User login
Asymptomatic Violaceous Plaques on the Face and Back
The Diagnosis: Cutaneous Sarcoidosis
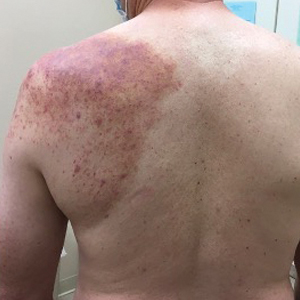
A biopsy of a plaque on the back confirmed cutaneous sarcoidosis (CS). A chest radiograph demonstrated hilar nodes, and a referral was placed for comanagement with a pulmonologist. Histopathology was critical in making the diagnosis, with well-circumscribed noncaseating granulomas present in the dermis. The granulomas in CS often are described as naked, as there are minimal lymphocytes present and plasma cells normally are absent.1 Because the lungs are the most common site of involvement, a chest radiograph is necessary to examine for systemic sarcoidosis. Laboratory workup is used to evaluate for lymphopenia, hypercalcemia, elevated blood sedimentation rate, and elevated angiotensin- converting enzyme levels, which are common in systemic sarcoidosis.1
Sarcoidosis is a multisystemic granulomatous disorder with an unknown etiology. It is believed to develop in genetically predisposed individuals as a reaction to unidentified antigens in the environment.1 Helper T cells (TH1) respond to these environmental antigens in those who are susceptible, which leads to the disease process, but paradoxically, even with the elevation of cellular immune activity at the sites of the granulomatous inflammation, the peripheral immune response in these patients is suppressed as shown by lymphopenia.2
Cutaneous sarcoidosis is found in approximately one-third of patients with systemic sarcoidosis but can occur without systemic involvement.1,2 Sarcoidosis is reported worldwide and affects patients of all races and ethnicities, ages, and sexes but does have a higher prevalence among Black individuals in the United States, patients younger than 40 years (peak incidence, 20–29 years of age), and females.2 In 80% of patients, CS occurs before systemic sarcoidosis develops, or they may develop simultaneously.1
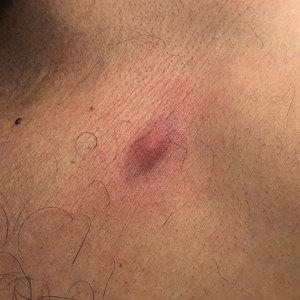
Cutaneous sarcoidosis has a wide range of clinical presentations that are classified as specific and nonspecific. Specific lesions in CS contain noncaseating granulomas while nonspecific lesions in CS appear as reactive processes.2 The most common specific presentation of CS includes papules that are brown in pigmentation in lighter skin tones and red to violaceous in darker skin tones (Figure). The most common nonspecific skin manifestation is erythema nodosum, which represents a hypersensitivity reaction. Cutaneous sarcoidosis can appear as hypopigmented or hyperpigmented patches or plaques.1
Treatments for CS vary based on the individual.1 For milder and more localized cases, topical or intralesional steroids may be used. If systemic sarcoidosis is suspected or if there is diffuse involvement of the skin, systemic steroids, antimalarials (eg, hydroxychloroquine), low-dose methotrexate, minocycline, allopurinol, azathioprine, isotretinoin, tumor necrosis factor α inhibitors, or psoralen plus long-wave UVA radiation may be used. If systemic sarcoidosis is present, referral to a pulmonologist is recommended for co-management.1
Cutaneous sarcoidosis is known as the “great imitator,” and there are multiple diseases to consider in the differential that are distinguished by the physical findings.1 In our case of a middle-aged Black woman with indurated plaques, a few diagnoses to consider were psoriasis, discoid lupus erythematosus (DLE), mycosis fungoides (MF), and tinea infection.
Psoriasis is a common disease, and 90% of patients have chronic plaquelike disease with well-demarcated erythematous plaques that have a silver-gray scale and a positive Auspitz sign (also known as pinpoint bleeding).3 Plaques often are distributed on the trunk, limb extensors, and scalp, along with nail changes. Some patients also have joint pain, indicating psoriatic arthritis. The etiology of psoriasis is unknown, but it develops due to unrestrained keratinocyte proliferation and defective differentiation, which leads to histopathology showing regular acanthosis and papillary dermal ectasia with rouleaux. Mild cases typically are treated with topical steroids or vitamin D, while more severe cases are treated with methotrexate, cyclosporine, retinoids, or biologics.3
Discoid lupus erythematosus occurs 4 times more often in Black patients than in White patients. Clinically, DLE begins as well-defined, erythematous, scaly patches that expand with hyperpigmentation at the periphery and leave an atrophic, scarred, hypopigmented center.4 It typically is localized to the head and neck, but in cases where it disseminates elsewhere on the body, the risk for systemic lupus erythematosus increases from 1.2% to 28%.5 Histopathology of DLE shows vacuolar degeneration of the basal cell layer in the epidermis along with patchy lymphocytic infiltrate in the dermis. Treatments range from topical steroids for mild cases to antimalarial agents, retinoids, anti-inflammatory drugs, and calcineurin inhibitors for more severe cases.4
Although there are multiple types of cutaneous T-cell lymphoma, the most common is MF, which traditionally is nonaggressive. The typical patient with MF is older than 60 years and presents with indolent, ongoing, flat to minimally indurated patches or plaques that have cigarette paper scale. As MF progresses, some plaques grow into tumors and can become more aggressive. Histologically, MF changes based on its clinical stage, with the initial phase showing epidermotropic atypical lymphocytes and later phases showing less epitheliotropic, larger, atypical lymphocytes. The treatment algorithm varies depending on cutaneous T-cell lymphoma staging.6
Tinea infections are caused by dermatophytes. In prepubertal children, they predominantly appear as tinea corporis (on the body) or tinea capitis (on the scalp), but in adults they appear as tinea cruris (on the groin), tinea pedis (on the feet), or tinea unguium (on the nails).7 Tinea infections classically are known to appear as an annular patch with an active erythematous scaling border and central clearing. The patches can be pruritic. Potassium hydroxide preparation of a skin scraping is a quick test to use in the office; if the results are inconclusive, a culture may be required. Treatment depends on the location of the infection but typically involves either topical or oral antifungal agents.7
- Tchernev G, Cardoso JC, Chokoeva AA, et al. The “mystery” of cutaneous sarcoidosis: facts and controversies. Int J Immunopathol Pharmacol. 2014;27:321-330. doi:10.1177/039463201402700302
- Ali MM, Atwan AA, Gonzalez ML. Cutaneous sarcoidosis: updates in the pathogenesis. J Eur Acad Dermatol Venereol. 2010;24:747-755. doi:10.1111/j.1468-3083.2009.03517.x
- Rendon A, Schäkel K. Psoriasis pathogenesis and treatment [published online March 23, 2019]. Int J Mol Sci. 2019;20:1475. doi:10.3390/ijms20061475
- McDaniel B, Sukumaran S, Koritala T, et al. Discoid lupus erythematosus. StatPearls [Internet]. StatPearls Publishing; 2023. Accessed December 11, 2023. https://www.ncbi.nlm.nih.gov/books/NBK493145/
- Bhat MR, Hulmani M, Dandakeri S, et al. Disseminated discoid lupus erythematosus leading to squamous cell carcinoma. Indian J Dermatol. 2012;57:158-161. doi:10.4103/0019-5154.94298
- Pulitzer M. Cutaneous T-cell Lymphoma. Clin Lab Med. 2017; 37:527-546. doi:10.1016/j.cll.2017.06.006
- Ely JW, Rosenfeld S, Seabury Stone M. Diagnosis and management of tinea infections. Am Fam Physician. 2014;90:702-710.
The Diagnosis: Cutaneous Sarcoidosis
A biopsy of a plaque on the back confirmed cutaneous sarcoidosis (CS). A chest radiograph demonstrated hilar nodes, and a referral was placed for comanagement with a pulmonologist. Histopathology was critical in making the diagnosis, with well-circumscribed noncaseating granulomas present in the dermis. The granulomas in CS often are described as naked, as there are minimal lymphocytes present and plasma cells normally are absent.1 Because the lungs are the most common site of involvement, a chest radiograph is necessary to examine for systemic sarcoidosis. Laboratory workup is used to evaluate for lymphopenia, hypercalcemia, elevated blood sedimentation rate, and elevated angiotensin- converting enzyme levels, which are common in systemic sarcoidosis.1
Sarcoidosis is a multisystemic granulomatous disorder with an unknown etiology. It is believed to develop in genetically predisposed individuals as a reaction to unidentified antigens in the environment.1 Helper T cells (TH1) respond to these environmental antigens in those who are susceptible, which leads to the disease process, but paradoxically, even with the elevation of cellular immune activity at the sites of the granulomatous inflammation, the peripheral immune response in these patients is suppressed as shown by lymphopenia.2
Cutaneous sarcoidosis is found in approximately one-third of patients with systemic sarcoidosis but can occur without systemic involvement.1,2 Sarcoidosis is reported worldwide and affects patients of all races and ethnicities, ages, and sexes but does have a higher prevalence among Black individuals in the United States, patients younger than 40 years (peak incidence, 20–29 years of age), and females.2 In 80% of patients, CS occurs before systemic sarcoidosis develops, or they may develop simultaneously.1
Cutaneous sarcoidosis has a wide range of clinical presentations that are classified as specific and nonspecific. Specific lesions in CS contain noncaseating granulomas while nonspecific lesions in CS appear as reactive processes.2 The most common specific presentation of CS includes papules that are brown in pigmentation in lighter skin tones and red to violaceous in darker skin tones (Figure). The most common nonspecific skin manifestation is erythema nodosum, which represents a hypersensitivity reaction. Cutaneous sarcoidosis can appear as hypopigmented or hyperpigmented patches or plaques.1

Treatments for CS vary based on the individual.1 For milder and more localized cases, topical or intralesional steroids may be used. If systemic sarcoidosis is suspected or if there is diffuse involvement of the skin, systemic steroids, antimalarials (eg, hydroxychloroquine), low-dose methotrexate, minocycline, allopurinol, azathioprine, isotretinoin, tumor necrosis factor α inhibitors, or psoralen plus long-wave UVA radiation may be used. If systemic sarcoidosis is present, referral to a pulmonologist is recommended for co-management.1
Cutaneous sarcoidosis is known as the “great imitator,” and there are multiple diseases to consider in the differential that are distinguished by the physical findings.1 In our case of a middle-aged Black woman with indurated plaques, a few diagnoses to consider were psoriasis, discoid lupus erythematosus (DLE), mycosis fungoides (MF), and tinea infection.
Psoriasis is a common disease, and 90% of patients have chronic plaquelike disease with well-demarcated erythematous plaques that have a silver-gray scale and a positive Auspitz sign (also known as pinpoint bleeding).3 Plaques often are distributed on the trunk, limb extensors, and scalp, along with nail changes. Some patients also have joint pain, indicating psoriatic arthritis. The etiology of psoriasis is unknown, but it develops due to unrestrained keratinocyte proliferation and defective differentiation, which leads to histopathology showing regular acanthosis and papillary dermal ectasia with rouleaux. Mild cases typically are treated with topical steroids or vitamin D, while more severe cases are treated with methotrexate, cyclosporine, retinoids, or biologics.3
Discoid lupus erythematosus occurs 4 times more often in Black patients than in White patients. Clinically, DLE begins as well-defined, erythematous, scaly patches that expand with hyperpigmentation at the periphery and leave an atrophic, scarred, hypopigmented center.4 It typically is localized to the head and neck, but in cases where it disseminates elsewhere on the body, the risk for systemic lupus erythematosus increases from 1.2% to 28%.5 Histopathology of DLE shows vacuolar degeneration of the basal cell layer in the epidermis along with patchy lymphocytic infiltrate in the dermis. Treatments range from topical steroids for mild cases to antimalarial agents, retinoids, anti-inflammatory drugs, and calcineurin inhibitors for more severe cases.4
Although there are multiple types of cutaneous T-cell lymphoma, the most common is MF, which traditionally is nonaggressive. The typical patient with MF is older than 60 years and presents with indolent, ongoing, flat to minimally indurated patches or plaques that have cigarette paper scale. As MF progresses, some plaques grow into tumors and can become more aggressive. Histologically, MF changes based on its clinical stage, with the initial phase showing epidermotropic atypical lymphocytes and later phases showing less epitheliotropic, larger, atypical lymphocytes. The treatment algorithm varies depending on cutaneous T-cell lymphoma staging.6
Tinea infections are caused by dermatophytes. In prepubertal children, they predominantly appear as tinea corporis (on the body) or tinea capitis (on the scalp), but in adults they appear as tinea cruris (on the groin), tinea pedis (on the feet), or tinea unguium (on the nails).7 Tinea infections classically are known to appear as an annular patch with an active erythematous scaling border and central clearing. The patches can be pruritic. Potassium hydroxide preparation of a skin scraping is a quick test to use in the office; if the results are inconclusive, a culture may be required. Treatment depends on the location of the infection but typically involves either topical or oral antifungal agents.7
The Diagnosis: Cutaneous Sarcoidosis
A biopsy of a plaque on the back confirmed cutaneous sarcoidosis (CS). A chest radiograph demonstrated hilar nodes, and a referral was placed for comanagement with a pulmonologist. Histopathology was critical in making the diagnosis, with well-circumscribed noncaseating granulomas present in the dermis. The granulomas in CS often are described as naked, as there are minimal lymphocytes present and plasma cells normally are absent.1 Because the lungs are the most common site of involvement, a chest radiograph is necessary to examine for systemic sarcoidosis. Laboratory workup is used to evaluate for lymphopenia, hypercalcemia, elevated blood sedimentation rate, and elevated angiotensin- converting enzyme levels, which are common in systemic sarcoidosis.1
Sarcoidosis is a multisystemic granulomatous disorder with an unknown etiology. It is believed to develop in genetically predisposed individuals as a reaction to unidentified antigens in the environment.1 Helper T cells (TH1) respond to these environmental antigens in those who are susceptible, which leads to the disease process, but paradoxically, even with the elevation of cellular immune activity at the sites of the granulomatous inflammation, the peripheral immune response in these patients is suppressed as shown by lymphopenia.2
Cutaneous sarcoidosis is found in approximately one-third of patients with systemic sarcoidosis but can occur without systemic involvement.1,2 Sarcoidosis is reported worldwide and affects patients of all races and ethnicities, ages, and sexes but does have a higher prevalence among Black individuals in the United States, patients younger than 40 years (peak incidence, 20–29 years of age), and females.2 In 80% of patients, CS occurs before systemic sarcoidosis develops, or they may develop simultaneously.1
Cutaneous sarcoidosis has a wide range of clinical presentations that are classified as specific and nonspecific. Specific lesions in CS contain noncaseating granulomas while nonspecific lesions in CS appear as reactive processes.2 The most common specific presentation of CS includes papules that are brown in pigmentation in lighter skin tones and red to violaceous in darker skin tones (Figure). The most common nonspecific skin manifestation is erythema nodosum, which represents a hypersensitivity reaction. Cutaneous sarcoidosis can appear as hypopigmented or hyperpigmented patches or plaques.1

Treatments for CS vary based on the individual.1 For milder and more localized cases, topical or intralesional steroids may be used. If systemic sarcoidosis is suspected or if there is diffuse involvement of the skin, systemic steroids, antimalarials (eg, hydroxychloroquine), low-dose methotrexate, minocycline, allopurinol, azathioprine, isotretinoin, tumor necrosis factor α inhibitors, or psoralen plus long-wave UVA radiation may be used. If systemic sarcoidosis is present, referral to a pulmonologist is recommended for co-management.1
Cutaneous sarcoidosis is known as the “great imitator,” and there are multiple diseases to consider in the differential that are distinguished by the physical findings.1 In our case of a middle-aged Black woman with indurated plaques, a few diagnoses to consider were psoriasis, discoid lupus erythematosus (DLE), mycosis fungoides (MF), and tinea infection.
Psoriasis is a common disease, and 90% of patients have chronic plaquelike disease with well-demarcated erythematous plaques that have a silver-gray scale and a positive Auspitz sign (also known as pinpoint bleeding).3 Plaques often are distributed on the trunk, limb extensors, and scalp, along with nail changes. Some patients also have joint pain, indicating psoriatic arthritis. The etiology of psoriasis is unknown, but it develops due to unrestrained keratinocyte proliferation and defective differentiation, which leads to histopathology showing regular acanthosis and papillary dermal ectasia with rouleaux. Mild cases typically are treated with topical steroids or vitamin D, while more severe cases are treated with methotrexate, cyclosporine, retinoids, or biologics.3
Discoid lupus erythematosus occurs 4 times more often in Black patients than in White patients. Clinically, DLE begins as well-defined, erythematous, scaly patches that expand with hyperpigmentation at the periphery and leave an atrophic, scarred, hypopigmented center.4 It typically is localized to the head and neck, but in cases where it disseminates elsewhere on the body, the risk for systemic lupus erythematosus increases from 1.2% to 28%.5 Histopathology of DLE shows vacuolar degeneration of the basal cell layer in the epidermis along with patchy lymphocytic infiltrate in the dermis. Treatments range from topical steroids for mild cases to antimalarial agents, retinoids, anti-inflammatory drugs, and calcineurin inhibitors for more severe cases.4
Although there are multiple types of cutaneous T-cell lymphoma, the most common is MF, which traditionally is nonaggressive. The typical patient with MF is older than 60 years and presents with indolent, ongoing, flat to minimally indurated patches or plaques that have cigarette paper scale. As MF progresses, some plaques grow into tumors and can become more aggressive. Histologically, MF changes based on its clinical stage, with the initial phase showing epidermotropic atypical lymphocytes and later phases showing less epitheliotropic, larger, atypical lymphocytes. The treatment algorithm varies depending on cutaneous T-cell lymphoma staging.6
Tinea infections are caused by dermatophytes. In prepubertal children, they predominantly appear as tinea corporis (on the body) or tinea capitis (on the scalp), but in adults they appear as tinea cruris (on the groin), tinea pedis (on the feet), or tinea unguium (on the nails).7 Tinea infections classically are known to appear as an annular patch with an active erythematous scaling border and central clearing. The patches can be pruritic. Potassium hydroxide preparation of a skin scraping is a quick test to use in the office; if the results are inconclusive, a culture may be required. Treatment depends on the location of the infection but typically involves either topical or oral antifungal agents.7
- Tchernev G, Cardoso JC, Chokoeva AA, et al. The “mystery” of cutaneous sarcoidosis: facts and controversies. Int J Immunopathol Pharmacol. 2014;27:321-330. doi:10.1177/039463201402700302
- Ali MM, Atwan AA, Gonzalez ML. Cutaneous sarcoidosis: updates in the pathogenesis. J Eur Acad Dermatol Venereol. 2010;24:747-755. doi:10.1111/j.1468-3083.2009.03517.x
- Rendon A, Schäkel K. Psoriasis pathogenesis and treatment [published online March 23, 2019]. Int J Mol Sci. 2019;20:1475. doi:10.3390/ijms20061475
- McDaniel B, Sukumaran S, Koritala T, et al. Discoid lupus erythematosus. StatPearls [Internet]. StatPearls Publishing; 2023. Accessed December 11, 2023. https://www.ncbi.nlm.nih.gov/books/NBK493145/
- Bhat MR, Hulmani M, Dandakeri S, et al. Disseminated discoid lupus erythematosus leading to squamous cell carcinoma. Indian J Dermatol. 2012;57:158-161. doi:10.4103/0019-5154.94298
- Pulitzer M. Cutaneous T-cell Lymphoma. Clin Lab Med. 2017; 37:527-546. doi:10.1016/j.cll.2017.06.006
- Ely JW, Rosenfeld S, Seabury Stone M. Diagnosis and management of tinea infections. Am Fam Physician. 2014;90:702-710.
- Tchernev G, Cardoso JC, Chokoeva AA, et al. The “mystery” of cutaneous sarcoidosis: facts and controversies. Int J Immunopathol Pharmacol. 2014;27:321-330. doi:10.1177/039463201402700302
- Ali MM, Atwan AA, Gonzalez ML. Cutaneous sarcoidosis: updates in the pathogenesis. J Eur Acad Dermatol Venereol. 2010;24:747-755. doi:10.1111/j.1468-3083.2009.03517.x
- Rendon A, Schäkel K. Psoriasis pathogenesis and treatment [published online March 23, 2019]. Int J Mol Sci. 2019;20:1475. doi:10.3390/ijms20061475
- McDaniel B, Sukumaran S, Koritala T, et al. Discoid lupus erythematosus. StatPearls [Internet]. StatPearls Publishing; 2023. Accessed December 11, 2023. https://www.ncbi.nlm.nih.gov/books/NBK493145/
- Bhat MR, Hulmani M, Dandakeri S, et al. Disseminated discoid lupus erythematosus leading to squamous cell carcinoma. Indian J Dermatol. 2012;57:158-161. doi:10.4103/0019-5154.94298
- Pulitzer M. Cutaneous T-cell Lymphoma. Clin Lab Med. 2017; 37:527-546. doi:10.1016/j.cll.2017.06.006
- Ely JW, Rosenfeld S, Seabury Stone M. Diagnosis and management of tinea infections. Am Fam Physician. 2014;90:702-710.
A 35-year-old Black woman presented to dermatology as a new patient for evaluation of an asymptomatic rash that had enlarged and spread to involve both the face and back over the last 4 months. She had not tried any treatments. She had no notable medical history and was uncertain of her family history. Physical examination showed indurated, flesh-colored to violaceous plaques around the alar-facial groove (top), nasal tip, chin, and back (bottom). The mucosae and nails were not involved.
Long COVID Has Caused Thousands of US Deaths: New CDC Data
While COVID has now claimed more than 1 million lives in the United States alone, these aren’t the only fatalities caused at least in part by the virus. A small but growing number of Americans are surviving acute infections only to succumb months later to the lingering health problems caused by long COVID.
Much of the attention on long COVID has centered on the sometimes debilitating symptoms that strike people with the condition, with no formal diagnostic tests or standard treatments available, and the effect it has on quality of life. But new figures from the US Centers for Disease Control and Prevention (CDC) show that long COVID can also be deadly.
More than 5000 Americans have died from long COVID since the start of the pandemic, according to new estimates from the CDC.
This total, based on death certificate data collected by the CDC, includes a preliminary tally of 1491 long COVID deaths in 2023 in addition to 3544 fatalities previously reported from January 2020 through June 2022.
Guidance issued in 2023 on how to formally report long COVID as a cause of death on death certificates should help get a more accurate count of these fatalities going forward, said Robert Anderson, PhD, chief mortality statistician for the CDC, Atlanta, Georgia.
“We hope that the guidance will help cause of death certifiers be more aware of the impact of long COVID and more likely to report long COVID as a cause of death when appropriate,” Dr. Anderson said. “That said, we do not expect that this guidance will have a dramatic impact on the trend.”
There’s no standard definition or diagnostic test for long COVID. It’s typically diagnosed when people have symptoms at least 3 months after an acute infection that weren’t present before they got sick. As of the end of last year, about 7% of American adults had experienced long COVID at some point, the CDC estimated in September 2023.
The new death tally indicates long COVID remains a significant public health threat and is likely to grow in the years ahead, even though the pandemic may no longer be considered a global health crisis, experts said.
For example, the death certificate figures indicate:
COVID-19 was the third leading cause of American deaths in 2020 and 2021, and the fourth leading cause of death in the United States in 2023.
Nearly 1% of the more than one million deaths related to COVID-19 since the start of the pandemic have been attributed to long COVID, according to data released by the CDC.
The proportion of COVID-related deaths from long COVID peaked in June 2021 at 1.2% and again in April 2022 at 3.8%, according to the CDC. Both of these peaks coincided with periods of declining fatalities from acute infections.
“I do expect that deaths associated with long COVID will make up an increasingly larger proportion of total deaths associated with COVID-19,” said Mark Czeisler, PhD, a researcher at Harvard Medical School, Boston, Massachusetts, who has studied long COVID fatalities.
Months and even years after an acute infection, long COVID can contribute to serious and potentially life-threatening conditions that impact nearly every major system in the body, according to the CDC guidelines for identifying the condition on death certificates.
This means long COVID may often be listed as an underlying cause of death when people with this condition die of issues related to their heart, lungs, brain or kidneys, the CDC guidelines noted.
The risk for long COVID fatalities remains elevated for at least 6 months for people with milder acute infections and for at least 2 years in severe cases that require hospitalization, some previous research suggested.
As happens with other acute infections, certain people are more at risk for fatal case of long COVID. Age, race, and ethnicity have all been cited as risk factors by researchers who have been tracking the condition since the start of the pandemic.
Half of long COVID fatalities from July 2021 to June 2022 occurred in people aged 65 years and older, and another 23% were recorded among people aged 50-64 years old, according a report from CDC.
Long COVID death rates also varied by race and ethnicity, from a high of 14.1 cases per million among America Indian and Alaskan natives to a low of 1.5 cases per million among Asian people, the CDC found. Death rates per million were 6.7 for White individuals, 6.4 for Black people, and 4.7 for Hispanic people.
The disproportionate share of Black and Hispanic people who developed and died from severe acute infections may have left fewer survivors to develop long COVID, limiting long COVID fatalities among these groups, the CDC report concluded.
It’s also possible that long COVID fatalities were undercounted in these populations because they faced challenges accessing healthcare or seeing providers who could recognize the hallmark symptoms of long COVID.
It’s also difficult to distinguish between how many deaths related to the virus ultimately occur as a result of long COVID rather than acute infections. That’s because it may depend on a variety of factors, including how consistently medical examiners follow the CDC guidelines, said Ziyad Al-Aly, MD, chief of research at the Veterans Affairs, St. Louis Health Care System and a senior clinical epidemiologist at Washington University in St. Louis.
“Long COVID remains massively underdiagnosed, and death in people with long COVID is misattributed to other things,” Dr. Al-Aly said.
An accurate test for long COVID could help lead to a more accurate count of these fatalities, Dr. Czeisler said. Some preliminary research suggests that it might one day be possible to diagnose long COVID with a blood test.
“The timeline for such a test and the extent to which it would be widely applied is uncertain,” Dr. Czeisler noted, “though that would certainly be a gamechanger.”
A version of this article appeared on Medscape.com.
