial comorbidities of anxiety, stress, and depression.
Study participants included 98 patients with episodic migraine, defined as fewer than 15 headache days a month, and 27 controls* matched by demographics to the patients and without any chronic pain conditions. The groups were 92% female and had similar ratios of whites (75% and 77%) and college graduates (95% and 96%).
Only the patients were randomized to the two interventions, one a training on MBSR and the other focusing on stress management for headache (SMH).
The MBSR training involved group sessions, eight 2.5-hour meditation sessions, at-home practice, a half-day retreat, and then an additional four biweekly sessions. The mindfulness training specifically focused on intentionally paying attention in the moment without judgment. The SMH arm focused on education for managing headache symptoms, stress, sleep hygiene, and diet, but it did not involve any specific skills training, such as relaxation training.
All participants, including healthy controls, underwent clinical assessment and baseline MRI and psychosocial questionnaires, followed by MRI and psychosocial questionnaire follow-ups at 3 and 6 months. MRI imaging focused on the whole brain and on the bilateral insula, dorsolateral prefrontal cortex, anterior cingulate cortex, and superior frontal gyrus. Patients also kept headache diaries throughout the trial.
Both intervention groups showed an increase in gray matter volume over 6 months, compared with healthy controls: 1.3% in the whole brain for SMH participants and 1.01% in the MBSR patients, compared with –1.37% in healthy participants. In the right superior frontal gyrus, gray matter volume also increased 2.62% in SMH participants and 1.25% in MBSR patients but decreased 0.19% in healthy participants.
Dr. Burrowes said she could not share specific findings on headache frequency and psychosocial outcomes because her team’s research is currently under review. Overall, however, headache frequency declined more than 50% post intervention, and 39% of migraine patients responded to the therapy.
In addition, anxiety, stress, and depression symptoms all saw improvements from MBSR and slightly but significantly mediated the effect of MBSR on migraine reduction.
Dr. Burrowes reported having no disclosures.
*The story was updated 6/20/2019.
SAN ANTONIO – Episodic migraine patients benefit from mindfulness-based stress reduction training, according to new research. The intervention reduced headache frequency, slightly increased whole-brain gray matter volume, and reduced symptoms of anxiety, depression, and stress.
The gray matter findings may indicate opportunities for therapeutic targets, while the psychosocial findings are important in understanding migraine burden, treatment response, and personalized medicine opportunities, Shana Burrowes, PhD, a postdoctoral associate at Boston University, said at the annual meeting of the College on Problems of Drug Dependence.
In a session focused on exploring alternatives to opioids for pain treatment, Dr. Burrowes described interim results of a randomized, controlled trial testing the effectiveness of mindfulness-based stress reduction (MBSR) training for managing migraine.
In discussing the rationale for study endpoints, she explained a three-pronged model for understanding migraine. Those elements include the symptoms themselves – unilateral throbbing pain, nausea, and photophobia – and the psychosocial symptoms and comorbidities, including anxiety, depression, stress, and catastrophizing. Up to 30%* of migraine patients have comorbid depression.
Those two prongs have a bidirectional relationship, since each increases the risk of the other. For example, frequent migraine can leave people feeling anxious about when their next migraine will occur, and that anxiety can increase the risk of it occurring.
Both elements lead to the third prong, which is change in gray matter volume. “If you’re a patient with migraine, an MRI on your brain is going to look different from somebody who does not have migraine,” Dr. Burrowes said. “With all these things going on in a patient, a migraine patient is actually pretty difficult to treat.”
Therefore, the researchers focused on outcomes from each of these three domains: gray matter volume in MRI; headache frequency as a clinical outcome; and the psychosocial comorbidities of anxiety, stress, and depression.
Study participants included 98 patients with episodic migraine, defined as fewer than 15 headache days a month, and 27 controls* matched by demographics to the patients and without any chronic pain conditions. The groups were 92% female and had similar ratios of whites (75% and 77%) and college graduates (95% and 96%).
Only the patients were randomized to the two interventions, one a training on MBSR and the other focusing on stress management for headache (SMH).
The MBSR training involved group sessions, eight 2.5-hour meditation sessions, at-home practice, a half-day retreat, and then an additional four biweekly sessions. The mindfulness training specifically focused on intentionally paying attention in the moment without judgment. The SMH arm focused on education for managing headache symptoms, stress, sleep hygiene, and diet, but it did not involve any specific skills training, such as relaxation training.
All participants, including healthy controls, underwent clinical assessment and baseline MRI and psychosocial questionnaires, followed by MRI and psychosocial questionnaire follow-ups at 3 and 6 months. MRI imaging focused on the whole brain and on the bilateral insula, dorsolateral prefrontal cortex, anterior cingulate cortex, and superior frontal gyrus. Patients also kept headache diaries throughout the trial.
Both intervention groups showed an increase in gray matter volume over 6 months, compared with healthy controls: 1.3% in the whole brain for SMH participants and 1.01% in the MBSR patients, compared with –1.37% in healthy participants. In the right superior frontal gyrus, gray matter volume also increased 2.62% in SMH participants and 1.25% in MBSR patients but decreased 0.19% in healthy participants.
Dr. Burrowes said she could not share specific findings on headache frequency and psychosocial outcomes because her team’s research is currently under review. Overall, however, headache frequency declined more than 50% post intervention, and 39% of migraine patients responded to the therapy.
In addition, anxiety, stress, and depression symptoms all saw improvements from MBSR and slightly but significantly mediated the effect of MBSR on migraine reduction.
Dr. Burrowes reported having no disclosures.
*The story was updated 6/20/2019.
REPORTING FROM CPDD 2019
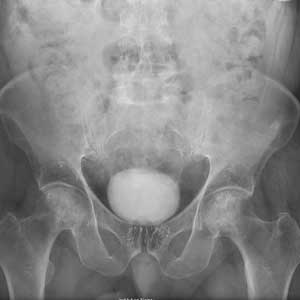
These Hips Don’t Lie
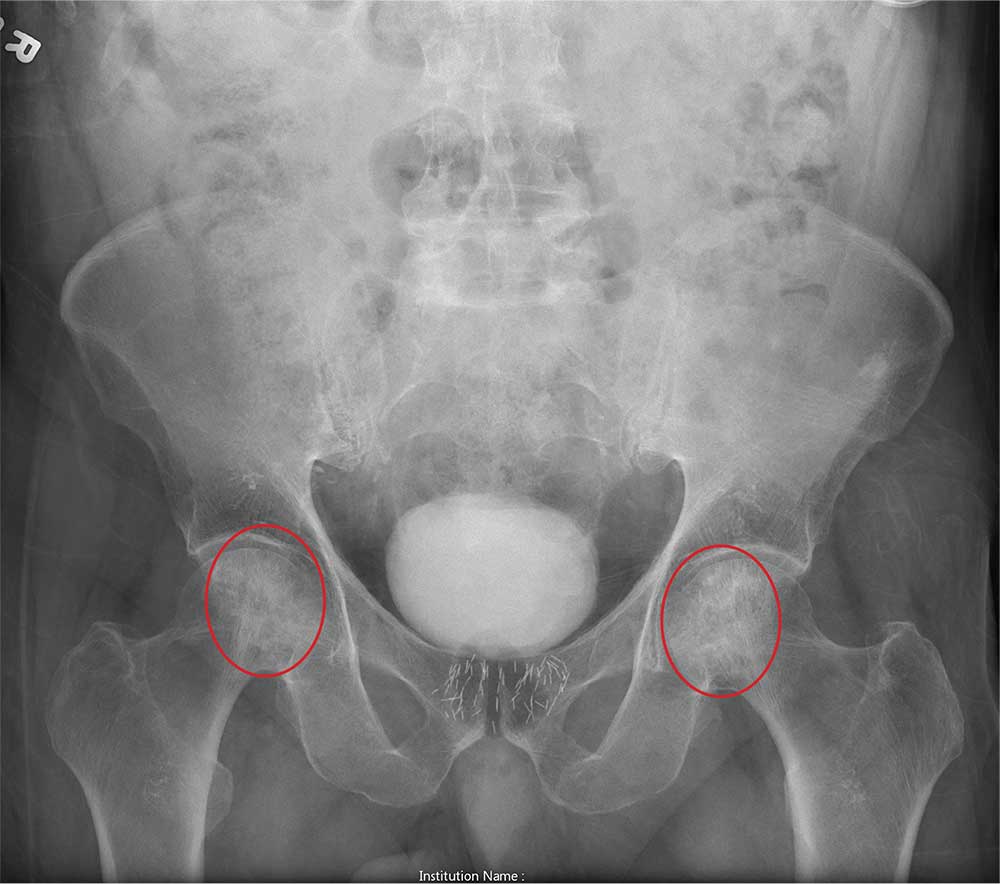
ANSWER
The radiograph shows no evidence of an acute fracture. Incidental findings include excreted contrast within the bladder and radiopaque markers from prostatic seed implants.
