User login
Enlarged Facial Pores: An Update on Treatments
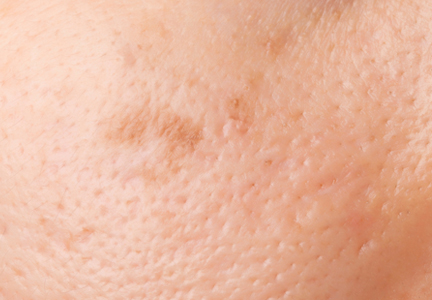
Enlarged facial pores are superficial skin structures that are visualized as small openings on the skin corresponding to the openings of the pilosebaceous apparatus. These openings may be impacted with horny follicular plugs consisting of sebaceous debris that appear as open comedones.1 Skin pores is a lay term that is poorly defined in the medical literature and often is categorized in terms of arbitrary circular diameters determined through cosmetic skin analyzers.2 The term refers to pilosebaceous follicular enlargements (with or without open comedonal horny impactions) that can be visualized by the naked eye, most commonly occurring on the face and scalp. These enlarged pores remain a pervasive cosmetic concern that impacts patient quality of life. Enlarged pores are difficult to treat, in part due to lack of knowledge of the pathophysiology; thus, we review the currently proposed causes of enlarged pilosebaceous openings and the treatments in the scope of this pathogenesis with a focus on therapeutic efficacy.
Pathogenesis of Enlarged Facial Pores
It is now thought that seborrhea, loss of skin elasticity and tension, and hair follicle size are most clinically relevant to the pathogenesis of enlarged pores.2 Other potential associated and causative factors include genetic predisposition, acne, comedogenic xenobiotics, chronic photodamage, chronic radiodermatitis, and vitamin A deficiency.1,3
The direct relationship between sebum output and pore size has been well established, particularly in men who generally have higher sebum output levels than women, which likely is testosterone driven.4,5 However, there are contradictory data on whether sex affects pore size, as females also exhibit contributory hormonal factors. Sebum output and pore size increase substantially during the ovulation phase of the female menstrual cycle, likely secondary to increased progesterone affecting sebaceous gland activity.2,4 The presence of acne also is associated with enlarged facial pores, though the extent of seborrhea as a confounding factor is unclear. Furthermore, acne severity does not correlate with increased pore size.5 However, the processes of acne and facial pores are interlinked, given the frequent occurrence of open comedones within the pores.
Skin elasticity and tensile strength when defined visually and mechanically has shown a negative correlation with facial pore size and density.5 It is well known that cutaneous aging and chronic photodamage cause perturbation in the collagen and elastin framework that allows for the skin to maintain its resilient properties.6 Aged and photodamaged skin also demonstrates decreased expression of microfibril-associated glycoprotein-1 (MAGP-1), a crucial component in elastic fiber assembly and skin elasticity in the dermis and perifollicular/pore areas.7
Pore density and size appears to range diversely across ethnicities, though Chinese women exhibit notably lower pore size and density across all ages as compared to other ethnicities.8 Black individuals have aberrant epidermal architecture, defined as the presence of stalagmitelike structures at the dermoepidermal junction, correlating with enlarged pore size compared to other ethnicities.2,8
Treating Enlarged Facial Pores
Treatments for enlarged facial pores primarily aim to decrease sebum production, rejuvenate skin, remove hair, and/or decrease follicular size. Evidence-based studies are limited, and many currently used therapies have not been studied with enlarged facial pores as a primary investigative outcome. Here, we include studies that report efficacy in decreasing pore size specifically. It is important to note the lack of a uniform and objective modality with which to report skin pore size. Studies use a wide range of techniques including patient self-reporting, physician observation, and software image analyzers.
Topical Therapies
Topical retinoids are vitamin A derivatives, and they are first-line therapies in reversing the aberrant collagen and elastin-associated epidermal and dermal changes that occur with chronological aging and photoaging. Tretinoin, isotretinoin, and tazarotene have shown efficacy in multiple parameters of skin rejuvenation, including facial pores, skin wrinkling, hyperpigmentation, skin laxity, and sebum production.9 However, it is important to note that retinoids treat keratinocyte atypia in acne, and efficacy in facial pores is confounded by improvement in follicular keratinization. Because studies have not distinctly uncoupled this association, it is erroneous to conclude that retinoids reduce facial pore size and density irrespective of concomitant acne vulgaris.
Tazarotene has been evaluated for use in reducing facial pore size. In one investigation, 568 patients with moderate wrinkling or hyperpigmentation were randomized to receive tazarotene cream 0.1% or placebo once daily for 24 weeks and were evaluated for enlarged facial pores as a secondary outcome using a double-blinded physician 5-point scale.10 At week 24, 42% of tazarotene-treated patients achieved improvement of at least 1 point compared to 20% of placebo-treated patients (P<.001). Adverse events were dermatitic, as can be expected of retinoids, leading to a 4% discontinuation rate in the tazarotene group compared to 1% in the placebo group.10
Tretinoin has long been used off label for antiaging treatments but has only recently shown efficacy for facial pores. In one study, 60 women who had previously sought antiaging procedures were treated with tretinoin cream 0.025% once daily and no other antiaging products or procedures for 90 days.11 Facial pore evaluations were determined by a modified dermatoscope with a polarized analyzer for clinical scoring using a photonumeric scale. Patients improved from a baseline average score of 3.2 in facial pores to a posttreatment average score of 2.0 (P<.05) at day 84. This improvement was sustained from day 28 of treatment and corresponded to patient self-perception. Adverse events included xerosis, desquamation, burning, and erythema, which led to 3 premature discontinuations.11
Various chemical peel formulations are used in skin rejuvenation and have shown application in enlarged facial pores. Chemical peels act at the epidermal or dermal level to induce temporary breakdown and regeneration of healthier cells and improved skin matrix.12 Twenty-two Japanese women applied glycolic acid (30% solution) every 2 weeks for a total of 5 treatments and exhibited reduced appearance of conspicuous, open, and dark pores, defined by surface area and shading as determined through dermatoscopic and software analysis, with mean improvement rates of 34.6%, 11%, and 34.3%, respectively. More than 70% of participants exhibited improvement in enlarged facial pores.13 A study involving a 40% glycolic acid and vitamin C formulation demonstrated significant improvement in facial pores (28.3%; P<.001).14
The newest topical therapies studied for use in minimizing facial pilosebaceous openings are natural plant-derived copper chlorophyllin complex sodium salt (CHLcu) and tetra-hydro-jasmonic acid (LR2412). Clinical trials of these botanicals are limited with small sample sizes but are included here as novel treatments requiring further investigation.
Chlorophyllin copper complex sodium salt is derived from chlorophyll, a green pigment found in plants, and has been investigated as a topical gel in liposomal dispersions for application in photodamaged and aged skin. Chlorophyllin copper complex sodium salt exerts in vitro hyaluronidase inhibitory activity to maintain hyaluronic acid in the extracellular matrix and counteract the structural breakdown of cutaneous aging.15 Two small single-center pilot trials enrolled 10 participants each in a 3-week study of CHLcu 0.1% twice daily and an 8-week study of CHLcu 0.066% twice daily.16,17 After 3 weeks, patients treated with CHLcu 0.1% exhibited a 22.2% improvement in facial pores by clinical assessment grading, though this improvement was not significant on software imaging analysis. Patients improved the most on parameters of facial seborrhea by clinical assessment.16 After 8 weeks, patients treated with CHLcu 0.066% exhibited 25.3% improvement in facial pores by clinical assessment grading.17 Treatments were reported to be well tolerated without noted adverse events in both studies.
Tetra-hydro-jasmonic acid is an analogue of jasmonic acid, a plant hormone derived from linoleic acid. Due to its favorable safety profile and bioavailability, penetration into epidermal and dermal layers, and potential effects in rejuvenating desquamation, LR2412 is currently being assessed for treatment of skin wrinkles, texture, and pores.18 Its effect is thought to relate to stimulation of laminin-5, collagen IV, and fibrillin deposition at the dermoepidermal junction.19 In an open-label trial of a topical preparation of LR2412, 15 participants were treated twice daily for 6 weeks and assessed through investigator clinical assessment scoring.20 Investigator scoring of pores improved by 25.2% from baseline (P<.05) after 6 weeks of treatment. Improvement in pores was seen as early as days 1 and 3. No serious adverse events were reported, though 2 participants developed acne on follow-up.20
Tetra-hydro-jasmonic acid also is formulated with retinol (retinol 0.2%/LR2412 2.0%) and demonstrated cosmetic efficacy in a noninferiority trial with tretinoin cream 0.025%.11 Sixty patients each were randomized to retinol/LR2412 or tretinoin at bedtime and treated for 90 days. At day 84, participants in the retinol/LR2412 group exhibited an improvement in investigator clinical assessment scoring from a baseline of 3.6 to 2.5 (P<.05). There were no significant differences in investigator-assessed efficacy between the treatment arms. Participants reported similar or better results and fewer side effects with retinol/LR2412 on self-questionnaires. Eight participants treated with retinol/LR2412 and 15 participants treated with tretinoin reported various incidences of skin irritation, burning, and desquamation.11
Oral Therapies
The most commonly used oral therapies for enlarged pores are antiandrogens, such as combined oral contraceptives, spironolactone, and cyproterone acetate, which modulate sebum production due to the presence of androgen receptors within sebaceous glands.21 Forty-four white women in an open-label, phase 4 study were treated with combined oral contraceptives containing chlormadinone acetate–ethinyl estradiol for 6 menstrual cycles, with standardized photography taken before and after the treatment period for software analysis. After 6 treatment cycles, 9.1% (4/44) of participants had visibly enlarged pores of the forehead and cheeks compared to 43.2% (19/44) of participants at baseline (P<.0001).22 The effects of other antiandrogens on facial pores have not been studied in this capacity.
Lasers, Radiofrequency, and Ultrasound Devices
The development of various devices that can deliver targeted thermal or ultrasound energy to the skin offers the newest and most robust modality in cosmetic therapy. The mechanism of their efficacy may be due to a combination of induced remodeling of collagen fibers near pilosebaceous openings to increase skin elasticity and decrease sebum production.2,23
Devices with established antiaging effects have been extensively reviewed and include the gold particle 800-nm diode laser, 1450-nm diode laser, microneedle apparatuses, fractional radiofrequency devices, 2790-nm erbium:YAG laser, nonablative 1410-nm fractionated erbium-doped fiber laser, and nonablative 1440-nm fractional laser.2
Literature on the use of these devices for minimizing facial pore size is limited. One treatment of intense focused ultrasound using a 3-mm transducer successfully improved overall pore appearance in 91% of sites at 6-week follow-up on a clinical grading scale.24 Three sessions of nonablative 1410-nm fractionated erbium-doped fiber laser treatments yielded facial skin pore minimization of greater than 51% in 14 of 15 participants.25
The nonablative 1440-nm diode fractional laser received 510(k) clearance by the US Food and Drug Administration in 2011 for aesthetic use in chronologically aged and photoaged skin. Twenty participants treated for 2 weeks and a total of 6 facial treatments with this laser system showed a 17% average improvement in facial pore score on software analysis (P≤.002). Adverse events were mild and included erythema and xerosis.26
Conclusion
The reliability of available literature on efficacy of various treatments in diminishing facial skin pores has been challenging given that most studies are low in power, lack control groups, use nonuniform methods of reporting outcomes, and do not report complete adverse events. Thus, all results should be interpreted with caution.
Overall, it is clear that the pathogenesis of enlarged facial pores is multifactorial and complex, necessitating a similar approach to therapeutics. Topical treatments offer a range of diverse therapies with proven benefit in facial pore reduction. The advent of lasers and devices offers constantly evolving therapeutic options with diffuse antiaging effects. Despite the numerous topical, oral, and device-oriented options, enlarged facial pores remain a challenging cosmetic concern. More robust efficacy studies on new treatments are necessary.
- Uhoda E, Pierard-Franchimont C, Petit L, et al. The conundrum of skin pores in dermocosmetology. Dermatology. 2005;210:3-7.
- Lee SJ, Seok J, Jeong SY, et al. Facial pores: definition, causes, and treatment options. Dermatol Surg. 2016;42:277-285.
- Pierard GE, Pierard-Franchimont C, Marks R, et al. EEMCO guidance for the in vivo assessment of skin greasiness. The EEMCO Group. Skin Pharmacol Appl Skin Physiol. 2000;13:372-389.
- Roh M, Han M, Kim D, et al. Sebum output as a factor contributing to the size of facial pores. Br J Dermatol. 2006;155:890-894.
- Kim BY, Choi JW, Park KC, et al. Sebum, acne, skin elasticity, and gender difference-which is the major influencing factor for facial pores? Skin Res Technol. 2013;19:E45-E53.
- Uitto J. The role of elastin and collagen in cutaneous aging: intrinsic aging versus photoexposure. J Drugs Dermatol. 2008;7(2 suppl):S12-S16.
- Zheng Q, Chen S, Chen Y, et al. Investigation of age-related decline of microfibril-associated glycoprotein-1 in human skin through immunohistochemistry study. Clin Cosmet Investig Dermatol. 2013;6:317-323.
- Sugiyama-Nakagiri Y, Sugata K, Hachiya A, et al. Ethnic differences in the structural properties of facial skin. J Dermatol Sci. 2009;53:135-139.
- Mukherjee S, Date A, Patravale V, et al. Retinoids in the treatment of skin aging: an overview of clinical efficacy and safety. Clin Interv Aging. 2006;1:327-348.
