User login
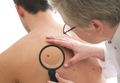
Sell skin care products to protect your patients
The ethics behind selling skin care products to patients has been hotly debated within the field of cosmetic dermatology for several decades. In 15 years of practice, I have come to the conclusion that patients want you to and need you to because otherwise they are easily taken advantage of. Other physicians are doing it but we – the dermatologists – are the most qualified to offer skin care advice. This article will discuss the reasons that you need to get over the ethical dilemma and offer skin care to your patients.
Using the correct skin care regimen for the face and body will improve outcomes
Whether a patient suffers from acne, rosacea, melasma, psoriasis, eczema, contact dermatitis, or even tinea versicolor, using the proper skin care regimen will improve outcomes by affecting the skin barrier, pH, hydration level, and function of the keratinocytes and fibroblasts. In fact, every personal care product that touches the skin has an impact on skin health. For example, if a patient uses a detergent-laden bar soap, the skin barrier will be impaired, which can cause them to react to allergens and irritants. Personal care products can affect the skin pH; this is shown to play a role in Malassezia colonization in atopic dermatitis patients (J Clin Med. 2015;4[6]:1217-28). As dermatologists, we know better than anyone that daily use of SPF improves skin health and lowers the risk of postinflammatory pigmentation. We all agree that patients should cleanse the skin and apply a SPF every day. Giving them guidance about which to choose is very important.
Giving the patient exact instructions will lead to improved compliance
Why should recommending skin care products be perceived differently than prescribing a prescription medication? We should prescribe to our patients in writing the exact skin care regimen they should use for their face or body to ensure that they understand the directions. I have been surprised by patients who have said, “I did not know I was supposed to wash my cleanser off,” or “I wash my face with hand soap.” We can help them by educating them and giving them specific instructions. Improved education and communication results in increased compliance. When you do surgery on a wound, you probably tell them to apply a topical antibiotic ointment, but do you direct them to what cleanser to use or tell them which SPF to use on the stitched wound? Providing written instructions for all dermatologic disorders and postprocedure care is necessary to improve compliance and outcomes.
Combine cosmeceuticals, prescription medications, and medical procedures
You (unlike the cosmetic counter salesperson) have the ability to combine cosmeceuticals with prescription medications and medical procedures. In fact, selling your patients the right skin care products to use after a procedure saves them a trip to the store and ensures that they use the correct products. Of course it makes sense that patients getting toxins and fillers should use a retinoid to improve skin aging; however, many general dermatologic diseases would improve with the proper skin care. For example, do you use biologics for psoriasis? Using the proper skin care to regulate skin pH and improve the skin barrier may help prevent colonization of yeast, fungus, and bacteria. The same is true for atopic patients. Do you use liquid nitrogen? Studies show that using a retinoid before a procedure speeds healing. Skin care goes way beyond wrinkles and dark circles under the eyes, so if you are not prescribing the patient an exact regimen, you are not maximizing outcomes.
I don’t have time to talk to my patients about skin care
The missing piece is that most of us don’t have the time to spend discussing skin care. This is where using a standardized scientific methodology is crucial. I developed and use a skin typing methodology in my office and have seen improved physician/patient relationships and increased patient satisfaction resulting in a significant amount of referrals. We also have noted decreased call backs and fewer adverse events from products because the patients have a better understanding of how to properly apply the cosmeceuticals and prescription products. The best part is, it does not add any time onto the patient visit when standardized methodologies are properly adopted.
What if I still do not feel comfortable profiting from the sale of skin care products?
First you need to realize that time is money and you are saving the patient the cost in time it would have taken them to go to a store, park, and shop for the correct product. I have seen data presented from several companies that show that patients usually spend a large amount of money on skin care products after they see their dermatologist. Without guidance, they will likely buy the incorrect products. If they buy the wrong product, you save them the hassle of having to make another office visit and the aggravation of the side effects from the incorrect product. These are often of poor quality or not appropriate for their skin issues. Counterfeit products are rampant on the Internet and many new companies tout worthless products with stem cells and other nonsense. Only you can help your patients make sure that money is spent on the proper products.
Conclusion
Do you really want someone else giving your patients skin care advice? Your patients deserve to have someone with your insights, knowledge, compassion, and honesty help them achieve optimal skin health through use of the proper cosmeceuticals and prescription medications. It is up to you and your staff to save your patients from falling prey to persuasive salespeople with no scientific knowledge or concern for long-term skin health.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. Dr. Baumann also developed and owns the Baumann Skin Type Solution skin typing systems and related products.
The ethics behind selling skin care products to patients has been hotly debated within the field of cosmetic dermatology for several decades. In 15 years of practice, I have come to the conclusion that patients want you to and need you to because otherwise they are easily taken advantage of. Other physicians are doing it but we – the dermatologists – are the most qualified to offer skin care advice. This article will discuss the reasons that you need to get over the ethical dilemma and offer skin care to your patients.
Using the correct skin care regimen for the face and body will improve outcomes
Whether a patient suffers from acne, rosacea, melasma, psoriasis, eczema, contact dermatitis, or even tinea versicolor, using the proper skin care regimen will improve outcomes by affecting the skin barrier, pH, hydration level, and function of the keratinocytes and fibroblasts. In fact, every personal care product that touches the skin has an impact on skin health. For example, if a patient uses a detergent-laden bar soap, the skin barrier will be impaired, which can cause them to react to allergens and irritants. Personal care products can affect the skin pH; this is shown to play a role in Malassezia colonization in atopic dermatitis patients (J Clin Med. 2015;4[6]:1217-28). As dermatologists, we know better than anyone that daily use of SPF improves skin health and lowers the risk of postinflammatory pigmentation. We all agree that patients should cleanse the skin and apply a SPF every day. Giving them guidance about which to choose is very important.
Giving the patient exact instructions will lead to improved compliance
Why should recommending skin care products be perceived differently than prescribing a prescription medication? We should prescribe to our patients in writing the exact skin care regimen they should use for their face or body to ensure that they understand the directions. I have been surprised by patients who have said, “I did not know I was supposed to wash my cleanser off,” or “I wash my face with hand soap.” We can help them by educating them and giving them specific instructions. Improved education and communication results in increased compliance. When you do surgery on a wound, you probably tell them to apply a topical antibiotic ointment, but do you direct them to what cleanser to use or tell them which SPF to use on the stitched wound? Providing written instructions for all dermatologic disorders and postprocedure care is necessary to improve compliance and outcomes.
Combine cosmeceuticals, prescription medications, and medical procedures
You (unlike the cosmetic counter salesperson) have the ability to combine cosmeceuticals with prescription medications and medical procedures. In fact, selling your patients the right skin care products to use after a procedure saves them a trip to the store and ensures that they use the correct products. Of course it makes sense that patients getting toxins and fillers should use a retinoid to improve skin aging; however, many general dermatologic diseases would improve with the proper skin care. For example, do you use biologics for psoriasis? Using the proper skin care to regulate skin pH and improve the skin barrier may help prevent colonization of yeast, fungus, and bacteria. The same is true for atopic patients. Do you use liquid nitrogen? Studies show that using a retinoid before a procedure speeds healing. Skin care goes way beyond wrinkles and dark circles under the eyes, so if you are not prescribing the patient an exact regimen, you are not maximizing outcomes.
I don’t have time to talk to my patients about skin care
The missing piece is that most of us don’t have the time to spend discussing skin care. This is where using a standardized scientific methodology is crucial. I developed and use a skin typing methodology in my office and have seen improved physician/patient relationships and increased patient satisfaction resulting in a significant amount of referrals. We also have noted decreased call backs and fewer adverse events from products because the patients have a better understanding of how to properly apply the cosmeceuticals and prescription products. The best part is, it does not add any time onto the patient visit when standardized methodologies are properly adopted.
What if I still do not feel comfortable profiting from the sale of skin care products?
First you need to realize that time is money and you are saving the patient the cost in time it would have taken them to go to a store, park, and shop for the correct product. I have seen data presented from several companies that show that patients usually spend a large amount of money on skin care products after they see their dermatologist. Without guidance, they will likely buy the incorrect products. If they buy the wrong product, you save them the hassle of having to make another office visit and the aggravation of the side effects from the incorrect product. These are often of poor quality or not appropriate for their skin issues. Counterfeit products are rampant on the Internet and many new companies tout worthless products with stem cells and other nonsense. Only you can help your patients make sure that money is spent on the proper products.
Conclusion
Do you really want someone else giving your patients skin care advice? Your patients deserve to have someone with your insights, knowledge, compassion, and honesty help them achieve optimal skin health through use of the proper cosmeceuticals and prescription medications. It is up to you and your staff to save your patients from falling prey to persuasive salespeople with no scientific knowledge or concern for long-term skin health.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. Dr. Baumann also developed and owns the Baumann Skin Type Solution skin typing systems and related products.
The ethics behind selling skin care products to patients has been hotly debated within the field of cosmetic dermatology for several decades. In 15 years of practice, I have come to the conclusion that patients want you to and need you to because otherwise they are easily taken advantage of. Other physicians are doing it but we – the dermatologists – are the most qualified to offer skin care advice. This article will discuss the reasons that you need to get over the ethical dilemma and offer skin care to your patients.
Using the correct skin care regimen for the face and body will improve outcomes
Whether a patient suffers from acne, rosacea, melasma, psoriasis, eczema, contact dermatitis, or even tinea versicolor, using the proper skin care regimen will improve outcomes by affecting the skin barrier, pH, hydration level, and function of the keratinocytes and fibroblasts. In fact, every personal care product that touches the skin has an impact on skin health. For example, if a patient uses a detergent-laden bar soap, the skin barrier will be impaired, which can cause them to react to allergens and irritants. Personal care products can affect the skin pH; this is shown to play a role in Malassezia colonization in atopic dermatitis patients (J Clin Med. 2015;4[6]:1217-28). As dermatologists, we know better than anyone that daily use of SPF improves skin health and lowers the risk of postinflammatory pigmentation. We all agree that patients should cleanse the skin and apply a SPF every day. Giving them guidance about which to choose is very important.
Giving the patient exact instructions will lead to improved compliance
Why should recommending skin care products be perceived differently than prescribing a prescription medication? We should prescribe to our patients in writing the exact skin care regimen they should use for their face or body to ensure that they understand the directions. I have been surprised by patients who have said, “I did not know I was supposed to wash my cleanser off,” or “I wash my face with hand soap.” We can help them by educating them and giving them specific instructions. Improved education and communication results in increased compliance. When you do surgery on a wound, you probably tell them to apply a topical antibiotic ointment, but do you direct them to what cleanser to use or tell them which SPF to use on the stitched wound? Providing written instructions for all dermatologic disorders and postprocedure care is necessary to improve compliance and outcomes.
Combine cosmeceuticals, prescription medications, and medical procedures
You (unlike the cosmetic counter salesperson) have the ability to combine cosmeceuticals with prescription medications and medical procedures. In fact, selling your patients the right skin care products to use after a procedure saves them a trip to the store and ensures that they use the correct products. Of course it makes sense that patients getting toxins and fillers should use a retinoid to improve skin aging; however, many general dermatologic diseases would improve with the proper skin care. For example, do you use biologics for psoriasis? Using the proper skin care to regulate skin pH and improve the skin barrier may help prevent colonization of yeast, fungus, and bacteria. The same is true for atopic patients. Do you use liquid nitrogen? Studies show that using a retinoid before a procedure speeds healing. Skin care goes way beyond wrinkles and dark circles under the eyes, so if you are not prescribing the patient an exact regimen, you are not maximizing outcomes.
I don’t have time to talk to my patients about skin care
The missing piece is that most of us don’t have the time to spend discussing skin care. This is where using a standardized scientific methodology is crucial. I developed and use a skin typing methodology in my office and have seen improved physician/patient relationships and increased patient satisfaction resulting in a significant amount of referrals. We also have noted decreased call backs and fewer adverse events from products because the patients have a better understanding of how to properly apply the cosmeceuticals and prescription products. The best part is, it does not add any time onto the patient visit when standardized methodologies are properly adopted.
What if I still do not feel comfortable profiting from the sale of skin care products?
First you need to realize that time is money and you are saving the patient the cost in time it would have taken them to go to a store, park, and shop for the correct product. I have seen data presented from several companies that show that patients usually spend a large amount of money on skin care products after they see their dermatologist. Without guidance, they will likely buy the incorrect products. If they buy the wrong product, you save them the hassle of having to make another office visit and the aggravation of the side effects from the incorrect product. These are often of poor quality or not appropriate for their skin issues. Counterfeit products are rampant on the Internet and many new companies tout worthless products with stem cells and other nonsense. Only you can help your patients make sure that money is spent on the proper products.
Conclusion
Do you really want someone else giving your patients skin care advice? Your patients deserve to have someone with your insights, knowledge, compassion, and honesty help them achieve optimal skin health through use of the proper cosmeceuticals and prescription medications. It is up to you and your staff to save your patients from falling prey to persuasive salespeople with no scientific knowledge or concern for long-term skin health.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. Dr. Baumann also developed and owns the Baumann Skin Type Solution skin typing systems and related products.
• Most skin care products that patients buy are not appropriate for their skin issues.
• Dermatologists have the most knowledge and insights to prescribe skin care.
• Giving specific skin care instructions helps improve communication.
• Increased communication improves outcomes.
Oxandrolone, propranolol combo increases growth in severely burned children
CHICAGO – Combination therapy with oxandrolone and propranolol can attenuate burn-induced growth arrest and increase growth rate in severely burned children, according to findings from a prospective, randomized clinical trial.
Of 612 children with burns over at least 30% of their total body surface area (average of more than 50%), 103 were randomized to receive treatment with both oxandrolone and propranolol, 67 received oxandrolone alone, 194 received propranolol alone, and 248 served as controls. After a minimum of 1 year of treatment, the average growth rate was 5.9 cm in the control group and 7.6 cm in the group receiving combination therapy, Dr. David N. Herndon of the University of Texas Medical Branch at Galveston reported at the annual meeting of the American Surgical Association.
“The rate of growth with combination therapy was significantly greater than with either of the individual drugs alone,” Dr. Herndon said.
Further, the period of growth arrest was significantly shorter – by 84 days – among those in the combination-treatment group, compared with those in the control group.
Study subjects were children treated at Shriners Hospitals for Children – Galveston from 1997 to 2015. Boys aged 6 months to 14 years and girls aged 6 months to 12 years were included to eliminate the variable onset of postpubescent growth delay. About two-thirds in each group were boys, and the ages in the patient groups were similar. Mortality was low and was similar across the groups, as was hospital length of stay.
Dr. Herndon and his colleagues controlled for heterogeneous burn distribution between the groups in the course of their analyses, as well as age.
In children with severe, extensive burn injury, the hypercatabolic response is mediated by increased production of catecholamines and corticosteroids, coupled with decreased production of testosterone. This contributes to growth arrest and to decreased strength for up to 2 years after burn injury, he explained. Children with burns over 50% of their total body surface routinely survive acute hospitalization but, at 3 months post injury, are thin, have difficulty walking, and require occupational and physical therapy to help them perform even the simplest activities of daily living.
At 1 year, a raised inflammatory mass covers their wounds, they experience itching, and they have, in large part, stunted growth; there is severe loss of lean body mass and strength, and fracture risk is increased, Dr. Herndon said.