Fairly extensive sclerosis is noted within both femoral heads, which is suggestive of osteonecrosis (also known as avascular necrosis). Orthopedic consult was requested for further workup of this specific problem.

ANSWER
The radiograph shows no evidence of an acute fracture. Incidental findings include excreted contrast within the bladder and radiopaque markers from prostatic seed implants.
Fairly extensive sclerosis is noted within both femoral heads, which is suggestive of osteonecrosis (also known as avascular necrosis). Orthopedic consult was requested for further workup of this specific problem.

ANSWER
The radiograph shows no evidence of an acute fracture. Incidental findings include excreted contrast within the bladder and radiopaque markers from prostatic seed implants.
Fairly extensive sclerosis is noted within both femoral heads, which is suggestive of osteonecrosis (also known as avascular necrosis). Orthopedic consult was requested for further workup of this specific problem.
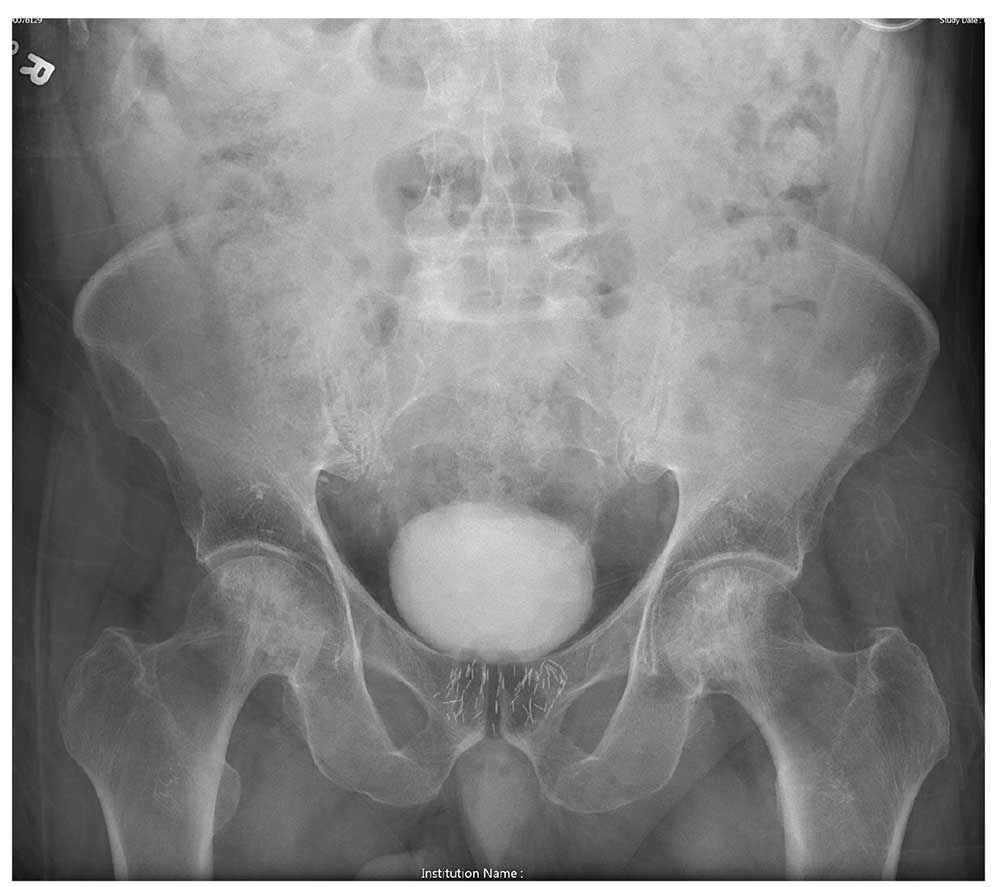
An 80-year-old man is transferred to your facility for evaluation of a lumbar compression fracture he sustained from a motor vehicle collision. The patient was a restrained driver in a vehicle that was broadsided at an unknown speed. His airbags deployed. In addition to mild back discomfort, he complains of severe right hip pain.
His medical history is significant for prostate cancer and coronary artery disease. Surgical history includes remote cardiac bypass surgery and recent revascularization with stents.
On examination, you note an elderly male who is awake and alert. His vital signs are stable. He is able to move all extremities
A portable pelvis radiograph is obtained (shown). What is your impression?
Gabapentinoids associated with suicidal behavior, overdose
Young patients might be at increased risk of suicidal behavior, unintentional overdose, injuries, and traffic incidents during treatment periods with gabapentinoids, compared with periods without treatment with those medications, a cohort study of almost 200,000 people shows. Pregabalin is associated with higher hazards of those outcomes than is gabapentin, and the associations are strongest in patients aged 15-24 years, the researchers reported.
“If our findings are triangulated with other forms of evidence, ,” wrote Yasmina Molero, PhD, and associates. “Further restrictions for off-label prescription may need consideration.” The study was published in BMJ.
The use of gabapentinoids has risen in the United States (JAMA Intern Med. 2018;178[2]:292-4), and overdose deaths tied to gabapentin have led some states to explore reclassification of the drug as a controlled substance (Risk Manag Healthc Policy. 2018;11:109-16). In the United Kingdom, gabapentinoids are being reclassified as a class C controlled drug because of concerns about the risk of addiction, overdose, and safety, wrote Dr. Molero of the department of psychiatry at Warneford Hospital at the University of Oxford, England, and associates.
To study associations between gabapentinoids and adverse outcomes related to coordination, mental health, and criminality, Dr. Molero and her associates analyzed data from 191,973 people from the Swedish Prescribed Drug Register who collected prescriptions for pregabalin or gabapentin between 2006 and 2013. The researchers included patients aged 15 years and older in their analyses.
They examined suicidal behavior, unintentional overdoses, head or body injuries, road traffic incidents and offenses, and arrests for violent crime using the Swedish Patient Register and the National Crime Register. In addition, they defined suicidal behavior as emergency hospital visits attributable to self-injurious behavior or suicide attempt, or death by suicide. Unintentional overdoses were defined as emergency hospital visits or death attributable to poisoning by illicit drugs, medications, or biologic substances; accidental poisoning by noxious substances; or acute intoxications and overdoses by alcohol and illicit drugs, excluding intentional self-poisoning, wrote Dr. Molero, who is affiliated with the Karolinska Institute in Stockholm, and her associates.
Of the nearly 192,000 participants who collected prescriptions of gabapentinoids on at least two consecutive occasions, 120,664 received pregabalin, and 85,360 received gabapentin; 14,051 of the participants received both drugs. Fifty-nine percent were women, and most patients were aged 45 or older.
During the study period, 10,026 participants (5.2%) were treated for suicidal behavior or died from suicide, 17,144 participants (8.9%) experienced an unintentional overdose, and 12,070 participants (6.3%) had a road traffic incident or offense. In addition, 70,522 participants (36.7%) had head or body injuries, and 7,984 participants (4.1%) were arrested for a violent crime.
The study used a within-individual design that compared when a person was taking a gabapentinoid with when he or she was not. During treatment periods, participants were at increased risk of suicidal behavior or death from suicide (age-adjusted hazard ratio, 1.26), unintentional overdose (1.24), head or body injuries (1.22), and road traffic incidents or offenses (1.13). Associations with arrests for violent crime were not significant.
