User login
Reversing Facial Fillers: How Much Hyaluronidase Is Needed to Dissolve Unwanted Filler?
Hyaluronic acid (HA) fillers are among the most commonly injected soft-tissue fillers worldwide. However, even with proper technique, there may be instances in which the filler becomes visible (Tyndall effect) or edematous, prompting the need to dissolve it. Not all HA fillers are identical; many differ in their degree of cross-linking or classification as monophasic or biphasic, which may affect the success in dissolving them with hyaluronidase. Rao et al (J Drugs Dermatol. 2014;13:1053-1056) published a study that looked at 2 commonly used hyaluronidase agents and how well they dissolved 4 commonly used HA fillers in the United States.
Rao et al performed an in vitro study using Vitrase (ovine testicular hyaluronidase)(Bausch & Lomb Incorporated) and Hylenex (recombinant human hyaluronidase)(Halozyme Therapeutics). The HA fillers tested were Restylane (Galderma Laboratories, LP), Juvéderm and Juvéderm Voluma (Allergan), and Belotero (Merz Aesthetics).
Phase 1 of the study looked at the volume of hyaluronidase on 3 of 4 fillers. The researchers utilized 0.1 mL of Vitrase or Hylenex to treat 0.2 mL of Restylane, Belotero, and Juvéderm. A control slide was kept for each filler. For Vitrase, 0.1 mL is 20 U of hyaluronidase. For Hylenex, 0.1 mL is 15 U of hyaluronidase. The filler and hyaluronidase were mixed together for 10 seconds using a 27-gauge needle. Photograph assessment of the slides was taken at 1 and 5 minutes.
Phase 2 of the study looked at the number of units of hyaluronidase on all 4 fillers. For this phase, 15 U and 30 U of Hylenex were mixed with 0.2 mL aliquots of each filler. Photographs and qualitative observations were taken at 1 and 5 minutes. The aliquots were observed for a total of 15 minutes.
For phase 1, the 2 hyaluronidase agents worked similarly on all 3 HA fillers. The greatest effect on dissolving or changing the shape of a filler was on Restylane, followed by Juvéderm and Belotero (a monophasic filler). The greatest effect was in the first minute and continued through the fifth minute. Even at 5 minutes, Belotero maintained most of its shape.
Phase 2 showed similar results with Restylane reacting the most to the hyaluronidase, followed by Juvéderm Voluma, Juvéderm, and Belotero. The higher dose (30 U) of hyaluronidase had a more dramatic effect on all fillers compared to the lower dose (15 U). Results were time dependent with the greatest changes seen at 5 minutes as opposed to 1 minute. The results of the 15-minute observation did not show any further changes. Belotero also maintained most of its shape during this phase.
What’s the issue?
With the increased number of HA fillers coming onto the market, it will become more important to understand the interaction between HA fillers and hyaluronidase. Hyaluronic acid fillers will vary in their degree of cross-linking, water absorption, and classification as monophasic (cohesive) or biphasic. Oftentimes, it is not until we use fillers in off-label manners that we realize some unintended consequences. We realized all too quickly that fillers, such as Restylane, placed superficially under eyelid skin gave an unsightly Tyndall effect. We then relied on hyaluronidase injections to resolve this issue. Furthermore, we learned that Juvéderm Ultra Plus XC could hold approximately 300% of its weight in water, causing unsightly eyelid edema in some patients. Luckily, the use of hyaluronidase can be a saving grace for physicians. However, the success of hyaluronidase injections varies. From the results of this study, it may be true that certain fillers need higher volumes or a higher number of units than other fillers to dissolve them. I would like to see this study expanded as newer HA fillers are brought onto the market.
Have you found that you have had to use varying amounts of hyaluronidase to address different filler complications?
Hyaluronic acid (HA) fillers are among the most commonly injected soft-tissue fillers worldwide. However, even with proper technique, there may be instances in which the filler becomes visible (Tyndall effect) or edematous, prompting the need to dissolve it. Not all HA fillers are identical; many differ in their degree of cross-linking or classification as monophasic or biphasic, which may affect the success in dissolving them with hyaluronidase. Rao et al (J Drugs Dermatol. 2014;13:1053-1056) published a study that looked at 2 commonly used hyaluronidase agents and how well they dissolved 4 commonly used HA fillers in the United States.
Rao et al performed an in vitro study using Vitrase (ovine testicular hyaluronidase)(Bausch & Lomb Incorporated) and Hylenex (recombinant human hyaluronidase)(Halozyme Therapeutics). The HA fillers tested were Restylane (Galderma Laboratories, LP), Juvéderm and Juvéderm Voluma (Allergan), and Belotero (Merz Aesthetics).
Phase 1 of the study looked at the volume of hyaluronidase on 3 of 4 fillers. The researchers utilized 0.1 mL of Vitrase or Hylenex to treat 0.2 mL of Restylane, Belotero, and Juvéderm. A control slide was kept for each filler. For Vitrase, 0.1 mL is 20 U of hyaluronidase. For Hylenex, 0.1 mL is 15 U of hyaluronidase. The filler and hyaluronidase were mixed together for 10 seconds using a 27-gauge needle. Photograph assessment of the slides was taken at 1 and 5 minutes.
Phase 2 of the study looked at the number of units of hyaluronidase on all 4 fillers. For this phase, 15 U and 30 U of Hylenex were mixed with 0.2 mL aliquots of each filler. Photographs and qualitative observations were taken at 1 and 5 minutes. The aliquots were observed for a total of 15 minutes.
For phase 1, the 2 hyaluronidase agents worked similarly on all 3 HA fillers. The greatest effect on dissolving or changing the shape of a filler was on Restylane, followed by Juvéderm and Belotero (a monophasic filler). The greatest effect was in the first minute and continued through the fifth minute. Even at 5 minutes, Belotero maintained most of its shape.
Phase 2 showed similar results with Restylane reacting the most to the hyaluronidase, followed by Juvéderm Voluma, Juvéderm, and Belotero. The higher dose (30 U) of hyaluronidase had a more dramatic effect on all fillers compared to the lower dose (15 U). Results were time dependent with the greatest changes seen at 5 minutes as opposed to 1 minute. The results of the 15-minute observation did not show any further changes. Belotero also maintained most of its shape during this phase.
What’s the issue?
With the increased number of HA fillers coming onto the market, it will become more important to understand the interaction between HA fillers and hyaluronidase. Hyaluronic acid fillers will vary in their degree of cross-linking, water absorption, and classification as monophasic (cohesive) or biphasic. Oftentimes, it is not until we use fillers in off-label manners that we realize some unintended consequences. We realized all too quickly that fillers, such as Restylane, placed superficially under eyelid skin gave an unsightly Tyndall effect. We then relied on hyaluronidase injections to resolve this issue. Furthermore, we learned that Juvéderm Ultra Plus XC could hold approximately 300% of its weight in water, causing unsightly eyelid edema in some patients. Luckily, the use of hyaluronidase can be a saving grace for physicians. However, the success of hyaluronidase injections varies. From the results of this study, it may be true that certain fillers need higher volumes or a higher number of units than other fillers to dissolve them. I would like to see this study expanded as newer HA fillers are brought onto the market.
Have you found that you have had to use varying amounts of hyaluronidase to address different filler complications?
Hyaluronic acid (HA) fillers are among the most commonly injected soft-tissue fillers worldwide. However, even with proper technique, there may be instances in which the filler becomes visible (Tyndall effect) or edematous, prompting the need to dissolve it. Not all HA fillers are identical; many differ in their degree of cross-linking or classification as monophasic or biphasic, which may affect the success in dissolving them with hyaluronidase. Rao et al (J Drugs Dermatol. 2014;13:1053-1056) published a study that looked at 2 commonly used hyaluronidase agents and how well they dissolved 4 commonly used HA fillers in the United States.
Rao et al performed an in vitro study using Vitrase (ovine testicular hyaluronidase)(Bausch & Lomb Incorporated) and Hylenex (recombinant human hyaluronidase)(Halozyme Therapeutics). The HA fillers tested were Restylane (Galderma Laboratories, LP), Juvéderm and Juvéderm Voluma (Allergan), and Belotero (Merz Aesthetics).
Phase 1 of the study looked at the volume of hyaluronidase on 3 of 4 fillers. The researchers utilized 0.1 mL of Vitrase or Hylenex to treat 0.2 mL of Restylane, Belotero, and Juvéderm. A control slide was kept for each filler. For Vitrase, 0.1 mL is 20 U of hyaluronidase. For Hylenex, 0.1 mL is 15 U of hyaluronidase. The filler and hyaluronidase were mixed together for 10 seconds using a 27-gauge needle. Photograph assessment of the slides was taken at 1 and 5 minutes.
Phase 2 of the study looked at the number of units of hyaluronidase on all 4 fillers. For this phase, 15 U and 30 U of Hylenex were mixed with 0.2 mL aliquots of each filler. Photographs and qualitative observations were taken at 1 and 5 minutes. The aliquots were observed for a total of 15 minutes.
For phase 1, the 2 hyaluronidase agents worked similarly on all 3 HA fillers. The greatest effect on dissolving or changing the shape of a filler was on Restylane, followed by Juvéderm and Belotero (a monophasic filler). The greatest effect was in the first minute and continued through the fifth minute. Even at 5 minutes, Belotero maintained most of its shape.
Phase 2 showed similar results with Restylane reacting the most to the hyaluronidase, followed by Juvéderm Voluma, Juvéderm, and Belotero. The higher dose (30 U) of hyaluronidase had a more dramatic effect on all fillers compared to the lower dose (15 U). Results were time dependent with the greatest changes seen at 5 minutes as opposed to 1 minute. The results of the 15-minute observation did not show any further changes. Belotero also maintained most of its shape during this phase.
What’s the issue?
With the increased number of HA fillers coming onto the market, it will become more important to understand the interaction between HA fillers and hyaluronidase. Hyaluronic acid fillers will vary in their degree of cross-linking, water absorption, and classification as monophasic (cohesive) or biphasic. Oftentimes, it is not until we use fillers in off-label manners that we realize some unintended consequences. We realized all too quickly that fillers, such as Restylane, placed superficially under eyelid skin gave an unsightly Tyndall effect. We then relied on hyaluronidase injections to resolve this issue. Furthermore, we learned that Juvéderm Ultra Plus XC could hold approximately 300% of its weight in water, causing unsightly eyelid edema in some patients. Luckily, the use of hyaluronidase can be a saving grace for physicians. However, the success of hyaluronidase injections varies. From the results of this study, it may be true that certain fillers need higher volumes or a higher number of units than other fillers to dissolve them. I would like to see this study expanded as newer HA fillers are brought onto the market.
Have you found that you have had to use varying amounts of hyaluronidase to address different filler complications?
Laser and electrocautery plume
A recent publication by Chuang et al. carefully characterizes the content of smoke (or plume) created from laser hair removal.1 At the University of California, Los Angeles, discarded terminal hairs from the trunk and extremities were collected from two adult volunteers. The hair samples were sealed in glass gas chromatography chambers and treated with either an 810-nm diode laser (Lightsheer, Lumenis) or 755-nm alexandrite laser (Gentlelase, Candela). During laser hair removal (LHR) treatment, two 6-L negative-pressure canisters were used to capture 30 seconds of laser plume, and a portable condensation particle counter was used to measure ultrafine particulates (less than 1 mcm). Ultrafine particle concentrations were measured within the treatment room, within the waiting room, and outside the building. The laser plume was then analyzed by gas chromatography–mass spectrometry (GC-MS) at the Boston University department of chemistry.
Analysis with GC-MS identified 377 chemical compounds. Sixty-two of the compounds exhibited strong absorption peaks, of which 13 are known or suspected carcinogens (including benzene, ethylbenzene, benzeneacetonitrile, acetonitrile, quinoline, isoquinoline, sterene, diethyl phthalate, 2-methylpyridine, naphthalene carbonitrile, and propene) and more than 20 are known environmental toxins causing acute toxic effects on exposure (including carbon monoxide, p-xylene, phenol, toluene, benzaldehyde, benzenedicarboxylic acid [phthalic acid], and long-chain and cyclic hydrocarbons).
During LHR, the portable condensation particle counters documented an eightfold increase, compared with the ambient room baseline level of ultrafine particle concentrations (ambient room baseline, 15,300 particles per cubic centimeter [ppc]; LHR with smoke evacuator, 129,376 ppc), even when a smoke evacuator was in close proximity (5.0 cm) to the procedure site. When the smoke evacuator was turned off for 30 seconds, there was a more than 26-fold increase in particulate count, compared with ambient baseline levels (ambient baseline, 15,300 ppc; LHR without smoke evacuator for 30 seconds, 435,888 ppc).
It has long been known that smoke created from electrocautery also may impose a risk on the health care worker. In 2011, Lewin et al. published a comprehensive review of the risk that surgical smoke and laser imposes on the dermatologist.2 At this time, most of the laser data was for the plume created by ablative CO2 lasers. In their review, it was stated that surgical smoke is composed of 95% water and 5% particulate matter (made up of chemicals, blood and tissue particles, viruses, and bacteria). The size of the particulate matter is dictated by the device used, with electrosurgical units creating particles of roughly 0.07 mcm and lasers liberating particles of 0.31 mcm. The size of liberated particles is important as those smaller than 100 mcm in diameter remain airborne, and particles less than 2 mcm are deposited in the bronchioles and alveoli.
Electrocautery plume is composed mostly of hydrocarbons, phenols, nitriles, and fatty acids, but most notably carbon monoxide, acrylonitrile, hydrogen cyanide, and benzene, which may have carcinogenic potential. On the mucosa of the canine tongue, the mutagenic effect of the smoke from 1 g of cauterized tissue with laser and electrocautery was equivalent to those from three or six cigarettes, respectively. Pulmonary changes also may occur. Blood vessel hypertrophy, alveolar congestion, and emphysematous changes were seen in rats after plume created from both electrocautery and Nd:Yag ablation of porcine skin. In that study, pulmonary changes were less severe in rats exposed to surgical smoke collected with single- and double-filtered smoke evacuators than unfiltered smoke.
Infection also poses a risk, with bovine papillomavirus and HPV detected in CO2 laser plume as early as 1988. In 1995, Gloster and Roenigk at the Mayo Clinic conducted a comparative study using questionnaires sent to members of the American Society for Laser Surgeons and the American Society of Dermatologic Surgery.3 The comparison groups were CO2 laser surgeons and two large groups of patients in the community with a diagnosis of warts. Analysis revealed that CO2 laser surgeons had a statistically significant greater risk of acquiring nasopharyngeal warts but were less likely to acquire plantar, genital, and perianal warts than the Mayo Clinic patient group did, demonstrating that laser plume is a likely means by which HPV can be transmitted to the upper airway, suggesting that those using lasers to treat HPV lesions are at greater risk. Staphylococcus, Corynebacterium, and Neisseria also have been detected during laser resurfacing.
Traditional surgical masks are able to capture particles greater than 5 mcm but offer no protection against particulate matter produced by electrosurgical and laser devices liberating byproducts less than 1 mcm. Laser masks or high-filtration masks provide greater protection than do standard surgical masks and are able to filter particles to 1.1 mcm; however, it has been shown that approximately 77% of particulate matter in surgical smoke is 1.1 mcm and smaller. Smoke evacuators consist of a suction unit (vacuum pump), filter, hose, and inlet nozzle. The smoke evacuator should have a capture velocity of approximately 30-45 m/min at the inlet nozzle. As the effectiveness of the smoke evacuator decreases with farther distance away from the procedure site, the current study of laser plume from LHR recommends that the smoke evacuator be placed within 5 cm of plume generation.4 In a study of warts treated with CO2 laser or electrocoagulation, smoke evacuators are 98.6% effective when placed 1 cm from the treatment site, with efficacy decreasing to 50% when moved to 2 cm from the treatment site.
As our specialty likely conducts the highest proportion of laser and electrosurgical procedures of any specialty, current recommendations for electrocautery, laser resurfacing, and LHR should include adequate air filtration and the use of high-filtration masks and smoke evacuators by the health care practitioner.
References
1. JAMA Dermatol. 2016 Jul 6. doi: 10.1001/jamadermatol.2016.2097. [Epub ahead of print]
2. J Am Acad Dermatol. 2011 Sep;65(3):636-41.
3. J Am Acad Dermatol. 1995 Mar;32(3):436-41.
4. J Am Acad Dermatol. 1989 Jul;21(1):41-9.
Dr. Wesley and Dr. Talakoub are co-contributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Wesley. Write to them at [email protected].
A recent publication by Chuang et al. carefully characterizes the content of smoke (or plume) created from laser hair removal.1 At the University of California, Los Angeles, discarded terminal hairs from the trunk and extremities were collected from two adult volunteers. The hair samples were sealed in glass gas chromatography chambers and treated with either an 810-nm diode laser (Lightsheer, Lumenis) or 755-nm alexandrite laser (Gentlelase, Candela). During laser hair removal (LHR) treatment, two 6-L negative-pressure canisters were used to capture 30 seconds of laser plume, and a portable condensation particle counter was used to measure ultrafine particulates (less than 1 mcm). Ultrafine particle concentrations were measured within the treatment room, within the waiting room, and outside the building. The laser plume was then analyzed by gas chromatography–mass spectrometry (GC-MS) at the Boston University department of chemistry.
Analysis with GC-MS identified 377 chemical compounds. Sixty-two of the compounds exhibited strong absorption peaks, of which 13 are known or suspected carcinogens (including benzene, ethylbenzene, benzeneacetonitrile, acetonitrile, quinoline, isoquinoline, sterene, diethyl phthalate, 2-methylpyridine, naphthalene carbonitrile, and propene) and more than 20 are known environmental toxins causing acute toxic effects on exposure (including carbon monoxide, p-xylene, phenol, toluene, benzaldehyde, benzenedicarboxylic acid [phthalic acid], and long-chain and cyclic hydrocarbons).
During LHR, the portable condensation particle counters documented an eightfold increase, compared with the ambient room baseline level of ultrafine particle concentrations (ambient room baseline, 15,300 particles per cubic centimeter [ppc]; LHR with smoke evacuator, 129,376 ppc), even when a smoke evacuator was in close proximity (5.0 cm) to the procedure site. When the smoke evacuator was turned off for 30 seconds, there was a more than 26-fold increase in particulate count, compared with ambient baseline levels (ambient baseline, 15,300 ppc; LHR without smoke evacuator for 30 seconds, 435,888 ppc).
It has long been known that smoke created from electrocautery also may impose a risk on the health care worker. In 2011, Lewin et al. published a comprehensive review of the risk that surgical smoke and laser imposes on the dermatologist.2 At this time, most of the laser data was for the plume created by ablative CO2 lasers. In their review, it was stated that surgical smoke is composed of 95% water and 5% particulate matter (made up of chemicals, blood and tissue particles, viruses, and bacteria). The size of the particulate matter is dictated by the device used, with electrosurgical units creating particles of roughly 0.07 mcm and lasers liberating particles of 0.31 mcm. The size of liberated particles is important as those smaller than 100 mcm in diameter remain airborne, and particles less than 2 mcm are deposited in the bronchioles and alveoli.
Electrocautery plume is composed mostly of hydrocarbons, phenols, nitriles, and fatty acids, but most notably carbon monoxide, acrylonitrile, hydrogen cyanide, and benzene, which may have carcinogenic potential. On the mucosa of the canine tongue, the mutagenic effect of the smoke from 1 g of cauterized tissue with laser and electrocautery was equivalent to those from three or six cigarettes, respectively. Pulmonary changes also may occur. Blood vessel hypertrophy, alveolar congestion, and emphysematous changes were seen in rats after plume created from both electrocautery and Nd:Yag ablation of porcine skin. In that study, pulmonary changes were less severe in rats exposed to surgical smoke collected with single- and double-filtered smoke evacuators than unfiltered smoke.
Infection also poses a risk, with bovine papillomavirus and HPV detected in CO2 laser plume as early as 1988. In 1995, Gloster and Roenigk at the Mayo Clinic conducted a comparative study using questionnaires sent to members of the American Society for Laser Surgeons and the American Society of Dermatologic Surgery.3 The comparison groups were CO2 laser surgeons and two large groups of patients in the community with a diagnosis of warts. Analysis revealed that CO2 laser surgeons had a statistically significant greater risk of acquiring nasopharyngeal warts but were less likely to acquire plantar, genital, and perianal warts than the Mayo Clinic patient group did, demonstrating that laser plume is a likely means by which HPV can be transmitted to the upper airway, suggesting that those using lasers to treat HPV lesions are at greater risk. Staphylococcus, Corynebacterium, and Neisseria also have been detected during laser resurfacing.
