User login
Psychotropic med use tied to ‘striking’ post-COVID dementia risk
, new research suggests.
Results from a large study of more than 1,700 patients who had been hospitalized with COVID showed a greater than twofold increased risk for post-COVID dementia in those taking antipsychotics and mood stabilizers/anticonvulsants – medications often used to treat schizophrenia, psychosis, bipolar disorder, and seizures.
“We know that pre-existing psychiatric illness is associated with poor COVID-19 outcomes, but our study is the first to show an association with certain psychiatric medications and dementia,” co-investigator Liron Sinvani, MD, the Feinstein Institutes for Medical Research, Manhasset, New York, said in an interview.
“Our study highlights the potential interaction between baseline neuropsychiatric disease, psychotropic medications, COVID-19, and dementia,” Dr. Sinvani added.
The findings were published online March 18 in Frontiers in Medicine.
‘Striking’ dementia rate
Using electronic health records, the researchers evaluated pre-COVID psychotropic medication use and post-COVID dementia onset in 1,755 adults aged 65 and older. All were hospitalized with COVID-19 at Northwell Health between March 1 and April 20, 2020.
A “striking” 13% of the participants (n = 223) developed dementia within 1-year of follow-up, the investigators report.
Among the 438 patients (25%) exposed to at least one psychotropic medication before COVID-19, 105 (24%) developed dementia in the year following COVID versus 118 of 1,317 (9%) patients with no pre-COVID exposure to psychotropic medication (odds ratio, 3.2; 95% confidence interval, 2.37-4.32).
Both pre-COVID psychotropic medication use (OR, 2.7; 95% CI, 1.8-4.0, P < .001) and delirium (OR, 3.0; 95% CI, 1.9-4.6, P < .001) were significantly associated with post-COVID dementia at 1 year.
In a sensitivity analysis in the subset of 423 patients with at least one documented neurologic or psychiatric diagnosis at the time of COVID admission, and after adjusting for confounding factors, pre-COVID psychotropic medication use remained significantly linked to post-COVID dementia onset (OR, 3.09; 95% CI, 1.5-6.6, P = .002).
Drug classes most strongly associated with 1-year post-COVID dementia onset were antipsychotics (OR, 2.8, 95% CI, 1.7-4.4, P < .001) and mood stabilizers/anticonvulsants (OR, 2.4, 95% CI, 1.39-4.02, P = .001).
In a further exploratory analysis, the psychotropics valproic acid (multiple brands) and haloperidol (Haldol) had the largest association with post-COVID dementia.
Antidepressants as a class were not associated with post-COVID dementia, but the potential effects of two commonly prescribed antidepressants in older adults, mirtazapine (Remeron) and escitalopram (Lexapro), “warrant further investigation,” the researchers note.
Predictive risk marker?
“This research shows that psychotropic medications can be considered a predictive risk marker for post-COVID dementia. In patients taking psychotropic medications, COVID-19 could have accelerated progression of dementia after hospitalization,” lead author Yun Freudenberg-Hua, MD, the Feinstein Institutes, said in a news release.
It is unclear why psychotropic medications may raise the risk for dementia onset after COVID, the investigators note.
“It is intuitive that psychotropic medications indicate pre-existing neuropsychiatric conditions in which COVID-19 occurs. It is possible that psychotropic medications may potentiate the neurostructural changes that have been found in the brain of those who have recovered from COVID-19,” they write.
The sensitivity analysis in patients with documented neurologic and psychiatric diagnoses supports this interpretation.
COVID-19 may also accelerate the underlying brain disorders for which psychotropic medications were prescribed, leading to the greater incidence of post-COVID dementia, the researchers write.
“It is important to note that this study is in no way recommending people should stop taking antipsychotics but simply that clinicians need to factor in a patient’s medication history while considering post-COVID aftereffects,” Dr. Freudenberg-Hua said.
“Given that the number of patients with dementia is projected to triple in the next 30 years, these findings have significant public health implications,” Dr. Sinvani added.
She noted that “care partners and health care professionals” should look for early signs of dementia, such as forgetfulness and depressive symptoms, in their patients.
“Future studies must continue to evaluate these associations, which are key for potential future interventions to prevent dementia,” Dr. Sinvani said.
The study was funded by the National Institutes of Health. Dr. Freudenberg-Hua co-owns stock and stock options from Regeneron Pharmaceuticals. Dr. Sinvani has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research suggests.
Results from a large study of more than 1,700 patients who had been hospitalized with COVID showed a greater than twofold increased risk for post-COVID dementia in those taking antipsychotics and mood stabilizers/anticonvulsants – medications often used to treat schizophrenia, psychosis, bipolar disorder, and seizures.
“We know that pre-existing psychiatric illness is associated with poor COVID-19 outcomes, but our study is the first to show an association with certain psychiatric medications and dementia,” co-investigator Liron Sinvani, MD, the Feinstein Institutes for Medical Research, Manhasset, New York, said in an interview.
“Our study highlights the potential interaction between baseline neuropsychiatric disease, psychotropic medications, COVID-19, and dementia,” Dr. Sinvani added.
The findings were published online March 18 in Frontiers in Medicine.
‘Striking’ dementia rate
Using electronic health records, the researchers evaluated pre-COVID psychotropic medication use and post-COVID dementia onset in 1,755 adults aged 65 and older. All were hospitalized with COVID-19 at Northwell Health between March 1 and April 20, 2020.
A “striking” 13% of the participants (n = 223) developed dementia within 1-year of follow-up, the investigators report.
Among the 438 patients (25%) exposed to at least one psychotropic medication before COVID-19, 105 (24%) developed dementia in the year following COVID versus 118 of 1,317 (9%) patients with no pre-COVID exposure to psychotropic medication (odds ratio, 3.2; 95% confidence interval, 2.37-4.32).
Both pre-COVID psychotropic medication use (OR, 2.7; 95% CI, 1.8-4.0, P < .001) and delirium (OR, 3.0; 95% CI, 1.9-4.6, P < .001) were significantly associated with post-COVID dementia at 1 year.
In a sensitivity analysis in the subset of 423 patients with at least one documented neurologic or psychiatric diagnosis at the time of COVID admission, and after adjusting for confounding factors, pre-COVID psychotropic medication use remained significantly linked to post-COVID dementia onset (OR, 3.09; 95% CI, 1.5-6.6, P = .002).
Drug classes most strongly associated with 1-year post-COVID dementia onset were antipsychotics (OR, 2.8, 95% CI, 1.7-4.4, P < .001) and mood stabilizers/anticonvulsants (OR, 2.4, 95% CI, 1.39-4.02, P = .001).
In a further exploratory analysis, the psychotropics valproic acid (multiple brands) and haloperidol (Haldol) had the largest association with post-COVID dementia.
Antidepressants as a class were not associated with post-COVID dementia, but the potential effects of two commonly prescribed antidepressants in older adults, mirtazapine (Remeron) and escitalopram (Lexapro), “warrant further investigation,” the researchers note.
Predictive risk marker?
“This research shows that psychotropic medications can be considered a predictive risk marker for post-COVID dementia. In patients taking psychotropic medications, COVID-19 could have accelerated progression of dementia after hospitalization,” lead author Yun Freudenberg-Hua, MD, the Feinstein Institutes, said in a news release.
It is unclear why psychotropic medications may raise the risk for dementia onset after COVID, the investigators note.
“It is intuitive that psychotropic medications indicate pre-existing neuropsychiatric conditions in which COVID-19 occurs. It is possible that psychotropic medications may potentiate the neurostructural changes that have been found in the brain of those who have recovered from COVID-19,” they write.
The sensitivity analysis in patients with documented neurologic and psychiatric diagnoses supports this interpretation.
COVID-19 may also accelerate the underlying brain disorders for which psychotropic medications were prescribed, leading to the greater incidence of post-COVID dementia, the researchers write.
“It is important to note that this study is in no way recommending people should stop taking antipsychotics but simply that clinicians need to factor in a patient’s medication history while considering post-COVID aftereffects,” Dr. Freudenberg-Hua said.
“Given that the number of patients with dementia is projected to triple in the next 30 years, these findings have significant public health implications,” Dr. Sinvani added.
She noted that “care partners and health care professionals” should look for early signs of dementia, such as forgetfulness and depressive symptoms, in their patients.
“Future studies must continue to evaluate these associations, which are key for potential future interventions to prevent dementia,” Dr. Sinvani said.
The study was funded by the National Institutes of Health. Dr. Freudenberg-Hua co-owns stock and stock options from Regeneron Pharmaceuticals. Dr. Sinvani has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research suggests.
Results from a large study of more than 1,700 patients who had been hospitalized with COVID showed a greater than twofold increased risk for post-COVID dementia in those taking antipsychotics and mood stabilizers/anticonvulsants – medications often used to treat schizophrenia, psychosis, bipolar disorder, and seizures.
“We know that pre-existing psychiatric illness is associated with poor COVID-19 outcomes, but our study is the first to show an association with certain psychiatric medications and dementia,” co-investigator Liron Sinvani, MD, the Feinstein Institutes for Medical Research, Manhasset, New York, said in an interview.
“Our study highlights the potential interaction between baseline neuropsychiatric disease, psychotropic medications, COVID-19, and dementia,” Dr. Sinvani added.
The findings were published online March 18 in Frontiers in Medicine.
‘Striking’ dementia rate
Using electronic health records, the researchers evaluated pre-COVID psychotropic medication use and post-COVID dementia onset in 1,755 adults aged 65 and older. All were hospitalized with COVID-19 at Northwell Health between March 1 and April 20, 2020.
A “striking” 13% of the participants (n = 223) developed dementia within 1-year of follow-up, the investigators report.
Among the 438 patients (25%) exposed to at least one psychotropic medication before COVID-19, 105 (24%) developed dementia in the year following COVID versus 118 of 1,317 (9%) patients with no pre-COVID exposure to psychotropic medication (odds ratio, 3.2; 95% confidence interval, 2.37-4.32).
Both pre-COVID psychotropic medication use (OR, 2.7; 95% CI, 1.8-4.0, P < .001) and delirium (OR, 3.0; 95% CI, 1.9-4.6, P < .001) were significantly associated with post-COVID dementia at 1 year.
In a sensitivity analysis in the subset of 423 patients with at least one documented neurologic or psychiatric diagnosis at the time of COVID admission, and after adjusting for confounding factors, pre-COVID psychotropic medication use remained significantly linked to post-COVID dementia onset (OR, 3.09; 95% CI, 1.5-6.6, P = .002).
Drug classes most strongly associated with 1-year post-COVID dementia onset were antipsychotics (OR, 2.8, 95% CI, 1.7-4.4, P < .001) and mood stabilizers/anticonvulsants (OR, 2.4, 95% CI, 1.39-4.02, P = .001).
In a further exploratory analysis, the psychotropics valproic acid (multiple brands) and haloperidol (Haldol) had the largest association with post-COVID dementia.
Antidepressants as a class were not associated with post-COVID dementia, but the potential effects of two commonly prescribed antidepressants in older adults, mirtazapine (Remeron) and escitalopram (Lexapro), “warrant further investigation,” the researchers note.
Predictive risk marker?
“This research shows that psychotropic medications can be considered a predictive risk marker for post-COVID dementia. In patients taking psychotropic medications, COVID-19 could have accelerated progression of dementia after hospitalization,” lead author Yun Freudenberg-Hua, MD, the Feinstein Institutes, said in a news release.
It is unclear why psychotropic medications may raise the risk for dementia onset after COVID, the investigators note.
“It is intuitive that psychotropic medications indicate pre-existing neuropsychiatric conditions in which COVID-19 occurs. It is possible that psychotropic medications may potentiate the neurostructural changes that have been found in the brain of those who have recovered from COVID-19,” they write.
The sensitivity analysis in patients with documented neurologic and psychiatric diagnoses supports this interpretation.
COVID-19 may also accelerate the underlying brain disorders for which psychotropic medications were prescribed, leading to the greater incidence of post-COVID dementia, the researchers write.
“It is important to note that this study is in no way recommending people should stop taking antipsychotics but simply that clinicians need to factor in a patient’s medication history while considering post-COVID aftereffects,” Dr. Freudenberg-Hua said.
“Given that the number of patients with dementia is projected to triple in the next 30 years, these findings have significant public health implications,” Dr. Sinvani added.
She noted that “care partners and health care professionals” should look for early signs of dementia, such as forgetfulness and depressive symptoms, in their patients.
“Future studies must continue to evaluate these associations, which are key for potential future interventions to prevent dementia,” Dr. Sinvani said.
The study was funded by the National Institutes of Health. Dr. Freudenberg-Hua co-owns stock and stock options from Regeneron Pharmaceuticals. Dr. Sinvani has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM FRONTIERS IN MEDICINE
Cellulitis care costly from misdiagnosis, needless hospitalizations
BOSTON – The cost of care for the more than 14 million cases of cellulitis that occur each year in the United States is in the billions of dollars, but there are multiple opportunities, many involving dermatologists, to dramatically reduce these costs, according to an outline of strategies presented at the American Academy of Dermatology 2022 annual meeting in Boston.
“Cellulitis is misdiagnosed about one-third of the time, and that cost is very high,” reported Jennifer L. Adams, MD, assistant professor of dermatology, University of Nebraska, Omaha. She sees opportunities for dermatological consults to help weed through the many cellulitis mimickers, such as venous insufficiency or psoriasiform drug reactions, to prevent unnecessary admissions and ineffective therapy.
“There is a huge need for diagnostic accuracy as a means to deliver more cost-effective care,” Dr. Adams said.
Solving misdiagnosis is only part of the story. Costs of care are also ramped up by unnecessary hospitalizations. According to Dr. Adams, published criteria to triage emergency room patients with cellulitis to outpatient care are not always followed. In one review, 14% of admitted patients had met the criteria for outpatient treatment.
Cellulitis is a common skin infection that causes redness, swelling, and pain in the infected area, most often on the legs and feet.
Unnecessary hospitalizations for misdiagnosed cellulitis, which is associated with an average 4-day hospital stay, “range from $200 million to $500 million in avoidable direct healthcare costs,” Dr. Adams said.
Even for justifiable hospitalizations, there are still opportunities for cost savings. In one study, blood cultures were ordered in 73% of patients even though only 2% produced a finding relevant to care. According to Dr. Adams, most cellulitis cases are caused by the “usual suspects” – group A beta-hemolytic streptococcus, Streptococcus pneumoniae, and Staphylococcus aureus. The exceptions stand out by clinical criteria, such as known neutropenia, history of an animal bite, signs of Systemic Inflammatory Response Syndrome (SIRS), or a purulent appearance.
“Blood cultures are not cost-effective in uncomplicated cellulitis,” Dr. Adams said. She said there are numerous published algorithms to guide clinicians on decision-making in the management of soft tissue infections, including cellulitis, including a much-cited algorithm first published more than 15 years ago and updated in 2014.
Similarly, labs and imaging are commonly ordered with no strong likelihood that they will change management, she said. These types of decisions are also covered in published algorithms.
Strategies to prevent rehospitalization are another area where there is a large opportunity to reduce health care resources consumed by cellulitis. The rehospitalization rate at 30 days is approximately 10%, but many patients have recurrent episodes over years, according to Dr. Adams. The risk factors and the preventative measures have been well described.
“Scrupulous clinical care can reduce recurrence, and it is cost-effective,” said Dr. Adams, referring to control of edema, control of underlying conditions associated with increased risk, such as diabetes, and managing dry skin and erosions with topical agents or even moisturizers. Compression socks are a simple but effective tool, she added.
For patients with repeat episodes of cellulitis over years, Dr. Adams referred to a double-blind trial that associated a twice-daily dose of 250 mg penicillin with a 45% reduction in the risk of cellulitis recurrence over 1 year. At approximately $10 a month for this treatment, she said it is very cost-effective, although she acknowledged that recurrence rates of cellulitis climb back up when the penicillin is stopped.
“I think of this as a bridge while you work on addressing the venous insufficiency or other risk factors for cellulitis,” Dr. Adams said.
For reducing the costs of cellulitis, there is evidence that dermatologists can play a role. Dr. Adams cited a study that evaluated the impact of a dermatologist consultation for suspected cellulitis in the emergency room or within 24 hours of admission. Of 34 patients already prescribed antibiotics for presumed cellulitis, discontinuation was recommended in 82%. Of 39 admissions, pseudocellulitis was identified in 51%.
Extrapolating these data to national rates of cellulitis, there was an estimated savings of up to $200 million annually without any apparent increased risk of adverse outcomes, according to Dr. Adams.
When contacted about his experience, the senior investigator of that study, Arash Mostaghimi, MD, director of the Inpatient Dermatology Consult Service, Brigham and Women’s Hospital, Boston, largely agreed with the premise of Adam’s analysis. In particular, he said, avoiding misdiagnosis of cellulitis offers a major opportunity to lower costs while possibly improving care.
True of national practice and at the local level, “misdiagnosis of noninfectious inflammatory reactions such as cellulitis has substantial cost impacts,” Dr. Mostaghimi said in an interview. Based on evidence, the savings are derived directly from “unnecessary antibiotic exposure as well as inappropriate hospitalization.”
Following publication of his study, he became involved in addressing this issue at his institution.
“At Brigham and Women’s, we collaborated with colleagues in infectious disease and in the emergency department to create cellulitis protocols that identify patients at risk for misdiagnosis and facilitate early dermatology consultation for diagnostic confirmation,” he said.
Although there are algorithms to achieve this goal, he indicated that the expertise of dermatologists can quickly and efficiently differentiate inflammatory skin reactions and expedite appropriate care.
Dr. Adams and Dr. Mostaghimi have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
BOSTON – The cost of care for the more than 14 million cases of cellulitis that occur each year in the United States is in the billions of dollars, but there are multiple opportunities, many involving dermatologists, to dramatically reduce these costs, according to an outline of strategies presented at the American Academy of Dermatology 2022 annual meeting in Boston.
“Cellulitis is misdiagnosed about one-third of the time, and that cost is very high,” reported Jennifer L. Adams, MD, assistant professor of dermatology, University of Nebraska, Omaha. She sees opportunities for dermatological consults to help weed through the many cellulitis mimickers, such as venous insufficiency or psoriasiform drug reactions, to prevent unnecessary admissions and ineffective therapy.
“There is a huge need for diagnostic accuracy as a means to deliver more cost-effective care,” Dr. Adams said.
Solving misdiagnosis is only part of the story. Costs of care are also ramped up by unnecessary hospitalizations. According to Dr. Adams, published criteria to triage emergency room patients with cellulitis to outpatient care are not always followed. In one review, 14% of admitted patients had met the criteria for outpatient treatment.
Cellulitis is a common skin infection that causes redness, swelling, and pain in the infected area, most often on the legs and feet.
Unnecessary hospitalizations for misdiagnosed cellulitis, which is associated with an average 4-day hospital stay, “range from $200 million to $500 million in avoidable direct healthcare costs,” Dr. Adams said.
Even for justifiable hospitalizations, there are still opportunities for cost savings. In one study, blood cultures were ordered in 73% of patients even though only 2% produced a finding relevant to care. According to Dr. Adams, most cellulitis cases are caused by the “usual suspects” – group A beta-hemolytic streptococcus, Streptococcus pneumoniae, and Staphylococcus aureus. The exceptions stand out by clinical criteria, such as known neutropenia, history of an animal bite, signs of Systemic Inflammatory Response Syndrome (SIRS), or a purulent appearance.