Gabapentinoid treatment was associated with increased hazards of suicidal behavior in people young than 55 years, but hazards were reduced or not significant in participants aged 55 years and older. Participants aged 15-24 years had the highest hazards of suicidal behavior (1.67).
In a sensitivity analysis, the researchers examined participants who had a diagnosis of comorbid epilepsy, psychiatric disorders, or musculoskeletal disorders before the start of gabapentinoid treatment. Among patients with comorbid epilepsy, gabapentinoids were not significantly associated with suicidal behavior and were associated with reduced hazards for all other outcomes.
“In comorbid psychiatric disorders, gabapentinoids were associated with lower risk for all outcomes,” the researchers said. Among patients with comorbid musculoskeletal disorders, gabapentinoids were associated with reductions in head or body injuries, traffic incidents, and arrests for violent crime.
Dr. Molero and her associates noted that they lacked information about alcohol and drug use, as well as treatment adherence and the conditions for which gabapentinoids had been prescribed. Furthermore, differences in prescription practices and outcome rates might affect the generalizability of the results to other countries.
The different results for pregabalin and gabapentin “could be due to their different pharmacodynamic and pharmacokinetic profiles; pregabalin has a higher potency, greater bioavailability, and quicker absorption than gabapentin. Pregabalin also has been associated with withdrawal symptoms following rapid discontinuation, which could be related to suicidal behavior,” Dr. Molero and colleagues said. “The reduced hazards in older people could reflect pharmacodynamic differences related to age, less concurrent use of alcohol or drugs, different indications for treatment, or reduced symptom severity of underlying conditions.”
The Wellcome Trust, Swedish Research Council, and Karolinska Institute supported the study. The authors had no disclosures relevant to the study. One author reported grants from Shire and Evolan and has served as a speaker for Shire.
SOURCE: Molero Y et al. BMJ. 2019 Jun 12. doi: 10.1136/bmj.l2147.
The findings by Molero et al. advance clinical knowledge about the drug class of gabapentinoids, wrote Derek K. Tracy, MB BCh. Though the study does not establish causality, it does rely on a solid, large dataset. The study shows the importance of uncoupling pregabalin and gabapentin. Both drugs are indeed gabapentinoids, but their use can lead to different outcomes, depending on the age of patients. For example, pregabalin – not gabapentin – appears tied to higher risks of harm. The demographic group that is most vulnerable is patients aged 15-24, the researchers found. Factors driving those age-related differences in risks tied to the drugs need to be understood.
Dr. Tracy is a consultant psychiatrist at Queen Mary’s Hospital in London. He is a trustee of the charity Mentor and has received honoraria from Janssen for delivering educational talks on novel psychoactive substances. His comments were adapted from an editorial (BMJ. 2019 Jun 12. doi: 10.1136/bmj.14021 ).
The findings by Molero et al. advance clinical knowledge about the drug class of gabapentinoids, wrote Derek K. Tracy, MB BCh. Though the study does not establish causality, it does rely on a solid, large dataset. The study shows the importance of uncoupling pregabalin and gabapentin. Both drugs are indeed gabapentinoids, but their use can lead to different outcomes, depending on the age of patients. For example, pregabalin – not gabapentin – appears tied to higher risks of harm. The demographic group that is most vulnerable is patients aged 15-24, the researchers found. Factors driving those age-related differences in risks tied to the drugs need to be understood.
Dr. Tracy is a consultant psychiatrist at Queen Mary’s Hospital in London. He is a trustee of the charity Mentor and has received honoraria from Janssen for delivering educational talks on novel psychoactive substances. His comments were adapted from an editorial (BMJ. 2019 Jun 12. doi: 10.1136/bmj.14021 ).
The findings by Molero et al. advance clinical knowledge about the drug class of gabapentinoids, wrote Derek K. Tracy, MB BCh. Though the study does not establish causality, it does rely on a solid, large dataset. The study shows the importance of uncoupling pregabalin and gabapentin. Both drugs are indeed gabapentinoids, but their use can lead to different outcomes, depending on the age of patients. For example, pregabalin – not gabapentin – appears tied to higher risks of harm. The demographic group that is most vulnerable is patients aged 15-24, the researchers found. Factors driving those age-related differences in risks tied to the drugs need to be understood.
Dr. Tracy is a consultant psychiatrist at Queen Mary’s Hospital in London. He is a trustee of the charity Mentor and has received honoraria from Janssen for delivering educational talks on novel psychoactive substances. His comments were adapted from an editorial (BMJ. 2019 Jun 12. doi: 10.1136/bmj.14021 ).
Young patients might be at increased risk of suicidal behavior, unintentional overdose, injuries, and traffic incidents during treatment periods with gabapentinoids, compared with periods without treatment with those medications, a cohort study of almost 200,000 people shows. Pregabalin is associated with higher hazards of those outcomes than is gabapentin, and the associations are strongest in patients aged 15-24 years, the researchers reported.
“If our findings are triangulated with other forms of evidence, ,” wrote Yasmina Molero, PhD, and associates. “Further restrictions for off-label prescription may need consideration.” The study was published in BMJ.
The use of gabapentinoids has risen in the United States (JAMA Intern Med. 2018;178[2]:292-4), and overdose deaths tied to gabapentin have led some states to explore reclassification of the drug as a controlled substance (Risk Manag Healthc Policy. 2018;11:109-16). In the United Kingdom, gabapentinoids are being reclassified as a class C controlled drug because of concerns about the risk of addiction, overdose, and safety, wrote Dr. Molero of the department of psychiatry at Warneford Hospital at the University of Oxford, England, and associates.
To study associations between gabapentinoids and adverse outcomes related to coordination, mental health, and criminality, Dr. Molero and her associates analyzed data from 191,973 people from the Swedish Prescribed Drug Register who collected prescriptions for pregabalin or gabapentin between 2006 and 2013. The researchers included patients aged 15 years and older in their analyses.
They examined suicidal behavior, unintentional overdoses, head or body injuries, road traffic incidents and offenses, and arrests for violent crime using the Swedish Patient Register and the National Crime Register. In addition, they defined suicidal behavior as emergency hospital visits attributable to self-injurious behavior or suicide attempt, or death by suicide. Unintentional overdoses were defined as emergency hospital visits or death attributable to poisoning by illicit drugs, medications, or biologic substances; accidental poisoning by noxious substances; or acute intoxications and overdoses by alcohol and illicit drugs, excluding intentional self-poisoning, wrote Dr. Molero, who is affiliated with the Karolinska Institute in Stockholm, and her associates.
Of the nearly 192,000 participants who collected prescriptions of gabapentinoids on at least two consecutive occasions, 120,664 received pregabalin, and 85,360 received gabapentin; 14,051 of the participants received both drugs. Fifty-nine percent were women, and most patients were aged 45 or older.
During the study period, 10,026 participants (5.2%) were treated for suicidal behavior or died from suicide, 17,144 participants (8.9%) experienced an unintentional overdose, and 12,070 participants (6.3%) had a road traffic incident or offense. In addition, 70,522 participants (36.7%) had head or body injuries, and 7,984 participants (4.1%) were arrested for a violent crime.
The study used a within-individual design that compared when a person was taking a gabapentinoid with when he or she was not. During treatment periods, participants were at increased risk of suicidal behavior or death from suicide (age-adjusted hazard ratio, 1.26), unintentional overdose (1.24), head or body injuries (1.22), and road traffic incidents or offenses (1.13). Associations with arrests for violent crime were not significant.
Gabapentinoid treatment was associated with increased hazards of suicidal behavior in people young than 55 years, but hazards were reduced or not significant in participants aged 55 years and older. Participants aged 15-24 years had the highest hazards of suicidal behavior (1.67).
In a sensitivity analysis, the researchers examined participants who had a diagnosis of comorbid epilepsy, psychiatric disorders, or musculoskeletal disorders before the start of gabapentinoid treatment. Among patients with comorbid epilepsy, gabapentinoids were not significantly associated with suicidal behavior and were associated with reduced hazards for all other outcomes.