In previous work, he and his colleagues showed that administration of propranolol at an average dose of 4 mg/kg per day for 1 year decreased cardiac work and resting energy expenditure while increasing peripheral lean mass. Further, they found that the testosterone analog oxandrolone, given at 0.1 mg/kg twice per day for 1 year, improved lean body mass accretion and bone mineral content.
The current study was conducted to test the effects of administering both agents in combination.
“The combined use of oxandrolone and propranolol in severely burned children confers an additional benefit on growth over either treatment alone,” Dr. Herndon said, adding that the additive effects of combination therapy may be due to the effects of oxandrolone on bone growth and the anti-inflammatory effects of propranolol.
“The additional benefits point out mechanistic changes that may be eventful in the treatment of hypermetabolism generally and in inflammatory states,” he concluded.
Dr. Herndon reported having no disclosures.
CHICAGO – Combination therapy with oxandrolone and propranolol can attenuate burn-induced growth arrest and increase growth rate in severely burned children, according to findings from a prospective, randomized clinical trial.
Of 612 children with burns over at least 30% of their total body surface area (average of more than 50%), 103 were randomized to receive treatment with both oxandrolone and propranolol, 67 received oxandrolone alone, 194 received propranolol alone, and 248 served as controls. After a minimum of 1 year of treatment, the average growth rate was 5.9 cm in the control group and 7.6 cm in the group receiving combination therapy, Dr. David N. Herndon of the University of Texas Medical Branch at Galveston reported at the annual meeting of the American Surgical Association.
“The rate of growth with combination therapy was significantly greater than with either of the individual drugs alone,” Dr. Herndon said.
Further, the period of growth arrest was significantly shorter – by 84 days – among those in the combination-treatment group, compared with those in the control group.
Study subjects were children treated at Shriners Hospitals for Children – Galveston from 1997 to 2015. Boys aged 6 months to 14 years and girls aged 6 months to 12 years were included to eliminate the variable onset of postpubescent growth delay. About two-thirds in each group were boys, and the ages in the patient groups were similar. Mortality was low and was similar across the groups, as was hospital length of stay.
Dr. Herndon and his colleagues controlled for heterogeneous burn distribution between the groups in the course of their analyses, as well as age.
In children with severe, extensive burn injury, the hypercatabolic response is mediated by increased production of catecholamines and corticosteroids, coupled with decreased production of testosterone. This contributes to growth arrest and to decreased strength for up to 2 years after burn injury, he explained. Children with burns over 50% of their total body surface routinely survive acute hospitalization but, at 3 months post injury, are thin, have difficulty walking, and require occupational and physical therapy to help them perform even the simplest activities of daily living.
At 1 year, a raised inflammatory mass covers their wounds, they experience itching, and they have, in large part, stunted growth; there is severe loss of lean body mass and strength, and fracture risk is increased, Dr. Herndon said.
In previous work, he and his colleagues showed that administration of propranolol at an average dose of 4 mg/kg per day for 1 year decreased cardiac work and resting energy expenditure while increasing peripheral lean mass. Further, they found that the testosterone analog oxandrolone, given at 0.1 mg/kg twice per day for 1 year, improved lean body mass accretion and bone mineral content.
The current study was conducted to test the effects of administering both agents in combination.
“The combined use of oxandrolone and propranolol in severely burned children confers an additional benefit on growth over either treatment alone,” Dr. Herndon said, adding that the additive effects of combination therapy may be due to the effects of oxandrolone on bone growth and the anti-inflammatory effects of propranolol.
“The additional benefits point out mechanistic changes that may be eventful in the treatment of hypermetabolism generally and in inflammatory states,” he concluded.
Dr. Herndon reported having no disclosures.
CHICAGO – Combination therapy with oxandrolone and propranolol can attenuate burn-induced growth arrest and increase growth rate in severely burned children, according to findings from a prospective, randomized clinical trial.
Of 612 children with burns over at least 30% of their total body surface area (average of more than 50%), 103 were randomized to receive treatment with both oxandrolone and propranolol, 67 received oxandrolone alone, 194 received propranolol alone, and 248 served as controls. After a minimum of 1 year of treatment, the average growth rate was 5.9 cm in the control group and 7.6 cm in the group receiving combination therapy, Dr. David N. Herndon of the University of Texas Medical Branch at Galveston reported at the annual meeting of the American Surgical Association.
“The rate of growth with combination therapy was significantly greater than with either of the individual drugs alone,” Dr. Herndon said.
Further, the period of growth arrest was significantly shorter – by 84 days – among those in the combination-treatment group, compared with those in the control group.
Study subjects were children treated at Shriners Hospitals for Children – Galveston from 1997 to 2015. Boys aged 6 months to 14 years and girls aged 6 months to 12 years were included to eliminate the variable onset of postpubescent growth delay. About two-thirds in each group were boys, and the ages in the patient groups were similar. Mortality was low and was similar across the groups, as was hospital length of stay.
Dr. Herndon and his colleagues controlled for heterogeneous burn distribution between the groups in the course of their analyses, as well as age.
In children with severe, extensive burn injury, the hypercatabolic response is mediated by increased production of catecholamines and corticosteroids, coupled with decreased production of testosterone. This contributes to growth arrest and to decreased strength for up to 2 years after burn injury, he explained. Children with burns over 50% of their total body surface routinely survive acute hospitalization but, at 3 months post injury, are thin, have difficulty walking, and require occupational and physical therapy to help them perform even the simplest activities of daily living.
At 1 year, a raised inflammatory mass covers their wounds, they experience itching, and they have, in large part, stunted growth; there is severe loss of lean body mass and strength, and fracture risk is increased, Dr. Herndon said.
In previous work, he and his colleagues showed that administration of propranolol at an average dose of 4 mg/kg per day for 1 year decreased cardiac work and resting energy expenditure while increasing peripheral lean mass. Further, they found that the testosterone analog oxandrolone, given at 0.1 mg/kg twice per day for 1 year, improved lean body mass accretion and bone mineral content.
The current study was conducted to test the effects of administering both agents in combination.
“The combined use of oxandrolone and propranolol in severely burned children confers an additional benefit on growth over either treatment alone,” Dr. Herndon said, adding that the additive effects of combination therapy may be due to the effects of oxandrolone on bone growth and the anti-inflammatory effects of propranolol.
“The additional benefits point out mechanistic changes that may be eventful in the treatment of hypermetabolism generally and in inflammatory states,” he concluded.
Dr. Herndon reported having no disclosures.
AT THE ASA ANNUAL MEETING
Key clinical point: Combination therapy with oxandrolone and propranolol can attenuate burn-induced growth arrest and increase growth rate in severely burned children, according to findings from a prospective, randomized clinical trial.
Major finding: After a minimum of 1 year of treatment, the average growth rate was 5.9 cm in the control group and 7.6 cm in the group receiving combination therapy.
Data source: A prospective, randomized clinical trial involving 612 children.
Disclosures: Dr. Herndon reported having no disclosures.
Ibuprofen plus acetaminophen works well for postop Mohs pain
EXPERT ANALYSIS FROM THE ACMS ANNUAL MEETING
ORLANDO – An alternating schedule of ibuprofen and acetaminophen every 3 hours is an excellent method of managing postoperative pain associated with Mohs surgery, especially if the initial dose is taken at the start of the procedure.
By starting with ibuprofen, the regimen capitalizes on the drug’s anti-inflammatory component to reduce overall postoperative analgesic requirement, Dr. Bryan Carroll said at the annual meeting of the American College of Mohs Surgery.
“This combination has even been shown to be superior to narcotics, both alone and in combination,” said Dr. Carroll, director of dermatologic surgery at Eastern Virginia Medical School, Norfolk. “This finding has been reinforced in the Mohs literature,” he added, citing a study that found the combination of acetaminophen and ibuprofen was more effective than acetaminophen alone or acetaminophen with codeine in controlling pain after Mohs surgery and reconstruction (Dermatol Surg. 2011 Jul;37[7]:1007-13).
Alternately layering the analgesics allows both to build to a maximum concentration in the blood without any nadirs where pain can get a foothold, an important concept in pain management, Dr. Carroll said.
And having two medications on board allows simultaneous targeting of different portions of the pain signaling pathway, he added. Ibuprofen works at the points of transduction and transmission, while acetaminophen works at the points of transmission and perception.
Dr. Carroll’s regimen starts at the time of surgery, when patients receive 400 mg ibuprofen. Three hours later, they receive 1 gram of acetaminophen; this dose should be adjusted for patients older than 60 years, who should not get more than 3 grams in 24 hours, and for those with liver failure, who should be limited to 2 grams over 24 hours.
This alternating dose is repeated every 3 hours. By the time of discharge, most patients have had at least two doses. This schedule is usually sufficient for patients at moderate to high risk of uncontrolled pain, who can then manage their discomfort with either drug the next day, he said.
Patients at higher risk of uncontrolled pain can use the regimen for the first day, and then titrate off according to their comfort. Some of these patients, however, may benefit from oxycodone, with the addition of laxative and an antiemetic, he noted.
The layering technique provides consistent postoperative pain relief that’s effective for most patients – even those who undergo substantial reconstruction, Dr. Carroll said in an interview. “This schedule is sufficient for all of our procedures, including larger reconstructions such as forehead flaps and cervicofacial rotation flaps. But additional interventions are indicated for patients with a high risk of uncontrolled pain. It’s the patient, not the procedure, which determines need for escalation.”
Teasing out those patients who may need more assertive pain management should be done in a preoperative assessment, Dr. Carroll said. A patient’s expectations of pain and history of chronic pain are some of the biggest factors in predicting a patient who will have uncontrolled pain.
“The experience of pain in Mohs surgery has limited studies,” he said. “Only a handful of investigations have looked at predictors that could help us plan. But of these, two things do stand out: a patient’s expectation of pain and a patient’s history of chronic pain.”
Surprisingly, he said, studies have determined that even a modestly elevated expectation of pain is enough to tip patients into a high-risk category. “If a patient predicted that his pain would be a 4 on a 1-10 scale, that was correlated with a lack of pain control during the operative experience. Maybe we’d expect this correlation if the expectation was an 8 or a 10, but a 4 was surprising. If a patient has even that amount of concern, I start thinking about additional interventions I can provide to maximize comfort.”
A patient’s past experience with pain is also a very large factor in how that person will experience postoperative pain. “Chronic pain does correlate with uncontrolled pain during surgery. I always ask about it. And this talk also helps drive your conversation about what you will be doing to keep them comfortable.”
That chat should include an explanation of how chronic and acute pain differ, Dr. Carroll said. “Chronic and acute pain involve different pathways and need different interventions. If the patient expresses fear, saying something like, ‘Tylenol is like water to me,’ believe him. It is like water for chronic pain. But you can also tell that patient that chronic pain is different from acute pain, and that acetaminophen will be a part of successfully managing it.”
Dr. Carroll had no financial disclosures.
EXPERT ANALYSIS FROM THE ACMS ANNUAL MEETING
ORLANDO – An alternating schedule of ibuprofen and acetaminophen every 3 hours is an excellent method of managing postoperative pain associated with Mohs surgery, especially if the initial dose is taken at the start of the procedure.
By starting with ibuprofen, the regimen capitalizes on the drug’s anti-inflammatory component to reduce overall postoperative analgesic requirement, Dr. Bryan Carroll said at the annual meeting of the American College of Mohs Surgery.
“This combination has even been shown to be superior to narcotics, both alone and in combination,” said Dr. Carroll, director of dermatologic surgery at Eastern Virginia Medical School, Norfolk. “This finding has been reinforced in the Mohs literature,” he added, citing a study that found the combination of acetaminophen and ibuprofen was more effective than acetaminophen alone or acetaminophen with codeine in controlling pain after Mohs surgery and reconstruction (Dermatol Surg. 2011 Jul;37[7]:1007-13).
Alternately layering the analgesics allows both to build to a maximum concentration in the blood without any nadirs where pain can get a foothold, an important concept in pain management, Dr. Carroll said.
And having two medications on board allows simultaneous targeting of different portions of the pain signaling pathway, he added. Ibuprofen works at the points of transduction and transmission, while acetaminophen works at the points of transmission and perception.
Dr. Carroll’s regimen starts at the time of surgery, when patients receive 400 mg ibuprofen. Three hours later, they receive 1 gram of acetaminophen; this dose should be adjusted for patients older than 60 years, who should not get more than 3 grams in 24 hours, and for those with liver failure, who should be limited to 2 grams over 24 hours.
This alternating dose is repeated every 3 hours. By the time of discharge, most patients have had at least two doses. This schedule is usually sufficient for patients at moderate to high risk of uncontrolled pain, who can then manage their discomfort with either drug the next day, he said.
Patients at higher risk of uncontrolled pain can use the regimen for the first day, and then titrate off according to their comfort. Some of these patients, however, may benefit from oxycodone, with the addition of laxative and an antiemetic, he noted.
The layering technique provides consistent postoperative pain relief that’s effective for most patients – even those who undergo substantial reconstruction, Dr. Carroll said in an interview. “This schedule is sufficient for all of our procedures, including larger reconstructions such as forehead flaps and cervicofacial rotation flaps. But additional interventions are indicated for patients with a high risk of uncontrolled pain. It’s the patient, not the procedure, which determines need for escalation.”
Teasing out those patients who may need more assertive pain management should be done in a preoperative assessment, Dr. Carroll said. A patient’s expectations of pain and history of chronic pain are some of the biggest factors in predicting a patient who will have uncontrolled pain.
“The experience of pain in Mohs surgery has limited studies,” he said. “Only a handful of investigations have looked at predictors that could help us plan. But of these, two things do stand out: a patient’s expectation of pain and a patient’s history of chronic pain.”
Surprisingly, he said, studies have determined that even a modestly elevated expectation of pain is enough to tip patients into a high-risk category. “If a patient predicted that his pain would be a 4 on a 1-10 scale, that was correlated with a lack of pain control during the operative experience. Maybe we’d expect this correlation if the expectation was an 8 or a 10, but a 4 was surprising. If a patient has even that amount of concern, I start thinking about additional interventions I can provide to maximize comfort.”
A patient’s past experience with pain is also a very large factor in how that person will experience postoperative pain. “Chronic pain does correlate with uncontrolled pain during surgery. I always ask about it. And this talk also helps drive your conversation about what you will be doing to keep them comfortable.”
That chat should include an explanation of how chronic and acute pain differ, Dr. Carroll said. “Chronic and acute pain involve different pathways and need different interventions. If the patient expresses fear, saying something like, ‘Tylenol is like water to me,’ believe him. It is like water for chronic pain. But you can also tell that patient that chronic pain is different from acute pain, and that acetaminophen will be a part of successfully managing it.”
Dr. Carroll had no financial disclosures.
EXPERT ANALYSIS FROM THE ACMS ANNUAL MEETING
ORLANDO – An alternating schedule of ibuprofen and acetaminophen every 3 hours is an excellent method of managing postoperative pain associated with Mohs surgery, especially if the initial dose is taken at the start of the procedure.
By starting with ibuprofen, the regimen capitalizes on the drug’s anti-inflammatory component to reduce overall postoperative analgesic requirement, Dr. Bryan Carroll said at the annual meeting of the American College of Mohs Surgery.