“Blood cultures are not cost-effective in uncomplicated cellulitis,” Dr. Adams said. She said there are numerous published algorithms to guide clinicians on decision-making in the management of soft tissue infections, including cellulitis, including a much-cited algorithm first published more than 15 years ago and updated in 2014.
Similarly, labs and imaging are commonly ordered with no strong likelihood that they will change management, she said. These types of decisions are also covered in published algorithms.
Strategies to prevent rehospitalization are another area where there is a large opportunity to reduce health care resources consumed by cellulitis. The rehospitalization rate at 30 days is approximately 10%, but many patients have recurrent episodes over years, according to Dr. Adams. The risk factors and the preventative measures have been well described.
“Scrupulous clinical care can reduce recurrence, and it is cost-effective,” said Dr. Adams, referring to control of edema, control of underlying conditions associated with increased risk, such as diabetes, and managing dry skin and erosions with topical agents or even moisturizers. Compression socks are a simple but effective tool, she added.
For patients with repeat episodes of cellulitis over years, Dr. Adams referred to a double-blind trial that associated a twice-daily dose of 250 mg penicillin with a 45% reduction in the risk of cellulitis recurrence over 1 year. At approximately $10 a month for this treatment, she said it is very cost-effective, although she acknowledged that recurrence rates of cellulitis climb back up when the penicillin is stopped.
“I think of this as a bridge while you work on addressing the venous insufficiency or other risk factors for cellulitis,” Dr. Adams said.
For reducing the costs of cellulitis, there is evidence that dermatologists can play a role. Dr. Adams cited a study that evaluated the impact of a dermatologist consultation for suspected cellulitis in the emergency room or within 24 hours of admission. Of 34 patients already prescribed antibiotics for presumed cellulitis, discontinuation was recommended in 82%. Of 39 admissions, pseudocellulitis was identified in 51%.
Extrapolating these data to national rates of cellulitis, there was an estimated savings of up to $200 million annually without any apparent increased risk of adverse outcomes, according to Dr. Adams.
When contacted about his experience, the senior investigator of that study, Arash Mostaghimi, MD, director of the Inpatient Dermatology Consult Service, Brigham and Women’s Hospital, Boston, largely agreed with the premise of Adam’s analysis. In particular, he said, avoiding misdiagnosis of cellulitis offers a major opportunity to lower costs while possibly improving care.
True of national practice and at the local level, “misdiagnosis of noninfectious inflammatory reactions such as cellulitis has substantial cost impacts,” Dr. Mostaghimi said in an interview. Based on evidence, the savings are derived directly from “unnecessary antibiotic exposure as well as inappropriate hospitalization.”
Following publication of his study, he became involved in addressing this issue at his institution.
“At Brigham and Women’s, we collaborated with colleagues in infectious disease and in the emergency department to create cellulitis protocols that identify patients at risk for misdiagnosis and facilitate early dermatology consultation for diagnostic confirmation,” he said.
Although there are algorithms to achieve this goal, he indicated that the expertise of dermatologists can quickly and efficiently differentiate inflammatory skin reactions and expedite appropriate care.
Dr. Adams and Dr. Mostaghimi have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
BOSTON – The cost of care for the more than 14 million cases of cellulitis that occur each year in the United States is in the billions of dollars, but there are multiple opportunities, many involving dermatologists, to dramatically reduce these costs, according to an outline of strategies presented at the American Academy of Dermatology 2022 annual meeting in Boston.
“Cellulitis is misdiagnosed about one-third of the time, and that cost is very high,” reported Jennifer L. Adams, MD, assistant professor of dermatology, University of Nebraska, Omaha. She sees opportunities for dermatological consults to help weed through the many cellulitis mimickers, such as venous insufficiency or psoriasiform drug reactions, to prevent unnecessary admissions and ineffective therapy.
“There is a huge need for diagnostic accuracy as a means to deliver more cost-effective care,” Dr. Adams said.
Solving misdiagnosis is only part of the story. Costs of care are also ramped up by unnecessary hospitalizations. According to Dr. Adams, published criteria to triage emergency room patients with cellulitis to outpatient care are not always followed. In one review, 14% of admitted patients had met the criteria for outpatient treatment.
Cellulitis is a common skin infection that causes redness, swelling, and pain in the infected area, most often on the legs and feet.
Unnecessary hospitalizations for misdiagnosed cellulitis, which is associated with an average 4-day hospital stay, “range from $200 million to $500 million in avoidable direct healthcare costs,” Dr. Adams said.
Even for justifiable hospitalizations, there are still opportunities for cost savings. In one study, blood cultures were ordered in 73% of patients even though only 2% produced a finding relevant to care. According to Dr. Adams, most cellulitis cases are caused by the “usual suspects” – group A beta-hemolytic streptococcus, Streptococcus pneumoniae, and Staphylococcus aureus. The exceptions stand out by clinical criteria, such as known neutropenia, history of an animal bite, signs of Systemic Inflammatory Response Syndrome (SIRS), or a purulent appearance.
“Blood cultures are not cost-effective in uncomplicated cellulitis,” Dr. Adams said. She said there are numerous published algorithms to guide clinicians on decision-making in the management of soft tissue infections, including cellulitis, including a much-cited algorithm first published more than 15 years ago and updated in 2014.
Similarly, labs and imaging are commonly ordered with no strong likelihood that they will change management, she said. These types of decisions are also covered in published algorithms.
Strategies to prevent rehospitalization are another area where there is a large opportunity to reduce health care resources consumed by cellulitis. The rehospitalization rate at 30 days is approximately 10%, but many patients have recurrent episodes over years, according to Dr. Adams. The risk factors and the preventative measures have been well described.
“Scrupulous clinical care can reduce recurrence, and it is cost-effective,” said Dr. Adams, referring to control of edema, control of underlying conditions associated with increased risk, such as diabetes, and managing dry skin and erosions with topical agents or even moisturizers. Compression socks are a simple but effective tool, she added.
For patients with repeat episodes of cellulitis over years, Dr. Adams referred to a double-blind trial that associated a twice-daily dose of 250 mg penicillin with a 45% reduction in the risk of cellulitis recurrence over 1 year. At approximately $10 a month for this treatment, she said it is very cost-effective, although she acknowledged that recurrence rates of cellulitis climb back up when the penicillin is stopped.
“I think of this as a bridge while you work on addressing the venous insufficiency or other risk factors for cellulitis,” Dr. Adams said.
For reducing the costs of cellulitis, there is evidence that dermatologists can play a role. Dr. Adams cited a study that evaluated the impact of a dermatologist consultation for suspected cellulitis in the emergency room or within 24 hours of admission. Of 34 patients already prescribed antibiotics for presumed cellulitis, discontinuation was recommended in 82%. Of 39 admissions, pseudocellulitis was identified in 51%.
Extrapolating these data to national rates of cellulitis, there was an estimated savings of up to $200 million annually without any apparent increased risk of adverse outcomes, according to Dr. Adams.
When contacted about his experience, the senior investigator of that study, Arash Mostaghimi, MD, director of the Inpatient Dermatology Consult Service, Brigham and Women’s Hospital, Boston, largely agreed with the premise of Adam’s analysis. In particular, he said, avoiding misdiagnosis of cellulitis offers a major opportunity to lower costs while possibly improving care.
True of national practice and at the local level, “misdiagnosis of noninfectious inflammatory reactions such as cellulitis has substantial cost impacts,” Dr. Mostaghimi said in an interview. Based on evidence, the savings are derived directly from “unnecessary antibiotic exposure as well as inappropriate hospitalization.”
Following publication of his study, he became involved in addressing this issue at his institution.
“At Brigham and Women’s, we collaborated with colleagues in infectious disease and in the emergency department to create cellulitis protocols that identify patients at risk for misdiagnosis and facilitate early dermatology consultation for diagnostic confirmation,” he said.
Although there are algorithms to achieve this goal, he indicated that the expertise of dermatologists can quickly and efficiently differentiate inflammatory skin reactions and expedite appropriate care.
Dr. Adams and Dr. Mostaghimi have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT AAD 2022
Global registry tracks COVID-19 outcomes in atopic dermatitis patients
BOSTON – , results from a global registry demonstrated.
Moreover, combination systemic treatment, especially those that included systemic corticosteroids, was associated with the highest risk of COVID-19–related hospitalization.
“Patients with inflammatory skin diseases such as AD may be at higher risk of COVID-19,” Annelie H. Musters, MD, said during a late-breaking abstract session at the annual meeting of the American Academy of Dermatology. “Another factor to consider is that AD patients are often treated with systemic immunomodulatory therapy, including systemic corticosteroids and nonsteroidal immunosuppressants such as methotrexate, cyclosporin, biologics, and Janus kinase inhibitors. Different mechanisms of action and levels of immunosuppression may impart variable risks of serious infections.”
On the other hand, some degree of immunomodulation may have beneficial effects on the course of COVID-19 in AD patients, said Dr. Musters, of the department of dermatology at Academic Medical Center, University of Amsterdam. Targeting of specific immune pathways could reduce the development of a hyperinflammatory state in severe COVID-19. Dual blockade of interleukin (IL)-4 and IL-13 with dupilumab may have a protective effect in the context of COVID-19 infection, because expression of Th2 cytokines, including IL-4 and IL-13, may be increased during COVID-19.
“At the start of the pandemic, many of us were faced with important questions, like do systemic immunomodulatory treatments influence outcomes of COVID-19 in patients with AD?” she said. “Do patients on dupilumab or other novel systemics fare better than those on conventional systemic treatment?”
To answer these questions, she and her colleagues launched a web-based registry in April 2020 to investigate COVID-19 outcomes in patients with AD treated with or without systemic immunomodulatory treatments. For the registry, known as Surveillance Epidemiology of Coronavirus Under Research Exclusion for Atopic Dermatitis (SECURE-AD), clinicians in 27 countries used a web-based form to enter anonymized data after patients had fully recovered from COVID-19. Eligibility criteria included having proven or highly suspected COVID-19, and there were no restrictions on age nor the type of AD treatment they were receiving.
Dr. Musters reported results from 442 patients who were recruited between April 2, 2020, and Oct. 31, 2021. Their mean age was 35.6 years, their median body mass index was 23.7 kg/m2, and there was an even sex distribution. Most patients were White and were recruited from Italy. Of the 442 patients, 216 (48.8%) received dupilumab monotherapy, 131 (29.6%) received topical treatments, and 14 (3.16%) received combination systemic treatments, including systemic corticosteroids. About 12% presented to the emergency department and 6% were hospitalized. Of those hospitalized, 2% required intensive care and/or ventilation, and no deaths have occurred in the registry to date.
By treatment group, hospitalization rates were highest among those on combination treatments (35.7%), followed by systemic corticosteroids (14.3%), topical treatments only (9.9%), other conventional systemics (3.6%), methotrexate (3.3%), and dupilumab (2.3%).
To further explore the differences between hospitalization rates in treatment groups, the researchers performed a multivariable logistic regression analysis, adjusted for age, sex, ethnicity, and comorbidity score. Compared with those who received dupilumab, the adjusted odds ratios (ORs) for hospitalization were highest among those who received topical treatments (OR, 4.95), followed by those who received systemic corticosteroids (OR, 2.81), and those who received other conventional systemic treatments (OR, 2.36).
Dr. Musters and colleagues also found that compared with patients on nonsteroidal immunosuppressive therapy, patients on combination systemic therapy had a significantly higher odds of hospitalization, specifically an OR of 45.75 for those on combination treatment including corticosteroids, an OR of 37.57 for those on combination treatment not including steroids, and an OR of 1.87 for those on systemic corticosteroids as monotherapy.
“Overall, the risk of COVID-19 complications appears to be low in patients with AD, even when treated with systemic immunomodulatory agents,” Dr. Musters concluded. “Dupilumab monotherapy was associated with lower odds of hospitalizations compared with other therapies. Moreover, combination systemic treatment, especially combinations including systemic corticosteroids, was associated with the highest risk of severe COVID-19.”
She added that other population-based study designs are more suitable to answer other important questions, such as whether the overall risk of COVID-19 in patients with AD is higher or lower compared to healthy controls.
Amy S. Paller, MD, professor and chair of the department of dermatology at Northwestern University, Chicago, who was asked to comment on the study, characterized the results as reassuring. In this patient population, “we expected that dupilumab would not cause any problems,” she said. “We wouldn’t necessarily expect it to [confer] a benefit, but I think it’s because the patients who need a systemic medication are going on something that’s very targeted (dupilumab) rather than something that has a broader immunosuppressing function. It was interesting but not surprising that those on systemic steroids had more of a problem. Get them on something that’s very targeted if you can and don’t suppress the immune systems that might be handling COVID-19.”
Dr. Musters reported having no disclosures. Dr. Paller disclosed that she is consultant to and/or an investigator for many pharmaceutical companies.
BOSTON – , results from a global registry demonstrated.
Moreover, combination systemic treatment, especially those that included systemic corticosteroids, was associated with the highest risk of COVID-19–related hospitalization.
“Patients with inflammatory skin diseases such as AD may be at higher risk of COVID-19,” Annelie H. Musters, MD, said during a late-breaking abstract session at the annual meeting of the American Academy of Dermatology. “Another factor to consider is that AD patients are often treated with systemic immunomodulatory therapy, including systemic corticosteroids and nonsteroidal immunosuppressants such as methotrexate, cyclosporin, biologics, and Janus kinase inhibitors. Different mechanisms of action and levels of immunosuppression may impart variable risks of serious infections.”
On the other hand, some degree of immunomodulation may have beneficial effects on the course of COVID-19 in AD patients, said Dr. Musters, of the department of dermatology at Academic Medical Center, University of Amsterdam. Targeting of specific immune pathways could reduce the development of a hyperinflammatory state in severe COVID-19. Dual blockade of interleukin (IL)-4 and IL-13 with dupilumab may have a protective effect in the context of COVID-19 infection, because expression of Th2 cytokines, including IL-4 and IL-13, may be increased during COVID-19.
“At the start of the pandemic, many of us were faced with important questions, like do systemic immunomodulatory treatments influence outcomes of COVID-19 in patients with AD?” she said. “Do patients on dupilumab or other novel systemics fare better than those on conventional systemic treatment?”
To answer these questions, she and her colleagues launched a web-based registry in April 2020 to investigate COVID-19 outcomes in patients with AD treated with or without systemic immunomodulatory treatments. For the registry, known as Surveillance Epidemiology of Coronavirus Under Research Exclusion for Atopic Dermatitis (SECURE-AD), clinicians in 27 countries used a web-based form to enter anonymized data after patients had fully recovered from COVID-19. Eligibility criteria included having proven or highly suspected COVID-19, and there were no restrictions on age nor the type of AD treatment they were receiving.
Dr. Musters reported results from 442 patients who were recruited between April 2, 2020, and Oct. 31, 2021. Their mean age was 35.6 years, their median body mass index was 23.7 kg/m2, and there was an even sex distribution. Most patients were White and were recruited from Italy. Of the 442 patients, 216 (48.8%) received dupilumab monotherapy, 131 (29.6%) received topical treatments, and 14 (3.16%) received combination systemic treatments, including systemic corticosteroids. About 12% presented to the emergency department and 6% were hospitalized. Of those hospitalized, 2% required intensive care and/or ventilation, and no deaths have occurred in the registry to date.
By treatment group, hospitalization rates were highest among those on combination treatments (35.7%), followed by systemic corticosteroids (14.3%), topical treatments only (9.9%), other conventional systemics (3.6%), methotrexate (3.3%), and dupilumab (2.3%).
To further explore the differences between hospitalization rates in treatment groups, the researchers performed a multivariable logistic regression analysis, adjusted for age, sex, ethnicity, and comorbidity score. Compared with those who received dupilumab, the adjusted odds ratios (ORs) for hospitalization were highest among those who received topical treatments (OR, 4.95), followed by those who received systemic corticosteroids (OR, 2.81), and those who received other conventional systemic treatments (OR, 2.36).
Dr. Musters and colleagues also found that compared with patients on nonsteroidal immunosuppressive therapy, patients on combination systemic therapy had a significantly higher odds of hospitalization, specifically an OR of 45.75 for those on combination treatment including corticosteroids, an OR of 37.57 for those on combination treatment not including steroids, and an OR of 1.87 for those on systemic corticosteroids as monotherapy.
“Overall, the risk of COVID-19 complications appears to be low in patients with AD, even when treated with systemic immunomodulatory agents,” Dr. Musters concluded. “Dupilumab monotherapy was associated with lower odds of hospitalizations compared with other therapies. Moreover, combination systemic treatment, especially combinations including systemic corticosteroids, was associated with the highest risk of severe COVID-19.”
She added that other population-based study designs are more suitable to answer other important questions, such as whether the overall risk of COVID-19 in patients with AD is higher or lower compared to healthy controls.
Amy S. Paller, MD, professor and chair of the department of dermatology at Northwestern University, Chicago, who was asked to comment on the study, characterized the results as reassuring. In this patient population, “we expected that dupilumab would not cause any problems,” she said. “We wouldn’t necessarily expect it to [confer] a benefit, but I think it’s because the patients who need a systemic medication are going on something that’s very targeted (dupilumab) rather than something that has a broader immunosuppressing function. It was interesting but not surprising that those on systemic steroids had more of a problem. Get them on something that’s very targeted if you can and don’t suppress the immune systems that might be handling COVID-19.”
Dr. Musters reported having no disclosures. Dr. Paller disclosed that she is consultant to and/or an investigator for many pharmaceutical companies.
BOSTON – , results from a global registry demonstrated.
Moreover, combination systemic treatment, especially those that included systemic corticosteroids, was associated with the highest risk of COVID-19–related hospitalization.
“Patients with inflammatory skin diseases such as AD may be at higher risk of COVID-19,” Annelie H. Musters, MD, said during a late-breaking abstract session at the annual meeting of the American Academy of Dermatology. “Another factor to consider is that AD patients are often treated with systemic immunomodulatory therapy, including systemic corticosteroids and nonsteroidal immunosuppressants such as methotrexate, cyclosporin, biologics, and Janus kinase inhibitors. Different mechanisms of action and levels of immunosuppression may impart variable risks of serious infections.”
On the other hand, some degree of immunomodulation may have beneficial effects on the course of COVID-19 in AD patients, said Dr. Musters, of the department of dermatology at Academic Medical Center, University of Amsterdam. Targeting of specific immune pathways could reduce the development of a hyperinflammatory state in severe COVID-19. Dual blockade of interleukin (IL)-4 and IL-13 with dupilumab may have a protective effect in the context of COVID-19 infection, because expression of Th2 cytokines, including IL-4 and IL-13, may be increased during COVID-19.
“At the start of the pandemic, many of us were faced with important questions, like do systemic immunomodulatory treatments influence outcomes of COVID-19 in patients with AD?” she said. “Do patients on dupilumab or other novel systemics fare better than those on conventional systemic treatment?”
To answer these questions, she and her colleagues launched a web-based registry in April 2020 to investigate COVID-19 outcomes in patients with AD treated with or without systemic immunomodulatory treatments. For the registry, known as Surveillance Epidemiology of Coronavirus Under Research Exclusion for Atopic Dermatitis (SECURE-AD), clinicians in 27 countries used a web-based form to enter anonymized data after patients had fully recovered from COVID-19. Eligibility criteria included having proven or highly suspected COVID-19, and there were no restrictions on age nor the type of AD treatment they were receiving.