“In comorbid psychiatric disorders, gabapentinoids were associated with lower risk for all outcomes,” the researchers said. Among patients with comorbid musculoskeletal disorders, gabapentinoids were associated with reductions in head or body injuries, traffic incidents, and arrests for violent crime.
Dr. Molero and her associates noted that they lacked information about alcohol and drug use, as well as treatment adherence and the conditions for which gabapentinoids had been prescribed. Furthermore, differences in prescription practices and outcome rates might affect the generalizability of the results to other countries.
The different results for pregabalin and gabapentin “could be due to their different pharmacodynamic and pharmacokinetic profiles; pregabalin has a higher potency, greater bioavailability, and quicker absorption than gabapentin. Pregabalin also has been associated with withdrawal symptoms following rapid discontinuation, which could be related to suicidal behavior,” Dr. Molero and colleagues said. “The reduced hazards in older people could reflect pharmacodynamic differences related to age, less concurrent use of alcohol or drugs, different indications for treatment, or reduced symptom severity of underlying conditions.”
The Wellcome Trust, Swedish Research Council, and Karolinska Institute supported the study. The authors had no disclosures relevant to the study. One author reported grants from Shire and Evolan and has served as a speaker for Shire.
SOURCE: Molero Y et al. BMJ. 2019 Jun 12. doi: 10.1136/bmj.l2147.
Young patients might be at increased risk of suicidal behavior, unintentional overdose, injuries, and traffic incidents during treatment periods with gabapentinoids, compared with periods without treatment with those medications, a cohort study of almost 200,000 people shows. Pregabalin is associated with higher hazards of those outcomes than is gabapentin, and the associations are strongest in patients aged 15-24 years, the researchers reported.
“If our findings are triangulated with other forms of evidence, ,” wrote Yasmina Molero, PhD, and associates. “Further restrictions for off-label prescription may need consideration.” The study was published in BMJ.
The use of gabapentinoids has risen in the United States (JAMA Intern Med. 2018;178[2]:292-4), and overdose deaths tied to gabapentin have led some states to explore reclassification of the drug as a controlled substance (Risk Manag Healthc Policy. 2018;11:109-16). In the United Kingdom, gabapentinoids are being reclassified as a class C controlled drug because of concerns about the risk of addiction, overdose, and safety, wrote Dr. Molero of the department of psychiatry at Warneford Hospital at the University of Oxford, England, and associates.
To study associations between gabapentinoids and adverse outcomes related to coordination, mental health, and criminality, Dr. Molero and her associates analyzed data from 191,973 people from the Swedish Prescribed Drug Register who collected prescriptions for pregabalin or gabapentin between 2006 and 2013. The researchers included patients aged 15 years and older in their analyses.
They examined suicidal behavior, unintentional overdoses, head or body injuries, road traffic incidents and offenses, and arrests for violent crime using the Swedish Patient Register and the National Crime Register. In addition, they defined suicidal behavior as emergency hospital visits attributable to self-injurious behavior or suicide attempt, or death by suicide. Unintentional overdoses were defined as emergency hospital visits or death attributable to poisoning by illicit drugs, medications, or biologic substances; accidental poisoning by noxious substances; or acute intoxications and overdoses by alcohol and illicit drugs, excluding intentional self-poisoning, wrote Dr. Molero, who is affiliated with the Karolinska Institute in Stockholm, and her associates.
Of the nearly 192,000 participants who collected prescriptions of gabapentinoids on at least two consecutive occasions, 120,664 received pregabalin, and 85,360 received gabapentin; 14,051 of the participants received both drugs. Fifty-nine percent were women, and most patients were aged 45 or older.
During the study period, 10,026 participants (5.2%) were treated for suicidal behavior or died from suicide, 17,144 participants (8.9%) experienced an unintentional overdose, and 12,070 participants (6.3%) had a road traffic incident or offense. In addition, 70,522 participants (36.7%) had head or body injuries, and 7,984 participants (4.1%) were arrested for a violent crime.
The study used a within-individual design that compared when a person was taking a gabapentinoid with when he or she was not. During treatment periods, participants were at increased risk of suicidal behavior or death from suicide (age-adjusted hazard ratio, 1.26), unintentional overdose (1.24), head or body injuries (1.22), and road traffic incidents or offenses (1.13). Associations with arrests for violent crime were not significant.
Gabapentinoid treatment was associated with increased hazards of suicidal behavior in people young than 55 years, but hazards were reduced or not significant in participants aged 55 years and older. Participants aged 15-24 years had the highest hazards of suicidal behavior (1.67).
In a sensitivity analysis, the researchers examined participants who had a diagnosis of comorbid epilepsy, psychiatric disorders, or musculoskeletal disorders before the start of gabapentinoid treatment. Among patients with comorbid epilepsy, gabapentinoids were not significantly associated with suicidal behavior and were associated with reduced hazards for all other outcomes.
“In comorbid psychiatric disorders, gabapentinoids were associated with lower risk for all outcomes,” the researchers said. Among patients with comorbid musculoskeletal disorders, gabapentinoids were associated with reductions in head or body injuries, traffic incidents, and arrests for violent crime.
Dr. Molero and her associates noted that they lacked information about alcohol and drug use, as well as treatment adherence and the conditions for which gabapentinoids had been prescribed. Furthermore, differences in prescription practices and outcome rates might affect the generalizability of the results to other countries.
The different results for pregabalin and gabapentin “could be due to their different pharmacodynamic and pharmacokinetic profiles; pregabalin has a higher potency, greater bioavailability, and quicker absorption than gabapentin. Pregabalin also has been associated with withdrawal symptoms following rapid discontinuation, which could be related to suicidal behavior,” Dr. Molero and colleagues said. “The reduced hazards in older people could reflect pharmacodynamic differences related to age, less concurrent use of alcohol or drugs, different indications for treatment, or reduced symptom severity of underlying conditions.”
The Wellcome Trust, Swedish Research Council, and Karolinska Institute supported the study. The authors had no disclosures relevant to the study. One author reported grants from Shire and Evolan and has served as a speaker for Shire.
SOURCE: Molero Y et al. BMJ. 2019 Jun 12. doi: 10.1136/bmj.l2147.
FROM BMJ
Key clinical point: Patients might be at increased risk of suicidal behavior, unintentional overdose, head and body injuries, and traffic incidents during periods of treatment with gabapentinoids. Pregabalin is associated with higher hazards of these outcomes than is gabapentin, and the associations are strongest in patients aged 15-24 years.
Major finding: During treatment periods, patients were at increased risk of suicidal behavior or death from suicide (age-adjusted hazard ratio, 1.26), unintentional overdose (1.24), head or body injuries (1.22), and road traffic incidents or offenses (1.13).
Study details: An analysis of data from 191,973 people from the Swedish Prescribed Drug Register, which collected prescriptions for pregabalin or gabapentin between 2006 and 2013.
Disclosures: The Wellcome Trust, Swedish Research Council, and Karolinska Institute supported the study. The authors had no relevant disclosures. One author reported grants from Shire and Evolan, and has served as a speaker for Shire.
Source: Molero Y et al. BMJ. 2019 Jun 12. doi: 10.1136/bmj.l2147.
Psychiatry residents not getting training in treating chronic pain
SAN FRANCISCO –
Given the unique role of psychiatrists in helping chronic pain patients with coping strategies and managing comorbid psychiatric illness, this void is concerning, said Ali Ahsan Ali, MD, a resident psychiatrist at the Micah School of Medicine at Mount Sinai/Elmhurst Hospital Center in New York, in an interview at the annual meeting of the American Psychiatric Association.
In a video interview, Dr. Ali spoke with Ahmar M. Butt, MD, about how and why Dr. Ali and his colleagues conducted the survey of all 221 U.S. psychiatry residency programs in January 2019. They also discuss the implications of these trends for patients, particularly in light of the country’s opioid crisis.
Dr. Ali had no disclosures. Dr. Butt is board certified in general psychiatry, child and adolescent psychiatry, and preventive medicine, with a subspecialty in addiction medicine. Dr. Butt is interim program director of the psychiatry residency program at Broadlawns UnityPointe Health, Des Moines, Iowa. He had no disclosures.