Traditional surgical masks are able to capture particles greater than 5 mcm but offer no protection against particulate matter produced by electrosurgical and laser devices liberating byproducts less than 1 mcm. Laser masks or high-filtration masks provide greater protection than do standard surgical masks and are able to filter particles to 1.1 mcm; however, it has been shown that approximately 77% of particulate matter in surgical smoke is 1.1 mcm and smaller. Smoke evacuators consist of a suction unit (vacuum pump), filter, hose, and inlet nozzle. The smoke evacuator should have a capture velocity of approximately 30-45 m/min at the inlet nozzle. As the effectiveness of the smoke evacuator decreases with farther distance away from the procedure site, the current study of laser plume from LHR recommends that the smoke evacuator be placed within 5 cm of plume generation.4 In a study of warts treated with CO2 laser or electrocoagulation, smoke evacuators are 98.6% effective when placed 1 cm from the treatment site, with efficacy decreasing to 50% when moved to 2 cm from the treatment site.
As our specialty likely conducts the highest proportion of laser and electrosurgical procedures of any specialty, current recommendations for electrocautery, laser resurfacing, and LHR should include adequate air filtration and the use of high-filtration masks and smoke evacuators by the health care practitioner.
References
1. JAMA Dermatol. 2016 Jul 6. doi: 10.1001/jamadermatol.2016.2097. [Epub ahead of print]
2. J Am Acad Dermatol. 2011 Sep;65(3):636-41.
3. J Am Acad Dermatol. 1995 Mar;32(3):436-41.
4. J Am Acad Dermatol. 1989 Jul;21(1):41-9.
Dr. Wesley and Dr. Talakoub are co-contributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Wesley. Write to them at [email protected].
A recent publication by Chuang et al. carefully characterizes the content of smoke (or plume) created from laser hair removal.1 At the University of California, Los Angeles, discarded terminal hairs from the trunk and extremities were collected from two adult volunteers. The hair samples were sealed in glass gas chromatography chambers and treated with either an 810-nm diode laser (Lightsheer, Lumenis) or 755-nm alexandrite laser (Gentlelase, Candela). During laser hair removal (LHR) treatment, two 6-L negative-pressure canisters were used to capture 30 seconds of laser plume, and a portable condensation particle counter was used to measure ultrafine particulates (less than 1 mcm). Ultrafine particle concentrations were measured within the treatment room, within the waiting room, and outside the building. The laser plume was then analyzed by gas chromatography–mass spectrometry (GC-MS) at the Boston University department of chemistry.
Analysis with GC-MS identified 377 chemical compounds. Sixty-two of the compounds exhibited strong absorption peaks, of which 13 are known or suspected carcinogens (including benzene, ethylbenzene, benzeneacetonitrile, acetonitrile, quinoline, isoquinoline, sterene, diethyl phthalate, 2-methylpyridine, naphthalene carbonitrile, and propene) and more than 20 are known environmental toxins causing acute toxic effects on exposure (including carbon monoxide, p-xylene, phenol, toluene, benzaldehyde, benzenedicarboxylic acid [phthalic acid], and long-chain and cyclic hydrocarbons).
During LHR, the portable condensation particle counters documented an eightfold increase, compared with the ambient room baseline level of ultrafine particle concentrations (ambient room baseline, 15,300 particles per cubic centimeter [ppc]; LHR with smoke evacuator, 129,376 ppc), even when a smoke evacuator was in close proximity (5.0 cm) to the procedure site. When the smoke evacuator was turned off for 30 seconds, there was a more than 26-fold increase in particulate count, compared with ambient baseline levels (ambient baseline, 15,300 ppc; LHR without smoke evacuator for 30 seconds, 435,888 ppc).
It has long been known that smoke created from electrocautery also may impose a risk on the health care worker. In 2011, Lewin et al. published a comprehensive review of the risk that surgical smoke and laser imposes on the dermatologist.2 At this time, most of the laser data was for the plume created by ablative CO2 lasers. In their review, it was stated that surgical smoke is composed of 95% water and 5% particulate matter (made up of chemicals, blood and tissue particles, viruses, and bacteria). The size of the particulate matter is dictated by the device used, with electrosurgical units creating particles of roughly 0.07 mcm and lasers liberating particles of 0.31 mcm. The size of liberated particles is important as those smaller than 100 mcm in diameter remain airborne, and particles less than 2 mcm are deposited in the bronchioles and alveoli.
Electrocautery plume is composed mostly of hydrocarbons, phenols, nitriles, and fatty acids, but most notably carbon monoxide, acrylonitrile, hydrogen cyanide, and benzene, which may have carcinogenic potential. On the mucosa of the canine tongue, the mutagenic effect of the smoke from 1 g of cauterized tissue with laser and electrocautery was equivalent to those from three or six cigarettes, respectively. Pulmonary changes also may occur. Blood vessel hypertrophy, alveolar congestion, and emphysematous changes were seen in rats after plume created from both electrocautery and Nd:Yag ablation of porcine skin. In that study, pulmonary changes were less severe in rats exposed to surgical smoke collected with single- and double-filtered smoke evacuators than unfiltered smoke.
Infection also poses a risk, with bovine papillomavirus and HPV detected in CO2 laser plume as early as 1988. In 1995, Gloster and Roenigk at the Mayo Clinic conducted a comparative study using questionnaires sent to members of the American Society for Laser Surgeons and the American Society of Dermatologic Surgery.3 The comparison groups were CO2 laser surgeons and two large groups of patients in the community with a diagnosis of warts. Analysis revealed that CO2 laser surgeons had a statistically significant greater risk of acquiring nasopharyngeal warts but were less likely to acquire plantar, genital, and perianal warts than the Mayo Clinic patient group did, demonstrating that laser plume is a likely means by which HPV can be transmitted to the upper airway, suggesting that those using lasers to treat HPV lesions are at greater risk. Staphylococcus, Corynebacterium, and Neisseria also have been detected during laser resurfacing.
Traditional surgical masks are able to capture particles greater than 5 mcm but offer no protection against particulate matter produced by electrosurgical and laser devices liberating byproducts less than 1 mcm. Laser masks or high-filtration masks provide greater protection than do standard surgical masks and are able to filter particles to 1.1 mcm; however, it has been shown that approximately 77% of particulate matter in surgical smoke is 1.1 mcm and smaller. Smoke evacuators consist of a suction unit (vacuum pump), filter, hose, and inlet nozzle. The smoke evacuator should have a capture velocity of approximately 30-45 m/min at the inlet nozzle. As the effectiveness of the smoke evacuator decreases with farther distance away from the procedure site, the current study of laser plume from LHR recommends that the smoke evacuator be placed within 5 cm of plume generation.4 In a study of warts treated with CO2 laser or electrocoagulation, smoke evacuators are 98.6% effective when placed 1 cm from the treatment site, with efficacy decreasing to 50% when moved to 2 cm from the treatment site.
As our specialty likely conducts the highest proportion of laser and electrosurgical procedures of any specialty, current recommendations for electrocautery, laser resurfacing, and LHR should include adequate air filtration and the use of high-filtration masks and smoke evacuators by the health care practitioner.
References
1. JAMA Dermatol. 2016 Jul 6. doi: 10.1001/jamadermatol.2016.2097. [Epub ahead of print]
2. J Am Acad Dermatol. 2011 Sep;65(3):636-41.
3. J Am Acad Dermatol. 1995 Mar;32(3):436-41.
4. J Am Acad Dermatol. 1989 Jul;21(1):41-9.
Dr. Wesley and Dr. Talakoub are co-contributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Wesley. Write to them at [email protected].
Expert advocates single pass CO2 laser resurfacing
NEWPORT BEACH, CALIF. – Facial CO2 laser resurfacing deserves a comeback, according to Victor Ross, MD, director of laser and cosmetic dermatology at the Scripps Clinic in San Diego.
The practice fell out of favor in the late 1990s because of long recovery times, hypopigmentation, and the risk of keloid scars, among other issues. Physicians were just being too aggressive, doing multiple full-field passes in one session, he said at the Summit in Aesthetic Medicine, held by Global Academy for Medical Education.
It turns out that doing one pass very conservatively – with maybe a second pass around the mouth for deeper wrinkles – delivers a lot of the benefits with none of the downsides. It takes maybe 45 minutes, and “we get very nice results, I think better than fractional [laser] results,” Dr. Ross said. The wrinkle-smoothing effect may not be as potent or durable as the old-school approach, “but it’s a more natural look and [there’s] much faster recovery. Patients go pink instead of red,” and can wear makeup sooner. “You don’t get delayed hypopigmentation.”
The general trend in cosmetic dermatology is to do multiple procedures in one office visit instead of spacing them out over several appointments. For instance, lentigines on the hand could be targeted with intense pulsed light and then hand crepiness could be treated with a fractional laser. You “can get a lot done and a very nice result in one” session, but it makes sense to dial settings back maybe 15%-20% when different devices are used on the same area, he said.
I keep several lasers in one room” and sometimes “go back and forth, back and forth like a mad chef,” he said.
Companies are helping further the approach. One company, for instance, has added a nonablative fractional laser to its intense pulsed light platform. Others are combining ablative and nonablative fractional lasers. It’s all about “allowing you to have more flexibility in how you deliver energy,” Dr. Ross said.
Meanwhile, another newer kid on the block, the picosecond laser, has helped a bit with tattoo removal, but “we are still not really hitting a home run. There’s no doubt that picosecond lasers are more efficient than nanosecond lasers, but they’re maybe 20%-30% better,” he said. The problem is that most commercially available picosecond lasers have pulse durations on the order of 500-800 picoseconds, which is not optimal for the smallest pigment particles. If pulse durations are too short and energies too high, “you get this kind of white plasma in the skin, which doesn’t” help.
Industry hasn’t solved the problem yet; for now “I would give us a C-plus on tattoos,” he said.
Dr. Ross had some advice for dermatologists looking to outfit a new cosmetic practice. Right off the bat, “you will need something for red and brown spots, because you are going to see a lot of that.” An intense pulsed light device or a large-spot potassium titanyl phosphate (KTP) laser fills the bill, he said.
He added that he would have a device for resurfacing, and as the third device, “I would get maybe a Q-switch laser for tattoos,” he said.
So armed, a dermatologist could handle much of what’s likely to come through the door.
Dr. Ross works with a number of companies, including Lutronic, Cynosure, and Ellipse. Global Academy and this news organization are owned by the same company.
NEWPORT BEACH, CALIF. – Facial CO2 laser resurfacing deserves a comeback, according to Victor Ross, MD, director of laser and cosmetic dermatology at the Scripps Clinic in San Diego.
The practice fell out of favor in the late 1990s because of long recovery times, hypopigmentation, and the risk of keloid scars, among other issues. Physicians were just being too aggressive, doing multiple full-field passes in one session, he said at the Summit in Aesthetic Medicine, held by Global Academy for Medical Education.
It turns out that doing one pass very conservatively – with maybe a second pass around the mouth for deeper wrinkles – delivers a lot of the benefits with none of the downsides. It takes maybe 45 minutes, and “we get very nice results, I think better than fractional [laser] results,” Dr. Ross said. The wrinkle-smoothing effect may not be as potent or durable as the old-school approach, “but it’s a more natural look and [there’s] much faster recovery. Patients go pink instead of red,” and can wear makeup sooner. “You don’t get delayed hypopigmentation.”
The general trend in cosmetic dermatology is to do multiple procedures in one office visit instead of spacing them out over several appointments. For instance, lentigines on the hand could be targeted with intense pulsed light and then hand crepiness could be treated with a fractional laser. You “can get a lot done and a very nice result in one” session, but it makes sense to dial settings back maybe 15%-20% when different devices are used on the same area, he said.
I keep several lasers in one room” and sometimes “go back and forth, back and forth like a mad chef,” he said.
Companies are helping further the approach. One company, for instance, has added a nonablative fractional laser to its intense pulsed light platform. Others are combining ablative and nonablative fractional lasers. It’s all about “allowing you to have more flexibility in how you deliver energy,” Dr. Ross said.
Meanwhile, another newer kid on the block, the picosecond laser, has helped a bit with tattoo removal, but “we are still not really hitting a home run. There’s no doubt that picosecond lasers are more efficient than nanosecond lasers, but they’re maybe 20%-30% better,” he said. The problem is that most commercially available picosecond lasers have pulse durations on the order of 500-800 picoseconds, which is not optimal for the smallest pigment particles. If pulse durations are too short and energies too high, “you get this kind of white plasma in the skin, which doesn’t” help.
Industry hasn’t solved the problem yet; for now “I would give us a C-plus on tattoos,” he said.
Dr. Ross had some advice for dermatologists looking to outfit a new cosmetic practice. Right off the bat, “you will need something for red and brown spots, because you are going to see a lot of that.” An intense pulsed light device or a large-spot potassium titanyl phosphate (KTP) laser fills the bill, he said.
He added that he would have a device for resurfacing, and as the third device, “I would get maybe a Q-switch laser for tattoos,” he said.
So armed, a dermatologist could handle much of what’s likely to come through the door.
Dr. Ross works with a number of companies, including Lutronic, Cynosure, and Ellipse. Global Academy and this news organization are owned by the same company.
NEWPORT BEACH, CALIF. – Facial CO2 laser resurfacing deserves a comeback, according to Victor Ross, MD, director of laser and cosmetic dermatology at the Scripps Clinic in San Diego.
The practice fell out of favor in the late 1990s because of long recovery times, hypopigmentation, and the risk of keloid scars, among other issues. Physicians were just being too aggressive, doing multiple full-field passes in one session, he said at the Summit in Aesthetic Medicine, held by Global Academy for Medical Education.
It turns out that doing one pass very conservatively – with maybe a second pass around the mouth for deeper wrinkles – delivers a lot of the benefits with none of the downsides. It takes maybe 45 minutes, and “we get very nice results, I think better than fractional [laser] results,” Dr. Ross said. The wrinkle-smoothing effect may not be as potent or durable as the old-school approach, “but it’s a more natural look and [there’s] much faster recovery. Patients go pink instead of red,” and can wear makeup sooner. “You don’t get delayed hypopigmentation.”
The general trend in cosmetic dermatology is to do multiple procedures in one office visit instead of spacing them out over several appointments. For instance, lentigines on the hand could be targeted with intense pulsed light and then hand crepiness could be treated with a fractional laser. You “can get a lot done and a very nice result in one” session, but it makes sense to dial settings back maybe 15%-20% when different devices are used on the same area, he said.
I keep several lasers in one room” and sometimes “go back and forth, back and forth like a mad chef,” he said.
Companies are helping further the approach. One company, for instance, has added a nonablative fractional laser to its intense pulsed light platform. Others are combining ablative and nonablative fractional lasers. It’s all about “allowing you to have more flexibility in how you deliver energy,” Dr. Ross said.
Meanwhile, another newer kid on the block, the picosecond laser, has helped a bit with tattoo removal, but “we are still not really hitting a home run. There’s no doubt that picosecond lasers are more efficient than nanosecond lasers, but they’re maybe 20%-30% better,” he said. The problem is that most commercially available picosecond lasers have pulse durations on the order of 500-800 picoseconds, which is not optimal for the smallest pigment particles. If pulse durations are too short and energies too high, “you get this kind of white plasma in the skin, which doesn’t” help.
Industry hasn’t solved the problem yet; for now “I would give us a C-plus on tattoos,” he said.
Dr. Ross had some advice for dermatologists looking to outfit a new cosmetic practice. Right off the bat, “you will need something for red and brown spots, because you are going to see a lot of that.” An intense pulsed light device or a large-spot potassium titanyl phosphate (KTP) laser fills the bill, he said.
He added that he would have a device for resurfacing, and as the third device, “I would get maybe a Q-switch laser for tattoos,” he said.
So armed, a dermatologist could handle much of what’s likely to come through the door.
Dr. Ross works with a number of companies, including Lutronic, Cynosure, and Ellipse. Global Academy and this news organization are owned by the same company.
EXPERT ANALYSIS FROM THE SUMMIT IN AESTHETIC MEDICINE
Lasers and topicals both lighten solar lentigines
Quality-switched (QS) laser therapy with a ruby laser and topical triple-combination therapy (TCT) were similarly effective in lightening solar lentigines, based on data from a prospective, open label study of 15 adults. The findings were published in Dermatologic Surgery.
The QS ruby laser (QSRL) delivered longer-lasting skin-lightening results in less time, but the topical therapy is less expensive and has a lower risk of adverse events, wrote Dr. Laurence Imhof of the department of dermatology, University Hospital Zürich, and colleagues (Dermatol Surg. 2016;42:853-57. doi: 10.1097/DSS.0000000000000793). “Although therapy [for solar lentigines] is not medically indicated, there is a rising treatment demand for aesthetic reasons. Therefore, the treatment should be very safe and affordable,” they wrote.
The researchers compared the safety and efficacy of treating solar lentigines with a 694-nm QSRL and TCT (hydroquinone 5%, tretinoin 0.03%, and dexamethasone 0.03%). The patients included 14 women and 1 man with evenly distributed solar lentigines on the backs of both hands. Each patient was treated with 1-2 QSRL sessions on the back of the right hand and 7 weeks of once-daily application of TCT on the back of the left hand. The average age of the patients was 61 years; 13 were Fitzpatrick Skin Types II and III. Adverse events were mild and transient with both treatments, although the QSRL caused significantly more crusting and hyperpigmentation than the TCT.
Both treatments significantly reduced pigment at the end of treatment (day 56) and 12 weeks’ post treatment (140 days), compared with baseline. Treatment areas were evaluated at days 28, 56, and 140 by the treating physician, the patient, and a blinded physician (based on photos).The 6-point grading scale rated the degree of lightening/percentage of clearing, ranging from worse (less than 0%) to excellent (76%-100%).
At 56 days, the mean macroscopic improvement with QSRL laser was rated 3.5 by the treating physician and patient and 3 by a blinded physician; the mean macroscopic improvement with TCT was rated 2.4 by the treating physician, 2.2 by the patient, and 1.7 by the blinded physician. At 140 days, the mean macroscopic improvements with the laser were rated 2.9 by the treating physician, 2.7 by the patient, and 2.8 by the blinded physician, compared with 1.2, 1.1, and 0.7, respectively, with TCT.
The results were limited by the small size, short follow-up period, and open-label design, the researchers noted. However, the study is the first known to compare a laser and a topical treatment for the management of solar lentigines, and the findings suggest that both therapies can be recommended for the aesthetic treatment of solar lentigines, they said.
The TCT cream used in the study was provided by Louis Widmer SA, which provided a grant to the University of Zürich. The researchers had no financial conflicts to disclose.

|
Dr. Nazanin Saedi |
“We commend Imhof and colleagues for providing a long-awaited direct comparison of the Q-switched laser and topical triple-combination therapy,” wrote Dr. Laura M. Schilling and Dr. Nazanin Saedi, in an accompanying editorial (Dermatol Surg. 2016;42:8580859. doi: 10.1097/DSS.0000000000000792). Both treatments are viable options for patients, they emphasized. “It is fundamental, especially in this era of patient-centered care, to recognize that treatment options must be individualized. There are numerous variables to be considered when choosing the appropriate treatment for a patient including efficacy, skin type, cost, availability, and patient preference, among others.”
Dr. Schilling and Dr. Saedi are with the department of dermatology and cutaneous biology, Thomas Jefferson University, Philadelphia. They had no financial conflicts to disclose.

|
Dr. Nazanin Saedi |
“We commend Imhof and colleagues for providing a long-awaited direct comparison of the Q-switched laser and topical triple-combination therapy,” wrote Dr. Laura M. Schilling and Dr. Nazanin Saedi, in an accompanying editorial (Dermatol Surg. 2016;42:8580859. doi: 10.1097/DSS.0000000000000792). Both treatments are viable options for patients, they emphasized. “It is fundamental, especially in this era of patient-centered care, to recognize that treatment options must be individualized. There are numerous variables to be considered when choosing the appropriate treatment for a patient including efficacy, skin type, cost, availability, and patient preference, among others.”
Dr. Schilling and Dr. Saedi are with the department of dermatology and cutaneous biology, Thomas Jefferson University, Philadelphia. They had no financial conflicts to disclose.

|
Dr. Nazanin Saedi |
“We commend Imhof and colleagues for providing a long-awaited direct comparison of the Q-switched laser and topical triple-combination therapy,” wrote Dr. Laura M. Schilling and Dr. Nazanin Saedi, in an accompanying editorial (Dermatol Surg. 2016;42:8580859. doi: 10.1097/DSS.0000000000000792). Both treatments are viable options for patients, they emphasized. “It is fundamental, especially in this era of patient-centered care, to recognize that treatment options must be individualized. There are numerous variables to be considered when choosing the appropriate treatment for a patient including efficacy, skin type, cost, availability, and patient preference, among others.”