Dr. Musters reported results from 442 patients who were recruited between April 2, 2020, and Oct. 31, 2021. Their mean age was 35.6 years, their median body mass index was 23.7 kg/m2, and there was an even sex distribution. Most patients were White and were recruited from Italy. Of the 442 patients, 216 (48.8%) received dupilumab monotherapy, 131 (29.6%) received topical treatments, and 14 (3.16%) received combination systemic treatments, including systemic corticosteroids. About 12% presented to the emergency department and 6% were hospitalized. Of those hospitalized, 2% required intensive care and/or ventilation, and no deaths have occurred in the registry to date.
By treatment group, hospitalization rates were highest among those on combination treatments (35.7%), followed by systemic corticosteroids (14.3%), topical treatments only (9.9%), other conventional systemics (3.6%), methotrexate (3.3%), and dupilumab (2.3%).
To further explore the differences between hospitalization rates in treatment groups, the researchers performed a multivariable logistic regression analysis, adjusted for age, sex, ethnicity, and comorbidity score. Compared with those who received dupilumab, the adjusted odds ratios (ORs) for hospitalization were highest among those who received topical treatments (OR, 4.95), followed by those who received systemic corticosteroids (OR, 2.81), and those who received other conventional systemic treatments (OR, 2.36).
Dr. Musters and colleagues also found that compared with patients on nonsteroidal immunosuppressive therapy, patients on combination systemic therapy had a significantly higher odds of hospitalization, specifically an OR of 45.75 for those on combination treatment including corticosteroids, an OR of 37.57 for those on combination treatment not including steroids, and an OR of 1.87 for those on systemic corticosteroids as monotherapy.
“Overall, the risk of COVID-19 complications appears to be low in patients with AD, even when treated with systemic immunomodulatory agents,” Dr. Musters concluded. “Dupilumab monotherapy was associated with lower odds of hospitalizations compared with other therapies. Moreover, combination systemic treatment, especially combinations including systemic corticosteroids, was associated with the highest risk of severe COVID-19.”
She added that other population-based study designs are more suitable to answer other important questions, such as whether the overall risk of COVID-19 in patients with AD is higher or lower compared to healthy controls.
Amy S. Paller, MD, professor and chair of the department of dermatology at Northwestern University, Chicago, who was asked to comment on the study, characterized the results as reassuring. In this patient population, “we expected that dupilumab would not cause any problems,” she said. “We wouldn’t necessarily expect it to [confer] a benefit, but I think it’s because the patients who need a systemic medication are going on something that’s very targeted (dupilumab) rather than something that has a broader immunosuppressing function. It was interesting but not surprising that those on systemic steroids had more of a problem. Get them on something that’s very targeted if you can and don’t suppress the immune systems that might be handling COVID-19.”
Dr. Musters reported having no disclosures. Dr. Paller disclosed that she is consultant to and/or an investigator for many pharmaceutical companies.
AT AAD 22
Children and COVID: The long goodbye continues
COVID-19 continues to be a diminishing issue for U.S. children, as the number of new cases declined for the ninth consecutive week, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.
the AAP and CHA said in their weekly COVID report. The most recently infected children brought the total number of COVID-19 cases to just over 12.8 million since the pandemic began.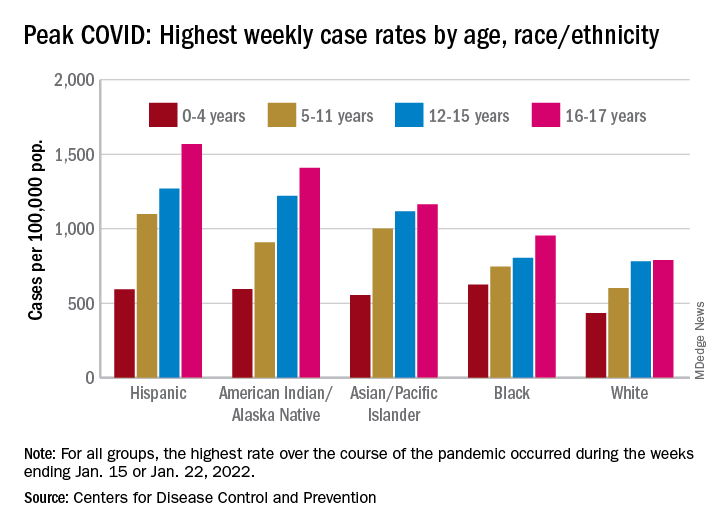
Other measures of COVID occurrence in children, such as hospital admissions and emergency department visits, also followed recent downward trends, although the sizes of the declines are beginning to decrease. Admissions dropped by 13.3% during the week ending March 26, but that followed declines of 25%, 20%, 26.5% and 24.4% for the 4 previous weeks, data from the Centers for Disease Control and Prevention show.
The slowdown in ED visits started a couple of weeks earlier, but the decline is still ongoing. As of March 25, ED visits with a confirmed COVID diagnosis represented just 0.4% of all visits for children aged 0-11 years, down from 1.1% on Feb. 25 and a peak of 14.3% on Jan. 15. For children aged 12-15, the latest figure is just 0.2%, compared with 0.5% on Feb. 25 and a peak of 14.3% on Jan. 9, the CDC reported on its COVID Data Tracker.
Although he was speaking of the nation as a whole and not specifically of children, Anthony Fauci, MD, the director of the National Institute of Allergy and Infectious Diseases, recently told the Washington Post that, “unless something changes dramatically,” another major surge isn’t on the horizon.
That sentiment, however, was not entirely shared by Moderna’s chief medical officer, Paul Burton, MD, PhD. In an interview with WebMD, he said that another COVID wave is inevitable and that it’s too soon to dismantle the vaccine infrastructure: “We’ve come so far. We’ve put so much into this to now take our foot off the gas. I think it would be a mistake for public health worldwide.”
Disparities during the Omicron surge
As the country puts Omicron in its rear view mirror, a quick look back at the CDC data shows some differences in how children were affected. At the surge’s peak in early to mid-January, Hispanic children were the most likely to get COVID-19, with incidence highest in the older groups. (See graph.)
At their peak week of Jan. 2-8, Hispanic children aged 16-17 years had a COVID rate of 1,568 cases per 100,000 population, versus 790 per 100,000 for White children, whose peak occurred a week later, from Jan. 9 to 15. Hispanic children aged 5-11 (1,098 per 100,000) and 12-15 (1,269 per 100,000) also had the highest recorded rates of the largest racial/ethnic groups, while Black children had the highest one-week rate, 625 per 100,000, among the 0- to 4-year-olds, according to the CDC.
COVID-19 continues to be a diminishing issue for U.S. children, as the number of new cases declined for the ninth consecutive week, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.
the AAP and CHA said in their weekly COVID report. The most recently infected children brought the total number of COVID-19 cases to just over 12.8 million since the pandemic began.
Other measures of COVID occurrence in children, such as hospital admissions and emergency department visits, also followed recent downward trends, although the sizes of the declines are beginning to decrease. Admissions dropped by 13.3% during the week ending March 26, but that followed declines of 25%, 20%, 26.5% and 24.4% for the 4 previous weeks, data from the Centers for Disease Control and Prevention show.
The slowdown in ED visits started a couple of weeks earlier, but the decline is still ongoing. As of March 25, ED visits with a confirmed COVID diagnosis represented just 0.4% of all visits for children aged 0-11 years, down from 1.1% on Feb. 25 and a peak of 14.3% on Jan. 15. For children aged 12-15, the latest figure is just 0.2%, compared with 0.5% on Feb. 25 and a peak of 14.3% on Jan. 9, the CDC reported on its COVID Data Tracker.
Although he was speaking of the nation as a whole and not specifically of children, Anthony Fauci, MD, the director of the National Institute of Allergy and Infectious Diseases, recently told the Washington Post that, “unless something changes dramatically,” another major surge isn’t on the horizon.
That sentiment, however, was not entirely shared by Moderna’s chief medical officer, Paul Burton, MD, PhD. In an interview with WebMD, he said that another COVID wave is inevitable and that it’s too soon to dismantle the vaccine infrastructure: “We’ve come so far. We’ve put so much into this to now take our foot off the gas. I think it would be a mistake for public health worldwide.”
Disparities during the Omicron surge
As the country puts Omicron in its rear view mirror, a quick look back at the CDC data shows some differences in how children were affected. At the surge’s peak in early to mid-January, Hispanic children were the most likely to get COVID-19, with incidence highest in the older groups. (See graph.)
At their peak week of Jan. 2-8, Hispanic children aged 16-17 years had a COVID rate of 1,568 cases per 100,000 population, versus 790 per 100,000 for White children, whose peak occurred a week later, from Jan. 9 to 15. Hispanic children aged 5-11 (1,098 per 100,000) and 12-15 (1,269 per 100,000) also had the highest recorded rates of the largest racial/ethnic groups, while Black children had the highest one-week rate, 625 per 100,000, among the 0- to 4-year-olds, according to the CDC.
COVID-19 continues to be a diminishing issue for U.S. children, as the number of new cases declined for the ninth consecutive week, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.
the AAP and CHA said in their weekly COVID report. The most recently infected children brought the total number of COVID-19 cases to just over 12.8 million since the pandemic began.
Other measures of COVID occurrence in children, such as hospital admissions and emergency department visits, also followed recent downward trends, although the sizes of the declines are beginning to decrease. Admissions dropped by 13.3% during the week ending March 26, but that followed declines of 25%, 20%, 26.5% and 24.4% for the 4 previous weeks, data from the Centers for Disease Control and Prevention show.
The slowdown in ED visits started a couple of weeks earlier, but the decline is still ongoing. As of March 25, ED visits with a confirmed COVID diagnosis represented just 0.4% of all visits for children aged 0-11 years, down from 1.1% on Feb. 25 and a peak of 14.3% on Jan. 15. For children aged 12-15, the latest figure is just 0.2%, compared with 0.5% on Feb. 25 and a peak of 14.3% on Jan. 9, the CDC reported on its COVID Data Tracker.
Although he was speaking of the nation as a whole and not specifically of children, Anthony Fauci, MD, the director of the National Institute of Allergy and Infectious Diseases, recently told the Washington Post that, “unless something changes dramatically,” another major surge isn’t on the horizon.
That sentiment, however, was not entirely shared by Moderna’s chief medical officer, Paul Burton, MD, PhD. In an interview with WebMD, he said that another COVID wave is inevitable and that it’s too soon to dismantle the vaccine infrastructure: “We’ve come so far. We’ve put so much into this to now take our foot off the gas. I think it would be a mistake for public health worldwide.”
Disparities during the Omicron surge
As the country puts Omicron in its rear view mirror, a quick look back at the CDC data shows some differences in how children were affected. At the surge’s peak in early to mid-January, Hispanic children were the most likely to get COVID-19, with incidence highest in the older groups. (See graph.)
At their peak week of Jan. 2-8, Hispanic children aged 16-17 years had a COVID rate of 1,568 cases per 100,000 population, versus 790 per 100,000 for White children, whose peak occurred a week later, from Jan. 9 to 15. Hispanic children aged 5-11 (1,098 per 100,000) and 12-15 (1,269 per 100,000) also had the highest recorded rates of the largest racial/ethnic groups, while Black children had the highest one-week rate, 625 per 100,000, among the 0- to 4-year-olds, according to the CDC.
FDA approves HIV injectable Cabenuva initiation without oral lead-in
Initiating treatment may become easier for adults living with HIV. a combination injectable, without a lead-in period of oral tablets, according to a press release from Janssen Pharmaceuticals.
Cabenuva combines rilpivirine (Janssen) and cabotegravir (ViiV Healthcare). The change offers patients and clinicians an option for a streamlined entry to treatment without the burden of daily pill taking, according to the release.
Cabenuva injections may be given as few as six times a year to manage HIV, according to Janssen. HIV patients with viral suppression previously had to complete an oral treatment regimen before starting monthly or bimonthly injections.
The injectable combination of cabotegravir, an HIV-1 integrase strand transfer inhibitor, and rilpivirine, an HIV-1 nonnucleoside reverse transcriptase inhibitor, is currently indicated as a complete treatment regimen to replace the current antiretroviral regimen for adults with HIV who are virologically suppressed,” according to the press release.
“Janssen and ViiV are exploring the future possibility of an ultra–long-acting version of Cabenuva, which could reduce the frequency of injections even further, according to the press release.
Access may improve, but barriers persist
“Despite advances in HIV care, many barriers remain, particularly for the most vulnerable populations,” Lina Rosengren-Hovee, MD, of the University of North Carolina at Chapel Hill, said in an interview.
“Care engagement has improved with the use of bridge counselors, rapid ART [antiretroviral therapy] initiation policies, and contact tracing,” she said. “Similarly, increasing access to multiple modalities of HIV treatment is critical to increase engagement in care.
“For patients, removing the oral lead-in primarily reduces the number of clinical visits to start injectable ART,” Dr. Rosengren-Hovee added. “It may also remove adherence barriers for patients who have difficulty taking a daily oral medication.”
But Dr. Rosengren-Hovee (who has no financial connection to the manufacturers) pointed out that access to Cabenuva may not be seamless. “Unless the medication is stocked in clinics, patients are not likely to receive their first injection during the initial visit. Labs are also required prior to initiation to ensure there is no contraindication to the medication, such as viral resistance to one of its components. Cost and insurance coverage are also likely to remain major obstacles.”
Dr. Rosengren-Hovee has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Initiating treatment may become easier for adults living with HIV. a combination injectable, without a lead-in period of oral tablets, according to a press release from Janssen Pharmaceuticals.
Cabenuva combines rilpivirine (Janssen) and cabotegravir (ViiV Healthcare). The change offers patients and clinicians an option for a streamlined entry to treatment without the burden of daily pill taking, according to the release.
Cabenuva injections may be given as few as six times a year to manage HIV, according to Janssen. HIV patients with viral suppression previously had to complete an oral treatment regimen before starting monthly or bimonthly injections.
The injectable combination of cabotegravir, an HIV-1 integrase strand transfer inhibitor, and rilpivirine, an HIV-1 nonnucleoside reverse transcriptase inhibitor, is currently indicated as a complete treatment regimen to replace the current antiretroviral regimen for adults with HIV who are virologically suppressed,” according to the press release.
“Janssen and ViiV are exploring the future possibility of an ultra–long-acting version of Cabenuva, which could reduce the frequency of injections even further, according to the press release.
Access may improve, but barriers persist
“Despite advances in HIV care, many barriers remain, particularly for the most vulnerable populations,” Lina Rosengren-Hovee, MD, of the University of North Carolina at Chapel Hill, said in an interview.
“Care engagement has improved with the use of bridge counselors, rapid ART [antiretroviral therapy] initiation policies, and contact tracing,” she said. “Similarly, increasing access to multiple modalities of HIV treatment is critical to increase engagement in care.
“For patients, removing the oral lead-in primarily reduces the number of clinical visits to start injectable ART,” Dr. Rosengren-Hovee added. “It may also remove adherence barriers for patients who have difficulty taking a daily oral medication.”
But Dr. Rosengren-Hovee (who has no financial connection to the manufacturers) pointed out that access to Cabenuva may not be seamless. “Unless the medication is stocked in clinics, patients are not likely to receive their first injection during the initial visit. Labs are also required prior to initiation to ensure there is no contraindication to the medication, such as viral resistance to one of its components. Cost and insurance coverage are also likely to remain major obstacles.”
Dr. Rosengren-Hovee has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Initiating treatment may become easier for adults living with HIV. a combination injectable, without a lead-in period of oral tablets, according to a press release from Janssen Pharmaceuticals.
Cabenuva combines rilpivirine (Janssen) and cabotegravir (ViiV Healthcare). The change offers patients and clinicians an option for a streamlined entry to treatment without the burden of daily pill taking, according to the release.
Cabenuva injections may be given as few as six times a year to manage HIV, according to Janssen. HIV patients with viral suppression previously had to complete an oral treatment regimen before starting monthly or bimonthly injections.
The injectable combination of cabotegravir, an HIV-1 integrase strand transfer inhibitor, and rilpivirine, an HIV-1 nonnucleoside reverse transcriptase inhibitor, is currently indicated as a complete treatment regimen to replace the current antiretroviral regimen for adults with HIV who are virologically suppressed,” according to the press release.
“Janssen and ViiV are exploring the future possibility of an ultra–long-acting version of Cabenuva, which could reduce the frequency of injections even further, according to the press release.
Access may improve, but barriers persist
“Despite advances in HIV care, many barriers remain, particularly for the most vulnerable populations,” Lina Rosengren-Hovee, MD, of the University of North Carolina at Chapel Hill, said in an interview.
“Care engagement has improved with the use of bridge counselors, rapid ART [antiretroviral therapy] initiation policies, and contact tracing,” she said. “Similarly, increasing access to multiple modalities of HIV treatment is critical to increase engagement in care.
“For patients, removing the oral lead-in primarily reduces the number of clinical visits to start injectable ART,” Dr. Rosengren-Hovee added. “It may also remove adherence barriers for patients who have difficulty taking a daily oral medication.”
But Dr. Rosengren-Hovee (who has no financial connection to the manufacturers) pointed out that access to Cabenuva may not be seamless. “Unless the medication is stocked in clinics, patients are not likely to receive their first injection during the initial visit. Labs are also required prior to initiation to ensure there is no contraindication to the medication, such as viral resistance to one of its components. Cost and insurance coverage are also likely to remain major obstacles.”
Dr. Rosengren-Hovee has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Tick-borne Heartland virus circulating in U.S., researchers say
published in Emerging Infectious Diseases.
People can get the virus after being bitten by an infected tick, which can lead to hospitalization and death. The virus has also been found among deer and other wild mammals.
“Heartland is an emerging infectious disease that is not well understood,” Gonzalo Vazquez-Prokopec, PhD, the senior study author and an expert in vector-borne diseases at Emory University, Atlanta, said in a statement.
“We’re trying to get ahead of this virus by learning everything that we can about it before it potentially becomes a bigger problem,” he said.
Researchers at Emory and the University of Georgia analyzed virus samples from nearly 10,000 ticks collected in central Georgia. They found that about 1 out of every 2,000 specimens had the Heartland virus, including the adult and nymph stages.
The virus, which was first identified in Missouri in 2009, has been documented in several states across the Southeast and Midwest. There have been more than 50 cases in people from 11 states, according to the Centers for Disease Control and Prevention, with most cases requiring hospitalization. Most people diagnosed with the disease became sick from May to September, the CDC reported. Symptoms can be a high fever, fatigue, diarrhea, muscle pain, and low counts of white blood cells and platelets. It can take up to 2 weeks for symptoms to appear after a bite from an infected tick.
There are no vaccines or medications to prevent or treat the Heartland virus, according to the CDC. Doctors may be able to provide medications to improve symptoms. Overall, though, experts recommend that people avoid tick bites as much as possible, particularly during “high tick season” between April and September.
“You should be thinking about them almost any time of the year. It’s something that should be on everybody’s mind,” Jonathan Larson, PhD, an extension entomologist at the University of Kentucky, Lexington, told USA Today.
The CDC recognizes 18 tick-borne diseases in the United States, including Lyme disease, which has become the most common vector-borne disease in the country. The black-legged tick, also known as the deer tick, typically transmits the bacteria that causes Lyme disease.