- Kang S, Krueger GG, Tanghetti EA, et al; Tazarotene Cream in Photodamage Study Group. A multicenter, randomized, double-blind trial of tazarotene 0.1% cream in the treatment of photodamage. J Am Acad Dermatol. 2005;52:268-274.
- Bouloc A, Vergnanini AL, Issa MC. A double-blind randomized study comparing the association of retinol and LR2412 with tretinoin 0.025% in photoaged skin. J Cosmet Dermatol. 2015;14:40-46.
- Fischer TC, Perosino E, Poli F, et al. Chemical peels in aesthetic dermatology: an update 2009 [published online September 8, 2009]. J Eur Acad Dermatol Venereol. 2010;24:281-292.
- Kakudo N, Kushida S, Tanaka N, et al. A novel method to measure conspicuous facial pores using computer analysis of digital-camera-captured images: the effect of glycolic acid chemical peeling. Skin Res Technol. 2011;17:427-433.
- Kim WS. Efficacy and safety of a new superficial chemical peel using alpha-hydroxy acid, vitamin C and oxygen for melasma. J Cosmet Laser Ther. 2013;15:21-24.
- McCook JP, Dorogi PL, Vasily DB, et al. In vitro inhibition of hyaluronidase by sodium copper chlorophyllin complex and chlorophyllin analogs. Clin Cosmet Investig Dermatol. 2015;8:443-448.
- Stephens TJ, McCook JP, Herndon JH Jr. Pilot study of topical copper chlorophyllin complex in subjects with facial acne and large pores. J Drugs Dermatol. 2015;14:589-592.
- Sigler ML, Stephens TJ. Assessment of the safety and efficacy of topical copper chlorophyllin in women with photodamaged facial skin. J Drugs Dermatol. 2015;14:401-404.
- Alexiades M. Jasmonates and tetrahydrojasmonic acid: a novel class of anti-aging molecules. J Drugs Dermatol. 2016;15:206-207.
- Tran C, Michelet JF, Simonetti L, et al. In vitro and in vivo studies with tetra-hydro-jasmonic acid (LR2412) reveal its potential to correct signs of skin ageing. J Eur Acad Dermatol Venereol. 2014;28:415-423.
- Alexiades M. Clinical assessment of a novel jasmonate cosmeceutical, LR2412-Cx, for the treatment of skin aging. J Drugs Dermatol. 2016;15:209-215.
- Lam C, Zaenglein AL. Contraceptive use in acne. Clin Dermatol. 2014;32:502-515.
- Kerscher M, Reuther T, Bayrhammer J, et al. Effects of an oral contraceptive containing chlormadinone and ethinylestradiol on acne-prone skin of women of different age groups: an open-label, single-centre, phase IV study. Clin Drug Investig. 2008;28:703-711.
- Schmults CD, Phelps R, Goldberg DJ. Nonablative facial remodeling: erythema reduction and histologic evidence of new collagen formation using a 300-microsecond 1064-nm Nd:YAG laser. Arch Dermatol. 2004;140:1373-1376.
- Lee HJ, Lee KR, Park JY, et al. The efficacy and safety of intense focused ultrasound in the treatment of enlarged facial pores in Asian skin. J Dermatolog Treat. 2015;26:73-77.
- Suh DH, Chang KY, Lee SJ, et al. Treatment of dilated pores with 1410-nm fractional erbium-doped fiber laser. Lasers Med Sci. 2015;30:1135-1139.
- Saedi N, Petrell K, Arndt K, et al. Evaluating facial pores and skin texture after low-energy nonablative fractional 1440-nm laser treatments. J Am Acad Dermatol. 2013;68:113-118.
Enlarged facial pores are superficial skin structures that are visualized as small openings on the skin corresponding to the openings of the pilosebaceous apparatus. These openings may be impacted with horny follicular plugs consisting of sebaceous debris that appear as open comedones.1 Skin pores is a lay term that is poorly defined in the medical literature and often is categorized in terms of arbitrary circular diameters determined through cosmetic skin analyzers.2 The term refers to pilosebaceous follicular enlargements (with or without open comedonal horny impactions) that can be visualized by the naked eye, most commonly occurring on the face and scalp. These enlarged pores remain a pervasive cosmetic concern that impacts patient quality of life. Enlarged pores are difficult to treat, in part due to lack of knowledge of the pathophysiology; thus, we review the currently proposed causes of enlarged pilosebaceous openings and the treatments in the scope of this pathogenesis with a focus on therapeutic efficacy.
Pathogenesis of Enlarged Facial Pores
It is now thought that seborrhea, loss of skin elasticity and tension, and hair follicle size are most clinically relevant to the pathogenesis of enlarged pores.2 Other potential associated and causative factors include genetic predisposition, acne, comedogenic xenobiotics, chronic photodamage, chronic radiodermatitis, and vitamin A deficiency.1,3
The direct relationship between sebum output and pore size has been well established, particularly in men who generally have higher sebum output levels than women, which likely is testosterone driven.4,5 However, there are contradictory data on whether sex affects pore size, as females also exhibit contributory hormonal factors. Sebum output and pore size increase substantially during the ovulation phase of the female menstrual cycle, likely secondary to increased progesterone affecting sebaceous gland activity.2,4 The presence of acne also is associated with enlarged facial pores, though the extent of seborrhea as a confounding factor is unclear. Furthermore, acne severity does not correlate with increased pore size.5 However, the processes of acne and facial pores are interlinked, given the frequent occurrence of open comedones within the pores.
Skin elasticity and tensile strength when defined visually and mechanically has shown a negative correlation with facial pore size and density.5 It is well known that cutaneous aging and chronic photodamage cause perturbation in the collagen and elastin framework that allows for the skin to maintain its resilient properties.6 Aged and photodamaged skin also demonstrates decreased expression of microfibril-associated glycoprotein-1 (MAGP-1), a crucial component in elastic fiber assembly and skin elasticity in the dermis and perifollicular/pore areas.7
Pore density and size appears to range diversely across ethnicities, though Chinese women exhibit notably lower pore size and density across all ages as compared to other ethnicities.8 Black individuals have aberrant epidermal architecture, defined as the presence of stalagmitelike structures at the dermoepidermal junction, correlating with enlarged pore size compared to other ethnicities.2,8
Treating Enlarged Facial Pores
Treatments for enlarged facial pores primarily aim to decrease sebum production, rejuvenate skin, remove hair, and/or decrease follicular size. Evidence-based studies are limited, and many currently used therapies have not been studied with enlarged facial pores as a primary investigative outcome. Here, we include studies that report efficacy in decreasing pore size specifically. It is important to note the lack of a uniform and objective modality with which to report skin pore size. Studies use a wide range of techniques including patient self-reporting, physician observation, and software image analyzers.
Topical Therapies
Topical retinoids are vitamin A derivatives, and they are first-line therapies in reversing the aberrant collagen and elastin-associated epidermal and dermal changes that occur with chronological aging and photoaging. Tretinoin, isotretinoin, and tazarotene have shown efficacy in multiple parameters of skin rejuvenation, including facial pores, skin wrinkling, hyperpigmentation, skin laxity, and sebum production.9 However, it is important to note that retinoids treat keratinocyte atypia in acne, and efficacy in facial pores is confounded by improvement in follicular keratinization. Because studies have not distinctly uncoupled this association, it is erroneous to conclude that retinoids reduce facial pore size and density irrespective of concomitant acne vulgaris.
Tazarotene has been evaluated for use in reducing facial pore size. In one investigation, 568 patients with moderate wrinkling or hyperpigmentation were randomized to receive tazarotene cream 0.1% or placebo once daily for 24 weeks and were evaluated for enlarged facial pores as a secondary outcome using a double-blinded physician 5-point scale.10 At week 24, 42% of tazarotene-treated patients achieved improvement of at least 1 point compared to 20% of placebo-treated patients (P<.001). Adverse events were dermatitic, as can be expected of retinoids, leading to a 4% discontinuation rate in the tazarotene group compared to 1% in the placebo group.10
Tretinoin has long been used off label for antiaging treatments but has only recently shown efficacy for facial pores. In one study, 60 women who had previously sought antiaging procedures were treated with tretinoin cream 0.025% once daily and no other antiaging products or procedures for 90 days.11 Facial pore evaluations were determined by a modified dermatoscope with a polarized analyzer for clinical scoring using a photonumeric scale. Patients improved from a baseline average score of 3.2 in facial pores to a posttreatment average score of 2.0 (P<.05) at day 84. This improvement was sustained from day 28 of treatment and corresponded to patient self-perception. Adverse events included xerosis, desquamation, burning, and erythema, which led to 3 premature discontinuations.11
Various chemical peel formulations are used in skin rejuvenation and have shown application in enlarged facial pores. Chemical peels act at the epidermal or dermal level to induce temporary breakdown and regeneration of healthier cells and improved skin matrix.12 Twenty-two Japanese women applied glycolic acid (30% solution) every 2 weeks for a total of 5 treatments and exhibited reduced appearance of conspicuous, open, and dark pores, defined by surface area and shading as determined through dermatoscopic and software analysis, with mean improvement rates of 34.6%, 11%, and 34.3%, respectively. More than 70% of participants exhibited improvement in enlarged facial pores.13 A study involving a 40% glycolic acid and vitamin C formulation demonstrated significant improvement in facial pores (28.3%; P<.001).14
The newest topical therapies studied for use in minimizing facial pilosebaceous openings are natural plant-derived copper chlorophyllin complex sodium salt (CHLcu) and tetra-hydro-jasmonic acid (LR2412). Clinical trials of these botanicals are limited with small sample sizes but are included here as novel treatments requiring further investigation.
Chlorophyllin copper complex sodium salt is derived from chlorophyll, a green pigment found in plants, and has been investigated as a topical gel in liposomal dispersions for application in photodamaged and aged skin. Chlorophyllin copper complex sodium salt exerts in vitro hyaluronidase inhibitory activity to maintain hyaluronic acid in the extracellular matrix and counteract the structural breakdown of cutaneous aging.15 Two small single-center pilot trials enrolled 10 participants each in a 3-week study of CHLcu 0.1% twice daily and an 8-week study of CHLcu 0.066% twice daily.16,17 After 3 weeks, patients treated with CHLcu 0.1% exhibited a 22.2% improvement in facial pores by clinical assessment grading, though this improvement was not significant on software imaging analysis. Patients improved the most on parameters of facial seborrhea by clinical assessment.16 After 8 weeks, patients treated with CHLcu 0.066% exhibited 25.3% improvement in facial pores by clinical assessment grading.17 Treatments were reported to be well tolerated without noted adverse events in both studies.
Tetra-hydro-jasmonic acid is an analogue of jasmonic acid, a plant hormone derived from linoleic acid. Due to its favorable safety profile and bioavailability, penetration into epidermal and dermal layers, and potential effects in rejuvenating desquamation, LR2412 is currently being assessed for treatment of skin wrinkles, texture, and pores.18 Its effect is thought to relate to stimulation of laminin-5, collagen IV, and fibrillin deposition at the dermoepidermal junction.19 In an open-label trial of a topical preparation of LR2412, 15 participants were treated twice daily for 6 weeks and assessed through investigator clinical assessment scoring.20 Investigator scoring of pores improved by 25.2% from baseline (P<.05) after 6 weeks of treatment. Improvement in pores was seen as early as days 1 and 3. No serious adverse events were reported, though 2 participants developed acne on follow-up.20
Tetra-hydro-jasmonic acid also is formulated with retinol (retinol 0.2%/LR2412 2.0%) and demonstrated cosmetic efficacy in a noninferiority trial with tretinoin cream 0.025%.11 Sixty patients each were randomized to retinol/LR2412 or tretinoin at bedtime and treated for 90 days. At day 84, participants in the retinol/LR2412 group exhibited an improvement in investigator clinical assessment scoring from a baseline of 3.6 to 2.5 (P<.05). There were no significant differences in investigator-assessed efficacy between the treatment arms. Participants reported similar or better results and fewer side effects with retinol/LR2412 on self-questionnaires. Eight participants treated with retinol/LR2412 and 15 participants treated with tretinoin reported various incidences of skin irritation, burning, and desquamation.11
Oral Therapies
The most commonly used oral therapies for enlarged pores are antiandrogens, such as combined oral contraceptives, spironolactone, and cyproterone acetate, which modulate sebum production due to the presence of androgen receptors within sebaceous glands.21 Forty-four white women in an open-label, phase 4 study were treated with combined oral contraceptives containing chlormadinone acetate–ethinyl estradiol for 6 menstrual cycles, with standardized photography taken before and after the treatment period for software analysis. After 6 treatment cycles, 9.1% (4/44) of participants had visibly enlarged pores of the forehead and cheeks compared to 43.2% (19/44) of participants at baseline (P<.0001).22 The effects of other antiandrogens on facial pores have not been studied in this capacity.
Lasers, Radiofrequency, and Ultrasound Devices
The development of various devices that can deliver targeted thermal or ultrasound energy to the skin offers the newest and most robust modality in cosmetic therapy. The mechanism of their efficacy may be due to a combination of induced remodeling of collagen fibers near pilosebaceous openings to increase skin elasticity and decrease sebum production.2,23
Devices with established antiaging effects have been extensively reviewed and include the gold particle 800-nm diode laser, 1450-nm diode laser, microneedle apparatuses, fractional radiofrequency devices, 2790-nm erbium:YAG laser, nonablative 1410-nm fractionated erbium-doped fiber laser, and nonablative 1440-nm fractional laser.2
Literature on the use of these devices for minimizing facial pore size is limited. One treatment of intense focused ultrasound using a 3-mm transducer successfully improved overall pore appearance in 91% of sites at 6-week follow-up on a clinical grading scale.24 Three sessions of nonablative 1410-nm fractionated erbium-doped fiber laser treatments yielded facial skin pore minimization of greater than 51% in 14 of 15 participants.25
The nonablative 1440-nm diode fractional laser received 510(k) clearance by the US Food and Drug Administration in 2011 for aesthetic use in chronologically aged and photoaged skin. Twenty participants treated for 2 weeks and a total of 6 facial treatments with this laser system showed a 17% average improvement in facial pore score on software analysis (P≤.002). Adverse events were mild and included erythema and xerosis.26
Conclusion
The reliability of available literature on efficacy of various treatments in diminishing facial skin pores has been challenging given that most studies are low in power, lack control groups, use nonuniform methods of reporting outcomes, and do not report complete adverse events. Thus, all results should be interpreted with caution.