Dr. Schilling and Dr. Saedi are with the department of dermatology and cutaneous biology, Thomas Jefferson University, Philadelphia. They had no financial conflicts to disclose.
Quality-switched (QS) laser therapy with a ruby laser and topical triple-combination therapy (TCT) were similarly effective in lightening solar lentigines, based on data from a prospective, open label study of 15 adults. The findings were published in Dermatologic Surgery.
The QS ruby laser (QSRL) delivered longer-lasting skin-lightening results in less time, but the topical therapy is less expensive and has a lower risk of adverse events, wrote Dr. Laurence Imhof of the department of dermatology, University Hospital Zürich, and colleagues (Dermatol Surg. 2016;42:853-57. doi: 10.1097/DSS.0000000000000793). “Although therapy [for solar lentigines] is not medically indicated, there is a rising treatment demand for aesthetic reasons. Therefore, the treatment should be very safe and affordable,” they wrote.
The researchers compared the safety and efficacy of treating solar lentigines with a 694-nm QSRL and TCT (hydroquinone 5%, tretinoin 0.03%, and dexamethasone 0.03%). The patients included 14 women and 1 man with evenly distributed solar lentigines on the backs of both hands. Each patient was treated with 1-2 QSRL sessions on the back of the right hand and 7 weeks of once-daily application of TCT on the back of the left hand. The average age of the patients was 61 years; 13 were Fitzpatrick Skin Types II and III. Adverse events were mild and transient with both treatments, although the QSRL caused significantly more crusting and hyperpigmentation than the TCT.
Both treatments significantly reduced pigment at the end of treatment (day 56) and 12 weeks’ post treatment (140 days), compared with baseline. Treatment areas were evaluated at days 28, 56, and 140 by the treating physician, the patient, and a blinded physician (based on photos).The 6-point grading scale rated the degree of lightening/percentage of clearing, ranging from worse (less than 0%) to excellent (76%-100%).
At 56 days, the mean macroscopic improvement with QSRL laser was rated 3.5 by the treating physician and patient and 3 by a blinded physician; the mean macroscopic improvement with TCT was rated 2.4 by the treating physician, 2.2 by the patient, and 1.7 by the blinded physician. At 140 days, the mean macroscopic improvements with the laser were rated 2.9 by the treating physician, 2.7 by the patient, and 2.8 by the blinded physician, compared with 1.2, 1.1, and 0.7, respectively, with TCT.
The results were limited by the small size, short follow-up period, and open-label design, the researchers noted. However, the study is the first known to compare a laser and a topical treatment for the management of solar lentigines, and the findings suggest that both therapies can be recommended for the aesthetic treatment of solar lentigines, they said.
The TCT cream used in the study was provided by Louis Widmer SA, which provided a grant to the University of Zürich. The researchers had no financial conflicts to disclose.
Quality-switched (QS) laser therapy with a ruby laser and topical triple-combination therapy (TCT) were similarly effective in lightening solar lentigines, based on data from a prospective, open label study of 15 adults. The findings were published in Dermatologic Surgery.
The QS ruby laser (QSRL) delivered longer-lasting skin-lightening results in less time, but the topical therapy is less expensive and has a lower risk of adverse events, wrote Dr. Laurence Imhof of the department of dermatology, University Hospital Zürich, and colleagues (Dermatol Surg. 2016;42:853-57. doi: 10.1097/DSS.0000000000000793). “Although therapy [for solar lentigines] is not medically indicated, there is a rising treatment demand for aesthetic reasons. Therefore, the treatment should be very safe and affordable,” they wrote.
The researchers compared the safety and efficacy of treating solar lentigines with a 694-nm QSRL and TCT (hydroquinone 5%, tretinoin 0.03%, and dexamethasone 0.03%). The patients included 14 women and 1 man with evenly distributed solar lentigines on the backs of both hands. Each patient was treated with 1-2 QSRL sessions on the back of the right hand and 7 weeks of once-daily application of TCT on the back of the left hand. The average age of the patients was 61 years; 13 were Fitzpatrick Skin Types II and III. Adverse events were mild and transient with both treatments, although the QSRL caused significantly more crusting and hyperpigmentation than the TCT.
Both treatments significantly reduced pigment at the end of treatment (day 56) and 12 weeks’ post treatment (140 days), compared with baseline. Treatment areas were evaluated at days 28, 56, and 140 by the treating physician, the patient, and a blinded physician (based on photos).The 6-point grading scale rated the degree of lightening/percentage of clearing, ranging from worse (less than 0%) to excellent (76%-100%).
At 56 days, the mean macroscopic improvement with QSRL laser was rated 3.5 by the treating physician and patient and 3 by a blinded physician; the mean macroscopic improvement with TCT was rated 2.4 by the treating physician, 2.2 by the patient, and 1.7 by the blinded physician. At 140 days, the mean macroscopic improvements with the laser were rated 2.9 by the treating physician, 2.7 by the patient, and 2.8 by the blinded physician, compared with 1.2, 1.1, and 0.7, respectively, with TCT.
The results were limited by the small size, short follow-up period, and open-label design, the researchers noted. However, the study is the first known to compare a laser and a topical treatment for the management of solar lentigines, and the findings suggest that both therapies can be recommended for the aesthetic treatment of solar lentigines, they said.
The TCT cream used in the study was provided by Louis Widmer SA, which provided a grant to the University of Zürich. The researchers had no financial conflicts to disclose.
FROM DERMATOLOGIC SURGERY
Key clinical point: Both treatments significantly reduced pigment at the study conclusion (day 56) and at 12 weeks post treatment, compared with baseline.
Major finding: The mean macroscopic improvement with QSRL laser at 56 days was rated 3.5 by the treating physician and patient and 3 by a blinded physician; the mean macroscopic improvement with TCT was rated 2.4 by the treating physician, 2.2 by the patient, and 1.7 by the blinded physician.
Data source: A prospective, open-label 20-week study of 15 patients with solar lentigines.
Disclosures: The TCT cream used in the study was provided by Louis Widmer SA, which provided a grant to the University of Zürich. The researchers had no financial conflicts to disclose.
Sunscreens May Fail to Meet SPF Claims on Product Labels
New data from Consumer Reports indicate that 48% of all sunscreens tested (N=104) over 4 years did not provide the sun protection factor (SPF) promised on product labels, leaving consumers with insufficient sun protection, which could lead to long-term sun damage including wrinkles or skin cancer. Furthermore, 42% of chemical sunscreens (n=85) and 74% of mineral sunscreens (n=19) did not meet their SPF claims.
The study also reveals that more than one-third (35%) of sunscreens registered below SPF 30, which is the minimum recommended by the American Academy of Dermatology (AAD). Although product labels featured claims of water resistance, nearly half of the sunscreens tested failed to meet their SPF claim following water immersion.
Dermatologists can educate patients about correct sunscreen use and product labels to ensure the highest level of protection against melanoma and other skin cancers. According to a 2016 AAD survey, only 32% of respondents knew that an SPF 30 sunscreen does not provide twice as much protection as an SPF 15 sunscreen. Furthermore, only 45% of respondents knew that a higher-SPF sunscreen does not protect skin from sun exposure longer than a lower-SPF sunscreen.
The AAD has issued a list of talking points highlighting key messages that dermatologists can share with patients and/or the media when asked about sun-protection techniques and the recent sunscreen data released by Consumer Reports.
New data from Consumer Reports indicate that 48% of all sunscreens tested (N=104) over 4 years did not provide the sun protection factor (SPF) promised on product labels, leaving consumers with insufficient sun protection, which could lead to long-term sun damage including wrinkles or skin cancer. Furthermore, 42% of chemical sunscreens (n=85) and 74% of mineral sunscreens (n=19) did not meet their SPF claims.
The study also reveals that more than one-third (35%) of sunscreens registered below SPF 30, which is the minimum recommended by the American Academy of Dermatology (AAD). Although product labels featured claims of water resistance, nearly half of the sunscreens tested failed to meet their SPF claim following water immersion.
Dermatologists can educate patients about correct sunscreen use and product labels to ensure the highest level of protection against melanoma and other skin cancers. According to a 2016 AAD survey, only 32% of respondents knew that an SPF 30 sunscreen does not provide twice as much protection as an SPF 15 sunscreen. Furthermore, only 45% of respondents knew that a higher-SPF sunscreen does not protect skin from sun exposure longer than a lower-SPF sunscreen.
The AAD has issued a list of talking points highlighting key messages that dermatologists can share with patients and/or the media when asked about sun-protection techniques and the recent sunscreen data released by Consumer Reports.
New data from Consumer Reports indicate that 48% of all sunscreens tested (N=104) over 4 years did not provide the sun protection factor (SPF) promised on product labels, leaving consumers with insufficient sun protection, which could lead to long-term sun damage including wrinkles or skin cancer. Furthermore, 42% of chemical sunscreens (n=85) and 74% of mineral sunscreens (n=19) did not meet their SPF claims.
The study also reveals that more than one-third (35%) of sunscreens registered below SPF 30, which is the minimum recommended by the American Academy of Dermatology (AAD). Although product labels featured claims of water resistance, nearly half of the sunscreens tested failed to meet their SPF claim following water immersion.
Dermatologists can educate patients about correct sunscreen use and product labels to ensure the highest level of protection against melanoma and other skin cancers. According to a 2016 AAD survey, only 32% of respondents knew that an SPF 30 sunscreen does not provide twice as much protection as an SPF 15 sunscreen. Furthermore, only 45% of respondents knew that a higher-SPF sunscreen does not protect skin from sun exposure longer than a lower-SPF sunscreen.
The AAD has issued a list of talking points highlighting key messages that dermatologists can share with patients and/or the media when asked about sun-protection techniques and the recent sunscreen data released by Consumer Reports.
Update on green tea
During the last 25 years, green tea, which is derived from Camellia sinensis (an evergreen member of the Theaceae family), has gained considerable attention because of its purported antioxidant and anticarcinogenic properties. Believed to have been used by human beings for 4,000 years,1 green tea is now one of the most heavily researched of the antioxidants, with numerous studies of its cutaneous effects appearing in the literature.2 Laden with plant polyphenols, orally administered or topically applied green tea has been shown to display significant antioxidant, chemopreventive, immunomodulatory, and anti-inflammatory activity, affecting the biochemical pathways important in cell proliferation.3-6 For this reason, and due to its global popularity as a beverage, green tea polyphenols are among the most frequently studied herbal agents used in medicine.
Polyphenols, many of which are potent antioxidants, are a large diverse family of thousands of chemical compounds present in plants. The four major polyphenolic catechins present in green tea include: ECG [(-)EpiCatechin-3-O-Gallate], GCG [(-)GalloCatechin-3-O-Gallate], EGC [(-)EpiGalloCatechin], and EGCG [(-)EpiGalloCatechin-3-O-Gallate], the most abundant and biologically active green tea constituent. In fact, EGCG is the component associated with the greatest anticarcinogenic and chemopreventive properties.6
A wide-ranging evidence-based review of the use of botanicals in dermatology, published in 2010, showed that the oral administration, in particular, as well as topical application of antioxidant plant extracts of green tea, among other botanicals, can protect skin against the harmful effects of UV exposure, including erythema, premature aging, and cancer.7
Green tea is thought to be challenging to formulate because of the inherent hydrophilicity of EGCG, which limits penetration into human skin.8,9 Nevertheless, green tea is thought to have great potential in traditional sunscreens to enhance photoprotection.10,11 The photoprotective activity of orally administered or topically applied green tea has been supported in various studies.12-15
The remainder of this column will focus on recent studies of topically applied green tea polyphenols in human beings as well as clinical uses of this agent.
Topical uses
Topical green tea appears to reduce skin inflammation and neutralize free radicals, which explains its popularity as an additive in rosacea and antiaging skin care products. The antiaging effects of green tea are difficult to measure because it functions as an antioxidant that prevents aging and does not have the capacity to increase collagen synthesis or ameliorate already existing wrinkles. However, there is relatively good evidence, in comparison to other antioxidants, suggesting that topically applied green tea can help protect skin from UV radiation.16
Investigators performed a thorough literature search of all in vitro, in vivo, and controlled clinical trials involving green tea formulations and their dermatologic applications, which was published in 2012. They evaluated 20 studies, with evidence suggesting that orally administered green tea displays a broad range of healthy activity, and supportive data for the use of topically applied green tea extract for treatment of various cutaneous conditions, including acne, rosacea, atopic dermatitis, androgenetic alopecia, hirsutism, candidiasis, keloids, leishmaniasis, and genital warts.17
Also, a green tea topical formulation, green tea sinecatechin Polyphenon E (Veregen) ointment, has recently been shown to exert antioxidant, antiviral, and antitumor activity, and has demonstrated efficacy in treating Condylomata acuminata (external anogenital warts).18 In addition, topically applying green tea catechins in the morning in combination with traditional sunscreens is believed to have the potential to protect the skin from UV-induced damage. Topical green tea may improve rosacea, prevent retinoid dermatitis, and play a role in managing pigmentation disorders. Few of the many over-the-counter products that contain green tea catechins have been tested in controlled clinical trials and the concentration of polyphenols in these products is too low to demonstrate efficacy. It is necessary to know the amount of green tea catechins in a formulation to judge its efficacy.
Acne
In 2009, in a 6-week study investigating the efficacy of 2% green tea lotion for the treatment of mild-to-moderate acne vulgaris in 20 patients, researchers reported statistically significant reductions in mean total lesion count and mean severity index (devised by the authors to correlate with total lesion count in increasing intensity, scaled from 1 to 3). They concluded that 2% green tea lotion is both an effective and cost effective approach for treating mild-to-moderate acne lesions.19
A 2012 study revealed that ethanol extracts of several herbs, including green tea, exhibited the potential for inhibiting acne when incorporated into a topical moisturizer, specifically acting against acne-causing bacteria without provoking irritation.20 Earlier that year, other investigators conducted in vitro and in vivo experiments to evaluate the effects against acne of polyphenon-60, which contains various green tea catechins (now referred to as sinecatechins in the United States.).21 In this clinical study, patients exhibited improvement in acne symptoms, including a reduction in the number of pustules and comedones.22
A study published in 2013, a single-blind, placebo-controlled, split-face comparative study in 22 individuals over 60 days, evaluated the efficacy of green tea, as well as green tea plus lotus, compared with placebo for controlling casual sebum secretions in healthy adults. Compared with placebo, consistent and statistically significant decreases in sebum secretions were observed in both treatment groups. The combination of green tea and lotus extracts also achieved statistically sounder results than green tea alone. The investigators concluded that a synergistic interaction between green tea and lotus extract constituents appears to hold promise for the treatment of skin conditions in which elevated sebum levels are involved.23
Anogenital warts
In 2006, the Food and Drug Administration approved for the first time a botanical drug formulation for the topical treatment of genital and perianal warts: sinecatechins, derived from green tea catechins and other C. sinensis constituents in a topical 15% ointment (Veregen).21, 24-28
Two years later, Tatti et al. conducted a randomized, double-blind, vehicle-controlled trial to evaluate the efficacy of topical sinecatechins in 502 male and female patients (aged 18 years and older) for the treatment of anogenital warts. For 16 weeks or until complete clearance, subjects applied sinecatechins ointment 15% or 10% or vehicle (placebo) three times daily. Complete clearance was achieved in 57.2% of patients treated with 15% ointment, 56.3% using 10% ointment, and 33.7% who used only the vehicle. Respective recurrence rates, after 12 weeks, were 6.5%, 8.3%, and 8.8%. The investigators concluded that topical sinecatechins in 15% and 10% concentrations represent effective and well-tolerated options for anogenital wart treatment.29
Similarly favorable results regarding polyphenon E 15% were reported in reference to three placebo-controlled clinical studies in 1,400 patients with genital warts from Europe, North and South America, and South Africa,30,31 and by Tatti et al. again in 2010 after randomized, double-blind, vehicle-controlled safety and efficacy trials in nearly 1,000 patients treated with polyphenon E 15% and 10% formulations.21
Two years later, investigators evaluated sinecatechins (Polyphenon E) 10% ointment in two double-blind, multinational studies in adults with external genital and perianal warts. Polyphenon E 10% was found to be significantly more effective than vehicle in completely or partially clearing all warts.32
Earlier that year, a review of the use of sinecatechins ointment for the treatment of external anogenital warts noted that while clearance rates are similar among sinecatechins and other indicated topical medications such as imiquimod and podophyllotoxin, recurrence rates are lower for patients treated with sinecatechins. The authors concluded that the use of sinecatechins for condylomata acuminata was safe and effective and its various molecular activities suggest broader applications to other viral and tumor lesions.33
In 2015, Gupta and Daigle reported that sinecatechins 10% ointment for the treatment of external genital warts was found in phase III trials to display greater efficacy and lower rates of recurrence in comparison to patient-applied treatments now available.28 Later that year, in a systematic PubMed and Embase review of clinical trials involving the use of polyphenol-based therapies, Tuong et al. identified cogent evidence suggesting the effectiveness of green tea polyphenols for the treatment of anogenital warts.34
Antiaging activity
Green tea has been shown to work in combination with red light to exert a rejuvenating effect on the skin, as Sommer and Zhu reported in 2009 that green tea filled cotton pads applied once daily for 20 minutes prior to treatment with light-emitting diodes (central wavelength 670 nm, dermal dose 4 J/cm2) reduced wrinkles in 1 month comparably to 10 months of light treatment alone.35
In 2013, Hong et al. studied the antiwrinkle effects of topically applied green tea extract with high antioxidant activity after tannase treatment. Study participants were randomly divided to receive either green tea extract or tannase-converted green tea extract on their crow’s feet for an 8-week period. The investigators found that tannase treatment elevated the antioxidant activity of green tea and imparted antiwrinkle effects.36
At around the same time, Gianeti conducted clinical studies in 24 volunteers to assess the effects of a cosmetic formulation containing 6% C. sinensis glycolic leaf extracts. Skin moisture was enhanced after 30 days of topical application as was the viscoelastic-to-elastic ratio compared with vehicle and control (a forearm area left untreated). Skin roughness was significantly diminished after 30 days. The investigators concluded that the topical cosmetic formulation with green tea yielded salient moisturizing and cutaneous microrelief benefits.37
Also in 2013, oral intake of green tea catechins in 16 healthy human subjects (with 14 completing the study) appeared to result in the integration of catechin metabolites into human skin linked to the negation of UV-induced 12-hydroxyeicosatetraenoic acid (12-HETE). The investigators speculated that this incorporation of catechins may render protection against sunburn inflammation and even cumulative UV-induced harm.38
After earlier showing the efficacy of green tea and lotus extracts in skin disorders involving excess sebum in a single-blinded, placebo-controlled, split-face comparative study,23 Mahmood and Akhtar conducted a 60-day placebo-controlled comparative split-face study in 33 healthy Asian men to evaluate the efficacy of two cosmetic formulations (green tea and lotus extract) for facial wrinkles. All of the formulations yielded improvements in skin roughness, scaliness, smoothness, and wrinkling, with the greatest reduction in wrinkling conferred by the combination formulation. The investigators concluded that the synergistic activity of green tea and lotus extracts exerted significant improvement along several skin parameters, suggesting the potential for these ingredients in antiaging products.38
In 2014, the synergistic effects of green tea and ginkgo biloba were explored in preclinical and clinical studies. In the clinical study, 48 participants applied the formulations on forearm skin and were evaluated before and after 3 hours and following 15- and 30-day use periods. Results showed a moisturizing effect and enhancement in skin microrelief, as well as improvements in skin elasticity and barrier function.3
Conclusion
Green tea remains one of the most researched antioxidants as benefits from its use continue to emerge. Indeed, green tea polyphenols are in use for a growing number of indications, especially acne and anogenital warts, and there is reason for optimism that topically applied green tea will gain momentum as an increasingly selected therapeutic option. More clinical studies are necessary to further establish the potential role of green tea for a wider range of cutaneous indications. Green tea holds particular promise in relation to photoprotection against UV-induced skin cancer and skin aging.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. She also developed and owns the Baumann Skin Type Solution skin typing systems and related products
References:
1. Cancer Lett. 1997 Mar 19;114(1-2):315-7.
2. J Am Acad Dermatol. 2005 Jun;52(6):1049-59.
3. Arch Dermatol. 2000 Aug;136(8):989-94.
4. Photochem Photobiol. 1995 Nov;62(5):855-61.
5. Oxid Med Cell Longev. 2012:2012:560682.
6. J Dtsch Dermatol Ges. 2015 Aug;13(8):768-75.
7. Am J Clin Dermatol. 2010;11(4):247-67.
8. Dermatol Ther. 2007 Sep-Oct;20(5):322-9.
9. J Clin Aesthet Dermatol. 2010 Feb;3(2):22-41.
10. Photodermatol Photoimmunol Photomed. 2007 Feb;23(1):48-56.
11. Skin Res Technol. 2009 Aug;15(3):338-45.
12. Exp Dermatol. 2009 Jan;18(1):69-77.
13. Exp Dermatol. 2009 Jun;18(6):522-6.
14. Arch Biochem Biophys. 2011 Apr 15;508(2):152-8.
15. Cancer Prev Res (Phila). 2010 Feb;3(2):179-89.
16. Complement Ther Clin Pract. 2014 Feb;20(1):11-5.
17. Skinmed. 2012 Nov-Dec;10(6):352-5.
18. J Eur Acad Dermatol Venereol. 2011 Mar;25(3):345-53.
19. J Drugs Dermatol. 2009 Apr;8(4):358-64.
20. Pak J Pharm Sci. 2012 Oct;25(4):867-70.
21. Br J Dermatol. 2010 Jan;162(1):176-84.
22. Arch Dermatol Res. 2012 Oct;304(8):655-63.
23. Hippokratia. 2013 Jan;17(1):64-7.
24. Food Chem Toxicol. 2008 Aug;46(8):2606-10.
25. Nat Biotechnol. 2008 Oct;26(10):1077-83.
26. Skin Therapy Lett. 2012 Apr;17(4):5-7.
27. J Clin Aesthet Dermatol. 2012 Jan;5(1):19-26.
28. Skin Therapy Lett. 2015 Jan-Feb;20(1):6-8.
29. Obstet Gynecol. 2008 Jun;111(6):1371-9.
30. Hautarzt. 2008 Jan;59(1):31-5.
31. J Eur Acad Dermatol Venereol. 2007 Nov;21(10):1404-12.
32. Am J Clin Dermatol. 2012 Aug 1:13(4):275-81.
33. Expert Opin Biol Ther. 2012 Jun;12(6):783-93.
34. J Dermatolog Treat. 2015;26(4):381-8.
35. Photomed Laser Surg. 2009 Dec;27(6):969-71.
36. J Cosmet Dermatol. 2013 Jun;12(2):137-43.
37. Dermatol Ther. 2013 May-Jun;26(3):267-71.
During the last 25 years, green tea, which is derived from Camellia sinensis (an evergreen member of the Theaceae family), has gained considerable attention because of its purported antioxidant and anticarcinogenic properties. Believed to have been used by human beings for 4,000 years,1 green tea is now one of the most heavily researched of the antioxidants, with numerous studies of its cutaneous effects appearing in the literature.2 Laden with plant polyphenols, orally administered or topically applied green tea has been shown to display significant antioxidant, chemopreventive, immunomodulatory, and anti-inflammatory activity, affecting the biochemical pathways important in cell proliferation.3-6 For this reason, and due to its global popularity as a beverage, green tea polyphenols are among the most frequently studied herbal agents used in medicine.