“This combination has even been shown to be superior to narcotics, both alone and in combination,” said Dr. Carroll, director of dermatologic surgery at Eastern Virginia Medical School, Norfolk. “This finding has been reinforced in the Mohs literature,” he added, citing a study that found the combination of acetaminophen and ibuprofen was more effective than acetaminophen alone or acetaminophen with codeine in controlling pain after Mohs surgery and reconstruction (Dermatol Surg. 2011 Jul;37[7]:1007-13).
Alternately layering the analgesics allows both to build to a maximum concentration in the blood without any nadirs where pain can get a foothold, an important concept in pain management, Dr. Carroll said.
And having two medications on board allows simultaneous targeting of different portions of the pain signaling pathway, he added. Ibuprofen works at the points of transduction and transmission, while acetaminophen works at the points of transmission and perception.
Dr. Carroll’s regimen starts at the time of surgery, when patients receive 400 mg ibuprofen. Three hours later, they receive 1 gram of acetaminophen; this dose should be adjusted for patients older than 60 years, who should not get more than 3 grams in 24 hours, and for those with liver failure, who should be limited to 2 grams over 24 hours.
This alternating dose is repeated every 3 hours. By the time of discharge, most patients have had at least two doses. This schedule is usually sufficient for patients at moderate to high risk of uncontrolled pain, who can then manage their discomfort with either drug the next day, he said.
Patients at higher risk of uncontrolled pain can use the regimen for the first day, and then titrate off according to their comfort. Some of these patients, however, may benefit from oxycodone, with the addition of laxative and an antiemetic, he noted.
The layering technique provides consistent postoperative pain relief that’s effective for most patients – even those who undergo substantial reconstruction, Dr. Carroll said in an interview. “This schedule is sufficient for all of our procedures, including larger reconstructions such as forehead flaps and cervicofacial rotation flaps. But additional interventions are indicated for patients with a high risk of uncontrolled pain. It’s the patient, not the procedure, which determines need for escalation.”
Teasing out those patients who may need more assertive pain management should be done in a preoperative assessment, Dr. Carroll said. A patient’s expectations of pain and history of chronic pain are some of the biggest factors in predicting a patient who will have uncontrolled pain.
“The experience of pain in Mohs surgery has limited studies,” he said. “Only a handful of investigations have looked at predictors that could help us plan. But of these, two things do stand out: a patient’s expectation of pain and a patient’s history of chronic pain.”
Surprisingly, he said, studies have determined that even a modestly elevated expectation of pain is enough to tip patients into a high-risk category. “If a patient predicted that his pain would be a 4 on a 1-10 scale, that was correlated with a lack of pain control during the operative experience. Maybe we’d expect this correlation if the expectation was an 8 or a 10, but a 4 was surprising. If a patient has even that amount of concern, I start thinking about additional interventions I can provide to maximize comfort.”
A patient’s past experience with pain is also a very large factor in how that person will experience postoperative pain. “Chronic pain does correlate with uncontrolled pain during surgery. I always ask about it. And this talk also helps drive your conversation about what you will be doing to keep them comfortable.”
That chat should include an explanation of how chronic and acute pain differ, Dr. Carroll said. “Chronic and acute pain involve different pathways and need different interventions. If the patient expresses fear, saying something like, ‘Tylenol is like water to me,’ believe him. It is like water for chronic pain. But you can also tell that patient that chronic pain is different from acute pain, and that acetaminophen will be a part of successfully managing it.”
Dr. Carroll had no financial disclosures.
Sentinel node biopsies may be useful in head and neck squamous cell carcinoma
ORLANDO – Sentinel node biopsies may be a useful staging tool for patients with cutaneous squamous cell carcinomas of the head and neck.
These patients – especially those with compromised immune systems – appear to be at sufficiently high risk of metastasis to justify the procedure, Dr. Jonathan Lopez said at the annual meeting of the American College of Mohs Surgery.
“We found that sentinel lymph node biopsy in our clinic had a 91% negative predictive value for local recurrence, nodal recurrence, and disease-specific death. It provides valuable prognostic information for patients at increased risk of nodal metastasis,” said Dr. Lopez, a dermatology resident at the Mayo Clinic, Rochester, Minn.
He and his associates conducted a chart review of 24 patients treated at the Mayo Clinic from 2000 to 2014 for a cutaneous squamous cell carcinoma (SCC) of the head or neck. Of these, 11 patients were immunosuppressed. Five had undergone a kidney transplant and three a lung transplant. One patient had Hodgkin’s lymphoma, one had cutaneous lymphocytic leukemia, and one, metastatic urothelial carcinoma.
Before sentinel node biopsy, eight patients had a wide local excision; 12 were treated with Mohs micrographic surgery only; and four had a Mohs procedure followed by resection for better margins.
The biopsies identified two patients with nodal disease, but failed to identify a third who had it, Dr. Lopez said.
Patient No. 1 had a primary SCC on the nasal tip that was stage 2, according to the American Joint Committee on Cancer (AJCC) staging system, and 2b according to the Brigham and Women’s Hospital (BWH) system. He had undergone a prior double lung transplant and his lymph node dissection showed no nodal metastasis. He declined radiotherapy and died within 2 months of the biopsy, of unclear causes that were not related to his skin cancer.
Patient No. 2 had a primary lesion on the right cheek, and a history of kidney transplant. His cancer was stage 2 by the AJCC system and 2b by the BWH system. His lymph node dissection of the right parotid and neck was negative. At last follow-up of 3.5 years, he was cancer free. However, Dr. Lopez noted, the patient died at 4 years’ follow-up of unknown causes.
The final patient had a primary lesion on the right conchal bowl. It was a stage 2 cancer by the AJCC system and 2a by the BWH system. His sentinel node biopsy was negative. However, the otolaryngologist who performed the biopsy also took seven superficial parotid nodes and one of those was positive. This patient had no recurrence at the last visit, 1.5 years after the biopsy.
The sentinel node biopsies were negative in the 21 other patients. Of these, 14 had no evidence of recurrence at a mean of 3 years’ follow-up after the sentinel lymph node biopsy. Two developed local recurrence and two others, both of whom had a history of multiple squamous cell carcinomas, developed nodal spread and died of metastatic disease. Three have died of causes unrelated to their cancer.
Dr. Lopez had no financial disclosures.
ORLANDO – Sentinel node biopsies may be a useful staging tool for patients with cutaneous squamous cell carcinomas of the head and neck.
These patients – especially those with compromised immune systems – appear to be at sufficiently high risk of metastasis to justify the procedure, Dr. Jonathan Lopez said at the annual meeting of the American College of Mohs Surgery.
“We found that sentinel lymph node biopsy in our clinic had a 91% negative predictive value for local recurrence, nodal recurrence, and disease-specific death. It provides valuable prognostic information for patients at increased risk of nodal metastasis,” said Dr. Lopez, a dermatology resident at the Mayo Clinic, Rochester, Minn.
He and his associates conducted a chart review of 24 patients treated at the Mayo Clinic from 2000 to 2014 for a cutaneous squamous cell carcinoma (SCC) of the head or neck. Of these, 11 patients were immunosuppressed. Five had undergone a kidney transplant and three a lung transplant. One patient had Hodgkin’s lymphoma, one had cutaneous lymphocytic leukemia, and one, metastatic urothelial carcinoma.
Before sentinel node biopsy, eight patients had a wide local excision; 12 were treated with Mohs micrographic surgery only; and four had a Mohs procedure followed by resection for better margins.
The biopsies identified two patients with nodal disease, but failed to identify a third who had it, Dr. Lopez said.
Patient No. 1 had a primary SCC on the nasal tip that was stage 2, according to the American Joint Committee on Cancer (AJCC) staging system, and 2b according to the Brigham and Women’s Hospital (BWH) system. He had undergone a prior double lung transplant and his lymph node dissection showed no nodal metastasis. He declined radiotherapy and died within 2 months of the biopsy, of unclear causes that were not related to his skin cancer.
Patient No. 2 had a primary lesion on the right cheek, and a history of kidney transplant. His cancer was stage 2 by the AJCC system and 2b by the BWH system. His lymph node dissection of the right parotid and neck was negative. At last follow-up of 3.5 years, he was cancer free. However, Dr. Lopez noted, the patient died at 4 years’ follow-up of unknown causes.
The final patient had a primary lesion on the right conchal bowl. It was a stage 2 cancer by the AJCC system and 2a by the BWH system. His sentinel node biopsy was negative. However, the otolaryngologist who performed the biopsy also took seven superficial parotid nodes and one of those was positive. This patient had no recurrence at the last visit, 1.5 years after the biopsy.
The sentinel node biopsies were negative in the 21 other patients. Of these, 14 had no evidence of recurrence at a mean of 3 years’ follow-up after the sentinel lymph node biopsy. Two developed local recurrence and two others, both of whom had a history of multiple squamous cell carcinomas, developed nodal spread and died of metastatic disease. Three have died of causes unrelated to their cancer.
Dr. Lopez had no financial disclosures.
ORLANDO – Sentinel node biopsies may be a useful staging tool for patients with cutaneous squamous cell carcinomas of the head and neck.
These patients – especially those with compromised immune systems – appear to be at sufficiently high risk of metastasis to justify the procedure, Dr. Jonathan Lopez said at the annual meeting of the American College of Mohs Surgery.
“We found that sentinel lymph node biopsy in our clinic had a 91% negative predictive value for local recurrence, nodal recurrence, and disease-specific death. It provides valuable prognostic information for patients at increased risk of nodal metastasis,” said Dr. Lopez, a dermatology resident at the Mayo Clinic, Rochester, Minn.
He and his associates conducted a chart review of 24 patients treated at the Mayo Clinic from 2000 to 2014 for a cutaneous squamous cell carcinoma (SCC) of the head or neck. Of these, 11 patients were immunosuppressed. Five had undergone a kidney transplant and three a lung transplant. One patient had Hodgkin’s lymphoma, one had cutaneous lymphocytic leukemia, and one, metastatic urothelial carcinoma.
Before sentinel node biopsy, eight patients had a wide local excision; 12 were treated with Mohs micrographic surgery only; and four had a Mohs procedure followed by resection for better margins.
The biopsies identified two patients with nodal disease, but failed to identify a third who had it, Dr. Lopez said.
Patient No. 1 had a primary SCC on the nasal tip that was stage 2, according to the American Joint Committee on Cancer (AJCC) staging system, and 2b according to the Brigham and Women’s Hospital (BWH) system. He had undergone a prior double lung transplant and his lymph node dissection showed no nodal metastasis. He declined radiotherapy and died within 2 months of the biopsy, of unclear causes that were not related to his skin cancer.
Patient No. 2 had a primary lesion on the right cheek, and a history of kidney transplant. His cancer was stage 2 by the AJCC system and 2b by the BWH system. His lymph node dissection of the right parotid and neck was negative. At last follow-up of 3.5 years, he was cancer free. However, Dr. Lopez noted, the patient died at 4 years’ follow-up of unknown causes.
The final patient had a primary lesion on the right conchal bowl. It was a stage 2 cancer by the AJCC system and 2a by the BWH system. His sentinel node biopsy was negative. However, the otolaryngologist who performed the biopsy also took seven superficial parotid nodes and one of those was positive. This patient had no recurrence at the last visit, 1.5 years after the biopsy.
The sentinel node biopsies were negative in the 21 other patients. Of these, 14 had no evidence of recurrence at a mean of 3 years’ follow-up after the sentinel lymph node biopsy. Two developed local recurrence and two others, both of whom had a history of multiple squamous cell carcinomas, developed nodal spread and died of metastatic disease. Three have died of causes unrelated to their cancer.
Dr. Lopez had no financial disclosures.
AT THE ACMS ANNUAL MEETING
Key clinical point: Sentinel node biopsies identified nodal spread in some patients with cutaneous SCC of the head and neck
Major finding: The procedure had a 91% negative predictive value for nodal spread and disease-specific death.
Data source: The retrospective chart review comprised of 24 patients, treated at the Mayo Clinic for cutaneous SCC of the head and neck from 2000 to 2014.
Disclosures: Dr. Lopez had no financial disclosures.
Matrilin-2 protein distinguished BCCs from benign tumors in study
ORLANDO – Matrilin-2 – a matrix protein found in peritumoral stroma – reliably distinguished invasive basal cell carcinoma from the often difficult-to-distinguish basaloid follicular hamartoma (BFH), in a study that evaluated the protein as a marker in this setting.
The protein marked 41 of 42 cancers and none of the hamartomas, Dr. Renato Goreshi reported at the annual meeting of the American College of Mohs Surgery. The one cancer it failed to identify was a superficial basal cell tumor – a finding that makes sense, since dermal fibroblasts appear to secrete matrilin-2 as a response to invasive skin tumors, said Dr. Goreshi of the Roger Williams Cancer Center, Providence, R.I.
Mohs surgery typically employs hematoxylin and eosin staining to delineate tumor boundary. But, Dr. Goreshi said, that stain doesn’t always reliably differentiate adnexal tumors from basal cell carcinomas. “Basaloid follicular hamartoma can be particularly difficult to distinguish from basal cell carcinoma,” he said.
BFH typically presents as individual or linearly arranged, small skin-colored to brown papules or plaques, or as multiple lesions in a generalized distribution on the face, scalp, and occasionally, the trunk (Arch Pathol Lab Med. 2010 Aug;134[8]:1215-9). These are often stable for many years. The differential diagnosis includes basal cell carcinoma and trichoepithelioma.
BFH sometimes occurs near a BCC, although there are no data on how often this happens.
Dr. Goreshi cited a 2007 case report of a young woman that illustrates this problem. The patient presented with a basal cell carcinoma on the side of her nose. The adjacent BFH was unrecognized, however. She underwent a multiple-stage Mohs that was unnecessarily extended because tumor margins included sections of the BFH.
“The lesion was interpreted as malignancy by both the Mohs surgeon and the dermatopathologist, but was later determined to have been a hamartoma. This highlights the importance of finding an effective marker,” Dr. Goreshi said.
He and his fellowship director, Dr. Satori Iwamoto, chief of Mohs micrographic surgery at Roger Williams, looked for a reliable way to differentiate these tumors, capitalizing on the invasive nature of BCC. The peritumoral stroma plays a role in tumor growth and invasion. It involves fibroblasts, inflammatory and endothelial cells, and extracellular matrix proteins. Matrilin-2, which is involved in the formation of filamentous networks, was a promising candidate and the initial investigations looked good, said Dr. Goreshi said.
Their confirmatory study comprised 42 BCC and seven BFH sections that were obtained during Mohs surgery. All were stained for matrilin-2 and scored for location and intensity of staining by two reviewers. The investigators also conducted flow cytometry to determine the source of the protein.
The BCC set consisted of 11 morpheaform/infiltrative BCCs, 25 nodular BCCs, and 6 superficial BCCs. With the exception of one superficial lesion, all of these stained positive for matrilin-2 in the peritumoral stroma. None of the BFH sections stained positive for the protein, however. Flow cytometry determined that the protein was coming from dermal fibroblasts in the stroma.
This is actually a key point, Dr. Goreshi noted. “Matrilin-2 is not acting as a conventional tumor marker would, but as a marker of invasion.”
This was again played out in the variation of staining intensity in the tumor subtypes. It was most intense around the infiltrative subtypes. There was also adnexal staining, but it was significantly less than what was seen in the peritumoral stroma. There was virtually no staining in or around the hamartoma.