Overall, it is clear that the pathogenesis of enlarged facial pores is multifactorial and complex, necessitating a similar approach to therapeutics. Topical treatments offer a range of diverse therapies with proven benefit in facial pore reduction. The advent of lasers and devices offers constantly evolving therapeutic options with diffuse antiaging effects. Despite the numerous topical, oral, and device-oriented options, enlarged facial pores remain a challenging cosmetic concern. More robust efficacy studies on new treatments are necessary.
Enlarged facial pores are superficial skin structures that are visualized as small openings on the skin corresponding to the openings of the pilosebaceous apparatus. These openings may be impacted with horny follicular plugs consisting of sebaceous debris that appear as open comedones.1 Skin pores is a lay term that is poorly defined in the medical literature and often is categorized in terms of arbitrary circular diameters determined through cosmetic skin analyzers.2 The term refers to pilosebaceous follicular enlargements (with or without open comedonal horny impactions) that can be visualized by the naked eye, most commonly occurring on the face and scalp. These enlarged pores remain a pervasive cosmetic concern that impacts patient quality of life. Enlarged pores are difficult to treat, in part due to lack of knowledge of the pathophysiology; thus, we review the currently proposed causes of enlarged pilosebaceous openings and the treatments in the scope of this pathogenesis with a focus on therapeutic efficacy.
Pathogenesis of Enlarged Facial Pores
It is now thought that seborrhea, loss of skin elasticity and tension, and hair follicle size are most clinically relevant to the pathogenesis of enlarged pores.2 Other potential associated and causative factors include genetic predisposition, acne, comedogenic xenobiotics, chronic photodamage, chronic radiodermatitis, and vitamin A deficiency.1,3
The direct relationship between sebum output and pore size has been well established, particularly in men who generally have higher sebum output levels than women, which likely is testosterone driven.4,5 However, there are contradictory data on whether sex affects pore size, as females also exhibit contributory hormonal factors. Sebum output and pore size increase substantially during the ovulation phase of the female menstrual cycle, likely secondary to increased progesterone affecting sebaceous gland activity.2,4 The presence of acne also is associated with enlarged facial pores, though the extent of seborrhea as a confounding factor is unclear. Furthermore, acne severity does not correlate with increased pore size.5 However, the processes of acne and facial pores are interlinked, given the frequent occurrence of open comedones within the pores.
Skin elasticity and tensile strength when defined visually and mechanically has shown a negative correlation with facial pore size and density.5 It is well known that cutaneous aging and chronic photodamage cause perturbation in the collagen and elastin framework that allows for the skin to maintain its resilient properties.6 Aged and photodamaged skin also demonstrates decreased expression of microfibril-associated glycoprotein-1 (MAGP-1), a crucial component in elastic fiber assembly and skin elasticity in the dermis and perifollicular/pore areas.7
Pore density and size appears to range diversely across ethnicities, though Chinese women exhibit notably lower pore size and density across all ages as compared to other ethnicities.8 Black individuals have aberrant epidermal architecture, defined as the presence of stalagmitelike structures at the dermoepidermal junction, correlating with enlarged pore size compared to other ethnicities.2,8
Treating Enlarged Facial Pores
Treatments for enlarged facial pores primarily aim to decrease sebum production, rejuvenate skin, remove hair, and/or decrease follicular size. Evidence-based studies are limited, and many currently used therapies have not been studied with enlarged facial pores as a primary investigative outcome. Here, we include studies that report efficacy in decreasing pore size specifically. It is important to note the lack of a uniform and objective modality with which to report skin pore size. Studies use a wide range of techniques including patient self-reporting, physician observation, and software image analyzers.
Topical Therapies
Topical retinoids are vitamin A derivatives, and they are first-line therapies in reversing the aberrant collagen and elastin-associated epidermal and dermal changes that occur with chronological aging and photoaging. Tretinoin, isotretinoin, and tazarotene have shown efficacy in multiple parameters of skin rejuvenation, including facial pores, skin wrinkling, hyperpigmentation, skin laxity, and sebum production.9 However, it is important to note that retinoids treat keratinocyte atypia in acne, and efficacy in facial pores is confounded by improvement in follicular keratinization. Because studies have not distinctly uncoupled this association, it is erroneous to conclude that retinoids reduce facial pore size and density irrespective of concomitant acne vulgaris.
Tazarotene has been evaluated for use in reducing facial pore size. In one investigation, 568 patients with moderate wrinkling or hyperpigmentation were randomized to receive tazarotene cream 0.1% or placebo once daily for 24 weeks and were evaluated for enlarged facial pores as a secondary outcome using a double-blinded physician 5-point scale.10 At week 24, 42% of tazarotene-treated patients achieved improvement of at least 1 point compared to 20% of placebo-treated patients (P<.001). Adverse events were dermatitic, as can be expected of retinoids, leading to a 4% discontinuation rate in the tazarotene group compared to 1% in the placebo group.10
Tretinoin has long been used off label for antiaging treatments but has only recently shown efficacy for facial pores. In one study, 60 women who had previously sought antiaging procedures were treated with tretinoin cream 0.025% once daily and no other antiaging products or procedures for 90 days.11 Facial pore evaluations were determined by a modified dermatoscope with a polarized analyzer for clinical scoring using a photonumeric scale. Patients improved from a baseline average score of 3.2 in facial pores to a posttreatment average score of 2.0 (P<.05) at day 84. This improvement was sustained from day 28 of treatment and corresponded to patient self-perception. Adverse events included xerosis, desquamation, burning, and erythema, which led to 3 premature discontinuations.11
Various chemical peel formulations are used in skin rejuvenation and have shown application in enlarged facial pores. Chemical peels act at the epidermal or dermal level to induce temporary breakdown and regeneration of healthier cells and improved skin matrix.12 Twenty-two Japanese women applied glycolic acid (30% solution) every 2 weeks for a total of 5 treatments and exhibited reduced appearance of conspicuous, open, and dark pores, defined by surface area and shading as determined through dermatoscopic and software analysis, with mean improvement rates of 34.6%, 11%, and 34.3%, respectively. More than 70% of participants exhibited improvement in enlarged facial pores.13 A study involving a 40% glycolic acid and vitamin C formulation demonstrated significant improvement in facial pores (28.3%; P<.001).14
The newest topical therapies studied for use in minimizing facial pilosebaceous openings are natural plant-derived copper chlorophyllin complex sodium salt (CHLcu) and tetra-hydro-jasmonic acid (LR2412). Clinical trials of these botanicals are limited with small sample sizes but are included here as novel treatments requiring further investigation.
Chlorophyllin copper complex sodium salt is derived from chlorophyll, a green pigment found in plants, and has been investigated as a topical gel in liposomal dispersions for application in photodamaged and aged skin. Chlorophyllin copper complex sodium salt exerts in vitro hyaluronidase inhibitory activity to maintain hyaluronic acid in the extracellular matrix and counteract the structural breakdown of cutaneous aging.15 Two small single-center pilot trials enrolled 10 participants each in a 3-week study of CHLcu 0.1% twice daily and an 8-week study of CHLcu 0.066% twice daily.16,17 After 3 weeks, patients treated with CHLcu 0.1% exhibited a 22.2% improvement in facial pores by clinical assessment grading, though this improvement was not significant on software imaging analysis. Patients improved the most on parameters of facial seborrhea by clinical assessment.16 After 8 weeks, patients treated with CHLcu 0.066% exhibited 25.3% improvement in facial pores by clinical assessment grading.17 Treatments were reported to be well tolerated without noted adverse events in both studies.
Tetra-hydro-jasmonic acid is an analogue of jasmonic acid, a plant hormone derived from linoleic acid. Due to its favorable safety profile and bioavailability, penetration into epidermal and dermal layers, and potential effects in rejuvenating desquamation, LR2412 is currently being assessed for treatment of skin wrinkles, texture, and pores.18 Its effect is thought to relate to stimulation of laminin-5, collagen IV, and fibrillin deposition at the dermoepidermal junction.19 In an open-label trial of a topical preparation of LR2412, 15 participants were treated twice daily for 6 weeks and assessed through investigator clinical assessment scoring.20 Investigator scoring of pores improved by 25.2% from baseline (P<.05) after 6 weeks of treatment. Improvement in pores was seen as early as days 1 and 3. No serious adverse events were reported, though 2 participants developed acne on follow-up.20
Tetra-hydro-jasmonic acid also is formulated with retinol (retinol 0.2%/LR2412 2.0%) and demonstrated cosmetic efficacy in a noninferiority trial with tretinoin cream 0.025%.11 Sixty patients each were randomized to retinol/LR2412 or tretinoin at bedtime and treated for 90 days. At day 84, participants in the retinol/LR2412 group exhibited an improvement in investigator clinical assessment scoring from a baseline of 3.6 to 2.5 (P<.05). There were no significant differences in investigator-assessed efficacy between the treatment arms. Participants reported similar or better results and fewer side effects with retinol/LR2412 on self-questionnaires. Eight participants treated with retinol/LR2412 and 15 participants treated with tretinoin reported various incidences of skin irritation, burning, and desquamation.11
Oral Therapies
The most commonly used oral therapies for enlarged pores are antiandrogens, such as combined oral contraceptives, spironolactone, and cyproterone acetate, which modulate sebum production due to the presence of androgen receptors within sebaceous glands.21 Forty-four white women in an open-label, phase 4 study were treated with combined oral contraceptives containing chlormadinone acetate–ethinyl estradiol for 6 menstrual cycles, with standardized photography taken before and after the treatment period for software analysis. After 6 treatment cycles, 9.1% (4/44) of participants had visibly enlarged pores of the forehead and cheeks compared to 43.2% (19/44) of participants at baseline (P<.0001).22 The effects of other antiandrogens on facial pores have not been studied in this capacity.
Lasers, Radiofrequency, and Ultrasound Devices
The development of various devices that can deliver targeted thermal or ultrasound energy to the skin offers the newest and most robust modality in cosmetic therapy. The mechanism of their efficacy may be due to a combination of induced remodeling of collagen fibers near pilosebaceous openings to increase skin elasticity and decrease sebum production.2,23
Devices with established antiaging effects have been extensively reviewed and include the gold particle 800-nm diode laser, 1450-nm diode laser, microneedle apparatuses, fractional radiofrequency devices, 2790-nm erbium:YAG laser, nonablative 1410-nm fractionated erbium-doped fiber laser, and nonablative 1440-nm fractional laser.2
Literature on the use of these devices for minimizing facial pore size is limited. One treatment of intense focused ultrasound using a 3-mm transducer successfully improved overall pore appearance in 91% of sites at 6-week follow-up on a clinical grading scale.24 Three sessions of nonablative 1410-nm fractionated erbium-doped fiber laser treatments yielded facial skin pore minimization of greater than 51% in 14 of 15 participants.25
The nonablative 1440-nm diode fractional laser received 510(k) clearance by the US Food and Drug Administration in 2011 for aesthetic use in chronologically aged and photoaged skin. Twenty participants treated for 2 weeks and a total of 6 facial treatments with this laser system showed a 17% average improvement in facial pore score on software analysis (P≤.002). Adverse events were mild and included erythema and xerosis.26
Conclusion
The reliability of available literature on efficacy of various treatments in diminishing facial skin pores has been challenging given that most studies are low in power, lack control groups, use nonuniform methods of reporting outcomes, and do not report complete adverse events. Thus, all results should be interpreted with caution.
Overall, it is clear that the pathogenesis of enlarged facial pores is multifactorial and complex, necessitating a similar approach to therapeutics. Topical treatments offer a range of diverse therapies with proven benefit in facial pore reduction. The advent of lasers and devices offers constantly evolving therapeutic options with diffuse antiaging effects. Despite the numerous topical, oral, and device-oriented options, enlarged facial pores remain a challenging cosmetic concern. More robust efficacy studies on new treatments are necessary.
- Uhoda E, Pierard-Franchimont C, Petit L, et al. The conundrum of skin pores in dermocosmetology. Dermatology. 2005;210:3-7.
- Lee SJ, Seok J, Jeong SY, et al. Facial pores: definition, causes, and treatment options. Dermatol Surg. 2016;42:277-285.
- Pierard GE, Pierard-Franchimont C, Marks R, et al. EEMCO guidance for the in vivo assessment of skin greasiness. The EEMCO Group. Skin Pharmacol Appl Skin Physiol. 2000;13:372-389.
- Roh M, Han M, Kim D, et al. Sebum output as a factor contributing to the size of facial pores. Br J Dermatol. 2006;155:890-894.
- Kim BY, Choi JW, Park KC, et al. Sebum, acne, skin elasticity, and gender difference-which is the major influencing factor for facial pores? Skin Res Technol. 2013;19:E45-E53.
- Uitto J. The role of elastin and collagen in cutaneous aging: intrinsic aging versus photoexposure. J Drugs Dermatol. 2008;7(2 suppl):S12-S16.