Polyphenols, many of which are potent antioxidants, are a large diverse family of thousands of chemical compounds present in plants. The four major polyphenolic catechins present in green tea include: ECG [(-)EpiCatechin-3-O-Gallate], GCG [(-)GalloCatechin-3-O-Gallate], EGC [(-)EpiGalloCatechin], and EGCG [(-)EpiGalloCatechin-3-O-Gallate], the most abundant and biologically active green tea constituent. In fact, EGCG is the component associated with the greatest anticarcinogenic and chemopreventive properties.6
A wide-ranging evidence-based review of the use of botanicals in dermatology, published in 2010, showed that the oral administration, in particular, as well as topical application of antioxidant plant extracts of green tea, among other botanicals, can protect skin against the harmful effects of UV exposure, including erythema, premature aging, and cancer.7
Green tea is thought to be challenging to formulate because of the inherent hydrophilicity of EGCG, which limits penetration into human skin.8,9 Nevertheless, green tea is thought to have great potential in traditional sunscreens to enhance photoprotection.10,11 The photoprotective activity of orally administered or topically applied green tea has been supported in various studies.12-15
The remainder of this column will focus on recent studies of topically applied green tea polyphenols in human beings as well as clinical uses of this agent.
Topical uses
Topical green tea appears to reduce skin inflammation and neutralize free radicals, which explains its popularity as an additive in rosacea and antiaging skin care products. The antiaging effects of green tea are difficult to measure because it functions as an antioxidant that prevents aging and does not have the capacity to increase collagen synthesis or ameliorate already existing wrinkles. However, there is relatively good evidence, in comparison to other antioxidants, suggesting that topically applied green tea can help protect skin from UV radiation.16
Investigators performed a thorough literature search of all in vitro, in vivo, and controlled clinical trials involving green tea formulations and their dermatologic applications, which was published in 2012. They evaluated 20 studies, with evidence suggesting that orally administered green tea displays a broad range of healthy activity, and supportive data for the use of topically applied green tea extract for treatment of various cutaneous conditions, including acne, rosacea, atopic dermatitis, androgenetic alopecia, hirsutism, candidiasis, keloids, leishmaniasis, and genital warts.17
Also, a green tea topical formulation, green tea sinecatechin Polyphenon E (Veregen) ointment, has recently been shown to exert antioxidant, antiviral, and antitumor activity, and has demonstrated efficacy in treating Condylomata acuminata (external anogenital warts).18 In addition, topically applying green tea catechins in the morning in combination with traditional sunscreens is believed to have the potential to protect the skin from UV-induced damage. Topical green tea may improve rosacea, prevent retinoid dermatitis, and play a role in managing pigmentation disorders. Few of the many over-the-counter products that contain green tea catechins have been tested in controlled clinical trials and the concentration of polyphenols in these products is too low to demonstrate efficacy. It is necessary to know the amount of green tea catechins in a formulation to judge its efficacy.
Acne
In 2009, in a 6-week study investigating the efficacy of 2% green tea lotion for the treatment of mild-to-moderate acne vulgaris in 20 patients, researchers reported statistically significant reductions in mean total lesion count and mean severity index (devised by the authors to correlate with total lesion count in increasing intensity, scaled from 1 to 3). They concluded that 2% green tea lotion is both an effective and cost effective approach for treating mild-to-moderate acne lesions.19
A 2012 study revealed that ethanol extracts of several herbs, including green tea, exhibited the potential for inhibiting acne when incorporated into a topical moisturizer, specifically acting against acne-causing bacteria without provoking irritation.20 Earlier that year, other investigators conducted in vitro and in vivo experiments to evaluate the effects against acne of polyphenon-60, which contains various green tea catechins (now referred to as sinecatechins in the United States.).21 In this clinical study, patients exhibited improvement in acne symptoms, including a reduction in the number of pustules and comedones.22
A study published in 2013, a single-blind, placebo-controlled, split-face comparative study in 22 individuals over 60 days, evaluated the efficacy of green tea, as well as green tea plus lotus, compared with placebo for controlling casual sebum secretions in healthy adults. Compared with placebo, consistent and statistically significant decreases in sebum secretions were observed in both treatment groups. The combination of green tea and lotus extracts also achieved statistically sounder results than green tea alone. The investigators concluded that a synergistic interaction between green tea and lotus extract constituents appears to hold promise for the treatment of skin conditions in which elevated sebum levels are involved.23
Anogenital warts
In 2006, the Food and Drug Administration approved for the first time a botanical drug formulation for the topical treatment of genital and perianal warts: sinecatechins, derived from green tea catechins and other C. sinensis constituents in a topical 15% ointment (Veregen).21, 24-28
Two years later, Tatti et al. conducted a randomized, double-blind, vehicle-controlled trial to evaluate the efficacy of topical sinecatechins in 502 male and female patients (aged 18 years and older) for the treatment of anogenital warts. For 16 weeks or until complete clearance, subjects applied sinecatechins ointment 15% or 10% or vehicle (placebo) three times daily. Complete clearance was achieved in 57.2% of patients treated with 15% ointment, 56.3% using 10% ointment, and 33.7% who used only the vehicle. Respective recurrence rates, after 12 weeks, were 6.5%, 8.3%, and 8.8%. The investigators concluded that topical sinecatechins in 15% and 10% concentrations represent effective and well-tolerated options for anogenital wart treatment.29
Similarly favorable results regarding polyphenon E 15% were reported in reference to three placebo-controlled clinical studies in 1,400 patients with genital warts from Europe, North and South America, and South Africa,30,31 and by Tatti et al. again in 2010 after randomized, double-blind, vehicle-controlled safety and efficacy trials in nearly 1,000 patients treated with polyphenon E 15% and 10% formulations.21
Two years later, investigators evaluated sinecatechins (Polyphenon E) 10% ointment in two double-blind, multinational studies in adults with external genital and perianal warts. Polyphenon E 10% was found to be significantly more effective than vehicle in completely or partially clearing all warts.32
Earlier that year, a review of the use of sinecatechins ointment for the treatment of external anogenital warts noted that while clearance rates are similar among sinecatechins and other indicated topical medications such as imiquimod and podophyllotoxin, recurrence rates are lower for patients treated with sinecatechins. The authors concluded that the use of sinecatechins for condylomata acuminata was safe and effective and its various molecular activities suggest broader applications to other viral and tumor lesions.33
In 2015, Gupta and Daigle reported that sinecatechins 10% ointment for the treatment of external genital warts was found in phase III trials to display greater efficacy and lower rates of recurrence in comparison to patient-applied treatments now available.28 Later that year, in a systematic PubMed and Embase review of clinical trials involving the use of polyphenol-based therapies, Tuong et al. identified cogent evidence suggesting the effectiveness of green tea polyphenols for the treatment of anogenital warts.34
Antiaging activity
Green tea has been shown to work in combination with red light to exert a rejuvenating effect on the skin, as Sommer and Zhu reported in 2009 that green tea filled cotton pads applied once daily for 20 minutes prior to treatment with light-emitting diodes (central wavelength 670 nm, dermal dose 4 J/cm2) reduced wrinkles in 1 month comparably to 10 months of light treatment alone.35
In 2013, Hong et al. studied the antiwrinkle effects of topically applied green tea extract with high antioxidant activity after tannase treatment. Study participants were randomly divided to receive either green tea extract or tannase-converted green tea extract on their crow’s feet for an 8-week period. The investigators found that tannase treatment elevated the antioxidant activity of green tea and imparted antiwrinkle effects.36
At around the same time, Gianeti conducted clinical studies in 24 volunteers to assess the effects of a cosmetic formulation containing 6% C. sinensis glycolic leaf extracts. Skin moisture was enhanced after 30 days of topical application as was the viscoelastic-to-elastic ratio compared with vehicle and control (a forearm area left untreated). Skin roughness was significantly diminished after 30 days. The investigators concluded that the topical cosmetic formulation with green tea yielded salient moisturizing and cutaneous microrelief benefits.37
Also in 2013, oral intake of green tea catechins in 16 healthy human subjects (with 14 completing the study) appeared to result in the integration of catechin metabolites into human skin linked to the negation of UV-induced 12-hydroxyeicosatetraenoic acid (12-HETE). The investigators speculated that this incorporation of catechins may render protection against sunburn inflammation and even cumulative UV-induced harm.38
After earlier showing the efficacy of green tea and lotus extracts in skin disorders involving excess sebum in a single-blinded, placebo-controlled, split-face comparative study,23 Mahmood and Akhtar conducted a 60-day placebo-controlled comparative split-face study in 33 healthy Asian men to evaluate the efficacy of two cosmetic formulations (green tea and lotus extract) for facial wrinkles. All of the formulations yielded improvements in skin roughness, scaliness, smoothness, and wrinkling, with the greatest reduction in wrinkling conferred by the combination formulation. The investigators concluded that the synergistic activity of green tea and lotus extracts exerted significant improvement along several skin parameters, suggesting the potential for these ingredients in antiaging products.38
In 2014, the synergistic effects of green tea and ginkgo biloba were explored in preclinical and clinical studies. In the clinical study, 48 participants applied the formulations on forearm skin and were evaluated before and after 3 hours and following 15- and 30-day use periods. Results showed a moisturizing effect and enhancement in skin microrelief, as well as improvements in skin elasticity and barrier function.3
Conclusion
Green tea remains one of the most researched antioxidants as benefits from its use continue to emerge. Indeed, green tea polyphenols are in use for a growing number of indications, especially acne and anogenital warts, and there is reason for optimism that topically applied green tea will gain momentum as an increasingly selected therapeutic option. More clinical studies are necessary to further establish the potential role of green tea for a wider range of cutaneous indications. Green tea holds particular promise in relation to photoprotection against UV-induced skin cancer and skin aging.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. She also developed and owns the Baumann Skin Type Solution skin typing systems and related products
References:
1. Cancer Lett. 1997 Mar 19;114(1-2):315-7.
2. J Am Acad Dermatol. 2005 Jun;52(6):1049-59.
3. Arch Dermatol. 2000 Aug;136(8):989-94.
4. Photochem Photobiol. 1995 Nov;62(5):855-61.
5. Oxid Med Cell Longev. 2012:2012:560682.
6. J Dtsch Dermatol Ges. 2015 Aug;13(8):768-75.
7. Am J Clin Dermatol. 2010;11(4):247-67.
8. Dermatol Ther. 2007 Sep-Oct;20(5):322-9.
9. J Clin Aesthet Dermatol. 2010 Feb;3(2):22-41.
10. Photodermatol Photoimmunol Photomed. 2007 Feb;23(1):48-56.
11. Skin Res Technol. 2009 Aug;15(3):338-45.
12. Exp Dermatol. 2009 Jan;18(1):69-77.
13. Exp Dermatol. 2009 Jun;18(6):522-6.
14. Arch Biochem Biophys. 2011 Apr 15;508(2):152-8.
15. Cancer Prev Res (Phila). 2010 Feb;3(2):179-89.
16. Complement Ther Clin Pract. 2014 Feb;20(1):11-5.
17. Skinmed. 2012 Nov-Dec;10(6):352-5.
18. J Eur Acad Dermatol Venereol. 2011 Mar;25(3):345-53.
19. J Drugs Dermatol. 2009 Apr;8(4):358-64.
20. Pak J Pharm Sci. 2012 Oct;25(4):867-70.
21. Br J Dermatol. 2010 Jan;162(1):176-84.
22. Arch Dermatol Res. 2012 Oct;304(8):655-63.
23. Hippokratia. 2013 Jan;17(1):64-7.
24. Food Chem Toxicol. 2008 Aug;46(8):2606-10.
25. Nat Biotechnol. 2008 Oct;26(10):1077-83.
26. Skin Therapy Lett. 2012 Apr;17(4):5-7.
27. J Clin Aesthet Dermatol. 2012 Jan;5(1):19-26.
28. Skin Therapy Lett. 2015 Jan-Feb;20(1):6-8.
29. Obstet Gynecol. 2008 Jun;111(6):1371-9.
30. Hautarzt. 2008 Jan;59(1):31-5.
31. J Eur Acad Dermatol Venereol. 2007 Nov;21(10):1404-12.
32. Am J Clin Dermatol. 2012 Aug 1:13(4):275-81.
33. Expert Opin Biol Ther. 2012 Jun;12(6):783-93.
34. J Dermatolog Treat. 2015;26(4):381-8.
35. Photomed Laser Surg. 2009 Dec;27(6):969-71.
36. J Cosmet Dermatol. 2013 Jun;12(2):137-43.
37. Dermatol Ther. 2013 May-Jun;26(3):267-71.
During the last 25 years, green tea, which is derived from Camellia sinensis (an evergreen member of the Theaceae family), has gained considerable attention because of its purported antioxidant and anticarcinogenic properties. Believed to have been used by human beings for 4,000 years,1 green tea is now one of the most heavily researched of the antioxidants, with numerous studies of its cutaneous effects appearing in the literature.2 Laden with plant polyphenols, orally administered or topically applied green tea has been shown to display significant antioxidant, chemopreventive, immunomodulatory, and anti-inflammatory activity, affecting the biochemical pathways important in cell proliferation.3-6 For this reason, and due to its global popularity as a beverage, green tea polyphenols are among the most frequently studied herbal agents used in medicine.
Polyphenols, many of which are potent antioxidants, are a large diverse family of thousands of chemical compounds present in plants. The four major polyphenolic catechins present in green tea include: ECG [(-)EpiCatechin-3-O-Gallate], GCG [(-)GalloCatechin-3-O-Gallate], EGC [(-)EpiGalloCatechin], and EGCG [(-)EpiGalloCatechin-3-O-Gallate], the most abundant and biologically active green tea constituent. In fact, EGCG is the component associated with the greatest anticarcinogenic and chemopreventive properties.6
A wide-ranging evidence-based review of the use of botanicals in dermatology, published in 2010, showed that the oral administration, in particular, as well as topical application of antioxidant plant extracts of green tea, among other botanicals, can protect skin against the harmful effects of UV exposure, including erythema, premature aging, and cancer.7
Green tea is thought to be challenging to formulate because of the inherent hydrophilicity of EGCG, which limits penetration into human skin.8,9 Nevertheless, green tea is thought to have great potential in traditional sunscreens to enhance photoprotection.10,11 The photoprotective activity of orally administered or topically applied green tea has been supported in various studies.12-15
The remainder of this column will focus on recent studies of topically applied green tea polyphenols in human beings as well as clinical uses of this agent.
Topical uses
Topical green tea appears to reduce skin inflammation and neutralize free radicals, which explains its popularity as an additive in rosacea and antiaging skin care products. The antiaging effects of green tea are difficult to measure because it functions as an antioxidant that prevents aging and does not have the capacity to increase collagen synthesis or ameliorate already existing wrinkles. However, there is relatively good evidence, in comparison to other antioxidants, suggesting that topically applied green tea can help protect skin from UV radiation.16
Investigators performed a thorough literature search of all in vitro, in vivo, and controlled clinical trials involving green tea formulations and their dermatologic applications, which was published in 2012. They evaluated 20 studies, with evidence suggesting that orally administered green tea displays a broad range of healthy activity, and supportive data for the use of topically applied green tea extract for treatment of various cutaneous conditions, including acne, rosacea, atopic dermatitis, androgenetic alopecia, hirsutism, candidiasis, keloids, leishmaniasis, and genital warts.17
Also, a green tea topical formulation, green tea sinecatechin Polyphenon E (Veregen) ointment, has recently been shown to exert antioxidant, antiviral, and antitumor activity, and has demonstrated efficacy in treating Condylomata acuminata (external anogenital warts).18 In addition, topically applying green tea catechins in the morning in combination with traditional sunscreens is believed to have the potential to protect the skin from UV-induced damage. Topical green tea may improve rosacea, prevent retinoid dermatitis, and play a role in managing pigmentation disorders. Few of the many over-the-counter products that contain green tea catechins have been tested in controlled clinical trials and the concentration of polyphenols in these products is too low to demonstrate efficacy. It is necessary to know the amount of green tea catechins in a formulation to judge its efficacy.