While COVID has now claimed more than 1 million lives in the United States alone, these aren’t the only fatalities caused at least in part by the virus. A small but growing number of Americans are surviving acute infections only to succumb months later to the lingering health problems caused by long COVID.
Much of the attention on long COVID has centered on the sometimes debilitating symptoms that strike people with the condition, with no formal diagnostic tests or standard treatments available, and the effect it has on quality of life. But new figures from the US Centers for Disease Control and Prevention (CDC) show that long COVID can also be deadly.
More than 5000 Americans have died from long COVID since the start of the pandemic, according to new estimates from the CDC.
This total, based on death certificate data collected by the CDC, includes a preliminary tally of 1491 long COVID deaths in 2023 in addition to 3544 fatalities previously reported from January 2020 through June 2022.
Guidance issued in 2023 on how to formally report long COVID as a cause of death on death certificates should help get a more accurate count of these fatalities going forward, said Robert Anderson, PhD, chief mortality statistician for the CDC, Atlanta, Georgia.
“We hope that the guidance will help cause of death certifiers be more aware of the impact of long COVID and more likely to report long COVID as a cause of death when appropriate,” Dr. Anderson said. “That said, we do not expect that this guidance will have a dramatic impact on the trend.”
There’s no standard definition or diagnostic test for long COVID. It’s typically diagnosed when people have symptoms at least 3 months after an acute infection that weren’t present before they got sick. As of the end of last year, about 7% of American adults had experienced long COVID at some point, the CDC estimated in September 2023.
The new death tally indicates long COVID remains a significant public health threat and is likely to grow in the years ahead, even though the pandemic may no longer be considered a global health crisis, experts said.
For example, the death certificate figures indicate:
COVID-19 was the third leading cause of American deaths in 2020 and 2021, and the fourth leading cause of death in the United States in 2023.
Nearly 1% of the more than one million deaths related to COVID-19 since the start of the pandemic have been attributed to long COVID, according to data released by the CDC.
The proportion of COVID-related deaths from long COVID peaked in June 2021 at 1.2% and again in April 2022 at 3.8%, according to the CDC. Both of these peaks coincided with periods of declining fatalities from acute infections.
“I do expect that deaths associated with long COVID will make up an increasingly larger proportion of total deaths associated with COVID-19,” said Mark Czeisler, PhD, a researcher at Harvard Medical School, Boston, Massachusetts, who has studied long COVID fatalities.
Months and even years after an acute infection, long COVID can contribute to serious and potentially life-threatening conditions that impact nearly every major system in the body, according to the CDC guidelines for identifying the condition on death certificates.
This means long COVID may often be listed as an underlying cause of death when people with this condition die of issues related to their heart, lungs, brain or kidneys, the CDC guidelines noted.
The risk for long COVID fatalities remains elevated for at least 6 months for people with milder acute infections and for at least 2 years in severe cases that require hospitalization, some previous research suggested.
As happens with other acute infections, certain people are more at risk for fatal case of long COVID. Age, race, and ethnicity have all been cited as risk factors by researchers who have been tracking the condition since the start of the pandemic.
Half of long COVID fatalities from July 2021 to June 2022 occurred in people aged 65 years and older, and another 23% were recorded among people aged 50-64 years old, according a report from CDC.
Long COVID death rates also varied by race and ethnicity, from a high of 14.1 cases per million among America Indian and Alaskan natives to a low of 1.5 cases per million among Asian people, the CDC found. Death rates per million were 6.7 for White individuals, 6.4 for Black people, and 4.7 for Hispanic people.
The disproportionate share of Black and Hispanic people who developed and died from severe acute infections may have left fewer survivors to develop long COVID, limiting long COVID fatalities among these groups, the CDC report concluded.
It’s also possible that long COVID fatalities were undercounted in these populations because they faced challenges accessing healthcare or seeing providers who could recognize the hallmark symptoms of long COVID.
It’s also difficult to distinguish between how many deaths related to the virus ultimately occur as a result of long COVID rather than acute infections. That’s because it may depend on a variety of factors, including how consistently medical examiners follow the CDC guidelines, said Ziyad Al-Aly, MD, chief of research at the Veterans Affairs, St. Louis Health Care System and a senior clinical epidemiologist at Washington University in St. Louis.
“Long COVID remains massively underdiagnosed, and death in people with long COVID is misattributed to other things,” Dr. Al-Aly said.
An accurate test for long COVID could help lead to a more accurate count of these fatalities, Dr. Czeisler said. Some preliminary research suggests that it might one day be possible to diagnose long COVID with a blood test.
“The timeline for such a test and the extent to which it would be widely applied is uncertain,” Dr. Czeisler noted, “though that would certainly be a gamechanger.”
A version of this article appeared on Medscape.com.
While COVID has now claimed more than 1 million lives in the United States alone, these aren’t the only fatalities caused at least in part by the virus. A small but growing number of Americans are surviving acute infections only to succumb months later to the lingering health problems caused by long COVID.
Much of the attention on long COVID has centered on the sometimes debilitating symptoms that strike people with the condition, with no formal diagnostic tests or standard treatments available, and the effect it has on quality of life. But new figures from the US Centers for Disease Control and Prevention (CDC) show that long COVID can also be deadly.
More than 5000 Americans have died from long COVID since the start of the pandemic, according to new estimates from the CDC.
This total, based on death certificate data collected by the CDC, includes a preliminary tally of 1491 long COVID deaths in 2023 in addition to 3544 fatalities previously reported from January 2020 through June 2022.
Guidance issued in 2023 on how to formally report long COVID as a cause of death on death certificates should help get a more accurate count of these fatalities going forward, said Robert Anderson, PhD, chief mortality statistician for the CDC, Atlanta, Georgia.
“We hope that the guidance will help cause of death certifiers be more aware of the impact of long COVID and more likely to report long COVID as a cause of death when appropriate,” Dr. Anderson said. “That said, we do not expect that this guidance will have a dramatic impact on the trend.”
There’s no standard definition or diagnostic test for long COVID. It’s typically diagnosed when people have symptoms at least 3 months after an acute infection that weren’t present before they got sick. As of the end of last year, about 7% of American adults had experienced long COVID at some point, the CDC estimated in September 2023.
The new death tally indicates long COVID remains a significant public health threat and is likely to grow in the years ahead, even though the pandemic may no longer be considered a global health crisis, experts said.
For example, the death certificate figures indicate:
COVID-19 was the third leading cause of American deaths in 2020 and 2021, and the fourth leading cause of death in the United States in 2023.
Nearly 1% of the more than one million deaths related to COVID-19 since the start of the pandemic have been attributed to long COVID, according to data released by the CDC.
The proportion of COVID-related deaths from long COVID peaked in June 2021 at 1.2% and again in April 2022 at 3.8%, according to the CDC. Both of these peaks coincided with periods of declining fatalities from acute infections.
“I do expect that deaths associated with long COVID will make up an increasingly larger proportion of total deaths associated with COVID-19,” said Mark Czeisler, PhD, a researcher at Harvard Medical School, Boston, Massachusetts, who has studied long COVID fatalities.
Months and even years after an acute infection, long COVID can contribute to serious and potentially life-threatening conditions that impact nearly every major system in the body, according to the CDC guidelines for identifying the condition on death certificates.
This means long COVID may often be listed as an underlying cause of death when people with this condition die of issues related to their heart, lungs, brain or kidneys, the CDC guidelines noted.
The risk for long COVID fatalities remains elevated for at least 6 months for people with milder acute infections and for at least 2 years in severe cases that require hospitalization, some previous research suggested.
As happens with other acute infections, certain people are more at risk for fatal case of long COVID. Age, race, and ethnicity have all been cited as risk factors by researchers who have been tracking the condition since the start of the pandemic.
Half of long COVID fatalities from July 2021 to June 2022 occurred in people aged 65 years and older, and another 23% were recorded among people aged 50-64 years old, according a report from CDC.
Long COVID death rates also varied by race and ethnicity, from a high of 14.1 cases per million among America Indian and Alaskan natives to a low of 1.5 cases per million among Asian people, the CDC found. Death rates per million were 6.7 for White individuals, 6.4 for Black people, and 4.7 for Hispanic people.
The disproportionate share of Black and Hispanic people who developed and died from severe acute infections may have left fewer survivors to develop long COVID, limiting long COVID fatalities among these groups, the CDC report concluded.
It’s also possible that long COVID fatalities were undercounted in these populations because they faced challenges accessing healthcare or seeing providers who could recognize the hallmark symptoms of long COVID.
It’s also difficult to distinguish between how many deaths related to the virus ultimately occur as a result of long COVID rather than acute infections. That’s because it may depend on a variety of factors, including how consistently medical examiners follow the CDC guidelines, said Ziyad Al-Aly, MD, chief of research at the Veterans Affairs, St. Louis Health Care System and a senior clinical epidemiologist at Washington University in St. Louis.
“Long COVID remains massively underdiagnosed, and death in people with long COVID is misattributed to other things,” Dr. Al-Aly said.
An accurate test for long COVID could help lead to a more accurate count of these fatalities, Dr. Czeisler said. Some preliminary research suggests that it might one day be possible to diagnose long COVID with a blood test.
“The timeline for such a test and the extent to which it would be widely applied is uncertain,” Dr. Czeisler noted, “though that would certainly be a gamechanger.”
A version of this article appeared on Medscape.com.
Short Course of Amoxicillin Shows Effectiveness for Febrile UTIs
Use of oral amoxicillin-clavulanic acid for 5 days was noninferior to a 10-day course of treatment among children with noncomplicated febrile urinary tract infections (UTIs), according to new research.
Well-appearing children with febrile UTIs are generally treated with a 10-day course of oral antibiotics, but the effectiveness of a 5-day course has not been evaluated, wrote Giovanni Montini, MD, of the University of Milan, Milan, Italy, and colleagues.
Robert W. Frenck Jr, MD, a director of the Center for Vaccine Research at Cincinnati Children’s Hospital Medical Center, Ohio, said he was not surprised that the shorter course was sufficient to treat these cases. The antibiotic concentration in the urine often significantly exceeds the levels in the blood, he said.
Dr. Frenck, who was not involved in the study, said that he saw no real barriers to the use of a shorter course of therapy in clinical practice.
“I think both parents and the medical team would be happy to be able to use a shorter course of therapy,” he said.
In the study published in Pediatrics , researchers randomized 142 children aged 3 months to 5 years with uncomplicated febrile UTIs to 50 mg/kg/d of amoxicillin-clavulanate for either the short or standard period. The study took place at eight pediatric emergency departments in Italy between May 2020 and September 2022. All patients received prescriptions for 5 days of antibiotics, and those randomized to the standard course received a second prescription after randomization.
The primary endpoint was recurrence of the UTI within 30 days of completion of therapy. Secondary endpoints included clinical recovery at the end of treatment, adverse events related to the therapy, and signs of antibiotic resistance.
The UTI recurrence rate within 30 days of treatment completion was 2.8% in the short-course group and 14.3% in the standard group. A post hoc analysis excluding patients with vesicoureteral reflux and non–Escherichia coli UTIs further confirmed the noninferiority of short-course treatment.
“It is a bit surprising that the short-course group had fewer relapses within 30 days of discontinuing antibiotics,” Dr. Frenck said. “However, the differences may be due to small sample sizes and do not appear to be statistically significant differences in recurrence rates.”
Resolution of symptoms was similar between the short-course and standard groups (97.2% and 92.9%, respectively), and indications of antibiotic resistance were similar between the groups. No adverse events were reported in the standard group, and one case of diarrhea occurred in the short-course group.
The findings were limited by the study’s unblinded randomization, so parents were aware of the trial and were potentially sensitized to look for signs of infection. Researchers also relied on parent reports of adverse drug effects rather than through a standardized questionnaire, the researchers noted.
Dr. Frenck said a potential benefit to shortening treatment is that adherence usually increases.
“But you only want to decrease the length of a course of medicine if you can do so without compromising the effectiveness of the treatment,” Dr. Frenck said.
Dr. Frenck also noted a recent study, which demonstrated that 5 days of antibiotics had equivalent efficacy as 10 days for uncomplicated pneumonia.
“The current paper further demonstrates that shorter courses of antibiotics may be possible for other mild forms of infections.”
Looking ahead, researchers could evaluate the use of short-course antibiotics for other common infections such as otitis media, he noted.
The study was supported by the Ministry of Health, Rome, Italy, in collaboration with the Institute for Maternal and Child Health IRCCS Burlo Garofolo, Trieste, Italy. The researchers report no financial conflicts. Dr. Frenck disclosed conducting clinical trials for Pfizer, Moderna, AstraZeneca, Merck, and GSK; none of those trials were for antibiotics or urinary tract infections.
A version of this article appeared on Medscape.com.
Use of oral amoxicillin-clavulanic acid for 5 days was noninferior to a 10-day course of treatment among children with noncomplicated febrile urinary tract infections (UTIs), according to new research.
Well-appearing children with febrile UTIs are generally treated with a 10-day course of oral antibiotics, but the effectiveness of a 5-day course has not been evaluated, wrote Giovanni Montini, MD, of the University of Milan, Milan, Italy, and colleagues.
Robert W. Frenck Jr, MD, a director of the Center for Vaccine Research at Cincinnati Children’s Hospital Medical Center, Ohio, said he was not surprised that the shorter course was sufficient to treat these cases. The antibiotic concentration in the urine often significantly exceeds the levels in the blood, he said.
Dr. Frenck, who was not involved in the study, said that he saw no real barriers to the use of a shorter course of therapy in clinical practice.
“I think both parents and the medical team would be happy to be able to use a shorter course of therapy,” he said.
In the study published in Pediatrics , researchers randomized 142 children aged 3 months to 5 years with uncomplicated febrile UTIs to 50 mg/kg/d of amoxicillin-clavulanate for either the short or standard period. The study took place at eight pediatric emergency departments in Italy between May 2020 and September 2022. All patients received prescriptions for 5 days of antibiotics, and those randomized to the standard course received a second prescription after randomization.
The primary endpoint was recurrence of the UTI within 30 days of completion of therapy. Secondary endpoints included clinical recovery at the end of treatment, adverse events related to the therapy, and signs of antibiotic resistance.
The UTI recurrence rate within 30 days of treatment completion was 2.8% in the short-course group and 14.3% in the standard group. A post hoc analysis excluding patients with vesicoureteral reflux and non–Escherichia coli UTIs further confirmed the noninferiority of short-course treatment.
“It is a bit surprising that the short-course group had fewer relapses within 30 days of discontinuing antibiotics,” Dr. Frenck said. “However, the differences may be due to small sample sizes and do not appear to be statistically significant differences in recurrence rates.”
Resolution of symptoms was similar between the short-course and standard groups (97.2% and 92.9%, respectively), and indications of antibiotic resistance were similar between the groups. No adverse events were reported in the standard group, and one case of diarrhea occurred in the short-course group.
The findings were limited by the study’s unblinded randomization, so parents were aware of the trial and were potentially sensitized to look for signs of infection. Researchers also relied on parent reports of adverse drug effects rather than through a standardized questionnaire, the researchers noted.
Dr. Frenck said a potential benefit to shortening treatment is that adherence usually increases.
“But you only want to decrease the length of a course of medicine if you can do so without compromising the effectiveness of the treatment,” Dr. Frenck said.
Dr. Frenck also noted a recent study, which demonstrated that 5 days of antibiotics had equivalent efficacy as 10 days for uncomplicated pneumonia.
“The current paper further demonstrates that shorter courses of antibiotics may be possible for other mild forms of infections.”
Looking ahead, researchers could evaluate the use of short-course antibiotics for other common infections such as otitis media, he noted.
The study was supported by the Ministry of Health, Rome, Italy, in collaboration with the Institute for Maternal and Child Health IRCCS Burlo Garofolo, Trieste, Italy. The researchers report no financial conflicts. Dr. Frenck disclosed conducting clinical trials for Pfizer, Moderna, AstraZeneca, Merck, and GSK; none of those trials were for antibiotics or urinary tract infections.
A version of this article appeared on Medscape.com.
Use of oral amoxicillin-clavulanic acid for 5 days was noninferior to a 10-day course of treatment among children with noncomplicated febrile urinary tract infections (UTIs), according to new research.
Well-appearing children with febrile UTIs are generally treated with a 10-day course of oral antibiotics, but the effectiveness of a 5-day course has not been evaluated, wrote Giovanni Montini, MD, of the University of Milan, Milan, Italy, and colleagues.
Robert W. Frenck Jr, MD, a director of the Center for Vaccine Research at Cincinnati Children’s Hospital Medical Center, Ohio, said he was not surprised that the shorter course was sufficient to treat these cases. The antibiotic concentration in the urine often significantly exceeds the levels in the blood, he said.
Dr. Frenck, who was not involved in the study, said that he saw no real barriers to the use of a shorter course of therapy in clinical practice.
“I think both parents and the medical team would be happy to be able to use a shorter course of therapy,” he said.
In the study published in Pediatrics , researchers randomized 142 children aged 3 months to 5 years with uncomplicated febrile UTIs to 50 mg/kg/d of amoxicillin-clavulanate for either the short or standard period. The study took place at eight pediatric emergency departments in Italy between May 2020 and September 2022. All patients received prescriptions for 5 days of antibiotics, and those randomized to the standard course received a second prescription after randomization.
The primary endpoint was recurrence of the UTI within 30 days of completion of therapy. Secondary endpoints included clinical recovery at the end of treatment, adverse events related to the therapy, and signs of antibiotic resistance.
The UTI recurrence rate within 30 days of treatment completion was 2.8% in the short-course group and 14.3% in the standard group. A post hoc analysis excluding patients with vesicoureteral reflux and non–Escherichia coli UTIs further confirmed the noninferiority of short-course treatment.
“It is a bit surprising that the short-course group had fewer relapses within 30 days of discontinuing antibiotics,” Dr. Frenck said. “However, the differences may be due to small sample sizes and do not appear to be statistically significant differences in recurrence rates.”
Resolution of symptoms was similar between the short-course and standard groups (97.2% and 92.9%, respectively), and indications of antibiotic resistance were similar between the groups. No adverse events were reported in the standard group, and one case of diarrhea occurred in the short-course group.
The findings were limited by the study’s unblinded randomization, so parents were aware of the trial and were potentially sensitized to look for signs of infection. Researchers also relied on parent reports of adverse drug effects rather than through a standardized questionnaire, the researchers noted.
Dr. Frenck said a potential benefit to shortening treatment is that adherence usually increases.
“But you only want to decrease the length of a course of medicine if you can do so without compromising the effectiveness of the treatment,” Dr. Frenck said.
Dr. Frenck also noted a recent study, which demonstrated that 5 days of antibiotics had equivalent efficacy as 10 days for uncomplicated pneumonia.
“The current paper further demonstrates that shorter courses of antibiotics may be possible for other mild forms of infections.”
Looking ahead, researchers could evaluate the use of short-course antibiotics for other common infections such as otitis media, he noted.
The study was supported by the Ministry of Health, Rome, Italy, in collaboration with the Institute for Maternal and Child Health IRCCS Burlo Garofolo, Trieste, Italy. The researchers report no financial conflicts. Dr. Frenck disclosed conducting clinical trials for Pfizer, Moderna, AstraZeneca, Merck, and GSK; none of those trials were for antibiotics or urinary tract infections.
A version of this article appeared on Medscape.com.
FROM PEDIATRICS
Hospital Adverse Events Rise After Private Equity Acquisition
Hospital-acquired adverse events or conditions including falls and infections increased by approximately 25% after hospitals’ acquisition by private equity compared with control hospitals, on the basis of a study of Medicare claims for more than 4,500,000 hospitalizations.
“Prior research on private equity in health care showed that acquisition is associated with higher charges, prices, and spending; however, the implications for quality of care and patient outcomes remained less understood,” corresponding author Zirui Song, MD, of Harvard Medical School, Boston, said in an interview. “This was particularly true for measures of clinical quality that were less susceptible to changes in patient mix or coding behavior, such as hospital-acquired adverse events.”
In the study, published in JAMA, the researchers compared data from 100% Medicare Part A claims for 662,095 hospitalizations at 51 hospitals acquired by private equities and 4,160,720 hospitalizations at 259 control hospitals. The hospitalizations occurred between 2009 and 2019. The researchers also used a difference-in-differences design to evaluate hospitalizations from 3 years before to 3 years after acquisition, controlling for patient and hospital attributes.
Hospital-acquired adverse events as defined by the US Centers for Medicare & Medicaid Services included falls, infections, stage III or IV pressure ulcers, foreign objects retained after surgery, air embolism, and blood incompatibility.
Overall, Medicare patients in private equity hospitals experienced a 25.4% increase in hospital-acquired conditions compared with those in control hospitals through a period of up to 3 years after acquisition, with a difference of 4.6 additional hospital-acquired conditions per 10,000 hospitalizations (P = .004). Central line-associated bloodstream infections accounted for 37.7% of the increase (P = .04), despite a 16.2% decrease in placement of central lines, and falls accounted for 27.3% (P = .02).
Notably, the incidence of surgical site infections increased from 10.8 per 10,000 hospitalizations before acquisition to 21.6 per 10,000 hospitalizations after acquisition, despite a reduction of 8.1% in surgical volume. By contrast, surgical site infections decreased at control hospitals over the study period.
In-hospital mortality decreased slightly at private equity hospitals compared with the control hospitals, but there was no differential change in mortality by 30 days after hospital discharge. The slight difference might be caused by the trend in slightly younger Medicare beneficiaries treated at private equity hospitals; these patients were less likely to be eligible for both Medicaid and Medicare and were more likely to be transferred to other hospitals, the researchers noted.
The findings were limited by several factors including the lack of generalizability to all private equity-acquired hospitals and to non-Medicare patients, the researchers noted. Other limitations include the use of the International Classification of Diseases, Ninth Revision (ICD-9) and Tenth Revision (ICD-10) codes that might have failed to capture all hospital-acquired conditions and the inability to account for all confounding factors.
However, the results suggest that private equity acquisition was associated with increased hospital-acquired adverse events and highlight concerns about the impact of private equity ownership on healthcare delivery, the researchers concluded.
In a related story published in July 2023, this news organization described a report showing an association between private equity ownership of medical practices and increased consumer prices for multiple medical specialties.
“Medicare patients admitted to private equity-owned hospitals experienced, on average, an 25% increase in hospital-acquired adverse events after the hospital was bought compared to similar patients at hospitals not acquired by private equity firms. We were surprised by the extent of this change relative to the comparison (non-private equity) hospitals, including the sizable increase in central line-associated bloodstream infections and the doubling of surgical site infections at private equity hospitals — both of which went down at the comparison hospitals during the same period,” Dr. Song said in an interview.
“A key implication is that patients, providers, and policymakers might be more attuned to the potential clinical impact of private equity ownership in the delivery system. Given that a plausible explanation for these findings is reductions in clinician staffing, clinical organizations and policymakers might also be more aware of cost-cutting strategies after acquisition,” Dr. Song said. “Prior research has shown that hospitals, nursing homes, and physician practices experience staffing cuts after private equity acquisition, which is a common way to reduce operating costs and boost the profitability of acquired entities,” he noted.
“More research is needed to understand the impact of private equity acquisitions across health care settings and the potential effects of policy levers that aim to protect patients and societal resources,” said Dr. Song, who coauthored an article outlining a policy framework for addressing private equity in healthcare, published in JAMA in April 2023. “Potential regulatory remedies include minimum staffing ratios, antitrust enforcement, mitigating the financial risk of such acquisitions, increasing the transparency of these acquisitions, and protecting patients and society from the higher prices of care attributed to this model of provider ownership,” he said.
Patients Pay the Price of Private Equity Acquisition
“The exponential growth in private equity ownership in hospital and physician practices in the past few decades has left a majority of health care providers disillusioned with cost-cutting practices resulting in staffing reductions and ratios that sacrifice patient care as part of their approach to running clinical operations ‘lean,’ ” Robert Glatter, MD, an emergency medicine physician at Lenox Hill Hospital, New York, NY, said in an interview.
“While private equity companies argue that such practices are essential to meet their bottom line and increase operating margins, it doesn’t translate into ideal care for patients; lean practices in staffing which focus on profits at the expense of patient safety and quality of care.
“When you look at patient outcomes, it is the patients who ultimately pay the price — not the shareholders,” Dr. Glatter said. “This translates to higher risks of hospital-acquired complications including falls and blood-borne infections, including surgical site infections, as noted by the authors of the current study when private equity took over operations in hospitals.
Dr. Glatter said he was not surprised by the findings. “In my world, patient care and safety come first. Period,” he said. “Would you want your family’s health and well-being sacrificed in the name of company profits? I think it’s a rhetorical question, but one that every health care provider who works in a hospital or practice run by private equity must consider.”
Despite a decline in utilization at private equity hospitals as noted in the current study, hospital-acquired infections and adverse outcomes still increased, illustrating a decline in quality of care, said Dr. Glatter. “While these disparities were not evident when looking at 30-day outcomes, they demonstrate how operational changes impact patient outcomes in the near term. Having younger and healthier patients, and fewer Medicare and Medicaid patients combined with more hospital transfers to non–private equity run hospitals, resulted in lower in-hospital mortality in the near term, which was not apparent at 30 days post discharge,” he said.
“The explosion of hospital mergers and consolidation in the past several decades has led to skyrocketing health care costs at the expense of patient satisfaction, but also health care providers’ autonomy to manage and maintain quality care for their patients,” Dr. Glatter said.
“It’s important to understand that private equity’s interests are primarily aligned with their shareholder’s interests, as opposed to patients’ outcomes and interests,” Dr. Glatter told this news organization. “Within 5-7 years, the goal is to increase operating margins and profits and then sell a practice or hospital, which is ultimately part of a ‘health care portfolio,’ ” he said.
Additional research is needed to examine whether other hospital-acquired conditions including pressure sores, catheter-associated UTIs, methicillin-resistant Staphylococcus aureus infections, Clostridium difficile infections, and nosocomial pneumonia have increased in hospitals following private equity acquisition, given the overall national decline in these events, he said.
“At the same time, it is vital to also look at management and readmission rates for patients with strokes, heart attacks, and congestive heart failure in hospitals that are run by private equity,” Dr. Glatter noted. “These are important benchmarks of care monitored by CMS that reflect the quality of care that payers ultimately factor into reimbursement.”