- Zheng Q, Chen S, Chen Y, et al. Investigation of age-related decline of microfibril-associated glycoprotein-1 in human skin through immunohistochemistry study. Clin Cosmet Investig Dermatol. 2013;6:317-323.
- Sugiyama-Nakagiri Y, Sugata K, Hachiya A, et al. Ethnic differences in the structural properties of facial skin. J Dermatol Sci. 2009;53:135-139.
- Mukherjee S, Date A, Patravale V, et al. Retinoids in the treatment of skin aging: an overview of clinical efficacy and safety. Clin Interv Aging. 2006;1:327-348.
- Kang S, Krueger GG, Tanghetti EA, et al; Tazarotene Cream in Photodamage Study Group. A multicenter, randomized, double-blind trial of tazarotene 0.1% cream in the treatment of photodamage. J Am Acad Dermatol. 2005;52:268-274.
- Bouloc A, Vergnanini AL, Issa MC. A double-blind randomized study comparing the association of retinol and LR2412 with tretinoin 0.025% in photoaged skin. J Cosmet Dermatol. 2015;14:40-46.
- Fischer TC, Perosino E, Poli F, et al. Chemical peels in aesthetic dermatology: an update 2009 [published online September 8, 2009]. J Eur Acad Dermatol Venereol. 2010;24:281-292.
- Kakudo N, Kushida S, Tanaka N, et al. A novel method to measure conspicuous facial pores using computer analysis of digital-camera-captured images: the effect of glycolic acid chemical peeling. Skin Res Technol. 2011;17:427-433.
- Kim WS. Efficacy and safety of a new superficial chemical peel using alpha-hydroxy acid, vitamin C and oxygen for melasma. J Cosmet Laser Ther. 2013;15:21-24.
- McCook JP, Dorogi PL, Vasily DB, et al. In vitro inhibition of hyaluronidase by sodium copper chlorophyllin complex and chlorophyllin analogs. Clin Cosmet Investig Dermatol. 2015;8:443-448.
- Stephens TJ, McCook JP, Herndon JH Jr. Pilot study of topical copper chlorophyllin complex in subjects with facial acne and large pores. J Drugs Dermatol. 2015;14:589-592.
- Sigler ML, Stephens TJ. Assessment of the safety and efficacy of topical copper chlorophyllin in women with photodamaged facial skin. J Drugs Dermatol. 2015;14:401-404.
- Alexiades M. Jasmonates and tetrahydrojasmonic acid: a novel class of anti-aging molecules. J Drugs Dermatol. 2016;15:206-207.
- Tran C, Michelet JF, Simonetti L, et al. In vitro and in vivo studies with tetra-hydro-jasmonic acid (LR2412) reveal its potential to correct signs of skin ageing. J Eur Acad Dermatol Venereol. 2014;28:415-423.
- Alexiades M. Clinical assessment of a novel jasmonate cosmeceutical, LR2412-Cx, for the treatment of skin aging. J Drugs Dermatol. 2016;15:209-215.
- Lam C, Zaenglein AL. Contraceptive use in acne. Clin Dermatol. 2014;32:502-515.
- Kerscher M, Reuther T, Bayrhammer J, et al. Effects of an oral contraceptive containing chlormadinone and ethinylestradiol on acne-prone skin of women of different age groups: an open-label, single-centre, phase IV study. Clin Drug Investig. 2008;28:703-711.
- Schmults CD, Phelps R, Goldberg DJ. Nonablative facial remodeling: erythema reduction and histologic evidence of new collagen formation using a 300-microsecond 1064-nm Nd:YAG laser. Arch Dermatol. 2004;140:1373-1376.
- Lee HJ, Lee KR, Park JY, et al. The efficacy and safety of intense focused ultrasound in the treatment of enlarged facial pores in Asian skin. J Dermatolog Treat. 2015;26:73-77.
- Suh DH, Chang KY, Lee SJ, et al. Treatment of dilated pores with 1410-nm fractional erbium-doped fiber laser. Lasers Med Sci. 2015;30:1135-1139.
- Saedi N, Petrell K, Arndt K, et al. Evaluating facial pores and skin texture after low-energy nonablative fractional 1440-nm laser treatments. J Am Acad Dermatol. 2013;68:113-118.
- Uhoda E, Pierard-Franchimont C, Petit L, et al. The conundrum of skin pores in dermocosmetology. Dermatology. 2005;210:3-7.
- Lee SJ, Seok J, Jeong SY, et al. Facial pores: definition, causes, and treatment options. Dermatol Surg. 2016;42:277-285.
- Pierard GE, Pierard-Franchimont C, Marks R, et al. EEMCO guidance for the in vivo assessment of skin greasiness. The EEMCO Group. Skin Pharmacol Appl Skin Physiol. 2000;13:372-389.
- Roh M, Han M, Kim D, et al. Sebum output as a factor contributing to the size of facial pores. Br J Dermatol. 2006;155:890-894.
- Kim BY, Choi JW, Park KC, et al. Sebum, acne, skin elasticity, and gender difference-which is the major influencing factor for facial pores? Skin Res Technol. 2013;19:E45-E53.
- Uitto J. The role of elastin and collagen in cutaneous aging: intrinsic aging versus photoexposure. J Drugs Dermatol. 2008;7(2 suppl):S12-S16.
- Zheng Q, Chen S, Chen Y, et al. Investigation of age-related decline of microfibril-associated glycoprotein-1 in human skin through immunohistochemistry study. Clin Cosmet Investig Dermatol. 2013;6:317-323.
- Sugiyama-Nakagiri Y, Sugata K, Hachiya A, et al. Ethnic differences in the structural properties of facial skin. J Dermatol Sci. 2009;53:135-139.
- Mukherjee S, Date A, Patravale V, et al. Retinoids in the treatment of skin aging: an overview of clinical efficacy and safety. Clin Interv Aging. 2006;1:327-348.
- Kang S, Krueger GG, Tanghetti EA, et al; Tazarotene Cream in Photodamage Study Group. A multicenter, randomized, double-blind trial of tazarotene 0.1% cream in the treatment of photodamage. J Am Acad Dermatol. 2005;52:268-274.
- Bouloc A, Vergnanini AL, Issa MC. A double-blind randomized study comparing the association of retinol and LR2412 with tretinoin 0.025% in photoaged skin. J Cosmet Dermatol. 2015;14:40-46.
- Fischer TC, Perosino E, Poli F, et al. Chemical peels in aesthetic dermatology: an update 2009 [published online September 8, 2009]. J Eur Acad Dermatol Venereol. 2010;24:281-292.
- Kakudo N, Kushida S, Tanaka N, et al. A novel method to measure conspicuous facial pores using computer analysis of digital-camera-captured images: the effect of glycolic acid chemical peeling. Skin Res Technol. 2011;17:427-433.
- Kim WS. Efficacy and safety of a new superficial chemical peel using alpha-hydroxy acid, vitamin C and oxygen for melasma. J Cosmet Laser Ther. 2013;15:21-24.
- McCook JP, Dorogi PL, Vasily DB, et al. In vitro inhibition of hyaluronidase by sodium copper chlorophyllin complex and chlorophyllin analogs. Clin Cosmet Investig Dermatol. 2015;8:443-448.
- Stephens TJ, McCook JP, Herndon JH Jr. Pilot study of topical copper chlorophyllin complex in subjects with facial acne and large pores. J Drugs Dermatol. 2015;14:589-592.
- Sigler ML, Stephens TJ. Assessment of the safety and efficacy of topical copper chlorophyllin in women with photodamaged facial skin. J Drugs Dermatol. 2015;14:401-404.
- Alexiades M. Jasmonates and tetrahydrojasmonic acid: a novel class of anti-aging molecules. J Drugs Dermatol. 2016;15:206-207.
- Tran C, Michelet JF, Simonetti L, et al. In vitro and in vivo studies with tetra-hydro-jasmonic acid (LR2412) reveal its potential to correct signs of skin ageing. J Eur Acad Dermatol Venereol. 2014;28:415-423.
- Alexiades M. Clinical assessment of a novel jasmonate cosmeceutical, LR2412-Cx, for the treatment of skin aging. J Drugs Dermatol. 2016;15:209-215.
- Lam C, Zaenglein AL. Contraceptive use in acne. Clin Dermatol. 2014;32:502-515.
- Kerscher M, Reuther T, Bayrhammer J, et al. Effects of an oral contraceptive containing chlormadinone and ethinylestradiol on acne-prone skin of women of different age groups: an open-label, single-centre, phase IV study. Clin Drug Investig. 2008;28:703-711.
- Schmults CD, Phelps R, Goldberg DJ. Nonablative facial remodeling: erythema reduction and histologic evidence of new collagen formation using a 300-microsecond 1064-nm Nd:YAG laser. Arch Dermatol. 2004;140:1373-1376.
- Lee HJ, Lee KR, Park JY, et al. The efficacy and safety of intense focused ultrasound in the treatment of enlarged facial pores in Asian skin. J Dermatolog Treat. 2015;26:73-77.
- Suh DH, Chang KY, Lee SJ, et al. Treatment of dilated pores with 1410-nm fractional erbium-doped fiber laser. Lasers Med Sci. 2015;30:1135-1139.
- Saedi N, Petrell K, Arndt K, et al. Evaluating facial pores and skin texture after low-energy nonablative fractional 1440-nm laser treatments. J Am Acad Dermatol. 2013;68:113-118.
Practice Points
- The pathogenesis of enlarged facial pores is speculated to be associated with sebum production, skin aging and photodamage, and hair follicle size, among other factors.
- Current treatment modalities for enlarged facial pores target these factors and include topical retinoids, chemical peels, oral antiandrogens, lasers, radiofrequency, and ultrasound devices, with the latter devices offering the most novel and robust choices.
- New botanically derived topical treatments, specifically copper chlorophyllin complex sodium salt and tetra-hydro-jasmonic acid, are in development with initial positive results, though studies are still limited.
Using a Combination of Therapies to Manage Rosacea

What do your patients need to know at the first visit?
Patients need to understand that rosacea has no gender, age, or race predilection. It is caused by a personal and genetic proinflammatory predisposition. Rosacea patients seem to have a genetic predisposition to overproduce cathelicidins, a small antimicrobial peptide that is produced by the action of stratum corneum tryptic enzyme. They have more production and abnormal forms of cathelicidins produced by high levels of stratum corneum tryptic enzyme. This upregulation of inflammatory cathelicidins on the dermis is associated with vascular instability and exacerbated by triggers such as sunlight, hot drinks, spicy foods, stress, and rapid changing weather.
I divide the condition into proinflammatory predisposition, vascular instability with redness, Demodex infection of the hair follicle, and sebaceous gland overgrowth. More recently, the bacterium Bacillus oleronius was isolated from inside a Demodex mite and was found to produce molecules provoking an immune reaction in rosacea patients (Erbaguci and Ozgöztaşi). Other studies have shown that patients with varying types of rosacea react to the molecules produced by this bacterium, exposing it as a likely trigger for the condition (Li et al). What’s more, this bacterium is sensitive to the antibiotics used to treat rosacea.
What are your go-to treatments? What are the side effects?
For inflammation I prescribe anti-inflammatory (low dose) or antibacterial (high dose) doses of doxycycline and/or anti-inflammatory azelaic acid gel 15% twice daily after application of barrier repair topical hyaluronic acid. For the vascular component I use the temporary relief from the application of brimonidine gel 0.33% in the morning in addition to the topical given for inflammatory rosacea, and the more durable excel V (532 and 1064 nm) laser. Ultimately, topical ivermectin is prescribed for those patients who do not respond to previously mentioned treatments for coverage of Demodex infestation. For rhinophyma I offer a surgical approach and laser treatments; surgical removal of the excess glandular growth is followed by fractional ablative and nonablative treatments for scar reduction after surgery.
All patients should apply an inorganic sun protection factor 50+ sunblock with titanium dioxide and zinc oxide to prevent sunlight from being a trigger. All patients are encouraged to avoid triggers.
I try to prevent the potential side effects associated with rosacea treatments. For example, applying barrier repair hyaluronic acid before azelaic acid to prevent irritation and telling patients they might have vascular rebound phenomena with more redness after brimonidine application wears off. I also explain to patients that laser treatments induce temporary erythema and swelling that may last 3 days.
How do you keep patients compliant with treatment?
In general, my patients are compliant with their treatments, which I ascribe to the simplicity of a twice-daily regimen that is written for them. They understand that I design a treatment regimen for each individual patient based on his/her presentation.
What resources do you recommend to patients for more information?
I recommend web-based resources that can provide further assistance and information, such as the American Academy of Dermatology website (https://www.aad.org/public/diseases/acne-and-rosacea/rosacea), National Rosacea Society (www.rosacea.org), and specific disease foundations (eg, International Rosacea Foundation [www.internationalrosaceafoundation.org]).Suggested Readings
- Erbaguci Z, Ozgöztaşi O. The significance of Demodex folliculorum density in rosacea. Int J Dermatol. 1998;37:421-425.
- Li J, O’Reilly N, Sheha H, et al. Correlation between ocular Demodex infestation and serum immunoreactivity to Bacillus proteins in patients with facial rosacea. Ophthalmology. 2010;117:870-877.

What do your patients need to know at the first visit?