SAN FRANCISCO –
Given the unique role of psychiatrists in helping chronic pain patients with coping strategies and managing comorbid psychiatric illness, this void is concerning, said Ali Ahsan Ali, MD, a resident psychiatrist at the Micah School of Medicine at Mount Sinai/Elmhurst Hospital Center in New York, in an interview at the annual meeting of the American Psychiatric Association.
In a video interview, Dr. Ali spoke with Ahmar M. Butt, MD, about how and why Dr. Ali and his colleagues conducted the survey of all 221 U.S. psychiatry residency programs in January 2019. They also discuss the implications of these trends for patients, particularly in light of the country’s opioid crisis.
Dr. Ali had no disclosures. Dr. Butt is board certified in general psychiatry, child and adolescent psychiatry, and preventive medicine, with a subspecialty in addiction medicine. Dr. Butt is interim program director of the psychiatry residency program at Broadlawns UnityPointe Health, Des Moines, Iowa. He had no disclosures.
SAN FRANCISCO –
Given the unique role of psychiatrists in helping chronic pain patients with coping strategies and managing comorbid psychiatric illness, this void is concerning, said Ali Ahsan Ali, MD, a resident psychiatrist at the Micah School of Medicine at Mount Sinai/Elmhurst Hospital Center in New York, in an interview at the annual meeting of the American Psychiatric Association.
In a video interview, Dr. Ali spoke with Ahmar M. Butt, MD, about how and why Dr. Ali and his colleagues conducted the survey of all 221 U.S. psychiatry residency programs in January 2019. They also discuss the implications of these trends for patients, particularly in light of the country’s opioid crisis.
Dr. Ali had no disclosures. Dr. Butt is board certified in general psychiatry, child and adolescent psychiatry, and preventive medicine, with a subspecialty in addiction medicine. Dr. Butt is interim program director of the psychiatry residency program at Broadlawns UnityPointe Health, Des Moines, Iowa. He had no disclosures.
REPORTING FROM APA 2019
Varicella vaccine delivers doubled benefit to children
than those in unvaccinated children.
The benefit became largely apparent after children received the second vaccination in the recommended series, and persisted throughout childhood, Sheila Weinmann, PhD, of Kaiser Permanente Northern California, Oakland, and colleagues said.*
The analysis included 6.37 million children in the Kaiser Permanente database, 50% of whom were vaccinated for all or some of the study period stretching from 2003 to 2014. Overall, the crude lab-confirmed herpes zoster (HZ) incidence rate was 74/100,000 person-years. When stratified by vaccine status, the crude rate of HZ among vaccinated children was 78% lower than among unvaccinated children (38 vs. 170 cases per 100,000 person years).
Herpes zoster was more common among girls than boys and up to six times more common in immunosuppressed children than in nonimmunosuppressed children.
The authors also found that unvaccinated children benefited from the high rate of vaccination around them. Although the HZ rate was always lower among vaccinated children, the rate among unvaccinated children fell sharply after 2007.
“The trend of decreasing HZ incidence among children who were unvaccinated is likely due to a lack of primary VZV [varicella-zoster virus] infection resulting from herd immunity in a highly vaccinated population,” Dr. Weinmann and her associates said.
There was some variability among age groups, especially among the youngest who were not fully vaccinated.
“In the group aged 1-2 years, the confirmation-adjusted HZ rate among children who were vaccinated was 70% higher than among those who were unvaccinated,” the authors said. In the “older groups, HZ rates were significantly higher in children who were unvaccinated than in those who were vaccinated,” the researchers noted.
The highest incidence was among vaccinated 1-year-olds, who had a 140% higher risk of HZ than did unvaccinated 1-year-olds. But this risk elevation disappeared by age 2 years. For everyone else, aged 2-17 years, the rate of HZ remained significantly lower in vaccinated children.
“Among the small number of children vaccinated at 11 months of age (for whom the vaccine is not recommended), the HZ incidence rate was significantly higher than in children vaccinated at 1 year of age and older. Similarly, children who contract wild-type varicella infection at younger than 1 year of age also have a higher risk of HZ (relative risk, 13.5). The immature adaptive T-cell response in children less than 1 year of age appears less able to contain VZV as a latent infection, compared with older children.
“Our findings for 11-month-olds who were vaccinated should be interpreted with caution because this population included only three cases of HZ and could have included children participating in a prelicensure study with a vaccine formulation different from Varivax,” Dr. Weinmann and her associates said.
Dr. Weinmann and her associates reported no relevant financial disclosures. The study was supported by the Centers for Disease Control and Prevention.
SOURCE: Weinmann S et al. Pediatrics. 2019 Jun 10. doi: 10.1542/peds.2018-2917.
* This article was updated 6/14/2019
The finding of a 78% lower incidence of zoster in varicella-vaccinated children is nothing short of “remarkable,” Anne A Gershon, MD, wrote in an accompanying editorial.
But the benefit could be in jeopardy, as parents question the safety and effectiveness of all vaccines, she wrote.
“That the varicella vaccine prevents not only varicella but zoster as well is an exciting dual benefit from the varicella vaccine, further improving the health of children by immunization,” Dr. Gershon said. “Additional studies will be necessary to show the mechanism for the protection against zoster (viral, immunologic, or both), how long this benefit lasts, and whether additional doses of some form of VZV [varicella-zoster virus] vaccine will be more useful.”
But, she suggested, in a time when cases of clinical varicella are dwindling, so is public awareness of the vaccine’s benefit. Clinical varicella is worse for adults than it is for children.
“Efforts to immunize all children against chickenpox must continue to be made to protect our population from wild-type VZV. Fortunately, antiviral therapy is also available for individuals who are unvaccinated and develop varicella or zoster, but immunization is, as usual, preferable,” Dr. Gershon concluded.
Dr. Gershon, a pediatric infectious disease specialist, is a professor of pediatrics at Columbia University, New York. She wrote a commentary to accompany the article by Weinmann et al. (Pediatrics. 2019 Jun 10. doi: 10.1542/peds.2018-3561). Dr. Gershon had no relevant financial disclosures. The commentary was funded by the National Institutes of Health.
The finding of a 78% lower incidence of zoster in varicella-vaccinated children is nothing short of “remarkable,” Anne A Gershon, MD, wrote in an accompanying editorial.
But the benefit could be in jeopardy, as parents question the safety and effectiveness of all vaccines, she wrote.
“That the varicella vaccine prevents not only varicella but zoster as well is an exciting dual benefit from the varicella vaccine, further improving the health of children by immunization,” Dr. Gershon said. “Additional studies will be necessary to show the mechanism for the protection against zoster (viral, immunologic, or both), how long this benefit lasts, and whether additional doses of some form of VZV [varicella-zoster virus] vaccine will be more useful.”
But, she suggested, in a time when cases of clinical varicella are dwindling, so is public awareness of the vaccine’s benefit. Clinical varicella is worse for adults than it is for children.
“Efforts to immunize all children against chickenpox must continue to be made to protect our population from wild-type VZV. Fortunately, antiviral therapy is also available for individuals who are unvaccinated and develop varicella or zoster, but immunization is, as usual, preferable,” Dr. Gershon concluded.
Dr. Gershon, a pediatric infectious disease specialist, is a professor of pediatrics at Columbia University, New York. She wrote a commentary to accompany the article by Weinmann et al. (Pediatrics. 2019 Jun 10. doi: 10.1542/peds.2018-3561). Dr. Gershon had no relevant financial disclosures. The commentary was funded by the National Institutes of Health.
The finding of a 78% lower incidence of zoster in varicella-vaccinated children is nothing short of “remarkable,” Anne A Gershon, MD, wrote in an accompanying editorial.
But the benefit could be in jeopardy, as parents question the safety and effectiveness of all vaccines, she wrote.
“That the varicella vaccine prevents not only varicella but zoster as well is an exciting dual benefit from the varicella vaccine, further improving the health of children by immunization,” Dr. Gershon said. “Additional studies will be necessary to show the mechanism for the protection against zoster (viral, immunologic, or both), how long this benefit lasts, and whether additional doses of some form of VZV [varicella-zoster virus] vaccine will be more useful.”
