User login
Acute myocarditis a possible complication of monkeypox
Clinicians in Portugal say a 31-year-old man with confirmed monkeypox developed acute myocarditis roughly 1 week after the eruption of the characteristic skin lesions of the disease.
Ana Isabel Pinho, MD, department of cardiology, São João University Hospital Centre, Porto, Portugal, said in a news release.
“We believe that reporting this potential causal relationship can raise more awareness of the scientific community and health professionals for acute myocarditis as a possible complication associated with monkeypox and might be helpful for close monitoring of affected patients for further recognition of other complications in the future,” Dr. Pinho adds.
Dr. Pinho and colleagues describe the case in a report published in JACC: Case Reports.
Case details
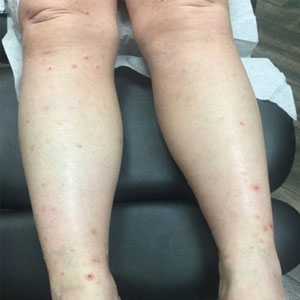
The patient presented with a 5-day history of malaise, myalgias, and fever followed by the eruption of multiple swollen skin lesions on his face, hands, and genitalia.
Monkeypox was confirmed by positive polymerase chain reaction assay of a swab sample from a skin lesion.
Three days later, the patient developed chest tightness that radiated through the left arm and which awoke him during the night. He was admitted to an intensive care unit with clinical suspicion of acute myocarditis.
The patient’s initial electrocardiogram showed sinus rhythm with nonspecific ventricular repolarization abnormalities.
On chest x-ray, the cardiothoracic index was normal, with no interstitial infiltrates, pleural effusion, or masses. On transthoracic echocardigraphy, biventricular systolic function was preserved, and there was no pericardial effusion.
Routine laboratory tests revealed elevated levels of C-reactive protein, creatine phosphokinase, high-sensitivity troponin I, and brain natriuretic peptide, suggesting stress injury to the heart.
Findings on cardiac magnetic resonance were consistent with myocardial inflammation and acute myocarditis.
The patient was treated with supportive care, and he made a full clinical recovery. He was discharged after 1 week. On discharge, cardiac enzymes were within the normal range. The patient showed sustained electric and hemodynamic stability, and the skin lesions had healed.
“Through this important case study, we are developing a deeper understanding of monkeypox, viral myocarditis, and how to accurately diagnose and manage this disease,” Julia Grapsa, MD, PhD, editor-in-chief of JACC: Case Reports, commented in the news release.
“I commend the authors on this valuable clinical case during a critical time as monkeypox continues to spread globally,” Dr. Grapsa added.
The researchers say further research is needed to identify the pathologic mechanism underlying monkeypox-associated cardiac injury.
By the numbers
According to the latest data, California has reported 3,629 cases, followed closely by New York with 3,367 cases, Florida with 1,957 cases, Texas with 1,698, Georgia with 1,418, and Illinois with 1,081. The other states have reported fewer than 600 cases.
The CDC says that globally, more than 52,000 monkeypox cases have been reported.
Monkeypox case counts appear to be slowing in the United States and globally.
Last week, the World Health Organization said the number of new cases worldwide declined by 21% between Aug. 15 and 21 after increasing for 4 straight weeks.
The research had no funding. Dr. Pinho and colleagues have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Clinicians in Portugal say a 31-year-old man with confirmed monkeypox developed acute myocarditis roughly 1 week after the eruption of the characteristic skin lesions of the disease.
Ana Isabel Pinho, MD, department of cardiology, São João University Hospital Centre, Porto, Portugal, said in a news release.
“We believe that reporting this potential causal relationship can raise more awareness of the scientific community and health professionals for acute myocarditis as a possible complication associated with monkeypox and might be helpful for close monitoring of affected patients for further recognition of other complications in the future,” Dr. Pinho adds.
Dr. Pinho and colleagues describe the case in a report published in JACC: Case Reports.
Case details
The patient presented with a 5-day history of malaise, myalgias, and fever followed by the eruption of multiple swollen skin lesions on his face, hands, and genitalia.
Monkeypox was confirmed by positive polymerase chain reaction assay of a swab sample from a skin lesion.
Three days later, the patient developed chest tightness that radiated through the left arm and which awoke him during the night. He was admitted to an intensive care unit with clinical suspicion of acute myocarditis.
The patient’s initial electrocardiogram showed sinus rhythm with nonspecific ventricular repolarization abnormalities.
On chest x-ray, the cardiothoracic index was normal, with no interstitial infiltrates, pleural effusion, or masses. On transthoracic echocardigraphy, biventricular systolic function was preserved, and there was no pericardial effusion.
Routine laboratory tests revealed elevated levels of C-reactive protein, creatine phosphokinase, high-sensitivity troponin I, and brain natriuretic peptide, suggesting stress injury to the heart.
Findings on cardiac magnetic resonance were consistent with myocardial inflammation and acute myocarditis.
The patient was treated with supportive care, and he made a full clinical recovery. He was discharged after 1 week. On discharge, cardiac enzymes were within the normal range. The patient showed sustained electric and hemodynamic stability, and the skin lesions had healed.
“Through this important case study, we are developing a deeper understanding of monkeypox, viral myocarditis, and how to accurately diagnose and manage this disease,” Julia Grapsa, MD, PhD, editor-in-chief of JACC: Case Reports, commented in the news release.
“I commend the authors on this valuable clinical case during a critical time as monkeypox continues to spread globally,” Dr. Grapsa added.
The researchers say further research is needed to identify the pathologic mechanism underlying monkeypox-associated cardiac injury.
By the numbers
According to the latest data, California has reported 3,629 cases, followed closely by New York with 3,367 cases, Florida with 1,957 cases, Texas with 1,698, Georgia with 1,418, and Illinois with 1,081. The other states have reported fewer than 600 cases.
The CDC says that globally, more than 52,000 monkeypox cases have been reported.
Monkeypox case counts appear to be slowing in the United States and globally.
Last week, the World Health Organization said the number of new cases worldwide declined by 21% between Aug. 15 and 21 after increasing for 4 straight weeks.
The research had no funding. Dr. Pinho and colleagues have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Clinicians in Portugal say a 31-year-old man with confirmed monkeypox developed acute myocarditis roughly 1 week after the eruption of the characteristic skin lesions of the disease.
Ana Isabel Pinho, MD, department of cardiology, São João University Hospital Centre, Porto, Portugal, said in a news release.
“We believe that reporting this potential causal relationship can raise more awareness of the scientific community and health professionals for acute myocarditis as a possible complication associated with monkeypox and might be helpful for close monitoring of affected patients for further recognition of other complications in the future,” Dr. Pinho adds.
Dr. Pinho and colleagues describe the case in a report published in JACC: Case Reports.
Case details
The patient presented with a 5-day history of malaise, myalgias, and fever followed by the eruption of multiple swollen skin lesions on his face, hands, and genitalia.
Monkeypox was confirmed by positive polymerase chain reaction assay of a swab sample from a skin lesion.
Three days later, the patient developed chest tightness that radiated through the left arm and which awoke him during the night. He was admitted to an intensive care unit with clinical suspicion of acute myocarditis.
The patient’s initial electrocardiogram showed sinus rhythm with nonspecific ventricular repolarization abnormalities.
On chest x-ray, the cardiothoracic index was normal, with no interstitial infiltrates, pleural effusion, or masses. On transthoracic echocardigraphy, biventricular systolic function was preserved, and there was no pericardial effusion.
Routine laboratory tests revealed elevated levels of C-reactive protein, creatine phosphokinase, high-sensitivity troponin I, and brain natriuretic peptide, suggesting stress injury to the heart.
Findings on cardiac magnetic resonance were consistent with myocardial inflammation and acute myocarditis.
The patient was treated with supportive care, and he made a full clinical recovery. He was discharged after 1 week. On discharge, cardiac enzymes were within the normal range. The patient showed sustained electric and hemodynamic stability, and the skin lesions had healed.
“Through this important case study, we are developing a deeper understanding of monkeypox, viral myocarditis, and how to accurately diagnose and manage this disease,” Julia Grapsa, MD, PhD, editor-in-chief of JACC: Case Reports, commented in the news release.
“I commend the authors on this valuable clinical case during a critical time as monkeypox continues to spread globally,” Dr. Grapsa added.
The researchers say further research is needed to identify the pathologic mechanism underlying monkeypox-associated cardiac injury.
By the numbers
According to the latest data, California has reported 3,629 cases, followed closely by New York with 3,367 cases, Florida with 1,957 cases, Texas with 1,698, Georgia with 1,418, and Illinois with 1,081. The other states have reported fewer than 600 cases.
The CDC says that globally, more than 52,000 monkeypox cases have been reported.
Monkeypox case counts appear to be slowing in the United States and globally.
Last week, the World Health Organization said the number of new cases worldwide declined by 21% between Aug. 15 and 21 after increasing for 4 straight weeks.
The research had no funding. Dr. Pinho and colleagues have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Dolutegravir in pregnant patients with HIV showed more viral suppression at delivery vs. other treatments
“Dolutegravir is increasingly used in pregnancy in the United States,” Kunjal Patel, DSc, one of the investigators, said in an interview. “While its effectiveness and safety in pregnancy have been compared to efavirenz in previous studies, including three randomized trials, efavirenz isn’t really used in the United States and Europe for treatment of HIV; it is mainly used in Africa,” she said. Therefore, it was important to compare dolutegravir use in pregnancy to the other antiretroviral regimens that are listed as being preferred for use in pregnancy in the U.S., including atazanavir/ritonavir, darunavir/ritonavir, and raltegravir, and others often used in the U.S. and Europe, she said.
In the study published in the New England Journal of Medicine, Dr. Patel, of Harvard T.H. Chan School of Public Health, Boston, and colleagues analyzed data from kids enrolled in the Surveillance and Monitoring for ART Toxicities Dynamic (SMARTT) cohort. This group is part of an ongoing research project focused on evaluating ART toxicities during pregnancy in children who were exposed to HIV perinatally but not infected. It included pregnancies from 2007 until January 2020 that involved use of the ARTs listed.
The study population of 1,257 pregnancies with observed birth outcomes included 120 individuals with an initial ART of dolutegravir (DTG), 464 started on atazanavir–ritonavir (ATV/r), 185 on darunavir–ritonavir (DRV/r), 243 on oral rilpivirine (RPV), 86 on raltegravir (RAL), and 159 on elvitegravir–cobicistat (EVG/c). In approximately half of the pregnancies (51%), ART was started before conception, and the initial ART was changed in 27%.
The primary outcomes were viral suppression at delivery, and adverse birth outcomes, including preterm and very preterm birth, low and very low birth weight, and neonatal death within 14 days.
The median age of the patients at conception was 29 years, and 66% were non-Hispanic Black, representative of persons with HIV of childbearing age in the United States, the researchers noted. Overall, 96.7% of the patients who received dolutegravir showed viral suppression at delivery, compared to 90.1% for darunavir–ritonavir, 89.8% for elvitegravir–cobicistat, 89.2% for raltegravir, and 84.0% for atazanavir–ritonavir.
“We expected that dolutegravir to be similar with regards to viral suppression at delivery compared to raltegravir so were surprised that we observed less viral suppression with raltegravir compared to dolutegravir,” Dr. Patel said in an interview. “Our results may be due to the higher pill burden and lower barrier to resistance with RAL compared to dolutegravir, but we did not assess adherence or resistance in our study,” she noted.
Across ART regimens, the observed risks of preterm birth ranged from 13.6% to 17.6%, risks of low birth weight ranged from 11.9% to 16.7%, and risks of being small for gestational age ranged from 9.1% to 12.5%. For the composite of any adverse birth outcome and any severe adverse birth outcome, the observed risks ranged from 22.6% to 27.9% and 0% to 4.2%, respectively.
A total of 20 very preterm births, including 15 infants with very low birth weight, occurred across patients receiving all ART regimens, and no neonatal deaths occurred. The researchers found no apparent patterns of differences in the observed risk of adverse birth outcomes across all groups related to the timing of ART initiation in pregnancy, but the risks were greater among those who began the drugs during pregnancy compared to those who began before conception.
“Our results confirm the recommendation of DTG as “preferred” in U.S. perinatal guidelines, and provide evidence suggesting ATV/r and RAL provides lower HIV viral suppression at delivery compared to DTG, and support DRV/r as a reasonable alternative when DTG use is not feasible,” Dr. Patel said in an interview.
“With regards to next steps, we are interested in comparing the effectiveness and safety of dolutegravir-based regimens that include tenofovir alafenamide (TAF) vs. tenofovir disoproxil fumarate (TDF) in our U.S. setting,” she said.
The study findings were limited by several factors including the lack of data on predictors of preterm birth and low birth weight, such as previous preterm birth and prepregnancy body mass index, the researchers noted.
However, the results indicate that other common ARTs provide less HIV viral suppression at delivery than dolutegravir, with similar adverse birth outcomes; the results also support darunavir–ritonavir as a reasonable alternative when dolutegravir use is not feasible, as it showed the next highest level of viral suppression after dolutegravir, the researchers concluded.
Findings fill a key research gap
The current study is important given the limited data on effectiveness and outcomes in pregnancy with the use of contemporary HIV regimens in the United States, Martina L. Badell, MD, a maternal-fetal medicine specialist at Emory University, Atlanta, said in an interview.
“Pregnancy is still among exclusion criteria for most drug studies,” said Dr. Badell, who was not involved in the current study. “Dolutegravir-based ART is first line in the U.S. today because of its effectiveness, lower side effects, and higher barrier to resistance; therefore understanding the benefits and birth outcomes in pregnancy is critical,” she explained.
Dr. Badell said she was not surprised by the study findings. “However it is very reassuring to see in a large observational study comparing the dolutegravir regimens to other contemporary regimens in pregnancy, such a high level of viral suppression and no increased risk of adverse perinatal outcomes,” she said.
The study findings will impact clinical practice by reaffirming patient counseling regarding the use of dolutegravir in pregnancy, said Dr. Badell. “The use of ART in pregnancy is complex given the number of drug choices, whether the patient was on ART prior to pregnancy or initiated during pregnancy, and the various factors other than ART that affect perinatal outcomes, such as preterm birth and congenital anomalies, she explained.
The finding that the risk of adverse outcomes was higher for those who initiated ART during pregnancy vs. those who were already on ARTs when they became pregnant contradicts some previous research, said Dr. Badell. But this is “reassuring, as we highly recommend ART with viral suppression prior to pregnancy or to start as early as possible in pregnancy.”
Adverse birth outcomes can be affected by many variables such as age, substance abuse, prior adverse birth outcome and other factors, and larger studies that control for these variables will allow better evaluation of the effect of the ART drugs, Dr. Badell added.
The study was funded by the Eunice Kennedy Shriver National Institute of Child Health and Human Development, along with the Office of the Director, National Institutes of Health; National Institute of Dental and Craniofacial Research; National Institute of Allergy and Infectious Diseases; National Institute of Neurological Disorders and Stroke; National Institute on Deafness and Other Communication Disorders; National Institute of Mental Health; National Institute on Drug Abuse; National Cancer Institute; National Institute on Alcohol Abuse and Alcoholism; and National Heart, Lung, and Blood Institute through cooperative agreements with the Harvard T.H. Chan School of Public Health and the Tulane University School of Medicine.
The researchers and Dr. Badell had no financial conflicts to disclose.
“Dolutegravir is increasingly used in pregnancy in the United States,” Kunjal Patel, DSc, one of the investigators, said in an interview. “While its effectiveness and safety in pregnancy have been compared to efavirenz in previous studies, including three randomized trials, efavirenz isn’t really used in the United States and Europe for treatment of HIV; it is mainly used in Africa,” she said. Therefore, it was important to compare dolutegravir use in pregnancy to the other antiretroviral regimens that are listed as being preferred for use in pregnancy in the U.S., including atazanavir/ritonavir, darunavir/ritonavir, and raltegravir, and others often used in the U.S. and Europe, she said.
In the study published in the New England Journal of Medicine, Dr. Patel, of Harvard T.H. Chan School of Public Health, Boston, and colleagues analyzed data from kids enrolled in the Surveillance and Monitoring for ART Toxicities Dynamic (SMARTT) cohort. This group is part of an ongoing research project focused on evaluating ART toxicities during pregnancy in children who were exposed to HIV perinatally but not infected. It included pregnancies from 2007 until January 2020 that involved use of the ARTs listed.
The study population of 1,257 pregnancies with observed birth outcomes included 120 individuals with an initial ART of dolutegravir (DTG), 464 started on atazanavir–ritonavir (ATV/r), 185 on darunavir–ritonavir (DRV/r), 243 on oral rilpivirine (RPV), 86 on raltegravir (RAL), and 159 on elvitegravir–cobicistat (EVG/c). In approximately half of the pregnancies (51%), ART was started before conception, and the initial ART was changed in 27%.
The primary outcomes were viral suppression at delivery, and adverse birth outcomes, including preterm and very preterm birth, low and very low birth weight, and neonatal death within 14 days.
The median age of the patients at conception was 29 years, and 66% were non-Hispanic Black, representative of persons with HIV of childbearing age in the United States, the researchers noted. Overall, 96.7% of the patients who received dolutegravir showed viral suppression at delivery, compared to 90.1% for darunavir–ritonavir, 89.8% for elvitegravir–cobicistat, 89.2% for raltegravir, and 84.0% for atazanavir–ritonavir.
“We expected that dolutegravir to be similar with regards to viral suppression at delivery compared to raltegravir so were surprised that we observed less viral suppression with raltegravir compared to dolutegravir,” Dr. Patel said in an interview. “Our results may be due to the higher pill burden and lower barrier to resistance with RAL compared to dolutegravir, but we did not assess adherence or resistance in our study,” she noted.
Across ART regimens, the observed risks of preterm birth ranged from 13.6% to 17.6%, risks of low birth weight ranged from 11.9% to 16.7%, and risks of being small for gestational age ranged from 9.1% to 12.5%. For the composite of any adverse birth outcome and any severe adverse birth outcome, the observed risks ranged from 22.6% to 27.9% and 0% to 4.2%, respectively.
A total of 20 very preterm births, including 15 infants with very low birth weight, occurred across patients receiving all ART regimens, and no neonatal deaths occurred. The researchers found no apparent patterns of differences in the observed risk of adverse birth outcomes across all groups related to the timing of ART initiation in pregnancy, but the risks were greater among those who began the drugs during pregnancy compared to those who began before conception.
“Our results confirm the recommendation of DTG as “preferred” in U.S. perinatal guidelines, and provide evidence suggesting ATV/r and RAL provides lower HIV viral suppression at delivery compared to DTG, and support DRV/r as a reasonable alternative when DTG use is not feasible,” Dr. Patel said in an interview.
“With regards to next steps, we are interested in comparing the effectiveness and safety of dolutegravir-based regimens that include tenofovir alafenamide (TAF) vs. tenofovir disoproxil fumarate (TDF) in our U.S. setting,” she said.
The study findings were limited by several factors including the lack of data on predictors of preterm birth and low birth weight, such as previous preterm birth and prepregnancy body mass index, the researchers noted.
However, the results indicate that other common ARTs provide less HIV viral suppression at delivery than dolutegravir, with similar adverse birth outcomes; the results also support darunavir–ritonavir as a reasonable alternative when dolutegravir use is not feasible, as it showed the next highest level of viral suppression after dolutegravir, the researchers concluded.
Findings fill a key research gap
The current study is important given the limited data on effectiveness and outcomes in pregnancy with the use of contemporary HIV regimens in the United States, Martina L. Badell, MD, a maternal-fetal medicine specialist at Emory University, Atlanta, said in an interview.