Acne
In 2009, in a 6-week study investigating the efficacy of 2% green tea lotion for the treatment of mild-to-moderate acne vulgaris in 20 patients, researchers reported statistically significant reductions in mean total lesion count and mean severity index (devised by the authors to correlate with total lesion count in increasing intensity, scaled from 1 to 3). They concluded that 2% green tea lotion is both an effective and cost effective approach for treating mild-to-moderate acne lesions.19
A 2012 study revealed that ethanol extracts of several herbs, including green tea, exhibited the potential for inhibiting acne when incorporated into a topical moisturizer, specifically acting against acne-causing bacteria without provoking irritation.20 Earlier that year, other investigators conducted in vitro and in vivo experiments to evaluate the effects against acne of polyphenon-60, which contains various green tea catechins (now referred to as sinecatechins in the United States.).21 In this clinical study, patients exhibited improvement in acne symptoms, including a reduction in the number of pustules and comedones.22
A study published in 2013, a single-blind, placebo-controlled, split-face comparative study in 22 individuals over 60 days, evaluated the efficacy of green tea, as well as green tea plus lotus, compared with placebo for controlling casual sebum secretions in healthy adults. Compared with placebo, consistent and statistically significant decreases in sebum secretions were observed in both treatment groups. The combination of green tea and lotus extracts also achieved statistically sounder results than green tea alone. The investigators concluded that a synergistic interaction between green tea and lotus extract constituents appears to hold promise for the treatment of skin conditions in which elevated sebum levels are involved.23
Anogenital warts
In 2006, the Food and Drug Administration approved for the first time a botanical drug formulation for the topical treatment of genital and perianal warts: sinecatechins, derived from green tea catechins and other C. sinensis constituents in a topical 15% ointment (Veregen).21, 24-28
Two years later, Tatti et al. conducted a randomized, double-blind, vehicle-controlled trial to evaluate the efficacy of topical sinecatechins in 502 male and female patients (aged 18 years and older) for the treatment of anogenital warts. For 16 weeks or until complete clearance, subjects applied sinecatechins ointment 15% or 10% or vehicle (placebo) three times daily. Complete clearance was achieved in 57.2% of patients treated with 15% ointment, 56.3% using 10% ointment, and 33.7% who used only the vehicle. Respective recurrence rates, after 12 weeks, were 6.5%, 8.3%, and 8.8%. The investigators concluded that topical sinecatechins in 15% and 10% concentrations represent effective and well-tolerated options for anogenital wart treatment.29
Similarly favorable results regarding polyphenon E 15% were reported in reference to three placebo-controlled clinical studies in 1,400 patients with genital warts from Europe, North and South America, and South Africa,30,31 and by Tatti et al. again in 2010 after randomized, double-blind, vehicle-controlled safety and efficacy trials in nearly 1,000 patients treated with polyphenon E 15% and 10% formulations.21
Two years later, investigators evaluated sinecatechins (Polyphenon E) 10% ointment in two double-blind, multinational studies in adults with external genital and perianal warts. Polyphenon E 10% was found to be significantly more effective than vehicle in completely or partially clearing all warts.32
Earlier that year, a review of the use of sinecatechins ointment for the treatment of external anogenital warts noted that while clearance rates are similar among sinecatechins and other indicated topical medications such as imiquimod and podophyllotoxin, recurrence rates are lower for patients treated with sinecatechins. The authors concluded that the use of sinecatechins for condylomata acuminata was safe and effective and its various molecular activities suggest broader applications to other viral and tumor lesions.33
In 2015, Gupta and Daigle reported that sinecatechins 10% ointment for the treatment of external genital warts was found in phase III trials to display greater efficacy and lower rates of recurrence in comparison to patient-applied treatments now available.28 Later that year, in a systematic PubMed and Embase review of clinical trials involving the use of polyphenol-based therapies, Tuong et al. identified cogent evidence suggesting the effectiveness of green tea polyphenols for the treatment of anogenital warts.34
Antiaging activity
Green tea has been shown to work in combination with red light to exert a rejuvenating effect on the skin, as Sommer and Zhu reported in 2009 that green tea filled cotton pads applied once daily for 20 minutes prior to treatment with light-emitting diodes (central wavelength 670 nm, dermal dose 4 J/cm2) reduced wrinkles in 1 month comparably to 10 months of light treatment alone.35
In 2013, Hong et al. studied the antiwrinkle effects of topically applied green tea extract with high antioxidant activity after tannase treatment. Study participants were randomly divided to receive either green tea extract or tannase-converted green tea extract on their crow’s feet for an 8-week period. The investigators found that tannase treatment elevated the antioxidant activity of green tea and imparted antiwrinkle effects.36
At around the same time, Gianeti conducted clinical studies in 24 volunteers to assess the effects of a cosmetic formulation containing 6% C. sinensis glycolic leaf extracts. Skin moisture was enhanced after 30 days of topical application as was the viscoelastic-to-elastic ratio compared with vehicle and control (a forearm area left untreated). Skin roughness was significantly diminished after 30 days. The investigators concluded that the topical cosmetic formulation with green tea yielded salient moisturizing and cutaneous microrelief benefits.37
Also in 2013, oral intake of green tea catechins in 16 healthy human subjects (with 14 completing the study) appeared to result in the integration of catechin metabolites into human skin linked to the negation of UV-induced 12-hydroxyeicosatetraenoic acid (12-HETE). The investigators speculated that this incorporation of catechins may render protection against sunburn inflammation and even cumulative UV-induced harm.38
After earlier showing the efficacy of green tea and lotus extracts in skin disorders involving excess sebum in a single-blinded, placebo-controlled, split-face comparative study,23 Mahmood and Akhtar conducted a 60-day placebo-controlled comparative split-face study in 33 healthy Asian men to evaluate the efficacy of two cosmetic formulations (green tea and lotus extract) for facial wrinkles. All of the formulations yielded improvements in skin roughness, scaliness, smoothness, and wrinkling, with the greatest reduction in wrinkling conferred by the combination formulation. The investigators concluded that the synergistic activity of green tea and lotus extracts exerted significant improvement along several skin parameters, suggesting the potential for these ingredients in antiaging products.38
In 2014, the synergistic effects of green tea and ginkgo biloba were explored in preclinical and clinical studies. In the clinical study, 48 participants applied the formulations on forearm skin and were evaluated before and after 3 hours and following 15- and 30-day use periods. Results showed a moisturizing effect and enhancement in skin microrelief, as well as improvements in skin elasticity and barrier function.3
Conclusion
Green tea remains one of the most researched antioxidants as benefits from its use continue to emerge. Indeed, green tea polyphenols are in use for a growing number of indications, especially acne and anogenital warts, and there is reason for optimism that topically applied green tea will gain momentum as an increasingly selected therapeutic option. More clinical studies are necessary to further establish the potential role of green tea for a wider range of cutaneous indications. Green tea holds particular promise in relation to photoprotection against UV-induced skin cancer and skin aging.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. She also developed and owns the Baumann Skin Type Solution skin typing systems and related products
References:
1. Cancer Lett. 1997 Mar 19;114(1-2):315-7.
2. J Am Acad Dermatol. 2005 Jun;52(6):1049-59.
3. Arch Dermatol. 2000 Aug;136(8):989-94.
4. Photochem Photobiol. 1995 Nov;62(5):855-61.
5. Oxid Med Cell Longev. 2012:2012:560682.
6. J Dtsch Dermatol Ges. 2015 Aug;13(8):768-75.
7. Am J Clin Dermatol. 2010;11(4):247-67.
8. Dermatol Ther. 2007 Sep-Oct;20(5):322-9.
9. J Clin Aesthet Dermatol. 2010 Feb;3(2):22-41.
10. Photodermatol Photoimmunol Photomed. 2007 Feb;23(1):48-56.
11. Skin Res Technol. 2009 Aug;15(3):338-45.
12. Exp Dermatol. 2009 Jan;18(1):69-77.
13. Exp Dermatol. 2009 Jun;18(6):522-6.
14. Arch Biochem Biophys. 2011 Apr 15;508(2):152-8.
15. Cancer Prev Res (Phila). 2010 Feb;3(2):179-89.
16. Complement Ther Clin Pract. 2014 Feb;20(1):11-5.
17. Skinmed. 2012 Nov-Dec;10(6):352-5.
18. J Eur Acad Dermatol Venereol. 2011 Mar;25(3):345-53.
19. J Drugs Dermatol. 2009 Apr;8(4):358-64.
20. Pak J Pharm Sci. 2012 Oct;25(4):867-70.
21. Br J Dermatol. 2010 Jan;162(1):176-84.
22. Arch Dermatol Res. 2012 Oct;304(8):655-63.
23. Hippokratia. 2013 Jan;17(1):64-7.
24. Food Chem Toxicol. 2008 Aug;46(8):2606-10.
25. Nat Biotechnol. 2008 Oct;26(10):1077-83.
26. Skin Therapy Lett. 2012 Apr;17(4):5-7.
27. J Clin Aesthet Dermatol. 2012 Jan;5(1):19-26.
28. Skin Therapy Lett. 2015 Jan-Feb;20(1):6-8.
29. Obstet Gynecol. 2008 Jun;111(6):1371-9.
30. Hautarzt. 2008 Jan;59(1):31-5.
31. J Eur Acad Dermatol Venereol. 2007 Nov;21(10):1404-12.
32. Am J Clin Dermatol. 2012 Aug 1:13(4):275-81.
33. Expert Opin Biol Ther. 2012 Jun;12(6):783-93.
34. J Dermatolog Treat. 2015;26(4):381-8.
35. Photomed Laser Surg. 2009 Dec;27(6):969-71.
36. J Cosmet Dermatol. 2013 Jun;12(2):137-43.
37. Dermatol Ther. 2013 May-Jun;26(3):267-71.
• Green tea is one of the most researched antioxidants, particularly its constituent polyphenolic catechins (notably epigallocatechin gallate, or EGCG).
• It is thought to be difficult to formulate in topical products because of the intrinsic hydrophilicity of EGCG.
• Topical application is thought to reduce inflammation and neutralize free radicals, but does not increase collagen production or reduce already existing wrinkles.
• It has been shown to be effective topically for treating acne, anogenital warts, and aging skin.
Resolution of Disseminated Granuloma Annulare With Removal of Surgical Hardware
To the Editor:
Disseminated granuloma annulare is a noninfectious granulomatous disease of unknown etiology. Reported precipitating factors include trauma, sun exposure, viral infection, vaccination, and malignancy.1 In contrast to a localized variant, disseminated granuloma annulare is associated with a later age of onset, longer duration, and recalcitrance to therapy.2 Although a variety of therapeutic approaches exist, there are limited efficacy data, which is complicated by the spontaneous, self-limited nature of the disease.3,4
A 47-year-old man presented with an eruption of a thick red plaque on the dorsal aspect of the left hand (Figure). The eruption began 6 weeks following fixation of a Galeazzi fracture of the right radius with a stainless steel volar plate. Subsequent to the initial eruption, similar indurated plaques developed on the left thenar area, bilateral axillae, and bilateral legs. A punch biopsy was conducted to rule out necrobiosis lipoidica diabeticorum and sarcoidosis as well as to histopathologically confirm the clinical diagnosis of disseminated granuloma annulare. Following diagnosis, the patient received topical clobetasol for application to the advancing borders of the plaques. At 4-month follow-up, additional plaques continued to develop. The patient was not interested in pursuing alternative courses of therapy and felt that the implantation of surgical hardware was the cause. To the best of our knowledge, there have been no reports of precipitation of disseminated granuloma annulare in response to surgical hardware. Given the time course of onset of the eruption it was plausible that the hardware was the inciting event. The orthopedist thought that the fracture had healed sufficiently to remove the volar plate. The patient elected to have the hardware removed to potentially resolve or arrest the progression of the plaques. Resolution of the plaques was observed by the patient 2 weeks following surgical removal of the volar plate. At 4 months following hardware removal, the patient only had 2 slightly pink, hyperpigmented lesions on the left hand in the areas most severely affected, with complete resolution of all other plaques. The patient was given topical clobetasol for the residual lesions.
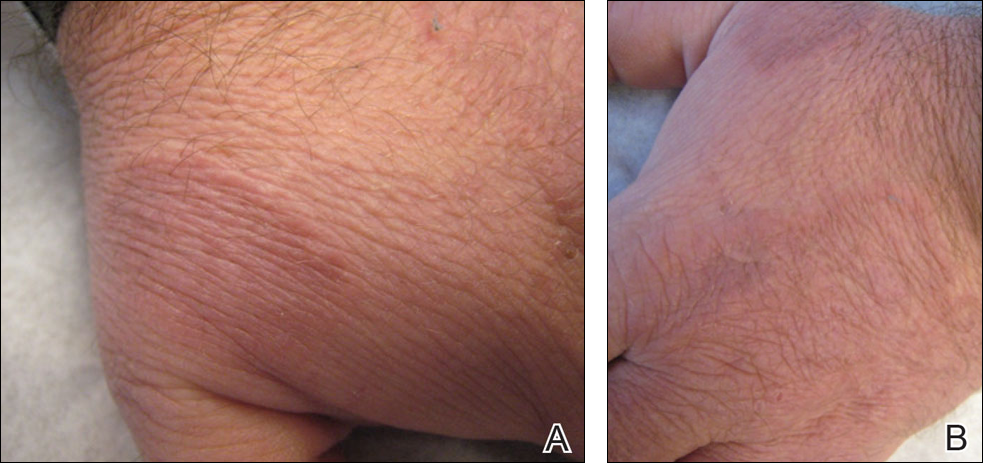
Precipitation and spontaneous resolution of disseminated granuloma annulare following the implantation and removal of surgical hardware is rare. Resolution following hardware removal is consistent with the theory that pathogenesis is due to a delayed-type hypersensitivity reaction to an inciting factor.5 Our case suggests that disseminated granuloma annulare may occur as a delayed-type hypersensitivity reaction to implanted surgical hardware, which should be considered in the etiology and potential therapeutic options for this disorder.
- Mills A, Chetty R. Auricular granuloma annulare. a consequence of trauma? Am J Dermatopathol. 1992;14:431-433.
- Dicken CH, Carrington SG, Winkelmann RK. Generalized granuloma annulare. Arch Dermatol. 1969;99:556-563.
- Yun JH, Lee JY, Kim MK, et al. Clinical and pathological features of generalized granuloma annulare with their correlation: a retrospective multicenter study in Korea [published online May 31, 2009]. Ann Dermatol. 2009:21:113-119
- Cyr PR. Diagnosis and management of granuloma annulare. Am Fam Physician. 2006;74:1729-1734.
- Buechner SA, Winkelmann RK, Banks PM. Identification of T-cell subpopulations in granuloma annulare. Arch Dermatol. 1983;119:125-128.
To the Editor:
Disseminated granuloma annulare is a noninfectious granulomatous disease of unknown etiology. Reported precipitating factors include trauma, sun exposure, viral infection, vaccination, and malignancy.1 In contrast to a localized variant, disseminated granuloma annulare is associated with a later age of onset, longer duration, and recalcitrance to therapy.2 Although a variety of therapeutic approaches exist, there are limited efficacy data, which is complicated by the spontaneous, self-limited nature of the disease.3,4
A 47-year-old man presented with an eruption of a thick red plaque on the dorsal aspect of the left hand (Figure). The eruption began 6 weeks following fixation of a Galeazzi fracture of the right radius with a stainless steel volar plate. Subsequent to the initial eruption, similar indurated plaques developed on the left thenar area, bilateral axillae, and bilateral legs. A punch biopsy was conducted to rule out necrobiosis lipoidica diabeticorum and sarcoidosis as well as to histopathologically confirm the clinical diagnosis of disseminated granuloma annulare. Following diagnosis, the patient received topical clobetasol for application to the advancing borders of the plaques. At 4-month follow-up, additional plaques continued to develop. The patient was not interested in pursuing alternative courses of therapy and felt that the implantation of surgical hardware was the cause. To the best of our knowledge, there have been no reports of precipitation of disseminated granuloma annulare in response to surgical hardware. Given the time course of onset of the eruption it was plausible that the hardware was the inciting event. The orthopedist thought that the fracture had healed sufficiently to remove the volar plate. The patient elected to have the hardware removed to potentially resolve or arrest the progression of the plaques. Resolution of the plaques was observed by the patient 2 weeks following surgical removal of the volar plate. At 4 months following hardware removal, the patient only had 2 slightly pink, hyperpigmented lesions on the left hand in the areas most severely affected, with complete resolution of all other plaques. The patient was given topical clobetasol for the residual lesions.

Precipitation and spontaneous resolution of disseminated granuloma annulare following the implantation and removal of surgical hardware is rare. Resolution following hardware removal is consistent with the theory that pathogenesis is due to a delayed-type hypersensitivity reaction to an inciting factor.5 Our case suggests that disseminated granuloma annulare may occur as a delayed-type hypersensitivity reaction to implanted surgical hardware, which should be considered in the etiology and potential therapeutic options for this disorder.
To the Editor:
Disseminated granuloma annulare is a noninfectious granulomatous disease of unknown etiology. Reported precipitating factors include trauma, sun exposure, viral infection, vaccination, and malignancy.1 In contrast to a localized variant, disseminated granuloma annulare is associated with a later age of onset, longer duration, and recalcitrance to therapy.2 Although a variety of therapeutic approaches exist, there are limited efficacy data, which is complicated by the spontaneous, self-limited nature of the disease.3,4
A 47-year-old man presented with an eruption of a thick red plaque on the dorsal aspect of the left hand (Figure). The eruption began 6 weeks following fixation of a Galeazzi fracture of the right radius with a stainless steel volar plate. Subsequent to the initial eruption, similar indurated plaques developed on the left thenar area, bilateral axillae, and bilateral legs. A punch biopsy was conducted to rule out necrobiosis lipoidica diabeticorum and sarcoidosis as well as to histopathologically confirm the clinical diagnosis of disseminated granuloma annulare. Following diagnosis, the patient received topical clobetasol for application to the advancing borders of the plaques. At 4-month follow-up, additional plaques continued to develop. The patient was not interested in pursuing alternative courses of therapy and felt that the implantation of surgical hardware was the cause. To the best of our knowledge, there have been no reports of precipitation of disseminated granuloma annulare in response to surgical hardware. Given the time course of onset of the eruption it was plausible that the hardware was the inciting event. The orthopedist thought that the fracture had healed sufficiently to remove the volar plate. The patient elected to have the hardware removed to potentially resolve or arrest the progression of the plaques. Resolution of the plaques was observed by the patient 2 weeks following surgical removal of the volar plate. At 4 months following hardware removal, the patient only had 2 slightly pink, hyperpigmented lesions on the left hand in the areas most severely affected, with complete resolution of all other plaques. The patient was given topical clobetasol for the residual lesions.

Precipitation and spontaneous resolution of disseminated granuloma annulare following the implantation and removal of surgical hardware is rare. Resolution following hardware removal is consistent with the theory that pathogenesis is due to a delayed-type hypersensitivity reaction to an inciting factor.5 Our case suggests that disseminated granuloma annulare may occur as a delayed-type hypersensitivity reaction to implanted surgical hardware, which should be considered in the etiology and potential therapeutic options for this disorder.
- Mills A, Chetty R. Auricular granuloma annulare. a consequence of trauma? Am J Dermatopathol. 1992;14:431-433.
- Dicken CH, Carrington SG, Winkelmann RK. Generalized granuloma annulare. Arch Dermatol. 1969;99:556-563.
- Yun JH, Lee JY, Kim MK, et al. Clinical and pathological features of generalized granuloma annulare with their correlation: a retrospective multicenter study in Korea [published online May 31, 2009]. Ann Dermatol. 2009:21:113-119
- Cyr PR. Diagnosis and management of granuloma annulare. Am Fam Physician. 2006;74:1729-1734.
- Buechner SA, Winkelmann RK, Banks PM. Identification of T-cell subpopulations in granuloma annulare. Arch Dermatol. 1983;119:125-128.
- Mills A, Chetty R. Auricular granuloma annulare. a consequence of trauma? Am J Dermatopathol. 1992;14:431-433.
- Dicken CH, Carrington SG, Winkelmann RK. Generalized granuloma annulare. Arch Dermatol. 1969;99:556-563.
- Yun JH, Lee JY, Kim MK, et al. Clinical and pathological features of generalized granuloma annulare with their correlation: a retrospective multicenter study in Korea [published online May 31, 2009]. Ann Dermatol. 2009:21:113-119
- Cyr PR. Diagnosis and management of granuloma annulare. Am Fam Physician. 2006;74:1729-1734.
- Buechner SA, Winkelmann RK, Banks PM. Identification of T-cell subpopulations in granuloma annulare. Arch Dermatol. 1983;119:125-128.
Practice Points
- Disseminated granuloma annulare may occur as a delayed-type hypersensitivity reaction to implanted surgical hardware.
- Resolution may occur following removal of surgical hardware.