But researchers are still studying how the Heartland virus spreads. In the latest study, they found the virus in the lone star tick, which is named for a distinctive white spot on its back and is the most common tick in Georgia. The tick is also widely distributed in wooded areas across the Southeast, Midwest, and Eastern United States.
The research team will now collect ticks across Georgia for testing to better understand what could raise the risk of getting the Heartland virus.
“We want to start filling in the huge gaps of knowledge of the transmission cycle for Heartland virus,” Dr. Vazquez-Prokopec said. “We need to better understand the key actors that transmit the virus and any environmental factors that may help it to persist within different habitats.”
A version of this article first appeared on WebMD.com.
published in Emerging Infectious Diseases.
People can get the virus after being bitten by an infected tick, which can lead to hospitalization and death. The virus has also been found among deer and other wild mammals.
“Heartland is an emerging infectious disease that is not well understood,” Gonzalo Vazquez-Prokopec, PhD, the senior study author and an expert in vector-borne diseases at Emory University, Atlanta, said in a statement.
“We’re trying to get ahead of this virus by learning everything that we can about it before it potentially becomes a bigger problem,” he said.
Researchers at Emory and the University of Georgia analyzed virus samples from nearly 10,000 ticks collected in central Georgia. They found that about 1 out of every 2,000 specimens had the Heartland virus, including the adult and nymph stages.
The virus, which was first identified in Missouri in 2009, has been documented in several states across the Southeast and Midwest. There have been more than 50 cases in people from 11 states, according to the Centers for Disease Control and Prevention, with most cases requiring hospitalization. Most people diagnosed with the disease became sick from May to September, the CDC reported. Symptoms can be a high fever, fatigue, diarrhea, muscle pain, and low counts of white blood cells and platelets. It can take up to 2 weeks for symptoms to appear after a bite from an infected tick.
There are no vaccines or medications to prevent or treat the Heartland virus, according to the CDC. Doctors may be able to provide medications to improve symptoms. Overall, though, experts recommend that people avoid tick bites as much as possible, particularly during “high tick season” between April and September.
“You should be thinking about them almost any time of the year. It’s something that should be on everybody’s mind,” Jonathan Larson, PhD, an extension entomologist at the University of Kentucky, Lexington, told USA Today.
The CDC recognizes 18 tick-borne diseases in the United States, including Lyme disease, which has become the most common vector-borne disease in the country. The black-legged tick, also known as the deer tick, typically transmits the bacteria that causes Lyme disease.
But researchers are still studying how the Heartland virus spreads. In the latest study, they found the virus in the lone star tick, which is named for a distinctive white spot on its back and is the most common tick in Georgia. The tick is also widely distributed in wooded areas across the Southeast, Midwest, and Eastern United States.
The research team will now collect ticks across Georgia for testing to better understand what could raise the risk of getting the Heartland virus.
“We want to start filling in the huge gaps of knowledge of the transmission cycle for Heartland virus,” Dr. Vazquez-Prokopec said. “We need to better understand the key actors that transmit the virus and any environmental factors that may help it to persist within different habitats.”
A version of this article first appeared on WebMD.com.
published in Emerging Infectious Diseases.
People can get the virus after being bitten by an infected tick, which can lead to hospitalization and death. The virus has also been found among deer and other wild mammals.
“Heartland is an emerging infectious disease that is not well understood,” Gonzalo Vazquez-Prokopec, PhD, the senior study author and an expert in vector-borne diseases at Emory University, Atlanta, said in a statement.
“We’re trying to get ahead of this virus by learning everything that we can about it before it potentially becomes a bigger problem,” he said.
Researchers at Emory and the University of Georgia analyzed virus samples from nearly 10,000 ticks collected in central Georgia. They found that about 1 out of every 2,000 specimens had the Heartland virus, including the adult and nymph stages.
The virus, which was first identified in Missouri in 2009, has been documented in several states across the Southeast and Midwest. There have been more than 50 cases in people from 11 states, according to the Centers for Disease Control and Prevention, with most cases requiring hospitalization. Most people diagnosed with the disease became sick from May to September, the CDC reported. Symptoms can be a high fever, fatigue, diarrhea, muscle pain, and low counts of white blood cells and platelets. It can take up to 2 weeks for symptoms to appear after a bite from an infected tick.
There are no vaccines or medications to prevent or treat the Heartland virus, according to the CDC. Doctors may be able to provide medications to improve symptoms. Overall, though, experts recommend that people avoid tick bites as much as possible, particularly during “high tick season” between April and September.
“You should be thinking about them almost any time of the year. It’s something that should be on everybody’s mind,” Jonathan Larson, PhD, an extension entomologist at the University of Kentucky, Lexington, told USA Today.
The CDC recognizes 18 tick-borne diseases in the United States, including Lyme disease, which has become the most common vector-borne disease in the country. The black-legged tick, also known as the deer tick, typically transmits the bacteria that causes Lyme disease.
But researchers are still studying how the Heartland virus spreads. In the latest study, they found the virus in the lone star tick, which is named for a distinctive white spot on its back and is the most common tick in Georgia. The tick is also widely distributed in wooded areas across the Southeast, Midwest, and Eastern United States.
The research team will now collect ticks across Georgia for testing to better understand what could raise the risk of getting the Heartland virus.
“We want to start filling in the huge gaps of knowledge of the transmission cycle for Heartland virus,” Dr. Vazquez-Prokopec said. “We need to better understand the key actors that transmit the virus and any environmental factors that may help it to persist within different habitats.”
A version of this article first appeared on WebMD.com.
FROM EMERGING INFECTIOUS DISEASES
More questions than answers when managing HIV and menopause
Note: In this article, “women” refers to ciswomen – those who identify as women and were assigned female sex at birth. Menopause also affects transmen and nonbinary people, but published research on the menopause experience has included only ciswomen participants.
Gina Brown was boarding an early morning flight in 2016 when suddenly she started to overheat. “As soon as I stepped on the plane, I immediately was drenched in sweat,” she said. Not knowing what to do, she stood still until a fellow female passenger noticed her alarm and asked a flight attendant to grab her a cup of ice. “Is this the first time this has happened to you?” the woman asked, and Ms. Brown nodded. “It’s called a hot flash,” the woman continued, “and you’re going to be okay.”
As soon as Ms. Brown returned from her trip, she visited her doctor for blood work and learned that her hormone levels were decreasing. “I knew something was going on, but [my provider and I] didn’t have a conversation about menopause,” she said. Ms. Brown, who is 56 years old, has been living with HIV for nearly 28 years, and is part of a growing group of women with HIV now entering menopause.
In 1996, a person diagnosed with HIV at 20 years of age could expect to live only to age 39. Because of antiretroviral therapy (ART), an HIV diagnosis is not nearly so dire. Now, someone with HIV who adheres to the ART regimen is estimated to have a lifespan close to that of the general population.
For women with HIV, this means going through menopause. Though this transition can be challenging for any woman, experiencing menopause with HIV adds another level of complication. On top of adhering to daily ART regimens, the woman must also deal with the hormonal changes of menopause and the symptoms that come with it. And the limited research in this area suggests that women with HIV and their clinicians may not be prepared.
“Those of us long-term survivors who have been around for a while never expected to be here, and I don’t think providers or the health care system expected us to be here,” said Vickie A. Lynn, PhD, 56, who has been living with HIV for 37 years and received an AIDS diagnosis in 1991. Her work focuses on health care interventions for people with HIV. “So now that we’re here, I don’t know that we have enough information or research to inform some of our treatment options.” Instead, these women are met with a series of unknowns due to limited studies and conflicting findings.
Earlier menopause?
The onset of menopause can be difficult to determine in women living with HIV, said Sara Looby, PhD, ANP-BC, a researcher at Massachusetts General Hospital and assistant professor of medicine at Harvard Medical School, both in Boston. Her research focuses on metabolic disorders, including bone loss, cardiovascular disease risk, and menopause in women living with HIV. This population is at an increased risk for amenorrhea, due to both behavioral and clinical factors, and sometimes this amenorrhea is mistakenly assumed to be menopause, she explained. A history of smoking, low weight, methadone use, or use of other psychotropic medications are common in women with HIV and can lead to missed periods. Some factors specific to HIV – including a low CD4 count and a history of an AIDS diagnosis – have also been linked to amenorrhea.
This is likely why research studies on the age of onset of menopause with women with HIV can reach conflicting conclusions. Some studies suggest that women with HIV tend to go through menopause 3-5 years earlier than women without HIV. Other studies suggest no difference in the age of onset in menopause between women living with and without HIV. But how menopause status has been accessed can vary from study to study, Dr. Looby said. Future research needs to consider participants’ complete menstrual and reproductive history, as well as relevant medical, social, and behavioral factors, she added, so that the findings are reliably capturing the age of onset of menopause rather than amenorrhea from other causes.
If menopause does occur earlier in women with HIV, there could be additional health implications. Estrogen regulates bone mass, and some research suggests the hormone may be cardioprotective. Estrogen is also thought to increase production of the neurotransmitter serotonin, which could affect mood and cognition. Women with HIV are already at higher risk for bone loss, cardiovascular disease, and depressed mood compared to women without HIV, Dr. Looby said, and as estrogen levels fall during menopause, these conditions may be deleteriously affected.
“If it is determined that women with HIV experience menopause at an earlier age, maybe early to mid-40s instead of 51 and older, they may be at increased risk for cardiovascular and bone conditions as well as mood symptoms associated with estrogen loss at an earlier age than women without HIV, which could be highly detrimental to their physical and mental health,” Dr. Looby said.
More frequent and severe menopausal symptoms?
Women with HIV may not only go through menopause earlier than women without HIV, but their symptoms may also be more frequent and more severe. In a 2017 study of both HIV-positive and HIV-negative Nigerian women, participants with HIV had more menopause symptoms overall and were three times as likely to report severe symptoms compared to women without HIV. A 2005 study conducted in New York found HIV-positive women were 24% more likely to report menopause symptoms compared to HIV-negative women in the study.
Looby’s own research has also found a similar pattern. In a study comparing 33 women with HIV to 33 women without HIV – all were close to menopause and matched for age, race, body mass index, and menstrual patterns – women with HIV reported more severe hot flashes and more days with hot flashes. These women also reported that their hot flashes interfered to a much greater degree with daily activities and quality of life compared to participants without HIV.
But studies of women with HIV who are entering menopause are rare, and most include only small numbers of women. As a result, many women with HIV do not know what to expect entering menopause. “I always say, I wish somebody would do some real research on HIV and menopause, because I want to know if it is worse for us or if it is the same,” said Ms. Brown, who works as the director of strategic partnership and community engagement at the Southern Aids Coalition in Powder Springs, Ga. “I would think it’s worse for me.”
More frequent and severe symptoms can have downstream effects, with some evidence suggesting that women with HIV who experience severe menopause symptoms are less likely to stick to their ART regimen. “There’s a clear picture emerging that menopausal symptoms in this group really matter,” said Shema Tariq, PhD, FRCP, an HIV physician-scientist at the University College London Institute for Global Health in England. “They really impact women’s well-being, as well as impacting their ability to look after their long-term condition.”
Providers wary of treating menopause symptoms in women with HIV
The little research we do have about women with HIV experiencing menopause suggests that this population could greatly benefit from treatment prescribed in women without HIV for menopause symptoms and conditions, including hormone replacement therapy (HRT). Women with HIV regularly experience night sweats and hot flashes during the menopause transition and may have more severe symptoms than women not living with the virus. If women with HIV also frequently enter early menopause (entering menopause before the age of 45), then this group meets two indications for hormone replacement therapy.
Despite the potential benefits of HRT in this population, some studies suggest this intervention is underutilized. In Dr. Tariq’s Positive Transitions through Menopause (PRIME) study, which explores how menopause affects more than 800 women living with HIV, only 8% of respondents reported using HRT. In a Canadian study that has not yet gone through peer review, 11.8% of perimenopausal and postmenopausal women reported ever using HRT, about half the rate of women in North America without HIV.
Provider discomfort with managing menopause-related care in women with HIV is one reason for such low HRT use in this population, Dr. Tariq said. In a survey of 88 general practitioners in the United Kingdom, nearly all (> 95%) respondents said they were comfortable managing menopause in a general population, but just 46% said they felt comfortable managing menopause in women with HIV. Their top concerns included the potential for drug-to-drug interactions between ART and HRT, missing an HIV-related diagnosis, and risks of menopausal hormone therapy in HIV. Nearly half of respondents (46%) said only specialists should be providing menopause-related care for women with HIV.
But specialists may also feel conflicted about managing menopause-related care in women with HIV, said Dr. Tariq. “If you’re looking at people who manage HIV, you’re looking primarily at infectious disease physicians and HIV physicians. We’re not trained as gynecologists. We’re not used to prescribing HRT,” she said. “And the problem is gynecologists aren’t used to managing HIV. They get nervous about prescribing anything when they see antiretroviral medication because all that people think of is a drug-drug interaction.”
This leaves women with HIV seeking care and treatment for menopause in a difficult situation, where they are “just being ping-ponged around between different health care providers,” said Susan Cole-Haley, 53, an HIV-activist in London who has been living with the virus for 23 years. “So many women with HIV have multiple health conditions and multiple health care providers, which can just make it really problematic and really exhausting in terms of getting help.”
Many unknowns
Providers may also be uncomfortable with prescribing hormone therapy because of alarming research in the early 2000s, which found that hormone replacement therapy increased the risk of breast cancer and cardiovascular disease. Later analyses have found no increased cardiovascular disease risk in women who were younger than 60 or were less than 10 years beyond the onset of menopause. Still, the “media frenzy” around the initial findings “has put off a whole load of patients and a whole load of clinicians from even thinking of HRT,” Dr. Tariq said.
Providers may be even more hesitant because people with HIV already have a higher risk for heart disease, due to behaviors such as smoking and HIV-specific factors. (Research has yet to tease out whether these cardiovascular effects are a result of the virus, a result of the antiretroviral therapy, or a result of both factors.) In addition, there have been no prospective studies looking directly at the efficacy and safety of hormone replacement therapy in women with HIV, so providers generally rely on the guidelines for the use of menopausal hormone therapy for women without HIV. While researchers from Canada and the United Kingdom have compiled recommendations for HRT in women with HIV, there is great need for a large-scale clinical trial to establish consistent guidelines for the use of HRT for women with HIV globally, Dr. Looby said.
There are also hormonal preparations and drug-to-drug interactions to consider, though none of the interactions identified so far rise to the level of contraindications. Because of how the liver metabolizes ART and HRT, hormone doses may need to be adjusted, or perhaps administered transdermally via a patch versus a pill form. (Estrogen delivered via skin patch may have reduced cardiovascular disease risk compared to other methods of delivery, some studies in women without HIV suggest.) These expected interactions are based on data from contraceptives, noted Elizabeth King, MD, whose research at the Women’s Health Research Institute at BC Women’s Hospital in Vancouver, B.C., focuses on menopause and HIV. Studies have not been done on drug-drug interactions between ART and HRT specifically, she said, and formulations for HRT are a bit different from contraceptives.
While these unknowns do need to be discussed in shared decision-making around starting HRT in women with HIV, they should not dissuade providers from considering the treatment, Dr. King said. “If women are having extremely troublesome symptoms, then withholding therapy that is potentially beneficial because of worries about some of the things we do not know – I don’t know if that is any better,” she said.
Many women with HIV may not want to start HRT – as was the case for Dr. Lynn. “I’ve taken a lot of medication in my time, and I really try to avoid it as much as possible,” she said. Uncertainties around drug interactions were the main concern for Dawn Averitt, 53, founder of the Well Project, an HIV nonprofit focused on women and girls. Ms. Averitt has lived with HIV for 34 years. “What if some of the things that I’m dealing with could be managed by HRT?” she said. “Or what if taking it exacerbates problems in a way that nobody knows to look for?” In this case, providers may work with patients to discuss nonhormonal treatment options for menopause symptom management.
While some women with HIV may not want HRT, “It’s important that women have that option, and from what we are seeing right now, not a lot of women are even being offered the therapy,” Dr. King said.
There are other nonhormonal treatments available for managing menopause symptoms, including selective serotonin reuptake inhibitors (SSRIs) as well as nonmedicinal interventions such as cognitive behavioral therapy, but these also have not been studied specifically in women with HIV.
The path forward
Dr. Tariq and Dr. Looby agreed the next step in expanding our knowledge around HIV and menopause should be to better engage women with HIV in research and clinical care around their experience with menopause. This includes studies on the symptoms they regularly experience and how these symptoms affect their quality of life, including their physical, psychological, cognitive, and social health. These studies could also help researchers and clinicians understand what these women with HIV want for their menopause care, whether that be medication, psychotherapy, and/or peer support groups. These interventions, whether pharmaceutical based or not, can then be assessed based on outcomes in women with HIV, Dr. Tariq noted.
Another important factor is increasing education, on both the patient and provider side, Dr. Looby said. Many women may not know what menopause is, what symptoms look like, and how these hormonal changes can affect their health. If providers keep an open dialogue with female patients around menopause throughout their adult care, that can better prepare women for the menopause transition and alert them to common symptoms they may experience. There also is a great need for provider education, Dr. Looby added. Infectious disease specialists may need further education on menopause management, while women’s health specialists may need additional training for managing care for patients with HIV. Ideally, this information could be shared among a team of providers, including infectious disease, primary care, and women’s health specialists, so that clinicians can collaborate in prescribing treatment for women with HIV, Dr. Looby said.
Lastly, there needs to be more research funding allocated toward answering questions related to menopause and HIV, including the age of onset of menopause in women with HIV, the severity of symptoms, how HIV may influence the menopause transition and vice versa, and regarding the effectiveness of treatment – pharmaceutical and nonpharmaceutical – for women with HIV going through the menopause transition. “If we don’t have funding for these studies, then we won’t have answers to establish clinical care guidelines necessary to support the health, well-being, and quality of life of women with HIV,” Dr. Looby said.
And the number of women living with HIV entering menopause is expected to keep growing, Dr. King added. “It was only a couple of decades ago when women were being told they wouldn’t even live to experience menopause, and now we are at a point where this is the highest proportion of menopausal women ever that we have seen in our HIV clinics,” she said. “It speaks to the success of antiretrovirals,” Dr. King acknowledged, but that also means identifying new challenges and addressing recognized gaps in care.
“We are charting a new course, in some ways,” she added. “There is a lot of work to be done.”
A version of this article first appeared on Medscape.com.
Note: In this article, “women” refers to ciswomen – those who identify as women and were assigned female sex at birth. Menopause also affects transmen and nonbinary people, but published research on the menopause experience has included only ciswomen participants.
Gina Brown was boarding an early morning flight in 2016 when suddenly she started to overheat. “As soon as I stepped on the plane, I immediately was drenched in sweat,” she said. Not knowing what to do, she stood still until a fellow female passenger noticed her alarm and asked a flight attendant to grab her a cup of ice. “Is this the first time this has happened to you?” the woman asked, and Ms. Brown nodded. “It’s called a hot flash,” the woman continued, “and you’re going to be okay.”
As soon as Ms. Brown returned from her trip, she visited her doctor for blood work and learned that her hormone levels were decreasing. “I knew something was going on, but [my provider and I] didn’t have a conversation about menopause,” she said. Ms. Brown, who is 56 years old, has been living with HIV for nearly 28 years, and is part of a growing group of women with HIV now entering menopause.