“Pregnancy is still among exclusion criteria for most drug studies,” said Dr. Badell, who was not involved in the current study. “Dolutegravir-based ART is first line in the U.S. today because of its effectiveness, lower side effects, and higher barrier to resistance; therefore understanding the benefits and birth outcomes in pregnancy is critical,” she explained.
Dr. Badell said she was not surprised by the study findings. “However it is very reassuring to see in a large observational study comparing the dolutegravir regimens to other contemporary regimens in pregnancy, such a high level of viral suppression and no increased risk of adverse perinatal outcomes,” she said.
The study findings will impact clinical practice by reaffirming patient counseling regarding the use of dolutegravir in pregnancy, said Dr. Badell. “The use of ART in pregnancy is complex given the number of drug choices, whether the patient was on ART prior to pregnancy or initiated during pregnancy, and the various factors other than ART that affect perinatal outcomes, such as preterm birth and congenital anomalies, she explained.
The finding that the risk of adverse outcomes was higher for those who initiated ART during pregnancy vs. those who were already on ARTs when they became pregnant contradicts some previous research, said Dr. Badell. But this is “reassuring, as we highly recommend ART with viral suppression prior to pregnancy or to start as early as possible in pregnancy.”
Adverse birth outcomes can be affected by many variables such as age, substance abuse, prior adverse birth outcome and other factors, and larger studies that control for these variables will allow better evaluation of the effect of the ART drugs, Dr. Badell added.
The study was funded by the Eunice Kennedy Shriver National Institute of Child Health and Human Development, along with the Office of the Director, National Institutes of Health; National Institute of Dental and Craniofacial Research; National Institute of Allergy and Infectious Diseases; National Institute of Neurological Disorders and Stroke; National Institute on Deafness and Other Communication Disorders; National Institute of Mental Health; National Institute on Drug Abuse; National Cancer Institute; National Institute on Alcohol Abuse and Alcoholism; and National Heart, Lung, and Blood Institute through cooperative agreements with the Harvard T.H. Chan School of Public Health and the Tulane University School of Medicine.
The researchers and Dr. Badell had no financial conflicts to disclose.
“Dolutegravir is increasingly used in pregnancy in the United States,” Kunjal Patel, DSc, one of the investigators, said in an interview. “While its effectiveness and safety in pregnancy have been compared to efavirenz in previous studies, including three randomized trials, efavirenz isn’t really used in the United States and Europe for treatment of HIV; it is mainly used in Africa,” she said. Therefore, it was important to compare dolutegravir use in pregnancy to the other antiretroviral regimens that are listed as being preferred for use in pregnancy in the U.S., including atazanavir/ritonavir, darunavir/ritonavir, and raltegravir, and others often used in the U.S. and Europe, she said.
In the study published in the New England Journal of Medicine, Dr. Patel, of Harvard T.H. Chan School of Public Health, Boston, and colleagues analyzed data from kids enrolled in the Surveillance and Monitoring for ART Toxicities Dynamic (SMARTT) cohort. This group is part of an ongoing research project focused on evaluating ART toxicities during pregnancy in children who were exposed to HIV perinatally but not infected. It included pregnancies from 2007 until January 2020 that involved use of the ARTs listed.
The study population of 1,257 pregnancies with observed birth outcomes included 120 individuals with an initial ART of dolutegravir (DTG), 464 started on atazanavir–ritonavir (ATV/r), 185 on darunavir–ritonavir (DRV/r), 243 on oral rilpivirine (RPV), 86 on raltegravir (RAL), and 159 on elvitegravir–cobicistat (EVG/c). In approximately half of the pregnancies (51%), ART was started before conception, and the initial ART was changed in 27%.
The primary outcomes were viral suppression at delivery, and adverse birth outcomes, including preterm and very preterm birth, low and very low birth weight, and neonatal death within 14 days.
The median age of the patients at conception was 29 years, and 66% were non-Hispanic Black, representative of persons with HIV of childbearing age in the United States, the researchers noted. Overall, 96.7% of the patients who received dolutegravir showed viral suppression at delivery, compared to 90.1% for darunavir–ritonavir, 89.8% for elvitegravir–cobicistat, 89.2% for raltegravir, and 84.0% for atazanavir–ritonavir.
“We expected that dolutegravir to be similar with regards to viral suppression at delivery compared to raltegravir so were surprised that we observed less viral suppression with raltegravir compared to dolutegravir,” Dr. Patel said in an interview. “Our results may be due to the higher pill burden and lower barrier to resistance with RAL compared to dolutegravir, but we did not assess adherence or resistance in our study,” she noted.
Across ART regimens, the observed risks of preterm birth ranged from 13.6% to 17.6%, risks of low birth weight ranged from 11.9% to 16.7%, and risks of being small for gestational age ranged from 9.1% to 12.5%. For the composite of any adverse birth outcome and any severe adverse birth outcome, the observed risks ranged from 22.6% to 27.9% and 0% to 4.2%, respectively.
A total of 20 very preterm births, including 15 infants with very low birth weight, occurred across patients receiving all ART regimens, and no neonatal deaths occurred. The researchers found no apparent patterns of differences in the observed risk of adverse birth outcomes across all groups related to the timing of ART initiation in pregnancy, but the risks were greater among those who began the drugs during pregnancy compared to those who began before conception.
“Our results confirm the recommendation of DTG as “preferred” in U.S. perinatal guidelines, and provide evidence suggesting ATV/r and RAL provides lower HIV viral suppression at delivery compared to DTG, and support DRV/r as a reasonable alternative when DTG use is not feasible,” Dr. Patel said in an interview.
“With regards to next steps, we are interested in comparing the effectiveness and safety of dolutegravir-based regimens that include tenofovir alafenamide (TAF) vs. tenofovir disoproxil fumarate (TDF) in our U.S. setting,” she said.
The study findings were limited by several factors including the lack of data on predictors of preterm birth and low birth weight, such as previous preterm birth and prepregnancy body mass index, the researchers noted.
However, the results indicate that other common ARTs provide less HIV viral suppression at delivery than dolutegravir, with similar adverse birth outcomes; the results also support darunavir–ritonavir as a reasonable alternative when dolutegravir use is not feasible, as it showed the next highest level of viral suppression after dolutegravir, the researchers concluded.
Findings fill a key research gap
The current study is important given the limited data on effectiveness and outcomes in pregnancy with the use of contemporary HIV regimens in the United States, Martina L. Badell, MD, a maternal-fetal medicine specialist at Emory University, Atlanta, said in an interview.
“Pregnancy is still among exclusion criteria for most drug studies,” said Dr. Badell, who was not involved in the current study. “Dolutegravir-based ART is first line in the U.S. today because of its effectiveness, lower side effects, and higher barrier to resistance; therefore understanding the benefits and birth outcomes in pregnancy is critical,” she explained.
Dr. Badell said she was not surprised by the study findings. “However it is very reassuring to see in a large observational study comparing the dolutegravir regimens to other contemporary regimens in pregnancy, such a high level of viral suppression and no increased risk of adverse perinatal outcomes,” she said.
The study findings will impact clinical practice by reaffirming patient counseling regarding the use of dolutegravir in pregnancy, said Dr. Badell. “The use of ART in pregnancy is complex given the number of drug choices, whether the patient was on ART prior to pregnancy or initiated during pregnancy, and the various factors other than ART that affect perinatal outcomes, such as preterm birth and congenital anomalies, she explained.
The finding that the risk of adverse outcomes was higher for those who initiated ART during pregnancy vs. those who were already on ARTs when they became pregnant contradicts some previous research, said Dr. Badell. But this is “reassuring, as we highly recommend ART with viral suppression prior to pregnancy or to start as early as possible in pregnancy.”
Adverse birth outcomes can be affected by many variables such as age, substance abuse, prior adverse birth outcome and other factors, and larger studies that control for these variables will allow better evaluation of the effect of the ART drugs, Dr. Badell added.
The study was funded by the Eunice Kennedy Shriver National Institute of Child Health and Human Development, along with the Office of the Director, National Institutes of Health; National Institute of Dental and Craniofacial Research; National Institute of Allergy and Infectious Diseases; National Institute of Neurological Disorders and Stroke; National Institute on Deafness and Other Communication Disorders; National Institute of Mental Health; National Institute on Drug Abuse; National Cancer Institute; National Institute on Alcohol Abuse and Alcoholism; and National Heart, Lung, and Blood Institute through cooperative agreements with the Harvard T.H. Chan School of Public Health and the Tulane University School of Medicine.
The researchers and Dr. Badell had no financial conflicts to disclose.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Consensus document aids schistosomiasis management
After malaria, human schistosomiasis is the parasitic disease with the highest morbidity and mortality worldwide. An estimated 236 million people are infected. Most are in sub-Saharan Africa. Complications lead to the deaths of 300,000 people each year. Pilot studies point to a high rate of underdiagnosis, whether in the sub-Saharan immigrant population residing in Spain or among individuals affected by outbreaks of autochthonous transmission (as happened in the 2003 case of four Spanish farmers who bathed in an artificial irrigation pool in Almería).
The “Consensus Document for the Management of Schistosomiasis in Primary Care” was recently published in the journal Atención Primaria [Primary Care]. Its aim is to establish clear recommendations so that primary care clinicians will be able to diagnose, manage, and treat this disease. The document was prepared by professionals who belong to the following five scientific societies: the Spanish Society of Family and Community Medicine, the Spanish Society of General Practitioners and Family Doctors, the Spanish Society of Primary Care Physicians, the Spanish Society for Pediatric Infectious Diseases, and the Spanish Society of Tropical Medicine and International Health (SEMTSI).
Agustín Benito Llanes, PhD, is the director of Spain’s National Center for Tropical Medicine (Carlos III Institute of Health) and the president of the SEMTSI. He told Univadis Spain, “The consensus document is invaluable for the management of cases imported by migrant populations coming from endemic areas and in the prevention of possible outbreaks in our country, especially urinary schistosomiasis.” He went on to explain, “This diagnostic strategy, which is also recommended by the European Centre for Disease Prevention and Control (ECDC), must be viewed in the context of the general management of patients with imported eosinophilia – eosinophilia being a condition that indicates that the individual may have a disease caused by a parasitic worm. I do know that primary care has been greatly affected and impacted by the pandemic, but new e-consultation and telemedicine models are making it possible for hospital specialists and primary care specialists to quickly get in touch with each other and work closely together. This technology can play a critical role in the shared care of patients with these types of diseases.”
The document recommends that serologic screening for schistosomiasis be considered for the following patients: asymptomatic individuals who have come from endemic regions and were exposed to freshwater sources; those who present with symptoms consistent with those of the disease; and patients for whom clinical exams or lab tests suggest acute schistosomiasis (eosinophilia is usually a sign). Screening for chronic schistosomiasis is indicated if the necessary resources for diagnosis and treatment are not available. The following considerations support screening asymptomatic individuals: the high prevalence of parasitic infection among migrants from endemic regions and among people who have traveled to those places; and the possibility of preventing serious complications and secondary transmissions.
The working group recommends that all at-risk individuals undergo screening, no matter how long it’s been since they were last in an endemic zone. This is because the parasites can live for over a decade. If primary care physicians don’t have access to diagnostic tests or to treatments, patients should be referred to specialists with experience in tropical diseases. A definitive diagnosis is made through the detection of blood fluke eggs in urine, stool, or body tissues. Through such detection, the species responsible for infection can be identified.
Primary care difficulties
To prevent and control the disease, the European health authorities recommend serologic screening of at-risk population groups. Because primary care is usually the first point of contact with the health care system for these infected patients, primary care physicians must know the main characteristics of schistosomiasis and be provided with the necessary means for its diagnosis and treatment. Yet physicians in health care centers face significant limitations when it comes to identifying and treating these patients.
Joaquín Salas, MD, director of the Tropical Medicine Unit at Poniente Hospital in El Ejido (Almería) and the document’s first author, explained these difficulties. “In Spain, we currently have the problem where the care of migrant patients varies greatly between the different autonomous communities – and even within an autonomous community, depending on geographical areas. This variability is caused, in large part, by the number of migrants that they serve. In places that have a large sub-Saharan migrant population, there are health centers that have gotten to a point where they’re able to request serologic testing for schistosomiasis. Unfortunately, in many instances, this testing is still only available to specialists. The objective of documents like ours is to make not only physicians but also managers aware of the importance of diagnosing and treating this disease as early as possible. Raising awareness is complicated, to a large extent, by the lack of knowledge about this disease – something that’s seen with many other ‘neglected diseases’ which primarily affect the poorest people in poor countries.”
Dr. Llanes explained that an autonomous community can individually approve serologic screenings and incorporate them into its primary care programs, regardless of whether they can be approved at the state level. He pointed out that this is what happened with Chagas disease. “To prevent vertical transmission, a protocol for pregnant women was implemented by several communities; it’s about to be approved on a national level.”
But there’s another obstacle to treating schistosomiasis. At the moment, the recommended antiparasitic treatments (e.g., praziquantel) are considered foreign medications. This makes it difficult for primary care physicians and specialists to have access to them. Even so, Dr. Salas believes “that in some places, pharmacy units facilitate things in such a way that physicians who prescribe those treatments are able to obtain them quite quickly and with less red tape. Be that as it may, ideally, the medication would be available in our country’s pharmacies, and it could be prescribed without these kinds of bureaucratic obstacles. The same thing happened with ivermectin, which is used, among other things, to treat strongyloidiasis, and which, for a few months now, has been on the market without restrictions. We hope the same will happen soon with praziquantel.”
Increasing risk
Although schistosomiasis is not endemic to Spain, various factors are contributing to an increase in the number of cases within its borders. Dr. Salas said that “without a doubt, climate change and global warming are influencing the expansion of vectors – mosquitoes, ticks, snails – that can transmit, to places like Europe, diseases referred to as ‘tropical.’ In the case of schistosomiasis, it’s been shown that Bulinus snails, intermediate hosts for Schistosoma, have adapted perfectly to Almería here in Spain and to the French island of Corsica, where winters are more and more temperate. But not only is this adaptation due to climate change, those same snails have acquired specific capabilities that allow them to better tolerate temperatures lower than those they initially had in the areas where they’re from in sub-Saharan Africa. To sum up, the colonization of new territories is due as much to a change in the climate – temperatures gradually rising – as to adaptations of the vectors themselves.”
Dr. Llanes noted that “the case involving the farmers in Almería shows that the vectors in Europe can, in fact, transmit the disease, basically because Schistosoma haematobium, human, can develop hybrids with schistosomes of cattle origin, Schistosoma bovis – hybrids that can be transmitted through European snails. This is what happened with the outbreak in Corsica, its vector being the freshwater snail Planorbarius metidjensis – as I said, shown to be implicated in Corsica’s significant outbreak, to which subsequent cases are still being traced. Obviously, the effects of climate change – temperatures rising and extreme meteorological phenomena increasing – on the infections transmitted by vectors are of the utmost importance and, together with the process of globalization, are what makes us consider these types of conditions to be emerging diseases or emerging infections.”
This article was translated from Univadis Spain. A version appeared on Medscape.com.
After malaria, human schistosomiasis is the parasitic disease with the highest morbidity and mortality worldwide. An estimated 236 million people are infected. Most are in sub-Saharan Africa. Complications lead to the deaths of 300,000 people each year. Pilot studies point to a high rate of underdiagnosis, whether in the sub-Saharan immigrant population residing in Spain or among individuals affected by outbreaks of autochthonous transmission (as happened in the 2003 case of four Spanish farmers who bathed in an artificial irrigation pool in Almería).
The “Consensus Document for the Management of Schistosomiasis in Primary Care” was recently published in the journal Atención Primaria [Primary Care]. Its aim is to establish clear recommendations so that primary care clinicians will be able to diagnose, manage, and treat this disease. The document was prepared by professionals who belong to the following five scientific societies: the Spanish Society of Family and Community Medicine, the Spanish Society of General Practitioners and Family Doctors, the Spanish Society of Primary Care Physicians, the Spanish Society for Pediatric Infectious Diseases, and the Spanish Society of Tropical Medicine and International Health (SEMTSI).
Agustín Benito Llanes, PhD, is the director of Spain’s National Center for Tropical Medicine (Carlos III Institute of Health) and the president of the SEMTSI. He told Univadis Spain, “The consensus document is invaluable for the management of cases imported by migrant populations coming from endemic areas and in the prevention of possible outbreaks in our country, especially urinary schistosomiasis.” He went on to explain, “This diagnostic strategy, which is also recommended by the European Centre for Disease Prevention and Control (ECDC), must be viewed in the context of the general management of patients with imported eosinophilia – eosinophilia being a condition that indicates that the individual may have a disease caused by a parasitic worm. I do know that primary care has been greatly affected and impacted by the pandemic, but new e-consultation and telemedicine models are making it possible for hospital specialists and primary care specialists to quickly get in touch with each other and work closely together. This technology can play a critical role in the shared care of patients with these types of diseases.”
The document recommends that serologic screening for schistosomiasis be considered for the following patients: asymptomatic individuals who have come from endemic regions and were exposed to freshwater sources; those who present with symptoms consistent with those of the disease; and patients for whom clinical exams or lab tests suggest acute schistosomiasis (eosinophilia is usually a sign). Screening for chronic schistosomiasis is indicated if the necessary resources for diagnosis and treatment are not available. The following considerations support screening asymptomatic individuals: the high prevalence of parasitic infection among migrants from endemic regions and among people who have traveled to those places; and the possibility of preventing serious complications and secondary transmissions.
The working group recommends that all at-risk individuals undergo screening, no matter how long it’s been since they were last in an endemic zone. This is because the parasites can live for over a decade. If primary care physicians don’t have access to diagnostic tests or to treatments, patients should be referred to specialists with experience in tropical diseases. A definitive diagnosis is made through the detection of blood fluke eggs in urine, stool, or body tissues. Through such detection, the species responsible for infection can be identified.
Primary care difficulties
To prevent and control the disease, the European health authorities recommend serologic screening of at-risk population groups. Because primary care is usually the first point of contact with the health care system for these infected patients, primary care physicians must know the main characteristics of schistosomiasis and be provided with the necessary means for its diagnosis and treatment. Yet physicians in health care centers face significant limitations when it comes to identifying and treating these patients.
Joaquín Salas, MD, director of the Tropical Medicine Unit at Poniente Hospital in El Ejido (Almería) and the document’s first author, explained these difficulties. “In Spain, we currently have the problem where the care of migrant patients varies greatly between the different autonomous communities – and even within an autonomous community, depending on geographical areas. This variability is caused, in large part, by the number of migrants that they serve. In places that have a large sub-Saharan migrant population, there are health centers that have gotten to a point where they’re able to request serologic testing for schistosomiasis. Unfortunately, in many instances, this testing is still only available to specialists. The objective of documents like ours is to make not only physicians but also managers aware of the importance of diagnosing and treating this disease as early as possible. Raising awareness is complicated, to a large extent, by the lack of knowledge about this disease – something that’s seen with many other ‘neglected diseases’ which primarily affect the poorest people in poor countries.”
Dr. Llanes explained that an autonomous community can individually approve serologic screenings and incorporate them into its primary care programs, regardless of whether they can be approved at the state level. He pointed out that this is what happened with Chagas disease. “To prevent vertical transmission, a protocol for pregnant women was implemented by several communities; it’s about to be approved on a national level.”