Cosmetic Corner: Dermatologists Weigh in on OTC Acne Treatments
To improve patient care and outcomes, leading dermatologists offered their recommendations on OTC acne treatments. Consideration must be given to:
- Acne Care Gel
LovelySkin
“I particularly love this product in combination with azelaic acid for pregnant patients.”—Linda Fromm, MD, Rapid City, South Dakota
- Cleardin Acne Spot Gel
Isdin
“This product is great for spot treatments. It can be used alone or in combination with other acne products.”—Gary Goldenberg, MD, New York, New York
- Effaclar Duo
La Roche-Posay Laboratoire Dermatologique
“This is a great product with highly bioavailable benzoyl peroxide.”—Gary Goldenberg, MD, New York, New York
- Glytone Acne Treatment Spray
Pierre Fabre Dermo-Cosmetique USA
“For breakouts on the body, particularly for my male patients because it is quick and easy to use.”—Linda Fromm, MD, Rapid City, South Dakota
- Micronized BPO Cleanser 5%
BrandMDskincare
“For breakouts on the body, particularly for my male patients because it is quick and easy to use.”— Linda Fromm, MD, Rapid City, South Dakota
- Neutrogena Clear Pore Cleanser/Mask
Johnson & Johnson Consumer Inc
“This product has benzoyl peroxide, which is a known acne fighter. It can be used as a mask, which I recommend once a week.”—Anthony M. Rossi, MD, New York, New York
- Neutrogena On-the-Spot Acne Treatment
Johnson & Johnson Consumer Inc
“This product is best used for spot treating acne lesions. It is formulated as benzoyl peroxide 2.5%, which is the equivalent low yet effective concentration found in many prescription acne creams.”— Cherise M. Levi, DO, New York, New York
- PanOxyl 4% Acne Creamy Wash
Stiefel, a GSK company
Recommended by Gary Goldenberg, MD, New York, New York
Cutis invites readers to send us their recommendations. Self-tanners and cleansing devices will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on OTC acne treatments. Consideration must be given to:
- Acne Care Gel
LovelySkin
“I particularly love this product in combination with azelaic acid for pregnant patients.”—Linda Fromm, MD, Rapid City, South Dakota
- Cleardin Acne Spot Gel
Isdin
“This product is great for spot treatments. It can be used alone or in combination with other acne products.”—Gary Goldenberg, MD, New York, New York
- Effaclar Duo
La Roche-Posay Laboratoire Dermatologique
“This is a great product with highly bioavailable benzoyl peroxide.”—Gary Goldenberg, MD, New York, New York
- Glytone Acne Treatment Spray
Pierre Fabre Dermo-Cosmetique USA
“For breakouts on the body, particularly for my male patients because it is quick and easy to use.”—Linda Fromm, MD, Rapid City, South Dakota
- Micronized BPO Cleanser 5%
BrandMDskincare
“For breakouts on the body, particularly for my male patients because it is quick and easy to use.”— Linda Fromm, MD, Rapid City, South Dakota
- Neutrogena Clear Pore Cleanser/Mask
Johnson & Johnson Consumer Inc
“This product has benzoyl peroxide, which is a known acne fighter. It can be used as a mask, which I recommend once a week.”—Anthony M. Rossi, MD, New York, New York
- Neutrogena On-the-Spot Acne Treatment
Johnson & Johnson Consumer Inc
“This product is best used for spot treating acne lesions. It is formulated as benzoyl peroxide 2.5%, which is the equivalent low yet effective concentration found in many prescription acne creams.”— Cherise M. Levi, DO, New York, New York
- PanOxyl 4% Acne Creamy Wash
Stiefel, a GSK company
Recommended by Gary Goldenberg, MD, New York, New York
Cutis invites readers to send us their recommendations. Self-tanners and cleansing devices will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on OTC acne treatments. Consideration must be given to:
- Acne Care Gel
LovelySkin
“I particularly love this product in combination with azelaic acid for pregnant patients.”—Linda Fromm, MD, Rapid City, South Dakota
- Cleardin Acne Spot Gel
Isdin
“This product is great for spot treatments. It can be used alone or in combination with other acne products.”—Gary Goldenberg, MD, New York, New York
- Effaclar Duo
La Roche-Posay Laboratoire Dermatologique
“This is a great product with highly bioavailable benzoyl peroxide.”—Gary Goldenberg, MD, New York, New York
- Glytone Acne Treatment Spray
Pierre Fabre Dermo-Cosmetique USA
“For breakouts on the body, particularly for my male patients because it is quick and easy to use.”—Linda Fromm, MD, Rapid City, South Dakota
- Micronized BPO Cleanser 5%
BrandMDskincare
“For breakouts on the body, particularly for my male patients because it is quick and easy to use.”— Linda Fromm, MD, Rapid City, South Dakota
- Neutrogena Clear Pore Cleanser/Mask
Johnson & Johnson Consumer Inc
“This product has benzoyl peroxide, which is a known acne fighter. It can be used as a mask, which I recommend once a week.”—Anthony M. Rossi, MD, New York, New York
- Neutrogena On-the-Spot Acne Treatment
Johnson & Johnson Consumer Inc
“This product is best used for spot treating acne lesions. It is formulated as benzoyl peroxide 2.5%, which is the equivalent low yet effective concentration found in many prescription acne creams.”— Cherise M. Levi, DO, New York, New York
- PanOxyl 4% Acne Creamy Wash
Stiefel, a GSK company
Recommended by Gary Goldenberg, MD, New York, New York
Cutis invites readers to send us their recommendations. Self-tanners and cleansing devices will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
Keep hyaluronidase in the office for filler complications
NEWPORT BEACH, CALIF. – If you inject fillers, Manhattan plastic surgeon Lawrence Bass, MD, recommends that you keep hyaluronidase handy.
It’s the go-to for intravascular injections and helpful for nodules and granulomas, the main filler worries, but some offices don’t stock it. “Complications are rare, but they can be devastating. You have to be prepared.”
Massage, aspiration, transdermal nitroglycerin, and other options might help when, for example, a patient’s lip turns purple after a nasolabial fold injection, but “really, it’s all about hyaluronidase,” whether the filler is hyaluronic acid or not. The enzyme digests hyaluronic acid – both the injected kind and what patients have naturally – and loosens and disperses other fillers.
A patient who says that the injection hurt more than usual is an indication that the vessel may have been injected, Dr. Bass said. In such cases, “you have to deal with it immediately,” and repeat dosing if skin color doesn’t improve within an hour, he noted. “If you manage patients correctly, you can usually settle it with little or no deformity.”
There are two hyaluronidase options in the United States: Vitrase, an ovine formulation, and Hylenex, a recombinant human formulation. It probably doesn’t matter which one is used, he said. Soft tissue vascular compromise generally requires 200 units, and retinal artery occlusion, 500 units or more. Restylane is the easiest filler to digest because it’s the least cross-linked; Juvederm is more difficult; and Belotero is the most difficult, Dr. Bass noted.
Nodules and granulomas from filler injections have a slower onset than vascular compromise. Infected nodules can appear months after injection, and granulomas – red or purplish hypersensitivity reactions with indistinct borders, often with all injection sites involved at once – can appear years afterwards, set off by the flu or some other inflammatory trigger, according to Miles Graivier, MD, a plastic surgeon in Roswell, Ga., who shared the podium with Dr. Bass at the meeting.
Dr. Graivier described his approach to granulomas as “very aggressive.” Patients with granulomas receive oral steroids plus intralesional triamcinolone/fluorouracil/lidocaine injections every 2 weeks or more frequently. Hyaluronidase is used to disperse the filler. Resistant patients also get oral allopurinol. Laser liquefaction can help with both granulomas and infected nodules, he added.
Retinal artery occlusion is probably the most feared filler complication. It’s most likely with glabellar or periorbital injections but possible with any facial injection. “It’s a real horror show when it happens. The bottom line is you must restore circulation within 60-90 minutes” with retrobulbar hyaluronidase injections “or patients will lose vision permanently,” Dr. Bass said. People might not feel comfortable with retrobulbar injections, “but when the alternative is blindness, maybe you want to give them a try,” he added.
A 25 gauge, 1.5 inch needle is introduced to the lateral limbus at the orbital rim. “You pass the needle along the orbital floor to the equator of the globe,” – about 13-14 mm – “angle it up to enter the muscular conus, and inject 500 units or more.” Injecting at the orbital floor is less risky, but probably less effective, he said.
Hard nodules, meanwhile, present early after injection and are usually a result of too much filler or poor technique, Dr. Graivier said. For hard nodules, “hyaluronidase is the number one treatment,” although cracking, massage, and saline injection might help.
Infected nodules look like abscesses, but usually grow nothing on culture. Hyaluronidase is also first line for infected nodules, along with drainage and broad spectrum oral antibiotics for at least 6 weeks, and, if they don’t work, intravenous antibiotics, he added.
Dr. Bass is an investigator and speaker for Cynosure; an investigator for Endo Pharmaceuticals and Neothetics, and an advisor to Merz. Dr. Graivier is an advisor for Merz, and an investigator for Merz, Galderma, Evolus, Ideal Implants, Exilis, and Cynosure. The Graivier Center for Plastic Surgery is a training center for Cynosure.
The Summit in Aesthetic Medicine is held by Global Academy for Medical Education. Global Academy and this news organization are owned by the same company.
NEWPORT BEACH, CALIF. – If you inject fillers, Manhattan plastic surgeon Lawrence Bass, MD, recommends that you keep hyaluronidase handy.
It’s the go-to for intravascular injections and helpful for nodules and granulomas, the main filler worries, but some offices don’t stock it. “Complications are rare, but they can be devastating. You have to be prepared.”
Massage, aspiration, transdermal nitroglycerin, and other options might help when, for example, a patient’s lip turns purple after a nasolabial fold injection, but “really, it’s all about hyaluronidase,” whether the filler is hyaluronic acid or not. The enzyme digests hyaluronic acid – both the injected kind and what patients have naturally – and loosens and disperses other fillers.
A patient who says that the injection hurt more than usual is an indication that the vessel may have been injected, Dr. Bass said. In such cases, “you have to deal with it immediately,” and repeat dosing if skin color doesn’t improve within an hour, he noted. “If you manage patients correctly, you can usually settle it with little or no deformity.”
There are two hyaluronidase options in the United States: Vitrase, an ovine formulation, and Hylenex, a recombinant human formulation. It probably doesn’t matter which one is used, he said. Soft tissue vascular compromise generally requires 200 units, and retinal artery occlusion, 500 units or more. Restylane is the easiest filler to digest because it’s the least cross-linked; Juvederm is more difficult; and Belotero is the most difficult, Dr. Bass noted.
Nodules and granulomas from filler injections have a slower onset than vascular compromise. Infected nodules can appear months after injection, and granulomas – red or purplish hypersensitivity reactions with indistinct borders, often with all injection sites involved at once – can appear years afterwards, set off by the flu or some other inflammatory trigger, according to Miles Graivier, MD, a plastic surgeon in Roswell, Ga., who shared the podium with Dr. Bass at the meeting.
Dr. Graivier described his approach to granulomas as “very aggressive.” Patients with granulomas receive oral steroids plus intralesional triamcinolone/fluorouracil/lidocaine injections every 2 weeks or more frequently. Hyaluronidase is used to disperse the filler. Resistant patients also get oral allopurinol. Laser liquefaction can help with both granulomas and infected nodules, he added.
Retinal artery occlusion is probably the most feared filler complication. It’s most likely with glabellar or periorbital injections but possible with any facial injection. “It’s a real horror show when it happens. The bottom line is you must restore circulation within 60-90 minutes” with retrobulbar hyaluronidase injections “or patients will lose vision permanently,” Dr. Bass said. People might not feel comfortable with retrobulbar injections, “but when the alternative is blindness, maybe you want to give them a try,” he added.
A 25 gauge, 1.5 inch needle is introduced to the lateral limbus at the orbital rim. “You pass the needle along the orbital floor to the equator of the globe,” – about 13-14 mm – “angle it up to enter the muscular conus, and inject 500 units or more.” Injecting at the orbital floor is less risky, but probably less effective, he said.
Hard nodules, meanwhile, present early after injection and are usually a result of too much filler or poor technique, Dr. Graivier said. For hard nodules, “hyaluronidase is the number one treatment,” although cracking, massage, and saline injection might help.
Infected nodules look like abscesses, but usually grow nothing on culture. Hyaluronidase is also first line for infected nodules, along with drainage and broad spectrum oral antibiotics for at least 6 weeks, and, if they don’t work, intravenous antibiotics, he added.
Dr. Bass is an investigator and speaker for Cynosure; an investigator for Endo Pharmaceuticals and Neothetics, and an advisor to Merz. Dr. Graivier is an advisor for Merz, and an investigator for Merz, Galderma, Evolus, Ideal Implants, Exilis, and Cynosure. The Graivier Center for Plastic Surgery is a training center for Cynosure.
The Summit in Aesthetic Medicine is held by Global Academy for Medical Education. Global Academy and this news organization are owned by the same company.
NEWPORT BEACH, CALIF. – If you inject fillers, Manhattan plastic surgeon Lawrence Bass, MD, recommends that you keep hyaluronidase handy.
It’s the go-to for intravascular injections and helpful for nodules and granulomas, the main filler worries, but some offices don’t stock it. “Complications are rare, but they can be devastating. You have to be prepared.”
Massage, aspiration, transdermal nitroglycerin, and other options might help when, for example, a patient’s lip turns purple after a nasolabial fold injection, but “really, it’s all about hyaluronidase,” whether the filler is hyaluronic acid or not. The enzyme digests hyaluronic acid – both the injected kind and what patients have naturally – and loosens and disperses other fillers.
A patient who says that the injection hurt more than usual is an indication that the vessel may have been injected, Dr. Bass said. In such cases, “you have to deal with it immediately,” and repeat dosing if skin color doesn’t improve within an hour, he noted. “If you manage patients correctly, you can usually settle it with little or no deformity.”
There are two hyaluronidase options in the United States: Vitrase, an ovine formulation, and Hylenex, a recombinant human formulation. It probably doesn’t matter which one is used, he said. Soft tissue vascular compromise generally requires 200 units, and retinal artery occlusion, 500 units or more. Restylane is the easiest filler to digest because it’s the least cross-linked; Juvederm is more difficult; and Belotero is the most difficult, Dr. Bass noted.
Nodules and granulomas from filler injections have a slower onset than vascular compromise. Infected nodules can appear months after injection, and granulomas – red or purplish hypersensitivity reactions with indistinct borders, often with all injection sites involved at once – can appear years afterwards, set off by the flu or some other inflammatory trigger, according to Miles Graivier, MD, a plastic surgeon in Roswell, Ga., who shared the podium with Dr. Bass at the meeting.
Dr. Graivier described his approach to granulomas as “very aggressive.” Patients with granulomas receive oral steroids plus intralesional triamcinolone/fluorouracil/lidocaine injections every 2 weeks or more frequently. Hyaluronidase is used to disperse the filler. Resistant patients also get oral allopurinol. Laser liquefaction can help with both granulomas and infected nodules, he added.
Retinal artery occlusion is probably the most feared filler complication. It’s most likely with glabellar or periorbital injections but possible with any facial injection. “It’s a real horror show when it happens. The bottom line is you must restore circulation within 60-90 minutes” with retrobulbar hyaluronidase injections “or patients will lose vision permanently,” Dr. Bass said. People might not feel comfortable with retrobulbar injections, “but when the alternative is blindness, maybe you want to give them a try,” he added.
A 25 gauge, 1.5 inch needle is introduced to the lateral limbus at the orbital rim. “You pass the needle along the orbital floor to the equator of the globe,” – about 13-14 mm – “angle it up to enter the muscular conus, and inject 500 units or more.” Injecting at the orbital floor is less risky, but probably less effective, he said.
Hard nodules, meanwhile, present early after injection and are usually a result of too much filler or poor technique, Dr. Graivier said. For hard nodules, “hyaluronidase is the number one treatment,” although cracking, massage, and saline injection might help.
Infected nodules look like abscesses, but usually grow nothing on culture. Hyaluronidase is also first line for infected nodules, along with drainage and broad spectrum oral antibiotics for at least 6 weeks, and, if they don’t work, intravenous antibiotics, he added.
Dr. Bass is an investigator and speaker for Cynosure; an investigator for Endo Pharmaceuticals and Neothetics, and an advisor to Merz. Dr. Graivier is an advisor for Merz, and an investigator for Merz, Galderma, Evolus, Ideal Implants, Exilis, and Cynosure. The Graivier Center for Plastic Surgery is a training center for Cynosure.
The Summit in Aesthetic Medicine is held by Global Academy for Medical Education. Global Academy and this news organization are owned by the same company.
EXPERT ANALYSIS FROM THE SUMMIT IN AESTHETIC MEDICINE
Comparison of Pneumatic Broadband Light Plus Adapalene Gel 0.3% Versus Adapalene Gel 0.3% Monotherapy in the Treatment of Mild to Moderate Acne
Acne is a common and distressing condition that typically presents in adolescents and young adults and has been associated with not only medical but also emotional and aesthetic consequences. Acne treatments that offer faster improvement are the coveted goal. Although clinical studies support the use of combination therapy with topical retinoids and antibiotics, the overuse of antibiotics raises caution for bacterial resistance.1 Therefore, adjunctive treatments such as chemical peels, light therapy, and laser treatments can hasten the response to traditional acne treatments and in some cases may potentially decrease use of both oral and topical antibiotics.
Light therapy, particularly with visible light, may improve acne outcomes. Pneumatic broadband light (PBBL) is a light treatment in the broadband range (400–1200 nm) combined with a vacuum. The suction created by the vacuum has several effects on acne lesions, such as creating a mechanical lysis of thin-walled pustules and dislodging pore impaction. The blue light with a wavelength of 410 nm targets endogenous porphyrins in Propionibacterium acnes and elicits singlet oxygen production, resulting in bacterial destruction.2,3 Studies showed that PBBL alone was effective in most patients with mild to moderate acne and caused minimal side effects.2-4
We sought to determine if PBBL combined with a topical retinoid can accelerate and prolong acne improvement. We evaluated the efficacy, safety, and tolerability of PBBL plus adapalene gel 0.3% versus adapalene gel 0.3% monotherapy in patients with mild to moderate acne.
METHODSPatient Population
Patients with mild to moderate acne were eligible for the study if they were 18 years or older at screening, in good health, had stopped oral isotretinoin for at least 1 year prior to treatment initiation, and were not taking oral or topical antibiotics or using any topical retinoid derivatives for at least 1 month prior to treatment initiation. Inclusion criteria included at least 10 acne lesions on the face. Patients were excluded if they had a history of receiving PBBL treatment; had a history of scarring, hypopigmentation, or hyperpigmentation from laser or light treatments; and/or were pregnant or refused use of contraception during the study period.
Study Design
This single-blind, randomized, split-face study was approved by the institutional review board of the University of Pennsylvania (Philadelphia, Pennsylvania). All participants provided informed consent before entering the study. Each participant was randomly assigned to receive PBBL on one side of the face for 6 consecutive weeks and apply adapalene gel 0.3% to both sides of the face nightly for 10 weeks. Pneumatic broadband light treatment was performed using the following settings: starting power 2 (approximately 4–6 J/cm2) and vacuum setting 3 (negative pressure, approximately 3 lb/in2). The power setting was increased to a maximum of 6 (12–14 J/cm2) at subsequent visits depending on tolerability of the participants.
All participants visited the clinic weekly for 6 weeks and also returned for follow-up at week 10 (4 weeks following last PBBL treatment). At each visit, the participants completed satisfaction questionnaires and were assessed by a dermatologist evaluator using several parameters including the modified Global Acne Grading Score (mGAGS), clinical photography, participant self-assessment, physician assessment, and Wong Baker FACES Pain Rating Scale (WBPRS). The physician evaluator was blinded to the side of the face receiving PBBL treatment. Clinical photographs were taken to compare the clinical outcome at each visit versus baseline.
Efficacy Evaluation
Acne Counts
The blinded evaluator counted acne lesions and assessed the mGAGS at each visit prior to administration of the PBBL treatment. Acne lesions were counted separately as noninflammatory (comedones) and inflammatory (papules, pustules, nodules) on the forehead, cheeks, nose, and chin.
Modified Global Acne Grading Score
The modified Global Acne Grading Score was modified from the Global Acne Grading Scale (GAGS) that has previously been used to evaluate acne severity.5 The original GAGS used the type and location of the acne lesions. The GAGS considers 6 locations on the face, chest, and upper back, with a grading factor for each location (forehead=2; cheeks=2; nose=1; chin=1). Another grading factor represented the lesion type (0=no lesion; 1=comedone; 2=papule; 3=pustule; 4=nodule). The local score was calculated by multiplying the location grading factor by the lesion type grading factor. The total score was the sum of the individual local scores for the 4 locations.
Given that the number of acne lesions is important, we modified the GAGS by adding a grading factor that represented the number of lesions to improve the accuracy of the test (1=0–10 lesions; 2=11–20 lesions; 3=21–30 lesions; 4=≥31 lesions). The local score of mGAGS was calculated by multiplying the grading factors for location, lesion type, and number of lesions. Each local score was then added to yield a total score. The mGAGS may be useful and more accurate to determine the severity of acne (0=none; 1–44=mild; 45–80=moderate; 81–132=severe; 133–176=very severe).
Participant Self-assessment
Participants assessed their acne lesions using an 11-point rating scale (–5=100% worsening; –4=76%–99% worsening; –3=51%–75% worsening; –2=26%–50% worsening; –1=1%–25% worsening; 0=no improvement; 1=1%–25% improvement; 2=26%–50% improvement; 3=51%–75% improvement; 4=76%–99% improvement; 5=100% acne clear) to compare their acne at each treatment visit and week 10 follow-up with a baseline photograph.
Physician Assessment
The blinded evaluator assessed acne lesions on the face using the same 11-point rating scale that was used for participant self-assessment. For each participant, assessments were made at each treatment visit and week 10 follow-up by comparing baseline photographs.
Safety Evaluation
The WBPRS score, a standardized 6-point scale (0=no pain; 1=hurts a little bit; 2=hurts a little bit more; 3=hurts even more; 4=hurts whole lot; 5=hurt worst),6 was used to evaluate pain toleration during PBBL treatments and was recorded along with adverse events throughout the study.
Statistical Analysis
Based on data from 2 prior studies,3,7 we expected that the favorable clinical outcome of adapalene gel 0.3% and PBBL therapy would be 23% and 78%, respectively. If the adjunctive therapy with PBBL was beneficial, the favorable outcome would be higher than 78%. To be able to detect this difference, the sample size of 11 patients was needed when 5% type I error and 20% type II error were accepted.