Staining was not as intense around the superficial BCC subtypes. In fact, it was not significantly different from what was seen in the adnexal structures. Again, however, there was no staining in or around the hamartoma.
“Now we are looking at the staining patterns of other lesions, including melanoma and squamous cell carcinoma, and trying to figure out why the dermal fibroblasts are secreting matrilin-2,” Dr. Goreshi said.
The study was the winner of the 2016 Theodore Tromovitch Award, presented for original research conducted by a fellow-in-training during his or her year of training.
Neither Dr. Goreshi nor Dr. Iwamoto had any relevant financial disclosures.
ORLANDO – Matrilin-2 – a matrix protein found in peritumoral stroma – reliably distinguished invasive basal cell carcinoma from the often difficult-to-distinguish basaloid follicular hamartoma (BFH), in a study that evaluated the protein as a marker in this setting.
The protein marked 41 of 42 cancers and none of the hamartomas, Dr. Renato Goreshi reported at the annual meeting of the American College of Mohs Surgery. The one cancer it failed to identify was a superficial basal cell tumor – a finding that makes sense, since dermal fibroblasts appear to secrete matrilin-2 as a response to invasive skin tumors, said Dr. Goreshi of the Roger Williams Cancer Center, Providence, R.I.
Mohs surgery typically employs hematoxylin and eosin staining to delineate tumor boundary. But, Dr. Goreshi said, that stain doesn’t always reliably differentiate adnexal tumors from basal cell carcinomas. “Basaloid follicular hamartoma can be particularly difficult to distinguish from basal cell carcinoma,” he said.
BFH typically presents as individual or linearly arranged, small skin-colored to brown papules or plaques, or as multiple lesions in a generalized distribution on the face, scalp, and occasionally, the trunk (Arch Pathol Lab Med. 2010 Aug;134[8]:1215-9). These are often stable for many years. The differential diagnosis includes basal cell carcinoma and trichoepithelioma.
BFH sometimes occurs near a BCC, although there are no data on how often this happens.
Dr. Goreshi cited a 2007 case report of a young woman that illustrates this problem. The patient presented with a basal cell carcinoma on the side of her nose. The adjacent BFH was unrecognized, however. She underwent a multiple-stage Mohs that was unnecessarily extended because tumor margins included sections of the BFH.
“The lesion was interpreted as malignancy by both the Mohs surgeon and the dermatopathologist, but was later determined to have been a hamartoma. This highlights the importance of finding an effective marker,” Dr. Goreshi said.
He and his fellowship director, Dr. Satori Iwamoto, chief of Mohs micrographic surgery at Roger Williams, looked for a reliable way to differentiate these tumors, capitalizing on the invasive nature of BCC. The peritumoral stroma plays a role in tumor growth and invasion. It involves fibroblasts, inflammatory and endothelial cells, and extracellular matrix proteins. Matrilin-2, which is involved in the formation of filamentous networks, was a promising candidate and the initial investigations looked good, said Dr. Goreshi said.
Their confirmatory study comprised 42 BCC and seven BFH sections that were obtained during Mohs surgery. All were stained for matrilin-2 and scored for location and intensity of staining by two reviewers. The investigators also conducted flow cytometry to determine the source of the protein.
The BCC set consisted of 11 morpheaform/infiltrative BCCs, 25 nodular BCCs, and 6 superficial BCCs. With the exception of one superficial lesion, all of these stained positive for matrilin-2 in the peritumoral stroma. None of the BFH sections stained positive for the protein, however. Flow cytometry determined that the protein was coming from dermal fibroblasts in the stroma.
This is actually a key point, Dr. Goreshi noted. “Matrilin-2 is not acting as a conventional tumor marker would, but as a marker of invasion.”
This was again played out in the variation of staining intensity in the tumor subtypes. It was most intense around the infiltrative subtypes. There was also adnexal staining, but it was significantly less than what was seen in the peritumoral stroma. There was virtually no staining in or around the hamartoma.
Staining was not as intense around the superficial BCC subtypes. In fact, it was not significantly different from what was seen in the adnexal structures. Again, however, there was no staining in or around the hamartoma.
“Now we are looking at the staining patterns of other lesions, including melanoma and squamous cell carcinoma, and trying to figure out why the dermal fibroblasts are secreting matrilin-2,” Dr. Goreshi said.
The study was the winner of the 2016 Theodore Tromovitch Award, presented for original research conducted by a fellow-in-training during his or her year of training.
Neither Dr. Goreshi nor Dr. Iwamoto had any relevant financial disclosures.
ORLANDO – Matrilin-2 – a matrix protein found in peritumoral stroma – reliably distinguished invasive basal cell carcinoma from the often difficult-to-distinguish basaloid follicular hamartoma (BFH), in a study that evaluated the protein as a marker in this setting.
The protein marked 41 of 42 cancers and none of the hamartomas, Dr. Renato Goreshi reported at the annual meeting of the American College of Mohs Surgery. The one cancer it failed to identify was a superficial basal cell tumor – a finding that makes sense, since dermal fibroblasts appear to secrete matrilin-2 as a response to invasive skin tumors, said Dr. Goreshi of the Roger Williams Cancer Center, Providence, R.I.
Mohs surgery typically employs hematoxylin and eosin staining to delineate tumor boundary. But, Dr. Goreshi said, that stain doesn’t always reliably differentiate adnexal tumors from basal cell carcinomas. “Basaloid follicular hamartoma can be particularly difficult to distinguish from basal cell carcinoma,” he said.
BFH typically presents as individual or linearly arranged, small skin-colored to brown papules or plaques, or as multiple lesions in a generalized distribution on the face, scalp, and occasionally, the trunk (Arch Pathol Lab Med. 2010 Aug;134[8]:1215-9). These are often stable for many years. The differential diagnosis includes basal cell carcinoma and trichoepithelioma.
BFH sometimes occurs near a BCC, although there are no data on how often this happens.
Dr. Goreshi cited a 2007 case report of a young woman that illustrates this problem. The patient presented with a basal cell carcinoma on the side of her nose. The adjacent BFH was unrecognized, however. She underwent a multiple-stage Mohs that was unnecessarily extended because tumor margins included sections of the BFH.
“The lesion was interpreted as malignancy by both the Mohs surgeon and the dermatopathologist, but was later determined to have been a hamartoma. This highlights the importance of finding an effective marker,” Dr. Goreshi said.
He and his fellowship director, Dr. Satori Iwamoto, chief of Mohs micrographic surgery at Roger Williams, looked for a reliable way to differentiate these tumors, capitalizing on the invasive nature of BCC. The peritumoral stroma plays a role in tumor growth and invasion. It involves fibroblasts, inflammatory and endothelial cells, and extracellular matrix proteins. Matrilin-2, which is involved in the formation of filamentous networks, was a promising candidate and the initial investigations looked good, said Dr. Goreshi said.
Their confirmatory study comprised 42 BCC and seven BFH sections that were obtained during Mohs surgery. All were stained for matrilin-2 and scored for location and intensity of staining by two reviewers. The investigators also conducted flow cytometry to determine the source of the protein.
The BCC set consisted of 11 morpheaform/infiltrative BCCs, 25 nodular BCCs, and 6 superficial BCCs. With the exception of one superficial lesion, all of these stained positive for matrilin-2 in the peritumoral stroma. None of the BFH sections stained positive for the protein, however. Flow cytometry determined that the protein was coming from dermal fibroblasts in the stroma.
This is actually a key point, Dr. Goreshi noted. “Matrilin-2 is not acting as a conventional tumor marker would, but as a marker of invasion.”
This was again played out in the variation of staining intensity in the tumor subtypes. It was most intense around the infiltrative subtypes. There was also adnexal staining, but it was significantly less than what was seen in the peritumoral stroma. There was virtually no staining in or around the hamartoma.
Staining was not as intense around the superficial BCC subtypes. In fact, it was not significantly different from what was seen in the adnexal structures. Again, however, there was no staining in or around the hamartoma.
“Now we are looking at the staining patterns of other lesions, including melanoma and squamous cell carcinoma, and trying to figure out why the dermal fibroblasts are secreting matrilin-2,” Dr. Goreshi said.
The study was the winner of the 2016 Theodore Tromovitch Award, presented for original research conducted by a fellow-in-training during his or her year of training.
Neither Dr. Goreshi nor Dr. Iwamoto had any relevant financial disclosures.
AT THE ACMS ANNUAL MEETING
Key clinical point: Matrilin-2 is the first marker of tumor invasion to be used in skin cancers.
Major finding: The protein bound to 41 of 42 BCCs, and to none of the hamartoma lesions studied, reliably distinguishing the two.
Data source: 42 frozen section BCCs and seven basaloid follicular hamartomas.
Disclosures: Neither Dr. Goreshi nor Dr. Iwamoto had any relevant financial disclosures.
Early Detection of Melanoma in Men
Men do not know as much about skin cancer prevention and detection techniques as women, according to a recent survey conducted by the American Academy of Dermatology (AAD). This lack of knowledge may delay or prevent early diagnosis and treatment of melanoma and other nonmelanoma skin cancers in this patient population.
The survey results showed that only 56% of men versus 76% of women know there is no such thing as a healthy tan, and only 54% of men versus 70% of women know that getting a base tan is not a healthy way to protect skin from the sun. Furthermore, only 56% of men surveyed were aware that skin cancer could occur on areas of the skin not typically exposed to the sun compared to 65% of women.
“While our survey results indicate that men don’t know as much about skin cancer prevention and detection as women, men over 50 have a higher risk of developing melanoma, so it’s especially important for them to be vigilant about protecting and monitoring their skin,” said AAD President Abel Torres, MD, JD.
May is skin cancer awareness month and the AAD is encouraging patients to make sure their skin is “Looking Good in 2016” by using sun protection and regularly examining skin for signs of skin cancer. The campaign features a public service announcement encouraging men to check their skin for signs of skin cancer and find a partner to help. The AAD also released a new infographic with tips on performing a skin cancer self-examination that dermatologists can share with patients to promote early detection of skin cancer.
Dermatologist intervention in catching skin cancers when they are easier to treat also is key. At the 74th Annual Meeting of the American Academy of Dermatology in Washington, DC, Dr. Orit Markowitz discussed noninvasive imaging tools that can help dermatologists diagnose skin cancers earlier. She noted that even when a lesion looks very small, tools such as dermoscopy can reveal features that indicate it already has depth and therefore may progress to a more serious malignancy. Early detection is particularly crucial in cases of rare aggressive tumors such as amelanotic melanoma. “If something is very pink clinically and then suddenly has pigmentation dermoscopically, you really have to be considering biopsying that lesion because you may be looking at an early amelanotic melanoma,” Dr. Markowitz explained. By the time the lesion develops more obvious clinical features suggesting malignancy, the tumor progression may be far more advanced.
Men do not know as much about skin cancer prevention and detection techniques as women, according to a recent survey conducted by the American Academy of Dermatology (AAD). This lack of knowledge may delay or prevent early diagnosis and treatment of melanoma and other nonmelanoma skin cancers in this patient population.
The survey results showed that only 56% of men versus 76% of women know there is no such thing as a healthy tan, and only 54% of men versus 70% of women know that getting a base tan is not a healthy way to protect skin from the sun. Furthermore, only 56% of men surveyed were aware that skin cancer could occur on areas of the skin not typically exposed to the sun compared to 65% of women.
“While our survey results indicate that men don’t know as much about skin cancer prevention and detection as women, men over 50 have a higher risk of developing melanoma, so it’s especially important for them to be vigilant about protecting and monitoring their skin,” said AAD President Abel Torres, MD, JD.
May is skin cancer awareness month and the AAD is encouraging patients to make sure their skin is “Looking Good in 2016” by using sun protection and regularly examining skin for signs of skin cancer. The campaign features a public service announcement encouraging men to check their skin for signs of skin cancer and find a partner to help. The AAD also released a new infographic with tips on performing a skin cancer self-examination that dermatologists can share with patients to promote early detection of skin cancer.
Dermatologist intervention in catching skin cancers when they are easier to treat also is key. At the 74th Annual Meeting of the American Academy of Dermatology in Washington, DC, Dr. Orit Markowitz discussed noninvasive imaging tools that can help dermatologists diagnose skin cancers earlier. She noted that even when a lesion looks very small, tools such as dermoscopy can reveal features that indicate it already has depth and therefore may progress to a more serious malignancy. Early detection is particularly crucial in cases of rare aggressive tumors such as amelanotic melanoma. “If something is very pink clinically and then suddenly has pigmentation dermoscopically, you really have to be considering biopsying that lesion because you may be looking at an early amelanotic melanoma,” Dr. Markowitz explained. By the time the lesion develops more obvious clinical features suggesting malignancy, the tumor progression may be far more advanced.
Men do not know as much about skin cancer prevention and detection techniques as women, according to a recent survey conducted by the American Academy of Dermatology (AAD). This lack of knowledge may delay or prevent early diagnosis and treatment of melanoma and other nonmelanoma skin cancers in this patient population.
The survey results showed that only 56% of men versus 76% of women know there is no such thing as a healthy tan, and only 54% of men versus 70% of women know that getting a base tan is not a healthy way to protect skin from the sun. Furthermore, only 56% of men surveyed were aware that skin cancer could occur on areas of the skin not typically exposed to the sun compared to 65% of women.
“While our survey results indicate that men don’t know as much about skin cancer prevention and detection as women, men over 50 have a higher risk of developing melanoma, so it’s especially important for them to be vigilant about protecting and monitoring their skin,” said AAD President Abel Torres, MD, JD.
May is skin cancer awareness month and the AAD is encouraging patients to make sure their skin is “Looking Good in 2016” by using sun protection and regularly examining skin for signs of skin cancer. The campaign features a public service announcement encouraging men to check their skin for signs of skin cancer and find a partner to help. The AAD also released a new infographic with tips on performing a skin cancer self-examination that dermatologists can share with patients to promote early detection of skin cancer.
Dermatologist intervention in catching skin cancers when they are easier to treat also is key. At the 74th Annual Meeting of the American Academy of Dermatology in Washington, DC, Dr. Orit Markowitz discussed noninvasive imaging tools that can help dermatologists diagnose skin cancers earlier. She noted that even when a lesion looks very small, tools such as dermoscopy can reveal features that indicate it already has depth and therefore may progress to a more serious malignancy. Early detection is particularly crucial in cases of rare aggressive tumors such as amelanotic melanoma. “If something is very pink clinically and then suddenly has pigmentation dermoscopically, you really have to be considering biopsying that lesion because you may be looking at an early amelanotic melanoma,” Dr. Markowitz explained. By the time the lesion develops more obvious clinical features suggesting malignancy, the tumor progression may be far more advanced.