But there’s another obstacle to treating schistosomiasis. At the moment, the recommended antiparasitic treatments (e.g., praziquantel) are considered foreign medications. This makes it difficult for primary care physicians and specialists to have access to them. Even so, Dr. Salas believes “that in some places, pharmacy units facilitate things in such a way that physicians who prescribe those treatments are able to obtain them quite quickly and with less red tape. Be that as it may, ideally, the medication would be available in our country’s pharmacies, and it could be prescribed without these kinds of bureaucratic obstacles. The same thing happened with ivermectin, which is used, among other things, to treat strongyloidiasis, and which, for a few months now, has been on the market without restrictions. We hope the same will happen soon with praziquantel.”
Increasing risk
Although schistosomiasis is not endemic to Spain, various factors are contributing to an increase in the number of cases within its borders. Dr. Salas said that “without a doubt, climate change and global warming are influencing the expansion of vectors – mosquitoes, ticks, snails – that can transmit, to places like Europe, diseases referred to as ‘tropical.’ In the case of schistosomiasis, it’s been shown that Bulinus snails, intermediate hosts for Schistosoma, have adapted perfectly to Almería here in Spain and to the French island of Corsica, where winters are more and more temperate. But not only is this adaptation due to climate change, those same snails have acquired specific capabilities that allow them to better tolerate temperatures lower than those they initially had in the areas where they’re from in sub-Saharan Africa. To sum up, the colonization of new territories is due as much to a change in the climate – temperatures gradually rising – as to adaptations of the vectors themselves.”
Dr. Llanes noted that “the case involving the farmers in Almería shows that the vectors in Europe can, in fact, transmit the disease, basically because Schistosoma haematobium, human, can develop hybrids with schistosomes of cattle origin, Schistosoma bovis – hybrids that can be transmitted through European snails. This is what happened with the outbreak in Corsica, its vector being the freshwater snail Planorbarius metidjensis – as I said, shown to be implicated in Corsica’s significant outbreak, to which subsequent cases are still being traced. Obviously, the effects of climate change – temperatures rising and extreme meteorological phenomena increasing – on the infections transmitted by vectors are of the utmost importance and, together with the process of globalization, are what makes us consider these types of conditions to be emerging diseases or emerging infections.”
This article was translated from Univadis Spain. A version appeared on Medscape.com.
After malaria, human schistosomiasis is the parasitic disease with the highest morbidity and mortality worldwide. An estimated 236 million people are infected. Most are in sub-Saharan Africa. Complications lead to the deaths of 300,000 people each year. Pilot studies point to a high rate of underdiagnosis, whether in the sub-Saharan immigrant population residing in Spain or among individuals affected by outbreaks of autochthonous transmission (as happened in the 2003 case of four Spanish farmers who bathed in an artificial irrigation pool in Almería).
The “Consensus Document for the Management of Schistosomiasis in Primary Care” was recently published in the journal Atención Primaria [Primary Care]. Its aim is to establish clear recommendations so that primary care clinicians will be able to diagnose, manage, and treat this disease. The document was prepared by professionals who belong to the following five scientific societies: the Spanish Society of Family and Community Medicine, the Spanish Society of General Practitioners and Family Doctors, the Spanish Society of Primary Care Physicians, the Spanish Society for Pediatric Infectious Diseases, and the Spanish Society of Tropical Medicine and International Health (SEMTSI).
Agustín Benito Llanes, PhD, is the director of Spain’s National Center for Tropical Medicine (Carlos III Institute of Health) and the president of the SEMTSI. He told Univadis Spain, “The consensus document is invaluable for the management of cases imported by migrant populations coming from endemic areas and in the prevention of possible outbreaks in our country, especially urinary schistosomiasis.” He went on to explain, “This diagnostic strategy, which is also recommended by the European Centre for Disease Prevention and Control (ECDC), must be viewed in the context of the general management of patients with imported eosinophilia – eosinophilia being a condition that indicates that the individual may have a disease caused by a parasitic worm. I do know that primary care has been greatly affected and impacted by the pandemic, but new e-consultation and telemedicine models are making it possible for hospital specialists and primary care specialists to quickly get in touch with each other and work closely together. This technology can play a critical role in the shared care of patients with these types of diseases.”
The document recommends that serologic screening for schistosomiasis be considered for the following patients: asymptomatic individuals who have come from endemic regions and were exposed to freshwater sources; those who present with symptoms consistent with those of the disease; and patients for whom clinical exams or lab tests suggest acute schistosomiasis (eosinophilia is usually a sign). Screening for chronic schistosomiasis is indicated if the necessary resources for diagnosis and treatment are not available. The following considerations support screening asymptomatic individuals: the high prevalence of parasitic infection among migrants from endemic regions and among people who have traveled to those places; and the possibility of preventing serious complications and secondary transmissions.
The working group recommends that all at-risk individuals undergo screening, no matter how long it’s been since they were last in an endemic zone. This is because the parasites can live for over a decade. If primary care physicians don’t have access to diagnostic tests or to treatments, patients should be referred to specialists with experience in tropical diseases. A definitive diagnosis is made through the detection of blood fluke eggs in urine, stool, or body tissues. Through such detection, the species responsible for infection can be identified.
Primary care difficulties
To prevent and control the disease, the European health authorities recommend serologic screening of at-risk population groups. Because primary care is usually the first point of contact with the health care system for these infected patients, primary care physicians must know the main characteristics of schistosomiasis and be provided with the necessary means for its diagnosis and treatment. Yet physicians in health care centers face significant limitations when it comes to identifying and treating these patients.
Joaquín Salas, MD, director of the Tropical Medicine Unit at Poniente Hospital in El Ejido (Almería) and the document’s first author, explained these difficulties. “In Spain, we currently have the problem where the care of migrant patients varies greatly between the different autonomous communities – and even within an autonomous community, depending on geographical areas. This variability is caused, in large part, by the number of migrants that they serve. In places that have a large sub-Saharan migrant population, there are health centers that have gotten to a point where they’re able to request serologic testing for schistosomiasis. Unfortunately, in many instances, this testing is still only available to specialists. The objective of documents like ours is to make not only physicians but also managers aware of the importance of diagnosing and treating this disease as early as possible. Raising awareness is complicated, to a large extent, by the lack of knowledge about this disease – something that’s seen with many other ‘neglected diseases’ which primarily affect the poorest people in poor countries.”
Dr. Llanes explained that an autonomous community can individually approve serologic screenings and incorporate them into its primary care programs, regardless of whether they can be approved at the state level. He pointed out that this is what happened with Chagas disease. “To prevent vertical transmission, a protocol for pregnant women was implemented by several communities; it’s about to be approved on a national level.”
But there’s another obstacle to treating schistosomiasis. At the moment, the recommended antiparasitic treatments (e.g., praziquantel) are considered foreign medications. This makes it difficult for primary care physicians and specialists to have access to them. Even so, Dr. Salas believes “that in some places, pharmacy units facilitate things in such a way that physicians who prescribe those treatments are able to obtain them quite quickly and with less red tape. Be that as it may, ideally, the medication would be available in our country’s pharmacies, and it could be prescribed without these kinds of bureaucratic obstacles. The same thing happened with ivermectin, which is used, among other things, to treat strongyloidiasis, and which, for a few months now, has been on the market without restrictions. We hope the same will happen soon with praziquantel.”
Increasing risk
Although schistosomiasis is not endemic to Spain, various factors are contributing to an increase in the number of cases within its borders. Dr. Salas said that “without a doubt, climate change and global warming are influencing the expansion of vectors – mosquitoes, ticks, snails – that can transmit, to places like Europe, diseases referred to as ‘tropical.’ In the case of schistosomiasis, it’s been shown that Bulinus snails, intermediate hosts for Schistosoma, have adapted perfectly to Almería here in Spain and to the French island of Corsica, where winters are more and more temperate. But not only is this adaptation due to climate change, those same snails have acquired specific capabilities that allow them to better tolerate temperatures lower than those they initially had in the areas where they’re from in sub-Saharan Africa. To sum up, the colonization of new territories is due as much to a change in the climate – temperatures gradually rising – as to adaptations of the vectors themselves.”
Dr. Llanes noted that “the case involving the farmers in Almería shows that the vectors in Europe can, in fact, transmit the disease, basically because Schistosoma haematobium, human, can develop hybrids with schistosomes of cattle origin, Schistosoma bovis – hybrids that can be transmitted through European snails. This is what happened with the outbreak in Corsica, its vector being the freshwater snail Planorbarius metidjensis – as I said, shown to be implicated in Corsica’s significant outbreak, to which subsequent cases are still being traced. Obviously, the effects of climate change – temperatures rising and extreme meteorological phenomena increasing – on the infections transmitted by vectors are of the utmost importance and, together with the process of globalization, are what makes us consider these types of conditions to be emerging diseases or emerging infections.”
This article was translated from Univadis Spain. A version appeared on Medscape.com.
AHA guidance on infective endocarditis with injection drug use
Prompted by the “unprecedented” increase in the occurrence of infective endocarditis (IE) cases among people who inject drugs, the American Heart Association has issued a scientific statement devoted solely to this challenging patient population.
The statement provides a more in-depth focus on the management of IE among this unique population than what has been provided in prior AHA IE-related documents.
The statement stresses that managing IE in people who inject drugs is complex and requires a unique multidisciplinary approach that includes consultation with an addiction specialist.
The statement was published online in Circulation.
Poor long-term prognosis
In the United States from 2002 to 2016, the proportion of patients hospitalized with IE related to injection drug use doubled from 8% to about 16%.
The long-term prognosis for this population is “currently dismal for this relatively young group of individuals,” writing group Chair Daniel C. DeSimone, MD, with the Mayo Clinic in Rochester, Minn., notes in a news release.
as well as addiction medicine or addiction psychiatry specialists, pharmacists, social workers, and nurse specialists.
Nurse specialists can coordinate care from the initial IE hospitalization to outpatient and community care to support substance use disorder.
“Clinical teams must recognize that substance use disorder is a treatable chronic, relapsing medical illness and many people are able to enter sustained remission, particularly when they receive effective treatments,” the writing group emphasizes.
Although not all patients with injection drug–related IE have opioid addiction, for those who do, the “best practice” is to offer buprenorphine or methadone “as soon as possible” after the patient presents to the hospital, they advise.
Antimicrobial therapy
The writing group says it’s “reasonable” to offer people with injection drug–related IE standard treatment for IE, which is 6 weeks of intravenous antibiotics. They recognize, however, that this regimen is often not feasible in this patient population and say there is growing evidence that partial intravenous therapy followed by oral antibiotic treatment to complete a total of 6 weeks is a possible option.
They also highlight the “critical” importance of preventive measures in people who inject drugs who are successfully treated for an initial bout of IE because they remain at “extremely” high risk for subsequent bouts of IE, regardless of whether injection drug use is continued.
The writing group also stresses that people with IE who inject drugs should be considered for heart valve repair or replacement surgery regardless of current drug use if they have indications for valve surgery.
“There’s no evidence that indications for valve surgery are different for people who inject drugs compared to those who don’t, however, some treatment centers don’t offer surgery, especially if the patient currently injects drugs or has had a previous valve surgery,” Dr. DeSimone says in the release.
“Those who develop infective endocarditis require complex care delivered by professionals who look beyond stigma and bias to provide optimal and equitable care,” Dr. DeSimone adds.
The writing group acknowledges that while addiction medicine and addiction psychiatry expertise are critical to managing IE in injection drug users, these specific resources are currently not widely available.
They call on health care systems to attract individuals with addiction training and support addiction medicine consultative services, particularly in centers where drug use–related IE is common and expected to continue to increase.
This AHA scientific statement was prepared by the volunteer writing group on behalf of the AHA Rheumatic Fever, Endocarditis and Kawasaki Disease Committee of the Council on Lifelong Congenital Heart Disease and Heart Health in the Young; the Council on Cardiovascular Surgery and Anesthesia; the Council on Cardiovascular and Stroke Nursing; the Council on Clinical Cardiology; and the Council on Peripheral Vascular Disease.
This research had no commercial funding. Dr. DeSimone has no relevant disclosures.
A version of this article first appeared on Medscape.com.
Prompted by the “unprecedented” increase in the occurrence of infective endocarditis (IE) cases among people who inject drugs, the American Heart Association has issued a scientific statement devoted solely to this challenging patient population.
The statement provides a more in-depth focus on the management of IE among this unique population than what has been provided in prior AHA IE-related documents.
The statement stresses that managing IE in people who inject drugs is complex and requires a unique multidisciplinary approach that includes consultation with an addiction specialist.
The statement was published online in Circulation.
Poor long-term prognosis
In the United States from 2002 to 2016, the proportion of patients hospitalized with IE related to injection drug use doubled from 8% to about 16%.
The long-term prognosis for this population is “currently dismal for this relatively young group of individuals,” writing group Chair Daniel C. DeSimone, MD, with the Mayo Clinic in Rochester, Minn., notes in a news release.
as well as addiction medicine or addiction psychiatry specialists, pharmacists, social workers, and nurse specialists.
Nurse specialists can coordinate care from the initial IE hospitalization to outpatient and community care to support substance use disorder.
“Clinical teams must recognize that substance use disorder is a treatable chronic, relapsing medical illness and many people are able to enter sustained remission, particularly when they receive effective treatments,” the writing group emphasizes.
Although not all patients with injection drug–related IE have opioid addiction, for those who do, the “best practice” is to offer buprenorphine or methadone “as soon as possible” after the patient presents to the hospital, they advise.
Antimicrobial therapy
The writing group says it’s “reasonable” to offer people with injection drug–related IE standard treatment for IE, which is 6 weeks of intravenous antibiotics. They recognize, however, that this regimen is often not feasible in this patient population and say there is growing evidence that partial intravenous therapy followed by oral antibiotic treatment to complete a total of 6 weeks is a possible option.
They also highlight the “critical” importance of preventive measures in people who inject drugs who are successfully treated for an initial bout of IE because they remain at “extremely” high risk for subsequent bouts of IE, regardless of whether injection drug use is continued.
The writing group also stresses that people with IE who inject drugs should be considered for heart valve repair or replacement surgery regardless of current drug use if they have indications for valve surgery.
“There’s no evidence that indications for valve surgery are different for people who inject drugs compared to those who don’t, however, some treatment centers don’t offer surgery, especially if the patient currently injects drugs or has had a previous valve surgery,” Dr. DeSimone says in the release.
“Those who develop infective endocarditis require complex care delivered by professionals who look beyond stigma and bias to provide optimal and equitable care,” Dr. DeSimone adds.
The writing group acknowledges that while addiction medicine and addiction psychiatry expertise are critical to managing IE in injection drug users, these specific resources are currently not widely available.
They call on health care systems to attract individuals with addiction training and support addiction medicine consultative services, particularly in centers where drug use–related IE is common and expected to continue to increase.
This AHA scientific statement was prepared by the volunteer writing group on behalf of the AHA Rheumatic Fever, Endocarditis and Kawasaki Disease Committee of the Council on Lifelong Congenital Heart Disease and Heart Health in the Young; the Council on Cardiovascular Surgery and Anesthesia; the Council on Cardiovascular and Stroke Nursing; the Council on Clinical Cardiology; and the Council on Peripheral Vascular Disease.
This research had no commercial funding. Dr. DeSimone has no relevant disclosures.
A version of this article first appeared on Medscape.com.
Prompted by the “unprecedented” increase in the occurrence of infective endocarditis (IE) cases among people who inject drugs, the American Heart Association has issued a scientific statement devoted solely to this challenging patient population.
The statement provides a more in-depth focus on the management of IE among this unique population than what has been provided in prior AHA IE-related documents.
The statement stresses that managing IE in people who inject drugs is complex and requires a unique multidisciplinary approach that includes consultation with an addiction specialist.
The statement was published online in Circulation.
Poor long-term prognosis
In the United States from 2002 to 2016, the proportion of patients hospitalized with IE related to injection drug use doubled from 8% to about 16%.
The long-term prognosis for this population is “currently dismal for this relatively young group of individuals,” writing group Chair Daniel C. DeSimone, MD, with the Mayo Clinic in Rochester, Minn., notes in a news release.
as well as addiction medicine or addiction psychiatry specialists, pharmacists, social workers, and nurse specialists.
Nurse specialists can coordinate care from the initial IE hospitalization to outpatient and community care to support substance use disorder.
“Clinical teams must recognize that substance use disorder is a treatable chronic, relapsing medical illness and many people are able to enter sustained remission, particularly when they receive effective treatments,” the writing group emphasizes.
Although not all patients with injection drug–related IE have opioid addiction, for those who do, the “best practice” is to offer buprenorphine or methadone “as soon as possible” after the patient presents to the hospital, they advise.
Antimicrobial therapy
The writing group says it’s “reasonable” to offer people with injection drug–related IE standard treatment for IE, which is 6 weeks of intravenous antibiotics. They recognize, however, that this regimen is often not feasible in this patient population and say there is growing evidence that partial intravenous therapy followed by oral antibiotic treatment to complete a total of 6 weeks is a possible option.
They also highlight the “critical” importance of preventive measures in people who inject drugs who are successfully treated for an initial bout of IE because they remain at “extremely” high risk for subsequent bouts of IE, regardless of whether injection drug use is continued.
The writing group also stresses that people with IE who inject drugs should be considered for heart valve repair or replacement surgery regardless of current drug use if they have indications for valve surgery.
“There’s no evidence that indications for valve surgery are different for people who inject drugs compared to those who don’t, however, some treatment centers don’t offer surgery, especially if the patient currently injects drugs or has had a previous valve surgery,” Dr. DeSimone says in the release.
“Those who develop infective endocarditis require complex care delivered by professionals who look beyond stigma and bias to provide optimal and equitable care,” Dr. DeSimone adds.
The writing group acknowledges that while addiction medicine and addiction psychiatry expertise are critical to managing IE in injection drug users, these specific resources are currently not widely available.
They call on health care systems to attract individuals with addiction training and support addiction medicine consultative services, particularly in centers where drug use–related IE is common and expected to continue to increase.
This AHA scientific statement was prepared by the volunteer writing group on behalf of the AHA Rheumatic Fever, Endocarditis and Kawasaki Disease Committee of the Council on Lifelong Congenital Heart Disease and Heart Health in the Young; the Council on Cardiovascular Surgery and Anesthesia; the Council on Cardiovascular and Stroke Nursing; the Council on Clinical Cardiology; and the Council on Peripheral Vascular Disease.
This research had no commercial funding. Dr. DeSimone has no relevant disclosures.
A version of this article first appeared on Medscape.com.
FROM CIRCULATION
Asymptomatic infections drive many epidemics, including monkeypox, polio, and COVID
Monkeypox, COVID, and polio: These three very different diseases have been dominating news cycles recently, but they share at least one common characteristic: some people can become infected – and in turn infect others – while showing no symptoms.
In 1883, the famous bacteriologist Friedrich Loeffler (1852-1915) recognized that an individual’s asymptomatic carriage of bacteria could lead to diphtheria in others.
“Typhoid Mary” is perhaps the quintessential example of asymptomatic transmission of infections leading to illness and death. At the turn of the 20th century, young Mary Mallon emigrated from Ireland to New York, where she soon became a cook for wealthy Manhattan families.
George Soper, a sanitary engineer, was hired by a stricken family to investigate. After epidemiologic study, he suspected that Mary was a carrier of Salmonella typhi, the bacterial cause of typhoid fever. He persuaded the New York Department of Health to test her – against her will – for infection. After her stool was found to test positive for Salmonella, Mary was forcibly moved to North Brother Island, where she remained largely isolated from others for the next 2 years. In 1910, she was released by a new commissioner after promising not to work as a cook.