In 1996, a person diagnosed with HIV at 20 years of age could expect to live only to age 39. Because of antiretroviral therapy (ART), an HIV diagnosis is not nearly so dire. Now, someone with HIV who adheres to the ART regimen is estimated to have a lifespan close to that of the general population.
For women with HIV, this means going through menopause. Though this transition can be challenging for any woman, experiencing menopause with HIV adds another level of complication. On top of adhering to daily ART regimens, the woman must also deal with the hormonal changes of menopause and the symptoms that come with it. And the limited research in this area suggests that women with HIV and their clinicians may not be prepared.
“Those of us long-term survivors who have been around for a while never expected to be here, and I don’t think providers or the health care system expected us to be here,” said Vickie A. Lynn, PhD, 56, who has been living with HIV for 37 years and received an AIDS diagnosis in 1991. Her work focuses on health care interventions for people with HIV. “So now that we’re here, I don’t know that we have enough information or research to inform some of our treatment options.” Instead, these women are met with a series of unknowns due to limited studies and conflicting findings.
Earlier menopause?
The onset of menopause can be difficult to determine in women living with HIV, said Sara Looby, PhD, ANP-BC, a researcher at Massachusetts General Hospital and assistant professor of medicine at Harvard Medical School, both in Boston. Her research focuses on metabolic disorders, including bone loss, cardiovascular disease risk, and menopause in women living with HIV. This population is at an increased risk for amenorrhea, due to both behavioral and clinical factors, and sometimes this amenorrhea is mistakenly assumed to be menopause, she explained. A history of smoking, low weight, methadone use, or use of other psychotropic medications are common in women with HIV and can lead to missed periods. Some factors specific to HIV – including a low CD4 count and a history of an AIDS diagnosis – have also been linked to amenorrhea.
This is likely why research studies on the age of onset of menopause with women with HIV can reach conflicting conclusions. Some studies suggest that women with HIV tend to go through menopause 3-5 years earlier than women without HIV. Other studies suggest no difference in the age of onset in menopause between women living with and without HIV. But how menopause status has been accessed can vary from study to study, Dr. Looby said. Future research needs to consider participants’ complete menstrual and reproductive history, as well as relevant medical, social, and behavioral factors, she added, so that the findings are reliably capturing the age of onset of menopause rather than amenorrhea from other causes.
If menopause does occur earlier in women with HIV, there could be additional health implications. Estrogen regulates bone mass, and some research suggests the hormone may be cardioprotective. Estrogen is also thought to increase production of the neurotransmitter serotonin, which could affect mood and cognition. Women with HIV are already at higher risk for bone loss, cardiovascular disease, and depressed mood compared to women without HIV, Dr. Looby said, and as estrogen levels fall during menopause, these conditions may be deleteriously affected.
“If it is determined that women with HIV experience menopause at an earlier age, maybe early to mid-40s instead of 51 and older, they may be at increased risk for cardiovascular and bone conditions as well as mood symptoms associated with estrogen loss at an earlier age than women without HIV, which could be highly detrimental to their physical and mental health,” Dr. Looby said.
More frequent and severe menopausal symptoms?
Women with HIV may not only go through menopause earlier than women without HIV, but their symptoms may also be more frequent and more severe. In a 2017 study of both HIV-positive and HIV-negative Nigerian women, participants with HIV had more menopause symptoms overall and were three times as likely to report severe symptoms compared to women without HIV. A 2005 study conducted in New York found HIV-positive women were 24% more likely to report menopause symptoms compared to HIV-negative women in the study.
Looby’s own research has also found a similar pattern. In a study comparing 33 women with HIV to 33 women without HIV – all were close to menopause and matched for age, race, body mass index, and menstrual patterns – women with HIV reported more severe hot flashes and more days with hot flashes. These women also reported that their hot flashes interfered to a much greater degree with daily activities and quality of life compared to participants without HIV.
But studies of women with HIV who are entering menopause are rare, and most include only small numbers of women. As a result, many women with HIV do not know what to expect entering menopause. “I always say, I wish somebody would do some real research on HIV and menopause, because I want to know if it is worse for us or if it is the same,” said Ms. Brown, who works as the director of strategic partnership and community engagement at the Southern Aids Coalition in Powder Springs, Ga. “I would think it’s worse for me.”
More frequent and severe symptoms can have downstream effects, with some evidence suggesting that women with HIV who experience severe menopause symptoms are less likely to stick to their ART regimen. “There’s a clear picture emerging that menopausal symptoms in this group really matter,” said Shema Tariq, PhD, FRCP, an HIV physician-scientist at the University College London Institute for Global Health in England. “They really impact women’s well-being, as well as impacting their ability to look after their long-term condition.”
Providers wary of treating menopause symptoms in women with HIV
The little research we do have about women with HIV experiencing menopause suggests that this population could greatly benefit from treatment prescribed in women without HIV for menopause symptoms and conditions, including hormone replacement therapy (HRT). Women with HIV regularly experience night sweats and hot flashes during the menopause transition and may have more severe symptoms than women not living with the virus. If women with HIV also frequently enter early menopause (entering menopause before the age of 45), then this group meets two indications for hormone replacement therapy.
Despite the potential benefits of HRT in this population, some studies suggest this intervention is underutilized. In Dr. Tariq’s Positive Transitions through Menopause (PRIME) study, which explores how menopause affects more than 800 women living with HIV, only 8% of respondents reported using HRT. In a Canadian study that has not yet gone through peer review, 11.8% of perimenopausal and postmenopausal women reported ever using HRT, about half the rate of women in North America without HIV.
Provider discomfort with managing menopause-related care in women with HIV is one reason for such low HRT use in this population, Dr. Tariq said. In a survey of 88 general practitioners in the United Kingdom, nearly all (> 95%) respondents said they were comfortable managing menopause in a general population, but just 46% said they felt comfortable managing menopause in women with HIV. Their top concerns included the potential for drug-to-drug interactions between ART and HRT, missing an HIV-related diagnosis, and risks of menopausal hormone therapy in HIV. Nearly half of respondents (46%) said only specialists should be providing menopause-related care for women with HIV.
But specialists may also feel conflicted about managing menopause-related care in women with HIV, said Dr. Tariq. “If you’re looking at people who manage HIV, you’re looking primarily at infectious disease physicians and HIV physicians. We’re not trained as gynecologists. We’re not used to prescribing HRT,” she said. “And the problem is gynecologists aren’t used to managing HIV. They get nervous about prescribing anything when they see antiretroviral medication because all that people think of is a drug-drug interaction.”
This leaves women with HIV seeking care and treatment for menopause in a difficult situation, where they are “just being ping-ponged around between different health care providers,” said Susan Cole-Haley, 53, an HIV-activist in London who has been living with the virus for 23 years. “So many women with HIV have multiple health conditions and multiple health care providers, which can just make it really problematic and really exhausting in terms of getting help.”
Many unknowns
Providers may also be uncomfortable with prescribing hormone therapy because of alarming research in the early 2000s, which found that hormone replacement therapy increased the risk of breast cancer and cardiovascular disease. Later analyses have found no increased cardiovascular disease risk in women who were younger than 60 or were less than 10 years beyond the onset of menopause. Still, the “media frenzy” around the initial findings “has put off a whole load of patients and a whole load of clinicians from even thinking of HRT,” Dr. Tariq said.
Providers may be even more hesitant because people with HIV already have a higher risk for heart disease, due to behaviors such as smoking and HIV-specific factors. (Research has yet to tease out whether these cardiovascular effects are a result of the virus, a result of the antiretroviral therapy, or a result of both factors.) In addition, there have been no prospective studies looking directly at the efficacy and safety of hormone replacement therapy in women with HIV, so providers generally rely on the guidelines for the use of menopausal hormone therapy for women without HIV. While researchers from Canada and the United Kingdom have compiled recommendations for HRT in women with HIV, there is great need for a large-scale clinical trial to establish consistent guidelines for the use of HRT for women with HIV globally, Dr. Looby said.
There are also hormonal preparations and drug-to-drug interactions to consider, though none of the interactions identified so far rise to the level of contraindications. Because of how the liver metabolizes ART and HRT, hormone doses may need to be adjusted, or perhaps administered transdermally via a patch versus a pill form. (Estrogen delivered via skin patch may have reduced cardiovascular disease risk compared to other methods of delivery, some studies in women without HIV suggest.) These expected interactions are based on data from contraceptives, noted Elizabeth King, MD, whose research at the Women’s Health Research Institute at BC Women’s Hospital in Vancouver, B.C., focuses on menopause and HIV. Studies have not been done on drug-drug interactions between ART and HRT specifically, she said, and formulations for HRT are a bit different from contraceptives.
While these unknowns do need to be discussed in shared decision-making around starting HRT in women with HIV, they should not dissuade providers from considering the treatment, Dr. King said. “If women are having extremely troublesome symptoms, then withholding therapy that is potentially beneficial because of worries about some of the things we do not know – I don’t know if that is any better,” she said.
Many women with HIV may not want to start HRT – as was the case for Dr. Lynn. “I’ve taken a lot of medication in my time, and I really try to avoid it as much as possible,” she said. Uncertainties around drug interactions were the main concern for Dawn Averitt, 53, founder of the Well Project, an HIV nonprofit focused on women and girls. Ms. Averitt has lived with HIV for 34 years. “What if some of the things that I’m dealing with could be managed by HRT?” she said. “Or what if taking it exacerbates problems in a way that nobody knows to look for?” In this case, providers may work with patients to discuss nonhormonal treatment options for menopause symptom management.
While some women with HIV may not want HRT, “It’s important that women have that option, and from what we are seeing right now, not a lot of women are even being offered the therapy,” Dr. King said.
There are other nonhormonal treatments available for managing menopause symptoms, including selective serotonin reuptake inhibitors (SSRIs) as well as nonmedicinal interventions such as cognitive behavioral therapy, but these also have not been studied specifically in women with HIV.
The path forward
Dr. Tariq and Dr. Looby agreed the next step in expanding our knowledge around HIV and menopause should be to better engage women with HIV in research and clinical care around their experience with menopause. This includes studies on the symptoms they regularly experience and how these symptoms affect their quality of life, including their physical, psychological, cognitive, and social health. These studies could also help researchers and clinicians understand what these women with HIV want for their menopause care, whether that be medication, psychotherapy, and/or peer support groups. These interventions, whether pharmaceutical based or not, can then be assessed based on outcomes in women with HIV, Dr. Tariq noted.
Another important factor is increasing education, on both the patient and provider side, Dr. Looby said. Many women may not know what menopause is, what symptoms look like, and how these hormonal changes can affect their health. If providers keep an open dialogue with female patients around menopause throughout their adult care, that can better prepare women for the menopause transition and alert them to common symptoms they may experience. There also is a great need for provider education, Dr. Looby added. Infectious disease specialists may need further education on menopause management, while women’s health specialists may need additional training for managing care for patients with HIV. Ideally, this information could be shared among a team of providers, including infectious disease, primary care, and women’s health specialists, so that clinicians can collaborate in prescribing treatment for women with HIV, Dr. Looby said.
Lastly, there needs to be more research funding allocated toward answering questions related to menopause and HIV, including the age of onset of menopause in women with HIV, the severity of symptoms, how HIV may influence the menopause transition and vice versa, and regarding the effectiveness of treatment – pharmaceutical and nonpharmaceutical – for women with HIV going through the menopause transition. “If we don’t have funding for these studies, then we won’t have answers to establish clinical care guidelines necessary to support the health, well-being, and quality of life of women with HIV,” Dr. Looby said.
And the number of women living with HIV entering menopause is expected to keep growing, Dr. King added. “It was only a couple of decades ago when women were being told they wouldn’t even live to experience menopause, and now we are at a point where this is the highest proportion of menopausal women ever that we have seen in our HIV clinics,” she said. “It speaks to the success of antiretrovirals,” Dr. King acknowledged, but that also means identifying new challenges and addressing recognized gaps in care.
“We are charting a new course, in some ways,” she added. “There is a lot of work to be done.”
A version of this article first appeared on Medscape.com.
Note: In this article, “women” refers to ciswomen – those who identify as women and were assigned female sex at birth. Menopause also affects transmen and nonbinary people, but published research on the menopause experience has included only ciswomen participants.
Gina Brown was boarding an early morning flight in 2016 when suddenly she started to overheat. “As soon as I stepped on the plane, I immediately was drenched in sweat,” she said. Not knowing what to do, she stood still until a fellow female passenger noticed her alarm and asked a flight attendant to grab her a cup of ice. “Is this the first time this has happened to you?” the woman asked, and Ms. Brown nodded. “It’s called a hot flash,” the woman continued, “and you’re going to be okay.”
As soon as Ms. Brown returned from her trip, she visited her doctor for blood work and learned that her hormone levels were decreasing. “I knew something was going on, but [my provider and I] didn’t have a conversation about menopause,” she said. Ms. Brown, who is 56 years old, has been living with HIV for nearly 28 years, and is part of a growing group of women with HIV now entering menopause.
In 1996, a person diagnosed with HIV at 20 years of age could expect to live only to age 39. Because of antiretroviral therapy (ART), an HIV diagnosis is not nearly so dire. Now, someone with HIV who adheres to the ART regimen is estimated to have a lifespan close to that of the general population.
For women with HIV, this means going through menopause. Though this transition can be challenging for any woman, experiencing menopause with HIV adds another level of complication. On top of adhering to daily ART regimens, the woman must also deal with the hormonal changes of menopause and the symptoms that come with it. And the limited research in this area suggests that women with HIV and their clinicians may not be prepared.
“Those of us long-term survivors who have been around for a while never expected to be here, and I don’t think providers or the health care system expected us to be here,” said Vickie A. Lynn, PhD, 56, who has been living with HIV for 37 years and received an AIDS diagnosis in 1991. Her work focuses on health care interventions for people with HIV. “So now that we’re here, I don’t know that we have enough information or research to inform some of our treatment options.” Instead, these women are met with a series of unknowns due to limited studies and conflicting findings.
Earlier menopause?
The onset of menopause can be difficult to determine in women living with HIV, said Sara Looby, PhD, ANP-BC, a researcher at Massachusetts General Hospital and assistant professor of medicine at Harvard Medical School, both in Boston. Her research focuses on metabolic disorders, including bone loss, cardiovascular disease risk, and menopause in women living with HIV. This population is at an increased risk for amenorrhea, due to both behavioral and clinical factors, and sometimes this amenorrhea is mistakenly assumed to be menopause, she explained. A history of smoking, low weight, methadone use, or use of other psychotropic medications are common in women with HIV and can lead to missed periods. Some factors specific to HIV – including a low CD4 count and a history of an AIDS diagnosis – have also been linked to amenorrhea.
This is likely why research studies on the age of onset of menopause with women with HIV can reach conflicting conclusions. Some studies suggest that women with HIV tend to go through menopause 3-5 years earlier than women without HIV. Other studies suggest no difference in the age of onset in menopause between women living with and without HIV. But how menopause status has been accessed can vary from study to study, Dr. Looby said. Future research needs to consider participants’ complete menstrual and reproductive history, as well as relevant medical, social, and behavioral factors, she added, so that the findings are reliably capturing the age of onset of menopause rather than amenorrhea from other causes.
If menopause does occur earlier in women with HIV, there could be additional health implications. Estrogen regulates bone mass, and some research suggests the hormone may be cardioprotective. Estrogen is also thought to increase production of the neurotransmitter serotonin, which could affect mood and cognition. Women with HIV are already at higher risk for bone loss, cardiovascular disease, and depressed mood compared to women without HIV, Dr. Looby said, and as estrogen levels fall during menopause, these conditions may be deleteriously affected.
“If it is determined that women with HIV experience menopause at an earlier age, maybe early to mid-40s instead of 51 and older, they may be at increased risk for cardiovascular and bone conditions as well as mood symptoms associated with estrogen loss at an earlier age than women without HIV, which could be highly detrimental to their physical and mental health,” Dr. Looby said.
More frequent and severe menopausal symptoms?
Women with HIV may not only go through menopause earlier than women without HIV, but their symptoms may also be more frequent and more severe. In a 2017 study of both HIV-positive and HIV-negative Nigerian women, participants with HIV had more menopause symptoms overall and were three times as likely to report severe symptoms compared to women without HIV. A 2005 study conducted in New York found HIV-positive women were 24% more likely to report menopause symptoms compared to HIV-negative women in the study.
Looby’s own research has also found a similar pattern. In a study comparing 33 women with HIV to 33 women without HIV – all were close to menopause and matched for age, race, body mass index, and menstrual patterns – women with HIV reported more severe hot flashes and more days with hot flashes. These women also reported that their hot flashes interfered to a much greater degree with daily activities and quality of life compared to participants without HIV.
But studies of women with HIV who are entering menopause are rare, and most include only small numbers of women. As a result, many women with HIV do not know what to expect entering menopause. “I always say, I wish somebody would do some real research on HIV and menopause, because I want to know if it is worse for us or if it is the same,” said Ms. Brown, who works as the director of strategic partnership and community engagement at the Southern Aids Coalition in Powder Springs, Ga. “I would think it’s worse for me.”
More frequent and severe symptoms can have downstream effects, with some evidence suggesting that women with HIV who experience severe menopause symptoms are less likely to stick to their ART regimen. “There’s a clear picture emerging that menopausal symptoms in this group really matter,” said Shema Tariq, PhD, FRCP, an HIV physician-scientist at the University College London Institute for Global Health in England. “They really impact women’s well-being, as well as impacting their ability to look after their long-term condition.”
Providers wary of treating menopause symptoms in women with HIV
The little research we do have about women with HIV experiencing menopause suggests that this population could greatly benefit from treatment prescribed in women without HIV for menopause symptoms and conditions, including hormone replacement therapy (HRT). Women with HIV regularly experience night sweats and hot flashes during the menopause transition and may have more severe symptoms than women not living with the virus. If women with HIV also frequently enter early menopause (entering menopause before the age of 45), then this group meets two indications for hormone replacement therapy.
Despite the potential benefits of HRT in this population, some studies suggest this intervention is underutilized. In Dr. Tariq’s Positive Transitions through Menopause (PRIME) study, which explores how menopause affects more than 800 women living with HIV, only 8% of respondents reported using HRT. In a Canadian study that has not yet gone through peer review, 11.8% of perimenopausal and postmenopausal women reported ever using HRT, about half the rate of women in North America without HIV.
Provider discomfort with managing menopause-related care in women with HIV is one reason for such low HRT use in this population, Dr. Tariq said. In a survey of 88 general practitioners in the United Kingdom, nearly all (> 95%) respondents said they were comfortable managing menopause in a general population, but just 46% said they felt comfortable managing menopause in women with HIV. Their top concerns included the potential for drug-to-drug interactions between ART and HRT, missing an HIV-related diagnosis, and risks of menopausal hormone therapy in HIV. Nearly half of respondents (46%) said only specialists should be providing menopause-related care for women with HIV.
But specialists may also feel conflicted about managing menopause-related care in women with HIV, said Dr. Tariq. “If you’re looking at people who manage HIV, you’re looking primarily at infectious disease physicians and HIV physicians. We’re not trained as gynecologists. We’re not used to prescribing HRT,” she said. “And the problem is gynecologists aren’t used to managing HIV. They get nervous about prescribing anything when they see antiretroviral medication because all that people think of is a drug-drug interaction.”
This leaves women with HIV seeking care and treatment for menopause in a difficult situation, where they are “just being ping-ponged around between different health care providers,” said Susan Cole-Haley, 53, an HIV-activist in London who has been living with the virus for 23 years. “So many women with HIV have multiple health conditions and multiple health care providers, which can just make it really problematic and really exhausting in terms of getting help.”