Management of Vitiligo Patients With Surgical Interventions
Vitiligo is a common, asymptomatic, acquired depigmentation disorder that is caused by an unknown etiology. Lesions appear as sharply demarcated, depigmented macules and patches that are scattered symmetrically or unsymmetrically over the body. The presentation can be delineated based on the segmental or nonsegmental nature of the disease. According to the revised classification/nomenclature of vitiligo,1 the disorder can be classified as nonsegmental, segmental, mixed, or unclassified. The pathogenesis of the vitiligo disease process is due to multiple modalities that contribute to melanocyte loss. Theories for melanocyte destruction include but are not limited to autoimmunity, biochemicals, epidermal cytokines, increased hydrogen peroxide and free radicals, and humoral and cellular immune alteration.2,3
Despite its long history, the most frustrating aspect of the vitiligo disease process remains its treatment due to limited efficacy, frequent application of topicals, and the need for high-potency steroids. Medical therapies usually are the first line of treatment and are most effective with few side effects for bilateral nonsegmental or evolving vitiligo.2 Some of the primary therapies with the highest efficacies appear to be calcipotriene and psoralen plus UVA, psoralen plus UVA as monotherapy, excimer laser, narrowband UVB, oral steroids, 8-methoxypsoralen, tacrolimus, and topical steroids.4 The theory is that these treatments would be successful if the patient had active melanocytes in the external root sheath that would be able to repigment a patch of vitiligo.5 Hence, it would be more difficult to treat areas such as the dorsal aspect of the fingers and toes because they lack hair-bearing areas with melanocytes.6 The alternative approach to treating vitiligo patches would be surgical intervention techniques, as they provide melanocytic cells to a previously depigmented area.3,5 The focus of this article is to evaluate the efficacy and appropriate use of some of the surgical procedures that can be used in the treatment of vitiligo patients.
Candidate Selection
First, vitiligo patients for whom first-line treatment with medical therapies has failed are candidates for surgical techniques. The second vital component is to clinically confirm the diagnosis of vitiligo as opposed to other genetic, infectious, or autoimmune causes of pigment loss. Lastly, the vitiligo patch should be stable. A stable vitiligo patch does not continue to progress and is no longer responsive to topical medications that are meant to repigment for a discernible period of time.7
Classification of Disease Stage
To classify the stage of vitiligo prior to surgical intervention, Gauthier8 created a basic grading system: grade I, with partial depletion of epidermal melanocytes in a vitiligo patch that responds to repigmentation in a follicular pattern evenly such as on the face and neck; grade II, with complete depletion of epidermal melanocytes with a usual follicular pattern of repigmentation; and grade III, indicating complete depletion of follicular melanocytes with no hope of response to medical therapy. According to Rusfianti and Wirohadidjodjo,2 the surgical techniques that have developed over the years for treatment of grade III vitiligo patients include split-thickness skin grafting, suction blister grafting, miniature punch grafting, and cultured melanocyte transplantation.
Surgical Techniques
Split-thickness skin grafting is an older procedure that entails the use of a harvesting graft site with no pigment loss and dermabrasion of the recipient area to allow interaction with the wound bed.9 With proper care and minimal movement or wrinkling of the graft site, patients can have repigmentation without skip areas.
Suction blister grafting is another tried and tested surgical intervention. Hasegawa et al10 conducted a study of 15 patients (13 males, 2 females; age range, 16–38 years) diagnosed with segmental vitiligo who were treated using the suction blister grafting technique with CO2 laser resurfacing. Patients were recruited 1 month prior to initiating therapy and no other treatments were used during the month or in conjunction with the surgical intervention. Suction blisters were harvested from the left thigh and transferred in saline to the recipient site, which was abraded with 1 pass of the short-pulse CO2 laser system. The recipient sites were then closed with 7-0 nylon sutures and covered tightly with tie-over dressings for at least 1 week. Within 6 months of the procedure, a treatment response of 100% was seen in 15 patients, making it an effective method for treatment-resistant vitiligo patients.10
Miniature punch grafting is another possible treatment option for resistant cases of vitiligo. Mapar et al11 conducted a study in 25 patients (21 women, 4 men; age range, 20–47 years) who had been diagnosed with stable vitiligo (ie, no progression in the last 2 years) and were treated with single hair follicle transplant versus miniature punch grafting. The theory behind the study was to use the melanocytic reservoir noted in the normal hair follicle to repigment the vitiligo patch. With follow-up of both methods of treatment, there was no statistical difference in treatment results.11 A similar study was conducted by Malakar and Lahiri12 in patients with lip leukoderma (a variant of vitiligo). One hundred eight patients (41 males, 67 females; age range, 14–62 years) who had been diagnosed with stable lip leukoderma (ie, stable vitiligo for at least 6 months) underwent treatment via autologous miniature punch grafting. Punch biopsies were performed in donor sites of the buttocks and upper thighs with 72% of patients noting complete repigmentation. Complications noted were herpes labialis–induced lip leukoderma, which ultimately led to rejection of the graft site.12 Overall, however, miniature punch grafting is a viable surgical option in stable vitiligo patients.
Cultured melanocyte transplantation, or a noncultured epidermal suspension, was first initiated in 1992.13 Silpa-Archa et al14 conducted an open, split-comparison study of 6 vitiligo patients (5 women, 1 man; age range, 20–65 years) with stable lesions. Fifty percent of patients received autologous pigmented skin cellular suspension, which was applied to vitiligo-affected skin that was treated with a fractionated CO2 laser, and 50% received dermabrasion. Composite dressing was placed overlying the site with dressing removal in 1 week. The degree of repigmentation was based on a modified vitiligo area scoring index scale of poor (0%–25%), fair (26%–50%), good (51%–75%), very good (76%–90%), or excellent (91%–100%). Overall repigmentation was very good to excellent in all 6 patients.14 Potentially, this method can far improve the surgical treatment options for future vitiligo patients.
Final Thoughts
Overall, when evaluating surgical interventions for the treatment of vitiligo, careful consideration of the patient’s disease progression, failed therapies, outcome expectations, and repigmentation is warranted prior to initiating any procedure. For appropriate candidates, a range of surgical methodologies has proven to be effective in treatment of stable vitiligo patients.
- Taïeb A, Picardo M; VETF members. The definition and assessment of vitiligo: a consensus report of the Vitiligo European Task Force. Pigment Cell Res. 2007;20:27-35. Cited by: Ezzedine K, Lim HW, Suzuki T, et al; Vitiligo Global Issue Consensus Conference Panelists. Revised classification/nomenclature of vitiligo and related issues: the Vitiligo Global Issues Consensus Conference. Pigment Cell Melanoma Res. 2012;25:E1-E13.
- Rusfianti M, Wirohadidjodjo YW. Dermatosurgical techniques for repigmentation of vitiligo. Int J Dermatol. 2006;45:411-417.
- Falabella R. Surgical therapies for vitiligo. Clin Dermatol. 1997;15:927-939.
- Whitton ME, Pinart M, Batchelor J, et al. Interventions for vitiligo. Cochrane Database Syst Rev. 2015;2:CD003263.
- Mulekar SV, Isedeh P. Surgical interventions for vitiligo: an evidence-based review. Br J Dermatol. 2013;169(suppl 3):57-66.
- Dutta AK, Mandal SB. A clinical study of 650 vitiligo cases and their classification. Indian J Dermatol. 1969;14:103-111.
- Falabella R, Arrunategui A, Barona MI, et al. The minigrafting test for vitiligo: detection of stable lesions for melanocyte transplantation. J Am Acad Dermatol. 1995;32:228-232.
- Gauthier Y. Le vitiligo. Gaz Med. 1994;101:8-12.
- Malakar S, Malakar RS. Surgical pearl: composite film and graft unit for the recipient area dressing after split-thickness skin grafting in vitiligo. J Am Acad Dermatol. 2001;44:856-858.
- Hasegawa T, Suga Y, Ikejima A, et al. Suction blister grafting with CO2 laser resurfacing of the graft recipient site for vitiligo. J Dermatol. 2007;34:490-492.
- Mapar MA, Safarpour M, Mapar M, et al. A comparative study of the mini-punch grafting and hair follicle transplantation in the treatment of refractory and stable vitiligo. J Am Acad Dermatol. 2014;70:743-747.
- Malakar S, Lahiri K. Punch grafting for lip leukoderma. Dermatology. 2004;208:125-128.
- Gauthier Y, Surleve-Bazeille JE. Autologous grafting with noncultured melanocytes: a simplified method for treatment of depigmented lesions. J Am Acad Dermatol. 1992;26(2, pt 1):191-194.
- Silpa-Archa N, Griffith JL, Williams MS, et al. Prospective comparison of recipient-site preparation with fractional carbon dioxide laser versus dermabrasion and recipient-site dressing composition in melanocyte-keratinocyte transplantation procedure in vitiligo: a preliminary study [published online January 24, 2016]. Br J Dermatol. 2016;174:895-897.
Vitiligo is a common, asymptomatic, acquired depigmentation disorder that is caused by an unknown etiology. Lesions appear as sharply demarcated, depigmented macules and patches that are scattered symmetrically or unsymmetrically over the body. The presentation can be delineated based on the segmental or nonsegmental nature of the disease. According to the revised classification/nomenclature of vitiligo,1 the disorder can be classified as nonsegmental, segmental, mixed, or unclassified. The pathogenesis of the vitiligo disease process is due to multiple modalities that contribute to melanocyte loss. Theories for melanocyte destruction include but are not limited to autoimmunity, biochemicals, epidermal cytokines, increased hydrogen peroxide and free radicals, and humoral and cellular immune alteration.2,3
Despite its long history, the most frustrating aspect of the vitiligo disease process remains its treatment due to limited efficacy, frequent application of topicals, and the need for high-potency steroids. Medical therapies usually are the first line of treatment and are most effective with few side effects for bilateral nonsegmental or evolving vitiligo.2 Some of the primary therapies with the highest efficacies appear to be calcipotriene and psoralen plus UVA, psoralen plus UVA as monotherapy, excimer laser, narrowband UVB, oral steroids, 8-methoxypsoralen, tacrolimus, and topical steroids.4 The theory is that these treatments would be successful if the patient had active melanocytes in the external root sheath that would be able to repigment a patch of vitiligo.5 Hence, it would be more difficult to treat areas such as the dorsal aspect of the fingers and toes because they lack hair-bearing areas with melanocytes.6 The alternative approach to treating vitiligo patches would be surgical intervention techniques, as they provide melanocytic cells to a previously depigmented area.3,5 The focus of this article is to evaluate the efficacy and appropriate use of some of the surgical procedures that can be used in the treatment of vitiligo patients.
Candidate Selection
First, vitiligo patients for whom first-line treatment with medical therapies has failed are candidates for surgical techniques. The second vital component is to clinically confirm the diagnosis of vitiligo as opposed to other genetic, infectious, or autoimmune causes of pigment loss. Lastly, the vitiligo patch should be stable. A stable vitiligo patch does not continue to progress and is no longer responsive to topical medications that are meant to repigment for a discernible period of time.7
Classification of Disease Stage
To classify the stage of vitiligo prior to surgical intervention, Gauthier8 created a basic grading system: grade I, with partial depletion of epidermal melanocytes in a vitiligo patch that responds to repigmentation in a follicular pattern evenly such as on the face and neck; grade II, with complete depletion of epidermal melanocytes with a usual follicular pattern of repigmentation; and grade III, indicating complete depletion of follicular melanocytes with no hope of response to medical therapy. According to Rusfianti and Wirohadidjodjo,2 the surgical techniques that have developed over the years for treatment of grade III vitiligo patients include split-thickness skin grafting, suction blister grafting, miniature punch grafting, and cultured melanocyte transplantation.
Surgical Techniques
Split-thickness skin grafting is an older procedure that entails the use of a harvesting graft site with no pigment loss and dermabrasion of the recipient area to allow interaction with the wound bed.9 With proper care and minimal movement or wrinkling of the graft site, patients can have repigmentation without skip areas.
Suction blister grafting is another tried and tested surgical intervention. Hasegawa et al10 conducted a study of 15 patients (13 males, 2 females; age range, 16–38 years) diagnosed with segmental vitiligo who were treated using the suction blister grafting technique with CO2 laser resurfacing. Patients were recruited 1 month prior to initiating therapy and no other treatments were used during the month or in conjunction with the surgical intervention. Suction blisters were harvested from the left thigh and transferred in saline to the recipient site, which was abraded with 1 pass of the short-pulse CO2 laser system. The recipient sites were then closed with 7-0 nylon sutures and covered tightly with tie-over dressings for at least 1 week. Within 6 months of the procedure, a treatment response of 100% was seen in 15 patients, making it an effective method for treatment-resistant vitiligo patients.10
Miniature punch grafting is another possible treatment option for resistant cases of vitiligo. Mapar et al11 conducted a study in 25 patients (21 women, 4 men; age range, 20–47 years) who had been diagnosed with stable vitiligo (ie, no progression in the last 2 years) and were treated with single hair follicle transplant versus miniature punch grafting. The theory behind the study was to use the melanocytic reservoir noted in the normal hair follicle to repigment the vitiligo patch. With follow-up of both methods of treatment, there was no statistical difference in treatment results.11 A similar study was conducted by Malakar and Lahiri12 in patients with lip leukoderma (a variant of vitiligo). One hundred eight patients (41 males, 67 females; age range, 14–62 years) who had been diagnosed with stable lip leukoderma (ie, stable vitiligo for at least 6 months) underwent treatment via autologous miniature punch grafting. Punch biopsies were performed in donor sites of the buttocks and upper thighs with 72% of patients noting complete repigmentation. Complications noted were herpes labialis–induced lip leukoderma, which ultimately led to rejection of the graft site.12 Overall, however, miniature punch grafting is a viable surgical option in stable vitiligo patients.
Cultured melanocyte transplantation, or a noncultured epidermal suspension, was first initiated in 1992.13 Silpa-Archa et al14 conducted an open, split-comparison study of 6 vitiligo patients (5 women, 1 man; age range, 20–65 years) with stable lesions. Fifty percent of patients received autologous pigmented skin cellular suspension, which was applied to vitiligo-affected skin that was treated with a fractionated CO2 laser, and 50% received dermabrasion. Composite dressing was placed overlying the site with dressing removal in 1 week. The degree of repigmentation was based on a modified vitiligo area scoring index scale of poor (0%–25%), fair (26%–50%), good (51%–75%), very good (76%–90%), or excellent (91%–100%). Overall repigmentation was very good to excellent in all 6 patients.14 Potentially, this method can far improve the surgical treatment options for future vitiligo patients.
Final Thoughts
Overall, when evaluating surgical interventions for the treatment of vitiligo, careful consideration of the patient’s disease progression, failed therapies, outcome expectations, and repigmentation is warranted prior to initiating any procedure. For appropriate candidates, a range of surgical methodologies has proven to be effective in treatment of stable vitiligo patients.
Vitiligo is a common, asymptomatic, acquired depigmentation disorder that is caused by an unknown etiology. Lesions appear as sharply demarcated, depigmented macules and patches that are scattered symmetrically or unsymmetrically over the body. The presentation can be delineated based on the segmental or nonsegmental nature of the disease. According to the revised classification/nomenclature of vitiligo,1 the disorder can be classified as nonsegmental, segmental, mixed, or unclassified. The pathogenesis of the vitiligo disease process is due to multiple modalities that contribute to melanocyte loss. Theories for melanocyte destruction include but are not limited to autoimmunity, biochemicals, epidermal cytokines, increased hydrogen peroxide and free radicals, and humoral and cellular immune alteration.2,3
Despite its long history, the most frustrating aspect of the vitiligo disease process remains its treatment due to limited efficacy, frequent application of topicals, and the need for high-potency steroids. Medical therapies usually are the first line of treatment and are most effective with few side effects for bilateral nonsegmental or evolving vitiligo.2 Some of the primary therapies with the highest efficacies appear to be calcipotriene and psoralen plus UVA, psoralen plus UVA as monotherapy, excimer laser, narrowband UVB, oral steroids, 8-methoxypsoralen, tacrolimus, and topical steroids.4 The theory is that these treatments would be successful if the patient had active melanocytes in the external root sheath that would be able to repigment a patch of vitiligo.5 Hence, it would be more difficult to treat areas such as the dorsal aspect of the fingers and toes because they lack hair-bearing areas with melanocytes.6 The alternative approach to treating vitiligo patches would be surgical intervention techniques, as they provide melanocytic cells to a previously depigmented area.3,5 The focus of this article is to evaluate the efficacy and appropriate use of some of the surgical procedures that can be used in the treatment of vitiligo patients.