However, working under an assumed name, Mary resumed cooking at the Sloane Hospital for Women in Manhattan. Over the next 3 months, at least 25 staff members became ill. Having been found out, Mary was again exiled to the island, where she spent the rest of her life. She died in 1938 after having infected at least 122 people, five of whom died.
COVID
Asymptomatic infections are primary drivers of COVID. Earlier in the pandemic, a meta-analysis suggested a 40% rate of asymptomatic infections, although some early reports arrived at lower estimates. A 2021 JAMA Network Open modeling study indicated a 60% rate.
Those rates are changing with the Omicron variants, of which even more cases are asymptomatic. Is this from a mutation in the virus? Some suggest that it is most likely attributable to prior vaccination resulting in boosted immunity and infections being milder. Of concern is that, although people may be asymptomatic, they still have the same viral load in their nose and can readily transmit infection.
Vincent Racaniello, PhD, a professor of virology at Columbia University in New York, told this news organization that “SARS-CoV-2 COVID is so effective at transmitting because it does this asymptomatic transmission. And so you’re out and about; you have no idea that you’re infected. You’re effectively doing what we call community transmission.”
This distinguishes SARS-CoV-2 from SARS-CoV-1. SARS-CoV-1 – which caused the SARS epidemic in 2002–2004 – had very little asymptomatic shedding. With COVID, on the other hand, “A lot of people are infected but never transmit,” Dr. Racaniello added. “I think 80% of transmissions are done by 20% of infected people because those are the ones who are shedding the most virus.”
Polio
The August case of paralytic polio in Rockland County, N.Y., is “the first case of polio reported in the United States in nearly 10 years, and only the second instance of community transmission identified in the U.S. since 1979,” a spokesperson for the Centers for Disease Control and Prevention said in an email. “Although no additional cases of polio have been reported at this time, recent wastewater findings elevate concerns that poliovirus is present in these communities, posing a risk to those who are unvaccinated.”
Poliovirus has now been found in the wastewater of New York City and three surrounding counties: Rockland, Orange, and Sullivan.
Unlike COVID, which is spread through air and respiratory secretions, polio has primarily fecal-oral transmission, meaning it is spread by people ingesting food or water contaminated with stool.
According to the World Health Organization, up to 90% of infections are unrecognized because the person has no to minimal symptoms. Symptoms are nonspecific in the remainder. Only a small proportion of those infected go on to develop paralysis.
Paul Offit, MD, a virologist and director of the Vaccine Education Center at the Children’s Hospital of Philadelphia, told this news organization that before widespread immunization, polio “caused 25,000 – 30,000 children every year to be paralyzed and 1,500 to die. Roughly 1 of every 200 children who was infected was paralyzed. We had the inactivated vaccine followed by the oral polio vaccine (OPV). The price that we paid for the OPV was that rarely it could revert to the so-called neurovirulent type, a paralytic type.”
Use of the OPV was discontinued in 2000 in the United States but is still widely used worldwide because it is inexpensive and easier to administer than injections. It appeared that we were close to completely eradicating polio, as we had smallpox, but then vaccine-derived polio virus (VDPV) started cropping up in Africa, the Middle East, and Asia. They are mainly from the type 2 virus, as is the New York case. There have been three other cases of VDPV in the United States since 2000.
Now, Dr. Offit estimates that only 1 in 2,000 of those infected become paralyzed. This is why the CDC and epidemiologists are so concerned about the Rockland patient – that one case of paralysis could represent a large pool of people who are infected with polio and are asymptomatic, continuing to shed infectious virus into the sewage.
The CDC confirmed that it began conducting wastewater testing for polio in August 2022. In their interviews for this article, Dr. Offit and Dr. Racaniello were both critical of this, stressing that it is essential to do wastewater testing nationally, since asymptomatic polio can be expected to crop up from international travelers who have received OPV.
Many countries conduct that kind of wastewater surveillance. Dr. Racaniello was particularly critical of the CDC. “We’ve been telling CDC for years, at least a decade, Why don’t you check the wastewater?,” Dr. Racaniello said, “It’s been known for many years that we should be looking to monitor the circulation of these viruses. So we are using paralysis as a sentinel to say that this virus is in the wastewater, which is just not acceptable!”
Apparently there was some concern that the public would not understand. Dr. Offit viewed it as one more piece of necessary education: “You shouldn’t be alarmed about this as long as you’re vaccinated. If you’re not vaccinated, realize that this is a risk you’re taking.”
Monkeypox
Monkeypox cases have been skyrocketing in the United States in recent weeks. More than 18,000 cases have been reported since the first case in Boston on May 19, 2022.
“Monkeypox was such a rare zoonotic disease, and the disease always historically was introduced through animal contact,” Stuart Isaacs, MD, a pox virologist at the University of Pennsylvania, said in an interview. “And then the infected person would have potential spread within the household as the most common human-to-human spread, The sexual transmission is driving a lot of this infection and potentially allowing this to efficiently spread from person to person.”
A recent study from Belgium, available only as a preprint, created concerns about potential asymptomatic transmission of monkeypox Three men had undergone testing for anogenital chlamydia and gonorrhea but showed no clinical signs of monkeypox. The same samples were later tested by polymerase chain reaction (PCR), and their viral load in anorectal swabs was similar to or slightly lower than that of symptomatic patients. While no cultures were done, the patients seroconverted by later antibody testing, confirming infection.
Via email, a CDC spokesperson noted, “At this time, CDC does not have enough data to support transmission from aerosolized virus for the ongoing monkeypox outbreak, or to assess the risks for transmission from asymptomatic people. The data supports the main source of transmission currently as close contact with someone who is infected with monkeypox.”
Dr. Isaacs agreed, saying studies of smallpox, a related orthopox virus, also suggested this.
In the United Kingdom, the Institute of Tropical Medicine is offering PCR testing for monkeypox to all patients who come for gonorrhea/chlamydia screening. Dr. Racaniello said, “I think that would be great to get a sense of who is infected. Then you could look at the results and say what fraction of people go on to develop lesions, and they give you a sense of the asymptomatic rate, which we don’t know at this point.”
Unfortunately, to be tested for monkeypox in the United States requires that the patient have a lesion. “This is part of the dropped ball of public health in the U.S.,” Dr. Racaniello said. “We’re not thinking about this. .... We need to be doing [infectivity] experiments. So then the question is, how much infectious virus do you need to transmit?”
Conclusion
We’ve seen that asymptomatic carriage of bacteria and viruses occurs readily with typhoid, COVID, diphtheria, and polio (among many other organisms, such as methicillin-resistant Staphylococcus aureus or group A strep) and is far less likely with monkeypox.
Two common denominators emerged from these interviews. The first and biggest hurdle is identifying asymptomatic carriers, which is hampered by the politicization of the CDC and funding cuts to public health. “It used to be the CDC was all about public health, and now it’s administrators, unfortunately,” said Dr. Racaniello, citing science writer Laurie Garrett, author of the influential 1994 book, “The Coming Plague”.
We don’t conduct proper surveillance, he pointed out. Wastewater surveillance has been neglected for more than a decade. It has been used for SARS-CoV-2 but is only now being initiated for polio and monkeypox. Norovirus testing would also be especially helpful in reducing foodborne outbreaks, Dr. Racaniello suggested.
The second common denominator is the need to increase the availability and uptake of vaccines. As Dr. Racaniello said about COVID, “The virus is here to stay. It’s never going to go away. It’s in humans. It’s in a lot of animals. So we’re stuck with it. We’re going to have outbreaks every year. So what do you do? Get vaccinated.” And he added, “Vaccination is the most important strategy to go on with our lives.”
Dr. Isaacs was a bit more tempered, not wanting to oversell the vaccine. He said, “The vaccine is just part of the toolkit,” which includes education, testing, isolation, and reducing risk, all of which decrease the transmission cycles.
Speaking of how antivaccine advocates had specifically targeted the Hasidic community in New York State’s Rockland County, Dr. Offit noted, “I don’t think it’s a knowledge deficit as much as a trust deficit.” He said officials should identify people in communities such as these who are trusted and have them become the influencers.
The final major hurdle to controlling these outbreaks remains global disparities in care. Monkeypox has been endemic in Nigeria for decades. It was only when it spread to Europe and America that it received attention. Polio has been actively circulating in Africa and the Middle East but is only getting attention because of the New York case.
Africa was unable to afford COVID vaccines until recently. While many in the United States are on their fourth booster, as of December 2021, more than 80% of people in Africa had not yet received a single dose, according to an article by Munyaradzi Makoni in The Lancet Respiratory Medicine.
Echoing Dr. Peter Hotez’s long-standing plea for “vaccine diplomacy,” Dr. Racaniello concluded, “My philosophy has always been we should give [vaccines] to them. I mean, we spend trillions on guns. Can’t we spend a few hundred million on vaccines? We should give away everything in terms of medicine to countries that need it, and people would like us a lot better than they do now. I think it would be such a great way of getting countries to like us. … So what if it costs a billion dollars a year? It’s a drop in the bucket for us.”
Given globalization, an infectious outbreak anywhere is a risk to all.
Dr. Racaniello and Dr. Offit report no relevant financial relationships. Dr. Isaacs receives royalties from UpToDate.
A version of this article first appeared on Medscape.com.
Monkeypox, COVID, and polio: These three very different diseases have been dominating news cycles recently, but they share at least one common characteristic: some people can become infected – and in turn infect others – while showing no symptoms.
In 1883, the famous bacteriologist Friedrich Loeffler (1852-1915) recognized that an individual’s asymptomatic carriage of bacteria could lead to diphtheria in others.
“Typhoid Mary” is perhaps the quintessential example of asymptomatic transmission of infections leading to illness and death. At the turn of the 20th century, young Mary Mallon emigrated from Ireland to New York, where she soon became a cook for wealthy Manhattan families.
George Soper, a sanitary engineer, was hired by a stricken family to investigate. After epidemiologic study, he suspected that Mary was a carrier of Salmonella typhi, the bacterial cause of typhoid fever. He persuaded the New York Department of Health to test her – against her will – for infection. After her stool was found to test positive for Salmonella, Mary was forcibly moved to North Brother Island, where she remained largely isolated from others for the next 2 years. In 1910, she was released by a new commissioner after promising not to work as a cook.
However, working under an assumed name, Mary resumed cooking at the Sloane Hospital for Women in Manhattan. Over the next 3 months, at least 25 staff members became ill. Having been found out, Mary was again exiled to the island, where she spent the rest of her life. She died in 1938 after having infected at least 122 people, five of whom died.
COVID
Asymptomatic infections are primary drivers of COVID. Earlier in the pandemic, a meta-analysis suggested a 40% rate of asymptomatic infections, although some early reports arrived at lower estimates. A 2021 JAMA Network Open modeling study indicated a 60% rate.
Those rates are changing with the Omicron variants, of which even more cases are asymptomatic. Is this from a mutation in the virus? Some suggest that it is most likely attributable to prior vaccination resulting in boosted immunity and infections being milder. Of concern is that, although people may be asymptomatic, they still have the same viral load in their nose and can readily transmit infection.
Vincent Racaniello, PhD, a professor of virology at Columbia University in New York, told this news organization that “SARS-CoV-2 COVID is so effective at transmitting because it does this asymptomatic transmission. And so you’re out and about; you have no idea that you’re infected. You’re effectively doing what we call community transmission.”
This distinguishes SARS-CoV-2 from SARS-CoV-1. SARS-CoV-1 – which caused the SARS epidemic in 2002–2004 – had very little asymptomatic shedding. With COVID, on the other hand, “A lot of people are infected but never transmit,” Dr. Racaniello added. “I think 80% of transmissions are done by 20% of infected people because those are the ones who are shedding the most virus.”
Polio
The August case of paralytic polio in Rockland County, N.Y., is “the first case of polio reported in the United States in nearly 10 years, and only the second instance of community transmission identified in the U.S. since 1979,” a spokesperson for the Centers for Disease Control and Prevention said in an email. “Although no additional cases of polio have been reported at this time, recent wastewater findings elevate concerns that poliovirus is present in these communities, posing a risk to those who are unvaccinated.”
Poliovirus has now been found in the wastewater of New York City and three surrounding counties: Rockland, Orange, and Sullivan.
Unlike COVID, which is spread through air and respiratory secretions, polio has primarily fecal-oral transmission, meaning it is spread by people ingesting food or water contaminated with stool.
According to the World Health Organization, up to 90% of infections are unrecognized because the person has no to minimal symptoms. Symptoms are nonspecific in the remainder. Only a small proportion of those infected go on to develop paralysis.
Paul Offit, MD, a virologist and director of the Vaccine Education Center at the Children’s Hospital of Philadelphia, told this news organization that before widespread immunization, polio “caused 25,000 – 30,000 children every year to be paralyzed and 1,500 to die. Roughly 1 of every 200 children who was infected was paralyzed. We had the inactivated vaccine followed by the oral polio vaccine (OPV). The price that we paid for the OPV was that rarely it could revert to the so-called neurovirulent type, a paralytic type.”
Use of the OPV was discontinued in 2000 in the United States but is still widely used worldwide because it is inexpensive and easier to administer than injections. It appeared that we were close to completely eradicating polio, as we had smallpox, but then vaccine-derived polio virus (VDPV) started cropping up in Africa, the Middle East, and Asia. They are mainly from the type 2 virus, as is the New York case. There have been three other cases of VDPV in the United States since 2000.
Now, Dr. Offit estimates that only 1 in 2,000 of those infected become paralyzed. This is why the CDC and epidemiologists are so concerned about the Rockland patient – that one case of paralysis could represent a large pool of people who are infected with polio and are asymptomatic, continuing to shed infectious virus into the sewage.
The CDC confirmed that it began conducting wastewater testing for polio in August 2022. In their interviews for this article, Dr. Offit and Dr. Racaniello were both critical of this, stressing that it is essential to do wastewater testing nationally, since asymptomatic polio can be expected to crop up from international travelers who have received OPV.
Many countries conduct that kind of wastewater surveillance. Dr. Racaniello was particularly critical of the CDC. “We’ve been telling CDC for years, at least a decade, Why don’t you check the wastewater?,” Dr. Racaniello said, “It’s been known for many years that we should be looking to monitor the circulation of these viruses. So we are using paralysis as a sentinel to say that this virus is in the wastewater, which is just not acceptable!”
Apparently there was some concern that the public would not understand. Dr. Offit viewed it as one more piece of necessary education: “You shouldn’t be alarmed about this as long as you’re vaccinated. If you’re not vaccinated, realize that this is a risk you’re taking.”
Monkeypox
Monkeypox cases have been skyrocketing in the United States in recent weeks. More than 18,000 cases have been reported since the first case in Boston on May 19, 2022.
“Monkeypox was such a rare zoonotic disease, and the disease always historically was introduced through animal contact,” Stuart Isaacs, MD, a pox virologist at the University of Pennsylvania, said in an interview. “And then the infected person would have potential spread within the household as the most common human-to-human spread, The sexual transmission is driving a lot of this infection and potentially allowing this to efficiently spread from person to person.”
A recent study from Belgium, available only as a preprint, created concerns about potential asymptomatic transmission of monkeypox Three men had undergone testing for anogenital chlamydia and gonorrhea but showed no clinical signs of monkeypox. The same samples were later tested by polymerase chain reaction (PCR), and their viral load in anorectal swabs was similar to or slightly lower than that of symptomatic patients. While no cultures were done, the patients seroconverted by later antibody testing, confirming infection.
Via email, a CDC spokesperson noted, “At this time, CDC does not have enough data to support transmission from aerosolized virus for the ongoing monkeypox outbreak, or to assess the risks for transmission from asymptomatic people. The data supports the main source of transmission currently as close contact with someone who is infected with monkeypox.”
Dr. Isaacs agreed, saying studies of smallpox, a related orthopox virus, also suggested this.
In the United Kingdom, the Institute of Tropical Medicine is offering PCR testing for monkeypox to all patients who come for gonorrhea/chlamydia screening. Dr. Racaniello said, “I think that would be great to get a sense of who is infected. Then you could look at the results and say what fraction of people go on to develop lesions, and they give you a sense of the asymptomatic rate, which we don’t know at this point.”
Unfortunately, to be tested for monkeypox in the United States requires that the patient have a lesion. “This is part of the dropped ball of public health in the U.S.,” Dr. Racaniello said. “We’re not thinking about this. .... We need to be doing [infectivity] experiments. So then the question is, how much infectious virus do you need to transmit?”
Conclusion
We’ve seen that asymptomatic carriage of bacteria and viruses occurs readily with typhoid, COVID, diphtheria, and polio (among many other organisms, such as methicillin-resistant Staphylococcus aureus or group A strep) and is far less likely with monkeypox.
Two common denominators emerged from these interviews. The first and biggest hurdle is identifying asymptomatic carriers, which is hampered by the politicization of the CDC and funding cuts to public health. “It used to be the CDC was all about public health, and now it’s administrators, unfortunately,” said Dr. Racaniello, citing science writer Laurie Garrett, author of the influential 1994 book, “The Coming Plague”.
We don’t conduct proper surveillance, he pointed out. Wastewater surveillance has been neglected for more than a decade. It has been used for SARS-CoV-2 but is only now being initiated for polio and monkeypox. Norovirus testing would also be especially helpful in reducing foodborne outbreaks, Dr. Racaniello suggested.
The second common denominator is the need to increase the availability and uptake of vaccines. As Dr. Racaniello said about COVID, “The virus is here to stay. It’s never going to go away. It’s in humans. It’s in a lot of animals. So we’re stuck with it. We’re going to have outbreaks every year. So what do you do? Get vaccinated.” And he added, “Vaccination is the most important strategy to go on with our lives.”
Dr. Isaacs was a bit more tempered, not wanting to oversell the vaccine. He said, “The vaccine is just part of the toolkit,” which includes education, testing, isolation, and reducing risk, all of which decrease the transmission cycles.
Speaking of how antivaccine advocates had specifically targeted the Hasidic community in New York State’s Rockland County, Dr. Offit noted, “I don’t think it’s a knowledge deficit as much as a trust deficit.” He said officials should identify people in communities such as these who are trusted and have them become the influencers.
The final major hurdle to controlling these outbreaks remains global disparities in care. Monkeypox has been endemic in Nigeria for decades. It was only when it spread to Europe and America that it received attention. Polio has been actively circulating in Africa and the Middle East but is only getting attention because of the New York case.
Africa was unable to afford COVID vaccines until recently. While many in the United States are on their fourth booster, as of December 2021, more than 80% of people in Africa had not yet received a single dose, according to an article by Munyaradzi Makoni in The Lancet Respiratory Medicine.
Echoing Dr. Peter Hotez’s long-standing plea for “vaccine diplomacy,” Dr. Racaniello concluded, “My philosophy has always been we should give [vaccines] to them. I mean, we spend trillions on guns. Can’t we spend a few hundred million on vaccines? We should give away everything in terms of medicine to countries that need it, and people would like us a lot better than they do now. I think it would be such a great way of getting countries to like us. … So what if it costs a billion dollars a year? It’s a drop in the bucket for us.”
Given globalization, an infectious outbreak anywhere is a risk to all.
Dr. Racaniello and Dr. Offit report no relevant financial relationships. Dr. Isaacs receives royalties from UpToDate.
A version of this article first appeared on Medscape.com.
Monkeypox, COVID, and polio: These three very different diseases have been dominating news cycles recently, but they share at least one common characteristic: some people can become infected – and in turn infect others – while showing no symptoms.