Categorical variables were expressed as percentages, while continuous variables were expressed in terms of median (range). The clinical outcomes between both treatment groups were compared using the Wilcoxon signed rank test. A 2-tailed P value of ≤.05 was considered statistically significant. All statistical calculations were performed using STATA software version 10.0.
RESULTS
Baseline Characteristics
Four male and 7 female patients aged 18 to 35 years (median, 23 years) with mild to moderate acne were enrolled in the study. Of the 11 participants, 7 were white, 2 were black, 1 was Asian, and 1 was Latin American. Baseline characteristics of both sides of the face were comparable in all participants (Table 1). Eight participants (73%) completed the study. Two black participants withdrew from the study due to hyperpigmentation following PBBL treatment; 1 participant did not return for follow-up at week 10, as she was out of the country.
Lesion Counts
At week 3, reduction in noninflammatory lesions was significantly greater on the side receiving the combination therapy compared to the monotherapy side (P=.04)(Table 2). However, there was no significant difference between the combination therapy and the adapalene monotherapy sides in the reduction of noninflammatory and inflammatory lesions at week 4 (Figure 1). There was a remarkable improvement of the combination therapy and adapalene monotherapy sides in acne lesions, but there was no significant difference between the combination therapy and the adapalene monotherapy sides (Figure 2).
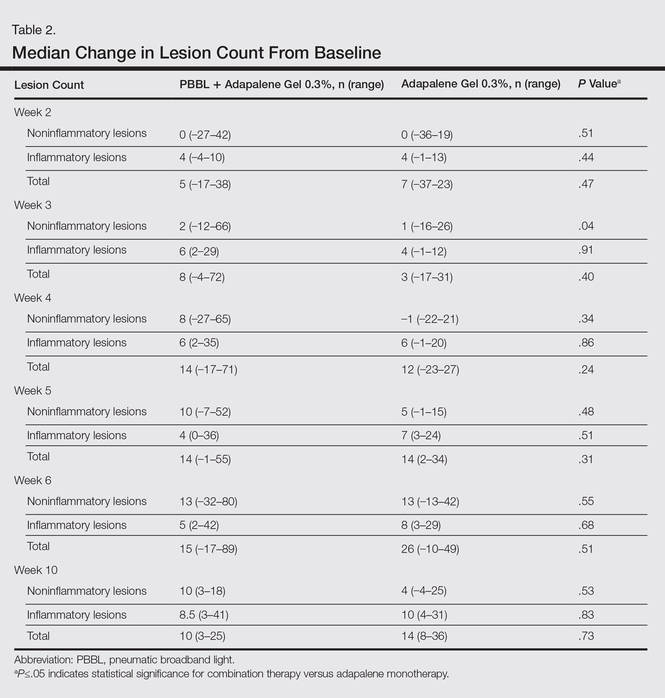
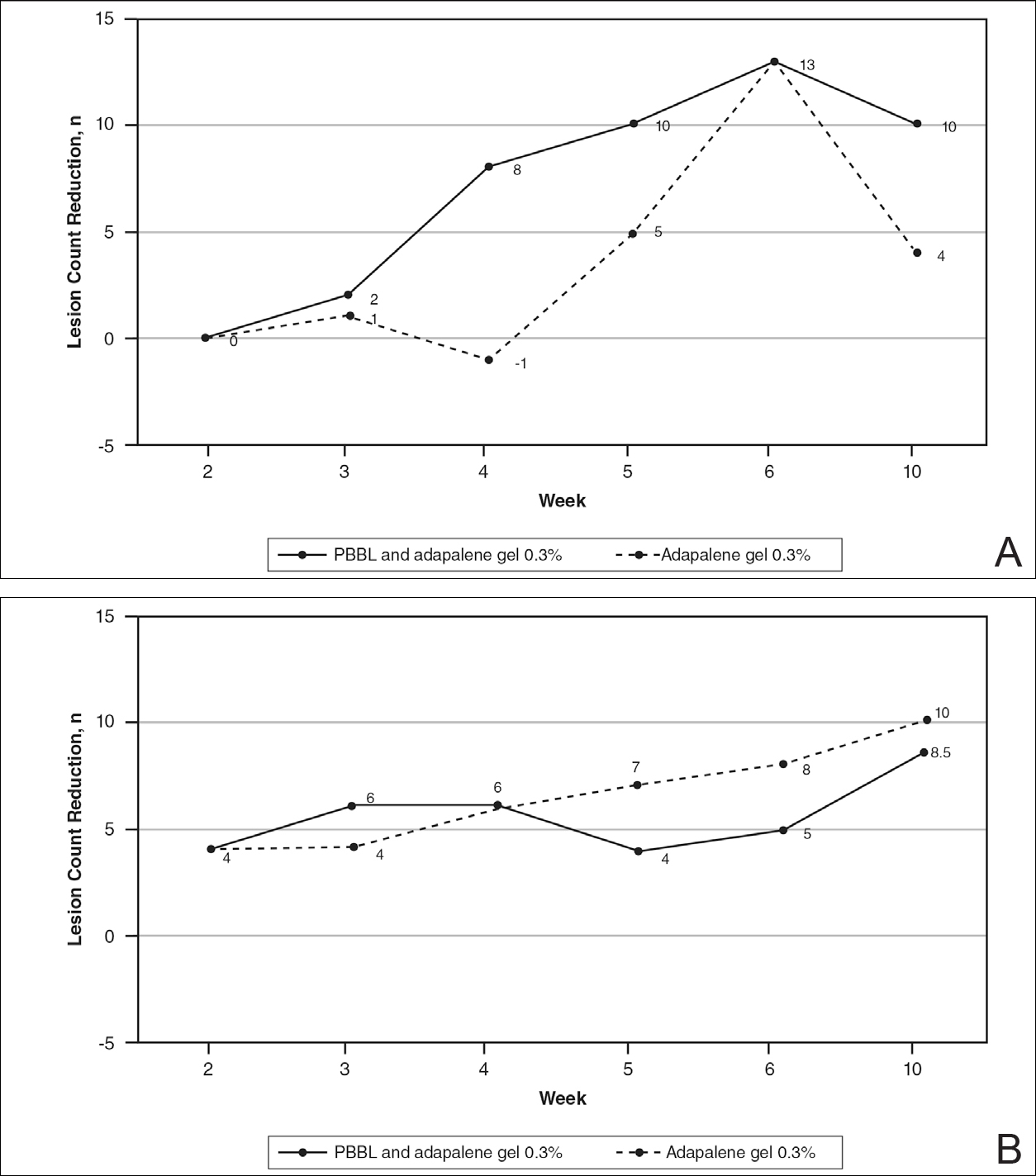
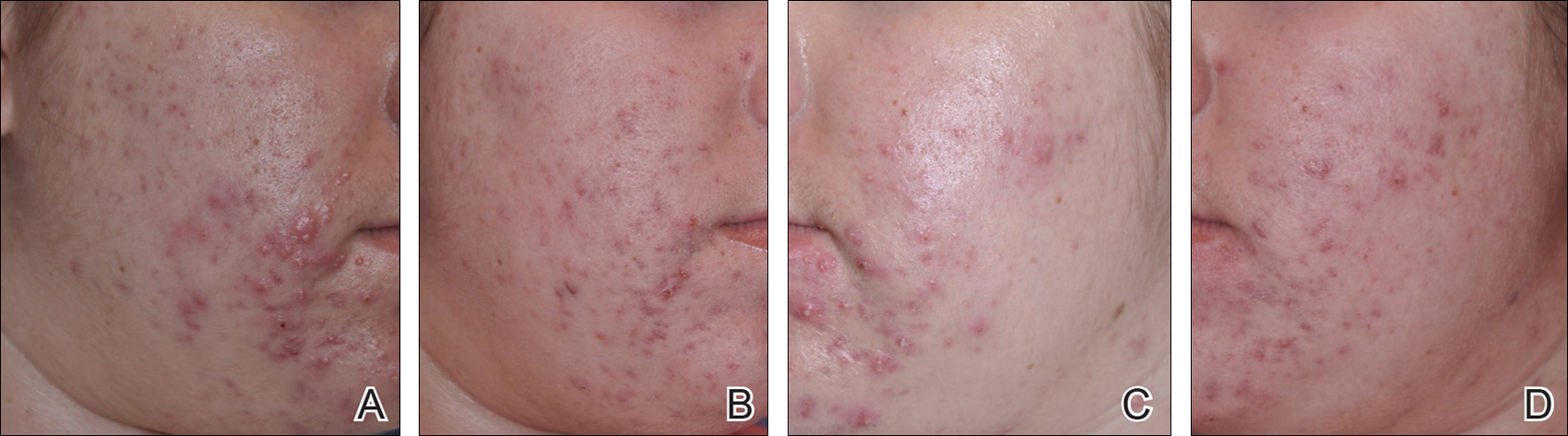
Modified Global Acne Grading Score
At weeks 3 and 4, the improvement of mGAGS was significantly greater on the side treated with the combination therapy (P=.05). However, this significant difference was not sustained (Table 3).
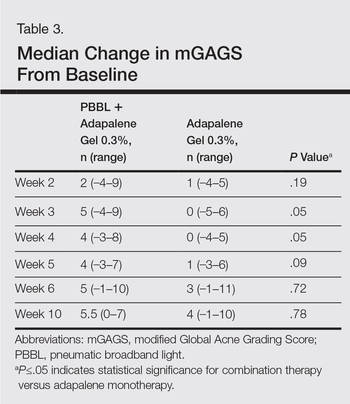
Participant Self-assessment and Physician Assessment
The rate of acne improvement according to participant self-assessment was slightly higher on the side receiving the combination therapy compared to the monotherapy side at week 2 (26%–50% vs 1%–25%) and week 6 (76%–99% vs 51%–75%). However, there was no statistically significant difference. For the physician assessment, there was no significant difference between the monotherapy and combination therapy sides.
Safety
The median WBPRS score was 1 (hurts a little bit) throughout all PBBL treatment visits. The maximum score was highest at week 1 (4=hurts whole lot) and subsequently decreased to 2 (hurts a little bit more) at week 6.
After the PBBL treatment, all participants experienced transient erythema in the treatment area. All participants noted their skin had become drier than usual from adapalene, except 1 participant (11%) who reported very dry skin on areas where adapalene gel 0.3% had been applied. However, the dryness was tolerable and relief was reported following application of a moisturizer. No participants withdrew from the study due to skin dryness.
Both black participants experienced hyperpigmentation caused by PBBL (1 on the treatment sites, the other on the test spot) and withdrew from the study. The hyperpigmentation resolved over time following application of a topical bleaching cream. One patient experienced purpura following PBBL treatment at week 4, which was associated with an increase in PBBL power. No other side effects (eg, scaling, stinging, burning, vesicle formation, blistering, crusting, scarring) were observed.
COMMENT
This 10-week study demonstrated that PBBL initially improved the appearance of acne in the first month of treatment, as determined by the significantly greater reduction in mGAGS for the combination side versus the adapalene monotherapy side. Differences in the reduction of acne lesions were not significant between the 2 treatments, except for noninflammatory lesion reduction at week 3. Analysis of physician assessment with photographs revealed acne improvement from baseline in the first month but no additional effects with the PBBL treatment at the end of study. Similarly, participant assessment indicated an improvement by week 2 with the combination therapy compared to adapalene monotherapy in their assessment of acne lesion reductions from baseline. By the end of the study, there was no significant difference between monotherapy and combination therapy.
These findings illustrate that combination therapy with PBBL plus adapalene improved the appearance of acne lesions within the first month of treatment, but there were no further signs of improvement at weeks 5 and 6. These results are consistent with at least 2 other studies that demonstrated acne reduction within the first 3 weeks of PBBL treatment.2,4 The current study was completed as planned with 6 weeks of combination therapy and patients continued adapalene application until the last follow-up visit in week 10. The length of the combination treatment was enough to determine that extension of treatment would not be necessary to gain any further benefits in this study. Because of the small sample size, we would not be able to detect any significant differences, as the difference between the combination therapy and the adapalene monotherapy was less than 55%. Therefore, a future study with a larger sample size is needed to draw a better conclusion.
Pneumatic broadband light has shown impressive results in acne treatment. However, some side effects need to be considered. Minimal adverse events have been reported such as erythema, dryness, peeling, burning, and itching.2-4 In this study, we found that all patients experienced transient erythema during and after PBBL treatment, but this effect disappeared in minutes. Purpura can occur if a higher power of PBBL is performed (6 or greater). Black patients experienced hyperpigmentation that can occur in darker skin types, as reported when light therapy is performed despite using the correct skin type tips.8 Therefore, care must be used in darker skin types, and we advocate a skin test in this population prior to general use.
Our study showed that PBBL can be safely combined with adapalene gel 0.3% and is well tolerated in the treatment of mild to moderate facial acne vulgaris for patients with Fitzpatrick skin types I to III. The combination of PBBL and adapalene reduces acne severity, as shown by the reduction in mGAGS during the first month of treatment. Patients noted faster improvement in their acne lesions with this combination. Although this study was limited by a relatively small sample size, this information may be useful in getting patients to be compliant overall, as they appeared to see results sooner, giving other therapies time to initiate their effect. It appears that 4 consecutive weekly treatments are enough to see that effect. Additionally, this combination therapy provides results without having to resort to oral antibiotics, as many patients today are concerned about creating future antibiotic resistance.
Conclusion
Adapalene gel 0.3% can be safely combined with PBBL for treatment of mild to moderate acne. Although the benefits of this combination therapy can be seen after 4 consecutive weekly treatments, the beneficial effect is not sustained.
Acknowledgment
The authors would like to thank Joyce Okawa, RN (Philadelphia, Pennsylvania), for her assistance in the submission to the University of Pennsylvania institutional review board.
- Thiboutot D, Gollnick H, Bettoli V, et al. New insights into the management of acne: an update from the Global Alliance to Improve Outcomes in Acne group. J Am Acad Dermatol. 2009;60(5 suppl):S1-S50.
- Gold MH, Biron J. Efficacy of a novel combination of pneumatic energy and broadband light for the treatment of acne. J Drugs Dermatol. 2008;7:639-642.
- Shamban AT, Enokibori M, Narurkar V, et al. Photopneumatic technology for the treatment of acne vulgaris. J Drugs Dermatol. 2008;7:139-145.
- Wanitphakdeedecha R, Tanzi EL, Alster TS. Photopneumatic therapy for the treatment of acne. J Drugs Dermatol. 2009;8:239-241.
- Doshi A, Zaheer A, Stiller MJ. A comparison of current acne grading systems and proposal of a novel system. Int J Dermatol. 1997;36:416-418.
- Wong DL, Baker CM. Pain in children: comparison of assessment scales. Pediatr Nurs. 1988;14:9-17.
- Thiboutot D, Pariser DM, Egan N, et al. Adapalene gel 0.3% for the treatment of acne vulgaris: a multicenter, randomized, double-blind, controlled, phase III trial. J Am Acad Dermatol. 2006;54:242-250.
- Yeung CK, Shek SY, Bjerring P, et al. A comparative study of intense pulsed light alone and its combination with photodynamic therapy for the treatment of facial acne in Asian skin. Lasers Surg Med. 2007;39:1-6.
Acne is a common and distressing condition that typically presents in adolescents and young adults and has been associated with not only medical but also emotional and aesthetic consequences. Acne treatments that offer faster improvement are the coveted goal. Although clinical studies support the use of combination therapy with topical retinoids and antibiotics, the overuse of antibiotics raises caution for bacterial resistance.1 Therefore, adjunctive treatments such as chemical peels, light therapy, and laser treatments can hasten the response to traditional acne treatments and in some cases may potentially decrease use of both oral and topical antibiotics.
Light therapy, particularly with visible light, may improve acne outcomes. Pneumatic broadband light (PBBL) is a light treatment in the broadband range (400–1200 nm) combined with a vacuum. The suction created by the vacuum has several effects on acne lesions, such as creating a mechanical lysis of thin-walled pustules and dislodging pore impaction. The blue light with a wavelength of 410 nm targets endogenous porphyrins in Propionibacterium acnes and elicits singlet oxygen production, resulting in bacterial destruction.2,3 Studies showed that PBBL alone was effective in most patients with mild to moderate acne and caused minimal side effects.2-4
We sought to determine if PBBL combined with a topical retinoid can accelerate and prolong acne improvement. We evaluated the efficacy, safety, and tolerability of PBBL plus adapalene gel 0.3% versus adapalene gel 0.3% monotherapy in patients with mild to moderate acne.
METHODSPatient Population
Patients with mild to moderate acne were eligible for the study if they were 18 years or older at screening, in good health, had stopped oral isotretinoin for at least 1 year prior to treatment initiation, and were not taking oral or topical antibiotics or using any topical retinoid derivatives for at least 1 month prior to treatment initiation. Inclusion criteria included at least 10 acne lesions on the face. Patients were excluded if they had a history of receiving PBBL treatment; had a history of scarring, hypopigmentation, or hyperpigmentation from laser or light treatments; and/or were pregnant or refused use of contraception during the study period.
Study Design
This single-blind, randomized, split-face study was approved by the institutional review board of the University of Pennsylvania (Philadelphia, Pennsylvania). All participants provided informed consent before entering the study. Each participant was randomly assigned to receive PBBL on one side of the face for 6 consecutive weeks and apply adapalene gel 0.3% to both sides of the face nightly for 10 weeks. Pneumatic broadband light treatment was performed using the following settings: starting power 2 (approximately 4–6 J/cm2) and vacuum setting 3 (negative pressure, approximately 3 lb/in2). The power setting was increased to a maximum of 6 (12–14 J/cm2) at subsequent visits depending on tolerability of the participants.
All participants visited the clinic weekly for 6 weeks and also returned for follow-up at week 10 (4 weeks following last PBBL treatment). At each visit, the participants completed satisfaction questionnaires and were assessed by a dermatologist evaluator using several parameters including the modified Global Acne Grading Score (mGAGS), clinical photography, participant self-assessment, physician assessment, and Wong Baker FACES Pain Rating Scale (WBPRS). The physician evaluator was blinded to the side of the face receiving PBBL treatment. Clinical photographs were taken to compare the clinical outcome at each visit versus baseline.
Efficacy Evaluation
Acne Counts
The blinded evaluator counted acne lesions and assessed the mGAGS at each visit prior to administration of the PBBL treatment. Acne lesions were counted separately as noninflammatory (comedones) and inflammatory (papules, pustules, nodules) on the forehead, cheeks, nose, and chin.
Modified Global Acne Grading Score
The modified Global Acne Grading Score was modified from the Global Acne Grading Scale (GAGS) that has previously been used to evaluate acne severity.5 The original GAGS used the type and location of the acne lesions. The GAGS considers 6 locations on the face, chest, and upper back, with a grading factor for each location (forehead=2; cheeks=2; nose=1; chin=1). Another grading factor represented the lesion type (0=no lesion; 1=comedone; 2=papule; 3=pustule; 4=nodule). The local score was calculated by multiplying the location grading factor by the lesion type grading factor. The total score was the sum of the individual local scores for the 4 locations.
Given that the number of acne lesions is important, we modified the GAGS by adding a grading factor that represented the number of lesions to improve the accuracy of the test (1=0–10 lesions; 2=11–20 lesions; 3=21–30 lesions; 4=≥31 lesions). The local score of mGAGS was calculated by multiplying the grading factors for location, lesion type, and number of lesions. Each local score was then added to yield a total score. The mGAGS may be useful and more accurate to determine the severity of acne (0=none; 1–44=mild; 45–80=moderate; 81–132=severe; 133–176=very severe).
Participant Self-assessment
Participants assessed their acne lesions using an 11-point rating scale (–5=100% worsening; –4=76%–99% worsening; –3=51%–75% worsening; –2=26%–50% worsening; –1=1%–25% worsening; 0=no improvement; 1=1%–25% improvement; 2=26%–50% improvement; 3=51%–75% improvement; 4=76%–99% improvement; 5=100% acne clear) to compare their acne at each treatment visit and week 10 follow-up with a baseline photograph.
Physician Assessment
The blinded evaluator assessed acne lesions on the face using the same 11-point rating scale that was used for participant self-assessment. For each participant, assessments were made at each treatment visit and week 10 follow-up by comparing baseline photographs.
Safety Evaluation
The WBPRS score, a standardized 6-point scale (0=no pain; 1=hurts a little bit; 2=hurts a little bit more; 3=hurts even more; 4=hurts whole lot; 5=hurt worst),6 was used to evaluate pain toleration during PBBL treatments and was recorded along with adverse events throughout the study.