Patients need to understand that rosacea has no gender, age, or race predilection. It is caused by a personal and genetic proinflammatory predisposition. Rosacea patients seem to have a genetic predisposition to overproduce cathelicidins, a small antimicrobial peptide that is produced by the action of stratum corneum tryptic enzyme. They have more production and abnormal forms of cathelicidins produced by high levels of stratum corneum tryptic enzyme. This upregulation of inflammatory cathelicidins on the dermis is associated with vascular instability and exacerbated by triggers such as sunlight, hot drinks, spicy foods, stress, and rapid changing weather.
I divide the condition into proinflammatory predisposition, vascular instability with redness, Demodex infection of the hair follicle, and sebaceous gland overgrowth. More recently, the bacterium Bacillus oleronius was isolated from inside a Demodex mite and was found to produce molecules provoking an immune reaction in rosacea patients (Erbaguci and Ozgöztaşi). Other studies have shown that patients with varying types of rosacea react to the molecules produced by this bacterium, exposing it as a likely trigger for the condition (Li et al). What’s more, this bacterium is sensitive to the antibiotics used to treat rosacea.
What are your go-to treatments? What are the side effects?
For inflammation I prescribe anti-inflammatory (low dose) or antibacterial (high dose) doses of doxycycline and/or anti-inflammatory azelaic acid gel 15% twice daily after application of barrier repair topical hyaluronic acid. For the vascular component I use the temporary relief from the application of brimonidine gel 0.33% in the morning in addition to the topical given for inflammatory rosacea, and the more durable excel V (532 and 1064 nm) laser. Ultimately, topical ivermectin is prescribed for those patients who do not respond to previously mentioned treatments for coverage of Demodex infestation. For rhinophyma I offer a surgical approach and laser treatments; surgical removal of the excess glandular growth is followed by fractional ablative and nonablative treatments for scar reduction after surgery.
All patients should apply an inorganic sun protection factor 50+ sunblock with titanium dioxide and zinc oxide to prevent sunlight from being a trigger. All patients are encouraged to avoid triggers.
I try to prevent the potential side effects associated with rosacea treatments. For example, applying barrier repair hyaluronic acid before azelaic acid to prevent irritation and telling patients they might have vascular rebound phenomena with more redness after brimonidine application wears off. I also explain to patients that laser treatments induce temporary erythema and swelling that may last 3 days.
How do you keep patients compliant with treatment?
In general, my patients are compliant with their treatments, which I ascribe to the simplicity of a twice-daily regimen that is written for them. They understand that I design a treatment regimen for each individual patient based on his/her presentation.
What resources do you recommend to patients for more information?
I recommend web-based resources that can provide further assistance and information, such as the American Academy of Dermatology website (https://www.aad.org/public/diseases/acne-and-rosacea/rosacea), National Rosacea Society (www.rosacea.org), and specific disease foundations (eg, International Rosacea Foundation [www.internationalrosaceafoundation.org]).Suggested Readings
- Erbaguci Z, Ozgöztaşi O. The significance of Demodex folliculorum density in rosacea. Int J Dermatol. 1998;37:421-425.
- Li J, O’Reilly N, Sheha H, et al. Correlation between ocular Demodex infestation and serum immunoreactivity to Bacillus proteins in patients with facial rosacea. Ophthalmology. 2010;117:870-877.

What do your patients need to know at the first visit?
Patients need to understand that rosacea has no gender, age, or race predilection. It is caused by a personal and genetic proinflammatory predisposition. Rosacea patients seem to have a genetic predisposition to overproduce cathelicidins, a small antimicrobial peptide that is produced by the action of stratum corneum tryptic enzyme. They have more production and abnormal forms of cathelicidins produced by high levels of stratum corneum tryptic enzyme. This upregulation of inflammatory cathelicidins on the dermis is associated with vascular instability and exacerbated by triggers such as sunlight, hot drinks, spicy foods, stress, and rapid changing weather.
I divide the condition into proinflammatory predisposition, vascular instability with redness, Demodex infection of the hair follicle, and sebaceous gland overgrowth. More recently, the bacterium Bacillus oleronius was isolated from inside a Demodex mite and was found to produce molecules provoking an immune reaction in rosacea patients (Erbaguci and Ozgöztaşi). Other studies have shown that patients with varying types of rosacea react to the molecules produced by this bacterium, exposing it as a likely trigger for the condition (Li et al). What’s more, this bacterium is sensitive to the antibiotics used to treat rosacea.
What are your go-to treatments? What are the side effects?
For inflammation I prescribe anti-inflammatory (low dose) or antibacterial (high dose) doses of doxycycline and/or anti-inflammatory azelaic acid gel 15% twice daily after application of barrier repair topical hyaluronic acid. For the vascular component I use the temporary relief from the application of brimonidine gel 0.33% in the morning in addition to the topical given for inflammatory rosacea, and the more durable excel V (532 and 1064 nm) laser. Ultimately, topical ivermectin is prescribed for those patients who do not respond to previously mentioned treatments for coverage of Demodex infestation. For rhinophyma I offer a surgical approach and laser treatments; surgical removal of the excess glandular growth is followed by fractional ablative and nonablative treatments for scar reduction after surgery.
All patients should apply an inorganic sun protection factor 50+ sunblock with titanium dioxide and zinc oxide to prevent sunlight from being a trigger. All patients are encouraged to avoid triggers.
I try to prevent the potential side effects associated with rosacea treatments. For example, applying barrier repair hyaluronic acid before azelaic acid to prevent irritation and telling patients they might have vascular rebound phenomena with more redness after brimonidine application wears off. I also explain to patients that laser treatments induce temporary erythema and swelling that may last 3 days.
How do you keep patients compliant with treatment?
In general, my patients are compliant with their treatments, which I ascribe to the simplicity of a twice-daily regimen that is written for them. They understand that I design a treatment regimen for each individual patient based on his/her presentation.
What resources do you recommend to patients for more information?
I recommend web-based resources that can provide further assistance and information, such as the American Academy of Dermatology website (https://www.aad.org/public/diseases/acne-and-rosacea/rosacea), National Rosacea Society (www.rosacea.org), and specific disease foundations (eg, International Rosacea Foundation [www.internationalrosaceafoundation.org]).Suggested Readings
- Erbaguci Z, Ozgöztaşi O. The significance of Demodex folliculorum density in rosacea. Int J Dermatol. 1998;37:421-425.
- Li J, O’Reilly N, Sheha H, et al. Correlation between ocular Demodex infestation and serum immunoreactivity to Bacillus proteins in patients with facial rosacea. Ophthalmology. 2010;117:870-877.
There’s still a place for HA fillers in fine facial lines
NEWPORT BEACH, CALIF. – Despite all the technical advances in aesthetic medicine, there’s still a place for treating fine facial lines with thin hyaluronic acid fillers, according to Dr. Mark G. Rubin, a dermatologist at the Lasky Skin Center in Beverly Hills, Calif.
“In the last couple of years, with all the volumizing products, a lot of people say they don’t treat wrinkles anymore. The idea is if you just put some volume in [a patient’s] nasojugal groove, you inflate the cheek, pull the skin tight, and the nasolabial fold will miraculously disappear. I don’t think that’s really true,” he said at the meeting held by Global Academy for Medical Education.
When patients don’t have good skin elasticity and tightness – and most older patients worried about fine lines don’t – volumizing will improve skin contour but not do much for atrophic lines.
That’s a good time to turn to fillers. “Basically, you are putting putty into a dent. Different wrinkles need different depths of material. We have fillers that are thin like sand, medium like pebbles, or thick like boulders.” Shallow lines need thinner material; deeper folds need volumizing boulders for lift. In some patients, “you need to layer them, boulders first then more superficial fillers to smooth out the surface,” Dr. Rubin said. “There are a lot of medium and deep fillers, but for really fine lines and superficial filling, there’s only Restylane Silk and Belotero.”
However, even superficial fillers need to be diluted sometimes with saline or lidocaine. “There isn’t a perfect filler; you need to create the one that works by changing its characteristics,” he said.
“With Restylane, if you dilute it too much, it turns into water, and you get no lift at all. You can dilute Juvederm down pretty well, but in some patients it still leaves ridges. You can dilute Belotero a lot without losing its ability to create lift, but I think in a lot of patients, Restylane Silk has a little better persistence than Belotero,” he said.
Staying in the dermis is important for superficial lines. “You need to come in at a very acute angle, and you have to see drug coming back at you through the pores. If you are not seeing that, you are definitely too deep. You also need to overcorrect, and see the area blanch a little bit,” he said.
Dr. Rubin warns patients that they might have what looks like a string of pearls under their skin after injection. “We massage [the beads] down with a Q-tip,” rolling it back and forth over the bulges, and they melt over time. A part of the reaction is histamine-related, so antihistamines like loratadine (Claritin) can help.
There can be more serious swelling as well, especially after perioral injections, and “it’s more common with Silk than with regular Restylane. Some patients have erythema that stays a long time, and some have a burning sensation. If it’s not horrific, you wait and see. If it’s still there in 10 days or 2 weeks, my inclination is to dissolve it out and switch drugs,” he said.
Dr. Rubin is a consultant for Merz, maker of Belotero, Radiesse, and other products. Global Academy and this news organization are owned by the same company.
NEWPORT BEACH, CALIF. – Despite all the technical advances in aesthetic medicine, there’s still a place for treating fine facial lines with thin hyaluronic acid fillers, according to Dr. Mark G. Rubin, a dermatologist at the Lasky Skin Center in Beverly Hills, Calif.
“In the last couple of years, with all the volumizing products, a lot of people say they don’t treat wrinkles anymore. The idea is if you just put some volume in [a patient’s] nasojugal groove, you inflate the cheek, pull the skin tight, and the nasolabial fold will miraculously disappear. I don’t think that’s really true,” he said at the meeting held by Global Academy for Medical Education.
When patients don’t have good skin elasticity and tightness – and most older patients worried about fine lines don’t – volumizing will improve skin contour but not do much for atrophic lines.
That’s a good time to turn to fillers. “Basically, you are putting putty into a dent. Different wrinkles need different depths of material. We have fillers that are thin like sand, medium like pebbles, or thick like boulders.” Shallow lines need thinner material; deeper folds need volumizing boulders for lift. In some patients, “you need to layer them, boulders first then more superficial fillers to smooth out the surface,” Dr. Rubin said. “There are a lot of medium and deep fillers, but for really fine lines and superficial filling, there’s only Restylane Silk and Belotero.”
However, even superficial fillers need to be diluted sometimes with saline or lidocaine. “There isn’t a perfect filler; you need to create the one that works by changing its characteristics,” he said.
“With Restylane, if you dilute it too much, it turns into water, and you get no lift at all. You can dilute Juvederm down pretty well, but in some patients it still leaves ridges. You can dilute Belotero a lot without losing its ability to create lift, but I think in a lot of patients, Restylane Silk has a little better persistence than Belotero,” he said.
Staying in the dermis is important for superficial lines. “You need to come in at a very acute angle, and you have to see drug coming back at you through the pores. If you are not seeing that, you are definitely too deep. You also need to overcorrect, and see the area blanch a little bit,” he said.
Dr. Rubin warns patients that they might have what looks like a string of pearls under their skin after injection. “We massage [the beads] down with a Q-tip,” rolling it back and forth over the bulges, and they melt over time. A part of the reaction is histamine-related, so antihistamines like loratadine (Claritin) can help.
There can be more serious swelling as well, especially after perioral injections, and “it’s more common with Silk than with regular Restylane. Some patients have erythema that stays a long time, and some have a burning sensation. If it’s not horrific, you wait and see. If it’s still there in 10 days or 2 weeks, my inclination is to dissolve it out and switch drugs,” he said.
Dr. Rubin is a consultant for Merz, maker of Belotero, Radiesse, and other products. Global Academy and this news organization are owned by the same company.
NEWPORT BEACH, CALIF. – Despite all the technical advances in aesthetic medicine, there’s still a place for treating fine facial lines with thin hyaluronic acid fillers, according to Dr. Mark G. Rubin, a dermatologist at the Lasky Skin Center in Beverly Hills, Calif.
“In the last couple of years, with all the volumizing products, a lot of people say they don’t treat wrinkles anymore. The idea is if you just put some volume in [a patient’s] nasojugal groove, you inflate the cheek, pull the skin tight, and the nasolabial fold will miraculously disappear. I don’t think that’s really true,” he said at the meeting held by Global Academy for Medical Education.
When patients don’t have good skin elasticity and tightness – and most older patients worried about fine lines don’t – volumizing will improve skin contour but not do much for atrophic lines.
That’s a good time to turn to fillers. “Basically, you are putting putty into a dent. Different wrinkles need different depths of material. We have fillers that are thin like sand, medium like pebbles, or thick like boulders.” Shallow lines need thinner material; deeper folds need volumizing boulders for lift. In some patients, “you need to layer them, boulders first then more superficial fillers to smooth out the surface,” Dr. Rubin said. “There are a lot of medium and deep fillers, but for really fine lines and superficial filling, there’s only Restylane Silk and Belotero.”
However, even superficial fillers need to be diluted sometimes with saline or lidocaine. “There isn’t a perfect filler; you need to create the one that works by changing its characteristics,” he said.
“With Restylane, if you dilute it too much, it turns into water, and you get no lift at all. You can dilute Juvederm down pretty well, but in some patients it still leaves ridges. You can dilute Belotero a lot without losing its ability to create lift, but I think in a lot of patients, Restylane Silk has a little better persistence than Belotero,” he said.
Staying in the dermis is important for superficial lines. “You need to come in at a very acute angle, and you have to see drug coming back at you through the pores. If you are not seeing that, you are definitely too deep. You also need to overcorrect, and see the area blanch a little bit,” he said.