In 1883, the famous bacteriologist Friedrich Loeffler (1852-1915) recognized that an individual’s asymptomatic carriage of bacteria could lead to diphtheria in others.
“Typhoid Mary” is perhaps the quintessential example of asymptomatic transmission of infections leading to illness and death. At the turn of the 20th century, young Mary Mallon emigrated from Ireland to New York, where she soon became a cook for wealthy Manhattan families.
George Soper, a sanitary engineer, was hired by a stricken family to investigate. After epidemiologic study, he suspected that Mary was a carrier of Salmonella typhi, the bacterial cause of typhoid fever. He persuaded the New York Department of Health to test her – against her will – for infection. After her stool was found to test positive for Salmonella, Mary was forcibly moved to North Brother Island, where she remained largely isolated from others for the next 2 years. In 1910, she was released by a new commissioner after promising not to work as a cook.
However, working under an assumed name, Mary resumed cooking at the Sloane Hospital for Women in Manhattan. Over the next 3 months, at least 25 staff members became ill. Having been found out, Mary was again exiled to the island, where she spent the rest of her life. She died in 1938 after having infected at least 122 people, five of whom died.
COVID
Asymptomatic infections are primary drivers of COVID. Earlier in the pandemic, a meta-analysis suggested a 40% rate of asymptomatic infections, although some early reports arrived at lower estimates. A 2021 JAMA Network Open modeling study indicated a 60% rate.
Those rates are changing with the Omicron variants, of which even more cases are asymptomatic. Is this from a mutation in the virus? Some suggest that it is most likely attributable to prior vaccination resulting in boosted immunity and infections being milder. Of concern is that, although people may be asymptomatic, they still have the same viral load in their nose and can readily transmit infection.
Vincent Racaniello, PhD, a professor of virology at Columbia University in New York, told this news organization that “SARS-CoV-2 COVID is so effective at transmitting because it does this asymptomatic transmission. And so you’re out and about; you have no idea that you’re infected. You’re effectively doing what we call community transmission.”
This distinguishes SARS-CoV-2 from SARS-CoV-1. SARS-CoV-1 – which caused the SARS epidemic in 2002–2004 – had very little asymptomatic shedding. With COVID, on the other hand, “A lot of people are infected but never transmit,” Dr. Racaniello added. “I think 80% of transmissions are done by 20% of infected people because those are the ones who are shedding the most virus.”
Polio
The August case of paralytic polio in Rockland County, N.Y., is “the first case of polio reported in the United States in nearly 10 years, and only the second instance of community transmission identified in the U.S. since 1979,” a spokesperson for the Centers for Disease Control and Prevention said in an email. “Although no additional cases of polio have been reported at this time, recent wastewater findings elevate concerns that poliovirus is present in these communities, posing a risk to those who are unvaccinated.”
Poliovirus has now been found in the wastewater of New York City and three surrounding counties: Rockland, Orange, and Sullivan.
Unlike COVID, which is spread through air and respiratory secretions, polio has primarily fecal-oral transmission, meaning it is spread by people ingesting food or water contaminated with stool.
According to the World Health Organization, up to 90% of infections are unrecognized because the person has no to minimal symptoms. Symptoms are nonspecific in the remainder. Only a small proportion of those infected go on to develop paralysis.
Paul Offit, MD, a virologist and director of the Vaccine Education Center at the Children’s Hospital of Philadelphia, told this news organization that before widespread immunization, polio “caused 25,000 – 30,000 children every year to be paralyzed and 1,500 to die. Roughly 1 of every 200 children who was infected was paralyzed. We had the inactivated vaccine followed by the oral polio vaccine (OPV). The price that we paid for the OPV was that rarely it could revert to the so-called neurovirulent type, a paralytic type.”
Use of the OPV was discontinued in 2000 in the United States but is still widely used worldwide because it is inexpensive and easier to administer than injections. It appeared that we were close to completely eradicating polio, as we had smallpox, but then vaccine-derived polio virus (VDPV) started cropping up in Africa, the Middle East, and Asia. They are mainly from the type 2 virus, as is the New York case. There have been three other cases of VDPV in the United States since 2000.
Now, Dr. Offit estimates that only 1 in 2,000 of those infected become paralyzed. This is why the CDC and epidemiologists are so concerned about the Rockland patient – that one case of paralysis could represent a large pool of people who are infected with polio and are asymptomatic, continuing to shed infectious virus into the sewage.
The CDC confirmed that it began conducting wastewater testing for polio in August 2022. In their interviews for this article, Dr. Offit and Dr. Racaniello were both critical of this, stressing that it is essential to do wastewater testing nationally, since asymptomatic polio can be expected to crop up from international travelers who have received OPV.
Many countries conduct that kind of wastewater surveillance. Dr. Racaniello was particularly critical of the CDC. “We’ve been telling CDC for years, at least a decade, Why don’t you check the wastewater?,” Dr. Racaniello said, “It’s been known for many years that we should be looking to monitor the circulation of these viruses. So we are using paralysis as a sentinel to say that this virus is in the wastewater, which is just not acceptable!”
Apparently there was some concern that the public would not understand. Dr. Offit viewed it as one more piece of necessary education: “You shouldn’t be alarmed about this as long as you’re vaccinated. If you’re not vaccinated, realize that this is a risk you’re taking.”
Monkeypox
Monkeypox cases have been skyrocketing in the United States in recent weeks. More than 18,000 cases have been reported since the first case in Boston on May 19, 2022.
“Monkeypox was such a rare zoonotic disease, and the disease always historically was introduced through animal contact,” Stuart Isaacs, MD, a pox virologist at the University of Pennsylvania, said in an interview. “And then the infected person would have potential spread within the household as the most common human-to-human spread, The sexual transmission is driving a lot of this infection and potentially allowing this to efficiently spread from person to person.”
A recent study from Belgium, available only as a preprint, created concerns about potential asymptomatic transmission of monkeypox Three men had undergone testing for anogenital chlamydia and gonorrhea but showed no clinical signs of monkeypox. The same samples were later tested by polymerase chain reaction (PCR), and their viral load in anorectal swabs was similar to or slightly lower than that of symptomatic patients. While no cultures were done, the patients seroconverted by later antibody testing, confirming infection.
Via email, a CDC spokesperson noted, “At this time, CDC does not have enough data to support transmission from aerosolized virus for the ongoing monkeypox outbreak, or to assess the risks for transmission from asymptomatic people. The data supports the main source of transmission currently as close contact with someone who is infected with monkeypox.”
Dr. Isaacs agreed, saying studies of smallpox, a related orthopox virus, also suggested this.
In the United Kingdom, the Institute of Tropical Medicine is offering PCR testing for monkeypox to all patients who come for gonorrhea/chlamydia screening. Dr. Racaniello said, “I think that would be great to get a sense of who is infected. Then you could look at the results and say what fraction of people go on to develop lesions, and they give you a sense of the asymptomatic rate, which we don’t know at this point.”
Unfortunately, to be tested for monkeypox in the United States requires that the patient have a lesion. “This is part of the dropped ball of public health in the U.S.,” Dr. Racaniello said. “We’re not thinking about this. .... We need to be doing [infectivity] experiments. So then the question is, how much infectious virus do you need to transmit?”
Conclusion
We’ve seen that asymptomatic carriage of bacteria and viruses occurs readily with typhoid, COVID, diphtheria, and polio (among many other organisms, such as methicillin-resistant Staphylococcus aureus or group A strep) and is far less likely with monkeypox.
Two common denominators emerged from these interviews. The first and biggest hurdle is identifying asymptomatic carriers, which is hampered by the politicization of the CDC and funding cuts to public health. “It used to be the CDC was all about public health, and now it’s administrators, unfortunately,” said Dr. Racaniello, citing science writer Laurie Garrett, author of the influential 1994 book, “The Coming Plague”.
We don’t conduct proper surveillance, he pointed out. Wastewater surveillance has been neglected for more than a decade. It has been used for SARS-CoV-2 but is only now being initiated for polio and monkeypox. Norovirus testing would also be especially helpful in reducing foodborne outbreaks, Dr. Racaniello suggested.
The second common denominator is the need to increase the availability and uptake of vaccines. As Dr. Racaniello said about COVID, “The virus is here to stay. It’s never going to go away. It’s in humans. It’s in a lot of animals. So we’re stuck with it. We’re going to have outbreaks every year. So what do you do? Get vaccinated.” And he added, “Vaccination is the most important strategy to go on with our lives.”
Dr. Isaacs was a bit more tempered, not wanting to oversell the vaccine. He said, “The vaccine is just part of the toolkit,” which includes education, testing, isolation, and reducing risk, all of which decrease the transmission cycles.
Speaking of how antivaccine advocates had specifically targeted the Hasidic community in New York State’s Rockland County, Dr. Offit noted, “I don’t think it’s a knowledge deficit as much as a trust deficit.” He said officials should identify people in communities such as these who are trusted and have them become the influencers.
The final major hurdle to controlling these outbreaks remains global disparities in care. Monkeypox has been endemic in Nigeria for decades. It was only when it spread to Europe and America that it received attention. Polio has been actively circulating in Africa and the Middle East but is only getting attention because of the New York case.
Africa was unable to afford COVID vaccines until recently. While many in the United States are on their fourth booster, as of December 2021, more than 80% of people in Africa had not yet received a single dose, according to an article by Munyaradzi Makoni in The Lancet Respiratory Medicine.
Echoing Dr. Peter Hotez’s long-standing plea for “vaccine diplomacy,” Dr. Racaniello concluded, “My philosophy has always been we should give [vaccines] to them. I mean, we spend trillions on guns. Can’t we spend a few hundred million on vaccines? We should give away everything in terms of medicine to countries that need it, and people would like us a lot better than they do now. I think it would be such a great way of getting countries to like us. … So what if it costs a billion dollars a year? It’s a drop in the bucket for us.”
Given globalization, an infectious outbreak anywhere is a risk to all.
Dr. Racaniello and Dr. Offit report no relevant financial relationships. Dr. Isaacs receives royalties from UpToDate.
A version of this article first appeared on Medscape.com.
U.S. life expectancy drops to lowest in decades
according to new CDC data.
In 2021, the average American could expect to live until age 76, which fell from 77 in 2020 and 79 in 2019. That marks the lowest age since 1996 and the largest 2-year decline since 1923.
“Even small declines in life expectancy of a tenth or two-tenths of a year mean that on a population level, a lot more people are dying prematurely,” Robert Anderson, PhD, chief of mortality statistics at the National Center for Health Statistics, which produced the report, told The New York Times.
“This signals a huge impact on the population in terms of increased mortality,” he said.
COVID-19 played a major role, with excess death from the coronavirus contributing to half of the decline during the past 2 years. Drug overdose deaths also reached a record high in 2021, rising to about 109,000 people. Unintentional injuries, with about half due to drug overdose, were a leading cause of the decline in life expectancy, along with deaths from heart disease, chronic liver disease, cirrhosis, and suicide.
The decrease has been particularly devastating among Native Americans and Alaska Natives. Average life expectancy dropped by 4 years in 2020 alone and more than 6.5 years since the beginning of the pandemic. Now their life expectancy is 65, which was the average for all Americans in 1944.
“When I saw that in the report, I just – my jaw dropped,” Dr. Anderson told CNN.
“It was hard enough to fathom a 2.7-year decline over 2 years overall,” he said. “But then to see a 6.6-year decline for the American Indian population, it just shows the substantial impact that the pandemic has had on that population.”
Longstanding health issues and systemic problems, such as poverty, discrimination, and poor access to health care, led to the major declines among Native Americans and Alaska Natives, CNN reported.
“A lot of the talk is going to be around the pandemic, but we need to think about what has driven the conditions that have allowed certain communities to be more vulnerable,” Ruben Cantu, an associate program director with Prevention Institute, a nonprofit focused on health equity, told CNN.
The gap in life expectancy between women and men also became wider in 2021, growing to 5.9 years and marking the largest gap since 1996. The life expectancy for men in 2021 was 73.2, as compared with 79.1 for women.
The decline in overall U.S. life expectancy would have been even greater if there weren’t “offsetting effects,” the researchers wrote, such as declines in death due to the flu, pneumonia, chronic lower respiratory diseases, and Alzheimer’s disease.
The drop in U.S. life expectancy is “historic,” Steven Woolf, MD, retired director of the Center on Society and Health and Virginia Commonwealth University, told the Times.
Other high-income countries also saw a drop in life expectancy in 2020 due to the pandemic, but most began to recover last year due to major vaccine campaigns and behavior changes such as wearing masks, he said.
“None of them experienced a continuing fall in life expectancy like the U.S. did, and a good number of them saw life expectancy start inching back to normal,” he said. “The U.S. is clearly an outlier.”
A version of this article first appeared on WebMD.com.
according to new CDC data.
In 2021, the average American could expect to live until age 76, which fell from 77 in 2020 and 79 in 2019. That marks the lowest age since 1996 and the largest 2-year decline since 1923.
“Even small declines in life expectancy of a tenth or two-tenths of a year mean that on a population level, a lot more people are dying prematurely,” Robert Anderson, PhD, chief of mortality statistics at the National Center for Health Statistics, which produced the report, told The New York Times.
“This signals a huge impact on the population in terms of increased mortality,” he said.
COVID-19 played a major role, with excess death from the coronavirus contributing to half of the decline during the past 2 years. Drug overdose deaths also reached a record high in 2021, rising to about 109,000 people. Unintentional injuries, with about half due to drug overdose, were a leading cause of the decline in life expectancy, along with deaths from heart disease, chronic liver disease, cirrhosis, and suicide.
The decrease has been particularly devastating among Native Americans and Alaska Natives. Average life expectancy dropped by 4 years in 2020 alone and more than 6.5 years since the beginning of the pandemic. Now their life expectancy is 65, which was the average for all Americans in 1944.
“When I saw that in the report, I just – my jaw dropped,” Dr. Anderson told CNN.
“It was hard enough to fathom a 2.7-year decline over 2 years overall,” he said. “But then to see a 6.6-year decline for the American Indian population, it just shows the substantial impact that the pandemic has had on that population.”
Longstanding health issues and systemic problems, such as poverty, discrimination, and poor access to health care, led to the major declines among Native Americans and Alaska Natives, CNN reported.
“A lot of the talk is going to be around the pandemic, but we need to think about what has driven the conditions that have allowed certain communities to be more vulnerable,” Ruben Cantu, an associate program director with Prevention Institute, a nonprofit focused on health equity, told CNN.
The gap in life expectancy between women and men also became wider in 2021, growing to 5.9 years and marking the largest gap since 1996. The life expectancy for men in 2021 was 73.2, as compared with 79.1 for women.
The decline in overall U.S. life expectancy would have been even greater if there weren’t “offsetting effects,” the researchers wrote, such as declines in death due to the flu, pneumonia, chronic lower respiratory diseases, and Alzheimer’s disease.
The drop in U.S. life expectancy is “historic,” Steven Woolf, MD, retired director of the Center on Society and Health and Virginia Commonwealth University, told the Times.
Other high-income countries also saw a drop in life expectancy in 2020 due to the pandemic, but most began to recover last year due to major vaccine campaigns and behavior changes such as wearing masks, he said.
“None of them experienced a continuing fall in life expectancy like the U.S. did, and a good number of them saw life expectancy start inching back to normal,” he said. “The U.S. is clearly an outlier.”
A version of this article first appeared on WebMD.com.
according to new CDC data.
In 2021, the average American could expect to live until age 76, which fell from 77 in 2020 and 79 in 2019. That marks the lowest age since 1996 and the largest 2-year decline since 1923.
“Even small declines in life expectancy of a tenth or two-tenths of a year mean that on a population level, a lot more people are dying prematurely,” Robert Anderson, PhD, chief of mortality statistics at the National Center for Health Statistics, which produced the report, told The New York Times.
“This signals a huge impact on the population in terms of increased mortality,” he said.
COVID-19 played a major role, with excess death from the coronavirus contributing to half of the decline during the past 2 years. Drug overdose deaths also reached a record high in 2021, rising to about 109,000 people. Unintentional injuries, with about half due to drug overdose, were a leading cause of the decline in life expectancy, along with deaths from heart disease, chronic liver disease, cirrhosis, and suicide.
The decrease has been particularly devastating among Native Americans and Alaska Natives. Average life expectancy dropped by 4 years in 2020 alone and more than 6.5 years since the beginning of the pandemic. Now their life expectancy is 65, which was the average for all Americans in 1944.
“When I saw that in the report, I just – my jaw dropped,” Dr. Anderson told CNN.
“It was hard enough to fathom a 2.7-year decline over 2 years overall,” he said. “But then to see a 6.6-year decline for the American Indian population, it just shows the substantial impact that the pandemic has had on that population.”
Longstanding health issues and systemic problems, such as poverty, discrimination, and poor access to health care, led to the major declines among Native Americans and Alaska Natives, CNN reported.
“A lot of the talk is going to be around the pandemic, but we need to think about what has driven the conditions that have allowed certain communities to be more vulnerable,” Ruben Cantu, an associate program director with Prevention Institute, a nonprofit focused on health equity, told CNN.
The gap in life expectancy between women and men also became wider in 2021, growing to 5.9 years and marking the largest gap since 1996. The life expectancy for men in 2021 was 73.2, as compared with 79.1 for women.
The decline in overall U.S. life expectancy would have been even greater if there weren’t “offsetting effects,” the researchers wrote, such as declines in death due to the flu, pneumonia, chronic lower respiratory diseases, and Alzheimer’s disease.
The drop in U.S. life expectancy is “historic,” Steven Woolf, MD, retired director of the Center on Society and Health and Virginia Commonwealth University, told the Times.
Other high-income countries also saw a drop in life expectancy in 2020 due to the pandemic, but most began to recover last year due to major vaccine campaigns and behavior changes such as wearing masks, he said.
“None of them experienced a continuing fall in life expectancy like the U.S. did, and a good number of them saw life expectancy start inching back to normal,” he said. “The U.S. is clearly an outlier.”
A version of this article first appeared on WebMD.com.
Many young kids with COVID may show no symptoms
BY WILL PASS
Just 14% of adults who tested positive for SARS-CoV-2 were asymptomatic, versus 37% of children aged 0-4 years, in the paper. This raises concern that parents, childcare providers, and preschools may be underestimating infection in seemingly healthy young kids who have been exposed to COVID, wrote lead author Ruth A. Karron, MD, and colleagues in JAMA Network Open.
Methods
The new research involved 690 individuals from 175 households in Maryland who were monitored closely between November 2020 and October 2021. Every week for 8 months, participants completed online symptom checks and underwent PCR testing using nasal swabs, with symptomatic individuals submitting additional swabs for analysis.
“What was different about our study [compared with previous studies] was the intensity of our collection, and the fact that we collected specimens from asymptomatic people,” said Dr. Karron, a pediatrician and professor in the department of international health, Johns Hopkins University, Baltimore, in an interview. “You shed more virus earlier in the infection than later, and the fact that we were sampling every single week meant that we could pick up those early infections.”
The study also stands out for its focus on young children, Dr. Karron said. Enrollment required all households to have at least one child aged 0-4 years, so 256 out of 690 participants (37.1%) were in this youngest age group. The remainder of the population consisted of 100 older children aged 5-17 years (14.5%) and 334 adults aged 18-74 years (48.4%).
Children 4 and under more than twice as likely to be asymptomatic
By the end of the study, 51 participants had tested positive for SARS-CoV-2, among whom 14 had no symptoms. A closer look showed that children 0-4 years of age who contracted COVID were more than twice as likely to be asymptomatic as infected adults (36.8% vs. 14.3%).