Examining the metrics associated with these diagnoses will help in understanding whether private equity-managed facilities are leading to adverse outcomes and mortality, increased length of stay, hospital readmissions, and increased nosocomial infections, apart from other aspects of patient experience, Dr. Glatter added.
The study was supported by the National Heart, Lung, and Blood Institute, the National Institute on Aging, and Arnold Ventures. The researchers had no financial conflicts to disclose. Dr. Glatter had no financial conflicts to disclose and serves on the Medscape Emergency Medicine Editorial Board.
A version of this article appeared on Medscape.com.
Hospital-acquired adverse events or conditions including falls and infections increased by approximately 25% after hospitals’ acquisition by private equity compared with control hospitals, on the basis of a study of Medicare claims for more than 4,500,000 hospitalizations.
“Prior research on private equity in health care showed that acquisition is associated with higher charges, prices, and spending; however, the implications for quality of care and patient outcomes remained less understood,” corresponding author Zirui Song, MD, of Harvard Medical School, Boston, said in an interview. “This was particularly true for measures of clinical quality that were less susceptible to changes in patient mix or coding behavior, such as hospital-acquired adverse events.”
In the study, published in JAMA, the researchers compared data from 100% Medicare Part A claims for 662,095 hospitalizations at 51 hospitals acquired by private equities and 4,160,720 hospitalizations at 259 control hospitals. The hospitalizations occurred between 2009 and 2019. The researchers also used a difference-in-differences design to evaluate hospitalizations from 3 years before to 3 years after acquisition, controlling for patient and hospital attributes.
Hospital-acquired adverse events as defined by the US Centers for Medicare & Medicaid Services included falls, infections, stage III or IV pressure ulcers, foreign objects retained after surgery, air embolism, and blood incompatibility.
Overall, Medicare patients in private equity hospitals experienced a 25.4% increase in hospital-acquired conditions compared with those in control hospitals through a period of up to 3 years after acquisition, with a difference of 4.6 additional hospital-acquired conditions per 10,000 hospitalizations (P = .004). Central line-associated bloodstream infections accounted for 37.7% of the increase (P = .04), despite a 16.2% decrease in placement of central lines, and falls accounted for 27.3% (P = .02).
Notably, the incidence of surgical site infections increased from 10.8 per 10,000 hospitalizations before acquisition to 21.6 per 10,000 hospitalizations after acquisition, despite a reduction of 8.1% in surgical volume. By contrast, surgical site infections decreased at control hospitals over the study period.
In-hospital mortality decreased slightly at private equity hospitals compared with the control hospitals, but there was no differential change in mortality by 30 days after hospital discharge. The slight difference might be caused by the trend in slightly younger Medicare beneficiaries treated at private equity hospitals; these patients were less likely to be eligible for both Medicaid and Medicare and were more likely to be transferred to other hospitals, the researchers noted.
The findings were limited by several factors including the lack of generalizability to all private equity-acquired hospitals and to non-Medicare patients, the researchers noted. Other limitations include the use of the International Classification of Diseases, Ninth Revision (ICD-9) and Tenth Revision (ICD-10) codes that might have failed to capture all hospital-acquired conditions and the inability to account for all confounding factors.
However, the results suggest that private equity acquisition was associated with increased hospital-acquired adverse events and highlight concerns about the impact of private equity ownership on healthcare delivery, the researchers concluded.
In a related story published in July 2023, this news organization described a report showing an association between private equity ownership of medical practices and increased consumer prices for multiple medical specialties.
“Medicare patients admitted to private equity-owned hospitals experienced, on average, an 25% increase in hospital-acquired adverse events after the hospital was bought compared to similar patients at hospitals not acquired by private equity firms. We were surprised by the extent of this change relative to the comparison (non-private equity) hospitals, including the sizable increase in central line-associated bloodstream infections and the doubling of surgical site infections at private equity hospitals — both of which went down at the comparison hospitals during the same period,” Dr. Song said in an interview.
“A key implication is that patients, providers, and policymakers might be more attuned to the potential clinical impact of private equity ownership in the delivery system. Given that a plausible explanation for these findings is reductions in clinician staffing, clinical organizations and policymakers might also be more aware of cost-cutting strategies after acquisition,” Dr. Song said. “Prior research has shown that hospitals, nursing homes, and physician practices experience staffing cuts after private equity acquisition, which is a common way to reduce operating costs and boost the profitability of acquired entities,” he noted.
“More research is needed to understand the impact of private equity acquisitions across health care settings and the potential effects of policy levers that aim to protect patients and societal resources,” said Dr. Song, who coauthored an article outlining a policy framework for addressing private equity in healthcare, published in JAMA in April 2023. “Potential regulatory remedies include minimum staffing ratios, antitrust enforcement, mitigating the financial risk of such acquisitions, increasing the transparency of these acquisitions, and protecting patients and society from the higher prices of care attributed to this model of provider ownership,” he said.
Patients Pay the Price of Private Equity Acquisition
“The exponential growth in private equity ownership in hospital and physician practices in the past few decades has left a majority of health care providers disillusioned with cost-cutting practices resulting in staffing reductions and ratios that sacrifice patient care as part of their approach to running clinical operations ‘lean,’ ” Robert Glatter, MD, an emergency medicine physician at Lenox Hill Hospital, New York, NY, said in an interview.
“While private equity companies argue that such practices are essential to meet their bottom line and increase operating margins, it doesn’t translate into ideal care for patients; lean practices in staffing which focus on profits at the expense of patient safety and quality of care.
“When you look at patient outcomes, it is the patients who ultimately pay the price — not the shareholders,” Dr. Glatter said. “This translates to higher risks of hospital-acquired complications including falls and blood-borne infections, including surgical site infections, as noted by the authors of the current study when private equity took over operations in hospitals.
Dr. Glatter said he was not surprised by the findings. “In my world, patient care and safety come first. Period,” he said. “Would you want your family’s health and well-being sacrificed in the name of company profits? I think it’s a rhetorical question, but one that every health care provider who works in a hospital or practice run by private equity must consider.”
Despite a decline in utilization at private equity hospitals as noted in the current study, hospital-acquired infections and adverse outcomes still increased, illustrating a decline in quality of care, said Dr. Glatter. “While these disparities were not evident when looking at 30-day outcomes, they demonstrate how operational changes impact patient outcomes in the near term. Having younger and healthier patients, and fewer Medicare and Medicaid patients combined with more hospital transfers to non–private equity run hospitals, resulted in lower in-hospital mortality in the near term, which was not apparent at 30 days post discharge,” he said.
“The explosion of hospital mergers and consolidation in the past several decades has led to skyrocketing health care costs at the expense of patient satisfaction, but also health care providers’ autonomy to manage and maintain quality care for their patients,” Dr. Glatter said.
“It’s important to understand that private equity’s interests are primarily aligned with their shareholder’s interests, as opposed to patients’ outcomes and interests,” Dr. Glatter told this news organization. “Within 5-7 years, the goal is to increase operating margins and profits and then sell a practice or hospital, which is ultimately part of a ‘health care portfolio,’ ” he said.
Additional research is needed to examine whether other hospital-acquired conditions including pressure sores, catheter-associated UTIs, methicillin-resistant Staphylococcus aureus infections, Clostridium difficile infections, and nosocomial pneumonia have increased in hospitals following private equity acquisition, given the overall national decline in these events, he said.
“At the same time, it is vital to also look at management and readmission rates for patients with strokes, heart attacks, and congestive heart failure in hospitals that are run by private equity,” Dr. Glatter noted. “These are important benchmarks of care monitored by CMS that reflect the quality of care that payers ultimately factor into reimbursement.”
Examining the metrics associated with these diagnoses will help in understanding whether private equity-managed facilities are leading to adverse outcomes and mortality, increased length of stay, hospital readmissions, and increased nosocomial infections, apart from other aspects of patient experience, Dr. Glatter added.
The study was supported by the National Heart, Lung, and Blood Institute, the National Institute on Aging, and Arnold Ventures. The researchers had no financial conflicts to disclose. Dr. Glatter had no financial conflicts to disclose and serves on the Medscape Emergency Medicine Editorial Board.
A version of this article appeared on Medscape.com.
Hospital-acquired adverse events or conditions including falls and infections increased by approximately 25% after hospitals’ acquisition by private equity compared with control hospitals, on the basis of a study of Medicare claims for more than 4,500,000 hospitalizations.
“Prior research on private equity in health care showed that acquisition is associated with higher charges, prices, and spending; however, the implications for quality of care and patient outcomes remained less understood,” corresponding author Zirui Song, MD, of Harvard Medical School, Boston, said in an interview. “This was particularly true for measures of clinical quality that were less susceptible to changes in patient mix or coding behavior, such as hospital-acquired adverse events.”
In the study, published in JAMA, the researchers compared data from 100% Medicare Part A claims for 662,095 hospitalizations at 51 hospitals acquired by private equities and 4,160,720 hospitalizations at 259 control hospitals. The hospitalizations occurred between 2009 and 2019. The researchers also used a difference-in-differences design to evaluate hospitalizations from 3 years before to 3 years after acquisition, controlling for patient and hospital attributes.
Hospital-acquired adverse events as defined by the US Centers for Medicare & Medicaid Services included falls, infections, stage III or IV pressure ulcers, foreign objects retained after surgery, air embolism, and blood incompatibility.
Overall, Medicare patients in private equity hospitals experienced a 25.4% increase in hospital-acquired conditions compared with those in control hospitals through a period of up to 3 years after acquisition, with a difference of 4.6 additional hospital-acquired conditions per 10,000 hospitalizations (P = .004). Central line-associated bloodstream infections accounted for 37.7% of the increase (P = .04), despite a 16.2% decrease in placement of central lines, and falls accounted for 27.3% (P = .02).
Notably, the incidence of surgical site infections increased from 10.8 per 10,000 hospitalizations before acquisition to 21.6 per 10,000 hospitalizations after acquisition, despite a reduction of 8.1% in surgical volume. By contrast, surgical site infections decreased at control hospitals over the study period.
In-hospital mortality decreased slightly at private equity hospitals compared with the control hospitals, but there was no differential change in mortality by 30 days after hospital discharge. The slight difference might be caused by the trend in slightly younger Medicare beneficiaries treated at private equity hospitals; these patients were less likely to be eligible for both Medicaid and Medicare and were more likely to be transferred to other hospitals, the researchers noted.
The findings were limited by several factors including the lack of generalizability to all private equity-acquired hospitals and to non-Medicare patients, the researchers noted. Other limitations include the use of the International Classification of Diseases, Ninth Revision (ICD-9) and Tenth Revision (ICD-10) codes that might have failed to capture all hospital-acquired conditions and the inability to account for all confounding factors.
However, the results suggest that private equity acquisition was associated with increased hospital-acquired adverse events and highlight concerns about the impact of private equity ownership on healthcare delivery, the researchers concluded.
In a related story published in July 2023, this news organization described a report showing an association between private equity ownership of medical practices and increased consumer prices for multiple medical specialties.
“Medicare patients admitted to private equity-owned hospitals experienced, on average, an 25% increase in hospital-acquired adverse events after the hospital was bought compared to similar patients at hospitals not acquired by private equity firms. We were surprised by the extent of this change relative to the comparison (non-private equity) hospitals, including the sizable increase in central line-associated bloodstream infections and the doubling of surgical site infections at private equity hospitals — both of which went down at the comparison hospitals during the same period,” Dr. Song said in an interview.
“A key implication is that patients, providers, and policymakers might be more attuned to the potential clinical impact of private equity ownership in the delivery system. Given that a plausible explanation for these findings is reductions in clinician staffing, clinical organizations and policymakers might also be more aware of cost-cutting strategies after acquisition,” Dr. Song said. “Prior research has shown that hospitals, nursing homes, and physician practices experience staffing cuts after private equity acquisition, which is a common way to reduce operating costs and boost the profitability of acquired entities,” he noted.
“More research is needed to understand the impact of private equity acquisitions across health care settings and the potential effects of policy levers that aim to protect patients and societal resources,” said Dr. Song, who coauthored an article outlining a policy framework for addressing private equity in healthcare, published in JAMA in April 2023. “Potential regulatory remedies include minimum staffing ratios, antitrust enforcement, mitigating the financial risk of such acquisitions, increasing the transparency of these acquisitions, and protecting patients and society from the higher prices of care attributed to this model of provider ownership,” he said.
Patients Pay the Price of Private Equity Acquisition
“The exponential growth in private equity ownership in hospital and physician practices in the past few decades has left a majority of health care providers disillusioned with cost-cutting practices resulting in staffing reductions and ratios that sacrifice patient care as part of their approach to running clinical operations ‘lean,’ ” Robert Glatter, MD, an emergency medicine physician at Lenox Hill Hospital, New York, NY, said in an interview.
“While private equity companies argue that such practices are essential to meet their bottom line and increase operating margins, it doesn’t translate into ideal care for patients; lean practices in staffing which focus on profits at the expense of patient safety and quality of care.
“When you look at patient outcomes, it is the patients who ultimately pay the price — not the shareholders,” Dr. Glatter said. “This translates to higher risks of hospital-acquired complications including falls and blood-borne infections, including surgical site infections, as noted by the authors of the current study when private equity took over operations in hospitals.
Dr. Glatter said he was not surprised by the findings. “In my world, patient care and safety come first. Period,” he said. “Would you want your family’s health and well-being sacrificed in the name of company profits? I think it’s a rhetorical question, but one that every health care provider who works in a hospital or practice run by private equity must consider.”
Despite a decline in utilization at private equity hospitals as noted in the current study, hospital-acquired infections and adverse outcomes still increased, illustrating a decline in quality of care, said Dr. Glatter. “While these disparities were not evident when looking at 30-day outcomes, they demonstrate how operational changes impact patient outcomes in the near term. Having younger and healthier patients, and fewer Medicare and Medicaid patients combined with more hospital transfers to non–private equity run hospitals, resulted in lower in-hospital mortality in the near term, which was not apparent at 30 days post discharge,” he said.
“The explosion of hospital mergers and consolidation in the past several decades has led to skyrocketing health care costs at the expense of patient satisfaction, but also health care providers’ autonomy to manage and maintain quality care for their patients,” Dr. Glatter said.
“It’s important to understand that private equity’s interests are primarily aligned with their shareholder’s interests, as opposed to patients’ outcomes and interests,” Dr. Glatter told this news organization. “Within 5-7 years, the goal is to increase operating margins and profits and then sell a practice or hospital, which is ultimately part of a ‘health care portfolio,’ ” he said.
Additional research is needed to examine whether other hospital-acquired conditions including pressure sores, catheter-associated UTIs, methicillin-resistant Staphylococcus aureus infections, Clostridium difficile infections, and nosocomial pneumonia have increased in hospitals following private equity acquisition, given the overall national decline in these events, he said.
“At the same time, it is vital to also look at management and readmission rates for patients with strokes, heart attacks, and congestive heart failure in hospitals that are run by private equity,” Dr. Glatter noted. “These are important benchmarks of care monitored by CMS that reflect the quality of care that payers ultimately factor into reimbursement.”
Examining the metrics associated with these diagnoses will help in understanding whether private equity-managed facilities are leading to adverse outcomes and mortality, increased length of stay, hospital readmissions, and increased nosocomial infections, apart from other aspects of patient experience, Dr. Glatter added.
The study was supported by the National Heart, Lung, and Blood Institute, the National Institute on Aging, and Arnold Ventures. The researchers had no financial conflicts to disclose. Dr. Glatter had no financial conflicts to disclose and serves on the Medscape Emergency Medicine Editorial Board.
A version of this article appeared on Medscape.com.
Migratory Nodules in a Traveler
The Diagnosis: Gnathostomiasis
The biopsy demonstrated a dense, eosinophilic, granulomatous infiltrate surrounding sections of a parasite with skeletal muscle bundles and intestines containing a brush border and luminal debris (Figure), which was consistent with a diagnosis of gnathostomiasis. Upon further questioning, he revealed that while in Peru he frequently consumed ceviche, which is a dish typically made from fresh raw fish cured in lemon or lime juice. He subsequently was treated with oral ivermectin 0.2 mg/kg once daily for 2 days with no evidence of recurrence 12 months later.
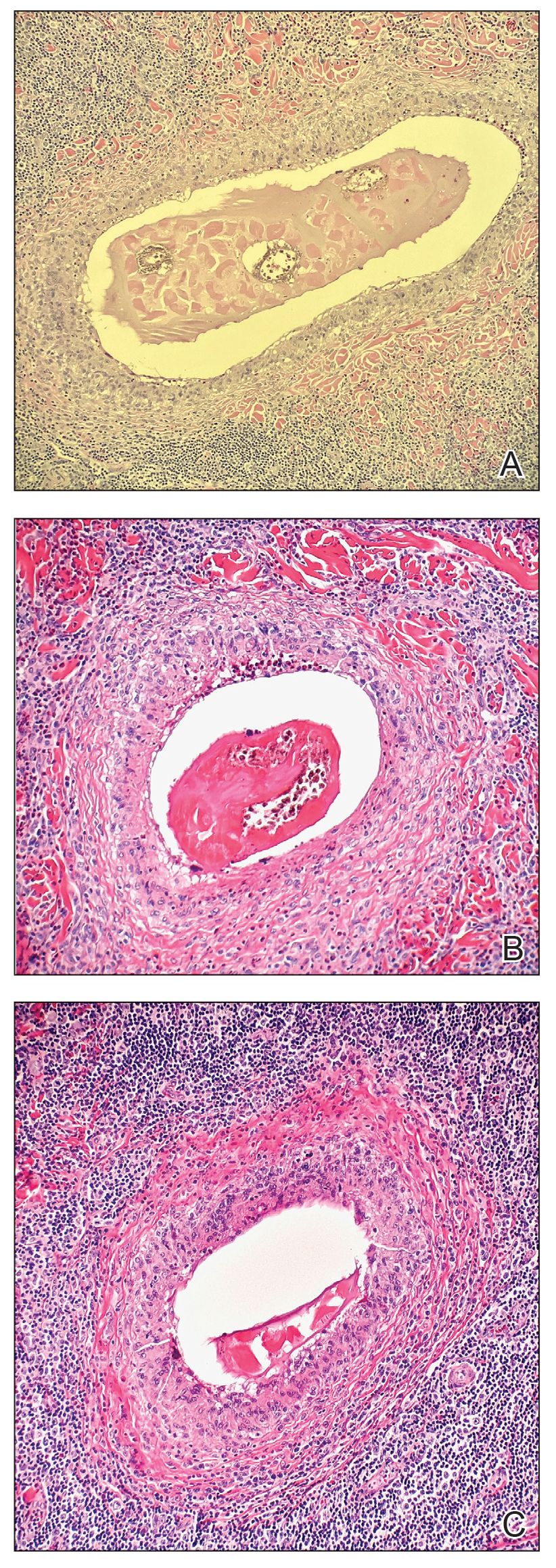
Cutaneous gnathostomiasis is the most common manifestation of infection caused by the third-stage larvae of the genus Gnathostoma. The nematode is endemic to tropical and subtropical regions of Japan and Southeast Asia, particularly Thailand. The disease has been increasingly observed in Central and South America. Humans can become infected through ingestion of undercooked meats, particularly freshwater fish but also poultry, snakes, or frogs. Few cases have been reported in North America and Europe presumably due to more stringent regulations governing the sourcing and storage of fish for consumption.1-3 Restaurants in endemic regions also may use cheaper local freshwater or brackish fish compared to restaurants in the West, which use more expensive saltwater fish that do not harbor Gnathostoma species.1 There is a false belief among restauranteurs and consumers that the larvae can be reliably killed by marinating meat in citrus juice or with concurrent consumption of alcohol or hot spices.2 Adequately cooking or freezing meat to 20 °C for 3 to 5 days are the only effective ways to ensure that the larvae are killed.1-3
The parasite requires its natural definitive hosts—fish-eating mammals such as pigs, cats, and dogs—to complete its life cycle and reproduce. Humans are accidental hosts in whom the parasite fails to reach sexual maturity.1-3 Consequently, symptoms commonly are due to the migration of only 1 larva, but occasionally infection with 2 or more has been observed.1,4
Human infection initially may result in malaise, fever, anorexia, abdominal pain, nausea, vomiting, and diarrhea as the parasite migrates through the stomach, intestines, and liver. After 2 to 4 weeks, larvae may reach the skin where they most commonly create ill-defined, erythematous, indurated, round or oval plaques or nodules described as nodular migratory panniculitis. These lesions tend to develop on the trunk or arms and correspond to the location of the migrating worm.1,3,5 The larvae have been observed to migrate at 1 cm/h.6 Symptoms often wax and wane, with individual nodules lasting approximately 1 to 2 weeks. Uniquely, larval migration can result in a trail of subcutaneous hemorrhage that is considered pathognomonic and helps to differentiate gnathostomiasis from other forms of parasitosis such as strongyloidiasis and sparganosis.1,3 Larvae are highly motile and invasive, and they are capable of producing a wide range of symptoms affecting virtually any part of the body.1,2 Depending on the anatomic location of the migrating worm, infection also may result in neurologic, gastrointestinal, pulmonary, or ocular symptoms.1-3,7 Eosinophilia is common but can subside in the chronic stage, as seen in our patient.1
The classic triad of intermittent migratory nodules, eosinophilia, and a history of travel to Southeast Asia or another endemic region should raise suspicion for gnathostomiasis.1-3,5,7 Unfortunately, confirmatory testing such as Gnathostoma serology is not readily available in the United States, and available serologic tests demonstrate frequent false positives and incomplete crossreactivity.1,2,8 Accordingly, the diagnosis most commonly is solidified by combining cardinal clinical features with histologic findings of a dense eosinophilic inflammatory infiltrate involving the dermis and hypodermis.2,5 In one study, the larva itself was only found in 12 of 66 (18%) skin biopsy specimens from patients with gnathostomiasis.5 If the larva is detected within the sections, it ranges from 2.5 to 12.5 mm in length and 0.4 to 1.2 mm in width and can exhibit cuticular spines, intestinal cells, and characteristic large lateral chords.1,5
The treatment of choice is surgical removal of the worm. Oral albendazole (400–800 mg/d for 21 days) also is considered a first-line treatment and results in clinical cure in approximately 90% of cases. Two doses of oral ivermectin (0.2 mg/kg) spaced 24 to 48 hours apart is an acceptable alternative with comparable efficacy.1-3 Care should be taken if involvement of the central nervous system is suspected, as antihelminthic treatment theoretically could be deleterious due to an inflammatory response to the dying larvae.1,2,9
In the differential diagnosis, loiasis can resemble gnathostomiasis, but the former is endemic to Africa.3 Cutaneous larva migrans most frequently is caused by hookworms from the genus Ancylostoma, which classically leads to superficial serpiginous linear plaques that migrate at a rate of several millimeters per day. However, the larvae are believed to lack the collagenase enzyme required to penetrate the epidermal basement membrane and thus are not capable of producing deep-seated nodules or visceral symptoms.3 Strongyloidiasis (larva currens) generally exhibits a more linear morphology, and infection would result in positive Strongyloides serology.7 Erythema nodosum is a septal panniculitis that can be triggered by infection, pregnancy, medications, connective tissue diseases, inflammatory conditions, and underlying malignancy.10
- Herman JS, Chiodini PL. Gnathostomiasis, another emerging imported disease. Clin Microbiol Rev. 2009;22:484-492.
- Liu GH, Sun MM, Elsheikha HM, et al. Human gnathostomiasis: a neglected food-borne zoonosis. Parasit Vectors. 2020;13:616.
- Tyring SK. Gnathostomiasis. In: Tyring SK, Lupi O, Hengge UR, eds. Tropical Dermatology. 2nd ed. Elsevier; 2017:77-78.
- Rusnak JM, Lucey DR. Clinical gnathostomiasis: case report and review of the English-language literature. Clin Infect Dis. 1993;16:33-50.
- Magaña M, Messina M, Bustamante F, et al. Gnathostomiasis: clinicopathologic study. Am J Dermatopathol. 2004;26:91-95.
- Chandenier J, Husson J, Canaple S, et al. Medullary gnathostomiasis in a white patient: use of immunodiagnosis and magnetic resonance imaging. Clin Infect Dis. 2001;32:E154-E157.
- Hamilton WL, Agranoff D. Imported gnathostomiasis manifesting as cutaneous larva migrans and Löffler’s syndrome. BMJ Case Rep. 2018;2018:bcr2017223132.
- Neumayr A, Ollague J, Bravo F, et al. Cross-reactivity pattern of Asian and American human gnathostomiasis in western blot assays using crude antigens prepared from Gnathostoma spinigerum and Gnathostoma binucleatum third-stage larvae. Am J Trop Med Hyg. 2016;95:413-416.
- Kraivichian K, Nuchprayoon S, Sitichalernchai P, et al. Treatment of cutaneous gnathostomiasis with ivermectin. Am J Trop Med Hyg. 2004;71:623-628.
- Pérez-Garza DM, Chavez-Alvarez S, Ocampo-Candiani J, et al. Erythema nodosum: a practical approach and diagnostic algorithm. Am J Clin Dermatol. 2021;22:367-378.
The Diagnosis: Gnathostomiasis
The biopsy demonstrated a dense, eosinophilic, granulomatous infiltrate surrounding sections of a parasite with skeletal muscle bundles and intestines containing a brush border and luminal debris (Figure), which was consistent with a diagnosis of gnathostomiasis. Upon further questioning, he revealed that while in Peru he frequently consumed ceviche, which is a dish typically made from fresh raw fish cured in lemon or lime juice. He subsequently was treated with oral ivermectin 0.2 mg/kg once daily for 2 days with no evidence of recurrence 12 months later.