Candidate Selection
First, vitiligo patients for whom first-line treatment with medical therapies has failed are candidates for surgical techniques. The second vital component is to clinically confirm the diagnosis of vitiligo as opposed to other genetic, infectious, or autoimmune causes of pigment loss. Lastly, the vitiligo patch should be stable. A stable vitiligo patch does not continue to progress and is no longer responsive to topical medications that are meant to repigment for a discernible period of time.7
Classification of Disease Stage
To classify the stage of vitiligo prior to surgical intervention, Gauthier8 created a basic grading system: grade I, with partial depletion of epidermal melanocytes in a vitiligo patch that responds to repigmentation in a follicular pattern evenly such as on the face and neck; grade II, with complete depletion of epidermal melanocytes with a usual follicular pattern of repigmentation; and grade III, indicating complete depletion of follicular melanocytes with no hope of response to medical therapy. According to Rusfianti and Wirohadidjodjo,2 the surgical techniques that have developed over the years for treatment of grade III vitiligo patients include split-thickness skin grafting, suction blister grafting, miniature punch grafting, and cultured melanocyte transplantation.
Surgical Techniques
Split-thickness skin grafting is an older procedure that entails the use of a harvesting graft site with no pigment loss and dermabrasion of the recipient area to allow interaction with the wound bed.9 With proper care and minimal movement or wrinkling of the graft site, patients can have repigmentation without skip areas.
Suction blister grafting is another tried and tested surgical intervention. Hasegawa et al10 conducted a study of 15 patients (13 males, 2 females; age range, 16–38 years) diagnosed with segmental vitiligo who were treated using the suction blister grafting technique with CO2 laser resurfacing. Patients were recruited 1 month prior to initiating therapy and no other treatments were used during the month or in conjunction with the surgical intervention. Suction blisters were harvested from the left thigh and transferred in saline to the recipient site, which was abraded with 1 pass of the short-pulse CO2 laser system. The recipient sites were then closed with 7-0 nylon sutures and covered tightly with tie-over dressings for at least 1 week. Within 6 months of the procedure, a treatment response of 100% was seen in 15 patients, making it an effective method for treatment-resistant vitiligo patients.10
Miniature punch grafting is another possible treatment option for resistant cases of vitiligo. Mapar et al11 conducted a study in 25 patients (21 women, 4 men; age range, 20–47 years) who had been diagnosed with stable vitiligo (ie, no progression in the last 2 years) and were treated with single hair follicle transplant versus miniature punch grafting. The theory behind the study was to use the melanocytic reservoir noted in the normal hair follicle to repigment the vitiligo patch. With follow-up of both methods of treatment, there was no statistical difference in treatment results.11 A similar study was conducted by Malakar and Lahiri12 in patients with lip leukoderma (a variant of vitiligo). One hundred eight patients (41 males, 67 females; age range, 14–62 years) who had been diagnosed with stable lip leukoderma (ie, stable vitiligo for at least 6 months) underwent treatment via autologous miniature punch grafting. Punch biopsies were performed in donor sites of the buttocks and upper thighs with 72% of patients noting complete repigmentation. Complications noted were herpes labialis–induced lip leukoderma, which ultimately led to rejection of the graft site.12 Overall, however, miniature punch grafting is a viable surgical option in stable vitiligo patients.
Cultured melanocyte transplantation, or a noncultured epidermal suspension, was first initiated in 1992.13 Silpa-Archa et al14 conducted an open, split-comparison study of 6 vitiligo patients (5 women, 1 man; age range, 20–65 years) with stable lesions. Fifty percent of patients received autologous pigmented skin cellular suspension, which was applied to vitiligo-affected skin that was treated with a fractionated CO2 laser, and 50% received dermabrasion. Composite dressing was placed overlying the site with dressing removal in 1 week. The degree of repigmentation was based on a modified vitiligo area scoring index scale of poor (0%–25%), fair (26%–50%), good (51%–75%), very good (76%–90%), or excellent (91%–100%). Overall repigmentation was very good to excellent in all 6 patients.14 Potentially, this method can far improve the surgical treatment options for future vitiligo patients.
Final Thoughts
Overall, when evaluating surgical interventions for the treatment of vitiligo, careful consideration of the patient’s disease progression, failed therapies, outcome expectations, and repigmentation is warranted prior to initiating any procedure. For appropriate candidates, a range of surgical methodologies has proven to be effective in treatment of stable vitiligo patients.
- Taïeb A, Picardo M; VETF members. The definition and assessment of vitiligo: a consensus report of the Vitiligo European Task Force. Pigment Cell Res. 2007;20:27-35. Cited by: Ezzedine K, Lim HW, Suzuki T, et al; Vitiligo Global Issue Consensus Conference Panelists. Revised classification/nomenclature of vitiligo and related issues: the Vitiligo Global Issues Consensus Conference. Pigment Cell Melanoma Res. 2012;25:E1-E13.
- Rusfianti M, Wirohadidjodjo YW. Dermatosurgical techniques for repigmentation of vitiligo. Int J Dermatol. 2006;45:411-417.
- Falabella R. Surgical therapies for vitiligo. Clin Dermatol. 1997;15:927-939.
- Whitton ME, Pinart M, Batchelor J, et al. Interventions for vitiligo. Cochrane Database Syst Rev. 2015;2:CD003263.
- Mulekar SV, Isedeh P. Surgical interventions for vitiligo: an evidence-based review. Br J Dermatol. 2013;169(suppl 3):57-66.
- Dutta AK, Mandal SB. A clinical study of 650 vitiligo cases and their classification. Indian J Dermatol. 1969;14:103-111.
- Falabella R, Arrunategui A, Barona MI, et al. The minigrafting test for vitiligo: detection of stable lesions for melanocyte transplantation. J Am Acad Dermatol. 1995;32:228-232.
- Gauthier Y. Le vitiligo. Gaz Med. 1994;101:8-12.
- Malakar S, Malakar RS. Surgical pearl: composite film and graft unit for the recipient area dressing after split-thickness skin grafting in vitiligo. J Am Acad Dermatol. 2001;44:856-858.
- Hasegawa T, Suga Y, Ikejima A, et al. Suction blister grafting with CO2 laser resurfacing of the graft recipient site for vitiligo. J Dermatol. 2007;34:490-492.
- Mapar MA, Safarpour M, Mapar M, et al. A comparative study of the mini-punch grafting and hair follicle transplantation in the treatment of refractory and stable vitiligo. J Am Acad Dermatol. 2014;70:743-747.
- Malakar S, Lahiri K. Punch grafting for lip leukoderma. Dermatology. 2004;208:125-128.
- Gauthier Y, Surleve-Bazeille JE. Autologous grafting with noncultured melanocytes: a simplified method for treatment of depigmented lesions. J Am Acad Dermatol. 1992;26(2, pt 1):191-194.
- Silpa-Archa N, Griffith JL, Williams MS, et al. Prospective comparison of recipient-site preparation with fractional carbon dioxide laser versus dermabrasion and recipient-site dressing composition in melanocyte-keratinocyte transplantation procedure in vitiligo: a preliminary study [published online January 24, 2016]. Br J Dermatol. 2016;174:895-897.
- Taïeb A, Picardo M; VETF members. The definition and assessment of vitiligo: a consensus report of the Vitiligo European Task Force. Pigment Cell Res. 2007;20:27-35. Cited by: Ezzedine K, Lim HW, Suzuki T, et al; Vitiligo Global Issue Consensus Conference Panelists. Revised classification/nomenclature of vitiligo and related issues: the Vitiligo Global Issues Consensus Conference. Pigment Cell Melanoma Res. 2012;25:E1-E13.
- Rusfianti M, Wirohadidjodjo YW. Dermatosurgical techniques for repigmentation of vitiligo. Int J Dermatol. 2006;45:411-417.
- Falabella R. Surgical therapies for vitiligo. Clin Dermatol. 1997;15:927-939.
- Whitton ME, Pinart M, Batchelor J, et al. Interventions for vitiligo. Cochrane Database Syst Rev. 2015;2:CD003263.
- Mulekar SV, Isedeh P. Surgical interventions for vitiligo: an evidence-based review. Br J Dermatol. 2013;169(suppl 3):57-66.
- Dutta AK, Mandal SB. A clinical study of 650 vitiligo cases and their classification. Indian J Dermatol. 1969;14:103-111.
- Falabella R, Arrunategui A, Barona MI, et al. The minigrafting test for vitiligo: detection of stable lesions for melanocyte transplantation. J Am Acad Dermatol. 1995;32:228-232.
- Gauthier Y. Le vitiligo. Gaz Med. 1994;101:8-12.
- Malakar S, Malakar RS. Surgical pearl: composite film and graft unit for the recipient area dressing after split-thickness skin grafting in vitiligo. J Am Acad Dermatol. 2001;44:856-858.
- Hasegawa T, Suga Y, Ikejima A, et al. Suction blister grafting with CO2 laser resurfacing of the graft recipient site for vitiligo. J Dermatol. 2007;34:490-492.
- Mapar MA, Safarpour M, Mapar M, et al. A comparative study of the mini-punch grafting and hair follicle transplantation in the treatment of refractory and stable vitiligo. J Am Acad Dermatol. 2014;70:743-747.
- Malakar S, Lahiri K. Punch grafting for lip leukoderma. Dermatology. 2004;208:125-128.
- Gauthier Y, Surleve-Bazeille JE. Autologous grafting with noncultured melanocytes: a simplified method for treatment of depigmented lesions. J Am Acad Dermatol. 1992;26(2, pt 1):191-194.
- Silpa-Archa N, Griffith JL, Williams MS, et al. Prospective comparison of recipient-site preparation with fractional carbon dioxide laser versus dermabrasion and recipient-site dressing composition in melanocyte-keratinocyte transplantation procedure in vitiligo: a preliminary study [published online January 24, 2016]. Br J Dermatol. 2016;174:895-897.
Aesthetic Dermatology: Effects of climate change on skin
Global climate appears to be changing at an unprecedented rate. Climate change can be caused by many factors, including variations in solar radiation received by the earth, oceanic circulation, plate tectonics, as well as human-induced alterations of the natural world. Many human activities, such as the use of fossil fuel and the consequent accumulation of greenhouse gases in the atmosphere, land consumption, deforestation, industrial processes, as well as some agriculture practices, are contributing to global climate change. Many have reported on the current trend toward global warming (average surface temperature has augmented by 0.6°C over the past 100 years), decreased precipitation, atmospheric humidity changes, and the rise in global extreme climatic events. The magnitude and cause of these changes and their impact on human activity have become important matters of debate worldwide, representing climate change as one of the greatest challenges of the modern age.
Although many articles have been written based on observations and various predictive models of how climate change could affect social, economic, and health systems, only a few studies exist about the effects of this change on skin and skin disease. However, the skin is the most highly exposed organ to the environment; therefore, cutaneous conditions are inclined to respond to changes in climate.
Skin cancer
The World Health Organization predicts that the depletion of the ozone layer could lead to further increased rates of melanoma and nonmelanoma skin cancer. In humans, it has been speculated that a long-term rise of temperature by 2°C could increase the carcinogenic effectiveness of solar UV by 10%.
Strictly speaking, stratospheric ozone depletion is not part of “global climate change,” which occurs in the troposphere. There are, however, several recently described interactions between ozone depletion and greenhouse gas–induced warming. Stratospheric ozone absorbs much of the incoming solar ultraviolet radiation, especially the biologically more damaging, shorter-wavelength UVB wavelengths. We now know that various industrial halogenated chemicals such as the chlorofluorocarbons or CFCs (used in refrigeration, insulation, and spray-can propellants) and methyl bromide, while inert at ambient Earth-surface temperatures, react with ozone in the extremely cold polar stratosphere. This destruction of ozone occurs especially in late winter and early spring.
During the 1980s and 1990s at northern midlatitudes (such as Europe), the average year-round ozone concentration declined by around 4% per decade; over the southern regions of Australia, New Zealand, Argentina, and South Africa, the figure approximated 6%-7%. UV exposures at northern midlatitudes are likely to peak around 2020, with an estimated 10% increase in effective ultraviolet radiation relative to 1980s levels.
The modeling of future ozone levels and UV radiation (UVR) exposures has estimated that, in consequence, a ‘European’ population living at around 45 degrees North will experience, by 2050, an approximate 5% excess of total skin cancer incidence (assuming, conservatively, no change in age distribution). The equivalent estimation for the U.S. population is for a 10% increase in skin cancer incidence by around 2050.
In the mid-1980s, governments recognized the emerging hazard from ozone depletion. The Montreal Protocol of 1987 was adopted, and the phasing out of major ozone-destroying gases began. Some anticipate a slow but near-complete recovery of stratospheric ozone by the middle of the twenty-first century; the Environmental Protection Agency (EPA) estimates recovery by 2065 with strict adherence to protection protocols.
Increased exposure to UVR also leads to increased rates of lens opacification, cataracts, and whole-body immunosuppression. UVR-induced immunosuppression could influence patterns of infectious disease. It may also influence the occurrence and progression of various autoimmune diseases and, less certainly, vaccine efficacy.
Extreme weather events
The International Society of Dermatology Task Force on Climate Change reports that weather phenomena such as El Niño also result in changes to dermatologic conditions. The El Niño Southern Oscillation (ENSO) is a complex climate phenomenon occurring in the Pacific Ocean at intervals of 2-7 years. The term refers to fluctuations in ocean temperatures in the tropical eastern Pacific Ocean (El Niño, the warm phase of ENSO, and La Niña, the cool phase of ENSO) and in atmospheric pressure across the Pacific basin (Southern Oscillation). This weather pattern is attributed with causing climate change in certain parts of the world and is associated with disease outbreaks.
El Niño has been associated with increases in the occurrence of actinic keratosis, tinea, pityriasis versicolor, miliaria, folliculitis, rosacea, dermatitis caused by Paederus irritans and Paederus sabaeus, and certain vector-borne and waterborne diseases (such as dengue fever, leishmaniasis, Chagas disease, Barmah Forest virus, and leptospirosis), and with decreases in the occurrence of dermatitis, scabies, psoriasis, and papular urticaria. La Niña has been associated with increases in the occurrence of varicella; hand, foot, and mouth disease; and Ross River virus (in certain areas), and decreases in viral warts and leishmaniasis.
Separately, global warming is expected to affect the start, duration, and intensity of the pollen season, and secondarily the rate of asthma exacerbations due to air pollution, respiratory infections, and/or cold air inhalation, with probable increases in eczema and other atopy-related conditions as well.