The relationship between symptoms and viral load also differed between adults and young children.
While adults with high viral loads – suggesting greater contagiousness – typically had more severe COVID symptoms, no correlation was found in young kids, meaning children with mild or no symptoms could still be highly contagious.
Dr. Karron said these findings should help parents and other stakeholders make better-informed decisions based on known risks. She recommended testing young, asymptomatic children for COVID if they have been exposed to infected individuals, then acting accordingly based on the results.
“If a family is infected with the virus, and the 2-year-old is asymptomatic, and people are thinking about a visit to elderly grandparents who may be frail, one shouldn’t assume that the 2-year-old is uninfected,” Dr. Karron said. “That child should be tested along with other family members.”
Testing should also be considered for young children exposed to COVID at childcare facilities, she added.
But not every expert consulted for this piece shared these opinions of Dr. Karron.
“I question whether that effort is worth it,” said Dean Blumberg, MD, professor and chief of the division of pediatric infectious diseases at UC Davis Health, Sacramento, Calif.
He noted that recent Food and Drug Administration guidance for COVID testing calls for three negative at-home antigen tests to confirm lack of infection.
“That would take 4 days to get those tests done,” he said. “So, it’s a lot of testing. It’s a lot of record keeping, it’s inconvenient, it’s uncomfortable to be tested, and I just question whether it’s worth that effort.”
Applicability of findings to today questioned
Dr. Blumberg also questioned whether the study, which was completed almost a year ago, reflects the current pandemic landscape.
“At the time this study was done, it was predominantly Delta [variant instead of Omicron],” Dr. Blumberg said. “The other issue [with the study] is that … most of the children didn’t have preexisting immunity, so you have to take that into account.”
Preexisting immunity – whether from exposure or vaccination – could lower viral loads, so asymptomatic children today really could be less contagious than they were when the study was done, according to Dr. Blumberg. Kids without symptoms are also less likely to spread the virus, because they aren’t coughing or sneezing, he added.
Sara R. Kim, MD, and Janet A. Englund, MD, of the Seattle Children’s Research Institute, University of Washington, said it’s challenging to know how applicable the findings are, although they sided more with the investigators than Dr. Blumberg.
“Given the higher rate of transmissibility and infectivity of the Omicron variant, it is difficult to make direct associations between findings reported during this study period and those present in the current era during which the Omicron variant is circulating,” they wrote in an accompanying editorial. “However, the higher rates of asymptomatic infection observed among children in this study are likely to be consistent with those observed for current and future viral variants.”
Although the experts offered different interpretations of the findings, they shared similar perspectives on vaccination.
“The most important thing that parents can do is get their kids vaccinated, be vaccinated themselves, and have everybody in the household vaccinated and up to date for all doses that are indicated,” Dr. Blumberg said.
Dr. Karron noted that vaccination will be increasingly important in the coming months.
“Summer is ending; school is starting,” she said. “We’re going to be in large groups indoors again very soon. To keep young children safe, I think it’s really important for them to get vaccinated.”
The study was funded by the CDC. The investigators disclosed no other relationships. Dr. Englund disclosed relationships with AstraZeneca, GlaxoSmithKline, Merck, and others. Dr. Kim and Dr. Blumberg disclosed no relevant conflicts of interest.
BY WILL PASS
Just 14% of adults who tested positive for SARS-CoV-2 were asymptomatic, versus 37% of children aged 0-4 years, in the paper. This raises concern that parents, childcare providers, and preschools may be underestimating infection in seemingly healthy young kids who have been exposed to COVID, wrote lead author Ruth A. Karron, MD, and colleagues in JAMA Network Open.
Methods
The new research involved 690 individuals from 175 households in Maryland who were monitored closely between November 2020 and October 2021. Every week for 8 months, participants completed online symptom checks and underwent PCR testing using nasal swabs, with symptomatic individuals submitting additional swabs for analysis.
“What was different about our study [compared with previous studies] was the intensity of our collection, and the fact that we collected specimens from asymptomatic people,” said Dr. Karron, a pediatrician and professor in the department of international health, Johns Hopkins University, Baltimore, in an interview. “You shed more virus earlier in the infection than later, and the fact that we were sampling every single week meant that we could pick up those early infections.”
The study also stands out for its focus on young children, Dr. Karron said. Enrollment required all households to have at least one child aged 0-4 years, so 256 out of 690 participants (37.1%) were in this youngest age group. The remainder of the population consisted of 100 older children aged 5-17 years (14.5%) and 334 adults aged 18-74 years (48.4%).
Children 4 and under more than twice as likely to be asymptomatic
By the end of the study, 51 participants had tested positive for SARS-CoV-2, among whom 14 had no symptoms. A closer look showed that children 0-4 years of age who contracted COVID were more than twice as likely to be asymptomatic as infected adults (36.8% vs. 14.3%).
The relationship between symptoms and viral load also differed between adults and young children.
While adults with high viral loads – suggesting greater contagiousness – typically had more severe COVID symptoms, no correlation was found in young kids, meaning children with mild or no symptoms could still be highly contagious.
Dr. Karron said these findings should help parents and other stakeholders make better-informed decisions based on known risks. She recommended testing young, asymptomatic children for COVID if they have been exposed to infected individuals, then acting accordingly based on the results.
“If a family is infected with the virus, and the 2-year-old is asymptomatic, and people are thinking about a visit to elderly grandparents who may be frail, one shouldn’t assume that the 2-year-old is uninfected,” Dr. Karron said. “That child should be tested along with other family members.”
Testing should also be considered for young children exposed to COVID at childcare facilities, she added.
But not every expert consulted for this piece shared these opinions of Dr. Karron.
“I question whether that effort is worth it,” said Dean Blumberg, MD, professor and chief of the division of pediatric infectious diseases at UC Davis Health, Sacramento, Calif.
He noted that recent Food and Drug Administration guidance for COVID testing calls for three negative at-home antigen tests to confirm lack of infection.
“That would take 4 days to get those tests done,” he said. “So, it’s a lot of testing. It’s a lot of record keeping, it’s inconvenient, it’s uncomfortable to be tested, and I just question whether it’s worth that effort.”
Applicability of findings to today questioned
Dr. Blumberg also questioned whether the study, which was completed almost a year ago, reflects the current pandemic landscape.
“At the time this study was done, it was predominantly Delta [variant instead of Omicron],” Dr. Blumberg said. “The other issue [with the study] is that … most of the children didn’t have preexisting immunity, so you have to take that into account.”
Preexisting immunity – whether from exposure or vaccination – could lower viral loads, so asymptomatic children today really could be less contagious than they were when the study was done, according to Dr. Blumberg. Kids without symptoms are also less likely to spread the virus, because they aren’t coughing or sneezing, he added.
Sara R. Kim, MD, and Janet A. Englund, MD, of the Seattle Children’s Research Institute, University of Washington, said it’s challenging to know how applicable the findings are, although they sided more with the investigators than Dr. Blumberg.
“Given the higher rate of transmissibility and infectivity of the Omicron variant, it is difficult to make direct associations between findings reported during this study period and those present in the current era during which the Omicron variant is circulating,” they wrote in an accompanying editorial. “However, the higher rates of asymptomatic infection observed among children in this study are likely to be consistent with those observed for current and future viral variants.”
Although the experts offered different interpretations of the findings, they shared similar perspectives on vaccination.
“The most important thing that parents can do is get their kids vaccinated, be vaccinated themselves, and have everybody in the household vaccinated and up to date for all doses that are indicated,” Dr. Blumberg said.
Dr. Karron noted that vaccination will be increasingly important in the coming months.
“Summer is ending; school is starting,” she said. “We’re going to be in large groups indoors again very soon. To keep young children safe, I think it’s really important for them to get vaccinated.”
The study was funded by the CDC. The investigators disclosed no other relationships. Dr. Englund disclosed relationships with AstraZeneca, GlaxoSmithKline, Merck, and others. Dr. Kim and Dr. Blumberg disclosed no relevant conflicts of interest.
BY WILL PASS
Just 14% of adults who tested positive for SARS-CoV-2 were asymptomatic, versus 37% of children aged 0-4 years, in the paper. This raises concern that parents, childcare providers, and preschools may be underestimating infection in seemingly healthy young kids who have been exposed to COVID, wrote lead author Ruth A. Karron, MD, and colleagues in JAMA Network Open.
Methods
The new research involved 690 individuals from 175 households in Maryland who were monitored closely between November 2020 and October 2021. Every week for 8 months, participants completed online symptom checks and underwent PCR testing using nasal swabs, with symptomatic individuals submitting additional swabs for analysis.
“What was different about our study [compared with previous studies] was the intensity of our collection, and the fact that we collected specimens from asymptomatic people,” said Dr. Karron, a pediatrician and professor in the department of international health, Johns Hopkins University, Baltimore, in an interview. “You shed more virus earlier in the infection than later, and the fact that we were sampling every single week meant that we could pick up those early infections.”
The study also stands out for its focus on young children, Dr. Karron said. Enrollment required all households to have at least one child aged 0-4 years, so 256 out of 690 participants (37.1%) were in this youngest age group. The remainder of the population consisted of 100 older children aged 5-17 years (14.5%) and 334 adults aged 18-74 years (48.4%).
Children 4 and under more than twice as likely to be asymptomatic
By the end of the study, 51 participants had tested positive for SARS-CoV-2, among whom 14 had no symptoms. A closer look showed that children 0-4 years of age who contracted COVID were more than twice as likely to be asymptomatic as infected adults (36.8% vs. 14.3%).
The relationship between symptoms and viral load also differed between adults and young children.
While adults with high viral loads – suggesting greater contagiousness – typically had more severe COVID symptoms, no correlation was found in young kids, meaning children with mild or no symptoms could still be highly contagious.
Dr. Karron said these findings should help parents and other stakeholders make better-informed decisions based on known risks. She recommended testing young, asymptomatic children for COVID if they have been exposed to infected individuals, then acting accordingly based on the results.
“If a family is infected with the virus, and the 2-year-old is asymptomatic, and people are thinking about a visit to elderly grandparents who may be frail, one shouldn’t assume that the 2-year-old is uninfected,” Dr. Karron said. “That child should be tested along with other family members.”
Testing should also be considered for young children exposed to COVID at childcare facilities, she added.
But not every expert consulted for this piece shared these opinions of Dr. Karron.
“I question whether that effort is worth it,” said Dean Blumberg, MD, professor and chief of the division of pediatric infectious diseases at UC Davis Health, Sacramento, Calif.
He noted that recent Food and Drug Administration guidance for COVID testing calls for three negative at-home antigen tests to confirm lack of infection.
“That would take 4 days to get those tests done,” he said. “So, it’s a lot of testing. It’s a lot of record keeping, it’s inconvenient, it’s uncomfortable to be tested, and I just question whether it’s worth that effort.”
Applicability of findings to today questioned
Dr. Blumberg also questioned whether the study, which was completed almost a year ago, reflects the current pandemic landscape.
“At the time this study was done, it was predominantly Delta [variant instead of Omicron],” Dr. Blumberg said. “The other issue [with the study] is that … most of the children didn’t have preexisting immunity, so you have to take that into account.”
Preexisting immunity – whether from exposure or vaccination – could lower viral loads, so asymptomatic children today really could be less contagious than they were when the study was done, according to Dr. Blumberg. Kids without symptoms are also less likely to spread the virus, because they aren’t coughing or sneezing, he added.
Sara R. Kim, MD, and Janet A. Englund, MD, of the Seattle Children’s Research Institute, University of Washington, said it’s challenging to know how applicable the findings are, although they sided more with the investigators than Dr. Blumberg.
“Given the higher rate of transmissibility and infectivity of the Omicron variant, it is difficult to make direct associations between findings reported during this study period and those present in the current era during which the Omicron variant is circulating,” they wrote in an accompanying editorial. “However, the higher rates of asymptomatic infection observed among children in this study are likely to be consistent with those observed for current and future viral variants.”
Although the experts offered different interpretations of the findings, they shared similar perspectives on vaccination.
“The most important thing that parents can do is get their kids vaccinated, be vaccinated themselves, and have everybody in the household vaccinated and up to date for all doses that are indicated,” Dr. Blumberg said.
Dr. Karron noted that vaccination will be increasingly important in the coming months.
“Summer is ending; school is starting,” she said. “We’re going to be in large groups indoors again very soon. To keep young children safe, I think it’s really important for them to get vaccinated.”
The study was funded by the CDC. The investigators disclosed no other relationships. Dr. Englund disclosed relationships with AstraZeneca, GlaxoSmithKline, Merck, and others. Dr. Kim and Dr. Blumberg disclosed no relevant conflicts of interest.
FROM JAMA NETWORK OPEN
FDA authorizes updated COVID boosters to target newest variants
The agency cited data to support the safety and efficacy of this next generation of mRNA vaccines targeted toward variants of concern.
The Pfizer EUA corresponds to the company’s combination booster shot that includes the original COVID-19 vaccine as well as a vaccine specifically designed to protect against the most recent Omicron variants, BA.4 and BA.5.
The Moderna combination vaccine will contain both the firm’s original COVID-19 vaccine and a vaccine to protect specifically against Omicron BA.4 and BA.5 subvariants.
As of Aug. 27, BA.4 and BA.4.6 account for about 11% of circulating variants and BA.5 accounts for almost all the remaining 89%, Centers for Disease Control and Prevention data show.
The next step will be review of the scientific data by the CDC’s Advisory Committee on Immunization Practices, which is set to meet Sept. 1 and 2. The final hurdle before distribution of the new vaccines will be sign-off on CDC recommendations for use by agency Director Rochelle Walensky, MD.
This is a developing story. A version of this article first appeared on WebMD.com.
The agency cited data to support the safety and efficacy of this next generation of mRNA vaccines targeted toward variants of concern.
The Pfizer EUA corresponds to the company’s combination booster shot that includes the original COVID-19 vaccine as well as a vaccine specifically designed to protect against the most recent Omicron variants, BA.4 and BA.5.
The Moderna combination vaccine will contain both the firm’s original COVID-19 vaccine and a vaccine to protect specifically against Omicron BA.4 and BA.5 subvariants.
As of Aug. 27, BA.4 and BA.4.6 account for about 11% of circulating variants and BA.5 accounts for almost all the remaining 89%, Centers for Disease Control and Prevention data show.
The next step will be review of the scientific data by the CDC’s Advisory Committee on Immunization Practices, which is set to meet Sept. 1 and 2. The final hurdle before distribution of the new vaccines will be sign-off on CDC recommendations for use by agency Director Rochelle Walensky, MD.
This is a developing story. A version of this article first appeared on WebMD.com.
The agency cited data to support the safety and efficacy of this next generation of mRNA vaccines targeted toward variants of concern.
The Pfizer EUA corresponds to the company’s combination booster shot that includes the original COVID-19 vaccine as well as a vaccine specifically designed to protect against the most recent Omicron variants, BA.4 and BA.5.
The Moderna combination vaccine will contain both the firm’s original COVID-19 vaccine and a vaccine to protect specifically against Omicron BA.4 and BA.5 subvariants.
As of Aug. 27, BA.4 and BA.4.6 account for about 11% of circulating variants and BA.5 accounts for almost all the remaining 89%, Centers for Disease Control and Prevention data show.
The next step will be review of the scientific data by the CDC’s Advisory Committee on Immunization Practices, which is set to meet Sept. 1 and 2. The final hurdle before distribution of the new vaccines will be sign-off on CDC recommendations for use by agency Director Rochelle Walensky, MD.
This is a developing story. A version of this article first appeared on WebMD.com.
Angiolymphoid Hyperplasia with Eosinophilia in a Patient With Coccidioidomycosis
Angiolymphoid hyperplasia with eosinophilia (ALHE) is a rare nodular unencapsulated mass that is characterized by benign anomalous vascular hyperplasia of epithelioidlike endothelial cells attached to dilated blood vessels. The mass is surrounded by lymphocytes and eosinophils that can present clinically as papules, plaques, or nodules.1 The etiology of ALHE is unknown; it is hypothesized that it is a vascular neoplasm or a lymphoproliferative disorder.
Coccidioidomycosis (CM) is a prevalent deep fungal infection endemic to the southwestern United States caused by Coccidioides immitis and Coccidioides posadasii. Infection can occur from direct inoculation through abrasions or direct trauma but usually occurs through the inhalation of spores and can result in a reactive rash (eg, Sweet syndrome, erythema nodosum, interstitial granulomatous dermatitis).2 Coccidioidomycosis also can result in respiratory pneumonia and dissemination from pulmonary infection of the skin. As such, it is important to distinguish CM and its immunologically mediated eruptions for accurate diagnosis and treatment.
We report a novel case of ALHE as a reactive dermatologic presentation in a patient with CM.
Case Report
A 72-year-old woman presented to the dermatology clinic with itchy papules and plaques on the arms and legs of 17 years’ duration. Her medical history included coronary artery disease and hypercholesterolemia as well as a remote history of cutaneous marginal zone B-cell lymphoma of the nose, which was confirmed by histology and treated more than 10 years prior and has remained in remission for 6 years. Her current medications included aspirin, atorvastatin, lisinopril, and metoprolol succinate.
Our patient first presented to our dermatology clinic for itchy nodules and papules on the legs and arms. The patient previously had been seen by another dermatologist 2 months prior for the same condition. At that time, biopsies of the lesions were reported as prurigo nodules. Physical examination at the current presentation revealed round, pink to flesh-colored, raised papules and plaques scattered on the arms and legs (Figure 1). The differential diagnosis included lymphomatoid papulosis, cutaneous B-cell lymphoma, pseudolymphoma, cutaneous CM, and papular mucinosis.
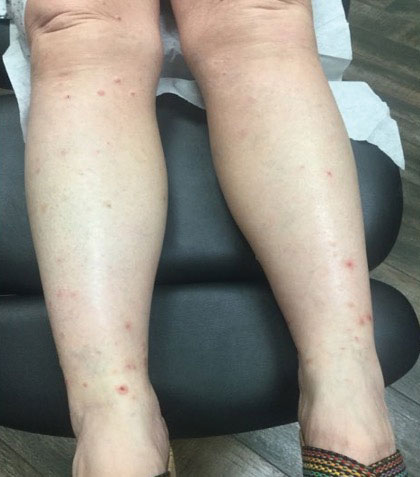
Four-mm punch biopsies of the right proximal pretibial region and left knee region were taken and sent for histologic analysis, direct immunofluorescence testing, and tissue culture. Testing for atypical mycobacteria and deep fungal infection was negative; bacterial cultures and sensitivity testing were negative. Direct immunofluorescence testing was negative. Microscopic examination of material from the right proximal pretibial region showed widely dilated, variously shaped, large blood vessels in a multinodular pattern; the vessels also were surrounded by an inflammatory cell infiltrate containing eosinophils. Histologic findings were consistent with ALHE.
Subsequent biopsies were completed 2 weeks and 1 month from the initial presentation. Both histology reports—from 2 different histopathology laboratories—were consistent with ALHE (Figure 2). Additional work-up during the patient’s initial visit to our clinic for the rash included CM serologic testing, which demonstrated IgM and IgG antibodies. Subsequently, chest radiography revealed a 2.2×2.3-cm mass in the right lower lobe of the lung. Follow-up computed tomography 1 month later confirmed the nodule in the same area to be 2.3×2.1×1.8 cm.