Dr. Rubin warns patients that they might have what looks like a string of pearls under their skin after injection. “We massage [the beads] down with a Q-tip,” rolling it back and forth over the bulges, and they melt over time. A part of the reaction is histamine-related, so antihistamines like loratadine (Claritin) can help.
There can be more serious swelling as well, especially after perioral injections, and “it’s more common with Silk than with regular Restylane. Some patients have erythema that stays a long time, and some have a burning sensation. If it’s not horrific, you wait and see. If it’s still there in 10 days or 2 weeks, my inclination is to dissolve it out and switch drugs,” he said.
Dr. Rubin is a consultant for Merz, maker of Belotero, Radiesse, and other products. Global Academy and this news organization are owned by the same company.
EXPERT ANALYSIS FROM THE SUMMIT IN AESTHETIC MEDICINE
Lasers for Darker Skin Types
Red Alert: Can Topical Skin Care Products Promote Melanoma Metastasis?

Le Gal et al (Sci Transl Med. 2015;7:308re8) discovered that antioxidant administration in mice not only increased lymph node metastases but also increased the migration and invasive properties of human melanoma cells. However, the antioxidant N-acetylcysteine (NAC) had no impact on the number and size of the primary tumors (in mice), and neither NAC nor Trolox (6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid), a structurally unrelated antioxidant and soluble vitamin E analogue, affected the proliferation of human melanoma cells. Hence, the progression of malignant melanoma (MM), a cancer that is sensitive to changes in reduction-oxidation status, may be influenced by exposure to antioxidants and vitamin E.
What’s the issue?
Healthy individuals and oncology patients commonly use supplements containing antioxidants to prevent cancer and fight malignancy, respectively. However, animal studies and human clinical trials have shown that antioxidants increase cancer risk and accelerate the progression of primary lung tumors. Le Gal et al’s study regarding progression of melanoma metastases following exposure to antioxidants extends the observations demonstrated for lung neoplasms. N-acetylcysteine was added to the drinking water of mice, whereas NAC and Trolox were added to a panel of human MM cell lines. N-acetylcysteine increased lymph node metastases in the endogenous mouse model of MM, and both NAC and Trolox markedly increased the migrations and invasive properties of human MM cells.
Cancers may be caused or exacerbated by free radicals. It has been assumed that antioxidants may protect against malignancy by destroying free radicals. Although prior studies have concluded that antioxidants prevent healthy cells from transforming into cancer after exposure to free radicals, Le Gal et al’s research suggests that antioxidants may not only protect but also enhance tumor progression once a cancer has developed.
If one extends the results of animal and tissue culture studies to humans, exposure to antioxidants may potentially influence the course of metastatic disease in patients who have already developed melanoma. In addition to systemic exposure after receiving oral antioxidants, melanoma patients also can be topically exposed to antioxidants. For example, nonprescription skin care products such as cutaneous rejuvenation treatments, emollients, and sunscreens can contain β-carotene, vitamin E, and other antioxidants. It remains to be determined whether topical exposure to antioxidants can cause the same observations that have occurred following systemic absorption in mice or tissue culture studies in human cell lines. Should we caution our melanoma patients with regards to what they apply to their skin?

Le Gal et al (Sci Transl Med. 2015;7:308re8) discovered that antioxidant administration in mice not only increased lymph node metastases but also increased the migration and invasive properties of human melanoma cells. However, the antioxidant N-acetylcysteine (NAC) had no impact on the number and size of the primary tumors (in mice), and neither NAC nor Trolox (6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid), a structurally unrelated antioxidant and soluble vitamin E analogue, affected the proliferation of human melanoma cells. Hence, the progression of malignant melanoma (MM), a cancer that is sensitive to changes in reduction-oxidation status, may be influenced by exposure to antioxidants and vitamin E.
What’s the issue?
Healthy individuals and oncology patients commonly use supplements containing antioxidants to prevent cancer and fight malignancy, respectively. However, animal studies and human clinical trials have shown that antioxidants increase cancer risk and accelerate the progression of primary lung tumors. Le Gal et al’s study regarding progression of melanoma metastases following exposure to antioxidants extends the observations demonstrated for lung neoplasms. N-acetylcysteine was added to the drinking water of mice, whereas NAC and Trolox were added to a panel of human MM cell lines. N-acetylcysteine increased lymph node metastases in the endogenous mouse model of MM, and both NAC and Trolox markedly increased the migrations and invasive properties of human MM cells.
Cancers may be caused or exacerbated by free radicals. It has been assumed that antioxidants may protect against malignancy by destroying free radicals. Although prior studies have concluded that antioxidants prevent healthy cells from transforming into cancer after exposure to free radicals, Le Gal et al’s research suggests that antioxidants may not only protect but also enhance tumor progression once a cancer has developed.
If one extends the results of animal and tissue culture studies to humans, exposure to antioxidants may potentially influence the course of metastatic disease in patients who have already developed melanoma. In addition to systemic exposure after receiving oral antioxidants, melanoma patients also can be topically exposed to antioxidants. For example, nonprescription skin care products such as cutaneous rejuvenation treatments, emollients, and sunscreens can contain β-carotene, vitamin E, and other antioxidants. It remains to be determined whether topical exposure to antioxidants can cause the same observations that have occurred following systemic absorption in mice or tissue culture studies in human cell lines. Should we caution our melanoma patients with regards to what they apply to their skin?

Le Gal et al (Sci Transl Med. 2015;7:308re8) discovered that antioxidant administration in mice not only increased lymph node metastases but also increased the migration and invasive properties of human melanoma cells. However, the antioxidant N-acetylcysteine (NAC) had no impact on the number and size of the primary tumors (in mice), and neither NAC nor Trolox (6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid), a structurally unrelated antioxidant and soluble vitamin E analogue, affected the proliferation of human melanoma cells. Hence, the progression of malignant melanoma (MM), a cancer that is sensitive to changes in reduction-oxidation status, may be influenced by exposure to antioxidants and vitamin E.
What’s the issue?
Healthy individuals and oncology patients commonly use supplements containing antioxidants to prevent cancer and fight malignancy, respectively. However, animal studies and human clinical trials have shown that antioxidants increase cancer risk and accelerate the progression of primary lung tumors. Le Gal et al’s study regarding progression of melanoma metastases following exposure to antioxidants extends the observations demonstrated for lung neoplasms. N-acetylcysteine was added to the drinking water of mice, whereas NAC and Trolox were added to a panel of human MM cell lines. N-acetylcysteine increased lymph node metastases in the endogenous mouse model of MM, and both NAC and Trolox markedly increased the migrations and invasive properties of human MM cells.
Cancers may be caused or exacerbated by free radicals. It has been assumed that antioxidants may protect against malignancy by destroying free radicals. Although prior studies have concluded that antioxidants prevent healthy cells from transforming into cancer after exposure to free radicals, Le Gal et al’s research suggests that antioxidants may not only protect but also enhance tumor progression once a cancer has developed.
If one extends the results of animal and tissue culture studies to humans, exposure to antioxidants may potentially influence the course of metastatic disease in patients who have already developed melanoma. In addition to systemic exposure after receiving oral antioxidants, melanoma patients also can be topically exposed to antioxidants. For example, nonprescription skin care products such as cutaneous rejuvenation treatments, emollients, and sunscreens can contain β-carotene, vitamin E, and other antioxidants. It remains to be determined whether topical exposure to antioxidants can cause the same observations that have occurred following systemic absorption in mice or tissue culture studies in human cell lines. Should we caution our melanoma patients with regards to what they apply to their skin?
Aesthetic Dermatology: Synthetic second skin
Hot off the presses, a new so-called “second skin” is being redeveloped and rebranded for use in both cosmetic and medical dermatology. But what is this substance, and will it hold up to all the claims the manufacturer and research team suggest?
Recently described in Nature Materials, the liquid polymer developed by chemical engineers at MIT is a synthetic, adherent silicone-based film that lies perfectly invisibly on the skin – providing a pulling or temporary tightening of the skin. The product was initially marketed by the company Living Proof as “Neotensil” [an acronym for (Neo) new, (T) transforming, (E) elastic, (N) non-invasive, (S) supportive, (I) invisible, and immediate (L) layer solution]. When applied to the area under the eye, the product creates a so-called “Spanx” effect or tightening of periorbital skin.
The material – called XPL – is delivered in a two-step sequential process. First, a polysiloxane cream is applied to the skin, followed by a platinum catalyst that induces the polymer to harden and tighten the skin underneath. The product uses patented Strateris technology, which is described as creating invisible “shapewear” for the eye; a film that tightens, smooths, and lifts the appearance of skin for up to 24 hours. It was briefly on the market in 2014-2015, then taken off the market to be redeveloped.
Does it work? Yes. Although it takes about an hour to take effect, the clinical results are jaw dropping. However, it also has its drawbacks. The polymer – which hardens within 2 minutes – must be applied to clean skin, with no creams or makeup whatsoever. And makeup cannot be applied over the treated area either, as the area looks irregular and uneven with makeup. This is a very difficult obstacle to overcome for many female patients.
Additionally, to take off the product, the polymer must be dissolved with a special chemical remover that is packaged with the product. Without this key component, it is very difficult and very irritating to remove. Although none of the patients I have used this product on have developed allergic reactions, any synthetic polymer, particularly one with adherent properties, has the potential to be an irritant and/or an allergen. Long-term clinical trials are needed to both validate its efficacy and side-effect profile.
The potential for clinical uses is vast. The product has been shown to provide a synthetic skin barrier that minimizes transepidermal water loss, improving skin hydration. Its uses in burns, atopic dermatitis, bullous disease, and psoriasis could help those with altered skin-barrier function. The researchers are also hoping to use this product for targeted drug delivery and for UV protection.
After a decade of research, the MIT team has developed a skinlike material that is invisible and mimics both the mechanical and elastic properties of the skin. Future clinical studies are essential to evaluating its broad applicability in both dermatology and general medicine.
1. Nature Materials 2016. doi:10.1038/nmat4635.
Dr. Wesley and Dr. Talakoub are co-contributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Dr. Talakoub has no disclosures related to the product. Write to them at [email protected].
Hot off the presses, a new so-called “second skin” is being redeveloped and rebranded for use in both cosmetic and medical dermatology. But what is this substance, and will it hold up to all the claims the manufacturer and research team suggest?
Recently described in Nature Materials, the liquid polymer developed by chemical engineers at MIT is a synthetic, adherent silicone-based film that lies perfectly invisibly on the skin – providing a pulling or temporary tightening of the skin. The product was initially marketed by the company Living Proof as “Neotensil” [an acronym for (Neo) new, (T) transforming, (E) elastic, (N) non-invasive, (S) supportive, (I) invisible, and immediate (L) layer solution]. When applied to the area under the eye, the product creates a so-called “Spanx” effect or tightening of periorbital skin.
The material – called XPL – is delivered in a two-step sequential process. First, a polysiloxane cream is applied to the skin, followed by a platinum catalyst that induces the polymer to harden and tighten the skin underneath. The product uses patented Strateris technology, which is described as creating invisible “shapewear” for the eye; a film that tightens, smooths, and lifts the appearance of skin for up to 24 hours. It was briefly on the market in 2014-2015, then taken off the market to be redeveloped.
Does it work? Yes. Although it takes about an hour to take effect, the clinical results are jaw dropping. However, it also has its drawbacks. The polymer – which hardens within 2 minutes – must be applied to clean skin, with no creams or makeup whatsoever. And makeup cannot be applied over the treated area either, as the area looks irregular and uneven with makeup. This is a very difficult obstacle to overcome for many female patients.
Additionally, to take off the product, the polymer must be dissolved with a special chemical remover that is packaged with the product. Without this key component, it is very difficult and very irritating to remove. Although none of the patients I have used this product on have developed allergic reactions, any synthetic polymer, particularly one with adherent properties, has the potential to be an irritant and/or an allergen. Long-term clinical trials are needed to both validate its efficacy and side-effect profile.
The potential for clinical uses is vast. The product has been shown to provide a synthetic skin barrier that minimizes transepidermal water loss, improving skin hydration. Its uses in burns, atopic dermatitis, bullous disease, and psoriasis could help those with altered skin-barrier function. The researchers are also hoping to use this product for targeted drug delivery and for UV protection.
After a decade of research, the MIT team has developed a skinlike material that is invisible and mimics both the mechanical and elastic properties of the skin. Future clinical studies are essential to evaluating its broad applicability in both dermatology and general medicine.
1. Nature Materials 2016. doi:10.1038/nmat4635.
Dr. Wesley and Dr. Talakoub are co-contributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Dr. Talakoub has no disclosures related to the product. Write to them at [email protected].
Hot off the presses, a new so-called “second skin” is being redeveloped and rebranded for use in both cosmetic and medical dermatology. But what is this substance, and will it hold up to all the claims the manufacturer and research team suggest?
Recently described in Nature Materials, the liquid polymer developed by chemical engineers at MIT is a synthetic, adherent silicone-based film that lies perfectly invisibly on the skin – providing a pulling or temporary tightening of the skin. The product was initially marketed by the company Living Proof as “Neotensil” [an acronym for (Neo) new, (T) transforming, (E) elastic, (N) non-invasive, (S) supportive, (I) invisible, and immediate (L) layer solution]. When applied to the area under the eye, the product creates a so-called “Spanx” effect or tightening of periorbital skin.
The material – called XPL – is delivered in a two-step sequential process. First, a polysiloxane cream is applied to the skin, followed by a platinum catalyst that induces the polymer to harden and tighten the skin underneath. The product uses patented Strateris technology, which is described as creating invisible “shapewear” for the eye; a film that tightens, smooths, and lifts the appearance of skin for up to 24 hours. It was briefly on the market in 2014-2015, then taken off the market to be redeveloped.