Cutaneous gnathostomiasis is the most common manifestation of infection caused by the third-stage larvae of the genus Gnathostoma. The nematode is endemic to tropical and subtropical regions of Japan and Southeast Asia, particularly Thailand. The disease has been increasingly observed in Central and South America. Humans can become infected through ingestion of undercooked meats, particularly freshwater fish but also poultry, snakes, or frogs. Few cases have been reported in North America and Europe presumably due to more stringent regulations governing the sourcing and storage of fish for consumption.1-3 Restaurants in endemic regions also may use cheaper local freshwater or brackish fish compared to restaurants in the West, which use more expensive saltwater fish that do not harbor Gnathostoma species.1 There is a false belief among restauranteurs and consumers that the larvae can be reliably killed by marinating meat in citrus juice or with concurrent consumption of alcohol or hot spices.2 Adequately cooking or freezing meat to 20 °C for 3 to 5 days are the only effective ways to ensure that the larvae are killed.1-3
The parasite requires its natural definitive hosts—fish-eating mammals such as pigs, cats, and dogs—to complete its life cycle and reproduce. Humans are accidental hosts in whom the parasite fails to reach sexual maturity.1-3 Consequently, symptoms commonly are due to the migration of only 1 larva, but occasionally infection with 2 or more has been observed.1,4
Human infection initially may result in malaise, fever, anorexia, abdominal pain, nausea, vomiting, and diarrhea as the parasite migrates through the stomach, intestines, and liver. After 2 to 4 weeks, larvae may reach the skin where they most commonly create ill-defined, erythematous, indurated, round or oval plaques or nodules described as nodular migratory panniculitis. These lesions tend to develop on the trunk or arms and correspond to the location of the migrating worm.1,3,5 The larvae have been observed to migrate at 1 cm/h.6 Symptoms often wax and wane, with individual nodules lasting approximately 1 to 2 weeks. Uniquely, larval migration can result in a trail of subcutaneous hemorrhage that is considered pathognomonic and helps to differentiate gnathostomiasis from other forms of parasitosis such as strongyloidiasis and sparganosis.1,3 Larvae are highly motile and invasive, and they are capable of producing a wide range of symptoms affecting virtually any part of the body.1,2 Depending on the anatomic location of the migrating worm, infection also may result in neurologic, gastrointestinal, pulmonary, or ocular symptoms.1-3,7 Eosinophilia is common but can subside in the chronic stage, as seen in our patient.1
The classic triad of intermittent migratory nodules, eosinophilia, and a history of travel to Southeast Asia or another endemic region should raise suspicion for gnathostomiasis.1-3,5,7 Unfortunately, confirmatory testing such as Gnathostoma serology is not readily available in the United States, and available serologic tests demonstrate frequent false positives and incomplete crossreactivity.1,2,8 Accordingly, the diagnosis most commonly is solidified by combining cardinal clinical features with histologic findings of a dense eosinophilic inflammatory infiltrate involving the dermis and hypodermis.2,5 In one study, the larva itself was only found in 12 of 66 (18%) skin biopsy specimens from patients with gnathostomiasis.5 If the larva is detected within the sections, it ranges from 2.5 to 12.5 mm in length and 0.4 to 1.2 mm in width and can exhibit cuticular spines, intestinal cells, and characteristic large lateral chords.1,5
The treatment of choice is surgical removal of the worm. Oral albendazole (400–800 mg/d for 21 days) also is considered a first-line treatment and results in clinical cure in approximately 90% of cases. Two doses of oral ivermectin (0.2 mg/kg) spaced 24 to 48 hours apart is an acceptable alternative with comparable efficacy.1-3 Care should be taken if involvement of the central nervous system is suspected, as antihelminthic treatment theoretically could be deleterious due to an inflammatory response to the dying larvae.1,2,9
In the differential diagnosis, loiasis can resemble gnathostomiasis, but the former is endemic to Africa.3 Cutaneous larva migrans most frequently is caused by hookworms from the genus Ancylostoma, which classically leads to superficial serpiginous linear plaques that migrate at a rate of several millimeters per day. However, the larvae are believed to lack the collagenase enzyme required to penetrate the epidermal basement membrane and thus are not capable of producing deep-seated nodules or visceral symptoms.3 Strongyloidiasis (larva currens) generally exhibits a more linear morphology, and infection would result in positive Strongyloides serology.7 Erythema nodosum is a septal panniculitis that can be triggered by infection, pregnancy, medications, connective tissue diseases, inflammatory conditions, and underlying malignancy.10
The Diagnosis: Gnathostomiasis
The biopsy demonstrated a dense, eosinophilic, granulomatous infiltrate surrounding sections of a parasite with skeletal muscle bundles and intestines containing a brush border and luminal debris (Figure), which was consistent with a diagnosis of gnathostomiasis. Upon further questioning, he revealed that while in Peru he frequently consumed ceviche, which is a dish typically made from fresh raw fish cured in lemon or lime juice. He subsequently was treated with oral ivermectin 0.2 mg/kg once daily for 2 days with no evidence of recurrence 12 months later.

Cutaneous gnathostomiasis is the most common manifestation of infection caused by the third-stage larvae of the genus Gnathostoma. The nematode is endemic to tropical and subtropical regions of Japan and Southeast Asia, particularly Thailand. The disease has been increasingly observed in Central and South America. Humans can become infected through ingestion of undercooked meats, particularly freshwater fish but also poultry, snakes, or frogs. Few cases have been reported in North America and Europe presumably due to more stringent regulations governing the sourcing and storage of fish for consumption.1-3 Restaurants in endemic regions also may use cheaper local freshwater or brackish fish compared to restaurants in the West, which use more expensive saltwater fish that do not harbor Gnathostoma species.1 There is a false belief among restauranteurs and consumers that the larvae can be reliably killed by marinating meat in citrus juice or with concurrent consumption of alcohol or hot spices.2 Adequately cooking or freezing meat to 20 °C for 3 to 5 days are the only effective ways to ensure that the larvae are killed.1-3
The parasite requires its natural definitive hosts—fish-eating mammals such as pigs, cats, and dogs—to complete its life cycle and reproduce. Humans are accidental hosts in whom the parasite fails to reach sexual maturity.1-3 Consequently, symptoms commonly are due to the migration of only 1 larva, but occasionally infection with 2 or more has been observed.1,4
Human infection initially may result in malaise, fever, anorexia, abdominal pain, nausea, vomiting, and diarrhea as the parasite migrates through the stomach, intestines, and liver. After 2 to 4 weeks, larvae may reach the skin where they most commonly create ill-defined, erythematous, indurated, round or oval plaques or nodules described as nodular migratory panniculitis. These lesions tend to develop on the trunk or arms and correspond to the location of the migrating worm.1,3,5 The larvae have been observed to migrate at 1 cm/h.6 Symptoms often wax and wane, with individual nodules lasting approximately 1 to 2 weeks. Uniquely, larval migration can result in a trail of subcutaneous hemorrhage that is considered pathognomonic and helps to differentiate gnathostomiasis from other forms of parasitosis such as strongyloidiasis and sparganosis.1,3 Larvae are highly motile and invasive, and they are capable of producing a wide range of symptoms affecting virtually any part of the body.1,2 Depending on the anatomic location of the migrating worm, infection also may result in neurologic, gastrointestinal, pulmonary, or ocular symptoms.1-3,7 Eosinophilia is common but can subside in the chronic stage, as seen in our patient.1
The classic triad of intermittent migratory nodules, eosinophilia, and a history of travel to Southeast Asia or another endemic region should raise suspicion for gnathostomiasis.1-3,5,7 Unfortunately, confirmatory testing such as Gnathostoma serology is not readily available in the United States, and available serologic tests demonstrate frequent false positives and incomplete crossreactivity.1,2,8 Accordingly, the diagnosis most commonly is solidified by combining cardinal clinical features with histologic findings of a dense eosinophilic inflammatory infiltrate involving the dermis and hypodermis.2,5 In one study, the larva itself was only found in 12 of 66 (18%) skin biopsy specimens from patients with gnathostomiasis.5 If the larva is detected within the sections, it ranges from 2.5 to 12.5 mm in length and 0.4 to 1.2 mm in width and can exhibit cuticular spines, intestinal cells, and characteristic large lateral chords.1,5
The treatment of choice is surgical removal of the worm. Oral albendazole (400–800 mg/d for 21 days) also is considered a first-line treatment and results in clinical cure in approximately 90% of cases. Two doses of oral ivermectin (0.2 mg/kg) spaced 24 to 48 hours apart is an acceptable alternative with comparable efficacy.1-3 Care should be taken if involvement of the central nervous system is suspected, as antihelminthic treatment theoretically could be deleterious due to an inflammatory response to the dying larvae.1,2,9
In the differential diagnosis, loiasis can resemble gnathostomiasis, but the former is endemic to Africa.3 Cutaneous larva migrans most frequently is caused by hookworms from the genus Ancylostoma, which classically leads to superficial serpiginous linear plaques that migrate at a rate of several millimeters per day. However, the larvae are believed to lack the collagenase enzyme required to penetrate the epidermal basement membrane and thus are not capable of producing deep-seated nodules or visceral symptoms.3 Strongyloidiasis (larva currens) generally exhibits a more linear morphology, and infection would result in positive Strongyloides serology.7 Erythema nodosum is a septal panniculitis that can be triggered by infection, pregnancy, medications, connective tissue diseases, inflammatory conditions, and underlying malignancy.10
- Herman JS, Chiodini PL. Gnathostomiasis, another emerging imported disease. Clin Microbiol Rev. 2009;22:484-492.
- Liu GH, Sun MM, Elsheikha HM, et al. Human gnathostomiasis: a neglected food-borne zoonosis. Parasit Vectors. 2020;13:616.
- Tyring SK. Gnathostomiasis. In: Tyring SK, Lupi O, Hengge UR, eds. Tropical Dermatology. 2nd ed. Elsevier; 2017:77-78.
- Rusnak JM, Lucey DR. Clinical gnathostomiasis: case report and review of the English-language literature. Clin Infect Dis. 1993;16:33-50.
- Magaña M, Messina M, Bustamante F, et al. Gnathostomiasis: clinicopathologic study. Am J Dermatopathol. 2004;26:91-95.
- Chandenier J, Husson J, Canaple S, et al. Medullary gnathostomiasis in a white patient: use of immunodiagnosis and magnetic resonance imaging. Clin Infect Dis. 2001;32:E154-E157.
- Hamilton WL, Agranoff D. Imported gnathostomiasis manifesting as cutaneous larva migrans and Löffler’s syndrome. BMJ Case Rep. 2018;2018:bcr2017223132.
- Neumayr A, Ollague J, Bravo F, et al. Cross-reactivity pattern of Asian and American human gnathostomiasis in western blot assays using crude antigens prepared from Gnathostoma spinigerum and Gnathostoma binucleatum third-stage larvae. Am J Trop Med Hyg. 2016;95:413-416.
- Kraivichian K, Nuchprayoon S, Sitichalernchai P, et al. Treatment of cutaneous gnathostomiasis with ivermectin. Am J Trop Med Hyg. 2004;71:623-628.
- Pérez-Garza DM, Chavez-Alvarez S, Ocampo-Candiani J, et al. Erythema nodosum: a practical approach and diagnostic algorithm. Am J Clin Dermatol. 2021;22:367-378.
- Herman JS, Chiodini PL. Gnathostomiasis, another emerging imported disease. Clin Microbiol Rev. 2009;22:484-492.
- Liu GH, Sun MM, Elsheikha HM, et al. Human gnathostomiasis: a neglected food-borne zoonosis. Parasit Vectors. 2020;13:616.
- Tyring SK. Gnathostomiasis. In: Tyring SK, Lupi O, Hengge UR, eds. Tropical Dermatology. 2nd ed. Elsevier; 2017:77-78.
- Rusnak JM, Lucey DR. Clinical gnathostomiasis: case report and review of the English-language literature. Clin Infect Dis. 1993;16:33-50.
- Magaña M, Messina M, Bustamante F, et al. Gnathostomiasis: clinicopathologic study. Am J Dermatopathol. 2004;26:91-95.
- Chandenier J, Husson J, Canaple S, et al. Medullary gnathostomiasis in a white patient: use of immunodiagnosis and magnetic resonance imaging. Clin Infect Dis. 2001;32:E154-E157.
- Hamilton WL, Agranoff D. Imported gnathostomiasis manifesting as cutaneous larva migrans and Löffler’s syndrome. BMJ Case Rep. 2018;2018:bcr2017223132.
- Neumayr A, Ollague J, Bravo F, et al. Cross-reactivity pattern of Asian and American human gnathostomiasis in western blot assays using crude antigens prepared from Gnathostoma spinigerum and Gnathostoma binucleatum third-stage larvae. Am J Trop Med Hyg. 2016;95:413-416.
- Kraivichian K, Nuchprayoon S, Sitichalernchai P, et al. Treatment of cutaneous gnathostomiasis with ivermectin. Am J Trop Med Hyg. 2004;71:623-628.
- Pérez-Garza DM, Chavez-Alvarez S, Ocampo-Candiani J, et al. Erythema nodosum: a practical approach and diagnostic algorithm. Am J Clin Dermatol. 2021;22:367-378.
A 41-year-old man presented to a dermatology clinic in the United States with a migratory subcutaneous nodule overlying the left upper chest that initially developed 12 months prior and continued to migrate along the trunk and proximal aspect of the arms. The patient had spent the last 3 years residing in Peru. He never observed more than 1 nodule at a time and denied associated fever, headache, visual changes, chest pain, cough, abdominal pain, and diarrhea. Laboratory studies including a blood eosinophil count and serum Strongyloides immunoglobulins were within reference range. An excisional biopsy was performed.
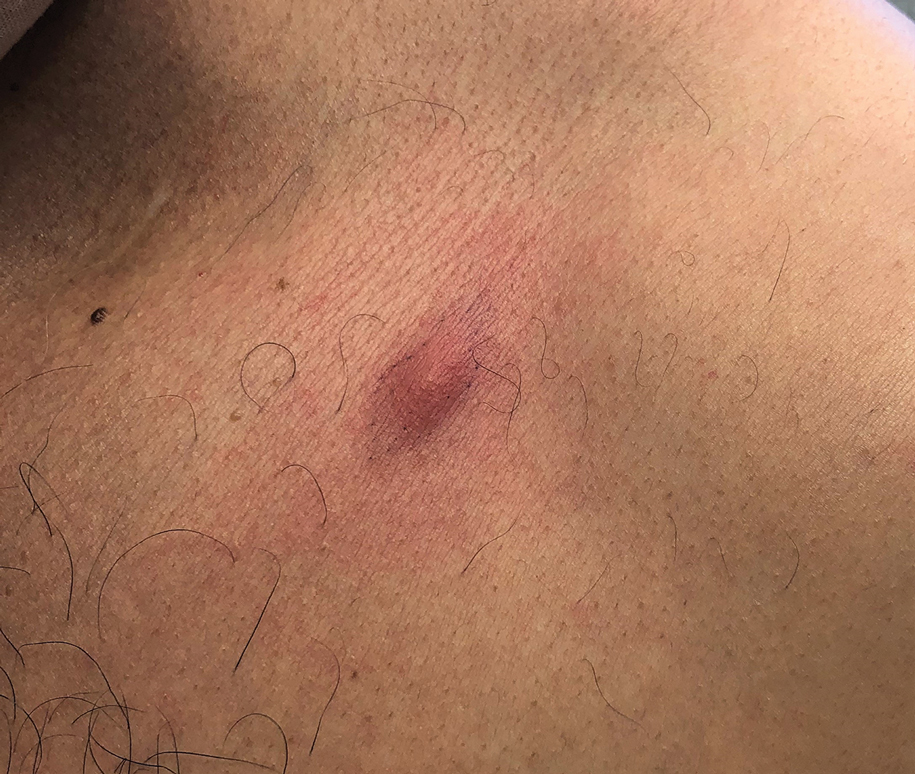
New Insights, New Standards: How 2023 Changed Care for Internists
The past year brought major changes in preventive standards for anxiety, HIV, and RSV along with new guidelines for the treatment of atrial fibrillation. For insight into the effect on internal medicine, we turned to Sarah Candler, MD, MPH, a Houston internist who specializes in the care of high-risk older adults.
Q: Which new prevention guidelines had the most impact on you over the past year?
A: I’m a primary care doctor, and most of the internal medicine updates that are interesting to me focus on how we can keep people from getting sick in the first place. That’s especially important in light of the fact that we had a decrease in life expectancy of 2 years [it finally rose slightly in 2022] and widening of the gender gap in life expectancy for men and women.
I’m excited to see new recommendations from the U.S. Preventive Services Task Force, including a new one about using PREP [pre-exposure prophylaxis] to preventively treat anyone who’s at risk for getting HIV. That’s a big one because it’s one of the first times that we’ve identified at-risk groups for screening based on social risk factors, not gender, age, or genetics.
The new recommendation is PREP for anyone who’s at risk for getting HIV because they have a partner with HIV, had an sexually transmitted infection in the last 6 months, or a history of inconsistent or no condom use with partners with unknown HIV status.
PREP therapy is something that most primary care physicians can either do or learn how to do pretty easily. But the treatment does require maintenance and monitoring.
Q: How firm is this recommendation?
A: The task force gives different grades for their recommendations based on how strong the evidence is. For the guidelines about PREP, they give a grade of A. That means this is top of the class: You should definitely do this.
Q: What are the best strategies to ask patients personal questions about their sex lives in order to evaluate their risk?
A: A lot of internal medicine physicians are getting pretty good at this. We see it as part of our job just the same way as we asked things like, “How often are you walking?” and “Have you been feeling down?”
There’s no one right way to have a conversation like that. But it’s key to say, as I do to my patients, that “I’m not here to judge anything. I am truly here to gather information and make recommendations to you as a partner in your care.”
Q: What other guidelines made an impact in 2023?
A: The U.S. Preventive Services Task Force made a recommendation to screen adults aged 18-64 for anxiety, and this guidance got a B grade. [The task force said there’s not enough evidence to support routine anxiety screening in adults 65 and older.]
The new recommendations is a sign that we’re doing a better job at making treatment of those diseases more acceptable. This is also another example of the medical community recognizing that internal medicine physicians are pretty good at identifying and treating mental health.
Q: How do you figure out whether to treat depression/anxiety yourself or refer patients to specialists?
A: As a primary care physician, I feel comfortable diagnosing and managing some mental health disease in my own practice. There are FDA-approved medications for both anxiety and depression that are easily managed by a primary care physician.
And there’s something to the therapeutic relationship, to naming and identifying these conditions with your patients. Some patients feel a bit of relief just knowing that they have a diagnosis.
Q: What should internists know about the new CDC guidelines that promote discussing RSV vaccines with patients who are over 60?
A: The vaccines are recommended for folks who have underlying conditions like lung disease or heart disease. Those are the ones who end up getting really, really sick. There are two adult vaccines that are available, and there’s not a preference for one over the other.
The vaccines are both protein-based, like the old-school versions of vaccines, not the mRNA vaccines that we’ve all been hearing more about through COVID. Anybody who’s reluctant to take an mRNA vaccine can rest assured that the RSV is not protein-based. And they are single-dose vaccines, which is helpful.
Q: What else should internists know about that was new in 2023?
A: I’m super excited about how cardiologists are thinking about atrial fibrillation. In 2023, the American College of Cardiology and the American Heart Association came up with a giant overhaul of how they look at atrial fibrillation. They classify it in stages and allows us to think about stopping it before it starts.
They’re talking about something they’re calling preclinical or subclinical atrial fibrillation, which you may detect on wearables like somebody’s watch or another tool used to monitor heart rate or exercise. It might be the first harbinger that there’s something wrong with the heart rate, and they may not even have symptoms of it. [A 2023 study in The New England Journal of Medicine linked the anticoagulant apixaban, or Eliquis, to a 37% lower risk of stroke and systemic embolism rates in older patients with subclinical atrial fibrillation but an 80% higher risk of major bleeding vs. aspirin therapy.]
And they’re now recommending early rhythm control.
Q: What does early rhythm control mean for patients and physicians?
A: For the longest time, we have thought about atrial fibrillation treatment in terms of rate control and not worrying too much about the rhythm. But now we recognize that it’s actually really important that we get the rhythm under control because physical changes to the heart can lead to permanent damage.
So now they’re recommending catheter ablation as first-line therapy in some patients as a class 1 recommendation because heart function is already decreased. Improving the ability of the heart to beat with a regular rhythm can lead to improvement of function. This was unheard of even 5 years ago.
Q: Should internists be more willing to refer patients with atrial fibrillation to cardiologists?
A: Yes, I think so. One of the biggest changes for me is that I am going to refer new diagnoses of atrial fibrillation to a cardiologist. And I’m going to ask patients if they have wearable devices because sometimes those things might tell me about something like subclinical atrial fibrillation.
Q: There’s also detailed data about atrial fibrillation risk factors, which include older age, smoking, sedentary lifestyle, alcohol use, diabetes, height, obesity, diabetes, and others. Is this information useful?
A: It’s a really great tool to have in the arsenal because it helps me have shared decision-making conversations with my patients in a way that’s much more convincing. A patient might say, “Why do you care if I drink so much? My liver levels are fine.” And I can say, “It’s going to be a risk factor for having problems with your heart.”
For better or worse, people really take the heart very seriously, I am an internal medicine physician, so I love all the organs equally. But man, people get pretty scared when you tell them something can affect their heart. So when I talk to patients about their risk factors, it’s going to really be helpful that I can remind them of the impact that some of these lifestyle behaviors can have on their heart health.
Dr. Candler has no disclosures.
The past year brought major changes in preventive standards for anxiety, HIV, and RSV along with new guidelines for the treatment of atrial fibrillation. For insight into the effect on internal medicine, we turned to Sarah Candler, MD, MPH, a Houston internist who specializes in the care of high-risk older adults.
Q: Which new prevention guidelines had the most impact on you over the past year?
A: I’m a primary care doctor, and most of the internal medicine updates that are interesting to me focus on how we can keep people from getting sick in the first place. That’s especially important in light of the fact that we had a decrease in life expectancy of 2 years [it finally rose slightly in 2022] and widening of the gender gap in life expectancy for men and women.
I’m excited to see new recommendations from the U.S. Preventive Services Task Force, including a new one about using PREP [pre-exposure prophylaxis] to preventively treat anyone who’s at risk for getting HIV. That’s a big one because it’s one of the first times that we’ve identified at-risk groups for screening based on social risk factors, not gender, age, or genetics.
The new recommendation is PREP for anyone who’s at risk for getting HIV because they have a partner with HIV, had an sexually transmitted infection in the last 6 months, or a history of inconsistent or no condom use with partners with unknown HIV status.
PREP therapy is something that most primary care physicians can either do or learn how to do pretty easily. But the treatment does require maintenance and monitoring.
Q: How firm is this recommendation?
A: The task force gives different grades for their recommendations based on how strong the evidence is. For the guidelines about PREP, they give a grade of A. That means this is top of the class: You should definitely do this.
Q: What are the best strategies to ask patients personal questions about their sex lives in order to evaluate their risk?
A: A lot of internal medicine physicians are getting pretty good at this. We see it as part of our job just the same way as we asked things like, “How often are you walking?” and “Have you been feeling down?”
There’s no one right way to have a conversation like that. But it’s key to say, as I do to my patients, that “I’m not here to judge anything. I am truly here to gather information and make recommendations to you as a partner in your care.”
Q: What other guidelines made an impact in 2023?
A: The U.S. Preventive Services Task Force made a recommendation to screen adults aged 18-64 for anxiety, and this guidance got a B grade. [The task force said there’s not enough evidence to support routine anxiety screening in adults 65 and older.]
The new recommendations is a sign that we’re doing a better job at making treatment of those diseases more acceptable. This is also another example of the medical community recognizing that internal medicine physicians are pretty good at identifying and treating mental health.
Q: How do you figure out whether to treat depression/anxiety yourself or refer patients to specialists?
A: As a primary care physician, I feel comfortable diagnosing and managing some mental health disease in my own practice. There are FDA-approved medications for both anxiety and depression that are easily managed by a primary care physician.
And there’s something to the therapeutic relationship, to naming and identifying these conditions with your patients. Some patients feel a bit of relief just knowing that they have a diagnosis.
Q: What should internists know about the new CDC guidelines that promote discussing RSV vaccines with patients who are over 60?
A: The vaccines are recommended for folks who have underlying conditions like lung disease or heart disease. Those are the ones who end up getting really, really sick. There are two adult vaccines that are available, and there’s not a preference for one over the other.
The vaccines are both protein-based, like the old-school versions of vaccines, not the mRNA vaccines that we’ve all been hearing more about through COVID. Anybody who’s reluctant to take an mRNA vaccine can rest assured that the RSV is not protein-based. And they are single-dose vaccines, which is helpful.
Q: What else should internists know about that was new in 2023?
A: I’m super excited about how cardiologists are thinking about atrial fibrillation. In 2023, the American College of Cardiology and the American Heart Association came up with a giant overhaul of how they look at atrial fibrillation. They classify it in stages and allows us to think about stopping it before it starts.
They’re talking about something they’re calling preclinical or subclinical atrial fibrillation, which you may detect on wearables like somebody’s watch or another tool used to monitor heart rate or exercise. It might be the first harbinger that there’s something wrong with the heart rate, and they may not even have symptoms of it. [A 2023 study in The New England Journal of Medicine linked the anticoagulant apixaban, or Eliquis, to a 37% lower risk of stroke and systemic embolism rates in older patients with subclinical atrial fibrillation but an 80% higher risk of major bleeding vs. aspirin therapy.]
And they’re now recommending early rhythm control.
Q: What does early rhythm control mean for patients and physicians?
A: For the longest time, we have thought about atrial fibrillation treatment in terms of rate control and not worrying too much about the rhythm. But now we recognize that it’s actually really important that we get the rhythm under control because physical changes to the heart can lead to permanent damage.
So now they’re recommending catheter ablation as first-line therapy in some patients as a class 1 recommendation because heart function is already decreased. Improving the ability of the heart to beat with a regular rhythm can lead to improvement of function. This was unheard of even 5 years ago.
Q: Should internists be more willing to refer patients with atrial fibrillation to cardiologists?
A: Yes, I think so. One of the biggest changes for me is that I am going to refer new diagnoses of atrial fibrillation to a cardiologist. And I’m going to ask patients if they have wearable devices because sometimes those things might tell me about something like subclinical atrial fibrillation.
Q: There’s also detailed data about atrial fibrillation risk factors, which include older age, smoking, sedentary lifestyle, alcohol use, diabetes, height, obesity, diabetes, and others. Is this information useful?