But, she suggested, in a time when cases of clinical varicella are dwindling, so is public awareness of the vaccine’s benefit. Clinical varicella is worse for adults than it is for children.
“Efforts to immunize all children against chickenpox must continue to be made to protect our population from wild-type VZV. Fortunately, antiviral therapy is also available for individuals who are unvaccinated and develop varicella or zoster, but immunization is, as usual, preferable,” Dr. Gershon concluded.
Dr. Gershon, a pediatric infectious disease specialist, is a professor of pediatrics at Columbia University, New York. She wrote a commentary to accompany the article by Weinmann et al. (Pediatrics. 2019 Jun 10. doi: 10.1542/peds.2018-3561). Dr. Gershon had no relevant financial disclosures. The commentary was funded by the National Institutes of Health.
than those in unvaccinated children.
The benefit became largely apparent after children received the second vaccination in the recommended series, and persisted throughout childhood, Sheila Weinmann, PhD, of Kaiser Permanente Northern California, Oakland, and colleagues said.*
The analysis included 6.37 million children in the Kaiser Permanente database, 50% of whom were vaccinated for all or some of the study period stretching from 2003 to 2014. Overall, the crude lab-confirmed herpes zoster (HZ) incidence rate was 74/100,000 person-years. When stratified by vaccine status, the crude rate of HZ among vaccinated children was 78% lower than among unvaccinated children (38 vs. 170 cases per 100,000 person years).
Herpes zoster was more common among girls than boys and up to six times more common in immunosuppressed children than in nonimmunosuppressed children.
The authors also found that unvaccinated children benefited from the high rate of vaccination around them. Although the HZ rate was always lower among vaccinated children, the rate among unvaccinated children fell sharply after 2007.
“The trend of decreasing HZ incidence among children who were unvaccinated is likely due to a lack of primary VZV [varicella-zoster virus] infection resulting from herd immunity in a highly vaccinated population,” Dr. Weinmann and her associates said.
There was some variability among age groups, especially among the youngest who were not fully vaccinated.
“In the group aged 1-2 years, the confirmation-adjusted HZ rate among children who were vaccinated was 70% higher than among those who were unvaccinated,” the authors said. In the “older groups, HZ rates were significantly higher in children who were unvaccinated than in those who were vaccinated,” the researchers noted.
The highest incidence was among vaccinated 1-year-olds, who had a 140% higher risk of HZ than did unvaccinated 1-year-olds. But this risk elevation disappeared by age 2 years. For everyone else, aged 2-17 years, the rate of HZ remained significantly lower in vaccinated children.
“Among the small number of children vaccinated at 11 months of age (for whom the vaccine is not recommended), the HZ incidence rate was significantly higher than in children vaccinated at 1 year of age and older. Similarly, children who contract wild-type varicella infection at younger than 1 year of age also have a higher risk of HZ (relative risk, 13.5). The immature adaptive T-cell response in children less than 1 year of age appears less able to contain VZV as a latent infection, compared with older children.
“Our findings for 11-month-olds who were vaccinated should be interpreted with caution because this population included only three cases of HZ and could have included children participating in a prelicensure study with a vaccine formulation different from Varivax,” Dr. Weinmann and her associates said.
Dr. Weinmann and her associates reported no relevant financial disclosures. The study was supported by the Centers for Disease Control and Prevention.
SOURCE: Weinmann S et al. Pediatrics. 2019 Jun 10. doi: 10.1542/peds.2018-2917.
* This article was updated 6/14/2019
than those in unvaccinated children.
The benefit became largely apparent after children received the second vaccination in the recommended series, and persisted throughout childhood, Sheila Weinmann, PhD, of Kaiser Permanente Northern California, Oakland, and colleagues said.*
The analysis included 6.37 million children in the Kaiser Permanente database, 50% of whom were vaccinated for all or some of the study period stretching from 2003 to 2014. Overall, the crude lab-confirmed herpes zoster (HZ) incidence rate was 74/100,000 person-years. When stratified by vaccine status, the crude rate of HZ among vaccinated children was 78% lower than among unvaccinated children (38 vs. 170 cases per 100,000 person years).
Herpes zoster was more common among girls than boys and up to six times more common in immunosuppressed children than in nonimmunosuppressed children.
The authors also found that unvaccinated children benefited from the high rate of vaccination around them. Although the HZ rate was always lower among vaccinated children, the rate among unvaccinated children fell sharply after 2007.
“The trend of decreasing HZ incidence among children who were unvaccinated is likely due to a lack of primary VZV [varicella-zoster virus] infection resulting from herd immunity in a highly vaccinated population,” Dr. Weinmann and her associates said.
There was some variability among age groups, especially among the youngest who were not fully vaccinated.
“In the group aged 1-2 years, the confirmation-adjusted HZ rate among children who were vaccinated was 70% higher than among those who were unvaccinated,” the authors said. In the “older groups, HZ rates were significantly higher in children who were unvaccinated than in those who were vaccinated,” the researchers noted.
The highest incidence was among vaccinated 1-year-olds, who had a 140% higher risk of HZ than did unvaccinated 1-year-olds. But this risk elevation disappeared by age 2 years. For everyone else, aged 2-17 years, the rate of HZ remained significantly lower in vaccinated children.
“Among the small number of children vaccinated at 11 months of age (for whom the vaccine is not recommended), the HZ incidence rate was significantly higher than in children vaccinated at 1 year of age and older. Similarly, children who contract wild-type varicella infection at younger than 1 year of age also have a higher risk of HZ (relative risk, 13.5). The immature adaptive T-cell response in children less than 1 year of age appears less able to contain VZV as a latent infection, compared with older children.
“Our findings for 11-month-olds who were vaccinated should be interpreted with caution because this population included only three cases of HZ and could have included children participating in a prelicensure study with a vaccine formulation different from Varivax,” Dr. Weinmann and her associates said.
Dr. Weinmann and her associates reported no relevant financial disclosures. The study was supported by the Centers for Disease Control and Prevention.
SOURCE: Weinmann S et al. Pediatrics. 2019 Jun 10. doi: 10.1542/peds.2018-2917.
* This article was updated 6/14/2019
FROM PEDIATRICS
Key clinical point: Varicella vaccine is preventing pediatric zoster among children aged 2-17 years.
Major finding: Varicella-vaccinated children have a 78% lower incidence of pediatric zoster than do unvaccinated children.
Study details: The population-based cohort study included more than 6.3 million children.
Disclosures: Dr. Weinmann and her associates reported no relevant financial disclosures. The study was supported by the Centers for Disease Control and Prevention.
Source: Weinmann S et al. Pediatrics. 2019. doi: 10.1542/peds.2018-2917.
Less Is More When It Comes to Ketorolac for Pain

A 46-year-old man with no significant medical history presents to the emergency department (ED) with right flank pain and nausea. CT reveals a 5-mm ureteral stone with no obstruction or hydronephrosis. You are planning to start him on IV ketorolac for pain. What is the most appropriate dose?
Ketorolac tromethamine is a highly effective NSAID. As a nonopiate analgesic, it is often the first choice for the treatment of acute pain in the flank, abdomen, musculoskeletal system, or head.2 While it is not associated with euphoria, withdrawal effects, or respiratory depression (like its opiate analgesic counterparts), ketorolac carries an FDA black-box warning for gastrointestinal, cardiovascular, renal, and bleeding risks.3
NSAIDs are known to have a “ceiling dose” at which maximum analgesic benefit is achieved; higher doses will not provide further pain relief. Higher doses of ketorolac may be used when the anti-inflammatory effects of NSAIDs are desired, but they are likely to cause more adverse effects.4 Available data describe the ceiling dose of ketorolac as 10 mg across dosage forms—yet the majority of research and most health care providers in current practice use higher doses (20 to 60 mg).4,5 The FDA-approved labeling provides for a maximum dose of 60 mg/d.3
In one recent study, ketorolac was prescribed above its ceiling dose in at least 97% of patients who received IV doses and at least 96% of those who received intramuscular (IM) doses in a US ED.6 If 10 mg of ketorolac is an effective analgesic dose, current practice exceeds the label recommendation to use the lowest effective dose. This study sought to determine the comparative efficacy of 3 different doses of IV ketorolac for acute pain management in an ED.