Does it work? Yes. Although it takes about an hour to take effect, the clinical results are jaw dropping. However, it also has its drawbacks. The polymer – which hardens within 2 minutes – must be applied to clean skin, with no creams or makeup whatsoever. And makeup cannot be applied over the treated area either, as the area looks irregular and uneven with makeup. This is a very difficult obstacle to overcome for many female patients.
Additionally, to take off the product, the polymer must be dissolved with a special chemical remover that is packaged with the product. Without this key component, it is very difficult and very irritating to remove. Although none of the patients I have used this product on have developed allergic reactions, any synthetic polymer, particularly one with adherent properties, has the potential to be an irritant and/or an allergen. Long-term clinical trials are needed to both validate its efficacy and side-effect profile.
The potential for clinical uses is vast. The product has been shown to provide a synthetic skin barrier that minimizes transepidermal water loss, improving skin hydration. Its uses in burns, atopic dermatitis, bullous disease, and psoriasis could help those with altered skin-barrier function. The researchers are also hoping to use this product for targeted drug delivery and for UV protection.
After a decade of research, the MIT team has developed a skinlike material that is invisible and mimics both the mechanical and elastic properties of the skin. Future clinical studies are essential to evaluating its broad applicability in both dermatology and general medicine.
1. Nature Materials 2016. doi:10.1038/nmat4635.
Dr. Wesley and Dr. Talakoub are co-contributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Dr. Talakoub has no disclosures related to the product. Write to them at [email protected].
Preventing, Identifying, and Managing Cosmetic Procedure Complications, Part 1: Soft-Tissue Augmentation and Botulinum Toxin Injections
The primary cosmetic procedures that dermatology residents will perform or assist in performing during their training are soft-tissue augmentation, botulinum toxin injections, chemical peels, and laser therapy. Because complications can occur from these procedures, it is important for residents to learn how to prevent, identify, and manage them for optimal patient outcomes. In part 1 of this 2-part series, soft-tissue augmentation and botulinum toxin injections are discussed. Chemical peels and laser therapy will be addressed in part 2.
Soft-Tissue Augmentation
Soft-tissue fillers include those that are made from collagen (bovine or human), hyaluronic acid (HA), poly-L-lactic acid, calcium hydroxylapatite, silicone, and polymethylmethacrylate. In general, acute complications of soft-tissue filler injections include erythema, swelling, and bruising.1-3 Patients who take blood thinners or supplements (eg, vitamin E, ginseng, garlic, ginger) should be asked to discontinue use 1 week prior to the procedure. Patients who take blood thinners also should be counseled to expect some bruising. Prior to injection, the skin should be thoroughly cleansed to avoid introducing skin bacteria into the injection site and to reduce infection risk. Postinjection erythema may be related to mast cell activation, which is temporary and should resolve after a few days.1-3
If you find yourself injecting the filler too superficially, you may notice that the skin begins to take on a blue-gray hue1-3 that is known as the Tyndall effect and can be prevented by injecting the filler at the proper level. For example, collagen-based fillers should be placed at the mid dermis, thicker HA fillers should be placed in the deep dermis, and calcium hydroxylapatite should be placed at the junction of the dermis and subcutaneous tissue. Polymethylmethacrylate and poly-L-lactic acid should both be placed subdermally.1-3
The gravest immediate complications associated with soft-tissue filler injections are occlusion of the central retinal artery and/or skin necrosis.1-4 Residents should never inject filler to the glabella or to the nose.1-3 Injections at these sites are sometimes performed but should only be performed by experienced dermatologists. The perioral and tear trough regions also are high-risk injection areas that require a high degree of experience and should only be injected with proper supervision by an experienced dermatologist.1-3 Residents generally can avoid these complications, though not with a 100% guarantee, by avoiding injections in high-risk areas, aspirating to check for blood, and slowly injecting a small amount of filler into the treatment area.1-3 A consensus statement on management of injection-induced necrosis advises to apply a nitropaste ointment 2% to the treatment site or administer an oral aspirin if the patient develops severe pain; vision loss; or acute skin discoloration, especially blanching.4 For HA-based fillers, at least 200 U of hyaluronidase should be injected. It has been suggested that saline can be injected to flush out calcium hydroxylapatite fillers.3 Warm compresses should be placed on the involved area. Following these interventions, any patient with vision loss or orbital pain should immediately undergo ophthalmologic evaluation.3 The most important intervention occurs in the first 24 hours.3,4 After 24 hours, careful wound care, oral anticoagulants, and hyperbaric oxygen therapy have been suggested as management options.3
There are 2 major chronic complications of soft-tissue filler injection, including delayed-onset infection, which occurs 2 weeks or more postinjection, and granuloma formation.1-3 Chronic low-grade infection at the injection site may be indicative of biofilm formation. If an HA filler was used, it should be dissolved with hyaluronidase to help break up the biofilm nidus.3 A course of oral antibiotics also may be indicated.1-3 Intralesional steroids may be used but only after antibiotics have been administered. A biofilm that develops from more permanent fillers may be more difficult to manage. Atypical mycobacterial infections have been known to develop at injection sites, which should be considered in refractory cases.1-3,5
Calcium hydroxylapatite, polymethylmethacrylate, and silicone can stimulate chronic immune system activation, which makes them more prone to granuloma formation.1-3 Once infection is ruled out, granulomas may be treated with intralesional steroids, surgical excision (if the results would be cosmetically acceptable), laser therapy, or potentially local injection of an immunosuppressant (eg, methotrexate, 5-fluorouracil).3
Botulinum Toxin Injections
Patients who are pregnant, lactating, or have neuromuscular disease are not candidates for botulinum toxin injections. There also is a risk that patients taking calcium channel blockers or aminoglycoside antibiotics may experience potentiated effects of the botulinum toxin.6
Patients should be informed that a postinjection headache may occur and should be treated with over-the-counter medications.6 Complication-free botulinum toxin procedures depend heavily on the physician’s knowledge of facial anatomy.1,6 The diagrams provided by Hirsch and Stier1 offer an excellent guide on where to place the injections. Brow droop, eyelid ptosis, and “Spock brow” (eyebrows that are overarched) largely can be avoided by proper injection point placement. A Spock brow may be corrected by injecting the lateral upper forehead with a few units to correct the exaggerated arch.6,7 For eyelid ptosis, apraclonidine 0.05% drops (1–2 drops 3 times daily) should be used until the ptosis resolves.6 Phenylephrine hydrochloride drops may be used should a patient have a documented sensitivity to apraclonidine but should not be used in a patient with acute angle-closure glaucoma or aneurysms.6
Final Thoughts
Learning to perform soft-tissue augmentation and botulinum toxin injections can be a satisfying and fun part of dermatology residency. Preventing, identifying, and managing any complications that may occur is an integral part of performing these procedures.
- Hirsch R, Stier M. Complications and their management in cosmetic dermatology. Dermatol Clin. 2009;27:507-520.
- Gladstone HB, Cohen JL. Adverse effects when injecting facial fillers. Semin Cutan Med Surg. 2007;26:34-39.
- Boulle K, Heydenrych I. Patient factors influencing dermal filler complications: prevention, assessment, and treatment. Clin Cosmet Investig Dermatol. 2015;8:205-214.
- Cohen JL, Biesman BS, Dayan SH, et al. Treatment of hyaluronic acid filler–induced impending necrosis with hyaluronidase: consensus recommendations [published online May 10, 2015]. Aesthet Surg J. 2015;35:844-849.
- Rodriguez JM, Xie YL, Winthrop KL, et al. Mycobacterium chelonae facial infections following injection of dermal filler. Aesthet Surg J. 2013;33:265-269.
- Nigam PK, Nigam A. Botulinum toxin. Indian J Dermatol. 2010;55:8-14.
- Carruthers A, Carruthers J. Update on the botulinum neurotoxins. Skin Therapy Lett. 2001;6:1-2.
The primary cosmetic procedures that dermatology residents will perform or assist in performing during their training are soft-tissue augmentation, botulinum toxin injections, chemical peels, and laser therapy. Because complications can occur from these procedures, it is important for residents to learn how to prevent, identify, and manage them for optimal patient outcomes. In part 1 of this 2-part series, soft-tissue augmentation and botulinum toxin injections are discussed. Chemical peels and laser therapy will be addressed in part 2.
Soft-Tissue Augmentation
Soft-tissue fillers include those that are made from collagen (bovine or human), hyaluronic acid (HA), poly-L-lactic acid, calcium hydroxylapatite, silicone, and polymethylmethacrylate. In general, acute complications of soft-tissue filler injections include erythema, swelling, and bruising.1-3 Patients who take blood thinners or supplements (eg, vitamin E, ginseng, garlic, ginger) should be asked to discontinue use 1 week prior to the procedure. Patients who take blood thinners also should be counseled to expect some bruising. Prior to injection, the skin should be thoroughly cleansed to avoid introducing skin bacteria into the injection site and to reduce infection risk. Postinjection erythema may be related to mast cell activation, which is temporary and should resolve after a few days.1-3
If you find yourself injecting the filler too superficially, you may notice that the skin begins to take on a blue-gray hue1-3 that is known as the Tyndall effect and can be prevented by injecting the filler at the proper level. For example, collagen-based fillers should be placed at the mid dermis, thicker HA fillers should be placed in the deep dermis, and calcium hydroxylapatite should be placed at the junction of the dermis and subcutaneous tissue. Polymethylmethacrylate and poly-L-lactic acid should both be placed subdermally.1-3
The gravest immediate complications associated with soft-tissue filler injections are occlusion of the central retinal artery and/or skin necrosis.1-4 Residents should never inject filler to the glabella or to the nose.1-3 Injections at these sites are sometimes performed but should only be performed by experienced dermatologists. The perioral and tear trough regions also are high-risk injection areas that require a high degree of experience and should only be injected with proper supervision by an experienced dermatologist.1-3 Residents generally can avoid these complications, though not with a 100% guarantee, by avoiding injections in high-risk areas, aspirating to check for blood, and slowly injecting a small amount of filler into the treatment area.1-3 A consensus statement on management of injection-induced necrosis advises to apply a nitropaste ointment 2% to the treatment site or administer an oral aspirin if the patient develops severe pain; vision loss; or acute skin discoloration, especially blanching.4 For HA-based fillers, at least 200 U of hyaluronidase should be injected. It has been suggested that saline can be injected to flush out calcium hydroxylapatite fillers.3 Warm compresses should be placed on the involved area. Following these interventions, any patient with vision loss or orbital pain should immediately undergo ophthalmologic evaluation.3 The most important intervention occurs in the first 24 hours.3,4 After 24 hours, careful wound care, oral anticoagulants, and hyperbaric oxygen therapy have been suggested as management options.3
There are 2 major chronic complications of soft-tissue filler injection, including delayed-onset infection, which occurs 2 weeks or more postinjection, and granuloma formation.1-3 Chronic low-grade infection at the injection site may be indicative of biofilm formation. If an HA filler was used, it should be dissolved with hyaluronidase to help break up the biofilm nidus.3 A course of oral antibiotics also may be indicated.1-3 Intralesional steroids may be used but only after antibiotics have been administered. A biofilm that develops from more permanent fillers may be more difficult to manage. Atypical mycobacterial infections have been known to develop at injection sites, which should be considered in refractory cases.1-3,5
Calcium hydroxylapatite, polymethylmethacrylate, and silicone can stimulate chronic immune system activation, which makes them more prone to granuloma formation.1-3 Once infection is ruled out, granulomas may be treated with intralesional steroids, surgical excision (if the results would be cosmetically acceptable), laser therapy, or potentially local injection of an immunosuppressant (eg, methotrexate, 5-fluorouracil).3
Botulinum Toxin Injections
Patients who are pregnant, lactating, or have neuromuscular disease are not candidates for botulinum toxin injections. There also is a risk that patients taking calcium channel blockers or aminoglycoside antibiotics may experience potentiated effects of the botulinum toxin.6
Patients should be informed that a postinjection headache may occur and should be treated with over-the-counter medications.6 Complication-free botulinum toxin procedures depend heavily on the physician’s knowledge of facial anatomy.1,6 The diagrams provided by Hirsch and Stier1 offer an excellent guide on where to place the injections. Brow droop, eyelid ptosis, and “Spock brow” (eyebrows that are overarched) largely can be avoided by proper injection point placement. A Spock brow may be corrected by injecting the lateral upper forehead with a few units to correct the exaggerated arch.6,7 For eyelid ptosis, apraclonidine 0.05% drops (1–2 drops 3 times daily) should be used until the ptosis resolves.6 Phenylephrine hydrochloride drops may be used should a patient have a documented sensitivity to apraclonidine but should not be used in a patient with acute angle-closure glaucoma or aneurysms.6
Final Thoughts
Learning to perform soft-tissue augmentation and botulinum toxin injections can be a satisfying and fun part of dermatology residency. Preventing, identifying, and managing any complications that may occur is an integral part of performing these procedures.
The primary cosmetic procedures that dermatology residents will perform or assist in performing during their training are soft-tissue augmentation, botulinum toxin injections, chemical peels, and laser therapy. Because complications can occur from these procedures, it is important for residents to learn how to prevent, identify, and manage them for optimal patient outcomes. In part 1 of this 2-part series, soft-tissue augmentation and botulinum toxin injections are discussed. Chemical peels and laser therapy will be addressed in part 2.