A: It’s a really great tool to have in the arsenal because it helps me have shared decision-making conversations with my patients in a way that’s much more convincing. A patient might say, “Why do you care if I drink so much? My liver levels are fine.” And I can say, “It’s going to be a risk factor for having problems with your heart.”
For better or worse, people really take the heart very seriously, I am an internal medicine physician, so I love all the organs equally. But man, people get pretty scared when you tell them something can affect their heart. So when I talk to patients about their risk factors, it’s going to really be helpful that I can remind them of the impact that some of these lifestyle behaviors can have on their heart health.
Dr. Candler has no disclosures.
The past year brought major changes in preventive standards for anxiety, HIV, and RSV along with new guidelines for the treatment of atrial fibrillation. For insight into the effect on internal medicine, we turned to Sarah Candler, MD, MPH, a Houston internist who specializes in the care of high-risk older adults.
Q: Which new prevention guidelines had the most impact on you over the past year?
A: I’m a primary care doctor, and most of the internal medicine updates that are interesting to me focus on how we can keep people from getting sick in the first place. That’s especially important in light of the fact that we had a decrease in life expectancy of 2 years [it finally rose slightly in 2022] and widening of the gender gap in life expectancy for men and women.
I’m excited to see new recommendations from the U.S. Preventive Services Task Force, including a new one about using PREP [pre-exposure prophylaxis] to preventively treat anyone who’s at risk for getting HIV. That’s a big one because it’s one of the first times that we’ve identified at-risk groups for screening based on social risk factors, not gender, age, or genetics.
The new recommendation is PREP for anyone who’s at risk for getting HIV because they have a partner with HIV, had an sexually transmitted infection in the last 6 months, or a history of inconsistent or no condom use with partners with unknown HIV status.
PREP therapy is something that most primary care physicians can either do or learn how to do pretty easily. But the treatment does require maintenance and monitoring.
Q: How firm is this recommendation?
A: The task force gives different grades for their recommendations based on how strong the evidence is. For the guidelines about PREP, they give a grade of A. That means this is top of the class: You should definitely do this.
Q: What are the best strategies to ask patients personal questions about their sex lives in order to evaluate their risk?
A: A lot of internal medicine physicians are getting pretty good at this. We see it as part of our job just the same way as we asked things like, “How often are you walking?” and “Have you been feeling down?”
There’s no one right way to have a conversation like that. But it’s key to say, as I do to my patients, that “I’m not here to judge anything. I am truly here to gather information and make recommendations to you as a partner in your care.”
Q: What other guidelines made an impact in 2023?
A: The U.S. Preventive Services Task Force made a recommendation to screen adults aged 18-64 for anxiety, and this guidance got a B grade. [The task force said there’s not enough evidence to support routine anxiety screening in adults 65 and older.]
The new recommendations is a sign that we’re doing a better job at making treatment of those diseases more acceptable. This is also another example of the medical community recognizing that internal medicine physicians are pretty good at identifying and treating mental health.
Q: How do you figure out whether to treat depression/anxiety yourself or refer patients to specialists?
A: As a primary care physician, I feel comfortable diagnosing and managing some mental health disease in my own practice. There are FDA-approved medications for both anxiety and depression that are easily managed by a primary care physician.
And there’s something to the therapeutic relationship, to naming and identifying these conditions with your patients. Some patients feel a bit of relief just knowing that they have a diagnosis.
Q: What should internists know about the new CDC guidelines that promote discussing RSV vaccines with patients who are over 60?
A: The vaccines are recommended for folks who have underlying conditions like lung disease or heart disease. Those are the ones who end up getting really, really sick. There are two adult vaccines that are available, and there’s not a preference for one over the other.
The vaccines are both protein-based, like the old-school versions of vaccines, not the mRNA vaccines that we’ve all been hearing more about through COVID. Anybody who’s reluctant to take an mRNA vaccine can rest assured that the RSV is not protein-based. And they are single-dose vaccines, which is helpful.
Q: What else should internists know about that was new in 2023?
A: I’m super excited about how cardiologists are thinking about atrial fibrillation. In 2023, the American College of Cardiology and the American Heart Association came up with a giant overhaul of how they look at atrial fibrillation. They classify it in stages and allows us to think about stopping it before it starts.
They’re talking about something they’re calling preclinical or subclinical atrial fibrillation, which you may detect on wearables like somebody’s watch or another tool used to monitor heart rate or exercise. It might be the first harbinger that there’s something wrong with the heart rate, and they may not even have symptoms of it. [A 2023 study in The New England Journal of Medicine linked the anticoagulant apixaban, or Eliquis, to a 37% lower risk of stroke and systemic embolism rates in older patients with subclinical atrial fibrillation but an 80% higher risk of major bleeding vs. aspirin therapy.]
And they’re now recommending early rhythm control.
Q: What does early rhythm control mean for patients and physicians?
A: For the longest time, we have thought about atrial fibrillation treatment in terms of rate control and not worrying too much about the rhythm. But now we recognize that it’s actually really important that we get the rhythm under control because physical changes to the heart can lead to permanent damage.
So now they’re recommending catheter ablation as first-line therapy in some patients as a class 1 recommendation because heart function is already decreased. Improving the ability of the heart to beat with a regular rhythm can lead to improvement of function. This was unheard of even 5 years ago.
Q: Should internists be more willing to refer patients with atrial fibrillation to cardiologists?
A: Yes, I think so. One of the biggest changes for me is that I am going to refer new diagnoses of atrial fibrillation to a cardiologist. And I’m going to ask patients if they have wearable devices because sometimes those things might tell me about something like subclinical atrial fibrillation.
Q: There’s also detailed data about atrial fibrillation risk factors, which include older age, smoking, sedentary lifestyle, alcohol use, diabetes, height, obesity, diabetes, and others. Is this information useful?
A: It’s a really great tool to have in the arsenal because it helps me have shared decision-making conversations with my patients in a way that’s much more convincing. A patient might say, “Why do you care if I drink so much? My liver levels are fine.” And I can say, “It’s going to be a risk factor for having problems with your heart.”
For better or worse, people really take the heart very seriously, I am an internal medicine physician, so I love all the organs equally. But man, people get pretty scared when you tell them something can affect their heart. So when I talk to patients about their risk factors, it’s going to really be helpful that I can remind them of the impact that some of these lifestyle behaviors can have on their heart health.
Dr. Candler has no disclosures.
ME/CFS and Long COVID: Research Aims to Identify Treatable, Druggable Pathways
BETHESDA, MD — New research into the mechanisms underlying myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) and long COVID is aimed at identifying potential approaches to treatment of the two overlapping illnesses.
According to a new data brief from the National Center for Health Statistics, in 2021-2022, 1.3% of US adults had ME/CFS, a complex, multisystem illness characterized by activity-limiting fatigue, worsening of symptoms after exertion, unrefreshing sleep, and other symptoms.
A 2-day conference, Advancing ME/CFS Research: Identifying Targets for Potential Intervention and Learning from Long COVID, was held in December 12-13 on the main campus of the US National Institutes of Health (NIH) and was livestreamed. The last such meeting, also featuring results from NIH-funded research, was held in April 2019.
“Things have changed since 2019 ... The idea of this meeting is to try and identify pathways that will be treatable and druggable and really make an impact for patients based on the things that we’ve learned over the last number of years and including, fortunately or unfortunately, the huge number of people who are suffering from long COVID, where the symptoms overlap so much with those who have been suffering for a long time with ME/CFS,” said Conference Chair Joe Breen, PhD, of the National Institute of Allergy and Infectious Diseases.
As in 2019, the meeting was preceded by a day of research presentations from young investigators, some of whom also presented their findings at the main meeting. New this year were four “lived experience” speakers who described their physical, emotional, and financial struggles with ME/CFS or long COVID. Two of them presented virtually because they were too ill to travel.
Social worker and patient advocate Terri Wilder of Minneapolis, Minnesota, reported some feedback she received on social media after she asked people with ME/CFS about their priorities for the research and clinical communities.
Among the top responses were the need to recognize and study the phenomenon of “post-exertional malaise” and to stop recommending exercise for people with these illnesses, to accelerate research to find effective treatments, and to put an end to stigma around the condition. “People don’t believe us when we tell them we’re sick, people make fun of us, misperceptions persist,” Wilder said.
One person commented, “[Clinicians] shouldn’t be afraid to try off-label meds with us if needed. There may be some secondary effects, but they are better options than us taking our own lives because we can’t stand the suffering.”
Research areas covered at the conference included immunology, virology, metabolism, gene regulation, and neurology of both ME/CFS and long COVID, as well as the latest findings regarding the overlap between the two conditions.
Oxidative Stress in Both ME/CFS and Long COVID: A Role for Metformin?
Mark M. Davis, PhD, professor and director of the Institute for Immunity, Transplantation, and Infection at Stanford University, Palo Alto, California, summarized published data suggesting that oxidative stress is a shared characteristic of both ME/CFS and long COVID. Most cellular reactive oxygen species (ROS) are produced in the cell’s mitochondria, and T-cell activation is ROS-dependent.
Women in particular with ME/CFS show high ROS levels with consistent T-cell hyperproliferation, “which can be suppressed with specific drugs such as metformin. This raises the prospect of optimizing drug treatment and drug discovery with a simple in vitro assay of the effects on a patient’s lymphocytes,” Dr. Davis said. He also cited a study suggesting that metformin may help prevent long COVID.
Asked to comment on that, longtime ME/CFS researcher Anthony L. Komaroff, MD, of Harvard University, Cambridge, Massachusetts, cautioned that although metformin is used safely by millions of people with type 2 diabetes worldwide, it’s possible that some people with ME/CFS may be more likely to experience its known adverse effects such as lactic acidosis.
To repurpose metformin or any other already-marketed drugs for ME/CFS and/or long COVID, Dr. Komaroff said, “We should entertain treatment trials.” However, as he and many others lamented at the conference, funding for off-patent drugs often isn’t forthcoming.
Addressing the Microbiome, Innate Immunity
W. Ian Lipkin, MD, of Columbia University, New York, NY, was one of two speakers who discussed the role of disruptions in the microbiome and innate immunity in ME/CFS. He presented data suggesting that “dysregulation of the gut microbiome in ME/CFS may interfere with butyrate production, resulting in inflammation and porosity to bacteria and bacterial products that trigger innate immunity.”
Dr. Lipkin highlighted a “really intriguing” paper in which exogenous administration of interleukin 37 (IL-37), a naturally occurring inhibitor of inflammation, reversed the decrease in exercise performance observed during inflammation-induced fatigue and increased exercise performance, both in mice.
“Although we do not fully understand the pathophysiology of ME/CFS, it is not premature to consider randomized clinical trials of pro- and pre-biotics that address dysbiosis as well as drugs that modify innate immune responses such as poly (I:C) and IL-37,” Dr. Lipkin said.
Alleviating Endoplasmic Reticulum (ER) Stress: A Strategy to Increase Energy?
Paul M. Hwang, MD, PhD, from the Cardiovascular Branch of the National Heart, Lung, and Blood Institute, Bethesda, Maryland, described work that he and his colleagues recently published around a case of a 38-year-old woman with Li-Fraumeni syndrome, a genetic early-onset cancer, who also had extensive fatigue, exercise intolerance, and post-exertional malaise that began after she contracted mononucleosis as a teenager.
Testing revealed that her cells had increased expression of Wiskott-Aldrich Syndrome Protein Family Member 3 (WASF3), a “top candidate” gene found to be associated with ME/CFS in a bioinformatics study published more than a decade ago. Moreover, immunoblotting of deidentified skeletal muscle biopsy samples obtained from patients with postinfectious ME/CFS also revealed significantly increased WASF3 levels.
Hwang and colleagues showed in mice that ER stress–induced WASF3 protein localizes to mitochondria and disrupts respiratory supercomplex assembly, leading to decreased oxygen consumption and exercise endurance.
However, use of the investigational protein phosphatase 1 inhibitor salubrinal in the female patient’s cells inhibited the ER stress, which in turn decreased WASF3 expression and improved mitochondrial supercomplex formation and respiration, “suggesting a treatment strategy in ME/CFS,” Dr. Hwang said.
Neurovascular Dysregulation During Exercise: A Role for Pyridostigmine?
David M. Systrom, MD, a pulmonary and critical care medicine specialist at the Brigham and Women’s Hospital, Boston, Massachusetts, gave an update of his work investigating neurovascular dysregulation during exercise in both ME/CFS and long COVID using invasive cardiopulmonary testing.
In a 2021 publication, Dr. Systrom and his colleagues identified the mechanism of “preload failure,” or lower filling pressures of blood in the heart chambers because of insufficient vein constriction and reduced return of blood to the right side of the heart in people with ME/CFS, compared with healthy controls.
Subsequently, in a randomized trial of 45 patients with ME/CFS, Systrom and his colleagues published in November 2022, use of the cholinesterase inhibitor pyridostigmine, currently approved for treating myasthenia gravis and related conditions, improved peak Vo2 by increasing cardiac output and filling pressures.
Now, Dr. Systrom’s team is conducting a randomized trial comparing 60 mg pyridostigmine with or without low-dose naltrexone (LDN) vs placebo in 160 patients with ME/CFS for 3 months. Metabolomic, transcriptomic, proteomic, and other assessments will be conducted on urine and blood samples. Participants will also wear devices that measure steps, sleep, heart rate, and other metrics.
Komaroff cautioned that pyridostigmine, too, has potential adverse effects. “I’m not sure pyridostigmine is ready for prime time ... It’s a drug developed for a very different purpose ... Now will it hold up in a larger trial, and will there be any side effects that turn up in larger studies? It’s not unreasonable to study.”
Brain Inflammation: Measuring and Treating It
Hannah F. Bues, clinical research coordinator at Massachusetts General Hospital, Boston, presented data now in preprint (ie, not yet peer-reviewed) in which researchers used [11C]PBR28 PET neuroimaging, a marker of neuroinflammation, to compare 12 individuals with long COVID vs 43 healthy controls. They found significantly increased neuroinflammation in several different brain regions in the long COVID group compared with controls.
Samples of peripheral blood plasma also showed significant correlations between neuroinflammation and circulating analytes related to vascular dysfunction. This work is ongoing in both long COVID and pre-COVID ME/CFS populations, Bues said.
Jarred Younger, PhD, of the Neuroinflammation, Pain, and Fatigue Laboratory at the University of Alabama, Birmingham, also gave an update of his ongoing work demonstrating significant brain inflammation seen in neuroimaging of people with ME/CFS compared with healthy controls.
Dr. Younger has been investigating the use of LDN for pain in fibromyalgia. Anecdotally, there have been reports of fatigue reduction with LDN in ME/CFS.
Dr. Younger conducted a post hoc analysis of his previous trial of LDN for 12 weeks in 30 patients with fibromyalgia. Of those, 16 met older CFS criteria. There was a significant reduction in their fatigue severity, with P <.0001 from baseline and P < .009 compared with placebo. The P values were high because the data included daily symptom reports. The average fatigue reduction was 25%.
“It wasn’t a study designed for ME/CFS, but I think it’s compelling evidence and enough with the other types of data we have to say we need to do a proper clinical trial of low-dose naltrexone in ME/CFS now,” Dr. Younger said.
‘We Need to Do Something’ About the Underfunding
Another NIH-funded ME/CFS researcher, Maureen Hanson, PhD, of Cornell University, Ithaca, NY, noted that the NIH currently funds ME/CFS research at about $13 million compared with $1.15 billion for the Researching COVID to Enhance Recovery Initiative granted to NIH by Congress for “post-acute sequelae of SARS-CoV-2 (PASC)” in 2021 “because of the urgency of studying this. Most of us here are well aware of the underfunding of ME/CFS relative to the burden of illness,” she said.
Current 2024 funding for AIDS research is $3294 million. “There are 1.2 million individuals living with HIV in the United States, and there are over 3 million who are barely living with ME/CFS in the United States. We need to do something about this ... It’s certainly possible that future funding for PASC is now going to disappear,” Dr. Hanson cautioned.
Wilder, the patient advocate, reminded the audience that “There is a cohort of people with ME who got sick in the 1980s and 1990s in the prime of their life ... They have dreamed of a day when there would be a major announcement that a treatment has been discovered to take away the suffering of this disease ... They keep waiting and waiting, year after year, missing more and more of their lives with each passing day ... We’re all depending on you.”
Dr. Systrom has received funding from the Solve ME/CFS Initiative, Department of Defense, and Open Medicine Foundation. Dr. Younger’s work is funded by the NIH, Department of Defense, SolveME, the American Fibromyalgia Association, and ME Research UK. Dr. Lipkin and Dr. Hanson receive NIH funding. Dr. Komaroff has no disclosures.
A version of this article appeared on Medscape.com.
BETHESDA, MD — New research into the mechanisms underlying myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) and long COVID is aimed at identifying potential approaches to treatment of the two overlapping illnesses.
According to a new data brief from the National Center for Health Statistics, in 2021-2022, 1.3% of US adults had ME/CFS, a complex, multisystem illness characterized by activity-limiting fatigue, worsening of symptoms after exertion, unrefreshing sleep, and other symptoms.
A 2-day conference, Advancing ME/CFS Research: Identifying Targets for Potential Intervention and Learning from Long COVID, was held in December 12-13 on the main campus of the US National Institutes of Health (NIH) and was livestreamed. The last such meeting, also featuring results from NIH-funded research, was held in April 2019.
“Things have changed since 2019 ... The idea of this meeting is to try and identify pathways that will be treatable and druggable and really make an impact for patients based on the things that we’ve learned over the last number of years and including, fortunately or unfortunately, the huge number of people who are suffering from long COVID, where the symptoms overlap so much with those who have been suffering for a long time with ME/CFS,” said Conference Chair Joe Breen, PhD, of the National Institute of Allergy and Infectious Diseases.
As in 2019, the meeting was preceded by a day of research presentations from young investigators, some of whom also presented their findings at the main meeting. New this year were four “lived experience” speakers who described their physical, emotional, and financial struggles with ME/CFS or long COVID. Two of them presented virtually because they were too ill to travel.
Social worker and patient advocate Terri Wilder of Minneapolis, Minnesota, reported some feedback she received on social media after she asked people with ME/CFS about their priorities for the research and clinical communities.
Among the top responses were the need to recognize and study the phenomenon of “post-exertional malaise” and to stop recommending exercise for people with these illnesses, to accelerate research to find effective treatments, and to put an end to stigma around the condition. “People don’t believe us when we tell them we’re sick, people make fun of us, misperceptions persist,” Wilder said.
One person commented, “[Clinicians] shouldn’t be afraid to try off-label meds with us if needed. There may be some secondary effects, but they are better options than us taking our own lives because we can’t stand the suffering.”
Research areas covered at the conference included immunology, virology, metabolism, gene regulation, and neurology of both ME/CFS and long COVID, as well as the latest findings regarding the overlap between the two conditions.
Oxidative Stress in Both ME/CFS and Long COVID: A Role for Metformin?
Mark M. Davis, PhD, professor and director of the Institute for Immunity, Transplantation, and Infection at Stanford University, Palo Alto, California, summarized published data suggesting that oxidative stress is a shared characteristic of both ME/CFS and long COVID. Most cellular reactive oxygen species (ROS) are produced in the cell’s mitochondria, and T-cell activation is ROS-dependent.
Women in particular with ME/CFS show high ROS levels with consistent T-cell hyperproliferation, “which can be suppressed with specific drugs such as metformin. This raises the prospect of optimizing drug treatment and drug discovery with a simple in vitro assay of the effects on a patient’s lymphocytes,” Dr. Davis said. He also cited a study suggesting that metformin may help prevent long COVID.
Asked to comment on that, longtime ME/CFS researcher Anthony L. Komaroff, MD, of Harvard University, Cambridge, Massachusetts, cautioned that although metformin is used safely by millions of people with type 2 diabetes worldwide, it’s possible that some people with ME/CFS may be more likely to experience its known adverse effects such as lactic acidosis.
To repurpose metformin or any other already-marketed drugs for ME/CFS and/or long COVID, Dr. Komaroff said, “We should entertain treatment trials.” However, as he and many others lamented at the conference, funding for off-patent drugs often isn’t forthcoming.
Addressing the Microbiome, Innate Immunity
W. Ian Lipkin, MD, of Columbia University, New York, NY, was one of two speakers who discussed the role of disruptions in the microbiome and innate immunity in ME/CFS. He presented data suggesting that “dysregulation of the gut microbiome in ME/CFS may interfere with butyrate production, resulting in inflammation and porosity to bacteria and bacterial products that trigger innate immunity.”
Dr. Lipkin highlighted a “really intriguing” paper in which exogenous administration of interleukin 37 (IL-37), a naturally occurring inhibitor of inflammation, reversed the decrease in exercise performance observed during inflammation-induced fatigue and increased exercise performance, both in mice.
“Although we do not fully understand the pathophysiology of ME/CFS, it is not premature to consider randomized clinical trials of pro- and pre-biotics that address dysbiosis as well as drugs that modify innate immune responses such as poly (I:C) and IL-37,” Dr. Lipkin said.
Alleviating Endoplasmic Reticulum (ER) Stress: A Strategy to Increase Energy?
Paul M. Hwang, MD, PhD, from the Cardiovascular Branch of the National Heart, Lung, and Blood Institute, Bethesda, Maryland, described work that he and his colleagues recently published around a case of a 38-year-old woman with Li-Fraumeni syndrome, a genetic early-onset cancer, who also had extensive fatigue, exercise intolerance, and post-exertional malaise that began after she contracted mononucleosis as a teenager.
Testing revealed that her cells had increased expression of Wiskott-Aldrich Syndrome Protein Family Member 3 (WASF3), a “top candidate” gene found to be associated with ME/CFS in a bioinformatics study published more than a decade ago. Moreover, immunoblotting of deidentified skeletal muscle biopsy samples obtained from patients with postinfectious ME/CFS also revealed significantly increased WASF3 levels.
Hwang and colleagues showed in mice that ER stress–induced WASF3 protein localizes to mitochondria and disrupts respiratory supercomplex assembly, leading to decreased oxygen consumption and exercise endurance.
However, use of the investigational protein phosphatase 1 inhibitor salubrinal in the female patient’s cells inhibited the ER stress, which in turn decreased WASF3 expression and improved mitochondrial supercomplex formation and respiration, “suggesting a treatment strategy in ME/CFS,” Dr. Hwang said.
Neurovascular Dysregulation During Exercise: A Role for Pyridostigmine?
David M. Systrom, MD, a pulmonary and critical care medicine specialist at the Brigham and Women’s Hospital, Boston, Massachusetts, gave an update of his work investigating neurovascular dysregulation during exercise in both ME/CFS and long COVID using invasive cardiopulmonary testing.
In a 2021 publication, Dr. Systrom and his colleagues identified the mechanism of “preload failure,” or lower filling pressures of blood in the heart chambers because of insufficient vein constriction and reduced return of blood to the right side of the heart in people with ME/CFS, compared with healthy controls.
Subsequently, in a randomized trial of 45 patients with ME/CFS, Systrom and his colleagues published in November 2022, use of the cholinesterase inhibitor pyridostigmine, currently approved for treating myasthenia gravis and related conditions, improved peak Vo2 by increasing cardiac output and filling pressures.
Now, Dr. Systrom’s team is conducting a randomized trial comparing 60 mg pyridostigmine with or without low-dose naltrexone (LDN) vs placebo in 160 patients with ME/CFS for 3 months. Metabolomic, transcriptomic, proteomic, and other assessments will be conducted on urine and blood samples. Participants will also wear devices that measure steps, sleep, heart rate, and other metrics.
Komaroff cautioned that pyridostigmine, too, has potential adverse effects. “I’m not sure pyridostigmine is ready for prime time ... It’s a drug developed for a very different purpose ... Now will it hold up in a larger trial, and will there be any side effects that turn up in larger studies? It’s not unreasonable to study.”
Brain Inflammation: Measuring and Treating It
Hannah F. Bues, clinical research coordinator at Massachusetts General Hospital, Boston, presented data now in preprint (ie, not yet peer-reviewed) in which researchers used [11C]PBR28 PET neuroimaging, a marker of neuroinflammation, to compare 12 individuals with long COVID vs 43 healthy controls. They found significantly increased neuroinflammation in several different brain regions in the long COVID group compared with controls.
Samples of peripheral blood plasma also showed significant correlations between neuroinflammation and circulating analytes related to vascular dysfunction. This work is ongoing in both long COVID and pre-COVID ME/CFS populations, Bues said.
Jarred Younger, PhD, of the Neuroinflammation, Pain, and Fatigue Laboratory at the University of Alabama, Birmingham, also gave an update of his ongoing work demonstrating significant brain inflammation seen in neuroimaging of people with ME/CFS compared with healthy controls.
Dr. Younger has been investigating the use of LDN for pain in fibromyalgia. Anecdotally, there have been reports of fatigue reduction with LDN in ME/CFS.
Dr. Younger conducted a post hoc analysis of his previous trial of LDN for 12 weeks in 30 patients with fibromyalgia. Of those, 16 met older CFS criteria. There was a significant reduction in their fatigue severity, with P <.0001 from baseline and P < .009 compared with placebo. The P values were high because the data included daily symptom reports. The average fatigue reduction was 25%.
“It wasn’t a study designed for ME/CFS, but I think it’s compelling evidence and enough with the other types of data we have to say we need to do a proper clinical trial of low-dose naltrexone in ME/CFS now,” Dr. Younger said.
‘We Need to Do Something’ About the Underfunding
Another NIH-funded ME/CFS researcher, Maureen Hanson, PhD, of Cornell University, Ithaca, NY, noted that the NIH currently funds ME/CFS research at about $13 million compared with $1.15 billion for the Researching COVID to Enhance Recovery Initiative granted to NIH by Congress for “post-acute sequelae of SARS-CoV-2 (PASC)” in 2021 “because of the urgency of studying this. Most of us here are well aware of the underfunding of ME/CFS relative to the burden of illness,” she said.
Current 2024 funding for AIDS research is $3294 million. “There are 1.2 million individuals living with HIV in the United States, and there are over 3 million who are barely living with ME/CFS in the United States. We need to do something about this ... It’s certainly possible that future funding for PASC is now going to disappear,” Dr. Hanson cautioned.