Many unknowns
Providers may also be uncomfortable with prescribing hormone therapy because of alarming research in the early 2000s, which found that hormone replacement therapy increased the risk of breast cancer and cardiovascular disease. Later analyses have found no increased cardiovascular disease risk in women who were younger than 60 or were less than 10 years beyond the onset of menopause. Still, the “media frenzy” around the initial findings “has put off a whole load of patients and a whole load of clinicians from even thinking of HRT,” Dr. Tariq said.
Providers may be even more hesitant because people with HIV already have a higher risk for heart disease, due to behaviors such as smoking and HIV-specific factors. (Research has yet to tease out whether these cardiovascular effects are a result of the virus, a result of the antiretroviral therapy, or a result of both factors.) In addition, there have been no prospective studies looking directly at the efficacy and safety of hormone replacement therapy in women with HIV, so providers generally rely on the guidelines for the use of menopausal hormone therapy for women without HIV. While researchers from Canada and the United Kingdom have compiled recommendations for HRT in women with HIV, there is great need for a large-scale clinical trial to establish consistent guidelines for the use of HRT for women with HIV globally, Dr. Looby said.
There are also hormonal preparations and drug-to-drug interactions to consider, though none of the interactions identified so far rise to the level of contraindications. Because of how the liver metabolizes ART and HRT, hormone doses may need to be adjusted, or perhaps administered transdermally via a patch versus a pill form. (Estrogen delivered via skin patch may have reduced cardiovascular disease risk compared to other methods of delivery, some studies in women without HIV suggest.) These expected interactions are based on data from contraceptives, noted Elizabeth King, MD, whose research at the Women’s Health Research Institute at BC Women’s Hospital in Vancouver, B.C., focuses on menopause and HIV. Studies have not been done on drug-drug interactions between ART and HRT specifically, she said, and formulations for HRT are a bit different from contraceptives.
While these unknowns do need to be discussed in shared decision-making around starting HRT in women with HIV, they should not dissuade providers from considering the treatment, Dr. King said. “If women are having extremely troublesome symptoms, then withholding therapy that is potentially beneficial because of worries about some of the things we do not know – I don’t know if that is any better,” she said.
Many women with HIV may not want to start HRT – as was the case for Dr. Lynn. “I’ve taken a lot of medication in my time, and I really try to avoid it as much as possible,” she said. Uncertainties around drug interactions were the main concern for Dawn Averitt, 53, founder of the Well Project, an HIV nonprofit focused on women and girls. Ms. Averitt has lived with HIV for 34 years. “What if some of the things that I’m dealing with could be managed by HRT?” she said. “Or what if taking it exacerbates problems in a way that nobody knows to look for?” In this case, providers may work with patients to discuss nonhormonal treatment options for menopause symptom management.
While some women with HIV may not want HRT, “It’s important that women have that option, and from what we are seeing right now, not a lot of women are even being offered the therapy,” Dr. King said.
There are other nonhormonal treatments available for managing menopause symptoms, including selective serotonin reuptake inhibitors (SSRIs) as well as nonmedicinal interventions such as cognitive behavioral therapy, but these also have not been studied specifically in women with HIV.
The path forward
Dr. Tariq and Dr. Looby agreed the next step in expanding our knowledge around HIV and menopause should be to better engage women with HIV in research and clinical care around their experience with menopause. This includes studies on the symptoms they regularly experience and how these symptoms affect their quality of life, including their physical, psychological, cognitive, and social health. These studies could also help researchers and clinicians understand what these women with HIV want for their menopause care, whether that be medication, psychotherapy, and/or peer support groups. These interventions, whether pharmaceutical based or not, can then be assessed based on outcomes in women with HIV, Dr. Tariq noted.
Another important factor is increasing education, on both the patient and provider side, Dr. Looby said. Many women may not know what menopause is, what symptoms look like, and how these hormonal changes can affect their health. If providers keep an open dialogue with female patients around menopause throughout their adult care, that can better prepare women for the menopause transition and alert them to common symptoms they may experience. There also is a great need for provider education, Dr. Looby added. Infectious disease specialists may need further education on menopause management, while women’s health specialists may need additional training for managing care for patients with HIV. Ideally, this information could be shared among a team of providers, including infectious disease, primary care, and women’s health specialists, so that clinicians can collaborate in prescribing treatment for women with HIV, Dr. Looby said.
Lastly, there needs to be more research funding allocated toward answering questions related to menopause and HIV, including the age of onset of menopause in women with HIV, the severity of symptoms, how HIV may influence the menopause transition and vice versa, and regarding the effectiveness of treatment – pharmaceutical and nonpharmaceutical – for women with HIV going through the menopause transition. “If we don’t have funding for these studies, then we won’t have answers to establish clinical care guidelines necessary to support the health, well-being, and quality of life of women with HIV,” Dr. Looby said.
And the number of women living with HIV entering menopause is expected to keep growing, Dr. King added. “It was only a couple of decades ago when women were being told they wouldn’t even live to experience menopause, and now we are at a point where this is the highest proportion of menopausal women ever that we have seen in our HIV clinics,” she said. “It speaks to the success of antiretrovirals,” Dr. King acknowledged, but that also means identifying new challenges and addressing recognized gaps in care.
“We are charting a new course, in some ways,” she added. “There is a lot of work to be done.”
A version of this article first appeared on Medscape.com.
Moderna reports positive COVID-19 vaccine response in kids down to 6 months
Moderna on March 23 released interim results indicating that its mRNA-1273 COVID vaccine produced “robust” neutralizing antibody titers in children aged 6 months to 6 years – levels similar to those seen in adults.
Vaccine efficacy against infection was 43.7% in children aged 6 months to 2 years and 37.5% among children aged 2-6 years, the new data from its phase 2/3 KidCOVE study show.
The company explained the lower efficacy numbers by noting that its study involving these younger children was conducted during the Omicron wave. The same decrease in efficacy against infection was reported in adults during the Omicron surge.
A majority of COVID-19 cases were mild in the approximately 6,900 children aged 6 months to 6 years in the study. No severe COVID-19 cases, hospitalizations, or deaths were reported.
The primary series of two 25-mcg doses of the vaccine given 28 days apart was generally well tolerated. Most adverse events were mild to moderate. For example, temperature greater than 38° C (>100.4° F) was reported for 17.0% of the 6-month-old to 2-year-old group and for 14.6% of the 2- to 6-year-old group. A few children, 0.2% of each group, experienced a temperature greater than 40° C (>104° F).
Moderna plans to include these response, efficacy, and safety data in an application to the Food and Drug Administration for emergency use authorization (EUA) of the vaccine in these younger children in the coming weeks.
“We now have clinical data on the performance of our vaccine from infants 6 months of age through older adults,” Moderna CEO Stephane Bancel said in a news release. He described the interim results as “good news for parents of children under 6 years of age.”
In other news
Moderna also announced that it began the FDA EUA submission process for a 50-μg two-dose primary series for children aged 6-12 years.
The company is also updating its EUA submission for a 100-mcg two-dose primary series for children and adolescents aged 12-18 years.
Similar to its booster research in adults, Moderna plans to evaluate the potential of a booster dose for all pediatric populations, including those aged 6 months to 6 years, 6-12 years, and adolescents. The company is evaluating both a booster dose of mRNA-1273 and its bivalent booster candidate (mRNA1273.214), which includes an Omicron variant booster and mRNA-1273.
A version of this article first appeared on Medscape.com.
Moderna on March 23 released interim results indicating that its mRNA-1273 COVID vaccine produced “robust” neutralizing antibody titers in children aged 6 months to 6 years – levels similar to those seen in adults.
Vaccine efficacy against infection was 43.7% in children aged 6 months to 2 years and 37.5% among children aged 2-6 years, the new data from its phase 2/3 KidCOVE study show.
The company explained the lower efficacy numbers by noting that its study involving these younger children was conducted during the Omicron wave. The same decrease in efficacy against infection was reported in adults during the Omicron surge.
A majority of COVID-19 cases were mild in the approximately 6,900 children aged 6 months to 6 years in the study. No severe COVID-19 cases, hospitalizations, or deaths were reported.
The primary series of two 25-mcg doses of the vaccine given 28 days apart was generally well tolerated. Most adverse events were mild to moderate. For example, temperature greater than 38° C (>100.4° F) was reported for 17.0% of the 6-month-old to 2-year-old group and for 14.6% of the 2- to 6-year-old group. A few children, 0.2% of each group, experienced a temperature greater than 40° C (>104° F).
Moderna plans to include these response, efficacy, and safety data in an application to the Food and Drug Administration for emergency use authorization (EUA) of the vaccine in these younger children in the coming weeks.
“We now have clinical data on the performance of our vaccine from infants 6 months of age through older adults,” Moderna CEO Stephane Bancel said in a news release. He described the interim results as “good news for parents of children under 6 years of age.”
In other news
Moderna also announced that it began the FDA EUA submission process for a 50-μg two-dose primary series for children aged 6-12 years.
The company is also updating its EUA submission for a 100-mcg two-dose primary series for children and adolescents aged 12-18 years.
Similar to its booster research in adults, Moderna plans to evaluate the potential of a booster dose for all pediatric populations, including those aged 6 months to 6 years, 6-12 years, and adolescents. The company is evaluating both a booster dose of mRNA-1273 and its bivalent booster candidate (mRNA1273.214), which includes an Omicron variant booster and mRNA-1273.
A version of this article first appeared on Medscape.com.
Moderna on March 23 released interim results indicating that its mRNA-1273 COVID vaccine produced “robust” neutralizing antibody titers in children aged 6 months to 6 years – levels similar to those seen in adults.
Vaccine efficacy against infection was 43.7% in children aged 6 months to 2 years and 37.5% among children aged 2-6 years, the new data from its phase 2/3 KidCOVE study show.
The company explained the lower efficacy numbers by noting that its study involving these younger children was conducted during the Omicron wave. The same decrease in efficacy against infection was reported in adults during the Omicron surge.
A majority of COVID-19 cases were mild in the approximately 6,900 children aged 6 months to 6 years in the study. No severe COVID-19 cases, hospitalizations, or deaths were reported.
The primary series of two 25-mcg doses of the vaccine given 28 days apart was generally well tolerated. Most adverse events were mild to moderate. For example, temperature greater than 38° C (>100.4° F) was reported for 17.0% of the 6-month-old to 2-year-old group and for 14.6% of the 2- to 6-year-old group. A few children, 0.2% of each group, experienced a temperature greater than 40° C (>104° F).
Moderna plans to include these response, efficacy, and safety data in an application to the Food and Drug Administration for emergency use authorization (EUA) of the vaccine in these younger children in the coming weeks.
“We now have clinical data on the performance of our vaccine from infants 6 months of age through older adults,” Moderna CEO Stephane Bancel said in a news release. He described the interim results as “good news for parents of children under 6 years of age.”
In other news
Moderna also announced that it began the FDA EUA submission process for a 50-μg two-dose primary series for children aged 6-12 years.
The company is also updating its EUA submission for a 100-mcg two-dose primary series for children and adolescents aged 12-18 years.
Similar to its booster research in adults, Moderna plans to evaluate the potential of a booster dose for all pediatric populations, including those aged 6 months to 6 years, 6-12 years, and adolescents. The company is evaluating both a booster dose of mRNA-1273 and its bivalent booster candidate (mRNA1273.214), which includes an Omicron variant booster and mRNA-1273.
A version of this article first appeared on Medscape.com.
Pneumonia decision tool reduces death in ED patients
a 3-year, pragmatic, cluster-controlled study shows.
“We designed the ePNa specifically to require minimal input from the clinician so everything it does is already in the electronic medical record,” Nathan Dean, MD, University of Utah, Salt Lake City, told this news organization.
“So it’s actually putting the guideline recommendations into effect for physicians so that they can make better decisions by having all this information – it’s a comprehensive best practice kind of tool where best practices are likely to make the biggest difference for patients with a high severity of illness,” he added.
The study was published online in the American Journal of Respiratory and Critical Care Medicine.
Guideline-based tool
The ePNa makes use of pneumonia guidelines of 2007 and 2019 from the American Thoracic Society/Infectious Disease Society of America. The system was deployed into six geographic clusters of 16 Intermountain hospital EDs at 2-month intervals between December 2017 and November 2018. Simultaneous deployment was impractical, as implementation of the tool takes education, monitoring, and feedback that can be facilitated by focusing on only a few hospitals at a time.
The decision support tool gathers key patient indicators including age, fever, oxygen saturation, vital signs, and laboratory and chest imaging results to offer recommendations on care, including appropriate antibiotic therapy, microbiology studies, and whether a given patient should be sent to the intensive care unit, admitted to hospital, or may safely be discharged home.
Investigators analyzed a total of 6,848 patients, of whom 4,536 were managed for pneumonia before the ePNa was deployed and 2,312 after deployment.
The median age of patients was 67 years (interquartile range, 50-79 years). Roughly half were female and almost all were White. “Observed 30-day all-cause mortality including both outpatients and inpatients was 8.6% before deployment versus 4.8% after deployment of ePNa,” Dr. Dean and colleagues reported.
Adjusted for severity of illness, the odds ratio for lower mortality post-ePNa launch was 0.62 (95% confidence interval, 0.49-0.79; P < .0010) “and lower morality was consistent across hospital clusters.”
Compared with patients who were discharged home, reductions in mortality were greatest in patients who were directly admitted to ICUs from the ED (OR, 0.32; 95% CI, 0.14-0.77; P = .01). The OR for patients admitted to the medical floor was 0.53 (95% CI, 0.25-1.1; P = .09), which did not reach statistical significance.
Dr. Dean explained that the reductions in mortality were seen among those with the most severe illness, in whom best practices would benefit the most. In contrast, patients who are sent home on an antibiotic are at low risk for mortality while patients admitted to the medical floor may well have another, more lethal illness from which they end up dying, rather than simple pneumonia.
“For me, this was a clear demonstration that these best practices made the biggest difference in patients who were sick and who did not have any underlying disease that was going to kill them anyway,” he emphasized. On the other hand, both 30-day mortality and 7-day secondary hospital admission were higher among patients the tool recommended for hospital ward admission but who were discharged home from the ED.
“This was an unexpected finding,” Dr. Dean observed. However, as he explained, the authors reviewed 25% of randomly selected patients who fell into this subgroup and discovered that the ePNa tool was used in only about 20% of patients – “so doctors did not use the tool in the majority of this group.”
In addition, some of these patients declined hospital admission, so the doctors may have recommended that they be admitted but the patients said no. “The hypothesis here is that if they had been admitted to the hospital, they may have had a lower mortality risk,” Dr. Dean said.
Noticeable changes
Another noticeable change following the introduction of the ePNa tool was that guideline-concordant antibiotic prescribing increased in the 8 hours after patients presented to the ED, from 79.5% prior to the tool’s launch to 87.9%, again after adjusting for pneumonia severity (P < .001). Use of broad-spectrum antibiotics was not significantly different between the two treatment intervals, but administration of antibiotics active against methicillin-resistant Staphylococcus aureus dropped significantly between the two treatment intervals (P < .001). And the mean time from admission to the ED to the first antibiotic taken was slightly faster, improving from 159.4 minutes (95% CI, 156.9-161.9 minutes) prior to the ePNa launch to 150.9 minutes (95% CI, 144.1-157.8) post deployment (P < .001).
“Overall outpatient disposition for treatment of pneumonia from the emergency department increased from 29.2% before ePNa to 46.9% [post ePNA],” the authors noted, while a similar increase was observed in patients for whom ePNA recommended outpatient care – from 49.2% pre-ePNA to 66.6% after ePNA.
Both hospital ward admission and admission to the ICU decreased after ePNa had been introduced. Despite a significant increase in the percentage of patients being discharged home, neither 7-day secondary hospital admission nor severity-adjusted, 30-day mortality were significantly different before versus after the introduction of ePNa, the authors stressed.
A limitation of the study was that the trial was confined to a single health care system in one region of the United States with a patient population that may differ from that in other regions.
Reason for its success
Asked to comment on the findings, Adam Balls, MD, emergency department chair, Intermountain Medical Center, Murray, Utah, suggested that the reason the ePNa tool has been so successful at improving care for pneumonia patients is that it puts the guidelines directly into the hands of individual providers and tells them what’s going on. (Dr. Balls was not involved in the study.) “The tool allows us to take into consideration various clinical features – a patient’s oxygen requirements and whether or not they had prior complicated pneumonias that required additional antibiotics, for example – and then it makes the best determination for not only the disposition for that patient but antibiotic treatment as well,” he said in an interview.
This then allows physicians to either appropriately discharge less severely ill patients and admit those who are more ill – “and in general, just do a better job of treating pneumonia with this tool,” Dr. Balls said. He himself uses the decision support tool when attending to his own patients with pneumonia, as he feels that the tool really does make his care of these patients better. “There is a disparity around how we treat pneumonia in the U.S.
“Clinicians sometimes have a bias or a preference for certain antibiotics and we may not be appropriately treating these patients with broad-spectrum antibiotics or are perhaps using antibiotics that are not as effective based on an individual patient scenario so this is definitely a user-friendly tool that hopefully can be deployed throughout other health care systems to improve the treatment of pneumonia overall,” Dr. Balls emphasized.
A version of this article first appeared on Medscape.com.
a 3-year, pragmatic, cluster-controlled study shows.
“We designed the ePNa specifically to require minimal input from the clinician so everything it does is already in the electronic medical record,” Nathan Dean, MD, University of Utah, Salt Lake City, told this news organization.
“So it’s actually putting the guideline recommendations into effect for physicians so that they can make better decisions by having all this information – it’s a comprehensive best practice kind of tool where best practices are likely to make the biggest difference for patients with a high severity of illness,” he added.
The study was published online in the American Journal of Respiratory and Critical Care Medicine.
Guideline-based tool
The ePNa makes use of pneumonia guidelines of 2007 and 2019 from the American Thoracic Society/Infectious Disease Society of America. The system was deployed into six geographic clusters of 16 Intermountain hospital EDs at 2-month intervals between December 2017 and November 2018. Simultaneous deployment was impractical, as implementation of the tool takes education, monitoring, and feedback that can be facilitated by focusing on only a few hospitals at a time.
The decision support tool gathers key patient indicators including age, fever, oxygen saturation, vital signs, and laboratory and chest imaging results to offer recommendations on care, including appropriate antibiotic therapy, microbiology studies, and whether a given patient should be sent to the intensive care unit, admitted to hospital, or may safely be discharged home.
Investigators analyzed a total of 6,848 patients, of whom 4,536 were managed for pneumonia before the ePNa was deployed and 2,312 after deployment.
The median age of patients was 67 years (interquartile range, 50-79 years). Roughly half were female and almost all were White. “Observed 30-day all-cause mortality including both outpatients and inpatients was 8.6% before deployment versus 4.8% after deployment of ePNa,” Dr. Dean and colleagues reported.
Adjusted for severity of illness, the odds ratio for lower mortality post-ePNa launch was 0.62 (95% confidence interval, 0.49-0.79; P < .0010) “and lower morality was consistent across hospital clusters.”
Compared with patients who were discharged home, reductions in mortality were greatest in patients who were directly admitted to ICUs from the ED (OR, 0.32; 95% CI, 0.14-0.77; P = .01). The OR for patients admitted to the medical floor was 0.53 (95% CI, 0.25-1.1; P = .09), which did not reach statistical significance.