STUDY SUMMARY
10 mg of ketorolac is enough for pain
This randomized double-blind trial evaluated the effectiveness of ketorolac in 240 adult patients (ages 18 to 65) presenting to an ED with acute flank, abdominal, musculoskeletal, or headache pain.1 Acute pain was defined as onset within the past 30 days.
Patients were randomly assigned to receive either 10, 15, or 30 mg of IV ketorolac in 10 mL of normal saline. A pharmacist prepared the medication in identical syringes, which were delivered in a blinded manner to the nurses caring for the patients. Pain (measured using a 0-to-10 scale), vital signs, and adverse effects were assessed at baseline and at 15, 30, 60, 90, and 120 minutes. If patients were still in pain at 30 minutes, IV morphine (0.1 mg/kg) was offered. The primary outcome was a numerical pain score at 30 minutes after ketorolac administration; secondary outcomes included the occurrence of adverse events and the use of rescue
The treatment groups were similar in terms of demographics and baseline vital signs. Mean age was 39 to 42. Across the 3 groups, 36% to 40% of patients had abdominal pain, 26% to 39% had flank pain, 20% to 26% had musculoskeletal pain, and 1% to 11% had headache pain. Patients had experienced pain for an average of 1.5 to 3.5 days.
Continue to: Baseline pain scores...
Baseline pain scores were similar for all 3 groups (7.5-7.8 on a 10-point scale). In the intention-to-treat analysis, all 3 doses of ketorolac decreased pain significantly at 30 minutes, with no difference between the groups: mean pain scores postintervention were 5.1 for the 10- and 15-mg group and 4.8 for the 30-mg group. There was no difference between the groups at any other time intervals. There was also no difference between groups in the number of patients who needed rescue medication at 30 minutes (4 patients in the 10-mg group, 3 patients in the 15-mg group, and 4 patients in the 30-mg group). In addition, adverse events (eg, dizziness, nausea, headache, itching, flushing) did not differ between the groups.
WHAT’S NEW
10 mg is just as effective as 30 mg
This trial confirms that a low dose of IV ketorolac is just as effective as higher doses for acute pain control.
CAVEATS
2-hour limit; no look at long-term effects
It isn’t known whether the higher dose would have provided greater pain relief beyond the 120 minutes evaluated in this trial, or if alternative dosage forms (oral or IM) would result in different outcomes. This study was not designed to compare serious long-term adverse effects such as bleeding, renal impairment, or cardiovascular events. Additionally, this study was not powered to look at specific therapeutic indications or anti-inflammatory response.
CHALLENGES TO IMPLEMENTATION
10-mg single-dose vial not readily available
Ketorolac tromethamine for injection is available in the United States in 15-, 30-, and 60-mg single-dose vials. Because a 10-mg dose is not available as a single-dose vial, it would need to be specially prepared (as it was in this study). However, this study should reassure providers that using the lowest available dose (eg, 15 mg IV if that is what is available) will relieve acute pain as well as higher doses will. CR
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
Copyright © 2019. The Family Physicians Inquiries Network. All rights reserved.
Reprinted with permission from the Family Physicians Inquiries Network and The Journal of Family Practice (2019;68[1]:41-42).
1. Motov S, Yasavolian M, Likourezos A, et al. Comparison of intravenous ketorolac at three single-dose regimens for treating acute pain in the emergency department: a randomized controlled trial. Ann Emerg Med. 2017; 70:177-184.
2. Buckley MM, Brogden RN. Ketorolac: a review of its pharmacodynamic and pharmacokinetic properties, and therapeutic potential. Drugs. 1990;39: 86-109.
3. Ketorolac tromethamine [package insert]. Bedford, OH: Bedford Laboratories; 2009.
4. Catapano MS. The analgesic efficacy of ketorolac for acute pain. J Emerg Med. 1996;14:67-75.
5. García Rodríguez LA, Cattaruzzi C, Troncon MG, et al. Risk of hospitalization for upper gastrointestinal tract bleeding associated with ketorolac, other nonsteroidal anti-inflammatory drugs, calcium antagonists, and other antihypertensive drugs. Arch Intern Med. 1998;158:33-39.
6. Soleyman-Zomalan E, Motov S, Likourezos A, et al. Patterns of ketorolac dosing by emergency physicians. World J Emerg Med. 2017;8:43-46.

A 46-year-old man with no significant medical history presents to the emergency department (ED) with right flank pain and nausea. CT reveals a 5-mm ureteral stone with no obstruction or hydronephrosis. You are planning to start him on IV ketorolac for pain. What is the most appropriate dose?
Ketorolac tromethamine is a highly effective NSAID. As a nonopiate analgesic, it is often the first choice for the treatment of acute pain in the flank, abdomen, musculoskeletal system, or head.2 While it is not associated with euphoria, withdrawal effects, or respiratory depression (like its opiate analgesic counterparts), ketorolac carries an FDA black-box warning for gastrointestinal, cardiovascular, renal, and bleeding risks.3
NSAIDs are known to have a “ceiling dose” at which maximum analgesic benefit is achieved; higher doses will not provide further pain relief. Higher doses of ketorolac may be used when the anti-inflammatory effects of NSAIDs are desired, but they are likely to cause more adverse effects.4 Available data describe the ceiling dose of ketorolac as 10 mg across dosage forms—yet the majority of research and most health care providers in current practice use higher doses (20 to 60 mg).4,5 The FDA-approved labeling provides for a maximum dose of 60 mg/d.3
In one recent study, ketorolac was prescribed above its ceiling dose in at least 97% of patients who received IV doses and at least 96% of those who received intramuscular (IM) doses in a US ED.6 If 10 mg of ketorolac is an effective analgesic dose, current practice exceeds the label recommendation to use the lowest effective dose. This study sought to determine the comparative efficacy of 3 different doses of IV ketorolac for acute pain management in an ED.
STUDY SUMMARY
10 mg of ketorolac is enough for pain
This randomized double-blind trial evaluated the effectiveness of ketorolac in 240 adult patients (ages 18 to 65) presenting to an ED with acute flank, abdominal, musculoskeletal, or headache pain.1 Acute pain was defined as onset within the past 30 days.
Patients were randomly assigned to receive either 10, 15, or 30 mg of IV ketorolac in 10 mL of normal saline. A pharmacist prepared the medication in identical syringes, which were delivered in a blinded manner to the nurses caring for the patients. Pain (measured using a 0-to-10 scale), vital signs, and adverse effects were assessed at baseline and at 15, 30, 60, 90, and 120 minutes. If patients were still in pain at 30 minutes, IV morphine (0.1 mg/kg) was offered. The primary outcome was a numerical pain score at 30 minutes after ketorolac administration; secondary outcomes included the occurrence of adverse events and the use of rescue
The treatment groups were similar in terms of demographics and baseline vital signs. Mean age was 39 to 42. Across the 3 groups, 36% to 40% of patients had abdominal pain, 26% to 39% had flank pain, 20% to 26% had musculoskeletal pain, and 1% to 11% had headache pain. Patients had experienced pain for an average of 1.5 to 3.5 days.
Continue to: Baseline pain scores...
Baseline pain scores were similar for all 3 groups (7.5-7.8 on a 10-point scale). In the intention-to-treat analysis, all 3 doses of ketorolac decreased pain significantly at 30 minutes, with no difference between the groups: mean pain scores postintervention were 5.1 for the 10- and 15-mg group and 4.8 for the 30-mg group. There was no difference between the groups at any other time intervals. There was also no difference between groups in the number of patients who needed rescue medication at 30 minutes (4 patients in the 10-mg group, 3 patients in the 15-mg group, and 4 patients in the 30-mg group). In addition, adverse events (eg, dizziness, nausea, headache, itching, flushing) did not differ between the groups.