Vector-borne diseases
In the past year, the largest Ebola virus outbreak in West Africa has resulted in importation of the virus to other countries and secondary local transmission. Autochthonous transmission of Chikungunya virus has occurred in nonendemic areas, including Europe, the Caribbean, and the Americas. Zika virus has re-emerged in the Pacific with local transmission from imported cases. Climate change, deforestation, and changes in precipitation have been linked to variations in the geographical distribution of vectors of some infectious diseases (leishmaniasis, Lyme disease, and now Zika virus) by changing their spread. A warm and humid environment from global warming can also encourage the colonization of the skin by bacteria and fungi.
Finally, there is a wider, ecological dimension to consider. UV radiation impairs the molecular chemistry of photosynthesis both on land (terrestrial plants) and at sea (phytoplankton). This could affect world food production, at least marginally, and thus contribute to nutritional and health problems in food-insecure populations.
Another reason of many, but from a dermatologist’s point of view, to be mindful of taking care of the planet and our environment.
References
1. World Health Organization (WHO).
2. Photochem Photobiol Sci. 2002 May;1(5):324-6.
3. G Ital Dermatol Venereol. 2013 Feb;148(1):135-46.
4. Int J Dermatol. 2012 Jun;51(6):656-61.
5. Int J Dermatol. 2015 Dec;54(12):1343-51.
6. Curr Opin Infect Dis. 2015 Apr;28(2):139-50.
Dr. Wesley and Dr. Talakoub are co-contributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Wesley. Write to them at [email protected].
Global climate appears to be changing at an unprecedented rate. Climate change can be caused by many factors, including variations in solar radiation received by the earth, oceanic circulation, plate tectonics, as well as human-induced alterations of the natural world. Many human activities, such as the use of fossil fuel and the consequent accumulation of greenhouse gases in the atmosphere, land consumption, deforestation, industrial processes, as well as some agriculture practices, are contributing to global climate change. Many have reported on the current trend toward global warming (average surface temperature has augmented by 0.6°C over the past 100 years), decreased precipitation, atmospheric humidity changes, and the rise in global extreme climatic events. The magnitude and cause of these changes and their impact on human activity have become important matters of debate worldwide, representing climate change as one of the greatest challenges of the modern age.
Although many articles have been written based on observations and various predictive models of how climate change could affect social, economic, and health systems, only a few studies exist about the effects of this change on skin and skin disease. However, the skin is the most highly exposed organ to the environment; therefore, cutaneous conditions are inclined to respond to changes in climate.
Skin cancer
The World Health Organization predicts that the depletion of the ozone layer could lead to further increased rates of melanoma and nonmelanoma skin cancer. In humans, it has been speculated that a long-term rise of temperature by 2°C could increase the carcinogenic effectiveness of solar UV by 10%.
Strictly speaking, stratospheric ozone depletion is not part of “global climate change,” which occurs in the troposphere. There are, however, several recently described interactions between ozone depletion and greenhouse gas–induced warming. Stratospheric ozone absorbs much of the incoming solar ultraviolet radiation, especially the biologically more damaging, shorter-wavelength UVB wavelengths. We now know that various industrial halogenated chemicals such as the chlorofluorocarbons or CFCs (used in refrigeration, insulation, and spray-can propellants) and methyl bromide, while inert at ambient Earth-surface temperatures, react with ozone in the extremely cold polar stratosphere. This destruction of ozone occurs especially in late winter and early spring.
During the 1980s and 1990s at northern midlatitudes (such as Europe), the average year-round ozone concentration declined by around 4% per decade; over the southern regions of Australia, New Zealand, Argentina, and South Africa, the figure approximated 6%-7%. UV exposures at northern midlatitudes are likely to peak around 2020, with an estimated 10% increase in effective ultraviolet radiation relative to 1980s levels.
The modeling of future ozone levels and UV radiation (UVR) exposures has estimated that, in consequence, a ‘European’ population living at around 45 degrees North will experience, by 2050, an approximate 5% excess of total skin cancer incidence (assuming, conservatively, no change in age distribution). The equivalent estimation for the U.S. population is for a 10% increase in skin cancer incidence by around 2050.
In the mid-1980s, governments recognized the emerging hazard from ozone depletion. The Montreal Protocol of 1987 was adopted, and the phasing out of major ozone-destroying gases began. Some anticipate a slow but near-complete recovery of stratospheric ozone by the middle of the twenty-first century; the Environmental Protection Agency (EPA) estimates recovery by 2065 with strict adherence to protection protocols.
Increased exposure to UVR also leads to increased rates of lens opacification, cataracts, and whole-body immunosuppression. UVR-induced immunosuppression could influence patterns of infectious disease. It may also influence the occurrence and progression of various autoimmune diseases and, less certainly, vaccine efficacy.
Extreme weather events
The International Society of Dermatology Task Force on Climate Change reports that weather phenomena such as El Niño also result in changes to dermatologic conditions. The El Niño Southern Oscillation (ENSO) is a complex climate phenomenon occurring in the Pacific Ocean at intervals of 2-7 years. The term refers to fluctuations in ocean temperatures in the tropical eastern Pacific Ocean (El Niño, the warm phase of ENSO, and La Niña, the cool phase of ENSO) and in atmospheric pressure across the Pacific basin (Southern Oscillation). This weather pattern is attributed with causing climate change in certain parts of the world and is associated with disease outbreaks.
El Niño has been associated with increases in the occurrence of actinic keratosis, tinea, pityriasis versicolor, miliaria, folliculitis, rosacea, dermatitis caused by Paederus irritans and Paederus sabaeus, and certain vector-borne and waterborne diseases (such as dengue fever, leishmaniasis, Chagas disease, Barmah Forest virus, and leptospirosis), and with decreases in the occurrence of dermatitis, scabies, psoriasis, and papular urticaria. La Niña has been associated with increases in the occurrence of varicella; hand, foot, and mouth disease; and Ross River virus (in certain areas), and decreases in viral warts and leishmaniasis.
Separately, global warming is expected to affect the start, duration, and intensity of the pollen season, and secondarily the rate of asthma exacerbations due to air pollution, respiratory infections, and/or cold air inhalation, with probable increases in eczema and other atopy-related conditions as well.
Vector-borne diseases
In the past year, the largest Ebola virus outbreak in West Africa has resulted in importation of the virus to other countries and secondary local transmission. Autochthonous transmission of Chikungunya virus has occurred in nonendemic areas, including Europe, the Caribbean, and the Americas. Zika virus has re-emerged in the Pacific with local transmission from imported cases. Climate change, deforestation, and changes in precipitation have been linked to variations in the geographical distribution of vectors of some infectious diseases (leishmaniasis, Lyme disease, and now Zika virus) by changing their spread. A warm and humid environment from global warming can also encourage the colonization of the skin by bacteria and fungi.
Finally, there is a wider, ecological dimension to consider. UV radiation impairs the molecular chemistry of photosynthesis both on land (terrestrial plants) and at sea (phytoplankton). This could affect world food production, at least marginally, and thus contribute to nutritional and health problems in food-insecure populations.
Another reason of many, but from a dermatologist’s point of view, to be mindful of taking care of the planet and our environment.
References
1. World Health Organization (WHO).
2. Photochem Photobiol Sci. 2002 May;1(5):324-6.
3. G Ital Dermatol Venereol. 2013 Feb;148(1):135-46.
4. Int J Dermatol. 2012 Jun;51(6):656-61.
5. Int J Dermatol. 2015 Dec;54(12):1343-51.
6. Curr Opin Infect Dis. 2015 Apr;28(2):139-50.
Dr. Wesley and Dr. Talakoub are co-contributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Wesley. Write to them at [email protected].
Global climate appears to be changing at an unprecedented rate. Climate change can be caused by many factors, including variations in solar radiation received by the earth, oceanic circulation, plate tectonics, as well as human-induced alterations of the natural world. Many human activities, such as the use of fossil fuel and the consequent accumulation of greenhouse gases in the atmosphere, land consumption, deforestation, industrial processes, as well as some agriculture practices, are contributing to global climate change. Many have reported on the current trend toward global warming (average surface temperature has augmented by 0.6°C over the past 100 years), decreased precipitation, atmospheric humidity changes, and the rise in global extreme climatic events. The magnitude and cause of these changes and their impact on human activity have become important matters of debate worldwide, representing climate change as one of the greatest challenges of the modern age.
Although many articles have been written based on observations and various predictive models of how climate change could affect social, economic, and health systems, only a few studies exist about the effects of this change on skin and skin disease. However, the skin is the most highly exposed organ to the environment; therefore, cutaneous conditions are inclined to respond to changes in climate.
Skin cancer
The World Health Organization predicts that the depletion of the ozone layer could lead to further increased rates of melanoma and nonmelanoma skin cancer. In humans, it has been speculated that a long-term rise of temperature by 2°C could increase the carcinogenic effectiveness of solar UV by 10%.
Strictly speaking, stratospheric ozone depletion is not part of “global climate change,” which occurs in the troposphere. There are, however, several recently described interactions between ozone depletion and greenhouse gas–induced warming. Stratospheric ozone absorbs much of the incoming solar ultraviolet radiation, especially the biologically more damaging, shorter-wavelength UVB wavelengths. We now know that various industrial halogenated chemicals such as the chlorofluorocarbons or CFCs (used in refrigeration, insulation, and spray-can propellants) and methyl bromide, while inert at ambient Earth-surface temperatures, react with ozone in the extremely cold polar stratosphere. This destruction of ozone occurs especially in late winter and early spring.
During the 1980s and 1990s at northern midlatitudes (such as Europe), the average year-round ozone concentration declined by around 4% per decade; over the southern regions of Australia, New Zealand, Argentina, and South Africa, the figure approximated 6%-7%. UV exposures at northern midlatitudes are likely to peak around 2020, with an estimated 10% increase in effective ultraviolet radiation relative to 1980s levels.
The modeling of future ozone levels and UV radiation (UVR) exposures has estimated that, in consequence, a ‘European’ population living at around 45 degrees North will experience, by 2050, an approximate 5% excess of total skin cancer incidence (assuming, conservatively, no change in age distribution). The equivalent estimation for the U.S. population is for a 10% increase in skin cancer incidence by around 2050.
In the mid-1980s, governments recognized the emerging hazard from ozone depletion. The Montreal Protocol of 1987 was adopted, and the phasing out of major ozone-destroying gases began. Some anticipate a slow but near-complete recovery of stratospheric ozone by the middle of the twenty-first century; the Environmental Protection Agency (EPA) estimates recovery by 2065 with strict adherence to protection protocols.
Increased exposure to UVR also leads to increased rates of lens opacification, cataracts, and whole-body immunosuppression. UVR-induced immunosuppression could influence patterns of infectious disease. It may also influence the occurrence and progression of various autoimmune diseases and, less certainly, vaccine efficacy.
Extreme weather events
The International Society of Dermatology Task Force on Climate Change reports that weather phenomena such as El Niño also result in changes to dermatologic conditions. The El Niño Southern Oscillation (ENSO) is a complex climate phenomenon occurring in the Pacific Ocean at intervals of 2-7 years. The term refers to fluctuations in ocean temperatures in the tropical eastern Pacific Ocean (El Niño, the warm phase of ENSO, and La Niña, the cool phase of ENSO) and in atmospheric pressure across the Pacific basin (Southern Oscillation). This weather pattern is attributed with causing climate change in certain parts of the world and is associated with disease outbreaks.
El Niño has been associated with increases in the occurrence of actinic keratosis, tinea, pityriasis versicolor, miliaria, folliculitis, rosacea, dermatitis caused by Paederus irritans and Paederus sabaeus, and certain vector-borne and waterborne diseases (such as dengue fever, leishmaniasis, Chagas disease, Barmah Forest virus, and leptospirosis), and with decreases in the occurrence of dermatitis, scabies, psoriasis, and papular urticaria. La Niña has been associated with increases in the occurrence of varicella; hand, foot, and mouth disease; and Ross River virus (in certain areas), and decreases in viral warts and leishmaniasis.
Separately, global warming is expected to affect the start, duration, and intensity of the pollen season, and secondarily the rate of asthma exacerbations due to air pollution, respiratory infections, and/or cold air inhalation, with probable increases in eczema and other atopy-related conditions as well.
Vector-borne diseases
In the past year, the largest Ebola virus outbreak in West Africa has resulted in importation of the virus to other countries and secondary local transmission. Autochthonous transmission of Chikungunya virus has occurred in nonendemic areas, including Europe, the Caribbean, and the Americas. Zika virus has re-emerged in the Pacific with local transmission from imported cases. Climate change, deforestation, and changes in precipitation have been linked to variations in the geographical distribution of vectors of some infectious diseases (leishmaniasis, Lyme disease, and now Zika virus) by changing their spread. A warm and humid environment from global warming can also encourage the colonization of the skin by bacteria and fungi.
Finally, there is a wider, ecological dimension to consider. UV radiation impairs the molecular chemistry of photosynthesis both on land (terrestrial plants) and at sea (phytoplankton). This could affect world food production, at least marginally, and thus contribute to nutritional and health problems in food-insecure populations.
Another reason of many, but from a dermatologist’s point of view, to be mindful of taking care of the planet and our environment.
References
1. World Health Organization (WHO).
2. Photochem Photobiol Sci. 2002 May;1(5):324-6.
3. G Ital Dermatol Venereol. 2013 Feb;148(1):135-46.
4. Int J Dermatol. 2012 Jun;51(6):656-61.
5. Int J Dermatol. 2015 Dec;54(12):1343-51.
6. Curr Opin Infect Dis. 2015 Apr;28(2):139-50.
Dr. Wesley and Dr. Talakoub are co-contributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Wesley. Write to them at [email protected].
Mohs surgeons, dermatopathologists nearly always agree on frozen section analysis
ORLANDO – Mohs fellowship–trained surgeons are highly skilled at reading frozen section slides obtained during surgery, agreeing with the dermatopathologist’s assessment more than 99% of the time, according to a review of more than 4,000 cases.
Among 4,145 slides in five datasets, only 28 were discordant. The concordance of surgeons’ and dermatopathologists’ assessments was quite consistent, ranging from 95%-of 99.7%.
“There is absolutely no detriment to our interpretation of these frozen section slides,” Dr. James Highsmith said at the annual meeting of the American College of Mohs Surgery. “These numbers give credence as to why we have these good cure rates. We can say with 99% accuracy that yes, we are removing these tumors from our patients.”
Dr. Highsmith, a Mohs surgeon in Birmingham, Ala., reviewed 10 years of his own data (1,720 cases), as well as four published cohorts comprising another 2,425 cases. All of the studies examined the concordance of frozen section assessments between fellowship-trained Mohs surgeons and the dermatopathologists working with them. All of the series reported concordance, discordance, sensitivity, and specificity.
His own database is the largest one yet compiled, spanning the longest period of time. Of 1,720 cases, there were eight discrepancies: one in which the surgeon identified tumor that was not supported by dermatopathology, and seven in which the surgeon missed tumor that dermatopathology confirmed was present. The concordance rate was 99.5%. This worked out to a 99.4% sensitivity and 99.8% specificity, Dr. Highsmith said.
Results were similar with the other four studies.
The largest was a 2009 retrospective review of 1,156 slides collected over 10 years; they were part of a preexisting randomized, blinded study. Of these, 32 slides (2.8%) were discordant between the Mohs surgeon and the dermatopathologist, for a 99.7% concordance rate (J Am Acad Dermatol. 2009 Jan;[1]: 94-8).