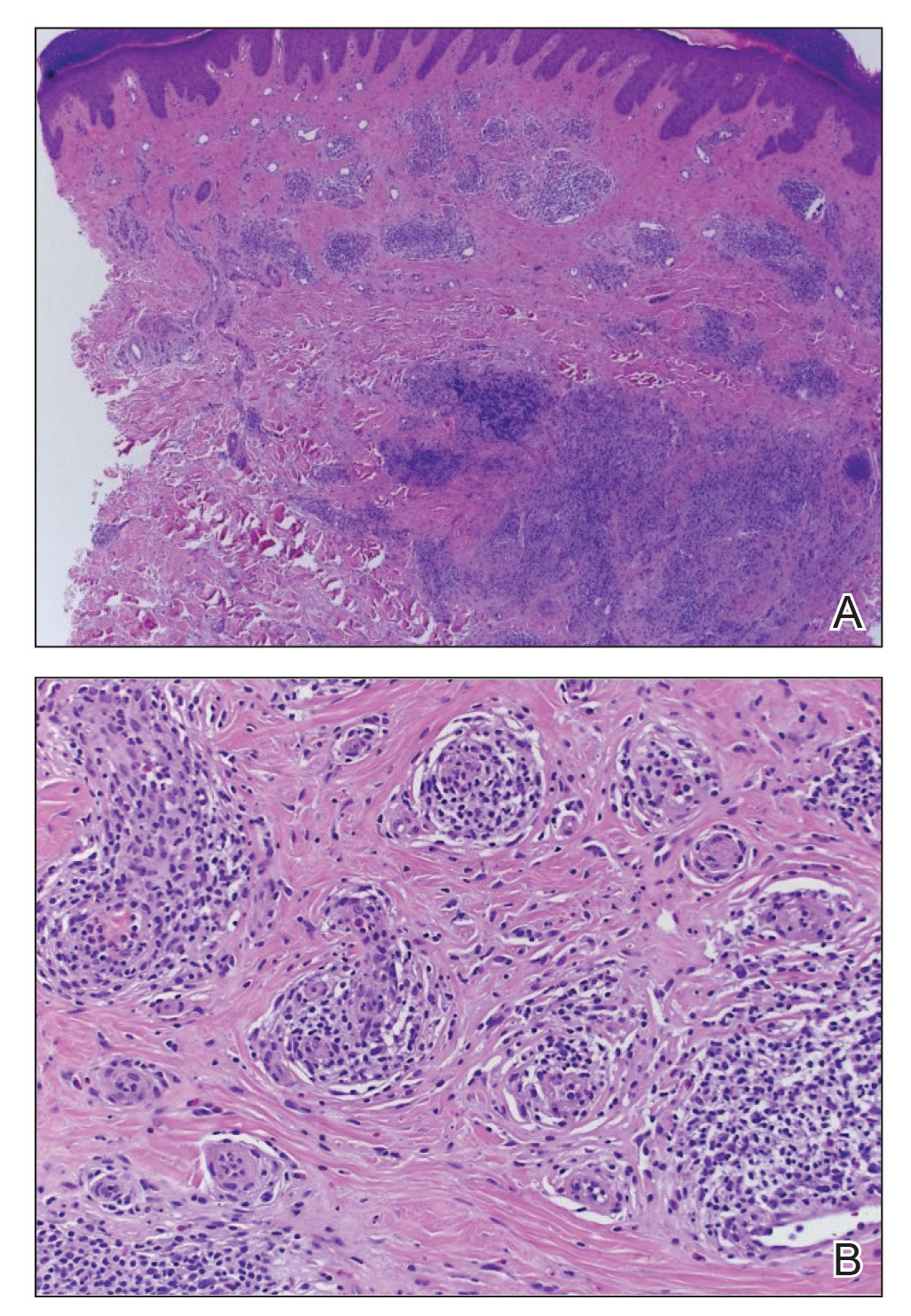
The patient was referred to pulmonology and was treated for pulmonary CM with oral fluconazole 200 mg twice daily for 4 months. Initial treatment also included clobetasol cream 0.05% applied twice daily, which did not produce marked improvement in pruritus. Narrowband UVB phototherapy was attempted, but the patient could not complete the course because of travel time to the office; however, the patient’s ALHE improved considerably with the fluconazole treatment for pulmonary CM.
Oral doxycycline 100 mg twice daily was added to the fluconazole 2 months after her initial visit to our office, which kept the ALHE at bay and helped with the pruritus (Figure 3). Pulmonology and primary care comanaged the pulmonary CM with oral fluconazole 200 mg twice daily. Repeat serologic testing for CM was negative for IgG and IgM after 14 months since the initial visit to the office.
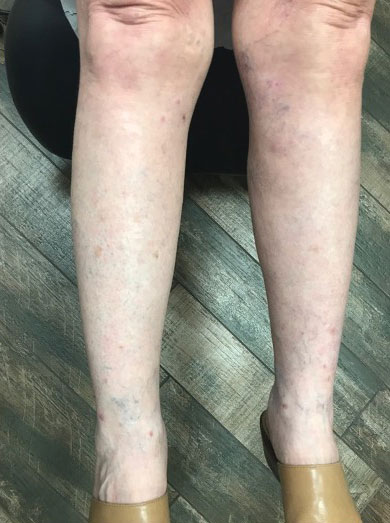
Comment
Pulmonary CM infection has varying dermatologic manifestations. A PubMed search of articles indexed for MEDLINE using the terms ALHE and coccidioidomycosis yielded no case reports; in fact, there have been few reported cases of ALHE at all. Notable conditions associated with ALHE include membranous nephropathy and arteriovenous malformations treated with corticosteroids and surgery, respectively.3,4 Our case is a rare presentation of CM infection manifesting with ALHE. Following treatment and remission for our patient’s CM infection, the ALHE lesion decreased in size.
Standard treatment of uncomplicated CM involves azole antifungals, typically oral fluconazole or itraconazole 400 to 600 mg/d. In more severe cases (eg, immunocompromised patients) amphotericin B can be used.5 Our patient was treated with oral fluconazole 200 mg twice daily for 4 months.
In the literature, treatment via surgical excision, steroid injection, pulsed-dye laser therapy, and radiotherapy also has been described.6-8 Antibiotics including clindamycin, doxycycline, and amoxicillin-clavulanate also have been shown to be effective.9
In our patient, ALHE improved when oral doxycycline 100 mg twice daily was added to the oral fluconazole. In fact, after 4 months of treatment, the CM infection and ALHE lesions both improved to a point at which the lesions were not visible. When those lesions recurred 15 months later, they responded with another course of doxycycline and fluconazole.
Upon recurrence, the patient was asked to have her care transferred to her pulmonologist, who then managed the fluconazole regimen. During the pulmonologist’s workup, no peripheral eosinophilia was found. This is important because eosinophils can be a marker for CM infection; in this case, however, the ALHE lesion was a reactive process to the infection. Classically known to play a reactive role in fungal infection, these white blood cells demonstrate reactivity to the environmental fungus Alternaria alternata by contact-dependent killing, utilizing β2 integrins and CD11b to recognize and adhere to β-glucan. Eosinophils react through contact-dependent killing, releasing cytotoxic granule proteins and proinflammatory mediators, and have been documented to occur in CM and Paracoccidioides brasiliensis infection, in which they deposit major basic protein on the organism.10 Most pertinent to our case with ALHE and CM is the ability of eosinophils to communicate with other immune cells. Eosinophils play a role in the active inflammation of CM through cytokine signaling, which may propagate formation of ALHE.
The function of eosinophils in ALHE is poorly understood; it is unclear whether they act as a primary driver of pathogenesis or are simply indicators of secondary infiltration or infection. Our review of the current literature suggests that eosinophils are unnecessary for progression of ALHE but might be involved at its onset. As reported, even monoclonal antibody therapy (eg, mepolizumab and benralizumab) that effectively depletes eosinophil levels by negating IL-5 signaling do not slow progression of ALHE.11 Symptomatic changes are modest at best (ie, simply softening the ALHE nodules).
Our patient had no peripheral eosinophilia, suggesting that the onset of ALHE might not be caused by eosinophilia but a different inflammatory process—in this patient, by CM. Because peripheral eosinophilia was not seen in our patient, the presence of eosinophils in the ALHE lesion likely is unnecessary for its onset or progression but is a secondary process that exacerbates the lesion. The pathogenesis is unknown but could be directed toward lymphocytes and plasma cells, with eosinophils as part of the dynamic process.11
Conclusion
Because reports of an association between CM and ALHE are limited, our case is distinguished by a unique clinical presentation of ALHE. When a patient is given a diagnosis of ALHE, it therefore is important to consider exposure to CM as a cause, especially in patients who reside in or travel to a region where CM is endemic.
- Wells GC, Whimster IW. Subcutaneous angiolymphoid hyperplasia with eosinophilia. Br J Dermatol. 1969;81:1-14. doi:10.1111/j.1365-2133.1969.tb15914.x
- DiCaudo D. Coccidioidomycosis. Semin Cutan Med Surg. 2014;33:140-145. doi:10.12788/j.sder.0111
- Onishi Y, Ohara K. Angiolymphoid hyperplasia with eosinophilia associated with arteriovenous malformation: a clinicopathological correlation with angiography and serial estimation of serum levels of renin, eosinophil cationic protein and interleukin 5. Br J Dermatol. 1999;140:1153-1156. doi:10.1046/j.1365-2133.1999.02880.x
- Matsumoto A, Matsui I, Namba T, et al. VEGF-A links angiolymphoid hyperplasia with eosinophilia (ALHE) to THSD7A membranous nephropathy: a report of 2 cases. Am J Kidney Dis. 2019;73:880-885. doi:10.1053/j.ajkd.2018.10.009
- Bercovitch RS, Catanzaro A, Schwartz BS, et al. Coccidioidomycosis during pregnancy: a review and recommendations for management. Clin Infect Dis. 2011;53:363-368. doi:10.1093/cid/cir410
- Youssef A, Hasan AR, Youssef Y, et al. Angiolymphoid hyperplasia with eosinophilia: a case report. J Med Case Rep. 2018;12:89. doi:10.1186/s13256-018-1599-x
- Abrahamson TG, Davis DA. Angiolymphoid hyperplasia witheosinophilia responsive to pulsed dye laser. J Am Acad Dermatol. 2003;49(2 suppl case reports):S195-S196. doi:10.1067/mjd.2003.314
- Lembo S, Balato A, Cirillo T, et al. A long-term follow-up of angiolymphoid hyperplasia with eosinophilia treated by corticosteroids: when a traditional therapy is still up-to-date. Case Rep Dermatol. 2011;3:64-67. doi:10.1159/000323182
- Cleveland E. Atypical presentation of angiolymphomatous hyperplasia with eosinophilia. J Am Acad Dermatol. 2018;79(3 suppl 1):AB53. doi:10.1016/j.jaad.2018.05.249
- Ravin KA, Loy M. The eosinophil in infection. Clin Rev Allergy Immunol. 2015;50:214-227. doi:10.1007/s12016-015-8525-4
- Grünewald M, Stölzl D, Wehkamp U, et al. Role of eosinophils in angiolymphoid hyperplasia with eosinophilia. JAMA Dermatol. 2021;157:1241-1243. doi:10.1001/jamadermatol.2021.2732
Angiolymphoid hyperplasia with eosinophilia (ALHE) is a rare nodular unencapsulated mass that is characterized by benign anomalous vascular hyperplasia of epithelioidlike endothelial cells attached to dilated blood vessels. The mass is surrounded by lymphocytes and eosinophils that can present clinically as papules, plaques, or nodules.1 The etiology of ALHE is unknown; it is hypothesized that it is a vascular neoplasm or a lymphoproliferative disorder.
Coccidioidomycosis (CM) is a prevalent deep fungal infection endemic to the southwestern United States caused by Coccidioides immitis and Coccidioides posadasii. Infection can occur from direct inoculation through abrasions or direct trauma but usually occurs through the inhalation of spores and can result in a reactive rash (eg, Sweet syndrome, erythema nodosum, interstitial granulomatous dermatitis).2 Coccidioidomycosis also can result in respiratory pneumonia and dissemination from pulmonary infection of the skin. As such, it is important to distinguish CM and its immunologically mediated eruptions for accurate diagnosis and treatment.
We report a novel case of ALHE as a reactive dermatologic presentation in a patient with CM.
Case Report
A 72-year-old woman presented to the dermatology clinic with itchy papules and plaques on the arms and legs of 17 years’ duration. Her medical history included coronary artery disease and hypercholesterolemia as well as a remote history of cutaneous marginal zone B-cell lymphoma of the nose, which was confirmed by histology and treated more than 10 years prior and has remained in remission for 6 years. Her current medications included aspirin, atorvastatin, lisinopril, and metoprolol succinate.
Our patient first presented to our dermatology clinic for itchy nodules and papules on the legs and arms. The patient previously had been seen by another dermatologist 2 months prior for the same condition. At that time, biopsies of the lesions were reported as prurigo nodules. Physical examination at the current presentation revealed round, pink to flesh-colored, raised papules and plaques scattered on the arms and legs (Figure 1). The differential diagnosis included lymphomatoid papulosis, cutaneous B-cell lymphoma, pseudolymphoma, cutaneous CM, and papular mucinosis.

Four-mm punch biopsies of the right proximal pretibial region and left knee region were taken and sent for histologic analysis, direct immunofluorescence testing, and tissue culture. Testing for atypical mycobacteria and deep fungal infection was negative; bacterial cultures and sensitivity testing were negative. Direct immunofluorescence testing was negative. Microscopic examination of material from the right proximal pretibial region showed widely dilated, variously shaped, large blood vessels in a multinodular pattern; the vessels also were surrounded by an inflammatory cell infiltrate containing eosinophils. Histologic findings were consistent with ALHE.
Subsequent biopsies were completed 2 weeks and 1 month from the initial presentation. Both histology reports—from 2 different histopathology laboratories—were consistent with ALHE (Figure 2). Additional work-up during the patient’s initial visit to our clinic for the rash included CM serologic testing, which demonstrated IgM and IgG antibodies. Subsequently, chest radiography revealed a 2.2×2.3-cm mass in the right lower lobe of the lung. Follow-up computed tomography 1 month later confirmed the nodule in the same area to be 2.3×2.1×1.8 cm.

The patient was referred to pulmonology and was treated for pulmonary CM with oral fluconazole 200 mg twice daily for 4 months. Initial treatment also included clobetasol cream 0.05% applied twice daily, which did not produce marked improvement in pruritus. Narrowband UVB phototherapy was attempted, but the patient could not complete the course because of travel time to the office; however, the patient’s ALHE improved considerably with the fluconazole treatment for pulmonary CM.
Oral doxycycline 100 mg twice daily was added to the fluconazole 2 months after her initial visit to our office, which kept the ALHE at bay and helped with the pruritus (Figure 3). Pulmonology and primary care comanaged the pulmonary CM with oral fluconazole 200 mg twice daily. Repeat serologic testing for CM was negative for IgG and IgM after 14 months since the initial visit to the office.

Comment
Pulmonary CM infection has varying dermatologic manifestations. A PubMed search of articles indexed for MEDLINE using the terms ALHE and coccidioidomycosis yielded no case reports; in fact, there have been few reported cases of ALHE at all. Notable conditions associated with ALHE include membranous nephropathy and arteriovenous malformations treated with corticosteroids and surgery, respectively.3,4 Our case is a rare presentation of CM infection manifesting with ALHE. Following treatment and remission for our patient’s CM infection, the ALHE lesion decreased in size.
Standard treatment of uncomplicated CM involves azole antifungals, typically oral fluconazole or itraconazole 400 to 600 mg/d. In more severe cases (eg, immunocompromised patients) amphotericin B can be used.5 Our patient was treated with oral fluconazole 200 mg twice daily for 4 months.
In the literature, treatment via surgical excision, steroid injection, pulsed-dye laser therapy, and radiotherapy also has been described.6-8 Antibiotics including clindamycin, doxycycline, and amoxicillin-clavulanate also have been shown to be effective.9
In our patient, ALHE improved when oral doxycycline 100 mg twice daily was added to the oral fluconazole. In fact, after 4 months of treatment, the CM infection and ALHE lesions both improved to a point at which the lesions were not visible. When those lesions recurred 15 months later, they responded with another course of doxycycline and fluconazole.
Upon recurrence, the patient was asked to have her care transferred to her pulmonologist, who then managed the fluconazole regimen. During the pulmonologist’s workup, no peripheral eosinophilia was found. This is important because eosinophils can be a marker for CM infection; in this case, however, the ALHE lesion was a reactive process to the infection. Classically known to play a reactive role in fungal infection, these white blood cells demonstrate reactivity to the environmental fungus Alternaria alternata by contact-dependent killing, utilizing β2 integrins and CD11b to recognize and adhere to β-glucan. Eosinophils react through contact-dependent killing, releasing cytotoxic granule proteins and proinflammatory mediators, and have been documented to occur in CM and Paracoccidioides brasiliensis infection, in which they deposit major basic protein on the organism.10 Most pertinent to our case with ALHE and CM is the ability of eosinophils to communicate with other immune cells. Eosinophils play a role in the active inflammation of CM through cytokine signaling, which may propagate formation of ALHE.
The function of eosinophils in ALHE is poorly understood; it is unclear whether they act as a primary driver of pathogenesis or are simply indicators of secondary infiltration or infection. Our review of the current literature suggests that eosinophils are unnecessary for progression of ALHE but might be involved at its onset. As reported, even monoclonal antibody therapy (eg, mepolizumab and benralizumab) that effectively depletes eosinophil levels by negating IL-5 signaling do not slow progression of ALHE.11 Symptomatic changes are modest at best (ie, simply softening the ALHE nodules).
Our patient had no peripheral eosinophilia, suggesting that the onset of ALHE might not be caused by eosinophilia but a different inflammatory process—in this patient, by CM. Because peripheral eosinophilia was not seen in our patient, the presence of eosinophils in the ALHE lesion likely is unnecessary for its onset or progression but is a secondary process that exacerbates the lesion. The pathogenesis is unknown but could be directed toward lymphocytes and plasma cells, with eosinophils as part of the dynamic process.11
Conclusion
Because reports of an association between CM and ALHE are limited, our case is distinguished by a unique clinical presentation of ALHE. When a patient is given a diagnosis of ALHE, it therefore is important to consider exposure to CM as a cause, especially in patients who reside in or travel to a region where CM is endemic.
Angiolymphoid hyperplasia with eosinophilia (ALHE) is a rare nodular unencapsulated mass that is characterized by benign anomalous vascular hyperplasia of epithelioidlike endothelial cells attached to dilated blood vessels. The mass is surrounded by lymphocytes and eosinophils that can present clinically as papules, plaques, or nodules.1 The etiology of ALHE is unknown; it is hypothesized that it is a vascular neoplasm or a lymphoproliferative disorder.
Coccidioidomycosis (CM) is a prevalent deep fungal infection endemic to the southwestern United States caused by Coccidioides immitis and Coccidioides posadasii. Infection can occur from direct inoculation through abrasions or direct trauma but usually occurs through the inhalation of spores and can result in a reactive rash (eg, Sweet syndrome, erythema nodosum, interstitial granulomatous dermatitis).2 Coccidioidomycosis also can result in respiratory pneumonia and dissemination from pulmonary infection of the skin. As such, it is important to distinguish CM and its immunologically mediated eruptions for accurate diagnosis and treatment.
We report a novel case of ALHE as a reactive dermatologic presentation in a patient with CM.