Dr. Dean explained that the reductions in mortality were seen among those with the most severe illness, in whom best practices would benefit the most. In contrast, patients who are sent home on an antibiotic are at low risk for mortality while patients admitted to the medical floor may well have another, more lethal illness from which they end up dying, rather than simple pneumonia.
“For me, this was a clear demonstration that these best practices made the biggest difference in patients who were sick and who did not have any underlying disease that was going to kill them anyway,” he emphasized. On the other hand, both 30-day mortality and 7-day secondary hospital admission were higher among patients the tool recommended for hospital ward admission but who were discharged home from the ED.
“This was an unexpected finding,” Dr. Dean observed. However, as he explained, the authors reviewed 25% of randomly selected patients who fell into this subgroup and discovered that the ePNa tool was used in only about 20% of patients – “so doctors did not use the tool in the majority of this group.”
In addition, some of these patients declined hospital admission, so the doctors may have recommended that they be admitted but the patients said no. “The hypothesis here is that if they had been admitted to the hospital, they may have had a lower mortality risk,” Dr. Dean said.
Noticeable changes
Another noticeable change following the introduction of the ePNa tool was that guideline-concordant antibiotic prescribing increased in the 8 hours after patients presented to the ED, from 79.5% prior to the tool’s launch to 87.9%, again after adjusting for pneumonia severity (P < .001). Use of broad-spectrum antibiotics was not significantly different between the two treatment intervals, but administration of antibiotics active against methicillin-resistant Staphylococcus aureus dropped significantly between the two treatment intervals (P < .001). And the mean time from admission to the ED to the first antibiotic taken was slightly faster, improving from 159.4 minutes (95% CI, 156.9-161.9 minutes) prior to the ePNa launch to 150.9 minutes (95% CI, 144.1-157.8) post deployment (P < .001).
“Overall outpatient disposition for treatment of pneumonia from the emergency department increased from 29.2% before ePNa to 46.9% [post ePNA],” the authors noted, while a similar increase was observed in patients for whom ePNA recommended outpatient care – from 49.2% pre-ePNA to 66.6% after ePNA.
Both hospital ward admission and admission to the ICU decreased after ePNa had been introduced. Despite a significant increase in the percentage of patients being discharged home, neither 7-day secondary hospital admission nor severity-adjusted, 30-day mortality were significantly different before versus after the introduction of ePNa, the authors stressed.
A limitation of the study was that the trial was confined to a single health care system in one region of the United States with a patient population that may differ from that in other regions.
Reason for its success
Asked to comment on the findings, Adam Balls, MD, emergency department chair, Intermountain Medical Center, Murray, Utah, suggested that the reason the ePNa tool has been so successful at improving care for pneumonia patients is that it puts the guidelines directly into the hands of individual providers and tells them what’s going on. (Dr. Balls was not involved in the study.) “The tool allows us to take into consideration various clinical features – a patient’s oxygen requirements and whether or not they had prior complicated pneumonias that required additional antibiotics, for example – and then it makes the best determination for not only the disposition for that patient but antibiotic treatment as well,” he said in an interview.
This then allows physicians to either appropriately discharge less severely ill patients and admit those who are more ill – “and in general, just do a better job of treating pneumonia with this tool,” Dr. Balls said. He himself uses the decision support tool when attending to his own patients with pneumonia, as he feels that the tool really does make his care of these patients better. “There is a disparity around how we treat pneumonia in the U.S.
“Clinicians sometimes have a bias or a preference for certain antibiotics and we may not be appropriately treating these patients with broad-spectrum antibiotics or are perhaps using antibiotics that are not as effective based on an individual patient scenario so this is definitely a user-friendly tool that hopefully can be deployed throughout other health care systems to improve the treatment of pneumonia overall,” Dr. Balls emphasized.
A version of this article first appeared on Medscape.com.
a 3-year, pragmatic, cluster-controlled study shows.
“We designed the ePNa specifically to require minimal input from the clinician so everything it does is already in the electronic medical record,” Nathan Dean, MD, University of Utah, Salt Lake City, told this news organization.
“So it’s actually putting the guideline recommendations into effect for physicians so that they can make better decisions by having all this information – it’s a comprehensive best practice kind of tool where best practices are likely to make the biggest difference for patients with a high severity of illness,” he added.
The study was published online in the American Journal of Respiratory and Critical Care Medicine.
Guideline-based tool
The ePNa makes use of pneumonia guidelines of 2007 and 2019 from the American Thoracic Society/Infectious Disease Society of America. The system was deployed into six geographic clusters of 16 Intermountain hospital EDs at 2-month intervals between December 2017 and November 2018. Simultaneous deployment was impractical, as implementation of the tool takes education, monitoring, and feedback that can be facilitated by focusing on only a few hospitals at a time.
The decision support tool gathers key patient indicators including age, fever, oxygen saturation, vital signs, and laboratory and chest imaging results to offer recommendations on care, including appropriate antibiotic therapy, microbiology studies, and whether a given patient should be sent to the intensive care unit, admitted to hospital, or may safely be discharged home.
Investigators analyzed a total of 6,848 patients, of whom 4,536 were managed for pneumonia before the ePNa was deployed and 2,312 after deployment.
The median age of patients was 67 years (interquartile range, 50-79 years). Roughly half were female and almost all were White. “Observed 30-day all-cause mortality including both outpatients and inpatients was 8.6% before deployment versus 4.8% after deployment of ePNa,” Dr. Dean and colleagues reported.
Adjusted for severity of illness, the odds ratio for lower mortality post-ePNa launch was 0.62 (95% confidence interval, 0.49-0.79; P < .0010) “and lower morality was consistent across hospital clusters.”
Compared with patients who were discharged home, reductions in mortality were greatest in patients who were directly admitted to ICUs from the ED (OR, 0.32; 95% CI, 0.14-0.77; P = .01). The OR for patients admitted to the medical floor was 0.53 (95% CI, 0.25-1.1; P = .09), which did not reach statistical significance.
Dr. Dean explained that the reductions in mortality were seen among those with the most severe illness, in whom best practices would benefit the most. In contrast, patients who are sent home on an antibiotic are at low risk for mortality while patients admitted to the medical floor may well have another, more lethal illness from which they end up dying, rather than simple pneumonia.
“For me, this was a clear demonstration that these best practices made the biggest difference in patients who were sick and who did not have any underlying disease that was going to kill them anyway,” he emphasized. On the other hand, both 30-day mortality and 7-day secondary hospital admission were higher among patients the tool recommended for hospital ward admission but who were discharged home from the ED.
“This was an unexpected finding,” Dr. Dean observed. However, as he explained, the authors reviewed 25% of randomly selected patients who fell into this subgroup and discovered that the ePNa tool was used in only about 20% of patients – “so doctors did not use the tool in the majority of this group.”
In addition, some of these patients declined hospital admission, so the doctors may have recommended that they be admitted but the patients said no. “The hypothesis here is that if they had been admitted to the hospital, they may have had a lower mortality risk,” Dr. Dean said.
Noticeable changes
Another noticeable change following the introduction of the ePNa tool was that guideline-concordant antibiotic prescribing increased in the 8 hours after patients presented to the ED, from 79.5% prior to the tool’s launch to 87.9%, again after adjusting for pneumonia severity (P < .001). Use of broad-spectrum antibiotics was not significantly different between the two treatment intervals, but administration of antibiotics active against methicillin-resistant Staphylococcus aureus dropped significantly between the two treatment intervals (P < .001). And the mean time from admission to the ED to the first antibiotic taken was slightly faster, improving from 159.4 minutes (95% CI, 156.9-161.9 minutes) prior to the ePNa launch to 150.9 minutes (95% CI, 144.1-157.8) post deployment (P < .001).
“Overall outpatient disposition for treatment of pneumonia from the emergency department increased from 29.2% before ePNa to 46.9% [post ePNA],” the authors noted, while a similar increase was observed in patients for whom ePNA recommended outpatient care – from 49.2% pre-ePNA to 66.6% after ePNA.
Both hospital ward admission and admission to the ICU decreased after ePNa had been introduced. Despite a significant increase in the percentage of patients being discharged home, neither 7-day secondary hospital admission nor severity-adjusted, 30-day mortality were significantly different before versus after the introduction of ePNa, the authors stressed.
A limitation of the study was that the trial was confined to a single health care system in one region of the United States with a patient population that may differ from that in other regions.
Reason for its success
Asked to comment on the findings, Adam Balls, MD, emergency department chair, Intermountain Medical Center, Murray, Utah, suggested that the reason the ePNa tool has been so successful at improving care for pneumonia patients is that it puts the guidelines directly into the hands of individual providers and tells them what’s going on. (Dr. Balls was not involved in the study.) “The tool allows us to take into consideration various clinical features – a patient’s oxygen requirements and whether or not they had prior complicated pneumonias that required additional antibiotics, for example – and then it makes the best determination for not only the disposition for that patient but antibiotic treatment as well,” he said in an interview.
This then allows physicians to either appropriately discharge less severely ill patients and admit those who are more ill – “and in general, just do a better job of treating pneumonia with this tool,” Dr. Balls said. He himself uses the decision support tool when attending to his own patients with pneumonia, as he feels that the tool really does make his care of these patients better. “There is a disparity around how we treat pneumonia in the U.S.
“Clinicians sometimes have a bias or a preference for certain antibiotics and we may not be appropriately treating these patients with broad-spectrum antibiotics or are perhaps using antibiotics that are not as effective based on an individual patient scenario so this is definitely a user-friendly tool that hopefully can be deployed throughout other health care systems to improve the treatment of pneumonia overall,” Dr. Balls emphasized.
A version of this article first appeared on Medscape.com.
FROM THE AMERICAN JOURNAL OF RESPIRATORY AND CRITICAL CARE MEDICINE
Are ‘antibiotic diets’ good practice?
Antibiotics are among the most commonly prescribed medications in both outpatient and hospital settings. Global efforts at curbing antibiotic-resistant strains have prompted clinicians to pursue better stewardship, whereby they limit their prescribing of such medications to those who truly need them.
Yet there’s another possible means of addressing antibiotic resistance – using dietary interventions to reduce the gastrointestinal complaints that so often accompany the use of antibiotics: vomiting, nausea, diarrhea, bloating/indigestion, abdominal pain, and loss of appetite. Far from being a mere nuisance, these complications can have major ramifications.
“Often [these side effects] will result in people stopping an antibiotic they need or taking a second- or third-generation one, which potentially causes even more problems with resistance,” explained Daniel J. Merenstein, MD, a professor of family medicine at Georgetown University, Washington, who has conducted multiple trials on antibiotic stewardship and probiotics.
And it’s not just clinicians who would like to find a way around these common complaints. On lifestyle blogs such as Livestrong and Goop, medical news websites, and via academic institutions, patients can learn how the right probiotic supplement or certain fermented or high-fiber foods, for example, might spare them the upset stomach that often accompanies a course of antibiotics.
Yet according to experts in the field, there are notable questions to be answered about whether there’s evidence to support this approach.
The cost of GI discomfort
Pinpointing the exact manner by which antibiotics upend GI tracts is a complex task, according to Gail Cresci, PhD, RD, a microbiome researcher in the department of pediatric gastroenterology, hepatology, and nutrition and director of nutrition research at the Cleveland Clinic.
“A lot of different mechanisms can go awry,” Dr. Cresci said. “But normally, the good microbes are there to help keep the balance, so when that balance is gone, the pathogens can take over and lead to this disruption.”
According to Lynne V. McFarland, PhD, a recently retired infectious disease epidemiologist who now serves as a private consultant, such complications can have substantial ramifications for patients and health care systems.
“If you’re an inpatient and develop antibiotic-associated diarrhea, it usually increases the length of stay from 8 to 20 days. It also increases the cost of care. And small children who have this can get severely dehydrated, which can become life threatening.”
Proponents of probiotics
Several of the researchers who were interviewed believe there’s convincing evidence supporting probiotics for the treatment of common antibiotic-related GI complaints. In many instances, they were involved in the studies themselves.
During Dr. McFarland’s 4-decade career in probiotics research, she has participated in early animal studies with strains such as Saccharomyces boulardii and was involved in meta-analyses of their role in Clostridioides difficile infection and associated diarrhea and even as a potential GI intervention for COVID-19 patients (Antibiotics (Basel). 2021 Apr 9;10(4):408).
In mouse model studies from 2013 and 2018, Dr. Cresci and colleagues showed that the probiotic strains Lactobacillus GG and Faecalibacterium prausnitzii reduced the structural gut changes that lead to antibiotic-associated diarrhea and minimized the risk of C. difficile infection.
In a 2021 randomized controlled trial led by Merenstein, healthy participants were given a trial of amoxicillin/clavulanate (days 1-7) in conjunction with either yogurt containing the probiotic Bifidobacterium animalis subsp lactis BB-12 or control yogurt (days 1-14). After assessing feces samples over a 30-day period, they found that those who received the probiotic yogurt had a significantly smaller decrease in short-chain fatty acid levels and a more stable taxonomic microbiota profile than control persons.
Dr. Merenstein said that, on the basis of results such as these and others, he’s comfortable being relatively definitive about the value of probiotics.
“I believe it’s close to standard of care that if you’re prescribing antibiotics, especially for more than 7 days, you really need to put people on probiotics that have been studied, simply because the evidence is robust enough now,” he said.
Even for proponents, there are caveats
However, all the researchers recommending the use of probiotics did so with caveats. First and foremost, they advise clinicians that the term “probiotics” is an imprecise catchall and is essentially meaningless.
“A lot of products label themselves as probiotic. It’s a great marketing scheme, but many of the products out there aren’t really probiotics; they’re not proven with randomized control trials and don’t have the scientific background,” said Dr. McFarland. “We’ve found that the efficacy is extremely strain specific and disease specific. A strain may work for one disease and not work for another.”
In 2018, Dr. McFarland coauthored an evidence-based practical guide to help clinicians and patients identify the specific strain that works in certain indications. Dr. Cresci recommends that clinicians consult websites such as Probiotics.org or the National Institutes of Health’s database to find the strains that have been proven to work in well-designed clinical trials.
There was also agreement that, to date, the most robust data support probiotics for the treatment of antibiotic-associated diarrhea.
Although the optimal timing of probiotics is a subject of debate, most proponents agreed that the general rule is “the sooner the better.”
Dr. McFarland recommended incorporating probiotics within 24 hours of starting an antibiotic “because the damage done to your GI tract microbiome is pretty quick, and the probiotics work best if they’re established before major disruption occurs.” She added that patients should continue taking probiotics for 2-8 weeks after stopping antibiotics.
“It takes a long time for your normal flora to get restored,” she said. “It’s best to cover your bases.”
For others, the evidence is not definitive
Opinions on the value of probiotics to combat antibiotic-related GI side effects are divergent, though.
“I would not recommend the routine use of probiotics, and certainly not in the prevention of C. difficile or antibiotic-related diarrhea,” said David A. Johnson, MD, professor of medicine and chief of gastroenterology at Eastern Virginia Medical School, Norfolk. “I think the evidence does not support that, and I stand strongly on that recommendation.”
Dr. Johnson cited the 2020 guidelines from the American Gastroenterological Association, which offer only a conditional recommendation for the use of specific probiotics and only in preventing antibiotic-associated C. difficile infection.
Geoffrey A. Preidis, MD, PhD, an assistant professor of pediatrics in the section of gastroenterology, hepatology, and nutrition at Baylor College of Medicine, Houston, served as a coauthor of the AGA’s guidelines. He noted that after reviewing 39 published trials of approximately 10,000 patients given probiotics while receiving antibiotics, the authors “did find some evidence that specific probiotics might decrease the risk of C. difficile diarrhea, but the quality of that evidence was low.”
Dr. Preidis attributed this to the lack of well-designed multicenter trials that can isolate the effects of certain strains and determine their benefit in this application.
“The majority of published trials have not reported safety data as rigorously as these data are reported in pharmaceutical trials, so the risk of side effects could be higher than we think,” said Dr. Preidis. “As living microbes, probiotics can move from the intestines into the bloodstream, causing sepsis. Contamination in the manufacturing process has been reported. There might be other long-term effects that we are not yet aware of.”
When asked to characterize the available data on probiotics, Dr. Johnson replied, “I’d generally label it, ‘caveat emptor.’ ”
Dr. McFarland agreed that the field would benefit from better-designed studies and called out meta-analyses that pool outcomes with various strains for particular criticism.
“When researchers do that, it’s no longer valid and shouldn’t have been published, in my opinion,” she told this news organization.
‘Antibiotic diets’ as a possible approach
A compromise between the two sides may be the so-called antibiotic diet. The theory behind such diets is that foods and beverages with biome-boosting properties may be a risk-free intervention that patients can adopt to alleviate antibiotic-related side effects.
“You want your diet to include more soluble fibers to help support the good bacteria, particularly when you’re taking antibiotics,” said Dr. Cresci. “You can get this through eating fresh fruits and vegetables, whole grains, and foods that have more prebiotic, like potatoes. You can also eat fermented food, such as kefir, kombucha, kimchi, and yogurt, so you’re adding more beneficial bacteria into your intestinal tract.”
There is ample published evidence that such foods can boost microbiome diversity and decrease inflammation, including a July 2021 study in Cell. However, the protection this may confer while taking antibiotics isn’t known. Establishing a clear role for the efficacy of such interventions is made additionally difficult by the well-established limitations of conducting dietary clinical trials.
Dr. Merenstein said that there is no compelling evidence that antibiotic-related complications can be offset by changing what goes onto our dinner plates. He joked: “We can’t say, ‘Here’s amoxicillin for your ear infection, now make sure you increase your fermented food, fiber, and water.’ ”
Dr. Johnson said he’s intrigued by studies of prebiotics – fibers that boost beneficial bacteria in the GI system.
“I would love to have more findings about prebiotic identification; that is, things we could do in a healthy way to keep the gut balanced while it’s subject to a change with antibiotics,” he said. “We’re just not there yet.”
Dr. Johnson added that he generally recommends that patients taking antibiotics eat “a bland diet, avoiding things that may have been provocative in the past.”
If patients are already enjoying foods with microbiome-boosting reputations, Dr. Johnson sees “very little downside to continuing that [while on antibiotics].” However, he noted that the period in which you’re taking antibiotics isn’t ideal for trying new foods, given the lack of experience with how the gut bacteria will react.
There are data about foods to avoid while taking antibiotics, which generally fall in line with common dietary knowledge. Many patients may know not to drink grapefruit juice with certain medications, but it’s worth a reminder. Certain antibiotics may also require delaying or avoiding dairy products, although this does not apply to yogurt.
A fiber-deficient diet can aggravate microbiota collapse following antibiotics. In a 2020 study, researchers showed that people on a high-fat diet who were using antibiotics were 8.6 times more likely to have preinflammatory bowel disease than those eating low-fat foods and who had no recent history of antibiotic use. Mouse model data from the same study indicated that poor diet and antibiotics may have worked in conjunction to reduce oxygen in the gut.
Dr. McFarland noted that building a healthy microbiome is a lifelong pursuit and that several factors (for example, environmental or genetic) are out of individuals’ hands. The general public might want a quick fix – ironically, one of the main drivers behind their requesting and receiving antibiotics when they’re not indicated – but it’s likely not available to them.