The second largest cohort was published in 1989. It comprised 1,000 slides that were evaluated by the Mohs surgeon and a general pathologist; disputed slides were read by two other Mohs surgeons, two pathologists, and a dermatopathologist. The overall concordance rate was 98.9% (J Am Acad Dermatol. 1989;20[4]:670-4).
In 2013, a Mohs surgeon published a set of 170 slides that had been randomly selected from his cases as part of a quality assurance audit. There was one discordant interpretation, for an overall concordance rate of 99.4% (Dermatol Surg. 2013;39[11]:1648-52).
The smallest study, published in 2011, reviewed 99 slides assessed by a single Mohs surgeon. The concordance rate with dermatopathology was 95%.
An overall analysis of these four cohorts determined a sensitivity of 99.4%, specificity of 99.2% and negative predictive value of 99.2%, Dr. Highsmith said.
“When a fellowship-trained Mohs surgeon looks at a slide and says there’s no more tumor present, then there’s a greater-than-99% chance that is accurate,” he said.
Dr. Highsmith had no financial disclosures.
ORLANDO – Mohs fellowship–trained surgeons are highly skilled at reading frozen section slides obtained during surgery, agreeing with the dermatopathologist’s assessment more than 99% of the time, according to a review of more than 4,000 cases.
Among 4,145 slides in five datasets, only 28 were discordant. The concordance of surgeons’ and dermatopathologists’ assessments was quite consistent, ranging from 95%-of 99.7%.
“There is absolutely no detriment to our interpretation of these frozen section slides,” Dr. James Highsmith said at the annual meeting of the American College of Mohs Surgery. “These numbers give credence as to why we have these good cure rates. We can say with 99% accuracy that yes, we are removing these tumors from our patients.”
Dr. Highsmith, a Mohs surgeon in Birmingham, Ala., reviewed 10 years of his own data (1,720 cases), as well as four published cohorts comprising another 2,425 cases. All of the studies examined the concordance of frozen section assessments between fellowship-trained Mohs surgeons and the dermatopathologists working with them. All of the series reported concordance, discordance, sensitivity, and specificity.
His own database is the largest one yet compiled, spanning the longest period of time. Of 1,720 cases, there were eight discrepancies: one in which the surgeon identified tumor that was not supported by dermatopathology, and seven in which the surgeon missed tumor that dermatopathology confirmed was present. The concordance rate was 99.5%. This worked out to a 99.4% sensitivity and 99.8% specificity, Dr. Highsmith said.
Results were similar with the other four studies.
The largest was a 2009 retrospective review of 1,156 slides collected over 10 years; they were part of a preexisting randomized, blinded study. Of these, 32 slides (2.8%) were discordant between the Mohs surgeon and the dermatopathologist, for a 99.7% concordance rate (J Am Acad Dermatol. 2009 Jan;[1]: 94-8).
The second largest cohort was published in 1989. It comprised 1,000 slides that were evaluated by the Mohs surgeon and a general pathologist; disputed slides were read by two other Mohs surgeons, two pathologists, and a dermatopathologist. The overall concordance rate was 98.9% (J Am Acad Dermatol. 1989;20[4]:670-4).
In 2013, a Mohs surgeon published a set of 170 slides that had been randomly selected from his cases as part of a quality assurance audit. There was one discordant interpretation, for an overall concordance rate of 99.4% (Dermatol Surg. 2013;39[11]:1648-52).
The smallest study, published in 2011, reviewed 99 slides assessed by a single Mohs surgeon. The concordance rate with dermatopathology was 95%.
An overall analysis of these four cohorts determined a sensitivity of 99.4%, specificity of 99.2% and negative predictive value of 99.2%, Dr. Highsmith said.
“When a fellowship-trained Mohs surgeon looks at a slide and says there’s no more tumor present, then there’s a greater-than-99% chance that is accurate,” he said.
Dr. Highsmith had no financial disclosures.
ORLANDO – Mohs fellowship–trained surgeons are highly skilled at reading frozen section slides obtained during surgery, agreeing with the dermatopathologist’s assessment more than 99% of the time, according to a review of more than 4,000 cases.
Among 4,145 slides in five datasets, only 28 were discordant. The concordance of surgeons’ and dermatopathologists’ assessments was quite consistent, ranging from 95%-of 99.7%.
“There is absolutely no detriment to our interpretation of these frozen section slides,” Dr. James Highsmith said at the annual meeting of the American College of Mohs Surgery. “These numbers give credence as to why we have these good cure rates. We can say with 99% accuracy that yes, we are removing these tumors from our patients.”
Dr. Highsmith, a Mohs surgeon in Birmingham, Ala., reviewed 10 years of his own data (1,720 cases), as well as four published cohorts comprising another 2,425 cases. All of the studies examined the concordance of frozen section assessments between fellowship-trained Mohs surgeons and the dermatopathologists working with them. All of the series reported concordance, discordance, sensitivity, and specificity.
His own database is the largest one yet compiled, spanning the longest period of time. Of 1,720 cases, there were eight discrepancies: one in which the surgeon identified tumor that was not supported by dermatopathology, and seven in which the surgeon missed tumor that dermatopathology confirmed was present. The concordance rate was 99.5%. This worked out to a 99.4% sensitivity and 99.8% specificity, Dr. Highsmith said.
Results were similar with the other four studies.
The largest was a 2009 retrospective review of 1,156 slides collected over 10 years; they were part of a preexisting randomized, blinded study. Of these, 32 slides (2.8%) were discordant between the Mohs surgeon and the dermatopathologist, for a 99.7% concordance rate (J Am Acad Dermatol. 2009 Jan;[1]: 94-8).
The second largest cohort was published in 1989. It comprised 1,000 slides that were evaluated by the Mohs surgeon and a general pathologist; disputed slides were read by two other Mohs surgeons, two pathologists, and a dermatopathologist. The overall concordance rate was 98.9% (J Am Acad Dermatol. 1989;20[4]:670-4).
In 2013, a Mohs surgeon published a set of 170 slides that had been randomly selected from his cases as part of a quality assurance audit. There was one discordant interpretation, for an overall concordance rate of 99.4% (Dermatol Surg. 2013;39[11]:1648-52).
The smallest study, published in 2011, reviewed 99 slides assessed by a single Mohs surgeon. The concordance rate with dermatopathology was 95%.
An overall analysis of these four cohorts determined a sensitivity of 99.4%, specificity of 99.2% and negative predictive value of 99.2%, Dr. Highsmith said.
“When a fellowship-trained Mohs surgeon looks at a slide and says there’s no more tumor present, then there’s a greater-than-99% chance that is accurate,” he said.
Dr. Highsmith had no financial disclosures.
AT THE ACMS ANNUAL MEETING
Key clinical point: Fellowship-trained Mohs surgeons and dermatopathologists almost always agree on slide interpretation.
Major finding: The concordance rate between surgeons and dermatopathologists was over 99% in a pooled review of studies.
Data source: A review of 4,145 frozen section slides analyzed for concordance rates between Mohs surgeons and dermatopathologists.
Disclosures: Dr. James Highsmith had no financial disclosures.
En bloc excision may be viable amputation alternative for nail melanoma
ORLANDO – En bloc excision can cure melanoma in situ of the nail apparatus without amputation, while also preserving function and quality of life, according to the results of a retrospective study of patients treated with this approach.
At 3 years’ follow-up, there were no melanoma recurrences in any of 29 patients treated with the digit-sparing excision, Dr. Thomas Knackstedt said at the annual meeting of the American College of Mohs Surgery. Follow-up telephone interviews found that patients retained full function of the affected finger, and reported virtually no quality of life impairment.
“Because treatment without disarticulation or amputation can provide these excellent long-term cure rates in nail apparatus melanoma in situ, it should be the first-line treatment whenever possible,” said Dr. Knackstedt, a Mohs surgeon in East Greenwich, R.I.
Although similar excellent outcomes have been reported with wide local excision, Dr. Knackstedt said it remains a controversial treatment for nail apparatus melanoma in situ (NAMis).
He reviewed his institution’s experience with treating 29 cases of NAMis with en bloc excision: removal of nail folds, matrix, bed, hyponychium, and margins. All of the surgeries were performed between 2005 and 2015. The current mean follow-up time is 35.5 months for clinic visits and 39 months for telephone contact. The longest follow-up is 103 months. There have been no melanoma recurrences.
The patients were a mean of 39 years old. Melanoma appeared most often on the thumb (seven patients) and hallux (six). The average duration of the lesion was almost 5 years, although that varied from 6 months to 13 years.
Twenty-four patients (83%) presented with longitudinal melanonychia, with an average diameter of 4.1 mm. Four patients (14%) had complete melanonychia, and one patient had erythronychia.
Most of the cases were repaired with a full-thickness skin graft. One was left to heal by second intention, and one case was repaired by a dorsal metacarpal artery flap.
There were two wound infections, one case of delayed graft necrosis, seven nail spicules, and two cysts. There were no cases of tendon injury. One patient complained about temperature sensitivity in the affected finger. There were no cases of disease recurrence, although one patient with a nail spicule did show disease persistence. The spicule was excised, and that patient remains disease free 5 years later, Dr. Knackstedt said.
The telephone survey collected data from 23 patients (79%). Patients were unanimous in reporting that they were “very satisfied” with their treatment. In an 11-question quality of life survey, 78% said that the surgery affected their quality of life “only a little” or “not at all.” On a 10-point Likert scale with 10 being most affected by the surgery, the mean rating was 1.3.
“In light of these results, and when we consider that even a distal joint amputation can reduce the functionality of a finger by 40%-80%, we conclude that en bloc excision must be considered as a viable alternative to amputation for NAMis,” Dr. Knackstedt said.
He had no relevant financial disclosures.
ORLANDO – En bloc excision can cure melanoma in situ of the nail apparatus without amputation, while also preserving function and quality of life, according to the results of a retrospective study of patients treated with this approach.
At 3 years’ follow-up, there were no melanoma recurrences in any of 29 patients treated with the digit-sparing excision, Dr. Thomas Knackstedt said at the annual meeting of the American College of Mohs Surgery. Follow-up telephone interviews found that patients retained full function of the affected finger, and reported virtually no quality of life impairment.
“Because treatment without disarticulation or amputation can provide these excellent long-term cure rates in nail apparatus melanoma in situ, it should be the first-line treatment whenever possible,” said Dr. Knackstedt, a Mohs surgeon in East Greenwich, R.I.
Although similar excellent outcomes have been reported with wide local excision, Dr. Knackstedt said it remains a controversial treatment for nail apparatus melanoma in situ (NAMis).
He reviewed his institution’s experience with treating 29 cases of NAMis with en bloc excision: removal of nail folds, matrix, bed, hyponychium, and margins. All of the surgeries were performed between 2005 and 2015. The current mean follow-up time is 35.5 months for clinic visits and 39 months for telephone contact. The longest follow-up is 103 months. There have been no melanoma recurrences.
The patients were a mean of 39 years old. Melanoma appeared most often on the thumb (seven patients) and hallux (six). The average duration of the lesion was almost 5 years, although that varied from 6 months to 13 years.
Twenty-four patients (83%) presented with longitudinal melanonychia, with an average diameter of 4.1 mm. Four patients (14%) had complete melanonychia, and one patient had erythronychia.
Most of the cases were repaired with a full-thickness skin graft. One was left to heal by second intention, and one case was repaired by a dorsal metacarpal artery flap.
There were two wound infections, one case of delayed graft necrosis, seven nail spicules, and two cysts. There were no cases of tendon injury. One patient complained about temperature sensitivity in the affected finger. There were no cases of disease recurrence, although one patient with a nail spicule did show disease persistence. The spicule was excised, and that patient remains disease free 5 years later, Dr. Knackstedt said.
The telephone survey collected data from 23 patients (79%). Patients were unanimous in reporting that they were “very satisfied” with their treatment. In an 11-question quality of life survey, 78% said that the surgery affected their quality of life “only a little” or “not at all.” On a 10-point Likert scale with 10 being most affected by the surgery, the mean rating was 1.3.
“In light of these results, and when we consider that even a distal joint amputation can reduce the functionality of a finger by 40%-80%, we conclude that en bloc excision must be considered as a viable alternative to amputation for NAMis,” Dr. Knackstedt said.
He had no relevant financial disclosures.
ORLANDO – En bloc excision can cure melanoma in situ of the nail apparatus without amputation, while also preserving function and quality of life, according to the results of a retrospective study of patients treated with this approach.
At 3 years’ follow-up, there were no melanoma recurrences in any of 29 patients treated with the digit-sparing excision, Dr. Thomas Knackstedt said at the annual meeting of the American College of Mohs Surgery. Follow-up telephone interviews found that patients retained full function of the affected finger, and reported virtually no quality of life impairment.
“Because treatment without disarticulation or amputation can provide these excellent long-term cure rates in nail apparatus melanoma in situ, it should be the first-line treatment whenever possible,” said Dr. Knackstedt, a Mohs surgeon in East Greenwich, R.I.
Although similar excellent outcomes have been reported with wide local excision, Dr. Knackstedt said it remains a controversial treatment for nail apparatus melanoma in situ (NAMis).
He reviewed his institution’s experience with treating 29 cases of NAMis with en bloc excision: removal of nail folds, matrix, bed, hyponychium, and margins. All of the surgeries were performed between 2005 and 2015. The current mean follow-up time is 35.5 months for clinic visits and 39 months for telephone contact. The longest follow-up is 103 months. There have been no melanoma recurrences.
The patients were a mean of 39 years old. Melanoma appeared most often on the thumb (seven patients) and hallux (six). The average duration of the lesion was almost 5 years, although that varied from 6 months to 13 years.
Twenty-four patients (83%) presented with longitudinal melanonychia, with an average diameter of 4.1 mm. Four patients (14%) had complete melanonychia, and one patient had erythronychia.
Most of the cases were repaired with a full-thickness skin graft. One was left to heal by second intention, and one case was repaired by a dorsal metacarpal artery flap.
There were two wound infections, one case of delayed graft necrosis, seven nail spicules, and two cysts. There were no cases of tendon injury. One patient complained about temperature sensitivity in the affected finger. There were no cases of disease recurrence, although one patient with a nail spicule did show disease persistence. The spicule was excised, and that patient remains disease free 5 years later, Dr. Knackstedt said.
The telephone survey collected data from 23 patients (79%). Patients were unanimous in reporting that they were “very satisfied” with their treatment. In an 11-question quality of life survey, 78% said that the surgery affected their quality of life “only a little” or “not at all.” On a 10-point Likert scale with 10 being most affected by the surgery, the mean rating was 1.3.
“In light of these results, and when we consider that even a distal joint amputation can reduce the functionality of a finger by 40%-80%, we conclude that en bloc excision must be considered as a viable alternative to amputation for NAMis,” Dr. Knackstedt said.
He had no relevant financial disclosures.
AT THE ACMS ANNUAL MEETING
Key clinical point: En bloc removal of the nail apparatus effected good clinical and quality of life outcomes in nail melanoma.
Major finding: There have been no recurrences of melanoma over a mean follow-up of 39 months.
Data source: A review evaluated 29 cases of nail apparatus melanoma in situ treated with en bloc excision at one institution.
Disclosures: Dr. Knackstedt had no relevant financial disclosures.