Statistical Analysis
Based on data from 2 prior studies,3,7 we expected that the favorable clinical outcome of adapalene gel 0.3% and PBBL therapy would be 23% and 78%, respectively. If the adjunctive therapy with PBBL was beneficial, the favorable outcome would be higher than 78%. To be able to detect this difference, the sample size of 11 patients was needed when 5% type I error and 20% type II error were accepted.
Categorical variables were expressed as percentages, while continuous variables were expressed in terms of median (range). The clinical outcomes between both treatment groups were compared using the Wilcoxon signed rank test. A 2-tailed P value of ≤.05 was considered statistically significant. All statistical calculations were performed using STATA software version 10.0.
RESULTS
Baseline Characteristics
Four male and 7 female patients aged 18 to 35 years (median, 23 years) with mild to moderate acne were enrolled in the study. Of the 11 participants, 7 were white, 2 were black, 1 was Asian, and 1 was Latin American. Baseline characteristics of both sides of the face were comparable in all participants (Table 1). Eight participants (73%) completed the study. Two black participants withdrew from the study due to hyperpigmentation following PBBL treatment; 1 participant did not return for follow-up at week 10, as she was out of the country.

Lesion Counts
At week 3, reduction in noninflammatory lesions was significantly greater on the side receiving the combination therapy compared to the monotherapy side (P=.04)(Table 2). However, there was no significant difference between the combination therapy and the adapalene monotherapy sides in the reduction of noninflammatory and inflammatory lesions at week 4 (Figure 1). There was a remarkable improvement of the combination therapy and adapalene monotherapy sides in acne lesions, but there was no significant difference between the combination therapy and the adapalene monotherapy sides (Figure 2).



Modified Global Acne Grading Score
At weeks 3 and 4, the improvement of mGAGS was significantly greater on the side treated with the combination therapy (P=.05). However, this significant difference was not sustained (Table 3).

Participant Self-assessment and Physician Assessment
The rate of acne improvement according to participant self-assessment was slightly higher on the side receiving the combination therapy compared to the monotherapy side at week 2 (26%–50% vs 1%–25%) and week 6 (76%–99% vs 51%–75%). However, there was no statistically significant difference. For the physician assessment, there was no significant difference between the monotherapy and combination therapy sides.
Safety
The median WBPRS score was 1 (hurts a little bit) throughout all PBBL treatment visits. The maximum score was highest at week 1 (4=hurts whole lot) and subsequently decreased to 2 (hurts a little bit more) at week 6.
After the PBBL treatment, all participants experienced transient erythema in the treatment area. All participants noted their skin had become drier than usual from adapalene, except 1 participant (11%) who reported very dry skin on areas where adapalene gel 0.3% had been applied. However, the dryness was tolerable and relief was reported following application of a moisturizer. No participants withdrew from the study due to skin dryness.
Both black participants experienced hyperpigmentation caused by PBBL (1 on the treatment sites, the other on the test spot) and withdrew from the study. The hyperpigmentation resolved over time following application of a topical bleaching cream. One patient experienced purpura following PBBL treatment at week 4, which was associated with an increase in PBBL power. No other side effects (eg, scaling, stinging, burning, vesicle formation, blistering, crusting, scarring) were observed.
COMMENT
This 10-week study demonstrated that PBBL initially improved the appearance of acne in the first month of treatment, as determined by the significantly greater reduction in mGAGS for the combination side versus the adapalene monotherapy side. Differences in the reduction of acne lesions were not significant between the 2 treatments, except for noninflammatory lesion reduction at week 3. Analysis of physician assessment with photographs revealed acne improvement from baseline in the first month but no additional effects with the PBBL treatment at the end of study. Similarly, participant assessment indicated an improvement by week 2 with the combination therapy compared to adapalene monotherapy in their assessment of acne lesion reductions from baseline. By the end of the study, there was no significant difference between monotherapy and combination therapy.
These findings illustrate that combination therapy with PBBL plus adapalene improved the appearance of acne lesions within the first month of treatment, but there were no further signs of improvement at weeks 5 and 6. These results are consistent with at least 2 other studies that demonstrated acne reduction within the first 3 weeks of PBBL treatment.2,4 The current study was completed as planned with 6 weeks of combination therapy and patients continued adapalene application until the last follow-up visit in week 10. The length of the combination treatment was enough to determine that extension of treatment would not be necessary to gain any further benefits in this study. Because of the small sample size, we would not be able to detect any significant differences, as the difference between the combination therapy and the adapalene monotherapy was less than 55%. Therefore, a future study with a larger sample size is needed to draw a better conclusion.
Pneumatic broadband light has shown impressive results in acne treatment. However, some side effects need to be considered. Minimal adverse events have been reported such as erythema, dryness, peeling, burning, and itching.2-4 In this study, we found that all patients experienced transient erythema during and after PBBL treatment, but this effect disappeared in minutes. Purpura can occur if a higher power of PBBL is performed (6 or greater). Black patients experienced hyperpigmentation that can occur in darker skin types, as reported when light therapy is performed despite using the correct skin type tips.8 Therefore, care must be used in darker skin types, and we advocate a skin test in this population prior to general use.
Our study showed that PBBL can be safely combined with adapalene gel 0.3% and is well tolerated in the treatment of mild to moderate facial acne vulgaris for patients with Fitzpatrick skin types I to III. The combination of PBBL and adapalene reduces acne severity, as shown by the reduction in mGAGS during the first month of treatment. Patients noted faster improvement in their acne lesions with this combination. Although this study was limited by a relatively small sample size, this information may be useful in getting patients to be compliant overall, as they appeared to see results sooner, giving other therapies time to initiate their effect. It appears that 4 consecutive weekly treatments are enough to see that effect. Additionally, this combination therapy provides results without having to resort to oral antibiotics, as many patients today are concerned about creating future antibiotic resistance.
Conclusion
Adapalene gel 0.3% can be safely combined with PBBL for treatment of mild to moderate acne. Although the benefits of this combination therapy can be seen after 4 consecutive weekly treatments, the beneficial effect is not sustained.
Acknowledgment
The authors would like to thank Joyce Okawa, RN (Philadelphia, Pennsylvania), for her assistance in the submission to the University of Pennsylvania institutional review board.
Acne is a common and distressing condition that typically presents in adolescents and young adults and has been associated with not only medical but also emotional and aesthetic consequences. Acne treatments that offer faster improvement are the coveted goal. Although clinical studies support the use of combination therapy with topical retinoids and antibiotics, the overuse of antibiotics raises caution for bacterial resistance.1 Therefore, adjunctive treatments such as chemical peels, light therapy, and laser treatments can hasten the response to traditional acne treatments and in some cases may potentially decrease use of both oral and topical antibiotics.
Light therapy, particularly with visible light, may improve acne outcomes. Pneumatic broadband light (PBBL) is a light treatment in the broadband range (400–1200 nm) combined with a vacuum. The suction created by the vacuum has several effects on acne lesions, such as creating a mechanical lysis of thin-walled pustules and dislodging pore impaction. The blue light with a wavelength of 410 nm targets endogenous porphyrins in Propionibacterium acnes and elicits singlet oxygen production, resulting in bacterial destruction.2,3 Studies showed that PBBL alone was effective in most patients with mild to moderate acne and caused minimal side effects.2-4
We sought to determine if PBBL combined with a topical retinoid can accelerate and prolong acne improvement. We evaluated the efficacy, safety, and tolerability of PBBL plus adapalene gel 0.3% versus adapalene gel 0.3% monotherapy in patients with mild to moderate acne.
METHODSPatient Population
Patients with mild to moderate acne were eligible for the study if they were 18 years or older at screening, in good health, had stopped oral isotretinoin for at least 1 year prior to treatment initiation, and were not taking oral or topical antibiotics or using any topical retinoid derivatives for at least 1 month prior to treatment initiation. Inclusion criteria included at least 10 acne lesions on the face. Patients were excluded if they had a history of receiving PBBL treatment; had a history of scarring, hypopigmentation, or hyperpigmentation from laser or light treatments; and/or were pregnant or refused use of contraception during the study period.
Study Design
This single-blind, randomized, split-face study was approved by the institutional review board of the University of Pennsylvania (Philadelphia, Pennsylvania). All participants provided informed consent before entering the study. Each participant was randomly assigned to receive PBBL on one side of the face for 6 consecutive weeks and apply adapalene gel 0.3% to both sides of the face nightly for 10 weeks. Pneumatic broadband light treatment was performed using the following settings: starting power 2 (approximately 4–6 J/cm2) and vacuum setting 3 (negative pressure, approximately 3 lb/in2). The power setting was increased to a maximum of 6 (12–14 J/cm2) at subsequent visits depending on tolerability of the participants.
All participants visited the clinic weekly for 6 weeks and also returned for follow-up at week 10 (4 weeks following last PBBL treatment). At each visit, the participants completed satisfaction questionnaires and were assessed by a dermatologist evaluator using several parameters including the modified Global Acne Grading Score (mGAGS), clinical photography, participant self-assessment, physician assessment, and Wong Baker FACES Pain Rating Scale (WBPRS). The physician evaluator was blinded to the side of the face receiving PBBL treatment. Clinical photographs were taken to compare the clinical outcome at each visit versus baseline.
Efficacy Evaluation
Acne Counts
The blinded evaluator counted acne lesions and assessed the mGAGS at each visit prior to administration of the PBBL treatment. Acne lesions were counted separately as noninflammatory (comedones) and inflammatory (papules, pustules, nodules) on the forehead, cheeks, nose, and chin.
Modified Global Acne Grading Score
The modified Global Acne Grading Score was modified from the Global Acne Grading Scale (GAGS) that has previously been used to evaluate acne severity.5 The original GAGS used the type and location of the acne lesions. The GAGS considers 6 locations on the face, chest, and upper back, with a grading factor for each location (forehead=2; cheeks=2; nose=1; chin=1). Another grading factor represented the lesion type (0=no lesion; 1=comedone; 2=papule; 3=pustule; 4=nodule). The local score was calculated by multiplying the location grading factor by the lesion type grading factor. The total score was the sum of the individual local scores for the 4 locations.
Given that the number of acne lesions is important, we modified the GAGS by adding a grading factor that represented the number of lesions to improve the accuracy of the test (1=0–10 lesions; 2=11–20 lesions; 3=21–30 lesions; 4=≥31 lesions). The local score of mGAGS was calculated by multiplying the grading factors for location, lesion type, and number of lesions. Each local score was then added to yield a total score. The mGAGS may be useful and more accurate to determine the severity of acne (0=none; 1–44=mild; 45–80=moderate; 81–132=severe; 133–176=very severe).
Participant Self-assessment
Participants assessed their acne lesions using an 11-point rating scale (–5=100% worsening; –4=76%–99% worsening; –3=51%–75% worsening; –2=26%–50% worsening; –1=1%–25% worsening; 0=no improvement; 1=1%–25% improvement; 2=26%–50% improvement; 3=51%–75% improvement; 4=76%–99% improvement; 5=100% acne clear) to compare their acne at each treatment visit and week 10 follow-up with a baseline photograph.
Physician Assessment
The blinded evaluator assessed acne lesions on the face using the same 11-point rating scale that was used for participant self-assessment. For each participant, assessments were made at each treatment visit and week 10 follow-up by comparing baseline photographs.
Safety Evaluation
The WBPRS score, a standardized 6-point scale (0=no pain; 1=hurts a little bit; 2=hurts a little bit more; 3=hurts even more; 4=hurts whole lot; 5=hurt worst),6 was used to evaluate pain toleration during PBBL treatments and was recorded along with adverse events throughout the study.
Statistical Analysis
Based on data from 2 prior studies,3,7 we expected that the favorable clinical outcome of adapalene gel 0.3% and PBBL therapy would be 23% and 78%, respectively. If the adjunctive therapy with PBBL was beneficial, the favorable outcome would be higher than 78%. To be able to detect this difference, the sample size of 11 patients was needed when 5% type I error and 20% type II error were accepted.
Categorical variables were expressed as percentages, while continuous variables were expressed in terms of median (range). The clinical outcomes between both treatment groups were compared using the Wilcoxon signed rank test. A 2-tailed P value of ≤.05 was considered statistically significant. All statistical calculations were performed using STATA software version 10.0.
RESULTS
Baseline Characteristics
Four male and 7 female patients aged 18 to 35 years (median, 23 years) with mild to moderate acne were enrolled in the study. Of the 11 participants, 7 were white, 2 were black, 1 was Asian, and 1 was Latin American. Baseline characteristics of both sides of the face were comparable in all participants (Table 1). Eight participants (73%) completed the study. Two black participants withdrew from the study due to hyperpigmentation following PBBL treatment; 1 participant did not return for follow-up at week 10, as she was out of the country.

Lesion Counts
At week 3, reduction in noninflammatory lesions was significantly greater on the side receiving the combination therapy compared to the monotherapy side (P=.04)(Table 2). However, there was no significant difference between the combination therapy and the adapalene monotherapy sides in the reduction of noninflammatory and inflammatory lesions at week 4 (Figure 1). There was a remarkable improvement of the combination therapy and adapalene monotherapy sides in acne lesions, but there was no significant difference between the combination therapy and the adapalene monotherapy sides (Figure 2).



Modified Global Acne Grading Score
At weeks 3 and 4, the improvement of mGAGS was significantly greater on the side treated with the combination therapy (P=.05). However, this significant difference was not sustained (Table 3).

Participant Self-assessment and Physician Assessment
The rate of acne improvement according to participant self-assessment was slightly higher on the side receiving the combination therapy compared to the monotherapy side at week 2 (26%–50% vs 1%–25%) and week 6 (76%–99% vs 51%–75%). However, there was no statistically significant difference. For the physician assessment, there was no significant difference between the monotherapy and combination therapy sides.
Safety
The median WBPRS score was 1 (hurts a little bit) throughout all PBBL treatment visits. The maximum score was highest at week 1 (4=hurts whole lot) and subsequently decreased to 2 (hurts a little bit more) at week 6.
After the PBBL treatment, all participants experienced transient erythema in the treatment area. All participants noted their skin had become drier than usual from adapalene, except 1 participant (11%) who reported very dry skin on areas where adapalene gel 0.3% had been applied. However, the dryness was tolerable and relief was reported following application of a moisturizer. No participants withdrew from the study due to skin dryness.
Both black participants experienced hyperpigmentation caused by PBBL (1 on the treatment sites, the other on the test spot) and withdrew from the study. The hyperpigmentation resolved over time following application of a topical bleaching cream. One patient experienced purpura following PBBL treatment at week 4, which was associated with an increase in PBBL power. No other side effects (eg, scaling, stinging, burning, vesicle formation, blistering, crusting, scarring) were observed.
COMMENT
This 10-week study demonstrated that PBBL initially improved the appearance of acne in the first month of treatment, as determined by the significantly greater reduction in mGAGS for the combination side versus the adapalene monotherapy side. Differences in the reduction of acne lesions were not significant between the 2 treatments, except for noninflammatory lesion reduction at week 3. Analysis of physician assessment with photographs revealed acne improvement from baseline in the first month but no additional effects with the PBBL treatment at the end of study. Similarly, participant assessment indicated an improvement by week 2 with the combination therapy compared to adapalene monotherapy in their assessment of acne lesion reductions from baseline. By the end of the study, there was no significant difference between monotherapy and combination therapy.
These findings illustrate that combination therapy with PBBL plus adapalene improved the appearance of acne lesions within the first month of treatment, but there were no further signs of improvement at weeks 5 and 6. These results are consistent with at least 2 other studies that demonstrated acne reduction within the first 3 weeks of PBBL treatment.2,4 The current study was completed as planned with 6 weeks of combination therapy and patients continued adapalene application until the last follow-up visit in week 10. The length of the combination treatment was enough to determine that extension of treatment would not be necessary to gain any further benefits in this study. Because of the small sample size, we would not be able to detect any significant differences, as the difference between the combination therapy and the adapalene monotherapy was less than 55%. Therefore, a future study with a larger sample size is needed to draw a better conclusion.
Pneumatic broadband light has shown impressive results in acne treatment. However, some side effects need to be considered. Minimal adverse events have been reported such as erythema, dryness, peeling, burning, and itching.2-4 In this study, we found that all patients experienced transient erythema during and after PBBL treatment, but this effect disappeared in minutes. Purpura can occur if a higher power of PBBL is performed (6 or greater). Black patients experienced hyperpigmentation that can occur in darker skin types, as reported when light therapy is performed despite using the correct skin type tips.8 Therefore, care must be used in darker skin types, and we advocate a skin test in this population prior to general use.
Our study showed that PBBL can be safely combined with adapalene gel 0.3% and is well tolerated in the treatment of mild to moderate facial acne vulgaris for patients with Fitzpatrick skin types I to III. The combination of PBBL and adapalene reduces acne severity, as shown by the reduction in mGAGS during the first month of treatment. Patients noted faster improvement in their acne lesions with this combination. Although this study was limited by a relatively small sample size, this information may be useful in getting patients to be compliant overall, as they appeared to see results sooner, giving other therapies time to initiate their effect. It appears that 4 consecutive weekly treatments are enough to see that effect. Additionally, this combination therapy provides results without having to resort to oral antibiotics, as many patients today are concerned about creating future antibiotic resistance.
Conclusion
Adapalene gel 0.3% can be safely combined with PBBL for treatment of mild to moderate acne. Although the benefits of this combination therapy can be seen after 4 consecutive weekly treatments, the beneficial effect is not sustained.
Acknowledgment
The authors would like to thank Joyce Okawa, RN (Philadelphia, Pennsylvania), for her assistance in the submission to the University of Pennsylvania institutional review board.
- Thiboutot D, Gollnick H, Bettoli V, et al. New insights into the management of acne: an update from the Global Alliance to Improve Outcomes in Acne group. J Am Acad Dermatol. 2009;60(5 suppl):S1-S50.
- Gold MH, Biron J. Efficacy of a novel combination of pneumatic energy and broadband light for the treatment of acne. J Drugs Dermatol. 2008;7:639-642.
- Shamban AT, Enokibori M, Narurkar V, et al. Photopneumatic technology for the treatment of acne vulgaris. J Drugs Dermatol. 2008;7:139-145.
- Wanitphakdeedecha R, Tanzi EL, Alster TS. Photopneumatic therapy for the treatment of acne. J Drugs Dermatol. 2009;8:239-241.
- Doshi A, Zaheer A, Stiller MJ. A comparison of current acne grading systems and proposal of a novel system. Int J Dermatol. 1997;36:416-418.
- Wong DL, Baker CM. Pain in children: comparison of assessment scales. Pediatr Nurs. 1988;14:9-17.
- Thiboutot D, Pariser DM, Egan N, et al. Adapalene gel 0.3% for the treatment of acne vulgaris: a multicenter, randomized, double-blind, controlled, phase III trial. J Am Acad Dermatol. 2006;54:242-250.
- Yeung CK, Shek SY, Bjerring P, et al. A comparative study of intense pulsed light alone and its combination with photodynamic therapy for the treatment of facial acne in Asian skin. Lasers Surg Med. 2007;39:1-6.
- Thiboutot D, Gollnick H, Bettoli V, et al. New insights into the management of acne: an update from the Global Alliance to Improve Outcomes in Acne group. J Am Acad Dermatol. 2009;60(5 suppl):S1-S50.
- Gold MH, Biron J. Efficacy of a novel combination of pneumatic energy and broadband light for the treatment of acne. J Drugs Dermatol. 2008;7:639-642.
- Shamban AT, Enokibori M, Narurkar V, et al. Photopneumatic technology for the treatment of acne vulgaris. J Drugs Dermatol. 2008;7:139-145.
- Wanitphakdeedecha R, Tanzi EL, Alster TS. Photopneumatic therapy for the treatment of acne. J Drugs Dermatol. 2009;8:239-241.
- Doshi A, Zaheer A, Stiller MJ. A comparison of current acne grading systems and proposal of a novel system. Int J Dermatol. 1997;36:416-418.
- Wong DL, Baker CM. Pain in children: comparison of assessment scales. Pediatr Nurs. 1988;14:9-17.
- Thiboutot D, Pariser DM, Egan N, et al. Adapalene gel 0.3% for the treatment of acne vulgaris: a multicenter, randomized, double-blind, controlled, phase III trial. J Am Acad Dermatol. 2006;54:242-250.
- Yeung CK, Shek SY, Bjerring P, et al. A comparative study of intense pulsed light alone and its combination with photodynamic therapy for the treatment of facial acne in Asian skin. Lasers Surg Med. 2007;39:1-6.
Practice Points
- Compliance is achieved when patients can see improvements with their acne treatments quickly.
- Combination therapy achieves the goal of a quicker visual improvement of acneform pustules and papules with pneumatic broadband light while topical acne treatments have a chance to work, thus increasing compliance.