“You can’t eat one salad and suddenly have a healthy gut, unfortunately.”
A version of this article first appeared on Medscape.com.
Antibiotics are among the most commonly prescribed medications in both outpatient and hospital settings. Global efforts at curbing antibiotic-resistant strains have prompted clinicians to pursue better stewardship, whereby they limit their prescribing of such medications to those who truly need them.
Yet there’s another possible means of addressing antibiotic resistance – using dietary interventions to reduce the gastrointestinal complaints that so often accompany the use of antibiotics: vomiting, nausea, diarrhea, bloating/indigestion, abdominal pain, and loss of appetite. Far from being a mere nuisance, these complications can have major ramifications.
“Often [these side effects] will result in people stopping an antibiotic they need or taking a second- or third-generation one, which potentially causes even more problems with resistance,” explained Daniel J. Merenstein, MD, a professor of family medicine at Georgetown University, Washington, who has conducted multiple trials on antibiotic stewardship and probiotics.
And it’s not just clinicians who would like to find a way around these common complaints. On lifestyle blogs such as Livestrong and Goop, medical news websites, and via academic institutions, patients can learn how the right probiotic supplement or certain fermented or high-fiber foods, for example, might spare them the upset stomach that often accompanies a course of antibiotics.
Yet according to experts in the field, there are notable questions to be answered about whether there’s evidence to support this approach.
The cost of GI discomfort
Pinpointing the exact manner by which antibiotics upend GI tracts is a complex task, according to Gail Cresci, PhD, RD, a microbiome researcher in the department of pediatric gastroenterology, hepatology, and nutrition and director of nutrition research at the Cleveland Clinic.
“A lot of different mechanisms can go awry,” Dr. Cresci said. “But normally, the good microbes are there to help keep the balance, so when that balance is gone, the pathogens can take over and lead to this disruption.”
According to Lynne V. McFarland, PhD, a recently retired infectious disease epidemiologist who now serves as a private consultant, such complications can have substantial ramifications for patients and health care systems.
“If you’re an inpatient and develop antibiotic-associated diarrhea, it usually increases the length of stay from 8 to 20 days. It also increases the cost of care. And small children who have this can get severely dehydrated, which can become life threatening.”
Proponents of probiotics
Several of the researchers who were interviewed believe there’s convincing evidence supporting probiotics for the treatment of common antibiotic-related GI complaints. In many instances, they were involved in the studies themselves.
During Dr. McFarland’s 4-decade career in probiotics research, she has participated in early animal studies with strains such as Saccharomyces boulardii and was involved in meta-analyses of their role in Clostridioides difficile infection and associated diarrhea and even as a potential GI intervention for COVID-19 patients (Antibiotics (Basel). 2021 Apr 9;10(4):408).
In mouse model studies from 2013 and 2018, Dr. Cresci and colleagues showed that the probiotic strains Lactobacillus GG and Faecalibacterium prausnitzii reduced the structural gut changes that lead to antibiotic-associated diarrhea and minimized the risk of C. difficile infection.
In a 2021 randomized controlled trial led by Merenstein, healthy participants were given a trial of amoxicillin/clavulanate (days 1-7) in conjunction with either yogurt containing the probiotic Bifidobacterium animalis subsp lactis BB-12 or control yogurt (days 1-14). After assessing feces samples over a 30-day period, they found that those who received the probiotic yogurt had a significantly smaller decrease in short-chain fatty acid levels and a more stable taxonomic microbiota profile than control persons.
Dr. Merenstein said that, on the basis of results such as these and others, he’s comfortable being relatively definitive about the value of probiotics.
“I believe it’s close to standard of care that if you’re prescribing antibiotics, especially for more than 7 days, you really need to put people on probiotics that have been studied, simply because the evidence is robust enough now,” he said.
Even for proponents, there are caveats
However, all the researchers recommending the use of probiotics did so with caveats. First and foremost, they advise clinicians that the term “probiotics” is an imprecise catchall and is essentially meaningless.
“A lot of products label themselves as probiotic. It’s a great marketing scheme, but many of the products out there aren’t really probiotics; they’re not proven with randomized control trials and don’t have the scientific background,” said Dr. McFarland. “We’ve found that the efficacy is extremely strain specific and disease specific. A strain may work for one disease and not work for another.”
In 2018, Dr. McFarland coauthored an evidence-based practical guide to help clinicians and patients identify the specific strain that works in certain indications. Dr. Cresci recommends that clinicians consult websites such as Probiotics.org or the National Institutes of Health’s database to find the strains that have been proven to work in well-designed clinical trials.
There was also agreement that, to date, the most robust data support probiotics for the treatment of antibiotic-associated diarrhea.
Although the optimal timing of probiotics is a subject of debate, most proponents agreed that the general rule is “the sooner the better.”
Dr. McFarland recommended incorporating probiotics within 24 hours of starting an antibiotic “because the damage done to your GI tract microbiome is pretty quick, and the probiotics work best if they’re established before major disruption occurs.” She added that patients should continue taking probiotics for 2-8 weeks after stopping antibiotics.
“It takes a long time for your normal flora to get restored,” she said. “It’s best to cover your bases.”
For others, the evidence is not definitive
Opinions on the value of probiotics to combat antibiotic-related GI side effects are divergent, though.
“I would not recommend the routine use of probiotics, and certainly not in the prevention of C. difficile or antibiotic-related diarrhea,” said David A. Johnson, MD, professor of medicine and chief of gastroenterology at Eastern Virginia Medical School, Norfolk. “I think the evidence does not support that, and I stand strongly on that recommendation.”
Dr. Johnson cited the 2020 guidelines from the American Gastroenterological Association, which offer only a conditional recommendation for the use of specific probiotics and only in preventing antibiotic-associated C. difficile infection.
Geoffrey A. Preidis, MD, PhD, an assistant professor of pediatrics in the section of gastroenterology, hepatology, and nutrition at Baylor College of Medicine, Houston, served as a coauthor of the AGA’s guidelines. He noted that after reviewing 39 published trials of approximately 10,000 patients given probiotics while receiving antibiotics, the authors “did find some evidence that specific probiotics might decrease the risk of C. difficile diarrhea, but the quality of that evidence was low.”
Dr. Preidis attributed this to the lack of well-designed multicenter trials that can isolate the effects of certain strains and determine their benefit in this application.
“The majority of published trials have not reported safety data as rigorously as these data are reported in pharmaceutical trials, so the risk of side effects could be higher than we think,” said Dr. Preidis. “As living microbes, probiotics can move from the intestines into the bloodstream, causing sepsis. Contamination in the manufacturing process has been reported. There might be other long-term effects that we are not yet aware of.”
When asked to characterize the available data on probiotics, Dr. Johnson replied, “I’d generally label it, ‘caveat emptor.’ ”
Dr. McFarland agreed that the field would benefit from better-designed studies and called out meta-analyses that pool outcomes with various strains for particular criticism.
“When researchers do that, it’s no longer valid and shouldn’t have been published, in my opinion,” she told this news organization.
‘Antibiotic diets’ as a possible approach
A compromise between the two sides may be the so-called antibiotic diet. The theory behind such diets is that foods and beverages with biome-boosting properties may be a risk-free intervention that patients can adopt to alleviate antibiotic-related side effects.
“You want your diet to include more soluble fibers to help support the good bacteria, particularly when you’re taking antibiotics,” said Dr. Cresci. “You can get this through eating fresh fruits and vegetables, whole grains, and foods that have more prebiotic, like potatoes. You can also eat fermented food, such as kefir, kombucha, kimchi, and yogurt, so you’re adding more beneficial bacteria into your intestinal tract.”
There is ample published evidence that such foods can boost microbiome diversity and decrease inflammation, including a July 2021 study in Cell. However, the protection this may confer while taking antibiotics isn’t known. Establishing a clear role for the efficacy of such interventions is made additionally difficult by the well-established limitations of conducting dietary clinical trials.
Dr. Merenstein said that there is no compelling evidence that antibiotic-related complications can be offset by changing what goes onto our dinner plates. He joked: “We can’t say, ‘Here’s amoxicillin for your ear infection, now make sure you increase your fermented food, fiber, and water.’ ”
Dr. Johnson said he’s intrigued by studies of prebiotics – fibers that boost beneficial bacteria in the GI system.
“I would love to have more findings about prebiotic identification; that is, things we could do in a healthy way to keep the gut balanced while it’s subject to a change with antibiotics,” he said. “We’re just not there yet.”
Dr. Johnson added that he generally recommends that patients taking antibiotics eat “a bland diet, avoiding things that may have been provocative in the past.”
If patients are already enjoying foods with microbiome-boosting reputations, Dr. Johnson sees “very little downside to continuing that [while on antibiotics].” However, he noted that the period in which you’re taking antibiotics isn’t ideal for trying new foods, given the lack of experience with how the gut bacteria will react.
There are data about foods to avoid while taking antibiotics, which generally fall in line with common dietary knowledge. Many patients may know not to drink grapefruit juice with certain medications, but it’s worth a reminder. Certain antibiotics may also require delaying or avoiding dairy products, although this does not apply to yogurt.
A fiber-deficient diet can aggravate microbiota collapse following antibiotics. In a 2020 study, researchers showed that people on a high-fat diet who were using antibiotics were 8.6 times more likely to have preinflammatory bowel disease than those eating low-fat foods and who had no recent history of antibiotic use. Mouse model data from the same study indicated that poor diet and antibiotics may have worked in conjunction to reduce oxygen in the gut.
Dr. McFarland noted that building a healthy microbiome is a lifelong pursuit and that several factors (for example, environmental or genetic) are out of individuals’ hands. The general public might want a quick fix – ironically, one of the main drivers behind their requesting and receiving antibiotics when they’re not indicated – but it’s likely not available to them.
“You can’t eat one salad and suddenly have a healthy gut, unfortunately.”
A version of this article first appeared on Medscape.com.
Antibiotics are among the most commonly prescribed medications in both outpatient and hospital settings. Global efforts at curbing antibiotic-resistant strains have prompted clinicians to pursue better stewardship, whereby they limit their prescribing of such medications to those who truly need them.
Yet there’s another possible means of addressing antibiotic resistance – using dietary interventions to reduce the gastrointestinal complaints that so often accompany the use of antibiotics: vomiting, nausea, diarrhea, bloating/indigestion, abdominal pain, and loss of appetite. Far from being a mere nuisance, these complications can have major ramifications.
“Often [these side effects] will result in people stopping an antibiotic they need or taking a second- or third-generation one, which potentially causes even more problems with resistance,” explained Daniel J. Merenstein, MD, a professor of family medicine at Georgetown University, Washington, who has conducted multiple trials on antibiotic stewardship and probiotics.
And it’s not just clinicians who would like to find a way around these common complaints. On lifestyle blogs such as Livestrong and Goop, medical news websites, and via academic institutions, patients can learn how the right probiotic supplement or certain fermented or high-fiber foods, for example, might spare them the upset stomach that often accompanies a course of antibiotics.
Yet according to experts in the field, there are notable questions to be answered about whether there’s evidence to support this approach.
The cost of GI discomfort
Pinpointing the exact manner by which antibiotics upend GI tracts is a complex task, according to Gail Cresci, PhD, RD, a microbiome researcher in the department of pediatric gastroenterology, hepatology, and nutrition and director of nutrition research at the Cleveland Clinic.
“A lot of different mechanisms can go awry,” Dr. Cresci said. “But normally, the good microbes are there to help keep the balance, so when that balance is gone, the pathogens can take over and lead to this disruption.”
According to Lynne V. McFarland, PhD, a recently retired infectious disease epidemiologist who now serves as a private consultant, such complications can have substantial ramifications for patients and health care systems.
“If you’re an inpatient and develop antibiotic-associated diarrhea, it usually increases the length of stay from 8 to 20 days. It also increases the cost of care. And small children who have this can get severely dehydrated, which can become life threatening.”
Proponents of probiotics
Several of the researchers who were interviewed believe there’s convincing evidence supporting probiotics for the treatment of common antibiotic-related GI complaints. In many instances, they were involved in the studies themselves.
During Dr. McFarland’s 4-decade career in probiotics research, she has participated in early animal studies with strains such as Saccharomyces boulardii and was involved in meta-analyses of their role in Clostridioides difficile infection and associated diarrhea and even as a potential GI intervention for COVID-19 patients (Antibiotics (Basel). 2021 Apr 9;10(4):408).
In mouse model studies from 2013 and 2018, Dr. Cresci and colleagues showed that the probiotic strains Lactobacillus GG and Faecalibacterium prausnitzii reduced the structural gut changes that lead to antibiotic-associated diarrhea and minimized the risk of C. difficile infection.
In a 2021 randomized controlled trial led by Merenstein, healthy participants were given a trial of amoxicillin/clavulanate (days 1-7) in conjunction with either yogurt containing the probiotic Bifidobacterium animalis subsp lactis BB-12 or control yogurt (days 1-14). After assessing feces samples over a 30-day period, they found that those who received the probiotic yogurt had a significantly smaller decrease in short-chain fatty acid levels and a more stable taxonomic microbiota profile than control persons.
Dr. Merenstein said that, on the basis of results such as these and others, he’s comfortable being relatively definitive about the value of probiotics.
“I believe it’s close to standard of care that if you’re prescribing antibiotics, especially for more than 7 days, you really need to put people on probiotics that have been studied, simply because the evidence is robust enough now,” he said.
Even for proponents, there are caveats
However, all the researchers recommending the use of probiotics did so with caveats. First and foremost, they advise clinicians that the term “probiotics” is an imprecise catchall and is essentially meaningless.
“A lot of products label themselves as probiotic. It’s a great marketing scheme, but many of the products out there aren’t really probiotics; they’re not proven with randomized control trials and don’t have the scientific background,” said Dr. McFarland. “We’ve found that the efficacy is extremely strain specific and disease specific. A strain may work for one disease and not work for another.”
In 2018, Dr. McFarland coauthored an evidence-based practical guide to help clinicians and patients identify the specific strain that works in certain indications. Dr. Cresci recommends that clinicians consult websites such as Probiotics.org or the National Institutes of Health’s database to find the strains that have been proven to work in well-designed clinical trials.
There was also agreement that, to date, the most robust data support probiotics for the treatment of antibiotic-associated diarrhea.
Although the optimal timing of probiotics is a subject of debate, most proponents agreed that the general rule is “the sooner the better.”
Dr. McFarland recommended incorporating probiotics within 24 hours of starting an antibiotic “because the damage done to your GI tract microbiome is pretty quick, and the probiotics work best if they’re established before major disruption occurs.” She added that patients should continue taking probiotics for 2-8 weeks after stopping antibiotics.
“It takes a long time for your normal flora to get restored,” she said. “It’s best to cover your bases.”
For others, the evidence is not definitive
Opinions on the value of probiotics to combat antibiotic-related GI side effects are divergent, though.
“I would not recommend the routine use of probiotics, and certainly not in the prevention of C. difficile or antibiotic-related diarrhea,” said David A. Johnson, MD, professor of medicine and chief of gastroenterology at Eastern Virginia Medical School, Norfolk. “I think the evidence does not support that, and I stand strongly on that recommendation.”
Dr. Johnson cited the 2020 guidelines from the American Gastroenterological Association, which offer only a conditional recommendation for the use of specific probiotics and only in preventing antibiotic-associated C. difficile infection.
Geoffrey A. Preidis, MD, PhD, an assistant professor of pediatrics in the section of gastroenterology, hepatology, and nutrition at Baylor College of Medicine, Houston, served as a coauthor of the AGA’s guidelines. He noted that after reviewing 39 published trials of approximately 10,000 patients given probiotics while receiving antibiotics, the authors “did find some evidence that specific probiotics might decrease the risk of C. difficile diarrhea, but the quality of that evidence was low.”
Dr. Preidis attributed this to the lack of well-designed multicenter trials that can isolate the effects of certain strains and determine their benefit in this application.
“The majority of published trials have not reported safety data as rigorously as these data are reported in pharmaceutical trials, so the risk of side effects could be higher than we think,” said Dr. Preidis. “As living microbes, probiotics can move from the intestines into the bloodstream, causing sepsis. Contamination in the manufacturing process has been reported. There might be other long-term effects that we are not yet aware of.”
When asked to characterize the available data on probiotics, Dr. Johnson replied, “I’d generally label it, ‘caveat emptor.’ ”
Dr. McFarland agreed that the field would benefit from better-designed studies and called out meta-analyses that pool outcomes with various strains for particular criticism.
“When researchers do that, it’s no longer valid and shouldn’t have been published, in my opinion,” she told this news organization.
‘Antibiotic diets’ as a possible approach
A compromise between the two sides may be the so-called antibiotic diet. The theory behind such diets is that foods and beverages with biome-boosting properties may be a risk-free intervention that patients can adopt to alleviate antibiotic-related side effects.
“You want your diet to include more soluble fibers to help support the good bacteria, particularly when you’re taking antibiotics,” said Dr. Cresci. “You can get this through eating fresh fruits and vegetables, whole grains, and foods that have more prebiotic, like potatoes. You can also eat fermented food, such as kefir, kombucha, kimchi, and yogurt, so you’re adding more beneficial bacteria into your intestinal tract.”
There is ample published evidence that such foods can boost microbiome diversity and decrease inflammation, including a July 2021 study in Cell. However, the protection this may confer while taking antibiotics isn’t known. Establishing a clear role for the efficacy of such interventions is made additionally difficult by the well-established limitations of conducting dietary clinical trials.
Dr. Merenstein said that there is no compelling evidence that antibiotic-related complications can be offset by changing what goes onto our dinner plates. He joked: “We can’t say, ‘Here’s amoxicillin for your ear infection, now make sure you increase your fermented food, fiber, and water.’ ”
Dr. Johnson said he’s intrigued by studies of prebiotics – fibers that boost beneficial bacteria in the GI system.
“I would love to have more findings about prebiotic identification; that is, things we could do in a healthy way to keep the gut balanced while it’s subject to a change with antibiotics,” he said. “We’re just not there yet.”
Dr. Johnson added that he generally recommends that patients taking antibiotics eat “a bland diet, avoiding things that may have been provocative in the past.”
If patients are already enjoying foods with microbiome-boosting reputations, Dr. Johnson sees “very little downside to continuing that [while on antibiotics].” However, he noted that the period in which you’re taking antibiotics isn’t ideal for trying new foods, given the lack of experience with how the gut bacteria will react.
There are data about foods to avoid while taking antibiotics, which generally fall in line with common dietary knowledge. Many patients may know not to drink grapefruit juice with certain medications, but it’s worth a reminder. Certain antibiotics may also require delaying or avoiding dairy products, although this does not apply to yogurt.
A fiber-deficient diet can aggravate microbiota collapse following antibiotics. In a 2020 study, researchers showed that people on a high-fat diet who were using antibiotics were 8.6 times more likely to have preinflammatory bowel disease than those eating low-fat foods and who had no recent history of antibiotic use. Mouse model data from the same study indicated that poor diet and antibiotics may have worked in conjunction to reduce oxygen in the gut.
Dr. McFarland noted that building a healthy microbiome is a lifelong pursuit and that several factors (for example, environmental or genetic) are out of individuals’ hands. The general public might want a quick fix – ironically, one of the main drivers behind their requesting and receiving antibiotics when they’re not indicated – but it’s likely not available to them.
“You can’t eat one salad and suddenly have a healthy gut, unfortunately.”
A version of this article first appeared on Medscape.com.