Wilder, the patient advocate, reminded the audience that “There is a cohort of people with ME who got sick in the 1980s and 1990s in the prime of their life ... They have dreamed of a day when there would be a major announcement that a treatment has been discovered to take away the suffering of this disease ... They keep waiting and waiting, year after year, missing more and more of their lives with each passing day ... We’re all depending on you.”
Dr. Systrom has received funding from the Solve ME/CFS Initiative, Department of Defense, and Open Medicine Foundation. Dr. Younger’s work is funded by the NIH, Department of Defense, SolveME, the American Fibromyalgia Association, and ME Research UK. Dr. Lipkin and Dr. Hanson receive NIH funding. Dr. Komaroff has no disclosures.
A version of this article appeared on Medscape.com.
BETHESDA, MD — New research into the mechanisms underlying myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) and long COVID is aimed at identifying potential approaches to treatment of the two overlapping illnesses.
According to a new data brief from the National Center for Health Statistics, in 2021-2022, 1.3% of US adults had ME/CFS, a complex, multisystem illness characterized by activity-limiting fatigue, worsening of symptoms after exertion, unrefreshing sleep, and other symptoms.
A 2-day conference, Advancing ME/CFS Research: Identifying Targets for Potential Intervention and Learning from Long COVID, was held in December 12-13 on the main campus of the US National Institutes of Health (NIH) and was livestreamed. The last such meeting, also featuring results from NIH-funded research, was held in April 2019.
“Things have changed since 2019 ... The idea of this meeting is to try and identify pathways that will be treatable and druggable and really make an impact for patients based on the things that we’ve learned over the last number of years and including, fortunately or unfortunately, the huge number of people who are suffering from long COVID, where the symptoms overlap so much with those who have been suffering for a long time with ME/CFS,” said Conference Chair Joe Breen, PhD, of the National Institute of Allergy and Infectious Diseases.
As in 2019, the meeting was preceded by a day of research presentations from young investigators, some of whom also presented their findings at the main meeting. New this year were four “lived experience” speakers who described their physical, emotional, and financial struggles with ME/CFS or long COVID. Two of them presented virtually because they were too ill to travel.
Social worker and patient advocate Terri Wilder of Minneapolis, Minnesota, reported some feedback she received on social media after she asked people with ME/CFS about their priorities for the research and clinical communities.
Among the top responses were the need to recognize and study the phenomenon of “post-exertional malaise” and to stop recommending exercise for people with these illnesses, to accelerate research to find effective treatments, and to put an end to stigma around the condition. “People don’t believe us when we tell them we’re sick, people make fun of us, misperceptions persist,” Wilder said.
One person commented, “[Clinicians] shouldn’t be afraid to try off-label meds with us if needed. There may be some secondary effects, but they are better options than us taking our own lives because we can’t stand the suffering.”
Research areas covered at the conference included immunology, virology, metabolism, gene regulation, and neurology of both ME/CFS and long COVID, as well as the latest findings regarding the overlap between the two conditions.
Oxidative Stress in Both ME/CFS and Long COVID: A Role for Metformin?
Mark M. Davis, PhD, professor and director of the Institute for Immunity, Transplantation, and Infection at Stanford University, Palo Alto, California, summarized published data suggesting that oxidative stress is a shared characteristic of both ME/CFS and long COVID. Most cellular reactive oxygen species (ROS) are produced in the cell’s mitochondria, and T-cell activation is ROS-dependent.
Women in particular with ME/CFS show high ROS levels with consistent T-cell hyperproliferation, “which can be suppressed with specific drugs such as metformin. This raises the prospect of optimizing drug treatment and drug discovery with a simple in vitro assay of the effects on a patient’s lymphocytes,” Dr. Davis said. He also cited a study suggesting that metformin may help prevent long COVID.
Asked to comment on that, longtime ME/CFS researcher Anthony L. Komaroff, MD, of Harvard University, Cambridge, Massachusetts, cautioned that although metformin is used safely by millions of people with type 2 diabetes worldwide, it’s possible that some people with ME/CFS may be more likely to experience its known adverse effects such as lactic acidosis.
To repurpose metformin or any other already-marketed drugs for ME/CFS and/or long COVID, Dr. Komaroff said, “We should entertain treatment trials.” However, as he and many others lamented at the conference, funding for off-patent drugs often isn’t forthcoming.
Addressing the Microbiome, Innate Immunity
W. Ian Lipkin, MD, of Columbia University, New York, NY, was one of two speakers who discussed the role of disruptions in the microbiome and innate immunity in ME/CFS. He presented data suggesting that “dysregulation of the gut microbiome in ME/CFS may interfere with butyrate production, resulting in inflammation and porosity to bacteria and bacterial products that trigger innate immunity.”
Dr. Lipkin highlighted a “really intriguing” paper in which exogenous administration of interleukin 37 (IL-37), a naturally occurring inhibitor of inflammation, reversed the decrease in exercise performance observed during inflammation-induced fatigue and increased exercise performance, both in mice.
“Although we do not fully understand the pathophysiology of ME/CFS, it is not premature to consider randomized clinical trials of pro- and pre-biotics that address dysbiosis as well as drugs that modify innate immune responses such as poly (I:C) and IL-37,” Dr. Lipkin said.
Alleviating Endoplasmic Reticulum (ER) Stress: A Strategy to Increase Energy?
Paul M. Hwang, MD, PhD, from the Cardiovascular Branch of the National Heart, Lung, and Blood Institute, Bethesda, Maryland, described work that he and his colleagues recently published around a case of a 38-year-old woman with Li-Fraumeni syndrome, a genetic early-onset cancer, who also had extensive fatigue, exercise intolerance, and post-exertional malaise that began after she contracted mononucleosis as a teenager.
Testing revealed that her cells had increased expression of Wiskott-Aldrich Syndrome Protein Family Member 3 (WASF3), a “top candidate” gene found to be associated with ME/CFS in a bioinformatics study published more than a decade ago. Moreover, immunoblotting of deidentified skeletal muscle biopsy samples obtained from patients with postinfectious ME/CFS also revealed significantly increased WASF3 levels.
Hwang and colleagues showed in mice that ER stress–induced WASF3 protein localizes to mitochondria and disrupts respiratory supercomplex assembly, leading to decreased oxygen consumption and exercise endurance.
However, use of the investigational protein phosphatase 1 inhibitor salubrinal in the female patient’s cells inhibited the ER stress, which in turn decreased WASF3 expression and improved mitochondrial supercomplex formation and respiration, “suggesting a treatment strategy in ME/CFS,” Dr. Hwang said.
Neurovascular Dysregulation During Exercise: A Role for Pyridostigmine?
David M. Systrom, MD, a pulmonary and critical care medicine specialist at the Brigham and Women’s Hospital, Boston, Massachusetts, gave an update of his work investigating neurovascular dysregulation during exercise in both ME/CFS and long COVID using invasive cardiopulmonary testing.
In a 2021 publication, Dr. Systrom and his colleagues identified the mechanism of “preload failure,” or lower filling pressures of blood in the heart chambers because of insufficient vein constriction and reduced return of blood to the right side of the heart in people with ME/CFS, compared with healthy controls.
Subsequently, in a randomized trial of 45 patients with ME/CFS, Systrom and his colleagues published in November 2022, use of the cholinesterase inhibitor pyridostigmine, currently approved for treating myasthenia gravis and related conditions, improved peak Vo2 by increasing cardiac output and filling pressures.
Now, Dr. Systrom’s team is conducting a randomized trial comparing 60 mg pyridostigmine with or without low-dose naltrexone (LDN) vs placebo in 160 patients with ME/CFS for 3 months. Metabolomic, transcriptomic, proteomic, and other assessments will be conducted on urine and blood samples. Participants will also wear devices that measure steps, sleep, heart rate, and other metrics.
Komaroff cautioned that pyridostigmine, too, has potential adverse effects. “I’m not sure pyridostigmine is ready for prime time ... It’s a drug developed for a very different purpose ... Now will it hold up in a larger trial, and will there be any side effects that turn up in larger studies? It’s not unreasonable to study.”
Brain Inflammation: Measuring and Treating It
Hannah F. Bues, clinical research coordinator at Massachusetts General Hospital, Boston, presented data now in preprint (ie, not yet peer-reviewed) in which researchers used [11C]PBR28 PET neuroimaging, a marker of neuroinflammation, to compare 12 individuals with long COVID vs 43 healthy controls. They found significantly increased neuroinflammation in several different brain regions in the long COVID group compared with controls.
Samples of peripheral blood plasma also showed significant correlations between neuroinflammation and circulating analytes related to vascular dysfunction. This work is ongoing in both long COVID and pre-COVID ME/CFS populations, Bues said.
Jarred Younger, PhD, of the Neuroinflammation, Pain, and Fatigue Laboratory at the University of Alabama, Birmingham, also gave an update of his ongoing work demonstrating significant brain inflammation seen in neuroimaging of people with ME/CFS compared with healthy controls.
Dr. Younger has been investigating the use of LDN for pain in fibromyalgia. Anecdotally, there have been reports of fatigue reduction with LDN in ME/CFS.
Dr. Younger conducted a post hoc analysis of his previous trial of LDN for 12 weeks in 30 patients with fibromyalgia. Of those, 16 met older CFS criteria. There was a significant reduction in their fatigue severity, with P <.0001 from baseline and P < .009 compared with placebo. The P values were high because the data included daily symptom reports. The average fatigue reduction was 25%.
“It wasn’t a study designed for ME/CFS, but I think it’s compelling evidence and enough with the other types of data we have to say we need to do a proper clinical trial of low-dose naltrexone in ME/CFS now,” Dr. Younger said.
‘We Need to Do Something’ About the Underfunding
Another NIH-funded ME/CFS researcher, Maureen Hanson, PhD, of Cornell University, Ithaca, NY, noted that the NIH currently funds ME/CFS research at about $13 million compared with $1.15 billion for the Researching COVID to Enhance Recovery Initiative granted to NIH by Congress for “post-acute sequelae of SARS-CoV-2 (PASC)” in 2021 “because of the urgency of studying this. Most of us here are well aware of the underfunding of ME/CFS relative to the burden of illness,” she said.
Current 2024 funding for AIDS research is $3294 million. “There are 1.2 million individuals living with HIV in the United States, and there are over 3 million who are barely living with ME/CFS in the United States. We need to do something about this ... It’s certainly possible that future funding for PASC is now going to disappear,” Dr. Hanson cautioned.
Wilder, the patient advocate, reminded the audience that “There is a cohort of people with ME who got sick in the 1980s and 1990s in the prime of their life ... They have dreamed of a day when there would be a major announcement that a treatment has been discovered to take away the suffering of this disease ... They keep waiting and waiting, year after year, missing more and more of their lives with each passing day ... We’re all depending on you.”
Dr. Systrom has received funding from the Solve ME/CFS Initiative, Department of Defense, and Open Medicine Foundation. Dr. Younger’s work is funded by the NIH, Department of Defense, SolveME, the American Fibromyalgia Association, and ME Research UK. Dr. Lipkin and Dr. Hanson receive NIH funding. Dr. Komaroff has no disclosures.
A version of this article appeared on Medscape.com.
FROM AN NIH RESEARCH CONFERENCE
H pylori Infection Linked to Increased Alzheimer’s Risk
TOPLINE:
results of a large and lengthy population-based study suggest.
METHODOLOGY:
- Researchers identified all cases with a first-time diagnosis of AD and matched each AD case to up to 40 AD-free control cases on the basis of age, sex, cohort entry date, and duration of follow-up.
- The exposure of interest was CAHPI, defined based on an algorithm using clinical guidelines and recommendations on the management of H pylori (HP) infection, with researchers focusing on infected individuals presenting with symptoms or developing serious complications from the infection.
- Researchers performed several sensitivity analyses, which included repeating the primary analysis using alternate lag periods, restricting the cohort to participants with AD (not vascular, alcoholic, and unspecified dementia), and using salmonellosis, an infection not previously associated with AD, as a negative control exposure.
TAKEAWAY:
- Compared with no exposure to CAHPI, exposure to CAHPI was associated with a moderately increased risk for AD (odds ratio [OR], 1.11; 95% CI, 1.01-1.21), with no major effect modification by demographics or socioeconomic status.
- The increased risk peaked 7.3-10.8 years after CAHPI onset (OR, 1.24; 95% CI, 1.05-1.47) before decreasing.
- Sensitivity analyses yielded findings that were overall consistent with those of the primary analysis.
- The analysis with salmonellosis as a negative control exposure showed no association with the risk for AD (OR, 1.03; 95% CI, 0.82-1.29).
IN PRACTICE:
“These results support the notion of HP infection as a potential modifiable risk factor of AD” and “pave the way for future randomized controlled trials that would assess the impact and cost-effectiveness of population-based targeted interventions such as individualized HP eradication programs, on the development of AD,” the authors write.
SOURCE:
The study was conducted by Antonios Douros, Department of Medicine, and Department of Epidemiology, Biostatistics, and Occupational Health, McGill University, Montreal, Quebec, Canada, and colleagues. It was published online in Alzheimer’s & Dementia.
LIMITATIONS:
Given the observational nature of the study, residual confounding is possible. Because the exposure definition was on the basis of CAHPI recorded by general practitioners, exposure misclassification due to symptomatic patients not seeking primary care is possible, as is outcome misclassification. The authors can’t rule out the possibility of an association between asymptomatic H pylori infection and AD risk.
DISCLOSURES:
The study received funding from the Canadian Institutes of Health Research. Douros has no relevant conflicts of interest; see paper for disclosures of other authors.
Pauline Anderson has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
TOPLINE:
results of a large and lengthy population-based study suggest.
METHODOLOGY:
- Researchers identified all cases with a first-time diagnosis of AD and matched each AD case to up to 40 AD-free control cases on the basis of age, sex, cohort entry date, and duration of follow-up.
- The exposure of interest was CAHPI, defined based on an algorithm using clinical guidelines and recommendations on the management of H pylori (HP) infection, with researchers focusing on infected individuals presenting with symptoms or developing serious complications from the infection.
- Researchers performed several sensitivity analyses, which included repeating the primary analysis using alternate lag periods, restricting the cohort to participants with AD (not vascular, alcoholic, and unspecified dementia), and using salmonellosis, an infection not previously associated with AD, as a negative control exposure.
TAKEAWAY:
- Compared with no exposure to CAHPI, exposure to CAHPI was associated with a moderately increased risk for AD (odds ratio [OR], 1.11; 95% CI, 1.01-1.21), with no major effect modification by demographics or socioeconomic status.
- The increased risk peaked 7.3-10.8 years after CAHPI onset (OR, 1.24; 95% CI, 1.05-1.47) before decreasing.
- Sensitivity analyses yielded findings that were overall consistent with those of the primary analysis.
- The analysis with salmonellosis as a negative control exposure showed no association with the risk for AD (OR, 1.03; 95% CI, 0.82-1.29).
IN PRACTICE:
“These results support the notion of HP infection as a potential modifiable risk factor of AD” and “pave the way for future randomized controlled trials that would assess the impact and cost-effectiveness of population-based targeted interventions such as individualized HP eradication programs, on the development of AD,” the authors write.
SOURCE:
The study was conducted by Antonios Douros, Department of Medicine, and Department of Epidemiology, Biostatistics, and Occupational Health, McGill University, Montreal, Quebec, Canada, and colleagues. It was published online in Alzheimer’s & Dementia.
LIMITATIONS:
Given the observational nature of the study, residual confounding is possible. Because the exposure definition was on the basis of CAHPI recorded by general practitioners, exposure misclassification due to symptomatic patients not seeking primary care is possible, as is outcome misclassification. The authors can’t rule out the possibility of an association between asymptomatic H pylori infection and AD risk.
DISCLOSURES:
The study received funding from the Canadian Institutes of Health Research. Douros has no relevant conflicts of interest; see paper for disclosures of other authors.
Pauline Anderson has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
TOPLINE:
results of a large and lengthy population-based study suggest.
METHODOLOGY:
- Researchers identified all cases with a first-time diagnosis of AD and matched each AD case to up to 40 AD-free control cases on the basis of age, sex, cohort entry date, and duration of follow-up.
- The exposure of interest was CAHPI, defined based on an algorithm using clinical guidelines and recommendations on the management of H pylori (HP) infection, with researchers focusing on infected individuals presenting with symptoms or developing serious complications from the infection.
- Researchers performed several sensitivity analyses, which included repeating the primary analysis using alternate lag periods, restricting the cohort to participants with AD (not vascular, alcoholic, and unspecified dementia), and using salmonellosis, an infection not previously associated with AD, as a negative control exposure.
TAKEAWAY:
- Compared with no exposure to CAHPI, exposure to CAHPI was associated with a moderately increased risk for AD (odds ratio [OR], 1.11; 95% CI, 1.01-1.21), with no major effect modification by demographics or socioeconomic status.
- The increased risk peaked 7.3-10.8 years after CAHPI onset (OR, 1.24; 95% CI, 1.05-1.47) before decreasing.
- Sensitivity analyses yielded findings that were overall consistent with those of the primary analysis.
- The analysis with salmonellosis as a negative control exposure showed no association with the risk for AD (OR, 1.03; 95% CI, 0.82-1.29).
IN PRACTICE:
“These results support the notion of HP infection as a potential modifiable risk factor of AD” and “pave the way for future randomized controlled trials that would assess the impact and cost-effectiveness of population-based targeted interventions such as individualized HP eradication programs, on the development of AD,” the authors write.
SOURCE:
The study was conducted by Antonios Douros, Department of Medicine, and Department of Epidemiology, Biostatistics, and Occupational Health, McGill University, Montreal, Quebec, Canada, and colleagues. It was published online in Alzheimer’s & Dementia.
LIMITATIONS:
Given the observational nature of the study, residual confounding is possible. Because the exposure definition was on the basis of CAHPI recorded by general practitioners, exposure misclassification due to symptomatic patients not seeking primary care is possible, as is outcome misclassification. The authors can’t rule out the possibility of an association between asymptomatic H pylori infection and AD risk.
DISCLOSURES:
The study received funding from the Canadian Institutes of Health Research. Douros has no relevant conflicts of interest; see paper for disclosures of other authors.
Pauline Anderson has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
COVID Strain JN.1 Is Now a ‘Variant of Interest,’ WHO Says
the global health agency has announced.
JN.1 was previously grouped with its relative, BA.2.86, but has increased so much in the past 4 weeks that the WHO moved it to standalone status, according to a summary published by the agency. The prevalence of JN.1 worldwide jumped from 3% for the week ending November 5 to 27% for the week ending December 3. During that same period, JN.1 rose from 1% to 66% of cases in the Western Pacific, which stretches across 37 countries, from China and Mongolia to Australia and New Zealand.
In the United States, JN.1 has been increasing rapidly. The variant accounted for an estimated 21% of cases for the 2-week period ending December 9, up from 8% during the 2 weeks prior.
SARS-CoV-2 is the virus that causes COVID, and like other viruses, it evolves over time, sometimes changing how the virus affects people or how well existing treatments and vaccines work against it.
The WHO and CDC have said the current COVID vaccine appears to protect people against severe symptoms due to JN.1, and the WHO called the rising variant’s public health risk “low.”
“As we observe the rise of the JN.1 variant, it’s important to note that while it may be spreading more widely, there is currently no significant evidence suggesting it is more severe or that it poses a substantial public health risk,” John Brownstein, PhD, chief innovation officer at Boston Children’s Hospital, told ABC News.
In its recent risk analysis, the WHO did acknowledge that it’s not certain whether JN.1 has a higher risk of evading immunity or causing more severe symptoms than other strains. The WHO advised countries to further study how much JN.1 can evade existing antibodies and whether the variant results in more severe disease.
The latest CDC data show that 11% of COVID tests reported to the agency are positive, and 23,432 people were hospitalized with severe symptoms within a 7-day period. The CDC urgently asked people to get vaccinated against respiratory illnesses like the flu and COVID-19 ahead of the holidays as cases rise nationwide.
“Getting vaccinated now can help prevent hospitalizations and save lives,” the agency advised.
A version of this article originally appeared on WebMD.com.
the global health agency has announced.
JN.1 was previously grouped with its relative, BA.2.86, but has increased so much in the past 4 weeks that the WHO moved it to standalone status, according to a summary published by the agency. The prevalence of JN.1 worldwide jumped from 3% for the week ending November 5 to 27% for the week ending December 3. During that same period, JN.1 rose from 1% to 66% of cases in the Western Pacific, which stretches across 37 countries, from China and Mongolia to Australia and New Zealand.
In the United States, JN.1 has been increasing rapidly. The variant accounted for an estimated 21% of cases for the 2-week period ending December 9, up from 8% during the 2 weeks prior.
SARS-CoV-2 is the virus that causes COVID, and like other viruses, it evolves over time, sometimes changing how the virus affects people or how well existing treatments and vaccines work against it.
The WHO and CDC have said the current COVID vaccine appears to protect people against severe symptoms due to JN.1, and the WHO called the rising variant’s public health risk “low.”
“As we observe the rise of the JN.1 variant, it’s important to note that while it may be spreading more widely, there is currently no significant evidence suggesting it is more severe or that it poses a substantial public health risk,” John Brownstein, PhD, chief innovation officer at Boston Children’s Hospital, told ABC News.
In its recent risk analysis, the WHO did acknowledge that it’s not certain whether JN.1 has a higher risk of evading immunity or causing more severe symptoms than other strains. The WHO advised countries to further study how much JN.1 can evade existing antibodies and whether the variant results in more severe disease.
The latest CDC data show that 11% of COVID tests reported to the agency are positive, and 23,432 people were hospitalized with severe symptoms within a 7-day period. The CDC urgently asked people to get vaccinated against respiratory illnesses like the flu and COVID-19 ahead of the holidays as cases rise nationwide.
“Getting vaccinated now can help prevent hospitalizations and save lives,” the agency advised.
A version of this article originally appeared on WebMD.com.
the global health agency has announced.
JN.1 was previously grouped with its relative, BA.2.86, but has increased so much in the past 4 weeks that the WHO moved it to standalone status, according to a summary published by the agency. The prevalence of JN.1 worldwide jumped from 3% for the week ending November 5 to 27% for the week ending December 3. During that same period, JN.1 rose from 1% to 66% of cases in the Western Pacific, which stretches across 37 countries, from China and Mongolia to Australia and New Zealand.
In the United States, JN.1 has been increasing rapidly. The variant accounted for an estimated 21% of cases for the 2-week period ending December 9, up from 8% during the 2 weeks prior.
SARS-CoV-2 is the virus that causes COVID, and like other viruses, it evolves over time, sometimes changing how the virus affects people or how well existing treatments and vaccines work against it.
The WHO and CDC have said the current COVID vaccine appears to protect people against severe symptoms due to JN.1, and the WHO called the rising variant’s public health risk “low.”
“As we observe the rise of the JN.1 variant, it’s important to note that while it may be spreading more widely, there is currently no significant evidence suggesting it is more severe or that it poses a substantial public health risk,” John Brownstein, PhD, chief innovation officer at Boston Children’s Hospital, told ABC News.
In its recent risk analysis, the WHO did acknowledge that it’s not certain whether JN.1 has a higher risk of evading immunity or causing more severe symptoms than other strains. The WHO advised countries to further study how much JN.1 can evade existing antibodies and whether the variant results in more severe disease.
The latest CDC data show that 11% of COVID tests reported to the agency are positive, and 23,432 people were hospitalized with severe symptoms within a 7-day period. The CDC urgently asked people to get vaccinated against respiratory illnesses like the flu and COVID-19 ahead of the holidays as cases rise nationwide.
“Getting vaccinated now can help prevent hospitalizations and save lives,” the agency advised.
A version of this article originally appeared on WebMD.com.
Reactive Angioendotheliomatosis Following Ad26.COV2.S Vaccination
To the Editor:
Reactive angioendotheliomatosis (RAE) is a rare self-limited cutaneous vascular proliferation of endothelial cells within blood vessels that manifests clinically as infiltrated red-blue patches and plaques with purpura that can progress to occlude vascular lumina. The etiology of RAE is mostly idiopathic; however, the disorder typically occurs in association with a range of systemic diseases, including infection, cryoglobulinemia, leukemia, antiphospholipid syndrome, peripheral vascular disease, and arteriovenous fistula. Histopathologic examination of these lesions shows marked proliferation of endothelial cells, including occlusion of the lumen of blood vessels over wide areas.
After ruling out malignancy, treatment of RAE focuses on targeting the underlying cause or disease, if any is present; 75% of reported cases occur in association with systemic disease.1 Onset can occur at any age without predilection for sex. Reactive angioendotheliomatosis commonly manifests on the extremities but may occur on the head and neck in rare instances.2
The rarity of the condition and its poorly defined clinical characteristics make it difficult to develop a treatment plan. There are no standardized treatment guidelines for the reactive form of angiomatosis. We report a case of RAE that developed 2 weeks after vaccination with the Ad26.COV2.S vaccine (Johnson & Johnson Innovative Medicine [formerly Janssen Pharmaceutical Companies of Johnson & Johnson]) that improved following 2 weeks of treatment with a topical corticosteroid and an oral antihistamine.
A 58-year-old man presented to an outpatient dermatology clinic with pruritus and occasional paresthesia associated with a rash over the left arm of 1 month’s duration. The patient suspected that the rash may have formed secondary to the bite of oak mites on the arms and chest while he was carrying milled wood. Further inquiry into the patient’s history revealed that he received the Ad26.COV2.S vaccine 2 weeks prior to the appearance of the rash. He denied mechanical trauma. His medical history included hypercholesterolemia and a mild COVID-19 infection 8 months prior to the appearance of the rash that did not require hospitalization. He denied fever or chills during the 2 weeks following vaccination. The pruritus was minimally relieved for short periods with over-the-counter calamine lotion. The patient’s medication regimen included daily pravastatin and loratadine at the time of the initial visit. He used acetaminophen as needed for knee pain.