Soft-Tissue Augmentation
Soft-tissue fillers include those that are made from collagen (bovine or human), hyaluronic acid (HA), poly-L-lactic acid, calcium hydroxylapatite, silicone, and polymethylmethacrylate. In general, acute complications of soft-tissue filler injections include erythema, swelling, and bruising.1-3 Patients who take blood thinners or supplements (eg, vitamin E, ginseng, garlic, ginger) should be asked to discontinue use 1 week prior to the procedure. Patients who take blood thinners also should be counseled to expect some bruising. Prior to injection, the skin should be thoroughly cleansed to avoid introducing skin bacteria into the injection site and to reduce infection risk. Postinjection erythema may be related to mast cell activation, which is temporary and should resolve after a few days.1-3
If you find yourself injecting the filler too superficially, you may notice that the skin begins to take on a blue-gray hue1-3 that is known as the Tyndall effect and can be prevented by injecting the filler at the proper level. For example, collagen-based fillers should be placed at the mid dermis, thicker HA fillers should be placed in the deep dermis, and calcium hydroxylapatite should be placed at the junction of the dermis and subcutaneous tissue. Polymethylmethacrylate and poly-L-lactic acid should both be placed subdermally.1-3
The gravest immediate complications associated with soft-tissue filler injections are occlusion of the central retinal artery and/or skin necrosis.1-4 Residents should never inject filler to the glabella or to the nose.1-3 Injections at these sites are sometimes performed but should only be performed by experienced dermatologists. The perioral and tear trough regions also are high-risk injection areas that require a high degree of experience and should only be injected with proper supervision by an experienced dermatologist.1-3 Residents generally can avoid these complications, though not with a 100% guarantee, by avoiding injections in high-risk areas, aspirating to check for blood, and slowly injecting a small amount of filler into the treatment area.1-3 A consensus statement on management of injection-induced necrosis advises to apply a nitropaste ointment 2% to the treatment site or administer an oral aspirin if the patient develops severe pain; vision loss; or acute skin discoloration, especially blanching.4 For HA-based fillers, at least 200 U of hyaluronidase should be injected. It has been suggested that saline can be injected to flush out calcium hydroxylapatite fillers.3 Warm compresses should be placed on the involved area. Following these interventions, any patient with vision loss or orbital pain should immediately undergo ophthalmologic evaluation.3 The most important intervention occurs in the first 24 hours.3,4 After 24 hours, careful wound care, oral anticoagulants, and hyperbaric oxygen therapy have been suggested as management options.3
There are 2 major chronic complications of soft-tissue filler injection, including delayed-onset infection, which occurs 2 weeks or more postinjection, and granuloma formation.1-3 Chronic low-grade infection at the injection site may be indicative of biofilm formation. If an HA filler was used, it should be dissolved with hyaluronidase to help break up the biofilm nidus.3 A course of oral antibiotics also may be indicated.1-3 Intralesional steroids may be used but only after antibiotics have been administered. A biofilm that develops from more permanent fillers may be more difficult to manage. Atypical mycobacterial infections have been known to develop at injection sites, which should be considered in refractory cases.1-3,5
Calcium hydroxylapatite, polymethylmethacrylate, and silicone can stimulate chronic immune system activation, which makes them more prone to granuloma formation.1-3 Once infection is ruled out, granulomas may be treated with intralesional steroids, surgical excision (if the results would be cosmetically acceptable), laser therapy, or potentially local injection of an immunosuppressant (eg, methotrexate, 5-fluorouracil).3
Botulinum Toxin Injections
Patients who are pregnant, lactating, or have neuromuscular disease are not candidates for botulinum toxin injections. There also is a risk that patients taking calcium channel blockers or aminoglycoside antibiotics may experience potentiated effects of the botulinum toxin.6
Patients should be informed that a postinjection headache may occur and should be treated with over-the-counter medications.6 Complication-free botulinum toxin procedures depend heavily on the physician’s knowledge of facial anatomy.1,6 The diagrams provided by Hirsch and Stier1 offer an excellent guide on where to place the injections. Brow droop, eyelid ptosis, and “Spock brow” (eyebrows that are overarched) largely can be avoided by proper injection point placement. A Spock brow may be corrected by injecting the lateral upper forehead with a few units to correct the exaggerated arch.6,7 For eyelid ptosis, apraclonidine 0.05% drops (1–2 drops 3 times daily) should be used until the ptosis resolves.6 Phenylephrine hydrochloride drops may be used should a patient have a documented sensitivity to apraclonidine but should not be used in a patient with acute angle-closure glaucoma or aneurysms.6
Final Thoughts
Learning to perform soft-tissue augmentation and botulinum toxin injections can be a satisfying and fun part of dermatology residency. Preventing, identifying, and managing any complications that may occur is an integral part of performing these procedures.
- Hirsch R, Stier M. Complications and their management in cosmetic dermatology. Dermatol Clin. 2009;27:507-520.
- Gladstone HB, Cohen JL. Adverse effects when injecting facial fillers. Semin Cutan Med Surg. 2007;26:34-39.
- Boulle K, Heydenrych I. Patient factors influencing dermal filler complications: prevention, assessment, and treatment. Clin Cosmet Investig Dermatol. 2015;8:205-214.
- Cohen JL, Biesman BS, Dayan SH, et al. Treatment of hyaluronic acid filler–induced impending necrosis with hyaluronidase: consensus recommendations [published online May 10, 2015]. Aesthet Surg J. 2015;35:844-849.
- Rodriguez JM, Xie YL, Winthrop KL, et al. Mycobacterium chelonae facial infections following injection of dermal filler. Aesthet Surg J. 2013;33:265-269.
- Nigam PK, Nigam A. Botulinum toxin. Indian J Dermatol. 2010;55:8-14.
- Carruthers A, Carruthers J. Update on the botulinum neurotoxins. Skin Therapy Lett. 2001;6:1-2.
- Hirsch R, Stier M. Complications and their management in cosmetic dermatology. Dermatol Clin. 2009;27:507-520.
- Gladstone HB, Cohen JL. Adverse effects when injecting facial fillers. Semin Cutan Med Surg. 2007;26:34-39.
- Boulle K, Heydenrych I. Patient factors influencing dermal filler complications: prevention, assessment, and treatment. Clin Cosmet Investig Dermatol. 2015;8:205-214.
- Cohen JL, Biesman BS, Dayan SH, et al. Treatment of hyaluronic acid filler–induced impending necrosis with hyaluronidase: consensus recommendations [published online May 10, 2015]. Aesthet Surg J. 2015;35:844-849.
- Rodriguez JM, Xie YL, Winthrop KL, et al. Mycobacterium chelonae facial infections following injection of dermal filler. Aesthet Surg J. 2013;33:265-269.
- Nigam PK, Nigam A. Botulinum toxin. Indian J Dermatol. 2010;55:8-14.
- Carruthers A, Carruthers J. Update on the botulinum neurotoxins. Skin Therapy Lett. 2001;6:1-2.
VIDEO: The ins and outs of JAK ihibitors for alopecia
NEWPORT BEACH, CALIF. – The promise of Janus kinase (JAK) inhibitors for alopecia seems to be holding up in the practice of Dr. Natasha Mesinkovska, a dermatologist at the University of California, Irvine.
There’s been much excitement about JAK inhibitors since Yale researchers reported in 2014 that tofacitinib (Xeljanz), a JAK inhibitor approved in the United States for rheumatoid arthritis, appeared to grow a full head of hair, plus body hair, in an essentially hairless 25-year-old man with plaque psoriasis. JAK inhibitors have been under investigation for alopecia ever since. Meanwhile, they are being used off label for hair loss around the country.
In her own practice, Dr. Mesinkovska estimates that about two-thirds of patients have some degree of hair regrowth, with particularly satisfying results in men. About 40 of her alopecia patients have opted for JAK inhibitors so far.
In an interview at the Summit in Aesthetic Medicine, Dr. Mesinkovska shared her insights and tips, as well as promising alopecia results for the psoriasis biologic ustekinumab (Stelara), an interleukin-12 and -23 antagonist. “This is a very exciting time for alopecia areata,” she said.
The Summit in Aesthetic Medicine is held by the Global Academy for Medical Education. Global Academy and this news organization are owned by the same company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
NEWPORT BEACH, CALIF. – The promise of Janus kinase (JAK) inhibitors for alopecia seems to be holding up in the practice of Dr. Natasha Mesinkovska, a dermatologist at the University of California, Irvine.
There’s been much excitement about JAK inhibitors since Yale researchers reported in 2014 that tofacitinib (Xeljanz), a JAK inhibitor approved in the United States for rheumatoid arthritis, appeared to grow a full head of hair, plus body hair, in an essentially hairless 25-year-old man with plaque psoriasis. JAK inhibitors have been under investigation for alopecia ever since. Meanwhile, they are being used off label for hair loss around the country.
In her own practice, Dr. Mesinkovska estimates that about two-thirds of patients have some degree of hair regrowth, with particularly satisfying results in men. About 40 of her alopecia patients have opted for JAK inhibitors so far.
In an interview at the Summit in Aesthetic Medicine, Dr. Mesinkovska shared her insights and tips, as well as promising alopecia results for the psoriasis biologic ustekinumab (Stelara), an interleukin-12 and -23 antagonist. “This is a very exciting time for alopecia areata,” she said.
The Summit in Aesthetic Medicine is held by the Global Academy for Medical Education. Global Academy and this news organization are owned by the same company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
NEWPORT BEACH, CALIF. – The promise of Janus kinase (JAK) inhibitors for alopecia seems to be holding up in the practice of Dr. Natasha Mesinkovska, a dermatologist at the University of California, Irvine.
There’s been much excitement about JAK inhibitors since Yale researchers reported in 2014 that tofacitinib (Xeljanz), a JAK inhibitor approved in the United States for rheumatoid arthritis, appeared to grow a full head of hair, plus body hair, in an essentially hairless 25-year-old man with plaque psoriasis. JAK inhibitors have been under investigation for alopecia ever since. Meanwhile, they are being used off label for hair loss around the country.
In her own practice, Dr. Mesinkovska estimates that about two-thirds of patients have some degree of hair regrowth, with particularly satisfying results in men. About 40 of her alopecia patients have opted for JAK inhibitors so far.
In an interview at the Summit in Aesthetic Medicine, Dr. Mesinkovska shared her insights and tips, as well as promising alopecia results for the psoriasis biologic ustekinumab (Stelara), an interleukin-12 and -23 antagonist. “This is a very exciting time for alopecia areata,” she said.
The Summit in Aesthetic Medicine is held by the Global Academy for Medical Education. Global Academy and this news organization are owned by the same company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
EXPERT ANALYSIS FROM THE SUMMIT IN AESTHETIC MEDICINE
VIDEO: How to treat vascular birthmarks
NEWPORT BEACH, CALIF. – A combination of propranolol and laser is more effective than propranolol alone for infantile hemangiomas, and rapamycin can improve pulse die laser results for port wine stains.
Meanwhile, lasers hurt, so general anesthesia is in order for children as long as they’re older than 6 months.
Those are just a few of the pearls Dr. Kristen Kelly, a University of California, Irvine, professor of dermatology and surgery, shared at the Summit in Aesthetic Medicine. Dr. Kelly explained the latest developments in an interview at the conference, held by Global Academy for Medical Education.
Global Academy and this news organization are owned the same company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
NEWPORT BEACH, CALIF. – A combination of propranolol and laser is more effective than propranolol alone for infantile hemangiomas, and rapamycin can improve pulse die laser results for port wine stains.
Meanwhile, lasers hurt, so general anesthesia is in order for children as long as they’re older than 6 months.
Those are just a few of the pearls Dr. Kristen Kelly, a University of California, Irvine, professor of dermatology and surgery, shared at the Summit in Aesthetic Medicine. Dr. Kelly explained the latest developments in an interview at the conference, held by Global Academy for Medical Education.
Global Academy and this news organization are owned the same company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
NEWPORT BEACH, CALIF. – A combination of propranolol and laser is more effective than propranolol alone for infantile hemangiomas, and rapamycin can improve pulse die laser results for port wine stains.
Meanwhile, lasers hurt, so general anesthesia is in order for children as long as they’re older than 6 months.
Those are just a few of the pearls Dr. Kristen Kelly, a University of California, Irvine, professor of dermatology and surgery, shared at the Summit in Aesthetic Medicine. Dr. Kelly explained the latest developments in an interview at the conference, held by Global Academy for Medical Education.
Global Academy and this news organization are owned the same company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
EXPERT ANALYSIS FROM THE SUMMIT IN AESTHETIC MEDICINE
Cosmetic Corner: Dermatologists Weigh in on Body Scrubs
To improve patient care and outcomes, leading dermatologists offered their recommendations on body scrubs. Consideration must be given to:
- Almond Shower Scrub
- Gently Exfoliating Body Scrub
- Peppermint Body Scrub
- Shea Butter Ultra Rich Body Scrub
- Ultimate Man Body Scrub Soap
Cutis invites readers to send us their recommendations. Body scrubs, OTC acne treatments, and cleansing devices will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on body scrubs. Consideration must be given to:
- Almond Shower Scrub
- Gently Exfoliating Body Scrub
- Peppermint Body Scrub
- Shea Butter Ultra Rich Body Scrub
- Ultimate Man Body Scrub Soap
Cutis invites readers to send us their recommendations. Body scrubs, OTC acne treatments, and cleansing devices will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on body scrubs. Consideration must be given to:
- Almond Shower Scrub
- Gently Exfoliating Body Scrub
- Peppermint Body Scrub
- Shea Butter Ultra Rich Body Scrub
- Ultimate Man Body Scrub Soap
Cutis invites readers to send us their recommendations. Body scrubs, OTC acne treatments, and cleansing devices will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.