WHAT’S NEW
10 mg is just as effective as 30 mg
This trial confirms that a low dose of IV ketorolac is just as effective as higher doses for acute pain control.
CAVEATS
2-hour limit; no look at long-term effects
It isn’t known whether the higher dose would have provided greater pain relief beyond the 120 minutes evaluated in this trial, or if alternative dosage forms (oral or IM) would result in different outcomes. This study was not designed to compare serious long-term adverse effects such as bleeding, renal impairment, or cardiovascular events. Additionally, this study was not powered to look at specific therapeutic indications or anti-inflammatory response.
CHALLENGES TO IMPLEMENTATION
10-mg single-dose vial not readily available
Ketorolac tromethamine for injection is available in the United States in 15-, 30-, and 60-mg single-dose vials. Because a 10-mg dose is not available as a single-dose vial, it would need to be specially prepared (as it was in this study). However, this study should reassure providers that using the lowest available dose (eg, 15 mg IV if that is what is available) will relieve acute pain as well as higher doses will. CR
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
Copyright © 2019. The Family Physicians Inquiries Network. All rights reserved.
Reprinted with permission from the Family Physicians Inquiries Network and The Journal of Family Practice (2019;68[1]:41-42).

A 46-year-old man with no significant medical history presents to the emergency department (ED) with right flank pain and nausea. CT reveals a 5-mm ureteral stone with no obstruction or hydronephrosis. You are planning to start him on IV ketorolac for pain. What is the most appropriate dose?
Ketorolac tromethamine is a highly effective NSAID. As a nonopiate analgesic, it is often the first choice for the treatment of acute pain in the flank, abdomen, musculoskeletal system, or head.2 While it is not associated with euphoria, withdrawal effects, or respiratory depression (like its opiate analgesic counterparts), ketorolac carries an FDA black-box warning for gastrointestinal, cardiovascular, renal, and bleeding risks.3
NSAIDs are known to have a “ceiling dose” at which maximum analgesic benefit is achieved; higher doses will not provide further pain relief. Higher doses of ketorolac may be used when the anti-inflammatory effects of NSAIDs are desired, but they are likely to cause more adverse effects.4 Available data describe the ceiling dose of ketorolac as 10 mg across dosage forms—yet the majority of research and most health care providers in current practice use higher doses (20 to 60 mg).4,5 The FDA-approved labeling provides for a maximum dose of 60 mg/d.3
In one recent study, ketorolac was prescribed above its ceiling dose in at least 97% of patients who received IV doses and at least 96% of those who received intramuscular (IM) doses in a US ED.6 If 10 mg of ketorolac is an effective analgesic dose, current practice exceeds the label recommendation to use the lowest effective dose. This study sought to determine the comparative efficacy of 3 different doses of IV ketorolac for acute pain management in an ED.
STUDY SUMMARY
10 mg of ketorolac is enough for pain
This randomized double-blind trial evaluated the effectiveness of ketorolac in 240 adult patients (ages 18 to 65) presenting to an ED with acute flank, abdominal, musculoskeletal, or headache pain.1 Acute pain was defined as onset within the past 30 days.
Patients were randomly assigned to receive either 10, 15, or 30 mg of IV ketorolac in 10 mL of normal saline. A pharmacist prepared the medication in identical syringes, which were delivered in a blinded manner to the nurses caring for the patients. Pain (measured using a 0-to-10 scale), vital signs, and adverse effects were assessed at baseline and at 15, 30, 60, 90, and 120 minutes. If patients were still in pain at 30 minutes, IV morphine (0.1 mg/kg) was offered. The primary outcome was a numerical pain score at 30 minutes after ketorolac administration; secondary outcomes included the occurrence of adverse events and the use of rescue
The treatment groups were similar in terms of demographics and baseline vital signs. Mean age was 39 to 42. Across the 3 groups, 36% to 40% of patients had abdominal pain, 26% to 39% had flank pain, 20% to 26% had musculoskeletal pain, and 1% to 11% had headache pain. Patients had experienced pain for an average of 1.5 to 3.5 days.
Continue to: Baseline pain scores...
Baseline pain scores were similar for all 3 groups (7.5-7.8 on a 10-point scale). In the intention-to-treat analysis, all 3 doses of ketorolac decreased pain significantly at 30 minutes, with no difference between the groups: mean pain scores postintervention were 5.1 for the 10- and 15-mg group and 4.8 for the 30-mg group. There was no difference between the groups at any other time intervals. There was also no difference between groups in the number of patients who needed rescue medication at 30 minutes (4 patients in the 10-mg group, 3 patients in the 15-mg group, and 4 patients in the 30-mg group). In addition, adverse events (eg, dizziness, nausea, headache, itching, flushing) did not differ between the groups.
WHAT’S NEW
10 mg is just as effective as 30 mg
This trial confirms that a low dose of IV ketorolac is just as effective as higher doses for acute pain control.
CAVEATS
2-hour limit; no look at long-term effects
It isn’t known whether the higher dose would have provided greater pain relief beyond the 120 minutes evaluated in this trial, or if alternative dosage forms (oral or IM) would result in different outcomes. This study was not designed to compare serious long-term adverse effects such as bleeding, renal impairment, or cardiovascular events. Additionally, this study was not powered to look at specific therapeutic indications or anti-inflammatory response.
CHALLENGES TO IMPLEMENTATION
10-mg single-dose vial not readily available
Ketorolac tromethamine for injection is available in the United States in 15-, 30-, and 60-mg single-dose vials. Because a 10-mg dose is not available as a single-dose vial, it would need to be specially prepared (as it was in this study). However, this study should reassure providers that using the lowest available dose (eg, 15 mg IV if that is what is available) will relieve acute pain as well as higher doses will. CR
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
Copyright © 2019. The Family Physicians Inquiries Network. All rights reserved.
Reprinted with permission from the Family Physicians Inquiries Network and The Journal of Family Practice (2019;68[1]:41-42).
1. Motov S, Yasavolian M, Likourezos A, et al. Comparison of intravenous ketorolac at three single-dose regimens for treating acute pain in the emergency department: a randomized controlled trial. Ann Emerg Med. 2017; 70:177-184.
2. Buckley MM, Brogden RN. Ketorolac: a review of its pharmacodynamic and pharmacokinetic properties, and therapeutic potential. Drugs. 1990;39: 86-109.
3. Ketorolac tromethamine [package insert]. Bedford, OH: Bedford Laboratories; 2009.
4. Catapano MS. The analgesic efficacy of ketorolac for acute pain. J Emerg Med. 1996;14:67-75.
5. García Rodríguez LA, Cattaruzzi C, Troncon MG, et al. Risk of hospitalization for upper gastrointestinal tract bleeding associated with ketorolac, other nonsteroidal anti-inflammatory drugs, calcium antagonists, and other antihypertensive drugs. Arch Intern Med. 1998;158:33-39.
6. Soleyman-Zomalan E, Motov S, Likourezos A, et al. Patterns of ketorolac dosing by emergency physicians. World J Emerg Med. 2017;8:43-46.
1. Motov S, Yasavolian M, Likourezos A, et al. Comparison of intravenous ketorolac at three single-dose regimens for treating acute pain in the emergency department: a randomized controlled trial. Ann Emerg Med. 2017; 70:177-184.
2. Buckley MM, Brogden RN. Ketorolac: a review of its pharmacodynamic and pharmacokinetic properties, and therapeutic potential. Drugs. 1990;39: 86-109.
3. Ketorolac tromethamine [package insert]. Bedford, OH: Bedford Laboratories; 2009.
4. Catapano MS. The analgesic efficacy of ketorolac for acute pain. J Emerg Med. 1996;14:67-75.
5. García Rodríguez LA, Cattaruzzi C, Troncon MG, et al. Risk of hospitalization for upper gastrointestinal tract bleeding associated with ketorolac, other nonsteroidal anti-inflammatory drugs, calcium antagonists, and other antihypertensive drugs. Arch Intern Med. 1998;158:33-39.
6. Soleyman-Zomalan E, Motov S, Likourezos A, et al. Patterns of ketorolac dosing by emergency physicians. World J Emerg Med. 2017;8:43-46.