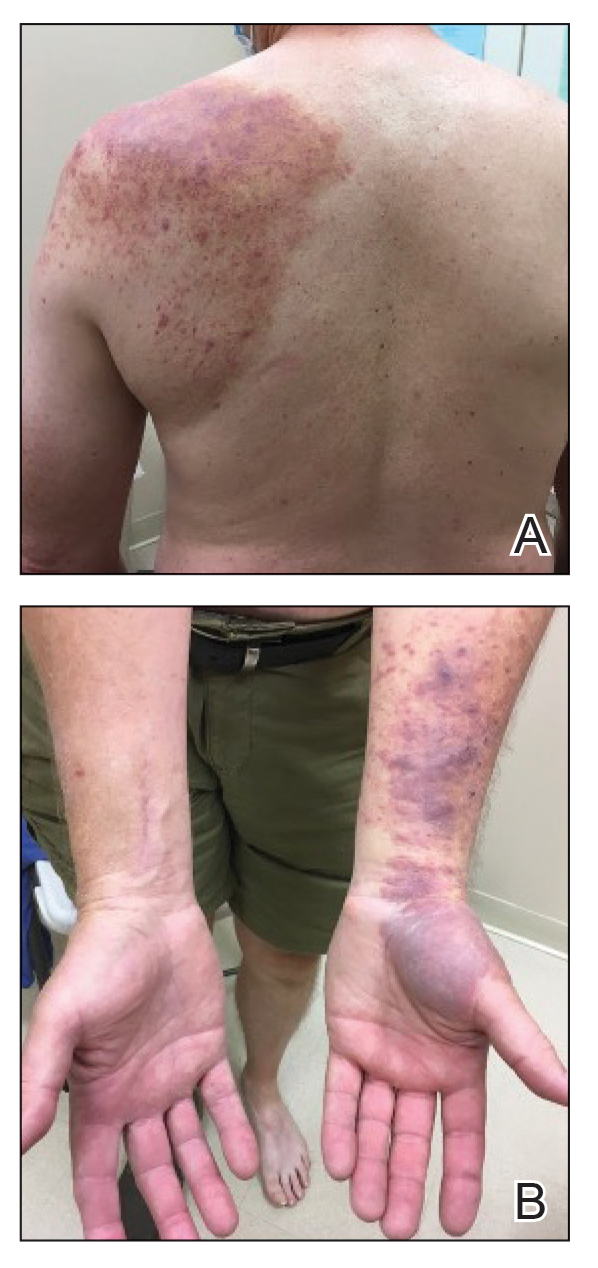
Physical examination revealed palpable purpura in a dermatomal distribution with nonpitting edema over the left scapula (Figure 1A), left anterolateral shoulder, left lateral volar forearm, and thenar eminence of the left hand (Figure 1B). Notably, the entire right arm, conjunctivae, tongue, lips, and bilateral fingernails were clear. Three 4-mm punch biopsies were performed at the initial presentation: 1 perilesional biopsy for direct immunofluorescence testing and 2 lesional biopsies for routine histologic evaluation. An extensive serologic workup failed to reveal abnormalities. An activated partial thromboplastin time, dilute Russell viper venom time, serum protein electrophoresis, and levels of rheumatoid factor and angiotensin-converting enzyme were within reference range. Anticardiolipin antibodies IgA, IgM, and IgG were negative. A cryoglobulin test was negative.
Histopathology revealed a proliferation of irregularly shaped vascular spaces with plump endothelium in the papillary dermis (Figure 2). Scattered leukocyte common antigen-positive lymphocytes were noted within lesions. The epidermis appeared normal, without evidence of spongiosis or alteration of the stratum corneum. Immunohistochemical studies of the perilesional skin biopsy revealed positivity for CD31 and D2-40 (Figure 3). Specimens were negative for CD20 and human herpesvirus 8. Direct immunofluorescence of the perilesional biopsy was negative.
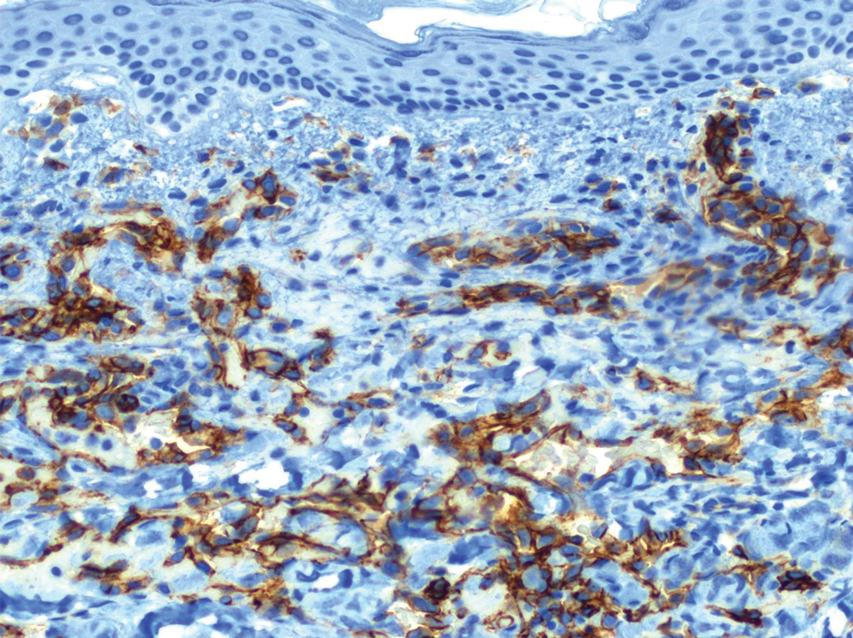
A diagnosis of RAE was made based on clinical and histologic findings. Treatment with triamcinolone ointment 0.1% twice daily and oral cetirizine 10 mg twice daily was initiated. Re-evaluation 2 weeks later revealed notable improvement in the affected areas, including decreased edema, improvement of the purpura, and absence of pruritus. The patient noted no further spread or blister formation while the active areas were being treated with the topical steroid. The treatment regimen was modified to triamcinolone ointment 0.1% once daily, and cetirizine was discontinued. At 3-month follow-up, active areas had completely resolved (Figure 4) and triamcinolone was discontinued. To date, the patient has not had recurrence of symptoms and remains healthy.
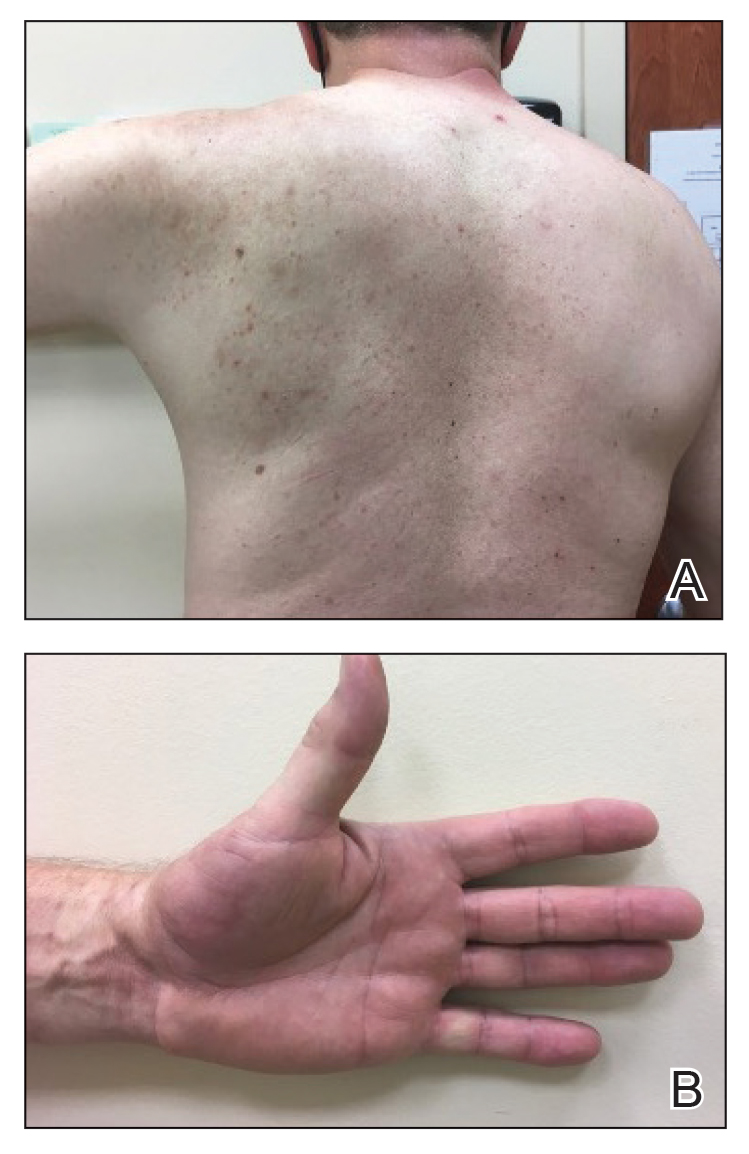
Gottron and Nikolowski3 reported the first case of RAE in an adult patient who presented with purpuric patches secondary to skin infarction. Current definitions use the umbrella term cutaneous reactive angiomatosis to cover 3 major subtypes: reactive angioendotheliomatosis, diffuse dermal angioendotheliomatosis, and acroangiodermatitis (pseudo-Kaposi sarcoma [KS]). The manifestation of these subgroups is clinically similar, and they must be differentiated through histologic evaluation.4
Reactive angioendotheliomatosis has an unknown pathogenesis and is poorly defined clinically. The exact pathophysiology is unknown but likely is linked to vaso-occlusion and hypoxia.1 A PubMed search of articles indexed for MEDLINE, as well as a review of Science Direct, Google Scholar, and Cochrane Library, using the terms reactive angioendotheliomatosis, COVID, vaccine, Ad26.COV2.S, and RAE in any combination revealed no prior cases of RAE in association with Ad26.COV2.S vaccination.
By the late 1980s, systemic angioendotheliomatosis was segregated into 2 distinct entities: malignant and reactive.4 The differential diagnosis of malignant systemic angioendotheliomatosis includes KS and angiosarcoma; nonmalignant causes are the variants of cutaneous reactive angiomatosis. It is important to rule out KS because of its malignant and deceptive nature. It is unknown if KS originates in blood vessels or lymphatic endothelial cells; however, evidence is strongly in favor of blood vessel origin using CD31 and CD34 endothelial markers.5 CD34 positivity is more reliable than CD31 in diagnosing KS, but the absence of both markers does not offer enough evidence to rule out KS on its own.6
In our patient, histopathology revealed cells positive for CD31 and D2-40; the latter is a lymphatic endothelial cell marker that stains the endothelium of lymphatic channels but not blood vessels.7 Positive D2-40 can be indicative of KS and non-KS lesions, each with a distinct staining pattern. D2-40 staining on non-KS lesions is confined to lymphatic vessels, as it was in our patient; in contrast, spindle-shaped cells also will be stained in KS lesions.8
Another cell marker, CD20, is a B cell–specific protein that can be measured to help diagnose malignant diseases such as B-cell lymphoma and leukemia. Human herpesvirus 8 (also known as KS-associated herpesvirus) is the infectious cause of KS and traditionally has been detected using methods such as the polymerase chain reaction.9,10
Most cases of RAE are idiopathic and occur in association with systemic disease, which was not the case in our patient. We speculated that his reaction was most likely triggered by vascular transfection of endothelial cells secondary to Ad26.COV2.S vaccination. Alternatively, vaccination may have caused vascular occlusion, though the lack of cyanosis, nail changes, and route of inoculant make this less likely.
All approved COVID-19 vaccines are designed solely for intramuscular injection. In comparison to other types of tissue, muscles have superior vascularity, allowing for enhanced mobilization of compounds, which results in faster systemic circulation.11 Alternative methods of injection, including intravascular, subcutaneous, and intradermal, may lead to decreased efficacy or adverse events, or both.
Prior cases of RAE have been treated with laser therapy, topical or systemic corticosteroids, excisional removal, or topical β-blockers, such as timolol.12 β-Blocking agents act on β-adrenergic receptors on endothelial cells to inhibit angiogenesis by reducing release of blood vessel growth-signaling molecules and triggering apoptosis. In this patient, topical steroids and oral antihistamines were sufficient treatment.
Vaccine-related adverse events have been reported but remain rare. The benefits of Ad26.COV2.S vaccination for protection against COVID-19 outweigh the extremely low risk for adverse events.13 For that reason, the Centers for Disease Control and Prevention recommends a booster for individuals who are eligible to maximize protection. Intramuscular injection of Ad26.COV2.S resulted in a lower incidence of moderate to severe COVID-19 cases in all age groups vs the placebo group. Hypersensitivity adverse events were reported in 0.4% of Ad26.COV2.S-vaccinated patients vs 0.4% of patients who received a placebo; the more common reactions were nonanaphylactic.13
There have been 12 reports of cerebral venous sinus thrombosis with thrombocytopenia after Ad26.COV2.S vaccination, which sparked nationwide controversy over the safety of the Ad26.COV2.S vaccine.14 After further investigation into those reports, the US Food and Drug Administration and the Centers for Disease Control and Prevention concluded that the benefits of the Ad26.COV2.S vaccine outweigh the low risk for associated thrombosis.15
Although adverse reactions are rare, it is important that health care providers take proper safety measures before and while administering any COVID-19 vaccine. Patients should be screened for contraindications to the COVID-19 vaccine to mitigate adverse effects seen in the small percentage of patients who may need to take alternative precautions.
The broad tissue tropism and high transmissibility of SARS-CoV-2 are the main contributors to its infection having reached pandemic scale. The spike (S) protein on SARS-CoV-2 binds to ACE2, the most thoroughly studied SARS-CoV-2 receptor, which is found in a range of tissues, including arterial endothelial cells, leading to its transfection. Several studies have proposed that expression of the S protein causes endothelial dysfunction through cytokine release, activation of complement, and ultimately microvascular occlusion.16
Recent developments in the use of viral-like particles, such as vesicular stomatitis virus, may mitigate future cases of RAE that are associated with endothelial cell transfection. Vesicular stomatitis virus is a popular model virus for research applications due to its glycoprotein and matrix protein contributing to its broad tropism. Recent efforts to alter these proteins have successfully limited the broad tropism of vesicular stomatitis virus.17
The SARS-CoV-2 virus must be handled in a Biosafety Level 3 laboratory. Conversely, pseudoviruses can be handled in lower containment facilities due to their safe and efficacious nature, offering an avenue to expedite vaccine development against many viral outbreaks, including SARS-CoV-2.18
An increasing number of cutaneous manifestations have been associated with COVID-19 infection and vaccination. Eruptive pseudoangiomatosis, a rare self-limiting exanthem, has been reported in association with COVID-19 vaccination.19 Eruptive pseudoangiomatosis manifests as erythematous blanchable papules that resemble angiomas, typically in a widespread distribution. Eruptive pseudoangiomatosis has striking similarities to RAE histologically; both manifest as dilated dermal blood vessels with plump endothelial cells.
Our case is unique because of the vasculitic palpable nature of the lesions, which were localized to the left arm. Eruptive pseudoangiomatosis formation after COVID-19 infection or SARS-CoV-2 vaccination may suggest alteration of ACE2 by binding of S protein.20 Such alteration of the ACE2 pathway would lead to inflammation of angiotensin II, causing proliferation of endothelial cells in the formation of angiomalike lesions. This hypothesis suggests a paraviral eruption secondary to an immunologic reaction, not a classical virtual eruption from direct contact of the virus on blood vessels. Although EPA and RAE are harmless and self-limiting, these reports will spread awareness of the increasing number of skin manifestations related to COVID-19 and SARS-CoV-2 virus vaccination.
Acknowledgment—Thoughtful insights and comments on this manuscript were provided by Christine J. Ko, MD (New Haven, Connecticut); Christine L. Egan, MD (Glen Mills, Pennsylvania); Howard A. Bueller, MD (Delray Beach, Florida); and Juan Pablo Robles, PhD (Juriquilla, Mexico).
- McMenamin ME, Fletcher CDM. Reactive angioendotheliomatosis: a study of 15 cases demonstrating a wide clinicopathologic spectrum. Am J Surg Pathol. 2002;26:686-697. doi:10.1097/00000478-200206000-00001
- Khan S, Pujani M, Jetley S, et al. Angiomatosis: a rare vascular proliferation of head and neck region. J Cutan Aesthet Surg. 2015;8:108-110. doi:10.4103/0974-2077.158448
- Gottron HA, Nikolowski W. Extrarenal Lohlein focal nephritis of the skin in endocarditis. Arch Klin Exp Dermatol. 1958;207:156-176.
- Cooper PH. Angioendotheliomatosis: two separate diseases. J Cutan Pathol. 1988;15:259. doi:10.1111/j.1600-0560.1988.tb00556.x
- Cancian L, Hansen A, Boshoff C. Cellular origin of Kaposi’s sarcoma and Kaposi’s sarcoma-associated herpesvirus-induced cell reprogramming. Trends Cell Biol. Sep 2013;23:421-32. doi:10.1016/j.tcb.2013.04.001
- Russell Jones R, Orchard G, Zelger B, et al. Immunostaining for CD31 and CD34 in Kaposi sarcoma. J Clin Pathol. 1995;48:1011-1016. doi:10.1136/jcp.48.11.1011
- Kahn HJ, Bailey D, Marks A. Monoclonal antibody D2-40, a new marker of lymphatic endothelium, reacts with Kaposi’s sarcoma and a subset of angiosarcomas. Mod Pathol. 2002;15:434-440. doi:10.1038/modpathol.3880543
- Genedy RM, Hamza AM, Abdel Latef AA, et al. Sensitivity and specificity of D2-40 in differentiating Kaposi sarcoma from its mimickers. J Egyptian Womens Dermatolog Soc. 2021;18:67-74. doi:10.4103/jewd.jewd_61_20
- Mesri EA, Cesarman E, Boshoff C. Kaposi’s sarcoma and its associated herpesvirus. Nat Rev Cancer. 2010;10:707-719. doi:10.1038/nrc2888
- Patel RM, Goldblum JR, Hsi ED. Immunohistochemical detection of human herpes virus-8 latent nuclear antigen-1 is useful in the diagnosis of Kaposi sarcoma. Mod Pathol. 2004;17:456-460. doi:10.1038/modpathol.3800061
- Zuckerman JN. The importance of injecting vaccines into muscle. Different patients need different needle sizes. BMJ. 2000;321:1237-1238. doi:10.1136/bmj.321.7271.1237
- Bhatia R, Hazarika N, Chandrasekaran D, et al. Treatment of posttraumatic reactive angioendotheliomatosis with topical timolol maleate. JAMA Dermatol. 2021;157:1002-1004. doi:10.1001/jamadermatol.2021.1770
- Sadoff J, Gray G, Vandebosch A, et al; ENSEMBLE Study Group. Safety and efficacy of single-dose Ad26.COV2.S vaccine against Covid-19. N Engl J Med. 2021;384:2187-2201. doi:10.1056/NEJMoa2101544
- See I, Su JR, Lale A, et al. US case reports of cerebral venous sinus thrombosis with thrombocytopenia after Ad26.COV2.S vaccination, March 2 to April 21, 2021. JAMA. 2021;325:2448-2456. doi:10.1001/jama.2021.7517
- Berry CT, Eliliwi M, Gallagher S, et al. Cutaneous small vessel vasculitis following single-dose Janssen Ad26.COV2.S vaccination. JAAD Case Rep. 2021;15:11-14. doi:10.1016/j.jdcr.2021.07.002
- Flaumenhaft R, Enjyoji K, Schmaier AA. Vasculopathy in COVID-19. Blood. 2022;140:222-235. doi:10.1182/blood.2021012250
- Hastie E, Cataldi M, Marriott I, et al. Understanding and altering cell tropism of vesicular stomatitis virus. Virus Res. 2013;176:16-32. doi:10.1016/j.virusres.2013.06.003
- Xiong H-L, Wu Y-T, Cao J-L, et al. Robust neutralization assay based on SARS-CoV-2 S-protein-bearing vesicular stomatitis virus (VSV) pseudovirus and ACE2-overexpressing BHK21 cells. Emerg Microbes Infect. 2020;9:2105-2113. doi:10.1080/22221751.2020.1815589
- Mohta A, Jain SK, Mehta RD, et al. Development of eruptive pseudoangiomatosis following COVID-19 immunization – apropos of 5 cases. J Eur Acad Dermatol Venereol. 2021;35:e722-e725. doi:10.1111/jdv.17499
- Angeli F, Spanevello A, Reboldi G, et al. SARS-CoV-2 vaccines: lights and shadows. Eur J Intern Med. 2021;88:1-8. doi:10.1016/j.ejim.2021.04.019
To the Editor:
Reactive angioendotheliomatosis (RAE) is a rare self-limited cutaneous vascular proliferation of endothelial cells within blood vessels that manifests clinically as infiltrated red-blue patches and plaques with purpura that can progress to occlude vascular lumina. The etiology of RAE is mostly idiopathic; however, the disorder typically occurs in association with a range of systemic diseases, including infection, cryoglobulinemia, leukemia, antiphospholipid syndrome, peripheral vascular disease, and arteriovenous fistula. Histopathologic examination of these lesions shows marked proliferation of endothelial cells, including occlusion of the lumen of blood vessels over wide areas.
After ruling out malignancy, treatment of RAE focuses on targeting the underlying cause or disease, if any is present; 75% of reported cases occur in association with systemic disease.1 Onset can occur at any age without predilection for sex. Reactive angioendotheliomatosis commonly manifests on the extremities but may occur on the head and neck in rare instances.2
The rarity of the condition and its poorly defined clinical characteristics make it difficult to develop a treatment plan. There are no standardized treatment guidelines for the reactive form of angiomatosis. We report a case of RAE that developed 2 weeks after vaccination with the Ad26.COV2.S vaccine (Johnson & Johnson Innovative Medicine [formerly Janssen Pharmaceutical Companies of Johnson & Johnson]) that improved following 2 weeks of treatment with a topical corticosteroid and an oral antihistamine.
A 58-year-old man presented to an outpatient dermatology clinic with pruritus and occasional paresthesia associated with a rash over the left arm of 1 month’s duration. The patient suspected that the rash may have formed secondary to the bite of oak mites on the arms and chest while he was carrying milled wood. Further inquiry into the patient’s history revealed that he received the Ad26.COV2.S vaccine 2 weeks prior to the appearance of the rash. He denied mechanical trauma. His medical history included hypercholesterolemia and a mild COVID-19 infection 8 months prior to the appearance of the rash that did not require hospitalization. He denied fever or chills during the 2 weeks following vaccination. The pruritus was minimally relieved for short periods with over-the-counter calamine lotion. The patient’s medication regimen included daily pravastatin and loratadine at the time of the initial visit. He used acetaminophen as needed for knee pain.

Physical examination revealed palpable purpura in a dermatomal distribution with nonpitting edema over the left scapula (Figure 1A), left anterolateral shoulder, left lateral volar forearm, and thenar eminence of the left hand (Figure 1B). Notably, the entire right arm, conjunctivae, tongue, lips, and bilateral fingernails were clear. Three 4-mm punch biopsies were performed at the initial presentation: 1 perilesional biopsy for direct immunofluorescence testing and 2 lesional biopsies for routine histologic evaluation. An extensive serologic workup failed to reveal abnormalities. An activated partial thromboplastin time, dilute Russell viper venom time, serum protein electrophoresis, and levels of rheumatoid factor and angiotensin-converting enzyme were within reference range. Anticardiolipin antibodies IgA, IgM, and IgG were negative. A cryoglobulin test was negative.

Histopathology revealed a proliferation of irregularly shaped vascular spaces with plump endothelium in the papillary dermis (Figure 2). Scattered leukocyte common antigen-positive lymphocytes were noted within lesions. The epidermis appeared normal, without evidence of spongiosis or alteration of the stratum corneum. Immunohistochemical studies of the perilesional skin biopsy revealed positivity for CD31 and D2-40 (Figure 3). Specimens were negative for CD20 and human herpesvirus 8. Direct immunofluorescence of the perilesional biopsy was negative.

A diagnosis of RAE was made based on clinical and histologic findings. Treatment with triamcinolone ointment 0.1% twice daily and oral cetirizine 10 mg twice daily was initiated. Re-evaluation 2 weeks later revealed notable improvement in the affected areas, including decreased edema, improvement of the purpura, and absence of pruritus. The patient noted no further spread or blister formation while the active areas were being treated with the topical steroid. The treatment regimen was modified to triamcinolone ointment 0.1% once daily, and cetirizine was discontinued. At 3-month follow-up, active areas had completely resolved (Figure 4) and triamcinolone was discontinued. To date, the patient has not had recurrence of symptoms and remains healthy.

Gottron and Nikolowski3 reported the first case of RAE in an adult patient who presented with purpuric patches secondary to skin infarction. Current definitions use the umbrella term cutaneous reactive angiomatosis to cover 3 major subtypes: reactive angioendotheliomatosis, diffuse dermal angioendotheliomatosis, and acroangiodermatitis (pseudo-Kaposi sarcoma [KS]). The manifestation of these subgroups is clinically similar, and they must be differentiated through histologic evaluation.4
Reactive angioendotheliomatosis has an unknown pathogenesis and is poorly defined clinically. The exact pathophysiology is unknown but likely is linked to vaso-occlusion and hypoxia.1 A PubMed search of articles indexed for MEDLINE, as well as a review of Science Direct, Google Scholar, and Cochrane Library, using the terms reactive angioendotheliomatosis, COVID, vaccine, Ad26.COV2.S, and RAE in any combination revealed no prior cases of RAE in association with Ad26.COV2.S vaccination.
By the late 1980s, systemic angioendotheliomatosis was segregated into 2 distinct entities: malignant and reactive.4 The differential diagnosis of malignant systemic angioendotheliomatosis includes KS and angiosarcoma; nonmalignant causes are the variants of cutaneous reactive angiomatosis. It is important to rule out KS because of its malignant and deceptive nature. It is unknown if KS originates in blood vessels or lymphatic endothelial cells; however, evidence is strongly in favor of blood vessel origin using CD31 and CD34 endothelial markers.5 CD34 positivity is more reliable than CD31 in diagnosing KS, but the absence of both markers does not offer enough evidence to rule out KS on its own.6
In our patient, histopathology revealed cells positive for CD31 and D2-40; the latter is a lymphatic endothelial cell marker that stains the endothelium of lymphatic channels but not blood vessels.7 Positive D2-40 can be indicative of KS and non-KS lesions, each with a distinct staining pattern. D2-40 staining on non-KS lesions is confined to lymphatic vessels, as it was in our patient; in contrast, spindle-shaped cells also will be stained in KS lesions.8
Another cell marker, CD20, is a B cell–specific protein that can be measured to help diagnose malignant diseases such as B-cell lymphoma and leukemia. Human herpesvirus 8 (also known as KS-associated herpesvirus) is the infectious cause of KS and traditionally has been detected using methods such as the polymerase chain reaction.9,10
Most cases of RAE are idiopathic and occur in association with systemic disease, which was not the case in our patient. We speculated that his reaction was most likely triggered by vascular transfection of endothelial cells secondary to Ad26.COV2.S vaccination. Alternatively, vaccination may have caused vascular occlusion, though the lack of cyanosis, nail changes, and route of inoculant make this less likely.