Case Report
A 72-year-old woman presented to the dermatology clinic with itchy papules and plaques on the arms and legs of 17 years’ duration. Her medical history included coronary artery disease and hypercholesterolemia as well as a remote history of cutaneous marginal zone B-cell lymphoma of the nose, which was confirmed by histology and treated more than 10 years prior and has remained in remission for 6 years. Her current medications included aspirin, atorvastatin, lisinopril, and metoprolol succinate.
Our patient first presented to our dermatology clinic for itchy nodules and papules on the legs and arms. The patient previously had been seen by another dermatologist 2 months prior for the same condition. At that time, biopsies of the lesions were reported as prurigo nodules. Physical examination at the current presentation revealed round, pink to flesh-colored, raised papules and plaques scattered on the arms and legs (Figure 1). The differential diagnosis included lymphomatoid papulosis, cutaneous B-cell lymphoma, pseudolymphoma, cutaneous CM, and papular mucinosis.

Four-mm punch biopsies of the right proximal pretibial region and left knee region were taken and sent for histologic analysis, direct immunofluorescence testing, and tissue culture. Testing for atypical mycobacteria and deep fungal infection was negative; bacterial cultures and sensitivity testing were negative. Direct immunofluorescence testing was negative. Microscopic examination of material from the right proximal pretibial region showed widely dilated, variously shaped, large blood vessels in a multinodular pattern; the vessels also were surrounded by an inflammatory cell infiltrate containing eosinophils. Histologic findings were consistent with ALHE.
Subsequent biopsies were completed 2 weeks and 1 month from the initial presentation. Both histology reports—from 2 different histopathology laboratories—were consistent with ALHE (Figure 2). Additional work-up during the patient’s initial visit to our clinic for the rash included CM serologic testing, which demonstrated IgM and IgG antibodies. Subsequently, chest radiography revealed a 2.2×2.3-cm mass in the right lower lobe of the lung. Follow-up computed tomography 1 month later confirmed the nodule in the same area to be 2.3×2.1×1.8 cm.

The patient was referred to pulmonology and was treated for pulmonary CM with oral fluconazole 200 mg twice daily for 4 months. Initial treatment also included clobetasol cream 0.05% applied twice daily, which did not produce marked improvement in pruritus. Narrowband UVB phototherapy was attempted, but the patient could not complete the course because of travel time to the office; however, the patient’s ALHE improved considerably with the fluconazole treatment for pulmonary CM.
Oral doxycycline 100 mg twice daily was added to the fluconazole 2 months after her initial visit to our office, which kept the ALHE at bay and helped with the pruritus (Figure 3). Pulmonology and primary care comanaged the pulmonary CM with oral fluconazole 200 mg twice daily. Repeat serologic testing for CM was negative for IgG and IgM after 14 months since the initial visit to the office.

Comment
Pulmonary CM infection has varying dermatologic manifestations. A PubMed search of articles indexed for MEDLINE using the terms ALHE and coccidioidomycosis yielded no case reports; in fact, there have been few reported cases of ALHE at all. Notable conditions associated with ALHE include membranous nephropathy and arteriovenous malformations treated with corticosteroids and surgery, respectively.3,4 Our case is a rare presentation of CM infection manifesting with ALHE. Following treatment and remission for our patient’s CM infection, the ALHE lesion decreased in size.
Standard treatment of uncomplicated CM involves azole antifungals, typically oral fluconazole or itraconazole 400 to 600 mg/d. In more severe cases (eg, immunocompromised patients) amphotericin B can be used.5 Our patient was treated with oral fluconazole 200 mg twice daily for 4 months.
In the literature, treatment via surgical excision, steroid injection, pulsed-dye laser therapy, and radiotherapy also has been described.6-8 Antibiotics including clindamycin, doxycycline, and amoxicillin-clavulanate also have been shown to be effective.9
In our patient, ALHE improved when oral doxycycline 100 mg twice daily was added to the oral fluconazole. In fact, after 4 months of treatment, the CM infection and ALHE lesions both improved to a point at which the lesions were not visible. When those lesions recurred 15 months later, they responded with another course of doxycycline and fluconazole.
Upon recurrence, the patient was asked to have her care transferred to her pulmonologist, who then managed the fluconazole regimen. During the pulmonologist’s workup, no peripheral eosinophilia was found. This is important because eosinophils can be a marker for CM infection; in this case, however, the ALHE lesion was a reactive process to the infection. Classically known to play a reactive role in fungal infection, these white blood cells demonstrate reactivity to the environmental fungus Alternaria alternata by contact-dependent killing, utilizing β2 integrins and CD11b to recognize and adhere to β-glucan. Eosinophils react through contact-dependent killing, releasing cytotoxic granule proteins and proinflammatory mediators, and have been documented to occur in CM and Paracoccidioides brasiliensis infection, in which they deposit major basic protein on the organism.10 Most pertinent to our case with ALHE and CM is the ability of eosinophils to communicate with other immune cells. Eosinophils play a role in the active inflammation of CM through cytokine signaling, which may propagate formation of ALHE.
The function of eosinophils in ALHE is poorly understood; it is unclear whether they act as a primary driver of pathogenesis or are simply indicators of secondary infiltration or infection. Our review of the current literature suggests that eosinophils are unnecessary for progression of ALHE but might be involved at its onset. As reported, even monoclonal antibody therapy (eg, mepolizumab and benralizumab) that effectively depletes eosinophil levels by negating IL-5 signaling do not slow progression of ALHE.11 Symptomatic changes are modest at best (ie, simply softening the ALHE nodules).
Our patient had no peripheral eosinophilia, suggesting that the onset of ALHE might not be caused by eosinophilia but a different inflammatory process—in this patient, by CM. Because peripheral eosinophilia was not seen in our patient, the presence of eosinophils in the ALHE lesion likely is unnecessary for its onset or progression but is a secondary process that exacerbates the lesion. The pathogenesis is unknown but could be directed toward lymphocytes and plasma cells, with eosinophils as part of the dynamic process.11
Conclusion
Because reports of an association between CM and ALHE are limited, our case is distinguished by a unique clinical presentation of ALHE. When a patient is given a diagnosis of ALHE, it therefore is important to consider exposure to CM as a cause, especially in patients who reside in or travel to a region where CM is endemic.
- Wells GC, Whimster IW. Subcutaneous angiolymphoid hyperplasia with eosinophilia. Br J Dermatol. 1969;81:1-14. doi:10.1111/j.1365-2133.1969.tb15914.x
- DiCaudo D. Coccidioidomycosis. Semin Cutan Med Surg. 2014;33:140-145. doi:10.12788/j.sder.0111
- Onishi Y, Ohara K. Angiolymphoid hyperplasia with eosinophilia associated with arteriovenous malformation: a clinicopathological correlation with angiography and serial estimation of serum levels of renin, eosinophil cationic protein and interleukin 5. Br J Dermatol. 1999;140:1153-1156. doi:10.1046/j.1365-2133.1999.02880.x
- Matsumoto A, Matsui I, Namba T, et al. VEGF-A links angiolymphoid hyperplasia with eosinophilia (ALHE) to THSD7A membranous nephropathy: a report of 2 cases. Am J Kidney Dis. 2019;73:880-885. doi:10.1053/j.ajkd.2018.10.009
- Bercovitch RS, Catanzaro A, Schwartz BS, et al. Coccidioidomycosis during pregnancy: a review and recommendations for management. Clin Infect Dis. 2011;53:363-368. doi:10.1093/cid/cir410
- Youssef A, Hasan AR, Youssef Y, et al. Angiolymphoid hyperplasia with eosinophilia: a case report. J Med Case Rep. 2018;12:89. doi:10.1186/s13256-018-1599-x
- Abrahamson TG, Davis DA. Angiolymphoid hyperplasia witheosinophilia responsive to pulsed dye laser. J Am Acad Dermatol. 2003;49(2 suppl case reports):S195-S196. doi:10.1067/mjd.2003.314
- Lembo S, Balato A, Cirillo T, et al. A long-term follow-up of angiolymphoid hyperplasia with eosinophilia treated by corticosteroids: when a traditional therapy is still up-to-date. Case Rep Dermatol. 2011;3:64-67. doi:10.1159/000323182
- Cleveland E. Atypical presentation of angiolymphomatous hyperplasia with eosinophilia. J Am Acad Dermatol. 2018;79(3 suppl 1):AB53. doi:10.1016/j.jaad.2018.05.249
- Ravin KA, Loy M. The eosinophil in infection. Clin Rev Allergy Immunol. 2015;50:214-227. doi:10.1007/s12016-015-8525-4
- Grünewald M, Stölzl D, Wehkamp U, et al. Role of eosinophils in angiolymphoid hyperplasia with eosinophilia. JAMA Dermatol. 2021;157:1241-1243. doi:10.1001/jamadermatol.2021.2732
- Wells GC, Whimster IW. Subcutaneous angiolymphoid hyperplasia with eosinophilia. Br J Dermatol. 1969;81:1-14. doi:10.1111/j.1365-2133.1969.tb15914.x
- DiCaudo D. Coccidioidomycosis. Semin Cutan Med Surg. 2014;33:140-145. doi:10.12788/j.sder.0111
- Onishi Y, Ohara K. Angiolymphoid hyperplasia with eosinophilia associated with arteriovenous malformation: a clinicopathological correlation with angiography and serial estimation of serum levels of renin, eosinophil cationic protein and interleukin 5. Br J Dermatol. 1999;140:1153-1156. doi:10.1046/j.1365-2133.1999.02880.x
- Matsumoto A, Matsui I, Namba T, et al. VEGF-A links angiolymphoid hyperplasia with eosinophilia (ALHE) to THSD7A membranous nephropathy: a report of 2 cases. Am J Kidney Dis. 2019;73:880-885. doi:10.1053/j.ajkd.2018.10.009
- Bercovitch RS, Catanzaro A, Schwartz BS, et al. Coccidioidomycosis during pregnancy: a review and recommendations for management. Clin Infect Dis. 2011;53:363-368. doi:10.1093/cid/cir410
- Youssef A, Hasan AR, Youssef Y, et al. Angiolymphoid hyperplasia with eosinophilia: a case report. J Med Case Rep. 2018;12:89. doi:10.1186/s13256-018-1599-x
- Abrahamson TG, Davis DA. Angiolymphoid hyperplasia witheosinophilia responsive to pulsed dye laser. J Am Acad Dermatol. 2003;49(2 suppl case reports):S195-S196. doi:10.1067/mjd.2003.314
- Lembo S, Balato A, Cirillo T, et al. A long-term follow-up of angiolymphoid hyperplasia with eosinophilia treated by corticosteroids: when a traditional therapy is still up-to-date. Case Rep Dermatol. 2011;3:64-67. doi:10.1159/000323182
- Cleveland E. Atypical presentation of angiolymphomatous hyperplasia with eosinophilia. J Am Acad Dermatol. 2018;79(3 suppl 1):AB53. doi:10.1016/j.jaad.2018.05.249
- Ravin KA, Loy M. The eosinophil in infection. Clin Rev Allergy Immunol. 2015;50:214-227. doi:10.1007/s12016-015-8525-4
- Grünewald M, Stölzl D, Wehkamp U, et al. Role of eosinophils in angiolymphoid hyperplasia with eosinophilia. JAMA Dermatol. 2021;157:1241-1243. doi:10.1001/jamadermatol.2021.2732
Practice Points
- Angiolymphoid hyperplasia with eosinophilia (ALHE) is a rare entity of unknown etiology.
- There is an association between ALHE and coccidioidomycosis (CM). Patients who present with ALHE and reside in a CM-endemic region should be examined for CM.
Children and COVID: New cases increase; hospital admissions could follow
New cases of COVID-19 in children were up again after 2 weeks of declines, and preliminary data suggest that hospitalizations may be on the rise as well.
, based on data collected by the American Academy of Pediatrics and the Children’s Hospital Association from state and territorial health departments.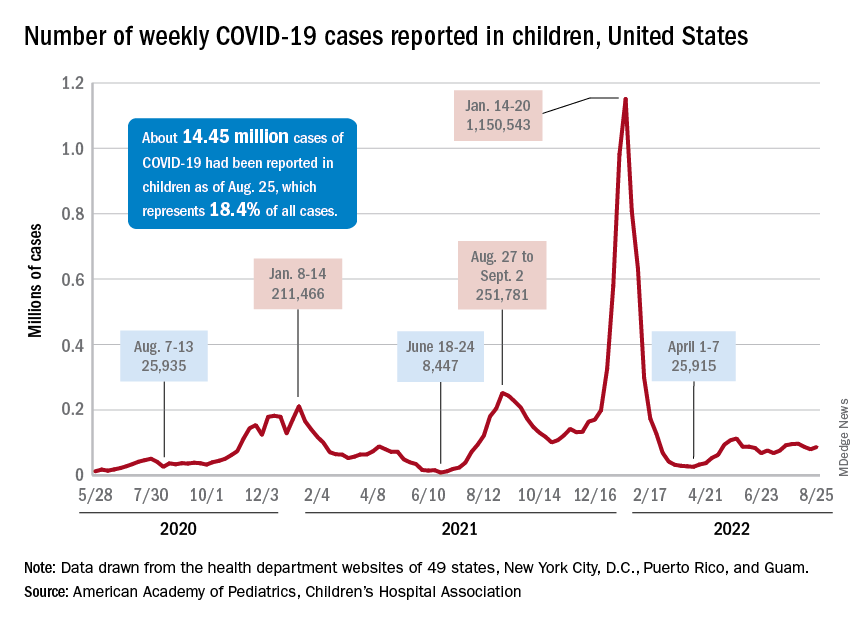
A similar increase seems to be reflected by hospital-level data. The latest 7-day (Aug. 21-27) average is 305 new admissions with diagnosed COVID per day among children aged 0-17 years, compared with 290 per day for the week of Aug. 14-20, the Centers for Disease Control and Prevention reported, while also noting the potential for reporting delays in the most recent 7-day period.
Daily hospital admissions for COVID had been headed downward through the first half of August, falling from 0.46 per 100,000 population at the end of July to 0.40 on Aug. 19, the CDC said on its COVID Data Tracker. Since then, however, admissions have gone the other way, with the preliminary nature of the latest data suggesting that the numbers will be even higher as more hospitals report over the next few days.
Vaccine initiations continue to fall
Initiations among school-age children have fallen for 3 consecutive weeks since Aug. 3, when numbers receiving their first vaccinations reached late-summer highs for those aged 5-11 and 12-17 years. Children under age 5, included in the CDC data for the first time on Aug. 11 as separate groups – under 2 years and 2-4 years – have had vaccine initiations drop by 8.0% and 19.8% over the 2 following weeks, the CDC said.
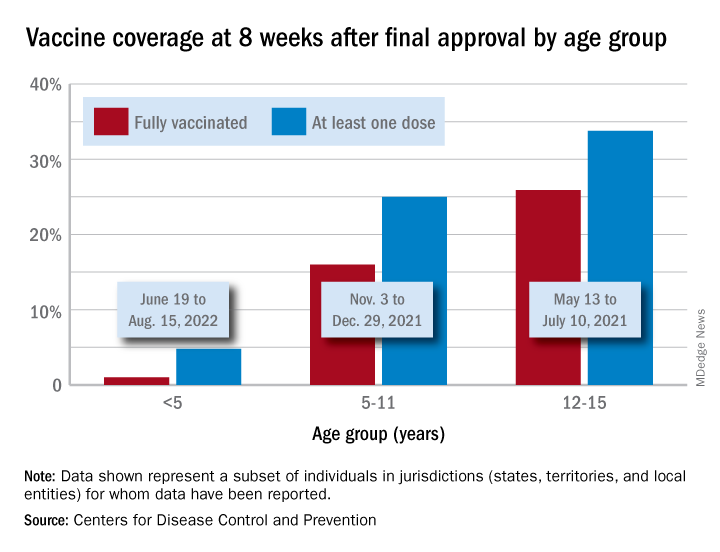
Through their first 8 weeks of vaccine eligibility (June 19 to Aug. 15), 4.8% of children under 5 years of age had received a first vaccination and 1.0% were fully vaccinated. For the two other age groups (5-11 and 12-15) who became eligible after the very first emergency authorization back in 2020, the respective proportions were 25.0% and 16.0% (5-11) and 33.8% and 26.1% (12-15) through the first 8 weeks, according to CDC data.
New cases of COVID-19 in children were up again after 2 weeks of declines, and preliminary data suggest that hospitalizations may be on the rise as well.
, based on data collected by the American Academy of Pediatrics and the Children’s Hospital Association from state and territorial health departments.
A similar increase seems to be reflected by hospital-level data. The latest 7-day (Aug. 21-27) average is 305 new admissions with diagnosed COVID per day among children aged 0-17 years, compared with 290 per day for the week of Aug. 14-20, the Centers for Disease Control and Prevention reported, while also noting the potential for reporting delays in the most recent 7-day period.
Daily hospital admissions for COVID had been headed downward through the first half of August, falling from 0.46 per 100,000 population at the end of July to 0.40 on Aug. 19, the CDC said on its COVID Data Tracker. Since then, however, admissions have gone the other way, with the preliminary nature of the latest data suggesting that the numbers will be even higher as more hospitals report over the next few days.
Vaccine initiations continue to fall
Initiations among school-age children have fallen for 3 consecutive weeks since Aug. 3, when numbers receiving their first vaccinations reached late-summer highs for those aged 5-11 and 12-17 years. Children under age 5, included in the CDC data for the first time on Aug. 11 as separate groups – under 2 years and 2-4 years – have had vaccine initiations drop by 8.0% and 19.8% over the 2 following weeks, the CDC said.

Through their first 8 weeks of vaccine eligibility (June 19 to Aug. 15), 4.8% of children under 5 years of age had received a first vaccination and 1.0% were fully vaccinated. For the two other age groups (5-11 and 12-15) who became eligible after the very first emergency authorization back in 2020, the respective proportions were 25.0% and 16.0% (5-11) and 33.8% and 26.1% (12-15) through the first 8 weeks, according to CDC data.
New cases of COVID-19 in children were up again after 2 weeks of declines, and preliminary data suggest that hospitalizations may be on the rise as well.
, based on data collected by the American Academy of Pediatrics and the Children’s Hospital Association from state and territorial health departments.
A similar increase seems to be reflected by hospital-level data. The latest 7-day (Aug. 21-27) average is 305 new admissions with diagnosed COVID per day among children aged 0-17 years, compared with 290 per day for the week of Aug. 14-20, the Centers for Disease Control and Prevention reported, while also noting the potential for reporting delays in the most recent 7-day period.
Daily hospital admissions for COVID had been headed downward through the first half of August, falling from 0.46 per 100,000 population at the end of July to 0.40 on Aug. 19, the CDC said on its COVID Data Tracker. Since then, however, admissions have gone the other way, with the preliminary nature of the latest data suggesting that the numbers will be even higher as more hospitals report over the next few days.
Vaccine initiations continue to fall
Initiations among school-age children have fallen for 3 consecutive weeks since Aug. 3, when numbers receiving their first vaccinations reached late-summer highs for those aged 5-11 and 12-17 years. Children under age 5, included in the CDC data for the first time on Aug. 11 as separate groups – under 2 years and 2-4 years – have had vaccine initiations drop by 8.0% and 19.8% over the 2 following weeks, the CDC said.

Through their first 8 weeks of vaccine eligibility (June 19 to Aug. 15), 4.8% of children under 5 years of age had received a first vaccination and 1.0% were fully vaccinated. For the two other age groups (5-11 and 12-15) who became eligible after the very first emergency authorization back in 2020, the respective proportions were 25.0% and 16.0% (5-11) and 33.8% and 26.1% (12-15) through the first 8 weeks, according to CDC data.