All approved COVID-19 vaccines are designed solely for intramuscular injection. In comparison to other types of tissue, muscles have superior vascularity, allowing for enhanced mobilization of compounds, which results in faster systemic circulation.11 Alternative methods of injection, including intravascular, subcutaneous, and intradermal, may lead to decreased efficacy or adverse events, or both.
Prior cases of RAE have been treated with laser therapy, topical or systemic corticosteroids, excisional removal, or topical β-blockers, such as timolol.12 β-Blocking agents act on β-adrenergic receptors on endothelial cells to inhibit angiogenesis by reducing release of blood vessel growth-signaling molecules and triggering apoptosis. In this patient, topical steroids and oral antihistamines were sufficient treatment.
Vaccine-related adverse events have been reported but remain rare. The benefits of Ad26.COV2.S vaccination for protection against COVID-19 outweigh the extremely low risk for adverse events.13 For that reason, the Centers for Disease Control and Prevention recommends a booster for individuals who are eligible to maximize protection. Intramuscular injection of Ad26.COV2.S resulted in a lower incidence of moderate to severe COVID-19 cases in all age groups vs the placebo group. Hypersensitivity adverse events were reported in 0.4% of Ad26.COV2.S-vaccinated patients vs 0.4% of patients who received a placebo; the more common reactions were nonanaphylactic.13
There have been 12 reports of cerebral venous sinus thrombosis with thrombocytopenia after Ad26.COV2.S vaccination, which sparked nationwide controversy over the safety of the Ad26.COV2.S vaccine.14 After further investigation into those reports, the US Food and Drug Administration and the Centers for Disease Control and Prevention concluded that the benefits of the Ad26.COV2.S vaccine outweigh the low risk for associated thrombosis.15
Although adverse reactions are rare, it is important that health care providers take proper safety measures before and while administering any COVID-19 vaccine. Patients should be screened for contraindications to the COVID-19 vaccine to mitigate adverse effects seen in the small percentage of patients who may need to take alternative precautions.
The broad tissue tropism and high transmissibility of SARS-CoV-2 are the main contributors to its infection having reached pandemic scale. The spike (S) protein on SARS-CoV-2 binds to ACE2, the most thoroughly studied SARS-CoV-2 receptor, which is found in a range of tissues, including arterial endothelial cells, leading to its transfection. Several studies have proposed that expression of the S protein causes endothelial dysfunction through cytokine release, activation of complement, and ultimately microvascular occlusion.16
Recent developments in the use of viral-like particles, such as vesicular stomatitis virus, may mitigate future cases of RAE that are associated with endothelial cell transfection. Vesicular stomatitis virus is a popular model virus for research applications due to its glycoprotein and matrix protein contributing to its broad tropism. Recent efforts to alter these proteins have successfully limited the broad tropism of vesicular stomatitis virus.17
The SARS-CoV-2 virus must be handled in a Biosafety Level 3 laboratory. Conversely, pseudoviruses can be handled in lower containment facilities due to their safe and efficacious nature, offering an avenue to expedite vaccine development against many viral outbreaks, including SARS-CoV-2.18
An increasing number of cutaneous manifestations have been associated with COVID-19 infection and vaccination. Eruptive pseudoangiomatosis, a rare self-limiting exanthem, has been reported in association with COVID-19 vaccination.19 Eruptive pseudoangiomatosis manifests as erythematous blanchable papules that resemble angiomas, typically in a widespread distribution. Eruptive pseudoangiomatosis has striking similarities to RAE histologically; both manifest as dilated dermal blood vessels with plump endothelial cells.
Our case is unique because of the vasculitic palpable nature of the lesions, which were localized to the left arm. Eruptive pseudoangiomatosis formation after COVID-19 infection or SARS-CoV-2 vaccination may suggest alteration of ACE2 by binding of S protein.20 Such alteration of the ACE2 pathway would lead to inflammation of angiotensin II, causing proliferation of endothelial cells in the formation of angiomalike lesions. This hypothesis suggests a paraviral eruption secondary to an immunologic reaction, not a classical virtual eruption from direct contact of the virus on blood vessels. Although EPA and RAE are harmless and self-limiting, these reports will spread awareness of the increasing number of skin manifestations related to COVID-19 and SARS-CoV-2 virus vaccination.
Acknowledgment—Thoughtful insights and comments on this manuscript were provided by Christine J. Ko, MD (New Haven, Connecticut); Christine L. Egan, MD (Glen Mills, Pennsylvania); Howard A. Bueller, MD (Delray Beach, Florida); and Juan Pablo Robles, PhD (Juriquilla, Mexico).
To the Editor:
Reactive angioendotheliomatosis (RAE) is a rare self-limited cutaneous vascular proliferation of endothelial cells within blood vessels that manifests clinically as infiltrated red-blue patches and plaques with purpura that can progress to occlude vascular lumina. The etiology of RAE is mostly idiopathic; however, the disorder typically occurs in association with a range of systemic diseases, including infection, cryoglobulinemia, leukemia, antiphospholipid syndrome, peripheral vascular disease, and arteriovenous fistula. Histopathologic examination of these lesions shows marked proliferation of endothelial cells, including occlusion of the lumen of blood vessels over wide areas.
After ruling out malignancy, treatment of RAE focuses on targeting the underlying cause or disease, if any is present; 75% of reported cases occur in association with systemic disease.1 Onset can occur at any age without predilection for sex. Reactive angioendotheliomatosis commonly manifests on the extremities but may occur on the head and neck in rare instances.2
The rarity of the condition and its poorly defined clinical characteristics make it difficult to develop a treatment plan. There are no standardized treatment guidelines for the reactive form of angiomatosis. We report a case of RAE that developed 2 weeks after vaccination with the Ad26.COV2.S vaccine (Johnson & Johnson Innovative Medicine [formerly Janssen Pharmaceutical Companies of Johnson & Johnson]) that improved following 2 weeks of treatment with a topical corticosteroid and an oral antihistamine.
A 58-year-old man presented to an outpatient dermatology clinic with pruritus and occasional paresthesia associated with a rash over the left arm of 1 month’s duration. The patient suspected that the rash may have formed secondary to the bite of oak mites on the arms and chest while he was carrying milled wood. Further inquiry into the patient’s history revealed that he received the Ad26.COV2.S vaccine 2 weeks prior to the appearance of the rash. He denied mechanical trauma. His medical history included hypercholesterolemia and a mild COVID-19 infection 8 months prior to the appearance of the rash that did not require hospitalization. He denied fever or chills during the 2 weeks following vaccination. The pruritus was minimally relieved for short periods with over-the-counter calamine lotion. The patient’s medication regimen included daily pravastatin and loratadine at the time of the initial visit. He used acetaminophen as needed for knee pain.

Physical examination revealed palpable purpura in a dermatomal distribution with nonpitting edema over the left scapula (Figure 1A), left anterolateral shoulder, left lateral volar forearm, and thenar eminence of the left hand (Figure 1B). Notably, the entire right arm, conjunctivae, tongue, lips, and bilateral fingernails were clear. Three 4-mm punch biopsies were performed at the initial presentation: 1 perilesional biopsy for direct immunofluorescence testing and 2 lesional biopsies for routine histologic evaluation. An extensive serologic workup failed to reveal abnormalities. An activated partial thromboplastin time, dilute Russell viper venom time, serum protein electrophoresis, and levels of rheumatoid factor and angiotensin-converting enzyme were within reference range. Anticardiolipin antibodies IgA, IgM, and IgG were negative. A cryoglobulin test was negative.

Histopathology revealed a proliferation of irregularly shaped vascular spaces with plump endothelium in the papillary dermis (Figure 2). Scattered leukocyte common antigen-positive lymphocytes were noted within lesions. The epidermis appeared normal, without evidence of spongiosis or alteration of the stratum corneum. Immunohistochemical studies of the perilesional skin biopsy revealed positivity for CD31 and D2-40 (Figure 3). Specimens were negative for CD20 and human herpesvirus 8. Direct immunofluorescence of the perilesional biopsy was negative.

A diagnosis of RAE was made based on clinical and histologic findings. Treatment with triamcinolone ointment 0.1% twice daily and oral cetirizine 10 mg twice daily was initiated. Re-evaluation 2 weeks later revealed notable improvement in the affected areas, including decreased edema, improvement of the purpura, and absence of pruritus. The patient noted no further spread or blister formation while the active areas were being treated with the topical steroid. The treatment regimen was modified to triamcinolone ointment 0.1% once daily, and cetirizine was discontinued. At 3-month follow-up, active areas had completely resolved (Figure 4) and triamcinolone was discontinued. To date, the patient has not had recurrence of symptoms and remains healthy.

Gottron and Nikolowski3 reported the first case of RAE in an adult patient who presented with purpuric patches secondary to skin infarction. Current definitions use the umbrella term cutaneous reactive angiomatosis to cover 3 major subtypes: reactive angioendotheliomatosis, diffuse dermal angioendotheliomatosis, and acroangiodermatitis (pseudo-Kaposi sarcoma [KS]). The manifestation of these subgroups is clinically similar, and they must be differentiated through histologic evaluation.4
Reactive angioendotheliomatosis has an unknown pathogenesis and is poorly defined clinically. The exact pathophysiology is unknown but likely is linked to vaso-occlusion and hypoxia.1 A PubMed search of articles indexed for MEDLINE, as well as a review of Science Direct, Google Scholar, and Cochrane Library, using the terms reactive angioendotheliomatosis, COVID, vaccine, Ad26.COV2.S, and RAE in any combination revealed no prior cases of RAE in association with Ad26.COV2.S vaccination.
By the late 1980s, systemic angioendotheliomatosis was segregated into 2 distinct entities: malignant and reactive.4 The differential diagnosis of malignant systemic angioendotheliomatosis includes KS and angiosarcoma; nonmalignant causes are the variants of cutaneous reactive angiomatosis. It is important to rule out KS because of its malignant and deceptive nature. It is unknown if KS originates in blood vessels or lymphatic endothelial cells; however, evidence is strongly in favor of blood vessel origin using CD31 and CD34 endothelial markers.5 CD34 positivity is more reliable than CD31 in diagnosing KS, but the absence of both markers does not offer enough evidence to rule out KS on its own.6
In our patient, histopathology revealed cells positive for CD31 and D2-40; the latter is a lymphatic endothelial cell marker that stains the endothelium of lymphatic channels but not blood vessels.7 Positive D2-40 can be indicative of KS and non-KS lesions, each with a distinct staining pattern. D2-40 staining on non-KS lesions is confined to lymphatic vessels, as it was in our patient; in contrast, spindle-shaped cells also will be stained in KS lesions.8
Another cell marker, CD20, is a B cell–specific protein that can be measured to help diagnose malignant diseases such as B-cell lymphoma and leukemia. Human herpesvirus 8 (also known as KS-associated herpesvirus) is the infectious cause of KS and traditionally has been detected using methods such as the polymerase chain reaction.9,10
Most cases of RAE are idiopathic and occur in association with systemic disease, which was not the case in our patient. We speculated that his reaction was most likely triggered by vascular transfection of endothelial cells secondary to Ad26.COV2.S vaccination. Alternatively, vaccination may have caused vascular occlusion, though the lack of cyanosis, nail changes, and route of inoculant make this less likely.
All approved COVID-19 vaccines are designed solely for intramuscular injection. In comparison to other types of tissue, muscles have superior vascularity, allowing for enhanced mobilization of compounds, which results in faster systemic circulation.11 Alternative methods of injection, including intravascular, subcutaneous, and intradermal, may lead to decreased efficacy or adverse events, or both.
Prior cases of RAE have been treated with laser therapy, topical or systemic corticosteroids, excisional removal, or topical β-blockers, such as timolol.12 β-Blocking agents act on β-adrenergic receptors on endothelial cells to inhibit angiogenesis by reducing release of blood vessel growth-signaling molecules and triggering apoptosis. In this patient, topical steroids and oral antihistamines were sufficient treatment.
Vaccine-related adverse events have been reported but remain rare. The benefits of Ad26.COV2.S vaccination for protection against COVID-19 outweigh the extremely low risk for adverse events.13 For that reason, the Centers for Disease Control and Prevention recommends a booster for individuals who are eligible to maximize protection. Intramuscular injection of Ad26.COV2.S resulted in a lower incidence of moderate to severe COVID-19 cases in all age groups vs the placebo group. Hypersensitivity adverse events were reported in 0.4% of Ad26.COV2.S-vaccinated patients vs 0.4% of patients who received a placebo; the more common reactions were nonanaphylactic.13
There have been 12 reports of cerebral venous sinus thrombosis with thrombocytopenia after Ad26.COV2.S vaccination, which sparked nationwide controversy over the safety of the Ad26.COV2.S vaccine.14 After further investigation into those reports, the US Food and Drug Administration and the Centers for Disease Control and Prevention concluded that the benefits of the Ad26.COV2.S vaccine outweigh the low risk for associated thrombosis.15
Although adverse reactions are rare, it is important that health care providers take proper safety measures before and while administering any COVID-19 vaccine. Patients should be screened for contraindications to the COVID-19 vaccine to mitigate adverse effects seen in the small percentage of patients who may need to take alternative precautions.
The broad tissue tropism and high transmissibility of SARS-CoV-2 are the main contributors to its infection having reached pandemic scale. The spike (S) protein on SARS-CoV-2 binds to ACE2, the most thoroughly studied SARS-CoV-2 receptor, which is found in a range of tissues, including arterial endothelial cells, leading to its transfection. Several studies have proposed that expression of the S protein causes endothelial dysfunction through cytokine release, activation of complement, and ultimately microvascular occlusion.16
Recent developments in the use of viral-like particles, such as vesicular stomatitis virus, may mitigate future cases of RAE that are associated with endothelial cell transfection. Vesicular stomatitis virus is a popular model virus for research applications due to its glycoprotein and matrix protein contributing to its broad tropism. Recent efforts to alter these proteins have successfully limited the broad tropism of vesicular stomatitis virus.17
The SARS-CoV-2 virus must be handled in a Biosafety Level 3 laboratory. Conversely, pseudoviruses can be handled in lower containment facilities due to their safe and efficacious nature, offering an avenue to expedite vaccine development against many viral outbreaks, including SARS-CoV-2.18
An increasing number of cutaneous manifestations have been associated with COVID-19 infection and vaccination. Eruptive pseudoangiomatosis, a rare self-limiting exanthem, has been reported in association with COVID-19 vaccination.19 Eruptive pseudoangiomatosis manifests as erythematous blanchable papules that resemble angiomas, typically in a widespread distribution. Eruptive pseudoangiomatosis has striking similarities to RAE histologically; both manifest as dilated dermal blood vessels with plump endothelial cells.
Our case is unique because of the vasculitic palpable nature of the lesions, which were localized to the left arm. Eruptive pseudoangiomatosis formation after COVID-19 infection or SARS-CoV-2 vaccination may suggest alteration of ACE2 by binding of S protein.20 Such alteration of the ACE2 pathway would lead to inflammation of angiotensin II, causing proliferation of endothelial cells in the formation of angiomalike lesions. This hypothesis suggests a paraviral eruption secondary to an immunologic reaction, not a classical virtual eruption from direct contact of the virus on blood vessels. Although EPA and RAE are harmless and self-limiting, these reports will spread awareness of the increasing number of skin manifestations related to COVID-19 and SARS-CoV-2 virus vaccination.
Acknowledgment—Thoughtful insights and comments on this manuscript were provided by Christine J. Ko, MD (New Haven, Connecticut); Christine L. Egan, MD (Glen Mills, Pennsylvania); Howard A. Bueller, MD (Delray Beach, Florida); and Juan Pablo Robles, PhD (Juriquilla, Mexico).
- McMenamin ME, Fletcher CDM. Reactive angioendotheliomatosis: a study of 15 cases demonstrating a wide clinicopathologic spectrum. Am J Surg Pathol. 2002;26:686-697. doi:10.1097/00000478-200206000-00001
- Khan S, Pujani M, Jetley S, et al. Angiomatosis: a rare vascular proliferation of head and neck region. J Cutan Aesthet Surg. 2015;8:108-110. doi:10.4103/0974-2077.158448
- Gottron HA, Nikolowski W. Extrarenal Lohlein focal nephritis of the skin in endocarditis. Arch Klin Exp Dermatol. 1958;207:156-176.
- Cooper PH. Angioendotheliomatosis: two separate diseases. J Cutan Pathol. 1988;15:259. doi:10.1111/j.1600-0560.1988.tb00556.x
- Cancian L, Hansen A, Boshoff C. Cellular origin of Kaposi’s sarcoma and Kaposi’s sarcoma-associated herpesvirus-induced cell reprogramming. Trends Cell Biol. Sep 2013;23:421-32. doi:10.1016/j.tcb.2013.04.001
- Russell Jones R, Orchard G, Zelger B, et al. Immunostaining for CD31 and CD34 in Kaposi sarcoma. J Clin Pathol. 1995;48:1011-1016. doi:10.1136/jcp.48.11.1011
- Kahn HJ, Bailey D, Marks A. Monoclonal antibody D2-40, a new marker of lymphatic endothelium, reacts with Kaposi’s sarcoma and a subset of angiosarcomas. Mod Pathol. 2002;15:434-440. doi:10.1038/modpathol.3880543
- Genedy RM, Hamza AM, Abdel Latef AA, et al. Sensitivity and specificity of D2-40 in differentiating Kaposi sarcoma from its mimickers. J Egyptian Womens Dermatolog Soc. 2021;18:67-74. doi:10.4103/jewd.jewd_61_20
- Mesri EA, Cesarman E, Boshoff C. Kaposi’s sarcoma and its associated herpesvirus. Nat Rev Cancer. 2010;10:707-719. doi:10.1038/nrc2888
- Patel RM, Goldblum JR, Hsi ED. Immunohistochemical detection of human herpes virus-8 latent nuclear antigen-1 is useful in the diagnosis of Kaposi sarcoma. Mod Pathol. 2004;17:456-460. doi:10.1038/modpathol.3800061
- Zuckerman JN. The importance of injecting vaccines into muscle. Different patients need different needle sizes. BMJ. 2000;321:1237-1238. doi:10.1136/bmj.321.7271.1237
- Bhatia R, Hazarika N, Chandrasekaran D, et al. Treatment of posttraumatic reactive angioendotheliomatosis with topical timolol maleate. JAMA Dermatol. 2021;157:1002-1004. doi:10.1001/jamadermatol.2021.1770
- Sadoff J, Gray G, Vandebosch A, et al; ENSEMBLE Study Group. Safety and efficacy of single-dose Ad26.COV2.S vaccine against Covid-19. N Engl J Med. 2021;384:2187-2201. doi:10.1056/NEJMoa2101544
- See I, Su JR, Lale A, et al. US case reports of cerebral venous sinus thrombosis with thrombocytopenia after Ad26.COV2.S vaccination, March 2 to April 21, 2021. JAMA. 2021;325:2448-2456. doi:10.1001/jama.2021.7517
- Berry CT, Eliliwi M, Gallagher S, et al. Cutaneous small vessel vasculitis following single-dose Janssen Ad26.COV2.S vaccination. JAAD Case Rep. 2021;15:11-14. doi:10.1016/j.jdcr.2021.07.002
- Flaumenhaft R, Enjyoji K, Schmaier AA. Vasculopathy in COVID-19. Blood. 2022;140:222-235. doi:10.1182/blood.2021012250
- Hastie E, Cataldi M, Marriott I, et al. Understanding and altering cell tropism of vesicular stomatitis virus. Virus Res. 2013;176:16-32. doi:10.1016/j.virusres.2013.06.003
- Xiong H-L, Wu Y-T, Cao J-L, et al. Robust neutralization assay based on SARS-CoV-2 S-protein-bearing vesicular stomatitis virus (VSV) pseudovirus and ACE2-overexpressing BHK21 cells. Emerg Microbes Infect. 2020;9:2105-2113. doi:10.1080/22221751.2020.1815589
- Mohta A, Jain SK, Mehta RD, et al. Development of eruptive pseudoangiomatosis following COVID-19 immunization – apropos of 5 cases. J Eur Acad Dermatol Venereol. 2021;35:e722-e725. doi:10.1111/jdv.17499
- Angeli F, Spanevello A, Reboldi G, et al. SARS-CoV-2 vaccines: lights and shadows. Eur J Intern Med. 2021;88:1-8. doi:10.1016/j.ejim.2021.04.019
- McMenamin ME, Fletcher CDM. Reactive angioendotheliomatosis: a study of 15 cases demonstrating a wide clinicopathologic spectrum. Am J Surg Pathol. 2002;26:686-697. doi:10.1097/00000478-200206000-00001
- Khan S, Pujani M, Jetley S, et al. Angiomatosis: a rare vascular proliferation of head and neck region. J Cutan Aesthet Surg. 2015;8:108-110. doi:10.4103/0974-2077.158448
- Gottron HA, Nikolowski W. Extrarenal Lohlein focal nephritis of the skin in endocarditis. Arch Klin Exp Dermatol. 1958;207:156-176.
- Cooper PH. Angioendotheliomatosis: two separate diseases. J Cutan Pathol. 1988;15:259. doi:10.1111/j.1600-0560.1988.tb00556.x
- Cancian L, Hansen A, Boshoff C. Cellular origin of Kaposi’s sarcoma and Kaposi’s sarcoma-associated herpesvirus-induced cell reprogramming. Trends Cell Biol. Sep 2013;23:421-32. doi:10.1016/j.tcb.2013.04.001
- Russell Jones R, Orchard G, Zelger B, et al. Immunostaining for CD31 and CD34 in Kaposi sarcoma. J Clin Pathol. 1995;48:1011-1016. doi:10.1136/jcp.48.11.1011
- Kahn HJ, Bailey D, Marks A. Monoclonal antibody D2-40, a new marker of lymphatic endothelium, reacts with Kaposi’s sarcoma and a subset of angiosarcomas. Mod Pathol. 2002;15:434-440. doi:10.1038/modpathol.3880543
- Genedy RM, Hamza AM, Abdel Latef AA, et al. Sensitivity and specificity of D2-40 in differentiating Kaposi sarcoma from its mimickers. J Egyptian Womens Dermatolog Soc. 2021;18:67-74. doi:10.4103/jewd.jewd_61_20
- Mesri EA, Cesarman E, Boshoff C. Kaposi’s sarcoma and its associated herpesvirus. Nat Rev Cancer. 2010;10:707-719. doi:10.1038/nrc2888
- Patel RM, Goldblum JR, Hsi ED. Immunohistochemical detection of human herpes virus-8 latent nuclear antigen-1 is useful in the diagnosis of Kaposi sarcoma. Mod Pathol. 2004;17:456-460. doi:10.1038/modpathol.3800061
- Zuckerman JN. The importance of injecting vaccines into muscle. Different patients need different needle sizes. BMJ. 2000;321:1237-1238. doi:10.1136/bmj.321.7271.1237
- Bhatia R, Hazarika N, Chandrasekaran D, et al. Treatment of posttraumatic reactive angioendotheliomatosis with topical timolol maleate. JAMA Dermatol. 2021;157:1002-1004. doi:10.1001/jamadermatol.2021.1770
- Sadoff J, Gray G, Vandebosch A, et al; ENSEMBLE Study Group. Safety and efficacy of single-dose Ad26.COV2.S vaccine against Covid-19. N Engl J Med. 2021;384:2187-2201. doi:10.1056/NEJMoa2101544
- See I, Su JR, Lale A, et al. US case reports of cerebral venous sinus thrombosis with thrombocytopenia after Ad26.COV2.S vaccination, March 2 to April 21, 2021. JAMA. 2021;325:2448-2456. doi:10.1001/jama.2021.7517
- Berry CT, Eliliwi M, Gallagher S, et al. Cutaneous small vessel vasculitis following single-dose Janssen Ad26.COV2.S vaccination. JAAD Case Rep. 2021;15:11-14. doi:10.1016/j.jdcr.2021.07.002
- Flaumenhaft R, Enjyoji K, Schmaier AA. Vasculopathy in COVID-19. Blood. 2022;140:222-235. doi:10.1182/blood.2021012250
- Hastie E, Cataldi M, Marriott I, et al. Understanding and altering cell tropism of vesicular stomatitis virus. Virus Res. 2013;176:16-32. doi:10.1016/j.virusres.2013.06.003
- Xiong H-L, Wu Y-T, Cao J-L, et al. Robust neutralization assay based on SARS-CoV-2 S-protein-bearing vesicular stomatitis virus (VSV) pseudovirus and ACE2-overexpressing BHK21 cells. Emerg Microbes Infect. 2020;9:2105-2113. doi:10.1080/22221751.2020.1815589
- Mohta A, Jain SK, Mehta RD, et al. Development of eruptive pseudoangiomatosis following COVID-19 immunization – apropos of 5 cases. J Eur Acad Dermatol Venereol. 2021;35:e722-e725. doi:10.1111/jdv.17499
- Angeli F, Spanevello A, Reboldi G, et al. SARS-CoV-2 vaccines: lights and shadows. Eur J Intern Med. 2021;88:1-8. doi:10.1016/j.ejim.2021.04.019
Practice points
- Reactive angioendotheliomatosis (RAE) is a rare benign vascular proliferation of endothelial cells lining blood vessels that clinically appears similar to Kaposi sarcoma and must be differentiated by microscopic evaluation.
- An increasing number of reports link SARS-CoV-2 viral infection or vaccination against this virus with various cutaneous manifestations. Our case offers a link between RAE and Ad26.COV2.S vaccination.